User login
Gestational diabetes: Treatment controversy rages on
WASHINGTON – Pharmacologic treatment of gestational diabetes remains controversial, with the American College of Obstetricians and Gynecologists and the American Diabetes Association firmly recommending insulin as the preferred first-line pharmacologic therapy, and the Society of Maternal-Fetal Medicine more accepting of metformin as a “reasonable and safe first-line” alternative to insulin and stating that there are no strong data supporting metformin over the sulfonylurea glyburide.
If there’s one main take-away, Mark B. Landon, MD, said at the biennial meeting of the Diabetes in Pregnancy Study Group of North America, it was that “the primary concern” about the use of oral agents for treating gestational diabetes mellitus (GDM) is that there is limited long-term follow-up of exposed offspring.
“The claim that long-term safety data are not available for any oral agent is probably the most valid warning [of any of the concerns voiced by professional organizations],” said Dr. Landon, Richard L. Meiling professor and chair of the department of obstetrics and gynecology at The Ohio State University Wexner Medical Center, Columbus.
Otherwise, he said, there are not enough data to firmly prioritize the drugs most commonly used for GDM, and “the superiority of insulin over oral agents simply remains questionable.”
ACOG’s 2017 level A recommendation for insulin as the first-line option when pharmacologic treatment is needed for treating GDM (Obstet Gynecol. 2017;130[1]:e17-37) was followed in 2018 by another updated practice bulletin on GDM (Obstet Gynecol. 2018;131[2]:e49-64) that considered several meta-analyses published in 2017 and reiterated a preference for insulin.
Those recent meta-analyses of pharmacologic treatment of GDM show that the available literature is generally of “poor trial quality,” and that studies are small and not designed to assess equivalence or noninferiority, Mark Turrentine, MD, chair of ACOG’s committee on practice bulletins, said in an interview. “Taking that into account and [considering] that oral antidiabetic medications are not approved by the Food and Drug Administration [for the treatment of GDM], that they cross the placenta, and that we currently lack long-term neonatal safety data ... we felt that insulin is the preferred treatment.”
In its 2017 and 2018 bulletins, ACOG said that metformin is a “reasonable alternative choice” for women who decline insulin therapy or who may be unable to safely administer it (a level B recommendation). The 2018 practice bulletin mentions one additional factor: affordability. “Insurance companies aren’t always covering [insulin],” said Dr. Turrentine, of the department of obstetrics and gynecology, Baylor College of Medicine, Houston. “It’s a challenge – no question.”
ACOG says glyburide should not be recommended as a first-line pharmacologic treatment, “because, in most studies, it does not yield outcomes equivalent to insulin or metformin,” Dr. Turrentine emphasized.
Glyburide’s role
Dr. Landon took issue with ACOG’s stance on the sulfonylurea. “Frankly, I think this [conclusion] is debatable,” he said. The trend in the United States – “at least after the 2017 ACOG document came out”– has been toward use of metformin over glyburide when an oral agent is [used], but “I think glyburide has been unfairly trashed. It probably still has a place.”
As Dr. Landon sees it, research published in 2015 put a damper on the use of glyburide, which “had become the number one agent” after an earlier, seminal trial, led by Oded Langer, MD, had shown equivalent glycemic control in about 400 women with GDM who were randomized to receive either insulin or glyburide (N Engl J Med. 2000;343;1134-8). The trial was not powered to evaluate other outcomes, but there were no significant differences in neonatal complications, Dr. Landon said.
One of the 2015 studies – a large, retrospective, population-based study of more than 9,000 women with GDM treated with glyburide or insulin – showed a higher risk of admission to the neonatal intensive care unit (relative risk, 1.41), hypoglycemia in the newborn (RR, 1.40), and large-for-gestational age (RR, 1.43) with glyburide, compared with insulin (JAMA Pediatr. 2015;169[5]:452-8).
A meta-analysis of glyburide, metformin, and insulin showed significant differences between glyburide and insulin in birth weight, macrosomia (RR, 2.62), and neonatal hypoglycemia (RR, 2.04; BMJ. 2015;350;h102). However, “this was basically a conglomeration of studies with about 50 [individuals] in each arm, and in which entry criteria for the diagnosis of GDM were rather heterogeneous,” said Dr. Landon. “There are real problems with this and other meta-analyses.”
The authors of a 2018 multicenter, noninferiority, randomized, controlled trial of about 900 women concluded that their study failed to show that the use of glyburide, compared with insulin, does not result in a greater frequency of perinatal complications. The authors also wrote, however, that the “increase in perinatal complications [with glyburide] may be no more than 10.5%, compared with insulin” (JAMA. 2018;319[17]:1773-80).
That increase, Dr. Landon said, was “not an absolute 10%, but 10% of the complication rate, which probably translates to about 2%.” The only component of a composite outcome (including macrosomia, hypoglycemia, and hyperbilirubinemia) that was significantly different, he noted, was hypoglycemia, which affected 12.2% of neonates in the glyburide group and 7.2% in the insulin group.
Glyburide’s role may well be substantiated in the future, Dr. Landon said during a discussion period at the meeting, through research underway at the University of Pittsburgh aimed at tailoring treatment to the underlying pathophysiology of a patient’s GDM.
The MATCh-GDM study (Metabolic Analysis for Treatment Choice in GDM) is randomizing women to receive usual, unmatched treatment or treatment matched to GDM mechanism – metformin for predominant insulin resistance, glyburide or insulin for predominant insulin secretion defects, and one of the three for combined mechanisms. The study’s principal investigator, Maisa Feghali, MD, of the department of obstetrics, gynecology, and reproductive sciences at the University of Pittsburgh, stressed in a presentation on the study that GDM is a heterogeneous condition and that research is needed to understand the impact of GDM subtypes on treatment response.
Metformin outcomes
Concerns about the impact of metformin on short-term perinatal outcomes focus on preterm birth, Dr. Landon said. The only study to date that has shown an increased rate of prematurity, however, is the “seminal” Metformin in Gestational Diabetes (MiG) trial led by Janet A. Rowan, MBChB, that randomized 751 women with GDM in Australia and New Zealand to treatment with metformin or insulin. The researchers found no significant differences between a composite of neonatal complications but did establish that severe hypoglycemia was less common in the metformin group and preterm birth was more common (N Engl J Med. 2008;358:2003-15).
A 2016 systematic review and meta-analysis of short- and long-term outcomes of metformin, compared with insulin, found that metformin did not increase preterm delivery (Diabet Med. 2017;34[1]:27-36). And while the 2015 BMJ meta-analysis found that metformin was associated with higher rates of preterm birth (RR, 1.50), the increased risk “was all driven by the Rowan study,” Dr. Landon said. The 2015 meta-analysis also found that metformin was associated with less maternal weight gain and fewer infants who were large for gestational age.
Metformin is also tainted by high rates of failure in GDM. In the 2008 Rowan study, 46% of patients on metformin failed to achieve glycemic control. “But this is a classic half-full, half-empty [phenomena],” Dr. Landon said. “Some people say this isn’t good, but on the other hand, 54% avoided insulin.”
Indeed, the Society of Maternal-Fetal Medicine (SMFM), in its 2018 statement on the pharmacologic treatment of GDM, said that oral hypoglycemic agents that are used as monotherapy work in “more than half” of GDM pregnancies. The need for adjunctive insulin to achieve glycemic control ranges between 26% and 46% for women using metformin, and 4% and 16% for women using glyburide, it says.
In the society’s view, recent meta-analyses and systemic reviews “support the efficacy and safety of oral agents,” and “although concerns have been raised for more frequent adverse neonatal outcomes with glyburide, including macrosomia and hypoglycemia, the evidence of benefit of one oral agent over the other remains limited.”
The society says that the difference between its statement and the ACOG recommendations is “based on the values placed by different experts and providers on the available evidence,” and it adds that more long-term data are needed.
But as Dr. Landon said, the SMFM is “a little more forgiving” in its interpretation of a limited body of literature. And clinicians, in the meantime, have to navigate the controversy. “The professional organizations don’t make it easy for [us],” he said. At this point, “insulin does not cross the placenta, and the oral agents do cross it. Informed consent is absolutely necessary when choosing oral agents for treating GDM.”
Offspring well-being
Of greater concern than neonatal outcomes are the potential long-term issues for offspring, Dr. Landon said. On the one hand, it is theorized that metformin may protect beta-cell function in offspring and thereby reduce the cross-generational effects of obesity and type 2 diabetes. On the other hand, it is theorized that the drug may cause a decrease in cell-cycle proliferation, which could have “unknown fetal programming effects,” and it may inhibit the mTOR signaling pathway, thus restricting the transport of glucose and amino acids across the placenta, he said. (Findings from in vitro research have suggested that glyburide treatment in GDM might be associated with enhanced transport across the placenta, he noted.)
Long-term follow-up studies of offspring are “clearly needed,” Dr. Landon said. At this point, in regard to long-term safety, he and other experts are concerned primarily about the potential for obesity and metabolic dysfunction in offspring who are exposed to metformin in utero. They are watching follow-up from Dr. Rowan’s MiG trial, as well as elsewhere in the literature, on metformin-exposed offspring from mothers with polycystic ovary syndrome.
A follow-up analysis of offspring from the MiG trial found that children of women with GDM who were exposed to metformin had larger measures of subcutaneous fat at age 2 years, compared with children of mothers treated with insulin alone, but that overall body fat was the same, Dr. Landon noted. The investigators postulated that these children may have less visceral fat and a more favorable pattern of fat distribution (Diab Care. 2011;34:2279-84).
A recently published follow-up analysis of two randomized, controlled trials of women with polycystic ovary syndrome is cause for more concern, he said. That analysis showed that offspring exposed to metformin in utero had a higher body mass index and an increased prevalence of obesity or overweight at age 4 years, compared with placebo groups (J Clin Endocrinol Metab. 2018;103[4]:1612-21).
That analysis of metformin-exposed offspring in the context of polycystic ovary syndrome was published after the SMFM statement, as was another follow-up analysis of MiG trial offspring – this one, at ages 7-9 years – that showed an increase in weight, size, and fat mass in one of two subsets analyzed, despite no difference in large-for-gestational age rates between the metformin- and insulin-exposed offspring (BMJ Open Diabetes Res Care. 2018;6[1]: e000456).
In 2018, a group of 17 prominent diabetes and maternal-fetal medicine researchers cited these findings in a response to the SMFM statement and cautioned against the widespread adoption of metformin use during pregnancy, writing that, based on “both pharmacologic and randomized trial evidence that metformin may create an atypical intrauterine environment ... we believe it is premature to embrace metformin as equivalent to insulin or as superior to glyburide, and that patients should be counseled on the limited long-term safety data and potential for adverse childhood metabolic effects” (Am J Obstet Gynecol. 2018;219[4]:367.e1-7).
WASHINGTON – Pharmacologic treatment of gestational diabetes remains controversial, with the American College of Obstetricians and Gynecologists and the American Diabetes Association firmly recommending insulin as the preferred first-line pharmacologic therapy, and the Society of Maternal-Fetal Medicine more accepting of metformin as a “reasonable and safe first-line” alternative to insulin and stating that there are no strong data supporting metformin over the sulfonylurea glyburide.
If there’s one main take-away, Mark B. Landon, MD, said at the biennial meeting of the Diabetes in Pregnancy Study Group of North America, it was that “the primary concern” about the use of oral agents for treating gestational diabetes mellitus (GDM) is that there is limited long-term follow-up of exposed offspring.
“The claim that long-term safety data are not available for any oral agent is probably the most valid warning [of any of the concerns voiced by professional organizations],” said Dr. Landon, Richard L. Meiling professor and chair of the department of obstetrics and gynecology at The Ohio State University Wexner Medical Center, Columbus.
Otherwise, he said, there are not enough data to firmly prioritize the drugs most commonly used for GDM, and “the superiority of insulin over oral agents simply remains questionable.”
ACOG’s 2017 level A recommendation for insulin as the first-line option when pharmacologic treatment is needed for treating GDM (Obstet Gynecol. 2017;130[1]:e17-37) was followed in 2018 by another updated practice bulletin on GDM (Obstet Gynecol. 2018;131[2]:e49-64) that considered several meta-analyses published in 2017 and reiterated a preference for insulin.
Those recent meta-analyses of pharmacologic treatment of GDM show that the available literature is generally of “poor trial quality,” and that studies are small and not designed to assess equivalence or noninferiority, Mark Turrentine, MD, chair of ACOG’s committee on practice bulletins, said in an interview. “Taking that into account and [considering] that oral antidiabetic medications are not approved by the Food and Drug Administration [for the treatment of GDM], that they cross the placenta, and that we currently lack long-term neonatal safety data ... we felt that insulin is the preferred treatment.”
In its 2017 and 2018 bulletins, ACOG said that metformin is a “reasonable alternative choice” for women who decline insulin therapy or who may be unable to safely administer it (a level B recommendation). The 2018 practice bulletin mentions one additional factor: affordability. “Insurance companies aren’t always covering [insulin],” said Dr. Turrentine, of the department of obstetrics and gynecology, Baylor College of Medicine, Houston. “It’s a challenge – no question.”
ACOG says glyburide should not be recommended as a first-line pharmacologic treatment, “because, in most studies, it does not yield outcomes equivalent to insulin or metformin,” Dr. Turrentine emphasized.
Glyburide’s role
Dr. Landon took issue with ACOG’s stance on the sulfonylurea. “Frankly, I think this [conclusion] is debatable,” he said. The trend in the United States – “at least after the 2017 ACOG document came out”– has been toward use of metformin over glyburide when an oral agent is [used], but “I think glyburide has been unfairly trashed. It probably still has a place.”
As Dr. Landon sees it, research published in 2015 put a damper on the use of glyburide, which “had become the number one agent” after an earlier, seminal trial, led by Oded Langer, MD, had shown equivalent glycemic control in about 400 women with GDM who were randomized to receive either insulin or glyburide (N Engl J Med. 2000;343;1134-8). The trial was not powered to evaluate other outcomes, but there were no significant differences in neonatal complications, Dr. Landon said.
One of the 2015 studies – a large, retrospective, population-based study of more than 9,000 women with GDM treated with glyburide or insulin – showed a higher risk of admission to the neonatal intensive care unit (relative risk, 1.41), hypoglycemia in the newborn (RR, 1.40), and large-for-gestational age (RR, 1.43) with glyburide, compared with insulin (JAMA Pediatr. 2015;169[5]:452-8).
A meta-analysis of glyburide, metformin, and insulin showed significant differences between glyburide and insulin in birth weight, macrosomia (RR, 2.62), and neonatal hypoglycemia (RR, 2.04; BMJ. 2015;350;h102). However, “this was basically a conglomeration of studies with about 50 [individuals] in each arm, and in which entry criteria for the diagnosis of GDM were rather heterogeneous,” said Dr. Landon. “There are real problems with this and other meta-analyses.”
The authors of a 2018 multicenter, noninferiority, randomized, controlled trial of about 900 women concluded that their study failed to show that the use of glyburide, compared with insulin, does not result in a greater frequency of perinatal complications. The authors also wrote, however, that the “increase in perinatal complications [with glyburide] may be no more than 10.5%, compared with insulin” (JAMA. 2018;319[17]:1773-80).
That increase, Dr. Landon said, was “not an absolute 10%, but 10% of the complication rate, which probably translates to about 2%.” The only component of a composite outcome (including macrosomia, hypoglycemia, and hyperbilirubinemia) that was significantly different, he noted, was hypoglycemia, which affected 12.2% of neonates in the glyburide group and 7.2% in the insulin group.
Glyburide’s role may well be substantiated in the future, Dr. Landon said during a discussion period at the meeting, through research underway at the University of Pittsburgh aimed at tailoring treatment to the underlying pathophysiology of a patient’s GDM.
The MATCh-GDM study (Metabolic Analysis for Treatment Choice in GDM) is randomizing women to receive usual, unmatched treatment or treatment matched to GDM mechanism – metformin for predominant insulin resistance, glyburide or insulin for predominant insulin secretion defects, and one of the three for combined mechanisms. The study’s principal investigator, Maisa Feghali, MD, of the department of obstetrics, gynecology, and reproductive sciences at the University of Pittsburgh, stressed in a presentation on the study that GDM is a heterogeneous condition and that research is needed to understand the impact of GDM subtypes on treatment response.
Metformin outcomes
Concerns about the impact of metformin on short-term perinatal outcomes focus on preterm birth, Dr. Landon said. The only study to date that has shown an increased rate of prematurity, however, is the “seminal” Metformin in Gestational Diabetes (MiG) trial led by Janet A. Rowan, MBChB, that randomized 751 women with GDM in Australia and New Zealand to treatment with metformin or insulin. The researchers found no significant differences between a composite of neonatal complications but did establish that severe hypoglycemia was less common in the metformin group and preterm birth was more common (N Engl J Med. 2008;358:2003-15).
A 2016 systematic review and meta-analysis of short- and long-term outcomes of metformin, compared with insulin, found that metformin did not increase preterm delivery (Diabet Med. 2017;34[1]:27-36). And while the 2015 BMJ meta-analysis found that metformin was associated with higher rates of preterm birth (RR, 1.50), the increased risk “was all driven by the Rowan study,” Dr. Landon said. The 2015 meta-analysis also found that metformin was associated with less maternal weight gain and fewer infants who were large for gestational age.
Metformin is also tainted by high rates of failure in GDM. In the 2008 Rowan study, 46% of patients on metformin failed to achieve glycemic control. “But this is a classic half-full, half-empty [phenomena],” Dr. Landon said. “Some people say this isn’t good, but on the other hand, 54% avoided insulin.”
Indeed, the Society of Maternal-Fetal Medicine (SMFM), in its 2018 statement on the pharmacologic treatment of GDM, said that oral hypoglycemic agents that are used as monotherapy work in “more than half” of GDM pregnancies. The need for adjunctive insulin to achieve glycemic control ranges between 26% and 46% for women using metformin, and 4% and 16% for women using glyburide, it says.
In the society’s view, recent meta-analyses and systemic reviews “support the efficacy and safety of oral agents,” and “although concerns have been raised for more frequent adverse neonatal outcomes with glyburide, including macrosomia and hypoglycemia, the evidence of benefit of one oral agent over the other remains limited.”
The society says that the difference between its statement and the ACOG recommendations is “based on the values placed by different experts and providers on the available evidence,” and it adds that more long-term data are needed.
But as Dr. Landon said, the SMFM is “a little more forgiving” in its interpretation of a limited body of literature. And clinicians, in the meantime, have to navigate the controversy. “The professional organizations don’t make it easy for [us],” he said. At this point, “insulin does not cross the placenta, and the oral agents do cross it. Informed consent is absolutely necessary when choosing oral agents for treating GDM.”
Offspring well-being
Of greater concern than neonatal outcomes are the potential long-term issues for offspring, Dr. Landon said. On the one hand, it is theorized that metformin may protect beta-cell function in offspring and thereby reduce the cross-generational effects of obesity and type 2 diabetes. On the other hand, it is theorized that the drug may cause a decrease in cell-cycle proliferation, which could have “unknown fetal programming effects,” and it may inhibit the mTOR signaling pathway, thus restricting the transport of glucose and amino acids across the placenta, he said. (Findings from in vitro research have suggested that glyburide treatment in GDM might be associated with enhanced transport across the placenta, he noted.)
Long-term follow-up studies of offspring are “clearly needed,” Dr. Landon said. At this point, in regard to long-term safety, he and other experts are concerned primarily about the potential for obesity and metabolic dysfunction in offspring who are exposed to metformin in utero. They are watching follow-up from Dr. Rowan’s MiG trial, as well as elsewhere in the literature, on metformin-exposed offspring from mothers with polycystic ovary syndrome.
A follow-up analysis of offspring from the MiG trial found that children of women with GDM who were exposed to metformin had larger measures of subcutaneous fat at age 2 years, compared with children of mothers treated with insulin alone, but that overall body fat was the same, Dr. Landon noted. The investigators postulated that these children may have less visceral fat and a more favorable pattern of fat distribution (Diab Care. 2011;34:2279-84).
A recently published follow-up analysis of two randomized, controlled trials of women with polycystic ovary syndrome is cause for more concern, he said. That analysis showed that offspring exposed to metformin in utero had a higher body mass index and an increased prevalence of obesity or overweight at age 4 years, compared with placebo groups (J Clin Endocrinol Metab. 2018;103[4]:1612-21).
That analysis of metformin-exposed offspring in the context of polycystic ovary syndrome was published after the SMFM statement, as was another follow-up analysis of MiG trial offspring – this one, at ages 7-9 years – that showed an increase in weight, size, and fat mass in one of two subsets analyzed, despite no difference in large-for-gestational age rates between the metformin- and insulin-exposed offspring (BMJ Open Diabetes Res Care. 2018;6[1]: e000456).
In 2018, a group of 17 prominent diabetes and maternal-fetal medicine researchers cited these findings in a response to the SMFM statement and cautioned against the widespread adoption of metformin use during pregnancy, writing that, based on “both pharmacologic and randomized trial evidence that metformin may create an atypical intrauterine environment ... we believe it is premature to embrace metformin as equivalent to insulin or as superior to glyburide, and that patients should be counseled on the limited long-term safety data and potential for adverse childhood metabolic effects” (Am J Obstet Gynecol. 2018;219[4]:367.e1-7).
WASHINGTON – Pharmacologic treatment of gestational diabetes remains controversial, with the American College of Obstetricians and Gynecologists and the American Diabetes Association firmly recommending insulin as the preferred first-line pharmacologic therapy, and the Society of Maternal-Fetal Medicine more accepting of metformin as a “reasonable and safe first-line” alternative to insulin and stating that there are no strong data supporting metformin over the sulfonylurea glyburide.
If there’s one main take-away, Mark B. Landon, MD, said at the biennial meeting of the Diabetes in Pregnancy Study Group of North America, it was that “the primary concern” about the use of oral agents for treating gestational diabetes mellitus (GDM) is that there is limited long-term follow-up of exposed offspring.
“The claim that long-term safety data are not available for any oral agent is probably the most valid warning [of any of the concerns voiced by professional organizations],” said Dr. Landon, Richard L. Meiling professor and chair of the department of obstetrics and gynecology at The Ohio State University Wexner Medical Center, Columbus.
Otherwise, he said, there are not enough data to firmly prioritize the drugs most commonly used for GDM, and “the superiority of insulin over oral agents simply remains questionable.”
ACOG’s 2017 level A recommendation for insulin as the first-line option when pharmacologic treatment is needed for treating GDM (Obstet Gynecol. 2017;130[1]:e17-37) was followed in 2018 by another updated practice bulletin on GDM (Obstet Gynecol. 2018;131[2]:e49-64) that considered several meta-analyses published in 2017 and reiterated a preference for insulin.
Those recent meta-analyses of pharmacologic treatment of GDM show that the available literature is generally of “poor trial quality,” and that studies are small and not designed to assess equivalence or noninferiority, Mark Turrentine, MD, chair of ACOG’s committee on practice bulletins, said in an interview. “Taking that into account and [considering] that oral antidiabetic medications are not approved by the Food and Drug Administration [for the treatment of GDM], that they cross the placenta, and that we currently lack long-term neonatal safety data ... we felt that insulin is the preferred treatment.”
In its 2017 and 2018 bulletins, ACOG said that metformin is a “reasonable alternative choice” for women who decline insulin therapy or who may be unable to safely administer it (a level B recommendation). The 2018 practice bulletin mentions one additional factor: affordability. “Insurance companies aren’t always covering [insulin],” said Dr. Turrentine, of the department of obstetrics and gynecology, Baylor College of Medicine, Houston. “It’s a challenge – no question.”
ACOG says glyburide should not be recommended as a first-line pharmacologic treatment, “because, in most studies, it does not yield outcomes equivalent to insulin or metformin,” Dr. Turrentine emphasized.
Glyburide’s role
Dr. Landon took issue with ACOG’s stance on the sulfonylurea. “Frankly, I think this [conclusion] is debatable,” he said. The trend in the United States – “at least after the 2017 ACOG document came out”– has been toward use of metformin over glyburide when an oral agent is [used], but “I think glyburide has been unfairly trashed. It probably still has a place.”
As Dr. Landon sees it, research published in 2015 put a damper on the use of glyburide, which “had become the number one agent” after an earlier, seminal trial, led by Oded Langer, MD, had shown equivalent glycemic control in about 400 women with GDM who were randomized to receive either insulin or glyburide (N Engl J Med. 2000;343;1134-8). The trial was not powered to evaluate other outcomes, but there were no significant differences in neonatal complications, Dr. Landon said.
One of the 2015 studies – a large, retrospective, population-based study of more than 9,000 women with GDM treated with glyburide or insulin – showed a higher risk of admission to the neonatal intensive care unit (relative risk, 1.41), hypoglycemia in the newborn (RR, 1.40), and large-for-gestational age (RR, 1.43) with glyburide, compared with insulin (JAMA Pediatr. 2015;169[5]:452-8).
A meta-analysis of glyburide, metformin, and insulin showed significant differences between glyburide and insulin in birth weight, macrosomia (RR, 2.62), and neonatal hypoglycemia (RR, 2.04; BMJ. 2015;350;h102). However, “this was basically a conglomeration of studies with about 50 [individuals] in each arm, and in which entry criteria for the diagnosis of GDM were rather heterogeneous,” said Dr. Landon. “There are real problems with this and other meta-analyses.”
The authors of a 2018 multicenter, noninferiority, randomized, controlled trial of about 900 women concluded that their study failed to show that the use of glyburide, compared with insulin, does not result in a greater frequency of perinatal complications. The authors also wrote, however, that the “increase in perinatal complications [with glyburide] may be no more than 10.5%, compared with insulin” (JAMA. 2018;319[17]:1773-80).
That increase, Dr. Landon said, was “not an absolute 10%, but 10% of the complication rate, which probably translates to about 2%.” The only component of a composite outcome (including macrosomia, hypoglycemia, and hyperbilirubinemia) that was significantly different, he noted, was hypoglycemia, which affected 12.2% of neonates in the glyburide group and 7.2% in the insulin group.
Glyburide’s role may well be substantiated in the future, Dr. Landon said during a discussion period at the meeting, through research underway at the University of Pittsburgh aimed at tailoring treatment to the underlying pathophysiology of a patient’s GDM.
The MATCh-GDM study (Metabolic Analysis for Treatment Choice in GDM) is randomizing women to receive usual, unmatched treatment or treatment matched to GDM mechanism – metformin for predominant insulin resistance, glyburide or insulin for predominant insulin secretion defects, and one of the three for combined mechanisms. The study’s principal investigator, Maisa Feghali, MD, of the department of obstetrics, gynecology, and reproductive sciences at the University of Pittsburgh, stressed in a presentation on the study that GDM is a heterogeneous condition and that research is needed to understand the impact of GDM subtypes on treatment response.
Metformin outcomes
Concerns about the impact of metformin on short-term perinatal outcomes focus on preterm birth, Dr. Landon said. The only study to date that has shown an increased rate of prematurity, however, is the “seminal” Metformin in Gestational Diabetes (MiG) trial led by Janet A. Rowan, MBChB, that randomized 751 women with GDM in Australia and New Zealand to treatment with metformin or insulin. The researchers found no significant differences between a composite of neonatal complications but did establish that severe hypoglycemia was less common in the metformin group and preterm birth was more common (N Engl J Med. 2008;358:2003-15).
A 2016 systematic review and meta-analysis of short- and long-term outcomes of metformin, compared with insulin, found that metformin did not increase preterm delivery (Diabet Med. 2017;34[1]:27-36). And while the 2015 BMJ meta-analysis found that metformin was associated with higher rates of preterm birth (RR, 1.50), the increased risk “was all driven by the Rowan study,” Dr. Landon said. The 2015 meta-analysis also found that metformin was associated with less maternal weight gain and fewer infants who were large for gestational age.
Metformin is also tainted by high rates of failure in GDM. In the 2008 Rowan study, 46% of patients on metformin failed to achieve glycemic control. “But this is a classic half-full, half-empty [phenomena],” Dr. Landon said. “Some people say this isn’t good, but on the other hand, 54% avoided insulin.”
Indeed, the Society of Maternal-Fetal Medicine (SMFM), in its 2018 statement on the pharmacologic treatment of GDM, said that oral hypoglycemic agents that are used as monotherapy work in “more than half” of GDM pregnancies. The need for adjunctive insulin to achieve glycemic control ranges between 26% and 46% for women using metformin, and 4% and 16% for women using glyburide, it says.
In the society’s view, recent meta-analyses and systemic reviews “support the efficacy and safety of oral agents,” and “although concerns have been raised for more frequent adverse neonatal outcomes with glyburide, including macrosomia and hypoglycemia, the evidence of benefit of one oral agent over the other remains limited.”
The society says that the difference between its statement and the ACOG recommendations is “based on the values placed by different experts and providers on the available evidence,” and it adds that more long-term data are needed.
But as Dr. Landon said, the SMFM is “a little more forgiving” in its interpretation of a limited body of literature. And clinicians, in the meantime, have to navigate the controversy. “The professional organizations don’t make it easy for [us],” he said. At this point, “insulin does not cross the placenta, and the oral agents do cross it. Informed consent is absolutely necessary when choosing oral agents for treating GDM.”
Offspring well-being
Of greater concern than neonatal outcomes are the potential long-term issues for offspring, Dr. Landon said. On the one hand, it is theorized that metformin may protect beta-cell function in offspring and thereby reduce the cross-generational effects of obesity and type 2 diabetes. On the other hand, it is theorized that the drug may cause a decrease in cell-cycle proliferation, which could have “unknown fetal programming effects,” and it may inhibit the mTOR signaling pathway, thus restricting the transport of glucose and amino acids across the placenta, he said. (Findings from in vitro research have suggested that glyburide treatment in GDM might be associated with enhanced transport across the placenta, he noted.)
Long-term follow-up studies of offspring are “clearly needed,” Dr. Landon said. At this point, in regard to long-term safety, he and other experts are concerned primarily about the potential for obesity and metabolic dysfunction in offspring who are exposed to metformin in utero. They are watching follow-up from Dr. Rowan’s MiG trial, as well as elsewhere in the literature, on metformin-exposed offspring from mothers with polycystic ovary syndrome.
A follow-up analysis of offspring from the MiG trial found that children of women with GDM who were exposed to metformin had larger measures of subcutaneous fat at age 2 years, compared with children of mothers treated with insulin alone, but that overall body fat was the same, Dr. Landon noted. The investigators postulated that these children may have less visceral fat and a more favorable pattern of fat distribution (Diab Care. 2011;34:2279-84).
A recently published follow-up analysis of two randomized, controlled trials of women with polycystic ovary syndrome is cause for more concern, he said. That analysis showed that offspring exposed to metformin in utero had a higher body mass index and an increased prevalence of obesity or overweight at age 4 years, compared with placebo groups (J Clin Endocrinol Metab. 2018;103[4]:1612-21).
That analysis of metformin-exposed offspring in the context of polycystic ovary syndrome was published after the SMFM statement, as was another follow-up analysis of MiG trial offspring – this one, at ages 7-9 years – that showed an increase in weight, size, and fat mass in one of two subsets analyzed, despite no difference in large-for-gestational age rates between the metformin- and insulin-exposed offspring (BMJ Open Diabetes Res Care. 2018;6[1]: e000456).
In 2018, a group of 17 prominent diabetes and maternal-fetal medicine researchers cited these findings in a response to the SMFM statement and cautioned against the widespread adoption of metformin use during pregnancy, writing that, based on “both pharmacologic and randomized trial evidence that metformin may create an atypical intrauterine environment ... we believe it is premature to embrace metformin as equivalent to insulin or as superior to glyburide, and that patients should be counseled on the limited long-term safety data and potential for adverse childhood metabolic effects” (Am J Obstet Gynecol. 2018;219[4]:367.e1-7).
EXPERT ANALYSIS FROM DPSG-NA 2019
High-dose chemo offers survival benefit only for highest-risk breast cancer
High-dose chemotherapy in the adjuvant setting offers a long-term survival advantage for women with very-high-risk stage III breast cancer, but does not improve survival odds for women with lower-risk cancers, an analysis of 20 years of follow-up data shows.
Among 885 women younger than 56 years at the time of treatment who had 4 or more involved axilliary lymph nodes, there was no overall survival difference over 2 decades between the total population of women randomized to receive adjuvant high-dose chemotherapy (HDCT) and those assigned to receive conventional-dose chemotherapy (CDCT).
However, women with 10 or more involved axilliary nodes and those with triple-negative breast cancer had an approximately 15% absolute improvement in 20-year overall survival with high-dose chemotherapy, although the difference for triple-negative disease fell just short of statistical significance, reported Tessa G. Steenbruggen, MD, from the Netherlands Cancer Institute in Amsterdam and colleagues.
