User login
FDA panel backs atezolizumab for mTNBC – at least for now
On the first day of a historic 3-day meeting about drugs that were granted an accelerated approval by the Food and Drug Administration for cancer indications, the first approval to come under discussion is staying in place, at least for now.
Members of the FDA’s Oncologic Drugs Advisory Committee voted 7-2 in favor of keeping in place the indication for atezolizumab (Tecentriq) for use in a certain form of breast cancer. At the same time, the committee urged the manufacturer, Genentech, to do the research needed to prove the medicine works for these patients.
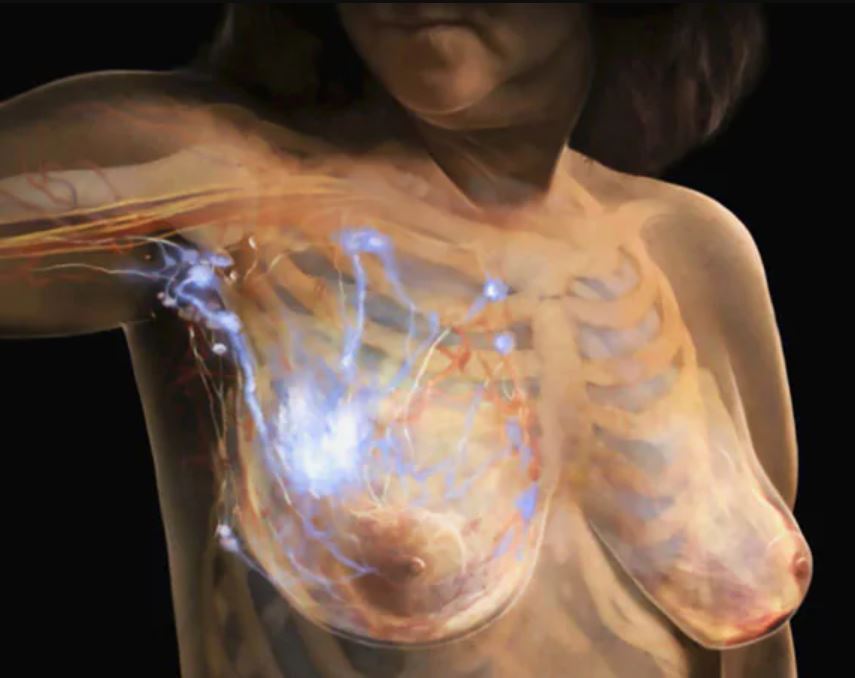
The specific indication is for atezolizumab as part of a combination with nab-paclitaxel for patients with unresectable locally advanced or metastatic triple-negative breast cancer (mTNBC) whose tumors are PD-L1 positive.
The FDA granted accelerated approval in 2019 for this use of atezolizumab, expecting Genentech to produce more extensive evidence of this benefit. But so far, Genentech has not produced the data proving to the FDA that atezolizumab provides the expected benefit.
The drug was already available for use in bladder cancer, having been granted a full approval for this indication in 2016.
Other accelerated approvals withdrawn
This week’s 3-day ODAC meeting is part of the FDA’s broader reconsideration of what it has described as “dangling accelerated approvals.”
Earlier discussions between the FDA and drugmakers have already triggered four voluntary withdrawals of cancer indications with these accelerated approvals, noted Julia A. Beaver, MD, and Richard Pazdur, MD, two of the FDA’s top regulators of oncology medicine, in an April 21 perspective article in the New England Journal of Medicine.
“The small percentage of drugs whose clinical benefit is ultimately not confirmed should be viewed not as a failure of accelerated approval but rather as an expected trade-off in expediting drug development that benefits patients with severe or life-threatening diseases,” Dr. Beaver and Dr. Pazdur wrote.
But making these calls can be tough. On the first day of the meeting, even ODAC panelists who backed Genentech’s bid to maintain an mTNBC indication for atezolizumab expressed discomfort with this choice.
The FDA granted the accelerated approval for use of this drug in March 2019 based on improved progression-free survival from the IMpassion130 trial. But the drug fell short in subsequent efforts to confirm the results seen in that study. The confirmatory IMpassion131 trial failed to meet the primary endpoint, the FDA staff noted in briefing materials for the ODAC meeting.
ODAC panelist Stan Lipkowitz, MD, PhD, of the National Cancer Institute, said he expected this vote had been a tough one for all members serving on ODAC that day.
“In some ways, the purist in me said I should have voted no. But when I looked at the data, there are a couple of things that struck me,” said Dr. Lipkowitz, who is the chief of the Women’s Malignancies Branch at NCI’s Center for Cancer Research. “First of all, the landscape hasn’t changed. There’s really no therapy in the first line for triple-negative metastatic that is shown to improve survival.”
Dr. Lipkowitz emphasized that Genentech needs to continue to try to prove atezolizumab works in this setting.
“There needs to be confirmatory study,” Dr. Lipkowitz concluded.
ODAC panelist Matthew Ellis, MD, PhD, of Baylor College of Medicine, Houston, said he also understood the difficult outlook for women fighting this cancer, but he voted against maintaining the approval.
“It’s not that I don’t feel the tragedy of these women,” said Dr. Ellis, citing his own decades of clinical experience.
“I just think that the data are the data,” Dr. Ellis said, adding that, in his view, “the only correct interpretation” of the evidence supported a vote against allowing the indication to stay.
The FDA considers the recommendations of its advisory committees but is not bound by them.
In a statement issued after the vote, Genentech said it intends to work with the FDA to determine the next steps for this indication of atezolizumab because “the clinically meaningful benefit demonstrated in the IMpassion130 study remains.”
The ODAC meeting continues for 2 more days, and will consider five more cancer indications that have been granted an accelerated approval.
A version of this article first appeared on Medscape.com.
On the first day of a historic 3-day meeting about drugs that were granted an accelerated approval by the Food and Drug Administration for cancer indications, the first approval to come under discussion is staying in place, at least for now.
Members of the FDA’s Oncologic Drugs Advisory Committee voted 7-2 in favor of keeping in place the indication for atezolizumab (Tecentriq) for use in a certain form of breast cancer. At the same time, the committee urged the manufacturer, Genentech, to do the research needed to prove the medicine works for these patients.
The specific indication is for atezolizumab as part of a combination with nab-paclitaxel for patients with unresectable locally advanced or metastatic triple-negative breast cancer (mTNBC) whose tumors are PD-L1 positive.
The FDA granted accelerated approval in 2019 for this use of atezolizumab, expecting Genentech to produce more extensive evidence of this benefit. But so far, Genentech has not produced the data proving to the FDA that atezolizumab provides the expected benefit.
The drug was already available for use in bladder cancer, having been granted a full approval for this indication in 2016.
Other accelerated approvals withdrawn
This week’s 3-day ODAC meeting is part of the FDA’s broader reconsideration of what it has described as “dangling accelerated approvals.”
Earlier discussions between the FDA and drugmakers have already triggered four voluntary withdrawals of cancer indications with these accelerated approvals, noted Julia A. Beaver, MD, and Richard Pazdur, MD, two of the FDA’s top regulators of oncology medicine, in an April 21 perspective article in the New England Journal of Medicine.
“The small percentage of drugs whose clinical benefit is ultimately not confirmed should be viewed not as a failure of accelerated approval but rather as an expected trade-off in expediting drug development that benefits patients with severe or life-threatening diseases,” Dr. Beaver and Dr. Pazdur wrote.
But making these calls can be tough. On the first day of the meeting, even ODAC panelists who backed Genentech’s bid to maintain an mTNBC indication for atezolizumab expressed discomfort with this choice.
The FDA granted the accelerated approval for use of this drug in March 2019 based on improved progression-free survival from the IMpassion130 trial. But the drug fell short in subsequent efforts to confirm the results seen in that study. The confirmatory IMpassion131 trial failed to meet the primary endpoint, the FDA staff noted in briefing materials for the ODAC meeting.
ODAC panelist Stan Lipkowitz, MD, PhD, of the National Cancer Institute, said he expected this vote had been a tough one for all members serving on ODAC that day.
“In some ways, the purist in me said I should have voted no. But when I looked at the data, there are a couple of things that struck me,” said Dr. Lipkowitz, who is the chief of the Women’s Malignancies Branch at NCI’s Center for Cancer Research. “First of all, the landscape hasn’t changed. There’s really no therapy in the first line for triple-negative metastatic that is shown to improve survival.”
Dr. Lipkowitz emphasized that Genentech needs to continue to try to prove atezolizumab works in this setting.
“There needs to be confirmatory study,” Dr. Lipkowitz concluded.
ODAC panelist Matthew Ellis, MD, PhD, of Baylor College of Medicine, Houston, said he also understood the difficult outlook for women fighting this cancer, but he voted against maintaining the approval.
“It’s not that I don’t feel the tragedy of these women,” said Dr. Ellis, citing his own decades of clinical experience.
“I just think that the data are the data,” Dr. Ellis said, adding that, in his view, “the only correct interpretation” of the evidence supported a vote against allowing the indication to stay.
The FDA considers the recommendations of its advisory committees but is not bound by them.
In a statement issued after the vote, Genentech said it intends to work with the FDA to determine the next steps for this indication of atezolizumab because “the clinically meaningful benefit demonstrated in the IMpassion130 study remains.”
The ODAC meeting continues for 2 more days, and will consider five more cancer indications that have been granted an accelerated approval.
A version of this article first appeared on Medscape.com.
On the first day of a historic 3-day meeting about drugs that were granted an accelerated approval by the Food and Drug Administration for cancer indications, the first approval to come under discussion is staying in place, at least for now.
Members of the FDA’s Oncologic Drugs Advisory Committee voted 7-2 in favor of keeping in place the indication for atezolizumab (Tecentriq) for use in a certain form of breast cancer. At the same time, the committee urged the manufacturer, Genentech, to do the research needed to prove the medicine works for these patients.
The specific indication is for atezolizumab as part of a combination with nab-paclitaxel for patients with unresectable locally advanced or metastatic triple-negative breast cancer (mTNBC) whose tumors are PD-L1 positive.
The FDA granted accelerated approval in 2019 for this use of atezolizumab, expecting Genentech to produce more extensive evidence of this benefit. But so far, Genentech has not produced the data proving to the FDA that atezolizumab provides the expected benefit.
The drug was already available for use in bladder cancer, having been granted a full approval for this indication in 2016.
Other accelerated approvals withdrawn
This week’s 3-day ODAC meeting is part of the FDA’s broader reconsideration of what it has described as “dangling accelerated approvals.”
Earlier discussions between the FDA and drugmakers have already triggered four voluntary withdrawals of cancer indications with these accelerated approvals, noted Julia A. Beaver, MD, and Richard Pazdur, MD, two of the FDA’s top regulators of oncology medicine, in an April 21 perspective article in the New England Journal of Medicine.
“The small percentage of drugs whose clinical benefit is ultimately not confirmed should be viewed not as a failure of accelerated approval but rather as an expected trade-off in expediting drug development that benefits patients with severe or life-threatening diseases,” Dr. Beaver and Dr. Pazdur wrote.
But making these calls can be tough. On the first day of the meeting, even ODAC panelists who backed Genentech’s bid to maintain an mTNBC indication for atezolizumab expressed discomfort with this choice.
The FDA granted the accelerated approval for use of this drug in March 2019 based on improved progression-free survival from the IMpassion130 trial. But the drug fell short in subsequent efforts to confirm the results seen in that study. The confirmatory IMpassion131 trial failed to meet the primary endpoint, the FDA staff noted in briefing materials for the ODAC meeting.
ODAC panelist Stan Lipkowitz, MD, PhD, of the National Cancer Institute, said he expected this vote had been a tough one for all members serving on ODAC that day.
“In some ways, the purist in me said I should have voted no. But when I looked at the data, there are a couple of things that struck me,” said Dr. Lipkowitz, who is the chief of the Women’s Malignancies Branch at NCI’s Center for Cancer Research. “First of all, the landscape hasn’t changed. There’s really no therapy in the first line for triple-negative metastatic that is shown to improve survival.”
Dr. Lipkowitz emphasized that Genentech needs to continue to try to prove atezolizumab works in this setting.
“There needs to be confirmatory study,” Dr. Lipkowitz concluded.
ODAC panelist Matthew Ellis, MD, PhD, of Baylor College of Medicine, Houston, said he also understood the difficult outlook for women fighting this cancer, but he voted against maintaining the approval.
“It’s not that I don’t feel the tragedy of these women,” said Dr. Ellis, citing his own decades of clinical experience.
“I just think that the data are the data,” Dr. Ellis said, adding that, in his view, “the only correct interpretation” of the evidence supported a vote against allowing the indication to stay.
The FDA considers the recommendations of its advisory committees but is not bound by them.
In a statement issued after the vote, Genentech said it intends to work with the FDA to determine the next steps for this indication of atezolizumab because “the clinically meaningful benefit demonstrated in the IMpassion130 study remains.”
The ODAC meeting continues for 2 more days, and will consider five more cancer indications that have been granted an accelerated approval.
A version of this article first appeared on Medscape.com.
Cell-free DNA improves response prediction in breast cancer
When the two techniques were in agreement, the accuracy of response prediction was 92.6% in the study, with a predictive value for complete response of 87.5% and a predictive value for absence of complete response of 94.7%, which was substantially better than either method alone.
“Our work identifies a new parameter that is easily combinable with MRI for a more accurate prediction of response following neoadjuvant treatment, with possible implications for current protocols for the evaluation of nodal residual disease,” researcher Francesco Ravera, MD, PhD, of the University of Genoa (Italy), said in a press release.
Dr. Ravera and colleagues presented their research in a poster at the American Association for Cancer Research Annual Meeting 2021: Week 1 (Abstract LB063).
Accurate response prediction is important because it guides subsequent surgical management, Dr. Ravera and colleagues noted. Pathological complete responders – generally about 25% of patients after neoadjuvant therapy – typically undergo a sentinel lymph node biopsy to ensure cancer hasn’t spread, while incomplete responders often have a complete axillary lymph node dissection.
Response is currently assessed by MRI, but accuracy is suboptimal, the researchers noted. A more accurate method might “allow the omission of sentinel lymph node biopsy in complete responders, which could be replaced by longitudinal radiologic monitoring. This would represent substantial progress in the pursuit of an effective, minimally invasive treatment,” Dr. Ravera said.
He and his colleagues turned to plasma cfDNA because it has shown potential for providing useful diagnostic, recurrence, and treatment response information in neoplastic patients.
When healthy cells die, they release similarly sized DNA fragments into the blood, but cancer cells release fragments of varying sizes. The heart of the research was using electrophoresis to assess the degree of fragmentation – called cfDNA integrity – in plasma samples from 38 patients after anthracycline/taxane-based regimens.
The researchers compared how well cfDNA, preoperative MRI, and the combination of the two methods predicted response according to surgical histology.
A total of 11 patients had pathological complete responses to neoadjuvant therapy.
The ratio of large 321-1,000 base pair sized fragments to smaller 150-220 base pair sized fragments, which the team dubbed the “cfDNA integrity index,” best predicted response. At a cutoff above 2.71, the index was 81.6% accurate in predicting pathological complete response, with a sensitivity of 81.8% and specificity of 81.5%.
The predictive power wasn’t much better than MRI, which was 77.1% accurate, with a sensitivity of 72.7% and a specificity of 81.5%.
The two techniques were concordant in their prediction in over two-thirds of patients. When the techniques agreed, accuracy was over 90%.
Prospective studies are needed to evaluate the cfDNA integrity index in combination with MRI, the researchers concluded.
The study was sponsored by the University of Genoa and others. Dr. Ravera disclosed no conflicts of interest.
When the two techniques were in agreement, the accuracy of response prediction was 92.6% in the study, with a predictive value for complete response of 87.5% and a predictive value for absence of complete response of 94.7%, which was substantially better than either method alone.
“Our work identifies a new parameter that is easily combinable with MRI for a more accurate prediction of response following neoadjuvant treatment, with possible implications for current protocols for the evaluation of nodal residual disease,” researcher Francesco Ravera, MD, PhD, of the University of Genoa (Italy), said in a press release.
Dr. Ravera and colleagues presented their research in a poster at the American Association for Cancer Research Annual Meeting 2021: Week 1 (Abstract LB063).
Accurate response prediction is important because it guides subsequent surgical management, Dr. Ravera and colleagues noted. Pathological complete responders – generally about 25% of patients after neoadjuvant therapy – typically undergo a sentinel lymph node biopsy to ensure cancer hasn’t spread, while incomplete responders often have a complete axillary lymph node dissection.
Response is currently assessed by MRI, but accuracy is suboptimal, the researchers noted. A more accurate method might “allow the omission of sentinel lymph node biopsy in complete responders, which could be replaced by longitudinal radiologic monitoring. This would represent substantial progress in the pursuit of an effective, minimally invasive treatment,” Dr. Ravera said.
He and his colleagues turned to plasma cfDNA because it has shown potential for providing useful diagnostic, recurrence, and treatment response information in neoplastic patients.
When healthy cells die, they release similarly sized DNA fragments into the blood, but cancer cells release fragments of varying sizes. The heart of the research was using electrophoresis to assess the degree of fragmentation – called cfDNA integrity – in plasma samples from 38 patients after anthracycline/taxane-based regimens.
The researchers compared how well cfDNA, preoperative MRI, and the combination of the two methods predicted response according to surgical histology.
A total of 11 patients had pathological complete responses to neoadjuvant therapy.
The ratio of large 321-1,000 base pair sized fragments to smaller 150-220 base pair sized fragments, which the team dubbed the “cfDNA integrity index,” best predicted response. At a cutoff above 2.71, the index was 81.6% accurate in predicting pathological complete response, with a sensitivity of 81.8% and specificity of 81.5%.
The predictive power wasn’t much better than MRI, which was 77.1% accurate, with a sensitivity of 72.7% and a specificity of 81.5%.
The two techniques were concordant in their prediction in over two-thirds of patients. When the techniques agreed, accuracy was over 90%.
