User login
Skin Cancer Screening: The Paradox of Melanoma and Improved All-Cause Mortality
In April 2023, the US Preventive Services Task Force (USPSTF) issued a controversial recommendation that the current evidence is insufficient to assess the benefits vs harms of visual skin examination by clinicians for skin cancer screening in adolescents and adults who do not have signs or symptoms of skin cancer.1,2 This recommendation by the USPSTF has not changed in a quarter century,3 but a recent study described an interesting paradox that should trigger wide evaluation and debate.
Patel et al4 analyzed data from the National Cancer Institute’s Surveillance, Epidemiology, and End Results Program from January 2000 to December 2018 to identify adults with a diagnosis of first primary melanoma in situ (MIS). Overall mortality was then determined through the National Vital Statistics System, which provides cause-of-death information for all deaths in the United States. The authors found 137,872 patients who had 1—and only 1—MIS discovered over the observation period. These patients predominantly were White (96.7%), and the mean (SD) age at diagnosis was 61.9 (16.5) years. During 910,308 total person-years of follow-up (mean [SD], 6.6 [5.1] years), 893 (0.6%) patients died of melanoma and 17,327 (12.6%) died of any cause. The 15-year melanoma-specific standardized mortality rate (SMR) was 1.89 (95% CI, 1.77-2.02), yet the 15-year overall survival relative to matched population controls was 112.4% (95% CI, 112.0%-112.8%), thus all-cause SMR was significantly lower at 0.68 (95% CI, 0.67-0.7). Although MIS was associated with a small increase in cohort melanoma mortality, overall mortality was actually lower than in the general population.4
Patel et al4 did a further broader search that included an additional 18,379 patients who also experienced a second primary melanoma, of which 6751 (36.7%) were invasive and 11,628 (63.3%) were in situ, with a melanoma-specific survival of 98.2% (95% CI, 97.6%-98.5%). Yet relative all-cause survival was significantly higher at 126.7% (95% CI, 125.5%-128.0%). Even among patients in whom a second primary melanoma was invasive, melanoma-specific survival was reduced to 91.1% (95% CI, 90.0%-92.1%), but relative all-cause survival was 116.7% (95% CI, 115%-118.4%). These data in the overall cohort of 155,251 patients showed a discordance between melanoma mortality, which was 4.27-times higher than in the general population (SMR, 4.27; 95% CI, 4.07-4.48), and a lower risk for death from all causes that was approximately 27% lower than in the general population (SMR, 0.73; 95% CI, 0.72-0.74). The authors showed that their findings were not associated with socioeconomic status.4
The analysis by Patel et al4 is now the second study in the literature to show this discordant melanoma survival pattern. In an earlier Australian study of 2452 melanoma patients, Watts et al5 reported that melanoma detection during routine skin checks was associated with a 25% lower all-cause mortality (hazard ratio, 0.75; 95% CI, 0.63-0.90) but not melanoma-specific mortality after multivariable adjustment for a variety of factors including socioeconomic status.These analyses by 2 different groups of investigators have broad implications. Both groups suggested that the improved life span in melanoma patients may be due to health-seeking behavior, which has been defined as “any action undertaken by individuals who perceive themselves to have a health problem or to be ill for the purpose of finding an appropriate remedy.”6
Once treated for melanoma, it is clear that patients are likely to return at regular intervals for thorough full-body skin examinations, but this activity alone could not be responsible for improved all-cause mortality in the face of increased melanoma-specific mortality. It seems the authors are implying a broader concept of good health behavior, originally defined by MacKian7 as encompassing “activities undertaken to maintain good health, to prevent ill health, as well as dealing with any departure from a good state of health,” such as overt behavioral patterns, actions, and habits with the goal of maintenance, restoration, and improvement of one’s health. A variety of behaviors fall within such a definition including smoking cessation, decreased alcohol use, good diet, more physical activity, safe sexual behavior, scheduling physician visits, medication adherence, vaccination, and yes—screening examinations for health problems.8
The concept that individuals who are diagnosed with melanoma fall into a pattern of good health behavior is an interesting hypothesis that must remain speculative until the multiple aspects of good health behavior are rigorously studied. This concept coexists with the hypothesis of melanoma “overdiagnosis”—the idea that many melanomas are detected that will never lead to death.9 Both concepts deserve further analysis. Unquestionably, a randomized controlled trial could never recruit patients willing to undergo long-term untreated observation of their melanomas to test the hypothesis that their melanoma diagnosis would eventually lead to death. Furthermore, Patel et al4 do suggest that even MIS carries a small but measurable increased risk for death from the disease, which is not particularly supportive of the overdiagnosis hypothesis; however, analysis of the concept that improved individual health behavior is at least in part responsible for the first discovery of melanomas is certainly approachable. Here is the key question: Did the melanoma diagnosis trigger a sudden change in multiple aspects of health behavior that led to significant all-cause mortality benefits? The average age of the population studied by Patel et al4 was approximately 62 years. One wonders whether the consequences of a lifetime of established health behavior patterns can be rapidly modified—certainly possible but again remains to be proven by further studies.
Conversely, the alternative hypothesis is that discovery of MIS was the result of active pursuit of self-examination and screening procedures as part of individually ingrained good health behavior over a lifetime. Goodwin et al10 carried out a study in a sample of the Medicare population aged 69 to 90 years looking at men who had prostate cancer screening via prostate-specific antigen measurement and women who had undergone mammography in older age, compared to the contrast population who had not had these screening procedures. They tracked date of death in Medicare enrollment files. They identified 543,970 women and 362,753 men who were aged 69 to 90 years as of January 1, 2003. Patients were stratified by life expectancy based on age and comorbidity. Within each stratum, the patients with cancer screening had higher actual median survival than those who were not screened, with differences ranging from 1.7 to 2.1 years for women and 0.9 to 1.1 years for men.10 These results were not the result of lower prostate or breast cancer mortality. Rather, one surmises that other health factors yielded lower mortality in the screened cohorts.
A full-body skin examination is a time-consuming process. Patients who come to their physician for a routine annual physical don’t expect a skin examination and very few physicians have the time for a long detailed full-body skin examination. When the patient presents to a dermatologist for an examination, it often is because they have real concerns; for example, they may have had a family member who died of skin cancer, or the patient themself may have noticed a worrisome lesion. Patients, not physicians, are the drivers of skin cancer screening, a fact that often is dismissed by those who are not necessarily supportive of the practice.
In light of the findings of Patel et al,4 it is essential that the USPSTF reviews be reanalyzed to compare skin cancer–specific mortality, all-cause mortality, and lifespan in individuals who pursue skin cancer screening; the reanalysis also should not be exclusively limited to survival. With the advent of the immune checkpoint inhibitors, patients with metastatic melanoma are living much longer.11 The burden of living with metastatic cancer must be characterized and measured to have a complete picture and a valid analysis.
After the release of the USPSTF recommendation, there have been calls for large-scale studies to prove the benefits of skin cancer screening.12 Such studies may be valuable; however, if the hypothesis that overall healthy behavior as the major outcome determinant is substantiated, it may prove quite challenging to perform tests of association with specific interventions. It has been shown that skin cancer screening does lead to discovery of more melanomas,13 yet in light of the paradox described by Patel et al,4 it also is likely that causes of death other than melanoma impact overall mortality. Patients who pursue skin examinations may engage in multiple different health activities that are beneficial in the long term, making it difficult to analyze the specific benefit of skin cancer screening in isolation. It may prove difficult to ask patients to omit selected aspects of healthy behavior to try to prove the point. At this time, there is much more work to be done prior to offering opinions on the importance of skin cancer examination in isolation to improve overall health care. In the meantime, dermatologists owe it to our patients to continue to diligently pursue thorough and detailed skin examinations.
- US Preventive Services Task Force; Mangione CM, Barry MJ, et al. Screening for skin cancer: US Preventive Services Task Force recommendation statement. JAMA. 2023;329:1290-1295.
- Henrikson NB, Ivlev I, Blasi PR, et al. Skin cancer screening: updated evidence report and systematic review for the US Preventive Services Task Force. JAMA. 2023;329:1296-1307.
- US Preventive Services Task Force Guide to Clinical Preventive Services. 2nd ed. Agency for Healthcare Research and Quality; 1996.
- Patel VR, Roberson ML, Pignone MP, et al. Risk of mortality after a diagnosis of melanoma in situ. JAMA Dermatol. 2023;169:703-710.
- Watts CG, McLoughlin K, Goumas C, et al. Association between melanoma detected during routine skin checks and mortality. JAMA Dermatol. 2021;157:1425-1436.
- Chrisman NJ. The health seeking process: an approach to the natural history of illness. Cult Med Psychiatry. 1977;1:351-773.
- MacKian S. A review of health seeking behaviour: problems and prospects. health systems development programme. University of Manchester; 2003. Accessed January 19, 2024. https://assets.publishing.service.gov.uk/media/57a08d1de5274a27b200163d/05-03_health_seeking_behaviour.pdf
- Conner M, Norman P. Health behaviour: current issues and challenges. Psychol Health. 2017;32:895-906.
- Welch HG, Black WC. Overdiagnosis in cancer. J Natl Cancer Inst. 2010;102:605-613.
- Goodwin JS, Sheffield K, Li S, et al. Receipt of cancer screening is a predictor of life expectancy. J Gen Intern Med. 2016;11:1308-1314.
- Johnson DB, Nebhan CA, Moslehi JJ, et al. Immune-checkpoint inhibitors: long-term implications of toxicity. Nat Rev Clin Oncol. 2022;19:254-267.
- Adamson AS. The USPSTF statement on skin cancer screening—not a disappointment but an opportunity. JAMA Dermatol. 2023;159:579-581. doi:10.1001/jamadermatol.2023.0706
- Katalinic A, Eisemann N, Waldmann A. Skin cancer screening in Germany. documenting melanoma incidence and mortality from 2008 to 2013. Dtsch Arztebl Int. 2015;112:629-634.
In April 2023, the US Preventive Services Task Force (USPSTF) issued a controversial recommendation that the current evidence is insufficient to assess the benefits vs harms of visual skin examination by clinicians for skin cancer screening in adolescents and adults who do not have signs or symptoms of skin cancer.1,2 This recommendation by the USPSTF has not changed in a quarter century,3 but a recent study described an interesting paradox that should trigger wide evaluation and debate.
Patel et al4 analyzed data from the National Cancer Institute’s Surveillance, Epidemiology, and End Results Program from January 2000 to December 2018 to identify adults with a diagnosis of first primary melanoma in situ (MIS). Overall mortality was then determined through the National Vital Statistics System, which provides cause-of-death information for all deaths in the United States. The authors found 137,872 patients who had 1—and only 1—MIS discovered over the observation period. These patients predominantly were White (96.7%), and the mean (SD) age at diagnosis was 61.9 (16.5) years. During 910,308 total person-years of follow-up (mean [SD], 6.6 [5.1] years), 893 (0.6%) patients died of melanoma and 17,327 (12.6%) died of any cause. The 15-year melanoma-specific standardized mortality rate (SMR) was 1.89 (95% CI, 1.77-2.02), yet the 15-year overall survival relative to matched population controls was 112.4% (95% CI, 112.0%-112.8%), thus all-cause SMR was significantly lower at 0.68 (95% CI, 0.67-0.7). Although MIS was associated with a small increase in cohort melanoma mortality, overall mortality was actually lower than in the general population.4
Patel et al4 did a further broader search that included an additional 18,379 patients who also experienced a second primary melanoma, of which 6751 (36.7%) were invasive and 11,628 (63.3%) were in situ, with a melanoma-specific survival of 98.2% (95% CI, 97.6%-98.5%). Yet relative all-cause survival was significantly higher at 126.7% (95% CI, 125.5%-128.0%). Even among patients in whom a second primary melanoma was invasive, melanoma-specific survival was reduced to 91.1% (95% CI, 90.0%-92.1%), but relative all-cause survival was 116.7% (95% CI, 115%-118.4%). These data in the overall cohort of 155,251 patients showed a discordance between melanoma mortality, which was 4.27-times higher than in the general population (SMR, 4.27; 95% CI, 4.07-4.48), and a lower risk for death from all causes that was approximately 27% lower than in the general population (SMR, 0.73; 95% CI, 0.72-0.74). The authors showed that their findings were not associated with socioeconomic status.4
The analysis by Patel et al4 is now the second study in the literature to show this discordant melanoma survival pattern. In an earlier Australian study of 2452 melanoma patients, Watts et al5 reported that melanoma detection during routine skin checks was associated with a 25% lower all-cause mortality (hazard ratio, 0.75; 95% CI, 0.63-0.90) but not melanoma-specific mortality after multivariable adjustment for a variety of factors including socioeconomic status.These analyses by 2 different groups of investigators have broad implications. Both groups suggested that the improved life span in melanoma patients may be due to health-seeking behavior, which has been defined as “any action undertaken by individuals who perceive themselves to have a health problem or to be ill for the purpose of finding an appropriate remedy.”6
Once treated for melanoma, it is clear that patients are likely to return at regular intervals for thorough full-body skin examinations, but this activity alone could not be responsible for improved all-cause mortality in the face of increased melanoma-specific mortality. It seems the authors are implying a broader concept of good health behavior, originally defined by MacKian7 as encompassing “activities undertaken to maintain good health, to prevent ill health, as well as dealing with any departure from a good state of health,” such as overt behavioral patterns, actions, and habits with the goal of maintenance, restoration, and improvement of one’s health. A variety of behaviors fall within such a definition including smoking cessation, decreased alcohol use, good diet, more physical activity, safe sexual behavior, scheduling physician visits, medication adherence, vaccination, and yes—screening examinations for health problems.8
The concept that individuals who are diagnosed with melanoma fall into a pattern of good health behavior is an interesting hypothesis that must remain speculative until the multiple aspects of good health behavior are rigorously studied. This concept coexists with the hypothesis of melanoma “overdiagnosis”—the idea that many melanomas are detected that will never lead to death.9 Both concepts deserve further analysis. Unquestionably, a randomized controlled trial could never recruit patients willing to undergo long-term untreated observation of their melanomas to test the hypothesis that their melanoma diagnosis would eventually lead to death. Furthermore, Patel et al4 do suggest that even MIS carries a small but measurable increased risk for death from the disease, which is not particularly supportive of the overdiagnosis hypothesis; however, analysis of the concept that improved individual health behavior is at least in part responsible for the first discovery of melanomas is certainly approachable. Here is the key question: Did the melanoma diagnosis trigger a sudden change in multiple aspects of health behavior that led to significant all-cause mortality benefits? The average age of the population studied by Patel et al4 was approximately 62 years. One wonders whether the consequences of a lifetime of established health behavior patterns can be rapidly modified—certainly possible but again remains to be proven by further studies.
Conversely, the alternative hypothesis is that discovery of MIS was the result of active pursuit of self-examination and screening procedures as part of individually ingrained good health behavior over a lifetime. Goodwin et al10 carried out a study in a sample of the Medicare population aged 69 to 90 years looking at men who had prostate cancer screening via prostate-specific antigen measurement and women who had undergone mammography in older age, compared to the contrast population who had not had these screening procedures. They tracked date of death in Medicare enrollment files. They identified 543,970 women and 362,753 men who were aged 69 to 90 years as of January 1, 2003. Patients were stratified by life expectancy based on age and comorbidity. Within each stratum, the patients with cancer screening had higher actual median survival than those who were not screened, with differences ranging from 1.7 to 2.1 years for women and 0.9 to 1.1 years for men.10 These results were not the result of lower prostate or breast cancer mortality. Rather, one surmises that other health factors yielded lower mortality in the screened cohorts.
A full-body skin examination is a time-consuming process. Patients who come to their physician for a routine annual physical don’t expect a skin examination and very few physicians have the time for a long detailed full-body skin examination. When the patient presents to a dermatologist for an examination, it often is because they have real concerns; for example, they may have had a family member who died of skin cancer, or the patient themself may have noticed a worrisome lesion. Patients, not physicians, are the drivers of skin cancer screening, a fact that often is dismissed by those who are not necessarily supportive of the practice.
In light of the findings of Patel et al,4 it is essential that the USPSTF reviews be reanalyzed to compare skin cancer–specific mortality, all-cause mortality, and lifespan in individuals who pursue skin cancer screening; the reanalysis also should not be exclusively limited to survival. With the advent of the immune checkpoint inhibitors, patients with metastatic melanoma are living much longer.11 The burden of living with metastatic cancer must be characterized and measured to have a complete picture and a valid analysis.
After the release of the USPSTF recommendation, there have been calls for large-scale studies to prove the benefits of skin cancer screening.12 Such studies may be valuable; however, if the hypothesis that overall healthy behavior as the major outcome determinant is substantiated, it may prove quite challenging to perform tests of association with specific interventions. It has been shown that skin cancer screening does lead to discovery of more melanomas,13 yet in light of the paradox described by Patel et al,4 it also is likely that causes of death other than melanoma impact overall mortality. Patients who pursue skin examinations may engage in multiple different health activities that are beneficial in the long term, making it difficult to analyze the specific benefit of skin cancer screening in isolation. It may prove difficult to ask patients to omit selected aspects of healthy behavior to try to prove the point. At this time, there is much more work to be done prior to offering opinions on the importance of skin cancer examination in isolation to improve overall health care. In the meantime, dermatologists owe it to our patients to continue to diligently pursue thorough and detailed skin examinations.
In April 2023, the US Preventive Services Task Force (USPSTF) issued a controversial recommendation that the current evidence is insufficient to assess the benefits vs harms of visual skin examination by clinicians for skin cancer screening in adolescents and adults who do not have signs or symptoms of skin cancer.1,2 This recommendation by the USPSTF has not changed in a quarter century,3 but a recent study described an interesting paradox that should trigger wide evaluation and debate.
Patel et al4 analyzed data from the National Cancer Institute’s Surveillance, Epidemiology, and End Results Program from January 2000 to December 2018 to identify adults with a diagnosis of first primary melanoma in situ (MIS). Overall mortality was then determined through the National Vital Statistics System, which provides cause-of-death information for all deaths in the United States. The authors found 137,872 patients who had 1—and only 1—MIS discovered over the observation period. These patients predominantly were White (96.7%), and the mean (SD) age at diagnosis was 61.9 (16.5) years. During 910,308 total person-years of follow-up (mean [SD], 6.6 [5.1] years), 893 (0.6%) patients died of melanoma and 17,327 (12.6%) died of any cause. The 15-year melanoma-specific standardized mortality rate (SMR) was 1.89 (95% CI, 1.77-2.02), yet the 15-year overall survival relative to matched population controls was 112.4% (95% CI, 112.0%-112.8%), thus all-cause SMR was significantly lower at 0.68 (95% CI, 0.67-0.7). Although MIS was associated with a small increase in cohort melanoma mortality, overall mortality was actually lower than in the general population.4
Patel et al4 did a further broader search that included an additional 18,379 patients who also experienced a second primary melanoma, of which 6751 (36.7%) were invasive and 11,628 (63.3%) were in situ, with a melanoma-specific survival of 98.2% (95% CI, 97.6%-98.5%). Yet relative all-cause survival was significantly higher at 126.7% (95% CI, 125.5%-128.0%). Even among patients in whom a second primary melanoma was invasive, melanoma-specific survival was reduced to 91.1% (95% CI, 90.0%-92.1%), but relative all-cause survival was 116.7% (95% CI, 115%-118.4%). These data in the overall cohort of 155,251 patients showed a discordance between melanoma mortality, which was 4.27-times higher than in the general population (SMR, 4.27; 95% CI, 4.07-4.48), and a lower risk for death from all causes that was approximately 27% lower than in the general population (SMR, 0.73; 95% CI, 0.72-0.74). The authors showed that their findings were not associated with socioeconomic status.4
The analysis by Patel et al4 is now the second study in the literature to show this discordant melanoma survival pattern. In an earlier Australian study of 2452 melanoma patients, Watts et al5 reported that melanoma detection during routine skin checks was associated with a 25% lower all-cause mortality (hazard ratio, 0.75; 95% CI, 0.63-0.90) but not melanoma-specific mortality after multivariable adjustment for a variety of factors including socioeconomic status.These analyses by 2 different groups of investigators have broad implications. Both groups suggested that the improved life span in melanoma patients may be due to health-seeking behavior, which has been defined as “any action undertaken by individuals who perceive themselves to have a health problem or to be ill for the purpose of finding an appropriate remedy.”6
Once treated for melanoma, it is clear that patients are likely to return at regular intervals for thorough full-body skin examinations, but this activity alone could not be responsible for improved all-cause mortality in the face of increased melanoma-specific mortality. It seems the authors are implying a broader concept of good health behavior, originally defined by MacKian7 as encompassing “activities undertaken to maintain good health, to prevent ill health, as well as dealing with any departure from a good state of health,” such as overt behavioral patterns, actions, and habits with the goal of maintenance, restoration, and improvement of one’s health. A variety of behaviors fall within such a definition including smoking cessation, decreased alcohol use, good diet, more physical activity, safe sexual behavior, scheduling physician visits, medication adherence, vaccination, and yes—screening examinations for health problems.8
The concept that individuals who are diagnosed with melanoma fall into a pattern of good health behavior is an interesting hypothesis that must remain speculative until the multiple aspects of good health behavior are rigorously studied. This concept coexists with the hypothesis of melanoma “overdiagnosis”—the idea that many melanomas are detected that will never lead to death.9 Both concepts deserve further analysis. Unquestionably, a randomized controlled trial could never recruit patients willing to undergo long-term untreated observation of their melanomas to test the hypothesis that their melanoma diagnosis would eventually lead to death. Furthermore, Patel et al4 do suggest that even MIS carries a small but measurable increased risk for death from the disease, which is not particularly supportive of the overdiagnosis hypothesis; however, analysis of the concept that improved individual health behavior is at least in part responsible for the first discovery of melanomas is certainly approachable. Here is the key question: Did the melanoma diagnosis trigger a sudden change in multiple aspects of health behavior that led to significant all-cause mortality benefits? The average age of the population studied by Patel et al4 was approximately 62 years. One wonders whether the consequences of a lifetime of established health behavior patterns can be rapidly modified—certainly possible but again remains to be proven by further studies.
Conversely, the alternative hypothesis is that discovery of MIS was the result of active pursuit of self-examination and screening procedures as part of individually ingrained good health behavior over a lifetime. Goodwin et al10 carried out a study in a sample of the Medicare population aged 69 to 90 years looking at men who had prostate cancer screening via prostate-specific antigen measurement and women who had undergone mammography in older age, compared to the contrast population who had not had these screening procedures. They tracked date of death in Medicare enrollment files. They identified 543,970 women and 362,753 men who were aged 69 to 90 years as of January 1, 2003. Patients were stratified by life expectancy based on age and comorbidity. Within each stratum, the patients with cancer screening had higher actual median survival than those who were not screened, with differences ranging from 1.7 to 2.1 years for women and 0.9 to 1.1 years for men.10 These results were not the result of lower prostate or breast cancer mortality. Rather, one surmises that other health factors yielded lower mortality in the screened cohorts.
