User login
Clinical pearls for administering cognitive exams during the pandemic
Patients have often been labeled as “poor historians” if they are not able to recollect their own medical history, whether through illness or difficulties in communication. But Fred Ovsiew, MD, speaking at Focus on Neuropsychiatry presented by Current Psychiatry and the American Academy of Clinical Psychiatrists, sees that label as an excuse on the part of the clinician.
“I strongly advise you to drop that phrase from your vocabulary if you do use it, because the patient is not the historian. The doctor, the clinician is the historian,” Dr. Ovsiew said at the meeting, presented by Global Academy for Medical Education. “It is the clinician’s job to put the story together using the account by the patient as one source, but [also] interviewing a collateral informant and/or reviewing records, which is necessary in almost every case of a neuropsychiatric illness.”
Rather, clinicians taking history at the bedside should focus on why the patients cannot give a narrative account of their illness. Patients can have narrative incapacity on a psychogenic basis, such as in patients with conversion or somatoform disorder, he explained. “I think this is a result of the narrative incapacity that develops in people who have had trauma or adverse experiences in childhood and insecure attachment. This is shown on the adult attachment interview as a disorganized account of their childhoods.”
Other patients might not be able to recount their medical history because they are amnestic, which leaves their account vague because of a lack of access to information. “It may be frozen in time in the sense that, up to a certain point in their life, they can recount the history,” Dr. Ovsiew said. “But in recent years, their account becomes vague.”
Patients with right hemisphere lesions might not know that their account has incongruity and is implausible, while patients with dorsolateral prefrontal lesions might be aspontaneous, use few words to describe their situation, and have poor insight. Those with ventromedial prefrontal lesions can be impulsive and have poor insight, not considering alternative possibilities, Dr. Ovsiew noted.
Asking open-ended questions of the patient is the first step to identifying any potential narrative incapacity, followed by a detailed medical history by the clinician. When taking a medical history, try avoiding what Dr. Ovsiew calls the “anything like that?” problem, where a clinician asks a question about a cluster of symptoms that would make sense to a doctor, but not a patient. For example, a doctor might ask whether a patient is experiencing “chest pain or leg swelling – anything like that?” because he or she knows what those symptoms have in common, but the patient might not know the relationship between those symptoms. “You can’t count on the patient to tell you all the relevant information,” he said. “You have to know what to ask about.”
“Patients with brain disease have subtle personality changes, sometimes more obvious personality changes. These need to be inquired about,” Dr. Ovsiew said. “The patient with apathy has reduced negative as well as positive emotions. The patient with depression has reduced positive emotions, but often tells you very clearly about the negative emotions of sadness, guilt. The patient with depression has diurnal variation in mood, a very telling symptom, especially when it’s disclosed spontaneously,” Dr. Ovsiew explained. “The point is, you need to know to ask about it.”
When taking a sleep history, clinicians should be aware of sleep disturbances apart from insomnia and early waking. REM sleep behavior disorder is a condition that should be inquired about. Obstructive sleep apnea is a condition that might not be immediately apparent to the patient, but a bed partner can identify whether a patient has problems breathing throughout the night.
“This is an important condition to uncover for the neuropsychiatrist because it contributes to treatment resistance and depression, and it contributes to cognitive impairment,” Dr. Ovsiew said. “These patients commonly have mild difficulties with attention and concentration.”
Always ask about head injury in every history, which can be relevant to later onset depression, PTSD, and cognitive impairment. Every head injury follows a trajectory of retrograde amnesia and altered state of consciousness (including coma), followed by a period of posttraumatic amnesia. Duration of these states can be used to assess the severity of brain injury, but the 15-point Glasgow Coma Scale is another way to assess injury severity, Dr. Ovsiew explained.
However, the two do not always overlap, he noted. “Someone may have a Glasgow Coma Scale score that is 9-12, predicting moderate brain injury, but they may have a short duration of amnesia. These don’t always follow the same path. There are many different ways of classifying how severe the brain injury is.”
Keep probes brief, straightforward
Cognitive exams of patients with suspected psychiatric disorders should be simple, easy to administer and focused on a single domain of cognition. “Probes should be brief. They should not require specialized equipment. The Purdue Pegboard Test might be a great neuropsychological instrument, but very few of us carry a pegboard around in our medical bags,” Dr. Ovsiew said.
The probe administered should also be accessible to the patient. The serial sevens clinical test, where a patient is asked to repeatedly subtract 7 from 100, is only effective at testing concentration if the patient is capable of completing the test. “There are going to be patients who can’t do the task, but it’s not because of concentration failure, it’s because of subtraction failure,” he said.
When assessing attention, effective tasks include having the patient perform the digit span test forward and backward, count backward from 20 to 1, listing the months of the year in reverse, and performing the Mental Alternation Test. However, Dr. Ovsiew explained there may be some barriers for patients in completing these tasks. “The person may be aphasic and not know the alphabet. The person may have English as a second language and not be skilled at giving the alphabet in English. In some cases, you may want to check and not assume that the patient can count and does know the alphabet.”
In assessing language, listen for aphasic abnormalities. “The patient, of course, is speaking throughout the interview, but you need to take a moment to listen for prosody, to listen to rate of speech, to listen for paraphasic errors or word-finding problems,” Dr. Ovsiew said. Any abnormalities should be probed further through confrontation naming tasks, which can be done in person and with some success through video, but not by phone. Naming to definition (“What do you call the part of a shirt that covers the arm?”) is one way of administering the test over the phone.
Visuospatial function can be assessed by clock drawing but also carries problems. Patients who do not plan their clock before beginning to draw, for example, may have an executive function problem instead of a visuospatial problem, Dr. Ovsiew noted. Patients in whom a clinician suspects hemineglect should be given a visual search task or line by section task. “I like doing clock drawing. It’s a nice screening test. It’s becoming, I think, less useful as people count on digital clocks and have trouble even imagining what an analog clock looks like.”
An approach that is better suited to in-person assessment, but also works by video, is the Poppelreuter figure visual perceptual function test, which is a prompt for the patient that involves common household items overlaying one another “in atypical positions and atypical configurations” where the patient is instructed to describe the items they see on the card. Another approach that works over video is the interlocking finger test, where the patient is asked to copy the hand positions made by the clinician.
Dr. Ovsiew admitted that visuospatial function is nearly impossible to assess over the phone. Asking topographical questions (“If you’re driving from Chicago to Los Angeles, is the Pacific Ocean in front of you, behind you, to your left, or to your right?”) may help judge visuospatial function, but this relies on the patient having the topographic knowledge to answer the questions. Some patients who are topographically disoriented can’t do them at all,” Dr. Ovsiew said.
Bedside neuropsychiatry assesses encoding of a memory, its retention and its retrieval as well as verbal and visual cues. Each one of these aspects of memory can be impaired on its own and should be explored separately, Dr. Ovsiew explained. “Neuropsychiatric clinicians have a rough-and-ready, seat-of-the-pants way of approaching this that wouldn’t pass muster if you’re a psychologist, but is the best we can do at the bedside.”
To test retrieval and retention, the Three Words–Three Shapes test works well in person, with some difficulty by video, and is not possible to administer over the phone. In lieu of that test, giving the patient a simple word list and asking them to repeat the list in order. Using the word list, “these different stages of memory function can be parsed out pretty well at the bedside or chairside, and even by the phone. Figuring out where the memory failure is diagnostically important,” Dr. Ovsiew said.
Executive function, which involves activation, planning, sequencing, maintaining, self-monitoring, and flexible employment of action and attention, is “complicated to evaluate because there are multiple aspects of executive function, multiple deficits that can be seen with executive dysfunction, and they don’t all correlate with each other.”
Within executive function evaluation, the Mental Alternation Test can assess working memory, motor sequencing can be assessed through the ring/fist, fist/edge/palm, alternating fist, and rampart tests. The Go/No-Go test can be used to assess response inhibition. For effortful retrieval evaluation, spontaneous word-list generation – such as thinking of all the items one can buy at a supermarket– can test category fluency, while a task to name all the words starting with a certain letter can assess letter stimulus.
Executive function “is of crucial importance in the neuropsychiatric evaluation because it’s strongly correlated with how well the person functions outside the office,” Dr. Ovsiew said.
Global Academy and this news organization are owned by the same parent company. Dr. Ovsiew reported relationships with Wolters Kluwer Health in the form of consulting, receiving royalty payments, and related activities.
Patients have often been labeled as “poor historians” if they are not able to recollect their own medical history, whether through illness or difficulties in communication. But Fred Ovsiew, MD, speaking at Focus on Neuropsychiatry presented by Current Psychiatry and the American Academy of Clinical Psychiatrists, sees that label as an excuse on the part of the clinician.
“I strongly advise you to drop that phrase from your vocabulary if you do use it, because the patient is not the historian. The doctor, the clinician is the historian,” Dr. Ovsiew said at the meeting, presented by Global Academy for Medical Education. “It is the clinician’s job to put the story together using the account by the patient as one source, but [also] interviewing a collateral informant and/or reviewing records, which is necessary in almost every case of a neuropsychiatric illness.”
Rather, clinicians taking history at the bedside should focus on why the patients cannot give a narrative account of their illness. Patients can have narrative incapacity on a psychogenic basis, such as in patients with conversion or somatoform disorder, he explained. “I think this is a result of the narrative incapacity that develops in people who have had trauma or adverse experiences in childhood and insecure attachment. This is shown on the adult attachment interview as a disorganized account of their childhoods.”
Other patients might not be able to recount their medical history because they are amnestic, which leaves their account vague because of a lack of access to information. “It may be frozen in time in the sense that, up to a certain point in their life, they can recount the history,” Dr. Ovsiew said. “But in recent years, their account becomes vague.”
Patients with right hemisphere lesions might not know that their account has incongruity and is implausible, while patients with dorsolateral prefrontal lesions might be aspontaneous, use few words to describe their situation, and have poor insight. Those with ventromedial prefrontal lesions can be impulsive and have poor insight, not considering alternative possibilities, Dr. Ovsiew noted.
Asking open-ended questions of the patient is the first step to identifying any potential narrative incapacity, followed by a detailed medical history by the clinician. When taking a medical history, try avoiding what Dr. Ovsiew calls the “anything like that?” problem, where a clinician asks a question about a cluster of symptoms that would make sense to a doctor, but not a patient. For example, a doctor might ask whether a patient is experiencing “chest pain or leg swelling – anything like that?” because he or she knows what those symptoms have in common, but the patient might not know the relationship between those symptoms. “You can’t count on the patient to tell you all the relevant information,” he said. “You have to know what to ask about.”
“Patients with brain disease have subtle personality changes, sometimes more obvious personality changes. These need to be inquired about,” Dr. Ovsiew said. “The patient with apathy has reduced negative as well as positive emotions. The patient with depression has reduced positive emotions, but often tells you very clearly about the negative emotions of sadness, guilt. The patient with depression has diurnal variation in mood, a very telling symptom, especially when it’s disclosed spontaneously,” Dr. Ovsiew explained. “The point is, you need to know to ask about it.”
When taking a sleep history, clinicians should be aware of sleep disturbances apart from insomnia and early waking. REM sleep behavior disorder is a condition that should be inquired about. Obstructive sleep apnea is a condition that might not be immediately apparent to the patient, but a bed partner can identify whether a patient has problems breathing throughout the night.
“This is an important condition to uncover for the neuropsychiatrist because it contributes to treatment resistance and depression, and it contributes to cognitive impairment,” Dr. Ovsiew said. “These patients commonly have mild difficulties with attention and concentration.”
Always ask about head injury in every history, which can be relevant to later onset depression, PTSD, and cognitive impairment. Every head injury follows a trajectory of retrograde amnesia and altered state of consciousness (including coma), followed by a period of posttraumatic amnesia. Duration of these states can be used to assess the severity of brain injury, but the 15-point Glasgow Coma Scale is another way to assess injury severity, Dr. Ovsiew explained.
However, the two do not always overlap, he noted. “Someone may have a Glasgow Coma Scale score that is 9-12, predicting moderate brain injury, but they may have a short duration of amnesia. These don’t always follow the same path. There are many different ways of classifying how severe the brain injury is.”
Keep probes brief, straightforward
Cognitive exams of patients with suspected psychiatric disorders should be simple, easy to administer and focused on a single domain of cognition. “Probes should be brief. They should not require specialized equipment. The Purdue Pegboard Test might be a great neuropsychological instrument, but very few of us carry a pegboard around in our medical bags,” Dr. Ovsiew said.
The probe administered should also be accessible to the patient. The serial sevens clinical test, where a patient is asked to repeatedly subtract 7 from 100, is only effective at testing concentration if the patient is capable of completing the test. “There are going to be patients who can’t do the task, but it’s not because of concentration failure, it’s because of subtraction failure,” he said.
When assessing attention, effective tasks include having the patient perform the digit span test forward and backward, count backward from 20 to 1, listing the months of the year in reverse, and performing the Mental Alternation Test. However, Dr. Ovsiew explained there may be some barriers for patients in completing these tasks. “The person may be aphasic and not know the alphabet. The person may have English as a second language and not be skilled at giving the alphabet in English. In some cases, you may want to check and not assume that the patient can count and does know the alphabet.”
In assessing language, listen for aphasic abnormalities. “The patient, of course, is speaking throughout the interview, but you need to take a moment to listen for prosody, to listen to rate of speech, to listen for paraphasic errors or word-finding problems,” Dr. Ovsiew said. Any abnormalities should be probed further through confrontation naming tasks, which can be done in person and with some success through video, but not by phone. Naming to definition (“What do you call the part of a shirt that covers the arm?”) is one way of administering the test over the phone.
Visuospatial function can be assessed by clock drawing but also carries problems. Patients who do not plan their clock before beginning to draw, for example, may have an executive function problem instead of a visuospatial problem, Dr. Ovsiew noted. Patients in whom a clinician suspects hemineglect should be given a visual search task or line by section task. “I like doing clock drawing. It’s a nice screening test. It’s becoming, I think, less useful as people count on digital clocks and have trouble even imagining what an analog clock looks like.”
An approach that is better suited to in-person assessment, but also works by video, is the Poppelreuter figure visual perceptual function test, which is a prompt for the patient that involves common household items overlaying one another “in atypical positions and atypical configurations” where the patient is instructed to describe the items they see on the card. Another approach that works over video is the interlocking finger test, where the patient is asked to copy the hand positions made by the clinician.
Dr. Ovsiew admitted that visuospatial function is nearly impossible to assess over the phone. Asking topographical questions (“If you’re driving from Chicago to Los Angeles, is the Pacific Ocean in front of you, behind you, to your left, or to your right?”) may help judge visuospatial function, but this relies on the patient having the topographic knowledge to answer the questions. Some patients who are topographically disoriented can’t do them at all,” Dr. Ovsiew said.
Bedside neuropsychiatry assesses encoding of a memory, its retention and its retrieval as well as verbal and visual cues. Each one of these aspects of memory can be impaired on its own and should be explored separately, Dr. Ovsiew explained. “Neuropsychiatric clinicians have a rough-and-ready, seat-of-the-pants way of approaching this that wouldn’t pass muster if you’re a psychologist, but is the best we can do at the bedside.”
To test retrieval and retention, the Three Words–Three Shapes test works well in person, with some difficulty by video, and is not possible to administer over the phone. In lieu of that test, giving the patient a simple word list and asking them to repeat the list in order. Using the word list, “these different stages of memory function can be parsed out pretty well at the bedside or chairside, and even by the phone. Figuring out where the memory failure is diagnostically important,” Dr. Ovsiew said.
Executive function, which involves activation, planning, sequencing, maintaining, self-monitoring, and flexible employment of action and attention, is “complicated to evaluate because there are multiple aspects of executive function, multiple deficits that can be seen with executive dysfunction, and they don’t all correlate with each other.”
Within executive function evaluation, the Mental Alternation Test can assess working memory, motor sequencing can be assessed through the ring/fist, fist/edge/palm, alternating fist, and rampart tests. The Go/No-Go test can be used to assess response inhibition. For effortful retrieval evaluation, spontaneous word-list generation – such as thinking of all the items one can buy at a supermarket– can test category fluency, while a task to name all the words starting with a certain letter can assess letter stimulus.
Executive function “is of crucial importance in the neuropsychiatric evaluation because it’s strongly correlated with how well the person functions outside the office,” Dr. Ovsiew said.
Global Academy and this news organization are owned by the same parent company. Dr. Ovsiew reported relationships with Wolters Kluwer Health in the form of consulting, receiving royalty payments, and related activities.
Patients have often been labeled as “poor historians” if they are not able to recollect their own medical history, whether through illness or difficulties in communication. But Fred Ovsiew, MD, speaking at Focus on Neuropsychiatry presented by Current Psychiatry and the American Academy of Clinical Psychiatrists, sees that label as an excuse on the part of the clinician.
“I strongly advise you to drop that phrase from your vocabulary if you do use it, because the patient is not the historian. The doctor, the clinician is the historian,” Dr. Ovsiew said at the meeting, presented by Global Academy for Medical Education. “It is the clinician’s job to put the story together using the account by the patient as one source, but [also] interviewing a collateral informant and/or reviewing records, which is necessary in almost every case of a neuropsychiatric illness.”
Rather, clinicians taking history at the bedside should focus on why the patients cannot give a narrative account of their illness. Patients can have narrative incapacity on a psychogenic basis, such as in patients with conversion or somatoform disorder, he explained. “I think this is a result of the narrative incapacity that develops in people who have had trauma or adverse experiences in childhood and insecure attachment. This is shown on the adult attachment interview as a disorganized account of their childhoods.”
Other patients might not be able to recount their medical history because they are amnestic, which leaves their account vague because of a lack of access to information. “It may be frozen in time in the sense that, up to a certain point in their life, they can recount the history,” Dr. Ovsiew said. “But in recent years, their account becomes vague.”
Patients with right hemisphere lesions might not know that their account has incongruity and is implausible, while patients with dorsolateral prefrontal lesions might be aspontaneous, use few words to describe their situation, and have poor insight. Those with ventromedial prefrontal lesions can be impulsive and have poor insight, not considering alternative possibilities, Dr. Ovsiew noted.
Asking open-ended questions of the patient is the first step to identifying any potential narrative incapacity, followed by a detailed medical history by the clinician. When taking a medical history, try avoiding what Dr. Ovsiew calls the “anything like that?” problem, where a clinician asks a question about a cluster of symptoms that would make sense to a doctor, but not a patient. For example, a doctor might ask whether a patient is experiencing “chest pain or leg swelling – anything like that?” because he or she knows what those symptoms have in common, but the patient might not know the relationship between those symptoms. “You can’t count on the patient to tell you all the relevant information,” he said. “You have to know what to ask about.”
“Patients with brain disease have subtle personality changes, sometimes more obvious personality changes. These need to be inquired about,” Dr. Ovsiew said. “The patient with apathy has reduced negative as well as positive emotions. The patient with depression has reduced positive emotions, but often tells you very clearly about the negative emotions of sadness, guilt. The patient with depression has diurnal variation in mood, a very telling symptom, especially when it’s disclosed spontaneously,” Dr. Ovsiew explained. “The point is, you need to know to ask about it.”
When taking a sleep history, clinicians should be aware of sleep disturbances apart from insomnia and early waking. REM sleep behavior disorder is a condition that should be inquired about. Obstructive sleep apnea is a condition that might not be immediately apparent to the patient, but a bed partner can identify whether a patient has problems breathing throughout the night.
“This is an important condition to uncover for the neuropsychiatrist because it contributes to treatment resistance and depression, and it contributes to cognitive impairment,” Dr. Ovsiew said. “These patients commonly have mild difficulties with attention and concentration.”
Always ask about head injury in every history, which can be relevant to later onset depression, PTSD, and cognitive impairment. Every head injury follows a trajectory of retrograde amnesia and altered state of consciousness (including coma), followed by a period of posttraumatic amnesia. Duration of these states can be used to assess the severity of brain injury, but the 15-point Glasgow Coma Scale is another way to assess injury severity, Dr. Ovsiew explained.
However, the two do not always overlap, he noted. “Someone may have a Glasgow Coma Scale score that is 9-12, predicting moderate brain injury, but they may have a short duration of amnesia. These don’t always follow the same path. There are many different ways of classifying how severe the brain injury is.”
Keep probes brief, straightforward
Cognitive exams of patients with suspected psychiatric disorders should be simple, easy to administer and focused on a single domain of cognition. “Probes should be brief. They should not require specialized equipment. The Purdue Pegboard Test might be a great neuropsychological instrument, but very few of us carry a pegboard around in our medical bags,” Dr. Ovsiew said.
The probe administered should also be accessible to the patient. The serial sevens clinical test, where a patient is asked to repeatedly subtract 7 from 100, is only effective at testing concentration if the patient is capable of completing the test. “There are going to be patients who can’t do the task, but it’s not because of concentration failure, it’s because of subtraction failure,” he said.
When assessing attention, effective tasks include having the patient perform the digit span test forward and backward, count backward from 20 to 1, listing the months of the year in reverse, and performing the Mental Alternation Test. However, Dr. Ovsiew explained there may be some barriers for patients in completing these tasks. “The person may be aphasic and not know the alphabet. The person may have English as a second language and not be skilled at giving the alphabet in English. In some cases, you may want to check and not assume that the patient can count and does know the alphabet.”
In assessing language, listen for aphasic abnormalities. “The patient, of course, is speaking throughout the interview, but you need to take a moment to listen for prosody, to listen to rate of speech, to listen for paraphasic errors or word-finding problems,” Dr. Ovsiew said. Any abnormalities should be probed further through confrontation naming tasks, which can be done in person and with some success through video, but not by phone. Naming to definition (“What do you call the part of a shirt that covers the arm?”) is one way of administering the test over the phone.
Visuospatial function can be assessed by clock drawing but also carries problems. Patients who do not plan their clock before beginning to draw, for example, may have an executive function problem instead of a visuospatial problem, Dr. Ovsiew noted. Patients in whom a clinician suspects hemineglect should be given a visual search task or line by section task. “I like doing clock drawing. It’s a nice screening test. It’s becoming, I think, less useful as people count on digital clocks and have trouble even imagining what an analog clock looks like.”
