User login
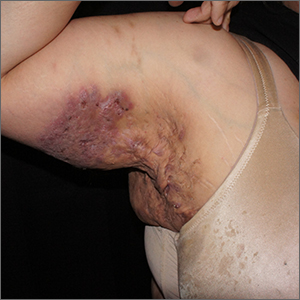
Dupilumab-associated lymphoid reactions require caution
, according to a study published in JAMA Dermatology
The potential for such reactions requires diagnosing AD carefully, monitoring patients on dupilumab for new and unusual symptoms, and thoroughly working up suspicious LRs, according to an accompanying editorial and experts interviewed for this article.
“Dupilumab has become such an important first-line systemic medication for our patients with moderate to severe atopic dermatitis. It’s important for us to understand everything we can about its use in the real world – both good and bad,”Raj Chovatiya, MD, PhD, MSCI, assistant professor of dermatology at Northwestern University, Chicago, said in an interview. He was uninvolved with either publication.
Robert Sidbury, MD, MPH, added that, although the affected patient group was small, studying lymphoid reactions associated with dupilumab is important because of the risk for diagnostic misadventure that these reactions carry. He is a professor of pediatrics and division head of dermatology at Seattle Children’s Hospital and the University of Washington, Seattle.
“AD and MF are easily confused for one another at baseline,” explained Dr. Sidbury, who was not involved with the study or editorial. “Dupilumab is known to make AD better and theoretically could help MF via its effect on interleukin (IL)–13, yet case reports of exacerbation and/or unmasking of MF are out there.”
For the study, researchers retrospectively examined records of 530 patients with AD treated with dupilumab at the University Medical Center Utrecht (the Netherlands). Reviewing pretreatment biopsies revealed that among 14 (2.6%) patients who developed clinical suspicion of cutaneous T-cell lymphoma (CTCL) while on treatment, three actually had preexisting MF.
All 14 patients with LR initially responded to dupilumab then developed worsening symptoms at a median of 4 months. Patients reported that the worsening lesions looked and felt different than did previous lesions, with symptoms including burning/pain and an appearance of generalized erythematous maculopapular plaques, sometimes with severe lichenification, on the lower trunk and upper thighs.
The 14 patients’ posttreatment biopsies showed an atypical lymphoid infiltrate with lichenoid or perivascular distribution and intraepithelial T-cell lymphocytes. Whereas patients with MF had hyperconvoluted cerebriform lymphocytes aligned in the epidermal basal layer at the dermoepidermal junction, the 11 with LR had similar-looking lesions dispersed throughout the upper epidermis.
Immunohistochemically, both groups had a dysregulated (mostly increased) CD4:CD8 ratio. CD30 overexpression, usually absent in early-stage MF, affected only patients with LR and one patient with advanced MF. In addition, patients with LR maintained pan–T-cell antigens (CD2, CD3, and CD5), whereas those with MF did not. The 11 patients with LR experienced biopsy-confirmed resolution once they discontinued dupilumab.
It is reassuring that the LRs resolved after dupilumab discontinuation, writes the author of the accompanying editorial, Joan Guitart, MD, chief of dermatopathology at Northwestern University. Nevertheless, he added, such patients deserve “a comprehensive workup including skin biopsy with T-cell receptor clonality assay, blood cell counts with flow cytometry analysis, serum lactate dehydrogenase, and documentation of possible adenopathy, followed with imaging studies and/or local biopsies in cases with abnormal results.”
The possibility that these LRs may represent a first step toward lymphoma requires dermatologists to remain vigilant in ruling out MF, Dr. Guitart wrote, particularly in atypical presentations such as adult-onset AD, cases lacking a history of AD, and cases involving erythrodermic and other uncharacteristic presentations such as plaques, nodules, or spared flexural sites.
For dermatopathologists, Dr. Guitart recommended a cautious approach that resists overdiagnosing MF and acknowledging that insufficient evidence exists to report such reactions as benign. The fact that one study patient had both MF and LR raises concerns that the LR may not always be reversible, Dr. Guitart added.
Clinicians and patients must consider the possibility of dupilumab-induced LR as part of the shared decision-making process and risk-benefit calculus, Dr. Sidbury said. In cases involving unexpected responses or atypical presentations, he added, clinicians must have a low threshold for stopping dupilumab.
For patients who must discontinue dupilumab because of LR, the list of treatment options is growing. “While more investigation is required to understand the role of newer IL-13–blocking biologics and JAK inhibitors among patients experiencing lymphoid reactions,” said Dr. Chovatiya, “traditional atopic dermatitis therapies like narrowband UVB phototherapy and the oral immunosuppressant methotrexate may be reassuring in this population.” Conversely, cyclosporine has been associated with progression of MF.
Also reassuring, said Dr. Sidbury and Dr. Chovatiya, is the rarity of LR overall. Dr. Sidbury said, “The numbers of patients in whom LR or onset/exacerbation of MF occurs is extraordinarily low when compared to those helped immeasurably by dupilumab.”
Dr. Sidbury added that the study and accompanying editorial also will alert clinicians to the potential for newer AD biologics that target solely IL-13 and not IL-4/13, as dupilumab does. “If the deregulated response leading to LR and potentially MF in the affected few is driven by IL-4 inhibition,” he said, “drugs such as tralokinumab (Adbry), lebrikizumab (once approved), and perhaps other newer options might calm AD without causing LRs.”
(Lebrikizumab is not yet approved. In an Oct. 2 press release, Eli Lilly and Company, developer of lebrikizumab, said that it would address issues the U.S. Food and Drug Administration had raised about a third-party manufacturing facility that arose during evaluation of the lebrikizumab biologic license application.)
Study limitations include the fact that most patients who experienced LR had already undergone skin biopsies before dupilumab treatment, which suggests that they had a more atypical AD presentation from the start. The authors add that their having treated all study patients in a tertiary referral hospital indicates a hard-to-treat AD subpopulation.
Study authors reported relationships with several biologic drug manufacturers including Sanofi and Regeneron (dupilumab), LEO Pharma (tralokinumab), and Eli Lilly (lebrikizumab). However, none of these companies provided support for the study.
Dr. Sidbury has been an investigator for Regeneron, Pfizer, and Galderma and a consultant for LEO Pharma and Eli Lilly. Dr. Chovatiya has served as an advisor, consultant, speaker, and investigator for Sanofi and Regeneron. Dr. Guitart reported no conflicts of interest.
A version of this article appeared on Medscape.com.
, according to a study published in JAMA Dermatology
The potential for such reactions requires diagnosing AD carefully, monitoring patients on dupilumab for new and unusual symptoms, and thoroughly working up suspicious LRs, according to an accompanying editorial and experts interviewed for this article.
“Dupilumab has become such an important first-line systemic medication for our patients with moderate to severe atopic dermatitis. It’s important for us to understand everything we can about its use in the real world – both good and bad,”Raj Chovatiya, MD, PhD, MSCI, assistant professor of dermatology at Northwestern University, Chicago, said in an interview. He was uninvolved with either publication.
Robert Sidbury, MD, MPH, added that, although the affected patient group was small, studying lymphoid reactions associated with dupilumab is important because of the risk for diagnostic misadventure that these reactions carry. He is a professor of pediatrics and division head of dermatology at Seattle Children’s Hospital and the University of Washington, Seattle.
“AD and MF are easily confused for one another at baseline,” explained Dr. Sidbury, who was not involved with the study or editorial. “Dupilumab is known to make AD better and theoretically could help MF via its effect on interleukin (IL)–13, yet case reports of exacerbation and/or unmasking of MF are out there.”
For the study, researchers retrospectively examined records of 530 patients with AD treated with dupilumab at the University Medical Center Utrecht (the Netherlands). Reviewing pretreatment biopsies revealed that among 14 (2.6%) patients who developed clinical suspicion of cutaneous T-cell lymphoma (CTCL) while on treatment, three actually had preexisting MF.
All 14 patients with LR initially responded to dupilumab then developed worsening symptoms at a median of 4 months. Patients reported that the worsening lesions looked and felt different than did previous lesions, with symptoms including burning/pain and an appearance of generalized erythematous maculopapular plaques, sometimes with severe lichenification, on the lower trunk and upper thighs.
The 14 patients’ posttreatment biopsies showed an atypical lymphoid infiltrate with lichenoid or perivascular distribution and intraepithelial T-cell lymphocytes. Whereas patients with MF had hyperconvoluted cerebriform lymphocytes aligned in the epidermal basal layer at the dermoepidermal junction, the 11 with LR had similar-looking lesions dispersed throughout the upper epidermis.
Immunohistochemically, both groups had a dysregulated (mostly increased) CD4:CD8 ratio. CD30 overexpression, usually absent in early-stage MF, affected only patients with LR and one patient with advanced MF. In addition, patients with LR maintained pan–T-cell antigens (CD2, CD3, and CD5), whereas those with MF did not. The 11 patients with LR experienced biopsy-confirmed resolution once they discontinued dupilumab.
It is reassuring that the LRs resolved after dupilumab discontinuation, writes the author of the accompanying editorial, Joan Guitart, MD, chief of dermatopathology at Northwestern University. Nevertheless, he added, such patients deserve “a comprehensive workup including skin biopsy with T-cell receptor clonality assay, blood cell counts with flow cytometry analysis, serum lactate dehydrogenase, and documentation of possible adenopathy, followed with imaging studies and/or local biopsies in cases with abnormal results.”
The possibility that these LRs may represent a first step toward lymphoma requires dermatologists to remain vigilant in ruling out MF, Dr. Guitart wrote, particularly in atypical presentations such as adult-onset AD, cases lacking a history of AD, and cases involving erythrodermic and other uncharacteristic presentations such as plaques, nodules, or spared flexural sites.
For dermatopathologists, Dr. Guitart recommended a cautious approach that resists overdiagnosing MF and acknowledging that insufficient evidence exists to report such reactions as benign. The fact that one study patient had both MF and LR raises concerns that the LR may not always be reversible, Dr. Guitart added.
Clinicians and patients must consider the possibility of dupilumab-induced LR as part of the shared decision-making process and risk-benefit calculus, Dr. Sidbury said. In cases involving unexpected responses or atypical presentations, he added, clinicians must have a low threshold for stopping dupilumab.
For patients who must discontinue dupilumab because of LR, the list of treatment options is growing. “While more investigation is required to understand the role of newer IL-13–blocking biologics and JAK inhibitors among patients experiencing lymphoid reactions,” said Dr. Chovatiya, “traditional atopic dermatitis therapies like narrowband UVB phototherapy and the oral immunosuppressant methotrexate may be reassuring in this population.” Conversely, cyclosporine has been associated with progression of MF.
Also reassuring, said Dr. Sidbury and Dr. Chovatiya, is the rarity of LR overall. Dr. Sidbury said, “The numbers of patients in whom LR or onset/exacerbation of MF occurs is extraordinarily low when compared to those helped immeasurably by dupilumab.”
Dr. Sidbury added that the study and accompanying editorial also will alert clinicians to the potential for newer AD biologics that target solely IL-13 and not IL-4/13, as dupilumab does. “If the deregulated response leading to LR and potentially MF in the affected few is driven by IL-4 inhibition,” he said, “drugs such as tralokinumab (Adbry), lebrikizumab (once approved), and perhaps other newer options might calm AD without causing LRs.”
(Lebrikizumab is not yet approved. In an Oct. 2 press release, Eli Lilly and Company, developer of lebrikizumab, said that it would address issues the U.S. Food and Drug Administration had raised about a third-party manufacturing facility that arose during evaluation of the lebrikizumab biologic license application.)
Study limitations include the fact that most patients who experienced LR had already undergone skin biopsies before dupilumab treatment, which suggests that they had a more atypical AD presentation from the start. The authors add that their having treated all study patients in a tertiary referral hospital indicates a hard-to-treat AD subpopulation.
Study authors reported relationships with several biologic drug manufacturers including Sanofi and Regeneron (dupilumab), LEO Pharma (tralokinumab), and Eli Lilly (lebrikizumab). However, none of these companies provided support for the study.
Dr. Sidbury has been an investigator for Regeneron, Pfizer, and Galderma and a consultant for LEO Pharma and Eli Lilly. Dr. Chovatiya has served as an advisor, consultant, speaker, and investigator for Sanofi and Regeneron. Dr. Guitart reported no conflicts of interest.
A version of this article appeared on Medscape.com.
, according to a study published in JAMA Dermatology
The potential for such reactions requires diagnosing AD carefully, monitoring patients on dupilumab for new and unusual symptoms, and thoroughly working up suspicious LRs, according to an accompanying editorial and experts interviewed for this article.
“Dupilumab has become such an important first-line systemic medication for our patients with moderate to severe atopic dermatitis. It’s important for us to understand everything we can about its use in the real world – both good and bad,”Raj Chovatiya, MD, PhD, MSCI, assistant professor of dermatology at Northwestern University, Chicago, said in an interview. He was uninvolved with either publication.
Robert Sidbury, MD, MPH, added that, although the affected patient group was small, studying lymphoid reactions associated with dupilumab is important because of the risk for diagnostic misadventure that these reactions carry. He is a professor of pediatrics and division head of dermatology at Seattle Children’s Hospital and the University of Washington, Seattle.
“AD and MF are easily confused for one another at baseline,” explained Dr. Sidbury, who was not involved with the study or editorial. “Dupilumab is known to make AD better and theoretically could help MF via its effect on interleukin (IL)–13, yet case reports of exacerbation and/or unmasking of MF are out there.”
For the study, researchers retrospectively examined records of 530 patients with AD treated with dupilumab at the University Medical Center Utrecht (the Netherlands). Reviewing pretreatment biopsies revealed that among 14 (2.6%) patients who developed clinical suspicion of cutaneous T-cell lymphoma (CTCL) while on treatment, three actually had preexisting MF.
All 14 patients with LR initially responded to dupilumab then developed worsening symptoms at a median of 4 months. Patients reported that the worsening lesions looked and felt different than did previous lesions, with symptoms including burning/pain and an appearance of generalized erythematous maculopapular plaques, sometimes with severe lichenification, on the lower trunk and upper thighs.
The 14 patients’ posttreatment biopsies showed an atypical lymphoid infiltrate with lichenoid or perivascular distribution and intraepithelial T-cell lymphocytes. Whereas patients with MF had hyperconvoluted cerebriform lymphocytes aligned in the epidermal basal layer at the dermoepidermal junction, the 11 with LR had similar-looking lesions dispersed throughout the upper epidermis.
Immunohistochemically, both groups had a dysregulated (mostly increased) CD4:CD8 ratio. CD30 overexpression, usually absent in early-stage MF, affected only patients with LR and one patient with advanced MF. In addition, patients with LR maintained pan–T-cell antigens (CD2, CD3, and CD5), whereas those with MF did not. The 11 patients with LR experienced biopsy-confirmed resolution once they discontinued dupilumab.
It is reassuring that the LRs resolved after dupilumab discontinuation, writes the author of the accompanying editorial, Joan Guitart, MD, chief of dermatopathology at Northwestern University. Nevertheless, he added, such patients deserve “a comprehensive workup including skin biopsy with T-cell receptor clonality assay, blood cell counts with flow cytometry analysis, serum lactate dehydrogenase, and documentation of possible adenopathy, followed with imaging studies and/or local biopsies in cases with abnormal results.”
