User login
Obesity boosts gestational diabetes risk in women with PCOS
In a population-based cohort study that included more than 1.2 million hospital live births, PCOS was associated with a 5% increase in risk for gestational diabetes. Almost 90% of this association was mediated by obesity.
“Women with PCOS are at higher risk, but it’s only 5% higher than the general population. However, that risk rises substantially with obesity,” senior author Maria P. Velez, MD, PhD, clinician-scientist and associate professor of obstetrics and gynecology at Queen’s University, Kingston, Ont., said in an interview. “Our study highlights the need for counseling our patients about the importance of weight optimization, ideally starting with lifestyle changes like diet and exercise.”The findings were published in the Journal of Obstetrics and Gynaecology Canada.
Major mediator
The estimated prevalence of PCOS is 8%-13%, and affected patients often present with anovulation, hyperandrogenism, obesity, metabolic syndrome, and infertility. Prepregnancy insulin resistance is common among women with PCOS and may play a major part in the pathogenesis of gestational diabetes. In addition, PCOS is often accompanied by excess weight gain; about 60% of women with PCOS are overweight or obese.
Previous research has shown that PCOS is a risk factor for gestational diabetes independent of obesity, while other research has shown that obesity has an important effect on this risk.
For the current study, the researchers used causal mediation analysis to elucidate more clearly the effect of obesity on the development of gestational diabetes among patients with PCOS. No previous study has used causal mediation analysis to examine this relationship.
Using data from linked universal health databases in Ontario, the researchers analyzed data on 1,268,901 births between 2006 and 2018. Of these births, 386,748 were associated with maternal PCOS.
The rate of gestational diabetes was higher among women with PCOS (60.2 per 1000 births), compared with women without PCOS (48.6 per 1,000 births). The finding resulted in an adjusted relative risk of 1.05. Obesity mediated 89.7% of this association.
“We hope that these data will inform preconception counseling and gestational diabetes screening in pregnant women with PCOS,” said Dr. Velez. “We have the data now to counsel our patients on the importance of weight management before pregnancy. But we need more resources, such as specialized clinics, to help these patients cope with managing their weight. We can tell our patients to work on their weight management, but they need much more support from the health care system.”
Results ‘not surprising’
Commenting on the study, Francine Hippolyte, MD, vice chair of obstetrics and gynecology at Long Island Jewish Medical Center, Katz Women’s Hospital, New Hyde Park, N.Y., said that the results are “not at all surprising.” Dr. Hippolyte was not involved in the research.
“We do know that PCOS is and should be treated as a metabolic syndrome. It’s a lot more than just infertility or changes or abnormalities with one’s menstrual cycle. It impacts a woman’s risk for diabetes, prediabetes, and abnormal lipid profile, regardless of whether or not she is obese,” said Dr. Hippolyte.
She agrees with the need for specialized clinics to help such vulnerable patients manage their weight.
“It would be great if insurances would cover things like nutritional counseling or have nutritionists on their roster so that patients can easily access that service. Many patients want to do right, especially preconceptually, but it is difficult without having access to resources. Unfortunately, as clinicians, we’re not as well versed in nutrition as we would like to be or should be, so we need a multidisciplinary approach. We need nutrition and weight loss clinics and proper services to really help these patients.”
The study was supported by the Canadian Institute of Health Research and ICES. Dr. Velez and Dr. Hippolyte reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In a population-based cohort study that included more than 1.2 million hospital live births, PCOS was associated with a 5% increase in risk for gestational diabetes. Almost 90% of this association was mediated by obesity.
“Women with PCOS are at higher risk, but it’s only 5% higher than the general population. However, that risk rises substantially with obesity,” senior author Maria P. Velez, MD, PhD, clinician-scientist and associate professor of obstetrics and gynecology at Queen’s University, Kingston, Ont., said in an interview. “Our study highlights the need for counseling our patients about the importance of weight optimization, ideally starting with lifestyle changes like diet and exercise.”The findings were published in the Journal of Obstetrics and Gynaecology Canada.
Major mediator
The estimated prevalence of PCOS is 8%-13%, and affected patients often present with anovulation, hyperandrogenism, obesity, metabolic syndrome, and infertility. Prepregnancy insulin resistance is common among women with PCOS and may play a major part in the pathogenesis of gestational diabetes. In addition, PCOS is often accompanied by excess weight gain; about 60% of women with PCOS are overweight or obese.
Previous research has shown that PCOS is a risk factor for gestational diabetes independent of obesity, while other research has shown that obesity has an important effect on this risk.
For the current study, the researchers used causal mediation analysis to elucidate more clearly the effect of obesity on the development of gestational diabetes among patients with PCOS. No previous study has used causal mediation analysis to examine this relationship.
Using data from linked universal health databases in Ontario, the researchers analyzed data on 1,268,901 births between 2006 and 2018. Of these births, 386,748 were associated with maternal PCOS.
The rate of gestational diabetes was higher among women with PCOS (60.2 per 1000 births), compared with women without PCOS (48.6 per 1,000 births). The finding resulted in an adjusted relative risk of 1.05. Obesity mediated 89.7% of this association.
“We hope that these data will inform preconception counseling and gestational diabetes screening in pregnant women with PCOS,” said Dr. Velez. “We have the data now to counsel our patients on the importance of weight management before pregnancy. But we need more resources, such as specialized clinics, to help these patients cope with managing their weight. We can tell our patients to work on their weight management, but they need much more support from the health care system.”
Results ‘not surprising’
Commenting on the study, Francine Hippolyte, MD, vice chair of obstetrics and gynecology at Long Island Jewish Medical Center, Katz Women’s Hospital, New Hyde Park, N.Y., said that the results are “not at all surprising.” Dr. Hippolyte was not involved in the research.
“We do know that PCOS is and should be treated as a metabolic syndrome. It’s a lot more than just infertility or changes or abnormalities with one’s menstrual cycle. It impacts a woman’s risk for diabetes, prediabetes, and abnormal lipid profile, regardless of whether or not she is obese,” said Dr. Hippolyte.
She agrees with the need for specialized clinics to help such vulnerable patients manage their weight.
“It would be great if insurances would cover things like nutritional counseling or have nutritionists on their roster so that patients can easily access that service. Many patients want to do right, especially preconceptually, but it is difficult without having access to resources. Unfortunately, as clinicians, we’re not as well versed in nutrition as we would like to be or should be, so we need a multidisciplinary approach. We need nutrition and weight loss clinics and proper services to really help these patients.”
The study was supported by the Canadian Institute of Health Research and ICES. Dr. Velez and Dr. Hippolyte reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In a population-based cohort study that included more than 1.2 million hospital live births, PCOS was associated with a 5% increase in risk for gestational diabetes. Almost 90% of this association was mediated by obesity.
“Women with PCOS are at higher risk, but it’s only 5% higher than the general population. However, that risk rises substantially with obesity,” senior author Maria P. Velez, MD, PhD, clinician-scientist and associate professor of obstetrics and gynecology at Queen’s University, Kingston, Ont., said in an interview. “Our study highlights the need for counseling our patients about the importance of weight optimization, ideally starting with lifestyle changes like diet and exercise.”The findings were published in the Journal of Obstetrics and Gynaecology Canada.
Major mediator
The estimated prevalence of PCOS is 8%-13%, and affected patients often present with anovulation, hyperandrogenism, obesity, metabolic syndrome, and infertility. Prepregnancy insulin resistance is common among women with PCOS and may play a major part in the pathogenesis of gestational diabetes. In addition, PCOS is often accompanied by excess weight gain; about 60% of women with PCOS are overweight or obese.
Previous research has shown that PCOS is a risk factor for gestational diabetes independent of obesity, while other research has shown that obesity has an important effect on this risk.
For the current study, the researchers used causal mediation analysis to elucidate more clearly the effect of obesity on the development of gestational diabetes among patients with PCOS. No previous study has used causal mediation analysis to examine this relationship.
Using data from linked universal health databases in Ontario, the researchers analyzed data on 1,268,901 births between 2006 and 2018. Of these births, 386,748 were associated with maternal PCOS.
The rate of gestational diabetes was higher among women with PCOS (60.2 per 1000 births), compared with women without PCOS (48.6 per 1,000 births). The finding resulted in an adjusted relative risk of 1.05. Obesity mediated 89.7% of this association.
“We hope that these data will inform preconception counseling and gestational diabetes screening in pregnant women with PCOS,” said Dr. Velez. “We have the data now to counsel our patients on the importance of weight management before pregnancy. But we need more resources, such as specialized clinics, to help these patients cope with managing their weight. We can tell our patients to work on their weight management, but they need much more support from the health care system.”
Results ‘not surprising’
Commenting on the study, Francine Hippolyte, MD, vice chair of obstetrics and gynecology at Long Island Jewish Medical Center, Katz Women’s Hospital, New Hyde Park, N.Y., said that the results are “not at all surprising.” Dr. Hippolyte was not involved in the research.
“We do know that PCOS is and should be treated as a metabolic syndrome. It’s a lot more than just infertility or changes or abnormalities with one’s menstrual cycle. It impacts a woman’s risk for diabetes, prediabetes, and abnormal lipid profile, regardless of whether or not she is obese,” said Dr. Hippolyte.
She agrees with the need for specialized clinics to help such vulnerable patients manage their weight.
“It would be great if insurances would cover things like nutritional counseling or have nutritionists on their roster so that patients can easily access that service. Many patients want to do right, especially preconceptually, but it is difficult without having access to resources. Unfortunately, as clinicians, we’re not as well versed in nutrition as we would like to be or should be, so we need a multidisciplinary approach. We need nutrition and weight loss clinics and proper services to really help these patients.”
The study was supported by the Canadian Institute of Health Research and ICES. Dr. Velez and Dr. Hippolyte reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM JOURNAL OF OBSTETRICS AND GYNAECOLOGY CANADA
SGLT2 inhibitors in type 2 diabetes linked to lower risk of developing GI cancers
VANCOUVER –
The SGLT2 inhibitors emerged superior to DPP4 inhibitors for reducing risk of colorectal, hepatic, esophageal, and other GI cancers except pancreatic cancer, said study investigator Shu-Yen Emily Chan, MD, a gastroenterologist in the departments of medicine and epidemiology at Weiss Memorial Hospital, Chicago.
On the basis of the findings, physicians could consider the SGLT2s canagliflozin, dapagliflozin, and empagliflozin or a GLP-1 as first-line therapy, particularly for people with T2D who are at elevated risk for GI cancers, Dr. Chan said in an interview at the American College of Gastroenterology (ACG): 2023 Annual Scientific Meeting.
Previous research focused on potential cardiovascular or renal benefits associated with SGLT2s, “but there are few looking at GI cancer risk and these medications,” she added. Most earlier studies in cancer have been preclinical and observational studies on colorectal cancer or hepatocellular carcinoma.
Using the TriNetX database of millions of medical claims from 92 hospitals across the United States, Dr. Chan and colleagues identified 706,390 adults who began first-line SGLT2 inhibitor therapy. They used propensity matching to link these patients with 706,390 other adults who began taking a DDP4 inhibitor (sitagliptin, saxagliptin, linagliptin, or alogliptin).
All participants had been diagnosed with type 2 diabetes. Patients were prescribed an SGLT2 inhibitor at least three times, and any cancer diagnosis that occurred at least 6 months after starting therapy was noted. Anyone with a history of cancer, cancer recurrence, or metastatic disease was excluded from the population-based cohort study.
In addition to evaluating a large number of patients, the study is notable for including people with ulcerative colitis and Crohn’s disease and for evaluating every GI cancer – esophageal, gastric, small intestinal, colorectal, rectal, anal, hepatic, biliary, and gallbladder malignancies.
Key findings
Among adults who received an SGLT2 inhibitor, there was a 15% decrease in overall risk of developing any GI cancer, compared with those who received a DPP4 inhibitor (hazard ratio, 0.85; 95% confidence interval, 0.82-0.88).
Colon cancer was the most common malignancy in the study. Dr. Chan and colleagues identified colon cancer among 1,789 people, or 0.25% of those taking an SGLT2 inhibitor, compared with 3,283 people, or 0.46%, of those taking a DPP4 inhibitor.
SGLT2 inhibitors were associated with a 16% decrease in risk of gastric cancer (HR, 0.84; 95% CI; 0.74-0.945; P = .005), a 13% decrease in risk of liver and intrahepatic bile duct cancer (HR, 0.87; 95% CI, 0.81-0.95), and a 22% decrease in risk of colon cancer (HR, 0.781; 95% CI, 0.74-0.83; P < .001), compared with the DPP4 medications.
The only cancer more likely in the SGLT2 inhibitor group than in the DPP4 inhibitor group was pancreatic cancer (HR, 1.035; 95% CI, 0.964-1.111; P = .340).
The SLGT2 inhibitor class also was superior to metformin for reducing risk of GI cancers.
Asked whether the study findings should alter current practice, Dr. Chan said that the study is new and hasn’t yet been published. “More studies will be needed and included in official guidelines before the findings become practice-changing,” she said.
Limitations of the study include residual confounding, absence of family cancer history, and information bias. Strengths include the large, national database and propensity score matching.
‘Eye-opening’ study
“It is a good study, and eye-opening because it shows that one class of diabetes medications is better than another one,” said session co-moderator Kenneth J. Vega, MD, professor of medicine and chief of the division of gastroenterology and hepatology at Augusta University–Medical College of Georgia.
Dr. Vega shared his theory on why diabetes medications could reduce risk of GI cancers. “I would think reducing diabetes means you can control inflammation ... and better controlling inflammation leads you to have less cancers.”
He added, “I think we need more long-term studies.”
The study was independently supported. Dr. Chan and Dr. Vega report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
VANCOUVER –
The SGLT2 inhibitors emerged superior to DPP4 inhibitors for reducing risk of colorectal, hepatic, esophageal, and other GI cancers except pancreatic cancer, said study investigator Shu-Yen Emily Chan, MD, a gastroenterologist in the departments of medicine and epidemiology at Weiss Memorial Hospital, Chicago.
On the basis of the findings, physicians could consider the SGLT2s canagliflozin, dapagliflozin, and empagliflozin or a GLP-1 as first-line therapy, particularly for people with T2D who are at elevated risk for GI cancers, Dr. Chan said in an interview at the American College of Gastroenterology (ACG): 2023 Annual Scientific Meeting.
Previous research focused on potential cardiovascular or renal benefits associated with SGLT2s, “but there are few looking at GI cancer risk and these medications,” she added. Most earlier studies in cancer have been preclinical and observational studies on colorectal cancer or hepatocellular carcinoma.
Using the TriNetX database of millions of medical claims from 92 hospitals across the United States, Dr. Chan and colleagues identified 706,390 adults who began first-line SGLT2 inhibitor therapy. They used propensity matching to link these patients with 706,390 other adults who began taking a DDP4 inhibitor (sitagliptin, saxagliptin, linagliptin, or alogliptin).
All participants had been diagnosed with type 2 diabetes. Patients were prescribed an SGLT2 inhibitor at least three times, and any cancer diagnosis that occurred at least 6 months after starting therapy was noted. Anyone with a history of cancer, cancer recurrence, or metastatic disease was excluded from the population-based cohort study.
In addition to evaluating a large number of patients, the study is notable for including people with ulcerative colitis and Crohn’s disease and for evaluating every GI cancer – esophageal, gastric, small intestinal, colorectal, rectal, anal, hepatic, biliary, and gallbladder malignancies.
Key findings
Among adults who received an SGLT2 inhibitor, there was a 15% decrease in overall risk of developing any GI cancer, compared with those who received a DPP4 inhibitor (hazard ratio, 0.85; 95% confidence interval, 0.82-0.88).
Colon cancer was the most common malignancy in the study. Dr. Chan and colleagues identified colon cancer among 1,789 people, or 0.25% of those taking an SGLT2 inhibitor, compared with 3,283 people, or 0.46%, of those taking a DPP4 inhibitor.
SGLT2 inhibitors were associated with a 16% decrease in risk of gastric cancer (HR, 0.84; 95% CI; 0.74-0.945; P = .005), a 13% decrease in risk of liver and intrahepatic bile duct cancer (HR, 0.87; 95% CI, 0.81-0.95), and a 22% decrease in risk of colon cancer (HR, 0.781; 95% CI, 0.74-0.83; P < .001), compared with the DPP4 medications.
The only cancer more likely in the SGLT2 inhibitor group than in the DPP4 inhibitor group was pancreatic cancer (HR, 1.035; 95% CI, 0.964-1.111; P = .340).
The SLGT2 inhibitor class also was superior to metformin for reducing risk of GI cancers.
Asked whether the study findings should alter current practice, Dr. Chan said that the study is new and hasn’t yet been published. “More studies will be needed and included in official guidelines before the findings become practice-changing,” she said.
Limitations of the study include residual confounding, absence of family cancer history, and information bias. Strengths include the large, national database and propensity score matching.
‘Eye-opening’ study
“It is a good study, and eye-opening because it shows that one class of diabetes medications is better than another one,” said session co-moderator Kenneth J. Vega, MD, professor of medicine and chief of the division of gastroenterology and hepatology at Augusta University–Medical College of Georgia.
Dr. Vega shared his theory on why diabetes medications could reduce risk of GI cancers. “I would think reducing diabetes means you can control inflammation ... and better controlling inflammation leads you to have less cancers.”
He added, “I think we need more long-term studies.”
The study was independently supported. Dr. Chan and Dr. Vega report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
VANCOUVER –
The SGLT2 inhibitors emerged superior to DPP4 inhibitors for reducing risk of colorectal, hepatic, esophageal, and other GI cancers except pancreatic cancer, said study investigator Shu-Yen Emily Chan, MD, a gastroenterologist in the departments of medicine and epidemiology at Weiss Memorial Hospital, Chicago.