An approach that is better suited to in-person assessment, but also works by video, is the Poppelreuter figure visual perceptual function test, which is a prompt for the patient that involves common household items overlaying one another “in atypical positions and atypical configurations” where the patient is instructed to describe the items they see on the card. Another approach that works over video is the interlocking finger test, where the patient is asked to copy the hand positions made by the clinician.
Dr. Ovsiew admitted that visuospatial function is nearly impossible to assess over the phone. Asking topographical questions (“If you’re driving from Chicago to Los Angeles, is the Pacific Ocean in front of you, behind you, to your left, or to your right?”) may help judge visuospatial function, but this relies on the patient having the topographic knowledge to answer the questions. Some patients who are topographically disoriented can’t do them at all,” Dr. Ovsiew said.
Bedside neuropsychiatry assesses encoding of a memory, its retention and its retrieval as well as verbal and visual cues. Each one of these aspects of memory can be impaired on its own and should be explored separately, Dr. Ovsiew explained. “Neuropsychiatric clinicians have a rough-and-ready, seat-of-the-pants way of approaching this that wouldn’t pass muster if you’re a psychologist, but is the best we can do at the bedside.”
To test retrieval and retention, the Three Words–Three Shapes test works well in person, with some difficulty by video, and is not possible to administer over the phone. In lieu of that test, giving the patient a simple word list and asking them to repeat the list in order. Using the word list, “these different stages of memory function can be parsed out pretty well at the bedside or chairside, and even by the phone. Figuring out where the memory failure is diagnostically important,” Dr. Ovsiew said.
Executive function, which involves activation, planning, sequencing, maintaining, self-monitoring, and flexible employment of action and attention, is “complicated to evaluate because there are multiple aspects of executive function, multiple deficits that can be seen with executive dysfunction, and they don’t all correlate with each other.”
Within executive function evaluation, the Mental Alternation Test can assess working memory, motor sequencing can be assessed through the ring/fist, fist/edge/palm, alternating fist, and rampart tests. The Go/No-Go test can be used to assess response inhibition. For effortful retrieval evaluation, spontaneous word-list generation – such as thinking of all the items one can buy at a supermarket– can test category fluency, while a task to name all the words starting with a certain letter can assess letter stimulus.
Executive function “is of crucial importance in the neuropsychiatric evaluation because it’s strongly correlated with how well the person functions outside the office,” Dr. Ovsiew said.
Global Academy and this news organization are owned by the same parent company. Dr. Ovsiew reported relationships with Wolters Kluwer Health in the form of consulting, receiving royalty payments, and related activities.
FROM FOCUS ON NEUROPSYCHIATRY 2020
COVID-19: Optimizing therapeutic strategies for children, adolescents with ADHD
Recently, the Yakima Health District (YHD), in collaboration with the Washington State Department of Health, issued dramatic revisions to its educational curriculum, opting for exclusively remote learning as an important next step in COVID-19 containment measures.
The newly implemented “enhanced” distance-learning paradigm has garnered considerable national attention. Even more noteworthy is how YHD addressed those with language barriers and learning differences such as ADHD as a “priority group”; these individuals are exempt from the newly implemented measures, and small instructional groups of no more than five “at-risk” students will be directly supervised by specialized educators.1,2 To overcome these new unprecedented challenges from the coronavirus pandemic, especially from the perspective of distance education and mental health for susceptible groups such as those with ADHD, it is of utmost importance to explore various programs of interest, as well as the targeted therapies being considered during this crisis.
From a therapeutic standpoint, individuals with learning differences are more likely to play catch-up with their age-matched peers. This puts them at significant risk for developmental delays with symptoms manifesting as disruptive behavioral issues. This is why ongoing parental guidance, coupled with a paradoxically stimulating environment, is critical for children and adolescents with ADHD.3 Accumulating evidence, based on a myriad of studies, demonstrates that childhood treatment with ADHD stimulants reduces the incidence of future substance use, as well as that of other negative outcomes.4,5
Therapeutic strategies that work
“The new normal” has forced unique challenges on clinicians for mitigating distress by novel means of health care delivery. Given the paucity of research exploring the interactions of individuals with ADHD within the context of COVID-19, Take for example, the suggested guidelines from the European ADHD Guidelines Group (EAGG) – such as the following:
- Telecommunications in general, and telepsychiatry in particular, should function as the primary mode of health care delivery to fulfill societal standards of physical distancing.
- Children and adolescents with ADHD should be designated as a “priority group” with respect to monitoring initiatives by educators in a school setting, be it virtual or otherwise.
- Implementation of behavioral strategies by parent or guardian to address psychological well-being and reduce the presence of comorbid behavioral conditions (such as oppositional defiant disorder).
In addition to the aforementioned guidance, EAGG maintains that individuals with ADHD may be initiated on medications after the completion of a baseline examination; if the patients in question are already on a treatment regimen, they should proceed with it as indicated. Interruptions to therapy are not ideal because patients are then subjected to health-related stressors of COVID-19. Reasonable regulations concerning access to medications, without unnecessary delays, undoubtedly will facilitate patient needs, allowing for a smooth transition in day-to-day activities. The family, as a cohesive unit, may benefit from reeducation because it contributes toward the therapeutic process. Neurofeedback, coping skills, and cognitive restructuring training are potential modalities that can augment medications.
Although it may seem counterintuitive, parents or caregivers should resist the urge to increase the medication dose during an outbreak with the intended goal of diminishing the psychosocial burden of ADHD symptomatology. Likewise, unless indicated by a specialist, antipsychotics and/or hypnotics should not be introduced for addressing behavioral dysregulation (such as agitation) during the confinement period.
Historically, numerous clinicians have suggested that patients undergo a routine cardiovascular examination and EKG before being prescribed psychostimulants (the rationale for this recommendation is that sympathomimetics unduly affect blood pressure and heart rate).6,7 However, the American Academy of Pediatrics (AAP) and the American Heart Association (AHA) eventually amended their previous stance by releasing a joint statement in which they deemed a baseline EKG necessary only in ADHD patients with preexisting cardiac risk. For all other patients, the use of EKGs was entirely contingent on physician discretion. However, given the nature of safety precautions for COVID-19, it is prudent to discourage or delay in-person cardiovascular examination/monitoring protocols altogether, especially in those patients without known heart conditions.
Another area of concern is sleep dysfunction, which might exist as an untoward effect of ADHD medication intake or because of the presence of COVID-19 psychosocial stressors. However, clinicians advise that unnecessary psychopharmacology (such as hypnotics or melatonin) be avoided. Instead, conservative lifestyle measures should be enacted, emphasizing the role of proper sleep hygiene in maintaining optimal behavioral health. Despite setbacks to in-person appointments, patients are expected to continue their pharmacotherapy with “parent-focused” ADHD interventions taking a primary role in facilitating compliance through remote monitoring.
ADMiRE, a tertiary-level, dedicated ADHD intervention program from South Dublin, Ireland, has identified several roadblocks with respect to streamlining health care for individuals with ADHD during the confinement period. The proposed resolution to these issues, some of which are derived from EAGG guidelines, might have universal applications elsewhere, thereby facilitating the development of therapeutic services of interest. ADMiRE has noted a correspondence between the guidelines established by EAGG and that of the Canadian ADHD Resource Alliance (CADDRA), including minimal in-person interactions (in favor of virtual teleconferencing) and a cardiovascular screen can be performed in lieu of baseline cardiac auscultation. Moreover, in the event that the patient is a low cardiac risk candidate for ADHD treatment, monitoring protocols may be continued from a home setting. However, if a physical examination is indicated, CADDRA recommends the use of precautionary PPE before commencing ADHD pharmacotherapy.
One of the most significant hurdles is that of school closures because teacher feedback for baseline behavior was traditionally instrumental in dictating patient medical management (for example, for titration schedule). It is expected that, for the time being, this role will be supplanted by parental reports. As well as disclosing information on behavioral dysregulation, family members should be trained to relay critical information about the development of stimulant-induced cardiovascular symptoms – namely, dyspnea, chest pain, and/or palpitations. Furthermore, as primary caregivers, parents should harbor a certain degree of emotional sensitivity because their mood state may influence the child’s overall behavioral course in terms of symptom exacerbation.8
Toward adopting an integrated model for care
Developing an effective assessment plan for patients with ADHD often proves to be a challenging task for clinicians, perhaps even more so in environments that enforce social distancing and limited physical contact by default. As a neurodevelopmental disorder from childhood, the symptoms (including inattention, hyperactivity, and/or impulsivity) of ADHD do not arise in a vacuum – comorbid conditions include mood and anxiety disorders, which are complicated further by a background risk for substance use and self-medicating tendencies.9 Unfortunately, the pandemic has limited the breadth of non-COVID doctors visits, which hinders the overall diagnostic and monitoring process for identifiable comorbid conditions, such as autism spectrum disorder, intellectual disability, oppositional defiant and conduct disorders, and so on.10 Since ADHD symptoms cannot be treated by pharmacotherapy or behavioral interventions alone, our team advocates that families provide additional emotional support and continuous encouragement during these uncertain times.
ADHD and the self-medication hypothesis
The Khantzian self-medication hypothesis posits that a drug seeker may subconsciously gravitate toward a particular agent only to discover a sense of relief concerning inner turmoil or restlessness after use. Observations support the notion that individuals with undiagnosed ADHD have sought cocaine or even recreational designer drugs (such as methylenedioxypyrovalerone, or “bath salts”).11 Given the similar mechanism of action between cocaine, methylenedioxypyrovalerone, and prescribed psychostimulants such as methylphenidate, the results are hardly surprising because these agents all work on the brain’s “reward center” (for example, the nucleus accumbens) by invoking dopamine release. Aside from the aforementioned self-medication hypothesis, “downers” such as Xanax recently have experienced a prescription spike during the outbreak. While there isn’t an immediate cause for concern of Xanax abuse in ADHD individuals, the potential for addiction is certainly real, especially when taking into account comorbid anxiety disorder or sleep dysfunction.
Because of limited resources and precautionary guidelines, clinicians are at a considerable disadvantage in terms of formulating a comprehensive diagnostic and treatment plan for children and adolescents with ADHD. This situation is further compounded by the recent closure of schools and the lack of feedback with respect to baseline behavior from teachers and specialized educators. This is why it is imperative for primary caregivers to closely monitor children with ADHD for developing changes in behavioral patterns (for example, mood or anxiety issues and drug-seeking or disruptive behavior) and work with health care professionals.
References
1. “Distance learning strongly recommended for all Yakima county schools.” NBC Right Now. 2020 Aug 5.
2. Retka J. “Enhanced” remote learning in Yakima county schools? What that means for students this fall. Yakima Herald-Republic. 2020 Aug 8.
3. Armstrong T. “To empower! Not Control! A holistic approach to ADHD.” American Institute for Learning and Development. 1998.
4. J Child Psychol Psychiatry. 2014 Aug;55(8):878-85.
5. Ir J Psychol Med. 2020 May 21:1-22.
6. Lancet Child Adolesc Health. 2020 Jun;4(6):412-4.
7. O’Keefe L. AAP News. 2008 Jun;29(6):1.
8. Asian J Psychiatr. 2020 Jun;51:102077.
9. Current Psychiatry. 2015 Dec;14(12):e3-4.
10. Encephale. 2020 Jun 7;46(3S):S85-92.
11. Current Psychiatry. 2014 Dec; 3(12): e3-4.
Dr. Islam is a medical adviser for the International Maternal and Child Health Foundation (IMCHF), Montreal, and is based in New York. He also is a postdoctoral fellow, psychopharmacologist, and a board-certified medical affairs specialist. Dr. Islam disclosed no relevant financial relationships. Zaid Ulhaq Choudhry is a research assistant at the IMCHF. He has no disclosures. Dr. Zia Choudhry is the chief scientific officer and head of the department of mental health and clini-cal research at the IMCHF and is Mr. Choudhry’s father. He has no disclosures.
Recently, the Yakima Health District (YHD), in collaboration with the Washington State Department of Health, issued dramatic revisions to its educational curriculum, opting for exclusively remote learning as an important next step in COVID-19 containment measures.
The newly implemented “enhanced” distance-learning paradigm has garnered considerable national attention. Even more noteworthy is how YHD addressed those with language barriers and learning differences such as ADHD as a “priority group”; these individuals are exempt from the newly implemented measures, and small instructional groups of no more than five “at-risk” students will be directly supervised by specialized educators.1,2 To overcome these new unprecedented challenges from the coronavirus pandemic, especially from the perspective of distance education and mental health for susceptible groups such as those with ADHD, it is of utmost importance to explore various programs of interest, as well as the targeted therapies being considered during this crisis.
From a therapeutic standpoint, individuals with learning differences are more likely to play catch-up with their age-matched peers. This puts them at significant risk for developmental delays with symptoms manifesting as disruptive behavioral issues. This is why ongoing parental guidance, coupled with a paradoxically stimulating environment, is critical for children and adolescents with ADHD.3 Accumulating evidence, based on a myriad of studies, demonstrates that childhood treatment with ADHD stimulants reduces the incidence of future substance use, as well as that of other negative outcomes.4,5
Therapeutic strategies that work
“The new normal” has forced unique challenges on clinicians for mitigating distress by novel means of health care delivery. Given the paucity of research exploring the interactions of individuals with ADHD within the context of COVID-19, Take for example, the suggested guidelines from the European ADHD Guidelines Group (EAGG) – such as the following:
- Telecommunications in general, and telepsychiatry in particular, should function as the primary mode of health care delivery to fulfill societal standards of physical distancing.
- Children and adolescents with ADHD should be designated as a “priority group” with respect to monitoring initiatives by educators in a school setting, be it virtual or otherwise.
- Implementation of behavioral strategies by parent or guardian to address psychological well-being and reduce the presence of comorbid behavioral conditions (such as oppositional defiant disorder).
In addition to the aforementioned guidance, EAGG maintains that individuals with ADHD may be initiated on medications after the completion of a baseline examination; if the patients in question are already on a treatment regimen, they should proceed with it as indicated. Interruptions to therapy are not ideal because patients are then subjected to health-related stressors of COVID-19. Reasonable regulations concerning access to medications, without unnecessary delays, undoubtedly will facilitate patient needs, allowing for a smooth transition in day-to-day activities. The family, as a cohesive unit, may benefit from reeducation because it contributes toward the therapeutic process. Neurofeedback, coping skills, and cognitive restructuring training are potential modalities that can augment medications.
Although it may seem counterintuitive, parents or caregivers should resist the urge to increase the medication dose during an outbreak with the intended goal of diminishing the psychosocial burden of ADHD symptomatology. Likewise, unless indicated by a specialist, antipsychotics and/or hypnotics should not be introduced for addressing behavioral dysregulation (such as agitation) during the confinement period.
Historically, numerous clinicians have suggested that patients undergo a routine cardiovascular examination and EKG before being prescribed psychostimulants (the rationale for this recommendation is that sympathomimetics unduly affect blood pressure and heart rate).6,7 However, the American Academy of Pediatrics (AAP) and the American Heart Association (AHA) eventually amended their previous stance by releasing a joint statement in which they deemed a baseline EKG necessary only in ADHD patients with preexisting cardiac risk. For all other patients, the use of EKGs was entirely contingent on physician discretion. However, given the nature of safety precautions for COVID-19, it is prudent to discourage or delay in-person cardiovascular examination/monitoring protocols altogether, especially in those patients without known heart conditions.
Another area of concern is sleep dysfunction, which might exist as an untoward effect of ADHD medication intake or because of the presence of COVID-19 psychosocial stressors. However, clinicians advise that unnecessary psychopharmacology (such as hypnotics or melatonin) be avoided. Instead, conservative lifestyle measures should be enacted, emphasizing the role of proper sleep hygiene in maintaining optimal behavioral health. Despite setbacks to in-person appointments, patients are expected to continue their pharmacotherapy with “parent-focused” ADHD interventions taking a primary role in facilitating compliance through remote monitoring.
ADMiRE, a tertiary-level, dedicated ADHD intervention program from South Dublin, Ireland, has identified several roadblocks with respect to streamlining health care for individuals with ADHD during the confinement period. The proposed resolution to these issues, some of which are derived from EAGG guidelines, might have universal applications elsewhere, thereby facilitating the development of therapeutic services of interest. ADMiRE has noted a correspondence between the guidelines established by EAGG and that of the Canadian ADHD Resource Alliance (CADDRA), including minimal in-person interactions (in favor of virtual teleconferencing) and a cardiovascular screen can be performed in lieu of baseline cardiac auscultation. Moreover, in the event that the patient is a low cardiac risk candidate for ADHD treatment, monitoring protocols may be continued from a home setting. However, if a physical examination is indicated, CADDRA recommends the use of precautionary PPE before commencing ADHD pharmacotherapy.
One of the most significant hurdles is that of school closures because teacher feedback for baseline behavior was traditionally instrumental in dictating patient medical management (for example, for titration schedule). It is expected that, for the time being, this role will be supplanted by parental reports. As well as disclosing information on behavioral dysregulation, family members should be trained to relay critical information about the development of stimulant-induced cardiovascular symptoms – namely, dyspnea, chest pain, and/or palpitations. Furthermore, as primary caregivers, parents should harbor a certain degree of emotional sensitivity because their mood state may influence the child’s overall behavioral course in terms of symptom exacerbation.8
Toward adopting an integrated model for care
Developing an effective assessment plan for patients with ADHD often proves to be a challenging task for clinicians, perhaps even more so in environments that enforce social distancing and limited physical contact by default. As a neurodevelopmental disorder from childhood, the symptoms (including inattention, hyperactivity, and/or impulsivity) of ADHD do not arise in a vacuum – comorbid conditions include mood and anxiety disorders, which are complicated further by a background risk for substance use and self-medicating tendencies.9 Unfortunately, the pandemic has limited the breadth of non-COVID doctors visits, which hinders the overall diagnostic and monitoring process for identifiable comorbid conditions, such as autism spectrum disorder, intellectual disability, oppositional defiant and conduct disorders, and so on.10 Since ADHD symptoms cannot be treated by pharmacotherapy or behavioral interventions alone, our team advocates that families provide additional emotional support and continuous encouragement during these uncertain times.
ADHD and the self-medication hypothesis
The Khantzian self-medication hypothesis posits that a drug seeker may subconsciously gravitate toward a particular agent only to discover a sense of relief concerning inner turmoil or restlessness after use. Observations support the notion that individuals with undiagnosed ADHD have sought cocaine or even recreational designer drugs (such as methylenedioxypyrovalerone, or “bath salts”).11 Given the similar mechanism of action between cocaine, methylenedioxypyrovalerone, and prescribed psychostimulants such as methylphenidate, the results are hardly surprising because these agents all work on the brain’s “reward center” (for example, the nucleus accumbens) by invoking dopamine release. Aside from the aforementioned self-medication hypothesis, “downers” such as Xanax recently have experienced a prescription spike during the outbreak. While there isn’t an immediate cause for concern of Xanax abuse in ADHD individuals, the potential for addiction is certainly real, especially when taking into account comorbid anxiety disorder or sleep dysfunction.
Because of limited resources and precautionary guidelines, clinicians are at a considerable disadvantage in terms of formulating a comprehensive diagnostic and treatment plan for children and adolescents with ADHD. This situation is further compounded by the recent closure of schools and the lack of feedback with respect to baseline behavior from teachers and specialized educators. This is why it is imperative for primary caregivers to closely monitor children with ADHD for developing changes in behavioral patterns (for example, mood or anxiety issues and drug-seeking or disruptive behavior) and work with health care professionals.
References
1. “Distance learning strongly recommended for all Yakima county schools.” NBC Right Now. 2020 Aug 5.
2. Retka J. “Enhanced” remote learning in Yakima county schools? What that means for students this fall. Yakima Herald-Republic. 2020 Aug 8.
3. Armstrong T. “To empower! Not Control! A holistic approach to ADHD.” American Institute for Learning and Development. 1998.
4. J Child Psychol Psychiatry. 2014 Aug;55(8):878-85.
5. Ir J Psychol Med. 2020 May 21:1-22.
6. Lancet Child Adolesc Health. 2020 Jun;4(6):412-4.
7. O’Keefe L. AAP News. 2008 Jun;29(6):1.
8. Asian J Psychiatr. 2020 Jun;51:102077.
9. Current Psychiatry. 2015 Dec;14(12):e3-4.
10. Encephale. 2020 Jun 7;46(3S):S85-92.
11. Current Psychiatry. 2014 Dec; 3(12): e3-4.
Dr. Islam is a medical adviser for the International Maternal and Child Health Foundation (IMCHF), Montreal, and is based in New York. He also is a postdoctoral fellow, psychopharmacologist, and a board-certified medical affairs specialist. Dr. Islam disclosed no relevant financial relationships. Zaid Ulhaq Choudhry is a research assistant at the IMCHF. He has no disclosures. Dr. Zia Choudhry is the chief scientific officer and head of the department of mental health and clini-cal research at the IMCHF and is Mr. Choudhry’s father. He has no disclosures.
Recently, the Yakima Health District (YHD), in collaboration with the Washington State Department of Health, issued dramatic revisions to its educational curriculum, opting for exclusively remote learning as an important next step in COVID-19 containment measures.
The newly implemented “enhanced” distance-learning paradigm has garnered considerable national attention. Even more noteworthy is how YHD addressed those with language barriers and learning differences such as ADHD as a “priority group”; these individuals are exempt from the newly implemented measures, and small instructional groups of no more than five “at-risk” students will be directly supervised by specialized educators.1,2 To overcome these new unprecedented challenges from the coronavirus pandemic, especially from the perspective of distance education and mental health for susceptible groups such as those with ADHD, it is of utmost importance to explore various programs of interest, as well as the targeted therapies being considered during this crisis.