The possibility that these LRs may represent a first step toward lymphoma requires dermatologists to remain vigilant in ruling out MF, Dr. Guitart wrote, particularly in atypical presentations such as adult-onset AD, cases lacking a history of AD, and cases involving erythrodermic and other uncharacteristic presentations such as plaques, nodules, or spared flexural sites.
For dermatopathologists, Dr. Guitart recommended a cautious approach that resists overdiagnosing MF and acknowledging that insufficient evidence exists to report such reactions as benign. The fact that one study patient had both MF and LR raises concerns that the LR may not always be reversible, Dr. Guitart added.
Clinicians and patients must consider the possibility of dupilumab-induced LR as part of the shared decision-making process and risk-benefit calculus, Dr. Sidbury said. In cases involving unexpected responses or atypical presentations, he added, clinicians must have a low threshold for stopping dupilumab.
For patients who must discontinue dupilumab because of LR, the list of treatment options is growing. “While more investigation is required to understand the role of newer IL-13–blocking biologics and JAK inhibitors among patients experiencing lymphoid reactions,” said Dr. Chovatiya, “traditional atopic dermatitis therapies like narrowband UVB phototherapy and the oral immunosuppressant methotrexate may be reassuring in this population.” Conversely, cyclosporine has been associated with progression of MF.
Also reassuring, said Dr. Sidbury and Dr. Chovatiya, is the rarity of LR overall. Dr. Sidbury said, “The numbers of patients in whom LR or onset/exacerbation of MF occurs is extraordinarily low when compared to those helped immeasurably by dupilumab.”
Dr. Sidbury added that the study and accompanying editorial also will alert clinicians to the potential for newer AD biologics that target solely IL-13 and not IL-4/13, as dupilumab does. “If the deregulated response leading to LR and potentially MF in the affected few is driven by IL-4 inhibition,” he said, “drugs such as tralokinumab (Adbry), lebrikizumab (once approved), and perhaps other newer options might calm AD without causing LRs.”
(Lebrikizumab is not yet approved. In an Oct. 2 press release, Eli Lilly and Company, developer of lebrikizumab, said that it would address issues the U.S. Food and Drug Administration had raised about a third-party manufacturing facility that arose during evaluation of the lebrikizumab biologic license application.)
Study limitations include the fact that most patients who experienced LR had already undergone skin biopsies before dupilumab treatment, which suggests that they had a more atypical AD presentation from the start. The authors add that their having treated all study patients in a tertiary referral hospital indicates a hard-to-treat AD subpopulation.
Study authors reported relationships with several biologic drug manufacturers including Sanofi and Regeneron (dupilumab), LEO Pharma (tralokinumab), and Eli Lilly (lebrikizumab). However, none of these companies provided support for the study.
Dr. Sidbury has been an investigator for Regeneron, Pfizer, and Galderma and a consultant for LEO Pharma and Eli Lilly. Dr. Chovatiya has served as an advisor, consultant, speaker, and investigator for Sanofi and Regeneron. Dr. Guitart reported no conflicts of interest.
A version of this article appeared on Medscape.com.
FROM JAMA DERMATOLOGY
More phase 3 data support use of nemolizumab for prurigo nodularis
reported at the annual Congress of the European Academy of Dermatology and Venereology.
In the OLYMPIA 1 study, clinically significant improvements in both itch and skin lesions were seen after 16 weeks of treatment with nemolizumab compared with placebo (P < .0001).
Indeed, among the 286 patients who participated in the trial (190 on nemolizumab and 96 on placebo), 58.4% of those treated with nemolizumab and 16.7% of those who received placebo had an improvement of 4 points or more in the weekly average peak pruritus numeric rating scale (PP-NRS) at week 16 (P < .0001).
Skin lesions were assessed using an investigators general assessment (IGA) score, where IGA success was defined as a score of 0/1 indicating clear or almost clear skin or where there had been at least a 2-point change from baseline values. Over a quarter (26.3%) of nemolizumab-treated patients met these criteria versus 7.3% for those on placebo (P = .0001).
“These results confirm the results of the OLYMPIA 2 study, the other phase 3 study, and now I hope you understand why we are so excited,” lead investigator Sonja Ständer, MD, of the Center for Chronic Pruritus at University Hospital Münster, Germany, said at the meeting, where she presented the data.
The OLYMPIA 2 study included 274 patients and the results showed a weekly average PP-NRS score improvement of 56.3% vs. 20.9% for placebo and IGA success in 37.7% and 11% of patients, respectively, at 16 weeks.
First-in-class therapy
“We know how difficult it is to treat patients; they are refractory to treatment, frustrated, and this really impacts them regarding their quality of life,” said Dr. Ständer. New options are needed to help patients, and nemolizumab, a first-in-class interleukin-31 (IL-31) receptor alpha antagonist, is one treatment that may answer this call.
Prurigo nodularis is a chronic neuroimmune skin condition characterized by severe itch and multiple nodular skin lesions, Dr. Ständer explained. She added that there is evidence that IL-31 has a key role to play in the development of itch, and in differentiation of keratinocytes, type 2 and type 17 immune responses, and fibrosis associated with the condition.
The OLYMPIA 1 and 2 trials are part of a large developmental program that includes two ongoing trials. One is assessing the durability of response over 24 weeks in 40 patients and the other is a long-term extension trial involving 450 patients from the OLYMPIA 1 and 2 trials.
Inclusion criteria and additional results
For inclusion in the study, adults with prurigo nodularis for at least 6 months had to have 20 or more nodules on the body with a bilateral distribution, an IGA score of 3 or more, and an average PP-NRS of 7 or higher. The latter “was really a high bar for them to qualify for the trial,” said Dr. Ständer.
After an initial 4-week screening period, patients were randomly assigned to 24 weeks of treatment with nemolizumab or placebo given as a subcutaneous injection every 4 weeks. An 8-week “off-treatment” period followed.
The nemolizumab dose was based on the patient’s body weight, with patients weighing less than 90 kg (198 pounds) getting a loading dose of 60 mg followed by further doses of 30 mg; while patients weighing 90 kg or more receiving 50 mg of nemolizumab.
Dr. Ständer reported that nemolizumab met all of the trials’ secondary endpoints; this included at least a 4-point improvement in sleep disturbance. She noted that changes in itch and subsequent sleep disturbance occurred early, at 4 weeks of treatment – after just one injection of nemolizumab.
The response rates seen in the moderate to severe prurigo nodularis population studies are quite unique when compared with conventional therapies, Dr. Ständer maintained. “We’ve never seen something like this before.”
No safety concerns
No significant difference in tolerability was seen between the nemolizumab and placebo groups, Dr. Ständer observed. Any adverse event occurred in 71.7% and 65.3% of patients, respectively, and serious adverse events in 8.6% and 10.5%.
There was a similar rate of adverse events leading to discontinuation, respectively (4.8% vs. 4.2%).
Headache was seen more frequently among those on nemolizumab than those on placebo (7.0% vs. 2.1%), and there was a higher number of eczema cases among those on nemolizumab (5.3% vs. 1.1%). The latter is somewhat paradoxical because nemolizumab is also being studied as a treatment for atopic dermatitis, with good results seen in phase 3 trials. Asked about this finding after her presentation, Dr. Ständer said “we are following up on that to know exactly what is going on; this is a side effect of nemolizumab that is seen also with other biologics.”
JAK inhibitor trial for PN, CPUO
Nemolizumab is not the only promising new approach to treating prurigo nodularis. During a separate late-breaking news session at the meeting, Shawn Kwatra, MD, director of the Johns Hopkins Itch Center in Baltimore, presented “dramatic” data from a “proof-of-concept” phase 2 study with the Janus kinase (JAK) inhibitor abrocitinib (Cibinqo), which is approved for atopic dermatitis in the United States and Europe.
The investigator-initiated trial took a different approach from most other trials, Dr. Kwatra said. The starting point was to look at studying multiple rather than single dermatologic diseases that were perhaps being left a little by the wayside but may share some common ground. Those two diseases were prurigo nodularis and chronic pruritus of unknown origin (CPUO).
“They’re actually very analogous conditions in the way we treat, so I thought those would be a good pair,” Dr. Kwatra said, noting that there were several studies that made him think that JAK inhibition “would be an interesting concept to try.”
On that basis, 10 women with prurigo nodularis (mean age, 58 years) and two women and eight men with CPUO (mean age, 70 years) were recruited and all were treated with abrocitinib at a once-daily oral dose of 200 mg for 12 weeks.
“They all had really intense itch,” before treatment, Dr. Kwatra said. The mean baseline PP-NRS was 9.2 and 8.2 in the prurigo nodularis and CPUO groups, respectively. By the end of treatment, however, “the improvement in itch was pretty dramatic,” especially for prurigo nodularis, he said.
At 12 weeks, the PP-NRS score had fallen to 2.0 in the prurigo nodularis group, equating to a significant 78% change from baseline (P < .001). And, in the CPUO group, the 12-week PP-NRS score was 3.8, nearly a 54% drop from baseline (P = .01).
Sleep disturbance was improved for both conditions, and in the patients with prurigo nodularis, there were improvements in skin lesions. Looking at the patients who responded to treatment, Dr. Kwatra noted that “if you responded, you respond fast, and you respond almost entirely.”
Additional findings from cutaneous transcriptome analysis showed that JAK inhibition with abrocitinib was modulating Th1-, Th2-, Th17-, and Th22-mediated pathways in both groups of patients.
The overall frequency of adverse events was low, and no serious adverse events occurred.
Commenting on the potential use of abrocitinib in managing patients with PN and CPUO, Tiago dos Reis Matos, MD, PhD, MSc, Amsterdam University Medical Centers, told this news organization that JAK1 inhibitors “are showing promising results in treating several diseases.”
Dr. Matos, who was not involved in the study, added that JAK inhibition was “of special interest in prurigo nodularis and chronic pruritus, since these are some of the most difficult diseases to treat with limited therapeutic options.”
Dr. Kwatra observed: “Obviously, we need further development. But we also have clues here about how to design phase 3 trials.”
Galderma funded the OLYMPIA 1 and 2 studies. Dr. Ständer was an investigator for the trial and reported serving as a consultant, speaker, or investigator for multiple pharmaceutical companies, including Galderma.
Johns Hopkins University supported the abrocitinib study with funding from Pfizer. Dr. Kwatra is an advisory board member or consultant to several pharmaceutical companies and is an investigator for Galderma, Incyte, Pfizer, and Sanofi.
A version of this article first appeared on Medscape.com.
reported at the annual Congress of the European Academy of Dermatology and Venereology.
In the OLYMPIA 1 study, clinically significant improvements in both itch and skin lesions were seen after 16 weeks of treatment with nemolizumab compared with placebo (P < .0001).
Indeed, among the 286 patients who participated in the trial (190 on nemolizumab and 96 on placebo), 58.4% of those treated with nemolizumab and 16.7% of those who received placebo had an improvement of 4 points or more in the weekly average peak pruritus numeric rating scale (PP-NRS) at week 16 (P < .0001).
Skin lesions were assessed using an investigators general assessment (IGA) score, where IGA success was defined as a score of 0/1 indicating clear or almost clear skin or where there had been at least a 2-point change from baseline values. Over a quarter (26.3%) of nemolizumab-treated patients met these criteria versus 7.3% for those on placebo (P = .0001).
“These results confirm the results of the OLYMPIA 2 study, the other phase 3 study, and now I hope you understand why we are so excited,” lead investigator Sonja Ständer, MD, of the Center for Chronic Pruritus at University Hospital Münster, Germany, said at the meeting, where she presented the data.
The OLYMPIA 2 study included 274 patients and the results showed a weekly average PP-NRS score improvement of 56.3% vs. 20.9% for placebo and IGA success in 37.7% and 11% of patients, respectively, at 16 weeks.
First-in-class therapy
“We know how difficult it is to treat patients; they are refractory to treatment, frustrated, and this really impacts them regarding their quality of life,” said Dr. Ständer. New options are needed to help patients, and nemolizumab, a first-in-class interleukin-31 (IL-31) receptor alpha antagonist, is one treatment that may answer this call.
Prurigo nodularis is a chronic neuroimmune skin condition characterized by severe itch and multiple nodular skin lesions, Dr. Ständer explained. She added that there is evidence that IL-31 has a key role to play in the development of itch, and in differentiation of keratinocytes, type 2 and type 17 immune responses, and fibrosis associated with the condition.
The OLYMPIA 1 and 2 trials are part of a large developmental program that includes two ongoing trials. One is assessing the durability of response over 24 weeks in 40 patients and the other is a long-term extension trial involving 450 patients from the OLYMPIA 1 and 2 trials.
Inclusion criteria and additional results
For inclusion in the study, adults with prurigo nodularis for at least 6 months had to have 20 or more nodules on the body with a bilateral distribution, an IGA score of 3 or more, and an average PP-NRS of 7 or higher. The latter “was really a high bar for them to qualify for the trial,” said Dr. Ständer.
After an initial 4-week screening period, patients were randomly assigned to 24 weeks of treatment with nemolizumab or placebo given as a subcutaneous injection every 4 weeks. An 8-week “off-treatment” period followed.
The nemolizumab dose was based on the patient’s body weight, with patients weighing less than 90 kg (198 pounds) getting a loading dose of 60 mg followed by further doses of 30 mg; while patients weighing 90 kg or more receiving 50 mg of nemolizumab.
Dr. Ständer reported that nemolizumab met all of the trials’ secondary endpoints; this included at least a 4-point improvement in sleep disturbance. She noted that changes in itch and subsequent sleep disturbance occurred early, at 4 weeks of treatment – after just one injection of nemolizumab.
The response rates seen in the moderate to severe prurigo nodularis population studies are quite unique when compared with conventional therapies, Dr. Ständer maintained. “We’ve never seen something like this before.”
No safety concerns
No significant difference in tolerability was seen between the nemolizumab and placebo groups, Dr. Ständer observed. Any adverse event occurred in 71.7% and 65.3% of patients, respectively, and serious adverse events in 8.6% and 10.5%.
There was a similar rate of adverse events leading to discontinuation, respectively (4.8% vs. 4.2%).
Headache was seen more frequently among those on nemolizumab than those on placebo (7.0% vs. 2.1%), and there was a higher number of eczema cases among those on nemolizumab (5.3% vs. 1.1%). The latter is somewhat paradoxical because nemolizumab is also being studied as a treatment for atopic dermatitis, with good results seen in phase 3 trials. Asked about this finding after her presentation, Dr. Ständer said “we are following up on that to know exactly what is going on; this is a side effect of nemolizumab that is seen also with other biologics.”
JAK inhibitor trial for PN, CPUO
Nemolizumab is not the only promising new approach to treating prurigo nodularis. During a separate late-breaking news session at the meeting, Shawn Kwatra, MD, director of the Johns Hopkins Itch Center in Baltimore, presented “dramatic” data from a “proof-of-concept” phase 2 study with the Janus kinase (JAK) inhibitor abrocitinib (Cibinqo), which is approved for atopic dermatitis in the United States and Europe.
The investigator-initiated trial took a different approach from most other trials, Dr. Kwatra said. The starting point was to look at studying multiple rather than single dermatologic diseases that were perhaps being left a little by the wayside but may share some common ground. Those two diseases were prurigo nodularis and chronic pruritus of unknown origin (CPUO).