Prospective studies are needed to evaluate the cfDNA integrity index in combination with MRI, the researchers concluded.
The study was sponsored by the University of Genoa and others. Dr. Ravera disclosed no conflicts of interest.
When the two techniques were in agreement, the accuracy of response prediction was 92.6% in the study, with a predictive value for complete response of 87.5% and a predictive value for absence of complete response of 94.7%, which was substantially better than either method alone.
“Our work identifies a new parameter that is easily combinable with MRI for a more accurate prediction of response following neoadjuvant treatment, with possible implications for current protocols for the evaluation of nodal residual disease,” researcher Francesco Ravera, MD, PhD, of the University of Genoa (Italy), said in a press release.
Dr. Ravera and colleagues presented their research in a poster at the American Association for Cancer Research Annual Meeting 2021: Week 1 (Abstract LB063).
Accurate response prediction is important because it guides subsequent surgical management, Dr. Ravera and colleagues noted. Pathological complete responders – generally about 25% of patients after neoadjuvant therapy – typically undergo a sentinel lymph node biopsy to ensure cancer hasn’t spread, while incomplete responders often have a complete axillary lymph node dissection.
Response is currently assessed by MRI, but accuracy is suboptimal, the researchers noted. A more accurate method might “allow the omission of sentinel lymph node biopsy in complete responders, which could be replaced by longitudinal radiologic monitoring. This would represent substantial progress in the pursuit of an effective, minimally invasive treatment,” Dr. Ravera said.
He and his colleagues turned to plasma cfDNA because it has shown potential for providing useful diagnostic, recurrence, and treatment response information in neoplastic patients.
When healthy cells die, they release similarly sized DNA fragments into the blood, but cancer cells release fragments of varying sizes. The heart of the research was using electrophoresis to assess the degree of fragmentation – called cfDNA integrity – in plasma samples from 38 patients after anthracycline/taxane-based regimens.
The researchers compared how well cfDNA, preoperative MRI, and the combination of the two methods predicted response according to surgical histology.
A total of 11 patients had pathological complete responses to neoadjuvant therapy.
The ratio of large 321-1,000 base pair sized fragments to smaller 150-220 base pair sized fragments, which the team dubbed the “cfDNA integrity index,” best predicted response. At a cutoff above 2.71, the index was 81.6% accurate in predicting pathological complete response, with a sensitivity of 81.8% and specificity of 81.5%.
The predictive power wasn’t much better than MRI, which was 77.1% accurate, with a sensitivity of 72.7% and a specificity of 81.5%.
The two techniques were concordant in their prediction in over two-thirds of patients. When the techniques agreed, accuracy was over 90%.
Prospective studies are needed to evaluate the cfDNA integrity index in combination with MRI, the researchers concluded.
The study was sponsored by the University of Genoa and others. Dr. Ravera disclosed no conflicts of interest.
FROM AACR 2021
Quicker fertility rebound in young women with breast cancer
Researchers found that omitting cyclophosphamide from a regimen of epirubicin and paclitaxel increased the likelihood of an early return of menses, and there was a trend toward improved disease-free survival.
The phase 3 SPECTRUM trial involved 521 women with estrogen receptor–positive, HER2-negative breast cancer who had undergone definitive surgery at one of eight institutions in China. The average age of the patients was 34 years.
Cyclophosphamide is a standard component of adjuvant chemotherapy, but it’s strongly associated with premature ovarian failure and infertility.
“For the first time, we demonstrate that a cyclophosphamide-free regimen [can] increase the rate of menses recovery without compromising survival,” said the researchers, led by Ke-Da Yu, MD, PhD, of the Fudan University Shanghai (China) Cancer Center.
They also reported that, among the women who tried to conceive at a later date, there was a higher pregnancy success rate among those who did not take cyclophosphamide.
“Our findings can be extrapolated to patients with other subtypes of breast cancer, such as triple-negative or HER2-enriched, because the effect of paclitaxel and cyclophosphamide on menstrual resumption is not subtype specific,” the investigators commented.
The results were published online in the Journal of the National Cancer Institute.
This is the first prospective trial specifically designed to find an adjuvant breast cancer regimen less toxic to the ovaries. The “investigators ... should be applauded,” wrote Matteo Lambertini, MD, PhD, of the University of Genova (Italy), and Ann Partridge, MD, of the Dana-Farber Cancer Institute in Boston, in an accompanying editorial.
Although promising, there are a few caveats, the editorialists wrote. In a past trial of doxorubicin and docetaxel in lieu of a cyclophosphamide regimen, disease outcomes were inferior. There is also a question as to whether the SPECTRUM results apply to non-Asian women.
The editorialists also noted that enrollment in this trial ended in 2016, before it was recommended that ovarian suppression be used in conjunction with adjuvant chemotherapy to prevent premature menopause.
“[It’s] notable that the absolute benefit in reducing [premature ovarian insufficiency] rates with the use of a cyclophosphamide-free regimen is similar to the effect demonstrated with the administration of a gonadotropin-releasing hormone agonist during cyclophosphamide-based chemotherapy,” they commented. It’s possible that combining the two approaches might have an additive effect, but for now the possibility remains unknown.
In addition, the SPECTRUM trial predates the widespread use of genetic testing to guide treatment, the editorialists pointed out.
“Therefore, caution should be taken in adopting wholesale such regimens,” Dr. Lambertini and Dr. Partridge said.
Switch to paclitaxel
The research team was inspired by previous reports that swapping out cyclophosphamide for paclitaxel did not reduce adjuvant efficacy in the general breast cancer population.
The SPECTRUM trial randomly assigned 260 women to receive a cyclophosphamide-free regimen of epirubicin (75 mg/m2) and paclitaxel (175 mg/m2) every 3 weeks for four cycles followed by weekly paclitaxel (80 mg/m2) for 12 weeks.
Another 261 women were randomly assigned to receive cyclophosphamide (600 mg/m2) and epirubicin (75 mg/m2) every 3 weeks for four cycles followed by weekly paclitaxel (80 mg/m2) for 12 weeks. These patients constituted the control group.
A year after completing chemotherapy, 63.1% of the cyclophosphamide-free arm versus 48.3% of the control group, had resumed menses, defined as having two consecutive menstrual cycles or one cycle but with premenopausal levels of estradiol and follicle-stimulating hormone (P < .001).
Another caveat of the study is that assessments of women who resumed menses were conducted at the 1-year point; rates may have been higher in the cyclophosphamide arm had the investigators conducted the assessments at 2 years, the editorialists said.
The 5-year disease-free survival was 84.7% in the cyclophosphamide-free arm versus 78.3% in the control group, an absolute difference of 14.8% (P = .07).
Patients with node-positive disease appeared to benefit the most from cyclophosphamide sparing.
There were no statistically significant differences in overall or distant disease-free survival.
Higher pregnancy rates
Almost 18% of women in the experimental arm reported trying to conceive, and 9.6% of them did so. About 10% of women in the cyclophosphamide arm tried to conceive, and 2.7% did so (P = .03).
The median interval between randomization and pregnancy was 42 months.
For all of the women who became pregnant, endocrine therapy was interrupted. “Women who temporarily interrupt endocrine therapy due to pregnancy should be reminded to resume endocrine therapy following attempted or successful pregnancy,” the investigators wrote.
The patients were taking tamoxifen at least 5 years after receiving chemotherapy, most often as monotherapy. About 5% of the patients underwent up-front ovarian suppression with an aromatase inhibitor, which is a current standard option.
The study was supported by the National Natural Science Foundation of China and other organizations. The investigators and Dr. Partridge disclosed no relevant financial relationships. Dr. Lambertini has consulted for and/or has received speakers fees from Roche, AstraZeneca, Lilly, Novartis, and other companies.
A version of this article first appeared on Medscape.com.
Researchers found that omitting cyclophosphamide from a regimen of epirubicin and paclitaxel increased the likelihood of an early return of menses, and there was a trend toward improved disease-free survival.
The phase 3 SPECTRUM trial involved 521 women with estrogen receptor–positive, HER2-negative breast cancer who had undergone definitive surgery at one of eight institutions in China. The average age of the patients was 34 years.
Cyclophosphamide is a standard component of adjuvant chemotherapy, but it’s strongly associated with premature ovarian failure and infertility.
“For the first time, we demonstrate that a cyclophosphamide-free regimen [can] increase the rate of menses recovery without compromising survival,” said the researchers, led by Ke-Da Yu, MD, PhD, of the Fudan University Shanghai (China) Cancer Center.
They also reported that, among the women who tried to conceive at a later date, there was a higher pregnancy success rate among those who did not take cyclophosphamide.
“Our findings can be extrapolated to patients with other subtypes of breast cancer, such as triple-negative or HER2-enriched, because the effect of paclitaxel and cyclophosphamide on menstrual resumption is not subtype specific,” the investigators commented.
The results were published online in the Journal of the National Cancer Institute.
This is the first prospective trial specifically designed to find an adjuvant breast cancer regimen less toxic to the ovaries. The “investigators ... should be applauded,” wrote Matteo Lambertini, MD, PhD, of the University of Genova (Italy), and Ann Partridge, MD, of the Dana-Farber Cancer Institute in Boston, in an accompanying editorial.
Although promising, there are a few caveats, the editorialists wrote. In a past trial of doxorubicin and docetaxel in lieu of a cyclophosphamide regimen, disease outcomes were inferior. There is also a question as to whether the SPECTRUM results apply to non-Asian women.
The editorialists also noted that enrollment in this trial ended in 2016, before it was recommended that ovarian suppression be used in conjunction with adjuvant chemotherapy to prevent premature menopause.
“[It’s] notable that the absolute benefit in reducing [premature ovarian insufficiency] rates with the use of a cyclophosphamide-free regimen is similar to the effect demonstrated with the administration of a gonadotropin-releasing hormone agonist during cyclophosphamide-based chemotherapy,” they commented. It’s possible that combining the two approaches might have an additive effect, but for now the possibility remains unknown.
In addition, the SPECTRUM trial predates the widespread use of genetic testing to guide treatment, the editorialists pointed out.
“Therefore, caution should be taken in adopting wholesale such regimens,” Dr. Lambertini and Dr. Partridge said.
Switch to paclitaxel
The research team was inspired by previous reports that swapping out cyclophosphamide for paclitaxel did not reduce adjuvant efficacy in the general breast cancer population.
The SPECTRUM trial randomly assigned 260 women to receive a cyclophosphamide-free regimen of epirubicin (75 mg/m2) and paclitaxel (175 mg/m2) every 3 weeks for four cycles followed by weekly paclitaxel (80 mg/m2) for 12 weeks.
Another 261 women were randomly assigned to receive cyclophosphamide (600 mg/m2) and epirubicin (75 mg/m2) every 3 weeks for four cycles followed by weekly paclitaxel (80 mg/m2) for 12 weeks. These patients constituted the control group.
A year after completing chemotherapy, 63.1% of the cyclophosphamide-free arm versus 48.3% of the control group, had resumed menses, defined as having two consecutive menstrual cycles or one cycle but with premenopausal levels of estradiol and follicle-stimulating hormone (P < .001).
Another caveat of the study is that assessments of women who resumed menses were conducted at the 1-year point; rates may have been higher in the cyclophosphamide arm had the investigators conducted the assessments at 2 years, the editorialists said.
The 5-year disease-free survival was 84.7% in the cyclophosphamide-free arm versus 78.3% in the control group, an absolute difference of 14.8% (P = .07).
Patients with node-positive disease appeared to benefit the most from cyclophosphamide sparing.
There were no statistically significant differences in overall or distant disease-free survival.
Higher pregnancy rates
Almost 18% of women in the experimental arm reported trying to conceive, and 9.6% of them did so. About 10% of women in the cyclophosphamide arm tried to conceive, and 2.7% did so (P = .03).
The median interval between randomization and pregnancy was 42 months.
For all of the women who became pregnant, endocrine therapy was interrupted. “Women who temporarily interrupt endocrine therapy due to pregnancy should be reminded to resume endocrine therapy following attempted or successful pregnancy,” the investigators wrote.
The patients were taking tamoxifen at least 5 years after receiving chemotherapy, most often as monotherapy. About 5% of the patients underwent up-front ovarian suppression with an aromatase inhibitor, which is a current standard option.
The study was supported by the National Natural Science Foundation of China and other organizations. The investigators and Dr. Partridge disclosed no relevant financial relationships. Dr. Lambertini has consulted for and/or has received speakers fees from Roche, AstraZeneca, Lilly, Novartis, and other companies.
A version of this article first appeared on Medscape.com.
Researchers found that omitting cyclophosphamide from a regimen of epirubicin and paclitaxel increased the likelihood of an early return of menses, and there was a trend toward improved disease-free survival.
The phase 3 SPECTRUM trial involved 521 women with estrogen receptor–positive, HER2-negative breast cancer who had undergone definitive surgery at one of eight institutions in China. The average age of the patients was 34 years.
Cyclophosphamide is a standard component of adjuvant chemotherapy, but it’s strongly associated with premature ovarian failure and infertility.
“For the first time, we demonstrate that a cyclophosphamide-free regimen [can] increase the rate of menses recovery without compromising survival,” said the researchers, led by Ke-Da Yu, MD, PhD, of the Fudan University Shanghai (China) Cancer Center.
They also reported that, among the women who tried to conceive at a later date, there was a higher pregnancy success rate among those who did not take cyclophosphamide.
“Our findings can be extrapolated to patients with other subtypes of breast cancer, such as triple-negative or HER2-enriched, because the effect of paclitaxel and cyclophosphamide on menstrual resumption is not subtype specific,” the investigators commented.
The results were published online in the Journal of the National Cancer Institute.
This is the first prospective trial specifically designed to find an adjuvant breast cancer regimen less toxic to the ovaries. The “investigators ... should be applauded,” wrote Matteo Lambertini, MD, PhD, of the University of Genova (Italy), and Ann Partridge, MD, of the Dana-Farber Cancer Institute in Boston, in an accompanying editorial.
Although promising, there are a few caveats, the editorialists wrote. In a past trial of doxorubicin and docetaxel in lieu of a cyclophosphamide regimen, disease outcomes were inferior. There is also a question as to whether the SPECTRUM results apply to non-Asian women.
The editorialists also noted that enrollment in this trial ended in 2016, before it was recommended that ovarian suppression be used in conjunction with adjuvant chemotherapy to prevent premature menopause.
“[It’s] notable that the absolute benefit in reducing [premature ovarian insufficiency] rates with the use of a cyclophosphamide-free regimen is similar to the effect demonstrated with the administration of a gonadotropin-releasing hormone agonist during cyclophosphamide-based chemotherapy,” they commented. It’s possible that combining the two approaches might have an additive effect, but for now the possibility remains unknown.
In addition, the SPECTRUM trial predates the widespread use of genetic testing to guide treatment, the editorialists pointed out.
“Therefore, caution should be taken in adopting wholesale such regimens,” Dr. Lambertini and Dr. Partridge said.
Switch to paclitaxel
The research team was inspired by previous reports that swapping out cyclophosphamide for paclitaxel did not reduce adjuvant efficacy in the general breast cancer population.
The SPECTRUM trial randomly assigned 260 women to receive a cyclophosphamide-free regimen of epirubicin (75 mg/m2) and paclitaxel (175 mg/m2) every 3 weeks for four cycles followed by weekly paclitaxel (80 mg/m2) for 12 weeks.
Another 261 women were randomly assigned to receive cyclophosphamide (600 mg/m2) and epirubicin (75 mg/m2) every 3 weeks for four cycles followed by weekly paclitaxel (80 mg/m2) for 12 weeks. These patients constituted the control group.
A year after completing chemotherapy, 63.1% of the cyclophosphamide-free arm versus 48.3% of the control group, had resumed menses, defined as having two consecutive menstrual cycles or one cycle but with premenopausal levels of estradiol and follicle-stimulating hormone (P < .001).
Another caveat of the study is that assessments of women who resumed menses were conducted at the 1-year point; rates may have been higher in the cyclophosphamide arm had the investigators conducted the assessments at 2 years, the editorialists said.
The 5-year disease-free survival was 84.7% in the cyclophosphamide-free arm versus 78.3% in the control group, an absolute difference of 14.8% (P = .07).
Patients with node-positive disease appeared to benefit the most from cyclophosphamide sparing.
There were no statistically significant differences in overall or distant disease-free survival.
Higher pregnancy rates
Almost 18% of women in the experimental arm reported trying to conceive, and 9.6% of them did so. About 10% of women in the cyclophosphamide arm tried to conceive, and 2.7% did so (P = .03).
The median interval between randomization and pregnancy was 42 months.
For all of the women who became pregnant, endocrine therapy was interrupted. “Women who temporarily interrupt endocrine therapy due to pregnancy should be reminded to resume endocrine therapy following attempted or successful pregnancy,” the investigators wrote.
The patients were taking tamoxifen at least 5 years after receiving chemotherapy, most often as monotherapy. About 5% of the patients underwent up-front ovarian suppression with an aromatase inhibitor, which is a current standard option.
The study was supported by the National Natural Science Foundation of China and other organizations. The investigators and Dr. Partridge disclosed no relevant financial relationships. Dr. Lambertini has consulted for and/or has received speakers fees from Roche, AstraZeneca, Lilly, Novartis, and other companies.
A version of this article first appeared on Medscape.com.
Personalized cancer vaccine shows early promise across tumor types
The vaccine, PGV-001, was given to 13 patients with solid tumors or multiple myeloma who had a high risk of recurrence after surgery or autologous stem cell transplant.
At last follow-up, four patients were still alive without evidence of disease and had not received subsequent therapy, four were alive and receiving therapy, three had died, and two were lost to follow-up.
Thomas Marron, MD, PhD , of Mount Sinai in New York presented these results in a poster at the American Association for Cancer Research Annual Meeting 2021: Week 1 ( Abstract LB048 ). Data in the abstract differ from the data presented.
