User login
Lorcaserin withdrawn from U.S. market due to cancer risk
The Food and Drug Administration asked Eisai to voluntary withdraw the weight-loss drug lorcaserin (Belviq and Belviq XR) on Feb. 13 after a post-marketing trial with more than 12,000 subjects revealed an increased occurrence of cancer.
In a Drug Safety Communication, the agency said “health care professionals should stop prescribing and dispensing lorcaserin to patients. Contact patients currently taking lorcaserin, inform them of the increased occurrence of cancer seen in the clinical trial, and ask them to stop taking the medicine. Discuss alternative weight-loss medicines or strategies with your patients.”
Eisai is complying with the withdrawal request.
The decision is based on the agency’s review of the 5-year trial, which was designed to evaluate cardiac risk with the drug and ended in June 2018. In total, 7.7% of patients randomized to 10 mg lorcaserin twice daily were diagnosed with 520 primary cancers, compared with 7.1% of placebo subjects diagnosed with 470 cancers, over a median follow-up of 3 years and 3 months. There was one additional cancer observed for every 470 patients treated for 1 year.
“There was no apparent difference in the incidence of cancer over the initial months of treatment, but the imbalance increased with longer duration on lorcaserin,” FDA said. Pancreatic, colorectal, and lung cancers were among those diagnosed.
In short, “we believe that the risks of lorcaserin outweigh its benefits based on our completed review of” the data, the agency said. The FDA is not recommending special cancer screenings for patients who have taken lorcaserin.
The action follows an FDA alert in January about a possible elevated cancer risk based on its preliminary analysis of the study.
Patients were also advised Feb. 13 to stop taking the drug and talk to their providers about alternative weight-loss medications and weight-management programs.
They were also told to dispose of the pills at a drug take-back location if available, but if not, to mix them with an “unappealing substance” such as dirt, cat litter, or used coffee grounds; seal them in plastic bag; and put them in the trash.
The Food and Drug Administration asked Eisai to voluntary withdraw the weight-loss drug lorcaserin (Belviq and Belviq XR) on Feb. 13 after a post-marketing trial with more than 12,000 subjects revealed an increased occurrence of cancer.
In a Drug Safety Communication, the agency said “health care professionals should stop prescribing and dispensing lorcaserin to patients. Contact patients currently taking lorcaserin, inform them of the increased occurrence of cancer seen in the clinical trial, and ask them to stop taking the medicine. Discuss alternative weight-loss medicines or strategies with your patients.”
Eisai is complying with the withdrawal request.
The decision is based on the agency’s review of the 5-year trial, which was designed to evaluate cardiac risk with the drug and ended in June 2018. In total, 7.7% of patients randomized to 10 mg lorcaserin twice daily were diagnosed with 520 primary cancers, compared with 7.1% of placebo subjects diagnosed with 470 cancers, over a median follow-up of 3 years and 3 months. There was one additional cancer observed for every 470 patients treated for 1 year.
“There was no apparent difference in the incidence of cancer over the initial months of treatment, but the imbalance increased with longer duration on lorcaserin,” FDA said. Pancreatic, colorectal, and lung cancers were among those diagnosed.
In short, “we believe that the risks of lorcaserin outweigh its benefits based on our completed review of” the data, the agency said. The FDA is not recommending special cancer screenings for patients who have taken lorcaserin.
The action follows an FDA alert in January about a possible elevated cancer risk based on its preliminary analysis of the study.
Patients were also advised Feb. 13 to stop taking the drug and talk to their providers about alternative weight-loss medications and weight-management programs.
They were also told to dispose of the pills at a drug take-back location if available, but if not, to mix them with an “unappealing substance” such as dirt, cat litter, or used coffee grounds; seal them in plastic bag; and put them in the trash.
The Food and Drug Administration asked Eisai to voluntary withdraw the weight-loss drug lorcaserin (Belviq and Belviq XR) on Feb. 13 after a post-marketing trial with more than 12,000 subjects revealed an increased occurrence of cancer.
In a Drug Safety Communication, the agency said “health care professionals should stop prescribing and dispensing lorcaserin to patients. Contact patients currently taking lorcaserin, inform them of the increased occurrence of cancer seen in the clinical trial, and ask them to stop taking the medicine. Discuss alternative weight-loss medicines or strategies with your patients.”
Eisai is complying with the withdrawal request.
The decision is based on the agency’s review of the 5-year trial, which was designed to evaluate cardiac risk with the drug and ended in June 2018. In total, 7.7% of patients randomized to 10 mg lorcaserin twice daily were diagnosed with 520 primary cancers, compared with 7.1% of placebo subjects diagnosed with 470 cancers, over a median follow-up of 3 years and 3 months. There was one additional cancer observed for every 470 patients treated for 1 year.
“There was no apparent difference in the incidence of cancer over the initial months of treatment, but the imbalance increased with longer duration on lorcaserin,” FDA said. Pancreatic, colorectal, and lung cancers were among those diagnosed.
In short, “we believe that the risks of lorcaserin outweigh its benefits based on our completed review of” the data, the agency said. The FDA is not recommending special cancer screenings for patients who have taken lorcaserin.
The action follows an FDA alert in January about a possible elevated cancer risk based on its preliminary analysis of the study.
Patients were also advised Feb. 13 to stop taking the drug and talk to their providers about alternative weight-loss medications and weight-management programs.
They were also told to dispose of the pills at a drug take-back location if available, but if not, to mix them with an “unappealing substance” such as dirt, cat litter, or used coffee grounds; seal them in plastic bag; and put them in the trash.
Pathways to new therapeutic agents for human coronaviruses
No specific treatment is currently available for human coronaviruses to date, but numerous antiviral agents are being identified through a variety of approaches, according to Thanigaimalai Pillaiyar, PhD, and colleagues in a review published in Drug Discovery Today.
Using the six previously discovered human coronaviruses – human CoV 229E (HCoV-229E), OC43 (HCoV-OC43), NL63 (HCoV-NL63), HKU1 (HCoV-HKU1); severe acute respiratory syndrome (SARS) CoV; and Middle East respiratory syndrome (MERS) CoV – the investigators examined progress in the use and development of therapeutic drugs, focusing on the potential roles of virus inhibitors.
“Research has mainly been focused on SARS- and MERS-CoV infections, because they were responsible for severe illness when compared with other CoVs,” Dr. Pillaiyar, of the department of pharmaceutical and medicinal chemistry at the University of Bonn (Germany), and colleagues wrote.
2019-nCov has been linked genomically as most closely related to SARS, and the Coronavirus Study Group of the International Committee on Virus Taxonomy, which has the responsibility for naming viruses, has designated the new virus SARS-CoV-2.
Examining extant drugs
The first approach to identifying possible antiviral agents reevaluates known, broadly acting antiviral drugs that have been used for other viral infections or other indications. The initial research into coronavirus therapeutics, in particular, has examined current antiviral therapeutics for their effectiveness against both SARS-CoV and MERS-CoV, but with mixed results.
For example, in a search of potential antiviral agents against CoVs, researchers identified four drugs – chloroquine, chlorpromazine, loperamide, and lopinavir – by screening drug libraries approved by the Food and Drug Administration. They were all able to inhibit the replication of MERS-CoV, SARS-CoV, and HCoV-229E in the low-micromolar range, which suggested that they could be used for broad-spectrum antiviral activity, according to Dr. Pillaiyar and colleagues.
Other research groups have also reported the discovery of antiviral drugs using this drug-repurposing approach, which included a number of broad-spectrum inhibitors of HCoVs (lycorine, emetine, monensin sodium, mycophenolate mofetil, mycophenolic acid, phenazopyridine, and pyrvinium pamoate) that showed strong inhibition of replication by four CoVs in vitro at low-micromolar concentrations and suppressed the replication of all CoVs in a dose-dependent manner. Findings from in vivo studies showed lycorine protected mice against lethal HCoV-OC43 infection.
Along with the aforementioned drugs, a number of others have also shown potential usefulness, but, as yet, none has been validated for use in humans.
Developing new antivirals
The second approach for anti-CoV drug discovery involves the development of new therapeutics based on the genomic and biophysical understanding of the individual CoV in order to interfere with the virus itself or to disrupt its direct metabolic requirements. This can take several approaches.
MERS-CoV and SARS-CoV PL protease inhibitors
Of particular interest are antiviral therapies that attack papain-like protease, which is an important target because it is a multifunctional protein involved in proteolytic deubiquitination and viral evasion of the innate immune response. One such potential therapeutic that takes advantage of this target is disulfiram, an FDA-approved drug for use in alcohol-aversion therapy. Disulfiram has been reported as an allosteric inhibitor of MERS-CoV papain-like protease. Numerous other drug categories are being examined, with promising results in targeting the papain-like protease enzymes of both SARS and MERS.
Replicase inhibitors
Helicase (nsP13) protein is a crucial component required for virus replication in host cells and could serve as a feasible target for anti-MERS and anti-SARS chemical therapies, the review authors wrote, citing as an example, the recent development of a small 1,2,4-triazole derivative that inhibited the viral NTPase/helicase of SARS- and MERS-CoVs and demonstrated high antiviral activity and low cytotoxicity.
Membrane-bound viral RNA synthesis inhibitors
Antiviral agents that target membrane-bound coronaviral RNA synthesis represent a novel and attractive approach, according to Dr. Pillaiyar and colleagues. And recently, an inhibitor was developed that targets membrane-bound coronaviral RNA synthesis and “showed potent antiviral activity of MERS-CoV infection with remarkable efficacy.”
Host-based, anti-CoV treatment options
An alternate therapeutic tactic is to bolster host defenses or to modify host susceptibilities to prevent virus infection or replication. The innate interferon response of the host is crucial for the control of viral replication after infection, and the addition of exogenous recombinant interferon or use of drugs to stimulate the normal host interferon response are both potential therapeutic avenues. For example, nitazoxanide is a potent type I interferon inducer that has been used in humans for parasitic infections, and a synthetic nitrothiazolyl-salicylamide derivative was found to exhibit broad-spectrum antiviral activities against RNA and DNA viruses, including some coronaviruses.
Numerous other host pathways are being investigated as potential areas to enhance defense against infection and replication, for example, using inhibitors to block nucleic acid synthesis has been shown to provide broad-spectrum activity against SARS-CoV and MERS-CoV.
One particular example is remdesivir, a novel nucleotide analog antiviral drug, that was developed as a therapy for Ebola virus disease and Marburg virus infections. It was later shown to provide “reasonable antiviral activity against more distantly related viruses, such as respiratory syncytial virus, Junin virus, Lassa fever virus, and MERS-CoV,” the authors wrote.
Also of interest regarding remdesivir’s potential broad-spectrum use is that it has shown potent in vitro “antiviral activity against Malaysian and Bangladesh genotypes of Nipah virus (an RNA virus, although not a coronavirus, that infects both humans and animals) and reduced replication of Malaysian Nipah virus in primary human lung microvascular endothelial cells by more than four orders of magnitude,” Dr. Pillaiyar and colleagues added. Of particular note, all remdesivir-treated, Nipah virus–infected animals “survived the lethal challenge, indicating that remdesivir represents a promising antiviral treatment.”
In a press briefing earlier this month, Anthony S. Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, reported that a randomized, controlled, phase 3 trial of the antiviral drug remdesivir is currently underway in China to establish whether the drug would be an effective and safe treatment for adults patients with mild or moderate 2019 Novel Coronavirus (2019-nCoV) disease.
“Our increasing understanding of novel emerging coronaviruses will be accompanied by increasing opportunities for the reasonable design of therapeutics. Importantly, understanding this basic information about CoV protease targets will not only aid the public health against SARS-CoV and MERS-CoV but also help in advance to target new coronaviruses that might emerge in the future,” the authors concluded.
Dr. Pillaiyar and colleagues reported that they had no financial conflicts of interest.
SOURCE: Pillaiyar T et al. Drug Discov Today. 2020 Jan 30. doi: 10.1016/j.drudis.2020.01.015.
No specific treatment is currently available for human coronaviruses to date, but numerous antiviral agents are being identified through a variety of approaches, according to Thanigaimalai Pillaiyar, PhD, and colleagues in a review published in Drug Discovery Today.
Using the six previously discovered human coronaviruses – human CoV 229E (HCoV-229E), OC43 (HCoV-OC43), NL63 (HCoV-NL63), HKU1 (HCoV-HKU1); severe acute respiratory syndrome (SARS) CoV; and Middle East respiratory syndrome (MERS) CoV – the investigators examined progress in the use and development of therapeutic drugs, focusing on the potential roles of virus inhibitors.
“Research has mainly been focused on SARS- and MERS-CoV infections, because they were responsible for severe illness when compared with other CoVs,” Dr. Pillaiyar, of the department of pharmaceutical and medicinal chemistry at the University of Bonn (Germany), and colleagues wrote.
2019-nCov has been linked genomically as most closely related to SARS, and the Coronavirus Study Group of the International Committee on Virus Taxonomy, which has the responsibility for naming viruses, has designated the new virus SARS-CoV-2.
Examining extant drugs
The first approach to identifying possible antiviral agents reevaluates known, broadly acting antiviral drugs that have been used for other viral infections or other indications. The initial research into coronavirus therapeutics, in particular, has examined current antiviral therapeutics for their effectiveness against both SARS-CoV and MERS-CoV, but with mixed results.
For example, in a search of potential antiviral agents against CoVs, researchers identified four drugs – chloroquine, chlorpromazine, loperamide, and lopinavir – by screening drug libraries approved by the Food and Drug Administration. They were all able to inhibit the replication of MERS-CoV, SARS-CoV, and HCoV-229E in the low-micromolar range, which suggested that they could be used for broad-spectrum antiviral activity, according to Dr. Pillaiyar and colleagues.
Other research groups have also reported the discovery of antiviral drugs using this drug-repurposing approach, which included a number of broad-spectrum inhibitors of HCoVs (lycorine, emetine, monensin sodium, mycophenolate mofetil, mycophenolic acid, phenazopyridine, and pyrvinium pamoate) that showed strong inhibition of replication by four CoVs in vitro at low-micromolar concentrations and suppressed the replication of all CoVs in a dose-dependent manner. Findings from in vivo studies showed lycorine protected mice against lethal HCoV-OC43 infection.
Along with the aforementioned drugs, a number of others have also shown potential usefulness, but, as yet, none has been validated for use in humans.
Developing new antivirals
The second approach for anti-CoV drug discovery involves the development of new therapeutics based on the genomic and biophysical understanding of the individual CoV in order to interfere with the virus itself or to disrupt its direct metabolic requirements. This can take several approaches.
MERS-CoV and SARS-CoV PL protease inhibitors
Of particular interest are antiviral therapies that attack papain-like protease, which is an important target because it is a multifunctional protein involved in proteolytic deubiquitination and viral evasion of the innate immune response. One such potential therapeutic that takes advantage of this target is disulfiram, an FDA-approved drug for use in alcohol-aversion therapy. Disulfiram has been reported as an allosteric inhibitor of MERS-CoV papain-like protease. Numerous other drug categories are being examined, with promising results in targeting the papain-like protease enzymes of both SARS and MERS.
Replicase inhibitors
Helicase (nsP13) protein is a crucial component required for virus replication in host cells and could serve as a feasible target for anti-MERS and anti-SARS chemical therapies, the review authors wrote, citing as an example, the recent development of a small 1,2,4-triazole derivative that inhibited the viral NTPase/helicase of SARS- and MERS-CoVs and demonstrated high antiviral activity and low cytotoxicity.
Membrane-bound viral RNA synthesis inhibitors
Antiviral agents that target membrane-bound coronaviral RNA synthesis represent a novel and attractive approach, according to Dr. Pillaiyar and colleagues. And recently, an inhibitor was developed that targets membrane-bound coronaviral RNA synthesis and “showed potent antiviral activity of MERS-CoV infection with remarkable efficacy.”
Host-based, anti-CoV treatment options
An alternate therapeutic tactic is to bolster host defenses or to modify host susceptibilities to prevent virus infection or replication. The innate interferon response of the host is crucial for the control of viral replication after infection, and the addition of exogenous recombinant interferon or use of drugs to stimulate the normal host interferon response are both potential therapeutic avenues. For example, nitazoxanide is a potent type I interferon inducer that has been used in humans for parasitic infections, and a synthetic nitrothiazolyl-salicylamide derivative was found to exhibit broad-spectrum antiviral activities against RNA and DNA viruses, including some coronaviruses.
Numerous other host pathways are being investigated as potential areas to enhance defense against infection and replication, for example, using inhibitors to block nucleic acid synthesis has been shown to provide broad-spectrum activity against SARS-CoV and MERS-CoV.
One particular example is remdesivir, a novel nucleotide analog antiviral drug, that was developed as a therapy for Ebola virus disease and Marburg virus infections. It was later shown to provide “reasonable antiviral activity against more distantly related viruses, such as respiratory syncytial virus, Junin virus, Lassa fever virus, and MERS-CoV,” the authors wrote.
Also of interest regarding remdesivir’s potential broad-spectrum use is that it has shown potent in vitro “antiviral activity against Malaysian and Bangladesh genotypes of Nipah virus (an RNA virus, although not a coronavirus, that infects both humans and animals) and reduced replication of Malaysian Nipah virus in primary human lung microvascular endothelial cells by more than four orders of magnitude,” Dr. Pillaiyar and colleagues added. Of particular note, all remdesivir-treated, Nipah virus–infected animals “survived the lethal challenge, indicating that remdesivir represents a promising antiviral treatment.”
In a press briefing earlier this month, Anthony S. Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, reported that a randomized, controlled, phase 3 trial of the antiviral drug remdesivir is currently underway in China to establish whether the drug would be an effective and safe treatment for adults patients with mild or moderate 2019 Novel Coronavirus (2019-nCoV) disease.
“Our increasing understanding of novel emerging coronaviruses will be accompanied by increasing opportunities for the reasonable design of therapeutics. Importantly, understanding this basic information about CoV protease targets will not only aid the public health against SARS-CoV and MERS-CoV but also help in advance to target new coronaviruses that might emerge in the future,” the authors concluded.
Dr. Pillaiyar and colleagues reported that they had no financial conflicts of interest.
SOURCE: Pillaiyar T et al. Drug Discov Today. 2020 Jan 30. doi: 10.1016/j.drudis.2020.01.015.
No specific treatment is currently available for human coronaviruses to date, but numerous antiviral agents are being identified through a variety of approaches, according to Thanigaimalai Pillaiyar, PhD, and colleagues in a review published in Drug Discovery Today.
Using the six previously discovered human coronaviruses – human CoV 229E (HCoV-229E), OC43 (HCoV-OC43), NL63 (HCoV-NL63), HKU1 (HCoV-HKU1); severe acute respiratory syndrome (SARS) CoV; and Middle East respiratory syndrome (MERS) CoV – the investigators examined progress in the use and development of therapeutic drugs, focusing on the potential roles of virus inhibitors.
“Research has mainly been focused on SARS- and MERS-CoV infections, because they were responsible for severe illness when compared with other CoVs,” Dr. Pillaiyar, of the department of pharmaceutical and medicinal chemistry at the University of Bonn (Germany), and colleagues wrote.
2019-nCov has been linked genomically as most closely related to SARS, and the Coronavirus Study Group of the International Committee on Virus Taxonomy, which has the responsibility for naming viruses, has designated the new virus SARS-CoV-2.
Examining extant drugs
The first approach to identifying possible antiviral agents reevaluates known, broadly acting antiviral drugs that have been used for other viral infections or other indications. The initial research into coronavirus therapeutics, in particular, has examined current antiviral therapeutics for their effectiveness against both SARS-CoV and MERS-CoV, but with mixed results.
For example, in a search of potential antiviral agents against CoVs, researchers identified four drugs – chloroquine, chlorpromazine, loperamide, and lopinavir – by screening drug libraries approved by the Food and Drug Administration. They were all able to inhibit the replication of MERS-CoV, SARS-CoV, and HCoV-229E in the low-micromolar range, which suggested that they could be used for broad-spectrum antiviral activity, according to Dr. Pillaiyar and colleagues.
Other research groups have also reported the discovery of antiviral drugs using this drug-repurposing approach, which included a number of broad-spectrum inhibitors of HCoVs (lycorine, emetine, monensin sodium, mycophenolate mofetil, mycophenolic acid, phenazopyridine, and pyrvinium pamoate) that showed strong inhibition of replication by four CoVs in vitro at low-micromolar concentrations and suppressed the replication of all CoVs in a dose-dependent manner. Findings from in vivo studies showed lycorine protected mice against lethal HCoV-OC43 infection.
Along with the aforementioned drugs, a number of others have also shown potential usefulness, but, as yet, none has been validated for use in humans.
Developing new antivirals
The second approach for anti-CoV drug discovery involves the development of new therapeutics based on the genomic and biophysical understanding of the individual CoV in order to interfere with the virus itself or to disrupt its direct metabolic requirements. This can take several approaches.
MERS-CoV and SARS-CoV PL protease inhibitors
Of particular interest are antiviral therapies that attack papain-like protease, which is an important target because it is a multifunctional protein involved in proteolytic deubiquitination and viral evasion of the innate immune response. One such potential therapeutic that takes advantage of this target is disulfiram, an FDA-approved drug for use in alcohol-aversion therapy. Disulfiram has been reported as an allosteric inhibitor of MERS-CoV papain-like protease. Numerous other drug categories are being examined, with promising results in targeting the papain-like protease enzymes of both SARS and MERS.
Replicase inhibitors
Helicase (nsP13) protein is a crucial component required for virus replication in host cells and could serve as a feasible target for anti-MERS and anti-SARS chemical therapies, the review authors wrote, citing as an example, the recent development of a small 1,2,4-triazole derivative that inhibited the viral NTPase/helicase of SARS- and MERS-CoVs and demonstrated high antiviral activity and low cytotoxicity.
Membrane-bound viral RNA synthesis inhibitors
Antiviral agents that target membrane-bound coronaviral RNA synthesis represent a novel and attractive approach, according to Dr. Pillaiyar and colleagues. And recently, an inhibitor was developed that targets membrane-bound coronaviral RNA synthesis and “showed potent antiviral activity of MERS-CoV infection with remarkable efficacy.”
Host-based, anti-CoV treatment options
An alternate therapeutic tactic is to bolster host defenses or to modify host susceptibilities to prevent virus infection or replication. The innate interferon response of the host is crucial for the control of viral replication after infection, and the addition of exogenous recombinant interferon or use of drugs to stimulate the normal host interferon response are both potential therapeutic avenues. For example, nitazoxanide is a potent type I interferon inducer that has been used in humans for parasitic infections, and a synthetic nitrothiazolyl-salicylamide derivative was found to exhibit broad-spectrum antiviral activities against RNA and DNA viruses, including some coronaviruses.
Numerous other host pathways are being investigated as potential areas to enhance defense against infection and replication, for example, using inhibitors to block nucleic acid synthesis has been shown to provide broad-spectrum activity against SARS-CoV and MERS-CoV.
One particular example is remdesivir, a novel nucleotide analog antiviral drug, that was developed as a therapy for Ebola virus disease and Marburg virus infections. It was later shown to provide “reasonable antiviral activity against more distantly related viruses, such as respiratory syncytial virus, Junin virus, Lassa fever virus, and MERS-CoV,” the authors wrote.
Also of interest regarding remdesivir’s potential broad-spectrum use is that it has shown potent in vitro “antiviral activity against Malaysian and Bangladesh genotypes of Nipah virus (an RNA virus, although not a coronavirus, that infects both humans and animals) and reduced replication of Malaysian Nipah virus in primary human lung microvascular endothelial cells by more than four orders of magnitude,” Dr. Pillaiyar and colleagues added. Of particular note, all remdesivir-treated, Nipah virus–infected animals “survived the lethal challenge, indicating that remdesivir represents a promising antiviral treatment.”
In a press briefing earlier this month, Anthony S. Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, reported that a randomized, controlled, phase 3 trial of the antiviral drug remdesivir is currently underway in China to establish whether the drug would be an effective and safe treatment for adults patients with mild or moderate 2019 Novel Coronavirus (2019-nCoV) disease.
“Our increasing understanding of novel emerging coronaviruses will be accompanied by increasing opportunities for the reasonable design of therapeutics. Importantly, understanding this basic information about CoV protease targets will not only aid the public health against SARS-CoV and MERS-CoV but also help in advance to target new coronaviruses that might emerge in the future,” the authors concluded.
Dr. Pillaiyar and colleagues reported that they had no financial conflicts of interest.
SOURCE: Pillaiyar T et al. Drug Discov Today. 2020 Jan 30. doi: 10.1016/j.drudis.2020.01.015.
FROM DRUG DISCOVERY TODAY
Pharmacologic prophylaxis fails in pediatric migraine
Clinicians hoped that medications used in adults – such as antidepressants, antiepileptics, antihypertensive agents, calcium channel blockers, and food supplements – would find similar success in children. Unfortunately, researchers found only short-term signs of efficacy over placebo, with no benefit lasting more than 6 months.
The study, conducted by a team led by Cosima Locher, PhD, of Boston Children’s Hospital, included 23 double-blind, randomized, controlled trials with a total of 2,217 patients; the mean age was 11 years. They compared 12 pharmacologic agents with each other or with placebo in the study, published online in JAMA Pediatrics.
In a main efficacy analysis that included 19 studies, only two treatments outperformed placebo: propranolol (standardized mean difference, 0.60; 95% confidence interval, 0.03-1.17) and topiramate (SMD, 0.59; 95% CI, 0.03-1.15). There were no statistically significant between-treatment differences.
The results had an overall low to moderate certainty.
When propranolol was compared to placebo, the 95% prediction interval (–0.62 to 1.82) was wider than the significant confidence interval (0.03-1.17), and comprised both beneficial and detrimental effects. A similar result was found with topiramate, with a prediction interval of –0.62 to 1.80 extending into nonsignificant effects (95% CI, 0.03-1.15). In both cases, significant effects were found only when the prediction interval was 70%.
In a long-term analysis (greater than 6 months), no treatment outperformed placebo.
The treatments generally were acceptable. The researchers found no significant difference in tolerability between any of the treatments and each other or placebo. Safety data analyzed from 13 trials revealed no significant differences between treatments and placebo.
“Because specific effects of drugs are associated with the size of the placebo effect, the lack of drug efficacy in our NMA [network meta-analysis] could be owing to a comparatively high placebo effect in children. In fact, there is indirect evidence [from other studies] that the placebo effect is more pronounced in children and adolescents than in adults,” Dr. Locher and associates said. They suggested that studies were needed to quantify the placebo effect in pediatric migraine, and if it was large, to develop innovative therapies making use of this.
The findings should lead to some changes in practice, Boris Zernikow, MD, PhD, of Children’s and Adolescents’ Hospital Datteln (Germany) wrote in an accompanying editorial.
Pharmacological prophylactic treatment of childhood migraine should be an exception rather than the rule, and nonpharmacologic approaches should be emphasized, particularly because the placebo effect is magnified in children, he said.
Many who suffer migraines in childhood will continue to be affected in adulthood, so pediatric intervention is a good opportunity to instill effective strategies. These include: using abortive medication early in an attack and using antimigraine medications for only that specific type of headache; engaging in physical activity to reduce migraine attacks; getting sufficient sleep; and learning relaxation and other psychological approaches to counter migraines.
Dr. Zernikow had no relevant financial disclosures. One study author received grants from Amgen and other support from Grunenthal and Akelos. The study received funding from the Sara Page Mayo Endowment for Pediatric Pain Research, Education, and Treatment; the Swiss National Science Foundation; the Schweizer-Arau-Foundation; and the Theophrastus Foundation.
SOURCES: Locher C et al. JAMA Pediatrics. 2020 Feb 10. doi: 10.1001/jamapediatrics.2019.5856; Zernikow B. JAMA Pediatrics. 2020 Feb 10. doi: 10.1001/jamapediatrics.2019.5907.
Clinicians hoped that medications used in adults – such as antidepressants, antiepileptics, antihypertensive agents, calcium channel blockers, and food supplements – would find similar success in children. Unfortunately, researchers found only short-term signs of efficacy over placebo, with no benefit lasting more than 6 months.
The study, conducted by a team led by Cosima Locher, PhD, of Boston Children’s Hospital, included 23 double-blind, randomized, controlled trials with a total of 2,217 patients; the mean age was 11 years. They compared 12 pharmacologic agents with each other or with placebo in the study, published online in JAMA Pediatrics.
In a main efficacy analysis that included 19 studies, only two treatments outperformed placebo: propranolol (standardized mean difference, 0.60; 95% confidence interval, 0.03-1.17) and topiramate (SMD, 0.59; 95% CI, 0.03-1.15). There were no statistically significant between-treatment differences.
The results had an overall low to moderate certainty.
When propranolol was compared to placebo, the 95% prediction interval (–0.62 to 1.82) was wider than the significant confidence interval (0.03-1.17), and comprised both beneficial and detrimental effects. A similar result was found with topiramate, with a prediction interval of –0.62 to 1.80 extending into nonsignificant effects (95% CI, 0.03-1.15). In both cases, significant effects were found only when the prediction interval was 70%.
In a long-term analysis (greater than 6 months), no treatment outperformed placebo.
The treatments generally were acceptable. The researchers found no significant difference in tolerability between any of the treatments and each other or placebo. Safety data analyzed from 13 trials revealed no significant differences between treatments and placebo.
“Because specific effects of drugs are associated with the size of the placebo effect, the lack of drug efficacy in our NMA [network meta-analysis] could be owing to a comparatively high placebo effect in children. In fact, there is indirect evidence [from other studies] that the placebo effect is more pronounced in children and adolescents than in adults,” Dr. Locher and associates said. They suggested that studies were needed to quantify the placebo effect in pediatric migraine, and if it was large, to develop innovative therapies making use of this.
The findings should lead to some changes in practice, Boris Zernikow, MD, PhD, of Children’s and Adolescents’ Hospital Datteln (Germany) wrote in an accompanying editorial.
Pharmacological prophylactic treatment of childhood migraine should be an exception rather than the rule, and nonpharmacologic approaches should be emphasized, particularly because the placebo effect is magnified in children, he said.
Many who suffer migraines in childhood will continue to be affected in adulthood, so pediatric intervention is a good opportunity to instill effective strategies. These include: using abortive medication early in an attack and using antimigraine medications for only that specific type of headache; engaging in physical activity to reduce migraine attacks; getting sufficient sleep; and learning relaxation and other psychological approaches to counter migraines.
Dr. Zernikow had no relevant financial disclosures. One study author received grants from Amgen and other support from Grunenthal and Akelos. The study received funding from the Sara Page Mayo Endowment for Pediatric Pain Research, Education, and Treatment; the Swiss National Science Foundation; the Schweizer-Arau-Foundation; and the Theophrastus Foundation.
SOURCES: Locher C et al. JAMA Pediatrics. 2020 Feb 10. doi: 10.1001/jamapediatrics.2019.5856; Zernikow B. JAMA Pediatrics. 2020 Feb 10. doi: 10.1001/jamapediatrics.2019.5907.
Clinicians hoped that medications used in adults – such as antidepressants, antiepileptics, antihypertensive agents, calcium channel blockers, and food supplements – would find similar success in children. Unfortunately, researchers found only short-term signs of efficacy over placebo, with no benefit lasting more than 6 months.
The study, conducted by a team led by Cosima Locher, PhD, of Boston Children’s Hospital, included 23 double-blind, randomized, controlled trials with a total of 2,217 patients; the mean age was 11 years. They compared 12 pharmacologic agents with each other or with placebo in the study, published online in JAMA Pediatrics.