“They’re actually very analogous conditions in the way we treat, so I thought those would be a good pair,” Dr. Kwatra said, noting that there were several studies that made him think that JAK inhibition “would be an interesting concept to try.”
On that basis, 10 women with prurigo nodularis (mean age, 58 years) and two women and eight men with CPUO (mean age, 70 years) were recruited and all were treated with abrocitinib at a once-daily oral dose of 200 mg for 12 weeks.
“They all had really intense itch,” before treatment, Dr. Kwatra said. The mean baseline PP-NRS was 9.2 and 8.2 in the prurigo nodularis and CPUO groups, respectively. By the end of treatment, however, “the improvement in itch was pretty dramatic,” especially for prurigo nodularis, he said.
At 12 weeks, the PP-NRS score had fallen to 2.0 in the prurigo nodularis group, equating to a significant 78% change from baseline (P < .001). And, in the CPUO group, the 12-week PP-NRS score was 3.8, nearly a 54% drop from baseline (P = .01).
Sleep disturbance was improved for both conditions, and in the patients with prurigo nodularis, there were improvements in skin lesions. Looking at the patients who responded to treatment, Dr. Kwatra noted that “if you responded, you respond fast, and you respond almost entirely.”
Additional findings from cutaneous transcriptome analysis showed that JAK inhibition with abrocitinib was modulating Th1-, Th2-, Th17-, and Th22-mediated pathways in both groups of patients.
The overall frequency of adverse events was low, and no serious adverse events occurred.
Commenting on the potential use of abrocitinib in managing patients with PN and CPUO, Tiago dos Reis Matos, MD, PhD, MSc, Amsterdam University Medical Centers, told this news organization that JAK1 inhibitors “are showing promising results in treating several diseases.”
Dr. Matos, who was not involved in the study, added that JAK inhibition was “of special interest in prurigo nodularis and chronic pruritus, since these are some of the most difficult diseases to treat with limited therapeutic options.”
Dr. Kwatra observed: “Obviously, we need further development. But we also have clues here about how to design phase 3 trials.”
Galderma funded the OLYMPIA 1 and 2 studies. Dr. Ständer was an investigator for the trial and reported serving as a consultant, speaker, or investigator for multiple pharmaceutical companies, including Galderma.
Johns Hopkins University supported the abrocitinib study with funding from Pfizer. Dr. Kwatra is an advisory board member or consultant to several pharmaceutical companies and is an investigator for Galderma, Incyte, Pfizer, and Sanofi.
A version of this article first appeared on Medscape.com.
reported at the annual Congress of the European Academy of Dermatology and Venereology.
In the OLYMPIA 1 study, clinically significant improvements in both itch and skin lesions were seen after 16 weeks of treatment with nemolizumab compared with placebo (P < .0001).
Indeed, among the 286 patients who participated in the trial (190 on nemolizumab and 96 on placebo), 58.4% of those treated with nemolizumab and 16.7% of those who received placebo had an improvement of 4 points or more in the weekly average peak pruritus numeric rating scale (PP-NRS) at week 16 (P < .0001).
Skin lesions were assessed using an investigators general assessment (IGA) score, where IGA success was defined as a score of 0/1 indicating clear or almost clear skin or where there had been at least a 2-point change from baseline values. Over a quarter (26.3%) of nemolizumab-treated patients met these criteria versus 7.3% for those on placebo (P = .0001).
“These results confirm the results of the OLYMPIA 2 study, the other phase 3 study, and now I hope you understand why we are so excited,” lead investigator Sonja Ständer, MD, of the Center for Chronic Pruritus at University Hospital Münster, Germany, said at the meeting, where she presented the data.
The OLYMPIA 2 study included 274 patients and the results showed a weekly average PP-NRS score improvement of 56.3% vs. 20.9% for placebo and IGA success in 37.7% and 11% of patients, respectively, at 16 weeks.
First-in-class therapy
“We know how difficult it is to treat patients; they are refractory to treatment, frustrated, and this really impacts them regarding their quality of life,” said Dr. Ständer. New options are needed to help patients, and nemolizumab, a first-in-class interleukin-31 (IL-31) receptor alpha antagonist, is one treatment that may answer this call.
Prurigo nodularis is a chronic neuroimmune skin condition characterized by severe itch and multiple nodular skin lesions, Dr. Ständer explained. She added that there is evidence that IL-31 has a key role to play in the development of itch, and in differentiation of keratinocytes, type 2 and type 17 immune responses, and fibrosis associated with the condition.
The OLYMPIA 1 and 2 trials are part of a large developmental program that includes two ongoing trials. One is assessing the durability of response over 24 weeks in 40 patients and the other is a long-term extension trial involving 450 patients from the OLYMPIA 1 and 2 trials.
Inclusion criteria and additional results
For inclusion in the study, adults with prurigo nodularis for at least 6 months had to have 20 or more nodules on the body with a bilateral distribution, an IGA score of 3 or more, and an average PP-NRS of 7 or higher. The latter “was really a high bar for them to qualify for the trial,” said Dr. Ständer.
After an initial 4-week screening period, patients were randomly assigned to 24 weeks of treatment with nemolizumab or placebo given as a subcutaneous injection every 4 weeks. An 8-week “off-treatment” period followed.
The nemolizumab dose was based on the patient’s body weight, with patients weighing less than 90 kg (198 pounds) getting a loading dose of 60 mg followed by further doses of 30 mg; while patients weighing 90 kg or more receiving 50 mg of nemolizumab.
Dr. Ständer reported that nemolizumab met all of the trials’ secondary endpoints; this included at least a 4-point improvement in sleep disturbance. She noted that changes in itch and subsequent sleep disturbance occurred early, at 4 weeks of treatment – after just one injection of nemolizumab.
The response rates seen in the moderate to severe prurigo nodularis population studies are quite unique when compared with conventional therapies, Dr. Ständer maintained. “We’ve never seen something like this before.”
No safety concerns
No significant difference in tolerability was seen between the nemolizumab and placebo groups, Dr. Ständer observed. Any adverse event occurred in 71.7% and 65.3% of patients, respectively, and serious adverse events in 8.6% and 10.5%.
There was a similar rate of adverse events leading to discontinuation, respectively (4.8% vs. 4.2%).
Headache was seen more frequently among those on nemolizumab than those on placebo (7.0% vs. 2.1%), and there was a higher number of eczema cases among those on nemolizumab (5.3% vs. 1.1%). The latter is somewhat paradoxical because nemolizumab is also being studied as a treatment for atopic dermatitis, with good results seen in phase 3 trials. Asked about this finding after her presentation, Dr. Ständer said “we are following up on that to know exactly what is going on; this is a side effect of nemolizumab that is seen also with other biologics.”
JAK inhibitor trial for PN, CPUO
Nemolizumab is not the only promising new approach to treating prurigo nodularis. During a separate late-breaking news session at the meeting, Shawn Kwatra, MD, director of the Johns Hopkins Itch Center in Baltimore, presented “dramatic” data from a “proof-of-concept” phase 2 study with the Janus kinase (JAK) inhibitor abrocitinib (Cibinqo), which is approved for atopic dermatitis in the United States and Europe.
The investigator-initiated trial took a different approach from most other trials, Dr. Kwatra said. The starting point was to look at studying multiple rather than single dermatologic diseases that were perhaps being left a little by the wayside but may share some common ground. Those two diseases were prurigo nodularis and chronic pruritus of unknown origin (CPUO).
“They’re actually very analogous conditions in the way we treat, so I thought those would be a good pair,” Dr. Kwatra said, noting that there were several studies that made him think that JAK inhibition “would be an interesting concept to try.”
On that basis, 10 women with prurigo nodularis (mean age, 58 years) and two women and eight men with CPUO (mean age, 70 years) were recruited and all were treated with abrocitinib at a once-daily oral dose of 200 mg for 12 weeks.
“They all had really intense itch,” before treatment, Dr. Kwatra said. The mean baseline PP-NRS was 9.2 and 8.2 in the prurigo nodularis and CPUO groups, respectively. By the end of treatment, however, “the improvement in itch was pretty dramatic,” especially for prurigo nodularis, he said.
At 12 weeks, the PP-NRS score had fallen to 2.0 in the prurigo nodularis group, equating to a significant 78% change from baseline (P < .001). And, in the CPUO group, the 12-week PP-NRS score was 3.8, nearly a 54% drop from baseline (P = .01).
Sleep disturbance was improved for both conditions, and in the patients with prurigo nodularis, there were improvements in skin lesions. Looking at the patients who responded to treatment, Dr. Kwatra noted that “if you responded, you respond fast, and you respond almost entirely.”
Additional findings from cutaneous transcriptome analysis showed that JAK inhibition with abrocitinib was modulating Th1-, Th2-, Th17-, and Th22-mediated pathways in both groups of patients.
The overall frequency of adverse events was low, and no serious adverse events occurred.
Commenting on the potential use of abrocitinib in managing patients with PN and CPUO, Tiago dos Reis Matos, MD, PhD, MSc, Amsterdam University Medical Centers, told this news organization that JAK1 inhibitors “are showing promising results in treating several diseases.”
Dr. Matos, who was not involved in the study, added that JAK inhibition was “of special interest in prurigo nodularis and chronic pruritus, since these are some of the most difficult diseases to treat with limited therapeutic options.”
Dr. Kwatra observed: “Obviously, we need further development. But we also have clues here about how to design phase 3 trials.”
Galderma funded the OLYMPIA 1 and 2 studies. Dr. Ständer was an investigator for the trial and reported serving as a consultant, speaker, or investigator for multiple pharmaceutical companies, including Galderma.
Johns Hopkins University supported the abrocitinib study with funding from Pfizer. Dr. Kwatra is an advisory board member or consultant to several pharmaceutical companies and is an investigator for Galderma, Incyte, Pfizer, and Sanofi.
A version of this article first appeared on Medscape.com.
FROM THE EADV CONGRESS
Birch bark–derived treatment reduces daily dressings in patients with epidermolysis bullosa
Additional when compared with a control gel.
In a final, post hoc analysis to come from the trial, 15 of 45 (33%) patients treated with Oleogel-S10 versus 5 of 48 (10.4%) treated with the control gel were reported as no longer needing daily dressing changes at 45 days of follow-up.
Moreover, the effect was sustained, with similar percentages of patients no longer requiring daily dressing changes at 60 days (34% vs. 13%, respectively) and 90 days (36% vs. 11%) of follow-up.
The mean reduction in daily dressing changes was 1.36 for Oleogel-S10 and 0.41 for the control gel (P = .005).
“Patients who, in the beginning, had daily dressing changes had almost three fewer dressing changes every 2 weeks if they were treated with Oleogel-S10,” Dimitra Kiritsi, MD, PhD, of the department of dermatology at the University of Freiburg (Germany), reported at the annual congress of the European Academy of Dermatology and Venereology. By comparison, patients in the control group had just one fewer daily dressing change in 2 weeks.
“You might say okay, but what does this mean in terms of time?” added Dr. Kiritsi. Using historical data on the time required for whole body care (Orphanet J Rare Dis. 2020 Jan 3. doi: 10.1186/s13023-019-1279-y), it was estimated that treatment with Oleogel-S10 was associated with an overall time-saving per week of 11 hours (6.6 hours for the patient and 4.4 hours for the caregiver) and use of the control gel was associated with an overall time-saving of 4 hours (2.4 hours for the patient and 1.6 hours for the caregiver).
“This is, for our patients, important,” said Dr. Kiritsi, as “it is time that they can spend doing something nice with the family” instead, avoiding the pain and distress associated with frequent dressing changes.
Approved in Europe, not in the United States
Oleogel-S10, classified as an herbal product, contains triterpenes derived from birch bark extract, which have been formulated with sunflower oil to form a gel.
Despite being approved for use in Europe, Oleogel-S10 has not yet been approved to treat EB in the United States. The FDA did not approve Amryt Pharma’s new drug application in February 2022. The application had included data from the EASE trial.
EASE included 223 patients with dystrophic or junctional EB, including 156 children, at 58 sites in 28 countries. As such, this makes it the largest treatment study in this rare genetic disease to date.
The trial had consisted of an initial 90-day, double-blind treatment period, during which time 109 patients had used Oleogel-S10 and 114 had used a control gel. This was followed by a 24-month open-label phase, during which time all remaining patients (n = 205) had used Oleogel-S10 on top of their standard of care.
Dr. Kiritsi summarized the main results of the EASE trial as follows.
- Complete healing of target wounds (primary endpoint) in 41.3% of patients treated with Oleogel-S10 and 28.9% of patients treated with the control gel (P = .013).
- Improved total body wound burden measured by both Epidermolysis Bullosa Disease Activity and Scarring Index and Body Surface Area Percentage scores.
- Reduced frequency of dressing changes (1 less per 2 weeks for Oleogel-S10 versus 0 less per 2 weeks for control gel).
- Improved pain among participants aged 4 years and older while their dressings were being changed.
- Reduced rates of wound infection (0.9% Oleogel-S10 vs. 4.4% control gel).
- Similar rates of treatment-emergent adverse events (24.8% vs. 22.8%, respectively), which were mostly deemed to be mild or moderate.
The EASE study – an important trial for EB
EASE is an important trial for EB, the study’s principal investigator Dédée Murrell, MD, DSc, University of New South Wales, Sydney, has pointed out previously.
“This was the first EB study to meet its primary endpoint and demonstrated a statistically significant acceleration of target wound healing by day 45,” Dr. Murrell said in a press release issued by Amryt Pharma to coincide with the online publication of the trial results.
“In addition, the favorable trends we see with key secondary endpoints such as reduced wound burden, pain, and frequency of dressing changes are considered as being very meaningful for patients,” Dr. Murrell said.
The EASE study was funded by Amryt Research Limited. Dr. Kiritsi reported receiving honoraria or consultation fees from Amryt, RHEACELL GmbH, and Fibrx Derm. She also acknowledged grant or research support from DEBRA International, EB Research Partnership, Fritz-Thyssen Foundation, German Research Foundation, and RHEACELL. Dr. Murrell has ties to Amryt and Amicus and is a co-owner of the patent for topical sirolimus for EB simplex.
A version of this article appeared on Medscape.com.
Additional when compared with a control gel.
In a final, post hoc analysis to come from the trial, 15 of 45 (33%) patients treated with Oleogel-S10 versus 5 of 48 (10.4%) treated with the control gel were reported as no longer needing daily dressing changes at 45 days of follow-up.
Moreover, the effect was sustained, with similar percentages of patients no longer requiring daily dressing changes at 60 days (34% vs. 13%, respectively) and 90 days (36% vs. 11%) of follow-up.
The mean reduction in daily dressing changes was 1.36 for Oleogel-S10 and 0.41 for the control gel (P = .005).
“Patients who, in the beginning, had daily dressing changes had almost three fewer dressing changes every 2 weeks if they were treated with Oleogel-S10,” Dimitra Kiritsi, MD, PhD, of the department of dermatology at the University of Freiburg (Germany), reported at the annual congress of the European Academy of Dermatology and Venereology. By comparison, patients in the control group had just one fewer daily dressing change in 2 weeks.