From a therapeutic standpoint, individuals with learning differences are more likely to play catch-up with their age-matched peers. This puts them at significant risk for developmental delays with symptoms manifesting as disruptive behavioral issues. This is why ongoing parental guidance, coupled with a paradoxically stimulating environment, is critical for children and adolescents with ADHD.3 Accumulating evidence, based on a myriad of studies, demonstrates that childhood treatment with ADHD stimulants reduces the incidence of future substance use, as well as that of other negative outcomes.4,5
Therapeutic strategies that work
“The new normal” has forced unique challenges on clinicians for mitigating distress by novel means of health care delivery. Given the paucity of research exploring the interactions of individuals with ADHD within the context of COVID-19, Take for example, the suggested guidelines from the European ADHD Guidelines Group (EAGG) – such as the following:
- Telecommunications in general, and telepsychiatry in particular, should function as the primary mode of health care delivery to fulfill societal standards of physical distancing.
- Children and adolescents with ADHD should be designated as a “priority group” with respect to monitoring initiatives by educators in a school setting, be it virtual or otherwise.
- Implementation of behavioral strategies by parent or guardian to address psychological well-being and reduce the presence of comorbid behavioral conditions (such as oppositional defiant disorder).
In addition to the aforementioned guidance, EAGG maintains that individuals with ADHD may be initiated on medications after the completion of a baseline examination; if the patients in question are already on a treatment regimen, they should proceed with it as indicated. Interruptions to therapy are not ideal because patients are then subjected to health-related stressors of COVID-19. Reasonable regulations concerning access to medications, without unnecessary delays, undoubtedly will facilitate patient needs, allowing for a smooth transition in day-to-day activities. The family, as a cohesive unit, may benefit from reeducation because it contributes toward the therapeutic process. Neurofeedback, coping skills, and cognitive restructuring training are potential modalities that can augment medications.
Although it may seem counterintuitive, parents or caregivers should resist the urge to increase the medication dose during an outbreak with the intended goal of diminishing the psychosocial burden of ADHD symptomatology. Likewise, unless indicated by a specialist, antipsychotics and/or hypnotics should not be introduced for addressing behavioral dysregulation (such as agitation) during the confinement period.
Historically, numerous clinicians have suggested that patients undergo a routine cardiovascular examination and EKG before being prescribed psychostimulants (the rationale for this recommendation is that sympathomimetics unduly affect blood pressure and heart rate).6,7 However, the American Academy of Pediatrics (AAP) and the American Heart Association (AHA) eventually amended their previous stance by releasing a joint statement in which they deemed a baseline EKG necessary only in ADHD patients with preexisting cardiac risk. For all other patients, the use of EKGs was entirely contingent on physician discretion. However, given the nature of safety precautions for COVID-19, it is prudent to discourage or delay in-person cardiovascular examination/monitoring protocols altogether, especially in those patients without known heart conditions.
Another area of concern is sleep dysfunction, which might exist as an untoward effect of ADHD medication intake or because of the presence of COVID-19 psychosocial stressors. However, clinicians advise that unnecessary psychopharmacology (such as hypnotics or melatonin) be avoided. Instead, conservative lifestyle measures should be enacted, emphasizing the role of proper sleep hygiene in maintaining optimal behavioral health. Despite setbacks to in-person appointments, patients are expected to continue their pharmacotherapy with “parent-focused” ADHD interventions taking a primary role in facilitating compliance through remote monitoring.
ADMiRE, a tertiary-level, dedicated ADHD intervention program from South Dublin, Ireland, has identified several roadblocks with respect to streamlining health care for individuals with ADHD during the confinement period. The proposed resolution to these issues, some of which are derived from EAGG guidelines, might have universal applications elsewhere, thereby facilitating the development of therapeutic services of interest. ADMiRE has noted a correspondence between the guidelines established by EAGG and that of the Canadian ADHD Resource Alliance (CADDRA), including minimal in-person interactions (in favor of virtual teleconferencing) and a cardiovascular screen can be performed in lieu of baseline cardiac auscultation. Moreover, in the event that the patient is a low cardiac risk candidate for ADHD treatment, monitoring protocols may be continued from a home setting. However, if a physical examination is indicated, CADDRA recommends the use of precautionary PPE before commencing ADHD pharmacotherapy.
One of the most significant hurdles is that of school closures because teacher feedback for baseline behavior was traditionally instrumental in dictating patient medical management (for example, for titration schedule). It is expected that, for the time being, this role will be supplanted by parental reports. As well as disclosing information on behavioral dysregulation, family members should be trained to relay critical information about the development of stimulant-induced cardiovascular symptoms – namely, dyspnea, chest pain, and/or palpitations. Furthermore, as primary caregivers, parents should harbor a certain degree of emotional sensitivity because their mood state may influence the child’s overall behavioral course in terms of symptom exacerbation.8
Toward adopting an integrated model for care
Developing an effective assessment plan for patients with ADHD often proves to be a challenging task for clinicians, perhaps even more so in environments that enforce social distancing and limited physical contact by default. As a neurodevelopmental disorder from childhood, the symptoms (including inattention, hyperactivity, and/or impulsivity) of ADHD do not arise in a vacuum – comorbid conditions include mood and anxiety disorders, which are complicated further by a background risk for substance use and self-medicating tendencies.9 Unfortunately, the pandemic has limited the breadth of non-COVID doctors visits, which hinders the overall diagnostic and monitoring process for identifiable comorbid conditions, such as autism spectrum disorder, intellectual disability, oppositional defiant and conduct disorders, and so on.10 Since ADHD symptoms cannot be treated by pharmacotherapy or behavioral interventions alone, our team advocates that families provide additional emotional support and continuous encouragement during these uncertain times.
ADHD and the self-medication hypothesis
The Khantzian self-medication hypothesis posits that a drug seeker may subconsciously gravitate toward a particular agent only to discover a sense of relief concerning inner turmoil or restlessness after use. Observations support the notion that individuals with undiagnosed ADHD have sought cocaine or even recreational designer drugs (such as methylenedioxypyrovalerone, or “bath salts”).11 Given the similar mechanism of action between cocaine, methylenedioxypyrovalerone, and prescribed psychostimulants such as methylphenidate, the results are hardly surprising because these agents all work on the brain’s “reward center” (for example, the nucleus accumbens) by invoking dopamine release. Aside from the aforementioned self-medication hypothesis, “downers” such as Xanax recently have experienced a prescription spike during the outbreak. While there isn’t an immediate cause for concern of Xanax abuse in ADHD individuals, the potential for addiction is certainly real, especially when taking into account comorbid anxiety disorder or sleep dysfunction.
Because of limited resources and precautionary guidelines, clinicians are at a considerable disadvantage in terms of formulating a comprehensive diagnostic and treatment plan for children and adolescents with ADHD. This situation is further compounded by the recent closure of schools and the lack of feedback with respect to baseline behavior from teachers and specialized educators. This is why it is imperative for primary caregivers to closely monitor children with ADHD for developing changes in behavioral patterns (for example, mood or anxiety issues and drug-seeking or disruptive behavior) and work with health care professionals.
References
1. “Distance learning strongly recommended for all Yakima county schools.” NBC Right Now. 2020 Aug 5.
2. Retka J. “Enhanced” remote learning in Yakima county schools? What that means for students this fall. Yakima Herald-Republic. 2020 Aug 8.
3. Armstrong T. “To empower! Not Control! A holistic approach to ADHD.” American Institute for Learning and Development. 1998.
4. J Child Psychol Psychiatry. 2014 Aug;55(8):878-85.
5. Ir J Psychol Med. 2020 May 21:1-22.
6. Lancet Child Adolesc Health. 2020 Jun;4(6):412-4.
7. O’Keefe L. AAP News. 2008 Jun;29(6):1.
8. Asian J Psychiatr. 2020 Jun;51:102077.
9. Current Psychiatry. 2015 Dec;14(12):e3-4.
10. Encephale. 2020 Jun 7;46(3S):S85-92.
11. Current Psychiatry. 2014 Dec; 3(12): e3-4.
Dr. Islam is a medical adviser for the International Maternal and Child Health Foundation (IMCHF), Montreal, and is based in New York. He also is a postdoctoral fellow, psychopharmacologist, and a board-certified medical affairs specialist. Dr. Islam disclosed no relevant financial relationships. Zaid Ulhaq Choudhry is a research assistant at the IMCHF. He has no disclosures. Dr. Zia Choudhry is the chief scientific officer and head of the department of mental health and clini-cal research at the IMCHF and is Mr. Choudhry’s father. He has no disclosures.
Frequent cannabis use in depression tripled over past decade
Not only are individuals with depression at significantly higher risk for cannabis use, compared with those without depression, this trend has increased dramatically over the last decade, new research shows.
Investigators analyzed data from more than 16,000 U.S. adults between the ages of 20 and 59 years and found that those with depression had almost twice the odds of any past-month cannabis use compared with those without depression. Odds rose from 1.5 in the 2005-2006 period to 2.3 in the 2015-2016 period.
Moreover, the odds ratio for daily or near-daily use almost tripled for those with versus without depression between the two periods.
“Clinicians should screen their depressed patients for cannabis use, since this is becoming more common and could actually make their depressive symptoms worse rather than better,” senior author Deborah Hasin, PhD, professor of epidemiology, Columbia University Irving Medical Center, New York City, told Medscape Medical News.
The results were published online August 18 in JAMA Network Open.
Misleading advertising
“Cannabis use is increasing in the U.S. and the potency of cannabis products is increasing as well,” Dr. Hasin said.
“Misleading media information and advertising suggests that cannabis is a good treatment for depression, although studies show that cannabis use may actually worsen depression symptoms, [so] we were interested in whether U.S. adults were increasingly likely to be cannabis users if they were depressed,” she reported.
To investigate, the researchers assessed data from the National Health and Nutrition Examination Survey (NHANES), with a final study sample consisting of 16,216 U.S. adults. The mean age was 39.12 years, 48.9% were men, 66.4% were non-Hispanic White, 65.6% had at least some college education, and 62.4% had an annual family income of less than $75,000.
Of these participants, 7.5% had “probable depression,” based on the Patient Health Questionnaire–9, the investigators report.
Past-month cannabis use was defined as using cannabis at least once during the past 20 days. Daily or near-daily past-month use was defined as using cannabis at least 20 times in the past 30 days.
Covariates included age, gender, race, education, marital status, annual family income, and past-year use of other substances, such as alcohol, heroin, and methamphetamine.
The researchers note that because the NHANES data were divided into six survey years (2005-2006, 2007-2008, 2009-2010, 2011-2012, 2013-2014, and 2015-2016), their analysis was based on a “new sample weight” that combined the datasets.
Especially pronounced
Results showed that the prevalence of any past-month cannabis use in the overall sample group increased from 12.2% in the 2005-2006 period to 17.3% in the 2015-2016 period (P < .001).
The investigators characterized this change as “significant,” adding that the estimated odds of cannabis use increased by approximately 9% between every 2-year time period.
The change was even more dramatic when the increase was examined across survey time periods (OR, 1.12; P < .001). The estimated odds of daily or near-daily use increased by approximately 12% between every 2-year period.
Interestingly, however, there were no significant changes in odds for depression when consecutive survey years were compared.
When the researchers specifically focused on the association between any past-month cannabis use and depression versus no depression, they found an adjusted OR of 1.90 (95% CI, 1.62-2.12; P < .001).
Individuals with depression also had 2.29 (95% CI, 1.80-2.92) times the odds for daily or near-daily cannabis use, compared with those without depression.
A post-hoc analysis looked at time trends in a sample group that included those missing information on at least one covariate (n = 17,724 participants). It showed similar results to those in the final sample that included no missing data.
People with depression have increased risk of using “most substances that can be abused,” Dr. Hasin said. “However, with the overall rates of cannabis use increasing in the general population, this is becoming especially pronounced for cannabis.”
Clear implications
Commenting on the findings for Medscape Medical News, Deepak D’Souza, MD, professor of psychiatry, Yale University, New Haven, Conn., said there is “concern about the unsubstantiated claims of cannabis having a beneficial effect in psychiatric disorders, the most common being depression.”
Dr. D’Souza, who was not involved with the study, called it “yet another piece of evidence suggesting that over the period of time during which cannabis laws have been liberalized, rates of past-month and daily cannabis use have increased, whereas rates of other substances, including alcohol, have remained stable.”
He suggested that a common limitation of epidemiological studies is that it is difficult to tell the direction of the association, “and it could be bidirectional.”
Nevertheless, there are clear implications for the practicing clinician, he added.
“If people have a history of depression, one should ask patients about the use of cannabis and also remind them about potential psychiatric negative effects of use,” Dr. D’Souza noted.
For the general public, “the point is that there is no good evidence to support cannabis use in depression treatment and, in fact, people with depression might be more likely to use it in problematic way,” he said.
Dr. Hasin agreed that it is “certainly possible that the relationship between cannabis use and depression is bidirectional, but the mechanism of this association requires more study.”
The study was supported by a grant from the National Institute on Drug Abuse to Dr. Hasin and by the New York State Psychiatric Institute. The study authors and Dr. D’Souza disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Not only are individuals with depression at significantly higher risk for cannabis use, compared with those without depression, this trend has increased dramatically over the last decade, new research shows.
Investigators analyzed data from more than 16,000 U.S. adults between the ages of 20 and 59 years and found that those with depression had almost twice the odds of any past-month cannabis use compared with those without depression. Odds rose from 1.5 in the 2005-2006 period to 2.3 in the 2015-2016 period.
Moreover, the odds ratio for daily or near-daily use almost tripled for those with versus without depression between the two periods.
“Clinicians should screen their depressed patients for cannabis use, since this is becoming more common and could actually make their depressive symptoms worse rather than better,” senior author Deborah Hasin, PhD, professor of epidemiology, Columbia University Irving Medical Center, New York City, told Medscape Medical News.
The results were published online August 18 in JAMA Network Open.
Misleading advertising
“Cannabis use is increasing in the U.S. and the potency of cannabis products is increasing as well,” Dr. Hasin said.
“Misleading media information and advertising suggests that cannabis is a good treatment for depression, although studies show that cannabis use may actually worsen depression symptoms, [so] we were interested in whether U.S. adults were increasingly likely to be cannabis users if they were depressed,” she reported.
To investigate, the researchers assessed data from the National Health and Nutrition Examination Survey (NHANES), with a final study sample consisting of 16,216 U.S. adults. The mean age was 39.12 years, 48.9% were men, 66.4% were non-Hispanic White, 65.6% had at least some college education, and 62.4% had an annual family income of less than $75,000.
Of these participants, 7.5% had “probable depression,” based on the Patient Health Questionnaire–9, the investigators report.
Past-month cannabis use was defined as using cannabis at least once during the past 20 days. Daily or near-daily past-month use was defined as using cannabis at least 20 times in the past 30 days.
Covariates included age, gender, race, education, marital status, annual family income, and past-year use of other substances, such as alcohol, heroin, and methamphetamine.
The researchers note that because the NHANES data were divided into six survey years (2005-2006, 2007-2008, 2009-2010, 2011-2012, 2013-2014, and 2015-2016), their analysis was based on a “new sample weight” that combined the datasets.
Especially pronounced
Results showed that the prevalence of any past-month cannabis use in the overall sample group increased from 12.2% in the 2005-2006 period to 17.3% in the 2015-2016 period (P < .001).
The investigators characterized this change as “significant,” adding that the estimated odds of cannabis use increased by approximately 9% between every 2-year time period.
The change was even more dramatic when the increase was examined across survey time periods (OR, 1.12; P < .001). The estimated odds of daily or near-daily use increased by approximately 12% between every 2-year period.
Interestingly, however, there were no significant changes in odds for depression when consecutive survey years were compared.
When the researchers specifically focused on the association between any past-month cannabis use and depression versus no depression, they found an adjusted OR of 1.90 (95% CI, 1.62-2.12; P < .001).
Individuals with depression also had 2.29 (95% CI, 1.80-2.92) times the odds for daily or near-daily cannabis use, compared with those without depression.
A post-hoc analysis looked at time trends in a sample group that included those missing information on at least one covariate (n = 17,724 participants). It showed similar results to those in the final sample that included no missing data.
People with depression have increased risk of using “most substances that can be abused,” Dr. Hasin said. “However, with the overall rates of cannabis use increasing in the general population, this is becoming especially pronounced for cannabis.”
Clear implications
Commenting on the findings for Medscape Medical News, Deepak D’Souza, MD, professor of psychiatry, Yale University, New Haven, Conn., said there is “concern about the unsubstantiated claims of cannabis having a beneficial effect in psychiatric disorders, the most common being depression.”
Dr. D’Souza, who was not involved with the study, called it “yet another piece of evidence suggesting that over the period of time during which cannabis laws have been liberalized, rates of past-month and daily cannabis use have increased, whereas rates of other substances, including alcohol, have remained stable.”
He suggested that a common limitation of epidemiological studies is that it is difficult to tell the direction of the association, “and it could be bidirectional.”
Nevertheless, there are clear implications for the practicing clinician, he added.
“If people have a history of depression, one should ask patients about the use of cannabis and also remind them about potential psychiatric negative effects of use,” Dr. D’Souza noted.
For the general public, “the point is that there is no good evidence to support cannabis use in depression treatment and, in fact, people with depression might be more likely to use it in problematic way,” he said.
Dr. Hasin agreed that it is “certainly possible that the relationship between cannabis use and depression is bidirectional, but the mechanism of this association requires more study.”
The study was supported by a grant from the National Institute on Drug Abuse to Dr. Hasin and by the New York State Psychiatric Institute. The study authors and Dr. D’Souza disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Not only are individuals with depression at significantly higher risk for cannabis use, compared with those without depression, this trend has increased dramatically over the last decade, new research shows.
Investigators analyzed data from more than 16,000 U.S. adults between the ages of 20 and 59 years and found that those with depression had almost twice the odds of any past-month cannabis use compared with those without depression. Odds rose from 1.5 in the 2005-2006 period to 2.3 in the 2015-2016 period.
Moreover, the odds ratio for daily or near-daily use almost tripled for those with versus without depression between the two periods.
“Clinicians should screen their depressed patients for cannabis use, since this is becoming more common and could actually make their depressive symptoms worse rather than better,” senior author Deborah Hasin, PhD, professor of epidemiology, Columbia University Irving Medical Center, New York City, told Medscape Medical News.
The results were published online August 18 in JAMA Network Open.
Misleading advertising
“Cannabis use is increasing in the U.S. and the potency of cannabis products is increasing as well,” Dr. Hasin said.
“Misleading media information and advertising suggests that cannabis is a good treatment for depression, although studies show that cannabis use may actually worsen depression symptoms, [so] we were interested in whether U.S. adults were increasingly likely to be cannabis users if they were depressed,” she reported.
To investigate, the researchers assessed data from the National Health and Nutrition Examination Survey (NHANES), with a final study sample consisting of 16,216 U.S. adults. The mean age was 39.12 years, 48.9% were men, 66.4% were non-Hispanic White, 65.6% had at least some college education, and 62.4% had an annual family income of less than $75,000.
Of these participants, 7.5% had “probable depression,” based on the Patient Health Questionnaire–9, the investigators report.
Past-month cannabis use was defined as using cannabis at least once during the past 20 days. Daily or near-daily past-month use was defined as using cannabis at least 20 times in the past 30 days.
Covariates included age, gender, race, education, marital status, annual family income, and past-year use of other substances, such as alcohol, heroin, and methamphetamine.
The researchers note that because the NHANES data were divided into six survey years (2005-2006, 2007-2008, 2009-2010, 2011-2012, 2013-2014, and 2015-2016), their analysis was based on a “new sample weight” that combined the datasets.
Especially pronounced
Results showed that the prevalence of any past-month cannabis use in the overall sample group increased from 12.2% in the 2005-2006 period to 17.3% in the 2015-2016 period (P < .001).
The investigators characterized this change as “significant,” adding that the estimated odds of cannabis use increased by approximately 9% between every 2-year time period.
The change was even more dramatic when the increase was examined across survey time periods (OR, 1.12; P < .001). The estimated odds of daily or near-daily use increased by approximately 12% between every 2-year period.
Interestingly, however, there were no significant changes in odds for depression when consecutive survey years were compared.
When the researchers specifically focused on the association between any past-month cannabis use and depression versus no depression, they found an adjusted OR of 1.90 (95% CI, 1.62-2.12; P < .001).
Individuals with depression also had 2.29 (95% CI, 1.80-2.92) times the odds for daily or near-daily cannabis use, compared with those without depression.
A post-hoc analysis looked at time trends in a sample group that included those missing information on at least one covariate (n = 17,724 participants). It showed similar results to those in the final sample that included no missing data.
People with depression have increased risk of using “most substances that can be abused,” Dr. Hasin said. “However, with the overall rates of cannabis use increasing in the general population, this is becoming especially pronounced for cannabis.”
Clear implications
Commenting on the findings for Medscape Medical News, Deepak D’Souza, MD, professor of psychiatry, Yale University, New Haven, Conn., said there is “concern about the unsubstantiated claims of cannabis having a beneficial effect in psychiatric disorders, the most common being depression.”
Dr. D’Souza, who was not involved with the study, called it “yet another piece of evidence suggesting that over the period of time during which cannabis laws have been liberalized, rates of past-month and daily cannabis use have increased, whereas rates of other substances, including alcohol, have remained stable.”
He suggested that a common limitation of epidemiological studies is that it is difficult to tell the direction of the association, “and it could be bidirectional.”
Nevertheless, there are clear implications for the practicing clinician, he added.
“If people have a history of depression, one should ask patients about the use of cannabis and also remind them about potential psychiatric negative effects of use,” Dr. D’Souza noted.
For the general public, “the point is that there is no good evidence to support cannabis use in depression treatment and, in fact, people with depression might be more likely to use it in problematic way,” he said.
Dr. Hasin agreed that it is “certainly possible that the relationship between cannabis use and depression is bidirectional, but the mechanism of this association requires more study.”
The study was supported by a grant from the National Institute on Drug Abuse to Dr. Hasin and by the New York State Psychiatric Institute. The study authors and Dr. D’Souza disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
The transitions of COVID-19
When I was preparing for the recent birth of my baby, I anticipated a period of transition for myself. As a reproductive psychiatrist, I have treated many women during the perinatal and postpartum periods, and have a unique appreciation for the life changes that accompany birth. What I did not expect, however, was the world transitioning with me.
