User login
Palmoplantar exanthema and liver dysfunction
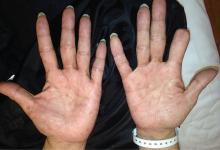
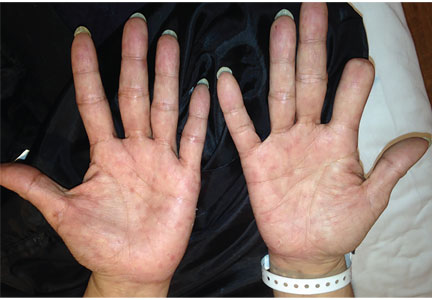
A 51-year-old man with type 2 diabetes was referred to our hospital because of liver dysfunction and nonpruritic exanthema, with papulosquamous, scaly, papular and macular lesions on his trunk and extremities, including his palms (Figure 1) and soles. Also noted were tiny grayish mucus patches on the oral mucosa. Axillary and inguinal superficial lymph nodes were palpable.
Laboratory testing revealed elevated serum levels of markers of liver disease, ie:
- Total bilirubin 9.8 mg/dL (reference range 0.2–1.3)
- Direct bilirubin 8.0 mg/dL (< 0.2)
- Aspartate aminotransferase 57 IU/L (13–35)
- Alanine aminotransferase 90 IU/L (10–54)
- Alkaline phosphatase 4,446 IU/L (36–108).
Possible causes of liver dysfunction such as legal and illicit drugs, alcohol abuse, obstructive biliary tract or liver disease, viral hepatitis, and primary biliary cirrhosis were ruled out by history, serologic testing, abdominal ultrasonography, and computed tomography.
Secondary syphilis was suspected in view of the characteristic distribution of exanthema involving the trunk and extremities, especially the palms and soles. On questioning, the patient admitted to having had unprotected sex with a female sex worker, which also raised the probability of syphilis infection.
The rapid plasma reagin test was positive at a titer of 1:16, and the Treponema pallidum agglutination test was positive at a signal-to-cutoff ratio of 27.02. Antibody testing for human immunodeficiency virus (HIV) was negative.
The patient was started on penicillin G, but 4 hours later, he developed a fever with a temperature of 100.2°F (37.9°C), which was assumed to be a Jarisch-Herxheimer reaction. The fever resolved by the next morning without further treatment.
His course was otherwise uneventful. The exanthema resolved within 3 months, and his liver function returned to normal. Five months later, the rapid plasma reagin test was repeated on an outpatient basis, and the result was normal.
SYPHILIS IS NOT A DISEASE OF THE PAST
Syphilis is caused by T pallidum and is mainly transmitted by sexual contact.1
The incidence of syphilis has substantially increased in recent years in Japan2,3 and worldwide.4 The typical patient is a young man who has sex with men, is infected with HIV, and has a history of syphilis infection.3 However, the rapid increase in syphilis infections in Japan in recent years is largely because of an increase in heterosexual transmission.3
Infectious in its early stages
Syphilis is potentially infectious in its early (primary, secondary, and early latent) stages.1,5 The secondary stage generally begins 6 to 8 weeks after the primary infection1 and presents with diverse symptoms, including arthralgia, condylomata lata, generalized lymphadenopathy, maculopapular and papulosquamous exanthema, myalgia, and pharyngitis.1
Liver dysfunction in secondary syphilis
Liver dysfunction is common in secondary syphilis, occurring in 25% to 50% of cases.5 The liver enzyme pattern in most cases is a disproportionate increase in the alkaline phosphatase level compared with modest elevations of aminotransferases and bilirubin.2,5 However, some cases may show predominant hepatocellular damage (with prominent elevations in aminotransferase levels), and others may present with severe cholestasis (with prominent elevations in alkaline phosphatase and bilirubin) or even fulminant hepatic failure.2,5
The diagnostic criteria for syphilitic hepatitis are abnormal liver enzyme levels, serologic evidence of syphilis in conjunction with acute clinical presentation of secondary syphilis, exclusion of alternative causes of liver dysfunction, and prompt recovery of liver function after antimicrobial therapy.2,5
Pathogenic mechanisms in syphilitic hepatitis include direct portal venous inoculation and immune complex-mediated disease.2 However, direct hepatotoxicity of the microorganism seems unlikely, as spirochetes are rarely detected in liver specimens.2,5
Jarisch-Herxheimer reaction
The Jarisch-Herxheimer reaction is an acute febrile illness during the first 24 hours of antimicrobial treatment.1,6 It is assumed to be due to lysis of large numbers of spirochetes, releasing lipopolysaccharides (endotoxins) that further incite the release of a range of cytokines, resulting in symptoms such as fever, chills, myalgias, headache, tachycardia, hyperventilation, vasodilation with flushing, and mild hypotension.6,7
The frequency of Jarisch-Herxheimer reaction in syphilis and other spirochetal infections has varied widely in different reports.8 It is common in primary and secondary syphilis but usually does not occur in latent syphilis.6
Consider the diagnosis
Physicians should consider secondary syphilis in patients who present with characteristic generalized reddish macules and papules with papulosquamous lesions, including on the palms and soles as in our patient, and also in patients who have had unprotected sexual contact. Syphilis is not a disease of the past.
Acknowledgment: The authors thank Dr. Joel Branch, Shonan Kamakura General Hospital, Japan, for his editorial assistance.
- Mattei PL, Beachkofsky TM, Gilson RT, Wisco OJ. Syphilis: a reemerging infection. Am Fam Physician 2012; 86(5):433–440. pmid:22963062
- Miura H, Nakano M, Ryu T, Kitamura S, Suzaki A. A case of syphilis presenting with initial syphilitic hepatitis and serological recurrence with cerebrospinal abnormality. Intern Med 2010; 49(14):1377–1381. pmid:20647651
- Nishijima T, Teruya K, Shibata S, et al. Incidence and risk factors for incident syphilis among HIV-1-infected men who have sex with men in a large urban HIV clinic in Tokyo, 2008-2015. PLoS One 2016; 11(12):e0168642. doi:10.1371/journal.pone.0168642
- US Preventive Services Task Force (USPSTF), Bibbins-Domingo K, Grossman DC, Curry SJ, et al. Screening for syphilis infection in nonpregnant adults and adolescents: US Preventive Services Task Force Recommendation Statement. JAMA 2016; 315(21):2321–2327. doi:10.1001/jama.2016.5824
- Aggarwal A, Sharma V, Vaiphei K, Duseja A, Chawla YK. An unusual cause of cholestatic hepatitis: syphilis. Dig Dis Sci 2013; 58(10):3049–3051. doi:10.1007/s10620-013-2581-5
- Belum GR, Belum VR, Chaitanya Arudra SK, Reddy BS. The Jarisch-Herxheimer reaction: revisited. Travel Med Infect Dis 2013; 11(4):231–237. doi:10.1016/j.tmaid.2013.04.001
- Nau R, Eiffert H. Modulation of release of proinflammatory bacterial compounds by antibacterials: potential impact on course of inflammation and outcome in sepsis and meningitis. Clin Microbiol Rev 2002; 15(1):95–110. pmid:11781269
- Butler T. The Jarisch-Herxheimer reaction after antibiotic treatment of spirochetal infections: a review of recent cases and our understanding of pathogenesis. Am J Trop Med Hyg 2017; 96(1):46–52. doi:10.4269/ajtmh.16-0434
A 51-year-old man with type 2 diabetes was referred to our hospital because of liver dysfunction and nonpruritic exanthema, with papulosquamous, scaly, papular and macular lesions on his trunk and extremities, including his palms (Figure 1) and soles. Also noted were tiny grayish mucus patches on the oral mucosa. Axillary and inguinal superficial lymph nodes were palpable.
Laboratory testing revealed elevated serum levels of markers of liver disease, ie:
- Total bilirubin 9.8 mg/dL (reference range 0.2–1.3)
- Direct bilirubin 8.0 mg/dL (< 0.2)
- Aspartate aminotransferase 57 IU/L (13–35)
- Alanine aminotransferase 90 IU/L (10–54)
- Alkaline phosphatase 4,446 IU/L (36–108).
Possible causes of liver dysfunction such as legal and illicit drugs, alcohol abuse, obstructive biliary tract or liver disease, viral hepatitis, and primary biliary cirrhosis were ruled out by history, serologic testing, abdominal ultrasonography, and computed tomography.
Secondary syphilis was suspected in view of the characteristic distribution of exanthema involving the trunk and extremities, especially the palms and soles. On questioning, the patient admitted to having had unprotected sex with a female sex worker, which also raised the probability of syphilis infection.
The rapid plasma reagin test was positive at a titer of 1:16, and the Treponema pallidum agglutination test was positive at a signal-to-cutoff ratio of 27.02. Antibody testing for human immunodeficiency virus (HIV) was negative.
The patient was started on penicillin G, but 4 hours later, he developed a fever with a temperature of 100.2°F (37.9°C), which was assumed to be a Jarisch-Herxheimer reaction. The fever resolved by the next morning without further treatment.
His course was otherwise uneventful. The exanthema resolved within 3 months, and his liver function returned to normal. Five months later, the rapid plasma reagin test was repeated on an outpatient basis, and the result was normal.
SYPHILIS IS NOT A DISEASE OF THE PAST
Syphilis is caused by T pallidum and is mainly transmitted by sexual contact.1
The incidence of syphilis has substantially increased in recent years in Japan2,3 and worldwide.4 The typical patient is a young man who has sex with men, is infected with HIV, and has a history of syphilis infection.3 However, the rapid increase in syphilis infections in Japan in recent years is largely because of an increase in heterosexual transmission.3
Infectious in its early stages
Syphilis is potentially infectious in its early (primary, secondary, and early latent) stages.1,5 The secondary stage generally begins 6 to 8 weeks after the primary infection1 and presents with diverse symptoms, including arthralgia, condylomata lata, generalized lymphadenopathy, maculopapular and papulosquamous exanthema, myalgia, and pharyngitis.1
Liver dysfunction in secondary syphilis
Liver dysfunction is common in secondary syphilis, occurring in 25% to 50% of cases.5 The liver enzyme pattern in most cases is a disproportionate increase in the alkaline phosphatase level compared with modest elevations of aminotransferases and bilirubin.2,5 However, some cases may show predominant hepatocellular damage (with prominent elevations in aminotransferase levels), and others may present with severe cholestasis (with prominent elevations in alkaline phosphatase and bilirubin) or even fulminant hepatic failure.2,5
The diagnostic criteria for syphilitic hepatitis are abnormal liver enzyme levels, serologic evidence of syphilis in conjunction with acute clinical presentation of secondary syphilis, exclusion of alternative causes of liver dysfunction, and prompt recovery of liver function after antimicrobial therapy.2,5
Pathogenic mechanisms in syphilitic hepatitis include direct portal venous inoculation and immune complex-mediated disease.2 However, direct hepatotoxicity of the microorganism seems unlikely, as spirochetes are rarely detected in liver specimens.2,5
Jarisch-Herxheimer reaction
The Jarisch-Herxheimer reaction is an acute febrile illness during the first 24 hours of antimicrobial treatment.1,6 It is assumed to be due to lysis of large numbers of spirochetes, releasing lipopolysaccharides (endotoxins) that further incite the release of a range of cytokines, resulting in symptoms such as fever, chills, myalgias, headache, tachycardia, hyperventilation, vasodilation with flushing, and mild hypotension.6,7
The frequency of Jarisch-Herxheimer reaction in syphilis and other spirochetal infections has varied widely in different reports.8 It is common in primary and secondary syphilis but usually does not occur in latent syphilis.6
Consider the diagnosis
Physicians should consider secondary syphilis in patients who present with characteristic generalized reddish macules and papules with papulosquamous lesions, including on the palms and soles as in our patient, and also in patients who have had unprotected sexual contact. Syphilis is not a disease of the past.
Acknowledgment: The authors thank Dr. Joel Branch, Shonan Kamakura General Hospital, Japan, for his editorial assistance.
A 51-year-old man with type 2 diabetes was referred to our hospital because of liver dysfunction and nonpruritic exanthema, with papulosquamous, scaly, papular and macular lesions on his trunk and extremities, including his palms (Figure 1) and soles. Also noted were tiny grayish mucus patches on the oral mucosa. Axillary and inguinal superficial lymph nodes were palpable.
Laboratory testing revealed elevated serum levels of markers of liver disease, ie:
- Total bilirubin 9.8 mg/dL (reference range 0.2–1.3)
- Direct bilirubin 8.0 mg/dL (< 0.2)
- Aspartate aminotransferase 57 IU/L (13–35)
- Alanine aminotransferase 90 IU/L (10–54)
- Alkaline phosphatase 4,446 IU/L (36–108).
Possible causes of liver dysfunction such as legal and illicit drugs, alcohol abuse, obstructive biliary tract or liver disease, viral hepatitis, and primary biliary cirrhosis were ruled out by history, serologic testing, abdominal ultrasonography, and computed tomography.
Secondary syphilis was suspected in view of the characteristic distribution of exanthema involving the trunk and extremities, especially the palms and soles. On questioning, the patient admitted to having had unprotected sex with a female sex worker, which also raised the probability of syphilis infection.
The rapid plasma reagin test was positive at a titer of 1:16, and the Treponema pallidum agglutination test was positive at a signal-to-cutoff ratio of 27.02. Antibody testing for human immunodeficiency virus (HIV) was negative.
The patient was started on penicillin G, but 4 hours later, he developed a fever with a temperature of 100.2°F (37.9°C), which was assumed to be a Jarisch-Herxheimer reaction. The fever resolved by the next morning without further treatment.
His course was otherwise uneventful. The exanthema resolved within 3 months, and his liver function returned to normal. Five months later, the rapid plasma reagin test was repeated on an outpatient basis, and the result was normal.