“You might say okay, but what does this mean in terms of time?” added Dr. Kiritsi. Using historical data on the time required for whole body care (Orphanet J Rare Dis. 2020 Jan 3. doi: 10.1186/s13023-019-1279-y), it was estimated that treatment with Oleogel-S10 was associated with an overall time-saving per week of 11 hours (6.6 hours for the patient and 4.4 hours for the caregiver) and use of the control gel was associated with an overall time-saving of 4 hours (2.4 hours for the patient and 1.6 hours for the caregiver).
“This is, for our patients, important,” said Dr. Kiritsi, as “it is time that they can spend doing something nice with the family” instead, avoiding the pain and distress associated with frequent dressing changes.
Approved in Europe, not in the United States
Oleogel-S10, classified as an herbal product, contains triterpenes derived from birch bark extract, which have been formulated with sunflower oil to form a gel.
Despite being approved for use in Europe, Oleogel-S10 has not yet been approved to treat EB in the United States. The FDA did not approve Amryt Pharma’s new drug application in February 2022. The application had included data from the EASE trial.
EASE included 223 patients with dystrophic or junctional EB, including 156 children, at 58 sites in 28 countries. As such, this makes it the largest treatment study in this rare genetic disease to date.
The trial had consisted of an initial 90-day, double-blind treatment period, during which time 109 patients had used Oleogel-S10 and 114 had used a control gel. This was followed by a 24-month open-label phase, during which time all remaining patients (n = 205) had used Oleogel-S10 on top of their standard of care.
Dr. Kiritsi summarized the main results of the EASE trial as follows.
- Complete healing of target wounds (primary endpoint) in 41.3% of patients treated with Oleogel-S10 and 28.9% of patients treated with the control gel (P = .013).
- Improved total body wound burden measured by both Epidermolysis Bullosa Disease Activity and Scarring Index and Body Surface Area Percentage scores.
- Reduced frequency of dressing changes (1 less per 2 weeks for Oleogel-S10 versus 0 less per 2 weeks for control gel).
- Improved pain among participants aged 4 years and older while their dressings were being changed.
- Reduced rates of wound infection (0.9% Oleogel-S10 vs. 4.4% control gel).
- Similar rates of treatment-emergent adverse events (24.8% vs. 22.8%, respectively), which were mostly deemed to be mild or moderate.
The EASE study – an important trial for EB
EASE is an important trial for EB, the study’s principal investigator Dédée Murrell, MD, DSc, University of New South Wales, Sydney, has pointed out previously.
“This was the first EB study to meet its primary endpoint and demonstrated a statistically significant acceleration of target wound healing by day 45,” Dr. Murrell said in a press release issued by Amryt Pharma to coincide with the online publication of the trial results.
“In addition, the favorable trends we see with key secondary endpoints such as reduced wound burden, pain, and frequency of dressing changes are considered as being very meaningful for patients,” Dr. Murrell said.
The EASE study was funded by Amryt Research Limited. Dr. Kiritsi reported receiving honoraria or consultation fees from Amryt, RHEACELL GmbH, and Fibrx Derm. She also acknowledged grant or research support from DEBRA International, EB Research Partnership, Fritz-Thyssen Foundation, German Research Foundation, and RHEACELL. Dr. Murrell has ties to Amryt and Amicus and is a co-owner of the patent for topical sirolimus for EB simplex.
A version of this article appeared on Medscape.com.
Additional when compared with a control gel.
In a final, post hoc analysis to come from the trial, 15 of 45 (33%) patients treated with Oleogel-S10 versus 5 of 48 (10.4%) treated with the control gel were reported as no longer needing daily dressing changes at 45 days of follow-up.
Moreover, the effect was sustained, with similar percentages of patients no longer requiring daily dressing changes at 60 days (34% vs. 13%, respectively) and 90 days (36% vs. 11%) of follow-up.
The mean reduction in daily dressing changes was 1.36 for Oleogel-S10 and 0.41 for the control gel (P = .005).
“Patients who, in the beginning, had daily dressing changes had almost three fewer dressing changes every 2 weeks if they were treated with Oleogel-S10,” Dimitra Kiritsi, MD, PhD, of the department of dermatology at the University of Freiburg (Germany), reported at the annual congress of the European Academy of Dermatology and Venereology. By comparison, patients in the control group had just one fewer daily dressing change in 2 weeks.
“You might say okay, but what does this mean in terms of time?” added Dr. Kiritsi. Using historical data on the time required for whole body care (Orphanet J Rare Dis. 2020 Jan 3. doi: 10.1186/s13023-019-1279-y), it was estimated that treatment with Oleogel-S10 was associated with an overall time-saving per week of 11 hours (6.6 hours for the patient and 4.4 hours for the caregiver) and use of the control gel was associated with an overall time-saving of 4 hours (2.4 hours for the patient and 1.6 hours for the caregiver).
“This is, for our patients, important,” said Dr. Kiritsi, as “it is time that they can spend doing something nice with the family” instead, avoiding the pain and distress associated with frequent dressing changes.
Approved in Europe, not in the United States
Oleogel-S10, classified as an herbal product, contains triterpenes derived from birch bark extract, which have been formulated with sunflower oil to form a gel.
Despite being approved for use in Europe, Oleogel-S10 has not yet been approved to treat EB in the United States. The FDA did not approve Amryt Pharma’s new drug application in February 2022. The application had included data from the EASE trial.
EASE included 223 patients with dystrophic or junctional EB, including 156 children, at 58 sites in 28 countries. As such, this makes it the largest treatment study in this rare genetic disease to date.
The trial had consisted of an initial 90-day, double-blind treatment period, during which time 109 patients had used Oleogel-S10 and 114 had used a control gel. This was followed by a 24-month open-label phase, during which time all remaining patients (n = 205) had used Oleogel-S10 on top of their standard of care.
Dr. Kiritsi summarized the main results of the EASE trial as follows.
- Complete healing of target wounds (primary endpoint) in 41.3% of patients treated with Oleogel-S10 and 28.9% of patients treated with the control gel (P = .013).
- Improved total body wound burden measured by both Epidermolysis Bullosa Disease Activity and Scarring Index and Body Surface Area Percentage scores.
- Reduced frequency of dressing changes (1 less per 2 weeks for Oleogel-S10 versus 0 less per 2 weeks for control gel).
- Improved pain among participants aged 4 years and older while their dressings were being changed.
- Reduced rates of wound infection (0.9% Oleogel-S10 vs. 4.4% control gel).
- Similar rates of treatment-emergent adverse events (24.8% vs. 22.8%, respectively), which were mostly deemed to be mild or moderate.
The EASE study – an important trial for EB
EASE is an important trial for EB, the study’s principal investigator Dédée Murrell, MD, DSc, University of New South Wales, Sydney, has pointed out previously.
“This was the first EB study to meet its primary endpoint and demonstrated a statistically significant acceleration of target wound healing by day 45,” Dr. Murrell said in a press release issued by Amryt Pharma to coincide with the online publication of the trial results.
“In addition, the favorable trends we see with key secondary endpoints such as reduced wound burden, pain, and frequency of dressing changes are considered as being very meaningful for patients,” Dr. Murrell said.
The EASE study was funded by Amryt Research Limited. Dr. Kiritsi reported receiving honoraria or consultation fees from Amryt, RHEACELL GmbH, and Fibrx Derm. She also acknowledged grant or research support from DEBRA International, EB Research Partnership, Fritz-Thyssen Foundation, German Research Foundation, and RHEACELL. Dr. Murrell has ties to Amryt and Amicus and is a co-owner of the patent for topical sirolimus for EB simplex.
A version of this article appeared on Medscape.com.
FROM THE EADV CONGRESS
Survey finds oral minoxidil shortage in Washington-area pharmacies
A .
Patients are not finding out until they go to pick up their prescription, which can result in an interruption of treatment – and, potentially a loss of hard-earned hair gain, said Adam Friedman, MD, professor and chair of dermatology at George Washington University, Washington, who was the lead author of the survey, published online on Oct. 26 as a research letter in the Journal of Drugs in Dermatology.
Going off low-dose oral minoxidil may spark a telogen effluvium event, and that is very disappointing to patients, Dr. Friedman told this news organization.
“There needs to be some system that alerts us,” he said. “Even if it’s a minor shortage, just so we’re aware. We can then prepare patients,” he added, noting that it would be better for someone to be taking a lower-than-normal dose rather than no medication at all while they wait for a refill.
Minoxidil has long been approved in a topical formulation to treat androgenetic alopecia, but a low-dose oral form has gained currency in the wake of findings that it might more effectively treat hair loss, and is without side effects. A New York Times article in August 2022 touting low-dose oral minoxidil as a cheap and effective hair loss drug appeared to ignite interest in this option. In May, 2023, researchers reporting in JAMA Network Open demonstrated a significant uptick in prescriptions for oral minoxidil in the wake of the article’s publication.
Oral minoxidil is approved by the Food and Drug Administration only for hypertension, but dermatologists are prescribing it off-label at a lower dose for hair loss. Dr. Friedman said it’s not clear whether the shortages his team found are national in scope, or whether they are a result of increased demand, or other factors.
After several patients told him they were having trouble filling minoxidil prescriptions, and colleagues said they’d had patients with similar experiences, Dr. Friedman and his colleagues undertook the survey. In the first week of October 2023, they contacted 277 pharmacies by phone in Washington and surrounding Virginia and Maryland counties. The pharmacies were CVS, Giant, Walgreens, and Harris Teeter.
Of the 277 pharmacies they contacted, 40% (111) reported availability of 2.5-mg tablets for a 30-day supply, and just under 30% (82) reported having 10-mg tablets for a 30-day supply.
For treating hair loss, most patients are prescribed 2.5-mg pills, with starting doses ranging from 0.625 mg to 5 mg twice a day, Dr. Friedman said. The 10-mg dose is more frequently prescribed for hypertension.
Only 28% (19 of 67) of the Maryland pharmacies had 30-day supplies of 2.5-mg tablets on hand, and just 22% (15) of the Maryland pharmacies had 30-day supplies of 10-mg tablets. In Northern Virginia, 44% (63 of 143) of the pharmacies had 30-day supplies of the 2.5 mg tablets, as did just 43% (29 of 67) of the Washington pharmacies.
Dr. Friedman said he has started giving patients paper prescriptions they can use to shop around, rather than electronically sending a prescription to a particular pharmacy.
Neither the Food and Drug Administration nor the American Society of Health System Pharmacists lists oral minoxidil as a drug in shortage.
Michael Ganio, PharmD, senior director of pharmacy practice and quality for ASHP, said the organization received a report from wholesalers in mid-September showing spotty oral minoxidil availability, with the drug on backorder with some manufacturers. ASHP's shortages list is compiled from reports from physicians, manufacturers and wholesalers, he said.
Under what he calls "blue sky conditions," pharmacies using a just-in-time inventory model should be able to fill prescriptions within hours or days, which might explain why some pharmacies in the Washington, DC area survey did not have a 30-day supply on hand, he said. However, Dr. Ganio noted that the causes of drug shortages are complex and multi-factorial. For now, he said there have been no oral minoxidil shortage reports since mid-September.
But Dr. Friedman said some of his patients have waited weeks for a new supply – and that no one is aware of the problem until the last moment.
The lack of alerts or transparency “also erodes the physician-patient relationship because there’s this expectation of the patient that we should have known this,” said Dr. Friedman.
Dr. Friedman reports no relevant financial relationships.
This story was updated on 11/2/2023.
A .
Patients are not finding out until they go to pick up their prescription, which can result in an interruption of treatment – and, potentially a loss of hard-earned hair gain, said Adam Friedman, MD, professor and chair of dermatology at George Washington University, Washington, who was the lead author of the survey, published online on Oct. 26 as a research letter in the Journal of Drugs in Dermatology.
Going off low-dose oral minoxidil may spark a telogen effluvium event, and that is very disappointing to patients, Dr. Friedman told this news organization.
“There needs to be some system that alerts us,” he said. “Even if it’s a minor shortage, just so we’re aware. We can then prepare patients,” he added, noting that it would be better for someone to be taking a lower-than-normal dose rather than no medication at all while they wait for a refill.
Minoxidil has long been approved in a topical formulation to treat androgenetic alopecia, but a low-dose oral form has gained currency in the wake of findings that it might more effectively treat hair loss, and is without side effects. A New York Times article in August 2022 touting low-dose oral minoxidil as a cheap and effective hair loss drug appeared to ignite interest in this option. In May, 2023, researchers reporting in JAMA Network Open demonstrated a significant uptick in prescriptions for oral minoxidil in the wake of the article’s publication.
Oral minoxidil is approved by the Food and Drug Administration only for hypertension, but dermatologists are prescribing it off-label at a lower dose for hair loss. Dr. Friedman said it’s not clear whether the shortages his team found are national in scope, or whether they are a result of increased demand, or other factors.
After several patients told him they were having trouble filling minoxidil prescriptions, and colleagues said they’d had patients with similar experiences, Dr. Friedman and his colleagues undertook the survey. In the first week of October 2023, they contacted 277 pharmacies by phone in Washington and surrounding Virginia and Maryland counties. The pharmacies were CVS, Giant, Walgreens, and Harris Teeter.
Of the 277 pharmacies they contacted, 40% (111) reported availability of 2.5-mg tablets for a 30-day supply, and just under 30% (82) reported having 10-mg tablets for a 30-day supply.
For treating hair loss, most patients are prescribed 2.5-mg pills, with starting doses ranging from 0.625 mg to 5 mg twice a day, Dr. Friedman said. The 10-mg dose is more frequently prescribed for hypertension.
Only 28% (19 of 67) of the Maryland pharmacies had 30-day supplies of 2.5-mg tablets on hand, and just 22% (15) of the Maryland pharmacies had 30-day supplies of 10-mg tablets. In Northern Virginia, 44% (63 of 143) of the pharmacies had 30-day supplies of the 2.5 mg tablets, as did just 43% (29 of 67) of the Washington pharmacies.
Dr. Friedman said he has started giving patients paper prescriptions they can use to shop around, rather than electronically sending a prescription to a particular pharmacy.
Neither the Food and Drug Administration nor the American Society of Health System Pharmacists lists oral minoxidil as a drug in shortage.
Michael Ganio, PharmD, senior director of pharmacy practice and quality for ASHP, said the organization received a report from wholesalers in mid-September showing spotty oral minoxidil availability, with the drug on backorder with some manufacturers. ASHP's shortages list is compiled from reports from physicians, manufacturers and wholesalers, he said.
Under what he calls "blue sky conditions," pharmacies using a just-in-time inventory model should be able to fill prescriptions within hours or days, which might explain why some pharmacies in the Washington, DC area survey did not have a 30-day supply on hand, he said. However, Dr. Ganio noted that the causes of drug shortages are complex and multi-factorial. For now, he said there have been no oral minoxidil shortage reports since mid-September.
But Dr. Friedman said some of his patients have waited weeks for a new supply – and that no one is aware of the problem until the last moment.
The lack of alerts or transparency “also erodes the physician-patient relationship because there’s this expectation of the patient that we should have known this,” said Dr. Friedman.
Dr. Friedman reports no relevant financial relationships.
This story was updated on 11/2/2023.
A .
Patients are not finding out until they go to pick up their prescription, which can result in an interruption of treatment – and, potentially a loss of hard-earned hair gain, said Adam Friedman, MD, professor and chair of dermatology at George Washington University, Washington, who was the lead author of the survey, published online on Oct. 26 as a research letter in the Journal of Drugs in Dermatology.