On the basis of the findings, physicians could consider the SGLT2s canagliflozin, dapagliflozin, and empagliflozin or a GLP-1 as first-line therapy, particularly for people with T2D who are at elevated risk for GI cancers, Dr. Chan said in an interview at the American College of Gastroenterology (ACG): 2023 Annual Scientific Meeting.
Previous research focused on potential cardiovascular or renal benefits associated with SGLT2s, “but there are few looking at GI cancer risk and these medications,” she added. Most earlier studies in cancer have been preclinical and observational studies on colorectal cancer or hepatocellular carcinoma.
Using the TriNetX database of millions of medical claims from 92 hospitals across the United States, Dr. Chan and colleagues identified 706,390 adults who began first-line SGLT2 inhibitor therapy. They used propensity matching to link these patients with 706,390 other adults who began taking a DDP4 inhibitor (sitagliptin, saxagliptin, linagliptin, or alogliptin).
All participants had been diagnosed with type 2 diabetes. Patients were prescribed an SGLT2 inhibitor at least three times, and any cancer diagnosis that occurred at least 6 months after starting therapy was noted. Anyone with a history of cancer, cancer recurrence, or metastatic disease was excluded from the population-based cohort study.
In addition to evaluating a large number of patients, the study is notable for including people with ulcerative colitis and Crohn’s disease and for evaluating every GI cancer – esophageal, gastric, small intestinal, colorectal, rectal, anal, hepatic, biliary, and gallbladder malignancies.
Key findings
Among adults who received an SGLT2 inhibitor, there was a 15% decrease in overall risk of developing any GI cancer, compared with those who received a DPP4 inhibitor (hazard ratio, 0.85; 95% confidence interval, 0.82-0.88).
Colon cancer was the most common malignancy in the study. Dr. Chan and colleagues identified colon cancer among 1,789 people, or 0.25% of those taking an SGLT2 inhibitor, compared with 3,283 people, or 0.46%, of those taking a DPP4 inhibitor.
SGLT2 inhibitors were associated with a 16% decrease in risk of gastric cancer (HR, 0.84; 95% CI; 0.74-0.945; P = .005), a 13% decrease in risk of liver and intrahepatic bile duct cancer (HR, 0.87; 95% CI, 0.81-0.95), and a 22% decrease in risk of colon cancer (HR, 0.781; 95% CI, 0.74-0.83; P < .001), compared with the DPP4 medications.
The only cancer more likely in the SGLT2 inhibitor group than in the DPP4 inhibitor group was pancreatic cancer (HR, 1.035; 95% CI, 0.964-1.111; P = .340).
The SLGT2 inhibitor class also was superior to metformin for reducing risk of GI cancers.
Asked whether the study findings should alter current practice, Dr. Chan said that the study is new and hasn’t yet been published. “More studies will be needed and included in official guidelines before the findings become practice-changing,” she said.
Limitations of the study include residual confounding, absence of family cancer history, and information bias. Strengths include the large, national database and propensity score matching.
‘Eye-opening’ study
“It is a good study, and eye-opening because it shows that one class of diabetes medications is better than another one,” said session co-moderator Kenneth J. Vega, MD, professor of medicine and chief of the division of gastroenterology and hepatology at Augusta University–Medical College of Georgia.
Dr. Vega shared his theory on why diabetes medications could reduce risk of GI cancers. “I would think reducing diabetes means you can control inflammation ... and better controlling inflammation leads you to have less cancers.”
He added, “I think we need more long-term studies.”
The study was independently supported. Dr. Chan and Dr. Vega report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
AT ACG 2023
Higher weight loss on tirzepatide links to seven factors
TOPLINE:
Among the 3,188 people with type 2 diabetes who were adherent to their tirzepatide (Mounjaro, Lilly) regimen in four pivotal trials of the agent, a quarter achieved at least a 15% cut from their baseline body weight after 40-42 weeks of treatment, and researchers found seven baseline variables that were significantly linked with a higher incidence of this level of weight loss.
say the authors.
METHODOLOGY:
- Investigators conducted a post hoc analysis of data collected from a total of 3,188 people with type 2 diabetes who had been adherent to their assigned tirzepatide regimen for 40-42 weeks in any one of four pivotal trials of the agent.
- The researchers aimed to identify predictors of a reduction in body weight of at least 15% with tirzepatide treatment at any of the three tested doses – 5 mg, 10 mg, or 15 mg – which were administered by subcutaneous injection once a week.
- All four trials that provided data prohibited concurrent therapy that would promote weight loss, and the people included in the analysis did not receive any rescue medications for controlling glycemia.
- The primary efficacy measure in all four studies was the ability of tirzepatide to improve glycemic control (measured by A1c level), compared with placebo, semaglutide (Ozempic) 1 mg SC once weekly, insulin degludec (Tresiba, Novo Nordisk), or insulin glargine (Basaglar, Lilly).
TAKEAWAY:
- Among the 3,188 people who remained adherent to their tirzepatide regimen for 40-42 weeks, 792 (25%) experienced a weight reduction of at least 15% from baseline.
- Multivariate analysis of baseline covariates showed that these seven factors were significantly linked with greater than or equal to 15% weight loss: higher tirzepatide dose, being female, being of White or Asian race, being of younger age, undergoing treatment with metformin, having better glycemic control (based on lower A1c and lower fasting serum glucose), and having lower non–high-density lipoprotein cholesterol level.
- During follow-up, achievement of at least a 15% cut in baseline body weight was significantly associated with greater reductions in A1c, fasting serum glucose level, waist circumference, blood pressure, serum triglyceride level, and serum level of the liver enzyme alanine transaminase.
IN PRACTICE:
“These findings may provide valuable information to clinicians and people with type 2 diabetes regarding the likelihood of achieving substantial body weight reduction with tirzepatide and also help to signal likely improvements to be seen in a range of cardiometabolic risk parameters with tirzepatide-induced weight loss,” the authors concluded in their report.
SOURCE:
The study was largely run by researchers who are employees of Lilly, the company that markets tirzepatide (Mounjaro). It was published in Diabetes Care.
LIMITATIONS:
- The analysis was post hoc.
- The follow-up was limited.
- The analysis focused entirely on baseline parameters as potential predictors of weight loss magnitude.
DISCLOSURES:
The study was funded by Eli Lilly, the company that markets tirzepatide (Mounjaro) and that sponsored the SURPASS trials. Six authors are employees of Lilly, one is a contractor for Lilly, and the two remaining authors have had financial relationships with Lilly and with several other companies.
A version of this article first appeared on Medscape.com.
TOPLINE:
Among the 3,188 people with type 2 diabetes who were adherent to their tirzepatide (Mounjaro, Lilly) regimen in four pivotal trials of the agent, a quarter achieved at least a 15% cut from their baseline body weight after 40-42 weeks of treatment, and researchers found seven baseline variables that were significantly linked with a higher incidence of this level of weight loss.
say the authors.
METHODOLOGY:
- Investigators conducted a post hoc analysis of data collected from a total of 3,188 people with type 2 diabetes who had been adherent to their assigned tirzepatide regimen for 40-42 weeks in any one of four pivotal trials of the agent.
- The researchers aimed to identify predictors of a reduction in body weight of at least 15% with tirzepatide treatment at any of the three tested doses – 5 mg, 10 mg, or 15 mg – which were administered by subcutaneous injection once a week.
- All four trials that provided data prohibited concurrent therapy that would promote weight loss, and the people included in the analysis did not receive any rescue medications for controlling glycemia.
- The primary efficacy measure in all four studies was the ability of tirzepatide to improve glycemic control (measured by A1c level), compared with placebo, semaglutide (Ozempic) 1 mg SC once weekly, insulin degludec (Tresiba, Novo Nordisk), or insulin glargine (Basaglar, Lilly).
TAKEAWAY:
- Among the 3,188 people who remained adherent to their tirzepatide regimen for 40-42 weeks, 792 (25%) experienced a weight reduction of at least 15% from baseline.
- Multivariate analysis of baseline covariates showed that these seven factors were significantly linked with greater than or equal to 15% weight loss: higher tirzepatide dose, being female, being of White or Asian race, being of younger age, undergoing treatment with metformin, having better glycemic control (based on lower A1c and lower fasting serum glucose), and having lower non–high-density lipoprotein cholesterol level.
- During follow-up, achievement of at least a 15% cut in baseline body weight was significantly associated with greater reductions in A1c, fasting serum glucose level, waist circumference, blood pressure, serum triglyceride level, and serum level of the liver enzyme alanine transaminase.
IN PRACTICE:
“These findings may provide valuable information to clinicians and people with type 2 diabetes regarding the likelihood of achieving substantial body weight reduction with tirzepatide and also help to signal likely improvements to be seen in a range of cardiometabolic risk parameters with tirzepatide-induced weight loss,” the authors concluded in their report.
SOURCE:
The study was largely run by researchers who are employees of Lilly, the company that markets tirzepatide (Mounjaro). It was published in Diabetes Care.
LIMITATIONS:
- The analysis was post hoc.
- The follow-up was limited.
- The analysis focused entirely on baseline parameters as potential predictors of weight loss magnitude.
DISCLOSURES:
The study was funded by Eli Lilly, the company that markets tirzepatide (Mounjaro) and that sponsored the SURPASS trials. Six authors are employees of Lilly, one is a contractor for Lilly, and the two remaining authors have had financial relationships with Lilly and with several other companies.
A version of this article first appeared on Medscape.com.
TOPLINE:
Among the 3,188 people with type 2 diabetes who were adherent to their tirzepatide (Mounjaro, Lilly) regimen in four pivotal trials of the agent, a quarter achieved at least a 15% cut from their baseline body weight after 40-42 weeks of treatment, and researchers found seven baseline variables that were significantly linked with a higher incidence of this level of weight loss.
say the authors.
METHODOLOGY:
- Investigators conducted a post hoc analysis of data collected from a total of 3,188 people with type 2 diabetes who had been adherent to their assigned tirzepatide regimen for 40-42 weeks in any one of four pivotal trials of the agent.
- The researchers aimed to identify predictors of a reduction in body weight of at least 15% with tirzepatide treatment at any of the three tested doses – 5 mg, 10 mg, or 15 mg – which were administered by subcutaneous injection once a week.
- All four trials that provided data prohibited concurrent therapy that would promote weight loss, and the people included in the analysis did not receive any rescue medications for controlling glycemia.
- The primary efficacy measure in all four studies was the ability of tirzepatide to improve glycemic control (measured by A1c level), compared with placebo, semaglutide (Ozempic) 1 mg SC once weekly, insulin degludec (Tresiba, Novo Nordisk), or insulin glargine (Basaglar, Lilly).
TAKEAWAY:
- Among the 3,188 people who remained adherent to their tirzepatide regimen for 40-42 weeks, 792 (25%) experienced a weight reduction of at least 15% from baseline.
- Multivariate analysis of baseline covariates showed that these seven factors were significantly linked with greater than or equal to 15% weight loss: higher tirzepatide dose, being female, being of White or Asian race, being of younger age, undergoing treatment with metformin, having better glycemic control (based on lower A1c and lower fasting serum glucose), and having lower non–high-density lipoprotein cholesterol level.
- During follow-up, achievement of at least a 15% cut in baseline body weight was significantly associated with greater reductions in A1c, fasting serum glucose level, waist circumference, blood pressure, serum triglyceride level, and serum level of the liver enzyme alanine transaminase.
IN PRACTICE:
“These findings may provide valuable information to clinicians and people with type 2 diabetes regarding the likelihood of achieving substantial body weight reduction with tirzepatide and also help to signal likely improvements to be seen in a range of cardiometabolic risk parameters with tirzepatide-induced weight loss,” the authors concluded in their report.
SOURCE:
The study was largely run by researchers who are employees of Lilly, the company that markets tirzepatide (Mounjaro). It was published in Diabetes Care.
LIMITATIONS:
- The analysis was post hoc.
- The follow-up was limited.
- The analysis focused entirely on baseline parameters as potential predictors of weight loss magnitude.
DISCLOSURES:
The study was funded by Eli Lilly, the company that markets tirzepatide (Mounjaro) and that sponsored the SURPASS trials. Six authors are employees of Lilly, one is a contractor for Lilly, and the two remaining authors have had financial relationships with Lilly and with several other companies.
A version of this article first appeared on Medscape.com.
Tricyclics may raise fracture risk in type 2 diabetes
VANCOUVER – , independent of any prevalent neuropathy, according to findings from an analysis of a large, randomized clinical trial.
Although the findings are suggestive, they don’t definitively pin blame on TCAs, said Rachel Elam, MD, who presented the study at the annual meeting of the American Society for Bone and Mineral Research. “I think that there’s not enough information to conclude that tricyclic antidepressants directly lead to fractures, but I think it opens the door [to] something we should look into more. Is it being mediated by a better predictor, or is it the medication itself? I think it’s more hypothesis generating,” said Dr. Elam, an assistant professor of medicine in the division of rheumatology at the Medical College of Georgia, Augusta.
Patients with type 2 diabetes are known to be at increased risk of fracture, but prediction tools tend to underestimate this risk, Dr. Elam said. “Type 2 diabetes–specific clinical risk factors may be helpful for finding out fracture risk in this population,” Dr. Elam said during her talk.
Glycemic control is one candidate risk factor because advanced glycation end products are linked to reduced bone strength. Other factors include antidiabetic medication use, neuropathy, and microvascular disease, which has been linked to increased cortical porosity.
The study examined a somewhat younger population than previous surveys, having drawn from the Look AHEAD-C clinical trial, which examined the effects of an intensive lifestyle intervention on type 2 diabetes. Look AHEAD-C included 4,697 participants aged 45-75 from 16 U.S. clinical sites. Participants had a body mass index of 25.0 kg/m2 or higher and hemoglobin A1c levels of 11% or below.
Dr. Elam cited the database’s inclusion of factors like A1c levels, renal parameters, and diabetic neuropathy. “It gave us a really good population to look at those risk factors” in a large group of people with type 2 diabetes, she said.
Over a median follow-up of 16.6 years, there were 649 participants with incident first clinical fracture(s). Statistically significant factors predicting fracture risk included TCA use (hazard ratio, 2.24; 95% confidence interval, 1.14-4.43), female gender (HR, 2.20; 95% CI, 1.83-2.66), insulin use (HR, 1.26; 95% CI, 1.02-1.57), increases in A1c level (per 1% increase: HR, 1.12; 95% CI, 1.04-1.20), age (HR, 1.02; 95% CI, 1.01-1.04), other or mixed race/ethnicity (HR, 0.68; 95% CI, 0.52-0.87), Hispanic White race/ethnicity (HR, 0.60; 95% CI, 0.39-0.91), non-Hispanic Black race/ethnicity (HR, 0.35; 95% CI, 0.26-0.47), and estrogen use (HR, 0.65; 95% CI, 0.44-0.98).
During the Q&A session following the presentation, Elsa Strotmeyer, PhD, commented that TCAs have been linked to central nervous system pathways in falls in other populations. “It’s a very nice study. It’s important to look at the diabetes complications related to the fracture risk, but I thought that they should have emphasized some more of the diabetes complications being related to fracture rather than these tricyclic antidepressants, because that is not a unique factor to that population,” said Dr. Strotmeyer, who is an associate professor of epidemiology at the University of Pittsburgh.
Instead, she noted a different strength of the study. “The study population is important because they’re a relatively young population with type 2 diabetes, compared to many studies [that] have been published in older populations. Showing similar things that we found in older populations was the unique piece and the important piece of this study,” Dr. Strotmeyer said.
Ultimately, the model wasn’t sufficient to be used as a fall risk predictor, but it should inform future work, according to Dr. Elam. “I think it does lay some new groundwork that when we’re looking forward, it may [help in building] other models to better predict fracture risk in type 2 diabetes. Things that would be important to include [in future models] would be medication use, such as tricyclic antidepressants,” and to make sure we include glycemic control, A1c, and insulin medication.
The study was independently funded. Dr. Elam and Dr. Strotmeyer report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
VANCOUVER – , independent of any prevalent neuropathy, according to findings from an analysis of a large, randomized clinical trial.
Although the findings are suggestive, they don’t definitively pin blame on TCAs, said Rachel Elam, MD, who presented the study at the annual meeting of the American Society for Bone and Mineral Research. “I think that there’s not enough information to conclude that tricyclic antidepressants directly lead to fractures, but I think it opens the door [to] something we should look into more. Is it being mediated by a better predictor, or is it the medication itself? I think it’s more hypothesis generating,” said Dr. Elam, an assistant professor of medicine in the division of rheumatology at the Medical College of Georgia, Augusta.
Patients with type 2 diabetes are known to be at increased risk of fracture, but prediction tools tend to underestimate this risk, Dr. Elam said. “Type 2 diabetes–specific clinical risk factors may be helpful for finding out fracture risk in this population,” Dr. Elam said during her talk.
Glycemic control is one candidate risk factor because advanced glycation end products are linked to reduced bone strength. Other factors include antidiabetic medication use, neuropathy, and microvascular disease, which has been linked to increased cortical porosity.
The study examined a somewhat younger population than previous surveys, having drawn from the Look AHEAD-C clinical trial, which examined the effects of an intensive lifestyle intervention on type 2 diabetes. Look AHEAD-C included 4,697 participants aged 45-75 from 16 U.S. clinical sites. Participants had a body mass index of 25.0 kg/m2 or higher and hemoglobin A1c levels of 11% or below.