“Our analysis confirms earlier results that HDCT has no significant overall survival benefit compared with CDCT for unselected patients with stage III [breast cancer]. However, we found a 14.6%improvement in 20-year OS estimates with HDCT in the predefined subgroup of patients with 10 or more involved [axilliary lymph nodes],” they wrote in JAMA Oncology.
And although other studies of chemotherapy regimens containing high doses of alkylating agents have shown increases in risk of late second malignancies and major cardiovascular events, there were no significant increases of either adverse event with HDCT in this study, the authors noted.
They reported 20-year follow-up results for 885 women who were enrolled in a 10-center randomized clinical trial conducted in the Netherlands from August 1, 1993, through July 31, 1999.
The participants were younger than age 56 years with breast cancer involving at least 4 axillary lymph nodes. All patients underwent surgery with complete axillary clearance and were then randomized to receive either conventional chemotherapy, which consisted of five cycles of fluorouracil 500mg/m2, epirubicin 90 mg/m2, and cyclophosphamide 500mg/m2 (FEC), or high-dose chemotherapy, with the first 4 cycles identical to conventional-dose chemotherapy but the fifth cycle consisting of cyclophosphamide 6000 mg/m2, thiotepa 480 mg/m2, and carboplatin 1600 mg/m2, supported with autologous hematopoietic stem cell transplant.
In addition, all patients received radiotherapy according to the local standard and 2 years of adjuvant tamoxifen.
After a median follow-up of 20.4 years, the 20-year overall survival (OS) rates were 45.3% for patients who had received high-dose chemotherapy and 41.5% for those who had received the conventional dose. This translated into a nonsignificant hazard ratio of 0.89.
However, for patients with 10 or more involved axillary nodes, the 20-year OS rates were 44.5% with HDCT and 29.9% with CDCT, translating into an absolute OS advantage for high-dose chemotherapy of 14.6% and an HR of 0.72 (P = .02).
Respective 20-year OS rates for women with triple-negative breast cancer were 52.9% and 37.5%, an absolute difference of 15.4% and a HR of 0.67, which fell just short of statistical significance, possibly because of the small number of patients with triple-negative breast cancer (140).
“In our 20-year follow-up analysis, there was no increase in cumulative risk for a second malignant neoplasm or for incidence of major cardiovascular events after HDCT,” the investigators wrote.
They noted that women randomized to high-dose chemotherapy had more frequent dysrhythmias, hypertension, and hypercholesterolemia, adding that the latter two adverse events may be partly attributable to a higher incidence of menopause induction among women who received HDCT.
The study was sponsored by University Medical Center Groningen and the The Netherlands Cancer Institute. Dr Steenbruggen reported receiving grants from the Dutch Health Insurance Council during the conduct of the study.
SOURCE: Steenbruggen TG et al. JAMA Oncology. 2020 Jan 30. doi: 10.1001/jamaoncol.2019.6276.
High-dose chemotherapy in the adjuvant setting offers a long-term survival advantage for women with very-high-risk stage III breast cancer, but does not improve survival odds for women with lower-risk cancers, an analysis of 20 years of follow-up data shows.
Among 885 women younger than 56 years at the time of treatment who had 4 or more involved axilliary lymph nodes, there was no overall survival difference over 2 decades between the total population of women randomized to receive adjuvant high-dose chemotherapy (HDCT) and those assigned to receive conventional-dose chemotherapy (CDCT).
However, women with 10 or more involved axilliary nodes and those with triple-negative breast cancer had an approximately 15% absolute improvement in 20-year overall survival with high-dose chemotherapy, although the difference for triple-negative disease fell just short of statistical significance, reported Tessa G. Steenbruggen, MD, from the Netherlands Cancer Institute in Amsterdam and colleagues.
“Our analysis confirms earlier results that HDCT has no significant overall survival benefit compared with CDCT for unselected patients with stage III [breast cancer]. However, we found a 14.6%improvement in 20-year OS estimates with HDCT in the predefined subgroup of patients with 10 or more involved [axilliary lymph nodes],” they wrote in JAMA Oncology.
And although other studies of chemotherapy regimens containing high doses of alkylating agents have shown increases in risk of late second malignancies and major cardiovascular events, there were no significant increases of either adverse event with HDCT in this study, the authors noted.
They reported 20-year follow-up results for 885 women who were enrolled in a 10-center randomized clinical trial conducted in the Netherlands from August 1, 1993, through July 31, 1999.
The participants were younger than age 56 years with breast cancer involving at least 4 axillary lymph nodes. All patients underwent surgery with complete axillary clearance and were then randomized to receive either conventional chemotherapy, which consisted of five cycles of fluorouracil 500mg/m2, epirubicin 90 mg/m2, and cyclophosphamide 500mg/m2 (FEC), or high-dose chemotherapy, with the first 4 cycles identical to conventional-dose chemotherapy but the fifth cycle consisting of cyclophosphamide 6000 mg/m2, thiotepa 480 mg/m2, and carboplatin 1600 mg/m2, supported with autologous hematopoietic stem cell transplant.
In addition, all patients received radiotherapy according to the local standard and 2 years of adjuvant tamoxifen.
After a median follow-up of 20.4 years, the 20-year overall survival (OS) rates were 45.3% for patients who had received high-dose chemotherapy and 41.5% for those who had received the conventional dose. This translated into a nonsignificant hazard ratio of 0.89.
However, for patients with 10 or more involved axillary nodes, the 20-year OS rates were 44.5% with HDCT and 29.9% with CDCT, translating into an absolute OS advantage for high-dose chemotherapy of 14.6% and an HR of 0.72 (P = .02).
Respective 20-year OS rates for women with triple-negative breast cancer were 52.9% and 37.5%, an absolute difference of 15.4% and a HR of 0.67, which fell just short of statistical significance, possibly because of the small number of patients with triple-negative breast cancer (140).
“In our 20-year follow-up analysis, there was no increase in cumulative risk for a second malignant neoplasm or for incidence of major cardiovascular events after HDCT,” the investigators wrote.
They noted that women randomized to high-dose chemotherapy had more frequent dysrhythmias, hypertension, and hypercholesterolemia, adding that the latter two adverse events may be partly attributable to a higher incidence of menopause induction among women who received HDCT.
The study was sponsored by University Medical Center Groningen and the The Netherlands Cancer Institute. Dr Steenbruggen reported receiving grants from the Dutch Health Insurance Council during the conduct of the study.
SOURCE: Steenbruggen TG et al. JAMA Oncology. 2020 Jan 30. doi: 10.1001/jamaoncol.2019.6276.
High-dose chemotherapy in the adjuvant setting offers a long-term survival advantage for women with very-high-risk stage III breast cancer, but does not improve survival odds for women with lower-risk cancers, an analysis of 20 years of follow-up data shows.
Among 885 women younger than 56 years at the time of treatment who had 4 or more involved axilliary lymph nodes, there was no overall survival difference over 2 decades between the total population of women randomized to receive adjuvant high-dose chemotherapy (HDCT) and those assigned to receive conventional-dose chemotherapy (CDCT).
However, women with 10 or more involved axilliary nodes and those with triple-negative breast cancer had an approximately 15% absolute improvement in 20-year overall survival with high-dose chemotherapy, although the difference for triple-negative disease fell just short of statistical significance, reported Tessa G. Steenbruggen, MD, from the Netherlands Cancer Institute in Amsterdam and colleagues.
“Our analysis confirms earlier results that HDCT has no significant overall survival benefit compared with CDCT for unselected patients with stage III [breast cancer]. However, we found a 14.6%improvement in 20-year OS estimates with HDCT in the predefined subgroup of patients with 10 or more involved [axilliary lymph nodes],” they wrote in JAMA Oncology.
And although other studies of chemotherapy regimens containing high doses of alkylating agents have shown increases in risk of late second malignancies and major cardiovascular events, there were no significant increases of either adverse event with HDCT in this study, the authors noted.
They reported 20-year follow-up results for 885 women who were enrolled in a 10-center randomized clinical trial conducted in the Netherlands from August 1, 1993, through July 31, 1999.
The participants were younger than age 56 years with breast cancer involving at least 4 axillary lymph nodes. All patients underwent surgery with complete axillary clearance and were then randomized to receive either conventional chemotherapy, which consisted of five cycles of fluorouracil 500mg/m2, epirubicin 90 mg/m2, and cyclophosphamide 500mg/m2 (FEC), or high-dose chemotherapy, with the first 4 cycles identical to conventional-dose chemotherapy but the fifth cycle consisting of cyclophosphamide 6000 mg/m2, thiotepa 480 mg/m2, and carboplatin 1600 mg/m2, supported with autologous hematopoietic stem cell transplant.
In addition, all patients received radiotherapy according to the local standard and 2 years of adjuvant tamoxifen.
After a median follow-up of 20.4 years, the 20-year overall survival (OS) rates were 45.3% for patients who had received high-dose chemotherapy and 41.5% for those who had received the conventional dose. This translated into a nonsignificant hazard ratio of 0.89.
However, for patients with 10 or more involved axillary nodes, the 20-year OS rates were 44.5% with HDCT and 29.9% with CDCT, translating into an absolute OS advantage for high-dose chemotherapy of 14.6% and an HR of 0.72 (P = .02).
Respective 20-year OS rates for women with triple-negative breast cancer were 52.9% and 37.5%, an absolute difference of 15.4% and a HR of 0.67, which fell just short of statistical significance, possibly because of the small number of patients with triple-negative breast cancer (140).
“In our 20-year follow-up analysis, there was no increase in cumulative risk for a second malignant neoplasm or for incidence of major cardiovascular events after HDCT,” the investigators wrote.
They noted that women randomized to high-dose chemotherapy had more frequent dysrhythmias, hypertension, and hypercholesterolemia, adding that the latter two adverse events may be partly attributable to a higher incidence of menopause induction among women who received HDCT.
The study was sponsored by University Medical Center Groningen and the The Netherlands Cancer Institute. Dr Steenbruggen reported receiving grants from the Dutch Health Insurance Council during the conduct of the study.
SOURCE: Steenbruggen TG et al. JAMA Oncology. 2020 Jan 30. doi: 10.1001/jamaoncol.2019.6276.
FROM JAMA ONCOLOGY
Key clinical point: High-dose chemotherapy offers a long-term breast cancer survival advantage only for women with very-high-risk disease.
Major finding: The absolute 20-year overall survival benefit for women with 10 or more involved lymph nodes was 14.6%.
Study details: Long-term, follow-up study of 885 women under age 56 years with stage III breast cancer treated with adjuvant high- or conventional-dose chemotherapy.
Disclosures: The study was sponsored by University Medical Center Groningen and the The Netherlands Cancer Institute. Dr. Steenbruggen reported receiving grants from the Dutch Health Insurance Council during the conduct of the study.
Source: Steenbruggen TG et al. JAMA Oncology. 2020 Jan 30. doi: 10.1001/jamaoncol.2019.6276.
Psoriasis: A look back over the past 50 years, and forward to next steps
Imagine a patient suffering with horrible psoriasis for decades having failed “every available treatment.” Imagine him living all that time with “flaking, cracking, painful, itchy skin,” only to develop cirrhosis after exposure to toxic therapies.
Then imagine the experience for that patient when, 2 weeks after initiating treatment with a new interleukin-17 inhibitor, his skin clears completely.
“Two weeks later it’s all gone – it was a moment to behold,” said Joel M. Gelfand, MD, professor of dermatology and epidemiology at the University of Pennsylvania, Philadelphia, who had cared for the man for many years before a psoriasis treatment revolution of sorts took the field of dermatology by storm.
“The progress has been breathtaking – there’s no other way to describe it – and it feels like a miracle every time I see a new patient who has tough disease and I have all these things to offer them,” he continued. “For most patients, I can really help them and make a major difference in their life.”
said Mark Lebwohl, MD, Waldman professor of dermatology and chair of the Kimberly and Eric J. Waldman department of dermatology at the Icahn School of Medicine at Mount Sinai, New York.
Dr. Lebwohl recounted some of his own experiences with psoriasis patients before the advent of treatments – particularly biologics – that have transformed practice.
There was a time when psoriasis patients had little more to turn to than the effective – but “disgusting” – Goeckerman Regimen involving cycles of UVB light exposure and topical crude coal tar application. Initially, the regimen, which was introduced in the 1920s, was used around the clock on an inpatient basis until the skin cleared, Dr. Lebwohl said.
In the 1970s, the immunosuppressive chemotherapy drug methotrexate became the first oral systemic therapy approved for severe psoriasis. For those with disabling disease, it offered some hope for relief, but only about 40% of patients achieved at least a 75% reduction in the Psoriasis Area and Severity Index score (PASI 75), he said, adding that they did so at the expense of the liver and bone marrow. “But it was the only thing we had for severe psoriasis other than light treatments.”
In the 1980s and 1990s, oral retinoids emerged as a treatment for psoriasis, and the immunosuppressive drug cyclosporine used to prevent organ rejection in some transplant patients was found to clear psoriasis in affected transplant recipients. Although they brought relief to some patients with severe, disabling disease, these also came with a high price. “It’s not that effective, and it has lots of side effects ... and causes kidney damage in essentially 100% of patients,” Dr. Lebwohl said of cyclosporine.
“So we had treatments that worked, but because the side effects were sufficiently severe, a lot of patients were not treated,” he said.
Enter the biologics era
The early 2000s brought the first two approvals for psoriasis: alefacept (Amevive), a “modestly effective, but quite safe” immunosuppressive dimeric fusion protein approved in early 2003 for moderate to severe plaque psoriasis, and efalizumab (Raptiva), a recombinant humanized monoclonal antibody approved in October 2003; both were T-cell–targeted therapies. The former was withdrawn from the market voluntarily as newer agents became available, and the latter was withdrawn in 2009 because of a link with development of progressive multifocal leukoencephalopathy.
Tumor necrosis factor (TNF) blockers, which had been used effectively for RA and Crohn’s disease, emerged next, and were highly effective, much safer than the systemic treatments, and gained “very widespread use,” Dr. Lebwohl said.
His colleague Alice B. Gottlieb, MD, PhD, was among the pioneers in the development of TNF blockers for the treatment of psoriasis. Her seminal, investigator-initiated paper on the efficacy and safety of infliximab (Remicade) monotherapy for plaque-type psoriasis published in the Lancet in 2001 helped launch the current era in which many psoriasis patients achieve 100% PASI responses with limited side effects, he said, explaining that subsequent research elucidated the role of IL-12 and -23 – leading to effective treatments like ustekinumab (Stelara), and later IL-17, which is, “in fact, the molecule closest to the pathogenesis of psoriasis.”
“If you block IL-17, you get rid of psoriasis,” he said, noting that there are now several companies with approved antibodies to IL-17. “Taltz [ixekizumab] and Cosentyx [secukinumab] are the leading ones, and Siliq [brodalumab] blocks the receptor for IL-17, so it is very effective.”
Another novel biologic – bimekizumab – is on the horizon. It blocks both IL-17a and IL-17f, and appears highly effective in psoriasis and psoriatic arthritis (PsA). “Biologics were the real start of the [psoriasis treatment] revolution,” he said. “When I started out I would speak at patient meetings and the patients were angry at their physicians; they thought they weren’t aggressive enough, they were very frustrated.”
Dr. Lebwohl described patients he would see at annual National Psoriasis Foundation meetings: “There were patients in wheel chairs, because they couldn’t walk. They would be red and scaly all over ... you could have literally swept up scale like it was snow after one of those meetings.
“You go forward to around 2010 – nobody’s in wheelchairs anymore, everybody has clear skin, and it’s become a party; patients are no longer angry – they are thrilled with the results they are getting from much safer and much more effective drugs,” he said. “So it’s been a pleasure taking care of those patients and going from a very difficult time of treating them, to a time where we’ve done a great job treating them.”
Dr. Lebwohl noted that a “large number of dermatologists have been involved with the development of these drugs and making sure they succeed, and that has also been a pleasure to see.”
Dr. Gottlieb, who Dr. Lebwohl has described as “a superstar” in the fields of dermatology and rheumatology, is one such researcher. In an interview, she looked back on her work and the ways that her work “opened the field,” led to many of her trainees also doing “great work,” and changed the lives of patients.
“It’s nice to feel that I really did change, fundamentally, how psoriasis patients are treated,” said Dr. Gottlieb, who is a clinical professor in the department of dermatology at the Icahn School of Medicine at Mount Sinai. “That obviously feels great.”
She recalled a patient – “a 6-foot-5 biker with bad psoriasis” – who “literally, the minute the door closed, he was crying about how horrible his disease was.”
“And I cleared him ... and then you get big hugs – it just feels extremely good ... giving somebody their life back,” she said.
Dr. Gottlieb has been involved in much of the work in developing biologics for psoriasis, including the ongoing work with bimekizumab for PsA as mentioned by Dr. Lebwohl.
If the phase 2 data with bimekizumab are replicated in the ongoing phase 3 trials now underway at her center, “that can really raise the bar ... so if it’s reproducible, it’s very exciting.”
“It’s exciting to have an IL-23 blocker that, at least in clinical trials, showed inhibition of radiographic progression [in PsA],” she said. “That’s guselkumab those data are already out, and I was involved with that.”
The early work of Dr. Gottlieb and others has also “spread to other diseases,” like hidradenitis suppurativa and atopic dermatitis, she said, noting that numerous studies are underway.
Aside from curing all patients, her ultimate goal is getting to a point where psoriasis has no effect on patients’ quality of life.
“And I see it already,” she said. “It’s happening, and it’s nice to see that it’s happening in children now, too; several of the drugs are approved in kids.”
Alan Menter, MD, chairman of the division of dermatology at Baylor University Medical Center, Dallas, also a prolific researcher – and chair of the guidelines committee that published two new sets of guidelines for psoriasis treatment in 2019 – said that the field of dermatology was “late to the biologic evolution,” as many of the early biologics were first approved for PsA.
“But over the last 10 years, things have changed dramatically,” he said. “After that we suddenly leapt ahead of everybody. ... We now have 11 biologic drugs approved for psoriasis, which is more than any other disease has available.”
It’s been “highly exciting” to see this “evolution and revolution,” he commented, adding that one of the next challenges is to address the comorbidities, such as cardiovascular disease, associated with psoriasis.
“The big question now ... is if you improve skin and you improve joints, can you potentially reduce the risk of coronary artery disease,” he said. “Everybody is looking at that, and to me it’s one of the most exciting things that we’re doing.”
Work is ongoing to look at whether the IL-17s and IL-23s have “other indications outside of the skin and joints,” both within and outside of dermatology.
Like Dr. Gottlieb, Dr. Menter also mentioned the potential for hidradenitis suppurativa, and also for a condition that is rarely discussed or studied: genital psoriasis. Ixekizumab has recently been shown to work in about 75% of patients with genital psoriasis, he noted.
Another important area of research is the identification of biomarkers for predicting response and relapse, he said. For now, biomarker research has disappointed, he added, predicting that it will take at least 3-5 years before biomarkers to help guide treatment are identified.
Indeed, Dr. Gelfand, who also is director of the Psoriasis and Phototherapy Treatment Center, vice chair of clinical research, and medical director of the dermatology clinical studies unit at the University of Pennsylvania, agreed there is a need for research to improve treatment selection.
Advances are being made in genetics – with more than 80 different genes now identified as being related to psoriasis – and in medical informatics – which allow thousands of patients to be followed for years, he said, noting that this could elucidate immunopathological features that can improve treatments, predict and prevent comorbidity, and further improve outcomes.
“We also need care that is more patient centered,” he said, describing the ongoing pragmatic LITE trial of home- or office-based phototherapy for which he is the lead investigator, and other studies that he hopes will expand access to care.
Kenneth Brian Gordon, MD, chair and professor of dermatology at the Medical College of Wisconsin, Milwaukee, whose career started in the basic science immunology arena, added the need for expanding benefit to patients with more-moderate disease. Like Dr. Menter, he identified psoriasis as the area in medicine that has had the greatest degree of advancement, except perhaps for hepatitis C.
He described the process not as a “bench-to-bedside” story, but as a bedside-to-bench, then “back-to-bedside” story.
It was really about taking those early T-cell–targeted biologics and anti-TNF agents from bedside to bench with the realization of the importance of the IL-23 and IL-17 pathways, and that understanding led back to the bedside with the development of the newest agents – and to a “huge difference in patient’s lives.”
“But we’ve gotten so good at treating patients with severe disease ... the question now is how to take care of those with more-moderate disease,” he said, noting that a focus on cost and better delivery systems will be needed for that population.
That research is underway, and the future looks bright – and clear.
“I think with psoriasis therapy and where we’ve come in the last 20 years ... we have a hard time remembering what it was like before we had biologic agents” he said. “Our perspective has changed a lot, and sometimes we forget that.”
In fact, “psoriasis has sort of dragged dermatology into the world of modern clinical trial science, and we can now apply that to all sorts of other diseases,” he said. “The psoriasis trials were the first really well-done large-scale trials in dermatology, and I think that has given dermatology a real leg up in how we do clinical research and how we do evidence-based medicine.”
All of the doctors interviewed for this story have received funds and/or honoraria from, consulted with, are employed with, or served on the advisory boards of manufacturers of biologics. Dr. Gelfand is a copatent holder of resiquimod for treatment of cutaneous T-cell lymphoma and is deputy editor of the Journal of Investigative Dermatology.
Imagine a patient suffering with horrible psoriasis for decades having failed “every available treatment.” Imagine him living all that time with “flaking, cracking, painful, itchy skin,” only to develop cirrhosis after exposure to toxic therapies.
Then imagine the experience for that patient when, 2 weeks after initiating treatment with a new interleukin-17 inhibitor, his skin clears completely.
“Two weeks later it’s all gone – it was a moment to behold,” said Joel M. Gelfand, MD, professor of dermatology and epidemiology at the University of Pennsylvania, Philadelphia, who had cared for the man for many years before a psoriasis treatment revolution of sorts took the field of dermatology by storm.
“The progress has been breathtaking – there’s no other way to describe it – and it feels like a miracle every time I see a new patient who has tough disease and I have all these things to offer them,” he continued. “For most patients, I can really help them and make a major difference in their life.”
said Mark Lebwohl, MD, Waldman professor of dermatology and chair of the Kimberly and Eric J. Waldman department of dermatology at the Icahn School of Medicine at Mount Sinai, New York.
Dr. Lebwohl recounted some of his own experiences with psoriasis patients before the advent of treatments – particularly biologics – that have transformed practice.
There was a time when psoriasis patients had little more to turn to than the effective – but “disgusting” – Goeckerman Regimen involving cycles of UVB light exposure and topical crude coal tar application. Initially, the regimen, which was introduced in the 1920s, was used around the clock on an inpatient basis until the skin cleared, Dr. Lebwohl said.
In the 1970s, the immunosuppressive chemotherapy drug methotrexate became the first oral systemic therapy approved for severe psoriasis. For those with disabling disease, it offered some hope for relief, but only about 40% of patients achieved at least a 75% reduction in the Psoriasis Area and Severity Index score (PASI 75), he said, adding that they did so at the expense of the liver and bone marrow. “But it was the only thing we had for severe psoriasis other than light treatments.”
In the 1980s and 1990s, oral retinoids emerged as a treatment for psoriasis, and the immunosuppressive drug cyclosporine used to prevent organ rejection in some transplant patients was found to clear psoriasis in affected transplant recipients. Although they brought relief to some patients with severe, disabling disease, these also came with a high price. “It’s not that effective, and it has lots of side effects ... and causes kidney damage in essentially 100% of patients,” Dr. Lebwohl said of cyclosporine.
“So we had treatments that worked, but because the side effects were sufficiently severe, a lot of patients were not treated,” he said.
Enter the biologics era
The early 2000s brought the first two approvals for psoriasis: alefacept (Amevive), a “modestly effective, but quite safe” immunosuppressive dimeric fusion protein approved in early 2003 for moderate to severe plaque psoriasis, and efalizumab (Raptiva), a recombinant humanized monoclonal antibody approved in October 2003; both were T-cell–targeted therapies. The former was withdrawn from the market voluntarily as newer agents became available, and the latter was withdrawn in 2009 because of a link with development of progressive multifocal leukoencephalopathy.
Tumor necrosis factor (TNF) blockers, which had been used effectively for RA and Crohn’s disease, emerged next, and were highly effective, much safer than the systemic treatments, and gained “very widespread use,” Dr. Lebwohl said.
His colleague Alice B. Gottlieb, MD, PhD, was among the pioneers in the development of TNF blockers for the treatment of psoriasis. Her seminal, investigator-initiated paper on the efficacy and safety of infliximab (Remicade) monotherapy for plaque-type psoriasis published in the Lancet in 2001 helped launch the current era in which many psoriasis patients achieve 100% PASI responses with limited side effects, he said, explaining that subsequent research elucidated the role of IL-12 and -23 – leading to effective treatments like ustekinumab (Stelara), and later IL-17, which is, “in fact, the molecule closest to the pathogenesis of psoriasis.”
“If you block IL-17, you get rid of psoriasis,” he said, noting that there are now several companies with approved antibodies to IL-17. “Taltz [ixekizumab] and Cosentyx [secukinumab] are the leading ones, and Siliq [brodalumab] blocks the receptor for IL-17, so it is very effective.”
Another novel biologic – bimekizumab – is on the horizon. It blocks both IL-17a and IL-17f, and appears highly effective in psoriasis and psoriatic arthritis (PsA). “Biologics were the real start of the [psoriasis treatment] revolution,” he said. “When I started out I would speak at patient meetings and the patients were angry at their physicians; they thought they weren’t aggressive enough, they were very frustrated.”
Dr. Lebwohl described patients he would see at annual National Psoriasis Foundation meetings: “There were patients in wheel chairs, because they couldn’t walk. They would be red and scaly all over ... you could have literally swept up scale like it was snow after one of those meetings.
“You go forward to around 2010 – nobody’s in wheelchairs anymore, everybody has clear skin, and it’s become a party; patients are no longer angry – they are thrilled with the results they are getting from much safer and much more effective drugs,” he said. “So it’s been a pleasure taking care of those patients and going from a very difficult time of treating them, to a time where we’ve done a great job treating them.”
Dr. Lebwohl noted that a “large number of dermatologists have been involved with the development of these drugs and making sure they succeed, and that has also been a pleasure to see.”
Dr. Gottlieb, who Dr. Lebwohl has described as “a superstar” in the fields of dermatology and rheumatology, is one such researcher. In an interview, she looked back on her work and the ways that her work “opened the field,” led to many of her trainees also doing “great work,” and changed the lives of patients.
“It’s nice to feel that I really did change, fundamentally, how psoriasis patients are treated,” said Dr. Gottlieb, who is a clinical professor in the department of dermatology at the Icahn School of Medicine at Mount Sinai. “That obviously feels great.”
She recalled a patient – “a 6-foot-5 biker with bad psoriasis” – who “literally, the minute the door closed, he was crying about how horrible his disease was.”
“And I cleared him ... and then you get big hugs – it just feels extremely good ... giving somebody their life back,” she said.
Dr. Gottlieb has been involved in much of the work in developing biologics for psoriasis, including the ongoing work with bimekizumab for PsA as mentioned by Dr. Lebwohl.
If the phase 2 data with bimekizumab are replicated in the ongoing phase 3 trials now underway at her center, “that can really raise the bar ... so if it’s reproducible, it’s very exciting.”
“It’s exciting to have an IL-23 blocker that, at least in clinical trials, showed inhibition of radiographic progression [in PsA],” she said. “That’s guselkumab those data are already out, and I was involved with that.”
The early work of Dr. Gottlieb and others has also “spread to other diseases,” like hidradenitis suppurativa and atopic dermatitis, she said, noting that numerous studies are underway.
Aside from curing all patients, her ultimate goal is getting to a point where psoriasis has no effect on patients’ quality of life.
“And I see it already,” she said. “It’s happening, and it’s nice to see that it’s happening in children now, too; several of the drugs are approved in kids.”
Alan Menter, MD, chairman of the division of dermatology at Baylor University Medical Center, Dallas, also a prolific researcher – and chair of the guidelines committee that published two new sets of guidelines for psoriasis treatment in 2019 – said that the field of dermatology was “late to the biologic evolution,” as many of the early biologics were first approved for PsA.
“But over the last 10 years, things have changed dramatically,” he said. “After that we suddenly leapt ahead of everybody. ... We now have 11 biologic drugs approved for psoriasis, which is more than any other disease has available.”
It’s been “highly exciting” to see this “evolution and revolution,” he commented, adding that one of the next challenges is to address the comorbidities, such as cardiovascular disease, associated with psoriasis.
“The big question now ... is if you improve skin and you improve joints, can you potentially reduce the risk of coronary artery disease,” he said. “Everybody is looking at that, and to me it’s one of the most exciting things that we’re doing.”
Work is ongoing to look at whether the IL-17s and IL-23s have “other indications outside of the skin and joints,” both within and outside of dermatology.
Like Dr. Gottlieb, Dr. Menter also mentioned the potential for hidradenitis suppurativa, and also for a condition that is rarely discussed or studied: genital psoriasis. Ixekizumab has recently been shown to work in about 75% of patients with genital psoriasis, he noted.
Another important area of research is the identification of biomarkers for predicting response and relapse, he said. For now, biomarker research has disappointed, he added, predicting that it will take at least 3-5 years before biomarkers to help guide treatment are identified.
Indeed, Dr. Gelfand, who also is director of the Psoriasis and Phototherapy Treatment Center, vice chair of clinical research, and medical director of the dermatology clinical studies unit at the University of Pennsylvania, agreed there is a need for research to improve treatment selection.
Advances are being made in genetics – with more than 80 different genes now identified as being related to psoriasis – and in medical informatics – which allow thousands of patients to be followed for years, he said, noting that this could elucidate immunopathological features that can improve treatments, predict and prevent comorbidity, and further improve outcomes.
“We also need care that is more patient centered,” he said, describing the ongoing pragmatic LITE trial of home- or office-based phototherapy for which he is the lead investigator, and other studies that he hopes will expand access to care.
Kenneth Brian Gordon, MD, chair and professor of dermatology at the Medical College of Wisconsin, Milwaukee, whose career started in the basic science immunology arena, added the need for expanding benefit to patients with more-moderate disease. Like Dr. Menter, he identified psoriasis as the area in medicine that has had the greatest degree of advancement, except perhaps for hepatitis C.
He described the process not as a “bench-to-bedside” story, but as a bedside-to-bench, then “back-to-bedside” story.
It was really about taking those early T-cell–targeted biologics and anti-TNF agents from bedside to bench with the realization of the importance of the IL-23 and IL-17 pathways, and that understanding led back to the bedside with the development of the newest agents – and to a “huge difference in patient’s lives.”
“But we’ve gotten so good at treating patients with severe disease ... the question now is how to take care of those with more-moderate disease,” he said, noting that a focus on cost and better delivery systems will be needed for that population.
That research is underway, and the future looks bright – and clear.
“I think with psoriasis therapy and where we’ve come in the last 20 years ... we have a hard time remembering what it was like before we had biologic agents” he said. “Our perspective has changed a lot, and sometimes we forget that.”
In fact, “psoriasis has sort of dragged dermatology into the world of modern clinical trial science, and we can now apply that to all sorts of other diseases,” he said. “The psoriasis trials were the first really well-done large-scale trials in dermatology, and I think that has given dermatology a real leg up in how we do clinical research and how we do evidence-based medicine.”
All of the doctors interviewed for this story have received funds and/or honoraria from, consulted with, are employed with, or served on the advisory boards of manufacturers of biologics. Dr. Gelfand is a copatent holder of resiquimod for treatment of cutaneous T-cell lymphoma and is deputy editor of the Journal of Investigative Dermatology.
Imagine a patient suffering with horrible psoriasis for decades having failed “every available treatment.” Imagine him living all that time with “flaking, cracking, painful, itchy skin,” only to develop cirrhosis after exposure to toxic therapies.
Then imagine the experience for that patient when, 2 weeks after initiating treatment with a new interleukin-17 inhibitor, his skin clears completely.
“Two weeks later it’s all gone – it was a moment to behold,” said Joel M. Gelfand, MD, professor of dermatology and epidemiology at the University of Pennsylvania, Philadelphia, who had cared for the man for many years before a psoriasis treatment revolution of sorts took the field of dermatology by storm.
“The progress has been breathtaking – there’s no other way to describe it – and it feels like a miracle every time I see a new patient who has tough disease and I have all these things to offer them,” he continued. “For most patients, I can really help them and make a major difference in their life.”
said Mark Lebwohl, MD, Waldman professor of dermatology and chair of the Kimberly and Eric J. Waldman department of dermatology at the Icahn School of Medicine at Mount Sinai, New York.
