User login
Bariatric Surgery + Medical Therapy: Effective Tx for T2DM?

A 46-year-old woman presents with a BMI of 28, a 4-year history of type 2 diabetes mellitus (T2DM), and an A1C of 9.8%. The patient is currently being treated with intensive medical therapy (IMT), including metformin 2000 mg/d, sitagliptin 100 mg/d, and insulin glargine 12 U/d, with minimal change in A1C. Should you recommend bariatric surgery?
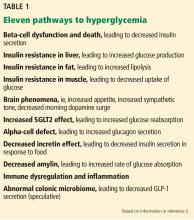
One in 11 Americans has diabetes, and at least 95% of those have T2DM.2,3 The treatment of T2DM is generally multimodal to target the various mechanisms that cause hyperglycemia. Strategies may include making lifestyle modifications, decreasing insulin resistance, increasing insulin secretion, replacing insulin, and targeting incretin-hormonal pathways.
The American Diabetes Association (ADA) recommends diet, exercise, and behavioral modifications as firstline therapy for diabetes management, but these methods are often inadequate.2 In addition to various pharmacotherapeutic strategies for some populations with T2DM, the ADA recommends bariatric surgery for those with a BMI ≥ 35 and uncontrolled hyperglycemia.2,4
However, this recommendation is based only on short-term studies. For example, in a single-center, nonblinded RCT of 60 patients with a BMI ≥ 35, the average baseline A1C levels of 8.65 ± 1.45% were reduced to 7.7 ± 0.6% in the IMT group and to 6.4 ± 1.4% in the gastric-bypass group at 2 years.5 In another study, a randomized double-blind trial involving 60 moderately obese patients (BMI, 25-35), gastric bypass yielded better outcomes than sleeve gastrectomy: 93% of patients in the former group and 47% of those in the latter group achieved remission of T2DM over a 12-month period.6
The current study by Schauer et al examined the long-term outcomes of IMT alone vs bariatric surgery with IMT for the treatment of T2DM in patients who are overweight or obese.1
STUDY SUMMARY
5-year follow-up: surgery + IMT works
This study was a 5-year follow-up of a nonblinded, single-center RCT comparing IMT alone to IMT with Roux-en-Y gastric bypass or sleeve gastrectomy in 150 patients with T2DM.1 Patients were included if they were ages 20 to 60, had a BMI of 27 to 43, and had an A1C > 7%. Patients with a history of bariatric surgery, complex abdominal surgery, or uncontrolled medical or psychiatric disorders were excluded.
Patients were randomly placed in a 1:1:1 fashion into 3 groups: IMT (as defined by the ADA) only, IMT and gastric bypass, or IMT and sleeve gastrectomy. The primary outcome was the number of patients with an A1C ≤ 6%. Secondary outcomes included weight loss, glucose control, lipid levels, blood pressure, medication use, renal function, adverse effects, ophthalmologic outcomes, and quality of life.
Continue to: Of the 150 patients...
Of the 150 patients, 1 died during the follow-up period, leaving 149. Of these, 134 completed the 5-year follow-up. Eight patients in the IMT group and 1 patient in the sleeve gastrectomy group never initiated assigned treatment, and 6 patients were lost to follow-up. One patient from the IMT group and 1 patient from the sleeve gastrectomy group crossed over to the gastric bypass group.
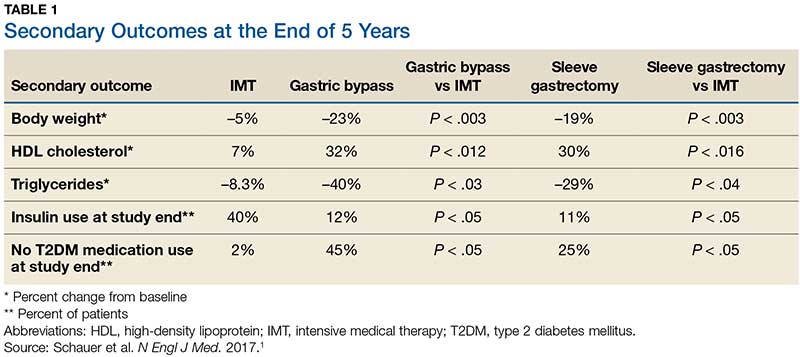
Results. More patients in the bariatric surgery and sleeve gastrectomy groups achieved an A1C of ≤ 6% than in the IMT group (14 of 49 gastric bypass patients, 11 of 47 sleeve gastrectomy patients, and 2 of 38 IMT patients). Compared with those in the IMT group, the patients in the 2 surgery groups showed greater reductions from baseline in body weight and triglyceride levels and greater increases from baseline in HDL cholesterol levels; they also required less antidiabetes medication for glycemic control (see Table).1
WHAT’S NEW?
Big benefits, minimal adverse effects
Prior studies evaluating the effect of gastric bypass surgery on diabetes were observational or had a shorter follow-up duration. This study demonstrates that bariatric surgery plus IMT has long-term benefits with minimal adverse events, compared with IMT alone.1,5 Additionally, this study supports recommendations for bariatric surgery as treatment for T2DM in patients with a BMI ≥ 27, which is below the starting BMI (35) recommended by the ADA.1,4
CAVEATS
Surgery is not without risks
The risk for surgical complications—eg, gastrointestinal bleeding, severe hypoglycemia requiring intervention, and ketoacidosis—in this patient population is significant.1 Other potential complications include gastrointestinal leak, stroke, and infection.1 Additionally, long-term complications from bariatric surgery are emerging and include choledocholithiasis, intestinal obstruction, and esophageal pathology.7 Extensive patient counseling is necessary to ensure that patients make an informed decision regarding surgery.
This study utilized surrogate markers (A1C, lipid levels, and body weight) as disease-oriented outcome measures. Patient-oriented outcomes, such as morbidity and mortality, were not explored in this study.
Continue to: Due to the small sample size...
Due to the small sample size of the study, it is unclear if the outcomes of the 2 surgery groups were significantly different. Patients who underwent gastric bypass surgery had more weight loss and used less diabetes medication at the end of follow-up, compared with patients who underwent sleeve gastrectomy. More information is needed to determine which gastric surgery is preferable for the treatment of T2DM while minimizing adverse effects. However, both of the procedures had outcomes superior to those of IMT, and selection of a particular type of surgery should be a joint decision between the patient and provider.
CHALLENGES TO IMPLEMENTATION
Access and cost may be barriers
The major barriers to implementation are access to, and cost of, bariatric surgery.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center For Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center For Research Resources or the National Institutes of Health.
Copyright © 2019. The Family Physicians Inquiries Network. All rights reserved.
Reprinted with permission from the Family Physicians Inquiries Network and The Journal of Family Practice (2019;68[2]:102-104).
1. Schauer PR, Bhatt DL, Kirwan JP, et al; STAMPEDE Investigators. Bariatric surgery versus intensive medical therapy for diabetes—5-year outcomes. N Engl J Med. 2017;376:641-651.
2. American Diabetes Association. Obesity management for the treatment of type 2 diabetes: standards of medical care in diabetes—2019. Diabetes Care. 2019;42(suppl 1):S81-S89.
3. CDC. National Diabetes Statistics Report, 2017. Atlanta, GA: CDC, US Department of Health and Human Services; 2017. www.cdc.gov/diabetes/pdfs/data/statistics/national-diabetes-statistics-report.pdf. Accessed June 27, 2019.
4. Rubino F, Nathan DM, Eckel RH, et al. Metabolic surgery in the treatment algorithm for type 2 diabetes: a joint statement by international diabetes organizations. Diabetes Care. 2016;39:861-877.
5. Mingrone G, Panunzi S, De Gaetano A, et al. Bariatric surgery versus conventional medical therapy for type 2 diabetes. N Engl J Med. 2012;366:1577-1585.
6. Lee WJ, Chong K, Ser KH, et al. Gastric bypass vs sleeve gastrectomy for type 2 diabetes mellitus: a randomized controlled trial. Arch Surg. 2011; 146:143-148.
7. Schulman AR, Thompson CC. Complications of bariatric surgery: what you can expect to see in your GI practice. Am J Gastroenterol. 2017;112:1640-1655.

A 46-year-old woman presents with a BMI of 28, a 4-year history of type 2 diabetes mellitus (T2DM), and an A1C of 9.8%. The patient is currently being treated with intensive medical therapy (IMT), including metformin 2000 mg/d, sitagliptin 100 mg/d, and insulin glargine 12 U/d, with minimal change in A1C. Should you recommend bariatric surgery?
One in 11 Americans has diabetes, and at least 95% of those have T2DM.2,3 The treatment of T2DM is generally multimodal to target the various mechanisms that cause hyperglycemia. Strategies may include making lifestyle modifications, decreasing insulin resistance, increasing insulin secretion, replacing insulin, and targeting incretin-hormonal pathways.
The American Diabetes Association (ADA) recommends diet, exercise, and behavioral modifications as firstline therapy for diabetes management, but these methods are often inadequate.2 In addition to various pharmacotherapeutic strategies for some populations with T2DM, the ADA recommends bariatric surgery for those with a BMI ≥ 35 and uncontrolled hyperglycemia.2,4
However, this recommendation is based only on short-term studies. For example, in a single-center, nonblinded RCT of 60 patients with a BMI ≥ 35, the average baseline A1C levels of 8.65 ± 1.45% were reduced to 7.7 ± 0.6% in the IMT group and to 6.4 ± 1.4% in the gastric-bypass group at 2 years.5 In another study, a randomized double-blind trial involving 60 moderately obese patients (BMI, 25-35), gastric bypass yielded better outcomes than sleeve gastrectomy: 93% of patients in the former group and 47% of those in the latter group achieved remission of T2DM over a 12-month period.6
The current study by Schauer et al examined the long-term outcomes of IMT alone vs bariatric surgery with IMT for the treatment of T2DM in patients who are overweight or obese.1
STUDY SUMMARY
5-year follow-up: surgery + IMT works
This study was a 5-year follow-up of a nonblinded, single-center RCT comparing IMT alone to IMT with Roux-en-Y gastric bypass or sleeve gastrectomy in 150 patients with T2DM.1 Patients were included if they were ages 20 to 60, had a BMI of 27 to 43, and had an A1C > 7%. Patients with a history of bariatric surgery, complex abdominal surgery, or uncontrolled medical or psychiatric disorders were excluded.
Patients were randomly placed in a 1:1:1 fashion into 3 groups: IMT (as defined by the ADA) only, IMT and gastric bypass, or IMT and sleeve gastrectomy. The primary outcome was the number of patients with an A1C ≤ 6%. Secondary outcomes included weight loss, glucose control, lipid levels, blood pressure, medication use, renal function, adverse effects, ophthalmologic outcomes, and quality of life.
Continue to: Of the 150 patients...
Of the 150 patients, 1 died during the follow-up period, leaving 149. Of these, 134 completed the 5-year follow-up. Eight patients in the IMT group and 1 patient in the sleeve gastrectomy group never initiated assigned treatment, and 6 patients were lost to follow-up. One patient from the IMT group and 1 patient from the sleeve gastrectomy group crossed over to the gastric bypass group.
Results. More patients in the bariatric surgery and sleeve gastrectomy groups achieved an A1C of ≤ 6% than in the IMT group (14 of 49 gastric bypass patients, 11 of 47 sleeve gastrectomy patients, and 2 of 38 IMT patients). Compared with those in the IMT group, the patients in the 2 surgery groups showed greater reductions from baseline in body weight and triglyceride levels and greater increases from baseline in HDL cholesterol levels; they also required less antidiabetes medication for glycemic control (see Table).1

WHAT’S NEW?
Big benefits, minimal adverse effects
Prior studies evaluating the effect of gastric bypass surgery on diabetes were observational or had a shorter follow-up duration. This study demonstrates that bariatric surgery plus IMT has long-term benefits with minimal adverse events, compared with IMT alone.1,5 Additionally, this study supports recommendations for bariatric surgery as treatment for T2DM in patients with a BMI ≥ 27, which is below the starting BMI (35) recommended by the ADA.1,4
CAVEATS
Surgery is not without risks
The risk for surgical complications—eg, gastrointestinal bleeding, severe hypoglycemia requiring intervention, and ketoacidosis—in this patient population is significant.1 Other potential complications include gastrointestinal leak, stroke, and infection.1 Additionally, long-term complications from bariatric surgery are emerging and include choledocholithiasis, intestinal obstruction, and esophageal pathology.7 Extensive patient counseling is necessary to ensure that patients make an informed decision regarding surgery.
This study utilized surrogate markers (A1C, lipid levels, and body weight) as disease-oriented outcome measures. Patient-oriented outcomes, such as morbidity and mortality, were not explored in this study.
Continue to: Due to the small sample size...
Due to the small sample size of the study, it is unclear if the outcomes of the 2 surgery groups were significantly different. Patients who underwent gastric bypass surgery had more weight loss and used less diabetes medication at the end of follow-up, compared with patients who underwent sleeve gastrectomy. More information is needed to determine which gastric surgery is preferable for the treatment of T2DM while minimizing adverse effects. However, both of the procedures had outcomes superior to those of IMT, and selection of a particular type of surgery should be a joint decision between the patient and provider.
CHALLENGES TO IMPLEMENTATION
Access and cost may be barriers
The major barriers to implementation are access to, and cost of, bariatric surgery.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center For Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center For Research Resources or the National Institutes of Health.
Copyright © 2019. The Family Physicians Inquiries Network. All rights reserved.
Reprinted with permission from the Family Physicians Inquiries Network and The Journal of Family Practice (2019;68[2]:102-104).

A 46-year-old woman presents with a BMI of 28, a 4-year history of type 2 diabetes mellitus (T2DM), and an A1C of 9.8%. The patient is currently being treated with intensive medical therapy (IMT), including metformin 2000 mg/d, sitagliptin 100 mg/d, and insulin glargine 12 U/d, with minimal change in A1C. Should you recommend bariatric surgery?
One in 11 Americans has diabetes, and at least 95% of those have T2DM.2,3 The treatment of T2DM is generally multimodal to target the various mechanisms that cause hyperglycemia. Strategies may include making lifestyle modifications, decreasing insulin resistance, increasing insulin secretion, replacing insulin, and targeting incretin-hormonal pathways.
The American Diabetes Association (ADA) recommends diet, exercise, and behavioral modifications as firstline therapy for diabetes management, but these methods are often inadequate.2 In addition to various pharmacotherapeutic strategies for some populations with T2DM, the ADA recommends bariatric surgery for those with a BMI ≥ 35 and uncontrolled hyperglycemia.2,4
However, this recommendation is based only on short-term studies. For example, in a single-center, nonblinded RCT of 60 patients with a BMI ≥ 35, the average baseline A1C levels of 8.65 ± 1.45% were reduced to 7.7 ± 0.6% in the IMT group and to 6.4 ± 1.4% in the gastric-bypass group at 2 years.5 In another study, a randomized double-blind trial involving 60 moderately obese patients (BMI, 25-35), gastric bypass yielded better outcomes than sleeve gastrectomy: 93% of patients in the former group and 47% of those in the latter group achieved remission of T2DM over a 12-month period.6
The current study by Schauer et al examined the long-term outcomes of IMT alone vs bariatric surgery with IMT for the treatment of T2DM in patients who are overweight or obese.1
STUDY SUMMARY
5-year follow-up: surgery + IMT works
This study was a 5-year follow-up of a nonblinded, single-center RCT comparing IMT alone to IMT with Roux-en-Y gastric bypass or sleeve gastrectomy in 150 patients with T2DM.1 Patients were included if they were ages 20 to 60, had a BMI of 27 to 43, and had an A1C > 7%. Patients with a history of bariatric surgery, complex abdominal surgery, or uncontrolled medical or psychiatric disorders were excluded.
Patients were randomly placed in a 1:1:1 fashion into 3 groups: IMT (as defined by the ADA) only, IMT and gastric bypass, or IMT and sleeve gastrectomy. The primary outcome was the number of patients with an A1C ≤ 6%. Secondary outcomes included weight loss, glucose control, lipid levels, blood pressure, medication use, renal function, adverse effects, ophthalmologic outcomes, and quality of life.
Continue to: Of the 150 patients...
Of the 150 patients, 1 died during the follow-up period, leaving 149. Of these, 134 completed the 5-year follow-up. Eight patients in the IMT group and 1 patient in the sleeve gastrectomy group never initiated assigned treatment, and 6 patients were lost to follow-up. One patient from the IMT group and 1 patient from the sleeve gastrectomy group crossed over to the gastric bypass group.
Results. More patients in the bariatric surgery and sleeve gastrectomy groups achieved an A1C of ≤ 6% than in the IMT group (14 of 49 gastric bypass patients, 11 of 47 sleeve gastrectomy patients, and 2 of 38 IMT patients). Compared with those in the IMT group, the patients in the 2 surgery groups showed greater reductions from baseline in body weight and triglyceride levels and greater increases from baseline in HDL cholesterol levels; they also required less antidiabetes medication for glycemic control (see Table).1

WHAT’S NEW?
Big benefits, minimal adverse effects
Prior studies evaluating the effect of gastric bypass surgery on diabetes were observational or had a shorter follow-up duration. This study demonstrates that bariatric surgery plus IMT has long-term benefits with minimal adverse events, compared with IMT alone.1,5 Additionally, this study supports recommendations for bariatric surgery as treatment for T2DM in patients with a BMI ≥ 27, which is below the starting BMI (35) recommended by the ADA.1,4
CAVEATS
Surgery is not without risks
The risk for surgical complications—eg, gastrointestinal bleeding, severe hypoglycemia requiring intervention, and ketoacidosis—in this patient population is significant.1 Other potential complications include gastrointestinal leak, stroke, and infection.1 Additionally, long-term complications from bariatric surgery are emerging and include choledocholithiasis, intestinal obstruction, and esophageal pathology.7 Extensive patient counseling is necessary to ensure that patients make an informed decision regarding surgery.
This study utilized surrogate markers (A1C, lipid levels, and body weight) as disease-oriented outcome measures. Patient-oriented outcomes, such as morbidity and mortality, were not explored in this study.
Continue to: Due to the small sample size...
Due to the small sample size of the study, it is unclear if the outcomes of the 2 surgery groups were significantly different. Patients who underwent gastric bypass surgery had more weight loss and used less diabetes medication at the end of follow-up, compared with patients who underwent sleeve gastrectomy. More information is needed to determine which gastric surgery is preferable for the treatment of T2DM while minimizing adverse effects. However, both of the procedures had outcomes superior to those of IMT, and selection of a particular type of surgery should be a joint decision between the patient and provider.
CHALLENGES TO IMPLEMENTATION
Access and cost may be barriers
The major barriers to implementation are access to, and cost of, bariatric surgery.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center For Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center For Research Resources or the National Institutes of Health.
Copyright © 2019. The Family Physicians Inquiries Network. All rights reserved.
Reprinted with permission from the Family Physicians Inquiries Network and The Journal of Family Practice (2019;68[2]:102-104).
1. Schauer PR, Bhatt DL, Kirwan JP, et al; STAMPEDE Investigators. Bariatric surgery versus intensive medical therapy for diabetes—5-year outcomes. N Engl J Med. 2017;376:641-651.
2. American Diabetes Association. Obesity management for the treatment of type 2 diabetes: standards of medical care in diabetes—2019. Diabetes Care. 2019;42(suppl 1):S81-S89.
3. CDC. National Diabetes Statistics Report, 2017. Atlanta, GA: CDC, US Department of Health and Human Services; 2017. www.cdc.gov/diabetes/pdfs/data/statistics/national-diabetes-statistics-report.pdf. Accessed June 27, 2019.
4. Rubino F, Nathan DM, Eckel RH, et al. Metabolic surgery in the treatment algorithm for type 2 diabetes: a joint statement by international diabetes organizations. Diabetes Care. 2016;39:861-877.
5. Mingrone G, Panunzi S, De Gaetano A, et al. Bariatric surgery versus conventional medical therapy for type 2 diabetes. N Engl J Med. 2012;366:1577-1585.
6. Lee WJ, Chong K, Ser KH, et al. Gastric bypass vs sleeve gastrectomy for type 2 diabetes mellitus: a randomized controlled trial. Arch Surg. 2011; 146:143-148.
7. Schulman AR, Thompson CC. Complications of bariatric surgery: what you can expect to see in your GI practice. Am J Gastroenterol. 2017;112:1640-1655.
1. Schauer PR, Bhatt DL, Kirwan JP, et al; STAMPEDE Investigators. Bariatric surgery versus intensive medical therapy for diabetes—5-year outcomes. N Engl J Med. 2017;376:641-651.
2. American Diabetes Association. Obesity management for the treatment of type 2 diabetes: standards of medical care in diabetes—2019. Diabetes Care. 2019;42(suppl 1):S81-S89.
3. CDC. National Diabetes Statistics Report, 2017. Atlanta, GA: CDC, US Department of Health and Human Services; 2017. www.cdc.gov/diabetes/pdfs/data/statistics/national-diabetes-statistics-report.pdf. Accessed June 27, 2019.
4. Rubino F, Nathan DM, Eckel RH, et al. Metabolic surgery in the treatment algorithm for type 2 diabetes: a joint statement by international diabetes organizations. Diabetes Care. 2016;39:861-877.
5. Mingrone G, Panunzi S, De Gaetano A, et al. Bariatric surgery versus conventional medical therapy for type 2 diabetes. N Engl J Med. 2012;366:1577-1585.
6. Lee WJ, Chong K, Ser KH, et al. Gastric bypass vs sleeve gastrectomy for type 2 diabetes mellitus: a randomized controlled trial. Arch Surg. 2011; 146:143-148.
7. Schulman AR, Thompson CC. Complications of bariatric surgery: what you can expect to see in your GI practice. Am J Gastroenterol. 2017;112:1640-1655.
Hyperthyroid? She sure doesn’t look like it
A 50-year-old woman returns for routine follow-up. She has a 15-year history of hypothyroidism, pernicious anemia, and celiac disease. She has had some recent abdominal pain, but no changes in her bowel patterns, and she has not experienced any problems with chest pain, palpitations, or weakness recently. The only medication she is taking is levothyroxine 125 mcg. She has reported no recent weight loss, her blood pressure is 100/60 mm Hg, pulse is 66 beats per minute, temperature is 36.8 degrees Celsius, body mass index is 20, and she does not have a neck goiter. Her cardiac exam was normal and her neurological exam revealed no tremor. Her lab for thyroid-stimulating hormone (TSH) was less than 0.03, and her lab for free thyroxine (FT4) was 2.2, while her TSH level had been 1.4 a year ago. Her levothyroxine dose was decreased to 100 mcg/day, and her repeat lab for TSH, which occurred 12 weeks later, was still less than 0.03. What is the best explanation for why this patient’s labs look like hyperthyroidism, but this patient clinically does not appear to have hyperthyroidism?
A) She was initially given too much levothyroxine; her TSH response is lagging to dose reduction.
B) She has Graves’ disease.
C) She has acute thyroiditis.
D) She is taking extra thyroid hormone.
E) She is taking biotin.
This patient has a history that includes multiple autoimmune diseases including hypothyroidism. It would be extremely unlikely that she would develop Graves' disease or develop acute thyroiditis in the setting of a gland that has been underfunctioning for years. She has no symptoms suggesting that she has hyperthyroidism, which makes taking more thyroid hormone than she is reporting less likely, although this could be possible. The TSH response can lag after dose adjustments of thyroid, but usually a 6-week interval is adequate. This patient’s testing was done 12 weeks after dose reduction making this very unlikely.
The cause for the labs that look like hyperthyroidism in this patient who appears clinically euthyroid is that she is taking biotin. Biotin (vitamin B7) has become a very popular supplement in the past few years for thin hair, brittle nails, and fatigue. The RDA for biotin is 30 mcg. It is widely available in high doses – 5,000-10,000 mcg – which are common doses for supplements.
Biotin has been used extensively as a key component of immunoassays. Streptavidin, a protein produced by the bacteria Streptomyces avidinii, binds biotin with an extremely high affinity, and this binding is utilized in a number of immunoassays, including the assays for thyroid hormone and TSH.1
High serum levels of biotin can make the assays inaccurate, with lower-than-actual TSH and higher-than-actual thyroid hormone levels. Multiple case reports have documented this happening clinically.1-3 I personally saw a case of this recently in my practice. Katzman and colleagues looked at the prevalence of biotin use in outpatients.4 They found that 7.7% were taking supplemental biotin, while 7.4% had levels of biotin in serum samples that were at a level that could interfere with biotin-based serum assays.
Theoretically, biotin can affect multiple other assays that use the streptavidin-biotin assay. The most concerning of these potential problems is with troponin assays. Biotin can falsely lower troponin assays and this can lead to missing the diagnosis of cardiac injury. The Food and Drug Administration released a warning about this and other biotin lab interactions in November 2017.5 Several studies have demonstrated that this effect can occur at serum levels achievable with available over-the-counter doses of biotin.6,7
Not all troponin assays are affected by high serum levels of biotin: The Gen 5 cTnT assay is the only troponin assay affected.7 I could not find any case reports that have been published where biotin had caused a clinical missed diagnosis with troponins.
Pearls
Consider biotin supplement use when you have patients whose labs look like hyperthyroidism, but clinically do not appear to be hyperthyroid.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and serves as third-year medical student clerkship director at the University of Washington. Contact Dr. Paauw at [email protected].
References
1. Charles S, Agrawal N, and Blum M. Erroneous thyroid diagnosis due to over-the-counter biotin. Nutrition 2019;57:257-8.
2. Elston MS et al. Factitious Graves’ disease due to a biotin immunoassay interference – a case and review of the literature. J Clin Endocrinol Metab 2016;101:3251-5.
3. Barbesino G. Misdiagnosis of Graves ’disease with apparent severe hyperthyroidism in a patient taking biotin megadoses. Thyroid 2016;26(6):860-3.
4. Katzman et al. Prevalence of biotin supplement usage in outpatients and plasma biotin concentrations in patients presenting to the emergency department. Clin Biochem. 2018 Sep;60:11-16.
5. “The FDA Warns that Biotin May Interfere with Lab Tests: FDA Safety Communication,” Nov. 28, 2017.
6. Trambas et al. Characterization of the scope and magnitude of biotin interference in susceptible Roche Elecsys competitive and sandwich immunoassays. Ann Clin Biochem. 2018 Mar;55(2):205-15.
7. Frame IJ et al. Susceptibility of cardiac troponin assays to biotin interference. Am J Clin Pathol. 2019 Apr 2;151(5):486-93.
A 50-year-old woman returns for routine follow-up. She has a 15-year history of hypothyroidism, pernicious anemia, and celiac disease. She has had some recent abdominal pain, but no changes in her bowel patterns, and she has not experienced any problems with chest pain, palpitations, or weakness recently. The only medication she is taking is levothyroxine 125 mcg. She has reported no recent weight loss, her blood pressure is 100/60 mm Hg, pulse is 66 beats per minute, temperature is 36.8 degrees Celsius, body mass index is 20, and she does not have a neck goiter. Her cardiac exam was normal and her neurological exam revealed no tremor. Her lab for thyroid-stimulating hormone (TSH) was less than 0.03, and her lab for free thyroxine (FT4) was 2.2, while her TSH level had been 1.4 a year ago. Her levothyroxine dose was decreased to 100 mcg/day, and her repeat lab for TSH, which occurred 12 weeks later, was still less than 0.03. What is the best explanation for why this patient’s labs look like hyperthyroidism, but this patient clinically does not appear to have hyperthyroidism?
A) She was initially given too much levothyroxine; her TSH response is lagging to dose reduction.
B) She has Graves’ disease.
C) She has acute thyroiditis.
D) She is taking extra thyroid hormone.
E) She is taking biotin.
This patient has a history that includes multiple autoimmune diseases including hypothyroidism. It would be extremely unlikely that she would develop Graves' disease or develop acute thyroiditis in the setting of a gland that has been underfunctioning for years. She has no symptoms suggesting that she has hyperthyroidism, which makes taking more thyroid hormone than she is reporting less likely, although this could be possible. The TSH response can lag after dose adjustments of thyroid, but usually a 6-week interval is adequate. This patient’s testing was done 12 weeks after dose reduction making this very unlikely.
The cause for the labs that look like hyperthyroidism in this patient who appears clinically euthyroid is that she is taking biotin. Biotin (vitamin B7) has become a very popular supplement in the past few years for thin hair, brittle nails, and fatigue. The RDA for biotin is 30 mcg. It is widely available in high doses – 5,000-10,000 mcg – which are common doses for supplements.
Biotin has been used extensively as a key component of immunoassays. Streptavidin, a protein produced by the bacteria Streptomyces avidinii, binds biotin with an extremely high affinity, and this binding is utilized in a number of immunoassays, including the assays for thyroid hormone and TSH.1
High serum levels of biotin can make the assays inaccurate, with lower-than-actual TSH and higher-than-actual thyroid hormone levels. Multiple case reports have documented this happening clinically.1-3 I personally saw a case of this recently in my practice. Katzman and colleagues looked at the prevalence of biotin use in outpatients.4 They found that 7.7% were taking supplemental biotin, while 7.4% had levels of biotin in serum samples that were at a level that could interfere with biotin-based serum assays.
Theoretically, biotin can affect multiple other assays that use the streptavidin-biotin assay. The most concerning of these potential problems is with troponin assays. Biotin can falsely lower troponin assays and this can lead to missing the diagnosis of cardiac injury. The Food and Drug Administration released a warning about this and other biotin lab interactions in November 2017.5 Several studies have demonstrated that this effect can occur at serum levels achievable with available over-the-counter doses of biotin.6,7
Not all troponin assays are affected by high serum levels of biotin: The Gen 5 cTnT assay is the only troponin assay affected.7 I could not find any case reports that have been published where biotin had caused a clinical missed diagnosis with troponins.
Pearls
Consider biotin supplement use when you have patients whose labs look like hyperthyroidism, but clinically do not appear to be hyperthyroid.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and serves as third-year medical student clerkship director at the University of Washington. Contact Dr. Paauw at [email protected].
References
1. Charles S, Agrawal N, and Blum M. Erroneous thyroid diagnosis due to over-the-counter biotin. Nutrition 2019;57:257-8.
2. Elston MS et al. Factitious Graves’ disease due to a biotin immunoassay interference – a case and review of the literature. J Clin Endocrinol Metab 2016;101:3251-5.
3. Barbesino G. Misdiagnosis of Graves ’disease with apparent severe hyperthyroidism in a patient taking biotin megadoses. Thyroid 2016;26(6):860-3.
4. Katzman et al. Prevalence of biotin supplement usage in outpatients and plasma biotin concentrations in patients presenting to the emergency department. Clin Biochem. 2018 Sep;60:11-16.
5. “The FDA Warns that Biotin May Interfere with Lab Tests: FDA Safety Communication,” Nov. 28, 2017.
6. Trambas et al. Characterization of the scope and magnitude of biotin interference in susceptible Roche Elecsys competitive and sandwich immunoassays. Ann Clin Biochem. 2018 Mar;55(2):205-15.
7. Frame IJ et al. Susceptibility of cardiac troponin assays to biotin interference. Am J Clin Pathol. 2019 Apr 2;151(5):486-93.
A 50-year-old woman returns for routine follow-up. She has a 15-year history of hypothyroidism, pernicious anemia, and celiac disease. She has had some recent abdominal pain, but no changes in her bowel patterns, and she has not experienced any problems with chest pain, palpitations, or weakness recently. The only medication she is taking is levothyroxine 125 mcg. She has reported no recent weight loss, her blood pressure is 100/60 mm Hg, pulse is 66 beats per minute, temperature is 36.8 degrees Celsius, body mass index is 20, and she does not have a neck goiter. Her cardiac exam was normal and her neurological exam revealed no tremor. Her lab for thyroid-stimulating hormone (TSH) was less than 0.03, and her lab for free thyroxine (FT4) was 2.2, while her TSH level had been 1.4 a year ago. Her levothyroxine dose was decreased to 100 mcg/day, and her repeat lab for TSH, which occurred 12 weeks later, was still less than 0.03. What is the best explanation for why this patient’s labs look like hyperthyroidism, but this patient clinically does not appear to have hyperthyroidism?
A) She was initially given too much levothyroxine; her TSH response is lagging to dose reduction.
B) She has Graves’ disease.
C) She has acute thyroiditis.
D) She is taking extra thyroid hormone.
E) She is taking biotin.
This patient has a history that includes multiple autoimmune diseases including hypothyroidism. It would be extremely unlikely that she would develop Graves' disease or develop acute thyroiditis in the setting of a gland that has been underfunctioning for years. She has no symptoms suggesting that she has hyperthyroidism, which makes taking more thyroid hormone than she is reporting less likely, although this could be possible. The TSH response can lag after dose adjustments of thyroid, but usually a 6-week interval is adequate. This patient’s testing was done 12 weeks after dose reduction making this very unlikely.
The cause for the labs that look like hyperthyroidism in this patient who appears clinically euthyroid is that she is taking biotin. Biotin (vitamin B7) has become a very popular supplement in the past few years for thin hair, brittle nails, and fatigue. The RDA for biotin is 30 mcg. It is widely available in high doses – 5,000-10,000 mcg – which are common doses for supplements.
Biotin has been used extensively as a key component of immunoassays. Streptavidin, a protein produced by the bacteria Streptomyces avidinii, binds biotin with an extremely high affinity, and this binding is utilized in a number of immunoassays, including the assays for thyroid hormone and TSH.1
High serum levels of biotin can make the assays inaccurate, with lower-than-actual TSH and higher-than-actual thyroid hormone levels. Multiple case reports have documented this happening clinically.1-3 I personally saw a case of this recently in my practice. Katzman and colleagues looked at the prevalence of biotin use in outpatients.4 They found that 7.7% were taking supplemental biotin, while 7.4% had levels of biotin in serum samples that were at a level that could interfere with biotin-based serum assays.
Theoretically, biotin can affect multiple other assays that use the streptavidin-biotin assay. The most concerning of these potential problems is with troponin assays. Biotin can falsely lower troponin assays and this can lead to missing the diagnosis of cardiac injury. The Food and Drug Administration released a warning about this and other biotin lab interactions in November 2017.5 Several studies have demonstrated that this effect can occur at serum levels achievable with available over-the-counter doses of biotin.6,7
Not all troponin assays are affected by high serum levels of biotin: The Gen 5 cTnT assay is the only troponin assay affected.7 I could not find any case reports that have been published where biotin had caused a clinical missed diagnosis with troponins.
Pearls
Consider biotin supplement use when you have patients whose labs look like hyperthyroidism, but clinically do not appear to be hyperthyroid.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and serves as third-year medical student clerkship director at the University of Washington. Contact Dr. Paauw at [email protected].
References
1. Charles S, Agrawal N, and Blum M. Erroneous thyroid diagnosis due to over-the-counter biotin. Nutrition 2019;57:257-8.
2. Elston MS et al. Factitious Graves’ disease due to a biotin immunoassay interference – a case and review of the literature. J Clin Endocrinol Metab 2016;101:3251-5.
3. Barbesino G. Misdiagnosis of Graves ’disease with apparent severe hyperthyroidism in a patient taking biotin megadoses. Thyroid 2016;26(6):860-3.
4. Katzman et al. Prevalence of biotin supplement usage in outpatients and plasma biotin concentrations in patients presenting to the emergency department. Clin Biochem. 2018 Sep;60:11-16.
5. “The FDA Warns that Biotin May Interfere with Lab Tests: FDA Safety Communication,” Nov. 28, 2017.
6. Trambas et al. Characterization of the scope and magnitude of biotin interference in susceptible Roche Elecsys competitive and sandwich immunoassays. Ann Clin Biochem. 2018 Mar;55(2):205-15.
7. Frame IJ et al. Susceptibility of cardiac troponin assays to biotin interference. Am J Clin Pathol. 2019 Apr 2;151(5):486-93.
Giant cell arteritis: An updated review of an old disease
Giant cell arteritis (GCA) is a systemic vasculitis involving medium-sized and large arteries, most commonly the temporal, ophthalmic, occipital, vertebral, posterior ciliary, and proximal vertebral arteries. Moreover, involvement of the ophthalmic artery and its branches results in loss of vision. GCA can also involve the aorta and its proximal branches, especially in the upper extremities.
GCA is the most common systemic vasculitis in adults. It occurs almost exclusively in patients over age 50 and affects women more than men. It is most frequent in populations of northern European ancestry, especially Scandinavian. In a retrospective cohort study in Norway, the average annual cumulative incidence rate of GCA was 16.7 per 100,000 people over age 50.1 Risk factors include older age, history of smoking, current smoking, early menopause, and, possibly, stress-related disorders.2
PATHOGENESIS IS NOT COMPLETELY UNDERSTOOD
The pathogenesis of GCA is not completely understood, but there is evidence of immune activation in the arterial wall leading to activation of macrophages and formation of multinucleated giant cells (which may not always be present in biopsies).
The most relevant cytokines in the ongoing pathogenesis are still being defined, but the presence of interferon gamma and interleukin 6 (IL-6) seem to be critical for the expression of the disease. The primary immunogenic triggers for the elaboration of these cytokines and the arteritis remain elusive.
A SPECTRUM OF PRESENTATIONS
The initial symptoms of GCA may be vague, such as malaise, fever, and night sweats, and are likely due to systemic inflammation. Features of vascular involvement include headache, scalp tenderness, and jaw claudication (cramping pain in the jaw while chewing).
A less common but serious feature associated with GCA is partial or complete vision loss affecting 1 or both eyes.3 Some patients suddenly go completely blind without any visual prodrome.
Overlapping GCA phenotypes exist, with a spectrum of presentations that include classic cranial arteritis, extracranial GCA (also called large-vessel GCA), and polymyalgia rheumatica.2
Cranial GCA, the best-characterized clinical presentation, causes symptoms such as headache or signs such as tenderness of the temporal artery. On examination, the temporal arteries may be tender or nodular, and the pulses may be felt above the zygomatic arch, above and in front of the tragus of the ear. About two-thirds of patients with cranial GCA present with new-onset headache, most often in the temporal area, but possibly anywhere throughout the head.
Visual disturbance, jaw claudication, and tongue pain are less common but, if present, increase the likelihood of this diagnosis.2
Large-vessel involvement in GCA is common and refers to involvement of the aorta and its proximal branches. Imaging methods used in diagnosing large-vessel GCA include color Doppler ultrasonography, computed tomography with angiography, magnetic resonance imaging with angiography, and positron emission tomography. In some centers, such imaging is performed in all patients diagnosed with GCA to survey for large-vessel involvement.
Depending on the imaging study, large-vessel involvement has been found in 30% to 80% of cases of GCA.4,5 It is often associated with nonspecific symptoms such as fever, weight loss, chills, and malaise, but it can also cause more specific symptoms such as unilateral extremity claudication. In contrast to patients with cranial GCA, patients with large-vessel GCA were younger at onset, less likely to have headaches, and more likely to have arm claudication at presentation.6 Aortitis of the ascending aorta can occur with a histopathologic pattern of GCA but without the clinical stigmata of GCA.
The finding of aortitis should prompt the clinician to question the patient about other symptoms of GCA and to order imaging of the whole vascular tree. Ultrasonography and biopsy of the temporal arteries can be considered. Whether idiopathic aortitis is part of the GCA spectrum remains to be seen.
Laboratory tests often show anemia, leukocytosis, and thrombocytosis. Acute-phase reactants such as C-reactive protein and the erythrocyte sedimentation rate are often elevated. The sedimentation rate often exceeds 50 mm/hour and sometimes 100 mm/hour.
In 2 retrospective studies, the number of patients with GCA whose sedimentation rate was less than 50 mm/hour ranged between 5% and 11%.7,8 However, a small percentage of patients with GCA have normal inflammatory markers. Therefore, if the suspicion for GCA is high, treatment should be started and biopsy pursued.9 In patients with paraproteinemia or other causes of a spuriously elevated or low erythrocyte sedimentation rate, C-reactive protein is a more reliable test.
Polymyalgia rheumatica is another rheumatologic condition that can occur independently or in conjunction with GCA. It is characterized by stiffness and pain in the proximal joints such as the hips and shoulders, typically worse in the morning and better with activity. Although the patient may subjectively feel weak, a close neurologic examination will reveal normal muscle strength.
Polymyalgia rheumatica is observed in 40% to 60% of patients with GCA at the time of diagnosis; 16% to 21% of patients with polymyalgia rheumatica may develop GCA, especially if untreated.2,10
Differential diagnosis
Other vasculitides (eg, Takayasu arteritis) can also present with unexplained fever, anemia, and constitutional symptoms.
Infection should be considered if fever is present. An infectious disease accompanied by fever, headache, and elevated inflammatory markers can mimic GCA.
Nonarteritic anterior ischemic optic neuropathy can present with sudden vision loss, prompting concern for underlying GCA. Risk factors include hypertension and diabetes mellitus; other features of GCA, including elevated inflammatory markers, are generally absent.
TEMPORAL ARTERY BIOPSY: THE GOLD STANDARD FOR DIAGNOSIS
Temporal artery biopsy remains the standard to confirm the diagnosis. However, because inflammation in the temporal arteries can affect some segments but not others, biopsy results on conventional hematoxylin and eosin staining can be falsely negative in patients with GCA. In one study,11 the mean sensitivity of unilateral temporal artery biopsy was 86.9%.
Typical positive histologic findings are inflammation with panarteritis, CD4-positive lymphocytes, macrophages, giant cells, and fragmentation of the internal elastic lamina.12
When GCA is suspected, treatment with glucocorticoids should be started immediately and biopsy performed as soon as possible. Delaying biopsy for 14 days or more may not affect the accuracy of biopsy study.13 Treatment should never be withheld while awaiting the results of biopsy study.
Biopsy is usually performed unilaterally, on the same side as the symptoms or abnormal findings on examination. Bilateral temporal artery biopsy is also performed and compared with unilateral biopsy; this approach increases the diagnostic yield by about 5%.14
IMAGING
In patients with suspected GCA, imaging is recommended early to complement the clinical criteria for the diagnosis of GCA.15 Positron emission tomography, computed tomography angiography, magnetic resonance angiography, or Doppler ultrasonography can reveal inflammation of the arteries in the proximal upper or lower limbs or the aorta.2
In patients with suspected cranial GCA, ultrasonography of the temporal and axillary arteries is recommended first. If ultrasonography is not available or is inconclusive, high-resolution magnetic resonance imaging of the cranial arteries can be used as an alternative. Computed tomography and positron emission tomography of the cranial arteries are not recommended.
In patients with suspected large-vessel GCA, ultrasonography, positron emission tomography, computed tomography, and magnetic resonance imaging may be used to screen for vessel wall inflammation, edema, and luminal narrowing in extracranial arteries. Ultrasonography is of limited value in assessing aortitis.
Color duplex ultrasonography can be applied to assess for vascular inflammation of the temporal or large arteries. The typical finding of the “halo” sign, a hypoechoic ring around the arterial lumen, represents the inflammation-induced thickening of the arterial wall. The “compression sign,” the persistence of the “halo” during compression of the vessel lumen by the ultrasound probe, has high specificity for the diagnosis.16
Ultrasonography of suspected GCA has yielded sensitivities of 55% to 100% and specificities of 78% to 100%. However, its sensitivity depends on the user’s level of expertise, so it should be done only in medical centers with a high number of GCA cases and with highly experienced sonographers. High-resolution magnetic resonance imaging is an alternative to ultrasonography and has shown similar sensitivity and specificity.3
TREATMENT WITH GLUCOCORTICOIDS
Glucocorticoids remain the standard for treatment of GCA. The therapeutic effect of glucocorticoids in GCA has been established by years of clinical experience, but has never been proven in a placebo-controlled trial. When started appropriately and expeditiously, glucocorticoids produce exquisite resolution of signs and symptoms and prevent the serious complication of vision loss. Rapid resolution of symptoms is so typical of GCA that if the patient’s symptoms persist more than a few days after starting a glucocorticoid, the diagnosis of GCA should be reconsidered.
In a retrospective study of 245 patients with biopsy-proven GCA treated with glucocorticoids, 34 had permanent loss of sight.17 In 32 (94%) of the 34, the vision loss occurred before glucocorticoids were started. Of the remaining 2 patients, 1 lost vision 8 days into treatment, and the other lost vision 3 years after diagnosis and 1 year after discontinuation of glucocorticoids.
In a series of 144 patients with biopsy-proven GCA, 51 had no vision loss at presentation and no vision loss after starting glucocorticoids, and 93 had vision loss at presentation. In the latter group, symptoms worsened within 5 days of starting glucocorticoids in 9 patients.18 If vision was intact at the time of presentation, prompt initiation of glucocorticoids reduced the risk of vision loss to less than 1%.
High doses, slowly tapered
The European League Against Rheumatism recommends early initiation of high-dose glucocorticoids for patients with large-vessel vasculitis,19 and it also recommends glucocorticoids for patients with polymyalgia rheumatica.20 The optimal initial and tapering dosage has never been formally evaluated, but regimens have been devised on the basis of expert opinion.21
For patients with GCA who do not have vision loss at the time of diagnosis, the initial dose is prednisone 1 mg/kg or its equivalent daily for 2 to 4 weeks, after which it is tapered.21 If the initial dosage is prednisone 60 mg orally daily for 2 to 4 weeks, our practice is to taper it to 50 mg daily for 2 weeks, then 40 mg daily for 2 weeks. Then, it is decreased by 5 mg every 2 weeks until it is 20 mg daily, and then by 2.5 mg every 2 weeks until it is 10 mg orally daily. Thereafter, the dosage is decreased by 1 mg every 2 to 4 weeks.
For patients with GCA who experience transient vision loss or diplopia at the time of diagnosis, intravenous pulse glucocorticoid therapy should be initiated to reduce the risk of vision loss as rapidly as possible.22 A typical pulse regimen is methylprednisolone 1 g intravenously daily for 3 days. Though not rigorously validated in studies, such an approach is used to avoid vision impairment due to GCA, which is rarely reversible.
RELAPSE OF DISEASE
Suspect a relapse of GCA if the patient’s initial symptoms recur, if inflammatory markers become elevated, or if classic symptoms of GCA or polymyalgia rheumatica occur. Elevations in inflammatory markers do not definitely indicate a flare of GCA, but they should trigger close monitoring of the patient’s symptoms.
Relapse is treated by increasing the glucocorticoid dosage as appropriate to the nature of the relapse. If vision is affected or the patient has symptoms of GCA, then increments of 30 to 60 mg of prednisone are warranted, whereas if the patient has symptoms of polymyalgia rheumatica, then increments of 5 to 10 mg of prednisone are usually used.
The incidence of relapses of GCA in multiple tertiary care centers has been reported to vary between 34% and 75%.23,24 Most relapses occur at prednisone dosages of less than 20 mg orally daily and within the first year after diagnosis. The most common symptoms are limb ischemia, jaw claudication, constitutional symptoms, headaches, and polymyalgia rheumatica. In a review of 286 patients,25 213 (74%) had at least 1 relapse. The first relapse occurred in the first year in 50%, by 2 years in 68%, and by 5 years in 79%.
ADVERSE EFFECTS OF GLUCOCORTICOIDS
In high doses, glucocorticoids have well-known adverse effects. In a population-based study of 120 patients, each patient treated with glucocorticoids experienced at least 1 adverse effect (cataract, fracture, infection, osteonecrosis, diabetes, hypertension, weight gain, capillary fragility, or hair loss).26 The effects were related to aging and cumulative dosage of prednisone but not to the initial dosage.
Glucocorticoids can affect many organs and systems:
- Eyes (cataracts, increased intraocular pressure, exophthalmos)
- Heart (premature atherosclerotic disease, hypertension, fluid retention, hyperlipidemia, arrhythmias)
- Gastrointestinal system (ulcer, gastrointestinal bleeding, gastritis, visceral perforation, hepatic steatosis, acute pancreatitis)
- Bone and muscle (osteopenia, osteoporosis, osteonecrosis, myopathy)
- Brain (mood disorder, psychosis, memory impairment)
- Endocrine system (hyperglycemia, hypothalamic-pituitary-adrenal axis suppression)
- Immune system (immunosuppression, leading to infection and leukocytosis).
Patients receiving a glucocorticoid dose equivalent to 20 mg or more of prednisone daily for 1 month or more who also have another cause of immunocompromise need prophylaxis against Pneumocystis jirovecii pneumonia.27 They should also receive appropriate immunizations before starting glucocorticoids. Live-virus vaccines should not be given to these patients until they have been off glucocorticoids for 1 month.
Glucocorticoids and bone loss
Glucocorticoids are associated with bone loss and fracture, which can occur within the first few months of use and with dosages as low as 2.5 to 7.5 mg orally daily.28 Therefore, glucocorticoid-induced bone loss has to be treated aggressively, particularly in patients who are older and have a history of fragility fracture.
For patients with GCA who need glucocorticoids in doses greater than 5 mg orally daily for more than 3 months, the following measures are advised to decrease the risk of bone loss:
- Weight-bearing exercise
- Smoking cessation
- Moderation in alcohol intake
- Measures to prevent falls29
- Supplementation with 1,200 mg of calcium and 800 IU of vitamin D.30
Pharmacologic therapy should be initiated in men over age 50 who have established osteoporosis and in postmenopausal women with established osteoporosis or osteopenia. For men over age 50 with established osteopenia, risk assessment with the glucocorticoid-corrected FRAX score (www.sheffield.ac.uk/FRAX) should be performed to identify those at high risk in whom pharmacologic therapy is warranted.31
Bisphosphonates are the first-line therapy for glucocorticoid-induced osteoporosis.32
Teriparatide is the second-line therapy and is used in patients who cannot tolerate bisphosphonates or other osteoporosis therapies, and in those who have severe osteoporosis, with T scores of –3.5 and below if they have not had a fracture, and –2.5 and below if they have had a fragility fracture.33
Denosumab, a monoclonal antibody to an osteoclast differentiating factor, may be beneficial for some patients with glucocorticoid-induced osteoporosis.34
To assess the efficacy of therapy, measuring bone mineral density at baseline and at 1 year of therapy is recommended. If density is stable or improved, then repeating the measurement at 2- to 3-year intervals is suggested.
TOCILIZUMAB: A STEROID-SPARING MEDICATION
Due to the adverse effects of long-term use of glucocorticoids and high rates of relapse, there is a pressing need for medications that are more efficacious and less toxic to treat GCA.
The European League Against Rheumatism, in its 2009 management guidelines for large-vessel vasculitis, recommend using an adjunctive immunosuppressant agent.19 In the case of GCA, they recommend using methotrexate 10 to 15 mg/week, which has shown modest evidence of reducing the relapse rate and lowering the cumulative doses of glucocorticoids needed.35,36
Studies of tumor necrosis factor inhibitors and abatacept have not yielded significant reductions in the relapse rate or decreased cumulative doses of prednisone.37,38
Advances in treatment for GCA have stagnated, but recent trials39,40 have evaluated the IL-6 receptor alpha inhibitor tocilizumab, given the central role of IL-6 in the pathogenesis of GCA. Case reports have revealed rapid induction and maintenance of remission in GCA using tocilizumab.41,42
Villiger et al39 performed a randomized, placebo-controlled trial to study the efficacy and safety of tocilizumab in induction and maintenance of disease remission in 30 patients with newly diagnosed GCA. The primary outcome, complete remission at 12 weeks, was achieved in 85% of patients who received tocilizumab plus tapered prednisolone, compared with 40% of patients who received placebo plus tapering prednisolone. The tocilizumab group also had favorable results in secondary outcomes including relapse-free survival at 52 weeks, time to first relapse after induction of remission, and cumulative dose of prednisolone.
The GiACTA trial. Stone et al40 studied the effect of tocilizumab on rates of relapse during glucocorticoid tapering in 251 GCA patients over the course of 52 weeks. Patients were randomized in a 2:1:1:1 ratio to 4 treatment groups:
- Tocilizumab weekly plus prednisone, with prednisone tapered over 26 weeks
- Tocilizumab every other week plus prednisone tapered over 26 weeks
- Placebo plus prednisone tapered over 26 weeks
- Placebo plus prednisone tapered over 52 weeks.
The primary outcome was the rate of sustained glucocorticoid-free remission at 52 weeks. Secondary outcomes included the remission rate, the cumulative glucocorticoid dose, and safety measures. At 52 weeks, the rates of sustained remission were:
- 56% with tocilizumab weekly
- 53% with tocilizumab every other week
- 14% with placebo plus 26-week prednisone taper
- 18% with placebo plus 52-week taper.
Differences between the active treatment groups and the placebo groups were statistically significant (P < .001).
The cumulative dose of prednisone in tocilizumab recipients was significantly less than in placebo recipients. Rates of adverse events were similar. Ultimately, the study showed that tocilizumab, either weekly or every other week, was more effective than prednisone alone at sustaining glucocorticoid-free remission in patients with GCA.
