User login
Iron Overload in Non-HFE Liver Disease: Not all Iron is Ready to Strike
Pathological iron overload with end-organ damage in hemochromatosis occurs in individuals who are homozygous for the major mutation C282Y. Phenotypic hemochromatosis occurs much less frequently in compound heterozygotes with one C282Y mutation and one H63D mutation. Iron overload can be confirmed by magnetic resonance imaging, which shows a loss of signal intensity in affected tissues and avoids the need for liver biopsy.
The serum ferritin level, an acute phase reactant, may be elevated for reasons other than iron overload, including infection and malignancy; in such cases, the iron saturation is usually normal. In patients with liver disease, iron overload is not restricted to patients with genetic hemochromatosis. In nonalcoholic fatty liver disease (NAFLD), up to one-third of patients have an elevated iron saturation (> 45%) and an elevated serum ferritin level. Iron accumulation in NAFLD can occur in hepatocytes, the reticuloendothelial system, or both. Deposition of iron in the reticuloendothelial system has been implicated in more severe liver disease (steatohepatitis and fibrosis) in NAFLD. Hepatic iron accumulation is also frequent in alcohol-associated liver disease. In chronic hepatitis B and C, accumulation of hepatic iron is also recognized.
Dr. Martin is chief of the division of digestive health and liver diseases at the Miller School of Medicine, University of Miami, where he is the Mandel Chair of Gastroenterology. Dr. Friedman is the Anton R. Fried, MD, Chair of the department of medicine at Newton-Wellesley Hospital in Newton, Massachusetts, and assistant chief of medicine at Massachusetts General Hospital, and a professor of medicine at Harvard Medical School and Tufts University School of Medicine, all in Boston. The authors disclosed no conflicts. Previously published in Gastro Hep Advances. 2023 Oct 12. doi: 10.1016/j.gastha.2023.10.004.
Pathological iron overload with end-organ damage in hemochromatosis occurs in individuals who are homozygous for the major mutation C282Y. Phenotypic hemochromatosis occurs much less frequently in compound heterozygotes with one C282Y mutation and one H63D mutation. Iron overload can be confirmed by magnetic resonance imaging, which shows a loss of signal intensity in affected tissues and avoids the need for liver biopsy.
The serum ferritin level, an acute phase reactant, may be elevated for reasons other than iron overload, including infection and malignancy; in such cases, the iron saturation is usually normal. In patients with liver disease, iron overload is not restricted to patients with genetic hemochromatosis. In nonalcoholic fatty liver disease (NAFLD), up to one-third of patients have an elevated iron saturation (> 45%) and an elevated serum ferritin level. Iron accumulation in NAFLD can occur in hepatocytes, the reticuloendothelial system, or both. Deposition of iron in the reticuloendothelial system has been implicated in more severe liver disease (steatohepatitis and fibrosis) in NAFLD. Hepatic iron accumulation is also frequent in alcohol-associated liver disease. In chronic hepatitis B and C, accumulation of hepatic iron is also recognized.
Dr. Martin is chief of the division of digestive health and liver diseases at the Miller School of Medicine, University of Miami, where he is the Mandel Chair of Gastroenterology. Dr. Friedman is the Anton R. Fried, MD, Chair of the department of medicine at Newton-Wellesley Hospital in Newton, Massachusetts, and assistant chief of medicine at Massachusetts General Hospital, and a professor of medicine at Harvard Medical School and Tufts University School of Medicine, all in Boston. The authors disclosed no conflicts. Previously published in Gastro Hep Advances. 2023 Oct 12. doi: 10.1016/j.gastha.2023.10.004.
Pathological iron overload with end-organ damage in hemochromatosis occurs in individuals who are homozygous for the major mutation C282Y. Phenotypic hemochromatosis occurs much less frequently in compound heterozygotes with one C282Y mutation and one H63D mutation. Iron overload can be confirmed by magnetic resonance imaging, which shows a loss of signal intensity in affected tissues and avoids the need for liver biopsy.
The serum ferritin level, an acute phase reactant, may be elevated for reasons other than iron overload, including infection and malignancy; in such cases, the iron saturation is usually normal. In patients with liver disease, iron overload is not restricted to patients with genetic hemochromatosis. In nonalcoholic fatty liver disease (NAFLD), up to one-third of patients have an elevated iron saturation (> 45%) and an elevated serum ferritin level. Iron accumulation in NAFLD can occur in hepatocytes, the reticuloendothelial system, or both. Deposition of iron in the reticuloendothelial system has been implicated in more severe liver disease (steatohepatitis and fibrosis) in NAFLD. Hepatic iron accumulation is also frequent in alcohol-associated liver disease. In chronic hepatitis B and C, accumulation of hepatic iron is also recognized.
Dr. Martin is chief of the division of digestive health and liver diseases at the Miller School of Medicine, University of Miami, where he is the Mandel Chair of Gastroenterology. Dr. Friedman is the Anton R. Fried, MD, Chair of the department of medicine at Newton-Wellesley Hospital in Newton, Massachusetts, and assistant chief of medicine at Massachusetts General Hospital, and a professor of medicine at Harvard Medical School and Tufts University School of Medicine, all in Boston. The authors disclosed no conflicts. Previously published in Gastro Hep Advances. 2023 Oct 12. doi: 10.1016/j.gastha.2023.10.004.
Guidelines Aren’t For Everybody
An 88-year-old man comes for clinic follow up. He has a medical history of type 2 diabetes, hypertension, heart failure with reduced ejection fraction, and chronic kidney disease. He recently had laboratory tests done: BUN, 32 mg/dL; creatinine, 2.3 mg/dL; potassium, 4.5 mmol/L; bicarbonate, 22 Eq/L; and A1c, 8.2%.
He checks his blood glucose daily (alternating between fasting blood glucose and before dinner) and his fasting blood glucose levels are around 130 mg/dL. His highest glucose reading was 240 mg/dL. He does not have polyuria or visual changes. Current medications: atorvastatin, irbesartan, empagliflozin, and amlodipine. On physical exam his blood pressure is 130/70 mm Hg, pulse is 80, and his BMI 20.
What medication adjustments would you recommend?
A. Begin insulin glargine at bedtime
B. Begin mealtime insulin aspart
C. Begin semaglutide
D. Begin metformin
E. No changes
I think the correct approach here would be no changes. Most physicians know guideline recommendations for A1c of less than 7% are used for patients with diabetes with few comorbid conditions, normal cognition, and functional status. Many of our elderly patients do not meet these criteria and the goal of intense medical treatment of diabetes is different in those patients. The American Diabetes Association has issued a thoughtful paper on treatment of diabetes in elderly people, stressing that patients should have very individualized goals, and that there is no one-size-fits all A1c goal.1
In this patient I would avoid adding insulin, given hypoglycemia risk. A GLP-1 agonist might appear attractive given his multiple cardiovascular risk factors, but his low BMI is a major concern for frailty that may well be worsened with reduced nutrient intake. Diabetes is the chronic condition that probably has the most guidance for management in elderly patients.
I recently saw a 92-year-old man with heart failure with reduced ejection fraction and atrial fibrillation who had been losing weight and becoming weaker. He had suffered several falls in the previous 2 weeks. His medication list included amiodarone, apixaban, sacubitril/valsartan, carvedilol, empagliflozin, spironolactone, and furosemide. He was extremely frail and had stopped eating. He was receiving all guideline-directed therapies, yet he was miserable and dying. Falls in this population are potentially as fatal as decompensated heart disease.
I stopped his amiodarone, furosemide, and spironolactone, and reduced his doses of sacubitril/valsartan and carvedilol. His appetite returned and his will to live returned. Heart failure guidelines do not include robust studies of very elderly patients because few studies exist in this population. Frailty assessment is crucial in decision making in your elderly patients.2,3 and frequent check-ins to make sure that they are not suffering from the effects of polypharmacy are crucial. Our goal in our very elderly patients is quality life-years. Polypharmacy has the potential to decrease the quality of life, as well as potentially shorten life.
The very elderly are at risk of the negative consequences of polypharmacy, especially if they have several diseases like diabetes, congestive heart failure, and hypertension that may require multiple medications. Gutierrez-Valencia and colleagues performed a systematic review of 25 articles on frailty and polypharmacy.4 Their findings demonstrated a significant association between an increased number of medications and frailty. They postulated that polypharmacy could actually be a contributor to frailty. There just isn’t enough evidence for the benefit of guidelines in the very aged and the risks of polypharmacy are real. We should use the lowest possible doses of medications in this population, frequently reassess goals, and monitor closely for side effects.
Pearl: Always consider the risks of polypharmacy when considering therapies for your elderly patients.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and he serves as third-year medical student clerkship director at the University of Washington. Contact Dr. Paauw at [email protected].
References
1. Older Adults: Standards of Medical Care in Diabetes — 2021. Diabetes Care 2021;44(Suppl 1):S168–S179.
2. Gaur A et al. Cardiogeriatrics: The current state of the art. Heart. 2024 Jan 11:heartjnl-2022-322117.
3. Denfeld QE et al. Assessing and managing frailty in advanced heart failure: An International Society for Heart and Lung Transplantation consensus statement. J Heart Lung Transplant. 2023 Nov 29:S1053-2498(23)02028-4.
4. Gutiérrez-Valencia M et al. The relationship between frailty and polypharmacy in older people: A systematic review. Br J Clin Pharmacol. 2018 Jul;84(7):1432-44.
An 88-year-old man comes for clinic follow up. He has a medical history of type 2 diabetes, hypertension, heart failure with reduced ejection fraction, and chronic kidney disease. He recently had laboratory tests done: BUN, 32 mg/dL; creatinine, 2.3 mg/dL; potassium, 4.5 mmol/L; bicarbonate, 22 Eq/L; and A1c, 8.2%.
He checks his blood glucose daily (alternating between fasting blood glucose and before dinner) and his fasting blood glucose levels are around 130 mg/dL. His highest glucose reading was 240 mg/dL. He does not have polyuria or visual changes. Current medications: atorvastatin, irbesartan, empagliflozin, and amlodipine. On physical exam his blood pressure is 130/70 mm Hg, pulse is 80, and his BMI 20.
What medication adjustments would you recommend?
A. Begin insulin glargine at bedtime
B. Begin mealtime insulin aspart
C. Begin semaglutide
D. Begin metformin
E. No changes
I think the correct approach here would be no changes. Most physicians know guideline recommendations for A1c of less than 7% are used for patients with diabetes with few comorbid conditions, normal cognition, and functional status. Many of our elderly patients do not meet these criteria and the goal of intense medical treatment of diabetes is different in those patients. The American Diabetes Association has issued a thoughtful paper on treatment of diabetes in elderly people, stressing that patients should have very individualized goals, and that there is no one-size-fits all A1c goal.1
In this patient I would avoid adding insulin, given hypoglycemia risk. A GLP-1 agonist might appear attractive given his multiple cardiovascular risk factors, but his low BMI is a major concern for frailty that may well be worsened with reduced nutrient intake. Diabetes is the chronic condition that probably has the most guidance for management in elderly patients.
I recently saw a 92-year-old man with heart failure with reduced ejection fraction and atrial fibrillation who had been losing weight and becoming weaker. He had suffered several falls in the previous 2 weeks. His medication list included amiodarone, apixaban, sacubitril/valsartan, carvedilol, empagliflozin, spironolactone, and furosemide. He was extremely frail and had stopped eating. He was receiving all guideline-directed therapies, yet he was miserable and dying. Falls in this population are potentially as fatal as decompensated heart disease.
I stopped his amiodarone, furosemide, and spironolactone, and reduced his doses of sacubitril/valsartan and carvedilol. His appetite returned and his will to live returned. Heart failure guidelines do not include robust studies of very elderly patients because few studies exist in this population. Frailty assessment is crucial in decision making in your elderly patients.2,3 and frequent check-ins to make sure that they are not suffering from the effects of polypharmacy are crucial. Our goal in our very elderly patients is quality life-years. Polypharmacy has the potential to decrease the quality of life, as well as potentially shorten life.
The very elderly are at risk of the negative consequences of polypharmacy, especially if they have several diseases like diabetes, congestive heart failure, and hypertension that may require multiple medications. Gutierrez-Valencia and colleagues performed a systematic review of 25 articles on frailty and polypharmacy.4 Their findings demonstrated a significant association between an increased number of medications and frailty. They postulated that polypharmacy could actually be a contributor to frailty. There just isn’t enough evidence for the benefit of guidelines in the very aged and the risks of polypharmacy are real. We should use the lowest possible doses of medications in this population, frequently reassess goals, and monitor closely for side effects.
Pearl: Always consider the risks of polypharmacy when considering therapies for your elderly patients.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and he serves as third-year medical student clerkship director at the University of Washington. Contact Dr. Paauw at [email protected].
References
1. Older Adults: Standards of Medical Care in Diabetes — 2021. Diabetes Care 2021;44(Suppl 1):S168–S179.
2. Gaur A et al. Cardiogeriatrics: The current state of the art. Heart. 2024 Jan 11:heartjnl-2022-322117.
3. Denfeld QE et al. Assessing and managing frailty in advanced heart failure: An International Society for Heart and Lung Transplantation consensus statement. J Heart Lung Transplant. 2023 Nov 29:S1053-2498(23)02028-4.
4. Gutiérrez-Valencia M et al. The relationship between frailty and polypharmacy in older people: A systematic review. Br J Clin Pharmacol. 2018 Jul;84(7):1432-44.
An 88-year-old man comes for clinic follow up. He has a medical history of type 2 diabetes, hypertension, heart failure with reduced ejection fraction, and chronic kidney disease. He recently had laboratory tests done: BUN, 32 mg/dL; creatinine, 2.3 mg/dL; potassium, 4.5 mmol/L; bicarbonate, 22 Eq/L; and A1c, 8.2%.
He checks his blood glucose daily (alternating between fasting blood glucose and before dinner) and his fasting blood glucose levels are around 130 mg/dL. His highest glucose reading was 240 mg/dL. He does not have polyuria or visual changes. Current medications: atorvastatin, irbesartan, empagliflozin, and amlodipine. On physical exam his blood pressure is 130/70 mm Hg, pulse is 80, and his BMI 20.
What medication adjustments would you recommend?
A. Begin insulin glargine at bedtime
B. Begin mealtime insulin aspart
C. Begin semaglutide
D. Begin metformin
E. No changes
I think the correct approach here would be no changes. Most physicians know guideline recommendations for A1c of less than 7% are used for patients with diabetes with few comorbid conditions, normal cognition, and functional status. Many of our elderly patients do not meet these criteria and the goal of intense medical treatment of diabetes is different in those patients. The American Diabetes Association has issued a thoughtful paper on treatment of diabetes in elderly people, stressing that patients should have very individualized goals, and that there is no one-size-fits all A1c goal.1
In this patient I would avoid adding insulin, given hypoglycemia risk. A GLP-1 agonist might appear attractive given his multiple cardiovascular risk factors, but his low BMI is a major concern for frailty that may well be worsened with reduced nutrient intake. Diabetes is the chronic condition that probably has the most guidance for management in elderly patients.
I recently saw a 92-year-old man with heart failure with reduced ejection fraction and atrial fibrillation who had been losing weight and becoming weaker. He had suffered several falls in the previous 2 weeks. His medication list included amiodarone, apixaban, sacubitril/valsartan, carvedilol, empagliflozin, spironolactone, and furosemide. He was extremely frail and had stopped eating. He was receiving all guideline-directed therapies, yet he was miserable and dying. Falls in this population are potentially as fatal as decompensated heart disease.
I stopped his amiodarone, furosemide, and spironolactone, and reduced his doses of sacubitril/valsartan and carvedilol. His appetite returned and his will to live returned. Heart failure guidelines do not include robust studies of very elderly patients because few studies exist in this population. Frailty assessment is crucial in decision making in your elderly patients.2,3 and frequent check-ins to make sure that they are not suffering from the effects of polypharmacy are crucial. Our goal in our very elderly patients is quality life-years. Polypharmacy has the potential to decrease the quality of life, as well as potentially shorten life.
The very elderly are at risk of the negative consequences of polypharmacy, especially if they have several diseases like diabetes, congestive heart failure, and hypertension that may require multiple medications. Gutierrez-Valencia and colleagues performed a systematic review of 25 articles on frailty and polypharmacy.4 Their findings demonstrated a significant association between an increased number of medications and frailty. They postulated that polypharmacy could actually be a contributor to frailty. There just isn’t enough evidence for the benefit of guidelines in the very aged and the risks of polypharmacy are real. We should use the lowest possible doses of medications in this population, frequently reassess goals, and monitor closely for side effects.
Pearl: Always consider the risks of polypharmacy when considering therapies for your elderly patients.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and he serves as third-year medical student clerkship director at the University of Washington. Contact Dr. Paauw at [email protected].
References
1. Older Adults: Standards of Medical Care in Diabetes — 2021. Diabetes Care 2021;44(Suppl 1):S168–S179.
2. Gaur A et al. Cardiogeriatrics: The current state of the art. Heart. 2024 Jan 11:heartjnl-2022-322117.
3. Denfeld QE et al. Assessing and managing frailty in advanced heart failure: An International Society for Heart and Lung Transplantation consensus statement. J Heart Lung Transplant. 2023 Nov 29:S1053-2498(23)02028-4.
4. Gutiérrez-Valencia M et al. The relationship between frailty and polypharmacy in older people: A systematic review. Br J Clin Pharmacol. 2018 Jul;84(7):1432-44.
New Injectable Weight Loss Drugs Pose Ethical Issues, Says Ethicist
This transcript has been edited for clarity.
There’s never been anything like the revolution in the treatment of obesity that we are now living through. Historically, there’s always been calorie counting and diets. Now, after a burst of interest in gastric bypass surgery, we have the amazing world of injectables. We all have heard about Ozempic, Mounjaro, and Wegovy.
These are being used by millions of Americans at this point, some on prescription for conditions like diabetes and some to bring about weight loss in prediabetes, or in some instances — as is often seen on American television — weight control or weight loss by people who just want to look better. Celebrities getting behind these injectables has really powered an explosion of use.
There still are ethical issues out there for practitioners. For one thing, there are some forms of semaglutide, a key ingredient in some of these injectables, that are made by compounding pharmacies. They’re not the name-brand prescription injectables made by large companies. They’re brewed up, if you will, by a specialty pharmacy trying to mimic the ingredient.
What we’ve seen in recent weeks is an explosion of overdoses. When a person uses one of these compounding pharmacies, usually in association with a spa or sometimes online sales of weight loss injectables, they’re not always certain about how to dose themselves, how much to give, and what to take. They could misread the instructions. The more that it’s up to them to determine the dose, the more there’s risk for error. Reports show as much as 1500% increases in poisoning of people who took, instead of a 10th of a milliliter, 10 mL of these compounded versions of the injectable drugs.
Everybody needs to be alert, and not only for adverse events from the prescription injectables. It is important to track that, make sure that people aren’t getting into trouble, and have contact with the FDA if you have a patient who reports some kind of adverse event they attribute to injectables.
It’s important to realize that there’s this generic, cheaper path, but it’s a more dangerous path. People need to know this if they’re going to try that route. Doctors should be aware of it. People should be ready to call the poison control center number in their area to make sure that they know what to do if they overdose on this stuff.
My own inclination is to try to discourage its use. I think it’s still too dangerous to have people self-dosing with ingredients that really are not yet FDA approved in terms of knowing that they’ve been tested in clinical trials.
The other big issue, aside from this Wild West world outside of prescribed injectables, is what to say to people who are obese or trying to manage their weight. I think people need to know all their options. It’s pretty easy to just say, “Let’s put you on one of these injectables” and prescribe it. For one thing, they may not be able to get it; there’s such huge demand that there are some shortages out there.
We don’t really know the long-term consequences of decades-long use of these drugs.
I think people should hear their options and maybe try something less invasive to begin with. If that doesn’t work, then move on to the injectables. It isn’t so clear to me — given the cost, some of the unknowns of long-term use, and some of the dangers of people sneaking around and trying to get things cheaper on the side — that going straight to injectables is our best answer.
I do think doctors should talk about weight with their patients, carefully, with the patient’s consent. Make sure there’s no stigma. Make sure we’re not doing anything to raise anxiety as we talk about this condition. After all, it is seen as a disease.
Then, maybe enter your way gradually into interventions, seeing if lifestyle change is possible. It’s cheap and easier to implement: better diet, better exercise, or calorie counting. Some people succeed. When they don’t, we should move on, but realize that we’ve got the equivalent of a black market. We need to encourage patients, if they use injectable weight loss drugs, to tell doctors so that they can be on alert about the dangers and risks of overdose.
