User login
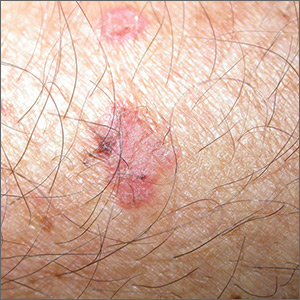
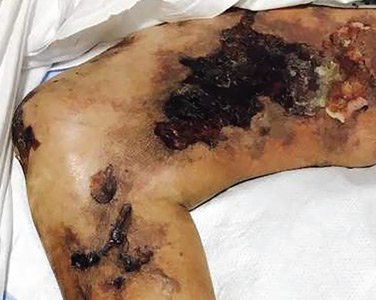
Scaling lesions on arm
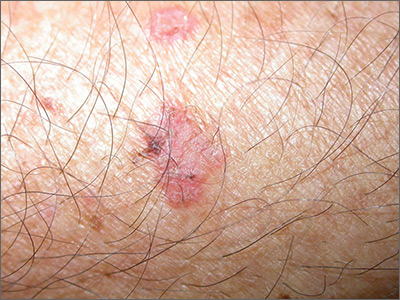
The FP also was concerned about a possible skin cancer, especially for the larger of the 2 lesions. The FP recommended 2 shave biopsies, which the patient agreed to. (See the Watch & Learn video on “Shave biopsy.”) The patient was directed to apply petrolatum once or twice daily to the biopsy sites and to cover them with dressings for the next 1 to 2 weeks. On the 2-week follow-up, the physician diagnosed the larger lesion as squamous cell carcinoma in situ (Bowen disease) and the top lesion as actinic keratosis.
The physician explained that the actinic keratosis did not need further treatment; however, the options for treating Bowen disease included cryosurgery, electrodesiccation and curettage, or elliptical excision. The patient chose an elliptical excision, which was performed without complications at the following visit.
The margins were clear and the surgery site healed without any problems. The patient said that he planned to wear long sleeves more often and use sunscreen when his arms were exposed to the sun.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Usatine R, Wah Y. Actinic keratosis and Bowen disease. In: Usatine R, Smith M, Mayeaux EJ, et al. Color Atlas of Family Medicine. 2nd ed. New York, NY: McGraw-Hill; 2013:969-976.
To learn more about the Color Atlas of Family Medicine, see: www.amazon.com/Color-Family-Medicine-Richard-Usatine/dp/0071769641/.
You can now get the second edition of the Color Atlas of Family Medicine as an app by clicking on this link: usatinemedia.com.

The FP also was concerned about a possible skin cancer, especially for the larger of the 2 lesions. The FP recommended 2 shave biopsies, which the patient agreed to. (See the Watch & Learn video on “Shave biopsy.”) The patient was directed to apply petrolatum once or twice daily to the biopsy sites and to cover them with dressings for the next 1 to 2 weeks. On the 2-week follow-up, the physician diagnosed the larger lesion as squamous cell carcinoma in situ (Bowen disease) and the top lesion as actinic keratosis.
The physician explained that the actinic keratosis did not need further treatment; however, the options for treating Bowen disease included cryosurgery, electrodesiccation and curettage, or elliptical excision. The patient chose an elliptical excision, which was performed without complications at the following visit.
The margins were clear and the surgery site healed without any problems. The patient said that he planned to wear long sleeves more often and use sunscreen when his arms were exposed to the sun.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Usatine R, Wah Y. Actinic keratosis and Bowen disease. In: Usatine R, Smith M, Mayeaux EJ, et al. Color Atlas of Family Medicine. 2nd ed. New York, NY: McGraw-Hill; 2013:969-976.
To learn more about the Color Atlas of Family Medicine, see: www.amazon.com/Color-Family-Medicine-Richard-Usatine/dp/0071769641/.
You can now get the second edition of the Color Atlas of Family Medicine as an app by clicking on this link: usatinemedia.com.

The FP also was concerned about a possible skin cancer, especially for the larger of the 2 lesions. The FP recommended 2 shave biopsies, which the patient agreed to. (See the Watch & Learn video on “Shave biopsy.”) The patient was directed to apply petrolatum once or twice daily to the biopsy sites and to cover them with dressings for the next 1 to 2 weeks. On the 2-week follow-up, the physician diagnosed the larger lesion as squamous cell carcinoma in situ (Bowen disease) and the top lesion as actinic keratosis.
The physician explained that the actinic keratosis did not need further treatment; however, the options for treating Bowen disease included cryosurgery, electrodesiccation and curettage, or elliptical excision. The patient chose an elliptical excision, which was performed without complications at the following visit.
The margins were clear and the surgery site healed without any problems. The patient said that he planned to wear long sleeves more often and use sunscreen when his arms were exposed to the sun.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Usatine R, Wah Y. Actinic keratosis and Bowen disease. In: Usatine R, Smith M, Mayeaux EJ, et al. Color Atlas of Family Medicine. 2nd ed. New York, NY: McGraw-Hill; 2013:969-976.
To learn more about the Color Atlas of Family Medicine, see: www.amazon.com/Color-Family-Medicine-Richard-Usatine/dp/0071769641/.
You can now get the second edition of the Color Atlas of Family Medicine as an app by clicking on this link: usatinemedia.com.
CUBE-C initiative aims to educate about atopic dermatitis
WASHINGTON – The National Eczema Association (NEA) has established the Coalition United for Better Eczema Care (CUBE-C) to provide practitioners with a resource for “trustworthy, up-to-date, state of the art” information on atopic dermatitis, with the goal of improving health outcomes, according to Julie Block, president and chief executive officer of the NEA.
In an interview at a symposium presented by CUBE-C, Ms. Block provided more information on CUBE-C, including how and why it started and what it can offer to dermatologists, as well as primary care physicians, who care for patients with atopic dermatitis. She said that the NEA convened dermatologists, allergists, immunologists, psychologists, nurse practitioners, physician assistants, and patients “to design a curriculum that provided an entire picture of the patient experience, so that we could go out and educate not only on the basics of eczema and atopic dermatitis for a variety of practitioners ... but also for the specialists who are now going to be engaging in new innovations and new therapies for their patients.”
She was joined by Adam Friedman, MD, professor of dermatology and residency program director at George Washington University, Washington, where the symposium, a resident’s boot camp, was held. The boot camp was somewhat unique in that it was geared more towards trainees; typically, the CUBE-C program is a CME program for practitioners, but this reflects the flexibility of the program, which can be tailored to the audience, Dr. Friedman pointed out. “The hope is that programs like these pop up all over the place ... anywhere you have a critical mass of individuals who want to learn about this,” where planners can choose from a menu of topics provided by CUBE-C – which include therapeutics, infections, pathogenesis, and access to care – and “easily formulate a conference like we held here today for the right audience.”
Topics covered at the George Washington University symposium included the impact of climate on the prevalence of childhood eczema, the diagnosis and differential diagnosis in children, infections in atopic dermatitis patients, and itch treatment.
More information on CUBE-C is available on the NEA website.
The symposium was supported by an educational grant from Sanofi Genzyme, Regeneron Pharmaceuticals, and Pfizer. Dr. Friedman reported serving as a speaker for Regeneron, Pfizer, and other companies. He also consults and serves on the advisory board for Pfizer and multiple other companies developing and marketing atopic dermatitis therapies and products.
WASHINGTON – The National Eczema Association (NEA) has established the Coalition United for Better Eczema Care (CUBE-C) to provide practitioners with a resource for “trustworthy, up-to-date, state of the art” information on atopic dermatitis, with the goal of improving health outcomes, according to Julie Block, president and chief executive officer of the NEA.
In an interview at a symposium presented by CUBE-C, Ms. Block provided more information on CUBE-C, including how and why it started and what it can offer to dermatologists, as well as primary care physicians, who care for patients with atopic dermatitis. She said that the NEA convened dermatologists, allergists, immunologists, psychologists, nurse practitioners, physician assistants, and patients “to design a curriculum that provided an entire picture of the patient experience, so that we could go out and educate not only on the basics of eczema and atopic dermatitis for a variety of practitioners ... but also for the specialists who are now going to be engaging in new innovations and new therapies for their patients.”
She was joined by Adam Friedman, MD, professor of dermatology and residency program director at George Washington University, Washington, where the symposium, a resident’s boot camp, was held. The boot camp was somewhat unique in that it was geared more towards trainees; typically, the CUBE-C program is a CME program for practitioners, but this reflects the flexibility of the program, which can be tailored to the audience, Dr. Friedman pointed out. “The hope is that programs like these pop up all over the place ... anywhere you have a critical mass of individuals who want to learn about this,” where planners can choose from a menu of topics provided by CUBE-C – which include therapeutics, infections, pathogenesis, and access to care – and “easily formulate a conference like we held here today for the right audience.”
Topics covered at the George Washington University symposium included the impact of climate on the prevalence of childhood eczema, the diagnosis and differential diagnosis in children, infections in atopic dermatitis patients, and itch treatment.
More information on CUBE-C is available on the NEA website.
The symposium was supported by an educational grant from Sanofi Genzyme, Regeneron Pharmaceuticals, and Pfizer. Dr. Friedman reported serving as a speaker for Regeneron, Pfizer, and other companies. He also consults and serves on the advisory board for Pfizer and multiple other companies developing and marketing atopic dermatitis therapies and products.
WASHINGTON – The National Eczema Association (NEA) has established the Coalition United for Better Eczema Care (CUBE-C) to provide practitioners with a resource for “trustworthy, up-to-date, state of the art” information on atopic dermatitis, with the goal of improving health outcomes, according to Julie Block, president and chief executive officer of the NEA.
In an interview at a symposium presented by CUBE-C, Ms. Block provided more information on CUBE-C, including how and why it started and what it can offer to dermatologists, as well as primary care physicians, who care for patients with atopic dermatitis. She said that the NEA convened dermatologists, allergists, immunologists, psychologists, nurse practitioners, physician assistants, and patients “to design a curriculum that provided an entire picture of the patient experience, so that we could go out and educate not only on the basics of eczema and atopic dermatitis for a variety of practitioners ... but also for the specialists who are now going to be engaging in new innovations and new therapies for their patients.”
She was joined by Adam Friedman, MD, professor of dermatology and residency program director at George Washington University, Washington, where the symposium, a resident’s boot camp, was held. The boot camp was somewhat unique in that it was geared more towards trainees; typically, the CUBE-C program is a CME program for practitioners, but this reflects the flexibility of the program, which can be tailored to the audience, Dr. Friedman pointed out. “The hope is that programs like these pop up all over the place ... anywhere you have a critical mass of individuals who want to learn about this,” where planners can choose from a menu of topics provided by CUBE-C – which include therapeutics, infections, pathogenesis, and access to care – and “easily formulate a conference like we held here today for the right audience.”
Topics covered at the George Washington University symposium included the impact of climate on the prevalence of childhood eczema, the diagnosis and differential diagnosis in children, infections in atopic dermatitis patients, and itch treatment.
More information on CUBE-C is available on the NEA website.
The symposium was supported by an educational grant from Sanofi Genzyme, Regeneron Pharmaceuticals, and Pfizer. Dr. Friedman reported serving as a speaker for Regeneron, Pfizer, and other companies. He also consults and serves on the advisory board for Pfizer and multiple other companies developing and marketing atopic dermatitis therapies and products.
Immunosuppression often triggers skin side effects
LAKE TAHOE, CALIF. – , and side effects of vemurafenib treatment.
In a presentation on the cutaneous sequelae of different immunosuppressive regimens at the annual meeting of the Society for Pediatric Dermatology, Carrie C. Coughlin, MD, opened with a discussion of AD triggered by the tumor necrosis factor (TNF) blocker infliximab, especially in the setting of therapy for Crohn’s disease. “In this patient population you often think of psoriasis as a consequence of infliximab and other TNF therapies,” said Dr. Coughlin, a pediatric dermatologist at Washington University, St. Louis. “But you can also get true atopic dermatitis with infliximab as well. Who’s more at risk for this? Patients with Crohn’s disease seem to be. Most of the literature is in adults, but there are a few series of children. In a series of children looking at cutaneous sequelae of infliximab therapy, about 20% of cutaneous eruptions were atopic dermatitis. I think it’s a great opportunity for us in dermatology to do a more research in this area.”
Some researchers have proposed that atopic disease could be a marker of over-suppression of TNF-alpha in young Crohn’s disease patients on infliximab (Inflamm Bowel Dis. 2014; 20[8]:1309-15). “One question you could ask is, could these patients actually tolerate a dose reduction?” Dr. Coughlin said. She promoted the role of dermatologists in working at managing side effects to keep patients on medications helping their GI disease, but acknowledged this is not always possible.