However, the study also raised questions about tocilizumab’s toxic effect profile and its long-term efficacy, as well as who are the optimal candidates for this therapy. Data on long-term use of tocilizumab are primarily taken from its use in rheumatoid arthritis.43 As of this writing, Stone et al are conducting an open-label trial to help provide long-term safety and efficacy data in patients with GCA. In the meantime, we must extrapolate data from the long-term use of tocilizumab in rheumatoid arthritis.
Tocilizumab and lower gastrointestinal tract perforation
One of the major adverse effects of long-term use of tocilizumab is lower gastrointestinal tract perforation.
Xie et al,44 in 2016, reported that the risk of perforation in patients on tocilizumab for rheumatoid arthritis was more than 2 times higher than in patients taking a tumor necrosis factor inhibitor. However, the absolute rates of perforation were low overall, roughly 1 to 3 per 1,000 patient-years in the tocilizumab group. Risk factors for perforation included older age, history of diverticulitis or other gastrointestinal tract condition, and prednisone doses of 7.5 mg or more a day.
Does tocilizumab prevent blindness?
Another consideration is that tocilizumab may not prevent optic neuropathy. In the GiACTA trial, 1 patient in the group receiving tocilizumab every other week developed optic neuropathy.40 Prednisone had been completely tapered off at the time, and the condition resolved when glucocorticoids were restarted. Thus, it is unknown if tocilizumab would be effective on its own without concomitant use of glucocorticoids.
Vision loss is one of the most severe complications of GCA, and it is still unclear whether tocilizumab can prevent vision loss in GCA. Also, we still have no data on the effect of tocilizumab on histopathologic findings, and whether biopsy yield diminishes over time. We hope future studies will help guide us in this regard.
No guidelines on tocilizumab yet
Clinical guidelines on the appropriate use of tocilizumab in GCA are lacking. The American College of Rheumatology and the European League Against Rheumatism have yet to publish updated guidelines with comments on use of tocilizumab. Therefore, it is unclear if tocilizumab is a first-line treatment in GCA, as its efficacy alone without glucocorticoids and its long-term safety in GCA patients have not been studied.
Treatment with tocilizumab should be individualized; it should be considered in patients who have had adverse effects from glucocorticoids, and in patients who experience a flare or cannot have their glucocorticoid dose lowered to an appropriate range.
The optimal duration of tocilizumab therapy is also unknown. However, using the GiACTA study as a rough guide, we try to limit its use to 1 year until additional data are available.
Patients on IL-6 inhibition may have suppressed C-reactive protein regardless of disease activity.43 Therefore, this laboratory value may not be reliable in determining active disease in patients on tocilizumab.
The GiACTA trial has shown an impressive improvement in the relapse-free remission period in patients with GCA taking tocilizumab. However, much work needs to be done to define the safety of this medication and determine which patients should be started on it. In the meantime, we recommend starting high-dose glucocorticoid therapy as soon as the diagnosis of GCA is suspected. In patients who do not tolerate glucocorticoids or whose disease flares during glucocorticoid taper, we recommend starting treatment with tocilizumab either once a week or every other week for at least 1 year.
- Brekke LK, Diamantopoulos AP, Fevang BT, Aßmus J, Esperø E, Gjesdal CG. Incidence of giant cell arteritis in Western Norway 1972–2012: a retrospective cohort study. Arthritis Res Ther 2017; 19(1):278. doi:10.1186/s13075-017-1479-6
- Dejaco C, Duftner C, Buttgereit F, Matteson EL, Dasgupta B. The spectrum of giant cell arteritis and polymyalgia rheumatica: revisiting the concept of the disease. Rheumatology (Oxford) 2017; 56(4):506–515. doi:10.1093/rheumatology/kew273
- Weyand CM, Goronzy JJ. Giant-cell arteritis and polymyalgia rheumatica. N Engl J Med 2014; 371(17):1653. doi:10.1056/NEJMc1409206
- Ghinoi A, Pipitone N, Nicolini A, et al. Large-vessel involvement in recent-onset giant cell arteritis: a case-control colour-Doppler sonography study. Rheumatology (Oxford) 2012; 51(4):730–734. doi:10.1093/rheumatology/ker329
- Prieto-González S, Depetris M, García-Martínez A, et al. Positron emission tomography assessment of large vessel inflammation in patients with newly diagnosed, biopsy-proven giant cell arteritis: a prospective, case-control study. Ann Rheum Dis 2014; 73(7):1388–1392. doi:10.1136/annrheumdis-2013-204572
- Brack A, Martinez-Taboada V, Stanson A, Goronzy JJ, Weyand CM. Disease pattern in cranial and large-vessel giant cell arteritis. Arthritis Rheum 1999; 42(2):311–317. doi:10.1002/1529-0131(199902)42:2<311::AID-ANR14>3.0.CO;2-F
- Salvarani C, Hunder GG. Giant cell arteritis with low erythrocyte sedimentation rate: frequency of occurence in a population-based study. Arthritis Rheum 2001; 45(2):140–145. doi:10.1002/1529-0131(200104)45:2<140::AID-ANR166>3.0.CO;2-2
- Liozon E, Jauberteau-Marchan MO, Ly K, Loustaud-Ratti V, Soria P, Vidal E. Giant cell arteritis with a low erythrocyte sedimentation rate: comments on the article by Salvarani and Hunder. Arthritis Rheum 2002; 47(6):692–694. doi:10.1002/art.10809
- Yu-Wai-Man P, Dayan MR. Giant cell arteritis with normal inflammatory markers. Acta Ophthalmol Scand 2007; 85(4):460. doi:10.1111/j.1600-0420.2006.00864.x
- Buttgereit F, Dejaco C, Matteson EL, Dasgupta B. Polymyalgia rheumatica and giant cell arteritis: a systematic review. JAMA 2016; 315(22):2442–2458. doi:10.1001/jama.2016.5444
- Niederkohr RD, Levin LA. Management of the patient with suspected temporal arteritis a decision-analytic approach. Ophthalmology 2005; 112(5):744–756. doi:10.1016/j.ophtha.2005.01.031
- Bowling K, Rait J, Atkinson J, Srinivas G. Temporal artery biopsy in the diagnosis of giant cell arteritis: does the end justify the means? Ann Med Surg (Lond) 2017; 20:1–5. doi:10.1016/j.amsu.2017.06.020
- Daily B, Dassow P, Haynes J, Nashelsky J. Giant cell arteritis: biopsy after corticosteroid initiation. Am Fam Physician 2017; 95(2):116–117. pmid:28084703
- Durling B, Toren A, Patel V, Gilberg S, Weis E, Jordan D. Incidence of discordant temporal artery biopsy in the diagnosis of giant cell arteritis. Can J Ophthalmol 2014; 49(2):157–161. doi:10.1016/j.jcjo.2013.12.008
- Dejaco C, Ramiro S, Duftner C, et al. EULAR recommendations for the use of imaging in large vessel vasculitis in clinical practice. Ann Rheum Dis 2018; 77(5):636–643. doi:10.1136/annrheumdis-2017-212649
- Aschwanden M, Imfeld S, Staub D, et al. The ultrasound compression sign to diagnose temporal giant cell arteritis shows an excellent interobserver agreement. Clin Exp Rheumatol 2015; 33(2 suppl 89):S-113–S-115. pmid:26016760
- Aiello PD, Trautmann JC, McPhee TJ, Kunselman AR, Hunder GG. Visual prognosis in giant cell arteritis. Ophthalmology 1993; 100(4):550–555. pmid:8479714
- Hayreh SS, Zimmerman B. Visual deterioration in giant cell arteritis patients while on high doses of corticosteroid therapy. Ophthalmology 2003; 110(6):1204–1215. doi:10.1016/S0161-6420(03)00228-8
- Mukhtyar C, Guillevin L, Cid MC, et al; European Vasculitis Study Group. EULAR recommendations for the management of large vessel vasculitis. Ann Rheum Dis 2009; 68(3):318–323. doi:10.1136/ard.2008.088351
- Dejaco C, Singh YP, Perel P, et al; European League Against Rheumatism; American College of Rheumatology. 2015 recommendations for the management of polymyalgia rheumatica: a European League Against Rheumatism/American College of Rheumatology collaborative initiative. Ann Rheum Dis 2015; 74(10):1799–1807. doi:10.1136/annrheumdis-2015-207492
- Bienvenu B, Ly KH, Lambert M, et al; Groupe d’Étude Français des Artérites des gros Vaisseaux, under the Aegis of the Filière des Maladies Auto-Immunes et Auto-Inflammatoires Rares. Management of giant cell arteritis: recommendations of the French Study Group for Large Vessel Vasculitis (GEFA). Rev Med Interne 2016; 37(3):154–165. doi:10.1016/j.revmed.2015.12.015
- Hayreh SS, Biousse V. Treatment of acute visual loss in giant cell arteritis: should we prescribe high-dose intravenous steroids or just oral steroids? J Neuroophthalmol 2012; 32(3):278–287. doi:10.1097/WNO.0b013e3182688218
- Restuccia G, Boiardi L, Cavazza A, et al. Flares in biopsy-proven giant cell arteritis in Northern Italy: characteristics and predictors in a long-term follow-up study. Medicine (Baltimore) 2016; 95(19):e3524. doi:10.1097/MD.0000000000003524
- Kermani TA, Warrington KJ, Cuthbertson D, et al; Vasculitis Clinical Research Consortium. Disease relapses among patients with giant cell arteritis: a prospective, longitudinal cohort study. J Rheumatol 2015; 42(7):1213–1217. doi:10.3899/jrheum.141347
- Labarca C, Koster MJ, Crowson CS, et al. Predictors of relapse and treatment outcomes in biopsy-proven giant cell arteritis: a retrospective cohort study. Rheumatology (Oxford) 2016; 55(2):347–356. doi:10.1093/rheumatology/kev348
- Proven A, Gabriel SE, Orces C, O’Fallon WM, Hunder GG. Glucocorticoid therapy in giant cell arteritis: duration and adverse outcomes. Arthritis Rheum 2003; 49(5):703–708. doi:10.1002/art.11388
- Sepkowitz KA. Opportunistic infections in patients with and patients without acquired immunodeficiency syndrome. Clin Infect Dis 2002; 34(8):1098–1107. doi:10.1086/339548
- van Staa TP, Leufkens HG, Cooper C. The epidemiology of corticosteroid-induced osteoporosis: a meta-analysis. Osteoporos Int 2002; 13(10):777–787. doi:10.1007/s001980200108
- Heffernan MP, Saag KG, Robinson JK, Callen JP. Prevention of osteoporosis associated with chronic glucocorticoid therapy. JAMA 2006; 295(11):1300–1303. pmid:16541489
- Buckley L, Guyatt G, Fink HA, et al. 2017 American College of Rheumatology guideline for the prevention and treatment of glucocorticoid-induced osteoporosis. Arthritis Care Res (Hoboken) 2017; 69(8):1095–1110. doi:10.1002/acr.23279
- Grossman JM, Gordon R, Ranganath VK, et al. American College of Rheumatology 2010 recommendations for the prevention and treatment of glucocorticoid-induced osteoporosis. Arthritis Care Res 201; 62(11):1515–1526. doi:10.1002/acr.20295
- Allen CS, Yeung JH, Vandermeer B, Homik J. Bisphosphonates for steroid-induced osteoporosis. Cochrane Database Syst Rev 2016; 10:CD001347. doi:10.1002/14651858.CD001347.pub2
- Carpinteri R, Porcelli T, Mejia C, et al. Glucocorticoid-induced osteoporosis and parathyroid hormone. J Endocrinol Invest 2010; 33(suppl 7):16–21. pmid:20938221
- Saag KG, Wagman RB, Geusens P, et al. Denosumab versus risedronate in glucocorticoid-induced osteoporosis: a multicentre, randomised, double-blind, active-controlled, double-dummy, non-inferiority study. Lancet Diabetes Endocrinol 2018; 6(6):445–454. doi:10.1016/S2213-8587(18)30075-5
- Hoffman GS, Cid MC, Hellmann DB, et al; International Network for the Study of Systemic Vasculitides. A multicenter, randomized, double-blind, placebo-controlled trial of adjuvant methotrexate treatment for giant cell arteritis. Arthritis Rheum 2002; 46(5):1309–1318. doi:10.1002/art.10262
- Spiera RF, Mitnick HJ, Kupersmith M, et al. A prospective, double-blind, randomized, placebo controlled trial of methotrexate in the treatment of giant cell arteritis (GCA). Clin Exp Rheumatol 2001; 19(5):495–501. pmid:11579707
- Hoffman GS, Cid MC, Rendt-Zagar KE, et al; Infliximab-GCA Study Group. Infliximab for maintenance of glucocorticosteroid-induced remission of giant cell arteritis: a randomized trial. Ann Intern Med 2007; 146(9):621–630. pmid:17470830
- Langford CA, Cuthbertson D, Ytterberg SR, et al; Vasculitis Clinical Research Consortium. A randomized, double-blind trial of abatacept (CTLA-4Ig) for the treatment of giant cell arteritis. Arthritis Rheumatol 2017; 69(4):837–845. doi:10.1002/art.40044
- Villiger PM, Adler S, Kuchen S, et al. Tocilizumab for induction and maintenance of remission in giant cell arteritis: a phase 2, randomised, double-blind, placebo-controlled trial. Lancet. 2016; 387(10031):1921–1927. doi:10.1016/S0140-6736(16)00560-2
- Stone JH, Tuckwell K, Dimonaco S, et al. Trial of tocilizumab in giant-cell arteritis. N Engl J Med 2017; 377(4):317–328. doi:10.1056/NEJMoa1613849
- Oliveira F, Butendieck RR, Ginsburg WW, Parikh K, Abril A. Tocilizumab, an effective treatment for relapsing giant cell arteritis. Clin Exp Rheumatol 2014; 32(3 suppl 82):S76–S78. pmid:24854376
- Loricera J, Blanco R, Hernández JL, et al. Tocilizumab in giant cell arteritis: multicenter open-label study of 22 patients. Semin Arthritis Rheum 2015; 44(6):717–723. doi:10.1016/j.semarthrit.2014.12.005
- Tamaki H, Hajj-Ali RA. Tocilizumab for giant cell arteritis—a new giant step in an old disease. JAMA Neurol 2018; 75(2):145–146. doi:10.1001/jamaneurol.2017.3811
- Xie F, Yun H, Bernatsky S, Curtis JR. Risk for gastrointestinal perforation among rheumatoid arthritis patients receiving tofacitinib, tocilizumab, or other biologics. Arthritis Rheumatol 2016; 68(11):2612–2617. doi:10.1002/art.39761
Giant cell arteritis (GCA) is a systemic vasculitis involving medium-sized and large arteries, most commonly the temporal, ophthalmic, occipital, vertebral, posterior ciliary, and proximal vertebral arteries. Moreover, involvement of the ophthalmic artery and its branches results in loss of vision. GCA can also involve the aorta and its proximal branches, especially in the upper extremities.
GCA is the most common systemic vasculitis in adults. It occurs almost exclusively in patients over age 50 and affects women more than men. It is most frequent in populations of northern European ancestry, especially Scandinavian. In a retrospective cohort study in Norway, the average annual cumulative incidence rate of GCA was 16.7 per 100,000 people over age 50.1 Risk factors include older age, history of smoking, current smoking, early menopause, and, possibly, stress-related disorders.2
PATHOGENESIS IS NOT COMPLETELY UNDERSTOOD
The pathogenesis of GCA is not completely understood, but there is evidence of immune activation in the arterial wall leading to activation of macrophages and formation of multinucleated giant cells (which may not always be present in biopsies).
The most relevant cytokines in the ongoing pathogenesis are still being defined, but the presence of interferon gamma and interleukin 6 (IL-6) seem to be critical for the expression of the disease. The primary immunogenic triggers for the elaboration of these cytokines and the arteritis remain elusive.
A SPECTRUM OF PRESENTATIONS
The initial symptoms of GCA may be vague, such as malaise, fever, and night sweats, and are likely due to systemic inflammation. Features of vascular involvement include headache, scalp tenderness, and jaw claudication (cramping pain in the jaw while chewing).
A less common but serious feature associated with GCA is partial or complete vision loss affecting 1 or both eyes.3 Some patients suddenly go completely blind without any visual prodrome.
Overlapping GCA phenotypes exist, with a spectrum of presentations that include classic cranial arteritis, extracranial GCA (also called large-vessel GCA), and polymyalgia rheumatica.2
Cranial GCA, the best-characterized clinical presentation, causes symptoms such as headache or signs such as tenderness of the temporal artery. On examination, the temporal arteries may be tender or nodular, and the pulses may be felt above the zygomatic arch, above and in front of the tragus of the ear. About two-thirds of patients with cranial GCA present with new-onset headache, most often in the temporal area, but possibly anywhere throughout the head.
Visual disturbance, jaw claudication, and tongue pain are less common but, if present, increase the likelihood of this diagnosis.2
Large-vessel involvement in GCA is common and refers to involvement of the aorta and its proximal branches. Imaging methods used in diagnosing large-vessel GCA include color Doppler ultrasonography, computed tomography with angiography, magnetic resonance imaging with angiography, and positron emission tomography. In some centers, such imaging is performed in all patients diagnosed with GCA to survey for large-vessel involvement.
Depending on the imaging study, large-vessel involvement has been found in 30% to 80% of cases of GCA.4,5 It is often associated with nonspecific symptoms such as fever, weight loss, chills, and malaise, but it can also cause more specific symptoms such as unilateral extremity claudication. In contrast to patients with cranial GCA, patients with large-vessel GCA were younger at onset, less likely to have headaches, and more likely to have arm claudication at presentation.6 Aortitis of the ascending aorta can occur with a histopathologic pattern of GCA but without the clinical stigmata of GCA.
The finding of aortitis should prompt the clinician to question the patient about other symptoms of GCA and to order imaging of the whole vascular tree. Ultrasonography and biopsy of the temporal arteries can be considered. Whether idiopathic aortitis is part of the GCA spectrum remains to be seen.
Laboratory tests often show anemia, leukocytosis, and thrombocytosis. Acute-phase reactants such as C-reactive protein and the erythrocyte sedimentation rate are often elevated. The sedimentation rate often exceeds 50 mm/hour and sometimes 100 mm/hour.
In 2 retrospective studies, the number of patients with GCA whose sedimentation rate was less than 50 mm/hour ranged between 5% and 11%.7,8 However, a small percentage of patients with GCA have normal inflammatory markers. Therefore, if the suspicion for GCA is high, treatment should be started and biopsy pursued.9 In patients with paraproteinemia or other causes of a spuriously elevated or low erythrocyte sedimentation rate, C-reactive protein is a more reliable test.
Polymyalgia rheumatica is another rheumatologic condition that can occur independently or in conjunction with GCA. It is characterized by stiffness and pain in the proximal joints such as the hips and shoulders, typically worse in the morning and better with activity. Although the patient may subjectively feel weak, a close neurologic examination will reveal normal muscle strength.
Polymyalgia rheumatica is observed in 40% to 60% of patients with GCA at the time of diagnosis; 16% to 21% of patients with polymyalgia rheumatica may develop GCA, especially if untreated.2,10
Differential diagnosis
Other vasculitides (eg, Takayasu arteritis) can also present with unexplained fever, anemia, and constitutional symptoms.
Infection should be considered if fever is present. An infectious disease accompanied by fever, headache, and elevated inflammatory markers can mimic GCA.
Nonarteritic anterior ischemic optic neuropathy can present with sudden vision loss, prompting concern for underlying GCA. Risk factors include hypertension and diabetes mellitus; other features of GCA, including elevated inflammatory markers, are generally absent.
TEMPORAL ARTERY BIOPSY: THE GOLD STANDARD FOR DIAGNOSIS
Temporal artery biopsy remains the standard to confirm the diagnosis. However, because inflammation in the temporal arteries can affect some segments but not others, biopsy results on conventional hematoxylin and eosin staining can be falsely negative in patients with GCA. In one study,11 the mean sensitivity of unilateral temporal artery biopsy was 86.9%.
Typical positive histologic findings are inflammation with panarteritis, CD4-positive lymphocytes, macrophages, giant cells, and fragmentation of the internal elastic lamina.12
When GCA is suspected, treatment with glucocorticoids should be started immediately and biopsy performed as soon as possible. Delaying biopsy for 14 days or more may not affect the accuracy of biopsy study.13 Treatment should never be withheld while awaiting the results of biopsy study.
Biopsy is usually performed unilaterally, on the same side as the symptoms or abnormal findings on examination. Bilateral temporal artery biopsy is also performed and compared with unilateral biopsy; this approach increases the diagnostic yield by about 5%.14
IMAGING
In patients with suspected GCA, imaging is recommended early to complement the clinical criteria for the diagnosis of GCA.15 Positron emission tomography, computed tomography angiography, magnetic resonance angiography, or Doppler ultrasonography can reveal inflammation of the arteries in the proximal upper or lower limbs or the aorta.2
In patients with suspected cranial GCA, ultrasonography of the temporal and axillary arteries is recommended first. If ultrasonography is not available or is inconclusive, high-resolution magnetic resonance imaging of the cranial arteries can be used as an alternative. Computed tomography and positron emission tomography of the cranial arteries are not recommended.
In patients with suspected large-vessel GCA, ultrasonography, positron emission tomography, computed tomography, and magnetic resonance imaging may be used to screen for vessel wall inflammation, edema, and luminal narrowing in extracranial arteries. Ultrasonography is of limited value in assessing aortitis.
Color duplex ultrasonography can be applied to assess for vascular inflammation of the temporal or large arteries. The typical finding of the “halo” sign, a hypoechoic ring around the arterial lumen, represents the inflammation-induced thickening of the arterial wall. The “compression sign,” the persistence of the “halo” during compression of the vessel lumen by the ultrasound probe, has high specificity for the diagnosis.16
Ultrasonography of suspected GCA has yielded sensitivities of 55% to 100% and specificities of 78% to 100%. However, its sensitivity depends on the user’s level of expertise, so it should be done only in medical centers with a high number of GCA cases and with highly experienced sonographers. High-resolution magnetic resonance imaging is an alternative to ultrasonography and has shown similar sensitivity and specificity.3
TREATMENT WITH GLUCOCORTICOIDS
Glucocorticoids remain the standard for treatment of GCA. The therapeutic effect of glucocorticoids in GCA has been established by years of clinical experience, but has never been proven in a placebo-controlled trial. When started appropriately and expeditiously, glucocorticoids produce exquisite resolution of signs and symptoms and prevent the serious complication of vision loss. Rapid resolution of symptoms is so typical of GCA that if the patient’s symptoms persist more than a few days after starting a glucocorticoid, the diagnosis of GCA should be reconsidered.
In a retrospective study of 245 patients with biopsy-proven GCA treated with glucocorticoids, 34 had permanent loss of sight.17 In 32 (94%) of the 34, the vision loss occurred before glucocorticoids were started. Of the remaining 2 patients, 1 lost vision 8 days into treatment, and the other lost vision 3 years after diagnosis and 1 year after discontinuation of glucocorticoids.
In a series of 144 patients with biopsy-proven GCA, 51 had no vision loss at presentation and no vision loss after starting glucocorticoids, and 93 had vision loss at presentation. In the latter group, symptoms worsened within 5 days of starting glucocorticoids in 9 patients.18 If vision was intact at the time of presentation, prompt initiation of glucocorticoids reduced the risk of vision loss to less than 1%.
High doses, slowly tapered
The European League Against Rheumatism recommends early initiation of high-dose glucocorticoids for patients with large-vessel vasculitis,19 and it also recommends glucocorticoids for patients with polymyalgia rheumatica.20 The optimal initial and tapering dosage has never been formally evaluated, but regimens have been devised on the basis of expert opinion.21
For patients with GCA who do not have vision loss at the time of diagnosis, the initial dose is prednisone 1 mg/kg or its equivalent daily for 2 to 4 weeks, after which it is tapered.21 If the initial dosage is prednisone 60 mg orally daily for 2 to 4 weeks, our practice is to taper it to 50 mg daily for 2 weeks, then 40 mg daily for 2 weeks. Then, it is decreased by 5 mg every 2 weeks until it is 20 mg daily, and then by 2.5 mg every 2 weeks until it is 10 mg orally daily. Thereafter, the dosage is decreased by 1 mg every 2 to 4 weeks.
For patients with GCA who experience transient vision loss or diplopia at the time of diagnosis, intravenous pulse glucocorticoid therapy should be initiated to reduce the risk of vision loss as rapidly as possible.22 A typical pulse regimen is methylprednisolone 1 g intravenously daily for 3 days. Though not rigorously validated in studies, such an approach is used to avoid vision impairment due to GCA, which is rarely reversible.
RELAPSE OF DISEASE
Suspect a relapse of GCA if the patient’s initial symptoms recur, if inflammatory markers become elevated, or if classic symptoms of GCA or polymyalgia rheumatica occur. Elevations in inflammatory markers do not definitely indicate a flare of GCA, but they should trigger close monitoring of the patient’s symptoms.
Relapse is treated by increasing the glucocorticoid dosage as appropriate to the nature of the relapse. If vision is affected or the patient has symptoms of GCA, then increments of 30 to 60 mg of prednisone are warranted, whereas if the patient has symptoms of polymyalgia rheumatica, then increments of 5 to 10 mg of prednisone are usually used.
The incidence of relapses of GCA in multiple tertiary care centers has been reported to vary between 34% and 75%.23,24 Most relapses occur at prednisone dosages of less than 20 mg orally daily and within the first year after diagnosis. The most common symptoms are limb ischemia, jaw claudication, constitutional symptoms, headaches, and polymyalgia rheumatica. In a review of 286 patients,25 213 (74%) had at least 1 relapse. The first relapse occurred in the first year in 50%, by 2 years in 68%, and by 5 years in 79%.
ADVERSE EFFECTS OF GLUCOCORTICOIDS
In high doses, glucocorticoids have well-known adverse effects. In a population-based study of 120 patients, each patient treated with glucocorticoids experienced at least 1 adverse effect (cataract, fracture, infection, osteonecrosis, diabetes, hypertension, weight gain, capillary fragility, or hair loss).26 The effects were related to aging and cumulative dosage of prednisone but not to the initial dosage.
Glucocorticoids can affect many organs and systems:
- Eyes (cataracts, increased intraocular pressure, exophthalmos)
- Heart (premature atherosclerotic disease, hypertension, fluid retention, hyperlipidemia, arrhythmias)
- Gastrointestinal system (ulcer, gastrointestinal bleeding, gastritis, visceral perforation, hepatic steatosis, acute pancreatitis)
- Bone and muscle (osteopenia, osteoporosis, osteonecrosis, myopathy)
- Brain (mood disorder, psychosis, memory impairment)
- Endocrine system (hyperglycemia, hypothalamic-pituitary-adrenal axis suppression)
- Immune system (immunosuppression, leading to infection and leukocytosis).
Patients receiving a glucocorticoid dose equivalent to 20 mg or more of prednisone daily for 1 month or more who also have another cause of immunocompromise need prophylaxis against Pneumocystis jirovecii pneumonia.27 They should also receive appropriate immunizations before starting glucocorticoids. Live-virus vaccines should not be given to these patients until they have been off glucocorticoids for 1 month.
Glucocorticoids and bone loss
Glucocorticoids are associated with bone loss and fracture, which can occur within the first few months of use and with dosages as low as 2.5 to 7.5 mg orally daily.28 Therefore, glucocorticoid-induced bone loss has to be treated aggressively, particularly in patients who are older and have a history of fragility fracture.
For patients with GCA who need glucocorticoids in doses greater than 5 mg orally daily for more than 3 months, the following measures are advised to decrease the risk of bone loss:
- Weight-bearing exercise
- Smoking cessation
- Moderation in alcohol intake
- Measures to prevent falls29
- Supplementation with 1,200 mg of calcium and 800 IU of vitamin D.30
Pharmacologic therapy should be initiated in men over age 50 who have established osteoporosis and in postmenopausal women with established osteoporosis or osteopenia. For men over age 50 with established osteopenia, risk assessment with the glucocorticoid-corrected FRAX score (www.sheffield.ac.uk/FRAX) should be performed to identify those at high risk in whom pharmacologic therapy is warranted.31
Bisphosphonates are the first-line therapy for glucocorticoid-induced osteoporosis.32
Teriparatide is the second-line therapy and is used in patients who cannot tolerate bisphosphonates or other osteoporosis therapies, and in those who have severe osteoporosis, with T scores of –3.5 and below if they have not had a fracture, and –2.5 and below if they have had a fragility fracture.33
Denosumab, a monoclonal antibody to an osteoclast differentiating factor, may be beneficial for some patients with glucocorticoid-induced osteoporosis.34
To assess the efficacy of therapy, measuring bone mineral density at baseline and at 1 year of therapy is recommended. If density is stable or improved, then repeating the measurement at 2- to 3-year intervals is suggested.
TOCILIZUMAB: A STEROID-SPARING MEDICATION
Due to the adverse effects of long-term use of glucocorticoids and high rates of relapse, there is a pressing need for medications that are more efficacious and less toxic to treat GCA.
The European League Against Rheumatism, in its 2009 management guidelines for large-vessel vasculitis, recommend using an adjunctive immunosuppressant agent.19 In the case of GCA, they recommend using methotrexate 10 to 15 mg/week, which has shown modest evidence of reducing the relapse rate and lowering the cumulative doses of glucocorticoids needed.35,36
Studies of tumor necrosis factor inhibitors and abatacept have not yielded significant reductions in the relapse rate or decreased cumulative doses of prednisone.37,38
Advances in treatment for GCA have stagnated, but recent trials39,40 have evaluated the IL-6 receptor alpha inhibitor tocilizumab, given the central role of IL-6 in the pathogenesis of GCA. Case reports have revealed rapid induction and maintenance of remission in GCA using tocilizumab.41,42
Villiger et al39 performed a randomized, placebo-controlled trial to study the efficacy and safety of tocilizumab in induction and maintenance of disease remission in 30 patients with newly diagnosed GCA. The primary outcome, complete remission at 12 weeks, was achieved in 85% of patients who received tocilizumab plus tapered prednisolone, compared with 40% of patients who received placebo plus tapering prednisolone. The tocilizumab group also had favorable results in secondary outcomes including relapse-free survival at 52 weeks, time to first relapse after induction of remission, and cumulative dose of prednisolone.
The GiACTA trial. Stone et al40 studied the effect of tocilizumab on rates of relapse during glucocorticoid tapering in 251 GCA patients over the course of 52 weeks. Patients were randomized in a 2:1:1:1 ratio to 4 treatment groups:
- Tocilizumab weekly plus prednisone, with prednisone tapered over 26 weeks
- Tocilizumab every other week plus prednisone tapered over 26 weeks
- Placebo plus prednisone tapered over 26 weeks
- Placebo plus prednisone tapered over 52 weeks.
The primary outcome was the rate of sustained glucocorticoid-free remission at 52 weeks. Secondary outcomes included the remission rate, the cumulative glucocorticoid dose, and safety measures. At 52 weeks, the rates of sustained remission were:
- 56% with tocilizumab weekly
- 53% with tocilizumab every other week
- 14% with placebo plus 26-week prednisone taper
- 18% with placebo plus 52-week taper.
Differences between the active treatment groups and the placebo groups were statistically significant (P < .001).
The cumulative dose of prednisone in tocilizumab recipients was significantly less than in placebo recipients. Rates of adverse events were similar. Ultimately, the study showed that tocilizumab, either weekly or every other week, was more effective than prednisone alone at sustaining glucocorticoid-free remission in patients with GCA.
However, the study also raised questions about tocilizumab’s toxic effect profile and its long-term efficacy, as well as who are the optimal candidates for this therapy. Data on long-term use of tocilizumab are primarily taken from its use in rheumatoid arthritis.43 As of this writing, Stone et al are conducting an open-label trial to help provide long-term safety and efficacy data in patients with GCA. In the meantime, we must extrapolate data from the long-term use of tocilizumab in rheumatoid arthritis.
Tocilizumab and lower gastrointestinal tract perforation
One of the major adverse effects of long-term use of tocilizumab is lower gastrointestinal tract perforation.
Xie et al,44 in 2016, reported that the risk of perforation in patients on tocilizumab for rheumatoid arthritis was more than 2 times higher than in patients taking a tumor necrosis factor inhibitor. However, the absolute rates of perforation were low overall, roughly 1 to 3 per 1,000 patient-years in the tocilizumab group. Risk factors for perforation included older age, history of diverticulitis or other gastrointestinal tract condition, and prednisone doses of 7.5 mg or more a day.
Does tocilizumab prevent blindness?
Another consideration is that tocilizumab may not prevent optic neuropathy. In the GiACTA trial, 1 patient in the group receiving tocilizumab every other week developed optic neuropathy.40 Prednisone had been completely tapered off at the time, and the condition resolved when glucocorticoids were restarted. Thus, it is unknown if tocilizumab would be effective on its own without concomitant use of glucocorticoids.
Vision loss is one of the most severe complications of GCA, and it is still unclear whether tocilizumab can prevent vision loss in GCA. Also, we still have no data on the effect of tocilizumab on histopathologic findings, and whether biopsy yield diminishes over time. We hope future studies will help guide us in this regard.
No guidelines on tocilizumab yet
Clinical guidelines on the appropriate use of tocilizumab in GCA are lacking. The American College of Rheumatology and the European League Against Rheumatism have yet to publish updated guidelines with comments on use of tocilizumab. Therefore, it is unclear if tocilizumab is a first-line treatment in GCA, as its efficacy alone without glucocorticoids and its long-term safety in GCA patients have not been studied.
Treatment with tocilizumab should be individualized; it should be considered in patients who have had adverse effects from glucocorticoids, and in patients who experience a flare or cannot have their glucocorticoid dose lowered to an appropriate range.
The optimal duration of tocilizumab therapy is also unknown. However, using the GiACTA study as a rough guide, we try to limit its use to 1 year until additional data are available.
Patients on IL-6 inhibition may have suppressed C-reactive protein regardless of disease activity.43 Therefore, this laboratory value may not be reliable in determining active disease in patients on tocilizumab.
The GiACTA trial has shown an impressive improvement in the relapse-free remission period in patients with GCA taking tocilizumab. However, much work needs to be done to define the safety of this medication and determine which patients should be started on it. In the meantime, we recommend starting high-dose glucocorticoid therapy as soon as the diagnosis of GCA is suspected. In patients who do not tolerate glucocorticoids or whose disease flares during glucocorticoid taper, we recommend starting treatment with tocilizumab either once a week or every other week for at least 1 year.
Giant cell arteritis (GCA) is a systemic vasculitis involving medium-sized and large arteries, most commonly the temporal, ophthalmic, occipital, vertebral, posterior ciliary, and proximal vertebral arteries. Moreover, involvement of the ophthalmic artery and its branches results in loss of vision. GCA can also involve the aorta and its proximal branches, especially in the upper extremities.
GCA is the most common systemic vasculitis in adults. It occurs almost exclusively in patients over age 50 and affects women more than men. It is most frequent in populations of northern European ancestry, especially Scandinavian. In a retrospective cohort study in Norway, the average annual cumulative incidence rate of GCA was 16.7 per 100,000 people over age 50.1 Risk factors include older age, history of smoking, current smoking, early menopause, and, possibly, stress-related disorders.2
PATHOGENESIS IS NOT COMPLETELY UNDERSTOOD
The pathogenesis of GCA is not completely understood, but there is evidence of immune activation in the arterial wall leading to activation of macrophages and formation of multinucleated giant cells (which may not always be present in biopsies).
The most relevant cytokines in the ongoing pathogenesis are still being defined, but the presence of interferon gamma and interleukin 6 (IL-6) seem to be critical for the expression of the disease. The primary immunogenic triggers for the elaboration of these cytokines and the arteritis remain elusive.
A SPECTRUM OF PRESENTATIONS
The initial symptoms of GCA may be vague, such as malaise, fever, and night sweats, and are likely due to systemic inflammation. Features of vascular involvement include headache, scalp tenderness, and jaw claudication (cramping pain in the jaw while chewing).
A less common but serious feature associated with GCA is partial or complete vision loss affecting 1 or both eyes.3 Some patients suddenly go completely blind without any visual prodrome.
Overlapping GCA phenotypes exist, with a spectrum of presentations that include classic cranial arteritis, extracranial GCA (also called large-vessel GCA), and polymyalgia rheumatica.2
Cranial GCA, the best-characterized clinical presentation, causes symptoms such as headache or signs such as tenderness of the temporal artery. On examination, the temporal arteries may be tender or nodular, and the pulses may be felt above the zygomatic arch, above and in front of the tragus of the ear. About two-thirds of patients with cranial GCA present with new-onset headache, most often in the temporal area, but possibly anywhere throughout the head.
Visual disturbance, jaw claudication, and tongue pain are less common but, if present, increase the likelihood of this diagnosis.2
Large-vessel involvement in GCA is common and refers to involvement of the aorta and its proximal branches. Imaging methods used in diagnosing large-vessel GCA include color Doppler ultrasonography, computed tomography with angiography, magnetic resonance imaging with angiography, and positron emission tomography. In some centers, such imaging is performed in all patients diagnosed with GCA to survey for large-vessel involvement.
Depending on the imaging study, large-vessel involvement has been found in 30% to 80% of cases of GCA.4,5 It is often associated with nonspecific symptoms such as fever, weight loss, chills, and malaise, but it can also cause more specific symptoms such as unilateral extremity claudication. In contrast to patients with cranial GCA, patients with large-vessel GCA were younger at onset, less likely to have headaches, and more likely to have arm claudication at presentation.6 Aortitis of the ascending aorta can occur with a histopathologic pattern of GCA but without the clinical stigmata of GCA.
The finding of aortitis should prompt the clinician to question the patient about other symptoms of GCA and to order imaging of the whole vascular tree. Ultrasonography and biopsy of the temporal arteries can be considered. Whether idiopathic aortitis is part of the GCA spectrum remains to be seen.
Laboratory tests often show anemia, leukocytosis, and thrombocytosis. Acute-phase reactants such as C-reactive protein and the erythrocyte sedimentation rate are often elevated. The sedimentation rate often exceeds 50 mm/hour and sometimes 100 mm/hour.
In 2 retrospective studies, the number of patients with GCA whose sedimentation rate was less than 50 mm/hour ranged between 5% and 11%.7,8 However, a small percentage of patients with GCA have normal inflammatory markers. Therefore, if the suspicion for GCA is high, treatment should be started and biopsy pursued.9 In patients with paraproteinemia or other causes of a spuriously elevated or low erythrocyte sedimentation rate, C-reactive protein is a more reliable test.
Polymyalgia rheumatica is another rheumatologic condition that can occur independently or in conjunction with GCA. It is characterized by stiffness and pain in the proximal joints such as the hips and shoulders, typically worse in the morning and better with activity. Although the patient may subjectively feel weak, a close neurologic examination will reveal normal muscle strength.
Polymyalgia rheumatica is observed in 40% to 60% of patients with GCA at the time of diagnosis; 16% to 21% of patients with polymyalgia rheumatica may develop GCA, especially if untreated.2,10
Differential diagnosis
Other vasculitides (eg, Takayasu arteritis) can also present with unexplained fever, anemia, and constitutional symptoms.
Infection should be considered if fever is present. An infectious disease accompanied by fever, headache, and elevated inflammatory markers can mimic GCA.
Nonarteritic anterior ischemic optic neuropathy can present with sudden vision loss, prompting concern for underlying GCA. Risk factors include hypertension and diabetes mellitus; other features of GCA, including elevated inflammatory markers, are generally absent.
TEMPORAL ARTERY BIOPSY: THE GOLD STANDARD FOR DIAGNOSIS
Temporal artery biopsy remains the standard to confirm the diagnosis. However, because inflammation in the temporal arteries can affect some segments but not others, biopsy results on conventional hematoxylin and eosin staining can be falsely negative in patients with GCA. In one study,11 the mean sensitivity of unilateral temporal artery biopsy was 86.9%.
Typical positive histologic findings are inflammation with panarteritis, CD4-positive lymphocytes, macrophages, giant cells, and fragmentation of the internal elastic lamina.12
When GCA is suspected, treatment with glucocorticoids should be started immediately and biopsy performed as soon as possible. Delaying biopsy for 14 days or more may not affect the accuracy of biopsy study.13 Treatment should never be withheld while awaiting the results of biopsy study.
Biopsy is usually performed unilaterally, on the same side as the symptoms or abnormal findings on examination. Bilateral temporal artery biopsy is also performed and compared with unilateral biopsy; this approach increases the diagnostic yield by about 5%.14
IMAGING
In patients with suspected GCA, imaging is recommended early to complement the clinical criteria for the diagnosis of GCA.15 Positron emission tomography, computed tomography angiography, magnetic resonance angiography, or Doppler ultrasonography can reveal inflammation of the arteries in the proximal upper or lower limbs or the aorta.2
In patients with suspected cranial GCA, ultrasonography of the temporal and axillary arteries is recommended first. If ultrasonography is not available or is inconclusive, high-resolution magnetic resonance imaging of the cranial arteries can be used as an alternative. Computed tomography and positron emission tomography of the cranial arteries are not recommended.
In patients with suspected large-vessel GCA, ultrasonography, positron emission tomography, computed tomography, and magnetic resonance imaging may be used to screen for vessel wall inflammation, edema, and luminal narrowing in extracranial arteries. Ultrasonography is of limited value in assessing aortitis.
Color duplex ultrasonography can be applied to assess for vascular inflammation of the temporal or large arteries. The typical finding of the “halo” sign, a hypoechoic ring around the arterial lumen, represents the inflammation-induced thickening of the arterial wall. The “compression sign,” the persistence of the “halo” during compression of the vessel lumen by the ultrasound probe, has high specificity for the diagnosis.16
Ultrasonography of suspected GCA has yielded sensitivities of 55% to 100% and specificities of 78% to 100%. However, its sensitivity depends on the user’s level of expertise, so it should be done only in medical centers with a high number of GCA cases and with highly experienced sonographers. High-resolution magnetic resonance imaging is an alternative to ultrasonography and has shown similar sensitivity and specificity.3
TREATMENT WITH GLUCOCORTICOIDS
Glucocorticoids remain the standard for treatment of GCA. The therapeutic effect of glucocorticoids in GCA has been established by years of clinical experience, but has never been proven in a placebo-controlled trial. When started appropriately and expeditiously, glucocorticoids produce exquisite resolution of signs and symptoms and prevent the serious complication of vision loss. Rapid resolution of symptoms is so typical of GCA that if the patient’s symptoms persist more than a few days after starting a glucocorticoid, the diagnosis of GCA should be reconsidered.
In a retrospective study of 245 patients with biopsy-proven GCA treated with glucocorticoids, 34 had permanent loss of sight.17 In 32 (94%) of the 34, the vision loss occurred before glucocorticoids were started. Of the remaining 2 patients, 1 lost vision 8 days into treatment, and the other lost vision 3 years after diagnosis and 1 year after discontinuation of glucocorticoids.
In a series of 144 patients with biopsy-proven GCA, 51 had no vision loss at presentation and no vision loss after starting glucocorticoids, and 93 had vision loss at presentation. In the latter group, symptoms worsened within 5 days of starting glucocorticoids in 9 patients.18 If vision was intact at the time of presentation, prompt initiation of glucocorticoids reduced the risk of vision loss to less than 1%.
High doses, slowly tapered
The European League Against Rheumatism recommends early initiation of high-dose glucocorticoids for patients with large-vessel vasculitis,19 and it also recommends glucocorticoids for patients with polymyalgia rheumatica.20 The optimal initial and tapering dosage has never been formally evaluated, but regimens have been devised on the basis of expert opinion.21
For patients with GCA who do not have vision loss at the time of diagnosis, the initial dose is prednisone 1 mg/kg or its equivalent daily for 2 to 4 weeks, after which it is tapered.21 If the initial dosage is prednisone 60 mg orally daily for 2 to 4 weeks, our practice is to taper it to 50 mg daily for 2 weeks, then 40 mg daily for 2 weeks. Then, it is decreased by 5 mg every 2 weeks until it is 20 mg daily, and then by 2.5 mg every 2 weeks until it is 10 mg orally daily. Thereafter, the dosage is decreased by 1 mg every 2 to 4 weeks.
For patients with GCA who experience transient vision loss or diplopia at the time of diagnosis, intravenous pulse glucocorticoid therapy should be initiated to reduce the risk of vision loss as rapidly as possible.22 A typical pulse regimen is methylprednisolone 1 g intravenously daily for 3 days. Though not rigorously validated in studies, such an approach is used to avoid vision impairment due to GCA, which is rarely reversible.
RELAPSE OF DISEASE
Suspect a relapse of GCA if the patient’s initial symptoms recur, if inflammatory markers become elevated, or if classic symptoms of GCA or polymyalgia rheumatica occur. Elevations in inflammatory markers do not definitely indicate a flare of GCA, but they should trigger close monitoring of the patient’s symptoms.
Relapse is treated by increasing the glucocorticoid dosage as appropriate to the nature of the relapse. If vision is affected or the patient has symptoms of GCA, then increments of 30 to 60 mg of prednisone are warranted, whereas if the patient has symptoms of polymyalgia rheumatica, then increments of 5 to 10 mg of prednisone are usually used.
The incidence of relapses of GCA in multiple tertiary care centers has been reported to vary between 34% and 75%.23,24 Most relapses occur at prednisone dosages of less than 20 mg orally daily and within the first year after diagnosis. The most common symptoms are limb ischemia, jaw claudication, constitutional symptoms, headaches, and polymyalgia rheumatica. In a review of 286 patients,25 213 (74%) had at least 1 relapse. The first relapse occurred in the first year in 50%, by 2 years in 68%, and by 5 years in 79%.
ADVERSE EFFECTS OF GLUCOCORTICOIDS
In high doses, glucocorticoids have well-known adverse effects. In a population-based study of 120 patients, each patient treated with glucocorticoids experienced at least 1 adverse effect (cataract, fracture, infection, osteonecrosis, diabetes, hypertension, weight gain, capillary fragility, or hair loss).26 The effects were related to aging and cumulative dosage of prednisone but not to the initial dosage.
Glucocorticoids can affect many organs and systems:
- Eyes (cataracts, increased intraocular pressure, exophthalmos)
- Heart (premature atherosclerotic disease, hypertension, fluid retention, hyperlipidemia, arrhythmias)
- Gastrointestinal system (ulcer, gastrointestinal bleeding, gastritis, visceral perforation, hepatic steatosis, acute pancreatitis)
- Bone and muscle (osteopenia, osteoporosis, osteonecrosis, myopathy)
- Brain (mood disorder, psychosis, memory impairment)
- Endocrine system (hyperglycemia, hypothalamic-pituitary-adrenal axis suppression)
- Immune system (immunosuppression, leading to infection and leukocytosis).
Patients receiving a glucocorticoid dose equivalent to 20 mg or more of prednisone daily for 1 month or more who also have another cause of immunocompromise need prophylaxis against Pneumocystis jirovecii pneumonia.27 They should also receive appropriate immunizations before starting glucocorticoids. Live-virus vaccines should not be given to these patients until they have been off glucocorticoids for 1 month.
Glucocorticoids and bone loss
Glucocorticoids are associated with bone loss and fracture, which can occur within the first few months of use and with dosages as low as 2.5 to 7.5 mg orally daily.28 Therefore, glucocorticoid-induced bone loss has to be treated aggressively, particularly in patients who are older and have a history of fragility fracture.
For patients with GCA who need glucocorticoids in doses greater than 5 mg orally daily for more than 3 months, the following measures are advised to decrease the risk of bone loss:
- Weight-bearing exercise
- Smoking cessation
- Moderation in alcohol intake
- Measures to prevent falls29
- Supplementation with 1,200 mg of calcium and 800 IU of vitamin D.30
Pharmacologic therapy should be initiated in men over age 50 who have established osteoporosis and in postmenopausal women with established osteoporosis or osteopenia. For men over age 50 with established osteopenia, risk assessment with the glucocorticoid-corrected FRAX score (www.sheffield.ac.uk/FRAX) should be performed to identify those at high risk in whom pharmacologic therapy is warranted.31
Bisphosphonates are the first-line therapy for glucocorticoid-induced osteoporosis.32
Teriparatide is the second-line therapy and is used in patients who cannot tolerate bisphosphonates or other osteoporosis therapies, and in those who have severe osteoporosis, with T scores of –3.5 and below if they have not had a fracture, and –2.5 and below if they have had a fragility fracture.33
Denosumab, a monoclonal antibody to an osteoclast differentiating factor, may be beneficial for some patients with glucocorticoid-induced osteoporosis.34
To assess the efficacy of therapy, measuring bone mineral density at baseline and at 1 year of therapy is recommended. If density is stable or improved, then repeating the measurement at 2- to 3-year intervals is suggested.
TOCILIZUMAB: A STEROID-SPARING MEDICATION
Due to the adverse effects of long-term use of glucocorticoids and high rates of relapse, there is a pressing need for medications that are more efficacious and less toxic to treat GCA.
The European League Against Rheumatism, in its 2009 management guidelines for large-vessel vasculitis, recommend using an adjunctive immunosuppressant agent.19 In the case of GCA, they recommend using methotrexate 10 to 15 mg/week, which has shown modest evidence of reducing the relapse rate and lowering the cumulative doses of glucocorticoids needed.35,36
Studies of tumor necrosis factor inhibitors and abatacept have not yielded significant reductions in the relapse rate or decreased cumulative doses of prednisone.37,38
Advances in treatment for GCA have stagnated, but recent trials39,40 have evaluated the IL-6 receptor alpha inhibitor tocilizumab, given the central role of IL-6 in the pathogenesis of GCA. Case reports have revealed rapid induction and maintenance of remission in GCA using tocilizumab.41,42
Villiger et al39 performed a randomized, placebo-controlled trial to study the efficacy and safety of tocilizumab in induction and maintenance of disease remission in 30 patients with newly diagnosed GCA. The primary outcome, complete remission at 12 weeks, was achieved in 85% of patients who received tocilizumab plus tapered prednisolone, compared with 40% of patients who received placebo plus tapering prednisolone. The tocilizumab group also had favorable results in secondary outcomes including relapse-free survival at 52 weeks, time to first relapse after induction of remission, and cumulative dose of prednisolone.
The GiACTA trial. Stone et al40 studied the effect of tocilizumab on rates of relapse during glucocorticoid tapering in 251 GCA patients over the course of 52 weeks. Patients were randomized in a 2:1:1:1 ratio to 4 treatment groups:
- Tocilizumab weekly plus prednisone, with prednisone tapered over 26 weeks
- Tocilizumab every other week plus prednisone tapered over 26 weeks
- Placebo plus prednisone tapered over 26 weeks
- Placebo plus prednisone tapered over 52 weeks.
The primary outcome was the rate of sustained glucocorticoid-free remission at 52 weeks. Secondary outcomes included the remission rate, the cumulative glucocorticoid dose, and safety measures. At 52 weeks, the rates of sustained remission were:
- 56% with tocilizumab weekly
- 53% with tocilizumab every other week
- 14% with placebo plus 26-week prednisone taper
- 18% with placebo plus 52-week taper.
Differences between the active treatment groups and the placebo groups were statistically significant (P < .001).
The cumulative dose of prednisone in tocilizumab recipients was significantly less than in placebo recipients. Rates of adverse events were similar. Ultimately, the study showed that tocilizumab, either weekly or every other week, was more effective than prednisone alone at sustaining glucocorticoid-free remission in patients with GCA.
However, the study also raised questions about tocilizumab’s toxic effect profile and its long-term efficacy, as well as who are the optimal candidates for this therapy. Data on long-term use of tocilizumab are primarily taken from its use in rheumatoid arthritis.43 As of this writing, Stone et al are conducting an open-label trial to help provide long-term safety and efficacy data in patients with GCA. In the meantime, we must extrapolate data from the long-term use of tocilizumab in rheumatoid arthritis.
Tocilizumab and lower gastrointestinal tract perforation
One of the major adverse effects of long-term use of tocilizumab is lower gastrointestinal tract perforation.
Xie et al,44 in 2016, reported that the risk of perforation in patients on tocilizumab for rheumatoid arthritis was more than 2 times higher than in patients taking a tumor necrosis factor inhibitor. However, the absolute rates of perforation were low overall, roughly 1 to 3 per 1,000 patient-years in the tocilizumab group. Risk factors for perforation included older age, history of diverticulitis or other gastrointestinal tract condition, and prednisone doses of 7.5 mg or more a day.
Does tocilizumab prevent blindness?
Another consideration is that tocilizumab may not prevent optic neuropathy. In the GiACTA trial, 1 patient in the group receiving tocilizumab every other week developed optic neuropathy.40 Prednisone had been completely tapered off at the time, and the condition resolved when glucocorticoids were restarted. Thus, it is unknown if tocilizumab would be effective on its own without concomitant use of glucocorticoids.
Vision loss is one of the most severe complications of GCA, and it is still unclear whether tocilizumab can prevent vision loss in GCA. Also, we still have no data on the effect of tocilizumab on histopathologic findings, and whether biopsy yield diminishes over time. We hope future studies will help guide us in this regard.
