User login
National poll shows ‘concerning’ impact of COVID on Americans’ mental health
Concern and anxiety around COVID-19 remains high among Americans, with more people reporting mental health effects from the pandemic this year than last, and parents concerned about the mental health of their children, results of a new poll by the American Psychiatric Association show. Although the overall level of anxiety has decreased from last year’s APA poll, “the degree to which anxiety still reigns is concerning,” APA President Jeffrey Geller, MD, MPH, told this news organization.
The results of the latest poll were presented at the American Psychiatric Association 2021 annual meeting and based on an online survey conducted March 26 to April 5 among a sample of 1,000 adults aged 18 years or older.
Serious mental health hit
In the new poll, about 4 in 10 Americans (41%) report they are more anxious than last year, down from just over 60%.
Young adults aged 18-29 years (49%) and Hispanic/Latinos (50%) are more likely to report being more anxious now than a year ago. Those 65 or older (30%) are less apt to say they feel more anxious than last year.
The latest poll also shows that Americans are more anxious about family and loved ones getting COVID-19 (64%) than about catching the virus themselves (49%).
Concern about family and loved ones contracting COVID-19 has increased since last year’s poll (conducted September 2020), rising from 56% then to 64% now. Hispanic/Latinx individuals (73%) and African American/Black individuals (76%) are more anxious about COVID-19 than White people (59%).
In the new poll, 43% of adults report the pandemic has had a serious impact on their mental health, up from 37% in 2020. Younger adults are more apt than older adults to report serious mental health effects.
Slightly fewer Americans report the pandemic is affecting their day-to-day life now as compared to a year ago, in ways such as problems sleeping (19% down from 22%), difficulty concentrating (18% down from 20%), and fighting more with loved ones (16% down from 17%).
The percentage of adults consuming more alcohol or other substances/drugs than normal increased slightly since last year (14%-17%). Additionally, 33% of adults (40% of women) report gaining weight during the pandemic.
Call to action
More than half of adults (53%) with children report they are concerned about the mental state of their children and almost half (48%) report the pandemic has caused mental health problems for one or more of their children, including minor problems for 29% and major problems for 19%.
More than a quarter (26%) of parents have sought professional mental health help for their children because of the pandemic.
; 23% received help from a primary care professional, 18% from a psychiatrist, 15% from a psychologist, 13% from a therapist, 10% from a social worker, and 10% from a school counselor or school psychologist.
More than 1 in 5 parents reported difficulty scheduling appointments for their child with a mental health professional.
“This poll shows that, even as vaccines become more widespread, Americans are still worried about the mental state of their children,” Dr. Geller said in a news release.
“This is a call to action for policymakers, who need to remember that, in our COVID-19 recovery, there’s no health without mental health,” he added.
Just over three-quarters (76%) of those surveyed say they have been or intend to get vaccinated; 22% say they don’t intend to get vaccinated; and 2% didn’t know.
For those who do not intend to get vaccinated, the primary concern (53%) is about side effects of the vaccine. Other reasons for not getting vaccinated include believing the vaccine is not effective (31%), believing the makers of the vaccine aren’t being honest about what’s in it (27%), and fear/anxiety about needles (12%).
Resiliency a finite resource
Reached for comment, Samoon Ahmad, MD, professor in the department of psychiatry, New York University, said it’s not surprising that Americans are still suffering more anxiety than normal.
“The Census Bureau’s Household Pulse Survey has shown that anxiety and depression levels have remained higher than normal since the pandemic began. That 43% of adults now say that the pandemic has had a serious impact on their mental health seems in line with what that survey has been reporting for over a year,” Dr. Ahmad, who serves as unit chief of inpatient psychiatry at Bellevue Hospital Center in New York, said in an interview.
He believes there are several reasons why anxiety levels remain high. One reason is something he’s noticed among his patients for years. “Most people struggle with anxiety especially at night when the noise and distractions of contemporary life fade away. This is the time of introspection,” he explained.
“Quarantine has been kind of like a protracted night because the distractions that are common in the so-called ‘rat race’ have been relatively muted for the past 14 months. I believe this has caused what you might call ‘forced introspection,’ and that this is giving rise to feelings of anxiety as people use their time alone to reassess their careers and their social lives and really begin to fret about some of the decisions that have led them to this point in their lives,” said Dr. Ahmad.
The other finding in the APA survey – that people are more concerned about their loved ones catching the virus than they were a year ago – is also not surprising, Dr. Ahmad said.
“Even though we seem to have turned a corner in the United States and the worst of the pandemic is behind us, the surge that went from roughly November through March of this year was more wide-reaching geographically than previous waves, and I think this made the severity of the virus far more real to people who lived in communities that had been spared severe outbreaks during the surges that we saw in the spring and summer of 2020,” Dr. Ahmad told this news organization.
“There’s also heightened concern over variants and the efficacy of the vaccine in treating these variants. Those who have families in other countries where the virus is surging, such as India or parts of Latin America, are likely experiencing additional stress and anxiety too,” he noted.
While the new APA poll findings are not surprising, they still are “deeply concerning,” Dr. Ahmad said.
“Resiliency is a finite resource, and people can only take so much stress before their mental health begins to suffer. For most people, this is not going to lead to some kind of overdramatic nervous breakdown. Instead, one may notice that they are more irritable than they once were, that they’re not sleeping particularly well, or that they have a nagging sense of discomfort and stress when doing activities that they used to think of as normal,” like taking a trip to the grocery store, meeting up with friends, or going to work, Dr. Ahmad said.
“Overcoming this kind of anxiety and reacclimating ourselves to social situations is going to take more time for some people than others, and that is perfectly natural,” said Dr. Ahmad, founder of the Integrative Center for Wellness in New York.
“I don’t think it’s wise to try to put a limit on what constitutes a normal amount of time to readjust, and I think everyone in the field of mental health needs to avoid pathologizing any lingering sense of unease. No one needs to be medicated or diagnosed with a mental illness because they are nervous about going into public spaces in the immediate aftermath of a pandemic. We need to show a lot of patience and encourage people to readjust at their own pace for the foreseeable future,” Dr. Ahmad said.
Dr. Geller and Dr. Ahmad have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Concern and anxiety around COVID-19 remains high among Americans, with more people reporting mental health effects from the pandemic this year than last, and parents concerned about the mental health of their children, results of a new poll by the American Psychiatric Association show. Although the overall level of anxiety has decreased from last year’s APA poll, “the degree to which anxiety still reigns is concerning,” APA President Jeffrey Geller, MD, MPH, told this news organization.
The results of the latest poll were presented at the American Psychiatric Association 2021 annual meeting and based on an online survey conducted March 26 to April 5 among a sample of 1,000 adults aged 18 years or older.
Serious mental health hit
In the new poll, about 4 in 10 Americans (41%) report they are more anxious than last year, down from just over 60%.
Young adults aged 18-29 years (49%) and Hispanic/Latinos (50%) are more likely to report being more anxious now than a year ago. Those 65 or older (30%) are less apt to say they feel more anxious than last year.
The latest poll also shows that Americans are more anxious about family and loved ones getting COVID-19 (64%) than about catching the virus themselves (49%).
Concern about family and loved ones contracting COVID-19 has increased since last year’s poll (conducted September 2020), rising from 56% then to 64% now. Hispanic/Latinx individuals (73%) and African American/Black individuals (76%) are more anxious about COVID-19 than White people (59%).
In the new poll, 43% of adults report the pandemic has had a serious impact on their mental health, up from 37% in 2020. Younger adults are more apt than older adults to report serious mental health effects.
Slightly fewer Americans report the pandemic is affecting their day-to-day life now as compared to a year ago, in ways such as problems sleeping (19% down from 22%), difficulty concentrating (18% down from 20%), and fighting more with loved ones (16% down from 17%).
The percentage of adults consuming more alcohol or other substances/drugs than normal increased slightly since last year (14%-17%). Additionally, 33% of adults (40% of women) report gaining weight during the pandemic.
Call to action
More than half of adults (53%) with children report they are concerned about the mental state of their children and almost half (48%) report the pandemic has caused mental health problems for one or more of their children, including minor problems for 29% and major problems for 19%.
More than a quarter (26%) of parents have sought professional mental health help for their children because of the pandemic.
; 23% received help from a primary care professional, 18% from a psychiatrist, 15% from a psychologist, 13% from a therapist, 10% from a social worker, and 10% from a school counselor or school psychologist.
More than 1 in 5 parents reported difficulty scheduling appointments for their child with a mental health professional.
“This poll shows that, even as vaccines become more widespread, Americans are still worried about the mental state of their children,” Dr. Geller said in a news release.
“This is a call to action for policymakers, who need to remember that, in our COVID-19 recovery, there’s no health without mental health,” he added.
Just over three-quarters (76%) of those surveyed say they have been or intend to get vaccinated; 22% say they don’t intend to get vaccinated; and 2% didn’t know.
For those who do not intend to get vaccinated, the primary concern (53%) is about side effects of the vaccine. Other reasons for not getting vaccinated include believing the vaccine is not effective (31%), believing the makers of the vaccine aren’t being honest about what’s in it (27%), and fear/anxiety about needles (12%).
Resiliency a finite resource
Reached for comment, Samoon Ahmad, MD, professor in the department of psychiatry, New York University, said it’s not surprising that Americans are still suffering more anxiety than normal.
“The Census Bureau’s Household Pulse Survey has shown that anxiety and depression levels have remained higher than normal since the pandemic began. That 43% of adults now say that the pandemic has had a serious impact on their mental health seems in line with what that survey has been reporting for over a year,” Dr. Ahmad, who serves as unit chief of inpatient psychiatry at Bellevue Hospital Center in New York, said in an interview.
He believes there are several reasons why anxiety levels remain high. One reason is something he’s noticed among his patients for years. “Most people struggle with anxiety especially at night when the noise and distractions of contemporary life fade away. This is the time of introspection,” he explained.
“Quarantine has been kind of like a protracted night because the distractions that are common in the so-called ‘rat race’ have been relatively muted for the past 14 months. I believe this has caused what you might call ‘forced introspection,’ and that this is giving rise to feelings of anxiety as people use their time alone to reassess their careers and their social lives and really begin to fret about some of the decisions that have led them to this point in their lives,” said Dr. Ahmad.
The other finding in the APA survey – that people are more concerned about their loved ones catching the virus than they were a year ago – is also not surprising, Dr. Ahmad said.
“Even though we seem to have turned a corner in the United States and the worst of the pandemic is behind us, the surge that went from roughly November through March of this year was more wide-reaching geographically than previous waves, and I think this made the severity of the virus far more real to people who lived in communities that had been spared severe outbreaks during the surges that we saw in the spring and summer of 2020,” Dr. Ahmad told this news organization.
“There’s also heightened concern over variants and the efficacy of the vaccine in treating these variants. Those who have families in other countries where the virus is surging, such as India or parts of Latin America, are likely experiencing additional stress and anxiety too,” he noted.
While the new APA poll findings are not surprising, they still are “deeply concerning,” Dr. Ahmad said.
“Resiliency is a finite resource, and people can only take so much stress before their mental health begins to suffer. For most people, this is not going to lead to some kind of overdramatic nervous breakdown. Instead, one may notice that they are more irritable than they once were, that they’re not sleeping particularly well, or that they have a nagging sense of discomfort and stress when doing activities that they used to think of as normal,” like taking a trip to the grocery store, meeting up with friends, or going to work, Dr. Ahmad said.
“Overcoming this kind of anxiety and reacclimating ourselves to social situations is going to take more time for some people than others, and that is perfectly natural,” said Dr. Ahmad, founder of the Integrative Center for Wellness in New York.
“I don’t think it’s wise to try to put a limit on what constitutes a normal amount of time to readjust, and I think everyone in the field of mental health needs to avoid pathologizing any lingering sense of unease. No one needs to be medicated or diagnosed with a mental illness because they are nervous about going into public spaces in the immediate aftermath of a pandemic. We need to show a lot of patience and encourage people to readjust at their own pace for the foreseeable future,” Dr. Ahmad said.
Dr. Geller and Dr. Ahmad have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Concern and anxiety around COVID-19 remains high among Americans, with more people reporting mental health effects from the pandemic this year than last, and parents concerned about the mental health of their children, results of a new poll by the American Psychiatric Association show. Although the overall level of anxiety has decreased from last year’s APA poll, “the degree to which anxiety still reigns is concerning,” APA President Jeffrey Geller, MD, MPH, told this news organization.
The results of the latest poll were presented at the American Psychiatric Association 2021 annual meeting and based on an online survey conducted March 26 to April 5 among a sample of 1,000 adults aged 18 years or older.
Serious mental health hit
In the new poll, about 4 in 10 Americans (41%) report they are more anxious than last year, down from just over 60%.
Young adults aged 18-29 years (49%) and Hispanic/Latinos (50%) are more likely to report being more anxious now than a year ago. Those 65 or older (30%) are less apt to say they feel more anxious than last year.
The latest poll also shows that Americans are more anxious about family and loved ones getting COVID-19 (64%) than about catching the virus themselves (49%).
Concern about family and loved ones contracting COVID-19 has increased since last year’s poll (conducted September 2020), rising from 56% then to 64% now. Hispanic/Latinx individuals (73%) and African American/Black individuals (76%) are more anxious about COVID-19 than White people (59%).
In the new poll, 43% of adults report the pandemic has had a serious impact on their mental health, up from 37% in 2020. Younger adults are more apt than older adults to report serious mental health effects.
Slightly fewer Americans report the pandemic is affecting their day-to-day life now as compared to a year ago, in ways such as problems sleeping (19% down from 22%), difficulty concentrating (18% down from 20%), and fighting more with loved ones (16% down from 17%).
The percentage of adults consuming more alcohol or other substances/drugs than normal increased slightly since last year (14%-17%). Additionally, 33% of adults (40% of women) report gaining weight during the pandemic.
Call to action
More than half of adults (53%) with children report they are concerned about the mental state of their children and almost half (48%) report the pandemic has caused mental health problems for one or more of their children, including minor problems for 29% and major problems for 19%.
More than a quarter (26%) of parents have sought professional mental health help for their children because of the pandemic.
; 23% received help from a primary care professional, 18% from a psychiatrist, 15% from a psychologist, 13% from a therapist, 10% from a social worker, and 10% from a school counselor or school psychologist.
More than 1 in 5 parents reported difficulty scheduling appointments for their child with a mental health professional.
“This poll shows that, even as vaccines become more widespread, Americans are still worried about the mental state of their children,” Dr. Geller said in a news release.
“This is a call to action for policymakers, who need to remember that, in our COVID-19 recovery, there’s no health without mental health,” he added.
Just over three-quarters (76%) of those surveyed say they have been or intend to get vaccinated; 22% say they don’t intend to get vaccinated; and 2% didn’t know.
For those who do not intend to get vaccinated, the primary concern (53%) is about side effects of the vaccine. Other reasons for not getting vaccinated include believing the vaccine is not effective (31%), believing the makers of the vaccine aren’t being honest about what’s in it (27%), and fear/anxiety about needles (12%).
Resiliency a finite resource
Reached for comment, Samoon Ahmad, MD, professor in the department of psychiatry, New York University, said it’s not surprising that Americans are still suffering more anxiety than normal.
“The Census Bureau’s Household Pulse Survey has shown that anxiety and depression levels have remained higher than normal since the pandemic began. That 43% of adults now say that the pandemic has had a serious impact on their mental health seems in line with what that survey has been reporting for over a year,” Dr. Ahmad, who serves as unit chief of inpatient psychiatry at Bellevue Hospital Center in New York, said in an interview.
He believes there are several reasons why anxiety levels remain high. One reason is something he’s noticed among his patients for years. “Most people struggle with anxiety especially at night when the noise and distractions of contemporary life fade away. This is the time of introspection,” he explained.
“Quarantine has been kind of like a protracted night because the distractions that are common in the so-called ‘rat race’ have been relatively muted for the past 14 months. I believe this has caused what you might call ‘forced introspection,’ and that this is giving rise to feelings of anxiety as people use their time alone to reassess their careers and their social lives and really begin to fret about some of the decisions that have led them to this point in their lives,” said Dr. Ahmad.
The other finding in the APA survey – that people are more concerned about their loved ones catching the virus than they were a year ago – is also not surprising, Dr. Ahmad said.
“Even though we seem to have turned a corner in the United States and the worst of the pandemic is behind us, the surge that went from roughly November through March of this year was more wide-reaching geographically than previous waves, and I think this made the severity of the virus far more real to people who lived in communities that had been spared severe outbreaks during the surges that we saw in the spring and summer of 2020,” Dr. Ahmad told this news organization.
“There’s also heightened concern over variants and the efficacy of the vaccine in treating these variants. Those who have families in other countries where the virus is surging, such as India or parts of Latin America, are likely experiencing additional stress and anxiety too,” he noted.
While the new APA poll findings are not surprising, they still are “deeply concerning,” Dr. Ahmad said.
“Resiliency is a finite resource, and people can only take so much stress before their mental health begins to suffer. For most people, this is not going to lead to some kind of overdramatic nervous breakdown. Instead, one may notice that they are more irritable than they once were, that they’re not sleeping particularly well, or that they have a nagging sense of discomfort and stress when doing activities that they used to think of as normal,” like taking a trip to the grocery store, meeting up with friends, or going to work, Dr. Ahmad said.
“Overcoming this kind of anxiety and reacclimating ourselves to social situations is going to take more time for some people than others, and that is perfectly natural,” said Dr. Ahmad, founder of the Integrative Center for Wellness in New York.
“I don’t think it’s wise to try to put a limit on what constitutes a normal amount of time to readjust, and I think everyone in the field of mental health needs to avoid pathologizing any lingering sense of unease. No one needs to be medicated or diagnosed with a mental illness because they are nervous about going into public spaces in the immediate aftermath of a pandemic. We need to show a lot of patience and encourage people to readjust at their own pace for the foreseeable future,” Dr. Ahmad said.
Dr. Geller and Dr. Ahmad have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FDA OKs higher-dose naloxone nasal spray for opioid overdose
The Food and Drug Administration has approved a higher-dose naloxone hydrochloride nasal spray (Kloxxado) for the emergency treatment of known or suspected opioid overdose, as manifested by respiratory and/or central nervous system depression.
Kloxxado delivers 8 mg of naloxone into the nasal cavity, which is twice as much as the 4 mg of naloxone contained in Narcan nasal spray.
When administered quickly, naloxone can counter opioid overdose effects, usually within minutes. A higher dose of naloxone provides an additional option for the treatment of opioid overdoses, the FDA said in a news release.
“This approval meets another critical need in combating opioid overdose,” Patrizia Cavazzoni, MD, director, FDA Center for Drug Evaluation and Research, said in the release.
“Addressing the opioid crisis is a top priority for the FDA, and we will continue our efforts to increase access to naloxone and place this important medicine in the hands of those who need it most,” said Dr. Cavazzoni.
In a company news release announcing the approval, manufacturer Hikma Pharmaceuticals noted that a recent survey of community organizations in which the 4-mg naloxone nasal spray had been distributed showed that for 34% of attempted reversals, two or more doses of naloxone were used.
A separate study found that the percentage of overdose-related emergency medical service calls in the United States that led to the administration of multiple doses of naloxone increased to 21% during the period of 2013-2016, which represents a 43% increase over 4 years.
“The approval of Kloxxado is an important step in providing patients, friends, and family members – as well as the public health community – with an important new option for treating opioid overdose,” Brian Hoffmann, president of Hikma Generics, said in the release.
The FDA approved Kloxxado through the 505(b)(2) regulatory pathway, which allows the agency to refer to previous findings of safety and efficacy for an already-approved product, as well as to review findings from further studies of the product.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration has approved a higher-dose naloxone hydrochloride nasal spray (Kloxxado) for the emergency treatment of known or suspected opioid overdose, as manifested by respiratory and/or central nervous system depression.
Kloxxado delivers 8 mg of naloxone into the nasal cavity, which is twice as much as the 4 mg of naloxone contained in Narcan nasal spray.
When administered quickly, naloxone can counter opioid overdose effects, usually within minutes. A higher dose of naloxone provides an additional option for the treatment of opioid overdoses, the FDA said in a news release.
“This approval meets another critical need in combating opioid overdose,” Patrizia Cavazzoni, MD, director, FDA Center for Drug Evaluation and Research, said in the release.
“Addressing the opioid crisis is a top priority for the FDA, and we will continue our efforts to increase access to naloxone and place this important medicine in the hands of those who need it most,” said Dr. Cavazzoni.
In a company news release announcing the approval, manufacturer Hikma Pharmaceuticals noted that a recent survey of community organizations in which the 4-mg naloxone nasal spray had been distributed showed that for 34% of attempted reversals, two or more doses of naloxone were used.
A separate study found that the percentage of overdose-related emergency medical service calls in the United States that led to the administration of multiple doses of naloxone increased to 21% during the period of 2013-2016, which represents a 43% increase over 4 years.
“The approval of Kloxxado is an important step in providing patients, friends, and family members – as well as the public health community – with an important new option for treating opioid overdose,” Brian Hoffmann, president of Hikma Generics, said in the release.
The FDA approved Kloxxado through the 505(b)(2) regulatory pathway, which allows the agency to refer to previous findings of safety and efficacy for an already-approved product, as well as to review findings from further studies of the product.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration has approved a higher-dose naloxone hydrochloride nasal spray (Kloxxado) for the emergency treatment of known or suspected opioid overdose, as manifested by respiratory and/or central nervous system depression.
Kloxxado delivers 8 mg of naloxone into the nasal cavity, which is twice as much as the 4 mg of naloxone contained in Narcan nasal spray.
When administered quickly, naloxone can counter opioid overdose effects, usually within minutes. A higher dose of naloxone provides an additional option for the treatment of opioid overdoses, the FDA said in a news release.
“This approval meets another critical need in combating opioid overdose,” Patrizia Cavazzoni, MD, director, FDA Center for Drug Evaluation and Research, said in the release.
“Addressing the opioid crisis is a top priority for the FDA, and we will continue our efforts to increase access to naloxone and place this important medicine in the hands of those who need it most,” said Dr. Cavazzoni.
In a company news release announcing the approval, manufacturer Hikma Pharmaceuticals noted that a recent survey of community organizations in which the 4-mg naloxone nasal spray had been distributed showed that for 34% of attempted reversals, two or more doses of naloxone were used.
A separate study found that the percentage of overdose-related emergency medical service calls in the United States that led to the administration of multiple doses of naloxone increased to 21% during the period of 2013-2016, which represents a 43% increase over 4 years.
“The approval of Kloxxado is an important step in providing patients, friends, and family members – as well as the public health community – with an important new option for treating opioid overdose,” Brian Hoffmann, president of Hikma Generics, said in the release.
The FDA approved Kloxxado through the 505(b)(2) regulatory pathway, which allows the agency to refer to previous findings of safety and efficacy for an already-approved product, as well as to review findings from further studies of the product.
A version of this article first appeared on Medscape.com.
The cloudy role of cannabis as a neuropsychiatric treatment
Although the healing properties of cannabis have been touted for millennia, research into its potential neuropsychiatric applications truly began to take off in the 1990s following the discovery of the cannabinoid system in the brain. This led to speculation that cannabis could play a therapeutic role in regulating dopamine, serotonin, and other neurotransmitters and offer a new means of treating various ailments.
At the same time, efforts to liberalize marijuana laws have successfully played out in several nations, including the United States, where, as of April 29, 36 states provide some access to cannabis. These dual tracks – medical and political – have made cannabis an increasingly accepted part of the cultural fabric.
Yet with this development has come a new quandary for clinicians. Medical cannabis has been made widely available to patients and has largely outpaced the clinical evidence, leaving it unclear how and for which indications it should be used.
The many forms of medical cannabis
Cannabis is a genus of plants that includes marijuana (Cannabis sativa) and hemp. These plants contain over 100 compounds, including terpenes, flavonoids, and – most importantly for medicinal applications – cannabinoids.
The most abundant cannabinoid in marijuana is the psychotropic delta-9-tetrahydrocannabinol (THC), which imparts the “high” sensation. The next most abundant cannabinoid is cannabidiol (CBD), which is the nonpsychotropic. THC and CBD are the most extensively studied cannabinoids, together and in isolation. Evidence suggests that other cannabinoids and terpenoids may also hold medical promise and that cannabis’ various compounds can work synergistically to produce a so-called entourage effect.
Patients walking into a typical medical cannabis dispensary will be faced with several plant-derived and synthetic options, which can differ considerably in terms of the ratios and amounts of THC and CBD they contain, as well in how they are consumed (i.e., via smoke, vapor, ingestion, topical administration, or oromucosal spray), all of which can alter their effects. Further complicating matters is the varying level of oversight each state and country has in how and whether they test for and accurately label products’ potency, cannabinoid content, and possible impurities.
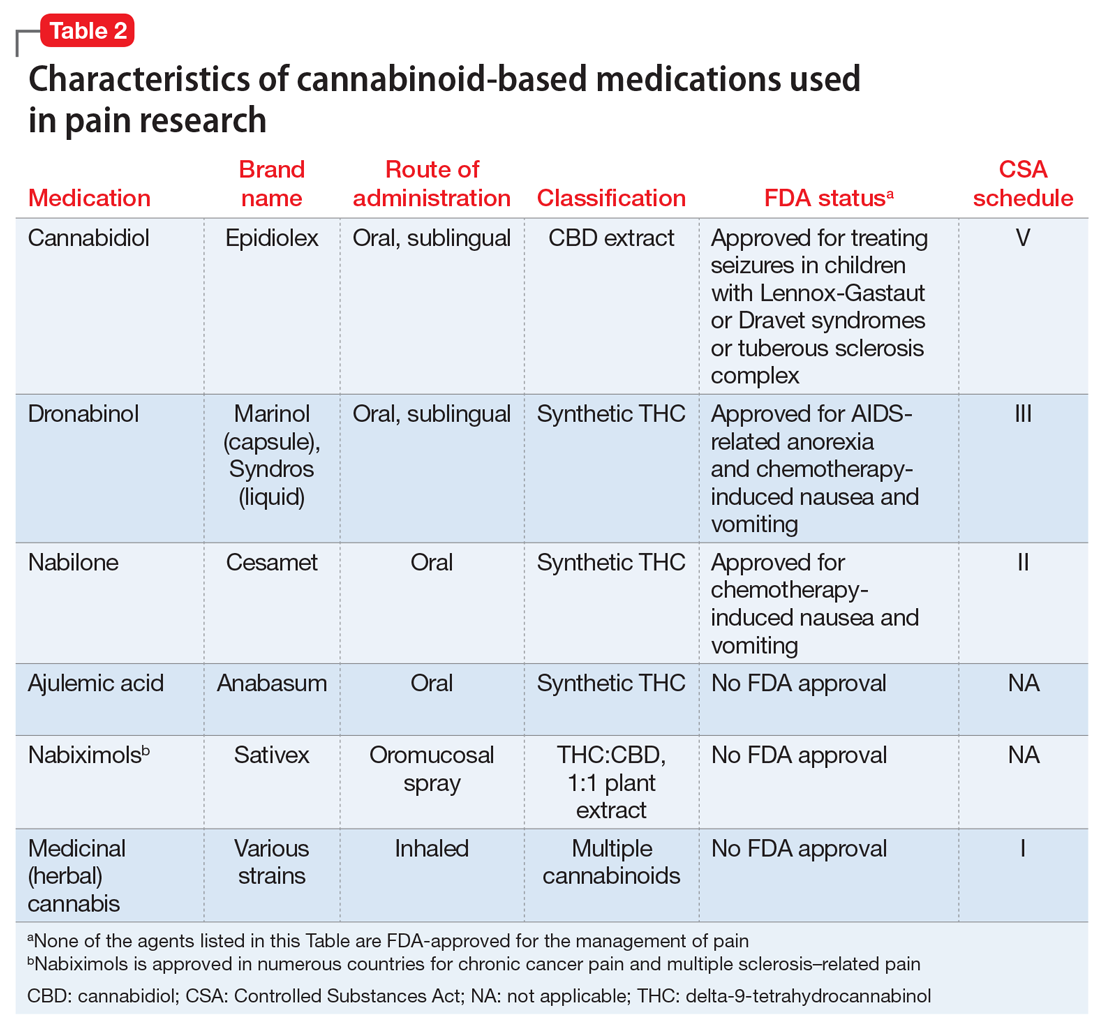
Medically authorized, prescription cannabis products go through an official regulatory review process, and indications/contraindications have been established for them. To date, the Food and Drug Administration has approved one cannabis-derived drug product – Epidiolex (purified CBD) – for the treatment of seizures associated with Lennox-Gastaut syndrome or Dravet syndrome in patients aged 2 years and older. The FDA has also approved three synthetic cannabis-related drug products – Marinol, Syndros (or dronabinol, created from synthetic THC), and Cesamet (or nabilone, a synthetic cannabinoid similar to THC) – all of which are indicated for treatment-related nausea and anorexia associated with weight loss in AIDS patients.
Surveys of medical cannabis consumers indicate that most people cannot distinguish between THC and CBD, so the first role that physicians find themselves in when recommending this treatment may be in helping patients navigate the volume of options.
Promising treatment for pain
Chronic pain is the leading reason patients seek out medical cannabis. It is also the indication that most researchers agree has the strongest evidence to support its use.
“In my mind, the most promising immediate use for medical cannabis is with THC for pain,” Diana M. Martinez, MD, a professor of psychiatry at Columbia University, New York, who specializes in addiction research, said in a recent MDedge podcast. “THC could be added to the armamentarium of pain medications that we use today.”
In a 2015 systematic literature review, researchers assessed 28 randomized, controlled trials (RCTs) of the use of cannabinoids for chronic pain. They reported that a variety of formulations resulted in at least a 30% reduction in the odds of pain, compared with placebo. A meta-analysis of five RCTs involving patients with neuropathic pain found a 30% reduction in pain over placebo with inhaled, vaporized cannabis. Varying results have been reported in additional studies for this indication. The National Academies of Sciences, Engineering, and Medicine concluded that there was a substantial body of evidence that cannabis is an effective treatment for chronic pain in adults.
The ongoing opioid epidemic has lent these results additional relevance.
Seeing this firsthand has caused Mark Steven Wallace, MD, a pain management specialist and chair of the division of pain medicine at the University of California San Diego Health, to reconsider offering cannabis to his patients.
“I think it’s probably more efficacious, just from my personal experience, and it’s a much lower risk of abuse and dependence than the opioids,” he said.
Dr. Wallace advised that clinicians who treat pain consider the ratios of cannabinoids.
“This is anecdotal, but we do find that with the combination of the two, CBD reduces the psychoactive effects of the THC. The ratios we use during the daytime range around 20 mg of CBD to 1 mg of THC,” he said.
In a recent secondary analysis of an RCT involving patients with painful diabetic peripheral neuropathy, Dr. Wallace and colleagues showed that THC’s effects appear to reverse themselves at a certain level.
“As the THC level goes up, the pain reduces until you reach about 16 ng/mL; then it starts going in the opposite direction, and pain will start to increase,” he said. “Even recreational cannabis users have reported that they avoid high doses because it’s very aversive. Using cannabis is all about, start low and go slow.”
A mixed bag for neurologic indications
There are relatively limited data on the use of medical cannabis for other neurologic conditions, and results have varied. For uses other than pain management, the evidence that does exist is strongest regarding epilepsy, said Daniel Freedman, DO, assistant professor of neurology at the University of Texas at Austin. He noted “multiple high-quality RCTs showing that pharmaceutical-grade CBD can reduce seizures associated with two particular epilepsy syndromes: Dravet Syndrome and Lennox Gastaut.”
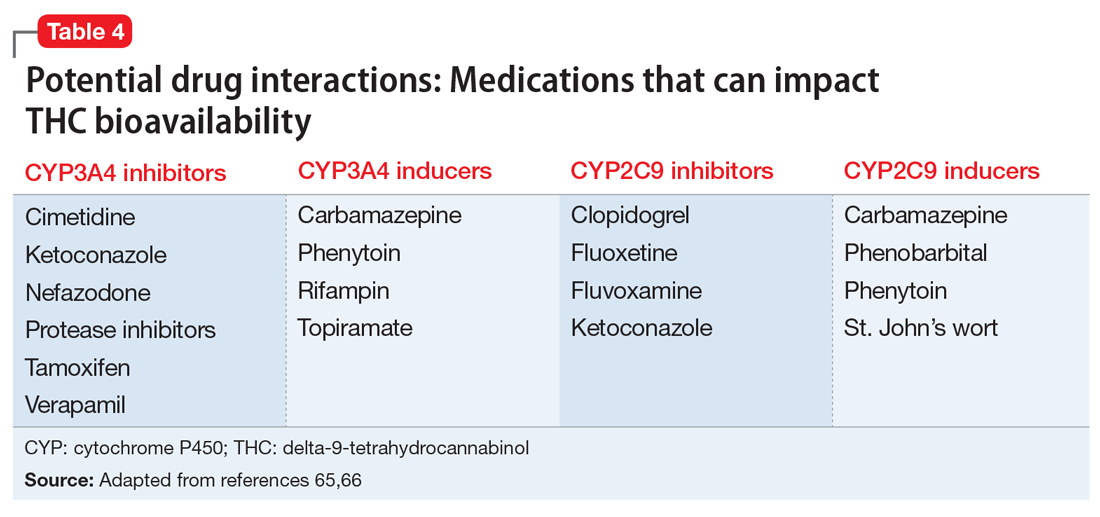
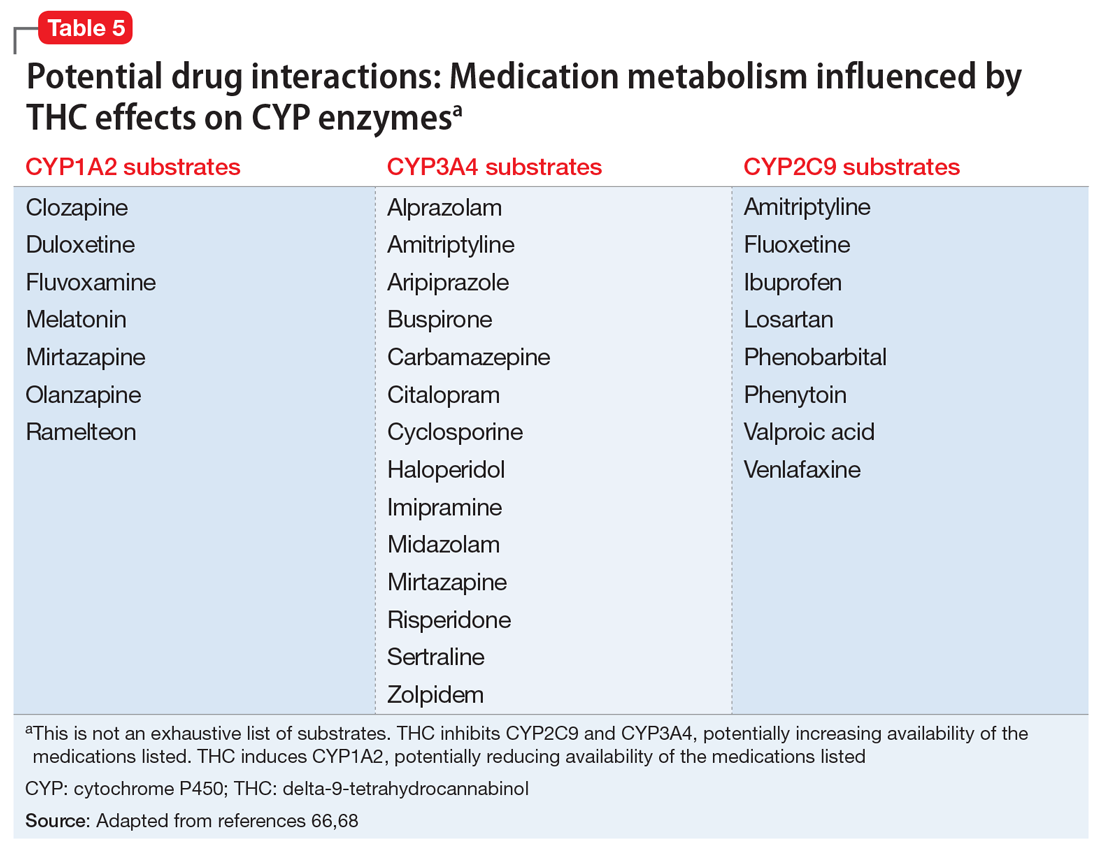
These findings led to the FDA’s 2018 approval of Epidiolex for these syndromes. In earlier years, interest in CBD for pediatric seizures was largely driven by anecdotal parental reports of its benefits. NASEM’s 2017 overview on medical cannabis found evidence from subsequent RCTs in this indication to be insufficient. Clinicians who prescribe CBD for this indication must be vigilant because it can interact with several commonly used antiepileptic drugs.
Cannabinoid treatments have also shown success in alleviating muscle spasticity resulting from multiple sclerosis, most prominently in the form of nabiximols (Sativex), a standardized oralmucosal spray containing approximately equal quantities of THC and CBD. Nabiximols is approved in Europe but not in the United States. Moderate evidence supports the efficacy of these and other treatments over placebo in reducing muscle spasticity. Patient ratings of its effects tend to be higher than clinician assessment.
Parkinson’s disease has not yet been approved as an indication for treatment with cannabis or cannabinoids, yet a growing body of preclinical data suggests these could influence the dopaminergic system, said Carsten Buhmann, MD, from the department of neurology at the University Medical Center Hamburg-Eppendorf (Germany).
“In general, cannabinoids modulate basal-ganglia function on two levels which are especially relevant in Parkinson’s disease, i.e., the glutamatergic/dopaminergic synaptic neurotransmission and the corticostriatal plasticity,” he said. “Furthermore, activation of the endocannabinoid system might induce neuroprotective effects related to direct receptor-independent mechanisms, activation of anti-inflammatory cascades in glial cells via the cannabinoid receptor type 2, and antiglutamatergic antiexcitotoxic properties.”
Dr. Buhmann said that currently, clinical evidence is scarce, consisting of only four double-blind, placebo-controlled RCTs involving 49 patients. Various cannabinoids and methods of administering treatment were employed. Improvement was only observed in one of these RCTs, which found that the cannabinoid receptor agonist nabilone significantly reduced levodopa-induced dyskinesia for patients with Parkinson’s disease. Subjective data support a beneficial effect. In a nationwide survey of 1,348 respondents conducted by Dr. Buhmann and colleagues, the majority of medical cannabis users reported that it improved their symptoms (54% with oral CBD and 68% with inhaled THC-containing cannabis).
