User login
Vitiligo
THE COMPARISON
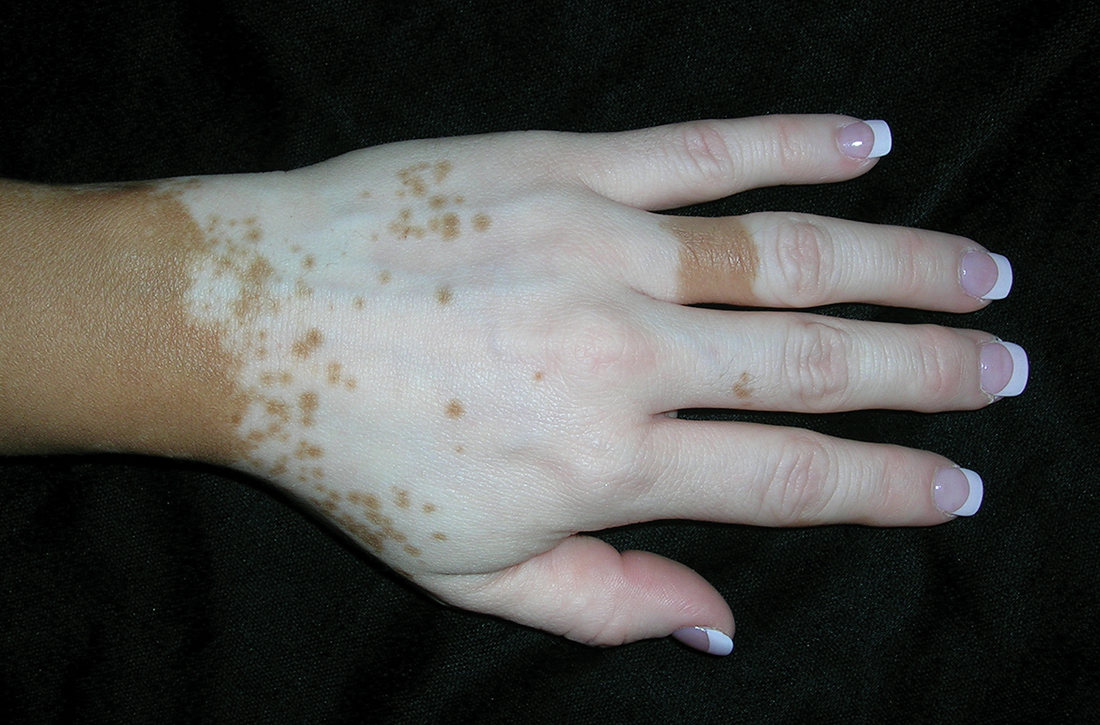
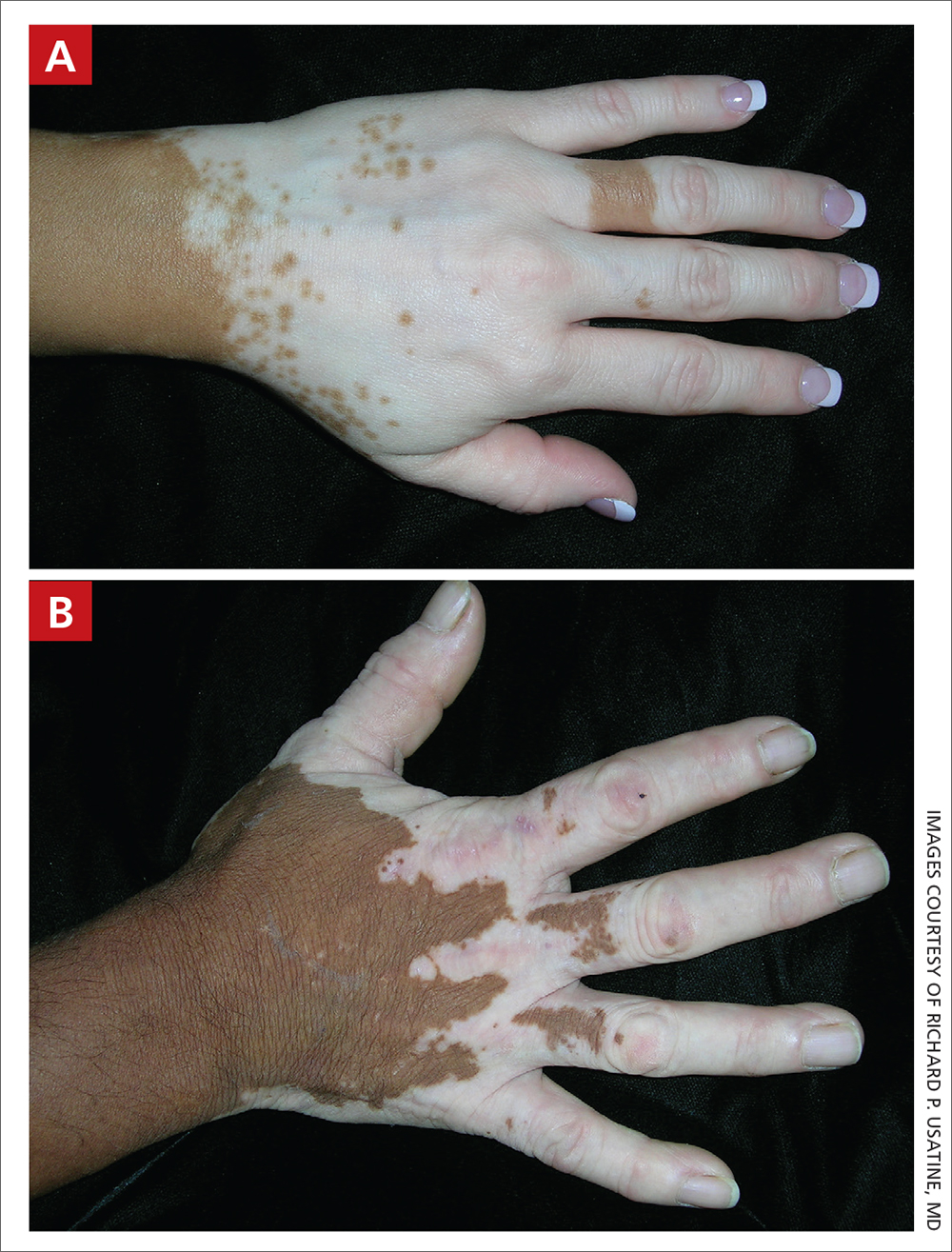
A Vitiligo in a young Hispanic female, which spared the area under a ring. The patient has spotty return of pigment on the hand after narrowband ultraviolet B (UVB) treatment.
B Vitiligo on the hand in a young Hispanic male.
Vitiligo is a chronic autoimmune disorder characterized by areas of depigmented white patches on the skin due to the loss of melanocytes in the epidermis. Various theories on the pathogenesis of vitiligo exist; however, autoimmune destruction of melanocytes remains the leading hypothesis, followed by intrinsic defects in melanocytes.1
Vitiligo is associated with various autoimmune diseases but is most frequently reported in conjunction with thyroid disorders.2
Epidemiology
Vitiligo affects approximately 1% of the US population and up to 8% worldwide.2 There is no difference in prevalence between races or genders. Females typically acquire the disease earlier than males. Onset may occur at any age, although about half of patients will have vitiligo by 20 years of age.1
Key clinical features in people with darker skin tones
Bright white patches are characteristic of vitiligo. The patches typically are asymptomatic and often affect the hands (FIGURES A and B), perioral skin, feet, and scalp, as well as areas more vulnerable to friction and trauma, such as the elbows and knees.2 Trichrome lesions—consisting of varying zones of white (depigmented), lighter brown (hypopigmented), and normal skin—are most commonly seen in individuals with darker skin. Trichrome vitiligo is considered an actively progressing variant of vitiligo.2
An important distinction when making the diagnosis is evaluating for segmental vs nonsegmental vitiligo. Although nonsegmental vitiligo—the more common subtype—is characterized by symmetric distribution and a less predictable course, segmental vitiligo manifests in a localized and unilateral distribution, often avoiding extension past the midline. Segmental vitiligo typically manifests at a younger age and follows a more rapidly stabilizing course.3
Worth noting
Given that stark contrasts between pigmented and depigmented lesions are more prominent in darker skin tones, vitiligo can be more socially stigmatizing and psychologically devastating in these patients.4,5
Continue to: Treatment of vitiligo...
Treatment of vitiligo includes narrowband UVB (NB-UVB) light phototherapy, excimer laser, topical corticosteroids, topical calcineurin inhibitors such as tacrolimus and pimecrolimus, and surgical melanocyte transplantation.1 In July 2022, ruxolitinib cream 1.5% was approved by the US Food and Drug Administration (FDA) for nonsegmental vitiligo in patients ages 12 years and older.6,7 It is the only FDA-approved therapy for vitiligo. It is thought to work by inhibiting the Janus kinase–signal transducers and activators of the transcription pathway.6 However, topical ruxolitinib is expensive, costing more than $2000 for 60 g.8
Health disparity highlight
A 2021 study reviewing the coverage policies of 15 commercial health care insurance companies, 50 BlueCross BlueShield plans, Medicaid, Medicare, and Veterans Affairs plans found inequities in the insurance coverage patterns for therapies used to treat vitiligo. There were 2 commonly cited reasons for denying coverage for therapies: vitiligo was considered cosmetic and therapies were not FDA approved.7 In comparison, NB-UVB light phototherapy for psoriasis is not considered cosmetic and has a much higher insurance coverage rate.9,10 The out-of-pocket cost for a patient to purchase their own NB-UVB light phototherapy is more than $5000.11 Not all patients of color are economically disadvantaged, but in the United States, Black and Hispanic populations experience disproportionately higher rates of poverty (19% and 17%, respectively) compared to their White counterparts (8%).12
Final thoughts
FDA approval of new drugs or new treatment indications comes after years of research discovery and large-scale trials. This pursuit of new discovery, however, is uneven. Vitiligo has historically been understudied and underfunded for research; this is common among several conditions adversely affecting people of color in the United States.13
1. Rashighi M, Harris JE. Vitiligo pathogenesis and emerging treatments. Dermatol Clin. 2017;35:257-265. doi: 10.1016/j.det. 2016.11.014
2. Alikhan A, Felsten LM, Daly M, et al. Vitiligo: a comprehensive overview part I. introduction, epidemiology, quality of life, diagnosis, differential diagnosis, associations, histopathology, etiology, and work-up. J Am Acad Dermatol. 2011;65:473-491. doi: 10.1016/j.jaad.2010.11.061
3. van Geel N, Speeckaert R. Segmental vitiligo. Dermatol Clin. 2017; 35:145-150. doi: 10.1016/j.det.2016.11.005
4. Grimes PE, Miller MM. Vitiligo: patient stories, self-esteem, and the psychological burden of disease. Int J Womens Dermatol. 2018;4:32-37. doi: 10.1016/j.ijwd.2017.11.005
5. Ezzedine K, Eleftheriadou V, Jones H, et al. Psychosocial effects of vitiligo: a systematic literature review. Am J Clin Dermatol. 2021; 22:757-774. doi: 10.1007/s40257-021-00631-6
6. FDA approves topical treatment addressing repigmentation in vitiligo in patients aged 12 and older. News release. US Food and Drug Administration; July 19, 2022. Accessed December 27, 2022. www.fda.gov/drugs/news-events-human-drugs/fda-approves-topical- treatment-addressing-repigmentation-vitiligo-patients-aged- 12-and-older
7. Blundell A, Sachar M, Gabel CK, et al. The scope of health insurance coverage of vitiligo treatments in the United States: implications for health care outcomes and disparities in children of color. Pediatr Dermatol. 2021;38(suppl 2):79-85. doi: 10.1111/ pde.14714
8. Opzelura prices, coupons, and patient assistance programs. Drugs.com. Accessed January 10, 2023. www.drugs.com/priceguide/opzelura
9. Bhutani T, Liao W. A practical approach to home UVB phototherapy for the treatment of generalized psoriasis. Pract Dermatol. 2010;7:31-35.
10. Castro Porto Silva Lopes F, Ahmed A. Insurance coverage for phototherapy for vitiligo in comparison to psoriasis and atopic dermatitis. SKIN The Journal of Cutaneous Medicine. 2022;6:217-224. doi: 10.25251/skin.6.3.6
11. Smith MP, Ly K, Thibodeaux Q, et al. Home phototherapy for patients with vitiligo: challenges and solutions. Clin Cosmet Investig Dermatol. 2019;12:451-459. doi: 10.2147/CCID.S185798
12. Shrider EA, Kollar M, Chen F, et al. Income and poverty in the United States: 2020. US Census Bureau. September 14, 2021. Accessed December 27, 2022. www.census.gov/library/publications/2021/demo/p60-273.html
13. Whitton ME, Pinart M, Batchelor J, et al. Interventions for vitiligo. Cochrane Database Syst Rev. 2010;(1):CD003263. doi: 10.1002/14651858.CD003263.pub4
THE COMPARISON
A Vitiligo in a young Hispanic female, which spared the area under a ring. The patient has spotty return of pigment on the hand after narrowband ultraviolet B (UVB) treatment.
B Vitiligo on the hand in a young Hispanic male.

Vitiligo is a chronic autoimmune disorder characterized by areas of depigmented white patches on the skin due to the loss of melanocytes in the epidermis. Various theories on the pathogenesis of vitiligo exist; however, autoimmune destruction of melanocytes remains the leading hypothesis, followed by intrinsic defects in melanocytes.1
Vitiligo is associated with various autoimmune diseases but is most frequently reported in conjunction with thyroid disorders.2
Epidemiology
Vitiligo affects approximately 1% of the US population and up to 8% worldwide.2 There is no difference in prevalence between races or genders. Females typically acquire the disease earlier than males. Onset may occur at any age, although about half of patients will have vitiligo by 20 years of age.1
Key clinical features in people with darker skin tones
Bright white patches are characteristic of vitiligo. The patches typically are asymptomatic and often affect the hands (FIGURES A and B), perioral skin, feet, and scalp, as well as areas more vulnerable to friction and trauma, such as the elbows and knees.2 Trichrome lesions—consisting of varying zones of white (depigmented), lighter brown (hypopigmented), and normal skin—are most commonly seen in individuals with darker skin. Trichrome vitiligo is considered an actively progressing variant of vitiligo.2
An important distinction when making the diagnosis is evaluating for segmental vs nonsegmental vitiligo. Although nonsegmental vitiligo—the more common subtype—is characterized by symmetric distribution and a less predictable course, segmental vitiligo manifests in a localized and unilateral distribution, often avoiding extension past the midline. Segmental vitiligo typically manifests at a younger age and follows a more rapidly stabilizing course.3
Worth noting
Given that stark contrasts between pigmented and depigmented lesions are more prominent in darker skin tones, vitiligo can be more socially stigmatizing and psychologically devastating in these patients.4,5
Continue to: Treatment of vitiligo...
Treatment of vitiligo includes narrowband UVB (NB-UVB) light phototherapy, excimer laser, topical corticosteroids, topical calcineurin inhibitors such as tacrolimus and pimecrolimus, and surgical melanocyte transplantation.1 In July 2022, ruxolitinib cream 1.5% was approved by the US Food and Drug Administration (FDA) for nonsegmental vitiligo in patients ages 12 years and older.6,7 It is the only FDA-approved therapy for vitiligo. It is thought to work by inhibiting the Janus kinase–signal transducers and activators of the transcription pathway.6 However, topical ruxolitinib is expensive, costing more than $2000 for 60 g.8
Health disparity highlight
A 2021 study reviewing the coverage policies of 15 commercial health care insurance companies, 50 BlueCross BlueShield plans, Medicaid, Medicare, and Veterans Affairs plans found inequities in the insurance coverage patterns for therapies used to treat vitiligo. There were 2 commonly cited reasons for denying coverage for therapies: vitiligo was considered cosmetic and therapies were not FDA approved.7 In comparison, NB-UVB light phototherapy for psoriasis is not considered cosmetic and has a much higher insurance coverage rate.9,10 The out-of-pocket cost for a patient to purchase their own NB-UVB light phototherapy is more than $5000.11 Not all patients of color are economically disadvantaged, but in the United States, Black and Hispanic populations experience disproportionately higher rates of poverty (19% and 17%, respectively) compared to their White counterparts (8%).12
Final thoughts
FDA approval of new drugs or new treatment indications comes after years of research discovery and large-scale trials. This pursuit of new discovery, however, is uneven. Vitiligo has historically been understudied and underfunded for research; this is common among several conditions adversely affecting people of color in the United States.13
THE COMPARISON
A Vitiligo in a young Hispanic female, which spared the area under a ring. The patient has spotty return of pigment on the hand after narrowband ultraviolet B (UVB) treatment.
B Vitiligo on the hand in a young Hispanic male.

Vitiligo is a chronic autoimmune disorder characterized by areas of depigmented white patches on the skin due to the loss of melanocytes in the epidermis. Various theories on the pathogenesis of vitiligo exist; however, autoimmune destruction of melanocytes remains the leading hypothesis, followed by intrinsic defects in melanocytes.1
Vitiligo is associated with various autoimmune diseases but is most frequently reported in conjunction with thyroid disorders.2
Epidemiology
Vitiligo affects approximately 1% of the US population and up to 8% worldwide.2 There is no difference in prevalence between races or genders. Females typically acquire the disease earlier than males. Onset may occur at any age, although about half of patients will have vitiligo by 20 years of age.1
Key clinical features in people with darker skin tones
Bright white patches are characteristic of vitiligo. The patches typically are asymptomatic and often affect the hands (FIGURES A and B), perioral skin, feet, and scalp, as well as areas more vulnerable to friction and trauma, such as the elbows and knees.2 Trichrome lesions—consisting of varying zones of white (depigmented), lighter brown (hypopigmented), and normal skin—are most commonly seen in individuals with darker skin. Trichrome vitiligo is considered an actively progressing variant of vitiligo.2
An important distinction when making the diagnosis is evaluating for segmental vs nonsegmental vitiligo. Although nonsegmental vitiligo—the more common subtype—is characterized by symmetric distribution and a less predictable course, segmental vitiligo manifests in a localized and unilateral distribution, often avoiding extension past the midline. Segmental vitiligo typically manifests at a younger age and follows a more rapidly stabilizing course.3
Worth noting
Given that stark contrasts between pigmented and depigmented lesions are more prominent in darker skin tones, vitiligo can be more socially stigmatizing and psychologically devastating in these patients.4,5
Continue to: Treatment of vitiligo...