A full-body skin examination is a time-consuming process. Patients who come to their physician for a routine annual physical don’t expect a skin examination and very few physicians have the time for a long detailed full-body skin examination. When the patient presents to a dermatologist for an examination, it often is because they have real concerns; for example, they may have had a family member who died of skin cancer, or the patient themself may have noticed a worrisome lesion. Patients, not physicians, are the drivers of skin cancer screening, a fact that often is dismissed by those who are not necessarily supportive of the practice.
In light of the findings of Patel et al,4 it is essential that the USPSTF reviews be reanalyzed to compare skin cancer–specific mortality, all-cause mortality, and lifespan in individuals who pursue skin cancer screening; the reanalysis also should not be exclusively limited to survival. With the advent of the immune checkpoint inhibitors, patients with metastatic melanoma are living much longer.11 The burden of living with metastatic cancer must be characterized and measured to have a complete picture and a valid analysis.
After the release of the USPSTF recommendation, there have been calls for large-scale studies to prove the benefits of skin cancer screening.12 Such studies may be valuable; however, if the hypothesis that overall healthy behavior as the major outcome determinant is substantiated, it may prove quite challenging to perform tests of association with specific interventions. It has been shown that skin cancer screening does lead to discovery of more melanomas,13 yet in light of the paradox described by Patel et al,4 it also is likely that causes of death other than melanoma impact overall mortality. Patients who pursue skin examinations may engage in multiple different health activities that are beneficial in the long term, making it difficult to analyze the specific benefit of skin cancer screening in isolation. It may prove difficult to ask patients to omit selected aspects of healthy behavior to try to prove the point. At this time, there is much more work to be done prior to offering opinions on the importance of skin cancer examination in isolation to improve overall health care. In the meantime, dermatologists owe it to our patients to continue to diligently pursue thorough and detailed skin examinations.
- US Preventive Services Task Force; Mangione CM, Barry MJ, et al. Screening for skin cancer: US Preventive Services Task Force recommendation statement. JAMA. 2023;329:1290-1295.
- Henrikson NB, Ivlev I, Blasi PR, et al. Skin cancer screening: updated evidence report and systematic review for the US Preventive Services Task Force. JAMA. 2023;329:1296-1307.
- US Preventive Services Task Force Guide to Clinical Preventive Services. 2nd ed. Agency for Healthcare Research and Quality; 1996.
- Patel VR, Roberson ML, Pignone MP, et al. Risk of mortality after a diagnosis of melanoma in situ. JAMA Dermatol. 2023;169:703-710.
- Watts CG, McLoughlin K, Goumas C, et al. Association between melanoma detected during routine skin checks and mortality. JAMA Dermatol. 2021;157:1425-1436.
- Chrisman NJ. The health seeking process: an approach to the natural history of illness. Cult Med Psychiatry. 1977;1:351-773.
- MacKian S. A review of health seeking behaviour: problems and prospects. health systems development programme. University of Manchester; 2003. Accessed January 19, 2024. https://assets.publishing.service.gov.uk/media/57a08d1de5274a27b200163d/05-03_health_seeking_behaviour.pdf
- Conner M, Norman P. Health behaviour: current issues and challenges. Psychol Health. 2017;32:895-906.
- Welch HG, Black WC. Overdiagnosis in cancer. J Natl Cancer Inst. 2010;102:605-613.
- Goodwin JS, Sheffield K, Li S, et al. Receipt of cancer screening is a predictor of life expectancy. J Gen Intern Med. 2016;11:1308-1314.
- Johnson DB, Nebhan CA, Moslehi JJ, et al. Immune-checkpoint inhibitors: long-term implications of toxicity. Nat Rev Clin Oncol. 2022;19:254-267.
- Adamson AS. The USPSTF statement on skin cancer screening—not a disappointment but an opportunity. JAMA Dermatol. 2023;159:579-581. doi:10.1001/jamadermatol.2023.0706
- Katalinic A, Eisemann N, Waldmann A. Skin cancer screening in Germany. documenting melanoma incidence and mortality from 2008 to 2013. Dtsch Arztebl Int. 2015;112:629-634.
- US Preventive Services Task Force; Mangione CM, Barry MJ, et al. Screening for skin cancer: US Preventive Services Task Force recommendation statement. JAMA. 2023;329:1290-1295.
- Henrikson NB, Ivlev I, Blasi PR, et al. Skin cancer screening: updated evidence report and systematic review for the US Preventive Services Task Force. JAMA. 2023;329:1296-1307.
- US Preventive Services Task Force Guide to Clinical Preventive Services. 2nd ed. Agency for Healthcare Research and Quality; 1996.
- Patel VR, Roberson ML, Pignone MP, et al. Risk of mortality after a diagnosis of melanoma in situ. JAMA Dermatol. 2023;169:703-710.
- Watts CG, McLoughlin K, Goumas C, et al. Association between melanoma detected during routine skin checks and mortality. JAMA Dermatol. 2021;157:1425-1436.
- Chrisman NJ. The health seeking process: an approach to the natural history of illness. Cult Med Psychiatry. 1977;1:351-773.
- MacKian S. A review of health seeking behaviour: problems and prospects. health systems development programme. University of Manchester; 2003. Accessed January 19, 2024. https://assets.publishing.service.gov.uk/media/57a08d1de5274a27b200163d/05-03_health_seeking_behaviour.pdf
- Conner M, Norman P. Health behaviour: current issues and challenges. Psychol Health. 2017;32:895-906.
- Welch HG, Black WC. Overdiagnosis in cancer. J Natl Cancer Inst. 2010;102:605-613.
- Goodwin JS, Sheffield K, Li S, et al. Receipt of cancer screening is a predictor of life expectancy. J Gen Intern Med. 2016;11:1308-1314.
- Johnson DB, Nebhan CA, Moslehi JJ, et al. Immune-checkpoint inhibitors: long-term implications of toxicity. Nat Rev Clin Oncol. 2022;19:254-267.
- Adamson AS. The USPSTF statement on skin cancer screening—not a disappointment but an opportunity. JAMA Dermatol. 2023;159:579-581. doi:10.1001/jamadermatol.2023.0706
- Katalinic A, Eisemann N, Waldmann A. Skin cancer screening in Germany. documenting melanoma incidence and mortality from 2008 to 2013. Dtsch Arztebl Int. 2015;112:629-634.
Practice Points
- Screening for skin cancer often is performed at the patient’s request.
- Patients who want full-body skin examinations may exhibit other health-promoting behaviors.
- Studies claiming “overdiagnosis” of skin cancer have not previously evaluated all-cause mortality.
Sodium vs Potassium for Lowering Blood Pressure?
A pair of dueling editorials in the journal Hypertension debate whether our focus should be on sodium or its often neglected partner, potassium.
A meta-analysis of 85 trials showed a consistent and linear. It may also depend on where you live and whether your concern is treating individuals or implementing effective food policy.
The Case for Sodium Restriction
Stephen Juraschek, MD, PhD, of the Beth Israel Deaconess Medical Center, Boston, Massachusetts, co-author of one editorial, told me in a zoom interview that he believes his side of the debate clearly has the stronger argument. Of the two cations in question, there has been infinitely more ink spilled about sodium.
Studies such as INTERSALT, the DASH diet, and TOHP may be the most well-known, but there are many, many intervention studies of sodium restriction’s effect on blood pressure. A meta-analysis of 85 trials of showed a consistent and linear relationship between sodium reduction and blood pressure. In contrast, the evidence base for potassium is more limited and less consistent. There are half as many trials with potassium, and its ability to lower blood pressure may depend on how much sodium is present in the diet.
An outlier in the sodium restriction evidence base is the PURE study, which suggested that extreme sodium restriction could increase cardiovascular mortality, but the trial suffered from two potential issues. First, it used a single spot urine specimen to measure sodium rather than the generally accepted more accurate 24-hour urine collection. A reanalysis of the TOHP study using a spot urine rather than a 24-hour urine collection changed the relationship between sodium intake and mortality and possibly explained the U-shaped association observed in PURE. Second, PURE was an observational cohort and was prone to confounding, or in this case, reverse causation. Why did people who consumed very little salt have an increased risk for cardiovascular disease? It is very possible that people with a high risk for cardiovascular disease were told to consume less salt to begin with. Hence B led to A rather than A leading to B.
The debate on sodium restriction has been bitter at times. Opposing camps formed, and people took sides in the “salt wars.” A group of researchers, termed the Jackson 6, met and decided to end the controversy by running a randomized trial in US prisons (having discounted the options of long-term care homes and military bases). They detailed their plan in an editorial in Hypertension. The study never came to fruition for two reasons: the obvious ethical problems of experimenting on prisoners and the revelation of undisclosed salt industry funding.
More recent studies have mercifully been more conventional. The SSaSS study, a randomized controlled trial of a salt substitute, provided the cardiovascular outcomes data that many were waiting for. And CARDIA-SSBP, a cross-over randomized trial recently presented at the American Heart Association meeting, showed that reducing dietary sodium was on par with medication when it came to lowering blood pressure.
For Dr. Juraschek, the evidence is clear: “If you were going to choose one, I would say the weight of the evidence is still really heavily on the sodium side.”
The Case for Potassium Supplementation
The evidence for salt restriction notwithstanding, Swapnil Hiremath, MD, MPH, from the University of Ottawa, Ontario, Canada, argued in his editorial that potassium supplementation has gotten short shrift. Though he admits the studies for potassium supplementation have been smaller and sometimes rely on observational evidence, the evidence is there. In the distal convoluted tubule, the sodium chloride cotransporter (NCC), aka the potassium switch, is turned on by low potassium levels and leads to sodium reabsorption by the kidney even in settings of high sodium intake (Figure). To nonnephrologists, renal physiology may be a black box. But if you quickly brush up on the mechanism of action of thiazide diuretics, the preceding descriptor will make more sense.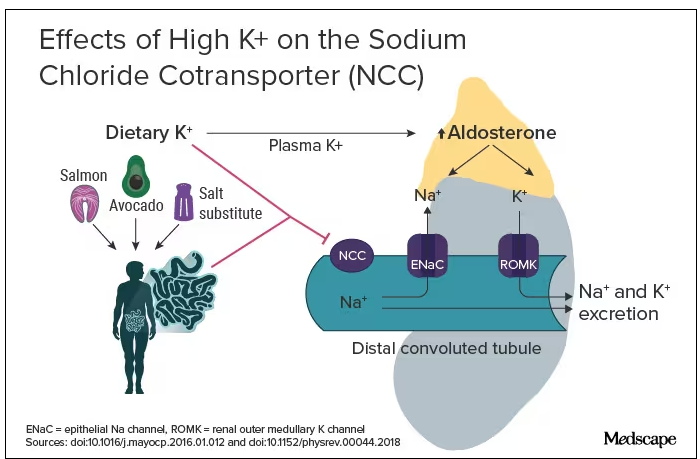
Dr. Hiremath points out that the DASH diet study also got patients to increase their potassium intake by eating more fruits and vegetables. Furthermore, the SSaSS study tested a salt substitute that was 25% potassium (and 75% sodium).
How much blood pressure lowering is due to sodium restriction vs potassium supplementation is a complex question because lowering sodium intake will invariably lead to more potassium intake. “It’s very hard to untangle the relationship,” Dr. Hiremath said in an interview. “It’s sort of synergistic but it’s not completely additive. It’s not as if you add four and four and get eight.” But he maintains there is more evidence regarding the benefit of potassium supplementation than many realize.
Realistic Diets and Taste Issues
“We know that increasing potassium, decreasing sodium is useful. The question is how do we do that?” says Dr. Hiremath. Should we encourage fruit and vegetable consumption in a healthy diet, give potassium supplements, or encourage the use of low-sodium salt substitutes?
Recommending a healthier diet with more fruits and vegetables is a no-brainer. But getting people to do it is hard. In a world where fruit is more expensive than junk food is, economic realities may drive food choice regardless of our best efforts. The 4700 mg of potassium in the DASH eating plan is the equivalent of eleven bananas daily; although not impossible, it would require a substantive shift in eating patterns for most people.
Given that we prescribe iron, vitamin B12, calcium, and vitamin D to patients who need them, why not potassium tablets to help with blood pressure? Granted, there are concerns about inducing hyperkalemia. Also, why not just prescribe a proven anti-hypertensive, such as ramipril, which has the added benefit of helping with renal protection or cardiac remodeling? Dr. Hiremath points out that patients are far less reluctant to take dietary supplements. Medication is something you take when sick. A supplement is seen as “natural” and “healthy” and might be more attractive to people resistant to prescription meds.
Another drawback of oral potassium supplementation is taste. In a Consumer Reports taste test, potassium chloride fared poorly. It was bitter and had a metallic aftertaste. At least one tester wouldn’t ever consume it again. Potassium citrate is slightly more palpable.
Salt substitutes, like the 75:25 ratio of sodium to potassium used in SSaSS, may be as high as you can go for potassium in any low-sodium salt alternative. If you go any higher than that, the taste will just turn people off, suggests Dr. Hiremath.
But SsaSS, which was done in China, may not be relevant to North America. In China, most sodium is added during cooking at home, and the consumption of processed foods is low. For the typical North American, roughly three quarters of the sodium eaten is added to their food by someone else; only about 15% is added during cooking at home or at the dinner table. If you aren’t someone who cooks, buying a salt substitute is probably not going to have much impact.
Given that reality, Dr. Juraschek thinks we need to target the sodium in processed foods. “There’s just so much sodium in so many products,” he says. “When you think about public policy, it’s most expeditious for there to be more regulation about how much is added to our food supply vs trying to get people to consume eight to 12 servings of fruit.”
No Salt War Here
Despite their different editorial takes, Dr. Hiremath and Dr. Juraschek largely agree on the broad strokes of the problem. This isn’t X (or Twitter) after all. Potassium supplementation may be useful in some parts of the world but may not address the underlying problem in countries where processed foods are the source of most dietary sodium.
The CARDIA-SSBP trial showed that a very low–sodium diet had the same blood pressure–lowering effect as a first-line antihypertensive, but most people will not be able to limit themselves to 500 mg of dietary sodium per day. In CARDIA-SSBP, just as in DASH, participants were provided with meals from study kitchens. They were not just told to eat less salt, which would almost certainly have failed.
“We should aim for stuff that is practical and doable rather than aim for stuff that cannot be done,” according to Dr. Hiremath. Whether that should be salt substitutes or policy change may depend on which part of the planet you live on.
One recent positive change may herald the beginning of a policy change, at least in the United States. In March 2023, the US Food and Drug Administration proposed a rule change to allow salt substitutes to be labeled as salt. This would make it easier for food manufacturers to swap out sodium chloride for a low-sodium alternative and reduce the amount of sodium in the US diet without having a large impact on taste and consumer uptake. Both Dr. Hiremath and Dr. Juraschek agree that it may not be enough on its own but that it’s a start.
Christopher Labos is a cardiologist with a degree in epidemiology. He spends most of his time doing things that he doesn’t get paid for, like research, teaching, and podcasting. Occasionally, he finds time to practice cardiology to pay the rent. He realizes that half of his research findings will be disproved in 5 years; he just doesn’t know which half. He is a regular contributor to the Montreal Gazette, CJAD radio, and CTV television in Montreal, and is host of the award-winning podcast The Body of Evidence.
A version of this article appeared on Medscape.com.
A pair of dueling editorials in the journal Hypertension debate whether our focus should be on sodium or its often neglected partner, potassium.
A meta-analysis of 85 trials showed a consistent and linear. It may also depend on where you live and whether your concern is treating individuals or implementing effective food policy.
The Case for Sodium Restriction
Stephen Juraschek, MD, PhD, of the Beth Israel Deaconess Medical Center, Boston, Massachusetts, co-author of one editorial, told me in a zoom interview that he believes his side of the debate clearly has the stronger argument. Of the two cations in question, there has been infinitely more ink spilled about sodium.
Studies such as INTERSALT, the DASH diet, and TOHP may be the most well-known, but there are many, many intervention studies of sodium restriction’s effect on blood pressure. A meta-analysis of 85 trials of showed a consistent and linear relationship between sodium reduction and blood pressure. In contrast, the evidence base for potassium is more limited and less consistent. There are half as many trials with potassium, and its ability to lower blood pressure may depend on how much sodium is present in the diet.
An outlier in the sodium restriction evidence base is the PURE study, which suggested that extreme sodium restriction could increase cardiovascular mortality, but the trial suffered from two potential issues. First, it used a single spot urine specimen to measure sodium rather than the generally accepted more accurate 24-hour urine collection. A reanalysis of the TOHP study using a spot urine rather than a 24-hour urine collection changed the relationship between sodium intake and mortality and possibly explained the U-shaped association observed in PURE. Second, PURE was an observational cohort and was prone to confounding, or in this case, reverse causation. Why did people who consumed very little salt have an increased risk for cardiovascular disease? It is very possible that people with a high risk for cardiovascular disease were told to consume less salt to begin with. Hence B led to A rather than A leading to B.
The debate on sodium restriction has been bitter at times. Opposing camps formed, and people took sides in the “salt wars.” A group of researchers, termed the Jackson 6, met and decided to end the controversy by running a randomized trial in US prisons (having discounted the options of long-term care homes and military bases). They detailed their plan in an editorial in Hypertension. The study never came to fruition for two reasons: the obvious ethical problems of experimenting on prisoners and the revelation of undisclosed salt industry funding.
More recent studies have mercifully been more conventional. The SSaSS study, a randomized controlled trial of a salt substitute, provided the cardiovascular outcomes data that many were waiting for. And CARDIA-SSBP, a cross-over randomized trial recently presented at the American Heart Association meeting, showed that reducing dietary sodium was on par with medication when it came to lowering blood pressure.
For Dr. Juraschek, the evidence is clear: “If you were going to choose one, I would say the weight of the evidence is still really heavily on the sodium side.”
The Case for Potassium Supplementation
The evidence for salt restriction notwithstanding, Swapnil Hiremath, MD, MPH, from the University of Ottawa, Ontario, Canada, argued in his editorial that potassium supplementation has gotten short shrift. Though he admits the studies for potassium supplementation have been smaller and sometimes rely on observational evidence, the evidence is there. In the distal convoluted tubule, the sodium chloride cotransporter (NCC), aka the potassium switch, is turned on by low potassium levels and leads to sodium reabsorption by the kidney even in settings of high sodium intake (Figure). To nonnephrologists, renal physiology may be a black box. But if you quickly brush up on the mechanism of action of thiazide diuretics, the preceding descriptor will make more sense.
Dr. Hiremath points out that the DASH diet study also got patients to increase their potassium intake by eating more fruits and vegetables. Furthermore, the SSaSS study tested a salt substitute that was 25% potassium (and 75% sodium).
How much blood pressure lowering is due to sodium restriction vs potassium supplementation is a complex question because lowering sodium intake will invariably lead to more potassium intake. “It’s very hard to untangle the relationship,” Dr. Hiremath said in an interview. “It’s sort of synergistic but it’s not completely additive. It’s not as if you add four and four and get eight.” But he maintains there is more evidence regarding the benefit of potassium supplementation than many realize.
Realistic Diets and Taste Issues
“We know that increasing potassium, decreasing sodium is useful. The question is how do we do that?” says Dr. Hiremath. Should we encourage fruit and vegetable consumption in a healthy diet, give potassium supplements, or encourage the use of low-sodium salt substitutes?
Recommending a healthier diet with more fruits and vegetables is a no-brainer. But getting people to do it is hard. In a world where fruit is more expensive than junk food is, economic realities may drive food choice regardless of our best efforts. The 4700 mg of potassium in the DASH eating plan is the equivalent of eleven bananas daily; although not impossible, it would require a substantive shift in eating patterns for most people.
Given that we prescribe iron, vitamin B12, calcium, and vitamin D to patients who need them, why not potassium tablets to help with blood pressure? Granted, there are concerns about inducing hyperkalemia. Also, why not just prescribe a proven anti-hypertensive, such as ramipril, which has the added benefit of helping with renal protection or cardiac remodeling? Dr. Hiremath points out that patients are far less reluctant to take dietary supplements. Medication is something you take when sick. A supplement is seen as “natural” and “healthy” and might be more attractive to people resistant to prescription meds.
Another drawback of oral potassium supplementation is taste. In a Consumer Reports taste test, potassium chloride fared poorly. It was bitter and had a metallic aftertaste. At least one tester wouldn’t ever consume it again. Potassium citrate is slightly more palpable.
Salt substitutes, like the 75:25 ratio of sodium to potassium used in SSaSS, may be as high as you can go for potassium in any low-sodium salt alternative. If you go any higher than that, the taste will just turn people off, suggests Dr. Hiremath.
But SsaSS, which was done in China, may not be relevant to North America. In China, most sodium is added during cooking at home, and the consumption of processed foods is low. For the typical North American, roughly three quarters of the sodium eaten is added to their food by someone else; only about 15% is added during cooking at home or at the dinner table. If you aren’t someone who cooks, buying a salt substitute is probably not going to have much impact.
Given that reality, Dr. Juraschek thinks we need to target the sodium in processed foods. “There’s just so much sodium in so many products,” he says. “When you think about public policy, it’s most expeditious for there to be more regulation about how much is added to our food supply vs trying to get people to consume eight to 12 servings of fruit.”
No Salt War Here
Despite their different editorial takes, Dr. Hiremath and Dr. Juraschek largely agree on the broad strokes of the problem. This isn’t X (or Twitter) after all. Potassium supplementation may be useful in some parts of the world but may not address the underlying problem in countries where processed foods are the source of most dietary sodium.
The CARDIA-SSBP trial showed that a very low–sodium diet had the same blood pressure–lowering effect as a first-line antihypertensive, but most people will not be able to limit themselves to 500 mg of dietary sodium per day. In CARDIA-SSBP, just as in DASH, participants were provided with meals from study kitchens. They were not just told to eat less salt, which would almost certainly have failed.
“We should aim for stuff that is practical and doable rather than aim for stuff that cannot be done,” according to Dr. Hiremath. Whether that should be salt substitutes or policy change may depend on which part of the planet you live on.
One recent positive change may herald the beginning of a policy change, at least in the United States. In March 2023, the US Food and Drug Administration proposed a rule change to allow salt substitutes to be labeled as salt. This would make it easier for food manufacturers to swap out sodium chloride for a low-sodium alternative and reduce the amount of sodium in the US diet without having a large impact on taste and consumer uptake. Both Dr. Hiremath and Dr. Juraschek agree that it may not be enough on its own but that it’s a start.
Christopher Labos is a cardiologist with a degree in epidemiology. He spends most of his time doing things that he doesn’t get paid for, like research, teaching, and podcasting. Occasionally, he finds time to practice cardiology to pay the rent. He realizes that half of his research findings will be disproved in 5 years; he just doesn’t know which half. He is a regular contributor to the Montreal Gazette, CJAD radio, and CTV television in Montreal, and is host of the award-winning podcast The Body of Evidence.
A version of this article appeared on Medscape.com.
A pair of dueling editorials in the journal Hypertension debate whether our focus should be on sodium or its often neglected partner, potassium.