NASEM concluded that there was insufficient evidence to support the efficacy of medical cannabis for other neurologic conditions, including Tourette syndrome, amyotrophic lateral sclerosis, Huntington disease, dystonia, or dementia. A 2020 position statement from the American Academy of Neurology cited the lack of sufficient peer-reviewed research as the reason it could not currently support the use of cannabis for neurologic disorders.
Yet, according to Dr. Freedman, who served as a coauthor of the AAN position statement, this hasn’t stymied research interest in the topic. He’s seen a substantial uptick in studies of CBD over the past 2 years. “The body of evidence grows, but I still see many claims being made without evidence. And no one seems to care about all the negative trials.”
Cannabis as a treatment for, and cause of, psychiatric disorders
Mental health problems – such as anxiety, depression, and PTSD – are among the most common reasons patients seek out medical cannabis. There is an understandable interest in using cannabis and cannabinoids to treat psychiatric disorders. Preclinical studies suggest that the endocannabinoid system plays a prominent role in modulating feelings of anxiety, mood, and fear. As with opioids and chronic pain management, there is hope that medical cannabis may provide a means of reducing prescription anxiolytics and their associated risks.
The authors of the first systematic review (BMC Psychiatry. 2020 Jan 16;20[1]:24) of the use of medical cannabis for major psychiatric disorders noted that the current evidence was “encouraging, albeit embryonic.”
Meta-analyses have indicated a small but positive association between cannabis use and anxiety, although this may reflect the fact that patients with anxiety sought out this treatment. Given the risks for substance use disorders among patients with anxiety, CBD may present a more viable option. Positive results have been shown as treatment for generalized social anxiety disorder.
Limited but encouraging results have also been reported regarding the alleviation of PTSD symptoms with both cannabis and CBD, although the body of high-quality evidence hasn’t notably progressed since 2017, when NASEM declared that the evidence was insufficient. Supportive evidence is similarly lacking regarding the treatment of depression. Longitudinal studies suggest that cannabis use, particularly heavy use, may increase the risk of developing this disorder. Because THC is psychoactive, it is advised that it be avoided by patients at risk for psychotic disorders. However, CBD has yielded limited benefits for patients with treatment-resistant schizophrenia and for young people at risk for psychosis.
The use of medical cannabis for psychiatric conditions requires a complex balancing act, inasmuch as these treatments may exacerbate the very problems they are intended to alleviate.
Marta Di Forti, MD, PhD, professor of psychiatric research at Kings College London, has been at the forefront of determining the mental health risks of continued cannabis use. In 2019, Dr. Di Forti developed the first and only Cannabis Clinic for Patients With Psychosis in London where she and her colleagues have continued to elucidate this connection.
Dr. Di Forti and colleagues have linked daily cannabis use to an increase in the risk of experiencing psychotic disorder, compared with never using it. That risk was further increased among users of high-potency cannabis (≥10% THC). The latter finding has troubling implications, because concentrations of THC have steadily risen since 1970. By contrast, CBD concentrations have remained generally stable. High-potency cannabis products are common in both recreational and medicinal settings.
“For somebody prescribing medicinal cannabis that has a ≥10% concentration of THC, I’d be particularly wary of the risk of psychosis,” said Dr. Di Forti. “If you’re expecting people to use a high content of THC daily to medicate pain or a chronic condition, you even more so need to be aware that this is a potential side effect.”
Dr. Di Forti noted that her findings come from a cohort of recreational users, most of whom were aged 18-35 years.
“There have actually not been studies developed from collecting data in this area from groups specifically using cannabis for medicinal rather than recreational purposes,” she said.
She added that she personally has no concerns about the use of medical cannabis but wants clinicians to be aware of the risk for psychosis, to structure their patient conversations to identify risk factors or family histories of psychosis, and to become knowledgeable in detecting the often subtle signs of its initial onset.
When cannabis-associated psychosis occurs, Dr. Di Forti said it is primarily treated with conventional means, such as antipsychotics and therapeutic interventions and by refraining from using cannabis. Achieving the latter goal can be a challenge for patients who are daily users of high-potency cannabis. Currently, there are no treatment options such as those offered to patients withdrawing from the use of alcohol or opioids. Dr. Di Forti and colleagues are currently researching a solution to that problem through the use of another medical cannabis, the oromucosal spray Sativex, which has been approved in the European Union.
The regulatory obstacles to clarifying cannabis’ role in medicine
That currently there is limited or no evidence to support the use of medical cannabis for the treatment of neuropsychiatric conditions points to the inherent difficulties in conducting high-level research in this area.
“There’s a tremendous shortage of reliable data, largely due to regulatory barriers,” said Dr. Martinez.
Since 1970, cannabis has been listed as a Schedule I drug that is illegal to prescribe (the Agriculture Improvement Act of 2018 removed hemp from such restrictions). The FDA has issued guidance for researchers who wish to investigate treatments using Cannabis sativa or its derivatives in which the THC content is greater than 0.3%. Such research requires regular interactions with several federal agencies, including the Drug Enforcement Administration.
“It’s impossible to do multicenter RCTs with large numbers of patients, because you can’t transport cannabis across state lines,” said Dr. Wallace.
Regulatory restrictions regarding medical cannabis vary considerably throughout the world (the European Monitoring Center for Drugs and Drug Addiction provides a useful breakdown of this on their website). The lack of consistency in regulatory oversight acts as an impediment for conducting large-scale international multicenter studies on the topic.
Dr. Buhmann noted that, in Germany, cannabis has been broadly approved for treatment-resistant conditions with severe symptoms that impair quality of life. In addition, it is easy to be reimbursed for the use of cannabis as a medical treatment. These factors serve as disincentives for the funding of high-quality studies.
“It’s likely that no pharmaceutical company will do an expensive RCT to get an approval for Parkinson’s disease because it is already possible to prescribe medical cannabis of any type of THC-containing cannabinoid, dose, or route of application,” Dr. Buhmann said.
In the face of such restrictions and barriers, researchers are turning to ambitious real-world data projects to better understand medical cannabis’ efficacy and safety. A notable example is ProjectTwenty21, which is supported by the Royal College of Psychiatrists. The project is collecting outcomes of the use of medical cannabis among 20,000 U.K. patients whose conventional treatments of chronic pain, anxiety disorder, epilepsy, multiple sclerosis, PTSD, substance use disorder, and Tourette syndrome failed.
Dr. Freedman noted that the continued lack of high-quality data creates a void that commercial interests fill with unfounded claims.
“The danger is that patients might abandon a medication or intervention backed by robust science in favor of something without any science or evidence behind it,” he said. “There is no reason not to expect the same level of data for claims about cannabis products as we would expect from pharmaceutical products.”
Getting to that point, however, will require that the authorities governing clinical trials begin to view cannabis as the research community does, as a possible treatment with potential value, rather than as an illicit drug that needs to be tamped down.
A version of this article first appeared on Medscape.com.
Although the healing properties of cannabis have been touted for millennia, research into its potential neuropsychiatric applications truly began to take off in the 1990s following the discovery of the cannabinoid system in the brain. This led to speculation that cannabis could play a therapeutic role in regulating dopamine, serotonin, and other neurotransmitters and offer a new means of treating various ailments.
At the same time, efforts to liberalize marijuana laws have successfully played out in several nations, including the United States, where, as of April 29, 36 states provide some access to cannabis. These dual tracks – medical and political – have made cannabis an increasingly accepted part of the cultural fabric.
Yet with this development has come a new quandary for clinicians. Medical cannabis has been made widely available to patients and has largely outpaced the clinical evidence, leaving it unclear how and for which indications it should be used.
The many forms of medical cannabis
Cannabis is a genus of plants that includes marijuana (Cannabis sativa) and hemp. These plants contain over 100 compounds, including terpenes, flavonoids, and – most importantly for medicinal applications – cannabinoids.
The most abundant cannabinoid in marijuana is the psychotropic delta-9-tetrahydrocannabinol (THC), which imparts the “high” sensation. The next most abundant cannabinoid is cannabidiol (CBD), which is the nonpsychotropic. THC and CBD are the most extensively studied cannabinoids, together and in isolation. Evidence suggests that other cannabinoids and terpenoids may also hold medical promise and that cannabis’ various compounds can work synergistically to produce a so-called entourage effect.
Patients walking into a typical medical cannabis dispensary will be faced with several plant-derived and synthetic options, which can differ considerably in terms of the ratios and amounts of THC and CBD they contain, as well in how they are consumed (i.e., via smoke, vapor, ingestion, topical administration, or oromucosal spray), all of which can alter their effects. Further complicating matters is the varying level of oversight each state and country has in how and whether they test for and accurately label products’ potency, cannabinoid content, and possible impurities.
Medically authorized, prescription cannabis products go through an official regulatory review process, and indications/contraindications have been established for them. To date, the Food and Drug Administration has approved one cannabis-derived drug product – Epidiolex (purified CBD) – for the treatment of seizures associated with Lennox-Gastaut syndrome or Dravet syndrome in patients aged 2 years and older. The FDA has also approved three synthetic cannabis-related drug products – Marinol, Syndros (or dronabinol, created from synthetic THC), and Cesamet (or nabilone, a synthetic cannabinoid similar to THC) – all of which are indicated for treatment-related nausea and anorexia associated with weight loss in AIDS patients.
Surveys of medical cannabis consumers indicate that most people cannot distinguish between THC and CBD, so the first role that physicians find themselves in when recommending this treatment may be in helping patients navigate the volume of options.
Promising treatment for pain
Chronic pain is the leading reason patients seek out medical cannabis. It is also the indication that most researchers agree has the strongest evidence to support its use.
“In my mind, the most promising immediate use for medical cannabis is with THC for pain,” Diana M. Martinez, MD, a professor of psychiatry at Columbia University, New York, who specializes in addiction research, said in a recent MDedge podcast. “THC could be added to the armamentarium of pain medications that we use today.”
In a 2015 systematic literature review, researchers assessed 28 randomized, controlled trials (RCTs) of the use of cannabinoids for chronic pain. They reported that a variety of formulations resulted in at least a 30% reduction in the odds of pain, compared with placebo. A meta-analysis of five RCTs involving patients with neuropathic pain found a 30% reduction in pain over placebo with inhaled, vaporized cannabis. Varying results have been reported in additional studies for this indication. The National Academies of Sciences, Engineering, and Medicine concluded that there was a substantial body of evidence that cannabis is an effective treatment for chronic pain in adults.
The ongoing opioid epidemic has lent these results additional relevance.
Seeing this firsthand has caused Mark Steven Wallace, MD, a pain management specialist and chair of the division of pain medicine at the University of California San Diego Health, to reconsider offering cannabis to his patients.
“I think it’s probably more efficacious, just from my personal experience, and it’s a much lower risk of abuse and dependence than the opioids,” he said.
Dr. Wallace advised that clinicians who treat pain consider the ratios of cannabinoids.
“This is anecdotal, but we do find that with the combination of the two, CBD reduces the psychoactive effects of the THC. The ratios we use during the daytime range around 20 mg of CBD to 1 mg of THC,” he said.
In a recent secondary analysis of an RCT involving patients with painful diabetic peripheral neuropathy, Dr. Wallace and colleagues showed that THC’s effects appear to reverse themselves at a certain level.
“As the THC level goes up, the pain reduces until you reach about 16 ng/mL; then it starts going in the opposite direction, and pain will start to increase,” he said. “Even recreational cannabis users have reported that they avoid high doses because it’s very aversive. Using cannabis is all about, start low and go slow.”
A mixed bag for neurologic indications
There are relatively limited data on the use of medical cannabis for other neurologic conditions, and results have varied. For uses other than pain management, the evidence that does exist is strongest regarding epilepsy, said Daniel Freedman, DO, assistant professor of neurology at the University of Texas at Austin. He noted “multiple high-quality RCTs showing that pharmaceutical-grade CBD can reduce seizures associated with two particular epilepsy syndromes: Dravet Syndrome and Lennox Gastaut.”
These findings led to the FDA’s 2018 approval of Epidiolex for these syndromes. In earlier years, interest in CBD for pediatric seizures was largely driven by anecdotal parental reports of its benefits. NASEM’s 2017 overview on medical cannabis found evidence from subsequent RCTs in this indication to be insufficient. Clinicians who prescribe CBD for this indication must be vigilant because it can interact with several commonly used antiepileptic drugs.
Cannabinoid treatments have also shown success in alleviating muscle spasticity resulting from multiple sclerosis, most prominently in the form of nabiximols (Sativex), a standardized oralmucosal spray containing approximately equal quantities of THC and CBD. Nabiximols is approved in Europe but not in the United States. Moderate evidence supports the efficacy of these and other treatments over placebo in reducing muscle spasticity. Patient ratings of its effects tend to be higher than clinician assessment.
Parkinson’s disease has not yet been approved as an indication for treatment with cannabis or cannabinoids, yet a growing body of preclinical data suggests these could influence the dopaminergic system, said Carsten Buhmann, MD, from the department of neurology at the University Medical Center Hamburg-Eppendorf (Germany).
“In general, cannabinoids modulate basal-ganglia function on two levels which are especially relevant in Parkinson’s disease, i.e., the glutamatergic/dopaminergic synaptic neurotransmission and the corticostriatal plasticity,” he said. “Furthermore, activation of the endocannabinoid system might induce neuroprotective effects related to direct receptor-independent mechanisms, activation of anti-inflammatory cascades in glial cells via the cannabinoid receptor type 2, and antiglutamatergic antiexcitotoxic properties.”
Dr. Buhmann said that currently, clinical evidence is scarce, consisting of only four double-blind, placebo-controlled RCTs involving 49 patients. Various cannabinoids and methods of administering treatment were employed. Improvement was only observed in one of these RCTs, which found that the cannabinoid receptor agonist nabilone significantly reduced levodopa-induced dyskinesia for patients with Parkinson’s disease. Subjective data support a beneficial effect. In a nationwide survey of 1,348 respondents conducted by Dr. Buhmann and colleagues, the majority of medical cannabis users reported that it improved their symptoms (54% with oral CBD and 68% with inhaled THC-containing cannabis).
NASEM concluded that there was insufficient evidence to support the efficacy of medical cannabis for other neurologic conditions, including Tourette syndrome, amyotrophic lateral sclerosis, Huntington disease, dystonia, or dementia. A 2020 position statement from the American Academy of Neurology cited the lack of sufficient peer-reviewed research as the reason it could not currently support the use of cannabis for neurologic disorders.
Yet, according to Dr. Freedman, who served as a coauthor of the AAN position statement, this hasn’t stymied research interest in the topic. He’s seen a substantial uptick in studies of CBD over the past 2 years. “The body of evidence grows, but I still see many claims being made without evidence. And no one seems to care about all the negative trials.”
Cannabis as a treatment for, and cause of, psychiatric disorders
Mental health problems – such as anxiety, depression, and PTSD – are among the most common reasons patients seek out medical cannabis. There is an understandable interest in using cannabis and cannabinoids to treat psychiatric disorders. Preclinical studies suggest that the endocannabinoid system plays a prominent role in modulating feelings of anxiety, mood, and fear. As with opioids and chronic pain management, there is hope that medical cannabis may provide a means of reducing prescription anxiolytics and their associated risks.
The authors of the first systematic review (BMC Psychiatry. 2020 Jan 16;20[1]:24) of the use of medical cannabis for major psychiatric disorders noted that the current evidence was “encouraging, albeit embryonic.”
Meta-analyses have indicated a small but positive association between cannabis use and anxiety, although this may reflect the fact that patients with anxiety sought out this treatment. Given the risks for substance use disorders among patients with anxiety, CBD may present a more viable option. Positive results have been shown as treatment for generalized social anxiety disorder.
Limited but encouraging results have also been reported regarding the alleviation of PTSD symptoms with both cannabis and CBD, although the body of high-quality evidence hasn’t notably progressed since 2017, when NASEM declared that the evidence was insufficient. Supportive evidence is similarly lacking regarding the treatment of depression. Longitudinal studies suggest that cannabis use, particularly heavy use, may increase the risk of developing this disorder. Because THC is psychoactive, it is advised that it be avoided by patients at risk for psychotic disorders. However, CBD has yielded limited benefits for patients with treatment-resistant schizophrenia and for young people at risk for psychosis.
The use of medical cannabis for psychiatric conditions requires a complex balancing act, inasmuch as these treatments may exacerbate the very problems they are intended to alleviate.
Marta Di Forti, MD, PhD, professor of psychiatric research at Kings College London, has been at the forefront of determining the mental health risks of continued cannabis use. In 2019, Dr. Di Forti developed the first and only Cannabis Clinic for Patients With Psychosis in London where she and her colleagues have continued to elucidate this connection.
Dr. Di Forti and colleagues have linked daily cannabis use to an increase in the risk of experiencing psychotic disorder, compared with never using it. That risk was further increased among users of high-potency cannabis (≥10% THC). The latter finding has troubling implications, because concentrations of THC have steadily risen since 1970. By contrast, CBD concentrations have remained generally stable. High-potency cannabis products are common in both recreational and medicinal settings.
“For somebody prescribing medicinal cannabis that has a ≥10% concentration of THC, I’d be particularly wary of the risk of psychosis,” said Dr. Di Forti. “If you’re expecting people to use a high content of THC daily to medicate pain or a chronic condition, you even more so need to be aware that this is a potential side effect.”
Dr. Di Forti noted that her findings come from a cohort of recreational users, most of whom were aged 18-35 years.
“There have actually not been studies developed from collecting data in this area from groups specifically using cannabis for medicinal rather than recreational purposes,” she said.
She added that she personally has no concerns about the use of medical cannabis but wants clinicians to be aware of the risk for psychosis, to structure their patient conversations to identify risk factors or family histories of psychosis, and to become knowledgeable in detecting the often subtle signs of its initial onset.
When cannabis-associated psychosis occurs, Dr. Di Forti said it is primarily treated with conventional means, such as antipsychotics and therapeutic interventions and by refraining from using cannabis. Achieving the latter goal can be a challenge for patients who are daily users of high-potency cannabis. Currently, there are no treatment options such as those offered to patients withdrawing from the use of alcohol or opioids. Dr. Di Forti and colleagues are currently researching a solution to that problem through the use of another medical cannabis, the oromucosal spray Sativex, which has been approved in the European Union.
The regulatory obstacles to clarifying cannabis’ role in medicine
That currently there is limited or no evidence to support the use of medical cannabis for the treatment of neuropsychiatric conditions points to the inherent difficulties in conducting high-level research in this area.
“There’s a tremendous shortage of reliable data, largely due to regulatory barriers,” said Dr. Martinez.
Since 1970, cannabis has been listed as a Schedule I drug that is illegal to prescribe (the Agriculture Improvement Act of 2018 removed hemp from such restrictions). The FDA has issued guidance for researchers who wish to investigate treatments using Cannabis sativa or its derivatives in which the THC content is greater than 0.3%. Such research requires regular interactions with several federal agencies, including the Drug Enforcement Administration.
“It’s impossible to do multicenter RCTs with large numbers of patients, because you can’t transport cannabis across state lines,” said Dr. Wallace.
Regulatory restrictions regarding medical cannabis vary considerably throughout the world (the European Monitoring Center for Drugs and Drug Addiction provides a useful breakdown of this on their website). The lack of consistency in regulatory oversight acts as an impediment for conducting large-scale international multicenter studies on the topic.
Dr. Buhmann noted that, in Germany, cannabis has been broadly approved for treatment-resistant conditions with severe symptoms that impair quality of life. In addition, it is easy to be reimbursed for the use of cannabis as a medical treatment. These factors serve as disincentives for the funding of high-quality studies.
“It’s likely that no pharmaceutical company will do an expensive RCT to get an approval for Parkinson’s disease because it is already possible to prescribe medical cannabis of any type of THC-containing cannabinoid, dose, or route of application,” Dr. Buhmann said.
In the face of such restrictions and barriers, researchers are turning to ambitious real-world data projects to better understand medical cannabis’ efficacy and safety. A notable example is ProjectTwenty21, which is supported by the Royal College of Psychiatrists. The project is collecting outcomes of the use of medical cannabis among 20,000 U.K. patients whose conventional treatments of chronic pain, anxiety disorder, epilepsy, multiple sclerosis, PTSD, substance use disorder, and Tourette syndrome failed.
Dr. Freedman noted that the continued lack of high-quality data creates a void that commercial interests fill with unfounded claims.
“The danger is that patients might abandon a medication or intervention backed by robust science in favor of something without any science or evidence behind it,” he said. “There is no reason not to expect the same level of data for claims about cannabis products as we would expect from pharmaceutical products.”
Getting to that point, however, will require that the authorities governing clinical trials begin to view cannabis as the research community does, as a possible treatment with potential value, rather than as an illicit drug that needs to be tamped down.
A version of this article first appeared on Medscape.com.
Although the healing properties of cannabis have been touted for millennia, research into its potential neuropsychiatric applications truly began to take off in the 1990s following the discovery of the cannabinoid system in the brain. This led to speculation that cannabis could play a therapeutic role in regulating dopamine, serotonin, and other neurotransmitters and offer a new means of treating various ailments.
At the same time, efforts to liberalize marijuana laws have successfully played out in several nations, including the United States, where, as of April 29, 36 states provide some access to cannabis. These dual tracks – medical and political – have made cannabis an increasingly accepted part of the cultural fabric.
Yet with this development has come a new quandary for clinicians. Medical cannabis has been made widely available to patients and has largely outpaced the clinical evidence, leaving it unclear how and for which indications it should be used.
The many forms of medical cannabis
Cannabis is a genus of plants that includes marijuana (Cannabis sativa) and hemp. These plants contain over 100 compounds, including terpenes, flavonoids, and – most importantly for medicinal applications – cannabinoids.
The most abundant cannabinoid in marijuana is the psychotropic delta-9-tetrahydrocannabinol (THC), which imparts the “high” sensation. The next most abundant cannabinoid is cannabidiol (CBD), which is the nonpsychotropic. THC and CBD are the most extensively studied cannabinoids, together and in isolation. Evidence suggests that other cannabinoids and terpenoids may also hold medical promise and that cannabis’ various compounds can work synergistically to produce a so-called entourage effect.
Patients walking into a typical medical cannabis dispensary will be faced with several plant-derived and synthetic options, which can differ considerably in terms of the ratios and amounts of THC and CBD they contain, as well in how they are consumed (i.e., via smoke, vapor, ingestion, topical administration, or oromucosal spray), all of which can alter their effects. Further complicating matters is the varying level of oversight each state and country has in how and whether they test for and accurately label products’ potency, cannabinoid content, and possible impurities.
Medically authorized, prescription cannabis products go through an official regulatory review process, and indications/contraindications have been established for them. To date, the Food and Drug Administration has approved one cannabis-derived drug product – Epidiolex (purified CBD) – for the treatment of seizures associated with Lennox-Gastaut syndrome or Dravet syndrome in patients aged 2 years and older. The FDA has also approved three synthetic cannabis-related drug products – Marinol, Syndros (or dronabinol, created from synthetic THC), and Cesamet (or nabilone, a synthetic cannabinoid similar to THC) – all of which are indicated for treatment-related nausea and anorexia associated with weight loss in AIDS patients.
Surveys of medical cannabis consumers indicate that most people cannot distinguish between THC and CBD, so the first role that physicians find themselves in when recommending this treatment may be in helping patients navigate the volume of options.
Promising treatment for pain
Chronic pain is the leading reason patients seek out medical cannabis. It is also the indication that most researchers agree has the strongest evidence to support its use.
“In my mind, the most promising immediate use for medical cannabis is with THC for pain,” Diana M. Martinez, MD, a professor of psychiatry at Columbia University, New York, who specializes in addiction research, said in a recent MDedge podcast. “THC could be added to the armamentarium of pain medications that we use today.”
In a 2015 systematic literature review, researchers assessed 28 randomized, controlled trials (RCTs) of the use of cannabinoids for chronic pain. They reported that a variety of formulations resulted in at least a 30% reduction in the odds of pain, compared with placebo. A meta-analysis of five RCTs involving patients with neuropathic pain found a 30% reduction in pain over placebo with inhaled, vaporized cannabis. Varying results have been reported in additional studies for this indication. The National Academies of Sciences, Engineering, and Medicine concluded that there was a substantial body of evidence that cannabis is an effective treatment for chronic pain in adults.
The ongoing opioid epidemic has lent these results additional relevance.
Seeing this firsthand has caused Mark Steven Wallace, MD, a pain management specialist and chair of the division of pain medicine at the University of California San Diego Health, to reconsider offering cannabis to his patients.
“I think it’s probably more efficacious, just from my personal experience, and it’s a much lower risk of abuse and dependence than the opioids,” he said.
Dr. Wallace advised that clinicians who treat pain consider the ratios of cannabinoids.
“This is anecdotal, but we do find that with the combination of the two, CBD reduces the psychoactive effects of the THC. The ratios we use during the daytime range around 20 mg of CBD to 1 mg of THC,” he said.
In a recent secondary analysis of an RCT involving patients with painful diabetic peripheral neuropathy, Dr. Wallace and colleagues showed that THC’s effects appear to reverse themselves at a certain level.
“As the THC level goes up, the pain reduces until you reach about 16 ng/mL; then it starts going in the opposite direction, and pain will start to increase,” he said. “Even recreational cannabis users have reported that they avoid high doses because it’s very aversive. Using cannabis is all about, start low and go slow.”
A mixed bag for neurologic indications
There are relatively limited data on the use of medical cannabis for other neurologic conditions, and results have varied. For uses other than pain management, the evidence that does exist is strongest regarding epilepsy, said Daniel Freedman, DO, assistant professor of neurology at the University of Texas at Austin. He noted “multiple high-quality RCTs showing that pharmaceutical-grade CBD can reduce seizures associated with two particular epilepsy syndromes: Dravet Syndrome and Lennox Gastaut.”
These findings led to the FDA’s 2018 approval of Epidiolex for these syndromes. In earlier years, interest in CBD for pediatric seizures was largely driven by anecdotal parental reports of its benefits. NASEM’s 2017 overview on medical cannabis found evidence from subsequent RCTs in this indication to be insufficient. Clinicians who prescribe CBD for this indication must be vigilant because it can interact with several commonly used antiepileptic drugs.
Cannabinoid treatments have also shown success in alleviating muscle spasticity resulting from multiple sclerosis, most prominently in the form of nabiximols (Sativex), a standardized oralmucosal spray containing approximately equal quantities of THC and CBD. Nabiximols is approved in Europe but not in the United States. Moderate evidence supports the efficacy of these and other treatments over placebo in reducing muscle spasticity. Patient ratings of its effects tend to be higher than clinician assessment.
Parkinson’s disease has not yet been approved as an indication for treatment with cannabis or cannabinoids, yet a growing body of preclinical data suggests these could influence the dopaminergic system, said Carsten Buhmann, MD, from the department of neurology at the University Medical Center Hamburg-Eppendorf (Germany).
“In general, cannabinoids modulate basal-ganglia function on two levels which are especially relevant in Parkinson’s disease, i.e., the glutamatergic/dopaminergic synaptic neurotransmission and the corticostriatal plasticity,” he said. “Furthermore, activation of the endocannabinoid system might induce neuroprotective effects related to direct receptor-independent mechanisms, activation of anti-inflammatory cascades in glial cells via the cannabinoid receptor type 2, and antiglutamatergic antiexcitotoxic properties.”
Dr. Buhmann said that currently, clinical evidence is scarce, consisting of only four double-blind, placebo-controlled RCTs involving 49 patients. Various cannabinoids and methods of administering treatment were employed. Improvement was only observed in one of these RCTs, which found that the cannabinoid receptor agonist nabilone significantly reduced levodopa-induced dyskinesia for patients with Parkinson’s disease. Subjective data support a beneficial effect. In a nationwide survey of 1,348 respondents conducted by Dr. Buhmann and colleagues, the majority of medical cannabis users reported that it improved their symptoms (54% with oral CBD and 68% with inhaled THC-containing cannabis).
NASEM concluded that there was insufficient evidence to support the efficacy of medical cannabis for other neurologic conditions, including Tourette syndrome, amyotrophic lateral sclerosis, Huntington disease, dystonia, or dementia. A 2020 position statement from the American Academy of Neurology cited the lack of sufficient peer-reviewed research as the reason it could not currently support the use of cannabis for neurologic disorders.
Yet, according to Dr. Freedman, who served as a coauthor of the AAN position statement, this hasn’t stymied research interest in the topic. He’s seen a substantial uptick in studies of CBD over the past 2 years. “The body of evidence grows, but I still see many claims being made without evidence. And no one seems to care about all the negative trials.”
Cannabis as a treatment for, and cause of, psychiatric disorders
Mental health problems – such as anxiety, depression, and PTSD – are among the most common reasons patients seek out medical cannabis. There is an understandable interest in using cannabis and cannabinoids to treat psychiatric disorders. Preclinical studies suggest that the endocannabinoid system plays a prominent role in modulating feelings of anxiety, mood, and fear. As with opioids and chronic pain management, there is hope that medical cannabis may provide a means of reducing prescription anxiolytics and their associated risks.
The authors of the first systematic review (BMC Psychiatry. 2020 Jan 16;20[1]:24) of the use of medical cannabis for major psychiatric disorders noted that the current evidence was “encouraging, albeit embryonic.”
Meta-analyses have indicated a small but positive association between cannabis use and anxiety, although this may reflect the fact that patients with anxiety sought out this treatment. Given the risks for substance use disorders among patients with anxiety, CBD may present a more viable option. Positive results have been shown as treatment for generalized social anxiety disorder.
Limited but encouraging results have also been reported regarding the alleviation of PTSD symptoms with both cannabis and CBD, although the body of high-quality evidence hasn’t notably progressed since 2017, when NASEM declared that the evidence was insufficient. Supportive evidence is similarly lacking regarding the treatment of depression. Longitudinal studies suggest that cannabis use, particularly heavy use, may increase the risk of developing this disorder. Because THC is psychoactive, it is advised that it be avoided by patients at risk for psychotic disorders. However, CBD has yielded limited benefits for patients with treatment-resistant schizophrenia and for young people at risk for psychosis.
The use of medical cannabis for psychiatric conditions requires a complex balancing act, inasmuch as these treatments may exacerbate the very problems they are intended to alleviate.
Marta Di Forti, MD, PhD, professor of psychiatric research at Kings College London, has been at the forefront of determining the mental health risks of continued cannabis use. In 2019, Dr. Di Forti developed the first and only Cannabis Clinic for Patients With Psychosis in London where she and her colleagues have continued to elucidate this connection.
Dr. Di Forti and colleagues have linked daily cannabis use to an increase in the risk of experiencing psychotic disorder, compared with never using it. That risk was further increased among users of high-potency cannabis (≥10% THC). The latter finding has troubling implications, because concentrations of THC have steadily risen since 1970. By contrast, CBD concentrations have remained generally stable. High-potency cannabis products are common in both recreational and medicinal settings.
“For somebody prescribing medicinal cannabis that has a ≥10% concentration of THC, I’d be particularly wary of the risk of psychosis,” said Dr. Di Forti. “If you’re expecting people to use a high content of THC daily to medicate pain or a chronic condition, you even more so need to be aware that this is a potential side effect.”
Dr. Di Forti noted that her findings come from a cohort of recreational users, most of whom were aged 18-35 years.
“There have actually not been studies developed from collecting data in this area from groups specifically using cannabis for medicinal rather than recreational purposes,” she said.
She added that she personally has no concerns about the use of medical cannabis but wants clinicians to be aware of the risk for psychosis, to structure their patient conversations to identify risk factors or family histories of psychosis, and to become knowledgeable in detecting the often subtle signs of its initial onset.
When cannabis-associated psychosis occurs, Dr. Di Forti said it is primarily treated with conventional means, such as antipsychotics and therapeutic interventions and by refraining from using cannabis. Achieving the latter goal can be a challenge for patients who are daily users of high-potency cannabis. Currently, there are no treatment options such as those offered to patients withdrawing from the use of alcohol or opioids. Dr. Di Forti and colleagues are currently researching a solution to that problem through the use of another medical cannabis, the oromucosal spray Sativex, which has been approved in the European Union.
The regulatory obstacles to clarifying cannabis’ role in medicine
That currently there is limited or no evidence to support the use of medical cannabis for the treatment of neuropsychiatric conditions points to the inherent difficulties in conducting high-level research in this area.
“There’s a tremendous shortage of reliable data, largely due to regulatory barriers,” said Dr. Martinez.
Since 1970, cannabis has been listed as a Schedule I drug that is illegal to prescribe (the Agriculture Improvement Act of 2018 removed hemp from such restrictions). The FDA has issued guidance for researchers who wish to investigate treatments using Cannabis sativa or its derivatives in which the THC content is greater than 0.3%. Such research requires regular interactions with several federal agencies, including the Drug Enforcement Administration.
“It’s impossible to do multicenter RCTs with large numbers of patients, because you can’t transport cannabis across state lines,” said Dr. Wallace.
Regulatory restrictions regarding medical cannabis vary considerably throughout the world (the European Monitoring Center for Drugs and Drug Addiction provides a useful breakdown of this on their website). The lack of consistency in regulatory oversight acts as an impediment for conducting large-scale international multicenter studies on the topic.
Dr. Buhmann noted that, in Germany, cannabis has been broadly approved for treatment-resistant conditions with severe symptoms that impair quality of life. In addition, it is easy to be reimbursed for the use of cannabis as a medical treatment. These factors serve as disincentives for the funding of high-quality studies.
“It’s likely that no pharmaceutical company will do an expensive RCT to get an approval for Parkinson’s disease because it is already possible to prescribe medical cannabis of any type of THC-containing cannabinoid, dose, or route of application,” Dr. Buhmann said.
In the face of such restrictions and barriers, researchers are turning to ambitious real-world data projects to better understand medical cannabis’ efficacy and safety. A notable example is ProjectTwenty21, which is supported by the Royal College of Psychiatrists. The project is collecting outcomes of the use of medical cannabis among 20,000 U.K. patients whose conventional treatments of chronic pain, anxiety disorder, epilepsy, multiple sclerosis, PTSD, substance use disorder, and Tourette syndrome failed.
Dr. Freedman noted that the continued lack of high-quality data creates a void that commercial interests fill with unfounded claims.
“The danger is that patients might abandon a medication or intervention backed by robust science in favor of something without any science or evidence behind it,” he said. “There is no reason not to expect the same level of data for claims about cannabis products as we would expect from pharmaceutical products.”
Getting to that point, however, will require that the authorities governing clinical trials begin to view cannabis as the research community does, as a possible treatment with potential value, rather than as an illicit drug that needs to be tamped down.
A version of this article first appeared on Medscape.com.
Measuring cotinine to monitor tobacco use and smoking cessation
Cigarette smoking is common among patients with schizophrenia, mood disorders, anxiety disorders,1-3 substance use disorders (SUDs),4 and other psychiatric disorders. Research suggests that compared with the general population, patients with SUDs consume more nicotine products and are more vulnerable to the effects of smoking.5 Despite the availability of effective treatments, many mental health professionals are reluctant to identify and treat tobacco use disorder,6-8 or they prioritize other disorders over tobacco use. Early detection and treatment of tobacco use disorder can improve patients’ health and reduce the incidence of acute and chronic illness.
Cotinine is a biomarker that can be used to detect tobacco use. It can be measured in routine clinical practice by collecting urinary, serum, or salivary specimens, and used to monitor psychiatric patients’ tobacco use. Monitoring cotinine levels is similar to using other biomarkers to assess medication adherence or identify illicit substance use. A growing body of evidence supports the utility of cotinine screening as a part of a comprehensive substance use disorder treatment plan,5,9,10 especially for:
- patients who have comorbid conditions that can be exacerbated by tobacco use, such as chronic obstructive pulmonary disease
- patients who are pregnant11,12
- patients who are less reliable in self-report or who require objective testing for validation.
Routine clinical screening of tobacco use is recommended for all patients and early detection may facilitate earlier treatment. Several FDA-approved medications are available for smoking cessation13; however, discussion of treatment options is beyond the scope of this review. In this article, we describe how cotinine is measured and analyzed, 3 case vignettes that illustrate its potential clinical utility, and limitations to its use as a biomarker of tobacco use.
Methods of measuring cotinine
Cigarette smoking is associated with the absorption of nicotine, which is mainly metabolized by cytochrome P450 (CYP) 2A6 to 6 primary metabolites: cotinine, hydroxycotinine, norcotinine, nornicotine, cotinine oxide, and nicotine oxide.14,15 Cotinine is the biomarker of choice for detecting use of tobacco/nicotine products due to its stability (it is not influenced by dietary or environmental factors), extended half-life (16 to 19 hours, compared with 2 hours for nicotine), and stable concentration throughout the day. Samples from saliva, urine, or blood can be analyzed through radioimmunoassay, enzyme-linked immunosorbent assay (ELISA), and gas/liquid chromatography.16 The specificity of cotinine for tobacco use is excellent, except for persons who are taking medications that contain nicotine.17
An advantage of cotinine over other biomarkers for smoking (such as carbon monoxide in expired air) is that the optimal cut-off points for cotinine are relatively uninfluenced by the prevalence of smoking in the population. The optimal cut-off levels used to detect current tobacco use may vary based on the sample or test used (saliva, urine, or plasma) and certain patient-specific factors (Box 111,16,18-21). However, for plasma or saliva cotinine, 16 ng/mL is the generally accepted cut-off level for detecting current tobacco use. A urinary cotinine cut-off level of 50 ng/mL is likely appropriate for most circumstances.17 Users of electronic nicotine delivery systems (electronic cigarettes) have been found to have cotinine levels similar to those of cigarette smokers.22
Box 1
Daily smokers typically have a serum/plasma cotinine concentration of ≥100 ng/mL. Individuals with heavy exposure to secondhand smoking may have plasma cotinine concentrations up to 25 ng/mL, and urine samples tend to be much more specific.16 However, serum cotinine has a wide cut-off range due to diverse racial/ethnic, gender, and pregnancy-related variations; the wide range is also associated with genetic polymorphisms of cytochrome P450 2A6 alleles and nicotine’s numerous metabolic pathways.11,18
Traditionally a serum/plasma cut-off point of approximately 15 ng/mL has been accepted to detect current tobacco use; however, recent studies21 recommend an average optimal cut-off point for US adults of 3 ng/mL. This possibly reflects differences in national cigarette smoking patterns and exposure.21 One study suggested optimal cut-off differences for men (1.78 ng/mL) and women (4.47 ng/mL).19 The same study also suggested different optimal cut-off levels for non-Hispanic White men (6.79 ng/ mL), non-Hispanic Black men (13.3 ng/mL), and Mexican-American men (0.79 ng/mL).19 These researchers also suggested different optimal cut-off levels for non-Hispanic White women (4.73 ng/mL), non-Hispanic Black women (5.91 ng/mL), and Mexican-American women (0.84 ng/mL).19 Genetic factors may also play a role in the progression of nicotine dependence and pose challenges that impact smoking persistence.20
Assessment of cotinine levels in saliva may be considered for outpatient monitoring due to its noninvasive nature, tolerability, and the ability to collect multiple samples over a limited period.23 Saliva cotinine levels correlate closely with blood concentrations. Urine cotinine levels offer some advantage because concentrations are 6 times higher in urine than in blood or saliva. For this reason, urine cotinine is the most widely used biomarker in individuals who use tobacco due to its high sensitivity, specificity, reliability, and noninvasive collection.23 By using a lower urinary cut-off of ≥2.47 ng/mL, ELISA kits detect the highest sensitivity and specificity, which is useful for monitoring daily tobacco use.24 This cut-off value was associated with 100% sensitivity and specificity, and these numbers declined with increases in the cut-off threshold.23
The following 3 clinical vignettes illustrate the impact of tobacco use disorder on patients, and how cotinine might help with their treatment.