Treatment of vitiligo includes narrowband UVB (NB-UVB) light phototherapy, excimer laser, topical corticosteroids, topical calcineurin inhibitors such as tacrolimus and pimecrolimus, and surgical melanocyte transplantation.1 In July 2022, ruxolitinib cream 1.5% was approved by the US Food and Drug Administration (FDA) for nonsegmental vitiligo in patients ages 12 years and older.6,7 It is the only FDA-approved therapy for vitiligo. It is thought to work by inhibiting the Janus kinase–signal transducers and activators of the transcription pathway.6 However, topical ruxolitinib is expensive, costing more than $2000 for 60 g.8
Health disparity highlight
A 2021 study reviewing the coverage policies of 15 commercial health care insurance companies, 50 BlueCross BlueShield plans, Medicaid, Medicare, and Veterans Affairs plans found inequities in the insurance coverage patterns for therapies used to treat vitiligo. There were 2 commonly cited reasons for denying coverage for therapies: vitiligo was considered cosmetic and therapies were not FDA approved.7 In comparison, NB-UVB light phototherapy for psoriasis is not considered cosmetic and has a much higher insurance coverage rate.9,10 The out-of-pocket cost for a patient to purchase their own NB-UVB light phototherapy is more than $5000.11 Not all patients of color are economically disadvantaged, but in the United States, Black and Hispanic populations experience disproportionately higher rates of poverty (19% and 17%, respectively) compared to their White counterparts (8%).12
Final thoughts
FDA approval of new drugs or new treatment indications comes after years of research discovery and large-scale trials. This pursuit of new discovery, however, is uneven. Vitiligo has historically been understudied and underfunded for research; this is common among several conditions adversely affecting people of color in the United States.13
1. Rashighi M, Harris JE. Vitiligo pathogenesis and emerging treatments. Dermatol Clin. 2017;35:257-265. doi: 10.1016/j.det. 2016.11.014
2. Alikhan A, Felsten LM, Daly M, et al. Vitiligo: a comprehensive overview part I. introduction, epidemiology, quality of life, diagnosis, differential diagnosis, associations, histopathology, etiology, and work-up. J Am Acad Dermatol. 2011;65:473-491. doi: 10.1016/j.jaad.2010.11.061
3. van Geel N, Speeckaert R. Segmental vitiligo. Dermatol Clin. 2017; 35:145-150. doi: 10.1016/j.det.2016.11.005
4. Grimes PE, Miller MM. Vitiligo: patient stories, self-esteem, and the psychological burden of disease. Int J Womens Dermatol. 2018;4:32-37. doi: 10.1016/j.ijwd.2017.11.005
5. Ezzedine K, Eleftheriadou V, Jones H, et al. Psychosocial effects of vitiligo: a systematic literature review. Am J Clin Dermatol. 2021; 22:757-774. doi: 10.1007/s40257-021-00631-6
6. FDA approves topical treatment addressing repigmentation in vitiligo in patients aged 12 and older. News release. US Food and Drug Administration; July 19, 2022. Accessed December 27, 2022. www.fda.gov/drugs/news-events-human-drugs/fda-approves-topical- treatment-addressing-repigmentation-vitiligo-patients-aged- 12-and-older
7. Blundell A, Sachar M, Gabel CK, et al. The scope of health insurance coverage of vitiligo treatments in the United States: implications for health care outcomes and disparities in children of color. Pediatr Dermatol. 2021;38(suppl 2):79-85. doi: 10.1111/ pde.14714
8. Opzelura prices, coupons, and patient assistance programs. Drugs.com. Accessed January 10, 2023. www.drugs.com/priceguide/opzelura
9. Bhutani T, Liao W. A practical approach to home UVB phototherapy for the treatment of generalized psoriasis. Pract Dermatol. 2010;7:31-35.
10. Castro Porto Silva Lopes F, Ahmed A. Insurance coverage for phototherapy for vitiligo in comparison to psoriasis and atopic dermatitis. SKIN The Journal of Cutaneous Medicine. 2022;6:217-224. doi: 10.25251/skin.6.3.6
11. Smith MP, Ly K, Thibodeaux Q, et al. Home phototherapy for patients with vitiligo: challenges and solutions. Clin Cosmet Investig Dermatol. 2019;12:451-459. doi: 10.2147/CCID.S185798
12. Shrider EA, Kollar M, Chen F, et al. Income and poverty in the United States: 2020. US Census Bureau. September 14, 2021. Accessed December 27, 2022. www.census.gov/library/publications/2021/demo/p60-273.html
13. Whitton ME, Pinart M, Batchelor J, et al. Interventions for vitiligo. Cochrane Database Syst Rev. 2010;(1):CD003263. doi: 10.1002/14651858.CD003263.pub4
1. Rashighi M, Harris JE. Vitiligo pathogenesis and emerging treatments. Dermatol Clin. 2017;35:257-265. doi: 10.1016/j.det. 2016.11.014
2. Alikhan A, Felsten LM, Daly M, et al. Vitiligo: a comprehensive overview part I. introduction, epidemiology, quality of life, diagnosis, differential diagnosis, associations, histopathology, etiology, and work-up. J Am Acad Dermatol. 2011;65:473-491. doi: 10.1016/j.jaad.2010.11.061
3. van Geel N, Speeckaert R. Segmental vitiligo. Dermatol Clin. 2017; 35:145-150. doi: 10.1016/j.det.2016.11.005
4. Grimes PE, Miller MM. Vitiligo: patient stories, self-esteem, and the psychological burden of disease. Int J Womens Dermatol. 2018;4:32-37. doi: 10.1016/j.ijwd.2017.11.005
5. Ezzedine K, Eleftheriadou V, Jones H, et al. Psychosocial effects of vitiligo: a systematic literature review. Am J Clin Dermatol. 2021; 22:757-774. doi: 10.1007/s40257-021-00631-6
6. FDA approves topical treatment addressing repigmentation in vitiligo in patients aged 12 and older. News release. US Food and Drug Administration; July 19, 2022. Accessed December 27, 2022. www.fda.gov/drugs/news-events-human-drugs/fda-approves-topical- treatment-addressing-repigmentation-vitiligo-patients-aged- 12-and-older
7. Blundell A, Sachar M, Gabel CK, et al. The scope of health insurance coverage of vitiligo treatments in the United States: implications for health care outcomes and disparities in children of color. Pediatr Dermatol. 2021;38(suppl 2):79-85. doi: 10.1111/ pde.14714
8. Opzelura prices, coupons, and patient assistance programs. Drugs.com. Accessed January 10, 2023. www.drugs.com/priceguide/opzelura
9. Bhutani T, Liao W. A practical approach to home UVB phototherapy for the treatment of generalized psoriasis. Pract Dermatol. 2010;7:31-35.
10. Castro Porto Silva Lopes F, Ahmed A. Insurance coverage for phototherapy for vitiligo in comparison to psoriasis and atopic dermatitis. SKIN The Journal of Cutaneous Medicine. 2022;6:217-224. doi: 10.25251/skin.6.3.6
11. Smith MP, Ly K, Thibodeaux Q, et al. Home phototherapy for patients with vitiligo: challenges and solutions. Clin Cosmet Investig Dermatol. 2019;12:451-459. doi: 10.2147/CCID.S185798
12. Shrider EA, Kollar M, Chen F, et al. Income and poverty in the United States: 2020. US Census Bureau. September 14, 2021. Accessed December 27, 2022. www.census.gov/library/publications/2021/demo/p60-273.html
13. Whitton ME, Pinart M, Batchelor J, et al. Interventions for vitiligo. Cochrane Database Syst Rev. 2010;(1):CD003263. doi: 10.1002/14651858.CD003263.pub4
Pigmentary disorder experts welcome research explosion
ORLANDO – , a panel of experts said in a session on this topic at the ODAC Dermatology, Aesthetic & Surgical Conference.
The arrival of ruxolitinib cream, a topical JAK inhibitor – and oral JAK inhibitors, including ritlecitinib, a JAK3/TEC (tyrosine kinase expressed in hepatocellular carcinoma) inhibitor in clinical trials – is a welcome development for treatment of vitiligo, said John E. Harris, MD, PhD, chair of dermatology and director of the Vitiligo Clinic and Research Center at the University of Massachusetts, Worcester. Also in the pipeline is a kit for melanocyte-keratinocyte transplantation, which involves transplanting epidermal cells from one part of the body to another. This can be a challenging procedure but a kit would make it easier for a wider range of practitioners. (Topical ruxolitinib was approved by the Food and Drug Administration for treating nonsegmental vitiligo in July, 2022.)
“In the last 10 years, it’s just blown up and people care about vitiligo now,” Dr. Harris said, noting that vitiligo is more than a cosmetic issue, like gray hair or wrinkles. “Vitiligo is an autoimmune disease and now is being treated as such.”
Nada Elbuluk, MD, MSc, associate professor of dermatology at the University of Southern California, Los Angeles, said she’s pleased at the increasing availability of treatment options for hyperpigmentation, aside from hydroquinone, which is associated with an increased risk of adverse effects.
“We have more and more nonhydroquinone agents ... which is really nice because it expands our treatment armamentarium and what we can use to cycle people off of hydroquinone,” she said.
Some of these options include tranexamic acid and products containing azelaic acid or vitamin C.
Iltefat H. Hamzavi, MD, senior staff physician at Henry Ford Health System, Detroit, said that pigmentary disorders are receiving the recognition they deserve.
“I’m excited just about the intersection of society and science, the awareness that pigmentary abnormalities mean something, and they mean something across our society,” he said.
Dr. Elbuluk said that hyperpigmentation has “profound effects on quality of life” for patients.
“They are often more bothered by the darkening of the skin than the primary process that caused it,” she said. “It’s not uncommon that the chief complaint will say ‘dark spots’ and I walk in a room and it’s a patient who has acne. They don’t even say they’re here for acne. They just put ‘dark spots’ [down] because that’s what bothers them. That’s what lasts for so long after the acne is gone.”
The experts offered suggestions for managing these cases. Among her tips, Dr. Elbuluk said that for hyperpigmentation, physicians should not be afraid to biopsy the face – but suggested small, 2-millimeter specimens. In addition, “you can get common conditions in uncommon places,” she noted. “If you see something that looks like melasma off the face, it actually could be, so keep that in your differential.”
Dr. Hamzavi, who spoke about hypopigmentation disorders, said clinicians need to use an algorithm for diagnosis, considering features such as localized or diffuse, scale or no scale, as well as patient history, and other factors. For instance, a hypopigmented area that is localized and has a reddish central papule might lead a clinician to a diagnosis of hypopigmented sarcoidosis.
Using the algorithms, “you actually have to categorize these and then use your own experience. ... All of these elements can help you become a really good taxonomist – ultimately that’s what physicians are.”
He said it’s also important to know when it’s time to reconsider a diagnosis, such as when patients do not respond to traditional treatments. “If they don’t respond, re-categorize,” he said.
Speaking about vitiligo, Dr. Harris said it’s crucial to differentiate active vitiligo from inactive vitiligo and if it’s active, steps need to be taken to keep it from worsening..
Four signs of active vitiligo are a “confetti” pattern of clustered tiny macules of depigmentation, which will coalesce quickly into huge patches; tri-chrome vitiligo that includes a hypopigmented zone; linear areas of depigmentation (Koebner’s phenomenon) that look like scratches on the skin; and inflammatory vitiligo, with an erythematous ring around the edges of a depigmented area.
Dr. Harris disclosed ties with Incyte, Pfizer, Abbvie, Genzyme/Sanofi and other companies. Dr. Elbuluk disclosed ties with Avita, Incyte, Beiersdorf, and other companies. Dr. Hamzavi disclosed ties with AbbVie, Pfizer, Incyte, and other companies.
ORLANDO – , a panel of experts said in a session on this topic at the ODAC Dermatology, Aesthetic & Surgical Conference.
The arrival of ruxolitinib cream, a topical JAK inhibitor – and oral JAK inhibitors, including ritlecitinib, a JAK3/TEC (tyrosine kinase expressed in hepatocellular carcinoma) inhibitor in clinical trials – is a welcome development for treatment of vitiligo, said John E. Harris, MD, PhD, chair of dermatology and director of the Vitiligo Clinic and Research Center at the University of Massachusetts, Worcester. Also in the pipeline is a kit for melanocyte-keratinocyte transplantation, which involves transplanting epidermal cells from one part of the body to another. This can be a challenging procedure but a kit would make it easier for a wider range of practitioners. (Topical ruxolitinib was approved by the Food and Drug Administration for treating nonsegmental vitiligo in July, 2022.)
“In the last 10 years, it’s just blown up and people care about vitiligo now,” Dr. Harris said, noting that vitiligo is more than a cosmetic issue, like gray hair or wrinkles. “Vitiligo is an autoimmune disease and now is being treated as such.”
Nada Elbuluk, MD, MSc, associate professor of dermatology at the University of Southern California, Los Angeles, said she’s pleased at the increasing availability of treatment options for hyperpigmentation, aside from hydroquinone, which is associated with an increased risk of adverse effects.
“We have more and more nonhydroquinone agents ... which is really nice because it expands our treatment armamentarium and what we can use to cycle people off of hydroquinone,” she said.
Some of these options include tranexamic acid and products containing azelaic acid or vitamin C.
Iltefat H. Hamzavi, MD, senior staff physician at Henry Ford Health System, Detroit, said that pigmentary disorders are receiving the recognition they deserve.
“I’m excited just about the intersection of society and science, the awareness that pigmentary abnormalities mean something, and they mean something across our society,” he said.
Dr. Elbuluk said that hyperpigmentation has “profound effects on quality of life” for patients.
“They are often more bothered by the darkening of the skin than the primary process that caused it,” she said. “It’s not uncommon that the chief complaint will say ‘dark spots’ and I walk in a room and it’s a patient who has acne. They don’t even say they’re here for acne. They just put ‘dark spots’ [down] because that’s what bothers them. That’s what lasts for so long after the acne is gone.”
The experts offered suggestions for managing these cases. Among her tips, Dr. Elbuluk said that for hyperpigmentation, physicians should not be afraid to biopsy the face – but suggested small, 2-millimeter specimens. In addition, “you can get common conditions in uncommon places,” she noted. “If you see something that looks like melasma off the face, it actually could be, so keep that in your differential.”
Dr. Hamzavi, who spoke about hypopigmentation disorders, said clinicians need to use an algorithm for diagnosis, considering features such as localized or diffuse, scale or no scale, as well as patient history, and other factors. For instance, a hypopigmented area that is localized and has a reddish central papule might lead a clinician to a diagnosis of hypopigmented sarcoidosis.
Using the algorithms, “you actually have to categorize these and then use your own experience. ... All of these elements can help you become a really good taxonomist – ultimately that’s what physicians are.”
He said it’s also important to know when it’s time to reconsider a diagnosis, such as when patients do not respond to traditional treatments. “If they don’t respond, re-categorize,” he said.
Speaking about vitiligo, Dr. Harris said it’s crucial to differentiate active vitiligo from inactive vitiligo and if it’s active, steps need to be taken to keep it from worsening..
Four signs of active vitiligo are a “confetti” pattern of clustered tiny macules of depigmentation, which will coalesce quickly into huge patches; tri-chrome vitiligo that includes a hypopigmented zone; linear areas of depigmentation (Koebner’s phenomenon) that look like scratches on the skin; and inflammatory vitiligo, with an erythematous ring around the edges of a depigmented area.
Dr. Harris disclosed ties with Incyte, Pfizer, Abbvie, Genzyme/Sanofi and other companies. Dr. Elbuluk disclosed ties with Avita, Incyte, Beiersdorf, and other companies. Dr. Hamzavi disclosed ties with AbbVie, Pfizer, Incyte, and other companies.
ORLANDO – , a panel of experts said in a session on this topic at the ODAC Dermatology, Aesthetic & Surgical Conference.
The arrival of ruxolitinib cream, a topical JAK inhibitor – and oral JAK inhibitors, including ritlecitinib, a JAK3/TEC (tyrosine kinase expressed in hepatocellular carcinoma) inhibitor in clinical trials – is a welcome development for treatment of vitiligo, said John E. Harris, MD, PhD, chair of dermatology and director of the Vitiligo Clinic and Research Center at the University of Massachusetts, Worcester. Also in the pipeline is a kit for melanocyte-keratinocyte transplantation, which involves transplanting epidermal cells from one part of the body to another. This can be a challenging procedure but a kit would make it easier for a wider range of practitioners. (Topical ruxolitinib was approved by the Food and Drug Administration for treating nonsegmental vitiligo in July, 2022.)
“In the last 10 years, it’s just blown up and people care about vitiligo now,” Dr. Harris said, noting that vitiligo is more than a cosmetic issue, like gray hair or wrinkles. “Vitiligo is an autoimmune disease and now is being treated as such.”
Nada Elbuluk, MD, MSc, associate professor of dermatology at the University of Southern California, Los Angeles, said she’s pleased at the increasing availability of treatment options for hyperpigmentation, aside from hydroquinone, which is associated with an increased risk of adverse effects.
“We have more and more nonhydroquinone agents ... which is really nice because it expands our treatment armamentarium and what we can use to cycle people off of hydroquinone,” she said.
Some of these options include tranexamic acid and products containing azelaic acid or vitamin C.
Iltefat H. Hamzavi, MD, senior staff physician at Henry Ford Health System, Detroit, said that pigmentary disorders are receiving the recognition they deserve.
“I’m excited just about the intersection of society and science, the awareness that pigmentary abnormalities mean something, and they mean something across our society,” he said.
Dr. Elbuluk said that hyperpigmentation has “profound effects on quality of life” for patients.
“They are often more bothered by the darkening of the skin than the primary process that caused it,” she said. “It’s not uncommon that the chief complaint will say ‘dark spots’ and I walk in a room and it’s a patient who has acne. They don’t even say they’re here for acne. They just put ‘dark spots’ [down] because that’s what bothers them. That’s what lasts for so long after the acne is gone.”
The experts offered suggestions for managing these cases. Among her tips, Dr. Elbuluk said that for hyperpigmentation, physicians should not be afraid to biopsy the face – but suggested small, 2-millimeter specimens. In addition, “you can get common conditions in uncommon places,” she noted. “If you see something that looks like melasma off the face, it actually could be, so keep that in your differential.”
Dr. Hamzavi, who spoke about hypopigmentation disorders, said clinicians need to use an algorithm for diagnosis, considering features such as localized or diffuse, scale or no scale, as well as patient history, and other factors. For instance, a hypopigmented area that is localized and has a reddish central papule might lead a clinician to a diagnosis of hypopigmented sarcoidosis.
Using the algorithms, “you actually have to categorize these and then use your own experience. ... All of these elements can help you become a really good taxonomist – ultimately that’s what physicians are.”
He said it’s also important to know when it’s time to reconsider a diagnosis, such as when patients do not respond to traditional treatments. “If they don’t respond, re-categorize,” he said.
Speaking about vitiligo, Dr. Harris said it’s crucial to differentiate active vitiligo from inactive vitiligo and if it’s active, steps need to be taken to keep it from worsening..