Dr. Elam cited the database’s inclusion of factors like A1c levels, renal parameters, and diabetic neuropathy. “It gave us a really good population to look at those risk factors” in a large group of people with type 2 diabetes, she said.
Over a median follow-up of 16.6 years, there were 649 participants with incident first clinical fracture(s). Statistically significant factors predicting fracture risk included TCA use (hazard ratio, 2.24; 95% confidence interval, 1.14-4.43), female gender (HR, 2.20; 95% CI, 1.83-2.66), insulin use (HR, 1.26; 95% CI, 1.02-1.57), increases in A1c level (per 1% increase: HR, 1.12; 95% CI, 1.04-1.20), age (HR, 1.02; 95% CI, 1.01-1.04), other or mixed race/ethnicity (HR, 0.68; 95% CI, 0.52-0.87), Hispanic White race/ethnicity (HR, 0.60; 95% CI, 0.39-0.91), non-Hispanic Black race/ethnicity (HR, 0.35; 95% CI, 0.26-0.47), and estrogen use (HR, 0.65; 95% CI, 0.44-0.98).
During the Q&A session following the presentation, Elsa Strotmeyer, PhD, commented that TCAs have been linked to central nervous system pathways in falls in other populations. “It’s a very nice study. It’s important to look at the diabetes complications related to the fracture risk, but I thought that they should have emphasized some more of the diabetes complications being related to fracture rather than these tricyclic antidepressants, because that is not a unique factor to that population,” said Dr. Strotmeyer, who is an associate professor of epidemiology at the University of Pittsburgh.
Instead, she noted a different strength of the study. “The study population is important because they’re a relatively young population with type 2 diabetes, compared to many studies [that] have been published in older populations. Showing similar things that we found in older populations was the unique piece and the important piece of this study,” Dr. Strotmeyer said.
Ultimately, the model wasn’t sufficient to be used as a fall risk predictor, but it should inform future work, according to Dr. Elam. “I think it does lay some new groundwork that when we’re looking forward, it may [help in building] other models to better predict fracture risk in type 2 diabetes. Things that would be important to include [in future models] would be medication use, such as tricyclic antidepressants,” and to make sure we include glycemic control, A1c, and insulin medication.
The study was independently funded. Dr. Elam and Dr. Strotmeyer report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
VANCOUVER – , independent of any prevalent neuropathy, according to findings from an analysis of a large, randomized clinical trial.
Although the findings are suggestive, they don’t definitively pin blame on TCAs, said Rachel Elam, MD, who presented the study at the annual meeting of the American Society for Bone and Mineral Research. “I think that there’s not enough information to conclude that tricyclic antidepressants directly lead to fractures, but I think it opens the door [to] something we should look into more. Is it being mediated by a better predictor, or is it the medication itself? I think it’s more hypothesis generating,” said Dr. Elam, an assistant professor of medicine in the division of rheumatology at the Medical College of Georgia, Augusta.
Patients with type 2 diabetes are known to be at increased risk of fracture, but prediction tools tend to underestimate this risk, Dr. Elam said. “Type 2 diabetes–specific clinical risk factors may be helpful for finding out fracture risk in this population,” Dr. Elam said during her talk.
Glycemic control is one candidate risk factor because advanced glycation end products are linked to reduced bone strength. Other factors include antidiabetic medication use, neuropathy, and microvascular disease, which has been linked to increased cortical porosity.
The study examined a somewhat younger population than previous surveys, having drawn from the Look AHEAD-C clinical trial, which examined the effects of an intensive lifestyle intervention on type 2 diabetes. Look AHEAD-C included 4,697 participants aged 45-75 from 16 U.S. clinical sites. Participants had a body mass index of 25.0 kg/m2 or higher and hemoglobin A1c levels of 11% or below.
Dr. Elam cited the database’s inclusion of factors like A1c levels, renal parameters, and diabetic neuropathy. “It gave us a really good population to look at those risk factors” in a large group of people with type 2 diabetes, she said.
Over a median follow-up of 16.6 years, there were 649 participants with incident first clinical fracture(s). Statistically significant factors predicting fracture risk included TCA use (hazard ratio, 2.24; 95% confidence interval, 1.14-4.43), female gender (HR, 2.20; 95% CI, 1.83-2.66), insulin use (HR, 1.26; 95% CI, 1.02-1.57), increases in A1c level (per 1% increase: HR, 1.12; 95% CI, 1.04-1.20), age (HR, 1.02; 95% CI, 1.01-1.04), other or mixed race/ethnicity (HR, 0.68; 95% CI, 0.52-0.87), Hispanic White race/ethnicity (HR, 0.60; 95% CI, 0.39-0.91), non-Hispanic Black race/ethnicity (HR, 0.35; 95% CI, 0.26-0.47), and estrogen use (HR, 0.65; 95% CI, 0.44-0.98).
During the Q&A session following the presentation, Elsa Strotmeyer, PhD, commented that TCAs have been linked to central nervous system pathways in falls in other populations. “It’s a very nice study. It’s important to look at the diabetes complications related to the fracture risk, but I thought that they should have emphasized some more of the diabetes complications being related to fracture rather than these tricyclic antidepressants, because that is not a unique factor to that population,” said Dr. Strotmeyer, who is an associate professor of epidemiology at the University of Pittsburgh.
Instead, she noted a different strength of the study. “The study population is important because they’re a relatively young population with type 2 diabetes, compared to many studies [that] have been published in older populations. Showing similar things that we found in older populations was the unique piece and the important piece of this study,” Dr. Strotmeyer said.
Ultimately, the model wasn’t sufficient to be used as a fall risk predictor, but it should inform future work, according to Dr. Elam. “I think it does lay some new groundwork that when we’re looking forward, it may [help in building] other models to better predict fracture risk in type 2 diabetes. Things that would be important to include [in future models] would be medication use, such as tricyclic antidepressants,” and to make sure we include glycemic control, A1c, and insulin medication.
The study was independently funded. Dr. Elam and Dr. Strotmeyer report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
AT ASBMR 2023
Common meds link to sudden cardiac arrest in type 2 diabetes
HAMBURG, Germany – , shows the first such analysis of real-world, primary care data.
People with type 2 diabetes who do not have a history of CVD have almost three times the risk of SCA if they take antipsychotic medications and nearly double the risk if they take certain antibiotics that prolong the QT interval, notably, macrolides and fluoroquinolones.
“These data show that commonly prescribed drugs - antipsychotic medications, used by about 3% of people with type 2 diabetes, and antibiotics, taken by 5% to 10%, convey an increased risk of sudden cardiac arrest in those without a history of cardiovascular disease,” said Peter Harms, MSc, who presented the study at the annual meeting of the European Association for the Study of Diabetes. Another drug associated with an increase in SCA among patients with diabetes was domperidone, an antinausea medication.
“Perhaps these drugs could be avoided in some cases, and GPs should be more aware of the possible consequences of their use,” he added. “If the patient has type 2 diabetes, then maybe it’s better to avoid some of these medications and try and cope without them, or at least find an alternative antibiotic.”
Mr. Harms, an epidemiologist from Amsterdam University Medical Centers, highlighted that their study was unique because the investigators drew upon primary care data. “These data are extensive, and we find a lot of associations which are very real.”
SCA is associated with 50% of all cardiac deaths and accounts for 20% of all mortality in high-income countries. Of those people who experience SCA, 80% of cases prove fatal.
“As the name suggests, it is difficult to predict because it is sudden, especially in people without a cardiovascular disease history,” Mr. Harms pointed out in an interview with this news organization. He highlighted that “around half of those who experience SCA, often between the ages of 40 and 60 years, have never seen a cardiologist, but many do have type 2 diabetes.
“We need to better understand how to recognize people at risk of SCA, know who to watch and how to prevent these events,” he emphasized.
Vladimira Fejfarova, MD, comoderated the session and commented on the study. “From the clinical point of view, it’s necessary to evaluate risk factors that can contribute to sudden cardiac arrest.”
Overall, the researchers found that, among people with type 2 diabetes who do not have a history of CVD, hypoglycemia, severe hypertension, dyslipidemia, and use of QTc-prolonging medications are associated with SCA risk. Among people with type 2 diabetes and CVD, albuminuria and heart failure are associated with SCA risk.
Dr. Fejfarova added: “With type 2 diabetes and also type 1, we need to look more at adverse events, especially when treating infections with macrolides, but also mycotic infections, because antimycotic drugs are known to influence QT intervals that could contribute to sudden cardiac arrest.
“We need to be more cautious with prescribing certain antibiotics that have these side effects in our patients with diabetes,” asserted Dr. Fejfarova, from the Diabetes Centre, Institute for Clinical and Experimental Medicine, Prague.
Type 2 diabetes doubles the risk of SCA
The researcher decided to investigate the population of people with type 2 diabetes because their risk of SCD is around twice that of those without type 2 diabetes. Because these patients have relatively frequent checkups with general practitioners, Mr. Harms turned to primary care databases that contained comprehensive and relatively routine information on risk indicators.
Longitudinal associations between clinical characteristics of 3,919 patients with type 2 diabetes – both those with and those without a history of CVD – and SCA (a total of 689 patients) were determined.
Cases were found in the AmsteRdam REsuscitation STtudies (ARREST) registry of out-of-hospital resuscitation attempts by emergency medical services in the Dutch region of Noord-Holland from 2010 to 2019. Case patients were matched with up to five control patients. The control group comprised people with type 2 diabetes who had not experienced an SCA. Control patients were sourced from the same primary care practices who were of similar age and sex. Clinical measurements, including blood pressure and blood glucose readings, medication use, and medical history for the 5 years leading up to an SCA, were obtained from general practice records. A multivariable analysis was performed, and results were stratified for people with and for those without a history of CVD.
Of particular interest were drugs that interfere with cardiac function, including some prokinetic, antibiotic, and antipsychotic medications. All of the drugs are known to be associated with a change in QTc prolongation. Examples include domperidone (QTc-prolonging prokinetic), macrolides and fluoroquinolones (QTc-prolonging antibiotics), and haloperidol (a QTc-prolonging antipsychotic).
Antibiotic and antipsychotic use might contribute to SCA in T2D
Case patients and control patients were similar in age, hemoglobin A1c level, and other characteristics with the exception that more patients with SCA had a history of CVD (40.0% vs. 29.4%).
“Looking at the associations in the overall population, insulin use was strongly associated with SCA risk [hazard ratio, 2.38] and perhaps this was an indicator of severity of type 2 diabetes,” remarked Mr. Harms. “Also, unsurprisingly, a history of arrhythmia [HR, 1.68] and, more surprisingly, prokinetic drug use [HR, 1.66; 95% confidence interval, 1.20-2.31], specifically those known for QTc-prolongation, were associated with SCA.”
Among people who had experienced an SCA and who did not have a history of CVD (337 case patients/2,023 control patients), QTc-prolonging antipsychotic medication use was associated with SCA at an HR of 2.87, and antibiotic medication use was associated with SCA at an HR of 1.66. A low fasting glucose level (< 4.5 mmol/mol) was associated with SCA at an HR of 2.5; severely high systolic blood pressure (> 180 mm Hg) was associated with SCA at an HR of 2.21; low HDL cholesterol level, with an HR of 1.35; and high LDL cholesterol level (> 2.6 mmol/L), with an HR of 1.64.
Among people with a history of CVD (352 case patients/1,207 control patients), associations between albuminuria and SCA were moderate (HR, 1.54) and severe (HR, 1.55); heart failure was associated with SCA at an HR of 1.85 (95% CI, 1.50-2.29).
Comoderator Dr. Fejfarova added that, in addition to the findings from Dr. Harms’ study, other research presented in the same session highlighted the importance of checking patients for the presence of arrhythmias that could lead to the development of atrioventricular blocks, sinus node diseases, and SCA.
Mr. Harms and Dr. Fejfarova have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
HAMBURG, Germany – , shows the first such analysis of real-world, primary care data.
People with type 2 diabetes who do not have a history of CVD have almost three times the risk of SCA if they take antipsychotic medications and nearly double the risk if they take certain antibiotics that prolong the QT interval, notably, macrolides and fluoroquinolones.
“These data show that commonly prescribed drugs - antipsychotic medications, used by about 3% of people with type 2 diabetes, and antibiotics, taken by 5% to 10%, convey an increased risk of sudden cardiac arrest in those without a history of cardiovascular disease,” said Peter Harms, MSc, who presented the study at the annual meeting of the European Association for the Study of Diabetes. Another drug associated with an increase in SCA among patients with diabetes was domperidone, an antinausea medication.
“Perhaps these drugs could be avoided in some cases, and GPs should be more aware of the possible consequences of their use,” he added. “If the patient has type 2 diabetes, then maybe it’s better to avoid some of these medications and try and cope without them, or at least find an alternative antibiotic.”
Mr. Harms, an epidemiologist from Amsterdam University Medical Centers, highlighted that their study was unique because the investigators drew upon primary care data. “These data are extensive, and we find a lot of associations which are very real.”
SCA is associated with 50% of all cardiac deaths and accounts for 20% of all mortality in high-income countries. Of those people who experience SCA, 80% of cases prove fatal.
“As the name suggests, it is difficult to predict because it is sudden, especially in people without a cardiovascular disease history,” Mr. Harms pointed out in an interview with this news organization. He highlighted that “around half of those who experience SCA, often between the ages of 40 and 60 years, have never seen a cardiologist, but many do have type 2 diabetes.
“We need to better understand how to recognize people at risk of SCA, know who to watch and how to prevent these events,” he emphasized.
Vladimira Fejfarova, MD, comoderated the session and commented on the study. “From the clinical point of view, it’s necessary to evaluate risk factors that can contribute to sudden cardiac arrest.”
Overall, the researchers found that, among people with type 2 diabetes who do not have a history of CVD, hypoglycemia, severe hypertension, dyslipidemia, and use of QTc-prolonging medications are associated with SCA risk. Among people with type 2 diabetes and CVD, albuminuria and heart failure are associated with SCA risk.
Dr. Fejfarova added: “With type 2 diabetes and also type 1, we need to look more at adverse events, especially when treating infections with macrolides, but also mycotic infections, because antimycotic drugs are known to influence QT intervals that could contribute to sudden cardiac arrest.
“We need to be more cautious with prescribing certain antibiotics that have these side effects in our patients with diabetes,” asserted Dr. Fejfarova, from the Diabetes Centre, Institute for Clinical and Experimental Medicine, Prague.
Type 2 diabetes doubles the risk of SCA
The researcher decided to investigate the population of people with type 2 diabetes because their risk of SCD is around twice that of those without type 2 diabetes. Because these patients have relatively frequent checkups with general practitioners, Mr. Harms turned to primary care databases that contained comprehensive and relatively routine information on risk indicators.
Longitudinal associations between clinical characteristics of 3,919 patients with type 2 diabetes – both those with and those without a history of CVD – and SCA (a total of 689 patients) were determined.
Cases were found in the AmsteRdam REsuscitation STtudies (ARREST) registry of out-of-hospital resuscitation attempts by emergency medical services in the Dutch region of Noord-Holland from 2010 to 2019. Case patients were matched with up to five control patients. The control group comprised people with type 2 diabetes who had not experienced an SCA. Control patients were sourced from the same primary care practices who were of similar age and sex. Clinical measurements, including blood pressure and blood glucose readings, medication use, and medical history for the 5 years leading up to an SCA, were obtained from general practice records. A multivariable analysis was performed, and results were stratified for people with and for those without a history of CVD.
Of particular interest were drugs that interfere with cardiac function, including some prokinetic, antibiotic, and antipsychotic medications. All of the drugs are known to be associated with a change in QTc prolongation. Examples include domperidone (QTc-prolonging prokinetic), macrolides and fluoroquinolones (QTc-prolonging antibiotics), and haloperidol (a QTc-prolonging antipsychotic).
Antibiotic and antipsychotic use might contribute to SCA in T2D
Case patients and control patients were similar in age, hemoglobin A1c level, and other characteristics with the exception that more patients with SCA had a history of CVD (40.0% vs. 29.4%).
“Looking at the associations in the overall population, insulin use was strongly associated with SCA risk [hazard ratio, 2.38] and perhaps this was an indicator of severity of type 2 diabetes,” remarked Mr. Harms. “Also, unsurprisingly, a history of arrhythmia [HR, 1.68] and, more surprisingly, prokinetic drug use [HR, 1.66; 95% confidence interval, 1.20-2.31], specifically those known for QTc-prolongation, were associated with SCA.”
Among people who had experienced an SCA and who did not have a history of CVD (337 case patients/2,023 control patients), QTc-prolonging antipsychotic medication use was associated with SCA at an HR of 2.87, and antibiotic medication use was associated with SCA at an HR of 1.66. A low fasting glucose level (< 4.5 mmol/mol) was associated with SCA at an HR of 2.5; severely high systolic blood pressure (> 180 mm Hg) was associated with SCA at an HR of 2.21; low HDL cholesterol level, with an HR of 1.35; and high LDL cholesterol level (> 2.6 mmol/L), with an HR of 1.64.
Among people with a history of CVD (352 case patients/1,207 control patients), associations between albuminuria and SCA were moderate (HR, 1.54) and severe (HR, 1.55); heart failure was associated with SCA at an HR of 1.85 (95% CI, 1.50-2.29).
Comoderator Dr. Fejfarova added that, in addition to the findings from Dr. Harms’ study, other research presented in the same session highlighted the importance of checking patients for the presence of arrhythmias that could lead to the development of atrioventricular blocks, sinus node diseases, and SCA.
Mr. Harms and Dr. Fejfarova have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
HAMBURG, Germany – , shows the first such analysis of real-world, primary care data.