No guidelines on tocilizumab yet
Clinical guidelines on the appropriate use of tocilizumab in GCA are lacking. The American College of Rheumatology and the European League Against Rheumatism have yet to publish updated guidelines with comments on use of tocilizumab. Therefore, it is unclear if tocilizumab is a first-line treatment in GCA, as its efficacy alone without glucocorticoids and its long-term safety in GCA patients have not been studied.
Treatment with tocilizumab should be individualized; it should be considered in patients who have had adverse effects from glucocorticoids, and in patients who experience a flare or cannot have their glucocorticoid dose lowered to an appropriate range.
The optimal duration of tocilizumab therapy is also unknown. However, using the GiACTA study as a rough guide, we try to limit its use to 1 year until additional data are available.
Patients on IL-6 inhibition may have suppressed C-reactive protein regardless of disease activity.43 Therefore, this laboratory value may not be reliable in determining active disease in patients on tocilizumab.
The GiACTA trial has shown an impressive improvement in the relapse-free remission period in patients with GCA taking tocilizumab. However, much work needs to be done to define the safety of this medication and determine which patients should be started on it. In the meantime, we recommend starting high-dose glucocorticoid therapy as soon as the diagnosis of GCA is suspected. In patients who do not tolerate glucocorticoids or whose disease flares during glucocorticoid taper, we recommend starting treatment with tocilizumab either once a week or every other week for at least 1 year.
- Brekke LK, Diamantopoulos AP, Fevang BT, Aßmus J, Esperø E, Gjesdal CG. Incidence of giant cell arteritis in Western Norway 1972–2012: a retrospective cohort study. Arthritis Res Ther 2017; 19(1):278. doi:10.1186/s13075-017-1479-6
- Dejaco C, Duftner C, Buttgereit F, Matteson EL, Dasgupta B. The spectrum of giant cell arteritis and polymyalgia rheumatica: revisiting the concept of the disease. Rheumatology (Oxford) 2017; 56(4):506–515. doi:10.1093/rheumatology/kew273
- Weyand CM, Goronzy JJ. Giant-cell arteritis and polymyalgia rheumatica. N Engl J Med 2014; 371(17):1653. doi:10.1056/NEJMc1409206
- Ghinoi A, Pipitone N, Nicolini A, et al. Large-vessel involvement in recent-onset giant cell arteritis: a case-control colour-Doppler sonography study. Rheumatology (Oxford) 2012; 51(4):730–734. doi:10.1093/rheumatology/ker329
- Prieto-González S, Depetris M, García-Martínez A, et al. Positron emission tomography assessment of large vessel inflammation in patients with newly diagnosed, biopsy-proven giant cell arteritis: a prospective, case-control study. Ann Rheum Dis 2014; 73(7):1388–1392. doi:10.1136/annrheumdis-2013-204572
- Brack A, Martinez-Taboada V, Stanson A, Goronzy JJ, Weyand CM. Disease pattern in cranial and large-vessel giant cell arteritis. Arthritis Rheum 1999; 42(2):311–317. doi:10.1002/1529-0131(199902)42:2<311::AID-ANR14>3.0.CO;2-F
- Salvarani C, Hunder GG. Giant cell arteritis with low erythrocyte sedimentation rate: frequency of occurence in a population-based study. Arthritis Rheum 2001; 45(2):140–145. doi:10.1002/1529-0131(200104)45:2<140::AID-ANR166>3.0.CO;2-2
- Liozon E, Jauberteau-Marchan MO, Ly K, Loustaud-Ratti V, Soria P, Vidal E. Giant cell arteritis with a low erythrocyte sedimentation rate: comments on the article by Salvarani and Hunder. Arthritis Rheum 2002; 47(6):692–694. doi:10.1002/art.10809
- Yu-Wai-Man P, Dayan MR. Giant cell arteritis with normal inflammatory markers. Acta Ophthalmol Scand 2007; 85(4):460. doi:10.1111/j.1600-0420.2006.00864.x
- Buttgereit F, Dejaco C, Matteson EL, Dasgupta B. Polymyalgia rheumatica and giant cell arteritis: a systematic review. JAMA 2016; 315(22):2442–2458. doi:10.1001/jama.2016.5444
- Niederkohr RD, Levin LA. Management of the patient with suspected temporal arteritis a decision-analytic approach. Ophthalmology 2005; 112(5):744–756. doi:10.1016/j.ophtha.2005.01.031
- Bowling K, Rait J, Atkinson J, Srinivas G. Temporal artery biopsy in the diagnosis of giant cell arteritis: does the end justify the means? Ann Med Surg (Lond) 2017; 20:1–5. doi:10.1016/j.amsu.2017.06.020
- Daily B, Dassow P, Haynes J, Nashelsky J. Giant cell arteritis: biopsy after corticosteroid initiation. Am Fam Physician 2017; 95(2):116–117. pmid:28084703
- Durling B, Toren A, Patel V, Gilberg S, Weis E, Jordan D. Incidence of discordant temporal artery biopsy in the diagnosis of giant cell arteritis. Can J Ophthalmol 2014; 49(2):157–161. doi:10.1016/j.jcjo.2013.12.008
- Dejaco C, Ramiro S, Duftner C, et al. EULAR recommendations for the use of imaging in large vessel vasculitis in clinical practice. Ann Rheum Dis 2018; 77(5):636–643. doi:10.1136/annrheumdis-2017-212649
- Aschwanden M, Imfeld S, Staub D, et al. The ultrasound compression sign to diagnose temporal giant cell arteritis shows an excellent interobserver agreement. Clin Exp Rheumatol 2015; 33(2 suppl 89):S-113–S-115. pmid:26016760
- Aiello PD, Trautmann JC, McPhee TJ, Kunselman AR, Hunder GG. Visual prognosis in giant cell arteritis. Ophthalmology 1993; 100(4):550–555. pmid:8479714
- Hayreh SS, Zimmerman B. Visual deterioration in giant cell arteritis patients while on high doses of corticosteroid therapy. Ophthalmology 2003; 110(6):1204–1215. doi:10.1016/S0161-6420(03)00228-8
- Mukhtyar C, Guillevin L, Cid MC, et al; European Vasculitis Study Group. EULAR recommendations for the management of large vessel vasculitis. Ann Rheum Dis 2009; 68(3):318–323. doi:10.1136/ard.2008.088351
- Dejaco C, Singh YP, Perel P, et al; European League Against Rheumatism; American College of Rheumatology. 2015 recommendations for the management of polymyalgia rheumatica: a European League Against Rheumatism/American College of Rheumatology collaborative initiative. Ann Rheum Dis 2015; 74(10):1799–1807. doi:10.1136/annrheumdis-2015-207492
- Bienvenu B, Ly KH, Lambert M, et al; Groupe d’Étude Français des Artérites des gros Vaisseaux, under the Aegis of the Filière des Maladies Auto-Immunes et Auto-Inflammatoires Rares. Management of giant cell arteritis: recommendations of the French Study Group for Large Vessel Vasculitis (GEFA). Rev Med Interne 2016; 37(3):154–165. doi:10.1016/j.revmed.2015.12.015
- Hayreh SS, Biousse V. Treatment of acute visual loss in giant cell arteritis: should we prescribe high-dose intravenous steroids or just oral steroids? J Neuroophthalmol 2012; 32(3):278–287. doi:10.1097/WNO.0b013e3182688218
- Restuccia G, Boiardi L, Cavazza A, et al. Flares in biopsy-proven giant cell arteritis in Northern Italy: characteristics and predictors in a long-term follow-up study. Medicine (Baltimore) 2016; 95(19):e3524. doi:10.1097/MD.0000000000003524
- Kermani TA, Warrington KJ, Cuthbertson D, et al; Vasculitis Clinical Research Consortium. Disease relapses among patients with giant cell arteritis: a prospective, longitudinal cohort study. J Rheumatol 2015; 42(7):1213–1217. doi:10.3899/jrheum.141347
- Labarca C, Koster MJ, Crowson CS, et al. Predictors of relapse and treatment outcomes in biopsy-proven giant cell arteritis: a retrospective cohort study. Rheumatology (Oxford) 2016; 55(2):347–356. doi:10.1093/rheumatology/kev348
- Proven A, Gabriel SE, Orces C, O’Fallon WM, Hunder GG. Glucocorticoid therapy in giant cell arteritis: duration and adverse outcomes. Arthritis Rheum 2003; 49(5):703–708. doi:10.1002/art.11388
- Sepkowitz KA. Opportunistic infections in patients with and patients without acquired immunodeficiency syndrome. Clin Infect Dis 2002; 34(8):1098–1107. doi:10.1086/339548
- van Staa TP, Leufkens HG, Cooper C. The epidemiology of corticosteroid-induced osteoporosis: a meta-analysis. Osteoporos Int 2002; 13(10):777–787. doi:10.1007/s001980200108
- Heffernan MP, Saag KG, Robinson JK, Callen JP. Prevention of osteoporosis associated with chronic glucocorticoid therapy. JAMA 2006; 295(11):1300–1303. pmid:16541489
- Buckley L, Guyatt G, Fink HA, et al. 2017 American College of Rheumatology guideline for the prevention and treatment of glucocorticoid-induced osteoporosis. Arthritis Care Res (Hoboken) 2017; 69(8):1095–1110. doi:10.1002/acr.23279
- Grossman JM, Gordon R, Ranganath VK, et al. American College of Rheumatology 2010 recommendations for the prevention and treatment of glucocorticoid-induced osteoporosis. Arthritis Care Res 201; 62(11):1515–1526. doi:10.1002/acr.20295
- Allen CS, Yeung JH, Vandermeer B, Homik J. Bisphosphonates for steroid-induced osteoporosis. Cochrane Database Syst Rev 2016; 10:CD001347. doi:10.1002/14651858.CD001347.pub2
- Carpinteri R, Porcelli T, Mejia C, et al. Glucocorticoid-induced osteoporosis and parathyroid hormone. J Endocrinol Invest 2010; 33(suppl 7):16–21. pmid:20938221
- Saag KG, Wagman RB, Geusens P, et al. Denosumab versus risedronate in glucocorticoid-induced osteoporosis: a multicentre, randomised, double-blind, active-controlled, double-dummy, non-inferiority study. Lancet Diabetes Endocrinol 2018; 6(6):445–454. doi:10.1016/S2213-8587(18)30075-5
- Hoffman GS, Cid MC, Hellmann DB, et al; International Network for the Study of Systemic Vasculitides. A multicenter, randomized, double-blind, placebo-controlled trial of adjuvant methotrexate treatment for giant cell arteritis. Arthritis Rheum 2002; 46(5):1309–1318. doi:10.1002/art.10262
- Spiera RF, Mitnick HJ, Kupersmith M, et al. A prospective, double-blind, randomized, placebo controlled trial of methotrexate in the treatment of giant cell arteritis (GCA). Clin Exp Rheumatol 2001; 19(5):495–501. pmid:11579707
- Hoffman GS, Cid MC, Rendt-Zagar KE, et al; Infliximab-GCA Study Group. Infliximab for maintenance of glucocorticosteroid-induced remission of giant cell arteritis: a randomized trial. Ann Intern Med 2007; 146(9):621–630. pmid:17470830
- Langford CA, Cuthbertson D, Ytterberg SR, et al; Vasculitis Clinical Research Consortium. A randomized, double-blind trial of abatacept (CTLA-4Ig) for the treatment of giant cell arteritis. Arthritis Rheumatol 2017; 69(4):837–845. doi:10.1002/art.40044
- Villiger PM, Adler S, Kuchen S, et al. Tocilizumab for induction and maintenance of remission in giant cell arteritis: a phase 2, randomised, double-blind, placebo-controlled trial. Lancet. 2016; 387(10031):1921–1927. doi:10.1016/S0140-6736(16)00560-2
- Stone JH, Tuckwell K, Dimonaco S, et al. Trial of tocilizumab in giant-cell arteritis. N Engl J Med 2017; 377(4):317–328. doi:10.1056/NEJMoa1613849
- Oliveira F, Butendieck RR, Ginsburg WW, Parikh K, Abril A. Tocilizumab, an effective treatment for relapsing giant cell arteritis. Clin Exp Rheumatol 2014; 32(3 suppl 82):S76–S78. pmid:24854376
- Loricera J, Blanco R, Hernández JL, et al. Tocilizumab in giant cell arteritis: multicenter open-label study of 22 patients. Semin Arthritis Rheum 2015; 44(6):717–723. doi:10.1016/j.semarthrit.2014.12.005
- Tamaki H, Hajj-Ali RA. Tocilizumab for giant cell arteritis—a new giant step in an old disease. JAMA Neurol 2018; 75(2):145–146. doi:10.1001/jamaneurol.2017.3811
- Xie F, Yun H, Bernatsky S, Curtis JR. Risk for gastrointestinal perforation among rheumatoid arthritis patients receiving tofacitinib, tocilizumab, or other biologics. Arthritis Rheumatol 2016; 68(11):2612–2617. doi:10.1002/art.39761
- Brekke LK, Diamantopoulos AP, Fevang BT, Aßmus J, Esperø E, Gjesdal CG. Incidence of giant cell arteritis in Western Norway 1972–2012: a retrospective cohort study. Arthritis Res Ther 2017; 19(1):278. doi:10.1186/s13075-017-1479-6
- Dejaco C, Duftner C, Buttgereit F, Matteson EL, Dasgupta B. The spectrum of giant cell arteritis and polymyalgia rheumatica: revisiting the concept of the disease. Rheumatology (Oxford) 2017; 56(4):506–515. doi:10.1093/rheumatology/kew273
- Weyand CM, Goronzy JJ. Giant-cell arteritis and polymyalgia rheumatica. N Engl J Med 2014; 371(17):1653. doi:10.1056/NEJMc1409206
- Ghinoi A, Pipitone N, Nicolini A, et al. Large-vessel involvement in recent-onset giant cell arteritis: a case-control colour-Doppler sonography study. Rheumatology (Oxford) 2012; 51(4):730–734. doi:10.1093/rheumatology/ker329
- Prieto-González S, Depetris M, García-Martínez A, et al. Positron emission tomography assessment of large vessel inflammation in patients with newly diagnosed, biopsy-proven giant cell arteritis: a prospective, case-control study. Ann Rheum Dis 2014; 73(7):1388–1392. doi:10.1136/annrheumdis-2013-204572
- Brack A, Martinez-Taboada V, Stanson A, Goronzy JJ, Weyand CM. Disease pattern in cranial and large-vessel giant cell arteritis. Arthritis Rheum 1999; 42(2):311–317. doi:10.1002/1529-0131(199902)42:2<311::AID-ANR14>3.0.CO;2-F
- Salvarani C, Hunder GG. Giant cell arteritis with low erythrocyte sedimentation rate: frequency of occurence in a population-based study. Arthritis Rheum 2001; 45(2):140–145. doi:10.1002/1529-0131(200104)45:2<140::AID-ANR166>3.0.CO;2-2
- Liozon E, Jauberteau-Marchan MO, Ly K, Loustaud-Ratti V, Soria P, Vidal E. Giant cell arteritis with a low erythrocyte sedimentation rate: comments on the article by Salvarani and Hunder. Arthritis Rheum 2002; 47(6):692–694. doi:10.1002/art.10809
- Yu-Wai-Man P, Dayan MR. Giant cell arteritis with normal inflammatory markers. Acta Ophthalmol Scand 2007; 85(4):460. doi:10.1111/j.1600-0420.2006.00864.x
- Buttgereit F, Dejaco C, Matteson EL, Dasgupta B. Polymyalgia rheumatica and giant cell arteritis: a systematic review. JAMA 2016; 315(22):2442–2458. doi:10.1001/jama.2016.5444
- Niederkohr RD, Levin LA. Management of the patient with suspected temporal arteritis a decision-analytic approach. Ophthalmology 2005; 112(5):744–756. doi:10.1016/j.ophtha.2005.01.031
- Bowling K, Rait J, Atkinson J, Srinivas G. Temporal artery biopsy in the diagnosis of giant cell arteritis: does the end justify the means? Ann Med Surg (Lond) 2017; 20:1–5. doi:10.1016/j.amsu.2017.06.020
- Daily B, Dassow P, Haynes J, Nashelsky J. Giant cell arteritis: biopsy after corticosteroid initiation. Am Fam Physician 2017; 95(2):116–117. pmid:28084703
- Durling B, Toren A, Patel V, Gilberg S, Weis E, Jordan D. Incidence of discordant temporal artery biopsy in the diagnosis of giant cell arteritis. Can J Ophthalmol 2014; 49(2):157–161. doi:10.1016/j.jcjo.2013.12.008
- Dejaco C, Ramiro S, Duftner C, et al. EULAR recommendations for the use of imaging in large vessel vasculitis in clinical practice. Ann Rheum Dis 2018; 77(5):636–643. doi:10.1136/annrheumdis-2017-212649
- Aschwanden M, Imfeld S, Staub D, et al. The ultrasound compression sign to diagnose temporal giant cell arteritis shows an excellent interobserver agreement. Clin Exp Rheumatol 2015; 33(2 suppl 89):S-113–S-115. pmid:26016760
- Aiello PD, Trautmann JC, McPhee TJ, Kunselman AR, Hunder GG. Visual prognosis in giant cell arteritis. Ophthalmology 1993; 100(4):550–555. pmid:8479714
- Hayreh SS, Zimmerman B. Visual deterioration in giant cell arteritis patients while on high doses of corticosteroid therapy. Ophthalmology 2003; 110(6):1204–1215. doi:10.1016/S0161-6420(03)00228-8
- Mukhtyar C, Guillevin L, Cid MC, et al; European Vasculitis Study Group. EULAR recommendations for the management of large vessel vasculitis. Ann Rheum Dis 2009; 68(3):318–323. doi:10.1136/ard.2008.088351
- Dejaco C, Singh YP, Perel P, et al; European League Against Rheumatism; American College of Rheumatology. 2015 recommendations for the management of polymyalgia rheumatica: a European League Against Rheumatism/American College of Rheumatology collaborative initiative. Ann Rheum Dis 2015; 74(10):1799–1807. doi:10.1136/annrheumdis-2015-207492
- Bienvenu B, Ly KH, Lambert M, et al; Groupe d’Étude Français des Artérites des gros Vaisseaux, under the Aegis of the Filière des Maladies Auto-Immunes et Auto-Inflammatoires Rares. Management of giant cell arteritis: recommendations of the French Study Group for Large Vessel Vasculitis (GEFA). Rev Med Interne 2016; 37(3):154–165. doi:10.1016/j.revmed.2015.12.015
- Hayreh SS, Biousse V. Treatment of acute visual loss in giant cell arteritis: should we prescribe high-dose intravenous steroids or just oral steroids? J Neuroophthalmol 2012; 32(3):278–287. doi:10.1097/WNO.0b013e3182688218
- Restuccia G, Boiardi L, Cavazza A, et al. Flares in biopsy-proven giant cell arteritis in Northern Italy: characteristics and predictors in a long-term follow-up study. Medicine (Baltimore) 2016; 95(19):e3524. doi:10.1097/MD.0000000000003524
- Kermani TA, Warrington KJ, Cuthbertson D, et al; Vasculitis Clinical Research Consortium. Disease relapses among patients with giant cell arteritis: a prospective, longitudinal cohort study. J Rheumatol 2015; 42(7):1213–1217. doi:10.3899/jrheum.141347
- Labarca C, Koster MJ, Crowson CS, et al. Predictors of relapse and treatment outcomes in biopsy-proven giant cell arteritis: a retrospective cohort study. Rheumatology (Oxford) 2016; 55(2):347–356. doi:10.1093/rheumatology/kev348
- Proven A, Gabriel SE, Orces C, O’Fallon WM, Hunder GG. Glucocorticoid therapy in giant cell arteritis: duration and adverse outcomes. Arthritis Rheum 2003; 49(5):703–708. doi:10.1002/art.11388
- Sepkowitz KA. Opportunistic infections in patients with and patients without acquired immunodeficiency syndrome. Clin Infect Dis 2002; 34(8):1098–1107. doi:10.1086/339548
- van Staa TP, Leufkens HG, Cooper C. The epidemiology of corticosteroid-induced osteoporosis: a meta-analysis. Osteoporos Int 2002; 13(10):777–787. doi:10.1007/s001980200108
- Heffernan MP, Saag KG, Robinson JK, Callen JP. Prevention of osteoporosis associated with chronic glucocorticoid therapy. JAMA 2006; 295(11):1300–1303. pmid:16541489
- Buckley L, Guyatt G, Fink HA, et al. 2017 American College of Rheumatology guideline for the prevention and treatment of glucocorticoid-induced osteoporosis. Arthritis Care Res (Hoboken) 2017; 69(8):1095–1110. doi:10.1002/acr.23279
- Grossman JM, Gordon R, Ranganath VK, et al. American College of Rheumatology 2010 recommendations for the prevention and treatment of glucocorticoid-induced osteoporosis. Arthritis Care Res 201; 62(11):1515–1526. doi:10.1002/acr.20295
- Allen CS, Yeung JH, Vandermeer B, Homik J. Bisphosphonates for steroid-induced osteoporosis. Cochrane Database Syst Rev 2016; 10:CD001347. doi:10.1002/14651858.CD001347.pub2
- Carpinteri R, Porcelli T, Mejia C, et al. Glucocorticoid-induced osteoporosis and parathyroid hormone. J Endocrinol Invest 2010; 33(suppl 7):16–21. pmid:20938221
- Saag KG, Wagman RB, Geusens P, et al. Denosumab versus risedronate in glucocorticoid-induced osteoporosis: a multicentre, randomised, double-blind, active-controlled, double-dummy, non-inferiority study. Lancet Diabetes Endocrinol 2018; 6(6):445–454. doi:10.1016/S2213-8587(18)30075-5
- Hoffman GS, Cid MC, Hellmann DB, et al; International Network for the Study of Systemic Vasculitides. A multicenter, randomized, double-blind, placebo-controlled trial of adjuvant methotrexate treatment for giant cell arteritis. Arthritis Rheum 2002; 46(5):1309–1318. doi:10.1002/art.10262
- Spiera RF, Mitnick HJ, Kupersmith M, et al. A prospective, double-blind, randomized, placebo controlled trial of methotrexate in the treatment of giant cell arteritis (GCA). Clin Exp Rheumatol 2001; 19(5):495–501. pmid:11579707
- Hoffman GS, Cid MC, Rendt-Zagar KE, et al; Infliximab-GCA Study Group. Infliximab for maintenance of glucocorticosteroid-induced remission of giant cell arteritis: a randomized trial. Ann Intern Med 2007; 146(9):621–630. pmid:17470830
- Langford CA, Cuthbertson D, Ytterberg SR, et al; Vasculitis Clinical Research Consortium. A randomized, double-blind trial of abatacept (CTLA-4Ig) for the treatment of giant cell arteritis. Arthritis Rheumatol 2017; 69(4):837–845. doi:10.1002/art.40044
- Villiger PM, Adler S, Kuchen S, et al. Tocilizumab for induction and maintenance of remission in giant cell arteritis: a phase 2, randomised, double-blind, placebo-controlled trial. Lancet. 2016; 387(10031):1921–1927. doi:10.1016/S0140-6736(16)00560-2
- Stone JH, Tuckwell K, Dimonaco S, et al. Trial of tocilizumab in giant-cell arteritis. N Engl J Med 2017; 377(4):317–328. doi:10.1056/NEJMoa1613849
- Oliveira F, Butendieck RR, Ginsburg WW, Parikh K, Abril A. Tocilizumab, an effective treatment for relapsing giant cell arteritis. Clin Exp Rheumatol 2014; 32(3 suppl 82):S76–S78. pmid:24854376
- Loricera J, Blanco R, Hernández JL, et al. Tocilizumab in giant cell arteritis: multicenter open-label study of 22 patients. Semin Arthritis Rheum 2015; 44(6):717–723. doi:10.1016/j.semarthrit.2014.12.005
- Tamaki H, Hajj-Ali RA. Tocilizumab for giant cell arteritis—a new giant step in an old disease. JAMA Neurol 2018; 75(2):145–146. doi:10.1001/jamaneurol.2017.3811
- Xie F, Yun H, Bernatsky S, Curtis JR. Risk for gastrointestinal perforation among rheumatoid arthritis patients receiving tofacitinib, tocilizumab, or other biologics. Arthritis Rheumatol 2016; 68(11):2612–2617. doi:10.1002/art.39761
KEY POINTS
- Giant cell arteritis can present with cranial symptoms, extracranial large-vessel involvement, or polymyalgia rheumatica.
- Temporal artery biopsy is the standard for diagnosis.
- Adverse effects of glucocorticoid treatment, particularly bone loss, need to be managed.
- In patients treated with glucocorticoids alone, the relapse rate is high when the drugs are tapered; thus, prolonged treatment is required.
Infertility: A practical framework
For millions of couples, a primary care physician may be the first point of contact for fertility concerns. Statistics from the US Centers for Disease Control and Prevention indicate that 12% of women ages 15 to 44 received fertility services from 2006 to 2010.1 Despite seeking services, most couples requested only advice or testing rather than treatments such as ovulation-inducing medications, surgery, or, rarely, assisted reproductive technologies including in vitro fertilization. Based on these data, primary care physicians are in a unique position to offer guidance and provide fertility services in most circumstances without the need for referral.
This article reviews the answers to questions patients frequently ask, and outlines a practical framework for the evaluation and management of the infertile couple.
MANY PATIENTS SEEK INFORMATION
At least 1 million medical visits per year are for women seeking help in becoming pregnant, with the number increasing over the last several decades.1 Reasons for the increase include delayed childbearing and the effects of aging on the female reproductive system (“female reproductive aging”), as well as the availability of increasingly effective treatments for infertility.
While the prevalence of infertility in US couples is widely quoted as 10% to 15%,2 there is no estimate for the number of fertility-related questions patients routinely pose to care providers. These questions often relate to coital timing, use of lubricants, positioning, and the use of fertility trackers and ovulation predictors.
A 2017 study of women with 12 months of infertility found that only 8% sought subspecialist care vs care from a general physician or provider, indicating that generalists are most often the first point of contact.3 The majority (92%) of women responding to a survey regarding fertility-awareness education indicated a preference for immediate counseling from their general practitioner.4
Although some healthcare providers may consider infertility simply a quality-of-life issue, the World Health Organization classifies it as a disease, and as such it warrants identification, assessment, and intervention.5 Further, patients with infertility are known to experience considerable psychological distress related to their condition. In a comparison study, women with infertility experienced levels of psychological distress similar to the level in patients with cancer and patients with chronic medical illness.6
In the current era, general practitioners and women’s health specialists may also now address patients’ questions about reproductive aging and egg-freezing, which is now an established technology.7
FAILURE TO CONCEIVE AFTER 1 YEAR
As women approach age 40, the potential for fertility decreases rapidly and significantly. Women in their later 30s have only half the fertility of women in their early 20s.10 Misperceptions of aging and female fertility have been fueled by widely publicized celebrity births from women in their 40s and even 50s, without disclosing the use of frozen or donor eggs. This unfortunate fact affects women actively trying to conceive as well as women who wish to delay childbearing due to lack of a partner or for personal or professional reasons. Primary care physicians should be able to provide counseling relevant to female reproductive aging and make suitable and timely referrals for fertility preservation if indicated.
AN EMOTIONAL ISSUE
In approaching the couple with infertility, it is important to proceed with great sensitivity for the socioemotional context of this diagnosis. For both the male and female partner, infertility can be highly stigmatizing, and can be viewed as a personal or relationship failure.
Couples should be encouraged to ask embarrassing or uncomfortable questions. Although this may not be feasible in many circumstances, interviews should ideally be conducted with both partners individually as well as together, to allow sensitive issues to be shared. In some cases, a partner may be unaware of a history of a sexually transmitted infection, a prior abortion, the use of testosterone supplements or medications to enhance male sexual performance, or a vasectomy or tubal ligation during a previous relationship.
It is not unusual that the anxiety of infertility can cause decreased libido and sexual and erectile dysfunction. These issues can further complicate the problem of conceiving, and couples counseling is not uncommonly required.11 Patients are often reassured to know that they are not alone in their diagnosis.
LOOK FOR CLUES
Before embarking on a series of tests, the primary care physician can carefully evaluate for clues that may guide the diagnostic evaluation. The approach can be individualized based on the patient’s age, duration of subfertility (ie, how long they have been trying to become pregnant), and risk factors. But as a general rule, regardless of age, couples who have been trying to conceive for more than 1 year should be encouraged to pursue additional testing.
Because each month presents a new cycle of hope (often followed by intense disappointment), the prevailing sentiment to “just give it a little more time” must be countered by education and counseling. The primary care physician must increase awareness that lack of pregnancy in the stated time periods is a compelling reason for evaluation.
History-taking in the infertile couple should include a complete gynecologic and menstrual history. A history of sexually transmitted diseases that can cause tubal disease, such as gonorrhea and Chlamydia, is significant. Both partners should be assessed for a history of prior conceptions, past medical or surgical problems, medications, and exposures to environmental toxins including alcohol, tobacco, and drugs.
A detailed physical examination can provide clues to the cause of subfertility, especially if signs of obesity, androgen excess, or insulin resistance are present.
QUESTIONS OFTEN ASKED BY COUPLES TRYING TO CONCEIVE
Clinicians are frequently asked questions related to sexual practices and lifestyle in relation to fertility and should be comfortable responding to questions in these areas.
Does frequent ejaculation ‘use up’ my sperm?
Men should be reassured that frequent ejaculations do not decrease sperm counts; even daily ejaculation does not deplete the concentration of sperm. Male partners can be reassured that “saving up” is not an effective strategy; in fact, abstinence periods of greater than 5 days can adversely affect semen parameters.12
How often should we have sex?
Infrequent intercourse (< 1 time per week) reduces the monthly chance of conceiving.13 There does not seem to be a significant improvement in fecundity with daily intercourse vs intercourse on alternate days. Strict schedules surrounding intercourse may increase stress, and reassurance should be offered that intercourse need not be regimented. Every 1 to 2 days should suffice.
Are any sexual positions better for conception?
There is no evidence that particular coital positioning or remaining supine after intercourse improves fertility. Sperm can be found within the endocervix within seconds of ejaculation, irrespective of sexual position.
What is the window of fertility?
There is good evidence that the fertile window lasts approximately 6 days and closes after ovulation.13,14 Women with regular cycles can determine their typical day of ovulation based on menstrual tracking. Intercourse should begin about 6 days before ovulation and should continue every 1 to 2 days for 1 week to fully capture this window.
Should we change our lifestyle?
Couples seeking pregnancy should be advised to limit alcohol and caffeine use, completely abstain from cigarette smoking or illicit drug use, and maintain a healthy body mass index.
Very few data exist to support particular diets or supplements to promote fertility, including antioxidants and herbal remedies. Folic acid supplementation is recommended in all women attempting to conceive to reduce the incidence of birth defects.
Do lubricants reduce fertility?
Although there seem to be no differences in fecundity rates in couples using commercial lubricants, most water-based lubricants are best avoided in couples with infertility, as adverse effects on sperm have been demonstrated in vitro.15 If lubrication is needed, couples may try mineral oil, canola oil, or hydroxyetylcellulose-based lubricants (eg, Pre-seed).
Do fertility trackers work?
Many couples with primary infertility perceive that coital timing is critical and worry that their infertility is due to poorly timed intercourse; in fact, this is seldom the case.
Despite widespread marketing of urinary luteinizing hormone (LH) detection kits and electronic trackers and monitors, there is no clear evidence that these methods improve monthly rates of conception.
Women with a regular menstrual cycle should be encouraged to take notice when their cervical mucus appears clear and slippery (a sign of ovulation). Not all women are able to detect these fluctuations; however, for those who can, observing cervical mucus changes appears to be equivalent or superior to predictor kits in predicting conception.16
A PRACTICAL FRAMEWORK FOR EVALUATING THE INFERTILE COUPLE
To assess for the common factors identified in Table 1, the essential investigation of the infertile couple includes:
- Semen analysis
- Confirmation of ovulation
- Hysterosalpingography.
Consideration can also be given to ovarian reserve testing in women at risk of diminished ovarian reserve. The above investigation can be performed simultaneously to allow for prompt identification of any issues. Further, infertility is often a combination of problems (eg, anovulation in the woman together with a problem in the man), so an incomplete evaluation may overlook a coexisting diagnosis and lead to delays in treatment and pregnancy.
Tests that are no longer typically used in clinical practice are outlined in Table 2.
OVARIAN RESERVE TESTING AND FEMALE REPRODUCTIVE AGING
Ovarian reserve refers to the number of fertilizable oocytes that remain in the ovary. This reserve changes over time, and changes occur rapidly as women approach and enter their 30s. Though not the case in men, the age of the female partner is an independent risk factor for infertility. This discrepancy is due to loss of ovarian reserve, chromosome abnormalities in embryos, and the development of medical conditions with age that affect fertility.
Testing for ovarian reserve does not necessarily predict an overall inability to achieve a live birth,17 but it can predict response to exogenous gonadotropins and, to some degree, the chance for successful pregnancy with assisted reproductive technology.18
The ASRM states that testing for diminished ovarian reserve may provide useful information in women who have had a previous poor response to gonadotropins and in women planning assisted reproductive technology.19 The ASRM also indicates that the following are risk factors for diminished ovarian reserve, and clinicians may target the assessment accordingly19:
- Age 35 or older
- History of exposure to chemotherapy or pelvic radiation
- Family history of early menopause (age < 40)
- History of ovarian surgery
- Unexplained or idiopathic fertility.
Although several tests of ovarian reserve exist, either an antimullerian hormone (AMH) test or a combined cycle day-3 follicle-stimulating hormone (FSH) and estradiol level are the 2 tests commonly used in clinical practice. Antral follicle counts are an ultrasonographic measure used by infertility specialists but rarely by primary care physicians. Assays such as inhibin are rarely ordered and have limited clinical utility.
The AMH test
Many reproductive endocrinologists rely on the AMH level as a single test of ovarian reserve as it is easy to obtain, has a relatively low cost, and offers stable results. AMH is produced by the granulosa cells of the ovarian antral follicles and is readily detected in serum samples.
Conveniently for the clinician, levels of this hormone remain stable throughout the menstrual cycle and therefore can be tested on any day and at any time of day. Lower serum AMH levels (< 1 ng/mL) have been shown to correspond to diminished ovarian stimulation with gonadotropins as well as decreased embryo quality and poor pregnancy outcomes with assisted reproductive technology.19
Nevertheless, despite overall stability, AMH levels can be falsely lowered in women using exogenous hormones or with a diagnosis of hypogonadotropic hypogonadism. Levels may be higher than expected in women with polycystic ovary syndrome due to higher numbers of antral and preantral follicles in the polycystic ovary.
The day-3 follicle-stimulating hormone test
FSH and 17-beta estradiol testing can be ordered in combination to assess function of the hypothalamic-pituitary-ovarian axis on day 3 of the menstrual cycle. There is some flexibility, however, and testing obtained on cycle day 2, 3, or 4 yields equivalent results.
Although there are no strict cutoffs, FSH levels that appear elevated (> 10–20 IU/L) are associated with lower chances of conceiving with in vitro fertilization in multiple studies.20
The test is limited by levels that may fluctuate cycle to cycle, and reassuring test results do not necessarily indicate that a woman will achieve a pregnancy. Although a serum estradiol value alone is not a useful test, it can be used in combination with day-3 FSH to screen for diminished ovarian reserve.
As premature recruitment of a follicle can cause an early follicular rise in estradiol, FSH may be falsely suppressed on day 3. For example, a “normal” day-3 FSH combined with an elevated day-3 17-beta estradiol level of 60 to 80 pg/mL is associated with a poor response to medical treatments for infertility.
Female reproductive aging
Aging of the female reproductive system is a central threat to fertility, and prompt assessment and referral are warranted for women age 35 or older who have been trying to conceive for more than 6 months. The ASRM recommends that women over age 40 be evaluated immediately.21
A prevailing misconception is that regular menstrual cycles correspond with normal fertility. In reality, women lose their ability to achieve a healthy live birth in the 5 to 10 years preceding menopause. Although all women who do not desire pregnancy should still use appropriate contraception to avoid unintended pregnancy, women who do desire pregnancy should be aware of these physiologic changes.
Classic age-related changes in ovarian reserve are accompanied by a steep rise in aneuploidy and miscarriage risk.22 This is particularly relevant as women increasingly delay childbearing in modern society. Loss of fertility begins at 32 and abruptly accelerates at age 3721; this fact is poorly communicated to and understood by patients. In a 2018 study of highly educated women, most respondents failed to identify that 45-year-old women can only rarely achieve a successful pregnancy.23
In recent decades, the percentage of women who delay childbearing until after age 35 has steadily increased. There is a widespread misconception that fertility treatments and assisted reproductive technology can compensate for female reproductive aging. Primary care physicians can play a central role in reminding couples that age remains the single greatest predictor of natural fertility and the chance of success with assisted reproduction.
Further, for women who desire future fertility and are without a partner, primary care physicians can counsel them regarding the availability of donor insemination or egg freezing. Studies confirm that women want clinicians to initiate information on reproductive health, and 80% of women undergoing elective egg-freezing for fertility preservation wished that they had done so at an earlier age.24,25
FEMALE PERITONEAL AND STRUCTURAL CAUSES
Women with endometriosis, fibroids, or a history of tubal disease have impaired fecundity. Pelvic imaging is an essential component of their evaluation. Although hysterosalpingography is the mainstay of tubal assessment, in select cases ultrasonography or hysteroscopy may be indicated.
Tubal disease and hysterosalpingography
Tubal disease remains one of the most common causes of infertility in the US females. In most cases, tubal damage is secondary to pelvic inflammatory disease from infection with gonorrhea or Chlamydia, or both.
Rates of confirmed tubal-factor infertility have been shown to increase with both the severity of the infection and the number of past infections.26 In a landmark study, 1 episode of pelvic inflammatory disease was associated with a 12% risk of tubal-factor infertility, whereas 3 infections carried a risk as high as 54%. Pelvic inflammatory disease is also known to increase the risk of ectopic pregnancy.
To assess tubal patency, hysterosalpingography, a radiographic procedure, is typically performed using fluoroscopy and injected contrast material. Some centers may offer sonohysterography as a radiation-free alternative, depending on sonographic skill and experience. Both tests are best scheduled in the window between the end of menstrual bleeding and ovulation. In practice, patients with regular cycles can typically schedule hysterosalpingography between cycle days 5 and 12.
In patients with known hydrosalpinx (a distended fallopian tube due to blockage) or a history of pelvic infection, doxycycline should be given before the procedure.27 Patients with demonstrated hydrosalpinx on hysterosalpingography should receive doxycycline 100 mg twice daily for 5 days to prevent posthysterosalpingography pelvic inflammatory disease.27 Patients with active pelvic or cervical infection should not undergo hysterosalpingography .
Women with confirmed hydrosalpinx or tubal obstruction can be referred for laparoscopy. Gynecologic surgeons will plan their approach based on whether the obstruction is proximal (near the uterus) or distal (near the ovary) as well as whether hydrosalpinx, abnormal tubal architecture, salpingitis isthmica nodosa, or peritubal adhesions are noted. Tubal surgery can be effective in mild cases of tubal disease; however, as in vitro fertilization is becoming more effective, patients with moderate or severe tubal disease are increasingly being referred directly for assisted reproductive technology. Before undergoing assisted reproductive technology, hydrosalpinx will need to be addressed, as it can decrease clinical pregnancy rates with in vitro fertilization.
Endometriosis
Endometriosis is found in 21% to 47% of women with subfertility28 and commonly causes pain, ovarian cysts, and tubal disease. There is often a delay of 7 to 8 years for diagnosis due to the misapprehension that severe dysmenorrhea is normal. Women with an affected first-degree family member are at substantially increased risk.
Although endometriosis is commonly thought to result from reflux of endometrial tissue into the peritoneal cavity with menses, there are multiple proposed mechanisms for the disease.29 The pathogenesis of endometriosis is enigmatic, and there are likely as yet undetermined immunologic and genetic predispositions that confer increased risk.
Common symptoms of endometriosis are dysmenorrhea, dyspareunia, and pelvic pain, and these are sometimes accompanied by bowel and bladder symptoms. Pelvic examination classically demonstrates an immobile uterus and uterosacral nodularity; palpation of these nodules can elicit pain. On laparoscopy, endometriosis can range from minimal to severe; however, stage of endometriosis correlates poorly with reported symptoms.30
Consideration of surgery is based on clinical history, results of the pelvic examination, and possible findings on ultrasonography or hysterosalpingography. Although positive findings on imaging can support a plan for intervention, endometriosis is largely a peritoneal disease, and evidence of tubal damage or ovarian cysts is rarely evident on ultrasonography. In women with menstrual complaints (eg, dysmenorrhea, heavy menstrual bleeding, abnormal uterine bleeding) and a history of infertility, ultrasonography may be useful in determining the presence of uterine pathology such as ovarian cyst or endometrioma, large hydrosalpinx, polyp, or substantial fibroid burden—any of which may have a significant impact on female fertility.
In the absence of a reliable blood test or imaging study, the gold standard for the diagnosis of endometriosis continues to be laparoscopic surgery. Hormonal treatments for endometriosis symptoms are not effective in improving infertility and will preclude pregnancy. Laparoscopic surgery is more successful in improving pregnancy rates in women with advanced disease: pregnancy rates after surgery can be as high as 60% in women with ovarian endometriomas but are significantly lower in women with removal of minimal to mild disease.30,31 Women over age 35 or who present with low ovarian reserve and whose male partner has semen abnormalities should consider moving directly to assisted reproductive technology rather than pursuing endometriosis surgery.
MALE FACTOR INFERTILITY
Although male partners are often highly engaged in and supportive of the fertility evaluation, some are reluctant to undergo testing, and some wish to undergo semen analysis only after female factors have been ruled out. Our practice is to evaluate male factors immediately, due to the high contribution of male factors (up to 40% of cases) either alone or in combination with female factors.32
Men at particularly increased risk of semen abnormalities include those with a history of chemotherapy or radiation or exposure to toxins (eg, environmental exposures, alcohol, tobacco, illicit substances) and prescribed medications.
At a minimum, for the male partner, a reproductive history should be taken and a semen analysis ordered. Men should be directly queried about testosterone use, as this often-used anabolic steroid hormone can severely impair sperm production.
Men who have low sperm counts, motility, or morphology scores based on World Health Organization criteria should not be deemed “infertile,” as there is significant variation from one analysis to the next, and normal fertility has been reported in men with notably low sperm counts. Particular caution should be exercised in interpreting low morphology scores in men with normal counts and motility, as this parameter appears to have the least prognostic value in this context. Men with abnormal semen analyses should be referred to a specialist for further urologic evaluation and treatment.
Treatments for male factor infertility include surgery, steroid hormones, and possibly intrauterine insemination or assisted reproductive technology. In even the most challenging cases, male infertility is now largely treatable with intracytoplasmic sperm injection with assisted reproductive technology. While most advances in in vitro fertilization have been evolutionary, intracytoplasmic sperm injection was revolutionary. This breakthrough technology allows a single sperm to be injected directly into the oocyte. Sperm for this procedure can be obtained either from the ejaculate or from microsurgical testicular sperm extraction.
ANOVULATION
A thorough menstrual history can be informative, as most females of reproductive age have a fairly predictable 25-to-35-day monthly menstrual cycle. Women presenting with menstrual charting with this pattern do not require laboratory confirmation of ovulation. Basal body temperatures are rarely used currently, as they are time-consuming, can induce stress, and are confirmatory rather than predictive of ovulation. Endometrial biopsy for endometrial “dating” is no longer performed in infertile women.
If laboratory confirmation is desired, LH kit testing with a commercially available test or a luteal phase serum progesterone obtained 7 days after suspected ovulation can be obtained. A serum progesterone level higher than 3 ng/mL is indicative of ovulation.19 Due to the notable fluctuations in ovulatory-appearing progesterone levels over several hours, caution must be taken in interpreting a lower-normal level as indicative of a luteal phase insufficiency.
Polycystic ovary syndrome
Polycystic ovary syndrome is important to understand because it is a metabolic condition that predisposes patients to a variety of health risks. Along with gynecologic consequences such as infertility, abnormal uterine bleeding, and endometrial pathology, it is often accompanied by alterations in glucose and lipid metabolism, obesity, hypertension, and cardiovascular disease.35
Despite its name, the syndrome does not involve the presence of classic ovarian cysts. In fact, the cysts associated with polycystic ovary syndrome are dense accumulations of antral follicles arranged peripherally in the ovarian cortex; they should not be removed surgically as they represent the ovarian reserve.
Although ovaries that appear polycystic on transvaginal ultrasonography are often associated with the syndrome, they are not invariably present and are not absolutely required for the diagnosis of polycystic ovary syndrome based on the most commonly used criteria.35 Several diagnostic criteria have been proposed for polycystic ovary syndrome and its phenotypes. The 2003 revised Rotterdam criteria require 2 out of the following 3 features:
- Oligo-ovulation or anovulation
- Evidence of hyperandrogenism, whether clinical (eg, acne or hirsutism) or based on laboratory testing
- Polycystic-appearing ovaries on ultrasonography.
There is no single test that can diagnose the disease. Although polycystic ovary syndrome is often characterized by elevated LH levels, LH–FSH ratios, and fasting insulin levels, these are not diagnostic criteria. The diagnosis hinges on excluding other causes of anovulation such as thyroid disease, hyperprolactinemia, 21-hydroxylase deficiency, androgen-producing neoplasms, and Cushing syndrome. In addition to checking serum testosterone levels, irregular menstrual cycles and infertility should be assessed at minimum with measurement of TSH, prolactin, and day-3 FSH. Obese women should be screened for metabolic syndrome, which should include an assessment of impaired glucose tolerance with a 2-hour oral glucose tolerance test.36
Women with polycystic ovary syndrome are known to have insulin resistance, which is difficult to assess and is independent of their body mass index.37 They often report a family history of diabetes or a personal history of gestational diabetes or giving birth to infants who are large for gestational age. Although most women diagnosed with insulin resistance and anovulatory infertility will not yet have a diagnosis of diabetes, women with polycystic ovary syndrome are 3 to 7 times more likely to develop type 2 diabetes later in life37 and are at increased risk of lipid abnormalities, cardiovascular disease, and stroke. Therefore, interventions to address the compounding influences of polycystic ovary syndrome and obesity can improve fertility outcomes and help prevent long-term sequelae that accompany the syndrome.
Treatment for women with polycystic ovary syndrome attempting conception includes lifestyle modifications, medications for ovulation induction, and possible use of insulin sensitizers. Metformin alone is not effective as a single agent for achieving pregnancy.38 Diet, weight loss, and exercise can have dramatic effects on ovulation and pregnancy and should be highly encouraged.
Ovulation induction is often required in anovulatory women, either in combination with lifestyle modifications or used subsequently if modifications are not successful. Letrozole is advised as the initial agent in women with obesity and anovulatory infertility rather than clomiphene citrate; a side-by-side comparison demonstrated increased rates of ovulation and live birth with letrozole.39
Once-daily letrozole 2.5 mg or clomiphene 50 mg can be prescribed for 5 days, from cycle days 3 through 7 to cycle days 5 through 9. If this initial dosing fails to result in ovulation, the dose can be increased. Known adverse effects are hot flashes, headaches, ovarian cysts, and increased risk of multiple gestation.
Metformin should be considered as an adjunct to fertility treatments in women with polycystic ovary syndrome, especially those with obesity or impaired glucose tolerance, or if there is no response to standard ovulation induction.
Ovarian hyperstimulation syndrome (cystic enlargement of the ovaries with potentially dangerous fluid and electrolyte imbalances) can occur in women with polycystic ovary syndrome; however, it rarely occurs with oral medications.
- Chandra A, Copen CE, Stephen EH. Infertility service use in the United States: data from the National Survey of Family Growth, 1982–2010. Natl Health Stat Report 2014; (73):1–21. pmid:24467919
- Mosher WD, Pratt WF. Fecundity and infertility in the United States: incidence and trends. Fertil Steril 1991; 56(2):192–193. pmid:2070846
- Boltz MW, Sanders JN, Simonsen SE, Stanford JB. Fertility treatment, use of in vitro fertilization, and time to live birth based on initial provider type. J Am Board Fam Med 2017; 30(2):230–238. doi:10.3122/jabfm.2017.02.160184
- Hampton K, Mazza D. Fertility-awareness knowledge, attitudes and practices of women attending general practice. Aust Fam Physician 2015; 44(11):840–845. pmid:26590626
- Zegers-Hochschild F, Adamson GD, de Mouzon J, et al; International Committee for Monitoring Assisted Reproductive Technology; World Health Organization. International Committee for Monitoring Assisted Reproductive Technology (ICMART) and the World Health Organization (WHO) revised glossary of ART terminology, 2009. Fertil Steril 2009; 92(5):1520–1524. doi:10.1016/j.fertnstert.2009.09.009
- Domar AD, Zuttermeister PC, Friedman R. The psychological impact of infertility: a comparison with patients with other medical conditions. J Psychosom Obstet Gynaecol 1993; 14(suppl):45–52. pmid:8142988
- Argyle CE, Harper JC, Davies MC. Oocyte cryopreservation: where are we now? Hum Reprod Update 2016; 22(4):440–449. doi:10.1093/humupd/dmw007
- Practice Committee of American Society for Reproductive Medicine. Definitions of infertility and recurrent pregnancy loss: a committee opinion. Fertil Steril 2013; 99(1):63. doi:10.1016/j.fertnstert.2012.09.023
- Guttmacher AF. Factors affecting normal expectancy of conception. J Am Med Assoc 1956; 161(9):855–860. pmid:13319020
- Dunson DB, Baird DD, Colombo B. Increased infertility with age in men and women. Obstet Gynecol 2004; 103(1):51–56. doi:10.1097/01.AOG.0000100153.24061.45
- National Collaborating Centre for Women’s and Children’s Health (UK). Fertility: assessment and treatment for people with fertility problems. London: Royal College of Obstetricians & Gynaecologists; 2013. www.ncbi.nlm.nih.gov/books/NBK247932. Accessed May 6, 2019.