A meta-analysis of 85 trials showed a consistent and linear. It may also depend on where you live and whether your concern is treating individuals or implementing effective food policy.
The Case for Sodium Restriction
Stephen Juraschek, MD, PhD, of the Beth Israel Deaconess Medical Center, Boston, Massachusetts, co-author of one editorial, told me in a zoom interview that he believes his side of the debate clearly has the stronger argument. Of the two cations in question, there has been infinitely more ink spilled about sodium.
Studies such as INTERSALT, the DASH diet, and TOHP may be the most well-known, but there are many, many intervention studies of sodium restriction’s effect on blood pressure. A meta-analysis of 85 trials of showed a consistent and linear relationship between sodium reduction and blood pressure. In contrast, the evidence base for potassium is more limited and less consistent. There are half as many trials with potassium, and its ability to lower blood pressure may depend on how much sodium is present in the diet.
An outlier in the sodium restriction evidence base is the PURE study, which suggested that extreme sodium restriction could increase cardiovascular mortality, but the trial suffered from two potential issues. First, it used a single spot urine specimen to measure sodium rather than the generally accepted more accurate 24-hour urine collection. A reanalysis of the TOHP study using a spot urine rather than a 24-hour urine collection changed the relationship between sodium intake and mortality and possibly explained the U-shaped association observed in PURE. Second, PURE was an observational cohort and was prone to confounding, or in this case, reverse causation. Why did people who consumed very little salt have an increased risk for cardiovascular disease? It is very possible that people with a high risk for cardiovascular disease were told to consume less salt to begin with. Hence B led to A rather than A leading to B.
The debate on sodium restriction has been bitter at times. Opposing camps formed, and people took sides in the “salt wars.” A group of researchers, termed the Jackson 6, met and decided to end the controversy by running a randomized trial in US prisons (having discounted the options of long-term care homes and military bases). They detailed their plan in an editorial in Hypertension. The study never came to fruition for two reasons: the obvious ethical problems of experimenting on prisoners and the revelation of undisclosed salt industry funding.
More recent studies have mercifully been more conventional. The SSaSS study, a randomized controlled trial of a salt substitute, provided the cardiovascular outcomes data that many were waiting for. And CARDIA-SSBP, a cross-over randomized trial recently presented at the American Heart Association meeting, showed that reducing dietary sodium was on par with medication when it came to lowering blood pressure.
For Dr. Juraschek, the evidence is clear: “If you were going to choose one, I would say the weight of the evidence is still really heavily on the sodium side.”
The Case for Potassium Supplementation
The evidence for salt restriction notwithstanding, Swapnil Hiremath, MD, MPH, from the University of Ottawa, Ontario, Canada, argued in his editorial that potassium supplementation has gotten short shrift. Though he admits the studies for potassium supplementation have been smaller and sometimes rely on observational evidence, the evidence is there. In the distal convoluted tubule, the sodium chloride cotransporter (NCC), aka the potassium switch, is turned on by low potassium levels and leads to sodium reabsorption by the kidney even in settings of high sodium intake (Figure). To nonnephrologists, renal physiology may be a black box. But if you quickly brush up on the mechanism of action of thiazide diuretics, the preceding descriptor will make more sense.
Dr. Hiremath points out that the DASH diet study also got patients to increase their potassium intake by eating more fruits and vegetables. Furthermore, the SSaSS study tested a salt substitute that was 25% potassium (and 75% sodium).
How much blood pressure lowering is due to sodium restriction vs potassium supplementation is a complex question because lowering sodium intake will invariably lead to more potassium intake. “It’s very hard to untangle the relationship,” Dr. Hiremath said in an interview. “It’s sort of synergistic but it’s not completely additive. It’s not as if you add four and four and get eight.” But he maintains there is more evidence regarding the benefit of potassium supplementation than many realize.
Realistic Diets and Taste Issues
“We know that increasing potassium, decreasing sodium is useful. The question is how do we do that?” says Dr. Hiremath. Should we encourage fruit and vegetable consumption in a healthy diet, give potassium supplements, or encourage the use of low-sodium salt substitutes?
Recommending a healthier diet with more fruits and vegetables is a no-brainer. But getting people to do it is hard. In a world where fruit is more expensive than junk food is, economic realities may drive food choice regardless of our best efforts. The 4700 mg of potassium in the DASH eating plan is the equivalent of eleven bananas daily; although not impossible, it would require a substantive shift in eating patterns for most people.
Given that we prescribe iron, vitamin B12, calcium, and vitamin D to patients who need them, why not potassium tablets to help with blood pressure? Granted, there are concerns about inducing hyperkalemia. Also, why not just prescribe a proven anti-hypertensive, such as ramipril, which has the added benefit of helping with renal protection or cardiac remodeling? Dr. Hiremath points out that patients are far less reluctant to take dietary supplements. Medication is something you take when sick. A supplement is seen as “natural” and “healthy” and might be more attractive to people resistant to prescription meds.
Another drawback of oral potassium supplementation is taste. In a Consumer Reports taste test, potassium chloride fared poorly. It was bitter and had a metallic aftertaste. At least one tester wouldn’t ever consume it again. Potassium citrate is slightly more palpable.
Salt substitutes, like the 75:25 ratio of sodium to potassium used in SSaSS, may be as high as you can go for potassium in any low-sodium salt alternative. If you go any higher than that, the taste will just turn people off, suggests Dr. Hiremath.
But SsaSS, which was done in China, may not be relevant to North America. In China, most sodium is added during cooking at home, and the consumption of processed foods is low. For the typical North American, roughly three quarters of the sodium eaten is added to their food by someone else; only about 15% is added during cooking at home or at the dinner table. If you aren’t someone who cooks, buying a salt substitute is probably not going to have much impact.
Given that reality, Dr. Juraschek thinks we need to target the sodium in processed foods. “There’s just so much sodium in so many products,” he says. “When you think about public policy, it’s most expeditious for there to be more regulation about how much is added to our food supply vs trying to get people to consume eight to 12 servings of fruit.”
No Salt War Here
Despite their different editorial takes, Dr. Hiremath and Dr. Juraschek largely agree on the broad strokes of the problem. This isn’t X (or Twitter) after all. Potassium supplementation may be useful in some parts of the world but may not address the underlying problem in countries where processed foods are the source of most dietary sodium.
The CARDIA-SSBP trial showed that a very low–sodium diet had the same blood pressure–lowering effect as a first-line antihypertensive, but most people will not be able to limit themselves to 500 mg of dietary sodium per day. In CARDIA-SSBP, just as in DASH, participants were provided with meals from study kitchens. They were not just told to eat less salt, which would almost certainly have failed.
“We should aim for stuff that is practical and doable rather than aim for stuff that cannot be done,” according to Dr. Hiremath. Whether that should be salt substitutes or policy change may depend on which part of the planet you live on.
One recent positive change may herald the beginning of a policy change, at least in the United States. In March 2023, the US Food and Drug Administration proposed a rule change to allow salt substitutes to be labeled as salt. This would make it easier for food manufacturers to swap out sodium chloride for a low-sodium alternative and reduce the amount of sodium in the US diet without having a large impact on taste and consumer uptake. Both Dr. Hiremath and Dr. Juraschek agree that it may not be enough on its own but that it’s a start.
Christopher Labos is a cardiologist with a degree in epidemiology. He spends most of his time doing things that he doesn’t get paid for, like research, teaching, and podcasting. Occasionally, he finds time to practice cardiology to pay the rent. He realizes that half of his research findings will be disproved in 5 years; he just doesn’t know which half. He is a regular contributor to the Montreal Gazette, CJAD radio, and CTV television in Montreal, and is host of the award-winning podcast The Body of Evidence.
A version of this article appeared on Medscape.com.
More Young Women Being Diagnosed With Breast Cancer Than Ever Before
This transcript has been edited for clarity.
From the year 2000 until around 2016, the incidence of breast cancer among young women — those under age 50 — rose steadily, if slowly.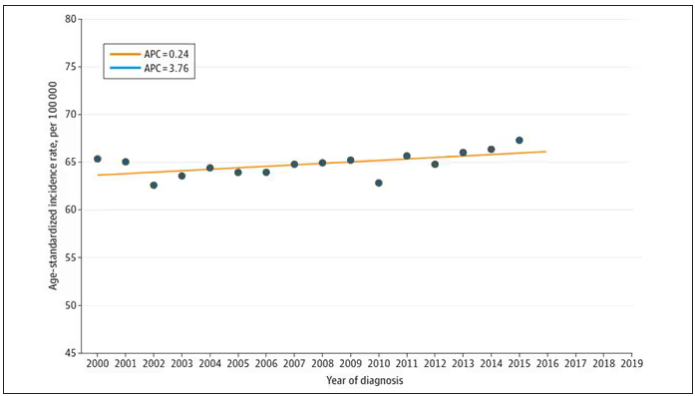
And then this happened: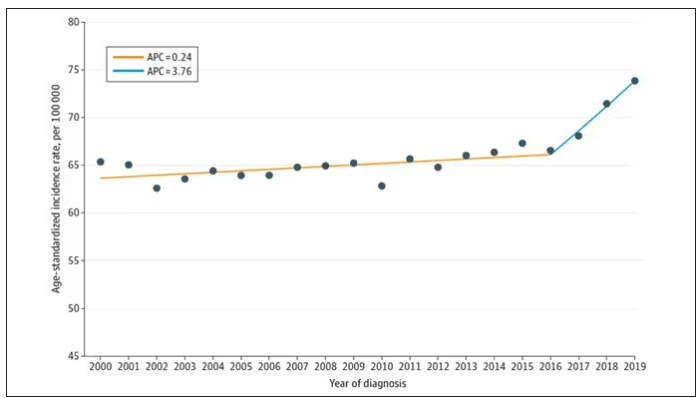
I look at a lot of graphs in my line of work, and it’s not too often that one actually makes me say “What the hell?” out loud. But this one did. Why are young women all of a sudden more likely to get breast cancer?
The graph comes from this paper, Breast cancer incidence among us women aged 20 to 49 years by race, stage, and hormone receptor status, appearing in JAMA Network Open
Researchers from Washington University in St. Louis utilized SEER registries to conduct their analyses. SEER is a public database from the National Cancer Institute with coverage of 27% of the US population and a long track record of statistical backbone to translate the data from SEER to numbers that are representative of the population at large.
From 2000 to 2019, more than 200,000 women were diagnosed with primary invasive breast cancer in the dataset, and I’ve already given you the top-line results. Of course, when you see a graph like this, the next question really needs to be why?
Fortunately, the SEER dataset contains a lot more information than simply whether someone was diagnosed with cancer. In the case of breast cancer, there is information about the patient’s demographics, the hormone status of the cancer, the stage, and so on. Using those additional data points can help the authors, and us, start to formulate some hypotheses as to what is happening here.
Let’s start with something a bit tricky about this kind of data. We see an uptick in new breast cancer diagnoses among young women in recent years. We need to tease that uptick apart a bit. It could be that it is the year that is the key factor here. In other words, it is simply that more women are getting breast cancer since 2016 and so more young women are getting breast cancer since 2016. These are known as period effects.
Or is there something unique to young women — something about their environmental exposures that put them at higher risk than they would have been had they been born at some other time? These are known as cohort effects.
The researchers teased these two effects apart, as you can see here, and concluded that, well, it’s both.
Stage of cancer at diagnosis can give us some more insight into what is happening. These results are pretty interesting. These higher cancer rates are due primarily to stage I and stage IV cancers, not stage II and stage III cancers.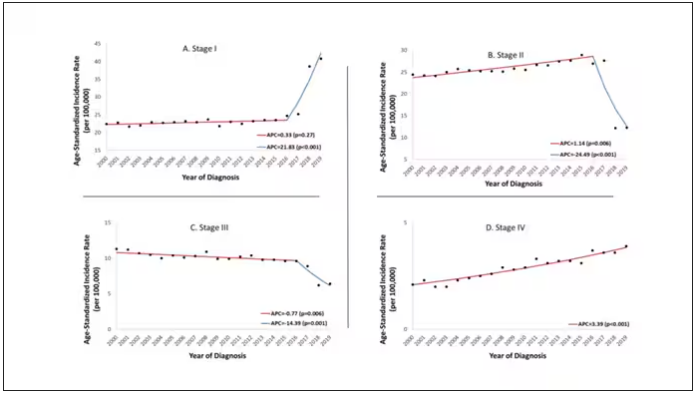
The rising incidence of stage I cancers could reflect better detection, though many of the women in this cohort would not have been old enough to quality for screening mammograms. That said, increased awareness about genetic risk and family history might be leading younger women to get screened, picking up more early cancers. Additionally, much of the increased incidence was with estrogen receptor–positive tumors, which might reflect the fact that women in this cohort are tending to have fewer children, and children later in life.
So why the rise in stage IV breast cancer? Well, precisely because younger women are not recommended to get screening mammograms; those who detect a lump on their own are likely to be at a more advanced stage. But I’m not sure why that would be changing recently. The authors argue that an increase in overweight and obesity in the country might be to blame here. Prior studies have shown that higher BMI is associated with higher stage at breast cancer diagnosis.
Of course, we can speculate as to multiple other causes as well: environmental toxins, pollution, hormone exposures, and so on. Figuring this out will be the work of multiple other studies. In the meantime, we should remember that the landscape of cancer is continuously changing. And that means we need to adapt to it. If these trends continue, national agencies may need to reconsider their guidelines for when screening mammography should begin — at least in some groups of young women.
Dr. F. Perry Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
From the year 2000 until around 2016, the incidence of breast cancer among young women — those under age 50 — rose steadily, if slowly.
And then this happened:
I look at a lot of graphs in my line of work, and it’s not too often that one actually makes me say “What the hell?” out loud. But this one did. Why are young women all of a sudden more likely to get breast cancer?
The graph comes from this paper, Breast cancer incidence among us women aged 20 to 49 years by race, stage, and hormone receptor status, appearing in JAMA Network Open
Researchers from Washington University in St. Louis utilized SEER registries to conduct their analyses. SEER is a public database from the National Cancer Institute with coverage of 27% of the US population and a long track record of statistical backbone to translate the data from SEER to numbers that are representative of the population at large.
From 2000 to 2019, more than 200,000 women were diagnosed with primary invasive breast cancer in the dataset, and I’ve already given you the top-line results. Of course, when you see a graph like this, the next question really needs to be why?
Fortunately, the SEER dataset contains a lot more information than simply whether someone was diagnosed with cancer. In the case of breast cancer, there is information about the patient’s demographics, the hormone status of the cancer, the stage, and so on. Using those additional data points can help the authors, and us, start to formulate some hypotheses as to what is happening here.
Let’s start with something a bit tricky about this kind of data. We see an uptick in new breast cancer diagnoses among young women in recent years. We need to tease that uptick apart a bit. It could be that it is the year that is the key factor here. In other words, it is simply that more women are getting breast cancer since 2016 and so more young women are getting breast cancer since 2016. These are known as period effects.
Or is there something unique to young women — something about their environmental exposures that put them at higher risk than they would have been had they been born at some other time? These are known as cohort effects.
The researchers teased these two effects apart, as you can see here, and concluded that, well, it’s both.
Stage of cancer at diagnosis can give us some more insight into what is happening. These results are pretty interesting. These higher cancer rates are due primarily to stage I and stage IV cancers, not stage II and stage III cancers.
The rising incidence of stage I cancers could reflect better detection, though many of the women in this cohort would not have been old enough to quality for screening mammograms. That said, increased awareness about genetic risk and family history might be leading younger women to get screened, picking up more early cancers. Additionally, much of the increased incidence was with estrogen receptor–positive tumors, which might reflect the fact that women in this cohort are tending to have fewer children, and children later in life.
So why the rise in stage IV breast cancer? Well, precisely because younger women are not recommended to get screening mammograms; those who detect a lump on their own are likely to be at a more advanced stage. But I’m not sure why that would be changing recently. The authors argue that an increase in overweight and obesity in the country might be to blame here. Prior studies have shown that higher BMI is associated with higher stage at breast cancer diagnosis.
Of course, we can speculate as to multiple other causes as well: environmental toxins, pollution, hormone exposures, and so on. Figuring this out will be the work of multiple other studies. In the meantime, we should remember that the landscape of cancer is continuously changing. And that means we need to adapt to it. If these trends continue, national agencies may need to reconsider their guidelines for when screening mammography should begin — at least in some groups of young women.
Dr. F. Perry Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
From the year 2000 until around 2016, the incidence of breast cancer among young women — those under age 50 — rose steadily, if slowly.
And then this happened:
I look at a lot of graphs in my line of work, and it’s not too often that one actually makes me say “What the hell?” out loud. But this one did. Why are young women all of a sudden more likely to get breast cancer?
The graph comes from this paper, Breast cancer incidence among us women aged 20 to 49 years by race, stage, and hormone receptor status, appearing in JAMA Network Open
Researchers from Washington University in St. Louis utilized SEER registries to conduct their analyses. SEER is a public database from the National Cancer Institute with coverage of 27% of the US population and a long track record of statistical backbone to translate the data from SEER to numbers that are representative of the population at large.
From 2000 to 2019, more than 200,000 women were diagnosed with primary invasive breast cancer in the dataset, and I’ve already given you the top-line results. Of course, when you see a graph like this, the next question really needs to be why?
Fortunately, the SEER dataset contains a lot more information than simply whether someone was diagnosed with cancer. In the case of breast cancer, there is information about the patient’s demographics, the hormone status of the cancer, the stage, and so on. Using those additional data points can help the authors, and us, start to formulate some hypotheses as to what is happening here.
Let’s start with something a bit tricky about this kind of data. We see an uptick in new breast cancer diagnoses among young women in recent years. We need to tease that uptick apart a bit. It could be that it is the year that is the key factor here. In other words, it is simply that more women are getting breast cancer since 2016 and so more young women are getting breast cancer since 2016. These are known as period effects.
Or is there something unique to young women — something about their environmental exposures that put them at higher risk than they would have been had they been born at some other time? These are known as cohort effects.
The researchers teased these two effects apart, as you can see here, and concluded that, well, it’s both.
Stage of cancer at diagnosis can give us some more insight into what is happening. These results are pretty interesting. These higher cancer rates are due primarily to stage I and stage IV cancers, not stage II and stage III cancers.
The rising incidence of stage I cancers could reflect better detection, though many of the women in this cohort would not have been old enough to quality for screening mammograms. That said, increased awareness about genetic risk and family history might be leading younger women to get screened, picking up more early cancers. Additionally, much of the increased incidence was with estrogen receptor–positive tumors, which might reflect the fact that women in this cohort are tending to have fewer children, and children later in life.
So why the rise in stage IV breast cancer? Well, precisely because younger women are not recommended to get screening mammograms; those who detect a lump on their own are likely to be at a more advanced stage. But I’m not sure why that would be changing recently. The authors argue that an increase in overweight and obesity in the country might be to blame here. Prior studies have shown that higher BMI is associated with higher stage at breast cancer diagnosis.
Of course, we can speculate as to multiple other causes as well: environmental toxins, pollution, hormone exposures, and so on. Figuring this out will be the work of multiple other studies. In the meantime, we should remember that the landscape of cancer is continuously changing. And that means we need to adapt to it. If these trends continue, national agencies may need to reconsider their guidelines for when screening mammography should begin — at least in some groups of young women.
Dr. F. Perry Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
How to Motivate Pain Patients to Try Nondrug Options
This transcript has been edited for clarity.
Neha Pathak, MD: Hello. Today, we’re talking to Dr. Daniel Clauw, a professor at the University of Michigan in Ann Arbor, who is running a major trial on treatments for chronic back pain. We’re talking today about managing back pain in the post-opioid world. Thank you so much, Dr. Clauw, for taking the time to be our resident pain consultant today. Managing chronic pain can lead to a large amount of burnout and helplessness in the clinic setting. That’s the reality with some of the modalities that patients are requesting; there is still confusion about what is optimal for a particular type of patient, this feeling that we’re not really helping people get better, and whenever patients come in, that’s always still their chief complaint.
How would you advise providers to think about that and to settle into their role as communicators about better strategies without the burnout?
Daniel Clauw, MD: The first thing is to broaden the number of other providers that you get involved in these individuals’ care as the evidence base for all of these nonpharmacologic therapies being effective in chronic pain increases and increases. As third-party payers begin to reimburse for more and more of these therapies, it’s really difficult to manage chronic pain patients if you’re trying to do it alone on an island.
If you can, identify the good physical therapists in your community that are going to really work with people to give them an exercise program that they can use at home; find a pain psychologist that can offer some cognitive-behavioral therapy (CBT) for insomnia and some CBT for pain; and in the subset of patients with trauma, give them the emotional awareness of the neural reprocessing therapy for that specific subset.
As you start to identify more and more of these nonpharmacologic therapies that you want your patients to try, each of those has a set of providers and they can be incredibly helpful so that you, as the primary care provider (PCP), don’t really feel overwhelmed that you’re it, that you’re the only one.
Many of these individuals have more time to spend, and they have more one-on-one in-person time than you do as a primary care physician in the current healthcare system. Many of those providers have become really good at doing amateur CBT, goal-setting, and some of the other things that you need to do when you manage chronic pain patients. Try to find that other group of people that you can send your patients to that are going to be offering some of these nonpharmacologic therapies, and they’ll really help you manage these individuals.
Dr. Pathak: I think a couple of things come up for me. One is that we have to maybe broaden thinking about pain management, not only as multimodal strategies but also as multidisciplinary strategies. To your point, I think that’s really important. I also worry and wonder about health equity concerns, because just as overburdened as many PCPs are, we’re seeing it’s very difficult to get into physical therapy or to get into a setting where you’d be able to receive CBT for your pain. Any thoughts on those types of considerations?
Dr. Clauw: That’s a huge problem. Our group and many other groups in the pain space are developing websites, smartphone apps, and things like that to try to get some of these things directly to individuals with pain, not only for the reasons that you stated but also so that persons with pain don’t have to become patients. Our healthcare systems often make pain worse rather than better.
There were some great articles in The Lancet about 5 years ago talking about low back pain and that in different countries, the healthcare systems, for different reasons, have a tendency to actually make low back pain worse because they do too much surgery, immobilize people, or things like that rather than just not make them better. I think we’ve overmedicalized chronic pain in some settings, and much of what we’re trying to lead people to are things that are parts of wellness programs. The NIH National Center for Complementary and Integrative Health director talks about whole person health often.
I think that these interdisciplinary, integrative approaches are what we have to be using for chronic pain patients. I tell pain patients that, among acupuncture, acupressure, mindfulness, five different forms of CBT, yoga, and tai chi, I don’t know which of those is going to work, but I know that about 1 in 3 individuals that tries each of those therapies gets a benefit. What I really should be doing most is incentivizing people and motivating people to keep trying some of those nonpharmacologic approaches that they haven’t yet tried, because when they find one that works for them, then they will integrate it into their day-to-day life.
The other trick I would use for primary care physicians or anyone managing chronic pain patients is, don’t try to incentivize a pain patient to go try a new nonpharmacologic therapy or start an exercise program because you want their pain score to go from a 6 to a 3. Incentivize them by asking them, what are two or three things that you’re not able to do now because you have chronic pain that you’d really like to be able to do?