Four signs of active vitiligo are a “confetti” pattern of clustered tiny macules of depigmentation, which will coalesce quickly into huge patches; tri-chrome vitiligo that includes a hypopigmented zone; linear areas of depigmentation (Koebner’s phenomenon) that look like scratches on the skin; and inflammatory vitiligo, with an erythematous ring around the edges of a depigmented area.
Dr. Harris disclosed ties with Incyte, Pfizer, Abbvie, Genzyme/Sanofi and other companies. Dr. Elbuluk disclosed ties with Avita, Incyte, Beiersdorf, and other companies. Dr. Hamzavi disclosed ties with AbbVie, Pfizer, Incyte, and other companies.
AT ODAC 2023
Long-pulsed 1,064 nm Nd:YAG for nonaggressive BCC ‘effective and easy’
SAN DIEGO – After Arisa E. Ortiz, MD, and colleagues published results of a multicenter study reporting that one treatment with the long-pulsed 1,064-nm Nd:YAG laser cleared nonaggressive basal cell carcinoma (BCC) on the trunk and extremities in 90% of patients, she heard from colleagues who were skeptical of the approach.
Maybe it’s just the biopsy alone that’s clearing these tumors, some told her. Others postulated that since the energy was delivered with a 5- to 6-mm spot size at a fluence of 125-140 J/cm2 and a 7- to 10-ms pulse duration, bulk heating likely disrupted the tumors. However, treatments were generally well tolerated, required no anesthesia, and caused no significant adverse events.
“It’s almost scarless,” Dr. Ortiz, director of laser and cosmetic dermatology at the University of California, San Diego, said at the annual Masters of Aesthetics Symposium. “Sometimes the treatment does leave a mark, but I think the scars are always acceptable. We do have good histologic evidence that we can penetrate 2.15 mm, which is a lot deeper than what the pulsed-dye laser or other superficial wavelengths are able to penetrate.”
Data is well powered to reject the null hypothesis that laser treatment does not have an effect on nodular and superficial BCC lesions, she continued, noting that it is at least comparable if not superior with clearance rates reported for methyl aminolevulinate–PDT (73%), imiquimod cream (83%), and fluorouracil cream (80%). “Maybe we’re not specifically targeting the vasculature [with this approach], but we did some optical coherence tomography imaging and saw that the blood vessels in the tumor were coagulated while the vasculature in the surrounding normal skin were spared,” said Dr. Ortiz, who is also vice president of the American Society for Laser Medicine and Surgery.
In a more recent analysis, she and her colleagues retrospectively analyzed long-term outcomes in 11 patients with BCC who had 16 lesions treated with the 1,064-nm Nd:YAG laser. At a mean of 9 months, 100% of lesions remained clear as determined by clinical observation.
In a subsequent, as yet unpublished study, she and her collaborators followed 34 patients with BCC one year following laser treatment. “Of these, 33 had no recurrence at 1-year follow-up,” Dr. Ortiz said, noting that the one patient with a recurrence was on a biologic agent for Crohn’s disease.
One key advantage of using the long-pulsed 1,064-nm Nd:YAG laser for nonaggressive BCC is the potential for one treatment visit. “They don’t have to come back for suture removal,” she said. “It’s a quick procedure, takes only about 5 minutes. There’s no limitation on activity and there’s minimal wound care, light ointment, and a band-aid; that’s it.”
In addition, she said, there is a lower risk of complications, infections, and bleeding, and there is minimal scarring. It is “also an alternative for treating patients with multiple tumors or those who are poor surgical candidates, such as the elderly and those with Gorlin syndrome.”
Dr. Ortiz avoids treating aggressive subtypes “because we don’t know what margin to treat,” she added. “Avoid the face. I do make some exceptions for patients if they’re elderly or if they’ve had multiple tumors. Monitor for recurrence like you would using any other modality.”
She uses lidocaine without epinephrine to avoid vasoconstriction and treats with the 1,064-nm Nd:YAG laser as follows: a 5-mm spot size, a fluence of 140 J/cm2, and a pulse duration of 8 ms, with no cooling, which are the settings for the Excel V Laser System, she noted. “If you’re using a different Nd:YAG laser, your pulse duration may vary. I do let the device cool in between pulses to avoid bulk heating.”
The immediate endpoint to strive for is slight greying and slight contraction, and the procedure is covered by insurance, billed as malignant destruction/EDC (CPT codes 17260-17266 trunk and 17280-17283 face). “I do biopsy prior to treatment,” she said. “I like the biopsy to be healed when I’m using the laser, so I’ll treat them about a month later.”
As for future directions, Dr. Ortiz and colleagues plan to evaluate the use of gold nanoparticles to more selectively target BCC during treatment with the 1,064-nm Nd:YAG laser. For now, she sees no downside of the procedure for proper candidates. “I do think that patients really like it,” she said. “It’s effective and easy.”
Dr. Ortiz disclosed having financial relationships with several pharmaceutical and device companies. She is also cochair of the MOAS.
SAN DIEGO – After Arisa E. Ortiz, MD, and colleagues published results of a multicenter study reporting that one treatment with the long-pulsed 1,064-nm Nd:YAG laser cleared nonaggressive basal cell carcinoma (BCC) on the trunk and extremities in 90% of patients, she heard from colleagues who were skeptical of the approach.
Maybe it’s just the biopsy alone that’s clearing these tumors, some told her. Others postulated that since the energy was delivered with a 5- to 6-mm spot size at a fluence of 125-140 J/cm2 and a 7- to 10-ms pulse duration, bulk heating likely disrupted the tumors. However, treatments were generally well tolerated, required no anesthesia, and caused no significant adverse events.
“It’s almost scarless,” Dr. Ortiz, director of laser and cosmetic dermatology at the University of California, San Diego, said at the annual Masters of Aesthetics Symposium. “Sometimes the treatment does leave a mark, but I think the scars are always acceptable. We do have good histologic evidence that we can penetrate 2.15 mm, which is a lot deeper than what the pulsed-dye laser or other superficial wavelengths are able to penetrate.”
Data is well powered to reject the null hypothesis that laser treatment does not have an effect on nodular and superficial BCC lesions, she continued, noting that it is at least comparable if not superior with clearance rates reported for methyl aminolevulinate–PDT (73%), imiquimod cream (83%), and fluorouracil cream (80%). “Maybe we’re not specifically targeting the vasculature [with this approach], but we did some optical coherence tomography imaging and saw that the blood vessels in the tumor were coagulated while the vasculature in the surrounding normal skin were spared,” said Dr. Ortiz, who is also vice president of the American Society for Laser Medicine and Surgery.
In a more recent analysis, she and her colleagues retrospectively analyzed long-term outcomes in 11 patients with BCC who had 16 lesions treated with the 1,064-nm Nd:YAG laser. At a mean of 9 months, 100% of lesions remained clear as determined by clinical observation.
In a subsequent, as yet unpublished study, she and her collaborators followed 34 patients with BCC one year following laser treatment. “Of these, 33 had no recurrence at 1-year follow-up,” Dr. Ortiz said, noting that the one patient with a recurrence was on a biologic agent for Crohn’s disease.
One key advantage of using the long-pulsed 1,064-nm Nd:YAG laser for nonaggressive BCC is the potential for one treatment visit. “They don’t have to come back for suture removal,” she said. “It’s a quick procedure, takes only about 5 minutes. There’s no limitation on activity and there’s minimal wound care, light ointment, and a band-aid; that’s it.”
In addition, she said, there is a lower risk of complications, infections, and bleeding, and there is minimal scarring. It is “also an alternative for treating patients with multiple tumors or those who are poor surgical candidates, such as the elderly and those with Gorlin syndrome.”
Dr. Ortiz avoids treating aggressive subtypes “because we don’t know what margin to treat,” she added. “Avoid the face. I do make some exceptions for patients if they’re elderly or if they’ve had multiple tumors. Monitor for recurrence like you would using any other modality.”
She uses lidocaine without epinephrine to avoid vasoconstriction and treats with the 1,064-nm Nd:YAG laser as follows: a 5-mm spot size, a fluence of 140 J/cm2, and a pulse duration of 8 ms, with no cooling, which are the settings for the Excel V Laser System, she noted. “If you’re using a different Nd:YAG laser, your pulse duration may vary. I do let the device cool in between pulses to avoid bulk heating.”
The immediate endpoint to strive for is slight greying and slight contraction, and the procedure is covered by insurance, billed as malignant destruction/EDC (CPT codes 17260-17266 trunk and 17280-17283 face). “I do biopsy prior to treatment,” she said. “I like the biopsy to be healed when I’m using the laser, so I’ll treat them about a month later.”
As for future directions, Dr. Ortiz and colleagues plan to evaluate the use of gold nanoparticles to more selectively target BCC during treatment with the 1,064-nm Nd:YAG laser. For now, she sees no downside of the procedure for proper candidates. “I do think that patients really like it,” she said. “It’s effective and easy.”
Dr. Ortiz disclosed having financial relationships with several pharmaceutical and device companies. She is also cochair of the MOAS.
SAN DIEGO – After Arisa E. Ortiz, MD, and colleagues published results of a multicenter study reporting that one treatment with the long-pulsed 1,064-nm Nd:YAG laser cleared nonaggressive basal cell carcinoma (BCC) on the trunk and extremities in 90% of patients, she heard from colleagues who were skeptical of the approach.
Maybe it’s just the biopsy alone that’s clearing these tumors, some told her. Others postulated that since the energy was delivered with a 5- to 6-mm spot size at a fluence of 125-140 J/cm2 and a 7- to 10-ms pulse duration, bulk heating likely disrupted the tumors. However, treatments were generally well tolerated, required no anesthesia, and caused no significant adverse events.
“It’s almost scarless,” Dr. Ortiz, director of laser and cosmetic dermatology at the University of California, San Diego, said at the annual Masters of Aesthetics Symposium. “Sometimes the treatment does leave a mark, but I think the scars are always acceptable. We do have good histologic evidence that we can penetrate 2.15 mm, which is a lot deeper than what the pulsed-dye laser or other superficial wavelengths are able to penetrate.”
Data is well powered to reject the null hypothesis that laser treatment does not have an effect on nodular and superficial BCC lesions, she continued, noting that it is at least comparable if not superior with clearance rates reported for methyl aminolevulinate–PDT (73%), imiquimod cream (83%), and fluorouracil cream (80%). “Maybe we’re not specifically targeting the vasculature [with this approach], but we did some optical coherence tomography imaging and saw that the blood vessels in the tumor were coagulated while the vasculature in the surrounding normal skin were spared,” said Dr. Ortiz, who is also vice president of the American Society for Laser Medicine and Surgery.
In a more recent analysis, she and her colleagues retrospectively analyzed long-term outcomes in 11 patients with BCC who had 16 lesions treated with the 1,064-nm Nd:YAG laser. At a mean of 9 months, 100% of lesions remained clear as determined by clinical observation.
In a subsequent, as yet unpublished study, she and her collaborators followed 34 patients with BCC one year following laser treatment. “Of these, 33 had no recurrence at 1-year follow-up,” Dr. Ortiz said, noting that the one patient with a recurrence was on a biologic agent for Crohn’s disease.
One key advantage of using the long-pulsed 1,064-nm Nd:YAG laser for nonaggressive BCC is the potential for one treatment visit. “They don’t have to come back for suture removal,” she said. “It’s a quick procedure, takes only about 5 minutes. There’s no limitation on activity and there’s minimal wound care, light ointment, and a band-aid; that’s it.”
In addition, she said, there is a lower risk of complications, infections, and bleeding, and there is minimal scarring. It is “also an alternative for treating patients with multiple tumors or those who are poor surgical candidates, such as the elderly and those with Gorlin syndrome.”
Dr. Ortiz avoids treating aggressive subtypes “because we don’t know what margin to treat,” she added. “Avoid the face. I do make some exceptions for patients if they’re elderly or if they’ve had multiple tumors. Monitor for recurrence like you would using any other modality.”
She uses lidocaine without epinephrine to avoid vasoconstriction and treats with the 1,064-nm Nd:YAG laser as follows: a 5-mm spot size, a fluence of 140 J/cm2, and a pulse duration of 8 ms, with no cooling, which are the settings for the Excel V Laser System, she noted. “If you’re using a different Nd:YAG laser, your pulse duration may vary. I do let the device cool in between pulses to avoid bulk heating.”
The immediate endpoint to strive for is slight greying and slight contraction, and the procedure is covered by insurance, billed as malignant destruction/EDC (CPT codes 17260-17266 trunk and 17280-17283 face). “I do biopsy prior to treatment,” she said. “I like the biopsy to be healed when I’m using the laser, so I’ll treat them about a month later.”
As for future directions, Dr. Ortiz and colleagues plan to evaluate the use of gold nanoparticles to more selectively target BCC during treatment with the 1,064-nm Nd:YAG laser. For now, she sees no downside of the procedure for proper candidates. “I do think that patients really like it,” she said. “It’s effective and easy.”
Dr. Ortiz disclosed having financial relationships with several pharmaceutical and device companies. She is also cochair of the MOAS.
AT MOAS 2022
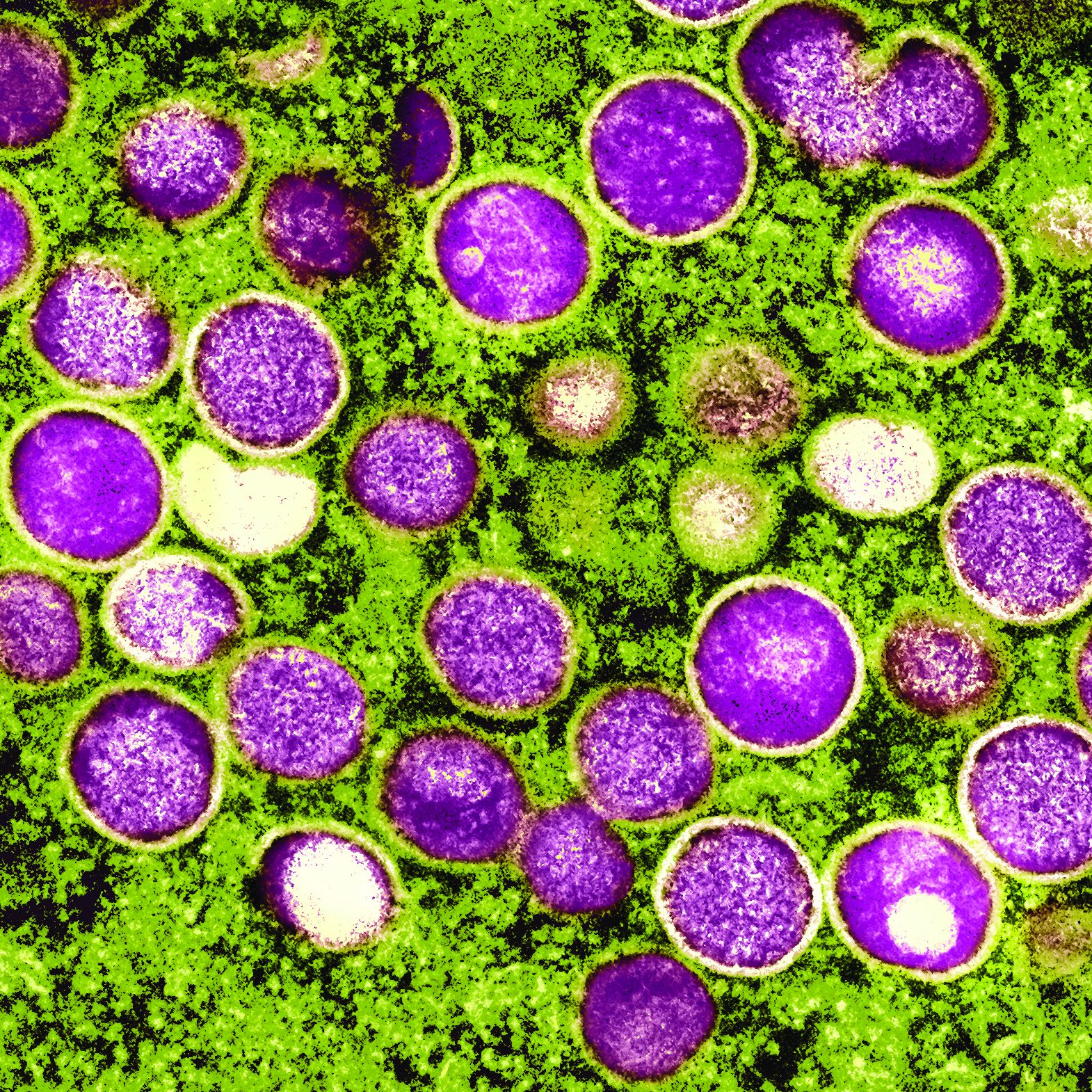
Mpox: Dermatology registry data pinpoints unique signs
that frequently appeared before systemic illness and a much lower overall numbers of lesions.
“Just these two findings alone show how important it is to remain clinically vigilant as dermatologists,” Esther Freeman, MD, PhD, director of global health dermatology at Massachusetts General Hospital, Boston, said in an interview. She is the corresponding author of the study, which analyzed 101 mpox cases from 13 countries and was published online on in the Journal of the American Academy of Dermatology.
“Mpox appeared to manifest differently than in previous outbreaks with morphologic and clinical evolutions much different than previously reported in endemic and prior outbreaks,” added Dr. Freeman. “Dermatologists should continue to keep mpox on the differential as it continues to circulate at low levels in the population and is a mimicker of many other common skin diseases.”
According to the Centers for Disease Control and Prevention, as of Jan. 20, 2023, there have been 30,061 cases of mpox in the United States during the outbreak that began in 2022; 23 people died. Worldwide, the number of cases neared 85,000.
Most of the affected cases were among gay, bisexual, and other men who have sex with men. A vaccination effort began last summer, and the number of cases soon plummeted. The national daily case count in January has been in the single digits.
For the new report, dermatologists tracked cases via the American Academy of Dermatology/International League of Dermatologic Societies (AAD/ILDS) Dermatology COVID-19, Monkeypox (mpox), and Emerging Infections Registry. The new report includes data about cases entered from Aug. 4 to Nov. 13. Of these cases, 97% were male, median age was 35 years, 62% were White, 20% were Hispanic, and 11% were Black.
Just over half (54%) of patients reported skin lesions as the first sign of disease, while others had signs such as fever (16%) and malaise (9%). “This is a sharp contrast to endemic or prior outbreaks in which a ‘flu-like’ prodrome preceded lesions,” Dr. Freeman said. “Dermatologists should be aware that patients may come in with mpox skin lesions as their only initial symptoms.”
In contrast to past outbreaks where patients may have had dozens or hundreds of lesions, 20% had only 1 lesion, while 52% had 2-5 lesions, and 20% had 6-20 lesions. “There may be only a few lesions, so index of suspicion needs to be high,” Dr. Freeman said.