Dr. Lebwohl recounted some of his own experiences with psoriasis patients before the advent of treatments – particularly biologics – that have transformed practice.
There was a time when psoriasis patients had little more to turn to than the effective – but “disgusting” – Goeckerman Regimen involving cycles of UVB light exposure and topical crude coal tar application. Initially, the regimen, which was introduced in the 1920s, was used around the clock on an inpatient basis until the skin cleared, Dr. Lebwohl said.
In the 1970s, the immunosuppressive chemotherapy drug methotrexate became the first oral systemic therapy approved for severe psoriasis. For those with disabling disease, it offered some hope for relief, but only about 40% of patients achieved at least a 75% reduction in the Psoriasis Area and Severity Index score (PASI 75), he said, adding that they did so at the expense of the liver and bone marrow. “But it was the only thing we had for severe psoriasis other than light treatments.”
In the 1980s and 1990s, oral retinoids emerged as a treatment for psoriasis, and the immunosuppressive drug cyclosporine used to prevent organ rejection in some transplant patients was found to clear psoriasis in affected transplant recipients. Although they brought relief to some patients with severe, disabling disease, these also came with a high price. “It’s not that effective, and it has lots of side effects ... and causes kidney damage in essentially 100% of patients,” Dr. Lebwohl said of cyclosporine.
“So we had treatments that worked, but because the side effects were sufficiently severe, a lot of patients were not treated,” he said.
Enter the biologics era
The early 2000s brought the first two approvals for psoriasis: alefacept (Amevive), a “modestly effective, but quite safe” immunosuppressive dimeric fusion protein approved in early 2003 for moderate to severe plaque psoriasis, and efalizumab (Raptiva), a recombinant humanized monoclonal antibody approved in October 2003; both were T-cell–targeted therapies. The former was withdrawn from the market voluntarily as newer agents became available, and the latter was withdrawn in 2009 because of a link with development of progressive multifocal leukoencephalopathy.
Tumor necrosis factor (TNF) blockers, which had been used effectively for RA and Crohn’s disease, emerged next, and were highly effective, much safer than the systemic treatments, and gained “very widespread use,” Dr. Lebwohl said.
His colleague Alice B. Gottlieb, MD, PhD, was among the pioneers in the development of TNF blockers for the treatment of psoriasis. Her seminal, investigator-initiated paper on the efficacy and safety of infliximab (Remicade) monotherapy for plaque-type psoriasis published in the Lancet in 2001 helped launch the current era in which many psoriasis patients achieve 100% PASI responses with limited side effects, he said, explaining that subsequent research elucidated the role of IL-12 and -23 – leading to effective treatments like ustekinumab (Stelara), and later IL-17, which is, “in fact, the molecule closest to the pathogenesis of psoriasis.”
“If you block IL-17, you get rid of psoriasis,” he said, noting that there are now several companies with approved antibodies to IL-17. “Taltz [ixekizumab] and Cosentyx [secukinumab] are the leading ones, and Siliq [brodalumab] blocks the receptor for IL-17, so it is very effective.”
Another novel biologic – bimekizumab – is on the horizon. It blocks both IL-17a and IL-17f, and appears highly effective in psoriasis and psoriatic arthritis (PsA). “Biologics were the real start of the [psoriasis treatment] revolution,” he said. “When I started out I would speak at patient meetings and the patients were angry at their physicians; they thought they weren’t aggressive enough, they were very frustrated.”
Dr. Lebwohl described patients he would see at annual National Psoriasis Foundation meetings: “There were patients in wheel chairs, because they couldn’t walk. They would be red and scaly all over ... you could have literally swept up scale like it was snow after one of those meetings.
“You go forward to around 2010 – nobody’s in wheelchairs anymore, everybody has clear skin, and it’s become a party; patients are no longer angry – they are thrilled with the results they are getting from much safer and much more effective drugs,” he said. “So it’s been a pleasure taking care of those patients and going from a very difficult time of treating them, to a time where we’ve done a great job treating them.”
Dr. Lebwohl noted that a “large number of dermatologists have been involved with the development of these drugs and making sure they succeed, and that has also been a pleasure to see.”
Dr. Gottlieb, who Dr. Lebwohl has described as “a superstar” in the fields of dermatology and rheumatology, is one such researcher. In an interview, she looked back on her work and the ways that her work “opened the field,” led to many of her trainees also doing “great work,” and changed the lives of patients.
“It’s nice to feel that I really did change, fundamentally, how psoriasis patients are treated,” said Dr. Gottlieb, who is a clinical professor in the department of dermatology at the Icahn School of Medicine at Mount Sinai. “That obviously feels great.”
She recalled a patient – “a 6-foot-5 biker with bad psoriasis” – who “literally, the minute the door closed, he was crying about how horrible his disease was.”
“And I cleared him ... and then you get big hugs – it just feels extremely good ... giving somebody their life back,” she said.
Dr. Gottlieb has been involved in much of the work in developing biologics for psoriasis, including the ongoing work with bimekizumab for PsA as mentioned by Dr. Lebwohl.
If the phase 2 data with bimekizumab are replicated in the ongoing phase 3 trials now underway at her center, “that can really raise the bar ... so if it’s reproducible, it’s very exciting.”
“It’s exciting to have an IL-23 blocker that, at least in clinical trials, showed inhibition of radiographic progression [in PsA],” she said. “That’s guselkumab those data are already out, and I was involved with that.”
The early work of Dr. Gottlieb and others has also “spread to other diseases,” like hidradenitis suppurativa and atopic dermatitis, she said, noting that numerous studies are underway.
Aside from curing all patients, her ultimate goal is getting to a point where psoriasis has no effect on patients’ quality of life.
“And I see it already,” she said. “It’s happening, and it’s nice to see that it’s happening in children now, too; several of the drugs are approved in kids.”
Alan Menter, MD, chairman of the division of dermatology at Baylor University Medical Center, Dallas, also a prolific researcher – and chair of the guidelines committee that published two new sets of guidelines for psoriasis treatment in 2019 – said that the field of dermatology was “late to the biologic evolution,” as many of the early biologics were first approved for PsA.
“But over the last 10 years, things have changed dramatically,” he said. “After that we suddenly leapt ahead of everybody. ... We now have 11 biologic drugs approved for psoriasis, which is more than any other disease has available.”
It’s been “highly exciting” to see this “evolution and revolution,” he commented, adding that one of the next challenges is to address the comorbidities, such as cardiovascular disease, associated with psoriasis.
“The big question now ... is if you improve skin and you improve joints, can you potentially reduce the risk of coronary artery disease,” he said. “Everybody is looking at that, and to me it’s one of the most exciting things that we’re doing.”
Work is ongoing to look at whether the IL-17s and IL-23s have “other indications outside of the skin and joints,” both within and outside of dermatology.
Like Dr. Gottlieb, Dr. Menter also mentioned the potential for hidradenitis suppurativa, and also for a condition that is rarely discussed or studied: genital psoriasis. Ixekizumab has recently been shown to work in about 75% of patients with genital psoriasis, he noted.
Another important area of research is the identification of biomarkers for predicting response and relapse, he said. For now, biomarker research has disappointed, he added, predicting that it will take at least 3-5 years before biomarkers to help guide treatment are identified.
Indeed, Dr. Gelfand, who also is director of the Psoriasis and Phototherapy Treatment Center, vice chair of clinical research, and medical director of the dermatology clinical studies unit at the University of Pennsylvania, agreed there is a need for research to improve treatment selection.
Advances are being made in genetics – with more than 80 different genes now identified as being related to psoriasis – and in medical informatics – which allow thousands of patients to be followed for years, he said, noting that this could elucidate immunopathological features that can improve treatments, predict and prevent comorbidity, and further improve outcomes.
“We also need care that is more patient centered,” he said, describing the ongoing pragmatic LITE trial of home- or office-based phototherapy for which he is the lead investigator, and other studies that he hopes will expand access to care.
Kenneth Brian Gordon, MD, chair and professor of dermatology at the Medical College of Wisconsin, Milwaukee, whose career started in the basic science immunology arena, added the need for expanding benefit to patients with more-moderate disease. Like Dr. Menter, he identified psoriasis as the area in medicine that has had the greatest degree of advancement, except perhaps for hepatitis C.
He described the process not as a “bench-to-bedside” story, but as a bedside-to-bench, then “back-to-bedside” story.
It was really about taking those early T-cell–targeted biologics and anti-TNF agents from bedside to bench with the realization of the importance of the IL-23 and IL-17 pathways, and that understanding led back to the bedside with the development of the newest agents – and to a “huge difference in patient’s lives.”
“But we’ve gotten so good at treating patients with severe disease ... the question now is how to take care of those with more-moderate disease,” he said, noting that a focus on cost and better delivery systems will be needed for that population.
That research is underway, and the future looks bright – and clear.
“I think with psoriasis therapy and where we’ve come in the last 20 years ... we have a hard time remembering what it was like before we had biologic agents” he said. “Our perspective has changed a lot, and sometimes we forget that.”
In fact, “psoriasis has sort of dragged dermatology into the world of modern clinical trial science, and we can now apply that to all sorts of other diseases,” he said. “The psoriasis trials were the first really well-done large-scale trials in dermatology, and I think that has given dermatology a real leg up in how we do clinical research and how we do evidence-based medicine.”
All of the doctors interviewed for this story have received funds and/or honoraria from, consulted with, are employed with, or served on the advisory boards of manufacturers of biologics. Dr. Gelfand is a copatent holder of resiquimod for treatment of cutaneous T-cell lymphoma and is deputy editor of the Journal of Investigative Dermatology.
Dual immunotherapy goes the distance in MSI-H colorectal cancer
SAN FRANCISCO – First-line dual immune checkpoint inhibitor therapy for microsatellite instability–high/DNA mismatch repair–deficient (MSI-H/dMMR) metastatic colorectal cancer has impressive durability, an update of the multicohort CheckMate 142 trial shows.
“We all know that MSI-H colorectal cancer patients have a poor prognosis,” said lead investigator Heinz-Josef Lenz, MD, codirector of the Colorectal Center and section head of the GI oncology program at the University of Southern California Norris Comprehensive Cancer Center, Los Angeles. First-line chemotherapy in this population yields median overall survival on the order of 20-22 months.
The cohort of 45 patients treated on the phase 2 trial received the programmed death–1 inhibitor nivolumab (Opdivo) every 2 weeks, plus a low dose of the CTLA4 inhibitor ipilimumab (Yervoy) every 6 weeks as first-line therapy for MSI-H metastatic colorectal cancer. (Nivolumab, with or without ipilimumab, has received Food and Drug Administration accelerated approval as second-line therapy based on data from other cohorts in the trial.)
Previously reported initial results, at a median follow-up of 13.8 months, showed that the investigator-assessed objective response rate was 60% (ESMO 2018, Abstract LBA18_PR). As of the update, now at a median follow-up of 19.9 months, that rate was 64%, according to data reported at the 2020 GI Cancers Symposium.
“Nivolumab and ipilimumab demonstrates clinically meaningful durable benefits and may present an option for first-line treatment for MSI-H metastatic colorectal cancer patients,” Dr. Lenz summarized.
“The incredible complete response rates and overall response rates seen are never seen with chemotherapy, and it’s very well tolerated, so if I have a choice, I would start with nivolumab-ipilimumab in first-line MSI-H,” he added. “The [National Comprehensive Cancer Network] guidelines recommend that you can start in first line with this combination if patients are not candidates for chemotherapy. So they leave this door open, that you have an opportunity to use immunotherapy in this patient population.”
Choosing single or dual immunotherapy
Given the so-called financial toxicity of dual nivolumab and ipilimumab therapy and lack of a randomized comparison against single-agent nivolumab, a session attendee said, can clinicians simply use the latter?
“Very importantly, in this clinical trial, ipilimumab was given every 6 weeks. As you know, the 2-week regimens are significantly more toxic,” Dr. Lenz noted. The combination using this low dose has safety similar to that seen with nivolumab alone. And in cross-trial comparisons, at least, “the combination seems to be a little bit more active, so I always choose both,” he said, adding that the financial aspects are beyond his purview.
However, session cochair Joseph J.Y. Sung, MD, PhD, MBBS, the Mok Hing Yiu Professor of Medicine in the department of medicine and therapeutics at the Chinese University of Hong Kong, had a different view.
“I think there is still room for a randomized study to prove the combination’s superiority against single immunotherapy,” he said in an interview. “If I’m treating an MSI patient, I would stick to single immunotherapy until I see more evidence that this combination has a substantial improvement in the outcome that is worth the money.”
Study details
The patients studied were unselected for baseline tumor expression of programmed death–ligand 1, with 27% having expression of 1% or greater and 58% having expression below that threshold.
As of the data cutoff for the update, 47% of patients were still on treatment, Dr. Lenz reported at the symposium, sponsored by the American Gastroenterological Association, American Society for Clinical Oncology, the American Society for Radiation Oncology, and the Society of Surgical Oncology. The most common reason for stopping treatment was disease progression.
The investigator-assessed objective response rate, the primary endpoint, consisted of complete responses in 9% of patients and partial responses in 56%. (The rate as assessed by blinded independent central reviewers was 58%, consisting of complete responses in 18% and partial responses in 40%.)
“The high response rate was consistent among all evaluated subgroups,” Dr. Lenz noted, with no differences detected by presence or absence of Lynch syndrome and/or specific microsatellite-related mutations.
Median time to response was 2.6 months, and median duration of response was not reached. Median progression-free and overall survival were also not reached, but 15-month rates were 75% and 84%, respectively.
The rate of grade 3-4 treatment-related adverse events was 20%, and the rate of grade 3-4 serious treatment-related adverse events was 11%. Only 4% of patients discontinued treatment because of such events.
Ongoing trials may take immunotherapy even further in this patient population, according to Dr. Lenz. “We are all waiting for the treatments with the immunotherapy in combination with chemo and bevacizumab in first line. Will that change the efficacy and toxicity?” he elaborated. “You can imagine, in the MSI-H patients with the immunotherapy alone, we haven’t reached median progression-free and overall survival, so it may take a long time to get this data.”
Dr. Lenz reported receiving honoraria from Bayer, Boehringer Ingelheim, Merck Serono, and Roche; consulting or advising with Bayer, Merck Serono, Pfizer, and Roche; receiving travel expenses from Bayer, Merck Serono, and Roche. The trial was funded by Bristol-Myers Squibb. Dr. Sung reported that he had no relevant conflicts of interest.
SOURCE: Lenz H-J et al. 2020 GI Cancers Symposium, Abstract 11.
SAN FRANCISCO – First-line dual immune checkpoint inhibitor therapy for microsatellite instability–high/DNA mismatch repair–deficient (MSI-H/dMMR) metastatic colorectal cancer has impressive durability, an update of the multicohort CheckMate 142 trial shows.
“We all know that MSI-H colorectal cancer patients have a poor prognosis,” said lead investigator Heinz-Josef Lenz, MD, codirector of the Colorectal Center and section head of the GI oncology program at the University of Southern California Norris Comprehensive Cancer Center, Los Angeles. First-line chemotherapy in this population yields median overall survival on the order of 20-22 months.
The cohort of 45 patients treated on the phase 2 trial received the programmed death–1 inhibitor nivolumab (Opdivo) every 2 weeks, plus a low dose of the CTLA4 inhibitor ipilimumab (Yervoy) every 6 weeks as first-line therapy for MSI-H metastatic colorectal cancer. (Nivolumab, with or without ipilimumab, has received Food and Drug Administration accelerated approval as second-line therapy based on data from other cohorts in the trial.)
Previously reported initial results, at a median follow-up of 13.8 months, showed that the investigator-assessed objective response rate was 60% (ESMO 2018, Abstract LBA18_PR). As of the update, now at a median follow-up of 19.9 months, that rate was 64%, according to data reported at the 2020 GI Cancers Symposium.
“Nivolumab and ipilimumab demonstrates clinically meaningful durable benefits and may present an option for first-line treatment for MSI-H metastatic colorectal cancer patients,” Dr. Lenz summarized.
“The incredible complete response rates and overall response rates seen are never seen with chemotherapy, and it’s very well tolerated, so if I have a choice, I would start with nivolumab-ipilimumab in first-line MSI-H,” he added. “The [National Comprehensive Cancer Network] guidelines recommend that you can start in first line with this combination if patients are not candidates for chemotherapy. So they leave this door open, that you have an opportunity to use immunotherapy in this patient population.”
Choosing single or dual immunotherapy
Given the so-called financial toxicity of dual nivolumab and ipilimumab therapy and lack of a randomized comparison against single-agent nivolumab, a session attendee said, can clinicians simply use the latter?
“Very importantly, in this clinical trial, ipilimumab was given every 6 weeks. As you know, the 2-week regimens are significantly more toxic,” Dr. Lenz noted. The combination using this low dose has safety similar to that seen with nivolumab alone. And in cross-trial comparisons, at least, “the combination seems to be a little bit more active, so I always choose both,” he said, adding that the financial aspects are beyond his purview.
However, session cochair Joseph J.Y. Sung, MD, PhD, MBBS, the Mok Hing Yiu Professor of Medicine in the department of medicine and therapeutics at the Chinese University of Hong Kong, had a different view.
“I think there is still room for a randomized study to prove the combination’s superiority against single immunotherapy,” he said in an interview. “If I’m treating an MSI patient, I would stick to single immunotherapy until I see more evidence that this combination has a substantial improvement in the outcome that is worth the money.”
Study details
The patients studied were unselected for baseline tumor expression of programmed death–ligand 1, with 27% having expression of 1% or greater and 58% having expression below that threshold.
As of the data cutoff for the update, 47% of patients were still on treatment, Dr. Lenz reported at the symposium, sponsored by the American Gastroenterological Association, American Society for Clinical Oncology, the American Society for Radiation Oncology, and the Society of Surgical Oncology. The most common reason for stopping treatment was disease progression.
The investigator-assessed objective response rate, the primary endpoint, consisted of complete responses in 9% of patients and partial responses in 56%. (The rate as assessed by blinded independent central reviewers was 58%, consisting of complete responses in 18% and partial responses in 40%.)
“The high response rate was consistent among all evaluated subgroups,” Dr. Lenz noted, with no differences detected by presence or absence of Lynch syndrome and/or specific microsatellite-related mutations.
Median time to response was 2.6 months, and median duration of response was not reached. Median progression-free and overall survival were also not reached, but 15-month rates were 75% and 84%, respectively.
The rate of grade 3-4 treatment-related adverse events was 20%, and the rate of grade 3-4 serious treatment-related adverse events was 11%. Only 4% of patients discontinued treatment because of such events.
Ongoing trials may take immunotherapy even further in this patient population, according to Dr. Lenz. “We are all waiting for the treatments with the immunotherapy in combination with chemo and bevacizumab in first line. Will that change the efficacy and toxicity?” he elaborated. “You can imagine, in the MSI-H patients with the immunotherapy alone, we haven’t reached median progression-free and overall survival, so it may take a long time to get this data.”
Dr. Lenz reported receiving honoraria from Bayer, Boehringer Ingelheim, Merck Serono, and Roche; consulting or advising with Bayer, Merck Serono, Pfizer, and Roche; receiving travel expenses from Bayer, Merck Serono, and Roche. The trial was funded by Bristol-Myers Squibb. Dr. Sung reported that he had no relevant conflicts of interest.
SOURCE: Lenz H-J et al. 2020 GI Cancers Symposium, Abstract 11.
SAN FRANCISCO – First-line dual immune checkpoint inhibitor therapy for microsatellite instability–high/DNA mismatch repair–deficient (MSI-H/dMMR) metastatic colorectal cancer has impressive durability, an update of the multicohort CheckMate 142 trial shows.
“We all know that MSI-H colorectal cancer patients have a poor prognosis,” said lead investigator Heinz-Josef Lenz, MD, codirector of the Colorectal Center and section head of the GI oncology program at the University of Southern California Norris Comprehensive Cancer Center, Los Angeles. First-line chemotherapy in this population yields median overall survival on the order of 20-22 months.
The cohort of 45 patients treated on the phase 2 trial received the programmed death–1 inhibitor nivolumab (Opdivo) every 2 weeks, plus a low dose of the CTLA4 inhibitor ipilimumab (Yervoy) every 6 weeks as first-line therapy for MSI-H metastatic colorectal cancer. (Nivolumab, with or without ipilimumab, has received Food and Drug Administration accelerated approval as second-line therapy based on data from other cohorts in the trial.)
Previously reported initial results, at a median follow-up of 13.8 months, showed that the investigator-assessed objective response rate was 60% (ESMO 2018, Abstract LBA18_PR). As of the update, now at a median follow-up of 19.9 months, that rate was 64%, according to data reported at the 2020 GI Cancers Symposium.
“Nivolumab and ipilimumab demonstrates clinically meaningful durable benefits and may present an option for first-line treatment for MSI-H metastatic colorectal cancer patients,” Dr. Lenz summarized.
“The incredible complete response rates and overall response rates seen are never seen with chemotherapy, and it’s very well tolerated, so if I have a choice, I would start with nivolumab-ipilimumab in first-line MSI-H,” he added. “The [National Comprehensive Cancer Network] guidelines recommend that you can start in first line with this combination if patients are not candidates for chemotherapy. So they leave this door open, that you have an opportunity to use immunotherapy in this patient population.”
Choosing single or dual immunotherapy
Given the so-called financial toxicity of dual nivolumab and ipilimumab therapy and lack of a randomized comparison against single-agent nivolumab, a session attendee said, can clinicians simply use the latter?
“Very importantly, in this clinical trial, ipilimumab was given every 6 weeks. As you know, the 2-week regimens are significantly more toxic,” Dr. Lenz noted. The combination using this low dose has safety similar to that seen with nivolumab alone. And in cross-trial comparisons, at least, “the combination seems to be a little bit more active, so I always choose both,” he said, adding that the financial aspects are beyond his purview.
However, session cochair Joseph J.Y. Sung, MD, PhD, MBBS, the Mok Hing Yiu Professor of Medicine in the department of medicine and therapeutics at the Chinese University of Hong Kong, had a different view.
“I think there is still room for a randomized study to prove the combination’s superiority against single immunotherapy,” he said in an interview. “If I’m treating an MSI patient, I would stick to single immunotherapy until I see more evidence that this combination has a substantial improvement in the outcome that is worth the money.”
Study details
The patients studied were unselected for baseline tumor expression of programmed death–ligand 1, with 27% having expression of 1% or greater and 58% having expression below that threshold.
As of the data cutoff for the update, 47% of patients were still on treatment, Dr. Lenz reported at the symposium, sponsored by the American Gastroenterological Association, American Society for Clinical Oncology, the American Society for Radiation Oncology, and the Society of Surgical Oncology. The most common reason for stopping treatment was disease progression.
The investigator-assessed objective response rate, the primary endpoint, consisted of complete responses in 9% of patients and partial responses in 56%. (The rate as assessed by blinded independent central reviewers was 58%, consisting of complete responses in 18% and partial responses in 40%.)
“The high response rate was consistent among all evaluated subgroups,” Dr. Lenz noted, with no differences detected by presence or absence of Lynch syndrome and/or specific microsatellite-related mutations.
Median time to response was 2.6 months, and median duration of response was not reached. Median progression-free and overall survival were also not reached, but 15-month rates were 75% and 84%, respectively.
The rate of grade 3-4 treatment-related adverse events was 20%, and the rate of grade 3-4 serious treatment-related adverse events was 11%. Only 4% of patients discontinued treatment because of such events.
Ongoing trials may take immunotherapy even further in this patient population, according to Dr. Lenz. “We are all waiting for the treatments with the immunotherapy in combination with chemo and bevacizumab in first line. Will that change the efficacy and toxicity?” he elaborated. “You can imagine, in the MSI-H patients with the immunotherapy alone, we haven’t reached median progression-free and overall survival, so it may take a long time to get this data.”
Dr. Lenz reported receiving honoraria from Bayer, Boehringer Ingelheim, Merck Serono, and Roche; consulting or advising with Bayer, Merck Serono, Pfizer, and Roche; receiving travel expenses from Bayer, Merck Serono, and Roche. The trial was funded by Bristol-Myers Squibb. Dr. Sung reported that he had no relevant conflicts of interest.
SOURCE: Lenz H-J et al. 2020 GI Cancers Symposium, Abstract 11.
REPORTING FROM THE 2020 GI CANCERS SYMPOSIUM
What 2019’s top five CAD trials tell us
SNOWMASS, COLO. – A repeated theme threading through much of one prominent interventional cardiologist’s personal list of the top five coronary artery disease (CAD) trials of the past year is that aspirin is very often more trouble than it’s worth.
“For some years I’ve been concerned that the only thing that aspirin does [in patients after percutaneous coronary intervention] is increase your risk of bleeding. It doesn’t really provide any additional ischemic protection,” Malcolm R. Bell, MBBS, said at the annual Cardiovascular Conference at Snowmass sponsored by the American College of Cardiology.
“I’ll remind you that, when we go back to the early stent days, observed Dr. Bell, professor of medicine and vice chair of the department of cardiovascular medicine at the Mayo Clinic in Rochester, Minn.
Here are the key takeaway messages from his five most important randomized trials in CAD during the last year.
AUGUSTUS
For years, cardiologists have grappled with how to best manage high-cardiovascular-risk patients with atrial fibrillation who seem like they might benefit from triple-antithrombotic therapy. AUGUSTUS supplied the answer: Don’t do it. Skip the aspirin and turn instead to a P2Y12 inhibitor plus a non–vitamin K antagonist oral anticoagulant (NOAC), rather than warfarin.
“I would like you to think of triple therapy as a triple threat. That’s really what triple therapy is all about”– a three-pronged threat to patient safety, Dr. Bell commented.
In AUGUSTUS, 4,614 patients with atrial fibrillation and CAD with an acute coronary syndrome (ACS) and/or percutaneous coronary intervention (PCI) in 33 countries were placed on a P2Y12 inhibitor – most often clopidogrel – and randomized double blind to either apixaban (Eliquis) or warfarin, and further to aspirin or placebo, for 6 months of antithrombotic therapy. The strategy of a P2Y12 inhibitor and apixaban without aspirin was the clear winner, resulting in significantly less major bleeding, mortality, and hospitalizations than treatment with a P2Y12 inhibitor and warfarin, with or without aspirin. Most importantly, ischemic event rates didn’t differ between the apixaban and warfarin groups. And patients randomized to aspirin had rates of ischemic events and death or hospitalization similar to placebo-treated controls, meaning aspirin accomplished nothing (N Engl J Med. 2019 Apr 18;380[16]:1509-24).
Dr. Bell noted that a meta-analysis of AUGUSTUS and three smaller randomized trials including more than 10,000 AUGUSTUS-type patients with atrial fibrillation concluded that a treatment strategy utilizing a NOAC and a P2Y12 inhibitor resulted in less bleeding than warfarin plus DAPT, and at no cost in terms of excess ischemic events. Moreover, regimens without aspirin resulted in less intracranial and other major bleeding without any difference in major adverse cardiovascular events (JAMA Cardiol. 2019 Jun 19. doi: 10.1001/jamacardio.2019.1880).
A key message of these four trials is that a NOAC is preferable to warfarin, so much so that, in high-risk patients who are already on warfarin, it’s worth considering a switch to a NOAC.
“And we should really be avoiding DAPT,” Dr. Bell added.
How soon after an ACS and/or PCI should patients with atrial fibrillation stop taking aspirin?
“In AUGUSTUS, randomization occurred at a median of 6 days, so we know that half the patients stopped their aspirin by then. In our own practice, we’re just dropping the aspirin for the most part before the patient leaves the hospital. I think if you leave them with instructions to stop the aspirin in a week’s time or a month’s time it just leads to confusion. And we should also remember that half of the major bleeding after PCI or ACS happens in the first 30 days, so it doesn’t make a lot of sense to say that we should continue it for a month and then drop it,” according to the cardiologist.
SMART-CHOICE and STOPDAPT-2
These two large multicenter studies demonstrate that DAPT can safely be stopped early if needed. SMART-CHOICE from South Korea and STOPDAPT-2 from Japan each randomized roughly 3,000 patients undergoing PCI to 12 months of DAPT or to DAPT for only 3 months or 1 month, respectively, at which point the aspirin was dropped and patients in the abbreviated DAPT arm continued on P2Y12 inhibitor monotherapy, mostly clopidogrel, for the remainder of the 12 months. In the Japanese STOPDAPT-2 trial, 1 month of DAPT proved superior to 12 months of DAPT for the primary composite endpoint of cardiovascular death, MI, stroke, definite stent thrombosis, or major or minor bleeding at 12 months (JAMA. 2019 Jun 25;321[24]:2414-27). In the South Korean SMART-CHOICE trial, 3 months of DAPT was noninferior to 12 months for major adverse cardiac and cerebrovascular events, and superior in terms of bleeding risk (JAMA. 2019 Jun 25;321[24]:2428-37). Of note, roughly half of patients in the two trials were lower-risk individuals undergoing PCI for stable angina.
Dr. Bell noted that, while the TWILIGHT trial (Ticagrelor With or Without Aspirin in High-Risk Patients After PCI) didn’t make his top-five list, it certainly fits well with the two East Asian studies. The TWILIGHT investigators randomized more than 7,000 patients to 12 months of DAPT or discontinuation of aspirin after 3 months. The result: a lower incidence of clinically relevant bleeding with ticagrelor monotherapy, and with no increased risk of death, MI, or stroke, compared with 12 months of DAPT (N Engl J Med. 2019 Nov 21;381[21]:2032-42).
“Again, I would just question what the added value of aspirin is here,” Dr. Bell commented. “Many interventional cardiologists are absolutely terrified of their patients having stent thrombosis, but with second-generation drug-eluting stents – the stents we’re putting in day in and day out – the risk of stent thrombosis is less than 1%. And in these two trials it was less than 0.5%. There’s more risk of having major bleeding events than there is of ischemia, so I think the balance is in favor of preventing bleeding. We know that major bleeding predicts short- and long-term mortality.”
COLCOT
This double-blind trial randomized 4,745 patients within 30 days post MI to low-dose colchicine or placebo on top of excellent rates of background guideline-directed medical therapy. The goal was to see if this anti-inflammatory agent could reduce cardiovascular events independent of any lipid-lowering effect, as was earlier seen with canakinumab in the CANTOS trial. It did so to a statistically significant but relatively modest degree, with a 5.5% rate of the composite cardiovascular events endpoint in the colchicine group and 7.1% in placebo-treated controls (N Engl J Med. 2019 Dec 26;381[26]:2497-505). But Dr. Bell was unimpressed.
“All-cause mortality was identical at 1.8% in both groups. So colchicine is not saving lives. In fact, the only real differences were in stroke – but the study wasn’t powered to look at stroke – and in urgent hospitalization for angina leading to revascularization, which is a soft endpoint,” he observed.
Plus, 2.5% of patients were lost to follow-up, which Dr. Bell considers “a little concerning” in a trial conducted in the current era.
“In my opinion, the evidence that colchicine is effective is weak, and I don’t think really supports the drug’s routine use post MI. We already send these patients out on numerous medications. We have to think about cost/benefit, and if a patient asks me: ‘Is this going to prevent another heart attack or make me live longer?’ I think the unequivocal answer is no,” he said.
These days colchicine is no longer an inexpensive drug, either, at an average cost of $300-$400 per month, the cardiologist added.
COMPLETE
This study randomized more than 4,000 patients with ST-segment elevation MI (STEMI) and multivessel disease to primary PCI of the culprit lesion only or to staged complete revascularization via PCI of all angiographically significant nonculprit lesions. Complete revascularization proved to be the superior strategy, with a 26% reduction in the risk of the composite of cardiovascular death or MI at a median of 3 years (N Engl J Med. 2019 Oct 10;381[15]:1411-21).
The optimal timing of the staged procedure remains unclear, since the study didn’t specify a protocol.
“I’m still a bit uncomfortable doing multivessel PCI at 2 o’clock in the morning in the setting of STEMI in someone I’ve never met before. I don’t think there’s a rush to do anything then. Often in this middle-of-the-night stuff, we miss things or we overinterpret things. I think it’s better to let the patient cool down, get to know them,” according to Dr. Bell.
EXCEL
Publication of the 5-year outcomes of the largest-ever randomized trial of PCI versus coronary artery bypass grafting (CABG) for left main coronary disease has led to furious controversy, with a few of the surgeons involved in the study opting to publically broadcast allegations of misbehavior on the part of the interventional cardiologist study leadership, charges that have been strongly denied.