Atopic disease can also occur after a solid organ transplant. In fact, the incidence of new-onset food allergies after a liver transplant is 6%-30% of cases, mainly in young patients (Pediatr Transplant. 2009;13[1]:63-9, Pediatr Transplant 2013;17[3]:251-5). “There are some mechanisms, including liver presentation of antigens, that spread through portal veins that could potentially put people at risk who have liver dysfunction,” Dr. Coughlin explained. “They could potentially have a higher risk for food allergies and AD. There is also some thought that tacrolimus potentially predisposes patients to having atopic dermatitis and atopy after their transplant. When you look at the mechanism of action of tacrolimus, you see increased levels of IL-5, IL-13, and skewing of IgE levels.”
Dr. Coughlin also discussed the possibilities of development of AD after transplant being a delayed presentation of an allergic sensitization that patients already had. Younger patients are at higher risk for AD post transplant. The renal transplant population, meanwhile, generally receives the transplant at a later age, “so they may not have that delay in terms of presentation; they may have already had their allergies and grown out of them by the time they’re getting their transplant,” she said. “I think there’s more for us to investigate.”
Solid organ transplant recipients also face an increased risk of skin cancer as a long-term side effect of immunosuppressive therapy. Risk factors include fair skin, sun exposure, and remote time from transplant. “Time from transplant is a risk factor,” Dr. Coughlin said. “Longer time on immunosuppression could predispose you to a risk for skin cancer. Our patients are living longer post transplant than they used to, so they have more potential years to develop their skin cancers.” She focused on the importance of educating transplant recipients and families early about photoprotection. “It’s interesting to think about how we can continue to intervene early on to continue to decrease risk.”
Young patients exposed to voriconazole also face an increased risk for skin cancer. “We know that longer-term dosing and higher cumulative dosing puts you at higher risk,” she said. Lung transplant recipients, who are often more likely to be treated with voriconazole, are thus at higher risk.
Dr. Coughlin ended her presentation by noting that side effects of the BRAF inhibitor vemurafenib (Zelboraf) used to treat advanced melanoma in children are similar to, but not the same as, those in adults. “We see BRAF mutations in multiple different tumor types: Langerhans cell histiocytosis, gliomas, and melanoma,” she said. “Trials of vemurafenib and dabrafenib are under way in the pediatric population. Vemurafenib can cause keratosis pilaris, panniculitis, alopecia, and granulomatous dermatitis.” In her experience, she has seen more alopecia in the older teenage population, but younger patients may not be asked about this side effect as frequently.
She counsels patients to expect keratosis pilaris–like eruptions and to take sun protection seriously. “It’s important to emphasize that each time they come in,” Dr. Coughlin said. She also discussed the potential for changing nevi and treatment options for vemurafenib-associated panniculitis.
Dr. Coughlin disclosed that she is the recipient of active pilot grants from the Pediatric Dermatology Research Alliance and the SPD.
LAKE TAHOE, CALIF. – , and side effects of vemurafenib treatment.
In a presentation on the cutaneous sequelae of different immunosuppressive regimens at the annual meeting of the Society for Pediatric Dermatology, Carrie C. Coughlin, MD, opened with a discussion of AD triggered by the tumor necrosis factor (TNF) blocker infliximab, especially in the setting of therapy for Crohn’s disease. “In this patient population you often think of psoriasis as a consequence of infliximab and other TNF therapies,” said Dr. Coughlin, a pediatric dermatologist at Washington University, St. Louis. “But you can also get true atopic dermatitis with infliximab as well. Who’s more at risk for this? Patients with Crohn’s disease seem to be. Most of the literature is in adults, but there are a few series of children. In a series of children looking at cutaneous sequelae of infliximab therapy, about 20% of cutaneous eruptions were atopic dermatitis. I think it’s a great opportunity for us in dermatology to do a more research in this area.”
Some researchers have proposed that atopic disease could be a marker of over-suppression of TNF-alpha in young Crohn’s disease patients on infliximab (Inflamm Bowel Dis. 2014; 20[8]:1309-15). “One question you could ask is, could these patients actually tolerate a dose reduction?” Dr. Coughlin said. She promoted the role of dermatologists in working at managing side effects to keep patients on medications helping their GI disease, but acknowledged this is not always possible.
Atopic disease can also occur after a solid organ transplant. In fact, the incidence of new-onset food allergies after a liver transplant is 6%-30% of cases, mainly in young patients (Pediatr Transplant. 2009;13[1]:63-9, Pediatr Transplant 2013;17[3]:251-5). “There are some mechanisms, including liver presentation of antigens, that spread through portal veins that could potentially put people at risk who have liver dysfunction,” Dr. Coughlin explained. “They could potentially have a higher risk for food allergies and AD. There is also some thought that tacrolimus potentially predisposes patients to having atopic dermatitis and atopy after their transplant. When you look at the mechanism of action of tacrolimus, you see increased levels of IL-5, IL-13, and skewing of IgE levels.”
Dr. Coughlin also discussed the possibilities of development of AD after transplant being a delayed presentation of an allergic sensitization that patients already had. Younger patients are at higher risk for AD post transplant. The renal transplant population, meanwhile, generally receives the transplant at a later age, “so they may not have that delay in terms of presentation; they may have already had their allergies and grown out of them by the time they’re getting their transplant,” she said. “I think there’s more for us to investigate.”
Solid organ transplant recipients also face an increased risk of skin cancer as a long-term side effect of immunosuppressive therapy. Risk factors include fair skin, sun exposure, and remote time from transplant. “Time from transplant is a risk factor,” Dr. Coughlin said. “Longer time on immunosuppression could predispose you to a risk for skin cancer. Our patients are living longer post transplant than they used to, so they have more potential years to develop their skin cancers.” She focused on the importance of educating transplant recipients and families early about photoprotection. “It’s interesting to think about how we can continue to intervene early on to continue to decrease risk.”
Young patients exposed to voriconazole also face an increased risk for skin cancer. “We know that longer-term dosing and higher cumulative dosing puts you at higher risk,” she said. Lung transplant recipients, who are often more likely to be treated with voriconazole, are thus at higher risk.
Dr. Coughlin ended her presentation by noting that side effects of the BRAF inhibitor vemurafenib (Zelboraf) used to treat advanced melanoma in children are similar to, but not the same as, those in adults. “We see BRAF mutations in multiple different tumor types: Langerhans cell histiocytosis, gliomas, and melanoma,” she said. “Trials of vemurafenib and dabrafenib are under way in the pediatric population. Vemurafenib can cause keratosis pilaris, panniculitis, alopecia, and granulomatous dermatitis.” In her experience, she has seen more alopecia in the older teenage population, but younger patients may not be asked about this side effect as frequently.
She counsels patients to expect keratosis pilaris–like eruptions and to take sun protection seriously. “It’s important to emphasize that each time they come in,” Dr. Coughlin said. She also discussed the potential for changing nevi and treatment options for vemurafenib-associated panniculitis.
Dr. Coughlin disclosed that she is the recipient of active pilot grants from the Pediatric Dermatology Research Alliance and the SPD.
LAKE TAHOE, CALIF. – , and side effects of vemurafenib treatment.
In a presentation on the cutaneous sequelae of different immunosuppressive regimens at the annual meeting of the Society for Pediatric Dermatology, Carrie C. Coughlin, MD, opened with a discussion of AD triggered by the tumor necrosis factor (TNF) blocker infliximab, especially in the setting of therapy for Crohn’s disease. “In this patient population you often think of psoriasis as a consequence of infliximab and other TNF therapies,” said Dr. Coughlin, a pediatric dermatologist at Washington University, St. Louis. “But you can also get true atopic dermatitis with infliximab as well. Who’s more at risk for this? Patients with Crohn’s disease seem to be. Most of the literature is in adults, but there are a few series of children. In a series of children looking at cutaneous sequelae of infliximab therapy, about 20% of cutaneous eruptions were atopic dermatitis. I think it’s a great opportunity for us in dermatology to do a more research in this area.”
Some researchers have proposed that atopic disease could be a marker of over-suppression of TNF-alpha in young Crohn’s disease patients on infliximab (Inflamm Bowel Dis. 2014; 20[8]:1309-15). “One question you could ask is, could these patients actually tolerate a dose reduction?” Dr. Coughlin said. She promoted the role of dermatologists in working at managing side effects to keep patients on medications helping their GI disease, but acknowledged this is not always possible.
Atopic disease can also occur after a solid organ transplant. In fact, the incidence of new-onset food allergies after a liver transplant is 6%-30% of cases, mainly in young patients (Pediatr Transplant. 2009;13[1]:63-9, Pediatr Transplant 2013;17[3]:251-5). “There are some mechanisms, including liver presentation of antigens, that spread through portal veins that could potentially put people at risk who have liver dysfunction,” Dr. Coughlin explained. “They could potentially have a higher risk for food allergies and AD. There is also some thought that tacrolimus potentially predisposes patients to having atopic dermatitis and atopy after their transplant. When you look at the mechanism of action of tacrolimus, you see increased levels of IL-5, IL-13, and skewing of IgE levels.”
Dr. Coughlin also discussed the possibilities of development of AD after transplant being a delayed presentation of an allergic sensitization that patients already had. Younger patients are at higher risk for AD post transplant. The renal transplant population, meanwhile, generally receives the transplant at a later age, “so they may not have that delay in terms of presentation; they may have already had their allergies and grown out of them by the time they’re getting their transplant,” she said. “I think there’s more for us to investigate.”
Solid organ transplant recipients also face an increased risk of skin cancer as a long-term side effect of immunosuppressive therapy. Risk factors include fair skin, sun exposure, and remote time from transplant. “Time from transplant is a risk factor,” Dr. Coughlin said. “Longer time on immunosuppression could predispose you to a risk for skin cancer. Our patients are living longer post transplant than they used to, so they have more potential years to develop their skin cancers.” She focused on the importance of educating transplant recipients and families early about photoprotection. “It’s interesting to think about how we can continue to intervene early on to continue to decrease risk.”
Young patients exposed to voriconazole also face an increased risk for skin cancer. “We know that longer-term dosing and higher cumulative dosing puts you at higher risk,” she said. Lung transplant recipients, who are often more likely to be treated with voriconazole, are thus at higher risk.
Dr. Coughlin ended her presentation by noting that side effects of the BRAF inhibitor vemurafenib (Zelboraf) used to treat advanced melanoma in children are similar to, but not the same as, those in adults. “We see BRAF mutations in multiple different tumor types: Langerhans cell histiocytosis, gliomas, and melanoma,” she said. “Trials of vemurafenib and dabrafenib are under way in the pediatric population. Vemurafenib can cause keratosis pilaris, panniculitis, alopecia, and granulomatous dermatitis.” In her experience, she has seen more alopecia in the older teenage population, but younger patients may not be asked about this side effect as frequently.
She counsels patients to expect keratosis pilaris–like eruptions and to take sun protection seriously. “It’s important to emphasize that each time they come in,” Dr. Coughlin said. She also discussed the potential for changing nevi and treatment options for vemurafenib-associated panniculitis.
Dr. Coughlin disclosed that she is the recipient of active pilot grants from the Pediatric Dermatology Research Alliance and the SPD.
REPORTING FROM SPD 2018
Tease out genetic and structural causes of children’s hair loss
according to Maria Hordinsky, MD, of the University of Minnesota, Minneapolis.
The ectodermal dysplasias are a heterogeneous group of disorders in which a main feature is the absent, incomplete, or delayed development of one or more of the appendages derived from ectoderm, such as the hair follicle, Dr. Hordinsky said in a presentation at Skin Disease Education Foundation’s Women’s & Pediatric Dermatology Seminar.
Patients with pure hair and nail ectodermal dysplasia generally present with absent or sparse eyebrows and eyelashes, as well as follicular papules on the scalp and fragile, irregular hair, Dr. Hordinsky said. The condition is caused by a mutation in a gene associated with the production of keratin. In another rare form of hereditary hair loss – hypotrichosis simplex – patients are born with normal hair but lose it gradually from the scalp during the middle of the first decade of life.
The inability to grow long hair characterizes short anagen syndrome, a congenital disorder not to be confused with loose anagen syndrome, Dr. Hordinsky said. Patients with short anagen syndrome experience an idiopathic short anagen phase and as a result, an increased number of hairs in the telogen phase. Children with short anagen syndrome have unusually short hair in early childhood. “Parents typically complain that their children exhibit short hair even though they have never had a haircut,” she explained.
Trichothiodystrophy, a rare autosomal recessive disease, is distinguished by hair that is brittle and sulfur deficient, Dr. Hordinsky said. She cited a review of 112 patients with trichothiodystrophy in which additional distinguishing features included developmental delay/intellectual impairment (86%), short stature (73%), and ichthyosis (65%).
Some cases of hair loss in children have a structural basis, Dr. Hordinsky noted. Structural hair abnormalities include fractures of the hair shaft, extraneous matter on the hair shaft, and hair shaft irregularities such as coiling or twisting, she said.
In trichoptilosis, extensive cuticle loss results in fraying and splitting of the hair shaft, while in patients with trichoclasis, a fractured hair is splinted by a partially intact cuticle.
In trichorrhexis nodosa, the most common type of structural hair abnormality, “intact nodes [of hair] resemble two paintbrushes thrust together,” Dr. Hordinsky explained. Trichorrhexis nodosa may be congenital or acquired, and occurs in children with mental retardation and argininosuccinic aciduria, she said.