In a main efficacy analysis that included 19 studies, only two treatments outperformed placebo: propranolol (standardized mean difference, 0.60; 95% confidence interval, 0.03-1.17) and topiramate (SMD, 0.59; 95% CI, 0.03-1.15). There were no statistically significant between-treatment differences.
The results had an overall low to moderate certainty.
When propranolol was compared to placebo, the 95% prediction interval (–0.62 to 1.82) was wider than the significant confidence interval (0.03-1.17), and comprised both beneficial and detrimental effects. A similar result was found with topiramate, with a prediction interval of –0.62 to 1.80 extending into nonsignificant effects (95% CI, 0.03-1.15). In both cases, significant effects were found only when the prediction interval was 70%.
In a long-term analysis (greater than 6 months), no treatment outperformed placebo.
The treatments generally were acceptable. The researchers found no significant difference in tolerability between any of the treatments and each other or placebo. Safety data analyzed from 13 trials revealed no significant differences between treatments and placebo.
“Because specific effects of drugs are associated with the size of the placebo effect, the lack of drug efficacy in our NMA [network meta-analysis] could be owing to a comparatively high placebo effect in children. In fact, there is indirect evidence [from other studies] that the placebo effect is more pronounced in children and adolescents than in adults,” Dr. Locher and associates said. They suggested that studies were needed to quantify the placebo effect in pediatric migraine, and if it was large, to develop innovative therapies making use of this.
The findings should lead to some changes in practice, Boris Zernikow, MD, PhD, of Children’s and Adolescents’ Hospital Datteln (Germany) wrote in an accompanying editorial.
Pharmacological prophylactic treatment of childhood migraine should be an exception rather than the rule, and nonpharmacologic approaches should be emphasized, particularly because the placebo effect is magnified in children, he said.
Many who suffer migraines in childhood will continue to be affected in adulthood, so pediatric intervention is a good opportunity to instill effective strategies. These include: using abortive medication early in an attack and using antimigraine medications for only that specific type of headache; engaging in physical activity to reduce migraine attacks; getting sufficient sleep; and learning relaxation and other psychological approaches to counter migraines.
Dr. Zernikow had no relevant financial disclosures. One study author received grants from Amgen and other support from Grunenthal and Akelos. The study received funding from the Sara Page Mayo Endowment for Pediatric Pain Research, Education, and Treatment; the Swiss National Science Foundation; the Schweizer-Arau-Foundation; and the Theophrastus Foundation.
SOURCES: Locher C et al. JAMA Pediatrics. 2020 Feb 10. doi: 10.1001/jamapediatrics.2019.5856; Zernikow B. JAMA Pediatrics. 2020 Feb 10. doi: 10.1001/jamapediatrics.2019.5907.
FROM JAMA PEDIATRICS
Shift in approach is encouraged in assessing chronic pain
In many cases, dietary interventions can lead to less inflammation
SAN DIEGO – When clinicians ask patients to quantify their level of chronic pain on a scale of 1-10, and they rate it as a 7, what does that really mean?
Robert A. Bonakdar, MD, said posing such a question as the main determinator of the treatment approach during a pain assessment “depersonalizes medicine to the point where you’re making a patient a number.” Dr. Bonakdar spoke at Natural Supplements: An Evidence-Based Update, presented by Scripps Center for Integrative Medicine.
“It considers areas that are often overlooked, such as the role of the gut microbiome, mood, and epigenetics.”
Over the past two decades, the number of American adults suffering from pain has increased from 120 million to 178 million, or to 41% of the adult population, said Dr. Bonakdar, a family physician who is director of pain management at the Scripps Center for Integrative Medicine. Data from the National Institutes of Health estimate that Americans spend more than $600 billion each year on the treatment of pain, which surpasses monies spent on cancer, heart disease, and diabetes. According to a 2016 report from the United States Bone and Joint Initiative, arthritis and rheumatologic conditions resulted in an estimated 6.7 million annual hospitalizations, and the average annual cost per person for treatment of a musculoskeletal condition is $7,800.
“If we continue on our current trajectory, we are choosing to accept more prevalence and incidence of these disorders, spiraling costs, restricted access to needed services, and less success in alleviating pain and suffering – a high cost,” Edward H. Yelin, PhD, cochair of the report’s steering committee, and professor of medicine and health policy at the University of California, San Francisco, said in a prepared statement in 2016. That same year, Brian F. Mandell, MD, PhD, editor of the Cleveland Clinic Journal of Medicine, penned an editorial in which he stated that “The time has come to move past using a one-size-fits-all fifth vital sign . . . and reflexively prescribing an opioid when pain is characterized as severe” (Clev Clin J Med. 2016. Jun;83[6]:400-1). A decade earlier, authors of a cross-sectional review at a single Department of Veterans Affairs medical center set out to assess the impact of the VA’s “Pain as the 5th Vital Sign” initiative on the quality of pain management (J Gen Intern Med. 2006;21[6]:607–12). They found that patients with substantial pain documented by the fifth vital sign often had inadequate pain management. The preponderance of existing evidence suggests that a different approach is needed to prescribing opioids, Dr. Bonakdar said. “It’s coming from every voice in pain care: that what we are doing is not working,” he said. “It’s not only not working; it’s dangerous. That’s the consequence of depersonalized medicine. What’s the consequence of depersonalized nutrition? It’s the same industrialized approach.”
The typical American diet, he continued, is rife with processed foods and lacks an adequate proportion of plant-based products. “It’s basically a setup for inflammation,” Dr. Bonakdar said. “Most people who come into our clinic are eating 63% processed foods, 25% animal foods, and 12% plant foods. When we are eating, we’re oversizing it because that’s the American thing to do. At the end of the day, this process is not only killing us from heart disease and stroke as causes of death, but it’s also killing us as far as pain. The same diet that’s causing heart disease is the same diet that’s increasing pain.”
Dr. Bonakdar said that the ingestion of ultra-processed foods over time jumpstarts the process of dysbiosis, which increases gut permeability. “When gut permeability happens, and you have high levels of polysaccharides and inflammatory markers such as zonulin and lipopolysaccharide (LPS), it not only goes on to affect adipose tissue and insulin resistance, it can affect the muscle and joints,” he explained. “That is a setup for sarcopenia, or muscle loss, which then makes it harder for patients to be fully functional and active. It goes on to cause joint problems as well.”
He likened an increase in gut permeability to “a bomb going off in the gut.” Routine consumption of highly processed foods “creates this wave of inflammation that goes throughout your body affecting joints and muscles, and causes an increased amount of pain. Over time, patients make the connection but it’s much easier to say, ‘take this NSAID’ or ‘take this Cox-2 inhibitor’ to suppress the pain. But if all you’re doing is suppressing, you’re not going to the source of the pain.”
Dr. Bonakdar cited several recent articles that help to make the connection between dysbiosis and pain, including a review that concluded that dysbiosis of gut microbiota can influence the onset and progression of chronic degenerative diseases (Nutrients. 2019;11[8]:1707). Authors of a separate review concluded that human microbiome studies strongly suggest an incriminating role of microbes in the pathophysiology and progression of RA. Lastly, several studies have noted that pain conditions such as fibromyalgia may have microbiome “signatures” related to dysbiosis, which may pave the way for interventions, such as dietary shifting and probiotics that target individuals with microbiome abnormalities (Pain. 2019 Nov;160[11]:2589-602 and EBioMedicine. 2019 Aug 1;46:499-511).
Clinicians can begin to help patients who present with pain complaints “by listening to what their current pattern is: strategies that have worked, and those that haven’t,” he said. “If we’re not understanding the person and we’re just ordering genetic studies or microbiome studies and going off of the assessment, we sometime miss what interventions to start. In many cases, a simple intervention like a dietary shift is all that’s required.”
A survey of more than 1 million individuals found that BMI and daily pain are positively correlated in the United States (Obesity 2012;20[7]:1491-5). “This is increased more significantly for women and the elderly,” said Dr. Bonakdar, who was not affiliated with the study. “If we can change the diet that person is taking, that’s going to begin the process of reversing this to the point where they’re having less pain from inflammation that’s affecting the adipose tissue and adipokines traveling to their joints, which can cause less dysbiosis. It is very much a vicious cycle that patients follow, but if you begin to unwind it, it’s going to help multiple areas.”
In the Intensive Diet and Exercise for Arthritis (IDEA) trial, researchers randomized 450 patients with osteoarthritis to intensive dietary restriction only, exercise only, or a combination of both (BMC Musculoskelet Disord. 2009;10:93). They found that a 5% weight loss over the course of 18 months led to a 30% reduction in pain and a 24% improvement in function.
Inspired by the IDEA trial design, Dr. Bonakdar and his colleagues completed an unpublished 12-week pilot program with 12 patients with a BMI of 27 kg/m2 or greater plus comorbidities. The program consisted of weekly group meetings, including a lecture by team clinicians, dietician, and fitness staff; group support sessions with a behavioral counselor; and a group exercise session. It also included weekly 1:1 personal training sessions and biweekly 1:1 dietitian meetings. The researchers also evaluated several deficiencies linked to pain, including magnesium, vitamin D, vitamins B1, B2, and B12, folate, calcium, amino acids, omega 3s, zinc, coenzyme Q10, carnitine, and vitamin C. The goal was a weight reduction of 5%.
The intervention consisted of a 28-day detox/protein shake consumed 1-3 times per day, which contained 17 g of protein per serving. Nutritional supplementation was added based on results of individual diagnostics.
According to preliminary results from the trial, the intended weight goal was achieved. “More importantly, there were significant improvements in markers of dysbiosis, including zonulin and lipopolysaccharide, as well as the adipokine leptin, which appeared to be associated with improvement in quality of life measures and pain,” Dr. Bonakdar said.
He concluded his presentation by highlighting a pilot study conducted in an Australian tertiary pain clinic. It found that a personalized dietitian-delivered dietary intervention can improve pain scores, quality of life, and dietary intake of people experiencing chronic pain (Nutrients. 2019 Jan 16;11[1] pii: E181). “This is another piece of the puzzle showing that these dietary interventions can be done in multiple settings, including tertiary centers with nutrition staff, and that this important step can improve pain and quality of life,” he said.
Dr. Bonakdar disclosed that he receives royalties from Oxford University Press, Lippincott, and Elsevier. He is also a consultant to Standard Process.
In many cases, dietary interventions can lead to less inflammation
In many cases, dietary interventions can lead to less inflammation
SAN DIEGO – When clinicians ask patients to quantify their level of chronic pain on a scale of 1-10, and they rate it as a 7, what does that really mean?
Robert A. Bonakdar, MD, said posing such a question as the main determinator of the treatment approach during a pain assessment “depersonalizes medicine to the point where you’re making a patient a number.” Dr. Bonakdar spoke at Natural Supplements: An Evidence-Based Update, presented by Scripps Center for Integrative Medicine.
“It considers areas that are often overlooked, such as the role of the gut microbiome, mood, and epigenetics.”
Over the past two decades, the number of American adults suffering from pain has increased from 120 million to 178 million, or to 41% of the adult population, said Dr. Bonakdar, a family physician who is director of pain management at the Scripps Center for Integrative Medicine. Data from the National Institutes of Health estimate that Americans spend more than $600 billion each year on the treatment of pain, which surpasses monies spent on cancer, heart disease, and diabetes. According to a 2016 report from the United States Bone and Joint Initiative, arthritis and rheumatologic conditions resulted in an estimated 6.7 million annual hospitalizations, and the average annual cost per person for treatment of a musculoskeletal condition is $7,800.
“If we continue on our current trajectory, we are choosing to accept more prevalence and incidence of these disorders, spiraling costs, restricted access to needed services, and less success in alleviating pain and suffering – a high cost,” Edward H. Yelin, PhD, cochair of the report’s steering committee, and professor of medicine and health policy at the University of California, San Francisco, said in a prepared statement in 2016. That same year, Brian F. Mandell, MD, PhD, editor of the Cleveland Clinic Journal of Medicine, penned an editorial in which he stated that “The time has come to move past using a one-size-fits-all fifth vital sign . . . and reflexively prescribing an opioid when pain is characterized as severe” (Clev Clin J Med. 2016. Jun;83[6]:400-1). A decade earlier, authors of a cross-sectional review at a single Department of Veterans Affairs medical center set out to assess the impact of the VA’s “Pain as the 5th Vital Sign” initiative on the quality of pain management (J Gen Intern Med. 2006;21[6]:607–12). They found that patients with substantial pain documented by the fifth vital sign often had inadequate pain management. The preponderance of existing evidence suggests that a different approach is needed to prescribing opioids, Dr. Bonakdar said. “It’s coming from every voice in pain care: that what we are doing is not working,” he said. “It’s not only not working; it’s dangerous. That’s the consequence of depersonalized medicine. What’s the consequence of depersonalized nutrition? It’s the same industrialized approach.”
The typical American diet, he continued, is rife with processed foods and lacks an adequate proportion of plant-based products. “It’s basically a setup for inflammation,” Dr. Bonakdar said. “Most people who come into our clinic are eating 63% processed foods, 25% animal foods, and 12% plant foods. When we are eating, we’re oversizing it because that’s the American thing to do. At the end of the day, this process is not only killing us from heart disease and stroke as causes of death, but it’s also killing us as far as pain. The same diet that’s causing heart disease is the same diet that’s increasing pain.”
Dr. Bonakdar said that the ingestion of ultra-processed foods over time jumpstarts the process of dysbiosis, which increases gut permeability. “When gut permeability happens, and you have high levels of polysaccharides and inflammatory markers such as zonulin and lipopolysaccharide (LPS), it not only goes on to affect adipose tissue and insulin resistance, it can affect the muscle and joints,” he explained. “That is a setup for sarcopenia, or muscle loss, which then makes it harder for patients to be fully functional and active. It goes on to cause joint problems as well.”
He likened an increase in gut permeability to “a bomb going off in the gut.” Routine consumption of highly processed foods “creates this wave of inflammation that goes throughout your body affecting joints and muscles, and causes an increased amount of pain. Over time, patients make the connection but it’s much easier to say, ‘take this NSAID’ or ‘take this Cox-2 inhibitor’ to suppress the pain. But if all you’re doing is suppressing, you’re not going to the source of the pain.”
Dr. Bonakdar cited several recent articles that help to make the connection between dysbiosis and pain, including a review that concluded that dysbiosis of gut microbiota can influence the onset and progression of chronic degenerative diseases (Nutrients. 2019;11[8]:1707). Authors of a separate review concluded that human microbiome studies strongly suggest an incriminating role of microbes in the pathophysiology and progression of RA. Lastly, several studies have noted that pain conditions such as fibromyalgia may have microbiome “signatures” related to dysbiosis, which may pave the way for interventions, such as dietary shifting and probiotics that target individuals with microbiome abnormalities (Pain. 2019 Nov;160[11]:2589-602 and EBioMedicine. 2019 Aug 1;46:499-511).
Clinicians can begin to help patients who present with pain complaints “by listening to what their current pattern is: strategies that have worked, and those that haven’t,” he said. “If we’re not understanding the person and we’re just ordering genetic studies or microbiome studies and going off of the assessment, we sometime miss what interventions to start. In many cases, a simple intervention like a dietary shift is all that’s required.”
A survey of more than 1 million individuals found that BMI and daily pain are positively correlated in the United States (Obesity 2012;20[7]:1491-5). “This is increased more significantly for women and the elderly,” said Dr. Bonakdar, who was not affiliated with the study. “If we can change the diet that person is taking, that’s going to begin the process of reversing this to the point where they’re having less pain from inflammation that’s affecting the adipose tissue and adipokines traveling to their joints, which can cause less dysbiosis. It is very much a vicious cycle that patients follow, but if you begin to unwind it, it’s going to help multiple areas.”
In the Intensive Diet and Exercise for Arthritis (IDEA) trial, researchers randomized 450 patients with osteoarthritis to intensive dietary restriction only, exercise only, or a combination of both (BMC Musculoskelet Disord. 2009;10:93). They found that a 5% weight loss over the course of 18 months led to a 30% reduction in pain and a 24% improvement in function.
Inspired by the IDEA trial design, Dr. Bonakdar and his colleagues completed an unpublished 12-week pilot program with 12 patients with a BMI of 27 kg/m2 or greater plus comorbidities. The program consisted of weekly group meetings, including a lecture by team clinicians, dietician, and fitness staff; group support sessions with a behavioral counselor; and a group exercise session. It also included weekly 1:1 personal training sessions and biweekly 1:1 dietitian meetings. The researchers also evaluated several deficiencies linked to pain, including magnesium, vitamin D, vitamins B1, B2, and B12, folate, calcium, amino acids, omega 3s, zinc, coenzyme Q10, carnitine, and vitamin C. The goal was a weight reduction of 5%.
The intervention consisted of a 28-day detox/protein shake consumed 1-3 times per day, which contained 17 g of protein per serving. Nutritional supplementation was added based on results of individual diagnostics.
According to preliminary results from the trial, the intended weight goal was achieved. “More importantly, there were significant improvements in markers of dysbiosis, including zonulin and lipopolysaccharide, as well as the adipokine leptin, which appeared to be associated with improvement in quality of life measures and pain,” Dr. Bonakdar said.
He concluded his presentation by highlighting a pilot study conducted in an Australian tertiary pain clinic. It found that a personalized dietitian-delivered dietary intervention can improve pain scores, quality of life, and dietary intake of people experiencing chronic pain (Nutrients. 2019 Jan 16;11[1] pii: E181). “This is another piece of the puzzle showing that these dietary interventions can be done in multiple settings, including tertiary centers with nutrition staff, and that this important step can improve pain and quality of life,” he said.
Dr. Bonakdar disclosed that he receives royalties from Oxford University Press, Lippincott, and Elsevier. He is also a consultant to Standard Process.
SAN DIEGO – When clinicians ask patients to quantify their level of chronic pain on a scale of 1-10, and they rate it as a 7, what does that really mean?
Robert A. Bonakdar, MD, said posing such a question as the main determinator of the treatment approach during a pain assessment “depersonalizes medicine to the point where you’re making a patient a number.” Dr. Bonakdar spoke at Natural Supplements: An Evidence-Based Update, presented by Scripps Center for Integrative Medicine.
“It considers areas that are often overlooked, such as the role of the gut microbiome, mood, and epigenetics.”
Over the past two decades, the number of American adults suffering from pain has increased from 120 million to 178 million, or to 41% of the adult population, said Dr. Bonakdar, a family physician who is director of pain management at the Scripps Center for Integrative Medicine. Data from the National Institutes of Health estimate that Americans spend more than $600 billion each year on the treatment of pain, which surpasses monies spent on cancer, heart disease, and diabetes. According to a 2016 report from the United States Bone and Joint Initiative, arthritis and rheumatologic conditions resulted in an estimated 6.7 million annual hospitalizations, and the average annual cost per person for treatment of a musculoskeletal condition is $7,800.
“If we continue on our current trajectory, we are choosing to accept more prevalence and incidence of these disorders, spiraling costs, restricted access to needed services, and less success in alleviating pain and suffering – a high cost,” Edward H. Yelin, PhD, cochair of the report’s steering committee, and professor of medicine and health policy at the University of California, San Francisco, said in a prepared statement in 2016. That same year, Brian F. Mandell, MD, PhD, editor of the Cleveland Clinic Journal of Medicine, penned an editorial in which he stated that “The time has come to move past using a one-size-fits-all fifth vital sign . . . and reflexively prescribing an opioid when pain is characterized as severe” (Clev Clin J Med. 2016. Jun;83[6]:400-1). A decade earlier, authors of a cross-sectional review at a single Department of Veterans Affairs medical center set out to assess the impact of the VA’s “Pain as the 5th Vital Sign” initiative on the quality of pain management (J Gen Intern Med. 2006;21[6]:607–12). They found that patients with substantial pain documented by the fifth vital sign often had inadequate pain management. The preponderance of existing evidence suggests that a different approach is needed to prescribing opioids, Dr. Bonakdar said. “It’s coming from every voice in pain care: that what we are doing is not working,” he said. “It’s not only not working; it’s dangerous. That’s the consequence of depersonalized medicine. What’s the consequence of depersonalized nutrition? It’s the same industrialized approach.”
The typical American diet, he continued, is rife with processed foods and lacks an adequate proportion of plant-based products. “It’s basically a setup for inflammation,” Dr. Bonakdar said. “Most people who come into our clinic are eating 63% processed foods, 25% animal foods, and 12% plant foods. When we are eating, we’re oversizing it because that’s the American thing to do. At the end of the day, this process is not only killing us from heart disease and stroke as causes of death, but it’s also killing us as far as pain. The same diet that’s causing heart disease is the same diet that’s increasing pain.”
Dr. Bonakdar said that the ingestion of ultra-processed foods over time jumpstarts the process of dysbiosis, which increases gut permeability. “When gut permeability happens, and you have high levels of polysaccharides and inflammatory markers such as zonulin and lipopolysaccharide (LPS), it not only goes on to affect adipose tissue and insulin resistance, it can affect the muscle and joints,” he explained. “That is a setup for sarcopenia, or muscle loss, which then makes it harder for patients to be fully functional and active. It goes on to cause joint problems as well.”
He likened an increase in gut permeability to “a bomb going off in the gut.” Routine consumption of highly processed foods “creates this wave of inflammation that goes throughout your body affecting joints and muscles, and causes an increased amount of pain. Over time, patients make the connection but it’s much easier to say, ‘take this NSAID’ or ‘take this Cox-2 inhibitor’ to suppress the pain. But if all you’re doing is suppressing, you’re not going to the source of the pain.”
Dr. Bonakdar cited several recent articles that help to make the connection between dysbiosis and pain, including a review that concluded that dysbiosis of gut microbiota can influence the onset and progression of chronic degenerative diseases (Nutrients. 2019;11[8]:1707). Authors of a separate review concluded that human microbiome studies strongly suggest an incriminating role of microbes in the pathophysiology and progression of RA. Lastly, several studies have noted that pain conditions such as fibromyalgia may have microbiome “signatures” related to dysbiosis, which may pave the way for interventions, such as dietary shifting and probiotics that target individuals with microbiome abnormalities (Pain. 2019 Nov;160[11]:2589-602 and EBioMedicine. 2019 Aug 1;46:499-511).
Clinicians can begin to help patients who present with pain complaints “by listening to what their current pattern is: strategies that have worked, and those that haven’t,” he said. “If we’re not understanding the person and we’re just ordering genetic studies or microbiome studies and going off of the assessment, we sometime miss what interventions to start. In many cases, a simple intervention like a dietary shift is all that’s required.”
A survey of more than 1 million individuals found that BMI and daily pain are positively correlated in the United States (Obesity 2012;20[7]:1491-5). “This is increased more significantly for women and the elderly,” said Dr. Bonakdar, who was not affiliated with the study. “If we can change the diet that person is taking, that’s going to begin the process of reversing this to the point where they’re having less pain from inflammation that’s affecting the adipose tissue and adipokines traveling to their joints, which can cause less dysbiosis. It is very much a vicious cycle that patients follow, but if you begin to unwind it, it’s going to help multiple areas.”
In the Intensive Diet and Exercise for Arthritis (IDEA) trial, researchers randomized 450 patients with osteoarthritis to intensive dietary restriction only, exercise only, or a combination of both (BMC Musculoskelet Disord. 2009;10:93). They found that a 5% weight loss over the course of 18 months led to a 30% reduction in pain and a 24% improvement in function.
Inspired by the IDEA trial design, Dr. Bonakdar and his colleagues completed an unpublished 12-week pilot program with 12 patients with a BMI of 27 kg/m2 or greater plus comorbidities. The program consisted of weekly group meetings, including a lecture by team clinicians, dietician, and fitness staff; group support sessions with a behavioral counselor; and a group exercise session. It also included weekly 1:1 personal training sessions and biweekly 1:1 dietitian meetings. The researchers also evaluated several deficiencies linked to pain, including magnesium, vitamin D, vitamins B1, B2, and B12, folate, calcium, amino acids, omega 3s, zinc, coenzyme Q10, carnitine, and vitamin C. The goal was a weight reduction of 5%.
The intervention consisted of a 28-day detox/protein shake consumed 1-3 times per day, which contained 17 g of protein per serving. Nutritional supplementation was added based on results of individual diagnostics.
According to preliminary results from the trial, the intended weight goal was achieved. “More importantly, there were significant improvements in markers of dysbiosis, including zonulin and lipopolysaccharide, as well as the adipokine leptin, which appeared to be associated with improvement in quality of life measures and pain,” Dr. Bonakdar said.
He concluded his presentation by highlighting a pilot study conducted in an Australian tertiary pain clinic. It found that a personalized dietitian-delivered dietary intervention can improve pain scores, quality of life, and dietary intake of people experiencing chronic pain (Nutrients. 2019 Jan 16;11[1] pii: E181). “This is another piece of the puzzle showing that these dietary interventions can be done in multiple settings, including tertiary centers with nutrition staff, and that this important step can improve pain and quality of life,” he said.
Dr. Bonakdar disclosed that he receives royalties from Oxford University Press, Lippincott, and Elsevier. He is also a consultant to Standard Process.
REPORTING FROM A NATURAL SUPPLEMENTS UPDATE
FDA not recommending recalls of diabetes drug metformin
The Food and Drug Administration says it has no plans to recall any metformin products, used for the treatment of type 2 diabetes, after tests it conducted did not show any evidence of contamination with N-nitrosodimethylamine (NDMA) at levels that would cause concern.
The FDA began testing samples of metformin for the carcinogen NDMA at the end of 2019. Contamination with this substance has led to recalls of hypertension and heartburn medications within the past 2 years.
That announcement came on the heels of a recall of three versions of metformin in Singapore and the European Medicines Agency’s request that manufacturers test for NDMA.
This week, the FDA posted laboratory results in which NDMA levels in some metformin products ranged from “not detectable to low.”
“To date, no sample of metformin that FDA has tested exceeds the acceptable daily intake for NDMA. FDA has not recommended metformin recalls in the U.S.,” the agency indicates.
More than 30 million people in the United States have diabetes; 90%-95% of cases are of type 2. Metformin is the fourth most prescribed drug in the United States.
“Patients should continue taking metformin to keep their diabetes under control,” the FDA emphasized. “It could be dangerous for patients with this serious condition to stop taking their metformin without first talking to their health care professionals.”
The agency plans to post the methods used in laboratory testing of metformin in the near future. The FDA is collaborating with international regulators to share testing results for metformin, along with testing results for other drugs.
The U.S. agency says it will continue to monitor NDMA in metformin, along with other drug products, and will provide timely updates of new developments, including product recalls.
For more information about NDMA, visit the FDA’s nitrosamines webpage.
The FDA also encourages health care professionals and patients to report adverse reactions or quality problems with any human drugs to the agency’s MedWatch Adverse Event Reporting program.
This article first appeared on Medscape.com.
The Food and Drug Administration says it has no plans to recall any metformin products, used for the treatment of type 2 diabetes, after tests it conducted did not show any evidence of contamination with N-nitrosodimethylamine (NDMA) at levels that would cause concern.
The FDA began testing samples of metformin for the carcinogen NDMA at the end of 2019. Contamination with this substance has led to recalls of hypertension and heartburn medications within the past 2 years.
That announcement came on the heels of a recall of three versions of metformin in Singapore and the European Medicines Agency’s request that manufacturers test for NDMA.
This week, the FDA posted laboratory results in which NDMA levels in some metformin products ranged from “not detectable to low.”
“To date, no sample of metformin that FDA has tested exceeds the acceptable daily intake for NDMA. FDA has not recommended metformin recalls in the U.S.,” the agency indicates.
More than 30 million people in the United States have diabetes; 90%-95% of cases are of type 2. Metformin is the fourth most prescribed drug in the United States.
“Patients should continue taking metformin to keep their diabetes under control,” the FDA emphasized. “It could be dangerous for patients with this serious condition to stop taking their metformin without first talking to their health care professionals.”
The agency plans to post the methods used in laboratory testing of metformin in the near future. The FDA is collaborating with international regulators to share testing results for metformin, along with testing results for other drugs.
The U.S. agency says it will continue to monitor NDMA in metformin, along with other drug products, and will provide timely updates of new developments, including product recalls.
For more information about NDMA, visit the FDA’s nitrosamines webpage.
The FDA also encourages health care professionals and patients to report adverse reactions or quality problems with any human drugs to the agency’s MedWatch Adverse Event Reporting program.
This article first appeared on Medscape.com.
The Food and Drug Administration says it has no plans to recall any metformin products, used for the treatment of type 2 diabetes, after tests it conducted did not show any evidence of contamination with N-nitrosodimethylamine (NDMA) at levels that would cause concern.
The FDA began testing samples of metformin for the carcinogen NDMA at the end of 2019. Contamination with this substance has led to recalls of hypertension and heartburn medications within the past 2 years.
That announcement came on the heels of a recall of three versions of metformin in Singapore and the European Medicines Agency’s request that manufacturers test for NDMA.
This week, the FDA posted laboratory results in which NDMA levels in some metformin products ranged from “not detectable to low.”
“To date, no sample of metformin that FDA has tested exceeds the acceptable daily intake for NDMA. FDA has not recommended metformin recalls in the U.S.,” the agency indicates.
More than 30 million people in the United States have diabetes; 90%-95% of cases are of type 2. Metformin is the fourth most prescribed drug in the United States.
“Patients should continue taking metformin to keep their diabetes under control,” the FDA emphasized. “It could be dangerous for patients with this serious condition to stop taking their metformin without first talking to their health care professionals.”
The agency plans to post the methods used in laboratory testing of metformin in the near future. The FDA is collaborating with international regulators to share testing results for metformin, along with testing results for other drugs.
The U.S. agency says it will continue to monitor NDMA in metformin, along with other drug products, and will provide timely updates of new developments, including product recalls.
For more information about NDMA, visit the FDA’s nitrosamines webpage.
The FDA also encourages health care professionals and patients to report adverse reactions or quality problems with any human drugs to the agency’s MedWatch Adverse Event Reporting program.
This article first appeared on Medscape.com.
Opioid use disorder in adolescents: An overview
Ms. L, age 17, seeks treatment because she has an ongoing struggle with multiple substances, including benzodiazepines, heroin, alcohol, cannabis, and prescription opioids.
She reports that she was 13 when she first used a prescription opioid that was not prescribed for her. She also reports engaging in unsafe sexual practices while using these substances, and has been diagnosed and treated for a sexually transmitted disease. She dropped out of school and is estranged from her family. She says that for a long time she has felt depressed and that she uses drugs to “self-medicate my emotions.” She endorses high anxiety and lack of motivation. Ms. L also reports having several criminal charges for theft, assault, and exchanging sex for drugs. She has undergone 3 admissions for detoxification, but promptly resumed using drugs, primarily heroin and oxycodone, immediately after discharge. Ms. L meets DSM-5 criteria for opioid use disorder (OUD).
Ms. L’s case illustrates a disturbing trend in the current opioid epidemic in the United States. Nearly 11.8 million individuals age ≥12 reported misuse of opioids in the last year.1 Adolescents who misuse prescription or illicit opioids are more likely to be involved with the legal system due to truancy, running away from home, physical altercations, prostitution, exchanging sex for drugs, robbery, and gang involvement. Adolescents who use opioids may also struggle with academic decline, drop out of school early, be unable to maintain a job, and have relationship difficulties, especially with family members.
In this article, I describe the scope of OUD among adolescents, including epidemiology, clinical manifestations, screening tools, and treatment approaches.
Scope of the problem
According to the most recent Monitoring the Future survey of more than 42,500 8th, 10th, and 12th grade students, 2.7% of 12th graders reported prescription opioid misuse (reported in the survey as “narcotics other than heroin”) in the past year.2 In addition, 0.4% of 12th graders reported heroin use over the same period.2 Although the prevalence of opioid use among adolescents has been declining over the past 5 years,2 it still represents a serious health crisis.