“While cancer immunotherapy has revolutionized the treatment of cancer, we know that the majority of patients fail to achieve significant clinical response,” Dr. Marron said during his presentation. “One reason for this may be due to lack of preexisting primed T-cell response needed for PD-1 blockade to have a significant effect. To address this, personalized neoantigen vaccines may help prime an improved immune response against tumor cells.”
With this in mind, Dr. Marron and colleagues developed PGV-001, a vaccine consisting of patient-specific synthetic neoantigen peptides given to patients in the adjuvant setting.
Creating a personalized vaccine
The researchers synthesized PGV-001 for 15 patients with advanced malignancies. The patients first underwent tumor and germline DNA sequencing as well as HLA typing. Bulk RNA sequencing was performed on patients’ tumors as well.
Then, the researchers used a computational pipeline called OpenVax to identify candidate neoantigens. This pipeline, developed at Mount Sinai, identified and prioritized candidate neoantigens using predicted MHC class I binding affinity and neoantigen abundance.
OpenVax identified an average of 71.5 neoantigens per patient (range, 7-193). The goal was to synthesize a maximum of 10 peptides per patient, but two patients did not have an adequate number of neoantigens.
Vaccine administration
The peptides were administered over the course of 27 weeks along with poly-ICLC and a tetanus helper peptide. Before receiving their vaccine doses, patients with solid tumors had undergone curative-intent surgery, and those with multiple myeloma had undergone autologous stem cell transplant.
“Most experimental personalized cancer vaccines are administered in the metastatic setting, but prior research indicates that immunotherapies tend to be more effective in patients who have less cancer spread,” principal investigator Nina Bhardwaj, MD, PhD , of Mount Sinai, explained in a press release .
“We have, therefore, developed a neoantigen vaccine that is administered after standard-of-care adjuvant therapy, such as surgery in solid tumors and bone marrow transplant in multiple myeloma, when patients have minimal, typically microscopic, residual disease.”
Feasibility, safety, and immunogenicity
PGV-001 was synthesized for 15 patients and administered to 13 of them. Six of the 13 patients had head and neck squamous cell carcinoma, three had multiple myeloma, two had non–small cell lung cancer, one had breast cancer, and one had urothelial carcinoma.
Eleven patients received all 10 intended doses, and two patients received at least 8 doses.
“The vaccine was well tolerated, with only half of patients experiencing mild, grade 1 adverse events,” Dr. Marron said.
Transient injection site reactions occurred in four patients, and grade 1 fever was reported in one patient.
Immune monitoring is ongoing, but an initial analysis in one patient showed “robust responses” in CD4 and CD8 T cells by intracellular cytokine staining for interferon-gamma, tumor necrosis factor–alpha, and interleukin-2 after in vitro expansion in the presence of vaccine antigens, according to the researchers.
Dr. Marron noted that robust T-cell reactivity was seen at the completion of all 10 doses but was not seen after the 6th dose, and this supports the need for a prolonged dosing schedule.
Survival and subsequent therapy
At a mean follow-up of 880 days, four patients had no evidence of disease and had not received subsequent therapy. This includes one patient with stage IIIA non–small cell lung cancer, one with stage IVA HER-2 positive breast cancer, one with stage II urothelial carcinoma, and one with multiple myeloma.
Four patients were alive and receiving subsequent lines of therapy. Two of these patients had significant responses to anti–PD-1 therapy.
Three patients have died, two of whom had documented recurrence of their malignancy. The last two patients were lost to follow-up without documented recurrence.
“Our results demonstrate that the OpenVax pipeline is a viable approach to generate a safe, personalized cancer vaccine, which could potentially be used to treat a range of tumor types,” Dr. Bhardwaj said.
Trials combining neoantigens identified with the OpenVax platform are ongoing in patients with urothelial carcinoma and glioblastoma multiforme, Dr. Marron said.
The current study ( NCT02721043 ) is sponsored by Dr. Bhardwaj. Dr. Marron and Dr. Bhardwaj reported having no disclosures. Their colleagues disclosed relationships with Bristol Myers Squibb, Sema4, and Related Sciences.
The vaccine, PGV-001, was given to 13 patients with solid tumors or multiple myeloma who had a high risk of recurrence after surgery or autologous stem cell transplant.
At last follow-up, four patients were still alive without evidence of disease and had not received subsequent therapy, four were alive and receiving therapy, three had died, and two were lost to follow-up.
Thomas Marron, MD, PhD , of Mount Sinai in New York presented these results in a poster at the American Association for Cancer Research Annual Meeting 2021: Week 1 ( Abstract LB048 ). Data in the abstract differ from the data presented.
“While cancer immunotherapy has revolutionized the treatment of cancer, we know that the majority of patients fail to achieve significant clinical response,” Dr. Marron said during his presentation. “One reason for this may be due to lack of preexisting primed T-cell response needed for PD-1 blockade to have a significant effect. To address this, personalized neoantigen vaccines may help prime an improved immune response against tumor cells.”
With this in mind, Dr. Marron and colleagues developed PGV-001, a vaccine consisting of patient-specific synthetic neoantigen peptides given to patients in the adjuvant setting.
Creating a personalized vaccine
The researchers synthesized PGV-001 for 15 patients with advanced malignancies. The patients first underwent tumor and germline DNA sequencing as well as HLA typing. Bulk RNA sequencing was performed on patients’ tumors as well.
Then, the researchers used a computational pipeline called OpenVax to identify candidate neoantigens. This pipeline, developed at Mount Sinai, identified and prioritized candidate neoantigens using predicted MHC class I binding affinity and neoantigen abundance.
OpenVax identified an average of 71.5 neoantigens per patient (range, 7-193). The goal was to synthesize a maximum of 10 peptides per patient, but two patients did not have an adequate number of neoantigens.
Vaccine administration
The peptides were administered over the course of 27 weeks along with poly-ICLC and a tetanus helper peptide. Before receiving their vaccine doses, patients with solid tumors had undergone curative-intent surgery, and those with multiple myeloma had undergone autologous stem cell transplant.
“Most experimental personalized cancer vaccines are administered in the metastatic setting, but prior research indicates that immunotherapies tend to be more effective in patients who have less cancer spread,” principal investigator Nina Bhardwaj, MD, PhD , of Mount Sinai, explained in a press release .
“We have, therefore, developed a neoantigen vaccine that is administered after standard-of-care adjuvant therapy, such as surgery in solid tumors and bone marrow transplant in multiple myeloma, when patients have minimal, typically microscopic, residual disease.”
Feasibility, safety, and immunogenicity
PGV-001 was synthesized for 15 patients and administered to 13 of them. Six of the 13 patients had head and neck squamous cell carcinoma, three had multiple myeloma, two had non–small cell lung cancer, one had breast cancer, and one had urothelial carcinoma.
Eleven patients received all 10 intended doses, and two patients received at least 8 doses.
“The vaccine was well tolerated, with only half of patients experiencing mild, grade 1 adverse events,” Dr. Marron said.
Transient injection site reactions occurred in four patients, and grade 1 fever was reported in one patient.
Immune monitoring is ongoing, but an initial analysis in one patient showed “robust responses” in CD4 and CD8 T cells by intracellular cytokine staining for interferon-gamma, tumor necrosis factor–alpha, and interleukin-2 after in vitro expansion in the presence of vaccine antigens, according to the researchers.
Dr. Marron noted that robust T-cell reactivity was seen at the completion of all 10 doses but was not seen after the 6th dose, and this supports the need for a prolonged dosing schedule.
Survival and subsequent therapy
At a mean follow-up of 880 days, four patients had no evidence of disease and had not received subsequent therapy. This includes one patient with stage IIIA non–small cell lung cancer, one with stage IVA HER-2 positive breast cancer, one with stage II urothelial carcinoma, and one with multiple myeloma.
Four patients were alive and receiving subsequent lines of therapy. Two of these patients had significant responses to anti–PD-1 therapy.
Three patients have died, two of whom had documented recurrence of their malignancy. The last two patients were lost to follow-up without documented recurrence.
“Our results demonstrate that the OpenVax pipeline is a viable approach to generate a safe, personalized cancer vaccine, which could potentially be used to treat a range of tumor types,” Dr. Bhardwaj said.
Trials combining neoantigens identified with the OpenVax platform are ongoing in patients with urothelial carcinoma and glioblastoma multiforme, Dr. Marron said.
The current study ( NCT02721043 ) is sponsored by Dr. Bhardwaj. Dr. Marron and Dr. Bhardwaj reported having no disclosures. Their colleagues disclosed relationships with Bristol Myers Squibb, Sema4, and Related Sciences.
The vaccine, PGV-001, was given to 13 patients with solid tumors or multiple myeloma who had a high risk of recurrence after surgery or autologous stem cell transplant.
At last follow-up, four patients were still alive without evidence of disease and had not received subsequent therapy, four were alive and receiving therapy, three had died, and two were lost to follow-up.
Thomas Marron, MD, PhD , of Mount Sinai in New York presented these results in a poster at the American Association for Cancer Research Annual Meeting 2021: Week 1 ( Abstract LB048 ). Data in the abstract differ from the data presented.
“While cancer immunotherapy has revolutionized the treatment of cancer, we know that the majority of patients fail to achieve significant clinical response,” Dr. Marron said during his presentation. “One reason for this may be due to lack of preexisting primed T-cell response needed for PD-1 blockade to have a significant effect. To address this, personalized neoantigen vaccines may help prime an improved immune response against tumor cells.”
With this in mind, Dr. Marron and colleagues developed PGV-001, a vaccine consisting of patient-specific synthetic neoantigen peptides given to patients in the adjuvant setting.
Creating a personalized vaccine
The researchers synthesized PGV-001 for 15 patients with advanced malignancies. The patients first underwent tumor and germline DNA sequencing as well as HLA typing. Bulk RNA sequencing was performed on patients’ tumors as well.
Then, the researchers used a computational pipeline called OpenVax to identify candidate neoantigens. This pipeline, developed at Mount Sinai, identified and prioritized candidate neoantigens using predicted MHC class I binding affinity and neoantigen abundance.
OpenVax identified an average of 71.5 neoantigens per patient (range, 7-193). The goal was to synthesize a maximum of 10 peptides per patient, but two patients did not have an adequate number of neoantigens.
Vaccine administration
The peptides were administered over the course of 27 weeks along with poly-ICLC and a tetanus helper peptide. Before receiving their vaccine doses, patients with solid tumors had undergone curative-intent surgery, and those with multiple myeloma had undergone autologous stem cell transplant.
“Most experimental personalized cancer vaccines are administered in the metastatic setting, but prior research indicates that immunotherapies tend to be more effective in patients who have less cancer spread,” principal investigator Nina Bhardwaj, MD, PhD , of Mount Sinai, explained in a press release .
“We have, therefore, developed a neoantigen vaccine that is administered after standard-of-care adjuvant therapy, such as surgery in solid tumors and bone marrow transplant in multiple myeloma, when patients have minimal, typically microscopic, residual disease.”
Feasibility, safety, and immunogenicity
PGV-001 was synthesized for 15 patients and administered to 13 of them. Six of the 13 patients had head and neck squamous cell carcinoma, three had multiple myeloma, two had non–small cell lung cancer, one had breast cancer, and one had urothelial carcinoma.
Eleven patients received all 10 intended doses, and two patients received at least 8 doses.
“The vaccine was well tolerated, with only half of patients experiencing mild, grade 1 adverse events,” Dr. Marron said.
Transient injection site reactions occurred in four patients, and grade 1 fever was reported in one patient.
Immune monitoring is ongoing, but an initial analysis in one patient showed “robust responses” in CD4 and CD8 T cells by intracellular cytokine staining for interferon-gamma, tumor necrosis factor–alpha, and interleukin-2 after in vitro expansion in the presence of vaccine antigens, according to the researchers.
Dr. Marron noted that robust T-cell reactivity was seen at the completion of all 10 doses but was not seen after the 6th dose, and this supports the need for a prolonged dosing schedule.
Survival and subsequent therapy
At a mean follow-up of 880 days, four patients had no evidence of disease and had not received subsequent therapy. This includes one patient with stage IIIA non–small cell lung cancer, one with stage IVA HER-2 positive breast cancer, one with stage II urothelial carcinoma, and one with multiple myeloma.
Four patients were alive and receiving subsequent lines of therapy. Two of these patients had significant responses to anti–PD-1 therapy.
Three patients have died, two of whom had documented recurrence of their malignancy. The last two patients were lost to follow-up without documented recurrence.
“Our results demonstrate that the OpenVax pipeline is a viable approach to generate a safe, personalized cancer vaccine, which could potentially be used to treat a range of tumor types,” Dr. Bhardwaj said.
Trials combining neoantigens identified with the OpenVax platform are ongoing in patients with urothelial carcinoma and glioblastoma multiforme, Dr. Marron said.
The current study ( NCT02721043 ) is sponsored by Dr. Bhardwaj. Dr. Marron and Dr. Bhardwaj reported having no disclosures. Their colleagues disclosed relationships with Bristol Myers Squibb, Sema4, and Related Sciences.
FROM AACR 2021
Rankings of most common cancers to shift over next 20 years
The next 20 years will see a big shift in cancer type rankings, researchers predict.
At the moment, the most common cancers in the United States are breast, lung, prostate, colorectal, and melanoma.
the study authors predicted. Breast cancer will remain the top cancer to be diagnosed, lung cancer will drop from second to third, and colorectal cancer will remain at fourth.
These predicted rankings of cancer types by their total number of annual cases were published online April 7, 2021, in JAMA Network Open.
The authors also rank cancer type by mortality. Currently, most cancer deaths are caused by lung cancer, followed by colorectal, pancreatic, and breast. By 2040, the most notable change in cancer deaths is that liver and intrahepatic bile duct cancer, currently at sixth, will jump up to third.
Two decades from now, the ranking in terms of cancer deaths will be lung, pancreatic, liver and intrahepatic bile duct, and colorectal.
“Our findings reflect the shifting dynamics of cancer screening and treatment,” lead author Lola Rahib, PhD, a pancreatic cancer scientist at Cancer Commons, the advocacy nonprofit, commented in a press statement.
The new analysis used population-growth projections (based on 2010 U.S. Census data) and current population-based cancer incidence and death rates (from Surveillance, Epidemiology, and End Results 2014-2016) to calculate the changes in incidences and deaths to the year 2040.
The projected, estimated numbers are not ironclad, the researchers acknowledged.
“Our projections assume that the observed rates and trends [from recent years] don’t change over time,” Dr. Rahib said in an interview, but she pointed out that change may indeed happen.
“Any long-term projections should be considered with a grain of salt,” said Kim Miller, MPH, a surveillance research scientist at the American Cancer Society, who was approached for comment.
Dr. Miller explained that “cancer trends can sometimes rapidly change within a few years.” Projections just 2-4 years ahead are “extremely difficult” and those 20 years ahead are even more so, she added in an interview.
“We’re encouraged to see the projected decreases in deaths from lung, colorectal, and breast cancer in the coming years,” said coauthor Lynn Matrisian, PhD, MBA, chief science officer at the Pancreatic Cancer Action Network. “It’s time to shift focus to some of the less commonly diagnosed cancers with the lowest survival rates, like pancreatic and liver cancer.”
Difference in opinion on prostate cancer
The huge fall in the incidence of prostate cancer that the authors predict will come about as a result of changes in prostate-specific antigen (PSA)–screening recommendations over the last 15 years, they suggested.
“The most recent change in 2018 recommends that men aged 55-69 can make their own decisions regarding screening, but previous changes recommended against PSA screening,” said Dr. Rahib.
“These changes in screening guidelines have influenced the number of diagnoses of prostate cancer in recent years and will continue to do so to 2040,” Dr. Rahib commented.
Dr. Miller casts doubt on this prediction.
Using data through 2017, “we have seen that the patterns in prostate cancer incidence are already shifting from the steep declines we saw in the early 2010s,” she said. “I would use caution when interpreting the overall trends for prostate, because this cancer in particular is dramatically affected by changes in recommendations for screening with the PSA test.”
Screening has also influenced colorectal cancer incidence, the authors pointed out, saying that the uptake of colorectal cancer screening is associated with a decrease in the number of colorectal cancers and deaths out to 2040, as a result of effectiveness of screening.
For breast cancer, the authors highlighted the fact that, although the number of breast cancers will continue to increase, the number of breast cancer deaths will decrease. That ongoing trend is most likely attributable to increased screening and advancements in treatment.
The study was supported by the National Institutes of Health, National Cancer Institute, the Cancer Prevention and Research Institute of Texas, Cancer Commons and the Pancreatic Cancer Action Network. The study authors and Dr. Miller disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The next 20 years will see a big shift in cancer type rankings, researchers predict.
At the moment, the most common cancers in the United States are breast, lung, prostate, colorectal, and melanoma.
the study authors predicted. Breast cancer will remain the top cancer to be diagnosed, lung cancer will drop from second to third, and colorectal cancer will remain at fourth.
These predicted rankings of cancer types by their total number of annual cases were published online April 7, 2021, in JAMA Network Open.
The authors also rank cancer type by mortality. Currently, most cancer deaths are caused by lung cancer, followed by colorectal, pancreatic, and breast. By 2040, the most notable change in cancer deaths is that liver and intrahepatic bile duct cancer, currently at sixth, will jump up to third.
Two decades from now, the ranking in terms of cancer deaths will be lung, pancreatic, liver and intrahepatic bile duct, and colorectal.
“Our findings reflect the shifting dynamics of cancer screening and treatment,” lead author Lola Rahib, PhD, a pancreatic cancer scientist at Cancer Commons, the advocacy nonprofit, commented in a press statement.
The new analysis used population-growth projections (based on 2010 U.S. Census data) and current population-based cancer incidence and death rates (from Surveillance, Epidemiology, and End Results 2014-2016) to calculate the changes in incidences and deaths to the year 2040.
The projected, estimated numbers are not ironclad, the researchers acknowledged.
“Our projections assume that the observed rates and trends [from recent years] don’t change over time,” Dr. Rahib said in an interview, but she pointed out that change may indeed happen.