A hair shaft abnormality is the culprit behind uncombable hair syndrome, which can be inherited or can occur sporadically, Dr. Hordinsky said. The key feature of the condition is unruly hair caused by a distinctive hair shaft defect, “possibly related to an abnormality in the inner root sheath.” Abnormal hairs usually become apparent at about 3-4 years of age, but eyebrows and eyelashes appear normal. Many patients have a silvery blonde tint to their hair because of how the abnormal hairs reflect light, she said.
Dr. Hordinsky is a consultant for P&G, Concert, Cassiopea, and BioAZ; and receives grant/research support from Aclaris, Allergan, and the National Alopecia Areata Foundation. SDEF and this news organization are owned by Frontline Medical Communications.
according to Maria Hordinsky, MD, of the University of Minnesota, Minneapolis.
The ectodermal dysplasias are a heterogeneous group of disorders in which a main feature is the absent, incomplete, or delayed development of one or more of the appendages derived from ectoderm, such as the hair follicle, Dr. Hordinsky said in a presentation at Skin Disease Education Foundation’s Women’s & Pediatric Dermatology Seminar.
Patients with pure hair and nail ectodermal dysplasia generally present with absent or sparse eyebrows and eyelashes, as well as follicular papules on the scalp and fragile, irregular hair, Dr. Hordinsky said. The condition is caused by a mutation in a gene associated with the production of keratin. In another rare form of hereditary hair loss – hypotrichosis simplex – patients are born with normal hair but lose it gradually from the scalp during the middle of the first decade of life.
The inability to grow long hair characterizes short anagen syndrome, a congenital disorder not to be confused with loose anagen syndrome, Dr. Hordinsky said. Patients with short anagen syndrome experience an idiopathic short anagen phase and as a result, an increased number of hairs in the telogen phase. Children with short anagen syndrome have unusually short hair in early childhood. “Parents typically complain that their children exhibit short hair even though they have never had a haircut,” she explained.
Trichothiodystrophy, a rare autosomal recessive disease, is distinguished by hair that is brittle and sulfur deficient, Dr. Hordinsky said. She cited a review of 112 patients with trichothiodystrophy in which additional distinguishing features included developmental delay/intellectual impairment (86%), short stature (73%), and ichthyosis (65%).
Some cases of hair loss in children have a structural basis, Dr. Hordinsky noted. Structural hair abnormalities include fractures of the hair shaft, extraneous matter on the hair shaft, and hair shaft irregularities such as coiling or twisting, she said.
In trichoptilosis, extensive cuticle loss results in fraying and splitting of the hair shaft, while in patients with trichoclasis, a fractured hair is splinted by a partially intact cuticle.
In trichorrhexis nodosa, the most common type of structural hair abnormality, “intact nodes [of hair] resemble two paintbrushes thrust together,” Dr. Hordinsky explained. Trichorrhexis nodosa may be congenital or acquired, and occurs in children with mental retardation and argininosuccinic aciduria, she said.
A hair shaft abnormality is the culprit behind uncombable hair syndrome, which can be inherited or can occur sporadically, Dr. Hordinsky said. The key feature of the condition is unruly hair caused by a distinctive hair shaft defect, “possibly related to an abnormality in the inner root sheath.” Abnormal hairs usually become apparent at about 3-4 years of age, but eyebrows and eyelashes appear normal. Many patients have a silvery blonde tint to their hair because of how the abnormal hairs reflect light, she said.
Dr. Hordinsky is a consultant for P&G, Concert, Cassiopea, and BioAZ; and receives grant/research support from Aclaris, Allergan, and the National Alopecia Areata Foundation. SDEF and this news organization are owned by Frontline Medical Communications.
according to Maria Hordinsky, MD, of the University of Minnesota, Minneapolis.
The ectodermal dysplasias are a heterogeneous group of disorders in which a main feature is the absent, incomplete, or delayed development of one or more of the appendages derived from ectoderm, such as the hair follicle, Dr. Hordinsky said in a presentation at Skin Disease Education Foundation’s Women’s & Pediatric Dermatology Seminar.
Patients with pure hair and nail ectodermal dysplasia generally present with absent or sparse eyebrows and eyelashes, as well as follicular papules on the scalp and fragile, irregular hair, Dr. Hordinsky said. The condition is caused by a mutation in a gene associated with the production of keratin. In another rare form of hereditary hair loss – hypotrichosis simplex – patients are born with normal hair but lose it gradually from the scalp during the middle of the first decade of life.
The inability to grow long hair characterizes short anagen syndrome, a congenital disorder not to be confused with loose anagen syndrome, Dr. Hordinsky said. Patients with short anagen syndrome experience an idiopathic short anagen phase and as a result, an increased number of hairs in the telogen phase. Children with short anagen syndrome have unusually short hair in early childhood. “Parents typically complain that their children exhibit short hair even though they have never had a haircut,” she explained.
Trichothiodystrophy, a rare autosomal recessive disease, is distinguished by hair that is brittle and sulfur deficient, Dr. Hordinsky said. She cited a review of 112 patients with trichothiodystrophy in which additional distinguishing features included developmental delay/intellectual impairment (86%), short stature (73%), and ichthyosis (65%).
Some cases of hair loss in children have a structural basis, Dr. Hordinsky noted. Structural hair abnormalities include fractures of the hair shaft, extraneous matter on the hair shaft, and hair shaft irregularities such as coiling or twisting, she said.
In trichoptilosis, extensive cuticle loss results in fraying and splitting of the hair shaft, while in patients with trichoclasis, a fractured hair is splinted by a partially intact cuticle.
In trichorrhexis nodosa, the most common type of structural hair abnormality, “intact nodes [of hair] resemble two paintbrushes thrust together,” Dr. Hordinsky explained. Trichorrhexis nodosa may be congenital or acquired, and occurs in children with mental retardation and argininosuccinic aciduria, she said.
A hair shaft abnormality is the culprit behind uncombable hair syndrome, which can be inherited or can occur sporadically, Dr. Hordinsky said. The key feature of the condition is unruly hair caused by a distinctive hair shaft defect, “possibly related to an abnormality in the inner root sheath.” Abnormal hairs usually become apparent at about 3-4 years of age, but eyebrows and eyelashes appear normal. Many patients have a silvery blonde tint to their hair because of how the abnormal hairs reflect light, she said.
Dr. Hordinsky is a consultant for P&G, Concert, Cassiopea, and BioAZ; and receives grant/research support from Aclaris, Allergan, and the National Alopecia Areata Foundation. SDEF and this news organization are owned by Frontline Medical Communications.
FROM SDEF WOMEN’S & PEDIATRIC DERMATOLOGY SEMINAR
Advise parents on validity of AD-associated conditions
including allergic rhinitis and asthma, according to Douglas W. Kress, MD, of the department of dermatology at the University of Pittsburgh.
Recent studies suggest that atopic dermatitis (AD) affects 10%-17% of the U.S. population, and 80%-90% of patients are diagnosed by the age of 5 years, Dr. Kress said in a presentation at Skin Disease Education Foundation’s Women’s & Pediatric Dermatology Seminar.
“There seem to be multiple pathways, which initiate and perpetuate the cutaneous inflammation of AD including exposure to allergens, irritants, and physical trauma, infection, stress, extremes in temperature and humidity,” Dr. Kress said. In addition, foods and airborne allergens may trigger AD.
Many parents may believe that certain factors are associated with AD, but most of these perceptions are not supported by evidence, said Dr. Kress, who is also chief of the division of pediatric dermatology at the Children’s Hospital of Pittsburgh. AD appears to be a disorder of T cell dysregulation dominated by Th2 lesions in acute cases and Th1 inflammation in patients with chronic lesions.
No associations have been proven between the development of AD in the first 18 months of life and any maternal dietary restrictions, according to a recent Cochrane review, nor is there evidence for an association between AD and the introduction of solid foods, exposure to fish oil, or exposure to animals at a young age, he said. In addition, a study published in 2016 showed a lack of evidence to support the use of specific allergen immunotherapy for AD.
However, evidence does support an association between the presence of AD in children and certain other conditions, Dr. Kress said. “Other associations include an increased incidence of alopecia areata, a threefold increase in autism spectrum disorders, and a twofold increase in ADHD in children with atopic dermatitis.”
The only known food allergy linked to AD severity is egg whites; reducing egg white exposure has been shown to improve AD in children with both conditions, he noted.
Although many patients with AD experience annoying but relatively mild symptoms, health care providers should be alert to the potential for infections, particularly with Staphylococcus aureus, and remember that an active egg white allergy has been associated with staphylococcal superantigen sensitization, said Dr. Kress. The increased risk for S. aureus in children with AD may stem from a tendency to underuse antibiotics in AD children, which results in a delayed treatment until the infection becomes overt. In addition, the increased pH in patients with AD might promote the development of pathogenic strains of staph. However, ceramide-based moisturizers could help inhibit these strains by increasing skin acidity.
For patients who have poor AD control with standard therapy, antibiotics may be used as adjunctive therapy. “Consider bleach baths and/or staph decolonization with mupirocin, both of which led to significant improvement in eczema severity compared to placebo,” Dr. Kress said. “Bleach may also have an anti-inflammatory effect.”
Dr. Kress disclosed relationships with Pfizer, Amgen, and Sanofi/Regeneron. SDEF and this news organization are owned by Frontline Medical Communications.
including allergic rhinitis and asthma, according to Douglas W. Kress, MD, of the department of dermatology at the University of Pittsburgh.
Recent studies suggest that atopic dermatitis (AD) affects 10%-17% of the U.S. population, and 80%-90% of patients are diagnosed by the age of 5 years, Dr. Kress said in a presentation at Skin Disease Education Foundation’s Women’s & Pediatric Dermatology Seminar.
“There seem to be multiple pathways, which initiate and perpetuate the cutaneous inflammation of AD including exposure to allergens, irritants, and physical trauma, infection, stress, extremes in temperature and humidity,” Dr. Kress said. In addition, foods and airborne allergens may trigger AD.
Many parents may believe that certain factors are associated with AD, but most of these perceptions are not supported by evidence, said Dr. Kress, who is also chief of the division of pediatric dermatology at the Children’s Hospital of Pittsburgh. AD appears to be a disorder of T cell dysregulation dominated by Th2 lesions in acute cases and Th1 inflammation in patients with chronic lesions.
No associations have been proven between the development of AD in the first 18 months of life and any maternal dietary restrictions, according to a recent Cochrane review, nor is there evidence for an association between AD and the introduction of solid foods, exposure to fish oil, or exposure to animals at a young age, he said. In addition, a study published in 2016 showed a lack of evidence to support the use of specific allergen immunotherapy for AD.
However, evidence does support an association between the presence of AD in children and certain other conditions, Dr. Kress said. “Other associations include an increased incidence of alopecia areata, a threefold increase in autism spectrum disorders, and a twofold increase in ADHD in children with atopic dermatitis.”
The only known food allergy linked to AD severity is egg whites; reducing egg white exposure has been shown to improve AD in children with both conditions, he noted.
Although many patients with AD experience annoying but relatively mild symptoms, health care providers should be alert to the potential for infections, particularly with Staphylococcus aureus, and remember that an active egg white allergy has been associated with staphylococcal superantigen sensitization, said Dr. Kress. The increased risk for S. aureus in children with AD may stem from a tendency to underuse antibiotics in AD children, which results in a delayed treatment until the infection becomes overt. In addition, the increased pH in patients with AD might promote the development of pathogenic strains of staph. However, ceramide-based moisturizers could help inhibit these strains by increasing skin acidity.
For patients who have poor AD control with standard therapy, antibiotics may be used as adjunctive therapy. “Consider bleach baths and/or staph decolonization with mupirocin, both of which led to significant improvement in eczema severity compared to placebo,” Dr. Kress said. “Bleach may also have an anti-inflammatory effect.”
Dr. Kress disclosed relationships with Pfizer, Amgen, and Sanofi/Regeneron. SDEF and this news organization are owned by Frontline Medical Communications.
including allergic rhinitis and asthma, according to Douglas W. Kress, MD, of the department of dermatology at the University of Pittsburgh.
Recent studies suggest that atopic dermatitis (AD) affects 10%-17% of the U.S. population, and 80%-90% of patients are diagnosed by the age of 5 years, Dr. Kress said in a presentation at Skin Disease Education Foundation’s Women’s & Pediatric Dermatology Seminar.
“There seem to be multiple pathways, which initiate and perpetuate the cutaneous inflammation of AD including exposure to allergens, irritants, and physical trauma, infection, stress, extremes in temperature and humidity,” Dr. Kress said. In addition, foods and airborne allergens may trigger AD.
Many parents may believe that certain factors are associated with AD, but most of these perceptions are not supported by evidence, said Dr. Kress, who is also chief of the division of pediatric dermatology at the Children’s Hospital of Pittsburgh. AD appears to be a disorder of T cell dysregulation dominated by Th2 lesions in acute cases and Th1 inflammation in patients with chronic lesions.