According to the study, “the most common skin lesion morphologies and secondary characteristics reported included papules, vesicles/blisters, pustules, erosions/ulcers and crust/scabs.” Dr. Freeman cautioned that “lesions may not go through the ‘typical’ progression from papule to pustule. The initial lesion could even be an ulceration or a crust. For dermatologists, this means you need to have a high index of suspicion, especially if you see a new onset lesion in the groin or perianal area, though they can also start elsewhere.”
She added that “the lesion you see on exam could be a classic pustule/pseudopustule, but it might not be – it could be a small perianal erosion or ulceration. If you have any concern it could be mpox, it’s a good idea to test by PCR.”
Morbilliform rash, scarring reported
The study also highlighted 10 cases of morbilliform rash. “A morbilliform exanthem is pretty nonspecific, and usually cases of mpox have more specific features,” dermatologist and study coauthor Misha Rosenbach, MD, of the University of Pennsylvania, Philadelphia, said in an interview.
“Given the current low rates of mpox, I do not think most dermatologists need to worry about mpox when evaluating morbilliform exanthems. However, in high-risk patients or patients with other morphologies, it is worth noting that there’s a chance that this may be related.”
Emory University dermatologist Howa Yeung, MD, MSc, who wasn’t involved with the study, said in an interview that morbilliform rashes in the mouth/tongue area, mostly on days 1-5, should be considered a possible sign of mpox. “While I didn’t typically think of monkeypox virus as a cause of viral exanthems, I will now add it to my differential diagnoses.”
In the report, 13% of patients had scarring, “an outcome underemphasized in the current literature” that could have long-term emotional and mental effects, the authors noted. “Some patients, particularly immunosuppressed patients, have had very large and/or ulceronecrotic lesions,” Dr. Rosenbach said. “Their scarring can be quite significant. There is, to date, very little guidance for clinicians or patients on how to mitigate this risk and, if scarring is developing, how best to manage it.”
As for lessons from the findings, Dr. Yeung said, “dermatologists need to be aware that patients with mpox can have multiple morphologies at the same time and lesions can skip stages.” And, he pointed out, it’s clear that wound care is important to prevent scarring.
The AAD has a resource page on skin care in patients with mpox that includes information about preventing scarring. Examples of mpox rashes are available on the CDC website.
The study was supported by a grant from the International League of Dermatologic Societies and in-kind support from the American Academy of Dermatology. Dr. Freeman is a coauthor for UpToDate. Dr. Freeman and Dr. Rosenbach are members of the AAD Ad Hoc Task Force to Create Monkeypox Content. Study authors reported no other disclosures, and Dr. Yeung has no disclosures.
that frequently appeared before systemic illness and a much lower overall numbers of lesions.
“Just these two findings alone show how important it is to remain clinically vigilant as dermatologists,” Esther Freeman, MD, PhD, director of global health dermatology at Massachusetts General Hospital, Boston, said in an interview. She is the corresponding author of the study, which analyzed 101 mpox cases from 13 countries and was published online on in the Journal of the American Academy of Dermatology.
“Mpox appeared to manifest differently than in previous outbreaks with morphologic and clinical evolutions much different than previously reported in endemic and prior outbreaks,” added Dr. Freeman. “Dermatologists should continue to keep mpox on the differential as it continues to circulate at low levels in the population and is a mimicker of many other common skin diseases.”
According to the Centers for Disease Control and Prevention, as of Jan. 20, 2023, there have been 30,061 cases of mpox in the United States during the outbreak that began in 2022; 23 people died. Worldwide, the number of cases neared 85,000.
Most of the affected cases were among gay, bisexual, and other men who have sex with men. A vaccination effort began last summer, and the number of cases soon plummeted. The national daily case count in January has been in the single digits.
For the new report, dermatologists tracked cases via the American Academy of Dermatology/International League of Dermatologic Societies (AAD/ILDS) Dermatology COVID-19, Monkeypox (mpox), and Emerging Infections Registry. The new report includes data about cases entered from Aug. 4 to Nov. 13. Of these cases, 97% were male, median age was 35 years, 62% were White, 20% were Hispanic, and 11% were Black.
Just over half (54%) of patients reported skin lesions as the first sign of disease, while others had signs such as fever (16%) and malaise (9%). “This is a sharp contrast to endemic or prior outbreaks in which a ‘flu-like’ prodrome preceded lesions,” Dr. Freeman said. “Dermatologists should be aware that patients may come in with mpox skin lesions as their only initial symptoms.”
In contrast to past outbreaks where patients may have had dozens or hundreds of lesions, 20% had only 1 lesion, while 52% had 2-5 lesions, and 20% had 6-20 lesions. “There may be only a few lesions, so index of suspicion needs to be high,” Dr. Freeman said.
According to the study, “the most common skin lesion morphologies and secondary characteristics reported included papules, vesicles/blisters, pustules, erosions/ulcers and crust/scabs.” Dr. Freeman cautioned that “lesions may not go through the ‘typical’ progression from papule to pustule. The initial lesion could even be an ulceration or a crust. For dermatologists, this means you need to have a high index of suspicion, especially if you see a new onset lesion in the groin or perianal area, though they can also start elsewhere.”
She added that “the lesion you see on exam could be a classic pustule/pseudopustule, but it might not be – it could be a small perianal erosion or ulceration. If you have any concern it could be mpox, it’s a good idea to test by PCR.”
Morbilliform rash, scarring reported
The study also highlighted 10 cases of morbilliform rash. “A morbilliform exanthem is pretty nonspecific, and usually cases of mpox have more specific features,” dermatologist and study coauthor Misha Rosenbach, MD, of the University of Pennsylvania, Philadelphia, said in an interview.
“Given the current low rates of mpox, I do not think most dermatologists need to worry about mpox when evaluating morbilliform exanthems. However, in high-risk patients or patients with other morphologies, it is worth noting that there’s a chance that this may be related.”
Emory University dermatologist Howa Yeung, MD, MSc, who wasn’t involved with the study, said in an interview that morbilliform rashes in the mouth/tongue area, mostly on days 1-5, should be considered a possible sign of mpox. “While I didn’t typically think of monkeypox virus as a cause of viral exanthems, I will now add it to my differential diagnoses.”
In the report, 13% of patients had scarring, “an outcome underemphasized in the current literature” that could have long-term emotional and mental effects, the authors noted. “Some patients, particularly immunosuppressed patients, have had very large and/or ulceronecrotic lesions,” Dr. Rosenbach said. “Their scarring can be quite significant. There is, to date, very little guidance for clinicians or patients on how to mitigate this risk and, if scarring is developing, how best to manage it.”
As for lessons from the findings, Dr. Yeung said, “dermatologists need to be aware that patients with mpox can have multiple morphologies at the same time and lesions can skip stages.” And, he pointed out, it’s clear that wound care is important to prevent scarring.
The AAD has a resource page on skin care in patients with mpox that includes information about preventing scarring. Examples of mpox rashes are available on the CDC website.
The study was supported by a grant from the International League of Dermatologic Societies and in-kind support from the American Academy of Dermatology. Dr. Freeman is a coauthor for UpToDate. Dr. Freeman and Dr. Rosenbach are members of the AAD Ad Hoc Task Force to Create Monkeypox Content. Study authors reported no other disclosures, and Dr. Yeung has no disclosures.
that frequently appeared before systemic illness and a much lower overall numbers of lesions.
“Just these two findings alone show how important it is to remain clinically vigilant as dermatologists,” Esther Freeman, MD, PhD, director of global health dermatology at Massachusetts General Hospital, Boston, said in an interview. She is the corresponding author of the study, which analyzed 101 mpox cases from 13 countries and was published online on in the Journal of the American Academy of Dermatology.
“Mpox appeared to manifest differently than in previous outbreaks with morphologic and clinical evolutions much different than previously reported in endemic and prior outbreaks,” added Dr. Freeman. “Dermatologists should continue to keep mpox on the differential as it continues to circulate at low levels in the population and is a mimicker of many other common skin diseases.”
According to the Centers for Disease Control and Prevention, as of Jan. 20, 2023, there have been 30,061 cases of mpox in the United States during the outbreak that began in 2022; 23 people died. Worldwide, the number of cases neared 85,000.
Most of the affected cases were among gay, bisexual, and other men who have sex with men. A vaccination effort began last summer, and the number of cases soon plummeted. The national daily case count in January has been in the single digits.
For the new report, dermatologists tracked cases via the American Academy of Dermatology/International League of Dermatologic Societies (AAD/ILDS) Dermatology COVID-19, Monkeypox (mpox), and Emerging Infections Registry. The new report includes data about cases entered from Aug. 4 to Nov. 13. Of these cases, 97% were male, median age was 35 years, 62% were White, 20% were Hispanic, and 11% were Black.
Just over half (54%) of patients reported skin lesions as the first sign of disease, while others had signs such as fever (16%) and malaise (9%). “This is a sharp contrast to endemic or prior outbreaks in which a ‘flu-like’ prodrome preceded lesions,” Dr. Freeman said. “Dermatologists should be aware that patients may come in with mpox skin lesions as their only initial symptoms.”
In contrast to past outbreaks where patients may have had dozens or hundreds of lesions, 20% had only 1 lesion, while 52% had 2-5 lesions, and 20% had 6-20 lesions. “There may be only a few lesions, so index of suspicion needs to be high,” Dr. Freeman said.
According to the study, “the most common skin lesion morphologies and secondary characteristics reported included papules, vesicles/blisters, pustules, erosions/ulcers and crust/scabs.” Dr. Freeman cautioned that “lesions may not go through the ‘typical’ progression from papule to pustule. The initial lesion could even be an ulceration or a crust. For dermatologists, this means you need to have a high index of suspicion, especially if you see a new onset lesion in the groin or perianal area, though they can also start elsewhere.”
She added that “the lesion you see on exam could be a classic pustule/pseudopustule, but it might not be – it could be a small perianal erosion or ulceration. If you have any concern it could be mpox, it’s a good idea to test by PCR.”
Morbilliform rash, scarring reported
The study also highlighted 10 cases of morbilliform rash. “A morbilliform exanthem is pretty nonspecific, and usually cases of mpox have more specific features,” dermatologist and study coauthor Misha Rosenbach, MD, of the University of Pennsylvania, Philadelphia, said in an interview.
“Given the current low rates of mpox, I do not think most dermatologists need to worry about mpox when evaluating morbilliform exanthems. However, in high-risk patients or patients with other morphologies, it is worth noting that there’s a chance that this may be related.”
Emory University dermatologist Howa Yeung, MD, MSc, who wasn’t involved with the study, said in an interview that morbilliform rashes in the mouth/tongue area, mostly on days 1-5, should be considered a possible sign of mpox. “While I didn’t typically think of monkeypox virus as a cause of viral exanthems, I will now add it to my differential diagnoses.”
In the report, 13% of patients had scarring, “an outcome underemphasized in the current literature” that could have long-term emotional and mental effects, the authors noted. “Some patients, particularly immunosuppressed patients, have had very large and/or ulceronecrotic lesions,” Dr. Rosenbach said. “Their scarring can be quite significant. There is, to date, very little guidance for clinicians or patients on how to mitigate this risk and, if scarring is developing, how best to manage it.”
As for lessons from the findings, Dr. Yeung said, “dermatologists need to be aware that patients with mpox can have multiple morphologies at the same time and lesions can skip stages.” And, he pointed out, it’s clear that wound care is important to prevent scarring.
The AAD has a resource page on skin care in patients with mpox that includes information about preventing scarring. Examples of mpox rashes are available on the CDC website.
The study was supported by a grant from the International League of Dermatologic Societies and in-kind support from the American Academy of Dermatology. Dr. Freeman is a coauthor for UpToDate. Dr. Freeman and Dr. Rosenbach are members of the AAD Ad Hoc Task Force to Create Monkeypox Content. Study authors reported no other disclosures, and Dr. Yeung has no disclosures.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
Age-related atopic dermatitis phenotypes evaluated in study
, while older adults tend to present with less flexural eczema and the fewest associated signs.
Those are key findings from a study conducted at a single academic medical center, which aimed to identify the age-related clinical phenotypes of AD.
“Previous studies have found differences in the clinical characteristics of AD depending on age of AD onset, ethnic background, and AD severity,” senior author Jonathan I. Silverberg, MD, PhD, MPH, director of clinical research in the department of dermatology at George Washington University, Washington, and his coauthor wrote in the study, which was published online in JAAD International. “However, none have prospectively compared the clinical characteristics and associated signs by age group. Improved understanding of the clinical phenotypes of AD may help guide choice of treatment and improve health outcomes,” they added.
Along with coauthor Sheena Chatrath, a dermatology research fellow in the department, Dr. Silverberg prospectively reviewed self-reported questionnaires that were completed by 380 patients prior to their visit at GWU’s eczema clinic between 2013 and 2019. Questions included age of AD onset, sociodemographics, Visual Analog Scale (VAS) itch and sleep for Scoring AD, and Numeric Rating Scale (NRS) for skin pain and itch. The researchers used the Eczema Area Severity Index to assess AD severity and a dermatologist conducted full body skin exams, noting the distribution of AD involvement as well as associated signs.
Of the 380 patients, 6.1% were younger than aged 18 years, 46.3% were young adults aged 18-39 years, and 47.6% were older adults 40 years of age and older.
Compared with pediatric patients, both young and older adults were less likely to experience AD on the ankles (adjusted odds ratio [aOR], 0.41 and 0.43, respectively), moderate to severe AD lesions on flexures (aOR, 0.47 and 0.30), pityriasis alba (aOR, 0.24 and 0.07), oozing lesions (aOR, 0.44 and 0.35), and moderate to severe excoriations (aOR, 0.49 and 0.44).
In children, severe itch was more common, reported in 47.1%, compared with 43.4% of the young adults and 38.6% of the older adults, and itch was less severe among the young and older adults. “Interestingly, despite increased itch in pediatric patients, we found no difference in the severity of skin pain across all age groups,” the researchers wrote. “Moreover, pediatric patients reported skin pain less often than adult patients. This may be due to age-related differences of pain perception.”
In other findings, compared with pediatric patients, young adults were more likely to experience AD around the eyes (aOR, 2.92), while older adults were less likely to experience AD on elbows (aOR, 0.34), nipples (aOR, 0.40), knees (aOR, 0.27), and less likely to have keratosis pilaris (aOR, 0.38), and lichenification (aOR, 0.47).
Dr. Silverberg and Ms. Chatrath used latent class analysis to identify four classes for distribution of AD lesions. In this model, class 1 had low probabilities of AD involvement at all sites examined and class 2 had low probabilities of scalp, face, and foot involvement, and intermediate probability of all other AD sites. Class 3 had low probabilities of hand and foot involvement, high probability of facial erythema, and intermediate probability of all other AD signs, while class 4 had intermediate probability of postauricular and foot involvement, and high probability of all other AD sites examined.
“Pediatric patients were most commonly in class 4 (33.3%), followed by class 1 and 2 (26.7%), and least commonly in class 3 (13.3%),” the authors wrote. “In young adults, class 4 and 1 were most common (32.4% and 29.4%), followed by class 2 (27.9%), and least commonly class 3 (10.3%). In older adults, class 1 was most common (40.3%), followed by class 4 (23.6%), and least commonly classes 2 and 3 (18.1%).”
The researchers also used latent class analysis to identify four classes for the signs and symptoms of AD. In this model, class 1 had zero-low probability of all AD signs and class 2 had low probability of all AD signs. Class 3 had high probability of oozing lesions and low probability of all other signs, while class 4 had high probability of xerosis, intermediate probability of ichthyosis and palmar hyperlinearity, and low probability of all other AD signs.
In all three groups, the most common class was class 1 (85.6% of older adults, 81.8% of younger adults, and 82.6% of pediatric patients). Among the pediatric patients, they wrote, “class 3 was the second most common (8.7%), followed by class 2 and 4 (4.4%).” Among the young adults, 9.7% were in class 2, 5.7% were in class 4, and 2.8% were in class 3; and among the older adults, 8.3% were in class 4, 4.4% were in class 2, and 1.67% were in class 3.
Zelma Chiesa Fuxench, MD, of the department of dermatology at the University of Pennsylvania, Philadelphia, who was asked to comment on the study, said that while AD is traditionally largely thought of as a disease of children primarily involving the flexural areas, “this study provides additional evidence to support that AD is more than just a disease of childhood with a fixed clinical presentation, but is a heterogeneous disease whose clinical presentation varies across different population groups.”
While the study provides insight into the clinical differences that may be observed across AD groups, “care must be taken when interpreting these results as the study was done in a single center with observations collected during one single visit,” she added. “AD is not a ‘static’ disease; its presentation can stay the same in one patient but can vary even in another patient throughout their lifetime. Therefore, studies of a more prospective nature that evaluate the change in clinical presentation using multiple measures throughout time in these individuals would be a step forward to better understand if these phenotypic differences truly exist and, as such, what implications could they have for treatment selection.”
This study was supported by grants from the Agency for Healthcare Research and Quality and the Dermatology Foundation. The researchers reported having no disclosures. Dr. Chiesa Fuxench disclosed serving as a consultant for the Asthma and Allergy Foundation of America, National Eczema Association, Pfizer, AbbVie, and Incyte, for which she has received honoraria for AD-related work. She is the recipient of research grants through Regeneron, Sanofi, Tioga, Vanda, Menlo Therapeutics, Leo Pharma, and Eli Lilly for work related to AD as well as honoraria for continuing medical education work related to AD sponsored through educational grants from Regeneron/Sanofi and Pfizer.
, while older adults tend to present with less flexural eczema and the fewest associated signs.
Those are key findings from a study conducted at a single academic medical center, which aimed to identify the age-related clinical phenotypes of AD.
“Previous studies have found differences in the clinical characteristics of AD depending on age of AD onset, ethnic background, and AD severity,” senior author Jonathan I. Silverberg, MD, PhD, MPH, director of clinical research in the department of dermatology at George Washington University, Washington, and his coauthor wrote in the study, which was published online in JAAD International. “However, none have prospectively compared the clinical characteristics and associated signs by age group. Improved understanding of the clinical phenotypes of AD may help guide choice of treatment and improve health outcomes,” they added.
Along with coauthor Sheena Chatrath, a dermatology research fellow in the department, Dr. Silverberg prospectively reviewed self-reported questionnaires that were completed by 380 patients prior to their visit at GWU’s eczema clinic between 2013 and 2019. Questions included age of AD onset, sociodemographics, Visual Analog Scale (VAS) itch and sleep for Scoring AD, and Numeric Rating Scale (NRS) for skin pain and itch. The researchers used the Eczema Area Severity Index to assess AD severity and a dermatologist conducted full body skin exams, noting the distribution of AD involvement as well as associated signs.