“Any long-term projections should be considered with a grain of salt,” said Kim Miller, MPH, a surveillance research scientist at the American Cancer Society, who was approached for comment.
Dr. Miller explained that “cancer trends can sometimes rapidly change within a few years.” Projections just 2-4 years ahead are “extremely difficult” and those 20 years ahead are even more so, she added in an interview.
“We’re encouraged to see the projected decreases in deaths from lung, colorectal, and breast cancer in the coming years,” said coauthor Lynn Matrisian, PhD, MBA, chief science officer at the Pancreatic Cancer Action Network. “It’s time to shift focus to some of the less commonly diagnosed cancers with the lowest survival rates, like pancreatic and liver cancer.”
Difference in opinion on prostate cancer
The huge fall in the incidence of prostate cancer that the authors predict will come about as a result of changes in prostate-specific antigen (PSA)–screening recommendations over the last 15 years, they suggested.
“The most recent change in 2018 recommends that men aged 55-69 can make their own decisions regarding screening, but previous changes recommended against PSA screening,” said Dr. Rahib.
“These changes in screening guidelines have influenced the number of diagnoses of prostate cancer in recent years and will continue to do so to 2040,” Dr. Rahib commented.
Dr. Miller casts doubt on this prediction.
Using data through 2017, “we have seen that the patterns in prostate cancer incidence are already shifting from the steep declines we saw in the early 2010s,” she said. “I would use caution when interpreting the overall trends for prostate, because this cancer in particular is dramatically affected by changes in recommendations for screening with the PSA test.”
Screening has also influenced colorectal cancer incidence, the authors pointed out, saying that the uptake of colorectal cancer screening is associated with a decrease in the number of colorectal cancers and deaths out to 2040, as a result of effectiveness of screening.
For breast cancer, the authors highlighted the fact that, although the number of breast cancers will continue to increase, the number of breast cancer deaths will decrease. That ongoing trend is most likely attributable to increased screening and advancements in treatment.
The study was supported by the National Institutes of Health, National Cancer Institute, the Cancer Prevention and Research Institute of Texas, Cancer Commons and the Pancreatic Cancer Action Network. The study authors and Dr. Miller disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The next 20 years will see a big shift in cancer type rankings, researchers predict.
At the moment, the most common cancers in the United States are breast, lung, prostate, colorectal, and melanoma.
the study authors predicted. Breast cancer will remain the top cancer to be diagnosed, lung cancer will drop from second to third, and colorectal cancer will remain at fourth.
These predicted rankings of cancer types by their total number of annual cases were published online April 7, 2021, in JAMA Network Open.
The authors also rank cancer type by mortality. Currently, most cancer deaths are caused by lung cancer, followed by colorectal, pancreatic, and breast. By 2040, the most notable change in cancer deaths is that liver and intrahepatic bile duct cancer, currently at sixth, will jump up to third.
Two decades from now, the ranking in terms of cancer deaths will be lung, pancreatic, liver and intrahepatic bile duct, and colorectal.
“Our findings reflect the shifting dynamics of cancer screening and treatment,” lead author Lola Rahib, PhD, a pancreatic cancer scientist at Cancer Commons, the advocacy nonprofit, commented in a press statement.
The new analysis used population-growth projections (based on 2010 U.S. Census data) and current population-based cancer incidence and death rates (from Surveillance, Epidemiology, and End Results 2014-2016) to calculate the changes in incidences and deaths to the year 2040.
The projected, estimated numbers are not ironclad, the researchers acknowledged.
“Our projections assume that the observed rates and trends [from recent years] don’t change over time,” Dr. Rahib said in an interview, but she pointed out that change may indeed happen.
“Any long-term projections should be considered with a grain of salt,” said Kim Miller, MPH, a surveillance research scientist at the American Cancer Society, who was approached for comment.
Dr. Miller explained that “cancer trends can sometimes rapidly change within a few years.” Projections just 2-4 years ahead are “extremely difficult” and those 20 years ahead are even more so, she added in an interview.
“We’re encouraged to see the projected decreases in deaths from lung, colorectal, and breast cancer in the coming years,” said coauthor Lynn Matrisian, PhD, MBA, chief science officer at the Pancreatic Cancer Action Network. “It’s time to shift focus to some of the less commonly diagnosed cancers with the lowest survival rates, like pancreatic and liver cancer.”
Difference in opinion on prostate cancer
The huge fall in the incidence of prostate cancer that the authors predict will come about as a result of changes in prostate-specific antigen (PSA)–screening recommendations over the last 15 years, they suggested.
“The most recent change in 2018 recommends that men aged 55-69 can make their own decisions regarding screening, but previous changes recommended against PSA screening,” said Dr. Rahib.
“These changes in screening guidelines have influenced the number of diagnoses of prostate cancer in recent years and will continue to do so to 2040,” Dr. Rahib commented.
Dr. Miller casts doubt on this prediction.
Using data through 2017, “we have seen that the patterns in prostate cancer incidence are already shifting from the steep declines we saw in the early 2010s,” she said. “I would use caution when interpreting the overall trends for prostate, because this cancer in particular is dramatically affected by changes in recommendations for screening with the PSA test.”
Screening has also influenced colorectal cancer incidence, the authors pointed out, saying that the uptake of colorectal cancer screening is associated with a decrease in the number of colorectal cancers and deaths out to 2040, as a result of effectiveness of screening.
For breast cancer, the authors highlighted the fact that, although the number of breast cancers will continue to increase, the number of breast cancer deaths will decrease. That ongoing trend is most likely attributable to increased screening and advancements in treatment.
The study was supported by the National Institutes of Health, National Cancer Institute, the Cancer Prevention and Research Institute of Texas, Cancer Commons and the Pancreatic Cancer Action Network. The study authors and Dr. Miller disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Steroid-refractory pneumonitis from ICIs: Experience at major centers
Pneumonitis is an uncommon and potentially life-threatening complication of immune checkpoint inhibitor (ICI) therapy. A fraction of patients with ICI-related pneumonitis fail to respond to initial therapy with high-dose systemic steroids.
The recently published experiences at two major cancer centers shed light on the outcomes from treatment and can provide some advice to clinicians for dealing with affected patients.
The Johns Hopkins experience
Because ICI-related pneumonitis typically improves within 48-72 hours of steroid therapy, at Johns Hopkins University, Baltimore, steroid-refractory pneumonitis is defined as pneumonitis that demonstrates no clinical improvement after high-dose corticosteroids for 2-14 days. If the immune toxicity–specialized, multidisciplinary management team implements additional immunosuppressive therapy, that is regarded as confirmatory evidence.
Aanika Balaji, a medical student at Johns Hopkins University, and colleagues retrospectively summarized the clinical course of 12 patients with ICI-related pneumonitis between 2011 and 2020. Clinical improvement with subsequent treatment was evidenced by reduction in either level of care or oxygen requirements.
Three-quarters of the patients were current or former smokers, and the same proportion had lung cancer. Most patients (91.6%) had received chemotherapy, 58.3% had prior chest radiotherapy, and 58.3% had achieved partial response or stable disease with an ICI.
Steroid-refractory ICI-related pneumonitis developed between 40 and 127 days (median, 85 days) after the first dose of ICI therapy. Subsequent immunosuppressive management included IVIg, infliximab, or the combination, in addition to ICU-level supportive care.
Among the seven patients who received IVIg alone, two patients (29%) achieved clinical improvement and hospital discharge. The remainder died.
The two patients treated with infliximab and the three patients treated with sequential IVIg and infliximab died. All deaths were attributed to ICI-related pneumonitis or infectious complications.
Overall, clinically relevant findings were:
- Steroid-refractory ICI-related pneumonitis was seen in 18.5% of patients referred for multidisciplinary care.
- Steroid-refractory ICI-related pneumonitis occurred at a median of 85 days into a patient’s ICI treatment.
- Some patients improved clinically after IVIg therapy, but mortality was high overall.
- Infliximab therapy, alone or in combination with IVIg, was ineffective.
The Memorial Sloan Kettering experience
Jason Beattie, MD, of Memorial Sloan Kettering Cancer Center, New York, and colleagues performed a retrospective study of patients who had pneumonitis after ICI therapy and/or received immune modulator therapy after corticosteroids in the setting of ICI cancer treatment.
Manual record review was performed to exclude cases of pneumonitis from other causes. The period reviewed was roughly contemporaneous with the Johns Hopkins series.
Patients with ICI-related pneumonitis were divided into “steroid refractory” (i.e., no response to high-dose corticosteroids) or “steroid resistant” (i.e., initial response, followed by worsening) categories.
The researchers identified 26 patients with ICI-related pneumonitis, all of whom had advanced malignancy (8 lung cancer, 4 malignant melanoma, 4 renal cell cancer, and 10 “other” cancers).
A majority of patients (85%) were current or former smokers, 73% had received ICI monotherapy, 35% had received prior chest radiation at a median interval of 4.9 months prior to pneumonitis onset, and 27% had preexisting pulmonary disease.
Twelve patients (46%) had steroid-refractory ICI-related pneumonitis, and 14 (54%) had steroid-resistant ICI-related pneumonitis.
The two groups differed in time to pneumonitis onset (a median of 68 days in the refractory group and 182 days in the resistant group) and time to immune modulator therapy after beginning steroids (median 7 days and 2.9 months, respectively). In the steroid-refractory cases, pneumonitis was more severe.
In addition to corticosteroids, most patients received infliximab monotherapy or infliximab with mycophenolate mofetil. In contrast to the Johns Hopkins series, IVIg was not used in the Memorial Sloan Kettering cases.
Outcomes from immune modulators were graded based on clinical evidence (progress notes, oxygen requirements, level of care, radiologic information, etc.) of resolution of pneumonitis on imaging at least 8 weeks after cessation of steroids and immune modulator therapy, durable improvement for at least 8 weeks after immune modulator therapy, transient improvement followed by pneumonitis relapse or inadequate follow-up because of death or hospice referral, or no improvement.
Ten patients (38%) had durable improvement of ICI-related pneumonitis, of whom three (12%) had complete resolution. Two of the patients with complete resolution had steroid-refractory pneumonitis, both of whom had received infliximab followed by mycophenolate mofetil.
Among the seven patients with durable improvement, four remained alive on immune modulators. Time to resolution of pneumonitis was protracted, ranging from 2.3 months to 8.4 months in the steroid-refractory patients.
Durable response was less common with steroid-refractory (25%) than steroid-resistant (50%) disease, with a significant difference in 90-day survival of 25% and 71%, respectively.
Among the 13 (50%) patients with transient improvement in ICI-related pneumonitis, 8 ultimately died, either because of recurrent ICI-related pneumonitis or infection. All three patients with no improvement from immune modulators died.
The 90-day all-cause mortality was 50%, with durable pneumonitis improvement and freedom from severe infectious complications occurring in only about a third of patients.
Lessons for clinicians
The National Comprehensive Cancer Network, the Society for Immunotherapy of Cancer, and the European Society of Medical Oncology have all published guidelines and recommendations for immunosuppression for steroid-refractory adverse events from ICIs.
Unfortunately, there is little experience with steroid-unresponsive ICI-related pneumonitis. The ideal sequence, dose, and duration of additional immune modulator therapy for ICI-related pneumonitis are unclear and may differ from the approaches to other immune-related toxicities.
This is important because, as suggested in an editorial by Margaret Gatti-Mays, MD, and James L. Gulley, MD, PhD, it is likely that ICI-related pneumonitis will be seen more in routine practice than in clinical trial populations. In addition, across all tumor types, ICI-related pneumonitis is the most common cause of ICI-associated death from toxicity.
The retrospective studies from Johns Hopkins and Memorial Sloan Kettering constitute the largest published experience with ICI-related pneumonitis and yield important clinical insights.
Uniform definitions of potentially important patient subgroups (e.g., steroid refractory vs. steroid resistant) are needed. The steroid-refractory and steroid-resistant subgroups have distinctly different clinical features and outcomes. Uniformity in the subgroup definitions would be a useful starting point from both clinical and research perspectives.
Preferred treatment choices need to be tested systematically in multi-institutional studies. Any potential impact of treatment for ICI-related pneumonitis on antitumor immune control should be identified.
Endpoints of interest need to be defined and measured prospectively. All-cause mortality after 90 days is important, but, as the authors of both reviews noted, there are vitally important narratives and differences in functionality that are completely concealed by restricting the focus to mortality.
Potential causal relationships with antecedent exposure to tobacco, radiation, intrathoracic tumor burden, or other factors need to be defined.
Clinicians need predictive biomarkers for ICI-related pneumonitis (e.g., in peripheral blood, pulmonary function testing, or bronchoscopy specimens). At-risk patients may benefit from early intervention.
The limitations of single-institution record reviews in guiding real-world patient management notwithstanding, these reviews illustrate the value of registries and prospective studies to guide the path forward. Taking these next steps will ensure for our patients that the success of immune-targeted therapy against their cancer never becomes a Pyrrhic victory.
The Johns Hopkins investigators and the editorialists reported having no disclosures. The Memorial Sloan Kettering investigators disclosed relationships with Targeted Oncology, Merck, Array BioPharma, Novartis, and many other companies.
Dr. Lyss was a community-based medical oncologist and clinical researcher for more than 35 years before his recent retirement. His clinical and research interests were focused on breast and lung cancers, as well as expanding clinical trial access to medically underserved populations. He is based in St. Louis. He has no conflicts of interest.
Pneumonitis is an uncommon and potentially life-threatening complication of immune checkpoint inhibitor (ICI) therapy. A fraction of patients with ICI-related pneumonitis fail to respond to initial therapy with high-dose systemic steroids.
The recently published experiences at two major cancer centers shed light on the outcomes from treatment and can provide some advice to clinicians for dealing with affected patients.
The Johns Hopkins experience
Because ICI-related pneumonitis typically improves within 48-72 hours of steroid therapy, at Johns Hopkins University, Baltimore, steroid-refractory pneumonitis is defined as pneumonitis that demonstrates no clinical improvement after high-dose corticosteroids for 2-14 days. If the immune toxicity–specialized, multidisciplinary management team implements additional immunosuppressive therapy, that is regarded as confirmatory evidence.
Aanika Balaji, a medical student at Johns Hopkins University, and colleagues retrospectively summarized the clinical course of 12 patients with ICI-related pneumonitis between 2011 and 2020. Clinical improvement with subsequent treatment was evidenced by reduction in either level of care or oxygen requirements.
Three-quarters of the patients were current or former smokers, and the same proportion had lung cancer. Most patients (91.6%) had received chemotherapy, 58.3% had prior chest radiotherapy, and 58.3% had achieved partial response or stable disease with an ICI.
Steroid-refractory ICI-related pneumonitis developed between 40 and 127 days (median, 85 days) after the first dose of ICI therapy. Subsequent immunosuppressive management included IVIg, infliximab, or the combination, in addition to ICU-level supportive care.
Among the seven patients who received IVIg alone, two patients (29%) achieved clinical improvement and hospital discharge. The remainder died.
The two patients treated with infliximab and the three patients treated with sequential IVIg and infliximab died. All deaths were attributed to ICI-related pneumonitis or infectious complications.
Overall, clinically relevant findings were:
- Steroid-refractory ICI-related pneumonitis was seen in 18.5% of patients referred for multidisciplinary care.
- Steroid-refractory ICI-related pneumonitis occurred at a median of 85 days into a patient’s ICI treatment.
- Some patients improved clinically after IVIg therapy, but mortality was high overall.
- Infliximab therapy, alone or in combination with IVIg, was ineffective.
The Memorial Sloan Kettering experience
Jason Beattie, MD, of Memorial Sloan Kettering Cancer Center, New York, and colleagues performed a retrospective study of patients who had pneumonitis after ICI therapy and/or received immune modulator therapy after corticosteroids in the setting of ICI cancer treatment.
Manual record review was performed to exclude cases of pneumonitis from other causes. The period reviewed was roughly contemporaneous with the Johns Hopkins series.
Patients with ICI-related pneumonitis were divided into “steroid refractory” (i.e., no response to high-dose corticosteroids) or “steroid resistant” (i.e., initial response, followed by worsening) categories.
The researchers identified 26 patients with ICI-related pneumonitis, all of whom had advanced malignancy (8 lung cancer, 4 malignant melanoma, 4 renal cell cancer, and 10 “other” cancers).
A majority of patients (85%) were current or former smokers, 73% had received ICI monotherapy, 35% had received prior chest radiation at a median interval of 4.9 months prior to pneumonitis onset, and 27% had preexisting pulmonary disease.
Twelve patients (46%) had steroid-refractory ICI-related pneumonitis, and 14 (54%) had steroid-resistant ICI-related pneumonitis.
The two groups differed in time to pneumonitis onset (a median of 68 days in the refractory group and 182 days in the resistant group) and time to immune modulator therapy after beginning steroids (median 7 days and 2.9 months, respectively). In the steroid-refractory cases, pneumonitis was more severe.
In addition to corticosteroids, most patients received infliximab monotherapy or infliximab with mycophenolate mofetil. In contrast to the Johns Hopkins series, IVIg was not used in the Memorial Sloan Kettering cases.
Outcomes from immune modulators were graded based on clinical evidence (progress notes, oxygen requirements, level of care, radiologic information, etc.) of resolution of pneumonitis on imaging at least 8 weeks after cessation of steroids and immune modulator therapy, durable improvement for at least 8 weeks after immune modulator therapy, transient improvement followed by pneumonitis relapse or inadequate follow-up because of death or hospice referral, or no improvement.
Ten patients (38%) had durable improvement of ICI-related pneumonitis, of whom three (12%) had complete resolution. Two of the patients with complete resolution had steroid-refractory pneumonitis, both of whom had received infliximab followed by mycophenolate mofetil.
Among the seven patients with durable improvement, four remained alive on immune modulators. Time to resolution of pneumonitis was protracted, ranging from 2.3 months to 8.4 months in the steroid-refractory patients.