No associations have been proven between the development of AD in the first 18 months of life and any maternal dietary restrictions, according to a recent Cochrane review, nor is there evidence for an association between AD and the introduction of solid foods, exposure to fish oil, or exposure to animals at a young age, he said. In addition, a study published in 2016 showed a lack of evidence to support the use of specific allergen immunotherapy for AD.
However, evidence does support an association between the presence of AD in children and certain other conditions, Dr. Kress said. “Other associations include an increased incidence of alopecia areata, a threefold increase in autism spectrum disorders, and a twofold increase in ADHD in children with atopic dermatitis.”
The only known food allergy linked to AD severity is egg whites; reducing egg white exposure has been shown to improve AD in children with both conditions, he noted.
Although many patients with AD experience annoying but relatively mild symptoms, health care providers should be alert to the potential for infections, particularly with Staphylococcus aureus, and remember that an active egg white allergy has been associated with staphylococcal superantigen sensitization, said Dr. Kress. The increased risk for S. aureus in children with AD may stem from a tendency to underuse antibiotics in AD children, which results in a delayed treatment until the infection becomes overt. In addition, the increased pH in patients with AD might promote the development of pathogenic strains of staph. However, ceramide-based moisturizers could help inhibit these strains by increasing skin acidity.
For patients who have poor AD control with standard therapy, antibiotics may be used as adjunctive therapy. “Consider bleach baths and/or staph decolonization with mupirocin, both of which led to significant improvement in eczema severity compared to placebo,” Dr. Kress said. “Bleach may also have an anti-inflammatory effect.”
Dr. Kress disclosed relationships with Pfizer, Amgen, and Sanofi/Regeneron. SDEF and this news organization are owned by Frontline Medical Communications.
EXPERT ANALYSIS FROM SDEF WOMEN’S & PEDIATRIC DERMATOLOGY SEMINAR
Study offers snapshot of esophageal strictures in EB patients
LAKE TAHOE, CALIF. – and direct visualization of these strictures is the preferred method of diagnosis. Those are key findings from a multicenter study that lead author Elena Pope, MD, discussed at the annual meeting of the Society for Pediatric Dermatology.
According to Dr. Pope, who heads the section of dermatology at the Hospital for Sick Children, Toronto, an estimated 10%-17% of epidermolysis bullosa (EB) patients experience strictures, with an overrepresentation in the recessive dystrophic EB subtype in up to 80% of cases. The risk increases with age. “What remains unknown is the best short- and long-term intervention to manage the strictures and predictors/associations for stricture-free episodes,” Dr. Pope said. “The objectives of the current study were to determine the prevalence and predisposing factors for strictures in EB, management options, patient outcomes, and predictors for recurrences and stricture-free intervals.”
She and her associates at seven centers worldwide collected data on 125 EB patients who experienced at least one episode of esophageal stricture. Data was analyzed descriptively and with ANOVA regression analysis for associations/predictors for recurrences/episode-free intervals.
The researchers evaluated 497 stricture events in the 125 patients. A slight female predominance was noted (53%), and the mean age of the first episode was 12.7 years, “which is a little bit older” than the age found in previously published data, Dr. Pope said. As expected, dystrophic EB patients made up most of the sample (98.4%); of these 123 patients, recessive dystrophic EB severe generalized subtype – approaching 50% – was the most common, followed by the recessive dystrophic EB severe intermediate subtype (almost 21%), the dominant dystrophic EB generalized subtype (7%), and other types of dystrophic EB (almost 26%).
The median body mass index percentile for age was 6.3, “so these were patients who were severely malnourished, probably as a result of their strictures as well as their underlying disease,” Dr. Pope said.
As expected, dysphagia was a presenting symptom in most patients (85.5%), while 29.8% presented with inability to swallow solids. The preferred method of evaluation was video fluoroscopy (57.7%), and less commonly with barium swallow (22.3%) or with clinical symptoms alone (0.1%). The mean number of strictures was 1.69; 76.7% were located in the cervical area, 56.7% were located in the thoracic area, and 9.7% were located in the abdominal area. Most patients (76%) had lesions that were 1 cm or longer in size.
Fluoroscopy guidance was the most common method of dilatation (in 45.2% of cases), followed by retrograde endoscopy was (33%), antegrade endoscopy (19.1%), and bougienage (0.1%). General anesthesia was used in most cases (87.6%), and corticosteroids were used around the dilatation in 90.4% of patients. The mean duration of medication use was about 5 days.
As for outcomes after dilatation, 92.2% of strictures completely resolved, 3.8% were partially resolved, 3.9% were not resolved, and 2.7% had complications. The median interval between dilatations was 7 months. Fluoroscopy-guided balloon dilatation was associated with the longest esophageal stricture-free duration (mean of 13.83 months vs. 8.75 months; P less than .001), followed by retrograde endoscopy (mean of 13.10 months vs. 7.85 months; P less than .001), and antegrade endoscopy (mean of 7.63 months vs. 11.46 months; P = .024). “I think this is interesting,” said Dr. Pope, who is also a professor of pediatrics at the University of Toronto. “I think the difference occurs because if you use the endoscopy, which a rigid tube, you can potentially cause more damage, and more long-term scarring.”
Another predictor of esophageal stricture-free episodes was systemic corticosteroid use (a mean of 25.28 months vs. 10.24 months; P less than .001) around the time of the dilatation procedure. “By using systemic steroids, you’re actually decreasing some of the inflammation associated with the trauma of the procedure decreasing the chances of strictures formation,” she said.
Dr. Pope recommended that future studies evaluate the benefit of periprocedural medical interventions on increasing the intervals between esophageal stricture occurrences.
The study was supported by an unrestricted grant from the Epidermolysis Bullosa Research Foundation. She reported having no financial disclosures.
LAKE TAHOE, CALIF. – and direct visualization of these strictures is the preferred method of diagnosis. Those are key findings from a multicenter study that lead author Elena Pope, MD, discussed at the annual meeting of the Society for Pediatric Dermatology.
According to Dr. Pope, who heads the section of dermatology at the Hospital for Sick Children, Toronto, an estimated 10%-17% of epidermolysis bullosa (EB) patients experience strictures, with an overrepresentation in the recessive dystrophic EB subtype in up to 80% of cases. The risk increases with age. “What remains unknown is the best short- and long-term intervention to manage the strictures and predictors/associations for stricture-free episodes,” Dr. Pope said. “The objectives of the current study were to determine the prevalence and predisposing factors for strictures in EB, management options, patient outcomes, and predictors for recurrences and stricture-free intervals.”
She and her associates at seven centers worldwide collected data on 125 EB patients who experienced at least one episode of esophageal stricture. Data was analyzed descriptively and with ANOVA regression analysis for associations/predictors for recurrences/episode-free intervals.
The researchers evaluated 497 stricture events in the 125 patients. A slight female predominance was noted (53%), and the mean age of the first episode was 12.7 years, “which is a little bit older” than the age found in previously published data, Dr. Pope said. As expected, dystrophic EB patients made up most of the sample (98.4%); of these 123 patients, recessive dystrophic EB severe generalized subtype – approaching 50% – was the most common, followed by the recessive dystrophic EB severe intermediate subtype (almost 21%), the dominant dystrophic EB generalized subtype (7%), and other types of dystrophic EB (almost 26%).
The median body mass index percentile for age was 6.3, “so these were patients who were severely malnourished, probably as a result of their strictures as well as their underlying disease,” Dr. Pope said.
As expected, dysphagia was a presenting symptom in most patients (85.5%), while 29.8% presented with inability to swallow solids. The preferred method of evaluation was video fluoroscopy (57.7%), and less commonly with barium swallow (22.3%) or with clinical symptoms alone (0.1%). The mean number of strictures was 1.69; 76.7% were located in the cervical area, 56.7% were located in the thoracic area, and 9.7% were located in the abdominal area. Most patients (76%) had lesions that were 1 cm or longer in size.
Fluoroscopy guidance was the most common method of dilatation (in 45.2% of cases), followed by retrograde endoscopy was (33%), antegrade endoscopy (19.1%), and bougienage (0.1%). General anesthesia was used in most cases (87.6%), and corticosteroids were used around the dilatation in 90.4% of patients. The mean duration of medication use was about 5 days.
As for outcomes after dilatation, 92.2% of strictures completely resolved, 3.8% were partially resolved, 3.9% were not resolved, and 2.7% had complications. The median interval between dilatations was 7 months. Fluoroscopy-guided balloon dilatation was associated with the longest esophageal stricture-free duration (mean of 13.83 months vs. 8.75 months; P less than .001), followed by retrograde endoscopy (mean of 13.10 months vs. 7.85 months; P less than .001), and antegrade endoscopy (mean of 7.63 months vs. 11.46 months; P = .024). “I think this is interesting,” said Dr. Pope, who is also a professor of pediatrics at the University of Toronto. “I think the difference occurs because if you use the endoscopy, which a rigid tube, you can potentially cause more damage, and more long-term scarring.”
Another predictor of esophageal stricture-free episodes was systemic corticosteroid use (a mean of 25.28 months vs. 10.24 months; P less than .001) around the time of the dilatation procedure. “By using systemic steroids, you’re actually decreasing some of the inflammation associated with the trauma of the procedure decreasing the chances of strictures formation,” she said.
Dr. Pope recommended that future studies evaluate the benefit of periprocedural medical interventions on increasing the intervals between esophageal stricture occurrences.
The study was supported by an unrestricted grant from the Epidermolysis Bullosa Research Foundation. She reported having no financial disclosures.
LAKE TAHOE, CALIF. – and direct visualization of these strictures is the preferred method of diagnosis. Those are key findings from a multicenter study that lead author Elena Pope, MD, discussed at the annual meeting of the Society for Pediatric Dermatology.
According to Dr. Pope, who heads the section of dermatology at the Hospital for Sick Children, Toronto, an estimated 10%-17% of epidermolysis bullosa (EB) patients experience strictures, with an overrepresentation in the recessive dystrophic EB subtype in up to 80% of cases. The risk increases with age. “What remains unknown is the best short- and long-term intervention to manage the strictures and predictors/associations for stricture-free episodes,” Dr. Pope said. “The objectives of the current study were to determine the prevalence and predisposing factors for strictures in EB, management options, patient outcomes, and predictors for recurrences and stricture-free intervals.”
She and her associates at seven centers worldwide collected data on 125 EB patients who experienced at least one episode of esophageal stricture. Data was analyzed descriptively and with ANOVA regression analysis for associations/predictors for recurrences/episode-free intervals.
The researchers evaluated 497 stricture events in the 125 patients. A slight female predominance was noted (53%), and the mean age of the first episode was 12.7 years, “which is a little bit older” than the age found in previously published data, Dr. Pope said. As expected, dystrophic EB patients made up most of the sample (98.4%); of these 123 patients, recessive dystrophic EB severe generalized subtype – approaching 50% – was the most common, followed by the recessive dystrophic EB severe intermediate subtype (almost 21%), the dominant dystrophic EB generalized subtype (7%), and other types of dystrophic EB (almost 26%).
The median body mass index percentile for age was 6.3, “so these were patients who were severely malnourished, probably as a result of their strictures as well as their underlying disease,” Dr. Pope said.
As expected, dysphagia was a presenting symptom in most patients (85.5%), while 29.8% presented with inability to swallow solids. The preferred method of evaluation was video fluoroscopy (57.7%), and less commonly with barium swallow (22.3%) or with clinical symptoms alone (0.1%). The mean number of strictures was 1.69; 76.7% were located in the cervical area, 56.7% were located in the thoracic area, and 9.7% were located in the abdominal area. Most patients (76%) had lesions that were 1 cm or longer in size.
Fluoroscopy guidance was the most common method of dilatation (in 45.2% of cases), followed by retrograde endoscopy was (33%), antegrade endoscopy (19.1%), and bougienage (0.1%). General anesthesia was used in most cases (87.6%), and corticosteroids were used around the dilatation in 90.4% of patients. The mean duration of medication use was about 5 days.
As for outcomes after dilatation, 92.2% of strictures completely resolved, 3.8% were partially resolved, 3.9% were not resolved, and 2.7% had complications. The median interval between dilatations was 7 months. Fluoroscopy-guided balloon dilatation was associated with the longest esophageal stricture-free duration (mean of 13.83 months vs. 8.75 months; P less than .001), followed by retrograde endoscopy (mean of 13.10 months vs. 7.85 months; P less than .001), and antegrade endoscopy (mean of 7.63 months vs. 11.46 months; P = .024). “I think this is interesting,” said Dr. Pope, who is also a professor of pediatrics at the University of Toronto. “I think the difference occurs because if you use the endoscopy, which a rigid tube, you can potentially cause more damage, and more long-term scarring.”
Another predictor of esophageal stricture-free episodes was systemic corticosteroid use (a mean of 25.28 months vs. 10.24 months; P less than .001) around the time of the dilatation procedure. “By using systemic steroids, you’re actually decreasing some of the inflammation associated with the trauma of the procedure decreasing the chances of strictures formation,” she said.
