User login
Study Identifies Oral Antibiotics Linked to Severe Cutaneous Reactions
according to a large, population-based, nested case-control study of older adults, spanning two decades.
The findings, published online in JAMA, “underscore the importance of judicious prescribing, with preferential use of antibiotics associated with a lower risk when clinically appropriate,” noted senior author David Juurlink, MD, PhD, professor of medicine; pediatrics; and health policy, management and evaluation at the University of Toronto, and head of the Clinical Pharmacology and Toxicology Division at Sunnybrook Health Sciences Centre, also in Toronto, Ontario, Canada, and coauthors.
“We hope our study raises awareness about the importance of drug allergy and gains support for future studies to improve drug allergy care,” lead author Erika Lee, MD, clinical immunology and allergy lecturer at the University of Toronto’s Drug Allergy Clinic, Sunnybrook Health Sciences Centre, said in an interview. “It is important to recognize symptoms and signs of a severe drug rash and promptly stop culprit drugs to prevent worsening reaction.”
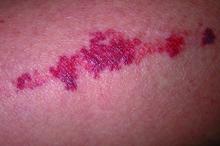
Serious cADRs are “a group of rare but potentially life-threatening drug hypersensitivity reactions involving the skin and, frequently, internal organs,” the authors wrote. “Typically delayed in onset, these reactions include drug reaction with eosinophilia and systemic symptoms, Stevens-Johnson syndrome (SJS), and toxic epidermal necrolysis (TEN) — the most severe cADR, which has a reported mortality of 20%-40%,” they noted.
Speculation Without Data
Although it has been speculated that some oral antibiotics are more likely than others to be associated with serious cADRs, there have been no population-based studies examining this, they added.
The study included adults aged 66 years or older and used administrative health databases in Ontario, spanning from April 1, 2002, to March 31, 2022. Data on antibiotic use were taken from the Ontario Drug Benefit database. The Canadian Institute for Health Information (CIHI) National Ambulatory Care Reporting System was used to obtain data on emergency department (ED) visits for cADRs, while the CIHI Discharge Abstract Database was used to identify hospitalizations for cADRs. Finally, demographic information and outpatient healthcare utilization data were obtained from the Registered Persons Database and the Ontario Health Insurance Plan database, respectively.
A cohort of 21,758 older adults (median age, 75 years; 64.1% women) who had an ED visit or hospitalization for serious cADRs within 60 days of receiving antibiotic therapy was matched by age and sex with 87,025 antibiotic-treated controls who did not have a cutaneous reaction.
The median duration of antibiotic prescription was 7 days among cases and controls, and among the cases, the median latency period between antibiotic prescriptions and hospital visits for cADRs was 14 days. Most of the case patients went to the ED only (86.9%), and the rest were hospitalized.
The most commonly prescribed antibiotic class was penicillins (28.9%), followed by cephalosporins (18.2%), fluoroquinolones (16.5%), macrolides (14.8%), nitrofurantoin (8.6%), and sulfonamides (6.2%). Less commonly used antibiotics (“other” antibiotics) accounted for 6.9%.
Macrolide antibiotics were used as the reference because they are rarely associated with serious cADRs, noted the authors, and the multivariable analysis, adjusted for risk factors associated with serious cADRs, including malignancy, chronic liver disease, chronic kidney disease, and HIV.
After multivariable adjustment, relative to macrolides, sulfonamides were most strongly associated with serious cADRs (adjusted odds ratio [aOR], 2.9) but so were all other antibiotic classes, including cephalosporins (aOR, 2.6), “other” antibiotics (aOR, 2.3), nitrofurantoin (aOR, 2.2), penicillins (aOR, 1.4), and fluoroquinolones (aOR,1.3).
In the secondary analysis, the crude rate of ED visits or hospitalizations for cADRs was highest for cephalosporins (4.92 per 1000 prescriptions), followed by sulfonamides (3.22 per 1000 prescriptions). Among hospitalized patients, the median length of stay was 6 days, with 9.6% requiring transfer to a critical care unit and 5.3% dying in the hospital.
Hospitalizations, ED Visits Not Studied Previously
“Notably, the rate of antibiotic-associated serious cADRs leading to an ED visit or hospitalization has not been previously studied,” noted the authors. “We found that at least two hospital encounters for serious cADRs ensued for every 1000 antibiotic prescriptions. This rate is considerably higher than suggested by studies that examine only SJS/TEN and drug reaction with eosinophilia and systemic symptoms.”
Dr. Lee also emphasized the previously unreported findings about nitrofurantoin. “It is surprising to find that nitrofurantoin, a commonly prescribed antibiotic for urinary tract infection, is also associated with an increased risk of severe drug rash,” she said in an interview.
“This finding highlights a potential novel risk at a population-based level and should be further explored in other populations to verify this association,” the authors wrote.
Amesh Adalja, MD, a senior scholar at the Johns Hopkins Center for Health Security in Baltimore, Maryland, and a spokesperson for the Infectious Diseases Society of America, who was not involved in the study, agreed that the nitrofurantoin finding was surprising, but he was not surprised that sulfonamides were high on the list.
“The study reinforces that antibiotics are not benign medications to be dispensed injudiciously,” he said in an interview. “Antibiotics have risks, including serious skin reactions, as well as the fostering of antibiotic resistance. Clinicians should always first ask themselves if their patient actually merits an antibiotic and then assess what is the safest antibiotic for the purpose, bearing in mind that certain antibiotics are more likely to result in adverse reactions than others.”
The study was supported by the Canadian Institutes of Health Research. The study was conducted at ICES, which is funded in part by an annual grant from the Ontario Ministry of Health and Long-Term Care. One coauthor reported receiving compensation from the British Journal of Dermatology as reviewer and section editor, the American Academy of Dermatology as guidelines writer, Canadian Dermatology Today as manuscript writer, and the National Eczema Association and the Canadian Agency for Drugs and Technologies in Health as consultant; as well as receiving research grants to the coauthor’s institution from the National Eczema Association, Eczema Society of Canada, Canadian Dermatology Foundation, Canadian Institutes of Health Research, US National Institutes of Health, and PSI Foundation. Another coauthor reported receiving grants from AbbVie, Bausch Health, Celgene, Lilly, Incyte, Janssen, LEO Pharma, L’Oréal, Novartis, Organon, Pfizer, Sandoz, Amgen, and Boehringer Ingelheim; receiving payment or honoraria for speaking from Sanofi China; participating on advisory boards for LEO Pharma, Novartis, Sanofi, and Union Therapeutics; and receiving equipment donation from L’Oréal. Dr. Adalja reported no relevant disclosures.
A version of this article first appeared on Medscape.com.
according to a large, population-based, nested case-control study of older adults, spanning two decades.
The findings, published online in JAMA, “underscore the importance of judicious prescribing, with preferential use of antibiotics associated with a lower risk when clinically appropriate,” noted senior author David Juurlink, MD, PhD, professor of medicine; pediatrics; and health policy, management and evaluation at the University of Toronto, and head of the Clinical Pharmacology and Toxicology Division at Sunnybrook Health Sciences Centre, also in Toronto, Ontario, Canada, and coauthors.
“We hope our study raises awareness about the importance of drug allergy and gains support for future studies to improve drug allergy care,” lead author Erika Lee, MD, clinical immunology and allergy lecturer at the University of Toronto’s Drug Allergy Clinic, Sunnybrook Health Sciences Centre, said in an interview. “It is important to recognize symptoms and signs of a severe drug rash and promptly stop culprit drugs to prevent worsening reaction.”
Serious cADRs are “a group of rare but potentially life-threatening drug hypersensitivity reactions involving the skin and, frequently, internal organs,” the authors wrote. “Typically delayed in onset, these reactions include drug reaction with eosinophilia and systemic symptoms, Stevens-Johnson syndrome (SJS), and toxic epidermal necrolysis (TEN) — the most severe cADR, which has a reported mortality of 20%-40%,” they noted.
Speculation Without Data
Although it has been speculated that some oral antibiotics are more likely than others to be associated with serious cADRs, there have been no population-based studies examining this, they added.
The study included adults aged 66 years or older and used administrative health databases in Ontario, spanning from April 1, 2002, to March 31, 2022. Data on antibiotic use were taken from the Ontario Drug Benefit database. The Canadian Institute for Health Information (CIHI) National Ambulatory Care Reporting System was used to obtain data on emergency department (ED) visits for cADRs, while the CIHI Discharge Abstract Database was used to identify hospitalizations for cADRs. Finally, demographic information and outpatient healthcare utilization data were obtained from the Registered Persons Database and the Ontario Health Insurance Plan database, respectively.
A cohort of 21,758 older adults (median age, 75 years; 64.1% women) who had an ED visit or hospitalization for serious cADRs within 60 days of receiving antibiotic therapy was matched by age and sex with 87,025 antibiotic-treated controls who did not have a cutaneous reaction.
The median duration of antibiotic prescription was 7 days among cases and controls, and among the cases, the median latency period between antibiotic prescriptions and hospital visits for cADRs was 14 days. Most of the case patients went to the ED only (86.9%), and the rest were hospitalized.
The most commonly prescribed antibiotic class was penicillins (28.9%), followed by cephalosporins (18.2%), fluoroquinolones (16.5%), macrolides (14.8%), nitrofurantoin (8.6%), and sulfonamides (6.2%). Less commonly used antibiotics (“other” antibiotics) accounted for 6.9%.
Macrolide antibiotics were used as the reference because they are rarely associated with serious cADRs, noted the authors, and the multivariable analysis, adjusted for risk factors associated with serious cADRs, including malignancy, chronic liver disease, chronic kidney disease, and HIV.
After multivariable adjustment, relative to macrolides, sulfonamides were most strongly associated with serious cADRs (adjusted odds ratio [aOR], 2.9) but so were all other antibiotic classes, including cephalosporins (aOR, 2.6), “other” antibiotics (aOR, 2.3), nitrofurantoin (aOR, 2.2), penicillins (aOR, 1.4), and fluoroquinolones (aOR,1.3).
In the secondary analysis, the crude rate of ED visits or hospitalizations for cADRs was highest for cephalosporins (4.92 per 1000 prescriptions), followed by sulfonamides (3.22 per 1000 prescriptions). Among hospitalized patients, the median length of stay was 6 days, with 9.6% requiring transfer to a critical care unit and 5.3% dying in the hospital.
Hospitalizations, ED Visits Not Studied Previously
“Notably, the rate of antibiotic-associated serious cADRs leading to an ED visit or hospitalization has not been previously studied,” noted the authors. “We found that at least two hospital encounters for serious cADRs ensued for every 1000 antibiotic prescriptions. This rate is considerably higher than suggested by studies that examine only SJS/TEN and drug reaction with eosinophilia and systemic symptoms.”
Dr. Lee also emphasized the previously unreported findings about nitrofurantoin. “It is surprising to find that nitrofurantoin, a commonly prescribed antibiotic for urinary tract infection, is also associated with an increased risk of severe drug rash,” she said in an interview.
“This finding highlights a potential novel risk at a population-based level and should be further explored in other populations to verify this association,” the authors wrote.
Amesh Adalja, MD, a senior scholar at the Johns Hopkins Center for Health Security in Baltimore, Maryland, and a spokesperson for the Infectious Diseases Society of America, who was not involved in the study, agreed that the nitrofurantoin finding was surprising, but he was not surprised that sulfonamides were high on the list.
“The study reinforces that antibiotics are not benign medications to be dispensed injudiciously,” he said in an interview. “Antibiotics have risks, including serious skin reactions, as well as the fostering of antibiotic resistance. Clinicians should always first ask themselves if their patient actually merits an antibiotic and then assess what is the safest antibiotic for the purpose, bearing in mind that certain antibiotics are more likely to result in adverse reactions than others.”
The study was supported by the Canadian Institutes of Health Research. The study was conducted at ICES, which is funded in part by an annual grant from the Ontario Ministry of Health and Long-Term Care. One coauthor reported receiving compensation from the British Journal of Dermatology as reviewer and section editor, the American Academy of Dermatology as guidelines writer, Canadian Dermatology Today as manuscript writer, and the National Eczema Association and the Canadian Agency for Drugs and Technologies in Health as consultant; as well as receiving research grants to the coauthor’s institution from the National Eczema Association, Eczema Society of Canada, Canadian Dermatology Foundation, Canadian Institutes of Health Research, US National Institutes of Health, and PSI Foundation. Another coauthor reported receiving grants from AbbVie, Bausch Health, Celgene, Lilly, Incyte, Janssen, LEO Pharma, L’Oréal, Novartis, Organon, Pfizer, Sandoz, Amgen, and Boehringer Ingelheim; receiving payment or honoraria for speaking from Sanofi China; participating on advisory boards for LEO Pharma, Novartis, Sanofi, and Union Therapeutics; and receiving equipment donation from L’Oréal. Dr. Adalja reported no relevant disclosures.
A version of this article first appeared on Medscape.com.
according to a large, population-based, nested case-control study of older adults, spanning two decades.
The findings, published online in JAMA, “underscore the importance of judicious prescribing, with preferential use of antibiotics associated with a lower risk when clinically appropriate,” noted senior author David Juurlink, MD, PhD, professor of medicine; pediatrics; and health policy, management and evaluation at the University of Toronto, and head of the Clinical Pharmacology and Toxicology Division at Sunnybrook Health Sciences Centre, also in Toronto, Ontario, Canada, and coauthors.
“We hope our study raises awareness about the importance of drug allergy and gains support for future studies to improve drug allergy care,” lead author Erika Lee, MD, clinical immunology and allergy lecturer at the University of Toronto’s Drug Allergy Clinic, Sunnybrook Health Sciences Centre, said in an interview. “It is important to recognize symptoms and signs of a severe drug rash and promptly stop culprit drugs to prevent worsening reaction.”
Serious cADRs are “a group of rare but potentially life-threatening drug hypersensitivity reactions involving the skin and, frequently, internal organs,” the authors wrote. “Typically delayed in onset, these reactions include drug reaction with eosinophilia and systemic symptoms, Stevens-Johnson syndrome (SJS), and toxic epidermal necrolysis (TEN) — the most severe cADR, which has a reported mortality of 20%-40%,” they noted.
Speculation Without Data
Although it has been speculated that some oral antibiotics are more likely than others to be associated with serious cADRs, there have been no population-based studies examining this, they added.
The study included adults aged 66 years or older and used administrative health databases in Ontario, spanning from April 1, 2002, to March 31, 2022. Data on antibiotic use were taken from the Ontario Drug Benefit database. The Canadian Institute for Health Information (CIHI) National Ambulatory Care Reporting System was used to obtain data on emergency department (ED) visits for cADRs, while the CIHI Discharge Abstract Database was used to identify hospitalizations for cADRs. Finally, demographic information and outpatient healthcare utilization data were obtained from the Registered Persons Database and the Ontario Health Insurance Plan database, respectively.
A cohort of 21,758 older adults (median age, 75 years; 64.1% women) who had an ED visit or hospitalization for serious cADRs within 60 days of receiving antibiotic therapy was matched by age and sex with 87,025 antibiotic-treated controls who did not have a cutaneous reaction.
The median duration of antibiotic prescription was 7 days among cases and controls, and among the cases, the median latency period between antibiotic prescriptions and hospital visits for cADRs was 14 days. Most of the case patients went to the ED only (86.9%), and the rest were hospitalized.
The most commonly prescribed antibiotic class was penicillins (28.9%), followed by cephalosporins (18.2%), fluoroquinolones (16.5%), macrolides (14.8%), nitrofurantoin (8.6%), and sulfonamides (6.2%). Less commonly used antibiotics (“other” antibiotics) accounted for 6.9%.
Macrolide antibiotics were used as the reference because they are rarely associated with serious cADRs, noted the authors, and the multivariable analysis, adjusted for risk factors associated with serious cADRs, including malignancy, chronic liver disease, chronic kidney disease, and HIV.
After multivariable adjustment, relative to macrolides, sulfonamides were most strongly associated with serious cADRs (adjusted odds ratio [aOR], 2.9) but so were all other antibiotic classes, including cephalosporins (aOR, 2.6), “other” antibiotics (aOR, 2.3), nitrofurantoin (aOR, 2.2), penicillins (aOR, 1.4), and fluoroquinolones (aOR,1.3).
In the secondary analysis, the crude rate of ED visits or hospitalizations for cADRs was highest for cephalosporins (4.92 per 1000 prescriptions), followed by sulfonamides (3.22 per 1000 prescriptions). Among hospitalized patients, the median length of stay was 6 days, with 9.6% requiring transfer to a critical care unit and 5.3% dying in the hospital.
Hospitalizations, ED Visits Not Studied Previously
“Notably, the rate of antibiotic-associated serious cADRs leading to an ED visit or hospitalization has not been previously studied,” noted the authors. “We found that at least two hospital encounters for serious cADRs ensued for every 1000 antibiotic prescriptions. This rate is considerably higher than suggested by studies that examine only SJS/TEN and drug reaction with eosinophilia and systemic symptoms.”
Dr. Lee also emphasized the previously unreported findings about nitrofurantoin. “It is surprising to find that nitrofurantoin, a commonly prescribed antibiotic for urinary tract infection, is also associated with an increased risk of severe drug rash,” she said in an interview.
“This finding highlights a potential novel risk at a population-based level and should be further explored in other populations to verify this association,” the authors wrote.
Amesh Adalja, MD, a senior scholar at the Johns Hopkins Center for Health Security in Baltimore, Maryland, and a spokesperson for the Infectious Diseases Society of America, who was not involved in the study, agreed that the nitrofurantoin finding was surprising, but he was not surprised that sulfonamides were high on the list.
“The study reinforces that antibiotics are not benign medications to be dispensed injudiciously,” he said in an interview. “Antibiotics have risks, including serious skin reactions, as well as the fostering of antibiotic resistance. Clinicians should always first ask themselves if their patient actually merits an antibiotic and then assess what is the safest antibiotic for the purpose, bearing in mind that certain antibiotics are more likely to result in adverse reactions than others.”
The study was supported by the Canadian Institutes of Health Research. The study was conducted at ICES, which is funded in part by an annual grant from the Ontario Ministry of Health and Long-Term Care. One coauthor reported receiving compensation from the British Journal of Dermatology as reviewer and section editor, the American Academy of Dermatology as guidelines writer, Canadian Dermatology Today as manuscript writer, and the National Eczema Association and the Canadian Agency for Drugs and Technologies in Health as consultant; as well as receiving research grants to the coauthor’s institution from the National Eczema Association, Eczema Society of Canada, Canadian Dermatology Foundation, Canadian Institutes of Health Research, US National Institutes of Health, and PSI Foundation. Another coauthor reported receiving grants from AbbVie, Bausch Health, Celgene, Lilly, Incyte, Janssen, LEO Pharma, L’Oréal, Novartis, Organon, Pfizer, Sandoz, Amgen, and Boehringer Ingelheim; receiving payment or honoraria for speaking from Sanofi China; participating on advisory boards for LEO Pharma, Novartis, Sanofi, and Union Therapeutics; and receiving equipment donation from L’Oréal. Dr. Adalja reported no relevant disclosures.
A version of this article first appeared on Medscape.com.
FROM JAMA
Management, Evaluation of Chronic Itch in Older Adults
WASHINGTON — , Shawn G. Kwatra, MD, said at the ElderDerm conference on dermatology in older patients hosted by the GW School of Medicine & Health Sciences.
“We found a few years ago that eosinophils seem to differentiate this group, and now we’re finding that IgE and CBC [complete blood count] differential can help you get a little better sense of who has an immune-driven itch vs something more neuropathic,” said Dr. Kwatra, professor and chair of dermatology at the University of Maryland, Baltimore, who founded and directed the Johns Hopkins Itch Center before coming to the University of Maryland in 2023. Not all patients with immune-driven itch will have these biomarkers, “but it’s a helpful tool,” he said.