Continue to: Vignette 1
Vignette 1
Mr. D, age 44, has a history of schizophrenia and has smoked 1 pack of cigarettes per day for the last 15 years. He was recently discharged from an inpatient psychiatric facility after his symptoms were stabilized. During his hospitalization, Mr. D used a nicotine-replacement product to comply with the hospital’s smoke-free policy. Unfortunately, since discharge, Mr. D reports worsening auditory hallucinations despite adherence with his antipsychotic medication, clozapine, 600 mg at bedtime. Collateral information gathered from Mr. D’s mother confirms that he has been adherent with the discharge medication regimen; however, Mr. D has resumed smoking 1 pack of cigarettes daily. The treatment team suspects that his worsening psychosis is related to the decrease of blood clozapine level due to CYP induction by cigarette smoke.
Cotinine and smoking-related drug interactions
Vignette 1 illustrates the significant impact tobacco smoke can have on the effectiveness of a psychotropic medication. This is caused by polycyclic aromatic hydrocarbons induction of hepatic CYP1A2 isoenzymes. Clinicians should routinely screen patients for smoking status due to the potential for drug interactions. Common major CYP1A2 substrates include
Vignette 2
Mr. B, age 34, has a history of cocaine use disorder and tobacco use disorder. He is referred to a treatment program and participates in a contingency management program for his substance use disorders. Biomarkers, including salivary cotinine, are used to assess Mr. B’s exposure to tobacco use. Mr. B and other participants in his program are eligible for prize draws if they are found to have samples that are negative for tobacco and other substances. There are other incentives in place for patients who show a reduced cotinine concentration.
Cotinine monitoring and contingency management
Clinicians can incorporate cotinine monitoring into existing SUD treatment. This is similar to the utilization of other biomarkers that are commonly used to identify recent illicit substance use or monitor adherence to treatment medications. For example, benzoylecgonine, a metabolite of cocaine, is frequently used to monitor abstinence from cocaine.
Treatments based on contingency management principles involve giving patients tangible rewards to reinforce desired (positive) behaviors. Smoking cessation can be confirmed by monitoring cotinine levels. Gayman et al9 found twice-weekly salivary testing was compatible with monitoring and promoting abstinence in a prize-based contingency management smoking cessation program. Most prior studies used urine cotinine measures to verify abstinence. Although highly reliable, urine samples require close monitoring to ensure sample validity, which can be a burden on staff and unpleasant for patients.9 It is also important to note that the rate of elimination of cotinine from saliva and urine are comparable. The half-life of cotinine is approximately 18 hours, and therefore the specificity of salivary test strips may be impacted during the first 4 to 5 days of abstinence. In the first few days of smoking cessation, a more intensive approach, such as quantifying urine cotinine levels and monitoring decline, may be appropriate.23
Continue to: Vignette 3
Vignette 3
Ms. C, age 34 and pregnant, is admitted to an outpatient treatment program for alcohol use disorder. She also has generalized anxiety disorder and tobacco use disorder. In addition to attending group therapy sessions and self-reporting any recent alcohol consumption, Ms. C also undergoes alcohol breathalyzer tests and urine studies of alcohol metabolites to monitor abstinence from alcohol. She says that the regular laboratory screening for alcohol use gives her a sense of accountability and tangible evidence of change that positively impacts her treatment. When the treating psychiatrist recommends that Ms. C also consider addressing her tobacco use disorder, she asks if there is some way to include laboratory testing to monitor her smoking cessation.
Cotinine as a predictor of smoking status
Smoking abstinence rates during pregnancy are lower than that for other substances, and pregnant women may not be aware of the impact of smoking on fetal development.30 Cotinine can be used to verify self-report of smoking status and severity.10,31,32
Salivary cotinine tests are commercially available, relatively economical, and convenient to use when frequent monitoring is required.32 In general, based on established cut-off values that are unique to the specimen collected, the overall high specificity and sensitivity of salivary testing allows clinicians to predict smoker vs nonsmoker status with confidence. For example, a 2008 study reported a salivary cotinine cut-off level of 12 ng/mL for smokers.21 The sensitivity and specificity of this cut-off value for distinguishing cigarette smokers from never smokers were 96.7% and 96.9%, respectively.21
Additionally, some studies suggest that cotinine levels may be predictive of treatment outcomes and retention in SUD treatment programs.33,34 One study of smoking cessation using nicotine replacement products found that compared with patients with lower baseline cotinine levels prior to treatment, patients with higher baseline cotinine plasma levels had lower smoking cessation success rates.34
A few caveats
There are several limitations to quantitative measures of cotinine (Box 221,23). These include (but are not limited to) potential errors related to sample collection, storage, shipping, and analysis.23 Compared with other methods, point-of-care cotinine measurement in saliva is noninvasive, simple, and requires less training to properly use.23
Box 2
Challenges in the collection of samples, storage, shipping, and instrumentation may limit cotinine consistency as a dependable biomarker in the clinical setting.23 Overall, quantitative measurements of cotinine have relative constructive utility in separating smokers from nonsmokers, because daily smokers typically have serum concentrations of 100 ng/mL or higher, in contrast to light/non-daily smokers, who have cotinine concentrations <10 ng/mL. Even heavy exposure to secondhand smoke typically yields plasma concentrations up to approximately 25 ng/mL. However, cotinine is a general metabolite found with the use of all nicotine products, which makes it extremely difficult to differentiate tobacco use from the use of nicotine replacement products, which are frequently used to treat tobacco use disorders.
One potential solution is to measure nicotine-derived nitrosamine ketone (NNK) and its metabolite 4-(methylnitrosamino)- 1-(3-pyridyl)-1-butanol (NNAL). Both NNK and NNAL are tobacco-specific lung carcinogens. NNAL can be measured in the urine. Although total NNAL represents only 15% of NNK dose intake, it has been quantified, with urine concentrations of ≥1,000 fmol/mL for daily smokers. NNAL also has an extremely high specificity to tobacco smoke, and thus allows differentiation of tobacco use from nicotine replacement treatment. Unfortunately, measurement for this biomarker requires specific chemical expertise and expensive equipment.
Another potential barrier to using cotinine in the clinical setting is the variable cut-off levels used in the United States, based on differences in race/ethnicity. This may be secondary to differences in smoking behaviors and/or differences in cotinine metabolism.21
Continue to: Confirmation of smoking cessation...
Confirmation of smoking cessation can be monitored reliably within the clinical setting using cotinine monitoring. However, this is not a routine test, and there are no guidelines or consensus on how or when it should be used. The clinical feasibility of cotinine monitoring for psychiatric patients will depend on the cost of testing, methods used, amount of reimbursement for performing the tests, and how clinicians value such testing.35
Bottom Line
Cotinine is a biomarker that can be used to detect tobacco use. Cotinine measurement can be used to monitor tobacco use and smoking cessation in psychiatric patients. Early detection and treatment of tobacco use disorder can improve patients’ health and reduce the incidence of acute and chronic illnesses. However, cotinine measurement is not a routine test, and there are no guidelines on how or when this test should be used.
Related Resources
- Peckham E, Brabyn S, Cook L, et al. Smoking cessation in severe mental ill health: what works? An updated systematic review and meta-analysis. BMC Psychiatry. 2017;17(1):252.
- Tidey JW, Miller ME. Smoking cessation and reduction in people with chronic mental illness. BMJ. 2015;351:h4065. doi: 10.1136/bmj.h4065
Drug Brand Names
Asenapine • Saphris
Buprenorphine • Sublocade
Clozapine • Clozaril
Duloxetine • Cymbalta
Haloperidol • Haldol
Mirtazapine • Remeron
Olanzapine • Zyprexa
Ziprasidone • Geodon
Zolpidem • Ambien
1. Prochaska JJ, Das S, Young-Wolff KC. Smoking, mental illness, and public health. Annu Rev Public Health. 2017;38:165-185.
2. Pal A, Balhara YP. A review of impact of tobacco use on patients with co-occurring psychiatric disorders. Tob Use Insights. 2016;9:7-12.
3. Lawrence D, Mitrou F, Zubrick SR. Smoking and mental illness: results from population surveys in Australia and the United States. BMC Public Health. 2009;9:285.
4. Kalman D, Morissette SB, George TP. Co-morbidity of smoking in patients with psychiatric and substance use disorders. Am J Addict. 2005;14(2):106-123.
5. Baca CT, Yahne CE. Smoking cessation during substance abuse treatment: what you need to know. J Subst Abuse Treat. 2009;36(2):205-219.
6. Hall SM, Tsoh JY, Prochaska JJ, et al. Treatment for cigarette smoking among depressed mental health outpatients: a randomized clinical trial. Am J Public Health. 2006;96(10):1808-1814.
7. McHugh RK, Votaw VR, Fulciniti F, et al. Perceived barriers to smoking cessation among adults with substance use disorders. J Subst Abuse Treat. 2017;74:48-53.
8. Strong DR, Uebelacker L, Fokas K, et al. Utilization of evidence-based smoking cessation treatments by psychiatric inpatient smokers with depression. J Addict Med. 2014;8(2):77-83.
9. Gayman C, Anderson K, Pietras C. Saliva cotinine as a measure of smoking abstinence in contingency management – a feasibility study. The Psychological Record. 2017;67(2):261-272.
10. Schepis TS, Duhig AM, Liss T, et al. Contingency management for smoking cessation: enhancing feasibility through use of immunoassay test strips measuring cotinine. Nicotine Tob Res. 2008;10(9):1495-1501.
11. Stragierowicz J, Mikolajewska K, Zawadzka-Stolarz M, et al. Estimation of cutoff values of cotinine in urine and saliva for pregnant women in Poland. Biomed Res Int. 2013;2013:386784. doi.org/10.1155/2013/386784
12. Shipton D, Tappin DM, Vadiveloo T, et al. Reliability of self reported smoking status by pregnant women for estimating smoking prevalence: a retrospective, cross sectional study. BMJ. 2009;339:b4347. doi.org/10.1136/bmj.b4347
13. Aubin HJ, Karila L, Reynaud M. Pharmacotherapy for smoking cessation: present and future. Curr Pharm Des. 2011;17(14):1343-1350.
14. McGuffey JE, Wei B, Bernert JT, et al. Validation of a LC-MS/MS method for quantifying urinary nicotine, six nicotine metabolites and the minor tobacco alkaloids--anatabine and anabasine--in smokers’ urine. PLoS One. 2014;9(7):e101816. doi: 10.1371/journal.pone.0101816
15. Duque A, Martinez PJ, Giraldo A, et al. Accuracy of cotinine serum test to detect the smoking habit and its association with periodontal disease in a multicenter study. Med Oral Patol Oral Cir Bucal. 2017;22(4):e425-e431. doi: 10.4317/medoral.21292
16. Avila-Tang E, Elf JL, Cummings KM, et al. Assessing secondhand smoke exposure with reported measures. Tob Control. 2013;22(3):156-163.
17. Benowitz NL, Bernert JT, Foulds J, et al. Biochemical verification of tobacco use and abstinence: 2019 Update. Nicotine Tob Res. 2020;22(7):1086-1097.
18. Nakajima M TY. Interindividual variability in nicotine metabolism: c-oxidation and glucuronidation. Drug Metab Pharmaokinet. 2005;20(4):227-235.
19. Benowitz NL, Bernert JT, Caraballo RS, et al. Optimal serum cotinine levels for distinguishing cigarette smokers and nonsmokers within different racial/ethnic groups in the United States between 1999 and 2004. Am J Epidemiol. 2009;169(2):236-248.
20. Schnoll R, Johnson TA, Lerman C. Genetics and smoking behavior. Curr Psychiatry Rep. 2007;9(5):349-357.
21. Kim S. Overview of cotinine cutoff values for smoking status classification. Int J Environ Res Public Health. 2016;13(12):1236.
22. Etter JF, Bullen C. Saliva cotinine levels in users of electronic cigarettes. Eur Respir J. 2011;38(5):1219-1220.
23. Raja M, Garg A, Yadav P, et al. Diagnostic methods for detection of cotinine level in tobacco users: a review. J Clin Diagn Res. 2016;10(3):ZE04-06. doi: 10.7860/JCDR/2016/17360.7423
24. Balhara YP, Jain R. A receiver operated curve-based evaluation of change in sensitivity and specificity of cotinine urinalysis for detecting active tobacco use. J Cancer Res Ther. 2013;9(1):84-89.
25. Fankhauser M. Drug interactions with tobacco smoke: implications for patient care. Current Psychiatry. 2013;12(1):12-16.
26. Scheuermann TS, Richter KP, Rigotti NA, et al. Accuracy of self-reported smoking abstinence in clinical trials of hospital-initiated smoking interventions. Addiction. 2017;112(12):2227-2236.
27. Holtyn AF, Knealing TW, Jarvis BP, et al. Monitoring cocaine use and abstinence among cocaine users for contingency management interventions. Psychol Rec. 2017;67(2):253-259.
28. Donroe JH, Holt SR, O’Connor PG, et al. Interpreting quantitative urine buprenorphine and norbuprenorphine levels in office-based clinical practice. Drug Alcohol Depend. 2017;180:46-51.
29. Sullivan M, Covey, LS. Current perspectives on smoking cessation among substance abusers. Curr Psychiatry Rep. 2002;4(5):388-396.
30. Forray A, Merry B, Lin H, et al. Perinatal substance use: a prospective evaluation of abstinence and relapse. Drug Alcohol Depend. 2015;150:147-155.
31. Parker DR, Lasater TM, Windsor R, et al. The accuracy of self-reported smoking status assessed by cotinine test strips. Nicotine Tob Res. 2002;4(3):305-309.
32. Asha V, Dhanya M. Immunochromatographic assessment of salivary cotinine and its correlation with nicotine dependence in tobacco chewers. J Cancer Prev. 2015;20(2):159-163.
33. Hall S, Herning RI, Jones RT, et al. Blood cotinine levels as indicators of smoking treatment outcome. Clin Pharmacol Ther. 1984;35(6):810-814.
34. Paoletti P, Fornai E, Maggiorelli F, et al. Importance of baseline cotinine plasma values in smoking cessation: results from a double-blind study with nicotine patch. Eur Respir J. 1996;9(4):643-651.
35. Montalto NJ, Wells WO. Validation of self-reported smoking status using saliva cotinine: a rapid semiquantitative dipstick method. Cancer Epidemiol Biomarkers Prev. 2007;16(9):1858-1862.
Cigarette smoking is common among patients with schizophrenia, mood disorders, anxiety disorders,1-3 substance use disorders (SUDs),4 and other psychiatric disorders. Research suggests that compared with the general population, patients with SUDs consume more nicotine products and are more vulnerable to the effects of smoking.5 Despite the availability of effective treatments, many mental health professionals are reluctant to identify and treat tobacco use disorder,6-8 or they prioritize other disorders over tobacco use. Early detection and treatment of tobacco use disorder can improve patients’ health and reduce the incidence of acute and chronic illness.
Cotinine is a biomarker that can be used to detect tobacco use. It can be measured in routine clinical practice by collecting urinary, serum, or salivary specimens, and used to monitor psychiatric patients’ tobacco use. Monitoring cotinine levels is similar to using other biomarkers to assess medication adherence or identify illicit substance use. A growing body of evidence supports the utility of cotinine screening as a part of a comprehensive substance use disorder treatment plan,5,9,10 especially for:
- patients who have comorbid conditions that can be exacerbated by tobacco use, such as chronic obstructive pulmonary disease
- patients who are pregnant11,12
- patients who are less reliable in self-report or who require objective testing for validation.
Routine clinical screening of tobacco use is recommended for all patients and early detection may facilitate earlier treatment. Several FDA-approved medications are available for smoking cessation13; however, discussion of treatment options is beyond the scope of this review. In this article, we describe how cotinine is measured and analyzed, 3 case vignettes that illustrate its potential clinical utility, and limitations to its use as a biomarker of tobacco use.
Methods of measuring cotinine
Cigarette smoking is associated with the absorption of nicotine, which is mainly metabolized by cytochrome P450 (CYP) 2A6 to 6 primary metabolites: cotinine, hydroxycotinine, norcotinine, nornicotine, cotinine oxide, and nicotine oxide.14,15 Cotinine is the biomarker of choice for detecting use of tobacco/nicotine products due to its stability (it is not influenced by dietary or environmental factors), extended half-life (16 to 19 hours, compared with 2 hours for nicotine), and stable concentration throughout the day. Samples from saliva, urine, or blood can be analyzed through radioimmunoassay, enzyme-linked immunosorbent assay (ELISA), and gas/liquid chromatography.16 The specificity of cotinine for tobacco use is excellent, except for persons who are taking medications that contain nicotine.17
An advantage of cotinine over other biomarkers for smoking (such as carbon monoxide in expired air) is that the optimal cut-off points for cotinine are relatively uninfluenced by the prevalence of smoking in the population. The optimal cut-off levels used to detect current tobacco use may vary based on the sample or test used (saliva, urine, or plasma) and certain patient-specific factors (Box 111,16,18-21). However, for plasma or saliva cotinine, 16 ng/mL is the generally accepted cut-off level for detecting current tobacco use. A urinary cotinine cut-off level of 50 ng/mL is likely appropriate for most circumstances.17 Users of electronic nicotine delivery systems (electronic cigarettes) have been found to have cotinine levels similar to those of cigarette smokers.22
Box 1
Daily smokers typically have a serum/plasma cotinine concentration of ≥100 ng/mL. Individuals with heavy exposure to secondhand smoking may have plasma cotinine concentrations up to 25 ng/mL, and urine samples tend to be much more specific.16 However, serum cotinine has a wide cut-off range due to diverse racial/ethnic, gender, and pregnancy-related variations; the wide range is also associated with genetic polymorphisms of cytochrome P450 2A6 alleles and nicotine’s numerous metabolic pathways.11,18
Traditionally a serum/plasma cut-off point of approximately 15 ng/mL has been accepted to detect current tobacco use; however, recent studies21 recommend an average optimal cut-off point for US adults of 3 ng/mL. This possibly reflects differences in national cigarette smoking patterns and exposure.21 One study suggested optimal cut-off differences for men (1.78 ng/mL) and women (4.47 ng/mL).19 The same study also suggested different optimal cut-off levels for non-Hispanic White men (6.79 ng/ mL), non-Hispanic Black men (13.3 ng/mL), and Mexican-American men (0.79 ng/mL).19 These researchers also suggested different optimal cut-off levels for non-Hispanic White women (4.73 ng/mL), non-Hispanic Black women (5.91 ng/mL), and Mexican-American women (0.84 ng/mL).19 Genetic factors may also play a role in the progression of nicotine dependence and pose challenges that impact smoking persistence.20
Assessment of cotinine levels in saliva may be considered for outpatient monitoring due to its noninvasive nature, tolerability, and the ability to collect multiple samples over a limited period.23 Saliva cotinine levels correlate closely with blood concentrations. Urine cotinine levels offer some advantage because concentrations are 6 times higher in urine than in blood or saliva. For this reason, urine cotinine is the most widely used biomarker in individuals who use tobacco due to its high sensitivity, specificity, reliability, and noninvasive collection.23 By using a lower urinary cut-off of ≥2.47 ng/mL, ELISA kits detect the highest sensitivity and specificity, which is useful for monitoring daily tobacco use.24 This cut-off value was associated with 100% sensitivity and specificity, and these numbers declined with increases in the cut-off threshold.23
The following 3 clinical vignettes illustrate the impact of tobacco use disorder on patients, and how cotinine might help with their treatment.
Continue to: Vignette 1
Vignette 1
Mr. D, age 44, has a history of schizophrenia and has smoked 1 pack of cigarettes per day for the last 15 years. He was recently discharged from an inpatient psychiatric facility after his symptoms were stabilized. During his hospitalization, Mr. D used a nicotine-replacement product to comply with the hospital’s smoke-free policy. Unfortunately, since discharge, Mr. D reports worsening auditory hallucinations despite adherence with his antipsychotic medication, clozapine, 600 mg at bedtime. Collateral information gathered from Mr. D’s mother confirms that he has been adherent with the discharge medication regimen; however, Mr. D has resumed smoking 1 pack of cigarettes daily. The treatment team suspects that his worsening psychosis is related to the decrease of blood clozapine level due to CYP induction by cigarette smoke.
Cotinine and smoking-related drug interactions
Vignette 1 illustrates the significant impact tobacco smoke can have on the effectiveness of a psychotropic medication. This is caused by polycyclic aromatic hydrocarbons induction of hepatic CYP1A2 isoenzymes. Clinicians should routinely screen patients for smoking status due to the potential for drug interactions. Common major CYP1A2 substrates include
Vignette 2
Mr. B, age 34, has a history of cocaine use disorder and tobacco use disorder. He is referred to a treatment program and participates in a contingency management program for his substance use disorders. Biomarkers, including salivary cotinine, are used to assess Mr. B’s exposure to tobacco use. Mr. B and other participants in his program are eligible for prize draws if they are found to have samples that are negative for tobacco and other substances. There are other incentives in place for patients who show a reduced cotinine concentration.
Cotinine monitoring and contingency management
Clinicians can incorporate cotinine monitoring into existing SUD treatment. This is similar to the utilization of other biomarkers that are commonly used to identify recent illicit substance use or monitor adherence to treatment medications. For example, benzoylecgonine, a metabolite of cocaine, is frequently used to monitor abstinence from cocaine.
Treatments based on contingency management principles involve giving patients tangible rewards to reinforce desired (positive) behaviors. Smoking cessation can be confirmed by monitoring cotinine levels. Gayman et al9 found twice-weekly salivary testing was compatible with monitoring and promoting abstinence in a prize-based contingency management smoking cessation program. Most prior studies used urine cotinine measures to verify abstinence. Although highly reliable, urine samples require close monitoring to ensure sample validity, which can be a burden on staff and unpleasant for patients.9 It is also important to note that the rate of elimination of cotinine from saliva and urine are comparable. The half-life of cotinine is approximately 18 hours, and therefore the specificity of salivary test strips may be impacted during the first 4 to 5 days of abstinence. In the first few days of smoking cessation, a more intensive approach, such as quantifying urine cotinine levels and monitoring decline, may be appropriate.23
Continue to: Vignette 3
Vignette 3
Ms. C, age 34 and pregnant, is admitted to an outpatient treatment program for alcohol use disorder. She also has generalized anxiety disorder and tobacco use disorder. In addition to attending group therapy sessions and self-reporting any recent alcohol consumption, Ms. C also undergoes alcohol breathalyzer tests and urine studies of alcohol metabolites to monitor abstinence from alcohol. She says that the regular laboratory screening for alcohol use gives her a sense of accountability and tangible evidence of change that positively impacts her treatment. When the treating psychiatrist recommends that Ms. C also consider addressing her tobacco use disorder, she asks if there is some way to include laboratory testing to monitor her smoking cessation.
Cotinine as a predictor of smoking status
Smoking abstinence rates during pregnancy are lower than that for other substances, and pregnant women may not be aware of the impact of smoking on fetal development.30 Cotinine can be used to verify self-report of smoking status and severity.10,31,32
Salivary cotinine tests are commercially available, relatively economical, and convenient to use when frequent monitoring is required.32 In general, based on established cut-off values that are unique to the specimen collected, the overall high specificity and sensitivity of salivary testing allows clinicians to predict smoker vs nonsmoker status with confidence. For example, a 2008 study reported a salivary cotinine cut-off level of 12 ng/mL for smokers.21 The sensitivity and specificity of this cut-off value for distinguishing cigarette smokers from never smokers were 96.7% and 96.9%, respectively.21
Additionally, some studies suggest that cotinine levels may be predictive of treatment outcomes and retention in SUD treatment programs.33,34 One study of smoking cessation using nicotine replacement products found that compared with patients with lower baseline cotinine levels prior to treatment, patients with higher baseline cotinine plasma levels had lower smoking cessation success rates.34
A few caveats
There are several limitations to quantitative measures of cotinine (Box 221,23). These include (but are not limited to) potential errors related to sample collection, storage, shipping, and analysis.23 Compared with other methods, point-of-care cotinine measurement in saliva is noninvasive, simple, and requires less training to properly use.23
Box 2
Challenges in the collection of samples, storage, shipping, and instrumentation may limit cotinine consistency as a dependable biomarker in the clinical setting.23 Overall, quantitative measurements of cotinine have relative constructive utility in separating smokers from nonsmokers, because daily smokers typically have serum concentrations of 100 ng/mL or higher, in contrast to light/non-daily smokers, who have cotinine concentrations <10 ng/mL. Even heavy exposure to secondhand smoke typically yields plasma concentrations up to approximately 25 ng/mL. However, cotinine is a general metabolite found with the use of all nicotine products, which makes it extremely difficult to differentiate tobacco use from the use of nicotine replacement products, which are frequently used to treat tobacco use disorders.
One potential solution is to measure nicotine-derived nitrosamine ketone (NNK) and its metabolite 4-(methylnitrosamino)- 1-(3-pyridyl)-1-butanol (NNAL). Both NNK and NNAL are tobacco-specific lung carcinogens. NNAL can be measured in the urine. Although total NNAL represents only 15% of NNK dose intake, it has been quantified, with urine concentrations of ≥1,000 fmol/mL for daily smokers. NNAL also has an extremely high specificity to tobacco smoke, and thus allows differentiation of tobacco use from nicotine replacement treatment. Unfortunately, measurement for this biomarker requires specific chemical expertise and expensive equipment.
Another potential barrier to using cotinine in the clinical setting is the variable cut-off levels used in the United States, based on differences in race/ethnicity. This may be secondary to differences in smoking behaviors and/or differences in cotinine metabolism.21
Continue to: Confirmation of smoking cessation...
Confirmation of smoking cessation can be monitored reliably within the clinical setting using cotinine monitoring. However, this is not a routine test, and there are no guidelines or consensus on how or when it should be used. The clinical feasibility of cotinine monitoring for psychiatric patients will depend on the cost of testing, methods used, amount of reimbursement for performing the tests, and how clinicians value such testing.35
Bottom Line
Cotinine is a biomarker that can be used to detect tobacco use. Cotinine measurement can be used to monitor tobacco use and smoking cessation in psychiatric patients. Early detection and treatment of tobacco use disorder can improve patients’ health and reduce the incidence of acute and chronic illnesses. However, cotinine measurement is not a routine test, and there are no guidelines on how or when this test should be used.
Related Resources
- Peckham E, Brabyn S, Cook L, et al. Smoking cessation in severe mental ill health: what works? An updated systematic review and meta-analysis. BMC Psychiatry. 2017;17(1):252.
- Tidey JW, Miller ME. Smoking cessation and reduction in people with chronic mental illness. BMJ. 2015;351:h4065. doi: 10.1136/bmj.h4065
Drug Brand Names
Asenapine • Saphris
Buprenorphine • Sublocade
Clozapine • Clozaril
Duloxetine • Cymbalta
Haloperidol • Haldol
Mirtazapine • Remeron
Olanzapine • Zyprexa
Ziprasidone • Geodon
Zolpidem • Ambien
Cigarette smoking is common among patients with schizophrenia, mood disorders, anxiety disorders,1-3 substance use disorders (SUDs),4 and other psychiatric disorders. Research suggests that compared with the general population, patients with SUDs consume more nicotine products and are more vulnerable to the effects of smoking.5 Despite the availability of effective treatments, many mental health professionals are reluctant to identify and treat tobacco use disorder,6-8 or they prioritize other disorders over tobacco use. Early detection and treatment of tobacco use disorder can improve patients’ health and reduce the incidence of acute and chronic illness.
Cotinine is a biomarker that can be used to detect tobacco use. It can be measured in routine clinical practice by collecting urinary, serum, or salivary specimens, and used to monitor psychiatric patients’ tobacco use. Monitoring cotinine levels is similar to using other biomarkers to assess medication adherence or identify illicit substance use. A growing body of evidence supports the utility of cotinine screening as a part of a comprehensive substance use disorder treatment plan,5,9,10 especially for:
- patients who have comorbid conditions that can be exacerbated by tobacco use, such as chronic obstructive pulmonary disease
- patients who are pregnant11,12
- patients who are less reliable in self-report or who require objective testing for validation.
Routine clinical screening of tobacco use is recommended for all patients and early detection may facilitate earlier treatment. Several FDA-approved medications are available for smoking cessation13; however, discussion of treatment options is beyond the scope of this review. In this article, we describe how cotinine is measured and analyzed, 3 case vignettes that illustrate its potential clinical utility, and limitations to its use as a biomarker of tobacco use.
Methods of measuring cotinine
Cigarette smoking is associated with the absorption of nicotine, which is mainly metabolized by cytochrome P450 (CYP) 2A6 to 6 primary metabolites: cotinine, hydroxycotinine, norcotinine, nornicotine, cotinine oxide, and nicotine oxide.14,15 Cotinine is the biomarker of choice for detecting use of tobacco/nicotine products due to its stability (it is not influenced by dietary or environmental factors), extended half-life (16 to 19 hours, compared with 2 hours for nicotine), and stable concentration throughout the day. Samples from saliva, urine, or blood can be analyzed through radioimmunoassay, enzyme-linked immunosorbent assay (ELISA), and gas/liquid chromatography.16 The specificity of cotinine for tobacco use is excellent, except for persons who are taking medications that contain nicotine.17
An advantage of cotinine over other biomarkers for smoking (such as carbon monoxide in expired air) is that the optimal cut-off points for cotinine are relatively uninfluenced by the prevalence of smoking in the population. The optimal cut-off levels used to detect current tobacco use may vary based on the sample or test used (saliva, urine, or plasma) and certain patient-specific factors (Box 111,16,18-21). However, for plasma or saliva cotinine, 16 ng/mL is the generally accepted cut-off level for detecting current tobacco use. A urinary cotinine cut-off level of 50 ng/mL is likely appropriate for most circumstances.17 Users of electronic nicotine delivery systems (electronic cigarettes) have been found to have cotinine levels similar to those of cigarette smokers.22
Box 1
Daily smokers typically have a serum/plasma cotinine concentration of ≥100 ng/mL. Individuals with heavy exposure to secondhand smoking may have plasma cotinine concentrations up to 25 ng/mL, and urine samples tend to be much more specific.16 However, serum cotinine has a wide cut-off range due to diverse racial/ethnic, gender, and pregnancy-related variations; the wide range is also associated with genetic polymorphisms of cytochrome P450 2A6 alleles and nicotine’s numerous metabolic pathways.11,18
Traditionally a serum/plasma cut-off point of approximately 15 ng/mL has been accepted to detect current tobacco use; however, recent studies21 recommend an average optimal cut-off point for US adults of 3 ng/mL. This possibly reflects differences in national cigarette smoking patterns and exposure.21 One study suggested optimal cut-off differences for men (1.78 ng/mL) and women (4.47 ng/mL).19 The same study also suggested different optimal cut-off levels for non-Hispanic White men (6.79 ng/ mL), non-Hispanic Black men (13.3 ng/mL), and Mexican-American men (0.79 ng/mL).19 These researchers also suggested different optimal cut-off levels for non-Hispanic White women (4.73 ng/mL), non-Hispanic Black women (5.91 ng/mL), and Mexican-American women (0.84 ng/mL).19 Genetic factors may also play a role in the progression of nicotine dependence and pose challenges that impact smoking persistence.20
Assessment of cotinine levels in saliva may be considered for outpatient monitoring due to its noninvasive nature, tolerability, and the ability to collect multiple samples over a limited period.23 Saliva cotinine levels correlate closely with blood concentrations. Urine cotinine levels offer some advantage because concentrations are 6 times higher in urine than in blood or saliva. For this reason, urine cotinine is the most widely used biomarker in individuals who use tobacco due to its high sensitivity, specificity, reliability, and noninvasive collection.23 By using a lower urinary cut-off of ≥2.47 ng/mL, ELISA kits detect the highest sensitivity and specificity, which is useful for monitoring daily tobacco use.24 This cut-off value was associated with 100% sensitivity and specificity, and these numbers declined with increases in the cut-off threshold.23
The following 3 clinical vignettes illustrate the impact of tobacco use disorder on patients, and how cotinine might help with their treatment.
Continue to: Vignette 1
Vignette 1
Mr. D, age 44, has a history of schizophrenia and has smoked 1 pack of cigarettes per day for the last 15 years. He was recently discharged from an inpatient psychiatric facility after his symptoms were stabilized. During his hospitalization, Mr. D used a nicotine-replacement product to comply with the hospital’s smoke-free policy. Unfortunately, since discharge, Mr. D reports worsening auditory hallucinations despite adherence with his antipsychotic medication, clozapine, 600 mg at bedtime. Collateral information gathered from Mr. D’s mother confirms that he has been adherent with the discharge medication regimen; however, Mr. D has resumed smoking 1 pack of cigarettes daily. The treatment team suspects that his worsening psychosis is related to the decrease of blood clozapine level due to CYP induction by cigarette smoke.
Cotinine and smoking-related drug interactions
Vignette 1 illustrates the significant impact tobacco smoke can have on the effectiveness of a psychotropic medication. This is caused by polycyclic aromatic hydrocarbons induction of hepatic CYP1A2 isoenzymes. Clinicians should routinely screen patients for smoking status due to the potential for drug interactions. Common major CYP1A2 substrates include
Vignette 2
Mr. B, age 34, has a history of cocaine use disorder and tobacco use disorder. He is referred to a treatment program and participates in a contingency management program for his substance use disorders. Biomarkers, including salivary cotinine, are used to assess Mr. B’s exposure to tobacco use. Mr. B and other participants in his program are eligible for prize draws if they are found to have samples that are negative for tobacco and other substances. There are other incentives in place for patients who show a reduced cotinine concentration.
Cotinine monitoring and contingency management
Clinicians can incorporate cotinine monitoring into existing SUD treatment. This is similar to the utilization of other biomarkers that are commonly used to identify recent illicit substance use or monitor adherence to treatment medications. For example, benzoylecgonine, a metabolite of cocaine, is frequently used to monitor abstinence from cocaine.
Treatments based on contingency management principles involve giving patients tangible rewards to reinforce desired (positive) behaviors. Smoking cessation can be confirmed by monitoring cotinine levels. Gayman et al9 found twice-weekly salivary testing was compatible with monitoring and promoting abstinence in a prize-based contingency management smoking cessation program. Most prior studies used urine cotinine measures to verify abstinence. Although highly reliable, urine samples require close monitoring to ensure sample validity, which can be a burden on staff and unpleasant for patients.9 It is also important to note that the rate of elimination of cotinine from saliva and urine are comparable. The half-life of cotinine is approximately 18 hours, and therefore the specificity of salivary test strips may be impacted during the first 4 to 5 days of abstinence. In the first few days of smoking cessation, a more intensive approach, such as quantifying urine cotinine levels and monitoring decline, may be appropriate.23
Continue to: Vignette 3
Vignette 3
Ms. C, age 34 and pregnant, is admitted to an outpatient treatment program for alcohol use disorder. She also has generalized anxiety disorder and tobacco use disorder. In addition to attending group therapy sessions and self-reporting any recent alcohol consumption, Ms. C also undergoes alcohol breathalyzer tests and urine studies of alcohol metabolites to monitor abstinence from alcohol. She says that the regular laboratory screening for alcohol use gives her a sense of accountability and tangible evidence of change that positively impacts her treatment. When the treating psychiatrist recommends that Ms. C also consider addressing her tobacco use disorder, she asks if there is some way to include laboratory testing to monitor her smoking cessation.
Cotinine as a predictor of smoking status
Smoking abstinence rates during pregnancy are lower than that for other substances, and pregnant women may not be aware of the impact of smoking on fetal development.30 Cotinine can be used to verify self-report of smoking status and severity.10,31,32
Salivary cotinine tests are commercially available, relatively economical, and convenient to use when frequent monitoring is required.32 In general, based on established cut-off values that are unique to the specimen collected, the overall high specificity and sensitivity of salivary testing allows clinicians to predict smoker vs nonsmoker status with confidence. For example, a 2008 study reported a salivary cotinine cut-off level of 12 ng/mL for smokers.21 The sensitivity and specificity of this cut-off value for distinguishing cigarette smokers from never smokers were 96.7% and 96.9%, respectively.21
Additionally, some studies suggest that cotinine levels may be predictive of treatment outcomes and retention in SUD treatment programs.33,34 One study of smoking cessation using nicotine replacement products found that compared with patients with lower baseline cotinine levels prior to treatment, patients with higher baseline cotinine plasma levels had lower smoking cessation success rates.34
A few caveats
There are several limitations to quantitative measures of cotinine (Box 221,23). These include (but are not limited to) potential errors related to sample collection, storage, shipping, and analysis.23 Compared with other methods, point-of-care cotinine measurement in saliva is noninvasive, simple, and requires less training to properly use.23
Box 2
Challenges in the collection of samples, storage, shipping, and instrumentation may limit cotinine consistency as a dependable biomarker in the clinical setting.23 Overall, quantitative measurements of cotinine have relative constructive utility in separating smokers from nonsmokers, because daily smokers typically have serum concentrations of 100 ng/mL or higher, in contrast to light/non-daily smokers, who have cotinine concentrations <10 ng/mL. Even heavy exposure to secondhand smoke typically yields plasma concentrations up to approximately 25 ng/mL. However, cotinine is a general metabolite found with the use of all nicotine products, which makes it extremely difficult to differentiate tobacco use from the use of nicotine replacement products, which are frequently used to treat tobacco use disorders.