Going off low-dose oral minoxidil may spark a telogen effluvium event, and that is very disappointing to patients, Dr. Friedman told this news organization.
“There needs to be some system that alerts us,” he said. “Even if it’s a minor shortage, just so we’re aware. We can then prepare patients,” he added, noting that it would be better for someone to be taking a lower-than-normal dose rather than no medication at all while they wait for a refill.
Minoxidil has long been approved in a topical formulation to treat androgenetic alopecia, but a low-dose oral form has gained currency in the wake of findings that it might more effectively treat hair loss, and is without side effects. A New York Times article in August 2022 touting low-dose oral minoxidil as a cheap and effective hair loss drug appeared to ignite interest in this option. In May, 2023, researchers reporting in JAMA Network Open demonstrated a significant uptick in prescriptions for oral minoxidil in the wake of the article’s publication.
Oral minoxidil is approved by the Food and Drug Administration only for hypertension, but dermatologists are prescribing it off-label at a lower dose for hair loss. Dr. Friedman said it’s not clear whether the shortages his team found are national in scope, or whether they are a result of increased demand, or other factors.
After several patients told him they were having trouble filling minoxidil prescriptions, and colleagues said they’d had patients with similar experiences, Dr. Friedman and his colleagues undertook the survey. In the first week of October 2023, they contacted 277 pharmacies by phone in Washington and surrounding Virginia and Maryland counties. The pharmacies were CVS, Giant, Walgreens, and Harris Teeter.
Of the 277 pharmacies they contacted, 40% (111) reported availability of 2.5-mg tablets for a 30-day supply, and just under 30% (82) reported having 10-mg tablets for a 30-day supply.
For treating hair loss, most patients are prescribed 2.5-mg pills, with starting doses ranging from 0.625 mg to 5 mg twice a day, Dr. Friedman said. The 10-mg dose is more frequently prescribed for hypertension.
Only 28% (19 of 67) of the Maryland pharmacies had 30-day supplies of 2.5-mg tablets on hand, and just 22% (15) of the Maryland pharmacies had 30-day supplies of 10-mg tablets. In Northern Virginia, 44% (63 of 143) of the pharmacies had 30-day supplies of the 2.5 mg tablets, as did just 43% (29 of 67) of the Washington pharmacies.
Dr. Friedman said he has started giving patients paper prescriptions they can use to shop around, rather than electronically sending a prescription to a particular pharmacy.
Neither the Food and Drug Administration nor the American Society of Health System Pharmacists lists oral minoxidil as a drug in shortage.
Michael Ganio, PharmD, senior director of pharmacy practice and quality for ASHP, said the organization received a report from wholesalers in mid-September showing spotty oral minoxidil availability, with the drug on backorder with some manufacturers. ASHP's shortages list is compiled from reports from physicians, manufacturers and wholesalers, he said.
Under what he calls "blue sky conditions," pharmacies using a just-in-time inventory model should be able to fill prescriptions within hours or days, which might explain why some pharmacies in the Washington, DC area survey did not have a 30-day supply on hand, he said. However, Dr. Ganio noted that the causes of drug shortages are complex and multi-factorial. For now, he said there have been no oral minoxidil shortage reports since mid-September.
But Dr. Friedman said some of his patients have waited weeks for a new supply – and that no one is aware of the problem until the last moment.
The lack of alerts or transparency “also erodes the physician-patient relationship because there’s this expectation of the patient that we should have known this,” said Dr. Friedman.
Dr. Friedman reports no relevant financial relationships.
This story was updated on 11/2/2023.
FROM THE JOURNAL OF DRUGS IN DERMATOLOGY
AI flagged skin cancer with near-perfect accuracy, in UK study
. AI detected more than 99% of all skin cancers.
The researchers tested the AI by integrating it into a clinical diagnosis process – anticipating a future in which AI helps doctors catch skin cancer faster and triage patients.
Skin cancer is the most common cancer in the United States; one in five 5 Americans develop skin cancer by age 70. With melanoma, the deadliest skin cancer, the 5-year survival rate is better than 99% if caught early, though only about three-quarters of melanomas are caught at this stage.
Amid rising skin cancer rates come concerns that the number of dermatologists in the workforce isn’t keeping pace. That may be why the average wait time for a dermatology appointment is trending up – in 2022, it reached 34.5 days.
The study, which was presented at the European Academy of Dermatology and Venereology Congress recently and has not yet been published, involved 6,900 patients in the United Kingdom with suspected skin cancer. The patients had been referred by their primary care physicians. The researchers took images of the suspicious areas and uploaded them to the AI software. The AI’s assessment was then shared with a dermatologist.
“Note that the diagnosis issued by the AI was not hidden from the dermatologist doing the second assessment,” said lead researcher Kashini Andrew, MBBS, a dermatologist and specialist registrar at University Hospitals Birmingham NHS Foundation Trust.
Dr. Andrew acknowledged that this may have influenced the dermatologist’s opinion. But that’s the vision of how doctors could use this tool.
The AI caught 59 of 59 melanomas and 189 of 190 total skin cancers (99.5%). (The one case that the AI missed was caught by the dermatologist.) It also flagged 541 of 585 precancerous lesions (92.5%). This represented a big improvement from a 2021 version of the model, which detected 86% of melanomas, 84% of all skin cancers, and 54% of precancerous lesions.
Over the 10-month period of the study, the system saved more than 1,000 face-to-face consultations, freeing dermatologists’ time to catch more cancers and serve more patients.
Limitations
The patients in the study were from “one hospital in a single region of the UK,” and the sample was not large enough to allow broad statements to be made about the use of AI in dermatology, Dr. Andrew said.
But it can open the conversation. Roxana Daneshjou, MD, PhD, a dermatologist at Stanford (Calif.) University who has studied the pros and cons of AI in medicine, had some concerns. For one thing, doctors can gather more in-depth information during an in-person exam than AI can glean from a photo, Dr. Daneshjou noted. They can examine skin texture, gather patient history, and take photos with special lighting and magnification.
And the AI needs to get better at ruling out malignancy, Dr. Daneshjou said. In this study, the AI identified 75% of benign lesions, a decline from the earlier version. The researchers noted in the abstract that this is a potential trade-off for increased sensitivity.
“[Unnecessary] biopsies can clog up the health care system, cost money, and cause stress and scarring,” said Dr. Daneshjou. “You don’t want to increase the burden of that.”
Still, if AI software such as the kind used in the study proves just as accurate in larger, more diverse sample sizes, then it could be a powerful tool for triage, Dr. Daneshjou said. “If AI gets particularly good at finding malignancy and also ruling it out, that would be a win.”
A version of this article appeared on Medscape.com.
. AI detected more than 99% of all skin cancers.
The researchers tested the AI by integrating it into a clinical diagnosis process – anticipating a future in which AI helps doctors catch skin cancer faster and triage patients.
Skin cancer is the most common cancer in the United States; one in five 5 Americans develop skin cancer by age 70. With melanoma, the deadliest skin cancer, the 5-year survival rate is better than 99% if caught early, though only about three-quarters of melanomas are caught at this stage.
Amid rising skin cancer rates come concerns that the number of dermatologists in the workforce isn’t keeping pace. That may be why the average wait time for a dermatology appointment is trending up – in 2022, it reached 34.5 days.
The study, which was presented at the European Academy of Dermatology and Venereology Congress recently and has not yet been published, involved 6,900 patients in the United Kingdom with suspected skin cancer. The patients had been referred by their primary care physicians. The researchers took images of the suspicious areas and uploaded them to the AI software. The AI’s assessment was then shared with a dermatologist.
“Note that the diagnosis issued by the AI was not hidden from the dermatologist doing the second assessment,” said lead researcher Kashini Andrew, MBBS, a dermatologist and specialist registrar at University Hospitals Birmingham NHS Foundation Trust.
Dr. Andrew acknowledged that this may have influenced the dermatologist’s opinion. But that’s the vision of how doctors could use this tool.
The AI caught 59 of 59 melanomas and 189 of 190 total skin cancers (99.5%). (The one case that the AI missed was caught by the dermatologist.) It also flagged 541 of 585 precancerous lesions (92.5%). This represented a big improvement from a 2021 version of the model, which detected 86% of melanomas, 84% of all skin cancers, and 54% of precancerous lesions.
Over the 10-month period of the study, the system saved more than 1,000 face-to-face consultations, freeing dermatologists’ time to catch more cancers and serve more patients.
Limitations
The patients in the study were from “one hospital in a single region of the UK,” and the sample was not large enough to allow broad statements to be made about the use of AI in dermatology, Dr. Andrew said.
But it can open the conversation. Roxana Daneshjou, MD, PhD, a dermatologist at Stanford (Calif.) University who has studied the pros and cons of AI in medicine, had some concerns. For one thing, doctors can gather more in-depth information during an in-person exam than AI can glean from a photo, Dr. Daneshjou noted. They can examine skin texture, gather patient history, and take photos with special lighting and magnification.
And the AI needs to get better at ruling out malignancy, Dr. Daneshjou said. In this study, the AI identified 75% of benign lesions, a decline from the earlier version. The researchers noted in the abstract that this is a potential trade-off for increased sensitivity.
“[Unnecessary] biopsies can clog up the health care system, cost money, and cause stress and scarring,” said Dr. Daneshjou. “You don’t want to increase the burden of that.”
Still, if AI software such as the kind used in the study proves just as accurate in larger, more diverse sample sizes, then it could be a powerful tool for triage, Dr. Daneshjou said. “If AI gets particularly good at finding malignancy and also ruling it out, that would be a win.”
A version of this article appeared on Medscape.com.
. AI detected more than 99% of all skin cancers.
The researchers tested the AI by integrating it into a clinical diagnosis process – anticipating a future in which AI helps doctors catch skin cancer faster and triage patients.
Skin cancer is the most common cancer in the United States; one in five 5 Americans develop skin cancer by age 70. With melanoma, the deadliest skin cancer, the 5-year survival rate is better than 99% if caught early, though only about three-quarters of melanomas are caught at this stage.
Amid rising skin cancer rates come concerns that the number of dermatologists in the workforce isn’t keeping pace. That may be why the average wait time for a dermatology appointment is trending up – in 2022, it reached 34.5 days.
The study, which was presented at the European Academy of Dermatology and Venereology Congress recently and has not yet been published, involved 6,900 patients in the United Kingdom with suspected skin cancer. The patients had been referred by their primary care physicians. The researchers took images of the suspicious areas and uploaded them to the AI software. The AI’s assessment was then shared with a dermatologist.
“Note that the diagnosis issued by the AI was not hidden from the dermatologist doing the second assessment,” said lead researcher Kashini Andrew, MBBS, a dermatologist and specialist registrar at University Hospitals Birmingham NHS Foundation Trust.
Dr. Andrew acknowledged that this may have influenced the dermatologist’s opinion. But that’s the vision of how doctors could use this tool.
The AI caught 59 of 59 melanomas and 189 of 190 total skin cancers (99.5%). (The one case that the AI missed was caught by the dermatologist.) It also flagged 541 of 585 precancerous lesions (92.5%). This represented a big improvement from a 2021 version of the model, which detected 86% of melanomas, 84% of all skin cancers, and 54% of precancerous lesions.
Over the 10-month period of the study, the system saved more than 1,000 face-to-face consultations, freeing dermatologists’ time to catch more cancers and serve more patients.
Limitations
The patients in the study were from “one hospital in a single region of the UK,” and the sample was not large enough to allow broad statements to be made about the use of AI in dermatology, Dr. Andrew said.
But it can open the conversation. Roxana Daneshjou, MD, PhD, a dermatologist at Stanford (Calif.) University who has studied the pros and cons of AI in medicine, had some concerns. For one thing, doctors can gather more in-depth information during an in-person exam than AI can glean from a photo, Dr. Daneshjou noted. They can examine skin texture, gather patient history, and take photos with special lighting and magnification.
And the AI needs to get better at ruling out malignancy, Dr. Daneshjou said. In this study, the AI identified 75% of benign lesions, a decline from the earlier version. The researchers noted in the abstract that this is a potential trade-off for increased sensitivity.
“[Unnecessary] biopsies can clog up the health care system, cost money, and cause stress and scarring,” said Dr. Daneshjou. “You don’t want to increase the burden of that.”
Still, if AI software such as the kind used in the study proves just as accurate in larger, more diverse sample sizes, then it could be a powerful tool for triage, Dr. Daneshjou said. “If AI gets particularly good at finding malignancy and also ruling it out, that would be a win.”
A version of this article appeared on Medscape.com.
FROM THE EADV CONGRESS
FDA OKs first ustekinumab biosimilar
The U.S. Food and Drug Administration has approved ustekinumab-auub (Wezlana) as a biosimilar to ustekinumab (Stelara) for the treatment of multiple inflammatory conditions. This is the first approval for a ustekinumab biosimilar in the United States.
Ustekinumab-auub was also granted an interchangeability designation, meaning that, depending on state law, a pharmacist may substitute the biosimilar for the reference product without consulting the prescribing provider.
“Today’s approval exemplifies the FDA’s longstanding commitment to support a competitive marketplace for biological products,” Sarah Yim, MD, director of the Office of Therapeutic Biologics and Biosimilars in the FDA’s Center for Drug Evaluation and Research, said in a statement. “This approval can empower patients by helping to increase access to safe, effective, and high-quality medications at potentially lower cost.”
Ustekinumab, manufactured by Johnson & Johnson, targets interleukin-12 and IL-23 and was first approved in 2009. Ustekinumab-auub was developed by Amgen.
Ustekinumab-auub is approved for the treatment of adult patients with moderate to severe plaque psoriasis who are candidates for phototherapy or systemic therapy, active psoriatic arthritis, moderate to severely active Crohn’s disease, and moderate to severely active ulcerative colitis. It is also approved for pediatric patients aged 6 years and older with moderate to severe plaque psoriasis who are candidates for phototherapy or systemic therapy and active psoriatic arthritis.
The approval was based on “comprehensive review of scientific evidence,” including “comparisons of the products on an analytical level using an extensive battery of chemical and biological tests and biological assays that confirmed similarity in the structural and functional features of Wezlana and Stelara (including those known to impact safety and efficacy), and comparative human pharmacokinetic data, clinical immunogenicity data, and other clinical safety and effectiveness data,” the FDA said.
Some common side effects of ustekinumab-auub include nasopharyngitis, upper respiratory tract infection, headache, fatigue, and nausea. The most severe side effect of the biosimilar, as with the reference drug ustekinumab, is infection.
The product launch of ustekinumab-auub will be delayed as a part of a settlement of Johnson & Johnson’s lawsuit against Amgen, according to Reuters. The details of the settlement are confidential, but it was stated that the biosimilar would be available by Jan. 1, 2025.
A version of this article first appeared on Medscape.com.
The U.S. Food and Drug Administration has approved ustekinumab-auub (Wezlana) as a biosimilar to ustekinumab (Stelara) for the treatment of multiple inflammatory conditions. This is the first approval for a ustekinumab biosimilar in the United States.
Ustekinumab-auub was also granted an interchangeability designation, meaning that, depending on state law, a pharmacist may substitute the biosimilar for the reference product without consulting the prescribing provider.