You’d like to play nine holes of golf; you’d like to be able to hug your grandchild; or you’d like to be able to do something else. Use those functional goals that are patient0driven to motivate your patients to do these things, because that will work much better. Again, any of us are inherently more likely to take the time and the effort to do some of these nonpharmacologic therapies if it’s for a reason that internally motivates us.
Dr. Pathak: I think that’s great. I’m very privileged to work within the Veterans Affairs (VA) healthcare system. I think that there’s been a huge shift within VA healthcare to provide these ancillary services, whether it’s yoga, tai chi, or acupuncture, as an adjunct to the pain management strategy.
Also, what comes up for me, as you’re saying, is grounding the point that instead of relying on a pain score — which can be objective and different from patient to patient and even within a patient — we should choose a smart goal that is almost more objective when it’s functional. Your goal is to walk two blocks to the mailbox. Can we achieve that as part of your pain control strategy?
I so appreciate your taking the time to be our pain consultant today. I really appreciate our discussion, and I’d like to hand it over to you for any final thoughts.
Dr. Clauw: I’d add that when you’re seeing chronic pain patients, many of them are going to have comorbid sleep problems. They’re going to have comorbid problems with fatigue and memory problems, especially the central nervous system–driven forms of pain that we now call nociplastic pain. Look at those as therapeutic targets.
If you’re befuddled because you’ve tried many different things for pain in this individual you’ve been seeing for a while, focus on their sleep and focus on getting them more active. Don’t use the word exercise — because that scares chronic pain patients — but focus on getting them more active.
There are many different tactics and strategies that you can use to motivate the patients to try some of these new nonpharmacologic approaches as the evidence base continues to increase.
Dr. Pathak: Thank you so much, again, to Dr. Clauw for joining us and being our pain consultant, really helping us to think about managing back pain in the postopioid world.
Dr. Pathak is Chief Physician Editor, Health and Lifestyle Medicine, WebMD. She has disclosed no relevant financial relationships. Dr. Clauw is Director, Chronic Pain and Fatigue Research Center, Department of Anesthesia, University of Michigan, Ann Arbor. He disclosed ties with Tonix and Viatris.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
Neha Pathak, MD: Hello. Today, we’re talking to Dr. Daniel Clauw, a professor at the University of Michigan in Ann Arbor, who is running a major trial on treatments for chronic back pain. We’re talking today about managing back pain in the post-opioid world. Thank you so much, Dr. Clauw, for taking the time to be our resident pain consultant today. Managing chronic pain can lead to a large amount of burnout and helplessness in the clinic setting. That’s the reality with some of the modalities that patients are requesting; there is still confusion about what is optimal for a particular type of patient, this feeling that we’re not really helping people get better, and whenever patients come in, that’s always still their chief complaint.
How would you advise providers to think about that and to settle into their role as communicators about better strategies without the burnout?
Daniel Clauw, MD: The first thing is to broaden the number of other providers that you get involved in these individuals’ care as the evidence base for all of these nonpharmacologic therapies being effective in chronic pain increases and increases. As third-party payers begin to reimburse for more and more of these therapies, it’s really difficult to manage chronic pain patients if you’re trying to do it alone on an island.
If you can, identify the good physical therapists in your community that are going to really work with people to give them an exercise program that they can use at home; find a pain psychologist that can offer some cognitive-behavioral therapy (CBT) for insomnia and some CBT for pain; and in the subset of patients with trauma, give them the emotional awareness of the neural reprocessing therapy for that specific subset.
As you start to identify more and more of these nonpharmacologic therapies that you want your patients to try, each of those has a set of providers and they can be incredibly helpful so that you, as the primary care provider (PCP), don’t really feel overwhelmed that you’re it, that you’re the only one.
Many of these individuals have more time to spend, and they have more one-on-one in-person time than you do as a primary care physician in the current healthcare system. Many of those providers have become really good at doing amateur CBT, goal-setting, and some of the other things that you need to do when you manage chronic pain patients. Try to find that other group of people that you can send your patients to that are going to be offering some of these nonpharmacologic therapies, and they’ll really help you manage these individuals.
Dr. Pathak: I think a couple of things come up for me. One is that we have to maybe broaden thinking about pain management, not only as multimodal strategies but also as multidisciplinary strategies. To your point, I think that’s really important. I also worry and wonder about health equity concerns, because just as overburdened as many PCPs are, we’re seeing it’s very difficult to get into physical therapy or to get into a setting where you’d be able to receive CBT for your pain. Any thoughts on those types of considerations?
Dr. Clauw: That’s a huge problem. Our group and many other groups in the pain space are developing websites, smartphone apps, and things like that to try to get some of these things directly to individuals with pain, not only for the reasons that you stated but also so that persons with pain don’t have to become patients. Our healthcare systems often make pain worse rather than better.
There were some great articles in The Lancet about 5 years ago talking about low back pain and that in different countries, the healthcare systems, for different reasons, have a tendency to actually make low back pain worse because they do too much surgery, immobilize people, or things like that rather than just not make them better. I think we’ve overmedicalized chronic pain in some settings, and much of what we’re trying to lead people to are things that are parts of wellness programs. The NIH National Center for Complementary and Integrative Health director talks about whole person health often.
I think that these interdisciplinary, integrative approaches are what we have to be using for chronic pain patients. I tell pain patients that, among acupuncture, acupressure, mindfulness, five different forms of CBT, yoga, and tai chi, I don’t know which of those is going to work, but I know that about 1 in 3 individuals that tries each of those therapies gets a benefit. What I really should be doing most is incentivizing people and motivating people to keep trying some of those nonpharmacologic approaches that they haven’t yet tried, because when they find one that works for them, then they will integrate it into their day-to-day life.
The other trick I would use for primary care physicians or anyone managing chronic pain patients is, don’t try to incentivize a pain patient to go try a new nonpharmacologic therapy or start an exercise program because you want their pain score to go from a 6 to a 3. Incentivize them by asking them, what are two or three things that you’re not able to do now because you have chronic pain that you’d really like to be able to do?
You’d like to play nine holes of golf; you’d like to be able to hug your grandchild; or you’d like to be able to do something else. Use those functional goals that are patient0driven to motivate your patients to do these things, because that will work much better. Again, any of us are inherently more likely to take the time and the effort to do some of these nonpharmacologic therapies if it’s for a reason that internally motivates us.
Dr. Pathak: I think that’s great. I’m very privileged to work within the Veterans Affairs (VA) healthcare system. I think that there’s been a huge shift within VA healthcare to provide these ancillary services, whether it’s yoga, tai chi, or acupuncture, as an adjunct to the pain management strategy.
Also, what comes up for me, as you’re saying, is grounding the point that instead of relying on a pain score — which can be objective and different from patient to patient and even within a patient — we should choose a smart goal that is almost more objective when it’s functional. Your goal is to walk two blocks to the mailbox. Can we achieve that as part of your pain control strategy?
I so appreciate your taking the time to be our pain consultant today. I really appreciate our discussion, and I’d like to hand it over to you for any final thoughts.
Dr. Clauw: I’d add that when you’re seeing chronic pain patients, many of them are going to have comorbid sleep problems. They’re going to have comorbid problems with fatigue and memory problems, especially the central nervous system–driven forms of pain that we now call nociplastic pain. Look at those as therapeutic targets.
If you’re befuddled because you’ve tried many different things for pain in this individual you’ve been seeing for a while, focus on their sleep and focus on getting them more active. Don’t use the word exercise — because that scares chronic pain patients — but focus on getting them more active.
There are many different tactics and strategies that you can use to motivate the patients to try some of these new nonpharmacologic approaches as the evidence base continues to increase.
Dr. Pathak: Thank you so much, again, to Dr. Clauw for joining us and being our pain consultant, really helping us to think about managing back pain in the postopioid world.
Dr. Pathak is Chief Physician Editor, Health and Lifestyle Medicine, WebMD. She has disclosed no relevant financial relationships. Dr. Clauw is Director, Chronic Pain and Fatigue Research Center, Department of Anesthesia, University of Michigan, Ann Arbor. He disclosed ties with Tonix and Viatris.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
Neha Pathak, MD: Hello. Today, we’re talking to Dr. Daniel Clauw, a professor at the University of Michigan in Ann Arbor, who is running a major trial on treatments for chronic back pain. We’re talking today about managing back pain in the post-opioid world. Thank you so much, Dr. Clauw, for taking the time to be our resident pain consultant today. Managing chronic pain can lead to a large amount of burnout and helplessness in the clinic setting. That’s the reality with some of the modalities that patients are requesting; there is still confusion about what is optimal for a particular type of patient, this feeling that we’re not really helping people get better, and whenever patients come in, that’s always still their chief complaint.
How would you advise providers to think about that and to settle into their role as communicators about better strategies without the burnout?
Daniel Clauw, MD: The first thing is to broaden the number of other providers that you get involved in these individuals’ care as the evidence base for all of these nonpharmacologic therapies being effective in chronic pain increases and increases. As third-party payers begin to reimburse for more and more of these therapies, it’s really difficult to manage chronic pain patients if you’re trying to do it alone on an island.
If you can, identify the good physical therapists in your community that are going to really work with people to give them an exercise program that they can use at home; find a pain psychologist that can offer some cognitive-behavioral therapy (CBT) for insomnia and some CBT for pain; and in the subset of patients with trauma, give them the emotional awareness of the neural reprocessing therapy for that specific subset.
As you start to identify more and more of these nonpharmacologic therapies that you want your patients to try, each of those has a set of providers and they can be incredibly helpful so that you, as the primary care provider (PCP), don’t really feel overwhelmed that you’re it, that you’re the only one.
Many of these individuals have more time to spend, and they have more one-on-one in-person time than you do as a primary care physician in the current healthcare system. Many of those providers have become really good at doing amateur CBT, goal-setting, and some of the other things that you need to do when you manage chronic pain patients. Try to find that other group of people that you can send your patients to that are going to be offering some of these nonpharmacologic therapies, and they’ll really help you manage these individuals.
Dr. Pathak: I think a couple of things come up for me. One is that we have to maybe broaden thinking about pain management, not only as multimodal strategies but also as multidisciplinary strategies. To your point, I think that’s really important. I also worry and wonder about health equity concerns, because just as overburdened as many PCPs are, we’re seeing it’s very difficult to get into physical therapy or to get into a setting where you’d be able to receive CBT for your pain. Any thoughts on those types of considerations?
Dr. Clauw: That’s a huge problem. Our group and many other groups in the pain space are developing websites, smartphone apps, and things like that to try to get some of these things directly to individuals with pain, not only for the reasons that you stated but also so that persons with pain don’t have to become patients. Our healthcare systems often make pain worse rather than better.
There were some great articles in The Lancet about 5 years ago talking about low back pain and that in different countries, the healthcare systems, for different reasons, have a tendency to actually make low back pain worse because they do too much surgery, immobilize people, or things like that rather than just not make them better. I think we’ve overmedicalized chronic pain in some settings, and much of what we’re trying to lead people to are things that are parts of wellness programs. The NIH National Center for Complementary and Integrative Health director talks about whole person health often.
I think that these interdisciplinary, integrative approaches are what we have to be using for chronic pain patients. I tell pain patients that, among acupuncture, acupressure, mindfulness, five different forms of CBT, yoga, and tai chi, I don’t know which of those is going to work, but I know that about 1 in 3 individuals that tries each of those therapies gets a benefit. What I really should be doing most is incentivizing people and motivating people to keep trying some of those nonpharmacologic approaches that they haven’t yet tried, because when they find one that works for them, then they will integrate it into their day-to-day life.
The other trick I would use for primary care physicians or anyone managing chronic pain patients is, don’t try to incentivize a pain patient to go try a new nonpharmacologic therapy or start an exercise program because you want their pain score to go from a 6 to a 3. Incentivize them by asking them, what are two or three things that you’re not able to do now because you have chronic pain that you’d really like to be able to do?
You’d like to play nine holes of golf; you’d like to be able to hug your grandchild; or you’d like to be able to do something else. Use those functional goals that are patient0driven to motivate your patients to do these things, because that will work much better. Again, any of us are inherently more likely to take the time and the effort to do some of these nonpharmacologic therapies if it’s for a reason that internally motivates us.
Dr. Pathak: I think that’s great. I’m very privileged to work within the Veterans Affairs (VA) healthcare system. I think that there’s been a huge shift within VA healthcare to provide these ancillary services, whether it’s yoga, tai chi, or acupuncture, as an adjunct to the pain management strategy.
Also, what comes up for me, as you’re saying, is grounding the point that instead of relying on a pain score — which can be objective and different from patient to patient and even within a patient — we should choose a smart goal that is almost more objective when it’s functional. Your goal is to walk two blocks to the mailbox. Can we achieve that as part of your pain control strategy?
I so appreciate your taking the time to be our pain consultant today. I really appreciate our discussion, and I’d like to hand it over to you for any final thoughts.
Dr. Clauw: I’d add that when you’re seeing chronic pain patients, many of them are going to have comorbid sleep problems. They’re going to have comorbid problems with fatigue and memory problems, especially the central nervous system–driven forms of pain that we now call nociplastic pain. Look at those as therapeutic targets.
If you’re befuddled because you’ve tried many different things for pain in this individual you’ve been seeing for a while, focus on their sleep and focus on getting them more active. Don’t use the word exercise — because that scares chronic pain patients — but focus on getting them more active.
There are many different tactics and strategies that you can use to motivate the patients to try some of these new nonpharmacologic approaches as the evidence base continues to increase.
Dr. Pathak: Thank you so much, again, to Dr. Clauw for joining us and being our pain consultant, really helping us to think about managing back pain in the postopioid world.
Dr. Pathak is Chief Physician Editor, Health and Lifestyle Medicine, WebMD. She has disclosed no relevant financial relationships. Dr. Clauw is Director, Chronic Pain and Fatigue Research Center, Department of Anesthesia, University of Michigan, Ann Arbor. He disclosed ties with Tonix and Viatris.
A version of this article appeared on Medscape.com.
VA Versus the Private Sector — No Contest? Think Again
Veterans Administration (VA) hospitals are a stepchild in the bizarre mishmash of the U.S. healthcare system. They’re best known (often justifiably so) for rather cantankerous patients, rigid rules, and other oddities (such as patients being able to go on leave and come back).
The majority of American doctors, including myself, did at least part of our training at a VA and have no shortage of stories about them. One I worked at (Omaha VA) was powered by its own nuclear reactor in the basement (no, really, it was, though sadly it’s since been taken out).
VA hospitals, in general, are no-frills — linoleum floors, no piano player in the lobby, very few private rooms, and occasionally in the news for scandals like the one at my hometown Carl T. Hayden VA hospital (I trained there, too).
Yet, . Their focus on patient care, rather than profits, allows them to run with 8% fewer administrative staff since they generally don’t have to deal with insurance billings, denials, and other paperwork (they also don’t have to deal with shareholders and investor demands or ridiculous CEO salaries, though the study didn’t address that).
On a national scale, this would mean roughly 900,000 fewer administrative jobs in the private sector. Granted, that also would mean those people would have to find other jobs, but let’s look at the patient side. If you had 900,000 fewer desk workers, you’d have the money to hire more nurses, respiratory techs, therapists, and other people directly involved in patient care. You’d also need a lot less office space, which further brings down overhead.
Part of the problem is that a lot of the current medical business is in marketing — how many ads do you see each day for different hospitals in your area? — and upcoding to extract more money from each billing. Neither of these has any clinical value on the patient side of things.
You don’t have to look back too far (2020) for the study that found U.S. healthcare spent four times as much money ($812 billion) per capita than our northern neighbors.
And, for all the jokes we make about the VA (myself included), a study last year found its care was on par (or even better than) most hospitals .
I’m not saying the VA is perfect. All of us who worked there can think of times it wasn’t. But we also remember plenty of issues we’ve had at other places we’ve practiced, too.
Maybe it’s time to stop laughing at the VA and realize they’re doing something right — and learn from it to make healthcare better at the other 6,000 or so hospitals in the U.S.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
Veterans Administration (VA) hospitals are a stepchild in the bizarre mishmash of the U.S. healthcare system. They’re best known (often justifiably so) for rather cantankerous patients, rigid rules, and other oddities (such as patients being able to go on leave and come back).
The majority of American doctors, including myself, did at least part of our training at a VA and have no shortage of stories about them. One I worked at (Omaha VA) was powered by its own nuclear reactor in the basement (no, really, it was, though sadly it’s since been taken out).
VA hospitals, in general, are no-frills — linoleum floors, no piano player in the lobby, very few private rooms, and occasionally in the news for scandals like the one at my hometown Carl T. Hayden VA hospital (I trained there, too).
Yet, . Their focus on patient care, rather than profits, allows them to run with 8% fewer administrative staff since they generally don’t have to deal with insurance billings, denials, and other paperwork (they also don’t have to deal with shareholders and investor demands or ridiculous CEO salaries, though the study didn’t address that).
On a national scale, this would mean roughly 900,000 fewer administrative jobs in the private sector. Granted, that also would mean those people would have to find other jobs, but let’s look at the patient side. If you had 900,000 fewer desk workers, you’d have the money to hire more nurses, respiratory techs, therapists, and other people directly involved in patient care. You’d also need a lot less office space, which further brings down overhead.
Part of the problem is that a lot of the current medical business is in marketing — how many ads do you see each day for different hospitals in your area? — and upcoding to extract more money from each billing. Neither of these has any clinical value on the patient side of things.
You don’t have to look back too far (2020) for the study that found U.S. healthcare spent four times as much money ($812 billion) per capita than our northern neighbors.
And, for all the jokes we make about the VA (myself included), a study last year found its care was on par (or even better than) most hospitals .
I’m not saying the VA is perfect. All of us who worked there can think of times it wasn’t. But we also remember plenty of issues we’ve had at other places we’ve practiced, too.
Maybe it’s time to stop laughing at the VA and realize they’re doing something right — and learn from it to make healthcare better at the other 6,000 or so hospitals in the U.S.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
Veterans Administration (VA) hospitals are a stepchild in the bizarre mishmash of the U.S. healthcare system. They’re best known (often justifiably so) for rather cantankerous patients, rigid rules, and other oddities (such as patients being able to go on leave and come back).
The majority of American doctors, including myself, did at least part of our training at a VA and have no shortage of stories about them. One I worked at (Omaha VA) was powered by its own nuclear reactor in the basement (no, really, it was, though sadly it’s since been taken out).
VA hospitals, in general, are no-frills — linoleum floors, no piano player in the lobby, very few private rooms, and occasionally in the news for scandals like the one at my hometown Carl T. Hayden VA hospital (I trained there, too).
Yet, . Their focus on patient care, rather than profits, allows them to run with 8% fewer administrative staff since they generally don’t have to deal with insurance billings, denials, and other paperwork (they also don’t have to deal with shareholders and investor demands or ridiculous CEO salaries, though the study didn’t address that).
On a national scale, this would mean roughly 900,000 fewer administrative jobs in the private sector. Granted, that also would mean those people would have to find other jobs, but let’s look at the patient side. If you had 900,000 fewer desk workers, you’d have the money to hire more nurses, respiratory techs, therapists, and other people directly involved in patient care. You’d also need a lot less office space, which further brings down overhead.
Part of the problem is that a lot of the current medical business is in marketing — how many ads do you see each day for different hospitals in your area? — and upcoding to extract more money from each billing. Neither of these has any clinical value on the patient side of things.
You don’t have to look back too far (2020) for the study that found U.S. healthcare spent four times as much money ($812 billion) per capita than our northern neighbors.
And, for all the jokes we make about the VA (myself included), a study last year found its care was on par (or even better than) most hospitals .
I’m not saying the VA is perfect. All of us who worked there can think of times it wasn’t. But we also remember plenty of issues we’ve had at other places we’ve practiced, too.
Maybe it’s time to stop laughing at the VA and realize they’re doing something right — and learn from it to make healthcare better at the other 6,000 or so hospitals in the U.S.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
Medical Aid in Dying Should Be Legal, Says Ethicist
This transcript has been edited for clarity.
Hi. I’m Art Caplan. I’m at the Division of Medical Ethics at the NYU Grossman School of Medicine.
Right now, there are 10 states and the District of Columbia that have had some version of medical assistance in dying approved and on the books. That basically means that about 20% of Americans have access where they live to a physician who can prescribe a lethal dose of medication to them if they’re terminally ill and can ingest the medication themselves. That leaves many Americans not covered by this kind of access to this kind of service.
Many of you watching this may live in states where it is legal, like Oregon, Washington, New Jersey, Colorado, and Hawaii. I know many doctors say, “I’m not going to do that.” It’s not something that anyone is compelling a doctor to do. For some Americans, access is not just about where they live but whether there is a doctor willing to participate with them in bringing about their accelerated death, knowing that they’re inevitably going to die.
There’s not much we can do about that. It’s up to the conscience of each physician as to what they’re comfortable with. Certainly, there are other things that can be done to extend the possibility of having this available.
One thing that’s taking place is that, after lawsuits were filed, Vermont and Oregon have given up on their residency requirement, so you don’t have to be there 6 months or a year in order to use this opportunity. It’s legal now to move to the state or visit the state, and as soon as you get there, sign up for this kind of end-of-life intervention.
New Jersey is also being sued. I’ll predict that every state that has a residency requirement, when sued in court, is going to lose because we’ve long recognized the right of Americans to seek out healthcare in the United States, wherever they want to go.
If some states have made this a legitimate medical procedure, courts are going to say you can’t restrict it only to state residents. If someone wants to use a service, they’re entitled to show up from another state or another place and use it. I’m not sure about foreign nationals, but I’m very sure that Americans can go state to state in search of legitimate medical procedures.
The other bills that are out there, however, are basically saying they want to emulate Oregon, Washington, and the other states and say that the terminally ill, with severe restrictions, are going to be able to get this service without going anywhere.
The restrictions include a diagnosis of terminal illness and that you have to be deemed mentally competent. You can’t use this if you have Alzheimer’s or severe depression. You have to make a request twice with a week or two in between to make sure that your request is authentic. And obviously, everyone is on board to make sure that you’re not being coerced or pushed somehow into requesting a somewhat earlier death than you would have experienced without having the availability of the pills.
You also have to take the pills yourself or be able to pull a switch so that you could use a feeding tube–type administration. If you can’t do that, say due to ALS, you’re not eligible to use medical aid in dying. It’s a pretty restricted intervention.
Many people who get pills after going through these restrictions in the states that permit it don’t use it. As many as one third say they like having it there as a safety valve or a parachute, but once they know they could end their life sooner, then they’re going to stick it out.
Should states make this legal? New York, Massachusetts, Florida, and many other states have bills that are moving through. I’m going to say yes. We’ve had Oregon and Washington since the late 1990s with medical aid in dying on the books. There doesn’t seem to be any evidence of pushing people to use this, of bias against the disabled, or bigotry against particular ethnic or racial groups being used to encourage people to end their life sooner.