Durable response was less common with steroid-refractory (25%) than steroid-resistant (50%) disease, with a significant difference in 90-day survival of 25% and 71%, respectively.
Among the 13 (50%) patients with transient improvement in ICI-related pneumonitis, 8 ultimately died, either because of recurrent ICI-related pneumonitis or infection. All three patients with no improvement from immune modulators died.
The 90-day all-cause mortality was 50%, with durable pneumonitis improvement and freedom from severe infectious complications occurring in only about a third of patients.
Lessons for clinicians
The National Comprehensive Cancer Network, the Society for Immunotherapy of Cancer, and the European Society of Medical Oncology have all published guidelines and recommendations for immunosuppression for steroid-refractory adverse events from ICIs.
Unfortunately, there is little experience with steroid-unresponsive ICI-related pneumonitis. The ideal sequence, dose, and duration of additional immune modulator therapy for ICI-related pneumonitis are unclear and may differ from the approaches to other immune-related toxicities.
This is important because, as suggested in an editorial by Margaret Gatti-Mays, MD, and James L. Gulley, MD, PhD, it is likely that ICI-related pneumonitis will be seen more in routine practice than in clinical trial populations. In addition, across all tumor types, ICI-related pneumonitis is the most common cause of ICI-associated death from toxicity.
The retrospective studies from Johns Hopkins and Memorial Sloan Kettering constitute the largest published experience with ICI-related pneumonitis and yield important clinical insights.
Uniform definitions of potentially important patient subgroups (e.g., steroid refractory vs. steroid resistant) are needed. The steroid-refractory and steroid-resistant subgroups have distinctly different clinical features and outcomes. Uniformity in the subgroup definitions would be a useful starting point from both clinical and research perspectives.
Preferred treatment choices need to be tested systematically in multi-institutional studies. Any potential impact of treatment for ICI-related pneumonitis on antitumor immune control should be identified.
Endpoints of interest need to be defined and measured prospectively. All-cause mortality after 90 days is important, but, as the authors of both reviews noted, there are vitally important narratives and differences in functionality that are completely concealed by restricting the focus to mortality.
Potential causal relationships with antecedent exposure to tobacco, radiation, intrathoracic tumor burden, or other factors need to be defined.
Clinicians need predictive biomarkers for ICI-related pneumonitis (e.g., in peripheral blood, pulmonary function testing, or bronchoscopy specimens). At-risk patients may benefit from early intervention.
The limitations of single-institution record reviews in guiding real-world patient management notwithstanding, these reviews illustrate the value of registries and prospective studies to guide the path forward. Taking these next steps will ensure for our patients that the success of immune-targeted therapy against their cancer never becomes a Pyrrhic victory.
The Johns Hopkins investigators and the editorialists reported having no disclosures. The Memorial Sloan Kettering investigators disclosed relationships with Targeted Oncology, Merck, Array BioPharma, Novartis, and many other companies.
Dr. Lyss was a community-based medical oncologist and clinical researcher for more than 35 years before his recent retirement. His clinical and research interests were focused on breast and lung cancers, as well as expanding clinical trial access to medically underserved populations. He is based in St. Louis. He has no conflicts of interest.
Pneumonitis is an uncommon and potentially life-threatening complication of immune checkpoint inhibitor (ICI) therapy. A fraction of patients with ICI-related pneumonitis fail to respond to initial therapy with high-dose systemic steroids.
The recently published experiences at two major cancer centers shed light on the outcomes from treatment and can provide some advice to clinicians for dealing with affected patients.
The Johns Hopkins experience
Because ICI-related pneumonitis typically improves within 48-72 hours of steroid therapy, at Johns Hopkins University, Baltimore, steroid-refractory pneumonitis is defined as pneumonitis that demonstrates no clinical improvement after high-dose corticosteroids for 2-14 days. If the immune toxicity–specialized, multidisciplinary management team implements additional immunosuppressive therapy, that is regarded as confirmatory evidence.
Aanika Balaji, a medical student at Johns Hopkins University, and colleagues retrospectively summarized the clinical course of 12 patients with ICI-related pneumonitis between 2011 and 2020. Clinical improvement with subsequent treatment was evidenced by reduction in either level of care or oxygen requirements.
Three-quarters of the patients were current or former smokers, and the same proportion had lung cancer. Most patients (91.6%) had received chemotherapy, 58.3% had prior chest radiotherapy, and 58.3% had achieved partial response or stable disease with an ICI.
Steroid-refractory ICI-related pneumonitis developed between 40 and 127 days (median, 85 days) after the first dose of ICI therapy. Subsequent immunosuppressive management included IVIg, infliximab, or the combination, in addition to ICU-level supportive care.
Among the seven patients who received IVIg alone, two patients (29%) achieved clinical improvement and hospital discharge. The remainder died.
The two patients treated with infliximab and the three patients treated with sequential IVIg and infliximab died. All deaths were attributed to ICI-related pneumonitis or infectious complications.
Overall, clinically relevant findings were:
- Steroid-refractory ICI-related pneumonitis was seen in 18.5% of patients referred for multidisciplinary care.
- Steroid-refractory ICI-related pneumonitis occurred at a median of 85 days into a patient’s ICI treatment.
- Some patients improved clinically after IVIg therapy, but mortality was high overall.
- Infliximab therapy, alone or in combination with IVIg, was ineffective.
The Memorial Sloan Kettering experience
Jason Beattie, MD, of Memorial Sloan Kettering Cancer Center, New York, and colleagues performed a retrospective study of patients who had pneumonitis after ICI therapy and/or received immune modulator therapy after corticosteroids in the setting of ICI cancer treatment.
Manual record review was performed to exclude cases of pneumonitis from other causes. The period reviewed was roughly contemporaneous with the Johns Hopkins series.
Patients with ICI-related pneumonitis were divided into “steroid refractory” (i.e., no response to high-dose corticosteroids) or “steroid resistant” (i.e., initial response, followed by worsening) categories.
The researchers identified 26 patients with ICI-related pneumonitis, all of whom had advanced malignancy (8 lung cancer, 4 malignant melanoma, 4 renal cell cancer, and 10 “other” cancers).
A majority of patients (85%) were current or former smokers, 73% had received ICI monotherapy, 35% had received prior chest radiation at a median interval of 4.9 months prior to pneumonitis onset, and 27% had preexisting pulmonary disease.
Twelve patients (46%) had steroid-refractory ICI-related pneumonitis, and 14 (54%) had steroid-resistant ICI-related pneumonitis.
The two groups differed in time to pneumonitis onset (a median of 68 days in the refractory group and 182 days in the resistant group) and time to immune modulator therapy after beginning steroids (median 7 days and 2.9 months, respectively). In the steroid-refractory cases, pneumonitis was more severe.
In addition to corticosteroids, most patients received infliximab monotherapy or infliximab with mycophenolate mofetil. In contrast to the Johns Hopkins series, IVIg was not used in the Memorial Sloan Kettering cases.
Outcomes from immune modulators were graded based on clinical evidence (progress notes, oxygen requirements, level of care, radiologic information, etc.) of resolution of pneumonitis on imaging at least 8 weeks after cessation of steroids and immune modulator therapy, durable improvement for at least 8 weeks after immune modulator therapy, transient improvement followed by pneumonitis relapse or inadequate follow-up because of death or hospice referral, or no improvement.
Ten patients (38%) had durable improvement of ICI-related pneumonitis, of whom three (12%) had complete resolution. Two of the patients with complete resolution had steroid-refractory pneumonitis, both of whom had received infliximab followed by mycophenolate mofetil.
Among the seven patients with durable improvement, four remained alive on immune modulators. Time to resolution of pneumonitis was protracted, ranging from 2.3 months to 8.4 months in the steroid-refractory patients.
Durable response was less common with steroid-refractory (25%) than steroid-resistant (50%) disease, with a significant difference in 90-day survival of 25% and 71%, respectively.
Among the 13 (50%) patients with transient improvement in ICI-related pneumonitis, 8 ultimately died, either because of recurrent ICI-related pneumonitis or infection. All three patients with no improvement from immune modulators died.
The 90-day all-cause mortality was 50%, with durable pneumonitis improvement and freedom from severe infectious complications occurring in only about a third of patients.
Lessons for clinicians
The National Comprehensive Cancer Network, the Society for Immunotherapy of Cancer, and the European Society of Medical Oncology have all published guidelines and recommendations for immunosuppression for steroid-refractory adverse events from ICIs.
Unfortunately, there is little experience with steroid-unresponsive ICI-related pneumonitis. The ideal sequence, dose, and duration of additional immune modulator therapy for ICI-related pneumonitis are unclear and may differ from the approaches to other immune-related toxicities.
This is important because, as suggested in an editorial by Margaret Gatti-Mays, MD, and James L. Gulley, MD, PhD, it is likely that ICI-related pneumonitis will be seen more in routine practice than in clinical trial populations. In addition, across all tumor types, ICI-related pneumonitis is the most common cause of ICI-associated death from toxicity.
The retrospective studies from Johns Hopkins and Memorial Sloan Kettering constitute the largest published experience with ICI-related pneumonitis and yield important clinical insights.
Uniform definitions of potentially important patient subgroups (e.g., steroid refractory vs. steroid resistant) are needed. The steroid-refractory and steroid-resistant subgroups have distinctly different clinical features and outcomes. Uniformity in the subgroup definitions would be a useful starting point from both clinical and research perspectives.
Preferred treatment choices need to be tested systematically in multi-institutional studies. Any potential impact of treatment for ICI-related pneumonitis on antitumor immune control should be identified.
Endpoints of interest need to be defined and measured prospectively. All-cause mortality after 90 days is important, but, as the authors of both reviews noted, there are vitally important narratives and differences in functionality that are completely concealed by restricting the focus to mortality.
Potential causal relationships with antecedent exposure to tobacco, radiation, intrathoracic tumor burden, or other factors need to be defined.
Clinicians need predictive biomarkers for ICI-related pneumonitis (e.g., in peripheral blood, pulmonary function testing, or bronchoscopy specimens). At-risk patients may benefit from early intervention.
The limitations of single-institution record reviews in guiding real-world patient management notwithstanding, these reviews illustrate the value of registries and prospective studies to guide the path forward. Taking these next steps will ensure for our patients that the success of immune-targeted therapy against their cancer never becomes a Pyrrhic victory.
The Johns Hopkins investigators and the editorialists reported having no disclosures. The Memorial Sloan Kettering investigators disclosed relationships with Targeted Oncology, Merck, Array BioPharma, Novartis, and many other companies.
Dr. Lyss was a community-based medical oncologist and clinical researcher for more than 35 years before his recent retirement. His clinical and research interests were focused on breast and lung cancers, as well as expanding clinical trial access to medically underserved populations. He is based in St. Louis. He has no conflicts of interest.
Cancer screening stopped by pandemic: Repercussions to come?
Last year, cancer screening programs around the world ground to a halt as SARS-CoV-2 infection rates surged globally. The effect of this slowdown is now becoming clear.
Thousands of cancer diagnoses are “missing,” and oncologists worry that this will lead to more advanced cancers and higher mortality for years to come.
“I feel like this is an earthquake that’s rocked our health care system. My guess is that you’ll probably still see repercussions of this over the next couple of years at least,” said Sharon Chang, MD, an attending surgical oncologist in the Permanente Medical Group, Fremont, Calif.
She was senior author of a study that analyzed the effects of the slowdown in mammography screening as a result of California’s “shelter-in-place” order on March 17, 2020. In the 2 months that followed, there were 64% fewer breast cancer diagnoses at 21 Kaiser Permanente medical centers, compared with the same period in 2019 (250 vs. 703).
In effect, approximately 450 breast cancer patients had “disappeared,” said coauthor Annie Tang, MD, a research fellow at the University of California, San Francisco, East Bay surgery program.
“What surprised me most from our data was the sheer number of breast cancer patients that were missing,” Dr. Tang said in an interview.
A similar picture has emerged elsewhere.
In Boston, an estimated 1,438 cancerous and precancerous lesions “went missing” during the first 3 months of pandemic shutdown, according to a study from the Massachusetts General Brigham health care system.
In this study, the investigators assessed screening rates for five cancers – breast cancer (mammography), prostate cancer (prostate-specific antigen testing), colorectal cancer (colonoscopy), cervical cancer (Papanicolaou tests), and lung cancer (low-dose CT).
Screening rates during the first peak of the pandemic (March 2 to June 2, 2020) were compared with those during the preceding and following 3 months and during the same 3 months in 2019.
The results showed a pronounced drop in screening rates during the peak pandemic period, compared with the three control periods. Decreases occurred for all screening tests and ranged from –60% to –82%.
There were also significant decreases in cancer diagnoses resulting from the decreases in screening tests, ranging from –19% to –78%.
“Quantifying the actual problem made us realize how much work needs to be done to get us back to prepandemic numbers,” said senior author Quoc-Dien Trinh, MD, FACS, codirector of the Dana Farber/Brigham and Women’s prostate cancer program.
In the Canadian province of Alberta, a similar decrease in cancer diagnoses occurred during the early days of the pandemic.
By the end of 2020, Alberta was “missing” approximately 2,000 cases of invasive cancers and 1,000 cases of noninvasive cancers, Doug Stewart, MD, senior medical director at the Cancer Strategic Clinical Network (SCN) of Alberta Health Services, told this news organization.
Dr. Stewart is able to track cancer diagnoses in Alberta almost in real time through a mandatory cancer registry. Within a month of shutdown, there was a 30% decrease in diagnoses of invasive cancers and a 50% decrease “in the kind of preinvasive cancers that, for the most part, are picked up by screening programs,” said Dr. Stewart.
After the health care system opened up again in the summer, Stewart said, noninvasive cancer diagnoses continued to be 20% lower than expected. There was a 10% shortfall in invasive cancer diagnoses.
The number of diagnoses had returned to normal by December 2020. However, Dr. Stewart is worried that this fact conceals a terrible truth.
The worry is over the backlog. Although the number of diagnoses is now similar to what it was before the pandemic, “people are presenting later, and maybe the cancer is more advanced,” he speculated.
His team at Alberta Health Services is assessing whether the cancers that are being diagnosed now are more advanced. Initial results are anticipated by late April 2021.
In the United Kingdom, there was a similar halt in cancer screening as a result of the country’s lockdown. Researchers now predict an uptick in cancer diagnoses.
Ajay Aggarwal, MD, PhD, consultant clinical oncologist and associate professor at the London School of Hygiene and Tropical Medicine, and colleagues have estimated that at least 3,500 deaths from breast, colorectal, esophageal, and lung cancer will occur during the next 5 years in England that could have been avoided had it not been for the lockdown measures necessitated by the pandemic.
Speaking to this news organization, Dr. Aggarwal warned that these numbers, which are from a modeling study published in August 2020, are “extremely conservative,” because the investigators considered diagnostic delays over only a 3-month period, the analysis involved only four cancers, and it did not reflect deferral of cancer treatment.
“It felt like it was the tip of the iceberg,” Dr. Aggarwal said. He warns that more recent data suggest that “diagnostic delays are probably worse than we predicted.”
He suspects that there is more at play than screening cancellations.
In another study conducted in the United Kingdom, data show “a falling edge of referrals” from primary care to cancer centers early in the pandemic. In that study, investigators analyzed real-time weekly hospital data from eight large British hospitals and found that urgent cancer referrals fell 70% at their lowest point.
“It really surprised me that the urgent referrals dropped so drastically,” said lead author Alvina Lai, PhD, a lecturer in health data analytics at University College London.
She attributed this in part to patients’ adherence to lockdown rules. “Patients are trying to follow government guidelines to stay home and not go to [general practitioners] unless necessary,” Dr. Lai explained in an interview.
Canada, like the United Kingdom, has a publicly funded health care system. Dr. Stewart came to a similar conclusion. “Some patients who have been diagnosed with cancer ... have told me it took them an extra couple of months to even contact the family doc, because they ... didn’t want to bother the family doctor with something that wasn’t COVID, this kind of guilt. They want to do something good for society. You know, most people are just really nice people, and they don’t want to bother the health care system if they don’t have COVID,” Dr. Stewart said.
Shelley Fuld Nasso, CEO of the National Coalition for Cancer Survivorship, a nonprofit organization based in Silver Spring, Md., agreed that screening shutdowns are not the only danger. “While we agree that screening is really important, we also want to make sure patients are following up with their physicians about symptoms that they have,” she said.
“Some of the speculation or concern about increased mortality for cancer is related to screening, but some of it is related to delayed diagnosis because of not following up on symptoms. ... What concerns me is not everyone has that ability or willingness to advocate for themselves,” she said.
Speaking at a press briefing held by the American Society for Radiation Oncology on March 30, Dr. Nasso related a case involving a patient who experienced severe arm pain. In a teleconsultation with her primary care physician, her condition was diagnosed as arthritis. She was subsequently diagnosed in the ED as having multiple myeloma.
Patients who “feel fine” may postpone their checkups to avoid going to the hospital and risking exposure to COVID-19.
“Some patients are still hesitant about returning for their mammograms or coming in if they feel a breast lump,” Dr. Tang said. “That fear of COVID-19 is still out there, and we don’t know how long patients are going to delay.”
In London, Dr. Aggarwal saw a similar response to the pandemic. “People were overestimating quite significantly what their risk of death was from acquiring COVID-19, and I think that balance was never [redressed] explicitly,” he said.
Public health initiatives to rebalance the messaging are now underway.
Public Health England and National Health Service England launched their Help Us Help You campaign in October 2020. The public information campaign urges people to speak to their doctors if they were “worried about a symptom that could be cancer.”
In Canada, the provincial government in Alberta has launched a public awareness campaign that conveys the message, “cancer has not gone away.”
“Cancer is still the No. 1 cause of potential life-years lost, despite COVID,” Dr. Stewart said. “We need to do what we can to make sure there’s no slippage in survival rates.”