People with type 2 diabetes who do not have a history of CVD have almost three times the risk of SCA if they take antipsychotic medications and nearly double the risk if they take certain antibiotics that prolong the QT interval, notably, macrolides and fluoroquinolones.
“These data show that commonly prescribed drugs - antipsychotic medications, used by about 3% of people with type 2 diabetes, and antibiotics, taken by 5% to 10%, convey an increased risk of sudden cardiac arrest in those without a history of cardiovascular disease,” said Peter Harms, MSc, who presented the study at the annual meeting of the European Association for the Study of Diabetes. Another drug associated with an increase in SCA among patients with diabetes was domperidone, an antinausea medication.
“Perhaps these drugs could be avoided in some cases, and GPs should be more aware of the possible consequences of their use,” he added. “If the patient has type 2 diabetes, then maybe it’s better to avoid some of these medications and try and cope without them, or at least find an alternative antibiotic.”
Mr. Harms, an epidemiologist from Amsterdam University Medical Centers, highlighted that their study was unique because the investigators drew upon primary care data. “These data are extensive, and we find a lot of associations which are very real.”
SCA is associated with 50% of all cardiac deaths and accounts for 20% of all mortality in high-income countries. Of those people who experience SCA, 80% of cases prove fatal.
“As the name suggests, it is difficult to predict because it is sudden, especially in people without a cardiovascular disease history,” Mr. Harms pointed out in an interview with this news organization. He highlighted that “around half of those who experience SCA, often between the ages of 40 and 60 years, have never seen a cardiologist, but many do have type 2 diabetes.
“We need to better understand how to recognize people at risk of SCA, know who to watch and how to prevent these events,” he emphasized.
Vladimira Fejfarova, MD, comoderated the session and commented on the study. “From the clinical point of view, it’s necessary to evaluate risk factors that can contribute to sudden cardiac arrest.”
Overall, the researchers found that, among people with type 2 diabetes who do not have a history of CVD, hypoglycemia, severe hypertension, dyslipidemia, and use of QTc-prolonging medications are associated with SCA risk. Among people with type 2 diabetes and CVD, albuminuria and heart failure are associated with SCA risk.
Dr. Fejfarova added: “With type 2 diabetes and also type 1, we need to look more at adverse events, especially when treating infections with macrolides, but also mycotic infections, because antimycotic drugs are known to influence QT intervals that could contribute to sudden cardiac arrest.
“We need to be more cautious with prescribing certain antibiotics that have these side effects in our patients with diabetes,” asserted Dr. Fejfarova, from the Diabetes Centre, Institute for Clinical and Experimental Medicine, Prague.
Type 2 diabetes doubles the risk of SCA
The researcher decided to investigate the population of people with type 2 diabetes because their risk of SCD is around twice that of those without type 2 diabetes. Because these patients have relatively frequent checkups with general practitioners, Mr. Harms turned to primary care databases that contained comprehensive and relatively routine information on risk indicators.
Longitudinal associations between clinical characteristics of 3,919 patients with type 2 diabetes – both those with and those without a history of CVD – and SCA (a total of 689 patients) were determined.
Cases were found in the AmsteRdam REsuscitation STtudies (ARREST) registry of out-of-hospital resuscitation attempts by emergency medical services in the Dutch region of Noord-Holland from 2010 to 2019. Case patients were matched with up to five control patients. The control group comprised people with type 2 diabetes who had not experienced an SCA. Control patients were sourced from the same primary care practices who were of similar age and sex. Clinical measurements, including blood pressure and blood glucose readings, medication use, and medical history for the 5 years leading up to an SCA, were obtained from general practice records. A multivariable analysis was performed, and results were stratified for people with and for those without a history of CVD.
Of particular interest were drugs that interfere with cardiac function, including some prokinetic, antibiotic, and antipsychotic medications. All of the drugs are known to be associated with a change in QTc prolongation. Examples include domperidone (QTc-prolonging prokinetic), macrolides and fluoroquinolones (QTc-prolonging antibiotics), and haloperidol (a QTc-prolonging antipsychotic).
Antibiotic and antipsychotic use might contribute to SCA in T2D
Case patients and control patients were similar in age, hemoglobin A1c level, and other characteristics with the exception that more patients with SCA had a history of CVD (40.0% vs. 29.4%).
“Looking at the associations in the overall population, insulin use was strongly associated with SCA risk [hazard ratio, 2.38] and perhaps this was an indicator of severity of type 2 diabetes,” remarked Mr. Harms. “Also, unsurprisingly, a history of arrhythmia [HR, 1.68] and, more surprisingly, prokinetic drug use [HR, 1.66; 95% confidence interval, 1.20-2.31], specifically those known for QTc-prolongation, were associated with SCA.”
Among people who had experienced an SCA and who did not have a history of CVD (337 case patients/2,023 control patients), QTc-prolonging antipsychotic medication use was associated with SCA at an HR of 2.87, and antibiotic medication use was associated with SCA at an HR of 1.66. A low fasting glucose level (< 4.5 mmol/mol) was associated with SCA at an HR of 2.5; severely high systolic blood pressure (> 180 mm Hg) was associated with SCA at an HR of 2.21; low HDL cholesterol level, with an HR of 1.35; and high LDL cholesterol level (> 2.6 mmol/L), with an HR of 1.64.
Among people with a history of CVD (352 case patients/1,207 control patients), associations between albuminuria and SCA were moderate (HR, 1.54) and severe (HR, 1.55); heart failure was associated with SCA at an HR of 1.85 (95% CI, 1.50-2.29).
Comoderator Dr. Fejfarova added that, in addition to the findings from Dr. Harms’ study, other research presented in the same session highlighted the importance of checking patients for the presence of arrhythmias that could lead to the development of atrioventricular blocks, sinus node diseases, and SCA.
Mr. Harms and Dr. Fejfarova have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
AT EASD 2023
Higher fracture risk not seen with SGLT2 inhibitors
VANCOUVER – In patients with type 2 diabetes, sodium-glucose cotransporter 2 (SGLT2) inhibitors as an adjunct to metformin were not associated with an increase in fracture risk, according to a new real-world study.
There have been some reports of an increase in fracture risk associated with SGLT2 inhibitors, and it was observed in the phase 3 CANVAS trial of canagliflozin (Invokana), which led to a Food and Drug Administration warning of fracture risks associated with canagliflozin use. Some ensuing studies did not show an increased risk, but these studies were generally less than a year in duration and may have missed longer-term risk, according to Veerle van Hulten, MSc.
“Fracture risk is something that takes a long time to develop, so we wanted to have a longer follow-up. We looked into the CPRD [Clinical Practice Research Datalink], which is a beautiful database containing real-world data from primary care practices,” said Ms. van Hulten, a PhD student at Maastricht (the Netherlands) University, who presented the study at the annual meeting of the American Society for Bone and Mineral Research.
Ms. van Hulten and colleagues compared SGLT2 inhibitors with dipeptidyl peptidase–4 (DPP-4) inhibitors because the latter are used in similar populations and have been shown to have no effect on fracture risk.
“What we found is that SGLT2 inhibitors are not associated with an increased fracture risk. Even with a duration of use of over 811 days, we did not observe an increased hazard ratio for fractures when compared DPP-4 inhibitor users,” Ms. van Hulten said.
SGLT2 inhibitors reduce blood sugar by increasing elimination of sugar in the urine. They also increase phosphate, reduce calcium, and increase parathyroid hormone, which could in turn negatively affect bone turnover, according to Ms. van Hulten.
In the new study, conducted between January 2013 and June 2020, the researchers used propensity score matching to compare adult patients, including 13,807 who were prescribed SGLT2 inhibitors and 28,524 who were prescribed DPP-4 inhibitors for the first time. They matched patients based on demographics, comorbidities, comedication, and lifestyle factors.
There was no association between SGLT2 inhibitor use and overall fracture risk or major osteoporotic, hip, vertebral, humerus, radius, or ulna fractures. There was no difference in risk for any duration of use, even with the longest duration of use of 811 days (adjusted hazard ratio, 1.0). There were no differences among specific SGLT2 inhibitors, including canagliflozin (aHR, 1.12; 95% confidence interval, 0.73-1.72). Analyses by sex and age also revealed no statistically significant differences between the two drug classes.
During the Q&A session after the presentation, Sarah Berry, MD, MPH, an associate professor of medicine at Harvard Medical School and a clinical researcher at the Marcus Institute for Aging Research, both in Boston, noted the trend toward an increase in fracture risk in the first 90 days. “It looked like there was something going on in the first 90 days, and then after that the results were much closer to the null. I would put out maybe another potential mechanism whereby the SGLT2 inhibitors might cause fracture, and that’s falls. They cause polyuria, and any drug you give that causes women to rush to the bathroom may well cause fractures, particularly in the short term,” Dr. Berry said.
Ms. van Hulten agreed, and also brought up that the drugs can cause osmotic diuresis. That can lead to hypovolemia, the symptoms of which include weakness, fatigue, and dizziness. “And increased falls, of course, increases fracture risk. We do not expect anything to happen to bone metabolism in the first 90 days. I think we can agree that there would be more time needed to alter the bone enough to increase fracture risk, so we expect that this trend toward an increased risk might be attributable to that increased fall risk that might occur with SGLT2 inhibitor use,” she said.
It’s possible that such a mechanism explains increased fracture risk seen in some earlier short-term studies, she added.
Overall, Ms. van Hulten said that the results should provide some confidence in SGLT2 inhibitors, though more work needs to be done. “I think we provide reassurance that SGLT2 inhibitors are safe to use. However, we still only have a median follow-up of 1.6 years. It’s not as long as we maybe would like, but it’s the best we can do with the data available, since the SGLT2 inhibitors have only been used since 2013. So maybe it’s best to prescribe it and keep [fall risk] in mind and look into the effects later on again, but it seems to be safe to use.”
The study received funding from the Novo Nordisk Foundation. Ms. van Hulten and Dr. Berry reported no relevant financial relationships.
A version of this article appeared on Medscape.com.
VANCOUVER – In patients with type 2 diabetes, sodium-glucose cotransporter 2 (SGLT2) inhibitors as an adjunct to metformin were not associated with an increase in fracture risk, according to a new real-world study.
There have been some reports of an increase in fracture risk associated with SGLT2 inhibitors, and it was observed in the phase 3 CANVAS trial of canagliflozin (Invokana), which led to a Food and Drug Administration warning of fracture risks associated with canagliflozin use. Some ensuing studies did not show an increased risk, but these studies were generally less than a year in duration and may have missed longer-term risk, according to Veerle van Hulten, MSc.
“Fracture risk is something that takes a long time to develop, so we wanted to have a longer follow-up. We looked into the CPRD [Clinical Practice Research Datalink], which is a beautiful database containing real-world data from primary care practices,” said Ms. van Hulten, a PhD student at Maastricht (the Netherlands) University, who presented the study at the annual meeting of the American Society for Bone and Mineral Research.
Ms. van Hulten and colleagues compared SGLT2 inhibitors with dipeptidyl peptidase–4 (DPP-4) inhibitors because the latter are used in similar populations and have been shown to have no effect on fracture risk.
“What we found is that SGLT2 inhibitors are not associated with an increased fracture risk. Even with a duration of use of over 811 days, we did not observe an increased hazard ratio for fractures when compared DPP-4 inhibitor users,” Ms. van Hulten said.
SGLT2 inhibitors reduce blood sugar by increasing elimination of sugar in the urine. They also increase phosphate, reduce calcium, and increase parathyroid hormone, which could in turn negatively affect bone turnover, according to Ms. van Hulten.
In the new study, conducted between January 2013 and June 2020, the researchers used propensity score matching to compare adult patients, including 13,807 who were prescribed SGLT2 inhibitors and 28,524 who were prescribed DPP-4 inhibitors for the first time. They matched patients based on demographics, comorbidities, comedication, and lifestyle factors.
There was no association between SGLT2 inhibitor use and overall fracture risk or major osteoporotic, hip, vertebral, humerus, radius, or ulna fractures. There was no difference in risk for any duration of use, even with the longest duration of use of 811 days (adjusted hazard ratio, 1.0). There were no differences among specific SGLT2 inhibitors, including canagliflozin (aHR, 1.12; 95% confidence interval, 0.73-1.72). Analyses by sex and age also revealed no statistically significant differences between the two drug classes.
During the Q&A session after the presentation, Sarah Berry, MD, MPH, an associate professor of medicine at Harvard Medical School and a clinical researcher at the Marcus Institute for Aging Research, both in Boston, noted the trend toward an increase in fracture risk in the first 90 days. “It looked like there was something going on in the first 90 days, and then after that the results were much closer to the null. I would put out maybe another potential mechanism whereby the SGLT2 inhibitors might cause fracture, and that’s falls. They cause polyuria, and any drug you give that causes women to rush to the bathroom may well cause fractures, particularly in the short term,” Dr. Berry said.
Ms. van Hulten agreed, and also brought up that the drugs can cause osmotic diuresis. That can lead to hypovolemia, the symptoms of which include weakness, fatigue, and dizziness. “And increased falls, of course, increases fracture risk. We do not expect anything to happen to bone metabolism in the first 90 days. I think we can agree that there would be more time needed to alter the bone enough to increase fracture risk, so we expect that this trend toward an increased risk might be attributable to that increased fall risk that might occur with SGLT2 inhibitor use,” she said.
It’s possible that such a mechanism explains increased fracture risk seen in some earlier short-term studies, she added.
Overall, Ms. van Hulten said that the results should provide some confidence in SGLT2 inhibitors, though more work needs to be done. “I think we provide reassurance that SGLT2 inhibitors are safe to use. However, we still only have a median follow-up of 1.6 years. It’s not as long as we maybe would like, but it’s the best we can do with the data available, since the SGLT2 inhibitors have only been used since 2013. So maybe it’s best to prescribe it and keep [fall risk] in mind and look into the effects later on again, but it seems to be safe to use.”
The study received funding from the Novo Nordisk Foundation. Ms. van Hulten and Dr. Berry reported no relevant financial relationships.
A version of this article appeared on Medscape.com.
VANCOUVER – In patients with type 2 diabetes, sodium-glucose cotransporter 2 (SGLT2) inhibitors as an adjunct to metformin were not associated with an increase in fracture risk, according to a new real-world study.
There have been some reports of an increase in fracture risk associated with SGLT2 inhibitors, and it was observed in the phase 3 CANVAS trial of canagliflozin (Invokana), which led to a Food and Drug Administration warning of fracture risks associated with canagliflozin use. Some ensuing studies did not show an increased risk, but these studies were generally less than a year in duration and may have missed longer-term risk, according to Veerle van Hulten, MSc.
“Fracture risk is something that takes a long time to develop, so we wanted to have a longer follow-up. We looked into the CPRD [Clinical Practice Research Datalink], which is a beautiful database containing real-world data from primary care practices,” said Ms. van Hulten, a PhD student at Maastricht (the Netherlands) University, who presented the study at the annual meeting of the American Society for Bone and Mineral Research.
Ms. van Hulten and colleagues compared SGLT2 inhibitors with dipeptidyl peptidase–4 (DPP-4) inhibitors because the latter are used in similar populations and have been shown to have no effect on fracture risk.
“What we found is that SGLT2 inhibitors are not associated with an increased fracture risk. Even with a duration of use of over 811 days, we did not observe an increased hazard ratio for fractures when compared DPP-4 inhibitor users,” Ms. van Hulten said.
SGLT2 inhibitors reduce blood sugar by increasing elimination of sugar in the urine. They also increase phosphate, reduce calcium, and increase parathyroid hormone, which could in turn negatively affect bone turnover, according to Ms. van Hulten.
In the new study, conducted between January 2013 and June 2020, the researchers used propensity score matching to compare adult patients, including 13,807 who were prescribed SGLT2 inhibitors and 28,524 who were prescribed DPP-4 inhibitors for the first time. They matched patients based on demographics, comorbidities, comedication, and lifestyle factors.
There was no association between SGLT2 inhibitor use and overall fracture risk or major osteoporotic, hip, vertebral, humerus, radius, or ulna fractures. There was no difference in risk for any duration of use, even with the longest duration of use of 811 days (adjusted hazard ratio, 1.0). There were no differences among specific SGLT2 inhibitors, including canagliflozin (aHR, 1.12; 95% confidence interval, 0.73-1.72). Analyses by sex and age also revealed no statistically significant differences between the two drug classes.
During the Q&A session after the presentation, Sarah Berry, MD, MPH, an associate professor of medicine at Harvard Medical School and a clinical researcher at the Marcus Institute for Aging Research, both in Boston, noted the trend toward an increase in fracture risk in the first 90 days. “It looked like there was something going on in the first 90 days, and then after that the results were much closer to the null. I would put out maybe another potential mechanism whereby the SGLT2 inhibitors might cause fracture, and that’s falls. They cause polyuria, and any drug you give that causes women to rush to the bathroom may well cause fractures, particularly in the short term,” Dr. Berry said.
Ms. van Hulten agreed, and also brought up that the drugs can cause osmotic diuresis. That can lead to hypovolemia, the symptoms of which include weakness, fatigue, and dizziness. “And increased falls, of course, increases fracture risk. We do not expect anything to happen to bone metabolism in the first 90 days. I think we can agree that there would be more time needed to alter the bone enough to increase fracture risk, so we expect that this trend toward an increased risk might be attributable to that increased fall risk that might occur with SGLT2 inhibitor use,” she said.
It’s possible that such a mechanism explains increased fracture risk seen in some earlier short-term studies, she added.