CPUO is the term that is increasingly being used, he said, to describe intense, chronic pruritus without primary skin lesions or rashes and without any known systemic cause. It becomes more common as people get older and is sometimes debilitating. The initial evaluation should be kept “simple and straightforward,” he advised, with heightened concern for underlying malignancy in those who present with an itch of less than 12 months’ duration.
Biologics, JAK Inhibitors: Case Reports, Ongoing Research
Research conducted by Dr. Kwatra and Jaya Manjunath, a fourth-year medical student at The George Washington University, Washington, documented higher levels of Th2-associated cytokines and inflammatory markers in patients with CPUO who had elevated IgE or eosinophil levels, or both than in patients with itch who had low IgE and eosinophil levels. The patients with higher levels also had a greater response to off-label treatment with immunomodulatory therapy.
“Multiple Th2-related inflammatory markers, like IL [interleukin]-5 and eotaxin-3, were reduced after dupilumab” in patients who responded to the therapy, said Ms. Manjunath, who co-presented the meeting session on chronic itch with Dr. Kwatra. Other changes in the plasma cytokine profile included a reduction in the serum level of thymus and activation-regulated chemokine, which is a biomarker for atopic dermatitis. The research is under review for publication.
Meanwhile, a phase 3 trial (LIBERTY-CPUO-CHIC) of dupilumab for CPUO is currently underway, Dr. Kwatra noted. Investigators are randomizing patients with severe pruritus (Worst Itch Numeric Rating Scale [WI-NRS] ≥ 7) to dupilumab or placebo for 12 or 24 weeks.
In one of several cases shared by Dr. Kwatra and Ms. Manjunath, a 71-year-old Black woman with a 6-month history of generalized itch (WI-NRS = 10) and a history of type 2 diabetes, hypertension, and chronic kidney disease was found to have elevated eosinophil levels and a negative malignancy workup. Previous therapies included antihistamines and topical steroids. She was started on a 600-mg loading dose of subcutaneous dupilumab followed by 300 mg every 14 days. At the 2-month follow-up, her WI-NRS score was 0.
Because “dupilumab is off label right now for this form of itch, oftentimes our first line is methotrexate,” Dr. Kwatra said. Patients “can have a good response with this therapeutic.”
He also described the case of a 72-year-old Black woman with total body itch for 2 years (WI-NRS = 10) and a history of seasonal allergies, thyroid disease, and hypertension. Previous therapies included prednisone, antihistamines, topical steroids, and gabapentin. The patient was found to have high IgE (447 kU/L) and eosinophil levels (4.9%), was started on methotrexate, and had an itch score of 0 at the 8-month follow-up.
JAK inhibitors may also have a role in the management of CPUO. A phase 2 nonrandomized controlled trial of abrocitinib for adults with prurigo nodularis (PN) or CPUO, recently published in JAMA Dermatology, showed itch scores decreased by 53.7% in the CPUO group (and 78.3% in the PN group) after 12 weeks of treatment with oral abrocitinib 200 mg daily. Patients had significant improvements in quality of life and no serious adverse events, said Dr. Kwatra, the lead author of the paper.
One of these patients was a 73-year-old White man who had experienced total body itch for 1.5 years (predominantly affecting his upper extremities; WI-NRS = 10) and a history of ascending aortic aneurysm, hypertension, and hyperlipidemia. Previous failed therapies included dupilumab (> 6 months), topical steroids, tacrolimus, and antihistamines. Labs showed elevated IgE (456 kU/L) and eosinophil levels (11.7%). After 12 weeks of treatment with abrocitinib, the WI-NRS decreased to 2.
PD-1 Inhibitors As a Trigger
Chronic pruritus caused by the anticancer PD-1 inhibitors is becoming more common as the utilization of these immune checkpoint inhibitors increases, Dr. Kwatra noted. “You don’t see much in the skin, but [these patients have] very high IgE and eosinophils,” he said. “We’ve been seeing more reports recently of utilizing agents that target type 2 inflammation off label for PD-1 inhibitor–related skin manifestations.”
One such patient with PD-1 inhibitor–induced pruritus was a 65-year-old White man with metastatic melanoma who reported a 6-month history of itching that began 3 weeks after the start of treatment with the PD-1 inhibitor pembrolizumab. His WI-NRS score was 10 despite treatment with topical steroids and antihistamines. He had a history of psoriasis. Labs showed elevated IgE (1350 kU/L) and eosinophil levels (4.5%). At a 4-month follow-up after treatment with off-label dupilumab (a 600-mg subcutaneous loading dose followed by 300 mg every 14 days), his WI-NRS score was 0.
In a paper recently published in JAAD International, Dr. Kwatra, Ms. Manjunath, and coinvestigators reported on a series of 15 patients who developed chronic pruritus following an immune stimulus exposure, including immunotherapy and vaccination (2024 Apr 7:16:97-102. doi: 10.1016/j.jdin.2024.03.022). Most immunotherapy-treated patients experienced pruritus during treatment or after 21-60 days of receiving treatment, and the patients with vaccine-stimulated pruritus (after Tdap and messenger RNA COVID-19 vaccination) developed pruritus within a week of vaccination.
In addition to the elevated levels of IgE and eosinophils, plasma cytokine analysis showed elevated levels of IL-5, thymic stromal lymphopoietin, and other Th2-related cytokines and inflammatory markers in patients with immune-stimulated pruritus compared with healthy controls, Ms. Manjunath said at the meeting.
When a Malignancy Workup Becomes Important
The initial part of any diagnostic workup for CPUO should include CBC with differential, liver function tests, renal function tests, and thyroid function testing, said Kwatra, referring to a diagnostic algorithm he developed, which was published as part of a CME review in the Journal of the American Academy of Dermatology in 2022.
Then, as indicated by risk factors in the history and physical, one could order other tests such as HIV serology, hepatitis B/C serologies, bullous pemphigoid testing, chest x-rays, evaluation for gammopathies, stool examination for ova and parasites, or heavy metal testing. “Do you do everything at once? We like to keep it straightforward,” Dr. Kwatra said. “Depending on the patient’s risk factors, you could order more or less.”
A malignancy workup should be strongly considered in patients whose itch duration is less than 12 months — and especially if the duration is less than 3 months — with an emphasis on cancers more frequently associated with itch: Hematologic and hepatobiliary cancers. This is “when concern should be heightened ... when there should be a lower threshold for workup,” he said.
The 12-month recommendation stems from a Danish cohort study published in 2014 that demonstrated a twofold increased incidence of cancer among patients with pruritus in the first 3 months after the diagnosis of pruritus. The 1-year absolute cancer risk was 1.63%.
Other risk factors for underlying malignancy or malignancy development in patients with CPUO include age older than 60 years, male sex, liver disease, and current or prior smoking, according to another study, noted Dr. Kwatra.
Dr. Kwatra disclosed that he is an advisory board member/consultant for Pfizer, Regeneron, Sanofi, and other companies and an investigator for Galderma, Incyte, Pfizer, and Sanofi. Manjunath served as the codirector of the ElderDerm conference.
A version of this article first appeared on Medscape.com.
WASHINGTON — , Shawn G. Kwatra, MD, said at the ElderDerm conference on dermatology in older patients hosted by the GW School of Medicine & Health Sciences.
“We found a few years ago that eosinophils seem to differentiate this group, and now we’re finding that IgE and CBC [complete blood count] differential can help you get a little better sense of who has an immune-driven itch vs something more neuropathic,” said Dr. Kwatra, professor and chair of dermatology at the University of Maryland, Baltimore, who founded and directed the Johns Hopkins Itch Center before coming to the University of Maryland in 2023. Not all patients with immune-driven itch will have these biomarkers, “but it’s a helpful tool,” he said.
CPUO is the term that is increasingly being used, he said, to describe intense, chronic pruritus without primary skin lesions or rashes and without any known systemic cause. It becomes more common as people get older and is sometimes debilitating. The initial evaluation should be kept “simple and straightforward,” he advised, with heightened concern for underlying malignancy in those who present with an itch of less than 12 months’ duration.
Biologics, JAK Inhibitors: Case Reports, Ongoing Research
Research conducted by Dr. Kwatra and Jaya Manjunath, a fourth-year medical student at The George Washington University, Washington, documented higher levels of Th2-associated cytokines and inflammatory markers in patients with CPUO who had elevated IgE or eosinophil levels, or both than in patients with itch who had low IgE and eosinophil levels. The patients with higher levels also had a greater response to off-label treatment with immunomodulatory therapy.
“Multiple Th2-related inflammatory markers, like IL [interleukin]-5 and eotaxin-3, were reduced after dupilumab” in patients who responded to the therapy, said Ms. Manjunath, who co-presented the meeting session on chronic itch with Dr. Kwatra. Other changes in the plasma cytokine profile included a reduction in the serum level of thymus and activation-regulated chemokine, which is a biomarker for atopic dermatitis. The research is under review for publication.
Meanwhile, a phase 3 trial (LIBERTY-CPUO-CHIC) of dupilumab for CPUO is currently underway, Dr. Kwatra noted. Investigators are randomizing patients with severe pruritus (Worst Itch Numeric Rating Scale [WI-NRS] ≥ 7) to dupilumab or placebo for 12 or 24 weeks.
In one of several cases shared by Dr. Kwatra and Ms. Manjunath, a 71-year-old Black woman with a 6-month history of generalized itch (WI-NRS = 10) and a history of type 2 diabetes, hypertension, and chronic kidney disease was found to have elevated eosinophil levels and a negative malignancy workup. Previous therapies included antihistamines and topical steroids. She was started on a 600-mg loading dose of subcutaneous dupilumab followed by 300 mg every 14 days. At the 2-month follow-up, her WI-NRS score was 0.
Because “dupilumab is off label right now for this form of itch, oftentimes our first line is methotrexate,” Dr. Kwatra said. Patients “can have a good response with this therapeutic.”
He also described the case of a 72-year-old Black woman with total body itch for 2 years (WI-NRS = 10) and a history of seasonal allergies, thyroid disease, and hypertension. Previous therapies included prednisone, antihistamines, topical steroids, and gabapentin. The patient was found to have high IgE (447 kU/L) and eosinophil levels (4.9%), was started on methotrexate, and had an itch score of 0 at the 8-month follow-up.
JAK inhibitors may also have a role in the management of CPUO. A phase 2 nonrandomized controlled trial of abrocitinib for adults with prurigo nodularis (PN) or CPUO, recently published in JAMA Dermatology, showed itch scores decreased by 53.7% in the CPUO group (and 78.3% in the PN group) after 12 weeks of treatment with oral abrocitinib 200 mg daily. Patients had significant improvements in quality of life and no serious adverse events, said Dr. Kwatra, the lead author of the paper.
One of these patients was a 73-year-old White man who had experienced total body itch for 1.5 years (predominantly affecting his upper extremities; WI-NRS = 10) and a history of ascending aortic aneurysm, hypertension, and hyperlipidemia. Previous failed therapies included dupilumab (> 6 months), topical steroids, tacrolimus, and antihistamines. Labs showed elevated IgE (456 kU/L) and eosinophil levels (11.7%). After 12 weeks of treatment with abrocitinib, the WI-NRS decreased to 2.
PD-1 Inhibitors As a Trigger
Chronic pruritus caused by the anticancer PD-1 inhibitors is becoming more common as the utilization of these immune checkpoint inhibitors increases, Dr. Kwatra noted. “You don’t see much in the skin, but [these patients have] very high IgE and eosinophils,” he said. “We’ve been seeing more reports recently of utilizing agents that target type 2 inflammation off label for PD-1 inhibitor–related skin manifestations.”
One such patient with PD-1 inhibitor–induced pruritus was a 65-year-old White man with metastatic melanoma who reported a 6-month history of itching that began 3 weeks after the start of treatment with the PD-1 inhibitor pembrolizumab. His WI-NRS score was 10 despite treatment with topical steroids and antihistamines. He had a history of psoriasis. Labs showed elevated IgE (1350 kU/L) and eosinophil levels (4.5%). At a 4-month follow-up after treatment with off-label dupilumab (a 600-mg subcutaneous loading dose followed by 300 mg every 14 days), his WI-NRS score was 0.
In a paper recently published in JAAD International, Dr. Kwatra, Ms. Manjunath, and coinvestigators reported on a series of 15 patients who developed chronic pruritus following an immune stimulus exposure, including immunotherapy and vaccination (2024 Apr 7:16:97-102. doi: 10.1016/j.jdin.2024.03.022). Most immunotherapy-treated patients experienced pruritus during treatment or after 21-60 days of receiving treatment, and the patients with vaccine-stimulated pruritus (after Tdap and messenger RNA COVID-19 vaccination) developed pruritus within a week of vaccination.
In addition to the elevated levels of IgE and eosinophils, plasma cytokine analysis showed elevated levels of IL-5, thymic stromal lymphopoietin, and other Th2-related cytokines and inflammatory markers in patients with immune-stimulated pruritus compared with healthy controls, Ms. Manjunath said at the meeting.
When a Malignancy Workup Becomes Important
The initial part of any diagnostic workup for CPUO should include CBC with differential, liver function tests, renal function tests, and thyroid function testing, said Kwatra, referring to a diagnostic algorithm he developed, which was published as part of a CME review in the Journal of the American Academy of Dermatology in 2022.
Then, as indicated by risk factors in the history and physical, one could order other tests such as HIV serology, hepatitis B/C serologies, bullous pemphigoid testing, chest x-rays, evaluation for gammopathies, stool examination for ova and parasites, or heavy metal testing. “Do you do everything at once? We like to keep it straightforward,” Dr. Kwatra said. “Depending on the patient’s risk factors, you could order more or less.”
A malignancy workup should be strongly considered in patients whose itch duration is less than 12 months — and especially if the duration is less than 3 months — with an emphasis on cancers more frequently associated with itch: Hematologic and hepatobiliary cancers. This is “when concern should be heightened ... when there should be a lower threshold for workup,” he said.
The 12-month recommendation stems from a Danish cohort study published in 2014 that demonstrated a twofold increased incidence of cancer among patients with pruritus in the first 3 months after the diagnosis of pruritus. The 1-year absolute cancer risk was 1.63%.
Other risk factors for underlying malignancy or malignancy development in patients with CPUO include age older than 60 years, male sex, liver disease, and current or prior smoking, according to another study, noted Dr. Kwatra.
Dr. Kwatra disclosed that he is an advisory board member/consultant for Pfizer, Regeneron, Sanofi, and other companies and an investigator for Galderma, Incyte, Pfizer, and Sanofi. Manjunath served as the codirector of the ElderDerm conference.
A version of this article first appeared on Medscape.com.
WASHINGTON — , Shawn G. Kwatra, MD, said at the ElderDerm conference on dermatology in older patients hosted by the GW School of Medicine & Health Sciences.
“We found a few years ago that eosinophils seem to differentiate this group, and now we’re finding that IgE and CBC [complete blood count] differential can help you get a little better sense of who has an immune-driven itch vs something more neuropathic,” said Dr. Kwatra, professor and chair of dermatology at the University of Maryland, Baltimore, who founded and directed the Johns Hopkins Itch Center before coming to the University of Maryland in 2023. Not all patients with immune-driven itch will have these biomarkers, “but it’s a helpful tool,” he said.
CPUO is the term that is increasingly being used, he said, to describe intense, chronic pruritus without primary skin lesions or rashes and without any known systemic cause. It becomes more common as people get older and is sometimes debilitating. The initial evaluation should be kept “simple and straightforward,” he advised, with heightened concern for underlying malignancy in those who present with an itch of less than 12 months’ duration.
Biologics, JAK Inhibitors: Case Reports, Ongoing Research
Research conducted by Dr. Kwatra and Jaya Manjunath, a fourth-year medical student at The George Washington University, Washington, documented higher levels of Th2-associated cytokines and inflammatory markers in patients with CPUO who had elevated IgE or eosinophil levels, or both than in patients with itch who had low IgE and eosinophil levels. The patients with higher levels also had a greater response to off-label treatment with immunomodulatory therapy.
“Multiple Th2-related inflammatory markers, like IL [interleukin]-5 and eotaxin-3, were reduced after dupilumab” in patients who responded to the therapy, said Ms. Manjunath, who co-presented the meeting session on chronic itch with Dr. Kwatra. Other changes in the plasma cytokine profile included a reduction in the serum level of thymus and activation-regulated chemokine, which is a biomarker for atopic dermatitis. The research is under review for publication.
Meanwhile, a phase 3 trial (LIBERTY-CPUO-CHIC) of dupilumab for CPUO is currently underway, Dr. Kwatra noted. Investigators are randomizing patients with severe pruritus (Worst Itch Numeric Rating Scale [WI-NRS] ≥ 7) to dupilumab or placebo for 12 or 24 weeks.
In one of several cases shared by Dr. Kwatra and Ms. Manjunath, a 71-year-old Black woman with a 6-month history of generalized itch (WI-NRS = 10) and a history of type 2 diabetes, hypertension, and chronic kidney disease was found to have elevated eosinophil levels and a negative malignancy workup. Previous therapies included antihistamines and topical steroids. She was started on a 600-mg loading dose of subcutaneous dupilumab followed by 300 mg every 14 days. At the 2-month follow-up, her WI-NRS score was 0.
Because “dupilumab is off label right now for this form of itch, oftentimes our first line is methotrexate,” Dr. Kwatra said. Patients “can have a good response with this therapeutic.”
He also described the case of a 72-year-old Black woman with total body itch for 2 years (WI-NRS = 10) and a history of seasonal allergies, thyroid disease, and hypertension. Previous therapies included prednisone, antihistamines, topical steroids, and gabapentin. The patient was found to have high IgE (447 kU/L) and eosinophil levels (4.9%), was started on methotrexate, and had an itch score of 0 at the 8-month follow-up.
JAK inhibitors may also have a role in the management of CPUO. A phase 2 nonrandomized controlled trial of abrocitinib for adults with prurigo nodularis (PN) or CPUO, recently published in JAMA Dermatology, showed itch scores decreased by 53.7% in the CPUO group (and 78.3% in the PN group) after 12 weeks of treatment with oral abrocitinib 200 mg daily. Patients had significant improvements in quality of life and no serious adverse events, said Dr. Kwatra, the lead author of the paper.
One of these patients was a 73-year-old White man who had experienced total body itch for 1.5 years (predominantly affecting his upper extremities; WI-NRS = 10) and a history of ascending aortic aneurysm, hypertension, and hyperlipidemia. Previous failed therapies included dupilumab (> 6 months), topical steroids, tacrolimus, and antihistamines. Labs showed elevated IgE (456 kU/L) and eosinophil levels (11.7%). After 12 weeks of treatment with abrocitinib, the WI-NRS decreased to 2.
PD-1 Inhibitors As a Trigger
Chronic pruritus caused by the anticancer PD-1 inhibitors is becoming more common as the utilization of these immune checkpoint inhibitors increases, Dr. Kwatra noted. “You don’t see much in the skin, but [these patients have] very high IgE and eosinophils,” he said. “We’ve been seeing more reports recently of utilizing agents that target type 2 inflammation off label for PD-1 inhibitor–related skin manifestations.”
One such patient with PD-1 inhibitor–induced pruritus was a 65-year-old White man with metastatic melanoma who reported a 6-month history of itching that began 3 weeks after the start of treatment with the PD-1 inhibitor pembrolizumab. His WI-NRS score was 10 despite treatment with topical steroids and antihistamines. He had a history of psoriasis. Labs showed elevated IgE (1350 kU/L) and eosinophil levels (4.5%). At a 4-month follow-up after treatment with off-label dupilumab (a 600-mg subcutaneous loading dose followed by 300 mg every 14 days), his WI-NRS score was 0.