One potential solution is to measure nicotine-derived nitrosamine ketone (NNK) and its metabolite 4-(methylnitrosamino)- 1-(3-pyridyl)-1-butanol (NNAL). Both NNK and NNAL are tobacco-specific lung carcinogens. NNAL can be measured in the urine. Although total NNAL represents only 15% of NNK dose intake, it has been quantified, with urine concentrations of ≥1,000 fmol/mL for daily smokers. NNAL also has an extremely high specificity to tobacco smoke, and thus allows differentiation of tobacco use from nicotine replacement treatment. Unfortunately, measurement for this biomarker requires specific chemical expertise and expensive equipment.
Another potential barrier to using cotinine in the clinical setting is the variable cut-off levels used in the United States, based on differences in race/ethnicity. This may be secondary to differences in smoking behaviors and/or differences in cotinine metabolism.21
Continue to: Confirmation of smoking cessation...
Confirmation of smoking cessation can be monitored reliably within the clinical setting using cotinine monitoring. However, this is not a routine test, and there are no guidelines or consensus on how or when it should be used. The clinical feasibility of cotinine monitoring for psychiatric patients will depend on the cost of testing, methods used, amount of reimbursement for performing the tests, and how clinicians value such testing.35
Bottom Line
Cotinine is a biomarker that can be used to detect tobacco use. Cotinine measurement can be used to monitor tobacco use and smoking cessation in psychiatric patients. Early detection and treatment of tobacco use disorder can improve patients’ health and reduce the incidence of acute and chronic illnesses. However, cotinine measurement is not a routine test, and there are no guidelines on how or when this test should be used.
Related Resources
- Peckham E, Brabyn S, Cook L, et al. Smoking cessation in severe mental ill health: what works? An updated systematic review and meta-analysis. BMC Psychiatry. 2017;17(1):252.
- Tidey JW, Miller ME. Smoking cessation and reduction in people with chronic mental illness. BMJ. 2015;351:h4065. doi: 10.1136/bmj.h4065
Drug Brand Names
Asenapine • Saphris
Buprenorphine • Sublocade
Clozapine • Clozaril
Duloxetine • Cymbalta
Haloperidol • Haldol
Mirtazapine • Remeron
Olanzapine • Zyprexa
Ziprasidone • Geodon
Zolpidem • Ambien
1. Prochaska JJ, Das S, Young-Wolff KC. Smoking, mental illness, and public health. Annu Rev Public Health. 2017;38:165-185.
2. Pal A, Balhara YP. A review of impact of tobacco use on patients with co-occurring psychiatric disorders. Tob Use Insights. 2016;9:7-12.
3. Lawrence D, Mitrou F, Zubrick SR. Smoking and mental illness: results from population surveys in Australia and the United States. BMC Public Health. 2009;9:285.
4. Kalman D, Morissette SB, George TP. Co-morbidity of smoking in patients with psychiatric and substance use disorders. Am J Addict. 2005;14(2):106-123.
5. Baca CT, Yahne CE. Smoking cessation during substance abuse treatment: what you need to know. J Subst Abuse Treat. 2009;36(2):205-219.
6. Hall SM, Tsoh JY, Prochaska JJ, et al. Treatment for cigarette smoking among depressed mental health outpatients: a randomized clinical trial. Am J Public Health. 2006;96(10):1808-1814.
7. McHugh RK, Votaw VR, Fulciniti F, et al. Perceived barriers to smoking cessation among adults with substance use disorders. J Subst Abuse Treat. 2017;74:48-53.
8. Strong DR, Uebelacker L, Fokas K, et al. Utilization of evidence-based smoking cessation treatments by psychiatric inpatient smokers with depression. J Addict Med. 2014;8(2):77-83.
9. Gayman C, Anderson K, Pietras C. Saliva cotinine as a measure of smoking abstinence in contingency management – a feasibility study. The Psychological Record. 2017;67(2):261-272.
10. Schepis TS, Duhig AM, Liss T, et al. Contingency management for smoking cessation: enhancing feasibility through use of immunoassay test strips measuring cotinine. Nicotine Tob Res. 2008;10(9):1495-1501.
11. Stragierowicz J, Mikolajewska K, Zawadzka-Stolarz M, et al. Estimation of cutoff values of cotinine in urine and saliva for pregnant women in Poland. Biomed Res Int. 2013;2013:386784. doi.org/10.1155/2013/386784
12. Shipton D, Tappin DM, Vadiveloo T, et al. Reliability of self reported smoking status by pregnant women for estimating smoking prevalence: a retrospective, cross sectional study. BMJ. 2009;339:b4347. doi.org/10.1136/bmj.b4347
13. Aubin HJ, Karila L, Reynaud M. Pharmacotherapy for smoking cessation: present and future. Curr Pharm Des. 2011;17(14):1343-1350.
14. McGuffey JE, Wei B, Bernert JT, et al. Validation of a LC-MS/MS method for quantifying urinary nicotine, six nicotine metabolites and the minor tobacco alkaloids--anatabine and anabasine--in smokers’ urine. PLoS One. 2014;9(7):e101816. doi: 10.1371/journal.pone.0101816
15. Duque A, Martinez PJ, Giraldo A, et al. Accuracy of cotinine serum test to detect the smoking habit and its association with periodontal disease in a multicenter study. Med Oral Patol Oral Cir Bucal. 2017;22(4):e425-e431. doi: 10.4317/medoral.21292
16. Avila-Tang E, Elf JL, Cummings KM, et al. Assessing secondhand smoke exposure with reported measures. Tob Control. 2013;22(3):156-163.
17. Benowitz NL, Bernert JT, Foulds J, et al. Biochemical verification of tobacco use and abstinence: 2019 Update. Nicotine Tob Res. 2020;22(7):1086-1097.
18. Nakajima M TY. Interindividual variability in nicotine metabolism: c-oxidation and glucuronidation. Drug Metab Pharmaokinet. 2005;20(4):227-235.
19. Benowitz NL, Bernert JT, Caraballo RS, et al. Optimal serum cotinine levels for distinguishing cigarette smokers and nonsmokers within different racial/ethnic groups in the United States between 1999 and 2004. Am J Epidemiol. 2009;169(2):236-248.
20. Schnoll R, Johnson TA, Lerman C. Genetics and smoking behavior. Curr Psychiatry Rep. 2007;9(5):349-357.
21. Kim S. Overview of cotinine cutoff values for smoking status classification. Int J Environ Res Public Health. 2016;13(12):1236.
22. Etter JF, Bullen C. Saliva cotinine levels in users of electronic cigarettes. Eur Respir J. 2011;38(5):1219-1220.
23. Raja M, Garg A, Yadav P, et al. Diagnostic methods for detection of cotinine level in tobacco users: a review. J Clin Diagn Res. 2016;10(3):ZE04-06. doi: 10.7860/JCDR/2016/17360.7423
24. Balhara YP, Jain R. A receiver operated curve-based evaluation of change in sensitivity and specificity of cotinine urinalysis for detecting active tobacco use. J Cancer Res Ther. 2013;9(1):84-89.
25. Fankhauser M. Drug interactions with tobacco smoke: implications for patient care. Current Psychiatry. 2013;12(1):12-16.
26. Scheuermann TS, Richter KP, Rigotti NA, et al. Accuracy of self-reported smoking abstinence in clinical trials of hospital-initiated smoking interventions. Addiction. 2017;112(12):2227-2236.
27. Holtyn AF, Knealing TW, Jarvis BP, et al. Monitoring cocaine use and abstinence among cocaine users for contingency management interventions. Psychol Rec. 2017;67(2):253-259.
28. Donroe JH, Holt SR, O’Connor PG, et al. Interpreting quantitative urine buprenorphine and norbuprenorphine levels in office-based clinical practice. Drug Alcohol Depend. 2017;180:46-51.
29. Sullivan M, Covey, LS. Current perspectives on smoking cessation among substance abusers. Curr Psychiatry Rep. 2002;4(5):388-396.
30. Forray A, Merry B, Lin H, et al. Perinatal substance use: a prospective evaluation of abstinence and relapse. Drug Alcohol Depend. 2015;150:147-155.
31. Parker DR, Lasater TM, Windsor R, et al. The accuracy of self-reported smoking status assessed by cotinine test strips. Nicotine Tob Res. 2002;4(3):305-309.
32. Asha V, Dhanya M. Immunochromatographic assessment of salivary cotinine and its correlation with nicotine dependence in tobacco chewers. J Cancer Prev. 2015;20(2):159-163.
33. Hall S, Herning RI, Jones RT, et al. Blood cotinine levels as indicators of smoking treatment outcome. Clin Pharmacol Ther. 1984;35(6):810-814.
34. Paoletti P, Fornai E, Maggiorelli F, et al. Importance of baseline cotinine plasma values in smoking cessation: results from a double-blind study with nicotine patch. Eur Respir J. 1996;9(4):643-651.
35. Montalto NJ, Wells WO. Validation of self-reported smoking status using saliva cotinine: a rapid semiquantitative dipstick method. Cancer Epidemiol Biomarkers Prev. 2007;16(9):1858-1862.
1. Prochaska JJ, Das S, Young-Wolff KC. Smoking, mental illness, and public health. Annu Rev Public Health. 2017;38:165-185.
2. Pal A, Balhara YP. A review of impact of tobacco use on patients with co-occurring psychiatric disorders. Tob Use Insights. 2016;9:7-12.
3. Lawrence D, Mitrou F, Zubrick SR. Smoking and mental illness: results from population surveys in Australia and the United States. BMC Public Health. 2009;9:285.
4. Kalman D, Morissette SB, George TP. Co-morbidity of smoking in patients with psychiatric and substance use disorders. Am J Addict. 2005;14(2):106-123.
5. Baca CT, Yahne CE. Smoking cessation during substance abuse treatment: what you need to know. J Subst Abuse Treat. 2009;36(2):205-219.
6. Hall SM, Tsoh JY, Prochaska JJ, et al. Treatment for cigarette smoking among depressed mental health outpatients: a randomized clinical trial. Am J Public Health. 2006;96(10):1808-1814.
7. McHugh RK, Votaw VR, Fulciniti F, et al. Perceived barriers to smoking cessation among adults with substance use disorders. J Subst Abuse Treat. 2017;74:48-53.
8. Strong DR, Uebelacker L, Fokas K, et al. Utilization of evidence-based smoking cessation treatments by psychiatric inpatient smokers with depression. J Addict Med. 2014;8(2):77-83.
9. Gayman C, Anderson K, Pietras C. Saliva cotinine as a measure of smoking abstinence in contingency management – a feasibility study. The Psychological Record. 2017;67(2):261-272.
10. Schepis TS, Duhig AM, Liss T, et al. Contingency management for smoking cessation: enhancing feasibility through use of immunoassay test strips measuring cotinine. Nicotine Tob Res. 2008;10(9):1495-1501.
11. Stragierowicz J, Mikolajewska K, Zawadzka-Stolarz M, et al. Estimation of cutoff values of cotinine in urine and saliva for pregnant women in Poland. Biomed Res Int. 2013;2013:386784. doi.org/10.1155/2013/386784
12. Shipton D, Tappin DM, Vadiveloo T, et al. Reliability of self reported smoking status by pregnant women for estimating smoking prevalence: a retrospective, cross sectional study. BMJ. 2009;339:b4347. doi.org/10.1136/bmj.b4347
13. Aubin HJ, Karila L, Reynaud M. Pharmacotherapy for smoking cessation: present and future. Curr Pharm Des. 2011;17(14):1343-1350.
14. McGuffey JE, Wei B, Bernert JT, et al. Validation of a LC-MS/MS method for quantifying urinary nicotine, six nicotine metabolites and the minor tobacco alkaloids--anatabine and anabasine--in smokers’ urine. PLoS One. 2014;9(7):e101816. doi: 10.1371/journal.pone.0101816
15. Duque A, Martinez PJ, Giraldo A, et al. Accuracy of cotinine serum test to detect the smoking habit and its association with periodontal disease in a multicenter study. Med Oral Patol Oral Cir Bucal. 2017;22(4):e425-e431. doi: 10.4317/medoral.21292
16. Avila-Tang E, Elf JL, Cummings KM, et al. Assessing secondhand smoke exposure with reported measures. Tob Control. 2013;22(3):156-163.
17. Benowitz NL, Bernert JT, Foulds J, et al. Biochemical verification of tobacco use and abstinence: 2019 Update. Nicotine Tob Res. 2020;22(7):1086-1097.
18. Nakajima M TY. Interindividual variability in nicotine metabolism: c-oxidation and glucuronidation. Drug Metab Pharmaokinet. 2005;20(4):227-235.
19. Benowitz NL, Bernert JT, Caraballo RS, et al. Optimal serum cotinine levels for distinguishing cigarette smokers and nonsmokers within different racial/ethnic groups in the United States between 1999 and 2004. Am J Epidemiol. 2009;169(2):236-248.
20. Schnoll R, Johnson TA, Lerman C. Genetics and smoking behavior. Curr Psychiatry Rep. 2007;9(5):349-357.
21. Kim S. Overview of cotinine cutoff values for smoking status classification. Int J Environ Res Public Health. 2016;13(12):1236.
22. Etter JF, Bullen C. Saliva cotinine levels in users of electronic cigarettes. Eur Respir J. 2011;38(5):1219-1220.
23. Raja M, Garg A, Yadav P, et al. Diagnostic methods for detection of cotinine level in tobacco users: a review. J Clin Diagn Res. 2016;10(3):ZE04-06. doi: 10.7860/JCDR/2016/17360.7423
24. Balhara YP, Jain R. A receiver operated curve-based evaluation of change in sensitivity and specificity of cotinine urinalysis for detecting active tobacco use. J Cancer Res Ther. 2013;9(1):84-89.
25. Fankhauser M. Drug interactions with tobacco smoke: implications for patient care. Current Psychiatry. 2013;12(1):12-16.
26. Scheuermann TS, Richter KP, Rigotti NA, et al. Accuracy of self-reported smoking abstinence in clinical trials of hospital-initiated smoking interventions. Addiction. 2017;112(12):2227-2236.
27. Holtyn AF, Knealing TW, Jarvis BP, et al. Monitoring cocaine use and abstinence among cocaine users for contingency management interventions. Psychol Rec. 2017;67(2):253-259.
28. Donroe JH, Holt SR, O’Connor PG, et al. Interpreting quantitative urine buprenorphine and norbuprenorphine levels in office-based clinical practice. Drug Alcohol Depend. 2017;180:46-51.
29. Sullivan M, Covey, LS. Current perspectives on smoking cessation among substance abusers. Curr Psychiatry Rep. 2002;4(5):388-396.
30. Forray A, Merry B, Lin H, et al. Perinatal substance use: a prospective evaluation of abstinence and relapse. Drug Alcohol Depend. 2015;150:147-155.
31. Parker DR, Lasater TM, Windsor R, et al. The accuracy of self-reported smoking status assessed by cotinine test strips. Nicotine Tob Res. 2002;4(3):305-309.
32. Asha V, Dhanya M. Immunochromatographic assessment of salivary cotinine and its correlation with nicotine dependence in tobacco chewers. J Cancer Prev. 2015;20(2):159-163.
33. Hall S, Herning RI, Jones RT, et al. Blood cotinine levels as indicators of smoking treatment outcome. Clin Pharmacol Ther. 1984;35(6):810-814.
34. Paoletti P, Fornai E, Maggiorelli F, et al. Importance of baseline cotinine plasma values in smoking cessation: results from a double-blind study with nicotine patch. Eur Respir J. 1996;9(4):643-651.
35. Montalto NJ, Wells WO. Validation of self-reported smoking status using saliva cotinine: a rapid semiquantitative dipstick method. Cancer Epidemiol Biomarkers Prev. 2007;16(9):1858-1862.
Cannabinoid-based medications for pain
Against the backdrop of an increasing opioid use epidemic and a marked acceleration of prescription opioid–related deaths,1,2 there has been an impetus to explore the usefulness of alternative and co-analgesic agents to assist patients with chronic pain. Preclinical studies employing animal-based models of human pain syndromes have demonstrated that cannabis and chemicals derived from cannabis extracts may mitigate several pain conditions.3
Because there are significant comorbidities between psychiatric disorders and chronic pain, psychiatrists are likely to care for patients with chronic pain. As the availability of and interest in cannabinoid-based medications (CBM) increases, psychiatrists will need to be apprised of the utility, adverse effects, and potential drug interactions of these agents.
The endocannabinoid system and cannabis receptors
The endogenous cannabinoid (endocannabinoid) system is abundantly present within the peripheral and central nervous systems. The first identified, and best studied, endocannabinoids are N-arachidonoyl-ethanolamine (AEA; anandamide) and 2-arachidonoylglycerol (2-AG).4 Unlike typical neurotransmitters, AEA and 2-AG are not stored within vesicles within presynaptic neuron axons. Instead, they are lipophilic molecules produced on demand, synthesized from phospholipids (ie, arachidonic acid derivatives) at the membranes of post-synaptic neurons, and released into the synapse directly.5
Acting as retrograde messengers, the endocannabinoids traverse the synapse, binding to receptors located on the axons of the presynaptic neuron. Two receptors—CB1 and CB2—have been most extensively studied and characterized.6,7 These receptors couple to Gi/o-proteins to inhibit adenylate cyclase, decreasing Ca2+ conductance and increasing K+ conductance.8 Once activated, cannabinoid receptors modulate neurotransmitter release from presynaptic axon terminals. Evidence points to a similar retrograde signaling between neurons and glial cells. Shortly after receptor activation, the endocannabinoids are deactivated by the actions of a transporter mechanism and enzyme degradation.9,10
The endocannabinoid system and pain transmission
Cannabinoid receptors are present in pain transmission circuits spanning from the peripheral sensory nerve endings (from which pain signals originate) to the spinal cord and supraspinal regions within the brain.11-14 CB1 receptors are abundantly present within the CNS, including regions involved in pain transmission. Binding to CB1 receptors, endocannabinoids modulate neurotransmission that impacts pain transmission centrally. Endocannabinoids can also indirectly modulate opiate and N-methyl-
By contrast, CB2 receptors are predominantly localized to peripheral tissues and immune cells, although there has been some discovery of their presence within the CNS (eg, on microglia). Endocannabinoid activation of CB2 receptors is thought to modulate the activity of peripheral afferent pain fibers and immune-mediated neuroinflammatory processes—such as inhibition of prostaglandin synthesis and mast cell degranulation—that can precipitate and maintain chronic pain states.16-18
Evidence garnered from preclinical (animal) studies points to the role of the endocannabinoid system in modulating normal pain transmission (see Manzanares et al3 for details). These studies offer a putative basis for understanding how exogenous cannabinoid congeners might serve to ameliorate pain transmission in pathophysiologic states, including chronic pain.
Continue to: Cannabinoid-based medications
Cannabinoid-based medications
Marijuana contains multiple components (cannabinoids). The most extensively studied are delta-9-tetrahydrocannabinol (THC) and cannabidiol (CBD). Because it predominantly binds CB1 receptors centrally, THC is the major psychoactive component of cannabis; it promotes sleep and appetite, influences anxiety, and produces the “high” associated with cannabis use. By contrast, CBD weakly binds CB1 and thus exerts minimal or no psychoactive effects.19
Cannabinoid absorption, metabolism, bioavailability, and clinical effects vary depending on the formulation and method of administration (Table 1).20-22 THC and CBD content and potency in inhaled cannabis can vary significantly depending on the strains of the cannabis plant and manner of cultivation.23 To standardize approaches for administering cannabinoids in clinical trials and for clinical use, researchers have developed pharmaceutical analogs that contain extracted chemicals or synthetic chemicals similar to THC and/or CBD.
In this article, CBM refers to smoked/vaporized herbal cannabis as well as pharmaceutical cannabis analogs. Table 2 summarizes the characteristics of CBM commonly used in studies investigating their use for managing pain conditions.
CBM for chronic pain
The literature base examining the role of CBM for managing chronic nonmalignant and malignant pain of varying etiologies is rapidly expanding. Randomized controlled trials (RCTs) have focused on inhaled/smoked products and related cannabinoid medications, some of which are FDA-approved (Table 2).
A multitude of other cannabinoid-based products are currently commercially available to consumers, including tincture and oil-based products; over-the-counter CBD products; and several other formulations of CBM (eg, edible and suppository products). Because such products are not standardized or quality-controlled,24 RCTs have not assessed their efficacy for mitigating pain. Consequently, the findings summarized in this article do not address the utility of these agents.
Continue to: CBM for non-cancer pain
CBM for non-cancer pain
Neuropathic pain. Randomized controlled trials have assessed the pain-mitigating effects of various CBM, including inhaled cannabis, synthetic THC, plant-extracted CBD, and a THC/CBD spray. Studies have shown that inhaled/vaporized cannabis can produce short-term pain reduction in patients with chronic neuropathic pain of diverse etiologies, including diabetes mellitus-, HIV-, trauma-, and medication-induced neuropathies.22,25,26 Similar beneficial effects have been observed with the use of cannabis analogues (eg, nabiximols).25,26-29
Meta-analyses and systematic reviews have determined that most of these RCTs were of low-to-moderate quality.26,30 Meta-analyses have revealed divergent and conflicting results because of differences in the inclusion and exclusion criteria used to select RCTs for analysis and differences in the standards with which the quality of evidence were determined.25,30
Overall, the benefit of CBM for mitigating neuropathic pain is promising, but the effectiveness may not be robust.30,31 Several noteworthy caveats limit the interpretation of the results of these RCTs:
- due to the small sample sizes and brief durations of study, questions remain regarding the extent to which effects are generalizable, whether the benefits are sustained, and whether adverse effects emerge over time with continued use
- most RCTs evaluated inhaled (herbal) cannabis and nabiximols; there is little data on the effectiveness of other CBM formulations25,26,30
- the pain-mitigating effects of CBM were usually compared with those of placebo; the comparative efficacy against agents commonly used to treat neuropathic pain remains largely unexamined
- these RCTs typically compared mean pain severity score differences between cannabis-treated and placebo groups using standard subjective rating scales of pain intensity, such as the Numerical Rating Scale or Visual Analogue Scale. Customarily, the pain literature has used a 30% or 50% reduction in pain severity from baseline as an indicator of significant clinical improvement.32,33 The RCTs of CBM for neuropathic pain rarely used this standard, which makes it unclear whether CBM results in clinically significant pain reductions30
- indirect measures of effectiveness (ie, whether using CBM reduces the need for opioids or other analgesics to manage pain) were seldom reported in these RCTs.
Due to these limitations, clinical guidelines and systematic reviews consider CBM as a third- or fourth-line therapy for patients experiencing chronic neuropathic pain for whom conventional agents such as anticonvulsants and antidepressants have failed.34,35
Spasticity in multiple sclerosis (MS). Several RCTs have assessed the use of CBM for MS-related spasticity, although few were deemed to be high quality. Nabiximols and synthetic THC were effective in managing spasticity and reducing pain severity associated with muscle spasms.36 Generally, investigations revealed that CBM were associated with improvements in subjective measures of spasticity, but these were not born out in clinical, objective measures.26,37 The efficacy of smoked cannabis was uncertain.37 The existing literature on CBM for MS-related spasticity does not address dosing, duration of effects, tolerability, or comparative effectiveness against conventional anti-spasm medications.36,37
Continue to: Other chronic pain conditions
Other chronic pain conditions. CBM have also been studied for their usefulness in several other noncancer chronic conditions, including Crohn’s disease, inflammatory bowel disease, fibromyalgia, and other rheumatologic pain conditions.22,31,38-40 However, a solid foundation of empirical work to inform their utility for managing pain in these conditions is lacking.
CBM for cancer pain
Anecdotal evidence suggests that inhaled cannabis has promising pain-mitigating effects in patients with advanced cancer.41-43 There is a dearth of high-quality RCTs assessing the utility of CBM in patients with cancer pain.43-45 The types of CBM used and dosing strategies varied across RCTs, which makes it difficult to infer how best to treat patients with cancer pain. The agents studied included nabiximols, THC spray, and synthetic THC capsules.43-45 Although some studies have demonstrated that synthetic THC and nabiximols have potential for reducing subjective pain ratings compared with placebo,46,47 these results were inconsistent.46,48 Oromucosal nabiximols did not appear to confer any additional analgesic benefit in patients who were already prescribed opioids.31,45
The benefit of CBM for mitigating cancer pain is promising, but it remains difficult to know how to position the use of CBM in managing cancer pain. Limitations in the cancer literature include:
- the RCTs addressing CBM use for cancer pain were often brief, which raises questions about the long-term effectiveness and adverse effects of these agents
- tolerability and dosing limits encountered due to adverse effects were seldom reported43,45
- the types of cancer pain that patients had were often quite diverse. The small sample sizes and the heterogeneity of conditions included in these RCTs limit the ability to determine whether pain-mitigating effects might vary according to type of cancer-related pain.31,45
Despite these limitations, some clinical guidelines and systematic reviews have suggested that CBM have some role in addressing refractory malignant pain conditions.49
Psychiatric considerations related to CBM
As of November 2020, 36 states had legalized the use of cannabis for medical purposes, typically for painful conditions, despite the fact that empirical evidence to support their efficacy is mixed.50 In light of recent changes in both the legal and popular attitudes regarding cannabis, the implications of legalizing CBM remains to be seen. For example, some research suggests that adults with pain are vulnerable to frequent nonmedical cannabis use and/or cannabis use disorder.51 Although well-intended, the legalization of CBM use might represent society’s next misstep in the quest to address the suffering of patients with chronic pain. Some evidence shows that cannabis use and cannabis use disorders increase in states that have legalized medical marijuana.52,53 Psychiatrists will be on the front lines of addressing any potential consequences arising from the use of CBM for treating pain.
Continue to: Psychiatric disorders and CBM
Psychiatric disorders and CBM. The psychological impact of CBM use among patients enduring chronic pain can include sedation, cognitive/attention disturbance, and fatigue. These adverse effects can limit the utility of such agents.22,29,45
Contraindications for CBM use, and conditions for which CBM ought to be used with caution, are listed in Table 354,55.The safety of CBM, particularly in patients with chronic pain and psychiatric disorders, has not been examined. Patients with psychiatric disorders may be poor candidates for medical cannabis. Epidemiologic data suggest that recreational cannabis use is positively associated both cross-sectionally and prospectively with psychotic spectrum disorders, depressive symptoms, and anxiety symptoms, including panic disorder.56 Psychotic reactions have also been associated with CBM (dronabinol and nabilone).57 Cannabis use also has been associated with an earlier onset of, and lower remission rates of, symptoms associated with bipolar disorder.58,59 Consequently, patients who have been diagnosed with or are at risk for developing any of the aforementioned conditions may not be suitable candidates for CBM. If CBM are used, patients should be closely monitored for the emergence/exacerbation of psychiatric symptoms. The frequency and extent of follow-up is not clear, however. Because of its reduced propensity to produce psychoactive effects, CBD may be safer than THC for managing pain in individuals who have or are vulnerable to developing psychiatric disorders.
There is a lack of evidence to support the use of CBM for treating primary depressive disorders, general anxiety disorder, posttraumatic stress disorder, or psychosis.60,61 Very low-quality evidence suggests that CBM could lead to a small improvement in anxiety among individuals with noncancer pain and MS.60 However, interpreting causality is complicated. It is plausible that, for some patients, subjective improvement in pain severity may be related to reduced anxiety.62 Conversely, it is equally plausible that reductions in emotional distress may reduce the propensity to attend to, and thus magnify, pain severity. In the latter case, the indirect impact of reducing pain by modifying emotional distress can be impacted by the type and dose of CBM used. For example, low concentrations of THC produce anxiolytic effects, but high concentrations may be anxiety-provoking.63,64
Several potential pharmacokinetic drug interactions may arise between herbal cannabis or CBM and other medications (Table 465,66). THC and CBD are both metabolized by cytochrome P450 (CYP) 2C19 and 3A4.65,66 In addition, THC is also metabolized by CYP2C9. Medications that inhibit or induce these enzymes can increase or decrease the bioavailability of THC and CBD.67
Simultaneously, cannabinoids can impact the bioavailability of co-prescribed medications (Table 566,68). Although such CYP enzyme interactions remain a theoretical possibility, it is uncertain whether significant perturbations in plasma concentrations (and clinical effects) have been encountered with prescription medications when co-administered with CBM.69 Nonetheless, patients receiving CBM should be closely monitored for their response to prescribed medications.70
Continue to: Potential CYP enzyme interactions...
Potential CYP enzyme interactions aside, clinicians need to consider the additive effects that may occur when CBM are combined with sympathomimetic agents (eg, tachycardia, hypertension); CNS depressants such as alcohol, benzodiazepines, and opioids (eg, drowsiness, ataxia); or anticholinergics (eg, tachycardia, confusion).71 Inhaled herbal cannabis contains mutagens and can result in lung damage, exacerbations of chronic bronchitis, and certain types of cancer.54,72 Co-prescribing benzodiazepines may be contraindicated in light of their effects on respiratory rate and effort.
The THC contained in CBM produces hormonal effects (ie, significantly increases plasma levels of ghrelin and leptin and decreases peptide YY levels)73 that affect appetite and can produce weight gain. This may be problematic for patients receiving psychoactive medications associated with increased risk of weight gain and dyslipidemia. Because of the association between cannabis use and motor vehicle accidents, patients whose jobs require them to drive or operate industrial equipment may not be ideal candidates for CBM, especially if such patients also consume alcohol or are prescribed benzodiazepines and/or sedative hypnotics.74 Lastly, due to their lipophilicity, cannabinoids cross the placental barrier and can be found in breast milk75 and therefore can affect pregnancy outcomes and neurodevelopment.
Bottom Line
The popularity of cannabinoid-based medications (CBM) for the treatment of chronic pain conditions is growing, but the interest in their use may be outpacing the evidence supporting their analgesic benefits. High-quality, well-controlled randomized controlled trials are needed to decipher whether, and to what extent, these agents can be positioned in chronic pain management. Because psychiatrists are likely to encounter patients considering, or receiving, CBM, they must be aware of the potential benefits, risks, and adverse effects of such treatments.
Related Resources
- Joshi KG. Cannabis-derived compounds: what you need to know. Current Psychiatry. 2020;19(10):64-65. doi:10.12788/ cp.0050
- Gupta S, Phalen T, Gupta S. Medical marijuana: do the benefits outweigh the risks? Current Psychiatry. 2018; 17(1):34-41.
Drug Brand Names
Ajulemic acid • Anabasum
Alprazolam • Xanax
Amitriptyline • Elavil
Aripiprazole • Abilify, Abilify Maintena
Buspirone • BuSpar
Cannabidiol • Epidiolex
Carbamazepine • Tegretol, Equetro
Cimetidine • Tagamet HB
Citalopram • Celexa
Clopidogrel • Plavix
Clozapine • Clozaril
Cyclosporine • Neoral, Sandimmune
Dronabinol • Marinol, Syndros
Duloxetine • Cymbalta
Fluoxetine • Prozac
Fluvoxamine • Luvox
Haloperidol • Haldol
Imipramine • Tofranil
Ketoconazole • Nizoral AD
Losartan • Cozaar
Midazolam • Versed
Mirtazapine • Remeron
Nabilone • Cesamet
Nabiximols • Sativex
Nefazodone • Serzone
Olanzapine • Zyprexa
Phenobarbital • Solfoton
Phenytoin • Dilantin
Ramelteon • Rozerem
Rifampin • Rifadin
Risperidone • Risperdal
Sertraline • Zoloft
Tamoxifen • Nolvadex
Topiramate • Topamax
Valproic acid • Depakote, Depakene
Venlafaxine • Effexor
Verapamil • Verelan
Zolpidem • Ambien
1. Okie S. A floor of opioids, a rising tide of deaths. N Engl J Med. 2010;363(21):1981-1985. doi:10.1056/NEJMp1011512
2. Powell D, Pacula RL, Taylor E. How increasing medical access to opioids contributes to the opioid epidemic: evidence from Medicare Part D. J Health Econ. 2020;71:102286. doi: 10.1016/j.jhealeco.2019.102286
3. Manzanares J, Julian MD, Carrascosa A. Role of the cannabinoid system in pain control and therapeutic implications for the management of acute and chronic pain episodes. Curr Neuropharmacol. 2006;4(3):239-257. doi: 10.2174/157015906778019527
4. Zou S, Kumar U. Cannabinoid receptors and the endocannabinoid system: signaling and function in the central nervous system. Int J Mol Sci. 2018;19(3):833. doi: 10.3390/ijms19030833
5. Huang WJ, Chen WW, Zhang X. Endocannabinoid system: role in depression, reward and pain control (Review). Mol Med Rep. 2016;14(4):2899-2903. doi:10.3892/mmr.2016.5585
6. Mechoulam R, Ben-Shabat S, Hanus L, et al. Identification of an endogenous 2-monoglyceride, present in canine gut, that binds to cannabinoid receptors. Biochem Pharmacol. 1995;50(1):83-90. doi:10.1016/0006-2952(95)00109-d
7. Walker JM, Krey JF, Chu CJ, et al. Endocannabinoids and related fatty acid derivatives in pain modulation. Chem Phys Lipids. 2002;121(1-2):159-172. doi: 10.1016/s0009-3084(02)00152-4
8. Howlett AC. Efficacy in CB1 receptor-mediated signal transduction. Br J Pharmacol. 2004;142(8):1209-1218. doi: 10.1038/sj.bjp.0705881
9. Giuffrida A, Beltramo M, Piomelli D. Mechanisms of endocannabinoid inactivation, biochemistry and pharmacology. J Pharmacol Exp Ther. 2001;298:7-14.
10. Piomelli D, Beltramo M, Giuffrida A, et al. Endogenous cannabinoid signaling. Neurobiol Dis. 1998;5(6 Pt B):462-473. doi: 10.1006/nbdi.1998.0221
11. Eggan SM, Lewis DA. Immunocytochemical distribution of the cannabinoid CB1 receptor in the primate neocortex: a regional and laminar analysis. Cereb Cortex. 2007;17(1):175-191. doi: 10.1093/cercor/bhj136
12. Jennings EA, Vaughan CW, Christie MJ. Cannabinoid actions on rat superficial medullary dorsal horn neurons in vitro. J Physiol. 2001;534(Pt 3):805-812. doi: 10.1111/j.1469-7793.2001.00805.x
13. Vaughan CW, Connor M, Bagley EE, et al. Actions of cannabinoids on membrane properties and synaptic transmission in rat periaqueductal gray neurons in vitro. Mol Pharmacol. 2000;57(2):288-295.
14. Vaughan CW, McGregor IS, Christie MJ. Cannabinoid receptor activation inhibits GABAergic neurotransmission in rostral ventromedial medulla neurons in vitro. Br J Pharmacol. 1999;127(4):935-940. doi: 10.1038/sj.bjp.0702636
15. Raichlen DA, Foster AD, Gerdeman GI, et al. Wired to run: exercise-induced endocannabinoid signaling in humans and cursorial mammals with implications for the “runner’s high.” J Exp Biol. 2012;215(Pt 8):1331-1336. doi: 10.1242/jeb.063677
16. Beltrano M. Cannabinoid type 2 receptor as a target for chronic pain. Mini Rev Chem. 2009;234:253-254.
17. Ibrahim MM, Deng H, Zvonok A, et al. Activation of CB2 cannabinoid receptors by AM1241 inhibits experimental neuropathic pain: pain inhibition by receptors not present in the CNS. Proc Natl Acad Sci U S A. 2003;100(18):10529-10533. doi: 10.1073/pnas.1834309100
18. Valenzano KJ, Tafessem L, Lee G, et al. Pharmacological and pharmacokinetic characterization of the cannabinoid receptor 2 agonist, GW405833, utilizing rodent models of acute and chronic pain, anxiety, ataxia and catalepsy. Neuropharmacology. 2005;48:658-672.
19. Pertwee RG, Howlett AC, Abood ME, et al. International union of basic and clinical pharmacology. LXXIX. Cannabinoid receptors and their ligands: beyond CB1 and CB2. Pharmacol Rev. 2010;62(4):588-631. doi: 10.1124/pr.110.003004
20. Carter GT, Weydt P, Kyashna-Tocha M, et al. Medicinal cannabis: rational guidelines for dosing. Drugs. 2004;7(5):464-470.