Part of the issue may relate to easier access to more potent opioids. For example, heroin available today can be >4 times purer than it was in the past. In 2002, t
Between 1997 and 2012, the annual incidence of youth (age 15 to 19) hospitalizations for prescription opioid poisoning increased >170%.5 Approximately 6% to 9% of youth involved in risky opioid use develop OUD 6 to 12 months after s
Continue to: In recent years...
In recent years, deaths from drug overdose have increased for all age groups; however, limited data is available regarding adolescent overdose deaths. According to the Centers for Disease Control and Prevention (CDC), from 2015 to 2016, drug overdose death rates for persons age 15 to 24 increased to 28%.9
How opioids work
Opioids activate specific transmembrane neurotransmitter receptors, including mu, kappa, and delta, in the CNS and peripheral nervous system (PNS). This leads to activation of G protein–mediated intracellular signal transduction. Mainly it is activation of endogenous mu opioid receptors that mediates the reward, withdrawal, and analgesic effects of opioids. These effects depend on the location of mu receptors. In the CNS, activation of mu opioid receptors may cause miosis, respiratory depression, euphoria, and analgesia.10
Different opioids vary in terms of their half-life; for most opioids, the half-life ranges from 2 to 4 hours.10 Heroin has a half-life of 30 minutes, but due to active metabolites its duration of action is 4 to 5 hours. Opioid metabolites can be detected in urine toxicology within approximately 1 to 2 days since last use.10
Chronic opioid use is associated with neurologic effects that change the function of areas of the brain that control pleasure/reward, stress, decision-making, and more. This leads to cravings, continued substance use, and dependence.11 After continued long-term use, patients report decreased euphoria, but typically they continue to use opioids to avoid withdrawal symptoms or worsening mood.
Criteria for opioid use disorder
In DSM-5, substance use disorders (SUDs)are no longer categorized as abuse or dependence.12 For opioids, the diagnosis is OUD. The Table12 outlines the DSM-5 criteria for OUD. Craving opioids is included for the first time in the OUD diagnosis. Having problems with the legal system is no longer considered a diagnostic criterion for OUD.
Continue to: A vulnerable population
A vulnerable population
As defined by Erik Erikson’s psychosocial stages of development, adolescents struggle between establishing their own identity vs role confusion.13 In an attempt to relate to peers or give in to peer pressure, some adolescents start by experimenting with nicotine, alcohol, and/or marijuana; however, some may move on to using other illicit drugs.14 Risk factors for the development of SUDs include early onset of substance use and a rapid progression through stages of substance use from experimentation to regular use, risky use, and dependence.15 In our case study, Ms. L’s substance use followed a similar pattern. Further, the comorbidity of SUDs and other psychiatric disorders may add a layer of complexity when caring for adolescents. Box 116-20 describes the relationship between comorbid psychiatric disorders and SUDs in adolescents.
Box 1
Disruptive behavior disorders are the most common coexisting psychiatric disorders in an adolescent with a substance use disorder (SUD), including opioid use disorder. These individuals typically present with aggression and other conduct disorder symptoms, and have early involvement with the legal system. Conversely, patients with conduct disorder are at high risk of early initiation of illicit substance use, including opioids. Early onset of substance use is a strong risk factor for developing an SUD.16
Mood disorders, particularly depression, can either precede or occur as a result of heavy and prolonged substance use.17 The estimated prevalence of major depressive disorder in individuals with an SUD is 24% to 50%. Among adolescents, an SUD is also a risk factor for suicidal ideation, suicide attempts, and completed suicide.18-20
Anxiety disorders, especially social phobia, and posttraumatic stress disorder are common in individuals with SUD.
Adolescents with SUD should be carefully evaluated for comorbid psychiatric disorders and treated accordingly.
Clinical manifestations
Common clinical manifestations of opioid use vary depending on when the patient is seen. An individual with OUD may appear acutely intoxicated, be in withdrawal, or show no effects. Chronic/prolonged use can lead to tolerance, such that a user needs to ingest larger amounts of the opioid to produce the same effects.
Acute intoxication can cause sedation, slurring of speech, and pinpoint pupils. Fresh injection sites may be visible on physical examination of IV users. The effects of acute intoxication usually depend on the half-life of the specific opioid and the individual’s tolerance.10 Tolerance to heroin can occur in 10 days and withdrawal can manifest in 3 to 7 hours after last use, depending on dose and purity.3 Tolerance can lead to unintentional overdose and death.
Withdrawal. Individuals experiencing withdrawal from opioids present with flu-like physical symptoms, including generalized body ache, rhinorrhea, diarrhea, goose bumps, lacrimation, and vomiting. Individuals also may experience irritability, restlessness, insomnia, anxiety, and depression during withdrawal.
Other manifestations. Excessive and chronic/prolonged opioid use can adversely impact socio-occupational functioning and cause academic decline in adolescents and youth. Personal relationships are significantly affected. Opioid users may have legal difficulties as a result of committing crimes such as theft, prostitution, or robbery in order to obtain opioids.
Continue to: Screening for OUD
Screening for OUD
Several screening tools are available to assess adolescents for SUDs, including OUD.
CRAFFT is a 6-item, clinician-administered screening tool that has been approved by American Academy of Pediatrics’ Committee on Substance Abuse for adolescents and young adults age <21.21-23 This commonly used tool can assess for alcohol, cannabis, and other drug use. A score ≥2 is considered positive for drug use, indicating that the individual would require further evaluation and assessment22,23 (Figure). There is also a self-administered CRAFFT questionnaire that can be completed by the patient.

NIDA-modified ASSIST. The American Psychiatric Association has adapted the National Institute on Drug Abuse (NIDA)-modified ASSIST. One version is designated for parents/guardians to administer to their children (age 6 to 17), and one is designated for adolescents (age 11 to 17) to self-administer.24,25 Each screening tool has 2 levels: Level 1 screens for substance use and other mental health symptoms, and Level 2 is more specific for substance use alone.
Drug Use Screening Inventory (DUSI) is a self-report questionnaire that has 149 items that assess the use of numerous drugs. It is designed to quantify the severity of consequences associated with drug and alcohol use.26,27
Problem-Oriented Screening Instrument for Teenagers (PO
Continue to: Personal Experience Screening Questionnaire (PESQ)...
Personal Experience Screening Questionnaire (PESQ) is a brief, 40-item, cost-effective, self-report questionnaire that can help identify adolescents (age 12 to 18) who should be referred for further evaluation.30
Addressing treatment expectations
For an adolescent with OUD, treatment should begin in the least restrictive environment that is perceived as safe for the patient. An adolescent’s readiness and motivation to achieve and maintain abstinence are crucial. Treatment planning should include the adolescent as well as his/her family to ensure they are able to verbalize their expectations. Start with a definitive treatment plan that addresses an individual’s needs. The plan should provide structure and an understanding of treatment expectations. The treatment team should clarify the realistic plan and goals based on empirical and clinical evidence. Treatment goals should include interventions to strengthen interpersonal relationships and assist with rehabilitation, such as establishing academic and/or vocational goals. Addressing readiness and working on a patient’s motivation is extremely important for most of these interventions.
In order for any intervention to be successful, clinicians need to establish and foster rapport with the adolescent. By law, substance use or behaviors related to substance use are not allowed to be shared outside the patient-clinician relationship, unless the adolescent gives consent or there are concerns that such behaviors might put the patient or others at risk. It is important to prime the adolescent and help them understand that any information pertaining to their safety or the safety of others may need to be shared outside the patient-clinician relationship.
Choosing an intervention
Less than 50% of a nationally representative sample of 345 addiction treatment programs serving adolescents and adults offer medications for treating OUD.31 Even in programs that offer pharmacotherapy, medications are significantly underutilized. Fewer than 30% of patients in addiction treatment programs receive medication, compared with 74% of patients receiving treatment for other mental health disorders.31 A
Psychotherapy may be used to treat OUD in adolescents. Several family therapies have been studied and are considered as critical psychotherapeutic interventions for treating SUDs, including structural family treatment and functional family therapy approaches.34 An integrated behavioral and family therapy model is also recommended for adolescent patients with SUDs. Cognitive distortions and use of self-deprecatory statements are common among adolescents.35 Therefore, using approaches of cognitive-behavioral therapy (CBT), or CBT plus motivational enhancement therapy, also might be effective for this population.36 The adolescent community reinforcement approach (A-CRA) is a behavioral treatment designed to help adolescents and their families learn how to lead a healthy and happy life without the use of drugs or alcohol by increasing access to social, familial, and educational/vocational reinforcers. Support groups and peer and family support should be encouraged as adjuncts to other interventions. In some areas, sober housing options for adolescents are also available.
Continue to: Harm-reduction strategies
Harm-reduction strategies. Although the primary goal of treatment for adolescents with OUD is to achieve and maintain abstinence from opioid use, implicit and explicit goals can be set. Short-term implicit goals may include harm-reduction strategies that emphasize decreasing the duration, frequency, and amount of substance use and limiting the chances of adverse effects, while the long-term explicit goal should be abstinence from opioid use.
Naloxone nasal spray is used as a harm-reduction strategy. It is an FDA-approved formulation that can reverse the effects of unintentional opioid overdoses and potentially prevent death from respiratory depression.37 Other harm-reduction strategies include needle exchange programs, which provide sterile needles to individuals who inject drugs in an effort to prevent or reduce the transmission of human immunodeficiency virus and other bloodborne viruses that can be spread via shared injection equipment. Fentanyl testing strips allow opioid users to test for the presence fentanyl and fentanyl analogs in the unregulated “street” opioid supply.
Pharmacologic interventions. Because there is limited empirical evidence on the efficacy of medication-assisted treatment (MAT) for adolescents with OUD, clinicians need to rely on evidence from research and experience with adults. Unfortunately, MAT is offered to adolescents considerably less often than it is to adults. Feder et al38 reported that only 2.4% of adolescents received MAT for heroin use and only 0.4% of adolescents received MAT for prescription opioid use, compared with 26.3% and 12% of adults, respectively.
Detoxification. Medications available for detoxification from opioids include opiates (such as methadone or buprenorphine) and clonidine (a central sympathomimetic). If the patient has used heroin for a short period (<1 year) and has no history of detoxification, consider a detoxification strategy with a longer-term taper (90 to 180 days) to allow for stabilization.
Maintenance treatment. Consider maintenance treatment for adolescents with a history of long-term opioid use and at least 2 prior short-term detoxification attempts or nonpharmacotherapy-based treatment within 12 months. Be sure to receive consent from a legal guardian and the patient. Maintenance treatment is usually recommended to continue for 1 to 6 years. Maintenance programs with longer durations have shown higher rates of abstinence, improved engagement, and retention in treatment.39
Continue to: According to guidelines from...
According to guidelines from the American Society of Addiction Medicine (ASAM), adolescents age >16 should be offered MAT; the first-line treatment is buprenorphine.40 To avoid risks of abuse and diversion, a combination of buprenorphine/naloxone may be administered.
Maintenance with buprenorphine
In order to prescribe and dispense buprenorphine, clinicians need to obtain a waiver from the Substance Abuse and Mental Health Services Administration. Before initiating buprenorphine, consider the type of opioid the individual used (short- or long-acting), the severity of the OUD, and the last reported use. The 3 phases of buprenorphine treatment are41:
- Induction phase. Buprenorphine can be initiated at 2 to 4 mg/d. Some patients may require up to 8 mg/d on the first day, which can be administered in divided doses.42 Evaluate and monitor patients carefully during the first few hours after the first dose. Patients should be in early withdrawal; otherwise, the buprenorphine might precipitate withdrawal. The induction phase can be completed in 2 to 4 days by titrating the dose so that the signs and symptoms of opioid withdrawal are minimal, and the patient is able to continue treatment. It may be helpful to have the patient’s legal guardian nearby in case the patient does not tolerate the medication or experiences withdrawal. The initial target dose for buprenorphine is approximately 12 to 16 mg/d.
- Stabilization phase. Patients no longer experience withdrawal symptoms and no longer have cravings. This phase can last 6 to 8 weeks. During this phase, patients should be seen weekly and doses should be adjusted if necessary. As a partial mu agonist, buprenorphine does not activate mu receptors fully and reaches a ceiling effect. Hence, doses >24 mg/d have limited added agonist properties.
- Maintenance phase. Because discontinuation of buprenorphine is associated with high relapse rates, patients may need to be maintained long-term on their stabilization dose, and for some patients, the length of time could be indefinite.39 During this phase, patients continue to undergo follow-up, but do so less frequently.
Methadone maintenance is generally not recommended for individuals age <18.
Preventing opioid diversion
Prescription medications that are kept in the home are a substantial source of opioids for adolescents. In 2014, 56% of 12th graders who did not need medications for medical purposes were able to acquire them from their friends or relatives; 36% of 12th graders used their own prescriptions.21 Limiting adolescents’ access to prescription opioids is the first line of prevention. Box 2 describes interventions and strategies to limit adolescents’ access to opioids.
Box 2
Many adolescents obtain opioids for recreational use from medications that were legitimately prescribed to family or friends. Both clinicians and parents/ guardians can take steps to reduce or prevent this type of diversion
Health care facilities. Regulating the number of pills dispensed to patients is crucial. It is highly recommended to prescribe only the minimal number of opioids necessary. In most cases, 3 to 7 days’ worth of opioids at a time might be sufficient, especially after surgical procedures.
Home. Families can limit adolescents’ access to prescription opioids in the home by keeping all medications in a lock box.
Proper disposal. Various entities offer locations for patients to drop off their unused opioids and other medications for safe disposal. These include police or fire departments and retail pharmacies. The US Drug Enforcement Administration sponsors a National Prescription Drug Take Back Day; see https://www.deadiversion.usdoj.gov/drug_disposal/takeback/index.html. The FDA also offers information on where and how to dispose of unused medicines at https://www.fda.gov/consumers/consumer-updates/where-and-how-dispose-unused-medicines.
CASE CONTINUED
Ms. L is initially prescribed, clonidine, 0.1 mg every 6 hours, to address opioid withdrawal. Clonidine is then tapered and maintained at 0.1 mg twice a day for irritability and impulse control. She is also prescribed sertraline, 100 mg/d, for depression and anxiety, and trazodone, 75 mg as needed at night, to assist with sleep.
Continue to: Following inpatient hospitalization...
Following inpatient hospitalization, during 12 weeks of partial hospital treatment, Ms. L participates in individual psychotherapy sessions 5 days/week; family therapy sessions once a week; and experiential therapy along with group sessions with other peers. She undergoes medication evaluations and adjustments on a weekly basis. Ms. L is now working at a store and is pursuing a high school equivalency certificate. She manages to avoid high-risk behaviors, although she reports having occasional cravings. Ms. L is actively involved in Narcotics Anonymous and has a sponsor. She has reconciled with her mother and moved back home, so she can stay away from her former acquaintances who are still using.
Bottom Line
Adolescents with opioid use disorder can benefit from an individualized treatment plan that includes psychosocial interventions, pharmacotherapy, or a combination of the two. Treatment planning should include the adolescent and his/her family to ensure they are able to verbalize their expectations. Treatment should focus on interventions that strengthen interpersonal relationships and assist with rehabilitation. Ongoing follow-up care is necessary for maintaining abstinence.
Related Resource
- Patkar AA, Weisler RH. Opioid abuse and overdose: Keep your patients safe. Current Psychiatry. 2017;16(8):8-12,14-16.
Drug Brand Names
Buprenorphine • Subutex, Sublocade
Buprenorphine/naloxone • Suboxone
Clonidine • Clorpres
Methadone • Methadose
Naloxone • Narcan
Oxycodone • OxyContin
Sertraline • Zoloft
Tramadol • Ultram
Trazodone • Desyrel, Oleptro
1. Davis JP, Prindle JJ, Eddie D, et al. Addressing the opioid epidemic with behavioral interventions for adolescents and young adults: a quasi-experimental design. J Consult Clin Psychol. 2019;87(10):941-951.
2. National Institute on Drug Abuse; National Institutes of Health; U.S. Department of Health and Human Services. Monitoring the Future Survey: High School and Youth Trends. https://www.drugabuse.gov/publications/drugfacts/monitoring-future-survey-high-school-youth-trends. Updated December 2019. Accessed January 13, 2020.
3. Hopfer CJ, Khuri E, Crowley TJ. Treating adolescent heroin use. J Am Acad Child Adolesc Psychiatry. 2003;42(5):609-611.
4. US Department of Justice, Drug Enforcement Agency, Diversion Control Division. https://www.deadiversion.usdoj.gov/. Accessed January 21, 2020.
5. Gaither JR, Leventhal JM, Ryan SA, et al. National trends in hospitalizations for opioid poisonings among children and adolescents, 1997-2012. JAMA Pediatr. 2016;170(12):1195-1201.
6. Parker MA, Anthony JC. Epidemiological evidence on extra-medical use of prescription pain relievers: transitions from newly incident use to dependence among 12-21 year olds in United States using meta-analysis, 2002-13. Peer J. 2015;3:e1340. doi: 10.7717/peerj.1340. eCollection 2015.
7. Subramaniam GA, Fishman MJ, Woody G. Treatment of opioid-dependent adolescents and young adults with buprenorphine. Curr Psychiatry Rep. 2009;11(5):360-363.
8. Borodovsky JT, Levy S, Fishman M. Buprenorphine treatment for adolescents and young adults with opioid use disorders: a narrative review. J Addict Med. 2018;12(3):170-183.
9. Centers for Disease Control and Prevention: National Center for Health Statistics. Drug overdose deaths in the United States, 1999-2016. https://www.cdc.gov/nchs/products/databriefs/db294.htm. Published December 2017. Accessed January 15, 2020.
10. Strain E. Opioid use disorder: epidemiology, pharmacology, clinical manifestation, course, screening, assessment, diagnosis. https://www.uptodate.com/contents/opioid-use-disorder-epidemiology-pharmacology-clinical-manifestations-course-screening-assessment-and-diagnosis. Updated August 15, 2019. Accessed January 21, 2020.
11. American Academy of Pediatrics Committee on Substance Use and Prevention. Policy statement: medication-assisted treatment of adolescents with opioid use disorder. Pediatrics. 2016;138(3):e20161893. doi: https://doi.org/10.1542/peds.2016-1893.
12. Diagnostic and Statistical Manual of Mental Disorders, 5th ed. Washington, DC: American Psychiatric Association; 2013:514.
13. Sadock BJ, Sadock VA. Chapter 6: Theories of personality and psychopathology. In: Sadock BJ, Sadock VA, eds. Kaplan and Sadock’s synopsis of psychiatry: behavioral sciences/clinical. 10th ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2007:209.
14. Kandel DB. Stages and pathways of drug involvement: examining the gateway hypothesis. Cambridge, United Kingdom: Cambridge University Press; 2002.
15. Robins LN, McEvoy L. Conduct problems as predictors of substance abuse. In: Robins LN, Rutter M, eds. Straight and devious pathways from childhood to adulthood. Cambridge, United Kingdom: Cambridge University Press; 1990;182-204.
16. Hopfer C, Salomonsen-Sautel S, Mikulich-Gilbertson S, et al. Conduct disorder and initiation of substance use: a prospective longitudinal study. J Am Acad Child Adolesc Psychiatry. 2013;52(5):511-518.e4.
17. Armstrong TD, Costello EJ. Community studies on adolescent substance use, abuse, or dependence and psychiatric comorbidity. J Consult Clin Psychol. 2002;70(6):1224-1239.
18. Crumley FE. Substance abuse and adolescent suicidal behavior. JAMA. 1990;263(22):3051-3056.
19. Lewinsohn PM, Rohde P, Seeley JR. Adolescent suicidal ideation and attempts: prevalence, risk factors, and clinical implications. Clinical Psychology: Science and Practice. 1996;3(1):25-46.
20. Kendler KS, Bulik CM, Silberg J, et al. Childhood sexual abuse and adult psychiatric and substance use disorder in women: an epidemiological and cotwin control analysis. Arch Gen Psychiatry. 2000;57(10):953-959.
21. Yule AM, Wilens TE, Rausch PK. The opioid epidemic: what a child psychiatrist is to do? J Am Acad Child Adolesc Psychiatry. 2017;56(7);541-543.
22. CRAFFT. https://crafft.org. Accessed January 21, 2020.
23. Knight JR, Sherritt L, Harris SK, et al. Validity of brief alcohol screening tests among adolescents: a comparison of the AUDIT, POSIT, CAGE, and CRAFFT. Alcohol Clin Exp Res. 2003;27(1):67-73.
24. American Psychiatric Association. Online assessment measures. https://www.psychiatry.org/psychiatrists/practice/dsm/educational-resources/assessment-measures. Accessed January 15, 2020.
25. National Institute of Drug Abuse. American Psychiatric Association adapted NIDA modified ASSIST tools. https://www.drugabuse.gov/nidamed-medical-health-professionals/tool-resources-your-practice/screening-assessment-drug-testing-resources/american-psychiatric-association-adapted-nida. Updated November 15, 2015. Accessed January 21, 2020.
26. Canada’s Mental Health & Addiction Network. Drug Use Screening Inventory (DUSI). https://www.porticonetwork.ca/web/knowledgex-archive/amh-specialists/screening-for-cd-in-youth/screening-both-mh-sud/dusi. Published 2009. Accessed January 21, 2020.
27. Tarter RE. Evaluation and treatment of adolescent substance abuse: a decision tree method. Am J Drug Alcohol Abuse. 1990;16(1-2):1-46.
28. Klitzner M, Gruenwald PJ, Taff GA, et al. The adolescent assessment referral system-final report. National Institute on Drug Abuse; Rockville, MD: 1993. NIDA Contract No. 271-89-8252.
29. Slesnick N, Tonigan JS. Assessment of alcohol and other drug use by runaway youths: a test-retest study of the Form 90. Alcohol Treat Q. 2004;22(2):21-34.
30. Winters KC, Kaminer Y. Screening and assessing adolescent substance use disorders in clinical populations. J Am Acad Child Adolesc Psychiatry. 2008;47(7):740-744.
31. Knudsen HK, Abraham AJ, Roman PM. Adoption and implementation of medications in addiction treatment programs. J Addict Med. 2011;5(1):21-27.
32. Deas D, Thomas SE. An overview of controlled study of adolescent substance abuse treatment. Am J Addiction. 2001;10(2):178-189.
33. William RJ, Chang, SY. A comprehensive and comparative review of adolescent substance abuse treatment outcome. Clinical Psychology: Science and Practice. 2000;7(2):138-166.
34. Bukstein OG, Work Group on Quality Issues. Practice parameters for the assessment and treatment of children and adolescents with substance use disorders. J Am Acad Child Adolesc Psychiatry. 2005;44(6):609-621.
35. Van Hasselt VB, Null JA, Kempton T, et al. Social skills and depression in adolescent substance abusers. Addict Behav. 1993;18(1):9-18.
36. Dennis M, Godley SH, Diamond G, et al. The Cannabis Youth Treatment (CYT) study: main findings from two randomized trials. J Subst Abuse Treat. 2004;27(3):197-213.
37. US Food and Drug Administration. Information about naloxone. https://www.fda.gov/drugs/postmarket-drug-safety-information-patients-and-providers/information-about-naloxone. Updated December 19, 2019. Accessed January 21, 2020.
38. Feder KA, Krawcyzk N, Saloner, B. Medication-assisted treatment for adolescents in specialty treatment for opioid use disorder. J Adolesc Health. 2018;60(6):747-750.
39. Woody GE, Poole SA, Subramaniam G, et al. Extended vs short-term buprenorphine-naloxone for treatment of opioid-addicted youth: a randomized trial. JAMA. 2008;300(17):2003-2011.
40. US Department of Health and Human Services. Substance Abuse and Mental Health Ser-vices Administration. Medication-assisted treatment for opioid addiction in opioid treatment programs: a treatment improvement protocol TIP 43. https://www.asam.org/docs/advocacy/samhsa_tip43_matforopioidaddiction.pdf?sfvrsn=0. Published 2005. Accessed January 15, 2020.
41. US Department of Health and Human Services. Substance Abuse and Mental Health Services Administration. Medication-assisted treatment (MAT). https://www.samhsa.gov/medication-assisted-treatment. Updated September 9, 2019. Accessed January 21, 2020.
42. Johnson RE, Strain EC, Amass L. Buprenorphine: how to use it right. Drug Alcohol Depend. 2003;70(suppl 2):S59-S77.
Ms. L, age 17, seeks treatment because she has an ongoing struggle with multiple substances, including benzodiazepines, heroin, alcohol, cannabis, and prescription opioids.
She reports that she was 13 when she first used a prescription opioid that was not prescribed for her. She also reports engaging in unsafe sexual practices while using these substances, and has been diagnosed and treated for a sexually transmitted disease. She dropped out of school and is estranged from her family. She says that for a long time she has felt depressed and that she uses drugs to “self-medicate my emotions.” She endorses high anxiety and lack of motivation. Ms. L also reports having several criminal charges for theft, assault, and exchanging sex for drugs. She has undergone 3 admissions for detoxification, but promptly resumed using drugs, primarily heroin and oxycodone, immediately after discharge. Ms. L meets DSM-5 criteria for opioid use disorder (OUD).
Ms. L’s case illustrates a disturbing trend in the current opioid epidemic in the United States. Nearly 11.8 million individuals age ≥12 reported misuse of opioids in the last year.1 Adolescents who misuse prescription or illicit opioids are more likely to be involved with the legal system due to truancy, running away from home, physical altercations, prostitution, exchanging sex for drugs, robbery, and gang involvement. Adolescents who use opioids may also struggle with academic decline, drop out of school early, be unable to maintain a job, and have relationship difficulties, especially with family members.
In this article, I describe the scope of OUD among adolescents, including epidemiology, clinical manifestations, screening tools, and treatment approaches.
Scope of the problem
According to the most recent Monitoring the Future survey of more than 42,500 8th, 10th, and 12th grade students, 2.7% of 12th graders reported prescription opioid misuse (reported in the survey as “narcotics other than heroin”) in the past year.2 In addition, 0.4% of 12th graders reported heroin use over the same period.2 Although the prevalence of opioid use among adolescents has been declining over the past 5 years,2 it still represents a serious health crisis.
Part of the issue may relate to easier access to more potent opioids. For example, heroin available today can be >4 times purer than it was in the past. In 2002, t
Between 1997 and 2012, the annual incidence of youth (age 15 to 19) hospitalizations for prescription opioid poisoning increased >170%.5 Approximately 6% to 9% of youth involved in risky opioid use develop OUD 6 to 12 months after s
Continue to: In recent years...
In recent years, deaths from drug overdose have increased for all age groups; however, limited data is available regarding adolescent overdose deaths. According to the Centers for Disease Control and Prevention (CDC), from 2015 to 2016, drug overdose death rates for persons age 15 to 24 increased to 28%.9
How opioids work
Opioids activate specific transmembrane neurotransmitter receptors, including mu, kappa, and delta, in the CNS and peripheral nervous system (PNS). This leads to activation of G protein–mediated intracellular signal transduction. Mainly it is activation of endogenous mu opioid receptors that mediates the reward, withdrawal, and analgesic effects of opioids. These effects depend on the location of mu receptors. In the CNS, activation of mu opioid receptors may cause miosis, respiratory depression, euphoria, and analgesia.10
Different opioids vary in terms of their half-life; for most opioids, the half-life ranges from 2 to 4 hours.10 Heroin has a half-life of 30 minutes, but due to active metabolites its duration of action is 4 to 5 hours. Opioid metabolites can be detected in urine toxicology within approximately 1 to 2 days since last use.10
Chronic opioid use is associated with neurologic effects that change the function of areas of the brain that control pleasure/reward, stress, decision-making, and more. This leads to cravings, continued substance use, and dependence.11 After continued long-term use, patients report decreased euphoria, but typically they continue to use opioids to avoid withdrawal symptoms or worsening mood.
Criteria for opioid use disorder
In DSM-5, substance use disorders (SUDs)are no longer categorized as abuse or dependence.12 For opioids, the diagnosis is OUD. The Table12 outlines the DSM-5 criteria for OUD. Craving opioids is included for the first time in the OUD diagnosis. Having problems with the legal system is no longer considered a diagnostic criterion for OUD.
Continue to: A vulnerable population
A vulnerable population
As defined by Erik Erikson’s psychosocial stages of development, adolescents struggle between establishing their own identity vs role confusion.13 In an attempt to relate to peers or give in to peer pressure, some adolescents start by experimenting with nicotine, alcohol, and/or marijuana; however, some may move on to using other illicit drugs.14 Risk factors for the development of SUDs include early onset of substance use and a rapid progression through stages of substance use from experimentation to regular use, risky use, and dependence.15 In our case study, Ms. L’s substance use followed a similar pattern. Further, the comorbidity of SUDs and other psychiatric disorders may add a layer of complexity when caring for adolescents. Box 116-20 describes the relationship between comorbid psychiatric disorders and SUDs in adolescents.
Box 1
Disruptive behavior disorders are the most common coexisting psychiatric disorders in an adolescent with a substance use disorder (SUD), including opioid use disorder. These individuals typically present with aggression and other conduct disorder symptoms, and have early involvement with the legal system. Conversely, patients with conduct disorder are at high risk of early initiation of illicit substance use, including opioids. Early onset of substance use is a strong risk factor for developing an SUD.16
Mood disorders, particularly depression, can either precede or occur as a result of heavy and prolonged substance use.17 The estimated prevalence of major depressive disorder in individuals with an SUD is 24% to 50%. Among adolescents, an SUD is also a risk factor for suicidal ideation, suicide attempts, and completed suicide.18-20
Anxiety disorders, especially social phobia, and posttraumatic stress disorder are common in individuals with SUD.
Adolescents with SUD should be carefully evaluated for comorbid psychiatric disorders and treated accordingly.
Clinical manifestations
Common clinical manifestations of opioid use vary depending on when the patient is seen. An individual with OUD may appear acutely intoxicated, be in withdrawal, or show no effects. Chronic/prolonged use can lead to tolerance, such that a user needs to ingest larger amounts of the opioid to produce the same effects.
Acute intoxication can cause sedation, slurring of speech, and pinpoint pupils. Fresh injection sites may be visible on physical examination of IV users. The effects of acute intoxication usually depend on the half-life of the specific opioid and the individual’s tolerance.10 Tolerance to heroin can occur in 10 days and withdrawal can manifest in 3 to 7 hours after last use, depending on dose and purity.3 Tolerance can lead to unintentional overdose and death.
Withdrawal. Individuals experiencing withdrawal from opioids present with flu-like physical symptoms, including generalized body ache, rhinorrhea, diarrhea, goose bumps, lacrimation, and vomiting. Individuals also may experience irritability, restlessness, insomnia, anxiety, and depression during withdrawal.
Other manifestations. Excessive and chronic/prolonged opioid use can adversely impact socio-occupational functioning and cause academic decline in adolescents and youth. Personal relationships are significantly affected. Opioid users may have legal difficulties as a result of committing crimes such as theft, prostitution, or robbery in order to obtain opioids.
Continue to: Screening for OUD
Screening for OUD
Several screening tools are available to assess adolescents for SUDs, including OUD.
CRAFFT is a 6-item, clinician-administered screening tool that has been approved by American Academy of Pediatrics’ Committee on Substance Abuse for adolescents and young adults age <21.21-23 This commonly used tool can assess for alcohol, cannabis, and other drug use. A score ≥2 is considered positive for drug use, indicating that the individual would require further evaluation and assessment22,23 (Figure). There is also a self-administered CRAFFT questionnaire that can be completed by the patient.