In a paper recently published in JAAD International, Dr. Kwatra, Ms. Manjunath, and coinvestigators reported on a series of 15 patients who developed chronic pruritus following an immune stimulus exposure, including immunotherapy and vaccination (2024 Apr 7:16:97-102. doi: 10.1016/j.jdin.2024.03.022). Most immunotherapy-treated patients experienced pruritus during treatment or after 21-60 days of receiving treatment, and the patients with vaccine-stimulated pruritus (after Tdap and messenger RNA COVID-19 vaccination) developed pruritus within a week of vaccination.
In addition to the elevated levels of IgE and eosinophils, plasma cytokine analysis showed elevated levels of IL-5, thymic stromal lymphopoietin, and other Th2-related cytokines and inflammatory markers in patients with immune-stimulated pruritus compared with healthy controls, Ms. Manjunath said at the meeting.
When a Malignancy Workup Becomes Important
The initial part of any diagnostic workup for CPUO should include CBC with differential, liver function tests, renal function tests, and thyroid function testing, said Kwatra, referring to a diagnostic algorithm he developed, which was published as part of a CME review in the Journal of the American Academy of Dermatology in 2022.
Then, as indicated by risk factors in the history and physical, one could order other tests such as HIV serology, hepatitis B/C serologies, bullous pemphigoid testing, chest x-rays, evaluation for gammopathies, stool examination for ova and parasites, or heavy metal testing. “Do you do everything at once? We like to keep it straightforward,” Dr. Kwatra said. “Depending on the patient’s risk factors, you could order more or less.”
A malignancy workup should be strongly considered in patients whose itch duration is less than 12 months — and especially if the duration is less than 3 months — with an emphasis on cancers more frequently associated with itch: Hematologic and hepatobiliary cancers. This is “when concern should be heightened ... when there should be a lower threshold for workup,” he said.
The 12-month recommendation stems from a Danish cohort study published in 2014 that demonstrated a twofold increased incidence of cancer among patients with pruritus in the first 3 months after the diagnosis of pruritus. The 1-year absolute cancer risk was 1.63%.
Other risk factors for underlying malignancy or malignancy development in patients with CPUO include age older than 60 years, male sex, liver disease, and current or prior smoking, according to another study, noted Dr. Kwatra.
Dr. Kwatra disclosed that he is an advisory board member/consultant for Pfizer, Regeneron, Sanofi, and other companies and an investigator for Galderma, Incyte, Pfizer, and Sanofi. Manjunath served as the codirector of the ElderDerm conference.
A version of this article first appeared on Medscape.com.
FROM ELDERDERM 2024
Several Skin Conditions More Likely in Children With Obesity
TORONTO — results of new research show.
The retrospective cohort study found markedly higher rates of skin infections, atopic dermatitis (AD), and acanthosis nigricans among children with overweight, compared with children with average weight.
“Many conditions associated with obesity are strong predictors of cardiovascular mortality as these children age, so doctors can play a key role in advocating for weight loss strategies in this population,” lead study author Samantha Epstein, third-year medical student at Case Western Reserve University, Cleveland, Ohio, said in an interview. The findings were presented at the annual meeting of the Society for Pediatric Dermatology.
Previous research has linked obesity, a chronic inflammatory condition, to psoriasis, AD, hidradenitis suppurativa (HS), acne vulgaris, infections, and rosacea in adults. However, there’s scant research exploring the connection between obesity and cutaneous conditions in children.
According to the Cleveland Clinic, childhood obesity is defined as a body mass index, which is weight in kg divided by the square of height in m2, at or above the 95th percentile for age and sex in children aged 2 years or older.
For the study, Ms. Epstein and coauthor Sonal D. Shah, MD, associate professor, Department of Dermatology, Case Western Reserve University, and a board-certified pediatric dermatologist accessed a large national research database and used diagnostic codes to identify over 1 million children (mean age, 8.5 years). Most (about 44%) were White; about one-quarter were Black. The groups were propensity matched, so there were about equal numbers of youngsters with and without obesity and of boys and girls.
They collected data on AD, HS, rosacea, psoriasis, and acanthosis nigricans (a thickened purplish discoloration typically found in body folds around the armpits, groin, and neck). They also gathered information on comorbidities.
Acanthosis nigricans, which is linked to metabolic syndrome, type 2 diabetes, and insulin resistance , was more prevalent among children with obesity (20,885 cases in the with-obesity group and 336 in the without-obesity group, for a relative risk [RR] of 62.16 and an odds ratio [OR] of 64.38).
Skin and subcutaneous tissue infections were also more common among those with obesity (14,795 cases) vs 4720 cases among those without obesity (RR, 3.14; OR, 3.2). As for AD, there were 11,892 cases in the with-obesity group and 2983 in the without-obesity group (RR, 3.99; OR, 4.06). There were 1166 cases of psoriasis among those with obesity and 408 among those without obesity (RR, 2.86; OR, 2.88).
HS (587 cases in the with-obesity group and 70 in the without-obesity group; RR, 8.39; OR, 8.39) and rosacea (351 in the with-obesity group and 138 in the without-obesity group; RR, 2.54; OR, 2.55) were the least common skin conditions.
Higher Comorbidity Rates
Compared with their average-weight counterparts, the children with obesity had higher rates of comorbidities, including type 2 diabetes. Ms. Epstein noted that children with diabetes and obesity had increased risks for every skin condition except for infections of the skin and subcutaneous tissue when compared with children without obesity.
Such infections were the most common skin conditions among children without obesity. “This was expected just due to the fact that children are outside, they’re playing in the grass and the dirt, and they get infected,” said Ms. Epstein. Still, these infections were three times more common in youngsters with obesity.
Although acanthosis nigricans is “highly correlated” with type 2 diabetes, “not as many children as we would expect in this population have developed type 2 diabetes,” said Ms. Epstein. This might make some sense, though, because these children are still quite young. “When dermatologists recognize this skin condition, they can advocate for weight loss management to try to prevent it.”
Other conditions seen more often in the overweight children with overweight included: hypertension, hyperlipidemia, obstructive sleep apnea, polycystic ovarian syndrome, attention-deficit/hyperactivity disorder, major depressive disorder, depressive episodes, and anxiety (all P < .001).
Commenting on the results, Sonia Havele, MD, a pediatrician and dermatology resident at Children’s Mercy Hospital, Kansas City, Missouri, said in an interview that the study reflects trends that she and her colleagues see in clinic: There are more common skin conditions in their patients with obesity.
She agreed that it offers an opening for education. “The results of this study highlight the opportunity we have as pediatric dermatologists to provide additional counseling on obesity and offer referrals to our colleagues in endocrinology, gastroenterology, and nutrition if needed.”
No conflicts of interest were reported.
TORONTO — results of new research show.
The retrospective cohort study found markedly higher rates of skin infections, atopic dermatitis (AD), and acanthosis nigricans among children with overweight, compared with children with average weight.
“Many conditions associated with obesity are strong predictors of cardiovascular mortality as these children age, so doctors can play a key role in advocating for weight loss strategies in this population,” lead study author Samantha Epstein, third-year medical student at Case Western Reserve University, Cleveland, Ohio, said in an interview. The findings were presented at the annual meeting of the Society for Pediatric Dermatology.
Previous research has linked obesity, a chronic inflammatory condition, to psoriasis, AD, hidradenitis suppurativa (HS), acne vulgaris, infections, and rosacea in adults. However, there’s scant research exploring the connection between obesity and cutaneous conditions in children.
According to the Cleveland Clinic, childhood obesity is defined as a body mass index, which is weight in kg divided by the square of height in m2, at or above the 95th percentile for age and sex in children aged 2 years or older.
For the study, Ms. Epstein and coauthor Sonal D. Shah, MD, associate professor, Department of Dermatology, Case Western Reserve University, and a board-certified pediatric dermatologist accessed a large national research database and used diagnostic codes to identify over 1 million children (mean age, 8.5 years). Most (about 44%) were White; about one-quarter were Black. The groups were propensity matched, so there were about equal numbers of youngsters with and without obesity and of boys and girls.
They collected data on AD, HS, rosacea, psoriasis, and acanthosis nigricans (a thickened purplish discoloration typically found in body folds around the armpits, groin, and neck). They also gathered information on comorbidities.
Acanthosis nigricans, which is linked to metabolic syndrome, type 2 diabetes, and insulin resistance , was more prevalent among children with obesity (20,885 cases in the with-obesity group and 336 in the without-obesity group, for a relative risk [RR] of 62.16 and an odds ratio [OR] of 64.38).
Skin and subcutaneous tissue infections were also more common among those with obesity (14,795 cases) vs 4720 cases among those without obesity (RR, 3.14; OR, 3.2). As for AD, there were 11,892 cases in the with-obesity group and 2983 in the without-obesity group (RR, 3.99; OR, 4.06). There were 1166 cases of psoriasis among those with obesity and 408 among those without obesity (RR, 2.86; OR, 2.88).
HS (587 cases in the with-obesity group and 70 in the without-obesity group; RR, 8.39; OR, 8.39) and rosacea (351 in the with-obesity group and 138 in the without-obesity group; RR, 2.54; OR, 2.55) were the least common skin conditions.
Higher Comorbidity Rates
Compared with their average-weight counterparts, the children with obesity had higher rates of comorbidities, including type 2 diabetes. Ms. Epstein noted that children with diabetes and obesity had increased risks for every skin condition except for infections of the skin and subcutaneous tissue when compared with children without obesity.
Such infections were the most common skin conditions among children without obesity. “This was expected just due to the fact that children are outside, they’re playing in the grass and the dirt, and they get infected,” said Ms. Epstein. Still, these infections were three times more common in youngsters with obesity.
Although acanthosis nigricans is “highly correlated” with type 2 diabetes, “not as many children as we would expect in this population have developed type 2 diabetes,” said Ms. Epstein. This might make some sense, though, because these children are still quite young. “When dermatologists recognize this skin condition, they can advocate for weight loss management to try to prevent it.”
Other conditions seen more often in the overweight children with overweight included: hypertension, hyperlipidemia, obstructive sleep apnea, polycystic ovarian syndrome, attention-deficit/hyperactivity disorder, major depressive disorder, depressive episodes, and anxiety (all P < .001).
Commenting on the results, Sonia Havele, MD, a pediatrician and dermatology resident at Children’s Mercy Hospital, Kansas City, Missouri, said in an interview that the study reflects trends that she and her colleagues see in clinic: There are more common skin conditions in their patients with obesity.
She agreed that it offers an opening for education. “The results of this study highlight the opportunity we have as pediatric dermatologists to provide additional counseling on obesity and offer referrals to our colleagues in endocrinology, gastroenterology, and nutrition if needed.”
No conflicts of interest were reported.
TORONTO — results of new research show.
The retrospective cohort study found markedly higher rates of skin infections, atopic dermatitis (AD), and acanthosis nigricans among children with overweight, compared with children with average weight.
“Many conditions associated with obesity are strong predictors of cardiovascular mortality as these children age, so doctors can play a key role in advocating for weight loss strategies in this population,” lead study author Samantha Epstein, third-year medical student at Case Western Reserve University, Cleveland, Ohio, said in an interview. The findings were presented at the annual meeting of the Society for Pediatric Dermatology.
Previous research has linked obesity, a chronic inflammatory condition, to psoriasis, AD, hidradenitis suppurativa (HS), acne vulgaris, infections, and rosacea in adults. However, there’s scant research exploring the connection between obesity and cutaneous conditions in children.
According to the Cleveland Clinic, childhood obesity is defined as a body mass index, which is weight in kg divided by the square of height in m2, at or above the 95th percentile for age and sex in children aged 2 years or older.
For the study, Ms. Epstein and coauthor Sonal D. Shah, MD, associate professor, Department of Dermatology, Case Western Reserve University, and a board-certified pediatric dermatologist accessed a large national research database and used diagnostic codes to identify over 1 million children (mean age, 8.5 years). Most (about 44%) were White; about one-quarter were Black. The groups were propensity matched, so there were about equal numbers of youngsters with and without obesity and of boys and girls.
They collected data on AD, HS, rosacea, psoriasis, and acanthosis nigricans (a thickened purplish discoloration typically found in body folds around the armpits, groin, and neck). They also gathered information on comorbidities.
Acanthosis nigricans, which is linked to metabolic syndrome, type 2 diabetes, and insulin resistance , was more prevalent among children with obesity (20,885 cases in the with-obesity group and 336 in the without-obesity group, for a relative risk [RR] of 62.16 and an odds ratio [OR] of 64.38).
Skin and subcutaneous tissue infections were also more common among those with obesity (14,795 cases) vs 4720 cases among those without obesity (RR, 3.14; OR, 3.2). As for AD, there were 11,892 cases in the with-obesity group and 2983 in the without-obesity group (RR, 3.99; OR, 4.06). There were 1166 cases of psoriasis among those with obesity and 408 among those without obesity (RR, 2.86; OR, 2.88).
HS (587 cases in the with-obesity group and 70 in the without-obesity group; RR, 8.39; OR, 8.39) and rosacea (351 in the with-obesity group and 138 in the without-obesity group; RR, 2.54; OR, 2.55) were the least common skin conditions.
Higher Comorbidity Rates
Compared with their average-weight counterparts, the children with obesity had higher rates of comorbidities, including type 2 diabetes. Ms. Epstein noted that children with diabetes and obesity had increased risks for every skin condition except for infections of the skin and subcutaneous tissue when compared with children without obesity.
Such infections were the most common skin conditions among children without obesity. “This was expected just due to the fact that children are outside, they’re playing in the grass and the dirt, and they get infected,” said Ms. Epstein. Still, these infections were three times more common in youngsters with obesity.
Although acanthosis nigricans is “highly correlated” with type 2 diabetes, “not as many children as we would expect in this population have developed type 2 diabetes,” said Ms. Epstein. This might make some sense, though, because these children are still quite young. “When dermatologists recognize this skin condition, they can advocate for weight loss management to try to prevent it.”
Other conditions seen more often in the overweight children with overweight included: hypertension, hyperlipidemia, obstructive sleep apnea, polycystic ovarian syndrome, attention-deficit/hyperactivity disorder, major depressive disorder, depressive episodes, and anxiety (all P < .001).
Commenting on the results, Sonia Havele, MD, a pediatrician and dermatology resident at Children’s Mercy Hospital, Kansas City, Missouri, said in an interview that the study reflects trends that she and her colleagues see in clinic: There are more common skin conditions in their patients with obesity.
She agreed that it offers an opening for education. “The results of this study highlight the opportunity we have as pediatric dermatologists to provide additional counseling on obesity and offer referrals to our colleagues in endocrinology, gastroenterology, and nutrition if needed.”
No conflicts of interest were reported.
FROM SPD 2024
Dermatoporosis in Older Adults: A Condition That Requires Holistic, Creative Management
WASHINGTON — and conveys the skin’s vulnerability to serious medical complications, said Adam Friedman, MD, at the ElderDerm conference on dermatology in the older patient.
Key features of dermatoporosis include atrophic skin, solar purpura, white pseudoscars, easily acquired skin lacerations and tears, bruises, and delayed healing. “We’re going to see more of this, and it will more and more be a chief complaint of patients,” said Dr. Friedman, professor and chair of dermatology at George Washington University (GWU) in Washington, and co-chair of the meeting. GWU hosted the conference, describing it as a first-of-its-kind meeting dedicated to improving dermatologic care for older adults.
Dermatoporosis was described in the literature in 2007 by dermatologists at the University of Geneva in Switzerland. “It is not only a cosmetic problem,” Dr. Friedman said. “This is a medical problem ... which can absolutely lead to comorbidities [such as deep dissecting hematomas] that are a huge strain on the healthcare system.”
Dermatologists can meet the moment with holistic, creative combination treatment and counseling approaches aimed at improving the mechanical strength of skin and preventing potential complications in older patients, Dr. Friedman said at the meeting.
He described the case of a 76-year-old woman who presented with dermatoporosis on her arms involving pronounced skin atrophy, solar purpura, and a small covered laceration. “This was a patient who was both devastated by the appearance” and impacted by the pain and burden of dressing frequent wounds, said Dr. Friedman, who is also the director of the Residency Program, of Translational Research, and of Supportive Oncodermatology, all within the Department of Dermatology at GWU.
With 11 months of topical treatment that included daily application of calcipotriene 0.05% ointment and nightly application of tazarotene 0.045% lotion and oral supplementation with 1000-mg vitamin C twice daily and 1000-mg citrus bioflavonoid complex daily, as well as no changes to the medications she took for various comorbidities, the solar purpura improved significantly and “we made a huge difference in the integrity of her skin,” he said.
Dr. Friedman also described this case in a recently published article in the Journal of Drugs in Dermatology titled “What’s Old Is New: An Emerging Focus on Dermatoporosis”.
Likely Pathophysiology
Advancing age and chronic ultraviolet (UV) radiation exposure are the chief drivers of dermatoporosis. In addition to UVA and UVB light, other secondary drivers include genetic susceptibility, topical and systematic corticosteroid use, and anticoagulant treatment.
Its pathogenesis is not well described in the literature but is easy to envision, Dr. Friedman said. For one, both advancing age and exposure to UV light lead to a reduction in hygroscopic glycosaminoglycans, including hyaluronate (HA), and the impact of this diminishment is believed to go “beyond [the loss of] buoyancy,” he noted. Researchers have “been showing these are not just water-loving molecules, they also have some biologic properties” relating to keratinocyte production and epidermal turnover that appear to be intricately linked to the pathogenesis of dermatoporosis.
HAs have been shown to interact with the cell surface receptor CD44 to stimulate keratinocyte proliferation, and low levels of CD44 have been reported in skin with dermatoporosis compared with a younger control population. (A newly characterized organelle, the hyaluronosome, serves as an HA factory and contains CD44 and heparin-binding epidermal growth factor, Dr. Friedman noted. Inadequate functioning may be involved in skin atrophy.)
Advancing age also brings an increase in matrix metalloproteinases (MMPs)–1, –2, and –3, which are “the demolition workers of the skin,” and downregulation of a tissue inhibitor of MMPs, he said.
Adding insult to injury, dermis-penetrating UVA also activates MMPs, “obliterating collagen and elastin.” UVB generates DNA photoproducts, including oxidative stress and damaging skin cell DNA. “That UV light induces breakdown [of the skin] through different mechanisms and inhibits buildup is a simple concept I think our patients can understand,” Dr. Friedman said.
Multifaceted Treatment
For an older adult, “there is never a wrong time to start sun-protective measures” to prevent or try to halt the progression of dermatoporosis, Dr. Friedman said, noting that “UV radiation is an immunosuppressant, so there are many good reasons to start” if the adult is not already taking measures on a regular basis.
Potential treatments for the syndrome of dermatoporosis are backed by few clinical studies, but dermatologists are skilled at translating the use of products from one disease state to another based on understandings of pathophysiology and mechanistic pathways, Dr. Friedman commented in an interview after the meeting.
For instance, “from decades of research, we know what retinoids will do to the skin,” he said in the interview. “We know they will turn on collagen-1 and -3 genes in the skin, and that they will increase the production of glycosaminoglycans ... By understanding the biology, we can translate this to dermatoporosis.” These changes were demonstrated, for instance, in a small study of topical retinol in older adults.
Studies of topical alpha hydroxy acid (AHA), moreover, have demonstrated epidermal thickening and firmness, and “some studies show they can limit steroid-induced atrophy,” Dr. Friedman said at the meeting. “And things like lactic acid and urea are super accessible.”
Topical dehydroepiandrosterone is backed by even less data than retinoids or AHAs are, “but it’s still something to consider” as part of a multimechanistic approach to dermatoporosis, Dr. Friedman shared, noting that a small study demonstrated beneficial effects on epidermal atrophy in aging skin.