SYPHILIS IS NOT A DISEASE OF THE PAST
Syphilis is caused by T pallidum and is mainly transmitted by sexual contact.1
The incidence of syphilis has substantially increased in recent years in Japan2,3 and worldwide.4 The typical patient is a young man who has sex with men, is infected with HIV, and has a history of syphilis infection.3 However, the rapid increase in syphilis infections in Japan in recent years is largely because of an increase in heterosexual transmission.3
Infectious in its early stages
Syphilis is potentially infectious in its early (primary, secondary, and early latent) stages.1,5 The secondary stage generally begins 6 to 8 weeks after the primary infection1 and presents with diverse symptoms, including arthralgia, condylomata lata, generalized lymphadenopathy, maculopapular and papulosquamous exanthema, myalgia, and pharyngitis.1
Liver dysfunction in secondary syphilis
Liver dysfunction is common in secondary syphilis, occurring in 25% to 50% of cases.5 The liver enzyme pattern in most cases is a disproportionate increase in the alkaline phosphatase level compared with modest elevations of aminotransferases and bilirubin.2,5 However, some cases may show predominant hepatocellular damage (with prominent elevations in aminotransferase levels), and others may present with severe cholestasis (with prominent elevations in alkaline phosphatase and bilirubin) or even fulminant hepatic failure.2,5
The diagnostic criteria for syphilitic hepatitis are abnormal liver enzyme levels, serologic evidence of syphilis in conjunction with acute clinical presentation of secondary syphilis, exclusion of alternative causes of liver dysfunction, and prompt recovery of liver function after antimicrobial therapy.2,5
Pathogenic mechanisms in syphilitic hepatitis include direct portal venous inoculation and immune complex-mediated disease.2 However, direct hepatotoxicity of the microorganism seems unlikely, as spirochetes are rarely detected in liver specimens.2,5
Jarisch-Herxheimer reaction
The Jarisch-Herxheimer reaction is an acute febrile illness during the first 24 hours of antimicrobial treatment.1,6 It is assumed to be due to lysis of large numbers of spirochetes, releasing lipopolysaccharides (endotoxins) that further incite the release of a range of cytokines, resulting in symptoms such as fever, chills, myalgias, headache, tachycardia, hyperventilation, vasodilation with flushing, and mild hypotension.6,7
The frequency of Jarisch-Herxheimer reaction in syphilis and other spirochetal infections has varied widely in different reports.8 It is common in primary and secondary syphilis but usually does not occur in latent syphilis.6
Consider the diagnosis
Physicians should consider secondary syphilis in patients who present with characteristic generalized reddish macules and papules with papulosquamous lesions, including on the palms and soles as in our patient, and also in patients who have had unprotected sexual contact. Syphilis is not a disease of the past.
Acknowledgment: The authors thank Dr. Joel Branch, Shonan Kamakura General Hospital, Japan, for his editorial assistance.
- Mattei PL, Beachkofsky TM, Gilson RT, Wisco OJ. Syphilis: a reemerging infection. Am Fam Physician 2012; 86(5):433–440. pmid:22963062
- Miura H, Nakano M, Ryu T, Kitamura S, Suzaki A. A case of syphilis presenting with initial syphilitic hepatitis and serological recurrence with cerebrospinal abnormality. Intern Med 2010; 49(14):1377–1381. pmid:20647651
- Nishijima T, Teruya K, Shibata S, et al. Incidence and risk factors for incident syphilis among HIV-1-infected men who have sex with men in a large urban HIV clinic in Tokyo, 2008-2015. PLoS One 2016; 11(12):e0168642. doi:10.1371/journal.pone.0168642
- US Preventive Services Task Force (USPSTF), Bibbins-Domingo K, Grossman DC, Curry SJ, et al. Screening for syphilis infection in nonpregnant adults and adolescents: US Preventive Services Task Force Recommendation Statement. JAMA 2016; 315(21):2321–2327. doi:10.1001/jama.2016.5824
- Aggarwal A, Sharma V, Vaiphei K, Duseja A, Chawla YK. An unusual cause of cholestatic hepatitis: syphilis. Dig Dis Sci 2013; 58(10):3049–3051. doi:10.1007/s10620-013-2581-5
- Belum GR, Belum VR, Chaitanya Arudra SK, Reddy BS. The Jarisch-Herxheimer reaction: revisited. Travel Med Infect Dis 2013; 11(4):231–237. doi:10.1016/j.tmaid.2013.04.001
- Nau R, Eiffert H. Modulation of release of proinflammatory bacterial compounds by antibacterials: potential impact on course of inflammation and outcome in sepsis and meningitis. Clin Microbiol Rev 2002; 15(1):95–110. pmid:11781269
- Butler T. The Jarisch-Herxheimer reaction after antibiotic treatment of spirochetal infections: a review of recent cases and our understanding of pathogenesis. Am J Trop Med Hyg 2017; 96(1):46–52. doi:10.4269/ajtmh.16-0434
- Mattei PL, Beachkofsky TM, Gilson RT, Wisco OJ. Syphilis: a reemerging infection. Am Fam Physician 2012; 86(5):433–440. pmid:22963062
- Miura H, Nakano M, Ryu T, Kitamura S, Suzaki A. A case of syphilis presenting with initial syphilitic hepatitis and serological recurrence with cerebrospinal abnormality. Intern Med 2010; 49(14):1377–1381. pmid:20647651
- Nishijima T, Teruya K, Shibata S, et al. Incidence and risk factors for incident syphilis among HIV-1-infected men who have sex with men in a large urban HIV clinic in Tokyo, 2008-2015. PLoS One 2016; 11(12):e0168642. doi:10.1371/journal.pone.0168642
- US Preventive Services Task Force (USPSTF), Bibbins-Domingo K, Grossman DC, Curry SJ, et al. Screening for syphilis infection in nonpregnant adults and adolescents: US Preventive Services Task Force Recommendation Statement. JAMA 2016; 315(21):2321–2327. doi:10.1001/jama.2016.5824
- Aggarwal A, Sharma V, Vaiphei K, Duseja A, Chawla YK. An unusual cause of cholestatic hepatitis: syphilis. Dig Dis Sci 2013; 58(10):3049–3051. doi:10.1007/s10620-013-2581-5
- Belum GR, Belum VR, Chaitanya Arudra SK, Reddy BS. The Jarisch-Herxheimer reaction: revisited. Travel Med Infect Dis 2013; 11(4):231–237. doi:10.1016/j.tmaid.2013.04.001
- Nau R, Eiffert H. Modulation of release of proinflammatory bacterial compounds by antibacterials: potential impact on course of inflammation and outcome in sepsis and meningitis. Clin Microbiol Rev 2002; 15(1):95–110. pmid:11781269
- Butler T. The Jarisch-Herxheimer reaction after antibiotic treatment of spirochetal infections: a review of recent cases and our understanding of pathogenesis. Am J Trop Med Hyg 2017; 96(1):46–52. doi:10.4269/ajtmh.16-0434
Angular cheilitis induced by iron deficiency anemia
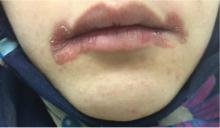
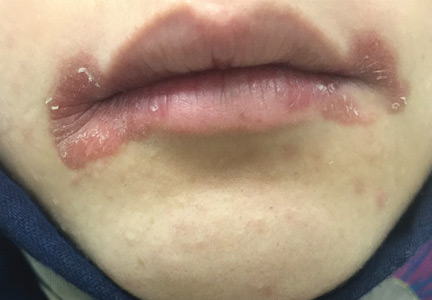
A 20-year-old woman had a 4-month history of painful red erosions around the mouth. She had no dysphagia or fatigue and no history of diarrhea, gluten intolerance, or diabetes mellitus. An antifungal-antibacterial ointment prescribed by her dentist had provided no relief.
- Hemoglobin 8.0 g/dL (reference range for females 12.3–15.3)
- Mean corpuscular volume 62 fL (80–100)
- Serum ferritin 1.3 ng/mL (15–200)
- Reticulocyte count 0.86% (0.5–1.5)
- White blood cell count 9.8 × 109/L (4.5–11.0)
- Platelet count 450 × 109/L (150–400).
Vitamin B12 and folate levels were normal, and tests for antitissue transglutaminase and antinuclear antibodies were negative.
Based on these results, the diagnosis was angular cheilitis from iron deficiency anemia. Treatment with oral ferrous gluconate 300 mg twice daily cleared the cheilitis, and after 4 weeks of this treatment, the hemoglobin level increased to 9.8 g/dL, the serum ferritin increased to 7 ng/mL, and the reticulocyte count increased to 2.6%. She was advised to continue taking oral iron tablets for another 3 months until the hemoglobin level reached 12.0 g/dL.
During 2 years of follow-up, she had no recurrence of angular cheilitis, and her hemoglobin and serum ferritin levels remained normal. Ferrous gluconate was her only medication from the time of her diagnosis.
A BROAD DIFFERENTIAL DIAGNOSIS
Angular cheilitis (perlèche) is an inflammatory condition characterized by erosive inflammation at one or both angles of the mouth. It typically presents as erythema, scaling, fissuring, and ulceration. A wide variety of factors, including nutritional deficiencies, local and systemic factors, and drug side effects, may produce cheilitis.1,2
Nutritional deficiencies account for 25% of all cases of angular cheilitis3 and include iron deficiency and deficiencies of the B vitamins riboflavin (B2), niacin (B3), pyridoxine (B6), and cyanocobalamin (B12).1
Local causes include infection with Candida albicans or Staphylococcus aureus and allergic contact dermatitis. Common causes of allergic contact dermatitis include lipstick, toothpaste, mouthwash, cosmetics, sunscreen, fragrance, metals such as nickel, and dental appliances.1
Systemic diseases associated with angular cheilitis include xerostomia, inflammatory bowel disease, Sjögren syndrome, glucagonoma, and human immunodeficiency virus.1
Drugs that cause angular cheilitis include isotretinoin, sorafenib (antineoplastic kinase inhibitor), and ointments or creams such as neomycin sulfate–polymyxin B sulfate, bacitracin, idoxuridine, and steroids.1,4
Conditions that mimic angular cheilitis include herpes simplex type 1 (herpes labialis) and actinic cheilitis. Herpes labialis, characterized by burning sensation, itching, or paresthesia, usually precedes a recurrence of vesicles that eventually ulcerate or form a crust and heal without a crust. Herpes labialis often recurs, affecting the vermilion border and lasting approximately 1 week.
Actinic cheilitis, a premalignant condition that commonly involves the lower lip with sparing of the corners of the mouth, is caused by excessive sun exposure. Patients often have persistent dryness and cracking of the lips.
In our patient, angular cheilitis was the main clinical manifestation of iron deficiency anemia, highlighting the importance of looking for iron deficiency in affected patients without a more obvious cause.
- Park KK, Brodell RT, Helms SE. Angular cheilitis, part 2: nutritional, systemic, and drug-related causes and treatment. Cutis 2011; 88(1):27–32. pmid:21877503
- Park KK, Brodell RT, Helms SE. Angular cheilitis, part 1: local etiologies. Cutis 2011; 87(6):289–295. pmid:21838086
- Konstantinidis AB, Hatziotis JH. Angular cheilosis: an analysis of 156 cases. J Oral Med 1984; 39(4):199–206. pmid:6594458
- Yang CH, Lin WC, Chuang CK, et al. Hand-foot skin reaction in patients treated with sorafenib: a clinicopathological study of cutaneous manifestations due to multitargeted kinase inhibitor therapy. Br J Dermatol 2008; 158(3):592–596. doi:10.1111/j.1365-2133.2007.08357.x
A 20-year-old woman had a 4-month history of painful red erosions around the mouth. She had no dysphagia or fatigue and no history of diarrhea, gluten intolerance, or diabetes mellitus. An antifungal-antibacterial ointment prescribed by her dentist had provided no relief.
- Hemoglobin 8.0 g/dL (reference range for females 12.3–15.3)
- Mean corpuscular volume 62 fL (80–100)
- Serum ferritin 1.3 ng/mL (15–200)
- Reticulocyte count 0.86% (0.5–1.5)
- White blood cell count 9.8 × 109/L (4.5–11.0)
- Platelet count 450 × 109/L (150–400).
Vitamin B12 and folate levels were normal, and tests for antitissue transglutaminase and antinuclear antibodies were negative.
Based on these results, the diagnosis was angular cheilitis from iron deficiency anemia. Treatment with oral ferrous gluconate 300 mg twice daily cleared the cheilitis, and after 4 weeks of this treatment, the hemoglobin level increased to 9.8 g/dL, the serum ferritin increased to 7 ng/mL, and the reticulocyte count increased to 2.6%. She was advised to continue taking oral iron tablets for another 3 months until the hemoglobin level reached 12.0 g/dL.
During 2 years of follow-up, she had no recurrence of angular cheilitis, and her hemoglobin and serum ferritin levels remained normal. Ferrous gluconate was her only medication from the time of her diagnosis.
A BROAD DIFFERENTIAL DIAGNOSIS
Angular cheilitis (perlèche) is an inflammatory condition characterized by erosive inflammation at one or both angles of the mouth. It typically presents as erythema, scaling, fissuring, and ulceration. A wide variety of factors, including nutritional deficiencies, local and systemic factors, and drug side effects, may produce cheilitis.1,2
Nutritional deficiencies account for 25% of all cases of angular cheilitis3 and include iron deficiency and deficiencies of the B vitamins riboflavin (B2), niacin (B3), pyridoxine (B6), and cyanocobalamin (B12).1
Local causes include infection with Candida albicans or Staphylococcus aureus and allergic contact dermatitis. Common causes of allergic contact dermatitis include lipstick, toothpaste, mouthwash, cosmetics, sunscreen, fragrance, metals such as nickel, and dental appliances.1
Systemic diseases associated with angular cheilitis include xerostomia, inflammatory bowel disease, Sjögren syndrome, glucagonoma, and human immunodeficiency virus.1
Drugs that cause angular cheilitis include isotretinoin, sorafenib (antineoplastic kinase inhibitor), and ointments or creams such as neomycin sulfate–polymyxin B sulfate, bacitracin, idoxuridine, and steroids.1,4
Conditions that mimic angular cheilitis include herpes simplex type 1 (herpes labialis) and actinic cheilitis. Herpes labialis, characterized by burning sensation, itching, or paresthesia, usually precedes a recurrence of vesicles that eventually ulcerate or form a crust and heal without a crust. Herpes labialis often recurs, affecting the vermilion border and lasting approximately 1 week.