Dr. Caplan is Director, Division of Medical Ethics, New York University Langone Medical Center, New York City. He disclosed an unpaid position with Johnson & Johnson’s Panel for Compassionate Drug Use, and serves as a contributing author and advisor for Medscape.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
There’s never been anything like the revolution in the treatment of obesity that we are now living through. Historically, there’s always been calorie counting and diets. Now, after a burst of interest in gastric bypass surgery, we have the amazing world of injectables. We all have heard about Ozempic, Mounjaro, and Wegovy.
These are being used by millions of Americans at this point, some on prescription for conditions like diabetes and some to bring about weight loss in prediabetes, or in some instances — as is often seen on American television — weight control or weight loss by people who just want to look better. Celebrities getting behind these injectables has really powered an explosion of use.
There still are ethical issues out there for practitioners. For one thing, there are some forms of semaglutide, a key ingredient in some of these injectables, that are made by compounding pharmacies. They’re not the name-brand prescription injectables made by large companies. They’re brewed up, if you will, by a specialty pharmacy trying to mimic the ingredient.
What we’ve seen in recent weeks is an explosion of overdoses. When a person uses one of these compounding pharmacies, usually in association with a spa or sometimes online sales of weight loss injectables, they’re not always certain about how to dose themselves, how much to give, and what to take. They could misread the instructions. The more that it’s up to them to determine the dose, the more there’s risk for error. Reports show as much as 1500% increases in poisoning of people who took, instead of a 10th of a milliliter, 10 mL of these compounded versions of the injectable drugs.
Everybody needs to be alert, and not only for adverse events from the prescription injectables. It is important to track that, make sure that people aren’t getting into trouble, and have contact with the FDA if you have a patient who reports some kind of adverse event they attribute to injectables.
It’s important to realize that there’s this generic, cheaper path, but it’s a more dangerous path. People need to know this if they’re going to try that route. Doctors should be aware of it. People should be ready to call the poison control center number in their area to make sure that they know what to do if they overdose on this stuff.
My own inclination is to try to discourage its use. I think it’s still too dangerous to have people self-dosing with ingredients that really are not yet FDA approved in terms of knowing that they’ve been tested in clinical trials.
The other big issue, aside from this Wild West world outside of prescribed injectables, is what to say to people who are obese or trying to manage their weight. I think people need to know all their options. It’s pretty easy to just say, “Let’s put you on one of these injectables” and prescribe it. For one thing, they may not be able to get it; there’s such huge demand that there are some shortages out there.
We don’t really know the long-term consequences of decades-long use of these drugs.
I think people should hear their options and maybe try something less invasive to begin with. If that doesn’t work, then move on to the injectables. It isn’t so clear to me — given the cost, some of the unknowns of long-term use, and some of the dangers of people sneaking around and trying to get things cheaper on the side — that going straight to injectables is our best answer.
I do think doctors should talk about weight with their patients, carefully, with the patient’s consent. Make sure there’s no stigma. Make sure we’re not doing anything to raise anxiety as we talk about this condition. After all, it is seen as a disease.
Then, maybe enter your way gradually into interventions, seeing if lifestyle change is possible. It’s cheap and easier to implement: better diet, better exercise, or calorie counting. Some people succeed. When they don’t, we should move on, but realize that we’ve got the equivalent of a black market. We need to encourage patients, if they use injectable weight loss drugs, to tell doctors so that they can be on alert about the dangers and risks of overdose.
Dr. Caplan is Director, Division of Medical Ethics, New York University Langone Medical Center, New York City. He disclosed an unpaid position with Johnson & Johnson’s Panel for Compassionate Drug Use, and serves as a contributing author and advisor for Medscape.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
There’s never been anything like the revolution in the treatment of obesity that we are now living through. Historically, there’s always been calorie counting and diets. Now, after a burst of interest in gastric bypass surgery, we have the amazing world of injectables. We all have heard about Ozempic, Mounjaro, and Wegovy.
These are being used by millions of Americans at this point, some on prescription for conditions like diabetes and some to bring about weight loss in prediabetes, or in some instances — as is often seen on American television — weight control or weight loss by people who just want to look better. Celebrities getting behind these injectables has really powered an explosion of use.
There still are ethical issues out there for practitioners. For one thing, there are some forms of semaglutide, a key ingredient in some of these injectables, that are made by compounding pharmacies. They’re not the name-brand prescription injectables made by large companies. They’re brewed up, if you will, by a specialty pharmacy trying to mimic the ingredient.
What we’ve seen in recent weeks is an explosion of overdoses. When a person uses one of these compounding pharmacies, usually in association with a spa or sometimes online sales of weight loss injectables, they’re not always certain about how to dose themselves, how much to give, and what to take. They could misread the instructions. The more that it’s up to them to determine the dose, the more there’s risk for error. Reports show as much as 1500% increases in poisoning of people who took, instead of a 10th of a milliliter, 10 mL of these compounded versions of the injectable drugs.
Everybody needs to be alert, and not only for adverse events from the prescription injectables. It is important to track that, make sure that people aren’t getting into trouble, and have contact with the FDA if you have a patient who reports some kind of adverse event they attribute to injectables.
It’s important to realize that there’s this generic, cheaper path, but it’s a more dangerous path. People need to know this if they’re going to try that route. Doctors should be aware of it. People should be ready to call the poison control center number in their area to make sure that they know what to do if they overdose on this stuff.
My own inclination is to try to discourage its use. I think it’s still too dangerous to have people self-dosing with ingredients that really are not yet FDA approved in terms of knowing that they’ve been tested in clinical trials.
The other big issue, aside from this Wild West world outside of prescribed injectables, is what to say to people who are obese or trying to manage their weight. I think people need to know all their options. It’s pretty easy to just say, “Let’s put you on one of these injectables” and prescribe it. For one thing, they may not be able to get it; there’s such huge demand that there are some shortages out there.
We don’t really know the long-term consequences of decades-long use of these drugs.
I think people should hear their options and maybe try something less invasive to begin with. If that doesn’t work, then move on to the injectables. It isn’t so clear to me — given the cost, some of the unknowns of long-term use, and some of the dangers of people sneaking around and trying to get things cheaper on the side — that going straight to injectables is our best answer.
I do think doctors should talk about weight with their patients, carefully, with the patient’s consent. Make sure there’s no stigma. Make sure we’re not doing anything to raise anxiety as we talk about this condition. After all, it is seen as a disease.
Then, maybe enter your way gradually into interventions, seeing if lifestyle change is possible. It’s cheap and easier to implement: better diet, better exercise, or calorie counting. Some people succeed. When they don’t, we should move on, but realize that we’ve got the equivalent of a black market. We need to encourage patients, if they use injectable weight loss drugs, to tell doctors so that they can be on alert about the dangers and risks of overdose.
Dr. Caplan is Director, Division of Medical Ethics, New York University Langone Medical Center, New York City. He disclosed an unpaid position with Johnson & Johnson’s Panel for Compassionate Drug Use, and serves as a contributing author and advisor for Medscape.
A version of this article appeared on Medscape.com.
Offsetting Side Effects of New Antiobesity Medications
It’s 2 a.m. and my phone wakes me up with a start. My patient, Christine Z*, is vomiting uncontrollably, and Dr Google has diagnosed her with acute pancreatitis from semaglutide (Wegovy). Ten hours, several imaging studies, one blood draw, and many bags of fluids later, the verdict is in: Christine is alarmingly constipated. In fact, her entire large intestine is packed to the brim with stool. In residency, we called this diagnosis FOS, and I’ll leave it to your imagination to figure out what it stands for.
In retrospect, Christine mentions that upon raising her Wegovy dose, her bowel movements had become increasingly smaller and infrequent. This begs the question:
Proper nutrition always starts with drinking copious amounts of water. In general, I recommend a minimum of 64 ounces of water daily in patients taking incretins such as semaglutide (Wegovy for weight loss, Ozempic and Rybelsus for type 2 diabetes) or tirzepatide (Zepbound for weight loss, Mounjaro for type 2 diabetes). While these medications don’t directly dehydrate patients, they can increase the risk for dehydration due to severe nausea. Drinking copious amounts of water can prevent dehydration, preserve kidney function, and minimize fatigue and dizziness. In addition, fluids help soften bowel movements, making them easier to pass.
Occasionally incretins make it so easy for patients to drop pounds that their eating patterns become sloppier — more sweets and simple carbohydrates. I recommend a realistic and low glycemic index meal plan. While no foods are strictly contraindicated, processed, high-sugar, and fatty foods are likely to worsen side effects like nausea and gastrointestinal distress. Similarly, alcohol not only worsens nausea, but it’s also likely to exacerbate reflux by relaxing the sphincter that separates the stomach from the esophagus.
The next most important dietary advice is consuming sufficient fiber. In the majority of patients, increasing fiber intake relieves constipation. There are two types of fiber: soluble and insoluble. In practical terms, most fiber-rich foods contain a mixture of these two types. The general recommendation is 38 g/d for men and 25 g/d for women. The caveat to this advice is that a minority of patients, such as those with irritable bowel syndrome, may develop worsening constipation with increasing fiber.
To minimize side effects, some patients find it useful to eat five small meals throughout the day rather than three larger meals. In addition, I recommend eating slowly and stopping before the point of satiety. Finally, because weight loss of any kind is inevitably associated with muscle loss, I stress the importance of adequate protein. In general, I advise 25-30 g of protein per meal.
Christine eventually restarted her Wegovy after recovering from her grueling night in the emergency room. As this was her second go-around on Wegovy, she dug out my “guide to preventing side effects of incretins” and followed it to a T. So far, she’s feeling great.
*The patient’s name has been changed.
A version of this article appeared on Medscape.com.
It’s 2 a.m. and my phone wakes me up with a start. My patient, Christine Z*, is vomiting uncontrollably, and Dr Google has diagnosed her with acute pancreatitis from semaglutide (Wegovy). Ten hours, several imaging studies, one blood draw, and many bags of fluids later, the verdict is in: Christine is alarmingly constipated. In fact, her entire large intestine is packed to the brim with stool. In residency, we called this diagnosis FOS, and I’ll leave it to your imagination to figure out what it stands for.
In retrospect, Christine mentions that upon raising her Wegovy dose, her bowel movements had become increasingly smaller and infrequent. This begs the question:
Proper nutrition always starts with drinking copious amounts of water. In general, I recommend a minimum of 64 ounces of water daily in patients taking incretins such as semaglutide (Wegovy for weight loss, Ozempic and Rybelsus for type 2 diabetes) or tirzepatide (Zepbound for weight loss, Mounjaro for type 2 diabetes). While these medications don’t directly dehydrate patients, they can increase the risk for dehydration due to severe nausea. Drinking copious amounts of water can prevent dehydration, preserve kidney function, and minimize fatigue and dizziness. In addition, fluids help soften bowel movements, making them easier to pass.
Occasionally incretins make it so easy for patients to drop pounds that their eating patterns become sloppier — more sweets and simple carbohydrates. I recommend a realistic and low glycemic index meal plan. While no foods are strictly contraindicated, processed, high-sugar, and fatty foods are likely to worsen side effects like nausea and gastrointestinal distress. Similarly, alcohol not only worsens nausea, but it’s also likely to exacerbate reflux by relaxing the sphincter that separates the stomach from the esophagus.
The next most important dietary advice is consuming sufficient fiber. In the majority of patients, increasing fiber intake relieves constipation. There are two types of fiber: soluble and insoluble. In practical terms, most fiber-rich foods contain a mixture of these two types. The general recommendation is 38 g/d for men and 25 g/d for women. The caveat to this advice is that a minority of patients, such as those with irritable bowel syndrome, may develop worsening constipation with increasing fiber.
To minimize side effects, some patients find it useful to eat five small meals throughout the day rather than three larger meals. In addition, I recommend eating slowly and stopping before the point of satiety. Finally, because weight loss of any kind is inevitably associated with muscle loss, I stress the importance of adequate protein. In general, I advise 25-30 g of protein per meal.
Christine eventually restarted her Wegovy after recovering from her grueling night in the emergency room. As this was her second go-around on Wegovy, she dug out my “guide to preventing side effects of incretins” and followed it to a T. So far, she’s feeling great.
*The patient’s name has been changed.
A version of this article appeared on Medscape.com.
It’s 2 a.m. and my phone wakes me up with a start. My patient, Christine Z*, is vomiting uncontrollably, and Dr Google has diagnosed her with acute pancreatitis from semaglutide (Wegovy). Ten hours, several imaging studies, one blood draw, and many bags of fluids later, the verdict is in: Christine is alarmingly constipated. In fact, her entire large intestine is packed to the brim with stool. In residency, we called this diagnosis FOS, and I’ll leave it to your imagination to figure out what it stands for.
In retrospect, Christine mentions that upon raising her Wegovy dose, her bowel movements had become increasingly smaller and infrequent. This begs the question:
Proper nutrition always starts with drinking copious amounts of water. In general, I recommend a minimum of 64 ounces of water daily in patients taking incretins such as semaglutide (Wegovy for weight loss, Ozempic and Rybelsus for type 2 diabetes) or tirzepatide (Zepbound for weight loss, Mounjaro for type 2 diabetes). While these medications don’t directly dehydrate patients, they can increase the risk for dehydration due to severe nausea. Drinking copious amounts of water can prevent dehydration, preserve kidney function, and minimize fatigue and dizziness. In addition, fluids help soften bowel movements, making them easier to pass.
Occasionally incretins make it so easy for patients to drop pounds that their eating patterns become sloppier — more sweets and simple carbohydrates. I recommend a realistic and low glycemic index meal plan. While no foods are strictly contraindicated, processed, high-sugar, and fatty foods are likely to worsen side effects like nausea and gastrointestinal distress. Similarly, alcohol not only worsens nausea, but it’s also likely to exacerbate reflux by relaxing the sphincter that separates the stomach from the esophagus.
The next most important dietary advice is consuming sufficient fiber. In the majority of patients, increasing fiber intake relieves constipation. There are two types of fiber: soluble and insoluble. In practical terms, most fiber-rich foods contain a mixture of these two types. The general recommendation is 38 g/d for men and 25 g/d for women. The caveat to this advice is that a minority of patients, such as those with irritable bowel syndrome, may develop worsening constipation with increasing fiber.
To minimize side effects, some patients find it useful to eat five small meals throughout the day rather than three larger meals. In addition, I recommend eating slowly and stopping before the point of satiety. Finally, because weight loss of any kind is inevitably associated with muscle loss, I stress the importance of adequate protein. In general, I advise 25-30 g of protein per meal.
Christine eventually restarted her Wegovy after recovering from her grueling night in the emergency room. As this was her second go-around on Wegovy, she dug out my “guide to preventing side effects of incretins” and followed it to a T. So far, she’s feeling great.
*The patient’s name has been changed.
A version of this article appeared on Medscape.com.
Unlikely Breakthrough of the Year: Chemo for Lung Cancer
This transcript has been edited for clarity.
I’ve been spending time recently reflecting on the biggest developments from last year. I have to say that the breakthrough of the year, based on the amount of data presented and the importance of the data, is chemotherapy. I never thought I would say that. Many folks have tried to relegate chemotherapy to the museum, but last year it came to the forefront.
Let’s start with neoadjuvant therapy. We now have multiple drug approvals for giving a checkpoint inhibitor and neoadjuvant therapy in what I would say is a new standard of care for patients with locally advanced lung cancers who are candidates for surgery. In all those trials, there was a clear improvement in progression-free survival by adding a checkpoint inhibitor to chemotherapy. The cornerstone of this regimen is chemotherapy.
What about adjuvant therapy? I think one of the most astounding pieces of data last year was in the adjuvant realm. In the trial comparing adjuvant osimertinib with placebo in patients with EGFR-mutant disease, patients who received chemotherapy in addition to osimertinib had a 7% improvement in 5-year survival. Patients who had placebo, who got chemotherapy vs didn’t, had a 9% improvement in 5-year survival. Those are huge numbers for that kind of metric, and it happened with chemotherapy.
What about targeted therapies? Again, I think people were astounded that, by adding chemotherapy to osimertinib compared with osimertinib alone, there was a 9-month improvement overall in progression-free survival. I think in the presentation of the data that has been made, the most remarkable piece of data is that, in patients with brain metastases, chemotherapy on top of osimertinib improved progression-free survival. Not only did it improve progression-free survival, but it did it with brain metastases, where people think it just doesn’t help at all.
What about other, newer agents with chemotherapy? Amivantamab, I would say, has hitched itself to chemotherapy. A trial in EGFR exon 20 compared chemo to amivantamab plus chemotherapy. There again, chemo is the common denominator. Amivantamab added approximately 5 months of improved progression-free survival. Again, chemo was used. In adjuvant, neoadjuvant, and targeted therapies, chemotherapy adds.
What about the second line? I think everybody was very disappointed when second-line sotorasib gave a very tiny amount of progression-free survival improvement over docetaxel. I think we all want more for our patients than we can deliver with docetaxel. The roughly 5-week improvement seen with sotorasib was one that raised a question about the place of sotorasib in this setting.
Clearly, we’ve all seen patients have an excellent result with sotorasib as an additional option for treating patients with long progression-free survival, high rates of response, and good tolerability even at the 960 mg dose. But in the randomized trial, it wasn’t better than docetaxel. Again, I think we were disappointed with tusamitamab ravtansine in that it could not beat docetaxel either. I think the idea here is that chemo still has a huge place and still remains the treatment that we have to beat.
We’re all very excited about the antibody-drug conjugates and I think everybody sees them as another advance. Many folks have said that they are just a more precise way of delivering chemotherapy, and when you look at the side effects, it supports that — they’re largely side effects of chemotherapy with these drugs across the board. Also, when you look at the patterns of resistance, the resistance really isn’t a resistance to the targeted therapy; it’s a resistance to chemotherapy more than anything else.
So we’re happy that the antibody-drug conjugates are available and we were disappointed with tusamitamab ravtansine because we thought that it could beat docetaxel. But in truth, it didn’t, and unfortunately, that pivotal trial led to the end of the entire development program for that agent, as stated in a press release.
The molecule or treatment of the year is chemotherapy — added to targeted therapies, used with immunotherapy, and now attached to antibodies as part of antibody-drug conjugates. I think it remains, more than any one treatment, a very effective treatment for patients and deserves to be used.
There are a lot of choices here. I think you have to be very careful to choose wisely, and you also have to be careful because chemotherapy has side effects. The nice thing is that many of those side effects can be ameliorated. We have to make sure that we use all the supportive medications we can.
Who would have thought that chemotherapy would be the treatment of the year in 2023 for lung cancers?
Dr. Kris is chief of the thoracic oncology service and the William and Joy Ruane Chair in Thoracic Oncology at Memorial Sloan Kettering Cancer Center in New York City. He disclosed ties with AstraZeneca, Roche/Genentech, Ariad Pharmaceuticals, Pfizer Inc, and PUMA.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
I’ve been spending time recently reflecting on the biggest developments from last year. I have to say that the breakthrough of the year, based on the amount of data presented and the importance of the data, is chemotherapy. I never thought I would say that. Many folks have tried to relegate chemotherapy to the museum, but last year it came to the forefront.
Let’s start with neoadjuvant therapy. We now have multiple drug approvals for giving a checkpoint inhibitor and neoadjuvant therapy in what I would say is a new standard of care for patients with locally advanced lung cancers who are candidates for surgery. In all those trials, there was a clear improvement in progression-free survival by adding a checkpoint inhibitor to chemotherapy. The cornerstone of this regimen is chemotherapy.
What about adjuvant therapy? I think one of the most astounding pieces of data last year was in the adjuvant realm. In the trial comparing adjuvant osimertinib with placebo in patients with EGFR-mutant disease, patients who received chemotherapy in addition to osimertinib had a 7% improvement in 5-year survival. Patients who had placebo, who got chemotherapy vs didn’t, had a 9% improvement in 5-year survival. Those are huge numbers for that kind of metric, and it happened with chemotherapy.
What about targeted therapies? Again, I think people were astounded that, by adding chemotherapy to osimertinib compared with osimertinib alone, there was a 9-month improvement overall in progression-free survival. I think in the presentation of the data that has been made, the most remarkable piece of data is that, in patients with brain metastases, chemotherapy on top of osimertinib improved progression-free survival. Not only did it improve progression-free survival, but it did it with brain metastases, where people think it just doesn’t help at all.
What about other, newer agents with chemotherapy? Amivantamab, I would say, has hitched itself to chemotherapy. A trial in EGFR exon 20 compared chemo to amivantamab plus chemotherapy. There again, chemo is the common denominator. Amivantamab added approximately 5 months of improved progression-free survival. Again, chemo was used. In adjuvant, neoadjuvant, and targeted therapies, chemotherapy adds.