“Today’s approval exemplifies the FDA’s longstanding commitment to support a competitive marketplace for biological products,” Sarah Yim, MD, director of the Office of Therapeutic Biologics and Biosimilars in the FDA’s Center for Drug Evaluation and Research, said in a statement. “This approval can empower patients by helping to increase access to safe, effective, and high-quality medications at potentially lower cost.”
Ustekinumab, manufactured by Johnson & Johnson, targets interleukin-12 and IL-23 and was first approved in 2009. Ustekinumab-auub was developed by Amgen.
Ustekinumab-auub is approved for the treatment of adult patients with moderate to severe plaque psoriasis who are candidates for phototherapy or systemic therapy, active psoriatic arthritis, moderate to severely active Crohn’s disease, and moderate to severely active ulcerative colitis. It is also approved for pediatric patients aged 6 years and older with moderate to severe plaque psoriasis who are candidates for phototherapy or systemic therapy and active psoriatic arthritis.
The approval was based on “comprehensive review of scientific evidence,” including “comparisons of the products on an analytical level using an extensive battery of chemical and biological tests and biological assays that confirmed similarity in the structural and functional features of Wezlana and Stelara (including those known to impact safety and efficacy), and comparative human pharmacokinetic data, clinical immunogenicity data, and other clinical safety and effectiveness data,” the FDA said.
Some common side effects of ustekinumab-auub include nasopharyngitis, upper respiratory tract infection, headache, fatigue, and nausea. The most severe side effect of the biosimilar, as with the reference drug ustekinumab, is infection.
The product launch of ustekinumab-auub will be delayed as a part of a settlement of Johnson & Johnson’s lawsuit against Amgen, according to Reuters. The details of the settlement are confidential, but it was stated that the biosimilar would be available by Jan. 1, 2025.
A version of this article first appeared on Medscape.com.
The U.S. Food and Drug Administration has approved ustekinumab-auub (Wezlana) as a biosimilar to ustekinumab (Stelara) for the treatment of multiple inflammatory conditions. This is the first approval for a ustekinumab biosimilar in the United States.
Ustekinumab-auub was also granted an interchangeability designation, meaning that, depending on state law, a pharmacist may substitute the biosimilar for the reference product without consulting the prescribing provider.
“Today’s approval exemplifies the FDA’s longstanding commitment to support a competitive marketplace for biological products,” Sarah Yim, MD, director of the Office of Therapeutic Biologics and Biosimilars in the FDA’s Center for Drug Evaluation and Research, said in a statement. “This approval can empower patients by helping to increase access to safe, effective, and high-quality medications at potentially lower cost.”
Ustekinumab, manufactured by Johnson & Johnson, targets interleukin-12 and IL-23 and was first approved in 2009. Ustekinumab-auub was developed by Amgen.
Ustekinumab-auub is approved for the treatment of adult patients with moderate to severe plaque psoriasis who are candidates for phototherapy or systemic therapy, active psoriatic arthritis, moderate to severely active Crohn’s disease, and moderate to severely active ulcerative colitis. It is also approved for pediatric patients aged 6 years and older with moderate to severe plaque psoriasis who are candidates for phototherapy or systemic therapy and active psoriatic arthritis.
The approval was based on “comprehensive review of scientific evidence,” including “comparisons of the products on an analytical level using an extensive battery of chemical and biological tests and biological assays that confirmed similarity in the structural and functional features of Wezlana and Stelara (including those known to impact safety and efficacy), and comparative human pharmacokinetic data, clinical immunogenicity data, and other clinical safety and effectiveness data,” the FDA said.
Some common side effects of ustekinumab-auub include nasopharyngitis, upper respiratory tract infection, headache, fatigue, and nausea. The most severe side effect of the biosimilar, as with the reference drug ustekinumab, is infection.
The product launch of ustekinumab-auub will be delayed as a part of a settlement of Johnson & Johnson’s lawsuit against Amgen, according to Reuters. The details of the settlement are confidential, but it was stated that the biosimilar would be available by Jan. 1, 2025.
A version of this article first appeared on Medscape.com.
FDA approves second treatment for adults with hidradenitis suppurativa
The U.S.
The development, which was announced Oct. 31, makes secukinumab the first and only interleukin (IL)–17A inhibitor approved by the FDA for HS, which affects an estimated 1% of the worldwide population. It joins the tumor necrosis factor blocker adalimumab as the only FDA-approved treatment options for HS.
Secukinumab (Cosentyx) was previously approved by the FDA for treatment of moderate-to-severe plaque psoriasis in adults, and several other indications including psoriatic arthritis and ankylosing spondylitis.
Approval for HS was based on the pivotal phase 3 SUNSHINE and SUNRISE trials, which had a combined enrollment of more than 1,000 patients with HS in 40 countries. The studies evaluated efficacy, safety, and tolerability of two dose regimens of the drug in adults with moderate to severe HS at 16 weeks and up to 52 weeks.
According to a press release from Novartis announcing the approval, results at week 16 showed that a significantly higher proportion of patients achieved a Hidradenitis Suppurativa Clinical Response (HiSCR) when treated with secukinumab 300 mg every 2 weeks, compared with placebo: 44.5% vs. 29.4%, respectively, in the SUNSHINE trial and 38.3.% vs. 26.1% in the SUNRISE trial (P < .05 for both associations).
Similarly, results at week 16 showed that a significantly higher proportion of patients achieved an HiSCR when treated with secukinumab 300 mg every 4 weeks, compared with placebo: 41.3% vs. 29.4% in the SUNSHINE trial and 42.5% vs. 26.1% in the SUNRISE trial (P < .05 for both associations).
In addition, in an exploratory analysis out to 52 weeks, HiSCR values observed at week 16 following either dose regimen of secukinumab were improved over time up to week 52. In SUNSHINE, the values improved by 56.4% in patients treated with secukinumab every 3 weeks and by 56.3% in those treated with secukinumab every 4 weeks. In SUNRISE, the values improved by 65% in patients who were treated with secukinumab every 2 weeks and by 62.2% in those who were treated with the drug every 4 weeks.
In an interview, Haley Naik, MD, a dermatologist who directs the hidradenitis suppurativa program at the University of California, San Francisco, characterized the approval as a win for HS patients. “Patients now not only have a second option for approved therapy for HS, but also an option that raises the bar for what we can expect from therapeutic response,” she told this news organization. “I am excited to see a novel therapy that improves HS and quality of life for patients make it through the regulatory pipeline.”
Dr. Naik disclosed that she has received grant support from AbbVie; consulting fees from 23andme, AbbVie, Aristea Therapeutics, Nimbus Therapeutics, Medscape, Sonoma Biotherapeutics, DAVA Oncology, Boehringer Ingelheim, Union Chimique Belge, and Novartis; investigator fees from Pfizer; and holds shares in Radera. She is also an associate editor for JAMA Dermatology and a board member of the Hidradenitis Suppurativa Foundation.
A version of this article first appeared on Medscape.com.
The U.S.
The development, which was announced Oct. 31, makes secukinumab the first and only interleukin (IL)–17A inhibitor approved by the FDA for HS, which affects an estimated 1% of the worldwide population. It joins the tumor necrosis factor blocker adalimumab as the only FDA-approved treatment options for HS.
Secukinumab (Cosentyx) was previously approved by the FDA for treatment of moderate-to-severe plaque psoriasis in adults, and several other indications including psoriatic arthritis and ankylosing spondylitis.
Approval for HS was based on the pivotal phase 3 SUNSHINE and SUNRISE trials, which had a combined enrollment of more than 1,000 patients with HS in 40 countries. The studies evaluated efficacy, safety, and tolerability of two dose regimens of the drug in adults with moderate to severe HS at 16 weeks and up to 52 weeks.
According to a press release from Novartis announcing the approval, results at week 16 showed that a significantly higher proportion of patients achieved a Hidradenitis Suppurativa Clinical Response (HiSCR) when treated with secukinumab 300 mg every 2 weeks, compared with placebo: 44.5% vs. 29.4%, respectively, in the SUNSHINE trial and 38.3.% vs. 26.1% in the SUNRISE trial (P < .05 for both associations).
Similarly, results at week 16 showed that a significantly higher proportion of patients achieved an HiSCR when treated with secukinumab 300 mg every 4 weeks, compared with placebo: 41.3% vs. 29.4% in the SUNSHINE trial and 42.5% vs. 26.1% in the SUNRISE trial (P < .05 for both associations).
In addition, in an exploratory analysis out to 52 weeks, HiSCR values observed at week 16 following either dose regimen of secukinumab were improved over time up to week 52. In SUNSHINE, the values improved by 56.4% in patients treated with secukinumab every 3 weeks and by 56.3% in those treated with secukinumab every 4 weeks. In SUNRISE, the values improved by 65% in patients who were treated with secukinumab every 2 weeks and by 62.2% in those who were treated with the drug every 4 weeks.
In an interview, Haley Naik, MD, a dermatologist who directs the hidradenitis suppurativa program at the University of California, San Francisco, characterized the approval as a win for HS patients. “Patients now not only have a second option for approved therapy for HS, but also an option that raises the bar for what we can expect from therapeutic response,” she told this news organization. “I am excited to see a novel therapy that improves HS and quality of life for patients make it through the regulatory pipeline.”
Dr. Naik disclosed that she has received grant support from AbbVie; consulting fees from 23andme, AbbVie, Aristea Therapeutics, Nimbus Therapeutics, Medscape, Sonoma Biotherapeutics, DAVA Oncology, Boehringer Ingelheim, Union Chimique Belge, and Novartis; investigator fees from Pfizer; and holds shares in Radera. She is also an associate editor for JAMA Dermatology and a board member of the Hidradenitis Suppurativa Foundation.
A version of this article first appeared on Medscape.com.
The U.S.
The development, which was announced Oct. 31, makes secukinumab the first and only interleukin (IL)–17A inhibitor approved by the FDA for HS, which affects an estimated 1% of the worldwide population. It joins the tumor necrosis factor blocker adalimumab as the only FDA-approved treatment options for HS.
Secukinumab (Cosentyx) was previously approved by the FDA for treatment of moderate-to-severe plaque psoriasis in adults, and several other indications including psoriatic arthritis and ankylosing spondylitis.
Approval for HS was based on the pivotal phase 3 SUNSHINE and SUNRISE trials, which had a combined enrollment of more than 1,000 patients with HS in 40 countries. The studies evaluated efficacy, safety, and tolerability of two dose regimens of the drug in adults with moderate to severe HS at 16 weeks and up to 52 weeks.
According to a press release from Novartis announcing the approval, results at week 16 showed that a significantly higher proportion of patients achieved a Hidradenitis Suppurativa Clinical Response (HiSCR) when treated with secukinumab 300 mg every 2 weeks, compared with placebo: 44.5% vs. 29.4%, respectively, in the SUNSHINE trial and 38.3.% vs. 26.1% in the SUNRISE trial (P < .05 for both associations).
Similarly, results at week 16 showed that a significantly higher proportion of patients achieved an HiSCR when treated with secukinumab 300 mg every 4 weeks, compared with placebo: 41.3% vs. 29.4% in the SUNSHINE trial and 42.5% vs. 26.1% in the SUNRISE trial (P < .05 for both associations).
In addition, in an exploratory analysis out to 52 weeks, HiSCR values observed at week 16 following either dose regimen of secukinumab were improved over time up to week 52. In SUNSHINE, the values improved by 56.4% in patients treated with secukinumab every 3 weeks and by 56.3% in those treated with secukinumab every 4 weeks. In SUNRISE, the values improved by 65% in patients who were treated with secukinumab every 2 weeks and by 62.2% in those who were treated with the drug every 4 weeks.
In an interview, Haley Naik, MD, a dermatologist who directs the hidradenitis suppurativa program at the University of California, San Francisco, characterized the approval as a win for HS patients. “Patients now not only have a second option for approved therapy for HS, but also an option that raises the bar for what we can expect from therapeutic response,” she told this news organization. “I am excited to see a novel therapy that improves HS and quality of life for patients make it through the regulatory pipeline.”
Dr. Naik disclosed that she has received grant support from AbbVie; consulting fees from 23andme, AbbVie, Aristea Therapeutics, Nimbus Therapeutics, Medscape, Sonoma Biotherapeutics, DAVA Oncology, Boehringer Ingelheim, Union Chimique Belge, and Novartis; investigator fees from Pfizer; and holds shares in Radera. She is also an associate editor for JAMA Dermatology and a board member of the Hidradenitis Suppurativa Foundation.
A version of this article first appeared on Medscape.com.
Review finds no CV or VTE risk signal with use of JAK inhibitors for skin indications
, results from a systematic literature review, and meta-analysis showed.
“There remains a knowledge gap regarding the risk of JAK inhibitor use and VTE and/or MACE in the dermatologic population,” researchers led by Michael S. Garshick, MD, a cardiologist at New York University Langone Health, wrote in their study, which was published online in JAMA Dermatology . “Pooled safety studies suggest that the risk of MACE and VTE may be lower in patients treated with JAK inhibitors for a dermatologic indication than the risk observed in the ORAL Surveillance study, which may be related to the younger age and better health status of those enrolled in trials for dermatologic indications.” The results of that study, which included patients with rheumatoid arthritis only, resulted in the addition of a boxed warning in the labels for topical and oral JAK inhibitors regarding the increased risk of MACE, VTE, serious infections, malignancies, and death .
For the review – thought to be the first to specifically evaluate these risks for dermatologic indications – the researchers searched PubMed and ClinicalTrials.gov from inception through April 1, 2023, for phase 3 dermatology randomized clinical trials (RCTs) to evaluate the risk of MACE, VTE, and all-cause mortality with JAK inhibitors, compared with placebo or an active comparator in the treatment of immune-mediated inflammatory skin diseases. They followed Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) guidelines and used a random-effects model and the DerSimonian-Laird method to calculate adverse events with odds ratios.
The database search yielded 35 RCTs with a total of 20,651 patients. Their mean age was 38.5 years, 54% were male, and the mean follow-up time was 4.9 months. Of the 35 trials, most (21) involved patients with atopic dermatitis, followed by psoriasis/psoriatic arthritis (9 trials), alopecia areata (3 trials) and vitiligo (2 trials).
The researchers found no significant difference between JAK inhibitors and placebo/active comparator in composite MACE and all-cause mortality (odds ratio, 0.83; 95% confidence interval, 0.44-1.57) or in VTE (OR, 0.52; 95% CI, 0.26-1.04).
In a secondary analysis, which included additional psoriatic arthritis RCTs, no significant differences between the treatment and placebo/active comparator groups were observed. Similarly, subgroup analyses of oral versus topical JAK inhibitors and a sensitivity analysis that excluded pediatric trials showed no significant differences between patients exposed to JAK inhibitors and those not exposed.
The researchers acknowledged certain limitations of the review, including the lack of access to patient-level data, the fact that most trials only included short-term follow-up, and that the findings have limited generalizability to an older patient population. “It remains unclear if the cardiovascular risks of JAK inhibitors are primarily due to patient level cardiovascular risk factors or are drug mediated,” they concluded. “Dermatologists should carefully select patients and assess baseline cardiovascular risk factors when considering JAK therapy. Cardiovascular risk assessment should continue for the duration of treatment.”