The use of vitamin D analogues such as calcipotriene, which is approved for the treatment of psoriasis, may also be promising. “One concept is that [vitamin D analogues] increase calcium concentrations in the epidermis, and calcium is so central to keratinocyte differentiation” and epidermal function that calcipotriene in combination with topical steroid therapy has been shown to limit skin atrophy, he noted.
Nutritionally, low protein intake is a known problem in the older population and is associated with increased skin fragility and poorer healing. From a prevention and treatment standpoint, therefore, patients can be counseled to be attentive to their diets, Dr. Friedman said. Experts have recommended a higher protein intake for older adults than for younger adults; in 2013, an international group recommended a protein intake of 1-1.5 g/kg/d for healthy older adults and more for those with acute or chronic illness.
“Patients love talking about diet and skin disease ... and they love over-the-counter nutraceuticals as well because they want something natural,” Dr. Friedman said. “I like using bioflavonoids in combination with vitamin C, which can be effective especially for solar purpura.”
A 6-week randomized, placebo-controlled, double-blind trial involving 67 patients with purpura associated with aging found a 50% reduction in purpura lesions among those took a particular citrus bioflavonoid blend twice daily. “I thought this was a pretty well-done study,” he said, noting that patient self-assessment and investigator global assessment were utilized.
Skin Injury and Wound Prevention
In addition to recommending gentle skin cleansers and daily moisturizing, dermatologists should talk to their older patients with dermatoporosis about their home environments. “What is it like? Is there furniture with sharp edges?” Dr. Friedman advised. If so, could they use sleeves or protectors on their arms or legs “to protect against injury?”
In a later meeting session about lower-extremity wounds on geriatric patients, Michael Stempel, DPM, assistant professor of medicine and surgery and chief of podiatry at GWU, said that he was happy to hear the term dermatoporosis being used because like diabetes, it’s a risk factor for developing lower-extremity wounds and poor wound healing.
He shared the case of an older woman with dermatoporosis who “tripped and skinned her knee against a step and then self-treated it for over a month by pouring hydrogen peroxide over it and letting air get to it.” The wound developed into “full-thickness tissue loss,” said Dr. Stempel, also medical director of the Wound Healing and Limb Preservation Center at GWU Hospital.
Misperceptions are common among older patients about how a simple wound should be managed; for instance, the adage “just let it get air” is not uncommon. This makes anticipatory guidance about basic wound care — such as the importance of a moist and occlusive environment and the safe use of hydrogen peroxide — especially important for patients with dermatoporosis, Dr. Friedman commented after the meeting.
Dermatoporosis is quantifiable, Dr. Friedman said during the meeting, with a scoring system having been developed by the researchers in Switzerland who originally coined the term. Its use in practice is unnecessary, but its existence is “nice to share with patients who feel bothered because oftentimes, patients feel it’s been dismissed by other providers,” he said. “Telling your patients there’s an actual name for their problem, and that there are ways to quantify and measure changes over time, is validating.”
Its recognition as a medical condition, Dr. Friedman added, also enables the dermatologist to bring it up and counsel appropriately — without a patient feeling shame — when it is identified in the context of a skin excision, treatment of a primary inflammatory skin disease, or management of another dermatologic problem.
Dr. Friedman disclosed that he is a consultant/advisory board member for L’Oréal, La Roche-Posay, Galderma, and other companies; a speaker for Regeneron/Sanofi, Incyte, BMD, and Janssen; and has grants from Pfizer, Lilly, Incyte, and other companies. Dr. Stempel reported no disclosures.
A version of this article first appeared on Medscape.com.
WASHINGTON — and conveys the skin’s vulnerability to serious medical complications, said Adam Friedman, MD, at the ElderDerm conference on dermatology in the older patient.
Key features of dermatoporosis include atrophic skin, solar purpura, white pseudoscars, easily acquired skin lacerations and tears, bruises, and delayed healing. “We’re going to see more of this, and it will more and more be a chief complaint of patients,” said Dr. Friedman, professor and chair of dermatology at George Washington University (GWU) in Washington, and co-chair of the meeting. GWU hosted the conference, describing it as a first-of-its-kind meeting dedicated to improving dermatologic care for older adults.
Dermatoporosis was described in the literature in 2007 by dermatologists at the University of Geneva in Switzerland. “It is not only a cosmetic problem,” Dr. Friedman said. “This is a medical problem ... which can absolutely lead to comorbidities [such as deep dissecting hematomas] that are a huge strain on the healthcare system.”
Dermatologists can meet the moment with holistic, creative combination treatment and counseling approaches aimed at improving the mechanical strength of skin and preventing potential complications in older patients, Dr. Friedman said at the meeting.
He described the case of a 76-year-old woman who presented with dermatoporosis on her arms involving pronounced skin atrophy, solar purpura, and a small covered laceration. “This was a patient who was both devastated by the appearance” and impacted by the pain and burden of dressing frequent wounds, said Dr. Friedman, who is also the director of the Residency Program, of Translational Research, and of Supportive Oncodermatology, all within the Department of Dermatology at GWU.
With 11 months of topical treatment that included daily application of calcipotriene 0.05% ointment and nightly application of tazarotene 0.045% lotion and oral supplementation with 1000-mg vitamin C twice daily and 1000-mg citrus bioflavonoid complex daily, as well as no changes to the medications she took for various comorbidities, the solar purpura improved significantly and “we made a huge difference in the integrity of her skin,” he said.
Dr. Friedman also described this case in a recently published article in the Journal of Drugs in Dermatology titled “What’s Old Is New: An Emerging Focus on Dermatoporosis”.
Likely Pathophysiology
Advancing age and chronic ultraviolet (UV) radiation exposure are the chief drivers of dermatoporosis. In addition to UVA and UVB light, other secondary drivers include genetic susceptibility, topical and systematic corticosteroid use, and anticoagulant treatment.
Its pathogenesis is not well described in the literature but is easy to envision, Dr. Friedman said. For one, both advancing age and exposure to UV light lead to a reduction in hygroscopic glycosaminoglycans, including hyaluronate (HA), and the impact of this diminishment is believed to go “beyond [the loss of] buoyancy,” he noted. Researchers have “been showing these are not just water-loving molecules, they also have some biologic properties” relating to keratinocyte production and epidermal turnover that appear to be intricately linked to the pathogenesis of dermatoporosis.
HAs have been shown to interact with the cell surface receptor CD44 to stimulate keratinocyte proliferation, and low levels of CD44 have been reported in skin with dermatoporosis compared with a younger control population. (A newly characterized organelle, the hyaluronosome, serves as an HA factory and contains CD44 and heparin-binding epidermal growth factor, Dr. Friedman noted. Inadequate functioning may be involved in skin atrophy.)
Advancing age also brings an increase in matrix metalloproteinases (MMPs)–1, –2, and –3, which are “the demolition workers of the skin,” and downregulation of a tissue inhibitor of MMPs, he said.
Adding insult to injury, dermis-penetrating UVA also activates MMPs, “obliterating collagen and elastin.” UVB generates DNA photoproducts, including oxidative stress and damaging skin cell DNA. “That UV light induces breakdown [of the skin] through different mechanisms and inhibits buildup is a simple concept I think our patients can understand,” Dr. Friedman said.
Multifaceted Treatment
For an older adult, “there is never a wrong time to start sun-protective measures” to prevent or try to halt the progression of dermatoporosis, Dr. Friedman said, noting that “UV radiation is an immunosuppressant, so there are many good reasons to start” if the adult is not already taking measures on a regular basis.
Potential treatments for the syndrome of dermatoporosis are backed by few clinical studies, but dermatologists are skilled at translating the use of products from one disease state to another based on understandings of pathophysiology and mechanistic pathways, Dr. Friedman commented in an interview after the meeting.
For instance, “from decades of research, we know what retinoids will do to the skin,” he said in the interview. “We know they will turn on collagen-1 and -3 genes in the skin, and that they will increase the production of glycosaminoglycans ... By understanding the biology, we can translate this to dermatoporosis.” These changes were demonstrated, for instance, in a small study of topical retinol in older adults.
Studies of topical alpha hydroxy acid (AHA), moreover, have demonstrated epidermal thickening and firmness, and “some studies show they can limit steroid-induced atrophy,” Dr. Friedman said at the meeting. “And things like lactic acid and urea are super accessible.”
Topical dehydroepiandrosterone is backed by even less data than retinoids or AHAs are, “but it’s still something to consider” as part of a multimechanistic approach to dermatoporosis, Dr. Friedman shared, noting that a small study demonstrated beneficial effects on epidermal atrophy in aging skin.
The use of vitamin D analogues such as calcipotriene, which is approved for the treatment of psoriasis, may also be promising. “One concept is that [vitamin D analogues] increase calcium concentrations in the epidermis, and calcium is so central to keratinocyte differentiation” and epidermal function that calcipotriene in combination with topical steroid therapy has been shown to limit skin atrophy, he noted.
Nutritionally, low protein intake is a known problem in the older population and is associated with increased skin fragility and poorer healing. From a prevention and treatment standpoint, therefore, patients can be counseled to be attentive to their diets, Dr. Friedman said. Experts have recommended a higher protein intake for older adults than for younger adults; in 2013, an international group recommended a protein intake of 1-1.5 g/kg/d for healthy older adults and more for those with acute or chronic illness.
“Patients love talking about diet and skin disease ... and they love over-the-counter nutraceuticals as well because they want something natural,” Dr. Friedman said. “I like using bioflavonoids in combination with vitamin C, which can be effective especially for solar purpura.”
A 6-week randomized, placebo-controlled, double-blind trial involving 67 patients with purpura associated with aging found a 50% reduction in purpura lesions among those took a particular citrus bioflavonoid blend twice daily. “I thought this was a pretty well-done study,” he said, noting that patient self-assessment and investigator global assessment were utilized.
Skin Injury and Wound Prevention
In addition to recommending gentle skin cleansers and daily moisturizing, dermatologists should talk to their older patients with dermatoporosis about their home environments. “What is it like? Is there furniture with sharp edges?” Dr. Friedman advised. If so, could they use sleeves or protectors on their arms or legs “to protect against injury?”
In a later meeting session about lower-extremity wounds on geriatric patients, Michael Stempel, DPM, assistant professor of medicine and surgery and chief of podiatry at GWU, said that he was happy to hear the term dermatoporosis being used because like diabetes, it’s a risk factor for developing lower-extremity wounds and poor wound healing.
He shared the case of an older woman with dermatoporosis who “tripped and skinned her knee against a step and then self-treated it for over a month by pouring hydrogen peroxide over it and letting air get to it.” The wound developed into “full-thickness tissue loss,” said Dr. Stempel, also medical director of the Wound Healing and Limb Preservation Center at GWU Hospital.
Misperceptions are common among older patients about how a simple wound should be managed; for instance, the adage “just let it get air” is not uncommon. This makes anticipatory guidance about basic wound care — such as the importance of a moist and occlusive environment and the safe use of hydrogen peroxide — especially important for patients with dermatoporosis, Dr. Friedman commented after the meeting.
Dermatoporosis is quantifiable, Dr. Friedman said during the meeting, with a scoring system having been developed by the researchers in Switzerland who originally coined the term. Its use in practice is unnecessary, but its existence is “nice to share with patients who feel bothered because oftentimes, patients feel it’s been dismissed by other providers,” he said. “Telling your patients there’s an actual name for their problem, and that there are ways to quantify and measure changes over time, is validating.”
Its recognition as a medical condition, Dr. Friedman added, also enables the dermatologist to bring it up and counsel appropriately — without a patient feeling shame — when it is identified in the context of a skin excision, treatment of a primary inflammatory skin disease, or management of another dermatologic problem.
Dr. Friedman disclosed that he is a consultant/advisory board member for L’Oréal, La Roche-Posay, Galderma, and other companies; a speaker for Regeneron/Sanofi, Incyte, BMD, and Janssen; and has grants from Pfizer, Lilly, Incyte, and other companies. Dr. Stempel reported no disclosures.
A version of this article first appeared on Medscape.com.
WASHINGTON — and conveys the skin’s vulnerability to serious medical complications, said Adam Friedman, MD, at the ElderDerm conference on dermatology in the older patient.
Key features of dermatoporosis include atrophic skin, solar purpura, white pseudoscars, easily acquired skin lacerations and tears, bruises, and delayed healing. “We’re going to see more of this, and it will more and more be a chief complaint of patients,” said Dr. Friedman, professor and chair of dermatology at George Washington University (GWU) in Washington, and co-chair of the meeting. GWU hosted the conference, describing it as a first-of-its-kind meeting dedicated to improving dermatologic care for older adults.
Dermatoporosis was described in the literature in 2007 by dermatologists at the University of Geneva in Switzerland. “It is not only a cosmetic problem,” Dr. Friedman said. “This is a medical problem ... which can absolutely lead to comorbidities [such as deep dissecting hematomas] that are a huge strain on the healthcare system.”
Dermatologists can meet the moment with holistic, creative combination treatment and counseling approaches aimed at improving the mechanical strength of skin and preventing potential complications in older patients, Dr. Friedman said at the meeting.
He described the case of a 76-year-old woman who presented with dermatoporosis on her arms involving pronounced skin atrophy, solar purpura, and a small covered laceration. “This was a patient who was both devastated by the appearance” and impacted by the pain and burden of dressing frequent wounds, said Dr. Friedman, who is also the director of the Residency Program, of Translational Research, and of Supportive Oncodermatology, all within the Department of Dermatology at GWU.
With 11 months of topical treatment that included daily application of calcipotriene 0.05% ointment and nightly application of tazarotene 0.045% lotion and oral supplementation with 1000-mg vitamin C twice daily and 1000-mg citrus bioflavonoid complex daily, as well as no changes to the medications she took for various comorbidities, the solar purpura improved significantly and “we made a huge difference in the integrity of her skin,” he said.
Dr. Friedman also described this case in a recently published article in the Journal of Drugs in Dermatology titled “What’s Old Is New: An Emerging Focus on Dermatoporosis”.
Likely Pathophysiology
Advancing age and chronic ultraviolet (UV) radiation exposure are the chief drivers of dermatoporosis. In addition to UVA and UVB light, other secondary drivers include genetic susceptibility, topical and systematic corticosteroid use, and anticoagulant treatment.
Its pathogenesis is not well described in the literature but is easy to envision, Dr. Friedman said. For one, both advancing age and exposure to UV light lead to a reduction in hygroscopic glycosaminoglycans, including hyaluronate (HA), and the impact of this diminishment is believed to go “beyond [the loss of] buoyancy,” he noted. Researchers have “been showing these are not just water-loving molecules, they also have some biologic properties” relating to keratinocyte production and epidermal turnover that appear to be intricately linked to the pathogenesis of dermatoporosis.
HAs have been shown to interact with the cell surface receptor CD44 to stimulate keratinocyte proliferation, and low levels of CD44 have been reported in skin with dermatoporosis compared with a younger control population. (A newly characterized organelle, the hyaluronosome, serves as an HA factory and contains CD44 and heparin-binding epidermal growth factor, Dr. Friedman noted. Inadequate functioning may be involved in skin atrophy.)
Advancing age also brings an increase in matrix metalloproteinases (MMPs)–1, –2, and –3, which are “the demolition workers of the skin,” and downregulation of a tissue inhibitor of MMPs, he said.
Adding insult to injury, dermis-penetrating UVA also activates MMPs, “obliterating collagen and elastin.” UVB generates DNA photoproducts, including oxidative stress and damaging skin cell DNA. “That UV light induces breakdown [of the skin] through different mechanisms and inhibits buildup is a simple concept I think our patients can understand,” Dr. Friedman said.
Multifaceted Treatment
For an older adult, “there is never a wrong time to start sun-protective measures” to prevent or try to halt the progression of dermatoporosis, Dr. Friedman said, noting that “UV radiation is an immunosuppressant, so there are many good reasons to start” if the adult is not already taking measures on a regular basis.
Potential treatments for the syndrome of dermatoporosis are backed by few clinical studies, but dermatologists are skilled at translating the use of products from one disease state to another based on understandings of pathophysiology and mechanistic pathways, Dr. Friedman commented in an interview after the meeting.
For instance, “from decades of research, we know what retinoids will do to the skin,” he said in the interview. “We know they will turn on collagen-1 and -3 genes in the skin, and that they will increase the production of glycosaminoglycans ... By understanding the biology, we can translate this to dermatoporosis.” These changes were demonstrated, for instance, in a small study of topical retinol in older adults.
Studies of topical alpha hydroxy acid (AHA), moreover, have demonstrated epidermal thickening and firmness, and “some studies show they can limit steroid-induced atrophy,” Dr. Friedman said at the meeting. “And things like lactic acid and urea are super accessible.”
Topical dehydroepiandrosterone is backed by even less data than retinoids or AHAs are, “but it’s still something to consider” as part of a multimechanistic approach to dermatoporosis, Dr. Friedman shared, noting that a small study demonstrated beneficial effects on epidermal atrophy in aging skin.
The use of vitamin D analogues such as calcipotriene, which is approved for the treatment of psoriasis, may also be promising. “One concept is that [vitamin D analogues] increase calcium concentrations in the epidermis, and calcium is so central to keratinocyte differentiation” and epidermal function that calcipotriene in combination with topical steroid therapy has been shown to limit skin atrophy, he noted.
Nutritionally, low protein intake is a known problem in the older population and is associated with increased skin fragility and poorer healing. From a prevention and treatment standpoint, therefore, patients can be counseled to be attentive to their diets, Dr. Friedman said. Experts have recommended a higher protein intake for older adults than for younger adults; in 2013, an international group recommended a protein intake of 1-1.5 g/kg/d for healthy older adults and more for those with acute or chronic illness.
“Patients love talking about diet and skin disease ... and they love over-the-counter nutraceuticals as well because they want something natural,” Dr. Friedman said. “I like using bioflavonoids in combination with vitamin C, which can be effective especially for solar purpura.”
A 6-week randomized, placebo-controlled, double-blind trial involving 67 patients with purpura associated with aging found a 50% reduction in purpura lesions among those took a particular citrus bioflavonoid blend twice daily. “I thought this was a pretty well-done study,” he said, noting that patient self-assessment and investigator global assessment were utilized.
Skin Injury and Wound Prevention
In addition to recommending gentle skin cleansers and daily moisturizing, dermatologists should talk to their older patients with dermatoporosis about their home environments. “What is it like? Is there furniture with sharp edges?” Dr. Friedman advised. If so, could they use sleeves or protectors on their arms or legs “to protect against injury?”
In a later meeting session about lower-extremity wounds on geriatric patients, Michael Stempel, DPM, assistant professor of medicine and surgery and chief of podiatry at GWU, said that he was happy to hear the term dermatoporosis being used because like diabetes, it’s a risk factor for developing lower-extremity wounds and poor wound healing.
He shared the case of an older woman with dermatoporosis who “tripped and skinned her knee against a step and then self-treated it for over a month by pouring hydrogen peroxide over it and letting air get to it.” The wound developed into “full-thickness tissue loss,” said Dr. Stempel, also medical director of the Wound Healing and Limb Preservation Center at GWU Hospital.
Misperceptions are common among older patients about how a simple wound should be managed; for instance, the adage “just let it get air” is not uncommon. This makes anticipatory guidance about basic wound care — such as the importance of a moist and occlusive environment and the safe use of hydrogen peroxide — especially important for patients with dermatoporosis, Dr. Friedman commented after the meeting.
Dermatoporosis is quantifiable, Dr. Friedman said during the meeting, with a scoring system having been developed by the researchers in Switzerland who originally coined the term. Its use in practice is unnecessary, but its existence is “nice to share with patients who feel bothered because oftentimes, patients feel it’s been dismissed by other providers,” he said. “Telling your patients there’s an actual name for their problem, and that there are ways to quantify and measure changes over time, is validating.”