Dr. Tang, Dr. Chang, Dr. Lai, Dr. Stewart, and Dr. Aggarwal have disclosed no relevant financial relationship. Dr. Trinh has received personal fees from Astellas, Bayer, and Janssen and grants from Intuitive Surgical.
A version of this article first appeared on Medscape.com.
Last year, cancer screening programs around the world ground to a halt as SARS-CoV-2 infection rates surged globally. The effect of this slowdown is now becoming clear.
Thousands of cancer diagnoses are “missing,” and oncologists worry that this will lead to more advanced cancers and higher mortality for years to come.
“I feel like this is an earthquake that’s rocked our health care system. My guess is that you’ll probably still see repercussions of this over the next couple of years at least,” said Sharon Chang, MD, an attending surgical oncologist in the Permanente Medical Group, Fremont, Calif.
She was senior author of a study that analyzed the effects of the slowdown in mammography screening as a result of California’s “shelter-in-place” order on March 17, 2020. In the 2 months that followed, there were 64% fewer breast cancer diagnoses at 21 Kaiser Permanente medical centers, compared with the same period in 2019 (250 vs. 703).
In effect, approximately 450 breast cancer patients had “disappeared,” said coauthor Annie Tang, MD, a research fellow at the University of California, San Francisco, East Bay surgery program.
“What surprised me most from our data was the sheer number of breast cancer patients that were missing,” Dr. Tang said in an interview.
A similar picture has emerged elsewhere.
In Boston, an estimated 1,438 cancerous and precancerous lesions “went missing” during the first 3 months of pandemic shutdown, according to a study from the Massachusetts General Brigham health care system.
In this study, the investigators assessed screening rates for five cancers – breast cancer (mammography), prostate cancer (prostate-specific antigen testing), colorectal cancer (colonoscopy), cervical cancer (Papanicolaou tests), and lung cancer (low-dose CT).
Screening rates during the first peak of the pandemic (March 2 to June 2, 2020) were compared with those during the preceding and following 3 months and during the same 3 months in 2019.
The results showed a pronounced drop in screening rates during the peak pandemic period, compared with the three control periods. Decreases occurred for all screening tests and ranged from –60% to –82%.
There were also significant decreases in cancer diagnoses resulting from the decreases in screening tests, ranging from –19% to –78%.
“Quantifying the actual problem made us realize how much work needs to be done to get us back to prepandemic numbers,” said senior author Quoc-Dien Trinh, MD, FACS, codirector of the Dana Farber/Brigham and Women’s prostate cancer program.
In the Canadian province of Alberta, a similar decrease in cancer diagnoses occurred during the early days of the pandemic.
By the end of 2020, Alberta was “missing” approximately 2,000 cases of invasive cancers and 1,000 cases of noninvasive cancers, Doug Stewart, MD, senior medical director at the Cancer Strategic Clinical Network (SCN) of Alberta Health Services, told this news organization.
Dr. Stewart is able to track cancer diagnoses in Alberta almost in real time through a mandatory cancer registry. Within a month of shutdown, there was a 30% decrease in diagnoses of invasive cancers and a 50% decrease “in the kind of preinvasive cancers that, for the most part, are picked up by screening programs,” said Dr. Stewart.
After the health care system opened up again in the summer, Stewart said, noninvasive cancer diagnoses continued to be 20% lower than expected. There was a 10% shortfall in invasive cancer diagnoses.
The number of diagnoses had returned to normal by December 2020. However, Dr. Stewart is worried that this fact conceals a terrible truth.
The worry is over the backlog. Although the number of diagnoses is now similar to what it was before the pandemic, “people are presenting later, and maybe the cancer is more advanced,” he speculated.
His team at Alberta Health Services is assessing whether the cancers that are being diagnosed now are more advanced. Initial results are anticipated by late April 2021.
In the United Kingdom, there was a similar halt in cancer screening as a result of the country’s lockdown. Researchers now predict an uptick in cancer diagnoses.
Ajay Aggarwal, MD, PhD, consultant clinical oncologist and associate professor at the London School of Hygiene and Tropical Medicine, and colleagues have estimated that at least 3,500 deaths from breast, colorectal, esophageal, and lung cancer will occur during the next 5 years in England that could have been avoided had it not been for the lockdown measures necessitated by the pandemic.
Speaking to this news organization, Dr. Aggarwal warned that these numbers, which are from a modeling study published in August 2020, are “extremely conservative,” because the investigators considered diagnostic delays over only a 3-month period, the analysis involved only four cancers, and it did not reflect deferral of cancer treatment.
“It felt like it was the tip of the iceberg,” Dr. Aggarwal said. He warns that more recent data suggest that “diagnostic delays are probably worse than we predicted.”
He suspects that there is more at play than screening cancellations.
In another study conducted in the United Kingdom, data show “a falling edge of referrals” from primary care to cancer centers early in the pandemic. In that study, investigators analyzed real-time weekly hospital data from eight large British hospitals and found that urgent cancer referrals fell 70% at their lowest point.
“It really surprised me that the urgent referrals dropped so drastically,” said lead author Alvina Lai, PhD, a lecturer in health data analytics at University College London.
She attributed this in part to patients’ adherence to lockdown rules. “Patients are trying to follow government guidelines to stay home and not go to [general practitioners] unless necessary,” Dr. Lai explained in an interview.
Canada, like the United Kingdom, has a publicly funded health care system. Dr. Stewart came to a similar conclusion. “Some patients who have been diagnosed with cancer ... have told me it took them an extra couple of months to even contact the family doc, because they ... didn’t want to bother the family doctor with something that wasn’t COVID, this kind of guilt. They want to do something good for society. You know, most people are just really nice people, and they don’t want to bother the health care system if they don’t have COVID,” Dr. Stewart said.
Shelley Fuld Nasso, CEO of the National Coalition for Cancer Survivorship, a nonprofit organization based in Silver Spring, Md., agreed that screening shutdowns are not the only danger. “While we agree that screening is really important, we also want to make sure patients are following up with their physicians about symptoms that they have,” she said.
“Some of the speculation or concern about increased mortality for cancer is related to screening, but some of it is related to delayed diagnosis because of not following up on symptoms. ... What concerns me is not everyone has that ability or willingness to advocate for themselves,” she said.
Speaking at a press briefing held by the American Society for Radiation Oncology on March 30, Dr. Nasso related a case involving a patient who experienced severe arm pain. In a teleconsultation with her primary care physician, her condition was diagnosed as arthritis. She was subsequently diagnosed in the ED as having multiple myeloma.
Patients who “feel fine” may postpone their checkups to avoid going to the hospital and risking exposure to COVID-19.
“Some patients are still hesitant about returning for their mammograms or coming in if they feel a breast lump,” Dr. Tang said. “That fear of COVID-19 is still out there, and we don’t know how long patients are going to delay.”
In London, Dr. Aggarwal saw a similar response to the pandemic. “People were overestimating quite significantly what their risk of death was from acquiring COVID-19, and I think that balance was never [redressed] explicitly,” he said.
Public health initiatives to rebalance the messaging are now underway.
Public Health England and National Health Service England launched their Help Us Help You campaign in October 2020. The public information campaign urges people to speak to their doctors if they were “worried about a symptom that could be cancer.”
In Canada, the provincial government in Alberta has launched a public awareness campaign that conveys the message, “cancer has not gone away.”
“Cancer is still the No. 1 cause of potential life-years lost, despite COVID,” Dr. Stewart said. “We need to do what we can to make sure there’s no slippage in survival rates.”
Dr. Tang, Dr. Chang, Dr. Lai, Dr. Stewart, and Dr. Aggarwal have disclosed no relevant financial relationship. Dr. Trinh has received personal fees from Astellas, Bayer, and Janssen and grants from Intuitive Surgical.
A version of this article first appeared on Medscape.com.
Last year, cancer screening programs around the world ground to a halt as SARS-CoV-2 infection rates surged globally. The effect of this slowdown is now becoming clear.
Thousands of cancer diagnoses are “missing,” and oncologists worry that this will lead to more advanced cancers and higher mortality for years to come.
“I feel like this is an earthquake that’s rocked our health care system. My guess is that you’ll probably still see repercussions of this over the next couple of years at least,” said Sharon Chang, MD, an attending surgical oncologist in the Permanente Medical Group, Fremont, Calif.
She was senior author of a study that analyzed the effects of the slowdown in mammography screening as a result of California’s “shelter-in-place” order on March 17, 2020. In the 2 months that followed, there were 64% fewer breast cancer diagnoses at 21 Kaiser Permanente medical centers, compared with the same period in 2019 (250 vs. 703).
In effect, approximately 450 breast cancer patients had “disappeared,” said coauthor Annie Tang, MD, a research fellow at the University of California, San Francisco, East Bay surgery program.
“What surprised me most from our data was the sheer number of breast cancer patients that were missing,” Dr. Tang said in an interview.
A similar picture has emerged elsewhere.
In Boston, an estimated 1,438 cancerous and precancerous lesions “went missing” during the first 3 months of pandemic shutdown, according to a study from the Massachusetts General Brigham health care system.
In this study, the investigators assessed screening rates for five cancers – breast cancer (mammography), prostate cancer (prostate-specific antigen testing), colorectal cancer (colonoscopy), cervical cancer (Papanicolaou tests), and lung cancer (low-dose CT).
Screening rates during the first peak of the pandemic (March 2 to June 2, 2020) were compared with those during the preceding and following 3 months and during the same 3 months in 2019.
The results showed a pronounced drop in screening rates during the peak pandemic period, compared with the three control periods. Decreases occurred for all screening tests and ranged from –60% to –82%.
There were also significant decreases in cancer diagnoses resulting from the decreases in screening tests, ranging from –19% to –78%.
“Quantifying the actual problem made us realize how much work needs to be done to get us back to prepandemic numbers,” said senior author Quoc-Dien Trinh, MD, FACS, codirector of the Dana Farber/Brigham and Women’s prostate cancer program.
In the Canadian province of Alberta, a similar decrease in cancer diagnoses occurred during the early days of the pandemic.
By the end of 2020, Alberta was “missing” approximately 2,000 cases of invasive cancers and 1,000 cases of noninvasive cancers, Doug Stewart, MD, senior medical director at the Cancer Strategic Clinical Network (SCN) of Alberta Health Services, told this news organization.
Dr. Stewart is able to track cancer diagnoses in Alberta almost in real time through a mandatory cancer registry. Within a month of shutdown, there was a 30% decrease in diagnoses of invasive cancers and a 50% decrease “in the kind of preinvasive cancers that, for the most part, are picked up by screening programs,” said Dr. Stewart.
After the health care system opened up again in the summer, Stewart said, noninvasive cancer diagnoses continued to be 20% lower than expected. There was a 10% shortfall in invasive cancer diagnoses.
The number of diagnoses had returned to normal by December 2020. However, Dr. Stewart is worried that this fact conceals a terrible truth.
The worry is over the backlog. Although the number of diagnoses is now similar to what it was before the pandemic, “people are presenting later, and maybe the cancer is more advanced,” he speculated.
His team at Alberta Health Services is assessing whether the cancers that are being diagnosed now are more advanced. Initial results are anticipated by late April 2021.
In the United Kingdom, there was a similar halt in cancer screening as a result of the country’s lockdown. Researchers now predict an uptick in cancer diagnoses.
Ajay Aggarwal, MD, PhD, consultant clinical oncologist and associate professor at the London School of Hygiene and Tropical Medicine, and colleagues have estimated that at least 3,500 deaths from breast, colorectal, esophageal, and lung cancer will occur during the next 5 years in England that could have been avoided had it not been for the lockdown measures necessitated by the pandemic.
Speaking to this news organization, Dr. Aggarwal warned that these numbers, which are from a modeling study published in August 2020, are “extremely conservative,” because the investigators considered diagnostic delays over only a 3-month period, the analysis involved only four cancers, and it did not reflect deferral of cancer treatment.
“It felt like it was the tip of the iceberg,” Dr. Aggarwal said. He warns that more recent data suggest that “diagnostic delays are probably worse than we predicted.”
He suspects that there is more at play than screening cancellations.
In another study conducted in the United Kingdom, data show “a falling edge of referrals” from primary care to cancer centers early in the pandemic. In that study, investigators analyzed real-time weekly hospital data from eight large British hospitals and found that urgent cancer referrals fell 70% at their lowest point.
“It really surprised me that the urgent referrals dropped so drastically,” said lead author Alvina Lai, PhD, a lecturer in health data analytics at University College London.
She attributed this in part to patients’ adherence to lockdown rules. “Patients are trying to follow government guidelines to stay home and not go to [general practitioners] unless necessary,” Dr. Lai explained in an interview.
Canada, like the United Kingdom, has a publicly funded health care system. Dr. Stewart came to a similar conclusion. “Some patients who have been diagnosed with cancer ... have told me it took them an extra couple of months to even contact the family doc, because they ... didn’t want to bother the family doctor with something that wasn’t COVID, this kind of guilt. They want to do something good for society. You know, most people are just really nice people, and they don’t want to bother the health care system if they don’t have COVID,” Dr. Stewart said.
Shelley Fuld Nasso, CEO of the National Coalition for Cancer Survivorship, a nonprofit organization based in Silver Spring, Md., agreed that screening shutdowns are not the only danger. “While we agree that screening is really important, we also want to make sure patients are following up with their physicians about symptoms that they have,” she said.
“Some of the speculation or concern about increased mortality for cancer is related to screening, but some of it is related to delayed diagnosis because of not following up on symptoms. ... What concerns me is not everyone has that ability or willingness to advocate for themselves,” she said.
Speaking at a press briefing held by the American Society for Radiation Oncology on March 30, Dr. Nasso related a case involving a patient who experienced severe arm pain. In a teleconsultation with her primary care physician, her condition was diagnosed as arthritis. She was subsequently diagnosed in the ED as having multiple myeloma.
Patients who “feel fine” may postpone their checkups to avoid going to the hospital and risking exposure to COVID-19.
“Some patients are still hesitant about returning for their mammograms or coming in if they feel a breast lump,” Dr. Tang said. “That fear of COVID-19 is still out there, and we don’t know how long patients are going to delay.”
In London, Dr. Aggarwal saw a similar response to the pandemic. “People were overestimating quite significantly what their risk of death was from acquiring COVID-19, and I think that balance was never [redressed] explicitly,” he said.
Public health initiatives to rebalance the messaging are now underway.
Public Health England and National Health Service England launched their Help Us Help You campaign in October 2020. The public information campaign urges people to speak to their doctors if they were “worried about a symptom that could be cancer.”
In Canada, the provincial government in Alberta has launched a public awareness campaign that conveys the message, “cancer has not gone away.”
“Cancer is still the No. 1 cause of potential life-years lost, despite COVID,” Dr. Stewart said. “We need to do what we can to make sure there’s no slippage in survival rates.”
Dr. Tang, Dr. Chang, Dr. Lai, Dr. Stewart, and Dr. Aggarwal have disclosed no relevant financial relationship. Dr. Trinh has received personal fees from Astellas, Bayer, and Janssen and grants from Intuitive Surgical.
A version of this article first appeared on Medscape.com.
Treating metastatic TNBC: Where are we now?
Treating triple-negative breast cancer (TNBC), one of the more lethal breast cancer subtypes, remains a challenge. By definition, TNBC lacks the three telltale molecular signatures known to spur tumor growth: estrogen receptor (ER), progesterone receptor (PR), and human epidermal growth factor receptor 2 (HER2). A growing amount of literature shows that these frequently aggressive tumors harbor a rich array of molecular characteristics but no clear oncogenic driver.
“TNBC is incredibly heterogeneous, which makes it challenging to treat,” said Rita Nanda, MD, director of the breast oncology program and associate professor of medicine at the University of Chicago. “We have subsets of TNBC that don’t respond to currently available therapies and, as of yet, have no identifiable therapeutic targets.”
Overall, about 40% of patients with TNBC show a pathologic complete response after first-line neoadjuvant chemotherapy – typically anthracycline and taxane-based agents. But for 50% of patients, chemotherapy leaves behind substantial residual cancer tissue. These patients subsequently face a 40%-80% risk for recurrence and progression to advanced disease.
When triple-negative disease metastasizes, survival rates plummet. The most recent data from the National Cancer Institute, which tracked patients by stage of diagnosis between 2010 and 2016, showed steep declines in 5-year survival as TNBC progressed from local (91.2%) to regional (65%) to advanced-stage disease (11.5%).
Experts have started to make headway identifying and targeting different molecular features of advanced TNBC. These approaches often focus on three key areas: targeting cell surface proteins or oncogenes, stimulating an anticancer immune response, or inhibiting an overactive signaling pathway.
“For a patient with metastatic breast cancer, finding a molecular target or an oncogenic driver is essential,” said Kelly McCann, MD, PhD, a hematologist/oncologist in the department of medicine at the University of California, Los Angeles. “Because TNBC encompasses many different molecular subsets of breast cancer, the development of effective new therapeutics is going to depend on subdividing TNBC into categories with more clear targets.”
A targeted strategy
The Food and Drug Administration’s approval of sacituzumab govitecan, the first antibody-drug conjugate to treat metastatic TNBC, marked an important addition to the TNBC drug armamentarium. “Sacituzumab govitecan is one of the most exciting drugs available for the treatment of metastatic disease,” Dr. Nanda said.
Sacituzumab govitecan, approved as third-line therapy for metastatic TNBC, works by targeting the cell surface protein TROP2, expressed in about 88% of TNBC tumors but rarely in healthy cells.
In the phase 1/2 ASCENT trial, the median progression-free survival was 5.5 months and overall survival was 13.0 months in 108 patients with metastatic TNBC who had received at least two therapies prior to sacituzumab govitecan.
A subsequent phase 3 trial showed progression-free survival of 5.6 months with sacituzumab govitecan and 1.7 months with physician’s choice of chemotherapy. The median overall survival was 12.1 months and 6.7 months, respectively.
But, according to the analysis, TROP2 expression did not necessarily predict who would benefit from sacituzumab govitecan. A biomarker study revealed that although patients with moderate to high TROP2 expression exhibited the strongest treatment response, those with low TROP2 expression also survived longer when given sacituzumab govitecan, compared with chemotherapy alone.