21. Huestis MA. Human cannabinoid pharmacokinetics. Chem Biodivers. 2007;4(8):1770-1804.
22. Johal H, Devji T, Chang Y, et al. cannabinoids in chronic non-cancer pain: a systematic review and meta-analysis. Clin Med Insights Arthritis Musculoskelet Disord. 2020;13:1179544120906461. doi: 10.1177/1179544120906461
23. Hillig KW, Mahlberg PG. A chemotaxonomic analysis of cannabinoid variation in Cannabis (Cannabaceae). Am J Bot. 2004;91(6):966-975. doi: 10.3732/ajb.91.6.966
24. Hazekamp A, Ware MA, Muller-Vahl KR, et al. The medicinal use of cannabis and cannabinoids--an international cross-sectional survey on administration forms. J Psychoactive Drugs. 2013;45(3):199-210. doi: 10.1080/02791072.2013.805976
25. Andreae MH, Carter GM, Shaparin N, et al. inhaled cannabis for chronic neuropathic pain: a meta-analysis of individual patient data. J Pain. 2015;16(12):1221-1232. doi: 10.1016/j.jpain.2015.07.009
26. Whiting PF, Wolff RF, Deshpande S, et al. Cannabinoids for medical use: a systematic review and meta-analysis. JAMA. 2015;313(24):2456-2473. doi: 10.1001/jama.2015.6358
27. Boychuk DG, Goddard G, Mauro G, et al. The effectiveness of cannabinoids in the management of chronic nonmalignant neuropathic pain: a systematic review. J Oral Facial Pain Headache. 2015;29(1):7-14. doi: 10.11607/ofph.1274
28. Lynch ME, Campbell F. Cannabinoids for treatment of chronic non-cancer pain; a systematic review of randomized trials. Br J Clin Pharmacol. 2011;72(5):735-744. doi: 10.1111/j.1365-2125.2011.03970.x
29. Stockings E, Campbell G, Hall WD, et al. Cannabis and cannabinoids for the treatment of people with chronic noncancer pain conditions: a systematic review and meta-analysis of controlled and observational studies. Pain. 2018;159(10):1932-1954. doi: 10.1097/j.pain.0000000000001293
30. Mücke M, Phillips T, Radbruch L, et al. Cannabis-based medicines for chronic neuropathic pain in adults. Cochrane Database Syst Rev. 2018;3(3):CD012182. doi: 10.1002/14651858.CD012182.pub2
31. Häuser W, Fitzcharles MA, Radbruch L, et al. Cannabinoids in pain management and palliative medicine. Dtsch Arztebl Int. 2017;114(38):627-634. doi: 10.3238/arztebl.2017.0627
32. Dworkin RH, Turk DC, Wyrwich KW, et al. Interpreting the clinical importance of treatment outcomes in chronic pain clinical trials: IMMPACT recommendations. J Pain. 2008;9(2):105-121. doi: 10.1016/j.jpain.2007.09.005
33. Farrar JT, Troxel AB, Stott C, et al. Validity, reliability, and clinical importance of change in a 0-10 numeric rating scale measure of spasticity: a post hoc analysis of a randomized, double-blind, placebo-controlled trial. Clin Ther. 2008;30(5):974-985. doi: 10.1016/j.clinthera.2008.05.011
34. Moulin D, Boulanger A, Clark AJ, et al. Pharmacological management of chronic neuropathic pain: revised consensus statement from the Canadian Pain Society. Pain Res Manag. 2014;19(6):328-335. doi: 10.1155/2014/754693
35. Petzke F, Enax-Krumova EK, Häuser W. Efficacy, tolerability and safety of cannabinoids for chronic neuropathic pain: a systematic review of randomized controlled studies. Schmerz. 2016;30(1):62-88. doi: 10.1007/s00482-015-0089-y
36. Rice J, Cameron M. Cannabinoids for treatment of MS symptoms: state of the evidence. Curr Neurol Neurosci Rep. 2018;18(8):50. doi: 10.1007/s11910-018-0859-x
37. Koppel BS, Brust JCM, Fife T, et al. Systematic review: efficacy and safety of medical marijuana in selected neurologic disorders. Report of the Guideline Development Subcommittee of the American Academy of Neurology. Neurology. 2014;82(17):1556-1563. doi: 10.1212/WNL.0000000000000363
38. Kafil TS, Nguyen TM, MacDonald JK, et al. Cannabis for the treatment of Crohn’s disease and ulcerative colitis: evidence from Cochrane Reviews. Inflamm Bowel Dis. 2020;26(4):502-509. doi: 10.1093/ibd/izz233
39. Katz-Talmor D, Katz I, Porat-Katz BS, et al. Cannabinoids for the treatment of rheumatic diseases - where do we stand? Nat Rev Rheumatol. 2018;14(8):488-498. doi: 10.1038/s41584-018-0025-5
40. Walitt B, Klose P, Fitzcharles MA, et al. Cannabinoids for fibromyalgia. Cochrane Database Syst Rev. 2016;7(7):CD011694. doi: 10.1002/14651858.CD011694.pub2
41. Bar-Lev Schleider L, Mechoulam R, Lederman V, et al. Prospective analysis of safety and efficacy of medical cannabis in large unselected population of patients with cancer. Eur J Intern Med. 2018;49:37‐43. doi: 10.1016/j.ejim.2018.01.023
42. Bennett M, Paice JA, Wallace M. Pain and opioids in cancer care: benefits, risks, and alternatives. Am Soc Clin Oncol Educ Book. 2017;37:705‐713. doi:10.1200/EDBK_180469
43. Blake A, Wan BA, Malek L, et al. A selective review of medical cannabis in cancer pain management. Ann Palliat Med. 2017;6(Suppl 2):5215-5222. doi: 10.21037/apm.2017.08.05
44. Aviram J, Samuelly-Lechtag G. Efficacy of cannabis-based medicines for pain management: a systematic review and meta-analysis of randomized controlled trials. Pain Physician. 2017;20(6):E755-E796.
45. Häuser W, Welsch P, Klose P, et al. Efficacy, tolerability and safety of cannabis-based medicines for cancer pain: a systematic review with meta-analysis of randomised controlled trials. Schmerz. 2019;33(5):424-436. doi: 10.1007/s00482-019-0373-3
46. Johnson JR, Burnell-Nugent M, Lossignol D, et al. Multicenter, double-blind, randomized, placebo-controlled, parallel-group study of the efficacy, safety, and tolerability of THC:CBD extract and THC extract in patients with intractable cancer-related pain. J Pain Symptom Manage 2010; 39:167-179.
47. Portenoy RK, Ganae-Motan ED, Allende S, et al. Nabiximols for opioid-treated cancer patients with poorly-controlled chronic pain: a randomized, placebo-controlled, graded-dose trial. J Pain. 2012;13(5):438-449. doi: 10.1016/j.jpain.2012.01.003
48. Lynch ME, Cesar-Rittenberg P, Hohmann AG. A double-blind, placebo-controlled, crossover pilot trial with extension using an oral mucosal cannabinoid extract for treatment of chemotherapy-induced neuropathic pain. J Pain Symptom Manage. 2014;47(1):166-173. doi: 10.1016/j.jpainsymman.2013.02.018
49. Kleckner AS, Kleckner IR, Kamen CS, et al. Opportunities for cannabis in supportive care in cancer. Ther Adv Med Oncol. 2019;11:1758835919866362. doi: 10.1177/1758835919866362
50. National Conference of State Legislatures (ncsl.org). State Medical Marijuana Laws. Accessed April 5, 2021. https://www.ncsl.org/research/health/state-medical-marijuana-laws.aspx
51. Hasin DS, Shmulewitz D, Cerda M, et al. US adults with pain, a group increasingly vulnerable to nonmedical cannabis use and cannabis use disorder: 2001-2002 and 2012-2013. Am J Psychiatry. 2020;177(7):611-618. doi: 10.1176/appi.ajp.2019.19030284
52. Hasin DS, Sarvet AL, Cerdá M, et al. US adult illicit cannabis use, cannabis use disorder, and medical marijuana laws: 1991-1992 to 2012-2013. JAMA Psychiatry. 2017;74(6):579-588. doi: 10.1001/jamapsychiatry.2017.0724
53. National Institute on Drug Abuse. Illicit cannabis use and use disorders increase in states with medical marijuana laws. April 26, 2017. Accessed October 24, 2020. https://archives.drugabuse.gov/news-events/news-releases/2017/04/illicit-cannabis-use-use-disorders-increase-in-states-medical-marijuana-laws
54. National Academies of Sciences, Engineering, and Medicine. The health effects of cannabis and cannabinoids: the current state of evidence and recommendations for research. The National Academies Press; 2017. https://doi.org/10.17226/24625
55. Stanford M. Physician recommended marijuana: contraindications & standards of care. A review of the literature. Accessed July 7, 2020. http://drneurosci.com/MedicalMarijuanaStandardsofCare.pdf
56. Repp K, Raich A. Marijuana and health: a comprehensive review of 20 years of research. Washington County Oregon Department of Health and Human Services. 2014. Accessed April 8, 2021. https://www.co.washington.or.us/CAO/upload/HHSmarijuana-review.pdf
57. Parmar JR, Forrest BD, Freeman RA. Medical marijuana patient counseling points for health care professionals based on trends in the medical uses, efficacy, and adverse effects of cannabis-based pharmaceutical drugs. Res Social Adm Pharm. 2016;12(4):638-654. doi: 10.1016/j.sapharm.2015.09.002.
58. Leite RT, Nogueira Sde O, do Nascimento JP, et al. The use of cannabis as a predictor of early onset of bipolar disorder and suicide attempts. Neural Plast. 2015;2015:434127. doi: 10.1155/2015/43412
59. Kim SW, Dodd S, Berk L, et al. Impact of cannabis use on long-term remission in bipolar I and schizoaffective disorder. Psychiatry Investig. 2015;12(3):349-355. doi: 10.4306/pi.2015.12.3.349
60. Black N, Stockings E, Campbell G, et al. Cannabinoids for the treatment of mental disorders and symptoms of mental disorders: a systematic review and meta-analysis. Lancet Psychiatry. 2019;6(12):995-1010.
61. Wilkinson ST, Radhakrishnan R, D’Souza DC. A systematic review of the evidence for medical marijuana in psychiatric indications. J Clin Psychiatry. 2016;77(8):1050-1064. doi: 10.4088/JCP.15r10036.
62. Woolf CJ, American College of Physicians. American Physiological Society Pain: moving from symptom control toward mechanism-specific pharmacologic management. Ann Intern Med. 2004;140(6):441-451.
63. Crippa JA, Zuardi AW, Martín-Santos R, et al. Cannabis and anxiety: a critical review of the evidence. Hum Psychopharmacol. 2009;24(7):515‐523. doi: 10.1002/hup.1048
64. Sachs J, McGlade E, Yurgelun-Todd D. Safety and toxicology of cannabinoids. Neurotherapeutics. 2015;12(4):735‐746. doi: 10.1007/s13311-015-0380-8
65. Antoniou T, Bodkin J, Ho JMW. Drug interactions with cannabinoids. CMAJ. 2020;2;192:E206. doi: 10.1503/cmaj.191097
66. Brown JD. Potential adverse drug events with tetrahydrocannabinol (THC) due to drug-drug interactions. J Clin Med. 2020;9(4):919. doi: 10.3390/jcm9040919.
67. Maida V, Daeninck P. A user’s guide to cannabinoid therapy in oncology. Curr Oncol. 2016;23(6):398-406. doi: http://dx.doi.org/10.3747/co.23.3487
68. Stout SM, Cimino NM. Exogenous cannabinoids as substrates, inhibitors, and inducers of human drug metabolizing enzymes: a systematic review. Drug Metab Rev. 2014;46(1):86-95. doi: 10.3109/03602532.2013.849268
69. Abrams DI. Integrating cannabis into clinical cancer care. Curr Oncol. 2016;23(52):S8-S14.
70. Alsherbiny MA, Li CG. Medicinal cannabis—potential drug interactions. Medicines. 2018;6(1):3. doi: 10.3390/medicines6010003
71. Lucas CJ, Galettis P, Schneider J. The pharmacokinetics and the pharmacodynamics of cannabinoids. Br J Clin Pharmacol. 2018;84:2477-2482.
72. Ghasemiesfe M, Barrow B, Leonard S, et al. Association between marijuana use and risk of cancer: a systematic review and meta-analysis. JAMA Netw Open. 2019;2(11):e1916318. doi: 10.1001/jamanetworkopen.2019.16318
73. Riggs PK, Vaida F, Rossi SS, et al. A pilot study of the effects of cannabis on appetite hormones in HIV-infected adult men. Brain Res. 2012;1431:46-52. doi: 10.1016/j.brainres.2011.11.001
74. Asbridge M, Hayden JA, Cartwright JL. Acute cannabis consumption and motor vehicle collision risk: systematic review of observational studies and meta-analysis. BMJ. 2012;344:e536. doi: 10.1136/bmj.e536
75. Carlier J, Huestis MA, Zaami S, et al. Monitoring perinatal exposure to cannabis and synthetic cannabinoids. Ther Drug Monit. 2020;42(2):194-204.
Against the backdrop of an increasing opioid use epidemic and a marked acceleration of prescription opioid–related deaths,1,2 there has been an impetus to explore the usefulness of alternative and co-analgesic agents to assist patients with chronic pain. Preclinical studies employing animal-based models of human pain syndromes have demonstrated that cannabis and chemicals derived from cannabis extracts may mitigate several pain conditions.3
Because there are significant comorbidities between psychiatric disorders and chronic pain, psychiatrists are likely to care for patients with chronic pain. As the availability of and interest in cannabinoid-based medications (CBM) increases, psychiatrists will need to be apprised of the utility, adverse effects, and potential drug interactions of these agents.
The endocannabinoid system and cannabis receptors
The endogenous cannabinoid (endocannabinoid) system is abundantly present within the peripheral and central nervous systems. The first identified, and best studied, endocannabinoids are N-arachidonoyl-ethanolamine (AEA; anandamide) and 2-arachidonoylglycerol (2-AG).4 Unlike typical neurotransmitters, AEA and 2-AG are not stored within vesicles within presynaptic neuron axons. Instead, they are lipophilic molecules produced on demand, synthesized from phospholipids (ie, arachidonic acid derivatives) at the membranes of post-synaptic neurons, and released into the synapse directly.5
Acting as retrograde messengers, the endocannabinoids traverse the synapse, binding to receptors located on the axons of the presynaptic neuron. Two receptors—CB1 and CB2—have been most extensively studied and characterized.6,7 These receptors couple to Gi/o-proteins to inhibit adenylate cyclase, decreasing Ca2+ conductance and increasing K+ conductance.8 Once activated, cannabinoid receptors modulate neurotransmitter release from presynaptic axon terminals. Evidence points to a similar retrograde signaling between neurons and glial cells. Shortly after receptor activation, the endocannabinoids are deactivated by the actions of a transporter mechanism and enzyme degradation.9,10
The endocannabinoid system and pain transmission
Cannabinoid receptors are present in pain transmission circuits spanning from the peripheral sensory nerve endings (from which pain signals originate) to the spinal cord and supraspinal regions within the brain.11-14 CB1 receptors are abundantly present within the CNS, including regions involved in pain transmission. Binding to CB1 receptors, endocannabinoids modulate neurotransmission that impacts pain transmission centrally. Endocannabinoids can also indirectly modulate opiate and N-methyl-
By contrast, CB2 receptors are predominantly localized to peripheral tissues and immune cells, although there has been some discovery of their presence within the CNS (eg, on microglia). Endocannabinoid activation of CB2 receptors is thought to modulate the activity of peripheral afferent pain fibers and immune-mediated neuroinflammatory processes—such as inhibition of prostaglandin synthesis and mast cell degranulation—that can precipitate and maintain chronic pain states.16-18
Evidence garnered from preclinical (animal) studies points to the role of the endocannabinoid system in modulating normal pain transmission (see Manzanares et al3 for details). These studies offer a putative basis for understanding how exogenous cannabinoid congeners might serve to ameliorate pain transmission in pathophysiologic states, including chronic pain.
Continue to: Cannabinoid-based medications
Cannabinoid-based medications
Marijuana contains multiple components (cannabinoids). The most extensively studied are delta-9-tetrahydrocannabinol (THC) and cannabidiol (CBD). Because it predominantly binds CB1 receptors centrally, THC is the major psychoactive component of cannabis; it promotes sleep and appetite, influences anxiety, and produces the “high” associated with cannabis use. By contrast, CBD weakly binds CB1 and thus exerts minimal or no psychoactive effects.19
Cannabinoid absorption, metabolism, bioavailability, and clinical effects vary depending on the formulation and method of administration (Table 1).20-22 THC and CBD content and potency in inhaled cannabis can vary significantly depending on the strains of the cannabis plant and manner of cultivation.23 To standardize approaches for administering cannabinoids in clinical trials and for clinical use, researchers have developed pharmaceutical analogs that contain extracted chemicals or synthetic chemicals similar to THC and/or CBD.
In this article, CBM refers to smoked/vaporized herbal cannabis as well as pharmaceutical cannabis analogs. Table 2 summarizes the characteristics of CBM commonly used in studies investigating their use for managing pain conditions.
CBM for chronic pain
The literature base examining the role of CBM for managing chronic nonmalignant and malignant pain of varying etiologies is rapidly expanding. Randomized controlled trials (RCTs) have focused on inhaled/smoked products and related cannabinoid medications, some of which are FDA-approved (Table 2).
A multitude of other cannabinoid-based products are currently commercially available to consumers, including tincture and oil-based products; over-the-counter CBD products; and several other formulations of CBM (eg, edible and suppository products). Because such products are not standardized or quality-controlled,24 RCTs have not assessed their efficacy for mitigating pain. Consequently, the findings summarized in this article do not address the utility of these agents.
Continue to: CBM for non-cancer pain
CBM for non-cancer pain
Neuropathic pain. Randomized controlled trials have assessed the pain-mitigating effects of various CBM, including inhaled cannabis, synthetic THC, plant-extracted CBD, and a THC/CBD spray. Studies have shown that inhaled/vaporized cannabis can produce short-term pain reduction in patients with chronic neuropathic pain of diverse etiologies, including diabetes mellitus-, HIV-, trauma-, and medication-induced neuropathies.22,25,26 Similar beneficial effects have been observed with the use of cannabis analogues (eg, nabiximols).25,26-29
Meta-analyses and systematic reviews have determined that most of these RCTs were of low-to-moderate quality.26,30 Meta-analyses have revealed divergent and conflicting results because of differences in the inclusion and exclusion criteria used to select RCTs for analysis and differences in the standards with which the quality of evidence were determined.25,30
Overall, the benefit of CBM for mitigating neuropathic pain is promising, but the effectiveness may not be robust.30,31 Several noteworthy caveats limit the interpretation of the results of these RCTs:
- due to the small sample sizes and brief durations of study, questions remain regarding the extent to which effects are generalizable, whether the benefits are sustained, and whether adverse effects emerge over time with continued use
- most RCTs evaluated inhaled (herbal) cannabis and nabiximols; there is little data on the effectiveness of other CBM formulations25,26,30
- the pain-mitigating effects of CBM were usually compared with those of placebo; the comparative efficacy against agents commonly used to treat neuropathic pain remains largely unexamined
- these RCTs typically compared mean pain severity score differences between cannabis-treated and placebo groups using standard subjective rating scales of pain intensity, such as the Numerical Rating Scale or Visual Analogue Scale. Customarily, the pain literature has used a 30% or 50% reduction in pain severity from baseline as an indicator of significant clinical improvement.32,33 The RCTs of CBM for neuropathic pain rarely used this standard, which makes it unclear whether CBM results in clinically significant pain reductions30
- indirect measures of effectiveness (ie, whether using CBM reduces the need for opioids or other analgesics to manage pain) were seldom reported in these RCTs.
Due to these limitations, clinical guidelines and systematic reviews consider CBM as a third- or fourth-line therapy for patients experiencing chronic neuropathic pain for whom conventional agents such as anticonvulsants and antidepressants have failed.34,35
Spasticity in multiple sclerosis (MS). Several RCTs have assessed the use of CBM for MS-related spasticity, although few were deemed to be high quality. Nabiximols and synthetic THC were effective in managing spasticity and reducing pain severity associated with muscle spasms.36 Generally, investigations revealed that CBM were associated with improvements in subjective measures of spasticity, but these were not born out in clinical, objective measures.26,37 The efficacy of smoked cannabis was uncertain.37 The existing literature on CBM for MS-related spasticity does not address dosing, duration of effects, tolerability, or comparative effectiveness against conventional anti-spasm medications.36,37
Continue to: Other chronic pain conditions
Other chronic pain conditions. CBM have also been studied for their usefulness in several other noncancer chronic conditions, including Crohn’s disease, inflammatory bowel disease, fibromyalgia, and other rheumatologic pain conditions.22,31,38-40 However, a solid foundation of empirical work to inform their utility for managing pain in these conditions is lacking.
CBM for cancer pain
Anecdotal evidence suggests that inhaled cannabis has promising pain-mitigating effects in patients with advanced cancer.41-43 There is a dearth of high-quality RCTs assessing the utility of CBM in patients with cancer pain.43-45 The types of CBM used and dosing strategies varied across RCTs, which makes it difficult to infer how best to treat patients with cancer pain. The agents studied included nabiximols, THC spray, and synthetic THC capsules.43-45 Although some studies have demonstrated that synthetic THC and nabiximols have potential for reducing subjective pain ratings compared with placebo,46,47 these results were inconsistent.46,48 Oromucosal nabiximols did not appear to confer any additional analgesic benefit in patients who were already prescribed opioids.31,45
The benefit of CBM for mitigating cancer pain is promising, but it remains difficult to know how to position the use of CBM in managing cancer pain. Limitations in the cancer literature include:
- the RCTs addressing CBM use for cancer pain were often brief, which raises questions about the long-term effectiveness and adverse effects of these agents
- tolerability and dosing limits encountered due to adverse effects were seldom reported43,45
- the types of cancer pain that patients had were often quite diverse. The small sample sizes and the heterogeneity of conditions included in these RCTs limit the ability to determine whether pain-mitigating effects might vary according to type of cancer-related pain.31,45
Despite these limitations, some clinical guidelines and systematic reviews have suggested that CBM have some role in addressing refractory malignant pain conditions.49
Psychiatric considerations related to CBM
As of November 2020, 36 states had legalized the use of cannabis for medical purposes, typically for painful conditions, despite the fact that empirical evidence to support their efficacy is mixed.50 In light of recent changes in both the legal and popular attitudes regarding cannabis, the implications of legalizing CBM remains to be seen. For example, some research suggests that adults with pain are vulnerable to frequent nonmedical cannabis use and/or cannabis use disorder.51 Although well-intended, the legalization of CBM use might represent society’s next misstep in the quest to address the suffering of patients with chronic pain. Some evidence shows that cannabis use and cannabis use disorders increase in states that have legalized medical marijuana.52,53 Psychiatrists will be on the front lines of addressing any potential consequences arising from the use of CBM for treating pain.
Continue to: Psychiatric disorders and CBM
Psychiatric disorders and CBM. The psychological impact of CBM use among patients enduring chronic pain can include sedation, cognitive/attention disturbance, and fatigue. These adverse effects can limit the utility of such agents.22,29,45
Contraindications for CBM use, and conditions for which CBM ought to be used with caution, are listed in Table 354,55.The safety of CBM, particularly in patients with chronic pain and psychiatric disorders, has not been examined. Patients with psychiatric disorders may be poor candidates for medical cannabis. Epidemiologic data suggest that recreational cannabis use is positively associated both cross-sectionally and prospectively with psychotic spectrum disorders, depressive symptoms, and anxiety symptoms, including panic disorder.56 Psychotic reactions have also been associated with CBM (dronabinol and nabilone).57 Cannabis use also has been associated with an earlier onset of, and lower remission rates of, symptoms associated with bipolar disorder.58,59 Consequently, patients who have been diagnosed with or are at risk for developing any of the aforementioned conditions may not be suitable candidates for CBM. If CBM are used, patients should be closely monitored for the emergence/exacerbation of psychiatric symptoms. The frequency and extent of follow-up is not clear, however. Because of its reduced propensity to produce psychoactive effects, CBD may be safer than THC for managing pain in individuals who have or are vulnerable to developing psychiatric disorders.
There is a lack of evidence to support the use of CBM for treating primary depressive disorders, general anxiety disorder, posttraumatic stress disorder, or psychosis.60,61 Very low-quality evidence suggests that CBM could lead to a small improvement in anxiety among individuals with noncancer pain and MS.60 However, interpreting causality is complicated. It is plausible that, for some patients, subjective improvement in pain severity may be related to reduced anxiety.62 Conversely, it is equally plausible that reductions in emotional distress may reduce the propensity to attend to, and thus magnify, pain severity. In the latter case, the indirect impact of reducing pain by modifying emotional distress can be impacted by the type and dose of CBM used. For example, low concentrations of THC produce anxiolytic effects, but high concentrations may be anxiety-provoking.63,64
Several potential pharmacokinetic drug interactions may arise between herbal cannabis or CBM and other medications (Table 465,66). THC and CBD are both metabolized by cytochrome P450 (CYP) 2C19 and 3A4.65,66 In addition, THC is also metabolized by CYP2C9. Medications that inhibit or induce these enzymes can increase or decrease the bioavailability of THC and CBD.67
Simultaneously, cannabinoids can impact the bioavailability of co-prescribed medications (Table 566,68). Although such CYP enzyme interactions remain a theoretical possibility, it is uncertain whether significant perturbations in plasma concentrations (and clinical effects) have been encountered with prescription medications when co-administered with CBM.69 Nonetheless, patients receiving CBM should be closely monitored for their response to prescribed medications.70
Continue to: Potential CYP enzyme interactions...
Potential CYP enzyme interactions aside, clinicians need to consider the additive effects that may occur when CBM are combined with sympathomimetic agents (eg, tachycardia, hypertension); CNS depressants such as alcohol, benzodiazepines, and opioids (eg, drowsiness, ataxia); or anticholinergics (eg, tachycardia, confusion).71 Inhaled herbal cannabis contains mutagens and can result in lung damage, exacerbations of chronic bronchitis, and certain types of cancer.54,72 Co-prescribing benzodiazepines may be contraindicated in light of their effects on respiratory rate and effort.
The THC contained in CBM produces hormonal effects (ie, significantly increases plasma levels of ghrelin and leptin and decreases peptide YY levels)73 that affect appetite and can produce weight gain. This may be problematic for patients receiving psychoactive medications associated with increased risk of weight gain and dyslipidemia. Because of the association between cannabis use and motor vehicle accidents, patients whose jobs require them to drive or operate industrial equipment may not be ideal candidates for CBM, especially if such patients also consume alcohol or are prescribed benzodiazepines and/or sedative hypnotics.74 Lastly, due to their lipophilicity, cannabinoids cross the placental barrier and can be found in breast milk75 and therefore can affect pregnancy outcomes and neurodevelopment.
Bottom Line
The popularity of cannabinoid-based medications (CBM) for the treatment of chronic pain conditions is growing, but the interest in their use may be outpacing the evidence supporting their analgesic benefits. High-quality, well-controlled randomized controlled trials are needed to decipher whether, and to what extent, these agents can be positioned in chronic pain management. Because psychiatrists are likely to encounter patients considering, or receiving, CBM, they must be aware of the potential benefits, risks, and adverse effects of such treatments.
Related Resources
- Joshi KG. Cannabis-derived compounds: what you need to know. Current Psychiatry. 2020;19(10):64-65. doi:10.12788/ cp.0050
- Gupta S, Phalen T, Gupta S. Medical marijuana: do the benefits outweigh the risks? Current Psychiatry. 2018; 17(1):34-41.
Drug Brand Names
Ajulemic acid • Anabasum
Alprazolam • Xanax
Amitriptyline • Elavil
Aripiprazole • Abilify, Abilify Maintena
Buspirone • BuSpar
Cannabidiol • Epidiolex
Carbamazepine • Tegretol, Equetro
Cimetidine • Tagamet HB
Citalopram • Celexa
Clopidogrel • Plavix
Clozapine • Clozaril
Cyclosporine • Neoral, Sandimmune
Dronabinol • Marinol, Syndros
Duloxetine • Cymbalta
Fluoxetine • Prozac
Fluvoxamine • Luvox
Haloperidol • Haldol
Imipramine • Tofranil
Ketoconazole • Nizoral AD
Losartan • Cozaar
Midazolam • Versed
Mirtazapine • Remeron
Nabilone • Cesamet
Nabiximols • Sativex
Nefazodone • Serzone
Olanzapine • Zyprexa
Phenobarbital • Solfoton
Phenytoin • Dilantin
Ramelteon • Rozerem
Rifampin • Rifadin
Risperidone • Risperdal
Sertraline • Zoloft
Tamoxifen • Nolvadex
Topiramate • Topamax
Valproic acid • Depakote, Depakene
Venlafaxine • Effexor
Verapamil • Verelan
Zolpidem • Ambien
Against the backdrop of an increasing opioid use epidemic and a marked acceleration of prescription opioid–related deaths,1,2 there has been an impetus to explore the usefulness of alternative and co-analgesic agents to assist patients with chronic pain. Preclinical studies employing animal-based models of human pain syndromes have demonstrated that cannabis and chemicals derived from cannabis extracts may mitigate several pain conditions.3
Because there are significant comorbidities between psychiatric disorders and chronic pain, psychiatrists are likely to care for patients with chronic pain. As the availability of and interest in cannabinoid-based medications (CBM) increases, psychiatrists will need to be apprised of the utility, adverse effects, and potential drug interactions of these agents.
The endocannabinoid system and cannabis receptors
The endogenous cannabinoid (endocannabinoid) system is abundantly present within the peripheral and central nervous systems. The first identified, and best studied, endocannabinoids are N-arachidonoyl-ethanolamine (AEA; anandamide) and 2-arachidonoylglycerol (2-AG).4 Unlike typical neurotransmitters, AEA and 2-AG are not stored within vesicles within presynaptic neuron axons. Instead, they are lipophilic molecules produced on demand, synthesized from phospholipids (ie, arachidonic acid derivatives) at the membranes of post-synaptic neurons, and released into the synapse directly.5
Acting as retrograde messengers, the endocannabinoids traverse the synapse, binding to receptors located on the axons of the presynaptic neuron. Two receptors—CB1 and CB2—have been most extensively studied and characterized.6,7 These receptors couple to Gi/o-proteins to inhibit adenylate cyclase, decreasing Ca2+ conductance and increasing K+ conductance.8 Once activated, cannabinoid receptors modulate neurotransmitter release from presynaptic axon terminals. Evidence points to a similar retrograde signaling between neurons and glial cells. Shortly after receptor activation, the endocannabinoids are deactivated by the actions of a transporter mechanism and enzyme degradation.9,10
The endocannabinoid system and pain transmission
Cannabinoid receptors are present in pain transmission circuits spanning from the peripheral sensory nerve endings (from which pain signals originate) to the spinal cord and supraspinal regions within the brain.11-14 CB1 receptors are abundantly present within the CNS, including regions involved in pain transmission. Binding to CB1 receptors, endocannabinoids modulate neurotransmission that impacts pain transmission centrally. Endocannabinoids can also indirectly modulate opiate and N-methyl-
By contrast, CB2 receptors are predominantly localized to peripheral tissues and immune cells, although there has been some discovery of their presence within the CNS (eg, on microglia). Endocannabinoid activation of CB2 receptors is thought to modulate the activity of peripheral afferent pain fibers and immune-mediated neuroinflammatory processes—such as inhibition of prostaglandin synthesis and mast cell degranulation—that can precipitate and maintain chronic pain states.16-18
Evidence garnered from preclinical (animal) studies points to the role of the endocannabinoid system in modulating normal pain transmission (see Manzanares et al3 for details). These studies offer a putative basis for understanding how exogenous cannabinoid congeners might serve to ameliorate pain transmission in pathophysiologic states, including chronic pain.
Continue to: Cannabinoid-based medications
Cannabinoid-based medications
Marijuana contains multiple components (cannabinoids). The most extensively studied are delta-9-tetrahydrocannabinol (THC) and cannabidiol (CBD). Because it predominantly binds CB1 receptors centrally, THC is the major psychoactive component of cannabis; it promotes sleep and appetite, influences anxiety, and produces the “high” associated with cannabis use. By contrast, CBD weakly binds CB1 and thus exerts minimal or no psychoactive effects.19
Cannabinoid absorption, metabolism, bioavailability, and clinical effects vary depending on the formulation and method of administration (Table 1).20-22 THC and CBD content and potency in inhaled cannabis can vary significantly depending on the strains of the cannabis plant and manner of cultivation.23 To standardize approaches for administering cannabinoids in clinical trials and for clinical use, researchers have developed pharmaceutical analogs that contain extracted chemicals or synthetic chemicals similar to THC and/or CBD.
In this article, CBM refers to smoked/vaporized herbal cannabis as well as pharmaceutical cannabis analogs. Table 2 summarizes the characteristics of CBM commonly used in studies investigating their use for managing pain conditions.
CBM for chronic pain
The literature base examining the role of CBM for managing chronic nonmalignant and malignant pain of varying etiologies is rapidly expanding. Randomized controlled trials (RCTs) have focused on inhaled/smoked products and related cannabinoid medications, some of which are FDA-approved (Table 2).
A multitude of other cannabinoid-based products are currently commercially available to consumers, including tincture and oil-based products; over-the-counter CBD products; and several other formulations of CBM (eg, edible and suppository products). Because such products are not standardized or quality-controlled,24 RCTs have not assessed their efficacy for mitigating pain. Consequently, the findings summarized in this article do not address the utility of these agents.
Continue to: CBM for non-cancer pain
CBM for non-cancer pain
Neuropathic pain. Randomized controlled trials have assessed the pain-mitigating effects of various CBM, including inhaled cannabis, synthetic THC, plant-extracted CBD, and a THC/CBD spray. Studies have shown that inhaled/vaporized cannabis can produce short-term pain reduction in patients with chronic neuropathic pain of diverse etiologies, including diabetes mellitus-, HIV-, trauma-, and medication-induced neuropathies.22,25,26 Similar beneficial effects have been observed with the use of cannabis analogues (eg, nabiximols).25,26-29
Meta-analyses and systematic reviews have determined that most of these RCTs were of low-to-moderate quality.26,30 Meta-analyses have revealed divergent and conflicting results because of differences in the inclusion and exclusion criteria used to select RCTs for analysis and differences in the standards with which the quality of evidence were determined.25,30
Overall, the benefit of CBM for mitigating neuropathic pain is promising, but the effectiveness may not be robust.30,31 Several noteworthy caveats limit the interpretation of the results of these RCTs:
- due to the small sample sizes and brief durations of study, questions remain regarding the extent to which effects are generalizable, whether the benefits are sustained, and whether adverse effects emerge over time with continued use
- most RCTs evaluated inhaled (herbal) cannabis and nabiximols; there is little data on the effectiveness of other CBM formulations25,26,30
- the pain-mitigating effects of CBM were usually compared with those of placebo; the comparative efficacy against agents commonly used to treat neuropathic pain remains largely unexamined
- these RCTs typically compared mean pain severity score differences between cannabis-treated and placebo groups using standard subjective rating scales of pain intensity, such as the Numerical Rating Scale or Visual Analogue Scale. Customarily, the pain literature has used a 30% or 50% reduction in pain severity from baseline as an indicator of significant clinical improvement.32,33 The RCTs of CBM for neuropathic pain rarely used this standard, which makes it unclear whether CBM results in clinically significant pain reductions30
- indirect measures of effectiveness (ie, whether using CBM reduces the need for opioids or other analgesics to manage pain) were seldom reported in these RCTs.
Due to these limitations, clinical guidelines and systematic reviews consider CBM as a third- or fourth-line therapy for patients experiencing chronic neuropathic pain for whom conventional agents such as anticonvulsants and antidepressants have failed.34,35
Spasticity in multiple sclerosis (MS). Several RCTs have assessed the use of CBM for MS-related spasticity, although few were deemed to be high quality. Nabiximols and synthetic THC were effective in managing spasticity and reducing pain severity associated with muscle spasms.36 Generally, investigations revealed that CBM were associated with improvements in subjective measures of spasticity, but these were not born out in clinical, objective measures.26,37 The efficacy of smoked cannabis was uncertain.37 The existing literature on CBM for MS-related spasticity does not address dosing, duration of effects, tolerability, or comparative effectiveness against conventional anti-spasm medications.36,37
Continue to: Other chronic pain conditions
Other chronic pain conditions. CBM have also been studied for their usefulness in several other noncancer chronic conditions, including Crohn’s disease, inflammatory bowel disease, fibromyalgia, and other rheumatologic pain conditions.22,31,38-40 However, a solid foundation of empirical work to inform their utility for managing pain in these conditions is lacking.
CBM for cancer pain
Anecdotal evidence suggests that inhaled cannabis has promising pain-mitigating effects in patients with advanced cancer.41-43 There is a dearth of high-quality RCTs assessing the utility of CBM in patients with cancer pain.43-45 The types of CBM used and dosing strategies varied across RCTs, which makes it difficult to infer how best to treat patients with cancer pain. The agents studied included nabiximols, THC spray, and synthetic THC capsules.43-45 Although some studies have demonstrated that synthetic THC and nabiximols have potential for reducing subjective pain ratings compared with placebo,46,47 these results were inconsistent.46,48 Oromucosal nabiximols did not appear to confer any additional analgesic benefit in patients who were already prescribed opioids.31,45
The benefit of CBM for mitigating cancer pain is promising, but it remains difficult to know how to position the use of CBM in managing cancer pain. Limitations in the cancer literature include:
- the RCTs addressing CBM use for cancer pain were often brief, which raises questions about the long-term effectiveness and adverse effects of these agents
- tolerability and dosing limits encountered due to adverse effects were seldom reported43,45
- the types of cancer pain that patients had were often quite diverse. The small sample sizes and the heterogeneity of conditions included in these RCTs limit the ability to determine whether pain-mitigating effects might vary according to type of cancer-related pain.31,45
Despite these limitations, some clinical guidelines and systematic reviews have suggested that CBM have some role in addressing refractory malignant pain conditions.49
Psychiatric considerations related to CBM
As of November 2020, 36 states had legalized the use of cannabis for medical purposes, typically for painful conditions, despite the fact that empirical evidence to support their efficacy is mixed.50 In light of recent changes in both the legal and popular attitudes regarding cannabis, the implications of legalizing CBM remains to be seen. For example, some research suggests that adults with pain are vulnerable to frequent nonmedical cannabis use and/or cannabis use disorder.51 Although well-intended, the legalization of CBM use might represent society’s next misstep in the quest to address the suffering of patients with chronic pain. Some evidence shows that cannabis use and cannabis use disorders increase in states that have legalized medical marijuana.52,53 Psychiatrists will be on the front lines of addressing any potential consequences arising from the use of CBM for treating pain.
Continue to: Psychiatric disorders and CBM
Psychiatric disorders and CBM. The psychological impact of CBM use among patients enduring chronic pain can include sedation, cognitive/attention disturbance, and fatigue. These adverse effects can limit the utility of such agents.22,29,45
Contraindications for CBM use, and conditions for which CBM ought to be used with caution, are listed in Table 354,55.The safety of CBM, particularly in patients with chronic pain and psychiatric disorders, has not been examined. Patients with psychiatric disorders may be poor candidates for medical cannabis. Epidemiologic data suggest that recreational cannabis use is positively associated both cross-sectionally and prospectively with psychotic spectrum disorders, depressive symptoms, and anxiety symptoms, including panic disorder.56 Psychotic reactions have also been associated with CBM (dronabinol and nabilone).57 Cannabis use also has been associated with an earlier onset of, and lower remission rates of, symptoms associated with bipolar disorder.58,59 Consequently, patients who have been diagnosed with or are at risk for developing any of the aforementioned conditions may not be suitable candidates for CBM. If CBM are used, patients should be closely monitored for the emergence/exacerbation of psychiatric symptoms. The frequency and extent of follow-up is not clear, however. Because of its reduced propensity to produce psychoactive effects, CBD may be safer than THC for managing pain in individuals who have or are vulnerable to developing psychiatric disorders.