The actual results are in line with findings reported from smaller randomized trials. At 5 years in EXCEL, there was no significant difference between the PCI and CABG groups in the primary composite endpoint of death, cerebrovascular accident, or MI (N Engl J Med. 2019 Nov 7;381[19]:1820-30). The all-cause mortality rate was 13% in the PCI arm and 9.9% with CABG, but this finding comes with a caveat.
“I’ll emphasize this trial was never powered to look at mortality. Neither were any of the other randomized trials. On the other hand, I don’t think you can necessarily ignore the finding of an absolute 3.1% difference,” Dr. Bell said.
PCI and CABG are both very good, mature therapies for left main disease, in his view. In the setting of more-complex coronary disease in younger patients, he often views the complete revascularization offered by surgery as the preferred option. On the other hand, in an 80-year-old with severe comorbidities, clearly PCI is attractive.
He considers the highly public nature of this interspecialty spat a regrettable black eye for the entire field of cardiovascular medicine. And he predicted that an ongoing outside neutral-party review of the study data and procedures will conclude, as he has, “there was no malfeasance at all in the trial.”
Dr. Bell reported having no financial conflicts regarding his presentation.
SNOWMASS, COLO. – A repeated theme threading through much of one prominent interventional cardiologist’s personal list of the top five coronary artery disease (CAD) trials of the past year is that aspirin is very often more trouble than it’s worth.
“For some years I’ve been concerned that the only thing that aspirin does [in patients after percutaneous coronary intervention] is increase your risk of bleeding. It doesn’t really provide any additional ischemic protection,” Malcolm R. Bell, MBBS, said at the annual Cardiovascular Conference at Snowmass sponsored by the American College of Cardiology.
“I’ll remind you that, when we go back to the early stent days, observed Dr. Bell, professor of medicine and vice chair of the department of cardiovascular medicine at the Mayo Clinic in Rochester, Minn.
Here are the key takeaway messages from his five most important randomized trials in CAD during the last year.
AUGUSTUS
For years, cardiologists have grappled with how to best manage high-cardiovascular-risk patients with atrial fibrillation who seem like they might benefit from triple-antithrombotic therapy. AUGUSTUS supplied the answer: Don’t do it. Skip the aspirin and turn instead to a P2Y12 inhibitor plus a non–vitamin K antagonist oral anticoagulant (NOAC), rather than warfarin.
“I would like you to think of triple therapy as a triple threat. That’s really what triple therapy is all about”– a three-pronged threat to patient safety, Dr. Bell commented.
In AUGUSTUS, 4,614 patients with atrial fibrillation and CAD with an acute coronary syndrome (ACS) and/or percutaneous coronary intervention (PCI) in 33 countries were placed on a P2Y12 inhibitor – most often clopidogrel – and randomized double blind to either apixaban (Eliquis) or warfarin, and further to aspirin or placebo, for 6 months of antithrombotic therapy. The strategy of a P2Y12 inhibitor and apixaban without aspirin was the clear winner, resulting in significantly less major bleeding, mortality, and hospitalizations than treatment with a P2Y12 inhibitor and warfarin, with or without aspirin. Most importantly, ischemic event rates didn’t differ between the apixaban and warfarin groups. And patients randomized to aspirin had rates of ischemic events and death or hospitalization similar to placebo-treated controls, meaning aspirin accomplished nothing (N Engl J Med. 2019 Apr 18;380[16]:1509-24).
Dr. Bell noted that a meta-analysis of AUGUSTUS and three smaller randomized trials including more than 10,000 AUGUSTUS-type patients with atrial fibrillation concluded that a treatment strategy utilizing a NOAC and a P2Y12 inhibitor resulted in less bleeding than warfarin plus DAPT, and at no cost in terms of excess ischemic events. Moreover, regimens without aspirin resulted in less intracranial and other major bleeding without any difference in major adverse cardiovascular events (JAMA Cardiol. 2019 Jun 19. doi: 10.1001/jamacardio.2019.1880).
A key message of these four trials is that a NOAC is preferable to warfarin, so much so that, in high-risk patients who are already on warfarin, it’s worth considering a switch to a NOAC.
“And we should really be avoiding DAPT,” Dr. Bell added.
How soon after an ACS and/or PCI should patients with atrial fibrillation stop taking aspirin?
“In AUGUSTUS, randomization occurred at a median of 6 days, so we know that half the patients stopped their aspirin by then. In our own practice, we’re just dropping the aspirin for the most part before the patient leaves the hospital. I think if you leave them with instructions to stop the aspirin in a week’s time or a month’s time it just leads to confusion. And we should also remember that half of the major bleeding after PCI or ACS happens in the first 30 days, so it doesn’t make a lot of sense to say that we should continue it for a month and then drop it,” according to the cardiologist.
SMART-CHOICE and STOPDAPT-2
These two large multicenter studies demonstrate that DAPT can safely be stopped early if needed. SMART-CHOICE from South Korea and STOPDAPT-2 from Japan each randomized roughly 3,000 patients undergoing PCI to 12 months of DAPT or to DAPT for only 3 months or 1 month, respectively, at which point the aspirin was dropped and patients in the abbreviated DAPT arm continued on P2Y12 inhibitor monotherapy, mostly clopidogrel, for the remainder of the 12 months. In the Japanese STOPDAPT-2 trial, 1 month of DAPT proved superior to 12 months of DAPT for the primary composite endpoint of cardiovascular death, MI, stroke, definite stent thrombosis, or major or minor bleeding at 12 months (JAMA. 2019 Jun 25;321[24]:2414-27). In the South Korean SMART-CHOICE trial, 3 months of DAPT was noninferior to 12 months for major adverse cardiac and cerebrovascular events, and superior in terms of bleeding risk (JAMA. 2019 Jun 25;321[24]:2428-37). Of note, roughly half of patients in the two trials were lower-risk individuals undergoing PCI for stable angina.
Dr. Bell noted that, while the TWILIGHT trial (Ticagrelor With or Without Aspirin in High-Risk Patients After PCI) didn’t make his top-five list, it certainly fits well with the two East Asian studies. The TWILIGHT investigators randomized more than 7,000 patients to 12 months of DAPT or discontinuation of aspirin after 3 months. The result: a lower incidence of clinically relevant bleeding with ticagrelor monotherapy, and with no increased risk of death, MI, or stroke, compared with 12 months of DAPT (N Engl J Med. 2019 Nov 21;381[21]:2032-42).
“Again, I would just question what the added value of aspirin is here,” Dr. Bell commented. “Many interventional cardiologists are absolutely terrified of their patients having stent thrombosis, but with second-generation drug-eluting stents – the stents we’re putting in day in and day out – the risk of stent thrombosis is less than 1%. And in these two trials it was less than 0.5%. There’s more risk of having major bleeding events than there is of ischemia, so I think the balance is in favor of preventing bleeding. We know that major bleeding predicts short- and long-term mortality.”
COLCOT
This double-blind trial randomized 4,745 patients within 30 days post MI to low-dose colchicine or placebo on top of excellent rates of background guideline-directed medical therapy. The goal was to see if this anti-inflammatory agent could reduce cardiovascular events independent of any lipid-lowering effect, as was earlier seen with canakinumab in the CANTOS trial. It did so to a statistically significant but relatively modest degree, with a 5.5% rate of the composite cardiovascular events endpoint in the colchicine group and 7.1% in placebo-treated controls (N Engl J Med. 2019 Dec 26;381[26]:2497-505). But Dr. Bell was unimpressed.
“All-cause mortality was identical at 1.8% in both groups. So colchicine is not saving lives. In fact, the only real differences were in stroke – but the study wasn’t powered to look at stroke – and in urgent hospitalization for angina leading to revascularization, which is a soft endpoint,” he observed.
Plus, 2.5% of patients were lost to follow-up, which Dr. Bell considers “a little concerning” in a trial conducted in the current era.
“In my opinion, the evidence that colchicine is effective is weak, and I don’t think really supports the drug’s routine use post MI. We already send these patients out on numerous medications. We have to think about cost/benefit, and if a patient asks me: ‘Is this going to prevent another heart attack or make me live longer?’ I think the unequivocal answer is no,” he said.
These days colchicine is no longer an inexpensive drug, either, at an average cost of $300-$400 per month, the cardiologist added.
COMPLETE
This study randomized more than 4,000 patients with ST-segment elevation MI (STEMI) and multivessel disease to primary PCI of the culprit lesion only or to staged complete revascularization via PCI of all angiographically significant nonculprit lesions. Complete revascularization proved to be the superior strategy, with a 26% reduction in the risk of the composite of cardiovascular death or MI at a median of 3 years (N Engl J Med. 2019 Oct 10;381[15]:1411-21).
The optimal timing of the staged procedure remains unclear, since the study didn’t specify a protocol.
“I’m still a bit uncomfortable doing multivessel PCI at 2 o’clock in the morning in the setting of STEMI in someone I’ve never met before. I don’t think there’s a rush to do anything then. Often in this middle-of-the-night stuff, we miss things or we overinterpret things. I think it’s better to let the patient cool down, get to know them,” according to Dr. Bell.
EXCEL
Publication of the 5-year outcomes of the largest-ever randomized trial of PCI versus coronary artery bypass grafting (CABG) for left main coronary disease has led to furious controversy, with a few of the surgeons involved in the study opting to publically broadcast allegations of misbehavior on the part of the interventional cardiologist study leadership, charges that have been strongly denied.
The actual results are in line with findings reported from smaller randomized trials. At 5 years in EXCEL, there was no significant difference between the PCI and CABG groups in the primary composite endpoint of death, cerebrovascular accident, or MI (N Engl J Med. 2019 Nov 7;381[19]:1820-30). The all-cause mortality rate was 13% in the PCI arm and 9.9% with CABG, but this finding comes with a caveat.
“I’ll emphasize this trial was never powered to look at mortality. Neither were any of the other randomized trials. On the other hand, I don’t think you can necessarily ignore the finding of an absolute 3.1% difference,” Dr. Bell said.
PCI and CABG are both very good, mature therapies for left main disease, in his view. In the setting of more-complex coronary disease in younger patients, he often views the complete revascularization offered by surgery as the preferred option. On the other hand, in an 80-year-old with severe comorbidities, clearly PCI is attractive.
He considers the highly public nature of this interspecialty spat a regrettable black eye for the entire field of cardiovascular medicine. And he predicted that an ongoing outside neutral-party review of the study data and procedures will conclude, as he has, “there was no malfeasance at all in the trial.”
Dr. Bell reported having no financial conflicts regarding his presentation.
SNOWMASS, COLO. – A repeated theme threading through much of one prominent interventional cardiologist’s personal list of the top five coronary artery disease (CAD) trials of the past year is that aspirin is very often more trouble than it’s worth.
“For some years I’ve been concerned that the only thing that aspirin does [in patients after percutaneous coronary intervention] is increase your risk of bleeding. It doesn’t really provide any additional ischemic protection,” Malcolm R. Bell, MBBS, said at the annual Cardiovascular Conference at Snowmass sponsored by the American College of Cardiology.
“I’ll remind you that, when we go back to the early stent days, observed Dr. Bell, professor of medicine and vice chair of the department of cardiovascular medicine at the Mayo Clinic in Rochester, Minn.
Here are the key takeaway messages from his five most important randomized trials in CAD during the last year.
AUGUSTUS
For years, cardiologists have grappled with how to best manage high-cardiovascular-risk patients with atrial fibrillation who seem like they might benefit from triple-antithrombotic therapy. AUGUSTUS supplied the answer: Don’t do it. Skip the aspirin and turn instead to a P2Y12 inhibitor plus a non–vitamin K antagonist oral anticoagulant (NOAC), rather than warfarin.
“I would like you to think of triple therapy as a triple threat. That’s really what triple therapy is all about”– a three-pronged threat to patient safety, Dr. Bell commented.
In AUGUSTUS, 4,614 patients with atrial fibrillation and CAD with an acute coronary syndrome (ACS) and/or percutaneous coronary intervention (PCI) in 33 countries were placed on a P2Y12 inhibitor – most often clopidogrel – and randomized double blind to either apixaban (Eliquis) or warfarin, and further to aspirin or placebo, for 6 months of antithrombotic therapy. The strategy of a P2Y12 inhibitor and apixaban without aspirin was the clear winner, resulting in significantly less major bleeding, mortality, and hospitalizations than treatment with a P2Y12 inhibitor and warfarin, with or without aspirin. Most importantly, ischemic event rates didn’t differ between the apixaban and warfarin groups. And patients randomized to aspirin had rates of ischemic events and death or hospitalization similar to placebo-treated controls, meaning aspirin accomplished nothing (N Engl J Med. 2019 Apr 18;380[16]:1509-24).
Dr. Bell noted that a meta-analysis of AUGUSTUS and three smaller randomized trials including more than 10,000 AUGUSTUS-type patients with atrial fibrillation concluded that a treatment strategy utilizing a NOAC and a P2Y12 inhibitor resulted in less bleeding than warfarin plus DAPT, and at no cost in terms of excess ischemic events. Moreover, regimens without aspirin resulted in less intracranial and other major bleeding without any difference in major adverse cardiovascular events (JAMA Cardiol. 2019 Jun 19. doi: 10.1001/jamacardio.2019.1880).
A key message of these four trials is that a NOAC is preferable to warfarin, so much so that, in high-risk patients who are already on warfarin, it’s worth considering a switch to a NOAC.
“And we should really be avoiding DAPT,” Dr. Bell added.
How soon after an ACS and/or PCI should patients with atrial fibrillation stop taking aspirin?
“In AUGUSTUS, randomization occurred at a median of 6 days, so we know that half the patients stopped their aspirin by then. In our own practice, we’re just dropping the aspirin for the most part before the patient leaves the hospital. I think if you leave them with instructions to stop the aspirin in a week’s time or a month’s time it just leads to confusion. And we should also remember that half of the major bleeding after PCI or ACS happens in the first 30 days, so it doesn’t make a lot of sense to say that we should continue it for a month and then drop it,” according to the cardiologist.
SMART-CHOICE and STOPDAPT-2
These two large multicenter studies demonstrate that DAPT can safely be stopped early if needed. SMART-CHOICE from South Korea and STOPDAPT-2 from Japan each randomized roughly 3,000 patients undergoing PCI to 12 months of DAPT or to DAPT for only 3 months or 1 month, respectively, at which point the aspirin was dropped and patients in the abbreviated DAPT arm continued on P2Y12 inhibitor monotherapy, mostly clopidogrel, for the remainder of the 12 months. In the Japanese STOPDAPT-2 trial, 1 month of DAPT proved superior to 12 months of DAPT for the primary composite endpoint of cardiovascular death, MI, stroke, definite stent thrombosis, or major or minor bleeding at 12 months (JAMA. 2019 Jun 25;321[24]:2414-27). In the South Korean SMART-CHOICE trial, 3 months of DAPT was noninferior to 12 months for major adverse cardiac and cerebrovascular events, and superior in terms of bleeding risk (JAMA. 2019 Jun 25;321[24]:2428-37). Of note, roughly half of patients in the two trials were lower-risk individuals undergoing PCI for stable angina.
Dr. Bell noted that, while the TWILIGHT trial (Ticagrelor With or Without Aspirin in High-Risk Patients After PCI) didn’t make his top-five list, it certainly fits well with the two East Asian studies. The TWILIGHT investigators randomized more than 7,000 patients to 12 months of DAPT or discontinuation of aspirin after 3 months. The result: a lower incidence of clinically relevant bleeding with ticagrelor monotherapy, and with no increased risk of death, MI, or stroke, compared with 12 months of DAPT (N Engl J Med. 2019 Nov 21;381[21]:2032-42).
“Again, I would just question what the added value of aspirin is here,” Dr. Bell commented. “Many interventional cardiologists are absolutely terrified of their patients having stent thrombosis, but with second-generation drug-eluting stents – the stents we’re putting in day in and day out – the risk of stent thrombosis is less than 1%. And in these two trials it was less than 0.5%. There’s more risk of having major bleeding events than there is of ischemia, so I think the balance is in favor of preventing bleeding. We know that major bleeding predicts short- and long-term mortality.”
COLCOT
This double-blind trial randomized 4,745 patients within 30 days post MI to low-dose colchicine or placebo on top of excellent rates of background guideline-directed medical therapy. The goal was to see if this anti-inflammatory agent could reduce cardiovascular events independent of any lipid-lowering effect, as was earlier seen with canakinumab in the CANTOS trial. It did so to a statistically significant but relatively modest degree, with a 5.5% rate of the composite cardiovascular events endpoint in the colchicine group and 7.1% in placebo-treated controls (N Engl J Med. 2019 Dec 26;381[26]:2497-505). But Dr. Bell was unimpressed.
“All-cause mortality was identical at 1.8% in both groups. So colchicine is not saving lives. In fact, the only real differences were in stroke – but the study wasn’t powered to look at stroke – and in urgent hospitalization for angina leading to revascularization, which is a soft endpoint,” he observed.
Plus, 2.5% of patients were lost to follow-up, which Dr. Bell considers “a little concerning” in a trial conducted in the current era.
“In my opinion, the evidence that colchicine is effective is weak, and I don’t think really supports the drug’s routine use post MI. We already send these patients out on numerous medications. We have to think about cost/benefit, and if a patient asks me: ‘Is this going to prevent another heart attack or make me live longer?’ I think the unequivocal answer is no,” he said.
These days colchicine is no longer an inexpensive drug, either, at an average cost of $300-$400 per month, the cardiologist added.
COMPLETE
This study randomized more than 4,000 patients with ST-segment elevation MI (STEMI) and multivessel disease to primary PCI of the culprit lesion only or to staged complete revascularization via PCI of all angiographically significant nonculprit lesions. Complete revascularization proved to be the superior strategy, with a 26% reduction in the risk of the composite of cardiovascular death or MI at a median of 3 years (N Engl J Med. 2019 Oct 10;381[15]:1411-21).
The optimal timing of the staged procedure remains unclear, since the study didn’t specify a protocol.
“I’m still a bit uncomfortable doing multivessel PCI at 2 o’clock in the morning in the setting of STEMI in someone I’ve never met before. I don’t think there’s a rush to do anything then. Often in this middle-of-the-night stuff, we miss things or we overinterpret things. I think it’s better to let the patient cool down, get to know them,” according to Dr. Bell.
EXCEL
Publication of the 5-year outcomes of the largest-ever randomized trial of PCI versus coronary artery bypass grafting (CABG) for left main coronary disease has led to furious controversy, with a few of the surgeons involved in the study opting to publically broadcast allegations of misbehavior on the part of the interventional cardiologist study leadership, charges that have been strongly denied.
The actual results are in line with findings reported from smaller randomized trials. At 5 years in EXCEL, there was no significant difference between the PCI and CABG groups in the primary composite endpoint of death, cerebrovascular accident, or MI (N Engl J Med. 2019 Nov 7;381[19]:1820-30). The all-cause mortality rate was 13% in the PCI arm and 9.9% with CABG, but this finding comes with a caveat.
“I’ll emphasize this trial was never powered to look at mortality. Neither were any of the other randomized trials. On the other hand, I don’t think you can necessarily ignore the finding of an absolute 3.1% difference,” Dr. Bell said.
PCI and CABG are both very good, mature therapies for left main disease, in his view. In the setting of more-complex coronary disease in younger patients, he often views the complete revascularization offered by surgery as the preferred option. On the other hand, in an 80-year-old with severe comorbidities, clearly PCI is attractive.
He considers the highly public nature of this interspecialty spat a regrettable black eye for the entire field of cardiovascular medicine. And he predicted that an ongoing outside neutral-party review of the study data and procedures will conclude, as he has, “there was no malfeasance at all in the trial.”
Dr. Bell reported having no financial conflicts regarding his presentation.
REPORTING FROM ACC SNOWMASS 2020
Evidence-based tools for premenstrual disorders
CASE
A 30-year-old G2P2 woman presents for a well-woman visit and reports 6 months of premenstrual symptoms including irritability, depression, breast pain, and headaches. She is not taking any medications or hormonal contraceptives. She is sexually active and currently not interested in becoming pregnant. She asks what you can do for her symptoms, as they are affecting her life at home and at work.
Symptoms and definitions vary
Although more than 150 premenstrual symptoms have been reported, the most common psychological and behavioral ones are mood swings, depression, anxiety, irritability, crying, social withdrawal, forgetfulness, and problems concentrating.1-3 The most common physical symptoms are fatigue, abdominal bloating, weight gain, breast tenderness, acne, change in appetite or food cravings, edema, headache, and gastrointestinal upset. The etiology of these symptoms is usually multifactorial, with some combination of hormonal, neurotransmitter, lifestyle, environmental, and psychosocial factors playing a role.
Premenstrual disorder. In reviewing diagnostic criteria for the various premenstrual syndromes and disorders from different organizations (eg, the International Society for Premenstrual Disorders; the American College of Obstetricians and Gynecologists; the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition), there is agreement on the following criteria for premenstrual syndrome (PMS)4-6:
- The woman must be ovulating. (Women who no longer menstruate [eg, because of hysterectomy or endometrial ablation] can have premenstrual disorders as long as ovarian function remains intact.)
- The woman experiences a constellation of disabling physical and/or psychological symptoms that appears in the luteal phase of her menstrual cycle.
- The symptoms improve soon after the onset of menses.
- There is a symptom-free interval before ovulation.
- There is prospective documentation of symptoms for at least 2 consecutive cycles.
- The symptoms are sufficient in severity to affect activities of daily living and/or important relationships.
Premenstrual dysphoric disorder. PMDD is another common premenstrual disorder. It is distinguished by significant premenstrual psychological symptoms and requires the presence of marked affective lability, marked irritability or anger, markedly depressed mood, and/or marked anxiety (TABLE 1).7

Exacerbation of other ailments. Another premenstrual disorder is the premenstrual exacerbation of underlying chronic medical or psychological problems such as migraines, seizures, asthma, diabetes, irritable bowel syndrome, fibromyalgia, anxiety, or depression.
Differences in interpretation lead to variations in prevalence
Differences in the interpretation of significant premenstrual symptoms have led to variations in estimated prevalence. For example, 80% to 95% of women report premenstrual symptoms, but only 30% to 40% meet criteria for PMS and only 3% to 8% meet criteria for PMDD.8 Many women who report premenstrual symptoms in a retrospective questionnaire do not meet criteria for PMS or PMDD based on prospective symptom charting. The Daily Record of Severity of Problems (DRSP), a prospective tracking tool for premenstrual symptoms, is sensitive and specific for diagnosing PMS and PMDD if administered on the first day of menstruation.9
Ask about symptoms and use a tracking tool
When you see a woman for a well-woman visit or a gynecologic problem, inquire about physical/emotional symptoms and their severity during the week that precedes menstruation. If a patient reports few symptoms of a mild nature, then no further work-up is needed.
Continue to: If patients report significant...
If patients report significant premenstrual symptoms, recommend the use of a tool to track the symptoms. Older tools such as the DRSP and the Premenstrual Symptoms Screening Tool (PSST), newer symptom diaries that can be used for both PMS and PMDD,and questionnaires that have been used in research situations can be time consuming and difficult for patients to complete.10-12 Instead, physicians can easily construct their own charting tool, as we did for patients to use when tracking their most bothersome symptoms (FIGURE 1). Tracking helps to confirm the diagnosis and helps you and the patient focus on treatment goals.

Keep in mind other diagnoses (eg, anemia, thyroid disorders, perimenopause, anxiety, depression, eating disorders, substance abuse) that can cause or exacerbate the psychological/physical symptoms the patient is reporting. If you suspect any of these other diagnoses, laboratory evaluation (eg, complete blood count, thyroid-stimulating hormone level or other hormonal testing, urine drug screen, etc) may be warranted to rule out other etiologies for the reported symptoms.
Develop a Tx plan that considers symptoms, family-planning needs
Focus treatment on the patient’s predominant symptoms whether they are physical, psychological, or mixed (FIGURE 2). The patient’s preferences regarding family planning are another important consideration. Women who are using a fertility awareness
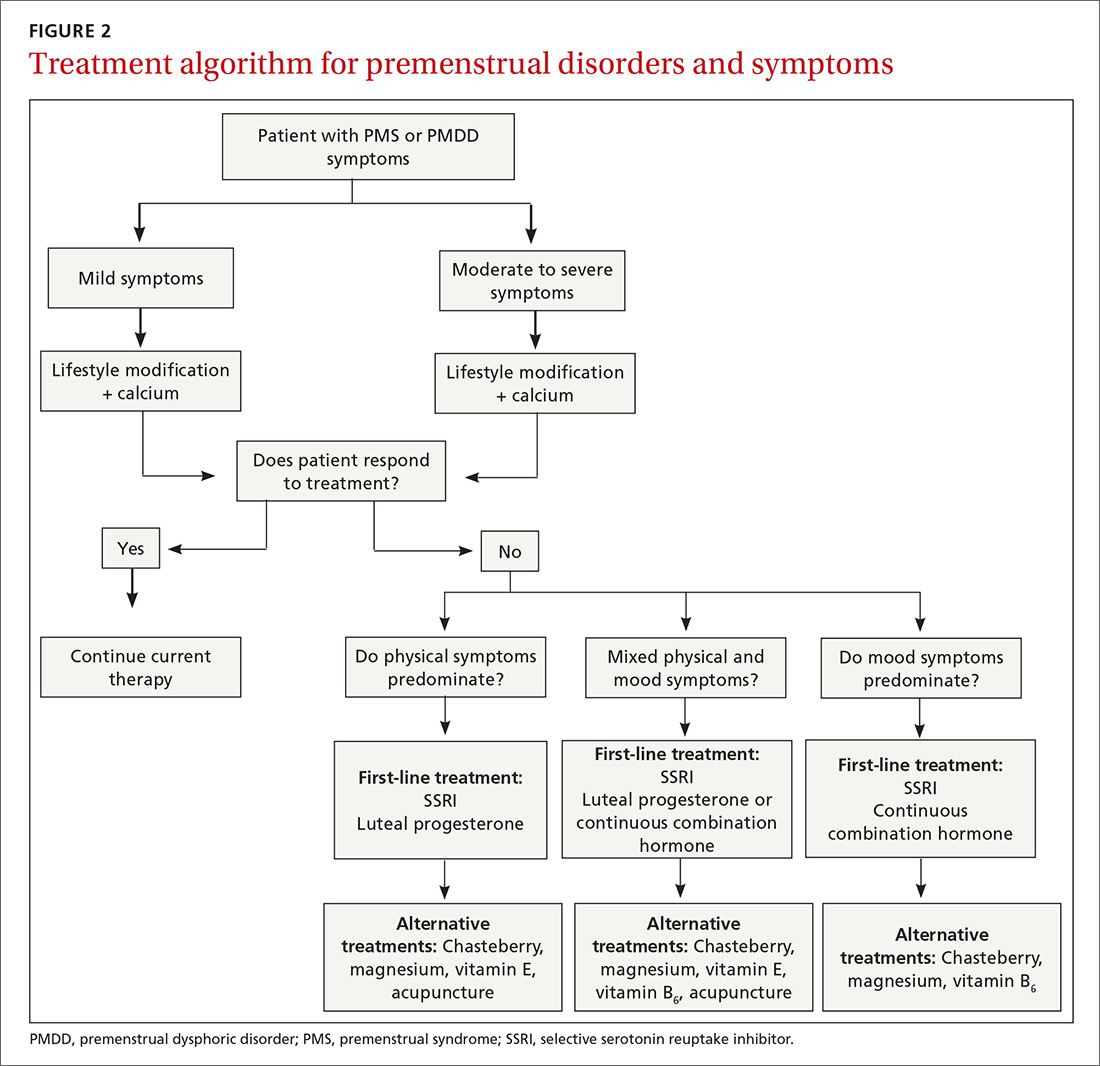
Although the definitions for PMS and PMDD require at least 2 cycles of prospective documentation of symptoms, dietary and lifestyle changes can begin immediately. Regular follow-up to document improvement of symptoms is important; using the patient’s symptoms charting tool can help with this.
Focus on diet and lifestyle right away
Experts in the field of PMS/PMDD suggest that simple dietary changes may be a reasonable first step to help improve symptoms. Researchers have found that diets high in fiber, vegetables, and whole grains are inversely related to PMS.13 Older studies have suggested an increased prevalence and severity of PMS with increased caffeine intake; however, a newer study found no such association.14
Continue to: A case-control study nested...
A case-control study nested within the Nurses’ Health Study II cohort showed that a high intake of both dietary calcium and vitamin D prevented the development of PMS in women ages 27 to 44.15 B vitamins, such as thiamine and riboflavin, from food sources have been associated with a lower risk of PMS.16 A variety of older clinical studies showed benefit from aerobic exercise on PMS symptoms,17-19 but a newer cross-sectional study of young adult women found no association between physical activity and the prevalence of PMS.20 Acupuncture has demonstrated efficacy for the treatment of the physical symptoms of PMS and PMDD, but more rigorous studies are needed.21,22 Cognitive behavioral therapy has been studied as a treatment, but data to support this approach are limited so it cannot be recommended at this time.23
Make the most of supplements—especially calcium
Calcium is the nutritional supplement with the most evidence to support its use to relieve symptoms of PMS and PMDD (TABLE 221,22,24-45). Research indicates that disturbances in calcium regulation and calcium deficiency may be responsible for various premenstrual symptoms. One study showed that, compared with placebo, women who took 1200 mg/d calcium carbonate for 3 menstrual cycles had a 48% decrease in both somatic and affective symptoms.24 Another trial demonstrated improvement in PMS symptoms of early tiredness, appetite changes, and depression with calcium therapy.25
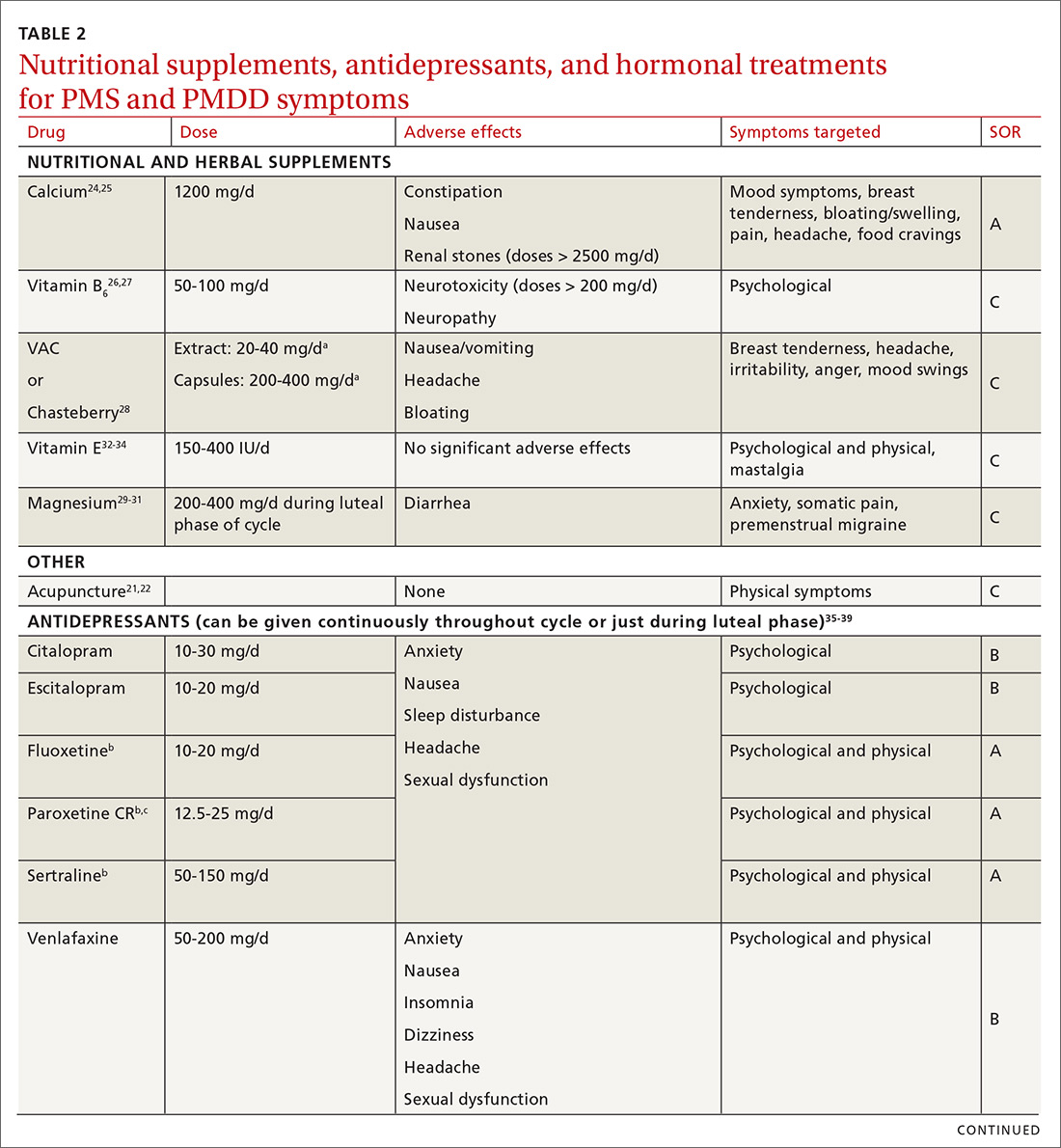
Pyridoxine (vitamin B6) has potential benefit in treating PMS due to its ability to increase levels of serotonin, norepinephrine, histamine, dopamine, and taurine.26 An older systematic review showed benefit for symptoms associated with PMS, but the authors concluded that larger randomized controlled trials (RCTs) were needed before definitive recommendations could be made.27
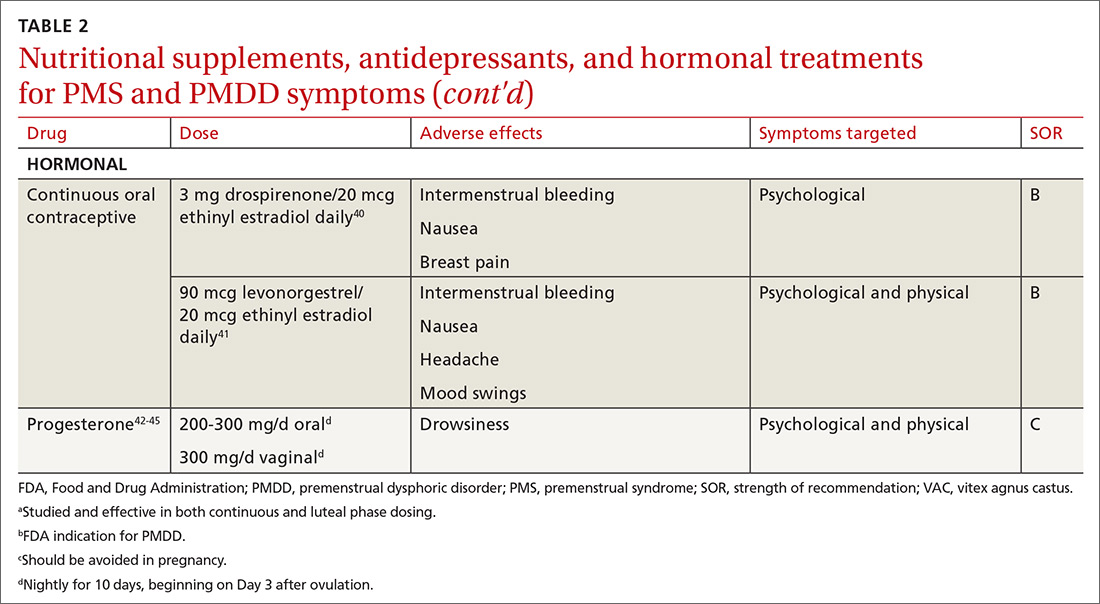
Chasteberry. A number of studies have evaluated the effect of vitex agnus castus (VAC), commonly referred to as chasteberry, on PMS and PMDD symptoms. The exact mechanism of VAC is unknown, but in vitro studies show binding of VAC extracts to dopamine-2 receptors and opioid receptors, and an affinity for estrogen receptors.28
A recent meta-analysis concluded that VAC extracts are not superior to selective serotonin reuptake inhibitors (SSRIs) or oral contraceptives (OCs) for PMS/PMDD.28 The authors suggested a possible benefit of VAC compared with placebo or other nutritional supplements; however, the studies supporting its use are limited by small sample size and potential bias.