Overall, Ms. van Hulten said that the results should provide some confidence in SGLT2 inhibitors, though more work needs to be done. “I think we provide reassurance that SGLT2 inhibitors are safe to use. However, we still only have a median follow-up of 1.6 years. It’s not as long as we maybe would like, but it’s the best we can do with the data available, since the SGLT2 inhibitors have only been used since 2013. So maybe it’s best to prescribe it and keep [fall risk] in mind and look into the effects later on again, but it seems to be safe to use.”
The study received funding from the Novo Nordisk Foundation. Ms. van Hulten and Dr. Berry reported no relevant financial relationships.
A version of this article appeared on Medscape.com.
AT ASBMR 2023
A focus on women with diabetes and their offspring
In 2021, diabetes and related complications was the 8th leading cause of death in the United States.1 As of 2022, more than 11% of the U.S. population had diabetes and 38% of the adult U.S. population had prediabetes.2 Diabetes is the most expensive chronic condition in the United States, where $1 of every $4 in health care costs is spent on care.3
Where this is most concerning is diabetes in pregnancy. While childbirth rates in the United States have decreased since the 2007 high of 4.32 million births4 to 3.66 million in 2021,5 the incidence of diabetes in pregnancy – both pregestational and gestational – has increased. The rate of pregestational diabetes in 2021 was 10.9 per 1,000 births, a 27% increase from 2016 (8.6 per 1,000).6 The percentage of those giving birth who also were diagnosed with gestational diabetes mellitus (GDM) was 8.3% in 2021, up from 6.0% in 2016.7
Adverse outcomes for an infant born to a mother with diabetes include a higher risk of obesity and diabetes as adults, potentially leading to a forward-feeding cycle.
We and our colleagues established the Diabetes in Pregnancy Study Group of North America in 1997 because we had witnessed too frequently the devastating diabetes-induced pregnancy complications in our patients. The mission we set forth was to provide a forum for dialogue among maternal-fetal medicine subspecialists. The three main goals we set forth to support this mission were to provide a catalyst for research, contribute to the creation and refinement of medical policies, and influence professional practices in diabetes in pregnancy.8
In the last quarter century, DPSG-NA, through its annual and biennial meetings, has brought together several hundred practitioners that include physicians, nurses, statisticians, researchers, nutritionists, and allied health professionals, among others. As a group, it has improved the detection and management of diabetes in pregnant women and their offspring through knowledge sharing and influencing policies on GDM screening, diagnosis, management, and treatment. Our members have shown that preconceptional counseling for women with diabetes can significantly reduce congenital malformation and perinatal mortality compared with those women with pregestational diabetes who receive no counseling.9,10
We have addressed a wide variety of topics including the paucity of data in determining the timing of delivery for women with diabetes and the Institute of Medicine/National Academy of Medicine recommendations of gestational weight gain and risks of not adhering to them. We have learned about new scientific discoveries that reveal underlying mechanisms to diabetes-related birth defects and potential therapeutic targets; and we have discussed the health literacy requirements, ethics, and opportunities for lifestyle intervention.11-16
But we need to do more.
Two risk factors are at play: Women continue to choose to have babies at later ages and their pregnancies continue to be complicated by the rising incidence of obesity (see Figure 1 and Figure 2). 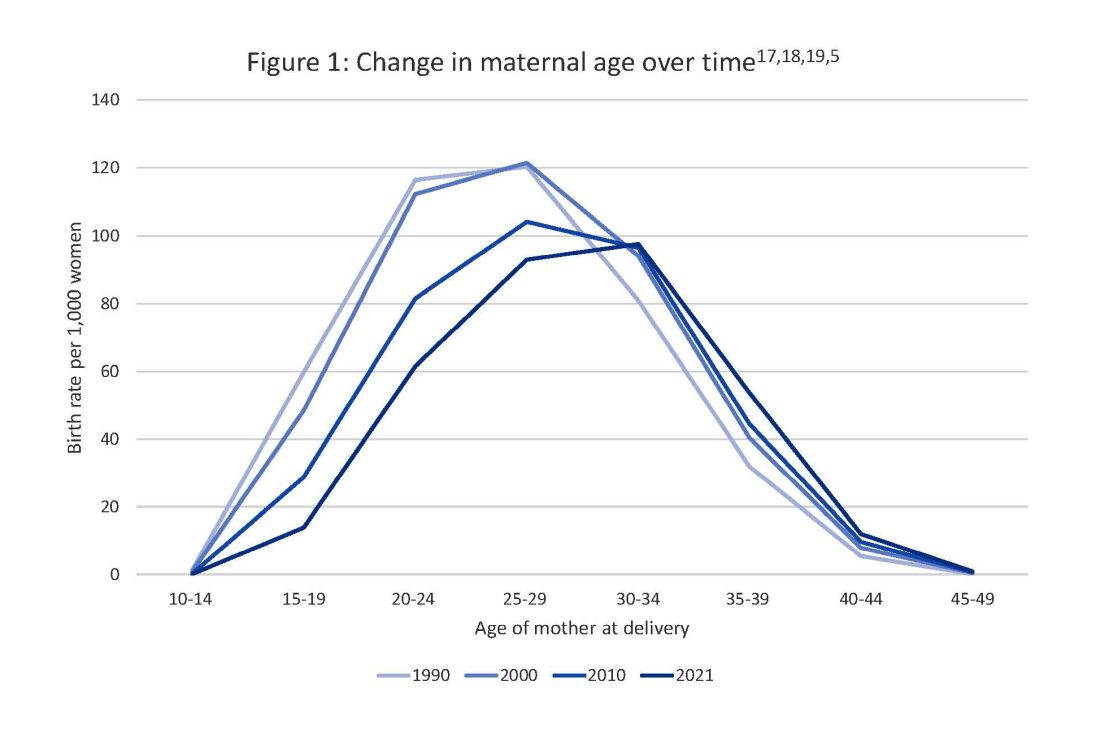
The global obesity epidemic has become a significant concern for all aspects of health and particularly for diabetes in pregnancy. 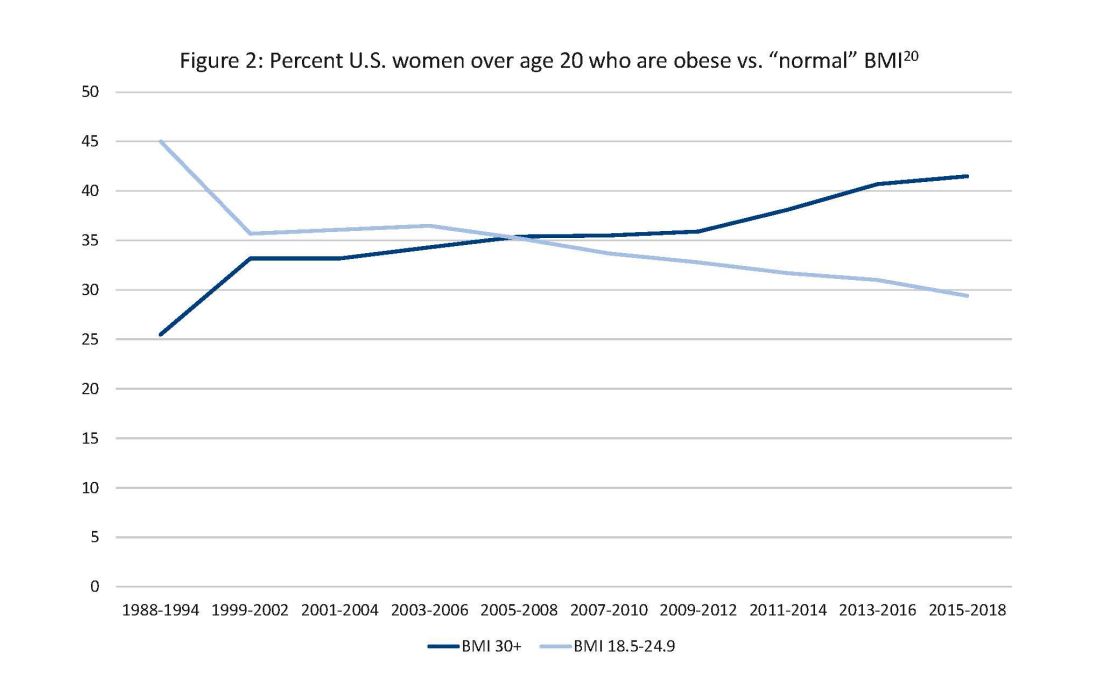
In 1990, 24.9% of women in the United States were obese; in 2010, 35.8%; and now more than 41%. Some experts project that by 2030 more than 80% of women in the United States will be overweight or obese.21
If we are to stop this cycle of diabetes begets more diabetes, now more than ever we need to come together and accelerate the research and education around the diabetes in pregnancy. Join us at this year’s DPSG-NA meeting Oct. 26-28 to take part in the knowledge sharing, discussions, and planning. More information can be found online at https://events.dpsg-na.com/home.
Dr. Miodovnik is adjunct professor of obstetrics, gynecology, and reproductive sciences at University of Maryland School of Medicine. Dr. Reece is professor of obstetrics, gynecology, and reproductive sciences and senior scientist at the Center for Birth Defects Research at University of Maryland School of Medicine.
References
1. Xu J et al. Mortality in the United States, 2021. NCHS Data Brief. 2022 Dec;(456):1-8. PMID: 36598387.
2. Centers for Disease Control and Prevention, diabetes data and statistics.
3. American Diabetes Association. The Cost of Diabetes.
4. Martin JA et al. Births: Final data for 2007. Natl Vital Stat Rep. 2010 Aug 9;58(24):1-85. PMID: 21254725.
5. Osterman MJK et al. Births: Final data for 2021. Natl Vital Stat Rep. 2023 Jan;72(1):1-53. PMID: 36723449.
6. Gregory ECW and Ely DM. Trends and characteristics in prepregnancy diabetes: United States, 2016-2021. Natl Vital Stat Rep. 2023 May;72(6):1-13. PMID: 37256333.
7. QuickStats: Percentage of mothers with gestational diabetes, by maternal age – National Vital Statistics System, United States, 2016 and 2021. MMWR Morb Mortal Wkly Rep. 2023 Jan 6;72(1):16. doi: 10.15585/mmwr.mm7201a4.
8. Langer O et al. The Diabetes in Pregnancy Study Group of North America – Introduction and summary statement. Prenat Neonat Med. 1998;3(6):514-6.
9. Willhoite MB et al. The impact of preconception counseling on pregnancy outcomes. The experience of the Maine Diabetes in Pregnancy Program. Diabetes Care. 1993 Feb;16(2):450-5. doi: 10.2337/diacare.16.2.450.
10. McElvy SS et al. A focused preconceptional and early pregnancy program in women with type 1 diabetes reduces perinatal mortality and malformation rates to general population levels. J Matern Fetal Med. 2000 Jan-Feb;9(1):14-20. doi: 10.1002/(SICI)1520-6661(200001/02)9:1<14::AID-MFM5>3.0.CO;2-K.
11. Rosen JA et al. The history and contributions of the Diabetes in Pregnancy Study Group of North America (1997-2015). Am J Perinatol. 2016 Nov;33(13):1223-6. doi: 10.1055/s-0036-1585082.
12. Driggers RW and Baschat A. The 12th meeting of the Diabetes in Pregnancy Study Group of North America (DPSG-NA): Introduction and overview. J Matern Fetal Neonatal Med. 2012 Jan;25(1):3-4. doi: 10.3109/14767058.2012.626917.
13. Langer O et al. The proceedings of the Diabetes in Pregnancy Study Group of North America 2009 conference. J Matern Fetal Neonatal Med. 2010 Mar;23(3):196-8. doi: 10.3109/14767050903550634.
14. Reece EA et al. A consensus report of the Diabetes in Pregnancy Study Group of North America Conference, Little Rock, Ark., May 2002. J Matern Fetal Neonatal Med. 2002 Dec;12(6):362-4. doi: 10.1080/jmf.12.6.362.364.
15. Reece EA and Maulik D. A consensus conference of the Diabetes in Pregnancy Study Group of North America. J Matern Fetal Neonatal Med. 2002 Dec;12(6):361. doi: 10.1080/jmf.12.6.361.361.
16. Gabbe SG. Summation of the second meeting of the Diabetes in Pregnancy Study Group of North America (DPSG-NA). J Matern Fetal Med. 2000 Jan-Feb;9(1):3-9.
17. Vital Statistics of the United States 1990: Volume I – Natality.
18. Martin JA et al. Births: final data for 2000. Natl Vital Stat Rep. 2002 Feb 12;50(5):1-101. PMID: 11876093.
19. Martin JA et al. Births: final data for 2010. Natl Vital Stat Rep. 2012 Aug 28;61(1):1-72. PMID: 24974589.
20. CDC Website. Normal weight, overweight, and obesity among adults aged 20 and over, by selected characteristics: United States.
21. Wang Y et al. Has the prevalence of overweight, obesity, and central obesity levelled off in the United States? Trends, patterns, disparities, and future projections for the obesity epidemic. Int J Epidemiol. 2020 Jun 1;49(3):810-23. doi: 10.1093/ije/dyz273.
In 2021, diabetes and related complications was the 8th leading cause of death in the United States.1 As of 2022, more than 11% of the U.S. population had diabetes and 38% of the adult U.S. population had prediabetes.2 Diabetes is the most expensive chronic condition in the United States, where $1 of every $4 in health care costs is spent on care.3
Where this is most concerning is diabetes in pregnancy. While childbirth rates in the United States have decreased since the 2007 high of 4.32 million births4 to 3.66 million in 2021,5 the incidence of diabetes in pregnancy – both pregestational and gestational – has increased. The rate of pregestational diabetes in 2021 was 10.9 per 1,000 births, a 27% increase from 2016 (8.6 per 1,000).6 The percentage of those giving birth who also were diagnosed with gestational diabetes mellitus (GDM) was 8.3% in 2021, up from 6.0% in 2016.7
Adverse outcomes for an infant born to a mother with diabetes include a higher risk of obesity and diabetes as adults, potentially leading to a forward-feeding cycle.
We and our colleagues established the Diabetes in Pregnancy Study Group of North America in 1997 because we had witnessed too frequently the devastating diabetes-induced pregnancy complications in our patients. The mission we set forth was to provide a forum for dialogue among maternal-fetal medicine subspecialists. The three main goals we set forth to support this mission were to provide a catalyst for research, contribute to the creation and refinement of medical policies, and influence professional practices in diabetes in pregnancy.8
In the last quarter century, DPSG-NA, through its annual and biennial meetings, has brought together several hundred practitioners that include physicians, nurses, statisticians, researchers, nutritionists, and allied health professionals, among others. As a group, it has improved the detection and management of diabetes in pregnant women and their offspring through knowledge sharing and influencing policies on GDM screening, diagnosis, management, and treatment. Our members have shown that preconceptional counseling for women with diabetes can significantly reduce congenital malformation and perinatal mortality compared with those women with pregestational diabetes who receive no counseling.9,10
We have addressed a wide variety of topics including the paucity of data in determining the timing of delivery for women with diabetes and the Institute of Medicine/National Academy of Medicine recommendations of gestational weight gain and risks of not adhering to them. We have learned about new scientific discoveries that reveal underlying mechanisms to diabetes-related birth defects and potential therapeutic targets; and we have discussed the health literacy requirements, ethics, and opportunities for lifestyle intervention.11-16
But we need to do more.
Two risk factors are at play: Women continue to choose to have babies at later ages and their pregnancies continue to be complicated by the rising incidence of obesity (see Figure 1 and Figure 2). 
The global obesity epidemic has become a significant concern for all aspects of health and particularly for diabetes in pregnancy. 
In 1990, 24.9% of women in the United States were obese; in 2010, 35.8%; and now more than 41%. Some experts project that by 2030 more than 80% of women in the United States will be overweight or obese.21
If we are to stop this cycle of diabetes begets more diabetes, now more than ever we need to come together and accelerate the research and education around the diabetes in pregnancy. Join us at this year’s DPSG-NA meeting Oct. 26-28 to take part in the knowledge sharing, discussions, and planning. More information can be found online at https://events.dpsg-na.com/home.
Dr. Miodovnik is adjunct professor of obstetrics, gynecology, and reproductive sciences at University of Maryland School of Medicine. Dr. Reece is professor of obstetrics, gynecology, and reproductive sciences and senior scientist at the Center for Birth Defects Research at University of Maryland School of Medicine.
References
1. Xu J et al. Mortality in the United States, 2021. NCHS Data Brief. 2022 Dec;(456):1-8. PMID: 36598387.
2. Centers for Disease Control and Prevention, diabetes data and statistics.
3. American Diabetes Association. The Cost of Diabetes.
4. Martin JA et al. Births: Final data for 2007. Natl Vital Stat Rep. 2010 Aug 9;58(24):1-85. PMID: 21254725.
5. Osterman MJK et al. Births: Final data for 2021. Natl Vital Stat Rep. 2023 Jan;72(1):1-53. PMID: 36723449.
6. Gregory ECW and Ely DM. Trends and characteristics in prepregnancy diabetes: United States, 2016-2021. Natl Vital Stat Rep. 2023 May;72(6):1-13. PMID: 37256333.
7. QuickStats: Percentage of mothers with gestational diabetes, by maternal age – National Vital Statistics System, United States, 2016 and 2021. MMWR Morb Mortal Wkly Rep. 2023 Jan 6;72(1):16. doi: 10.15585/mmwr.mm7201a4.
8. Langer O et al. The Diabetes in Pregnancy Study Group of North America – Introduction and summary statement. Prenat Neonat Med. 1998;3(6):514-6.
9. Willhoite MB et al. The impact of preconception counseling on pregnancy outcomes. The experience of the Maine Diabetes in Pregnancy Program. Diabetes Care. 1993 Feb;16(2):450-5. doi: 10.2337/diacare.16.2.450.