NIDA-modified ASSIST. The American Psychiatric Association has adapted the National Institute on Drug Abuse (NIDA)-modified ASSIST. One version is designated for parents/guardians to administer to their children (age 6 to 17), and one is designated for adolescents (age 11 to 17) to self-administer.24,25 Each screening tool has 2 levels: Level 1 screens for substance use and other mental health symptoms, and Level 2 is more specific for substance use alone.
Drug Use Screening Inventory (DUSI) is a self-report questionnaire that has 149 items that assess the use of numerous drugs. It is designed to quantify the severity of consequences associated with drug and alcohol use.26,27
Problem-Oriented Screening Instrument for Teenagers (PO
Continue to: Personal Experience Screening Questionnaire (PESQ)...
Personal Experience Screening Questionnaire (PESQ) is a brief, 40-item, cost-effective, self-report questionnaire that can help identify adolescents (age 12 to 18) who should be referred for further evaluation.30
Addressing treatment expectations
For an adolescent with OUD, treatment should begin in the least restrictive environment that is perceived as safe for the patient. An adolescent’s readiness and motivation to achieve and maintain abstinence are crucial. Treatment planning should include the adolescent as well as his/her family to ensure they are able to verbalize their expectations. Start with a definitive treatment plan that addresses an individual’s needs. The plan should provide structure and an understanding of treatment expectations. The treatment team should clarify the realistic plan and goals based on empirical and clinical evidence. Treatment goals should include interventions to strengthen interpersonal relationships and assist with rehabilitation, such as establishing academic and/or vocational goals. Addressing readiness and working on a patient’s motivation is extremely important for most of these interventions.
In order for any intervention to be successful, clinicians need to establish and foster rapport with the adolescent. By law, substance use or behaviors related to substance use are not allowed to be shared outside the patient-clinician relationship, unless the adolescent gives consent or there are concerns that such behaviors might put the patient or others at risk. It is important to prime the adolescent and help them understand that any information pertaining to their safety or the safety of others may need to be shared outside the patient-clinician relationship.
Choosing an intervention
Less than 50% of a nationally representative sample of 345 addiction treatment programs serving adolescents and adults offer medications for treating OUD.31 Even in programs that offer pharmacotherapy, medications are significantly underutilized. Fewer than 30% of patients in addiction treatment programs receive medication, compared with 74% of patients receiving treatment for other mental health disorders.31 A
Psychotherapy may be used to treat OUD in adolescents. Several family therapies have been studied and are considered as critical psychotherapeutic interventions for treating SUDs, including structural family treatment and functional family therapy approaches.34 An integrated behavioral and family therapy model is also recommended for adolescent patients with SUDs. Cognitive distortions and use of self-deprecatory statements are common among adolescents.35 Therefore, using approaches of cognitive-behavioral therapy (CBT), or CBT plus motivational enhancement therapy, also might be effective for this population.36 The adolescent community reinforcement approach (A-CRA) is a behavioral treatment designed to help adolescents and their families learn how to lead a healthy and happy life without the use of drugs or alcohol by increasing access to social, familial, and educational/vocational reinforcers. Support groups and peer and family support should be encouraged as adjuncts to other interventions. In some areas, sober housing options for adolescents are also available.
Continue to: Harm-reduction strategies
Harm-reduction strategies. Although the primary goal of treatment for adolescents with OUD is to achieve and maintain abstinence from opioid use, implicit and explicit goals can be set. Short-term implicit goals may include harm-reduction strategies that emphasize decreasing the duration, frequency, and amount of substance use and limiting the chances of adverse effects, while the long-term explicit goal should be abstinence from opioid use.
Naloxone nasal spray is used as a harm-reduction strategy. It is an FDA-approved formulation that can reverse the effects of unintentional opioid overdoses and potentially prevent death from respiratory depression.37 Other harm-reduction strategies include needle exchange programs, which provide sterile needles to individuals who inject drugs in an effort to prevent or reduce the transmission of human immunodeficiency virus and other bloodborne viruses that can be spread via shared injection equipment. Fentanyl testing strips allow opioid users to test for the presence fentanyl and fentanyl analogs in the unregulated “street” opioid supply.
Pharmacologic interventions. Because there is limited empirical evidence on the efficacy of medication-assisted treatment (MAT) for adolescents with OUD, clinicians need to rely on evidence from research and experience with adults. Unfortunately, MAT is offered to adolescents considerably less often than it is to adults. Feder et al38 reported that only 2.4% of adolescents received MAT for heroin use and only 0.4% of adolescents received MAT for prescription opioid use, compared with 26.3% and 12% of adults, respectively.
Detoxification. Medications available for detoxification from opioids include opiates (such as methadone or buprenorphine) and clonidine (a central sympathomimetic). If the patient has used heroin for a short period (<1 year) and has no history of detoxification, consider a detoxification strategy with a longer-term taper (90 to 180 days) to allow for stabilization.
Maintenance treatment. Consider maintenance treatment for adolescents with a history of long-term opioid use and at least 2 prior short-term detoxification attempts or nonpharmacotherapy-based treatment within 12 months. Be sure to receive consent from a legal guardian and the patient. Maintenance treatment is usually recommended to continue for 1 to 6 years. Maintenance programs with longer durations have shown higher rates of abstinence, improved engagement, and retention in treatment.39
Continue to: According to guidelines from...
According to guidelines from the American Society of Addiction Medicine (ASAM), adolescents age >16 should be offered MAT; the first-line treatment is buprenorphine.40 To avoid risks of abuse and diversion, a combination of buprenorphine/naloxone may be administered.
Maintenance with buprenorphine
In order to prescribe and dispense buprenorphine, clinicians need to obtain a waiver from the Substance Abuse and Mental Health Services Administration. Before initiating buprenorphine, consider the type of opioid the individual used (short- or long-acting), the severity of the OUD, and the last reported use. The 3 phases of buprenorphine treatment are41:
- Induction phase. Buprenorphine can be initiated at 2 to 4 mg/d. Some patients may require up to 8 mg/d on the first day, which can be administered in divided doses.42 Evaluate and monitor patients carefully during the first few hours after the first dose. Patients should be in early withdrawal; otherwise, the buprenorphine might precipitate withdrawal. The induction phase can be completed in 2 to 4 days by titrating the dose so that the signs and symptoms of opioid withdrawal are minimal, and the patient is able to continue treatment. It may be helpful to have the patient’s legal guardian nearby in case the patient does not tolerate the medication or experiences withdrawal. The initial target dose for buprenorphine is approximately 12 to 16 mg/d.
- Stabilization phase. Patients no longer experience withdrawal symptoms and no longer have cravings. This phase can last 6 to 8 weeks. During this phase, patients should be seen weekly and doses should be adjusted if necessary. As a partial mu agonist, buprenorphine does not activate mu receptors fully and reaches a ceiling effect. Hence, doses >24 mg/d have limited added agonist properties.
- Maintenance phase. Because discontinuation of buprenorphine is associated with high relapse rates, patients may need to be maintained long-term on their stabilization dose, and for some patients, the length of time could be indefinite.39 During this phase, patients continue to undergo follow-up, but do so less frequently.
Methadone maintenance is generally not recommended for individuals age <18.
Preventing opioid diversion
Prescription medications that are kept in the home are a substantial source of opioids for adolescents. In 2014, 56% of 12th graders who did not need medications for medical purposes were able to acquire them from their friends or relatives; 36% of 12th graders used their own prescriptions.21 Limiting adolescents’ access to prescription opioids is the first line of prevention. Box 2 describes interventions and strategies to limit adolescents’ access to opioids.
Box 2
Many adolescents obtain opioids for recreational use from medications that were legitimately prescribed to family or friends. Both clinicians and parents/ guardians can take steps to reduce or prevent this type of diversion
Health care facilities. Regulating the number of pills dispensed to patients is crucial. It is highly recommended to prescribe only the minimal number of opioids necessary. In most cases, 3 to 7 days’ worth of opioids at a time might be sufficient, especially after surgical procedures.
Home. Families can limit adolescents’ access to prescription opioids in the home by keeping all medications in a lock box.
Proper disposal. Various entities offer locations for patients to drop off their unused opioids and other medications for safe disposal. These include police or fire departments and retail pharmacies. The US Drug Enforcement Administration sponsors a National Prescription Drug Take Back Day; see https://www.deadiversion.usdoj.gov/drug_disposal/takeback/index.html. The FDA also offers information on where and how to dispose of unused medicines at https://www.fda.gov/consumers/consumer-updates/where-and-how-dispose-unused-medicines.
CASE CONTINUED
Ms. L is initially prescribed, clonidine, 0.1 mg every 6 hours, to address opioid withdrawal. Clonidine is then tapered and maintained at 0.1 mg twice a day for irritability and impulse control. She is also prescribed sertraline, 100 mg/d, for depression and anxiety, and trazodone, 75 mg as needed at night, to assist with sleep.
Continue to: Following inpatient hospitalization...
Following inpatient hospitalization, during 12 weeks of partial hospital treatment, Ms. L participates in individual psychotherapy sessions 5 days/week; family therapy sessions once a week; and experiential therapy along with group sessions with other peers. She undergoes medication evaluations and adjustments on a weekly basis. Ms. L is now working at a store and is pursuing a high school equivalency certificate. She manages to avoid high-risk behaviors, although she reports having occasional cravings. Ms. L is actively involved in Narcotics Anonymous and has a sponsor. She has reconciled with her mother and moved back home, so she can stay away from her former acquaintances who are still using.
Bottom Line
Adolescents with opioid use disorder can benefit from an individualized treatment plan that includes psychosocial interventions, pharmacotherapy, or a combination of the two. Treatment planning should include the adolescent and his/her family to ensure they are able to verbalize their expectations. Treatment should focus on interventions that strengthen interpersonal relationships and assist with rehabilitation. Ongoing follow-up care is necessary for maintaining abstinence.
Related Resource
- Patkar AA, Weisler RH. Opioid abuse and overdose: Keep your patients safe. Current Psychiatry. 2017;16(8):8-12,14-16.
Drug Brand Names
Buprenorphine • Subutex, Sublocade
Buprenorphine/naloxone • Suboxone
Clonidine • Clorpres
Methadone • Methadose
Naloxone • Narcan
Oxycodone • OxyContin
Sertraline • Zoloft
Tramadol • Ultram
Trazodone • Desyrel, Oleptro
Ms. L, age 17, seeks treatment because she has an ongoing struggle with multiple substances, including benzodiazepines, heroin, alcohol, cannabis, and prescription opioids.
She reports that she was 13 when she first used a prescription opioid that was not prescribed for her. She also reports engaging in unsafe sexual practices while using these substances, and has been diagnosed and treated for a sexually transmitted disease. She dropped out of school and is estranged from her family. She says that for a long time she has felt depressed and that she uses drugs to “self-medicate my emotions.” She endorses high anxiety and lack of motivation. Ms. L also reports having several criminal charges for theft, assault, and exchanging sex for drugs. She has undergone 3 admissions for detoxification, but promptly resumed using drugs, primarily heroin and oxycodone, immediately after discharge. Ms. L meets DSM-5 criteria for opioid use disorder (OUD).
Ms. L’s case illustrates a disturbing trend in the current opioid epidemic in the United States. Nearly 11.8 million individuals age ≥12 reported misuse of opioids in the last year.1 Adolescents who misuse prescription or illicit opioids are more likely to be involved with the legal system due to truancy, running away from home, physical altercations, prostitution, exchanging sex for drugs, robbery, and gang involvement. Adolescents who use opioids may also struggle with academic decline, drop out of school early, be unable to maintain a job, and have relationship difficulties, especially with family members.
In this article, I describe the scope of OUD among adolescents, including epidemiology, clinical manifestations, screening tools, and treatment approaches.
Scope of the problem
According to the most recent Monitoring the Future survey of more than 42,500 8th, 10th, and 12th grade students, 2.7% of 12th graders reported prescription opioid misuse (reported in the survey as “narcotics other than heroin”) in the past year.2 In addition, 0.4% of 12th graders reported heroin use over the same period.2 Although the prevalence of opioid use among adolescents has been declining over the past 5 years,2 it still represents a serious health crisis.
Part of the issue may relate to easier access to more potent opioids. For example, heroin available today can be >4 times purer than it was in the past. In 2002, t
Between 1997 and 2012, the annual incidence of youth (age 15 to 19) hospitalizations for prescription opioid poisoning increased >170%.5 Approximately 6% to 9% of youth involved in risky opioid use develop OUD 6 to 12 months after s
Continue to: In recent years...
In recent years, deaths from drug overdose have increased for all age groups; however, limited data is available regarding adolescent overdose deaths. According to the Centers for Disease Control and Prevention (CDC), from 2015 to 2016, drug overdose death rates for persons age 15 to 24 increased to 28%.9
How opioids work
Opioids activate specific transmembrane neurotransmitter receptors, including mu, kappa, and delta, in the CNS and peripheral nervous system (PNS). This leads to activation of G protein–mediated intracellular signal transduction. Mainly it is activation of endogenous mu opioid receptors that mediates the reward, withdrawal, and analgesic effects of opioids. These effects depend on the location of mu receptors. In the CNS, activation of mu opioid receptors may cause miosis, respiratory depression, euphoria, and analgesia.10
Different opioids vary in terms of their half-life; for most opioids, the half-life ranges from 2 to 4 hours.10 Heroin has a half-life of 30 minutes, but due to active metabolites its duration of action is 4 to 5 hours. Opioid metabolites can be detected in urine toxicology within approximately 1 to 2 days since last use.10
Chronic opioid use is associated with neurologic effects that change the function of areas of the brain that control pleasure/reward, stress, decision-making, and more. This leads to cravings, continued substance use, and dependence.11 After continued long-term use, patients report decreased euphoria, but typically they continue to use opioids to avoid withdrawal symptoms or worsening mood.
Criteria for opioid use disorder
In DSM-5, substance use disorders (SUDs)are no longer categorized as abuse or dependence.12 For opioids, the diagnosis is OUD. The Table12 outlines the DSM-5 criteria for OUD. Craving opioids is included for the first time in the OUD diagnosis. Having problems with the legal system is no longer considered a diagnostic criterion for OUD.
Continue to: A vulnerable population
A vulnerable population
As defined by Erik Erikson’s psychosocial stages of development, adolescents struggle between establishing their own identity vs role confusion.13 In an attempt to relate to peers or give in to peer pressure, some adolescents start by experimenting with nicotine, alcohol, and/or marijuana; however, some may move on to using other illicit drugs.14 Risk factors for the development of SUDs include early onset of substance use and a rapid progression through stages of substance use from experimentation to regular use, risky use, and dependence.15 In our case study, Ms. L’s substance use followed a similar pattern. Further, the comorbidity of SUDs and other psychiatric disorders may add a layer of complexity when caring for adolescents. Box 116-20 describes the relationship between comorbid psychiatric disorders and SUDs in adolescents.
Box 1
Disruptive behavior disorders are the most common coexisting psychiatric disorders in an adolescent with a substance use disorder (SUD), including opioid use disorder. These individuals typically present with aggression and other conduct disorder symptoms, and have early involvement with the legal system. Conversely, patients with conduct disorder are at high risk of early initiation of illicit substance use, including opioids. Early onset of substance use is a strong risk factor for developing an SUD.16
Mood disorders, particularly depression, can either precede or occur as a result of heavy and prolonged substance use.17 The estimated prevalence of major depressive disorder in individuals with an SUD is 24% to 50%. Among adolescents, an SUD is also a risk factor for suicidal ideation, suicide attempts, and completed suicide.18-20
Anxiety disorders, especially social phobia, and posttraumatic stress disorder are common in individuals with SUD.
Adolescents with SUD should be carefully evaluated for comorbid psychiatric disorders and treated accordingly.
Clinical manifestations
Common clinical manifestations of opioid use vary depending on when the patient is seen. An individual with OUD may appear acutely intoxicated, be in withdrawal, or show no effects. Chronic/prolonged use can lead to tolerance, such that a user needs to ingest larger amounts of the opioid to produce the same effects.
Acute intoxication can cause sedation, slurring of speech, and pinpoint pupils. Fresh injection sites may be visible on physical examination of IV users. The effects of acute intoxication usually depend on the half-life of the specific opioid and the individual’s tolerance.10 Tolerance to heroin can occur in 10 days and withdrawal can manifest in 3 to 7 hours after last use, depending on dose and purity.3 Tolerance can lead to unintentional overdose and death.
Withdrawal. Individuals experiencing withdrawal from opioids present with flu-like physical symptoms, including generalized body ache, rhinorrhea, diarrhea, goose bumps, lacrimation, and vomiting. Individuals also may experience irritability, restlessness, insomnia, anxiety, and depression during withdrawal.
Other manifestations. Excessive and chronic/prolonged opioid use can adversely impact socio-occupational functioning and cause academic decline in adolescents and youth. Personal relationships are significantly affected. Opioid users may have legal difficulties as a result of committing crimes such as theft, prostitution, or robbery in order to obtain opioids.
Continue to: Screening for OUD
Screening for OUD
Several screening tools are available to assess adolescents for SUDs, including OUD.
CRAFFT is a 6-item, clinician-administered screening tool that has been approved by American Academy of Pediatrics’ Committee on Substance Abuse for adolescents and young adults age <21.21-23 This commonly used tool can assess for alcohol, cannabis, and other drug use. A score ≥2 is considered positive for drug use, indicating that the individual would require further evaluation and assessment22,23 (Figure). There is also a self-administered CRAFFT questionnaire that can be completed by the patient.

NIDA-modified ASSIST. The American Psychiatric Association has adapted the National Institute on Drug Abuse (NIDA)-modified ASSIST. One version is designated for parents/guardians to administer to their children (age 6 to 17), and one is designated for adolescents (age 11 to 17) to self-administer.24,25 Each screening tool has 2 levels: Level 1 screens for substance use and other mental health symptoms, and Level 2 is more specific for substance use alone.
Drug Use Screening Inventory (DUSI) is a self-report questionnaire that has 149 items that assess the use of numerous drugs. It is designed to quantify the severity of consequences associated with drug and alcohol use.26,27
Problem-Oriented Screening Instrument for Teenagers (PO
Continue to: Personal Experience Screening Questionnaire (PESQ)...
Personal Experience Screening Questionnaire (PESQ) is a brief, 40-item, cost-effective, self-report questionnaire that can help identify adolescents (age 12 to 18) who should be referred for further evaluation.30
Addressing treatment expectations
For an adolescent with OUD, treatment should begin in the least restrictive environment that is perceived as safe for the patient. An adolescent’s readiness and motivation to achieve and maintain abstinence are crucial. Treatment planning should include the adolescent as well as his/her family to ensure they are able to verbalize their expectations. Start with a definitive treatment plan that addresses an individual’s needs. The plan should provide structure and an understanding of treatment expectations. The treatment team should clarify the realistic plan and goals based on empirical and clinical evidence. Treatment goals should include interventions to strengthen interpersonal relationships and assist with rehabilitation, such as establishing academic and/or vocational goals. Addressing readiness and working on a patient’s motivation is extremely important for most of these interventions.
In order for any intervention to be successful, clinicians need to establish and foster rapport with the adolescent. By law, substance use or behaviors related to substance use are not allowed to be shared outside the patient-clinician relationship, unless the adolescent gives consent or there are concerns that such behaviors might put the patient or others at risk. It is important to prime the adolescent and help them understand that any information pertaining to their safety or the safety of others may need to be shared outside the patient-clinician relationship.
Choosing an intervention
Less than 50% of a nationally representative sample of 345 addiction treatment programs serving adolescents and adults offer medications for treating OUD.31 Even in programs that offer pharmacotherapy, medications are significantly underutilized. Fewer than 30% of patients in addiction treatment programs receive medication, compared with 74% of patients receiving treatment for other mental health disorders.31 A
Psychotherapy may be used to treat OUD in adolescents. Several family therapies have been studied and are considered as critical psychotherapeutic interventions for treating SUDs, including structural family treatment and functional family therapy approaches.34 An integrated behavioral and family therapy model is also recommended for adolescent patients with SUDs. Cognitive distortions and use of self-deprecatory statements are common among adolescents.35 Therefore, using approaches of cognitive-behavioral therapy (CBT), or CBT plus motivational enhancement therapy, also might be effective for this population.36 The adolescent community reinforcement approach (A-CRA) is a behavioral treatment designed to help adolescents and their families learn how to lead a healthy and happy life without the use of drugs or alcohol by increasing access to social, familial, and educational/vocational reinforcers. Support groups and peer and family support should be encouraged as adjuncts to other interventions. In some areas, sober housing options for adolescents are also available.
Continue to: Harm-reduction strategies
Harm-reduction strategies. Although the primary goal of treatment for adolescents with OUD is to achieve and maintain abstinence from opioid use, implicit and explicit goals can be set. Short-term implicit goals may include harm-reduction strategies that emphasize decreasing the duration, frequency, and amount of substance use and limiting the chances of adverse effects, while the long-term explicit goal should be abstinence from opioid use.
Naloxone nasal spray is used as a harm-reduction strategy. It is an FDA-approved formulation that can reverse the effects of unintentional opioid overdoses and potentially prevent death from respiratory depression.37 Other harm-reduction strategies include needle exchange programs, which provide sterile needles to individuals who inject drugs in an effort to prevent or reduce the transmission of human immunodeficiency virus and other bloodborne viruses that can be spread via shared injection equipment. Fentanyl testing strips allow opioid users to test for the presence fentanyl and fentanyl analogs in the unregulated “street” opioid supply.
Pharmacologic interventions. Because there is limited empirical evidence on the efficacy of medication-assisted treatment (MAT) for adolescents with OUD, clinicians need to rely on evidence from research and experience with adults. Unfortunately, MAT is offered to adolescents considerably less often than it is to adults. Feder et al38 reported that only 2.4% of adolescents received MAT for heroin use and only 0.4% of adolescents received MAT for prescription opioid use, compared with 26.3% and 12% of adults, respectively.
Detoxification. Medications available for detoxification from opioids include opiates (such as methadone or buprenorphine) and clonidine (a central sympathomimetic). If the patient has used heroin for a short period (<1 year) and has no history of detoxification, consider a detoxification strategy with a longer-term taper (90 to 180 days) to allow for stabilization.
Maintenance treatment. Consider maintenance treatment for adolescents with a history of long-term opioid use and at least 2 prior short-term detoxification attempts or nonpharmacotherapy-based treatment within 12 months. Be sure to receive consent from a legal guardian and the patient. Maintenance treatment is usually recommended to continue for 1 to 6 years. Maintenance programs with longer durations have shown higher rates of abstinence, improved engagement, and retention in treatment.39
Continue to: According to guidelines from...
According to guidelines from the American Society of Addiction Medicine (ASAM), adolescents age >16 should be offered MAT; the first-line treatment is buprenorphine.40 To avoid risks of abuse and diversion, a combination of buprenorphine/naloxone may be administered.
Maintenance with buprenorphine
In order to prescribe and dispense buprenorphine, clinicians need to obtain a waiver from the Substance Abuse and Mental Health Services Administration. Before initiating buprenorphine, consider the type of opioid the individual used (short- or long-acting), the severity of the OUD, and the last reported use. The 3 phases of buprenorphine treatment are41:
- Induction phase. Buprenorphine can be initiated at 2 to 4 mg/d. Some patients may require up to 8 mg/d on the first day, which can be administered in divided doses.42 Evaluate and monitor patients carefully during the first few hours after the first dose. Patients should be in early withdrawal; otherwise, the buprenorphine might precipitate withdrawal. The induction phase can be completed in 2 to 4 days by titrating the dose so that the signs and symptoms of opioid withdrawal are minimal, and the patient is able to continue treatment. It may be helpful to have the patient’s legal guardian nearby in case the patient does not tolerate the medication or experiences withdrawal. The initial target dose for buprenorphine is approximately 12 to 16 mg/d.
- Stabilization phase. Patients no longer experience withdrawal symptoms and no longer have cravings. This phase can last 6 to 8 weeks. During this phase, patients should be seen weekly and doses should be adjusted if necessary. As a partial mu agonist, buprenorphine does not activate mu receptors fully and reaches a ceiling effect. Hence, doses >24 mg/d have limited added agonist properties.
- Maintenance phase. Because discontinuation of buprenorphine is associated with high relapse rates, patients may need to be maintained long-term on their stabilization dose, and for some patients, the length of time could be indefinite.39 During this phase, patients continue to undergo follow-up, but do so less frequently.
Methadone maintenance is generally not recommended for individuals age <18.
Preventing opioid diversion
Prescription medications that are kept in the home are a substantial source of opioids for adolescents. In 2014, 56% of 12th graders who did not need medications for medical purposes were able to acquire them from their friends or relatives; 36% of 12th graders used their own prescriptions.21 Limiting adolescents’ access to prescription opioids is the first line of prevention. Box 2 describes interventions and strategies to limit adolescents’ access to opioids.
Box 2
Many adolescents obtain opioids for recreational use from medications that were legitimately prescribed to family or friends. Both clinicians and parents/ guardians can take steps to reduce or prevent this type of diversion
Health care facilities. Regulating the number of pills dispensed to patients is crucial. It is highly recommended to prescribe only the minimal number of opioids necessary. In most cases, 3 to 7 days’ worth of opioids at a time might be sufficient, especially after surgical procedures.
Home. Families can limit adolescents’ access to prescription opioids in the home by keeping all medications in a lock box.
Proper disposal. Various entities offer locations for patients to drop off their unused opioids and other medications for safe disposal. These include police or fire departments and retail pharmacies. The US Drug Enforcement Administration sponsors a National Prescription Drug Take Back Day; see https://www.deadiversion.usdoj.gov/drug_disposal/takeback/index.html. The FDA also offers information on where and how to dispose of unused medicines at https://www.fda.gov/consumers/consumer-updates/where-and-how-dispose-unused-medicines.
CASE CONTINUED
Ms. L is initially prescribed, clonidine, 0.1 mg every 6 hours, to address opioid withdrawal. Clonidine is then tapered and maintained at 0.1 mg twice a day for irritability and impulse control. She is also prescribed sertraline, 100 mg/d, for depression and anxiety, and trazodone, 75 mg as needed at night, to assist with sleep.
Continue to: Following inpatient hospitalization...
Following inpatient hospitalization, during 12 weeks of partial hospital treatment, Ms. L participates in individual psychotherapy sessions 5 days/week; family therapy sessions once a week; and experiential therapy along with group sessions with other peers. She undergoes medication evaluations and adjustments on a weekly basis. Ms. L is now working at a store and is pursuing a high school equivalency certificate. She manages to avoid high-risk behaviors, although she reports having occasional cravings. Ms. L is actively involved in Narcotics Anonymous and has a sponsor. She has reconciled with her mother and moved back home, so she can stay away from her former acquaintances who are still using.
Bottom Line
Adolescents with opioid use disorder can benefit from an individualized treatment plan that includes psychosocial interventions, pharmacotherapy, or a combination of the two. Treatment planning should include the adolescent and his/her family to ensure they are able to verbalize their expectations. Treatment should focus on interventions that strengthen interpersonal relationships and assist with rehabilitation. Ongoing follow-up care is necessary for maintaining abstinence.
Related Resource
- Patkar AA, Weisler RH. Opioid abuse and overdose: Keep your patients safe. Current Psychiatry. 2017;16(8):8-12,14-16.
Drug Brand Names
Buprenorphine • Subutex, Sublocade
Buprenorphine/naloxone • Suboxone
Clonidine • Clorpres
Methadone • Methadose
Naloxone • Narcan
Oxycodone • OxyContin
Sertraline • Zoloft
Tramadol • Ultram
Trazodone • Desyrel, Oleptro
1. Davis JP, Prindle JJ, Eddie D, et al. Addressing the opioid epidemic with behavioral interventions for adolescents and young adults: a quasi-experimental design. J Consult Clin Psychol. 2019;87(10):941-951.
2. National Institute on Drug Abuse; National Institutes of Health; U.S. Department of Health and Human Services. Monitoring the Future Survey: High School and Youth Trends. https://www.drugabuse.gov/publications/drugfacts/monitoring-future-survey-high-school-youth-trends. Updated December 2019. Accessed January 13, 2020.
3. Hopfer CJ, Khuri E, Crowley TJ. Treating adolescent heroin use. J Am Acad Child Adolesc Psychiatry. 2003;42(5):609-611.
4. US Department of Justice, Drug Enforcement Agency, Diversion Control Division. https://www.deadiversion.usdoj.gov/. Accessed January 21, 2020.
5. Gaither JR, Leventhal JM, Ryan SA, et al. National trends in hospitalizations for opioid poisonings among children and adolescents, 1997-2012. JAMA Pediatr. 2016;170(12):1195-1201.
6. Parker MA, Anthony JC. Epidemiological evidence on extra-medical use of prescription pain relievers: transitions from newly incident use to dependence among 12-21 year olds in United States using meta-analysis, 2002-13. Peer J. 2015;3:e1340. doi: 10.7717/peerj.1340. eCollection 2015.
7. Subramaniam GA, Fishman MJ, Woody G. Treatment of opioid-dependent adolescents and young adults with buprenorphine. Curr Psychiatry Rep. 2009;11(5):360-363.
8. Borodovsky JT, Levy S, Fishman M. Buprenorphine treatment for adolescents and young adults with opioid use disorders: a narrative review. J Addict Med. 2018;12(3):170-183.
9. Centers for Disease Control and Prevention: National Center for Health Statistics. Drug overdose deaths in the United States, 1999-2016. https://www.cdc.gov/nchs/products/databriefs/db294.htm. Published December 2017. Accessed January 15, 2020.
10. Strain E. Opioid use disorder: epidemiology, pharmacology, clinical manifestation, course, screening, assessment, diagnosis. https://www.uptodate.com/contents/opioid-use-disorder-epidemiology-pharmacology-clinical-manifestations-course-screening-assessment-and-diagnosis. Updated August 15, 2019. Accessed January 21, 2020.
11. American Academy of Pediatrics Committee on Substance Use and Prevention. Policy statement: medication-assisted treatment of adolescents with opioid use disorder. Pediatrics. 2016;138(3):e20161893. doi: https://doi.org/10.1542/peds.2016-1893.
12. Diagnostic and Statistical Manual of Mental Disorders, 5th ed. Washington, DC: American Psychiatric Association; 2013:514.
13. Sadock BJ, Sadock VA. Chapter 6: Theories of personality and psychopathology. In: Sadock BJ, Sadock VA, eds. Kaplan and Sadock’s synopsis of psychiatry: behavioral sciences/clinical. 10th ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2007:209.
14. Kandel DB. Stages and pathways of drug involvement: examining the gateway hypothesis. Cambridge, United Kingdom: Cambridge University Press; 2002.
15. Robins LN, McEvoy L. Conduct problems as predictors of substance abuse. In: Robins LN, Rutter M, eds. Straight and devious pathways from childhood to adulthood. Cambridge, United Kingdom: Cambridge University Press; 1990;182-204.
16. Hopfer C, Salomonsen-Sautel S, Mikulich-Gilbertson S, et al. Conduct disorder and initiation of substance use: a prospective longitudinal study. J Am Acad Child Adolesc Psychiatry. 2013;52(5):511-518.e4.
17. Armstrong TD, Costello EJ. Community studies on adolescent substance use, abuse, or dependence and psychiatric comorbidity. J Consult Clin Psychol. 2002;70(6):1224-1239.
18. Crumley FE. Substance abuse and adolescent suicidal behavior. JAMA. 1990;263(22):3051-3056.
19. Lewinsohn PM, Rohde P, Seeley JR. Adolescent suicidal ideation and attempts: prevalence, risk factors, and clinical implications. Clinical Psychology: Science and Practice. 1996;3(1):25-46.