Continue to: Magnesium
Magnesium. Many small studies have evaluated the role of other herbal and nutritional supplements for the treatment of PMS/PMDD. A systematic review of studies on the effect of magnesium supplementation on anxiety and stress showed that magnesium may have a potential role in the treatment of the premenstrual symptom of anxiety.29 Other studies have demonstrated a potential role in the treatment of premenstrual migraine.30,31
Vitamin E has demonstrated benefit in the treatment of cyclic mastalgia; however, evidence for using vitamin E for mood and depressive symptoms associated with PMS and PMDD is inconsistent.32-34 Other studies involving vitamin D, St. John’s wort, black cohosh, evening primrose oil, saffron, and ginkgo biloba either showed these agents to be nonefficacious in relieving PMS/PMDD symptoms or to require more data before they can be recommended for use.34,46
Patient doesn’t respond? Start an SSRI
Pharmacotherapy with antidepressants is typically reserved for those who do not respond to nonpharmacologic therapies and are experiencing more moderate to severe symptoms of PMS or PMDD. Reduced levels of serotonin and serotonergic activity in the brain may be linked to symptoms of PMS and PMDD.47 Studies have shown SSRIs to be effective in reducing many psychological symptoms (eg, depression, anxiety, lethargy, irritability) and some physical symptoms (eg, headache, breast tenderness, muscle or joint pain) associated with PMS and PMDD.
A Cochrane review of 31 RCTs compared various SSRIs to placebo. When taken either continuously or intermittently (administration during luteal phase), SSRIs were similarly effective in relieving symptoms when compared with placebo.35 Psychological symptoms are more likely to improve with both low and moderate doses of SSRIs, while physical symptoms may only improve with moderate or higher doses. A direct comparison of the various SSRIs for the treatment of PMS or PMDD is lacking; therefore, the selection of SSRI may be based on patient characteristics and preference.
The benefits of SSRIs are noted much earlier in the treatment of PMS/PMDD than they are observed in their use for depression or anxiety.36 This suggests that the mechanism by which SSRIs relieve PMS/PMDD symptoms is different than that for depression or anxiety. Intermittent dosing capitalizes upon the rapid effect seen with these medications and the cyclical nature of these disorders. In most studies, the benefit of intermittent dosing is similar to continuous dosing; however, one meta-analysis did note that continuous dosing had a larger effect.37
Continue to: The doses of SSRIs...
The doses of SSRIs used in most PMS/PMDD trials were lower than those typically used for the treatment of depression and anxiety. The withdrawal effect that can be seen with abrupt cessation of SSRIs has not been reported in the intermittent-dosing studies for PMS/PMDD.38 While this might imply a more tolerable safety profile, the most common adverse effects reported in trials were still as expected: sleep disturbances, headache, nausea, and sexual dysfunction. It is important to note that SSRIs should be used with caution during pregnancy, and paroxetine should be avoided in women considering pregnancy in the near future.
Other antidepressant classes have been studied to a lesser extent than SSRIs. Continuously dosed venlafaxine, a serotonin and norepinephrine reuptake inhibitor, demonstrated efficacy in PMS/PMDD treatment when compared with placebo within the first cycle of therapy.39 The response seen was comparable to that associated with SSRI treatments in other trials.
Buspirone, an anxiolytic with serotonin receptor activity that is different from that of the SSRIs, demonstrated efficacy in reducing the symptom of irritability.48 Buspirone may have a role to play in those presenting with irritability as a primary symptom or in those who are unable to tolerate the adverse effects of SSRIs. Tricyclic antidepressants, bupropion, and alprazolam have either limited data regarding efficacy or are associated with adverse effects that limit their use.38
Hormonal treatments may be worth considering
One commonly prescribed hormonal therapy for PMS and PMDD is continuous OCs. A 2012 Cochrane review of OCs containing drospirenone evaluated 5 trials and a total of 1920 women.40 Two placebo-controlled trials of women with severe premenstrual symptoms (PMDD) showed improvement after 3 months of taking daily drospirenone 3 mg with ethinyl estradiol 20 mcg, compared with placebo.
While experiencing greater benefit, these groups also experienced significantly more adverse effects including nausea, intermenstrual bleeding, and breast pain. The respective odds ratios for the 3 adverse effects were 3.15 (95% confidence interval [CI], 1.90-5.22), 4.92 (95% CI, 3.03-7.96), and 2.67 (95% CI, 1.50-4.78). The review concluded that drospirenone 3 mg with ethinyl estradiol 20 mcg may help in the treatment of severe premenstrual symptoms (PMDD) but that it is unknown whether this treatment is appropriate for patients with less severe premenstrual symptoms.
Continue to: Another multicenter RCT
Another multicenter RCT evaluated women with PMDD who received levonorgestrel 90 mcg with ethinyl estradiol 20 mcg or placebo daily for 112 days.41 Symptoms were recorded utilizing the DRSP. Significantly more women taking the daily combination hormone (52%) than placebo (40%) had a positive response (≥ 50% improvement in the DRSP 7-day late luteal phase score and Clinical Global Impression of Severity score of ≥ 1 improvement, evaluated at the last “on-therapy” cycle [P = .025]). Twenty-three of 186 patients in the treatment arm dropped out because of adverse effects.
Noncontraceptive estrogen-containing preparations. Hormone therapy preparations containing lower doses of estrogen than seen in OC preparations have also been studied for PMS management. A 2017 Cochrane review of noncontraceptive estrogen-containing preparations found very low-quality evidence to support the effectiveness of continuous estrogen (transdermal patches or subcutaneous implants) plus progestogen.49
Progesterone. The cyclic use of progesterone in the luteal phase has been reviewed as a hormonal treatment for PMS. A 2012 Cochrane review of the efficacy of progesterone for PMS was inconclusive; however, route of administration, dose, and duration differed across studies.42
Another systematic review of 10 trials involving 531 women concluded that progesterone was no better than placebo in the treatment of PMS.43 However, it should be noted that each trial evaluated a different dose of progesterone, and all but 1 of the trials administered progesterone by using the calendar method to predict the beginning of the luteal phase. The only trial to use an objective confirmation of ovulation prior to beginning progesterone therapy did demonstrate significant improvement in premenstrual symptoms.
This 1985 study by Dennerstein et al44 prescribed progesterone for 10 days of each menstrual cycle starting 3 days after ovulation. In each cycle, ovulation was confirmed by determinations of urinary 24-hour pregnanediol and total estrogen concentrations. Progesterone was then prescribed during the objectively identified luteal phase, resulting in significant improvement in symptoms.
Continue to: Another study evaluated...
Another study evaluated the post-ovulatory progesterone profiles of 77 women with symptoms of PMS and found lower levels of progesterone and a sharper rate of decline in the women with PMS vs the control group.45 Subsequent progesterone treatment during the objectively identified luteal phase significantly improved PMS symptoms. These studies would seem to suggest that progesterone replacement when administered during an objectively identified luteal phase may offer some benefit in the treatment of PMS, but larger RCTs are needed to confirm this.
CASE
You provide the patient with diet and lifestyle education as well as a recommendation for calcium supplementation. The patient agrees to prospectively chart her most significant premenstrual symptoms. You review additional treatment options including SSRI medications and hormonal approaches. She is using a fertility awareness–based method of family planning that allows her to confidently identify her luteal phase. She agrees to take sertraline 50 mg/d during the luteal phase of her cycle. At her follow-up office visit 3 months later, she reports improvement in her premenstrual symptoms. Her charting of symptoms confirms this.
CORRESPONDENCE
Peter Danis, MD, Mercy Family Medicine St. Louis, 12680 Olive Boulevard, St. Louis, MO 63141; [email protected].
1. Woods NF, Most A, Dery GK. Prevalence of perimenstrual symptoms. Am J Public Health. 1982;72:1257-1264.
2. Johnson SR, McChesney C, Bean JA. Epidemiology of premenstrual symptoms in a nonclinical sample. 1. Prevalence, natural history and help-seeking behavior. J Repro Med. 1988;33:340-346.
3. Campbell EM, Peterkin D, O’Grady K, et al. Premenstrual symptoms in general practice patients. Prevalence and treatment. J Reprod Med. 1997;42:637-646.
4. O’Brien PM, Bäckström T, Brown C, et al. Towards a consensus on diagnostic criteria, measurement, and trial design of the premenstrual disorders: the ISPMD Montreal consensus. Arch Womens Ment Health. 2011;14:13-21.
5. Epperson CN, Steiner M, Hartlage SA, et al. Premenstrual dysphoric disorder: evidence for a new category for DSM-5. Am J Psychiatry. 2012;169:465-475.
6. American College of Obstetricians and Gynecologists. Guidelines for Women’s Health Care: A Resource Manual. 4th ed. Washington, DC: American College of Obstetricians and Gynecologists; 2014:607-613.
7. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition. Arlington, VA: American Psychiatric Association, 2013.
8. Dennerstein L, Lehert P, Heinemann K. Epidemiology of premenstrual symptoms and disorders. Menopause Int. 2012;18:48-51.
9. Borenstein JE, Dean BB, Yonkers KA, et al. Using the daily record of severity of problems as a screening instrument for premenstrual syndrome. Obstet Gynecol. 2007;109:1068-1075.
10. Steiner M, Macdougall M, Brown E. The premenstrual symptoms screening tool (PSST) for clinicians. Arch Womens Ment Health. 2003;6:203-209.
11. Endicott J, Nee J, Harrison W. Daily Record of Severity of Problems (DRSP): reliability and validity. Arch Womens Ment Health. 2006;9:41-49.
12. Janda C, Kues JN, Andersson G, et al. A symptom diary to assess severe premenstrual syndrome and premenstrual dysphoric disorder. Women Health. 2017;57:837-854.
13. Farasati N, Siassi F, Koohdani F, et al. Western dietary pattern is related to premenstrual syndrome: a case-control study. Brit J Nutr. 2015;114:2016-2021.
14. Purdue-Smithe AC, Manson JE, Hankinson SE, et al. A prospective study of caffeine and coffee intake and premenstrual syndrome. Am J Clin Nutr. 2016;104:499-507.
15. Bertone-Johnson ER, Hankinson SE, Bendich A, et al. Calcium and vitamin D intake and risk of incident premenstrual syndrome. Arch Intern Med. 2005;165:1246-1252.
16. Chocano-Bedoya PO, Manson JE, Hankinson SE, et al. Dietary B vitamin intake and incident premenstrual syndrome. Am J Clin Nutr. 2011;93:1080-1086.
17. Prior JC, Vigna Y. Conditioning exercise and premenstrual symptoms. J Reprod Med. 1987;32:423-428.
18. Aganoff JA, Boyle GJ. Aerobic exercise, mood states, and menstrual cycle symptoms. J Psychosom Res. 1994;38:183-192.
19. El-Lithy A, El-Mazny A, Sabbour A, et al. Effect of aerobic exercise on premenstrual symptoms, haematological and hormonal parameters in young women. J Obstet Gynaecol. 2015;35:389-392.
20. Kroll-Desrosiers AR, Ronnenberg AG, Zagarins SE, et al. Recreational physical activity and premenstrual syndrome in young adult women: a cross-sectional study. PLoS One. 2017;12:1-13.
21. Jang SH, Kim DI, Choi MS. Effects and treatment methods of acupuncture and herbal medicine for premenstrual syndrome/premenstrual dysphoric disorder: systematic review. BMC Complement Altern Med. 2014;14:11.
22. Kim SY, Park HJ, Lee H, et al. Acupuncture for premenstrual syndrome: a systematic review and meta-analysis of randomized controlled trials. BJOG. 2011;118:899-915.
23. Lustyk MK, Gerrish WG, Shaver S, et al. Cognitive-behavioral therapy for premenstrual syndrome and premenstrual dysphoric disorder: a systematic review. Arch Womens Ment Health. 2009;12:85-96.
24. Thys-Jacob S, Starkey P, Bernstein D, et al. Calcium carbonate and the premenstrual syndrome: effects on premenstrual and menstrual syndromes. Am J Obstet Gynecol. 1998;179:444-452.
25. Ghanbari Z, Haghollahi F, Shariat M, et al. Effects of calcium supplement therapy in women with premenstrual syndrome. Taiwan J Obstet Gynecol. 2009;48:124-129.
26. Girman A, Lee R, Kligler B. An integrative medicine approach to premenstrual syndrome. Am J Obstet Gynecol. 2003;188(5 suppl):s56-s65.
27. Wyatt KM, Dimmock PW, Jones PW, et al. Efficacy of vitamin B-6 in the treatment of premenstrual syndrome: systematic review. BMJ. 1999;318:1375-1381.
28. Verkaik S, Kamperman AM, van Westrhenen R, et al. The treatment of premenstrual syndrome with preparations of vitex agnus castus: a systematic review and meta-analysis. Am J Obstet Gynecol. 2017;217:150-166.
29. Boyle NB, Lawton C, Dye L. The effects of magnesium supplementation on subjective anxiety and stress—a systematic review. Nutrients. 2017;9:429-450.
30. Mauskop A, Altura BT, Altura BM. Serum ionized magnesium levels and serum ionized calcium/ionized magnesium ratios in women with menstrual migraine. Headache. 2002;42:242-248.
31. Facchinetti F, Sances C, Borella P, et al. Magnesium prophylaxis of menstrual migraine: effects on intracellular magnesium. Headache. 1991;31:298-301.
32. Parsay S, Olfati F, Nahidi S. Therapeutic effects of vitamin E on cyclic mastalgia. Breast J. 2009;15:510-514.
33. London RS, Murphy L, Kitlowski KE, et al. Efficacy of alpha-tocopherol in the treatment of the premenstrual syndrome. J Reprod Med. 1987;32:400-404.
34. Whelan AM, Jurgens TM, Naylor H. Herbs, vitamins, and minerals in the treatment of premenstrual syndrome: a systematic review. Can J Clin Pharmacol. 2009;16:e407-e429.
, , , . Selective serotonin reuptake inhibitors for premenstrual syndrome. Cochrane Database Syst Rev. 2013;(6): CD001396.
36. Dimmock P, Wyatt K, Jones P, et al. Efficacy of selective serotonin-reuptake inhibitors in premenstrual syndrome: a systematic review. Lancet. 2000;356:1131-1136.
37. Shah NR, Jones JB, Aperi J, et al. Selective serotonin reuptake inhibitors for premenstrual syndrome and premenstrual dysphoric disorder. Obstet Gynecol. 2008;111:1175-1182.
38. Freeman EW. Luteal phase administration of agents for the treatment of premenstrual dysphoric disorder. CNS Drugs. 2004;18:453-468.
39. Freeman EW, Rickels K, Yonkers KA, et al. Venlafaxine in the treatment of premenstrual dysphoric disorder. Obstet Gynecol. 2001;98:737-744.
40. Lopez LM, Kaptein AA, Helmerhorst FM. Oral contraceptives containing drospirenone for premenstrual syndrome. Cochrane Database Syst Rev. 2012;(2):CD006586.
41. Halbreich U, Freeman EW, Rapkin AJ, et al. Continuous oral levonorgestrel/ethinyl estradiol for treating premenstrual dysphoric disorder. Contraception. 2012;85:19-27.
, , , . Progesterone for premenstrual syndrome. Cochrane Database Syst Rev. 2012;(3):CD003415.
43. Wyatt K, Dimmock P, Jones P, et al. Efficacy of progesterone and progestogens in management of premenstrual syndrome: systematic review. BMJ. 2001;323: 776-780.
44. Dennerstein L, Spencer-Gardner C, Gotts G, et al. Progesterone and the premenstrual syndrome: a double-blind crossover trial. Br Med J (Clin Res Ed). 1985;290:1617-1621.
45. NaProTECHNOLOGY. The Medical and Surgical Practice of NaProTECHNOLOGY. Premenstrual Syndrome: Evaluation and Treatment. Omaha, NE: Pope Paul VI Institute Press. 2004;29:345-368. https://www.naprotechnology.com/naprotext.htm. Accessed January 23, 2020.
46. Dante G, Facchinetti F. Herbal treatments for alleviating premenstrual symptoms: a systematic review. J Psychosom Obstet Gynaecol. 2011;32:42-51.
47. Jarvis CI, Lynch AM, Morin AK. Management strategies for premenstrual syndrome/premenstrual dysphoric disorder. Ann Pharmacother. 2008;42:967-978.
48. Landen M, Eriksson O, Sundblad C, et al. Compounds with affinity for serotonergic receptors in the treatment of premenstrual dysphoria: a comparison of buspirone, nefazodone and placebo. Psychopharmacology (Berl). 2001;155:292-298.
, , , . Non-contraceptive oestrogen-containing preparations for controlling symptoms of premenstrual syndrome . Cochrane Database Syst Rev . 2017 ;( 3) :CD010503.
CASE
A 30-year-old G2P2 woman presents for a well-woman visit and reports 6 months of premenstrual symptoms including irritability, depression, breast pain, and headaches. She is not taking any medications or hormonal contraceptives. She is sexually active and currently not interested in becoming pregnant. She asks what you can do for her symptoms, as they are affecting her life at home and at work.
Symptoms and definitions vary
Although more than 150 premenstrual symptoms have been reported, the most common psychological and behavioral ones are mood swings, depression, anxiety, irritability, crying, social withdrawal, forgetfulness, and problems concentrating.1-3 The most common physical symptoms are fatigue, abdominal bloating, weight gain, breast tenderness, acne, change in appetite or food cravings, edema, headache, and gastrointestinal upset. The etiology of these symptoms is usually multifactorial, with some combination of hormonal, neurotransmitter, lifestyle, environmental, and psychosocial factors playing a role.
Premenstrual disorder. In reviewing diagnostic criteria for the various premenstrual syndromes and disorders from different organizations (eg, the International Society for Premenstrual Disorders; the American College of Obstetricians and Gynecologists; the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition), there is agreement on the following criteria for premenstrual syndrome (PMS)4-6:
- The woman must be ovulating. (Women who no longer menstruate [eg, because of hysterectomy or endometrial ablation] can have premenstrual disorders as long as ovarian function remains intact.)
- The woman experiences a constellation of disabling physical and/or psychological symptoms that appears in the luteal phase of her menstrual cycle.
- The symptoms improve soon after the onset of menses.
- There is a symptom-free interval before ovulation.
- There is prospective documentation of symptoms for at least 2 consecutive cycles.
- The symptoms are sufficient in severity to affect activities of daily living and/or important relationships.
Premenstrual dysphoric disorder. PMDD is another common premenstrual disorder. It is distinguished by significant premenstrual psychological symptoms and requires the presence of marked affective lability, marked irritability or anger, markedly depressed mood, and/or marked anxiety (TABLE 1).7

Exacerbation of other ailments. Another premenstrual disorder is the premenstrual exacerbation of underlying chronic medical or psychological problems such as migraines, seizures, asthma, diabetes, irritable bowel syndrome, fibromyalgia, anxiety, or depression.
Differences in interpretation lead to variations in prevalence
Differences in the interpretation of significant premenstrual symptoms have led to variations in estimated prevalence. For example, 80% to 95% of women report premenstrual symptoms, but only 30% to 40% meet criteria for PMS and only 3% to 8% meet criteria for PMDD.8 Many women who report premenstrual symptoms in a retrospective questionnaire do not meet criteria for PMS or PMDD based on prospective symptom charting. The Daily Record of Severity of Problems (DRSP), a prospective tracking tool for premenstrual symptoms, is sensitive and specific for diagnosing PMS and PMDD if administered on the first day of menstruation.9
Ask about symptoms and use a tracking tool
When you see a woman for a well-woman visit or a gynecologic problem, inquire about physical/emotional symptoms and their severity during the week that precedes menstruation. If a patient reports few symptoms of a mild nature, then no further work-up is needed.
Continue to: If patients report significant...
If patients report significant premenstrual symptoms, recommend the use of a tool to track the symptoms. Older tools such as the DRSP and the Premenstrual Symptoms Screening Tool (PSST), newer symptom diaries that can be used for both PMS and PMDD,and questionnaires that have been used in research situations can be time consuming and difficult for patients to complete.10-12 Instead, physicians can easily construct their own charting tool, as we did for patients to use when tracking their most bothersome symptoms (FIGURE 1). Tracking helps to confirm the diagnosis and helps you and the patient focus on treatment goals.

Keep in mind other diagnoses (eg, anemia, thyroid disorders, perimenopause, anxiety, depression, eating disorders, substance abuse) that can cause or exacerbate the psychological/physical symptoms the patient is reporting. If you suspect any of these other diagnoses, laboratory evaluation (eg, complete blood count, thyroid-stimulating hormone level or other hormonal testing, urine drug screen, etc) may be warranted to rule out other etiologies for the reported symptoms.
Develop a Tx plan that considers symptoms, family-planning needs
Focus treatment on the patient’s predominant symptoms whether they are physical, psychological, or mixed (FIGURE 2). The patient’s preferences regarding family planning are another important consideration. Women who are using a fertility awareness

Although the definitions for PMS and PMDD require at least 2 cycles of prospective documentation of symptoms, dietary and lifestyle changes can begin immediately. Regular follow-up to document improvement of symptoms is important; using the patient’s symptoms charting tool can help with this.
Focus on diet and lifestyle right away
Experts in the field of PMS/PMDD suggest that simple dietary changes may be a reasonable first step to help improve symptoms. Researchers have found that diets high in fiber, vegetables, and whole grains are inversely related to PMS.13 Older studies have suggested an increased prevalence and severity of PMS with increased caffeine intake; however, a newer study found no such association.14
Continue to: A case-control study nested...
A case-control study nested within the Nurses’ Health Study II cohort showed that a high intake of both dietary calcium and vitamin D prevented the development of PMS in women ages 27 to 44.15 B vitamins, such as thiamine and riboflavin, from food sources have been associated with a lower risk of PMS.16 A variety of older clinical studies showed benefit from aerobic exercise on PMS symptoms,17-19 but a newer cross-sectional study of young adult women found no association between physical activity and the prevalence of PMS.20 Acupuncture has demonstrated efficacy for the treatment of the physical symptoms of PMS and PMDD, but more rigorous studies are needed.21,22 Cognitive behavioral therapy has been studied as a treatment, but data to support this approach are limited so it cannot be recommended at this time.23
Make the most of supplements—especially calcium
Calcium is the nutritional supplement with the most evidence to support its use to relieve symptoms of PMS and PMDD (TABLE 221,22,24-45). Research indicates that disturbances in calcium regulation and calcium deficiency may be responsible for various premenstrual symptoms. One study showed that, compared with placebo, women who took 1200 mg/d calcium carbonate for 3 menstrual cycles had a 48% decrease in both somatic and affective symptoms.24 Another trial demonstrated improvement in PMS symptoms of early tiredness, appetite changes, and depression with calcium therapy.25

Pyridoxine (vitamin B6) has potential benefit in treating PMS due to its ability to increase levels of serotonin, norepinephrine, histamine, dopamine, and taurine.26 An older systematic review showed benefit for symptoms associated with PMS, but the authors concluded that larger randomized controlled trials (RCTs) were needed before definitive recommendations could be made.27

Chasteberry. A number of studies have evaluated the effect of vitex agnus castus (VAC), commonly referred to as chasteberry, on PMS and PMDD symptoms. The exact mechanism of VAC is unknown, but in vitro studies show binding of VAC extracts to dopamine-2 receptors and opioid receptors, and an affinity for estrogen receptors.28
A recent meta-analysis concluded that VAC extracts are not superior to selective serotonin reuptake inhibitors (SSRIs) or oral contraceptives (OCs) for PMS/PMDD.28 The authors suggested a possible benefit of VAC compared with placebo or other nutritional supplements; however, the studies supporting its use are limited by small sample size and potential bias.
Continue to: Magnesium
Magnesium. Many small studies have evaluated the role of other herbal and nutritional supplements for the treatment of PMS/PMDD. A systematic review of studies on the effect of magnesium supplementation on anxiety and stress showed that magnesium may have a potential role in the treatment of the premenstrual symptom of anxiety.29 Other studies have demonstrated a potential role in the treatment of premenstrual migraine.30,31
Vitamin E has demonstrated benefit in the treatment of cyclic mastalgia; however, evidence for using vitamin E for mood and depressive symptoms associated with PMS and PMDD is inconsistent.32-34 Other studies involving vitamin D, St. John’s wort, black cohosh, evening primrose oil, saffron, and ginkgo biloba either showed these agents to be nonefficacious in relieving PMS/PMDD symptoms or to require more data before they can be recommended for use.34,46
Patient doesn’t respond? Start an SSRI
Pharmacotherapy with antidepressants is typically reserved for those who do not respond to nonpharmacologic therapies and are experiencing more moderate to severe symptoms of PMS or PMDD. Reduced levels of serotonin and serotonergic activity in the brain may be linked to symptoms of PMS and PMDD.47 Studies have shown SSRIs to be effective in reducing many psychological symptoms (eg, depression, anxiety, lethargy, irritability) and some physical symptoms (eg, headache, breast tenderness, muscle or joint pain) associated with PMS and PMDD.
A Cochrane review of 31 RCTs compared various SSRIs to placebo. When taken either continuously or intermittently (administration during luteal phase), SSRIs were similarly effective in relieving symptoms when compared with placebo.35 Psychological symptoms are more likely to improve with both low and moderate doses of SSRIs, while physical symptoms may only improve with moderate or higher doses. A direct comparison of the various SSRIs for the treatment of PMS or PMDD is lacking; therefore, the selection of SSRI may be based on patient characteristics and preference.
The benefits of SSRIs are noted much earlier in the treatment of PMS/PMDD than they are observed in their use for depression or anxiety.36 This suggests that the mechanism by which SSRIs relieve PMS/PMDD symptoms is different than that for depression or anxiety. Intermittent dosing capitalizes upon the rapid effect seen with these medications and the cyclical nature of these disorders. In most studies, the benefit of intermittent dosing is similar to continuous dosing; however, one meta-analysis did note that continuous dosing had a larger effect.37
Continue to: The doses of SSRIs...
The doses of SSRIs used in most PMS/PMDD trials were lower than those typically used for the treatment of depression and anxiety. The withdrawal effect that can be seen with abrupt cessation of SSRIs has not been reported in the intermittent-dosing studies for PMS/PMDD.38 While this might imply a more tolerable safety profile, the most common adverse effects reported in trials were still as expected: sleep disturbances, headache, nausea, and sexual dysfunction. It is important to note that SSRIs should be used with caution during pregnancy, and paroxetine should be avoided in women considering pregnancy in the near future.
Other antidepressant classes have been studied to a lesser extent than SSRIs. Continuously dosed venlafaxine, a serotonin and norepinephrine reuptake inhibitor, demonstrated efficacy in PMS/PMDD treatment when compared with placebo within the first cycle of therapy.39 The response seen was comparable to that associated with SSRI treatments in other trials.
Buspirone, an anxiolytic with serotonin receptor activity that is different from that of the SSRIs, demonstrated efficacy in reducing the symptom of irritability.48 Buspirone may have a role to play in those presenting with irritability as a primary symptom or in those who are unable to tolerate the adverse effects of SSRIs. Tricyclic antidepressants, bupropion, and alprazolam have either limited data regarding efficacy or are associated with adverse effects that limit their use.38
Hormonal treatments may be worth considering
One commonly prescribed hormonal therapy for PMS and PMDD is continuous OCs. A 2012 Cochrane review of OCs containing drospirenone evaluated 5 trials and a total of 1920 women.40 Two placebo-controlled trials of women with severe premenstrual symptoms (PMDD) showed improvement after 3 months of taking daily drospirenone 3 mg with ethinyl estradiol 20 mcg, compared with placebo.
While experiencing greater benefit, these groups also experienced significantly more adverse effects including nausea, intermenstrual bleeding, and breast pain. The respective odds ratios for the 3 adverse effects were 3.15 (95% confidence interval [CI], 1.90-5.22), 4.92 (95% CI, 3.03-7.96), and 2.67 (95% CI, 1.50-4.78). The review concluded that drospirenone 3 mg with ethinyl estradiol 20 mcg may help in the treatment of severe premenstrual symptoms (PMDD) but that it is unknown whether this treatment is appropriate for patients with less severe premenstrual symptoms.
Continue to: Another multicenter RCT
Another multicenter RCT evaluated women with PMDD who received levonorgestrel 90 mcg with ethinyl estradiol 20 mcg or placebo daily for 112 days.41 Symptoms were recorded utilizing the DRSP. Significantly more women taking the daily combination hormone (52%) than placebo (40%) had a positive response (≥ 50% improvement in the DRSP 7-day late luteal phase score and Clinical Global Impression of Severity score of ≥ 1 improvement, evaluated at the last “on-therapy” cycle [P = .025]). Twenty-three of 186 patients in the treatment arm dropped out because of adverse effects.