“The new normal” is an economic phrase that describes the COVID-19 era. The pandemic has engendered economic instability, collapsed industries, challenged health care systems, and has led to many deaths worldwide. The COVID-19 pandemic also has been associated with overall increases in anxiety and depression.1 Emerging research suggests that frontline medical workers are especially at risk for developing psychological distress.2
COVID-19 has also created immense challenges for families. Because of concern for the spread of the virus, schools have been suspended, older grandparents isolated, and many parents continue to work remotely. For families in psychiatric care, this time has also been a time of change. Telepsychiatry might be more accessible, but the transition has been an adjustment for patients and clinicians.
As psychiatrists, how do we best treat families during this time? What are some ways to support our psychiatric colleagues? How do we ensure our own emotional well-being amid the tremendous changes occurring around us?
Background of interpersonal psychotherapy
Interpersonal psychotherapy (IPT) is a form of psychotherapy designed to treat depression following periods of transition. Its main goals include improving interpersonal connection and reducing psychological distress. Originally developed in the 1970s by Gerald Klerman, MD; Myrna Weissman, PhD; and Eugene Paykel, MD, IPT is a structured, time-limited form of psychotherapy.3
Conceptualizing depression as a treatable illness, Pim Cuijpers, PhD, and associates summarized the division of IPT into three phases.4 The initial phase involves history taking, forming an alliance, and choosing an interpersonal focus for treatment. The middle phase focuses on applying interpersonal problem-specific therapeutic techniques. The concluding phase of treatment involves consolidation of gains as well as formulating contingency plans for relapse of symptoms. Over the course of treatment, an IPT clinician focuses on life transitions and emphasizes that isolation and antagonistic relationships increase an individual’s vulnerability for a depressive episode.3
Randomized, controlled trials support IPT’s efficacy as a treatment for depression. Research also suggests it can possibly prevent the development of depression.4 Although IPT initially was designed as an individual form of psychotherapy, it has been adapted to both family and group contexts.5,6 IPT is also an empirically valid form of psychotherapy for postpartum depression.7
Interpersonal psychotherapy for families
Given IPT’s role for treating depression following times of transition, clinicians should consider adapting interpersonal psychotherapy to family treatment during this time. Addressing social isolation, managing complex family relationships, and monitoring the family’s overall emotional health should be prioritized. Families under quarantine or who are grieving the death of family members may especially benefit from improved interpersonal connection. Consistent with the IPT model, contingency plans for the family should also be explored to prepare for potential future waves of the pandemic.
In addition to supporting and strengthening families, psychiatrists can use IPT themes to identify positive changes for families tied to COVID-19. Despite its difficulties, the stay-at-home order provided some families a unique chance to slow down and adapt a more relaxed routine. Busy families were suddenly given the opportunity to spend more time with one another. Although many older grandparents were isolated, creative uses of technology provided a chance for grandparents to remain an integral part of family life. Psychiatrists can assist families in transitioning back to previous schedules, while also exploring ways to incorporate the positive changes gained during this time.
Interpersonal psychotherapy for psychiatrists
An interpersonal focus could also be helpful for clinicians to adapt to changes in psychiatric practice. Many clinicians have been thrust into telepsychiatry practice, some with little to no preparation. Because of the trauma associated with frontline work, some psychiatrists have expanded their patient panel to treat physician colleagues. For consult-liaison psychiatrists, the possible neuropsychiatric effects of COVID-19 are new symptoms to consider when evaluating patients in a medical hospital setting.8 Fundamentally, modern day psychiatrists have never encountered a pandemic nor attempted to treat its psychological implications. Prioritizing seeking support from colleagues and caring for one’s personal relationships are helpful tools for clinicians to maintain their own emotional health during this challenging period.
Personal reflection
When I reflect on my baby’s recent birth, I recognize the importance of interpersonal relationships. COVID-19 developed shortly after I gave birth, during the initial haze of the newborn period. Initially, I felt overwhelmed by the many transitions and emotions that were occurring simultaneously. However, as I began to prioritize socialization for myself and my family (albeit creatively at times while socially distancing), I witnessed its positive effects on my emotional well-being and recognized its value in managing times of transition.
Using IPT for families, colleagues, and ourselves
As general psychiatrists, there are several ways to utilize IPT-related themes during this time:
- Connect with families: Although families may recognize they are struggling emotionally, some may find it difficult to navigate the sea of mental health resources. This is particularly true when a family’s financial situation is also stressed. Reaching out to local religious services and community medical resources or inquiring about the mental health of other family members are ways for psychiatrists to engage more families in mental health treatment.
- Reach out to colleagues: Psychiatrists are not immune to developing psychiatric disorders,and it is important to support each other.9 This is also an unusual time when psychiatrists are treating symptoms in patients that they themselves may be also experiencing. Supporting help groups and hot lines, reaching out to colleagues who appear to be struggling and addressing interpersonal conflicts within one’s practice are crucial practices for psychiatrists during this time.
- Explore within ourselves: Evaluating our own interpersonal relationships as well as areas for improvement are critical skills to maintain our own emotional well-being. Setting aside time to connect with friends in a nonclinical setting and prioritizing our family connections are helpful tools. In addition, exploring our reactions to past life transitions could improve our own level of insight into our response to COVID-19.
Conclusion
Conceptualizing COVID-19 as a period of transition and using IPT themes are helpful tools to mitigate the potential adverse psychological effects of COVID-19 on families. Similarly, they can also be helpful in supporting our colleagues and helping ourselves cope during this difficult period.
References
1. Qiu J et al. Gen Psychiatr. 2020 Mar 6;33(2):e100213.
2. Gautam M et al. Psychosomatics. 2020 Apr 20. doi: 10.1016/j.psym.2020.04.009.
3. Markowitz JC, Weissman MM. Clin Psychol Psychother. 2012 Mar-Apr;19(2):99-105.
4. Cuijpers P et al. Am J Psychiatry. 2016 Jul;173(7):680-7.
5. Dietz LJ et al. J Am Acad Child Adolesc Psychiatry. 2015 Mar;54(3):191-9.
6. Verdeli H et al. Child Adolesc Psychiatr Clin N Am. 2008 Jul;17(3):605-24.
7. Stuart S. Clin Psychol Psychother. 2012 Mar-Apr;19(2):134-40.
8. Rogers JP et al. Lancet Psychiatry. 2020 Jul;7(7):611-27.
9. Korkeila JA et al. Scand J Public Health. 2003;31(2):85-91.
Dr. Reinstein is a psychiatry attending at Zucker Hillside Hospital, New York. Her clinical interests include reproductive psychiatry and family therapy, with a specific focus on maternal mental health. She is one of the recipients of the 4th Annual Resident Recognition Award for Excellence in Family Oriented Care. Dr. Reinstein has no conflicts of interest. Alison M. Heru, MD, the Families in Psychiatry columnist, invited Dr. Reinstein to address this topic.
When I was preparing for the recent birth of my baby, I anticipated a period of transition for myself. As a reproductive psychiatrist, I have treated many women during the perinatal and postpartum periods, and have a unique appreciation for the life changes that accompany birth. What I did not expect, however, was the world transitioning with me.
“The new normal” is an economic phrase that describes the COVID-19 era. The pandemic has engendered economic instability, collapsed industries, challenged health care systems, and has led to many deaths worldwide. The COVID-19 pandemic also has been associated with overall increases in anxiety and depression.1 Emerging research suggests that frontline medical workers are especially at risk for developing psychological distress.2
COVID-19 has also created immense challenges for families. Because of concern for the spread of the virus, schools have been suspended, older grandparents isolated, and many parents continue to work remotely. For families in psychiatric care, this time has also been a time of change. Telepsychiatry might be more accessible, but the transition has been an adjustment for patients and clinicians.
As psychiatrists, how do we best treat families during this time? What are some ways to support our psychiatric colleagues? How do we ensure our own emotional well-being amid the tremendous changes occurring around us?
Background of interpersonal psychotherapy
Interpersonal psychotherapy (IPT) is a form of psychotherapy designed to treat depression following periods of transition. Its main goals include improving interpersonal connection and reducing psychological distress. Originally developed in the 1970s by Gerald Klerman, MD; Myrna Weissman, PhD; and Eugene Paykel, MD, IPT is a structured, time-limited form of psychotherapy.3
Conceptualizing depression as a treatable illness, Pim Cuijpers, PhD, and associates summarized the division of IPT into three phases.4 The initial phase involves history taking, forming an alliance, and choosing an interpersonal focus for treatment. The middle phase focuses on applying interpersonal problem-specific therapeutic techniques. The concluding phase of treatment involves consolidation of gains as well as formulating contingency plans for relapse of symptoms. Over the course of treatment, an IPT clinician focuses on life transitions and emphasizes that isolation and antagonistic relationships increase an individual’s vulnerability for a depressive episode.3
Randomized, controlled trials support IPT’s efficacy as a treatment for depression. Research also suggests it can possibly prevent the development of depression.4 Although IPT initially was designed as an individual form of psychotherapy, it has been adapted to both family and group contexts.5,6 IPT is also an empirically valid form of psychotherapy for postpartum depression.7
Interpersonal psychotherapy for families
Given IPT’s role for treating depression following times of transition, clinicians should consider adapting interpersonal psychotherapy to family treatment during this time. Addressing social isolation, managing complex family relationships, and monitoring the family’s overall emotional health should be prioritized. Families under quarantine or who are grieving the death of family members may especially benefit from improved interpersonal connection. Consistent with the IPT model, contingency plans for the family should also be explored to prepare for potential future waves of the pandemic.
In addition to supporting and strengthening families, psychiatrists can use IPT themes to identify positive changes for families tied to COVID-19. Despite its difficulties, the stay-at-home order provided some families a unique chance to slow down and adapt a more relaxed routine. Busy families were suddenly given the opportunity to spend more time with one another. Although many older grandparents were isolated, creative uses of technology provided a chance for grandparents to remain an integral part of family life. Psychiatrists can assist families in transitioning back to previous schedules, while also exploring ways to incorporate the positive changes gained during this time.
Interpersonal psychotherapy for psychiatrists
An interpersonal focus could also be helpful for clinicians to adapt to changes in psychiatric practice. Many clinicians have been thrust into telepsychiatry practice, some with little to no preparation. Because of the trauma associated with frontline work, some psychiatrists have expanded their patient panel to treat physician colleagues. For consult-liaison psychiatrists, the possible neuropsychiatric effects of COVID-19 are new symptoms to consider when evaluating patients in a medical hospital setting.8 Fundamentally, modern day psychiatrists have never encountered a pandemic nor attempted to treat its psychological implications. Prioritizing seeking support from colleagues and caring for one’s personal relationships are helpful tools for clinicians to maintain their own emotional health during this challenging period.
Personal reflection
When I reflect on my baby’s recent birth, I recognize the importance of interpersonal relationships. COVID-19 developed shortly after I gave birth, during the initial haze of the newborn period. Initially, I felt overwhelmed by the many transitions and emotions that were occurring simultaneously. However, as I began to prioritize socialization for myself and my family (albeit creatively at times while socially distancing), I witnessed its positive effects on my emotional well-being and recognized its value in managing times of transition.
Using IPT for families, colleagues, and ourselves
As general psychiatrists, there are several ways to utilize IPT-related themes during this time:
- Connect with families: Although families may recognize they are struggling emotionally, some may find it difficult to navigate the sea of mental health resources. This is particularly true when a family’s financial situation is also stressed. Reaching out to local religious services and community medical resources or inquiring about the mental health of other family members are ways for psychiatrists to engage more families in mental health treatment.
- Reach out to colleagues: Psychiatrists are not immune to developing psychiatric disorders,and it is important to support each other.9 This is also an unusual time when psychiatrists are treating symptoms in patients that they themselves may be also experiencing. Supporting help groups and hot lines, reaching out to colleagues who appear to be struggling and addressing interpersonal conflicts within one’s practice are crucial practices for psychiatrists during this time.
- Explore within ourselves: Evaluating our own interpersonal relationships as well as areas for improvement are critical skills to maintain our own emotional well-being. Setting aside time to connect with friends in a nonclinical setting and prioritizing our family connections are helpful tools. In addition, exploring our reactions to past life transitions could improve our own level of insight into our response to COVID-19.
Conclusion
Conceptualizing COVID-19 as a period of transition and using IPT themes are helpful tools to mitigate the potential adverse psychological effects of COVID-19 on families. Similarly, they can also be helpful in supporting our colleagues and helping ourselves cope during this difficult period.
References
1. Qiu J et al. Gen Psychiatr. 2020 Mar 6;33(2):e100213.
2. Gautam M et al. Psychosomatics. 2020 Apr 20. doi: 10.1016/j.psym.2020.04.009.
3. Markowitz JC, Weissman MM. Clin Psychol Psychother. 2012 Mar-Apr;19(2):99-105.
4. Cuijpers P et al. Am J Psychiatry. 2016 Jul;173(7):680-7.
5. Dietz LJ et al. J Am Acad Child Adolesc Psychiatry. 2015 Mar;54(3):191-9.
6. Verdeli H et al. Child Adolesc Psychiatr Clin N Am. 2008 Jul;17(3):605-24.
7. Stuart S. Clin Psychol Psychother. 2012 Mar-Apr;19(2):134-40.
8. Rogers JP et al. Lancet Psychiatry. 2020 Jul;7(7):611-27.
9. Korkeila JA et al. Scand J Public Health. 2003;31(2):85-91.
Dr. Reinstein is a psychiatry attending at Zucker Hillside Hospital, New York. Her clinical interests include reproductive psychiatry and family therapy, with a specific focus on maternal mental health. She is one of the recipients of the 4th Annual Resident Recognition Award for Excellence in Family Oriented Care. Dr. Reinstein has no conflicts of interest. Alison M. Heru, MD, the Families in Psychiatry columnist, invited Dr. Reinstein to address this topic.
When I was preparing for the recent birth of my baby, I anticipated a period of transition for myself. As a reproductive psychiatrist, I have treated many women during the perinatal and postpartum periods, and have a unique appreciation for the life changes that accompany birth. What I did not expect, however, was the world transitioning with me.
“The new normal” is an economic phrase that describes the COVID-19 era. The pandemic has engendered economic instability, collapsed industries, challenged health care systems, and has led to many deaths worldwide. The COVID-19 pandemic also has been associated with overall increases in anxiety and depression.1 Emerging research suggests that frontline medical workers are especially at risk for developing psychological distress.2
COVID-19 has also created immense challenges for families. Because of concern for the spread of the virus, schools have been suspended, older grandparents isolated, and many parents continue to work remotely. For families in psychiatric care, this time has also been a time of change. Telepsychiatry might be more accessible, but the transition has been an adjustment for patients and clinicians.
As psychiatrists, how do we best treat families during this time? What are some ways to support our psychiatric colleagues? How do we ensure our own emotional well-being amid the tremendous changes occurring around us?
Background of interpersonal psychotherapy
Interpersonal psychotherapy (IPT) is a form of psychotherapy designed to treat depression following periods of transition. Its main goals include improving interpersonal connection and reducing psychological distress. Originally developed in the 1970s by Gerald Klerman, MD; Myrna Weissman, PhD; and Eugene Paykel, MD, IPT is a structured, time-limited form of psychotherapy.3
Conceptualizing depression as a treatable illness, Pim Cuijpers, PhD, and associates summarized the division of IPT into three phases.4 The initial phase involves history taking, forming an alliance, and choosing an interpersonal focus for treatment. The middle phase focuses on applying interpersonal problem-specific therapeutic techniques. The concluding phase of treatment involves consolidation of gains as well as formulating contingency plans for relapse of symptoms. Over the course of treatment, an IPT clinician focuses on life transitions and emphasizes that isolation and antagonistic relationships increase an individual’s vulnerability for a depressive episode.3
Randomized, controlled trials support IPT’s efficacy as a treatment for depression. Research also suggests it can possibly prevent the development of depression.4 Although IPT initially was designed as an individual form of psychotherapy, it has been adapted to both family and group contexts.5,6 IPT is also an empirically valid form of psychotherapy for postpartum depression.7
Interpersonal psychotherapy for families
Given IPT’s role for treating depression following times of transition, clinicians should consider adapting interpersonal psychotherapy to family treatment during this time. Addressing social isolation, managing complex family relationships, and monitoring the family’s overall emotional health should be prioritized. Families under quarantine or who are grieving the death of family members may especially benefit from improved interpersonal connection. Consistent with the IPT model, contingency plans for the family should also be explored to prepare for potential future waves of the pandemic.
In addition to supporting and strengthening families, psychiatrists can use IPT themes to identify positive changes for families tied to COVID-19. Despite its difficulties, the stay-at-home order provided some families a unique chance to slow down and adapt a more relaxed routine. Busy families were suddenly given the opportunity to spend more time with one another. Although many older grandparents were isolated, creative uses of technology provided a chance for grandparents to remain an integral part of family life. Psychiatrists can assist families in transitioning back to previous schedules, while also exploring ways to incorporate the positive changes gained during this time.
Interpersonal psychotherapy for psychiatrists
An interpersonal focus could also be helpful for clinicians to adapt to changes in psychiatric practice. Many clinicians have been thrust into telepsychiatry practice, some with little to no preparation. Because of the trauma associated with frontline work, some psychiatrists have expanded their patient panel to treat physician colleagues. For consult-liaison psychiatrists, the possible neuropsychiatric effects of COVID-19 are new symptoms to consider when evaluating patients in a medical hospital setting.8 Fundamentally, modern day psychiatrists have never encountered a pandemic nor attempted to treat its psychological implications. Prioritizing seeking support from colleagues and caring for one’s personal relationships are helpful tools for clinicians to maintain their own emotional health during this challenging period.
Personal reflection
When I reflect on my baby’s recent birth, I recognize the importance of interpersonal relationships. COVID-19 developed shortly after I gave birth, during the initial haze of the newborn period. Initially, I felt overwhelmed by the many transitions and emotions that were occurring simultaneously. However, as I began to prioritize socialization for myself and my family (albeit creatively at times while socially distancing), I witnessed its positive effects on my emotional well-being and recognized its value in managing times of transition.
Using IPT for families, colleagues, and ourselves
As general psychiatrists, there are several ways to utilize IPT-related themes during this time:
- Connect with families: Although families may recognize they are struggling emotionally, some may find it difficult to navigate the sea of mental health resources. This is particularly true when a family’s financial situation is also stressed. Reaching out to local religious services and community medical resources or inquiring about the mental health of other family members are ways for psychiatrists to engage more families in mental health treatment.
- Reach out to colleagues: Psychiatrists are not immune to developing psychiatric disorders,and it is important to support each other.9 This is also an unusual time when psychiatrists are treating symptoms in patients that they themselves may be also experiencing. Supporting help groups and hot lines, reaching out to colleagues who appear to be struggling and addressing interpersonal conflicts within one’s practice are crucial practices for psychiatrists during this time.
- Explore within ourselves: Evaluating our own interpersonal relationships as well as areas for improvement are critical skills to maintain our own emotional well-being. Setting aside time to connect with friends in a nonclinical setting and prioritizing our family connections are helpful tools. In addition, exploring our reactions to past life transitions could improve our own level of insight into our response to COVID-19.
Conclusion
Conceptualizing COVID-19 as a period of transition and using IPT themes are helpful tools to mitigate the potential adverse psychological effects of COVID-19 on families. Similarly, they can also be helpful in supporting our colleagues and helping ourselves cope during this difficult period.
References
1. Qiu J et al. Gen Psychiatr. 2020 Mar 6;33(2):e100213.
2. Gautam M et al. Psychosomatics. 2020 Apr 20. doi: 10.1016/j.psym.2020.04.009.
3. Markowitz JC, Weissman MM. Clin Psychol Psychother. 2012 Mar-Apr;19(2):99-105.
4. Cuijpers P et al. Am J Psychiatry. 2016 Jul;173(7):680-7.
5. Dietz LJ et al. J Am Acad Child Adolesc Psychiatry. 2015 Mar;54(3):191-9.
6. Verdeli H et al. Child Adolesc Psychiatr Clin N Am. 2008 Jul;17(3):605-24.
7. Stuart S. Clin Psychol Psychother. 2012 Mar-Apr;19(2):134-40.
8. Rogers JP et al. Lancet Psychiatry. 2020 Jul;7(7):611-27.
9. Korkeila JA et al. Scand J Public Health. 2003;31(2):85-91.
Dr. Reinstein is a psychiatry attending at Zucker Hillside Hospital, New York. Her clinical interests include reproductive psychiatry and family therapy, with a specific focus on maternal mental health. She is one of the recipients of the 4th Annual Resident Recognition Award for Excellence in Family Oriented Care. Dr. Reinstein has no conflicts of interest. Alison M. Heru, MD, the Families in Psychiatry columnist, invited Dr. Reinstein to address this topic.
Anxiety disorders begin earlier in life, differ by gender
Anxiety disorders start very early in life and may manifest themselves first as other conditions like social anxiety disorder, according to Jeffrey R. Strawn, MD.
An adolescent presenting to a mental health clinician with anxiety at 16 years old, for example has likely struggled with her anxiety for years before visiting a clinic. “That child may have been someone who had separation anxiety earlier in life and who as, even an infant, had behavioral inhibitions, that reluctance or timidness to explore new things, that tendency to retreat from novel stimuli,” Dr. Strawn, associate professor of psychiatry, pediatrics and clinical pharmacology at Cincinnati Children’s Hospital Medical Center, said at Focus on Neuropsychiatry presented by Current Psychiatry and the American Academy of Clinical Psychiatrists. “Anxiety disorders are enduring and persistent, and they begin very early in life.”
Social anxiety disorder is one of the first anxiety disorders that appear in childhood or adolescents, which rises during puberty and during a time in a child’s life when they are dealing with new social pressures and challenges, such as graduating from elementary to middle school, Dr. Strawn noted. Generalized anxiety disorder is usually the next to emerge, followed by panic disorder. On the other hand, agoraphobia, another anxiety disorder that begins in childhood, “often represents behavioral avoidance as opposed to agoraphobia as we classically think about it as adult psychiatrists.”