20. Kendler KS, Bulik CM, Silberg J, et al. Childhood sexual abuse and adult psychiatric and substance use disorder in women: an epidemiological and cotwin control analysis. Arch Gen Psychiatry. 2000;57(10):953-959.
21. Yule AM, Wilens TE, Rausch PK. The opioid epidemic: what a child psychiatrist is to do? J Am Acad Child Adolesc Psychiatry. 2017;56(7);541-543.
22. CRAFFT. https://crafft.org. Accessed January 21, 2020.
23. Knight JR, Sherritt L, Harris SK, et al. Validity of brief alcohol screening tests among adolescents: a comparison of the AUDIT, POSIT, CAGE, and CRAFFT. Alcohol Clin Exp Res. 2003;27(1):67-73.
24. American Psychiatric Association. Online assessment measures. https://www.psychiatry.org/psychiatrists/practice/dsm/educational-resources/assessment-measures. Accessed January 15, 2020.
25. National Institute of Drug Abuse. American Psychiatric Association adapted NIDA modified ASSIST tools. https://www.drugabuse.gov/nidamed-medical-health-professionals/tool-resources-your-practice/screening-assessment-drug-testing-resources/american-psychiatric-association-adapted-nida. Updated November 15, 2015. Accessed January 21, 2020.
26. Canada’s Mental Health & Addiction Network. Drug Use Screening Inventory (DUSI). https://www.porticonetwork.ca/web/knowledgex-archive/amh-specialists/screening-for-cd-in-youth/screening-both-mh-sud/dusi. Published 2009. Accessed January 21, 2020.
27. Tarter RE. Evaluation and treatment of adolescent substance abuse: a decision tree method. Am J Drug Alcohol Abuse. 1990;16(1-2):1-46.
28. Klitzner M, Gruenwald PJ, Taff GA, et al. The adolescent assessment referral system-final report. National Institute on Drug Abuse; Rockville, MD: 1993. NIDA Contract No. 271-89-8252.
29. Slesnick N, Tonigan JS. Assessment of alcohol and other drug use by runaway youths: a test-retest study of the Form 90. Alcohol Treat Q. 2004;22(2):21-34.
30. Winters KC, Kaminer Y. Screening and assessing adolescent substance use disorders in clinical populations. J Am Acad Child Adolesc Psychiatry. 2008;47(7):740-744.
31. Knudsen HK, Abraham AJ, Roman PM. Adoption and implementation of medications in addiction treatment programs. J Addict Med. 2011;5(1):21-27.
32. Deas D, Thomas SE. An overview of controlled study of adolescent substance abuse treatment. Am J Addiction. 2001;10(2):178-189.
33. William RJ, Chang, SY. A comprehensive and comparative review of adolescent substance abuse treatment outcome. Clinical Psychology: Science and Practice. 2000;7(2):138-166.
34. Bukstein OG, Work Group on Quality Issues. Practice parameters for the assessment and treatment of children and adolescents with substance use disorders. J Am Acad Child Adolesc Psychiatry. 2005;44(6):609-621.
35. Van Hasselt VB, Null JA, Kempton T, et al. Social skills and depression in adolescent substance abusers. Addict Behav. 1993;18(1):9-18.
36. Dennis M, Godley SH, Diamond G, et al. The Cannabis Youth Treatment (CYT) study: main findings from two randomized trials. J Subst Abuse Treat. 2004;27(3):197-213.
37. US Food and Drug Administration. Information about naloxone. https://www.fda.gov/drugs/postmarket-drug-safety-information-patients-and-providers/information-about-naloxone. Updated December 19, 2019. Accessed January 21, 2020.
38. Feder KA, Krawcyzk N, Saloner, B. Medication-assisted treatment for adolescents in specialty treatment for opioid use disorder. J Adolesc Health. 2018;60(6):747-750.
39. Woody GE, Poole SA, Subramaniam G, et al. Extended vs short-term buprenorphine-naloxone for treatment of opioid-addicted youth: a randomized trial. JAMA. 2008;300(17):2003-2011.
40. US Department of Health and Human Services. Substance Abuse and Mental Health Ser-vices Administration. Medication-assisted treatment for opioid addiction in opioid treatment programs: a treatment improvement protocol TIP 43. https://www.asam.org/docs/advocacy/samhsa_tip43_matforopioidaddiction.pdf?sfvrsn=0. Published 2005. Accessed January 15, 2020.
41. US Department of Health and Human Services. Substance Abuse and Mental Health Services Administration. Medication-assisted treatment (MAT). https://www.samhsa.gov/medication-assisted-treatment. Updated September 9, 2019. Accessed January 21, 2020.
42. Johnson RE, Strain EC, Amass L. Buprenorphine: how to use it right. Drug Alcohol Depend. 2003;70(suppl 2):S59-S77.
1. Davis JP, Prindle JJ, Eddie D, et al. Addressing the opioid epidemic with behavioral interventions for adolescents and young adults: a quasi-experimental design. J Consult Clin Psychol. 2019;87(10):941-951.
2. National Institute on Drug Abuse; National Institutes of Health; U.S. Department of Health and Human Services. Monitoring the Future Survey: High School and Youth Trends. https://www.drugabuse.gov/publications/drugfacts/monitoring-future-survey-high-school-youth-trends. Updated December 2019. Accessed January 13, 2020.
3. Hopfer CJ, Khuri E, Crowley TJ. Treating adolescent heroin use. J Am Acad Child Adolesc Psychiatry. 2003;42(5):609-611.
4. US Department of Justice, Drug Enforcement Agency, Diversion Control Division. https://www.deadiversion.usdoj.gov/. Accessed January 21, 2020.
5. Gaither JR, Leventhal JM, Ryan SA, et al. National trends in hospitalizations for opioid poisonings among children and adolescents, 1997-2012. JAMA Pediatr. 2016;170(12):1195-1201.
6. Parker MA, Anthony JC. Epidemiological evidence on extra-medical use of prescription pain relievers: transitions from newly incident use to dependence among 12-21 year olds in United States using meta-analysis, 2002-13. Peer J. 2015;3:e1340. doi: 10.7717/peerj.1340. eCollection 2015.
7. Subramaniam GA, Fishman MJ, Woody G. Treatment of opioid-dependent adolescents and young adults with buprenorphine. Curr Psychiatry Rep. 2009;11(5):360-363.
8. Borodovsky JT, Levy S, Fishman M. Buprenorphine treatment for adolescents and young adults with opioid use disorders: a narrative review. J Addict Med. 2018;12(3):170-183.
9. Centers for Disease Control and Prevention: National Center for Health Statistics. Drug overdose deaths in the United States, 1999-2016. https://www.cdc.gov/nchs/products/databriefs/db294.htm. Published December 2017. Accessed January 15, 2020.
10. Strain E. Opioid use disorder: epidemiology, pharmacology, clinical manifestation, course, screening, assessment, diagnosis. https://www.uptodate.com/contents/opioid-use-disorder-epidemiology-pharmacology-clinical-manifestations-course-screening-assessment-and-diagnosis. Updated August 15, 2019. Accessed January 21, 2020.
11. American Academy of Pediatrics Committee on Substance Use and Prevention. Policy statement: medication-assisted treatment of adolescents with opioid use disorder. Pediatrics. 2016;138(3):e20161893. doi: https://doi.org/10.1542/peds.2016-1893.
12. Diagnostic and Statistical Manual of Mental Disorders, 5th ed. Washington, DC: American Psychiatric Association; 2013:514.
13. Sadock BJ, Sadock VA. Chapter 6: Theories of personality and psychopathology. In: Sadock BJ, Sadock VA, eds. Kaplan and Sadock’s synopsis of psychiatry: behavioral sciences/clinical. 10th ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2007:209.
14. Kandel DB. Stages and pathways of drug involvement: examining the gateway hypothesis. Cambridge, United Kingdom: Cambridge University Press; 2002.
15. Robins LN, McEvoy L. Conduct problems as predictors of substance abuse. In: Robins LN, Rutter M, eds. Straight and devious pathways from childhood to adulthood. Cambridge, United Kingdom: Cambridge University Press; 1990;182-204.
16. Hopfer C, Salomonsen-Sautel S, Mikulich-Gilbertson S, et al. Conduct disorder and initiation of substance use: a prospective longitudinal study. J Am Acad Child Adolesc Psychiatry. 2013;52(5):511-518.e4.
17. Armstrong TD, Costello EJ. Community studies on adolescent substance use, abuse, or dependence and psychiatric comorbidity. J Consult Clin Psychol. 2002;70(6):1224-1239.
18. Crumley FE. Substance abuse and adolescent suicidal behavior. JAMA. 1990;263(22):3051-3056.
19. Lewinsohn PM, Rohde P, Seeley JR. Adolescent suicidal ideation and attempts: prevalence, risk factors, and clinical implications. Clinical Psychology: Science and Practice. 1996;3(1):25-46.
20. Kendler KS, Bulik CM, Silberg J, et al. Childhood sexual abuse and adult psychiatric and substance use disorder in women: an epidemiological and cotwin control analysis. Arch Gen Psychiatry. 2000;57(10):953-959.
21. Yule AM, Wilens TE, Rausch PK. The opioid epidemic: what a child psychiatrist is to do? J Am Acad Child Adolesc Psychiatry. 2017;56(7);541-543.
22. CRAFFT. https://crafft.org. Accessed January 21, 2020.
23. Knight JR, Sherritt L, Harris SK, et al. Validity of brief alcohol screening tests among adolescents: a comparison of the AUDIT, POSIT, CAGE, and CRAFFT. Alcohol Clin Exp Res. 2003;27(1):67-73.
24. American Psychiatric Association. Online assessment measures. https://www.psychiatry.org/psychiatrists/practice/dsm/educational-resources/assessment-measures. Accessed January 15, 2020.
25. National Institute of Drug Abuse. American Psychiatric Association adapted NIDA modified ASSIST tools. https://www.drugabuse.gov/nidamed-medical-health-professionals/tool-resources-your-practice/screening-assessment-drug-testing-resources/american-psychiatric-association-adapted-nida. Updated November 15, 2015. Accessed January 21, 2020.
26. Canada’s Mental Health & Addiction Network. Drug Use Screening Inventory (DUSI). https://www.porticonetwork.ca/web/knowledgex-archive/amh-specialists/screening-for-cd-in-youth/screening-both-mh-sud/dusi. Published 2009. Accessed January 21, 2020.
27. Tarter RE. Evaluation and treatment of adolescent substance abuse: a decision tree method. Am J Drug Alcohol Abuse. 1990;16(1-2):1-46.
28. Klitzner M, Gruenwald PJ, Taff GA, et al. The adolescent assessment referral system-final report. National Institute on Drug Abuse; Rockville, MD: 1993. NIDA Contract No. 271-89-8252.
29. Slesnick N, Tonigan JS. Assessment of alcohol and other drug use by runaway youths: a test-retest study of the Form 90. Alcohol Treat Q. 2004;22(2):21-34.
30. Winters KC, Kaminer Y. Screening and assessing adolescent substance use disorders in clinical populations. J Am Acad Child Adolesc Psychiatry. 2008;47(7):740-744.
31. Knudsen HK, Abraham AJ, Roman PM. Adoption and implementation of medications in addiction treatment programs. J Addict Med. 2011;5(1):21-27.
32. Deas D, Thomas SE. An overview of controlled study of adolescent substance abuse treatment. Am J Addiction. 2001;10(2):178-189.
33. William RJ, Chang, SY. A comprehensive and comparative review of adolescent substance abuse treatment outcome. Clinical Psychology: Science and Practice. 2000;7(2):138-166.
34. Bukstein OG, Work Group on Quality Issues. Practice parameters for the assessment and treatment of children and adolescents with substance use disorders. J Am Acad Child Adolesc Psychiatry. 2005;44(6):609-621.
35. Van Hasselt VB, Null JA, Kempton T, et al. Social skills and depression in adolescent substance abusers. Addict Behav. 1993;18(1):9-18.
36. Dennis M, Godley SH, Diamond G, et al. The Cannabis Youth Treatment (CYT) study: main findings from two randomized trials. J Subst Abuse Treat. 2004;27(3):197-213.
37. US Food and Drug Administration. Information about naloxone. https://www.fda.gov/drugs/postmarket-drug-safety-information-patients-and-providers/information-about-naloxone. Updated December 19, 2019. Accessed January 21, 2020.
38. Feder KA, Krawcyzk N, Saloner, B. Medication-assisted treatment for adolescents in specialty treatment for opioid use disorder. J Adolesc Health. 2018;60(6):747-750.
39. Woody GE, Poole SA, Subramaniam G, et al. Extended vs short-term buprenorphine-naloxone for treatment of opioid-addicted youth: a randomized trial. JAMA. 2008;300(17):2003-2011.
40. US Department of Health and Human Services. Substance Abuse and Mental Health Ser-vices Administration. Medication-assisted treatment for opioid addiction in opioid treatment programs: a treatment improvement protocol TIP 43. https://www.asam.org/docs/advocacy/samhsa_tip43_matforopioidaddiction.pdf?sfvrsn=0. Published 2005. Accessed January 15, 2020.
41. US Department of Health and Human Services. Substance Abuse and Mental Health Services Administration. Medication-assisted treatment (MAT). https://www.samhsa.gov/medication-assisted-treatment. Updated September 9, 2019. Accessed January 21, 2020.
42. Johnson RE, Strain EC, Amass L. Buprenorphine: how to use it right. Drug Alcohol Depend. 2003;70(suppl 2):S59-S77.
Improving Nephropathy Screening in Appalachian Patients With Diabetes Using Practice-Wide Outreach
From West Virginia University, Morgantown, WV.
Abstract
Objective: To describe the strategies a family medicine clinic in Appalachia utilized to increase nephropathy screening rates as well as to explore the factors predictive of nephropathy screening in patients with diabetes.
Design: This quality improvement project targeted the points in the care process when patients are lost to follow-up for nephropathy screening.
Setting and participants: Patients with diabetes cared for by a primary care provider (PCP) at an academic family medicine practice in Appalachia from January 2018 to November 2018.
Interventions: Bulk orders for albumin-to-creatinine (ACR) testing and urine collection during clinic visit, enhanced patient communication through bulk communication reminders and individual patient outreach, and education of clinic providers.
Measurements: Demographic data and monthly nephropathy screening rates.
Results: The nephropathy screening rate increased by 6.2% during the project. Older patients living closer to the clinic who visited their PCP 3 or more times per year were the most likely to be screened.
Conclusion: Combining team-based interventions with quality control monitoring can significantly improve compliance with recommended nephropathy treatment and screening in rural patients with diabetes at a family medicine clinic.
Keywords: rural; kidney disease; albumin-to-creatinine ratio; electronic health record.
According to the Centers for Disease Control and Prevention (CDC), an estimated 30.3 million people in the United States—about 9.4% of the population—have been diagnosed with diabetes.1 Diabetes is the seventh leading cause of death in the United States, and it contributes to other leading causes of death: heart disease and stroke.1 Diabetes also is related to high morbidity risk and is a leading cause of chronic kidney disease.1 The total cost of diagnosed diabetes was estimated at $327 billion in direct medical costs and reduced productivity.2
Residents of Appalachia bear a disproportionate burden of diabetes and other related negative health outcomes; these outcomes are influenced by a number of factors, including socioeconomic status, poverty, rurality, and health care access. Rates of chronic disease, such as diabetes, are most pronounced in Appalachia’s most economically distressed counties.3-5 In 2011, the CDC labeled a 644-county area the “diabetes belt,” which included most of Appalachia.6 As a result of this elevated prevalence of diabetes in Appalachia as compared to the rest of the country, complications directly associated with diabetes are more commonly observed in Appalachian residents. One of the most damaging complications is diabetic nephropathy.
Diabetic nephropathy results from damage to the microvasculature of the kidney due to inadequately controlled blood glucose. This, in turn, leads to decreased renal function, eventually leading to clinically significant renal disease. The long-term complications associated with nephropathy can include many comorbid conditions, the most serious of which are progression to end-stage renal disease, dialysis requirement, and early mortality. Diabetic nephropathy affects approximately 40% of patients with type 1 and type 2 diabetes.7,8
One way to prevent complications of diabetic nephropathy, in addition to good glycemic control in patients with diabetes, is early and regular screening. Currently, the American Diabetes Association (ADA) recommends yearly screening for diabetic nephropathy in the form of a urine albumin-to-creatinine ratio (ACR) for patients 18 to 75 years of age.2 This screening to detect diabetic nephropathy is recognized as a marker of quality care by many public and private insurance agencies and medical specialty associations, such as the Centers for Medicare and Medicaid Services.
Many patients with diabetes are cared for by primary care providers (PCP), and these PCP appointments provide an opportune time to screen and appropriately treat nephropathy. Screening opportunities are often missed, however, due to time constraints and competing health priorities. There are also a number of other factors specific to the Appalachian region that reduce the likelihood of screening for diabetic nephropathy, such as a lack of health insurance, the need to travel long distances to see a PCP, work and household responsibilities, low levels of education and health literacy, and a mistrust of outsiders regarding personal matters, including health.9-11 While nephropathy can have a detrimental impact on patients across populations, it is of particular concern for a state located in the heart of Appalachia, such as West Virginia.
Given the disproportionate burden of diabetes in this region and the potentially severe consequences of undetected nephropathy, clinicians from an academic family medicine clinic in West Virginia undertook a quality improvement project to increase the rate of nephropathy screening and treatment among patients with diabetes. This article describes the intervention strategies the team utilized to increase nephropathy screening and treatment in patients 18 to 75 years of age who met quality measures for nephropathy screening or treatment in the previous 12 months and explores the factors most predictive of nephropathy screening in Appalachian patients in this age group. It also reports the challenges and opportunities encountered and offers suggestions for other providers and clinics attempting to increase their nephropathy screening rates.
Methods
Setting and Study Population
The study population included patients ages 18 to 75 years under the care of providers in an academic family medicine practice in West Virginia who had been diagnosed with diabetes mellitus. The study focused on those patients overdue for diabetic nephropathy screening (ie, had not been screened in previous 12 months). The project began in January 2018 with a screening rate of 83.8%. The goal of this project was to increase this compliance metric by at least 5%. The project protocol was submitted to the West Virginia University Institutional Review Board, and, because it is a quality improvement project, permission was given to proceed without a board review.
Interventions
The team identified and implemented several interventions intended to reduce screening barriers and increase the screening rate.
Bulk orders for ACR and urine collection during clinic visits. Prior to initiation of this project, it was left to individual clinic providers to order nephropathy screening for patients with diabetes during a clinic visit; after receiving the order for “random urine microalbumin/creatinine ratio,” patients then had to travel to a lab to provide a urine sample. For this project and moving forward, the team changed to the procedure of initiating bulk ACR orders and collecting urine samples during clinic visits from all patients ages 18 to 75 years who have diabetes.
Bulk communication reminders. Since many patients with diabetes may not have realized they were overdue for nephropathy screening, the team began sending out bulk communication reminders through either the institution’s electronic health record (EHR; MyChart) or postal service–delivered physical letters (according to patient communication preferences) to remind patients that they were due for screening and to encourage them to schedule an appointment or keep a previously scheduled appointment with their PCP.
Individual patient outreach. A team of pharmacy students led by a licensed pharmacist in the family medicine clinic contacted patients overdue for screening even after bulk communication reminders went out. The students telephoned patients 2 to 3 months following the bulk communication. The students obtained an updated list of patients with diabetes ages 18 to 75 years from an EHR quality report. They began by prescreening the patients on the overdue list for potential candidacy for an angiotensin-converting enzyme (ACE) inhibitor or an angiotensin II receptor blocker (ARB). Screening for candidacy included evaluation of recent blood pressure readings, electrolytes (ie, basic metabolic panel), and ACR. If the students determined a patient was a candidate, they presented the patient to the preceptor for verification and then reached out to the provider with a recommendation. If the provider agreed, the student contacted the patient by telephone for medication counseling and education. The remaining patients determined not to be candidates for ACE inhibitors or ARBs were contacted by the pharmacy students by telephone to remind them that laboratory work was pending. Up to 3 phone call attempts were made before patients were determined to be unreachable. Students left voice mails with generic reminders if a patient could not be reached. If a patient answered, the student provided a reminder but also reviewed indications for lab work, the reason why the provider wished for follow-up, and updated lab hours. Students also followed up with the results of the work-up, as appropriate. During this outreach process, the student team encountered a number of patients who had moved or changed to a PCP outside of the family medicine clinic. In these cases, the EHR was updated and those patients were removed from the list of patients altogether.
Education of clinic providers. Clinic providers were educated during faculty and resident meetings and didactic learning sessions on identifying patients within the EHR who are due for nephropathy screening. They also received instruction on how to update the EHR to reflect completed screenings.
Data Analysis
All analyses in this study were conducted using SAS (version 9.4, 2013, SAS Institute Inc., Cary, NC). Descriptive analyses were conducted to summarize basic patient demographic information. To compare patients screened within the previous 12 months to those patients overdue for screening, 2-sample t-tests were used to examine differences in patients’ age, HbA1c, ACR, and creatinine level and the distance (in miles) between the patient’s home and the clinic. Chi-square analyses were used to examine the relationship between whether a patient was recently screened for nephropathy and the patient’s insurance, number of patient visits in the previous 12 months, and provider level. Logistic regression analyses were conducted to control for covariates and to explore which factors were most predictive of nephropathy screening. All tests were 2-tailed, and P values less than 0.05 were considered statistically significant.
Results
Patient Characteristics
There were 1676 family medicine clinic patients with diabetes between 18 and 75 years of age (Table 1 and Table 2). Of the total sample, 1489 (88.8%) had completed screening for nephropathy in the 12 months prior to evaluation, and 67.5%, 23.7%, and 8.8% of patients had private insurance, Medicare, and Medicaid, respectively. 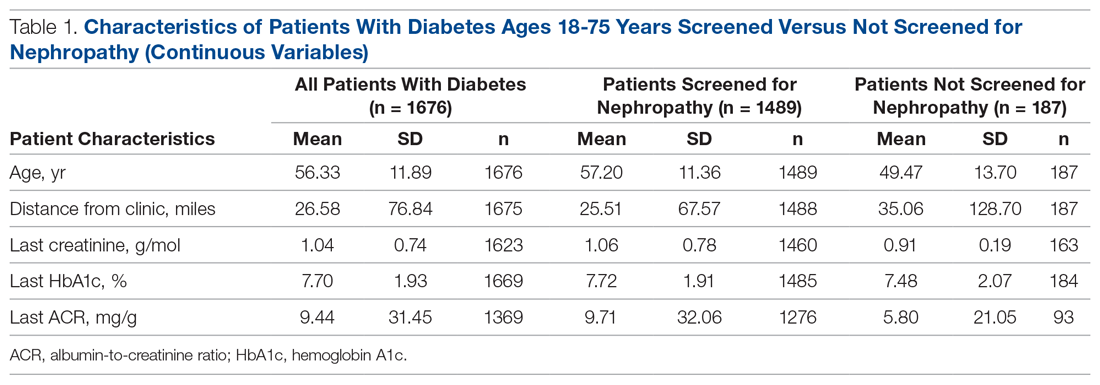
The mean (SD) age of the patients was 56.3 (11.9) years. The mean distance between the patient’s home and the clinic was 26.6 (76.8) miles. The mean number of visits was 3.6 (2.9) per year, and 43.0% of the patientvisited the clinic more than 3 times in a year. The mean values for HbA1c (%), creatinine (g/mol), and ACR (mg/g) were 7.7 (1.9), 1.0 (0.7), and 9.4 (31.4), respectively.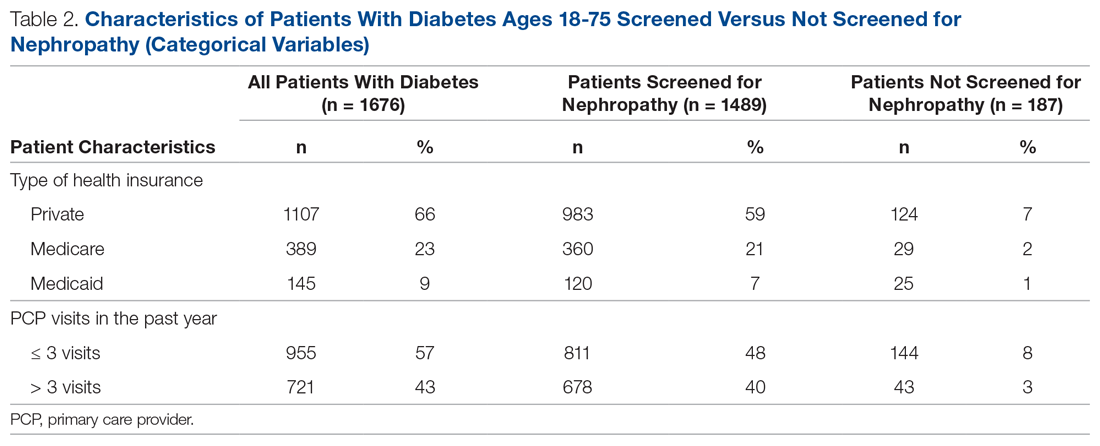
Screening of Patients for Nephropathy
Patients with Medicare and private insurance were more likely to have completed the nephropathy screening than those with Medicaid (92.5% versus 88.8% versus 82.8%, P = 0.004; Table 3 and Table 4). 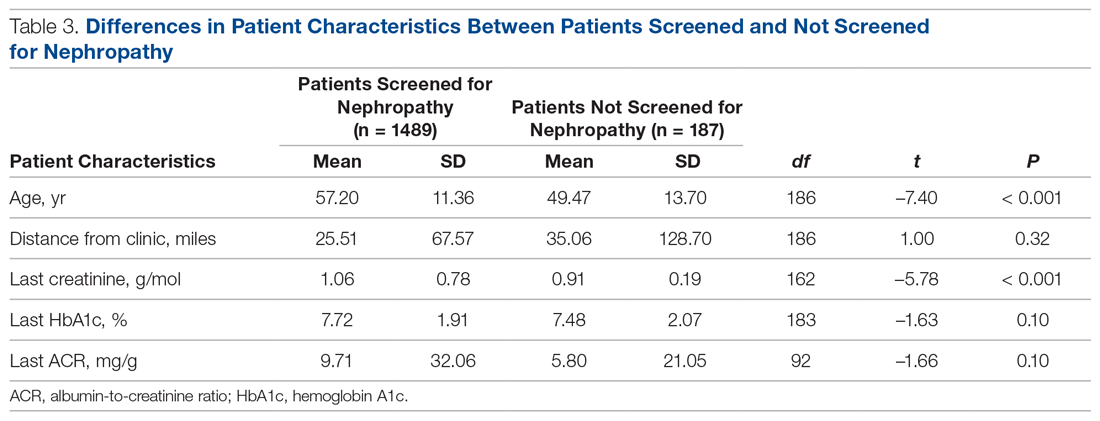
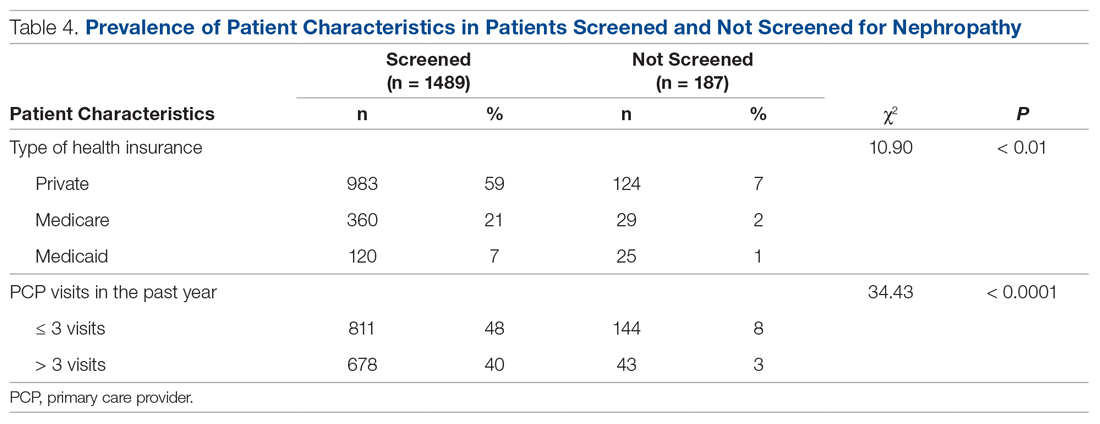
Changes in Screening Rate
The practice-wide screening rate was 83.8% at the start of this project in January 2018. The screening rate steadily increased throughout 2018, reaching 90.3% in August 2018, and then leveled off around 90% when the project was concluded at the end of November 2018 (Figure). As an added benefit of the increased screening rates, a number of patients were initiated on an ACE inhibitor or ARB based on the team’s screening efforts.
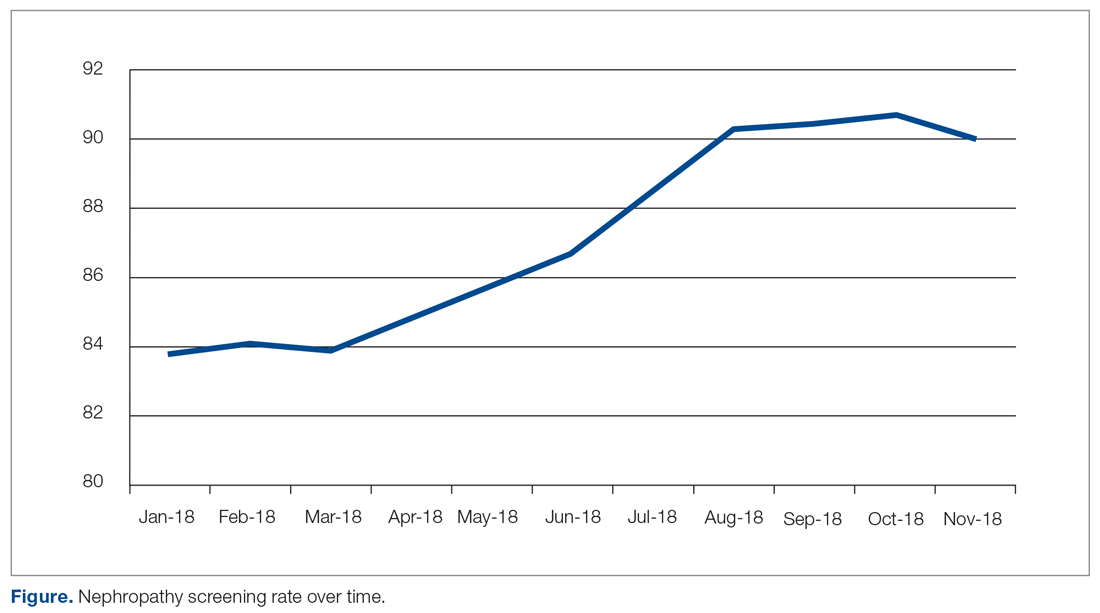
Predictors of Nephropathy Screening
A logistic regression analysis was conducted with nephropathy screening (screened or not screened) as the outcome and 7 patient characteristics as predictors: type of insurance (private, Medicare, or Medicaid), PCP visits in the past 12 months (≤ 3 or > 3), distance in miles of the patient’s residence from the clinic, age, last HbA1c value, last ACR value, and last creatinine value. A test of the full model with all 7 predictors was statistically significant (χ2 (8) = 57.77, P < 0.001). Table 5 shows regression coefficients, Wald statistics, and 95% confidence intervals for odds ratios for each of the 7 predictors. According to the Wald criterion, 3 patient characteristics were significant predictors of nephropathy screening: age, distance between the patient’s home and clinic, and number of PCP visits in the past 12 months. After adjusting for the covariates, there were still significant associations between the nephropathy screening status and age ( χ2(1) = 9.64, P < 0.01); distance between the patient’s home and the clinic (χ2(1) = 3.98, P < 0.05); and the number of PCP visits in the previous year (χ2(1) = 21.74, P < 0.001). With each 1-year increment in age, the odds of completing the nephropathy screening increased by 3.2%. With each 1-mile increase in the distance between the patient’s home and clinic, the odds of completing the nephropathy screening decreased by 0.2%. Patients who visited the clinic more than 3 times in a year were 3.9 times (95% confidence interval, 2.2-7.0) more likely to complete the nephropathy screening than their counterparts who visited fewer than 3 times per year.