Noncontraceptive estrogen-containing preparations. Hormone therapy preparations containing lower doses of estrogen than seen in OC preparations have also been studied for PMS management. A 2017 Cochrane review of noncontraceptive estrogen-containing preparations found very low-quality evidence to support the effectiveness of continuous estrogen (transdermal patches or subcutaneous implants) plus progestogen.49
Progesterone. The cyclic use of progesterone in the luteal phase has been reviewed as a hormonal treatment for PMS. A 2012 Cochrane review of the efficacy of progesterone for PMS was inconclusive; however, route of administration, dose, and duration differed across studies.42
Another systematic review of 10 trials involving 531 women concluded that progesterone was no better than placebo in the treatment of PMS.43 However, it should be noted that each trial evaluated a different dose of progesterone, and all but 1 of the trials administered progesterone by using the calendar method to predict the beginning of the luteal phase. The only trial to use an objective confirmation of ovulation prior to beginning progesterone therapy did demonstrate significant improvement in premenstrual symptoms.
This 1985 study by Dennerstein et al44 prescribed progesterone for 10 days of each menstrual cycle starting 3 days after ovulation. In each cycle, ovulation was confirmed by determinations of urinary 24-hour pregnanediol and total estrogen concentrations. Progesterone was then prescribed during the objectively identified luteal phase, resulting in significant improvement in symptoms.
Continue to: Another study evaluated...
Another study evaluated the post-ovulatory progesterone profiles of 77 women with symptoms of PMS and found lower levels of progesterone and a sharper rate of decline in the women with PMS vs the control group.45 Subsequent progesterone treatment during the objectively identified luteal phase significantly improved PMS symptoms. These studies would seem to suggest that progesterone replacement when administered during an objectively identified luteal phase may offer some benefit in the treatment of PMS, but larger RCTs are needed to confirm this.
CASE
You provide the patient with diet and lifestyle education as well as a recommendation for calcium supplementation. The patient agrees to prospectively chart her most significant premenstrual symptoms. You review additional treatment options including SSRI medications and hormonal approaches. She is using a fertility awareness–based method of family planning that allows her to confidently identify her luteal phase. She agrees to take sertraline 50 mg/d during the luteal phase of her cycle. At her follow-up office visit 3 months later, she reports improvement in her premenstrual symptoms. Her charting of symptoms confirms this.
CORRESPONDENCE
Peter Danis, MD, Mercy Family Medicine St. Louis, 12680 Olive Boulevard, St. Louis, MO 63141; [email protected].
CASE
A 30-year-old G2P2 woman presents for a well-woman visit and reports 6 months of premenstrual symptoms including irritability, depression, breast pain, and headaches. She is not taking any medications or hormonal contraceptives. She is sexually active and currently not interested in becoming pregnant. She asks what you can do for her symptoms, as they are affecting her life at home and at work.
Symptoms and definitions vary
Although more than 150 premenstrual symptoms have been reported, the most common psychological and behavioral ones are mood swings, depression, anxiety, irritability, crying, social withdrawal, forgetfulness, and problems concentrating.1-3 The most common physical symptoms are fatigue, abdominal bloating, weight gain, breast tenderness, acne, change in appetite or food cravings, edema, headache, and gastrointestinal upset. The etiology of these symptoms is usually multifactorial, with some combination of hormonal, neurotransmitter, lifestyle, environmental, and psychosocial factors playing a role.
Premenstrual disorder. In reviewing diagnostic criteria for the various premenstrual syndromes and disorders from different organizations (eg, the International Society for Premenstrual Disorders; the American College of Obstetricians and Gynecologists; the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition), there is agreement on the following criteria for premenstrual syndrome (PMS)4-6:
- The woman must be ovulating. (Women who no longer menstruate [eg, because of hysterectomy or endometrial ablation] can have premenstrual disorders as long as ovarian function remains intact.)
- The woman experiences a constellation of disabling physical and/or psychological symptoms that appears in the luteal phase of her menstrual cycle.
- The symptoms improve soon after the onset of menses.
- There is a symptom-free interval before ovulation.
- There is prospective documentation of symptoms for at least 2 consecutive cycles.
- The symptoms are sufficient in severity to affect activities of daily living and/or important relationships.
Premenstrual dysphoric disorder. PMDD is another common premenstrual disorder. It is distinguished by significant premenstrual psychological symptoms and requires the presence of marked affective lability, marked irritability or anger, markedly depressed mood, and/or marked anxiety (TABLE 1).7

Exacerbation of other ailments. Another premenstrual disorder is the premenstrual exacerbation of underlying chronic medical or psychological problems such as migraines, seizures, asthma, diabetes, irritable bowel syndrome, fibromyalgia, anxiety, or depression.
Differences in interpretation lead to variations in prevalence
Differences in the interpretation of significant premenstrual symptoms have led to variations in estimated prevalence. For example, 80% to 95% of women report premenstrual symptoms, but only 30% to 40% meet criteria for PMS and only 3% to 8% meet criteria for PMDD.8 Many women who report premenstrual symptoms in a retrospective questionnaire do not meet criteria for PMS or PMDD based on prospective symptom charting. The Daily Record of Severity of Problems (DRSP), a prospective tracking tool for premenstrual symptoms, is sensitive and specific for diagnosing PMS and PMDD if administered on the first day of menstruation.9
Ask about symptoms and use a tracking tool
When you see a woman for a well-woman visit or a gynecologic problem, inquire about physical/emotional symptoms and their severity during the week that precedes menstruation. If a patient reports few symptoms of a mild nature, then no further work-up is needed.
Continue to: If patients report significant...
If patients report significant premenstrual symptoms, recommend the use of a tool to track the symptoms. Older tools such as the DRSP and the Premenstrual Symptoms Screening Tool (PSST), newer symptom diaries that can be used for both PMS and PMDD,and questionnaires that have been used in research situations can be time consuming and difficult for patients to complete.10-12 Instead, physicians can easily construct their own charting tool, as we did for patients to use when tracking their most bothersome symptoms (FIGURE 1). Tracking helps to confirm the diagnosis and helps you and the patient focus on treatment goals.

Keep in mind other diagnoses (eg, anemia, thyroid disorders, perimenopause, anxiety, depression, eating disorders, substance abuse) that can cause or exacerbate the psychological/physical symptoms the patient is reporting. If you suspect any of these other diagnoses, laboratory evaluation (eg, complete blood count, thyroid-stimulating hormone level or other hormonal testing, urine drug screen, etc) may be warranted to rule out other etiologies for the reported symptoms.
Develop a Tx plan that considers symptoms, family-planning needs
Focus treatment on the patient’s predominant symptoms whether they are physical, psychological, or mixed (FIGURE 2). The patient’s preferences regarding family planning are another important consideration. Women who are using a fertility awareness

Although the definitions for PMS and PMDD require at least 2 cycles of prospective documentation of symptoms, dietary and lifestyle changes can begin immediately. Regular follow-up to document improvement of symptoms is important; using the patient’s symptoms charting tool can help with this.
Focus on diet and lifestyle right away
Experts in the field of PMS/PMDD suggest that simple dietary changes may be a reasonable first step to help improve symptoms. Researchers have found that diets high in fiber, vegetables, and whole grains are inversely related to PMS.13 Older studies have suggested an increased prevalence and severity of PMS with increased caffeine intake; however, a newer study found no such association.14
Continue to: A case-control study nested...
A case-control study nested within the Nurses’ Health Study II cohort showed that a high intake of both dietary calcium and vitamin D prevented the development of PMS in women ages 27 to 44.15 B vitamins, such as thiamine and riboflavin, from food sources have been associated with a lower risk of PMS.16 A variety of older clinical studies showed benefit from aerobic exercise on PMS symptoms,17-19 but a newer cross-sectional study of young adult women found no association between physical activity and the prevalence of PMS.20 Acupuncture has demonstrated efficacy for the treatment of the physical symptoms of PMS and PMDD, but more rigorous studies are needed.21,22 Cognitive behavioral therapy has been studied as a treatment, but data to support this approach are limited so it cannot be recommended at this time.23
Make the most of supplements—especially calcium
Calcium is the nutritional supplement with the most evidence to support its use to relieve symptoms of PMS and PMDD (TABLE 221,22,24-45). Research indicates that disturbances in calcium regulation and calcium deficiency may be responsible for various premenstrual symptoms. One study showed that, compared with placebo, women who took 1200 mg/d calcium carbonate for 3 menstrual cycles had a 48% decrease in both somatic and affective symptoms.24 Another trial demonstrated improvement in PMS symptoms of early tiredness, appetite changes, and depression with calcium therapy.25

Pyridoxine (vitamin B6) has potential benefit in treating PMS due to its ability to increase levels of serotonin, norepinephrine, histamine, dopamine, and taurine.26 An older systematic review showed benefit for symptoms associated with PMS, but the authors concluded that larger randomized controlled trials (RCTs) were needed before definitive recommendations could be made.27

Chasteberry. A number of studies have evaluated the effect of vitex agnus castus (VAC), commonly referred to as chasteberry, on PMS and PMDD symptoms. The exact mechanism of VAC is unknown, but in vitro studies show binding of VAC extracts to dopamine-2 receptors and opioid receptors, and an affinity for estrogen receptors.28
A recent meta-analysis concluded that VAC extracts are not superior to selective serotonin reuptake inhibitors (SSRIs) or oral contraceptives (OCs) for PMS/PMDD.28 The authors suggested a possible benefit of VAC compared with placebo or other nutritional supplements; however, the studies supporting its use are limited by small sample size and potential bias.
Continue to: Magnesium
Magnesium. Many small studies have evaluated the role of other herbal and nutritional supplements for the treatment of PMS/PMDD. A systematic review of studies on the effect of magnesium supplementation on anxiety and stress showed that magnesium may have a potential role in the treatment of the premenstrual symptom of anxiety.29 Other studies have demonstrated a potential role in the treatment of premenstrual migraine.30,31
Vitamin E has demonstrated benefit in the treatment of cyclic mastalgia; however, evidence for using vitamin E for mood and depressive symptoms associated with PMS and PMDD is inconsistent.32-34 Other studies involving vitamin D, St. John’s wort, black cohosh, evening primrose oil, saffron, and ginkgo biloba either showed these agents to be nonefficacious in relieving PMS/PMDD symptoms or to require more data before they can be recommended for use.34,46
Patient doesn’t respond? Start an SSRI
Pharmacotherapy with antidepressants is typically reserved for those who do not respond to nonpharmacologic therapies and are experiencing more moderate to severe symptoms of PMS or PMDD. Reduced levels of serotonin and serotonergic activity in the brain may be linked to symptoms of PMS and PMDD.47 Studies have shown SSRIs to be effective in reducing many psychological symptoms (eg, depression, anxiety, lethargy, irritability) and some physical symptoms (eg, headache, breast tenderness, muscle or joint pain) associated with PMS and PMDD.
A Cochrane review of 31 RCTs compared various SSRIs to placebo. When taken either continuously or intermittently (administration during luteal phase), SSRIs were similarly effective in relieving symptoms when compared with placebo.35 Psychological symptoms are more likely to improve with both low and moderate doses of SSRIs, while physical symptoms may only improve with moderate or higher doses. A direct comparison of the various SSRIs for the treatment of PMS or PMDD is lacking; therefore, the selection of SSRI may be based on patient characteristics and preference.
The benefits of SSRIs are noted much earlier in the treatment of PMS/PMDD than they are observed in their use for depression or anxiety.36 This suggests that the mechanism by which SSRIs relieve PMS/PMDD symptoms is different than that for depression or anxiety. Intermittent dosing capitalizes upon the rapid effect seen with these medications and the cyclical nature of these disorders. In most studies, the benefit of intermittent dosing is similar to continuous dosing; however, one meta-analysis did note that continuous dosing had a larger effect.37
Continue to: The doses of SSRIs...
The doses of SSRIs used in most PMS/PMDD trials were lower than those typically used for the treatment of depression and anxiety. The withdrawal effect that can be seen with abrupt cessation of SSRIs has not been reported in the intermittent-dosing studies for PMS/PMDD.38 While this might imply a more tolerable safety profile, the most common adverse effects reported in trials were still as expected: sleep disturbances, headache, nausea, and sexual dysfunction. It is important to note that SSRIs should be used with caution during pregnancy, and paroxetine should be avoided in women considering pregnancy in the near future.
Other antidepressant classes have been studied to a lesser extent than SSRIs. Continuously dosed venlafaxine, a serotonin and norepinephrine reuptake inhibitor, demonstrated efficacy in PMS/PMDD treatment when compared with placebo within the first cycle of therapy.39 The response seen was comparable to that associated with SSRI treatments in other trials.
Buspirone, an anxiolytic with serotonin receptor activity that is different from that of the SSRIs, demonstrated efficacy in reducing the symptom of irritability.48 Buspirone may have a role to play in those presenting with irritability as a primary symptom or in those who are unable to tolerate the adverse effects of SSRIs. Tricyclic antidepressants, bupropion, and alprazolam have either limited data regarding efficacy or are associated with adverse effects that limit their use.38
Hormonal treatments may be worth considering
One commonly prescribed hormonal therapy for PMS and PMDD is continuous OCs. A 2012 Cochrane review of OCs containing drospirenone evaluated 5 trials and a total of 1920 women.40 Two placebo-controlled trials of women with severe premenstrual symptoms (PMDD) showed improvement after 3 months of taking daily drospirenone 3 mg with ethinyl estradiol 20 mcg, compared with placebo.
While experiencing greater benefit, these groups also experienced significantly more adverse effects including nausea, intermenstrual bleeding, and breast pain. The respective odds ratios for the 3 adverse effects were 3.15 (95% confidence interval [CI], 1.90-5.22), 4.92 (95% CI, 3.03-7.96), and 2.67 (95% CI, 1.50-4.78). The review concluded that drospirenone 3 mg with ethinyl estradiol 20 mcg may help in the treatment of severe premenstrual symptoms (PMDD) but that it is unknown whether this treatment is appropriate for patients with less severe premenstrual symptoms.
Continue to: Another multicenter RCT
Another multicenter RCT evaluated women with PMDD who received levonorgestrel 90 mcg with ethinyl estradiol 20 mcg or placebo daily for 112 days.41 Symptoms were recorded utilizing the DRSP. Significantly more women taking the daily combination hormone (52%) than placebo (40%) had a positive response (≥ 50% improvement in the DRSP 7-day late luteal phase score and Clinical Global Impression of Severity score of ≥ 1 improvement, evaluated at the last “on-therapy” cycle [P = .025]). Twenty-three of 186 patients in the treatment arm dropped out because of adverse effects.
Noncontraceptive estrogen-containing preparations. Hormone therapy preparations containing lower doses of estrogen than seen in OC preparations have also been studied for PMS management. A 2017 Cochrane review of noncontraceptive estrogen-containing preparations found very low-quality evidence to support the effectiveness of continuous estrogen (transdermal patches or subcutaneous implants) plus progestogen.49
Progesterone. The cyclic use of progesterone in the luteal phase has been reviewed as a hormonal treatment for PMS. A 2012 Cochrane review of the efficacy of progesterone for PMS was inconclusive; however, route of administration, dose, and duration differed across studies.42
Another systematic review of 10 trials involving 531 women concluded that progesterone was no better than placebo in the treatment of PMS.43 However, it should be noted that each trial evaluated a different dose of progesterone, and all but 1 of the trials administered progesterone by using the calendar method to predict the beginning of the luteal phase. The only trial to use an objective confirmation of ovulation prior to beginning progesterone therapy did demonstrate significant improvement in premenstrual symptoms.
This 1985 study by Dennerstein et al44 prescribed progesterone for 10 days of each menstrual cycle starting 3 days after ovulation. In each cycle, ovulation was confirmed by determinations of urinary 24-hour pregnanediol and total estrogen concentrations. Progesterone was then prescribed during the objectively identified luteal phase, resulting in significant improvement in symptoms.
Continue to: Another study evaluated...
Another study evaluated the post-ovulatory progesterone profiles of 77 women with symptoms of PMS and found lower levels of progesterone and a sharper rate of decline in the women with PMS vs the control group.45 Subsequent progesterone treatment during the objectively identified luteal phase significantly improved PMS symptoms. These studies would seem to suggest that progesterone replacement when administered during an objectively identified luteal phase may offer some benefit in the treatment of PMS, but larger RCTs are needed to confirm this.
CASE
You provide the patient with diet and lifestyle education as well as a recommendation for calcium supplementation. The patient agrees to prospectively chart her most significant premenstrual symptoms. You review additional treatment options including SSRI medications and hormonal approaches. She is using a fertility awareness–based method of family planning that allows her to confidently identify her luteal phase. She agrees to take sertraline 50 mg/d during the luteal phase of her cycle. At her follow-up office visit 3 months later, she reports improvement in her premenstrual symptoms. Her charting of symptoms confirms this.
CORRESPONDENCE
Peter Danis, MD, Mercy Family Medicine St. Louis, 12680 Olive Boulevard, St. Louis, MO 63141; [email protected].
1. Woods NF, Most A, Dery GK. Prevalence of perimenstrual symptoms. Am J Public Health. 1982;72:1257-1264.
2. Johnson SR, McChesney C, Bean JA. Epidemiology of premenstrual symptoms in a nonclinical sample. 1. Prevalence, natural history and help-seeking behavior. J Repro Med. 1988;33:340-346.
3. Campbell EM, Peterkin D, O’Grady K, et al. Premenstrual symptoms in general practice patients. Prevalence and treatment. J Reprod Med. 1997;42:637-646.
4. O’Brien PM, Bäckström T, Brown C, et al. Towards a consensus on diagnostic criteria, measurement, and trial design of the premenstrual disorders: the ISPMD Montreal consensus. Arch Womens Ment Health. 2011;14:13-21.
5. Epperson CN, Steiner M, Hartlage SA, et al. Premenstrual dysphoric disorder: evidence for a new category for DSM-5. Am J Psychiatry. 2012;169:465-475.
6. American College of Obstetricians and Gynecologists. Guidelines for Women’s Health Care: A Resource Manual. 4th ed. Washington, DC: American College of Obstetricians and Gynecologists; 2014:607-613.
7. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition. Arlington, VA: American Psychiatric Association, 2013.
8. Dennerstein L, Lehert P, Heinemann K. Epidemiology of premenstrual symptoms and disorders. Menopause Int. 2012;18:48-51.
9. Borenstein JE, Dean BB, Yonkers KA, et al. Using the daily record of severity of problems as a screening instrument for premenstrual syndrome. Obstet Gynecol. 2007;109:1068-1075.
10. Steiner M, Macdougall M, Brown E. The premenstrual symptoms screening tool (PSST) for clinicians. Arch Womens Ment Health. 2003;6:203-209.
11. Endicott J, Nee J, Harrison W. Daily Record of Severity of Problems (DRSP): reliability and validity. Arch Womens Ment Health. 2006;9:41-49.
12. Janda C, Kues JN, Andersson G, et al. A symptom diary to assess severe premenstrual syndrome and premenstrual dysphoric disorder. Women Health. 2017;57:837-854.
13. Farasati N, Siassi F, Koohdani F, et al. Western dietary pattern is related to premenstrual syndrome: a case-control study. Brit J Nutr. 2015;114:2016-2021.
14. Purdue-Smithe AC, Manson JE, Hankinson SE, et al. A prospective study of caffeine and coffee intake and premenstrual syndrome. Am J Clin Nutr. 2016;104:499-507.
15. Bertone-Johnson ER, Hankinson SE, Bendich A, et al. Calcium and vitamin D intake and risk of incident premenstrual syndrome. Arch Intern Med. 2005;165:1246-1252.
16. Chocano-Bedoya PO, Manson JE, Hankinson SE, et al. Dietary B vitamin intake and incident premenstrual syndrome. Am J Clin Nutr. 2011;93:1080-1086.
17. Prior JC, Vigna Y. Conditioning exercise and premenstrual symptoms. J Reprod Med. 1987;32:423-428.
18. Aganoff JA, Boyle GJ. Aerobic exercise, mood states, and menstrual cycle symptoms. J Psychosom Res. 1994;38:183-192.
19. El-Lithy A, El-Mazny A, Sabbour A, et al. Effect of aerobic exercise on premenstrual symptoms, haematological and hormonal parameters in young women. J Obstet Gynaecol. 2015;35:389-392.
20. Kroll-Desrosiers AR, Ronnenberg AG, Zagarins SE, et al. Recreational physical activity and premenstrual syndrome in young adult women: a cross-sectional study. PLoS One. 2017;12:1-13.
21. Jang SH, Kim DI, Choi MS. Effects and treatment methods of acupuncture and herbal medicine for premenstrual syndrome/premenstrual dysphoric disorder: systematic review. BMC Complement Altern Med. 2014;14:11.
22. Kim SY, Park HJ, Lee H, et al. Acupuncture for premenstrual syndrome: a systematic review and meta-analysis of randomized controlled trials. BJOG. 2011;118:899-915.
23. Lustyk MK, Gerrish WG, Shaver S, et al. Cognitive-behavioral therapy for premenstrual syndrome and premenstrual dysphoric disorder: a systematic review. Arch Womens Ment Health. 2009;12:85-96.
24. Thys-Jacob S, Starkey P, Bernstein D, et al. Calcium carbonate and the premenstrual syndrome: effects on premenstrual and menstrual syndromes. Am J Obstet Gynecol. 1998;179:444-452.
25. Ghanbari Z, Haghollahi F, Shariat M, et al. Effects of calcium supplement therapy in women with premenstrual syndrome. Taiwan J Obstet Gynecol. 2009;48:124-129.
26. Girman A, Lee R, Kligler B. An integrative medicine approach to premenstrual syndrome. Am J Obstet Gynecol. 2003;188(5 suppl):s56-s65.
27. Wyatt KM, Dimmock PW, Jones PW, et al. Efficacy of vitamin B-6 in the treatment of premenstrual syndrome: systematic review. BMJ. 1999;318:1375-1381.
28. Verkaik S, Kamperman AM, van Westrhenen R, et al. The treatment of premenstrual syndrome with preparations of vitex agnus castus: a systematic review and meta-analysis. Am J Obstet Gynecol. 2017;217:150-166.
29. Boyle NB, Lawton C, Dye L. The effects of magnesium supplementation on subjective anxiety and stress—a systematic review. Nutrients. 2017;9:429-450.
30. Mauskop A, Altura BT, Altura BM. Serum ionized magnesium levels and serum ionized calcium/ionized magnesium ratios in women with menstrual migraine. Headache. 2002;42:242-248.
31. Facchinetti F, Sances C, Borella P, et al. Magnesium prophylaxis of menstrual migraine: effects on intracellular magnesium. Headache. 1991;31:298-301.
32. Parsay S, Olfati F, Nahidi S. Therapeutic effects of vitamin E on cyclic mastalgia. Breast J. 2009;15:510-514.
33. London RS, Murphy L, Kitlowski KE, et al. Efficacy of alpha-tocopherol in the treatment of the premenstrual syndrome. J Reprod Med. 1987;32:400-404.
34. Whelan AM, Jurgens TM, Naylor H. Herbs, vitamins, and minerals in the treatment of premenstrual syndrome: a systematic review. Can J Clin Pharmacol. 2009;16:e407-e429.
, , , . Selective serotonin reuptake inhibitors for premenstrual syndrome. Cochrane Database Syst Rev. 2013;(6): CD001396.
36. Dimmock P, Wyatt K, Jones P, et al. Efficacy of selective serotonin-reuptake inhibitors in premenstrual syndrome: a systematic review. Lancet. 2000;356:1131-1136.
37. Shah NR, Jones JB, Aperi J, et al. Selective serotonin reuptake inhibitors for premenstrual syndrome and premenstrual dysphoric disorder. Obstet Gynecol. 2008;111:1175-1182.
38. Freeman EW. Luteal phase administration of agents for the treatment of premenstrual dysphoric disorder. CNS Drugs. 2004;18:453-468.
39. Freeman EW, Rickels K, Yonkers KA, et al. Venlafaxine in the treatment of premenstrual dysphoric disorder. Obstet Gynecol. 2001;98:737-744.
40. Lopez LM, Kaptein AA, Helmerhorst FM. Oral contraceptives containing drospirenone for premenstrual syndrome. Cochrane Database Syst Rev. 2012;(2):CD006586.
41. Halbreich U, Freeman EW, Rapkin AJ, et al. Continuous oral levonorgestrel/ethinyl estradiol for treating premenstrual dysphoric disorder. Contraception. 2012;85:19-27.
, , , . Progesterone for premenstrual syndrome. Cochrane Database Syst Rev. 2012;(3):CD003415.
43. Wyatt K, Dimmock P, Jones P, et al. Efficacy of progesterone and progestogens in management of premenstrual syndrome: systematic review. BMJ. 2001;323: 776-780.
44. Dennerstein L, Spencer-Gardner C, Gotts G, et al. Progesterone and the premenstrual syndrome: a double-blind crossover trial. Br Med J (Clin Res Ed). 1985;290:1617-1621.
45. NaProTECHNOLOGY. The Medical and Surgical Practice of NaProTECHNOLOGY. Premenstrual Syndrome: Evaluation and Treatment. Omaha, NE: Pope Paul VI Institute Press. 2004;29:345-368. https://www.naprotechnology.com/naprotext.htm. Accessed January 23, 2020.
46. Dante G, Facchinetti F. Herbal treatments for alleviating premenstrual symptoms: a systematic review. J Psychosom Obstet Gynaecol. 2011;32:42-51.
47. Jarvis CI, Lynch AM, Morin AK. Management strategies for premenstrual syndrome/premenstrual dysphoric disorder. Ann Pharmacother. 2008;42:967-978.
48. Landen M, Eriksson O, Sundblad C, et al. Compounds with affinity for serotonergic receptors in the treatment of premenstrual dysphoria: a comparison of buspirone, nefazodone and placebo. Psychopharmacology (Berl). 2001;155:292-298.
, , , . Non-contraceptive oestrogen-containing preparations for controlling symptoms of premenstrual syndrome . Cochrane Database Syst Rev . 2017 ;( 3) :CD010503.
1. Woods NF, Most A, Dery GK. Prevalence of perimenstrual symptoms. Am J Public Health. 1982;72:1257-1264.
2. Johnson SR, McChesney C, Bean JA. Epidemiology of premenstrual symptoms in a nonclinical sample. 1. Prevalence, natural history and help-seeking behavior. J Repro Med. 1988;33:340-346.
3. Campbell EM, Peterkin D, O’Grady K, et al. Premenstrual symptoms in general practice patients. Prevalence and treatment. J Reprod Med. 1997;42:637-646.
4. O’Brien PM, Bäckström T, Brown C, et al. Towards a consensus on diagnostic criteria, measurement, and trial design of the premenstrual disorders: the ISPMD Montreal consensus. Arch Womens Ment Health. 2011;14:13-21.
5. Epperson CN, Steiner M, Hartlage SA, et al. Premenstrual dysphoric disorder: evidence for a new category for DSM-5. Am J Psychiatry. 2012;169:465-475.
6. American College of Obstetricians and Gynecologists. Guidelines for Women’s Health Care: A Resource Manual. 4th ed. Washington, DC: American College of Obstetricians and Gynecologists; 2014:607-613.
7. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition. Arlington, VA: American Psychiatric Association, 2013.
8. Dennerstein L, Lehert P, Heinemann K. Epidemiology of premenstrual symptoms and disorders. Menopause Int. 2012;18:48-51.
9. Borenstein JE, Dean BB, Yonkers KA, et al. Using the daily record of severity of problems as a screening instrument for premenstrual syndrome. Obstet Gynecol. 2007;109:1068-1075.
10. Steiner M, Macdougall M, Brown E. The premenstrual symptoms screening tool (PSST) for clinicians. Arch Womens Ment Health. 2003;6:203-209.
11. Endicott J, Nee J, Harrison W. Daily Record of Severity of Problems (DRSP): reliability and validity. Arch Womens Ment Health. 2006;9:41-49.
12. Janda C, Kues JN, Andersson G, et al. A symptom diary to assess severe premenstrual syndrome and premenstrual dysphoric disorder. Women Health. 2017;57:837-854.
13. Farasati N, Siassi F, Koohdani F, et al. Western dietary pattern is related to premenstrual syndrome: a case-control study. Brit J Nutr. 2015;114:2016-2021.
14. Purdue-Smithe AC, Manson JE, Hankinson SE, et al. A prospective study of caffeine and coffee intake and premenstrual syndrome. Am J Clin Nutr. 2016;104:499-507.
15. Bertone-Johnson ER, Hankinson SE, Bendich A, et al. Calcium and vitamin D intake and risk of incident premenstrual syndrome. Arch Intern Med. 2005;165:1246-1252.
16. Chocano-Bedoya PO, Manson JE, Hankinson SE, et al. Dietary B vitamin intake and incident premenstrual syndrome. Am J Clin Nutr. 2011;93:1080-1086.
17. Prior JC, Vigna Y. Conditioning exercise and premenstrual symptoms. J Reprod Med. 1987;32:423-428.
18. Aganoff JA, Boyle GJ. Aerobic exercise, mood states, and menstrual cycle symptoms. J Psychosom Res. 1994;38:183-192.
19. El-Lithy A, El-Mazny A, Sabbour A, et al. Effect of aerobic exercise on premenstrual symptoms, haematological and hormonal parameters in young women. J Obstet Gynaecol. 2015;35:389-392.
20. Kroll-Desrosiers AR, Ronnenberg AG, Zagarins SE, et al. Recreational physical activity and premenstrual syndrome in young adult women: a cross-sectional study. PLoS One. 2017;12:1-13.
21. Jang SH, Kim DI, Choi MS. Effects and treatment methods of acupuncture and herbal medicine for premenstrual syndrome/premenstrual dysphoric disorder: systematic review. BMC Complement Altern Med. 2014;14:11.
22. Kim SY, Park HJ, Lee H, et al. Acupuncture for premenstrual syndrome: a systematic review and meta-analysis of randomized controlled trials. BJOG. 2011;118:899-915.
23. Lustyk MK, Gerrish WG, Shaver S, et al. Cognitive-behavioral therapy for premenstrual syndrome and premenstrual dysphoric disorder: a systematic review. Arch Womens Ment Health. 2009;12:85-96.
24. Thys-Jacob S, Starkey P, Bernstein D, et al. Calcium carbonate and the premenstrual syndrome: effects on premenstrual and menstrual syndromes. Am J Obstet Gynecol. 1998;179:444-452.
25. Ghanbari Z, Haghollahi F, Shariat M, et al. Effects of calcium supplement therapy in women with premenstrual syndrome. Taiwan J Obstet Gynecol. 2009;48:124-129.
26. Girman A, Lee R, Kligler B. An integrative medicine approach to premenstrual syndrome. Am J Obstet Gynecol. 2003;188(5 suppl):s56-s65.
27. Wyatt KM, Dimmock PW, Jones PW, et al. Efficacy of vitamin B-6 in the treatment of premenstrual syndrome: systematic review. BMJ. 1999;318:1375-1381.
28. Verkaik S, Kamperman AM, van Westrhenen R, et al. The treatment of premenstrual syndrome with preparations of vitex agnus castus: a systematic review and meta-analysis. Am J Obstet Gynecol. 2017;217:150-166.
29. Boyle NB, Lawton C, Dye L. The effects of magnesium supplementation on subjective anxiety and stress—a systematic review. Nutrients. 2017;9:429-450.
30. Mauskop A, Altura BT, Altura BM. Serum ionized magnesium levels and serum ionized calcium/ionized magnesium ratios in women with menstrual migraine. Headache. 2002;42:242-248.
31. Facchinetti F, Sances C, Borella P, et al. Magnesium prophylaxis of menstrual migraine: effects on intracellular magnesium. Headache. 1991;31:298-301.
32. Parsay S, Olfati F, Nahidi S. Therapeutic effects of vitamin E on cyclic mastalgia. Breast J. 2009;15:510-514.
33. London RS, Murphy L, Kitlowski KE, et al. Efficacy of alpha-tocopherol in the treatment of the premenstrual syndrome. J Reprod Med. 1987;32:400-404.
34. Whelan AM, Jurgens TM, Naylor H. Herbs, vitamins, and minerals in the treatment of premenstrual syndrome: a systematic review. Can J Clin Pharmacol. 2009;16:e407-e429.
, , , . Selective serotonin reuptake inhibitors for premenstrual syndrome. Cochrane Database Syst Rev. 2013;(6): CD001396.
36. Dimmock P, Wyatt K, Jones P, et al. Efficacy of selective serotonin-reuptake inhibitors in premenstrual syndrome: a systematic review. Lancet. 2000;356:1131-1136.