There is a lack of evidence to support the use of CBM for treating primary depressive disorders, general anxiety disorder, posttraumatic stress disorder, or psychosis.60,61 Very low-quality evidence suggests that CBM could lead to a small improvement in anxiety among individuals with noncancer pain and MS.60 However, interpreting causality is complicated. It is plausible that, for some patients, subjective improvement in pain severity may be related to reduced anxiety.62 Conversely, it is equally plausible that reductions in emotional distress may reduce the propensity to attend to, and thus magnify, pain severity. In the latter case, the indirect impact of reducing pain by modifying emotional distress can be impacted by the type and dose of CBM used. For example, low concentrations of THC produce anxiolytic effects, but high concentrations may be anxiety-provoking.63,64
Several potential pharmacokinetic drug interactions may arise between herbal cannabis or CBM and other medications (Table 465,66). THC and CBD are both metabolized by cytochrome P450 (CYP) 2C19 and 3A4.65,66 In addition, THC is also metabolized by CYP2C9. Medications that inhibit or induce these enzymes can increase or decrease the bioavailability of THC and CBD.67
Simultaneously, cannabinoids can impact the bioavailability of co-prescribed medications (Table 566,68). Although such CYP enzyme interactions remain a theoretical possibility, it is uncertain whether significant perturbations in plasma concentrations (and clinical effects) have been encountered with prescription medications when co-administered with CBM.69 Nonetheless, patients receiving CBM should be closely monitored for their response to prescribed medications.70
Continue to: Potential CYP enzyme interactions...
Potential CYP enzyme interactions aside, clinicians need to consider the additive effects that may occur when CBM are combined with sympathomimetic agents (eg, tachycardia, hypertension); CNS depressants such as alcohol, benzodiazepines, and opioids (eg, drowsiness, ataxia); or anticholinergics (eg, tachycardia, confusion).71 Inhaled herbal cannabis contains mutagens and can result in lung damage, exacerbations of chronic bronchitis, and certain types of cancer.54,72 Co-prescribing benzodiazepines may be contraindicated in light of their effects on respiratory rate and effort.
The THC contained in CBM produces hormonal effects (ie, significantly increases plasma levels of ghrelin and leptin and decreases peptide YY levels)73 that affect appetite and can produce weight gain. This may be problematic for patients receiving psychoactive medications associated with increased risk of weight gain and dyslipidemia. Because of the association between cannabis use and motor vehicle accidents, patients whose jobs require them to drive or operate industrial equipment may not be ideal candidates for CBM, especially if such patients also consume alcohol or are prescribed benzodiazepines and/or sedative hypnotics.74 Lastly, due to their lipophilicity, cannabinoids cross the placental barrier and can be found in breast milk75 and therefore can affect pregnancy outcomes and neurodevelopment.
Bottom Line
The popularity of cannabinoid-based medications (CBM) for the treatment of chronic pain conditions is growing, but the interest in their use may be outpacing the evidence supporting their analgesic benefits. High-quality, well-controlled randomized controlled trials are needed to decipher whether, and to what extent, these agents can be positioned in chronic pain management. Because psychiatrists are likely to encounter patients considering, or receiving, CBM, they must be aware of the potential benefits, risks, and adverse effects of such treatments.
Related Resources
- Joshi KG. Cannabis-derived compounds: what you need to know. Current Psychiatry. 2020;19(10):64-65. doi:10.12788/ cp.0050
- Gupta S, Phalen T, Gupta S. Medical marijuana: do the benefits outweigh the risks? Current Psychiatry. 2018; 17(1):34-41.
Drug Brand Names
Ajulemic acid • Anabasum
Alprazolam • Xanax
Amitriptyline • Elavil
Aripiprazole • Abilify, Abilify Maintena
Buspirone • BuSpar
Cannabidiol • Epidiolex
Carbamazepine • Tegretol, Equetro
Cimetidine • Tagamet HB
Citalopram • Celexa
Clopidogrel • Plavix
Clozapine • Clozaril
Cyclosporine • Neoral, Sandimmune
Dronabinol • Marinol, Syndros
Duloxetine • Cymbalta
Fluoxetine • Prozac
Fluvoxamine • Luvox
Haloperidol • Haldol
Imipramine • Tofranil
Ketoconazole • Nizoral AD
Losartan • Cozaar
Midazolam • Versed
Mirtazapine • Remeron
Nabilone • Cesamet
Nabiximols • Sativex
Nefazodone • Serzone
Olanzapine • Zyprexa
Phenobarbital • Solfoton
Phenytoin • Dilantin
Ramelteon • Rozerem
Rifampin • Rifadin
Risperidone • Risperdal
Sertraline • Zoloft
Tamoxifen • Nolvadex
Topiramate • Topamax
Valproic acid • Depakote, Depakene
Venlafaxine • Effexor
Verapamil • Verelan
Zolpidem • Ambien
1. Okie S. A floor of opioids, a rising tide of deaths. N Engl J Med. 2010;363(21):1981-1985. doi:10.1056/NEJMp1011512
2. Powell D, Pacula RL, Taylor E. How increasing medical access to opioids contributes to the opioid epidemic: evidence from Medicare Part D. J Health Econ. 2020;71:102286. doi: 10.1016/j.jhealeco.2019.102286
3. Manzanares J, Julian MD, Carrascosa A. Role of the cannabinoid system in pain control and therapeutic implications for the management of acute and chronic pain episodes. Curr Neuropharmacol. 2006;4(3):239-257. doi: 10.2174/157015906778019527
4. Zou S, Kumar U. Cannabinoid receptors and the endocannabinoid system: signaling and function in the central nervous system. Int J Mol Sci. 2018;19(3):833. doi: 10.3390/ijms19030833
5. Huang WJ, Chen WW, Zhang X. Endocannabinoid system: role in depression, reward and pain control (Review). Mol Med Rep. 2016;14(4):2899-2903. doi:10.3892/mmr.2016.5585
6. Mechoulam R, Ben-Shabat S, Hanus L, et al. Identification of an endogenous 2-monoglyceride, present in canine gut, that binds to cannabinoid receptors. Biochem Pharmacol. 1995;50(1):83-90. doi:10.1016/0006-2952(95)00109-d
7. Walker JM, Krey JF, Chu CJ, et al. Endocannabinoids and related fatty acid derivatives in pain modulation. Chem Phys Lipids. 2002;121(1-2):159-172. doi: 10.1016/s0009-3084(02)00152-4
8. Howlett AC. Efficacy in CB1 receptor-mediated signal transduction. Br J Pharmacol. 2004;142(8):1209-1218. doi: 10.1038/sj.bjp.0705881
9. Giuffrida A, Beltramo M, Piomelli D. Mechanisms of endocannabinoid inactivation, biochemistry and pharmacology. J Pharmacol Exp Ther. 2001;298:7-14.
10. Piomelli D, Beltramo M, Giuffrida A, et al. Endogenous cannabinoid signaling. Neurobiol Dis. 1998;5(6 Pt B):462-473. doi: 10.1006/nbdi.1998.0221
11. Eggan SM, Lewis DA. Immunocytochemical distribution of the cannabinoid CB1 receptor in the primate neocortex: a regional and laminar analysis. Cereb Cortex. 2007;17(1):175-191. doi: 10.1093/cercor/bhj136
12. Jennings EA, Vaughan CW, Christie MJ. Cannabinoid actions on rat superficial medullary dorsal horn neurons in vitro. J Physiol. 2001;534(Pt 3):805-812. doi: 10.1111/j.1469-7793.2001.00805.x
13. Vaughan CW, Connor M, Bagley EE, et al. Actions of cannabinoids on membrane properties and synaptic transmission in rat periaqueductal gray neurons in vitro. Mol Pharmacol. 2000;57(2):288-295.
14. Vaughan CW, McGregor IS, Christie MJ. Cannabinoid receptor activation inhibits GABAergic neurotransmission in rostral ventromedial medulla neurons in vitro. Br J Pharmacol. 1999;127(4):935-940. doi: 10.1038/sj.bjp.0702636
15. Raichlen DA, Foster AD, Gerdeman GI, et al. Wired to run: exercise-induced endocannabinoid signaling in humans and cursorial mammals with implications for the “runner’s high.” J Exp Biol. 2012;215(Pt 8):1331-1336. doi: 10.1242/jeb.063677
16. Beltrano M. Cannabinoid type 2 receptor as a target for chronic pain. Mini Rev Chem. 2009;234:253-254.
17. Ibrahim MM, Deng H, Zvonok A, et al. Activation of CB2 cannabinoid receptors by AM1241 inhibits experimental neuropathic pain: pain inhibition by receptors not present in the CNS. Proc Natl Acad Sci U S A. 2003;100(18):10529-10533. doi: 10.1073/pnas.1834309100
18. Valenzano KJ, Tafessem L, Lee G, et al. Pharmacological and pharmacokinetic characterization of the cannabinoid receptor 2 agonist, GW405833, utilizing rodent models of acute and chronic pain, anxiety, ataxia and catalepsy. Neuropharmacology. 2005;48:658-672.
19. Pertwee RG, Howlett AC, Abood ME, et al. International union of basic and clinical pharmacology. LXXIX. Cannabinoid receptors and their ligands: beyond CB1 and CB2. Pharmacol Rev. 2010;62(4):588-631. doi: 10.1124/pr.110.003004
20. Carter GT, Weydt P, Kyashna-Tocha M, et al. Medicinal cannabis: rational guidelines for dosing. Drugs. 2004;7(5):464-470.
21. Huestis MA. Human cannabinoid pharmacokinetics. Chem Biodivers. 2007;4(8):1770-1804.
22. Johal H, Devji T, Chang Y, et al. cannabinoids in chronic non-cancer pain: a systematic review and meta-analysis. Clin Med Insights Arthritis Musculoskelet Disord. 2020;13:1179544120906461. doi: 10.1177/1179544120906461
23. Hillig KW, Mahlberg PG. A chemotaxonomic analysis of cannabinoid variation in Cannabis (Cannabaceae). Am J Bot. 2004;91(6):966-975. doi: 10.3732/ajb.91.6.966
24. Hazekamp A, Ware MA, Muller-Vahl KR, et al. The medicinal use of cannabis and cannabinoids--an international cross-sectional survey on administration forms. J Psychoactive Drugs. 2013;45(3):199-210. doi: 10.1080/02791072.2013.805976
25. Andreae MH, Carter GM, Shaparin N, et al. inhaled cannabis for chronic neuropathic pain: a meta-analysis of individual patient data. J Pain. 2015;16(12):1221-1232. doi: 10.1016/j.jpain.2015.07.009
26. Whiting PF, Wolff RF, Deshpande S, et al. Cannabinoids for medical use: a systematic review and meta-analysis. JAMA. 2015;313(24):2456-2473. doi: 10.1001/jama.2015.6358
27. Boychuk DG, Goddard G, Mauro G, et al. The effectiveness of cannabinoids in the management of chronic nonmalignant neuropathic pain: a systematic review. J Oral Facial Pain Headache. 2015;29(1):7-14. doi: 10.11607/ofph.1274
28. Lynch ME, Campbell F. Cannabinoids for treatment of chronic non-cancer pain; a systematic review of randomized trials. Br J Clin Pharmacol. 2011;72(5):735-744. doi: 10.1111/j.1365-2125.2011.03970.x
29. Stockings E, Campbell G, Hall WD, et al. Cannabis and cannabinoids for the treatment of people with chronic noncancer pain conditions: a systematic review and meta-analysis of controlled and observational studies. Pain. 2018;159(10):1932-1954. doi: 10.1097/j.pain.0000000000001293
30. Mücke M, Phillips T, Radbruch L, et al. Cannabis-based medicines for chronic neuropathic pain in adults. Cochrane Database Syst Rev. 2018;3(3):CD012182. doi: 10.1002/14651858.CD012182.pub2
31. Häuser W, Fitzcharles MA, Radbruch L, et al. Cannabinoids in pain management and palliative medicine. Dtsch Arztebl Int. 2017;114(38):627-634. doi: 10.3238/arztebl.2017.0627
32. Dworkin RH, Turk DC, Wyrwich KW, et al. Interpreting the clinical importance of treatment outcomes in chronic pain clinical trials: IMMPACT recommendations. J Pain. 2008;9(2):105-121. doi: 10.1016/j.jpain.2007.09.005
33. Farrar JT, Troxel AB, Stott C, et al. Validity, reliability, and clinical importance of change in a 0-10 numeric rating scale measure of spasticity: a post hoc analysis of a randomized, double-blind, placebo-controlled trial. Clin Ther. 2008;30(5):974-985. doi: 10.1016/j.clinthera.2008.05.011
34. Moulin D, Boulanger A, Clark AJ, et al. Pharmacological management of chronic neuropathic pain: revised consensus statement from the Canadian Pain Society. Pain Res Manag. 2014;19(6):328-335. doi: 10.1155/2014/754693
35. Petzke F, Enax-Krumova EK, Häuser W. Efficacy, tolerability and safety of cannabinoids for chronic neuropathic pain: a systematic review of randomized controlled studies. Schmerz. 2016;30(1):62-88. doi: 10.1007/s00482-015-0089-y
36. Rice J, Cameron M. Cannabinoids for treatment of MS symptoms: state of the evidence. Curr Neurol Neurosci Rep. 2018;18(8):50. doi: 10.1007/s11910-018-0859-x
37. Koppel BS, Brust JCM, Fife T, et al. Systematic review: efficacy and safety of medical marijuana in selected neurologic disorders. Report of the Guideline Development Subcommittee of the American Academy of Neurology. Neurology. 2014;82(17):1556-1563. doi: 10.1212/WNL.0000000000000363
38. Kafil TS, Nguyen TM, MacDonald JK, et al. Cannabis for the treatment of Crohn’s disease and ulcerative colitis: evidence from Cochrane Reviews. Inflamm Bowel Dis. 2020;26(4):502-509. doi: 10.1093/ibd/izz233
39. Katz-Talmor D, Katz I, Porat-Katz BS, et al. Cannabinoids for the treatment of rheumatic diseases - where do we stand? Nat Rev Rheumatol. 2018;14(8):488-498. doi: 10.1038/s41584-018-0025-5
40. Walitt B, Klose P, Fitzcharles MA, et al. Cannabinoids for fibromyalgia. Cochrane Database Syst Rev. 2016;7(7):CD011694. doi: 10.1002/14651858.CD011694.pub2
41. Bar-Lev Schleider L, Mechoulam R, Lederman V, et al. Prospective analysis of safety and efficacy of medical cannabis in large unselected population of patients with cancer. Eur J Intern Med. 2018;49:37‐43. doi: 10.1016/j.ejim.2018.01.023
42. Bennett M, Paice JA, Wallace M. Pain and opioids in cancer care: benefits, risks, and alternatives. Am Soc Clin Oncol Educ Book. 2017;37:705‐713. doi:10.1200/EDBK_180469
43. Blake A, Wan BA, Malek L, et al. A selective review of medical cannabis in cancer pain management. Ann Palliat Med. 2017;6(Suppl 2):5215-5222. doi: 10.21037/apm.2017.08.05
44. Aviram J, Samuelly-Lechtag G. Efficacy of cannabis-based medicines for pain management: a systematic review and meta-analysis of randomized controlled trials. Pain Physician. 2017;20(6):E755-E796.
45. Häuser W, Welsch P, Klose P, et al. Efficacy, tolerability and safety of cannabis-based medicines for cancer pain: a systematic review with meta-analysis of randomised controlled trials. Schmerz. 2019;33(5):424-436. doi: 10.1007/s00482-019-0373-3
46. Johnson JR, Burnell-Nugent M, Lossignol D, et al. Multicenter, double-blind, randomized, placebo-controlled, parallel-group study of the efficacy, safety, and tolerability of THC:CBD extract and THC extract in patients with intractable cancer-related pain. J Pain Symptom Manage 2010; 39:167-179.
47. Portenoy RK, Ganae-Motan ED, Allende S, et al. Nabiximols for opioid-treated cancer patients with poorly-controlled chronic pain: a randomized, placebo-controlled, graded-dose trial. J Pain. 2012;13(5):438-449. doi: 10.1016/j.jpain.2012.01.003
48. Lynch ME, Cesar-Rittenberg P, Hohmann AG. A double-blind, placebo-controlled, crossover pilot trial with extension using an oral mucosal cannabinoid extract for treatment of chemotherapy-induced neuropathic pain. J Pain Symptom Manage. 2014;47(1):166-173. doi: 10.1016/j.jpainsymman.2013.02.018
49. Kleckner AS, Kleckner IR, Kamen CS, et al. Opportunities for cannabis in supportive care in cancer. Ther Adv Med Oncol. 2019;11:1758835919866362. doi: 10.1177/1758835919866362
50. National Conference of State Legislatures (ncsl.org). State Medical Marijuana Laws. Accessed April 5, 2021. https://www.ncsl.org/research/health/state-medical-marijuana-laws.aspx
51. Hasin DS, Shmulewitz D, Cerda M, et al. US adults with pain, a group increasingly vulnerable to nonmedical cannabis use and cannabis use disorder: 2001-2002 and 2012-2013. Am J Psychiatry. 2020;177(7):611-618. doi: 10.1176/appi.ajp.2019.19030284
52. Hasin DS, Sarvet AL, Cerdá M, et al. US adult illicit cannabis use, cannabis use disorder, and medical marijuana laws: 1991-1992 to 2012-2013. JAMA Psychiatry. 2017;74(6):579-588. doi: 10.1001/jamapsychiatry.2017.0724
53. National Institute on Drug Abuse. Illicit cannabis use and use disorders increase in states with medical marijuana laws. April 26, 2017. Accessed October 24, 2020. https://archives.drugabuse.gov/news-events/news-releases/2017/04/illicit-cannabis-use-use-disorders-increase-in-states-medical-marijuana-laws
54. National Academies of Sciences, Engineering, and Medicine. The health effects of cannabis and cannabinoids: the current state of evidence and recommendations for research. The National Academies Press; 2017. https://doi.org/10.17226/24625
55. Stanford M. Physician recommended marijuana: contraindications & standards of care. A review of the literature. Accessed July 7, 2020. http://drneurosci.com/MedicalMarijuanaStandardsofCare.pdf
56. Repp K, Raich A. Marijuana and health: a comprehensive review of 20 years of research. Washington County Oregon Department of Health and Human Services. 2014. Accessed April 8, 2021. https://www.co.washington.or.us/CAO/upload/HHSmarijuana-review.pdf
57. Parmar JR, Forrest BD, Freeman RA. Medical marijuana patient counseling points for health care professionals based on trends in the medical uses, efficacy, and adverse effects of cannabis-based pharmaceutical drugs. Res Social Adm Pharm. 2016;12(4):638-654. doi: 10.1016/j.sapharm.2015.09.002.
58. Leite RT, Nogueira Sde O, do Nascimento JP, et al. The use of cannabis as a predictor of early onset of bipolar disorder and suicide attempts. Neural Plast. 2015;2015:434127. doi: 10.1155/2015/43412
59. Kim SW, Dodd S, Berk L, et al. Impact of cannabis use on long-term remission in bipolar I and schizoaffective disorder. Psychiatry Investig. 2015;12(3):349-355. doi: 10.4306/pi.2015.12.3.349
60. Black N, Stockings E, Campbell G, et al. Cannabinoids for the treatment of mental disorders and symptoms of mental disorders: a systematic review and meta-analysis. Lancet Psychiatry. 2019;6(12):995-1010.
61. Wilkinson ST, Radhakrishnan R, D’Souza DC. A systematic review of the evidence for medical marijuana in psychiatric indications. J Clin Psychiatry. 2016;77(8):1050-1064. doi: 10.4088/JCP.15r10036.
62. Woolf CJ, American College of Physicians. American Physiological Society Pain: moving from symptom control toward mechanism-specific pharmacologic management. Ann Intern Med. 2004;140(6):441-451.
63. Crippa JA, Zuardi AW, Martín-Santos R, et al. Cannabis and anxiety: a critical review of the evidence. Hum Psychopharmacol. 2009;24(7):515‐523. doi: 10.1002/hup.1048
64. Sachs J, McGlade E, Yurgelun-Todd D. Safety and toxicology of cannabinoids. Neurotherapeutics. 2015;12(4):735‐746. doi: 10.1007/s13311-015-0380-8
65. Antoniou T, Bodkin J, Ho JMW. Drug interactions with cannabinoids. CMAJ. 2020;2;192:E206. doi: 10.1503/cmaj.191097
66. Brown JD. Potential adverse drug events with tetrahydrocannabinol (THC) due to drug-drug interactions. J Clin Med. 2020;9(4):919. doi: 10.3390/jcm9040919.
67. Maida V, Daeninck P. A user’s guide to cannabinoid therapy in oncology. Curr Oncol. 2016;23(6):398-406. doi: http://dx.doi.org/10.3747/co.23.3487
68. Stout SM, Cimino NM. Exogenous cannabinoids as substrates, inhibitors, and inducers of human drug metabolizing enzymes: a systematic review. Drug Metab Rev. 2014;46(1):86-95. doi: 10.3109/03602532.2013.849268
69. Abrams DI. Integrating cannabis into clinical cancer care. Curr Oncol. 2016;23(52):S8-S14.
70. Alsherbiny MA, Li CG. Medicinal cannabis—potential drug interactions. Medicines. 2018;6(1):3. doi: 10.3390/medicines6010003
71. Lucas CJ, Galettis P, Schneider J. The pharmacokinetics and the pharmacodynamics of cannabinoids. Br J Clin Pharmacol. 2018;84:2477-2482.
72. Ghasemiesfe M, Barrow B, Leonard S, et al. Association between marijuana use and risk of cancer: a systematic review and meta-analysis. JAMA Netw Open. 2019;2(11):e1916318. doi: 10.1001/jamanetworkopen.2019.16318
73. Riggs PK, Vaida F, Rossi SS, et al. A pilot study of the effects of cannabis on appetite hormones in HIV-infected adult men. Brain Res. 2012;1431:46-52. doi: 10.1016/j.brainres.2011.11.001
74. Asbridge M, Hayden JA, Cartwright JL. Acute cannabis consumption and motor vehicle collision risk: systematic review of observational studies and meta-analysis. BMJ. 2012;344:e536. doi: 10.1136/bmj.e536
75. Carlier J, Huestis MA, Zaami S, et al. Monitoring perinatal exposure to cannabis and synthetic cannabinoids. Ther Drug Monit. 2020;42(2):194-204.
1. Okie S. A floor of opioids, a rising tide of deaths. N Engl J Med. 2010;363(21):1981-1985. doi:10.1056/NEJMp1011512
2. Powell D, Pacula RL, Taylor E. How increasing medical access to opioids contributes to the opioid epidemic: evidence from Medicare Part D. J Health Econ. 2020;71:102286. doi: 10.1016/j.jhealeco.2019.102286
3. Manzanares J, Julian MD, Carrascosa A. Role of the cannabinoid system in pain control and therapeutic implications for the management of acute and chronic pain episodes. Curr Neuropharmacol. 2006;4(3):239-257. doi: 10.2174/157015906778019527
4. Zou S, Kumar U. Cannabinoid receptors and the endocannabinoid system: signaling and function in the central nervous system. Int J Mol Sci. 2018;19(3):833. doi: 10.3390/ijms19030833
5. Huang WJ, Chen WW, Zhang X. Endocannabinoid system: role in depression, reward and pain control (Review). Mol Med Rep. 2016;14(4):2899-2903. doi:10.3892/mmr.2016.5585
6. Mechoulam R, Ben-Shabat S, Hanus L, et al. Identification of an endogenous 2-monoglyceride, present in canine gut, that binds to cannabinoid receptors. Biochem Pharmacol. 1995;50(1):83-90. doi:10.1016/0006-2952(95)00109-d
7. Walker JM, Krey JF, Chu CJ, et al. Endocannabinoids and related fatty acid derivatives in pain modulation. Chem Phys Lipids. 2002;121(1-2):159-172. doi: 10.1016/s0009-3084(02)00152-4
8. Howlett AC. Efficacy in CB1 receptor-mediated signal transduction. Br J Pharmacol. 2004;142(8):1209-1218. doi: 10.1038/sj.bjp.0705881
9. Giuffrida A, Beltramo M, Piomelli D. Mechanisms of endocannabinoid inactivation, biochemistry and pharmacology. J Pharmacol Exp Ther. 2001;298:7-14.
10. Piomelli D, Beltramo M, Giuffrida A, et al. Endogenous cannabinoid signaling. Neurobiol Dis. 1998;5(6 Pt B):462-473. doi: 10.1006/nbdi.1998.0221
11. Eggan SM, Lewis DA. Immunocytochemical distribution of the cannabinoid CB1 receptor in the primate neocortex: a regional and laminar analysis. Cereb Cortex. 2007;17(1):175-191. doi: 10.1093/cercor/bhj136
12. Jennings EA, Vaughan CW, Christie MJ. Cannabinoid actions on rat superficial medullary dorsal horn neurons in vitro. J Physiol. 2001;534(Pt 3):805-812. doi: 10.1111/j.1469-7793.2001.00805.x
13. Vaughan CW, Connor M, Bagley EE, et al. Actions of cannabinoids on membrane properties and synaptic transmission in rat periaqueductal gray neurons in vitro. Mol Pharmacol. 2000;57(2):288-295.
14. Vaughan CW, McGregor IS, Christie MJ. Cannabinoid receptor activation inhibits GABAergic neurotransmission in rostral ventromedial medulla neurons in vitro. Br J Pharmacol. 1999;127(4):935-940. doi: 10.1038/sj.bjp.0702636
15. Raichlen DA, Foster AD, Gerdeman GI, et al. Wired to run: exercise-induced endocannabinoid signaling in humans and cursorial mammals with implications for the “runner’s high.” J Exp Biol. 2012;215(Pt 8):1331-1336. doi: 10.1242/jeb.063677
16. Beltrano M. Cannabinoid type 2 receptor as a target for chronic pain. Mini Rev Chem. 2009;234:253-254.
17. Ibrahim MM, Deng H, Zvonok A, et al. Activation of CB2 cannabinoid receptors by AM1241 inhibits experimental neuropathic pain: pain inhibition by receptors not present in the CNS. Proc Natl Acad Sci U S A. 2003;100(18):10529-10533. doi: 10.1073/pnas.1834309100
18. Valenzano KJ, Tafessem L, Lee G, et al. Pharmacological and pharmacokinetic characterization of the cannabinoid receptor 2 agonist, GW405833, utilizing rodent models of acute and chronic pain, anxiety, ataxia and catalepsy. Neuropharmacology. 2005;48:658-672.
19. Pertwee RG, Howlett AC, Abood ME, et al. International union of basic and clinical pharmacology. LXXIX. Cannabinoid receptors and their ligands: beyond CB1 and CB2. Pharmacol Rev. 2010;62(4):588-631. doi: 10.1124/pr.110.003004
20. Carter GT, Weydt P, Kyashna-Tocha M, et al. Medicinal cannabis: rational guidelines for dosing. Drugs. 2004;7(5):464-470.
21. Huestis MA. Human cannabinoid pharmacokinetics. Chem Biodivers. 2007;4(8):1770-1804.
22. Johal H, Devji T, Chang Y, et al. cannabinoids in chronic non-cancer pain: a systematic review and meta-analysis. Clin Med Insights Arthritis Musculoskelet Disord. 2020;13:1179544120906461. doi: 10.1177/1179544120906461
23. Hillig KW, Mahlberg PG. A chemotaxonomic analysis of cannabinoid variation in Cannabis (Cannabaceae). Am J Bot. 2004;91(6):966-975. doi: 10.3732/ajb.91.6.966
24. Hazekamp A, Ware MA, Muller-Vahl KR, et al. The medicinal use of cannabis and cannabinoids--an international cross-sectional survey on administration forms. J Psychoactive Drugs. 2013;45(3):199-210. doi: 10.1080/02791072.2013.805976
25. Andreae MH, Carter GM, Shaparin N, et al. inhaled cannabis for chronic neuropathic pain: a meta-analysis of individual patient data. J Pain. 2015;16(12):1221-1232. doi: 10.1016/j.jpain.2015.07.009
26. Whiting PF, Wolff RF, Deshpande S, et al. Cannabinoids for medical use: a systematic review and meta-analysis. JAMA. 2015;313(24):2456-2473. doi: 10.1001/jama.2015.6358
27. Boychuk DG, Goddard G, Mauro G, et al. The effectiveness of cannabinoids in the management of chronic nonmalignant neuropathic pain: a systematic review. J Oral Facial Pain Headache. 2015;29(1):7-14. doi: 10.11607/ofph.1274
28. Lynch ME, Campbell F. Cannabinoids for treatment of chronic non-cancer pain; a systematic review of randomized trials. Br J Clin Pharmacol. 2011;72(5):735-744. doi: 10.1111/j.1365-2125.2011.03970.x
29. Stockings E, Campbell G, Hall WD, et al. Cannabis and cannabinoids for the treatment of people with chronic noncancer pain conditions: a systematic review and meta-analysis of controlled and observational studies. Pain. 2018;159(10):1932-1954. doi: 10.1097/j.pain.0000000000001293
30. Mücke M, Phillips T, Radbruch L, et al. Cannabis-based medicines for chronic neuropathic pain in adults. Cochrane Database Syst Rev. 2018;3(3):CD012182. doi: 10.1002/14651858.CD012182.pub2
31. Häuser W, Fitzcharles MA, Radbruch L, et al. Cannabinoids in pain management and palliative medicine. Dtsch Arztebl Int. 2017;114(38):627-634. doi: 10.3238/arztebl.2017.0627
32. Dworkin RH, Turk DC, Wyrwich KW, et al. Interpreting the clinical importance of treatment outcomes in chronic pain clinical trials: IMMPACT recommendations. J Pain. 2008;9(2):105-121. doi: 10.1016/j.jpain.2007.09.005
33. Farrar JT, Troxel AB, Stott C, et al. Validity, reliability, and clinical importance of change in a 0-10 numeric rating scale measure of spasticity: a post hoc analysis of a randomized, double-blind, placebo-controlled trial. Clin Ther. 2008;30(5):974-985. doi: 10.1016/j.clinthera.2008.05.011
34. Moulin D, Boulanger A, Clark AJ, et al. Pharmacological management of chronic neuropathic pain: revised consensus statement from the Canadian Pain Society. Pain Res Manag. 2014;19(6):328-335. doi: 10.1155/2014/754693
35. Petzke F, Enax-Krumova EK, Häuser W. Efficacy, tolerability and safety of cannabinoids for chronic neuropathic pain: a systematic review of randomized controlled studies. Schmerz. 2016;30(1):62-88. doi: 10.1007/s00482-015-0089-y
36. Rice J, Cameron M. Cannabinoids for treatment of MS symptoms: state of the evidence. Curr Neurol Neurosci Rep. 2018;18(8):50. doi: 10.1007/s11910-018-0859-x
37. Koppel BS, Brust JCM, Fife T, et al. Systematic review: efficacy and safety of medical marijuana in selected neurologic disorders. Report of the Guideline Development Subcommittee of the American Academy of Neurology. Neurology. 2014;82(17):1556-1563. doi: 10.1212/WNL.0000000000000363
38. Kafil TS, Nguyen TM, MacDonald JK, et al. Cannabis for the treatment of Crohn’s disease and ulcerative colitis: evidence from Cochrane Reviews. Inflamm Bowel Dis. 2020;26(4):502-509. doi: 10.1093/ibd/izz233
39. Katz-Talmor D, Katz I, Porat-Katz BS, et al. Cannabinoids for the treatment of rheumatic diseases - where do we stand? Nat Rev Rheumatol. 2018;14(8):488-498. doi: 10.1038/s41584-018-0025-5
40. Walitt B, Klose P, Fitzcharles MA, et al. Cannabinoids for fibromyalgia. Cochrane Database Syst Rev. 2016;7(7):CD011694. doi: 10.1002/14651858.CD011694.pub2
41. Bar-Lev Schleider L, Mechoulam R, Lederman V, et al. Prospective analysis of safety and efficacy of medical cannabis in large unselected population of patients with cancer. Eur J Intern Med. 2018;49:37‐43. doi: 10.1016/j.ejim.2018.01.023
42. Bennett M, Paice JA, Wallace M. Pain and opioids in cancer care: benefits, risks, and alternatives. Am Soc Clin Oncol Educ Book. 2017;37:705‐713. doi:10.1200/EDBK_180469
43. Blake A, Wan BA, Malek L, et al. A selective review of medical cannabis in cancer pain management. Ann Palliat Med. 2017;6(Suppl 2):5215-5222. doi: 10.21037/apm.2017.08.05
44. Aviram J, Samuelly-Lechtag G. Efficacy of cannabis-based medicines for pain management: a systematic review and meta-analysis of randomized controlled trials. Pain Physician. 2017;20(6):E755-E796.
45. Häuser W, Welsch P, Klose P, et al. Efficacy, tolerability and safety of cannabis-based medicines for cancer pain: a systematic review with meta-analysis of randomised controlled trials. Schmerz. 2019;33(5):424-436. doi: 10.1007/s00482-019-0373-3
46. Johnson JR, Burnell-Nugent M, Lossignol D, et al. Multicenter, double-blind, randomized, placebo-controlled, parallel-group study of the efficacy, safety, and tolerability of THC:CBD extract and THC extract in patients with intractable cancer-related pain. J Pain Symptom Manage 2010; 39:167-179.
47. Portenoy RK, Ganae-Motan ED, Allende S, et al. Nabiximols for opioid-treated cancer patients with poorly-controlled chronic pain: a randomized, placebo-controlled, graded-dose trial. J Pain. 2012;13(5):438-449. doi: 10.1016/j.jpain.2012.01.003
48. Lynch ME, Cesar-Rittenberg P, Hohmann AG. A double-blind, placebo-controlled, crossover pilot trial with extension using an oral mucosal cannabinoid extract for treatment of chemotherapy-induced neuropathic pain. J Pain Symptom Manage. 2014;47(1):166-173. doi: 10.1016/j.jpainsymman.2013.02.018
49. Kleckner AS, Kleckner IR, Kamen CS, et al. Opportunities for cannabis in supportive care in cancer. Ther Adv Med Oncol. 2019;11:1758835919866362. doi: 10.1177/1758835919866362
50. National Conference of State Legislatures (ncsl.org). State Medical Marijuana Laws. Accessed April 5, 2021. https://www.ncsl.org/research/health/state-medical-marijuana-laws.aspx
51. Hasin DS, Shmulewitz D, Cerda M, et al. US adults with pain, a group increasingly vulnerable to nonmedical cannabis use and cannabis use disorder: 2001-2002 and 2012-2013. Am J Psychiatry. 2020;177(7):611-618. doi: 10.1176/appi.ajp.2019.19030284
52. Hasin DS, Sarvet AL, Cerdá M, et al. US adult illicit cannabis use, cannabis use disorder, and medical marijuana laws: 1991-1992 to 2012-2013. JAMA Psychiatry. 2017;74(6):579-588. doi: 10.1001/jamapsychiatry.2017.0724
53. National Institute on Drug Abuse. Illicit cannabis use and use disorders increase in states with medical marijuana laws. April 26, 2017. Accessed October 24, 2020. https://archives.drugabuse.gov/news-events/news-releases/2017/04/illicit-cannabis-use-use-disorders-increase-in-states-medical-marijuana-laws
54. National Academies of Sciences, Engineering, and Medicine. The health effects of cannabis and cannabinoids: the current state of evidence and recommendations for research. The National Academies Press; 2017. https://doi.org/10.17226/24625
55. Stanford M. Physician recommended marijuana: contraindications & standards of care. A review of the literature. Accessed July 7, 2020. http://drneurosci.com/MedicalMarijuanaStandardsofCare.pdf
56. Repp K, Raich A. Marijuana and health: a comprehensive review of 20 years of research. Washington County Oregon Department of Health and Human Services. 2014. Accessed April 8, 2021. https://www.co.washington.or.us/CAO/upload/HHSmarijuana-review.pdf
57. Parmar JR, Forrest BD, Freeman RA. Medical marijuana patient counseling points for health care professionals based on trends in the medical uses, efficacy, and adverse effects of cannabis-based pharmaceutical drugs. Res Social Adm Pharm. 2016;12(4):638-654. doi: 10.1016/j.sapharm.2015.09.002.
58. Leite RT, Nogueira Sde O, do Nascimento JP, et al. The use of cannabis as a predictor of early onset of bipolar disorder and suicide attempts. Neural Plast. 2015;2015:434127. doi: 10.1155/2015/43412
59. Kim SW, Dodd S, Berk L, et al. Impact of cannabis use on long-term remission in bipolar I and schizoaffective disorder. Psychiatry Investig. 2015;12(3):349-355. doi: 10.4306/pi.2015.12.3.349
60. Black N, Stockings E, Campbell G, et al. Cannabinoids for the treatment of mental disorders and symptoms of mental disorders: a systematic review and meta-analysis. Lancet Psychiatry. 2019;6(12):995-1010.
61. Wilkinson ST, Radhakrishnan R, D’Souza DC. A systematic review of the evidence for medical marijuana in psychiatric indications. J Clin Psychiatry. 2016;77(8):1050-1064. doi: 10.4088/JCP.15r10036.
62. Woolf CJ, American College of Physicians. American Physiological Society Pain: moving from symptom control toward mechanism-specific pharmacologic management. Ann Intern Med. 2004;140(6):441-451.
63. Crippa JA, Zuardi AW, Martín-Santos R, et al. Cannabis and anxiety: a critical review of the evidence. Hum Psychopharmacol. 2009;24(7):515‐523. doi: 10.1002/hup.1048
64. Sachs J, McGlade E, Yurgelun-Todd D. Safety and toxicology of cannabinoids. Neurotherapeutics. 2015;12(4):735‐746. doi: 10.1007/s13311-015-0380-8
65. Antoniou T, Bodkin J, Ho JMW. Drug interactions with cannabinoids. CMAJ. 2020;2;192:E206. doi: 10.1503/cmaj.191097
66. Brown JD. Potential adverse drug events with tetrahydrocannabinol (THC) due to drug-drug interactions. J Clin Med. 2020;9(4):919. doi: 10.3390/jcm9040919.