Raj Chovatiya, MD, PhD, assistant professor of dermatology and director of the center for eczema and itch at Northwestern University, Chicago, who was asked to comment on the study results, characterized the findings as reassuring to dermatologists who may be reluctant to initiate therapy with JAK inhibitors based on concerns about safety signals for MACE, VTE, and all-cause mortality.
“These data systematically show that across medications and across conditions, there doesn’t appear to be an increased signal for these events during the short-term, placebo-controlled period which generally spans a few months in most studies,” he told this news organization. The findings, he added, “align well with our clinical experience to date for JAK inhibitor use in inflammatory skin disease. Short-term safety, particularly in relation to boxed warning events such MACE, VTE, and all-cause mortality, have generally been favorable with real-world use. It’s good to have a rigorous statistical analysis to refer to when setting patient expectations.”
However, he noted that these data only examined short-term safety during the placebo or active comparator-controlled periods. “Considering that events like MACE or VTE may take many months or years to manifest, continued long-term data generation is needed to fully answer the question of risk,” he said.
Dr. Garshick disclosed that he received grants from Pfizer and personal fees from Bristol Myers Squibb during the conduct of the study and personal fees from Kiniksa Pharmaceuticals outside the submitted work. Several other coauthors reported having advisory board roles and/or having received funding or support from several pharmaceutical companies. Dr. Chovatiya disclosed that he is a consultant to, a speaker for, investigator, and/or a member of the advisory board for several pharmaceutical companies, including those that develop JAK inhibitors.
, results from a systematic literature review, and meta-analysis showed.
“There remains a knowledge gap regarding the risk of JAK inhibitor use and VTE and/or MACE in the dermatologic population,” researchers led by Michael S. Garshick, MD, a cardiologist at New York University Langone Health, wrote in their study, which was published online in JAMA Dermatology . “Pooled safety studies suggest that the risk of MACE and VTE may be lower in patients treated with JAK inhibitors for a dermatologic indication than the risk observed in the ORAL Surveillance study, which may be related to the younger age and better health status of those enrolled in trials for dermatologic indications.” The results of that study, which included patients with rheumatoid arthritis only, resulted in the addition of a boxed warning in the labels for topical and oral JAK inhibitors regarding the increased risk of MACE, VTE, serious infections, malignancies, and death .
For the review – thought to be the first to specifically evaluate these risks for dermatologic indications – the researchers searched PubMed and ClinicalTrials.gov from inception through April 1, 2023, for phase 3 dermatology randomized clinical trials (RCTs) to evaluate the risk of MACE, VTE, and all-cause mortality with JAK inhibitors, compared with placebo or an active comparator in the treatment of immune-mediated inflammatory skin diseases. They followed Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) guidelines and used a random-effects model and the DerSimonian-Laird method to calculate adverse events with odds ratios.
The database search yielded 35 RCTs with a total of 20,651 patients. Their mean age was 38.5 years, 54% were male, and the mean follow-up time was 4.9 months. Of the 35 trials, most (21) involved patients with atopic dermatitis, followed by psoriasis/psoriatic arthritis (9 trials), alopecia areata (3 trials) and vitiligo (2 trials).
The researchers found no significant difference between JAK inhibitors and placebo/active comparator in composite MACE and all-cause mortality (odds ratio, 0.83; 95% confidence interval, 0.44-1.57) or in VTE (OR, 0.52; 95% CI, 0.26-1.04).
In a secondary analysis, which included additional psoriatic arthritis RCTs, no significant differences between the treatment and placebo/active comparator groups were observed. Similarly, subgroup analyses of oral versus topical JAK inhibitors and a sensitivity analysis that excluded pediatric trials showed no significant differences between patients exposed to JAK inhibitors and those not exposed.
The researchers acknowledged certain limitations of the review, including the lack of access to patient-level data, the fact that most trials only included short-term follow-up, and that the findings have limited generalizability to an older patient population. “It remains unclear if the cardiovascular risks of JAK inhibitors are primarily due to patient level cardiovascular risk factors or are drug mediated,” they concluded. “Dermatologists should carefully select patients and assess baseline cardiovascular risk factors when considering JAK therapy. Cardiovascular risk assessment should continue for the duration of treatment.”
Raj Chovatiya, MD, PhD, assistant professor of dermatology and director of the center for eczema and itch at Northwestern University, Chicago, who was asked to comment on the study results, characterized the findings as reassuring to dermatologists who may be reluctant to initiate therapy with JAK inhibitors based on concerns about safety signals for MACE, VTE, and all-cause mortality.
“These data systematically show that across medications and across conditions, there doesn’t appear to be an increased signal for these events during the short-term, placebo-controlled period which generally spans a few months in most studies,” he told this news organization. The findings, he added, “align well with our clinical experience to date for JAK inhibitor use in inflammatory skin disease. Short-term safety, particularly in relation to boxed warning events such MACE, VTE, and all-cause mortality, have generally been favorable with real-world use. It’s good to have a rigorous statistical analysis to refer to when setting patient expectations.”
However, he noted that these data only examined short-term safety during the placebo or active comparator-controlled periods. “Considering that events like MACE or VTE may take many months or years to manifest, continued long-term data generation is needed to fully answer the question of risk,” he said.
Dr. Garshick disclosed that he received grants from Pfizer and personal fees from Bristol Myers Squibb during the conduct of the study and personal fees from Kiniksa Pharmaceuticals outside the submitted work. Several other coauthors reported having advisory board roles and/or having received funding or support from several pharmaceutical companies. Dr. Chovatiya disclosed that he is a consultant to, a speaker for, investigator, and/or a member of the advisory board for several pharmaceutical companies, including those that develop JAK inhibitors.
, results from a systematic literature review, and meta-analysis showed.
“There remains a knowledge gap regarding the risk of JAK inhibitor use and VTE and/or MACE in the dermatologic population,” researchers led by Michael S. Garshick, MD, a cardiologist at New York University Langone Health, wrote in their study, which was published online in JAMA Dermatology . “Pooled safety studies suggest that the risk of MACE and VTE may be lower in patients treated with JAK inhibitors for a dermatologic indication than the risk observed in the ORAL Surveillance study, which may be related to the younger age and better health status of those enrolled in trials for dermatologic indications.” The results of that study, which included patients with rheumatoid arthritis only, resulted in the addition of a boxed warning in the labels for topical and oral JAK inhibitors regarding the increased risk of MACE, VTE, serious infections, malignancies, and death .
For the review – thought to be the first to specifically evaluate these risks for dermatologic indications – the researchers searched PubMed and ClinicalTrials.gov from inception through April 1, 2023, for phase 3 dermatology randomized clinical trials (RCTs) to evaluate the risk of MACE, VTE, and all-cause mortality with JAK inhibitors, compared with placebo or an active comparator in the treatment of immune-mediated inflammatory skin diseases. They followed Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) guidelines and used a random-effects model and the DerSimonian-Laird method to calculate adverse events with odds ratios.
The database search yielded 35 RCTs with a total of 20,651 patients. Their mean age was 38.5 years, 54% were male, and the mean follow-up time was 4.9 months. Of the 35 trials, most (21) involved patients with atopic dermatitis, followed by psoriasis/psoriatic arthritis (9 trials), alopecia areata (3 trials) and vitiligo (2 trials).
The researchers found no significant difference between JAK inhibitors and placebo/active comparator in composite MACE and all-cause mortality (odds ratio, 0.83; 95% confidence interval, 0.44-1.57) or in VTE (OR, 0.52; 95% CI, 0.26-1.04).
In a secondary analysis, which included additional psoriatic arthritis RCTs, no significant differences between the treatment and placebo/active comparator groups were observed. Similarly, subgroup analyses of oral versus topical JAK inhibitors and a sensitivity analysis that excluded pediatric trials showed no significant differences between patients exposed to JAK inhibitors and those not exposed.
The researchers acknowledged certain limitations of the review, including the lack of access to patient-level data, the fact that most trials only included short-term follow-up, and that the findings have limited generalizability to an older patient population. “It remains unclear if the cardiovascular risks of JAK inhibitors are primarily due to patient level cardiovascular risk factors or are drug mediated,” they concluded. “Dermatologists should carefully select patients and assess baseline cardiovascular risk factors when considering JAK therapy. Cardiovascular risk assessment should continue for the duration of treatment.”
Raj Chovatiya, MD, PhD, assistant professor of dermatology and director of the center for eczema and itch at Northwestern University, Chicago, who was asked to comment on the study results, characterized the findings as reassuring to dermatologists who may be reluctant to initiate therapy with JAK inhibitors based on concerns about safety signals for MACE, VTE, and all-cause mortality.
“These data systematically show that across medications and across conditions, there doesn’t appear to be an increased signal for these events during the short-term, placebo-controlled period which generally spans a few months in most studies,” he told this news organization. The findings, he added, “align well with our clinical experience to date for JAK inhibitor use in inflammatory skin disease. Short-term safety, particularly in relation to boxed warning events such MACE, VTE, and all-cause mortality, have generally been favorable with real-world use. It’s good to have a rigorous statistical analysis to refer to when setting patient expectations.”
However, he noted that these data only examined short-term safety during the placebo or active comparator-controlled periods. “Considering that events like MACE or VTE may take many months or years to manifest, continued long-term data generation is needed to fully answer the question of risk,” he said.
Dr. Garshick disclosed that he received grants from Pfizer and personal fees from Bristol Myers Squibb during the conduct of the study and personal fees from Kiniksa Pharmaceuticals outside the submitted work. Several other coauthors reported having advisory board roles and/or having received funding or support from several pharmaceutical companies. Dr. Chovatiya disclosed that he is a consultant to, a speaker for, investigator, and/or a member of the advisory board for several pharmaceutical companies, including those that develop JAK inhibitors.
FROM JAMA DERMATOLOGY
Phase 3 trial supports topical JAK inhibitor for AD in young children
BERLIN – as previously shown in adolescents and adults for whom it already has an approved indication.
In this study – TRUE-AD3 – systemic exposure to ruxolitinib, which is selective for JAK1 and 2, was followed closely, and the low mean plasma concentrations “suggest systemic JAK inhibition is highly unlikely,” Lawrence F. Eichenfield, MD, professor of dermatology and pediatrics at the University of California, San Diego, said at the annual congress of the European Academy of Dermatology and Venereology.
For example, at a plasma concentration no greater than 27 nM in both younger and older patients at 4 weeks and again at 8 weeks, the systemic exposure was about a tenth of that (281 nM) previously associated with myelosuppression, he reported.
Given the boxed warning for oral JAK inhibitors, which was based largely on a 2022 study in adults with rheumatoid arthritis that associated tofacitinib, a nonspecific JAK inhibitor, with an increased risk of thrombotic events in adults already at risk for these events, safety was a focus of this phase 3 trial. The boxed warning is also in the labeling for topical ruxolitinib, 1.5% (Opzelura), approved for treating to mild to moderate atopic dermatitis in patients 12 years of age and older.
Dr. Eichenfield said there were no significant safety signals in the younger pediatric population. “There were no treatment-emergent adverse events suggestive of systemic JAK inhibition,” he said. This not only included the absence of serious infections, cardiac events, thromboses, or malignancies, but there was no signal of hematologic abnormalities, such as change in hemoglobin or neutrophil count.
Application site reactions
Rather, in the study of children ages 2-11, the only adverse events associated with topical ruxolitinib not observed in the control arm, which received the vehicle alone, were application site reactions, such as pain, erythema, and irritation. None of these occurred in more than 3% of those randomized to ruxolitinib regardless of dose.
Overall, in the trial, which randomized 329 patients ages from 2 to under 12 years with mild to moderate AD to ruxolitinib 1.5% cream, ruxolitinib 0.75% cream, or vehicle in a 2:2:1 fashion, there were just two (0.8%) discontinuations in the ruxolitinib groups (one in each dosing arm). There were none in the vehicle arm.
The safety supports an expansion of the AD indication for topical ruxolitinib in young children, because the rates of response were very similar to that seen in adolescents and adults in the previously published TRUE AD-1 and TRUE AD-2 trials, he said.
For the primary endpoint of Investigator’s Global Assessment (IGA) score of 0 (clear) or 1 (almost clear) with at least a 2 grade improvement in IGA score from baseline, the response rates were 56.5%, 36.6%, and 10.8% for ruxolitinib 1.5%, ruxolitinib 0.75%, and vehicle respectively, at 8 weeks (P < .0001 for both doses relative to vehicle).
For the secondary efficacy endpoint of 75% or greater clearance on the Eczema Area and Severity Index, the rates were 67.2%, 51.5%, and 15.4%, for ruxolitinib 1.5%, ruxolitinib 0.75%, and vehicle respectively. Again, the advantage of both doses of ruxolitinib relative to vehicle was highly statistically significant (P < .0001).
Control of itch, evaluated with the Numerical Rating Scale was only evaluated in children 6-2 because of concern of the reliability of reporting in younger children. Control was defined as at least a 4-point improvement from baseline. It was achieved by 43.4%, 37.5%, and 29.7% by week 8 in the arms receiving the higher dose of ruxolitinib, the lower dose, and vehicle, respectively. The median time to achieving itch control was 11 days, 13 days, and 23 days, respectively. For all of these endpoints, the separation of the curves was readily apparent within the first 2 weeks.
The efficacy and tolerability of ruxolitinib appeared to be similar in younger children (ages 2-6) relative to older children.
Extension study in children near completion
Most of the patients who participated in TRUE AD-3 have been rolled over to the open-label extension trial, which is nearing completion. Those originally randomized to vehicle have been rerandomized to the lower or higher dose of ruxolitinib.
While this trial was focused on ruxolitinib as monotherapy, Thrasyvoulos Tzellos, MD, head of the department of dermatology, Nordland Hospital Trust, Bødo, Norway, questioned whether this is will be how it will be used in clinical practice. With the increasing array of therapies for AD, the “concept of combination therapy becomes more and more relevant,” he said after Dr. Eichenfield’s presentation.
Questioning whether an effective nonsteroidal anti-inflammatory agent like ruxolitinib should be considered a first-line treatment in mild disease or an adjunctive treatment for AD of any severity, he suggested that it might be best considered within a combination.
Dr. Eichenfield agreed. “Once we get the drug approved in a controlled trial, I think we then figure out how to use it in clinical practice.” Based on his own use of ruxolitinib in adults, he noted that he has not seen this drug replace other therapies so much as provide another option for control.
“We have an increasing armamentarium of drugs to use for involvement in different areas of the body in order to get more long-term control of disease,” he said. As an effective topical nonsteroidal drug, he believes its addition to clinical care in younger children, if approved, will be meaningful.
Dr. Eichenfield disclosed financial relationships with more multiple pharmaceutical companies, including Incyte, the manufacturer of ruxolitinib cream that provided funding for the True-AD trials. Dr. Tzellos reported financial relationships with AbbVie and UCB.
BERLIN – as previously shown in adolescents and adults for whom it already has an approved indication.
In this study – TRUE-AD3 – systemic exposure to ruxolitinib, which is selective for JAK1 and 2, was followed closely, and the low mean plasma concentrations “suggest systemic JAK inhibition is highly unlikely,” Lawrence F. Eichenfield, MD, professor of dermatology and pediatrics at the University of California, San Diego, said at the annual congress of the European Academy of Dermatology and Venereology.