- Elzanaty S, Malm J, Giwercman A. Duration of sexual abstinence: epididymal and accessory sex gland secretions and their relationship to sperm motility. Hum Reprod 2005; 20(1):221–225. doi:10.1093/humrep/deh586
- Wilcox AJ, Weinberg CR, Baird DD. Timing of sexual intercourse in relation to ovulation. Effects on the probability of conception, survival of the pregnancy, and sex of the baby. N Engl J Med 1995; 333(23):1517–1521. doi:10.1056/NEJM199512073332301
- Practice Committee of the American Society for Reproductive Medicine in collaboration with the Society for Reproductive Endocrinology and Infertility. Optimizing natural fertility: a committee opinion. Fertil Steril 2017; 107(1):52–58. doi:10.1016/j.fertnstert.2016.09.029
- Kutteh WH, Chao CH, Ritter JO, Byrd W. Vaginal lubricants for the infertile couple: effect on sperm activity. Int J Fertil Menopausal Stud 1996; 41(4):400–404. pmid:8894797
- Bigelow JL, Dunson DB, Stanford JB, Ecochard R, Gnoth C, Colombo B. Mucus observations in the fertile window: a better predictor of conception than timing of intercourse. Hum Reprod 2004; 19(4):889–892. doi:10.1093/humrep/deh173
- Steiner AZ, Pritchard D, Stanczyk FZ, et al. Association between biomarkers of ovarian reserve and infertility among older women of reproductive age. JAMA 2017; 318(14):1367–1376. doi:10.1001/jama.2017.14588
- Broekmans FJ, Kwee J, Hendriks DJ, Mol BW, Lambalk CB. A systematic review of tests predicting ovarian reserve and IVF outcome. Hum Reprod Update 2006; 12(6):685–718. doi:10.1093/humupd/dml034
- Practice Committee of the American Society for Reproductive Medicine. Diagnostic evaluation of the infertile female: a committee opinion. Fertil Steril 2015; 103(6):e44–e50. doi:10.1016/j.fertnstert.2015.03.019
- Sharara FI, Scott RT Jr, Seifer DB. The detection of diminished ovarian reserve in infertile women. Am J Obstet Gynecol 1998; 179(3 Pt 1):804–812. pmid:9757994
- American College of Obstetricians and Gynecologists Committee on Gynecologic Practice and Practice Committee. Female age-related fertility decline. Committee Opinion No. 589. Fertil Steril 2014; 101(3):633–634. doi:10.1016/j.fertnstert.2013.12.032
- Balasch J, Gratacós E. Delayed childbearing: effects on fertility and the outcome of pregnancy. Curr Opin Obstet Gynecol 2012; 24(3):187–193. doi:10.1097/GCO.0b013e3283517908
- Hickman LC, Fortin C, Goodman L, Liu X, Flyckt R. Fertility and fertility preservation: knowledge, awareness and attitudes of female graduate students. Eur J Contracept Reprod Health Care 2018; 23(2):130–138. doi:10.1080/13625187.2018.1455085
- Lundsberg LS, Pal L, Gariepy AM, Xu X, Chu MC, Illuzzi JL. Knowledge, attitudes, and practices regarding conception and fertility: a population-based survey among reproductive-age United States women. Fertil Steril 2014; 101(3):767–774. doi:10.1016/j.fertnstert.2013.12.006
- Hodes-Wertz B, Druckenmiller S, Smith M, Noyes N. What do reproductive-age women who undergo oocyte cryopreservation think about the process as a means to preserve fertility? Fertil Steril 2013; 100(5):1343–1349. doi:10.1016/j.fertnstert.2013.07.201
- Weström L, Joesoef R, Reynolds G, Hagdu A, Thompson SE. Pelvic inflammatory disease and fertility. A cohort study of 1,844 women with laparoscopically verified disease and 657 control women with normal laparoscopic results. Sex Transm Dis 1992; 19(4):185–192. pmid:1411832
- ACOG Practice Bulletin No. 195: prevention of infection after gynecologic procedures. Obstet Gynecol 2018; 131(6):e172–e189. doi:10.1097/AOG.0000000000002670
- Balasch J, Creus M, Fábregues F, et al. Visible and non-visible endometriosis at laparoscopy in fertile and infertile women and in patients with chronic pelvic pain: a prospective study. Hum Reprod 1996; 11(2):387–391. pmid:8671229
- Falcone T, Flyckt R. Clinical management of endometriosis. Obstet Gynecol 2018; 131(3):557–571. doi:10.1097/AOG.0000000000002469
- Flyckt R, Kim S, Falcone T. Surgical management of endometriosis in patients with chronic pelvic pain. Semin Reprod Med 2017; 35(1):54–64. doi:10.1055/s-0036-1597306
- Practice Committee of the American Society for Reproductive Medicine. Endometriosis and infertility: a committee opinion. Fertil Steril 2012; 98(3):591–598. doi:10.1016/j.fertnstert.2012.05.031
- Thonneau P, Marchand S, Tallec A, et al. Incidence and main causes of infertility in a resident population (1,850,000) of three French regions (1988–1989). Hum Reprod 1991; 6(6):811–816. pmid:1757519
- Cooper TG, Noonan E, von Eckardstein S, et al. World Health Organization reference values for human semen characteristics. Hum Reprod Update 2010; 16(3):231–245. doi:10.1093/humupd/dmp048
- Practice Committee of American Society for Reproductive Medicine. Diagnostic evaluation of the infertile male: a committee opinion. Fertil Steril 2012; 98(2):294–301. doi:10.1016/j.fertnstert.2012.05.033
- Rotterdam ESHRE/ASRM-Sponsored PCOS consensus workshop group. Revised 2003 consensus on diagnostic criteria and long-term health risks related to polycystic ovary syndrome (PCOS). Hum Reprod 2004; 19(1):41–47. pmid:14688154
- Falcone T, Finegood DT, Fantus IG, Morris D. Androgen response to endogenous insulin secretion during the frequently sampled intravenous glucose tolerance test in normal and hyperandrogenic women. J Clin Endocrinol Metab 1990; 71(6):1653–1657. doi:10.1210/jcem-71-6-1653
- Daniilidis A, Dinas K. Long term health consequences of polycystic ovarian syndrome: a review analysis. Hippokratia 2009; 13(2):90–92. pmid:19561777
- Legro RS, Barnhart HX, Schlaff WD, et al; Cooperative Multicenter Reproductive Medicine Network. Clomiphene, metformin, or both for infertility in the polycystic ovary syndrome. N Engl J Med 2007; 356(6):551–566. doi:10.1056/NEJMoa063971
- Legro RS, Brzyski RG, Diamond MP, et al; NICHD Reproductive Medicine Network. Letrozole versus clomiphene for infertility in the polycystic ovary syndrome. N Engl J Med 2014; 371(2):119–129. doi:10.1056/NEJMoa1313517
For millions of couples, a primary care physician may be the first point of contact for fertility concerns. Statistics from the US Centers for Disease Control and Prevention indicate that 12% of women ages 15 to 44 received fertility services from 2006 to 2010.1 Despite seeking services, most couples requested only advice or testing rather than treatments such as ovulation-inducing medications, surgery, or, rarely, assisted reproductive technologies including in vitro fertilization. Based on these data, primary care physicians are in a unique position to offer guidance and provide fertility services in most circumstances without the need for referral.
This article reviews the answers to questions patients frequently ask, and outlines a practical framework for the evaluation and management of the infertile couple.
MANY PATIENTS SEEK INFORMATION
At least 1 million medical visits per year are for women seeking help in becoming pregnant, with the number increasing over the last several decades.1 Reasons for the increase include delayed childbearing and the effects of aging on the female reproductive system (“female reproductive aging”), as well as the availability of increasingly effective treatments for infertility.
While the prevalence of infertility in US couples is widely quoted as 10% to 15%,2 there is no estimate for the number of fertility-related questions patients routinely pose to care providers. These questions often relate to coital timing, use of lubricants, positioning, and the use of fertility trackers and ovulation predictors.
A 2017 study of women with 12 months of infertility found that only 8% sought subspecialist care vs care from a general physician or provider, indicating that generalists are most often the first point of contact.3 The majority (92%) of women responding to a survey regarding fertility-awareness education indicated a preference for immediate counseling from their general practitioner.4
Although some healthcare providers may consider infertility simply a quality-of-life issue, the World Health Organization classifies it as a disease, and as such it warrants identification, assessment, and intervention.5 Further, patients with infertility are known to experience considerable psychological distress related to their condition. In a comparison study, women with infertility experienced levels of psychological distress similar to the level in patients with cancer and patients with chronic medical illness.6
In the current era, general practitioners and women’s health specialists may also now address patients’ questions about reproductive aging and egg-freezing, which is now an established technology.7
FAILURE TO CONCEIVE AFTER 1 YEAR
As women approach age 40, the potential for fertility decreases rapidly and significantly. Women in their later 30s have only half the fertility of women in their early 20s.10 Misperceptions of aging and female fertility have been fueled by widely publicized celebrity births from women in their 40s and even 50s, without disclosing the use of frozen or donor eggs. This unfortunate fact affects women actively trying to conceive as well as women who wish to delay childbearing due to lack of a partner or for personal or professional reasons. Primary care physicians should be able to provide counseling relevant to female reproductive aging and make suitable and timely referrals for fertility preservation if indicated.
AN EMOTIONAL ISSUE
In approaching the couple with infertility, it is important to proceed with great sensitivity for the socioemotional context of this diagnosis. For both the male and female partner, infertility can be highly stigmatizing, and can be viewed as a personal or relationship failure.
Couples should be encouraged to ask embarrassing or uncomfortable questions. Although this may not be feasible in many circumstances, interviews should ideally be conducted with both partners individually as well as together, to allow sensitive issues to be shared. In some cases, a partner may be unaware of a history of a sexually transmitted infection, a prior abortion, the use of testosterone supplements or medications to enhance male sexual performance, or a vasectomy or tubal ligation during a previous relationship.
It is not unusual that the anxiety of infertility can cause decreased libido and sexual and erectile dysfunction. These issues can further complicate the problem of conceiving, and couples counseling is not uncommonly required.11 Patients are often reassured to know that they are not alone in their diagnosis.
LOOK FOR CLUES
Before embarking on a series of tests, the primary care physician can carefully evaluate for clues that may guide the diagnostic evaluation. The approach can be individualized based on the patient’s age, duration of subfertility (ie, how long they have been trying to become pregnant), and risk factors. But as a general rule, regardless of age, couples who have been trying to conceive for more than 1 year should be encouraged to pursue additional testing.
Because each month presents a new cycle of hope (often followed by intense disappointment), the prevailing sentiment to “just give it a little more time” must be countered by education and counseling. The primary care physician must increase awareness that lack of pregnancy in the stated time periods is a compelling reason for evaluation.
History-taking in the infertile couple should include a complete gynecologic and menstrual history. A history of sexually transmitted diseases that can cause tubal disease, such as gonorrhea and Chlamydia, is significant. Both partners should be assessed for a history of prior conceptions, past medical or surgical problems, medications, and exposures to environmental toxins including alcohol, tobacco, and drugs.
A detailed physical examination can provide clues to the cause of subfertility, especially if signs of obesity, androgen excess, or insulin resistance are present.
QUESTIONS OFTEN ASKED BY COUPLES TRYING TO CONCEIVE
Clinicians are frequently asked questions related to sexual practices and lifestyle in relation to fertility and should be comfortable responding to questions in these areas.
Does frequent ejaculation ‘use up’ my sperm?
Men should be reassured that frequent ejaculations do not decrease sperm counts; even daily ejaculation does not deplete the concentration of sperm. Male partners can be reassured that “saving up” is not an effective strategy; in fact, abstinence periods of greater than 5 days can adversely affect semen parameters.12
How often should we have sex?
Infrequent intercourse (< 1 time per week) reduces the monthly chance of conceiving.13 There does not seem to be a significant improvement in fecundity with daily intercourse vs intercourse on alternate days. Strict schedules surrounding intercourse may increase stress, and reassurance should be offered that intercourse need not be regimented. Every 1 to 2 days should suffice.
Are any sexual positions better for conception?
There is no evidence that particular coital positioning or remaining supine after intercourse improves fertility. Sperm can be found within the endocervix within seconds of ejaculation, irrespective of sexual position.
What is the window of fertility?
There is good evidence that the fertile window lasts approximately 6 days and closes after ovulation.13,14 Women with regular cycles can determine their typical day of ovulation based on menstrual tracking. Intercourse should begin about 6 days before ovulation and should continue every 1 to 2 days for 1 week to fully capture this window.
Should we change our lifestyle?
Couples seeking pregnancy should be advised to limit alcohol and caffeine use, completely abstain from cigarette smoking or illicit drug use, and maintain a healthy body mass index.
Very few data exist to support particular diets or supplements to promote fertility, including antioxidants and herbal remedies. Folic acid supplementation is recommended in all women attempting to conceive to reduce the incidence of birth defects.
Do lubricants reduce fertility?
Although there seem to be no differences in fecundity rates in couples using commercial lubricants, most water-based lubricants are best avoided in couples with infertility, as adverse effects on sperm have been demonstrated in vitro.15 If lubrication is needed, couples may try mineral oil, canola oil, or hydroxyetylcellulose-based lubricants (eg, Pre-seed).
Do fertility trackers work?
Many couples with primary infertility perceive that coital timing is critical and worry that their infertility is due to poorly timed intercourse; in fact, this is seldom the case.
Despite widespread marketing of urinary luteinizing hormone (LH) detection kits and electronic trackers and monitors, there is no clear evidence that these methods improve monthly rates of conception.
Women with a regular menstrual cycle should be encouraged to take notice when their cervical mucus appears clear and slippery (a sign of ovulation). Not all women are able to detect these fluctuations; however, for those who can, observing cervical mucus changes appears to be equivalent or superior to predictor kits in predicting conception.16
A PRACTICAL FRAMEWORK FOR EVALUATING THE INFERTILE COUPLE
To assess for the common factors identified in Table 1, the essential investigation of the infertile couple includes:
- Semen analysis
- Confirmation of ovulation
- Hysterosalpingography.
Consideration can also be given to ovarian reserve testing in women at risk of diminished ovarian reserve. The above investigation can be performed simultaneously to allow for prompt identification of any issues. Further, infertility is often a combination of problems (eg, anovulation in the woman together with a problem in the man), so an incomplete evaluation may overlook a coexisting diagnosis and lead to delays in treatment and pregnancy.
Tests that are no longer typically used in clinical practice are outlined in Table 2.
OVARIAN RESERVE TESTING AND FEMALE REPRODUCTIVE AGING
Ovarian reserve refers to the number of fertilizable oocytes that remain in the ovary. This reserve changes over time, and changes occur rapidly as women approach and enter their 30s. Though not the case in men, the age of the female partner is an independent risk factor for infertility. This discrepancy is due to loss of ovarian reserve, chromosome abnormalities in embryos, and the development of medical conditions with age that affect fertility.
Testing for ovarian reserve does not necessarily predict an overall inability to achieve a live birth,17 but it can predict response to exogenous gonadotropins and, to some degree, the chance for successful pregnancy with assisted reproductive technology.18
The ASRM states that testing for diminished ovarian reserve may provide useful information in women who have had a previous poor response to gonadotropins and in women planning assisted reproductive technology.19 The ASRM also indicates that the following are risk factors for diminished ovarian reserve, and clinicians may target the assessment accordingly19:
- Age 35 or older
- History of exposure to chemotherapy or pelvic radiation
- Family history of early menopause (age < 40)
- History of ovarian surgery
- Unexplained or idiopathic fertility.
Although several tests of ovarian reserve exist, either an antimullerian hormone (AMH) test or a combined cycle day-3 follicle-stimulating hormone (FSH) and estradiol level are the 2 tests commonly used in clinical practice. Antral follicle counts are an ultrasonographic measure used by infertility specialists but rarely by primary care physicians. Assays such as inhibin are rarely ordered and have limited clinical utility.
The AMH test
Many reproductive endocrinologists rely on the AMH level as a single test of ovarian reserve as it is easy to obtain, has a relatively low cost, and offers stable results. AMH is produced by the granulosa cells of the ovarian antral follicles and is readily detected in serum samples.
Conveniently for the clinician, levels of this hormone remain stable throughout the menstrual cycle and therefore can be tested on any day and at any time of day. Lower serum AMH levels (< 1 ng/mL) have been shown to correspond to diminished ovarian stimulation with gonadotropins as well as decreased embryo quality and poor pregnancy outcomes with assisted reproductive technology.19
Nevertheless, despite overall stability, AMH levels can be falsely lowered in women using exogenous hormones or with a diagnosis of hypogonadotropic hypogonadism. Levels may be higher than expected in women with polycystic ovary syndrome due to higher numbers of antral and preantral follicles in the polycystic ovary.
The day-3 follicle-stimulating hormone test
FSH and 17-beta estradiol testing can be ordered in combination to assess function of the hypothalamic-pituitary-ovarian axis on day 3 of the menstrual cycle. There is some flexibility, however, and testing obtained on cycle day 2, 3, or 4 yields equivalent results.
Although there are no strict cutoffs, FSH levels that appear elevated (> 10–20 IU/L) are associated with lower chances of conceiving with in vitro fertilization in multiple studies.20
The test is limited by levels that may fluctuate cycle to cycle, and reassuring test results do not necessarily indicate that a woman will achieve a pregnancy. Although a serum estradiol value alone is not a useful test, it can be used in combination with day-3 FSH to screen for diminished ovarian reserve.
As premature recruitment of a follicle can cause an early follicular rise in estradiol, FSH may be falsely suppressed on day 3. For example, a “normal” day-3 FSH combined with an elevated day-3 17-beta estradiol level of 60 to 80 pg/mL is associated with a poor response to medical treatments for infertility.
Female reproductive aging
Aging of the female reproductive system is a central threat to fertility, and prompt assessment and referral are warranted for women age 35 or older who have been trying to conceive for more than 6 months. The ASRM recommends that women over age 40 be evaluated immediately.21
A prevailing misconception is that regular menstrual cycles correspond with normal fertility. In reality, women lose their ability to achieve a healthy live birth in the 5 to 10 years preceding menopause. Although all women who do not desire pregnancy should still use appropriate contraception to avoid unintended pregnancy, women who do desire pregnancy should be aware of these physiologic changes.
Classic age-related changes in ovarian reserve are accompanied by a steep rise in aneuploidy and miscarriage risk.22 This is particularly relevant as women increasingly delay childbearing in modern society. Loss of fertility begins at 32 and abruptly accelerates at age 3721; this fact is poorly communicated to and understood by patients. In a 2018 study of highly educated women, most respondents failed to identify that 45-year-old women can only rarely achieve a successful pregnancy.23
In recent decades, the percentage of women who delay childbearing until after age 35 has steadily increased. There is a widespread misconception that fertility treatments and assisted reproductive technology can compensate for female reproductive aging. Primary care physicians can play a central role in reminding couples that age remains the single greatest predictor of natural fertility and the chance of success with assisted reproduction.
Further, for women who desire future fertility and are without a partner, primary care physicians can counsel them regarding the availability of donor insemination or egg freezing. Studies confirm that women want clinicians to initiate information on reproductive health, and 80% of women undergoing elective egg-freezing for fertility preservation wished that they had done so at an earlier age.24,25
FEMALE PERITONEAL AND STRUCTURAL CAUSES
Women with endometriosis, fibroids, or a history of tubal disease have impaired fecundity. Pelvic imaging is an essential component of their evaluation. Although hysterosalpingography is the mainstay of tubal assessment, in select cases ultrasonography or hysteroscopy may be indicated.
Tubal disease and hysterosalpingography
Tubal disease remains one of the most common causes of infertility in the US females. In most cases, tubal damage is secondary to pelvic inflammatory disease from infection with gonorrhea or Chlamydia, or both.
Rates of confirmed tubal-factor infertility have been shown to increase with both the severity of the infection and the number of past infections.26 In a landmark study, 1 episode of pelvic inflammatory disease was associated with a 12% risk of tubal-factor infertility, whereas 3 infections carried a risk as high as 54%. Pelvic inflammatory disease is also known to increase the risk of ectopic pregnancy.
To assess tubal patency, hysterosalpingography, a radiographic procedure, is typically performed using fluoroscopy and injected contrast material. Some centers may offer sonohysterography as a radiation-free alternative, depending on sonographic skill and experience. Both tests are best scheduled in the window between the end of menstrual bleeding and ovulation. In practice, patients with regular cycles can typically schedule hysterosalpingography between cycle days 5 and 12.
In patients with known hydrosalpinx (a distended fallopian tube due to blockage) or a history of pelvic infection, doxycycline should be given before the procedure.27 Patients with demonstrated hydrosalpinx on hysterosalpingography should receive doxycycline 100 mg twice daily for 5 days to prevent posthysterosalpingography pelvic inflammatory disease.27 Patients with active pelvic or cervical infection should not undergo hysterosalpingography .
Women with confirmed hydrosalpinx or tubal obstruction can be referred for laparoscopy. Gynecologic surgeons will plan their approach based on whether the obstruction is proximal (near the uterus) or distal (near the ovary) as well as whether hydrosalpinx, abnormal tubal architecture, salpingitis isthmica nodosa, or peritubal adhesions are noted. Tubal surgery can be effective in mild cases of tubal disease; however, as in vitro fertilization is becoming more effective, patients with moderate or severe tubal disease are increasingly being referred directly for assisted reproductive technology. Before undergoing assisted reproductive technology, hydrosalpinx will need to be addressed, as it can decrease clinical pregnancy rates with in vitro fertilization.
Endometriosis
Endometriosis is found in 21% to 47% of women with subfertility28 and commonly causes pain, ovarian cysts, and tubal disease. There is often a delay of 7 to 8 years for diagnosis due to the misapprehension that severe dysmenorrhea is normal. Women with an affected first-degree family member are at substantially increased risk.
Although endometriosis is commonly thought to result from reflux of endometrial tissue into the peritoneal cavity with menses, there are multiple proposed mechanisms for the disease.29 The pathogenesis of endometriosis is enigmatic, and there are likely as yet undetermined immunologic and genetic predispositions that confer increased risk.
Common symptoms of endometriosis are dysmenorrhea, dyspareunia, and pelvic pain, and these are sometimes accompanied by bowel and bladder symptoms. Pelvic examination classically demonstrates an immobile uterus and uterosacral nodularity; palpation of these nodules can elicit pain. On laparoscopy, endometriosis can range from minimal to severe; however, stage of endometriosis correlates poorly with reported symptoms.30
Consideration of surgery is based on clinical history, results of the pelvic examination, and possible findings on ultrasonography or hysterosalpingography. Although positive findings on imaging can support a plan for intervention, endometriosis is largely a peritoneal disease, and evidence of tubal damage or ovarian cysts is rarely evident on ultrasonography. In women with menstrual complaints (eg, dysmenorrhea, heavy menstrual bleeding, abnormal uterine bleeding) and a history of infertility, ultrasonography may be useful in determining the presence of uterine pathology such as ovarian cyst or endometrioma, large hydrosalpinx, polyp, or substantial fibroid burden—any of which may have a significant impact on female fertility.
In the absence of a reliable blood test or imaging study, the gold standard for the diagnosis of endometriosis continues to be laparoscopic surgery. Hormonal treatments for endometriosis symptoms are not effective in improving infertility and will preclude pregnancy. Laparoscopic surgery is more successful in improving pregnancy rates in women with advanced disease: pregnancy rates after surgery can be as high as 60% in women with ovarian endometriomas but are significantly lower in women with removal of minimal to mild disease.30,31 Women over age 35 or who present with low ovarian reserve and whose male partner has semen abnormalities should consider moving directly to assisted reproductive technology rather than pursuing endometriosis surgery.
MALE FACTOR INFERTILITY
Although male partners are often highly engaged in and supportive of the fertility evaluation, some are reluctant to undergo testing, and some wish to undergo semen analysis only after female factors have been ruled out. Our practice is to evaluate male factors immediately, due to the high contribution of male factors (up to 40% of cases) either alone or in combination with female factors.32
Men at particularly increased risk of semen abnormalities include those with a history of chemotherapy or radiation or exposure to toxins (eg, environmental exposures, alcohol, tobacco, illicit substances) and prescribed medications.
At a minimum, for the male partner, a reproductive history should be taken and a semen analysis ordered. Men should be directly queried about testosterone use, as this often-used anabolic steroid hormone can severely impair sperm production.
Men who have low sperm counts, motility, or morphology scores based on World Health Organization criteria should not be deemed “infertile,” as there is significant variation from one analysis to the next, and normal fertility has been reported in men with notably low sperm counts. Particular caution should be exercised in interpreting low morphology scores in men with normal counts and motility, as this parameter appears to have the least prognostic value in this context. Men with abnormal semen analyses should be referred to a specialist for further urologic evaluation and treatment.
Treatments for male factor infertility include surgery, steroid hormones, and possibly intrauterine insemination or assisted reproductive technology. In even the most challenging cases, male infertility is now largely treatable with intracytoplasmic sperm injection with assisted reproductive technology. While most advances in in vitro fertilization have been evolutionary, intracytoplasmic sperm injection was revolutionary. This breakthrough technology allows a single sperm to be injected directly into the oocyte. Sperm for this procedure can be obtained either from the ejaculate or from microsurgical testicular sperm extraction.
ANOVULATION
A thorough menstrual history can be informative, as most females of reproductive age have a fairly predictable 25-to-35-day monthly menstrual cycle. Women presenting with menstrual charting with this pattern do not require laboratory confirmation of ovulation. Basal body temperatures are rarely used currently, as they are time-consuming, can induce stress, and are confirmatory rather than predictive of ovulation. Endometrial biopsy for endometrial “dating” is no longer performed in infertile women.
If laboratory confirmation is desired, LH kit testing with a commercially available test or a luteal phase serum progesterone obtained 7 days after suspected ovulation can be obtained. A serum progesterone level higher than 3 ng/mL is indicative of ovulation.19 Due to the notable fluctuations in ovulatory-appearing progesterone levels over several hours, caution must be taken in interpreting a lower-normal level as indicative of a luteal phase insufficiency.
Polycystic ovary syndrome
Polycystic ovary syndrome is important to understand because it is a metabolic condition that predisposes patients to a variety of health risks. Along with gynecologic consequences such as infertility, abnormal uterine bleeding, and endometrial pathology, it is often accompanied by alterations in glucose and lipid metabolism, obesity, hypertension, and cardiovascular disease.35
Despite its name, the syndrome does not involve the presence of classic ovarian cysts. In fact, the cysts associated with polycystic ovary syndrome are dense accumulations of antral follicles arranged peripherally in the ovarian cortex; they should not be removed surgically as they represent the ovarian reserve.
Although ovaries that appear polycystic on transvaginal ultrasonography are often associated with the syndrome, they are not invariably present and are not absolutely required for the diagnosis of polycystic ovary syndrome based on the most commonly used criteria.35 Several diagnostic criteria have been proposed for polycystic ovary syndrome and its phenotypes. The 2003 revised Rotterdam criteria require 2 out of the following 3 features:
- Oligo-ovulation or anovulation
- Evidence of hyperandrogenism, whether clinical (eg, acne or hirsutism) or based on laboratory testing
- Polycystic-appearing ovaries on ultrasonography.
There is no single test that can diagnose the disease. Although polycystic ovary syndrome is often characterized by elevated LH levels, LH–FSH ratios, and fasting insulin levels, these are not diagnostic criteria. The diagnosis hinges on excluding other causes of anovulation such as thyroid disease, hyperprolactinemia, 21-hydroxylase deficiency, androgen-producing neoplasms, and Cushing syndrome. In addition to checking serum testosterone levels, irregular menstrual cycles and infertility should be assessed at minimum with measurement of TSH, prolactin, and day-3 FSH. Obese women should be screened for metabolic syndrome, which should include an assessment of impaired glucose tolerance with a 2-hour oral glucose tolerance test.36
Women with polycystic ovary syndrome are known to have insulin resistance, which is difficult to assess and is independent of their body mass index.37 They often report a family history of diabetes or a personal history of gestational diabetes or giving birth to infants who are large for gestational age. Although most women diagnosed with insulin resistance and anovulatory infertility will not yet have a diagnosis of diabetes, women with polycystic ovary syndrome are 3 to 7 times more likely to develop type 2 diabetes later in life37 and are at increased risk of lipid abnormalities, cardiovascular disease, and stroke. Therefore, interventions to address the compounding influences of polycystic ovary syndrome and obesity can improve fertility outcomes and help prevent long-term sequelae that accompany the syndrome.
Treatment for women with polycystic ovary syndrome attempting conception includes lifestyle modifications, medications for ovulation induction, and possible use of insulin sensitizers. Metformin alone is not effective as a single agent for achieving pregnancy.38 Diet, weight loss, and exercise can have dramatic effects on ovulation and pregnancy and should be highly encouraged.
Ovulation induction is often required in anovulatory women, either in combination with lifestyle modifications or used subsequently if modifications are not successful. Letrozole is advised as the initial agent in women with obesity and anovulatory infertility rather than clomiphene citrate; a side-by-side comparison demonstrated increased rates of ovulation and live birth with letrozole.39
Once-daily letrozole 2.5 mg or clomiphene 50 mg can be prescribed for 5 days, from cycle days 3 through 7 to cycle days 5 through 9. If this initial dosing fails to result in ovulation, the dose can be increased. Known adverse effects are hot flashes, headaches, ovarian cysts, and increased risk of multiple gestation.
Metformin should be considered as an adjunct to fertility treatments in women with polycystic ovary syndrome, especially those with obesity or impaired glucose tolerance, or if there is no response to standard ovulation induction.
Ovarian hyperstimulation syndrome (cystic enlargement of the ovaries with potentially dangerous fluid and electrolyte imbalances) can occur in women with polycystic ovary syndrome; however, it rarely occurs with oral medications.
For millions of couples, a primary care physician may be the first point of contact for fertility concerns. Statistics from the US Centers for Disease Control and Prevention indicate that 12% of women ages 15 to 44 received fertility services from 2006 to 2010.1 Despite seeking services, most couples requested only advice or testing rather than treatments such as ovulation-inducing medications, surgery, or, rarely, assisted reproductive technologies including in vitro fertilization. Based on these data, primary care physicians are in a unique position to offer guidance and provide fertility services in most circumstances without the need for referral.
This article reviews the answers to questions patients frequently ask, and outlines a practical framework for the evaluation and management of the infertile couple.
MANY PATIENTS SEEK INFORMATION
At least 1 million medical visits per year are for women seeking help in becoming pregnant, with the number increasing over the last several decades.1 Reasons for the increase include delayed childbearing and the effects of aging on the female reproductive system (“female reproductive aging”), as well as the availability of increasingly effective treatments for infertility.
While the prevalence of infertility in US couples is widely quoted as 10% to 15%,2 there is no estimate for the number of fertility-related questions patients routinely pose to care providers. These questions often relate to coital timing, use of lubricants, positioning, and the use of fertility trackers and ovulation predictors.
A 2017 study of women with 12 months of infertility found that only 8% sought subspecialist care vs care from a general physician or provider, indicating that generalists are most often the first point of contact.3 The majority (92%) of women responding to a survey regarding fertility-awareness education indicated a preference for immediate counseling from their general practitioner.4
Although some healthcare providers may consider infertility simply a quality-of-life issue, the World Health Organization classifies it as a disease, and as such it warrants identification, assessment, and intervention.5 Further, patients with infertility are known to experience considerable psychological distress related to their condition. In a comparison study, women with infertility experienced levels of psychological distress similar to the level in patients with cancer and patients with chronic medical illness.6
In the current era, general practitioners and women’s health specialists may also now address patients’ questions about reproductive aging and egg-freezing, which is now an established technology.7
FAILURE TO CONCEIVE AFTER 1 YEAR
As women approach age 40, the potential for fertility decreases rapidly and significantly. Women in their later 30s have only half the fertility of women in their early 20s.10 Misperceptions of aging and female fertility have been fueled by widely publicized celebrity births from women in their 40s and even 50s, without disclosing the use of frozen or donor eggs. This unfortunate fact affects women actively trying to conceive as well as women who wish to delay childbearing due to lack of a partner or for personal or professional reasons. Primary care physicians should be able to provide counseling relevant to female reproductive aging and make suitable and timely referrals for fertility preservation if indicated.
AN EMOTIONAL ISSUE
In approaching the couple with infertility, it is important to proceed with great sensitivity for the socioemotional context of this diagnosis. For both the male and female partner, infertility can be highly stigmatizing, and can be viewed as a personal or relationship failure.
Couples should be encouraged to ask embarrassing or uncomfortable questions. Although this may not be feasible in many circumstances, interviews should ideally be conducted with both partners individually as well as together, to allow sensitive issues to be shared. In some cases, a partner may be unaware of a history of a sexually transmitted infection, a prior abortion, the use of testosterone supplements or medications to enhance male sexual performance, or a vasectomy or tubal ligation during a previous relationship.
It is not unusual that the anxiety of infertility can cause decreased libido and sexual and erectile dysfunction. These issues can further complicate the problem of conceiving, and couples counseling is not uncommonly required.11 Patients are often reassured to know that they are not alone in their diagnosis.
LOOK FOR CLUES
Before embarking on a series of tests, the primary care physician can carefully evaluate for clues that may guide the diagnostic evaluation. The approach can be individualized based on the patient’s age, duration of subfertility (ie, how long they have been trying to become pregnant), and risk factors. But as a general rule, regardless of age, couples who have been trying to conceive for more than 1 year should be encouraged to pursue additional testing.
Because each month presents a new cycle of hope (often followed by intense disappointment), the prevailing sentiment to “just give it a little more time” must be countered by education and counseling. The primary care physician must increase awareness that lack of pregnancy in the stated time periods is a compelling reason for evaluation.
History-taking in the infertile couple should include a complete gynecologic and menstrual history. A history of sexually transmitted diseases that can cause tubal disease, such as gonorrhea and Chlamydia, is significant. Both partners should be assessed for a history of prior conceptions, past medical or surgical problems, medications, and exposures to environmental toxins including alcohol, tobacco, and drugs.
A detailed physical examination can provide clues to the cause of subfertility, especially if signs of obesity, androgen excess, or insulin resistance are present.
QUESTIONS OFTEN ASKED BY COUPLES TRYING TO CONCEIVE
Clinicians are frequently asked questions related to sexual practices and lifestyle in relation to fertility and should be comfortable responding to questions in these areas.
Does frequent ejaculation ‘use up’ my sperm?
Men should be reassured that frequent ejaculations do not decrease sperm counts; even daily ejaculation does not deplete the concentration of sperm. Male partners can be reassured that “saving up” is not an effective strategy; in fact, abstinence periods of greater than 5 days can adversely affect semen parameters.12
How often should we have sex?
Infrequent intercourse (< 1 time per week) reduces the monthly chance of conceiving.13 There does not seem to be a significant improvement in fecundity with daily intercourse vs intercourse on alternate days. Strict schedules surrounding intercourse may increase stress, and reassurance should be offered that intercourse need not be regimented. Every 1 to 2 days should suffice.
Are any sexual positions better for conception?
There is no evidence that particular coital positioning or remaining supine after intercourse improves fertility. Sperm can be found within the endocervix within seconds of ejaculation, irrespective of sexual position.
What is the window of fertility?
There is good evidence that the fertile window lasts approximately 6 days and closes after ovulation.13,14 Women with regular cycles can determine their typical day of ovulation based on menstrual tracking. Intercourse should begin about 6 days before ovulation and should continue every 1 to 2 days for 1 week to fully capture this window.
Should we change our lifestyle?
Couples seeking pregnancy should be advised to limit alcohol and caffeine use, completely abstain from cigarette smoking or illicit drug use, and maintain a healthy body mass index.
Very few data exist to support particular diets or supplements to promote fertility, including antioxidants and herbal remedies. Folic acid supplementation is recommended in all women attempting to conceive to reduce the incidence of birth defects.
Do lubricants reduce fertility?
Although there seem to be no differences in fecundity rates in couples using commercial lubricants, most water-based lubricants are best avoided in couples with infertility, as adverse effects on sperm have been demonstrated in vitro.15 If lubrication is needed, couples may try mineral oil, canola oil, or hydroxyetylcellulose-based lubricants (eg, Pre-seed).
Do fertility trackers work?
Many couples with primary infertility perceive that coital timing is critical and worry that their infertility is due to poorly timed intercourse; in fact, this is seldom the case.
Despite widespread marketing of urinary luteinizing hormone (LH) detection kits and electronic trackers and monitors, there is no clear evidence that these methods improve monthly rates of conception.
Women with a regular menstrual cycle should be encouraged to take notice when their cervical mucus appears clear and slippery (a sign of ovulation). Not all women are able to detect these fluctuations; however, for those who can, observing cervical mucus changes appears to be equivalent or superior to predictor kits in predicting conception.16
A PRACTICAL FRAMEWORK FOR EVALUATING THE INFERTILE COUPLE
To assess for the common factors identified in Table 1, the essential investigation of the infertile couple includes:
- Semen analysis
- Confirmation of ovulation
- Hysterosalpingography.
Consideration can also be given to ovarian reserve testing in women at risk of diminished ovarian reserve. The above investigation can be performed simultaneously to allow for prompt identification of any issues. Further, infertility is often a combination of problems (eg, anovulation in the woman together with a problem in the man), so an incomplete evaluation may overlook a coexisting diagnosis and lead to delays in treatment and pregnancy.
Tests that are no longer typically used in clinical practice are outlined in Table 2.
OVARIAN RESERVE TESTING AND FEMALE REPRODUCTIVE AGING
Ovarian reserve refers to the number of fertilizable oocytes that remain in the ovary. This reserve changes over time, and changes occur rapidly as women approach and enter their 30s. Though not the case in men, the age of the female partner is an independent risk factor for infertility. This discrepancy is due to loss of ovarian reserve, chromosome abnormalities in embryos, and the development of medical conditions with age that affect fertility.
Testing for ovarian reserve does not necessarily predict an overall inability to achieve a live birth,17 but it can predict response to exogenous gonadotropins and, to some degree, the chance for successful pregnancy with assisted reproductive technology.18
The ASRM states that testing for diminished ovarian reserve may provide useful information in women who have had a previous poor response to gonadotropins and in women planning assisted reproductive technology.19 The ASRM also indicates that the following are risk factors for diminished ovarian reserve, and clinicians may target the assessment accordingly19:
- Age 35 or older
- History of exposure to chemotherapy or pelvic radiation
- Family history of early menopause (age < 40)
- History of ovarian surgery
- Unexplained or idiopathic fertility.
Although several tests of ovarian reserve exist, either an antimullerian hormone (AMH) test or a combined cycle day-3 follicle-stimulating hormone (FSH) and estradiol level are the 2 tests commonly used in clinical practice. Antral follicle counts are an ultrasonographic measure used by infertility specialists but rarely by primary care physicians. Assays such as inhibin are rarely ordered and have limited clinical utility.
The AMH test
Many reproductive endocrinologists rely on the AMH level as a single test of ovarian reserve as it is easy to obtain, has a relatively low cost, and offers stable results. AMH is produced by the granulosa cells of the ovarian antral follicles and is readily detected in serum samples.
Conveniently for the clinician, levels of this hormone remain stable throughout the menstrual cycle and therefore can be tested on any day and at any time of day. Lower serum AMH levels (< 1 ng/mL) have been shown to correspond to diminished ovarian stimulation with gonadotropins as well as decreased embryo quality and poor pregnancy outcomes with assisted reproductive technology.19
Nevertheless, despite overall stability, AMH levels can be falsely lowered in women using exogenous hormones or with a diagnosis of hypogonadotropic hypogonadism. Levels may be higher than expected in women with polycystic ovary syndrome due to higher numbers of antral and preantral follicles in the polycystic ovary.
The day-3 follicle-stimulating hormone test
FSH and 17-beta estradiol testing can be ordered in combination to assess function of the hypothalamic-pituitary-ovarian axis on day 3 of the menstrual cycle. There is some flexibility, however, and testing obtained on cycle day 2, 3, or 4 yields equivalent results.
Although there are no strict cutoffs, FSH levels that appear elevated (> 10–20 IU/L) are associated with lower chances of conceiving with in vitro fertilization in multiple studies.20
The test is limited by levels that may fluctuate cycle to cycle, and reassuring test results do not necessarily indicate that a woman will achieve a pregnancy. Although a serum estradiol value alone is not a useful test, it can be used in combination with day-3 FSH to screen for diminished ovarian reserve.
As premature recruitment of a follicle can cause an early follicular rise in estradiol, FSH may be falsely suppressed on day 3. For example, a “normal” day-3 FSH combined with an elevated day-3 17-beta estradiol level of 60 to 80 pg/mL is associated with a poor response to medical treatments for infertility.
Female reproductive aging
Aging of the female reproductive system is a central threat to fertility, and prompt assessment and referral are warranted for women age 35 or older who have been trying to conceive for more than 6 months. The ASRM recommends that women over age 40 be evaluated immediately.21
A prevailing misconception is that regular menstrual cycles correspond with normal fertility. In reality, women lose their ability to achieve a healthy live birth in the 5 to 10 years preceding menopause. Although all women who do not desire pregnancy should still use appropriate contraception to avoid unintended pregnancy, women who do desire pregnancy should be aware of these physiologic changes.
Classic age-related changes in ovarian reserve are accompanied by a steep rise in aneuploidy and miscarriage risk.22 This is particularly relevant as women increasingly delay childbearing in modern society. Loss of fertility begins at 32 and abruptly accelerates at age 3721; this fact is poorly communicated to and understood by patients. In a 2018 study of highly educated women, most respondents failed to identify that 45-year-old women can only rarely achieve a successful pregnancy.23
In recent decades, the percentage of women who delay childbearing until after age 35 has steadily increased. There is a widespread misconception that fertility treatments and assisted reproductive technology can compensate for female reproductive aging. Primary care physicians can play a central role in reminding couples that age remains the single greatest predictor of natural fertility and the chance of success with assisted reproduction.
Further, for women who desire future fertility and are without a partner, primary care physicians can counsel them regarding the availability of donor insemination or egg freezing. Studies confirm that women want clinicians to initiate information on reproductive health, and 80% of women undergoing elective egg-freezing for fertility preservation wished that they had done so at an earlier age.24,25
FEMALE PERITONEAL AND STRUCTURAL CAUSES
Women with endometriosis, fibroids, or a history of tubal disease have impaired fecundity. Pelvic imaging is an essential component of their evaluation. Although hysterosalpingography is the mainstay of tubal assessment, in select cases ultrasonography or hysteroscopy may be indicated.
Tubal disease and hysterosalpingography
Tubal disease remains one of the most common causes of infertility in the US females. In most cases, tubal damage is secondary to pelvic inflammatory disease from infection with gonorrhea or Chlamydia, or both.
Rates of confirmed tubal-factor infertility have been shown to increase with both the severity of the infection and the number of past infections.26 In a landmark study, 1 episode of pelvic inflammatory disease was associated with a 12% risk of tubal-factor infertility, whereas 3 infections carried a risk as high as 54%. Pelvic inflammatory disease is also known to increase the risk of ectopic pregnancy.
To assess tubal patency, hysterosalpingography, a radiographic procedure, is typically performed using fluoroscopy and injected contrast material. Some centers may offer sonohysterography as a radiation-free alternative, depending on sonographic skill and experience. Both tests are best scheduled in the window between the end of menstrual bleeding and ovulation. In practice, patients with regular cycles can typically schedule hysterosalpingography between cycle days 5 and 12.
In patients with known hydrosalpinx (a distended fallopian tube due to blockage) or a history of pelvic infection, doxycycline should be given before the procedure.27 Patients with demonstrated hydrosalpinx on hysterosalpingography should receive doxycycline 100 mg twice daily for 5 days to prevent posthysterosalpingography pelvic inflammatory disease.27 Patients with active pelvic or cervical infection should not undergo hysterosalpingography .
Women with confirmed hydrosalpinx or tubal obstruction can be referred for laparoscopy. Gynecologic surgeons will plan their approach based on whether the obstruction is proximal (near the uterus) or distal (near the ovary) as well as whether hydrosalpinx, abnormal tubal architecture, salpingitis isthmica nodosa, or peritubal adhesions are noted. Tubal surgery can be effective in mild cases of tubal disease; however, as in vitro fertilization is becoming more effective, patients with moderate or severe tubal disease are increasingly being referred directly for assisted reproductive technology. Before undergoing assisted reproductive technology, hydrosalpinx will need to be addressed, as it can decrease clinical pregnancy rates with in vitro fertilization.
Endometriosis
Endometriosis is found in 21% to 47% of women with subfertility28 and commonly causes pain, ovarian cysts, and tubal disease. There is often a delay of 7 to 8 years for diagnosis due to the misapprehension that severe dysmenorrhea is normal. Women with an affected first-degree family member are at substantially increased risk.
Although endometriosis is commonly thought to result from reflux of endometrial tissue into the peritoneal cavity with menses, there are multiple proposed mechanisms for the disease.29 The pathogenesis of endometriosis is enigmatic, and there are likely as yet undetermined immunologic and genetic predispositions that confer increased risk.
Common symptoms of endometriosis are dysmenorrhea, dyspareunia, and pelvic pain, and these are sometimes accompanied by bowel and bladder symptoms. Pelvic examination classically demonstrates an immobile uterus and uterosacral nodularity; palpation of these nodules can elicit pain. On laparoscopy, endometriosis can range from minimal to severe; however, stage of endometriosis correlates poorly with reported symptoms.30
Consideration of surgery is based on clinical history, results of the pelvic examination, and possible findings on ultrasonography or hysterosalpingography. Although positive findings on imaging can support a plan for intervention, endometriosis is largely a peritoneal disease, and evidence of tubal damage or ovarian cysts is rarely evident on ultrasonography. In women with menstrual complaints (eg, dysmenorrhea, heavy menstrual bleeding, abnormal uterine bleeding) and a history of infertility, ultrasonography may be useful in determining the presence of uterine pathology such as ovarian cyst or endometrioma, large hydrosalpinx, polyp, or substantial fibroid burden—any of which may have a significant impact on female fertility.
In the absence of a reliable blood test or imaging study, the gold standard for the diagnosis of endometriosis continues to be laparoscopic surgery. Hormonal treatments for endometriosis symptoms are not effective in improving infertility and will preclude pregnancy. Laparoscopic surgery is more successful in improving pregnancy rates in women with advanced disease: pregnancy rates after surgery can be as high as 60% in women with ovarian endometriomas but are significantly lower in women with removal of minimal to mild disease.30,31 Women over age 35 or who present with low ovarian reserve and whose male partner has semen abnormalities should consider moving directly to assisted reproductive technology rather than pursuing endometriosis surgery.
MALE FACTOR INFERTILITY
Although male partners are often highly engaged in and supportive of the fertility evaluation, some are reluctant to undergo testing, and some wish to undergo semen analysis only after female factors have been ruled out. Our practice is to evaluate male factors immediately, due to the high contribution of male factors (up to 40% of cases) either alone or in combination with female factors.32
Men at particularly increased risk of semen abnormalities include those with a history of chemotherapy or radiation or exposure to toxins (eg, environmental exposures, alcohol, tobacco, illicit substances) and prescribed medications.
At a minimum, for the male partner, a reproductive history should be taken and a semen analysis ordered. Men should be directly queried about testosterone use, as this often-used anabolic steroid hormone can severely impair sperm production.
Men who have low sperm counts, motility, or morphology scores based on World Health Organization criteria should not be deemed “infertile,” as there is significant variation from one analysis to the next, and normal fertility has been reported in men with notably low sperm counts. Particular caution should be exercised in interpreting low morphology scores in men with normal counts and motility, as this parameter appears to have the least prognostic value in this context. Men with abnormal semen analyses should be referred to a specialist for further urologic evaluation and treatment.
Treatments for male factor infertility include surgery, steroid hormones, and possibly intrauterine insemination or assisted reproductive technology. In even the most challenging cases, male infertility is now largely treatable with intracytoplasmic sperm injection with assisted reproductive technology. While most advances in in vitro fertilization have been evolutionary, intracytoplasmic sperm injection was revolutionary. This breakthrough technology allows a single sperm to be injected directly into the oocyte. Sperm for this procedure can be obtained either from the ejaculate or from microsurgical testicular sperm extraction.
ANOVULATION
A thorough menstrual history can be informative, as most females of reproductive age have a fairly predictable 25-to-35-day monthly menstrual cycle. Women presenting with menstrual charting with this pattern do not require laboratory confirmation of ovulation. Basal body temperatures are rarely used currently, as they are time-consuming, can induce stress, and are confirmatory rather than predictive of ovulation. Endometrial biopsy for endometrial “dating” is no longer performed in infertile women.
If laboratory confirmation is desired, LH kit testing with a commercially available test or a luteal phase serum progesterone obtained 7 days after suspected ovulation can be obtained. A serum progesterone level higher than 3 ng/mL is indicative of ovulation.19 Due to the notable fluctuations in ovulatory-appearing progesterone levels over several hours, caution must be taken in interpreting a lower-normal level as indicative of a luteal phase insufficiency.
Polycystic ovary syndrome
Polycystic ovary syndrome is important to understand because it is a metabolic condition that predisposes patients to a variety of health risks. Along with gynecologic consequences such as infertility, abnormal uterine bleeding, and endometrial pathology, it is often accompanied by alterations in glucose and lipid metabolism, obesity, hypertension, and cardiovascular disease.35
Despite its name, the syndrome does not involve the presence of classic ovarian cysts. In fact, the cysts associated with polycystic ovary syndrome are dense accumulations of antral follicles arranged peripherally in the ovarian cortex; they should not be removed surgically as they represent the ovarian reserve.
Although ovaries that appear polycystic on transvaginal ultrasonography are often associated with the syndrome, they are not invariably present and are not absolutely required for the diagnosis of polycystic ovary syndrome based on the most commonly used criteria.35 Several diagnostic criteria have been proposed for polycystic ovary syndrome and its phenotypes. The 2003 revised Rotterdam criteria require 2 out of the following 3 features:
- Oligo-ovulation or anovulation
- Evidence of hyperandrogenism, whether clinical (eg, acne or hirsutism) or based on laboratory testing
- Polycystic-appearing ovaries on ultrasonography.
There is no single test that can diagnose the disease. Although polycystic ovary syndrome is often characterized by elevated LH levels, LH–FSH ratios, and fasting insulin levels, these are not diagnostic criteria. The diagnosis hinges on excluding other causes of anovulation such as thyroid disease, hyperprolactinemia, 21-hydroxylase deficiency, androgen-producing neoplasms, and Cushing syndrome. In addition to checking serum testosterone levels, irregular menstrual cycles and infertility should be assessed at minimum with measurement of TSH, prolactin, and day-3 FSH. Obese women should be screened for metabolic syndrome, which should include an assessment of impaired glucose tolerance with a 2-hour oral glucose tolerance test.36
Women with polycystic ovary syndrome are known to have insulin resistance, which is difficult to assess and is independent of their body mass index.37 They often report a family history of diabetes or a personal history of gestational diabetes or giving birth to infants who are large for gestational age. Although most women diagnosed with insulin resistance and anovulatory infertility will not yet have a diagnosis of diabetes, women with polycystic ovary syndrome are 3 to 7 times more likely to develop type 2 diabetes later in life37 and are at increased risk of lipid abnormalities, cardiovascular disease, and stroke. Therefore, interventions to address the compounding influences of polycystic ovary syndrome and obesity can improve fertility outcomes and help prevent long-term sequelae that accompany the syndrome.
Treatment for women with polycystic ovary syndrome attempting conception includes lifestyle modifications, medications for ovulation induction, and possible use of insulin sensitizers. Metformin alone is not effective as a single agent for achieving pregnancy.38 Diet, weight loss, and exercise can have dramatic effects on ovulation and pregnancy and should be highly encouraged.
Ovulation induction is often required in anovulatory women, either in combination with lifestyle modifications or used subsequently if modifications are not successful. Letrozole is advised as the initial agent in women with obesity and anovulatory infertility rather than clomiphene citrate; a side-by-side comparison demonstrated increased rates of ovulation and live birth with letrozole.39
Once-daily letrozole 2.5 mg or clomiphene 50 mg can be prescribed for 5 days, from cycle days 3 through 7 to cycle days 5 through 9. If this initial dosing fails to result in ovulation, the dose can be increased. Known adverse effects are hot flashes, headaches, ovarian cysts, and increased risk of multiple gestation.
Metformin should be considered as an adjunct to fertility treatments in women with polycystic ovary syndrome, especially those with obesity or impaired glucose tolerance, or if there is no response to standard ovulation induction.
Ovarian hyperstimulation syndrome (cystic enlargement of the ovaries with potentially dangerous fluid and electrolyte imbalances) can occur in women with polycystic ovary syndrome; however, it rarely occurs with oral medications.
- Chandra A, Copen CE, Stephen EH. Infertility service use in the United States: data from the National Survey of Family Growth, 1982–2010. Natl Health Stat Report 2014; (73):1–21. pmid:24467919
- Mosher WD, Pratt WF. Fecundity and infertility in the United States: incidence and trends. Fertil Steril 1991; 56(2):192–193. pmid:2070846
- Boltz MW, Sanders JN, Simonsen SE, Stanford JB. Fertility treatment, use of in vitro fertilization, and time to live birth based on initial provider type. J Am Board Fam Med 2017; 30(2):230–238. doi:10.3122/jabfm.2017.02.160184
- Hampton K, Mazza D. Fertility-awareness knowledge, attitudes and practices of women attending general practice. Aust Fam Physician 2015; 44(11):840–845. pmid:26590626
- Zegers-Hochschild F, Adamson GD, de Mouzon J, et al; International Committee for Monitoring Assisted Reproductive Technology; World Health Organization. International Committee for Monitoring Assisted Reproductive Technology (ICMART) and the World Health Organization (WHO) revised glossary of ART terminology, 2009. Fertil Steril 2009; 92(5):1520–1524. doi:10.1016/j.fertnstert.2009.09.009
- Domar AD, Zuttermeister PC, Friedman R. The psychological impact of infertility: a comparison with patients with other medical conditions. J Psychosom Obstet Gynaecol 1993; 14(suppl):45–52. pmid:8142988
- Argyle CE, Harper JC, Davies MC. Oocyte cryopreservation: where are we now? Hum Reprod Update 2016; 22(4):440–449. doi:10.1093/humupd/dmw007
- Practice Committee of American Society for Reproductive Medicine. Definitions of infertility and recurrent pregnancy loss: a committee opinion. Fertil Steril 2013; 99(1):63. doi:10.1016/j.fertnstert.2012.09.023
- Guttmacher AF. Factors affecting normal expectancy of conception. J Am Med Assoc 1956; 161(9):855–860. pmid:13319020
- Dunson DB, Baird DD, Colombo B. Increased infertility with age in men and women. Obstet Gynecol 2004; 103(1):51–56. doi:10.1097/01.AOG.0000100153.24061.45
- National Collaborating Centre for Women’s and Children’s Health (UK). Fertility: assessment and treatment for people with fertility problems. London: Royal College of Obstetricians & Gynaecologists; 2013. www.ncbi.nlm.nih.gov/books/NBK247932. Accessed May 6, 2019.