I think it is an option that Americans want. I think it’s an option that makes some sense. I’m well aware that we also have to make sure that people know about hospice. In some of these states, medical aid in dying is offered as a part of hospice — not all, but a few. Not everybody wants hospice once they realize that they’re dying and that it is coming relatively soon. They may want to leave with family present, with a ceremony, or with a quality of life that they desire.
Past experience says let’s continue to expand availability in each state. Let’s also realize that we have to keep the restrictions in place on how it’s used because they have protected us against abuse. Let’s understand that every doctor has an option to do this or not do this. It’s a matter of conscience and a matter of comfort.
I think legalization is the direction we’re going to be going in. Getting rid of the residency requirements that have been around, as I think courts are going to overturn them, also gives a push to the idea that once the service is in this many states, it’s something that should be available if there are doctors willing to do it.
I’m Art Caplan at the Division of Medical Ethics at NYU Grossman School of Medicine. New York, NY. Thank you for watching.
Arthur L. Caplan, PhD, has disclosed the following relevant financial relationships:
- Served as a director, officer, partner, employee, advisor, consultant, or trustee for: Johnson & Johnson’s Panel for Compassionate Drug Use (unpaid position)
- Serves as a contributing author and adviser for: Medscape
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
Hi. I’m Art Caplan. I’m at the Division of Medical Ethics at the NYU Grossman School of Medicine.
Right now, there are 10 states and the District of Columbia that have had some version of medical assistance in dying approved and on the books. That basically means that about 20% of Americans have access where they live to a physician who can prescribe a lethal dose of medication to them if they’re terminally ill and can ingest the medication themselves. That leaves many Americans not covered by this kind of access to this kind of service.
Many of you watching this may live in states where it is legal, like Oregon, Washington, New Jersey, Colorado, and Hawaii. I know many doctors say, “I’m not going to do that.” It’s not something that anyone is compelling a doctor to do. For some Americans, access is not just about where they live but whether there is a doctor willing to participate with them in bringing about their accelerated death, knowing that they’re inevitably going to die.
There’s not much we can do about that. It’s up to the conscience of each physician as to what they’re comfortable with. Certainly, there are other things that can be done to extend the possibility of having this available.
One thing that’s taking place is that, after lawsuits were filed, Vermont and Oregon have given up on their residency requirement, so you don’t have to be there 6 months or a year in order to use this opportunity. It’s legal now to move to the state or visit the state, and as soon as you get there, sign up for this kind of end-of-life intervention.
New Jersey is also being sued. I’ll predict that every state that has a residency requirement, when sued in court, is going to lose because we’ve long recognized the right of Americans to seek out healthcare in the United States, wherever they want to go.
If some states have made this a legitimate medical procedure, courts are going to say you can’t restrict it only to state residents. If someone wants to use a service, they’re entitled to show up from another state or another place and use it. I’m not sure about foreign nationals, but I’m very sure that Americans can go state to state in search of legitimate medical procedures.
The other bills that are out there, however, are basically saying they want to emulate Oregon, Washington, and the other states and say that the terminally ill, with severe restrictions, are going to be able to get this service without going anywhere.
The restrictions include a diagnosis of terminal illness and that you have to be deemed mentally competent. You can’t use this if you have Alzheimer’s or severe depression. You have to make a request twice with a week or two in between to make sure that your request is authentic. And obviously, everyone is on board to make sure that you’re not being coerced or pushed somehow into requesting a somewhat earlier death than you would have experienced without having the availability of the pills.
You also have to take the pills yourself or be able to pull a switch so that you could use a feeding tube–type administration. If you can’t do that, say due to ALS, you’re not eligible to use medical aid in dying. It’s a pretty restricted intervention.
Many people who get pills after going through these restrictions in the states that permit it don’t use it. As many as one third say they like having it there as a safety valve or a parachute, but once they know they could end their life sooner, then they’re going to stick it out.
Should states make this legal? New York, Massachusetts, Florida, and many other states have bills that are moving through. I’m going to say yes. We’ve had Oregon and Washington since the late 1990s with medical aid in dying on the books. There doesn’t seem to be any evidence of pushing people to use this, of bias against the disabled, or bigotry against particular ethnic or racial groups being used to encourage people to end their life sooner.
I think it is an option that Americans want. I think it’s an option that makes some sense. I’m well aware that we also have to make sure that people know about hospice. In some of these states, medical aid in dying is offered as a part of hospice — not all, but a few. Not everybody wants hospice once they realize that they’re dying and that it is coming relatively soon. They may want to leave with family present, with a ceremony, or with a quality of life that they desire.
Past experience says let’s continue to expand availability in each state. Let’s also realize that we have to keep the restrictions in place on how it’s used because they have protected us against abuse. Let’s understand that every doctor has an option to do this or not do this. It’s a matter of conscience and a matter of comfort.
I think legalization is the direction we’re going to be going in. Getting rid of the residency requirements that have been around, as I think courts are going to overturn them, also gives a push to the idea that once the service is in this many states, it’s something that should be available if there are doctors willing to do it.
I’m Art Caplan at the Division of Medical Ethics at NYU Grossman School of Medicine. New York, NY. Thank you for watching.
Arthur L. Caplan, PhD, has disclosed the following relevant financial relationships:
- Served as a director, officer, partner, employee, advisor, consultant, or trustee for: Johnson & Johnson’s Panel for Compassionate Drug Use (unpaid position)
- Serves as a contributing author and adviser for: Medscape
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
Hi. I’m Art Caplan. I’m at the Division of Medical Ethics at the NYU Grossman School of Medicine.
Right now, there are 10 states and the District of Columbia that have had some version of medical assistance in dying approved and on the books. That basically means that about 20% of Americans have access where they live to a physician who can prescribe a lethal dose of medication to them if they’re terminally ill and can ingest the medication themselves. That leaves many Americans not covered by this kind of access to this kind of service.
Many of you watching this may live in states where it is legal, like Oregon, Washington, New Jersey, Colorado, and Hawaii. I know many doctors say, “I’m not going to do that.” It’s not something that anyone is compelling a doctor to do. For some Americans, access is not just about where they live but whether there is a doctor willing to participate with them in bringing about their accelerated death, knowing that they’re inevitably going to die.
There’s not much we can do about that. It’s up to the conscience of each physician as to what they’re comfortable with. Certainly, there are other things that can be done to extend the possibility of having this available.
One thing that’s taking place is that, after lawsuits were filed, Vermont and Oregon have given up on their residency requirement, so you don’t have to be there 6 months or a year in order to use this opportunity. It’s legal now to move to the state or visit the state, and as soon as you get there, sign up for this kind of end-of-life intervention.
New Jersey is also being sued. I’ll predict that every state that has a residency requirement, when sued in court, is going to lose because we’ve long recognized the right of Americans to seek out healthcare in the United States, wherever they want to go.
If some states have made this a legitimate medical procedure, courts are going to say you can’t restrict it only to state residents. If someone wants to use a service, they’re entitled to show up from another state or another place and use it. I’m not sure about foreign nationals, but I’m very sure that Americans can go state to state in search of legitimate medical procedures.
The other bills that are out there, however, are basically saying they want to emulate Oregon, Washington, and the other states and say that the terminally ill, with severe restrictions, are going to be able to get this service without going anywhere.
The restrictions include a diagnosis of terminal illness and that you have to be deemed mentally competent. You can’t use this if you have Alzheimer’s or severe depression. You have to make a request twice with a week or two in between to make sure that your request is authentic. And obviously, everyone is on board to make sure that you’re not being coerced or pushed somehow into requesting a somewhat earlier death than you would have experienced without having the availability of the pills.
You also have to take the pills yourself or be able to pull a switch so that you could use a feeding tube–type administration. If you can’t do that, say due to ALS, you’re not eligible to use medical aid in dying. It’s a pretty restricted intervention.
Many people who get pills after going through these restrictions in the states that permit it don’t use it. As many as one third say they like having it there as a safety valve or a parachute, but once they know they could end their life sooner, then they’re going to stick it out.
Should states make this legal? New York, Massachusetts, Florida, and many other states have bills that are moving through. I’m going to say yes. We’ve had Oregon and Washington since the late 1990s with medical aid in dying on the books. There doesn’t seem to be any evidence of pushing people to use this, of bias against the disabled, or bigotry against particular ethnic or racial groups being used to encourage people to end their life sooner.
I think it is an option that Americans want. I think it’s an option that makes some sense. I’m well aware that we also have to make sure that people know about hospice. In some of these states, medical aid in dying is offered as a part of hospice — not all, but a few. Not everybody wants hospice once they realize that they’re dying and that it is coming relatively soon. They may want to leave with family present, with a ceremony, or with a quality of life that they desire.
Past experience says let’s continue to expand availability in each state. Let’s also realize that we have to keep the restrictions in place on how it’s used because they have protected us against abuse. Let’s understand that every doctor has an option to do this or not do this. It’s a matter of conscience and a matter of comfort.
I think legalization is the direction we’re going to be going in. Getting rid of the residency requirements that have been around, as I think courts are going to overturn them, also gives a push to the idea that once the service is in this many states, it’s something that should be available if there are doctors willing to do it.
I’m Art Caplan at the Division of Medical Ethics at NYU Grossman School of Medicine. New York, NY. Thank you for watching.
Arthur L. Caplan, PhD, has disclosed the following relevant financial relationships:
- Served as a director, officer, partner, employee, advisor, consultant, or trustee for: Johnson & Johnson’s Panel for Compassionate Drug Use (unpaid position)
- Serves as a contributing author and adviser for: Medscape
A version of this article appeared on Medscape.com.
The Breakthrough Drug Whose Full Promise Remains Unrealized
Celebrating a Decade of Sofosbuvir for Hepatitis C
Prior to 2013, the backbone of hepatitis C virus (HCV) therapy was pegylated interferon (PEG) in combination with ribavirin (RBV). This year-long therapy was associated with significant side effects and abysmal cure rates. Although efficacy improved with the addition of first-generation protease inhibitors, cure rates remained suboptimal and treatment side effects continued to be significant.
Clinicians and patients needed better options and looked to the drug pipeline with hope. However, even among the most optimistic, the idea that HCV therapy could evolve into an all-oral option seemed a relative pipe dream.
The Sofosbuvir Revolution Begins
The Liver Meeting held in 2013 changed everything.
Several presentations featured compelling data with sofosbuvir, a new polymerase inhibitor that, when combined with RBV, offered an all-oral option to patients with genotypes 2 and 3, as well as improved efficacy for patients with genotypes 1, 4, 5, and 6 when it was combined with 12 weeks of PEG/RBV.
However, the glass ceiling of HCV care was truly shattered with the randomized COSMOS trial, a late-breaker abstract that revealed 12-week functional cure rates in patients receiving sofosbuvir in combination with the protease inhibitor simeprevir.
This phase 2a trial in treatment-naive and -experienced genotype 1 patients with and without cirrhosis showed that an all-oral option was not only viable for the most common strain of HCV but was also safe and efficacious, even in difficult-to-treat populations.
On December 6, 2013, the US Food and Drug Administration (FDA) approved sofosbuvir for the treatment of HCV, ushering in a new era of therapy.
Guidelines quickly changed to advocate for both expansive HCV screening and generous treatment. Yet, as this more permissive approach was being recommended, the high price tag and large anticipated volume of those seeking prescriptions were setting off alarms. The drug cost triggered extensive restrictions based on degree of fibrosis, sobriety, and provider type in an effort to prevent immediate healthcare expenditures.
Given its high cost, rules restricting a patient to only one course of sofosbuvir-based therapy also surfaced. Although treatment with first-generation protease inhibitors carried a hefty price of $161,813.49 per sustained virologic response (SVR), compared with $66,000-$100,000 for 12 weeks of all-oral therapy, its uptake was low and limited by side effects and comorbid conditions. All-oral treatment appeared to have few medical barriers, leading payers to find ways to slow utilization. These restrictions are now gradually being eliminated.
Because of high SVR rates and few contraindications to therapy, most patients who gained access to treatment achieved cure. This included patients who had previously not responded to treatment and prioritized those with more advanced disease.
This quickly led to a significant shift in the population in need of treatment. Prior to 2013, many patients with HCV had advanced disease and did not respond to prior treatment options. After uptake of all-oral therapy, individuals in need were typically treatment naive without advanced disease.
This shift also added new psychosocial dimensions, as many of the newly infected individuals were struggling with active substance abuse. HCV treatment providers needed to change, with increasing recruitment of advanced practice providers, primary care physicians, and addiction medication specialists.
Progress, but Far From Reaching Targets
Fast-forward to 2023.
Ten years after FDA approval, 13.2 million individuals infected with HCV have been treated globally, 82% with sofosbuvir-based regimens and most in lower-middle-income countries. This is absolutely cause for celebration, but not complacency.
In 2016, the World Health Assembly adopted a resolution of elimination of viral hepatitis by 2030. The World Health Organization (WHO) defined elimination of HCV as 90% reduction in new cases of infection, 90% diagnosis of those infected, 80% of eligible individuals treated, and 65% reduction of deaths by 2030.
Despite all the success thus far, the CDA Foundation estimates that the WHO elimination targets will not be achieved until after the year 2050. They also note that in 2020, over 50 million individuals were infected with HCV, of which only 20% were diagnosed and 1% annually treated.
The HCV care cascade, by which the patient journeys from screening to cure, is complicated, and a one-size-fits-all solution is not possible. Reflex testing (an automatic transition to HCV polymerase chain reaction [PCR] testing in the lab for those who are HCV antibody positive) has significantly improved diagnosis. However, communicating these results and linking a patient to curative therapy remain significant obstacles.
Models and real-life experience show that multiple strategies can be successful. They include leveraging the electronic medical record, simplified treatment algorithms, test-and-treat strategies (screening high-risk populations with a point-of-care test that allows treatment initiation at the same visit), and co-localizing HCV screening and treatment with addiction services and relinkage programs (finding those who are already diagnosed and linking them to treatment).
In addition, focusing on populations at high risk for HCV infection — such as people who inject drugs, men who have sex with men, and incarcerated individuals — allows for better resource utilization.
Though daunting, HCV elimination is not impossible. There are several examples of success, including in the countries of Georgia and Iceland. Although, comparatively, the United States remains behind the curve, the White House has asked Congress for $11 billion to fund HCV elimination domestically.
As we await action at the national level, clinicians are reminded that there are several things we can do in caring for patients with HCV:
- A one-time HCV screening is recommended in all individuals aged 18 or older, including pregnant people with each pregnancy.
- HCV antibody testing with reflex to PCR should be used as the screening test.
- Pan-genotypic all-oral therapy is recommended for patients with HCV. Cure rates are greater than 95%, and there are few contraindications to treatment.
- Most people are eligible for simplified treatment algorithms that allow minimal on-treatment monitoring.
Without increased screening and linkage to curative therapy, we will not meet the WHO goals for HCV elimination.
Dr. Reau is chief of the hepatology section at Rush University Medical Center in Chicago and a regular contributor to this news organization. She serves as editor of Clinical Liver Disease, a multimedia review journal, and recently as a member of HCVGuidelines.org, a web-based resource from the American Association for the Study of Liver Diseases (AASLD) and the Infectious Diseases Society of America, as well as educational chair of the AASLD hepatitis C special interest group. She continues to have an active role in the hepatology interest group of the World Gastroenterology Organisation and the American Liver Foundation at the regional and national levels. She disclosed ties with AbbVie, Gilead, Arbutus, Intercept, and Salix.
A version of this article appeared on Medscape.com.
Celebrating a Decade of Sofosbuvir for Hepatitis C
Celebrating a Decade of Sofosbuvir for Hepatitis C
Prior to 2013, the backbone of hepatitis C virus (HCV) therapy was pegylated interferon (PEG) in combination with ribavirin (RBV). This year-long therapy was associated with significant side effects and abysmal cure rates. Although efficacy improved with the addition of first-generation protease inhibitors, cure rates remained suboptimal and treatment side effects continued to be significant.
Clinicians and patients needed better options and looked to the drug pipeline with hope. However, even among the most optimistic, the idea that HCV therapy could evolve into an all-oral option seemed a relative pipe dream.
The Sofosbuvir Revolution Begins
The Liver Meeting held in 2013 changed everything.
Several presentations featured compelling data with sofosbuvir, a new polymerase inhibitor that, when combined with RBV, offered an all-oral option to patients with genotypes 2 and 3, as well as improved efficacy for patients with genotypes 1, 4, 5, and 6 when it was combined with 12 weeks of PEG/RBV.
However, the glass ceiling of HCV care was truly shattered with the randomized COSMOS trial, a late-breaker abstract that revealed 12-week functional cure rates in patients receiving sofosbuvir in combination with the protease inhibitor simeprevir.
This phase 2a trial in treatment-naive and -experienced genotype 1 patients with and without cirrhosis showed that an all-oral option was not only viable for the most common strain of HCV but was also safe and efficacious, even in difficult-to-treat populations.
On December 6, 2013, the US Food and Drug Administration (FDA) approved sofosbuvir for the treatment of HCV, ushering in a new era of therapy.
Guidelines quickly changed to advocate for both expansive HCV screening and generous treatment. Yet, as this more permissive approach was being recommended, the high price tag and large anticipated volume of those seeking prescriptions were setting off alarms. The drug cost triggered extensive restrictions based on degree of fibrosis, sobriety, and provider type in an effort to prevent immediate healthcare expenditures.
Given its high cost, rules restricting a patient to only one course of sofosbuvir-based therapy also surfaced. Although treatment with first-generation protease inhibitors carried a hefty price of $161,813.49 per sustained virologic response (SVR), compared with $66,000-$100,000 for 12 weeks of all-oral therapy, its uptake was low and limited by side effects and comorbid conditions. All-oral treatment appeared to have few medical barriers, leading payers to find ways to slow utilization. These restrictions are now gradually being eliminated.
Because of high SVR rates and few contraindications to therapy, most patients who gained access to treatment achieved cure. This included patients who had previously not responded to treatment and prioritized those with more advanced disease.
This quickly led to a significant shift in the population in need of treatment. Prior to 2013, many patients with HCV had advanced disease and did not respond to prior treatment options. After uptake of all-oral therapy, individuals in need were typically treatment naive without advanced disease.
This shift also added new psychosocial dimensions, as many of the newly infected individuals were struggling with active substance abuse. HCV treatment providers needed to change, with increasing recruitment of advanced practice providers, primary care physicians, and addiction medication specialists.
Progress, but Far From Reaching Targets
Fast-forward to 2023.
Ten years after FDA approval, 13.2 million individuals infected with HCV have been treated globally, 82% with sofosbuvir-based regimens and most in lower-middle-income countries. This is absolutely cause for celebration, but not complacency.
In 2016, the World Health Assembly adopted a resolution of elimination of viral hepatitis by 2030. The World Health Organization (WHO) defined elimination of HCV as 90% reduction in new cases of infection, 90% diagnosis of those infected, 80% of eligible individuals treated, and 65% reduction of deaths by 2030.
Despite all the success thus far, the CDA Foundation estimates that the WHO elimination targets will not be achieved until after the year 2050. They also note that in 2020, over 50 million individuals were infected with HCV, of which only 20% were diagnosed and 1% annually treated.
The HCV care cascade, by which the patient journeys from screening to cure, is complicated, and a one-size-fits-all solution is not possible. Reflex testing (an automatic transition to HCV polymerase chain reaction [PCR] testing in the lab for those who are HCV antibody positive) has significantly improved diagnosis. However, communicating these results and linking a patient to curative therapy remain significant obstacles.
Models and real-life experience show that multiple strategies can be successful. They include leveraging the electronic medical record, simplified treatment algorithms, test-and-treat strategies (screening high-risk populations with a point-of-care test that allows treatment initiation at the same visit), and co-localizing HCV screening and treatment with addiction services and relinkage programs (finding those who are already diagnosed and linking them to treatment).
In addition, focusing on populations at high risk for HCV infection — such as people who inject drugs, men who have sex with men, and incarcerated individuals — allows for better resource utilization.
Though daunting, HCV elimination is not impossible. There are several examples of success, including in the countries of Georgia and Iceland. Although, comparatively, the United States remains behind the curve, the White House has asked Congress for $11 billion to fund HCV elimination domestically.
As we await action at the national level, clinicians are reminded that there are several things we can do in caring for patients with HCV:
- A one-time HCV screening is recommended in all individuals aged 18 or older, including pregnant people with each pregnancy.
- HCV antibody testing with reflex to PCR should be used as the screening test.
- Pan-genotypic all-oral therapy is recommended for patients with HCV. Cure rates are greater than 95%, and there are few contraindications to treatment.
- Most people are eligible for simplified treatment algorithms that allow minimal on-treatment monitoring.
Without increased screening and linkage to curative therapy, we will not meet the WHO goals for HCV elimination.
Dr. Reau is chief of the hepatology section at Rush University Medical Center in Chicago and a regular contributor to this news organization. She serves as editor of Clinical Liver Disease, a multimedia review journal, and recently as a member of HCVGuidelines.org, a web-based resource from the American Association for the Study of Liver Diseases (AASLD) and the Infectious Diseases Society of America, as well as educational chair of the AASLD hepatitis C special interest group. She continues to have an active role in the hepatology interest group of the World Gastroenterology Organisation and the American Liver Foundation at the regional and national levels. She disclosed ties with AbbVie, Gilead, Arbutus, Intercept, and Salix.
A version of this article appeared on Medscape.com.
Prior to 2013, the backbone of hepatitis C virus (HCV) therapy was pegylated interferon (PEG) in combination with ribavirin (RBV). This year-long therapy was associated with significant side effects and abysmal cure rates. Although efficacy improved with the addition of first-generation protease inhibitors, cure rates remained suboptimal and treatment side effects continued to be significant.
Clinicians and patients needed better options and looked to the drug pipeline with hope. However, even among the most optimistic, the idea that HCV therapy could evolve into an all-oral option seemed a relative pipe dream.
The Sofosbuvir Revolution Begins
The Liver Meeting held in 2013 changed everything.
Several presentations featured compelling data with sofosbuvir, a new polymerase inhibitor that, when combined with RBV, offered an all-oral option to patients with genotypes 2 and 3, as well as improved efficacy for patients with genotypes 1, 4, 5, and 6 when it was combined with 12 weeks of PEG/RBV.
However, the glass ceiling of HCV care was truly shattered with the randomized COSMOS trial, a late-breaker abstract that revealed 12-week functional cure rates in patients receiving sofosbuvir in combination with the protease inhibitor simeprevir.
This phase 2a trial in treatment-naive and -experienced genotype 1 patients with and without cirrhosis showed that an all-oral option was not only viable for the most common strain of HCV but was also safe and efficacious, even in difficult-to-treat populations.
On December 6, 2013, the US Food and Drug Administration (FDA) approved sofosbuvir for the treatment of HCV, ushering in a new era of therapy.
Guidelines quickly changed to advocate for both expansive HCV screening and generous treatment. Yet, as this more permissive approach was being recommended, the high price tag and large anticipated volume of those seeking prescriptions were setting off alarms. The drug cost triggered extensive restrictions based on degree of fibrosis, sobriety, and provider type in an effort to prevent immediate healthcare expenditures.