Its recognition as a medical condition, Dr. Friedman added, also enables the dermatologist to bring it up and counsel appropriately — without a patient feeling shame — when it is identified in the context of a skin excision, treatment of a primary inflammatory skin disease, or management of another dermatologic problem.
Dr. Friedman disclosed that he is a consultant/advisory board member for L’Oréal, La Roche-Posay, Galderma, and other companies; a speaker for Regeneron/Sanofi, Incyte, BMD, and Janssen; and has grants from Pfizer, Lilly, Incyte, and other companies. Dr. Stempel reported no disclosures.
A version of this article first appeared on Medscape.com.
FROM ELDERDERM 2024
Managing Atopic Dermatitis in Older Adults: A Common, Unique Challenge
WASHINGTON, DC — Jonathan I. Silverberg, MD, PhD, MPH, said at the ElderDerm Conference on dermatology in the older patient hosted by the George Washington University School of Medicine and Health Sciences, Washington, DC.
“I walked out of residency under the impression that if it didn’t start in the first year or two of life, it’s not AD,” said Dr. Silverberg, professor of dermatology and director of clinical research at George Washington University. “The numbers tell us a very different story.”
The prevalence of AD in the United States fluctuates between 6% and 8% through adulthood, including age categories up to 81-85 years, according to 2012 National Health Interview Survey data. And while persistence of childhood-onset AD is common, a systematic review and meta-analysis published in 2018 concluded that one in four adults with AD report adult-onset disease.
The investigators, including Dr. Silverberg, identified 25 observational studies — studies conducted across 16 countries and published during 1956-2017 — that included an analysis of age of onset beyond 10 years of age, and other inclusion criteria. Of the 25 studies, 17 reported age of onset after 16 years of age and had sufficient data for the meta-analysis. Using random-effects weighting, the investigators found a pooled proportion of adult-onset AD of 26.1% (95% CI, 16.5%-37.2%).
The research demonstrates that “the age of onset is distributed well throughout the lifespan,” Dr. Silverberg said, with the data “indicating there are many elderly-onset cases of true AD as well.” (Thirteen of the studies analyzed an age of onset from age ≥ 65, and several looked beyond age 80).
A 2021 study of a primary care database in the United Kingdom of 3.85 million children and adults found a “fascinating” bimodal distribution of incidence across the lifespan, with peaks in both infancy and older adulthood, he said. Incidence in adulthood was relatively stable from ages 18-49 years, after which, “into the 50s, 60s and beyond, you started to see a steady climb again.”
Also intriguing, Dr. Silverberg continued, are findings from a study of outpatient healthcare utilization for AD in which he and his coinvestigator analyzed data from the National Ambulatory Medical Care Survey (NAMCS). In the article, published in 2023 covering data from the 1993-2015 NAMCS, they reported that AD visits were more common among children aged 0-4 years (32.0%) and 5-9 years of age (10.6%), then decreased in adolescents aged 10-19 years (11.6%), remained fairly steady in patients aged 20-89 years (1.0%-4.7%), and increased in patients aged > 90 years (20.7%).
“The peak usage for dermatologists, primary care physicians, etc., is happening in the first few years of life, partially because that’s when the disease is more common and more severe but also partially because that’s when parents and caregivers are first learning [about] the disease and trying to understand how to gain control,” Dr. Silverberg said at the meeting, presenting data from an expanded, unpublished analysis of NAMCS data showing these same outpatient utilization patterns.
“It’s fascinating — there’s a much greater utilization in the elderly population. Why? The short answer is, we don’t know,” he said.
Risk Factors, Immune Differences
People with adult-onset AD were more likely to be women, smokers in adulthood, and have a lower childhood socioeconomic status than those whose AD started in childhood in a longitudinal study of two large birth cohorts from the United Kingdom , Dr. Silverberg pointed out.
Patients with childhood-onset AD, meanwhile, were more likely to have asthma, allergen-specific immunoglobulin E (IgE), and known genetic polymorphisms previously associated with AD. (Each cohort — the 1958 British Cohort Study and the 1970 British Cohort Study — had more than 17,000 participants who were followed from birth through middle age.)
Data is limited, but “mechanistically,” AD in much older adults appears to have a unique serum cytokine pattern, Dr. Silverberg said. He pointed to a cross-sectional study in China of 1312 children and adults with AD in which researchers analyzed clinical features, serum samples, and skin biopsy samples.
Adults aged > 60 years showed more lesions on the trunk and extensor sites of the extremities and lower levels of serum IgE and peripheral eosinophil counts than those in younger age groups. And “interestingly,” compared with healthy controls, older patients with AD had “higher levels of serum expression of a variety of cytokines, including IL [interleukin]-4 but also high TARC levels ... and a variety of cytokines related to the Th17, TH1 axes, etc.,” he said.
“So, we’re seeing a fascinating new profile that may be a little different than younger-onset cases,” he said, noting that TARC (thymus and activation-regulated chemokine) is regarded as a “decent biomarker” for AD.
In addition to higher levels of IL-4 and TARC, the study investigators reported significantly higher levels of IL-17A, IL-6, IL-22, IL-33, and thymic stromal lymphopoietin in older patients, compared with healthy controls.
Research also suggests that air pollution may play a role in the onset of AD in older age, Dr. Silverberg said, referencing a 2023 study that explored the association of air pollution and genetic risk with the onset of AD after age 50. The study analyzed 337,910 participants from the UK Biobank, with a median 12-year follow-up. Genetic risks were assessed as low, intermediate, and high, based on tertiles of polygenic risk scores. Exposure to various air pollutants was assessed using weighted quantile sum and also categorized into tertiles.
The incidence of older adult-onset AD was associated with medium and high air pollution compared with low air pollution, with hazard ratios (HRs) of 1.182 (P = .003) and 1.359 (P < .001), respectively. And “to a lesser extent,” Dr. Silverberg said, incidence was associated with medium and high genetic susceptibility, with HRs of 1.065 (P = .249) and 1.153 (P = .008).
The researchers calculated a greater population-attributable fraction of air pollution (15.5%) than genetic risk (6.4%). “This means that yes, genetics can contribute even to later-onset disease ... but environment may play an even more important role,” Dr. Silverberg said.
In the Clinic
In all patients, and especially in older adults, sleep disturbance associated with AD is a consideration for care. Data collected at the eczema clinic of Northwestern University, Chicago, Illinois, between 2014 and 2019 through previsit, self-administered questionnaires show that patients ≥ 65 years of age have more profound sleep disturbance (especially trouble staying asleep) than patients aged 18-64 years, despite having similar AD severity, said Dr. Silverberg, a coinvestigator of the study.
Older age was associated with having an increased number of nights of sleep disturbance (3-7 nights in the previous week) because of eczema (adjusted odds ratio [aOR], 2.14; 95% CI, 1.16-3.92). It was also associated with itching-attributed delays in falling asleep and nighttime awakenings in the prior 2 weeks (aOR, 1.88; 95% CI, 1.05-3.39).
“The aging population has dysregulated sleep patterns and altered circadian rhythms, so some of this is just natural predisposition,” Dr. Silverberg said. “But it’s amplified [with AD and itching], and it becomes a big clinical problem when we get into treatment because it’s our natural inclination to prescribe antihistamines for their sedative properties.”
Antihistamines can cause more profound sedation, more forgetfulness, and more anticholinergic side effects, he said, noting that “there’s some evidence that high-dose antihistamines may exacerbate dementia.”
Medication side effects and medication interactions, comorbidities, and decreased renal and hepatic clearance all can complicate treatment of AD in older adults. So can mobility, the extent of social/caregiving support, and other aspects of aging. For example, “I’m a big fan of ‘soak and smears’ ... but you have to ask, can you get out of a bathtub safely?” Dr. Silverberg said. “And you have to ask, can you reach the areas you need to [in order] to apply topicals?”
With oral Janus kinase inhibitors and other systemic medications, as with other drugs, “our older population is the most vulnerable from a safety perspective,” he said. A recently published post hoc analysis of four randomized trials of dupilumab in adults ≥ 60 years of age with moderate to severe AD demonstrated efficacy comparable with that in younger patients and “a really clean safety profile,” said Dr. Silverberg, the lead author. “We really need more of these types of post hocs to have some relative contextualization” for older adults.
Dr. Silverberg reported being a speaker for AbbVie, Eli Lilly, Leo Pharma, Pfizer, Regeneron, and Sanofi-Genzyme; a consultant and/or advisory board member for Regeneron, Sanofi-Genzyme, and other companies; and an investigator for several companies.
A version of this article first appeared on Medscape.com.
WASHINGTON, DC — Jonathan I. Silverberg, MD, PhD, MPH, said at the ElderDerm Conference on dermatology in the older patient hosted by the George Washington University School of Medicine and Health Sciences, Washington, DC.
“I walked out of residency under the impression that if it didn’t start in the first year or two of life, it’s not AD,” said Dr. Silverberg, professor of dermatology and director of clinical research at George Washington University. “The numbers tell us a very different story.”
The prevalence of AD in the United States fluctuates between 6% and 8% through adulthood, including age categories up to 81-85 years, according to 2012 National Health Interview Survey data. And while persistence of childhood-onset AD is common, a systematic review and meta-analysis published in 2018 concluded that one in four adults with AD report adult-onset disease.
The investigators, including Dr. Silverberg, identified 25 observational studies — studies conducted across 16 countries and published during 1956-2017 — that included an analysis of age of onset beyond 10 years of age, and other inclusion criteria. Of the 25 studies, 17 reported age of onset after 16 years of age and had sufficient data for the meta-analysis. Using random-effects weighting, the investigators found a pooled proportion of adult-onset AD of 26.1% (95% CI, 16.5%-37.2%).
The research demonstrates that “the age of onset is distributed well throughout the lifespan,” Dr. Silverberg said, with the data “indicating there are many elderly-onset cases of true AD as well.” (Thirteen of the studies analyzed an age of onset from age ≥ 65, and several looked beyond age 80).
A 2021 study of a primary care database in the United Kingdom of 3.85 million children and adults found a “fascinating” bimodal distribution of incidence across the lifespan, with peaks in both infancy and older adulthood, he said. Incidence in adulthood was relatively stable from ages 18-49 years, after which, “into the 50s, 60s and beyond, you started to see a steady climb again.”
Also intriguing, Dr. Silverberg continued, are findings from a study of outpatient healthcare utilization for AD in which he and his coinvestigator analyzed data from the National Ambulatory Medical Care Survey (NAMCS). In the article, published in 2023 covering data from the 1993-2015 NAMCS, they reported that AD visits were more common among children aged 0-4 years (32.0%) and 5-9 years of age (10.6%), then decreased in adolescents aged 10-19 years (11.6%), remained fairly steady in patients aged 20-89 years (1.0%-4.7%), and increased in patients aged > 90 years (20.7%).
“The peak usage for dermatologists, primary care physicians, etc., is happening in the first few years of life, partially because that’s when the disease is more common and more severe but also partially because that’s when parents and caregivers are first learning [about] the disease and trying to understand how to gain control,” Dr. Silverberg said at the meeting, presenting data from an expanded, unpublished analysis of NAMCS data showing these same outpatient utilization patterns.
“It’s fascinating — there’s a much greater utilization in the elderly population. Why? The short answer is, we don’t know,” he said.
Risk Factors, Immune Differences
People with adult-onset AD were more likely to be women, smokers in adulthood, and have a lower childhood socioeconomic status than those whose AD started in childhood in a longitudinal study of two large birth cohorts from the United Kingdom , Dr. Silverberg pointed out.
Patients with childhood-onset AD, meanwhile, were more likely to have asthma, allergen-specific immunoglobulin E (IgE), and known genetic polymorphisms previously associated with AD. (Each cohort — the 1958 British Cohort Study and the 1970 British Cohort Study — had more than 17,000 participants who were followed from birth through middle age.)
Data is limited, but “mechanistically,” AD in much older adults appears to have a unique serum cytokine pattern, Dr. Silverberg said. He pointed to a cross-sectional study in China of 1312 children and adults with AD in which researchers analyzed clinical features, serum samples, and skin biopsy samples.
Adults aged > 60 years showed more lesions on the trunk and extensor sites of the extremities and lower levels of serum IgE and peripheral eosinophil counts than those in younger age groups. And “interestingly,” compared with healthy controls, older patients with AD had “higher levels of serum expression of a variety of cytokines, including IL [interleukin]-4 but also high TARC levels ... and a variety of cytokines related to the Th17, TH1 axes, etc.,” he said.
“So, we’re seeing a fascinating new profile that may be a little different than younger-onset cases,” he said, noting that TARC (thymus and activation-regulated chemokine) is regarded as a “decent biomarker” for AD.
In addition to higher levels of IL-4 and TARC, the study investigators reported significantly higher levels of IL-17A, IL-6, IL-22, IL-33, and thymic stromal lymphopoietin in older patients, compared with healthy controls.
Research also suggests that air pollution may play a role in the onset of AD in older age, Dr. Silverberg said, referencing a 2023 study that explored the association of air pollution and genetic risk with the onset of AD after age 50. The study analyzed 337,910 participants from the UK Biobank, with a median 12-year follow-up. Genetic risks were assessed as low, intermediate, and high, based on tertiles of polygenic risk scores. Exposure to various air pollutants was assessed using weighted quantile sum and also categorized into tertiles.
The incidence of older adult-onset AD was associated with medium and high air pollution compared with low air pollution, with hazard ratios (HRs) of 1.182 (P = .003) and 1.359 (P < .001), respectively. And “to a lesser extent,” Dr. Silverberg said, incidence was associated with medium and high genetic susceptibility, with HRs of 1.065 (P = .249) and 1.153 (P = .008).
The researchers calculated a greater population-attributable fraction of air pollution (15.5%) than genetic risk (6.4%). “This means that yes, genetics can contribute even to later-onset disease ... but environment may play an even more important role,” Dr. Silverberg said.
In the Clinic
In all patients, and especially in older adults, sleep disturbance associated with AD is a consideration for care. Data collected at the eczema clinic of Northwestern University, Chicago, Illinois, between 2014 and 2019 through previsit, self-administered questionnaires show that patients ≥ 65 years of age have more profound sleep disturbance (especially trouble staying asleep) than patients aged 18-64 years, despite having similar AD severity, said Dr. Silverberg, a coinvestigator of the study.
Older age was associated with having an increased number of nights of sleep disturbance (3-7 nights in the previous week) because of eczema (adjusted odds ratio [aOR], 2.14; 95% CI, 1.16-3.92). It was also associated with itching-attributed delays in falling asleep and nighttime awakenings in the prior 2 weeks (aOR, 1.88; 95% CI, 1.05-3.39).
“The aging population has dysregulated sleep patterns and altered circadian rhythms, so some of this is just natural predisposition,” Dr. Silverberg said. “But it’s amplified [with AD and itching], and it becomes a big clinical problem when we get into treatment because it’s our natural inclination to prescribe antihistamines for their sedative properties.”
Antihistamines can cause more profound sedation, more forgetfulness, and more anticholinergic side effects, he said, noting that “there’s some evidence that high-dose antihistamines may exacerbate dementia.”
Medication side effects and medication interactions, comorbidities, and decreased renal and hepatic clearance all can complicate treatment of AD in older adults. So can mobility, the extent of social/caregiving support, and other aspects of aging. For example, “I’m a big fan of ‘soak and smears’ ... but you have to ask, can you get out of a bathtub safely?” Dr. Silverberg said. “And you have to ask, can you reach the areas you need to [in order] to apply topicals?”
With oral Janus kinase inhibitors and other systemic medications, as with other drugs, “our older population is the most vulnerable from a safety perspective,” he said. A recently published post hoc analysis of four randomized trials of dupilumab in adults ≥ 60 years of age with moderate to severe AD demonstrated efficacy comparable with that in younger patients and “a really clean safety profile,” said Dr. Silverberg, the lead author. “We really need more of these types of post hocs to have some relative contextualization” for older adults.
Dr. Silverberg reported being a speaker for AbbVie, Eli Lilly, Leo Pharma, Pfizer, Regeneron, and Sanofi-Genzyme; a consultant and/or advisory board member for Regeneron, Sanofi-Genzyme, and other companies; and an investigator for several companies.
A version of this article first appeared on Medscape.com.
WASHINGTON, DC — Jonathan I. Silverberg, MD, PhD, MPH, said at the ElderDerm Conference on dermatology in the older patient hosted by the George Washington University School of Medicine and Health Sciences, Washington, DC.
“I walked out of residency under the impression that if it didn’t start in the first year or two of life, it’s not AD,” said Dr. Silverberg, professor of dermatology and director of clinical research at George Washington University. “The numbers tell us a very different story.”
The prevalence of AD in the United States fluctuates between 6% and 8% through adulthood, including age categories up to 81-85 years, according to 2012 National Health Interview Survey data. And while persistence of childhood-onset AD is common, a systematic review and meta-analysis published in 2018 concluded that one in four adults with AD report adult-onset disease.
The investigators, including Dr. Silverberg, identified 25 observational studies — studies conducted across 16 countries and published during 1956-2017 — that included an analysis of age of onset beyond 10 years of age, and other inclusion criteria. Of the 25 studies, 17 reported age of onset after 16 years of age and had sufficient data for the meta-analysis. Using random-effects weighting, the investigators found a pooled proportion of adult-onset AD of 26.1% (95% CI, 16.5%-37.2%).
The research demonstrates that “the age of onset is distributed well throughout the lifespan,” Dr. Silverberg said, with the data “indicating there are many elderly-onset cases of true AD as well.” (Thirteen of the studies analyzed an age of onset from age ≥ 65, and several looked beyond age 80).
A 2021 study of a primary care database in the United Kingdom of 3.85 million children and adults found a “fascinating” bimodal distribution of incidence across the lifespan, with peaks in both infancy and older adulthood, he said. Incidence in adulthood was relatively stable from ages 18-49 years, after which, “into the 50s, 60s and beyond, you started to see a steady climb again.”
Also intriguing, Dr. Silverberg continued, are findings from a study of outpatient healthcare utilization for AD in which he and his coinvestigator analyzed data from the National Ambulatory Medical Care Survey (NAMCS). In the article, published in 2023 covering data from the 1993-2015 NAMCS, they reported that AD visits were more common among children aged 0-4 years (32.0%) and 5-9 years of age (10.6%), then decreased in adolescents aged 10-19 years (11.6%), remained fairly steady in patients aged 20-89 years (1.0%-4.7%), and increased in patients aged > 90 years (20.7%).
“The peak usage for dermatologists, primary care physicians, etc., is happening in the first few years of life, partially because that’s when the disease is more common and more severe but also partially because that’s when parents and caregivers are first learning [about] the disease and trying to understand how to gain control,” Dr. Silverberg said at the meeting, presenting data from an expanded, unpublished analysis of NAMCS data showing these same outpatient utilization patterns.
“It’s fascinating — there’s a much greater utilization in the elderly population. Why? The short answer is, we don’t know,” he said.
Risk Factors, Immune Differences
People with adult-onset AD were more likely to be women, smokers in adulthood, and have a lower childhood socioeconomic status than those whose AD started in childhood in a longitudinal study of two large birth cohorts from the United Kingdom , Dr. Silverberg pointed out.
Patients with childhood-onset AD, meanwhile, were more likely to have asthma, allergen-specific immunoglobulin E (IgE), and known genetic polymorphisms previously associated with AD. (Each cohort — the 1958 British Cohort Study and the 1970 British Cohort Study — had more than 17,000 participants who were followed from birth through middle age.)
Data is limited, but “mechanistically,” AD in much older adults appears to have a unique serum cytokine pattern, Dr. Silverberg said. He pointed to a cross-sectional study in China of 1312 children and adults with AD in which researchers analyzed clinical features, serum samples, and skin biopsy samples.