Onset of anxiety disorders also differ by gender. “In terms of the emergence of these anxiety disorders, another thing that’s important to know is that the onset seems to be a bit different with regard to girls and boys. We see that break there emerging really around the time of puberty or as people are moving into late puberty, at least for girls,” Dr. Strawn said at the meeting presented by Global Academy for Medical Education. .
A shift occurs in amygdala prefrontal circuitry as children age, Dr. Strawn explained. Younger children do not have the ability to modulate the amygdala with their prefrontal cortex, but this amygdala–medial prefrontal cortex functional connectivity will change as children grow. A study by Dylan G. Gee, PhD, and colleagues found positive amygdala–medial prefrontal cortex functional connectivity at younger than 10 years old, and a “steady decline in amygdala activity” from 10-13 years to adulthood at 22 years old (J Neurosci. 2013 Mar 6;33[10]:4584-93).
“In essence, what we’re seeing is that there’s improvement or more effectiveness in terms of that connection between the prefrontal cortex, the amygdala, and that ability to amplify the brake to the amygdala,” Dr. Strawn said.
SSRIs, SNRIs for pediatric patients
Selective serotonin reuptake inhibitors can be effective for pediatric patients with anxiety disorders. Results from the Child/Adolescent Anxiety Multimodal Study (CAMS) show that patients with generalized separation or social anxiety disorder treated with sertraline or cognitive-behavioral therapy (CBT) for 3 months responded better to treatment than placebo. A combination of sertraline and CBT performing best, compared with either intervention alone (N Engl J Med. 2008;359:2753-66).
When examining treatment response in 76 patients from CAMS, the researchers saw improvement at 4 weeks from baseline in patients with anxiety symptoms receiving CBT, but no significant change in improvement after 4 weeks up to 12 weeks (J Child Adolesc Psychopharm. 2017 Aug 1. doi: 10.1089/cap.2016.0198).
“What that actually means is that your improvement at week 4 is better than your improvement at baseline, and your improvement at week 8 is greater than your improvement at week 4. Similarly, in your improvement, week 12 is greater than your improvement at week 8,” Dr. Strawn said.
However, “that’s not the case for aggressively titrated sertraline,” which had no statistically significant difference in improvement at 8 weeks and 12 weeks, he explained. “What this actually means is that, if I have not had improvement by week 8, there is a three-to-one odds against improvement over those next 4 weeks. The take-home message here is really that an adequate trial for an SSRI in pediatric anxiety disorders is probably about 8 weeks – not 12, not longer.”
Serotonin norepinephrine reuptake inhibitors (SNRIs) are also effective in pediatric patients with anxiety disorders.
“Both SNRIs as well as SSRIs have certainly demonstrated efficacy in terms of treating pediatric patients with anxiety, but there is a very important difference here with regard to the trajectory of improvement and also the magnitude of improvement,” Dr. Strawn said. SNRIs like atomoxetine, duloxetine, or venlafaxine “do not improve as rapidly and do not improve to the same extent as kids who are treated with an SSRI.”
Dose is another factor that affects symptom improvement in patients with pediatric anxiety disorders. In a 2018 meta-analysis, Dr. Strawn and colleagues found that patients treated with a higher dose of SSRIs demonstrated more rapid improvement at 2 weeks, compared with patients who received SNRIs (P = .002), but there was no significant difference in overall response trajectory (J Am Acad Child Adolesc Psychiatry. 2018 Apr;57[4]:235-44.E2).
Response to SSRIs can depend a patient’s genotype, Dr. Strawn said. The serotonin transporter promotor polymorphism has received “considerable attention in adults with depressive disorders primarily” but also might play a role in anxiety disorder response in pediatric patients. One study presented by his group at the 2019 annual meeting of the American Academy of Child & Adolescent Psychiatry showed that patients with a short-short copy of the serotonin transporter promoter polymorphism instead of a long copy had “shallower and less improvement over the course of treatment” when taking escitalopram.
“This is something that doesn’t necessarily compel us to use an SNRI over an SSRI, but it’s something that does give us some important information in terms of the trajectory of improvement,” he said.
When it comes to side effects of SNRIs and SSRIs, the profile is “pretty consistent with what we know to be the side effect profile in adults with depressive and anxiety disorders,” Dr. Strawn noted. “SNRIs tend to be a little bit better tolerated, both in terms of adverse event–related discontinuation and also in terms of their likelihood of producing activation.”
Patient and caregiver expectations can further affect response to treatment. “I think this has implications in terms of how we actively manage expectations and discussions about the evidence for interventions with our patients in the clinic.”
Global Academy and this news organization are owned by the same parent company. Dr. Strawn reported receiving research support from Edgemont Pharmaceuticals, Eli Lilly, Forest Research Laboratories, Lundbeck, the National Institutes of Health, Neuronetics, and Shire. He also reported receiving royalties from Springer Publishing, and is a consultant for and receives material support from Assurex/Genesight.
Anxiety disorders start very early in life and may manifest themselves first as other conditions like social anxiety disorder, according to Jeffrey R. Strawn, MD.
An adolescent presenting to a mental health clinician with anxiety at 16 years old, for example has likely struggled with her anxiety for years before visiting a clinic. “That child may have been someone who had separation anxiety earlier in life and who as, even an infant, had behavioral inhibitions, that reluctance or timidness to explore new things, that tendency to retreat from novel stimuli,” Dr. Strawn, associate professor of psychiatry, pediatrics and clinical pharmacology at Cincinnati Children’s Hospital Medical Center, said at Focus on Neuropsychiatry presented by Current Psychiatry and the American Academy of Clinical Psychiatrists. “Anxiety disorders are enduring and persistent, and they begin very early in life.”
Social anxiety disorder is one of the first anxiety disorders that appear in childhood or adolescents, which rises during puberty and during a time in a child’s life when they are dealing with new social pressures and challenges, such as graduating from elementary to middle school, Dr. Strawn noted. Generalized anxiety disorder is usually the next to emerge, followed by panic disorder. On the other hand, agoraphobia, another anxiety disorder that begins in childhood, “often represents behavioral avoidance as opposed to agoraphobia as we classically think about it as adult psychiatrists.”
Onset of anxiety disorders also differ by gender. “In terms of the emergence of these anxiety disorders, another thing that’s important to know is that the onset seems to be a bit different with regard to girls and boys. We see that break there emerging really around the time of puberty or as people are moving into late puberty, at least for girls,” Dr. Strawn said at the meeting presented by Global Academy for Medical Education. .
A shift occurs in amygdala prefrontal circuitry as children age, Dr. Strawn explained. Younger children do not have the ability to modulate the amygdala with their prefrontal cortex, but this amygdala–medial prefrontal cortex functional connectivity will change as children grow. A study by Dylan G. Gee, PhD, and colleagues found positive amygdala–medial prefrontal cortex functional connectivity at younger than 10 years old, and a “steady decline in amygdala activity” from 10-13 years to adulthood at 22 years old (J Neurosci. 2013 Mar 6;33[10]:4584-93).
“In essence, what we’re seeing is that there’s improvement or more effectiveness in terms of that connection between the prefrontal cortex, the amygdala, and that ability to amplify the brake to the amygdala,” Dr. Strawn said.
SSRIs, SNRIs for pediatric patients
Selective serotonin reuptake inhibitors can be effective for pediatric patients with anxiety disorders. Results from the Child/Adolescent Anxiety Multimodal Study (CAMS) show that patients with generalized separation or social anxiety disorder treated with sertraline or cognitive-behavioral therapy (CBT) for 3 months responded better to treatment than placebo. A combination of sertraline and CBT performing best, compared with either intervention alone (N Engl J Med. 2008;359:2753-66).
When examining treatment response in 76 patients from CAMS, the researchers saw improvement at 4 weeks from baseline in patients with anxiety symptoms receiving CBT, but no significant change in improvement after 4 weeks up to 12 weeks (J Child Adolesc Psychopharm. 2017 Aug 1. doi: 10.1089/cap.2016.0198).
“What that actually means is that your improvement at week 4 is better than your improvement at baseline, and your improvement at week 8 is greater than your improvement at week 4. Similarly, in your improvement, week 12 is greater than your improvement at week 8,” Dr. Strawn said.
However, “that’s not the case for aggressively titrated sertraline,” which had no statistically significant difference in improvement at 8 weeks and 12 weeks, he explained. “What this actually means is that, if I have not had improvement by week 8, there is a three-to-one odds against improvement over those next 4 weeks. The take-home message here is really that an adequate trial for an SSRI in pediatric anxiety disorders is probably about 8 weeks – not 12, not longer.”
Serotonin norepinephrine reuptake inhibitors (SNRIs) are also effective in pediatric patients with anxiety disorders.
“Both SNRIs as well as SSRIs have certainly demonstrated efficacy in terms of treating pediatric patients with anxiety, but there is a very important difference here with regard to the trajectory of improvement and also the magnitude of improvement,” Dr. Strawn said. SNRIs like atomoxetine, duloxetine, or venlafaxine “do not improve as rapidly and do not improve to the same extent as kids who are treated with an SSRI.”
Dose is another factor that affects symptom improvement in patients with pediatric anxiety disorders. In a 2018 meta-analysis, Dr. Strawn and colleagues found that patients treated with a higher dose of SSRIs demonstrated more rapid improvement at 2 weeks, compared with patients who received SNRIs (P = .002), but there was no significant difference in overall response trajectory (J Am Acad Child Adolesc Psychiatry. 2018 Apr;57[4]:235-44.E2).
Response to SSRIs can depend a patient’s genotype, Dr. Strawn said. The serotonin transporter promotor polymorphism has received “considerable attention in adults with depressive disorders primarily” but also might play a role in anxiety disorder response in pediatric patients. One study presented by his group at the 2019 annual meeting of the American Academy of Child & Adolescent Psychiatry showed that patients with a short-short copy of the serotonin transporter promoter polymorphism instead of a long copy had “shallower and less improvement over the course of treatment” when taking escitalopram.
“This is something that doesn’t necessarily compel us to use an SNRI over an SSRI, but it’s something that does give us some important information in terms of the trajectory of improvement,” he said.
When it comes to side effects of SNRIs and SSRIs, the profile is “pretty consistent with what we know to be the side effect profile in adults with depressive and anxiety disorders,” Dr. Strawn noted. “SNRIs tend to be a little bit better tolerated, both in terms of adverse event–related discontinuation and also in terms of their likelihood of producing activation.”
Patient and caregiver expectations can further affect response to treatment. “I think this has implications in terms of how we actively manage expectations and discussions about the evidence for interventions with our patients in the clinic.”
Global Academy and this news organization are owned by the same parent company. Dr. Strawn reported receiving research support from Edgemont Pharmaceuticals, Eli Lilly, Forest Research Laboratories, Lundbeck, the National Institutes of Health, Neuronetics, and Shire. He also reported receiving royalties from Springer Publishing, and is a consultant for and receives material support from Assurex/Genesight.
Anxiety disorders start very early in life and may manifest themselves first as other conditions like social anxiety disorder, according to Jeffrey R. Strawn, MD.
An adolescent presenting to a mental health clinician with anxiety at 16 years old, for example has likely struggled with her anxiety for years before visiting a clinic. “That child may have been someone who had separation anxiety earlier in life and who as, even an infant, had behavioral inhibitions, that reluctance or timidness to explore new things, that tendency to retreat from novel stimuli,” Dr. Strawn, associate professor of psychiatry, pediatrics and clinical pharmacology at Cincinnati Children’s Hospital Medical Center, said at Focus on Neuropsychiatry presented by Current Psychiatry and the American Academy of Clinical Psychiatrists. “Anxiety disorders are enduring and persistent, and they begin very early in life.”
Social anxiety disorder is one of the first anxiety disorders that appear in childhood or adolescents, which rises during puberty and during a time in a child’s life when they are dealing with new social pressures and challenges, such as graduating from elementary to middle school, Dr. Strawn noted. Generalized anxiety disorder is usually the next to emerge, followed by panic disorder. On the other hand, agoraphobia, another anxiety disorder that begins in childhood, “often represents behavioral avoidance as opposed to agoraphobia as we classically think about it as adult psychiatrists.”
Onset of anxiety disorders also differ by gender. “In terms of the emergence of these anxiety disorders, another thing that’s important to know is that the onset seems to be a bit different with regard to girls and boys. We see that break there emerging really around the time of puberty or as people are moving into late puberty, at least for girls,” Dr. Strawn said at the meeting presented by Global Academy for Medical Education. .
A shift occurs in amygdala prefrontal circuitry as children age, Dr. Strawn explained. Younger children do not have the ability to modulate the amygdala with their prefrontal cortex, but this amygdala–medial prefrontal cortex functional connectivity will change as children grow. A study by Dylan G. Gee, PhD, and colleagues found positive amygdala–medial prefrontal cortex functional connectivity at younger than 10 years old, and a “steady decline in amygdala activity” from 10-13 years to adulthood at 22 years old (J Neurosci. 2013 Mar 6;33[10]:4584-93).
“In essence, what we’re seeing is that there’s improvement or more effectiveness in terms of that connection between the prefrontal cortex, the amygdala, and that ability to amplify the brake to the amygdala,” Dr. Strawn said.
SSRIs, SNRIs for pediatric patients
Selective serotonin reuptake inhibitors can be effective for pediatric patients with anxiety disorders. Results from the Child/Adolescent Anxiety Multimodal Study (CAMS) show that patients with generalized separation or social anxiety disorder treated with sertraline or cognitive-behavioral therapy (CBT) for 3 months responded better to treatment than placebo. A combination of sertraline and CBT performing best, compared with either intervention alone (N Engl J Med. 2008;359:2753-66).
When examining treatment response in 76 patients from CAMS, the researchers saw improvement at 4 weeks from baseline in patients with anxiety symptoms receiving CBT, but no significant change in improvement after 4 weeks up to 12 weeks (J Child Adolesc Psychopharm. 2017 Aug 1. doi: 10.1089/cap.2016.0198).
“What that actually means is that your improvement at week 4 is better than your improvement at baseline, and your improvement at week 8 is greater than your improvement at week 4. Similarly, in your improvement, week 12 is greater than your improvement at week 8,” Dr. Strawn said.
However, “that’s not the case for aggressively titrated sertraline,” which had no statistically significant difference in improvement at 8 weeks and 12 weeks, he explained. “What this actually means is that, if I have not had improvement by week 8, there is a three-to-one odds against improvement over those next 4 weeks. The take-home message here is really that an adequate trial for an SSRI in pediatric anxiety disorders is probably about 8 weeks – not 12, not longer.”
Serotonin norepinephrine reuptake inhibitors (SNRIs) are also effective in pediatric patients with anxiety disorders.
“Both SNRIs as well as SSRIs have certainly demonstrated efficacy in terms of treating pediatric patients with anxiety, but there is a very important difference here with regard to the trajectory of improvement and also the magnitude of improvement,” Dr. Strawn said. SNRIs like atomoxetine, duloxetine, or venlafaxine “do not improve as rapidly and do not improve to the same extent as kids who are treated with an SSRI.”
Dose is another factor that affects symptom improvement in patients with pediatric anxiety disorders. In a 2018 meta-analysis, Dr. Strawn and colleagues found that patients treated with a higher dose of SSRIs demonstrated more rapid improvement at 2 weeks, compared with patients who received SNRIs (P = .002), but there was no significant difference in overall response trajectory (J Am Acad Child Adolesc Psychiatry. 2018 Apr;57[4]:235-44.E2).
Response to SSRIs can depend a patient’s genotype, Dr. Strawn said. The serotonin transporter promotor polymorphism has received “considerable attention in adults with depressive disorders primarily” but also might play a role in anxiety disorder response in pediatric patients. One study presented by his group at the 2019 annual meeting of the American Academy of Child & Adolescent Psychiatry showed that patients with a short-short copy of the serotonin transporter promoter polymorphism instead of a long copy had “shallower and less improvement over the course of treatment” when taking escitalopram.
“This is something that doesn’t necessarily compel us to use an SNRI over an SSRI, but it’s something that does give us some important information in terms of the trajectory of improvement,” he said.
When it comes to side effects of SNRIs and SSRIs, the profile is “pretty consistent with what we know to be the side effect profile in adults with depressive and anxiety disorders,” Dr. Strawn noted. “SNRIs tend to be a little bit better tolerated, both in terms of adverse event–related discontinuation and also in terms of their likelihood of producing activation.”
Patient and caregiver expectations can further affect response to treatment. “I think this has implications in terms of how we actively manage expectations and discussions about the evidence for interventions with our patients in the clinic.”
Global Academy and this news organization are owned by the same parent company. Dr. Strawn reported receiving research support from Edgemont Pharmaceuticals, Eli Lilly, Forest Research Laboratories, Lundbeck, the National Institutes of Health, Neuronetics, and Shire. He also reported receiving royalties from Springer Publishing, and is a consultant for and receives material support from Assurex/Genesight.
FROM Focus on Neuropsychiatry 2020
CDC data confirm mental health is suffering during COVID-19
The ongoing COVID-19 pandemic continues to exact a huge toll on mental health in the United States, according to results of a survey released Aug. 13 by the Centers for Disease Control and Prevention.
During late June, about two in five U.S. adults surveyed said they were struggling with mental health or substance use. Younger adults, racial/ethnic minorities, essential workers, and those with preexisting psychiatric conditions were suffering the most.
“Addressing mental health disparities and preparing support systems to mitigate mental health consequences as the pandemic evolves will continue to be needed urgently,” write Rashon Lane, with the CDC COVID-19 Response Team, and colleagues in an article published online in the CDC’s Morbidity and Mortality Weekly Report.
During the period of June 24-30, 2020, 5,412 U.S. adults aged 18 and older completed online surveys that gauged mental health, substance use, and suicidal ideation.
Overall, 40.9% of respondents reported having at least one adverse mental or behavioral health condition; 31% reported symptoms of anxiety or depressive disorder; and 26% reported symptoms of a trauma- and stressor-related disorder related to the pandemic.
The prevalence of symptoms of anxiety disorder alone was roughly three times that reported in the second quarter of 2019, the authors noted.
In addition, , and nearly 11% reported having seriously considered suicide in the preceding 30 days.
Approximately twice as many respondents reported seriously considering suicide in the prior month compared with adults in the United States in 2018 (referring to the previous 12 months), the authors noted.
Suicidal ideation was significantly higher among younger respondents (aged 18-24 years, 26%), Hispanic persons (19%), non-Hispanic Black persons (15%), unpaid caregivers for adults (31%), and essential workers (22%).
The survey results are in line with recent data from Mental Health America, which indicate dramatic increases in depression, anxiety, and suicidality since the start of the COVID-19 pandemic.
The “markedly elevated” prevalence of adverse mental and behavioral health conditions associated with the COVID-19 pandemic highlights the “broad impact of the pandemic and the need to prevent and treat these conditions,” the researchers wrote.
The survey also highlights populations at increased risk for psychological distress and unhealthy coping.
“Future studies should identify drivers of adverse mental and behavioral health during the COVID-19 pandemic and whether factors such as social isolation, absence of school structure, unemployment and other financial worries, and various forms of violence (e.g., physical, emotional, mental, or sexual abuse) serve as additional stressors,” they suggested.
A version of this article originally appeared on Medscape.com.
The ongoing COVID-19 pandemic continues to exact a huge toll on mental health in the United States, according to results of a survey released Aug. 13 by the Centers for Disease Control and Prevention.
During late June, about two in five U.S. adults surveyed said they were struggling with mental health or substance use. Younger adults, racial/ethnic minorities, essential workers, and those with preexisting psychiatric conditions were suffering the most.
“Addressing mental health disparities and preparing support systems to mitigate mental health consequences as the pandemic evolves will continue to be needed urgently,” write Rashon Lane, with the CDC COVID-19 Response Team, and colleagues in an article published online in the CDC’s Morbidity and Mortality Weekly Report.
During the period of June 24-30, 2020, 5,412 U.S. adults aged 18 and older completed online surveys that gauged mental health, substance use, and suicidal ideation.
Overall, 40.9% of respondents reported having at least one adverse mental or behavioral health condition; 31% reported symptoms of anxiety or depressive disorder; and 26% reported symptoms of a trauma- and stressor-related disorder related to the pandemic.
The prevalence of symptoms of anxiety disorder alone was roughly three times that reported in the second quarter of 2019, the authors noted.
In addition, , and nearly 11% reported having seriously considered suicide in the preceding 30 days.
Approximately twice as many respondents reported seriously considering suicide in the prior month compared with adults in the United States in 2018 (referring to the previous 12 months), the authors noted.
Suicidal ideation was significantly higher among younger respondents (aged 18-24 years, 26%), Hispanic persons (19%), non-Hispanic Black persons (15%), unpaid caregivers for adults (31%), and essential workers (22%).
The survey results are in line with recent data from Mental Health America, which indicate dramatic increases in depression, anxiety, and suicidality since the start of the COVID-19 pandemic.
The “markedly elevated” prevalence of adverse mental and behavioral health conditions associated with the COVID-19 pandemic highlights the “broad impact of the pandemic and the need to prevent and treat these conditions,” the researchers wrote.
The survey also highlights populations at increased risk for psychological distress and unhealthy coping.
“Future studies should identify drivers of adverse mental and behavioral health during the COVID-19 pandemic and whether factors such as social isolation, absence of school structure, unemployment and other financial worries, and various forms of violence (e.g., physical, emotional, mental, or sexual abuse) serve as additional stressors,” they suggested.
A version of this article originally appeared on Medscape.com.
The ongoing COVID-19 pandemic continues to exact a huge toll on mental health in the United States, according to results of a survey released Aug. 13 by the Centers for Disease Control and Prevention.
During late June, about two in five U.S. adults surveyed said they were struggling with mental health or substance use. Younger adults, racial/ethnic minorities, essential workers, and those with preexisting psychiatric conditions were suffering the most.