What about the second line? I think everybody was very disappointed when second-line sotorasib gave a very tiny amount of progression-free survival improvement over docetaxel. I think we all want more for our patients than we can deliver with docetaxel. The roughly 5-week improvement seen with sotorasib was one that raised a question about the place of sotorasib in this setting.
Clearly, we’ve all seen patients have an excellent result with sotorasib as an additional option for treating patients with long progression-free survival, high rates of response, and good tolerability even at the 960 mg dose. But in the randomized trial, it wasn’t better than docetaxel. Again, I think we were disappointed with tusamitamab ravtansine in that it could not beat docetaxel either. I think the idea here is that chemo still has a huge place and still remains the treatment that we have to beat.
We’re all very excited about the antibody-drug conjugates and I think everybody sees them as another advance. Many folks have said that they are just a more precise way of delivering chemotherapy, and when you look at the side effects, it supports that — they’re largely side effects of chemotherapy with these drugs across the board. Also, when you look at the patterns of resistance, the resistance really isn’t a resistance to the targeted therapy; it’s a resistance to chemotherapy more than anything else.
So we’re happy that the antibody-drug conjugates are available and we were disappointed with tusamitamab ravtansine because we thought that it could beat docetaxel. But in truth, it didn’t, and unfortunately, that pivotal trial led to the end of the entire development program for that agent, as stated in a press release.
The molecule or treatment of the year is chemotherapy — added to targeted therapies, used with immunotherapy, and now attached to antibodies as part of antibody-drug conjugates. I think it remains, more than any one treatment, a very effective treatment for patients and deserves to be used.
There are a lot of choices here. I think you have to be very careful to choose wisely, and you also have to be careful because chemotherapy has side effects. The nice thing is that many of those side effects can be ameliorated. We have to make sure that we use all the supportive medications we can.
Who would have thought that chemotherapy would be the treatment of the year in 2023 for lung cancers?
Dr. Kris is chief of the thoracic oncology service and the William and Joy Ruane Chair in Thoracic Oncology at Memorial Sloan Kettering Cancer Center in New York City. He disclosed ties with AstraZeneca, Roche/Genentech, Ariad Pharmaceuticals, Pfizer Inc, and PUMA.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
I’ve been spending time recently reflecting on the biggest developments from last year. I have to say that the breakthrough of the year, based on the amount of data presented and the importance of the data, is chemotherapy. I never thought I would say that. Many folks have tried to relegate chemotherapy to the museum, but last year it came to the forefront.
Let’s start with neoadjuvant therapy. We now have multiple drug approvals for giving a checkpoint inhibitor and neoadjuvant therapy in what I would say is a new standard of care for patients with locally advanced lung cancers who are candidates for surgery. In all those trials, there was a clear improvement in progression-free survival by adding a checkpoint inhibitor to chemotherapy. The cornerstone of this regimen is chemotherapy.
What about adjuvant therapy? I think one of the most astounding pieces of data last year was in the adjuvant realm. In the trial comparing adjuvant osimertinib with placebo in patients with EGFR-mutant disease, patients who received chemotherapy in addition to osimertinib had a 7% improvement in 5-year survival. Patients who had placebo, who got chemotherapy vs didn’t, had a 9% improvement in 5-year survival. Those are huge numbers for that kind of metric, and it happened with chemotherapy.
What about targeted therapies? Again, I think people were astounded that, by adding chemotherapy to osimertinib compared with osimertinib alone, there was a 9-month improvement overall in progression-free survival. I think in the presentation of the data that has been made, the most remarkable piece of data is that, in patients with brain metastases, chemotherapy on top of osimertinib improved progression-free survival. Not only did it improve progression-free survival, but it did it with brain metastases, where people think it just doesn’t help at all.
What about other, newer agents with chemotherapy? Amivantamab, I would say, has hitched itself to chemotherapy. A trial in EGFR exon 20 compared chemo to amivantamab plus chemotherapy. There again, chemo is the common denominator. Amivantamab added approximately 5 months of improved progression-free survival. Again, chemo was used. In adjuvant, neoadjuvant, and targeted therapies, chemotherapy adds.
What about the second line? I think everybody was very disappointed when second-line sotorasib gave a very tiny amount of progression-free survival improvement over docetaxel. I think we all want more for our patients than we can deliver with docetaxel. The roughly 5-week improvement seen with sotorasib was one that raised a question about the place of sotorasib in this setting.
Clearly, we’ve all seen patients have an excellent result with sotorasib as an additional option for treating patients with long progression-free survival, high rates of response, and good tolerability even at the 960 mg dose. But in the randomized trial, it wasn’t better than docetaxel. Again, I think we were disappointed with tusamitamab ravtansine in that it could not beat docetaxel either. I think the idea here is that chemo still has a huge place and still remains the treatment that we have to beat.
We’re all very excited about the antibody-drug conjugates and I think everybody sees them as another advance. Many folks have said that they are just a more precise way of delivering chemotherapy, and when you look at the side effects, it supports that — they’re largely side effects of chemotherapy with these drugs across the board. Also, when you look at the patterns of resistance, the resistance really isn’t a resistance to the targeted therapy; it’s a resistance to chemotherapy more than anything else.
So we’re happy that the antibody-drug conjugates are available and we were disappointed with tusamitamab ravtansine because we thought that it could beat docetaxel. But in truth, it didn’t, and unfortunately, that pivotal trial led to the end of the entire development program for that agent, as stated in a press release.
The molecule or treatment of the year is chemotherapy — added to targeted therapies, used with immunotherapy, and now attached to antibodies as part of antibody-drug conjugates. I think it remains, more than any one treatment, a very effective treatment for patients and deserves to be used.
There are a lot of choices here. I think you have to be very careful to choose wisely, and you also have to be careful because chemotherapy has side effects. The nice thing is that many of those side effects can be ameliorated. We have to make sure that we use all the supportive medications we can.
Who would have thought that chemotherapy would be the treatment of the year in 2023 for lung cancers?
Dr. Kris is chief of the thoracic oncology service and the William and Joy Ruane Chair in Thoracic Oncology at Memorial Sloan Kettering Cancer Center in New York City. He disclosed ties with AstraZeneca, Roche/Genentech, Ariad Pharmaceuticals, Pfizer Inc, and PUMA.
A version of this article appeared on Medscape.com.
Implementing Trustworthy AI in VA High Reliability Health Care Organizations
Artificial intelligence (AI) has lagged in health care but has considerable potential to improve quality, safety, clinician experience, and access to care. It is being tested in areas like billing, hospital operations, and preventing adverse events (eg, sepsis mortality) with some early success. However, there are still many barriers preventing the widespread use of AI, such as data problems, mismatched rewards, and workplace obstacles. Innovative projects, partnerships, better rewards, and more investment could remove barriers. Implemented reliably and safely, AI can add to what clinicians know, help them work faster, cut costs, and, most importantly, improve patient care.1
AI can potentially bring several clinical benefits, such as reducing the administrative strain on clinicians and granting them more time for direct patient care. It can also improve diagnostic accuracy by analyzing patient data and diagnostic images, providing differential diagnoses, and increasing access to care by providing medical information and essential online services to patients.2
High Reliability Organizations
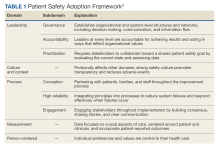
High reliability health care organizations have considerable experience safely launching new programs. For example, the Patient Safety Adoption Framework gives practical tips for smoothly rolling out safety initiatives (Table 1). Developed with experts and diverse views, this framework has 5 key areas: leadership, culture and context, process, measurement, and person-centeredness. These address adoption problems, guide leaders step-by-step, and focus on leadership buy-in, safety culture, cooperation, and local customization. Checklists and tools make it systematic to go from ideas to action on patient safety.3
Leadership involves establishing organizational commitment behind new safety programs. This visible commitment signals importance and priorities to others. Leaders model desired behaviors and language around safety, allocate resources, remove obstacles, and keep initiatives energized over time through consistent messaging.4 Culture and context recognizes that safety culture differs across units and facilities. Local input tailors programs to fit and examines strengths to build on, like psychological safety. Surveys gauge the existing culture and its need for change. Process details how to plan, design, test, implement, and improve new safety practices and provides a phased roadmap from idea to results. Measurement collects data to drive improvement and show impact. Metrics track progress and allow benchmarking. Person-centeredness puts patients first in safety efforts through participation, education, and transparency.
The Veterans Health Administration piloted a comprehensive high reliability hospital (HRH) model. Over 3 years, the Veterans Health Administration focused on leadership, culture, and process improvement at a hospital. After initiating the model, the pilot hospital improved its safety culture, reported more minor safety issues, and reduced deaths and complications better than other hospitals. The high-reliability approach successfully instilled principles and improved culture and outcomes. The HRH model is set to be expanded to 18 more US Department of Veterans Affairs (VA) sites for further evaluation across diverse settings.5
Trustworthy AI Framework
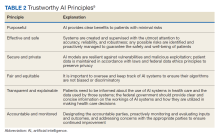
AI systems are growing more powerful and widespread, including in health care. Unfortunately, irresponsible AI can introduce new harm. ChatGPT and other large language models, for example, sometimes are known to provide erroneous information in a compelling way. Clinicians and patients who use such programs can act on such information, which would lead to unforeseen negative consequences. Several frameworks on ethical AI have come from governmental groups.6-9 In 2023, the VA National AI Institute suggested a Trustworthy AI Framework based on core principles tailored for federal health care. The framework has 6 key principles: purposeful, effective and safe, secure and private, fair and equitable, transparent and explainable, and accountable and monitored (Table 2).10
First, AI must clearly help veterans while minimizing risks. To ensure purpose, the VA will assess patient and clinician needs and design AI that targets meaningful problems to avoid scope creep or feature bloat. For example, adding new features to the AI software after release can clutter and complicate the interface, making it difficult to use. Rigorous testing will confirm that AI meets intent prior to deployment. Second, AI is designed and checked for effectiveness, safety, and reliability. The VA pledges to monitor AI’s impact to ensure it performs as expected without unintended consequences. Algorithms will be stress tested across representative datasets and approval processes will screen for safety issues. Third, AI models are secured from vulnerabilities and misuse. Technical controls will prevent unauthorized access or changes to AI systems. Audits will check for appropriate internal usage per policies. Continual patches and upgrades will maintain security. Fourth, the VA manages AI for fairness, avoiding bias. They will proactively assess datasets and algorithms for potential biases based on protected attributes like race, gender, or age. Biased outputs will be addressed through techniques such as data augmentation, reweighting, and algorithm tweaks. Fifth, transparency explains AI’s role in care. Documentation will detail an AI system’s data sources, methodology, testing, limitations, and integration with clinical workflows. Clinicians and patients will receive education on interpreting AI outputs. Finally, the VA pledges to closely monitor AI systems to sustain trust. The VA will establish oversight processes to quickly identify any declines in reliability or unfair impacts on subgroups. AI models will be retrained as needed based on incoming data patterns.
Each Trustworthy AI Framework principle connects to others in existing frameworks. The purpose principle aligns with human-centric AI focused on benefits. Effectiveness and safety link to technical robustness and risk management principles. Security maps to privacy protection principles. Fairness connects to principles of avoiding bias and discrimination. Transparency corresponds with accountable and explainable AI. Monitoring and accountability tie back to governance principles. Overall, the VA framework aims to guide ethical AI based on context. It offers a model for managing risks and building trust in health care AI.
Combining VA principles with high-reliability safety principles can ensure that AI benefits veterans. The leadership and culture aspects will drive commitment to trustworthy AI practices. Leaders will communicate the importance of responsible AI through words and actions. Culture surveys can assess baseline awareness of AI ethics issues to target education. AI security and fairness will be emphasized as safety critical. The process aspect will institute policies and procedures to uphold AI principles through the project lifecycle. For example, structured testing processes will validate safety. Measurement will collect data on principles like transparency and fairness. Dashboards can track metrics like explainability and biases. A patient-centered approach will incorporate veteran perspectives on AI through participatory design and advisory councils. They can give input on AI explainability and potential biases based on their diverse backgrounds.
Conclusions
Joint principles will lead to successful AI that improves care while proactively managing risks. Involve leaders to stress the necessity of eliminating biases. Build security into the AI development process. Co-design AI transparency features with end users. Closely monitor the impact of AI across safety, fairness, and other principles. Adhering to both Trustworthy AI and high reliability organizations principles will earn veterans’ confidence. Health care organizations like the VA can integrate ethical AI safely via established frameworks. With responsible design and implementation, AI’s potential to enhance care quality, safety, and access can be realized.
Acknowledgments
We would like to acknowledge Joshua Mueller, Theo Tiffney, John Zachary, and Gil Alterovitz for their excellent work creating the VA Trustworthy Principles. This material is the result of work supported by resources and the use of facilities at the James A. Haley Veterans’ Hospital.
1. Sahni NR, Carrus B. Artificial intelligence in U.S. health care delivery. N Engl J Med. 2023;389(4):348-358. doi:10.1056/NEJMra2204673
2. Borkowski AA, Jakey CE, Mastorides SM, et al. Applications of ChatGPT and large language models in medicine and health care: benefits and pitfalls. Fed Pract. 2023;40(6):170-173. doi:10.12788/fp.0386
3. Moyal-Smith R, Margo J, Maloney FL, et al. The patient safety adoption framework: a practical framework to bridge the know-do gap. J Patient Saf. 2023;19(4):243-248. doi:10.1097/PTS.0000000000001118
4. Isaacks DB, Anderson TM, Moore SC, Patterson W, Govindan S. High reliability organization principles improve VA workplace burnout: the Truman THRIVE2 model. Am J Med Qual. 2021;36(6):422-428. doi:10.1097/01.JMQ.0000735516.35323.97
5. Sculli GL, Pendley-Louis R, Neily J, et al. A high-reliability organization framework for health care: a multiyear implementation strategy and associated outcomes. J Patient Saf. 2022;18(1):64-70. doi:10.1097/PTS.0000000000000788
6. National Institute of Standards and Technology. AI risk management framework. Accessed January 2, 2024. https://www.nist.gov/itl/ai-risk-management-framework
7. Executive Office of the President, Office of Science and Technology Policy. Blueprint for an AI Bill of Rights. Accessed January 11, 2024. https://www.whitehouse.gov/ostp/ai-bill-of-rights
8. Executive Office of the President. Executive Order 13960: promoting the use of trustworthy artificial intelligence in the federal government. Fed Regist. 2020;89(236):78939-78943.
9. Biden JR. Executive Order on the safe, secure, and trustworthy development and use of artificial intelligence. Published October 30, 2023. Accessed January 11, 2024. https://www.whitehouse.gov/briefing-room/presidential-actions/2023/10/30/executive-order-on-the-safe-secure-and-trustworthy-development-and-use-of-artificial-intelligence/
10. US Department of Veterans Affairs. Trustworthy AI. Accessed January 11, 2024. https://department.va.gov/ai/trustworthy/
Artificial intelligence (AI) has lagged in health care but has considerable potential to improve quality, safety, clinician experience, and access to care. It is being tested in areas like billing, hospital operations, and preventing adverse events (eg, sepsis mortality) with some early success. However, there are still many barriers preventing the widespread use of AI, such as data problems, mismatched rewards, and workplace obstacles. Innovative projects, partnerships, better rewards, and more investment could remove barriers. Implemented reliably and safely, AI can add to what clinicians know, help them work faster, cut costs, and, most importantly, improve patient care.1
AI can potentially bring several clinical benefits, such as reducing the administrative strain on clinicians and granting them more time for direct patient care. It can also improve diagnostic accuracy by analyzing patient data and diagnostic images, providing differential diagnoses, and increasing access to care by providing medical information and essential online services to patients.2
High Reliability Organizations
High reliability health care organizations have considerable experience safely launching new programs. For example, the Patient Safety Adoption Framework gives practical tips for smoothly rolling out safety initiatives (Table 1). Developed with experts and diverse views, this framework has 5 key areas: leadership, culture and context, process, measurement, and person-centeredness. These address adoption problems, guide leaders step-by-step, and focus on leadership buy-in, safety culture, cooperation, and local customization. Checklists and tools make it systematic to go from ideas to action on patient safety.3
Leadership involves establishing organizational commitment behind new safety programs. This visible commitment signals importance and priorities to others. Leaders model desired behaviors and language around safety, allocate resources, remove obstacles, and keep initiatives energized over time through consistent messaging.4 Culture and context recognizes that safety culture differs across units and facilities. Local input tailors programs to fit and examines strengths to build on, like psychological safety. Surveys gauge the existing culture and its need for change. Process details how to plan, design, test, implement, and improve new safety practices and provides a phased roadmap from idea to results. Measurement collects data to drive improvement and show impact. Metrics track progress and allow benchmarking. Person-centeredness puts patients first in safety efforts through participation, education, and transparency.
The Veterans Health Administration piloted a comprehensive high reliability hospital (HRH) model. Over 3 years, the Veterans Health Administration focused on leadership, culture, and process improvement at a hospital. After initiating the model, the pilot hospital improved its safety culture, reported more minor safety issues, and reduced deaths and complications better than other hospitals. The high-reliability approach successfully instilled principles and improved culture and outcomes. The HRH model is set to be expanded to 18 more US Department of Veterans Affairs (VA) sites for further evaluation across diverse settings.5
Trustworthy AI Framework
AI systems are growing more powerful and widespread, including in health care. Unfortunately, irresponsible AI can introduce new harm. ChatGPT and other large language models, for example, sometimes are known to provide erroneous information in a compelling way. Clinicians and patients who use such programs can act on such information, which would lead to unforeseen negative consequences. Several frameworks on ethical AI have come from governmental groups.6-9 In 2023, the VA National AI Institute suggested a Trustworthy AI Framework based on core principles tailored for federal health care. The framework has 6 key principles: purposeful, effective and safe, secure and private, fair and equitable, transparent and explainable, and accountable and monitored (Table 2).10
First, AI must clearly help veterans while minimizing risks. To ensure purpose, the VA will assess patient and clinician needs and design AI that targets meaningful problems to avoid scope creep or feature bloat. For example, adding new features to the AI software after release can clutter and complicate the interface, making it difficult to use. Rigorous testing will confirm that AI meets intent prior to deployment. Second, AI is designed and checked for effectiveness, safety, and reliability. The VA pledges to monitor AI’s impact to ensure it performs as expected without unintended consequences. Algorithms will be stress tested across representative datasets and approval processes will screen for safety issues. Third, AI models are secured from vulnerabilities and misuse. Technical controls will prevent unauthorized access or changes to AI systems. Audits will check for appropriate internal usage per policies. Continual patches and upgrades will maintain security. Fourth, the VA manages AI for fairness, avoiding bias. They will proactively assess datasets and algorithms for potential biases based on protected attributes like race, gender, or age. Biased outputs will be addressed through techniques such as data augmentation, reweighting, and algorithm tweaks. Fifth, transparency explains AI’s role in care. Documentation will detail an AI system’s data sources, methodology, testing, limitations, and integration with clinical workflows. Clinicians and patients will receive education on interpreting AI outputs. Finally, the VA pledges to closely monitor AI systems to sustain trust. The VA will establish oversight processes to quickly identify any declines in reliability or unfair impacts on subgroups. AI models will be retrained as needed based on incoming data patterns.
Each Trustworthy AI Framework principle connects to others in existing frameworks. The purpose principle aligns with human-centric AI focused on benefits. Effectiveness and safety link to technical robustness and risk management principles. Security maps to privacy protection principles. Fairness connects to principles of avoiding bias and discrimination. Transparency corresponds with accountable and explainable AI. Monitoring and accountability tie back to governance principles. Overall, the VA framework aims to guide ethical AI based on context. It offers a model for managing risks and building trust in health care AI.
Combining VA principles with high-reliability safety principles can ensure that AI benefits veterans. The leadership and culture aspects will drive commitment to trustworthy AI practices. Leaders will communicate the importance of responsible AI through words and actions. Culture surveys can assess baseline awareness of AI ethics issues to target education. AI security and fairness will be emphasized as safety critical. The process aspect will institute policies and procedures to uphold AI principles through the project lifecycle. For example, structured testing processes will validate safety. Measurement will collect data on principles like transparency and fairness. Dashboards can track metrics like explainability and biases. A patient-centered approach will incorporate veteran perspectives on AI through participatory design and advisory councils. They can give input on AI explainability and potential biases based on their diverse backgrounds.