In summary, older patients living within about 164 miles of the clinic (ie, within 1 standard deviation from the average miles between patient’s homes and the clinic) who visited their PCP 3 or more times per year were the most likely to be screened.
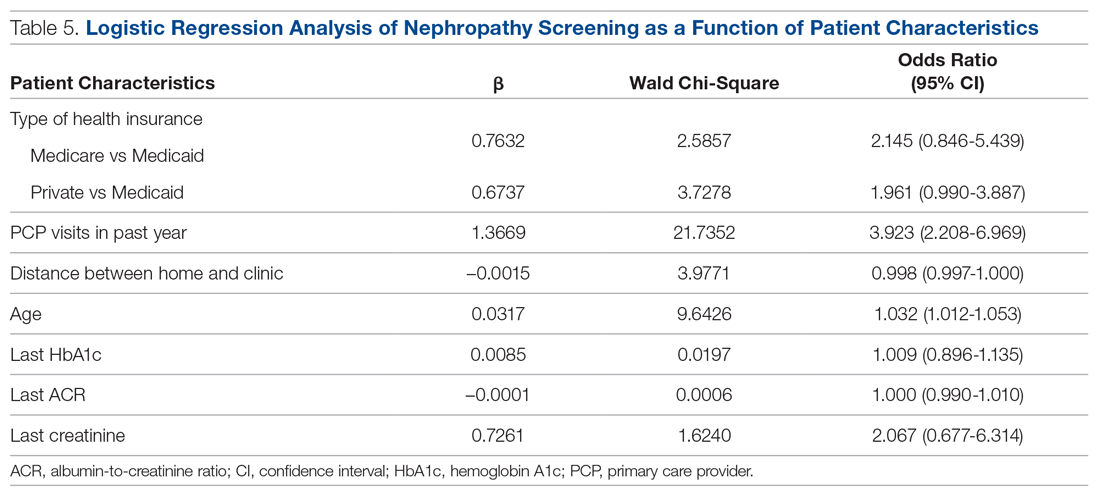
Discussion
Diabetic nephropathy is a critical issue facing family medicine providers and patients. The morbidity and mortality costs are significant, as diabetic nephropathy is the leading cause of end-stage renal disease. While the ADA recommends annual ACR screening in patients with diabetes and prescription of ACE inhibitors or ARBs in patients who qualify, many patients do not receive these interventions, despite following up with a provider.12-15 There is no current literature that indicates the compliance rates in the rural setting. Due to health disparities in the rural setting noted in the literature, it could be hypothesized that these individuals are at high risk of not meeting these screening and treatment recommendations.16,17 Limited access to care and resources, gaps in insurance coverage, and lower health literacy are a few barriers identified in the rural population that may influence whether these measures are met.17
Considering the disease burden of diabetes and its related complications, including nephropathy, consistent screening is necessary to reduce diabetes-related burdens and cost, while also increasing the quality of life for patients with diabetes. All parties must be involved to ensure appropriate compliance and treatment. Our institution’s implementation of quality improvement strategies has key implications for nephropathy screening and treatment efforts in rural settings.
An additional step of having a health care provider (other than the PCP) screen all patients who are not meeting the standard allows for identification of gaps in care. In our quality improvement workflow, the clinical pharmacist screened all patients for candidacy for ACE inhibitor/ARB therapy. While only a small percentage of patients qualified, many of these patients had previously been on therapy and were discontinued for an unknown reason or were stopped due to an acute condition (eg, acute kidney injury) and never restarted after recovery. Other patients required additional education that therapy would be utilized for nephroprotection versus blood pressure management (secondary to an elevated ACR). This highlights the importance of transitions of care and ongoing, intensive education, not only during initial diagnosis but also throughout the disease-state progression.
Utilization of EHRs and telephone outreach are additional aspects of care that can be provided. Our improved rates of compliance with these care interventions parallel findings from previous studies.15,18 Optimization of an institution’s EHR can aid in standardization of care, workflow management, and communication with patients, as well as alert nursing or support staff of screening needs. Techniques such as best practice reminders, patient chart messages, and nursing-entered physician alerts on daily schedules have been shown to increase rates of compliance with nephropathy standards. These findings underscore an additional opportunity for nursing and support staff to be better integrated into care.
Despite the success of this quality improvement initiative, there remain some limitations. The processes we used in this project may not be applicable to every institution and may have limited external validity. Primarily, while these processes may be implemented at some sites, without additional support staff (ie, extra nursing staff, pharmacists) and students to aid in patient outreach, success may be limited due to provider time constraints. Additionally, our workflow process demonstrates significant incorporation of an EHR system for patient outreach. Institutions and/or clinics that heavily rely on paper charts and paper outreach may face barriers with bulk orders (eg, ACR) and messages, interventions that streamlined our population health management. Finally, this project focuses on only 1 aspect of population health management for patients with diabetes. While nephropathy is a critical aspect of caring for individuals with diabetes, this patient outreach does not address retinopathy screening, HbA1c control, or vaccination rates, which are other components of care.
Conclusion
Although this evaluation does not provide insight into why patients were not treated or screened, it demonstrates processes to improve compliance in patients with diabetic nephropathy. Rural health care facilities require an ongoing program of change and evaluation, with the aim to improve the provision of services, increase screening, and encourage team member involvement in health promotion. This study demonstrates that combining team-based interventions with quality control monitoring can significantly improve compliance with recommended nephropathy screening and treatment in rural patients with diabetes at a family medicine clinic.
Corresponding author: Amie M. Ashcraft, West Virginia University, Department of Family Medicine, 1 Medical Center Drive, Box 9152, Morgantown, WV 26506; [email protected].
Financial disclosures: None.
Acknowledgment: The authors thank the faculty, residents, nurses, and clinic staff for their hard work and dedication to this effort: Umama Sadia, Michelle Prestoza, Richard Dattola, Greg Doyle, Dana King, Mike Maroon, Kendra Under, Judy Siebert, Christine Snyder, Rachel Burge, Meagan Gribble, Lisa Metts, Kelsey Samek, Sarah Deavers, Amber Kitzmiller, Angela Lamp, Tina Waldeck, and Andrea Sukeruksa.
1. Centers for Disease Control and Prevention (CDC). National diabetes statistics report. Estimates of diabetes and its burden in the United States. Atlanta, GA: CDC; 2017www.cdc.gov/diabetes/pdfs/data/statistics/national-diabetes-statistics-report.pdf. Accessed December 20, 2020.
2. American Diabetes Association (ADA). Economic costs of diabetes in the U.S. in 2017. Diabetes Care. 2018;41:917-928.
3. Wood L. Trends in national and regional economic distress, 1960-2000. Washington, DC: Appalachian Regional Commission; 2005.
4. Barker L, Crespo R, Gerzoff RB, et al. Residence in a distressed county in Appalachia as a risk factor for diabetes, Behavioral Risk Factor Surveillance System, 2006-2007. Prev Chronic Dis. 2010;7:A104.
5. Barker L, Kirtland KA, Gregg E, et al. Geographic distribution of diagnosed diabetes in the United States: A diabetes belt. Am J Prev Med. 2011;40:434-439.
6. Gross JL, de Azevedo MJ, Silveiro SP, et al. Diabetic nephropathy: Diagnosis, prevention, and treatment. Diabetes Care. 2005;28:164-176.
7. United States Renal Data System (USRDS). Annual data report. Ann Arbor, MI: USRDS; 2018. www.usrds.org/2018/view/Default.aspx. Accessed December 20, 2020.
8. Halverson JA, Bichak G. Underlying socioeconomic factors influencing health disparities in the Appalachian region. Washington, DC: Appalachian Regional Commission; 2008.
9. Shell R, Tudiver F. Barriers to cancer screening by rural Appalachian primary care providers. J Rural Health. 2004;20:368-373.
10. Hatcher J, Dignan MB, Schoenberg N. How do rural health care providers and patients view barriers to colorectal cancer screening? Insights from Appalachian Kentucky. Nurs Clin North Am. 2011;46:181-192.
11. Scott S, McSpirit S. The suspicious, untrusting hillbilly in political-economic contexts: Stereotypes and social trust in the Appalachian coalfields. Pract Anthropol. 2014;36:42-46.
12. Kirkman MS, Williams SR, Caffrey HH, Marrero DG. Impact of a program to improve adherence to diabetes guidelines by primary care physicians. Diabetes Care. 2002;25:1946-1951.
13. Byun SH, Ma SH, Jun JK, et al. Screening for diabetic retinopathy and nephropathy in patients with diabetes: A nationwide survey in Korea. PLoS One. 2013;8:e62991.
14. Flood D, Garcia P, Douglas K, et al. Screening for chronic kidney disease in a community-based diabetes cohort in rural Guatemala: A cross-sectional study. BMJ Open. 2018;8:e019778.
15. Anabtawi A, Mathew LM. Improving compliance with screening of diabetic patients for microalbuminuria in primary care practice. ISRN Endocrinology. 2013:893913.
16. Tonks SA, Makwana S, Salanitro AH, et al. Quality of diabetes mellitus care by rural primary care physicians. J Rural Health. 2012;28:364-371.
17. Douthit N, Kiv S, Dwolatzky T, Biswas S. Exposing some important barriers to health care access in the rural USA. Public Health. 2015;129:611-620.
18. Weber V, Bloom F, Pierdon S, Wood C. Employing the electronic health record to improve diabetes care: a multifaceted intervention in an integrated delivery system. J Gen Intern Med. 2008;23:379-382.
From West Virginia University, Morgantown, WV.
Abstract
Objective: To describe the strategies a family medicine clinic in Appalachia utilized to increase nephropathy screening rates as well as to explore the factors predictive of nephropathy screening in patients with diabetes.
Design: This quality improvement project targeted the points in the care process when patients are lost to follow-up for nephropathy screening.
Setting and participants: Patients with diabetes cared for by a primary care provider (PCP) at an academic family medicine practice in Appalachia from January 2018 to November 2018.
Interventions: Bulk orders for albumin-to-creatinine (ACR) testing and urine collection during clinic visit, enhanced patient communication through bulk communication reminders and individual patient outreach, and education of clinic providers.
Measurements: Demographic data and monthly nephropathy screening rates.
Results: The nephropathy screening rate increased by 6.2% during the project. Older patients living closer to the clinic who visited their PCP 3 or more times per year were the most likely to be screened.
Conclusion: Combining team-based interventions with quality control monitoring can significantly improve compliance with recommended nephropathy treatment and screening in rural patients with diabetes at a family medicine clinic.
Keywords: rural; kidney disease; albumin-to-creatinine ratio; electronic health record.
According to the Centers for Disease Control and Prevention (CDC), an estimated 30.3 million people in the United States—about 9.4% of the population—have been diagnosed with diabetes.1 Diabetes is the seventh leading cause of death in the United States, and it contributes to other leading causes of death: heart disease and stroke.1 Diabetes also is related to high morbidity risk and is a leading cause of chronic kidney disease.1 The total cost of diagnosed diabetes was estimated at $327 billion in direct medical costs and reduced productivity.2
Residents of Appalachia bear a disproportionate burden of diabetes and other related negative health outcomes; these outcomes are influenced by a number of factors, including socioeconomic status, poverty, rurality, and health care access. Rates of chronic disease, such as diabetes, are most pronounced in Appalachia’s most economically distressed counties.3-5 In 2011, the CDC labeled a 644-county area the “diabetes belt,” which included most of Appalachia.6 As a result of this elevated prevalence of diabetes in Appalachia as compared to the rest of the country, complications directly associated with diabetes are more commonly observed in Appalachian residents. One of the most damaging complications is diabetic nephropathy.
Diabetic nephropathy results from damage to the microvasculature of the kidney due to inadequately controlled blood glucose. This, in turn, leads to decreased renal function, eventually leading to clinically significant renal disease. The long-term complications associated with nephropathy can include many comorbid conditions, the most serious of which are progression to end-stage renal disease, dialysis requirement, and early mortality. Diabetic nephropathy affects approximately 40% of patients with type 1 and type 2 diabetes.7,8
One way to prevent complications of diabetic nephropathy, in addition to good glycemic control in patients with diabetes, is early and regular screening. Currently, the American Diabetes Association (ADA) recommends yearly screening for diabetic nephropathy in the form of a urine albumin-to-creatinine ratio (ACR) for patients 18 to 75 years of age.2 This screening to detect diabetic nephropathy is recognized as a marker of quality care by many public and private insurance agencies and medical specialty associations, such as the Centers for Medicare and Medicaid Services.
Many patients with diabetes are cared for by primary care providers (PCP), and these PCP appointments provide an opportune time to screen and appropriately treat nephropathy. Screening opportunities are often missed, however, due to time constraints and competing health priorities. There are also a number of other factors specific to the Appalachian region that reduce the likelihood of screening for diabetic nephropathy, such as a lack of health insurance, the need to travel long distances to see a PCP, work and household responsibilities, low levels of education and health literacy, and a mistrust of outsiders regarding personal matters, including health.9-11 While nephropathy can have a detrimental impact on patients across populations, it is of particular concern for a state located in the heart of Appalachia, such as West Virginia.
Given the disproportionate burden of diabetes in this region and the potentially severe consequences of undetected nephropathy, clinicians from an academic family medicine clinic in West Virginia undertook a quality improvement project to increase the rate of nephropathy screening and treatment among patients with diabetes. This article describes the intervention strategies the team utilized to increase nephropathy screening and treatment in patients 18 to 75 years of age who met quality measures for nephropathy screening or treatment in the previous 12 months and explores the factors most predictive of nephropathy screening in Appalachian patients in this age group. It also reports the challenges and opportunities encountered and offers suggestions for other providers and clinics attempting to increase their nephropathy screening rates.
Methods
Setting and Study Population
The study population included patients ages 18 to 75 years under the care of providers in an academic family medicine practice in West Virginia who had been diagnosed with diabetes mellitus. The study focused on those patients overdue for diabetic nephropathy screening (ie, had not been screened in previous 12 months). The project began in January 2018 with a screening rate of 83.8%. The goal of this project was to increase this compliance metric by at least 5%. The project protocol was submitted to the West Virginia University Institutional Review Board, and, because it is a quality improvement project, permission was given to proceed without a board review.
Interventions
The team identified and implemented several interventions intended to reduce screening barriers and increase the screening rate.
Bulk orders for ACR and urine collection during clinic visits. Prior to initiation of this project, it was left to individual clinic providers to order nephropathy screening for patients with diabetes during a clinic visit; after receiving the order for “random urine microalbumin/creatinine ratio,” patients then had to travel to a lab to provide a urine sample. For this project and moving forward, the team changed to the procedure of initiating bulk ACR orders and collecting urine samples during clinic visits from all patients ages 18 to 75 years who have diabetes.
Bulk communication reminders. Since many patients with diabetes may not have realized they were overdue for nephropathy screening, the team began sending out bulk communication reminders through either the institution’s electronic health record (EHR; MyChart) or postal service–delivered physical letters (according to patient communication preferences) to remind patients that they were due for screening and to encourage them to schedule an appointment or keep a previously scheduled appointment with their PCP.
Individual patient outreach. A team of pharmacy students led by a licensed pharmacist in the family medicine clinic contacted patients overdue for screening even after bulk communication reminders went out. The students telephoned patients 2 to 3 months following the bulk communication. The students obtained an updated list of patients with diabetes ages 18 to 75 years from an EHR quality report. They began by prescreening the patients on the overdue list for potential candidacy for an angiotensin-converting enzyme (ACE) inhibitor or an angiotensin II receptor blocker (ARB). Screening for candidacy included evaluation of recent blood pressure readings, electrolytes (ie, basic metabolic panel), and ACR. If the students determined a patient was a candidate, they presented the patient to the preceptor for verification and then reached out to the provider with a recommendation. If the provider agreed, the student contacted the patient by telephone for medication counseling and education. The remaining patients determined not to be candidates for ACE inhibitors or ARBs were contacted by the pharmacy students by telephone to remind them that laboratory work was pending. Up to 3 phone call attempts were made before patients were determined to be unreachable. Students left voice mails with generic reminders if a patient could not be reached. If a patient answered, the student provided a reminder but also reviewed indications for lab work, the reason why the provider wished for follow-up, and updated lab hours. Students also followed up with the results of the work-up, as appropriate. During this outreach process, the student team encountered a number of patients who had moved or changed to a PCP outside of the family medicine clinic. In these cases, the EHR was updated and those patients were removed from the list of patients altogether.
Education of clinic providers. Clinic providers were educated during faculty and resident meetings and didactic learning sessions on identifying patients within the EHR who are due for nephropathy screening. They also received instruction on how to update the EHR to reflect completed screenings.
Data Analysis
All analyses in this study were conducted using SAS (version 9.4, 2013, SAS Institute Inc., Cary, NC). Descriptive analyses were conducted to summarize basic patient demographic information. To compare patients screened within the previous 12 months to those patients overdue for screening, 2-sample t-tests were used to examine differences in patients’ age, HbA1c, ACR, and creatinine level and the distance (in miles) between the patient’s home and the clinic. Chi-square analyses were used to examine the relationship between whether a patient was recently screened for nephropathy and the patient’s insurance, number of patient visits in the previous 12 months, and provider level. Logistic regression analyses were conducted to control for covariates and to explore which factors were most predictive of nephropathy screening. All tests were 2-tailed, and P values less than 0.05 were considered statistically significant.
Results
Patient Characteristics
There were 1676 family medicine clinic patients with diabetes between 18 and 75 years of age (Table 1 and Table 2). Of the total sample, 1489 (88.8%) had completed screening for nephropathy in the 12 months prior to evaluation, and 67.5%, 23.7%, and 8.8% of patients had private insurance, Medicare, and Medicaid, respectively. 
The mean (SD) age of the patients was 56.3 (11.9) years. The mean distance between the patient’s home and the clinic was 26.6 (76.8) miles. The mean number of visits was 3.6 (2.9) per year, and 43.0% of the patientvisited the clinic more than 3 times in a year. The mean values for HbA1c (%), creatinine (g/mol), and ACR (mg/g) were 7.7 (1.9), 1.0 (0.7), and 9.4 (31.4), respectively.
Screening of Patients for Nephropathy
Patients with Medicare and private insurance were more likely to have completed the nephropathy screening than those with Medicaid (92.5% versus 88.8% versus 82.8%, P = 0.004; Table 3 and Table 4). 

Changes in Screening Rate
The practice-wide screening rate was 83.8% at the start of this project in January 2018. The screening rate steadily increased throughout 2018, reaching 90.3% in August 2018, and then leveled off around 90% when the project was concluded at the end of November 2018 (Figure). As an added benefit of the increased screening rates, a number of patients were initiated on an ACE inhibitor or ARB based on the team’s screening efforts.

Predictors of Nephropathy Screening
A logistic regression analysis was conducted with nephropathy screening (screened or not screened) as the outcome and 7 patient characteristics as predictors: type of insurance (private, Medicare, or Medicaid), PCP visits in the past 12 months (≤ 3 or > 3), distance in miles of the patient’s residence from the clinic, age, last HbA1c value, last ACR value, and last creatinine value. A test of the full model with all 7 predictors was statistically significant (χ2 (8) = 57.77, P < 0.001). Table 5 shows regression coefficients, Wald statistics, and 95% confidence intervals for odds ratios for each of the 7 predictors. According to the Wald criterion, 3 patient characteristics were significant predictors of nephropathy screening: age, distance between the patient’s home and clinic, and number of PCP visits in the past 12 months. After adjusting for the covariates, there were still significant associations between the nephropathy screening status and age ( χ2(1) = 9.64, P < 0.01); distance between the patient’s home and the clinic (χ2(1) = 3.98, P < 0.05); and the number of PCP visits in the previous year (χ2(1) = 21.74, P < 0.001). With each 1-year increment in age, the odds of completing the nephropathy screening increased by 3.2%. With each 1-mile increase in the distance between the patient’s home and clinic, the odds of completing the nephropathy screening decreased by 0.2%. Patients who visited the clinic more than 3 times in a year were 3.9 times (95% confidence interval, 2.2-7.0) more likely to complete the nephropathy screening than their counterparts who visited fewer than 3 times per year.
In summary, older patients living within about 164 miles of the clinic (ie, within 1 standard deviation from the average miles between patient’s homes and the clinic) who visited their PCP 3 or more times per year were the most likely to be screened.

Discussion
Diabetic nephropathy is a critical issue facing family medicine providers and patients. The morbidity and mortality costs are significant, as diabetic nephropathy is the leading cause of end-stage renal disease. While the ADA recommends annual ACR screening in patients with diabetes and prescription of ACE inhibitors or ARBs in patients who qualify, many patients do not receive these interventions, despite following up with a provider.12-15 There is no current literature that indicates the compliance rates in the rural setting. Due to health disparities in the rural setting noted in the literature, it could be hypothesized that these individuals are at high risk of not meeting these screening and treatment recommendations.16,17 Limited access to care and resources, gaps in insurance coverage, and lower health literacy are a few barriers identified in the rural population that may influence whether these measures are met.17
Considering the disease burden of diabetes and its related complications, including nephropathy, consistent screening is necessary to reduce diabetes-related burdens and cost, while also increasing the quality of life for patients with diabetes. All parties must be involved to ensure appropriate compliance and treatment. Our institution’s implementation of quality improvement strategies has key implications for nephropathy screening and treatment efforts in rural settings.
An additional step of having a health care provider (other than the PCP) screen all patients who are not meeting the standard allows for identification of gaps in care. In our quality improvement workflow, the clinical pharmacist screened all patients for candidacy for ACE inhibitor/ARB therapy. While only a small percentage of patients qualified, many of these patients had previously been on therapy and were discontinued for an unknown reason or were stopped due to an acute condition (eg, acute kidney injury) and never restarted after recovery. Other patients required additional education that therapy would be utilized for nephroprotection versus blood pressure management (secondary to an elevated ACR). This highlights the importance of transitions of care and ongoing, intensive education, not only during initial diagnosis but also throughout the disease-state progression.
Utilization of EHRs and telephone outreach are additional aspects of care that can be provided. Our improved rates of compliance with these care interventions parallel findings from previous studies.15,18 Optimization of an institution’s EHR can aid in standardization of care, workflow management, and communication with patients, as well as alert nursing or support staff of screening needs. Techniques such as best practice reminders, patient chart messages, and nursing-entered physician alerts on daily schedules have been shown to increase rates of compliance with nephropathy standards. These findings underscore an additional opportunity for nursing and support staff to be better integrated into care.
Despite the success of this quality improvement initiative, there remain some limitations. The processes we used in this project may not be applicable to every institution and may have limited external validity. Primarily, while these processes may be implemented at some sites, without additional support staff (ie, extra nursing staff, pharmacists) and students to aid in patient outreach, success may be limited due to provider time constraints. Additionally, our workflow process demonstrates significant incorporation of an EHR system for patient outreach. Institutions and/or clinics that heavily rely on paper charts and paper outreach may face barriers with bulk orders (eg, ACR) and messages, interventions that streamlined our population health management. Finally, this project focuses on only 1 aspect of population health management for patients with diabetes. While nephropathy is a critical aspect of caring for individuals with diabetes, this patient outreach does not address retinopathy screening, HbA1c control, or vaccination rates, which are other components of care.
Conclusion
Although this evaluation does not provide insight into why patients were not treated or screened, it demonstrates processes to improve compliance in patients with diabetic nephropathy. Rural health care facilities require an ongoing program of change and evaluation, with the aim to improve the provision of services, increase screening, and encourage team member involvement in health promotion. This study demonstrates that combining team-based interventions with quality control monitoring can significantly improve compliance with recommended nephropathy screening and treatment in rural patients with diabetes at a family medicine clinic.
Corresponding author: Amie M. Ashcraft, West Virginia University, Department of Family Medicine, 1 Medical Center Drive, Box 9152, Morgantown, WV 26506; [email protected].
Financial disclosures: None.
Acknowledgment: The authors thank the faculty, residents, nurses, and clinic staff for their hard work and dedication to this effort: Umama Sadia, Michelle Prestoza, Richard Dattola, Greg Doyle, Dana King, Mike Maroon, Kendra Under, Judy Siebert, Christine Snyder, Rachel Burge, Meagan Gribble, Lisa Metts, Kelsey Samek, Sarah Deavers, Amber Kitzmiller, Angela Lamp, Tina Waldeck, and Andrea Sukeruksa.
From West Virginia University, Morgantown, WV.
Abstract
Objective: To describe the strategies a family medicine clinic in Appalachia utilized to increase nephropathy screening rates as well as to explore the factors predictive of nephropathy screening in patients with diabetes.
Design: This quality improvement project targeted the points in the care process when patients are lost to follow-up for nephropathy screening.
Setting and participants: Patients with diabetes cared for by a primary care provider (PCP) at an academic family medicine practice in Appalachia from January 2018 to November 2018.
Interventions: Bulk orders for albumin-to-creatinine (ACR) testing and urine collection during clinic visit, enhanced patient communication through bulk communication reminders and individual patient outreach, and education of clinic providers.
Measurements: Demographic data and monthly nephropathy screening rates.
Results: The nephropathy screening rate increased by 6.2% during the project. Older patients living closer to the clinic who visited their PCP 3 or more times per year were the most likely to be screened.
Conclusion: Combining team-based interventions with quality control monitoring can significantly improve compliance with recommended nephropathy treatment and screening in rural patients with diabetes at a family medicine clinic.
Keywords: rural; kidney disease; albumin-to-creatinine ratio; electronic health record.
According to the Centers for Disease Control and Prevention (CDC), an estimated 30.3 million people in the United States—about 9.4% of the population—have been diagnosed with diabetes.1 Diabetes is the seventh leading cause of death in the United States, and it contributes to other leading causes of death: heart disease and stroke.1 Diabetes also is related to high morbidity risk and is a leading cause of chronic kidney disease.1 The total cost of diagnosed diabetes was estimated at $327 billion in direct medical costs and reduced productivity.2
Residents of Appalachia bear a disproportionate burden of diabetes and other related negative health outcomes; these outcomes are influenced by a number of factors, including socioeconomic status, poverty, rurality, and health care access. Rates of chronic disease, such as diabetes, are most pronounced in Appalachia’s most economically distressed counties.3-5 In 2011, the CDC labeled a 644-county area the “diabetes belt,” which included most of Appalachia.6 As a result of this elevated prevalence of diabetes in Appalachia as compared to the rest of the country, complications directly associated with diabetes are more commonly observed in Appalachian residents. One of the most damaging complications is diabetic nephropathy.
Diabetic nephropathy results from damage to the microvasculature of the kidney due to inadequately controlled blood glucose. This, in turn, leads to decreased renal function, eventually leading to clinically significant renal disease. The long-term complications associated with nephropathy can include many comorbid conditions, the most serious of which are progression to end-stage renal disease, dialysis requirement, and early mortality. Diabetic nephropathy affects approximately 40% of patients with type 1 and type 2 diabetes.7,8
One way to prevent complications of diabetic nephropathy, in addition to good glycemic control in patients with diabetes, is early and regular screening. Currently, the American Diabetes Association (ADA) recommends yearly screening for diabetic nephropathy in the form of a urine albumin-to-creatinine ratio (ACR) for patients 18 to 75 years of age.2 This screening to detect diabetic nephropathy is recognized as a marker of quality care by many public and private insurance agencies and medical specialty associations, such as the Centers for Medicare and Medicaid Services.
Many patients with diabetes are cared for by primary care providers (PCP), and these PCP appointments provide an opportune time to screen and appropriately treat nephropathy. Screening opportunities are often missed, however, due to time constraints and competing health priorities. There are also a number of other factors specific to the Appalachian region that reduce the likelihood of screening for diabetic nephropathy, such as a lack of health insurance, the need to travel long distances to see a PCP, work and household responsibilities, low levels of education and health literacy, and a mistrust of outsiders regarding personal matters, including health.9-11 While nephropathy can have a detrimental impact on patients across populations, it is of particular concern for a state located in the heart of Appalachia, such as West Virginia.
Given the disproportionate burden of diabetes in this region and the potentially severe consequences of undetected nephropathy, clinicians from an academic family medicine clinic in West Virginia undertook a quality improvement project to increase the rate of nephropathy screening and treatment among patients with diabetes. This article describes the intervention strategies the team utilized to increase nephropathy screening and treatment in patients 18 to 75 years of age who met quality measures for nephropathy screening or treatment in the previous 12 months and explores the factors most predictive of nephropathy screening in Appalachian patients in this age group. It also reports the challenges and opportunities encountered and offers suggestions for other providers and clinics attempting to increase their nephropathy screening rates.
Methods
Setting and Study Population
The study population included patients ages 18 to 75 years under the care of providers in an academic family medicine practice in West Virginia who had been diagnosed with diabetes mellitus. The study focused on those patients overdue for diabetic nephropathy screening (ie, had not been screened in previous 12 months). The project began in January 2018 with a screening rate of 83.8%. The goal of this project was to increase this compliance metric by at least 5%. The project protocol was submitted to the West Virginia University Institutional Review Board, and, because it is a quality improvement project, permission was given to proceed without a board review.
Interventions
The team identified and implemented several interventions intended to reduce screening barriers and increase the screening rate.
Bulk orders for ACR and urine collection during clinic visits. Prior to initiation of this project, it was left to individual clinic providers to order nephropathy screening for patients with diabetes during a clinic visit; after receiving the order for “random urine microalbumin/creatinine ratio,” patients then had to travel to a lab to provide a urine sample. For this project and moving forward, the team changed to the procedure of initiating bulk ACR orders and collecting urine samples during clinic visits from all patients ages 18 to 75 years who have diabetes.
Bulk communication reminders. Since many patients with diabetes may not have realized they were overdue for nephropathy screening, the team began sending out bulk communication reminders through either the institution’s electronic health record (EHR; MyChart) or postal service–delivered physical letters (according to patient communication preferences) to remind patients that they were due for screening and to encourage them to schedule an appointment or keep a previously scheduled appointment with their PCP.
Individual patient outreach. A team of pharmacy students led by a licensed pharmacist in the family medicine clinic contacted patients overdue for screening even after bulk communication reminders went out. The students telephoned patients 2 to 3 months following the bulk communication. The students obtained an updated list of patients with diabetes ages 18 to 75 years from an EHR quality report. They began by prescreening the patients on the overdue list for potential candidacy for an angiotensin-converting enzyme (ACE) inhibitor or an angiotensin II receptor blocker (ARB). Screening for candidacy included evaluation of recent blood pressure readings, electrolytes (ie, basic metabolic panel), and ACR. If the students determined a patient was a candidate, they presented the patient to the preceptor for verification and then reached out to the provider with a recommendation. If the provider agreed, the student contacted the patient by telephone for medication counseling and education. The remaining patients determined not to be candidates for ACE inhibitors or ARBs were contacted by the pharmacy students by telephone to remind them that laboratory work was pending. Up to 3 phone call attempts were made before patients were determined to be unreachable. Students left voice mails with generic reminders if a patient could not be reached. If a patient answered, the student provided a reminder but also reviewed indications for lab work, the reason why the provider wished for follow-up, and updated lab hours. Students also followed up with the results of the work-up, as appropriate. During this outreach process, the student team encountered a number of patients who had moved or changed to a PCP outside of the family medicine clinic. In these cases, the EHR was updated and those patients were removed from the list of patients altogether.