Of the 380 patients, 6.1% were younger than aged 18 years, 46.3% were young adults aged 18-39 years, and 47.6% were older adults 40 years of age and older.
Compared with pediatric patients, both young and older adults were less likely to experience AD on the ankles (adjusted odds ratio [aOR], 0.41 and 0.43, respectively), moderate to severe AD lesions on flexures (aOR, 0.47 and 0.30), pityriasis alba (aOR, 0.24 and 0.07), oozing lesions (aOR, 0.44 and 0.35), and moderate to severe excoriations (aOR, 0.49 and 0.44).
In children, severe itch was more common, reported in 47.1%, compared with 43.4% of the young adults and 38.6% of the older adults, and itch was less severe among the young and older adults. “Interestingly, despite increased itch in pediatric patients, we found no difference in the severity of skin pain across all age groups,” the researchers wrote. “Moreover, pediatric patients reported skin pain less often than adult patients. This may be due to age-related differences of pain perception.”
In other findings, compared with pediatric patients, young adults were more likely to experience AD around the eyes (aOR, 2.92), while older adults were less likely to experience AD on elbows (aOR, 0.34), nipples (aOR, 0.40), knees (aOR, 0.27), and less likely to have keratosis pilaris (aOR, 0.38), and lichenification (aOR, 0.47).
Dr. Silverberg and Ms. Chatrath used latent class analysis to identify four classes for distribution of AD lesions. In this model, class 1 had low probabilities of AD involvement at all sites examined and class 2 had low probabilities of scalp, face, and foot involvement, and intermediate probability of all other AD sites. Class 3 had low probabilities of hand and foot involvement, high probability of facial erythema, and intermediate probability of all other AD signs, while class 4 had intermediate probability of postauricular and foot involvement, and high probability of all other AD sites examined.
“Pediatric patients were most commonly in class 4 (33.3%), followed by class 1 and 2 (26.7%), and least commonly in class 3 (13.3%),” the authors wrote. “In young adults, class 4 and 1 were most common (32.4% and 29.4%), followed by class 2 (27.9%), and least commonly class 3 (10.3%). In older adults, class 1 was most common (40.3%), followed by class 4 (23.6%), and least commonly classes 2 and 3 (18.1%).”
The researchers also used latent class analysis to identify four classes for the signs and symptoms of AD. In this model, class 1 had zero-low probability of all AD signs and class 2 had low probability of all AD signs. Class 3 had high probability of oozing lesions and low probability of all other signs, while class 4 had high probability of xerosis, intermediate probability of ichthyosis and palmar hyperlinearity, and low probability of all other AD signs.
In all three groups, the most common class was class 1 (85.6% of older adults, 81.8% of younger adults, and 82.6% of pediatric patients). Among the pediatric patients, they wrote, “class 3 was the second most common (8.7%), followed by class 2 and 4 (4.4%).” Among the young adults, 9.7% were in class 2, 5.7% were in class 4, and 2.8% were in class 3; and among the older adults, 8.3% were in class 4, 4.4% were in class 2, and 1.67% were in class 3.
Zelma Chiesa Fuxench, MD, of the department of dermatology at the University of Pennsylvania, Philadelphia, who was asked to comment on the study, said that while AD is traditionally largely thought of as a disease of children primarily involving the flexural areas, “this study provides additional evidence to support that AD is more than just a disease of childhood with a fixed clinical presentation, but is a heterogeneous disease whose clinical presentation varies across different population groups.”
While the study provides insight into the clinical differences that may be observed across AD groups, “care must be taken when interpreting these results as the study was done in a single center with observations collected during one single visit,” she added. “AD is not a ‘static’ disease; its presentation can stay the same in one patient but can vary even in another patient throughout their lifetime. Therefore, studies of a more prospective nature that evaluate the change in clinical presentation using multiple measures throughout time in these individuals would be a step forward to better understand if these phenotypic differences truly exist and, as such, what implications could they have for treatment selection.”
This study was supported by grants from the Agency for Healthcare Research and Quality and the Dermatology Foundation. The researchers reported having no disclosures. Dr. Chiesa Fuxench disclosed serving as a consultant for the Asthma and Allergy Foundation of America, National Eczema Association, Pfizer, AbbVie, and Incyte, for which she has received honoraria for AD-related work. She is the recipient of research grants through Regeneron, Sanofi, Tioga, Vanda, Menlo Therapeutics, Leo Pharma, and Eli Lilly for work related to AD as well as honoraria for continuing medical education work related to AD sponsored through educational grants from Regeneron/Sanofi and Pfizer.
, while older adults tend to present with less flexural eczema and the fewest associated signs.
Those are key findings from a study conducted at a single academic medical center, which aimed to identify the age-related clinical phenotypes of AD.
“Previous studies have found differences in the clinical characteristics of AD depending on age of AD onset, ethnic background, and AD severity,” senior author Jonathan I. Silverberg, MD, PhD, MPH, director of clinical research in the department of dermatology at George Washington University, Washington, and his coauthor wrote in the study, which was published online in JAAD International. “However, none have prospectively compared the clinical characteristics and associated signs by age group. Improved understanding of the clinical phenotypes of AD may help guide choice of treatment and improve health outcomes,” they added.
Along with coauthor Sheena Chatrath, a dermatology research fellow in the department, Dr. Silverberg prospectively reviewed self-reported questionnaires that were completed by 380 patients prior to their visit at GWU’s eczema clinic between 2013 and 2019. Questions included age of AD onset, sociodemographics, Visual Analog Scale (VAS) itch and sleep for Scoring AD, and Numeric Rating Scale (NRS) for skin pain and itch. The researchers used the Eczema Area Severity Index to assess AD severity and a dermatologist conducted full body skin exams, noting the distribution of AD involvement as well as associated signs.
Of the 380 patients, 6.1% were younger than aged 18 years, 46.3% were young adults aged 18-39 years, and 47.6% were older adults 40 years of age and older.
Compared with pediatric patients, both young and older adults were less likely to experience AD on the ankles (adjusted odds ratio [aOR], 0.41 and 0.43, respectively), moderate to severe AD lesions on flexures (aOR, 0.47 and 0.30), pityriasis alba (aOR, 0.24 and 0.07), oozing lesions (aOR, 0.44 and 0.35), and moderate to severe excoriations (aOR, 0.49 and 0.44).
In children, severe itch was more common, reported in 47.1%, compared with 43.4% of the young adults and 38.6% of the older adults, and itch was less severe among the young and older adults. “Interestingly, despite increased itch in pediatric patients, we found no difference in the severity of skin pain across all age groups,” the researchers wrote. “Moreover, pediatric patients reported skin pain less often than adult patients. This may be due to age-related differences of pain perception.”
In other findings, compared with pediatric patients, young adults were more likely to experience AD around the eyes (aOR, 2.92), while older adults were less likely to experience AD on elbows (aOR, 0.34), nipples (aOR, 0.40), knees (aOR, 0.27), and less likely to have keratosis pilaris (aOR, 0.38), and lichenification (aOR, 0.47).
Dr. Silverberg and Ms. Chatrath used latent class analysis to identify four classes for distribution of AD lesions. In this model, class 1 had low probabilities of AD involvement at all sites examined and class 2 had low probabilities of scalp, face, and foot involvement, and intermediate probability of all other AD sites. Class 3 had low probabilities of hand and foot involvement, high probability of facial erythema, and intermediate probability of all other AD signs, while class 4 had intermediate probability of postauricular and foot involvement, and high probability of all other AD sites examined.
“Pediatric patients were most commonly in class 4 (33.3%), followed by class 1 and 2 (26.7%), and least commonly in class 3 (13.3%),” the authors wrote. “In young adults, class 4 and 1 were most common (32.4% and 29.4%), followed by class 2 (27.9%), and least commonly class 3 (10.3%). In older adults, class 1 was most common (40.3%), followed by class 4 (23.6%), and least commonly classes 2 and 3 (18.1%).”
The researchers also used latent class analysis to identify four classes for the signs and symptoms of AD. In this model, class 1 had zero-low probability of all AD signs and class 2 had low probability of all AD signs. Class 3 had high probability of oozing lesions and low probability of all other signs, while class 4 had high probability of xerosis, intermediate probability of ichthyosis and palmar hyperlinearity, and low probability of all other AD signs.
In all three groups, the most common class was class 1 (85.6% of older adults, 81.8% of younger adults, and 82.6% of pediatric patients). Among the pediatric patients, they wrote, “class 3 was the second most common (8.7%), followed by class 2 and 4 (4.4%).” Among the young adults, 9.7% were in class 2, 5.7% were in class 4, and 2.8% were in class 3; and among the older adults, 8.3% were in class 4, 4.4% were in class 2, and 1.67% were in class 3.
Zelma Chiesa Fuxench, MD, of the department of dermatology at the University of Pennsylvania, Philadelphia, who was asked to comment on the study, said that while AD is traditionally largely thought of as a disease of children primarily involving the flexural areas, “this study provides additional evidence to support that AD is more than just a disease of childhood with a fixed clinical presentation, but is a heterogeneous disease whose clinical presentation varies across different population groups.”
While the study provides insight into the clinical differences that may be observed across AD groups, “care must be taken when interpreting these results as the study was done in a single center with observations collected during one single visit,” she added. “AD is not a ‘static’ disease; its presentation can stay the same in one patient but can vary even in another patient throughout their lifetime. Therefore, studies of a more prospective nature that evaluate the change in clinical presentation using multiple measures throughout time in these individuals would be a step forward to better understand if these phenotypic differences truly exist and, as such, what implications could they have for treatment selection.”
This study was supported by grants from the Agency for Healthcare Research and Quality and the Dermatology Foundation. The researchers reported having no disclosures. Dr. Chiesa Fuxench disclosed serving as a consultant for the Asthma and Allergy Foundation of America, National Eczema Association, Pfizer, AbbVie, and Incyte, for which she has received honoraria for AD-related work. She is the recipient of research grants through Regeneron, Sanofi, Tioga, Vanda, Menlo Therapeutics, Leo Pharma, and Eli Lilly for work related to AD as well as honoraria for continuing medical education work related to AD sponsored through educational grants from Regeneron/Sanofi and Pfizer.
FROM JAAD INTERNATIONAL
Best estimates made for hydroxychloroquine retinopathy risk
A new study likely makes the best estimate yet of the degree of retinopathy risk that patients who take the antimalarial drug hydroxychloroquine (HCQ) can expect, deriving mainly from the cumulative dose taken during the first 5 years of use, according to a study published in Annals of Internal Medicine.
HCQ works to decrease activity in a patient’s immune system, which is effective in many cases of systemic lupus erythematosus, one of the most common indications for the drug. However, an adverse outcome of treatment can be HCQ retinopathy, a progressive form of vision loss in patients taking HCQ over an extended period (mostly for longer than 5 years). The disease is often asymptomatic, although some patients do present a paracentral scotoma and a decrease in color vision. Patients may also notice flashing shapes in their vision and find that they have difficulty reading. Eventually, HCQ retinopathy can lead to loss of visual acuity, loss of peripheral vision, and loss of night vision.
Researchers from Kaiser Permanente Northern California and Harvard Medical School analyzed 3,325 persons who received HCQ for 5 or more years between 2004 and 2020. Their goal was to both characterize the long-term risk for incident HCQ retinopathy and examine the degree to which average HCQ dose within the first 5 years of treatment serves as a prediction of the risk.
The researchers then estimated the risk for developing retinopathy after 15 years, according to patients’ average dosing levels during the first 5 years of therapy. Overall, 81 participants developed HCQ retinopathy with overall cumulative incidences of 2.5% after 10 years and 8.6% after 15 years; the risk was greater for those given a higher dose during the first 5 years of treatment.
The mechanism of how HCQ toxicity may occur is still not completely known. There is evidence that toxicity happens because HCQ binds to melanin in both the retinal pigment epithelium and uvea in high concentrations. HCQ can interfere with lysosomal function, leading to oxidation and accumulation of lysosomes, which can cause dysfunction of the retinal pigment epithelium.
Progressive retinopathy can continue even after the drug is stopped. “It’s thought to be a very mild but important risk,” said Nilanjana Bose, MD, MBA, a rheumatologist with Memorial Hermann Health System in Houston. “Patients taking HCQ must be screened for retinal issues, most certainly elderly patients and patients with any kind of comorbidities.”
A 2021 joint position statement from the American College of Rheumatology, American Academy of Dermatology, the Rheumatologic Dermatology Society, and the American Academy of Ophthalmology recommends a baseline eye exam within a few months after starting therapy, then additional screening at 5 years on HCQ and annually thereafter.
“Early detection of retinopathy is important in overall visual prognosis, because toxicity can continue even after discontinuation of the medication,” said Rukhsana G. Mirza, MD, professor of ophthalmology and medical education at Northwestern University in Chicago.
“Examination alone is not sufficient to evaluate early changes, and specialized testing must be done. These include color photos, visual field tests, optical coherence tomography, fundus autofluorescence and in some cases, multifocal electroretinogram. Also, the AAO [American Academy of Ophthalmology] has specific recommendations related to Asian patients as they may have a different pattern of retinopathy that must also be considered.”
More accurate risk measurements
This news organization asked study coauthor April Jorge, MD, assistant professor of medicine in the division of rheumatology, allergy, and immunology at Massachusetts General Hospital and Harvard Medical School, Boston, to discuss the study, how it correlates to past research, and what it adds that’s new and useful to rheumatologists and ophthalmologists:
Question: Your research found that a higher dose of HCQ in the first 5 years of treatment led to a greater risk of retinopathy. Is there any indication that a lower dose given more frequently, either within that 5-year period or longer, would pose a similar risk?
Answer: In our study, we assessed the HCQ dose in the first 5 years of use but followed patients who continued the medication longer than 5 years, through up to 15 years of use. Therefore, we compared the risk of HCQ retinopathy associated with different HCQ dosages but for the same duration of use. We found that for any dose of HCQ, the risk of retinopathy increases the longer the medication is used. However, patients who used a higher dose of HCQ had a higher risk of developing retinopathy over time.
Although current guidelines recommend avoiding any HCQ dose over 5 mg/kg per day to reduce the risk of retinopathy, we found a higher risk of retinopathy associated with dosing over 6 mg/kg per day than between 5 and 6 mg/kg per day and the lowest risk with dosing under 5 mg/kg per day.
Q: How does your study align with and/or expand upon previous research regarding HCQ risk?
A: An important prior study of hydroxychloroquine retinopathy was the 2014 study by Ronald B. Melles, MD, and Michael F. Marmor, MD, published in JAMA Ophthalmology. Prior to our present study, that was the largest study to use the modern screening method (optical coherence tomography) to detect HCQ retinopathy. That screening tool is more sensitive than older methods, so it can detect early/mild cases of retinopathy that are typically asymptomatic. Compared to older studies, that 2014 study found a much higher risk of HCQ retinopathy than was previously appreciated.
However, that 2014 study did have some key limitations that could affect the risk estimates, such as using prevalent cases. A key feature of our present study is that we took several important steps to generate more accurate risk estimates. This included using an incident user cohort and detecting incident retinopathy cases through serial review of optical coherence tomography (screening) studies.
To achieve a high degree of methodologic rigor in correctly identifying retinopathy outcomes, we had expert ophthalmologists perform masked adjudication of all screening studies, and we assessed the intra-rater reliability of these study interpretations. Therefore, our study adds to the literature more accurate estimates of retinopathy risk. We found a lower cumulative incidence of retinopathy than was identified in the 2014 study, but the risk is still noteworthy.
Also unique to our study, we graded the severity of HCQ retinopathy outcomes. This was important, as we found that the majority of retinopathy cases detected through routine screening are mild and presumed to be asymptomatic. This will likely be reassuring news for patients that we can screen for this adverse event to detect it early and prevent vision loss.
Another important difference was that we assessed the risk of retinopathy associated with using over 6 mg/kg per day, between 5 and 6 mg/kg per day, and less than 5 mg/kg per day, whereas the highest dosing group assessed in the 2014 study included all patients using over 5 mg/kg per day. The risk was considerably higher in the > 6 mg/kg per day group than in the 5-6 mg/kg per day group.
Q: How can rheumatologists and ophthalmologists use this new information specifically to better treat their patients?
A: Our study provides more accurate estimates of the risk of HCQ retinopathy than in prior studies. These risk estimates can be used when rheumatologists (and other clinicians who prescribe HCQ) consider the risks and benefits of this otherwise important and well-tolerated medication. The risk associated with different dose ranges could also inform dosing decisions, since dosing over 6 mg/kg per day may be more of a concern than using doses in the 5-6 mg/kg range. Ophthalmologists can also use these new risk estimates to counsel patients of the importance of HCQ retinopathy screening and can also hopefully provide some reassurance to patients that the risk of severe retinopathy is low as long as they are being monitored.
The study authors were supported by grants from the National Institute of Arthritis and Musculoskeletal and Skin Diseases and the Rheumatology Research Foundation. The authors report no relevant financial relationships. Dr. Bose and Dr. Mirza had no relevant disclosures.
A version of this article first appeared on Medscape.com.
A new study likely makes the best estimate yet of the degree of retinopathy risk that patients who take the antimalarial drug hydroxychloroquine (HCQ) can expect, deriving mainly from the cumulative dose taken during the first 5 years of use, according to a study published in Annals of Internal Medicine.
HCQ works to decrease activity in a patient’s immune system, which is effective in many cases of systemic lupus erythematosus, one of the most common indications for the drug. However, an adverse outcome of treatment can be HCQ retinopathy, a progressive form of vision loss in patients taking HCQ over an extended period (mostly for longer than 5 years). The disease is often asymptomatic, although some patients do present a paracentral scotoma and a decrease in color vision. Patients may also notice flashing shapes in their vision and find that they have difficulty reading. Eventually, HCQ retinopathy can lead to loss of visual acuity, loss of peripheral vision, and loss of night vision.
Researchers from Kaiser Permanente Northern California and Harvard Medical School analyzed 3,325 persons who received HCQ for 5 or more years between 2004 and 2020. Their goal was to both characterize the long-term risk for incident HCQ retinopathy and examine the degree to which average HCQ dose within the first 5 years of treatment serves as a prediction of the risk.
The researchers then estimated the risk for developing retinopathy after 15 years, according to patients’ average dosing levels during the first 5 years of therapy. Overall, 81 participants developed HCQ retinopathy with overall cumulative incidences of 2.5% after 10 years and 8.6% after 15 years; the risk was greater for those given a higher dose during the first 5 years of treatment.
The mechanism of how HCQ toxicity may occur is still not completely known. There is evidence that toxicity happens because HCQ binds to melanin in both the retinal pigment epithelium and uvea in high concentrations. HCQ can interfere with lysosomal function, leading to oxidation and accumulation of lysosomes, which can cause dysfunction of the retinal pigment epithelium.