Conclusions
Joint principles will lead to successful AI that improves care while proactively managing risks. Involve leaders to stress the necessity of eliminating biases. Build security into the AI development process. Co-design AI transparency features with end users. Closely monitor the impact of AI across safety, fairness, and other principles. Adhering to both Trustworthy AI and high reliability organizations principles will earn veterans’ confidence. Health care organizations like the VA can integrate ethical AI safely via established frameworks. With responsible design and implementation, AI’s potential to enhance care quality, safety, and access can be realized.
Acknowledgments
We would like to acknowledge Joshua Mueller, Theo Tiffney, John Zachary, and Gil Alterovitz for their excellent work creating the VA Trustworthy Principles. This material is the result of work supported by resources and the use of facilities at the James A. Haley Veterans’ Hospital.
Artificial intelligence (AI) has lagged in health care but has considerable potential to improve quality, safety, clinician experience, and access to care. It is being tested in areas like billing, hospital operations, and preventing adverse events (eg, sepsis mortality) with some early success. However, there are still many barriers preventing the widespread use of AI, such as data problems, mismatched rewards, and workplace obstacles. Innovative projects, partnerships, better rewards, and more investment could remove barriers. Implemented reliably and safely, AI can add to what clinicians know, help them work faster, cut costs, and, most importantly, improve patient care.1
AI can potentially bring several clinical benefits, such as reducing the administrative strain on clinicians and granting them more time for direct patient care. It can also improve diagnostic accuracy by analyzing patient data and diagnostic images, providing differential diagnoses, and increasing access to care by providing medical information and essential online services to patients.2
High Reliability Organizations
High reliability health care organizations have considerable experience safely launching new programs. For example, the Patient Safety Adoption Framework gives practical tips for smoothly rolling out safety initiatives (Table 1). Developed with experts and diverse views, this framework has 5 key areas: leadership, culture and context, process, measurement, and person-centeredness. These address adoption problems, guide leaders step-by-step, and focus on leadership buy-in, safety culture, cooperation, and local customization. Checklists and tools make it systematic to go from ideas to action on patient safety.3
Leadership involves establishing organizational commitment behind new safety programs. This visible commitment signals importance and priorities to others. Leaders model desired behaviors and language around safety, allocate resources, remove obstacles, and keep initiatives energized over time through consistent messaging.4 Culture and context recognizes that safety culture differs across units and facilities. Local input tailors programs to fit and examines strengths to build on, like psychological safety. Surveys gauge the existing culture and its need for change. Process details how to plan, design, test, implement, and improve new safety practices and provides a phased roadmap from idea to results. Measurement collects data to drive improvement and show impact. Metrics track progress and allow benchmarking. Person-centeredness puts patients first in safety efforts through participation, education, and transparency.
The Veterans Health Administration piloted a comprehensive high reliability hospital (HRH) model. Over 3 years, the Veterans Health Administration focused on leadership, culture, and process improvement at a hospital. After initiating the model, the pilot hospital improved its safety culture, reported more minor safety issues, and reduced deaths and complications better than other hospitals. The high-reliability approach successfully instilled principles and improved culture and outcomes. The HRH model is set to be expanded to 18 more US Department of Veterans Affairs (VA) sites for further evaluation across diverse settings.5
Trustworthy AI Framework
AI systems are growing more powerful and widespread, including in health care. Unfortunately, irresponsible AI can introduce new harm. ChatGPT and other large language models, for example, sometimes are known to provide erroneous information in a compelling way. Clinicians and patients who use such programs can act on such information, which would lead to unforeseen negative consequences. Several frameworks on ethical AI have come from governmental groups.6-9 In 2023, the VA National AI Institute suggested a Trustworthy AI Framework based on core principles tailored for federal health care. The framework has 6 key principles: purposeful, effective and safe, secure and private, fair and equitable, transparent and explainable, and accountable and monitored (Table 2).10
First, AI must clearly help veterans while minimizing risks. To ensure purpose, the VA will assess patient and clinician needs and design AI that targets meaningful problems to avoid scope creep or feature bloat. For example, adding new features to the AI software after release can clutter and complicate the interface, making it difficult to use. Rigorous testing will confirm that AI meets intent prior to deployment. Second, AI is designed and checked for effectiveness, safety, and reliability. The VA pledges to monitor AI’s impact to ensure it performs as expected without unintended consequences. Algorithms will be stress tested across representative datasets and approval processes will screen for safety issues. Third, AI models are secured from vulnerabilities and misuse. Technical controls will prevent unauthorized access or changes to AI systems. Audits will check for appropriate internal usage per policies. Continual patches and upgrades will maintain security. Fourth, the VA manages AI for fairness, avoiding bias. They will proactively assess datasets and algorithms for potential biases based on protected attributes like race, gender, or age. Biased outputs will be addressed through techniques such as data augmentation, reweighting, and algorithm tweaks. Fifth, transparency explains AI’s role in care. Documentation will detail an AI system’s data sources, methodology, testing, limitations, and integration with clinical workflows. Clinicians and patients will receive education on interpreting AI outputs. Finally, the VA pledges to closely monitor AI systems to sustain trust. The VA will establish oversight processes to quickly identify any declines in reliability or unfair impacts on subgroups. AI models will be retrained as needed based on incoming data patterns.
Each Trustworthy AI Framework principle connects to others in existing frameworks. The purpose principle aligns with human-centric AI focused on benefits. Effectiveness and safety link to technical robustness and risk management principles. Security maps to privacy protection principles. Fairness connects to principles of avoiding bias and discrimination. Transparency corresponds with accountable and explainable AI. Monitoring and accountability tie back to governance principles. Overall, the VA framework aims to guide ethical AI based on context. It offers a model for managing risks and building trust in health care AI.
Combining VA principles with high-reliability safety principles can ensure that AI benefits veterans. The leadership and culture aspects will drive commitment to trustworthy AI practices. Leaders will communicate the importance of responsible AI through words and actions. Culture surveys can assess baseline awareness of AI ethics issues to target education. AI security and fairness will be emphasized as safety critical. The process aspect will institute policies and procedures to uphold AI principles through the project lifecycle. For example, structured testing processes will validate safety. Measurement will collect data on principles like transparency and fairness. Dashboards can track metrics like explainability and biases. A patient-centered approach will incorporate veteran perspectives on AI through participatory design and advisory councils. They can give input on AI explainability and potential biases based on their diverse backgrounds.
Conclusions
Joint principles will lead to successful AI that improves care while proactively managing risks. Involve leaders to stress the necessity of eliminating biases. Build security into the AI development process. Co-design AI transparency features with end users. Closely monitor the impact of AI across safety, fairness, and other principles. Adhering to both Trustworthy AI and high reliability organizations principles will earn veterans’ confidence. Health care organizations like the VA can integrate ethical AI safely via established frameworks. With responsible design and implementation, AI’s potential to enhance care quality, safety, and access can be realized.
Acknowledgments
We would like to acknowledge Joshua Mueller, Theo Tiffney, John Zachary, and Gil Alterovitz for their excellent work creating the VA Trustworthy Principles. This material is the result of work supported by resources and the use of facilities at the James A. Haley Veterans’ Hospital.
1. Sahni NR, Carrus B. Artificial intelligence in U.S. health care delivery. N Engl J Med. 2023;389(4):348-358. doi:10.1056/NEJMra2204673
2. Borkowski AA, Jakey CE, Mastorides SM, et al. Applications of ChatGPT and large language models in medicine and health care: benefits and pitfalls. Fed Pract. 2023;40(6):170-173. doi:10.12788/fp.0386
3. Moyal-Smith R, Margo J, Maloney FL, et al. The patient safety adoption framework: a practical framework to bridge the know-do gap. J Patient Saf. 2023;19(4):243-248. doi:10.1097/PTS.0000000000001118
4. Isaacks DB, Anderson TM, Moore SC, Patterson W, Govindan S. High reliability organization principles improve VA workplace burnout: the Truman THRIVE2 model. Am J Med Qual. 2021;36(6):422-428. doi:10.1097/01.JMQ.0000735516.35323.97
5. Sculli GL, Pendley-Louis R, Neily J, et al. A high-reliability organization framework for health care: a multiyear implementation strategy and associated outcomes. J Patient Saf. 2022;18(1):64-70. doi:10.1097/PTS.0000000000000788
6. National Institute of Standards and Technology. AI risk management framework. Accessed January 2, 2024. https://www.nist.gov/itl/ai-risk-management-framework
7. Executive Office of the President, Office of Science and Technology Policy. Blueprint for an AI Bill of Rights. Accessed January 11, 2024. https://www.whitehouse.gov/ostp/ai-bill-of-rights
8. Executive Office of the President. Executive Order 13960: promoting the use of trustworthy artificial intelligence in the federal government. Fed Regist. 2020;89(236):78939-78943.
9. Biden JR. Executive Order on the safe, secure, and trustworthy development and use of artificial intelligence. Published October 30, 2023. Accessed January 11, 2024. https://www.whitehouse.gov/briefing-room/presidential-actions/2023/10/30/executive-order-on-the-safe-secure-and-trustworthy-development-and-use-of-artificial-intelligence/
10. US Department of Veterans Affairs. Trustworthy AI. Accessed January 11, 2024. https://department.va.gov/ai/trustworthy/
1. Sahni NR, Carrus B. Artificial intelligence in U.S. health care delivery. N Engl J Med. 2023;389(4):348-358. doi:10.1056/NEJMra2204673
2. Borkowski AA, Jakey CE, Mastorides SM, et al. Applications of ChatGPT and large language models in medicine and health care: benefits and pitfalls. Fed Pract. 2023;40(6):170-173. doi:10.12788/fp.0386
3. Moyal-Smith R, Margo J, Maloney FL, et al. The patient safety adoption framework: a practical framework to bridge the know-do gap. J Patient Saf. 2023;19(4):243-248. doi:10.1097/PTS.0000000000001118
4. Isaacks DB, Anderson TM, Moore SC, Patterson W, Govindan S. High reliability organization principles improve VA workplace burnout: the Truman THRIVE2 model. Am J Med Qual. 2021;36(6):422-428. doi:10.1097/01.JMQ.0000735516.35323.97
5. Sculli GL, Pendley-Louis R, Neily J, et al. A high-reliability organization framework for health care: a multiyear implementation strategy and associated outcomes. J Patient Saf. 2022;18(1):64-70. doi:10.1097/PTS.0000000000000788
6. National Institute of Standards and Technology. AI risk management framework. Accessed January 2, 2024. https://www.nist.gov/itl/ai-risk-management-framework
7. Executive Office of the President, Office of Science and Technology Policy. Blueprint for an AI Bill of Rights. Accessed January 11, 2024. https://www.whitehouse.gov/ostp/ai-bill-of-rights
8. Executive Office of the President. Executive Order 13960: promoting the use of trustworthy artificial intelligence in the federal government. Fed Regist. 2020;89(236):78939-78943.
9. Biden JR. Executive Order on the safe, secure, and trustworthy development and use of artificial intelligence. Published October 30, 2023. Accessed January 11, 2024. https://www.whitehouse.gov/briefing-room/presidential-actions/2023/10/30/executive-order-on-the-safe-secure-and-trustworthy-development-and-use-of-artificial-intelligence/
10. US Department of Veterans Affairs. Trustworthy AI. Accessed January 11, 2024. https://department.va.gov/ai/trustworthy/
Moving the Field FORWARD
As an organization, AGA has invested heavily in programs and initiatives to support the professional development of its members across career stages. This includes programs such as the AGA-AASLD Academic Skills Workshop (in which I was fortunate to participate in 2016), Women’s Leadership and Executive Leadership Conferences (with the Midwest Women in GI Regional Workshop taking place later this month), and the AGA Research Foundation Awards Program, which distributes over $2 million in funding annually to support promising early career and senior investigators.
AGA’s Fostering Opportunities Resulting in Workforce and Research Diversity (FORWARD) Program, which was first funded by the National Institutes of Health in 2018 and is focused on improving the diversity of the GI research workforce, is another shining example. Led by Dr. Byron Cryer and Dr. Sandra Quezada, the program recently welcomed its 3rd cohort of participants, including 14 mentees and 28 senior and near-peer mentors.
We are pleased to frequently highlight these programs in the pages of GI & Hepatology News, and hope you enjoy learning more about each of these initiatives in future issues.
In this month’s issue of GIHN, we highlight AGA’s newest Clinical Practice Guideline focused on management of pouchitis. We also report on the results of a recent RCT published in the New England Journal of Medicine demonstrating the efficacy of thalidomide as a treatment for recurrent bleeding due to small-intestinal angiodysplasia and summarize other key journal content impacting your clinical practice. In our February Member Spotlight, we feature Dr. Rajeev Jain of Texas Digestive Disease Consultants, a former AGA Governing Board member, and learn about his advocacy work to improve patient care and reduce physician burnout through insurance coverage and MOC reform. We hope you enjoy this, and all the exciting content included in our February issue!
Megan A. Adams, MD, JD, MSc
Editor-in-Chief
As an organization, AGA has invested heavily in programs and initiatives to support the professional development of its members across career stages. This includes programs such as the AGA-AASLD Academic Skills Workshop (in which I was fortunate to participate in 2016), Women’s Leadership and Executive Leadership Conferences (with the Midwest Women in GI Regional Workshop taking place later this month), and the AGA Research Foundation Awards Program, which distributes over $2 million in funding annually to support promising early career and senior investigators.
AGA’s Fostering Opportunities Resulting in Workforce and Research Diversity (FORWARD) Program, which was first funded by the National Institutes of Health in 2018 and is focused on improving the diversity of the GI research workforce, is another shining example. Led by Dr. Byron Cryer and Dr. Sandra Quezada, the program recently welcomed its 3rd cohort of participants, including 14 mentees and 28 senior and near-peer mentors.
We are pleased to frequently highlight these programs in the pages of GI & Hepatology News, and hope you enjoy learning more about each of these initiatives in future issues.
In this month’s issue of GIHN, we highlight AGA’s newest Clinical Practice Guideline focused on management of pouchitis. We also report on the results of a recent RCT published in the New England Journal of Medicine demonstrating the efficacy of thalidomide as a treatment for recurrent bleeding due to small-intestinal angiodysplasia and summarize other key journal content impacting your clinical practice. In our February Member Spotlight, we feature Dr. Rajeev Jain of Texas Digestive Disease Consultants, a former AGA Governing Board member, and learn about his advocacy work to improve patient care and reduce physician burnout through insurance coverage and MOC reform. We hope you enjoy this, and all the exciting content included in our February issue!
Megan A. Adams, MD, JD, MSc
Editor-in-Chief
As an organization, AGA has invested heavily in programs and initiatives to support the professional development of its members across career stages. This includes programs such as the AGA-AASLD Academic Skills Workshop (in which I was fortunate to participate in 2016), Women’s Leadership and Executive Leadership Conferences (with the Midwest Women in GI Regional Workshop taking place later this month), and the AGA Research Foundation Awards Program, which distributes over $2 million in funding annually to support promising early career and senior investigators.
AGA’s Fostering Opportunities Resulting in Workforce and Research Diversity (FORWARD) Program, which was first funded by the National Institutes of Health in 2018 and is focused on improving the diversity of the GI research workforce, is another shining example. Led by Dr. Byron Cryer and Dr. Sandra Quezada, the program recently welcomed its 3rd cohort of participants, including 14 mentees and 28 senior and near-peer mentors.
We are pleased to frequently highlight these programs in the pages of GI & Hepatology News, and hope you enjoy learning more about each of these initiatives in future issues.
In this month’s issue of GIHN, we highlight AGA’s newest Clinical Practice Guideline focused on management of pouchitis. We also report on the results of a recent RCT published in the New England Journal of Medicine demonstrating the efficacy of thalidomide as a treatment for recurrent bleeding due to small-intestinal angiodysplasia and summarize other key journal content impacting your clinical practice. In our February Member Spotlight, we feature Dr. Rajeev Jain of Texas Digestive Disease Consultants, a former AGA Governing Board member, and learn about his advocacy work to improve patient care and reduce physician burnout through insurance coverage and MOC reform. We hope you enjoy this, and all the exciting content included in our February issue!
Megan A. Adams, MD, JD, MSc
Editor-in-Chief
Top 5 Medications That Can Increase Blood Glucose Levels
It’s that time of the year, when social media is rife with many top 5 and top 10 lists. Let’s revisit some of the most commonly used medications known to increase glucose levels and look at some practical tips on overcoming these.
1. Glucocorticoids
Without a doubt, corticosteroids are at the top of the list when it comes to the potential for increasing blood glucose levels. High-dose glucocorticoid therapy is known to lead to new-onset diabetes (steroid-induced diabetes). Similarly, people with preexisting diabetes may notice significant worsening of glycemic control when they start on glucocorticoid therapy. The extent of glucose elevation depends on their glycemic status prior to initiation on steroids, the dose and duration of glucocorticoid therapy, and comorbid conditions, among other factors.
Management tip: For those with previously well-controlled diabetes or borderline diabetes, glucocorticoid-induced hyperglycemia may be managed by metformin with or without sulfonylurea therapy, especially if corticosteroid treatment is low-dose and for a shorter duration. However, for many individuals with preexisting poorly controlled diabetes or those initiated on high-dose corticosteroids, insulin therapy would perhaps be the treatment of choice. Glucocorticoid therapy generally leads to more pronounced postprandial hyperglycemia compared with fasting hyperglycemia; hence, the use of short-acting insulin therapy or perhaps NPH insulin in the morning might be a better option for many individuals. Dietary modification plays an important role in limiting the extent of postprandial hyperglycemia. Use of continuous glucose monitoring (CGM) devices may also be very helpful for understanding glycemic excursions and how to adjust insulin. In individuals for whom glucocorticoid therapy is tapered down, it is important to adjust the dose of medications with potential to cause hypoglycemia, such as insulin/sulfonylurea therapy, as the degree of hyperglycemia may decrease with decreased dose of the glucocorticoid therapy.
2. Antipsychotic Therapy
Antipsychotic medications can be obesogenic; between 15% and 72% of people who take second-generation antipsychotics experience weight gain of 7% or more. Increases in weight are not the only factor contributing to an elevated risk of developing type 2 diabetes. Antipsychotics are thought to cause downregulation of intracellular insulin signaling, leading to insulin resistance. At the same time, there seems to be a direct effect on the pancreatic beta cells. Antagonism of the dopamine D2, serotonin 5-HT2C, and muscarinic M3 receptors impairs beta-cell response to changes in blood glucose. In addition to the pharmacologic effects, cell culture experiments have shown that antipsychotics increase apoptosis of beta cells. Increased weight and concomitant development of type 2 diabetes is seen particularly in agents that exhibit high muscarinic M3 and histamine H1 receptor blockade. The effect on glucose metabolism is seen the most with agents such as clozapine, olanzapine, and haloperidol and the least with agents such as ziprasidone.
Management tip: Given the ongoing change in the understanding of increases in weight and their association with the risk of developing type 2 diabetes, a metabolically safer approach involves starting with medications that have a lower propensity for weight gain, and the partial agonists/third-generation antipsychotics as a family presently have the best overall data.
3. Thiazide Diuretics
Thiazide diuretics are commonly used for the management of hypertension and are associated with metabolic complications including hypokalemia; higher cholesterol, triglycerides, and other circulating lipids; and elevated glucose. It’s thought that the reduced potassium level occurring as a result of these medications might contribute to new-onset diabetes. The hypokalemia occurring from these medications is thought to lead to a decrease in insulin secretion and sensitivity, which is dose dependent. Studies show that the number needed to harm for chlorthalidone-induced diabetes is 29 over 1 year. There is believed to be no additional risk beyond 1 year.
Management tip: It’s important to monitor potassium levels for those initiated on thiazide diuretics. If hypokalemia occurs, it would be pertinent to correct the hypokalemia with potassium supplements to mitigate the risk for new-onset diabetes.
4. Statin Therapy
Statin therapy is thought to be associated with decreased insulin sensitivity and impairment in insulin secretion. The overall incidence of diabetes is pegged to be between 9% and 12% on statin therapy on the basis of meta-analysis studies, and higher on the basis of population-based studies. Overall, the estimated number needed to harm is: 1 out of every 255 patients on statin therapy for 4 years may develop new-onset diabetes. Compare this with the extremely strong evidence for number needed to treat being 39 for 5 years with statin therapy in patients with preexisting heart disease to prevent one occurrence of a nonfatal myocardial infarction.
Management tip: Although statins are associated with a small incident increase in the risk of developing diabetes, the potential benefits of using statin therapy for both primary and secondary prevention of cardiovascular disease significantly outweigh any of the potential risks associated with hyperglycemia. This is an important discussion to have with patients who are reluctant to use statin therapy because of the potential risk for new-onset diabetes as a side effect.