“Addressing mental health disparities and preparing support systems to mitigate mental health consequences as the pandemic evolves will continue to be needed urgently,” write Rashon Lane, with the CDC COVID-19 Response Team, and colleagues in an article published online in the CDC’s Morbidity and Mortality Weekly Report.
During the period of June 24-30, 2020, 5,412 U.S. adults aged 18 and older completed online surveys that gauged mental health, substance use, and suicidal ideation.
Overall, 40.9% of respondents reported having at least one adverse mental or behavioral health condition; 31% reported symptoms of anxiety or depressive disorder; and 26% reported symptoms of a trauma- and stressor-related disorder related to the pandemic.
The prevalence of symptoms of anxiety disorder alone was roughly three times that reported in the second quarter of 2019, the authors noted.
In addition, , and nearly 11% reported having seriously considered suicide in the preceding 30 days.
Approximately twice as many respondents reported seriously considering suicide in the prior month compared with adults in the United States in 2018 (referring to the previous 12 months), the authors noted.
Suicidal ideation was significantly higher among younger respondents (aged 18-24 years, 26%), Hispanic persons (19%), non-Hispanic Black persons (15%), unpaid caregivers for adults (31%), and essential workers (22%).
The survey results are in line with recent data from Mental Health America, which indicate dramatic increases in depression, anxiety, and suicidality since the start of the COVID-19 pandemic.
The “markedly elevated” prevalence of adverse mental and behavioral health conditions associated with the COVID-19 pandemic highlights the “broad impact of the pandemic and the need to prevent and treat these conditions,” the researchers wrote.
The survey also highlights populations at increased risk for psychological distress and unhealthy coping.
“Future studies should identify drivers of adverse mental and behavioral health during the COVID-19 pandemic and whether factors such as social isolation, absence of school structure, unemployment and other financial worries, and various forms of violence (e.g., physical, emotional, mental, or sexual abuse) serve as additional stressors,” they suggested.
A version of this article originally appeared on Medscape.com.
Chloroquine linked to serious psychiatric side effects
Chloroquine may be associated with serious psychiatric side effects, even in patients with no family or personal history of psychiatric disorders, a new review suggests.
In a letter to the editor published online July 28 in The Journal of Clinical Psychiatry, the authors summarize data from several studies published as far back as 1993 and as recently as May 2020.
“In addition to previously reported side effects, chloroquine could also induce psychiatric side effects which are polymorphic and can persist even after stopping the drug,” lead author Florence Gressier, MD, PhD, CESP, Inserm, department of psychiatry, Le Kremlin Bicêtre, France, said in an interview.
“In COVID-19 patients who may still be [undergoing treatment] with chloroquine, close psychiatric assessment and monitoring should be performed,” she said.
Heated controversy
Following findings of a small French study that suggested efficacy in lowering the viral load in patients with COVID-19, President Donald Trump expressed optimism regarding the role of hydroxychloroquine in treating COVID-19, calling it a “game changer”.
Other studies, however, have called into question both the efficacy and the safety of hydroxychloroquine in treating COVID-19. On June 15, the Food and Drug Administration revoked the emergency use authorization it had given in March to chloroquine and hydroxychloroquine for the treatment of COVID-19.
Nevertheless, hydroxychloroquine continues to be prescribed for COVID-19. For example, an article that appeared in Click2Houston on June 15 quoted the chief medical officer of Houston’s United Memorial Center as saying he plans to continue prescribing hydroxychloroquine for patients with COVID-19 until he finds a better alternative.
As discussed in a Medscape expert commentary, a group of physicians who held a “white coat summit” in front of the U.S. Supreme Court building promoted the use of hydroxychloroquine for the treatment of COVID-19. The video of their summit was retweeted by President Trump and garnered millions of views before it was taken down by Twitter, Facebook, and YouTube.
Sudden onset
For the new review, “we wanted to alert the public and practitioners on the potentially psychiatric risks induced by chloroquine, as it could be taken as self-medication or potentially still prescribed,” Dr. Gressier said.
“We think the format of the letter to the editor allows information to be provided in a concise and clear manner,” she added.
According to the FDA’s Adverse Event Reporting System database, 12% of reported adverse events (520 of 4,336) following the use of chloroquine that occurred between the fourth quarter of 2012 and the fourth quarter of 2019 were neuropsychiatric. These events included amnesia, delirium, hallucinations, depression, and loss of consciousness, the authors write.
The researchers acknowledged that the incidence of psychiatric adverse effects associated with the use of chloroquine is “unclear in the absence of high-quality, randomized placebo-controlled trials of its safety.” Nevertheless, they pointed out that there have been reports of insomnia and depression when the drug was used as prophylaxis against malaria .
Moreover, some case series or case reports describe symptoms such as depression, anxiety, agitation, violent outburst, suicidal ideation, and psychosis in patients who have been treated with chloroquine for malaria, lupus erythematosus, and rheumatoid arthritis .
“In contrast to many other psychoses, chloroquine psychosis may be more affective and include prominent visual hallucinations, symptoms of derealization, and disorders of thought, with preserved insight,” the authors wrote.
They noted that the frequency of symptoms does not appear to be connected to the cumulative dose or the duration of treatment, and the onset of psychosis or other adverse effects is usually “sudden.”
In addition, they warn that the drug’s psychiatric effects may go unnoticed, especially because COVID-19 itself has been associated with neuropsychiatric symptoms, making it hard to distinguish between symptoms caused by the illness and those caused by the drug.
Although the psychiatric symptoms typically occur early after treatment initiation, some “subtle” symptoms might persist after stopping the drug, possibly owing to its “extremely long” half-life, the authors stated.
Dr. Gressier noted that practicing clinicians should look up reports about self-medication with chloroquine “and warn their patients about the risk induced by chloroquine.”
Safe but ‘not benign’
Nilanjana Bose, MD, MBA, a rheumatologist at the Rheumatology Center of Houston, said she uses hydroxychloroquine “all the time” in clinical practice to treat patients with rheumatic conditions.
“I cannot comment on whether it [hydroxychloroquine or chloroquine] is a potential prophylactic or treatment for COVID-19, but I can say that, from a safety point of view, as a rheumatologist who uses hydroxychloroquine at a dose of 400 mg/day, I do not think we need to worry about serious [psychiatric] side effects,” Dr. Bose said in an interview.
Because clinicians are trying all types of possible treatments for COVID-19, “if this medication has possible efficacy, it is a great medicine from a rheumatologic perspective and is safe,” she added.
Nevertheless, the drug is “not benign, and regular side effects will be there, and of course, higher doses will cause more side effects,” said Dr. Bose, who was not involved in authoring the letter.
She counsels patients about potential psychiatric side effects of hydroxychloroquine because some of her patients have complained about irritability, worsening anxiety and depression, and difficulty sleeping.
Be wary
James “Jimmy” Potash, MD, MPH, Henry Phipps Professor of Psychiatry and Behavioral Sciences, Johns Hopkins Medicine, Baltimore, said in an interview that the “take-home message of this letter is that serious psychiatric effects, psychotic illness in particular,” can occur in individuals who take chloroquine and hydroxychloroquine.
In addition, “these are potentially very concerning side effects that psychiatrists should be aware of,” noted Dr. Potash, department director and psychiatrist-in-chief at Johns Hopkins.
He said that one of his patients who had been “completely psychiatrically healthy” took chloroquine prophylactically prior to traveling overseas. After she began taking the drug, she had an episode of mania that resolved once she discontinued the medication and received treatment for the mania.
“If you add potential psychiatric side effects to the other side effects that can result from these medications, that adds up to a pretty important reason to be wary of taking them, particularly for the indication of COVID-19, where the level of evidence that it helps in any way is still quite weak,” Dr. Potash said.
In an interview, Remington Nevin, MD, MPH, DrPH, executive director at the Quinism Foundation, White River Junction, Vt., a nonprofit organization that supports and promotes education and research on disorders caused by poisoning by quinoline drugs; and faculty associate in the department of mental health at Johns Hopkins Bloomberg School of Public Health, said that the authors of the letter “are to be commended for their efforts in raising awareness of the potentially lasting and disabling psychiatric effects of chloroquine and hydroxychloroquine, which, as with similar effects from other synthetic quinoline antimalarials, have occasionally been overlooked or misattributed to other conditions.”
He added: “I have proposed that the chronic neuropsychiatric effects of this class of drug are best considered not as side effects but as signs and symptoms of a disorder known as chronic quinoline encephalopathy caused by poisoning of the central nervous system.”
Dr. Gressier and the other letter authors, Dr. Bose, and Dr. Potash have reported no relevant financial relationships. Dr. Nevin has been retained as a consultant and expert witness in legal cases involving claims of adverse effects from quinoline antimalarial drugs.
A version of this article originally appeared on Medscape.com.
Chloroquine may be associated with serious psychiatric side effects, even in patients with no family or personal history of psychiatric disorders, a new review suggests.
In a letter to the editor published online July 28 in The Journal of Clinical Psychiatry, the authors summarize data from several studies published as far back as 1993 and as recently as May 2020.
“In addition to previously reported side effects, chloroquine could also induce psychiatric side effects which are polymorphic and can persist even after stopping the drug,” lead author Florence Gressier, MD, PhD, CESP, Inserm, department of psychiatry, Le Kremlin Bicêtre, France, said in an interview.
“In COVID-19 patients who may still be [undergoing treatment] with chloroquine, close psychiatric assessment and monitoring should be performed,” she said.
Heated controversy
Following findings of a small French study that suggested efficacy in lowering the viral load in patients with COVID-19, President Donald Trump expressed optimism regarding the role of hydroxychloroquine in treating COVID-19, calling it a “game changer”.
Other studies, however, have called into question both the efficacy and the safety of hydroxychloroquine in treating COVID-19. On June 15, the Food and Drug Administration revoked the emergency use authorization it had given in March to chloroquine and hydroxychloroquine for the treatment of COVID-19.
Nevertheless, hydroxychloroquine continues to be prescribed for COVID-19. For example, an article that appeared in Click2Houston on June 15 quoted the chief medical officer of Houston’s United Memorial Center as saying he plans to continue prescribing hydroxychloroquine for patients with COVID-19 until he finds a better alternative.
As discussed in a Medscape expert commentary, a group of physicians who held a “white coat summit” in front of the U.S. Supreme Court building promoted the use of hydroxychloroquine for the treatment of COVID-19. The video of their summit was retweeted by President Trump and garnered millions of views before it was taken down by Twitter, Facebook, and YouTube.
Sudden onset
For the new review, “we wanted to alert the public and practitioners on the potentially psychiatric risks induced by chloroquine, as it could be taken as self-medication or potentially still prescribed,” Dr. Gressier said.
“We think the format of the letter to the editor allows information to be provided in a concise and clear manner,” she added.
According to the FDA’s Adverse Event Reporting System database, 12% of reported adverse events (520 of 4,336) following the use of chloroquine that occurred between the fourth quarter of 2012 and the fourth quarter of 2019 were neuropsychiatric. These events included amnesia, delirium, hallucinations, depression, and loss of consciousness, the authors write.
The researchers acknowledged that the incidence of psychiatric adverse effects associated with the use of chloroquine is “unclear in the absence of high-quality, randomized placebo-controlled trials of its safety.” Nevertheless, they pointed out that there have been reports of insomnia and depression when the drug was used as prophylaxis against malaria .
Moreover, some case series or case reports describe symptoms such as depression, anxiety, agitation, violent outburst, suicidal ideation, and psychosis in patients who have been treated with chloroquine for malaria, lupus erythematosus, and rheumatoid arthritis .
“In contrast to many other psychoses, chloroquine psychosis may be more affective and include prominent visual hallucinations, symptoms of derealization, and disorders of thought, with preserved insight,” the authors wrote.
They noted that the frequency of symptoms does not appear to be connected to the cumulative dose or the duration of treatment, and the onset of psychosis or other adverse effects is usually “sudden.”
In addition, they warn that the drug’s psychiatric effects may go unnoticed, especially because COVID-19 itself has been associated with neuropsychiatric symptoms, making it hard to distinguish between symptoms caused by the illness and those caused by the drug.
Although the psychiatric symptoms typically occur early after treatment initiation, some “subtle” symptoms might persist after stopping the drug, possibly owing to its “extremely long” half-life, the authors stated.
Dr. Gressier noted that practicing clinicians should look up reports about self-medication with chloroquine “and warn their patients about the risk induced by chloroquine.”
Safe but ‘not benign’
Nilanjana Bose, MD, MBA, a rheumatologist at the Rheumatology Center of Houston, said she uses hydroxychloroquine “all the time” in clinical practice to treat patients with rheumatic conditions.
“I cannot comment on whether it [hydroxychloroquine or chloroquine] is a potential prophylactic or treatment for COVID-19, but I can say that, from a safety point of view, as a rheumatologist who uses hydroxychloroquine at a dose of 400 mg/day, I do not think we need to worry about serious [psychiatric] side effects,” Dr. Bose said in an interview.
Because clinicians are trying all types of possible treatments for COVID-19, “if this medication has possible efficacy, it is a great medicine from a rheumatologic perspective and is safe,” she added.
Nevertheless, the drug is “not benign, and regular side effects will be there, and of course, higher doses will cause more side effects,” said Dr. Bose, who was not involved in authoring the letter.
She counsels patients about potential psychiatric side effects of hydroxychloroquine because some of her patients have complained about irritability, worsening anxiety and depression, and difficulty sleeping.
Be wary
James “Jimmy” Potash, MD, MPH, Henry Phipps Professor of Psychiatry and Behavioral Sciences, Johns Hopkins Medicine, Baltimore, said in an interview that the “take-home message of this letter is that serious psychiatric effects, psychotic illness in particular,” can occur in individuals who take chloroquine and hydroxychloroquine.
In addition, “these are potentially very concerning side effects that psychiatrists should be aware of,” noted Dr. Potash, department director and psychiatrist-in-chief at Johns Hopkins.
He said that one of his patients who had been “completely psychiatrically healthy” took chloroquine prophylactically prior to traveling overseas. After she began taking the drug, she had an episode of mania that resolved once she discontinued the medication and received treatment for the mania.
“If you add potential psychiatric side effects to the other side effects that can result from these medications, that adds up to a pretty important reason to be wary of taking them, particularly for the indication of COVID-19, where the level of evidence that it helps in any way is still quite weak,” Dr. Potash said.
In an interview, Remington Nevin, MD, MPH, DrPH, executive director at the Quinism Foundation, White River Junction, Vt., a nonprofit organization that supports and promotes education and research on disorders caused by poisoning by quinoline drugs; and faculty associate in the department of mental health at Johns Hopkins Bloomberg School of Public Health, said that the authors of the letter “are to be commended for their efforts in raising awareness of the potentially lasting and disabling psychiatric effects of chloroquine and hydroxychloroquine, which, as with similar effects from other synthetic quinoline antimalarials, have occasionally been overlooked or misattributed to other conditions.”
He added: “I have proposed that the chronic neuropsychiatric effects of this class of drug are best considered not as side effects but as signs and symptoms of a disorder known as chronic quinoline encephalopathy caused by poisoning of the central nervous system.”
Dr. Gressier and the other letter authors, Dr. Bose, and Dr. Potash have reported no relevant financial relationships. Dr. Nevin has been retained as a consultant and expert witness in legal cases involving claims of adverse effects from quinoline antimalarial drugs.
A version of this article originally appeared on Medscape.com.
Chloroquine may be associated with serious psychiatric side effects, even in patients with no family or personal history of psychiatric disorders, a new review suggests.
In a letter to the editor published online July 28 in The Journal of Clinical Psychiatry, the authors summarize data from several studies published as far back as 1993 and as recently as May 2020.
“In addition to previously reported side effects, chloroquine could also induce psychiatric side effects which are polymorphic and can persist even after stopping the drug,” lead author Florence Gressier, MD, PhD, CESP, Inserm, department of psychiatry, Le Kremlin Bicêtre, France, said in an interview.
“In COVID-19 patients who may still be [undergoing treatment] with chloroquine, close psychiatric assessment and monitoring should be performed,” she said.
Heated controversy
Following findings of a small French study that suggested efficacy in lowering the viral load in patients with COVID-19, President Donald Trump expressed optimism regarding the role of hydroxychloroquine in treating COVID-19, calling it a “game changer”.
Other studies, however, have called into question both the efficacy and the safety of hydroxychloroquine in treating COVID-19. On June 15, the Food and Drug Administration revoked the emergency use authorization it had given in March to chloroquine and hydroxychloroquine for the treatment of COVID-19.
Nevertheless, hydroxychloroquine continues to be prescribed for COVID-19. For example, an article that appeared in Click2Houston on June 15 quoted the chief medical officer of Houston’s United Memorial Center as saying he plans to continue prescribing hydroxychloroquine for patients with COVID-19 until he finds a better alternative.
As discussed in a Medscape expert commentary, a group of physicians who held a “white coat summit” in front of the U.S. Supreme Court building promoted the use of hydroxychloroquine for the treatment of COVID-19. The video of their summit was retweeted by President Trump and garnered millions of views before it was taken down by Twitter, Facebook, and YouTube.
Sudden onset
For the new review, “we wanted to alert the public and practitioners on the potentially psychiatric risks induced by chloroquine, as it could be taken as self-medication or potentially still prescribed,” Dr. Gressier said.
“We think the format of the letter to the editor allows information to be provided in a concise and clear manner,” she added.
According to the FDA’s Adverse Event Reporting System database, 12% of reported adverse events (520 of 4,336) following the use of chloroquine that occurred between the fourth quarter of 2012 and the fourth quarter of 2019 were neuropsychiatric. These events included amnesia, delirium, hallucinations, depression, and loss of consciousness, the authors write.
The researchers acknowledged that the incidence of psychiatric adverse effects associated with the use of chloroquine is “unclear in the absence of high-quality, randomized placebo-controlled trials of its safety.” Nevertheless, they pointed out that there have been reports of insomnia and depression when the drug was used as prophylaxis against malaria .
Moreover, some case series or case reports describe symptoms such as depression, anxiety, agitation, violent outburst, suicidal ideation, and psychosis in patients who have been treated with chloroquine for malaria, lupus erythematosus, and rheumatoid arthritis .
“In contrast to many other psychoses, chloroquine psychosis may be more affective and include prominent visual hallucinations, symptoms of derealization, and disorders of thought, with preserved insight,” the authors wrote.
They noted that the frequency of symptoms does not appear to be connected to the cumulative dose or the duration of treatment, and the onset of psychosis or other adverse effects is usually “sudden.”
In addition, they warn that the drug’s psychiatric effects may go unnoticed, especially because COVID-19 itself has been associated with neuropsychiatric symptoms, making it hard to distinguish between symptoms caused by the illness and those caused by the drug.
Although the psychiatric symptoms typically occur early after treatment initiation, some “subtle” symptoms might persist after stopping the drug, possibly owing to its “extremely long” half-life, the authors stated.
Dr. Gressier noted that practicing clinicians should look up reports about self-medication with chloroquine “and warn their patients about the risk induced by chloroquine.”
Safe but ‘not benign’
Nilanjana Bose, MD, MBA, a rheumatologist at the Rheumatology Center of Houston, said she uses hydroxychloroquine “all the time” in clinical practice to treat patients with rheumatic conditions.
“I cannot comment on whether it [hydroxychloroquine or chloroquine] is a potential prophylactic or treatment for COVID-19, but I can say that, from a safety point of view, as a rheumatologist who uses hydroxychloroquine at a dose of 400 mg/day, I do not think we need to worry about serious [psychiatric] side effects,” Dr. Bose said in an interview.
Because clinicians are trying all types of possible treatments for COVID-19, “if this medication has possible efficacy, it is a great medicine from a rheumatologic perspective and is safe,” she added.
Nevertheless, the drug is “not benign, and regular side effects will be there, and of course, higher doses will cause more side effects,” said Dr. Bose, who was not involved in authoring the letter.
She counsels patients about potential psychiatric side effects of hydroxychloroquine because some of her patients have complained about irritability, worsening anxiety and depression, and difficulty sleeping.
Be wary
James “Jimmy” Potash, MD, MPH, Henry Phipps Professor of Psychiatry and Behavioral Sciences, Johns Hopkins Medicine, Baltimore, said in an interview that the “take-home message of this letter is that serious psychiatric effects, psychotic illness in particular,” can occur in individuals who take chloroquine and hydroxychloroquine.
In addition, “these are potentially very concerning side effects that psychiatrists should be aware of,” noted Dr. Potash, department director and psychiatrist-in-chief at Johns Hopkins.
He said that one of his patients who had been “completely psychiatrically healthy” took chloroquine prophylactically prior to traveling overseas. After she began taking the drug, she had an episode of mania that resolved once she discontinued the medication and received treatment for the mania.
“If you add potential psychiatric side effects to the other side effects that can result from these medications, that adds up to a pretty important reason to be wary of taking them, particularly for the indication of COVID-19, where the level of evidence that it helps in any way is still quite weak,” Dr. Potash said.
In an interview, Remington Nevin, MD, MPH, DrPH, executive director at the Quinism Foundation, White River Junction, Vt., a nonprofit organization that supports and promotes education and research on disorders caused by poisoning by quinoline drugs; and faculty associate in the department of mental health at Johns Hopkins Bloomberg School of Public Health, said that the authors of the letter “are to be commended for their efforts in raising awareness of the potentially lasting and disabling psychiatric effects of chloroquine and hydroxychloroquine, which, as with similar effects from other synthetic quinoline antimalarials, have occasionally been overlooked or misattributed to other conditions.”
He added: “I have proposed that the chronic neuropsychiatric effects of this class of drug are best considered not as side effects but as signs and symptoms of a disorder known as chronic quinoline encephalopathy caused by poisoning of the central nervous system.”
Dr. Gressier and the other letter authors, Dr. Bose, and Dr. Potash have reported no relevant financial relationships. Dr. Nevin has been retained as a consultant and expert witness in legal cases involving claims of adverse effects from quinoline antimalarial drugs.
A version of this article originally appeared on Medscape.com.
Botulinum toxin associated with antidepressant effects across indications, injection sites
, according to the study’s authors.