- Elzanaty S, Malm J, Giwercman A. Duration of sexual abstinence: epididymal and accessory sex gland secretions and their relationship to sperm motility. Hum Reprod 2005; 20(1):221–225. doi:10.1093/humrep/deh586
- Wilcox AJ, Weinberg CR, Baird DD. Timing of sexual intercourse in relation to ovulation. Effects on the probability of conception, survival of the pregnancy, and sex of the baby. N Engl J Med 1995; 333(23):1517–1521. doi:10.1056/NEJM199512073332301
- Practice Committee of the American Society for Reproductive Medicine in collaboration with the Society for Reproductive Endocrinology and Infertility. Optimizing natural fertility: a committee opinion. Fertil Steril 2017; 107(1):52–58. doi:10.1016/j.fertnstert.2016.09.029
- Kutteh WH, Chao CH, Ritter JO, Byrd W. Vaginal lubricants for the infertile couple: effect on sperm activity. Int J Fertil Menopausal Stud 1996; 41(4):400–404. pmid:8894797
- Bigelow JL, Dunson DB, Stanford JB, Ecochard R, Gnoth C, Colombo B. Mucus observations in the fertile window: a better predictor of conception than timing of intercourse. Hum Reprod 2004; 19(4):889–892. doi:10.1093/humrep/deh173
- Steiner AZ, Pritchard D, Stanczyk FZ, et al. Association between biomarkers of ovarian reserve and infertility among older women of reproductive age. JAMA 2017; 318(14):1367–1376. doi:10.1001/jama.2017.14588
- Broekmans FJ, Kwee J, Hendriks DJ, Mol BW, Lambalk CB. A systematic review of tests predicting ovarian reserve and IVF outcome. Hum Reprod Update 2006; 12(6):685–718. doi:10.1093/humupd/dml034
- Practice Committee of the American Society for Reproductive Medicine. Diagnostic evaluation of the infertile female: a committee opinion. Fertil Steril 2015; 103(6):e44–e50. doi:10.1016/j.fertnstert.2015.03.019
- Sharara FI, Scott RT Jr, Seifer DB. The detection of diminished ovarian reserve in infertile women. Am J Obstet Gynecol 1998; 179(3 Pt 1):804–812. pmid:9757994
- American College of Obstetricians and Gynecologists Committee on Gynecologic Practice and Practice Committee. Female age-related fertility decline. Committee Opinion No. 589. Fertil Steril 2014; 101(3):633–634. doi:10.1016/j.fertnstert.2013.12.032
- Balasch J, Gratacós E. Delayed childbearing: effects on fertility and the outcome of pregnancy. Curr Opin Obstet Gynecol 2012; 24(3):187–193. doi:10.1097/GCO.0b013e3283517908
- Hickman LC, Fortin C, Goodman L, Liu X, Flyckt R. Fertility and fertility preservation: knowledge, awareness and attitudes of female graduate students. Eur J Contracept Reprod Health Care 2018; 23(2):130–138. doi:10.1080/13625187.2018.1455085
- Lundsberg LS, Pal L, Gariepy AM, Xu X, Chu MC, Illuzzi JL. Knowledge, attitudes, and practices regarding conception and fertility: a population-based survey among reproductive-age United States women. Fertil Steril 2014; 101(3):767–774. doi:10.1016/j.fertnstert.2013.12.006
- Hodes-Wertz B, Druckenmiller S, Smith M, Noyes N. What do reproductive-age women who undergo oocyte cryopreservation think about the process as a means to preserve fertility? Fertil Steril 2013; 100(5):1343–1349. doi:10.1016/j.fertnstert.2013.07.201
- Weström L, Joesoef R, Reynolds G, Hagdu A, Thompson SE. Pelvic inflammatory disease and fertility. A cohort study of 1,844 women with laparoscopically verified disease and 657 control women with normal laparoscopic results. Sex Transm Dis 1992; 19(4):185–192. pmid:1411832
- ACOG Practice Bulletin No. 195: prevention of infection after gynecologic procedures. Obstet Gynecol 2018; 131(6):e172–e189. doi:10.1097/AOG.0000000000002670
- Balasch J, Creus M, Fábregues F, et al. Visible and non-visible endometriosis at laparoscopy in fertile and infertile women and in patients with chronic pelvic pain: a prospective study. Hum Reprod 1996; 11(2):387–391. pmid:8671229
- Falcone T, Flyckt R. Clinical management of endometriosis. Obstet Gynecol 2018; 131(3):557–571. doi:10.1097/AOG.0000000000002469
- Flyckt R, Kim S, Falcone T. Surgical management of endometriosis in patients with chronic pelvic pain. Semin Reprod Med 2017; 35(1):54–64. doi:10.1055/s-0036-1597306
- Practice Committee of the American Society for Reproductive Medicine. Endometriosis and infertility: a committee opinion. Fertil Steril 2012; 98(3):591–598. doi:10.1016/j.fertnstert.2012.05.031
- Thonneau P, Marchand S, Tallec A, et al. Incidence and main causes of infertility in a resident population (1,850,000) of three French regions (1988–1989). Hum Reprod 1991; 6(6):811–816. pmid:1757519
- Cooper TG, Noonan E, von Eckardstein S, et al. World Health Organization reference values for human semen characteristics. Hum Reprod Update 2010; 16(3):231–245. doi:10.1093/humupd/dmp048
- Practice Committee of American Society for Reproductive Medicine. Diagnostic evaluation of the infertile male: a committee opinion. Fertil Steril 2012; 98(2):294–301. doi:10.1016/j.fertnstert.2012.05.033
- Rotterdam ESHRE/ASRM-Sponsored PCOS consensus workshop group. Revised 2003 consensus on diagnostic criteria and long-term health risks related to polycystic ovary syndrome (PCOS). Hum Reprod 2004; 19(1):41–47. pmid:14688154
- Falcone T, Finegood DT, Fantus IG, Morris D. Androgen response to endogenous insulin secretion during the frequently sampled intravenous glucose tolerance test in normal and hyperandrogenic women. J Clin Endocrinol Metab 1990; 71(6):1653–1657. doi:10.1210/jcem-71-6-1653
- Daniilidis A, Dinas K. Long term health consequences of polycystic ovarian syndrome: a review analysis. Hippokratia 2009; 13(2):90–92. pmid:19561777
- Legro RS, Barnhart HX, Schlaff WD, et al; Cooperative Multicenter Reproductive Medicine Network. Clomiphene, metformin, or both for infertility in the polycystic ovary syndrome. N Engl J Med 2007; 356(6):551–566. doi:10.1056/NEJMoa063971
- Legro RS, Brzyski RG, Diamond MP, et al; NICHD Reproductive Medicine Network. Letrozole versus clomiphene for infertility in the polycystic ovary syndrome. N Engl J Med 2014; 371(2):119–129. doi:10.1056/NEJMoa1313517
- Chandra A, Copen CE, Stephen EH. Infertility service use in the United States: data from the National Survey of Family Growth, 1982–2010. Natl Health Stat Report 2014; (73):1–21. pmid:24467919
- Mosher WD, Pratt WF. Fecundity and infertility in the United States: incidence and trends. Fertil Steril 1991; 56(2):192–193. pmid:2070846
- Boltz MW, Sanders JN, Simonsen SE, Stanford JB. Fertility treatment, use of in vitro fertilization, and time to live birth based on initial provider type. J Am Board Fam Med 2017; 30(2):230–238. doi:10.3122/jabfm.2017.02.160184
- Hampton K, Mazza D. Fertility-awareness knowledge, attitudes and practices of women attending general practice. Aust Fam Physician 2015; 44(11):840–845. pmid:26590626
- Zegers-Hochschild F, Adamson GD, de Mouzon J, et al; International Committee for Monitoring Assisted Reproductive Technology; World Health Organization. International Committee for Monitoring Assisted Reproductive Technology (ICMART) and the World Health Organization (WHO) revised glossary of ART terminology, 2009. Fertil Steril 2009; 92(5):1520–1524. doi:10.1016/j.fertnstert.2009.09.009
- Domar AD, Zuttermeister PC, Friedman R. The psychological impact of infertility: a comparison with patients with other medical conditions. J Psychosom Obstet Gynaecol 1993; 14(suppl):45–52. pmid:8142988
- Argyle CE, Harper JC, Davies MC. Oocyte cryopreservation: where are we now? Hum Reprod Update 2016; 22(4):440–449. doi:10.1093/humupd/dmw007
- Practice Committee of American Society for Reproductive Medicine. Definitions of infertility and recurrent pregnancy loss: a committee opinion. Fertil Steril 2013; 99(1):63. doi:10.1016/j.fertnstert.2012.09.023
- Guttmacher AF. Factors affecting normal expectancy of conception. J Am Med Assoc 1956; 161(9):855–860. pmid:13319020
- Dunson DB, Baird DD, Colombo B. Increased infertility with age in men and women. Obstet Gynecol 2004; 103(1):51–56. doi:10.1097/01.AOG.0000100153.24061.45
- National Collaborating Centre for Women’s and Children’s Health (UK). Fertility: assessment and treatment for people with fertility problems. London: Royal College of Obstetricians & Gynaecologists; 2013. www.ncbi.nlm.nih.gov/books/NBK247932. Accessed May 6, 2019.
- Elzanaty S, Malm J, Giwercman A. Duration of sexual abstinence: epididymal and accessory sex gland secretions and their relationship to sperm motility. Hum Reprod 2005; 20(1):221–225. doi:10.1093/humrep/deh586
- Wilcox AJ, Weinberg CR, Baird DD. Timing of sexual intercourse in relation to ovulation. Effects on the probability of conception, survival of the pregnancy, and sex of the baby. N Engl J Med 1995; 333(23):1517–1521. doi:10.1056/NEJM199512073332301
- Practice Committee of the American Society for Reproductive Medicine in collaboration with the Society for Reproductive Endocrinology and Infertility. Optimizing natural fertility: a committee opinion. Fertil Steril 2017; 107(1):52–58. doi:10.1016/j.fertnstert.2016.09.029
- Kutteh WH, Chao CH, Ritter JO, Byrd W. Vaginal lubricants for the infertile couple: effect on sperm activity. Int J Fertil Menopausal Stud 1996; 41(4):400–404. pmid:8894797
- Bigelow JL, Dunson DB, Stanford JB, Ecochard R, Gnoth C, Colombo B. Mucus observations in the fertile window: a better predictor of conception than timing of intercourse. Hum Reprod 2004; 19(4):889–892. doi:10.1093/humrep/deh173
- Steiner AZ, Pritchard D, Stanczyk FZ, et al. Association between biomarkers of ovarian reserve and infertility among older women of reproductive age. JAMA 2017; 318(14):1367–1376. doi:10.1001/jama.2017.14588
- Broekmans FJ, Kwee J, Hendriks DJ, Mol BW, Lambalk CB. A systematic review of tests predicting ovarian reserve and IVF outcome. Hum Reprod Update 2006; 12(6):685–718. doi:10.1093/humupd/dml034
- Practice Committee of the American Society for Reproductive Medicine. Diagnostic evaluation of the infertile female: a committee opinion. Fertil Steril 2015; 103(6):e44–e50. doi:10.1016/j.fertnstert.2015.03.019
- Sharara FI, Scott RT Jr, Seifer DB. The detection of diminished ovarian reserve in infertile women. Am J Obstet Gynecol 1998; 179(3 Pt 1):804–812. pmid:9757994
- American College of Obstetricians and Gynecologists Committee on Gynecologic Practice and Practice Committee. Female age-related fertility decline. Committee Opinion No. 589. Fertil Steril 2014; 101(3):633–634. doi:10.1016/j.fertnstert.2013.12.032
- Balasch J, Gratacós E. Delayed childbearing: effects on fertility and the outcome of pregnancy. Curr Opin Obstet Gynecol 2012; 24(3):187–193. doi:10.1097/GCO.0b013e3283517908
- Hickman LC, Fortin C, Goodman L, Liu X, Flyckt R. Fertility and fertility preservation: knowledge, awareness and attitudes of female graduate students. Eur J Contracept Reprod Health Care 2018; 23(2):130–138. doi:10.1080/13625187.2018.1455085
- Lundsberg LS, Pal L, Gariepy AM, Xu X, Chu MC, Illuzzi JL. Knowledge, attitudes, and practices regarding conception and fertility: a population-based survey among reproductive-age United States women. Fertil Steril 2014; 101(3):767–774. doi:10.1016/j.fertnstert.2013.12.006
- Hodes-Wertz B, Druckenmiller S, Smith M, Noyes N. What do reproductive-age women who undergo oocyte cryopreservation think about the process as a means to preserve fertility? Fertil Steril 2013; 100(5):1343–1349. doi:10.1016/j.fertnstert.2013.07.201
- Weström L, Joesoef R, Reynolds G, Hagdu A, Thompson SE. Pelvic inflammatory disease and fertility. A cohort study of 1,844 women with laparoscopically verified disease and 657 control women with normal laparoscopic results. Sex Transm Dis 1992; 19(4):185–192. pmid:1411832
- ACOG Practice Bulletin No. 195: prevention of infection after gynecologic procedures. Obstet Gynecol 2018; 131(6):e172–e189. doi:10.1097/AOG.0000000000002670
- Balasch J, Creus M, Fábregues F, et al. Visible and non-visible endometriosis at laparoscopy in fertile and infertile women and in patients with chronic pelvic pain: a prospective study. Hum Reprod 1996; 11(2):387–391. pmid:8671229
- Falcone T, Flyckt R. Clinical management of endometriosis. Obstet Gynecol 2018; 131(3):557–571. doi:10.1097/AOG.0000000000002469
- Flyckt R, Kim S, Falcone T. Surgical management of endometriosis in patients with chronic pelvic pain. Semin Reprod Med 2017; 35(1):54–64. doi:10.1055/s-0036-1597306
- Practice Committee of the American Society for Reproductive Medicine. Endometriosis and infertility: a committee opinion. Fertil Steril 2012; 98(3):591–598. doi:10.1016/j.fertnstert.2012.05.031
- Thonneau P, Marchand S, Tallec A, et al. Incidence and main causes of infertility in a resident population (1,850,000) of three French regions (1988–1989). Hum Reprod 1991; 6(6):811–816. pmid:1757519
- Cooper TG, Noonan E, von Eckardstein S, et al. World Health Organization reference values for human semen characteristics. Hum Reprod Update 2010; 16(3):231–245. doi:10.1093/humupd/dmp048
- Practice Committee of American Society for Reproductive Medicine. Diagnostic evaluation of the infertile male: a committee opinion. Fertil Steril 2012; 98(2):294–301. doi:10.1016/j.fertnstert.2012.05.033
- Rotterdam ESHRE/ASRM-Sponsored PCOS consensus workshop group. Revised 2003 consensus on diagnostic criteria and long-term health risks related to polycystic ovary syndrome (PCOS). Hum Reprod 2004; 19(1):41–47. pmid:14688154
- Falcone T, Finegood DT, Fantus IG, Morris D. Androgen response to endogenous insulin secretion during the frequently sampled intravenous glucose tolerance test in normal and hyperandrogenic women. J Clin Endocrinol Metab 1990; 71(6):1653–1657. doi:10.1210/jcem-71-6-1653
- Daniilidis A, Dinas K. Long term health consequences of polycystic ovarian syndrome: a review analysis. Hippokratia 2009; 13(2):90–92. pmid:19561777
- Legro RS, Barnhart HX, Schlaff WD, et al; Cooperative Multicenter Reproductive Medicine Network. Clomiphene, metformin, or both for infertility in the polycystic ovary syndrome. N Engl J Med 2007; 356(6):551–566. doi:10.1056/NEJMoa063971
- Legro RS, Brzyski RG, Diamond MP, et al; NICHD Reproductive Medicine Network. Letrozole versus clomiphene for infertility in the polycystic ovary syndrome. N Engl J Med 2014; 371(2):119–129. doi:10.1056/NEJMoa1313517
KEY POINTS
- A primary care physician can provide advice and testing regarding most fertility concerns.
- Female reproductive aging is a central threat to fertility, and prompt assessment and referral are warranted for women age 35 and older.
- Male factor infertility can now often be overcome with assisted reproductive technologies.
- Polycystic ovary syndrome can cause anovulation and has metabolic effects that can evolve into metabolic syndrome, with serious health consequences.
Type 2 diabetes: Evolving concepts and treatment
Insights from basic and clinical research are changing the way we treat diabetes mellitus. In 2016, several key diabetes organizations, ie, the American Diabetes Association (ADA), the Juvenile Diabetes Research Foundation (JDRF), the European Association for the Study of Diabetes (EASD), and the American Association of Clinical Endocrinologists (AACE), called for bringing therapeutic approaches in line with our updated understanding of disease pathophysiology, replacing “one-size-fits-all” management with a tailored approach.1 This message has since been reiterated.2
Here, we review advances in our understanding of diabetes and how these inform a new model of diabetes treatment.
BETA CELLS ARE KEY
High levels of glucose and lipids damage and eventually kill beta cells through mechanisms including that of oxidative stress, so that glucose control deteriorates over time. The same processes are active in the target-organ damage seen in diabetes.3,4 These 2 insights—that the disease arises from combinatorial, nondiscrete pressures and that it proceeds through common processes of cell damage—leads us to a more unified understanding of the mechanism of diabetes, and may eventually replace current classifications of type 1, type 2, or latent autoimmune diabetes in adults, as well as nomenclature such as “microvascular” and “macrovascular” disease.3
FIRST-LINE LIFESTYLE INTERVENTIONS
Lifestyle interventions are the first-line therapy for elevated blood glucose. Achieving and maintaining a healthy body mass index is essential to help correct insulin resistance and minimize beta-cell dysfunction.
Lifestyle modifications for overweight or obese patients with diabetes mellitus include optimal caloric intake, decreased intake of simple carbohydrates, increased physical activity, and a 3% to 5% reduction in body weight.5 Weight-loss drugs may be indicated in obese patients. Normalization of lipids and hypertension should be an early goal.
RIGHT MEDICATIONS, RIGHT PATIENTS
While all of the drugs approved for treating diabetes lower glucose levels, some are more beneficial than others, possessing actions beyond their effect on plasma glucose levels, both good and bad.
The AACE guideline for use of various antidiabetic medications6 grades factors such as risks of hypoglycemia, ketoacidosis, weight gain, cardiovascular events, and renal, gastrointestinal, and bone concerns. This represents a much-needed first step toward guidance on selecting the right medications for the right patients. Risk factors (such as heart failure) and comorbidities (such as nonalcoholic fatty liver disease and nonalcoholic steatohepatitis) are among the considerations for choosing treatment.
Two principles
We propose 2 principles when choosing treatment:
Use “gentle” agents, ie, those that are least likely to exhaust beta cells or damage the organs involved in diabetes-related complications. Since the disease course depends on the health of the beta cells, give preference to agents that appear to best support beta cells—ie, agents that create the least oxidative stress or wear-and-tear—as will be outlined in this article.
Diabetes is associated with risks of cardiovascular disease, cardiac events, heart failure, and accelerated renal decompensation. Thus, it is equally important to prevent damage to the cardiovascular system, kidneys, and other tissues subject to damage through glucolipotoxicity.
Balancing glycemic control and risk
The hemoglobin A1c level is the chief target of care and an important barometer of risk of diabetes-related complications. In 2018, the American College of Physicians (ACP) relaxed its target for hemoglobin A1c from 7% to 8%.8 This move was apparently to give physicians greater “wiggle room” for achieving goals in hypoglycemia-prone patients. This, however, may take a toll.
Hypoglycemia is closely tied to cardiovascular disease. Even mild and asymptomatic hypoglycemia that goes undiagnosed and unnoticed by patients has been found to be associated with higher rates of all-cause mortality, prolonged QT interval, angina, arrhythmias, myocardial dysfunction, disturbances in autonomic balance, and sudden death.9–11
However, the ADA, AACE, American Association of Diabetes Educators (AADE), and the Endocrine Society jointly issued a strong indictment of the ACP recommendation.12 They argue that tight glucose control and its well-documented “legacy effects” on long-term outcomes should not be sacrificed.12,13 Indeed, there is no need to abandon evidence-based best practices in care when at least 8 of the 11 classes of antidiabetes agents do not introduce the same level of risk for hypoglycemia.
Current guidelines argue for tight glucose control but generally stop short of discriminating or stratifying the mechanisms of action of the individual classes of drugs. These guidelines also do not stress targeting the particular pathways of hyperglycemia present in any given patient. However, the 2016 ADA joint statement acknowledges the need to “characterize the many paths to beta-cell dysfunction or demise and identify therapeutic approaches that best target each path.”1
PROFILES OF DIABETES DRUGS
The sections below highlight some of the recent data on the profiles of most of the currently available agents.
Metformin: Still the first-line treatment
Current guidelines from the ACP, ADA, and AACE keep metformin14 as the backbone of treatment, although debate continues as to whether newer agents such as GLP-1 receptor agonists are superior for first-line therapy.
Pathways affected. Metformin improves insulin resistance in the liver, increases endogenous GLP-1 levels via the gut, and appears to modulate gut flora composition, which is increasingly suspected to contribute to dysmetabolism.
Advantages, benefits. Metformin is easy to use and does not cause hypoglycemia. It was found to modestly reduce the number of cardiovascular events and deaths in a number of clinical outcome studies.15–19
Disadvantages, adverse effects. In some patients, tolerability restricts the use of this drug at higher doses. The most common adverse effects of metformin are gastrointestinal symptoms (diarrhea, nausea, vomiting, flatulence); other risks include lactic acidosis in patients with impaired kidney function, heart failure, hypoxemia, alcoholism, cirrhosis, contrast exposure, sepsis, and shock.
GLP-1 receptor agonists
GLP-1 receptor agonists20–25 are injectable medications approved for adults with type 2 diabetes. Exenatide and liraglutide lower hemoglobin A1c by 1 to 1.5 absolute percentage points and reduce body weight; these effects persist over the long term.26 Newer once-weekly GLP-1 receptor agonists (albiglutide,20 dulaglutide,21 and semaglutide25) have similar benefits. In 2019, new drug applications were submitted to the FDA for the first-in-kind oral GLP-1 receptor agonists, which would improve convenience and adherence and make this class even more attractive.
Pathways affected. GLP-1 receptor agonists address multiple pathways of hyperglycemia. They increase insulin production and release, promote weight loss, and reduce insulin resistance, glucagon secretion, and inflammation. They also increase amylin, help overcome GLP-1 resistance, slow gastric emptying, and favorably modify gut flora.27
Advantages, benefits. The cardioprotective actions of GLP-1 receptor agonists include reducing inflammation and dysfunction in endothelial and myocardial cells; slowing atherosclerosis; reducing oxidative stress-induced injury and scavenging of reactive oxygen species in coronary endothelial, smooth muscle, and other cells; and enhancing endogenous antioxidant defenses.27 GLP-1 receptor agonism has also been found to inhibit apoptosis in cardiomyocytes, as well as in beta cells.
Several large-scale studies have shown improved outcomes with GLP-1 receptor agonists. The Liraglutide Effect and Action in Diabetes: Evaluation of Cardiovascular Outcome Results (LEADER) trial26 found that liraglutide reduced major adverse cardiovascular events by 13% and myocardial infarctions by 22% in more than 9,000 adults with type 2 diabetes who were at high risk of major adverse cardiovascular events compared with placebo. Rates of microvascular outcomes were also reduced.
A retrospective database analysis of 39,275 patients with type 2 diabetes who were treated with exenatide reported a lower incidence of cardiovascular events than in patients not treated with exenatide.28
However, no effect on cardiovascular outcomes was found with a third GLP-1 agent, lixisenatide, in a large-scale trial in high-risk patients with diabetes.29
The most recently evaluated GLP-1 receptor agonist is semaglutide. The Trial to Evaluate Cardiovascular and Other Long-term Outcomes With Semaglutide in Subjects With Type 2 Diabetes (SUSTAIN-6) demonstrated a reduced risk of major adverse cardiovascular events.30
Disadvantages, adverse effects. The most common adverse effects in this class include nausea, hypoglycemia, diarrhea, constipation, vomiting, headache, decreased appetite, dyspepsia, fatigue, dizziness, abdominal pain, and increased lipase. The nausea can be mitigated by advising patients to stop eating at first sensation of stomach fullness.
DPP-4 inhibitors
Dipeptidyl peptidase 4 (DPP-4) is a ubiquitous enzyme that rapidly degrades GLP-1 and other endogenous peptides.31 Saxagliptin,32 sitagliptin,33 linagliptin,34 and alogliptin35 are approved for use in the United States, and vildagliptin36 is available in Europe.
Pathways affected. These agents modify 3 pathways of hyperglycemia: they increase insulin secretion, decrease glucagon levels, and help overcome GLP-1 resistance.
Advantages, benefits. DPP-4 inhibitors have been used safely and effectively in clinically challenging populations of patients with long-standing type 2 diabetes (> 10 years).
Disadvantages, adverse effects. As this class increases GLP-1 levels only 2- to 4-fold, their efficacy is more modest than that of GLP-1 receptor agonists (hemoglobin A1c reductions of 0.5% to 1%; neutral effects on weight).37
Outcome trials have largely been neutral. Saxagliptin has been associated with an increase in admissions for heart failure. There have been a very small but statistically significant number of drug-related cases of acute pancreatitis.38
The most common adverse effects with this class include headache, nasopharyngitis, urinary tract infection, upper respiratory tract infection, and elevated liver enzymes.
SGLT2 inhibitors
Drugs of this class currently available in the United States are canagliflozin,39 dapagliflozin,40 empagliflozin,41 and ertugliflozin.42
Pathways affected. SGLT2 inhibitors lower the glucose reabsorption threshold in the kidney so that more glucose is excreted in the urine; they also decrease insulin resistance in muscle, liver, and fat cells (via weight loss) and possibly preserve beta-cell function by reducing glucotoxicity. A nonrenal mechanism—delayed gut absorption reducing postprandial glucose excursion—has been proposed to contribute to the glucose-lowering effects of canagliflozin.43
Advantages, benefits. These agents reduce hemoglobin A1c by about 0.5% to 1.0% from a baseline of about 8%. Because their action is independent of insulin, they can be used at any stage of type 2 diabetes, even after insulin secretion has significantly waned. Additional potential advantages include weight loss (up to 3.5% of body mass index) and lowering of systolic blood pressure (2–4 mm Hg) and diastolic blood pressure (1–2 mm Hg).39–42
Canagliflozin was shown in the Canagliflozin Cardiovascular Assessment Study (CANVAS)44 to significantly reduce the overall risk of cardiovascular disease by 14% and risk of heart failure hospitalization by 33% while significantly slowing the progression of renal disease.
In the BI 10773 (Empagliflozin) Cardiovascular Outcome Event Trial in Type 2 Diabetes Mellitus Patients (EMPA-REG OUTCOME),45 empagliflozin reduced heart failure hospitalizations by 35%, cardiovascular deaths by 38%, and all-cause mortality by about 32%. These benefits are thought to be due to less arterial stiffness, lower sympathetic tone, and decreased arrhythmias. Notably, these dramatic benefits accrued in only about 3 years with use of add-on therapy, even though the reduction in hemoglobin A1c was modest (0.6%), suggesting that pleiotropic effects are at work.
Disadvantages, adverse effects. The most common adverse effects of this class include urinary tract infections, yeast infections, dehydration, and hypovolemic symptoms; these can often be prevented. A trend toward increased incidence of amputations in earlier studies was not borne out in a 2018 meta-analysis of 4 observational databases.46
Thiazolidinediones
There are currently 2 approved thiazolidinediones in the United States, pioglitazone47 and rosiglitazone.48 Only pioglitazone is in common use, as rosiglitazone is associated with safety issues.49
Pathways affected. Pioglitazone reduces insulin resistance in muscle, liver, and adipose tissue.
Advantages, benefits. Decreased levels of low-density lipoprotein cholesterol and triglycerides and increased high-density lipoprotein cholesterol levels49 could plausibly account for the cardiovascular benefits reported in the Prospective Pioglitazone Clinical Trial in Macrovascular Events.50 Pioglitazone has also been found to improve insulin secretion, endothelial function, and diastolic dysfunction; reduce inflammation; decrease plasminogen activator inhibitor 1; reverse lipotoxicity; and help correct nonalcoholic fatty liver disease and steatohepatitis.
Pioglitazone has also been found to reduce plaque in carotid and coronary arteries51; improve outcomes in patients with heart failure and myocardial infarction compared with insulin-sensitizing drugs52; and reduce stroke and myocardial infarction in patients with insulin resistance (but not diabetes) and a recent history of ischemic stroke or transient ischemic attack (in the Insulin Resistance Intervention After Stroke trial).53 It may also help maintain beta-cell function; the Actos Now for the Prevention of Diabetes Study found that pioglitazone reduced the risk of conversion of impaired glucose tolerance to frank diabetes by 72%.54
Disadvantages, adverse effects. The most common adverse effects seen with this class include weight gain and salt retention, swelling, edema,55 and related cardiovascular consequences in certain patients. While this may be mitigatable with lifestyle changes or use in combination with a GLP-1 receptor agonist or SGLT2 inhibitor,56 pioglitazone is contraindicated in patients with heart failure, hemodynamic instability, or hepatic dysfunction.
Concerns that pioglitazone might increase the risk of bladder cancer seem to have been put to rest when a study in nearly 200,000 patients found no statistically significant association,57 but the warning remains in the US label.
Long-term use of this class of drugs has been associated with an increased risk of bone fractures,58 which warrants a risk-benefit assessment in each patient.
Injected insulin: Less safe than thought
Recent research suggests that injected insulin has a less favorable safety profile than previously thought.15–19,59 Studies of the long-term safety of insulin therapy have had inconsistent results but suggest that injected insulin is associated with poorer cardiovascular and renal outcomes (in some of the same studies that showed metformin or other agents to improve outcomes),17–19 and the association was dose-dependent. Several studies attempted to cancel out the poorer outcomes by adjusting for hemoglobin A1c levels, stage of disease,17–19,26,27 or severe hypoglycemic episodes.60 However, it may be inappropriate to reduce the impact of these variables, as these may themselves be the mediators of any deleterious effects of exogenous insulin.
When exogenous insulin is introduced into the peripheral circulation it causes a state of persistent iatrogenic hyperinsulinemia, which leads to insulin resistance and also appears to compromise the cardiovascular system. In contrast, endogenous insulin is released into the portal system in tightly controlled amounts.5,61 This suggests that the same insulin peptide may not be equivalently beneficial when introduced in an artificial manner.
Before starting insulin therapy, consider its side effects such as weight gain and hypoglycemia. Most (about 85%) episodes of hypoglycemia occur with basal-bolus insulin regimens.62 Moreover, iatrogenic hyperinsulinemia can damage the vascular system.63,64
We recommend. Insulin therapy is used early in the course of the disease as a short-term intervention for glucolipotoxicity. However, this can be accomplished without attendant risks of hypoglycemia and weight gain by using agents such as SGLT2 inhibitors and incretins. When insulin therapy is necessary, using it as add-on therapy might be considered instead of drug-switching. We have found alternate pharmacologic approaches successful in avoiding or delaying bolus insulin therapy. And in some patients taking insulin, we have had success in progressively introducing a noninsulin agent and were ultimately able to eliminate insulin altogether.
Bromocriptine-QR
Bromocriptine-QR (quick release)65 is a short-acting dopamine agonist that mimics the morning dopamine surge in the suprachiasmatic nucleus—the biologic clock.
Pathways affected. Bromocriptine addresses part of the brain contribution to hyperglycemia, with resultant reductions in both peripheral insulin resistance and sympathetic tone. This reduces muscle, liver, and adipose insulin resistance. It is moderately effective in glucose-lowering, especially in patients with significant insulin resistance.66
Advantages, benefits. A 1-year clinical trial reported that bromocriptine reduced cardiovascular adverse outcomes by 39%, and the composite end point of myocardial infarction, stroke, and cardiovascular death by 52% compared with placebo.67
Disadvantages, adverse effects. The most common adverse effects are nausea, rhinitis, headache, asthenia, dizziness, constipation, and sinusitis.
Alpha-glucosidase inhibitors
Alpha-glucosidase inhibitors (acarbose,68 miglitol69) work by decreasing the rate of absorption of glucose from the gastrointestinal tract.
Advantages, benefits. These drugs decrease hemoglobin A1c by 0.5% to 0.8%.70 They are weight-neutral and do not pose a risk of hypoglycemia. Clinical studies suggest that they may delay or prevent diabetes progression. They were also found to reduce cardiovascular events, acute myocardial infarction, and the onset of hypertension.69
Disadvantages, adverse effects. Their use remains limited due to gastrointestinal adverse effects. They may be contraindicated in patients with inflammatory bowel disease, partial bowel obstruction, or severe renal or hepatic disease.
Pramlintide
Pramlintide71 is an injectable amylin analogue. It is used as monotherapy or in combination with a sulfonylurea, metformin, or insulin glargine.
Pathways affected. Pramlintide decreases appetite, reduces glucagon levels, and minimizes absorption of glucose in the gut.
Disadvantages, adverse effects. Common side effects include mild to moderate hypoglycemia and nausea. Nausea may help explain the ability of pramlintide to confer weight loss when used in combination with insulin.
Sulfonylureas and meglitinides
These classes are still widely used in the treatment of type 2 diabetes, although the AACE6 and ADA72 guidelines de-emphasize their use based on associated risks of hypoglycemia, weight gain, morbidity, mortality, and loss of effect over time.
Pathways affected. Sulfonylureas stimulate insulin secretion from beta cells.
Disadvantages, adverse effects. Sulfonylureas and glinides are associated with poorer outcomes than newer agents in clinical trials15–19,59,60 and may be generally less beta-cell friendly.73 Their harmful effects are difficult to measure in vivo, but these drugs sometimes appear to be associated with more rapid beta-cell failure and progression to insulin dependence compared with newer ones. Several large-scale registry studies have found sulfonylureas and glinides to be associated with poorer outcomes (reviewed by Herman et al).74
Adverse effects include asthenia, headache, dizziness, nausea, diarrhea, epigastric fullness, and heartburn. Although they are often selected based on their low cost, other factors may offset their cost-effectiveness, such as need for glucose monitoring and hospital charges due to sulfonylurea-induced hypoglycemia. Their utility is also limited by dependence on beta-cell function.
Colesevelam
Colesevelam75 is a bile acid sequestrant and low-density lipoprotein cholesterol-reducing agent that has been approved for use in diabetes. The mode of action of colesevelam in this capacity is under investigation. Its effect on hemoglobin A1c is modest. It is associated with gastrointestinal adverse effects, particularly constipation.
Ranolazine
Ranolazine76 is an antianginal drug that also lowers glucose by increasing insulin release. It also possesses cardioprotective properties. In patients with diabetes and non-ST-segment elevation acute coronary syndromes, ranolazine reduced hemoglobin A1c by 1.2% and appeared to be weight-neutral.76 Ranolazine is under clinical development for use in diabetes. Adverse effects include dizziness, headache, constipation, and nausea.
Rational combinations of agents
The ideal strategy would use combinations of agents that mechanistically complement one another and address each path of hyperglycemia present in a patient. This approach should supplant the former approaches of adding-on agents only after treatment failure or sequentially trying first-, second-, and third-line treatments.
Examples of synergistic combinations include those that target fasting plasma glucose and postprandial glucose, reduce reliance on insulin with add-on therapies, or manage hyperglycemia in specific patient groups, such as renal-impaired patients.
Large-scale long-term clinical studies are needed to determine the safety, efficacy, and outcomes of various combinations and whether they confer additive benefits. Some studies have begun to explore possible combinations.
Combined metformin, pioglitazone, and exenatide was reported to delay progression of diabetes in early dysglycemia.77,78 Notably, this combination addresses multiple mediating pathways of hyperglycemia (Table 1).
A GLP-1 receptor agonist with an SGLT2 inhibitor would be another intriguing combination, as the mechanisms of action of these 2 classes complement one another. In limited clinical trials—the DURATION-8 study (lasting 26 weeks),79 the Canagliflozin Cardiovascular Assessment Study (18 weeks),80 and a 24-week study in nondiabetic obese patients81—additive benefits were also seen in systolic blood pressure, body weight, and cardiac risk factors by adding an SGLT2 inhibitor to a GLP-1 receptor agonist, compared with either agent alone. In theory, these improvements might slow or reverse cardiorenal compromise. Lower doses of 1 or more may be possible, and the regimen could prove cost-effective and life-sparing should it slow the progression of the disease and the onset of its complications. A clinical study of this combination is under way (Ralph DeFronzo, personal communication, July 2018). Similarly, the combination of metformin, saxagliptin and dapagliflozin has been shown to be effective.82
CONCLUSION
Care for diabetes mellitus can be particularly challenging for the primary care physician. The progressive nature of diabetes, with worsening hyperglycemia over the course of the disease, further complicates disease management.
Best practices for care nonetheless need to evolve with well-evidenced data, and without years of delay for “trickle-down” education from the specialties to primary care. We have arrived at a juncture to leverage therapies that address the 11 mediating pathways of hyperglycemia, optimally protect beta cells, minimize hypoglycemia, manage risk factors associated with diabetes, and improve diabetes-related outcomes.
- Skyler JS, Bakris GL, Bonifacio E, et al. Differentiation of diabetes by pathophysiology, natural history, and prognosis. Diabetes 2017; 66(2):241–255. doi:10.2337/db16-0806
- Davies MJ, D’Alessio DA, Fradkin J, et al. Management of hyperglycemia in type 2 diabetes, 2018. A consensus report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetes Care 2018; 41(12):2669–2701. doi:10.2337/dci18-0033
- Schwartz SS, Epstein S, Corkey BE, Grant SF, Gavin JR 3rd, Aguilar RB. The time is right for a new classification system for diabetes mellitus: rationale and implications of the beta-cell centric classification schema. Diabetes Care 2016; 39(2):179–186. doi:10.2337/dc15-1585
- Shah MS, Brownlee M. Molecular and cellular mechanisms of cardiovascular disorders in diabetes. Circ Res 2016; 118(11):1808–1829. doi:10.1161/CIRCRESAHA.116.306923
- Schwartz SS, Jellinger PS, Herman ME. Obviating much of the need for insulin therapy in type 2 diabetes mellitus: a re-assessment of insulin therapy’s safety profile. Postgrad Med 2016; 128(6):609–619. doi:10.1080/00325481.2016.1191955
- Garber AJ, Abrahamson MJ, Barzilay JE, et al. Consensus statement by the American Association of Clinical Endocrinologists and American College of Endocrinology on the comprehensive type 2 diabetes management algorithm – 2019 executive summary. Endocr Pract 2019; 25(1):69–100. doi:10.4158/CS-2018-0535
- Sniderman AD, LaChapelle KJ, Rachon NA , Furberg CD. The necessity for clinical reasoning in the era of evidence-based medicine. Mayo Clin Proc 2013; 88(10):1108–1114. doi:10.1016/j.mayocp.2013.07.012
- Qaseem A, Wilt TJ, Kansagara D, Horwitch C, Barry MJ, Forciea MA; Clinical Guidelines Committee of the American College of Physicians. Hemoglobin A1c targets for glycemic control with pharmacologic therapy for nonpregnant adults with type 2 diabetes mellitus: a guidance statement update from the American College of Physicians. Ann Intern Med 2018; 168(8):569–576. doi:10.7326/M17-0939
- Frier BM, Schernthaner G, Heller SR. Hypoglycemia and cardiovascular risks. Diabetes Care 2011; 34(suppl 2):S132–S137. doi:10.2337/dc11-s220
- Chico A, Vidal-Ríos P, Subira M, Novials A. The continuous glucose monitoring system is useful for detecting unrecognized hypoglycemias in patients with type 1 and type 2 diabetes but is not better than frequent capillary glucose measurements for improving metabolic control. Diabetes Care 2003; 26(4):1153–1157. pmid:12663589
- Weber KK, Lohmann T, Busch K, Donati-Hirsch I, Riel R. High frequency of unrecognized hypoglycaemias in patients with type 2 diabetes is discovered by continuous glucose monitoring. Exp Clin Endocrinol Diabetes 2007; 115(8):491–494. doi:10.1055/s-2007-984452
- American Diabetes Association (ADA). The American Diabetes Association, the American Association of Clinical Endocrinologists, the American Association of Diabetes Educators and the Endocrine Society strongly disagree with the American College of Physicians’ guidance for higher blood glucose targets for people with type 2 diabetes www.diabetes.org/newsroom/press-releases/2018/joint-acp-guidance-response.html. Accessed June 6, 2019.
- Freed S; Diabetes in Control. American College of Physicians recommending controversial increase in A1c of 7% to 8%. www.diabetesincontrol.com/american-college-of-physicians-recommending-controversial-increase-in-a1c-of-7-to-8. Accessed June 6, 2019.
- Glucophage XR (metformin hydrochloride) extended release tablets prescribing information. Princeton, NJ, Bristol-Myers Squibb Company, 2009.
- Mellbin LG, Malmberg K, Norhammar A, Wedel H, Rydén L; DIGAMI 2 Investigators. The impact of glucose lowering treatment on long-term prognosis in patients with type 2 diabetes and myocardial infarction: a report from the DIGAMI 2 trial. Eur Heart J 2008; 29(2):166–176. doi:10.1093/eurheartj/ehm518
- Anselmino M, Ohrvik J, Malmberg K, Standl E, Rydén L; Euro Heart Survey Investigators. Glucose lowering treatment in patients with coronary artery disease is prognostically important not only in established but also in newly detected diabetes mellitus: a report from the Euro Heart Survey on Diabetes and the Heart. Eur Heart J 2008; 29(2):177–184. doi:10.1093/eurheartj/ehm519
- Smooke S, Horwich TB, Fonarow GC. Insulin-treated diabetes is associated with a marked increase in mortality in patients with advanced heart failure. Am Heart J 2005; 149(1):168–174. doi:10.1016/j.ahj.2004.07.005
- Colayco DC, Niu F, McCombs JS, Cheetham TC. A1C and cardiovascular outcomes in type 2 diabetes: a nested case-control study. Diabetes Care 2011; 34(1):77–83. doi:10.2337/dc10-1318
- Holden SE, Jenkins-Jones S, Morgan CL, Schernthaner G, Currie CJ. Glucose-lowering with exogenous insulin monotherapy in type 2 diabetes: dose association with all-cause mortality, cardiovascular events and cancer. Diabetes Obes Metab 2015; 17(4):350–362. doi:10.1111/dom.12412
- Tanzeum (albiglutide) prescribing information. Wilmington, DE, GlaxoSmithKline LLC, 2014.
- Trulicity (dulaglutide) prescribing information. Indianapolis, IN, Eli Lilly and Company, 2014.
- Byetta (exenatide) prescribing information. Wilmington, DE, AstraZeneca Pharmaceuticals LP, 2014.
- Victoza (liraglutide injection) prescribing information. Plainsboro, NJ, Novo Nordisk Inc, 2013.
- Adlyxin (lixisenatide injection) prescribing information. Bridgewater, NJ, Sanofi, 2016.
- Ozempic (semaglutide) prescribing information. Plainsboro, NJ, Novo Nordisk, 2017.
- Marso SP, Daniels GH, Brown-Frandsen K, et al; LEADER Steering Committee; LEADER Trial Investigators. Liraglutide and cardiovascular outcomes in type 2 diabetes. N Engl J Med 2016; 375(4):311–322. doi:10.1056/NEJMoa1603827
- Chang G, Zhang D, Yu H, et al. Cardioprotective effects of exenatide against oxidative stress-induced injury. Int J Mol Med 2013; 32(5):1011–1020. doi:10.3892/ijmm.2013.1475
- Best JH, Hoogwerf BJ, Herman WH, et al. Risk of cardiovascular disease events in patients with type 2 diabetes prescribed the glucagon-like peptide 1 (GLP-1) receptor agonist exenatide twice daily or other glucose-lowering therapies: a retrospective analysis of the LifeLink database. Diabetes Care; 34(1):90–95. doi:10.2337/dc10-1393
- Pfeffer MA, Claggett B, Diaz R, et al; ELIXA Investigators. Lixisenatide in patients with type 2 diabetes and acute coronary syndrome. N Engl J Med 2015; 373(23):2247–2257. doi:10.1056/NEJMoa1509225
- Marso SP, Bain SC, Consoli A, et al; SUSTAIN-6 Investigators. Semaglutide and cardiovascular outcomes in patients with type 2 diabetes. N Engl J Med 2016; 375(19):1834–1844. doi:10.1056/NEJMoa1607141
- Mentlein R. Mechanisms underlying the rapid degradation and elimination of the incretin hormones GLP-1 and GIP. Best Pract Res Clin Endocrinol Metab 2009; 23(4):443–452. doi:10.1016/j.beem.2009.03.005
- Onglyza (saxagliptin) tablets prescribing information. Wilmington, DE, AstraZeneca Pharmaceuticals LP, 2014.
- Januvia (sitagliptin) tablets prescribing information. Whitehouse Station, NJ, Merck & Co., Inc, 2014.
- Tradjenta (linagliptin) tablets prescribing information. Ingelheim, Germany, Boehringer Ingelheim International GmbH, 2014.
- Nesina (alogliptin) tablets prescribing information. Deerfield, IL, Takeda Pharmaceuticals America, Inc, 2013.
- Galvus (vildagliptin) prescribing information. North Ryde, Australia, Novartis Pharmaceuticals, 2014.
- Drucker DJ, Nauck MA. The incretin system: glucagon-like peptide-1 receptor agonists and dipeptidyl peptidase-4 inhibitors in type 2 diabetes. Lancet 2006; 368(9548):1696–1705. doi:10.1016/S0140-6736(06)69705-5
- Scirica BM, Bhatt DL, Braunwald E, et al; SAVOR-TIMI 53 Steering Committee and Investigators. Saxagliptin and cardiovascular outcomes in patients with type 2 diabetes mellitus. N Engl J Med 2013; 369(14):1317–1326. doi:10.1056/NEJMoa1307684
- Invokana (canagliflozin) tablets prescribing information. Titusville, NJ: Janssen Pharmaceuticals, Inc, 2013.
- Farxiga (dapagliflozin) prescribing information. Princeton, NJ, Bristol-Myers Squibb, 2014.
- Jardiance (empagliflozin) prescribing information. Ridgefield, CT, Boehringer Ingelheim Pharmaceuticals, Inc, 2014.
- Steglatro (ertugliflozin) prescribing information. Whitehouse Station, NJ, Merck, Sharp & Dohme Corp, 2017.
- Vasilakou D, Karagiannis T, Athanasiadou E, et al. Sodium-glucose cotransporter 2 inhibitors for type 2 diabetes: a systematic review and meta-analysis. Ann Intern Med 2013; 159(4):262–274. doi:10.7326/0003-4819-159-4-201308200-00007
- Neal B, Perkovic V, Mahaffey KW, et al; CANVAS Program Collaborative Group. Canagliflozin and cardiovascular and renal events in type 2 diabetes. N Engl J Med 2017; 377(7):644–657. doi:10.1056/NEJMoa1611925
- Zinman B, Wanner C, Lachin JM, et al; EMPA-REG OUTCOME Investigators. Empagliflozin, cardiovascular outcomes, and mortality in type 2 diabetes. N Engl J Med 2015; 373(22):2117–2128. doi:10.1056/NEJMoa1504720
- Ryan PB, Buse JB, Schuemie MJ, et al. Comparative effectiveness of canagliflozin, SGLT2 inhibitors and non-SGLT2 inhibitors on the risk of hospitalization for heart failure and amputation in patients with type 2 diabetes mellitus: a real-world meta-analysis of 4 observational databases (OBSERVE-4D). Diabetes Obes Metab 2018; 20(11):2485–2597. doi:10.1111/dom.13424
- Actos (pioglitazone) tablets for oral use prescribing information. Deerfield, IL, Takeda Pharmaceuticals America, Inc, 2013.
- Avandia (rosiglitazone maleate tablets) prescribing information. Research Triangle Park, NC, GlaxoSmithKline, 1999.
- Goldberg RB, Kendall DK, Deeg MA, et al; GLAI Study Investigators. A comparison of lipid and glycemic effects of pioglitazone and rosiglitazone in patients with type 2 diabetes and dyslipidemia. Diabetes Care 2005; 28(7):1547–1554. pmid:15983299
- Dormandy JA, Charbonnel B, Eckland DJ, et al; PROactive Investigators. Secondary prevention of macrovascular events in patients with type 2 diabetes in the PROactive Study (PROspective pioglitAzone clinical trial in macroVascular Events): a randomised controlled trial. Lancet 2005; 366:1279–1289. doi:10.1016/S0140-6736(05)67528-9
- Nissen SE, Nicholls SJ, Wolski K, et al; PERISCOPE Investigators. Comparison of pioglitazone vs glimepiride on progression of coronary atherosclerosis in patients with type 2 diabetes the PERISCOPE randomized controlled trial. JAMA 2008; 299(13):1561–1573. doi:10.1001/jama.299.13.1561
- Masoudi FA, Inzucchi SE, Wang Y, Havranek EP, Foody JM, Krumholz HM. Thiazolidinediones, metformin, and outcomes in older patients with diabetes and heart failure: an observational study. Circulation 2005; 111(5):583–590. doi:10.1161/01.CIR.0000154542.13412.B1
- Kernan WN, Viscoli CM, Furie KL, et al; IRIS Trial Investigators. Pioglitazone after ischemic stroke or transient ischemic attack. N Engl J Med 2016; 374(14):1321–1331. doi:10.1056/NEJMoa1506930
- DeFronzo RA, Tripathy D, Schwenke DC, et al; ACT NOW Study. Pioglitazone for diabetes prevention in impaired glucose tolerance. N Engl J Med 2011; 364(12):1104–1115. doi:10.1056/NEJMoa1010949
- Nesto RW, Bell D, Bonow RO, et al; American Heart Association; American Diabetes Association. Thiazolidinedione use, fluid retention, and congestive heart failure: a consensus statement from the American Heart Association and American Diabetes Association. October 7, 2003. Circulation 2003; 108(23):2941–2948. doi:10.1161/01.CIR.0000103683.99399.7E
- Kushner RF, Sujak M. Prevention of weight gain in adult patients with type 2 diabetes treated with pioglitazone. Obesity (Silver Spring) 2009; 17(5):1017–1022. doi:10.1038/oby.2008.651
- Lewis JD, Habel LA, Quesenberry CP, et al. Pioglitazone use and risk of bladder cancer and other common cancers in persons with diabetes. JAMA 2015; 314(3):265–277. doi:10.1001/jama.2015.7996
- Meier C, Kraenzlin ME, Bodmer M, Jick SS, Jick H, Meier CR. Use of thiazolidinediones and fracture risk. Arch Intern Med 2008; 168(8):820–825. doi:10.1001/archinte.168.8.820
- Gamble JM, Chibrikov E, Twells LK, et al. Association of insulin dosage with mortality or major adverse cardiovascular events: a retrospective cohort study. Lancet Diabetes Endocrinol 2017; 5(1):43–52. doi:10.1016/S2213-8587(16)30316-3
- Bonds DE, Miller ME, Bergenstal RM, et al. The association between symptomatic, severe hypoglycaemia and mortality in type 2 diabetes: retrospective epidemiological analysis of the ACCORD study. BMJ 2010; 340:b4909. doi:10.1136/bmj.b4909
- Wang X, Yu C, Zhang B, Wang Y. The injurious effects of hyperinsulinism on blood vessels. Cell Biochem Biophys 2014; 69(2):213–218. doi:10.1007/s12013-013-9810-6
- Garber AJ, King AB, Del Prato S, et al; NN1250-3582 (BEGIN BB T2D) Trial Investigators. Insulin degludec, an ultra-longacting basal insulin, versus insulin glargine in basal-bolus treatment with mealtime insulin aspart in type 2 diabetes (BEGIN Basal-Bolus Type 2): a phase 3, randomised, open-label, treat-to-target non-inferiority trial. Lancet 2012; 379(9825):1498–1507. doi:10.1016/S0140-6736(12)60205-0
- Hanefeld M, Monnier L, Schnell O, Owens D. Early treatment with basal insulin glargine in people with type 2 diabetes: lessons from ORIGIN and other cardiovascular trials. Diabetes Ther 2016; 7(2):187–201. doi:10.1007/s13300-016-0153-3
- Nolan CJ, Ruderman NB, Prentki M. Intensive insulin for type 2 diabetes: the risk of causing harm. Lancet Diabetes Endocrinol 2013; 1(1):9–10. doi:10.1016/S2213-8587(13)70027-5
- Cycloset (bromocriptine mesylate) tablets prescribing information. Tiverton, RI, VeroScience LLC, 2019.
- Schwartz S, Zangeneh F. Evidence-based practice use of quick-release bromocriptine across the natural history of type 2 diabetes mellitus. Postgrad Med 2016; 128(8):828–838. doi:10.1080/00325481.2016.1214059
- Gaziano JM, Cincotta AH, Vinik A, Blonde L, Bohannon N, Scranton R. Effect of bromocriptine-QR (a quick-release formulation of bromocriptine mesylate) on major adverse cardiovascular events in type 2 diabetes subjects. J Am Heart Assoc 2012; 1(5):e002279. doi:10.1161/JAHA.112.002279
- Precose (acarbose) tablets prescribing information. Germany, Bayer HealthCare Pharmaceuticals Inc, 2011.
- Glyset (miglitol) tablets prescribing information. Germany, Bayer HealthCare Pharmaceuticals, Inc, 2012.