Dr. Pope recommended that future studies evaluate the benefit of periprocedural medical interventions on increasing the intervals between esophageal stricture occurrences.
The study was supported by an unrestricted grant from the Epidermolysis Bullosa Research Foundation. She reported having no financial disclosures.
REPORTING FROM SPD 2018
Key clinical point: Esophageal strictures are common complications of patients with severe types of epidermolysis bullosa.
Major finding: Most epidermolysis bullosa patients (85.5%) presented with dysphagia, while the preferred method of evaluation was video fluoroscopy (57.7%).
Study details: A multicenter study of 497 stricture events in 125 patients with epidermolysis bullosa.
Disclosures: The study was supported by an unrestricted grant from the Epidermolysis Bullosa Research Foundation. Dr. Pope reported having no financial disclosures.
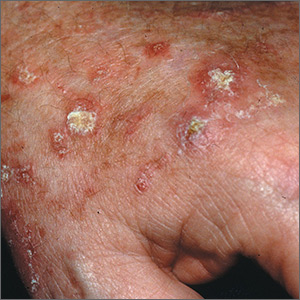
Rough skin on hands
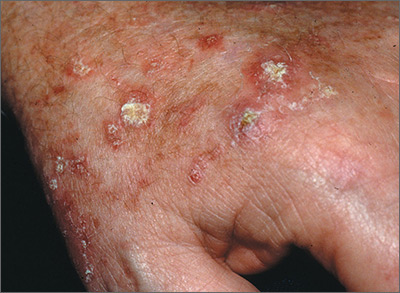
The FP recognized that the lesions on the back of the hands were due to sun damage and included actinic keratosis. However, he was concerned that some of the thicker lesions could be squamous cell carcinoma or SCC in situ.
He discussed options with the patient, which included biopsy of the thickest lesions, cryotherapy, and/or field treatment with a topical agent. The decision was made to biopsy the 2 thickest and whitest lesions and do cryotherapy on some of the other lesions. Shave biopsies were performed and the patient was encouraged to wear sunscreen and minimize his sun exposure. (See the Watch & Learn video on “Shave biopsy.”) On the follow-up visit 2 weeks later, the FP noted that the biopsies and frozen areas were healing. Both biopsies were hypertrophic actinic keratoses only.
The FP then prescribed 5-fluorouracil to be used for 3 to 4 weeks on the remaining lesions on the backs of his hands and forearms. The patient was directed to treat the right arm first to allow more time for the left hand to heal. He was told to stop the 5-fluorouracil if the area became too painful. The patient was also told to treat his left arm the same way after allowing the right arm to heal. Any areas of skin ulceration could be treated with plain petrolatum, but no topical steroids are needed.
Six months later, the patient’s arms and hands looked much better. The patient required regular follow-up.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Usatine R, Wah Y. Actinic keratosis and Bowen disease. In: Usatine R, Smith M, Mayeaux EJ, et al. Color Atlas of Family Medicine. 2nd ed. New York, NY: McGraw-Hill; 2013:969-976.
To learn more about the Color Atlas of Family Medicine, see: www.amazon.com/Color-Family-Medicine-Richard-Usatine/dp/0071769641/.
You can now get the second edition of the Color Atlas of Family Medicine as an app by clicking on this link: usatinemedia.com.

The FP recognized that the lesions on the back of the hands were due to sun damage and included actinic keratosis. However, he was concerned that some of the thicker lesions could be squamous cell carcinoma or SCC in situ.
He discussed options with the patient, which included biopsy of the thickest lesions, cryotherapy, and/or field treatment with a topical agent. The decision was made to biopsy the 2 thickest and whitest lesions and do cryotherapy on some of the other lesions. Shave biopsies were performed and the patient was encouraged to wear sunscreen and minimize his sun exposure. (See the Watch & Learn video on “Shave biopsy.”) On the follow-up visit 2 weeks later, the FP noted that the biopsies and frozen areas were healing. Both biopsies were hypertrophic actinic keratoses only.
The FP then prescribed 5-fluorouracil to be used for 3 to 4 weeks on the remaining lesions on the backs of his hands and forearms. The patient was directed to treat the right arm first to allow more time for the left hand to heal. He was told to stop the 5-fluorouracil if the area became too painful. The patient was also told to treat his left arm the same way after allowing the right arm to heal. Any areas of skin ulceration could be treated with plain petrolatum, but no topical steroids are needed.
Six months later, the patient’s arms and hands looked much better. The patient required regular follow-up.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Usatine R, Wah Y. Actinic keratosis and Bowen disease. In: Usatine R, Smith M, Mayeaux EJ, et al. Color Atlas of Family Medicine. 2nd ed. New York, NY: McGraw-Hill; 2013:969-976.
To learn more about the Color Atlas of Family Medicine, see: www.amazon.com/Color-Family-Medicine-Richard-Usatine/dp/0071769641/.
You can now get the second edition of the Color Atlas of Family Medicine as an app by clicking on this link: usatinemedia.com.

The FP recognized that the lesions on the back of the hands were due to sun damage and included actinic keratosis. However, he was concerned that some of the thicker lesions could be squamous cell carcinoma or SCC in situ.
He discussed options with the patient, which included biopsy of the thickest lesions, cryotherapy, and/or field treatment with a topical agent. The decision was made to biopsy the 2 thickest and whitest lesions and do cryotherapy on some of the other lesions. Shave biopsies were performed and the patient was encouraged to wear sunscreen and minimize his sun exposure. (See the Watch & Learn video on “Shave biopsy.”) On the follow-up visit 2 weeks later, the FP noted that the biopsies and frozen areas were healing. Both biopsies were hypertrophic actinic keratoses only.
The FP then prescribed 5-fluorouracil to be used for 3 to 4 weeks on the remaining lesions on the backs of his hands and forearms. The patient was directed to treat the right arm first to allow more time for the left hand to heal. He was told to stop the 5-fluorouracil if the area became too painful. The patient was also told to treat his left arm the same way after allowing the right arm to heal. Any areas of skin ulceration could be treated with plain petrolatum, but no topical steroids are needed.
Six months later, the patient’s arms and hands looked much better. The patient required regular follow-up.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Usatine R, Wah Y. Actinic keratosis and Bowen disease. In: Usatine R, Smith M, Mayeaux EJ, et al. Color Atlas of Family Medicine. 2nd ed. New York, NY: McGraw-Hill; 2013:969-976.
To learn more about the Color Atlas of Family Medicine, see: www.amazon.com/Color-Family-Medicine-Richard-Usatine/dp/0071769641/.
You can now get the second edition of the Color Atlas of Family Medicine as an app by clicking on this link: usatinemedia.com.
The Mother of All Skin Problems
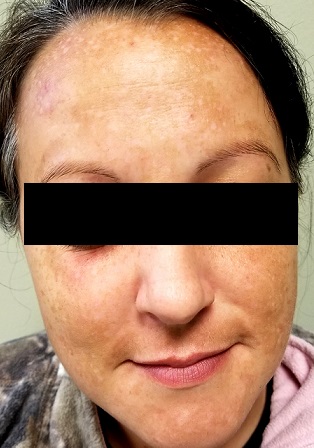
Each time this 32-year-old woman has a baby—she’s had four to date—she notices that sections of her face darken. Early on, she observed a pattern in which the coming of winter coincided with a lightening of these affected areas—but now the effect lasts year-round, with progressive darkening. She has not tried any products (OTC or prescription) for this problem.
Growing up in the South, the patient and her family spent most summers boating, swimming, and fishing. Her use of sunscreen was sporadic, but she would tan easily regardless.
Her health is good, aside from a 15-year history of smoking.
EXAMINATION
There is excessive hyperpigmentation (brown) on the patient’s face. It follows a mask-like pattern, including her maxilla and the periphery of her face.
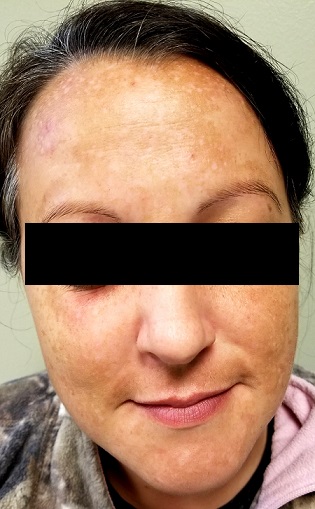
Elsewhere, there is abundant evidence of excessive sun exposure, with focal hyperpigmentation and telangiectasias on her arms. She has type IV skin, consistent with her Native American ancestry.
What is the diagnosis?
DISCUSSION
Melasma, also known as chloasma and dubbed the “mask of pregnancy,” is an extremely common problem that results from a combination of naturally dark skin, lots of sun exposure, and increased levels of estrogenic hormones. The latter can result from pregnancy or from oral contraceptive or estrogen replacement therapy use. Another precipitating factor is thyroid disease, certain types of which lead to an increase in melanocytic stimulating hormone.
Melasma, as one might expect, is seen almost exclusively in women, though a rare male is affected. It is especially common among Latina, Native American, and African-American women, whose melanocytes are especially able to produce pigment.
There are several treatments for melasma, none of them perfect, including tretinoin, azelaic acid, chemical peels, dermabrasion, and lasers. The most common treatment is hydroquinone cream, available in the US in both OTC (2%) and prescription (4%) strengths. However, hydroquinone is available OTC in stronger formulations (15% to 20%) in many Central and South American countries; unfortunately, many women who obtain and use these products experience exogenous ochronosis—a worsening or even precipitation of melasma, resulting from excessive production of tyrosinase.
Any treatment must be used in conjunction with rigorous sunscreen application. A full-spectrum product, with titanium dioxide and zinc oxide as the only active ingredients, must be used, because chemical-laden sunscreens don’t do as good a job covering UVA, UVB, and visible light. Convincing women who are unaccustomed to needing sunscreen to use it religiously is part of what makes treating melasma difficult.
The differential for melasma includes postinflammatory hyperpigmentation (eg, following an episode of contact dermatitis) and simple solar lentigines.
This patient was treated with hydroquinone 4% cream bid, plus sunscreen. She was also given information about other treatment options, such as laser and dermabrasion.
TAKE-HOME LEARNING POINTS
- Melasma, also known at chloasma, is quite common, especially among women with darker skin who live in sunny parts of the world.
- It results from a combination of dark skin, an increased level of estrogenic hormones (eg, with pregnancy, birth control pills, or estrogen replacement therapy), and excessive exposure to UV light.
- While hydroquinone cream can be an effective treatment, the maximum strength should be 4%; overuse of stronger concentrations (available in other countries, such as Mexico), can actually cause melasma to worsen.
Each time this 32-year-old woman has a baby—she’s had four to date—she notices that sections of her face darken. Early on, she observed a pattern in which the coming of winter coincided with a lightening of these affected areas—but now the effect lasts year-round, with progressive darkening. She has not tried any products (OTC or prescription) for this problem.
Growing up in the South, the patient and her family spent most summers boating, swimming, and fishing. Her use of sunscreen was sporadic, but she would tan easily regardless.
Her health is good, aside from a 15-year history of smoking.
EXAMINATION
There is excessive hyperpigmentation (brown) on the patient’s face. It follows a mask-like pattern, including her maxilla and the periphery of her face.

Elsewhere, there is abundant evidence of excessive sun exposure, with focal hyperpigmentation and telangiectasias on her arms. She has type IV skin, consistent with her Native American ancestry.
What is the diagnosis?
DISCUSSION
Melasma, also known as chloasma and dubbed the “mask of pregnancy,” is an extremely common problem that results from a combination of naturally dark skin, lots of sun exposure, and increased levels of estrogenic hormones. The latter can result from pregnancy or from oral contraceptive or estrogen replacement therapy use. Another precipitating factor is thyroid disease, certain types of which lead to an increase in melanocytic stimulating hormone.
Melasma, as one might expect, is seen almost exclusively in women, though a rare male is affected. It is especially common among Latina, Native American, and African-American women, whose melanocytes are especially able to produce pigment.
There are several treatments for melasma, none of them perfect, including tretinoin, azelaic acid, chemical peels, dermabrasion, and lasers. The most common treatment is hydroquinone cream, available in the US in both OTC (2%) and prescription (4%) strengths. However, hydroquinone is available OTC in stronger formulations (15% to 20%) in many Central and South American countries; unfortunately, many women who obtain and use these products experience exogenous ochronosis—a worsening or even precipitation of melasma, resulting from excessive production of tyrosinase.
Any treatment must be used in conjunction with rigorous sunscreen application. A full-spectrum product, with titanium dioxide and zinc oxide as the only active ingredients, must be used, because chemical-laden sunscreens don’t do as good a job covering UVA, UVB, and visible light. Convincing women who are unaccustomed to needing sunscreen to use it religiously is part of what makes treating melasma difficult.
The differential for melasma includes postinflammatory hyperpigmentation (eg, following an episode of contact dermatitis) and simple solar lentigines.
This patient was treated with hydroquinone 4% cream bid, plus sunscreen. She was also given information about other treatment options, such as laser and dermabrasion.