37. Shah NR, Jones JB, Aperi J, et al. Selective serotonin reuptake inhibitors for premenstrual syndrome and premenstrual dysphoric disorder. Obstet Gynecol. 2008;111:1175-1182.
38. Freeman EW. Luteal phase administration of agents for the treatment of premenstrual dysphoric disorder. CNS Drugs. 2004;18:453-468.
39. Freeman EW, Rickels K, Yonkers KA, et al. Venlafaxine in the treatment of premenstrual dysphoric disorder. Obstet Gynecol. 2001;98:737-744.
40. Lopez LM, Kaptein AA, Helmerhorst FM. Oral contraceptives containing drospirenone for premenstrual syndrome. Cochrane Database Syst Rev. 2012;(2):CD006586.
41. Halbreich U, Freeman EW, Rapkin AJ, et al. Continuous oral levonorgestrel/ethinyl estradiol for treating premenstrual dysphoric disorder. Contraception. 2012;85:19-27.
, , , . Progesterone for premenstrual syndrome. Cochrane Database Syst Rev. 2012;(3):CD003415.
43. Wyatt K, Dimmock P, Jones P, et al. Efficacy of progesterone and progestogens in management of premenstrual syndrome: systematic review. BMJ. 2001;323: 776-780.
44. Dennerstein L, Spencer-Gardner C, Gotts G, et al. Progesterone and the premenstrual syndrome: a double-blind crossover trial. Br Med J (Clin Res Ed). 1985;290:1617-1621.
45. NaProTECHNOLOGY. The Medical and Surgical Practice of NaProTECHNOLOGY. Premenstrual Syndrome: Evaluation and Treatment. Omaha, NE: Pope Paul VI Institute Press. 2004;29:345-368. https://www.naprotechnology.com/naprotext.htm. Accessed January 23, 2020.
46. Dante G, Facchinetti F. Herbal treatments for alleviating premenstrual symptoms: a systematic review. J Psychosom Obstet Gynaecol. 2011;32:42-51.
47. Jarvis CI, Lynch AM, Morin AK. Management strategies for premenstrual syndrome/premenstrual dysphoric disorder. Ann Pharmacother. 2008;42:967-978.
48. Landen M, Eriksson O, Sundblad C, et al. Compounds with affinity for serotonergic receptors in the treatment of premenstrual dysphoria: a comparison of buspirone, nefazodone and placebo. Psychopharmacology (Berl). 2001;155:292-298.
, , , . Non-contraceptive oestrogen-containing preparations for controlling symptoms of premenstrual syndrome . Cochrane Database Syst Rev . 2017 ;( 3) :CD010503.
PRACTICE RECOMMENDATIONS
› Start calcium supplementation in all patients who report significant premenstrual symptoms. A
› Add a selective serotonin reuptake inhibitor (SSRI) to calcium supplementationfor patients who have more severe premenstrual psychological symptoms. A
› Consider hormonal treatment options for patients who require treatment beyond calcium and an SSRI. B
› Provide nutrition and exercise information to all patients who report significant premenstrual symptoms. C
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
FDA approves fidaxomicin for treatment of C. difficile–associated diarrhea
The Food and Drug Administration has approved fidaxomicin (Dificid) for the treatment of Clostridioides difficile–associated diarrhea in children aged 6 months and older.
Approval was based on results from SUNSHINE, a phase 3, multicenter, investigator-blind, randomized, parallel-group study in 142 pediatric patients aged between 6 months and 18 years with confirmed C. difficile infection who received either fidaxomicin or vancomycin for 10 days. Clinical response 2 days after the conclusion of treatment was similar in both groups (77.6% for fidaxomicin vs. 70.5% for vancomycin), and fidaxomicin had a superior sustained response 30 days after the conclusion of treatment (68.4% vs. 50.0%).
The safety of fidaxomicin was assessed in a pair of clinical trials involving 136 patients; the most common adverse events were pyrexia, abdominal pain, vomiting, diarrhea, constipation, increased aminotransferases, and rash. Four patients discontinued fidaxomicin treatment because of adverse events, and four patients died during the trials, though all deaths were in patients aged younger than 2 years and seemed to be related to other comorbidities.
“C. difficile is an important cause of health care– and community-associated diarrheal illness in children, and sustained cure is difficult to achieve in some patients. The fidaxomicin pediatric trial was the first randomized, controlled trial of C. difficile infection treatment in children,” Larry K. Kociolek, MD, associate medical director of infection prevention and control at Ann & Robert H. Lurie Children’s Hospital of Chicago, said in the press release from Merck, manufacturer of fidaxomicin.
*This story was updated on 1/27/2020.
The Food and Drug Administration has approved fidaxomicin (Dificid) for the treatment of Clostridioides difficile–associated diarrhea in children aged 6 months and older.
Approval was based on results from SUNSHINE, a phase 3, multicenter, investigator-blind, randomized, parallel-group study in 142 pediatric patients aged between 6 months and 18 years with confirmed C. difficile infection who received either fidaxomicin or vancomycin for 10 days. Clinical response 2 days after the conclusion of treatment was similar in both groups (77.6% for fidaxomicin vs. 70.5% for vancomycin), and fidaxomicin had a superior sustained response 30 days after the conclusion of treatment (68.4% vs. 50.0%).
The safety of fidaxomicin was assessed in a pair of clinical trials involving 136 patients; the most common adverse events were pyrexia, abdominal pain, vomiting, diarrhea, constipation, increased aminotransferases, and rash. Four patients discontinued fidaxomicin treatment because of adverse events, and four patients died during the trials, though all deaths were in patients aged younger than 2 years and seemed to be related to other comorbidities.
“C. difficile is an important cause of health care– and community-associated diarrheal illness in children, and sustained cure is difficult to achieve in some patients. The fidaxomicin pediatric trial was the first randomized, controlled trial of C. difficile infection treatment in children,” Larry K. Kociolek, MD, associate medical director of infection prevention and control at Ann & Robert H. Lurie Children’s Hospital of Chicago, said in the press release from Merck, manufacturer of fidaxomicin.
*This story was updated on 1/27/2020.
The Food and Drug Administration has approved fidaxomicin (Dificid) for the treatment of Clostridioides difficile–associated diarrhea in children aged 6 months and older.
Approval was based on results from SUNSHINE, a phase 3, multicenter, investigator-blind, randomized, parallel-group study in 142 pediatric patients aged between 6 months and 18 years with confirmed C. difficile infection who received either fidaxomicin or vancomycin for 10 days. Clinical response 2 days after the conclusion of treatment was similar in both groups (77.6% for fidaxomicin vs. 70.5% for vancomycin), and fidaxomicin had a superior sustained response 30 days after the conclusion of treatment (68.4% vs. 50.0%).
The safety of fidaxomicin was assessed in a pair of clinical trials involving 136 patients; the most common adverse events were pyrexia, abdominal pain, vomiting, diarrhea, constipation, increased aminotransferases, and rash. Four patients discontinued fidaxomicin treatment because of adverse events, and four patients died during the trials, though all deaths were in patients aged younger than 2 years and seemed to be related to other comorbidities.
“C. difficile is an important cause of health care– and community-associated diarrheal illness in children, and sustained cure is difficult to achieve in some patients. The fidaxomicin pediatric trial was the first randomized, controlled trial of C. difficile infection treatment in children,” Larry K. Kociolek, MD, associate medical director of infection prevention and control at Ann & Robert H. Lurie Children’s Hospital of Chicago, said in the press release from Merck, manufacturer of fidaxomicin.
*This story was updated on 1/27/2020.
New Vascepa indication opens up treatment to millions; “Most significant event since statins”
The newly approved U.S. indication for icosapent ethyl (Vascepa; Amarin) is broadly in line with the entry criteria for the REDUCE-IT trial and includes a large high-risk primary-prevention population, as well as those with established cardiovascular disease (CVD). The drug, thus, could well be used by millions of patients in the United States alone.
The high-dose, purified eicosapentaenoic acid product was approved last week by the Food and Drug Administration for cardiovascular risk reduction among adults already taking maximally tolerated statins with triglyceride levels of 150 mg/dL or higher who have either established CVD or diabetes and two or more additional risk factors for CVD.
The approval is based largely on the REDUCE-IT trial’s finding of a 25% reduction in risk for major adverse cardiovascular events versus placebo. The FDA stated that the approval is the first for an agent with this specific indication.
Noting that it recognizes the need for additional medical treatments for CVD, the FDA says the new approval “will give patients with elevated triglycerides and other important risk factors, including heart disease, stroke, and diabetes, an adjunctive treatment option that can help decrease their risk of cardiovascular events.”
The drug was unanimously recommended for approval by the FDA’s Endocrinologic and Metabolic Drugs Advisory Committee last month. But while the committee all agreed on its use in patients with established CVD, which made up 70% of the REDUCE-IT population, they were divided on whether the indication should be extended to the high-risk primary-prevention population, who made up just 30% of patients in the study.
Nonetheless, the FDA has gone for a broad indication based on the whole REDUCE-IT population.
In a conference call following the approval, Steven Ketchum, PhD, chief scientific officer at Amarin, pointed out that the primary-prevention population stipulated in the new approval differed very slightly from the REDUCE-IT enrollment criteria.
The trial specified that patients with diabetes should be older than 50 with one other cardiovascular risk factor, whereas the approved population is for diabetes and two cardiovascular risk factors. But as these two risk factors are not specified, they could include age, cigarette smoking, hypertension or use of an antihypertensive agent, low HDL cholesterol, high C-reactive protein, body mass index above 25 kg/m2, renal dysfunction, retinopathy, albuminuria, or an ankle branchial index below 0.9, Dr. Ketchum said.
“So while the label asks for two other risk factors, one of these could be age; so we believe the label is actually slightly broader than the REDUCE-IT inclusion criteria, and doctors have been left with significant leeway to decide which risk factors to consider on top of diabetes.”
Deepak Bhatt, MD, the lead investigator of REDUCE-IT, described the Vascepa approval as “the most significant event in the field of cardiovascular prevention since the introduction of statins nearly 3 decades ago.”
He commended the FDA on “a very evidence-based, prescriber-friendly, and most importantly, patient-friendly label,” which he said was in line with guidelines from multiple professional societies that have already incorporated the REDUCE-IT findings for secondary prevention and diabetic primary prevention.
Dr. Bhatt, who is a professor of medicine at Brigham and Women’s Hospital and Harvard Medical School, Boston, said the label essentially matches the REDUCE-IT population.
“The entry criteria for REDUCE-IT was fasting triglycerides greater than or equal to 150 mg/dL, with a 10% variance allowed (giving a minimum triglyceride value of 135 mg/dL). In actuality, we ended up with about 10% of the population with triglycerides between 100 and 150 mg/dL, and they had a similar degree of benefit as those with higher levels,” he reported.
“In the label, the 150 mg/dL does not specify fasting, and in fact many practices have moved away from fasting lipid measurements for the sake of patient comfort,” Dr. Bhatt added. “On average, nonfasting levels are about 50 mg/dL higher, so the label essentially mirrors those we studied, with the FDA applying good common sense and not being overly dogmatic about the exact wording of the trial inclusion criteria.”
No price change foreseen
Vascepa is already on the market for patients with very high triglyceride levels, and the company says it is not increasing the current price of about $300 a month, which is “relatively low, compared to other new breakthrough drugs.” However, it says it expects sales to grow from vastly increased volume based on the new indication.
Dr. Bhatt noted that REDUCE-IT cost-effectiveness data presented at the recent American Heart Association scientific sessions found the drug to be cost saving in the majority of cases. “That is something that is quite rare in cost-effectiveness research,” he said.
“Now, the key challenge is to identify and treat appropriate patients,” Dr. Bhatt noted. He says this task will largely fall on cardiologists, endocrinologists, and primary care physicians, though stroke neurologists, nephrologists, and vascular medicine specialists will also have patients for whom the data are relevant.
“I believe the drug will ultimately be widely prescribed, initially by subspecialists, but by primary care physicians also. It is overall very well tolerated, safe, and easy to use,” he said. “Much like statin prescription started in subspecialty practices but then became quite common in primary care, I envision the same happening with icosapent ethyl.”
Lipid expert Roger Blumenthal, MD, who was not involved in the REDUCE-IT trial, also welcomed the new approval for Vascepa.
“The indication is very appropriate; it is great to have another disease-modifying medication in our prevention toolkit,” Dr. Blumenthal, who is director of the Johns Hopkins Ciccarone Center for the Prevention of Cardiovascular Disease in Baltimore, said in an interview.
Some still unsure
But not everyone is in full agreement with the broad indication granted.
One expert who has reservations is James de Lemos, MD, professor of medicine at the University of Texas, Dallas, who sat on the FDA advisory committee that assessed the drug last month.
“I would have preferred a narrower label for now, limited to the secondary prevention indication, because I felt that REDUCE-IT did not include sufficient numbers of patients to justify the high-risk primary-prevention indication. We need an adequately powered, randomized, controlled trial to establish the risk/benefit and cost/benefit in primary prevention, and with this broad label, I worry there will be little incentive for the company to pursue this,” Dr. de Lemos commented in an interview.
“This is a slippery slope, and we should not allow broad indications that extend to primary prevention for drugs that were studied in mixed secondary- and primary-prevention patients, with the results driven by the secondary-prevention subset. These two subgroups are fundamentally different populations in whom the pathophysiology and the background treatments are very different,” he added.
However, Dr. de Lemos acknowledged that he would use Vascepa for some high-risk primary-prevention patients in his practice – those with diabetes, high triglycerides, and multiple risk factors. “I just wish we had more data coming so that I could be more certain of the benefit in this group,” he said.
Dr. Bhatt disclosed sitting on advisory boards for Cardax, Cereno Scientific, Medscape Cardiology, PhaseBio, and Regado Biosciences; conducting unfunded research in association with FlowCo, Fractyl, Merck, Novo Nordisk, PLx Pharma, and Takeda; and receiving research funding from Abbott, Afimmune, Amarin, Amgen, AstraZeneca, Bayer, Boehringer Ingelheim, Bristol-Myers Squibb, Chiesi, CSL Behring, Eisai, Ethicon, Ferring Pharmaceuticals, and Forest Laboratories. Dr. De Lemos reported receiving grant and consulting income from Roche Diagnostics and Abbott Diagnostics; and consulting for Ortho Clinical Diagnostics. Dr. Blumenthal has disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
The newly approved U.S. indication for icosapent ethyl (Vascepa; Amarin) is broadly in line with the entry criteria for the REDUCE-IT trial and includes a large high-risk primary-prevention population, as well as those with established cardiovascular disease (CVD). The drug, thus, could well be used by millions of patients in the United States alone.
The high-dose, purified eicosapentaenoic acid product was approved last week by the Food and Drug Administration for cardiovascular risk reduction among adults already taking maximally tolerated statins with triglyceride levels of 150 mg/dL or higher who have either established CVD or diabetes and two or more additional risk factors for CVD.
The approval is based largely on the REDUCE-IT trial’s finding of a 25% reduction in risk for major adverse cardiovascular events versus placebo. The FDA stated that the approval is the first for an agent with this specific indication.
Noting that it recognizes the need for additional medical treatments for CVD, the FDA says the new approval “will give patients with elevated triglycerides and other important risk factors, including heart disease, stroke, and diabetes, an adjunctive treatment option that can help decrease their risk of cardiovascular events.”
The drug was unanimously recommended for approval by the FDA’s Endocrinologic and Metabolic Drugs Advisory Committee last month. But while the committee all agreed on its use in patients with established CVD, which made up 70% of the REDUCE-IT population, they were divided on whether the indication should be extended to the high-risk primary-prevention population, who made up just 30% of patients in the study.
Nonetheless, the FDA has gone for a broad indication based on the whole REDUCE-IT population.
In a conference call following the approval, Steven Ketchum, PhD, chief scientific officer at Amarin, pointed out that the primary-prevention population stipulated in the new approval differed very slightly from the REDUCE-IT enrollment criteria.
The trial specified that patients with diabetes should be older than 50 with one other cardiovascular risk factor, whereas the approved population is for diabetes and two cardiovascular risk factors. But as these two risk factors are not specified, they could include age, cigarette smoking, hypertension or use of an antihypertensive agent, low HDL cholesterol, high C-reactive protein, body mass index above 25 kg/m2, renal dysfunction, retinopathy, albuminuria, or an ankle branchial index below 0.9, Dr. Ketchum said.
“So while the label asks for two other risk factors, one of these could be age; so we believe the label is actually slightly broader than the REDUCE-IT inclusion criteria, and doctors have been left with significant leeway to decide which risk factors to consider on top of diabetes.”
Deepak Bhatt, MD, the lead investigator of REDUCE-IT, described the Vascepa approval as “the most significant event in the field of cardiovascular prevention since the introduction of statins nearly 3 decades ago.”
He commended the FDA on “a very evidence-based, prescriber-friendly, and most importantly, patient-friendly label,” which he said was in line with guidelines from multiple professional societies that have already incorporated the REDUCE-IT findings for secondary prevention and diabetic primary prevention.
Dr. Bhatt, who is a professor of medicine at Brigham and Women’s Hospital and Harvard Medical School, Boston, said the label essentially matches the REDUCE-IT population.
“The entry criteria for REDUCE-IT was fasting triglycerides greater than or equal to 150 mg/dL, with a 10% variance allowed (giving a minimum triglyceride value of 135 mg/dL). In actuality, we ended up with about 10% of the population with triglycerides between 100 and 150 mg/dL, and they had a similar degree of benefit as those with higher levels,” he reported.
“In the label, the 150 mg/dL does not specify fasting, and in fact many practices have moved away from fasting lipid measurements for the sake of patient comfort,” Dr. Bhatt added. “On average, nonfasting levels are about 50 mg/dL higher, so the label essentially mirrors those we studied, with the FDA applying good common sense and not being overly dogmatic about the exact wording of the trial inclusion criteria.”
No price change foreseen
Vascepa is already on the market for patients with very high triglyceride levels, and the company says it is not increasing the current price of about $300 a month, which is “relatively low, compared to other new breakthrough drugs.” However, it says it expects sales to grow from vastly increased volume based on the new indication.
Dr. Bhatt noted that REDUCE-IT cost-effectiveness data presented at the recent American Heart Association scientific sessions found the drug to be cost saving in the majority of cases. “That is something that is quite rare in cost-effectiveness research,” he said.
“Now, the key challenge is to identify and treat appropriate patients,” Dr. Bhatt noted. He says this task will largely fall on cardiologists, endocrinologists, and primary care physicians, though stroke neurologists, nephrologists, and vascular medicine specialists will also have patients for whom the data are relevant.
“I believe the drug will ultimately be widely prescribed, initially by subspecialists, but by primary care physicians also. It is overall very well tolerated, safe, and easy to use,” he said. “Much like statin prescription started in subspecialty practices but then became quite common in primary care, I envision the same happening with icosapent ethyl.”
Lipid expert Roger Blumenthal, MD, who was not involved in the REDUCE-IT trial, also welcomed the new approval for Vascepa.
“The indication is very appropriate; it is great to have another disease-modifying medication in our prevention toolkit,” Dr. Blumenthal, who is director of the Johns Hopkins Ciccarone Center for the Prevention of Cardiovascular Disease in Baltimore, said in an interview.
Some still unsure
But not everyone is in full agreement with the broad indication granted.
One expert who has reservations is James de Lemos, MD, professor of medicine at the University of Texas, Dallas, who sat on the FDA advisory committee that assessed the drug last month.
“I would have preferred a narrower label for now, limited to the secondary prevention indication, because I felt that REDUCE-IT did not include sufficient numbers of patients to justify the high-risk primary-prevention indication. We need an adequately powered, randomized, controlled trial to establish the risk/benefit and cost/benefit in primary prevention, and with this broad label, I worry there will be little incentive for the company to pursue this,” Dr. de Lemos commented in an interview.
“This is a slippery slope, and we should not allow broad indications that extend to primary prevention for drugs that were studied in mixed secondary- and primary-prevention patients, with the results driven by the secondary-prevention subset. These two subgroups are fundamentally different populations in whom the pathophysiology and the background treatments are very different,” he added.
However, Dr. de Lemos acknowledged that he would use Vascepa for some high-risk primary-prevention patients in his practice – those with diabetes, high triglycerides, and multiple risk factors. “I just wish we had more data coming so that I could be more certain of the benefit in this group,” he said.
Dr. Bhatt disclosed sitting on advisory boards for Cardax, Cereno Scientific, Medscape Cardiology, PhaseBio, and Regado Biosciences; conducting unfunded research in association with FlowCo, Fractyl, Merck, Novo Nordisk, PLx Pharma, and Takeda; and receiving research funding from Abbott, Afimmune, Amarin, Amgen, AstraZeneca, Bayer, Boehringer Ingelheim, Bristol-Myers Squibb, Chiesi, CSL Behring, Eisai, Ethicon, Ferring Pharmaceuticals, and Forest Laboratories. Dr. De Lemos reported receiving grant and consulting income from Roche Diagnostics and Abbott Diagnostics; and consulting for Ortho Clinical Diagnostics. Dr. Blumenthal has disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
The newly approved U.S. indication for icosapent ethyl (Vascepa; Amarin) is broadly in line with the entry criteria for the REDUCE-IT trial and includes a large high-risk primary-prevention population, as well as those with established cardiovascular disease (CVD). The drug, thus, could well be used by millions of patients in the United States alone.
The high-dose, purified eicosapentaenoic acid product was approved last week by the Food and Drug Administration for cardiovascular risk reduction among adults already taking maximally tolerated statins with triglyceride levels of 150 mg/dL or higher who have either established CVD or diabetes and two or more additional risk factors for CVD.
The approval is based largely on the REDUCE-IT trial’s finding of a 25% reduction in risk for major adverse cardiovascular events versus placebo. The FDA stated that the approval is the first for an agent with this specific indication.
Noting that it recognizes the need for additional medical treatments for CVD, the FDA says the new approval “will give patients with elevated triglycerides and other important risk factors, including heart disease, stroke, and diabetes, an adjunctive treatment option that can help decrease their risk of cardiovascular events.”
The drug was unanimously recommended for approval by the FDA’s Endocrinologic and Metabolic Drugs Advisory Committee last month. But while the committee all agreed on its use in patients with established CVD, which made up 70% of the REDUCE-IT population, they were divided on whether the indication should be extended to the high-risk primary-prevention population, who made up just 30% of patients in the study.
Nonetheless, the FDA has gone for a broad indication based on the whole REDUCE-IT population.
In a conference call following the approval, Steven Ketchum, PhD, chief scientific officer at Amarin, pointed out that the primary-prevention population stipulated in the new approval differed very slightly from the REDUCE-IT enrollment criteria.
The trial specified that patients with diabetes should be older than 50 with one other cardiovascular risk factor, whereas the approved population is for diabetes and two cardiovascular risk factors. But as these two risk factors are not specified, they could include age, cigarette smoking, hypertension or use of an antihypertensive agent, low HDL cholesterol, high C-reactive protein, body mass index above 25 kg/m2, renal dysfunction, retinopathy, albuminuria, or an ankle branchial index below 0.9, Dr. Ketchum said.
“So while the label asks for two other risk factors, one of these could be age; so we believe the label is actually slightly broader than the REDUCE-IT inclusion criteria, and doctors have been left with significant leeway to decide which risk factors to consider on top of diabetes.”
Deepak Bhatt, MD, the lead investigator of REDUCE-IT, described the Vascepa approval as “the most significant event in the field of cardiovascular prevention since the introduction of statins nearly 3 decades ago.”
He commended the FDA on “a very evidence-based, prescriber-friendly, and most importantly, patient-friendly label,” which he said was in line with guidelines from multiple professional societies that have already incorporated the REDUCE-IT findings for secondary prevention and diabetic primary prevention.
Dr. Bhatt, who is a professor of medicine at Brigham and Women’s Hospital and Harvard Medical School, Boston, said the label essentially matches the REDUCE-IT population.
“The entry criteria for REDUCE-IT was fasting triglycerides greater than or equal to 150 mg/dL, with a 10% variance allowed (giving a minimum triglyceride value of 135 mg/dL). In actuality, we ended up with about 10% of the population with triglycerides between 100 and 150 mg/dL, and they had a similar degree of benefit as those with higher levels,” he reported.
“In the label, the 150 mg/dL does not specify fasting, and in fact many practices have moved away from fasting lipid measurements for the sake of patient comfort,” Dr. Bhatt added. “On average, nonfasting levels are about 50 mg/dL higher, so the label essentially mirrors those we studied, with the FDA applying good common sense and not being overly dogmatic about the exact wording of the trial inclusion criteria.”
No price change foreseen
Vascepa is already on the market for patients with very high triglyceride levels, and the company says it is not increasing the current price of about $300 a month, which is “relatively low, compared to other new breakthrough drugs.” However, it says it expects sales to grow from vastly increased volume based on the new indication.
Dr. Bhatt noted that REDUCE-IT cost-effectiveness data presented at the recent American Heart Association scientific sessions found the drug to be cost saving in the majority of cases. “That is something that is quite rare in cost-effectiveness research,” he said.
“Now, the key challenge is to identify and treat appropriate patients,” Dr. Bhatt noted. He says this task will largely fall on cardiologists, endocrinologists, and primary care physicians, though stroke neurologists, nephrologists, and vascular medicine specialists will also have patients for whom the data are relevant.
“I believe the drug will ultimately be widely prescribed, initially by subspecialists, but by primary care physicians also. It is overall very well tolerated, safe, and easy to use,” he said. “Much like statin prescription started in subspecialty practices but then became quite common in primary care, I envision the same happening with icosapent ethyl.”
Lipid expert Roger Blumenthal, MD, who was not involved in the REDUCE-IT trial, also welcomed the new approval for Vascepa.
“The indication is very appropriate; it is great to have another disease-modifying medication in our prevention toolkit,” Dr. Blumenthal, who is director of the Johns Hopkins Ciccarone Center for the Prevention of Cardiovascular Disease in Baltimore, said in an interview.
Some still unsure
But not everyone is in full agreement with the broad indication granted.
One expert who has reservations is James de Lemos, MD, professor of medicine at the University of Texas, Dallas, who sat on the FDA advisory committee that assessed the drug last month.
“I would have preferred a narrower label for now, limited to the secondary prevention indication, because I felt that REDUCE-IT did not include sufficient numbers of patients to justify the high-risk primary-prevention indication. We need an adequately powered, randomized, controlled trial to establish the risk/benefit and cost/benefit in primary prevention, and with this broad label, I worry there will be little incentive for the company to pursue this,” Dr. de Lemos commented in an interview.
“This is a slippery slope, and we should not allow broad indications that extend to primary prevention for drugs that were studied in mixed secondary- and primary-prevention patients, with the results driven by the secondary-prevention subset. These two subgroups are fundamentally different populations in whom the pathophysiology and the background treatments are very different,” he added.
However, Dr. de Lemos acknowledged that he would use Vascepa for some high-risk primary-prevention patients in his practice – those with diabetes, high triglycerides, and multiple risk factors. “I just wish we had more data coming so that I could be more certain of the benefit in this group,” he said.
Dr. Bhatt disclosed sitting on advisory boards for Cardax, Cereno Scientific, Medscape Cardiology, PhaseBio, and Regado Biosciences; conducting unfunded research in association with FlowCo, Fractyl, Merck, Novo Nordisk, PLx Pharma, and Takeda; and receiving research funding from Abbott, Afimmune, Amarin, Amgen, AstraZeneca, Bayer, Boehringer Ingelheim, Bristol-Myers Squibb, Chiesi, CSL Behring, Eisai, Ethicon, Ferring Pharmaceuticals, and Forest Laboratories. Dr. De Lemos reported receiving grant and consulting income from Roche Diagnostics and Abbott Diagnostics; and consulting for Ortho Clinical Diagnostics. Dr. Blumenthal has disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Cardiac biomarkers refine antihypertensive drug initiation decisions
PHILADELPHIA – Incorporation of cardiac biomarkers into current guideline-based decision-making regarding initiation of antihypertensive medication in patients with previously untreated mild or moderate high blood pressure leads to more appropriate and selective matching of intensive blood pressure control with true patient risk, Ambarish Pandey, MD, reported at the American Heart Association scientific sessions.
That’s because the 2017 American College of Cardiology/AHA blood pressure guidelines recommend incorporating the ACC/AHA 10-Year Atherosclerotic Cardiovascular Disease (ASCVD) Risk Calculator into decision making as to whether to start antihypertensive drug therapy in patients with stage 1 hypertension (130-139/80-89 mm Hg), but the risk calculator doesn’t account for the risk of heart failure.
Yet by far the greatest benefit of intensive BP lowering is in reducing the risk of developing heart failure, as demonstrated in the landmark SPRINT trial, which showed that intensive BP lowering achieved much greater risk reduction in new-onset heart failure than in atherosclerotic cardiovascular events.
Thus, there’s a need for better strategies to guide antihypertensive therapy. And therein lies the rationale for incorporating into the risk assessment an individual’s values for N-terminal pro–brain natriuretic peptide (NT-proBNP), which reflects chronic myocardial stress, and high-sensitivity cardiac troponin T (hs-cTnT), which when elevated signals myocardial injury.
“Cardiac biomarkers are intermediate phenotypes from hypertension to future cardiovascular events. They can identify individuals at increased risk for atherosclerotic events, and at even higher risk for heart failure events,” explained Dr. Pandey, a cardiologist at the University of Texas Southwestern Medical Center, Dallas.
He presented a study of 12,987 participants in three major U.S. cohort studies: the Atherosclerosis Risk In Communities (ARIC) study, the Multi-Ethnic Study of Atherosclerosis (MESA), and the Dallas Heart Study. At baseline, none of the participants were on antihypertensive therapy or had known cardiovascular disease. During 10 years of prospective follow-up, 825 of them experienced a first cardiovascular disease event: 251 developed heart failure and 574 had an MI, stroke, or cardiovascular death. Dr. Pandey and his coworkers calculated the cardiovascular event incidence rate and number-needed-to-treat with intensive antihypertensive drug therapy to prevent a first cardiovascular disease event on the basis of whether patients in the various BP categories were positive or negative for one or more biomarkers.
The results
Fifty-four percent of subjects had normal BP, defined in the guidelines as less than 120/80 mm Hg. Another 3% had BP in excess of 160/100 mm Hg. No controversy exists regarding pharmacotherapy in either of these groups: It’s not warranted in the former, essential in the latter.
Another 3,000 individuals had what the ACC/AHA guidelines define as elevated BP, meaning 120-129/<80 mm Hg, or low-risk stage 1 hypertension of 130-139/80-89 mm Hg and a 10-year ASCVD risk score of less than 10%. Initiation of antihypertensive medication in these groups is not recommended in the guidelines. Yet 36% of these individuals had at least one positive cardiac biomarker. And here’s the eye-opening finding: Notably, the 10-year cardiovascular event incidence rate in this biomarker group not currently recommended for antihypertensive pharmacotherapy was 11%, more than double the 4.6% rate in the biomarker-negative group, which in turn was comparable to the 3.8% in the normal BP participants.
Antihypertensive therapy was recommended according to the guidelines in 20% of the total study population, comprising patients with stage 1 hypertension who had an ASCVD risk score of 10% or more as well as those with stage 2 hypertension, defined as BP greater than 140/90 mm Hg but less than 160/100 mm Hg. Forty-eight percent of these subjects were positive for at least one biomarker. Their cardiovascular incidence rate was 15.1%, compared to the 7.9% rate in biomarker-negative individuals.
The estimated number-needed-to-treat (NNT) with intensive blood pressure–lowering therapy to a target systolic BP of less than 120 mm Hg, as in SPRINT, to prevent one cardiovascular event in individuals not currently guideline-recommended for antihypertensive medications was 86 in those who were biomarker-negative. The NNT dropped to 36 in the biomarker-positive subgroup, a far more attractive figure that suggests a reasonable likelihood of benefit from intensive blood pressure control, in Dr. Pandey’s view.
Similarly, among individuals currently recommended for pharmacotherapy initiation, the NNTs were 49 if biomarker-negative, improving to 26 in those positive for one or both biomarkers, which was comparable to the NNT of 22 in the group with blood pressures greater than 160/100 mm Hg. The NNT of 49 in the biomarker-negative subgroup is in a borderline gray zone warranting individualized shared decision-making regarding pharmacotherapy, Dr. Pandey said.
In this study, an elevated hs-cTnT was defined as 6 ng/L or more, while an elevated NT-proBNP was considered to be at least 100 pg/mL.
“It’s noteworthy that the degree of elevation in hs-cTnT and NT-proBNP which were observed in our study were pretty subtle and much below the threshold used for diagnosis of ischemic events or heart failure. Thus, these elevations were largely representative of subtle chronic injury and not acute events,” according to the cardiologist.