10. McElvy SS et al. A focused preconceptional and early pregnancy program in women with type 1 diabetes reduces perinatal mortality and malformation rates to general population levels. J Matern Fetal Med. 2000 Jan-Feb;9(1):14-20. doi: 10.1002/(SICI)1520-6661(200001/02)9:1<14::AID-MFM5>3.0.CO;2-K.
11. Rosen JA et al. The history and contributions of the Diabetes in Pregnancy Study Group of North America (1997-2015). Am J Perinatol. 2016 Nov;33(13):1223-6. doi: 10.1055/s-0036-1585082.
12. Driggers RW and Baschat A. The 12th meeting of the Diabetes in Pregnancy Study Group of North America (DPSG-NA): Introduction and overview. J Matern Fetal Neonatal Med. 2012 Jan;25(1):3-4. doi: 10.3109/14767058.2012.626917.
13. Langer O et al. The proceedings of the Diabetes in Pregnancy Study Group of North America 2009 conference. J Matern Fetal Neonatal Med. 2010 Mar;23(3):196-8. doi: 10.3109/14767050903550634.
14. Reece EA et al. A consensus report of the Diabetes in Pregnancy Study Group of North America Conference, Little Rock, Ark., May 2002. J Matern Fetal Neonatal Med. 2002 Dec;12(6):362-4. doi: 10.1080/jmf.12.6.362.364.
15. Reece EA and Maulik D. A consensus conference of the Diabetes in Pregnancy Study Group of North America. J Matern Fetal Neonatal Med. 2002 Dec;12(6):361. doi: 10.1080/jmf.12.6.361.361.
16. Gabbe SG. Summation of the second meeting of the Diabetes in Pregnancy Study Group of North America (DPSG-NA). J Matern Fetal Med. 2000 Jan-Feb;9(1):3-9.
17. Vital Statistics of the United States 1990: Volume I – Natality.
18. Martin JA et al. Births: final data for 2000. Natl Vital Stat Rep. 2002 Feb 12;50(5):1-101. PMID: 11876093.
19. Martin JA et al. Births: final data for 2010. Natl Vital Stat Rep. 2012 Aug 28;61(1):1-72. PMID: 24974589.
20. CDC Website. Normal weight, overweight, and obesity among adults aged 20 and over, by selected characteristics: United States.
21. Wang Y et al. Has the prevalence of overweight, obesity, and central obesity levelled off in the United States? Trends, patterns, disparities, and future projections for the obesity epidemic. Int J Epidemiol. 2020 Jun 1;49(3):810-23. doi: 10.1093/ije/dyz273.
In 2021, diabetes and related complications was the 8th leading cause of death in the United States.1 As of 2022, more than 11% of the U.S. population had diabetes and 38% of the adult U.S. population had prediabetes.2 Diabetes is the most expensive chronic condition in the United States, where $1 of every $4 in health care costs is spent on care.3
Where this is most concerning is diabetes in pregnancy. While childbirth rates in the United States have decreased since the 2007 high of 4.32 million births4 to 3.66 million in 2021,5 the incidence of diabetes in pregnancy – both pregestational and gestational – has increased. The rate of pregestational diabetes in 2021 was 10.9 per 1,000 births, a 27% increase from 2016 (8.6 per 1,000).6 The percentage of those giving birth who also were diagnosed with gestational diabetes mellitus (GDM) was 8.3% in 2021, up from 6.0% in 2016.7
Adverse outcomes for an infant born to a mother with diabetes include a higher risk of obesity and diabetes as adults, potentially leading to a forward-feeding cycle.
We and our colleagues established the Diabetes in Pregnancy Study Group of North America in 1997 because we had witnessed too frequently the devastating diabetes-induced pregnancy complications in our patients. The mission we set forth was to provide a forum for dialogue among maternal-fetal medicine subspecialists. The three main goals we set forth to support this mission were to provide a catalyst for research, contribute to the creation and refinement of medical policies, and influence professional practices in diabetes in pregnancy.8
In the last quarter century, DPSG-NA, through its annual and biennial meetings, has brought together several hundred practitioners that include physicians, nurses, statisticians, researchers, nutritionists, and allied health professionals, among others. As a group, it has improved the detection and management of diabetes in pregnant women and their offspring through knowledge sharing and influencing policies on GDM screening, diagnosis, management, and treatment. Our members have shown that preconceptional counseling for women with diabetes can significantly reduce congenital malformation and perinatal mortality compared with those women with pregestational diabetes who receive no counseling.9,10
We have addressed a wide variety of topics including the paucity of data in determining the timing of delivery for women with diabetes and the Institute of Medicine/National Academy of Medicine recommendations of gestational weight gain and risks of not adhering to them. We have learned about new scientific discoveries that reveal underlying mechanisms to diabetes-related birth defects and potential therapeutic targets; and we have discussed the health literacy requirements, ethics, and opportunities for lifestyle intervention.11-16
But we need to do more.
Two risk factors are at play: Women continue to choose to have babies at later ages and their pregnancies continue to be complicated by the rising incidence of obesity (see Figure 1 and Figure 2). 
The global obesity epidemic has become a significant concern for all aspects of health and particularly for diabetes in pregnancy. 
In 1990, 24.9% of women in the United States were obese; in 2010, 35.8%; and now more than 41%. Some experts project that by 2030 more than 80% of women in the United States will be overweight or obese.21
If we are to stop this cycle of diabetes begets more diabetes, now more than ever we need to come together and accelerate the research and education around the diabetes in pregnancy. Join us at this year’s DPSG-NA meeting Oct. 26-28 to take part in the knowledge sharing, discussions, and planning. More information can be found online at https://events.dpsg-na.com/home.
Dr. Miodovnik is adjunct professor of obstetrics, gynecology, and reproductive sciences at University of Maryland School of Medicine. Dr. Reece is professor of obstetrics, gynecology, and reproductive sciences and senior scientist at the Center for Birth Defects Research at University of Maryland School of Medicine.
References
1. Xu J et al. Mortality in the United States, 2021. NCHS Data Brief. 2022 Dec;(456):1-8. PMID: 36598387.
2. Centers for Disease Control and Prevention, diabetes data and statistics.
3. American Diabetes Association. The Cost of Diabetes.
4. Martin JA et al. Births: Final data for 2007. Natl Vital Stat Rep. 2010 Aug 9;58(24):1-85. PMID: 21254725.
5. Osterman MJK et al. Births: Final data for 2021. Natl Vital Stat Rep. 2023 Jan;72(1):1-53. PMID: 36723449.
6. Gregory ECW and Ely DM. Trends and characteristics in prepregnancy diabetes: United States, 2016-2021. Natl Vital Stat Rep. 2023 May;72(6):1-13. PMID: 37256333.
7. QuickStats: Percentage of mothers with gestational diabetes, by maternal age – National Vital Statistics System, United States, 2016 and 2021. MMWR Morb Mortal Wkly Rep. 2023 Jan 6;72(1):16. doi: 10.15585/mmwr.mm7201a4.
8. Langer O et al. The Diabetes in Pregnancy Study Group of North America – Introduction and summary statement. Prenat Neonat Med. 1998;3(6):514-6.
9. Willhoite MB et al. The impact of preconception counseling on pregnancy outcomes. The experience of the Maine Diabetes in Pregnancy Program. Diabetes Care. 1993 Feb;16(2):450-5. doi: 10.2337/diacare.16.2.450.
10. McElvy SS et al. A focused preconceptional and early pregnancy program in women with type 1 diabetes reduces perinatal mortality and malformation rates to general population levels. J Matern Fetal Med. 2000 Jan-Feb;9(1):14-20. doi: 10.1002/(SICI)1520-6661(200001/02)9:1<14::AID-MFM5>3.0.CO;2-K.
11. Rosen JA et al. The history and contributions of the Diabetes in Pregnancy Study Group of North America (1997-2015). Am J Perinatol. 2016 Nov;33(13):1223-6. doi: 10.1055/s-0036-1585082.
12. Driggers RW and Baschat A. The 12th meeting of the Diabetes in Pregnancy Study Group of North America (DPSG-NA): Introduction and overview. J Matern Fetal Neonatal Med. 2012 Jan;25(1):3-4. doi: 10.3109/14767058.2012.626917.
13. Langer O et al. The proceedings of the Diabetes in Pregnancy Study Group of North America 2009 conference. J Matern Fetal Neonatal Med. 2010 Mar;23(3):196-8. doi: 10.3109/14767050903550634.
14. Reece EA et al. A consensus report of the Diabetes in Pregnancy Study Group of North America Conference, Little Rock, Ark., May 2002. J Matern Fetal Neonatal Med. 2002 Dec;12(6):362-4. doi: 10.1080/jmf.12.6.362.364.
15. Reece EA and Maulik D. A consensus conference of the Diabetes in Pregnancy Study Group of North America. J Matern Fetal Neonatal Med. 2002 Dec;12(6):361. doi: 10.1080/jmf.12.6.361.361.
16. Gabbe SG. Summation of the second meeting of the Diabetes in Pregnancy Study Group of North America (DPSG-NA). J Matern Fetal Med. 2000 Jan-Feb;9(1):3-9.
17. Vital Statistics of the United States 1990: Volume I – Natality.
18. Martin JA et al. Births: final data for 2000. Natl Vital Stat Rep. 2002 Feb 12;50(5):1-101. PMID: 11876093.
19. Martin JA et al. Births: final data for 2010. Natl Vital Stat Rep. 2012 Aug 28;61(1):1-72. PMID: 24974589.
20. CDC Website. Normal weight, overweight, and obesity among adults aged 20 and over, by selected characteristics: United States.
21. Wang Y et al. Has the prevalence of overweight, obesity, and central obesity levelled off in the United States? Trends, patterns, disparities, and future projections for the obesity epidemic. Int J Epidemiol. 2020 Jun 1;49(3):810-23. doi: 10.1093/ije/dyz273.
Antidepressants ‘don’t blunt’ semaglutide and weight loss
in a post hoc analysis of the Semaglutide Treatment Effect in People with Obesity (STEP) program.
Adverse events, including psychiatric events, were slightly more usual in the patients on antidepressants, Robert Kushner, MD, noted, in an oral session at the annual meeting of the Obesity Society.
“It is very common that patients who present for weight management are taking antidepressants for various reasons, including depression, anxiety, insomnia, or chronic pain,”Dr. Kushner, from Northwestern University in Chicago, said in an email. “We wanted to see if these participants responded differently to semaglutide, compared to those not on antidepressants.”
“We found that antidepressants do not blunt the effect of semaglutide for weight loss,” he said. “However, there is a slight increase in reported adverse effects.”
“Semaglutide 2.4 mg provides an effective treatment option for weight management, regardless of antidepressant use at baseline,” Dr. Kushner summarized. “Clinicians should be assured that we can use semaglutide in this population of patients.”
Jack Yanovski, MD, PhD, said this was a “great presentation,” noting that “it’s really important that we understand what goes on in patients with depression.”
“Of course, all these trials still had rules that prevent the folks with the most severe depressive symptoms or past suicidality to participate,” added Dr. Yanovski, chief of the Growth and Obesity Section, Eunice Kennedy Shriver National Institute of Child Health and Human Development, Rockville, Md. “We need specific trials to know exactly how well we do.”
Dr. Kushner agreed, but also noted that, ever since some earlier antidepressants were associated with risk for suicidal ideation and death, strict guidelines were put in place that exclude certain patients from participating in clinical trials.
Dr. Yanovski suggested that now that the drugs are approved, it would be possible to study this, and the information would be important for clinicians.
Dr. Kushner said he hopes that such studies are forthcoming. In the meantime, “data like this will add some support and understanding,” he suggested.
36,000 Patients with obesity, 500 on antidepressants
Many people living with obesity report taking antidepressants for depression, anxiety, chronic pain, obsessive-compulsive disorder, sleep disturbance, neuropathy, panic disorder, or posttraumatic stress disorder, Dr. Kushner noted.
However, some of these medications can cause weight gain, and little is known about treatment outcomes for people with obesity who are on antidepressants, since most weight-loss studies exclude people with active major depressive disorder.
The researchers analyzed data from 1,961 patients in STEP 1 and 807 patients in STEP 2 as well as 611 patients in STEP 3 and 304 patients in STEP 5 – 3,683 participants in total, of which 539 were on antidepressants at baseline.
The patients were randomly assigned to 2.4 mg semaglutide vs. placebo plus a lifestyle intervention (STEP 1, 2, and 5) or intensive behavioral therapy (STEP 3 only), for 68 weeks, except STEP 5, which was 104 weeks.
Patients were included if they were aged 18 or older with a body mass index ≥30 kg/m2, or ≥27 kg/m2 with more than one weight-related complication (STEP 1, 3, and 5) or BMI ≥27 kg/m2 with type 2 diabetes (STEP 2 only), and at least one self-reported unsuccessful effort to lose weight by diet.
They were excluded if they had active major depressive disorder within 2 years prior to screening (or other severe psychiatric disorders such as schizophrenia or bipolar disorder) or a Patient Health Questionnaire-9 score of 15 or higher (indicating moderately severe or severe depression), or suicide ideation (type 4 or 5 on the Columbia Suicide Severity Rating Scale) or suicide behavior, within 30 days of screening.
From baseline to week 68, patients on semaglutide (with/without baseline antidepressant use) had a significantly greater change in weight vs. patients on placebo (with/without baseline antidepressant use), respectively:
- STEP 1: –15.7% / –14.7% vs. –0.2% / –2.8%
- STEP 2: –10.7% / –9.5% vs. –3.3% / –3.4%
- STEP 3: –16.2% / –15.9% vs. –5.0% / –5.9%
- STEP 5: –19.0% / –14.1% vs. +1.6% / – 4.0%.
The proportion of reported adverse events was generally slightly greater in patients receiving semaglutide (with/without baseline antidepressant use) than those on placebo (with/without baseline antidepressant use), respectively:
- STEP 1: 97.7% vs 88.6% and 92.9% vs. 86%
- STEP 2: 97.6% vs 86.5% and 88.6% vs. 77.2%
- STEP 3: 97.6% vs 95.3% and 100% vs. 95.8%
- STEP 5: 100% vs 94.8% and 95.5% vs. 89.2%.
Gastrointestinal adverse events were more frequently reported in the semaglutide group and in patients on antidepressants at baseline. The proportion of patients with psychiatric adverse events was greater in participants on antidepressants at baseline. There were no differences in suicidal ideation/behavior in patients with/without antidepressant use at baseline.
The STEP trials were funded by Novo Nordisk. Dr. Kushner discloses that he served as a consultant for Novo Nordisk, WeightWatchers, Eli Lilly, and Pfizer, and received a research grant from Epitomee.
A version of this article appeared on Medscape.com.
in a post hoc analysis of the Semaglutide Treatment Effect in People with Obesity (STEP) program.
Adverse events, including psychiatric events, were slightly more usual in the patients on antidepressants, Robert Kushner, MD, noted, in an oral session at the annual meeting of the Obesity Society.
“It is very common that patients who present for weight management are taking antidepressants for various reasons, including depression, anxiety, insomnia, or chronic pain,”Dr. Kushner, from Northwestern University in Chicago, said in an email. “We wanted to see if these participants responded differently to semaglutide, compared to those not on antidepressants.”
“We found that antidepressants do not blunt the effect of semaglutide for weight loss,” he said. “However, there is a slight increase in reported adverse effects.”
“Semaglutide 2.4 mg provides an effective treatment option for weight management, regardless of antidepressant use at baseline,” Dr. Kushner summarized. “Clinicians should be assured that we can use semaglutide in this population of patients.”
Jack Yanovski, MD, PhD, said this was a “great presentation,” noting that “it’s really important that we understand what goes on in patients with depression.”
“Of course, all these trials still had rules that prevent the folks with the most severe depressive symptoms or past suicidality to participate,” added Dr. Yanovski, chief of the Growth and Obesity Section, Eunice Kennedy Shriver National Institute of Child Health and Human Development, Rockville, Md. “We need specific trials to know exactly how well we do.”
Dr. Kushner agreed, but also noted that, ever since some earlier antidepressants were associated with risk for suicidal ideation and death, strict guidelines were put in place that exclude certain patients from participating in clinical trials.
Dr. Yanovski suggested that now that the drugs are approved, it would be possible to study this, and the information would be important for clinicians.
Dr. Kushner said he hopes that such studies are forthcoming. In the meantime, “data like this will add some support and understanding,” he suggested.
36,000 Patients with obesity, 500 on antidepressants
Many people living with obesity report taking antidepressants for depression, anxiety, chronic pain, obsessive-compulsive disorder, sleep disturbance, neuropathy, panic disorder, or posttraumatic stress disorder, Dr. Kushner noted.
However, some of these medications can cause weight gain, and little is known about treatment outcomes for people with obesity who are on antidepressants, since most weight-loss studies exclude people with active major depressive disorder.
The researchers analyzed data from 1,961 patients in STEP 1 and 807 patients in STEP 2 as well as 611 patients in STEP 3 and 304 patients in STEP 5 – 3,683 participants in total, of which 539 were on antidepressants at baseline.
The patients were randomly assigned to 2.4 mg semaglutide vs. placebo plus a lifestyle intervention (STEP 1, 2, and 5) or intensive behavioral therapy (STEP 3 only), for 68 weeks, except STEP 5, which was 104 weeks.
Patients were included if they were aged 18 or older with a body mass index ≥30 kg/m2, or ≥27 kg/m2 with more than one weight-related complication (STEP 1, 3, and 5) or BMI ≥27 kg/m2 with type 2 diabetes (STEP 2 only), and at least one self-reported unsuccessful effort to lose weight by diet.