5. Beta-Blockers
Beta-blockers are another commonly used group of medications for managing hypertension, heart failure, coronary artery disease, and arrhythmia. Nonvasodilating beta-blockers such as metoprolol and atenolol are more likely to be associated with increases in A1c, mean plasma glucose, body weight, and triglycerides compared with vasodilating beta-blockers such as carvedilol, nebivolol, and labetalol (Bakris GL et al; Giugliano D et al). Similarly, studies have also shown that atenolol and metoprolol are associated with increased odds of hypoglycemia compared with carvedilol. People on beta-blockers may have masking of some of the symptoms of hypoglycemia, such as tremor, irritability, and palpitations, while other symptoms such as diaphoresis may remain unaffected on beta-blockers.
Management tip: Education on recognizing and managing hypoglycemia would be important when starting patients on beta-blockers if they are on preexisting insulin/sulfonylurea therapy. Use of CGM devices may be helpful if there is a high risk for hypoglycemia, especially as symptoms of hypoglycemia are often masked.
Honorable Mention
Several other medications — including antiretroviral therapy, tyrosine kinase inhibitors, mechanistic target of rapamycin (mTOR) inhibitors, immunosuppressants, and interferon alpha — are associated with worsening glycemic control and new-onset diabetes. Consider these agents’ effects on blood glucose, especially in people with an elevated risk of developing diabetes or those with preexisting diabetes, when prescribing.
A special mention should also be made of androgen deprivation therapy. These include treatment options like goserelin and leuprolide, which are gonadotropin-releasing hormone (GnRH) agonist therapies and are commonly used for prostate cancer management. Depending on the patient, these agents may be used for prolonged duration. Androgen deprivation therapy, by definition, decreases testosterone levels in men, thereby leading to worsening insulin resistance. Increase in fat mass and concomitant muscle wasting have been associated with the use of these medications; these, in turn, lead to peripheral insulin resistance. Nearly 1 out of every 5 men treated with long-term androgen deprivation therapy may be prone to developing worsening of A1c by 1% or more.
Management tip: Men on androgen deprivation therapy should be encouraged to participate in regular physical activity to reduce the burden of insulin resistance and to promote cardiovascular health.
Drug-induced diabetes is potentially reversible in many cases. Similarly, worsening of glycemic control due to medications in people with preexisting diabetes may also attenuate once the effect of the drug wears off. Blood glucose should be monitored on an ongoing basis so that diabetes medications can be adjusted. For some individuals, however, the worsening of glycemic status may be more chronic and may require long-term use of antihyperglycemic agents, especially if the benefits of continuation of the medication leading to hyperglycemia far exceed any potential risks.
Dr. Jain is Clinical Instructor, Department of Endocrinology, University of British Columbia; Endocrinologist, Fraser River Endocrinology, Vancouver, British Columbia, Canada. He disclosed ties with Abbott, Amgen, Boehringer Ingelheim, Dexcom, Eli Lilly, Janssen, Medtronic, Merck, and Novo Nordisk.
A version of this article appeared on Medscape.com.
It’s that time of the year, when social media is rife with many top 5 and top 10 lists. Let’s revisit some of the most commonly used medications known to increase glucose levels and look at some practical tips on overcoming these.
1. Glucocorticoids
Without a doubt, corticosteroids are at the top of the list when it comes to the potential for increasing blood glucose levels. High-dose glucocorticoid therapy is known to lead to new-onset diabetes (steroid-induced diabetes). Similarly, people with preexisting diabetes may notice significant worsening of glycemic control when they start on glucocorticoid therapy. The extent of glucose elevation depends on their glycemic status prior to initiation on steroids, the dose and duration of glucocorticoid therapy, and comorbid conditions, among other factors.
Management tip: For those with previously well-controlled diabetes or borderline diabetes, glucocorticoid-induced hyperglycemia may be managed by metformin with or without sulfonylurea therapy, especially if corticosteroid treatment is low-dose and for a shorter duration. However, for many individuals with preexisting poorly controlled diabetes or those initiated on high-dose corticosteroids, insulin therapy would perhaps be the treatment of choice. Glucocorticoid therapy generally leads to more pronounced postprandial hyperglycemia compared with fasting hyperglycemia; hence, the use of short-acting insulin therapy or perhaps NPH insulin in the morning might be a better option for many individuals. Dietary modification plays an important role in limiting the extent of postprandial hyperglycemia. Use of continuous glucose monitoring (CGM) devices may also be very helpful for understanding glycemic excursions and how to adjust insulin. In individuals for whom glucocorticoid therapy is tapered down, it is important to adjust the dose of medications with potential to cause hypoglycemia, such as insulin/sulfonylurea therapy, as the degree of hyperglycemia may decrease with decreased dose of the glucocorticoid therapy.
2. Antipsychotic Therapy
Antipsychotic medications can be obesogenic; between 15% and 72% of people who take second-generation antipsychotics experience weight gain of 7% or more. Increases in weight are not the only factor contributing to an elevated risk of developing type 2 diabetes. Antipsychotics are thought to cause downregulation of intracellular insulin signaling, leading to insulin resistance. At the same time, there seems to be a direct effect on the pancreatic beta cells. Antagonism of the dopamine D2, serotonin 5-HT2C, and muscarinic M3 receptors impairs beta-cell response to changes in blood glucose. In addition to the pharmacologic effects, cell culture experiments have shown that antipsychotics increase apoptosis of beta cells. Increased weight and concomitant development of type 2 diabetes is seen particularly in agents that exhibit high muscarinic M3 and histamine H1 receptor blockade. The effect on glucose metabolism is seen the most with agents such as clozapine, olanzapine, and haloperidol and the least with agents such as ziprasidone.
Management tip: Given the ongoing change in the understanding of increases in weight and their association with the risk of developing type 2 diabetes, a metabolically safer approach involves starting with medications that have a lower propensity for weight gain, and the partial agonists/third-generation antipsychotics as a family presently have the best overall data.
3. Thiazide Diuretics
Thiazide diuretics are commonly used for the management of hypertension and are associated with metabolic complications including hypokalemia; higher cholesterol, triglycerides, and other circulating lipids; and elevated glucose. It’s thought that the reduced potassium level occurring as a result of these medications might contribute to new-onset diabetes. The hypokalemia occurring from these medications is thought to lead to a decrease in insulin secretion and sensitivity, which is dose dependent. Studies show that the number needed to harm for chlorthalidone-induced diabetes is 29 over 1 year. There is believed to be no additional risk beyond 1 year.
Management tip: It’s important to monitor potassium levels for those initiated on thiazide diuretics. If hypokalemia occurs, it would be pertinent to correct the hypokalemia with potassium supplements to mitigate the risk for new-onset diabetes.
4. Statin Therapy
Statin therapy is thought to be associated with decreased insulin sensitivity and impairment in insulin secretion. The overall incidence of diabetes is pegged to be between 9% and 12% on statin therapy on the basis of meta-analysis studies, and higher on the basis of population-based studies. Overall, the estimated number needed to harm is: 1 out of every 255 patients on statin therapy for 4 years may develop new-onset diabetes. Compare this with the extremely strong evidence for number needed to treat being 39 for 5 years with statin therapy in patients with preexisting heart disease to prevent one occurrence of a nonfatal myocardial infarction.
Management tip: Although statins are associated with a small incident increase in the risk of developing diabetes, the potential benefits of using statin therapy for both primary and secondary prevention of cardiovascular disease significantly outweigh any of the potential risks associated with hyperglycemia. This is an important discussion to have with patients who are reluctant to use statin therapy because of the potential risk for new-onset diabetes as a side effect.
5. Beta-Blockers
Beta-blockers are another commonly used group of medications for managing hypertension, heart failure, coronary artery disease, and arrhythmia. Nonvasodilating beta-blockers such as metoprolol and atenolol are more likely to be associated with increases in A1c, mean plasma glucose, body weight, and triglycerides compared with vasodilating beta-blockers such as carvedilol, nebivolol, and labetalol (Bakris GL et al; Giugliano D et al). Similarly, studies have also shown that atenolol and metoprolol are associated with increased odds of hypoglycemia compared with carvedilol. People on beta-blockers may have masking of some of the symptoms of hypoglycemia, such as tremor, irritability, and palpitations, while other symptoms such as diaphoresis may remain unaffected on beta-blockers.
Management tip: Education on recognizing and managing hypoglycemia would be important when starting patients on beta-blockers if they are on preexisting insulin/sulfonylurea therapy. Use of CGM devices may be helpful if there is a high risk for hypoglycemia, especially as symptoms of hypoglycemia are often masked.
Honorable Mention
Several other medications — including antiretroviral therapy, tyrosine kinase inhibitors, mechanistic target of rapamycin (mTOR) inhibitors, immunosuppressants, and interferon alpha — are associated with worsening glycemic control and new-onset diabetes. Consider these agents’ effects on blood glucose, especially in people with an elevated risk of developing diabetes or those with preexisting diabetes, when prescribing.
A special mention should also be made of androgen deprivation therapy. These include treatment options like goserelin and leuprolide, which are gonadotropin-releasing hormone (GnRH) agonist therapies and are commonly used for prostate cancer management. Depending on the patient, these agents may be used for prolonged duration. Androgen deprivation therapy, by definition, decreases testosterone levels in men, thereby leading to worsening insulin resistance. Increase in fat mass and concomitant muscle wasting have been associated with the use of these medications; these, in turn, lead to peripheral insulin resistance. Nearly 1 out of every 5 men treated with long-term androgen deprivation therapy may be prone to developing worsening of A1c by 1% or more.
Management tip: Men on androgen deprivation therapy should be encouraged to participate in regular physical activity to reduce the burden of insulin resistance and to promote cardiovascular health.
Drug-induced diabetes is potentially reversible in many cases. Similarly, worsening of glycemic control due to medications in people with preexisting diabetes may also attenuate once the effect of the drug wears off. Blood glucose should be monitored on an ongoing basis so that diabetes medications can be adjusted. For some individuals, however, the worsening of glycemic status may be more chronic and may require long-term use of antihyperglycemic agents, especially if the benefits of continuation of the medication leading to hyperglycemia far exceed any potential risks.
Dr. Jain is Clinical Instructor, Department of Endocrinology, University of British Columbia; Endocrinologist, Fraser River Endocrinology, Vancouver, British Columbia, Canada. He disclosed ties with Abbott, Amgen, Boehringer Ingelheim, Dexcom, Eli Lilly, Janssen, Medtronic, Merck, and Novo Nordisk.
A version of this article appeared on Medscape.com.
It’s that time of the year, when social media is rife with many top 5 and top 10 lists. Let’s revisit some of the most commonly used medications known to increase glucose levels and look at some practical tips on overcoming these.
1. Glucocorticoids
Without a doubt, corticosteroids are at the top of the list when it comes to the potential for increasing blood glucose levels. High-dose glucocorticoid therapy is known to lead to new-onset diabetes (steroid-induced diabetes). Similarly, people with preexisting diabetes may notice significant worsening of glycemic control when they start on glucocorticoid therapy. The extent of glucose elevation depends on their glycemic status prior to initiation on steroids, the dose and duration of glucocorticoid therapy, and comorbid conditions, among other factors.
Management tip: For those with previously well-controlled diabetes or borderline diabetes, glucocorticoid-induced hyperglycemia may be managed by metformin with or without sulfonylurea therapy, especially if corticosteroid treatment is low-dose and for a shorter duration. However, for many individuals with preexisting poorly controlled diabetes or those initiated on high-dose corticosteroids, insulin therapy would perhaps be the treatment of choice. Glucocorticoid therapy generally leads to more pronounced postprandial hyperglycemia compared with fasting hyperglycemia; hence, the use of short-acting insulin therapy or perhaps NPH insulin in the morning might be a better option for many individuals. Dietary modification plays an important role in limiting the extent of postprandial hyperglycemia. Use of continuous glucose monitoring (CGM) devices may also be very helpful for understanding glycemic excursions and how to adjust insulin. In individuals for whom glucocorticoid therapy is tapered down, it is important to adjust the dose of medications with potential to cause hypoglycemia, such as insulin/sulfonylurea therapy, as the degree of hyperglycemia may decrease with decreased dose of the glucocorticoid therapy.
2. Antipsychotic Therapy
Antipsychotic medications can be obesogenic; between 15% and 72% of people who take second-generation antipsychotics experience weight gain of 7% or more. Increases in weight are not the only factor contributing to an elevated risk of developing type 2 diabetes. Antipsychotics are thought to cause downregulation of intracellular insulin signaling, leading to insulin resistance. At the same time, there seems to be a direct effect on the pancreatic beta cells. Antagonism of the dopamine D2, serotonin 5-HT2C, and muscarinic M3 receptors impairs beta-cell response to changes in blood glucose. In addition to the pharmacologic effects, cell culture experiments have shown that antipsychotics increase apoptosis of beta cells. Increased weight and concomitant development of type 2 diabetes is seen particularly in agents that exhibit high muscarinic M3 and histamine H1 receptor blockade. The effect on glucose metabolism is seen the most with agents such as clozapine, olanzapine, and haloperidol and the least with agents such as ziprasidone.
Management tip: Given the ongoing change in the understanding of increases in weight and their association with the risk of developing type 2 diabetes, a metabolically safer approach involves starting with medications that have a lower propensity for weight gain, and the partial agonists/third-generation antipsychotics as a family presently have the best overall data.
3. Thiazide Diuretics
Thiazide diuretics are commonly used for the management of hypertension and are associated with metabolic complications including hypokalemia; higher cholesterol, triglycerides, and other circulating lipids; and elevated glucose. It’s thought that the reduced potassium level occurring as a result of these medications might contribute to new-onset diabetes. The hypokalemia occurring from these medications is thought to lead to a decrease in insulin secretion and sensitivity, which is dose dependent. Studies show that the number needed to harm for chlorthalidone-induced diabetes is 29 over 1 year. There is believed to be no additional risk beyond 1 year.
Management tip: It’s important to monitor potassium levels for those initiated on thiazide diuretics. If hypokalemia occurs, it would be pertinent to correct the hypokalemia with potassium supplements to mitigate the risk for new-onset diabetes.
4. Statin Therapy
Statin therapy is thought to be associated with decreased insulin sensitivity and impairment in insulin secretion. The overall incidence of diabetes is pegged to be between 9% and 12% on statin therapy on the basis of meta-analysis studies, and higher on the basis of population-based studies. Overall, the estimated number needed to harm is: 1 out of every 255 patients on statin therapy for 4 years may develop new-onset diabetes. Compare this with the extremely strong evidence for number needed to treat being 39 for 5 years with statin therapy in patients with preexisting heart disease to prevent one occurrence of a nonfatal myocardial infarction.
Management tip: Although statins are associated with a small incident increase in the risk of developing diabetes, the potential benefits of using statin therapy for both primary and secondary prevention of cardiovascular disease significantly outweigh any of the potential risks associated with hyperglycemia. This is an important discussion to have with patients who are reluctant to use statin therapy because of the potential risk for new-onset diabetes as a side effect.
5. Beta-Blockers
Beta-blockers are another commonly used group of medications for managing hypertension, heart failure, coronary artery disease, and arrhythmia. Nonvasodilating beta-blockers such as metoprolol and atenolol are more likely to be associated with increases in A1c, mean plasma glucose, body weight, and triglycerides compared with vasodilating beta-blockers such as carvedilol, nebivolol, and labetalol (Bakris GL et al; Giugliano D et al). Similarly, studies have also shown that atenolol and metoprolol are associated with increased odds of hypoglycemia compared with carvedilol. People on beta-blockers may have masking of some of the symptoms of hypoglycemia, such as tremor, irritability, and palpitations, while other symptoms such as diaphoresis may remain unaffected on beta-blockers.
Management tip: Education on recognizing and managing hypoglycemia would be important when starting patients on beta-blockers if they are on preexisting insulin/sulfonylurea therapy. Use of CGM devices may be helpful if there is a high risk for hypoglycemia, especially as symptoms of hypoglycemia are often masked.
Honorable Mention
Several other medications — including antiretroviral therapy, tyrosine kinase inhibitors, mechanistic target of rapamycin (mTOR) inhibitors, immunosuppressants, and interferon alpha — are associated with worsening glycemic control and new-onset diabetes. Consider these agents’ effects on blood glucose, especially in people with an elevated risk of developing diabetes or those with preexisting diabetes, when prescribing.
A special mention should also be made of androgen deprivation therapy. These include treatment options like goserelin and leuprolide, which are gonadotropin-releasing hormone (GnRH) agonist therapies and are commonly used for prostate cancer management. Depending on the patient, these agents may be used for prolonged duration. Androgen deprivation therapy, by definition, decreases testosterone levels in men, thereby leading to worsening insulin resistance. Increase in fat mass and concomitant muscle wasting have been associated with the use of these medications; these, in turn, lead to peripheral insulin resistance. Nearly 1 out of every 5 men treated with long-term androgen deprivation therapy may be prone to developing worsening of A1c by 1% or more.
Management tip: Men on androgen deprivation therapy should be encouraged to participate in regular physical activity to reduce the burden of insulin resistance and to promote cardiovascular health.
Drug-induced diabetes is potentially reversible in many cases. Similarly, worsening of glycemic control due to medications in people with preexisting diabetes may also attenuate once the effect of the drug wears off. Blood glucose should be monitored on an ongoing basis so that diabetes medications can be adjusted. For some individuals, however, the worsening of glycemic status may be more chronic and may require long-term use of antihyperglycemic agents, especially if the benefits of continuation of the medication leading to hyperglycemia far exceed any potential risks.
Dr. Jain is Clinical Instructor, Department of Endocrinology, University of British Columbia; Endocrinologist, Fraser River Endocrinology, Vancouver, British Columbia, Canada. He disclosed ties with Abbott, Amgen, Boehringer Ingelheim, Dexcom, Eli Lilly, Janssen, Medtronic, Merck, and Novo Nordisk.
A version of this article appeared on Medscape.com.
The Potential Benefits of Dietary Changes in Psoriasis Patients
Psoriasis is a chronic inflammatory skin disease for which several lifestyle factors—smoking, alcohol use, and psychological stress—are associated with higher incidence and more severe disease.1-3 Diet also has been implicated as a factor that can affect psoriasis,4 and many patients have shown interest in possible dietary interventions to help their disease.5
In 2018, the National Psoriasis Foundation (NPF) presented dietary recommendations for patients based on results from a systematic review. From the available literature, only dietary weight reduction with hypocaloric diets in overweight or obese patients could be strongly recommended, and it has been proven that obesity is associated with worse psoriasis severity.6 Other more recent studies have shown that dietary modifications such as intermittent fasting and the ketogenic diet also led to weight loss and improved psoriasis severity in overweight patients; however, it is difficult to discern if the improvement was due to weight loss alone or if the dietary patterns themselves played a role.7,8 The paucity of well-designed studies evaluating the effects of other dietary changes has prevented further guidelines from being written. We propose that dietary patterns such as the Mediterranean diet (MeD) and vegan/vegetarian diets—even without strong data showing benefits in skin disease—may help to decrease systemic inflammation, improve gut dysbiosis, and help decrease the risk for cardiometabolic comorbidities that are associated with psoriasis.
Mediterranean Diet
The MeD is based on the dietary tendencies of inhabitants from the regions surrounding the Mediterranean Sea and is centered around nutrient-rich foods such as vegetables, olive oil, and legumes while limiting meat and dairy.9 The NPF recommended considering a trial of the MeD based on low-quality evidence.6 Observational studies have indicated that psoriasis patients are less likely to adhere to the MeD, but those who do have less severe disease.8 However, a search of PubMed articles indexed for MEDLINE using the terms Mediterranean diet and psoriasis yielded no prospective interventional studies. Given the association of the MeD with less severe disease, it is important to understand which specific foods in the MeD could be beneficial. Intake of omega-3 fatty acids, such as those found in fatty fish, are important for modulation of systemic inflammation.7 High intake of polyphenols—found in fruits and vegetables, extra-virgin olive oil, and wine—also have been implicated in improving inflammatory diseases due to potent antioxidant and anti-inflammatory properties. Individually, fruits, vegetables, whole grains, and sea fish have been associated with lowering C-reactive protein levels, which also is indicative of the benefits of these foods on systemic inflammation.7
Vegan/Vegetarian Diets
Although fruits, vegetables, legumes, and whole grains are a substantial component of the MeD, there are limited data on vegetarian or purely vegan plant-based diets. An observational study from the NPF found that only 48.4% (15/31) of patients on the MeD vs 69.0% (20/29) on a vegan diet reported a favorable skin response.5 Two case reports also have shown beneficial results of a strict vegan diet for psoriasis and psoriatic arthritis, where whole-food plant-based diets also improved joint symptoms.10-12 As with any diet, those who pursue a plant-based diet should strive to consume a variety of foods to avoid nutrient deficiencies. A recent systematic meta-analysis of 141 studies evaluated nutrient status of vegan and vegetarian diets compared to pescovegetarians and those who consume meat. All dietary patterns showed varying degrees of low levels of different nutrients.13 Of note, the researchers found that vitamin B12, vitamin D, iron, zinc, iodine, calcium, and docosahexaenoic acid were lower in plant-based diets. In contrast, folate; vitamins B1, B6, C, and E; polyunsaturated fatty acids; α-linolenic acid; and magnesium intake were higher. Those who consumed meat were at risk for inadequate intake of fiber, polyunsaturated fatty acids, α-linolenic acid, folate, vitamin E, calcium, magnesium, and vitamin D, though vitamin D intake was higher than in vegans/vegetarians.13 The results of this meta-analysis indicated the importance of educating patients on what constitutes a well-rounded, micronutrient-rich diet or appropriate supplementation for any diet.