67. Maida V, Daeninck P. A user’s guide to cannabinoid therapy in oncology. Curr Oncol. 2016;23(6):398-406. doi: http://dx.doi.org/10.3747/co.23.3487
68. Stout SM, Cimino NM. Exogenous cannabinoids as substrates, inhibitors, and inducers of human drug metabolizing enzymes: a systematic review. Drug Metab Rev. 2014;46(1):86-95. doi: 10.3109/03602532.2013.849268
69. Abrams DI. Integrating cannabis into clinical cancer care. Curr Oncol. 2016;23(52):S8-S14.
70. Alsherbiny MA, Li CG. Medicinal cannabis—potential drug interactions. Medicines. 2018;6(1):3. doi: 10.3390/medicines6010003
71. Lucas CJ, Galettis P, Schneider J. The pharmacokinetics and the pharmacodynamics of cannabinoids. Br J Clin Pharmacol. 2018;84:2477-2482.
72. Ghasemiesfe M, Barrow B, Leonard S, et al. Association between marijuana use and risk of cancer: a systematic review and meta-analysis. JAMA Netw Open. 2019;2(11):e1916318. doi: 10.1001/jamanetworkopen.2019.16318
73. Riggs PK, Vaida F, Rossi SS, et al. A pilot study of the effects of cannabis on appetite hormones in HIV-infected adult men. Brain Res. 2012;1431:46-52. doi: 10.1016/j.brainres.2011.11.001
74. Asbridge M, Hayden JA, Cartwright JL. Acute cannabis consumption and motor vehicle collision risk: systematic review of observational studies and meta-analysis. BMJ. 2012;344:e536. doi: 10.1136/bmj.e536
75. Carlier J, Huestis MA, Zaami S, et al. Monitoring perinatal exposure to cannabis and synthetic cannabinoids. Ther Drug Monit. 2020;42(2):194-204.
Marijuana vaping more common among Hispanic teens
Hispanic adolescents were more likely to use e-cigarettes to vape marijuana than were their Black and White counterparts in 2020, according to a recent study conducted by the Centers for Disease Control and Prevention and published in JAMA Pediatrics.
Researchers found that 25.6% of Hispanic students reported vaping marijuana, compared to 19.4% of Black students and 18.2% of White students. The study, which is an analysis of 2017, 2018, and 2020 results from the National Youth Tobacco Survey, also revealed that increases in this recreational practice occurred among all racial and ethnic groups within those 3 years, with Hispanic students having the largest percent increase, 11.6%, followed by Black students at 8.8% and White students at 7.4%.
“The initial motivation [to do this study] was to gain a better understanding of the prevalence of use of marijuana in e-cigarettes among youth, particularly given the context of the 2019 outbreak of e-cigarette, or vaping, product use–associated lung injury (EVALI),” study author Christina Vaughan Watson, DrPH, health scientist at the CDC’s National Center for Chronic Disease Prevention and Health Promotion, said in an interview.
The findings could help clinicians and physicians understand demographic variations among marijuana vapers and help inform targeted interventions for specific populations.
“Understanding demographic variations among those who are using marijuana in e-cigarettes can help inform evidenced-based interventions that may resonate with specific populations,” Dr. Watson explained.
Margaret Thew, DNP, medical director of adolescent medicine at Children’s Wisconsin in Milwaukee, who was not involved in the study, said in an interview that the findings were “eye opening” and revealed a pattern she hasn’t seen before in her adolescent clinic.
“I would have thought African-American or non-Hispanic Blacks would’ve been a higher group of use, because when we screen kids that’s what we tend to get from the population we see here,” Ms. Thew said.
Ms. Thew said the findings also had made her reconsider her clinic’s approach to screening adolescents for marijuana use as well as address possible language barriers.
“We are probably missing access to some of the kids that we may need to seek out,” she explained. “I also thought it sends a good message that we need to direct some of our education probably a little differently, especially if it’s a Hispanic population and English may not be the primary language.”
Dr. Watson said more research is needed to assess why differences in marijuana use in e-cigarettes exist among youth.
Marijuana use in e-cigarettes has become increasingly popular among U.S. teens, with one in five students in grades 10 and 12 reporting vaping marijuana within the past year in a 2019 study conducted by the National Institute on Drug Abuse.
Dr. Watson and colleagues also found statistically significant increases in vaping marijuana, with 19.5% of students reporting smoking marijuana via e-cigarettes in 2020, compared to 11.1% of them vaping the drug in 2017. They believe the rise in marijuana vaping among youth may be attributed to states increasingly legalizing adult marijuana sales, which could impact ease of access and social acceptance.
Ms. Thew believes the rise in marijuana vaping among youth can be attributed to the legalization of marijuana, which may send “a message to adolescents that it must be safe for them to use,” as well as the increasing popularity of e-cigarettes.
In fact, as of April 2021, marijuana is legal for adults in 16 states and the District of Columbia. Meanwhile, medical marijuana is legal in 36 states, according to the National Conference of State Legislatures.
“I mean, there’s just definitely been a lot more use of [e-cigarettes]. Vaping and things like that definitely took off between 2019 and 2020,” Ms. Thew explained. “And I think marijuana use in itself is going up tremendously, I think more kids who would have used alcohol in the past use weed.”
Although public attitudes toward marijuana have relaxed, previous studies have linked it to memory dysfunction, as well as long-term cognitive effects that can interfere with perception of time and motor function. However, studies also have shown that cannabis use can combat age-related cognitive decline and help with pain reduction.
However, when it comes to adolescents, Dr. Watson and colleagues said e-cigarette use among youth and young adults is unsafe, regardless of the substances used in these products, including marijuana. Furthermore, they said marijuana use can lead to higher risks of more problematic use later in life, adding that evidence-based strategies to reduce marijuana use in e-cigarettes are important for protecting young people.
The study author and experts disclosed no relevant financial relationships.
Hispanic adolescents were more likely to use e-cigarettes to vape marijuana than were their Black and White counterparts in 2020, according to a recent study conducted by the Centers for Disease Control and Prevention and published in JAMA Pediatrics.
Researchers found that 25.6% of Hispanic students reported vaping marijuana, compared to 19.4% of Black students and 18.2% of White students. The study, which is an analysis of 2017, 2018, and 2020 results from the National Youth Tobacco Survey, also revealed that increases in this recreational practice occurred among all racial and ethnic groups within those 3 years, with Hispanic students having the largest percent increase, 11.6%, followed by Black students at 8.8% and White students at 7.4%.
“The initial motivation [to do this study] was to gain a better understanding of the prevalence of use of marijuana in e-cigarettes among youth, particularly given the context of the 2019 outbreak of e-cigarette, or vaping, product use–associated lung injury (EVALI),” study author Christina Vaughan Watson, DrPH, health scientist at the CDC’s National Center for Chronic Disease Prevention and Health Promotion, said in an interview.
The findings could help clinicians and physicians understand demographic variations among marijuana vapers and help inform targeted interventions for specific populations.
“Understanding demographic variations among those who are using marijuana in e-cigarettes can help inform evidenced-based interventions that may resonate with specific populations,” Dr. Watson explained.
Margaret Thew, DNP, medical director of adolescent medicine at Children’s Wisconsin in Milwaukee, who was not involved in the study, said in an interview that the findings were “eye opening” and revealed a pattern she hasn’t seen before in her adolescent clinic.
“I would have thought African-American or non-Hispanic Blacks would’ve been a higher group of use, because when we screen kids that’s what we tend to get from the population we see here,” Ms. Thew said.
Ms. Thew said the findings also had made her reconsider her clinic’s approach to screening adolescents for marijuana use as well as address possible language barriers.
“We are probably missing access to some of the kids that we may need to seek out,” she explained. “I also thought it sends a good message that we need to direct some of our education probably a little differently, especially if it’s a Hispanic population and English may not be the primary language.”
Dr. Watson said more research is needed to assess why differences in marijuana use in e-cigarettes exist among youth.
Marijuana use in e-cigarettes has become increasingly popular among U.S. teens, with one in five students in grades 10 and 12 reporting vaping marijuana within the past year in a 2019 study conducted by the National Institute on Drug Abuse.
Dr. Watson and colleagues also found statistically significant increases in vaping marijuana, with 19.5% of students reporting smoking marijuana via e-cigarettes in 2020, compared to 11.1% of them vaping the drug in 2017. They believe the rise in marijuana vaping among youth may be attributed to states increasingly legalizing adult marijuana sales, which could impact ease of access and social acceptance.
Ms. Thew believes the rise in marijuana vaping among youth can be attributed to the legalization of marijuana, which may send “a message to adolescents that it must be safe for them to use,” as well as the increasing popularity of e-cigarettes.
In fact, as of April 2021, marijuana is legal for adults in 16 states and the District of Columbia. Meanwhile, medical marijuana is legal in 36 states, according to the National Conference of State Legislatures.
“I mean, there’s just definitely been a lot more use of [e-cigarettes]. Vaping and things like that definitely took off between 2019 and 2020,” Ms. Thew explained. “And I think marijuana use in itself is going up tremendously, I think more kids who would have used alcohol in the past use weed.”
Although public attitudes toward marijuana have relaxed, previous studies have linked it to memory dysfunction, as well as long-term cognitive effects that can interfere with perception of time and motor function. However, studies also have shown that cannabis use can combat age-related cognitive decline and help with pain reduction.
However, when it comes to adolescents, Dr. Watson and colleagues said e-cigarette use among youth and young adults is unsafe, regardless of the substances used in these products, including marijuana. Furthermore, they said marijuana use can lead to higher risks of more problematic use later in life, adding that evidence-based strategies to reduce marijuana use in e-cigarettes are important for protecting young people.
The study author and experts disclosed no relevant financial relationships.
Hispanic adolescents were more likely to use e-cigarettes to vape marijuana than were their Black and White counterparts in 2020, according to a recent study conducted by the Centers for Disease Control and Prevention and published in JAMA Pediatrics.
Researchers found that 25.6% of Hispanic students reported vaping marijuana, compared to 19.4% of Black students and 18.2% of White students. The study, which is an analysis of 2017, 2018, and 2020 results from the National Youth Tobacco Survey, also revealed that increases in this recreational practice occurred among all racial and ethnic groups within those 3 years, with Hispanic students having the largest percent increase, 11.6%, followed by Black students at 8.8% and White students at 7.4%.
“The initial motivation [to do this study] was to gain a better understanding of the prevalence of use of marijuana in e-cigarettes among youth, particularly given the context of the 2019 outbreak of e-cigarette, or vaping, product use–associated lung injury (EVALI),” study author Christina Vaughan Watson, DrPH, health scientist at the CDC’s National Center for Chronic Disease Prevention and Health Promotion, said in an interview.
The findings could help clinicians and physicians understand demographic variations among marijuana vapers and help inform targeted interventions for specific populations.
“Understanding demographic variations among those who are using marijuana in e-cigarettes can help inform evidenced-based interventions that may resonate with specific populations,” Dr. Watson explained.
Margaret Thew, DNP, medical director of adolescent medicine at Children’s Wisconsin in Milwaukee, who was not involved in the study, said in an interview that the findings were “eye opening” and revealed a pattern she hasn’t seen before in her adolescent clinic.
“I would have thought African-American or non-Hispanic Blacks would’ve been a higher group of use, because when we screen kids that’s what we tend to get from the population we see here,” Ms. Thew said.
Ms. Thew said the findings also had made her reconsider her clinic’s approach to screening adolescents for marijuana use as well as address possible language barriers.
“We are probably missing access to some of the kids that we may need to seek out,” she explained. “I also thought it sends a good message that we need to direct some of our education probably a little differently, especially if it’s a Hispanic population and English may not be the primary language.”
Dr. Watson said more research is needed to assess why differences in marijuana use in e-cigarettes exist among youth.
Marijuana use in e-cigarettes has become increasingly popular among U.S. teens, with one in five students in grades 10 and 12 reporting vaping marijuana within the past year in a 2019 study conducted by the National Institute on Drug Abuse.
Dr. Watson and colleagues also found statistically significant increases in vaping marijuana, with 19.5% of students reporting smoking marijuana via e-cigarettes in 2020, compared to 11.1% of them vaping the drug in 2017. They believe the rise in marijuana vaping among youth may be attributed to states increasingly legalizing adult marijuana sales, which could impact ease of access and social acceptance.
Ms. Thew believes the rise in marijuana vaping among youth can be attributed to the legalization of marijuana, which may send “a message to adolescents that it must be safe for them to use,” as well as the increasing popularity of e-cigarettes.
In fact, as of April 2021, marijuana is legal for adults in 16 states and the District of Columbia. Meanwhile, medical marijuana is legal in 36 states, according to the National Conference of State Legislatures.
“I mean, there’s just definitely been a lot more use of [e-cigarettes]. Vaping and things like that definitely took off between 2019 and 2020,” Ms. Thew explained. “And I think marijuana use in itself is going up tremendously, I think more kids who would have used alcohol in the past use weed.”
Although public attitudes toward marijuana have relaxed, previous studies have linked it to memory dysfunction, as well as long-term cognitive effects that can interfere with perception of time and motor function. However, studies also have shown that cannabis use can combat age-related cognitive decline and help with pain reduction.
However, when it comes to adolescents, Dr. Watson and colleagues said e-cigarette use among youth and young adults is unsafe, regardless of the substances used in these products, including marijuana. Furthermore, they said marijuana use can lead to higher risks of more problematic use later in life, adding that evidence-based strategies to reduce marijuana use in e-cigarettes are important for protecting young people.
The study author and experts disclosed no relevant financial relationships.
Doctors more likely to prescribe opioids to COVID ‘long-haulers,’ raising addiction fears
COVID-19 survivors are at risk from a possible second pandemic, this time of opioid addiction, given the high rate of painkillers being prescribed to these patients, health experts say.
A new study in Nature found alarmingly high rates of opioid use among COVID survivors with lingering symptoms at Veterans Affairs facilities. About 10% of COVID survivors develop “long COVID,” struggling with often disabling health problems even 6 months or longer after a diagnosis.
For every 1,000 long-COVID patients, known as “long-haulers,” who were treated at a VA facility, doctors wrote nine more prescriptions for opioids than they otherwise would have, along with 22 additional prescriptions for benzodiazepines, which include Xanax and other addictive pills used to treat anxiety.
Although previous studies have found many COVID survivors experience persistent health problems, the new article is the first to show they’re using more addictive medications, said Ziyad Al-Aly, MD, the paper’s lead author.
He’s concerned that even an apparently small increase in the inappropriate use of addictive pain pills will lead to a resurgence of the prescription opioid crisis, given the large number of COVID survivors. More than 3 million of the 31 million Americans infected with COVID develop long-term symptoms, which can include fatigue, shortness of breath, depression, anxiety, and memory problems known as “brain fog.”
The new study also found many patients have significant muscle and bone pain.
The frequent use of opioids was surprising, given concerns about their potential for addiction, said Dr. Al-Aly, chief of research and education service at the VA St. Louis Health Care System.
“Physicians now are supposed to shy away from prescribing opioids,” said Dr. Al-Aly, who studied more than 73,000 patients in the VA system. When Dr. Al-Aly saw the number of opioids prescriptions, he said, he thought to himself: “Is this really happening all over again?”
Doctors need to act now, before “it’s too late to do something,” Dr. Al-Aly said. “We must act now and ensure that people are getting the care they need. We do not want this to balloon into a suicide crisis or another opioid epidemic.”
As more doctors became aware of their addictive potential, new opioid prescriptions fell, by more than half since 2012. But said Andrew Kolodny, MD, medical director of opioid policy research at Brandeis University, Waltham, Mass.
Some patients who became addicted to prescription painkillers switched to heroin, either because it was cheaper or because they could no longer obtain opioids from their doctors. Overdose deaths surged in recent years as drug dealers began spiking heroin with a powerful synthetic opioid called fentanyl.
More than 88,000 Americans died from overdoses during the 12 months ending in August 2020, according to the Centers for Disease Control and Prevention. Health experts now advise doctors to avoid prescribing opioids for long periods.
The new study “suggests to me that many clinicians still don’t get it,” Dr. Kolodny said. “Many clinicians are under the false impression that opioids are appropriate for chronic pain patients.”
Hospitalized COVID patients often receive a lot of medication to control pain and anxiety, especially in ICUs, said Greg Martin, MD, president of the Society of Critical Care Medicine. Patients placed on ventilators, for example, are often sedated to make them more comfortable.
Martin said he’s concerned by the study’s findings, which suggest patients are unnecessarily continuing medications after leaving the hospital.
“I worry that COVID-19 patients, especially those who are severely and critically ill, receive a lot of medications during the hospitalization, and because they have persistent symptoms, the medications are continued after hospital discharge,” Dr. Martin said.
While some COVID patients are experiencing muscle and bone pain for the first time, others say the illness has intensified their preexisting pain.
Rachael Sunshine Burnett has suffered from chronic pain in her back and feet for 20 years, ever since an accident at a warehouse where she once worked. But Ms. Burnett, who first was diagnosed with COVID in April 2020, said the pain soon became 10 times worse and spread to the area between her shoulders and spine. Although she was already taking long-acting OxyContin twice a day, her doctor prescribed an additional opioid called oxycodone, which relieves pain immediately. She was reinfected with COVID in December.
“It’s been a horrible, horrible year,” said Ms. Burnett, 43, of Coxsackie, N.Y.
Doctors should recognize that pain can be a part of long COVID, Dr. Martin said. “We need to find the proper nonnarcotic treatment for it, just like we do with other forms of chronic pain,” he said.
The CDC recommends a number of alternatives to opioids – from physical therapy to biofeedback, over-the-counter anti-inflammatories, antidepressants, and antiseizure drugs that also relieve nerve pain.
The country also needs an overall strategy to cope with the wave of post-COVID complications, Dr. Al-Aly said.
“It’s better to be prepared than to be caught off guard years from now, when doctors realize: ‘Oh, we have a resurgence in opioids,’ ” Dr. Al-Aly said.
Dr. Al-Aly noted that his study may not capture the full complexity of post-COVID patient needs. Although women make up the majority of long-COVID patients in most studies, most patients in the VA system are men.
The study of VA patients makes it “abundantly clear that we are not prepared to meet the needs of 3 million Americans with long COVID,” said Eric Topol, MD, founder and director of the Scripps Research Translational Institute in San Diego. “We desperately need an intervention that will effectively treat these individuals.”
Dr. Al-Aly said COVID survivors may need care for years.
“That’s going to be a huge, significant burden on the health care system,” Dr. Al-Aly said. “Long COVID will reverberate in the health system for years or even decades to come.”
Kaiser Health News is a nonprofit news service covering health issues. It is an editorially independent program of KFF (Kaiser Family Foundation), which is not affiliated with Kaiser Permanente.
COVID-19 survivors are at risk from a possible second pandemic, this time of opioid addiction, given the high rate of painkillers being prescribed to these patients, health experts say.
A new study in Nature found alarmingly high rates of opioid use among COVID survivors with lingering symptoms at Veterans Affairs facilities. About 10% of COVID survivors develop “long COVID,” struggling with often disabling health problems even 6 months or longer after a diagnosis.
For every 1,000 long-COVID patients, known as “long-haulers,” who were treated at a VA facility, doctors wrote nine more prescriptions for opioids than they otherwise would have, along with 22 additional prescriptions for benzodiazepines, which include Xanax and other addictive pills used to treat anxiety.
Although previous studies have found many COVID survivors experience persistent health problems, the new article is the first to show they’re using more addictive medications, said Ziyad Al-Aly, MD, the paper’s lead author.
He’s concerned that even an apparently small increase in the inappropriate use of addictive pain pills will lead to a resurgence of the prescription opioid crisis, given the large number of COVID survivors. More than 3 million of the 31 million Americans infected with COVID develop long-term symptoms, which can include fatigue, shortness of breath, depression, anxiety, and memory problems known as “brain fog.”
The new study also found many patients have significant muscle and bone pain.
The frequent use of opioids was surprising, given concerns about their potential for addiction, said Dr. Al-Aly, chief of research and education service at the VA St. Louis Health Care System.
“Physicians now are supposed to shy away from prescribing opioids,” said Dr. Al-Aly, who studied more than 73,000 patients in the VA system. When Dr. Al-Aly saw the number of opioids prescriptions, he said, he thought to himself: “Is this really happening all over again?”
Doctors need to act now, before “it’s too late to do something,” Dr. Al-Aly said. “We must act now and ensure that people are getting the care they need. We do not want this to balloon into a suicide crisis or another opioid epidemic.”
As more doctors became aware of their addictive potential, new opioid prescriptions fell, by more than half since 2012. But said Andrew Kolodny, MD, medical director of opioid policy research at Brandeis University, Waltham, Mass.
Some patients who became addicted to prescription painkillers switched to heroin, either because it was cheaper or because they could no longer obtain opioids from their doctors. Overdose deaths surged in recent years as drug dealers began spiking heroin with a powerful synthetic opioid called fentanyl.
More than 88,000 Americans died from overdoses during the 12 months ending in August 2020, according to the Centers for Disease Control and Prevention. Health experts now advise doctors to avoid prescribing opioids for long periods.
The new study “suggests to me that many clinicians still don’t get it,” Dr. Kolodny said. “Many clinicians are under the false impression that opioids are appropriate for chronic pain patients.”
Hospitalized COVID patients often receive a lot of medication to control pain and anxiety, especially in ICUs, said Greg Martin, MD, president of the Society of Critical Care Medicine. Patients placed on ventilators, for example, are often sedated to make them more comfortable.
Martin said he’s concerned by the study’s findings, which suggest patients are unnecessarily continuing medications after leaving the hospital.
“I worry that COVID-19 patients, especially those who are severely and critically ill, receive a lot of medications during the hospitalization, and because they have persistent symptoms, the medications are continued after hospital discharge,” Dr. Martin said.
While some COVID patients are experiencing muscle and bone pain for the first time, others say the illness has intensified their preexisting pain.
Rachael Sunshine Burnett has suffered from chronic pain in her back and feet for 20 years, ever since an accident at a warehouse where she once worked. But Ms. Burnett, who first was diagnosed with COVID in April 2020, said the pain soon became 10 times worse and spread to the area between her shoulders and spine. Although she was already taking long-acting OxyContin twice a day, her doctor prescribed an additional opioid called oxycodone, which relieves pain immediately. She was reinfected with COVID in December.
“It’s been a horrible, horrible year,” said Ms. Burnett, 43, of Coxsackie, N.Y.
Doctors should recognize that pain can be a part of long COVID, Dr. Martin said. “We need to find the proper nonnarcotic treatment for it, just like we do with other forms of chronic pain,” he said.
The CDC recommends a number of alternatives to opioids – from physical therapy to biofeedback, over-the-counter anti-inflammatories, antidepressants, and antiseizure drugs that also relieve nerve pain.
The country also needs an overall strategy to cope with the wave of post-COVID complications, Dr. Al-Aly said.
“It’s better to be prepared than to be caught off guard years from now, when doctors realize: ‘Oh, we have a resurgence in opioids,’ ” Dr. Al-Aly said.
Dr. Al-Aly noted that his study may not capture the full complexity of post-COVID patient needs. Although women make up the majority of long-COVID patients in most studies, most patients in the VA system are men.
The study of VA patients makes it “abundantly clear that we are not prepared to meet the needs of 3 million Americans with long COVID,” said Eric Topol, MD, founder and director of the Scripps Research Translational Institute in San Diego. “We desperately need an intervention that will effectively treat these individuals.”
Dr. Al-Aly said COVID survivors may need care for years.
“That’s going to be a huge, significant burden on the health care system,” Dr. Al-Aly said. “Long COVID will reverberate in the health system for years or even decades to come.”
Kaiser Health News is a nonprofit news service covering health issues. It is an editorially independent program of KFF (Kaiser Family Foundation), which is not affiliated with Kaiser Permanente.
COVID-19 survivors are at risk from a possible second pandemic, this time of opioid addiction, given the high rate of painkillers being prescribed to these patients, health experts say.
A new study in Nature found alarmingly high rates of opioid use among COVID survivors with lingering symptoms at Veterans Affairs facilities. About 10% of COVID survivors develop “long COVID,” struggling with often disabling health problems even 6 months or longer after a diagnosis.
For every 1,000 long-COVID patients, known as “long-haulers,” who were treated at a VA facility, doctors wrote nine more prescriptions for opioids than they otherwise would have, along with 22 additional prescriptions for benzodiazepines, which include Xanax and other addictive pills used to treat anxiety.
Although previous studies have found many COVID survivors experience persistent health problems, the new article is the first to show they’re using more addictive medications, said Ziyad Al-Aly, MD, the paper’s lead author.
He’s concerned that even an apparently small increase in the inappropriate use of addictive pain pills will lead to a resurgence of the prescription opioid crisis, given the large number of COVID survivors. More than 3 million of the 31 million Americans infected with COVID develop long-term symptoms, which can include fatigue, shortness of breath, depression, anxiety, and memory problems known as “brain fog.”
The new study also found many patients have significant muscle and bone pain.
The frequent use of opioids was surprising, given concerns about their potential for addiction, said Dr. Al-Aly, chief of research and education service at the VA St. Louis Health Care System.
“Physicians now are supposed to shy away from prescribing opioids,” said Dr. Al-Aly, who studied more than 73,000 patients in the VA system. When Dr. Al-Aly saw the number of opioids prescriptions, he said, he thought to himself: “Is this really happening all over again?”
Doctors need to act now, before “it’s too late to do something,” Dr. Al-Aly said. “We must act now and ensure that people are getting the care they need. We do not want this to balloon into a suicide crisis or another opioid epidemic.”
As more doctors became aware of their addictive potential, new opioid prescriptions fell, by more than half since 2012. But said Andrew Kolodny, MD, medical director of opioid policy research at Brandeis University, Waltham, Mass.
Some patients who became addicted to prescription painkillers switched to heroin, either because it was cheaper or because they could no longer obtain opioids from their doctors. Overdose deaths surged in recent years as drug dealers began spiking heroin with a powerful synthetic opioid called fentanyl.
More than 88,000 Americans died from overdoses during the 12 months ending in August 2020, according to the Centers for Disease Control and Prevention. Health experts now advise doctors to avoid prescribing opioids for long periods.
The new study “suggests to me that many clinicians still don’t get it,” Dr. Kolodny said. “Many clinicians are under the false impression that opioids are appropriate for chronic pain patients.”
Hospitalized COVID patients often receive a lot of medication to control pain and anxiety, especially in ICUs, said Greg Martin, MD, president of the Society of Critical Care Medicine. Patients placed on ventilators, for example, are often sedated to make them more comfortable.
Martin said he’s concerned by the study’s findings, which suggest patients are unnecessarily continuing medications after leaving the hospital.
“I worry that COVID-19 patients, especially those who are severely and critically ill, receive a lot of medications during the hospitalization, and because they have persistent symptoms, the medications are continued after hospital discharge,” Dr. Martin said.
While some COVID patients are experiencing muscle and bone pain for the first time, others say the illness has intensified their preexisting pain.
Rachael Sunshine Burnett has suffered from chronic pain in her back and feet for 20 years, ever since an accident at a warehouse where she once worked. But Ms. Burnett, who first was diagnosed with COVID in April 2020, said the pain soon became 10 times worse and spread to the area between her shoulders and spine. Although she was already taking long-acting OxyContin twice a day, her doctor prescribed an additional opioid called oxycodone, which relieves pain immediately. She was reinfected with COVID in December.
“It’s been a horrible, horrible year,” said Ms. Burnett, 43, of Coxsackie, N.Y.
Doctors should recognize that pain can be a part of long COVID, Dr. Martin said. “We need to find the proper nonnarcotic treatment for it, just like we do with other forms of chronic pain,” he said.
The CDC recommends a number of alternatives to opioids – from physical therapy to biofeedback, over-the-counter anti-inflammatories, antidepressants, and antiseizure drugs that also relieve nerve pain.
The country also needs an overall strategy to cope with the wave of post-COVID complications, Dr. Al-Aly said.
“It’s better to be prepared than to be caught off guard years from now, when doctors realize: ‘Oh, we have a resurgence in opioids,’ ” Dr. Al-Aly said.
Dr. Al-Aly noted that his study may not capture the full complexity of post-COVID patient needs. Although women make up the majority of long-COVID patients in most studies, most patients in the VA system are men.
The study of VA patients makes it “abundantly clear that we are not prepared to meet the needs of 3 million Americans with long COVID,” said Eric Topol, MD, founder and director of the Scripps Research Translational Institute in San Diego. “We desperately need an intervention that will effectively treat these individuals.”
Dr. Al-Aly said COVID survivors may need care for years.
“That’s going to be a huge, significant burden on the health care system,” Dr. Al-Aly said. “Long COVID will reverberate in the health system for years or even decades to come.”
Kaiser Health News is a nonprofit news service covering health issues. It is an editorially independent program of KFF (Kaiser Family Foundation), which is not affiliated with Kaiser Permanente.
Adolescent substance use and the COVID-19 pandemic
During the past year, adolescents, families, educators, and health care providers have had to press forward through myriad challenges and stressors with flexibility and adaptability. With appropriate concern, we ask ourselves how children and youth are coping emotionally with the unprecedented changes of the past year.
Adolescent substance use represents an important area of concern. What has happened during the pandemic? Has youth substance use increased or decreased? Has access to substances increased or decreased, has monitoring and support for at-risk youth increased or decreased?
The answers to these questions are mixed. If anything, the pandemic has highlighted the heterogeneity of adolescent substance use. Now is a key time for assessment, support, and conversation with teens and families.
Monitoring the Future (MTF), a nationally representative annual survey, has provided a broad perspective on trends of adolescent substance use for decades.1 The MTF data is usually collected from February to May and was cut short in 2020 because of school closures associated with the pandemic. The sample size, though still nationally representative, was about a quarter of the typical volume. Some of the data are encouraging, including a flattening out of previous years’ stark increase in vaping of both nicotine and cannabis products (though overall numbers remain alarmingly high). Other data are more concerning including a continued increase in misuse of cough medicine, amphetamines, and inhalants among the youngest cohort surveyed (eighth graders). However, these data were largely representative of prepandemic circumstances.
The COVID-19 pandemic has significantly affected risk and protective factors for teen drug and alcohol use. Most notably, it has had a widely observed negative impact on adolescent mental health, across multiple disease categories.2 In addition, the cancellation of in-person academic and extracurricular activities such as arts and athletics markedly increased unstructured time, a known associated factor for higher-risk activities including substance use. This has also led to decreased contact with many supportive adults such as teachers and coaches. On the other hand, some adolescents now have more time with supportive parents and caregivers, more meals together, and more supervision, all of which are associated with decreased likelihood of substance use disorders.
The highly variable reasons for substance use affect highly variable pandemic-related changes in use. Understanding the impetus for use is a good place to start conversation and can help providers assess risk of escalation during the pandemic. Some teens primarily use for social enhancement while others use as a means of coping with stress or to mask or escape negative emotions. Still others continue use because of physiological dependence, craving, and other symptoms consistent with use disorders.
Highlighting the heterogeneity of this issue, one study assessing use early in the pandemic showed a decrease in the percentage of teens who use substances but an increase in frequency of use for those who are using.3 Though expected, an increase in frequency of use by oneself as compared with peers was also notable. Using substances alone is associated with more severe use disorders, carries greater risk of overdose, and can increase shame and secrecy, further fueling use disorders.
The pandemic has thus represented a protective pause for some experimental or socially motivated substance-using teens who have experienced a period of abstinence even if not fully by choice. For others, it has represented an acute amplification of risk factors and use has accelerated. This latter group includes those whose use represents an effort to cope with depression, anxiety, and loneliness or for whom isolation at home represents less monitoring, increased access, and greater exposure to substances.
Over the past year, in the treatment of adolescents struggling with substance use, many clinicians have observed a sifting effect during these unprecedented social changes. Many youth, who no longer have access to substances, have found they can “take it or leave it”. Other youth have been observed engaging in additional risk or going to greater lengths to access substances and continue their use. For both groups and everyone in between, this is an important time for screening, clinical assessment, and support.
While anticipating further research and data regarding broad substance use trends, including MTF data from 2021, recognizing that the impact of the COVID-19 pandemic is individual, with marked differences from adolescent to adolescent, will help us continue to act now to assess this important area of adolescent health. The first step for primary care providers is unchanged: to routinely screen for and discuss substance use in clinical settings.
Two brief, validated, easily accessible screening tools are available for primary care settings. They can both be self-administered and take less than 2 minutes to complete. Screening, Brief Intervention and Referral to Treatment and the Brief Screener for Tobacco, Alcohol and other Drugs can both be used for youth aged 12-17 years.4,5 Both screens are available online at drugabuse.gov.6
Routine screening will normalize conversations about substance use and healthy choices, provide opportunities for positive reinforcement, identify adolescents at risk, increase comfort and competence in providing brief intervention, and expedite referrals for additional support and treatment.
A false assumption that a particular adolescent isn’t using substances creates a missed opportunity to offer guidance and treatment. An oft-overlooked opportunity is that of providing positive reinforcement for an adolescent who isn’t using any substances or experimenting at all. Positive reinforcement is a strong component of reinforcing health maintenance.
Parent guidance and family assessment will also be critical tools. Parents and caregivers play a primary role in substance use treatment for teens and have a contributory impact on risk through both genes and environment. Of note, research suggests a moderate overall increase in adult substance use during the pandemic, particularly substances that are widely available such as alcohol. Adolescents may thus have greater access and exposure to substance use. A remarkably high percentage, 42%, of substance-using teens surveyed early in the pandemic indicated that they were using substances with their parents.3 Parents, who have equally been challenged by the pandemic, may need guidance in balancing compassion and support for struggling youth, while setting appropriate limits and maintaining expectations of healthy activities.
Unprecedented change and uncertainty provide an opportunity to reassess risks and openly discuss substance use with youth and families. Even with much on our minds during the COVID-19 pandemic, we can maintain focus on this significant risk to adolescent health and wellness. Our efforts now, from screening to treatment for adolescent substance use should be reinforced rather than delayed.
Dr. Jackson is assistant professor of psychiatry at the University of Vermont, Burlington.
References
1. Monitoringthefuture.org
2. Jones EAK et al. Int J Environ Res Public Health, 2021;18(5):2470.
3. Dumas TM et al. J Adolesc Health, 2020;67(3):354-61.
4. Levy S et al. JAMA Pediatr. 2014;168(9):822-8.
5. Kelly SM et al. Pediatrics. 2014;133(5):819-26.
6. National Institute on Drug Abuse. Adolescent Substance Use Screening Tools. 2016 Apr 27. https://www.drugabuse.gov/nidamed-medical-health-professionals/screening-tools-prevention/screening-tools-adolescent-substance-use/adolescent-substance-use-screening-tools
During the past year, adolescents, families, educators, and health care providers have had to press forward through myriad challenges and stressors with flexibility and adaptability. With appropriate concern, we ask ourselves how children and youth are coping emotionally with the unprecedented changes of the past year.
Adolescent substance use represents an important area of concern. What has happened during the pandemic? Has youth substance use increased or decreased? Has access to substances increased or decreased, has monitoring and support for at-risk youth increased or decreased?
The answers to these questions are mixed. If anything, the pandemic has highlighted the heterogeneity of adolescent substance use. Now is a key time for assessment, support, and conversation with teens and families.
Monitoring the Future (MTF), a nationally representative annual survey, has provided a broad perspective on trends of adolescent substance use for decades.1 The MTF data is usually collected from February to May and was cut short in 2020 because of school closures associated with the pandemic. The sample size, though still nationally representative, was about a quarter of the typical volume. Some of the data are encouraging, including a flattening out of previous years’ stark increase in vaping of both nicotine and cannabis products (though overall numbers remain alarmingly high). Other data are more concerning including a continued increase in misuse of cough medicine, amphetamines, and inhalants among the youngest cohort surveyed (eighth graders). However, these data were largely representative of prepandemic circumstances.
The COVID-19 pandemic has significantly affected risk and protective factors for teen drug and alcohol use. Most notably, it has had a widely observed negative impact on adolescent mental health, across multiple disease categories.2 In addition, the cancellation of in-person academic and extracurricular activities such as arts and athletics markedly increased unstructured time, a known associated factor for higher-risk activities including substance use. This has also led to decreased contact with many supportive adults such as teachers and coaches. On the other hand, some adolescents now have more time with supportive parents and caregivers, more meals together, and more supervision, all of which are associated with decreased likelihood of substance use disorders.
The highly variable reasons for substance use affect highly variable pandemic-related changes in use. Understanding the impetus for use is a good place to start conversation and can help providers assess risk of escalation during the pandemic. Some teens primarily use for social enhancement while others use as a means of coping with stress or to mask or escape negative emotions. Still others continue use because of physiological dependence, craving, and other symptoms consistent with use disorders.
Highlighting the heterogeneity of this issue, one study assessing use early in the pandemic showed a decrease in the percentage of teens who use substances but an increase in frequency of use for those who are using.3 Though expected, an increase in frequency of use by oneself as compared with peers was also notable. Using substances alone is associated with more severe use disorders, carries greater risk of overdose, and can increase shame and secrecy, further fueling use disorders.
The pandemic has thus represented a protective pause for some experimental or socially motivated substance-using teens who have experienced a period of abstinence even if not fully by choice. For others, it has represented an acute amplification of risk factors and use has accelerated. This latter group includes those whose use represents an effort to cope with depression, anxiety, and loneliness or for whom isolation at home represents less monitoring, increased access, and greater exposure to substances.
Over the past year, in the treatment of adolescents struggling with substance use, many clinicians have observed a sifting effect during these unprecedented social changes. Many youth, who no longer have access to substances, have found they can “take it or leave it”. Other youth have been observed engaging in additional risk or going to greater lengths to access substances and continue their use. For both groups and everyone in between, this is an important time for screening, clinical assessment, and support.
While anticipating further research and data regarding broad substance use trends, including MTF data from 2021, recognizing that the impact of the COVID-19 pandemic is individual, with marked differences from adolescent to adolescent, will help us continue to act now to assess this important area of adolescent health. The first step for primary care providers is unchanged: to routinely screen for and discuss substance use in clinical settings.
Two brief, validated, easily accessible screening tools are available for primary care settings. They can both be self-administered and take less than 2 minutes to complete. Screening, Brief Intervention and Referral to Treatment and the Brief Screener for Tobacco, Alcohol and other Drugs can both be used for youth aged 12-17 years.4,5 Both screens are available online at drugabuse.gov.6
Routine screening will normalize conversations about substance use and healthy choices, provide opportunities for positive reinforcement, identify adolescents at risk, increase comfort and competence in providing brief intervention, and expedite referrals for additional support and treatment.