Adults aged > 60 years showed more lesions on the trunk and extensor sites of the extremities and lower levels of serum IgE and peripheral eosinophil counts than those in younger age groups. And “interestingly,” compared with healthy controls, older patients with AD had “higher levels of serum expression of a variety of cytokines, including IL [interleukin]-4 but also high TARC levels ... and a variety of cytokines related to the Th17, TH1 axes, etc.,” he said.
“So, we’re seeing a fascinating new profile that may be a little different than younger-onset cases,” he said, noting that TARC (thymus and activation-regulated chemokine) is regarded as a “decent biomarker” for AD.
In addition to higher levels of IL-4 and TARC, the study investigators reported significantly higher levels of IL-17A, IL-6, IL-22, IL-33, and thymic stromal lymphopoietin in older patients, compared with healthy controls.
Research also suggests that air pollution may play a role in the onset of AD in older age, Dr. Silverberg said, referencing a 2023 study that explored the association of air pollution and genetic risk with the onset of AD after age 50. The study analyzed 337,910 participants from the UK Biobank, with a median 12-year follow-up. Genetic risks were assessed as low, intermediate, and high, based on tertiles of polygenic risk scores. Exposure to various air pollutants was assessed using weighted quantile sum and also categorized into tertiles.
The incidence of older adult-onset AD was associated with medium and high air pollution compared with low air pollution, with hazard ratios (HRs) of 1.182 (P = .003) and 1.359 (P < .001), respectively. And “to a lesser extent,” Dr. Silverberg said, incidence was associated with medium and high genetic susceptibility, with HRs of 1.065 (P = .249) and 1.153 (P = .008).
The researchers calculated a greater population-attributable fraction of air pollution (15.5%) than genetic risk (6.4%). “This means that yes, genetics can contribute even to later-onset disease ... but environment may play an even more important role,” Dr. Silverberg said.
In the Clinic
In all patients, and especially in older adults, sleep disturbance associated with AD is a consideration for care. Data collected at the eczema clinic of Northwestern University, Chicago, Illinois, between 2014 and 2019 through previsit, self-administered questionnaires show that patients ≥ 65 years of age have more profound sleep disturbance (especially trouble staying asleep) than patients aged 18-64 years, despite having similar AD severity, said Dr. Silverberg, a coinvestigator of the study.
Older age was associated with having an increased number of nights of sleep disturbance (3-7 nights in the previous week) because of eczema (adjusted odds ratio [aOR], 2.14; 95% CI, 1.16-3.92). It was also associated with itching-attributed delays in falling asleep and nighttime awakenings in the prior 2 weeks (aOR, 1.88; 95% CI, 1.05-3.39).
“The aging population has dysregulated sleep patterns and altered circadian rhythms, so some of this is just natural predisposition,” Dr. Silverberg said. “But it’s amplified [with AD and itching], and it becomes a big clinical problem when we get into treatment because it’s our natural inclination to prescribe antihistamines for their sedative properties.”
Antihistamines can cause more profound sedation, more forgetfulness, and more anticholinergic side effects, he said, noting that “there’s some evidence that high-dose antihistamines may exacerbate dementia.”
Medication side effects and medication interactions, comorbidities, and decreased renal and hepatic clearance all can complicate treatment of AD in older adults. So can mobility, the extent of social/caregiving support, and other aspects of aging. For example, “I’m a big fan of ‘soak and smears’ ... but you have to ask, can you get out of a bathtub safely?” Dr. Silverberg said. “And you have to ask, can you reach the areas you need to [in order] to apply topicals?”
With oral Janus kinase inhibitors and other systemic medications, as with other drugs, “our older population is the most vulnerable from a safety perspective,” he said. A recently published post hoc analysis of four randomized trials of dupilumab in adults ≥ 60 years of age with moderate to severe AD demonstrated efficacy comparable with that in younger patients and “a really clean safety profile,” said Dr. Silverberg, the lead author. “We really need more of these types of post hocs to have some relative contextualization” for older adults.
Dr. Silverberg reported being a speaker for AbbVie, Eli Lilly, Leo Pharma, Pfizer, Regeneron, and Sanofi-Genzyme; a consultant and/or advisory board member for Regeneron, Sanofi-Genzyme, and other companies; and an investigator for several companies.
A version of this article first appeared on Medscape.com.
FROM ELDERDERM 2024
Studies Show Dupilumab Effects In Children with Both Atopic Dermatitis and Alopecia
TOPLINE:
(AA) in a review.
METHODOLOGY:
- Researchers conducted a scoping review of seven studies, a result of a MEDLINE and Embase search on March 1, 2024, which included 31 patients aged 4-17 years with both AD and AA (average age, 11.4 years; 64.5% women).
- The review included four case reports, two case series, and one retrospective chart review.
- Patients had an average duration of AA and AD of 3.31 years and 5.33 years, respectively, before starting dupilumab.
- The type of AA was listed in 22 patients; among these patients, alopecia universalis was the most common (50%), followed by alopecia ophiasis (22.7%), patchy alopecia (18.2%), and alopecia totalis (9.09%).
TAKEAWAY:
- Overall, 77.4% of patients in the trials achieved hair regrowth with dupilumab treatment with a mean 42.6 reduction in SALT score (measuring scalp hair loss on a scale of 0-100) over an average of 3.21 months (P < .01).
- Severity of AD was reduced by an average of 2.14 units to an average of 0.857 (clear or almost clear AD; P < .01) on the AD Investigator Global Assessment dropping from an average of 3 (severe disease) before treatment.
- There were no characteristics that significantly distinguished patients with AA who responded to treatment from those who did not.
- Four patients reported worsening of preexisting AA after starting dupilumab; two of these continued dupilumab and showed improvement at subsequent follow-ups.
IN PRACTICE:
“Our review highlights the efficacy of dupilumab in pediatric AA with concurrent AD,” wrote the authors, noting that “the exact mechanism for this efficacy remains speculative.” Although there have been reports of new or worsening AA with dupilumab, they added, its “favorable safety profile in pediatrics enhances its appeal for AA treatment, as monotherapy or in combination with other AA medications.”
SOURCE:
The study was led by Dea Metko, Michael G. DeGroote School of Medicine in Hamilton, Ontario, Canada. It was published online on July 4, 2024, in Pediatric Dermatology.
LIMITATIONS:
Potential publication bias, inconsistent data reporting, the small number of patients, and short follow-up duration were the main limitations of this study.
DISCLOSURES:
The study funding source was not disclosed. One author received honoraria outside this work. Other authors declared no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
(AA) in a review.
METHODOLOGY:
- Researchers conducted a scoping review of seven studies, a result of a MEDLINE and Embase search on March 1, 2024, which included 31 patients aged 4-17 years with both AD and AA (average age, 11.4 years; 64.5% women).
- The review included four case reports, two case series, and one retrospective chart review.
- Patients had an average duration of AA and AD of 3.31 years and 5.33 years, respectively, before starting dupilumab.
- The type of AA was listed in 22 patients; among these patients, alopecia universalis was the most common (50%), followed by alopecia ophiasis (22.7%), patchy alopecia (18.2%), and alopecia totalis (9.09%).
TAKEAWAY:
- Overall, 77.4% of patients in the trials achieved hair regrowth with dupilumab treatment with a mean 42.6 reduction in SALT score (measuring scalp hair loss on a scale of 0-100) over an average of 3.21 months (P < .01).
- Severity of AD was reduced by an average of 2.14 units to an average of 0.857 (clear or almost clear AD; P < .01) on the AD Investigator Global Assessment dropping from an average of 3 (severe disease) before treatment.
- There were no characteristics that significantly distinguished patients with AA who responded to treatment from those who did not.
- Four patients reported worsening of preexisting AA after starting dupilumab; two of these continued dupilumab and showed improvement at subsequent follow-ups.
IN PRACTICE:
“Our review highlights the efficacy of dupilumab in pediatric AA with concurrent AD,” wrote the authors, noting that “the exact mechanism for this efficacy remains speculative.” Although there have been reports of new or worsening AA with dupilumab, they added, its “favorable safety profile in pediatrics enhances its appeal for AA treatment, as monotherapy or in combination with other AA medications.”
SOURCE:
The study was led by Dea Metko, Michael G. DeGroote School of Medicine in Hamilton, Ontario, Canada. It was published online on July 4, 2024, in Pediatric Dermatology.
LIMITATIONS:
Potential publication bias, inconsistent data reporting, the small number of patients, and short follow-up duration were the main limitations of this study.
DISCLOSURES:
The study funding source was not disclosed. One author received honoraria outside this work. Other authors declared no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
(AA) in a review.
METHODOLOGY:
- Researchers conducted a scoping review of seven studies, a result of a MEDLINE and Embase search on March 1, 2024, which included 31 patients aged 4-17 years with both AD and AA (average age, 11.4 years; 64.5% women).
- The review included four case reports, two case series, and one retrospective chart review.
- Patients had an average duration of AA and AD of 3.31 years and 5.33 years, respectively, before starting dupilumab.
- The type of AA was listed in 22 patients; among these patients, alopecia universalis was the most common (50%), followed by alopecia ophiasis (22.7%), patchy alopecia (18.2%), and alopecia totalis (9.09%).
TAKEAWAY:
- Overall, 77.4% of patients in the trials achieved hair regrowth with dupilumab treatment with a mean 42.6 reduction in SALT score (measuring scalp hair loss on a scale of 0-100) over an average of 3.21 months (P < .01).
- Severity of AD was reduced by an average of 2.14 units to an average of 0.857 (clear or almost clear AD; P < .01) on the AD Investigator Global Assessment dropping from an average of 3 (severe disease) before treatment.
- There were no characteristics that significantly distinguished patients with AA who responded to treatment from those who did not.
- Four patients reported worsening of preexisting AA after starting dupilumab; two of these continued dupilumab and showed improvement at subsequent follow-ups.
IN PRACTICE:
“Our review highlights the efficacy of dupilumab in pediatric AA with concurrent AD,” wrote the authors, noting that “the exact mechanism for this efficacy remains speculative.” Although there have been reports of new or worsening AA with dupilumab, they added, its “favorable safety profile in pediatrics enhances its appeal for AA treatment, as monotherapy or in combination with other AA medications.”
SOURCE:
The study was led by Dea Metko, Michael G. DeGroote School of Medicine in Hamilton, Ontario, Canada. It was published online on July 4, 2024, in Pediatric Dermatology.
LIMITATIONS:
Potential publication bias, inconsistent data reporting, the small number of patients, and short follow-up duration were the main limitations of this study.
DISCLOSURES:
The study funding source was not disclosed. One author received honoraria outside this work. Other authors declared no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
Topical PDE4 Inhibitor Now Approved for Atopic Dermatitis in Children, Adults
On July 9, the aged 6 years or older.
Roflumilast cream 0.15%, which has been developed by Arcutis Biotherapeutics and is marketed under the brand name Zoryve, is a steroid-free topical phosphodiesterase-4 inhibitor that was previously approved in a higher concentration to treat seborrheic dermatitis and plaque psoriasis.
According to a press release from Arcutis, approval for AD was supported by positive results from three phase 3 studies, a phase 2 dose-ranging study, and two phase 1 pharmacokinetic trials. In two identical phase 3 studies known as INTEGUMENT-1 and INTEGUMENT-2, about 40% of children and adults treated with roflumilast cream 0.15% achieved a Validated Investigator Global Assessment for Atopic Dermatitis score of clear (0) or almost clear (1) at week 4 (INTEGUMENT-1: 41.5% vs 25.2%; P < .0001; INTEGUMENT-2: 39% vs 16.9%; P < .0001), with significant improvement as early as week 1 (P < .0001).
Among children and adults who participated in the INTEGUMENT studies for 28 and 56 weeks, 61.3% and 65.7% achieved a 75% reduction in their Eczema Area and Severity Index scores, respectively. According to the company, there were no adverse reactions in the combined phase 3 pivotal trials that occurred in more than 2.9% of participants in either arm. The most common adverse reactions included headache (2.9%), nausea (1.9%), application-site pain (1.5%), diarrhea (1.5%), and vomiting (1.5%).
The product is expected to be available commercially at the end of July 2024, according to Arcutis. Roflumilast cream 0.3% is indicated for topical treatment of plaque psoriasis, including intertriginous areas, in adult and pediatric patients aged 6 years or older; roflumilast foam 0.3% is indicated for the treatment of seborrheic dermatitis in adult and pediatric patients aged 9 years or older.
A version of this article first appeared on Medscape.com.
On July 9, the aged 6 years or older.
Roflumilast cream 0.15%, which has been developed by Arcutis Biotherapeutics and is marketed under the brand name Zoryve, is a steroid-free topical phosphodiesterase-4 inhibitor that was previously approved in a higher concentration to treat seborrheic dermatitis and plaque psoriasis.
According to a press release from Arcutis, approval for AD was supported by positive results from three phase 3 studies, a phase 2 dose-ranging study, and two phase 1 pharmacokinetic trials. In two identical phase 3 studies known as INTEGUMENT-1 and INTEGUMENT-2, about 40% of children and adults treated with roflumilast cream 0.15% achieved a Validated Investigator Global Assessment for Atopic Dermatitis score of clear (0) or almost clear (1) at week 4 (INTEGUMENT-1: 41.5% vs 25.2%; P < .0001; INTEGUMENT-2: 39% vs 16.9%; P < .0001), with significant improvement as early as week 1 (P < .0001).
Among children and adults who participated in the INTEGUMENT studies for 28 and 56 weeks, 61.3% and 65.7% achieved a 75% reduction in their Eczema Area and Severity Index scores, respectively. According to the company, there were no adverse reactions in the combined phase 3 pivotal trials that occurred in more than 2.9% of participants in either arm. The most common adverse reactions included headache (2.9%), nausea (1.9%), application-site pain (1.5%), diarrhea (1.5%), and vomiting (1.5%).
The product is expected to be available commercially at the end of July 2024, according to Arcutis. Roflumilast cream 0.3% is indicated for topical treatment of plaque psoriasis, including intertriginous areas, in adult and pediatric patients aged 6 years or older; roflumilast foam 0.3% is indicated for the treatment of seborrheic dermatitis in adult and pediatric patients aged 9 years or older.
A version of this article first appeared on Medscape.com.
On July 9, the aged 6 years or older.
Roflumilast cream 0.15%, which has been developed by Arcutis Biotherapeutics and is marketed under the brand name Zoryve, is a steroid-free topical phosphodiesterase-4 inhibitor that was previously approved in a higher concentration to treat seborrheic dermatitis and plaque psoriasis.
According to a press release from Arcutis, approval for AD was supported by positive results from three phase 3 studies, a phase 2 dose-ranging study, and two phase 1 pharmacokinetic trials. In two identical phase 3 studies known as INTEGUMENT-1 and INTEGUMENT-2, about 40% of children and adults treated with roflumilast cream 0.15% achieved a Validated Investigator Global Assessment for Atopic Dermatitis score of clear (0) or almost clear (1) at week 4 (INTEGUMENT-1: 41.5% vs 25.2%; P < .0001; INTEGUMENT-2: 39% vs 16.9%; P < .0001), with significant improvement as early as week 1 (P < .0001).
Among children and adults who participated in the INTEGUMENT studies for 28 and 56 weeks, 61.3% and 65.7% achieved a 75% reduction in their Eczema Area and Severity Index scores, respectively. According to the company, there were no adverse reactions in the combined phase 3 pivotal trials that occurred in more than 2.9% of participants in either arm. The most common adverse reactions included headache (2.9%), nausea (1.9%), application-site pain (1.5%), diarrhea (1.5%), and vomiting (1.5%).
The product is expected to be available commercially at the end of July 2024, according to Arcutis. Roflumilast cream 0.3% is indicated for topical treatment of plaque psoriasis, including intertriginous areas, in adult and pediatric patients aged 6 years or older; roflumilast foam 0.3% is indicated for the treatment of seborrheic dermatitis in adult and pediatric patients aged 9 years or older.
A version of this article first appeared on Medscape.com.
Brazilian Peppertree: Watch Out for This Lesser-Known Relative of Poison Ivy
Brazilian peppertree (Schinus terebinthifolia), a member of the Anacardiaceae family, is an internationally invasive plant that causes allergic contact dermatitis (ACD) in susceptible individuals. This noxious weed has settled into the landscape of the southern United States and continues to expand. Its key identifying features include its year-round white flowers as well as a peppery and turpentinelike aroma created by cracking its bright red berries. The ACD associated with contact—primarily with the plant’s sap—stems from known alkenyl phenols, cardol and cardanol. Treatment of Brazilian peppertree–associated ACD parallels that for poison ivy. As this pest increases its range, dermatologists living in endemic areas should familiarize themselves with Brazilian peppertree and its potential for harm.
Brazilian Peppertree Morphology and Geography
Plants in the Anacardiaceae family contribute to more ACD than any other family, and its 80 genera include most of the urushiol-containing plants, such as Toxicodendron (poison ivy, poison oak, poison sumac, Japanese lacquer tree), Anacardium (cashew tree), Mangifera (mango fruit), Semecarpus (India marking nut tree), and Schinus (Brazilian peppertree). Deciduous and evergreen tree members of the Anacardiaceae family grow primarily in tropical and subtropical locations and produce thick resins, 5-petalled flowers, and small fruit known as drupes. The genus name for Brazilian peppertree, Schinus, derives from Latin and Greek words meaning “mastic tree,” a relative of the pistachio tree that the Brazilian peppertree resembles.1 Brazilian peppertree leaves look and smell similar to Pistacia terebinthus (turpentine tree or terebinth), from which the species name terebinthifolia derives.2
Brazilian peppertree originated in South America, particularly Brazil, Paraguay, and Argentina.3 Since the 1840s,4 it has been an invasive weed in the United States, notably in Florida, California, Hawaii, Alabama, Georgia,5 Arizona,6 Nevada,3 and Texas.5,7 The plant also grows throughout the world, including parts of Africa, Asia, Central America, Europe,6 New Zealand,8 Australia, and various islands.9 The plant expertly outcompetes neighboring plants and has prompted control and eradication efforts in many locations.3
Identifying Features and Allergenic Plant Parts
Brazilian peppertree can be either a shrub or tree up to 30 feet tall.4 As an evergreen, it retains its leaves year-round. During fruiting seasons (primarily December through March7), bright red or pink (depending on the variety3) berries appear (Figure 1A) and contribute to its nickname “Florida holly.” Although generally considered an unwelcome guest in Florida, it does display white flowers (Figure 1B) year-round, especially from September to November.9 It characteristically exhibits 3 to 13 leaflets per leaf.10 The leaflets’ ovoid and ridged edges, netlike vasculature, shiny hue, and aroma can help identify the plant (Figure 2A). For decades, the sap of the Brazilian peppertree has been associated with skin irritation (Figure 2B).6 Although the sap of the plant serves as the main culprit of Brazilian peppertree–associated ACD, it appears that other parts of the plant, including the fruit, can cause irritating effects to skin on contact.11,12 The leaves, trunk, and fruit can be harmful to both humans and animals.6 Chemicals from flowers and crushed fruit also can lead to irritating effects in the respiratory tract if aspirated.13


Urushiol, an oily resin present in most plants of the Anacardiaceae family,14 contains many chemicals, including allergenic phenols, catechols, and resorcinols.15 Urushiol-allergic individuals develop dermatitis upon exposure to Brazilian peppertree sap.6 Alkenyl phenols found in Brazilian peppertree lead to the cutaneous manifestations in sensitized patients.11,12 In 1983, Stahl et al11 identified a phenol, cardanol (chemical name 3-pentadecylphenol16) C15:1, in Brazilian peppertree fruit. The group further tested this compound’s effect on skin via patch testing, which showed an allergic response.11 Cashew nut shells (Anacardium occidentale) contain cardanol, anacardic acid (a phenolic acid), and cardol (a phenol with the chemical name 5-pentadecylresorcinol),15,16 though Stahl et al11 were unable to extract these 2 substances (if present) from Brazilian peppertree fruit. When exposed to cardol and anacardic acid, those allergic to poison ivy often develop ACD,15 and these 2 substances are more irritating than cardanol.11 A later study did identify cardol in addition to cardanol in Brazilian peppertree.12
Cutaneous Manifestations
Brazilian peppertree–induced ACD appears similar to other plant-induced ACD with linear streaks of erythema, juicy papules, vesicles, coalescing erythematous plaques, and/or occasional edema and bullae accompanied by intense pruritus.