Actinic cheilitis, a premalignant condition that commonly involves the lower lip with sparing of the corners of the mouth, is caused by excessive sun exposure. Patients often have persistent dryness and cracking of the lips.
In our patient, angular cheilitis was the main clinical manifestation of iron deficiency anemia, highlighting the importance of looking for iron deficiency in affected patients without a more obvious cause.
A 20-year-old woman had a 4-month history of painful red erosions around the mouth. She had no dysphagia or fatigue and no history of diarrhea, gluten intolerance, or diabetes mellitus. An antifungal-antibacterial ointment prescribed by her dentist had provided no relief.
- Hemoglobin 8.0 g/dL (reference range for females 12.3–15.3)
- Mean corpuscular volume 62 fL (80–100)
- Serum ferritin 1.3 ng/mL (15–200)
- Reticulocyte count 0.86% (0.5–1.5)
- White blood cell count 9.8 × 109/L (4.5–11.0)
- Platelet count 450 × 109/L (150–400).
Vitamin B12 and folate levels were normal, and tests for antitissue transglutaminase and antinuclear antibodies were negative.
Based on these results, the diagnosis was angular cheilitis from iron deficiency anemia. Treatment with oral ferrous gluconate 300 mg twice daily cleared the cheilitis, and after 4 weeks of this treatment, the hemoglobin level increased to 9.8 g/dL, the serum ferritin increased to 7 ng/mL, and the reticulocyte count increased to 2.6%. She was advised to continue taking oral iron tablets for another 3 months until the hemoglobin level reached 12.0 g/dL.
During 2 years of follow-up, she had no recurrence of angular cheilitis, and her hemoglobin and serum ferritin levels remained normal. Ferrous gluconate was her only medication from the time of her diagnosis.
A BROAD DIFFERENTIAL DIAGNOSIS
Angular cheilitis (perlèche) is an inflammatory condition characterized by erosive inflammation at one or both angles of the mouth. It typically presents as erythema, scaling, fissuring, and ulceration. A wide variety of factors, including nutritional deficiencies, local and systemic factors, and drug side effects, may produce cheilitis.1,2
Nutritional deficiencies account for 25% of all cases of angular cheilitis3 and include iron deficiency and deficiencies of the B vitamins riboflavin (B2), niacin (B3), pyridoxine (B6), and cyanocobalamin (B12).1
Local causes include infection with Candida albicans or Staphylococcus aureus and allergic contact dermatitis. Common causes of allergic contact dermatitis include lipstick, toothpaste, mouthwash, cosmetics, sunscreen, fragrance, metals such as nickel, and dental appliances.1
Systemic diseases associated with angular cheilitis include xerostomia, inflammatory bowel disease, Sjögren syndrome, glucagonoma, and human immunodeficiency virus.1
Drugs that cause angular cheilitis include isotretinoin, sorafenib (antineoplastic kinase inhibitor), and ointments or creams such as neomycin sulfate–polymyxin B sulfate, bacitracin, idoxuridine, and steroids.1,4
Conditions that mimic angular cheilitis include herpes simplex type 1 (herpes labialis) and actinic cheilitis. Herpes labialis, characterized by burning sensation, itching, or paresthesia, usually precedes a recurrence of vesicles that eventually ulcerate or form a crust and heal without a crust. Herpes labialis often recurs, affecting the vermilion border and lasting approximately 1 week.
Actinic cheilitis, a premalignant condition that commonly involves the lower lip with sparing of the corners of the mouth, is caused by excessive sun exposure. Patients often have persistent dryness and cracking of the lips.
In our patient, angular cheilitis was the main clinical manifestation of iron deficiency anemia, highlighting the importance of looking for iron deficiency in affected patients without a more obvious cause.
- Park KK, Brodell RT, Helms SE. Angular cheilitis, part 2: nutritional, systemic, and drug-related causes and treatment. Cutis 2011; 88(1):27–32. pmid:21877503
- Park KK, Brodell RT, Helms SE. Angular cheilitis, part 1: local etiologies. Cutis 2011; 87(6):289–295. pmid:21838086
- Konstantinidis AB, Hatziotis JH. Angular cheilosis: an analysis of 156 cases. J Oral Med 1984; 39(4):199–206. pmid:6594458
- Yang CH, Lin WC, Chuang CK, et al. Hand-foot skin reaction in patients treated with sorafenib: a clinicopathological study of cutaneous manifestations due to multitargeted kinase inhibitor therapy. Br J Dermatol 2008; 158(3):592–596. doi:10.1111/j.1365-2133.2007.08357.x
- Park KK, Brodell RT, Helms SE. Angular cheilitis, part 2: nutritional, systemic, and drug-related causes and treatment. Cutis 2011; 88(1):27–32. pmid:21877503
- Park KK, Brodell RT, Helms SE. Angular cheilitis, part 1: local etiologies. Cutis 2011; 87(6):289–295. pmid:21838086
- Konstantinidis AB, Hatziotis JH. Angular cheilosis: an analysis of 156 cases. J Oral Med 1984; 39(4):199–206. pmid:6594458
- Yang CH, Lin WC, Chuang CK, et al. Hand-foot skin reaction in patients treated with sorafenib: a clinicopathological study of cutaneous manifestations due to multitargeted kinase inhibitor therapy. Br J Dermatol 2008; 158(3):592–596. doi:10.1111/j.1365-2133.2007.08357.x
What is your diagnosis?
Laboratory work revealed a normal CBC and differential, an elevated C-reactive protein (CRP) and sedimentation rate (ESR), negative antistreptolysin O (ASO) titers, negative pregnancy test, a normal urinalysis, and negative blood, throat, and urine cultures. A chest x-ray also was negative as well as angiotensin-converting enzyme (ACE) levels. Tuberculosis interferon-gamma release essay was negative.
The patient was diagnosed with erythema nodosum (EN), based on physical exam and history of the lesions. In her particular case, infectious causes including streptococcus infection, tuberculosis, and coccidioidomycosis were ruled out. There were no x-ray findings that suggested sarcoidosis and her ACE level was within normal limits. The pregnancy test also was negative. Given her recent start on OCs, this was thought to be the cause of the lesions.
She was treated with elevation, compression stockings, and NSAIDs and discontinuation of OCs. The lesions resolved after 6 weeks leaving bruiselike patches (erythema contusiformis).
EN is a delayed-type hypersensitivity reaction, causing inflammation on the fat (panniculitis) most commonly on the shins, but it can also occur on the arms, face, neck, and thighs. It is the most common type of panniculitis and is usually seen more often in women from the second to fourth decade of life. Erythematous tender nodules in crops commonly located on the shins are the characteristic physical finding. Systemic symptoms can occur including fever, malaise, and joint pain. The lesions usually last up to 6-8 weeks and may leave bruiselike patches or postinflammatory hyperpigmentation that can take months to improve.1
The diagnosis of EN usually is made by physical examination and natural history. In unusual severe cases or lesions in atypical locations, a skin biopsy is indicated. Histologic examination of one of the lesions reveals a septal panniculitis without vasculitis. Miescher’s radial granulomas (grouped macrophages around neutrophils or septa-like spaces) often are present and are a characteristic feature of EN.
EN can be triggered by different types of infections such as streptococcus, mycoplasma, tuberculosis, or bacterial gastroenteritis; medications such as OCs, sulfonamides, iodides, penicillin, or bromides; medical conditions that include inflammatory bowel disease, pregnancy, or sarcoidosis; or neutrophilic dermatosis and malignancy such as leukemia and Hodgkin disease.2,3 A third of the cases are idiopathic. In children, streptococcal infections are responsible for most cases of EN.4
Recommended work-up to investigate possible triggers includes a CBC with differential, sedimentation rate, CRP, ASO titers or anti-DNase B titers, tuberculin skin test or interferon-gamma TB test and a chest X ray. If there are any other symptoms, physical signs, or risk factors are present for the other not so common causes, further ancillary testing may be warranted.
Erythematous nodules and papules on the shin in children are commonly caused by arthropod bites also known as papular urticaria. These lesions are pruritic rather than tender and usually respond to topical corticosteroids and oral antihistamines. Subcutaneous bacterial, fungal, or atypical mycobacterial infections can present with tender nodules that can ulcerate and drain on the shins, feet, or any other body part. These patients may have a history of immunodeficiency and usually systemic symptoms of infection are present. Cutaneous polyarteritis nodosa (PAN) also can present with tender nodules on the legs but these lesions usually necrose and ulcerate and may be associated with livedo racemosa, a transient or persistent, blotchy, reddish-blue to purple, netlike cyanotic pattern. On pathology, PAN presents with necrotizing medium vessel vasculitis. Malignant nodules also can occur on the shin. Pathology will show atypical cells. Other forms of panniculitis, such as erythema induratum and pancreatic panniculitis, can present with tender nodules but these lesions usually occur on the calves and ulcerate.
Management of EN starts with treating the underlying infection or stopping the causative medication. Initial measures include bed rest, leg elevation, compression bandages, and NSAIDs. Potassium iodide is a very effective therapy as it may control the symptoms within 24 hours. When there is no response to the above, or the patient has severe symptoms, a short course of systemic glucocorticoids can be started. Other medications for recalcitrant or recurrent lesions include colchicine, dapsone, or hydroxychloroquine.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego.
References
1. Panniculitis, in “Dermatology,” 3rd ed. (Philadelphia: Elsevier Saunders, 2012, p. 1641).
2. Arthritis Rheum. 2000 Mar;43(3):584-92.
3. J Clin Oncol. 2007 Sep 1;25(25):4011-2.
4. Turk J Pediatr. 2014 Mar-Apr;56(2):144-9.
Laboratory work revealed a normal CBC and differential, an elevated C-reactive protein (CRP) and sedimentation rate (ESR), negative antistreptolysin O (ASO) titers, negative pregnancy test, a normal urinalysis, and negative blood, throat, and urine cultures. A chest x-ray also was negative as well as angiotensin-converting enzyme (ACE) levels. Tuberculosis interferon-gamma release essay was negative.
The patient was diagnosed with erythema nodosum (EN), based on physical exam and history of the lesions. In her particular case, infectious causes including streptococcus infection, tuberculosis, and coccidioidomycosis were ruled out. There were no x-ray findings that suggested sarcoidosis and her ACE level was within normal limits. The pregnancy test also was negative. Given her recent start on OCs, this was thought to be the cause of the lesions.
She was treated with elevation, compression stockings, and NSAIDs and discontinuation of OCs. The lesions resolved after 6 weeks leaving bruiselike patches (erythema contusiformis).
EN is a delayed-type hypersensitivity reaction, causing inflammation on the fat (panniculitis) most commonly on the shins, but it can also occur on the arms, face, neck, and thighs. It is the most common type of panniculitis and is usually seen more often in women from the second to fourth decade of life. Erythematous tender nodules in crops commonly located on the shins are the characteristic physical finding. Systemic symptoms can occur including fever, malaise, and joint pain. The lesions usually last up to 6-8 weeks and may leave bruiselike patches or postinflammatory hyperpigmentation that can take months to improve.1
The diagnosis of EN usually is made by physical examination and natural history. In unusual severe cases or lesions in atypical locations, a skin biopsy is indicated. Histologic examination of one of the lesions reveals a septal panniculitis without vasculitis. Miescher’s radial granulomas (grouped macrophages around neutrophils or septa-like spaces) often are present and are a characteristic feature of EN.
EN can be triggered by different types of infections such as streptococcus, mycoplasma, tuberculosis, or bacterial gastroenteritis; medications such as OCs, sulfonamides, iodides, penicillin, or bromides; medical conditions that include inflammatory bowel disease, pregnancy, or sarcoidosis; or neutrophilic dermatosis and malignancy such as leukemia and Hodgkin disease.2,3 A third of the cases are idiopathic. In children, streptococcal infections are responsible for most cases of EN.4
Recommended work-up to investigate possible triggers includes a CBC with differential, sedimentation rate, CRP, ASO titers or anti-DNase B titers, tuberculin skin test or interferon-gamma TB test and a chest X ray. If there are any other symptoms, physical signs, or risk factors are present for the other not so common causes, further ancillary testing may be warranted.
Erythematous nodules and papules on the shin in children are commonly caused by arthropod bites also known as papular urticaria. These lesions are pruritic rather than tender and usually respond to topical corticosteroids and oral antihistamines. Subcutaneous bacterial, fungal, or atypical mycobacterial infections can present with tender nodules that can ulcerate and drain on the shins, feet, or any other body part. These patients may have a history of immunodeficiency and usually systemic symptoms of infection are present. Cutaneous polyarteritis nodosa (PAN) also can present with tender nodules on the legs but these lesions usually necrose and ulcerate and may be associated with livedo racemosa, a transient or persistent, blotchy, reddish-blue to purple, netlike cyanotic pattern. On pathology, PAN presents with necrotizing medium vessel vasculitis. Malignant nodules also can occur on the shin. Pathology will show atypical cells. Other forms of panniculitis, such as erythema induratum and pancreatic panniculitis, can present with tender nodules but these lesions usually occur on the calves and ulcerate.
Management of EN starts with treating the underlying infection or stopping the causative medication. Initial measures include bed rest, leg elevation, compression bandages, and NSAIDs. Potassium iodide is a very effective therapy as it may control the symptoms within 24 hours. When there is no response to the above, or the patient has severe symptoms, a short course of systemic glucocorticoids can be started. Other medications for recalcitrant or recurrent lesions include colchicine, dapsone, or hydroxychloroquine.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego.