Progressive retinopathy can continue even after the drug is stopped. “It’s thought to be a very mild but important risk,” said Nilanjana Bose, MD, MBA, a rheumatologist with Memorial Hermann Health System in Houston. “Patients taking HCQ must be screened for retinal issues, most certainly elderly patients and patients with any kind of comorbidities.”
A 2021 joint position statement from the American College of Rheumatology, American Academy of Dermatology, the Rheumatologic Dermatology Society, and the American Academy of Ophthalmology recommends a baseline eye exam within a few months after starting therapy, then additional screening at 5 years on HCQ and annually thereafter.
“Early detection of retinopathy is important in overall visual prognosis, because toxicity can continue even after discontinuation of the medication,” said Rukhsana G. Mirza, MD, professor of ophthalmology and medical education at Northwestern University in Chicago.
“Examination alone is not sufficient to evaluate early changes, and specialized testing must be done. These include color photos, visual field tests, optical coherence tomography, fundus autofluorescence and in some cases, multifocal electroretinogram. Also, the AAO [American Academy of Ophthalmology] has specific recommendations related to Asian patients as they may have a different pattern of retinopathy that must also be considered.”
More accurate risk measurements
This news organization asked study coauthor April Jorge, MD, assistant professor of medicine in the division of rheumatology, allergy, and immunology at Massachusetts General Hospital and Harvard Medical School, Boston, to discuss the study, how it correlates to past research, and what it adds that’s new and useful to rheumatologists and ophthalmologists:
Question: Your research found that a higher dose of HCQ in the first 5 years of treatment led to a greater risk of retinopathy. Is there any indication that a lower dose given more frequently, either within that 5-year period or longer, would pose a similar risk?
Answer: In our study, we assessed the HCQ dose in the first 5 years of use but followed patients who continued the medication longer than 5 years, through up to 15 years of use. Therefore, we compared the risk of HCQ retinopathy associated with different HCQ dosages but for the same duration of use. We found that for any dose of HCQ, the risk of retinopathy increases the longer the medication is used. However, patients who used a higher dose of HCQ had a higher risk of developing retinopathy over time.
Although current guidelines recommend avoiding any HCQ dose over 5 mg/kg per day to reduce the risk of retinopathy, we found a higher risk of retinopathy associated with dosing over 6 mg/kg per day than between 5 and 6 mg/kg per day and the lowest risk with dosing under 5 mg/kg per day.
Q: How does your study align with and/or expand upon previous research regarding HCQ risk?
A: An important prior study of hydroxychloroquine retinopathy was the 2014 study by Ronald B. Melles, MD, and Michael F. Marmor, MD, published in JAMA Ophthalmology. Prior to our present study, that was the largest study to use the modern screening method (optical coherence tomography) to detect HCQ retinopathy. That screening tool is more sensitive than older methods, so it can detect early/mild cases of retinopathy that are typically asymptomatic. Compared to older studies, that 2014 study found a much higher risk of HCQ retinopathy than was previously appreciated.
However, that 2014 study did have some key limitations that could affect the risk estimates, such as using prevalent cases. A key feature of our present study is that we took several important steps to generate more accurate risk estimates. This included using an incident user cohort and detecting incident retinopathy cases through serial review of optical coherence tomography (screening) studies.
To achieve a high degree of methodologic rigor in correctly identifying retinopathy outcomes, we had expert ophthalmologists perform masked adjudication of all screening studies, and we assessed the intra-rater reliability of these study interpretations. Therefore, our study adds to the literature more accurate estimates of retinopathy risk. We found a lower cumulative incidence of retinopathy than was identified in the 2014 study, but the risk is still noteworthy.
Also unique to our study, we graded the severity of HCQ retinopathy outcomes. This was important, as we found that the majority of retinopathy cases detected through routine screening are mild and presumed to be asymptomatic. This will likely be reassuring news for patients that we can screen for this adverse event to detect it early and prevent vision loss.
Another important difference was that we assessed the risk of retinopathy associated with using over 6 mg/kg per day, between 5 and 6 mg/kg per day, and less than 5 mg/kg per day, whereas the highest dosing group assessed in the 2014 study included all patients using over 5 mg/kg per day. The risk was considerably higher in the > 6 mg/kg per day group than in the 5-6 mg/kg per day group.
Q: How can rheumatologists and ophthalmologists use this new information specifically to better treat their patients?
A: Our study provides more accurate estimates of the risk of HCQ retinopathy than in prior studies. These risk estimates can be used when rheumatologists (and other clinicians who prescribe HCQ) consider the risks and benefits of this otherwise important and well-tolerated medication. The risk associated with different dose ranges could also inform dosing decisions, since dosing over 6 mg/kg per day may be more of a concern than using doses in the 5-6 mg/kg range. Ophthalmologists can also use these new risk estimates to counsel patients of the importance of HCQ retinopathy screening and can also hopefully provide some reassurance to patients that the risk of severe retinopathy is low as long as they are being monitored.
The study authors were supported by grants from the National Institute of Arthritis and Musculoskeletal and Skin Diseases and the Rheumatology Research Foundation. The authors report no relevant financial relationships. Dr. Bose and Dr. Mirza had no relevant disclosures.
A version of this article first appeared on Medscape.com.
A new study likely makes the best estimate yet of the degree of retinopathy risk that patients who take the antimalarial drug hydroxychloroquine (HCQ) can expect, deriving mainly from the cumulative dose taken during the first 5 years of use, according to a study published in Annals of Internal Medicine.
HCQ works to decrease activity in a patient’s immune system, which is effective in many cases of systemic lupus erythematosus, one of the most common indications for the drug. However, an adverse outcome of treatment can be HCQ retinopathy, a progressive form of vision loss in patients taking HCQ over an extended period (mostly for longer than 5 years). The disease is often asymptomatic, although some patients do present a paracentral scotoma and a decrease in color vision. Patients may also notice flashing shapes in their vision and find that they have difficulty reading. Eventually, HCQ retinopathy can lead to loss of visual acuity, loss of peripheral vision, and loss of night vision.
Researchers from Kaiser Permanente Northern California and Harvard Medical School analyzed 3,325 persons who received HCQ for 5 or more years between 2004 and 2020. Their goal was to both characterize the long-term risk for incident HCQ retinopathy and examine the degree to which average HCQ dose within the first 5 years of treatment serves as a prediction of the risk.
The researchers then estimated the risk for developing retinopathy after 15 years, according to patients’ average dosing levels during the first 5 years of therapy. Overall, 81 participants developed HCQ retinopathy with overall cumulative incidences of 2.5% after 10 years and 8.6% after 15 years; the risk was greater for those given a higher dose during the first 5 years of treatment.
The mechanism of how HCQ toxicity may occur is still not completely known. There is evidence that toxicity happens because HCQ binds to melanin in both the retinal pigment epithelium and uvea in high concentrations. HCQ can interfere with lysosomal function, leading to oxidation and accumulation of lysosomes, which can cause dysfunction of the retinal pigment epithelium.
Progressive retinopathy can continue even after the drug is stopped. “It’s thought to be a very mild but important risk,” said Nilanjana Bose, MD, MBA, a rheumatologist with Memorial Hermann Health System in Houston. “Patients taking HCQ must be screened for retinal issues, most certainly elderly patients and patients with any kind of comorbidities.”
A 2021 joint position statement from the American College of Rheumatology, American Academy of Dermatology, the Rheumatologic Dermatology Society, and the American Academy of Ophthalmology recommends a baseline eye exam within a few months after starting therapy, then additional screening at 5 years on HCQ and annually thereafter.
“Early detection of retinopathy is important in overall visual prognosis, because toxicity can continue even after discontinuation of the medication,” said Rukhsana G. Mirza, MD, professor of ophthalmology and medical education at Northwestern University in Chicago.
“Examination alone is not sufficient to evaluate early changes, and specialized testing must be done. These include color photos, visual field tests, optical coherence tomography, fundus autofluorescence and in some cases, multifocal electroretinogram. Also, the AAO [American Academy of Ophthalmology] has specific recommendations related to Asian patients as they may have a different pattern of retinopathy that must also be considered.”
More accurate risk measurements
This news organization asked study coauthor April Jorge, MD, assistant professor of medicine in the division of rheumatology, allergy, and immunology at Massachusetts General Hospital and Harvard Medical School, Boston, to discuss the study, how it correlates to past research, and what it adds that’s new and useful to rheumatologists and ophthalmologists:
Question: Your research found that a higher dose of HCQ in the first 5 years of treatment led to a greater risk of retinopathy. Is there any indication that a lower dose given more frequently, either within that 5-year period or longer, would pose a similar risk?
Answer: In our study, we assessed the HCQ dose in the first 5 years of use but followed patients who continued the medication longer than 5 years, through up to 15 years of use. Therefore, we compared the risk of HCQ retinopathy associated with different HCQ dosages but for the same duration of use. We found that for any dose of HCQ, the risk of retinopathy increases the longer the medication is used. However, patients who used a higher dose of HCQ had a higher risk of developing retinopathy over time.
Although current guidelines recommend avoiding any HCQ dose over 5 mg/kg per day to reduce the risk of retinopathy, we found a higher risk of retinopathy associated with dosing over 6 mg/kg per day than between 5 and 6 mg/kg per day and the lowest risk with dosing under 5 mg/kg per day.
Q: How does your study align with and/or expand upon previous research regarding HCQ risk?
A: An important prior study of hydroxychloroquine retinopathy was the 2014 study by Ronald B. Melles, MD, and Michael F. Marmor, MD, published in JAMA Ophthalmology. Prior to our present study, that was the largest study to use the modern screening method (optical coherence tomography) to detect HCQ retinopathy. That screening tool is more sensitive than older methods, so it can detect early/mild cases of retinopathy that are typically asymptomatic. Compared to older studies, that 2014 study found a much higher risk of HCQ retinopathy than was previously appreciated.
However, that 2014 study did have some key limitations that could affect the risk estimates, such as using prevalent cases. A key feature of our present study is that we took several important steps to generate more accurate risk estimates. This included using an incident user cohort and detecting incident retinopathy cases through serial review of optical coherence tomography (screening) studies.
To achieve a high degree of methodologic rigor in correctly identifying retinopathy outcomes, we had expert ophthalmologists perform masked adjudication of all screening studies, and we assessed the intra-rater reliability of these study interpretations. Therefore, our study adds to the literature more accurate estimates of retinopathy risk. We found a lower cumulative incidence of retinopathy than was identified in the 2014 study, but the risk is still noteworthy.
Also unique to our study, we graded the severity of HCQ retinopathy outcomes. This was important, as we found that the majority of retinopathy cases detected through routine screening are mild and presumed to be asymptomatic. This will likely be reassuring news for patients that we can screen for this adverse event to detect it early and prevent vision loss.
Another important difference was that we assessed the risk of retinopathy associated with using over 6 mg/kg per day, between 5 and 6 mg/kg per day, and less than 5 mg/kg per day, whereas the highest dosing group assessed in the 2014 study included all patients using over 5 mg/kg per day. The risk was considerably higher in the > 6 mg/kg per day group than in the 5-6 mg/kg per day group.
Q: How can rheumatologists and ophthalmologists use this new information specifically to better treat their patients?
A: Our study provides more accurate estimates of the risk of HCQ retinopathy than in prior studies. These risk estimates can be used when rheumatologists (and other clinicians who prescribe HCQ) consider the risks and benefits of this otherwise important and well-tolerated medication. The risk associated with different dose ranges could also inform dosing decisions, since dosing over 6 mg/kg per day may be more of a concern than using doses in the 5-6 mg/kg range. Ophthalmologists can also use these new risk estimates to counsel patients of the importance of HCQ retinopathy screening and can also hopefully provide some reassurance to patients that the risk of severe retinopathy is low as long as they are being monitored.
The study authors were supported by grants from the National Institute of Arthritis and Musculoskeletal and Skin Diseases and the Rheumatology Research Foundation. The authors report no relevant financial relationships. Dr. Bose and Dr. Mirza had no relevant disclosures.
A version of this article first appeared on Medscape.com.
FROM ANNALS OF INTERNAL MEDICINE
A toddler presents with a dark line on a fingernail
Given the over 1-year history of an unchanging longitudinal band of pigment without extension to the proximal or lateral nailfolds or any other nail findings, the most likely diagnosis is benign longitudinal melanonychia.
Longitudinal melanonychia, also known as melanonychia striata, describes a brown to black streak of pigment extending from the nail matrix to the free edge of the nail.1,2
This disorder can occur secondary to a wide variety of benign and pathologic causes including lentigines, nevi, melanoma, chronic trauma, inflammatory skin diseases, systemic diseases, iatrogenic causes, and genetic syndromes.3 In melanocytic causes of longitudinal melanonychia, either melanocytic activation or hyperplasia drive pigmentary development leading to the brown to black band seen in the nail.4 Benign causes of longitudinal melanonychia include benign melanocyte activation, lentigo, and benign nevus.1
What’s the differential diagnosis?
The differential diagnosis for longitudinal melanonychia can include a wide variety of local and systemic causes. For our discussion, we will limit our differential to other locally involved disorders of the nail including subungual melanoma, subungual hematoma, onychomycosis, and glomus tumor.
Subungual melanoma is a rare subtype of acral lentiginous melanoma that most often presents as longitudinal melanonychia. Subungual melanoma is more common in those aged 50-70 years, individuals with personal or family history of melanoma or dysplastic nevus syndrome, and persons with African American, Native American, and Asian descent. Longitudinal melanonychia features that can be concerning for subungual melanoma include the presence of multiple colors, width greater than or equal to 3 mm, blurry borders, rapid increase in size, and extension to the proximal or lateral nailfolds (Hutchinson’s sign). Biopsy is required to make the diagnosis of subungual melanoma but is not necessary for melanonychia without atypical features.
Treatment of subungual melanoma depends on disease stage and can range from wide local excision of the nail apparatus to amputation of the affected digit and management with a medical oncologist. Given the absence of concerning neoplastic findings or personal or family history of melanoma, subungual melanoma is unlikely in this patient.
Subungual hematoma is an accumulation of blood underneath the nail plate that is typically the result of acute or chronic trauma to the distal phalanx. It can present as purple, red, pink, brown, or black discoloration under the nail plate and is most commonly found on the first toe. With acute trauma, pain is usually present upon initial injury. Subungual hematomas typically resolve on their own with normal nail growth. The absence of a history of trauma or pain, and the linear appearance of the lesion in our patient are inconsistent with a subungual hematoma.
Onychomycosis is a fungal infection of the nail caused by dermatophytes, nondermatophytes, or yeasts. It may present with longitudinal melanonychia; however, it more often presents with other nail abnormalities such as nail thickening, yellow discoloration, onycholysis, splitting, subungual hyperkeratosis, and nail plate destruction, which are not present in this patient. Furthermore, onychomycosis is more common in adults than children. Diagnosis is usually made with potassium hydroxide (KOH) preparations, histopathologic examination of nail clippings with a periodic acid-Schiff stain, fungal culture, or PCR.
Glomus tumor is a rare, benign neoplasm originating from cells of the glomus body. It is often found in the subungual region, in addition to other areas rich in glomus bodies such as the fingertips, palms, wrists, and forearms. Subungual glomus tumors present as a red, purple, or blueish lesions under the nail plate. Distal notching or an overlying longitudinal fissure may be present. Subungual glomus tumors are typically associated with pinpoint tenderness, paroxysmal pain, and cold sensitivity, features that are not present in our patient. The history and examination of our patient are much more consistent with benign longitudinal melanonychia.
It appears that melanoma associated with longitudinal melanonychia is very rare in children. According to one review published in 2020, only 12 cases of pediatric subungual melanoma have been reported.5 Recent series have observed longitudinal melanonychia in large sets of children, with findings that demonstrate that the vast majority of longitudinal melanonychia either stops progressing or regresses. These investigations therefore recommend serial observation of longitudinal melanonychia except in rare circumstances.6,7
Given the lack of troubling findings or concerning history, our patient was managed with observation. On follow-up 6 months later, he was found to have no change in his nail pigmentation.
Dr. Haft is an inflammatory skin disease fellow in the division of pediatric and adolescent dermatology; Ms. Sui is a research associate in the department of dermatology, division of pediatric and adolescent dermatology; and Dr. Eichenfield is vice chair of the department of dermatology and professor of dermatology and pediatrics, all at the University of California and Rady Children’s Hospital, San Diego. They have no relevant disclosures.
References
1. Mannava KA et al. Hand Surg. 2013;18(1):133-9.
2. Leung AKC et al. Int J Dermatol. 2019;58(11):1239-45.
3. Andre J and Lateur N. Dermatol Clin. 2006;24(3):329-39.
4. Lee DK and Lipner SR. Ann Med. 2022;54(1):694-712.
5. Smith RJ and Rubin AI. Curr Opin Pediatr. 2020;32(4):506-15. .
6. Matsui Y et al. J Am Acad Dermatol. 2022;86(4):946-8.
7. Lee JS et al. J Am Acad Dermatol. 2022;87(2):366-72.
Given the over 1-year history of an unchanging longitudinal band of pigment without extension to the proximal or lateral nailfolds or any other nail findings, the most likely diagnosis is benign longitudinal melanonychia.
Longitudinal melanonychia, also known as melanonychia striata, describes a brown to black streak of pigment extending from the nail matrix to the free edge of the nail.1,2
This disorder can occur secondary to a wide variety of benign and pathologic causes including lentigines, nevi, melanoma, chronic trauma, inflammatory skin diseases, systemic diseases, iatrogenic causes, and genetic syndromes.3 In melanocytic causes of longitudinal melanonychia, either melanocytic activation or hyperplasia drive pigmentary development leading to the brown to black band seen in the nail.4 Benign causes of longitudinal melanonychia include benign melanocyte activation, lentigo, and benign nevus.1
What’s the differential diagnosis?
The differential diagnosis for longitudinal melanonychia can include a wide variety of local and systemic causes. For our discussion, we will limit our differential to other locally involved disorders of the nail including subungual melanoma, subungual hematoma, onychomycosis, and glomus tumor.
Subungual melanoma is a rare subtype of acral lentiginous melanoma that most often presents as longitudinal melanonychia. Subungual melanoma is more common in those aged 50-70 years, individuals with personal or family history of melanoma or dysplastic nevus syndrome, and persons with African American, Native American, and Asian descent. Longitudinal melanonychia features that can be concerning for subungual melanoma include the presence of multiple colors, width greater than or equal to 3 mm, blurry borders, rapid increase in size, and extension to the proximal or lateral nailfolds (Hutchinson’s sign). Biopsy is required to make the diagnosis of subungual melanoma but is not necessary for melanonychia without atypical features.
Treatment of subungual melanoma depends on disease stage and can range from wide local excision of the nail apparatus to amputation of the affected digit and management with a medical oncologist. Given the absence of concerning neoplastic findings or personal or family history of melanoma, subungual melanoma is unlikely in this patient.