TAKE-HOME LEARNING POINTS
- Melasma, also known at chloasma, is quite common, especially among women with darker skin who live in sunny parts of the world.
- It results from a combination of dark skin, an increased level of estrogenic hormones (eg, with pregnancy, birth control pills, or estrogen replacement therapy), and excessive exposure to UV light.
- While hydroquinone cream can be an effective treatment, the maximum strength should be 4%; overuse of stronger concentrations (available in other countries, such as Mexico), can actually cause melasma to worsen.
Each time this 32-year-old woman has a baby—she’s had four to date—she notices that sections of her face darken. Early on, she observed a pattern in which the coming of winter coincided with a lightening of these affected areas—but now the effect lasts year-round, with progressive darkening. She has not tried any products (OTC or prescription) for this problem.
Growing up in the South, the patient and her family spent most summers boating, swimming, and fishing. Her use of sunscreen was sporadic, but she would tan easily regardless.
Her health is good, aside from a 15-year history of smoking.
EXAMINATION
There is excessive hyperpigmentation (brown) on the patient’s face. It follows a mask-like pattern, including her maxilla and the periphery of her face.

Elsewhere, there is abundant evidence of excessive sun exposure, with focal hyperpigmentation and telangiectasias on her arms. She has type IV skin, consistent with her Native American ancestry.
What is the diagnosis?
DISCUSSION
Melasma, also known as chloasma and dubbed the “mask of pregnancy,” is an extremely common problem that results from a combination of naturally dark skin, lots of sun exposure, and increased levels of estrogenic hormones. The latter can result from pregnancy or from oral contraceptive or estrogen replacement therapy use. Another precipitating factor is thyroid disease, certain types of which lead to an increase in melanocytic stimulating hormone.
Melasma, as one might expect, is seen almost exclusively in women, though a rare male is affected. It is especially common among Latina, Native American, and African-American women, whose melanocytes are especially able to produce pigment.
There are several treatments for melasma, none of them perfect, including tretinoin, azelaic acid, chemical peels, dermabrasion, and lasers. The most common treatment is hydroquinone cream, available in the US in both OTC (2%) and prescription (4%) strengths. However, hydroquinone is available OTC in stronger formulations (15% to 20%) in many Central and South American countries; unfortunately, many women who obtain and use these products experience exogenous ochronosis—a worsening or even precipitation of melasma, resulting from excessive production of tyrosinase.
Any treatment must be used in conjunction with rigorous sunscreen application. A full-spectrum product, with titanium dioxide and zinc oxide as the only active ingredients, must be used, because chemical-laden sunscreens don’t do as good a job covering UVA, UVB, and visible light. Convincing women who are unaccustomed to needing sunscreen to use it religiously is part of what makes treating melasma difficult.
The differential for melasma includes postinflammatory hyperpigmentation (eg, following an episode of contact dermatitis) and simple solar lentigines.
This patient was treated with hydroquinone 4% cream bid, plus sunscreen. She was also given information about other treatment options, such as laser and dermabrasion.
TAKE-HOME LEARNING POINTS
- Melasma, also known at chloasma, is quite common, especially among women with darker skin who live in sunny parts of the world.
- It results from a combination of dark skin, an increased level of estrogenic hormones (eg, with pregnancy, birth control pills, or estrogen replacement therapy), and excessive exposure to UV light.
- While hydroquinone cream can be an effective treatment, the maximum strength should be 4%; overuse of stronger concentrations (available in other countries, such as Mexico), can actually cause melasma to worsen.
Foster cultural competence when examining hair, scalp of ethnic patients
LAKE TAHOE, CALIF. – The way Susan C. Taylor, MD, sees it,
At the annual meeting of the Society for Pediatric Dermatology, Dr. Taylor, a dermatologist at the University of Pennsylvania, Philadelphia, defined culturally competent care as a patient-centered approach in which clinicians establish a rapport with the patient and the caregiver. “It’s important that we ask the right questions,” she said. “In doing so, we have to be familiar with common hair care practices. It’s important that we respect our patients’ values, their goals, their health needs, and, of course, their cultural background. Finally, we have to engage in shared decision making. That’s where we can improve compliance and lead to an overall very satisfactory patient visit.”
To illustrate this point, she discussed the case of a four-year-old black female with a 9-month history of bad dandruff. The child’s mother reports thick flakes that never go away. She takes pride in caring for her daughter’s hair, and shampoos it every two weeks. “She tells me that during the 2.5 hours that it takes her on Saturdays to shampoo, detangle, and comb her daughter’s hair, which she then braids or cornrows and adorns with barrettes or balls, it is a great bonding experience for the two of them,” Dr. Taylor said. “The flakes are temporarily better after she ‘greases’ her daughter’s scalp, but after 2-3 days, they are back.”
In a case like this, Dr. Taylor recommends asking the parent or caregiver to join you while you examine the scalp. This way, the focus becomes the child’s scalp, and the parent is not just staring at your expressions. “The child also observes the pediatric dermatologist and parent/caregiver working together as a team,” she said. “You also want to ask the parent to remove the hair adornments. This makes the child feel more comfortable. It also allows you to observe how the hair is being managed. Are the adornments being removed gently? Is there aggressive pulling of the hair when they take out the braids? Is the child visibly wincing in pain? If the latter two happen this is a teachable moment. You can point out, ‘It looks like Susie is in pain. Let’s do it a little more gently. That might prevent further hair breakage.’ ”
The differential diagnosis of a scaly pediatric scalp includes infrequent shampooing, seborrheic dermatitis, tinea capitis, atopic dermatitis, psoriasis, and sebopsoriasis. The type of hairstyle also factors in. For example, cornrows are a popular hair styling option among ethnic patients. “These are very popular and very time consuming and may lead to infrequent shampooing,” she said. “If they’re put in very tightly or if they have beads or other adornments, it can lead to traction alopecia.” Twists, meanwhile, can create tension on the hair, while puffs can cause traction alopecia if they’re pulled too tightly. “Although dreadlocks are more common among adolescents, we’re seeing them more commonly in young children,” she said. “They can be very long and wavy and lead to traction alopecia.”
The time required to shampoo, detangle, and style tightly coiled African American hair can be significant. For example, in children with cornrows or braids with extensions, Dr. Taylor said that it might require 30 minutes to as long as 2 or 3 hours to remove their current hairstyle, followed by shampooing and conditioning. “Detangling can take at least 15 minutes. In tightly coiled African American hair, studies have demonstrated that detangling while the hair is wet is best, because you have fewer forces on that comb and the hair is less likely to break, as opposed to detangling when the hair is dry. After the wet hair is detangled and the conditioner is rinsed out, a leave-in conditioner is often applied. Then the hair is detangled again, which can take up to an hour, followed by styling, which can take 1-3 hours. That gives you some insight as to why there can often be infrequent shampooing.”
The recommended frequency of shampooing depends on the hairstyle selected. Many children with braids and extensions will have those braids and extensions taken out every six to 12 weeks, “but that doesn’t mean that the scalp can’t be shampooed,” Dr. Taylor said. “The scalp should be shampooed more often. Economics and socioeconomic status play into the frequency of shampooing. For example, if a parent or a caregiver sends a child to a hair stylist, that can range in price from $45 to $65 or more. Time also factors in. In the black community in particular, it’s a ritual on a Saturday to get your hair done. If the parent or caregiver works on weekends, that’s going to impact the frequency of shampooing.”
Dr. Taylor underscored the importance of framing the history-taking process to avoid common pitfall questions like “Do you wash the child’s hair every day or every other day?” or “Do you use dandruff shampoo every day?” It is important to remember that “the parent’s inherent perception is of a doctor who does not have my hair probably does not understand my hair or my child’s hair,” she said. “It’s unlikely that you’re going to find a parent who shampoos their child’s hair every day or every other day. Maybe once a week, probably biweekly. It’s important to ask culturally competent questions.”
She also advises against asking about shampooing when you’re examining the child’s hair, “because there’s going to be the perception that you may think the scalp is dirty,” Dr. Taylor explained. “You probably want to ask that when gathering the history of present illness. The culturally competent question is going to be, ‘Do you wash her/his hair weekly, every other week, monthly, or does it depend on the hair style?’ ”
Body language is also important. “Don’t lean in from afar when examining the patient,” she said. “Get up close and touch the child’s hair.” If you choose to wear surgical gloves for the exam “don’t hold your hands in the surgical scrub position,” she recommended. “Hold your hands in a more neutral position. I think it’s important to touch the hair.”
Referring back to the 4-year-old black child with bad dandruff, she said that a diagnosis of seborrheic dermatitis is unlikely since that condition usually occurs during puberty. “You should have a high index of suspicion for tinea capitis,” she said. “If the patient has occipital lymphadenopathy plus scaling of the scalp or alopecia, that’s enough to presumptively treat for tinea capitis. There are studies that support that.”
For established tinea capitis, Dr. Taylor advises parents to wash barrettes and other hair adornments in hot soapy water or in the dishwasher. She also recommends disposal of hair oil, pomade, and grease and shampooing the child’s hair with ketoconazole and use a conditioner to decrease household and patient spread, which decreases transmissible fungal spores. “There’s a misperception that the application of hair oils and grease can increase the rate of tinea capitis,” she noted. “That’s not true. However, if hair grease and hair oil is applied to the scalp within one week of culture, it could produce a false negative culture.”
For established seborrheic dermatitis, antifungal shampoos including ketoconazole, ciclopirox, and selenium sulfide may be too drying for ethnic hair, “which already has a propensity to break,” she said. “Instead, we recommend a 5-10 minute scalp contact time with the shampoo and avoid contact with strands of hair. Shampoo hair strands with a conditioning shampoo followed by a conditioner to limit hair breakage. We suggest once weekly or biweekly shampooing.”
Dr. Taylor disclosed that she has advisory board and/or investigator relationships with Aclaris Therapeutics, Allergan, Beiersdorf, Croma Pharmaceuticals, Galderma, Isdin, Johnson & Johnson, and Unilever. She also acknowledged Candrice R. Heath, MD, a dermatologist based in Newark, Delaware, for her assistance with the presentation content.
[email protected]
LAKE TAHOE, CALIF. – The way Susan C. Taylor, MD, sees it,
At the annual meeting of the Society for Pediatric Dermatology, Dr. Taylor, a dermatologist at the University of Pennsylvania, Philadelphia, defined culturally competent care as a patient-centered approach in which clinicians establish a rapport with the patient and the caregiver. “It’s important that we ask the right questions,” she said. “In doing so, we have to be familiar with common hair care practices. It’s important that we respect our patients’ values, their goals, their health needs, and, of course, their cultural background. Finally, we have to engage in shared decision making. That’s where we can improve compliance and lead to an overall very satisfactory patient visit.”
To illustrate this point, she discussed the case of a four-year-old black female with a 9-month history of bad dandruff. The child’s mother reports thick flakes that never go away. She takes pride in caring for her daughter’s hair, and shampoos it every two weeks. “She tells me that during the 2.5 hours that it takes her on Saturdays to shampoo, detangle, and comb her daughter’s hair, which she then braids or cornrows and adorns with barrettes or balls, it is a great bonding experience for the two of them,” Dr. Taylor said. “The flakes are temporarily better after she ‘greases’ her daughter’s scalp, but after 2-3 days, they are back.”
In a case like this, Dr. Taylor recommends asking the parent or caregiver to join you while you examine the scalp. This way, the focus becomes the child’s scalp, and the parent is not just staring at your expressions. “The child also observes the pediatric dermatologist and parent/caregiver working together as a team,” she said. “You also want to ask the parent to remove the hair adornments. This makes the child feel more comfortable. It also allows you to observe how the hair is being managed. Are the adornments being removed gently? Is there aggressive pulling of the hair when they take out the braids? Is the child visibly wincing in pain? If the latter two happen this is a teachable moment. You can point out, ‘It looks like Susie is in pain. Let’s do it a little more gently. That might prevent further hair breakage.’ ”
The differential diagnosis of a scaly pediatric scalp includes infrequent shampooing, seborrheic dermatitis, tinea capitis, atopic dermatitis, psoriasis, and sebopsoriasis. The type of hairstyle also factors in. For example, cornrows are a popular hair styling option among ethnic patients. “These are very popular and very time consuming and may lead to infrequent shampooing,” she said. “If they’re put in very tightly or if they have beads or other adornments, it can lead to traction alopecia.” Twists, meanwhile, can create tension on the hair, while puffs can cause traction alopecia if they’re pulled too tightly. “Although dreadlocks are more common among adolescents, we’re seeing them more commonly in young children,” she said. “They can be very long and wavy and lead to traction alopecia.”
The time required to shampoo, detangle, and style tightly coiled African American hair can be significant. For example, in children with cornrows or braids with extensions, Dr. Taylor said that it might require 30 minutes to as long as 2 or 3 hours to remove their current hairstyle, followed by shampooing and conditioning. “Detangling can take at least 15 minutes. In tightly coiled African American hair, studies have demonstrated that detangling while the hair is wet is best, because you have fewer forces on that comb and the hair is less likely to break, as opposed to detangling when the hair is dry. After the wet hair is detangled and the conditioner is rinsed out, a leave-in conditioner is often applied. Then the hair is detangled again, which can take up to an hour, followed by styling, which can take 1-3 hours. That gives you some insight as to why there can often be infrequent shampooing.”