- Van de Laar FA, Lucassen PL, Akkermans RP, Van de Lisdonk EH, Rutten GE, Van Weel C. Alpha-glucosidase inhibitors for type 2 diabetes mellitus. Cochrane Database Syst Rev 2005; (2):CD003639. doi:10.1002/14651858.CD003639.pub2
- Symlin (pramlintide acetate) injection for subcutaneous use prescribing information. Wilmongton, DE, AstraZeneca Pharmaceuticals LP, 2014.
- Inzucchi SE, Bergenstal RM, Buse JB, et al. Management of hyperglycaemia in type 2 diabetes, 2015: a patient-centred approach. Update to a position statement of the American Diabetes Association and the European Association for the Study of Diabetes. Diabetologia 2015; 58(3):429–442. doi:10.1007/s00125-014-3460-0
- Wajchenberg BL. Beta-cell failure in diabetes and preservation by clinical treatment. Endocr Rev 2007; 28(2):187–218. doi:10.1210/10.1210/er.2006-0038
- Herman ME, O’Keefe JH, Bell DSH, Schwartz SS. Insulin therapy increases cardiovascular risk in type 2 diabetes. Prog Cardiovasc Dis 2017; 60(3):422–434. doi:10.1016/j.pcad.2017.09.001
- Welchol (colesevelam hydrochloride) prescribing information. Parsippany, NJ, Daiichi Sankyo Inc, 2014.
- Ranexa (ranolazine) prescribing information. Foster City, CA: Gilead Sciences, Inc, 2016.
- Armato J, DeFronzo R, Abdul-Ghani M, Ruby R. Successful treatment of prediabetes in clinical practice: targeting insulin resistance and beta-cell dysfunction. Endocr Pract 2012; 18(3):342–350. doi:10.4158/EP11194.OR
- Abdul-Ghani MA, Puckett C, Triplitt C, et al. Initial combination therapy with metformin, pioglitazone and exenatide is more effective than sequential add-on therapy in subjects with new-onset diabetes. Results from the efficacy and durability of initial combination therapy for type 2 diabetes (EDICT): a randomized trial. Diabetes Obes Metab 2015; 17(3):268–275. doi:10.1111/dom.12417
- Frías JP, Guja C, Hardy E, et al. Exenatide once weekly plus dapagliflozin once daily versus exenatide or dapagliflozin alone in patients with type 2 diabetes inadequately controlled with metformin monotherapy (DURATION-8): a 28 week, multicentre, double-blind, phase 3, randomised controlled trial. Lancet Diabetes Endocrinol 2016; 4(12):1004–1016. doi:10.1016/S2213-8587(16)30267-4
- Fulcher G, Matthews DR, Perkovic V, et al; CANVAS trial collaborative group. Efficacy and safety of canagliflozin when used in conjunction with incretin-mimetic therapy in patients with type 2 diabetes. Diabetes Obes Metab 2016; 18(1):82–91. doi:10.1111/dom.12589
- Lundkvist P, Sjöström CD, Amini S, Pereira MJ, Johnsson E, Eriksson JW. Dapagliflozin once-daily and exenatide once-weekly dual therapy: a 24-week randomized, placebo-controlled, phase II study examining effects on body weight and prediabetes in obese adults without diabetes. Diabetes Obes Metab 2017; 19(1):49–60. doi:10.1111/dom.12779
- Del Prato S, Rosenstock J, Garcia-Sanchez R, et al. Safety and tolerability of dapagliflozin, saxagliptin and metformin in combination: post-hoc analysis of concomitant add-on versus sequential add-on to metformin and of triple versus dual therapy with metformin. Diabetes Obes Metab 2018; 20(6):1542–1546. doi:10.1111/dom.13258
Insights from basic and clinical research are changing the way we treat diabetes mellitus. In 2016, several key diabetes organizations, ie, the American Diabetes Association (ADA), the Juvenile Diabetes Research Foundation (JDRF), the European Association for the Study of Diabetes (EASD), and the American Association of Clinical Endocrinologists (AACE), called for bringing therapeutic approaches in line with our updated understanding of disease pathophysiology, replacing “one-size-fits-all” management with a tailored approach.1 This message has since been reiterated.2
Here, we review advances in our understanding of diabetes and how these inform a new model of diabetes treatment.
BETA CELLS ARE KEY
High levels of glucose and lipids damage and eventually kill beta cells through mechanisms including that of oxidative stress, so that glucose control deteriorates over time. The same processes are active in the target-organ damage seen in diabetes.3,4 These 2 insights—that the disease arises from combinatorial, nondiscrete pressures and that it proceeds through common processes of cell damage—leads us to a more unified understanding of the mechanism of diabetes, and may eventually replace current classifications of type 1, type 2, or latent autoimmune diabetes in adults, as well as nomenclature such as “microvascular” and “macrovascular” disease.3
FIRST-LINE LIFESTYLE INTERVENTIONS
Lifestyle interventions are the first-line therapy for elevated blood glucose. Achieving and maintaining a healthy body mass index is essential to help correct insulin resistance and minimize beta-cell dysfunction.
Lifestyle modifications for overweight or obese patients with diabetes mellitus include optimal caloric intake, decreased intake of simple carbohydrates, increased physical activity, and a 3% to 5% reduction in body weight.5 Weight-loss drugs may be indicated in obese patients. Normalization of lipids and hypertension should be an early goal.
RIGHT MEDICATIONS, RIGHT PATIENTS
While all of the drugs approved for treating diabetes lower glucose levels, some are more beneficial than others, possessing actions beyond their effect on plasma glucose levels, both good and bad.
The AACE guideline for use of various antidiabetic medications6 grades factors such as risks of hypoglycemia, ketoacidosis, weight gain, cardiovascular events, and renal, gastrointestinal, and bone concerns. This represents a much-needed first step toward guidance on selecting the right medications for the right patients. Risk factors (such as heart failure) and comorbidities (such as nonalcoholic fatty liver disease and nonalcoholic steatohepatitis) are among the considerations for choosing treatment.
Two principles
We propose 2 principles when choosing treatment:
Use “gentle” agents, ie, those that are least likely to exhaust beta cells or damage the organs involved in diabetes-related complications. Since the disease course depends on the health of the beta cells, give preference to agents that appear to best support beta cells—ie, agents that create the least oxidative stress or wear-and-tear—as will be outlined in this article.
Diabetes is associated with risks of cardiovascular disease, cardiac events, heart failure, and accelerated renal decompensation. Thus, it is equally important to prevent damage to the cardiovascular system, kidneys, and other tissues subject to damage through glucolipotoxicity.
Balancing glycemic control and risk
The hemoglobin A1c level is the chief target of care and an important barometer of risk of diabetes-related complications. In 2018, the American College of Physicians (ACP) relaxed its target for hemoglobin A1c from 7% to 8%.8 This move was apparently to give physicians greater “wiggle room” for achieving goals in hypoglycemia-prone patients. This, however, may take a toll.
Hypoglycemia is closely tied to cardiovascular disease. Even mild and asymptomatic hypoglycemia that goes undiagnosed and unnoticed by patients has been found to be associated with higher rates of all-cause mortality, prolonged QT interval, angina, arrhythmias, myocardial dysfunction, disturbances in autonomic balance, and sudden death.9–11
However, the ADA, AACE, American Association of Diabetes Educators (AADE), and the Endocrine Society jointly issued a strong indictment of the ACP recommendation.12 They argue that tight glucose control and its well-documented “legacy effects” on long-term outcomes should not be sacrificed.12,13 Indeed, there is no need to abandon evidence-based best practices in care when at least 8 of the 11 classes of antidiabetes agents do not introduce the same level of risk for hypoglycemia.
Current guidelines argue for tight glucose control but generally stop short of discriminating or stratifying the mechanisms of action of the individual classes of drugs. These guidelines also do not stress targeting the particular pathways of hyperglycemia present in any given patient. However, the 2016 ADA joint statement acknowledges the need to “characterize the many paths to beta-cell dysfunction or demise and identify therapeutic approaches that best target each path.”1
PROFILES OF DIABETES DRUGS
The sections below highlight some of the recent data on the profiles of most of the currently available agents.
Metformin: Still the first-line treatment
Current guidelines from the ACP, ADA, and AACE keep metformin14 as the backbone of treatment, although debate continues as to whether newer agents such as GLP-1 receptor agonists are superior for first-line therapy.
Pathways affected. Metformin improves insulin resistance in the liver, increases endogenous GLP-1 levels via the gut, and appears to modulate gut flora composition, which is increasingly suspected to contribute to dysmetabolism.
Advantages, benefits. Metformin is easy to use and does not cause hypoglycemia. It was found to modestly reduce the number of cardiovascular events and deaths in a number of clinical outcome studies.15–19
Disadvantages, adverse effects. In some patients, tolerability restricts the use of this drug at higher doses. The most common adverse effects of metformin are gastrointestinal symptoms (diarrhea, nausea, vomiting, flatulence); other risks include lactic acidosis in patients with impaired kidney function, heart failure, hypoxemia, alcoholism, cirrhosis, contrast exposure, sepsis, and shock.
GLP-1 receptor agonists
GLP-1 receptor agonists20–25 are injectable medications approved for adults with type 2 diabetes. Exenatide and liraglutide lower hemoglobin A1c by 1 to 1.5 absolute percentage points and reduce body weight; these effects persist over the long term.26 Newer once-weekly GLP-1 receptor agonists (albiglutide,20 dulaglutide,21 and semaglutide25) have similar benefits. In 2019, new drug applications were submitted to the FDA for the first-in-kind oral GLP-1 receptor agonists, which would improve convenience and adherence and make this class even more attractive.
Pathways affected. GLP-1 receptor agonists address multiple pathways of hyperglycemia. They increase insulin production and release, promote weight loss, and reduce insulin resistance, glucagon secretion, and inflammation. They also increase amylin, help overcome GLP-1 resistance, slow gastric emptying, and favorably modify gut flora.27
Advantages, benefits. The cardioprotective actions of GLP-1 receptor agonists include reducing inflammation and dysfunction in endothelial and myocardial cells; slowing atherosclerosis; reducing oxidative stress-induced injury and scavenging of reactive oxygen species in coronary endothelial, smooth muscle, and other cells; and enhancing endogenous antioxidant defenses.27 GLP-1 receptor agonism has also been found to inhibit apoptosis in cardiomyocytes, as well as in beta cells.
Several large-scale studies have shown improved outcomes with GLP-1 receptor agonists. The Liraglutide Effect and Action in Diabetes: Evaluation of Cardiovascular Outcome Results (LEADER) trial26 found that liraglutide reduced major adverse cardiovascular events by 13% and myocardial infarctions by 22% in more than 9,000 adults with type 2 diabetes who were at high risk of major adverse cardiovascular events compared with placebo. Rates of microvascular outcomes were also reduced.
A retrospective database analysis of 39,275 patients with type 2 diabetes who were treated with exenatide reported a lower incidence of cardiovascular events than in patients not treated with exenatide.28
However, no effect on cardiovascular outcomes was found with a third GLP-1 agent, lixisenatide, in a large-scale trial in high-risk patients with diabetes.29
The most recently evaluated GLP-1 receptor agonist is semaglutide. The Trial to Evaluate Cardiovascular and Other Long-term Outcomes With Semaglutide in Subjects With Type 2 Diabetes (SUSTAIN-6) demonstrated a reduced risk of major adverse cardiovascular events.30
Disadvantages, adverse effects. The most common adverse effects in this class include nausea, hypoglycemia, diarrhea, constipation, vomiting, headache, decreased appetite, dyspepsia, fatigue, dizziness, abdominal pain, and increased lipase. The nausea can be mitigated by advising patients to stop eating at first sensation of stomach fullness.
DPP-4 inhibitors
Dipeptidyl peptidase 4 (DPP-4) is a ubiquitous enzyme that rapidly degrades GLP-1 and other endogenous peptides.31 Saxagliptin,32 sitagliptin,33 linagliptin,34 and alogliptin35 are approved for use in the United States, and vildagliptin36 is available in Europe.
Pathways affected. These agents modify 3 pathways of hyperglycemia: they increase insulin secretion, decrease glucagon levels, and help overcome GLP-1 resistance.
Advantages, benefits. DPP-4 inhibitors have been used safely and effectively in clinically challenging populations of patients with long-standing type 2 diabetes (> 10 years).
Disadvantages, adverse effects. As this class increases GLP-1 levels only 2- to 4-fold, their efficacy is more modest than that of GLP-1 receptor agonists (hemoglobin A1c reductions of 0.5% to 1%; neutral effects on weight).37
Outcome trials have largely been neutral. Saxagliptin has been associated with an increase in admissions for heart failure. There have been a very small but statistically significant number of drug-related cases of acute pancreatitis.38
The most common adverse effects with this class include headache, nasopharyngitis, urinary tract infection, upper respiratory tract infection, and elevated liver enzymes.
SGLT2 inhibitors
Drugs of this class currently available in the United States are canagliflozin,39 dapagliflozin,40 empagliflozin,41 and ertugliflozin.42
Pathways affected. SGLT2 inhibitors lower the glucose reabsorption threshold in the kidney so that more glucose is excreted in the urine; they also decrease insulin resistance in muscle, liver, and fat cells (via weight loss) and possibly preserve beta-cell function by reducing glucotoxicity. A nonrenal mechanism—delayed gut absorption reducing postprandial glucose excursion—has been proposed to contribute to the glucose-lowering effects of canagliflozin.43
Advantages, benefits. These agents reduce hemoglobin A1c by about 0.5% to 1.0% from a baseline of about 8%. Because their action is independent of insulin, they can be used at any stage of type 2 diabetes, even after insulin secretion has significantly waned. Additional potential advantages include weight loss (up to 3.5% of body mass index) and lowering of systolic blood pressure (2–4 mm Hg) and diastolic blood pressure (1–2 mm Hg).39–42
Canagliflozin was shown in the Canagliflozin Cardiovascular Assessment Study (CANVAS)44 to significantly reduce the overall risk of cardiovascular disease by 14% and risk of heart failure hospitalization by 33% while significantly slowing the progression of renal disease.
In the BI 10773 (Empagliflozin) Cardiovascular Outcome Event Trial in Type 2 Diabetes Mellitus Patients (EMPA-REG OUTCOME),45 empagliflozin reduced heart failure hospitalizations by 35%, cardiovascular deaths by 38%, and all-cause mortality by about 32%. These benefits are thought to be due to less arterial stiffness, lower sympathetic tone, and decreased arrhythmias. Notably, these dramatic benefits accrued in only about 3 years with use of add-on therapy, even though the reduction in hemoglobin A1c was modest (0.6%), suggesting that pleiotropic effects are at work.
Disadvantages, adverse effects. The most common adverse effects of this class include urinary tract infections, yeast infections, dehydration, and hypovolemic symptoms; these can often be prevented. A trend toward increased incidence of amputations in earlier studies was not borne out in a 2018 meta-analysis of 4 observational databases.46
Thiazolidinediones
There are currently 2 approved thiazolidinediones in the United States, pioglitazone47 and rosiglitazone.48 Only pioglitazone is in common use, as rosiglitazone is associated with safety issues.49
Pathways affected. Pioglitazone reduces insulin resistance in muscle, liver, and adipose tissue.
Advantages, benefits. Decreased levels of low-density lipoprotein cholesterol and triglycerides and increased high-density lipoprotein cholesterol levels49 could plausibly account for the cardiovascular benefits reported in the Prospective Pioglitazone Clinical Trial in Macrovascular Events.50 Pioglitazone has also been found to improve insulin secretion, endothelial function, and diastolic dysfunction; reduce inflammation; decrease plasminogen activator inhibitor 1; reverse lipotoxicity; and help correct nonalcoholic fatty liver disease and steatohepatitis.
Pioglitazone has also been found to reduce plaque in carotid and coronary arteries51; improve outcomes in patients with heart failure and myocardial infarction compared with insulin-sensitizing drugs52; and reduce stroke and myocardial infarction in patients with insulin resistance (but not diabetes) and a recent history of ischemic stroke or transient ischemic attack (in the Insulin Resistance Intervention After Stroke trial).53 It may also help maintain beta-cell function; the Actos Now for the Prevention of Diabetes Study found that pioglitazone reduced the risk of conversion of impaired glucose tolerance to frank diabetes by 72%.54
Disadvantages, adverse effects. The most common adverse effects seen with this class include weight gain and salt retention, swelling, edema,55 and related cardiovascular consequences in certain patients. While this may be mitigatable with lifestyle changes or use in combination with a GLP-1 receptor agonist or SGLT2 inhibitor,56 pioglitazone is contraindicated in patients with heart failure, hemodynamic instability, or hepatic dysfunction.
Concerns that pioglitazone might increase the risk of bladder cancer seem to have been put to rest when a study in nearly 200,000 patients found no statistically significant association,57 but the warning remains in the US label.
Long-term use of this class of drugs has been associated with an increased risk of bone fractures,58 which warrants a risk-benefit assessment in each patient.
Injected insulin: Less safe than thought
Recent research suggests that injected insulin has a less favorable safety profile than previously thought.15–19,59 Studies of the long-term safety of insulin therapy have had inconsistent results but suggest that injected insulin is associated with poorer cardiovascular and renal outcomes (in some of the same studies that showed metformin or other agents to improve outcomes),17–19 and the association was dose-dependent. Several studies attempted to cancel out the poorer outcomes by adjusting for hemoglobin A1c levels, stage of disease,17–19,26,27 or severe hypoglycemic episodes.60 However, it may be inappropriate to reduce the impact of these variables, as these may themselves be the mediators of any deleterious effects of exogenous insulin.
When exogenous insulin is introduced into the peripheral circulation it causes a state of persistent iatrogenic hyperinsulinemia, which leads to insulin resistance and also appears to compromise the cardiovascular system. In contrast, endogenous insulin is released into the portal system in tightly controlled amounts.5,61 This suggests that the same insulin peptide may not be equivalently beneficial when introduced in an artificial manner.
Before starting insulin therapy, consider its side effects such as weight gain and hypoglycemia. Most (about 85%) episodes of hypoglycemia occur with basal-bolus insulin regimens.62 Moreover, iatrogenic hyperinsulinemia can damage the vascular system.63,64
We recommend. Insulin therapy is used early in the course of the disease as a short-term intervention for glucolipotoxicity. However, this can be accomplished without attendant risks of hypoglycemia and weight gain by using agents such as SGLT2 inhibitors and incretins. When insulin therapy is necessary, using it as add-on therapy might be considered instead of drug-switching. We have found alternate pharmacologic approaches successful in avoiding or delaying bolus insulin therapy. And in some patients taking insulin, we have had success in progressively introducing a noninsulin agent and were ultimately able to eliminate insulin altogether.
Bromocriptine-QR
Bromocriptine-QR (quick release)65 is a short-acting dopamine agonist that mimics the morning dopamine surge in the suprachiasmatic nucleus—the biologic clock.
Pathways affected. Bromocriptine addresses part of the brain contribution to hyperglycemia, with resultant reductions in both peripheral insulin resistance and sympathetic tone. This reduces muscle, liver, and adipose insulin resistance. It is moderately effective in glucose-lowering, especially in patients with significant insulin resistance.66
Advantages, benefits. A 1-year clinical trial reported that bromocriptine reduced cardiovascular adverse outcomes by 39%, and the composite end point of myocardial infarction, stroke, and cardiovascular death by 52% compared with placebo.67
Disadvantages, adverse effects. The most common adverse effects are nausea, rhinitis, headache, asthenia, dizziness, constipation, and sinusitis.
Alpha-glucosidase inhibitors
Alpha-glucosidase inhibitors (acarbose,68 miglitol69) work by decreasing the rate of absorption of glucose from the gastrointestinal tract.
Advantages, benefits. These drugs decrease hemoglobin A1c by 0.5% to 0.8%.70 They are weight-neutral and do not pose a risk of hypoglycemia. Clinical studies suggest that they may delay or prevent diabetes progression. They were also found to reduce cardiovascular events, acute myocardial infarction, and the onset of hypertension.69
Disadvantages, adverse effects. Their use remains limited due to gastrointestinal adverse effects. They may be contraindicated in patients with inflammatory bowel disease, partial bowel obstruction, or severe renal or hepatic disease.
Pramlintide
Pramlintide71 is an injectable amylin analogue. It is used as monotherapy or in combination with a sulfonylurea, metformin, or insulin glargine.
Pathways affected. Pramlintide decreases appetite, reduces glucagon levels, and minimizes absorption of glucose in the gut.
Disadvantages, adverse effects. Common side effects include mild to moderate hypoglycemia and nausea. Nausea may help explain the ability of pramlintide to confer weight loss when used in combination with insulin.
Sulfonylureas and meglitinides
These classes are still widely used in the treatment of type 2 diabetes, although the AACE6 and ADA72 guidelines de-emphasize their use based on associated risks of hypoglycemia, weight gain, morbidity, mortality, and loss of effect over time.
Pathways affected. Sulfonylureas stimulate insulin secretion from beta cells.
Disadvantages, adverse effects. Sulfonylureas and glinides are associated with poorer outcomes than newer agents in clinical trials15–19,59,60 and may be generally less beta-cell friendly.73 Their harmful effects are difficult to measure in vivo, but these drugs sometimes appear to be associated with more rapid beta-cell failure and progression to insulin dependence compared with newer ones. Several large-scale registry studies have found sulfonylureas and glinides to be associated with poorer outcomes (reviewed by Herman et al).74
Adverse effects include asthenia, headache, dizziness, nausea, diarrhea, epigastric fullness, and heartburn. Although they are often selected based on their low cost, other factors may offset their cost-effectiveness, such as need for glucose monitoring and hospital charges due to sulfonylurea-induced hypoglycemia. Their utility is also limited by dependence on beta-cell function.
Colesevelam
Colesevelam75 is a bile acid sequestrant and low-density lipoprotein cholesterol-reducing agent that has been approved for use in diabetes. The mode of action of colesevelam in this capacity is under investigation. Its effect on hemoglobin A1c is modest. It is associated with gastrointestinal adverse effects, particularly constipation.
Ranolazine
Ranolazine76 is an antianginal drug that also lowers glucose by increasing insulin release. It also possesses cardioprotective properties. In patients with diabetes and non-ST-segment elevation acute coronary syndromes, ranolazine reduced hemoglobin A1c by 1.2% and appeared to be weight-neutral.76 Ranolazine is under clinical development for use in diabetes. Adverse effects include dizziness, headache, constipation, and nausea.
Rational combinations of agents
The ideal strategy would use combinations of agents that mechanistically complement one another and address each path of hyperglycemia present in a patient. This approach should supplant the former approaches of adding-on agents only after treatment failure or sequentially trying first-, second-, and third-line treatments.
Examples of synergistic combinations include those that target fasting plasma glucose and postprandial glucose, reduce reliance on insulin with add-on therapies, or manage hyperglycemia in specific patient groups, such as renal-impaired patients.
Large-scale long-term clinical studies are needed to determine the safety, efficacy, and outcomes of various combinations and whether they confer additive benefits. Some studies have begun to explore possible combinations.
Combined metformin, pioglitazone, and exenatide was reported to delay progression of diabetes in early dysglycemia.77,78 Notably, this combination addresses multiple mediating pathways of hyperglycemia (Table 1).
A GLP-1 receptor agonist with an SGLT2 inhibitor would be another intriguing combination, as the mechanisms of action of these 2 classes complement one another. In limited clinical trials—the DURATION-8 study (lasting 26 weeks),79 the Canagliflozin Cardiovascular Assessment Study (18 weeks),80 and a 24-week study in nondiabetic obese patients81—additive benefits were also seen in systolic blood pressure, body weight, and cardiac risk factors by adding an SGLT2 inhibitor to a GLP-1 receptor agonist, compared with either agent alone. In theory, these improvements might slow or reverse cardiorenal compromise. Lower doses of 1 or more may be possible, and the regimen could prove cost-effective and life-sparing should it slow the progression of the disease and the onset of its complications. A clinical study of this combination is under way (Ralph DeFronzo, personal communication, July 2018). Similarly, the combination of metformin, saxagliptin and dapagliflozin has been shown to be effective.82
CONCLUSION
Care for diabetes mellitus can be particularly challenging for the primary care physician. The progressive nature of diabetes, with worsening hyperglycemia over the course of the disease, further complicates disease management.
Best practices for care nonetheless need to evolve with well-evidenced data, and without years of delay for “trickle-down” education from the specialties to primary care. We have arrived at a juncture to leverage therapies that address the 11 mediating pathways of hyperglycemia, optimally protect beta cells, minimize hypoglycemia, manage risk factors associated with diabetes, and improve diabetes-related outcomes.
Insights from basic and clinical research are changing the way we treat diabetes mellitus. In 2016, several key diabetes organizations, ie, the American Diabetes Association (ADA), the Juvenile Diabetes Research Foundation (JDRF), the European Association for the Study of Diabetes (EASD), and the American Association of Clinical Endocrinologists (AACE), called for bringing therapeutic approaches in line with our updated understanding of disease pathophysiology, replacing “one-size-fits-all” management with a tailored approach.1 This message has since been reiterated.2
Here, we review advances in our understanding of diabetes and how these inform a new model of diabetes treatment.
BETA CELLS ARE KEY
High levels of glucose and lipids damage and eventually kill beta cells through mechanisms including that of oxidative stress, so that glucose control deteriorates over time. The same processes are active in the target-organ damage seen in diabetes.3,4 These 2 insights—that the disease arises from combinatorial, nondiscrete pressures and that it proceeds through common processes of cell damage—leads us to a more unified understanding of the mechanism of diabetes, and may eventually replace current classifications of type 1, type 2, or latent autoimmune diabetes in adults, as well as nomenclature such as “microvascular” and “macrovascular” disease.3
FIRST-LINE LIFESTYLE INTERVENTIONS
Lifestyle interventions are the first-line therapy for elevated blood glucose. Achieving and maintaining a healthy body mass index is essential to help correct insulin resistance and minimize beta-cell dysfunction.
Lifestyle modifications for overweight or obese patients with diabetes mellitus include optimal caloric intake, decreased intake of simple carbohydrates, increased physical activity, and a 3% to 5% reduction in body weight.5 Weight-loss drugs may be indicated in obese patients. Normalization of lipids and hypertension should be an early goal.
RIGHT MEDICATIONS, RIGHT PATIENTS
While all of the drugs approved for treating diabetes lower glucose levels, some are more beneficial than others, possessing actions beyond their effect on plasma glucose levels, both good and bad.
The AACE guideline for use of various antidiabetic medications6 grades factors such as risks of hypoglycemia, ketoacidosis, weight gain, cardiovascular events, and renal, gastrointestinal, and bone concerns. This represents a much-needed first step toward guidance on selecting the right medications for the right patients. Risk factors (such as heart failure) and comorbidities (such as nonalcoholic fatty liver disease and nonalcoholic steatohepatitis) are among the considerations for choosing treatment.
Two principles
We propose 2 principles when choosing treatment:
Use “gentle” agents, ie, those that are least likely to exhaust beta cells or damage the organs involved in diabetes-related complications. Since the disease course depends on the health of the beta cells, give preference to agents that appear to best support beta cells—ie, agents that create the least oxidative stress or wear-and-tear—as will be outlined in this article.
Diabetes is associated with risks of cardiovascular disease, cardiac events, heart failure, and accelerated renal decompensation. Thus, it is equally important to prevent damage to the cardiovascular system, kidneys, and other tissues subject to damage through glucolipotoxicity.
Balancing glycemic control and risk
The hemoglobin A1c level is the chief target of care and an important barometer of risk of diabetes-related complications. In 2018, the American College of Physicians (ACP) relaxed its target for hemoglobin A1c from 7% to 8%.8 This move was apparently to give physicians greater “wiggle room” for achieving goals in hypoglycemia-prone patients. This, however, may take a toll.
Hypoglycemia is closely tied to cardiovascular disease. Even mild and asymptomatic hypoglycemia that goes undiagnosed and unnoticed by patients has been found to be associated with higher rates of all-cause mortality, prolonged QT interval, angina, arrhythmias, myocardial dysfunction, disturbances in autonomic balance, and sudden death.9–11
However, the ADA, AACE, American Association of Diabetes Educators (AADE), and the Endocrine Society jointly issued a strong indictment of the ACP recommendation.12 They argue that tight glucose control and its well-documented “legacy effects” on long-term outcomes should not be sacrificed.12,13 Indeed, there is no need to abandon evidence-based best practices in care when at least 8 of the 11 classes of antidiabetes agents do not introduce the same level of risk for hypoglycemia.
Current guidelines argue for tight glucose control but generally stop short of discriminating or stratifying the mechanisms of action of the individual classes of drugs. These guidelines also do not stress targeting the particular pathways of hyperglycemia present in any given patient. However, the 2016 ADA joint statement acknowledges the need to “characterize the many paths to beta-cell dysfunction or demise and identify therapeutic approaches that best target each path.”1
PROFILES OF DIABETES DRUGS
The sections below highlight some of the recent data on the profiles of most of the currently available agents.
Metformin: Still the first-line treatment
Current guidelines from the ACP, ADA, and AACE keep metformin14 as the backbone of treatment, although debate continues as to whether newer agents such as GLP-1 receptor agonists are superior for first-line therapy.
Pathways affected. Metformin improves insulin resistance in the liver, increases endogenous GLP-1 levels via the gut, and appears to modulate gut flora composition, which is increasingly suspected to contribute to dysmetabolism.
Advantages, benefits. Metformin is easy to use and does not cause hypoglycemia. It was found to modestly reduce the number of cardiovascular events and deaths in a number of clinical outcome studies.15–19
Disadvantages, adverse effects. In some patients, tolerability restricts the use of this drug at higher doses. The most common adverse effects of metformin are gastrointestinal symptoms (diarrhea, nausea, vomiting, flatulence); other risks include lactic acidosis in patients with impaired kidney function, heart failure, hypoxemia, alcoholism, cirrhosis, contrast exposure, sepsis, and shock.
GLP-1 receptor agonists
GLP-1 receptor agonists20–25 are injectable medications approved for adults with type 2 diabetes. Exenatide and liraglutide lower hemoglobin A1c by 1 to 1.5 absolute percentage points and reduce body weight; these effects persist over the long term.26 Newer once-weekly GLP-1 receptor agonists (albiglutide,20 dulaglutide,21 and semaglutide25) have similar benefits. In 2019, new drug applications were submitted to the FDA for the first-in-kind oral GLP-1 receptor agonists, which would improve convenience and adherence and make this class even more attractive.
Pathways affected. GLP-1 receptor agonists address multiple pathways of hyperglycemia. They increase insulin production and release, promote weight loss, and reduce insulin resistance, glucagon secretion, and inflammation. They also increase amylin, help overcome GLP-1 resistance, slow gastric emptying, and favorably modify gut flora.27
Advantages, benefits. The cardioprotective actions of GLP-1 receptor agonists include reducing inflammation and dysfunction in endothelial and myocardial cells; slowing atherosclerosis; reducing oxidative stress-induced injury and scavenging of reactive oxygen species in coronary endothelial, smooth muscle, and other cells; and enhancing endogenous antioxidant defenses.27 GLP-1 receptor agonism has also been found to inhibit apoptosis in cardiomyocytes, as well as in beta cells.
Several large-scale studies have shown improved outcomes with GLP-1 receptor agonists. The Liraglutide Effect and Action in Diabetes: Evaluation of Cardiovascular Outcome Results (LEADER) trial26 found that liraglutide reduced major adverse cardiovascular events by 13% and myocardial infarctions by 22% in more than 9,000 adults with type 2 diabetes who were at high risk of major adverse cardiovascular events compared with placebo. Rates of microvascular outcomes were also reduced.
A retrospective database analysis of 39,275 patients with type 2 diabetes who were treated with exenatide reported a lower incidence of cardiovascular events than in patients not treated with exenatide.28
However, no effect on cardiovascular outcomes was found with a third GLP-1 agent, lixisenatide, in a large-scale trial in high-risk patients with diabetes.29
The most recently evaluated GLP-1 receptor agonist is semaglutide. The Trial to Evaluate Cardiovascular and Other Long-term Outcomes With Semaglutide in Subjects With Type 2 Diabetes (SUSTAIN-6) demonstrated a reduced risk of major adverse cardiovascular events.30
Disadvantages, adverse effects. The most common adverse effects in this class include nausea, hypoglycemia, diarrhea, constipation, vomiting, headache, decreased appetite, dyspepsia, fatigue, dizziness, abdominal pain, and increased lipase. The nausea can be mitigated by advising patients to stop eating at first sensation of stomach fullness.
DPP-4 inhibitors
Dipeptidyl peptidase 4 (DPP-4) is a ubiquitous enzyme that rapidly degrades GLP-1 and other endogenous peptides.31 Saxagliptin,32 sitagliptin,33 linagliptin,34 and alogliptin35 are approved for use in the United States, and vildagliptin36 is available in Europe.
Pathways affected. These agents modify 3 pathways of hyperglycemia: they increase insulin secretion, decrease glucagon levels, and help overcome GLP-1 resistance.
Advantages, benefits. DPP-4 inhibitors have been used safely and effectively in clinically challenging populations of patients with long-standing type 2 diabetes (> 10 years).
Disadvantages, adverse effects. As this class increases GLP-1 levels only 2- to 4-fold, their efficacy is more modest than that of GLP-1 receptor agonists (hemoglobin A1c reductions of 0.5% to 1%; neutral effects on weight).37
Outcome trials have largely been neutral. Saxagliptin has been associated with an increase in admissions for heart failure. There have been a very small but statistically significant number of drug-related cases of acute pancreatitis.38
The most common adverse effects with this class include headache, nasopharyngitis, urinary tract infection, upper respiratory tract infection, and elevated liver enzymes.
SGLT2 inhibitors
Drugs of this class currently available in the United States are canagliflozin,39 dapagliflozin,40 empagliflozin,41 and ertugliflozin.42
Pathways affected. SGLT2 inhibitors lower the glucose reabsorption threshold in the kidney so that more glucose is excreted in the urine; they also decrease insulin resistance in muscle, liver, and fat cells (via weight loss) and possibly preserve beta-cell function by reducing glucotoxicity. A nonrenal mechanism—delayed gut absorption reducing postprandial glucose excursion—has been proposed to contribute to the glucose-lowering effects of canagliflozin.43
Advantages, benefits. These agents reduce hemoglobin A1c by about 0.5% to 1.0% from a baseline of about 8%. Because their action is independent of insulin, they can be used at any stage of type 2 diabetes, even after insulin secretion has significantly waned. Additional potential advantages include weight loss (up to 3.5% of body mass index) and lowering of systolic blood pressure (2–4 mm Hg) and diastolic blood pressure (1–2 mm Hg).39–42
Canagliflozin was shown in the Canagliflozin Cardiovascular Assessment Study (CANVAS)44 to significantly reduce the overall risk of cardiovascular disease by 14% and risk of heart failure hospitalization by 33% while significantly slowing the progression of renal disease.
In the BI 10773 (Empagliflozin) Cardiovascular Outcome Event Trial in Type 2 Diabetes Mellitus Patients (EMPA-REG OUTCOME),45 empagliflozin reduced heart failure hospitalizations by 35%, cardiovascular deaths by 38%, and all-cause mortality by about 32%. These benefits are thought to be due to less arterial stiffness, lower sympathetic tone, and decreased arrhythmias. Notably, these dramatic benefits accrued in only about 3 years with use of add-on therapy, even though the reduction in hemoglobin A1c was modest (0.6%), suggesting that pleiotropic effects are at work.
Disadvantages, adverse effects. The most common adverse effects of this class include urinary tract infections, yeast infections, dehydration, and hypovolemic symptoms; these can often be prevented. A trend toward increased incidence of amputations in earlier studies was not borne out in a 2018 meta-analysis of 4 observational databases.46
Thiazolidinediones
There are currently 2 approved thiazolidinediones in the United States, pioglitazone47 and rosiglitazone.48 Only pioglitazone is in common use, as rosiglitazone is associated with safety issues.49
Pathways affected. Pioglitazone reduces insulin resistance in muscle, liver, and adipose tissue.
Advantages, benefits. Decreased levels of low-density lipoprotein cholesterol and triglycerides and increased high-density lipoprotein cholesterol levels49 could plausibly account for the cardiovascular benefits reported in the Prospective Pioglitazone Clinical Trial in Macrovascular Events.50 Pioglitazone has also been found to improve insulin secretion, endothelial function, and diastolic dysfunction; reduce inflammation; decrease plasminogen activator inhibitor 1; reverse lipotoxicity; and help correct nonalcoholic fatty liver disease and steatohepatitis.
Pioglitazone has also been found to reduce plaque in carotid and coronary arteries51; improve outcomes in patients with heart failure and myocardial infarction compared with insulin-sensitizing drugs52; and reduce stroke and myocardial infarction in patients with insulin resistance (but not diabetes) and a recent history of ischemic stroke or transient ischemic attack (in the Insulin Resistance Intervention After Stroke trial).53 It may also help maintain beta-cell function; the Actos Now for the Prevention of Diabetes Study found that pioglitazone reduced the risk of conversion of impaired glucose tolerance to frank diabetes by 72%.54
Disadvantages, adverse effects. The most common adverse effects seen with this class include weight gain and salt retention, swelling, edema,55 and related cardiovascular consequences in certain patients. While this may be mitigatable with lifestyle changes or use in combination with a GLP-1 receptor agonist or SGLT2 inhibitor,56 pioglitazone is contraindicated in patients with heart failure, hemodynamic instability, or hepatic dysfunction.
Concerns that pioglitazone might increase the risk of bladder cancer seem to have been put to rest when a study in nearly 200,000 patients found no statistically significant association,57 but the warning remains in the US label.
Long-term use of this class of drugs has been associated with an increased risk of bone fractures,58 which warrants a risk-benefit assessment in each patient.
Injected insulin: Less safe than thought
Recent research suggests that injected insulin has a less favorable safety profile than previously thought.15–19,59 Studies of the long-term safety of insulin therapy have had inconsistent results but suggest that injected insulin is associated with poorer cardiovascular and renal outcomes (in some of the same studies that showed metformin or other agents to improve outcomes),17–19 and the association was dose-dependent. Several studies attempted to cancel out the poorer outcomes by adjusting for hemoglobin A1c levels, stage of disease,17–19,26,27 or severe hypoglycemic episodes.60 However, it may be inappropriate to reduce the impact of these variables, as these may themselves be the mediators of any deleterious effects of exogenous insulin.
When exogenous insulin is introduced into the peripheral circulation it causes a state of persistent iatrogenic hyperinsulinemia, which leads to insulin resistance and also appears to compromise the cardiovascular system. In contrast, endogenous insulin is released into the portal system in tightly controlled amounts.5,61 This suggests that the same insulin peptide may not be equivalently beneficial when introduced in an artificial manner.
Before starting insulin therapy, consider its side effects such as weight gain and hypoglycemia. Most (about 85%) episodes of hypoglycemia occur with basal-bolus insulin regimens.62 Moreover, iatrogenic hyperinsulinemia can damage the vascular system.63,64
We recommend. Insulin therapy is used early in the course of the disease as a short-term intervention for glucolipotoxicity. However, this can be accomplished without attendant risks of hypoglycemia and weight gain by using agents such as SGLT2 inhibitors and incretins. When insulin therapy is necessary, using it as add-on therapy might be considered instead of drug-switching. We have found alternate pharmacologic approaches successful in avoiding or delaying bolus insulin therapy. And in some patients taking insulin, we have had success in progressively introducing a noninsulin agent and were ultimately able to eliminate insulin altogether.
Bromocriptine-QR
Bromocriptine-QR (quick release)65 is a short-acting dopamine agonist that mimics the morning dopamine surge in the suprachiasmatic nucleus—the biologic clock.
Pathways affected. Bromocriptine addresses part of the brain contribution to hyperglycemia, with resultant reductions in both peripheral insulin resistance and sympathetic tone. This reduces muscle, liver, and adipose insulin resistance. It is moderately effective in glucose-lowering, especially in patients with significant insulin resistance.66
Advantages, benefits. A 1-year clinical trial reported that bromocriptine reduced cardiovascular adverse outcomes by 39%, and the composite end point of myocardial infarction, stroke, and cardiovascular death by 52% compared with placebo.67
Disadvantages, adverse effects. The most common adverse effects are nausea, rhinitis, headache, asthenia, dizziness, constipation, and sinusitis.
Alpha-glucosidase inhibitors
Alpha-glucosidase inhibitors (acarbose,68 miglitol69) work by decreasing the rate of absorption of glucose from the gastrointestinal tract.
Advantages, benefits. These drugs decrease hemoglobin A1c by 0.5% to 0.8%.70 They are weight-neutral and do not pose a risk of hypoglycemia. Clinical studies suggest that they may delay or prevent diabetes progression. They were also found to reduce cardiovascular events, acute myocardial infarction, and the onset of hypertension.69
Disadvantages, adverse effects. Their use remains limited due to gastrointestinal adverse effects. They may be contraindicated in patients with inflammatory bowel disease, partial bowel obstruction, or severe renal or hepatic disease.
Pramlintide
Pramlintide71 is an injectable amylin analogue. It is used as monotherapy or in combination with a sulfonylurea, metformin, or insulin glargine.
Pathways affected. Pramlintide decreases appetite, reduces glucagon levels, and minimizes absorption of glucose in the gut.
Disadvantages, adverse effects. Common side effects include mild to moderate hypoglycemia and nausea. Nausea may help explain the ability of pramlintide to confer weight loss when used in combination with insulin.
Sulfonylureas and meglitinides
These classes are still widely used in the treatment of type 2 diabetes, although the AACE6 and ADA72 guidelines de-emphasize their use based on associated risks of hypoglycemia, weight gain, morbidity, mortality, and loss of effect over time.
Pathways affected. Sulfonylureas stimulate insulin secretion from beta cells.
Disadvantages, adverse effects. Sulfonylureas and glinides are associated with poorer outcomes than newer agents in clinical trials15–19,59,60 and may be generally less beta-cell friendly.73 Their harmful effects are difficult to measure in vivo, but these drugs sometimes appear to be associated with more rapid beta-cell failure and progression to insulin dependence compared with newer ones. Several large-scale registry studies have found sulfonylureas and glinides to be associated with poorer outcomes (reviewed by Herman et al).74
Adverse effects include asthenia, headache, dizziness, nausea, diarrhea, epigastric fullness, and heartburn. Although they are often selected based on their low cost, other factors may offset their cost-effectiveness, such as need for glucose monitoring and hospital charges due to sulfonylurea-induced hypoglycemia. Their utility is also limited by dependence on beta-cell function.
Colesevelam
Colesevelam75 is a bile acid sequestrant and low-density lipoprotein cholesterol-reducing agent that has been approved for use in diabetes. The mode of action of colesevelam in this capacity is under investigation. Its effect on hemoglobin A1c is modest. It is associated with gastrointestinal adverse effects, particularly constipation.
Ranolazine
Ranolazine76 is an antianginal drug that also lowers glucose by increasing insulin release. It also possesses cardioprotective properties. In patients with diabetes and non-ST-segment elevation acute coronary syndromes, ranolazine reduced hemoglobin A1c by 1.2% and appeared to be weight-neutral.76 Ranolazine is under clinical development for use in diabetes. Adverse effects include dizziness, headache, constipation, and nausea.
Rational combinations of agents
The ideal strategy would use combinations of agents that mechanistically complement one another and address each path of hyperglycemia present in a patient. This approach should supplant the former approaches of adding-on agents only after treatment failure or sequentially trying first-, second-, and third-line treatments.
Examples of synergistic combinations include those that target fasting plasma glucose and postprandial glucose, reduce reliance on insulin with add-on therapies, or manage hyperglycemia in specific patient groups, such as renal-impaired patients.
Large-scale long-term clinical studies are needed to determine the safety, efficacy, and outcomes of various combinations and whether they confer additive benefits. Some studies have begun to explore possible combinations.
Combined metformin, pioglitazone, and exenatide was reported to delay progression of diabetes in early dysglycemia.77,78 Notably, this combination addresses multiple mediating pathways of hyperglycemia (Table 1).
A GLP-1 receptor agonist with an SGLT2 inhibitor would be another intriguing combination, as the mechanisms of action of these 2 classes complement one another. In limited clinical trials—the DURATION-8 study (lasting 26 weeks),79 the Canagliflozin Cardiovascular Assessment Study (18 weeks),80 and a 24-week study in nondiabetic obese patients81—additive benefits were also seen in systolic blood pressure, body weight, and cardiac risk factors by adding an SGLT2 inhibitor to a GLP-1 receptor agonist, compared with either agent alone. In theory, these improvements might slow or reverse cardiorenal compromise. Lower doses of 1 or more may be possible, and the regimen could prove cost-effective and life-sparing should it slow the progression of the disease and the onset of its complications. A clinical study of this combination is under way (Ralph DeFronzo, personal communication, July 2018). Similarly, the combination of metformin, saxagliptin and dapagliflozin has been shown to be effective.82
CONCLUSION
Care for diabetes mellitus can be particularly challenging for the primary care physician. The progressive nature of diabetes, with worsening hyperglycemia over the course of the disease, further complicates disease management.
Best practices for care nonetheless need to evolve with well-evidenced data, and without years of delay for “trickle-down” education from the specialties to primary care. We have arrived at a juncture to leverage therapies that address the 11 mediating pathways of hyperglycemia, optimally protect beta cells, minimize hypoglycemia, manage risk factors associated with diabetes, and improve diabetes-related outcomes.
- Skyler JS, Bakris GL, Bonifacio E, et al. Differentiation of diabetes by pathophysiology, natural history, and prognosis. Diabetes 2017; 66(2):241–255. doi:10.2337/db16-0806
- Davies MJ, D’Alessio DA, Fradkin J, et al. Management of hyperglycemia in type 2 diabetes, 2018. A consensus report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetes Care 2018; 41(12):2669–2701. doi:10.2337/dci18-0033
- Schwartz SS, Epstein S, Corkey BE, Grant SF, Gavin JR 3rd, Aguilar RB. The time is right for a new classification system for diabetes mellitus: rationale and implications of the beta-cell centric classification schema. Diabetes Care 2016; 39(2):179–186. doi:10.2337/dc15-1585
- Shah MS, Brownlee M. Molecular and cellular mechanisms of cardiovascular disorders in diabetes. Circ Res 2016; 118(11):1808–1829. doi:10.1161/CIRCRESAHA.116.306923
- Schwartz SS, Jellinger PS, Herman ME. Obviating much of the need for insulin therapy in type 2 diabetes mellitus: a re-assessment of insulin therapy’s safety profile. Postgrad Med 2016; 128(6):609–619. doi:10.1080/00325481.2016.1191955
- Garber AJ, Abrahamson MJ, Barzilay JE, et al. Consensus statement by the American Association of Clinical Endocrinologists and American College of Endocrinology on the comprehensive type 2 diabetes management algorithm – 2019 executive summary. Endocr Pract 2019; 25(1):69–100. doi:10.4158/CS-2018-0535
- Sniderman AD, LaChapelle KJ, Rachon NA , Furberg CD. The necessity for clinical reasoning in the era of evidence-based medicine. Mayo Clin Proc 2013; 88(10):1108–1114. doi:10.1016/j.mayocp.2013.07.012
- Qaseem A, Wilt TJ, Kansagara D, Horwitch C, Barry MJ, Forciea MA; Clinical Guidelines Committee of the American College of Physicians. Hemoglobin A1c targets for glycemic control with pharmacologic therapy for nonpregnant adults with type 2 diabetes mellitus: a guidance statement update from the American College of Physicians. Ann Intern Med 2018; 168(8):569–576. doi:10.7326/M17-0939
- Frier BM, Schernthaner G, Heller SR. Hypoglycemia and cardiovascular risks. Diabetes Care 2011; 34(suppl 2):S132–S137. doi:10.2337/dc11-s220
- Chico A, Vidal-Ríos P, Subira M, Novials A. The continuous glucose monitoring system is useful for detecting unrecognized hypoglycemias in patients with type 1 and type 2 diabetes but is not better than frequent capillary glucose measurements for improving metabolic control. Diabetes Care 2003; 26(4):1153–1157. pmid:12663589
- Weber KK, Lohmann T, Busch K, Donati-Hirsch I, Riel R. High frequency of unrecognized hypoglycaemias in patients with type 2 diabetes is discovered by continuous glucose monitoring. Exp Clin Endocrinol Diabetes 2007; 115(8):491–494. doi:10.1055/s-2007-984452
- American Diabetes Association (ADA). The American Diabetes Association, the American Association of Clinical Endocrinologists, the American Association of Diabetes Educators and the Endocrine Society strongly disagree with the American College of Physicians’ guidance for higher blood glucose targets for people with type 2 diabetes www.diabetes.org/newsroom/press-releases/2018/joint-acp-guidance-response.html. Accessed June 6, 2019.
- Freed S; Diabetes in Control. American College of Physicians recommending controversial increase in A1c of 7% to 8%. www.diabetesincontrol.com/american-college-of-physicians-recommending-controversial-increase-in-a1c-of-7-to-8. Accessed June 6, 2019.
- Glucophage XR (metformin hydrochloride) extended release tablets prescribing information. Princeton, NJ, Bristol-Myers Squibb Company, 2009.
- Mellbin LG, Malmberg K, Norhammar A, Wedel H, Rydén L; DIGAMI 2 Investigators. The impact of glucose lowering treatment on long-term prognosis in patients with type 2 diabetes and myocardial infarction: a report from the DIGAMI 2 trial. Eur Heart J 2008; 29(2):166–176. doi:10.1093/eurheartj/ehm518
- Anselmino M, Ohrvik J, Malmberg K, Standl E, Rydén L; Euro Heart Survey Investigators. Glucose lowering treatment in patients with coronary artery disease is prognostically important not only in established but also in newly detected diabetes mellitus: a report from the Euro Heart Survey on Diabetes and the Heart. Eur Heart J 2008; 29(2):177–184. doi:10.1093/eurheartj/ehm519
- Smooke S, Horwich TB, Fonarow GC. Insulin-treated diabetes is associated with a marked increase in mortality in patients with advanced heart failure. Am Heart J 2005; 149(1):168–174. doi:10.1016/j.ahj.2004.07.005
- Colayco DC, Niu F, McCombs JS, Cheetham TC. A1C and cardiovascular outcomes in type 2 diabetes: a nested case-control study. Diabetes Care 2011; 34(1):77–83. doi:10.2337/dc10-1318
- Holden SE, Jenkins-Jones S, Morgan CL, Schernthaner G, Currie CJ. Glucose-lowering with exogenous insulin monotherapy in type 2 diabetes: dose association with all-cause mortality, cardiovascular events and cancer. Diabetes Obes Metab 2015; 17(4):350–362. doi:10.1111/dom.12412
- Tanzeum (albiglutide) prescribing information. Wilmington, DE, GlaxoSmithKline LLC, 2014.
- Trulicity (dulaglutide) prescribing information. Indianapolis, IN, Eli Lilly and Company, 2014.
- Byetta (exenatide) prescribing information. Wilmington, DE, AstraZeneca Pharmaceuticals LP, 2014.
- Victoza (liraglutide injection) prescribing information. Plainsboro, NJ, Novo Nordisk Inc, 2013.
- Adlyxin (lixisenatide injection) prescribing information. Bridgewater, NJ, Sanofi, 2016.
- Ozempic (semaglutide) prescribing information. Plainsboro, NJ, Novo Nordisk, 2017.
- Marso SP, Daniels GH, Brown-Frandsen K, et al; LEADER Steering Committee; LEADER Trial Investigators. Liraglutide and cardiovascular outcomes in type 2 diabetes. N Engl J Med 2016; 375(4):311–322. doi:10.1056/NEJMoa1603827
- Chang G, Zhang D, Yu H, et al. Cardioprotective effects of exenatide against oxidative stress-induced injury. Int J Mol Med 2013; 32(5):1011–1020. doi:10.3892/ijmm.2013.1475
- Best JH, Hoogwerf BJ, Herman WH, et al. Risk of cardiovascular disease events in patients with type 2 diabetes prescribed the glucagon-like peptide 1 (GLP-1) receptor agonist exenatide twice daily or other glucose-lowering therapies: a retrospective analysis of the LifeLink database. Diabetes Care; 34(1):90–95. doi:10.2337/dc10-1393
- Pfeffer MA, Claggett B, Diaz R, et al; ELIXA Investigators. Lixisenatide in patients with type 2 diabetes and acute coronary syndrome. N Engl J Med 2015; 373(23):2247–2257. doi:10.1056/NEJMoa1509225
- Marso SP, Bain SC, Consoli A, et al; SUSTAIN-6 Investigators. Semaglutide and cardiovascular outcomes in patients with type 2 diabetes. N Engl J Med 2016; 375(19):1834–1844. doi:10.1056/NEJMoa1607141
- Mentlein R. Mechanisms underlying the rapid degradation and elimination of the incretin hormones GLP-1 and GIP. Best Pract Res Clin Endocrinol Metab 2009; 23(4):443–452. doi:10.1016/j.beem.2009.03.005
- Onglyza (saxagliptin) tablets prescribing information. Wilmington, DE, AstraZeneca Pharmaceuticals LP, 2014.