References
1. Panniculitis, in “Dermatology,” 3rd ed. (Philadelphia: Elsevier Saunders, 2012, p. 1641).
2. Arthritis Rheum. 2000 Mar;43(3):584-92.
3. J Clin Oncol. 2007 Sep 1;25(25):4011-2.
4. Turk J Pediatr. 2014 Mar-Apr;56(2):144-9.
Laboratory work revealed a normal CBC and differential, an elevated C-reactive protein (CRP) and sedimentation rate (ESR), negative antistreptolysin O (ASO) titers, negative pregnancy test, a normal urinalysis, and negative blood, throat, and urine cultures. A chest x-ray also was negative as well as angiotensin-converting enzyme (ACE) levels. Tuberculosis interferon-gamma release essay was negative.
The patient was diagnosed with erythema nodosum (EN), based on physical exam and history of the lesions. In her particular case, infectious causes including streptococcus infection, tuberculosis, and coccidioidomycosis were ruled out. There were no x-ray findings that suggested sarcoidosis and her ACE level was within normal limits. The pregnancy test also was negative. Given her recent start on OCs, this was thought to be the cause of the lesions.
She was treated with elevation, compression stockings, and NSAIDs and discontinuation of OCs. The lesions resolved after 6 weeks leaving bruiselike patches (erythema contusiformis).
EN is a delayed-type hypersensitivity reaction, causing inflammation on the fat (panniculitis) most commonly on the shins, but it can also occur on the arms, face, neck, and thighs. It is the most common type of panniculitis and is usually seen more often in women from the second to fourth decade of life. Erythematous tender nodules in crops commonly located on the shins are the characteristic physical finding. Systemic symptoms can occur including fever, malaise, and joint pain. The lesions usually last up to 6-8 weeks and may leave bruiselike patches or postinflammatory hyperpigmentation that can take months to improve.1
The diagnosis of EN usually is made by physical examination and natural history. In unusual severe cases or lesions in atypical locations, a skin biopsy is indicated. Histologic examination of one of the lesions reveals a septal panniculitis without vasculitis. Miescher’s radial granulomas (grouped macrophages around neutrophils or septa-like spaces) often are present and are a characteristic feature of EN.
EN can be triggered by different types of infections such as streptococcus, mycoplasma, tuberculosis, or bacterial gastroenteritis; medications such as OCs, sulfonamides, iodides, penicillin, or bromides; medical conditions that include inflammatory bowel disease, pregnancy, or sarcoidosis; or neutrophilic dermatosis and malignancy such as leukemia and Hodgkin disease.2,3 A third of the cases are idiopathic. In children, streptococcal infections are responsible for most cases of EN.4
Recommended work-up to investigate possible triggers includes a CBC with differential, sedimentation rate, CRP, ASO titers or anti-DNase B titers, tuberculin skin test or interferon-gamma TB test and a chest X ray. If there are any other symptoms, physical signs, or risk factors are present for the other not so common causes, further ancillary testing may be warranted.
Erythematous nodules and papules on the shin in children are commonly caused by arthropod bites also known as papular urticaria. These lesions are pruritic rather than tender and usually respond to topical corticosteroids and oral antihistamines. Subcutaneous bacterial, fungal, or atypical mycobacterial infections can present with tender nodules that can ulcerate and drain on the shins, feet, or any other body part. These patients may have a history of immunodeficiency and usually systemic symptoms of infection are present. Cutaneous polyarteritis nodosa (PAN) also can present with tender nodules on the legs but these lesions usually necrose and ulcerate and may be associated with livedo racemosa, a transient or persistent, blotchy, reddish-blue to purple, netlike cyanotic pattern. On pathology, PAN presents with necrotizing medium vessel vasculitis. Malignant nodules also can occur on the shin. Pathology will show atypical cells. Other forms of panniculitis, such as erythema induratum and pancreatic panniculitis, can present with tender nodules but these lesions usually occur on the calves and ulcerate.
Management of EN starts with treating the underlying infection or stopping the causative medication. Initial measures include bed rest, leg elevation, compression bandages, and NSAIDs. Potassium iodide is a very effective therapy as it may control the symptoms within 24 hours. When there is no response to the above, or the patient has severe symptoms, a short course of systemic glucocorticoids can be started. Other medications for recalcitrant or recurrent lesions include colchicine, dapsone, or hydroxychloroquine.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego.
References
1. Panniculitis, in “Dermatology,” 3rd ed. (Philadelphia: Elsevier Saunders, 2012, p. 1641).
2. Arthritis Rheum. 2000 Mar;43(3):584-92.
3. J Clin Oncol. 2007 Sep 1;25(25):4011-2.
4. Turk J Pediatr. 2014 Mar-Apr;56(2):144-9.
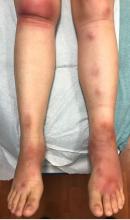
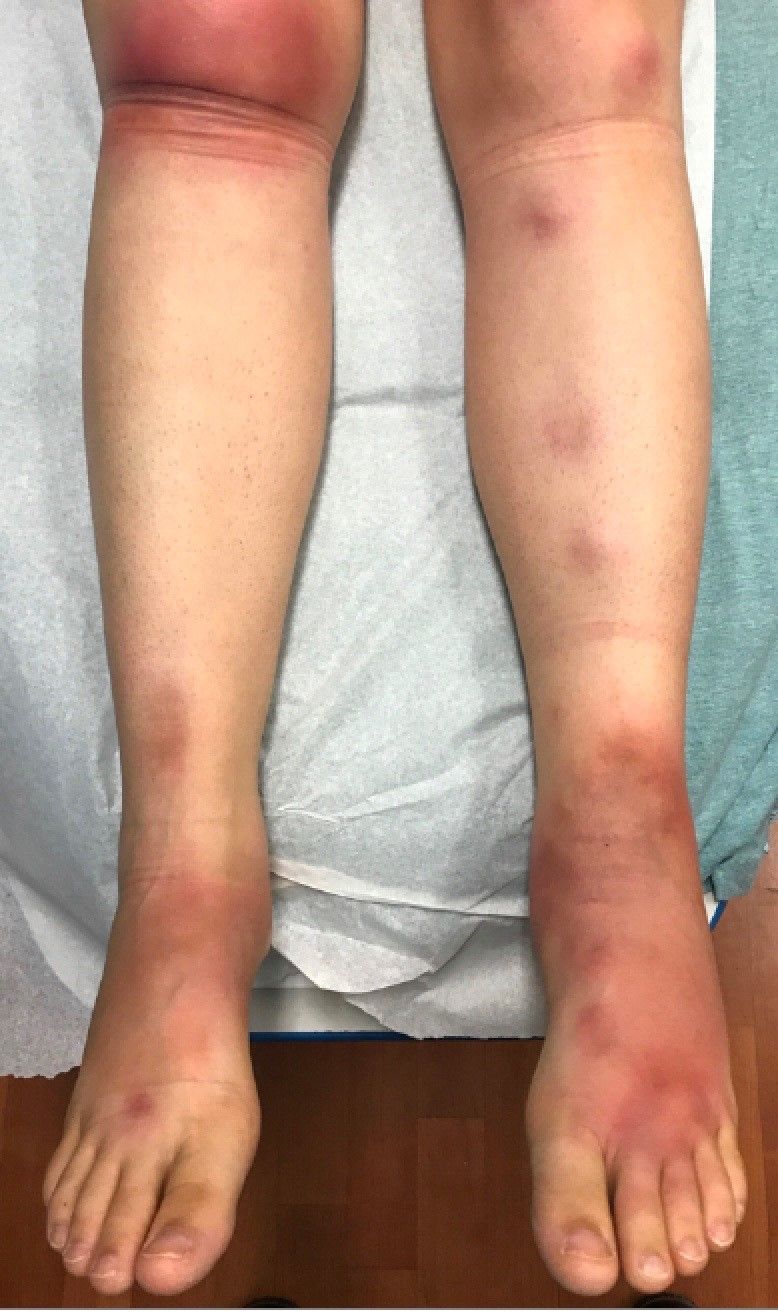
A 16-year-old female came to the dermatology clinic for acne follow-up. She reported some improvement on her acne since she started taking OCs. She also had been using benzoyl peroxide and tretinoin on her face. In addition to the acne, she also wanted us to check some tender bumps she had been getting on her shins after she came back from a camping trip. Initially she thought they were bug bites, but the lesions were getting larger, more tender, and not improving with diphenhydramine.
The physical exam did not reveal acute distress. She was afebrile. On skin examination, she had comedones, papules and scars on her face, chest, and back. On her shins there were several erythematous tender nodules and plaques. There was no edema on her legs and pulses were present.
Respect is key when treating dermatologic conditions in transgender youth
LAKE TAHOE, CALIF. – The way Stanley Vance Jr., MD, sees it, the No. 1 priority in the care of transgender youth is respecting their gender identity.
“This can really help with rapport and also help them continue to engage with your care,” he said at the annual meeting of the Society for Pediatric Dermatology.
One of the first steps is to establish the patient’s chosen name and pronouns. “Ask, use, and be consistent,” said Dr. Vance, an adolescent medicine specialist at the University of California, San Francisco. “Taking it to another level, you can implement system-level tools to ensure that all of your staff consistently use the chosen name and pronouns. and working with the IT department to have a place in our electronic medical record to put the chosen name and preferred pronouns.”
In a study published in the Journal of Adolescent Health, researchers found that the use of chosen names and pronouns for transgender use was associated with reduced depressive symptoms, suicidal ideation, and suicidal behavior among transgender youth.
Dr. Vance, who also holds a staff position at the UCSF Child and Adolescent Clinic, went on to discuss dermatologic considerations for gender diverse youth. In transgender females, estrogens can reduce the quantity and density of body and facial hair, “but it doesn’t necessarily get rid of the hair, so we may refer to dermatology for hair removal or hair reduction. There can also be a decrease in sebum production, which can lead to dry skin for those who are at risk.”
Transgender females often seek laser hair removal or electrolysis to aid in “blendability,” or how they perceive as being female or feminine. “We know that this can help in psychosocial outcomes for these young people,” Dr. Vance said. “Another reason why hair reduction and removal may be important is preoperatively for vaginoplasty.”
In transgender males, testosterone increases male pattern hair growth and can increase male pattern hair loss. “Minoxidil does not interact with gender-affirming hormone treatment. If finasteride needs to be considered, it may interfere with the development of secondary sex characteristics.” Testosterone also increases sebum production and can increase acne, particularly in the first 6 months to 1 year after initiation, and with increased titration. “Some transmasculine youth may need oral isotretinoin, as stopping testosterone can be psychologically damaging,” Dr. Vance said.
“Unfortunately, the iPLEDGE program requirements can be perceived as gender nonaffirming, because patients must register by the sex assigned to them at birth, they must take pregnancy tests, and there can be provider assumptions about sexuality which does not equate with gender identity.”
He recommended having “open and honest” conversations with patients about the requirements and limitations of dispensing oral isotretinoin. “Assure the patient that you will be respectful and affirming of their gender identity while they’re in your office,” Dr. Vance advised. “If the patient has a mental health provider, you can strategize with them to reduce gender dysphoria around this process. Finally, advocating to change the system can not only be helpful for the patient in front of you, but for other patients who are in the same situation.”
He concluded his presentation by describing transgender youth as “some of the most resilient young people I’ve had the pleasure of working with.
“I think that we can all work to make sure they feel supported in who they are,” he said.
Dr. Vance reported having no relevant financial disclosures.
[email protected]
LAKE TAHOE, CALIF. – The way Stanley Vance Jr., MD, sees it, the No. 1 priority in the care of transgender youth is respecting their gender identity.
“This can really help with rapport and also help them continue to engage with your care,” he said at the annual meeting of the Society for Pediatric Dermatology.
One of the first steps is to establish the patient’s chosen name and pronouns. “Ask, use, and be consistent,” said Dr. Vance, an adolescent medicine specialist at the University of California, San Francisco. “Taking it to another level, you can implement system-level tools to ensure that all of your staff consistently use the chosen name and pronouns. and working with the IT department to have a place in our electronic medical record to put the chosen name and preferred pronouns.”
In a study published in the Journal of Adolescent Health, researchers found that the use of chosen names and pronouns for transgender use was associated with reduced depressive symptoms, suicidal ideation, and suicidal behavior among transgender youth.
Dr. Vance, who also holds a staff position at the UCSF Child and Adolescent Clinic, went on to discuss dermatologic considerations for gender diverse youth. In transgender females, estrogens can reduce the quantity and density of body and facial hair, “but it doesn’t necessarily get rid of the hair, so we may refer to dermatology for hair removal or hair reduction. There can also be a decrease in sebum production, which can lead to dry skin for those who are at risk.”
Transgender females often seek laser hair removal or electrolysis to aid in “blendability,” or how they perceive as being female or feminine. “We know that this can help in psychosocial outcomes for these young people,” Dr. Vance said. “Another reason why hair reduction and removal may be important is preoperatively for vaginoplasty.”
In transgender males, testosterone increases male pattern hair growth and can increase male pattern hair loss. “Minoxidil does not interact with gender-affirming hormone treatment. If finasteride needs to be considered, it may interfere with the development of secondary sex characteristics.” Testosterone also increases sebum production and can increase acne, particularly in the first 6 months to 1 year after initiation, and with increased titration. “Some transmasculine youth may need oral isotretinoin, as stopping testosterone can be psychologically damaging,” Dr. Vance said.
“Unfortunately, the iPLEDGE program requirements can be perceived as gender nonaffirming, because patients must register by the sex assigned to them at birth, they must take pregnancy tests, and there can be provider assumptions about sexuality which does not equate with gender identity.”