The recommended frequency of shampooing depends on the hairstyle selected. Many children with braids and extensions will have those braids and extensions taken out every six to 12 weeks, “but that doesn’t mean that the scalp can’t be shampooed,” Dr. Taylor said. “The scalp should be shampooed more often. Economics and socioeconomic status play into the frequency of shampooing. For example, if a parent or a caregiver sends a child to a hair stylist, that can range in price from $45 to $65 or more. Time also factors in. In the black community in particular, it’s a ritual on a Saturday to get your hair done. If the parent or caregiver works on weekends, that’s going to impact the frequency of shampooing.”
Dr. Taylor underscored the importance of framing the history-taking process to avoid common pitfall questions like “Do you wash the child’s hair every day or every other day?” or “Do you use dandruff shampoo every day?” It is important to remember that “the parent’s inherent perception is of a doctor who does not have my hair probably does not understand my hair or my child’s hair,” she said. “It’s unlikely that you’re going to find a parent who shampoos their child’s hair every day or every other day. Maybe once a week, probably biweekly. It’s important to ask culturally competent questions.”
She also advises against asking about shampooing when you’re examining the child’s hair, “because there’s going to be the perception that you may think the scalp is dirty,” Dr. Taylor explained. “You probably want to ask that when gathering the history of present illness. The culturally competent question is going to be, ‘Do you wash her/his hair weekly, every other week, monthly, or does it depend on the hair style?’ ”
Body language is also important. “Don’t lean in from afar when examining the patient,” she said. “Get up close and touch the child’s hair.” If you choose to wear surgical gloves for the exam “don’t hold your hands in the surgical scrub position,” she recommended. “Hold your hands in a more neutral position. I think it’s important to touch the hair.”
Referring back to the 4-year-old black child with bad dandruff, she said that a diagnosis of seborrheic dermatitis is unlikely since that condition usually occurs during puberty. “You should have a high index of suspicion for tinea capitis,” she said. “If the patient has occipital lymphadenopathy plus scaling of the scalp or alopecia, that’s enough to presumptively treat for tinea capitis. There are studies that support that.”
For established tinea capitis, Dr. Taylor advises parents to wash barrettes and other hair adornments in hot soapy water or in the dishwasher. She also recommends disposal of hair oil, pomade, and grease and shampooing the child’s hair with ketoconazole and use a conditioner to decrease household and patient spread, which decreases transmissible fungal spores. “There’s a misperception that the application of hair oils and grease can increase the rate of tinea capitis,” she noted. “That’s not true. However, if hair grease and hair oil is applied to the scalp within one week of culture, it could produce a false negative culture.”
For established seborrheic dermatitis, antifungal shampoos including ketoconazole, ciclopirox, and selenium sulfide may be too drying for ethnic hair, “which already has a propensity to break,” she said. “Instead, we recommend a 5-10 minute scalp contact time with the shampoo and avoid contact with strands of hair. Shampoo hair strands with a conditioning shampoo followed by a conditioner to limit hair breakage. We suggest once weekly or biweekly shampooing.”
Dr. Taylor disclosed that she has advisory board and/or investigator relationships with Aclaris Therapeutics, Allergan, Beiersdorf, Croma Pharmaceuticals, Galderma, Isdin, Johnson & Johnson, and Unilever. She also acknowledged Candrice R. Heath, MD, a dermatologist based in Newark, Delaware, for her assistance with the presentation content.
[email protected]
LAKE TAHOE, CALIF. – The way Susan C. Taylor, MD, sees it,
At the annual meeting of the Society for Pediatric Dermatology, Dr. Taylor, a dermatologist at the University of Pennsylvania, Philadelphia, defined culturally competent care as a patient-centered approach in which clinicians establish a rapport with the patient and the caregiver. “It’s important that we ask the right questions,” she said. “In doing so, we have to be familiar with common hair care practices. It’s important that we respect our patients’ values, their goals, their health needs, and, of course, their cultural background. Finally, we have to engage in shared decision making. That’s where we can improve compliance and lead to an overall very satisfactory patient visit.”
To illustrate this point, she discussed the case of a four-year-old black female with a 9-month history of bad dandruff. The child’s mother reports thick flakes that never go away. She takes pride in caring for her daughter’s hair, and shampoos it every two weeks. “She tells me that during the 2.5 hours that it takes her on Saturdays to shampoo, detangle, and comb her daughter’s hair, which she then braids or cornrows and adorns with barrettes or balls, it is a great bonding experience for the two of them,” Dr. Taylor said. “The flakes are temporarily better after she ‘greases’ her daughter’s scalp, but after 2-3 days, they are back.”
In a case like this, Dr. Taylor recommends asking the parent or caregiver to join you while you examine the scalp. This way, the focus becomes the child’s scalp, and the parent is not just staring at your expressions. “The child also observes the pediatric dermatologist and parent/caregiver working together as a team,” she said. “You also want to ask the parent to remove the hair adornments. This makes the child feel more comfortable. It also allows you to observe how the hair is being managed. Are the adornments being removed gently? Is there aggressive pulling of the hair when they take out the braids? Is the child visibly wincing in pain? If the latter two happen this is a teachable moment. You can point out, ‘It looks like Susie is in pain. Let’s do it a little more gently. That might prevent further hair breakage.’ ”
The differential diagnosis of a scaly pediatric scalp includes infrequent shampooing, seborrheic dermatitis, tinea capitis, atopic dermatitis, psoriasis, and sebopsoriasis. The type of hairstyle also factors in. For example, cornrows are a popular hair styling option among ethnic patients. “These are very popular and very time consuming and may lead to infrequent shampooing,” she said. “If they’re put in very tightly or if they have beads or other adornments, it can lead to traction alopecia.” Twists, meanwhile, can create tension on the hair, while puffs can cause traction alopecia if they’re pulled too tightly. “Although dreadlocks are more common among adolescents, we’re seeing them more commonly in young children,” she said. “They can be very long and wavy and lead to traction alopecia.”
The time required to shampoo, detangle, and style tightly coiled African American hair can be significant. For example, in children with cornrows or braids with extensions, Dr. Taylor said that it might require 30 minutes to as long as 2 or 3 hours to remove their current hairstyle, followed by shampooing and conditioning. “Detangling can take at least 15 minutes. In tightly coiled African American hair, studies have demonstrated that detangling while the hair is wet is best, because you have fewer forces on that comb and the hair is less likely to break, as opposed to detangling when the hair is dry. After the wet hair is detangled and the conditioner is rinsed out, a leave-in conditioner is often applied. Then the hair is detangled again, which can take up to an hour, followed by styling, which can take 1-3 hours. That gives you some insight as to why there can often be infrequent shampooing.”
The recommended frequency of shampooing depends on the hairstyle selected. Many children with braids and extensions will have those braids and extensions taken out every six to 12 weeks, “but that doesn’t mean that the scalp can’t be shampooed,” Dr. Taylor said. “The scalp should be shampooed more often. Economics and socioeconomic status play into the frequency of shampooing. For example, if a parent or a caregiver sends a child to a hair stylist, that can range in price from $45 to $65 or more. Time also factors in. In the black community in particular, it’s a ritual on a Saturday to get your hair done. If the parent or caregiver works on weekends, that’s going to impact the frequency of shampooing.”
Dr. Taylor underscored the importance of framing the history-taking process to avoid common pitfall questions like “Do you wash the child’s hair every day or every other day?” or “Do you use dandruff shampoo every day?” It is important to remember that “the parent’s inherent perception is of a doctor who does not have my hair probably does not understand my hair or my child’s hair,” she said. “It’s unlikely that you’re going to find a parent who shampoos their child’s hair every day or every other day. Maybe once a week, probably biweekly. It’s important to ask culturally competent questions.”
She also advises against asking about shampooing when you’re examining the child’s hair, “because there’s going to be the perception that you may think the scalp is dirty,” Dr. Taylor explained. “You probably want to ask that when gathering the history of present illness. The culturally competent question is going to be, ‘Do you wash her/his hair weekly, every other week, monthly, or does it depend on the hair style?’ ”
Body language is also important. “Don’t lean in from afar when examining the patient,” she said. “Get up close and touch the child’s hair.” If you choose to wear surgical gloves for the exam “don’t hold your hands in the surgical scrub position,” she recommended. “Hold your hands in a more neutral position. I think it’s important to touch the hair.”
Referring back to the 4-year-old black child with bad dandruff, she said that a diagnosis of seborrheic dermatitis is unlikely since that condition usually occurs during puberty. “You should have a high index of suspicion for tinea capitis,” she said. “If the patient has occipital lymphadenopathy plus scaling of the scalp or alopecia, that’s enough to presumptively treat for tinea capitis. There are studies that support that.”
For established tinea capitis, Dr. Taylor advises parents to wash barrettes and other hair adornments in hot soapy water or in the dishwasher. She also recommends disposal of hair oil, pomade, and grease and shampooing the child’s hair with ketoconazole and use a conditioner to decrease household and patient spread, which decreases transmissible fungal spores. “There’s a misperception that the application of hair oils and grease can increase the rate of tinea capitis,” she noted. “That’s not true. However, if hair grease and hair oil is applied to the scalp within one week of culture, it could produce a false negative culture.”
For established seborrheic dermatitis, antifungal shampoos including ketoconazole, ciclopirox, and selenium sulfide may be too drying for ethnic hair, “which already has a propensity to break,” she said. “Instead, we recommend a 5-10 minute scalp contact time with the shampoo and avoid contact with strands of hair. Shampoo hair strands with a conditioning shampoo followed by a conditioner to limit hair breakage. We suggest once weekly or biweekly shampooing.”
Dr. Taylor disclosed that she has advisory board and/or investigator relationships with Aclaris Therapeutics, Allergan, Beiersdorf, Croma Pharmaceuticals, Galderma, Isdin, Johnson & Johnson, and Unilever. She also acknowledged Candrice R. Heath, MD, a dermatologist based in Newark, Delaware, for her assistance with the presentation content.
[email protected]
EXPERT ANALYSIS FROM THE SPD ANNUAL MEETING
Calcific uremic arteriolopathy
A 51-year-old man with end-stage renal disease, on peritoneal dialysis for the past 4 years, presented to the emergency department with severe pain in both legs. The pain had started 2 months previously and had progressively worsened. After multiple admissions in the past for hyperkalemia and volume overload due to noncompliance, he had been advised to switch to hemodialysis.
See related article and editorial
Laboratory analysis revealed the following values:
- Serum creatinine 12.62 mg/dL (reference range 0.73–1.22)
- Blood urea nitrogen 159 mg/dL (9–24)
- Serum calcium corrected for serum albumin 8.1 mg/dL (8.4–10.0)
- Serum phosphorus 10.6 mg/dL (2.7–4.8).
His history of end-stage renal disease, failure of peritoneal dialysis, high calcium-phosphorus product (8.1 mg/dL × 10.6 mg/dL = 85.9 mg2/dL2, reference range ≤ 55), and characteristic physical findings led to the diagnosis of calcific uremic arteriolopathy.
CALCIFIC UREMIC ARTERIOLOPATHY
Calcific uremic arteriolopathy or “calciphylaxis,” seen most often in patients with end-stage renal disease, is caused by calcium deposition in the media of the dermo-hypodermic arterioles, leading to infarction of adjacent tissue.1–3 A high calcium-phosphorus product (> 55) has been implicated in its development; however, the calcium-phosphorus product can be normal despite hyperphosphatemia, which itself may promote ectopic calcification.
Early ischemic manifestations include livedo reticularis and painful retiform purpura on the thighs and other areas of high adiposity. Lesions evolve into violaceous plaquelike subcutaneous nodules that can infarct, become necrotic, ulcerate, and become infected. Punch biopsy demonstrating arteriolar calcification, subintimal fibrosis, and thrombosis confirms the diagnosis.
Differential diagnosis
Warfarin necrosis can cause large, irregular, bloody bullae that ulcerate and turn into eschar that may resemble lesions of calcific uremic arteriolopathy. Our patient, however, had no exposure to warfarin.
Pemphigus foliaceus, an immunoglobulin G4-mediated autoimmune disorder targeted against desmoglein-1, leads to the formation of fragile blisters that easily rupture when rubbed (Nikolsky sign). Lesions evolve into scaling, crusty erosions on an erythematous base. With tender blisters and lack of mucous membrane involvement, pemphigus foliaceus shares similarities with calcific uremic arteriolopathy, but the presence of necrotic eschar surrounded by violaceous plaques in our patient made it an unlikely diagnosis.
Cryofibrinogenemia. In the right clinical scenario, ie, in a patient with vasculitis, malignancy, infection, cryoglobulinemia, or collagen diseases, cryofibrinogen-mediated cold-induced occlusive lesions may mimic calcific uremic arteriolopathy, with painful or pruritic erythema, purpura, livedo reticularis, necrosis, and ulceration.4 Our patient had no color changes with exposure to cold, nor any history of Raynaud phenomenon or joint pain, making the diagnosis of cryofibrinogenemia less likely.