Given its high cost, rules restricting a patient to only one course of sofosbuvir-based therapy also surfaced. Although treatment with first-generation protease inhibitors carried a hefty price of $161,813.49 per sustained virologic response (SVR), compared with $66,000-$100,000 for 12 weeks of all-oral therapy, its uptake was low and limited by side effects and comorbid conditions. All-oral treatment appeared to have few medical barriers, leading payers to find ways to slow utilization. These restrictions are now gradually being eliminated.
Because of high SVR rates and few contraindications to therapy, most patients who gained access to treatment achieved cure. This included patients who had previously not responded to treatment and prioritized those with more advanced disease.
This quickly led to a significant shift in the population in need of treatment. Prior to 2013, many patients with HCV had advanced disease and did not respond to prior treatment options. After uptake of all-oral therapy, individuals in need were typically treatment naive without advanced disease.
This shift also added new psychosocial dimensions, as many of the newly infected individuals were struggling with active substance abuse. HCV treatment providers needed to change, with increasing recruitment of advanced practice providers, primary care physicians, and addiction medication specialists.
Progress, but Far From Reaching Targets
Fast-forward to 2023.
Ten years after FDA approval, 13.2 million individuals infected with HCV have been treated globally, 82% with sofosbuvir-based regimens and most in lower-middle-income countries. This is absolutely cause for celebration, but not complacency.
In 2016, the World Health Assembly adopted a resolution of elimination of viral hepatitis by 2030. The World Health Organization (WHO) defined elimination of HCV as 90% reduction in new cases of infection, 90% diagnosis of those infected, 80% of eligible individuals treated, and 65% reduction of deaths by 2030.
Despite all the success thus far, the CDA Foundation estimates that the WHO elimination targets will not be achieved until after the year 2050. They also note that in 2020, over 50 million individuals were infected with HCV, of which only 20% were diagnosed and 1% annually treated.
The HCV care cascade, by which the patient journeys from screening to cure, is complicated, and a one-size-fits-all solution is not possible. Reflex testing (an automatic transition to HCV polymerase chain reaction [PCR] testing in the lab for those who are HCV antibody positive) has significantly improved diagnosis. However, communicating these results and linking a patient to curative therapy remain significant obstacles.
Models and real-life experience show that multiple strategies can be successful. They include leveraging the electronic medical record, simplified treatment algorithms, test-and-treat strategies (screening high-risk populations with a point-of-care test that allows treatment initiation at the same visit), and co-localizing HCV screening and treatment with addiction services and relinkage programs (finding those who are already diagnosed and linking them to treatment).
In addition, focusing on populations at high risk for HCV infection — such as people who inject drugs, men who have sex with men, and incarcerated individuals — allows for better resource utilization.
Though daunting, HCV elimination is not impossible. There are several examples of success, including in the countries of Georgia and Iceland. Although, comparatively, the United States remains behind the curve, the White House has asked Congress for $11 billion to fund HCV elimination domestically.
As we await action at the national level, clinicians are reminded that there are several things we can do in caring for patients with HCV:
- A one-time HCV screening is recommended in all individuals aged 18 or older, including pregnant people with each pregnancy.
- HCV antibody testing with reflex to PCR should be used as the screening test.
- Pan-genotypic all-oral therapy is recommended for patients with HCV. Cure rates are greater than 95%, and there are few contraindications to treatment.
- Most people are eligible for simplified treatment algorithms that allow minimal on-treatment monitoring.
Without increased screening and linkage to curative therapy, we will not meet the WHO goals for HCV elimination.
Dr. Reau is chief of the hepatology section at Rush University Medical Center in Chicago and a regular contributor to this news organization. She serves as editor of Clinical Liver Disease, a multimedia review journal, and recently as a member of HCVGuidelines.org, a web-based resource from the American Association for the Study of Liver Diseases (AASLD) and the Infectious Diseases Society of America, as well as educational chair of the AASLD hepatitis C special interest group. She continues to have an active role in the hepatology interest group of the World Gastroenterology Organisation and the American Liver Foundation at the regional and national levels. She disclosed ties with AbbVie, Gilead, Arbutus, Intercept, and Salix.
A version of this article appeared on Medscape.com.
A Military Nurse Saves a Life After a Brutal Rollover Crash
Emergencies happen anywhere and anytime, and sometimes, medical professionals find themselves in situations where they are the only ones who can help. Is There a Doctor in the House? is a series telling these stories.
A week earlier I’d had a heart surgery and was heading out for a post-op appointment when I saw it: I had a flat tire. It didn’t make sense. The tire was brand new, and there was no puncture. But it was flat.
I swapped out the flat for the spare and went off base to a tire shop. While I was there, my surgeon’s office called and rescheduled my appointment for a couple of hours later. That was lucky because by the time the tire was fixed, I had just enough time to get there.
The hospital is right near I-35 in San Antonio, Texas. I got off the freeway and onto the access road and paused to turn into the parking lot. That’s when I heard an enormous crash.
I saw a big poof of white smoke, and a car barreled off the freeway and came rolling down the embankment.
When the car hit the access road, I saw a woman ejected through the windshield. She bounced and landed in the road about 25 feet in front of me.
I put my car in park, grabbed my face mask and gloves, and started running toward her. But another vehicle — a truck towing a trailer — came from behind to drive around me. The driver didn’t realize what had happened and couldn’t stop in time…
The trailer ran over her.
I didn’t know if anyone could’ve survived that, but I went to her. I saw several other bystanders, but they were frozen in shock. I was praying, dear God, if she’s alive, let me do whatever I need to do to save her life.
It was a horrible scene. This poor lady was in a bloody heap in the middle of the road. Her right arm was twisted up under her neck so tightly, she was choking herself. So, the first thing I did was straighten her arm out to protect her airway.
I started yelling at people, “Call 9-1-1! Run to the hospital! Let them know there’s an accident out here, and I need help!”
The woman had a pulse, but it was super rapid. On first glance, she clearly had multiple fractures and a bad head bleed. With the sheer number of times she’d been injured, I didn’t know what was going on internally, but it was bad. She was gargling on her own blood and spitting it up. She was drowning.
A couple of technicians from the hospital came and brought me a tiny emergency kit. It had a blood pressure cuff and an oral airway. All the vital signs indicated the lady was going into shock. She’d lost a lot of blood on the pavement.
I was able to get the oral airway in. A few minutes later, a fire chief showed up. By now, the traffic had backed up so badly, the emergency vehicles couldn’t get in. But he managed to get there another way and gave me a cervical collar (C collar) and an Ambu bag.
I was hyper-focused on what I could do at that moment and what I needed to do next. Her stats were going down, but she still had a pulse. If she lost the pulse or went into a lethal rhythm, I’d have to start cardiopulmonary resuscitation (CPR). I asked the other people, but nobody else knew CPR, so I wouldn’t have help.
I could tell the lady had a pelvic fracture, and we needed to stabilize her. I directed people how to hold her neck safely and log-roll her flat on the ground. I also needed to put pressure on the back of her head because of all the bleeding. I got people to give me their clothes and tried to do that as I was bagging her.
The windows of her vehicle had all been blown out. I asked somebody to go find her purse with her ID. Then I noticed something …
My heart jumped into my stomach.
A car seat. There was an empty child’s car seat in the back of the car.
I started yelling at everyone, “Look for a baby! Go up and down the embankment and across the road. There might have been a baby in the car!”
But there wasn’t. Thank God. She hadn’t been driving with her child.
At that point, a paramedic came running from behind all the traffic. We did life support together until the ambulance finally arrived.
Emergency medical services got an intravenous line in and used medical anti-shock trousers. Thankfully, I already had the C collar on, and we’d been bagging her, so they could load her very quickly.
I got rid of my bloody gloves. I told a police officer I would come back. And then I went to my doctor’s appointment.
The window at my doctor’s office faced the access road, so the people there had seen all the traffic. They asked me what happened, and I said, “It was me. I saw it happen. I tried to help.” I was a little frazzled.
When I got back to the scene, the police and the fire chief kept thanking me for stopping. Why wouldn’t I stop? It was astounding to realize that they imagined somebody wouldn’t stop in a situation like this.
They told me the lady was alive. She was in the intensive care unit in critical condition, but she had survived. At that moment, I had this overwhelming feeling: God had put me in this exact place at the exact time to save her life.
Looking back, I think about how God ordered my steps. Without the mysterious flat tire, I would’ve gone to the hospital earlier. If my appointment hadn’t been rescheduled, I wouldn’t have been on the access road. All those events brought me there.
Several months later, the woman’s family contacted me and asked if we could meet. I found out more about her injuries. She’d had multiple skull fractures, facial fractures, and a broken jaw. Her upper arm was broken in three places. Her clavicle was broken. She had internal bleeding, a pelvic fracture, and a broken leg. She was 28 years old.
She’d had multiple surgeries, spent 2 months in the ICU, and another 3 months in intensive rehab. But she survived. It was incredible.
We all met up at a McDonald’s. First, her little son — who was the baby I thought might have been in the car — ran up to me and said, “Thank you for saving my mommy’s life.”
Then I turned, and there she was — a beautiful lady looking at me with awe and crying, saying, “It’s me.”
She obviously had gone through a transformation from all the injuries and the medications. She had a little bit of a speech delay, but mentally, she was there. She could walk.
She said, “You’re my angel. God put you there to save my life.” Her family all came up and hugged me. It was so beautiful.
She told me about the accident. She’d been speeding that day, zigzagging through lanes to get around the traffic. And she didn’t have her seatbelt on. She’d driven onto the shoulder to try to pass everyone, but it started narrowing. She clipped somebody’s bumper, went into a tailspin, and collided with a second vehicle, which caused her to flip over and down the embankment.
“God’s given me a new lease on life,” she said, “a fresh start. I will forever wear my seatbelt. And I’m going to do whatever I can to give back to other people because I don’t even feel like I deserve this.”
I just cried.
I’ve been a nurse for 29 years, first on the civilian side and later in the military. I’ve led codes and responded to trauma in a hospital setting or a deployed environment. I was well prepared to do what I did. But doing it under such stress with adrenaline bombarding me ... I’m amazed. I just think God’s hand was on me.
At that time, I was personally going through some things. After my heart surgery, I was in an emotional place where I didn’t feel loved or valued. But when I had that realization — when I knew that I was meant to be there to save her life, I also got the very clear message that I was valued and loved so much.
I know I have a very strong purpose. That day changed my life.
US Air Force Lt. Col. Anne Staley is the officer in charge of the Military Training Network, a division of the Defense Health Agency Education and Training Directorate in San Antonio, Texas.
A version of this article appeared on Medscape.com.
Emergencies happen anywhere and anytime, and sometimes, medical professionals find themselves in situations where they are the only ones who can help. Is There a Doctor in the House? is a series telling these stories.
A week earlier I’d had a heart surgery and was heading out for a post-op appointment when I saw it: I had a flat tire. It didn’t make sense. The tire was brand new, and there was no puncture. But it was flat.
I swapped out the flat for the spare and went off base to a tire shop. While I was there, my surgeon’s office called and rescheduled my appointment for a couple of hours later. That was lucky because by the time the tire was fixed, I had just enough time to get there.
The hospital is right near I-35 in San Antonio, Texas. I got off the freeway and onto the access road and paused to turn into the parking lot. That’s when I heard an enormous crash.
I saw a big poof of white smoke, and a car barreled off the freeway and came rolling down the embankment.
When the car hit the access road, I saw a woman ejected through the windshield. She bounced and landed in the road about 25 feet in front of me.
I put my car in park, grabbed my face mask and gloves, and started running toward her. But another vehicle — a truck towing a trailer — came from behind to drive around me. The driver didn’t realize what had happened and couldn’t stop in time…
The trailer ran over her.
I didn’t know if anyone could’ve survived that, but I went to her. I saw several other bystanders, but they were frozen in shock. I was praying, dear God, if she’s alive, let me do whatever I need to do to save her life.
It was a horrible scene. This poor lady was in a bloody heap in the middle of the road. Her right arm was twisted up under her neck so tightly, she was choking herself. So, the first thing I did was straighten her arm out to protect her airway.
I started yelling at people, “Call 9-1-1! Run to the hospital! Let them know there’s an accident out here, and I need help!”
The woman had a pulse, but it was super rapid. On first glance, she clearly had multiple fractures and a bad head bleed. With the sheer number of times she’d been injured, I didn’t know what was going on internally, but it was bad. She was gargling on her own blood and spitting it up. She was drowning.
A couple of technicians from the hospital came and brought me a tiny emergency kit. It had a blood pressure cuff and an oral airway. All the vital signs indicated the lady was going into shock. She’d lost a lot of blood on the pavement.
I was able to get the oral airway in. A few minutes later, a fire chief showed up. By now, the traffic had backed up so badly, the emergency vehicles couldn’t get in. But he managed to get there another way and gave me a cervical collar (C collar) and an Ambu bag.
I was hyper-focused on what I could do at that moment and what I needed to do next. Her stats were going down, but she still had a pulse. If she lost the pulse or went into a lethal rhythm, I’d have to start cardiopulmonary resuscitation (CPR). I asked the other people, but nobody else knew CPR, so I wouldn’t have help.
I could tell the lady had a pelvic fracture, and we needed to stabilize her. I directed people how to hold her neck safely and log-roll her flat on the ground. I also needed to put pressure on the back of her head because of all the bleeding. I got people to give me their clothes and tried to do that as I was bagging her.
The windows of her vehicle had all been blown out. I asked somebody to go find her purse with her ID. Then I noticed something …
My heart jumped into my stomach.
A car seat. There was an empty child’s car seat in the back of the car.
I started yelling at everyone, “Look for a baby! Go up and down the embankment and across the road. There might have been a baby in the car!”
But there wasn’t. Thank God. She hadn’t been driving with her child.
At that point, a paramedic came running from behind all the traffic. We did life support together until the ambulance finally arrived.
Emergency medical services got an intravenous line in and used medical anti-shock trousers. Thankfully, I already had the C collar on, and we’d been bagging her, so they could load her very quickly.
I got rid of my bloody gloves. I told a police officer I would come back. And then I went to my doctor’s appointment.
The window at my doctor’s office faced the access road, so the people there had seen all the traffic. They asked me what happened, and I said, “It was me. I saw it happen. I tried to help.” I was a little frazzled.
When I got back to the scene, the police and the fire chief kept thanking me for stopping. Why wouldn’t I stop? It was astounding to realize that they imagined somebody wouldn’t stop in a situation like this.
They told me the lady was alive. She was in the intensive care unit in critical condition, but she had survived. At that moment, I had this overwhelming feeling: God had put me in this exact place at the exact time to save her life.
Looking back, I think about how God ordered my steps. Without the mysterious flat tire, I would’ve gone to the hospital earlier. If my appointment hadn’t been rescheduled, I wouldn’t have been on the access road. All those events brought me there.
Several months later, the woman’s family contacted me and asked if we could meet. I found out more about her injuries. She’d had multiple skull fractures, facial fractures, and a broken jaw. Her upper arm was broken in three places. Her clavicle was broken. She had internal bleeding, a pelvic fracture, and a broken leg. She was 28 years old.
She’d had multiple surgeries, spent 2 months in the ICU, and another 3 months in intensive rehab. But she survived. It was incredible.
We all met up at a McDonald’s. First, her little son — who was the baby I thought might have been in the car — ran up to me and said, “Thank you for saving my mommy’s life.”
Then I turned, and there she was — a beautiful lady looking at me with awe and crying, saying, “It’s me.”
She obviously had gone through a transformation from all the injuries and the medications. She had a little bit of a speech delay, but mentally, she was there. She could walk.
She said, “You’re my angel. God put you there to save my life.” Her family all came up and hugged me. It was so beautiful.
She told me about the accident. She’d been speeding that day, zigzagging through lanes to get around the traffic. And she didn’t have her seatbelt on. She’d driven onto the shoulder to try to pass everyone, but it started narrowing. She clipped somebody’s bumper, went into a tailspin, and collided with a second vehicle, which caused her to flip over and down the embankment.
“God’s given me a new lease on life,” she said, “a fresh start. I will forever wear my seatbelt. And I’m going to do whatever I can to give back to other people because I don’t even feel like I deserve this.”
I just cried.
I’ve been a nurse for 29 years, first on the civilian side and later in the military. I’ve led codes and responded to trauma in a hospital setting or a deployed environment. I was well prepared to do what I did. But doing it under such stress with adrenaline bombarding me ... I’m amazed. I just think God’s hand was on me.
At that time, I was personally going through some things. After my heart surgery, I was in an emotional place where I didn’t feel loved or valued. But when I had that realization — when I knew that I was meant to be there to save her life, I also got the very clear message that I was valued and loved so much.
I know I have a very strong purpose. That day changed my life.
US Air Force Lt. Col. Anne Staley is the officer in charge of the Military Training Network, a division of the Defense Health Agency Education and Training Directorate in San Antonio, Texas.
A version of this article appeared on Medscape.com.
Emergencies happen anywhere and anytime, and sometimes, medical professionals find themselves in situations where they are the only ones who can help. Is There a Doctor in the House? is a series telling these stories.
A week earlier I’d had a heart surgery and was heading out for a post-op appointment when I saw it: I had a flat tire. It didn’t make sense. The tire was brand new, and there was no puncture. But it was flat.
I swapped out the flat for the spare and went off base to a tire shop. While I was there, my surgeon’s office called and rescheduled my appointment for a couple of hours later. That was lucky because by the time the tire was fixed, I had just enough time to get there.
The hospital is right near I-35 in San Antonio, Texas. I got off the freeway and onto the access road and paused to turn into the parking lot. That’s when I heard an enormous crash.
I saw a big poof of white smoke, and a car barreled off the freeway and came rolling down the embankment.
When the car hit the access road, I saw a woman ejected through the windshield. She bounced and landed in the road about 25 feet in front of me.
I put my car in park, grabbed my face mask and gloves, and started running toward her. But another vehicle — a truck towing a trailer — came from behind to drive around me. The driver didn’t realize what had happened and couldn’t stop in time…
The trailer ran over her.
I didn’t know if anyone could’ve survived that, but I went to her. I saw several other bystanders, but they were frozen in shock. I was praying, dear God, if she’s alive, let me do whatever I need to do to save her life.
It was a horrible scene. This poor lady was in a bloody heap in the middle of the road. Her right arm was twisted up under her neck so tightly, she was choking herself. So, the first thing I did was straighten her arm out to protect her airway.
I started yelling at people, “Call 9-1-1! Run to the hospital! Let them know there’s an accident out here, and I need help!”
The woman had a pulse, but it was super rapid. On first glance, she clearly had multiple fractures and a bad head bleed. With the sheer number of times she’d been injured, I didn’t know what was going on internally, but it was bad. She was gargling on her own blood and spitting it up. She was drowning.
A couple of technicians from the hospital came and brought me a tiny emergency kit. It had a blood pressure cuff and an oral airway. All the vital signs indicated the lady was going into shock. She’d lost a lot of blood on the pavement.
I was able to get the oral airway in. A few minutes later, a fire chief showed up. By now, the traffic had backed up so badly, the emergency vehicles couldn’t get in. But he managed to get there another way and gave me a cervical collar (C collar) and an Ambu bag.
I was hyper-focused on what I could do at that moment and what I needed to do next. Her stats were going down, but she still had a pulse. If she lost the pulse or went into a lethal rhythm, I’d have to start cardiopulmonary resuscitation (CPR). I asked the other people, but nobody else knew CPR, so I wouldn’t have help.
I could tell the lady had a pelvic fracture, and we needed to stabilize her. I directed people how to hold her neck safely and log-roll her flat on the ground. I also needed to put pressure on the back of her head because of all the bleeding. I got people to give me their clothes and tried to do that as I was bagging her.
The windows of her vehicle had all been blown out. I asked somebody to go find her purse with her ID. Then I noticed something …
My heart jumped into my stomach.
A car seat. There was an empty child’s car seat in the back of the car.
I started yelling at everyone, “Look for a baby! Go up and down the embankment and across the road. There might have been a baby in the car!”
But there wasn’t. Thank God. She hadn’t been driving with her child.
At that point, a paramedic came running from behind all the traffic. We did life support together until the ambulance finally arrived.
Emergency medical services got an intravenous line in and used medical anti-shock trousers. Thankfully, I already had the C collar on, and we’d been bagging her, so they could load her very quickly.
I got rid of my bloody gloves. I told a police officer I would come back. And then I went to my doctor’s appointment.
The window at my doctor’s office faced the access road, so the people there had seen all the traffic. They asked me what happened, and I said, “It was me. I saw it happen. I tried to help.” I was a little frazzled.
When I got back to the scene, the police and the fire chief kept thanking me for stopping. Why wouldn’t I stop? It was astounding to realize that they imagined somebody wouldn’t stop in a situation like this.
They told me the lady was alive. She was in the intensive care unit in critical condition, but she had survived. At that moment, I had this overwhelming feeling: God had put me in this exact place at the exact time to save her life.
Looking back, I think about how God ordered my steps. Without the mysterious flat tire, I would’ve gone to the hospital earlier. If my appointment hadn’t been rescheduled, I wouldn’t have been on the access road. All those events brought me there.
Several months later, the woman’s family contacted me and asked if we could meet. I found out more about her injuries. She’d had multiple skull fractures, facial fractures, and a broken jaw. Her upper arm was broken in three places. Her clavicle was broken. She had internal bleeding, a pelvic fracture, and a broken leg. She was 28 years old.
She’d had multiple surgeries, spent 2 months in the ICU, and another 3 months in intensive rehab. But she survived. It was incredible.
We all met up at a McDonald’s. First, her little son — who was the baby I thought might have been in the car — ran up to me and said, “Thank you for saving my mommy’s life.”
Then I turned, and there she was — a beautiful lady looking at me with awe and crying, saying, “It’s me.”
She obviously had gone through a transformation from all the injuries and the medications. She had a little bit of a speech delay, but mentally, she was there. She could walk.
She said, “You’re my angel. God put you there to save my life.” Her family all came up and hugged me. It was so beautiful.
She told me about the accident. She’d been speeding that day, zigzagging through lanes to get around the traffic. And she didn’t have her seatbelt on. She’d driven onto the shoulder to try to pass everyone, but it started narrowing. She clipped somebody’s bumper, went into a tailspin, and collided with a second vehicle, which caused her to flip over and down the embankment.
“God’s given me a new lease on life,” she said, “a fresh start. I will forever wear my seatbelt. And I’m going to do whatever I can to give back to other people because I don’t even feel like I deserve this.”
I just cried.
I’ve been a nurse for 29 years, first on the civilian side and later in the military. I’ve led codes and responded to trauma in a hospital setting or a deployed environment. I was well prepared to do what I did. But doing it under such stress with adrenaline bombarding me ... I’m amazed. I just think God’s hand was on me.
At that time, I was personally going through some things. After my heart surgery, I was in an emotional place where I didn’t feel loved or valued. But when I had that realization — when I knew that I was meant to be there to save her life, I also got the very clear message that I was valued and loved so much.
I know I have a very strong purpose. That day changed my life.