Education of clinic providers. Clinic providers were educated during faculty and resident meetings and didactic learning sessions on identifying patients within the EHR who are due for nephropathy screening. They also received instruction on how to update the EHR to reflect completed screenings.
Data Analysis
All analyses in this study were conducted using SAS (version 9.4, 2013, SAS Institute Inc., Cary, NC). Descriptive analyses were conducted to summarize basic patient demographic information. To compare patients screened within the previous 12 months to those patients overdue for screening, 2-sample t-tests were used to examine differences in patients’ age, HbA1c, ACR, and creatinine level and the distance (in miles) between the patient’s home and the clinic. Chi-square analyses were used to examine the relationship between whether a patient was recently screened for nephropathy and the patient’s insurance, number of patient visits in the previous 12 months, and provider level. Logistic regression analyses were conducted to control for covariates and to explore which factors were most predictive of nephropathy screening. All tests were 2-tailed, and P values less than 0.05 were considered statistically significant.
Results
Patient Characteristics
There were 1676 family medicine clinic patients with diabetes between 18 and 75 years of age (Table 1 and Table 2). Of the total sample, 1489 (88.8%) had completed screening for nephropathy in the 12 months prior to evaluation, and 67.5%, 23.7%, and 8.8% of patients had private insurance, Medicare, and Medicaid, respectively. 
The mean (SD) age of the patients was 56.3 (11.9) years. The mean distance between the patient’s home and the clinic was 26.6 (76.8) miles. The mean number of visits was 3.6 (2.9) per year, and 43.0% of the patientvisited the clinic more than 3 times in a year. The mean values for HbA1c (%), creatinine (g/mol), and ACR (mg/g) were 7.7 (1.9), 1.0 (0.7), and 9.4 (31.4), respectively.
Screening of Patients for Nephropathy
Patients with Medicare and private insurance were more likely to have completed the nephropathy screening than those with Medicaid (92.5% versus 88.8% versus 82.8%, P = 0.004; Table 3 and Table 4). 

Changes in Screening Rate
The practice-wide screening rate was 83.8% at the start of this project in January 2018. The screening rate steadily increased throughout 2018, reaching 90.3% in August 2018, and then leveled off around 90% when the project was concluded at the end of November 2018 (Figure). As an added benefit of the increased screening rates, a number of patients were initiated on an ACE inhibitor or ARB based on the team’s screening efforts.

Predictors of Nephropathy Screening
A logistic regression analysis was conducted with nephropathy screening (screened or not screened) as the outcome and 7 patient characteristics as predictors: type of insurance (private, Medicare, or Medicaid), PCP visits in the past 12 months (≤ 3 or > 3), distance in miles of the patient’s residence from the clinic, age, last HbA1c value, last ACR value, and last creatinine value. A test of the full model with all 7 predictors was statistically significant (χ2 (8) = 57.77, P < 0.001). Table 5 shows regression coefficients, Wald statistics, and 95% confidence intervals for odds ratios for each of the 7 predictors. According to the Wald criterion, 3 patient characteristics were significant predictors of nephropathy screening: age, distance between the patient’s home and clinic, and number of PCP visits in the past 12 months. After adjusting for the covariates, there were still significant associations between the nephropathy screening status and age ( χ2(1) = 9.64, P < 0.01); distance between the patient’s home and the clinic (χ2(1) = 3.98, P < 0.05); and the number of PCP visits in the previous year (χ2(1) = 21.74, P < 0.001). With each 1-year increment in age, the odds of completing the nephropathy screening increased by 3.2%. With each 1-mile increase in the distance between the patient’s home and clinic, the odds of completing the nephropathy screening decreased by 0.2%. Patients who visited the clinic more than 3 times in a year were 3.9 times (95% confidence interval, 2.2-7.0) more likely to complete the nephropathy screening than their counterparts who visited fewer than 3 times per year.
In summary, older patients living within about 164 miles of the clinic (ie, within 1 standard deviation from the average miles between patient’s homes and the clinic) who visited their PCP 3 or more times per year were the most likely to be screened.

Discussion
Diabetic nephropathy is a critical issue facing family medicine providers and patients. The morbidity and mortality costs are significant, as diabetic nephropathy is the leading cause of end-stage renal disease. While the ADA recommends annual ACR screening in patients with diabetes and prescription of ACE inhibitors or ARBs in patients who qualify, many patients do not receive these interventions, despite following up with a provider.12-15 There is no current literature that indicates the compliance rates in the rural setting. Due to health disparities in the rural setting noted in the literature, it could be hypothesized that these individuals are at high risk of not meeting these screening and treatment recommendations.16,17 Limited access to care and resources, gaps in insurance coverage, and lower health literacy are a few barriers identified in the rural population that may influence whether these measures are met.17
Considering the disease burden of diabetes and its related complications, including nephropathy, consistent screening is necessary to reduce diabetes-related burdens and cost, while also increasing the quality of life for patients with diabetes. All parties must be involved to ensure appropriate compliance and treatment. Our institution’s implementation of quality improvement strategies has key implications for nephropathy screening and treatment efforts in rural settings.
An additional step of having a health care provider (other than the PCP) screen all patients who are not meeting the standard allows for identification of gaps in care. In our quality improvement workflow, the clinical pharmacist screened all patients for candidacy for ACE inhibitor/ARB therapy. While only a small percentage of patients qualified, many of these patients had previously been on therapy and were discontinued for an unknown reason or were stopped due to an acute condition (eg, acute kidney injury) and never restarted after recovery. Other patients required additional education that therapy would be utilized for nephroprotection versus blood pressure management (secondary to an elevated ACR). This highlights the importance of transitions of care and ongoing, intensive education, not only during initial diagnosis but also throughout the disease-state progression.
Utilization of EHRs and telephone outreach are additional aspects of care that can be provided. Our improved rates of compliance with these care interventions parallel findings from previous studies.15,18 Optimization of an institution’s EHR can aid in standardization of care, workflow management, and communication with patients, as well as alert nursing or support staff of screening needs. Techniques such as best practice reminders, patient chart messages, and nursing-entered physician alerts on daily schedules have been shown to increase rates of compliance with nephropathy standards. These findings underscore an additional opportunity for nursing and support staff to be better integrated into care.
Despite the success of this quality improvement initiative, there remain some limitations. The processes we used in this project may not be applicable to every institution and may have limited external validity. Primarily, while these processes may be implemented at some sites, without additional support staff (ie, extra nursing staff, pharmacists) and students to aid in patient outreach, success may be limited due to provider time constraints. Additionally, our workflow process demonstrates significant incorporation of an EHR system for patient outreach. Institutions and/or clinics that heavily rely on paper charts and paper outreach may face barriers with bulk orders (eg, ACR) and messages, interventions that streamlined our population health management. Finally, this project focuses on only 1 aspect of population health management for patients with diabetes. While nephropathy is a critical aspect of caring for individuals with diabetes, this patient outreach does not address retinopathy screening, HbA1c control, or vaccination rates, which are other components of care.
Conclusion
Although this evaluation does not provide insight into why patients were not treated or screened, it demonstrates processes to improve compliance in patients with diabetic nephropathy. Rural health care facilities require an ongoing program of change and evaluation, with the aim to improve the provision of services, increase screening, and encourage team member involvement in health promotion. This study demonstrates that combining team-based interventions with quality control monitoring can significantly improve compliance with recommended nephropathy screening and treatment in rural patients with diabetes at a family medicine clinic.
Corresponding author: Amie M. Ashcraft, West Virginia University, Department of Family Medicine, 1 Medical Center Drive, Box 9152, Morgantown, WV 26506; [email protected].
Financial disclosures: None.
Acknowledgment: The authors thank the faculty, residents, nurses, and clinic staff for their hard work and dedication to this effort: Umama Sadia, Michelle Prestoza, Richard Dattola, Greg Doyle, Dana King, Mike Maroon, Kendra Under, Judy Siebert, Christine Snyder, Rachel Burge, Meagan Gribble, Lisa Metts, Kelsey Samek, Sarah Deavers, Amber Kitzmiller, Angela Lamp, Tina Waldeck, and Andrea Sukeruksa.
1. Centers for Disease Control and Prevention (CDC). National diabetes statistics report. Estimates of diabetes and its burden in the United States. Atlanta, GA: CDC; 2017www.cdc.gov/diabetes/pdfs/data/statistics/national-diabetes-statistics-report.pdf. Accessed December 20, 2020.
2. American Diabetes Association (ADA). Economic costs of diabetes in the U.S. in 2017. Diabetes Care. 2018;41:917-928.
3. Wood L. Trends in national and regional economic distress, 1960-2000. Washington, DC: Appalachian Regional Commission; 2005.
4. Barker L, Crespo R, Gerzoff RB, et al. Residence in a distressed county in Appalachia as a risk factor for diabetes, Behavioral Risk Factor Surveillance System, 2006-2007. Prev Chronic Dis. 2010;7:A104.
5. Barker L, Kirtland KA, Gregg E, et al. Geographic distribution of diagnosed diabetes in the United States: A diabetes belt. Am J Prev Med. 2011;40:434-439.
6. Gross JL, de Azevedo MJ, Silveiro SP, et al. Diabetic nephropathy: Diagnosis, prevention, and treatment. Diabetes Care. 2005;28:164-176.
7. United States Renal Data System (USRDS). Annual data report. Ann Arbor, MI: USRDS; 2018. www.usrds.org/2018/view/Default.aspx. Accessed December 20, 2020.
8. Halverson JA, Bichak G. Underlying socioeconomic factors influencing health disparities in the Appalachian region. Washington, DC: Appalachian Regional Commission; 2008.
9. Shell R, Tudiver F. Barriers to cancer screening by rural Appalachian primary care providers. J Rural Health. 2004;20:368-373.
10. Hatcher J, Dignan MB, Schoenberg N. How do rural health care providers and patients view barriers to colorectal cancer screening? Insights from Appalachian Kentucky. Nurs Clin North Am. 2011;46:181-192.
11. Scott S, McSpirit S. The suspicious, untrusting hillbilly in political-economic contexts: Stereotypes and social trust in the Appalachian coalfields. Pract Anthropol. 2014;36:42-46.
12. Kirkman MS, Williams SR, Caffrey HH, Marrero DG. Impact of a program to improve adherence to diabetes guidelines by primary care physicians. Diabetes Care. 2002;25:1946-1951.
13. Byun SH, Ma SH, Jun JK, et al. Screening for diabetic retinopathy and nephropathy in patients with diabetes: A nationwide survey in Korea. PLoS One. 2013;8:e62991.
14. Flood D, Garcia P, Douglas K, et al. Screening for chronic kidney disease in a community-based diabetes cohort in rural Guatemala: A cross-sectional study. BMJ Open. 2018;8:e019778.
15. Anabtawi A, Mathew LM. Improving compliance with screening of diabetic patients for microalbuminuria in primary care practice. ISRN Endocrinology. 2013:893913.
16. Tonks SA, Makwana S, Salanitro AH, et al. Quality of diabetes mellitus care by rural primary care physicians. J Rural Health. 2012;28:364-371.
17. Douthit N, Kiv S, Dwolatzky T, Biswas S. Exposing some important barriers to health care access in the rural USA. Public Health. 2015;129:611-620.
18. Weber V, Bloom F, Pierdon S, Wood C. Employing the electronic health record to improve diabetes care: a multifaceted intervention in an integrated delivery system. J Gen Intern Med. 2008;23:379-382.
1. Centers for Disease Control and Prevention (CDC). National diabetes statistics report. Estimates of diabetes and its burden in the United States. Atlanta, GA: CDC; 2017www.cdc.gov/diabetes/pdfs/data/statistics/national-diabetes-statistics-report.pdf. Accessed December 20, 2020.
2. American Diabetes Association (ADA). Economic costs of diabetes in the U.S. in 2017. Diabetes Care. 2018;41:917-928.
3. Wood L. Trends in national and regional economic distress, 1960-2000. Washington, DC: Appalachian Regional Commission; 2005.
4. Barker L, Crespo R, Gerzoff RB, et al. Residence in a distressed county in Appalachia as a risk factor for diabetes, Behavioral Risk Factor Surveillance System, 2006-2007. Prev Chronic Dis. 2010;7:A104.
5. Barker L, Kirtland KA, Gregg E, et al. Geographic distribution of diagnosed diabetes in the United States: A diabetes belt. Am J Prev Med. 2011;40:434-439.
6. Gross JL, de Azevedo MJ, Silveiro SP, et al. Diabetic nephropathy: Diagnosis, prevention, and treatment. Diabetes Care. 2005;28:164-176.
7. United States Renal Data System (USRDS). Annual data report. Ann Arbor, MI: USRDS; 2018. www.usrds.org/2018/view/Default.aspx. Accessed December 20, 2020.
8. Halverson JA, Bichak G. Underlying socioeconomic factors influencing health disparities in the Appalachian region. Washington, DC: Appalachian Regional Commission; 2008.
9. Shell R, Tudiver F. Barriers to cancer screening by rural Appalachian primary care providers. J Rural Health. 2004;20:368-373.
10. Hatcher J, Dignan MB, Schoenberg N. How do rural health care providers and patients view barriers to colorectal cancer screening? Insights from Appalachian Kentucky. Nurs Clin North Am. 2011;46:181-192.
11. Scott S, McSpirit S. The suspicious, untrusting hillbilly in political-economic contexts: Stereotypes and social trust in the Appalachian coalfields. Pract Anthropol. 2014;36:42-46.
12. Kirkman MS, Williams SR, Caffrey HH, Marrero DG. Impact of a program to improve adherence to diabetes guidelines by primary care physicians. Diabetes Care. 2002;25:1946-1951.
13. Byun SH, Ma SH, Jun JK, et al. Screening for diabetic retinopathy and nephropathy in patients with diabetes: A nationwide survey in Korea. PLoS One. 2013;8:e62991.
14. Flood D, Garcia P, Douglas K, et al. Screening for chronic kidney disease in a community-based diabetes cohort in rural Guatemala: A cross-sectional study. BMJ Open. 2018;8:e019778.
15. Anabtawi A, Mathew LM. Improving compliance with screening of diabetic patients for microalbuminuria in primary care practice. ISRN Endocrinology. 2013:893913.
16. Tonks SA, Makwana S, Salanitro AH, et al. Quality of diabetes mellitus care by rural primary care physicians. J Rural Health. 2012;28:364-371.
17. Douthit N, Kiv S, Dwolatzky T, Biswas S. Exposing some important barriers to health care access in the rural USA. Public Health. 2015;129:611-620.
18. Weber V, Bloom F, Pierdon S, Wood C. Employing the electronic health record to improve diabetes care: a multifaceted intervention in an integrated delivery system. J Gen Intern Med. 2008;23:379-382.
Switching from TDF- to TAF-Containing Antiretroviral Therapy: Impact on Bone Mineral Density in Older Patients Living With HIV
Study Overview
Objective. To evaluate the effect of changing from tenofovir disoproxil fumarate (TDF) –containing antiretroviral therapy (ART) to tenofovir alafenamide (TAF) –containing ART in patients ages 60 years and older living with HIV.
Design. Prospective, open-label, multicenter, randomized controlled trial.
Setting and participants. The study was completed across 36 European centers over 48 weeks. Patients were enrolled from December 12, 2015, to March 21, 2018, and were eligible to participate if they were diagnosed with HIV-1; virologically suppressed to < 50 copies/mL; on a TDF-containing ART regimen; and ≥ 60 years of age.
Intervention. Participants (n = 167) were randomly assigned in a 2:1 ratio to ART with TAF (10 mg), elvitegravir (EVG; 150 mg), cobicistat (COB; 150 mg), and emtricitabine (FTC; 200 mg) or to continued therapy with a TDF-containing ART regimen (300 mg TDF).
Main outcome measures. Primary outcome measures were the change in spine and hip bone mineral density from baseline at week 48. Secondary outcome measures included bone mineral density changes from baseline at week 24, HIV viral suppression and change in CD4 count at weeks 24 and 48, and the assessment of safety and tolerability of each ART regimen until week 48.
Main results. At 48 weeks, patients (n = 111) in the TAF+EVG+COB+FTC group had a mean 2.24% (SD, 3.27) increase in spine bone mineral density, while those in the TDF-containing group (n = 56) had a mean 0.10% decrease (SD, 3.39), a difference of 2.43% (95% confidence interval [CI], 1.34-3.52; P < 0.0001). In addition, at 48 weeks patients in the TAF+EVG+COB+FTC group had a mean 1.33% increase (SD, 2.20) in hip bone mineral density, as compared with a mean 0.73% decrease (SD, 3.21) in the TDF-containing group, a difference of 2.04% (95% CI, 1.17-2.90; P < 0.0001).
Similar results were seen in spine and hip bone mineral density in the TAF+EVG+COB+FTC group at week 24, with increases of 1.75% (P = 0.00080) and 1.35% (P = 0.00040), respectively. Both treatment groups maintained high virologic suppression. The TAF+EVG+COB+FTC group maintained 94.5% virologic suppression at week 24 and 93.6% at week 48, as compared with virologic suppression of 100% and 94.5% at weeks 24 and 48, respectively, in the TDF-containing group. However, the TAF+EVG+COB+FTC group had an increase in CD4 count from baseline (56 cells/µL), with no real change in the TDF-containing group (–1 cell/µL). Patients in the TAF+EVG+COB+FTC group had a mean 27.8 mg/g decrease in urine albumin-to-creatinine ratio (UACR) versus a 7.7 mg/g decrease in the TDF-containing group (P = 0.0042). In addition, patients in the TAF+EVG+COB+FTC group had a mean 49.8 mg/g decrease in urine protein-to-creatinine ratio (UPCR) versus a 3.8 mg/g decrease in the TDF-containing group (P = 0.0042).
Conclusion. Patients 60 years of age or older living with virologically suppressed HIV may benefit from improved bone mineral density by switching from a TDF-containing ART regimen to a TAF-containing regimen after 48 weeks, which, in turn, may help to reduce the risk for osteoporosis. Patients who were switched to a TAF-containing regimen also had favorable improvements in UACR and UPCR, which could indicate better renal function.
The Centers for Disease Control and Prevention estimated that in 2018 nearly half of those living with HIV in the United States were older than 50 years.1 Today, the life expectancy of patients living with HIV on ART in developed countries is similar to that of patients not living with HIV. A meta-analysis published in 2017 estimated that patients diagnosed with HIV at age 20 beginning ART have a life expectancy of 63 years, and another study estimated that life expectancy in such patients is 89.1% of that of the general population in Canada.2,3 Overall, most people living with HIV infection are aging and at risk for medical conditions similar to persons without HIV disease. However, rates of osteoporosis in elderly patients with HIV are estimated to be 3 times greater than rates in persons without HIV.4 As a result, it is becoming increasingly important to find ways to decrease the risk of osteoporosis in these patients.
ART typically includes a nucleoside reverse transcriptase inhibitor (NRTI) combination and a third agent, such as an integrase strand inhibitor. Tenofovir is a commonly used backbone NRTI that comes in 2 forms, TDF (tenofovir disoproxil fumarate) and TAF (tenofovir alafenamide). Both are prodrugs that are converted to tenofovir diphosphate. TDF specifically is associated with an increased risk of bone loss and nephrotoxicity. The loss in bone mineral density is most similar to the bone loss seen with oral glucocorticoids.5 TDF has been shown to increase plasma levels of RANKL and tumor necrosis factor-α, leading to increased bone resorption.6 The long-term effects of TDF- versus TAF-containing ART on bone mineral density have, to our knowledge, not been compared previously in a randomized control study. The significance of demonstrating an increase in bone mineral density in the prevention of osteoporotic bone fracture in people living with HIV is less clear. A long-term cohort study completed in Japan looking at patients on TDF showed an increased risk of bone fractures in both older postmenopausal women and younger men.7 However, a retrospective cohort study looking at 1981 patients with HIV found no association between bone fractures and TDF.8
This randomized controlled trial used appropriate methods to measure the reported primary and secondary endpoints; however, it would be of benefit to continue following these patients to measure their true long-term risk of osteoporosis-related complications. In terms of the study’s secondary endpoints, it is notable that the patients maintained HIV viral suppression after the switch and CD4 counts remained stable (with a slight increase observed in the TAF-containing ART cohort).
In regard to the patient’s renal function, patients in the TAF group had significantly improved UACR and UPCR, which likely reflects improved glomerular filtration. Improved renal function is also increasingly important for patients with HIV, as up to 48.5% have some form of chronic kidney disease.9
Applications for Clinical Practice
This study shows that making the switch from TDF- to TAF-containing ART can lead to improved bone mineral density. We can extrapolate that switching may lead to a decreased risk of osteoporosis and osteoporosis-related complications, such as bone fracture, but this needs to be investigated in more detail. As demonstrated in this study, switching from a TDF- to a TAF-containing regimen can also lead to improved renal function while maintaining HIV viral suppression and CD4 counts.
Unfortunately, the regimen selected with TAF in this study (elvitegravir, cobicistat, and emtricitabine) includes cobicistat, which is no longer recommended as initial therapy due to its risk of drug-drug interactions, and elvitegravir, which has a lower barrier to resistance than other integrase strand inhibitors.10,11 The United States Department of Health and Human Services guidelines and the International Antiviral Society-USA Panel suggest using several other TAF-containing regimens for beginning or even switching therapy in older patients.10,11
When choosing between either a TAF- or a TDF-containing regimen to treat HIV infection in older patients, increasing evidence shows that using a TAF-containing ART regimen may be more beneficial for people living and aging with virologically suppressed HIV infection.
–Sean P. Bliven, and Norman L. Beatty, MD, University of Florida College of Medicine, Division of Infectious Diseases and Global Medicine, Gainesville, FL
1. Centers for Disease Control and Prevention. HIV among people aged 50 and over. 2018. https://www.cdc.gov/hiv/group/age/olderamericans/index.html. Accessed on November 22, 2019.
2. Teeraananchai S, Kerr S, Amin J, et al. Life expectancy of HIV-positive people after starting combination antiretroviral therapy: a meta-analysis. HIV Medicine. 2016;18:256-266.
3. Wandeler G, Johnson LF, Egger M. Trends in life expectancy of HIV-positive adults on antiretroviral therapy across the globe. Curr Opin HIV AIDS. 2016;11:492-500.
4. Brown TT, Qaqish RB. Antiretroviral therapy and the prevalence of osteopenia and osteoporosis: a meta-analytic review. AIDS. 2006;20:2165-2174.
5. Bolland MJ, Grey A, Reid IR. Skeletal health in adults with HIV infection. Lancet Diabetes Endocrinol. 2015;3:63-74.
6. Ofotokun I, Titanji K, Vunnava A, et al. Antiretroviral therapy induces a rapid increase in bone resorption that is positively associated with the magnitude of immune reconstitution in HIV infection. AIDS. 2016;30:405-414.
7. Komatsu A, Ikeda A, Kikuchi A, et al. Osteoporosis-related fractures in HIV-infected patients receiving long-term tenofovir disoproxil fumarate: an observational cohort study. Drug Saf. 2018;41:843-848.
8. Gediminas L, Wright EA, Dong Y, et al. Factors associated with fractures in HIV-infected persons: which factors matter? Osteoporos Int. 201728:239-244.
9. Naicker S, Rahmania, Kopp JB. HIV and chronic kidney disease. Clin Nephrol. 2015; 83(Suppl 1):S32-S38.
10. United States Department of Health and Human Services. Guidelines for the use of antiretroviral agents in adults and adolescents living with HIV. https://aidsinfo.nih.gov/guidelines/html/1/adult-and-adolescent-arv/0. Accessed December 10, 2019.
11. Saag MS, Benson CA, Gandhi RT, et al. Antiretroviral drugs for treatment and prevention of HIV infection in adults: 2018 recommendations of the International Antiviral Society-USA Panel. JAMA. 2018;320:379-396.
Study Overview
Objective. To evaluate the effect of changing from tenofovir disoproxil fumarate (TDF) –containing antiretroviral therapy (ART) to tenofovir alafenamide (TAF) –containing ART in patients ages 60 years and older living with HIV.
Design. Prospective, open-label, multicenter, randomized controlled trial.
Setting and participants. The study was completed across 36 European centers over 48 weeks. Patients were enrolled from December 12, 2015, to March 21, 2018, and were eligible to participate if they were diagnosed with HIV-1; virologically suppressed to < 50 copies/mL; on a TDF-containing ART regimen; and ≥ 60 years of age.
Intervention. Participants (n = 167) were randomly assigned in a 2:1 ratio to ART with TAF (10 mg), elvitegravir (EVG; 150 mg), cobicistat (COB; 150 mg), and emtricitabine (FTC; 200 mg) or to continued therapy with a TDF-containing ART regimen (300 mg TDF).
Main outcome measures. Primary outcome measures were the change in spine and hip bone mineral density from baseline at week 48. Secondary outcome measures included bone mineral density changes from baseline at week 24, HIV viral suppression and change in CD4 count at weeks 24 and 48, and the assessment of safety and tolerability of each ART regimen until week 48.
Main results. At 48 weeks, patients (n = 111) in the TAF+EVG+COB+FTC group had a mean 2.24% (SD, 3.27) increase in spine bone mineral density, while those in the TDF-containing group (n = 56) had a mean 0.10% decrease (SD, 3.39), a difference of 2.43% (95% confidence interval [CI], 1.34-3.52; P < 0.0001). In addition, at 48 weeks patients in the TAF+EVG+COB+FTC group had a mean 1.33% increase (SD, 2.20) in hip bone mineral density, as compared with a mean 0.73% decrease (SD, 3.21) in the TDF-containing group, a difference of 2.04% (95% CI, 1.17-2.90; P < 0.0001).
Similar results were seen in spine and hip bone mineral density in the TAF+EVG+COB+FTC group at week 24, with increases of 1.75% (P = 0.00080) and 1.35% (P = 0.00040), respectively. Both treatment groups maintained high virologic suppression. The TAF+EVG+COB+FTC group maintained 94.5% virologic suppression at week 24 and 93.6% at week 48, as compared with virologic suppression of 100% and 94.5% at weeks 24 and 48, respectively, in the TDF-containing group. However, the TAF+EVG+COB+FTC group had an increase in CD4 count from baseline (56 cells/µL), with no real change in the TDF-containing group (–1 cell/µL). Patients in the TAF+EVG+COB+FTC group had a mean 27.8 mg/g decrease in urine albumin-to-creatinine ratio (UACR) versus a 7.7 mg/g decrease in the TDF-containing group (P = 0.0042). In addition, patients in the TAF+EVG+COB+FTC group had a mean 49.8 mg/g decrease in urine protein-to-creatinine ratio (UPCR) versus a 3.8 mg/g decrease in the TDF-containing group (P = 0.0042).
Conclusion. Patients 60 years of age or older living with virologically suppressed HIV may benefit from improved bone mineral density by switching from a TDF-containing ART regimen to a TAF-containing regimen after 48 weeks, which, in turn, may help to reduce the risk for osteoporosis. Patients who were switched to a TAF-containing regimen also had favorable improvements in UACR and UPCR, which could indicate better renal function.
The Centers for Disease Control and Prevention estimated that in 2018 nearly half of those living with HIV in the United States were older than 50 years.1 Today, the life expectancy of patients living with HIV on ART in developed countries is similar to that of patients not living with HIV. A meta-analysis published in 2017 estimated that patients diagnosed with HIV at age 20 beginning ART have a life expectancy of 63 years, and another study estimated that life expectancy in such patients is 89.1% of that of the general population in Canada.2,3 Overall, most people living with HIV infection are aging and at risk for medical conditions similar to persons without HIV disease. However, rates of osteoporosis in elderly patients with HIV are estimated to be 3 times greater than rates in persons without HIV.4 As a result, it is becoming increasingly important to find ways to decrease the risk of osteoporosis in these patients.
ART typically includes a nucleoside reverse transcriptase inhibitor (NRTI) combination and a third agent, such as an integrase strand inhibitor. Tenofovir is a commonly used backbone NRTI that comes in 2 forms, TDF (tenofovir disoproxil fumarate) and TAF (tenofovir alafenamide). Both are prodrugs that are converted to tenofovir diphosphate. TDF specifically is associated with an increased risk of bone loss and nephrotoxicity. The loss in bone mineral density is most similar to the bone loss seen with oral glucocorticoids.5 TDF has been shown to increase plasma levels of RANKL and tumor necrosis factor-α, leading to increased bone resorption.6 The long-term effects of TDF- versus TAF-containing ART on bone mineral density have, to our knowledge, not been compared previously in a randomized control study. The significance of demonstrating an increase in bone mineral density in the prevention of osteoporotic bone fracture in people living with HIV is less clear. A long-term cohort study completed in Japan looking at patients on TDF showed an increased risk of bone fractures in both older postmenopausal women and younger men.7 However, a retrospective cohort study looking at 1981 patients with HIV found no association between bone fractures and TDF.8
This randomized controlled trial used appropriate methods to measure the reported primary and secondary endpoints; however, it would be of benefit to continue following these patients to measure their true long-term risk of osteoporosis-related complications. In terms of the study’s secondary endpoints, it is notable that the patients maintained HIV viral suppression after the switch and CD4 counts remained stable (with a slight increase observed in the TAF-containing ART cohort).
In regard to the patient’s renal function, patients in the TAF group had significantly improved UACR and UPCR, which likely reflects improved glomerular filtration. Improved renal function is also increasingly important for patients with HIV, as up to 48.5% have some form of chronic kidney disease.9
Applications for Clinical Practice
This study shows that making the switch from TDF- to TAF-containing ART can lead to improved bone mineral density. We can extrapolate that switching may lead to a decreased risk of osteoporosis and osteoporosis-related complications, such as bone fracture, but this needs to be investigated in more detail. As demonstrated in this study, switching from a TDF- to a TAF-containing regimen can also lead to improved renal function while maintaining HIV viral suppression and CD4 counts.
Unfortunately, the regimen selected with TAF in this study (elvitegravir, cobicistat, and emtricitabine) includes cobicistat, which is no longer recommended as initial therapy due to its risk of drug-drug interactions, and elvitegravir, which has a lower barrier to resistance than other integrase strand inhibitors.10,11 The United States Department of Health and Human Services guidelines and the International Antiviral Society-USA Panel suggest using several other TAF-containing regimens for beginning or even switching therapy in older patients.10,11
When choosing between either a TAF- or a TDF-containing regimen to treat HIV infection in older patients, increasing evidence shows that using a TAF-containing ART regimen may be more beneficial for people living and aging with virologically suppressed HIV infection.
–Sean P. Bliven, and Norman L. Beatty, MD, University of Florida College of Medicine, Division of Infectious Diseases and Global Medicine, Gainesville, FL
Study Overview
Objective. To evaluate the effect of changing from tenofovir disoproxil fumarate (TDF) –containing antiretroviral therapy (ART) to tenofovir alafenamide (TAF) –containing ART in patients ages 60 years and older living with HIV.
Design. Prospective, open-label, multicenter, randomized controlled trial.
Setting and participants. The study was completed across 36 European centers over 48 weeks. Patients were enrolled from December 12, 2015, to March 21, 2018, and were eligible to participate if they were diagnosed with HIV-1; virologically suppressed to < 50 copies/mL; on a TDF-containing ART regimen; and ≥ 60 years of age.