- Januvia (sitagliptin) tablets prescribing information. Whitehouse Station, NJ, Merck & Co., Inc, 2014.
- Tradjenta (linagliptin) tablets prescribing information. Ingelheim, Germany, Boehringer Ingelheim International GmbH, 2014.
- Nesina (alogliptin) tablets prescribing information. Deerfield, IL, Takeda Pharmaceuticals America, Inc, 2013.
- Galvus (vildagliptin) prescribing information. North Ryde, Australia, Novartis Pharmaceuticals, 2014.
- Drucker DJ, Nauck MA. The incretin system: glucagon-like peptide-1 receptor agonists and dipeptidyl peptidase-4 inhibitors in type 2 diabetes. Lancet 2006; 368(9548):1696–1705. doi:10.1016/S0140-6736(06)69705-5
- Scirica BM, Bhatt DL, Braunwald E, et al; SAVOR-TIMI 53 Steering Committee and Investigators. Saxagliptin and cardiovascular outcomes in patients with type 2 diabetes mellitus. N Engl J Med 2013; 369(14):1317–1326. doi:10.1056/NEJMoa1307684
- Invokana (canagliflozin) tablets prescribing information. Titusville, NJ: Janssen Pharmaceuticals, Inc, 2013.
- Farxiga (dapagliflozin) prescribing information. Princeton, NJ, Bristol-Myers Squibb, 2014.
- Jardiance (empagliflozin) prescribing information. Ridgefield, CT, Boehringer Ingelheim Pharmaceuticals, Inc, 2014.
- Steglatro (ertugliflozin) prescribing information. Whitehouse Station, NJ, Merck, Sharp & Dohme Corp, 2017.
- Vasilakou D, Karagiannis T, Athanasiadou E, et al. Sodium-glucose cotransporter 2 inhibitors for type 2 diabetes: a systematic review and meta-analysis. Ann Intern Med 2013; 159(4):262–274. doi:10.7326/0003-4819-159-4-201308200-00007
- Neal B, Perkovic V, Mahaffey KW, et al; CANVAS Program Collaborative Group. Canagliflozin and cardiovascular and renal events in type 2 diabetes. N Engl J Med 2017; 377(7):644–657. doi:10.1056/NEJMoa1611925
- Zinman B, Wanner C, Lachin JM, et al; EMPA-REG OUTCOME Investigators. Empagliflozin, cardiovascular outcomes, and mortality in type 2 diabetes. N Engl J Med 2015; 373(22):2117–2128. doi:10.1056/NEJMoa1504720
- Ryan PB, Buse JB, Schuemie MJ, et al. Comparative effectiveness of canagliflozin, SGLT2 inhibitors and non-SGLT2 inhibitors on the risk of hospitalization for heart failure and amputation in patients with type 2 diabetes mellitus: a real-world meta-analysis of 4 observational databases (OBSERVE-4D). Diabetes Obes Metab 2018; 20(11):2485–2597. doi:10.1111/dom.13424
- Actos (pioglitazone) tablets for oral use prescribing information. Deerfield, IL, Takeda Pharmaceuticals America, Inc, 2013.
- Avandia (rosiglitazone maleate tablets) prescribing information. Research Triangle Park, NC, GlaxoSmithKline, 1999.
- Goldberg RB, Kendall DK, Deeg MA, et al; GLAI Study Investigators. A comparison of lipid and glycemic effects of pioglitazone and rosiglitazone in patients with type 2 diabetes and dyslipidemia. Diabetes Care 2005; 28(7):1547–1554. pmid:15983299
- Dormandy JA, Charbonnel B, Eckland DJ, et al; PROactive Investigators. Secondary prevention of macrovascular events in patients with type 2 diabetes in the PROactive Study (PROspective pioglitAzone clinical trial in macroVascular Events): a randomised controlled trial. Lancet 2005; 366:1279–1289. doi:10.1016/S0140-6736(05)67528-9
- Nissen SE, Nicholls SJ, Wolski K, et al; PERISCOPE Investigators. Comparison of pioglitazone vs glimepiride on progression of coronary atherosclerosis in patients with type 2 diabetes the PERISCOPE randomized controlled trial. JAMA 2008; 299(13):1561–1573. doi:10.1001/jama.299.13.1561
- Masoudi FA, Inzucchi SE, Wang Y, Havranek EP, Foody JM, Krumholz HM. Thiazolidinediones, metformin, and outcomes in older patients with diabetes and heart failure: an observational study. Circulation 2005; 111(5):583–590. doi:10.1161/01.CIR.0000154542.13412.B1
- Kernan WN, Viscoli CM, Furie KL, et al; IRIS Trial Investigators. Pioglitazone after ischemic stroke or transient ischemic attack. N Engl J Med 2016; 374(14):1321–1331. doi:10.1056/NEJMoa1506930
- DeFronzo RA, Tripathy D, Schwenke DC, et al; ACT NOW Study. Pioglitazone for diabetes prevention in impaired glucose tolerance. N Engl J Med 2011; 364(12):1104–1115. doi:10.1056/NEJMoa1010949
- Nesto RW, Bell D, Bonow RO, et al; American Heart Association; American Diabetes Association. Thiazolidinedione use, fluid retention, and congestive heart failure: a consensus statement from the American Heart Association and American Diabetes Association. October 7, 2003. Circulation 2003; 108(23):2941–2948. doi:10.1161/01.CIR.0000103683.99399.7E
- Kushner RF, Sujak M. Prevention of weight gain in adult patients with type 2 diabetes treated with pioglitazone. Obesity (Silver Spring) 2009; 17(5):1017–1022. doi:10.1038/oby.2008.651
- Lewis JD, Habel LA, Quesenberry CP, et al. Pioglitazone use and risk of bladder cancer and other common cancers in persons with diabetes. JAMA 2015; 314(3):265–277. doi:10.1001/jama.2015.7996
- Meier C, Kraenzlin ME, Bodmer M, Jick SS, Jick H, Meier CR. Use of thiazolidinediones and fracture risk. Arch Intern Med 2008; 168(8):820–825. doi:10.1001/archinte.168.8.820
- Gamble JM, Chibrikov E, Twells LK, et al. Association of insulin dosage with mortality or major adverse cardiovascular events: a retrospective cohort study. Lancet Diabetes Endocrinol 2017; 5(1):43–52. doi:10.1016/S2213-8587(16)30316-3
- Bonds DE, Miller ME, Bergenstal RM, et al. The association between symptomatic, severe hypoglycaemia and mortality in type 2 diabetes: retrospective epidemiological analysis of the ACCORD study. BMJ 2010; 340:b4909. doi:10.1136/bmj.b4909
- Wang X, Yu C, Zhang B, Wang Y. The injurious effects of hyperinsulinism on blood vessels. Cell Biochem Biophys 2014; 69(2):213–218. doi:10.1007/s12013-013-9810-6
- Garber AJ, King AB, Del Prato S, et al; NN1250-3582 (BEGIN BB T2D) Trial Investigators. Insulin degludec, an ultra-longacting basal insulin, versus insulin glargine in basal-bolus treatment with mealtime insulin aspart in type 2 diabetes (BEGIN Basal-Bolus Type 2): a phase 3, randomised, open-label, treat-to-target non-inferiority trial. Lancet 2012; 379(9825):1498–1507. doi:10.1016/S0140-6736(12)60205-0
- Hanefeld M, Monnier L, Schnell O, Owens D. Early treatment with basal insulin glargine in people with type 2 diabetes: lessons from ORIGIN and other cardiovascular trials. Diabetes Ther 2016; 7(2):187–201. doi:10.1007/s13300-016-0153-3
- Nolan CJ, Ruderman NB, Prentki M. Intensive insulin for type 2 diabetes: the risk of causing harm. Lancet Diabetes Endocrinol 2013; 1(1):9–10. doi:10.1016/S2213-8587(13)70027-5
- Cycloset (bromocriptine mesylate) tablets prescribing information. Tiverton, RI, VeroScience LLC, 2019.
- Schwartz S, Zangeneh F. Evidence-based practice use of quick-release bromocriptine across the natural history of type 2 diabetes mellitus. Postgrad Med 2016; 128(8):828–838. doi:10.1080/00325481.2016.1214059
- Gaziano JM, Cincotta AH, Vinik A, Blonde L, Bohannon N, Scranton R. Effect of bromocriptine-QR (a quick-release formulation of bromocriptine mesylate) on major adverse cardiovascular events in type 2 diabetes subjects. J Am Heart Assoc 2012; 1(5):e002279. doi:10.1161/JAHA.112.002279
- Precose (acarbose) tablets prescribing information. Germany, Bayer HealthCare Pharmaceuticals Inc, 2011.
- Glyset (miglitol) tablets prescribing information. Germany, Bayer HealthCare Pharmaceuticals, Inc, 2012.
- Van de Laar FA, Lucassen PL, Akkermans RP, Van de Lisdonk EH, Rutten GE, Van Weel C. Alpha-glucosidase inhibitors for type 2 diabetes mellitus. Cochrane Database Syst Rev 2005; (2):CD003639. doi:10.1002/14651858.CD003639.pub2
- Symlin (pramlintide acetate) injection for subcutaneous use prescribing information. Wilmongton, DE, AstraZeneca Pharmaceuticals LP, 2014.
- Inzucchi SE, Bergenstal RM, Buse JB, et al. Management of hyperglycaemia in type 2 diabetes, 2015: a patient-centred approach. Update to a position statement of the American Diabetes Association and the European Association for the Study of Diabetes. Diabetologia 2015; 58(3):429–442. doi:10.1007/s00125-014-3460-0
- Wajchenberg BL. Beta-cell failure in diabetes and preservation by clinical treatment. Endocr Rev 2007; 28(2):187–218. doi:10.1210/10.1210/er.2006-0038
- Herman ME, O’Keefe JH, Bell DSH, Schwartz SS. Insulin therapy increases cardiovascular risk in type 2 diabetes. Prog Cardiovasc Dis 2017; 60(3):422–434. doi:10.1016/j.pcad.2017.09.001
- Welchol (colesevelam hydrochloride) prescribing information. Parsippany, NJ, Daiichi Sankyo Inc, 2014.
- Ranexa (ranolazine) prescribing information. Foster City, CA: Gilead Sciences, Inc, 2016.
- Armato J, DeFronzo R, Abdul-Ghani M, Ruby R. Successful treatment of prediabetes in clinical practice: targeting insulin resistance and beta-cell dysfunction. Endocr Pract 2012; 18(3):342–350. doi:10.4158/EP11194.OR
- Abdul-Ghani MA, Puckett C, Triplitt C, et al. Initial combination therapy with metformin, pioglitazone and exenatide is more effective than sequential add-on therapy in subjects with new-onset diabetes. Results from the efficacy and durability of initial combination therapy for type 2 diabetes (EDICT): a randomized trial. Diabetes Obes Metab 2015; 17(3):268–275. doi:10.1111/dom.12417
- Frías JP, Guja C, Hardy E, et al. Exenatide once weekly plus dapagliflozin once daily versus exenatide or dapagliflozin alone in patients with type 2 diabetes inadequately controlled with metformin monotherapy (DURATION-8): a 28 week, multicentre, double-blind, phase 3, randomised controlled trial. Lancet Diabetes Endocrinol 2016; 4(12):1004–1016. doi:10.1016/S2213-8587(16)30267-4
- Fulcher G, Matthews DR, Perkovic V, et al; CANVAS trial collaborative group. Efficacy and safety of canagliflozin when used in conjunction with incretin-mimetic therapy in patients with type 2 diabetes. Diabetes Obes Metab 2016; 18(1):82–91. doi:10.1111/dom.12589
- Lundkvist P, Sjöström CD, Amini S, Pereira MJ, Johnsson E, Eriksson JW. Dapagliflozin once-daily and exenatide once-weekly dual therapy: a 24-week randomized, placebo-controlled, phase II study examining effects on body weight and prediabetes in obese adults without diabetes. Diabetes Obes Metab 2017; 19(1):49–60. doi:10.1111/dom.12779
- Del Prato S, Rosenstock J, Garcia-Sanchez R, et al. Safety and tolerability of dapagliflozin, saxagliptin and metformin in combination: post-hoc analysis of concomitant add-on versus sequential add-on to metformin and of triple versus dual therapy with metformin. Diabetes Obes Metab 2018; 20(6):1542–1546. doi:10.1111/dom.13258
- Skyler JS, Bakris GL, Bonifacio E, et al. Differentiation of diabetes by pathophysiology, natural history, and prognosis. Diabetes 2017; 66(2):241–255. doi:10.2337/db16-0806
- Davies MJ, D’Alessio DA, Fradkin J, et al. Management of hyperglycemia in type 2 diabetes, 2018. A consensus report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetes Care 2018; 41(12):2669–2701. doi:10.2337/dci18-0033
- Schwartz SS, Epstein S, Corkey BE, Grant SF, Gavin JR 3rd, Aguilar RB. The time is right for a new classification system for diabetes mellitus: rationale and implications of the beta-cell centric classification schema. Diabetes Care 2016; 39(2):179–186. doi:10.2337/dc15-1585
- Shah MS, Brownlee M. Molecular and cellular mechanisms of cardiovascular disorders in diabetes. Circ Res 2016; 118(11):1808–1829. doi:10.1161/CIRCRESAHA.116.306923
- Schwartz SS, Jellinger PS, Herman ME. Obviating much of the need for insulin therapy in type 2 diabetes mellitus: a re-assessment of insulin therapy’s safety profile. Postgrad Med 2016; 128(6):609–619. doi:10.1080/00325481.2016.1191955
- Garber AJ, Abrahamson MJ, Barzilay JE, et al. Consensus statement by the American Association of Clinical Endocrinologists and American College of Endocrinology on the comprehensive type 2 diabetes management algorithm – 2019 executive summary. Endocr Pract 2019; 25(1):69–100. doi:10.4158/CS-2018-0535
- Sniderman AD, LaChapelle KJ, Rachon NA , Furberg CD. The necessity for clinical reasoning in the era of evidence-based medicine. Mayo Clin Proc 2013; 88(10):1108–1114. doi:10.1016/j.mayocp.2013.07.012
- Qaseem A, Wilt TJ, Kansagara D, Horwitch C, Barry MJ, Forciea MA; Clinical Guidelines Committee of the American College of Physicians. Hemoglobin A1c targets for glycemic control with pharmacologic therapy for nonpregnant adults with type 2 diabetes mellitus: a guidance statement update from the American College of Physicians. Ann Intern Med 2018; 168(8):569–576. doi:10.7326/M17-0939
- Frier BM, Schernthaner G, Heller SR. Hypoglycemia and cardiovascular risks. Diabetes Care 2011; 34(suppl 2):S132–S137. doi:10.2337/dc11-s220
- Chico A, Vidal-Ríos P, Subira M, Novials A. The continuous glucose monitoring system is useful for detecting unrecognized hypoglycemias in patients with type 1 and type 2 diabetes but is not better than frequent capillary glucose measurements for improving metabolic control. Diabetes Care 2003; 26(4):1153–1157. pmid:12663589
- Weber KK, Lohmann T, Busch K, Donati-Hirsch I, Riel R. High frequency of unrecognized hypoglycaemias in patients with type 2 diabetes is discovered by continuous glucose monitoring. Exp Clin Endocrinol Diabetes 2007; 115(8):491–494. doi:10.1055/s-2007-984452
- American Diabetes Association (ADA). The American Diabetes Association, the American Association of Clinical Endocrinologists, the American Association of Diabetes Educators and the Endocrine Society strongly disagree with the American College of Physicians’ guidance for higher blood glucose targets for people with type 2 diabetes www.diabetes.org/newsroom/press-releases/2018/joint-acp-guidance-response.html. Accessed June 6, 2019.
- Freed S; Diabetes in Control. American College of Physicians recommending controversial increase in A1c of 7% to 8%. www.diabetesincontrol.com/american-college-of-physicians-recommending-controversial-increase-in-a1c-of-7-to-8. Accessed June 6, 2019.
- Glucophage XR (metformin hydrochloride) extended release tablets prescribing information. Princeton, NJ, Bristol-Myers Squibb Company, 2009.
- Mellbin LG, Malmberg K, Norhammar A, Wedel H, Rydén L; DIGAMI 2 Investigators. The impact of glucose lowering treatment on long-term prognosis in patients with type 2 diabetes and myocardial infarction: a report from the DIGAMI 2 trial. Eur Heart J 2008; 29(2):166–176. doi:10.1093/eurheartj/ehm518
- Anselmino M, Ohrvik J, Malmberg K, Standl E, Rydén L; Euro Heart Survey Investigators. Glucose lowering treatment in patients with coronary artery disease is prognostically important not only in established but also in newly detected diabetes mellitus: a report from the Euro Heart Survey on Diabetes and the Heart. Eur Heart J 2008; 29(2):177–184. doi:10.1093/eurheartj/ehm519
- Smooke S, Horwich TB, Fonarow GC. Insulin-treated diabetes is associated with a marked increase in mortality in patients with advanced heart failure. Am Heart J 2005; 149(1):168–174. doi:10.1016/j.ahj.2004.07.005
- Colayco DC, Niu F, McCombs JS, Cheetham TC. A1C and cardiovascular outcomes in type 2 diabetes: a nested case-control study. Diabetes Care 2011; 34(1):77–83. doi:10.2337/dc10-1318
- Holden SE, Jenkins-Jones S, Morgan CL, Schernthaner G, Currie CJ. Glucose-lowering with exogenous insulin monotherapy in type 2 diabetes: dose association with all-cause mortality, cardiovascular events and cancer. Diabetes Obes Metab 2015; 17(4):350–362. doi:10.1111/dom.12412
- Tanzeum (albiglutide) prescribing information. Wilmington, DE, GlaxoSmithKline LLC, 2014.
- Trulicity (dulaglutide) prescribing information. Indianapolis, IN, Eli Lilly and Company, 2014.
- Byetta (exenatide) prescribing information. Wilmington, DE, AstraZeneca Pharmaceuticals LP, 2014.
- Victoza (liraglutide injection) prescribing information. Plainsboro, NJ, Novo Nordisk Inc, 2013.
- Adlyxin (lixisenatide injection) prescribing information. Bridgewater, NJ, Sanofi, 2016.
- Ozempic (semaglutide) prescribing information. Plainsboro, NJ, Novo Nordisk, 2017.
- Marso SP, Daniels GH, Brown-Frandsen K, et al; LEADER Steering Committee; LEADER Trial Investigators. Liraglutide and cardiovascular outcomes in type 2 diabetes. N Engl J Med 2016; 375(4):311–322. doi:10.1056/NEJMoa1603827
- Chang G, Zhang D, Yu H, et al. Cardioprotective effects of exenatide against oxidative stress-induced injury. Int J Mol Med 2013; 32(5):1011–1020. doi:10.3892/ijmm.2013.1475
- Best JH, Hoogwerf BJ, Herman WH, et al. Risk of cardiovascular disease events in patients with type 2 diabetes prescribed the glucagon-like peptide 1 (GLP-1) receptor agonist exenatide twice daily or other glucose-lowering therapies: a retrospective analysis of the LifeLink database. Diabetes Care; 34(1):90–95. doi:10.2337/dc10-1393
- Pfeffer MA, Claggett B, Diaz R, et al; ELIXA Investigators. Lixisenatide in patients with type 2 diabetes and acute coronary syndrome. N Engl J Med 2015; 373(23):2247–2257. doi:10.1056/NEJMoa1509225
- Marso SP, Bain SC, Consoli A, et al; SUSTAIN-6 Investigators. Semaglutide and cardiovascular outcomes in patients with type 2 diabetes. N Engl J Med 2016; 375(19):1834–1844. doi:10.1056/NEJMoa1607141
- Mentlein R. Mechanisms underlying the rapid degradation and elimination of the incretin hormones GLP-1 and GIP. Best Pract Res Clin Endocrinol Metab 2009; 23(4):443–452. doi:10.1016/j.beem.2009.03.005
- Onglyza (saxagliptin) tablets prescribing information. Wilmington, DE, AstraZeneca Pharmaceuticals LP, 2014.
- Januvia (sitagliptin) tablets prescribing information. Whitehouse Station, NJ, Merck & Co., Inc, 2014.
- Tradjenta (linagliptin) tablets prescribing information. Ingelheim, Germany, Boehringer Ingelheim International GmbH, 2014.
- Nesina (alogliptin) tablets prescribing information. Deerfield, IL, Takeda Pharmaceuticals America, Inc, 2013.
- Galvus (vildagliptin) prescribing information. North Ryde, Australia, Novartis Pharmaceuticals, 2014.
- Drucker DJ, Nauck MA. The incretin system: glucagon-like peptide-1 receptor agonists and dipeptidyl peptidase-4 inhibitors in type 2 diabetes. Lancet 2006; 368(9548):1696–1705. doi:10.1016/S0140-6736(06)69705-5
- Scirica BM, Bhatt DL, Braunwald E, et al; SAVOR-TIMI 53 Steering Committee and Investigators. Saxagliptin and cardiovascular outcomes in patients with type 2 diabetes mellitus. N Engl J Med 2013; 369(14):1317–1326. doi:10.1056/NEJMoa1307684
- Invokana (canagliflozin) tablets prescribing information. Titusville, NJ: Janssen Pharmaceuticals, Inc, 2013.
- Farxiga (dapagliflozin) prescribing information. Princeton, NJ, Bristol-Myers Squibb, 2014.
- Jardiance (empagliflozin) prescribing information. Ridgefield, CT, Boehringer Ingelheim Pharmaceuticals, Inc, 2014.
- Steglatro (ertugliflozin) prescribing information. Whitehouse Station, NJ, Merck, Sharp & Dohme Corp, 2017.
- Vasilakou D, Karagiannis T, Athanasiadou E, et al. Sodium-glucose cotransporter 2 inhibitors for type 2 diabetes: a systematic review and meta-analysis. Ann Intern Med 2013; 159(4):262–274. doi:10.7326/0003-4819-159-4-201308200-00007
- Neal B, Perkovic V, Mahaffey KW, et al; CANVAS Program Collaborative Group. Canagliflozin and cardiovascular and renal events in type 2 diabetes. N Engl J Med 2017; 377(7):644–657. doi:10.1056/NEJMoa1611925
- Zinman B, Wanner C, Lachin JM, et al; EMPA-REG OUTCOME Investigators. Empagliflozin, cardiovascular outcomes, and mortality in type 2 diabetes. N Engl J Med 2015; 373(22):2117–2128. doi:10.1056/NEJMoa1504720
- Ryan PB, Buse JB, Schuemie MJ, et al. Comparative effectiveness of canagliflozin, SGLT2 inhibitors and non-SGLT2 inhibitors on the risk of hospitalization for heart failure and amputation in patients with type 2 diabetes mellitus: a real-world meta-analysis of 4 observational databases (OBSERVE-4D). Diabetes Obes Metab 2018; 20(11):2485–2597. doi:10.1111/dom.13424
- Actos (pioglitazone) tablets for oral use prescribing information. Deerfield, IL, Takeda Pharmaceuticals America, Inc, 2013.
- Avandia (rosiglitazone maleate tablets) prescribing information. Research Triangle Park, NC, GlaxoSmithKline, 1999.
- Goldberg RB, Kendall DK, Deeg MA, et al; GLAI Study Investigators. A comparison of lipid and glycemic effects of pioglitazone and rosiglitazone in patients with type 2 diabetes and dyslipidemia. Diabetes Care 2005; 28(7):1547–1554. pmid:15983299
- Dormandy JA, Charbonnel B, Eckland DJ, et al; PROactive Investigators. Secondary prevention of macrovascular events in patients with type 2 diabetes in the PROactive Study (PROspective pioglitAzone clinical trial in macroVascular Events): a randomised controlled trial. Lancet 2005; 366:1279–1289. doi:10.1016/S0140-6736(05)67528-9
- Nissen SE, Nicholls SJ, Wolski K, et al; PERISCOPE Investigators. Comparison of pioglitazone vs glimepiride on progression of coronary atherosclerosis in patients with type 2 diabetes the PERISCOPE randomized controlled trial. JAMA 2008; 299(13):1561–1573. doi:10.1001/jama.299.13.1561
- Masoudi FA, Inzucchi SE, Wang Y, Havranek EP, Foody JM, Krumholz HM. Thiazolidinediones, metformin, and outcomes in older patients with diabetes and heart failure: an observational study. Circulation 2005; 111(5):583–590. doi:10.1161/01.CIR.0000154542.13412.B1
- Kernan WN, Viscoli CM, Furie KL, et al; IRIS Trial Investigators. Pioglitazone after ischemic stroke or transient ischemic attack. N Engl J Med 2016; 374(14):1321–1331. doi:10.1056/NEJMoa1506930
- DeFronzo RA, Tripathy D, Schwenke DC, et al; ACT NOW Study. Pioglitazone for diabetes prevention in impaired glucose tolerance. N Engl J Med 2011; 364(12):1104–1115. doi:10.1056/NEJMoa1010949
- Nesto RW, Bell D, Bonow RO, et al; American Heart Association; American Diabetes Association. Thiazolidinedione use, fluid retention, and congestive heart failure: a consensus statement from the American Heart Association and American Diabetes Association. October 7, 2003. Circulation 2003; 108(23):2941–2948. doi:10.1161/01.CIR.0000103683.99399.7E
- Kushner RF, Sujak M. Prevention of weight gain in adult patients with type 2 diabetes treated with pioglitazone. Obesity (Silver Spring) 2009; 17(5):1017–1022. doi:10.1038/oby.2008.651
- Lewis JD, Habel LA, Quesenberry CP, et al. Pioglitazone use and risk of bladder cancer and other common cancers in persons with diabetes. JAMA 2015; 314(3):265–277. doi:10.1001/jama.2015.7996
- Meier C, Kraenzlin ME, Bodmer M, Jick SS, Jick H, Meier CR. Use of thiazolidinediones and fracture risk. Arch Intern Med 2008; 168(8):820–825. doi:10.1001/archinte.168.8.820
- Gamble JM, Chibrikov E, Twells LK, et al. Association of insulin dosage with mortality or major adverse cardiovascular events: a retrospective cohort study. Lancet Diabetes Endocrinol 2017; 5(1):43–52. doi:10.1016/S2213-8587(16)30316-3
- Bonds DE, Miller ME, Bergenstal RM, et al. The association between symptomatic, severe hypoglycaemia and mortality in type 2 diabetes: retrospective epidemiological analysis of the ACCORD study. BMJ 2010; 340:b4909. doi:10.1136/bmj.b4909
- Wang X, Yu C, Zhang B, Wang Y. The injurious effects of hyperinsulinism on blood vessels. Cell Biochem Biophys 2014; 69(2):213–218. doi:10.1007/s12013-013-9810-6
- Garber AJ, King AB, Del Prato S, et al; NN1250-3582 (BEGIN BB T2D) Trial Investigators. Insulin degludec, an ultra-longacting basal insulin, versus insulin glargine in basal-bolus treatment with mealtime insulin aspart in type 2 diabetes (BEGIN Basal-Bolus Type 2): a phase 3, randomised, open-label, treat-to-target non-inferiority trial. Lancet 2012; 379(9825):1498–1507. doi:10.1016/S0140-6736(12)60205-0
- Hanefeld M, Monnier L, Schnell O, Owens D. Early treatment with basal insulin glargine in people with type 2 diabetes: lessons from ORIGIN and other cardiovascular trials. Diabetes Ther 2016; 7(2):187–201. doi:10.1007/s13300-016-0153-3
- Nolan CJ, Ruderman NB, Prentki M. Intensive insulin for type 2 diabetes: the risk of causing harm. Lancet Diabetes Endocrinol 2013; 1(1):9–10. doi:10.1016/S2213-8587(13)70027-5
- Cycloset (bromocriptine mesylate) tablets prescribing information. Tiverton, RI, VeroScience LLC, 2019.
- Schwartz S, Zangeneh F. Evidence-based practice use of quick-release bromocriptine across the natural history of type 2 diabetes mellitus. Postgrad Med 2016; 128(8):828–838. doi:10.1080/00325481.2016.1214059
- Gaziano JM, Cincotta AH, Vinik A, Blonde L, Bohannon N, Scranton R. Effect of bromocriptine-QR (a quick-release formulation of bromocriptine mesylate) on major adverse cardiovascular events in type 2 diabetes subjects. J Am Heart Assoc 2012; 1(5):e002279. doi:10.1161/JAHA.112.002279
- Precose (acarbose) tablets prescribing information. Germany, Bayer HealthCare Pharmaceuticals Inc, 2011.
- Glyset (miglitol) tablets prescribing information. Germany, Bayer HealthCare Pharmaceuticals, Inc, 2012.
- Van de Laar FA, Lucassen PL, Akkermans RP, Van de Lisdonk EH, Rutten GE, Van Weel C. Alpha-glucosidase inhibitors for type 2 diabetes mellitus. Cochrane Database Syst Rev 2005; (2):CD003639. doi:10.1002/14651858.CD003639.pub2
- Symlin (pramlintide acetate) injection for subcutaneous use prescribing information. Wilmongton, DE, AstraZeneca Pharmaceuticals LP, 2014.
- Inzucchi SE, Bergenstal RM, Buse JB, et al. Management of hyperglycaemia in type 2 diabetes, 2015: a patient-centred approach. Update to a position statement of the American Diabetes Association and the European Association for the Study of Diabetes. Diabetologia 2015; 58(3):429–442. doi:10.1007/s00125-014-3460-0
- Wajchenberg BL. Beta-cell failure in diabetes and preservation by clinical treatment. Endocr Rev 2007; 28(2):187–218. doi:10.1210/10.1210/er.2006-0038
- Herman ME, O’Keefe JH, Bell DSH, Schwartz SS. Insulin therapy increases cardiovascular risk in type 2 diabetes. Prog Cardiovasc Dis 2017; 60(3):422–434. doi:10.1016/j.pcad.2017.09.001
- Welchol (colesevelam hydrochloride) prescribing information. Parsippany, NJ, Daiichi Sankyo Inc, 2014.
- Ranexa (ranolazine) prescribing information. Foster City, CA: Gilead Sciences, Inc, 2016.
- Armato J, DeFronzo R, Abdul-Ghani M, Ruby R. Successful treatment of prediabetes in clinical practice: targeting insulin resistance and beta-cell dysfunction. Endocr Pract 2012; 18(3):342–350. doi:10.4158/EP11194.OR
- Abdul-Ghani MA, Puckett C, Triplitt C, et al. Initial combination therapy with metformin, pioglitazone and exenatide is more effective than sequential add-on therapy in subjects with new-onset diabetes. Results from the efficacy and durability of initial combination therapy for type 2 diabetes (EDICT): a randomized trial. Diabetes Obes Metab 2015; 17(3):268–275. doi:10.1111/dom.12417
- Frías JP, Guja C, Hardy E, et al. Exenatide once weekly plus dapagliflozin once daily versus exenatide or dapagliflozin alone in patients with type 2 diabetes inadequately controlled with metformin monotherapy (DURATION-8): a 28 week, multicentre, double-blind, phase 3, randomised controlled trial. Lancet Diabetes Endocrinol 2016; 4(12):1004–1016. doi:10.1016/S2213-8587(16)30267-4
- Fulcher G, Matthews DR, Perkovic V, et al; CANVAS trial collaborative group. Efficacy and safety of canagliflozin when used in conjunction with incretin-mimetic therapy in patients with type 2 diabetes. Diabetes Obes Metab 2016; 18(1):82–91. doi:10.1111/dom.12589
- Lundkvist P, Sjöström CD, Amini S, Pereira MJ, Johnsson E, Eriksson JW. Dapagliflozin once-daily and exenatide once-weekly dual therapy: a 24-week randomized, placebo-controlled, phase II study examining effects on body weight and prediabetes in obese adults without diabetes. Diabetes Obes Metab 2017; 19(1):49–60. doi:10.1111/dom.12779
- Del Prato S, Rosenstock J, Garcia-Sanchez R, et al. Safety and tolerability of dapagliflozin, saxagliptin and metformin in combination: post-hoc analysis of concomitant add-on versus sequential add-on to metformin and of triple versus dual therapy with metformin. Diabetes Obes Metab 2018; 20(6):1542–1546. doi:10.1111/dom.13258
KEY POINTS
- At least 11 pathways lead to hyperglycemia; of these, beta-cell dysfunction is central.
- As different classes of diabetes drugs act on different pathways, we can target the pathways contributing to hyperglycemia in the individual patient, using fewer agents and lessening the risk of hypoglycemic episodes.
- In selecting treatment, we should favor drugs that are “gentle” on beta cells, do not cause dangerous hypoglycemia, and improve long-term outcomes as shown in randomized clinical trials.
FDA issues warning on insulin pump cybersecurity weakness
The Food and Drug Administration has issued a warning to patients and health care providers that a pair of Medtronic insulin pumps are being recalled because of potential cybersecurity risks, according to a press release.
The affected devices are the MiniMed 508 and MiniMed Paradigm series insulin pumps, which wirelessly connect to both the patient’s blood glucose meter and continuous glucose monitoring system. A remote controller and CareLink USB – a thumb-sized wireless device that plugs into a computer – are used to operate the devices; the remote controller sends insulin dosing commands to the pump and the CareLink USB can be used to download and share data with the patient’s health care provider.
The potential risk involves the wireless communication between the pumps and related devices such as the blood glucose meter and remote controller. The FDA has identified a cybersecurity vulnerability within the insulin pumps, and is concerned that a third party could connect to the device and change the pump’s settings. Insulin could be given in excess, causing hypoglycemia, or stopped, causing hyperglycemia or diabetic ketoacidosis.
Medtronic has identified 4,000 patients in the United States who are affected by the security weakness. Because the company is unable to adequately update or patch the device to remove the weakness, the FDA is working to ensure that Medtronic addresses the issue in any way possible, including helping patients with affected pumps switch to newer models.
“While we are not aware of patients who may have been harmed by this particular cybersecurity vulnerability, the risk of patient harm if such a vulnerability were left unaddressed is significant. The safety communication issued today contains recommendations for what actions patients and health care providers should take to avoid the risk this vulnerability could pose,” said Suzanne Schwartz, MD, MBA, deputy director of the Office of Strategic Partnerships and Technology Innovation.
Find the full press release on the FDA website.
The Food and Drug Administration has issued a warning to patients and health care providers that a pair of Medtronic insulin pumps are being recalled because of potential cybersecurity risks, according to a press release.
The affected devices are the MiniMed 508 and MiniMed Paradigm series insulin pumps, which wirelessly connect to both the patient’s blood glucose meter and continuous glucose monitoring system. A remote controller and CareLink USB – a thumb-sized wireless device that plugs into a computer – are used to operate the devices; the remote controller sends insulin dosing commands to the pump and the CareLink USB can be used to download and share data with the patient’s health care provider.
The potential risk involves the wireless communication between the pumps and related devices such as the blood glucose meter and remote controller. The FDA has identified a cybersecurity vulnerability within the insulin pumps, and is concerned that a third party could connect to the device and change the pump’s settings. Insulin could be given in excess, causing hypoglycemia, or stopped, causing hyperglycemia or diabetic ketoacidosis.
Medtronic has identified 4,000 patients in the United States who are affected by the security weakness. Because the company is unable to adequately update or patch the device to remove the weakness, the FDA is working to ensure that Medtronic addresses the issue in any way possible, including helping patients with affected pumps switch to newer models.
“While we are not aware of patients who may have been harmed by this particular cybersecurity vulnerability, the risk of patient harm if such a vulnerability were left unaddressed is significant. The safety communication issued today contains recommendations for what actions patients and health care providers should take to avoid the risk this vulnerability could pose,” said Suzanne Schwartz, MD, MBA, deputy director of the Office of Strategic Partnerships and Technology Innovation.
Find the full press release on the FDA website.
The Food and Drug Administration has issued a warning to patients and health care providers that a pair of Medtronic insulin pumps are being recalled because of potential cybersecurity risks, according to a press release.
The affected devices are the MiniMed 508 and MiniMed Paradigm series insulin pumps, which wirelessly connect to both the patient’s blood glucose meter and continuous glucose monitoring system. A remote controller and CareLink USB – a thumb-sized wireless device that plugs into a computer – are used to operate the devices; the remote controller sends insulin dosing commands to the pump and the CareLink USB can be used to download and share data with the patient’s health care provider.
The potential risk involves the wireless communication between the pumps and related devices such as the blood glucose meter and remote controller. The FDA has identified a cybersecurity vulnerability within the insulin pumps, and is concerned that a third party could connect to the device and change the pump’s settings. Insulin could be given in excess, causing hypoglycemia, or stopped, causing hyperglycemia or diabetic ketoacidosis.
Medtronic has identified 4,000 patients in the United States who are affected by the security weakness. Because the company is unable to adequately update or patch the device to remove the weakness, the FDA is working to ensure that Medtronic addresses the issue in any way possible, including helping patients with affected pumps switch to newer models.
“While we are not aware of patients who may have been harmed by this particular cybersecurity vulnerability, the risk of patient harm if such a vulnerability were left unaddressed is significant. The safety communication issued today contains recommendations for what actions patients and health care providers should take to avoid the risk this vulnerability could pose,” said Suzanne Schwartz, MD, MBA, deputy director of the Office of Strategic Partnerships and Technology Innovation.
Find the full press release on the FDA website.
Bisphosphonates before denosumab may prevent postdenosumab BMD rebound effect
MADRID – Results from an ongoing study of postmenopausal women who discontinue osteoporosis treatment with denosumab (Prolia) so far support the use of denosumab as a second-line therapy after a bisphosphonate, unless otherwise indicated, in order to reduce the loss of bone mineral density (BMD) after its discontinuation and also to support treatment to reduce bone turnover biomarkers as much as possible after stopping denosumab.
“We saw in our study that, even if you give bisphosphonates after denosumab discontinuation, [patients] could lose bone, and the group that controlled the loss of bone had very high control of bone turnover markers,” study author and presenter Bérengère Rozier Aubry, MD, said in an interview at the European Congress of Rheumatology.
She and her colleagues at the Center of Bone Diseases at Lausanne (Switzerland) University Hospital are conducting the ReoLaus (Rebound Effect Observatory in Lausanne) Bone Project to determine whether giving a bisphosphonate to postmenopausal women with osteoporosis after they have discontinued denosumab can stop the loss of bone mineral density (BMD) observed in many patients up to 2 years after stopping denosumab. This postdenosumab BMD loss has also been observed to occur with multiple spontaneous vertebral fractures.
Nearly half of patients who start denosumab discontinue it within 1 year, and 64% by 2 years, according to U.S. administrative claims data (Osteoporos Int. 2017 Apr. doi: 10.1007/s00198-016-3886-y), even though it can be taken for up to 10 years. The discontinuation is either because the patient wishes to do so or there’s a medical indication such as stopping aromatase inhibitor treatment, resolution of osteoporosis, or side effects, Dr. Rozier Aubry said in a press conference at the European Congress of Rheumatology.
Upon discontinuing denosumab, there’s a marked rebound effect in which levels of bone turnover markers rise for 2 years, and some or all of the BMD that was gained is lost (J Clin Endocrinol Metab. 2011 Apr. doi: 10.1210/jc.2010-1502). Multiple spontaneous vertebral fractures also have been reported in 5%-7%, as Dr. Rozier Aubry and colleagues first described in 2016 (Osteoporos Int. 2016 May. doi: 10.1007/s00198-015-3380-y) and others have reported subsequently.
Recommendations from the Endocrine Society in March 2019, a 2017 position statement from the European Calcified Tissue Society, and guidelines from other groups advise giving antiresorptive treatment (bisphosphonates, hormone therapy, or selective estrogen-receptor modulators) but do not say which one, in what dose, when, or for how long, Dr. Rozier Aubry noted.
Treatment with zoledronate 6 months after the last denosumab injection achieves partial preservation of BMD, but multiple vertebral fractures have still been reported when raloxifene, ibandronate, or alendronate have been given after stopping denosumab, she said.
In the ReoLaus Bone Project, Dr. Rozier Aubry and associates are following 170 postmenopausal women with osteoporosis at Lausanne University Hospital who are taking denosumab therapy. At the congress, she reported on the first 71 women in the cohort with 1 year of follow-up. They had a mean age of 64 years, had fewer than one prevalent fracture before starting denosumab, and stopped denosumab after a mean of 7.7 injections. Overall, 8% took glucocorticoids, and 22% took aromatase inhibitors.
The investigators collected data on what treatment was used after denosumab, how bone turnover markers changed 1-3 months after the last denosumab injection and then regularly afterward, how bone mineral density changed after 1 year, and any new osteoporotic fractures.
At the time of denosumab discontinuation, 59% received zoledronate, 24% alendronate, 3% other drugs, and 14% nothing. At a mean of about 17 months after the last denosumab injection, the investigators classified 30 patients as BMD losers (losing at least 3.96%), and 41 had stable BMD. The researchers found that BMD losers were younger (61.4 years vs. 65.5 years), were less likely to use zoledronate before starting denosumab (0% vs. 12%), and had greater serum CTX (C-telopeptide cross-linked type 1 collagen) levels at denosumab initiation (644 ng/mL vs. 474 ng/mL) and 12.8 months after stopping denosumab (592 ng/mL vs. 336 ng/mL) than did those with stable BMD. All differences were statistically significant.
“Our results support the use of denosumab in second line after bisphosphonate therapy to restrain the BMD loss at its discontinuation ... and a strategy to maintain the bone turnover marker serum CTX as low as possible after denosumab discontinuation,” she concluded.
“Our proposition is to start with 1 or 2 years of bisphosphonates, and if the osteoporosis is severe, to switch to denosumab treatment for 4, 6 years. … We can use denosumab for 10 years without side effects, and after that we give bisphosphonates to consolidate the treatment,” she said.
Dr. Rozier Aubry and her associates plan to follow patients in their study for 2 years.
Dr. Rozier Aubry disclosed serving on speakers bureaus for Eli Lilly, Pfizer, Amgen, and Novartis.
SOURCE: Rozier Aubry B et al. Ann Rheum Dis. Jun 2019;78(Suppl 2):115; Abstract OP0085. doi: 10.1136/annrheumdis-2019-eular.4175.
MADRID – Results from an ongoing study of postmenopausal women who discontinue osteoporosis treatment with denosumab (Prolia) so far support the use of denosumab as a second-line therapy after a bisphosphonate, unless otherwise indicated, in order to reduce the loss of bone mineral density (BMD) after its discontinuation and also to support treatment to reduce bone turnover biomarkers as much as possible after stopping denosumab.
“We saw in our study that, even if you give bisphosphonates after denosumab discontinuation, [patients] could lose bone, and the group that controlled the loss of bone had very high control of bone turnover markers,” study author and presenter Bérengère Rozier Aubry, MD, said in an interview at the European Congress of Rheumatology.
She and her colleagues at the Center of Bone Diseases at Lausanne (Switzerland) University Hospital are conducting the ReoLaus (Rebound Effect Observatory in Lausanne) Bone Project to determine whether giving a bisphosphonate to postmenopausal women with osteoporosis after they have discontinued denosumab can stop the loss of bone mineral density (BMD) observed in many patients up to 2 years after stopping denosumab. This postdenosumab BMD loss has also been observed to occur with multiple spontaneous vertebral fractures.
Nearly half of patients who start denosumab discontinue it within 1 year, and 64% by 2 years, according to U.S. administrative claims data (Osteoporos Int. 2017 Apr. doi: 10.1007/s00198-016-3886-y), even though it can be taken for up to 10 years. The discontinuation is either because the patient wishes to do so or there’s a medical indication such as stopping aromatase inhibitor treatment, resolution of osteoporosis, or side effects, Dr. Rozier Aubry said in a press conference at the European Congress of Rheumatology.
Upon discontinuing denosumab, there’s a marked rebound effect in which levels of bone turnover markers rise for 2 years, and some or all of the BMD that was gained is lost (J Clin Endocrinol Metab. 2011 Apr. doi: 10.1210/jc.2010-1502). Multiple spontaneous vertebral fractures also have been reported in 5%-7%, as Dr. Rozier Aubry and colleagues first described in 2016 (Osteoporos Int. 2016 May. doi: 10.1007/s00198-015-3380-y) and others have reported subsequently.
Recommendations from the Endocrine Society in March 2019, a 2017 position statement from the European Calcified Tissue Society, and guidelines from other groups advise giving antiresorptive treatment (bisphosphonates, hormone therapy, or selective estrogen-receptor modulators) but do not say which one, in what dose, when, or for how long, Dr. Rozier Aubry noted.
Treatment with zoledronate 6 months after the last denosumab injection achieves partial preservation of BMD, but multiple vertebral fractures have still been reported when raloxifene, ibandronate, or alendronate have been given after stopping denosumab, she said.
In the ReoLaus Bone Project, Dr. Rozier Aubry and associates are following 170 postmenopausal women with osteoporosis at Lausanne University Hospital who are taking denosumab therapy. At the congress, she reported on the first 71 women in the cohort with 1 year of follow-up. They had a mean age of 64 years, had fewer than one prevalent fracture before starting denosumab, and stopped denosumab after a mean of 7.7 injections. Overall, 8% took glucocorticoids, and 22% took aromatase inhibitors.
The investigators collected data on what treatment was used after denosumab, how bone turnover markers changed 1-3 months after the last denosumab injection and then regularly afterward, how bone mineral density changed after 1 year, and any new osteoporotic fractures.
At the time of denosumab discontinuation, 59% received zoledronate, 24% alendronate, 3% other drugs, and 14% nothing. At a mean of about 17 months after the last denosumab injection, the investigators classified 30 patients as BMD losers (losing at least 3.96%), and 41 had stable BMD. The researchers found that BMD losers were younger (61.4 years vs. 65.5 years), were less likely to use zoledronate before starting denosumab (0% vs. 12%), and had greater serum CTX (C-telopeptide cross-linked type 1 collagen) levels at denosumab initiation (644 ng/mL vs. 474 ng/mL) and 12.8 months after stopping denosumab (592 ng/mL vs. 336 ng/mL) than did those with stable BMD. All differences were statistically significant.
“Our results support the use of denosumab in second line after bisphosphonate therapy to restrain the BMD loss at its discontinuation ... and a strategy to maintain the bone turnover marker serum CTX as low as possible after denosumab discontinuation,” she concluded.
“Our proposition is to start with 1 or 2 years of bisphosphonates, and if the osteoporosis is severe, to switch to denosumab treatment for 4, 6 years. … We can use denosumab for 10 years without side effects, and after that we give bisphosphonates to consolidate the treatment,” she said.
Dr. Rozier Aubry and her associates plan to follow patients in their study for 2 years.
Dr. Rozier Aubry disclosed serving on speakers bureaus for Eli Lilly, Pfizer, Amgen, and Novartis.
SOURCE: Rozier Aubry B et al. Ann Rheum Dis. Jun 2019;78(Suppl 2):115; Abstract OP0085. doi: 10.1136/annrheumdis-2019-eular.4175.
MADRID – Results from an ongoing study of postmenopausal women who discontinue osteoporosis treatment with denosumab (Prolia) so far support the use of denosumab as a second-line therapy after a bisphosphonate, unless otherwise indicated, in order to reduce the loss of bone mineral density (BMD) after its discontinuation and also to support treatment to reduce bone turnover biomarkers as much as possible after stopping denosumab.
“We saw in our study that, even if you give bisphosphonates after denosumab discontinuation, [patients] could lose bone, and the group that controlled the loss of bone had very high control of bone turnover markers,” study author and presenter Bérengère Rozier Aubry, MD, said in an interview at the European Congress of Rheumatology.
She and her colleagues at the Center of Bone Diseases at Lausanne (Switzerland) University Hospital are conducting the ReoLaus (Rebound Effect Observatory in Lausanne) Bone Project to determine whether giving a bisphosphonate to postmenopausal women with osteoporosis after they have discontinued denosumab can stop the loss of bone mineral density (BMD) observed in many patients up to 2 years after stopping denosumab. This postdenosumab BMD loss has also been observed to occur with multiple spontaneous vertebral fractures.
Nearly half of patients who start denosumab discontinue it within 1 year, and 64% by 2 years, according to U.S. administrative claims data (Osteoporos Int. 2017 Apr. doi: 10.1007/s00198-016-3886-y), even though it can be taken for up to 10 years. The discontinuation is either because the patient wishes to do so or there’s a medical indication such as stopping aromatase inhibitor treatment, resolution of osteoporosis, or side effects, Dr. Rozier Aubry said in a press conference at the European Congress of Rheumatology.
Upon discontinuing denosumab, there’s a marked rebound effect in which levels of bone turnover markers rise for 2 years, and some or all of the BMD that was gained is lost (J Clin Endocrinol Metab. 2011 Apr. doi: 10.1210/jc.2010-1502). Multiple spontaneous vertebral fractures also have been reported in 5%-7%, as Dr. Rozier Aubry and colleagues first described in 2016 (Osteoporos Int. 2016 May. doi: 10.1007/s00198-015-3380-y) and others have reported subsequently.
Recommendations from the Endocrine Society in March 2019, a 2017 position statement from the European Calcified Tissue Society, and guidelines from other groups advise giving antiresorptive treatment (bisphosphonates, hormone therapy, or selective estrogen-receptor modulators) but do not say which one, in what dose, when, or for how long, Dr. Rozier Aubry noted.
Treatment with zoledronate 6 months after the last denosumab injection achieves partial preservation of BMD, but multiple vertebral fractures have still been reported when raloxifene, ibandronate, or alendronate have been given after stopping denosumab, she said.
In the ReoLaus Bone Project, Dr. Rozier Aubry and associates are following 170 postmenopausal women with osteoporosis at Lausanne University Hospital who are taking denosumab therapy. At the congress, she reported on the first 71 women in the cohort with 1 year of follow-up. They had a mean age of 64 years, had fewer than one prevalent fracture before starting denosumab, and stopped denosumab after a mean of 7.7 injections. Overall, 8% took glucocorticoids, and 22% took aromatase inhibitors.
The investigators collected data on what treatment was used after denosumab, how bone turnover markers changed 1-3 months after the last denosumab injection and then regularly afterward, how bone mineral density changed after 1 year, and any new osteoporotic fractures.
At the time of denosumab discontinuation, 59% received zoledronate, 24% alendronate, 3% other drugs, and 14% nothing. At a mean of about 17 months after the last denosumab injection, the investigators classified 30 patients as BMD losers (losing at least 3.96%), and 41 had stable BMD. The researchers found that BMD losers were younger (61.4 years vs. 65.5 years), were less likely to use zoledronate before starting denosumab (0% vs. 12%), and had greater serum CTX (C-telopeptide cross-linked type 1 collagen) levels at denosumab initiation (644 ng/mL vs. 474 ng/mL) and 12.8 months after stopping denosumab (592 ng/mL vs. 336 ng/mL) than did those with stable BMD. All differences were statistically significant.
“Our results support the use of denosumab in second line after bisphosphonate therapy to restrain the BMD loss at its discontinuation ... and a strategy to maintain the bone turnover marker serum CTX as low as possible after denosumab discontinuation,” she concluded.
“Our proposition is to start with 1 or 2 years of bisphosphonates, and if the osteoporosis is severe, to switch to denosumab treatment for 4, 6 years. … We can use denosumab for 10 years without side effects, and after that we give bisphosphonates to consolidate the treatment,” she said.
Dr. Rozier Aubry and her associates plan to follow patients in their study for 2 years.
Dr. Rozier Aubry disclosed serving on speakers bureaus for Eli Lilly, Pfizer, Amgen, and Novartis.
SOURCE: Rozier Aubry B et al. Ann Rheum Dis. Jun 2019;78(Suppl 2):115; Abstract OP0085. doi: 10.1136/annrheumdis-2019-eular.4175.
REPORTING FROM EULAR 2019 CONGRESS
Levothyroxine did not reduce fatigue in older patients with hypothyroidism
WASHINGTON – according to a new study.
The double-blind, randomized, placebo-controlled, parallel-group trial used subsets of data from the TRUST study. This new research, which was presented at the Annual Meeting of the Society of General Internal Medicine, provides further evidence that prescribing levothyroxine to older patients with subclinical hypothyroidism will not reduce fatigue.
Like the findings in the TRUST study, the new study, “Effect of Thyroid Hormone Replacement on Fatigability in Older Adults with Subclinical Hypothyroidism: A Randomized Placebo Controlled Trial,” showed that the use of levothyroxine in subclinical hypothyroidism was not effective in changing physical and mental fatigability in older adults.
“This study shows that levothyroxine treatment may not be necessary and results in high costs and side effects,” noted Mirah Stuber, MD, during her presentation of the findings at the meeting.