Nephrogenic systemic fibrosis. Gadolinium contrast medium in magnetic resonance imaging can cause nephrogenic systemic fibrosis, characterized by erythematous papules that coalesce into brawny plaques with surrounding woody induration, which may resemble lesions of calcific uremic arteriolopathy.5 However, our patient had not been exposed to gadolinium.
Management
Management is multidisciplinary and includes the following1:
- Hemodialysis, modified to optimize calcium balance2
- Intravenous sodium thiosulfate: the exact mechanism of action remains unclear, but it is thought to play a role in chelating calcium from tissue deposits, thus decreasing pain and promoting regression of skin lesions3
- Wound care, including chemical debridement agents, negative-pressure wound therapy, and surgical debridement for infected wounds6
- Pain management with opioid analgesics.
The patient was treated with all these measures. However, he died of sudden cardiac arrest during the same admission.
- Weenig RH, Sewell LD, Davis MD, McCarthy JT, Pittelkow MR. Calciphylaxis: natural history, risk factor analysis, and outcome. J Am Acad Dermatol 2007; 56(4):569–579. doi:10.1016/j.jaad.2006.08.065
- Nigwekar SU, Kroshinsky D, Nazarian RM, et al. Calciphylaxis: risk factors, diagnosis, and treatment. Am J Kidney Dis 2015; 66(1):133–146. doi:10.1053/j.ajkd.2015.01.034
- Janigan DT, Hirsch DJ, Klassen GA, MacDonald AS. Calcified subcutaneous arterioles with infarcts of the subcutis and skin (“calciphylaxis”) in chronic renal failure. Am J Kidney Dis 2000; 35(4):588–597. pmid:10739777
- Michaud M, Pourrat J. Cryofibrinogenemia. J Clin Rheumatol 2013; 19(3):142–148. doi:10.1097/RHU.0b013e318289e06e
- Galan A, Cowper SE, Bucala R. Nephrogenic systemic fibrosis (nephrogenic fibrosing dermopathy). Curr Opin Rheumatol 2006; 18(6):614–617. doi:10.1097/01.bor.0000245725.94887.8d
- Freedberg IM, Eisen AZ, Wolff K, Austen KF, Goldsmith LA, Katz SI, eds. Fitzpatrick’s Dermatology in General Medicine. 6th ed. New York, NY: McGraw-Hill Professional; 2003:558–562.
A 51-year-old man with end-stage renal disease, on peritoneal dialysis for the past 4 years, presented to the emergency department with severe pain in both legs. The pain had started 2 months previously and had progressively worsened. After multiple admissions in the past for hyperkalemia and volume overload due to noncompliance, he had been advised to switch to hemodialysis.
See related article and editorial
Laboratory analysis revealed the following values:
- Serum creatinine 12.62 mg/dL (reference range 0.73–1.22)
- Blood urea nitrogen 159 mg/dL (9–24)
- Serum calcium corrected for serum albumin 8.1 mg/dL (8.4–10.0)
- Serum phosphorus 10.6 mg/dL (2.7–4.8).
His history of end-stage renal disease, failure of peritoneal dialysis, high calcium-phosphorus product (8.1 mg/dL × 10.6 mg/dL = 85.9 mg2/dL2, reference range ≤ 55), and characteristic physical findings led to the diagnosis of calcific uremic arteriolopathy.
CALCIFIC UREMIC ARTERIOLOPATHY
Calcific uremic arteriolopathy or “calciphylaxis,” seen most often in patients with end-stage renal disease, is caused by calcium deposition in the media of the dermo-hypodermic arterioles, leading to infarction of adjacent tissue.1–3 A high calcium-phosphorus product (> 55) has been implicated in its development; however, the calcium-phosphorus product can be normal despite hyperphosphatemia, which itself may promote ectopic calcification.
Early ischemic manifestations include livedo reticularis and painful retiform purpura on the thighs and other areas of high adiposity. Lesions evolve into violaceous plaquelike subcutaneous nodules that can infarct, become necrotic, ulcerate, and become infected. Punch biopsy demonstrating arteriolar calcification, subintimal fibrosis, and thrombosis confirms the diagnosis.
Differential diagnosis
Warfarin necrosis can cause large, irregular, bloody bullae that ulcerate and turn into eschar that may resemble lesions of calcific uremic arteriolopathy. Our patient, however, had no exposure to warfarin.
Pemphigus foliaceus, an immunoglobulin G4-mediated autoimmune disorder targeted against desmoglein-1, leads to the formation of fragile blisters that easily rupture when rubbed (Nikolsky sign). Lesions evolve into scaling, crusty erosions on an erythematous base. With tender blisters and lack of mucous membrane involvement, pemphigus foliaceus shares similarities with calcific uremic arteriolopathy, but the presence of necrotic eschar surrounded by violaceous plaques in our patient made it an unlikely diagnosis.
Cryofibrinogenemia. In the right clinical scenario, ie, in a patient with vasculitis, malignancy, infection, cryoglobulinemia, or collagen diseases, cryofibrinogen-mediated cold-induced occlusive lesions may mimic calcific uremic arteriolopathy, with painful or pruritic erythema, purpura, livedo reticularis, necrosis, and ulceration.4 Our patient had no color changes with exposure to cold, nor any history of Raynaud phenomenon or joint pain, making the diagnosis of cryofibrinogenemia less likely.
Nephrogenic systemic fibrosis. Gadolinium contrast medium in magnetic resonance imaging can cause nephrogenic systemic fibrosis, characterized by erythematous papules that coalesce into brawny plaques with surrounding woody induration, which may resemble lesions of calcific uremic arteriolopathy.5 However, our patient had not been exposed to gadolinium.
Management
Management is multidisciplinary and includes the following1:
- Hemodialysis, modified to optimize calcium balance2
- Intravenous sodium thiosulfate: the exact mechanism of action remains unclear, but it is thought to play a role in chelating calcium from tissue deposits, thus decreasing pain and promoting regression of skin lesions3
- Wound care, including chemical debridement agents, negative-pressure wound therapy, and surgical debridement for infected wounds6
- Pain management with opioid analgesics.
The patient was treated with all these measures. However, he died of sudden cardiac arrest during the same admission.
A 51-year-old man with end-stage renal disease, on peritoneal dialysis for the past 4 years, presented to the emergency department with severe pain in both legs. The pain had started 2 months previously and had progressively worsened. After multiple admissions in the past for hyperkalemia and volume overload due to noncompliance, he had been advised to switch to hemodialysis.
See related article and editorial
Laboratory analysis revealed the following values:
- Serum creatinine 12.62 mg/dL (reference range 0.73–1.22)
- Blood urea nitrogen 159 mg/dL (9–24)
- Serum calcium corrected for serum albumin 8.1 mg/dL (8.4–10.0)
- Serum phosphorus 10.6 mg/dL (2.7–4.8).
His history of end-stage renal disease, failure of peritoneal dialysis, high calcium-phosphorus product (8.1 mg/dL × 10.6 mg/dL = 85.9 mg2/dL2, reference range ≤ 55), and characteristic physical findings led to the diagnosis of calcific uremic arteriolopathy.
CALCIFIC UREMIC ARTERIOLOPATHY
Calcific uremic arteriolopathy or “calciphylaxis,” seen most often in patients with end-stage renal disease, is caused by calcium deposition in the media of the dermo-hypodermic arterioles, leading to infarction of adjacent tissue.1–3 A high calcium-phosphorus product (> 55) has been implicated in its development; however, the calcium-phosphorus product can be normal despite hyperphosphatemia, which itself may promote ectopic calcification.
Early ischemic manifestations include livedo reticularis and painful retiform purpura on the thighs and other areas of high adiposity. Lesions evolve into violaceous plaquelike subcutaneous nodules that can infarct, become necrotic, ulcerate, and become infected. Punch biopsy demonstrating arteriolar calcification, subintimal fibrosis, and thrombosis confirms the diagnosis.
Differential diagnosis
Warfarin necrosis can cause large, irregular, bloody bullae that ulcerate and turn into eschar that may resemble lesions of calcific uremic arteriolopathy. Our patient, however, had no exposure to warfarin.
Pemphigus foliaceus, an immunoglobulin G4-mediated autoimmune disorder targeted against desmoglein-1, leads to the formation of fragile blisters that easily rupture when rubbed (Nikolsky sign). Lesions evolve into scaling, crusty erosions on an erythematous base. With tender blisters and lack of mucous membrane involvement, pemphigus foliaceus shares similarities with calcific uremic arteriolopathy, but the presence of necrotic eschar surrounded by violaceous plaques in our patient made it an unlikely diagnosis.
Cryofibrinogenemia. In the right clinical scenario, ie, in a patient with vasculitis, malignancy, infection, cryoglobulinemia, or collagen diseases, cryofibrinogen-mediated cold-induced occlusive lesions may mimic calcific uremic arteriolopathy, with painful or pruritic erythema, purpura, livedo reticularis, necrosis, and ulceration.4 Our patient had no color changes with exposure to cold, nor any history of Raynaud phenomenon or joint pain, making the diagnosis of cryofibrinogenemia less likely.
Nephrogenic systemic fibrosis. Gadolinium contrast medium in magnetic resonance imaging can cause nephrogenic systemic fibrosis, characterized by erythematous papules that coalesce into brawny plaques with surrounding woody induration, which may resemble lesions of calcific uremic arteriolopathy.5 However, our patient had not been exposed to gadolinium.
Management
Management is multidisciplinary and includes the following1:
- Hemodialysis, modified to optimize calcium balance2
- Intravenous sodium thiosulfate: the exact mechanism of action remains unclear, but it is thought to play a role in chelating calcium from tissue deposits, thus decreasing pain and promoting regression of skin lesions3
- Wound care, including chemical debridement agents, negative-pressure wound therapy, and surgical debridement for infected wounds6
- Pain management with opioid analgesics.
The patient was treated with all these measures. However, he died of sudden cardiac arrest during the same admission.
- Weenig RH, Sewell LD, Davis MD, McCarthy JT, Pittelkow MR. Calciphylaxis: natural history, risk factor analysis, and outcome. J Am Acad Dermatol 2007; 56(4):569–579. doi:10.1016/j.jaad.2006.08.065
- Nigwekar SU, Kroshinsky D, Nazarian RM, et al. Calciphylaxis: risk factors, diagnosis, and treatment. Am J Kidney Dis 2015; 66(1):133–146. doi:10.1053/j.ajkd.2015.01.034
- Janigan DT, Hirsch DJ, Klassen GA, MacDonald AS. Calcified subcutaneous arterioles with infarcts of the subcutis and skin (“calciphylaxis”) in chronic renal failure. Am J Kidney Dis 2000; 35(4):588–597. pmid:10739777
- Michaud M, Pourrat J. Cryofibrinogenemia. J Clin Rheumatol 2013; 19(3):142–148. doi:10.1097/RHU.0b013e318289e06e
- Galan A, Cowper SE, Bucala R. Nephrogenic systemic fibrosis (nephrogenic fibrosing dermopathy). Curr Opin Rheumatol 2006; 18(6):614–617. doi:10.1097/01.bor.0000245725.94887.8d
- Freedberg IM, Eisen AZ, Wolff K, Austen KF, Goldsmith LA, Katz SI, eds. Fitzpatrick’s Dermatology in General Medicine. 6th ed. New York, NY: McGraw-Hill Professional; 2003:558–562.
- Weenig RH, Sewell LD, Davis MD, McCarthy JT, Pittelkow MR. Calciphylaxis: natural history, risk factor analysis, and outcome. J Am Acad Dermatol 2007; 56(4):569–579. doi:10.1016/j.jaad.2006.08.065
- Nigwekar SU, Kroshinsky D, Nazarian RM, et al. Calciphylaxis: risk factors, diagnosis, and treatment. Am J Kidney Dis 2015; 66(1):133–146. doi:10.1053/j.ajkd.2015.01.034
- Janigan DT, Hirsch DJ, Klassen GA, MacDonald AS. Calcified subcutaneous arterioles with infarcts of the subcutis and skin (“calciphylaxis”) in chronic renal failure. Am J Kidney Dis 2000; 35(4):588–597. pmid:10739777
- Michaud M, Pourrat J. Cryofibrinogenemia. J Clin Rheumatol 2013; 19(3):142–148. doi:10.1097/RHU.0b013e318289e06e
- Galan A, Cowper SE, Bucala R. Nephrogenic systemic fibrosis (nephrogenic fibrosing dermopathy). Curr Opin Rheumatol 2006; 18(6):614–617. doi:10.1097/01.bor.0000245725.94887.8d
- Freedberg IM, Eisen AZ, Wolff K, Austen KF, Goldsmith LA, Katz SI, eds. Fitzpatrick’s Dermatology in General Medicine. 6th ed. New York, NY: McGraw-Hill Professional; 2003:558–562.