He recommended having “open and honest” conversations with patients about the requirements and limitations of dispensing oral isotretinoin. “Assure the patient that you will be respectful and affirming of their gender identity while they’re in your office,” Dr. Vance advised. “If the patient has a mental health provider, you can strategize with them to reduce gender dysphoria around this process. Finally, advocating to change the system can not only be helpful for the patient in front of you, but for other patients who are in the same situation.”
He concluded his presentation by describing transgender youth as “some of the most resilient young people I’ve had the pleasure of working with.
“I think that we can all work to make sure they feel supported in who they are,” he said.
Dr. Vance reported having no relevant financial disclosures.
[email protected]
LAKE TAHOE, CALIF. – The way Stanley Vance Jr., MD, sees it, the No. 1 priority in the care of transgender youth is respecting their gender identity.
“This can really help with rapport and also help them continue to engage with your care,” he said at the annual meeting of the Society for Pediatric Dermatology.
One of the first steps is to establish the patient’s chosen name and pronouns. “Ask, use, and be consistent,” said Dr. Vance, an adolescent medicine specialist at the University of California, San Francisco. “Taking it to another level, you can implement system-level tools to ensure that all of your staff consistently use the chosen name and pronouns. and working with the IT department to have a place in our electronic medical record to put the chosen name and preferred pronouns.”
In a study published in the Journal of Adolescent Health, researchers found that the use of chosen names and pronouns for transgender use was associated with reduced depressive symptoms, suicidal ideation, and suicidal behavior among transgender youth.
Dr. Vance, who also holds a staff position at the UCSF Child and Adolescent Clinic, went on to discuss dermatologic considerations for gender diverse youth. In transgender females, estrogens can reduce the quantity and density of body and facial hair, “but it doesn’t necessarily get rid of the hair, so we may refer to dermatology for hair removal or hair reduction. There can also be a decrease in sebum production, which can lead to dry skin for those who are at risk.”
Transgender females often seek laser hair removal or electrolysis to aid in “blendability,” or how they perceive as being female or feminine. “We know that this can help in psychosocial outcomes for these young people,” Dr. Vance said. “Another reason why hair reduction and removal may be important is preoperatively for vaginoplasty.”
In transgender males, testosterone increases male pattern hair growth and can increase male pattern hair loss. “Minoxidil does not interact with gender-affirming hormone treatment. If finasteride needs to be considered, it may interfere with the development of secondary sex characteristics.” Testosterone also increases sebum production and can increase acne, particularly in the first 6 months to 1 year after initiation, and with increased titration. “Some transmasculine youth may need oral isotretinoin, as stopping testosterone can be psychologically damaging,” Dr. Vance said.
“Unfortunately, the iPLEDGE program requirements can be perceived as gender nonaffirming, because patients must register by the sex assigned to them at birth, they must take pregnancy tests, and there can be provider assumptions about sexuality which does not equate with gender identity.”
He recommended having “open and honest” conversations with patients about the requirements and limitations of dispensing oral isotretinoin. “Assure the patient that you will be respectful and affirming of their gender identity while they’re in your office,” Dr. Vance advised. “If the patient has a mental health provider, you can strategize with them to reduce gender dysphoria around this process. Finally, advocating to change the system can not only be helpful for the patient in front of you, but for other patients who are in the same situation.”
He concluded his presentation by describing transgender youth as “some of the most resilient young people I’ve had the pleasure of working with.
“I think that we can all work to make sure they feel supported in who they are,” he said.
Dr. Vance reported having no relevant financial disclosures.
[email protected]
EXPERT ANALYSIS FROM SPD 2018
Pediatric vitiligo primarily affects those aged 10-17
LAKE TAHOE, CALIF. – Among children and adolescents, vitiligo appears to predominately affect nonwhite boys and girls between the ages of 10 and 17 years, results from a large cross-sectional analysis demonstrated.
During an interview at the annual meeting of the Society for Pediatric Dermatology, lead study author Jessica Haber, MD, said that, while it’s known vitiligo can have its onset in childhood, there have been no population-based analyses in the United States specific to children and adolescents with the condition.
“We wanted to examine disease burden in the U.S. specifically, because we have such a diverse population,” said Dr. Haber, a second-year resident in the department of dermatology at Northwell Health, New York.
For the study, she and her associates used IBM’s Explorys research analytics platform to conduct a cross-sectional analysis of more than 55 million unique patients across all census regions of the United States. There were 1,630 vitiligo cases identified from a total of 4,242,400 pediatric patients, for an overall standard prevalence of 0.04%, or 40.1 per 100,000 children and adolescents. The proportion of female and male patients with vitiligo was similar (49.1% and 50.9%, respectively), and nearly three-fourths (72.3%) were 10 years of age or older.
The researchers observed no significant difference in the prevalence of vitiligo between males and females (40.2 per 100,000 vs. 40 per 100,000, respectively). The standardized prevalence of vitiligo was greatest in pediatric patients who were of “other” races and ethnicities (including Asian, Hispanic, multiracial, and other; 69.1 per 100,000), followed by African Americans (51.5 per 100,000) and whites (37.9 per 100,000). There were too few vitiligo cases among biracial patients to determine standardized estimates, but the crude prevalence was greatest in this group (68.7 per 100,000).
Two factors could contribute to the increased prevalence of vitiligo observed in nonwhite children and adolescents, Dr. Haber said. One is selection bias.
“It has been reported that both children and adults with higher Fitzpatrick skin types tend to have increased morbidity of their vitiligo, so it may be a selection bias that these patients are seeking out treatment for their disease,” she said. (J Am Acad Dermatol. 2017;77[1]:1-13). That might explain some of our findings, as well.”
While the study findings “don’t necessarily change clinical practice, it is good for us to have a sense of the burden of disease in the pediatric patient population of vitiligo, and to be aware that this is a disease that predominately affects non-Caucasian children and adolescents,” Dr. Haber concluded.
She reported having no financial disclosures.
LAKE TAHOE, CALIF. – Among children and adolescents, vitiligo appears to predominately affect nonwhite boys and girls between the ages of 10 and 17 years, results from a large cross-sectional analysis demonstrated.
During an interview at the annual meeting of the Society for Pediatric Dermatology, lead study author Jessica Haber, MD, said that, while it’s known vitiligo can have its onset in childhood, there have been no population-based analyses in the United States specific to children and adolescents with the condition.
“We wanted to examine disease burden in the U.S. specifically, because we have such a diverse population,” said Dr. Haber, a second-year resident in the department of dermatology at Northwell Health, New York.
For the study, she and her associates used IBM’s Explorys research analytics platform to conduct a cross-sectional analysis of more than 55 million unique patients across all census regions of the United States. There were 1,630 vitiligo cases identified from a total of 4,242,400 pediatric patients, for an overall standard prevalence of 0.04%, or 40.1 per 100,000 children and adolescents. The proportion of female and male patients with vitiligo was similar (49.1% and 50.9%, respectively), and nearly three-fourths (72.3%) were 10 years of age or older.
The researchers observed no significant difference in the prevalence of vitiligo between males and females (40.2 per 100,000 vs. 40 per 100,000, respectively). The standardized prevalence of vitiligo was greatest in pediatric patients who were of “other” races and ethnicities (including Asian, Hispanic, multiracial, and other; 69.1 per 100,000), followed by African Americans (51.5 per 100,000) and whites (37.9 per 100,000). There were too few vitiligo cases among biracial patients to determine standardized estimates, but the crude prevalence was greatest in this group (68.7 per 100,000).
Two factors could contribute to the increased prevalence of vitiligo observed in nonwhite children and adolescents, Dr. Haber said. One is selection bias.
“It has been reported that both children and adults with higher Fitzpatrick skin types tend to have increased morbidity of their vitiligo, so it may be a selection bias that these patients are seeking out treatment for their disease,” she said. (J Am Acad Dermatol. 2017;77[1]:1-13). That might explain some of our findings, as well.”
While the study findings “don’t necessarily change clinical practice, it is good for us to have a sense of the burden of disease in the pediatric patient population of vitiligo, and to be aware that this is a disease that predominately affects non-Caucasian children and adolescents,” Dr. Haber concluded.
She reported having no financial disclosures.
LAKE TAHOE, CALIF. – Among children and adolescents, vitiligo appears to predominately affect nonwhite boys and girls between the ages of 10 and 17 years, results from a large cross-sectional analysis demonstrated.
During an interview at the annual meeting of the Society for Pediatric Dermatology, lead study author Jessica Haber, MD, said that, while it’s known vitiligo can have its onset in childhood, there have been no population-based analyses in the United States specific to children and adolescents with the condition.
“We wanted to examine disease burden in the U.S. specifically, because we have such a diverse population,” said Dr. Haber, a second-year resident in the department of dermatology at Northwell Health, New York.
For the study, she and her associates used IBM’s Explorys research analytics platform to conduct a cross-sectional analysis of more than 55 million unique patients across all census regions of the United States. There were 1,630 vitiligo cases identified from a total of 4,242,400 pediatric patients, for an overall standard prevalence of 0.04%, or 40.1 per 100,000 children and adolescents. The proportion of female and male patients with vitiligo was similar (49.1% and 50.9%, respectively), and nearly three-fourths (72.3%) were 10 years of age or older.
The researchers observed no significant difference in the prevalence of vitiligo between males and females (40.2 per 100,000 vs. 40 per 100,000, respectively). The standardized prevalence of vitiligo was greatest in pediatric patients who were of “other” races and ethnicities (including Asian, Hispanic, multiracial, and other; 69.1 per 100,000), followed by African Americans (51.5 per 100,000) and whites (37.9 per 100,000). There were too few vitiligo cases among biracial patients to determine standardized estimates, but the crude prevalence was greatest in this group (68.7 per 100,000).
Two factors could contribute to the increased prevalence of vitiligo observed in nonwhite children and adolescents, Dr. Haber said. One is selection bias.
“It has been reported that both children and adults with higher Fitzpatrick skin types tend to have increased morbidity of their vitiligo, so it may be a selection bias that these patients are seeking out treatment for their disease,” she said. (J Am Acad Dermatol. 2017;77[1]:1-13). That might explain some of our findings, as well.”
While the study findings “don’t necessarily change clinical practice, it is good for us to have a sense of the burden of disease in the pediatric patient population of vitiligo, and to be aware that this is a disease that predominately affects non-Caucasian children and adolescents,” Dr. Haber concluded.
She reported having no financial disclosures.
REPORTING FROM SPD 2018
Key clinical point: Vitiligo appears to predominately affect nonwhite boys and girls 10 years of age and older in the pediatric population.
Major finding: Of pediatric patients with vitiligo, 72.3% were 10 years of age or older.
Study details: A cross-sectional analysis of 1,630 vitiligo cases identified from a total of 4,242,400 pediatric patients.
Disclosures: Dr. Haber reported having no relevant financial disclosures.
When fingernails are the clue to a bigger problem
CHICAGO – When a child or adolescent comes to the dermatologist’s office with a concern about fingernails or toenails, physician antennae may go up. “The world is different in the world of pediatrics – and even in the world of adolescents,” said Sheila Fallon Friedlander, MD.
In adults, the most common cause of nail dystrophy is tinea, but for younger pediatric patients, less than 1% of nail problems are attributable to fungus, so dermatologists may need to look further.
“It’s so important in kids to do a good history and physical exam,” said Dr. Friedlander, professor of dermatology and pediatrics at the University of California, San Diego. History-taking should include determining whether the condition has been present since birth and how nail appearance has changed over time.
For Dr. Friedlander, the approach to nail abnormalities includes a full head and skin exam. “I always look at the teeth, the hair, the skin,” she said; underlying bony anomalies also may surface. A complete exam often will turn up important clues if a syndrome underpins the nail abnormalities, she said, speaking at the American Academy of Dermatology summer meeting.
Her exemplar patient, she said, is a 19-year-old male who comes in with a parent because he’s bothered by his fingernails, which are dystrophic and small. A head-to-toe exam shows micronychia of both toes and fingers, with lunulae that are triangularly shaped. The hair, skin, and teeth of the patient all were normal in appearance. However, “The knees and elbows were odd,” Dr. Friedlander said.
This patient has nail-patella syndrome. “Even though it’s rare, I want you to think about it,” Dr. Friedlander said. The autosomal dominant condition is seen in about 1 in 50,000 patients. It’s thought to be caused by heterozygous loss-of-function mutations in gene LMX1B, she said, that codes for a LIM homeobox transcription factor 1 beta.
Though the small nails and triangular lunulae may be what brings the patient to the dermatologist’s office, a careful exam and one radiograph can pick up a tetrad of anomalies, Dr. Friedlander said. Abnormalities can be seen in both the knees and elbows; the patellae are often small, and may even be absent. In addition, a hip radiograph will show characteristic “horns” on the posterior iliac crests.
Coming back to the dermatologic exam, Dr. Friedlander said nails may be absent, hypoplastic, and dystrophic – but those are features that can be shared with other nail disorders, inherited and acquired. The pathognomonic finding for nail-patella syndrome is the presence of the triangular lunula, she said.
Now that the diagnosis has been made, Dr. Friedlander asked about this young man: “Where will you refer him?” Knowing the diagnosis means that there are a lot of calls for your staff to make, she said.
The patient with knee patella syndrome should be referred to an orthopedist to assess knees and elbows; radial head subluxation also is common in these patients, she said.
An ophthalmologic referral is important as well; hyperpigmentation of the pupillary margin – a “Lester iris” – can be seen, and increased rates of cataracts and glaucoma also are associated with nail-patella syndrome.
“,” Dr. Friedlander said. Up to half of nail-patella syndrome patients will have kidney involvement that initially presents with hematuria and proteinuria. Because the LMX1B mutation impairs how podocytes and glomerular filtration slits develop and function, up to 10% can develop end-stage renal failure, she said.
Parents also should be on the lookout for associated behavioral issues: “The other thing that’s interesting is that these kids have an increased risk of [attention-deficit/hyperactivity disorder] and major depression,” Dr. Friedlander said.
Dr. Friedlander reported that she had no relevant conflicts of interest.