Their results show that the antidepressant effect of botulinum toxin “administered for various indications goes beyond the control of the intended disease states and does not depend on the location of the injection,” according to Tigran Makunts, PharmD, of the Skaggs School of Pharmacy and Pharmaceutical Sciences at the University of California, San Diego, and coauthors.
Previous high-quality studies have found botulinum toxin treatment has been associated with antidepressant effects when administered to the glabellar region of the face, they noted. The study was published in Scientific Reports.
The researchers evaluated adverse events reported to the Food and Drug Administration’s current adverse event reporting system (FAERS) between September 2012 and December 2019, and the FDA’s previous adverse event reporting system between January 2004 and August 2012. Overall, they analyzed 174,243 reports, which were divided into eight treatment-related groups based on the indication for botulinum toxin: Cosmetic use (20,684 patients), migraine (4,180 patients), spasms and spasticity not involving facial muscles (2,335 patients), neurological and urinary bladder disorders (915 patients), torticollis (1,360 patients), hyperhidrosis (601 patients), blepharospasm (487 patients), and sialorrhea (157 patients). Each group was matched to controls from the FAERS database, who had different treatments for the same indications. (Reports in which patients were on an antidepressant or where depression was listed as an indication were not included).
In nearly all treatment groups, reports of depression and depression-related adverse events were significantly lower among those who received botulinum toxin, compared with controls: For those who received botulinum toxin injections in facial muscles for cosmetic uses, the reporting odds ratio was 0.46 (95% confidence interval, 0.27-0.78). Significant effects were also see in the following groups: those who received injections into facial and head muscles for migraine (ROR, 0.60; 95% CI, 0.48-0.74), injections into the upper and lower limbs for spasms and spasticity (ROR, 0.28; 95% CI, 0.18-0.42), injections into neck muscles for torticollis and neck pain (ROR, 0.30; 95% CI, 0.20-0.44), injections into eyelid muscles for blepharospasm (ROR, 0.13; 95% CI, 0.05-0.39), and injections into the axilla and palm for hyperhidrosis (ROR, 0.12; 95% CI, 0.04-0.33).
There were no cases of depression or depression-related adverse event reports among those treated with botulinum toxin for sialorrhea with injections into the parotid and submandibular glands, and there were decreased reports of depression among those who received detrusor muscle injections for neurological and urinary bladder disorders, but the results in both groups were not statistically significant, according to the researchers.
In an interview, Ruben Abagyan, PhD, study coauthor and professor at Skaggs School of Pharmacy and Pharmaceutical Sciences, said the study’s finding go “beyond breaking a positive feedback loop between depression and the ‘frown’ wrinkles in the glabellar region of the forehead.” The data showing efficacy with botulinum toxin injected in other areas of the body can help clinicians “expand their search for the most effective injection location and dose beyond the facial injections to improve the depression-related therapeutic outcomes.”
Another takeaway from the study, he noted, is that botulinum toxin can have effects beyond the local effect seen near an injection site. Administering botulinum toxin for spasms and spasticity, excessive sweating, migraine, urinary bladder disorders, blepharospasm, or excessive salivation/drooling could result in reduced depression and improved systemic neurological effects.
“Severe depression remains a very difficult condition to treat. The existing drugs have dangerous side effects, the onset of the therapeutic action is delayed by at least a month, and the adherence to the medication is suboptimal. Therefore, finding new ways to treat depression is critical,” Dr. Abagyan said. “Botulinum toxin opens up a new physiological mechanism to be tried to reduce depression.”
Michelle Magid, MD, MBA, of the department of psychiatry at the University of Texas at Austin, said in an interview that, although the study was retrospective, “physicians can feel confident that botulinum toxin treatment will not cause depression; it may very well lead to improved mood in some of their patients.” Dr. Magid was not an author of this study, but has studied botulinum toxin as a possible treatment of depression.
“Previous studies have shown that botulinum toxin injected into the forehead region can improve symptoms of depression. The studies were small and confined to treating the glabellar region only,” she added. “This is a large retrospective study showing that botulinum toxin injected into other regions, such as the neck, underarms, bladder, hands, arms, and legs, can also have an antidepressant effect.”
Dr. Magid agreed that the use of botulinum toxin as an antidepressant should be investigated further, and could be a tool for patients who do not respond well to traditional antidepressant medications.
In their paper, the authors offered several plausible mechanisms for the antidepressant effects of botulinum toxin, including transneuronal transport to the parts of the central nervous system that regulate mood and emotion, systemic distribution, distributed muscle stress memory, and efficacy in the primary indication treatment. Although the mechanism of action is not well understood, Dr. Magid noted it could be the removal of somatic symptoms that contribute to an improvement in mood.
“It is possible that alleviating the psychological distress associated with neck spasms, excessive sweating [and so on] can be causing the antidepressive effects,” she said. “However, it is also possible that depression is actualized by a series of somatic symptoms – body aches, insomnia, sweating, for example. By removing somatic symptoms, one may also remove the correlating mood dysregulation.”
The study “certainly raises a lot of questions,” particularly about the “apparent multiple mechanisms of action of BoNT that we don’t understand yet,” Mark Rubin, MD, a cosmetic dermatologist who practices in Beverly Hills, Calif., said in an interview. “I believe it lends great deal of credence to the use of [botulinum toxin] for depression and certainly validates the need for more robust clinical trials for that indication,” he added.
“I think what we all as clinicians need to take away from this paper is that there is a great deal we don’t understand about the global pharmacologic effects of [botulinum toxin] and equally important, that there are apparently other pharmacologic pathways we need to explore in the treatment of depression, said Dr. Rubin, of the department of dermatology at the University of California, San Diego, who was not an investigator in the study.
One author reported being a consultant for Allergan. Dr. Makunts and the other author report no relevant conflicts of interest; Dr. Magid reported being a consultant for Allergan and a speaker for Ipsen. Dr. Rubin had no related disclosures.
SOURCE: Makunts T et al. Sci Rep. 2020 Jul 30;10(1):12851. doi: 10.1038/s41598-020-69773-7.
, according to the study’s authors.
Their results show that the antidepressant effect of botulinum toxin “administered for various indications goes beyond the control of the intended disease states and does not depend on the location of the injection,” according to Tigran Makunts, PharmD, of the Skaggs School of Pharmacy and Pharmaceutical Sciences at the University of California, San Diego, and coauthors.
Previous high-quality studies have found botulinum toxin treatment has been associated with antidepressant effects when administered to the glabellar region of the face, they noted. The study was published in Scientific Reports.
The researchers evaluated adverse events reported to the Food and Drug Administration’s current adverse event reporting system (FAERS) between September 2012 and December 2019, and the FDA’s previous adverse event reporting system between January 2004 and August 2012. Overall, they analyzed 174,243 reports, which were divided into eight treatment-related groups based on the indication for botulinum toxin: Cosmetic use (20,684 patients), migraine (4,180 patients), spasms and spasticity not involving facial muscles (2,335 patients), neurological and urinary bladder disorders (915 patients), torticollis (1,360 patients), hyperhidrosis (601 patients), blepharospasm (487 patients), and sialorrhea (157 patients). Each group was matched to controls from the FAERS database, who had different treatments for the same indications. (Reports in which patients were on an antidepressant or where depression was listed as an indication were not included).
In nearly all treatment groups, reports of depression and depression-related adverse events were significantly lower among those who received botulinum toxin, compared with controls: For those who received botulinum toxin injections in facial muscles for cosmetic uses, the reporting odds ratio was 0.46 (95% confidence interval, 0.27-0.78). Significant effects were also see in the following groups: those who received injections into facial and head muscles for migraine (ROR, 0.60; 95% CI, 0.48-0.74), injections into the upper and lower limbs for spasms and spasticity (ROR, 0.28; 95% CI, 0.18-0.42), injections into neck muscles for torticollis and neck pain (ROR, 0.30; 95% CI, 0.20-0.44), injections into eyelid muscles for blepharospasm (ROR, 0.13; 95% CI, 0.05-0.39), and injections into the axilla and palm for hyperhidrosis (ROR, 0.12; 95% CI, 0.04-0.33).
There were no cases of depression or depression-related adverse event reports among those treated with botulinum toxin for sialorrhea with injections into the parotid and submandibular glands, and there were decreased reports of depression among those who received detrusor muscle injections for neurological and urinary bladder disorders, but the results in both groups were not statistically significant, according to the researchers.
In an interview, Ruben Abagyan, PhD, study coauthor and professor at Skaggs School of Pharmacy and Pharmaceutical Sciences, said the study’s finding go “beyond breaking a positive feedback loop between depression and the ‘frown’ wrinkles in the glabellar region of the forehead.” The data showing efficacy with botulinum toxin injected in other areas of the body can help clinicians “expand their search for the most effective injection location and dose beyond the facial injections to improve the depression-related therapeutic outcomes.”
Another takeaway from the study, he noted, is that botulinum toxin can have effects beyond the local effect seen near an injection site. Administering botulinum toxin for spasms and spasticity, excessive sweating, migraine, urinary bladder disorders, blepharospasm, or excessive salivation/drooling could result in reduced depression and improved systemic neurological effects.
“Severe depression remains a very difficult condition to treat. The existing drugs have dangerous side effects, the onset of the therapeutic action is delayed by at least a month, and the adherence to the medication is suboptimal. Therefore, finding new ways to treat depression is critical,” Dr. Abagyan said. “Botulinum toxin opens up a new physiological mechanism to be tried to reduce depression.”
Michelle Magid, MD, MBA, of the department of psychiatry at the University of Texas at Austin, said in an interview that, although the study was retrospective, “physicians can feel confident that botulinum toxin treatment will not cause depression; it may very well lead to improved mood in some of their patients.” Dr. Magid was not an author of this study, but has studied botulinum toxin as a possible treatment of depression.
“Previous studies have shown that botulinum toxin injected into the forehead region can improve symptoms of depression. The studies were small and confined to treating the glabellar region only,” she added. “This is a large retrospective study showing that botulinum toxin injected into other regions, such as the neck, underarms, bladder, hands, arms, and legs, can also have an antidepressant effect.”
Dr. Magid agreed that the use of botulinum toxin as an antidepressant should be investigated further, and could be a tool for patients who do not respond well to traditional antidepressant medications.
In their paper, the authors offered several plausible mechanisms for the antidepressant effects of botulinum toxin, including transneuronal transport to the parts of the central nervous system that regulate mood and emotion, systemic distribution, distributed muscle stress memory, and efficacy in the primary indication treatment. Although the mechanism of action is not well understood, Dr. Magid noted it could be the removal of somatic symptoms that contribute to an improvement in mood.
“It is possible that alleviating the psychological distress associated with neck spasms, excessive sweating [and so on] can be causing the antidepressive effects,” she said. “However, it is also possible that depression is actualized by a series of somatic symptoms – body aches, insomnia, sweating, for example. By removing somatic symptoms, one may also remove the correlating mood dysregulation.”
The study “certainly raises a lot of questions,” particularly about the “apparent multiple mechanisms of action of BoNT that we don’t understand yet,” Mark Rubin, MD, a cosmetic dermatologist who practices in Beverly Hills, Calif., said in an interview. “I believe it lends great deal of credence to the use of [botulinum toxin] for depression and certainly validates the need for more robust clinical trials for that indication,” he added.
“I think what we all as clinicians need to take away from this paper is that there is a great deal we don’t understand about the global pharmacologic effects of [botulinum toxin] and equally important, that there are apparently other pharmacologic pathways we need to explore in the treatment of depression, said Dr. Rubin, of the department of dermatology at the University of California, San Diego, who was not an investigator in the study.
One author reported being a consultant for Allergan. Dr. Makunts and the other author report no relevant conflicts of interest; Dr. Magid reported being a consultant for Allergan and a speaker for Ipsen. Dr. Rubin had no related disclosures.
SOURCE: Makunts T et al. Sci Rep. 2020 Jul 30;10(1):12851. doi: 10.1038/s41598-020-69773-7.
, according to the study’s authors.
Their results show that the antidepressant effect of botulinum toxin “administered for various indications goes beyond the control of the intended disease states and does not depend on the location of the injection,” according to Tigran Makunts, PharmD, of the Skaggs School of Pharmacy and Pharmaceutical Sciences at the University of California, San Diego, and coauthors.
Previous high-quality studies have found botulinum toxin treatment has been associated with antidepressant effects when administered to the glabellar region of the face, they noted. The study was published in Scientific Reports.
The researchers evaluated adverse events reported to the Food and Drug Administration’s current adverse event reporting system (FAERS) between September 2012 and December 2019, and the FDA’s previous adverse event reporting system between January 2004 and August 2012. Overall, they analyzed 174,243 reports, which were divided into eight treatment-related groups based on the indication for botulinum toxin: Cosmetic use (20,684 patients), migraine (4,180 patients), spasms and spasticity not involving facial muscles (2,335 patients), neurological and urinary bladder disorders (915 patients), torticollis (1,360 patients), hyperhidrosis (601 patients), blepharospasm (487 patients), and sialorrhea (157 patients). Each group was matched to controls from the FAERS database, who had different treatments for the same indications. (Reports in which patients were on an antidepressant or where depression was listed as an indication were not included).
In nearly all treatment groups, reports of depression and depression-related adverse events were significantly lower among those who received botulinum toxin, compared with controls: For those who received botulinum toxin injections in facial muscles for cosmetic uses, the reporting odds ratio was 0.46 (95% confidence interval, 0.27-0.78). Significant effects were also see in the following groups: those who received injections into facial and head muscles for migraine (ROR, 0.60; 95% CI, 0.48-0.74), injections into the upper and lower limbs for spasms and spasticity (ROR, 0.28; 95% CI, 0.18-0.42), injections into neck muscles for torticollis and neck pain (ROR, 0.30; 95% CI, 0.20-0.44), injections into eyelid muscles for blepharospasm (ROR, 0.13; 95% CI, 0.05-0.39), and injections into the axilla and palm for hyperhidrosis (ROR, 0.12; 95% CI, 0.04-0.33).
There were no cases of depression or depression-related adverse event reports among those treated with botulinum toxin for sialorrhea with injections into the parotid and submandibular glands, and there were decreased reports of depression among those who received detrusor muscle injections for neurological and urinary bladder disorders, but the results in both groups were not statistically significant, according to the researchers.
In an interview, Ruben Abagyan, PhD, study coauthor and professor at Skaggs School of Pharmacy and Pharmaceutical Sciences, said the study’s finding go “beyond breaking a positive feedback loop between depression and the ‘frown’ wrinkles in the glabellar region of the forehead.” The data showing efficacy with botulinum toxin injected in other areas of the body can help clinicians “expand their search for the most effective injection location and dose beyond the facial injections to improve the depression-related therapeutic outcomes.”
Another takeaway from the study, he noted, is that botulinum toxin can have effects beyond the local effect seen near an injection site. Administering botulinum toxin for spasms and spasticity, excessive sweating, migraine, urinary bladder disorders, blepharospasm, or excessive salivation/drooling could result in reduced depression and improved systemic neurological effects.
“Severe depression remains a very difficult condition to treat. The existing drugs have dangerous side effects, the onset of the therapeutic action is delayed by at least a month, and the adherence to the medication is suboptimal. Therefore, finding new ways to treat depression is critical,” Dr. Abagyan said. “Botulinum toxin opens up a new physiological mechanism to be tried to reduce depression.”
Michelle Magid, MD, MBA, of the department of psychiatry at the University of Texas at Austin, said in an interview that, although the study was retrospective, “physicians can feel confident that botulinum toxin treatment will not cause depression; it may very well lead to improved mood in some of their patients.” Dr. Magid was not an author of this study, but has studied botulinum toxin as a possible treatment of depression.
“Previous studies have shown that botulinum toxin injected into the forehead region can improve symptoms of depression. The studies were small and confined to treating the glabellar region only,” she added. “This is a large retrospective study showing that botulinum toxin injected into other regions, such as the neck, underarms, bladder, hands, arms, and legs, can also have an antidepressant effect.”
Dr. Magid agreed that the use of botulinum toxin as an antidepressant should be investigated further, and could be a tool for patients who do not respond well to traditional antidepressant medications.
In their paper, the authors offered several plausible mechanisms for the antidepressant effects of botulinum toxin, including transneuronal transport to the parts of the central nervous system that regulate mood and emotion, systemic distribution, distributed muscle stress memory, and efficacy in the primary indication treatment. Although the mechanism of action is not well understood, Dr. Magid noted it could be the removal of somatic symptoms that contribute to an improvement in mood.
“It is possible that alleviating the psychological distress associated with neck spasms, excessive sweating [and so on] can be causing the antidepressive effects,” she said. “However, it is also possible that depression is actualized by a series of somatic symptoms – body aches, insomnia, sweating, for example. By removing somatic symptoms, one may also remove the correlating mood dysregulation.”
The study “certainly raises a lot of questions,” particularly about the “apparent multiple mechanisms of action of BoNT that we don’t understand yet,” Mark Rubin, MD, a cosmetic dermatologist who practices in Beverly Hills, Calif., said in an interview. “I believe it lends great deal of credence to the use of [botulinum toxin] for depression and certainly validates the need for more robust clinical trials for that indication,” he added.
“I think what we all as clinicians need to take away from this paper is that there is a great deal we don’t understand about the global pharmacologic effects of [botulinum toxin] and equally important, that there are apparently other pharmacologic pathways we need to explore in the treatment of depression, said Dr. Rubin, of the department of dermatology at the University of California, San Diego, who was not an investigator in the study.
One author reported being a consultant for Allergan. Dr. Makunts and the other author report no relevant conflicts of interest; Dr. Magid reported being a consultant for Allergan and a speaker for Ipsen. Dr. Rubin had no related disclosures.
SOURCE: Makunts T et al. Sci Rep. 2020 Jul 30;10(1):12851. doi: 10.1038/s41598-020-69773-7.
FROM SCIENTIFIC REPORTS
Vitamin D fails to prevent late-life depression, boost mood
Findings from a large randomized, controlled trial do not support the use of vitamin D3 supplementation for adults for the sole purpose of preventing depression.
Among adults aged 50 years or older who were without clinically relevant depressive symptoms at baseline, vitamin D3 supplementation taken over 5 years did not reduce the risk for depression or make a difference in the quality of mood.
“The study is among the largest of its kind ever, and it was able to address whether vitamin D3 supplementation is useful for what we call ‘universal prevention’ of depression,” Olivia Okereke, MD, Massachusetts General Hospital, Boston, said in an interview.
“These results tell us that there is no benefit to using vitamin D3 supplements for the sole purpose of preventing depression in the general population of middle-aged and older adults,” said Dr. Okereke.
“Because of the high dose and long duration of treatment and the randomized placebo-controlled design, we can have high confidence in results,” she added.
The study was published online August 4 in JAMA.
The VITAL-DEP trial
The findings are based on 18,353 older adults (mean age, 67.5 years; 49% women) in the VITAL-DEP study; 16,657 were at risk for incident depression (ie, had no history of depression), and 1696 were at risk for recurrent depression (i.e., had a history of depression but had not undergone treatment for depression within the past 2 years).
Roughly half were randomly allocated to receive vitamin D3 (2000 IU/d of cholecalciferol) and half to receive matching placebo for a median of 5.3 years. The participants’ mean level of 25-hydroxyvitamin D was 31.1 ng/mL; for about 12%, levels were lower than 20 ng/mL.
The risk for depression or clinically relevant depressive symptoms (total of incident and recurrent cases) was not significantly different between the vitamin D3 group (609 depression or clinically relevant depressive symptom events; 12.9/1000 person-years) and the placebo group (625 depression or clinically relevant depressive symptom events; 13.3/1000 person-years). The hazard ratio was 0.97 (95% confidence interval, 0.87-1.09; P = .62).
“Cumulative incidence curves showed lack of separation between treatment groups over the entire follow-up,” the researchers report.
There was also no significant between-group difference in the other primary outcome – the mean difference in mood scores on the eight-item Patient Health Questionnaire depression scale (PHQ-8).
The mean difference for change between treatment groups in PHQ-8 scores was not significantly different from 0 over the entire follow-up (0.01 points; 95% CI, −0.04 to 0.05 points) or at any point during follow-up.
To date, 13 randomized clinical trials have examined the effects of vitamin D3 supplementation on depression or mood during middle age or in older adults, and all except one reported null findings, Dr. Okereke and colleagues noted in their article.
The current study is the only one large enough to examine vitamin D3 supplementation for the universal prevention of depression, they point out.
Although the findings do not support vitamin D3 supplementation for depression prevention, Dr. Okereke said, We also know that vitamin D is essential for bone health, and this study does not tell us whether vitamin D3 is useful for prevention of other health outcomes.”
VITAL-DEP was supported by a grant from the National Institute of Mental Health. Pharmavite donated the vitamin D3, matching placebos, and packaging in the form of calendar packs. Dr. Okereke reported receiving royalties from Springer Publishing for a book on the prevention of late-life depression.
This article first appeared on Medscape.com.
Findings from a large randomized, controlled trial do not support the use of vitamin D3 supplementation for adults for the sole purpose of preventing depression.
Among adults aged 50 years or older who were without clinically relevant depressive symptoms at baseline, vitamin D3 supplementation taken over 5 years did not reduce the risk for depression or make a difference in the quality of mood.
“The study is among the largest of its kind ever, and it was able to address whether vitamin D3 supplementation is useful for what we call ‘universal prevention’ of depression,” Olivia Okereke, MD, Massachusetts General Hospital, Boston, said in an interview.
“These results tell us that there is no benefit to using vitamin D3 supplements for the sole purpose of preventing depression in the general population of middle-aged and older adults,” said Dr. Okereke.
“Because of the high dose and long duration of treatment and the randomized placebo-controlled design, we can have high confidence in results,” she added.
The study was published online August 4 in JAMA.
The VITAL-DEP trial
The findings are based on 18,353 older adults (mean age, 67.5 years; 49% women) in the VITAL-DEP study; 16,657 were at risk for incident depression (ie, had no history of depression), and 1696 were at risk for recurrent depression (i.e., had a history of depression but had not undergone treatment for depression within the past 2 years).