In other words, “patients did better on sacituzumab govitecan regardless of TROP2 expression, which suggests we do not have a good biomarker for identifying who will benefit,” Dr. Nanda said.
Two other investigational antibody-drug conjugates, trastuzumab deruxtecan and ladiratuzumab vedotin, show promise in the metastatic space as well. For instance, the recent phase 2 trial evaluating trastuzumab deruxtecan in patients with HER2-positive breast cancer reported treatment response in 44% of patients with HER2-low tumors.
Given that about 36.6% of TNBC tumors exhibit low levels of HER2 expression, “trastuzumab deruxtecan represents potential in treating HER2-low TNBC,” said Yuan Yuan, MD, PhD, medical oncologist at City of Hope, a comprehensive cancer center in Los Angeles County.
Early results from a phase 1b study showed that trastuzumab deruxtecan produced a response rate of 37% in patients with HER2-low breast cancer.
Investigators are now recruiting for an open-label phase 3 trial to determine whether trastuzumab deruxtecan extends survival in patients with HER2-low metastatic breast cancers.
Immunotherapy advances
Immune checkpoint inhibitors represent another promising treatment avenue for metastatic TNBC. Pembrolizumab and atezolizumab, recently approved by the FDA, show moderate progression-free and overall survival benefits in patients with metastatic TNBC expressing PD-L1. Estimates of PD-L1 immune cells present in TNBC tumors vary widely, from about 20% to 65%.
Yet, data on which patients will benefit are not so clear-cut. “These drugs give us more choices and represent the fast-evolving therapeutic landscape in TNBC, but they also leave a lot of unanswered questions about PD-L1 as a biomarker,” Dr. Yuan said.
Take two recent phase 3 trials evaluating atezolizumab: IMpassion130 and IMpassion131. In IMpassion130, patients with PDL1–positive tumors exhibited significantly longer median overall survival on atezolizumab plus nab-paclitaxel (25.0 months) compared with nab-paclitaxel alone (15.5 months). As with the trend observed in the TROP2 data for sacituzumab govitecan, all patients survived longer on atezolizumab plus nab-paclitaxel regardless of PD-L1 status: 21.3 months vs. 17.6 months with nab-paclitaxel alone.
However, in IMpassion131, neither progression-free survival nor overall survival significantly improved in the PD-L1–positive group receiving atezolizumab plus paclitaxel compared with paclitaxel alone: Progression-free survival was 5.7 months vs. 6 months, respectively, and overall survival was 28.3 months vs. 22.1 months.
“It is unclear why this study failed to demonstrate a significant improvement in progression-free survival with the addition of atezolizumab to paclitaxel,” Dr. Nanda said. “Perhaps the negative finding has to do with how the trial was conducted, or perhaps the PD-L1 assay used is an unreliable biomarker of immunotherapy benefit.”
Continued efforts to understand TNBC
Given the diversity of metastatic TNBC and the absence of clear molecular targets, researchers are exploring a host of therapeutic strategies in addition to antibody-drug conjugates and immunotherapies.
On the oncogene front, researchers are investigating common mutations in TNBC. About 11% of TNBC tumors, for instance, carry germline mutations in BRCA1 and BRCA2. These tumors may be more likely to respond to platinum agents and PARP inhibitors, such as FDA-approved olaparib. In a phase 3 trial, patients with metastatic HER2-negative breast cancer and a germline BRCA mutation who received olaparib exhibited a 2.8-month longer median progression-free survival and a 42% reduced risk for disease progression or death compared with those on standard chemotherapy.
When considering signaling pathways, the PI3K/AKT/mTOR pathway has been the target of numerous clinical trials. Dysregulation of signaling through the PI3K and AKT signaling pathway occurs in 25%-30% of patients with advanced TNBC, and AKT inhibitors have been shown to extend survival in these patients. Data show, for instance, that adding capivasertib to first-line paclitaxel therapy in patients with metastatic TNBC led to longer overall survival – 19.1 months vs. 12.6 with placebo plus paclitaxel – with better survival results in patients with PIK3CA/AKT1/PTEN altered tumors.
But there’s more to learn about treating metastatic TNBC. “Relapses tend to occur early in TNBC, and some tumors are inherently resistant to chemotherapy from the get-go,” said Charles Shapiro, MD, medical oncologist, Icahn School of Medicine at Mount Sinai, New York. “Understanding the causes of drug response and resistance in patients with metastatic TNBC represents the holy grail.”
Dr. Nanda agreed, noting that advancing treatments for TNBC will hinge on identifying the key factors driving metastasis. “For TNBC, we are still trying to elucidate the best molecular targets, while at the same time trying to identify robust biomarkers to predict benefit from therapies we already have available,” she said.
A version of this article first appeared on Medscape.com.
Treating triple-negative breast cancer (TNBC), one of the more lethal breast cancer subtypes, remains a challenge. By definition, TNBC lacks the three telltale molecular signatures known to spur tumor growth: estrogen receptor (ER), progesterone receptor (PR), and human epidermal growth factor receptor 2 (HER2). A growing amount of literature shows that these frequently aggressive tumors harbor a rich array of molecular characteristics but no clear oncogenic driver.
“TNBC is incredibly heterogeneous, which makes it challenging to treat,” said Rita Nanda, MD, director of the breast oncology program and associate professor of medicine at the University of Chicago. “We have subsets of TNBC that don’t respond to currently available therapies and, as of yet, have no identifiable therapeutic targets.”
Overall, about 40% of patients with TNBC show a pathologic complete response after first-line neoadjuvant chemotherapy – typically anthracycline and taxane-based agents. But for 50% of patients, chemotherapy leaves behind substantial residual cancer tissue. These patients subsequently face a 40%-80% risk for recurrence and progression to advanced disease.
When triple-negative disease metastasizes, survival rates plummet. The most recent data from the National Cancer Institute, which tracked patients by stage of diagnosis between 2010 and 2016, showed steep declines in 5-year survival as TNBC progressed from local (91.2%) to regional (65%) to advanced-stage disease (11.5%).
Experts have started to make headway identifying and targeting different molecular features of advanced TNBC. These approaches often focus on three key areas: targeting cell surface proteins or oncogenes, stimulating an anticancer immune response, or inhibiting an overactive signaling pathway.
“For a patient with metastatic breast cancer, finding a molecular target or an oncogenic driver is essential,” said Kelly McCann, MD, PhD, a hematologist/oncologist in the department of medicine at the University of California, Los Angeles. “Because TNBC encompasses many different molecular subsets of breast cancer, the development of effective new therapeutics is going to depend on subdividing TNBC into categories with more clear targets.”
A targeted strategy
The Food and Drug Administration’s approval of sacituzumab govitecan, the first antibody-drug conjugate to treat metastatic TNBC, marked an important addition to the TNBC drug armamentarium. “Sacituzumab govitecan is one of the most exciting drugs available for the treatment of metastatic disease,” Dr. Nanda said.
Sacituzumab govitecan, approved as third-line therapy for metastatic TNBC, works by targeting the cell surface protein TROP2, expressed in about 88% of TNBC tumors but rarely in healthy cells.
In the phase 1/2 ASCENT trial, the median progression-free survival was 5.5 months and overall survival was 13.0 months in 108 patients with metastatic TNBC who had received at least two therapies prior to sacituzumab govitecan.
A subsequent phase 3 trial showed progression-free survival of 5.6 months with sacituzumab govitecan and 1.7 months with physician’s choice of chemotherapy. The median overall survival was 12.1 months and 6.7 months, respectively.
But, according to the analysis, TROP2 expression did not necessarily predict who would benefit from sacituzumab govitecan. A biomarker study revealed that although patients with moderate to high TROP2 expression exhibited the strongest treatment response, those with low TROP2 expression also survived longer when given sacituzumab govitecan, compared with chemotherapy alone.
In other words, “patients did better on sacituzumab govitecan regardless of TROP2 expression, which suggests we do not have a good biomarker for identifying who will benefit,” Dr. Nanda said.
Two other investigational antibody-drug conjugates, trastuzumab deruxtecan and ladiratuzumab vedotin, show promise in the metastatic space as well. For instance, the recent phase 2 trial evaluating trastuzumab deruxtecan in patients with HER2-positive breast cancer reported treatment response in 44% of patients with HER2-low tumors.
Given that about 36.6% of TNBC tumors exhibit low levels of HER2 expression, “trastuzumab deruxtecan represents potential in treating HER2-low TNBC,” said Yuan Yuan, MD, PhD, medical oncologist at City of Hope, a comprehensive cancer center in Los Angeles County.
Early results from a phase 1b study showed that trastuzumab deruxtecan produced a response rate of 37% in patients with HER2-low breast cancer.
Investigators are now recruiting for an open-label phase 3 trial to determine whether trastuzumab deruxtecan extends survival in patients with HER2-low metastatic breast cancers.
Immunotherapy advances
Immune checkpoint inhibitors represent another promising treatment avenue for metastatic TNBC. Pembrolizumab and atezolizumab, recently approved by the FDA, show moderate progression-free and overall survival benefits in patients with metastatic TNBC expressing PD-L1. Estimates of PD-L1 immune cells present in TNBC tumors vary widely, from about 20% to 65%.
Yet, data on which patients will benefit are not so clear-cut. “These drugs give us more choices and represent the fast-evolving therapeutic landscape in TNBC, but they also leave a lot of unanswered questions about PD-L1 as a biomarker,” Dr. Yuan said.
Take two recent phase 3 trials evaluating atezolizumab: IMpassion130 and IMpassion131. In IMpassion130, patients with PDL1–positive tumors exhibited significantly longer median overall survival on atezolizumab plus nab-paclitaxel (25.0 months) compared with nab-paclitaxel alone (15.5 months). As with the trend observed in the TROP2 data for sacituzumab govitecan, all patients survived longer on atezolizumab plus nab-paclitaxel regardless of PD-L1 status: 21.3 months vs. 17.6 months with nab-paclitaxel alone.
However, in IMpassion131, neither progression-free survival nor overall survival significantly improved in the PD-L1–positive group receiving atezolizumab plus paclitaxel compared with paclitaxel alone: Progression-free survival was 5.7 months vs. 6 months, respectively, and overall survival was 28.3 months vs. 22.1 months.
“It is unclear why this study failed to demonstrate a significant improvement in progression-free survival with the addition of atezolizumab to paclitaxel,” Dr. Nanda said. “Perhaps the negative finding has to do with how the trial was conducted, or perhaps the PD-L1 assay used is an unreliable biomarker of immunotherapy benefit.”
Continued efforts to understand TNBC
Given the diversity of metastatic TNBC and the absence of clear molecular targets, researchers are exploring a host of therapeutic strategies in addition to antibody-drug conjugates and immunotherapies.
On the oncogene front, researchers are investigating common mutations in TNBC. About 11% of TNBC tumors, for instance, carry germline mutations in BRCA1 and BRCA2. These tumors may be more likely to respond to platinum agents and PARP inhibitors, such as FDA-approved olaparib. In a phase 3 trial, patients with metastatic HER2-negative breast cancer and a germline BRCA mutation who received olaparib exhibited a 2.8-month longer median progression-free survival and a 42% reduced risk for disease progression or death compared with those on standard chemotherapy.
When considering signaling pathways, the PI3K/AKT/mTOR pathway has been the target of numerous clinical trials. Dysregulation of signaling through the PI3K and AKT signaling pathway occurs in 25%-30% of patients with advanced TNBC, and AKT inhibitors have been shown to extend survival in these patients. Data show, for instance, that adding capivasertib to first-line paclitaxel therapy in patients with metastatic TNBC led to longer overall survival – 19.1 months vs. 12.6 with placebo plus paclitaxel – with better survival results in patients with PIK3CA/AKT1/PTEN altered tumors.
But there’s more to learn about treating metastatic TNBC. “Relapses tend to occur early in TNBC, and some tumors are inherently resistant to chemotherapy from the get-go,” said Charles Shapiro, MD, medical oncologist, Icahn School of Medicine at Mount Sinai, New York. “Understanding the causes of drug response and resistance in patients with metastatic TNBC represents the holy grail.”
Dr. Nanda agreed, noting that advancing treatments for TNBC will hinge on identifying the key factors driving metastasis. “For TNBC, we are still trying to elucidate the best molecular targets, while at the same time trying to identify robust biomarkers to predict benefit from therapies we already have available,” she said.
A version of this article first appeared on Medscape.com.
Treating triple-negative breast cancer (TNBC), one of the more lethal breast cancer subtypes, remains a challenge. By definition, TNBC lacks the three telltale molecular signatures known to spur tumor growth: estrogen receptor (ER), progesterone receptor (PR), and human epidermal growth factor receptor 2 (HER2). A growing amount of literature shows that these frequently aggressive tumors harbor a rich array of molecular characteristics but no clear oncogenic driver.
“TNBC is incredibly heterogeneous, which makes it challenging to treat,” said Rita Nanda, MD, director of the breast oncology program and associate professor of medicine at the University of Chicago. “We have subsets of TNBC that don’t respond to currently available therapies and, as of yet, have no identifiable therapeutic targets.”
Overall, about 40% of patients with TNBC show a pathologic complete response after first-line neoadjuvant chemotherapy – typically anthracycline and taxane-based agents. But for 50% of patients, chemotherapy leaves behind substantial residual cancer tissue. These patients subsequently face a 40%-80% risk for recurrence and progression to advanced disease.
When triple-negative disease metastasizes, survival rates plummet. The most recent data from the National Cancer Institute, which tracked patients by stage of diagnosis between 2010 and 2016, showed steep declines in 5-year survival as TNBC progressed from local (91.2%) to regional (65%) to advanced-stage disease (11.5%).
Experts have started to make headway identifying and targeting different molecular features of advanced TNBC. These approaches often focus on three key areas: targeting cell surface proteins or oncogenes, stimulating an anticancer immune response, or inhibiting an overactive signaling pathway.
“For a patient with metastatic breast cancer, finding a molecular target or an oncogenic driver is essential,” said Kelly McCann, MD, PhD, a hematologist/oncologist in the department of medicine at the University of California, Los Angeles. “Because TNBC encompasses many different molecular subsets of breast cancer, the development of effective new therapeutics is going to depend on subdividing TNBC into categories with more clear targets.”
A targeted strategy
The Food and Drug Administration’s approval of sacituzumab govitecan, the first antibody-drug conjugate to treat metastatic TNBC, marked an important addition to the TNBC drug armamentarium. “Sacituzumab govitecan is one of the most exciting drugs available for the treatment of metastatic disease,” Dr. Nanda said.
Sacituzumab govitecan, approved as third-line therapy for metastatic TNBC, works by targeting the cell surface protein TROP2, expressed in about 88% of TNBC tumors but rarely in healthy cells.
In the phase 1/2 ASCENT trial, the median progression-free survival was 5.5 months and overall survival was 13.0 months in 108 patients with metastatic TNBC who had received at least two therapies prior to sacituzumab govitecan.
A subsequent phase 3 trial showed progression-free survival of 5.6 months with sacituzumab govitecan and 1.7 months with physician’s choice of chemotherapy. The median overall survival was 12.1 months and 6.7 months, respectively.
But, according to the analysis, TROP2 expression did not necessarily predict who would benefit from sacituzumab govitecan. A biomarker study revealed that although patients with moderate to high TROP2 expression exhibited the strongest treatment response, those with low TROP2 expression also survived longer when given sacituzumab govitecan, compared with chemotherapy alone.
In other words, “patients did better on sacituzumab govitecan regardless of TROP2 expression, which suggests we do not have a good biomarker for identifying who will benefit,” Dr. Nanda said.
Two other investigational antibody-drug conjugates, trastuzumab deruxtecan and ladiratuzumab vedotin, show promise in the metastatic space as well. For instance, the recent phase 2 trial evaluating trastuzumab deruxtecan in patients with HER2-positive breast cancer reported treatment response in 44% of patients with HER2-low tumors.
Given that about 36.6% of TNBC tumors exhibit low levels of HER2 expression, “trastuzumab deruxtecan represents potential in treating HER2-low TNBC,” said Yuan Yuan, MD, PhD, medical oncologist at City of Hope, a comprehensive cancer center in Los Angeles County.
Early results from a phase 1b study showed that trastuzumab deruxtecan produced a response rate of 37% in patients with HER2-low breast cancer.
Investigators are now recruiting for an open-label phase 3 trial to determine whether trastuzumab deruxtecan extends survival in patients with HER2-low metastatic breast cancers.
Immunotherapy advances
Immune checkpoint inhibitors represent another promising treatment avenue for metastatic TNBC. Pembrolizumab and atezolizumab, recently approved by the FDA, show moderate progression-free and overall survival benefits in patients with metastatic TNBC expressing PD-L1. Estimates of PD-L1 immune cells present in TNBC tumors vary widely, from about 20% to 65%.
Yet, data on which patients will benefit are not so clear-cut. “These drugs give us more choices and represent the fast-evolving therapeutic landscape in TNBC, but they also leave a lot of unanswered questions about PD-L1 as a biomarker,” Dr. Yuan said.
Take two recent phase 3 trials evaluating atezolizumab: IMpassion130 and IMpassion131. In IMpassion130, patients with PDL1–positive tumors exhibited significantly longer median overall survival on atezolizumab plus nab-paclitaxel (25.0 months) compared with nab-paclitaxel alone (15.5 months). As with the trend observed in the TROP2 data for sacituzumab govitecan, all patients survived longer on atezolizumab plus nab-paclitaxel regardless of PD-L1 status: 21.3 months vs. 17.6 months with nab-paclitaxel alone.
However, in IMpassion131, neither progression-free survival nor overall survival significantly improved in the PD-L1–positive group receiving atezolizumab plus paclitaxel compared with paclitaxel alone: Progression-free survival was 5.7 months vs. 6 months, respectively, and overall survival was 28.3 months vs. 22.1 months.