SOURCE: Friedlander, S. Summer AAD 2018. Session F004.
CHICAGO – When a child or adolescent comes to the dermatologist’s office with a concern about fingernails or toenails, physician antennae may go up. “The world is different in the world of pediatrics – and even in the world of adolescents,” said Sheila Fallon Friedlander, MD.
In adults, the most common cause of nail dystrophy is tinea, but for younger pediatric patients, less than 1% of nail problems are attributable to fungus, so dermatologists may need to look further.
“It’s so important in kids to do a good history and physical exam,” said Dr. Friedlander, professor of dermatology and pediatrics at the University of California, San Diego. History-taking should include determining whether the condition has been present since birth and how nail appearance has changed over time.
For Dr. Friedlander, the approach to nail abnormalities includes a full head and skin exam. “I always look at the teeth, the hair, the skin,” she said; underlying bony anomalies also may surface. A complete exam often will turn up important clues if a syndrome underpins the nail abnormalities, she said, speaking at the American Academy of Dermatology summer meeting.
Her exemplar patient, she said, is a 19-year-old male who comes in with a parent because he’s bothered by his fingernails, which are dystrophic and small. A head-to-toe exam shows micronychia of both toes and fingers, with lunulae that are triangularly shaped. The hair, skin, and teeth of the patient all were normal in appearance. However, “The knees and elbows were odd,” Dr. Friedlander said.
This patient has nail-patella syndrome. “Even though it’s rare, I want you to think about it,” Dr. Friedlander said. The autosomal dominant condition is seen in about 1 in 50,000 patients. It’s thought to be caused by heterozygous loss-of-function mutations in gene LMX1B, she said, that codes for a LIM homeobox transcription factor 1 beta.
Though the small nails and triangular lunulae may be what brings the patient to the dermatologist’s office, a careful exam and one radiograph can pick up a tetrad of anomalies, Dr. Friedlander said. Abnormalities can be seen in both the knees and elbows; the patellae are often small, and may even be absent. In addition, a hip radiograph will show characteristic “horns” on the posterior iliac crests.
Coming back to the dermatologic exam, Dr. Friedlander said nails may be absent, hypoplastic, and dystrophic – but those are features that can be shared with other nail disorders, inherited and acquired. The pathognomonic finding for nail-patella syndrome is the presence of the triangular lunula, she said.
Now that the diagnosis has been made, Dr. Friedlander asked about this young man: “Where will you refer him?” Knowing the diagnosis means that there are a lot of calls for your staff to make, she said.
The patient with knee patella syndrome should be referred to an orthopedist to assess knees and elbows; radial head subluxation also is common in these patients, she said.
An ophthalmologic referral is important as well; hyperpigmentation of the pupillary margin – a “Lester iris” – can be seen, and increased rates of cataracts and glaucoma also are associated with nail-patella syndrome.
“,” Dr. Friedlander said. Up to half of nail-patella syndrome patients will have kidney involvement that initially presents with hematuria and proteinuria. Because the LMX1B mutation impairs how podocytes and glomerular filtration slits develop and function, up to 10% can develop end-stage renal failure, she said.
Parents also should be on the lookout for associated behavioral issues: “The other thing that’s interesting is that these kids have an increased risk of [attention-deficit/hyperactivity disorder] and major depression,” Dr. Friedlander said.
Dr. Friedlander reported that she had no relevant conflicts of interest.
SOURCE: Friedlander, S. Summer AAD 2018. Session F004.
CHICAGO – When a child or adolescent comes to the dermatologist’s office with a concern about fingernails or toenails, physician antennae may go up. “The world is different in the world of pediatrics – and even in the world of adolescents,” said Sheila Fallon Friedlander, MD.
In adults, the most common cause of nail dystrophy is tinea, but for younger pediatric patients, less than 1% of nail problems are attributable to fungus, so dermatologists may need to look further.
“It’s so important in kids to do a good history and physical exam,” said Dr. Friedlander, professor of dermatology and pediatrics at the University of California, San Diego. History-taking should include determining whether the condition has been present since birth and how nail appearance has changed over time.
For Dr. Friedlander, the approach to nail abnormalities includes a full head and skin exam. “I always look at the teeth, the hair, the skin,” she said; underlying bony anomalies also may surface. A complete exam often will turn up important clues if a syndrome underpins the nail abnormalities, she said, speaking at the American Academy of Dermatology summer meeting.
Her exemplar patient, she said, is a 19-year-old male who comes in with a parent because he’s bothered by his fingernails, which are dystrophic and small. A head-to-toe exam shows micronychia of both toes and fingers, with lunulae that are triangularly shaped. The hair, skin, and teeth of the patient all were normal in appearance. However, “The knees and elbows were odd,” Dr. Friedlander said.
This patient has nail-patella syndrome. “Even though it’s rare, I want you to think about it,” Dr. Friedlander said. The autosomal dominant condition is seen in about 1 in 50,000 patients. It’s thought to be caused by heterozygous loss-of-function mutations in gene LMX1B, she said, that codes for a LIM homeobox transcription factor 1 beta.
Though the small nails and triangular lunulae may be what brings the patient to the dermatologist’s office, a careful exam and one radiograph can pick up a tetrad of anomalies, Dr. Friedlander said. Abnormalities can be seen in both the knees and elbows; the patellae are often small, and may even be absent. In addition, a hip radiograph will show characteristic “horns” on the posterior iliac crests.
Coming back to the dermatologic exam, Dr. Friedlander said nails may be absent, hypoplastic, and dystrophic – but those are features that can be shared with other nail disorders, inherited and acquired. The pathognomonic finding for nail-patella syndrome is the presence of the triangular lunula, she said.
Now that the diagnosis has been made, Dr. Friedlander asked about this young man: “Where will you refer him?” Knowing the diagnosis means that there are a lot of calls for your staff to make, she said.
The patient with knee patella syndrome should be referred to an orthopedist to assess knees and elbows; radial head subluxation also is common in these patients, she said.
An ophthalmologic referral is important as well; hyperpigmentation of the pupillary margin – a “Lester iris” – can be seen, and increased rates of cataracts and glaucoma also are associated with nail-patella syndrome.
“,” Dr. Friedlander said. Up to half of nail-patella syndrome patients will have kidney involvement that initially presents with hematuria and proteinuria. Because the LMX1B mutation impairs how podocytes and glomerular filtration slits develop and function, up to 10% can develop end-stage renal failure, she said.
Parents also should be on the lookout for associated behavioral issues: “The other thing that’s interesting is that these kids have an increased risk of [attention-deficit/hyperactivity disorder] and major depression,” Dr. Friedlander said.
Dr. Friedlander reported that she had no relevant conflicts of interest.
SOURCE: Friedlander, S. Summer AAD 2018. Session F004.
REPORTING FROM SUMMER AAD 2018
Oral drug seen preventing angioedema attacks
An experimental agent reduced swelling episodes markedly in patients with hereditary angioedema, according to results from a phase 2 randomized, dose-ranging, placebo-controlled trial.
The drug BCX7353, developed by BioCryst Pharmaceuticals, is taken orally and works by inhibiting plasma kallikrein, an enzyme overexpressed in hereditary angioedema, a rare genetic disease that causes severe tissue swelling. In research published July 26 in the New England Journal of Medicine (N Engl J Med. 2018;379:352-62), Emel Aygören-Pürsün, MD, of Goethe University in Frankfurt, Germany, and colleagues, randomized 77 patients with type 1 or II hereditary angioedema and a pattern of frequent attacks to one of four doses of daily BCX7353, or placebo, for 28 days.
Dr. Aygören-Pürsün’s group found significant reductions in the number of monthly attacks for the three doses used in the study, with the best response seen in the group receiving the second-lowest dose of 125 mg. These patients saw a reduction of 73.8% (P less than .001) in monthly attacks from baseline, and 43% of patients receiving that dose had no attacks during the study period. The higher-dose groups saw more adverse events and apparently less efficacy, with the 250-mg dose associated with a reduction in attacks of 44.6% (P = .01), and for the 350-mg group, a 45.5% reduction (P = .006).
Patients receiving the lowest dose in the study, 62.5 mg, saw a small (about 10%) reduction in attacks that did not reach statistical significance. Gastrointestinal adverse events were reported in the two highest dose groups, and three patients in the 350-mg group dropped out after reporting serious adverse events, including one liver disorder considered likely related to the trial regimen.
The efficacy of the highest doses “was probably masked by gastrointestinal adverse events that may have been misattributed as early symptoms of abdominal angioedema attacks,” the investigators wrote in their analysis. Improvements in angioedema-related quality of life scores, a secondary trial endpoint, reached statistical significance for the 125- and 250-mg doses.
The authors cautioned that the safety of long-term dosing would need to be studied in longer-term trials.
The study was sponsored by the drug manufacturer, BioCryst Pharmaceuticals. All of the study’s authors, including the lead author, disclosed financial relationships in the form of grant support, fees, or employment with the study sponsor.
SOURCE: Aygören-Pürsün et al. N Engl J Med. 2018;379:352-62.
An experimental agent reduced swelling episodes markedly in patients with hereditary angioedema, according to results from a phase 2 randomized, dose-ranging, placebo-controlled trial.
The drug BCX7353, developed by BioCryst Pharmaceuticals, is taken orally and works by inhibiting plasma kallikrein, an enzyme overexpressed in hereditary angioedema, a rare genetic disease that causes severe tissue swelling. In research published July 26 in the New England Journal of Medicine (N Engl J Med. 2018;379:352-62), Emel Aygören-Pürsün, MD, of Goethe University in Frankfurt, Germany, and colleagues, randomized 77 patients with type 1 or II hereditary angioedema and a pattern of frequent attacks to one of four doses of daily BCX7353, or placebo, for 28 days.
Dr. Aygören-Pürsün’s group found significant reductions in the number of monthly attacks for the three doses used in the study, with the best response seen in the group receiving the second-lowest dose of 125 mg. These patients saw a reduction of 73.8% (P less than .001) in monthly attacks from baseline, and 43% of patients receiving that dose had no attacks during the study period. The higher-dose groups saw more adverse events and apparently less efficacy, with the 250-mg dose associated with a reduction in attacks of 44.6% (P = .01), and for the 350-mg group, a 45.5% reduction (P = .006).
Patients receiving the lowest dose in the study, 62.5 mg, saw a small (about 10%) reduction in attacks that did not reach statistical significance. Gastrointestinal adverse events were reported in the two highest dose groups, and three patients in the 350-mg group dropped out after reporting serious adverse events, including one liver disorder considered likely related to the trial regimen.
The efficacy of the highest doses “was probably masked by gastrointestinal adverse events that may have been misattributed as early symptoms of abdominal angioedema attacks,” the investigators wrote in their analysis. Improvements in angioedema-related quality of life scores, a secondary trial endpoint, reached statistical significance for the 125- and 250-mg doses.
The authors cautioned that the safety of long-term dosing would need to be studied in longer-term trials.
The study was sponsored by the drug manufacturer, BioCryst Pharmaceuticals. All of the study’s authors, including the lead author, disclosed financial relationships in the form of grant support, fees, or employment with the study sponsor.
SOURCE: Aygören-Pürsün et al. N Engl J Med. 2018;379:352-62.
An experimental agent reduced swelling episodes markedly in patients with hereditary angioedema, according to results from a phase 2 randomized, dose-ranging, placebo-controlled trial.
The drug BCX7353, developed by BioCryst Pharmaceuticals, is taken orally and works by inhibiting plasma kallikrein, an enzyme overexpressed in hereditary angioedema, a rare genetic disease that causes severe tissue swelling. In research published July 26 in the New England Journal of Medicine (N Engl J Med. 2018;379:352-62), Emel Aygören-Pürsün, MD, of Goethe University in Frankfurt, Germany, and colleagues, randomized 77 patients with type 1 or II hereditary angioedema and a pattern of frequent attacks to one of four doses of daily BCX7353, or placebo, for 28 days.
Dr. Aygören-Pürsün’s group found significant reductions in the number of monthly attacks for the three doses used in the study, with the best response seen in the group receiving the second-lowest dose of 125 mg. These patients saw a reduction of 73.8% (P less than .001) in monthly attacks from baseline, and 43% of patients receiving that dose had no attacks during the study period. The higher-dose groups saw more adverse events and apparently less efficacy, with the 250-mg dose associated with a reduction in attacks of 44.6% (P = .01), and for the 350-mg group, a 45.5% reduction (P = .006).
Patients receiving the lowest dose in the study, 62.5 mg, saw a small (about 10%) reduction in attacks that did not reach statistical significance. Gastrointestinal adverse events were reported in the two highest dose groups, and three patients in the 350-mg group dropped out after reporting serious adverse events, including one liver disorder considered likely related to the trial regimen.
The efficacy of the highest doses “was probably masked by gastrointestinal adverse events that may have been misattributed as early symptoms of abdominal angioedema attacks,” the investigators wrote in their analysis. Improvements in angioedema-related quality of life scores, a secondary trial endpoint, reached statistical significance for the 125- and 250-mg doses.
The authors cautioned that the safety of long-term dosing would need to be studied in longer-term trials.
The study was sponsored by the drug manufacturer, BioCryst Pharmaceuticals. All of the study’s authors, including the lead author, disclosed financial relationships in the form of grant support, fees, or employment with the study sponsor.
SOURCE: Aygören-Pürsün et al. N Engl J Med. 2018;379:352-62.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Key clinical point: An experimental kallikrein inhibitor, taken daily, prevented swelling attacks in people with hereditary angioedema.
Major finding: Patients in a moderate-dose group saw a 73.8% reduction in monthly swelling attacks from baseline, and 43% of saw no attacks.
Study details: A phase 2 study randomizing 77 patients with hereditary angioedema in three countries to one of four escalating doses or placebo.
Disclosures: All authors disclosed some relationship (current or past grant or fee support, or employment) with sponsor, BioCryst Pharmaceuticals.