Effects on Gut Microbiome
Any changes in diet can lead to alterations in the gut microbiome, which may impact skin disease, as evidence indicates a bidirectional relationship between gut and skin health.10 A metagenomic analysis of the gut microbiota in patients with untreated plaque psoriasis revealed a signature dysbiosis for which the researchers developed a psoriasis microbiota index, suggesting the gut microbiota may play a role in psoriasis pathophysiology.14 Research shows that both the MeD and vegan/vegetarian diets, which are relatively rich in fiber and omega-3 fatty acids and low in saturated fat and animal protein compared to many diets, cause increases in dietary fiber–metabolizing bacteria that produce short-chain fatty acids. These short-chain fatty acids improve gut epithelial integrity and alleviate both gut and systemic inflammation.10
The changes to the gut microbiome induced by a high-fat diet also are concerning. In contrast to the MeD or vegan/vegetarian diets, consumption of a high-fat diet induces alterations in the composition of the gut microbiota that in turn increase the release of proinflammatory cytokines and promote higher intestinal permeability.10 Similarly, high sugar consumption promotes increased intestinal permeability and shifts the gut microbiota to organisms that can rapidly utilize simple carbohydrates at the expense of other beneficial organisms, reducing bacterial diversity.15 The Western diet, which is notable for both high fat and high sugar content, is sometimes referred to as a proinflammatory diet and has been shown to worsen psoriasiformlike lesions in mice.16 Importantly, most research indicates that high fat and high sugar consumption appear to be more prevalent in psoriasis patients,8 but the type of fat consumed in the diet matters. The Western diet includes abundant saturated fat found in meat, dairy products, palm and coconut oils, and processed foods, as well as omega-6 fatty acids that are found in meat, poultry, and eggs. Saturated fat has been shown to promote helper T cell (TH17) accumulation in the skin, and omega-6 fatty acids serve as precursors to various inflammatory lipid mediators.4 This distinction of sources of fat between the Western diet and MeD is important in understanding the diets’ different effects on psoriasis and overall health. As previously discussed, the high intake of omega-3 acids in the MeD is one of the ways it may exert its anti-inflammatory benefits.7
Next Steps in Advising Psoriasis Patients
A major limitation of the data for MeD and vegan/vegetarian diets is limited randomized controlled trials evaluating the impact of these diets on psoriasis. Thus, dietary recommendations for psoriasis are not as strong as for other diseases for which more conclusive data exist.8 Although the data on diet and psoriasis are not definitive, perhaps dermatologists should shift the question from “Does this diet definitely improve psoriasis?” to “Does this diet definitely improve my patient’s health as a whole and maybe also their psoriasis?” For instance, the MeD has been shown to reduce the risk for type 2 diabetes mellitus and cardiovascular disease as well as to slow cognitive decline.17 Vegan/vegetarian diets focusing on whole vs processed foods have been shown to be highly effective in combatting obesity, type 2 diabetes mellitus, coronary artery disease including severe atherosclerosis, and hypertension.18 Psoriasis patients are at increased risk for many of the ailments that the MeD and plant-based diets protect against, making these diets potentially even more impactful than for someone without psoriasis.19 Dietary recommendations should still be made in conjunction with continuing traditional therapies for psoriasis and in consultation with the patient’s primary care physician and/or dietitian; however, rather than waiting for more randomized controlled trials before making health-promoting recommendations, what would be the downside of starting now? At worst, the dietary change decreases their risk for several metabolic conditions, and at best they may even see an improvement in their psoriasis.
- Naldi L, Chatenoud L, Linder D, et al. Cigarette smoking, body mass index, and stressful life events as risk factors for psoriasis: results from an Italian case–control study. J Invest Dermatol. 2005;125:61-67. doi:10.1111/j.0022-202X.2005.23681.x
- Armstrong AW, Harskamp CT, Dhillon JS, et al. Psoriasis and smoking: a systematic review and meta‐analysis. Br J Dermatol. 2014;170:304-314. doi:10.1111/bjd.12670
- Zhu K, Zhu C, Fan Y. Alcohol consumption and psoriatic risk: a meta‐analysis of case–control studies. J Dermatol. 2012;39:770-773. doi:10.1111/j.1346-8138.2012.01577.x
- Kanda N, Hoashi T, Saeki H. Nutrition and psoriasis. Int J Mol Sci. 2020;21:5405. doi:10.3390/ijms21155405
- Afifi L, Danesh MJ, Lee KM, et al. Dietary behaviors in psoriasis: patient-reported outcomes from a U.S. national survey. Dermatol Ther. 2017;7:227-242. doi:10.1007/s13555-017-0183-4
- Ford AR, Siegel M, Bagel J, et al. Dietary recommendations for adults with psoriasis or psoriatic arthritis from the medical board of the National Psoriasis Foundation: a systematic review. JAMA Dermatol. 2018;154:934. doi:10.1001/jamadermatol.2018.1412
- Duchnik E, Kruk J, Tuchowska A, et al. The impact of diet and physical activity on psoriasis: a narrative review of the current evidence. Nutrients. 2023;15:840. doi:10.3390/nu15040840
- Chung M, Bartholomew E, Yeroushalmi S, et al. Dietary intervention and supplements in the management of psoriasis: current perspectives. Psoriasis Targets Ther. 2022;12:151-176. doi:10.2147/PTT.S328581
- Mazza E, Ferro Y, Pujia R, et al. Mediterranean diet in healthy aging. J Nutr Health Aging. 2021;25:1076-1083. doi:10.1007/s12603-021-1675-6
- Flores-Balderas X, Peña-Peña M, Rada KM, et al. Beneficial effects of plant-based diets on skin health and inflammatory skin diseases. Nutrients. 2023;15:2842. doi:10.3390/nu15132842
- Bonjour M, Gabriel S, Valencia A, et al. Challenging case in clinical practice: prolonged water-only fasting followed by an exclusively whole-plant-food diet in the management of severe plaque psoriasis. Integr Complement Ther. 2022;28:85-87. doi:10.1089/ict.2022.29010.mbo
- Lewandowska M, Dunbar K, Kassam S. Managing psoriatic arthritis with a whole food plant-based diet: a case study. Am J Lifestyle Med. 2021;15:402-406. doi:10.1177/1559827621993435
- Neufingerl N, Eilander A. Nutrient intake and status in adults consuming plant-based diets compared to meat-eaters: a systematic review. Nutrients. 2021;14:29. doi:10.3390/nu14010029
- Dei-Cas I, Giliberto F, Luce L, et al. Metagenomic analysis of gut microbiota in non-treated plaque psoriasis patients stratified by disease severity: development of a new psoriasis-microbiome index. Sci Rep. 2020;10:12754. doi:10.1038/s41598-020-69537-3
- Satokari R. High intake of sugar and the balance between pro- and anti-inflammatory gut bacteria. Nutrients. 2020;12:1348. doi:10.3390/nu12051348
- Shi Z, Wu X, Santos Rocha C, et al. Short-term Western diet intake promotes IL-23–mediated skin and joint inflammation accompanied by changes to the gut microbiota in mice. J Invest Dermatol. 2021;141:1780-1791. doi:10.1016/j.jid.2020.11.032
- Romagnolo DF, Selmin OI. Mediterranean diet and prevention of chronic diseases. Nutr Today. 2017;52:208-222. doi:10.1097/NT.0000000000000228
- Tuso PJ, Ismail MH, Ha BP, et al. Nutritional update for physicians: plant-based diets. Perm J. 2013;17:61-66. doi:10.7812/TPP/12-085
- Parisi R, Symmons DPM, Griffiths CEM, et al. Global epidemiology of psoriasis: a systematic review of incidence and prevalence. J Invest Dermatol. 2013;133:377-385. doi:10.1038/jid.2012.339
Psoriasis is a chronic inflammatory skin disease for which several lifestyle factors—smoking, alcohol use, and psychological stress—are associated with higher incidence and more severe disease.1-3 Diet also has been implicated as a factor that can affect psoriasis,4 and many patients have shown interest in possible dietary interventions to help their disease.5
In 2018, the National Psoriasis Foundation (NPF) presented dietary recommendations for patients based on results from a systematic review. From the available literature, only dietary weight reduction with hypocaloric diets in overweight or obese patients could be strongly recommended, and it has been proven that obesity is associated with worse psoriasis severity.6 Other more recent studies have shown that dietary modifications such as intermittent fasting and the ketogenic diet also led to weight loss and improved psoriasis severity in overweight patients; however, it is difficult to discern if the improvement was due to weight loss alone or if the dietary patterns themselves played a role.7,8 The paucity of well-designed studies evaluating the effects of other dietary changes has prevented further guidelines from being written. We propose that dietary patterns such as the Mediterranean diet (MeD) and vegan/vegetarian diets—even without strong data showing benefits in skin disease—may help to decrease systemic inflammation, improve gut dysbiosis, and help decrease the risk for cardiometabolic comorbidities that are associated with psoriasis.
Mediterranean Diet
The MeD is based on the dietary tendencies of inhabitants from the regions surrounding the Mediterranean Sea and is centered around nutrient-rich foods such as vegetables, olive oil, and legumes while limiting meat and dairy.9 The NPF recommended considering a trial of the MeD based on low-quality evidence.6 Observational studies have indicated that psoriasis patients are less likely to adhere to the MeD, but those who do have less severe disease.8 However, a search of PubMed articles indexed for MEDLINE using the terms Mediterranean diet and psoriasis yielded no prospective interventional studies. Given the association of the MeD with less severe disease, it is important to understand which specific foods in the MeD could be beneficial. Intake of omega-3 fatty acids, such as those found in fatty fish, are important for modulation of systemic inflammation.7 High intake of polyphenols—found in fruits and vegetables, extra-virgin olive oil, and wine—also have been implicated in improving inflammatory diseases due to potent antioxidant and anti-inflammatory properties. Individually, fruits, vegetables, whole grains, and sea fish have been associated with lowering C-reactive protein levels, which also is indicative of the benefits of these foods on systemic inflammation.7
Vegan/Vegetarian Diets
Although fruits, vegetables, legumes, and whole grains are a substantial component of the MeD, there are limited data on vegetarian or purely vegan plant-based diets. An observational study from the NPF found that only 48.4% (15/31) of patients on the MeD vs 69.0% (20/29) on a vegan diet reported a favorable skin response.5 Two case reports also have shown beneficial results of a strict vegan diet for psoriasis and psoriatic arthritis, where whole-food plant-based diets also improved joint symptoms.10-12 As with any diet, those who pursue a plant-based diet should strive to consume a variety of foods to avoid nutrient deficiencies. A recent systematic meta-analysis of 141 studies evaluated nutrient status of vegan and vegetarian diets compared to pescovegetarians and those who consume meat. All dietary patterns showed varying degrees of low levels of different nutrients.13 Of note, the researchers found that vitamin B12, vitamin D, iron, zinc, iodine, calcium, and docosahexaenoic acid were lower in plant-based diets. In contrast, folate; vitamins B1, B6, C, and E; polyunsaturated fatty acids; α-linolenic acid; and magnesium intake were higher. Those who consumed meat were at risk for inadequate intake of fiber, polyunsaturated fatty acids, α-linolenic acid, folate, vitamin E, calcium, magnesium, and vitamin D, though vitamin D intake was higher than in vegans/vegetarians.13 The results of this meta-analysis indicated the importance of educating patients on what constitutes a well-rounded, micronutrient-rich diet or appropriate supplementation for any diet.
Effects on Gut Microbiome
Any changes in diet can lead to alterations in the gut microbiome, which may impact skin disease, as evidence indicates a bidirectional relationship between gut and skin health.10 A metagenomic analysis of the gut microbiota in patients with untreated plaque psoriasis revealed a signature dysbiosis for which the researchers developed a psoriasis microbiota index, suggesting the gut microbiota may play a role in psoriasis pathophysiology.14 Research shows that both the MeD and vegan/vegetarian diets, which are relatively rich in fiber and omega-3 fatty acids and low in saturated fat and animal protein compared to many diets, cause increases in dietary fiber–metabolizing bacteria that produce short-chain fatty acids. These short-chain fatty acids improve gut epithelial integrity and alleviate both gut and systemic inflammation.10
The changes to the gut microbiome induced by a high-fat diet also are concerning. In contrast to the MeD or vegan/vegetarian diets, consumption of a high-fat diet induces alterations in the composition of the gut microbiota that in turn increase the release of proinflammatory cytokines and promote higher intestinal permeability.10 Similarly, high sugar consumption promotes increased intestinal permeability and shifts the gut microbiota to organisms that can rapidly utilize simple carbohydrates at the expense of other beneficial organisms, reducing bacterial diversity.15 The Western diet, which is notable for both high fat and high sugar content, is sometimes referred to as a proinflammatory diet and has been shown to worsen psoriasiformlike lesions in mice.16 Importantly, most research indicates that high fat and high sugar consumption appear to be more prevalent in psoriasis patients,8 but the type of fat consumed in the diet matters. The Western diet includes abundant saturated fat found in meat, dairy products, palm and coconut oils, and processed foods, as well as omega-6 fatty acids that are found in meat, poultry, and eggs. Saturated fat has been shown to promote helper T cell (TH17) accumulation in the skin, and omega-6 fatty acids serve as precursors to various inflammatory lipid mediators.4 This distinction of sources of fat between the Western diet and MeD is important in understanding the diets’ different effects on psoriasis and overall health. As previously discussed, the high intake of omega-3 acids in the MeD is one of the ways it may exert its anti-inflammatory benefits.7
Next Steps in Advising Psoriasis Patients
A major limitation of the data for MeD and vegan/vegetarian diets is limited randomized controlled trials evaluating the impact of these diets on psoriasis. Thus, dietary recommendations for psoriasis are not as strong as for other diseases for which more conclusive data exist.8 Although the data on diet and psoriasis are not definitive, perhaps dermatologists should shift the question from “Does this diet definitely improve psoriasis?” to “Does this diet definitely improve my patient’s health as a whole and maybe also their psoriasis?” For instance, the MeD has been shown to reduce the risk for type 2 diabetes mellitus and cardiovascular disease as well as to slow cognitive decline.17 Vegan/vegetarian diets focusing on whole vs processed foods have been shown to be highly effective in combatting obesity, type 2 diabetes mellitus, coronary artery disease including severe atherosclerosis, and hypertension.18 Psoriasis patients are at increased risk for many of the ailments that the MeD and plant-based diets protect against, making these diets potentially even more impactful than for someone without psoriasis.19 Dietary recommendations should still be made in conjunction with continuing traditional therapies for psoriasis and in consultation with the patient’s primary care physician and/or dietitian; however, rather than waiting for more randomized controlled trials before making health-promoting recommendations, what would be the downside of starting now? At worst, the dietary change decreases their risk for several metabolic conditions, and at best they may even see an improvement in their psoriasis.
Psoriasis is a chronic inflammatory skin disease for which several lifestyle factors—smoking, alcohol use, and psychological stress—are associated with higher incidence and more severe disease.1-3 Diet also has been implicated as a factor that can affect psoriasis,4 and many patients have shown interest in possible dietary interventions to help their disease.5
In 2018, the National Psoriasis Foundation (NPF) presented dietary recommendations for patients based on results from a systematic review. From the available literature, only dietary weight reduction with hypocaloric diets in overweight or obese patients could be strongly recommended, and it has been proven that obesity is associated with worse psoriasis severity.6 Other more recent studies have shown that dietary modifications such as intermittent fasting and the ketogenic diet also led to weight loss and improved psoriasis severity in overweight patients; however, it is difficult to discern if the improvement was due to weight loss alone or if the dietary patterns themselves played a role.7,8 The paucity of well-designed studies evaluating the effects of other dietary changes has prevented further guidelines from being written. We propose that dietary patterns such as the Mediterranean diet (MeD) and vegan/vegetarian diets—even without strong data showing benefits in skin disease—may help to decrease systemic inflammation, improve gut dysbiosis, and help decrease the risk for cardiometabolic comorbidities that are associated with psoriasis.
Mediterranean Diet
The MeD is based on the dietary tendencies of inhabitants from the regions surrounding the Mediterranean Sea and is centered around nutrient-rich foods such as vegetables, olive oil, and legumes while limiting meat and dairy.9 The NPF recommended considering a trial of the MeD based on low-quality evidence.6 Observational studies have indicated that psoriasis patients are less likely to adhere to the MeD, but those who do have less severe disease.8 However, a search of PubMed articles indexed for MEDLINE using the terms Mediterranean diet and psoriasis yielded no prospective interventional studies. Given the association of the MeD with less severe disease, it is important to understand which specific foods in the MeD could be beneficial. Intake of omega-3 fatty acids, such as those found in fatty fish, are important for modulation of systemic inflammation.7 High intake of polyphenols—found in fruits and vegetables, extra-virgin olive oil, and wine—also have been implicated in improving inflammatory diseases due to potent antioxidant and anti-inflammatory properties. Individually, fruits, vegetables, whole grains, and sea fish have been associated with lowering C-reactive protein levels, which also is indicative of the benefits of these foods on systemic inflammation.7
Vegan/Vegetarian Diets
Although fruits, vegetables, legumes, and whole grains are a substantial component of the MeD, there are limited data on vegetarian or purely vegan plant-based diets. An observational study from the NPF found that only 48.4% (15/31) of patients on the MeD vs 69.0% (20/29) on a vegan diet reported a favorable skin response.5 Two case reports also have shown beneficial results of a strict vegan diet for psoriasis and psoriatic arthritis, where whole-food plant-based diets also improved joint symptoms.10-12 As with any diet, those who pursue a plant-based diet should strive to consume a variety of foods to avoid nutrient deficiencies. A recent systematic meta-analysis of 141 studies evaluated nutrient status of vegan and vegetarian diets compared to pescovegetarians and those who consume meat. All dietary patterns showed varying degrees of low levels of different nutrients.13 Of note, the researchers found that vitamin B12, vitamin D, iron, zinc, iodine, calcium, and docosahexaenoic acid were lower in plant-based diets. In contrast, folate; vitamins B1, B6, C, and E; polyunsaturated fatty acids; α-linolenic acid; and magnesium intake were higher. Those who consumed meat were at risk for inadequate intake of fiber, polyunsaturated fatty acids, α-linolenic acid, folate, vitamin E, calcium, magnesium, and vitamin D, though vitamin D intake was higher than in vegans/vegetarians.13 The results of this meta-analysis indicated the importance of educating patients on what constitutes a well-rounded, micronutrient-rich diet or appropriate supplementation for any diet.