Subungual hematoma is an accumulation of blood underneath the nail plate that is typically the result of acute or chronic trauma to the distal phalanx. It can present as purple, red, pink, brown, or black discoloration under the nail plate and is most commonly found on the first toe. With acute trauma, pain is usually present upon initial injury. Subungual hematomas typically resolve on their own with normal nail growth. The absence of a history of trauma or pain, and the linear appearance of the lesion in our patient are inconsistent with a subungual hematoma.
Onychomycosis is a fungal infection of the nail caused by dermatophytes, nondermatophytes, or yeasts. It may present with longitudinal melanonychia; however, it more often presents with other nail abnormalities such as nail thickening, yellow discoloration, onycholysis, splitting, subungual hyperkeratosis, and nail plate destruction, which are not present in this patient. Furthermore, onychomycosis is more common in adults than children. Diagnosis is usually made with potassium hydroxide (KOH) preparations, histopathologic examination of nail clippings with a periodic acid-Schiff stain, fungal culture, or PCR.
Glomus tumor is a rare, benign neoplasm originating from cells of the glomus body. It is often found in the subungual region, in addition to other areas rich in glomus bodies such as the fingertips, palms, wrists, and forearms. Subungual glomus tumors present as a red, purple, or blueish lesions under the nail plate. Distal notching or an overlying longitudinal fissure may be present. Subungual glomus tumors are typically associated with pinpoint tenderness, paroxysmal pain, and cold sensitivity, features that are not present in our patient. The history and examination of our patient are much more consistent with benign longitudinal melanonychia.
It appears that melanoma associated with longitudinal melanonychia is very rare in children. According to one review published in 2020, only 12 cases of pediatric subungual melanoma have been reported.5 Recent series have observed longitudinal melanonychia in large sets of children, with findings that demonstrate that the vast majority of longitudinal melanonychia either stops progressing or regresses. These investigations therefore recommend serial observation of longitudinal melanonychia except in rare circumstances.6,7
Given the lack of troubling findings or concerning history, our patient was managed with observation. On follow-up 6 months later, he was found to have no change in his nail pigmentation.
Dr. Haft is an inflammatory skin disease fellow in the division of pediatric and adolescent dermatology; Ms. Sui is a research associate in the department of dermatology, division of pediatric and adolescent dermatology; and Dr. Eichenfield is vice chair of the department of dermatology and professor of dermatology and pediatrics, all at the University of California and Rady Children’s Hospital, San Diego. They have no relevant disclosures.
References
1. Mannava KA et al. Hand Surg. 2013;18(1):133-9.
2. Leung AKC et al. Int J Dermatol. 2019;58(11):1239-45.
3. Andre J and Lateur N. Dermatol Clin. 2006;24(3):329-39.
4. Lee DK and Lipner SR. Ann Med. 2022;54(1):694-712.
5. Smith RJ and Rubin AI. Curr Opin Pediatr. 2020;32(4):506-15. .
6. Matsui Y et al. J Am Acad Dermatol. 2022;86(4):946-8.
7. Lee JS et al. J Am Acad Dermatol. 2022;87(2):366-72.
Given the over 1-year history of an unchanging longitudinal band of pigment without extension to the proximal or lateral nailfolds or any other nail findings, the most likely diagnosis is benign longitudinal melanonychia.
Longitudinal melanonychia, also known as melanonychia striata, describes a brown to black streak of pigment extending from the nail matrix to the free edge of the nail.1,2
This disorder can occur secondary to a wide variety of benign and pathologic causes including lentigines, nevi, melanoma, chronic trauma, inflammatory skin diseases, systemic diseases, iatrogenic causes, and genetic syndromes.3 In melanocytic causes of longitudinal melanonychia, either melanocytic activation or hyperplasia drive pigmentary development leading to the brown to black band seen in the nail.4 Benign causes of longitudinal melanonychia include benign melanocyte activation, lentigo, and benign nevus.1
What’s the differential diagnosis?
The differential diagnosis for longitudinal melanonychia can include a wide variety of local and systemic causes. For our discussion, we will limit our differential to other locally involved disorders of the nail including subungual melanoma, subungual hematoma, onychomycosis, and glomus tumor.
Subungual melanoma is a rare subtype of acral lentiginous melanoma that most often presents as longitudinal melanonychia. Subungual melanoma is more common in those aged 50-70 years, individuals with personal or family history of melanoma or dysplastic nevus syndrome, and persons with African American, Native American, and Asian descent. Longitudinal melanonychia features that can be concerning for subungual melanoma include the presence of multiple colors, width greater than or equal to 3 mm, blurry borders, rapid increase in size, and extension to the proximal or lateral nailfolds (Hutchinson’s sign). Biopsy is required to make the diagnosis of subungual melanoma but is not necessary for melanonychia without atypical features.
Treatment of subungual melanoma depends on disease stage and can range from wide local excision of the nail apparatus to amputation of the affected digit and management with a medical oncologist. Given the absence of concerning neoplastic findings or personal or family history of melanoma, subungual melanoma is unlikely in this patient.
Subungual hematoma is an accumulation of blood underneath the nail plate that is typically the result of acute or chronic trauma to the distal phalanx. It can present as purple, red, pink, brown, or black discoloration under the nail plate and is most commonly found on the first toe. With acute trauma, pain is usually present upon initial injury. Subungual hematomas typically resolve on their own with normal nail growth. The absence of a history of trauma or pain, and the linear appearance of the lesion in our patient are inconsistent with a subungual hematoma.
Onychomycosis is a fungal infection of the nail caused by dermatophytes, nondermatophytes, or yeasts. It may present with longitudinal melanonychia; however, it more often presents with other nail abnormalities such as nail thickening, yellow discoloration, onycholysis, splitting, subungual hyperkeratosis, and nail plate destruction, which are not present in this patient. Furthermore, onychomycosis is more common in adults than children. Diagnosis is usually made with potassium hydroxide (KOH) preparations, histopathologic examination of nail clippings with a periodic acid-Schiff stain, fungal culture, or PCR.
Glomus tumor is a rare, benign neoplasm originating from cells of the glomus body. It is often found in the subungual region, in addition to other areas rich in glomus bodies such as the fingertips, palms, wrists, and forearms. Subungual glomus tumors present as a red, purple, or blueish lesions under the nail plate. Distal notching or an overlying longitudinal fissure may be present. Subungual glomus tumors are typically associated with pinpoint tenderness, paroxysmal pain, and cold sensitivity, features that are not present in our patient. The history and examination of our patient are much more consistent with benign longitudinal melanonychia.
It appears that melanoma associated with longitudinal melanonychia is very rare in children. According to one review published in 2020, only 12 cases of pediatric subungual melanoma have been reported.5 Recent series have observed longitudinal melanonychia in large sets of children, with findings that demonstrate that the vast majority of longitudinal melanonychia either stops progressing or regresses. These investigations therefore recommend serial observation of longitudinal melanonychia except in rare circumstances.6,7
Given the lack of troubling findings or concerning history, our patient was managed with observation. On follow-up 6 months later, he was found to have no change in his nail pigmentation.
Dr. Haft is an inflammatory skin disease fellow in the division of pediatric and adolescent dermatology; Ms. Sui is a research associate in the department of dermatology, division of pediatric and adolescent dermatology; and Dr. Eichenfield is vice chair of the department of dermatology and professor of dermatology and pediatrics, all at the University of California and Rady Children’s Hospital, San Diego. They have no relevant disclosures.
References
1. Mannava KA et al. Hand Surg. 2013;18(1):133-9.
2. Leung AKC et al. Int J Dermatol. 2019;58(11):1239-45.
3. Andre J and Lateur N. Dermatol Clin. 2006;24(3):329-39.
4. Lee DK and Lipner SR. Ann Med. 2022;54(1):694-712.
5. Smith RJ and Rubin AI. Curr Opin Pediatr. 2020;32(4):506-15. .
6. Matsui Y et al. J Am Acad Dermatol. 2022;86(4):946-8.
7. Lee JS et al. J Am Acad Dermatol. 2022;87(2):366-72.
Examination findings reveal a 2-mm brown longitudinal band on the radial aspect of the right thumbnail that does not extend into the proximal or lateral nailfolds. The rest of the skin and nail exam is unremarkable.
AD outcomes improved with lebrikizumab and topical steroids
, according to results of the 16-week phase 3 ADhere trial.
“Lebrikizumab, a monoclonal antibody inhibiting interleukin-13, combined with TCS was associated with reduced overall disease severity of moderate to severe AD in adolescents and adults, and had a safety profile consistent with previous lebrikizumab AD studies,” noted lead author Eric L. Simpson, MD, professor of dermatology at Oregon Health & Science University, Portland, and coauthors in their article on the study, which was published in JAMA Dermatology.
The double-blind trial, conducted at 54 sites across Germany, Poland, Canada, and the United States, included 211 patients, mean age 37.2 years, of whom 48.8% were female and roughly 22% were adolescents. Almost 15% were Asian, and about 13% were Black.
At baseline, participants had a score of 16 or higher on the Eczema Area and Severity Index (EASI), a score of 3 or higher on the Investigator’s Global Assessment (IGA) scale, AD covering a body surface area of 10% or greater, and a history of inadequate response to treatment with topical medications.
After a minimum 1-week washout period from topical and systemic therapy, participants were randomized in a 2:1 ratio to receive lebrikizumab plus TCS (n = 145) or placebo plus TCS (n = 66) for 16 weeks.
Lebrikizumab or placebo was administered by subcutaneous injection every 2 weeks; the loading and week-2 doses of lebrikizumab were 500 mg, followed by 250 mg thereafter. All patients were instructed to use low- to mid-potency TCS at their own discretion. Study sites provided a mid-potency TCS (triamcinolone acetonide 0.1% cream) and a low-potency TCS (hydrocortisone 1% cream), with topical calcineurin inhibitors permitted for sensitive skin areas.
Primary outcomes at 16 weeks included a 2-point or more reduction in IGA score from baseline and EASI-75 response. Patients in the lebrikizumab arm had superior responses on both of these outcomes, with statistical significance achieved as early as week 8 and week 4, respectively, and maintained through week 16. Specifically, 41.2% of those treated with lebrikizumab had an IGA reduction of 2 points or more, compared with 22.1% of those receiving placebo plus TCS (P = .01), and the proportion of patients achieving EASI-75 responses was 69.5% vs. 42.2%, respectively (P < .001).
Patients treated with lebrikizumab also showed statistically significant improvements, compared with TCS alone in all key secondary endpoints, “including skin clearance, improvement in itch, itch interference on sleep, and enhanced QoL [quality of life],” noted the authors. “This study captured the clinical benefit of lebrikizumab through the combined end point of physician-assessed clinical sign of skin clearance (EASI-75) and patient-reported outcome of improvement in itch (Pruritus NRS).”
The percentage of patients who achieved the combined endpoint was more than double for the lebrikizumab plus TCS group vs. the group on TCS alone, indicating that patients treated with lebrikizumab plus TCS “were more likely to experience improvement in skin symptoms and itch,” the investigators added.
The authors noted that most treatment-emergent adverse events “were nonserious, mild, or moderate in severity, and did not lead to study discontinuation.” These included conjunctivitis (4.8%), headache (4.8%), hypertension (2.8%), injection-site reactions (2.8%), and herpes infection (3.4%) – all of which occurred in 1.5% or less of patients in the placebo group.
“The higher incidence of conjunctivitis has also been reported in other biologics inhibiting IL [interleukin]–13 and/or IL-4 signaling, as well as lebrikizumab monotherapy studies,” they noted. The 4.8% rate of conjunctivitis reported in the combination study, they added, is “compared with 7.5% frequency in 16-week data from the lebrikizumab monotherapy studies. Although the mechanism remains unclear, it has been reported that conjunctival goblet cell scarcity due to IL-13 and IL-4 inhibition, and subsequent effects on the homeostasis of the conjunctival mucosal surface, results in ocular AEs [adverse events].”
“This truly is a time of great hope and promise for our patients with AD,” commented Zelma Chiesa Fuxench, MD, who was not involved in the study. “The advent of newer, targeted therapeutic agents for AD continues to revolutionize the treatment experience for our patients, offering the possibility of greater AD disease control with a favorable risk profile and less need for blood work monitoring compared to traditional systemic agents.”
On the basis of the study results, Dr. Chiesa Fuxench, of the department of dermatology at the University of Pennsylvania, Philadelphia, said in an interview that “lebrikizumab represents an additional option in the treatment armamentarium for providers who care for patients with AD.” She added that, “while head-to-head trials comparing lebrikizumab to dupilumab, the first FDA-approved biologic for AD, would be beneficial, to the best of my knowledge this data is currently lacking. However, based on the results of this study, we would expect lebrikizumab to work at least similarly to dupilumab, based on the reported improvements in IGA and EASI score.”
Additionally, lebrikizumab showed a favorable safety profile, “with most treatment-emergent adverse effects reported as nonserious and not leading to drug discontinuation,” she said. “Of interest to clinicians may be the reported rates of conjunctivitis in this study. Rates of conjunctivitis for lebrikizumab appear to be lower than those reported in the LIBERTY AD CHRONOS study for dupilumab – a finding that merits further scrutiny in my opinion, as this one of the most frequent treatment-emergent adverse events that I encounter in my clinical practice.”
The study was funded by Dermira, a subsidiary of Eli Lilly. Dr. Simpson reported personal fees and grants from multiple sources, including Dermira and Eli Lilly, the companies developing lebrikizumab. Several authors were employees of Eli Lilly. Dr. Fuxench disclosed serving as a consultant for the Asthma and Allergy Foundation of America, National Eczema Association, Pfizer, AbbVie, and Incyte, for which she has received honoraria for AD-related work. She is the recipient of research grants through Regeneron, Sanofi, Tioga, Vanda, Menlo Therapeutics, Leo Pharma, and Eli Lilly for work related to AD as well as honoraria for continuing medical education work related to AD sponsored through educational grants from Regeneron/Sanofi and Pfizer.
A version of this article first appeared on Medscape.com.
, according to results of the 16-week phase 3 ADhere trial.
“Lebrikizumab, a monoclonal antibody inhibiting interleukin-13, combined with TCS was associated with reduced overall disease severity of moderate to severe AD in adolescents and adults, and had a safety profile consistent with previous lebrikizumab AD studies,” noted lead author Eric L. Simpson, MD, professor of dermatology at Oregon Health & Science University, Portland, and coauthors in their article on the study, which was published in JAMA Dermatology.
The double-blind trial, conducted at 54 sites across Germany, Poland, Canada, and the United States, included 211 patients, mean age 37.2 years, of whom 48.8% were female and roughly 22% were adolescents. Almost 15% were Asian, and about 13% were Black.
At baseline, participants had a score of 16 or higher on the Eczema Area and Severity Index (EASI), a score of 3 or higher on the Investigator’s Global Assessment (IGA) scale, AD covering a body surface area of 10% or greater, and a history of inadequate response to treatment with topical medications.
After a minimum 1-week washout period from topical and systemic therapy, participants were randomized in a 2:1 ratio to receive lebrikizumab plus TCS (n = 145) or placebo plus TCS (n = 66) for 16 weeks.
Lebrikizumab or placebo was administered by subcutaneous injection every 2 weeks; the loading and week-2 doses of lebrikizumab were 500 mg, followed by 250 mg thereafter. All patients were instructed to use low- to mid-potency TCS at their own discretion. Study sites provided a mid-potency TCS (triamcinolone acetonide 0.1% cream) and a low-potency TCS (hydrocortisone 1% cream), with topical calcineurin inhibitors permitted for sensitive skin areas.
Primary outcomes at 16 weeks included a 2-point or more reduction in IGA score from baseline and EASI-75 response. Patients in the lebrikizumab arm had superior responses on both of these outcomes, with statistical significance achieved as early as week 8 and week 4, respectively, and maintained through week 16. Specifically, 41.2% of those treated with lebrikizumab had an IGA reduction of 2 points or more, compared with 22.1% of those receiving placebo plus TCS (P = .01), and the proportion of patients achieving EASI-75 responses was 69.5% vs. 42.2%, respectively (P < .001).
Patients treated with lebrikizumab also showed statistically significant improvements, compared with TCS alone in all key secondary endpoints, “including skin clearance, improvement in itch, itch interference on sleep, and enhanced QoL [quality of life],” noted the authors. “This study captured the clinical benefit of lebrikizumab through the combined end point of physician-assessed clinical sign of skin clearance (EASI-75) and patient-reported outcome of improvement in itch (Pruritus NRS).”
The percentage of patients who achieved the combined endpoint was more than double for the lebrikizumab plus TCS group vs. the group on TCS alone, indicating that patients treated with lebrikizumab plus TCS “were more likely to experience improvement in skin symptoms and itch,” the investigators added.
The authors noted that most treatment-emergent adverse events “were nonserious, mild, or moderate in severity, and did not lead to study discontinuation.” These included conjunctivitis (4.8%), headache (4.8%), hypertension (2.8%), injection-site reactions (2.8%), and herpes infection (3.4%) – all of which occurred in 1.5% or less of patients in the placebo group.
“The higher incidence of conjunctivitis has also been reported in other biologics inhibiting IL [interleukin]–13 and/or IL-4 signaling, as well as lebrikizumab monotherapy studies,” they noted. The 4.8% rate of conjunctivitis reported in the combination study, they added, is “compared with 7.5% frequency in 16-week data from the lebrikizumab monotherapy studies. Although the mechanism remains unclear, it has been reported that conjunctival goblet cell scarcity due to IL-13 and IL-4 inhibition, and subsequent effects on the homeostasis of the conjunctival mucosal surface, results in ocular AEs [adverse events].”
“This truly is a time of great hope and promise for our patients with AD,” commented Zelma Chiesa Fuxench, MD, who was not involved in the study. “The advent of newer, targeted therapeutic agents for AD continues to revolutionize the treatment experience for our patients, offering the possibility of greater AD disease control with a favorable risk profile and less need for blood work monitoring compared to traditional systemic agents.”
On the basis of the study results, Dr. Chiesa Fuxench, of the department of dermatology at the University of Pennsylvania, Philadelphia, said in an interview that “lebrikizumab represents an additional option in the treatment armamentarium for providers who care for patients with AD.” She added that, “while head-to-head trials comparing lebrikizumab to dupilumab, the first FDA-approved biologic for AD, would be beneficial, to the best of my knowledge this data is currently lacking. However, based on the results of this study, we would expect lebrikizumab to work at least similarly to dupilumab, based on the reported improvements in IGA and EASI score.”