Source: Aygören-Pürsün et. al. N Engl J Med. 2018;379:352-62.
Study explores adolescents’ views on their skin tone, pressure to tan
LAKE TAHOE, CALIF. –
During an interview at the annual meeting of the Society for Pediatric Dermatology, lead study author Shivani Patel, MD, said that prior research on skin color had focused mainly on adults and its impact on self-esteem and perceived attractiveness, yet little data are available on perceptions of skin color among adolescents.
“During puberty, adolescents receive pressure from friends, family, and social media to conform to a certain acceptable standard of skin tone,” said Dr. Patel, a chief resident in the department of dermatology at Johns Hopkins University, Baltimore. “They will often engage in risky behaviors such as tanning bed use, suntanning, and use of skin lightening creams.”
In an effort to characterize the attitudes of adolescents about their skin tone, she and her associates recruited 50 patients aged 12-19 years who were seen at the Johns Hopkins dermatology clinics. Slightly more than half (56%) were female. They were asked to complete surveys on their use of sunscreen, tanning beds, and skin-lightening creams, as well as to report any family or friends who have used these interventions.
Next, the researchers used Pantone’s Capsure device to record each subject’s skin tone according to a palate of 110 skin colors available from Pantone’s SkinTone Guide, which is intended to match and reproduce lifelike skin tones in a variety of industries. The adolescents were then given the palette and asked which skin tone they felt best represented their skin and which skin tone they wished they had. These differences were compared with their objective measurement by the study team.
Of all respondents, 20% indicated that they felt pressure to have tan skin and were likely to engage in suntanning (P less than .001), a feeling they said started around the age of 12 years and stemmed from perceived pressure from friends and celebrity figures. Those who suntanned were more likely to wear sunscreen (P less than .01), a finding “that was reassuring and showed that they are aware of sunscreen and sun safety,” Dr. Patel said. However, about half of the respondents reported never wearing sunscreen and only two reported wearing sunscreen daily. No one reported using tanning beds, but 8% reported that a family member used them. One adolescent reported using skin lightening creams, and three reported that their mothers used them.
The researchers also found that black and Asian study participants were significantly more likely to desire a skin tone lighter than what they perceived their skin tone to be, while white participants were significantly more likely to desire a darker skin tone (P less than .011 for both associations).
The findings suggest that sun safety initiatives should target prepubertal patients before they engage in risky behaviors, Dr. Patel said. She acknowledged that the small sample size is a limitation of the study, but said that she and her associates hope to conduct a larger-scale analysis.
She reported having no financial disclosures.
LAKE TAHOE, CALIF. –
During an interview at the annual meeting of the Society for Pediatric Dermatology, lead study author Shivani Patel, MD, said that prior research on skin color had focused mainly on adults and its impact on self-esteem and perceived attractiveness, yet little data are available on perceptions of skin color among adolescents.
“During puberty, adolescents receive pressure from friends, family, and social media to conform to a certain acceptable standard of skin tone,” said Dr. Patel, a chief resident in the department of dermatology at Johns Hopkins University, Baltimore. “They will often engage in risky behaviors such as tanning bed use, suntanning, and use of skin lightening creams.”
In an effort to characterize the attitudes of adolescents about their skin tone, she and her associates recruited 50 patients aged 12-19 years who were seen at the Johns Hopkins dermatology clinics. Slightly more than half (56%) were female. They were asked to complete surveys on their use of sunscreen, tanning beds, and skin-lightening creams, as well as to report any family or friends who have used these interventions.
Next, the researchers used Pantone’s Capsure device to record each subject’s skin tone according to a palate of 110 skin colors available from Pantone’s SkinTone Guide, which is intended to match and reproduce lifelike skin tones in a variety of industries. The adolescents were then given the palette and asked which skin tone they felt best represented their skin and which skin tone they wished they had. These differences were compared with their objective measurement by the study team.
Of all respondents, 20% indicated that they felt pressure to have tan skin and were likely to engage in suntanning (P less than .001), a feeling they said started around the age of 12 years and stemmed from perceived pressure from friends and celebrity figures. Those who suntanned were more likely to wear sunscreen (P less than .01), a finding “that was reassuring and showed that they are aware of sunscreen and sun safety,” Dr. Patel said. However, about half of the respondents reported never wearing sunscreen and only two reported wearing sunscreen daily. No one reported using tanning beds, but 8% reported that a family member used them. One adolescent reported using skin lightening creams, and three reported that their mothers used them.
The researchers also found that black and Asian study participants were significantly more likely to desire a skin tone lighter than what they perceived their skin tone to be, while white participants were significantly more likely to desire a darker skin tone (P less than .011 for both associations).
The findings suggest that sun safety initiatives should target prepubertal patients before they engage in risky behaviors, Dr. Patel said. She acknowledged that the small sample size is a limitation of the study, but said that she and her associates hope to conduct a larger-scale analysis.
She reported having no financial disclosures.
LAKE TAHOE, CALIF. –
During an interview at the annual meeting of the Society for Pediatric Dermatology, lead study author Shivani Patel, MD, said that prior research on skin color had focused mainly on adults and its impact on self-esteem and perceived attractiveness, yet little data are available on perceptions of skin color among adolescents.
“During puberty, adolescents receive pressure from friends, family, and social media to conform to a certain acceptable standard of skin tone,” said Dr. Patel, a chief resident in the department of dermatology at Johns Hopkins University, Baltimore. “They will often engage in risky behaviors such as tanning bed use, suntanning, and use of skin lightening creams.”
In an effort to characterize the attitudes of adolescents about their skin tone, she and her associates recruited 50 patients aged 12-19 years who were seen at the Johns Hopkins dermatology clinics. Slightly more than half (56%) were female. They were asked to complete surveys on their use of sunscreen, tanning beds, and skin-lightening creams, as well as to report any family or friends who have used these interventions.
Next, the researchers used Pantone’s Capsure device to record each subject’s skin tone according to a palate of 110 skin colors available from Pantone’s SkinTone Guide, which is intended to match and reproduce lifelike skin tones in a variety of industries. The adolescents were then given the palette and asked which skin tone they felt best represented their skin and which skin tone they wished they had. These differences were compared with their objective measurement by the study team.
Of all respondents, 20% indicated that they felt pressure to have tan skin and were likely to engage in suntanning (P less than .001), a feeling they said started around the age of 12 years and stemmed from perceived pressure from friends and celebrity figures. Those who suntanned were more likely to wear sunscreen (P less than .01), a finding “that was reassuring and showed that they are aware of sunscreen and sun safety,” Dr. Patel said. However, about half of the respondents reported never wearing sunscreen and only two reported wearing sunscreen daily. No one reported using tanning beds, but 8% reported that a family member used them. One adolescent reported using skin lightening creams, and three reported that their mothers used them.
The researchers also found that black and Asian study participants were significantly more likely to desire a skin tone lighter than what they perceived their skin tone to be, while white participants were significantly more likely to desire a darker skin tone (P less than .011 for both associations).
The findings suggest that sun safety initiatives should target prepubertal patients before they engage in risky behaviors, Dr. Patel said. She acknowledged that the small sample size is a limitation of the study, but said that she and her associates hope to conduct a larger-scale analysis.
She reported having no financial disclosures.
REPORTING FROM SPD 2018
Key clinical point: Sun safety initiatives should target prepubertal patients before they engage in risky behaviors.
Major finding: One in five adolescents indicated that they felt pressure to have tan skin and were likely to engage in suntanning (P less than .001).
Study details: A survey of 50 patients aged 12-19 years.
Disclosures: Dr. Patel reported having no financial disclosures.
Atypical pigmented lesions on back
The FP recognized that this patient had multiple dysplastic nevi (DN) and was concerned about a possible melanoma.
Although this patient did not have the familial atypical mole and melanoma (FAMM) syndrome, she was at higher risk of developing a melanoma based on the numerous DN that were so visibly apparent on her trunk. (A diagnosis of FAMM requires the occurrence of melanoma in one or more first- or second-degree relatives.)
Total body photography is the current state-of-the-art technology for monitoring patients with multiple atypical moles at higher risk for skin cancer. Small changes in DN and new nevi can be seen best with this technology. Individual flat lesions also can be monitored with dermoscopy over 3 to 4 months.
It is not recommended, however, to follow a suspicious raised lesion over time with dermoscopy and/or photography because a raised lesion might be a nodular melanoma and a delay of diagnosis of over 3 months may worsen the prognosis.
In this case, the patient was happy to be referred to a dermatologist with total body photography. Fortunately, the patient reported back to her FP that the dermatologist did not detect any melanomas, and 2 biopsies that were performed came back as DN only.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Smith M. Epidermal nevus and nevus sebaceous. In: Usatine R, Smith M, Mayeaux EJ, et al. Color Atlas of Family Medicine. 2nd ed. New York, NY: McGraw-Hill; 2013:958-962.
To learn more about the Color Atlas of Family Medicine, see: www.amazon.com/Color-Family-Medicine-Richard-Usatine/dp/0071769641/.
You can now get the second edition of the Color Atlas of Family Medicine as an app by clicking on this link: usatinemedia.com.

The FP recognized that this patient had multiple dysplastic nevi (DN) and was concerned about a possible melanoma.
Although this patient did not have the familial atypical mole and melanoma (FAMM) syndrome, she was at higher risk of developing a melanoma based on the numerous DN that were so visibly apparent on her trunk. (A diagnosis of FAMM requires the occurrence of melanoma in one or more first- or second-degree relatives.)
Total body photography is the current state-of-the-art technology for monitoring patients with multiple atypical moles at higher risk for skin cancer. Small changes in DN and new nevi can be seen best with this technology. Individual flat lesions also can be monitored with dermoscopy over 3 to 4 months.
It is not recommended, however, to follow a suspicious raised lesion over time with dermoscopy and/or photography because a raised lesion might be a nodular melanoma and a delay of diagnosis of over 3 months may worsen the prognosis.
In this case, the patient was happy to be referred to a dermatologist with total body photography. Fortunately, the patient reported back to her FP that the dermatologist did not detect any melanomas, and 2 biopsies that were performed came back as DN only.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Smith M. Epidermal nevus and nevus sebaceous. In: Usatine R, Smith M, Mayeaux EJ, et al. Color Atlas of Family Medicine. 2nd ed. New York, NY: McGraw-Hill; 2013:958-962.
To learn more about the Color Atlas of Family Medicine, see: www.amazon.com/Color-Family-Medicine-Richard-Usatine/dp/0071769641/.
You can now get the second edition of the Color Atlas of Family Medicine as an app by clicking on this link: usatinemedia.com.

The FP recognized that this patient had multiple dysplastic nevi (DN) and was concerned about a possible melanoma.
Although this patient did not have the familial atypical mole and melanoma (FAMM) syndrome, she was at higher risk of developing a melanoma based on the numerous DN that were so visibly apparent on her trunk. (A diagnosis of FAMM requires the occurrence of melanoma in one or more first- or second-degree relatives.)
Total body photography is the current state-of-the-art technology for monitoring patients with multiple atypical moles at higher risk for skin cancer. Small changes in DN and new nevi can be seen best with this technology. Individual flat lesions also can be monitored with dermoscopy over 3 to 4 months.
It is not recommended, however, to follow a suspicious raised lesion over time with dermoscopy and/or photography because a raised lesion might be a nodular melanoma and a delay of diagnosis of over 3 months may worsen the prognosis.
In this case, the patient was happy to be referred to a dermatologist with total body photography. Fortunately, the patient reported back to her FP that the dermatologist did not detect any melanomas, and 2 biopsies that were performed came back as DN only.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Smith M. Epidermal nevus and nevus sebaceous. In: Usatine R, Smith M, Mayeaux EJ, et al. Color Atlas of Family Medicine. 2nd ed. New York, NY: McGraw-Hill; 2013:958-962.
To learn more about the Color Atlas of Family Medicine, see: www.amazon.com/Color-Family-Medicine-Richard-Usatine/dp/0071769641/.
You can now get the second edition of the Color Atlas of Family Medicine as an app by clicking on this link: usatinemedia.com.
Early-onset atopic dermatitis linked to elevated risk for seasonal allergies and asthma
LAKE TAHOE, CALIF. – results from a large, retrospective cohort study demonstrated.
“The atopic march is characterized by a progression from atopic dermatitis, usually early in childhood, to subsequent development of allergic rhinitis and asthma, lead study author Joy Wan, MD, said at the annual meeting of the Society for Pediatric Dermatology. “It is thought that the skin acts as the site of primary sensitization through a defective epithelial barrier, which then allows for allergic sensitization to occur in the airways. It is estimated that 30%-60% of AD patients go on to develop asthma and/or allergic rhinitis. However, not all patients complete the so-called atopic march, and this variation in the risk of asthma and allergic rhinitis among AD patients is not very well understood. Better ways to risk stratify these patients are needed.”
One possible explanation for this variation in the risk of atopy in AD patients could be the timing of their dermatitis onset. “We know that atopic dermatitis begins in infancy, but it can start at any age,” said Dr. Wan, who is a fellow in the section of pediatric dermatology at the Children’s Hospital of Philadelphia. “There has been a distinction between early-onset versus late-onset AD. Some past studies have also suggested that there is an increased risk of asthma and allergic rhinitis in children who have early-onset AD before the age of 1 or 2. This suggests that perhaps the model of the atopic march varies between early- and late-onset AD. However, past studies have had several limitations. They’ve often had short durations of follow-up, they’ve only examined narrow ranges of age of onset for AD, and most of them have been designed to primarily evaluate other exposures and outcomes, rather than looking at the timing of AD onset itself.”
For the current study, Dr. Wan and her associates set out to examine the risk of seasonal allergies and asthma among children with AD with respect to the age of AD onset. They used data from the Pediatric Eczema Elective Registry (PEER), an ongoing, prospective U.S. cohort of more than 7,700 children with physician-confirmed AD (JAMA Dermatol. 2014 Jun;150:593-600). All registry participants had used pimecrolimus cream in the past, but children with lymphoproliferative disease were excluded from the registry, as were those with malignancy or those who required the use of systemic immunosuppression.