Treatment
Avoiding contact with Brazilian peppertree is the first line of defense, and treatment for a reaction associated with exposure is similar to that of poison ivy.17 Application of cool compresses, calamine lotion, and topical astringents offer symptom alleviation, and topical steroids (eg, clobetasol propionate 0.05% twice daily) can improve mild localized ACD when given prior to formation of blisters. For more severe and diffuse ACD, oral steroids (eg, prednisone 1 mg/kg/d tapered over 2–3 weeks) likely are necessary, though intramuscular options greatly alleviate discomfort in more severe cases (eg, intramuscular triamcinolone acetonide 1 mg/kg combined with betamethasone 0.1 mg/kg). Physicians should monitor sites for any signs of superimposed bacterial infection and initiate antibiotics as necessary.17
- Zona S. The correct gender of Schinus (Anacardiaceae). Phytotaxa. 2015;222:075-077.
- Terebinth. Encyclopedia.com website. Updated May 17, 2018. Accessed July 9, 2024. https://www.encyclopedia.com/plants-and-animals/plants/plants/terebinth
- Brazilian pepper tree. iNaturalist website. Accessed July 1, 2024. https://www.inaturalist.org/guide_taxa/841531#:~:text=Throughout% 20South%20and%20Central%20America,and%20as%20a%20topical%20antiseptic
- Center for Aquatic and Invasive Plants. Schinus terebinthifolia. Brazilian peppertree. Accessed July 1, 2024. https://plants.ifas.ufl.edu/plant-directory/schinus-terebinthifolia/#:~:text=Species%20Overview&text=People%20sensitive%20to%20poison%20ivy,associated%20with%20its%20bloom%20period
- Brazilian peppertree (Schinus terebinthifolia). Early Detection & Distribution Mapping System. Accessed July 4, 2024. https://www.eddmaps.org/distribution/usstate.cfm?sub=78819
- Morton F. Brazilian pepper: its impact on people, animals, and the environment. Econ Bot. 1978;32:353-359.
- Fire Effects Information System. Schinus terebinthifolius. US Department of Agriculture website. Accessed July 4, 2024. https://www.fs.usda.gov/database/feis/plants/shrub/schter/all.html
- New Zealand Plant Conservation Network. Schinus terebinthifolius. Accessed July 1, 2024. https://www.nzpcn.org.nz/flora/species/schinus-terebinthifolius
- Rojas-Sandoval J, Acevedo-Rodriguez P. Schinus terebinthifolius (Brazilian pepper tree). CABI Compendium. July 23, 2014. Accessed July 1, 2024. https://www.cabidigitallibrary.org/doi/10.1079/cabicompendium.49031
- Patocka J, Diz de Almeida J. Brazilian peppertree: review of pharmacology. Mil Med Sci Lett. 2017;86:32-41.
- Stahl E, Keller K, Blinn C. Cardanol, a skin irritant in pink pepper. Plant Medica. 1983;48:5-9.
- Skopp G, Opferkuch H-J, Schqenker G. n-Alkylphenols from Schinus terebinthifolius Raddi (Anacardiaceae). In German. Zeitschrift für Naturforschung C. 1987;42:1-16. https://doi.org/10.1515/znc-1987-1-203.
- Lloyd HA, Jaouni TM, Evans SL, et al. Terpenes of Schinus terebinthifolius. Phytochemistry. 1977;16:1301-1302.
- Goon ATJ, Goh CL. Plant dermatitis: Asian perspective. Indian J Dermatol. 2011;56:707-710.
- Rozas-Muñoz E, Lepoittevin JP, Pujol RM, et al. Allergic contact dermatitis to plants: understanding the chemistry will help our diagnostic approach. Actas Dermosifiliogr. 2012;103:456-477.
- Caillol S. Cardanol: a promising building block for biobased polymers and additives. Curr Opin Green Sustain Chem. 2018;14: 26-32.
- Prok L, McGovern T. Poison ivy (Toxicodendron) dermatitis. UpToDate. Updated June 21, 2024. Accessed July 7, 2024. https://www.uptodate.com/contents/poison-ivy-toxicodendron-dermatitis#
Brazilian peppertree (Schinus terebinthifolia), a member of the Anacardiaceae family, is an internationally invasive plant that causes allergic contact dermatitis (ACD) in susceptible individuals. This noxious weed has settled into the landscape of the southern United States and continues to expand. Its key identifying features include its year-round white flowers as well as a peppery and turpentinelike aroma created by cracking its bright red berries. The ACD associated with contact—primarily with the plant’s sap—stems from known alkenyl phenols, cardol and cardanol. Treatment of Brazilian peppertree–associated ACD parallels that for poison ivy. As this pest increases its range, dermatologists living in endemic areas should familiarize themselves with Brazilian peppertree and its potential for harm.
Brazilian Peppertree Morphology and Geography
Plants in the Anacardiaceae family contribute to more ACD than any other family, and its 80 genera include most of the urushiol-containing plants, such as Toxicodendron (poison ivy, poison oak, poison sumac, Japanese lacquer tree), Anacardium (cashew tree), Mangifera (mango fruit), Semecarpus (India marking nut tree), and Schinus (Brazilian peppertree). Deciduous and evergreen tree members of the Anacardiaceae family grow primarily in tropical and subtropical locations and produce thick resins, 5-petalled flowers, and small fruit known as drupes. The genus name for Brazilian peppertree, Schinus, derives from Latin and Greek words meaning “mastic tree,” a relative of the pistachio tree that the Brazilian peppertree resembles.1 Brazilian peppertree leaves look and smell similar to Pistacia terebinthus (turpentine tree or terebinth), from which the species name terebinthifolia derives.2
Brazilian peppertree originated in South America, particularly Brazil, Paraguay, and Argentina.3 Since the 1840s,4 it has been an invasive weed in the United States, notably in Florida, California, Hawaii, Alabama, Georgia,5 Arizona,6 Nevada,3 and Texas.5,7 The plant also grows throughout the world, including parts of Africa, Asia, Central America, Europe,6 New Zealand,8 Australia, and various islands.9 The plant expertly outcompetes neighboring plants and has prompted control and eradication efforts in many locations.3
Identifying Features and Allergenic Plant Parts
Brazilian peppertree can be either a shrub or tree up to 30 feet tall.4 As an evergreen, it retains its leaves year-round. During fruiting seasons (primarily December through March7), bright red or pink (depending on the variety3) berries appear (Figure 1A) and contribute to its nickname “Florida holly.” Although generally considered an unwelcome guest in Florida, it does display white flowers (Figure 1B) year-round, especially from September to November.9 It characteristically exhibits 3 to 13 leaflets per leaf.10 The leaflets’ ovoid and ridged edges, netlike vasculature, shiny hue, and aroma can help identify the plant (Figure 2A). For decades, the sap of the Brazilian peppertree has been associated with skin irritation (Figure 2B).6 Although the sap of the plant serves as the main culprit of Brazilian peppertree–associated ACD, it appears that other parts of the plant, including the fruit, can cause irritating effects to skin on contact.11,12 The leaves, trunk, and fruit can be harmful to both humans and animals.6 Chemicals from flowers and crushed fruit also can lead to irritating effects in the respiratory tract if aspirated.13


Urushiol, an oily resin present in most plants of the Anacardiaceae family,14 contains many chemicals, including allergenic phenols, catechols, and resorcinols.15 Urushiol-allergic individuals develop dermatitis upon exposure to Brazilian peppertree sap.6 Alkenyl phenols found in Brazilian peppertree lead to the cutaneous manifestations in sensitized patients.11,12 In 1983, Stahl et al11 identified a phenol, cardanol (chemical name 3-pentadecylphenol16) C15:1, in Brazilian peppertree fruit. The group further tested this compound’s effect on skin via patch testing, which showed an allergic response.11 Cashew nut shells (Anacardium occidentale) contain cardanol, anacardic acid (a phenolic acid), and cardol (a phenol with the chemical name 5-pentadecylresorcinol),15,16 though Stahl et al11 were unable to extract these 2 substances (if present) from Brazilian peppertree fruit. When exposed to cardol and anacardic acid, those allergic to poison ivy often develop ACD,15 and these 2 substances are more irritating than cardanol.11 A later study did identify cardol in addition to cardanol in Brazilian peppertree.12
Cutaneous Manifestations
Brazilian peppertree–induced ACD appears similar to other plant-induced ACD with linear streaks of erythema, juicy papules, vesicles, coalescing erythematous plaques, and/or occasional edema and bullae accompanied by intense pruritus.
Treatment
Avoiding contact with Brazilian peppertree is the first line of defense, and treatment for a reaction associated with exposure is similar to that of poison ivy.17 Application of cool compresses, calamine lotion, and topical astringents offer symptom alleviation, and topical steroids (eg, clobetasol propionate 0.05% twice daily) can improve mild localized ACD when given prior to formation of blisters. For more severe and diffuse ACD, oral steroids (eg, prednisone 1 mg/kg/d tapered over 2–3 weeks) likely are necessary, though intramuscular options greatly alleviate discomfort in more severe cases (eg, intramuscular triamcinolone acetonide 1 mg/kg combined with betamethasone 0.1 mg/kg). Physicians should monitor sites for any signs of superimposed bacterial infection and initiate antibiotics as necessary.17
Brazilian peppertree (Schinus terebinthifolia), a member of the Anacardiaceae family, is an internationally invasive plant that causes allergic contact dermatitis (ACD) in susceptible individuals. This noxious weed has settled into the landscape of the southern United States and continues to expand. Its key identifying features include its year-round white flowers as well as a peppery and turpentinelike aroma created by cracking its bright red berries. The ACD associated with contact—primarily with the plant’s sap—stems from known alkenyl phenols, cardol and cardanol. Treatment of Brazilian peppertree–associated ACD parallels that for poison ivy. As this pest increases its range, dermatologists living in endemic areas should familiarize themselves with Brazilian peppertree and its potential for harm.
Brazilian Peppertree Morphology and Geography
Plants in the Anacardiaceae family contribute to more ACD than any other family, and its 80 genera include most of the urushiol-containing plants, such as Toxicodendron (poison ivy, poison oak, poison sumac, Japanese lacquer tree), Anacardium (cashew tree), Mangifera (mango fruit), Semecarpus (India marking nut tree), and Schinus (Brazilian peppertree). Deciduous and evergreen tree members of the Anacardiaceae family grow primarily in tropical and subtropical locations and produce thick resins, 5-petalled flowers, and small fruit known as drupes. The genus name for Brazilian peppertree, Schinus, derives from Latin and Greek words meaning “mastic tree,” a relative of the pistachio tree that the Brazilian peppertree resembles.1 Brazilian peppertree leaves look and smell similar to Pistacia terebinthus (turpentine tree or terebinth), from which the species name terebinthifolia derives.2
Brazilian peppertree originated in South America, particularly Brazil, Paraguay, and Argentina.3 Since the 1840s,4 it has been an invasive weed in the United States, notably in Florida, California, Hawaii, Alabama, Georgia,5 Arizona,6 Nevada,3 and Texas.5,7 The plant also grows throughout the world, including parts of Africa, Asia, Central America, Europe,6 New Zealand,8 Australia, and various islands.9 The plant expertly outcompetes neighboring plants and has prompted control and eradication efforts in many locations.3
Identifying Features and Allergenic Plant Parts
Brazilian peppertree can be either a shrub or tree up to 30 feet tall.4 As an evergreen, it retains its leaves year-round. During fruiting seasons (primarily December through March7), bright red or pink (depending on the variety3) berries appear (Figure 1A) and contribute to its nickname “Florida holly.” Although generally considered an unwelcome guest in Florida, it does display white flowers (Figure 1B) year-round, especially from September to November.9 It characteristically exhibits 3 to 13 leaflets per leaf.10 The leaflets’ ovoid and ridged edges, netlike vasculature, shiny hue, and aroma can help identify the plant (Figure 2A). For decades, the sap of the Brazilian peppertree has been associated with skin irritation (Figure 2B).6 Although the sap of the plant serves as the main culprit of Brazilian peppertree–associated ACD, it appears that other parts of the plant, including the fruit, can cause irritating effects to skin on contact.11,12 The leaves, trunk, and fruit can be harmful to both humans and animals.6 Chemicals from flowers and crushed fruit also can lead to irritating effects in the respiratory tract if aspirated.13


Urushiol, an oily resin present in most plants of the Anacardiaceae family,14 contains many chemicals, including allergenic phenols, catechols, and resorcinols.15 Urushiol-allergic individuals develop dermatitis upon exposure to Brazilian peppertree sap.6 Alkenyl phenols found in Brazilian peppertree lead to the cutaneous manifestations in sensitized patients.11,12 In 1983, Stahl et al11 identified a phenol, cardanol (chemical name 3-pentadecylphenol16) C15:1, in Brazilian peppertree fruit. The group further tested this compound’s effect on skin via patch testing, which showed an allergic response.11 Cashew nut shells (Anacardium occidentale) contain cardanol, anacardic acid (a phenolic acid), and cardol (a phenol with the chemical name 5-pentadecylresorcinol),15,16 though Stahl et al11 were unable to extract these 2 substances (if present) from Brazilian peppertree fruit. When exposed to cardol and anacardic acid, those allergic to poison ivy often develop ACD,15 and these 2 substances are more irritating than cardanol.11 A later study did identify cardol in addition to cardanol in Brazilian peppertree.12
Cutaneous Manifestations
Brazilian peppertree–induced ACD appears similar to other plant-induced ACD with linear streaks of erythema, juicy papules, vesicles, coalescing erythematous plaques, and/or occasional edema and bullae accompanied by intense pruritus.
Treatment
Avoiding contact with Brazilian peppertree is the first line of defense, and treatment for a reaction associated with exposure is similar to that of poison ivy.17 Application of cool compresses, calamine lotion, and topical astringents offer symptom alleviation, and topical steroids (eg, clobetasol propionate 0.05% twice daily) can improve mild localized ACD when given prior to formation of blisters. For more severe and diffuse ACD, oral steroids (eg, prednisone 1 mg/kg/d tapered over 2–3 weeks) likely are necessary, though intramuscular options greatly alleviate discomfort in more severe cases (eg, intramuscular triamcinolone acetonide 1 mg/kg combined with betamethasone 0.1 mg/kg). Physicians should monitor sites for any signs of superimposed bacterial infection and initiate antibiotics as necessary.17
- Zona S. The correct gender of Schinus (Anacardiaceae). Phytotaxa. 2015;222:075-077.
- Terebinth. Encyclopedia.com website. Updated May 17, 2018. Accessed July 9, 2024. https://www.encyclopedia.com/plants-and-animals/plants/plants/terebinth
- Brazilian pepper tree. iNaturalist website. Accessed July 1, 2024. https://www.inaturalist.org/guide_taxa/841531#:~:text=Throughout% 20South%20and%20Central%20America,and%20as%20a%20topical%20antiseptic
- Center for Aquatic and Invasive Plants. Schinus terebinthifolia. Brazilian peppertree. Accessed July 1, 2024. https://plants.ifas.ufl.edu/plant-directory/schinus-terebinthifolia/#:~:text=Species%20Overview&text=People%20sensitive%20to%20poison%20ivy,associated%20with%20its%20bloom%20period
- Brazilian peppertree (Schinus terebinthifolia). Early Detection & Distribution Mapping System. Accessed July 4, 2024. https://www.eddmaps.org/distribution/usstate.cfm?sub=78819
- Morton F. Brazilian pepper: its impact on people, animals, and the environment. Econ Bot. 1978;32:353-359.
- Fire Effects Information System. Schinus terebinthifolius. US Department of Agriculture website. Accessed July 4, 2024. https://www.fs.usda.gov/database/feis/plants/shrub/schter/all.html
- New Zealand Plant Conservation Network. Schinus terebinthifolius. Accessed July 1, 2024. https://www.nzpcn.org.nz/flora/species/schinus-terebinthifolius
- Rojas-Sandoval J, Acevedo-Rodriguez P. Schinus terebinthifolius (Brazilian pepper tree). CABI Compendium. July 23, 2014. Accessed July 1, 2024. https://www.cabidigitallibrary.org/doi/10.1079/cabicompendium.49031
- Patocka J, Diz de Almeida J. Brazilian peppertree: review of pharmacology. Mil Med Sci Lett. 2017;86:32-41.
- Stahl E, Keller K, Blinn C. Cardanol, a skin irritant in pink pepper. Plant Medica. 1983;48:5-9.
- Skopp G, Opferkuch H-J, Schqenker G. n-Alkylphenols from Schinus terebinthifolius Raddi (Anacardiaceae). In German. Zeitschrift für Naturforschung C. 1987;42:1-16. https://doi.org/10.1515/znc-1987-1-203.
- Lloyd HA, Jaouni TM, Evans SL, et al. Terpenes of Schinus terebinthifolius. Phytochemistry. 1977;16:1301-1302.
- Goon ATJ, Goh CL. Plant dermatitis: Asian perspective. Indian J Dermatol. 2011;56:707-710.
- Rozas-Muñoz E, Lepoittevin JP, Pujol RM, et al. Allergic contact dermatitis to plants: understanding the chemistry will help our diagnostic approach. Actas Dermosifiliogr. 2012;103:456-477.
- Caillol S. Cardanol: a promising building block for biobased polymers and additives. Curr Opin Green Sustain Chem. 2018;14: 26-32.
- Prok L, McGovern T. Poison ivy (Toxicodendron) dermatitis. UpToDate. Updated June 21, 2024. Accessed July 7, 2024. https://www.uptodate.com/contents/poison-ivy-toxicodendron-dermatitis#
- Zona S. The correct gender of Schinus (Anacardiaceae). Phytotaxa. 2015;222:075-077.
- Terebinth. Encyclopedia.com website. Updated May 17, 2018. Accessed July 9, 2024. https://www.encyclopedia.com/plants-and-animals/plants/plants/terebinth
- Brazilian pepper tree. iNaturalist website. Accessed July 1, 2024. https://www.inaturalist.org/guide_taxa/841531#:~:text=Throughout% 20South%20and%20Central%20America,and%20as%20a%20topical%20antiseptic
- Center for Aquatic and Invasive Plants. Schinus terebinthifolia. Brazilian peppertree. Accessed July 1, 2024. https://plants.ifas.ufl.edu/plant-directory/schinus-terebinthifolia/#:~:text=Species%20Overview&text=People%20sensitive%20to%20poison%20ivy,associated%20with%20its%20bloom%20period
- Brazilian peppertree (Schinus terebinthifolia). Early Detection & Distribution Mapping System. Accessed July 4, 2024. https://www.eddmaps.org/distribution/usstate.cfm?sub=78819
- Morton F. Brazilian pepper: its impact on people, animals, and the environment. Econ Bot. 1978;32:353-359.