US Air Force Lt. Col. Anne Staley is the officer in charge of the Military Training Network, a division of the Defense Health Agency Education and Training Directorate in San Antonio, Texas.
A version of this article appeared on Medscape.com.
Even Intentional Weight Loss Linked With Cancer
This transcript has been edited for clarity.
As anyone who has been through medical training will tell you, some little scenes just stick with you. I had been seeing a patient in our resident clinic in West Philly for a couple of years. She was in her mid-60s with diabetes and hypertension and a distant smoking history. She was overweight and had been trying to improve her diet and lose weight since I started seeing her. One day she came in and was delighted to report that she had finally started shedding some pounds — about 15 in the past 2 months.
I enthusiastically told my preceptor that my careful dietary counseling had finally done the job. She looked through the chart for a moment and asked, “Is she up to date on her cancer screening?” A workup revealed adenocarcinoma of the lung. The patient did well, actually, but the story stuck with me.
The textbooks call it “unintentional weight loss,” often in big, scary letters, and every doctor will go just a bit pale if a patient tells them that, despite efforts not to, they are losing weight. But true unintentional weight loss is not that common. After all, most of us are at least half-heartedly trying to lose weight all the time. Should doctors be worried when we are successful?
A new study suggests that perhaps they should. We’re talking about this study, appearing in JAMA, which combined participants from two long-running observational cohorts: 120,000 women from the Nurses’ Health Study, and 50,000 men from the Health Professionals Follow-Up Study. (These cohorts started in the 1970s and 1980s, so we’ll give them a pass on the gender-specific study designs.)
The rationale of enrolling healthcare providers in these cohort studies is that they would be reliable witnesses of their own health status. If a nurse or doctor says they have pancreatic cancer, it’s likely that they truly have pancreatic cancer. Detailed health surveys were distributed to the participants every other year, and the average follow-up was more than a decade.
Participants recorded their weight — as an aside, a nested study found that self-reported rate was extremely well correlated with professionally measured weight — and whether they had received a cancer diagnosis since the last survey.
This allowed researchers to look at the phenomenon described above. Would weight loss precede a new diagnosis of cancer? And, more interestingly, would intentional weight loss precede a new diagnosis of cancer.
I don’t think it will surprise you to hear that individuals in the highest category of weight loss, those who lost more than 10% of their body weight over a 2-year period, had a larger risk of being diagnosed with cancer in the next year. That’s the yellow line in this graph. In fact, they had about a 40% higher risk than those who did not lose weight.
Increased risk was found across multiple cancer types, though cancers of the gastrointestinal tract, not surprisingly, were most strongly associated with antecedent weight loss.
What about intentionality of weight loss? Unfortunately, the surveys did not ask participants whether they were trying to lose weight. Rather, the surveys asked about exercise and dietary habits. The researchers leveraged these responses to create three categories of participants: those who seemed to be trying to lose weight (defined as people who had increased their exercise and dietary quality); those who didn’t seem to be trying to lose weight (they changed neither exercise nor dietary behaviors); and a middle group, which changed one or the other of these behaviors but not both.
Let’s look at those who really seemed to be trying to lose weight. Over 2 years, they got more exercise and improved their diet.
If they succeeded in losing 10% or more of their body weight, they still had a higher risk for cancer than those who had not lost weight — about 30% higher, which is not that different from the 40% increased risk when you include those folks who weren’t changing their lifestyle.
This is why this study is important. The classic teaching is that unintentional weight loss is a bad thing and needs a workup. That’s fine. But we live in a world where perhaps the majority of people are, at any given time, trying to lose weight.
We need to be careful here. I am not by any means trying to say that people who have successfully lost weight have cancer. Both of the following statements can be true:
Significant weight loss, whether intentional or not, is associated with a higher risk for cancer.
Most people with significant weight loss will not have cancer.
Both of these can be true because cancer is, fortunately, rare. Of people who lose weight, the vast majority will lose weight because they are engaging in healthier behaviors. A small number may lose weight because something else is wrong. It’s just hard to tell the two apart.
Out of the nearly 200,000 people in this study, only around 16,000 developed cancer during follow-up. Again, although the chance of having cancer is slightly higher if someone has experienced weight loss, the chance is still very low.
We also need to avoid suggesting that weight loss causes cancer. Some people lose weight because of an existing, as of yet undiagnosed cancer and its metabolic effects. This is borne out if you look at the risk of being diagnosed with cancer as you move further away from the interval of weight loss.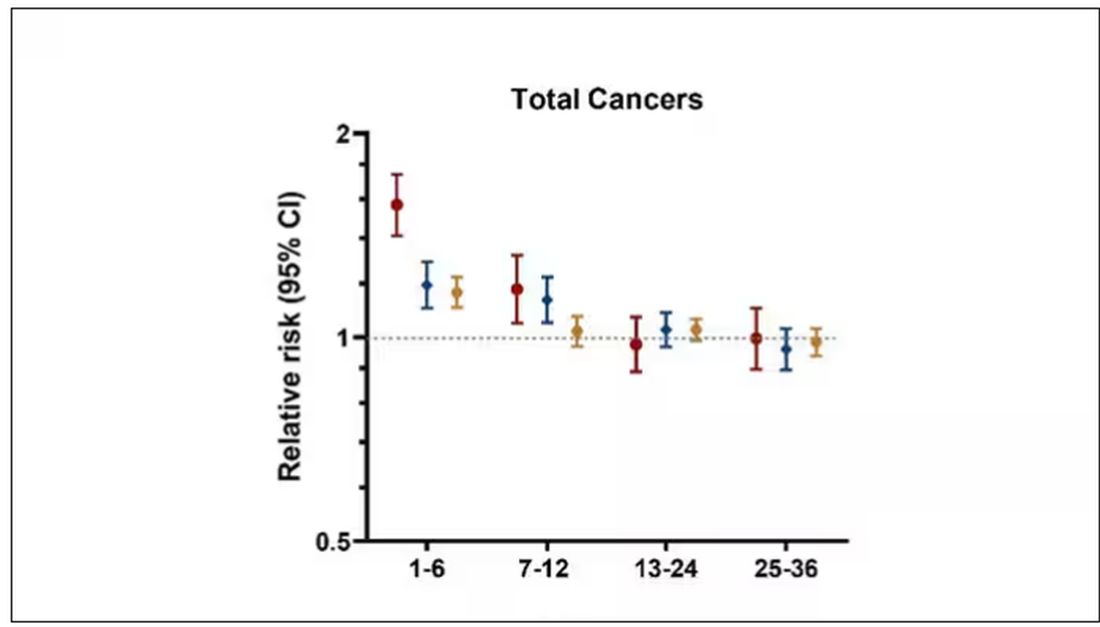
The further you get from the year of that 10% weight loss, the less likely you are to be diagnosed with cancer. Most of these cancers are diagnosed within a year of losing weight. In other words, if you’re reading this and getting worried that you lost weight 10 years ago, you’re probably out of the woods. That was, most likely, just you getting healthier.
Last thing: We have methods for weight loss now that are way more effective than diet or exercise. I’m looking at you, Ozempic. But aside from the weight loss wonder drugs, we have surgery and other interventions. This study did not capture any of that data. Ozempic wasn’t even on the market during this study, so we can’t say anything about the relationship between weight loss and cancer among people using nonlifestyle mechanisms to lose weight.
It’s a complicated system. But the clinically actionable point here is to notice if patients have lost weight. If they’ve lost it without trying, further workup is reasonable. If they’ve lost it but were trying to lose it, tell them “good job.” And consider a workup anyway.
Dr. Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
As anyone who has been through medical training will tell you, some little scenes just stick with you. I had been seeing a patient in our resident clinic in West Philly for a couple of years. She was in her mid-60s with diabetes and hypertension and a distant smoking history. She was overweight and had been trying to improve her diet and lose weight since I started seeing her. One day she came in and was delighted to report that she had finally started shedding some pounds — about 15 in the past 2 months.
I enthusiastically told my preceptor that my careful dietary counseling had finally done the job. She looked through the chart for a moment and asked, “Is she up to date on her cancer screening?” A workup revealed adenocarcinoma of the lung. The patient did well, actually, but the story stuck with me.
The textbooks call it “unintentional weight loss,” often in big, scary letters, and every doctor will go just a bit pale if a patient tells them that, despite efforts not to, they are losing weight. But true unintentional weight loss is not that common. After all, most of us are at least half-heartedly trying to lose weight all the time. Should doctors be worried when we are successful?
A new study suggests that perhaps they should. We’re talking about this study, appearing in JAMA, which combined participants from two long-running observational cohorts: 120,000 women from the Nurses’ Health Study, and 50,000 men from the Health Professionals Follow-Up Study. (These cohorts started in the 1970s and 1980s, so we’ll give them a pass on the gender-specific study designs.)
The rationale of enrolling healthcare providers in these cohort studies is that they would be reliable witnesses of their own health status. If a nurse or doctor says they have pancreatic cancer, it’s likely that they truly have pancreatic cancer. Detailed health surveys were distributed to the participants every other year, and the average follow-up was more than a decade.
Participants recorded their weight — as an aside, a nested study found that self-reported rate was extremely well correlated with professionally measured weight — and whether they had received a cancer diagnosis since the last survey.
This allowed researchers to look at the phenomenon described above. Would weight loss precede a new diagnosis of cancer? And, more interestingly, would intentional weight loss precede a new diagnosis of cancer.
I don’t think it will surprise you to hear that individuals in the highest category of weight loss, those who lost more than 10% of their body weight over a 2-year period, had a larger risk of being diagnosed with cancer in the next year. That’s the yellow line in this graph. In fact, they had about a 40% higher risk than those who did not lose weight.
Increased risk was found across multiple cancer types, though cancers of the gastrointestinal tract, not surprisingly, were most strongly associated with antecedent weight loss.
What about intentionality of weight loss? Unfortunately, the surveys did not ask participants whether they were trying to lose weight. Rather, the surveys asked about exercise and dietary habits. The researchers leveraged these responses to create three categories of participants: those who seemed to be trying to lose weight (defined as people who had increased their exercise and dietary quality); those who didn’t seem to be trying to lose weight (they changed neither exercise nor dietary behaviors); and a middle group, which changed one or the other of these behaviors but not both.
Let’s look at those who really seemed to be trying to lose weight. Over 2 years, they got more exercise and improved their diet.
If they succeeded in losing 10% or more of their body weight, they still had a higher risk for cancer than those who had not lost weight — about 30% higher, which is not that different from the 40% increased risk when you include those folks who weren’t changing their lifestyle.
This is why this study is important. The classic teaching is that unintentional weight loss is a bad thing and needs a workup. That’s fine. But we live in a world where perhaps the majority of people are, at any given time, trying to lose weight.
We need to be careful here. I am not by any means trying to say that people who have successfully lost weight have cancer. Both of the following statements can be true:
Significant weight loss, whether intentional or not, is associated with a higher risk for cancer.
Most people with significant weight loss will not have cancer.
Both of these can be true because cancer is, fortunately, rare. Of people who lose weight, the vast majority will lose weight because they are engaging in healthier behaviors. A small number may lose weight because something else is wrong. It’s just hard to tell the two apart.
Out of the nearly 200,000 people in this study, only around 16,000 developed cancer during follow-up. Again, although the chance of having cancer is slightly higher if someone has experienced weight loss, the chance is still very low.
We also need to avoid suggesting that weight loss causes cancer. Some people lose weight because of an existing, as of yet undiagnosed cancer and its metabolic effects. This is borne out if you look at the risk of being diagnosed with cancer as you move further away from the interval of weight loss.
The further you get from the year of that 10% weight loss, the less likely you are to be diagnosed with cancer. Most of these cancers are diagnosed within a year of losing weight. In other words, if you’re reading this and getting worried that you lost weight 10 years ago, you’re probably out of the woods. That was, most likely, just you getting healthier.
Last thing: We have methods for weight loss now that are way more effective than diet or exercise. I’m looking at you, Ozempic. But aside from the weight loss wonder drugs, we have surgery and other interventions. This study did not capture any of that data. Ozempic wasn’t even on the market during this study, so we can’t say anything about the relationship between weight loss and cancer among people using nonlifestyle mechanisms to lose weight.
It’s a complicated system. But the clinically actionable point here is to notice if patients have lost weight. If they’ve lost it without trying, further workup is reasonable. If they’ve lost it but were trying to lose it, tell them “good job.” And consider a workup anyway.
Dr. Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
As anyone who has been through medical training will tell you, some little scenes just stick with you. I had been seeing a patient in our resident clinic in West Philly for a couple of years. She was in her mid-60s with diabetes and hypertension and a distant smoking history. She was overweight and had been trying to improve her diet and lose weight since I started seeing her. One day she came in and was delighted to report that she had finally started shedding some pounds — about 15 in the past 2 months.
I enthusiastically told my preceptor that my careful dietary counseling had finally done the job. She looked through the chart for a moment and asked, “Is she up to date on her cancer screening?” A workup revealed adenocarcinoma of the lung. The patient did well, actually, but the story stuck with me.
The textbooks call it “unintentional weight loss,” often in big, scary letters, and every doctor will go just a bit pale if a patient tells them that, despite efforts not to, they are losing weight. But true unintentional weight loss is not that common. After all, most of us are at least half-heartedly trying to lose weight all the time. Should doctors be worried when we are successful?
A new study suggests that perhaps they should. We’re talking about this study, appearing in JAMA, which combined participants from two long-running observational cohorts: 120,000 women from the Nurses’ Health Study, and 50,000 men from the Health Professionals Follow-Up Study. (These cohorts started in the 1970s and 1980s, so we’ll give them a pass on the gender-specific study designs.)
The rationale of enrolling healthcare providers in these cohort studies is that they would be reliable witnesses of their own health status. If a nurse or doctor says they have pancreatic cancer, it’s likely that they truly have pancreatic cancer. Detailed health surveys were distributed to the participants every other year, and the average follow-up was more than a decade.
Participants recorded their weight — as an aside, a nested study found that self-reported rate was extremely well correlated with professionally measured weight — and whether they had received a cancer diagnosis since the last survey.
This allowed researchers to look at the phenomenon described above. Would weight loss precede a new diagnosis of cancer? And, more interestingly, would intentional weight loss precede a new diagnosis of cancer.
I don’t think it will surprise you to hear that individuals in the highest category of weight loss, those who lost more than 10% of their body weight over a 2-year period, had a larger risk of being diagnosed with cancer in the next year. That’s the yellow line in this graph. In fact, they had about a 40% higher risk than those who did not lose weight.
Increased risk was found across multiple cancer types, though cancers of the gastrointestinal tract, not surprisingly, were most strongly associated with antecedent weight loss.
What about intentionality of weight loss? Unfortunately, the surveys did not ask participants whether they were trying to lose weight. Rather, the surveys asked about exercise and dietary habits. The researchers leveraged these responses to create three categories of participants: those who seemed to be trying to lose weight (defined as people who had increased their exercise and dietary quality); those who didn’t seem to be trying to lose weight (they changed neither exercise nor dietary behaviors); and a middle group, which changed one or the other of these behaviors but not both.
Let’s look at those who really seemed to be trying to lose weight. Over 2 years, they got more exercise and improved their diet.
If they succeeded in losing 10% or more of their body weight, they still had a higher risk for cancer than those who had not lost weight — about 30% higher, which is not that different from the 40% increased risk when you include those folks who weren’t changing their lifestyle.
This is why this study is important. The classic teaching is that unintentional weight loss is a bad thing and needs a workup. That’s fine. But we live in a world where perhaps the majority of people are, at any given time, trying to lose weight.
We need to be careful here. I am not by any means trying to say that people who have successfully lost weight have cancer. Both of the following statements can be true:
Significant weight loss, whether intentional or not, is associated with a higher risk for cancer.
Most people with significant weight loss will not have cancer.
Both of these can be true because cancer is, fortunately, rare. Of people who lose weight, the vast majority will lose weight because they are engaging in healthier behaviors. A small number may lose weight because something else is wrong. It’s just hard to tell the two apart.
Out of the nearly 200,000 people in this study, only around 16,000 developed cancer during follow-up. Again, although the chance of having cancer is slightly higher if someone has experienced weight loss, the chance is still very low.
We also need to avoid suggesting that weight loss causes cancer. Some people lose weight because of an existing, as of yet undiagnosed cancer and its metabolic effects. This is borne out if you look at the risk of being diagnosed with cancer as you move further away from the interval of weight loss.
The further you get from the year of that 10% weight loss, the less likely you are to be diagnosed with cancer. Most of these cancers are diagnosed within a year of losing weight. In other words, if you’re reading this and getting worried that you lost weight 10 years ago, you’re probably out of the woods. That was, most likely, just you getting healthier.
Last thing: We have methods for weight loss now that are way more effective than diet or exercise. I’m looking at you, Ozempic. But aside from the weight loss wonder drugs, we have surgery and other interventions. This study did not capture any of that data. Ozempic wasn’t even on the market during this study, so we can’t say anything about the relationship between weight loss and cancer among people using nonlifestyle mechanisms to lose weight.
It’s a complicated system. But the clinically actionable point here is to notice if patients have lost weight. If they’ve lost it without trying, further workup is reasonable. If they’ve lost it but were trying to lose it, tell them “good job.” And consider a workup anyway.
Dr. Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
Tackling Sexism in Medicine
This interview was recorded Dec. 8, 2023. The transcript has been edited for clarity.
Kathrin LaFaver, MD: I have the pleasure of talking with Dr. Elizabeth Loder today. Dr. Loder is the vice chair of academic affairs in the department of neurology and a staff physician at Graham Headache Center at Brigham and Women’s Hospital in Boston. She’s also a professor of neurology at Harvard Medical School and has been a mentor to many headache specialists in the field.
We’ll be talking about the topic of sexism in medicine.
Elizabeth W. Loder, MD, MPH: Thank you so much, Dr. LaFaver. I’m very pleased to be here to talk about something that I think is very important.
Dr. LaFaver: Dr. Loder, you were a speaker at our Women in Neurology third annual conference in Florida, giving a talk on this very topic, sexism in medicine. It was very helpful, especially for many younger women entering the field of neurology, to hear that some of the experiences they’re having are not unique to them, but unfortunately remain a more systemic issue.
To get us started, could you define sexism in medicine for us, and how you got interested in this topic?
Sexism Can Be Subtle
Dr. Loder: You’re absolutely right. It’s nice for women, particularly those entering the field, to know that they’re not alone.
What is sexism? Well, it’s generally considered to be prejudice or discrimination against someone based on their sex or gender. One thing that’s important to remember is it’s not always extremely obvious. It’s not always intentional. I think most of us, I’d include myself in that, have sexist views. We may not always be aware of them. This is part of the unconscious bias that many of us have been trained to think.
As one example, we’re socialized — or I certainly was — to think of doctors as male. When a patient says, “I saw a cardiologist,” in my mind, I think that that person might have been male. Sometimes, I will ask, “What did he say?” That’s an example of sexism.
Sexism can be internalized. It can come from any source. We’re all implicated in it. I think that’s very important to remember. This is not a case of them doing something to us. This is something that is much more widespread and engendered in the society that we live in.
Dr. LaFaver: I think that’s really important to realize. Could you mention some other examples, specifically in the field of neurology, that you have encountered?
Dr. Loder: Certainly, looking over my own career — and I’m at a point now where I can look at early, mid, and late career — things were, of course, very different when I began. I would say that sexism was much more overt.
Very early in my career, I experienced a large amount of hostility from a boss. I ended up having to leave. This was, I believe, based on my sex. It turned out that leaving was a good thing for me, but it was a very unpleasant experience.
I also became a leader in my professional society. During my path to leadership and seeing other women attempt to make progress within the organization, I certainly saw behavior that I would characterize as rooted in sexism. To its credit, though, the professional society to which I belong, I think, has become one of the most progressive societies out there. The women within have been able to affect quite a bit of change in that professional organization.
I dealt with a case of sexual harassment in my own division when I was a division chief, which gave me insight into institutional policies and procedures meant to deal with situations like that, which, I think, leave something to be desired. That’s changing.
Now, I work in academic affairs, and I see how likely or unlikely people, based on their sex, are to put themselves forward for promotion, how likely they are to believe in their own capabilities. I also work as a medical journal editor, and I see generally that women are more hesitant to proclaim themselves experts and to accept invitations to provide or write editorial commentary. Sometimes, they do not perceive themselves as being expert enough to do so. I have seen many of these things over my career.
Dr. LaFaver: Thank you for sharing. I think it’s important to realize that it›s not just unidirectional but sometimes affects women and kind of goes both ways.
As you mentioned, these issues are not always overt. In recent years, microaggressions has become more of a term that has been used. Could you talk a bit about that? What are the microaggressions and how do they affect women?
Microaggressions and Migraine Mavens
Dr. Loder: The term microaggression has become very popular, and I think many people find it somewhat irritating. That, to me, is inherent in what a microaggression is. Microaggressions, by definition, are small things. It’s hard to prove that they were rooted in sexism. Sometimes, there’s a large amount of ambiguity about it. It can be as simple as inviting a pregnant woman to sit down because you perceive that she needs to sit, commenting on somebody’s shoes, or things like that.
Often, they’re unintentional. Sometimes, they come from a place of what we might term benevolent sexism, people trying to be helpful to a woman because they perceive that she’s weaker or she might not be able to do something, or maybe she has family responsibilities. They think that they’re being helpful. These things happen when we perceive people to be different in some way, as women are perceived to be different in terms of their responsibilities in the home or different in terms of what we expect from them in the workplace.
The problem with microaggressions is because they’re small, each one of them, I think the temptation is often to say, “Oh you know, they didn’t mean it. It’s not that big a deal. It was just a comment on your shoes.” If a woman brings that up, she’s often made to feel, sometimes by other women, that she should just chill out. This isn’t a big deal.
The problem is that they may not be a big deal in and of themselves, but when they are repeated over a long period of time, they can really sap somebody’s confidence in herself, make her question her own competence, and can have a cumulative effect that is very negative.
Although I think many people are skeptical about microaggressions as an important contribution to how women do in the workplace and in other settings, they are, in my opinion, important. I’ll just emphasize again that they come from everywhere, including other women and colleagues who mean to be helpful.
Dr. LaFaver: I know you have led a team of headache specialists and wrote a fantastic article about navigating sexism at work and what not to say to your female colleagues. Could you share some tips for them to navigate sexism at work and, specifically, as it applies to our careers in medicine.
Dr. Loder: Thank you for calling out that article. It actually grew organically out of a Facebook group that I started called Migraine Mavens. I’ve worked in the headache field throughout my career, and I experienced something that, to me, was kind of discouraging that I felt was sexist within our field. I just thought, What can I do? I thought, Well, maybe there’s some strength to be had in sharing this with other women, and maybe we should have our own social media community, so I formed this group.
After I gave a talk at the American Headache Society about sexism in the headache field, one of the members sent me a message saying, “Your talk was very timely. Immediately after you gave this talk, somebody stopped me in the hall and said, ‘Congratulations on your leadership position in the society, but are you really sure you can do this because you’ve got young children at home?’ Your talk was very needed.”