Effects on Gut Microbiome
Any changes in diet can lead to alterations in the gut microbiome, which may impact skin disease, as evidence indicates a bidirectional relationship between gut and skin health.10 A metagenomic analysis of the gut microbiota in patients with untreated plaque psoriasis revealed a signature dysbiosis for which the researchers developed a psoriasis microbiota index, suggesting the gut microbiota may play a role in psoriasis pathophysiology.14 Research shows that both the MeD and vegan/vegetarian diets, which are relatively rich in fiber and omega-3 fatty acids and low in saturated fat and animal protein compared to many diets, cause increases in dietary fiber–metabolizing bacteria that produce short-chain fatty acids. These short-chain fatty acids improve gut epithelial integrity and alleviate both gut and systemic inflammation.10
The changes to the gut microbiome induced by a high-fat diet also are concerning. In contrast to the MeD or vegan/vegetarian diets, consumption of a high-fat diet induces alterations in the composition of the gut microbiota that in turn increase the release of proinflammatory cytokines and promote higher intestinal permeability.10 Similarly, high sugar consumption promotes increased intestinal permeability and shifts the gut microbiota to organisms that can rapidly utilize simple carbohydrates at the expense of other beneficial organisms, reducing bacterial diversity.15 The Western diet, which is notable for both high fat and high sugar content, is sometimes referred to as a proinflammatory diet and has been shown to worsen psoriasiformlike lesions in mice.16 Importantly, most research indicates that high fat and high sugar consumption appear to be more prevalent in psoriasis patients,8 but the type of fat consumed in the diet matters. The Western diet includes abundant saturated fat found in meat, dairy products, palm and coconut oils, and processed foods, as well as omega-6 fatty acids that are found in meat, poultry, and eggs. Saturated fat has been shown to promote helper T cell (TH17) accumulation in the skin, and omega-6 fatty acids serve as precursors to various inflammatory lipid mediators.4 This distinction of sources of fat between the Western diet and MeD is important in understanding the diets’ different effects on psoriasis and overall health. As previously discussed, the high intake of omega-3 acids in the MeD is one of the ways it may exert its anti-inflammatory benefits.7
Next Steps in Advising Psoriasis Patients
A major limitation of the data for MeD and vegan/vegetarian diets is limited randomized controlled trials evaluating the impact of these diets on psoriasis. Thus, dietary recommendations for psoriasis are not as strong as for other diseases for which more conclusive data exist.8 Although the data on diet and psoriasis are not definitive, perhaps dermatologists should shift the question from “Does this diet definitely improve psoriasis?” to “Does this diet definitely improve my patient’s health as a whole and maybe also their psoriasis?” For instance, the MeD has been shown to reduce the risk for type 2 diabetes mellitus and cardiovascular disease as well as to slow cognitive decline.17 Vegan/vegetarian diets focusing on whole vs processed foods have been shown to be highly effective in combatting obesity, type 2 diabetes mellitus, coronary artery disease including severe atherosclerosis, and hypertension.18 Psoriasis patients are at increased risk for many of the ailments that the MeD and plant-based diets protect against, making these diets potentially even more impactful than for someone without psoriasis.19 Dietary recommendations should still be made in conjunction with continuing traditional therapies for psoriasis and in consultation with the patient’s primary care physician and/or dietitian; however, rather than waiting for more randomized controlled trials before making health-promoting recommendations, what would be the downside of starting now? At worst, the dietary change decreases their risk for several metabolic conditions, and at best they may even see an improvement in their psoriasis.
- Naldi L, Chatenoud L, Linder D, et al. Cigarette smoking, body mass index, and stressful life events as risk factors for psoriasis: results from an Italian case–control study. J Invest Dermatol. 2005;125:61-67. doi:10.1111/j.0022-202X.2005.23681.x
- Armstrong AW, Harskamp CT, Dhillon JS, et al. Psoriasis and smoking: a systematic review and meta‐analysis. Br J Dermatol. 2014;170:304-314. doi:10.1111/bjd.12670
- Zhu K, Zhu C, Fan Y. Alcohol consumption and psoriatic risk: a meta‐analysis of case–control studies. J Dermatol. 2012;39:770-773. doi:10.1111/j.1346-8138.2012.01577.x
- Kanda N, Hoashi T, Saeki H. Nutrition and psoriasis. Int J Mol Sci. 2020;21:5405. doi:10.3390/ijms21155405
- Afifi L, Danesh MJ, Lee KM, et al. Dietary behaviors in psoriasis: patient-reported outcomes from a U.S. national survey. Dermatol Ther. 2017;7:227-242. doi:10.1007/s13555-017-0183-4
- Ford AR, Siegel M, Bagel J, et al. Dietary recommendations for adults with psoriasis or psoriatic arthritis from the medical board of the National Psoriasis Foundation: a systematic review. JAMA Dermatol. 2018;154:934. doi:10.1001/jamadermatol.2018.1412
- Duchnik E, Kruk J, Tuchowska A, et al. The impact of diet and physical activity on psoriasis: a narrative review of the current evidence. Nutrients. 2023;15:840. doi:10.3390/nu15040840
- Chung M, Bartholomew E, Yeroushalmi S, et al. Dietary intervention and supplements in the management of psoriasis: current perspectives. Psoriasis Targets Ther. 2022;12:151-176. doi:10.2147/PTT.S328581
- Mazza E, Ferro Y, Pujia R, et al. Mediterranean diet in healthy aging. J Nutr Health Aging. 2021;25:1076-1083. doi:10.1007/s12603-021-1675-6
- Flores-Balderas X, Peña-Peña M, Rada KM, et al. Beneficial effects of plant-based diets on skin health and inflammatory skin diseases. Nutrients. 2023;15:2842. doi:10.3390/nu15132842
- Bonjour M, Gabriel S, Valencia A, et al. Challenging case in clinical practice: prolonged water-only fasting followed by an exclusively whole-plant-food diet in the management of severe plaque psoriasis. Integr Complement Ther. 2022;28:85-87. doi:10.1089/ict.2022.29010.mbo
- Lewandowska M, Dunbar K, Kassam S. Managing psoriatic arthritis with a whole food plant-based diet: a case study. Am J Lifestyle Med. 2021;15:402-406. doi:10.1177/1559827621993435
- Neufingerl N, Eilander A. Nutrient intake and status in adults consuming plant-based diets compared to meat-eaters: a systematic review. Nutrients. 2021;14:29. doi:10.3390/nu14010029
- Dei-Cas I, Giliberto F, Luce L, et al. Metagenomic analysis of gut microbiota in non-treated plaque psoriasis patients stratified by disease severity: development of a new psoriasis-microbiome index. Sci Rep. 2020;10:12754. doi:10.1038/s41598-020-69537-3
- Satokari R. High intake of sugar and the balance between pro- and anti-inflammatory gut bacteria. Nutrients. 2020;12:1348. doi:10.3390/nu12051348
- Shi Z, Wu X, Santos Rocha C, et al. Short-term Western diet intake promotes IL-23–mediated skin and joint inflammation accompanied by changes to the gut microbiota in mice. J Invest Dermatol. 2021;141:1780-1791. doi:10.1016/j.jid.2020.11.032
- Romagnolo DF, Selmin OI. Mediterranean diet and prevention of chronic diseases. Nutr Today. 2017;52:208-222. doi:10.1097/NT.0000000000000228
- Tuso PJ, Ismail MH, Ha BP, et al. Nutritional update for physicians: plant-based diets. Perm J. 2013;17:61-66. doi:10.7812/TPP/12-085
- Parisi R, Symmons DPM, Griffiths CEM, et al. Global epidemiology of psoriasis: a systematic review of incidence and prevalence. J Invest Dermatol. 2013;133:377-385. doi:10.1038/jid.2012.339
- Naldi L, Chatenoud L, Linder D, et al. Cigarette smoking, body mass index, and stressful life events as risk factors for psoriasis: results from an Italian case–control study. J Invest Dermatol. 2005;125:61-67. doi:10.1111/j.0022-202X.2005.23681.x
- Armstrong AW, Harskamp CT, Dhillon JS, et al. Psoriasis and smoking: a systematic review and meta‐analysis. Br J Dermatol. 2014;170:304-314. doi:10.1111/bjd.12670
- Zhu K, Zhu C, Fan Y. Alcohol consumption and psoriatic risk: a meta‐analysis of case–control studies. J Dermatol. 2012;39:770-773. doi:10.1111/j.1346-8138.2012.01577.x
- Kanda N, Hoashi T, Saeki H. Nutrition and psoriasis. Int J Mol Sci. 2020;21:5405. doi:10.3390/ijms21155405
- Afifi L, Danesh MJ, Lee KM, et al. Dietary behaviors in psoriasis: patient-reported outcomes from a U.S. national survey. Dermatol Ther. 2017;7:227-242. doi:10.1007/s13555-017-0183-4
- Ford AR, Siegel M, Bagel J, et al. Dietary recommendations for adults with psoriasis or psoriatic arthritis from the medical board of the National Psoriasis Foundation: a systematic review. JAMA Dermatol. 2018;154:934. doi:10.1001/jamadermatol.2018.1412
- Duchnik E, Kruk J, Tuchowska A, et al. The impact of diet and physical activity on psoriasis: a narrative review of the current evidence. Nutrients. 2023;15:840. doi:10.3390/nu15040840
- Chung M, Bartholomew E, Yeroushalmi S, et al. Dietary intervention and supplements in the management of psoriasis: current perspectives. Psoriasis Targets Ther. 2022;12:151-176. doi:10.2147/PTT.S328581
- Mazza E, Ferro Y, Pujia R, et al. Mediterranean diet in healthy aging. J Nutr Health Aging. 2021;25:1076-1083. doi:10.1007/s12603-021-1675-6
- Flores-Balderas X, Peña-Peña M, Rada KM, et al. Beneficial effects of plant-based diets on skin health and inflammatory skin diseases. Nutrients. 2023;15:2842. doi:10.3390/nu15132842
- Bonjour M, Gabriel S, Valencia A, et al. Challenging case in clinical practice: prolonged water-only fasting followed by an exclusively whole-plant-food diet in the management of severe plaque psoriasis. Integr Complement Ther. 2022;28:85-87. doi:10.1089/ict.2022.29010.mbo
- Lewandowska M, Dunbar K, Kassam S. Managing psoriatic arthritis with a whole food plant-based diet: a case study. Am J Lifestyle Med. 2021;15:402-406. doi:10.1177/1559827621993435
- Neufingerl N, Eilander A. Nutrient intake and status in adults consuming plant-based diets compared to meat-eaters: a systematic review. Nutrients. 2021;14:29. doi:10.3390/nu14010029
- Dei-Cas I, Giliberto F, Luce L, et al. Metagenomic analysis of gut microbiota in non-treated plaque psoriasis patients stratified by disease severity: development of a new psoriasis-microbiome index. Sci Rep. 2020;10:12754. doi:10.1038/s41598-020-69537-3
- Satokari R. High intake of sugar and the balance between pro- and anti-inflammatory gut bacteria. Nutrients. 2020;12:1348. doi:10.3390/nu12051348
- Shi Z, Wu X, Santos Rocha C, et al. Short-term Western diet intake promotes IL-23–mediated skin and joint inflammation accompanied by changes to the gut microbiota in mice. J Invest Dermatol. 2021;141:1780-1791. doi:10.1016/j.jid.2020.11.032
- Romagnolo DF, Selmin OI. Mediterranean diet and prevention of chronic diseases. Nutr Today. 2017;52:208-222. doi:10.1097/NT.0000000000000228
- Tuso PJ, Ismail MH, Ha BP, et al. Nutritional update for physicians: plant-based diets. Perm J. 2013;17:61-66. doi:10.7812/TPP/12-085
- Parisi R, Symmons DPM, Griffiths CEM, et al. Global epidemiology of psoriasis: a systematic review of incidence and prevalence. J Invest Dermatol. 2013;133:377-385. doi:10.1038/jid.2012.339
Practice Points
- Psoriasis is affected by lifestyle factors such as diet, which is an area of interest for many patients.
- Low-calorie diets are strongly recommended for overweight/obese patients with psoriasis to improve their disease.
- Changes in dietary patterns, such as adopting a Mediterranean diet or a plant-based diet, also have shown promise.
Navigating Psoriasis Treatment Innovations
Psoriasis is a chronic autoimmune skin condition that affects approximately 2% to 4% of the US population and notably impacts overall quality of life.1,2 There is no cure for this long-lasting condition. Fortunately, recent developments in research have led to more targeted therapies, paving the way for a more promising transformative landscape of psoriasis management. Herein, we explore the most up-to-date advancements and developments in the realm of psoriasis care.
Emerging Systemic Therapies
Biologics are cutting-edge treatments available for moderate to severe plaque psoriasis, as IL-17A, IL-23, and tumor necrosis factor α (TNF-α) have been recognized as key targets.3
IL-17—Bimekizumab is a unique monoclonal antibody that inhibits the activity of both IL-17A and IL-17F cytokines.3 This treatment was approved by the US Food and Drug Administration (FDA) in October 2023 for patients with moderate to severe plaque psoriasis who are candidates for systemic therapy or phototherapy.4
Bimekizumab outperformed ustekinumab in the BE VIVID phase 3 trial, with 273 of 321 patients (85%) receiving bimekizumab vs 81 of 163 patients (50%) receiving ustekinumab experiencing at least 90% improvement in psoriasis area and severity index (PASI) score at week 16.4 In a 2020 observational study (PSO-BIO-REAL), the efficacy rate of skin clearance after 6 months of treatment with biologics was only 25% (1/4).5 Aside from moderate to severe plaque psoriasis, bimekizumab demonstrated notable improvement in patients with psoriatic arthritis who had inadequate response or intolerance to TNF-α inhibitors compared to a placebo group in the BE COMPLETE phase 3 trial.6
IL-23—Guselkumab, risankizumab, and tildrakizumab are injectable therapies approved by the FDA in 2017 for moderate to severe plaque psoriasis.3 They inhibit IL-23 signaling by targeting the p19 subunit in addition to sparing IL-12.3,7
A novel oral therapeutic peptide, JNJ-2113—the first oral IL-23 receptor antagonist peptide that blocks IL-23 signaling—has been developed, offering a new way to treat moderate to severe plaque psoriasis. Trial results from a phase 2 study (FRONTIER1) have supported JNJ-2113’s advancement into phase 3.7,8 Patients who received JNJ-2113 successfully achieved PASI75 in addition to surpassing PASI90 and PASI100 at greater proportions compared to placebo at week 16.7
The promising early results of JNJ-2113 provide patients with greater flexibility and convenience for treatment options to address the manifestations of psoriasis. Although a considerable number of patients with moderate to severe plaque psoriasis qualify for advanced therapies, a substantial proportion remain untreated. Introducing an oral route of medication administration may help overcome barriers to therapy access due to a greater preference for pills over injections.9
TNF-α Inhibitors—Adalimumab is a TNF-α inhibitor that is used to treat moderate to severe chronic plaque psoriasis in adults who are candidates for systemic phototherapy.1,10 However, one of the main barriers to initiating treatment has been cost. Biosimilars contribute to market competition, thus allowing the possibility of lower drug prices.10
There are 9 FDA-approved biosimilar products for adalimumab, with 2 having interchangeable designation. The first interchangeable biosimilar to enter the US market, adalimumab-adbm, became available in July 2023. In October 2023, adalimumab-afzb was granted interchangeable designation,11 which enables pharmacists to swiftly substitute brand products for lower-cost biosimilars, providing patients with equally safe and effective alternatives without the delay of involving the prescribing clinician.12 Pricing information indicates an initial 5% discount, which may later increase to 60%, from brand name adalimumab. Hopefully, reduced drug costs due to market competition will allow more patients to overcome barriers to therapy access.
IL-12/IL-23—Ustekinumab is a monoclonal antibody that targets IL-12 and IL-23. The FDA recently approved ustekinumab-auub as the first interchangeable ustekinumab biosimilar for the treatment of various inflammatory diseases, including moderate to severe plaque psoriasis and psoriatic arthritis.12,13 The approval of ustekinumab-auub expands therapeutic options for the treatment of diverse inflammatory diseases. As the first interchangeable biosimilar in its category, this development underscores the importance of biosimilars in providing effective and accessible treatment.12,14
Topical Innovations
In October 2023, the FDA approved an expanded indication for roflumilast cream 0.3% to treat children as young as 6 years for plaque psoriasis, even for use in intertriginous areas,15 which is a milestone given the lack of treatment options for the pediatric population because topical steroids, the most common treatment option for plaque psoriasis, can have safety concerns related to long-term use. With the advent of this steroid-free topical agent, pediatric patients have a safe and well-tolerated option for managing plaque psoriasis.16 This promising effort will now expand to trials in children as young as 2 years to test efficacy.16
Engel et al17 proposed a new algorithmic approach to the topical management of psoriasis with roflumilast cream and tapinarof cream as first-line treatments for mild disease due to their novelty in treating intertriginous areas, whereas traditional topical steroids in these areas would be inapt.17 The latest indication for roflumilast cream suggests that this proposed recommendation could be a promising and convenient enhancement to psoriasis management, potentially outperforming traditional topical corticosteroids.15,17
Final Thoughts
Innovative targeted therapies ranging from new biologic agents to broader applications of topical treatments hold the potential to transform conventional psoriasis management with greater efficacy and safety, which can help create a more effective and personalized approach with greater patient satisfaction, ultimately enhancing overall quality of life. The choice of treatment is dependent not only on the severity of the disease but also on accessibility considerations such as cost. Overall, these innovative therapies add substantial value to the treatment armamentarium for psoriasis.
- Li C, Sunhe Y, Zhou H, Dong W. Efficacy and safety evaluations of adalimumab biosimilars in the treatment of psoriasis. J Dermatolog Treat. 2023;34:2249145. doi:10.1080/09546634.2023.2249145
- Liu J, Thatiparthi A, Martin A, et al. Association between psoriasis and thyroid dysfunction among US adults in the 2009-2014 National Health and Nutrition Examination Survey [published online Mary 17, 2021]. J Am Acad Dermatol. 2022;86:897-899. doi:10.1016/j.jaad.2021.03.030
- Lee EB, Amin M, Bhutani T, et al. Emerging therapies in psoriasis: a systematic review. Cutis. 2018;101(3S):5-9.
- Reich K, Papp KA, Blauvelt A, et al. Bimekizumab versus ustekinumab for the treatment of moderate to severe plaque psoriasis (BE VIVID): efficacy and safety from a 52-week, multicentre, double-blind, active comparator and placebo-controlled phase 3 trial. Lancet. 2021;397:487-498. doi:10.1016/S0140-6736(21)00125-2
- Seneschal J, Lacour JP, Bewley A, et al. A multinational, prospective, observational study to estimate complete skin clearance in patients with moderate-to-severe plaque PSOriasis treated with BIOlogics in a REAL world setting (PSO-BIO-REAL) [published online June 8, 2020]. J Eur Acad Dermatol Venereol. 2020;34:2566-2573. doi:10.1111/jdv.16568
- Merola JF, Landewé R, McInnes IB, et al. Bimekizumab in patients with active psoriatic arthritis and previous inadequate response or intolerance to tumour necrosis factor-α inhibitors: a randomised, double-blind, placebo-controlled, phase 3 trial (BE COMPLETE)[published online December 6, 2022]. Lancet. 2023;401:38-48. doi:10.1016/S0140-6736(22)02303-0
- Janssen announces positive topline results for JNJ-2113—a novel, first and only oral IL-23 receptor antagonist peptide in development for moderate-to-severe plaque psoriasis. News release. Janssen Pharmaceutical Companies; July 4, 2023.
- Bissonnette R, Pinter A, Ferris L, et al. A Phase 2, randomized, placebo-controlled, dose-ranging study of oral JNJ-77242113 for the treatment of moderate-to-severe plaque psoriasis: FRONTIER 1. Abstract presented at: World Congress of Dermatology, July 3-8, 2023; Singapore.
- Xu Y, Sudharshan L, Hsu MA, et al. Patient preferences associated with therapies for psoriatic arthritis: a conjoint analysis. Am Health Drug Benefits. 2018;11:408-417.
- Maurelli M, Girolomoni G, Gisondi P. Cost per responder of adalimumab biosimilars versus methotrexate in patients with psoriasis: a real-life experience. J Dermatolog Treat. 2023;34:2218504. doi:10.1080/09546634.2023.2218504
- Food and Drug Administration/Center for Drug Evaluation and Research. Expiration of first interchangeable exclusivity (“FIE”) when section 351(l)(6) litigation ends prior to the submission of an application for interchangeability [memorandum]. Published October 3, 2023. Accessed January 18, 2024. https://www.fda.gov/media/173749/download
- US Food & Drug Administration. Biosimilar and interchangeable biologics: more treatment choices. Accessed January 18, 2024. https://www.fda.gov/consumers/consumer-updates/biosimilar-and-interchangeable-biologics-more-treatment-choices
- Chow V, Mytych DT, Das S, et al. Pharmacokinetic similarity of ABP 654, an ustekinumab biosimilar candidate: results from a randomized, double-blind study in healthy subjects [published online July 7, 2023]. Clin Pharmacol Drug Dev. 2023;12:863-873. doi:10.1002/cpdd.1301
- Wezlana (ustekinumab-auub) [prescribing information]. Published October 2023. Accessed January 18, 2024. www.accessdata.fda.gov/drugsatfda_docs/label/2023/761285s000,761331s000lbl.pdf
- ZORYVE (roflumilast) topical cream [prescribing information]. Westlake Village, CA: Arcutis Biotherapeutics. Revised October 2023. Accessed January 18, 2024. https://www.arcutis.com/wp-content/uploads/USPI-roflumilast-cream.pdf
- Lie E, Choi M, Wang SP, et al. Topical management of pediatric psoriasis: a review of new developments and existing therapies. Paediatr Drugs. 2024;26:9-18. doi:10.1007/s40272-023-00592-9
- Engel PV, Smith B, Javadi SS, et al. It is time to consider anew topical algorithm for psoriasis. J Am Acad Dermatol. 2023:S0190-9622(23)02906-7. doi:10.1016/j.jaad.2023.07.1048
Psoriasis is a chronic autoimmune skin condition that affects approximately 2% to 4% of the US population and notably impacts overall quality of life.1,2 There is no cure for this long-lasting condition. Fortunately, recent developments in research have led to more targeted therapies, paving the way for a more promising transformative landscape of psoriasis management. Herein, we explore the most up-to-date advancements and developments in the realm of psoriasis care.