Additionally, lebrikizumab showed a favorable safety profile, “with most treatment-emergent adverse effects reported as nonserious and not leading to drug discontinuation,” she said. “Of interest to clinicians may be the reported rates of conjunctivitis in this study. Rates of conjunctivitis for lebrikizumab appear to be lower than those reported in the LIBERTY AD CHRONOS study for dupilumab – a finding that merits further scrutiny in my opinion, as this one of the most frequent treatment-emergent adverse events that I encounter in my clinical practice.”
The study was funded by Dermira, a subsidiary of Eli Lilly. Dr. Simpson reported personal fees and grants from multiple sources, including Dermira and Eli Lilly, the companies developing lebrikizumab. Several authors were employees of Eli Lilly. Dr. Fuxench disclosed serving as a consultant for the Asthma and Allergy Foundation of America, National Eczema Association, Pfizer, AbbVie, and Incyte, for which she has received honoraria for AD-related work. She is the recipient of research grants through Regeneron, Sanofi, Tioga, Vanda, Menlo Therapeutics, Leo Pharma, and Eli Lilly for work related to AD as well as honoraria for continuing medical education work related to AD sponsored through educational grants from Regeneron/Sanofi and Pfizer.
A version of this article first appeared on Medscape.com.
, according to results of the 16-week phase 3 ADhere trial.
“Lebrikizumab, a monoclonal antibody inhibiting interleukin-13, combined with TCS was associated with reduced overall disease severity of moderate to severe AD in adolescents and adults, and had a safety profile consistent with previous lebrikizumab AD studies,” noted lead author Eric L. Simpson, MD, professor of dermatology at Oregon Health & Science University, Portland, and coauthors in their article on the study, which was published in JAMA Dermatology.
The double-blind trial, conducted at 54 sites across Germany, Poland, Canada, and the United States, included 211 patients, mean age 37.2 years, of whom 48.8% were female and roughly 22% were adolescents. Almost 15% were Asian, and about 13% were Black.
At baseline, participants had a score of 16 or higher on the Eczema Area and Severity Index (EASI), a score of 3 or higher on the Investigator’s Global Assessment (IGA) scale, AD covering a body surface area of 10% or greater, and a history of inadequate response to treatment with topical medications.
After a minimum 1-week washout period from topical and systemic therapy, participants were randomized in a 2:1 ratio to receive lebrikizumab plus TCS (n = 145) or placebo plus TCS (n = 66) for 16 weeks.
Lebrikizumab or placebo was administered by subcutaneous injection every 2 weeks; the loading and week-2 doses of lebrikizumab were 500 mg, followed by 250 mg thereafter. All patients were instructed to use low- to mid-potency TCS at their own discretion. Study sites provided a mid-potency TCS (triamcinolone acetonide 0.1% cream) and a low-potency TCS (hydrocortisone 1% cream), with topical calcineurin inhibitors permitted for sensitive skin areas.
Primary outcomes at 16 weeks included a 2-point or more reduction in IGA score from baseline and EASI-75 response. Patients in the lebrikizumab arm had superior responses on both of these outcomes, with statistical significance achieved as early as week 8 and week 4, respectively, and maintained through week 16. Specifically, 41.2% of those treated with lebrikizumab had an IGA reduction of 2 points or more, compared with 22.1% of those receiving placebo plus TCS (P = .01), and the proportion of patients achieving EASI-75 responses was 69.5% vs. 42.2%, respectively (P < .001).
Patients treated with lebrikizumab also showed statistically significant improvements, compared with TCS alone in all key secondary endpoints, “including skin clearance, improvement in itch, itch interference on sleep, and enhanced QoL [quality of life],” noted the authors. “This study captured the clinical benefit of lebrikizumab through the combined end point of physician-assessed clinical sign of skin clearance (EASI-75) and patient-reported outcome of improvement in itch (Pruritus NRS).”
The percentage of patients who achieved the combined endpoint was more than double for the lebrikizumab plus TCS group vs. the group on TCS alone, indicating that patients treated with lebrikizumab plus TCS “were more likely to experience improvement in skin symptoms and itch,” the investigators added.
The authors noted that most treatment-emergent adverse events “were nonserious, mild, or moderate in severity, and did not lead to study discontinuation.” These included conjunctivitis (4.8%), headache (4.8%), hypertension (2.8%), injection-site reactions (2.8%), and herpes infection (3.4%) – all of which occurred in 1.5% or less of patients in the placebo group.
“The higher incidence of conjunctivitis has also been reported in other biologics inhibiting IL [interleukin]–13 and/or IL-4 signaling, as well as lebrikizumab monotherapy studies,” they noted. The 4.8% rate of conjunctivitis reported in the combination study, they added, is “compared with 7.5% frequency in 16-week data from the lebrikizumab monotherapy studies. Although the mechanism remains unclear, it has been reported that conjunctival goblet cell scarcity due to IL-13 and IL-4 inhibition, and subsequent effects on the homeostasis of the conjunctival mucosal surface, results in ocular AEs [adverse events].”
“This truly is a time of great hope and promise for our patients with AD,” commented Zelma Chiesa Fuxench, MD, who was not involved in the study. “The advent of newer, targeted therapeutic agents for AD continues to revolutionize the treatment experience for our patients, offering the possibility of greater AD disease control with a favorable risk profile and less need for blood work monitoring compared to traditional systemic agents.”
On the basis of the study results, Dr. Chiesa Fuxench, of the department of dermatology at the University of Pennsylvania, Philadelphia, said in an interview that “lebrikizumab represents an additional option in the treatment armamentarium for providers who care for patients with AD.” She added that, “while head-to-head trials comparing lebrikizumab to dupilumab, the first FDA-approved biologic for AD, would be beneficial, to the best of my knowledge this data is currently lacking. However, based on the results of this study, we would expect lebrikizumab to work at least similarly to dupilumab, based on the reported improvements in IGA and EASI score.”
Additionally, lebrikizumab showed a favorable safety profile, “with most treatment-emergent adverse effects reported as nonserious and not leading to drug discontinuation,” she said. “Of interest to clinicians may be the reported rates of conjunctivitis in this study. Rates of conjunctivitis for lebrikizumab appear to be lower than those reported in the LIBERTY AD CHRONOS study for dupilumab – a finding that merits further scrutiny in my opinion, as this one of the most frequent treatment-emergent adverse events that I encounter in my clinical practice.”
The study was funded by Dermira, a subsidiary of Eli Lilly. Dr. Simpson reported personal fees and grants from multiple sources, including Dermira and Eli Lilly, the companies developing lebrikizumab. Several authors were employees of Eli Lilly. Dr. Fuxench disclosed serving as a consultant for the Asthma and Allergy Foundation of America, National Eczema Association, Pfizer, AbbVie, and Incyte, for which she has received honoraria for AD-related work. She is the recipient of research grants through Regeneron, Sanofi, Tioga, Vanda, Menlo Therapeutics, Leo Pharma, and Eli Lilly for work related to AD as well as honoraria for continuing medical education work related to AD sponsored through educational grants from Regeneron/Sanofi and Pfizer.
A version of this article first appeared on Medscape.com.
FROM JAMA DERMATOLOGY
Manicure gone wrong leads to cancer diagnosis
. Now, she and her doctor are spreading the word about her ordeal as a lesson that speed and persistence in seeking treatment are the keys that make her type of cancer – squamous cell carcinoma – completely curable.
“She cut me, and the cut wasn’t just a regular cuticle cut. She cut me deep, and that was one of the first times that happened to me,” Grace Garcia, 50, told TODAY.com, recalling the November 2021 incident.
Ms. Garcia had been getting her nails done regularly for 20 years, she said, but happened to go to a different salon than her usual spot because she couldn’t get an appointment during the busy pre-Thanksgiving season. She doesn’t recall whether the technician opened packaging that signals unused tools.
She put antibiotic ointment on the cut, but it didn’t heal after a few days. Eventually, the skin closed and a darkened bump formed. It was painful. She went to her doctor, who said it was a “callus from writing,” she told TODAY.com. But it was on her ring finger, which didn’t seem connected to writing. Her doctor said to keep an eye on it.
Five months after the cut occurred, she mentioned it during a gynecology appointment and was referred to a dermatologist, who also advised keeping an eye on it. A wart developed. She went back to her primary care physician and then to another dermatologist. The spot was biopsied.
Squamous cell carcinoma is a common type of skin cancer, according to the American Academy of Dermatology. It can have many causes, but the cause in Ms. Garcia’s case was both very common and very rare: human papillomavirus, or HPV. HPV is a virus that infects millions of people every year, but it’s not a typical cause of skin cancer.
“It’s pretty rare for several reasons. Generally speaking, the strains that cause cancer from an HPV standpoint tend to be more sexually transmitted,” dermatologist Teo Soleymani told TODAY.com. “In Grace’s case, she had an injury, which became the portal of entry. So that thick skin that we have on our hands and feet that acts as a natural barrier against infections and things like that was no longer the case, and the virus was able to infect her skin.”
Dr. Soleymani said Ms. Garcia’s persistence to get answers likely saved her from losing a finger.
“Your outcomes are entirely dictated by how early you catch them, and very often they’re completely curable,” he said. “Her persistence – not only was she able to have a great outcome, she probably saved herself from having her finger amputated.”
. Now, she and her doctor are spreading the word about her ordeal as a lesson that speed and persistence in seeking treatment are the keys that make her type of cancer – squamous cell carcinoma – completely curable.
“She cut me, and the cut wasn’t just a regular cuticle cut. She cut me deep, and that was one of the first times that happened to me,” Grace Garcia, 50, told TODAY.com, recalling the November 2021 incident.
Ms. Garcia had been getting her nails done regularly for 20 years, she said, but happened to go to a different salon than her usual spot because she couldn’t get an appointment during the busy pre-Thanksgiving season. She doesn’t recall whether the technician opened packaging that signals unused tools.
She put antibiotic ointment on the cut, but it didn’t heal after a few days. Eventually, the skin closed and a darkened bump formed. It was painful. She went to her doctor, who said it was a “callus from writing,” she told TODAY.com. But it was on her ring finger, which didn’t seem connected to writing. Her doctor said to keep an eye on it.
Five months after the cut occurred, she mentioned it during a gynecology appointment and was referred to a dermatologist, who also advised keeping an eye on it. A wart developed. She went back to her primary care physician and then to another dermatologist. The spot was biopsied.
Squamous cell carcinoma is a common type of skin cancer, according to the American Academy of Dermatology. It can have many causes, but the cause in Ms. Garcia’s case was both very common and very rare: human papillomavirus, or HPV. HPV is a virus that infects millions of people every year, but it’s not a typical cause of skin cancer.
“It’s pretty rare for several reasons. Generally speaking, the strains that cause cancer from an HPV standpoint tend to be more sexually transmitted,” dermatologist Teo Soleymani told TODAY.com. “In Grace’s case, she had an injury, which became the portal of entry. So that thick skin that we have on our hands and feet that acts as a natural barrier against infections and things like that was no longer the case, and the virus was able to infect her skin.”
Dr. Soleymani said Ms. Garcia’s persistence to get answers likely saved her from losing a finger.
“Your outcomes are entirely dictated by how early you catch them, and very often they’re completely curable,” he said. “Her persistence – not only was she able to have a great outcome, she probably saved herself from having her finger amputated.”
. Now, she and her doctor are spreading the word about her ordeal as a lesson that speed and persistence in seeking treatment are the keys that make her type of cancer – squamous cell carcinoma – completely curable.
“She cut me, and the cut wasn’t just a regular cuticle cut. She cut me deep, and that was one of the first times that happened to me,” Grace Garcia, 50, told TODAY.com, recalling the November 2021 incident.
Ms. Garcia had been getting her nails done regularly for 20 years, she said, but happened to go to a different salon than her usual spot because she couldn’t get an appointment during the busy pre-Thanksgiving season. She doesn’t recall whether the technician opened packaging that signals unused tools.
She put antibiotic ointment on the cut, but it didn’t heal after a few days. Eventually, the skin closed and a darkened bump formed. It was painful. She went to her doctor, who said it was a “callus from writing,” she told TODAY.com. But it was on her ring finger, which didn’t seem connected to writing. Her doctor said to keep an eye on it.
Five months after the cut occurred, she mentioned it during a gynecology appointment and was referred to a dermatologist, who also advised keeping an eye on it. A wart developed. She went back to her primary care physician and then to another dermatologist. The spot was biopsied.
Squamous cell carcinoma is a common type of skin cancer, according to the American Academy of Dermatology. It can have many causes, but the cause in Ms. Garcia’s case was both very common and very rare: human papillomavirus, or HPV. HPV is a virus that infects millions of people every year, but it’s not a typical cause of skin cancer.
“It’s pretty rare for several reasons. Generally speaking, the strains that cause cancer from an HPV standpoint tend to be more sexually transmitted,” dermatologist Teo Soleymani told TODAY.com. “In Grace’s case, she had an injury, which became the portal of entry. So that thick skin that we have on our hands and feet that acts as a natural barrier against infections and things like that was no longer the case, and the virus was able to infect her skin.”
Dr. Soleymani said Ms. Garcia’s persistence to get answers likely saved her from losing a finger.
“Your outcomes are entirely dictated by how early you catch them, and very often they’re completely curable,” he said. “Her persistence – not only was she able to have a great outcome, she probably saved herself from having her finger amputated.”
Scaly facial plaques
This patient was experiencing a flare of his psoriasis. Three factors contributed to the flare: noncompliance with his treatment regimen, decreased sunlight in the winter, and his lithium therapy. Though carcinogenic, certain wavelengths of UV light are beneficial for psoriasis, and the shorter days of winter can cause flaring of psoriasis (or relative flaring). In addition, lithium—the most effective therapy for this patient’s bipolar disorder—can worsen psoriasis.
Psoriasis is a chronic multisystem inflammatory disorder with characteristic skin findings that include well-demarcated micaceous plaques, nail pitting, and sometimes tendon pain and inflammatory arthritis. Severity can range from small, thin plaques that are intermittently noticeable on the elbows or knees to widespread ash-like plaques covering most of the body.
Good topical choices for facial skin include hydrocortisone 2.5% cream or desonide 0.05%. Nonsteroidal topical therapies that are safe for facial skin include tacrolimus 0.1% ointment or pimecrolimus 1% cream.1 These options may be used twice daily until the disease is controlled.
In many cases (as in this one), the patient’s previous psoriasis outbreaks could not be controlled with topical therapy alone. The patient had not responded to a previous methotrexate regimen, and more recently had been clear for several years on systemic ustekinumab, a monoclonal antibody. Dosed every 12 weeks, or sometimes every 8 weeks, ustekinumab is given by subcutaneous injection, usually in the abdomen, through normal skin. Ustekinumab was recently approved for home use with just 4 injections per year for maintenance therapy. However, the infrequency of the injections sometimes leads to noncompliance, as occurred with this patient. He had missed 2 doses since taking over his own dosing regimen.
Ultimately, the patient’s flare resolved when he was transitioned back to in-office treatment with ustekinumab.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.
1. Woo SM, Choi JW, Yoon HS, et al. Classification of facial psoriasis based on the distributions of facial lesions. J Am Acad Dermatol. 2008;58:959-63. doi: 10.1016/j.jaad.2008.02.006

This patient was experiencing a flare of his psoriasis. Three factors contributed to the flare: noncompliance with his treatment regimen, decreased sunlight in the winter, and his lithium therapy. Though carcinogenic, certain wavelengths of UV light are beneficial for psoriasis, and the shorter days of winter can cause flaring of psoriasis (or relative flaring). In addition, lithium—the most effective therapy for this patient’s bipolar disorder—can worsen psoriasis.
Psoriasis is a chronic multisystem inflammatory disorder with characteristic skin findings that include well-demarcated micaceous plaques, nail pitting, and sometimes tendon pain and inflammatory arthritis. Severity can range from small, thin plaques that are intermittently noticeable on the elbows or knees to widespread ash-like plaques covering most of the body.
Good topical choices for facial skin include hydrocortisone 2.5% cream or desonide 0.05%. Nonsteroidal topical therapies that are safe for facial skin include tacrolimus 0.1% ointment or pimecrolimus 1% cream.1 These options may be used twice daily until the disease is controlled.
In many cases (as in this one), the patient’s previous psoriasis outbreaks could not be controlled with topical therapy alone. The patient had not responded to a previous methotrexate regimen, and more recently had been clear for several years on systemic ustekinumab, a monoclonal antibody. Dosed every 12 weeks, or sometimes every 8 weeks, ustekinumab is given by subcutaneous injection, usually in the abdomen, through normal skin. Ustekinumab was recently approved for home use with just 4 injections per year for maintenance therapy. However, the infrequency of the injections sometimes leads to noncompliance, as occurred with this patient. He had missed 2 doses since taking over his own dosing regimen.
Ultimately, the patient’s flare resolved when he was transitioned back to in-office treatment with ustekinumab.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.

This patient was experiencing a flare of his psoriasis. Three factors contributed to the flare: noncompliance with his treatment regimen, decreased sunlight in the winter, and his lithium therapy. Though carcinogenic, certain wavelengths of UV light are beneficial for psoriasis, and the shorter days of winter can cause flaring of psoriasis (or relative flaring). In addition, lithium—the most effective therapy for this patient’s bipolar disorder—can worsen psoriasis.
Psoriasis is a chronic multisystem inflammatory disorder with characteristic skin findings that include well-demarcated micaceous plaques, nail pitting, and sometimes tendon pain and inflammatory arthritis. Severity can range from small, thin plaques that are intermittently noticeable on the elbows or knees to widespread ash-like plaques covering most of the body.
Good topical choices for facial skin include hydrocortisone 2.5% cream or desonide 0.05%. Nonsteroidal topical therapies that are safe for facial skin include tacrolimus 0.1% ointment or pimecrolimus 1% cream.1 These options may be used twice daily until the disease is controlled.
In many cases (as in this one), the patient’s previous psoriasis outbreaks could not be controlled with topical therapy alone. The patient had not responded to a previous methotrexate regimen, and more recently had been clear for several years on systemic ustekinumab, a monoclonal antibody. Dosed every 12 weeks, or sometimes every 8 weeks, ustekinumab is given by subcutaneous injection, usually in the abdomen, through normal skin. Ustekinumab was recently approved for home use with just 4 injections per year for maintenance therapy. However, the infrequency of the injections sometimes leads to noncompliance, as occurred with this patient. He had missed 2 doses since taking over his own dosing regimen.
Ultimately, the patient’s flare resolved when he was transitioned back to in-office treatment with ustekinumab.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.
1. Woo SM, Choi JW, Yoon HS, et al. Classification of facial psoriasis based on the distributions of facial lesions. J Am Acad Dermatol. 2008;58:959-63. doi: 10.1016/j.jaad.2008.02.006
1. Woo SM, Choi JW, Yoon HS, et al. Classification of facial psoriasis based on the distributions of facial lesions. J Am Acad Dermatol. 2008;58:959-63. doi: 10.1016/j.jaad.2008.02.006