Roughly half were randomly allocated to receive vitamin D3 (2000 IU/d of cholecalciferol) and half to receive matching placebo for a median of 5.3 years. The participants’ mean level of 25-hydroxyvitamin D was 31.1 ng/mL; for about 12%, levels were lower than 20 ng/mL.
The risk for depression or clinically relevant depressive symptoms (total of incident and recurrent cases) was not significantly different between the vitamin D3 group (609 depression or clinically relevant depressive symptom events; 12.9/1000 person-years) and the placebo group (625 depression or clinically relevant depressive symptom events; 13.3/1000 person-years). The hazard ratio was 0.97 (95% confidence interval, 0.87-1.09; P = .62).
“Cumulative incidence curves showed lack of separation between treatment groups over the entire follow-up,” the researchers report.
There was also no significant between-group difference in the other primary outcome – the mean difference in mood scores on the eight-item Patient Health Questionnaire depression scale (PHQ-8).
The mean difference for change between treatment groups in PHQ-8 scores was not significantly different from 0 over the entire follow-up (0.01 points; 95% CI, −0.04 to 0.05 points) or at any point during follow-up.
To date, 13 randomized clinical trials have examined the effects of vitamin D3 supplementation on depression or mood during middle age or in older adults, and all except one reported null findings, Dr. Okereke and colleagues noted in their article.
The current study is the only one large enough to examine vitamin D3 supplementation for the universal prevention of depression, they point out.
Although the findings do not support vitamin D3 supplementation for depression prevention, Dr. Okereke said, We also know that vitamin D is essential for bone health, and this study does not tell us whether vitamin D3 is useful for prevention of other health outcomes.”
VITAL-DEP was supported by a grant from the National Institute of Mental Health. Pharmavite donated the vitamin D3, matching placebos, and packaging in the form of calendar packs. Dr. Okereke reported receiving royalties from Springer Publishing for a book on the prevention of late-life depression.
This article first appeared on Medscape.com.
Findings from a large randomized, controlled trial do not support the use of vitamin D3 supplementation for adults for the sole purpose of preventing depression.
Among adults aged 50 years or older who were without clinically relevant depressive symptoms at baseline, vitamin D3 supplementation taken over 5 years did not reduce the risk for depression or make a difference in the quality of mood.
“The study is among the largest of its kind ever, and it was able to address whether vitamin D3 supplementation is useful for what we call ‘universal prevention’ of depression,” Olivia Okereke, MD, Massachusetts General Hospital, Boston, said in an interview.
“These results tell us that there is no benefit to using vitamin D3 supplements for the sole purpose of preventing depression in the general population of middle-aged and older adults,” said Dr. Okereke.
“Because of the high dose and long duration of treatment and the randomized placebo-controlled design, we can have high confidence in results,” she added.
The study was published online August 4 in JAMA.
The VITAL-DEP trial
The findings are based on 18,353 older adults (mean age, 67.5 years; 49% women) in the VITAL-DEP study; 16,657 were at risk for incident depression (ie, had no history of depression), and 1696 were at risk for recurrent depression (i.e., had a history of depression but had not undergone treatment for depression within the past 2 years).
Roughly half were randomly allocated to receive vitamin D3 (2000 IU/d of cholecalciferol) and half to receive matching placebo for a median of 5.3 years. The participants’ mean level of 25-hydroxyvitamin D was 31.1 ng/mL; for about 12%, levels were lower than 20 ng/mL.
The risk for depression or clinically relevant depressive symptoms (total of incident and recurrent cases) was not significantly different between the vitamin D3 group (609 depression or clinically relevant depressive symptom events; 12.9/1000 person-years) and the placebo group (625 depression or clinically relevant depressive symptom events; 13.3/1000 person-years). The hazard ratio was 0.97 (95% confidence interval, 0.87-1.09; P = .62).
“Cumulative incidence curves showed lack of separation between treatment groups over the entire follow-up,” the researchers report.
There was also no significant between-group difference in the other primary outcome – the mean difference in mood scores on the eight-item Patient Health Questionnaire depression scale (PHQ-8).
The mean difference for change between treatment groups in PHQ-8 scores was not significantly different from 0 over the entire follow-up (0.01 points; 95% CI, −0.04 to 0.05 points) or at any point during follow-up.
To date, 13 randomized clinical trials have examined the effects of vitamin D3 supplementation on depression or mood during middle age or in older adults, and all except one reported null findings, Dr. Okereke and colleagues noted in their article.
The current study is the only one large enough to examine vitamin D3 supplementation for the universal prevention of depression, they point out.
Although the findings do not support vitamin D3 supplementation for depression prevention, Dr. Okereke said, We also know that vitamin D is essential for bone health, and this study does not tell us whether vitamin D3 is useful for prevention of other health outcomes.”
VITAL-DEP was supported by a grant from the National Institute of Mental Health. Pharmavite donated the vitamin D3, matching placebos, and packaging in the form of calendar packs. Dr. Okereke reported receiving royalties from Springer Publishing for a book on the prevention of late-life depression.
This article first appeared on Medscape.com.
ED visits for mental health, substance use doubled in 1 decade
ED visits related to mental health conditions increased nearly twofold from 2007-2008 to 2015-2016, new research suggests.
Data from the National Hospital Ambulatory Medical Care Survey (NHAMCS) showed that, over the 10-year study period, the proportion of ED visits for mental health diagnoses increased from 6.6% to 10.9%, with substance use accounting for much of the increase.
Although there have been policy efforts, such as expanding access to mental health care as part of the Affordable Care Act (ACA) of 2011, the senior author Taeho Greg Rhee, PhD, MSW, said in an interview.
“Treating mental health conditions in EDs is often considered suboptimal” because of limited time for full psychiatric assessment, lack of trained providers, and limited privacy in EDs, said Dr. Rhee of Yale University, New Haven, Conn.
The findings were published online July 28 in The Journal of Clinical Psychiatry.
“Outdated” research
Roughly one-fifth of U.S. adults experience some type of mental, behavioral, or emotional disorder annually. Moreover, the suicide rate has been steadily increasing, and there continues to be a “raging opioid epidemic,” the researchers wrote.
Despite these alarming figures, 57.4% of adults with mental illness reported in 2017 that they had not received any mental health treatment in the past year, reported the investigators.
Previous research has suggested that many adults have difficulty seeking outpatient mental health treatment and may turn to EDs instead. However, most studies of mental health ED use “are by now outdated, as they used data from years prior to the full implementation of the ACA,” the researchers noted.
“More Americans are suffering from mental illness, and given the recent policy efforts of expanding access to mental health care, we were questioning if ED visits due to mental health has changed or not,” Dr. Rhee said.
To investigate the question, the researchers conducted a cross-sectional analysis of data from the NHAMCS, a publicly available dataset provided by the National Center for Health Statistics of the Centers for Disease Control and Prevention.
They grouped psychiatric diagnoses into five categories: mood disorders, anxiety disorders, psychosis or schizophrenia, suicide attempt or ideation, or other/unspecified. Substance use diagnoses were grouped into six categories: alcohol, amphetamine, cannabis, cocaine, opioid, or other/unspecified.
These categories were used to determine the type of disorder a patient had, whether the patient had both psychiatric and substance-related diagnoses, and whether the patient received multiple mental health diagnoses at the time of the ED visit.
Sociodemographic covariates included age, sex, race/ethnicity, and insurance coverage.
Twofold and fourfold increases
Of 100.9 million outpatient ED visits that took place between 2007 and 2016, approximately 8.4 million (8.3%) were for psychiatric or substance use–related diagnoses. Also, the visits were more likely from adults who were younger than 45 years, male, non-Hispanic White, and covered by Medicaid or other public insurance types (58.5%, 52.5%, 65.2%, and 58.6%, respectively).
The overall rate of ED visits for any mental health diagnosis nearly doubled between 2007-2008 and 2015-2016. The rate of visits in which both psychiatric and substance use–related diagnoses increased fourfold during that time span. ED visits involving at least two mental health diagnoses increased twofold.
Additional changes in the number of visits are listed below (for each, P < .001).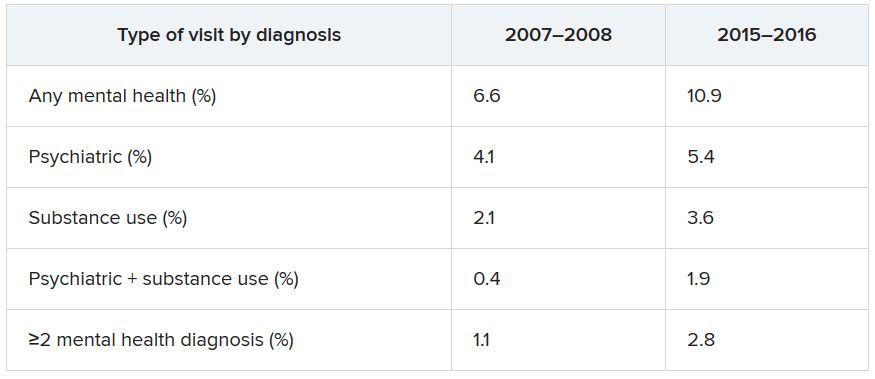
When these comparisons were adjusted for age, sex, and race/ethnicity, “linearly increasing trends of mental health–related ED visits were consistently found in all categories,” the authors reported. No trends were found regarding age, sex, or race/ethnicity. By contrast, mental health–related ED visits in which Medicaid was identified as the primary source of insurance nearly doubled between 2007–2008 and 2015–2016 (from 27.2% to 42.8%).
Other/unspecified psychiatric diagnoses, such as adjustment disorder and personality disorders, almost tripled between 2007-2008 and 2015-2016 (from 1,040 to 2,961 per 100,000 ED visits). ED visits for mood disorders and anxiety disorders also increased over time.
Alcohol-related ED visits were the most common substance use visits, increasing from 1,669 in 2007-2008 to 3,007 per 100,000 visits in 2015-2016. Amphetamine- and opioid-related ED visits more than doubled, and other/unspecified–related ED visits more than tripled during that time.
“One explanation why ED visits for mental health conditions have increased is that substance-related problems, which include overdose/self-injury issues, have increased over time,” Dr. Rhee noted, which “makes sense,” inasmuch as opioid, cannabis, and amphetamine use has increased across the country.
Another explanation is that, although mental health care access has been expanded through the ACA, “people, especially those with lower socioeconomic backgrounds, do not know how to get access to care and are still underserved,” he said.
“If mental health–related ED visits continue to increase in the future, there are several steps to be made. ED providers need to be better equipped with mental health care, and behavioral health should be better integrated as part of the care coordination,” said Dr. Rhee.
He added that reimbursement models across different insurance types, such as Medicare, Medicaid, and private insurance, “should consider expanding their coverage of mental health treatment in ED settings.”
“Canary in the coal mine”
Commenting on the study in an interview, Benjamin Druss, MD, MPH, professor and Rosalynn Carter Chair in Mental Health, Rollins School of Public Health, Emory University, Atlanta, called EDs the “canaries in the coal mine” for the broader health system.
The growing number of ED visits for behavioral problems “could represent both a rise in acute conditions such as substance use and lack of access to outpatient treatment,” said Dr. Druss, who was not involved with the research.
The findings “suggest the importance of strategies to effectively manage patients with behavioral conditions in ED settings and to effectively link them with high-quality outpatient care,” he noted.
Dr. Rhee has received funding from the National Institute on Aging and the American Foundation for Suicide Prevention. The other study authors and Dr. Druss report no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
ED visits related to mental health conditions increased nearly twofold from 2007-2008 to 2015-2016, new research suggests.
Data from the National Hospital Ambulatory Medical Care Survey (NHAMCS) showed that, over the 10-year study period, the proportion of ED visits for mental health diagnoses increased from 6.6% to 10.9%, with substance use accounting for much of the increase.
Although there have been policy efforts, such as expanding access to mental health care as part of the Affordable Care Act (ACA) of 2011, the senior author Taeho Greg Rhee, PhD, MSW, said in an interview.
“Treating mental health conditions in EDs is often considered suboptimal” because of limited time for full psychiatric assessment, lack of trained providers, and limited privacy in EDs, said Dr. Rhee of Yale University, New Haven, Conn.
The findings were published online July 28 in The Journal of Clinical Psychiatry.
“Outdated” research
Roughly one-fifth of U.S. adults experience some type of mental, behavioral, or emotional disorder annually. Moreover, the suicide rate has been steadily increasing, and there continues to be a “raging opioid epidemic,” the researchers wrote.
Despite these alarming figures, 57.4% of adults with mental illness reported in 2017 that they had not received any mental health treatment in the past year, reported the investigators.
Previous research has suggested that many adults have difficulty seeking outpatient mental health treatment and may turn to EDs instead. However, most studies of mental health ED use “are by now outdated, as they used data from years prior to the full implementation of the ACA,” the researchers noted.
“More Americans are suffering from mental illness, and given the recent policy efforts of expanding access to mental health care, we were questioning if ED visits due to mental health has changed or not,” Dr. Rhee said.
To investigate the question, the researchers conducted a cross-sectional analysis of data from the NHAMCS, a publicly available dataset provided by the National Center for Health Statistics of the Centers for Disease Control and Prevention.
They grouped psychiatric diagnoses into five categories: mood disorders, anxiety disorders, psychosis or schizophrenia, suicide attempt or ideation, or other/unspecified. Substance use diagnoses were grouped into six categories: alcohol, amphetamine, cannabis, cocaine, opioid, or other/unspecified.
These categories were used to determine the type of disorder a patient had, whether the patient had both psychiatric and substance-related diagnoses, and whether the patient received multiple mental health diagnoses at the time of the ED visit.
Sociodemographic covariates included age, sex, race/ethnicity, and insurance coverage.
Twofold and fourfold increases
Of 100.9 million outpatient ED visits that took place between 2007 and 2016, approximately 8.4 million (8.3%) were for psychiatric or substance use–related diagnoses. Also, the visits were more likely from adults who were younger than 45 years, male, non-Hispanic White, and covered by Medicaid or other public insurance types (58.5%, 52.5%, 65.2%, and 58.6%, respectively).
The overall rate of ED visits for any mental health diagnosis nearly doubled between 2007-2008 and 2015-2016. The rate of visits in which both psychiatric and substance use–related diagnoses increased fourfold during that time span. ED visits involving at least two mental health diagnoses increased twofold.
Additional changes in the number of visits are listed below (for each, P < .001).
When these comparisons were adjusted for age, sex, and race/ethnicity, “linearly increasing trends of mental health–related ED visits were consistently found in all categories,” the authors reported. No trends were found regarding age, sex, or race/ethnicity. By contrast, mental health–related ED visits in which Medicaid was identified as the primary source of insurance nearly doubled between 2007–2008 and 2015–2016 (from 27.2% to 42.8%).
Other/unspecified psychiatric diagnoses, such as adjustment disorder and personality disorders, almost tripled between 2007-2008 and 2015-2016 (from 1,040 to 2,961 per 100,000 ED visits). ED visits for mood disorders and anxiety disorders also increased over time.
Alcohol-related ED visits were the most common substance use visits, increasing from 1,669 in 2007-2008 to 3,007 per 100,000 visits in 2015-2016. Amphetamine- and opioid-related ED visits more than doubled, and other/unspecified–related ED visits more than tripled during that time.
“One explanation why ED visits for mental health conditions have increased is that substance-related problems, which include overdose/self-injury issues, have increased over time,” Dr. Rhee noted, which “makes sense,” inasmuch as opioid, cannabis, and amphetamine use has increased across the country.
Another explanation is that, although mental health care access has been expanded through the ACA, “people, especially those with lower socioeconomic backgrounds, do not know how to get access to care and are still underserved,” he said.
“If mental health–related ED visits continue to increase in the future, there are several steps to be made. ED providers need to be better equipped with mental health care, and behavioral health should be better integrated as part of the care coordination,” said Dr. Rhee.
He added that reimbursement models across different insurance types, such as Medicare, Medicaid, and private insurance, “should consider expanding their coverage of mental health treatment in ED settings.”
“Canary in the coal mine”
Commenting on the study in an interview, Benjamin Druss, MD, MPH, professor and Rosalynn Carter Chair in Mental Health, Rollins School of Public Health, Emory University, Atlanta, called EDs the “canaries in the coal mine” for the broader health system.
The growing number of ED visits for behavioral problems “could represent both a rise in acute conditions such as substance use and lack of access to outpatient treatment,” said Dr. Druss, who was not involved with the research.
The findings “suggest the importance of strategies to effectively manage patients with behavioral conditions in ED settings and to effectively link them with high-quality outpatient care,” he noted.
Dr. Rhee has received funding from the National Institute on Aging and the American Foundation for Suicide Prevention. The other study authors and Dr. Druss report no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
ED visits related to mental health conditions increased nearly twofold from 2007-2008 to 2015-2016, new research suggests.
Data from the National Hospital Ambulatory Medical Care Survey (NHAMCS) showed that, over the 10-year study period, the proportion of ED visits for mental health diagnoses increased from 6.6% to 10.9%, with substance use accounting for much of the increase.
Although there have been policy efforts, such as expanding access to mental health care as part of the Affordable Care Act (ACA) of 2011, the senior author Taeho Greg Rhee, PhD, MSW, said in an interview.
“Treating mental health conditions in EDs is often considered suboptimal” because of limited time for full psychiatric assessment, lack of trained providers, and limited privacy in EDs, said Dr. Rhee of Yale University, New Haven, Conn.
The findings were published online July 28 in The Journal of Clinical Psychiatry.
“Outdated” research
Roughly one-fifth of U.S. adults experience some type of mental, behavioral, or emotional disorder annually. Moreover, the suicide rate has been steadily increasing, and there continues to be a “raging opioid epidemic,” the researchers wrote.
Despite these alarming figures, 57.4% of adults with mental illness reported in 2017 that they had not received any mental health treatment in the past year, reported the investigators.
Previous research has suggested that many adults have difficulty seeking outpatient mental health treatment and may turn to EDs instead. However, most studies of mental health ED use “are by now outdated, as they used data from years prior to the full implementation of the ACA,” the researchers noted.
“More Americans are suffering from mental illness, and given the recent policy efforts of expanding access to mental health care, we were questioning if ED visits due to mental health has changed or not,” Dr. Rhee said.
To investigate the question, the researchers conducted a cross-sectional analysis of data from the NHAMCS, a publicly available dataset provided by the National Center for Health Statistics of the Centers for Disease Control and Prevention.
They grouped psychiatric diagnoses into five categories: mood disorders, anxiety disorders, psychosis or schizophrenia, suicide attempt or ideation, or other/unspecified. Substance use diagnoses were grouped into six categories: alcohol, amphetamine, cannabis, cocaine, opioid, or other/unspecified.
These categories were used to determine the type of disorder a patient had, whether the patient had both psychiatric and substance-related diagnoses, and whether the patient received multiple mental health diagnoses at the time of the ED visit.
Sociodemographic covariates included age, sex, race/ethnicity, and insurance coverage.
Twofold and fourfold increases
Of 100.9 million outpatient ED visits that took place between 2007 and 2016, approximately 8.4 million (8.3%) were for psychiatric or substance use–related diagnoses. Also, the visits were more likely from adults who were younger than 45 years, male, non-Hispanic White, and covered by Medicaid or other public insurance types (58.5%, 52.5%, 65.2%, and 58.6%, respectively).
The overall rate of ED visits for any mental health diagnosis nearly doubled between 2007-2008 and 2015-2016. The rate of visits in which both psychiatric and substance use–related diagnoses increased fourfold during that time span. ED visits involving at least two mental health diagnoses increased twofold.
Additional changes in the number of visits are listed below (for each, P < .001).
When these comparisons were adjusted for age, sex, and race/ethnicity, “linearly increasing trends of mental health–related ED visits were consistently found in all categories,” the authors reported. No trends were found regarding age, sex, or race/ethnicity. By contrast, mental health–related ED visits in which Medicaid was identified as the primary source of insurance nearly doubled between 2007–2008 and 2015–2016 (from 27.2% to 42.8%).
Other/unspecified psychiatric diagnoses, such as adjustment disorder and personality disorders, almost tripled between 2007-2008 and 2015-2016 (from 1,040 to 2,961 per 100,000 ED visits). ED visits for mood disorders and anxiety disorders also increased over time.
Alcohol-related ED visits were the most common substance use visits, increasing from 1,669 in 2007-2008 to 3,007 per 100,000 visits in 2015-2016. Amphetamine- and opioid-related ED visits more than doubled, and other/unspecified–related ED visits more than tripled during that time.
“One explanation why ED visits for mental health conditions have increased is that substance-related problems, which include overdose/self-injury issues, have increased over time,” Dr. Rhee noted, which “makes sense,” inasmuch as opioid, cannabis, and amphetamine use has increased across the country.
Another explanation is that, although mental health care access has been expanded through the ACA, “people, especially those with lower socioeconomic backgrounds, do not know how to get access to care and are still underserved,” he said.
“If mental health–related ED visits continue to increase in the future, there are several steps to be made. ED providers need to be better equipped with mental health care, and behavioral health should be better integrated as part of the care coordination,” said Dr. Rhee.
He added that reimbursement models across different insurance types, such as Medicare, Medicaid, and private insurance, “should consider expanding their coverage of mental health treatment in ED settings.”
“Canary in the coal mine”
Commenting on the study in an interview, Benjamin Druss, MD, MPH, professor and Rosalynn Carter Chair in Mental Health, Rollins School of Public Health, Emory University, Atlanta, called EDs the “canaries in the coal mine” for the broader health system.
The growing number of ED visits for behavioral problems “could represent both a rise in acute conditions such as substance use and lack of access to outpatient treatment,” said Dr. Druss, who was not involved with the research.
The findings “suggest the importance of strategies to effectively manage patients with behavioral conditions in ED settings and to effectively link them with high-quality outpatient care,” he noted.
Dr. Rhee has received funding from the National Institute on Aging and the American Foundation for Suicide Prevention. The other study authors and Dr. Druss report no relevant financial relationships.
A version of this article originally appeared on Medscape.com.