Emerging Systemic Therapies
Biologics are cutting-edge treatments available for moderate to severe plaque psoriasis, as IL-17A, IL-23, and tumor necrosis factor α (TNF-α) have been recognized as key targets.3
IL-17—Bimekizumab is a unique monoclonal antibody that inhibits the activity of both IL-17A and IL-17F cytokines.3 This treatment was approved by the US Food and Drug Administration (FDA) in October 2023 for patients with moderate to severe plaque psoriasis who are candidates for systemic therapy or phototherapy.4
Bimekizumab outperformed ustekinumab in the BE VIVID phase 3 trial, with 273 of 321 patients (85%) receiving bimekizumab vs 81 of 163 patients (50%) receiving ustekinumab experiencing at least 90% improvement in psoriasis area and severity index (PASI) score at week 16.4 In a 2020 observational study (PSO-BIO-REAL), the efficacy rate of skin clearance after 6 months of treatment with biologics was only 25% (1/4).5 Aside from moderate to severe plaque psoriasis, bimekizumab demonstrated notable improvement in patients with psoriatic arthritis who had inadequate response or intolerance to TNF-α inhibitors compared to a placebo group in the BE COMPLETE phase 3 trial.6
IL-23—Guselkumab, risankizumab, and tildrakizumab are injectable therapies approved by the FDA in 2017 for moderate to severe plaque psoriasis.3 They inhibit IL-23 signaling by targeting the p19 subunit in addition to sparing IL-12.3,7
A novel oral therapeutic peptide, JNJ-2113—the first oral IL-23 receptor antagonist peptide that blocks IL-23 signaling—has been developed, offering a new way to treat moderate to severe plaque psoriasis. Trial results from a phase 2 study (FRONTIER1) have supported JNJ-2113’s advancement into phase 3.7,8 Patients who received JNJ-2113 successfully achieved PASI75 in addition to surpassing PASI90 and PASI100 at greater proportions compared to placebo at week 16.7
The promising early results of JNJ-2113 provide patients with greater flexibility and convenience for treatment options to address the manifestations of psoriasis. Although a considerable number of patients with moderate to severe plaque psoriasis qualify for advanced therapies, a substantial proportion remain untreated. Introducing an oral route of medication administration may help overcome barriers to therapy access due to a greater preference for pills over injections.9
TNF-α Inhibitors—Adalimumab is a TNF-α inhibitor that is used to treat moderate to severe chronic plaque psoriasis in adults who are candidates for systemic phototherapy.1,10 However, one of the main barriers to initiating treatment has been cost. Biosimilars contribute to market competition, thus allowing the possibility of lower drug prices.10
There are 9 FDA-approved biosimilar products for adalimumab, with 2 having interchangeable designation. The first interchangeable biosimilar to enter the US market, adalimumab-adbm, became available in July 2023. In October 2023, adalimumab-afzb was granted interchangeable designation,11 which enables pharmacists to swiftly substitute brand products for lower-cost biosimilars, providing patients with equally safe and effective alternatives without the delay of involving the prescribing clinician.12 Pricing information indicates an initial 5% discount, which may later increase to 60%, from brand name adalimumab. Hopefully, reduced drug costs due to market competition will allow more patients to overcome barriers to therapy access.
IL-12/IL-23—Ustekinumab is a monoclonal antibody that targets IL-12 and IL-23. The FDA recently approved ustekinumab-auub as the first interchangeable ustekinumab biosimilar for the treatment of various inflammatory diseases, including moderate to severe plaque psoriasis and psoriatic arthritis.12,13 The approval of ustekinumab-auub expands therapeutic options for the treatment of diverse inflammatory diseases. As the first interchangeable biosimilar in its category, this development underscores the importance of biosimilars in providing effective and accessible treatment.12,14
Topical Innovations
In October 2023, the FDA approved an expanded indication for roflumilast cream 0.3% to treat children as young as 6 years for plaque psoriasis, even for use in intertriginous areas,15 which is a milestone given the lack of treatment options for the pediatric population because topical steroids, the most common treatment option for plaque psoriasis, can have safety concerns related to long-term use. With the advent of this steroid-free topical agent, pediatric patients have a safe and well-tolerated option for managing plaque psoriasis.16 This promising effort will now expand to trials in children as young as 2 years to test efficacy.16
Engel et al17 proposed a new algorithmic approach to the topical management of psoriasis with roflumilast cream and tapinarof cream as first-line treatments for mild disease due to their novelty in treating intertriginous areas, whereas traditional topical steroids in these areas would be inapt.17 The latest indication for roflumilast cream suggests that this proposed recommendation could be a promising and convenient enhancement to psoriasis management, potentially outperforming traditional topical corticosteroids.15,17
Final Thoughts
Innovative targeted therapies ranging from new biologic agents to broader applications of topical treatments hold the potential to transform conventional psoriasis management with greater efficacy and safety, which can help create a more effective and personalized approach with greater patient satisfaction, ultimately enhancing overall quality of life. The choice of treatment is dependent not only on the severity of the disease but also on accessibility considerations such as cost. Overall, these innovative therapies add substantial value to the treatment armamentarium for psoriasis.
Psoriasis is a chronic autoimmune skin condition that affects approximately 2% to 4% of the US population and notably impacts overall quality of life.1,2 There is no cure for this long-lasting condition. Fortunately, recent developments in research have led to more targeted therapies, paving the way for a more promising transformative landscape of psoriasis management. Herein, we explore the most up-to-date advancements and developments in the realm of psoriasis care.
Emerging Systemic Therapies
Biologics are cutting-edge treatments available for moderate to severe plaque psoriasis, as IL-17A, IL-23, and tumor necrosis factor α (TNF-α) have been recognized as key targets.3
IL-17—Bimekizumab is a unique monoclonal antibody that inhibits the activity of both IL-17A and IL-17F cytokines.3 This treatment was approved by the US Food and Drug Administration (FDA) in October 2023 for patients with moderate to severe plaque psoriasis who are candidates for systemic therapy or phototherapy.4
Bimekizumab outperformed ustekinumab in the BE VIVID phase 3 trial, with 273 of 321 patients (85%) receiving bimekizumab vs 81 of 163 patients (50%) receiving ustekinumab experiencing at least 90% improvement in psoriasis area and severity index (PASI) score at week 16.4 In a 2020 observational study (PSO-BIO-REAL), the efficacy rate of skin clearance after 6 months of treatment with biologics was only 25% (1/4).5 Aside from moderate to severe plaque psoriasis, bimekizumab demonstrated notable improvement in patients with psoriatic arthritis who had inadequate response or intolerance to TNF-α inhibitors compared to a placebo group in the BE COMPLETE phase 3 trial.6
IL-23—Guselkumab, risankizumab, and tildrakizumab are injectable therapies approved by the FDA in 2017 for moderate to severe plaque psoriasis.3 They inhibit IL-23 signaling by targeting the p19 subunit in addition to sparing IL-12.3,7
A novel oral therapeutic peptide, JNJ-2113—the first oral IL-23 receptor antagonist peptide that blocks IL-23 signaling—has been developed, offering a new way to treat moderate to severe plaque psoriasis. Trial results from a phase 2 study (FRONTIER1) have supported JNJ-2113’s advancement into phase 3.7,8 Patients who received JNJ-2113 successfully achieved PASI75 in addition to surpassing PASI90 and PASI100 at greater proportions compared to placebo at week 16.7
The promising early results of JNJ-2113 provide patients with greater flexibility and convenience for treatment options to address the manifestations of psoriasis. Although a considerable number of patients with moderate to severe plaque psoriasis qualify for advanced therapies, a substantial proportion remain untreated. Introducing an oral route of medication administration may help overcome barriers to therapy access due to a greater preference for pills over injections.9
TNF-α Inhibitors—Adalimumab is a TNF-α inhibitor that is used to treat moderate to severe chronic plaque psoriasis in adults who are candidates for systemic phototherapy.1,10 However, one of the main barriers to initiating treatment has been cost. Biosimilars contribute to market competition, thus allowing the possibility of lower drug prices.10
There are 9 FDA-approved biosimilar products for adalimumab, with 2 having interchangeable designation. The first interchangeable biosimilar to enter the US market, adalimumab-adbm, became available in July 2023. In October 2023, adalimumab-afzb was granted interchangeable designation,11 which enables pharmacists to swiftly substitute brand products for lower-cost biosimilars, providing patients with equally safe and effective alternatives without the delay of involving the prescribing clinician.12 Pricing information indicates an initial 5% discount, which may later increase to 60%, from brand name adalimumab. Hopefully, reduced drug costs due to market competition will allow more patients to overcome barriers to therapy access.
IL-12/IL-23—Ustekinumab is a monoclonal antibody that targets IL-12 and IL-23. The FDA recently approved ustekinumab-auub as the first interchangeable ustekinumab biosimilar for the treatment of various inflammatory diseases, including moderate to severe plaque psoriasis and psoriatic arthritis.12,13 The approval of ustekinumab-auub expands therapeutic options for the treatment of diverse inflammatory diseases. As the first interchangeable biosimilar in its category, this development underscores the importance of biosimilars in providing effective and accessible treatment.12,14
Topical Innovations
In October 2023, the FDA approved an expanded indication for roflumilast cream 0.3% to treat children as young as 6 years for plaque psoriasis, even for use in intertriginous areas,15 which is a milestone given the lack of treatment options for the pediatric population because topical steroids, the most common treatment option for plaque psoriasis, can have safety concerns related to long-term use. With the advent of this steroid-free topical agent, pediatric patients have a safe and well-tolerated option for managing plaque psoriasis.16 This promising effort will now expand to trials in children as young as 2 years to test efficacy.16
Engel et al17 proposed a new algorithmic approach to the topical management of psoriasis with roflumilast cream and tapinarof cream as first-line treatments for mild disease due to their novelty in treating intertriginous areas, whereas traditional topical steroids in these areas would be inapt.17 The latest indication for roflumilast cream suggests that this proposed recommendation could be a promising and convenient enhancement to psoriasis management, potentially outperforming traditional topical corticosteroids.15,17
Final Thoughts
Innovative targeted therapies ranging from new biologic agents to broader applications of topical treatments hold the potential to transform conventional psoriasis management with greater efficacy and safety, which can help create a more effective and personalized approach with greater patient satisfaction, ultimately enhancing overall quality of life. The choice of treatment is dependent not only on the severity of the disease but also on accessibility considerations such as cost. Overall, these innovative therapies add substantial value to the treatment armamentarium for psoriasis.
- Li C, Sunhe Y, Zhou H, Dong W. Efficacy and safety evaluations of adalimumab biosimilars in the treatment of psoriasis. J Dermatolog Treat. 2023;34:2249145. doi:10.1080/09546634.2023.2249145
- Liu J, Thatiparthi A, Martin A, et al. Association between psoriasis and thyroid dysfunction among US adults in the 2009-2014 National Health and Nutrition Examination Survey [published online Mary 17, 2021]. J Am Acad Dermatol. 2022;86:897-899. doi:10.1016/j.jaad.2021.03.030
- Lee EB, Amin M, Bhutani T, et al. Emerging therapies in psoriasis: a systematic review. Cutis. 2018;101(3S):5-9.
- Reich K, Papp KA, Blauvelt A, et al. Bimekizumab versus ustekinumab for the treatment of moderate to severe plaque psoriasis (BE VIVID): efficacy and safety from a 52-week, multicentre, double-blind, active comparator and placebo-controlled phase 3 trial. Lancet. 2021;397:487-498. doi:10.1016/S0140-6736(21)00125-2
- Seneschal J, Lacour JP, Bewley A, et al. A multinational, prospective, observational study to estimate complete skin clearance in patients with moderate-to-severe plaque PSOriasis treated with BIOlogics in a REAL world setting (PSO-BIO-REAL) [published online June 8, 2020]. J Eur Acad Dermatol Venereol. 2020;34:2566-2573. doi:10.1111/jdv.16568
- Merola JF, Landewé R, McInnes IB, et al. Bimekizumab in patients with active psoriatic arthritis and previous inadequate response or intolerance to tumour necrosis factor-α inhibitors: a randomised, double-blind, placebo-controlled, phase 3 trial (BE COMPLETE)[published online December 6, 2022]. Lancet. 2023;401:38-48. doi:10.1016/S0140-6736(22)02303-0
- Janssen announces positive topline results for JNJ-2113—a novel, first and only oral IL-23 receptor antagonist peptide in development for moderate-to-severe plaque psoriasis. News release. Janssen Pharmaceutical Companies; July 4, 2023.
- Bissonnette R, Pinter A, Ferris L, et al. A Phase 2, randomized, placebo-controlled, dose-ranging study of oral JNJ-77242113 for the treatment of moderate-to-severe plaque psoriasis: FRONTIER 1. Abstract presented at: World Congress of Dermatology, July 3-8, 2023; Singapore.
- Xu Y, Sudharshan L, Hsu MA, et al. Patient preferences associated with therapies for psoriatic arthritis: a conjoint analysis. Am Health Drug Benefits. 2018;11:408-417.
- Maurelli M, Girolomoni G, Gisondi P. Cost per responder of adalimumab biosimilars versus methotrexate in patients with psoriasis: a real-life experience. J Dermatolog Treat. 2023;34:2218504. doi:10.1080/09546634.2023.2218504
- Food and Drug Administration/Center for Drug Evaluation and Research. Expiration of first interchangeable exclusivity (“FIE”) when section 351(l)(6) litigation ends prior to the submission of an application for interchangeability [memorandum]. Published October 3, 2023. Accessed January 18, 2024. https://www.fda.gov/media/173749/download
- US Food & Drug Administration. Biosimilar and interchangeable biologics: more treatment choices. Accessed January 18, 2024. https://www.fda.gov/consumers/consumer-updates/biosimilar-and-interchangeable-biologics-more-treatment-choices
- Chow V, Mytych DT, Das S, et al. Pharmacokinetic similarity of ABP 654, an ustekinumab biosimilar candidate: results from a randomized, double-blind study in healthy subjects [published online July 7, 2023]. Clin Pharmacol Drug Dev. 2023;12:863-873. doi:10.1002/cpdd.1301
- Wezlana (ustekinumab-auub) [prescribing information]. Published October 2023. Accessed January 18, 2024. www.accessdata.fda.gov/drugsatfda_docs/label/2023/761285s000,761331s000lbl.pdf
- ZORYVE (roflumilast) topical cream [prescribing information]. Westlake Village, CA: Arcutis Biotherapeutics. Revised October 2023. Accessed January 18, 2024. https://www.arcutis.com/wp-content/uploads/USPI-roflumilast-cream.pdf
- Lie E, Choi M, Wang SP, et al. Topical management of pediatric psoriasis: a review of new developments and existing therapies. Paediatr Drugs. 2024;26:9-18. doi:10.1007/s40272-023-00592-9
- Engel PV, Smith B, Javadi SS, et al. It is time to consider anew topical algorithm for psoriasis. J Am Acad Dermatol. 2023:S0190-9622(23)02906-7. doi:10.1016/j.jaad.2023.07.1048
- Li C, Sunhe Y, Zhou H, Dong W. Efficacy and safety evaluations of adalimumab biosimilars in the treatment of psoriasis. J Dermatolog Treat. 2023;34:2249145. doi:10.1080/09546634.2023.2249145
- Liu J, Thatiparthi A, Martin A, et al. Association between psoriasis and thyroid dysfunction among US adults in the 2009-2014 National Health and Nutrition Examination Survey [published online Mary 17, 2021]. J Am Acad Dermatol. 2022;86:897-899. doi:10.1016/j.jaad.2021.03.030
- Lee EB, Amin M, Bhutani T, et al. Emerging therapies in psoriasis: a systematic review. Cutis. 2018;101(3S):5-9.
- Reich K, Papp KA, Blauvelt A, et al. Bimekizumab versus ustekinumab for the treatment of moderate to severe plaque psoriasis (BE VIVID): efficacy and safety from a 52-week, multicentre, double-blind, active comparator and placebo-controlled phase 3 trial. Lancet. 2021;397:487-498. doi:10.1016/S0140-6736(21)00125-2
- Seneschal J, Lacour JP, Bewley A, et al. A multinational, prospective, observational study to estimate complete skin clearance in patients with moderate-to-severe plaque PSOriasis treated with BIOlogics in a REAL world setting (PSO-BIO-REAL) [published online June 8, 2020]. J Eur Acad Dermatol Venereol. 2020;34:2566-2573. doi:10.1111/jdv.16568
- Merola JF, Landewé R, McInnes IB, et al. Bimekizumab in patients with active psoriatic arthritis and previous inadequate response or intolerance to tumour necrosis factor-α inhibitors: a randomised, double-blind, placebo-controlled, phase 3 trial (BE COMPLETE)[published online December 6, 2022]. Lancet. 2023;401:38-48. doi:10.1016/S0140-6736(22)02303-0
- Janssen announces positive topline results for JNJ-2113—a novel, first and only oral IL-23 receptor antagonist peptide in development for moderate-to-severe plaque psoriasis. News release. Janssen Pharmaceutical Companies; July 4, 2023.
- Bissonnette R, Pinter A, Ferris L, et al. A Phase 2, randomized, placebo-controlled, dose-ranging study of oral JNJ-77242113 for the treatment of moderate-to-severe plaque psoriasis: FRONTIER 1. Abstract presented at: World Congress of Dermatology, July 3-8, 2023; Singapore.
- Xu Y, Sudharshan L, Hsu MA, et al. Patient preferences associated with therapies for psoriatic arthritis: a conjoint analysis. Am Health Drug Benefits. 2018;11:408-417.
- Maurelli M, Girolomoni G, Gisondi P. Cost per responder of adalimumab biosimilars versus methotrexate in patients with psoriasis: a real-life experience. J Dermatolog Treat. 2023;34:2218504. doi:10.1080/09546634.2023.2218504
- Food and Drug Administration/Center for Drug Evaluation and Research. Expiration of first interchangeable exclusivity (“FIE”) when section 351(l)(6) litigation ends prior to the submission of an application for interchangeability [memorandum]. Published October 3, 2023. Accessed January 18, 2024. https://www.fda.gov/media/173749/download
- US Food & Drug Administration. Biosimilar and interchangeable biologics: more treatment choices. Accessed January 18, 2024. https://www.fda.gov/consumers/consumer-updates/biosimilar-and-interchangeable-biologics-more-treatment-choices
- Chow V, Mytych DT, Das S, et al. Pharmacokinetic similarity of ABP 654, an ustekinumab biosimilar candidate: results from a randomized, double-blind study in healthy subjects [published online July 7, 2023]. Clin Pharmacol Drug Dev. 2023;12:863-873. doi:10.1002/cpdd.1301
- Wezlana (ustekinumab-auub) [prescribing information]. Published October 2023. Accessed January 18, 2024. www.accessdata.fda.gov/drugsatfda_docs/label/2023/761285s000,761331s000lbl.pdf
- ZORYVE (roflumilast) topical cream [prescribing information]. Westlake Village, CA: Arcutis Biotherapeutics. Revised October 2023. Accessed January 18, 2024. https://www.arcutis.com/wp-content/uploads/USPI-roflumilast-cream.pdf
- Lie E, Choi M, Wang SP, et al. Topical management of pediatric psoriasis: a review of new developments and existing therapies. Paediatr Drugs. 2024;26:9-18. doi:10.1007/s40272-023-00592-9
- Engel PV, Smith B, Javadi SS, et al. It is time to consider anew topical algorithm for psoriasis. J Am Acad Dermatol. 2023:S0190-9622(23)02906-7. doi:10.1016/j.jaad.2023.07.1048