For example, at a plasma concentration no greater than 27 nM in both younger and older patients at 4 weeks and again at 8 weeks, the systemic exposure was about a tenth of that (281 nM) previously associated with myelosuppression, he reported.
Given the boxed warning for oral JAK inhibitors, which was based largely on a 2022 study in adults with rheumatoid arthritis that associated tofacitinib, a nonspecific JAK inhibitor, with an increased risk of thrombotic events in adults already at risk for these events, safety was a focus of this phase 3 trial. The boxed warning is also in the labeling for topical ruxolitinib, 1.5% (Opzelura), approved for treating to mild to moderate atopic dermatitis in patients 12 years of age and older.
Dr. Eichenfield said there were no significant safety signals in the younger pediatric population. “There were no treatment-emergent adverse events suggestive of systemic JAK inhibition,” he said. This not only included the absence of serious infections, cardiac events, thromboses, or malignancies, but there was no signal of hematologic abnormalities, such as change in hemoglobin or neutrophil count.
Application site reactions
Rather, in the study of children ages 2-11, the only adverse events associated with topical ruxolitinib not observed in the control arm, which received the vehicle alone, were application site reactions, such as pain, erythema, and irritation. None of these occurred in more than 3% of those randomized to ruxolitinib regardless of dose.
Overall, in the trial, which randomized 329 patients ages from 2 to under 12 years with mild to moderate AD to ruxolitinib 1.5% cream, ruxolitinib 0.75% cream, or vehicle in a 2:2:1 fashion, there were just two (0.8%) discontinuations in the ruxolitinib groups (one in each dosing arm). There were none in the vehicle arm.
The safety supports an expansion of the AD indication for topical ruxolitinib in young children, because the rates of response were very similar to that seen in adolescents and adults in the previously published TRUE AD-1 and TRUE AD-2 trials, he said.
For the primary endpoint of Investigator’s Global Assessment (IGA) score of 0 (clear) or 1 (almost clear) with at least a 2 grade improvement in IGA score from baseline, the response rates were 56.5%, 36.6%, and 10.8% for ruxolitinib 1.5%, ruxolitinib 0.75%, and vehicle respectively, at 8 weeks (P < .0001 for both doses relative to vehicle).
For the secondary efficacy endpoint of 75% or greater clearance on the Eczema Area and Severity Index, the rates were 67.2%, 51.5%, and 15.4%, for ruxolitinib 1.5%, ruxolitinib 0.75%, and vehicle respectively. Again, the advantage of both doses of ruxolitinib relative to vehicle was highly statistically significant (P < .0001).
Control of itch, evaluated with the Numerical Rating Scale was only evaluated in children 6-2 because of concern of the reliability of reporting in younger children. Control was defined as at least a 4-point improvement from baseline. It was achieved by 43.4%, 37.5%, and 29.7% by week 8 in the arms receiving the higher dose of ruxolitinib, the lower dose, and vehicle, respectively. The median time to achieving itch control was 11 days, 13 days, and 23 days, respectively. For all of these endpoints, the separation of the curves was readily apparent within the first 2 weeks.
The efficacy and tolerability of ruxolitinib appeared to be similar in younger children (ages 2-6) relative to older children.
Extension study in children near completion
Most of the patients who participated in TRUE AD-3 have been rolled over to the open-label extension trial, which is nearing completion. Those originally randomized to vehicle have been rerandomized to the lower or higher dose of ruxolitinib.
While this trial was focused on ruxolitinib as monotherapy, Thrasyvoulos Tzellos, MD, head of the department of dermatology, Nordland Hospital Trust, Bødo, Norway, questioned whether this is will be how it will be used in clinical practice. With the increasing array of therapies for AD, the “concept of combination therapy becomes more and more relevant,” he said after Dr. Eichenfield’s presentation.
Questioning whether an effective nonsteroidal anti-inflammatory agent like ruxolitinib should be considered a first-line treatment in mild disease or an adjunctive treatment for AD of any severity, he suggested that it might be best considered within a combination.
Dr. Eichenfield agreed. “Once we get the drug approved in a controlled trial, I think we then figure out how to use it in clinical practice.” Based on his own use of ruxolitinib in adults, he noted that he has not seen this drug replace other therapies so much as provide another option for control.
“We have an increasing armamentarium of drugs to use for involvement in different areas of the body in order to get more long-term control of disease,” he said. As an effective topical nonsteroidal drug, he believes its addition to clinical care in younger children, if approved, will be meaningful.
Dr. Eichenfield disclosed financial relationships with more multiple pharmaceutical companies, including Incyte, the manufacturer of ruxolitinib cream that provided funding for the True-AD trials. Dr. Tzellos reported financial relationships with AbbVie and UCB.
BERLIN – as previously shown in adolescents and adults for whom it already has an approved indication.
In this study – TRUE-AD3 – systemic exposure to ruxolitinib, which is selective for JAK1 and 2, was followed closely, and the low mean plasma concentrations “suggest systemic JAK inhibition is highly unlikely,” Lawrence F. Eichenfield, MD, professor of dermatology and pediatrics at the University of California, San Diego, said at the annual congress of the European Academy of Dermatology and Venereology.
For example, at a plasma concentration no greater than 27 nM in both younger and older patients at 4 weeks and again at 8 weeks, the systemic exposure was about a tenth of that (281 nM) previously associated with myelosuppression, he reported.
Given the boxed warning for oral JAK inhibitors, which was based largely on a 2022 study in adults with rheumatoid arthritis that associated tofacitinib, a nonspecific JAK inhibitor, with an increased risk of thrombotic events in adults already at risk for these events, safety was a focus of this phase 3 trial. The boxed warning is also in the labeling for topical ruxolitinib, 1.5% (Opzelura), approved for treating to mild to moderate atopic dermatitis in patients 12 years of age and older.
Dr. Eichenfield said there were no significant safety signals in the younger pediatric population. “There were no treatment-emergent adverse events suggestive of systemic JAK inhibition,” he said. This not only included the absence of serious infections, cardiac events, thromboses, or malignancies, but there was no signal of hematologic abnormalities, such as change in hemoglobin or neutrophil count.
Application site reactions
Rather, in the study of children ages 2-11, the only adverse events associated with topical ruxolitinib not observed in the control arm, which received the vehicle alone, were application site reactions, such as pain, erythema, and irritation. None of these occurred in more than 3% of those randomized to ruxolitinib regardless of dose.
Overall, in the trial, which randomized 329 patients ages from 2 to under 12 years with mild to moderate AD to ruxolitinib 1.5% cream, ruxolitinib 0.75% cream, or vehicle in a 2:2:1 fashion, there were just two (0.8%) discontinuations in the ruxolitinib groups (one in each dosing arm). There were none in the vehicle arm.
The safety supports an expansion of the AD indication for topical ruxolitinib in young children, because the rates of response were very similar to that seen in adolescents and adults in the previously published TRUE AD-1 and TRUE AD-2 trials, he said.
For the primary endpoint of Investigator’s Global Assessment (IGA) score of 0 (clear) or 1 (almost clear) with at least a 2 grade improvement in IGA score from baseline, the response rates were 56.5%, 36.6%, and 10.8% for ruxolitinib 1.5%, ruxolitinib 0.75%, and vehicle respectively, at 8 weeks (P < .0001 for both doses relative to vehicle).
For the secondary efficacy endpoint of 75% or greater clearance on the Eczema Area and Severity Index, the rates were 67.2%, 51.5%, and 15.4%, for ruxolitinib 1.5%, ruxolitinib 0.75%, and vehicle respectively. Again, the advantage of both doses of ruxolitinib relative to vehicle was highly statistically significant (P < .0001).
Control of itch, evaluated with the Numerical Rating Scale was only evaluated in children 6-2 because of concern of the reliability of reporting in younger children. Control was defined as at least a 4-point improvement from baseline. It was achieved by 43.4%, 37.5%, and 29.7% by week 8 in the arms receiving the higher dose of ruxolitinib, the lower dose, and vehicle, respectively. The median time to achieving itch control was 11 days, 13 days, and 23 days, respectively. For all of these endpoints, the separation of the curves was readily apparent within the first 2 weeks.
The efficacy and tolerability of ruxolitinib appeared to be similar in younger children (ages 2-6) relative to older children.
Extension study in children near completion
Most of the patients who participated in TRUE AD-3 have been rolled over to the open-label extension trial, which is nearing completion. Those originally randomized to vehicle have been rerandomized to the lower or higher dose of ruxolitinib.
While this trial was focused on ruxolitinib as monotherapy, Thrasyvoulos Tzellos, MD, head of the department of dermatology, Nordland Hospital Trust, Bødo, Norway, questioned whether this is will be how it will be used in clinical practice. With the increasing array of therapies for AD, the “concept of combination therapy becomes more and more relevant,” he said after Dr. Eichenfield’s presentation.
Questioning whether an effective nonsteroidal anti-inflammatory agent like ruxolitinib should be considered a first-line treatment in mild disease or an adjunctive treatment for AD of any severity, he suggested that it might be best considered within a combination.
Dr. Eichenfield agreed. “Once we get the drug approved in a controlled trial, I think we then figure out how to use it in clinical practice.” Based on his own use of ruxolitinib in adults, he noted that he has not seen this drug replace other therapies so much as provide another option for control.
“We have an increasing armamentarium of drugs to use for involvement in different areas of the body in order to get more long-term control of disease,” he said. As an effective topical nonsteroidal drug, he believes its addition to clinical care in younger children, if approved, will be meaningful.
Dr. Eichenfield disclosed financial relationships with more multiple pharmaceutical companies, including Incyte, the manufacturer of ruxolitinib cream that provided funding for the True-AD trials. Dr. Tzellos reported financial relationships with AbbVie and UCB.
AT THE EADV CONGRESS
Painful axillary plaque
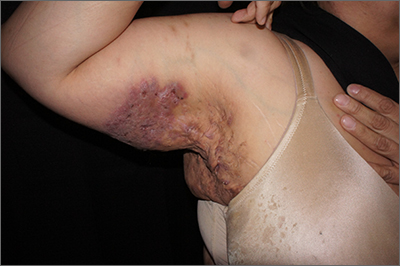
The persistent scars with recurrent abscesses and sinuses are indicative of advanced hidradenitis suppurativa. This painful and debilitating disease is characterized by the recurrent formation and inflammation of papules, cysts, sinuses, and scars in the axillae, inguinal folds, gluteal cleft, and inframammary folds. Pain, social isolation, depression, increased risk of substance abuse, and increased suicidality are all associated with hidradenitis suppurativa.
The disease may be graded based on severity, which can guide medical treatment options. The earliest stage appears similar to acne without significant sinus tract or scar formation and may be treated with topical therapies—including clindamycin 1% lotion or gel. When larger cysts associated with sinus tracts occur, systemic options with oral antibiotics (including doxycycline 100 mg bid for 3 months or combination clindamycin 300 mg and rifampin 300 mg, both bid for 3 months) are reasonable options. Intralesional triamcinolone in a concentration of 10 mg/mL injected directly into an inflamed cyst can provide acute relief. Severe disease is characterized by diffuse scars and sinus tracts. The TNF-alpha inhibitors adalimumab and infliximab are excellent options for severe disease that does not respond to antibiotics.
Surgical treatment may include either “deroofing” the sinuses or performing a wide excision of the whole area of involvement. Widely excised areas may be grafted, allowed to granulate, or closed if small enough. Although these options create significant wounds, patients experience good results; there is a 27% recurrence with deroofing and a 13% recurrence with wide excision.1
This patient underwent wide local excision of both axillae and the areas of involvement were allowed to granulate. Secondary intention healing occurred over 12 weeks.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.
1. Orenstein LAV, Nguyen TV, Damiani G, et al. Medical and surgical management of hidradenitis suppurativa: a review of international treatment guidelines and implementation in general dermatology practice. Dermatology. 2020;236:393-412. doi: 10.1159/000507323

The persistent scars with recurrent abscesses and sinuses are indicative of advanced hidradenitis suppurativa. This painful and debilitating disease is characterized by the recurrent formation and inflammation of papules, cysts, sinuses, and scars in the axillae, inguinal folds, gluteal cleft, and inframammary folds. Pain, social isolation, depression, increased risk of substance abuse, and increased suicidality are all associated with hidradenitis suppurativa.
The disease may be graded based on severity, which can guide medical treatment options. The earliest stage appears similar to acne without significant sinus tract or scar formation and may be treated with topical therapies—including clindamycin 1% lotion or gel. When larger cysts associated with sinus tracts occur, systemic options with oral antibiotics (including doxycycline 100 mg bid for 3 months or combination clindamycin 300 mg and rifampin 300 mg, both bid for 3 months) are reasonable options. Intralesional triamcinolone in a concentration of 10 mg/mL injected directly into an inflamed cyst can provide acute relief. Severe disease is characterized by diffuse scars and sinus tracts. The TNF-alpha inhibitors adalimumab and infliximab are excellent options for severe disease that does not respond to antibiotics.
Surgical treatment may include either “deroofing” the sinuses or performing a wide excision of the whole area of involvement. Widely excised areas may be grafted, allowed to granulate, or closed if small enough. Although these options create significant wounds, patients experience good results; there is a 27% recurrence with deroofing and a 13% recurrence with wide excision.1
This patient underwent wide local excision of both axillae and the areas of involvement were allowed to granulate. Secondary intention healing occurred over 12 weeks.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.

The persistent scars with recurrent abscesses and sinuses are indicative of advanced hidradenitis suppurativa. This painful and debilitating disease is characterized by the recurrent formation and inflammation of papules, cysts, sinuses, and scars in the axillae, inguinal folds, gluteal cleft, and inframammary folds. Pain, social isolation, depression, increased risk of substance abuse, and increased suicidality are all associated with hidradenitis suppurativa.
The disease may be graded based on severity, which can guide medical treatment options. The earliest stage appears similar to acne without significant sinus tract or scar formation and may be treated with topical therapies—including clindamycin 1% lotion or gel. When larger cysts associated with sinus tracts occur, systemic options with oral antibiotics (including doxycycline 100 mg bid for 3 months or combination clindamycin 300 mg and rifampin 300 mg, both bid for 3 months) are reasonable options. Intralesional triamcinolone in a concentration of 10 mg/mL injected directly into an inflamed cyst can provide acute relief. Severe disease is characterized by diffuse scars and sinus tracts. The TNF-alpha inhibitors adalimumab and infliximab are excellent options for severe disease that does not respond to antibiotics.
Surgical treatment may include either “deroofing” the sinuses or performing a wide excision of the whole area of involvement. Widely excised areas may be grafted, allowed to granulate, or closed if small enough. Although these options create significant wounds, patients experience good results; there is a 27% recurrence with deroofing and a 13% recurrence with wide excision.1
This patient underwent wide local excision of both axillae and the areas of involvement were allowed to granulate. Secondary intention healing occurred over 12 weeks.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.
1. Orenstein LAV, Nguyen TV, Damiani G, et al. Medical and surgical management of hidradenitis suppurativa: a review of international treatment guidelines and implementation in general dermatology practice. Dermatology. 2020;236:393-412. doi: 10.1159/000507323
1. Orenstein LAV, Nguyen TV, Damiani G, et al. Medical and surgical management of hidradenitis suppurativa: a review of international treatment guidelines and implementation in general dermatology practice. Dermatology. 2020;236:393-412. doi: 10.1159/000507323