A false assumption that a particular adolescent isn’t using substances creates a missed opportunity to offer guidance and treatment. An oft-overlooked opportunity is that of providing positive reinforcement for an adolescent who isn’t using any substances or experimenting at all. Positive reinforcement is a strong component of reinforcing health maintenance.
Parent guidance and family assessment will also be critical tools. Parents and caregivers play a primary role in substance use treatment for teens and have a contributory impact on risk through both genes and environment. Of note, research suggests a moderate overall increase in adult substance use during the pandemic, particularly substances that are widely available such as alcohol. Adolescents may thus have greater access and exposure to substance use. A remarkably high percentage, 42%, of substance-using teens surveyed early in the pandemic indicated that they were using substances with their parents.3 Parents, who have equally been challenged by the pandemic, may need guidance in balancing compassion and support for struggling youth, while setting appropriate limits and maintaining expectations of healthy activities.
Unprecedented change and uncertainty provide an opportunity to reassess risks and openly discuss substance use with youth and families. Even with much on our minds during the COVID-19 pandemic, we can maintain focus on this significant risk to adolescent health and wellness. Our efforts now, from screening to treatment for adolescent substance use should be reinforced rather than delayed.
Dr. Jackson is assistant professor of psychiatry at the University of Vermont, Burlington.
References
1. Monitoringthefuture.org
2. Jones EAK et al. Int J Environ Res Public Health, 2021;18(5):2470.
3. Dumas TM et al. J Adolesc Health, 2020;67(3):354-61.
4. Levy S et al. JAMA Pediatr. 2014;168(9):822-8.
5. Kelly SM et al. Pediatrics. 2014;133(5):819-26.
6. National Institute on Drug Abuse. Adolescent Substance Use Screening Tools. 2016 Apr 27. https://www.drugabuse.gov/nidamed-medical-health-professionals/screening-tools-prevention/screening-tools-adolescent-substance-use/adolescent-substance-use-screening-tools
During the past year, adolescents, families, educators, and health care providers have had to press forward through myriad challenges and stressors with flexibility and adaptability. With appropriate concern, we ask ourselves how children and youth are coping emotionally with the unprecedented changes of the past year.
Adolescent substance use represents an important area of concern. What has happened during the pandemic? Has youth substance use increased or decreased? Has access to substances increased or decreased, has monitoring and support for at-risk youth increased or decreased?
The answers to these questions are mixed. If anything, the pandemic has highlighted the heterogeneity of adolescent substance use. Now is a key time for assessment, support, and conversation with teens and families.
Monitoring the Future (MTF), a nationally representative annual survey, has provided a broad perspective on trends of adolescent substance use for decades.1 The MTF data is usually collected from February to May and was cut short in 2020 because of school closures associated with the pandemic. The sample size, though still nationally representative, was about a quarter of the typical volume. Some of the data are encouraging, including a flattening out of previous years’ stark increase in vaping of both nicotine and cannabis products (though overall numbers remain alarmingly high). Other data are more concerning including a continued increase in misuse of cough medicine, amphetamines, and inhalants among the youngest cohort surveyed (eighth graders). However, these data were largely representative of prepandemic circumstances.
The COVID-19 pandemic has significantly affected risk and protective factors for teen drug and alcohol use. Most notably, it has had a widely observed negative impact on adolescent mental health, across multiple disease categories.2 In addition, the cancellation of in-person academic and extracurricular activities such as arts and athletics markedly increased unstructured time, a known associated factor for higher-risk activities including substance use. This has also led to decreased contact with many supportive adults such as teachers and coaches. On the other hand, some adolescents now have more time with supportive parents and caregivers, more meals together, and more supervision, all of which are associated with decreased likelihood of substance use disorders.
The highly variable reasons for substance use affect highly variable pandemic-related changes in use. Understanding the impetus for use is a good place to start conversation and can help providers assess risk of escalation during the pandemic. Some teens primarily use for social enhancement while others use as a means of coping with stress or to mask or escape negative emotions. Still others continue use because of physiological dependence, craving, and other symptoms consistent with use disorders.
Highlighting the heterogeneity of this issue, one study assessing use early in the pandemic showed a decrease in the percentage of teens who use substances but an increase in frequency of use for those who are using.3 Though expected, an increase in frequency of use by oneself as compared with peers was also notable. Using substances alone is associated with more severe use disorders, carries greater risk of overdose, and can increase shame and secrecy, further fueling use disorders.
The pandemic has thus represented a protective pause for some experimental or socially motivated substance-using teens who have experienced a period of abstinence even if not fully by choice. For others, it has represented an acute amplification of risk factors and use has accelerated. This latter group includes those whose use represents an effort to cope with depression, anxiety, and loneliness or for whom isolation at home represents less monitoring, increased access, and greater exposure to substances.
Over the past year, in the treatment of adolescents struggling with substance use, many clinicians have observed a sifting effect during these unprecedented social changes. Many youth, who no longer have access to substances, have found they can “take it or leave it”. Other youth have been observed engaging in additional risk or going to greater lengths to access substances and continue their use. For both groups and everyone in between, this is an important time for screening, clinical assessment, and support.
While anticipating further research and data regarding broad substance use trends, including MTF data from 2021, recognizing that the impact of the COVID-19 pandemic is individual, with marked differences from adolescent to adolescent, will help us continue to act now to assess this important area of adolescent health. The first step for primary care providers is unchanged: to routinely screen for and discuss substance use in clinical settings.
Two brief, validated, easily accessible screening tools are available for primary care settings. They can both be self-administered and take less than 2 minutes to complete. Screening, Brief Intervention and Referral to Treatment and the Brief Screener for Tobacco, Alcohol and other Drugs can both be used for youth aged 12-17 years.4,5 Both screens are available online at drugabuse.gov.6
Routine screening will normalize conversations about substance use and healthy choices, provide opportunities for positive reinforcement, identify adolescents at risk, increase comfort and competence in providing brief intervention, and expedite referrals for additional support and treatment.
A false assumption that a particular adolescent isn’t using substances creates a missed opportunity to offer guidance and treatment. An oft-overlooked opportunity is that of providing positive reinforcement for an adolescent who isn’t using any substances or experimenting at all. Positive reinforcement is a strong component of reinforcing health maintenance.
Parent guidance and family assessment will also be critical tools. Parents and caregivers play a primary role in substance use treatment for teens and have a contributory impact on risk through both genes and environment. Of note, research suggests a moderate overall increase in adult substance use during the pandemic, particularly substances that are widely available such as alcohol. Adolescents may thus have greater access and exposure to substance use. A remarkably high percentage, 42%, of substance-using teens surveyed early in the pandemic indicated that they were using substances with their parents.3 Parents, who have equally been challenged by the pandemic, may need guidance in balancing compassion and support for struggling youth, while setting appropriate limits and maintaining expectations of healthy activities.
Unprecedented change and uncertainty provide an opportunity to reassess risks and openly discuss substance use with youth and families. Even with much on our minds during the COVID-19 pandemic, we can maintain focus on this significant risk to adolescent health and wellness. Our efforts now, from screening to treatment for adolescent substance use should be reinforced rather than delayed.
Dr. Jackson is assistant professor of psychiatry at the University of Vermont, Burlington.
References
1. Monitoringthefuture.org
2. Jones EAK et al. Int J Environ Res Public Health, 2021;18(5):2470.
3. Dumas TM et al. J Adolesc Health, 2020;67(3):354-61.
4. Levy S et al. JAMA Pediatr. 2014;168(9):822-8.
5. Kelly SM et al. Pediatrics. 2014;133(5):819-26.
6. National Institute on Drug Abuse. Adolescent Substance Use Screening Tools. 2016 Apr 27. https://www.drugabuse.gov/nidamed-medical-health-professionals/screening-tools-prevention/screening-tools-adolescent-substance-use/adolescent-substance-use-screening-tools
A thoughtful approach to drug screening and addiction
Reading the excellent article on urine drug screening by Drs. Hayes and Fox reminds me of 2 important aspects of primary care: (1) Diagnosing and treating patients with drug addiction is an important service we provide, and (2) interpreting laboratory tests requires training, skill, and clinical judgment.
Drs. Hayes and Fox describe the proper use of urine drug testing in the management of patients for whom we prescribe opioids, whether for chronic pain or for addiction treatment. Combining a review of the literature with their own professional experience treating these patients, Drs. Hayes and Fox highlight the potential pitfalls in interpreting urine drug screening results and admonish us to use good clinical judgment in applying those results to patient care. They emphasize the need to avoid racial bias and blaming the patient.
This article is very timely because, amidst the COVID-19 pandemic, the opioid epidemic has continued unabated. The most recent data from the National Center for Health Statistics shows that the estimated number of opioid overdose deaths increased by a whopping 32%, from 47,772 for the 1-year period ending August 2019 to 62,972 for the 1-year period ending August 2020.1 Although this increase began in fall 2019, there can be little doubt that the COVID-19 pandemic is partly responsible. A positive sign, however, is that opioid prescribing in the United States is trending downward, reaching its lowest level in 14 years in 2019.2 In fact, use of cheap street fentanyl, rather than prescription drugs, accounts for nearly all of the increase in opioid overdose deaths.1
Despite this positive news, the number of deaths associated with opioid use remains sobering. The statistics continue to underscore the fact that there simply are not enough addiction treatment centers to manage all of those who need and want help. All primary care physicians are eligible to prescribe suboxone to treat patients with opioid addiction—a treatment that can be highly effective in reducing the use of street opioids and, therefore, reducing deaths from overdose. Fewer than 10% of primary care physicians prescribed suboxone in 2017.3 I hope that more of you will take the required training and become involved in assisting your patients who struggle with opioid addiction.
1. National Center for Health Statistics. Provisional drug overdose death counts. Updated March 17, 2021. Accessed March 22, 2021. www.cdc.gov/nchs/nvss/vsrr/drug-overdose-data.htm
2. CDC. US opioid dispensing rate maps. Updated December 7, 2020. Accessed March 22, 2021. www.cdc.gov/drugoverdose/maps/rxrate-maps.html
3. McBain RK, Dick A, Sorbero M, et al. Growth and distribution of buprenorphine-waivered providers in the United States, 2007-2017. Ann Intern Med. 2020;172:504-506.
Reading the excellent article on urine drug screening by Drs. Hayes and Fox reminds me of 2 important aspects of primary care: (1) Diagnosing and treating patients with drug addiction is an important service we provide, and (2) interpreting laboratory tests requires training, skill, and clinical judgment.
Drs. Hayes and Fox describe the proper use of urine drug testing in the management of patients for whom we prescribe opioids, whether for chronic pain or for addiction treatment. Combining a review of the literature with their own professional experience treating these patients, Drs. Hayes and Fox highlight the potential pitfalls in interpreting urine drug screening results and admonish us to use good clinical judgment in applying those results to patient care. They emphasize the need to avoid racial bias and blaming the patient.
This article is very timely because, amidst the COVID-19 pandemic, the opioid epidemic has continued unabated. The most recent data from the National Center for Health Statistics shows that the estimated number of opioid overdose deaths increased by a whopping 32%, from 47,772 for the 1-year period ending August 2019 to 62,972 for the 1-year period ending August 2020.1 Although this increase began in fall 2019, there can be little doubt that the COVID-19 pandemic is partly responsible. A positive sign, however, is that opioid prescribing in the United States is trending downward, reaching its lowest level in 14 years in 2019.2 In fact, use of cheap street fentanyl, rather than prescription drugs, accounts for nearly all of the increase in opioid overdose deaths.1
Despite this positive news, the number of deaths associated with opioid use remains sobering. The statistics continue to underscore the fact that there simply are not enough addiction treatment centers to manage all of those who need and want help. All primary care physicians are eligible to prescribe suboxone to treat patients with opioid addiction—a treatment that can be highly effective in reducing the use of street opioids and, therefore, reducing deaths from overdose. Fewer than 10% of primary care physicians prescribed suboxone in 2017.3 I hope that more of you will take the required training and become involved in assisting your patients who struggle with opioid addiction.
Reading the excellent article on urine drug screening by Drs. Hayes and Fox reminds me of 2 important aspects of primary care: (1) Diagnosing and treating patients with drug addiction is an important service we provide, and (2) interpreting laboratory tests requires training, skill, and clinical judgment.
Drs. Hayes and Fox describe the proper use of urine drug testing in the management of patients for whom we prescribe opioids, whether for chronic pain or for addiction treatment. Combining a review of the literature with their own professional experience treating these patients, Drs. Hayes and Fox highlight the potential pitfalls in interpreting urine drug screening results and admonish us to use good clinical judgment in applying those results to patient care. They emphasize the need to avoid racial bias and blaming the patient.
This article is very timely because, amidst the COVID-19 pandemic, the opioid epidemic has continued unabated. The most recent data from the National Center for Health Statistics shows that the estimated number of opioid overdose deaths increased by a whopping 32%, from 47,772 for the 1-year period ending August 2019 to 62,972 for the 1-year period ending August 2020.1 Although this increase began in fall 2019, there can be little doubt that the COVID-19 pandemic is partly responsible. A positive sign, however, is that opioid prescribing in the United States is trending downward, reaching its lowest level in 14 years in 2019.2 In fact, use of cheap street fentanyl, rather than prescription drugs, accounts for nearly all of the increase in opioid overdose deaths.1
Despite this positive news, the number of deaths associated with opioid use remains sobering. The statistics continue to underscore the fact that there simply are not enough addiction treatment centers to manage all of those who need and want help. All primary care physicians are eligible to prescribe suboxone to treat patients with opioid addiction—a treatment that can be highly effective in reducing the use of street opioids and, therefore, reducing deaths from overdose. Fewer than 10% of primary care physicians prescribed suboxone in 2017.3 I hope that more of you will take the required training and become involved in assisting your patients who struggle with opioid addiction.
1. National Center for Health Statistics. Provisional drug overdose death counts. Updated March 17, 2021. Accessed March 22, 2021. www.cdc.gov/nchs/nvss/vsrr/drug-overdose-data.htm
2. CDC. US opioid dispensing rate maps. Updated December 7, 2020. Accessed March 22, 2021. www.cdc.gov/drugoverdose/maps/rxrate-maps.html
3. McBain RK, Dick A, Sorbero M, et al. Growth and distribution of buprenorphine-waivered providers in the United States, 2007-2017. Ann Intern Med. 2020;172:504-506.
1. National Center for Health Statistics. Provisional drug overdose death counts. Updated March 17, 2021. Accessed March 22, 2021. www.cdc.gov/nchs/nvss/vsrr/drug-overdose-data.htm
2. CDC. US opioid dispensing rate maps. Updated December 7, 2020. Accessed March 22, 2021. www.cdc.gov/drugoverdose/maps/rxrate-maps.html
3. McBain RK, Dick A, Sorbero M, et al. Growth and distribution of buprenorphine-waivered providers in the United States, 2007-2017. Ann Intern Med. 2020;172:504-506.
How physicians can provide better care to transgender patients
People who identify as transgender experience many health disparities, in addition to lack of access to quality care. The most commonly cited barrier is the lack of providers who are knowledgeable about transgender health care, according to past surveys.
Even those who do seek care often have unpleasant experiences. A 2015 survey conducted by the National Center for Transgender Equality found that 33% of those who saw a health care provider reported at least one unfavorable experience related to being transgender, such as being verbally harassed or refused treatment because of their gender identity. In fact, 23% of those surveyed say they did not seek health care they needed in the past year because of fear of being mistreated as a transgender person.
This interview has been edited for length and clarity.
Question: Surveys have shown that many people who identify as transgender will seek only transition care, not primary or preventive care. Why is that?
Dr. Brandt: My answer is multifactorial. Transgender patients do seek primary care – just not as readily. There’s a lot of misconceptions about health care needs for the LGBT community in general. For example, lesbian or bisexual women may be not as well informed about the need for Pap smears compared with their heterosexual counterparts. These misconceptions are further exacerbated in the transgender community.
The fact that a lot of patients seek only transition-related care, but not preventive services, such as primary care and gynecologic care, is also related to fears of discrimination and lack of education of providers. These patients are afraid when they walk into an office that they will be misgendered or their physician won’t be familiar with their health care needs.
What can clinics and clinicians do to create a safe and welcoming environment?
Dr. Brandt: It starts with educating office staff about terminology and gender identities.
A key feature of our EHR is the sexual orientation and gender identity platform, which asks questions about a patient’s gender identity, sexual orientation, sex assigned at birth, and organ inventory. These data are then found in the patient information tab and are just as relevant as their insurance status, age, and date of birth.
There are many ways a doctor’s office can signal to patients that they are inclusive. They can hang LGBTQ-friendly flags or symbols or a sign saying, “We have an anti-discrimination policy” in the waiting room. A welcoming environment can also be achieved by revising patient questionnaires or forms so that they aren’t gender-specific or binary.
Given that the patient may have limited contact with a primary care clinician, how do you prioritize what you address during the visit?
Dr. Brandt: Similar to cisgender patients, it depends initially on the age of the patient and the reason for the visit. The priorities of an otherwise healthy transgender patient in their 20s are going to be largely the same as for a cisgender patient of the same age. As patients age in the primary care world, you’re addressing more issues, such as colorectal screening, lipid disorders, and mammograms, and that doesn’t change. For the most part, the problems that you address should be specific for that age group.
It becomes more complicated when you add in factors such as hormone therapy and whether patients have had any type of gender-affirming surgery. Those things can change the usual recommendations for screening or risk assessment. We try to figure out what routine health maintenance and cancer screening a patient needs based on age and risk factors, in addition to hormone status and surgical state.
Do you think that many physicians are educated about the care of underserved populations such as transgender patients?
Dr. Brandt: Yes and no. We are definitely getting better at it. For example, the American College of Obstetricians and Gynecologists published a committee opinion highlighting transgender care. So organizations are starting to prioritize these populations and recognize that they are, in fact, underserved and they have special health care needs.
However, the knowledge gaps are still pretty big. I get calls daily from providers asking questions about how to manage patients on hormones, or how to examine a patient who has undergone a vaginoplasty. I hear a lot of horror stories from transgender patients who had their hormones stopped for absurd and medically misinformed reasons.
But I definitely think it’s getting better and it’s being addressed at all levels – the medical school level, the residency level, and the attending level. It just takes time to inform people and for people to get used to the health care needs of these patients.
What should physicians keep in mind when treating patients who identify as transgender?
Dr. Brandt: First and foremost, understanding the terminology and the difference between gender identity, sex, and sexual orientation. Being familiar with that language and being able to speak that language very comfortably and not being awkward about it is a really important thing for primary care physicians and indeed any physician who treats transgender patients.
Physicians should also be aware that any underserved population has higher rates of mental health issues, such as depression and anxiety. Obviously, that goes along with being underserved and the stigma and the disparities that exist for these patients. Having providers educate themselves about what those disparities are and how they impact a patient’s daily life and health is paramount to knowing how to treat patients.
What are your top health concerns for these patients and how do you address them?
Dr. Brandt: I think mental health and safety is probably the number one for me. About 41% of transgender adults have attempted suicide. That number is roughly 51% in transgender youth. That is an astonishing number. These patients have much higher rates of domestic violence, intimate partner violence, and sexual assault, especially trans women and trans women of color. So understanding those statistics is huge.
Obesity, smoking, and substance abuse are my next three. Again, those are things that should be addressed at any visit, regardless of the gender identity or sexual orientation of the patient, but those rates are particularly high in this population.
Fertility and long-term care for patients should be addressed. Many patients who identify as transgender are told they can’t have a family. As a primary care physician, you may see a patient before they are seen by an ob.gyn. or surgeon. Talking about what a patient’s long-term life goals are with fertility and family planning, and what that looks like for them, is a big thing for me. Other providers may not feel that’s a concern, but I believe it should be discussed before initiation of hormone therapy, which can significantly impact fertility in some patients.
Are there nuances to the physical examination that primary care physicians should be aware of when dealing with transmasculine patients vs. transfeminine patients?
Dr. Brandt: Absolutely. And this interview can’t cover the scope of those nuances. An example that comes to mind is the genital exam. For transgender women who have undergone a vaginoplasty, the pelvic exam can be very affirming. Whereas for transgender men, a gynecologic exam can significantly exacerbate dysphoria and there are ways to conduct the exam to limit this discomfort and avoid creating a traumatic experience for the patient. It’s important to be aware that the genital exam, or any type of genitourinary exam, can be either affirming or not affirming.
Sexually transmitted infections are up in the general population, and the trans population is at even higher risk. What should physicians think about when they assess this risk?
Dr. Brandt: It’s really important for primary care clinicians and for gynecologists to learn to be comfortable talking about sexual practices, because what people do behind closed doors is really a key to how to counsel patients about safe sex.
People are well aware of the need to have safe sex. However, depending on the type of sex that you’re having, what body parts go where, what is truly safe can vary and people may not know, for example, to wear a condom when sex toys are involved or that a transgender male on testosterone can become pregnant during penile-vaginal intercourse. Providers really should be very educated on the array of sexual practices that people have and how to counsel them about those. They should know how to ask patients the gender identity of their sexual partners, the sexual orientation of their partners, and what parts go where during sex.
Providers should also talk to patients about PrEP [pre-exposure prophylaxis], whether they identify as cisgender or transgender. My trans patients tend to be a lot more educated about PrEP than other patients. It’s something that many of the residents, even in a standard gynecologic clinic, for example, don’t talk to cisgender patients about because of the stigma surrounding HIV. Many providers still think that the only people who are at risk for HIV are men who have sex with men. And while those rates are higher in some populations, depending on sexual practices, those aren’t the only patients who qualify for PrEP.
Overall, in order to counsel patients about STIs and safe sexual practices, providers should learn to be comfortable talking about sex.
Do you have any strategies on how to make the appointment more successful in addressing those issues?
Dr. Brandt: Bedside manner is a hard thing to teach, and comfort in talking about sex, gender identity, and sexual orientation can vary – but there are a lot of continuing medical education courses that physicians can utilize through the World Professional Association for Transgender Health.
If providers start to notice an influx of patients who identify as transgender or if they want to start seeing transgender patients, it’s really important for them to have that training before they start interacting with patients. In all of medicine, we sort of learn as we go, but this patient population has been subjected to discrimination, violence, error, and misgendering. They have dealt with providers who didn’t understand their health care needs. While this field is evolving, knowing how to appropriately address a patient (using their correct name, pronouns, etc.) is an absolute must.
That needs to be part of a provider’s routine vernacular and not something that they sort of stumble through. You can scare a patient away as soon as they walk into the office with an uneducated front desk staff and things that are seen in the office. Seeking out those educational tools, being aware of your own deficits as a provider and the educational needs of your office, and addressing those needs is really key.
A version of this article first appeared on Medscape.com.
People who identify as transgender experience many health disparities, in addition to lack of access to quality care. The most commonly cited barrier is the lack of providers who are knowledgeable about transgender health care, according to past surveys.
Even those who do seek care often have unpleasant experiences. A 2015 survey conducted by the National Center for Transgender Equality found that 33% of those who saw a health care provider reported at least one unfavorable experience related to being transgender, such as being verbally harassed or refused treatment because of their gender identity. In fact, 23% of those surveyed say they did not seek health care they needed in the past year because of fear of being mistreated as a transgender person.
This interview has been edited for length and clarity.
Question: Surveys have shown that many people who identify as transgender will seek only transition care, not primary or preventive care. Why is that?
Dr. Brandt: My answer is multifactorial. Transgender patients do seek primary care – just not as readily. There’s a lot of misconceptions about health care needs for the LGBT community in general. For example, lesbian or bisexual women may be not as well informed about the need for Pap smears compared with their heterosexual counterparts. These misconceptions are further exacerbated in the transgender community.
The fact that a lot of patients seek only transition-related care, but not preventive services, such as primary care and gynecologic care, is also related to fears of discrimination and lack of education of providers. These patients are afraid when they walk into an office that they will be misgendered or their physician won’t be familiar with their health care needs.
What can clinics and clinicians do to create a safe and welcoming environment?
Dr. Brandt: It starts with educating office staff about terminology and gender identities.
A key feature of our EHR is the sexual orientation and gender identity platform, which asks questions about a patient’s gender identity, sexual orientation, sex assigned at birth, and organ inventory. These data are then found in the patient information tab and are just as relevant as their insurance status, age, and date of birth.
There are many ways a doctor’s office can signal to patients that they are inclusive. They can hang LGBTQ-friendly flags or symbols or a sign saying, “We have an anti-discrimination policy” in the waiting room. A welcoming environment can also be achieved by revising patient questionnaires or forms so that they aren’t gender-specific or binary.
Given that the patient may have limited contact with a primary care clinician, how do you prioritize what you address during the visit?
Dr. Brandt: Similar to cisgender patients, it depends initially on the age of the patient and the reason for the visit. The priorities of an otherwise healthy transgender patient in their 20s are going to be largely the same as for a cisgender patient of the same age. As patients age in the primary care world, you’re addressing more issues, such as colorectal screening, lipid disorders, and mammograms, and that doesn’t change. For the most part, the problems that you address should be specific for that age group.
It becomes more complicated when you add in factors such as hormone therapy and whether patients have had any type of gender-affirming surgery. Those things can change the usual recommendations for screening or risk assessment. We try to figure out what routine health maintenance and cancer screening a patient needs based on age and risk factors, in addition to hormone status and surgical state.
Do you think that many physicians are educated about the care of underserved populations such as transgender patients?
Dr. Brandt: Yes and no. We are definitely getting better at it. For example, the American College of Obstetricians and Gynecologists published a committee opinion highlighting transgender care. So organizations are starting to prioritize these populations and recognize that they are, in fact, underserved and they have special health care needs.
However, the knowledge gaps are still pretty big. I get calls daily from providers asking questions about how to manage patients on hormones, or how to examine a patient who has undergone a vaginoplasty. I hear a lot of horror stories from transgender patients who had their hormones stopped for absurd and medically misinformed reasons.
But I definitely think it’s getting better and it’s being addressed at all levels – the medical school level, the residency level, and the attending level. It just takes time to inform people and for people to get used to the health care needs of these patients.
What should physicians keep in mind when treating patients who identify as transgender?
Dr. Brandt: First and foremost, understanding the terminology and the difference between gender identity, sex, and sexual orientation. Being familiar with that language and being able to speak that language very comfortably and not being awkward about it is a really important thing for primary care physicians and indeed any physician who treats transgender patients.
Physicians should also be aware that any underserved population has higher rates of mental health issues, such as depression and anxiety. Obviously, that goes along with being underserved and the stigma and the disparities that exist for these patients. Having providers educate themselves about what those disparities are and how they impact a patient’s daily life and health is paramount to knowing how to treat patients.
What are your top health concerns for these patients and how do you address them?
Dr. Brandt: I think mental health and safety is probably the number one for me. About 41% of transgender adults have attempted suicide. That number is roughly 51% in transgender youth. That is an astonishing number. These patients have much higher rates of domestic violence, intimate partner violence, and sexual assault, especially trans women and trans women of color. So understanding those statistics is huge.
Obesity, smoking, and substance abuse are my next three. Again, those are things that should be addressed at any visit, regardless of the gender identity or sexual orientation of the patient, but those rates are particularly high in this population.
Fertility and long-term care for patients should be addressed. Many patients who identify as transgender are told they can’t have a family. As a primary care physician, you may see a patient before they are seen by an ob.gyn. or surgeon. Talking about what a patient’s long-term life goals are with fertility and family planning, and what that looks like for them, is a big thing for me. Other providers may not feel that’s a concern, but I believe it should be discussed before initiation of hormone therapy, which can significantly impact fertility in some patients.
Are there nuances to the physical examination that primary care physicians should be aware of when dealing with transmasculine patients vs. transfeminine patients?
Dr. Brandt: Absolutely. And this interview can’t cover the scope of those nuances. An example that comes to mind is the genital exam. For transgender women who have undergone a vaginoplasty, the pelvic exam can be very affirming. Whereas for transgender men, a gynecologic exam can significantly exacerbate dysphoria and there are ways to conduct the exam to limit this discomfort and avoid creating a traumatic experience for the patient. It’s important to be aware that the genital exam, or any type of genitourinary exam, can be either affirming or not affirming.
Sexually transmitted infections are up in the general population, and the trans population is at even higher risk. What should physicians think about when they assess this risk?
Dr. Brandt: It’s really important for primary care clinicians and for gynecologists to learn to be comfortable talking about sexual practices, because what people do behind closed doors is really a key to how to counsel patients about safe sex.
People are well aware of the need to have safe sex. However, depending on the type of sex that you’re having, what body parts go where, what is truly safe can vary and people may not know, for example, to wear a condom when sex toys are involved or that a transgender male on testosterone can become pregnant during penile-vaginal intercourse. Providers really should be very educated on the array of sexual practices that people have and how to counsel them about those. They should know how to ask patients the gender identity of their sexual partners, the sexual orientation of their partners, and what parts go where during sex.
Providers should also talk to patients about PrEP [pre-exposure prophylaxis], whether they identify as cisgender or transgender. My trans patients tend to be a lot more educated about PrEP than other patients. It’s something that many of the residents, even in a standard gynecologic clinic, for example, don’t talk to cisgender patients about because of the stigma surrounding HIV. Many providers still think that the only people who are at risk for HIV are men who have sex with men. And while those rates are higher in some populations, depending on sexual practices, those aren’t the only patients who qualify for PrEP.
Overall, in order to counsel patients about STIs and safe sexual practices, providers should learn to be comfortable talking about sex.
Do you have any strategies on how to make the appointment more successful in addressing those issues?
Dr. Brandt: Bedside manner is a hard thing to teach, and comfort in talking about sex, gender identity, and sexual orientation can vary – but there are a lot of continuing medical education courses that physicians can utilize through the World Professional Association for Transgender Health.
If providers start to notice an influx of patients who identify as transgender or if they want to start seeing transgender patients, it’s really important for them to have that training before they start interacting with patients. In all of medicine, we sort of learn as we go, but this patient population has been subjected to discrimination, violence, error, and misgendering. They have dealt with providers who didn’t understand their health care needs. While this field is evolving, knowing how to appropriately address a patient (using their correct name, pronouns, etc.) is an absolute must.
That needs to be part of a provider’s routine vernacular and not something that they sort of stumble through. You can scare a patient away as soon as they walk into the office with an uneducated front desk staff and things that are seen in the office. Seeking out those educational tools, being aware of your own deficits as a provider and the educational needs of your office, and addressing those needs is really key.
A version of this article first appeared on Medscape.com.
People who identify as transgender experience many health disparities, in addition to lack of access to quality care. The most commonly cited barrier is the lack of providers who are knowledgeable about transgender health care, according to past surveys.
Even those who do seek care often have unpleasant experiences. A 2015 survey conducted by the National Center for Transgender Equality found that 33% of those who saw a health care provider reported at least one unfavorable experience related to being transgender, such as being verbally harassed or refused treatment because of their gender identity. In fact, 23% of those surveyed say they did not seek health care they needed in the past year because of fear of being mistreated as a transgender person.
This interview has been edited for length and clarity.
Question: Surveys have shown that many people who identify as transgender will seek only transition care, not primary or preventive care. Why is that?
Dr. Brandt: My answer is multifactorial. Transgender patients do seek primary care – just not as readily. There’s a lot of misconceptions about health care needs for the LGBT community in general. For example, lesbian or bisexual women may be not as well informed about the need for Pap smears compared with their heterosexual counterparts. These misconceptions are further exacerbated in the transgender community.
The fact that a lot of patients seek only transition-related care, but not preventive services, such as primary care and gynecologic care, is also related to fears of discrimination and lack of education of providers. These patients are afraid when they walk into an office that they will be misgendered or their physician won’t be familiar with their health care needs.
What can clinics and clinicians do to create a safe and welcoming environment?
Dr. Brandt: It starts with educating office staff about terminology and gender identities.
A key feature of our EHR is the sexual orientation and gender identity platform, which asks questions about a patient’s gender identity, sexual orientation, sex assigned at birth, and organ inventory. These data are then found in the patient information tab and are just as relevant as their insurance status, age, and date of birth.
There are many ways a doctor’s office can signal to patients that they are inclusive. They can hang LGBTQ-friendly flags or symbols or a sign saying, “We have an anti-discrimination policy” in the waiting room. A welcoming environment can also be achieved by revising patient questionnaires or forms so that they aren’t gender-specific or binary.
Given that the patient may have limited contact with a primary care clinician, how do you prioritize what you address during the visit?
Dr. Brandt: Similar to cisgender patients, it depends initially on the age of the patient and the reason for the visit. The priorities of an otherwise healthy transgender patient in their 20s are going to be largely the same as for a cisgender patient of the same age. As patients age in the primary care world, you’re addressing more issues, such as colorectal screening, lipid disorders, and mammograms, and that doesn’t change. For the most part, the problems that you address should be specific for that age group.
It becomes more complicated when you add in factors such as hormone therapy and whether patients have had any type of gender-affirming surgery. Those things can change the usual recommendations for screening or risk assessment. We try to figure out what routine health maintenance and cancer screening a patient needs based on age and risk factors, in addition to hormone status and surgical state.
Do you think that many physicians are educated about the care of underserved populations such as transgender patients?
Dr. Brandt: Yes and no. We are definitely getting better at it. For example, the American College of Obstetricians and Gynecologists published a committee opinion highlighting transgender care. So organizations are starting to prioritize these populations and recognize that they are, in fact, underserved and they have special health care needs.
However, the knowledge gaps are still pretty big. I get calls daily from providers asking questions about how to manage patients on hormones, or how to examine a patient who has undergone a vaginoplasty. I hear a lot of horror stories from transgender patients who had their hormones stopped for absurd and medically misinformed reasons.
But I definitely think it’s getting better and it’s being addressed at all levels – the medical school level, the residency level, and the attending level. It just takes time to inform people and for people to get used to the health care needs of these patients.
What should physicians keep in mind when treating patients who identify as transgender?
Dr. Brandt: First and foremost, understanding the terminology and the difference between gender identity, sex, and sexual orientation. Being familiar with that language and being able to speak that language very comfortably and not being awkward about it is a really important thing for primary care physicians and indeed any physician who treats transgender patients.
Physicians should also be aware that any underserved population has higher rates of mental health issues, such as depression and anxiety. Obviously, that goes along with being underserved and the stigma and the disparities that exist for these patients. Having providers educate themselves about what those disparities are and how they impact a patient’s daily life and health is paramount to knowing how to treat patients.
What are your top health concerns for these patients and how do you address them?
Dr. Brandt: I think mental health and safety is probably the number one for me. About 41% of transgender adults have attempted suicide. That number is roughly 51% in transgender youth. That is an astonishing number. These patients have much higher rates of domestic violence, intimate partner violence, and sexual assault, especially trans women and trans women of color. So understanding those statistics is huge.
Obesity, smoking, and substance abuse are my next three. Again, those are things that should be addressed at any visit, regardless of the gender identity or sexual orientation of the patient, but those rates are particularly high in this population.
Fertility and long-term care for patients should be addressed. Many patients who identify as transgender are told they can’t have a family. As a primary care physician, you may see a patient before they are seen by an ob.gyn. or surgeon. Talking about what a patient’s long-term life goals are with fertility and family planning, and what that looks like for them, is a big thing for me. Other providers may not feel that’s a concern, but I believe it should be discussed before initiation of hormone therapy, which can significantly impact fertility in some patients.
Are there nuances to the physical examination that primary care physicians should be aware of when dealing with transmasculine patients vs. transfeminine patients?
Dr. Brandt: Absolutely. And this interview can’t cover the scope of those nuances. An example that comes to mind is the genital exam. For transgender women who have undergone a vaginoplasty, the pelvic exam can be very affirming. Whereas for transgender men, a gynecologic exam can significantly exacerbate dysphoria and there are ways to conduct the exam to limit this discomfort and avoid creating a traumatic experience for the patient. It’s important to be aware that the genital exam, or any type of genitourinary exam, can be either affirming or not affirming.
Sexually transmitted infections are up in the general population, and the trans population is at even higher risk. What should physicians think about when they assess this risk?
Dr. Brandt: It’s really important for primary care clinicians and for gynecologists to learn to be comfortable talking about sexual practices, because what people do behind closed doors is really a key to how to counsel patients about safe sex.
People are well aware of the need to have safe sex. However, depending on the type of sex that you’re having, what body parts go where, what is truly safe can vary and people may not know, for example, to wear a condom when sex toys are involved or that a transgender male on testosterone can become pregnant during penile-vaginal intercourse. Providers really should be very educated on the array of sexual practices that people have and how to counsel them about those. They should know how to ask patients the gender identity of their sexual partners, the sexual orientation of their partners, and what parts go where during sex.
Providers should also talk to patients about PrEP [pre-exposure prophylaxis], whether they identify as cisgender or transgender. My trans patients tend to be a lot more educated about PrEP than other patients. It’s something that many of the residents, even in a standard gynecologic clinic, for example, don’t talk to cisgender patients about because of the stigma surrounding HIV. Many providers still think that the only people who are at risk for HIV are men who have sex with men. And while those rates are higher in some populations, depending on sexual practices, those aren’t the only patients who qualify for PrEP.
Overall, in order to counsel patients about STIs and safe sexual practices, providers should learn to be comfortable talking about sex.
Do you have any strategies on how to make the appointment more successful in addressing those issues?
Dr. Brandt: Bedside manner is a hard thing to teach, and comfort in talking about sex, gender identity, and sexual orientation can vary – but there are a lot of continuing medical education courses that physicians can utilize through the World Professional Association for Transgender Health.
If providers start to notice an influx of patients who identify as transgender or if they want to start seeing transgender patients, it’s really important for them to have that training before they start interacting with patients. In all of medicine, we sort of learn as we go, but this patient population has been subjected to discrimination, violence, error, and misgendering. They have dealt with providers who didn’t understand their health care needs. While this field is evolving, knowing how to appropriately address a patient (using their correct name, pronouns, etc.) is an absolute must.
That needs to be part of a provider’s routine vernacular and not something that they sort of stumble through. You can scare a patient away as soon as they walk into the office with an uneducated front desk staff and things that are seen in the office. Seeking out those educational tools, being aware of your own deficits as a provider and the educational needs of your office, and addressing those needs is really key.
A version of this article first appeared on Medscape.com.