Intervention. Participants (n = 167) were randomly assigned in a 2:1 ratio to ART with TAF (10 mg), elvitegravir (EVG; 150 mg), cobicistat (COB; 150 mg), and emtricitabine (FTC; 200 mg) or to continued therapy with a TDF-containing ART regimen (300 mg TDF).
Main outcome measures. Primary outcome measures were the change in spine and hip bone mineral density from baseline at week 48. Secondary outcome measures included bone mineral density changes from baseline at week 24, HIV viral suppression and change in CD4 count at weeks 24 and 48, and the assessment of safety and tolerability of each ART regimen until week 48.
Main results. At 48 weeks, patients (n = 111) in the TAF+EVG+COB+FTC group had a mean 2.24% (SD, 3.27) increase in spine bone mineral density, while those in the TDF-containing group (n = 56) had a mean 0.10% decrease (SD, 3.39), a difference of 2.43% (95% confidence interval [CI], 1.34-3.52; P < 0.0001). In addition, at 48 weeks patients in the TAF+EVG+COB+FTC group had a mean 1.33% increase (SD, 2.20) in hip bone mineral density, as compared with a mean 0.73% decrease (SD, 3.21) in the TDF-containing group, a difference of 2.04% (95% CI, 1.17-2.90; P < 0.0001).
Similar results were seen in spine and hip bone mineral density in the TAF+EVG+COB+FTC group at week 24, with increases of 1.75% (P = 0.00080) and 1.35% (P = 0.00040), respectively. Both treatment groups maintained high virologic suppression. The TAF+EVG+COB+FTC group maintained 94.5% virologic suppression at week 24 and 93.6% at week 48, as compared with virologic suppression of 100% and 94.5% at weeks 24 and 48, respectively, in the TDF-containing group. However, the TAF+EVG+COB+FTC group had an increase in CD4 count from baseline (56 cells/µL), with no real change in the TDF-containing group (–1 cell/µL). Patients in the TAF+EVG+COB+FTC group had a mean 27.8 mg/g decrease in urine albumin-to-creatinine ratio (UACR) versus a 7.7 mg/g decrease in the TDF-containing group (P = 0.0042). In addition, patients in the TAF+EVG+COB+FTC group had a mean 49.8 mg/g decrease in urine protein-to-creatinine ratio (UPCR) versus a 3.8 mg/g decrease in the TDF-containing group (P = 0.0042).
Conclusion. Patients 60 years of age or older living with virologically suppressed HIV may benefit from improved bone mineral density by switching from a TDF-containing ART regimen to a TAF-containing regimen after 48 weeks, which, in turn, may help to reduce the risk for osteoporosis. Patients who were switched to a TAF-containing regimen also had favorable improvements in UACR and UPCR, which could indicate better renal function.
The Centers for Disease Control and Prevention estimated that in 2018 nearly half of those living with HIV in the United States were older than 50 years.1 Today, the life expectancy of patients living with HIV on ART in developed countries is similar to that of patients not living with HIV. A meta-analysis published in 2017 estimated that patients diagnosed with HIV at age 20 beginning ART have a life expectancy of 63 years, and another study estimated that life expectancy in such patients is 89.1% of that of the general population in Canada.2,3 Overall, most people living with HIV infection are aging and at risk for medical conditions similar to persons without HIV disease. However, rates of osteoporosis in elderly patients with HIV are estimated to be 3 times greater than rates in persons without HIV.4 As a result, it is becoming increasingly important to find ways to decrease the risk of osteoporosis in these patients.
ART typically includes a nucleoside reverse transcriptase inhibitor (NRTI) combination and a third agent, such as an integrase strand inhibitor. Tenofovir is a commonly used backbone NRTI that comes in 2 forms, TDF (tenofovir disoproxil fumarate) and TAF (tenofovir alafenamide). Both are prodrugs that are converted to tenofovir diphosphate. TDF specifically is associated with an increased risk of bone loss and nephrotoxicity. The loss in bone mineral density is most similar to the bone loss seen with oral glucocorticoids.5 TDF has been shown to increase plasma levels of RANKL and tumor necrosis factor-α, leading to increased bone resorption.6 The long-term effects of TDF- versus TAF-containing ART on bone mineral density have, to our knowledge, not been compared previously in a randomized control study. The significance of demonstrating an increase in bone mineral density in the prevention of osteoporotic bone fracture in people living with HIV is less clear. A long-term cohort study completed in Japan looking at patients on TDF showed an increased risk of bone fractures in both older postmenopausal women and younger men.7 However, a retrospective cohort study looking at 1981 patients with HIV found no association between bone fractures and TDF.8
This randomized controlled trial used appropriate methods to measure the reported primary and secondary endpoints; however, it would be of benefit to continue following these patients to measure their true long-term risk of osteoporosis-related complications. In terms of the study’s secondary endpoints, it is notable that the patients maintained HIV viral suppression after the switch and CD4 counts remained stable (with a slight increase observed in the TAF-containing ART cohort).
In regard to the patient’s renal function, patients in the TAF group had significantly improved UACR and UPCR, which likely reflects improved glomerular filtration. Improved renal function is also increasingly important for patients with HIV, as up to 48.5% have some form of chronic kidney disease.9
Applications for Clinical Practice
This study shows that making the switch from TDF- to TAF-containing ART can lead to improved bone mineral density. We can extrapolate that switching may lead to a decreased risk of osteoporosis and osteoporosis-related complications, such as bone fracture, but this needs to be investigated in more detail. As demonstrated in this study, switching from a TDF- to a TAF-containing regimen can also lead to improved renal function while maintaining HIV viral suppression and CD4 counts.
Unfortunately, the regimen selected with TAF in this study (elvitegravir, cobicistat, and emtricitabine) includes cobicistat, which is no longer recommended as initial therapy due to its risk of drug-drug interactions, and elvitegravir, which has a lower barrier to resistance than other integrase strand inhibitors.10,11 The United States Department of Health and Human Services guidelines and the International Antiviral Society-USA Panel suggest using several other TAF-containing regimens for beginning or even switching therapy in older patients.10,11
When choosing between either a TAF- or a TDF-containing regimen to treat HIV infection in older patients, increasing evidence shows that using a TAF-containing ART regimen may be more beneficial for people living and aging with virologically suppressed HIV infection.
–Sean P. Bliven, and Norman L. Beatty, MD, University of Florida College of Medicine, Division of Infectious Diseases and Global Medicine, Gainesville, FL
1. Centers for Disease Control and Prevention. HIV among people aged 50 and over. 2018. https://www.cdc.gov/hiv/group/age/olderamericans/index.html. Accessed on November 22, 2019.
2. Teeraananchai S, Kerr S, Amin J, et al. Life expectancy of HIV-positive people after starting combination antiretroviral therapy: a meta-analysis. HIV Medicine. 2016;18:256-266.
3. Wandeler G, Johnson LF, Egger M. Trends in life expectancy of HIV-positive adults on antiretroviral therapy across the globe. Curr Opin HIV AIDS. 2016;11:492-500.
4. Brown TT, Qaqish RB. Antiretroviral therapy and the prevalence of osteopenia and osteoporosis: a meta-analytic review. AIDS. 2006;20:2165-2174.
5. Bolland MJ, Grey A, Reid IR. Skeletal health in adults with HIV infection. Lancet Diabetes Endocrinol. 2015;3:63-74.
6. Ofotokun I, Titanji K, Vunnava A, et al. Antiretroviral therapy induces a rapid increase in bone resorption that is positively associated with the magnitude of immune reconstitution in HIV infection. AIDS. 2016;30:405-414.
7. Komatsu A, Ikeda A, Kikuchi A, et al. Osteoporosis-related fractures in HIV-infected patients receiving long-term tenofovir disoproxil fumarate: an observational cohort study. Drug Saf. 2018;41:843-848.
8. Gediminas L, Wright EA, Dong Y, et al. Factors associated with fractures in HIV-infected persons: which factors matter? Osteoporos Int. 201728:239-244.
9. Naicker S, Rahmania, Kopp JB. HIV and chronic kidney disease. Clin Nephrol. 2015; 83(Suppl 1):S32-S38.
10. United States Department of Health and Human Services. Guidelines for the use of antiretroviral agents in adults and adolescents living with HIV. https://aidsinfo.nih.gov/guidelines/html/1/adult-and-adolescent-arv/0. Accessed December 10, 2019.
11. Saag MS, Benson CA, Gandhi RT, et al. Antiretroviral drugs for treatment and prevention of HIV infection in adults: 2018 recommendations of the International Antiviral Society-USA Panel. JAMA. 2018;320:379-396.
1. Centers for Disease Control and Prevention. HIV among people aged 50 and over. 2018. https://www.cdc.gov/hiv/group/age/olderamericans/index.html. Accessed on November 22, 2019.
2. Teeraananchai S, Kerr S, Amin J, et al. Life expectancy of HIV-positive people after starting combination antiretroviral therapy: a meta-analysis. HIV Medicine. 2016;18:256-266.
3. Wandeler G, Johnson LF, Egger M. Trends in life expectancy of HIV-positive adults on antiretroviral therapy across the globe. Curr Opin HIV AIDS. 2016;11:492-500.
4. Brown TT, Qaqish RB. Antiretroviral therapy and the prevalence of osteopenia and osteoporosis: a meta-analytic review. AIDS. 2006;20:2165-2174.
5. Bolland MJ, Grey A, Reid IR. Skeletal health in adults with HIV infection. Lancet Diabetes Endocrinol. 2015;3:63-74.
6. Ofotokun I, Titanji K, Vunnava A, et al. Antiretroviral therapy induces a rapid increase in bone resorption that is positively associated with the magnitude of immune reconstitution in HIV infection. AIDS. 2016;30:405-414.
7. Komatsu A, Ikeda A, Kikuchi A, et al. Osteoporosis-related fractures in HIV-infected patients receiving long-term tenofovir disoproxil fumarate: an observational cohort study. Drug Saf. 2018;41:843-848.
8. Gediminas L, Wright EA, Dong Y, et al. Factors associated with fractures in HIV-infected persons: which factors matter? Osteoporos Int. 201728:239-244.
9. Naicker S, Rahmania, Kopp JB. HIV and chronic kidney disease. Clin Nephrol. 2015; 83(Suppl 1):S32-S38.
10. United States Department of Health and Human Services. Guidelines for the use of antiretroviral agents in adults and adolescents living with HIV. https://aidsinfo.nih.gov/guidelines/html/1/adult-and-adolescent-arv/0. Accessed December 10, 2019.
11. Saag MS, Benson CA, Gandhi RT, et al. Antiretroviral drugs for treatment and prevention of HIV infection in adults: 2018 recommendations of the International Antiviral Society-USA Panel. JAMA. 2018;320:379-396.
ECHELON-1 update: A+AVD bests ABVD in Hodgkin lymphoma
Brentuximab vedotin plus doxorubicin, vinblastine, and dacarbazine (A+AVD) provides “robust, sustained efficacy” in patients with Hodgkin lymphoma, according to investigators.
In the ECHELON-1 trial, investigators compared A+AVD to doxorubicin, bleomycin, vinblastine, and dacarbazine (ABVD) as frontline treatment for stage III or IV Hodgkin lymphoma. The 3-year progression-free survival (PFS) was superior in patients who received A+AVD, and this benefit was seen across most subgroups.
David J. Straus, MD, of Memorial Sloan Kettering Cancer Center in New York and his colleagues detailed these findings in Blood.
The phase 3 trial (NCT01712490) enrolled 1,334 patients with stage III or IV classical Hodgkin lymphoma. They were randomized to receive A+AVD (n = 664) or ABVD (n = 670). Baseline characteristics were similar between the treatment arms.
Positron emission tomography status after cycle 2 (PET2) was similar between the treatment arms as well. Most patients – 89% of the A+AVD arm and 86% of the ABVD arm – were PET2 negative. Treating physicians used PET2 status as a guide to potentially switch patients to an alternative regimen (radiotherapy or chemotherapy with or without transplant).
In a prior analysis, the study’s primary endpoint was modified PFS (time to progression, death, or noncomplete response after frontline therapy) per an independent review committee (N Engl J Med. 2018;378:331-44). The 2-year modified PFS rate was 82.1% in the A+AVD arm and 77.2% in the ABVD arm (hazard ratio, 0.77; P = .04).
PFS update
In the current analysis, the main exploratory endpoint was PFS per investigator. The 3-year PFS rate was significantly higher in the A+AVD arm than in the ABVD arm – 83.1% and 76.0%, respectively (HR, 0.704; P = .005).
The investigators observed a “consistent improvement in PFS” in the A+AVD arm, regardless of disease stage, International Prognostic score, Eastern Cooperative Oncology Group status, sex, or age. There was a significant improvement in PFS with A+AVD in PET2-negative patients and a trend toward improvement in PET2-positive patients. In the PET2-negative patients, the 3-year PFS was 85.8% in the A+AVD arm and 79.5% in the ABVD arm (HR, 0.69; P = .009). In PET2-positive patients, the 3-year PFS was 67.7% and 51.5%, respectively (HR, 0.59; P = .077).
“These data highlight that A+AVD provides a durable efficacy benefit, compared with ABVD, for frontline stage III/IV cHL [classical Hodgkin lymphoma], which is consistent across key subgroups regardless of patient status at PET2,” Dr. Straus and his colleagues wrote.
Safety update
In both treatment arms, peripheral neuropathy continued to improve or resolve with longer follow-up. Among patients who developed peripheral neuropathy, 78% in the A+AVD arm and 83% in the ABVD arm had improvement or resolution of the condition at 3 years.
Most patients had complete resolution of peripheral neuropathy; 62% in the A+AVD arm and 73% in the ABVD arm. The median time to complete resolution was 28 weeks (range, 0-167 weeks) after stopping A+AVD and 14 weeks (range, 0-188 weeks) after stopping ABVD.
The incidence of secondary malignancies was similar between the treatment arms. There were 14 secondary malignancies in the A+AVD arm (6 solid tumors, 8 hematologic malignancies) and 20 in the ABVD arm (9 solid tumors, 11 hematologic malignancies).
“A+AVD provided a sustained PFS benefit with a predictable and manageable safety profile,” Dr. Straus and colleagues wrote. “These data further support the advantages of A+AVD versus ABVD as frontline treatment of patients with advanced stage III or IV cHL [classical Hodgkin lymphoma].”
The ECHELON-1 trial was sponsored by Millennium Pharmaceuticals (a subsidiary of Takeda) and Seattle Genetics. The investigators disclosed relationships with Millennium, Takeda, Seattle Genetics, and a range of other companies.
SOURCE: Straus DJ et al. Blood. 2020 Jan 16. pii: blood.2019003127. doi: 10.1182/blood.2019003127.
Brentuximab vedotin plus doxorubicin, vinblastine, and dacarbazine (A+AVD) provides “robust, sustained efficacy” in patients with Hodgkin lymphoma, according to investigators.
In the ECHELON-1 trial, investigators compared A+AVD to doxorubicin, bleomycin, vinblastine, and dacarbazine (ABVD) as frontline treatment for stage III or IV Hodgkin lymphoma. The 3-year progression-free survival (PFS) was superior in patients who received A+AVD, and this benefit was seen across most subgroups.
David J. Straus, MD, of Memorial Sloan Kettering Cancer Center in New York and his colleagues detailed these findings in Blood.
The phase 3 trial (NCT01712490) enrolled 1,334 patients with stage III or IV classical Hodgkin lymphoma. They were randomized to receive A+AVD (n = 664) or ABVD (n = 670). Baseline characteristics were similar between the treatment arms.
Positron emission tomography status after cycle 2 (PET2) was similar between the treatment arms as well. Most patients – 89% of the A+AVD arm and 86% of the ABVD arm – were PET2 negative. Treating physicians used PET2 status as a guide to potentially switch patients to an alternative regimen (radiotherapy or chemotherapy with or without transplant).
In a prior analysis, the study’s primary endpoint was modified PFS (time to progression, death, or noncomplete response after frontline therapy) per an independent review committee (N Engl J Med. 2018;378:331-44). The 2-year modified PFS rate was 82.1% in the A+AVD arm and 77.2% in the ABVD arm (hazard ratio, 0.77; P = .04).
PFS update
In the current analysis, the main exploratory endpoint was PFS per investigator. The 3-year PFS rate was significantly higher in the A+AVD arm than in the ABVD arm – 83.1% and 76.0%, respectively (HR, 0.704; P = .005).
The investigators observed a “consistent improvement in PFS” in the A+AVD arm, regardless of disease stage, International Prognostic score, Eastern Cooperative Oncology Group status, sex, or age. There was a significant improvement in PFS with A+AVD in PET2-negative patients and a trend toward improvement in PET2-positive patients. In the PET2-negative patients, the 3-year PFS was 85.8% in the A+AVD arm and 79.5% in the ABVD arm (HR, 0.69; P = .009). In PET2-positive patients, the 3-year PFS was 67.7% and 51.5%, respectively (HR, 0.59; P = .077).
“These data highlight that A+AVD provides a durable efficacy benefit, compared with ABVD, for frontline stage III/IV cHL [classical Hodgkin lymphoma], which is consistent across key subgroups regardless of patient status at PET2,” Dr. Straus and his colleagues wrote.
Safety update
In both treatment arms, peripheral neuropathy continued to improve or resolve with longer follow-up. Among patients who developed peripheral neuropathy, 78% in the A+AVD arm and 83% in the ABVD arm had improvement or resolution of the condition at 3 years.
Most patients had complete resolution of peripheral neuropathy; 62% in the A+AVD arm and 73% in the ABVD arm. The median time to complete resolution was 28 weeks (range, 0-167 weeks) after stopping A+AVD and 14 weeks (range, 0-188 weeks) after stopping ABVD.
The incidence of secondary malignancies was similar between the treatment arms. There were 14 secondary malignancies in the A+AVD arm (6 solid tumors, 8 hematologic malignancies) and 20 in the ABVD arm (9 solid tumors, 11 hematologic malignancies).
“A+AVD provided a sustained PFS benefit with a predictable and manageable safety profile,” Dr. Straus and colleagues wrote. “These data further support the advantages of A+AVD versus ABVD as frontline treatment of patients with advanced stage III or IV cHL [classical Hodgkin lymphoma].”
The ECHELON-1 trial was sponsored by Millennium Pharmaceuticals (a subsidiary of Takeda) and Seattle Genetics. The investigators disclosed relationships with Millennium, Takeda, Seattle Genetics, and a range of other companies.
SOURCE: Straus DJ et al. Blood. 2020 Jan 16. pii: blood.2019003127. doi: 10.1182/blood.2019003127.
Brentuximab vedotin plus doxorubicin, vinblastine, and dacarbazine (A+AVD) provides “robust, sustained efficacy” in patients with Hodgkin lymphoma, according to investigators.
In the ECHELON-1 trial, investigators compared A+AVD to doxorubicin, bleomycin, vinblastine, and dacarbazine (ABVD) as frontline treatment for stage III or IV Hodgkin lymphoma. The 3-year progression-free survival (PFS) was superior in patients who received A+AVD, and this benefit was seen across most subgroups.
David J. Straus, MD, of Memorial Sloan Kettering Cancer Center in New York and his colleagues detailed these findings in Blood.
The phase 3 trial (NCT01712490) enrolled 1,334 patients with stage III or IV classical Hodgkin lymphoma. They were randomized to receive A+AVD (n = 664) or ABVD (n = 670). Baseline characteristics were similar between the treatment arms.
Positron emission tomography status after cycle 2 (PET2) was similar between the treatment arms as well. Most patients – 89% of the A+AVD arm and 86% of the ABVD arm – were PET2 negative. Treating physicians used PET2 status as a guide to potentially switch patients to an alternative regimen (radiotherapy or chemotherapy with or without transplant).
In a prior analysis, the study’s primary endpoint was modified PFS (time to progression, death, or noncomplete response after frontline therapy) per an independent review committee (N Engl J Med. 2018;378:331-44). The 2-year modified PFS rate was 82.1% in the A+AVD arm and 77.2% in the ABVD arm (hazard ratio, 0.77; P = .04).
PFS update
In the current analysis, the main exploratory endpoint was PFS per investigator. The 3-year PFS rate was significantly higher in the A+AVD arm than in the ABVD arm – 83.1% and 76.0%, respectively (HR, 0.704; P = .005).
The investigators observed a “consistent improvement in PFS” in the A+AVD arm, regardless of disease stage, International Prognostic score, Eastern Cooperative Oncology Group status, sex, or age. There was a significant improvement in PFS with A+AVD in PET2-negative patients and a trend toward improvement in PET2-positive patients. In the PET2-negative patients, the 3-year PFS was 85.8% in the A+AVD arm and 79.5% in the ABVD arm (HR, 0.69; P = .009). In PET2-positive patients, the 3-year PFS was 67.7% and 51.5%, respectively (HR, 0.59; P = .077).
“These data highlight that A+AVD provides a durable efficacy benefit, compared with ABVD, for frontline stage III/IV cHL [classical Hodgkin lymphoma], which is consistent across key subgroups regardless of patient status at PET2,” Dr. Straus and his colleagues wrote.
Safety update
In both treatment arms, peripheral neuropathy continued to improve or resolve with longer follow-up. Among patients who developed peripheral neuropathy, 78% in the A+AVD arm and 83% in the ABVD arm had improvement or resolution of the condition at 3 years.
Most patients had complete resolution of peripheral neuropathy; 62% in the A+AVD arm and 73% in the ABVD arm. The median time to complete resolution was 28 weeks (range, 0-167 weeks) after stopping A+AVD and 14 weeks (range, 0-188 weeks) after stopping ABVD.
The incidence of secondary malignancies was similar between the treatment arms. There were 14 secondary malignancies in the A+AVD arm (6 solid tumors, 8 hematologic malignancies) and 20 in the ABVD arm (9 solid tumors, 11 hematologic malignancies).
“A+AVD provided a sustained PFS benefit with a predictable and manageable safety profile,” Dr. Straus and colleagues wrote. “These data further support the advantages of A+AVD versus ABVD as frontline treatment of patients with advanced stage III or IV cHL [classical Hodgkin lymphoma].”
The ECHELON-1 trial was sponsored by Millennium Pharmaceuticals (a subsidiary of Takeda) and Seattle Genetics. The investigators disclosed relationships with Millennium, Takeda, Seattle Genetics, and a range of other companies.
SOURCE: Straus DJ et al. Blood. 2020 Jan 16. pii: blood.2019003127. doi: 10.1182/blood.2019003127.
FROM BLOOD
FDA okays Palforzia, first drug for peanut allergy in children
The Food and Drug Administration has approved the first drug to combat peanut allergy in children, (Palforzia, Aimmune Therapeutics), although those who take it must continue to avoid peanuts in their diets.
The peanut (Arachis hypogaea) allergen powder is also the first drug ever approved to treat a food allergy. It is not a cure, but it mitigates allergic reactions, including anaphylaxis, that may occur with accidental exposure to peanuts, the FDA said in a news release.
Treatment with the oral powder, which is mixed into semisolid food – such as applesauce or yogurt – can be started in children aged 4 through 17 years who have a confirmed peanut allergy and then continued as a maintenance medication. Some 1 million American children have peanut allergy, and only a fifth will outgrow the allergy, the agency said.
“Because there is no cure, allergic individuals must strictly avoid exposure to prevent severe and potentially life-threatening reactions,” said Peter Marks, MD, PhD, director of the FDA’s Center for Biologics Evaluation and Research, in the statement.
An FDA advisory panel backed the medication in September 2019, but some committee members expressed concern about the large number of children in clinical trials who required epinephrine after receiving a dose of Palforzia.
The initial dose phase is given on a single day, while updosing consists of 11 increasing doses over several months. If the patient tolerates the first administration of an increased dose level, they may continue that dose daily at home. Daily maintenance begins after the completion of all updosing levels.
Palforzia will be available only through specially certified health care providers, health care settings, and pharmacies to patients enrolled in the REMS program, the agency said. Also, the initial dose escalation and first dose of each updosing level can be given only in a certified setting.
The agency said that patients or parents or caregivers must be counseled on the need for constant availability of injectable epinephrine, the need for continued dietary peanut avoidance, and on how to recognize the signs and symptoms of anaphylaxis.
‘Eagerly’ awaited
Palforzia’s effectiveness was based on a randomized, double-blind, placebo-controlled study involving about 500 peanut-allergic individuals that found that 67.2% of allergic patients tolerated an oral challenge with a single 600-mg dose of peanut protein with no more than mild allergic symptoms after 6 months of maintenance treatment, compared with 4% of placebo recipients, the FDA said.
In two double-blind, placebo-controlled studies looking at safety, the most commonly reported side effects among about 700 individuals involved in the research were abdominal pain, vomiting, nausea, tingling in the mouth, itching (including in the mouth and ears), cough, runny nose, throat irritation and tightness, hives, wheezing and shortness of breath, and anaphylaxis.
Palforzia should not be given to those with uncontrolled asthma and can’t be used for emergency treatment of allergic reactions, including anaphylaxis.
“The food allergy community has been eagerly awaiting an FDA-approved treatment that can help mitigate allergic reactions to peanut and, as allergists, we want nothing more than to have a treatment option to offer our patients that has demonstrated both the safety and efficacy to truly impact the lives of patients who live with peanut allergy,” said Christina Ciaccio, MD, chief of Allergy/Immunology and Pediatric Pulmonary Medicine at the University of Chicago Medical Center and Biological Sciences, in a company statement from Aimmune. “With today’s approval of Palforzia, we can – for the first time – offer children and teens with peanut allergy a proven medicine that employs an established therapeutic approach.”
This article first appeared on Medscape.com.
The Food and Drug Administration has approved the first drug to combat peanut allergy in children, (Palforzia, Aimmune Therapeutics), although those who take it must continue to avoid peanuts in their diets.
The peanut (Arachis hypogaea) allergen powder is also the first drug ever approved to treat a food allergy. It is not a cure, but it mitigates allergic reactions, including anaphylaxis, that may occur with accidental exposure to peanuts, the FDA said in a news release.
Treatment with the oral powder, which is mixed into semisolid food – such as applesauce or yogurt – can be started in children aged 4 through 17 years who have a confirmed peanut allergy and then continued as a maintenance medication. Some 1 million American children have peanut allergy, and only a fifth will outgrow the allergy, the agency said.
“Because there is no cure, allergic individuals must strictly avoid exposure to prevent severe and potentially life-threatening reactions,” said Peter Marks, MD, PhD, director of the FDA’s Center for Biologics Evaluation and Research, in the statement.
An FDA advisory panel backed the medication in September 2019, but some committee members expressed concern about the large number of children in clinical trials who required epinephrine after receiving a dose of Palforzia.
The initial dose phase is given on a single day, while updosing consists of 11 increasing doses over several months. If the patient tolerates the first administration of an increased dose level, they may continue that dose daily at home. Daily maintenance begins after the completion of all updosing levels.
Palforzia will be available only through specially certified health care providers, health care settings, and pharmacies to patients enrolled in the REMS program, the agency said. Also, the initial dose escalation and first dose of each updosing level can be given only in a certified setting.
The agency said that patients or parents or caregivers must be counseled on the need for constant availability of injectable epinephrine, the need for continued dietary peanut avoidance, and on how to recognize the signs and symptoms of anaphylaxis.
‘Eagerly’ awaited
Palforzia’s effectiveness was based on a randomized, double-blind, placebo-controlled study involving about 500 peanut-allergic individuals that found that 67.2% of allergic patients tolerated an oral challenge with a single 600-mg dose of peanut protein with no more than mild allergic symptoms after 6 months of maintenance treatment, compared with 4% of placebo recipients, the FDA said.
In two double-blind, placebo-controlled studies looking at safety, the most commonly reported side effects among about 700 individuals involved in the research were abdominal pain, vomiting, nausea, tingling in the mouth, itching (including in the mouth and ears), cough, runny nose, throat irritation and tightness, hives, wheezing and shortness of breath, and anaphylaxis.
Palforzia should not be given to those with uncontrolled asthma and can’t be used for emergency treatment of allergic reactions, including anaphylaxis.
“The food allergy community has been eagerly awaiting an FDA-approved treatment that can help mitigate allergic reactions to peanut and, as allergists, we want nothing more than to have a treatment option to offer our patients that has demonstrated both the safety and efficacy to truly impact the lives of patients who live with peanut allergy,” said Christina Ciaccio, MD, chief of Allergy/Immunology and Pediatric Pulmonary Medicine at the University of Chicago Medical Center and Biological Sciences, in a company statement from Aimmune. “With today’s approval of Palforzia, we can – for the first time – offer children and teens with peanut allergy a proven medicine that employs an established therapeutic approach.”
This article first appeared on Medscape.com.
The Food and Drug Administration has approved the first drug to combat peanut allergy in children, (Palforzia, Aimmune Therapeutics), although those who take it must continue to avoid peanuts in their diets.
The peanut (Arachis hypogaea) allergen powder is also the first drug ever approved to treat a food allergy. It is not a cure, but it mitigates allergic reactions, including anaphylaxis, that may occur with accidental exposure to peanuts, the FDA said in a news release.
Treatment with the oral powder, which is mixed into semisolid food – such as applesauce or yogurt – can be started in children aged 4 through 17 years who have a confirmed peanut allergy and then continued as a maintenance medication. Some 1 million American children have peanut allergy, and only a fifth will outgrow the allergy, the agency said.
“Because there is no cure, allergic individuals must strictly avoid exposure to prevent severe and potentially life-threatening reactions,” said Peter Marks, MD, PhD, director of the FDA’s Center for Biologics Evaluation and Research, in the statement.
An FDA advisory panel backed the medication in September 2019, but some committee members expressed concern about the large number of children in clinical trials who required epinephrine after receiving a dose of Palforzia.
The initial dose phase is given on a single day, while updosing consists of 11 increasing doses over several months. If the patient tolerates the first administration of an increased dose level, they may continue that dose daily at home. Daily maintenance begins after the completion of all updosing levels.
Palforzia will be available only through specially certified health care providers, health care settings, and pharmacies to patients enrolled in the REMS program, the agency said. Also, the initial dose escalation and first dose of each updosing level can be given only in a certified setting.
The agency said that patients or parents or caregivers must be counseled on the need for constant availability of injectable epinephrine, the need for continued dietary peanut avoidance, and on how to recognize the signs and symptoms of anaphylaxis.
‘Eagerly’ awaited
Palforzia’s effectiveness was based on a randomized, double-blind, placebo-controlled study involving about 500 peanut-allergic individuals that found that 67.2% of allergic patients tolerated an oral challenge with a single 600-mg dose of peanut protein with no more than mild allergic symptoms after 6 months of maintenance treatment, compared with 4% of placebo recipients, the FDA said.
In two double-blind, placebo-controlled studies looking at safety, the most commonly reported side effects among about 700 individuals involved in the research were abdominal pain, vomiting, nausea, tingling in the mouth, itching (including in the mouth and ears), cough, runny nose, throat irritation and tightness, hives, wheezing and shortness of breath, and anaphylaxis.
Palforzia should not be given to those with uncontrolled asthma and can’t be used for emergency treatment of allergic reactions, including anaphylaxis.
“The food allergy community has been eagerly awaiting an FDA-approved treatment that can help mitigate allergic reactions to peanut and, as allergists, we want nothing more than to have a treatment option to offer our patients that has demonstrated both the safety and efficacy to truly impact the lives of patients who live with peanut allergy,” said Christina Ciaccio, MD, chief of Allergy/Immunology and Pediatric Pulmonary Medicine at the University of Chicago Medical Center and Biological Sciences, in a company statement from Aimmune. “With today’s approval of Palforzia, we can – for the first time – offer children and teens with peanut allergy a proven medicine that employs an established therapeutic approach.”
This article first appeared on Medscape.com.