They were excluded if they had active major depressive disorder within 2 years prior to screening (or other severe psychiatric disorders such as schizophrenia or bipolar disorder) or a Patient Health Questionnaire-9 score of 15 or higher (indicating moderately severe or severe depression), or suicide ideation (type 4 or 5 on the Columbia Suicide Severity Rating Scale) or suicide behavior, within 30 days of screening.
From baseline to week 68, patients on semaglutide (with/without baseline antidepressant use) had a significantly greater change in weight vs. patients on placebo (with/without baseline antidepressant use), respectively:
- STEP 1: –15.7% / –14.7% vs. –0.2% / –2.8%
- STEP 2: –10.7% / –9.5% vs. –3.3% / –3.4%
- STEP 3: –16.2% / –15.9% vs. –5.0% / –5.9%
- STEP 5: –19.0% / –14.1% vs. +1.6% / – 4.0%.
The proportion of reported adverse events was generally slightly greater in patients receiving semaglutide (with/without baseline antidepressant use) than those on placebo (with/without baseline antidepressant use), respectively:
- STEP 1: 97.7% vs 88.6% and 92.9% vs. 86%
- STEP 2: 97.6% vs 86.5% and 88.6% vs. 77.2%
- STEP 3: 97.6% vs 95.3% and 100% vs. 95.8%
- STEP 5: 100% vs 94.8% and 95.5% vs. 89.2%.
Gastrointestinal adverse events were more frequently reported in the semaglutide group and in patients on antidepressants at baseline. The proportion of patients with psychiatric adverse events was greater in participants on antidepressants at baseline. There were no differences in suicidal ideation/behavior in patients with/without antidepressant use at baseline.
The STEP trials were funded by Novo Nordisk. Dr. Kushner discloses that he served as a consultant for Novo Nordisk, WeightWatchers, Eli Lilly, and Pfizer, and received a research grant from Epitomee.
A version of this article appeared on Medscape.com.
in a post hoc analysis of the Semaglutide Treatment Effect in People with Obesity (STEP) program.
Adverse events, including psychiatric events, were slightly more usual in the patients on antidepressants, Robert Kushner, MD, noted, in an oral session at the annual meeting of the Obesity Society.
“It is very common that patients who present for weight management are taking antidepressants for various reasons, including depression, anxiety, insomnia, or chronic pain,”Dr. Kushner, from Northwestern University in Chicago, said in an email. “We wanted to see if these participants responded differently to semaglutide, compared to those not on antidepressants.”
“We found that antidepressants do not blunt the effect of semaglutide for weight loss,” he said. “However, there is a slight increase in reported adverse effects.”
“Semaglutide 2.4 mg provides an effective treatment option for weight management, regardless of antidepressant use at baseline,” Dr. Kushner summarized. “Clinicians should be assured that we can use semaglutide in this population of patients.”
Jack Yanovski, MD, PhD, said this was a “great presentation,” noting that “it’s really important that we understand what goes on in patients with depression.”
“Of course, all these trials still had rules that prevent the folks with the most severe depressive symptoms or past suicidality to participate,” added Dr. Yanovski, chief of the Growth and Obesity Section, Eunice Kennedy Shriver National Institute of Child Health and Human Development, Rockville, Md. “We need specific trials to know exactly how well we do.”
Dr. Kushner agreed, but also noted that, ever since some earlier antidepressants were associated with risk for suicidal ideation and death, strict guidelines were put in place that exclude certain patients from participating in clinical trials.
Dr. Yanovski suggested that now that the drugs are approved, it would be possible to study this, and the information would be important for clinicians.
Dr. Kushner said he hopes that such studies are forthcoming. In the meantime, “data like this will add some support and understanding,” he suggested.
36,000 Patients with obesity, 500 on antidepressants
Many people living with obesity report taking antidepressants for depression, anxiety, chronic pain, obsessive-compulsive disorder, sleep disturbance, neuropathy, panic disorder, or posttraumatic stress disorder, Dr. Kushner noted.
However, some of these medications can cause weight gain, and little is known about treatment outcomes for people with obesity who are on antidepressants, since most weight-loss studies exclude people with active major depressive disorder.
The researchers analyzed data from 1,961 patients in STEP 1 and 807 patients in STEP 2 as well as 611 patients in STEP 3 and 304 patients in STEP 5 – 3,683 participants in total, of which 539 were on antidepressants at baseline.
The patients were randomly assigned to 2.4 mg semaglutide vs. placebo plus a lifestyle intervention (STEP 1, 2, and 5) or intensive behavioral therapy (STEP 3 only), for 68 weeks, except STEP 5, which was 104 weeks.
Patients were included if they were aged 18 or older with a body mass index ≥30 kg/m2, or ≥27 kg/m2 with more than one weight-related complication (STEP 1, 3, and 5) or BMI ≥27 kg/m2 with type 2 diabetes (STEP 2 only), and at least one self-reported unsuccessful effort to lose weight by diet.
They were excluded if they had active major depressive disorder within 2 years prior to screening (or other severe psychiatric disorders such as schizophrenia or bipolar disorder) or a Patient Health Questionnaire-9 score of 15 or higher (indicating moderately severe or severe depression), or suicide ideation (type 4 or 5 on the Columbia Suicide Severity Rating Scale) or suicide behavior, within 30 days of screening.
From baseline to week 68, patients on semaglutide (with/without baseline antidepressant use) had a significantly greater change in weight vs. patients on placebo (with/without baseline antidepressant use), respectively:
- STEP 1: –15.7% / –14.7% vs. –0.2% / –2.8%
- STEP 2: –10.7% / –9.5% vs. –3.3% / –3.4%
- STEP 3: –16.2% / –15.9% vs. –5.0% / –5.9%
- STEP 5: –19.0% / –14.1% vs. +1.6% / – 4.0%.
The proportion of reported adverse events was generally slightly greater in patients receiving semaglutide (with/without baseline antidepressant use) than those on placebo (with/without baseline antidepressant use), respectively:
- STEP 1: 97.7% vs 88.6% and 92.9% vs. 86%
- STEP 2: 97.6% vs 86.5% and 88.6% vs. 77.2%
- STEP 3: 97.6% vs 95.3% and 100% vs. 95.8%
- STEP 5: 100% vs 94.8% and 95.5% vs. 89.2%.
Gastrointestinal adverse events were more frequently reported in the semaglutide group and in patients on antidepressants at baseline. The proportion of patients with psychiatric adverse events was greater in participants on antidepressants at baseline. There were no differences in suicidal ideation/behavior in patients with/without antidepressant use at baseline.
The STEP trials were funded by Novo Nordisk. Dr. Kushner discloses that he served as a consultant for Novo Nordisk, WeightWatchers, Eli Lilly, and Pfizer, and received a research grant from Epitomee.
A version of this article appeared on Medscape.com.
FROM OBESITYWEEK® 2023
Taking a new obesity drug and birth control pills? Be careful
For women who are obese, daily life is wrought with landmines. Whether it’s the challenges of air travel because plane seats are too small, the need to shield themselves from the world’s discriminating eyes, or the great lengths many will go to achieve better health and the promise of longevity, navigating life as an obese person requires a thick skin.
So, it’s no wonder so many are willing to pay more than $1,000 a month out of pocket to get their hands on drugs like semaglutide (Ozempic and Wegovy) or tirzepatide (Mounjaro). The benefits of these drugs, which are part of a new class called glucagonlike peptide–1 (GLP-1) receptor agonists, include significant and rapid weight loss, blood sugar control, and improved life quality; they are unprecedented in a setting where surgery has long been considered the most effective long-term option.
On the flip side, the desire for rapid weight loss and better blood sugar control also comes with an unexpected cost. , making an unintended pregnancy more likely.
Neel Shah, MD, an endocrinologist and associate professor at the University of Texas Health Science Center at Houston, said he has had several patients become pregnant without intending to.
“It was when Mounjaro came out on the market when we started using it,” he said of the drug the Food and Drug Administration approved for type 2 diabetes in 2022. “It [the warning] was in the product insert, but clinically speaking, I don’t know if it was at the top of providers’ minds when they were prescribing Mounjaro.”
When asked if he believed that we were going to be seeing a significant increase in so-called Mounjaro babies, Dr. Shah was sure in his response.
“Absolutely. We will because the sheer volume [of patients] will increase,” he said.
It’s all in the gut
One of the ways that drugs like Mounjaro work is by delaying the time that it takes for food to move from the stomach to the small intestine. Although data are still evolving, it is believed that this process – delayed gastric emptying – may affect the absorption of birth control pills.
Dr. Shah said another theory is that vomiting, which is a common side effect of these types of drugs, also affects the pills’ ability to prevent pregnancy.
And “there’s a prolonged period of ramping up the dose because of the GI side effects,” said Pinar Kodaman, MD, PhD, a reproductive endocrinologist and assistant professor of gynecology at Yale University in New Haven, Conn.
“Initially, at the lowest dose, there may not be a lot of potential effect on absorption and gastric emptying. But as the dose goes up, it becomes more common, and it can cause diarrhea, which is another condition that can affect the absorption of any medication,” she said.
Unanticipated outcomes, extra prevention
Roughly 42% of women in the United States are obese, 40% of whom are between the ages of 20 and 39. Although these new drugs can improve fertility outcomes for women who are obese (especially those with polycystic ovary syndrome, or PCOS), only one – Mounjaro – currently carries a warning about birth control pill effectiveness on its label. Unfortunately, it appears that some doctors are unaware or not counseling patients about this risk, and the data are unclear about whether other drugs in this class, like Ozempic and Wegovy, have the same risks.
“To date, it hasn’t been a typical thing that we counsel about,” said Dr. Kodaman. “It’s all fairly new, but when we have patients on birth control pills, we do review other medications that they are on because some can affect efficacy, and it’s something to keep in mind.”
It’s also unclear if other forms of birth control – for example, birth control patches that deliver through the skin – might carry similar pregnancy risks. Dr. Shah said some of his patients who became pregnant without intending to were using these patches. This raises even more questions, since they deliver drugs through the skin directly into the bloodstream and not through the GI system.
What can women do to help ensure that they don’t become pregnant while using these drugs?
“I really think that if patients want to protect themselves from an unplanned pregnancy, that as soon as they start the GLP receptor agonists, it wouldn’t be a bad idea to use condoms, because the onset of action is pretty quick,” said Dr. Kodaman, noting also that “at the lowest dose there may not be a lot of potential effect on gastric emptying. But as the dose goes up, it becomes much more common or can cause diarrhea.”
Dr. Shah said that in his practice he’s “been telling patients to add barrier contraception” 4 weeks before they start their first dose “and at any dose adjustment.”
Zoobia Chaudhry, an obesity medicine doctor and assistant professor of medicine at Johns Hopkins University in Baltimore, recommends that “patients just make sure that the injection and medication that they take are at least 1 hour apart.”
“Most of the time, patients do take birth control before bedtime, so if the two are spaced, it should be OK,” she said.
Another option is for women to speak to their doctors about other contraceptive options like IUDs or implantable rods, where gastric absorption is not going to be an issue.
“There’s very little research on this class of drugs,” said Emily Goodstein, a 40-year-old small-business owner in Washington, who recently switched from Ozempic to Mounjaro. “Being a person who lives in a larger body is such a horrifying experience because of the way that the world discriminates against you.”
She appreciates the feeling of being proactive that these new drugs grant. It has “opened up a bunch of opportunities for me to be seen as a full individual by the medical establishment,” she said. “I was willing to take the risk, knowing that I would be on these drugs for the rest of my life.”
In addition to being what Dr. Goodstein refers to as a guinea pig, she said she made sure that her primary care doctor was aware that she was not trying or planning to become pregnant again. (She has a 3-year-old child.) Still, her doctor mentioned only the most common side effects linked to these drugs, like nausea, vomiting, and diarrhea, and did not mention the risk of pregnancy.
“Folks are really not talking about the reproductive implications,” she said, referring to members of a Facebook group on these drugs that she belongs to.
Like patients themselves, many doctors are just beginning to get their arms around these agents. “Awareness, education, provider involvement, and having a multidisciplinary team could help patients achieve the goals that they set out for themselves,” said Dr. Shah.
Clear conversations are key.
A version of this article first appeared on WebMD.com.
For women who are obese, daily life is wrought with landmines. Whether it’s the challenges of air travel because plane seats are too small, the need to shield themselves from the world’s discriminating eyes, or the great lengths many will go to achieve better health and the promise of longevity, navigating life as an obese person requires a thick skin.
So, it’s no wonder so many are willing to pay more than $1,000 a month out of pocket to get their hands on drugs like semaglutide (Ozempic and Wegovy) or tirzepatide (Mounjaro). The benefits of these drugs, which are part of a new class called glucagonlike peptide–1 (GLP-1) receptor agonists, include significant and rapid weight loss, blood sugar control, and improved life quality; they are unprecedented in a setting where surgery has long been considered the most effective long-term option.
On the flip side, the desire for rapid weight loss and better blood sugar control also comes with an unexpected cost. , making an unintended pregnancy more likely.
Neel Shah, MD, an endocrinologist and associate professor at the University of Texas Health Science Center at Houston, said he has had several patients become pregnant without intending to.
“It was when Mounjaro came out on the market when we started using it,” he said of the drug the Food and Drug Administration approved for type 2 diabetes in 2022. “It [the warning] was in the product insert, but clinically speaking, I don’t know if it was at the top of providers’ minds when they were prescribing Mounjaro.”
When asked if he believed that we were going to be seeing a significant increase in so-called Mounjaro babies, Dr. Shah was sure in his response.
“Absolutely. We will because the sheer volume [of patients] will increase,” he said.
It’s all in the gut
One of the ways that drugs like Mounjaro work is by delaying the time that it takes for food to move from the stomach to the small intestine. Although data are still evolving, it is believed that this process – delayed gastric emptying – may affect the absorption of birth control pills.
Dr. Shah said another theory is that vomiting, which is a common side effect of these types of drugs, also affects the pills’ ability to prevent pregnancy.
And “there’s a prolonged period of ramping up the dose because of the GI side effects,” said Pinar Kodaman, MD, PhD, a reproductive endocrinologist and assistant professor of gynecology at Yale University in New Haven, Conn.
“Initially, at the lowest dose, there may not be a lot of potential effect on absorption and gastric emptying. But as the dose goes up, it becomes more common, and it can cause diarrhea, which is another condition that can affect the absorption of any medication,” she said.
Unanticipated outcomes, extra prevention
Roughly 42% of women in the United States are obese, 40% of whom are between the ages of 20 and 39. Although these new drugs can improve fertility outcomes for women who are obese (especially those with polycystic ovary syndrome, or PCOS), only one – Mounjaro – currently carries a warning about birth control pill effectiveness on its label. Unfortunately, it appears that some doctors are unaware or not counseling patients about this risk, and the data are unclear about whether other drugs in this class, like Ozempic and Wegovy, have the same risks.
“To date, it hasn’t been a typical thing that we counsel about,” said Dr. Kodaman. “It’s all fairly new, but when we have patients on birth control pills, we do review other medications that they are on because some can affect efficacy, and it’s something to keep in mind.”
It’s also unclear if other forms of birth control – for example, birth control patches that deliver through the skin – might carry similar pregnancy risks. Dr. Shah said some of his patients who became pregnant without intending to were using these patches. This raises even more questions, since they deliver drugs through the skin directly into the bloodstream and not through the GI system.
What can women do to help ensure that they don’t become pregnant while using these drugs?
“I really think that if patients want to protect themselves from an unplanned pregnancy, that as soon as they start the GLP receptor agonists, it wouldn’t be a bad idea to use condoms, because the onset of action is pretty quick,” said Dr. Kodaman, noting also that “at the lowest dose there may not be a lot of potential effect on gastric emptying. But as the dose goes up, it becomes much more common or can cause diarrhea.”
Dr. Shah said that in his practice he’s “been telling patients to add barrier contraception” 4 weeks before they start their first dose “and at any dose adjustment.”
Zoobia Chaudhry, an obesity medicine doctor and assistant professor of medicine at Johns Hopkins University in Baltimore, recommends that “patients just make sure that the injection and medication that they take are at least 1 hour apart.”
“Most of the time, patients do take birth control before bedtime, so if the two are spaced, it should be OK,” she said.
Another option is for women to speak to their doctors about other contraceptive options like IUDs or implantable rods, where gastric absorption is not going to be an issue.
“There’s very little research on this class of drugs,” said Emily Goodstein, a 40-year-old small-business owner in Washington, who recently switched from Ozempic to Mounjaro. “Being a person who lives in a larger body is such a horrifying experience because of the way that the world discriminates against you.”
She appreciates the feeling of being proactive that these new drugs grant. It has “opened up a bunch of opportunities for me to be seen as a full individual by the medical establishment,” she said. “I was willing to take the risk, knowing that I would be on these drugs for the rest of my life.”
In addition to being what Dr. Goodstein refers to as a guinea pig, she said she made sure that her primary care doctor was aware that she was not trying or planning to become pregnant again. (She has a 3-year-old child.) Still, her doctor mentioned only the most common side effects linked to these drugs, like nausea, vomiting, and diarrhea, and did not mention the risk of pregnancy.
“Folks are really not talking about the reproductive implications,” she said, referring to members of a Facebook group on these drugs that she belongs to.
Like patients themselves, many doctors are just beginning to get their arms around these agents. “Awareness, education, provider involvement, and having a multidisciplinary team could help patients achieve the goals that they set out for themselves,” said Dr. Shah.
Clear conversations are key.
A version of this article first appeared on WebMD.com.