People started just discussing within the group, “Oh, this happened to me,” “That happened to me.” We began to brainstorm what we could have done differently. When these things happen, people are not necessarily prepared for them, and later on, you lie awake thinking of what you could have said.
We decided to write a paper. It ended up being published in The Journal of Head and Face Pain, and it was based on real vignettes from people in the group. We anonymized them so that people would not recognize themselves necessarily, but they’re all real. They’re things that really happened to women in the group.
We first describe the vignette — what happened. Then we explain, for those who need the explanation, what’s wrong with it. Why is it wrong to tell somebody to smile, for example. What could you do differently? What could somebody who sees this happen do, the so-called bystander or upstander? If you witness something like that, can you help the person toward whom this problematic behavior is directed? We came up with some examples. The all-purpose thing is to say, “What did you just say?” Make them repeat it, which often helps people to realize how inappropriate it is.
This got published as an article. It became quite the subject of attention on X [formerly Twitter] and elsewhere. I guess the term going viral would apply to this. It included a large amount of real-world advice. The thing I really loved about it was that it was written by the women in the group who had experienced these things. I would characterize it as having been somewhat therapeutic. We got many messages from women in other disciplines, outside medicine, saying that this happens in oceanography, for example. I think what we described really resonated beyond the field of headache medicine and neurology.
Institutional Sexism
Dr. LaFaver: Looking beyond the individual person, do you have any recommendations for medical institutions to share in order to do better and maybe create an environment that is less sexist?
Dr. Loder: Yes. I think many institutions try to deal with the problem of lack of diversity, whether it›s women or other underrepresented groups, by hiring. That’s one way to go about it, but I think retention strategies are also very important, and they need to pay attention to the work environment.
Every institution now has guidelines about harassment, bullying, sexism, racism, and so on. In general though — and I’m not speaking about any particular institution — these policies and procedures are often crafted with an eye toward protecting the institution. I would advise institutions to be a bit more genuine about this, and not to think so quickly about what can we do to prevent the institution from facing a charge of sexism, or what can we do to deny or dismiss these allegations, toward thinking, what can we do to really and truly be helpful to these women? In other words, approach it from the point of view of trying to help the people involved instead of trying to help the institution. That will make a very big difference.
I also think that citizenship activities, serving on committees, doing thankless tasks behind the scenes, and clinical work, seeing patients ... Women are overrepresented among clinicians. Of course, we know that research and bringing in big grants is often prioritized over clinical care, despite what institutions may say about that in public. I think those activities should be valued more highly, both in terms of pay and in terms of academic recognition.
In regard to the issue of salary, I would encourage institutions to publish salaries or at least make them easily findable by people within the institution. I think there should be objective criteria for salary determinations. The most important thing is that I do not think that women should be expected to negotiate their salaries. Women are judged differently than men when they attempt to negotiate salaries. It often backfires for them. It also is something that many women do not feel comfortable doing.
Waiting until somebody tries to negotiate a higher salary really guarantees, embeds, and operationalizes sex differences in salaries. We need to move away from the idea that you have to be a go-getter and that you have to ask for this raise. There should be objective criteria, salaries should be revisited on a regular basis, and the kinds of activities that women do that are undervalued should be more recognized in terms of money and advancement within academia.
Dr. LaFaver: I couldn’t agree more. One of the other topics discussed at the Women in Neurology conference was mid-career development, retention, and career paths. I think it’s known that many women leave academic medicine mid-career. I think these are excellent suggestions and hopefully will help to make careers successful for men and women, without needing to worry about being treated differently or unfairly.
Thank you so much. This was a wonderful overview of this topic.
Dr. Loder: You’re welcome, Dr. LaFaver. Thank you so much for asking me to speak on this topic. I really appreciate it.
Dr. LaFaver: Thanks, everyone.
Dr. LaFaver is a neurologist at Saratoga Hospital Medical Group, Saratoga Springs, New York. Dr. LaFaver and Dr. Loder disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
This interview was recorded Dec. 8, 2023. The transcript has been edited for clarity.
Kathrin LaFaver, MD: I have the pleasure of talking with Dr. Elizabeth Loder today. Dr. Loder is the vice chair of academic affairs in the department of neurology and a staff physician at Graham Headache Center at Brigham and Women’s Hospital in Boston. She’s also a professor of neurology at Harvard Medical School and has been a mentor to many headache specialists in the field.
We’ll be talking about the topic of sexism in medicine.
Elizabeth W. Loder, MD, MPH: Thank you so much, Dr. LaFaver. I’m very pleased to be here to talk about something that I think is very important.
Dr. LaFaver: Dr. Loder, you were a speaker at our Women in Neurology third annual conference in Florida, giving a talk on this very topic, sexism in medicine. It was very helpful, especially for many younger women entering the field of neurology, to hear that some of the experiences they’re having are not unique to them, but unfortunately remain a more systemic issue.
To get us started, could you define sexism in medicine for us, and how you got interested in this topic?
Sexism Can Be Subtle
Dr. Loder: You’re absolutely right. It’s nice for women, particularly those entering the field, to know that they’re not alone.
What is sexism? Well, it’s generally considered to be prejudice or discrimination against someone based on their sex or gender. One thing that’s important to remember is it’s not always extremely obvious. It’s not always intentional. I think most of us, I’d include myself in that, have sexist views. We may not always be aware of them. This is part of the unconscious bias that many of us have been trained to think.
As one example, we’re socialized — or I certainly was — to think of doctors as male. When a patient says, “I saw a cardiologist,” in my mind, I think that that person might have been male. Sometimes, I will ask, “What did he say?” That’s an example of sexism.
Sexism can be internalized. It can come from any source. We’re all implicated in it. I think that’s very important to remember. This is not a case of them doing something to us. This is something that is much more widespread and engendered in the society that we live in.
Dr. LaFaver: I think that’s really important to realize. Could you mention some other examples, specifically in the field of neurology, that you have encountered?
Dr. Loder: Certainly, looking over my own career — and I’m at a point now where I can look at early, mid, and late career — things were, of course, very different when I began. I would say that sexism was much more overt.
Very early in my career, I experienced a large amount of hostility from a boss. I ended up having to leave. This was, I believe, based on my sex. It turned out that leaving was a good thing for me, but it was a very unpleasant experience.
I also became a leader in my professional society. During my path to leadership and seeing other women attempt to make progress within the organization, I certainly saw behavior that I would characterize as rooted in sexism. To its credit, though, the professional society to which I belong, I think, has become one of the most progressive societies out there. The women within have been able to affect quite a bit of change in that professional organization.
I dealt with a case of sexual harassment in my own division when I was a division chief, which gave me insight into institutional policies and procedures meant to deal with situations like that, which, I think, leave something to be desired. That’s changing.
Now, I work in academic affairs, and I see how likely or unlikely people, based on their sex, are to put themselves forward for promotion, how likely they are to believe in their own capabilities. I also work as a medical journal editor, and I see generally that women are more hesitant to proclaim themselves experts and to accept invitations to provide or write editorial commentary. Sometimes, they do not perceive themselves as being expert enough to do so. I have seen many of these things over my career.
Dr. LaFaver: Thank you for sharing. I think it’s important to realize that it›s not just unidirectional but sometimes affects women and kind of goes both ways.
As you mentioned, these issues are not always overt. In recent years, microaggressions has become more of a term that has been used. Could you talk a bit about that? What are the microaggressions and how do they affect women?
Microaggressions and Migraine Mavens
Dr. Loder: The term microaggression has become very popular, and I think many people find it somewhat irritating. That, to me, is inherent in what a microaggression is. Microaggressions, by definition, are small things. It’s hard to prove that they were rooted in sexism. Sometimes, there’s a large amount of ambiguity about it. It can be as simple as inviting a pregnant woman to sit down because you perceive that she needs to sit, commenting on somebody’s shoes, or things like that.
Often, they’re unintentional. Sometimes, they come from a place of what we might term benevolent sexism, people trying to be helpful to a woman because they perceive that she’s weaker or she might not be able to do something, or maybe she has family responsibilities. They think that they’re being helpful. These things happen when we perceive people to be different in some way, as women are perceived to be different in terms of their responsibilities in the home or different in terms of what we expect from them in the workplace.
The problem with microaggressions is because they’re small, each one of them, I think the temptation is often to say, “Oh you know, they didn’t mean it. It’s not that big a deal. It was just a comment on your shoes.” If a woman brings that up, she’s often made to feel, sometimes by other women, that she should just chill out. This isn’t a big deal.
The problem is that they may not be a big deal in and of themselves, but when they are repeated over a long period of time, they can really sap somebody’s confidence in herself, make her question her own competence, and can have a cumulative effect that is very negative.
Although I think many people are skeptical about microaggressions as an important contribution to how women do in the workplace and in other settings, they are, in my opinion, important. I’ll just emphasize again that they come from everywhere, including other women and colleagues who mean to be helpful.
Dr. LaFaver: I know you have led a team of headache specialists and wrote a fantastic article about navigating sexism at work and what not to say to your female colleagues. Could you share some tips for them to navigate sexism at work and, specifically, as it applies to our careers in medicine.
Dr. Loder: Thank you for calling out that article. It actually grew organically out of a Facebook group that I started called Migraine Mavens. I’ve worked in the headache field throughout my career, and I experienced something that, to me, was kind of discouraging that I felt was sexist within our field. I just thought, What can I do? I thought, Well, maybe there’s some strength to be had in sharing this with other women, and maybe we should have our own social media community, so I formed this group.
After I gave a talk at the American Headache Society about sexism in the headache field, one of the members sent me a message saying, “Your talk was very timely. Immediately after you gave this talk, somebody stopped me in the hall and said, ‘Congratulations on your leadership position in the society, but are you really sure you can do this because you’ve got young children at home?’ Your talk was very needed.”
People started just discussing within the group, “Oh, this happened to me,” “That happened to me.” We began to brainstorm what we could have done differently. When these things happen, people are not necessarily prepared for them, and later on, you lie awake thinking of what you could have said.
We decided to write a paper. It ended up being published in The Journal of Head and Face Pain, and it was based on real vignettes from people in the group. We anonymized them so that people would not recognize themselves necessarily, but they’re all real. They’re things that really happened to women in the group.
We first describe the vignette — what happened. Then we explain, for those who need the explanation, what’s wrong with it. Why is it wrong to tell somebody to smile, for example. What could you do differently? What could somebody who sees this happen do, the so-called bystander or upstander? If you witness something like that, can you help the person toward whom this problematic behavior is directed? We came up with some examples. The all-purpose thing is to say, “What did you just say?” Make them repeat it, which often helps people to realize how inappropriate it is.
This got published as an article. It became quite the subject of attention on X [formerly Twitter] and elsewhere. I guess the term going viral would apply to this. It included a large amount of real-world advice. The thing I really loved about it was that it was written by the women in the group who had experienced these things. I would characterize it as having been somewhat therapeutic. We got many messages from women in other disciplines, outside medicine, saying that this happens in oceanography, for example. I think what we described really resonated beyond the field of headache medicine and neurology.
Institutional Sexism
Dr. LaFaver: Looking beyond the individual person, do you have any recommendations for medical institutions to share in order to do better and maybe create an environment that is less sexist?
Dr. Loder: Yes. I think many institutions try to deal with the problem of lack of diversity, whether it›s women or other underrepresented groups, by hiring. That’s one way to go about it, but I think retention strategies are also very important, and they need to pay attention to the work environment.
Every institution now has guidelines about harassment, bullying, sexism, racism, and so on. In general though — and I’m not speaking about any particular institution — these policies and procedures are often crafted with an eye toward protecting the institution. I would advise institutions to be a bit more genuine about this, and not to think so quickly about what can we do to prevent the institution from facing a charge of sexism, or what can we do to deny or dismiss these allegations, toward thinking, what can we do to really and truly be helpful to these women? In other words, approach it from the point of view of trying to help the people involved instead of trying to help the institution. That will make a very big difference.
I also think that citizenship activities, serving on committees, doing thankless tasks behind the scenes, and clinical work, seeing patients ... Women are overrepresented among clinicians. Of course, we know that research and bringing in big grants is often prioritized over clinical care, despite what institutions may say about that in public. I think those activities should be valued more highly, both in terms of pay and in terms of academic recognition.
In regard to the issue of salary, I would encourage institutions to publish salaries or at least make them easily findable by people within the institution. I think there should be objective criteria for salary determinations. The most important thing is that I do not think that women should be expected to negotiate their salaries. Women are judged differently than men when they attempt to negotiate salaries. It often backfires for them. It also is something that many women do not feel comfortable doing.
Waiting until somebody tries to negotiate a higher salary really guarantees, embeds, and operationalizes sex differences in salaries. We need to move away from the idea that you have to be a go-getter and that you have to ask for this raise. There should be objective criteria, salaries should be revisited on a regular basis, and the kinds of activities that women do that are undervalued should be more recognized in terms of money and advancement within academia.
Dr. LaFaver: I couldn’t agree more. One of the other topics discussed at the Women in Neurology conference was mid-career development, retention, and career paths. I think it’s known that many women leave academic medicine mid-career. I think these are excellent suggestions and hopefully will help to make careers successful for men and women, without needing to worry about being treated differently or unfairly.
Thank you so much. This was a wonderful overview of this topic.
Dr. Loder: You’re welcome, Dr. LaFaver. Thank you so much for asking me to speak on this topic. I really appreciate it.
Dr. LaFaver: Thanks, everyone.
Dr. LaFaver is a neurologist at Saratoga Hospital Medical Group, Saratoga Springs, New York. Dr. LaFaver and Dr. Loder disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
This interview was recorded Dec. 8, 2023. The transcript has been edited for clarity.
Kathrin LaFaver, MD: I have the pleasure of talking with Dr. Elizabeth Loder today. Dr. Loder is the vice chair of academic affairs in the department of neurology and a staff physician at Graham Headache Center at Brigham and Women’s Hospital in Boston. She’s also a professor of neurology at Harvard Medical School and has been a mentor to many headache specialists in the field.
We’ll be talking about the topic of sexism in medicine.
Elizabeth W. Loder, MD, MPH: Thank you so much, Dr. LaFaver. I’m very pleased to be here to talk about something that I think is very important.
Dr. LaFaver: Dr. Loder, you were a speaker at our Women in Neurology third annual conference in Florida, giving a talk on this very topic, sexism in medicine. It was very helpful, especially for many younger women entering the field of neurology, to hear that some of the experiences they’re having are not unique to them, but unfortunately remain a more systemic issue.
To get us started, could you define sexism in medicine for us, and how you got interested in this topic?
Sexism Can Be Subtle
Dr. Loder: You’re absolutely right. It’s nice for women, particularly those entering the field, to know that they’re not alone.
What is sexism? Well, it’s generally considered to be prejudice or discrimination against someone based on their sex or gender. One thing that’s important to remember is it’s not always extremely obvious. It’s not always intentional. I think most of us, I’d include myself in that, have sexist views. We may not always be aware of them. This is part of the unconscious bias that many of us have been trained to think.
As one example, we’re socialized — or I certainly was — to think of doctors as male. When a patient says, “I saw a cardiologist,” in my mind, I think that that person might have been male. Sometimes, I will ask, “What did he say?” That’s an example of sexism.
Sexism can be internalized. It can come from any source. We’re all implicated in it. I think that’s very important to remember. This is not a case of them doing something to us. This is something that is much more widespread and engendered in the society that we live in.
Dr. LaFaver: I think that’s really important to realize. Could you mention some other examples, specifically in the field of neurology, that you have encountered?
Dr. Loder: Certainly, looking over my own career — and I’m at a point now where I can look at early, mid, and late career — things were, of course, very different when I began. I would say that sexism was much more overt.
Very early in my career, I experienced a large amount of hostility from a boss. I ended up having to leave. This was, I believe, based on my sex. It turned out that leaving was a good thing for me, but it was a very unpleasant experience.
I also became a leader in my professional society. During my path to leadership and seeing other women attempt to make progress within the organization, I certainly saw behavior that I would characterize as rooted in sexism. To its credit, though, the professional society to which I belong, I think, has become one of the most progressive societies out there. The women within have been able to affect quite a bit of change in that professional organization.
I dealt with a case of sexual harassment in my own division when I was a division chief, which gave me insight into institutional policies and procedures meant to deal with situations like that, which, I think, leave something to be desired. That’s changing.
Now, I work in academic affairs, and I see how likely or unlikely people, based on their sex, are to put themselves forward for promotion, how likely they are to believe in their own capabilities. I also work as a medical journal editor, and I see generally that women are more hesitant to proclaim themselves experts and to accept invitations to provide or write editorial commentary. Sometimes, they do not perceive themselves as being expert enough to do so. I have seen many of these things over my career.
Dr. LaFaver: Thank you for sharing. I think it’s important to realize that it›s not just unidirectional but sometimes affects women and kind of goes both ways.
As you mentioned, these issues are not always overt. In recent years, microaggressions has become more of a term that has been used. Could you talk a bit about that? What are the microaggressions and how do they affect women?
Microaggressions and Migraine Mavens
Dr. Loder: The term microaggression has become very popular, and I think many people find it somewhat irritating. That, to me, is inherent in what a microaggression is. Microaggressions, by definition, are small things. It’s hard to prove that they were rooted in sexism. Sometimes, there’s a large amount of ambiguity about it. It can be as simple as inviting a pregnant woman to sit down because you perceive that she needs to sit, commenting on somebody’s shoes, or things like that.
Often, they’re unintentional. Sometimes, they come from a place of what we might term benevolent sexism, people trying to be helpful to a woman because they perceive that she’s weaker or she might not be able to do something, or maybe she has family responsibilities. They think that they’re being helpful. These things happen when we perceive people to be different in some way, as women are perceived to be different in terms of their responsibilities in the home or different in terms of what we expect from them in the workplace.
The problem with microaggressions is because they’re small, each one of them, I think the temptation is often to say, “Oh you know, they didn’t mean it. It’s not that big a deal. It was just a comment on your shoes.” If a woman brings that up, she’s often made to feel, sometimes by other women, that she should just chill out. This isn’t a big deal.
The problem is that they may not be a big deal in and of themselves, but when they are repeated over a long period of time, they can really sap somebody’s confidence in herself, make her question her own competence, and can have a cumulative effect that is very negative.
Although I think many people are skeptical about microaggressions as an important contribution to how women do in the workplace and in other settings, they are, in my opinion, important. I’ll just emphasize again that they come from everywhere, including other women and colleagues who mean to be helpful.
Dr. LaFaver: I know you have led a team of headache specialists and wrote a fantastic article about navigating sexism at work and what not to say to your female colleagues. Could you share some tips for them to navigate sexism at work and, specifically, as it applies to our careers in medicine.
Dr. Loder: Thank you for calling out that article. It actually grew organically out of a Facebook group that I started called Migraine Mavens. I’ve worked in the headache field throughout my career, and I experienced something that, to me, was kind of discouraging that I felt was sexist within our field. I just thought, What can I do? I thought, Well, maybe there’s some strength to be had in sharing this with other women, and maybe we should have our own social media community, so I formed this group.
After I gave a talk at the American Headache Society about sexism in the headache field, one of the members sent me a message saying, “Your talk was very timely. Immediately after you gave this talk, somebody stopped me in the hall and said, ‘Congratulations on your leadership position in the society, but are you really sure you can do this because you’ve got young children at home?’ Your talk was very needed.”
People started just discussing within the group, “Oh, this happened to me,” “That happened to me.” We began to brainstorm what we could have done differently. When these things happen, people are not necessarily prepared for them, and later on, you lie awake thinking of what you could have said.
We decided to write a paper. It ended up being published in The Journal of Head and Face Pain, and it was based on real vignettes from people in the group. We anonymized them so that people would not recognize themselves necessarily, but they’re all real. They’re things that really happened to women in the group.
We first describe the vignette — what happened. Then we explain, for those who need the explanation, what’s wrong with it. Why is it wrong to tell somebody to smile, for example. What could you do differently? What could somebody who sees this happen do, the so-called bystander or upstander? If you witness something like that, can you help the person toward whom this problematic behavior is directed? We came up with some examples. The all-purpose thing is to say, “What did you just say?” Make them repeat it, which often helps people to realize how inappropriate it is.
This got published as an article. It became quite the subject of attention on X [formerly Twitter] and elsewhere. I guess the term going viral would apply to this. It included a large amount of real-world advice. The thing I really loved about it was that it was written by the women in the group who had experienced these things. I would characterize it as having been somewhat therapeutic. We got many messages from women in other disciplines, outside medicine, saying that this happens in oceanography, for example. I think what we described really resonated beyond the field of headache medicine and neurology.
Institutional Sexism
Dr. LaFaver: Looking beyond the individual person, do you have any recommendations for medical institutions to share in order to do better and maybe create an environment that is less sexist?
Dr. Loder: Yes. I think many institutions try to deal with the problem of lack of diversity, whether it›s women or other underrepresented groups, by hiring. That’s one way to go about it, but I think retention strategies are also very important, and they need to pay attention to the work environment.
Every institution now has guidelines about harassment, bullying, sexism, racism, and so on. In general though — and I’m not speaking about any particular institution — these policies and procedures are often crafted with an eye toward protecting the institution. I would advise institutions to be a bit more genuine about this, and not to think so quickly about what can we do to prevent the institution from facing a charge of sexism, or what can we do to deny or dismiss these allegations, toward thinking, what can we do to really and truly be helpful to these women? In other words, approach it from the point of view of trying to help the people involved instead of trying to help the institution. That will make a very big difference.
I also think that citizenship activities, serving on committees, doing thankless tasks behind the scenes, and clinical work, seeing patients ... Women are overrepresented among clinicians. Of course, we know that research and bringing in big grants is often prioritized over clinical care, despite what institutions may say about that in public. I think those activities should be valued more highly, both in terms of pay and in terms of academic recognition.
In regard to the issue of salary, I would encourage institutions to publish salaries or at least make them easily findable by people within the institution. I think there should be objective criteria for salary determinations. The most important thing is that I do not think that women should be expected to negotiate their salaries. Women are judged differently than men when they attempt to negotiate salaries. It often backfires for them. It also is something that many women do not feel comfortable doing.
Waiting until somebody tries to negotiate a higher salary really guarantees, embeds, and operationalizes sex differences in salaries. We need to move away from the idea that you have to be a go-getter and that you have to ask for this raise. There should be objective criteria, salaries should be revisited on a regular basis, and the kinds of activities that women do that are undervalued should be more recognized in terms of money and advancement within academia.
Dr. LaFaver: I couldn’t agree more. One of the other topics discussed at the Women in Neurology conference was mid-career development, retention, and career paths. I think it’s known that many women leave academic medicine mid-career. I think these are excellent suggestions and hopefully will help to make careers successful for men and women, without needing to worry about being treated differently or unfairly.
Thank you so much. This was a wonderful overview of this topic.
Dr. Loder: You’re welcome, Dr. LaFaver. Thank you so much for asking me to speak on this topic. I really appreciate it.
Dr. LaFaver: Thanks, everyone.
Dr. LaFaver is a neurologist at Saratoga Hospital Medical Group, Saratoga Springs, New York. Dr. LaFaver and Dr. Loder disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.