The researchers evaluated 3,966 subjects in PEER with at least 3 years of follow-up. The exposure of interest was age of AD onset, and they divided patients into three broad age categories: early onset (age 2 years or younger), mid onset (3-7 years), and late onset (8-17 years). Primary outcomes were prevalent seasonal allergies and asthma at the time of registry enrollment, and incident seasonal allergies and asthma during follow-up, assessed via patient surveys every 3 years.
The study population included high proportions of white and black children, and there was a slight predominance of females. The median age at PEER enrollment increased with advancing age of AD onset (5.2 years in the early-onset group vs. 8.2 years in the mid-onset group and 13.1 years in the late-onset group), while the duration of follow-up was fairly similar across the three groups (a median of about 8.3 months). Family history of AD was common across all three groups, while patients in the late-onset group tended to have better control of their AD, compared with their younger counterparts.
At baseline, the prevalence of seasonal allergies was highest among the early-onset group at 74.6%, compared with 69.9% among the mid-onset group and 70.1% among the late-onset group. After adjusting for sex, race, and age at registry enrollment, the relative risk for prevalent seasonal allergies was 9% lower in the mid-onset group (0.91) and 18% lower in the late-onset group (0.82), compared with those in the early-onset group. Next, Dr. Wan and her associates calculated the incidence of seasonal allergies among 1,054 patients who did not have allergies at baseline. The cumulative incidence was highest among the early-onset group (56.1%), followed by the mid-onset group (46.8%), and the late-onset group (30.6%). On adjusted analysis, the relative risk for seasonal allergies among patients who had no allergies at baseline was 18% lower in the mid-onset group (0.82) and 36% lower in the late-onset group (0.64), compared with those in the early-onset group.
In the analysis of asthma risk by age of AD onset, prevalence was highest among patients in the early-onset group at 51.5%, compared with 44.7% among the mid-onset age group and 43% among the late-onset age group. On adjusted analysis, the relative risk for asthma was 15% lower in the mid-onset group (0.85) and 29% lower in the late-onset group (0.71), compared with those in the early-onset group. Meanwhile, the cumulative incidence of asthma among patients without asthma at baseline was also highest in the early-onset group (39.2%), compared with 31.9% in the mid-onset group and 29.9% in the late-onset group.
On adjusted analysis, the relative risk for asthma among this subset of patients was 4% lower in the mid-onset group (0.96) and 8% lower in the late-onset group (0.92), compared with those in the early-onset group, a difference that was not statistically significant. “One possible explanation for this is that asthma tends to develop soon after AD does, and the rates of developing asthma later on, as detected by our study, are nondifferential,” Dr. Wan said. “Another possibility is that the impact of early-onset versus late-onset AD is just different for asthma than it is for seasonal allergies.”
She acknowledged certain limitations of the study, including the risk of misclassification bias and limitations in recall with self-reported data, and the fact that the findings may not be generalizable to all patients with AD.
“Future studies with longer follow-up and studies of adult-onset AD will help extend our findings,” she concluded. “Nevertheless, our findings may inform how we risk stratify patients for AD treatment or atopic march prevention efforts in the future.”
PEER is funded by a grant from Valeant Pharmaceuticals, but Valeant had no role in this study. Dr. Wan reported having no financial disclosures. The study won an award at the meeting for best research presented by a dermatology resident or fellow.
LAKE TAHOE, CALIF. – results from a large, retrospective cohort study demonstrated.
“The atopic march is characterized by a progression from atopic dermatitis, usually early in childhood, to subsequent development of allergic rhinitis and asthma, lead study author Joy Wan, MD, said at the annual meeting of the Society for Pediatric Dermatology. “It is thought that the skin acts as the site of primary sensitization through a defective epithelial barrier, which then allows for allergic sensitization to occur in the airways. It is estimated that 30%-60% of AD patients go on to develop asthma and/or allergic rhinitis. However, not all patients complete the so-called atopic march, and this variation in the risk of asthma and allergic rhinitis among AD patients is not very well understood. Better ways to risk stratify these patients are needed.”
One possible explanation for this variation in the risk of atopy in AD patients could be the timing of their dermatitis onset. “We know that atopic dermatitis begins in infancy, but it can start at any age,” said Dr. Wan, who is a fellow in the section of pediatric dermatology at the Children’s Hospital of Philadelphia. “There has been a distinction between early-onset versus late-onset AD. Some past studies have also suggested that there is an increased risk of asthma and allergic rhinitis in children who have early-onset AD before the age of 1 or 2. This suggests that perhaps the model of the atopic march varies between early- and late-onset AD. However, past studies have had several limitations. They’ve often had short durations of follow-up, they’ve only examined narrow ranges of age of onset for AD, and most of them have been designed to primarily evaluate other exposures and outcomes, rather than looking at the timing of AD onset itself.”
For the current study, Dr. Wan and her associates set out to examine the risk of seasonal allergies and asthma among children with AD with respect to the age of AD onset. They used data from the Pediatric Eczema Elective Registry (PEER), an ongoing, prospective U.S. cohort of more than 7,700 children with physician-confirmed AD (JAMA Dermatol. 2014 Jun;150:593-600). All registry participants had used pimecrolimus cream in the past, but children with lymphoproliferative disease were excluded from the registry, as were those with malignancy or those who required the use of systemic immunosuppression.
The researchers evaluated 3,966 subjects in PEER with at least 3 years of follow-up. The exposure of interest was age of AD onset, and they divided patients into three broad age categories: early onset (age 2 years or younger), mid onset (3-7 years), and late onset (8-17 years). Primary outcomes were prevalent seasonal allergies and asthma at the time of registry enrollment, and incident seasonal allergies and asthma during follow-up, assessed via patient surveys every 3 years.
The study population included high proportions of white and black children, and there was a slight predominance of females. The median age at PEER enrollment increased with advancing age of AD onset (5.2 years in the early-onset group vs. 8.2 years in the mid-onset group and 13.1 years in the late-onset group), while the duration of follow-up was fairly similar across the three groups (a median of about 8.3 months). Family history of AD was common across all three groups, while patients in the late-onset group tended to have better control of their AD, compared with their younger counterparts.
At baseline, the prevalence of seasonal allergies was highest among the early-onset group at 74.6%, compared with 69.9% among the mid-onset group and 70.1% among the late-onset group. After adjusting for sex, race, and age at registry enrollment, the relative risk for prevalent seasonal allergies was 9% lower in the mid-onset group (0.91) and 18% lower in the late-onset group (0.82), compared with those in the early-onset group. Next, Dr. Wan and her associates calculated the incidence of seasonal allergies among 1,054 patients who did not have allergies at baseline. The cumulative incidence was highest among the early-onset group (56.1%), followed by the mid-onset group (46.8%), and the late-onset group (30.6%). On adjusted analysis, the relative risk for seasonal allergies among patients who had no allergies at baseline was 18% lower in the mid-onset group (0.82) and 36% lower in the late-onset group (0.64), compared with those in the early-onset group.
In the analysis of asthma risk by age of AD onset, prevalence was highest among patients in the early-onset group at 51.5%, compared with 44.7% among the mid-onset age group and 43% among the late-onset age group. On adjusted analysis, the relative risk for asthma was 15% lower in the mid-onset group (0.85) and 29% lower in the late-onset group (0.71), compared with those in the early-onset group. Meanwhile, the cumulative incidence of asthma among patients without asthma at baseline was also highest in the early-onset group (39.2%), compared with 31.9% in the mid-onset group and 29.9% in the late-onset group.
On adjusted analysis, the relative risk for asthma among this subset of patients was 4% lower in the mid-onset group (0.96) and 8% lower in the late-onset group (0.92), compared with those in the early-onset group, a difference that was not statistically significant. “One possible explanation for this is that asthma tends to develop soon after AD does, and the rates of developing asthma later on, as detected by our study, are nondifferential,” Dr. Wan said. “Another possibility is that the impact of early-onset versus late-onset AD is just different for asthma than it is for seasonal allergies.”
She acknowledged certain limitations of the study, including the risk of misclassification bias and limitations in recall with self-reported data, and the fact that the findings may not be generalizable to all patients with AD.
“Future studies with longer follow-up and studies of adult-onset AD will help extend our findings,” she concluded. “Nevertheless, our findings may inform how we risk stratify patients for AD treatment or atopic march prevention efforts in the future.”
PEER is funded by a grant from Valeant Pharmaceuticals, but Valeant had no role in this study. Dr. Wan reported having no financial disclosures. The study won an award at the meeting for best research presented by a dermatology resident or fellow.
LAKE TAHOE, CALIF. – results from a large, retrospective cohort study demonstrated.
“The atopic march is characterized by a progression from atopic dermatitis, usually early in childhood, to subsequent development of allergic rhinitis and asthma, lead study author Joy Wan, MD, said at the annual meeting of the Society for Pediatric Dermatology. “It is thought that the skin acts as the site of primary sensitization through a defective epithelial barrier, which then allows for allergic sensitization to occur in the airways. It is estimated that 30%-60% of AD patients go on to develop asthma and/or allergic rhinitis. However, not all patients complete the so-called atopic march, and this variation in the risk of asthma and allergic rhinitis among AD patients is not very well understood. Better ways to risk stratify these patients are needed.”
One possible explanation for this variation in the risk of atopy in AD patients could be the timing of their dermatitis onset. “We know that atopic dermatitis begins in infancy, but it can start at any age,” said Dr. Wan, who is a fellow in the section of pediatric dermatology at the Children’s Hospital of Philadelphia. “There has been a distinction between early-onset versus late-onset AD. Some past studies have also suggested that there is an increased risk of asthma and allergic rhinitis in children who have early-onset AD before the age of 1 or 2. This suggests that perhaps the model of the atopic march varies between early- and late-onset AD. However, past studies have had several limitations. They’ve often had short durations of follow-up, they’ve only examined narrow ranges of age of onset for AD, and most of them have been designed to primarily evaluate other exposures and outcomes, rather than looking at the timing of AD onset itself.”
For the current study, Dr. Wan and her associates set out to examine the risk of seasonal allergies and asthma among children with AD with respect to the age of AD onset. They used data from the Pediatric Eczema Elective Registry (PEER), an ongoing, prospective U.S. cohort of more than 7,700 children with physician-confirmed AD (JAMA Dermatol. 2014 Jun;150:593-600). All registry participants had used pimecrolimus cream in the past, but children with lymphoproliferative disease were excluded from the registry, as were those with malignancy or those who required the use of systemic immunosuppression.
The researchers evaluated 3,966 subjects in PEER with at least 3 years of follow-up. The exposure of interest was age of AD onset, and they divided patients into three broad age categories: early onset (age 2 years or younger), mid onset (3-7 years), and late onset (8-17 years). Primary outcomes were prevalent seasonal allergies and asthma at the time of registry enrollment, and incident seasonal allergies and asthma during follow-up, assessed via patient surveys every 3 years.
The study population included high proportions of white and black children, and there was a slight predominance of females. The median age at PEER enrollment increased with advancing age of AD onset (5.2 years in the early-onset group vs. 8.2 years in the mid-onset group and 13.1 years in the late-onset group), while the duration of follow-up was fairly similar across the three groups (a median of about 8.3 months). Family history of AD was common across all three groups, while patients in the late-onset group tended to have better control of their AD, compared with their younger counterparts.
At baseline, the prevalence of seasonal allergies was highest among the early-onset group at 74.6%, compared with 69.9% among the mid-onset group and 70.1% among the late-onset group. After adjusting for sex, race, and age at registry enrollment, the relative risk for prevalent seasonal allergies was 9% lower in the mid-onset group (0.91) and 18% lower in the late-onset group (0.82), compared with those in the early-onset group. Next, Dr. Wan and her associates calculated the incidence of seasonal allergies among 1,054 patients who did not have allergies at baseline. The cumulative incidence was highest among the early-onset group (56.1%), followed by the mid-onset group (46.8%), and the late-onset group (30.6%). On adjusted analysis, the relative risk for seasonal allergies among patients who had no allergies at baseline was 18% lower in the mid-onset group (0.82) and 36% lower in the late-onset group (0.64), compared with those in the early-onset group.
In the analysis of asthma risk by age of AD onset, prevalence was highest among patients in the early-onset group at 51.5%, compared with 44.7% among the mid-onset age group and 43% among the late-onset age group. On adjusted analysis, the relative risk for asthma was 15% lower in the mid-onset group (0.85) and 29% lower in the late-onset group (0.71), compared with those in the early-onset group. Meanwhile, the cumulative incidence of asthma among patients without asthma at baseline was also highest in the early-onset group (39.2%), compared with 31.9% in the mid-onset group and 29.9% in the late-onset group.
On adjusted analysis, the relative risk for asthma among this subset of patients was 4% lower in the mid-onset group (0.96) and 8% lower in the late-onset group (0.92), compared with those in the early-onset group, a difference that was not statistically significant. “One possible explanation for this is that asthma tends to develop soon after AD does, and the rates of developing asthma later on, as detected by our study, are nondifferential,” Dr. Wan said. “Another possibility is that the impact of early-onset versus late-onset AD is just different for asthma than it is for seasonal allergies.”
She acknowledged certain limitations of the study, including the risk of misclassification bias and limitations in recall with self-reported data, and the fact that the findings may not be generalizable to all patients with AD.
“Future studies with longer follow-up and studies of adult-onset AD will help extend our findings,” she concluded. “Nevertheless, our findings may inform how we risk stratify patients for AD treatment or atopic march prevention efforts in the future.”
PEER is funded by a grant from Valeant Pharmaceuticals, but Valeant had no role in this study. Dr. Wan reported having no financial disclosures. The study won an award at the meeting for best research presented by a dermatology resident or fellow.
AT SPD 2018