For women who are obese, daily life is wrought with landmines. Whether it’s the challenges of air travel because plane seats are too small, the need to shield themselves from the world’s discriminating eyes, or the great lengths many will go to achieve better health and the promise of longevity, navigating life as an obese person requires a thick skin.
So, it’s no wonder so many are willing to pay more than $1,000 a month out of pocket to get their hands on drugs like semaglutide (Ozempic and Wegovy) or tirzepatide (Mounjaro). The benefits of these drugs, which are part of a new class called glucagonlike peptide–1 (GLP-1) receptor agonists, include significant and rapid weight loss, blood sugar control, and improved life quality; they are unprecedented in a setting where surgery has long been considered the most effective long-term option.
On the flip side, the desire for rapid weight loss and better blood sugar control also comes with an unexpected cost. , making an unintended pregnancy more likely.
Neel Shah, MD, an endocrinologist and associate professor at the University of Texas Health Science Center at Houston, said he has had several patients become pregnant without intending to.
“It was when Mounjaro came out on the market when we started using it,” he said of the drug the Food and Drug Administration approved for type 2 diabetes in 2022. “It [the warning] was in the product insert, but clinically speaking, I don’t know if it was at the top of providers’ minds when they were prescribing Mounjaro.”
When asked if he believed that we were going to be seeing a significant increase in so-called Mounjaro babies, Dr. Shah was sure in his response.
“Absolutely. We will because the sheer volume [of patients] will increase,” he said.
It’s all in the gut
One of the ways that drugs like Mounjaro work is by delaying the time that it takes for food to move from the stomach to the small intestine. Although data are still evolving, it is believed that this process – delayed gastric emptying – may affect the absorption of birth control pills.
Dr. Shah said another theory is that vomiting, which is a common side effect of these types of drugs, also affects the pills’ ability to prevent pregnancy.
And “there’s a prolonged period of ramping up the dose because of the GI side effects,” said Pinar Kodaman, MD, PhD, a reproductive endocrinologist and assistant professor of gynecology at Yale University in New Haven, Conn.
“Initially, at the lowest dose, there may not be a lot of potential effect on absorption and gastric emptying. But as the dose goes up, it becomes more common, and it can cause diarrhea, which is another condition that can affect the absorption of any medication,” she said.
Unanticipated outcomes, extra prevention
Roughly 42% of women in the United States are obese, 40% of whom are between the ages of 20 and 39. Although these new drugs can improve fertility outcomes for women who are obese (especially those with polycystic ovary syndrome, or PCOS), only one – Mounjaro – currently carries a warning about birth control pill effectiveness on its label. Unfortunately, it appears that some doctors are unaware or not counseling patients about this risk, and the data are unclear about whether other drugs in this class, like Ozempic and Wegovy, have the same risks.
“To date, it hasn’t been a typical thing that we counsel about,” said Dr. Kodaman. “It’s all fairly new, but when we have patients on birth control pills, we do review other medications that they are on because some can affect efficacy, and it’s something to keep in mind.”
It’s also unclear if other forms of birth control – for example, birth control patches that deliver through the skin – might carry similar pregnancy risks. Dr. Shah said some of his patients who became pregnant without intending to were using these patches. This raises even more questions, since they deliver drugs through the skin directly into the bloodstream and not through the GI system.
What can women do to help ensure that they don’t become pregnant while using these drugs?
“I really think that if patients want to protect themselves from an unplanned pregnancy, that as soon as they start the GLP receptor agonists, it wouldn’t be a bad idea to use condoms, because the onset of action is pretty quick,” said Dr. Kodaman, noting also that “at the lowest dose there may not be a lot of potential effect on gastric emptying. But as the dose goes up, it becomes much more common or can cause diarrhea.”
Dr. Shah said that in his practice he’s “been telling patients to add barrier contraception” 4 weeks before they start their first dose “and at any dose adjustment.”
Zoobia Chaudhry, an obesity medicine doctor and assistant professor of medicine at Johns Hopkins University in Baltimore, recommends that “patients just make sure that the injection and medication that they take are at least 1 hour apart.”
“Most of the time, patients do take birth control before bedtime, so if the two are spaced, it should be OK,” she said.
Another option is for women to speak to their doctors about other contraceptive options like IUDs or implantable rods, where gastric absorption is not going to be an issue.
“There’s very little research on this class of drugs,” said Emily Goodstein, a 40-year-old small-business owner in Washington, who recently switched from Ozempic to Mounjaro. “Being a person who lives in a larger body is such a horrifying experience because of the way that the world discriminates against you.”
She appreciates the feeling of being proactive that these new drugs grant. It has “opened up a bunch of opportunities for me to be seen as a full individual by the medical establishment,” she said. “I was willing to take the risk, knowing that I would be on these drugs for the rest of my life.”
In addition to being what Dr. Goodstein refers to as a guinea pig, she said she made sure that her primary care doctor was aware that she was not trying or planning to become pregnant again. (She has a 3-year-old child.) Still, her doctor mentioned only the most common side effects linked to these drugs, like nausea, vomiting, and diarrhea, and did not mention the risk of pregnancy.
“Folks are really not talking about the reproductive implications,” she said, referring to members of a Facebook group on these drugs that she belongs to.
Like patients themselves, many doctors are just beginning to get their arms around these agents. “Awareness, education, provider involvement, and having a multidisciplinary team could help patients achieve the goals that they set out for themselves,” said Dr. Shah.
Clear conversations are key.
A version of this article first appeared on WebMD.com.
Testosterone replacement benefits men with type 2 diabetes
HAMBURG, GERMANY – Testosterone replacement therapy was associated with significant reductions in hemoglobin A1c at 1 and 2 years among men with type 2 diabetes, a multinational audit shows.
“If you have a patient with type 2 diabetes, sexual dysfunction, or fatigue, please consider checking their testosterone level. And if they fulfill criteria for testosterone deficiency and have had their [prostate-specific antigen] checked, consider a trial of treatment and follow them,” study lead author T. Hugh Jones, MD, consultant physician and endocrinologist at Barnsley (England) Hospital NHS Foundation Trust advised, speaking with this news organization.
Dr. Jones also urges clinicians worldwide to enter their patients’ data into the ABCD Testosterone Audit, which aims to identify long-term outcomes and predictors of response to testosterone replacement therapy.
Dr. Jones, who is also professor of andrology at the University of Sheffield, presented the preliminary data analysis at the annual meeting of the European Association for the Study of Diabetes.
Thus far, a total of 428 men with type 2 diabetes and hypogonadism are entered into the audit, from 34 centers in eight countries: the United Kingdom, Germany, Canada, Brazil, South Africa, New Zealand, Malaysia, and Vietnam. Among 121 of the men at 12 months, there was a drop in A1c from a baseline level of 71.27 mmol/mol (8.7%) to 61.26 mmol/mol (7.8%). Among 104 men at 24 months, the drop was from 71.4 mmol/mol (8.7%) to 55.97 mmol/mol (7.3%). Both decreases were significant (P < .001).
Prior data from Dr. Jones’ group showed that about 40% of men with type 2 diabetes have symptomatic testosterone deficiency. Testosterone deficiency is also associated with adverse effects on cardiovascular risk factors, bone health, muscle strength, sexual function, and psychological well-being, yet it is often overlooked, Dr. Jones noted.
“It’s not typically measured in routine clinical practice. ... Deficiency is very common, but a lot of practitioners don’t treat it and don’t ask about it. But in fact, treatment has very significant benefits for patients. ... We know from sildenafil (Viagra) studies that 60%of people who didn’t respond were testosterone deficient. After being given testosterone, they converted to Viagra responders,” he noted.
Regarding safety concerns, the recent findings from the TRAVERSE study, in which about 70% of participants had type 2 diabetes, demonstrated no increased cardiovascular risk. There was also no association with prostate cancer, although it’s important to monitor prostate-specific antigen in patients for the first year on testosterone replacement, Dr. Jones said.
Asked to comment, endocrinologist Bradley D. Anawalt, MD, chief of medicine at the University of Washington Medical Center, Seattle, told this news organization, “This ‘worldwide survey’ confirms many studies from around the world over the past 20 years. ... [T]he association is due to ‘reverse causation,’ in that diabetes type 2 and obesity lower testosterone concentrations. Weight loss of 5%-10% may raise testosterone concentrations in men with high body mass indices, large waist circumferences, and low blood testosterone concentrations.”
At the same time, Dr. Anawalt pointed to data suggesting that “[t]reatment of androgen deficiency may facilitate lifestyle measures in men with high [body mass indexes] and high risk of type 2 diabetes to prevent, or more likely delay, the development of type 2 diabetes.”
However, both Dr. Jones and Dr. Anawalt emphasized that testosterone therapy would not be expected to affect blood glucose levels or any other cardiometabolic parameters in men who are not testosterone deficient, regardless of diabetes status.
“It’s important when you give testosterone to replace it to the normal level. Adequate treatment gives the greatest benefit,”Dr. Jones said.
As more centers contribute data to the ABCD audit, Jones anticipates collecting clinical practice data on a variety of clinical parameters, including complications, total insulin dose, kidney function, and eventually cardiovascular outcomes.
In the meantime, he said, giving testosterone replacement to men with deficiency can be very rewarding for many reasons. “People feel better. Individual patients come back and say ‘thank you doctor, you’ve given me my life back.’ It’s not often you get that. And the compliance is excellent.”
Dr. Jones is a speaker for, advisory board member for, and/or travel grant recipient of Besins Healthcare, Grantss, Grunenthal, and Simple Pharma. Dr. Anawalt has no disclosures.
A version of this article first appeared on Medscape.com.
HAMBURG, GERMANY – Testosterone replacement therapy was associated with significant reductions in hemoglobin A1c at 1 and 2 years among men with type 2 diabetes, a multinational audit shows.
“If you have a patient with type 2 diabetes, sexual dysfunction, or fatigue, please consider checking their testosterone level. And if they fulfill criteria for testosterone deficiency and have had their [prostate-specific antigen] checked, consider a trial of treatment and follow them,” study lead author T. Hugh Jones, MD, consultant physician and endocrinologist at Barnsley (England) Hospital NHS Foundation Trust advised, speaking with this news organization.
Dr. Jones also urges clinicians worldwide to enter their patients’ data into the ABCD Testosterone Audit, which aims to identify long-term outcomes and predictors of response to testosterone replacement therapy.
Dr. Jones, who is also professor of andrology at the University of Sheffield, presented the preliminary data analysis at the annual meeting of the European Association for the Study of Diabetes.
Thus far, a total of 428 men with type 2 diabetes and hypogonadism are entered into the audit, from 34 centers in eight countries: the United Kingdom, Germany, Canada, Brazil, South Africa, New Zealand, Malaysia, and Vietnam. Among 121 of the men at 12 months, there was a drop in A1c from a baseline level of 71.27 mmol/mol (8.7%) to 61.26 mmol/mol (7.8%). Among 104 men at 24 months, the drop was from 71.4 mmol/mol (8.7%) to 55.97 mmol/mol (7.3%). Both decreases were significant (P < .001).
Prior data from Dr. Jones’ group showed that about 40% of men with type 2 diabetes have symptomatic testosterone deficiency. Testosterone deficiency is also associated with adverse effects on cardiovascular risk factors, bone health, muscle strength, sexual function, and psychological well-being, yet it is often overlooked, Dr. Jones noted.
“It’s not typically measured in routine clinical practice. ... Deficiency is very common, but a lot of practitioners don’t treat it and don’t ask about it. But in fact, treatment has very significant benefits for patients. ... We know from sildenafil (Viagra) studies that 60%of people who didn’t respond were testosterone deficient. After being given testosterone, they converted to Viagra responders,” he noted.
Regarding safety concerns, the recent findings from the TRAVERSE study, in which about 70% of participants had type 2 diabetes, demonstrated no increased cardiovascular risk. There was also no association with prostate cancer, although it’s important to monitor prostate-specific antigen in patients for the first year on testosterone replacement, Dr. Jones said.
Asked to comment, endocrinologist Bradley D. Anawalt, MD, chief of medicine at the University of Washington Medical Center, Seattle, told this news organization, “This ‘worldwide survey’ confirms many studies from around the world over the past 20 years. ... [T]he association is due to ‘reverse causation,’ in that diabetes type 2 and obesity lower testosterone concentrations. Weight loss of 5%-10% may raise testosterone concentrations in men with high body mass indices, large waist circumferences, and low blood testosterone concentrations.”
At the same time, Dr. Anawalt pointed to data suggesting that “[t]reatment of androgen deficiency may facilitate lifestyle measures in men with high [body mass indexes] and high risk of type 2 diabetes to prevent, or more likely delay, the development of type 2 diabetes.”
However, both Dr. Jones and Dr. Anawalt emphasized that testosterone therapy would not be expected to affect blood glucose levels or any other cardiometabolic parameters in men who are not testosterone deficient, regardless of diabetes status.
“It’s important when you give testosterone to replace it to the normal level. Adequate treatment gives the greatest benefit,”Dr. Jones said.
As more centers contribute data to the ABCD audit, Jones anticipates collecting clinical practice data on a variety of clinical parameters, including complications, total insulin dose, kidney function, and eventually cardiovascular outcomes.
In the meantime, he said, giving testosterone replacement to men with deficiency can be very rewarding for many reasons. “People feel better. Individual patients come back and say ‘thank you doctor, you’ve given me my life back.’ It’s not often you get that. And the compliance is excellent.”
Dr. Jones is a speaker for, advisory board member for, and/or travel grant recipient of Besins Healthcare, Grantss, Grunenthal, and Simple Pharma. Dr. Anawalt has no disclosures.
A version of this article first appeared on Medscape.com.
HAMBURG, GERMANY – Testosterone replacement therapy was associated with significant reductions in hemoglobin A1c at 1 and 2 years among men with type 2 diabetes, a multinational audit shows.
“If you have a patient with type 2 diabetes, sexual dysfunction, or fatigue, please consider checking their testosterone level. And if they fulfill criteria for testosterone deficiency and have had their [prostate-specific antigen] checked, consider a trial of treatment and follow them,” study lead author T. Hugh Jones, MD, consultant physician and endocrinologist at Barnsley (England) Hospital NHS Foundation Trust advised, speaking with this news organization.
Dr. Jones also urges clinicians worldwide to enter their patients’ data into the ABCD Testosterone Audit, which aims to identify long-term outcomes and predictors of response to testosterone replacement therapy.
Dr. Jones, who is also professor of andrology at the University of Sheffield, presented the preliminary data analysis at the annual meeting of the European Association for the Study of Diabetes.
Thus far, a total of 428 men with type 2 diabetes and hypogonadism are entered into the audit, from 34 centers in eight countries: the United Kingdom, Germany, Canada, Brazil, South Africa, New Zealand, Malaysia, and Vietnam. Among 121 of the men at 12 months, there was a drop in A1c from a baseline level of 71.27 mmol/mol (8.7%) to 61.26 mmol/mol (7.8%). Among 104 men at 24 months, the drop was from 71.4 mmol/mol (8.7%) to 55.97 mmol/mol (7.3%). Both decreases were significant (P < .001).
Prior data from Dr. Jones’ group showed that about 40% of men with type 2 diabetes have symptomatic testosterone deficiency. Testosterone deficiency is also associated with adverse effects on cardiovascular risk factors, bone health, muscle strength, sexual function, and psychological well-being, yet it is often overlooked, Dr. Jones noted.
“It’s not typically measured in routine clinical practice. ... Deficiency is very common, but a lot of practitioners don’t treat it and don’t ask about it. But in fact, treatment has very significant benefits for patients. ... We know from sildenafil (Viagra) studies that 60%of people who didn’t respond were testosterone deficient. After being given testosterone, they converted to Viagra responders,” he noted.
Regarding safety concerns, the recent findings from the TRAVERSE study, in which about 70% of participants had type 2 diabetes, demonstrated no increased cardiovascular risk. There was also no association with prostate cancer, although it’s important to monitor prostate-specific antigen in patients for the first year on testosterone replacement, Dr. Jones said.
Asked to comment, endocrinologist Bradley D. Anawalt, MD, chief of medicine at the University of Washington Medical Center, Seattle, told this news organization, “This ‘worldwide survey’ confirms many studies from around the world over the past 20 years. ... [T]he association is due to ‘reverse causation,’ in that diabetes type 2 and obesity lower testosterone concentrations. Weight loss of 5%-10% may raise testosterone concentrations in men with high body mass indices, large waist circumferences, and low blood testosterone concentrations.”
At the same time, Dr. Anawalt pointed to data suggesting that “[t]reatment of androgen deficiency may facilitate lifestyle measures in men with high [body mass indexes] and high risk of type 2 diabetes to prevent, or more likely delay, the development of type 2 diabetes.”
However, both Dr. Jones and Dr. Anawalt emphasized that testosterone therapy would not be expected to affect blood glucose levels or any other cardiometabolic parameters in men who are not testosterone deficient, regardless of diabetes status.
“It’s important when you give testosterone to replace it to the normal level. Adequate treatment gives the greatest benefit,”Dr. Jones said.
As more centers contribute data to the ABCD audit, Jones anticipates collecting clinical practice data on a variety of clinical parameters, including complications, total insulin dose, kidney function, and eventually cardiovascular outcomes.
In the meantime, he said, giving testosterone replacement to men with deficiency can be very rewarding for many reasons. “People feel better. Individual patients come back and say ‘thank you doctor, you’ve given me my life back.’ It’s not often you get that. And the compliance is excellent.”
Dr. Jones is a speaker for, advisory board member for, and/or travel grant recipient of Besins Healthcare, Grantss, Grunenthal, and Simple Pharma. Dr. Anawalt has no disclosures.
A version of this article first appeared on Medscape.com.
AT EASD 2023