One audience member asked if the elevated biomarkers could simply be a surrogate for longer duration of exposure of the heart to high BP. Sure, Dr. Pandey replied, pointing to the 6-year greater average age of the biomarker-positive participants.
“It is likely that biomarker-positive status is capturing the culmination of longstanding exposure. But the thing about hypertension is there are no symptoms that can signal to the patient or the doctor that they have this disease, so testing for the biomarkers can actually capture the high-risk group that may have had hypertension for a long duration but now needs to be treated in order to prevent the advance of downstream adverse events,” he said.
Dr. Pandey reported having no financial conflicts of interest regarding his study, conducted free of commercial support.
SOURCE: Pandey A. AHA 2019 Abstract EP.AOS.521.141
PHILADELPHIA – Incorporation of cardiac biomarkers into current guideline-based decision-making regarding initiation of antihypertensive medication in patients with previously untreated mild or moderate high blood pressure leads to more appropriate and selective matching of intensive blood pressure control with true patient risk, Ambarish Pandey, MD, reported at the American Heart Association scientific sessions.
That’s because the 2017 American College of Cardiology/AHA blood pressure guidelines recommend incorporating the ACC/AHA 10-Year Atherosclerotic Cardiovascular Disease (ASCVD) Risk Calculator into decision making as to whether to start antihypertensive drug therapy in patients with stage 1 hypertension (130-139/80-89 mm Hg), but the risk calculator doesn’t account for the risk of heart failure.
Yet by far the greatest benefit of intensive BP lowering is in reducing the risk of developing heart failure, as demonstrated in the landmark SPRINT trial, which showed that intensive BP lowering achieved much greater risk reduction in new-onset heart failure than in atherosclerotic cardiovascular events.
Thus, there’s a need for better strategies to guide antihypertensive therapy. And therein lies the rationale for incorporating into the risk assessment an individual’s values for N-terminal pro–brain natriuretic peptide (NT-proBNP), which reflects chronic myocardial stress, and high-sensitivity cardiac troponin T (hs-cTnT), which when elevated signals myocardial injury.
“Cardiac biomarkers are intermediate phenotypes from hypertension to future cardiovascular events. They can identify individuals at increased risk for atherosclerotic events, and at even higher risk for heart failure events,” explained Dr. Pandey, a cardiologist at the University of Texas Southwestern Medical Center, Dallas.
He presented a study of 12,987 participants in three major U.S. cohort studies: the Atherosclerosis Risk In Communities (ARIC) study, the Multi-Ethnic Study of Atherosclerosis (MESA), and the Dallas Heart Study. At baseline, none of the participants were on antihypertensive therapy or had known cardiovascular disease. During 10 years of prospective follow-up, 825 of them experienced a first cardiovascular disease event: 251 developed heart failure and 574 had an MI, stroke, or cardiovascular death. Dr. Pandey and his coworkers calculated the cardiovascular event incidence rate and number-needed-to-treat with intensive antihypertensive drug therapy to prevent a first cardiovascular disease event on the basis of whether patients in the various BP categories were positive or negative for one or more biomarkers.
The results
Fifty-four percent of subjects had normal BP, defined in the guidelines as less than 120/80 mm Hg. Another 3% had BP in excess of 160/100 mm Hg. No controversy exists regarding pharmacotherapy in either of these groups: It’s not warranted in the former, essential in the latter.
Another 3,000 individuals had what the ACC/AHA guidelines define as elevated BP, meaning 120-129/<80 mm Hg, or low-risk stage 1 hypertension of 130-139/80-89 mm Hg and a 10-year ASCVD risk score of less than 10%. Initiation of antihypertensive medication in these groups is not recommended in the guidelines. Yet 36% of these individuals had at least one positive cardiac biomarker. And here’s the eye-opening finding: Notably, the 10-year cardiovascular event incidence rate in this biomarker group not currently recommended for antihypertensive pharmacotherapy was 11%, more than double the 4.6% rate in the biomarker-negative group, which in turn was comparable to the 3.8% in the normal BP participants.
Antihypertensive therapy was recommended according to the guidelines in 20% of the total study population, comprising patients with stage 1 hypertension who had an ASCVD risk score of 10% or more as well as those with stage 2 hypertension, defined as BP greater than 140/90 mm Hg but less than 160/100 mm Hg. Forty-eight percent of these subjects were positive for at least one biomarker. Their cardiovascular incidence rate was 15.1%, compared to the 7.9% rate in biomarker-negative individuals.
The estimated number-needed-to-treat (NNT) with intensive blood pressure–lowering therapy to a target systolic BP of less than 120 mm Hg, as in SPRINT, to prevent one cardiovascular event in individuals not currently guideline-recommended for antihypertensive medications was 86 in those who were biomarker-negative. The NNT dropped to 36 in the biomarker-positive subgroup, a far more attractive figure that suggests a reasonable likelihood of benefit from intensive blood pressure control, in Dr. Pandey’s view.
Similarly, among individuals currently recommended for pharmacotherapy initiation, the NNTs were 49 if biomarker-negative, improving to 26 in those positive for one or both biomarkers, which was comparable to the NNT of 22 in the group with blood pressures greater than 160/100 mm Hg. The NNT of 49 in the biomarker-negative subgroup is in a borderline gray zone warranting individualized shared decision-making regarding pharmacotherapy, Dr. Pandey said.
In this study, an elevated hs-cTnT was defined as 6 ng/L or more, while an elevated NT-proBNP was considered to be at least 100 pg/mL.
“It’s noteworthy that the degree of elevation in hs-cTnT and NT-proBNP which were observed in our study were pretty subtle and much below the threshold used for diagnosis of ischemic events or heart failure. Thus, these elevations were largely representative of subtle chronic injury and not acute events,” according to the cardiologist.
One audience member asked if the elevated biomarkers could simply be a surrogate for longer duration of exposure of the heart to high BP. Sure, Dr. Pandey replied, pointing to the 6-year greater average age of the biomarker-positive participants.
“It is likely that biomarker-positive status is capturing the culmination of longstanding exposure. But the thing about hypertension is there are no symptoms that can signal to the patient or the doctor that they have this disease, so testing for the biomarkers can actually capture the high-risk group that may have had hypertension for a long duration but now needs to be treated in order to prevent the advance of downstream adverse events,” he said.
Dr. Pandey reported having no financial conflicts of interest regarding his study, conducted free of commercial support.
SOURCE: Pandey A. AHA 2019 Abstract EP.AOS.521.141
PHILADELPHIA – Incorporation of cardiac biomarkers into current guideline-based decision-making regarding initiation of antihypertensive medication in patients with previously untreated mild or moderate high blood pressure leads to more appropriate and selective matching of intensive blood pressure control with true patient risk, Ambarish Pandey, MD, reported at the American Heart Association scientific sessions.
That’s because the 2017 American College of Cardiology/AHA blood pressure guidelines recommend incorporating the ACC/AHA 10-Year Atherosclerotic Cardiovascular Disease (ASCVD) Risk Calculator into decision making as to whether to start antihypertensive drug therapy in patients with stage 1 hypertension (130-139/80-89 mm Hg), but the risk calculator doesn’t account for the risk of heart failure.
Yet by far the greatest benefit of intensive BP lowering is in reducing the risk of developing heart failure, as demonstrated in the landmark SPRINT trial, which showed that intensive BP lowering achieved much greater risk reduction in new-onset heart failure than in atherosclerotic cardiovascular events.
Thus, there’s a need for better strategies to guide antihypertensive therapy. And therein lies the rationale for incorporating into the risk assessment an individual’s values for N-terminal pro–brain natriuretic peptide (NT-proBNP), which reflects chronic myocardial stress, and high-sensitivity cardiac troponin T (hs-cTnT), which when elevated signals myocardial injury.
“Cardiac biomarkers are intermediate phenotypes from hypertension to future cardiovascular events. They can identify individuals at increased risk for atherosclerotic events, and at even higher risk for heart failure events,” explained Dr. Pandey, a cardiologist at the University of Texas Southwestern Medical Center, Dallas.
He presented a study of 12,987 participants in three major U.S. cohort studies: the Atherosclerosis Risk In Communities (ARIC) study, the Multi-Ethnic Study of Atherosclerosis (MESA), and the Dallas Heart Study. At baseline, none of the participants were on antihypertensive therapy or had known cardiovascular disease. During 10 years of prospective follow-up, 825 of them experienced a first cardiovascular disease event: 251 developed heart failure and 574 had an MI, stroke, or cardiovascular death. Dr. Pandey and his coworkers calculated the cardiovascular event incidence rate and number-needed-to-treat with intensive antihypertensive drug therapy to prevent a first cardiovascular disease event on the basis of whether patients in the various BP categories were positive or negative for one or more biomarkers.
The results
Fifty-four percent of subjects had normal BP, defined in the guidelines as less than 120/80 mm Hg. Another 3% had BP in excess of 160/100 mm Hg. No controversy exists regarding pharmacotherapy in either of these groups: It’s not warranted in the former, essential in the latter.
Another 3,000 individuals had what the ACC/AHA guidelines define as elevated BP, meaning 120-129/<80 mm Hg, or low-risk stage 1 hypertension of 130-139/80-89 mm Hg and a 10-year ASCVD risk score of less than 10%. Initiation of antihypertensive medication in these groups is not recommended in the guidelines. Yet 36% of these individuals had at least one positive cardiac biomarker. And here’s the eye-opening finding: Notably, the 10-year cardiovascular event incidence rate in this biomarker group not currently recommended for antihypertensive pharmacotherapy was 11%, more than double the 4.6% rate in the biomarker-negative group, which in turn was comparable to the 3.8% in the normal BP participants.
Antihypertensive therapy was recommended according to the guidelines in 20% of the total study population, comprising patients with stage 1 hypertension who had an ASCVD risk score of 10% or more as well as those with stage 2 hypertension, defined as BP greater than 140/90 mm Hg but less than 160/100 mm Hg. Forty-eight percent of these subjects were positive for at least one biomarker. Their cardiovascular incidence rate was 15.1%, compared to the 7.9% rate in biomarker-negative individuals.
The estimated number-needed-to-treat (NNT) with intensive blood pressure–lowering therapy to a target systolic BP of less than 120 mm Hg, as in SPRINT, to prevent one cardiovascular event in individuals not currently guideline-recommended for antihypertensive medications was 86 in those who were biomarker-negative. The NNT dropped to 36 in the biomarker-positive subgroup, a far more attractive figure that suggests a reasonable likelihood of benefit from intensive blood pressure control, in Dr. Pandey’s view.
Similarly, among individuals currently recommended for pharmacotherapy initiation, the NNTs were 49 if biomarker-negative, improving to 26 in those positive for one or both biomarkers, which was comparable to the NNT of 22 in the group with blood pressures greater than 160/100 mm Hg. The NNT of 49 in the biomarker-negative subgroup is in a borderline gray zone warranting individualized shared decision-making regarding pharmacotherapy, Dr. Pandey said.
In this study, an elevated hs-cTnT was defined as 6 ng/L or more, while an elevated NT-proBNP was considered to be at least 100 pg/mL.
“It’s noteworthy that the degree of elevation in hs-cTnT and NT-proBNP which were observed in our study were pretty subtle and much below the threshold used for diagnosis of ischemic events or heart failure. Thus, these elevations were largely representative of subtle chronic injury and not acute events,” according to the cardiologist.
One audience member asked if the elevated biomarkers could simply be a surrogate for longer duration of exposure of the heart to high BP. Sure, Dr. Pandey replied, pointing to the 6-year greater average age of the biomarker-positive participants.
“It is likely that biomarker-positive status is capturing the culmination of longstanding exposure. But the thing about hypertension is there are no symptoms that can signal to the patient or the doctor that they have this disease, so testing for the biomarkers can actually capture the high-risk group that may have had hypertension for a long duration but now needs to be treated in order to prevent the advance of downstream adverse events,” he said.
Dr. Pandey reported having no financial conflicts of interest regarding his study, conducted free of commercial support.
SOURCE: Pandey A. AHA 2019 Abstract EP.AOS.521.141
REPORTING FROM AHA 2019
Value analysis of JAK inhibitors for RA hampered by limited data
Adequate evidence shows that adding a Janus kinase (JAK) inhibitor to conventional disease-modifying antirheumatic drug therapy provides a net health benefit for patients with rheumatoid arthritis, compared with conventional drugs alone, according to a report by an independent research institute. But the long-term economic value of JAK inhibitors for rheumatoid arthritis is less clear, the report by the Institute for Clinical and Economic Review (ICER) indicates.
ICER on Jan. 9 released a finalized report and policy recommendations on JAK inhibitors and biosimilars for rheumatoid arthritis. The report reviews current evidence for JAK inhibitors for adults with moderately active to severely active rheumatoid arthritis.
Since the nonprofit’s 2017 review of targeted immune modulators for rheumatoid arthritis, two JAK inhibitors, baricitinib (Olumiant) and upadacitinib (Rinvoq), were approved by the Food and Drug Administration. At a December 2019 public meeting of the California Technology Assessment Forum (CTAF), one of ICER’s independent evidence appraisal committees, panelists reviewed recent evidence.
A pricey comparator
In ICER’s analysis, the JAK inhibitor upadacitinib reached common thresholds for cost-effectiveness when compared with adalimumab (Humira). Nevertheless, the 14 members of the independent evidence appraisal committee voted that upadacitinib’s long-term economic value was “low” (8 votes) or “intermediate” (6 votes). Concerns about the generalizability of phase 3 clinical trial data to patients in the real world were among the reservations noted by panelists. Furthermore, “legitimate questions remain about whether or not adalimumab, launched 17 years ago, is fairly priced to begin with,” Pamela Bradt, MD, MPH, ICER’s chief scientific officer, said in a news release.
The panel did not vote on the economic value of tofacitinib (Xeljanz) or baricitinib, the two other JAK inhibitors that are approved for rheumatoid arthritis, because head-to-head evidence against adalimumab was insufficient, ICER said.
“Rheumatoid arthritis is a progressively disabling condition, and patients are fortunate to have multiple therapy options – including biosimilars – that effectively slow disease progression,” Dr. Bradt said. “Many economists might expect medicines to become more affordable in an increasingly crowded therapeutic class; however, because the current rebate structure has erected barriers between patients and several emerging RA therapies, traditional market dynamics have been unable to drive down prices.”
Weighing efficacy and cost
Panelists found that the net health benefit provided by upadacitinib is superior to that provided by adalimumab. At the same time, they said that there is insufficient head-to-head evidence to distinguish between the net health benefit of upadacitinib and tofacitinib or to demonstrate that tofacitinib is superior to adalimumab. Evidence comparing baricitinib to adalimumab does not exist.
CTAF members unanimously agreed that adequate evidence demonstrates that the biosimilar infliximab-dyyb (Inflectra) is clinically equivalent to its reference biologic, infliximab (Remicade).
Economic modeling demonstrated that upadacitinib plus a conventional drug achieves marginally higher quality of life than adalimumab plus a conventional drug does, at similar costs. “Based on this comparison with adalimumab, ICER’s value-based price benchmark range for upadacitinib is between $44,000 and $45,000,” according to the ICER news release. “This benchmark represents a 25% discount off of upadacitinib’s annual list price of $59,860, a suggested discount that is consistent with the rebates we assume the manufacturer is currently offering.”
After the voting session, various experts, including clinicians, patient advocates, and representatives from manufacturers and insurance companies, made the following policy recommendations:
- Regulatory intervention may be needed to ensure that drug prices do not continue to increase further from reasonable alignment with added benefits for patients.
- Insurers, pharmacy benefit managers, and employers should increase transparency around the role of discounts and rebates in formulary design.
- Policymakers should aim to create a system that rewards lower-priced biosimilar treatment options.
The findings of the clinical review by the Institute for Clinical and Economic Review (ICER) are generally in line with our clinical perceptions. We have an increasing number of treatment options for our RA patients, and the results of this review support the efficacy of tofacitinib and upadacitinib, compared with currently available biologic treatments. While ICER’s voting panel did find the data supported the superiority of upadacitinib over adalimumab, the cost analysis notes a WAC (wholesale acquisition cost) for upadacitinib of $59,860. While at expected discounted rates it is felt to be cost effective when compared with adalimumab, it is difficult to know what this means since ICER found adalimumab itself not to be cost effective, compared with conventional synthetic disease-modifying antirheumatic drugs (csDMARDs), in its 2017 review.
ICER’s focus is drug pricing and cost effectiveness, so obviously our biologic drugs are in the institute’s crosshairs. This review provided context for a policy roundtable discussion that included patient, payer, and manufacturer input, as well as American College of Rheumatology (ACR) input. We are thankful that ACR had a seat at the table, and thankful ICER is attempting to bring light to the important issues and barriers that perpetuate high drug prices in our marketplace. The discussion was wide ranging but focused on step-edit policies, the role of pharmacy benefit managers (PBMs) in perpetuating high drug prices and the relatively slow uptake of biosimilars in our marketplace.
These issues are critical to every practicing rheumatologist because we each deal daily with the hassles of step-edit/fail-first policies, which hijack our otherwise thoughtful and evidence-based decision making regarding the best treatments for our patients. We know how much (unreimbursed) time it takes our staff to sort through these step edits and prior authorizations, and we have seen recent data regarding how these policies delay care and harm patients. We were thankful to see ICER validate these concerns and note that their suggested guidelines for rational step therapy somewhat mirror those in the Safe Step Act, which ACR supports on a federal legislative level. ACR continues to vigorously support the grandfathering of any patient on an effective treatment, regardless of changes in insurance or formulary; this was an issue of robust debate at their meeting, and this patient-centric position is not uniformly held among policymakers, unfortunately.
ACR agrees with ICER’s conclusion that transparency in the PBM system regarding rebates should be promoted and that opaque rebate negotiations between PBMs and manufacturers both incentivize higher prices and block access to the marketplace for cheaper biosimilar options.
Additionally, ICER and ACR agree about the critical role that biosimilar uptake will play in controlling drug costs. While we do not yet have any biosimilars that have been deemed interchangeable by the Food and Drug Administration, we agree with ICER that data regarding comparable efficacy and safety of biosimilars to their originator products is very reassuring. While the decision to switch to a biosimilar should be an individual decision between a provider and patient, and while we recognize with frustration that many FDA-approved biosimilars are not commercially available because of patent law, it is clear that the current costs of our biologic drugs are not sustainable and the uptake of biosimilars will be critical if we hope our health care economy can continue to support coverage of these life-changing drugs in years to come. We agree with ICER that it is incumbent upon prescribers to reassure our patients regarding the safety and efficacy of these drugs.
Christopher Phillips, MD , is a community rheumatologist in Paducah, Ky., who serves as chair of the insurance subcommittee of the ACR, under the guidance of the Committee on Rheumatologic Care. He attended the initial ICER rheumatoid arthritis review meeting in 2017 on behalf of ACR. In 2019, Dr. Phillips served as a reviewer and clinical expert to the ICER panel and participated in the policy roundtable discussion.
The findings of the clinical review by the Institute for Clinical and Economic Review (ICER) are generally in line with our clinical perceptions. We have an increasing number of treatment options for our RA patients, and the results of this review support the efficacy of tofacitinib and upadacitinib, compared with currently available biologic treatments. While ICER’s voting panel did find the data supported the superiority of upadacitinib over adalimumab, the cost analysis notes a WAC (wholesale acquisition cost) for upadacitinib of $59,860. While at expected discounted rates it is felt to be cost effective when compared with adalimumab, it is difficult to know what this means since ICER found adalimumab itself not to be cost effective, compared with conventional synthetic disease-modifying antirheumatic drugs (csDMARDs), in its 2017 review.
ICER’s focus is drug pricing and cost effectiveness, so obviously our biologic drugs are in the institute’s crosshairs. This review provided context for a policy roundtable discussion that included patient, payer, and manufacturer input, as well as American College of Rheumatology (ACR) input. We are thankful that ACR had a seat at the table, and thankful ICER is attempting to bring light to the important issues and barriers that perpetuate high drug prices in our marketplace. The discussion was wide ranging but focused on step-edit policies, the role of pharmacy benefit managers (PBMs) in perpetuating high drug prices and the relatively slow uptake of biosimilars in our marketplace.
These issues are critical to every practicing rheumatologist because we each deal daily with the hassles of step-edit/fail-first policies, which hijack our otherwise thoughtful and evidence-based decision making regarding the best treatments for our patients. We know how much (unreimbursed) time it takes our staff to sort through these step edits and prior authorizations, and we have seen recent data regarding how these policies delay care and harm patients. We were thankful to see ICER validate these concerns and note that their suggested guidelines for rational step therapy somewhat mirror those in the Safe Step Act, which ACR supports on a federal legislative level. ACR continues to vigorously support the grandfathering of any patient on an effective treatment, regardless of changes in insurance or formulary; this was an issue of robust debate at their meeting, and this patient-centric position is not uniformly held among policymakers, unfortunately.
ACR agrees with ICER’s conclusion that transparency in the PBM system regarding rebates should be promoted and that opaque rebate negotiations between PBMs and manufacturers both incentivize higher prices and block access to the marketplace for cheaper biosimilar options.
Additionally, ICER and ACR agree about the critical role that biosimilar uptake will play in controlling drug costs. While we do not yet have any biosimilars that have been deemed interchangeable by the Food and Drug Administration, we agree with ICER that data regarding comparable efficacy and safety of biosimilars to their originator products is very reassuring. While the decision to switch to a biosimilar should be an individual decision between a provider and patient, and while we recognize with frustration that many FDA-approved biosimilars are not commercially available because of patent law, it is clear that the current costs of our biologic drugs are not sustainable and the uptake of biosimilars will be critical if we hope our health care economy can continue to support coverage of these life-changing drugs in years to come. We agree with ICER that it is incumbent upon prescribers to reassure our patients regarding the safety and efficacy of these drugs.
Christopher Phillips, MD , is a community rheumatologist in Paducah, Ky., who serves as chair of the insurance subcommittee of the ACR, under the guidance of the Committee on Rheumatologic Care. He attended the initial ICER rheumatoid arthritis review meeting in 2017 on behalf of ACR. In 2019, Dr. Phillips served as a reviewer and clinical expert to the ICER panel and participated in the policy roundtable discussion.
The findings of the clinical review by the Institute for Clinical and Economic Review (ICER) are generally in line with our clinical perceptions. We have an increasing number of treatment options for our RA patients, and the results of this review support the efficacy of tofacitinib and upadacitinib, compared with currently available biologic treatments. While ICER’s voting panel did find the data supported the superiority of upadacitinib over adalimumab, the cost analysis notes a WAC (wholesale acquisition cost) for upadacitinib of $59,860. While at expected discounted rates it is felt to be cost effective when compared with adalimumab, it is difficult to know what this means since ICER found adalimumab itself not to be cost effective, compared with conventional synthetic disease-modifying antirheumatic drugs (csDMARDs), in its 2017 review.
ICER’s focus is drug pricing and cost effectiveness, so obviously our biologic drugs are in the institute’s crosshairs. This review provided context for a policy roundtable discussion that included patient, payer, and manufacturer input, as well as American College of Rheumatology (ACR) input. We are thankful that ACR had a seat at the table, and thankful ICER is attempting to bring light to the important issues and barriers that perpetuate high drug prices in our marketplace. The discussion was wide ranging but focused on step-edit policies, the role of pharmacy benefit managers (PBMs) in perpetuating high drug prices and the relatively slow uptake of biosimilars in our marketplace.
These issues are critical to every practicing rheumatologist because we each deal daily with the hassles of step-edit/fail-first policies, which hijack our otherwise thoughtful and evidence-based decision making regarding the best treatments for our patients. We know how much (unreimbursed) time it takes our staff to sort through these step edits and prior authorizations, and we have seen recent data regarding how these policies delay care and harm patients. We were thankful to see ICER validate these concerns and note that their suggested guidelines for rational step therapy somewhat mirror those in the Safe Step Act, which ACR supports on a federal legislative level. ACR continues to vigorously support the grandfathering of any patient on an effective treatment, regardless of changes in insurance or formulary; this was an issue of robust debate at their meeting, and this patient-centric position is not uniformly held among policymakers, unfortunately.
ACR agrees with ICER’s conclusion that transparency in the PBM system regarding rebates should be promoted and that opaque rebate negotiations between PBMs and manufacturers both incentivize higher prices and block access to the marketplace for cheaper biosimilar options.
Additionally, ICER and ACR agree about the critical role that biosimilar uptake will play in controlling drug costs. While we do not yet have any biosimilars that have been deemed interchangeable by the Food and Drug Administration, we agree with ICER that data regarding comparable efficacy and safety of biosimilars to their originator products is very reassuring. While the decision to switch to a biosimilar should be an individual decision between a provider and patient, and while we recognize with frustration that many FDA-approved biosimilars are not commercially available because of patent law, it is clear that the current costs of our biologic drugs are not sustainable and the uptake of biosimilars will be critical if we hope our health care economy can continue to support coverage of these life-changing drugs in years to come. We agree with ICER that it is incumbent upon prescribers to reassure our patients regarding the safety and efficacy of these drugs.
Christopher Phillips, MD , is a community rheumatologist in Paducah, Ky., who serves as chair of the insurance subcommittee of the ACR, under the guidance of the Committee on Rheumatologic Care. He attended the initial ICER rheumatoid arthritis review meeting in 2017 on behalf of ACR. In 2019, Dr. Phillips served as a reviewer and clinical expert to the ICER panel and participated in the policy roundtable discussion.
Adequate evidence shows that adding a Janus kinase (JAK) inhibitor to conventional disease-modifying antirheumatic drug therapy provides a net health benefit for patients with rheumatoid arthritis, compared with conventional drugs alone, according to a report by an independent research institute. But the long-term economic value of JAK inhibitors for rheumatoid arthritis is less clear, the report by the Institute for Clinical and Economic Review (ICER) indicates.
ICER on Jan. 9 released a finalized report and policy recommendations on JAK inhibitors and biosimilars for rheumatoid arthritis. The report reviews current evidence for JAK inhibitors for adults with moderately active to severely active rheumatoid arthritis.
Since the nonprofit’s 2017 review of targeted immune modulators for rheumatoid arthritis, two JAK inhibitors, baricitinib (Olumiant) and upadacitinib (Rinvoq), were approved by the Food and Drug Administration. At a December 2019 public meeting of the California Technology Assessment Forum (CTAF), one of ICER’s independent evidence appraisal committees, panelists reviewed recent evidence.
A pricey comparator
In ICER’s analysis, the JAK inhibitor upadacitinib reached common thresholds for cost-effectiveness when compared with adalimumab (Humira). Nevertheless, the 14 members of the independent evidence appraisal committee voted that upadacitinib’s long-term economic value was “low” (8 votes) or “intermediate” (6 votes). Concerns about the generalizability of phase 3 clinical trial data to patients in the real world were among the reservations noted by panelists. Furthermore, “legitimate questions remain about whether or not adalimumab, launched 17 years ago, is fairly priced to begin with,” Pamela Bradt, MD, MPH, ICER’s chief scientific officer, said in a news release.
The panel did not vote on the economic value of tofacitinib (Xeljanz) or baricitinib, the two other JAK inhibitors that are approved for rheumatoid arthritis, because head-to-head evidence against adalimumab was insufficient, ICER said.
“Rheumatoid arthritis is a progressively disabling condition, and patients are fortunate to have multiple therapy options – including biosimilars – that effectively slow disease progression,” Dr. Bradt said. “Many economists might expect medicines to become more affordable in an increasingly crowded therapeutic class; however, because the current rebate structure has erected barriers between patients and several emerging RA therapies, traditional market dynamics have been unable to drive down prices.”
Weighing efficacy and cost
Panelists found that the net health benefit provided by upadacitinib is superior to that provided by adalimumab. At the same time, they said that there is insufficient head-to-head evidence to distinguish between the net health benefit of upadacitinib and tofacitinib or to demonstrate that tofacitinib is superior to adalimumab. Evidence comparing baricitinib to adalimumab does not exist.
CTAF members unanimously agreed that adequate evidence demonstrates that the biosimilar infliximab-dyyb (Inflectra) is clinically equivalent to its reference biologic, infliximab (Remicade).
Economic modeling demonstrated that upadacitinib plus a conventional drug achieves marginally higher quality of life than adalimumab plus a conventional drug does, at similar costs. “Based on this comparison with adalimumab, ICER’s value-based price benchmark range for upadacitinib is between $44,000 and $45,000,” according to the ICER news release. “This benchmark represents a 25% discount off of upadacitinib’s annual list price of $59,860, a suggested discount that is consistent with the rebates we assume the manufacturer is currently offering.”
After the voting session, various experts, including clinicians, patient advocates, and representatives from manufacturers and insurance companies, made the following policy recommendations:
- Regulatory intervention may be needed to ensure that drug prices do not continue to increase further from reasonable alignment with added benefits for patients.
- Insurers, pharmacy benefit managers, and employers should increase transparency around the role of discounts and rebates in formulary design.
- Policymakers should aim to create a system that rewards lower-priced biosimilar treatment options.
Adequate evidence shows that adding a Janus kinase (JAK) inhibitor to conventional disease-modifying antirheumatic drug therapy provides a net health benefit for patients with rheumatoid arthritis, compared with conventional drugs alone, according to a report by an independent research institute. But the long-term economic value of JAK inhibitors for rheumatoid arthritis is less clear, the report by the Institute for Clinical and Economic Review (ICER) indicates.
ICER on Jan. 9 released a finalized report and policy recommendations on JAK inhibitors and biosimilars for rheumatoid arthritis. The report reviews current evidence for JAK inhibitors for adults with moderately active to severely active rheumatoid arthritis.
Since the nonprofit’s 2017 review of targeted immune modulators for rheumatoid arthritis, two JAK inhibitors, baricitinib (Olumiant) and upadacitinib (Rinvoq), were approved by the Food and Drug Administration. At a December 2019 public meeting of the California Technology Assessment Forum (CTAF), one of ICER’s independent evidence appraisal committees, panelists reviewed recent evidence.
A pricey comparator
In ICER’s analysis, the JAK inhibitor upadacitinib reached common thresholds for cost-effectiveness when compared with adalimumab (Humira). Nevertheless, the 14 members of the independent evidence appraisal committee voted that upadacitinib’s long-term economic value was “low” (8 votes) or “intermediate” (6 votes). Concerns about the generalizability of phase 3 clinical trial data to patients in the real world were among the reservations noted by panelists. Furthermore, “legitimate questions remain about whether or not adalimumab, launched 17 years ago, is fairly priced to begin with,” Pamela Bradt, MD, MPH, ICER’s chief scientific officer, said in a news release.
The panel did not vote on the economic value of tofacitinib (Xeljanz) or baricitinib, the two other JAK inhibitors that are approved for rheumatoid arthritis, because head-to-head evidence against adalimumab was insufficient, ICER said.
“Rheumatoid arthritis is a progressively disabling condition, and patients are fortunate to have multiple therapy options – including biosimilars – that effectively slow disease progression,” Dr. Bradt said. “Many economists might expect medicines to become more affordable in an increasingly crowded therapeutic class; however, because the current rebate structure has erected barriers between patients and several emerging RA therapies, traditional market dynamics have been unable to drive down prices.”
Weighing efficacy and cost
Panelists found that the net health benefit provided by upadacitinib is superior to that provided by adalimumab. At the same time, they said that there is insufficient head-to-head evidence to distinguish between the net health benefit of upadacitinib and tofacitinib or to demonstrate that tofacitinib is superior to adalimumab. Evidence comparing baricitinib to adalimumab does not exist.
CTAF members unanimously agreed that adequate evidence demonstrates that the biosimilar infliximab-dyyb (Inflectra) is clinically equivalent to its reference biologic, infliximab (Remicade).
Economic modeling demonstrated that upadacitinib plus a conventional drug achieves marginally higher quality of life than adalimumab plus a conventional drug does, at similar costs. “Based on this comparison with adalimumab, ICER’s value-based price benchmark range for upadacitinib is between $44,000 and $45,000,” according to the ICER news release. “This benchmark represents a 25% discount off of upadacitinib’s annual list price of $59,860, a suggested discount that is consistent with the rebates we assume the manufacturer is currently offering.”
After the voting session, various experts, including clinicians, patient advocates, and representatives from manufacturers and insurance companies, made the following policy recommendations:
- Regulatory intervention may be needed to ensure that drug prices do not continue to increase further from reasonable alignment with added benefits for patients.
- Insurers, pharmacy benefit managers, and employers should increase transparency around the role of discounts and rebates in formulary design.
- Policymakers should aim to create a system that rewards lower-priced biosimilar treatment options.