“It is unclear why this study failed to demonstrate a significant improvement in progression-free survival with the addition of atezolizumab to paclitaxel,” Dr. Nanda said. “Perhaps the negative finding has to do with how the trial was conducted, or perhaps the PD-L1 assay used is an unreliable biomarker of immunotherapy benefit.”
Continued efforts to understand TNBC
Given the diversity of metastatic TNBC and the absence of clear molecular targets, researchers are exploring a host of therapeutic strategies in addition to antibody-drug conjugates and immunotherapies.
On the oncogene front, researchers are investigating common mutations in TNBC. About 11% of TNBC tumors, for instance, carry germline mutations in BRCA1 and BRCA2. These tumors may be more likely to respond to platinum agents and PARP inhibitors, such as FDA-approved olaparib. In a phase 3 trial, patients with metastatic HER2-negative breast cancer and a germline BRCA mutation who received olaparib exhibited a 2.8-month longer median progression-free survival and a 42% reduced risk for disease progression or death compared with those on standard chemotherapy.
When considering signaling pathways, the PI3K/AKT/mTOR pathway has been the target of numerous clinical trials. Dysregulation of signaling through the PI3K and AKT signaling pathway occurs in 25%-30% of patients with advanced TNBC, and AKT inhibitors have been shown to extend survival in these patients. Data show, for instance, that adding capivasertib to first-line paclitaxel therapy in patients with metastatic TNBC led to longer overall survival – 19.1 months vs. 12.6 with placebo plus paclitaxel – with better survival results in patients with PIK3CA/AKT1/PTEN altered tumors.
But there’s more to learn about treating metastatic TNBC. “Relapses tend to occur early in TNBC, and some tumors are inherently resistant to chemotherapy from the get-go,” said Charles Shapiro, MD, medical oncologist, Icahn School of Medicine at Mount Sinai, New York. “Understanding the causes of drug response and resistance in patients with metastatic TNBC represents the holy grail.”
Dr. Nanda agreed, noting that advancing treatments for TNBC will hinge on identifying the key factors driving metastasis. “For TNBC, we are still trying to elucidate the best molecular targets, while at the same time trying to identify robust biomarkers to predict benefit from therapies we already have available,” she said.
A version of this article first appeared on Medscape.com.
Clinical Edge Journal Scan Commentary: Breast Cancer April 2021
Adjuvant endocrine therapy (ET) has been shown to reduce risk of recurrence and improve survival in HR+ early breast cancer (EBC). A retrospective matched cohort analysis of 1,972 patients ≥70 years of age with HR+/HER2-negative, node-negative EBC and multiple medical co-morbidities, demonstrated higher median overall survival (OS) in patients who received ET versus those who did not (79.2 months compared to 67.7 months, p<0.0001). Older women with multiple medical co-morbidities may be at greater risk of toxicities related to ET, which can impact quality of life and adherence. The probability of initiating ET has been shown to be greater among patients who underwent radiation, thus highlighting the need to identify those who will adhere to ET when making decisions regarding radiation therapy to optimize care. ET should be discussed with all women who are candidates for this therapy, with a focus on individualized benefit and risk of these agents.
Studies of immunotherapy in metastatic triple-negative breast cancer have shown promising results, notably in patients with PD-L1 enriched tumors (IMpassion 130 and KEYNOTE-355). KEYNOTE-119 was a phase 3 study including 1,098 patients with metastatic TNBC who had received one or two previous systemic therapies for metastatic disease and progressed on most recent treatment, with randomization to pembrolizumab or physician’s choice chemotherapy . Median OS for pembrolizumab compared to chemotherapy was similar in the overall population (9.9 months versus 10.8 months, HR 0.97) and in patients with PD-L1 CPS score of ≥10 (12.7 months versus 11.6 months, HR 0.78, p=0.057). Although not statistically significant, greater PD-L1 expression was associated with longer median OS with pembrolizumab. Responses to immunotherapy may be durable in select patients, and these agents have an overall favorable toxicity profile. Novel immunotherapy combinations as well as biomarkers to predict response are certainly desired in this space.
Combination endocrine therapy plus a CDK 4/6 inhibitor is standard first-line treatment for metastatic HR+/HER2-negative breast cancer. Neutropenia is a common adverse event (AE) seen with the CDK 4/6 inhibitor palbociclib, and dosing guidelines provide a clear algorithm for drug monitoring and adjustment for cytopenias. Cutaneous toxicities, although not commonly seen, have been reported in the literature. Chawla and colleagues performed a retrospective analysis including 324 patients with advanced HR+/HER2-negative breast cancer who received palbociclib plus endocrine therapy. Cutaneous AEs were seen in 14.2%, with a significant proportion occurring early (41% occurred during or after the first cycle) and 50% resolved within 14 days (average 43 days). Of those who developed cutaneous AEs (n=46), only 15% and 4% required temporary hold and permanent cessation of therapy, respectively. These findings indicate a low overall incidence of cutaneous AEs associated with palbociclib, however highlight the importance of prompt recognition, management, and dermatology referral as appropriate, to help maintain patients on effective cancer-directed therapy.
References:
Yau C, van der Noordaa M, Wei J, et al. Residual cancer burden after neoadjuvant therapy and long-term survival outcomes in breast cancer: a multi-center pooled analysis. Presented at: 2019 San Antonio Breast Cancer Symposium; December 10-14, 2019; San Antonio, TX. Abstract GS5-01.
Wei M, Wang X, Zimmerman DN, Burt LM, Haaland B, Henry NL. Endocrine therapy and radiotherapy use among older women with hormone receptor-positive, clinically node-negative breast cancer. Breast Cancer Res Treat. 2021 Jan 9. doi: 10.1007/s10549-020-06071-w.Epub ahead of print.
Emens LA, Adams S, Barrios CH, et al. IMpassion130: Final OS analysis from the pivotal phase III study of atezolizumab plus nabpaclitaxel versus placebo plus nabpaclitaxel in previously untreated locally advanced or metastatic triple-negative breast cancer. Presented at: ESMO Virtual Congress 2020, Ann Oncol. 2020;31S:ESMO #LBA16.
Khan NAJ, Alsharedi M. Bullous Skin Rash: A Rare Case of Palbociclib-Induced Dermatological Toxicity. Cureus. 2020 Sep 3;12(9):e10229. doi: 10.7759/cureus.10229.
Adjuvant endocrine therapy (ET) has been shown to reduce risk of recurrence and improve survival in HR+ early breast cancer (EBC). A retrospective matched cohort analysis of 1,972 patients ≥70 years of age with HR+/HER2-negative, node-negative EBC and multiple medical co-morbidities, demonstrated higher median overall survival (OS) in patients who received ET versus those who did not (79.2 months compared to 67.7 months, p<0.0001). Older women with multiple medical co-morbidities may be at greater risk of toxicities related to ET, which can impact quality of life and adherence. The probability of initiating ET has been shown to be greater among patients who underwent radiation, thus highlighting the need to identify those who will adhere to ET when making decisions regarding radiation therapy to optimize care. ET should be discussed with all women who are candidates for this therapy, with a focus on individualized benefit and risk of these agents.
Studies of immunotherapy in metastatic triple-negative breast cancer have shown promising results, notably in patients with PD-L1 enriched tumors (IMpassion 130 and KEYNOTE-355). KEYNOTE-119 was a phase 3 study including 1,098 patients with metastatic TNBC who had received one or two previous systemic therapies for metastatic disease and progressed on most recent treatment, with randomization to pembrolizumab or physician’s choice chemotherapy . Median OS for pembrolizumab compared to chemotherapy was similar in the overall population (9.9 months versus 10.8 months, HR 0.97) and in patients with PD-L1 CPS score of ≥10 (12.7 months versus 11.6 months, HR 0.78, p=0.057). Although not statistically significant, greater PD-L1 expression was associated with longer median OS with pembrolizumab. Responses to immunotherapy may be durable in select patients, and these agents have an overall favorable toxicity profile. Novel immunotherapy combinations as well as biomarkers to predict response are certainly desired in this space.
Combination endocrine therapy plus a CDK 4/6 inhibitor is standard first-line treatment for metastatic HR+/HER2-negative breast cancer. Neutropenia is a common adverse event (AE) seen with the CDK 4/6 inhibitor palbociclib, and dosing guidelines provide a clear algorithm for drug monitoring and adjustment for cytopenias. Cutaneous toxicities, although not commonly seen, have been reported in the literature. Chawla and colleagues performed a retrospective analysis including 324 patients with advanced HR+/HER2-negative breast cancer who received palbociclib plus endocrine therapy. Cutaneous AEs were seen in 14.2%, with a significant proportion occurring early (41% occurred during or after the first cycle) and 50% resolved within 14 days (average 43 days). Of those who developed cutaneous AEs (n=46), only 15% and 4% required temporary hold and permanent cessation of therapy, respectively. These findings indicate a low overall incidence of cutaneous AEs associated with palbociclib, however highlight the importance of prompt recognition, management, and dermatology referral as appropriate, to help maintain patients on effective cancer-directed therapy.
References:
Yau C, van der Noordaa M, Wei J, et al. Residual cancer burden after neoadjuvant therapy and long-term survival outcomes in breast cancer: a multi-center pooled analysis. Presented at: 2019 San Antonio Breast Cancer Symposium; December 10-14, 2019; San Antonio, TX. Abstract GS5-01.
Wei M, Wang X, Zimmerman DN, Burt LM, Haaland B, Henry NL. Endocrine therapy and radiotherapy use among older women with hormone receptor-positive, clinically node-negative breast cancer. Breast Cancer Res Treat. 2021 Jan 9. doi: 10.1007/s10549-020-06071-w.Epub ahead of print.
Emens LA, Adams S, Barrios CH, et al. IMpassion130: Final OS analysis from the pivotal phase III study of atezolizumab plus nabpaclitaxel versus placebo plus nabpaclitaxel in previously untreated locally advanced or metastatic triple-negative breast cancer. Presented at: ESMO Virtual Congress 2020, Ann Oncol. 2020;31S:ESMO #LBA16.
Khan NAJ, Alsharedi M. Bullous Skin Rash: A Rare Case of Palbociclib-Induced Dermatological Toxicity. Cureus. 2020 Sep 3;12(9):e10229. doi: 10.7759/cureus.10229.
Adjuvant endocrine therapy (ET) has been shown to reduce risk of recurrence and improve survival in HR+ early breast cancer (EBC). A retrospective matched cohort analysis of 1,972 patients ≥70 years of age with HR+/HER2-negative, node-negative EBC and multiple medical co-morbidities, demonstrated higher median overall survival (OS) in patients who received ET versus those who did not (79.2 months compared to 67.7 months, p<0.0001). Older women with multiple medical co-morbidities may be at greater risk of toxicities related to ET, which can impact quality of life and adherence. The probability of initiating ET has been shown to be greater among patients who underwent radiation, thus highlighting the need to identify those who will adhere to ET when making decisions regarding radiation therapy to optimize care. ET should be discussed with all women who are candidates for this therapy, with a focus on individualized benefit and risk of these agents.
Studies of immunotherapy in metastatic triple-negative breast cancer have shown promising results, notably in patients with PD-L1 enriched tumors (IMpassion 130 and KEYNOTE-355). KEYNOTE-119 was a phase 3 study including 1,098 patients with metastatic TNBC who had received one or two previous systemic therapies for metastatic disease and progressed on most recent treatment, with randomization to pembrolizumab or physician’s choice chemotherapy . Median OS for pembrolizumab compared to chemotherapy was similar in the overall population (9.9 months versus 10.8 months, HR 0.97) and in patients with PD-L1 CPS score of ≥10 (12.7 months versus 11.6 months, HR 0.78, p=0.057). Although not statistically significant, greater PD-L1 expression was associated with longer median OS with pembrolizumab. Responses to immunotherapy may be durable in select patients, and these agents have an overall favorable toxicity profile. Novel immunotherapy combinations as well as biomarkers to predict response are certainly desired in this space.
Combination endocrine therapy plus a CDK 4/6 inhibitor is standard first-line treatment for metastatic HR+/HER2-negative breast cancer. Neutropenia is a common adverse event (AE) seen with the CDK 4/6 inhibitor palbociclib, and dosing guidelines provide a clear algorithm for drug monitoring and adjustment for cytopenias. Cutaneous toxicities, although not commonly seen, have been reported in the literature. Chawla and colleagues performed a retrospective analysis including 324 patients with advanced HR+/HER2-negative breast cancer who received palbociclib plus endocrine therapy. Cutaneous AEs were seen in 14.2%, with a significant proportion occurring early (41% occurred during or after the first cycle) and 50% resolved within 14 days (average 43 days). Of those who developed cutaneous AEs (n=46), only 15% and 4% required temporary hold and permanent cessation of therapy, respectively. These findings indicate a low overall incidence of cutaneous AEs associated with palbociclib, however highlight the importance of prompt recognition, management, and dermatology referral as appropriate, to help maintain patients on effective cancer-directed therapy.
References:
Yau C, van der Noordaa M, Wei J, et al. Residual cancer burden after neoadjuvant therapy and long-term survival outcomes in breast cancer: a multi-center pooled analysis. Presented at: 2019 San Antonio Breast Cancer Symposium; December 10-14, 2019; San Antonio, TX. Abstract GS5-01.
Wei M, Wang X, Zimmerman DN, Burt LM, Haaland B, Henry NL. Endocrine therapy and radiotherapy use among older women with hormone receptor-positive, clinically node-negative breast cancer. Breast Cancer Res Treat. 2021 Jan 9. doi: 10.1007/s10549-020-06071-w.Epub ahead of print.
Emens LA, Adams S, Barrios CH, et al. IMpassion130: Final OS analysis from the pivotal phase III study of atezolizumab plus nabpaclitaxel versus placebo plus nabpaclitaxel in previously untreated locally advanced or metastatic triple-negative breast cancer. Presented at: ESMO Virtual Congress 2020, Ann Oncol. 2020;31S:ESMO #LBA16.
Khan NAJ, Alsharedi M. Bullous Skin Rash: A Rare Case of Palbociclib-Induced Dermatological Toxicity. Cureus. 2020 Sep 3;12(9):e10229. doi: 10.7759/cureus.10229.
HER2+ BC: Recurrence risk remains even after achieving pCR with neoadjuvant pertuzumab+trastuzumab
Key clinical point: Patients with human epidermal growth factor receptor 2-positive (HER2+) early breast cancer (BC) who achieved a pathologic complete response (pCR) with neoadjuvant pertuzumab, trastuzumab, and chemotherapy (nPTC) and then received adjuvant trastuzumab remained at risk for recurrence, indicating the need for alternative therapies.
Major finding: At 4 years, the invasive disease-free survival rates for the overall, node-positive, and node-negative cohorts were 90.0% (95% confidence interval [CI], 84.6%-93.6%), 86.2% (95% CI, 78.1%-91.4%), and 96.0% (95% CI, 88.0%-98.7%), respectively.
Study details: Findings are from a real-world, retrospective analysis of 217 patients with HER2+ locally advanced, high-risk early-stage BC, including 135 patients with node-positive and 77 with node-negative disease. Patients received nPTC, had pCR, and then received adjuvant trastuzumab.
Disclosures: This study was funded by Genentech, Inc. Some investigators including the lead author reported ties with various pharmaceutical companies, including Genentech. J Sussell, A Cheng, and A Fung declared being employees of Genentech, Inc.
Source: O’Shaughnessy J et al. Breast Cancer Res Treat. 2021 Mar 1. doi: 10.1007/s10549-021-06137-3.
Key clinical point: Patients with human epidermal growth factor receptor 2-positive (HER2+) early breast cancer (BC) who achieved a pathologic complete response (pCR) with neoadjuvant pertuzumab, trastuzumab, and chemotherapy (nPTC) and then received adjuvant trastuzumab remained at risk for recurrence, indicating the need for alternative therapies.
Major finding: At 4 years, the invasive disease-free survival rates for the overall, node-positive, and node-negative cohorts were 90.0% (95% confidence interval [CI], 84.6%-93.6%), 86.2% (95% CI, 78.1%-91.4%), and 96.0% (95% CI, 88.0%-98.7%), respectively.
Study details: Findings are from a real-world, retrospective analysis of 217 patients with HER2+ locally advanced, high-risk early-stage BC, including 135 patients with node-positive and 77 with node-negative disease. Patients received nPTC, had pCR, and then received adjuvant trastuzumab.
Disclosures: This study was funded by Genentech, Inc. Some investigators including the lead author reported ties with various pharmaceutical companies, including Genentech. J Sussell, A Cheng, and A Fung declared being employees of Genentech, Inc.
Source: O’Shaughnessy J et al. Breast Cancer Res Treat. 2021 Mar 1. doi: 10.1007/s10549-021-06137-3.
Key clinical point: Patients with human epidermal growth factor receptor 2-positive (HER2+) early breast cancer (BC) who achieved a pathologic complete response (pCR) with neoadjuvant pertuzumab, trastuzumab, and chemotherapy (nPTC) and then received adjuvant trastuzumab remained at risk for recurrence, indicating the need for alternative therapies.
Major finding: At 4 years, the invasive disease-free survival rates for the overall, node-positive, and node-negative cohorts were 90.0% (95% confidence interval [CI], 84.6%-93.6%), 86.2% (95% CI, 78.1%-91.4%), and 96.0% (95% CI, 88.0%-98.7%), respectively.
Study details: Findings are from a real-world, retrospective analysis of 217 patients with HER2+ locally advanced, high-risk early-stage BC, including 135 patients with node-positive and 77 with node-negative disease. Patients received nPTC, had pCR, and then received adjuvant trastuzumab.
Disclosures: This study was funded by Genentech, Inc. Some investigators including the lead author reported ties with various pharmaceutical companies, including Genentech. J Sussell, A Cheng, and A Fung declared being employees of Genentech, Inc.
Source: O’Shaughnessy J et al. Breast Cancer Res Treat. 2021 Mar 1. doi: 10.1007/s10549-021-06137-3.