- Fire Effects Information System. Schinus terebinthifolius. US Department of Agriculture website. Accessed July 4, 2024. https://www.fs.usda.gov/database/feis/plants/shrub/schter/all.html
- New Zealand Plant Conservation Network. Schinus terebinthifolius. Accessed July 1, 2024. https://www.nzpcn.org.nz/flora/species/schinus-terebinthifolius
- Rojas-Sandoval J, Acevedo-Rodriguez P. Schinus terebinthifolius (Brazilian pepper tree). CABI Compendium. July 23, 2014. Accessed July 1, 2024. https://www.cabidigitallibrary.org/doi/10.1079/cabicompendium.49031
- Patocka J, Diz de Almeida J. Brazilian peppertree: review of pharmacology. Mil Med Sci Lett. 2017;86:32-41.
- Stahl E, Keller K, Blinn C. Cardanol, a skin irritant in pink pepper. Plant Medica. 1983;48:5-9.
- Skopp G, Opferkuch H-J, Schqenker G. n-Alkylphenols from Schinus terebinthifolius Raddi (Anacardiaceae). In German. Zeitschrift für Naturforschung C. 1987;42:1-16. https://doi.org/10.1515/znc-1987-1-203.
- Lloyd HA, Jaouni TM, Evans SL, et al. Terpenes of Schinus terebinthifolius. Phytochemistry. 1977;16:1301-1302.
- Goon ATJ, Goh CL. Plant dermatitis: Asian perspective. Indian J Dermatol. 2011;56:707-710.
- Rozas-Muñoz E, Lepoittevin JP, Pujol RM, et al. Allergic contact dermatitis to plants: understanding the chemistry will help our diagnostic approach. Actas Dermosifiliogr. 2012;103:456-477.
- Caillol S. Cardanol: a promising building block for biobased polymers and additives. Curr Opin Green Sustain Chem. 2018;14: 26-32.
- Prok L, McGovern T. Poison ivy (Toxicodendron) dermatitis. UpToDate. Updated June 21, 2024. Accessed July 7, 2024. https://www.uptodate.com/contents/poison-ivy-toxicodendron-dermatitis#
Practice Points
- The Anacardiaceae family contains several plants, including Brazilian peppertree and poison ivy, that have the potential to cause allergic contact dermatitis (ACD).
- Hot spots for Brazilian peppertree include Florida and California, though it also has been reported in Texas, Hawaii, Georgia, Alabama, Arkansas, Nevada, and Arizona.
- Alkenyl phenols (eg, cardol, cardanol) are the key sensitizers found in Brazilian peppertree.
- Treatment consists of supportive care and either topical, oral, or intramuscular steroids depending on the extent and severity of the ACD.
Dupilumab Safe, Effective Over 5 Years in Moderate to Severe Atopic Dermatitis
TOPLINE:
Over 5 years, dupilumab demonstrated acceptable safety and sustained efficacy, with significant improvements in the signs and symptoms of AD, in the treatment of moderate to severe atopic dermatitis (AD).
METHODOLOGY:
- The phase 3 multinational LIBERTY AD open-label extension study evaluated the safety and efficacy of dupilumab in 2677 adults with moderate to severe AD who had previously participated in dupilumab trials over 5 years; 334 patients (12.5%) completed treatment up to 5 years.
- Patients started with subcutaneous dupilumab, initially dosed weekly after a loading dose, then every 2 weeks in 2019.
- The primary outcomes were the incidence and rate of treatment-emergent adverse events (TEAEs).
TAKEAWAY:
- Overall, 14,717 TEAEs were reported over 5 years. The exposure-adjusted incidence rate decreased over time and was 252.48 events per 100 patient-years.
- The most common TEAEs were nasopharyngitis (28.9%), worsening AD (16.7%), upper respiratory tract infection (13.6%), conjunctivitis (10.3%), allergic conjunctivitis (9%), headache (8.1%), oral herpes (7.5%), and injection-site reactions (5.2%).
- Serious and severe TEAE rates were 10.6% and 10.0%, respectively. Exposure-adjusted incidence rates were 6.66 and 6.71 events per 100 patient-years, respectively.
- At week 260, 67.5% of patients had achieved clear or almost clear skin according to the Investigator’s Global Assessment, and 88.9% experienced a 75% or greater improvement in the Eczema Area and Severity Index.
IN PRACTICE:
“Safety and efficacy results from up to 5 years of dupilumab treatment in the LIBERTY AD open-label extension study support dupilumab as a continuous long-term treatment for adults with moderate to severe AD,” the authors concluded.
SOURCE:
The study was led by Lisa A. Beck, MD, University of Rochester, Rochester, New York, and was published online in JAMA Dermatology.
LIMITATIONS:
Study limitations included the absence of a placebo arm and treatment interruptions stemming from protocol changes. The number of patients who received biweekly doses was small. The early conclusion of the trial by the sponsor because of regulatory approval also resulted in a lower number of patients at later stages.
DISCLOSURES:
This study was funded by dupilumab manufacturers Sanofi and Regeneron Pharmaceuticals. Several authors declared ties with various pharmaceutical companies including Sanofi and Regeneron, and several authors were employees of Sanofi or Regeneron. No disclosures were reported by other authors.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
Over 5 years, dupilumab demonstrated acceptable safety and sustained efficacy, with significant improvements in the signs and symptoms of AD, in the treatment of moderate to severe atopic dermatitis (AD).
METHODOLOGY:
- The phase 3 multinational LIBERTY AD open-label extension study evaluated the safety and efficacy of dupilumab in 2677 adults with moderate to severe AD who had previously participated in dupilumab trials over 5 years; 334 patients (12.5%) completed treatment up to 5 years.
- Patients started with subcutaneous dupilumab, initially dosed weekly after a loading dose, then every 2 weeks in 2019.
- The primary outcomes were the incidence and rate of treatment-emergent adverse events (TEAEs).
TAKEAWAY:
- Overall, 14,717 TEAEs were reported over 5 years. The exposure-adjusted incidence rate decreased over time and was 252.48 events per 100 patient-years.
- The most common TEAEs were nasopharyngitis (28.9%), worsening AD (16.7%), upper respiratory tract infection (13.6%), conjunctivitis (10.3%), allergic conjunctivitis (9%), headache (8.1%), oral herpes (7.5%), and injection-site reactions (5.2%).
- Serious and severe TEAE rates were 10.6% and 10.0%, respectively. Exposure-adjusted incidence rates were 6.66 and 6.71 events per 100 patient-years, respectively.
- At week 260, 67.5% of patients had achieved clear or almost clear skin according to the Investigator’s Global Assessment, and 88.9% experienced a 75% or greater improvement in the Eczema Area and Severity Index.
IN PRACTICE:
“Safety and efficacy results from up to 5 years of dupilumab treatment in the LIBERTY AD open-label extension study support dupilumab as a continuous long-term treatment for adults with moderate to severe AD,” the authors concluded.
SOURCE:
The study was led by Lisa A. Beck, MD, University of Rochester, Rochester, New York, and was published online in JAMA Dermatology.
LIMITATIONS:
Study limitations included the absence of a placebo arm and treatment interruptions stemming from protocol changes. The number of patients who received biweekly doses was small. The early conclusion of the trial by the sponsor because of regulatory approval also resulted in a lower number of patients at later stages.
DISCLOSURES:
This study was funded by dupilumab manufacturers Sanofi and Regeneron Pharmaceuticals. Several authors declared ties with various pharmaceutical companies including Sanofi and Regeneron, and several authors were employees of Sanofi or Regeneron. No disclosures were reported by other authors.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
Over 5 years, dupilumab demonstrated acceptable safety and sustained efficacy, with significant improvements in the signs and symptoms of AD, in the treatment of moderate to severe atopic dermatitis (AD).
METHODOLOGY:
- The phase 3 multinational LIBERTY AD open-label extension study evaluated the safety and efficacy of dupilumab in 2677 adults with moderate to severe AD who had previously participated in dupilumab trials over 5 years; 334 patients (12.5%) completed treatment up to 5 years.
- Patients started with subcutaneous dupilumab, initially dosed weekly after a loading dose, then every 2 weeks in 2019.
- The primary outcomes were the incidence and rate of treatment-emergent adverse events (TEAEs).
TAKEAWAY:
- Overall, 14,717 TEAEs were reported over 5 years. The exposure-adjusted incidence rate decreased over time and was 252.48 events per 100 patient-years.
- The most common TEAEs were nasopharyngitis (28.9%), worsening AD (16.7%), upper respiratory tract infection (13.6%), conjunctivitis (10.3%), allergic conjunctivitis (9%), headache (8.1%), oral herpes (7.5%), and injection-site reactions (5.2%).
- Serious and severe TEAE rates were 10.6% and 10.0%, respectively. Exposure-adjusted incidence rates were 6.66 and 6.71 events per 100 patient-years, respectively.
- At week 260, 67.5% of patients had achieved clear or almost clear skin according to the Investigator’s Global Assessment, and 88.9% experienced a 75% or greater improvement in the Eczema Area and Severity Index.
IN PRACTICE:
“Safety and efficacy results from up to 5 years of dupilumab treatment in the LIBERTY AD open-label extension study support dupilumab as a continuous long-term treatment for adults with moderate to severe AD,” the authors concluded.
SOURCE:
The study was led by Lisa A. Beck, MD, University of Rochester, Rochester, New York, and was published online in JAMA Dermatology.
LIMITATIONS:
Study limitations included the absence of a placebo arm and treatment interruptions stemming from protocol changes. The number of patients who received biweekly doses was small. The early conclusion of the trial by the sponsor because of regulatory approval also resulted in a lower number of patients at later stages.
DISCLOSURES:
This study was funded by dupilumab manufacturers Sanofi and Regeneron Pharmaceuticals. Several authors declared ties with various pharmaceutical companies including Sanofi and Regeneron, and several authors were employees of Sanofi or Regeneron. No disclosures were reported by other authors.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
Survey Highlights Real-World Use of Upadacitinib in Adults With Atopic Dermatitis
, while 7.8% rated their itch as minimally improved.
Also, 27.5% reported itch improvement within one day of taking upadacitinib (Rinvoq), an oral Janus kinase inhibitor that was approved to treat moderate to severe AD in adults and children aged ≥ 12 years in January 2022.
“We have a lot of data about upadacitinib from clinical trials, but sometimes there’s a concern that when you start using a medication in the real world, the effectiveness doesn’t match up with the efficacy observed in clinical trials,” the study’s first author, Jonathan I. Silverberg, MD, PhD, professor of dermatology at George Washington University, Washington, said in an interview after the Revolutionizing Atopic Dermatitis conference, where the study was presented during a late-breaking abstract session. “We always want to confirm or reaffirm clinical trial results with real-world data.”
In SCALE-UP, 6191 adults with moderate to severe AD participating in the patient support program for upadacitinib in the United States were invited to complete a one-time online survey about their experience with upadacitinib, including the degree of and time to itch improvement and skin clearance. The researchers reported on 204 patients who completed the survey questions, for a response rate of 3.3%. The mean age of respondents was 45.3 years, their mean age when diagnosed with AD was 30.3 years; 70.1% were women, and 37% were using topical corticosteroids. In addition, 68.6% were White individuals, 12.3% were Black individuals, 8.8% were Asian individuals or Pacific Islanders, and 0.5% were Native Americans/Alaska Natives.
Duration of upadacitinib treatment was 2-6 months for 50.5% of the patients and 7-12 months for the remaining patients. Starting upadacitinib dose was 15 mg for about 95% of patients and 30 mg for nearly 4% of patients. At the time of the survey, 79.4% of patients were receiving upadacitinib 15 mg once a day, and 19.6% were receiving upadacitinib 30 mg once a day.
Improvements in Itch, Skin Clearance
Nearly all experienced improvements in itch, with 86.8% reporting “very much” or “much” improved itch. Relief was rapid, with 87% noticing improvement in itch within 7 days and 27.5% noticing improvement within 1 day. “This is something I have clinically seen,” Dr. Silverberg said.
After receiving upadacitinib, 87% and 86% of patients indicated they were “extremely” or “very” satisfied with the degree and speed of itch improvement, respectively.
In findings related to skin clearance, 90.7% of respondents reported clearer skin after initiating upadacitinib, with 81.4% reporting “very much” or “much” clearer skin. Skin clearance occurred rapidly, with 30.8% of patients noticing clearer skin within 3 days of starting upadacitinib and 89.2% of patients noticing clearer skin within 14 days. The proportions of patients who were “extremely” or “very” satisfied with the degree and speed of skin clearance were 83.8% and 83.2%, respectively.
“What we’re seeing is that the real-world effectiveness [of upadacitinib] aligns with the clinical trial efficacy,” Dr. Silverberg told this news organization. “This study adds even more data to help inform shared decision-making discussion with our patients in trying to decide what medication is best for them.”
He acknowledged certain limitations of the survey, including the lack of a control group of other treatments for comparison, a low response rate, and the potential for response bias. “That said, I think the results remain important, but we value having even more real-world data in the future from prospective registries,” he said. “Those kinds of studies are ongoing, and we look forward to getting more real-world data readouts.”
AbbVie, the manufacturer of upadacitinib, funded the study. Dr. Silverberg reported having served as an advisor, consultant, speaker, and/or investigator for several pharmaceutical companies, including AbbVie. Two authors are AbbVie employees.
A version of this article appeared on Medscape.com.
, while 7.8% rated their itch as minimally improved.
Also, 27.5% reported itch improvement within one day of taking upadacitinib (Rinvoq), an oral Janus kinase inhibitor that was approved to treat moderate to severe AD in adults and children aged ≥ 12 years in January 2022.
“We have a lot of data about upadacitinib from clinical trials, but sometimes there’s a concern that when you start using a medication in the real world, the effectiveness doesn’t match up with the efficacy observed in clinical trials,” the study’s first author, Jonathan I. Silverberg, MD, PhD, professor of dermatology at George Washington University, Washington, said in an interview after the Revolutionizing Atopic Dermatitis conference, where the study was presented during a late-breaking abstract session. “We always want to confirm or reaffirm clinical trial results with real-world data.”
In SCALE-UP, 6191 adults with moderate to severe AD participating in the patient support program for upadacitinib in the United States were invited to complete a one-time online survey about their experience with upadacitinib, including the degree of and time to itch improvement and skin clearance. The researchers reported on 204 patients who completed the survey questions, for a response rate of 3.3%. The mean age of respondents was 45.3 years, their mean age when diagnosed with AD was 30.3 years; 70.1% were women, and 37% were using topical corticosteroids. In addition, 68.6% were White individuals, 12.3% were Black individuals, 8.8% were Asian individuals or Pacific Islanders, and 0.5% were Native Americans/Alaska Natives.
Duration of upadacitinib treatment was 2-6 months for 50.5% of the patients and 7-12 months for the remaining patients. Starting upadacitinib dose was 15 mg for about 95% of patients and 30 mg for nearly 4% of patients. At the time of the survey, 79.4% of patients were receiving upadacitinib 15 mg once a day, and 19.6% were receiving upadacitinib 30 mg once a day.
Improvements in Itch, Skin Clearance
Nearly all experienced improvements in itch, with 86.8% reporting “very much” or “much” improved itch. Relief was rapid, with 87% noticing improvement in itch within 7 days and 27.5% noticing improvement within 1 day. “This is something I have clinically seen,” Dr. Silverberg said.
After receiving upadacitinib, 87% and 86% of patients indicated they were “extremely” or “very” satisfied with the degree and speed of itch improvement, respectively.
In findings related to skin clearance, 90.7% of respondents reported clearer skin after initiating upadacitinib, with 81.4% reporting “very much” or “much” clearer skin. Skin clearance occurred rapidly, with 30.8% of patients noticing clearer skin within 3 days of starting upadacitinib and 89.2% of patients noticing clearer skin within 14 days. The proportions of patients who were “extremely” or “very” satisfied with the degree and speed of skin clearance were 83.8% and 83.2%, respectively.
“What we’re seeing is that the real-world effectiveness [of upadacitinib] aligns with the clinical trial efficacy,” Dr. Silverberg told this news organization. “This study adds even more data to help inform shared decision-making discussion with our patients in trying to decide what medication is best for them.”
He acknowledged certain limitations of the survey, including the lack of a control group of other treatments for comparison, a low response rate, and the potential for response bias. “That said, I think the results remain important, but we value having even more real-world data in the future from prospective registries,” he said. “Those kinds of studies are ongoing, and we look forward to getting more real-world data readouts.”
AbbVie, the manufacturer of upadacitinib, funded the study. Dr. Silverberg reported having served as an advisor, consultant, speaker, and/or investigator for several pharmaceutical companies, including AbbVie. Two authors are AbbVie employees.
A version of this article appeared on Medscape.com.
, while 7.8% rated their itch as minimally improved.
Also, 27.5% reported itch improvement within one day of taking upadacitinib (Rinvoq), an oral Janus kinase inhibitor that was approved to treat moderate to severe AD in adults and children aged ≥ 12 years in January 2022.
“We have a lot of data about upadacitinib from clinical trials, but sometimes there’s a concern that when you start using a medication in the real world, the effectiveness doesn’t match up with the efficacy observed in clinical trials,” the study’s first author, Jonathan I. Silverberg, MD, PhD, professor of dermatology at George Washington University, Washington, said in an interview after the Revolutionizing Atopic Dermatitis conference, where the study was presented during a late-breaking abstract session. “We always want to confirm or reaffirm clinical trial results with real-world data.”
In SCALE-UP, 6191 adults with moderate to severe AD participating in the patient support program for upadacitinib in the United States were invited to complete a one-time online survey about their experience with upadacitinib, including the degree of and time to itch improvement and skin clearance. The researchers reported on 204 patients who completed the survey questions, for a response rate of 3.3%. The mean age of respondents was 45.3 years, their mean age when diagnosed with AD was 30.3 years; 70.1% were women, and 37% were using topical corticosteroids. In addition, 68.6% were White individuals, 12.3% were Black individuals, 8.8% were Asian individuals or Pacific Islanders, and 0.5% were Native Americans/Alaska Natives.
Duration of upadacitinib treatment was 2-6 months for 50.5% of the patients and 7-12 months for the remaining patients. Starting upadacitinib dose was 15 mg for about 95% of patients and 30 mg for nearly 4% of patients. At the time of the survey, 79.4% of patients were receiving upadacitinib 15 mg once a day, and 19.6% were receiving upadacitinib 30 mg once a day.
Improvements in Itch, Skin Clearance
Nearly all experienced improvements in itch, with 86.8% reporting “very much” or “much” improved itch. Relief was rapid, with 87% noticing improvement in itch within 7 days and 27.5% noticing improvement within 1 day. “This is something I have clinically seen,” Dr. Silverberg said.
After receiving upadacitinib, 87% and 86% of patients indicated they were “extremely” or “very” satisfied with the degree and speed of itch improvement, respectively.
In findings related to skin clearance, 90.7% of respondents reported clearer skin after initiating upadacitinib, with 81.4% reporting “very much” or “much” clearer skin. Skin clearance occurred rapidly, with 30.8% of patients noticing clearer skin within 3 days of starting upadacitinib and 89.2% of patients noticing clearer skin within 14 days. The proportions of patients who were “extremely” or “very” satisfied with the degree and speed of skin clearance were 83.8% and 83.2%, respectively.
“What we’re seeing is that the real-world effectiveness [of upadacitinib] aligns with the clinical trial efficacy,” Dr. Silverberg told this news organization. “This study adds even more data to help inform shared decision-making discussion with our patients in trying to decide what medication is best for them.”
He acknowledged certain limitations of the survey, including the lack of a control group of other treatments for comparison, a low response rate, and the potential for response bias. “That said, I think the results remain important, but we value having even more real-world data in the future from prospective registries,” he said. “Those kinds of studies are ongoing, and we look forward to getting more real-world data readouts.”
AbbVie, the manufacturer of upadacitinib, funded the study. Dr. Silverberg reported having served as an advisor, consultant, speaker, and/or investigator for several pharmaceutical companies, including AbbVie. Two authors are AbbVie employees.
A version of this article appeared on Medscape.com.