The new study examined the effect of levothyroxine on the tiredness of older adults but with a more detailed analysis of fatigue than the full TRUST study provided. In this new study, investigators used a new assessment tool, called the Pittsburgh Fatigability Scale (PFS), a 10-item self-administered questionnaire that can measure both physical and mental fatigability. Perceived fatigability anchors tiredness to a set of activities and has been shown to be a more sensitive measure than global fatigue, explained Dr. Stuber of the University of Chicago.
The scores range from 0-50, with higher scores indicating higher fatigability. The scales were divided between mental and physical activities and measured fatigue in relation to a defined activity of a specific duration and intensity. These activities included a leisurely 30-minute walk and participation in a 1-hour social activity.
This study involved 230 participants from Switzerland and Ireland, with a mean age of 73.4 years, who had persistent hypothyroidism. The population was randomized to 119 patients who were administered levothyroxine and 111 patients who received a placebo.
At baseline, the levothyroxine group had a mean physical PFS score of 14.7 ± 9.3, and the placebo group had a score of 11.1 ± 9.1. The baseline mean mental PFS score for the levothyroxine group was 7.4 ± 8.0, while it was 5.1 ± 6.9 for the placebo group.
After 12 months of the participants’ use of levothyroxine or placebo, the physical PFS scores increased for both the treatment and placebo groups. For the levothyroxine group, the mean physical PFS score was 14.8 ± 9.6, while it was 12.4 ± 9.3 for the placebo group (P = 0.88). The investigators found no significant differences between these scores for the levothyroxine and placebo groups.
The mean mental PFS score slightly decreased to 6.0 ± 7.8 for the levothyroxine group, while it slightly increased to 6.0 ± 8.0 for the placebo group (P = 0.26) at 12 months. The difference between the mental fatigability scores for the levothyroxine and placebo arms at 12 months was also not significant.
The physical fatigability between-group difference was 0.2 (95% confidence interval, –1.8 to 2.1; P = 0.88), while the mental fatigability difference was –1.0 (95% CI, –2.8 to 0.8; P = 0.26).
The study was funded by the European Union FP7 and the Swiss National Science Foundation. Merck KGaA, Darmstadt (Germany) provided levothyroxine and the placebo. Dr. Stuber has no conflicts of interest.
WASHINGTON – according to a new study.
The double-blind, randomized, placebo-controlled, parallel-group trial used subsets of data from the TRUST study. This new research, which was presented at the Annual Meeting of the Society of General Internal Medicine, provides further evidence that prescribing levothyroxine to older patients with subclinical hypothyroidism will not reduce fatigue.
Like the findings in the TRUST study, the new study, “Effect of Thyroid Hormone Replacement on Fatigability in Older Adults with Subclinical Hypothyroidism: A Randomized Placebo Controlled Trial,” showed that the use of levothyroxine in subclinical hypothyroidism was not effective in changing physical and mental fatigability in older adults.
“This study shows that levothyroxine treatment may not be necessary and results in high costs and side effects,” noted Mirah Stuber, MD, during her presentation of the findings at the meeting.
The new study examined the effect of levothyroxine on the tiredness of older adults but with a more detailed analysis of fatigue than the full TRUST study provided. In this new study, investigators used a new assessment tool, called the Pittsburgh Fatigability Scale (PFS), a 10-item self-administered questionnaire that can measure both physical and mental fatigability. Perceived fatigability anchors tiredness to a set of activities and has been shown to be a more sensitive measure than global fatigue, explained Dr. Stuber of the University of Chicago.
The scores range from 0-50, with higher scores indicating higher fatigability. The scales were divided between mental and physical activities and measured fatigue in relation to a defined activity of a specific duration and intensity. These activities included a leisurely 30-minute walk and participation in a 1-hour social activity.
This study involved 230 participants from Switzerland and Ireland, with a mean age of 73.4 years, who had persistent hypothyroidism. The population was randomized to 119 patients who were administered levothyroxine and 111 patients who received a placebo.
At baseline, the levothyroxine group had a mean physical PFS score of 14.7 ± 9.3, and the placebo group had a score of 11.1 ± 9.1. The baseline mean mental PFS score for the levothyroxine group was 7.4 ± 8.0, while it was 5.1 ± 6.9 for the placebo group.
After 12 months of the participants’ use of levothyroxine or placebo, the physical PFS scores increased for both the treatment and placebo groups. For the levothyroxine group, the mean physical PFS score was 14.8 ± 9.6, while it was 12.4 ± 9.3 for the placebo group (P = 0.88). The investigators found no significant differences between these scores for the levothyroxine and placebo groups.
The mean mental PFS score slightly decreased to 6.0 ± 7.8 for the levothyroxine group, while it slightly increased to 6.0 ± 8.0 for the placebo group (P = 0.26) at 12 months. The difference between the mental fatigability scores for the levothyroxine and placebo arms at 12 months was also not significant.
The physical fatigability between-group difference was 0.2 (95% confidence interval, –1.8 to 2.1; P = 0.88), while the mental fatigability difference was –1.0 (95% CI, –2.8 to 0.8; P = 0.26).
The study was funded by the European Union FP7 and the Swiss National Science Foundation. Merck KGaA, Darmstadt (Germany) provided levothyroxine and the placebo. Dr. Stuber has no conflicts of interest.
WASHINGTON – according to a new study.
The double-blind, randomized, placebo-controlled, parallel-group trial used subsets of data from the TRUST study. This new research, which was presented at the Annual Meeting of the Society of General Internal Medicine, provides further evidence that prescribing levothyroxine to older patients with subclinical hypothyroidism will not reduce fatigue.
Like the findings in the TRUST study, the new study, “Effect of Thyroid Hormone Replacement on Fatigability in Older Adults with Subclinical Hypothyroidism: A Randomized Placebo Controlled Trial,” showed that the use of levothyroxine in subclinical hypothyroidism was not effective in changing physical and mental fatigability in older adults.
“This study shows that levothyroxine treatment may not be necessary and results in high costs and side effects,” noted Mirah Stuber, MD, during her presentation of the findings at the meeting.
The new study examined the effect of levothyroxine on the tiredness of older adults but with a more detailed analysis of fatigue than the full TRUST study provided. In this new study, investigators used a new assessment tool, called the Pittsburgh Fatigability Scale (PFS), a 10-item self-administered questionnaire that can measure both physical and mental fatigability. Perceived fatigability anchors tiredness to a set of activities and has been shown to be a more sensitive measure than global fatigue, explained Dr. Stuber of the University of Chicago.
The scores range from 0-50, with higher scores indicating higher fatigability. The scales were divided between mental and physical activities and measured fatigue in relation to a defined activity of a specific duration and intensity. These activities included a leisurely 30-minute walk and participation in a 1-hour social activity.
This study involved 230 participants from Switzerland and Ireland, with a mean age of 73.4 years, who had persistent hypothyroidism. The population was randomized to 119 patients who were administered levothyroxine and 111 patients who received a placebo.
At baseline, the levothyroxine group had a mean physical PFS score of 14.7 ± 9.3, and the placebo group had a score of 11.1 ± 9.1. The baseline mean mental PFS score for the levothyroxine group was 7.4 ± 8.0, while it was 5.1 ± 6.9 for the placebo group.
After 12 months of the participants’ use of levothyroxine or placebo, the physical PFS scores increased for both the treatment and placebo groups. For the levothyroxine group, the mean physical PFS score was 14.8 ± 9.6, while it was 12.4 ± 9.3 for the placebo group (P = 0.88). The investigators found no significant differences between these scores for the levothyroxine and placebo groups.
The mean mental PFS score slightly decreased to 6.0 ± 7.8 for the levothyroxine group, while it slightly increased to 6.0 ± 8.0 for the placebo group (P = 0.26) at 12 months. The difference between the mental fatigability scores for the levothyroxine and placebo arms at 12 months was also not significant.
The physical fatigability between-group difference was 0.2 (95% confidence interval, –1.8 to 2.1; P = 0.88), while the mental fatigability difference was –1.0 (95% CI, –2.8 to 0.8; P = 0.26).
The study was funded by the European Union FP7 and the Swiss National Science Foundation. Merck KGaA, Darmstadt (Germany) provided levothyroxine and the placebo. Dr. Stuber has no conflicts of interest.
REPORTING FROM SGIM 2019
CGMs on the rise: New goals set time in range
SAN FRANCISCO – Most patients with diabetes should aim to spend at least 17 hours a day – more than 70% of their time – in a blood glucose range of 70-180 mg/dL, according to
Time spent below 70 mg/dL should be less than 1 hour a day, under 4% of the time in other words, and time spent below 54 mg/dL – the cut point for potentially serious hypoglycemia – less than 15 minutes (1%). Time spent at or above 180 mg/dL should be less than 6 hours (25%) a day, and above 250 mg/dL – the cut point for potentially serious hyperglycemia – less than 1 hour and 15 minutes (5%).
The advice comes from an international panel of diabetologists, researchers, and patients convened at the Advanced Technologies and Treatments for Diabetes Congress in Berlin earlier this year.
The goal was to give clinicians and patients handy treatment targets for continuous glucose monitors (CGMs), something that has been missing until now. The targets “should be considered an integral component of CGM data analysis and day-to-day treatment decision making,” said lead author Tadej Battelino, MD, PhD, head of the department of pediatric and adolescent endocrinology at Ljubljana (Slovenia) University, who presented the guidelines at the meeting.
The ADA, the European Association for the Study of Diabetes, and other leading diabetes groups have endorsed them.
CGMs have always offered the promise of tighter glycemic control, and their use is expanding, but they still have not led to a robust improvement in diabetes management, and in at least one study, they actually deteriorated control. There has been doubt about how to use them.
Dr. Battelino and associates thought that the main problem was a lack of clear, easy-to-understand treatment goals. The ADA and others previously recommended a CGM target of 70-180 mg/dL, but stopped short of saying how long people should be in that and other ranges. The new guidelines close the gap by adding the key element of duration.
“We ... pretty much defined what we believe is a safe way to live with diabetes,” Dr. Battelino said at the meeting. The work was based on literature review and expert opinion.
He and his colleagues noted in their journal write-up that for many the goals will be aspirational, but patients and doctors should not give up. The important thing is incremental change, with the hope of eventually meeting the targets. Even a small time-in-range increase reduces the risk of retinopathy and nephropathy, and improves hemoglobin A1c levels.
“You don’t have to get there all at once. Everyone needs to know that, whether they’re 14 or 44,” said coauthor Irl B. Hirsch, MD, chair of diabetes treatment and teaching at the University of Washington, Seattle, who moderated Dr. Battelino’s presentation.
To make the guidelines operational, the team created a simple, intuitive version of the ambulatory glucose profile they hope will be accepted as the new standard by CGM makers and included in device software. It reports the percentage of time in the 70-180 mg/dL range in green, the percentage below range in red, and the percentage above range in yellow. With a glance, both patients and doctors will know what is going on day by day, and what, if anything, needs to change.
The time-in-range bar was set at 50% for older and sicker patients, but their time-below-range goal was reduced from 4% to 1%, to emphasize the need to prevent hypoglycemia.
The target range was lowered for pregnant women to 63-140 mg/dL at least 70% of the time, because blood glucose levels are lower in pregnancy. However, “greater emphasis should be placed on getting to goal as soon as possible” with pregnant women and those planning to get pregnant, the panel said.
The work was funded by a number of companies, including Abbott, AstraZeneca, Dexcom, Eli Lilly, Medtronic, and Novo Nordisk. Dr. Battelino, Dr. Hirsch, and their coauthors reported various ties to those and other companies.
SOURCE: Battelino T et al. Diabetes Care. 2019 Jun 8. doi: 10.2337/dci19-0028.
SAN FRANCISCO – Most patients with diabetes should aim to spend at least 17 hours a day – more than 70% of their time – in a blood glucose range of 70-180 mg/dL, according to
Time spent below 70 mg/dL should be less than 1 hour a day, under 4% of the time in other words, and time spent below 54 mg/dL – the cut point for potentially serious hypoglycemia – less than 15 minutes (1%). Time spent at or above 180 mg/dL should be less than 6 hours (25%) a day, and above 250 mg/dL – the cut point for potentially serious hyperglycemia – less than 1 hour and 15 minutes (5%).
The advice comes from an international panel of diabetologists, researchers, and patients convened at the Advanced Technologies and Treatments for Diabetes Congress in Berlin earlier this year.
The goal was to give clinicians and patients handy treatment targets for continuous glucose monitors (CGMs), something that has been missing until now. The targets “should be considered an integral component of CGM data analysis and day-to-day treatment decision making,” said lead author Tadej Battelino, MD, PhD, head of the department of pediatric and adolescent endocrinology at Ljubljana (Slovenia) University, who presented the guidelines at the meeting.
The ADA, the European Association for the Study of Diabetes, and other leading diabetes groups have endorsed them.
CGMs have always offered the promise of tighter glycemic control, and their use is expanding, but they still have not led to a robust improvement in diabetes management, and in at least one study, they actually deteriorated control. There has been doubt about how to use them.
Dr. Battelino and associates thought that the main problem was a lack of clear, easy-to-understand treatment goals. The ADA and others previously recommended a CGM target of 70-180 mg/dL, but stopped short of saying how long people should be in that and other ranges. The new guidelines close the gap by adding the key element of duration.
“We ... pretty much defined what we believe is a safe way to live with diabetes,” Dr. Battelino said at the meeting. The work was based on literature review and expert opinion.
He and his colleagues noted in their journal write-up that for many the goals will be aspirational, but patients and doctors should not give up. The important thing is incremental change, with the hope of eventually meeting the targets. Even a small time-in-range increase reduces the risk of retinopathy and nephropathy, and improves hemoglobin A1c levels.
“You don’t have to get there all at once. Everyone needs to know that, whether they’re 14 or 44,” said coauthor Irl B. Hirsch, MD, chair of diabetes treatment and teaching at the University of Washington, Seattle, who moderated Dr. Battelino’s presentation.
To make the guidelines operational, the team created a simple, intuitive version of the ambulatory glucose profile they hope will be accepted as the new standard by CGM makers and included in device software. It reports the percentage of time in the 70-180 mg/dL range in green, the percentage below range in red, and the percentage above range in yellow. With a glance, both patients and doctors will know what is going on day by day, and what, if anything, needs to change.
The time-in-range bar was set at 50% for older and sicker patients, but their time-below-range goal was reduced from 4% to 1%, to emphasize the need to prevent hypoglycemia.
The target range was lowered for pregnant women to 63-140 mg/dL at least 70% of the time, because blood glucose levels are lower in pregnancy. However, “greater emphasis should be placed on getting to goal as soon as possible” with pregnant women and those planning to get pregnant, the panel said.
The work was funded by a number of companies, including Abbott, AstraZeneca, Dexcom, Eli Lilly, Medtronic, and Novo Nordisk. Dr. Battelino, Dr. Hirsch, and their coauthors reported various ties to those and other companies.
SOURCE: Battelino T et al. Diabetes Care. 2019 Jun 8. doi: 10.2337/dci19-0028.
SAN FRANCISCO – Most patients with diabetes should aim to spend at least 17 hours a day – more than 70% of their time – in a blood glucose range of 70-180 mg/dL, according to
Time spent below 70 mg/dL should be less than 1 hour a day, under 4% of the time in other words, and time spent below 54 mg/dL – the cut point for potentially serious hypoglycemia – less than 15 minutes (1%). Time spent at or above 180 mg/dL should be less than 6 hours (25%) a day, and above 250 mg/dL – the cut point for potentially serious hyperglycemia – less than 1 hour and 15 minutes (5%).
The advice comes from an international panel of diabetologists, researchers, and patients convened at the Advanced Technologies and Treatments for Diabetes Congress in Berlin earlier this year.
The goal was to give clinicians and patients handy treatment targets for continuous glucose monitors (CGMs), something that has been missing until now. The targets “should be considered an integral component of CGM data analysis and day-to-day treatment decision making,” said lead author Tadej Battelino, MD, PhD, head of the department of pediatric and adolescent endocrinology at Ljubljana (Slovenia) University, who presented the guidelines at the meeting.
The ADA, the European Association for the Study of Diabetes, and other leading diabetes groups have endorsed them.
CGMs have always offered the promise of tighter glycemic control, and their use is expanding, but they still have not led to a robust improvement in diabetes management, and in at least one study, they actually deteriorated control. There has been doubt about how to use them.
Dr. Battelino and associates thought that the main problem was a lack of clear, easy-to-understand treatment goals. The ADA and others previously recommended a CGM target of 70-180 mg/dL, but stopped short of saying how long people should be in that and other ranges. The new guidelines close the gap by adding the key element of duration.
“We ... pretty much defined what we believe is a safe way to live with diabetes,” Dr. Battelino said at the meeting. The work was based on literature review and expert opinion.
He and his colleagues noted in their journal write-up that for many the goals will be aspirational, but patients and doctors should not give up. The important thing is incremental change, with the hope of eventually meeting the targets. Even a small time-in-range increase reduces the risk of retinopathy and nephropathy, and improves hemoglobin A1c levels.
“You don’t have to get there all at once. Everyone needs to know that, whether they’re 14 or 44,” said coauthor Irl B. Hirsch, MD, chair of diabetes treatment and teaching at the University of Washington, Seattle, who moderated Dr. Battelino’s presentation.
To make the guidelines operational, the team created a simple, intuitive version of the ambulatory glucose profile they hope will be accepted as the new standard by CGM makers and included in device software. It reports the percentage of time in the 70-180 mg/dL range in green, the percentage below range in red, and the percentage above range in yellow. With a glance, both patients and doctors will know what is going on day by day, and what, if anything, needs to change.
The time-in-range bar was set at 50% for older and sicker patients, but their time-below-range goal was reduced from 4% to 1%, to emphasize the need to prevent hypoglycemia.
The target range was lowered for pregnant women to 63-140 mg/dL at least 70% of the time, because blood glucose levels are lower in pregnancy. However, “greater emphasis should be placed on getting to goal as soon as possible” with pregnant women and those planning to get pregnant, the panel said.
The work was funded by a number of companies, including Abbott, AstraZeneca, Dexcom, Eli Lilly, Medtronic, and Novo Nordisk. Dr. Battelino, Dr. Hirsch, and their coauthors reported various ties to those and other companies.
SOURCE: Battelino T et al. Diabetes Care. 2019 Jun 8. doi: 10.2337/dci19-0028.
REPORTING FROM ADA 2019
Screening for Endocrine Hypertension
Hypertension is one of the most common reasons for patient visits.1 According to the US Preventive Services Task Force, more than 70 million individuals older than 20 have hypertension, which is defined as a blood pressure (BP) of ≥ 130/85 mm Hg.2 Essential hypertension is the most common form of this condition; most affected patients will show improvement with evidence-based pharmacologic treatment, lifestyle modifications, and risk factor reductions.
For patients with refractory hypertension, however, identifying what steps to take in screening and diagnosis can be daunting for clinicians. It is important to identify cases of secondary hypertension, because if it is left undiagnosed and untreated, serious complications—such as cardiovascular and renal disease—are likely to occur.3,4
Secondary hypertension can be caused by myriad disease states and disorders, including endocrine disorders, renal disease, neurologic disorders, acute stress, and drug-induced hypertension.5 Endocrine hypertension is most commonly caused by adrenal gland disorders, including primary aldosteronism, Cushing syndrome, and pheochromocytoma. (Of note, Cushing syndrome is caused by glucocorticoid-secreting adrenal tumors, while Cushing disease is a condition in which there is glucocorticoid excess caused by oversecretion of pituitary adrenocorticotropic hormone.6 Cushing disease is more common than Cushing syndrome, which is rare.7) While nonadrenal endocrine disorders are not as common, they pose significant health issues, including growth hormone excess or deficiency, thyroid disorders, testosterone deficiency, obesity, insulin resistance, and metabolic syndrome.8
Understanding the endocrine causes of hypertension is a valuable resource for clinicians to have in their toolbox. Although the negative consequences of endocrine disorders are significant, these conditions are often recognizable, and pharmacologic treatment and/or surgical interventions can potentially resolve or improve hypertension and reduce risk for other comorbidities. This article summarizes screening and diagnosis guidelines for several possible causes of endocrine hypertension: primary aldosteronism, Cushing syndrome, and pheochromocytoma.
PRIMARY ALDOSTERONISM
Primary aldosteronism occurs in 5% to 10% of all hypertensive patients and is a common cause of secondary and endocrine hypertension (although in younger—particularly female—patients, it most commonly causes renal artery stenosis).9,10 Historically, primary aldosteronism was considered rare and not generally included in a differential diagnosis for patients presenting with resistant hypertension. However, clinical investigations have indicated that primary aldosteronism is more prevalent than previously thought.11
Patients develop this condition when there is increased aldosterone production independent of the renin-angiotensin system. The resulting sodium retention can lead to hypertension, hypokalemia, and high plasma aldosterone/renin ratio (ARR).12 Clinical findings and symptoms can be vague, increasing the difficulty in identifying primary aldosteronism as the diagnosis. Patients may be asymptomatic, with the only abnormal lab finding being hypokalemia (an infrequent finding, affecting < 25% of patients).13 If hypokalemia is present, symptoms can include nocturia, polyuria, muscle weakness, cramps, paresthesias, and palpitations.11
The Endocrine Society has identified 8 characteristics that increase the likelihood of primary aldosteronism. Patients require further screening if they
- Have a sustained elevated BP (≥ 150 mm Hg [systolic] and/or 100 mm Hg [diastolic])
- Have hypertension (BP > 140/90 mm Hg) that is resistant to 3 conventional antihypertensive drugs, including a diuretic
- Have controlled BP (BP < 140/90 mm Hg) with ≥ 4 antihypertensive drugs
- Have hypertension and spontaneous or diuretic-induced hypokalemia
- Have hypertension and adrenal incidentaloma
- Have hypertension and obstructive sleep apnea
- Have hypertension and a family history of early-onset hypertension or a cerebrovascular accident at a young age (< 40 years)
- Are hypertensive and a first-degree relative of a patient with primary aldosteronism.14
Continue to: The most reliable screening test...
The most reliable screening test for primary aldosterone is the ARR, although false-negative and false-positive results are possible.11 False-negative results can be caused by dietary salt restriction, hypokalemia, and use of medications including diuretics, calcium channel blockers, ACE inhibitors, and angiotensin receptor antagonists. Use of ß-adrenergic blockers, α-methyldopa, or NSAIDs can cause false-positive results.15 Patients should be encouraged to follow a liberal sodium diet before ARR testing, and efforts to correct hypokalemia should be implemented. Before ARR is measured, diuretics (specifically spironolactone) should be stopped for at least 4 weeks; other possible interfering medications should be stopped for at least 2 weeks.16
The ARR should be obtained multiple times to confirm elevated readings.16 Reference ranges vary, but generally plasma aldosterone concentrations > 20 ng/dL and plasma renin activity < 1 ng/mL/h indicate whether confirmatory testing should be completed.14 Further confirmatory testing can be achieved with efforts to suppress plasma aldosterone to < 10 ng/dL after an IV infusion of 2 L isotonic saline over 4 hours.12 Oral sodium load is used as well and usually before IV infusion.
CUSHING SYNDROME
Cushing syndrome is caused by excess circulating levels of glucocorticoids and affects < 0.1% of the world population.17 Signs and symptoms include centripetal obesity, moon facies, facial plethora, easy bruising, buffalo hump (or posterior cervical fat pad), hirsutism, and wide-purple striae.18 Up to 80% of these patients also have hypertension.19 If these patients have chronic exposure to high levels of glucocorticoid (the most common source being therapeutic administration of exogenous glucocorticoids), multiple complications can occur.6,20
The Endocrine Society Clinical Practice Guideline recommends the following patient groups be tested for Cushing syndrome:
- Young patients with unusual medical conditions, such as osteoporosis and resistant hypertension
- Patients with classic signs and symptoms, such as easy bruising, weight gain, facial plethora, and purple striae
- Children with decreasing height percentile and increasing weight
- Patients with adrenal incidentaloma compatible with adenoma.18
If Cushing syndrome is suspected, 1 of the following 3 initial tests can be completed: 24-hour, urine-free cortisol and creatinine; late-night salivary cortisol; or 1-mg overnight dexamethasone suppression test. Two of these tests must have abnormal results for confirmation before appropriate pituitary or adrenal imaging. If a patient has clinical features indicating Cushing syndrome but test results are normal, he or she should be referred to an endocrinologist. If a patient has ≥ 2 normal tests and probability of Cushing syndrome is unlikely, patients should be recommended for follow-up in 6 months to evaluate for any worsening of symptoms.18
Continue to: PHEOCHROMOCYTOMA
PHEOCHROMOCYTOMA
Pheochromocytoma is a condition in which there is secretion of excess catecholamines, epinephrine, norepinephrine, and dopamine due to a tumor of the adrenal medulla.21 This is a rare disease and accounts for only 0.2% to 0.6% of all causes of hypertension.22 Hypertension (persistent or paroxysmal) is the most common finding for patients with pheochromocytoma, with 80% to 90% presenting with this finding.23 It is important to note that approximately 10% of these patients will be normotensive. Three of the condition’s classic symptoms are headache, sweating, and palpitations.24 Additional symptoms include anxiety, sense of impending doom, fever, nausea, or vomiting.21
If left untreated, there is risk for hypertensive retinopathy, nephropathy, myocardial infarction, stroke from cerebral infarction, intracranial hemorrhage, or embolism.25 Due to the high rate of morbidity and mortality with untreated pheochromocytoma, laboratory testing should be initiated immediately upon suspicion of this diagnosis or if the patient has relevant family history.11
Patients should be screened for pheochromocytoma if they have ≥ 1 of the following factors:
- Resistant hypertension and hyperadrenergic symptoms (palpitations, perspiration, pallor, or headache)
- Family history of pheochromocytoma
- Any genetic syndrome with a known association to pheochromocytoma
- An adrenal mass that is > 4 cm, is cystic, or has hemorrhagic changes.19
Pheochromocytoma is diagnosed by identifying high concentrations of plasma-free metanephrines or 24-hour fractionated metanephrines and catecholamines. Some medications can interfere with the accuracy of lab results and therefore may need to be temporarily stopped; it is important to check the specific lab guidelines and review the patient’s medication lists before tests are ordered and conducted.25
ALWAYS SCREEN THE PATIENT
Although the causes of endocrine-related hypertension are very rare, screening for endocrine hypertension in patients who present with signs and symptoms of these conditions can greatly improve their lives. The endocrine disorders discussed in this article can be treated or controlled with appropriate diagnosis and treatment. In addition, resolving uncontrolled hypertension by addressing endocrine disorders can reduce the risk for long-term sequelae. It is important for clinicians to consider referral to an endocrine specialist if a patient has endocrine-related hypertension. In particular, patients with pheochromocytoma require quick referral due to a risk for high morbidity and mortality if left untreated.11
1. Smith MA, Schrager S, WinklerPrins V. Essentials of Family Medicine. 7th ed. Baltimore, MD: Lipincott Williams & Wilkins; 2019.
2. US Preventive Services Task Force. High blood pressure in adults: screening [final recommendation statement]. www.uspreventiveservicestaskforce.org/Page/Document/RecommendationStatementFinal/high-blood-pressure-in-adults-screening. Accessed May 20, 2019.
3. Puar T, Mok Y, Debajyoti R, et al. Secondary hypertension in adults. Singapore Med J. 2016;57:228-232.
4. Poulter NR, Prabhakaran D, Caulfield M. Hypertension. Lancet. 2015;386:801-812.
5. Faselis C, Doumas M, Papademetriou V. Common secondary causes of resistant hypertension and rational for treatment. Int J Hypertens. 2010;2011: doi: 10.4061/2011/236239.
6. Else T, Hammer GD. Disorders of the Adrenal Cortex. In: Hammer GD, McPhee SJ, eds. Pathophysiology of Disease: An Introduction to Clinical Medicine. 8th ed. New York, NY: McGraw-Hill; 2014.
7. Nieman L, Swearingen B; the Pituitary Society. Cushing’s syndrome and Cushing’s disease: your questions answered. www.pituitarysociety.org/sites/all/pdfs/Pituitary_Society_Cushings_brochure.pdf. Accessed May 20, 2019.
8. Koch, C. Chrousos, G. Overview of endocrine hypertension. In: Feingold KR, Anawalt B, Boyce A, et al, eds. Endotext. South Dartmouth, MA: MDText.com; 2016.
9. Barlow M, Abdel-Latif A. The forgotten cause of hypertension: a case report and literature review of the prevalence, diagnosis and management of primary aldosteronism. Case Rep Intern Med. 2018;5:4-7.
10. Viera A, Neutze D. Diagnosis of secondary hypertension: an age-based approach. Am Fam Physician. 2010;82:1471-1478.
11. Young WF, Calhoun DA, Lenders JWM, et al. Screening for endocrine hypertension: an Endocrine Society scientific statement. Endocr Rev. 2017;38:103-122.
12. Kotchen TA. Hypertensive vascular disease. In: Jameson JL, Fauci AS, Kasper DL, et al, eds. Harrison’s Principles of Internal Medicine. 20th ed. New York, NY: McGraw-Hill Education; 2018.
13. Rossi GP, Bernini G, Caliumi C, et al; PAPY Study Investigators. A prospective study of the prevalence of primary aldosteronism in 1,125 hypertensive patients. J Am Coll Cardiol. 2006;48:2293-2300.
14. Funder JW, Carey RM, Mantero F, et al. The management of primary aldosteronism: case detection, diagnosis, and treatment: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2016;101:1889-1916.
15. Stowasser M, Taylor PJ, Pimenta E, et al. Laboratory investigation of primary aldosteronism. Clin Biochem Rev. 2010;31:39-56.
16. Stowasser M, Gordon RD. The aldosterone-renin ratio for screening for primary aldosteronism. Endocrinologist. 2004;14:267-276.
17. Newell-Price J, Bertagna X, Grossman AB, et al. Cushing’s syndrome. Lancet. 2006;367:1605-1617.
18. Nieman LK, Biller BM, Findling JW, et al. The diagnosis of Cushing’s syndrome: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2008;93: 1526-1540.
19. Rimoldi S, Scherrer U, Messerli F. Secondary arterial hypertension: when, who, and how to screen? Eur Heart J. 2014;35:1245-1254.
20. Kirk L, Hash R, Harold K. Cushing’s syndrome and Cushing’s disease. Am Fam Physician. 2000;62:1133-1134.
21. Thomas RM, Ruel E, Shantavasinkul PC. Endocrine hypertension: an overview on the current etiopathogenesis and management options. World J Hypertens. 2015;5:14-27.
22. Ariton M, Juan CS, AvRuskin TW. Pheochromocytoma: clinical observations from a Brooklyn tertiary hospital. Endocr Pract. 2000;6:249-252.
23. Calhoun DA, Jones D, Textor S, et al. Resistant hypertension: diagnosis, evaluation, and treatment: a scientific statement from the American Heart Association Professional Education Committee of the Council for High Blood Pressure Research. Hypertension. 2008;51(6):1403-1419.
24. Lenders JW, Eisenhofer G, Mannelli M, et al. Pheochromocytoma. Lancet. 2005;366:665-675.
25. Fishbein L, Else T. Disorders of the adrenal medulla. In: Hammer GD, McPhee SJ, eds. Pathophysiology of Disease: An Introduction to Clinical Medicine. 8th ed. New York, NY: McGraw-Hill; 2014.
Hypertension is one of the most common reasons for patient visits.1 According to the US Preventive Services Task Force, more than 70 million individuals older than 20 have hypertension, which is defined as a blood pressure (BP) of ≥ 130/85 mm Hg.2 Essential hypertension is the most common form of this condition; most affected patients will show improvement with evidence-based pharmacologic treatment, lifestyle modifications, and risk factor reductions.
For patients with refractory hypertension, however, identifying what steps to take in screening and diagnosis can be daunting for clinicians. It is important to identify cases of secondary hypertension, because if it is left undiagnosed and untreated, serious complications—such as cardiovascular and renal disease—are likely to occur.3,4
Secondary hypertension can be caused by myriad disease states and disorders, including endocrine disorders, renal disease, neurologic disorders, acute stress, and drug-induced hypertension.5 Endocrine hypertension is most commonly caused by adrenal gland disorders, including primary aldosteronism, Cushing syndrome, and pheochromocytoma. (Of note, Cushing syndrome is caused by glucocorticoid-secreting adrenal tumors, while Cushing disease is a condition in which there is glucocorticoid excess caused by oversecretion of pituitary adrenocorticotropic hormone.6 Cushing disease is more common than Cushing syndrome, which is rare.7) While nonadrenal endocrine disorders are not as common, they pose significant health issues, including growth hormone excess or deficiency, thyroid disorders, testosterone deficiency, obesity, insulin resistance, and metabolic syndrome.8
Understanding the endocrine causes of hypertension is a valuable resource for clinicians to have in their toolbox. Although the negative consequences of endocrine disorders are significant, these conditions are often recognizable, and pharmacologic treatment and/or surgical interventions can potentially resolve or improve hypertension and reduce risk for other comorbidities. This article summarizes screening and diagnosis guidelines for several possible causes of endocrine hypertension: primary aldosteronism, Cushing syndrome, and pheochromocytoma.
PRIMARY ALDOSTERONISM
Primary aldosteronism occurs in 5% to 10% of all hypertensive patients and is a common cause of secondary and endocrine hypertension (although in younger—particularly female—patients, it most commonly causes renal artery stenosis).9,10 Historically, primary aldosteronism was considered rare and not generally included in a differential diagnosis for patients presenting with resistant hypertension. However, clinical investigations have indicated that primary aldosteronism is more prevalent than previously thought.11
Patients develop this condition when there is increased aldosterone production independent of the renin-angiotensin system. The resulting sodium retention can lead to hypertension, hypokalemia, and high plasma aldosterone/renin ratio (ARR).12 Clinical findings and symptoms can be vague, increasing the difficulty in identifying primary aldosteronism as the diagnosis. Patients may be asymptomatic, with the only abnormal lab finding being hypokalemia (an infrequent finding, affecting < 25% of patients).13 If hypokalemia is present, symptoms can include nocturia, polyuria, muscle weakness, cramps, paresthesias, and palpitations.11
The Endocrine Society has identified 8 characteristics that increase the likelihood of primary aldosteronism. Patients require further screening if they
- Have a sustained elevated BP (≥ 150 mm Hg [systolic] and/or 100 mm Hg [diastolic])
- Have hypertension (BP > 140/90 mm Hg) that is resistant to 3 conventional antihypertensive drugs, including a diuretic
- Have controlled BP (BP < 140/90 mm Hg) with ≥ 4 antihypertensive drugs
- Have hypertension and spontaneous or diuretic-induced hypokalemia
- Have hypertension and adrenal incidentaloma
- Have hypertension and obstructive sleep apnea
- Have hypertension and a family history of early-onset hypertension or a cerebrovascular accident at a young age (< 40 years)
- Are hypertensive and a first-degree relative of a patient with primary aldosteronism.14
Continue to: The most reliable screening test...
The most reliable screening test for primary aldosterone is the ARR, although false-negative and false-positive results are possible.11 False-negative results can be caused by dietary salt restriction, hypokalemia, and use of medications including diuretics, calcium channel blockers, ACE inhibitors, and angiotensin receptor antagonists. Use of ß-adrenergic blockers, α-methyldopa, or NSAIDs can cause false-positive results.15 Patients should be encouraged to follow a liberal sodium diet before ARR testing, and efforts to correct hypokalemia should be implemented. Before ARR is measured, diuretics (specifically spironolactone) should be stopped for at least 4 weeks; other possible interfering medications should be stopped for at least 2 weeks.16
The ARR should be obtained multiple times to confirm elevated readings.16 Reference ranges vary, but generally plasma aldosterone concentrations > 20 ng/dL and plasma renin activity < 1 ng/mL/h indicate whether confirmatory testing should be completed.14 Further confirmatory testing can be achieved with efforts to suppress plasma aldosterone to < 10 ng/dL after an IV infusion of 2 L isotonic saline over 4 hours.12 Oral sodium load is used as well and usually before IV infusion.
CUSHING SYNDROME
Cushing syndrome is caused by excess circulating levels of glucocorticoids and affects < 0.1% of the world population.17 Signs and symptoms include centripetal obesity, moon facies, facial plethora, easy bruising, buffalo hump (or posterior cervical fat pad), hirsutism, and wide-purple striae.18 Up to 80% of these patients also have hypertension.19 If these patients have chronic exposure to high levels of glucocorticoid (the most common source being therapeutic administration of exogenous glucocorticoids), multiple complications can occur.6,20
The Endocrine Society Clinical Practice Guideline recommends the following patient groups be tested for Cushing syndrome:
- Young patients with unusual medical conditions, such as osteoporosis and resistant hypertension
- Patients with classic signs and symptoms, such as easy bruising, weight gain, facial plethora, and purple striae
- Children with decreasing height percentile and increasing weight
- Patients with adrenal incidentaloma compatible with adenoma.18
If Cushing syndrome is suspected, 1 of the following 3 initial tests can be completed: 24-hour, urine-free cortisol and creatinine; late-night salivary cortisol; or 1-mg overnight dexamethasone suppression test. Two of these tests must have abnormal results for confirmation before appropriate pituitary or adrenal imaging. If a patient has clinical features indicating Cushing syndrome but test results are normal, he or she should be referred to an endocrinologist. If a patient has ≥ 2 normal tests and probability of Cushing syndrome is unlikely, patients should be recommended for follow-up in 6 months to evaluate for any worsening of symptoms.18
Continue to: PHEOCHROMOCYTOMA
PHEOCHROMOCYTOMA
Pheochromocytoma is a condition in which there is secretion of excess catecholamines, epinephrine, norepinephrine, and dopamine due to a tumor of the adrenal medulla.21 This is a rare disease and accounts for only 0.2% to 0.6% of all causes of hypertension.22 Hypertension (persistent or paroxysmal) is the most common finding for patients with pheochromocytoma, with 80% to 90% presenting with this finding.23 It is important to note that approximately 10% of these patients will be normotensive. Three of the condition’s classic symptoms are headache, sweating, and palpitations.24 Additional symptoms include anxiety, sense of impending doom, fever, nausea, or vomiting.21
If left untreated, there is risk for hypertensive retinopathy, nephropathy, myocardial infarction, stroke from cerebral infarction, intracranial hemorrhage, or embolism.25 Due to the high rate of morbidity and mortality with untreated pheochromocytoma, laboratory testing should be initiated immediately upon suspicion of this diagnosis or if the patient has relevant family history.11
Patients should be screened for pheochromocytoma if they have ≥ 1 of the following factors:
- Resistant hypertension and hyperadrenergic symptoms (palpitations, perspiration, pallor, or headache)
- Family history of pheochromocytoma
- Any genetic syndrome with a known association to pheochromocytoma
- An adrenal mass that is > 4 cm, is cystic, or has hemorrhagic changes.19
Pheochromocytoma is diagnosed by identifying high concentrations of plasma-free metanephrines or 24-hour fractionated metanephrines and catecholamines. Some medications can interfere with the accuracy of lab results and therefore may need to be temporarily stopped; it is important to check the specific lab guidelines and review the patient’s medication lists before tests are ordered and conducted.25
ALWAYS SCREEN THE PATIENT
Although the causes of endocrine-related hypertension are very rare, screening for endocrine hypertension in patients who present with signs and symptoms of these conditions can greatly improve their lives. The endocrine disorders discussed in this article can be treated or controlled with appropriate diagnosis and treatment. In addition, resolving uncontrolled hypertension by addressing endocrine disorders can reduce the risk for long-term sequelae. It is important for clinicians to consider referral to an endocrine specialist if a patient has endocrine-related hypertension. In particular, patients with pheochromocytoma require quick referral due to a risk for high morbidity and mortality if left untreated.11
Hypertension is one of the most common reasons for patient visits.1 According to the US Preventive Services Task Force, more than 70 million individuals older than 20 have hypertension, which is defined as a blood pressure (BP) of ≥ 130/85 mm Hg.2 Essential hypertension is the most common form of this condition; most affected patients will show improvement with evidence-based pharmacologic treatment, lifestyle modifications, and risk factor reductions.
For patients with refractory hypertension, however, identifying what steps to take in screening and diagnosis can be daunting for clinicians. It is important to identify cases of secondary hypertension, because if it is left undiagnosed and untreated, serious complications—such as cardiovascular and renal disease—are likely to occur.3,4
Secondary hypertension can be caused by myriad disease states and disorders, including endocrine disorders, renal disease, neurologic disorders, acute stress, and drug-induced hypertension.5 Endocrine hypertension is most commonly caused by adrenal gland disorders, including primary aldosteronism, Cushing syndrome, and pheochromocytoma. (Of note, Cushing syndrome is caused by glucocorticoid-secreting adrenal tumors, while Cushing disease is a condition in which there is glucocorticoid excess caused by oversecretion of pituitary adrenocorticotropic hormone.6 Cushing disease is more common than Cushing syndrome, which is rare.7) While nonadrenal endocrine disorders are not as common, they pose significant health issues, including growth hormone excess or deficiency, thyroid disorders, testosterone deficiency, obesity, insulin resistance, and metabolic syndrome.8
Understanding the endocrine causes of hypertension is a valuable resource for clinicians to have in their toolbox. Although the negative consequences of endocrine disorders are significant, these conditions are often recognizable, and pharmacologic treatment and/or surgical interventions can potentially resolve or improve hypertension and reduce risk for other comorbidities. This article summarizes screening and diagnosis guidelines for several possible causes of endocrine hypertension: primary aldosteronism, Cushing syndrome, and pheochromocytoma.
PRIMARY ALDOSTERONISM
Primary aldosteronism occurs in 5% to 10% of all hypertensive patients and is a common cause of secondary and endocrine hypertension (although in younger—particularly female—patients, it most commonly causes renal artery stenosis).9,10 Historically, primary aldosteronism was considered rare and not generally included in a differential diagnosis for patients presenting with resistant hypertension. However, clinical investigations have indicated that primary aldosteronism is more prevalent than previously thought.11
Patients develop this condition when there is increased aldosterone production independent of the renin-angiotensin system. The resulting sodium retention can lead to hypertension, hypokalemia, and high plasma aldosterone/renin ratio (ARR).12 Clinical findings and symptoms can be vague, increasing the difficulty in identifying primary aldosteronism as the diagnosis. Patients may be asymptomatic, with the only abnormal lab finding being hypokalemia (an infrequent finding, affecting < 25% of patients).13 If hypokalemia is present, symptoms can include nocturia, polyuria, muscle weakness, cramps, paresthesias, and palpitations.11
The Endocrine Society has identified 8 characteristics that increase the likelihood of primary aldosteronism. Patients require further screening if they
- Have a sustained elevated BP (≥ 150 mm Hg [systolic] and/or 100 mm Hg [diastolic])
- Have hypertension (BP > 140/90 mm Hg) that is resistant to 3 conventional antihypertensive drugs, including a diuretic
- Have controlled BP (BP < 140/90 mm Hg) with ≥ 4 antihypertensive drugs
- Have hypertension and spontaneous or diuretic-induced hypokalemia
- Have hypertension and adrenal incidentaloma
- Have hypertension and obstructive sleep apnea
- Have hypertension and a family history of early-onset hypertension or a cerebrovascular accident at a young age (< 40 years)
- Are hypertensive and a first-degree relative of a patient with primary aldosteronism.14
Continue to: The most reliable screening test...
The most reliable screening test for primary aldosterone is the ARR, although false-negative and false-positive results are possible.11 False-negative results can be caused by dietary salt restriction, hypokalemia, and use of medications including diuretics, calcium channel blockers, ACE inhibitors, and angiotensin receptor antagonists. Use of ß-adrenergic blockers, α-methyldopa, or NSAIDs can cause false-positive results.15 Patients should be encouraged to follow a liberal sodium diet before ARR testing, and efforts to correct hypokalemia should be implemented. Before ARR is measured, diuretics (specifically spironolactone) should be stopped for at least 4 weeks; other possible interfering medications should be stopped for at least 2 weeks.16
The ARR should be obtained multiple times to confirm elevated readings.16 Reference ranges vary, but generally plasma aldosterone concentrations > 20 ng/dL and plasma renin activity < 1 ng/mL/h indicate whether confirmatory testing should be completed.14 Further confirmatory testing can be achieved with efforts to suppress plasma aldosterone to < 10 ng/dL after an IV infusion of 2 L isotonic saline over 4 hours.12 Oral sodium load is used as well and usually before IV infusion.
CUSHING SYNDROME
Cushing syndrome is caused by excess circulating levels of glucocorticoids and affects < 0.1% of the world population.17 Signs and symptoms include centripetal obesity, moon facies, facial plethora, easy bruising, buffalo hump (or posterior cervical fat pad), hirsutism, and wide-purple striae.18 Up to 80% of these patients also have hypertension.19 If these patients have chronic exposure to high levels of glucocorticoid (the most common source being therapeutic administration of exogenous glucocorticoids), multiple complications can occur.6,20
The Endocrine Society Clinical Practice Guideline recommends the following patient groups be tested for Cushing syndrome:
- Young patients with unusual medical conditions, such as osteoporosis and resistant hypertension
- Patients with classic signs and symptoms, such as easy bruising, weight gain, facial plethora, and purple striae
- Children with decreasing height percentile and increasing weight
- Patients with adrenal incidentaloma compatible with adenoma.18
If Cushing syndrome is suspected, 1 of the following 3 initial tests can be completed: 24-hour, urine-free cortisol and creatinine; late-night salivary cortisol; or 1-mg overnight dexamethasone suppression test. Two of these tests must have abnormal results for confirmation before appropriate pituitary or adrenal imaging. If a patient has clinical features indicating Cushing syndrome but test results are normal, he or she should be referred to an endocrinologist. If a patient has ≥ 2 normal tests and probability of Cushing syndrome is unlikely, patients should be recommended for follow-up in 6 months to evaluate for any worsening of symptoms.18
Continue to: PHEOCHROMOCYTOMA
PHEOCHROMOCYTOMA
Pheochromocytoma is a condition in which there is secretion of excess catecholamines, epinephrine, norepinephrine, and dopamine due to a tumor of the adrenal medulla.21 This is a rare disease and accounts for only 0.2% to 0.6% of all causes of hypertension.22 Hypertension (persistent or paroxysmal) is the most common finding for patients with pheochromocytoma, with 80% to 90% presenting with this finding.23 It is important to note that approximately 10% of these patients will be normotensive. Three of the condition’s classic symptoms are headache, sweating, and palpitations.24 Additional symptoms include anxiety, sense of impending doom, fever, nausea, or vomiting.21
If left untreated, there is risk for hypertensive retinopathy, nephropathy, myocardial infarction, stroke from cerebral infarction, intracranial hemorrhage, or embolism.25 Due to the high rate of morbidity and mortality with untreated pheochromocytoma, laboratory testing should be initiated immediately upon suspicion of this diagnosis or if the patient has relevant family history.11
Patients should be screened for pheochromocytoma if they have ≥ 1 of the following factors:
- Resistant hypertension and hyperadrenergic symptoms (palpitations, perspiration, pallor, or headache)
- Family history of pheochromocytoma
- Any genetic syndrome with a known association to pheochromocytoma
- An adrenal mass that is > 4 cm, is cystic, or has hemorrhagic changes.19
Pheochromocytoma is diagnosed by identifying high concentrations of plasma-free metanephrines or 24-hour fractionated metanephrines and catecholamines. Some medications can interfere with the accuracy of lab results and therefore may need to be temporarily stopped; it is important to check the specific lab guidelines and review the patient’s medication lists before tests are ordered and conducted.25
ALWAYS SCREEN THE PATIENT
Although the causes of endocrine-related hypertension are very rare, screening for endocrine hypertension in patients who present with signs and symptoms of these conditions can greatly improve their lives. The endocrine disorders discussed in this article can be treated or controlled with appropriate diagnosis and treatment. In addition, resolving uncontrolled hypertension by addressing endocrine disorders can reduce the risk for long-term sequelae. It is important for clinicians to consider referral to an endocrine specialist if a patient has endocrine-related hypertension. In particular, patients with pheochromocytoma require quick referral due to a risk for high morbidity and mortality if left untreated.11
1. Smith MA, Schrager S, WinklerPrins V. Essentials of Family Medicine. 7th ed. Baltimore, MD: Lipincott Williams & Wilkins; 2019.
2. US Preventive Services Task Force. High blood pressure in adults: screening [final recommendation statement]. www.uspreventiveservicestaskforce.org/Page/Document/RecommendationStatementFinal/high-blood-pressure-in-adults-screening. Accessed May 20, 2019.
3. Puar T, Mok Y, Debajyoti R, et al. Secondary hypertension in adults. Singapore Med J. 2016;57:228-232.
4. Poulter NR, Prabhakaran D, Caulfield M. Hypertension. Lancet. 2015;386:801-812.
5. Faselis C, Doumas M, Papademetriou V. Common secondary causes of resistant hypertension and rational for treatment. Int J Hypertens. 2010;2011: doi: 10.4061/2011/236239.
6. Else T, Hammer GD. Disorders of the Adrenal Cortex. In: Hammer GD, McPhee SJ, eds. Pathophysiology of Disease: An Introduction to Clinical Medicine. 8th ed. New York, NY: McGraw-Hill; 2014.
7. Nieman L, Swearingen B; the Pituitary Society. Cushing’s syndrome and Cushing’s disease: your questions answered. www.pituitarysociety.org/sites/all/pdfs/Pituitary_Society_Cushings_brochure.pdf. Accessed May 20, 2019.
8. Koch, C. Chrousos, G. Overview of endocrine hypertension. In: Feingold KR, Anawalt B, Boyce A, et al, eds. Endotext. South Dartmouth, MA: MDText.com; 2016.
9. Barlow M, Abdel-Latif A. The forgotten cause of hypertension: a case report and literature review of the prevalence, diagnosis and management of primary aldosteronism. Case Rep Intern Med. 2018;5:4-7.
10. Viera A, Neutze D. Diagnosis of secondary hypertension: an age-based approach. Am Fam Physician. 2010;82:1471-1478.
11. Young WF, Calhoun DA, Lenders JWM, et al. Screening for endocrine hypertension: an Endocrine Society scientific statement. Endocr Rev. 2017;38:103-122.
12. Kotchen TA. Hypertensive vascular disease. In: Jameson JL, Fauci AS, Kasper DL, et al, eds. Harrison’s Principles of Internal Medicine. 20th ed. New York, NY: McGraw-Hill Education; 2018.
13. Rossi GP, Bernini G, Caliumi C, et al; PAPY Study Investigators. A prospective study of the prevalence of primary aldosteronism in 1,125 hypertensive patients. J Am Coll Cardiol. 2006;48:2293-2300.
14. Funder JW, Carey RM, Mantero F, et al. The management of primary aldosteronism: case detection, diagnosis, and treatment: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2016;101:1889-1916.
15. Stowasser M, Taylor PJ, Pimenta E, et al. Laboratory investigation of primary aldosteronism. Clin Biochem Rev. 2010;31:39-56.
16. Stowasser M, Gordon RD. The aldosterone-renin ratio for screening for primary aldosteronism. Endocrinologist. 2004;14:267-276.
17. Newell-Price J, Bertagna X, Grossman AB, et al. Cushing’s syndrome. Lancet. 2006;367:1605-1617.
18. Nieman LK, Biller BM, Findling JW, et al. The diagnosis of Cushing’s syndrome: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2008;93: 1526-1540.
19. Rimoldi S, Scherrer U, Messerli F. Secondary arterial hypertension: when, who, and how to screen? Eur Heart J. 2014;35:1245-1254.
20. Kirk L, Hash R, Harold K. Cushing’s syndrome and Cushing’s disease. Am Fam Physician. 2000;62:1133-1134.
21. Thomas RM, Ruel E, Shantavasinkul PC. Endocrine hypertension: an overview on the current etiopathogenesis and management options. World J Hypertens. 2015;5:14-27.
22. Ariton M, Juan CS, AvRuskin TW. Pheochromocytoma: clinical observations from a Brooklyn tertiary hospital. Endocr Pract. 2000;6:249-252.
23. Calhoun DA, Jones D, Textor S, et al. Resistant hypertension: diagnosis, evaluation, and treatment: a scientific statement from the American Heart Association Professional Education Committee of the Council for High Blood Pressure Research. Hypertension. 2008;51(6):1403-1419.
24. Lenders JW, Eisenhofer G, Mannelli M, et al. Pheochromocytoma. Lancet. 2005;366:665-675.
25. Fishbein L, Else T. Disorders of the adrenal medulla. In: Hammer GD, McPhee SJ, eds. Pathophysiology of Disease: An Introduction to Clinical Medicine. 8th ed. New York, NY: McGraw-Hill; 2014.
1. Smith MA, Schrager S, WinklerPrins V. Essentials of Family Medicine. 7th ed. Baltimore, MD: Lipincott Williams & Wilkins; 2019.
2. US Preventive Services Task Force. High blood pressure in adults: screening [final recommendation statement]. www.uspreventiveservicestaskforce.org/Page/Document/RecommendationStatementFinal/high-blood-pressure-in-adults-screening. Accessed May 20, 2019.
3. Puar T, Mok Y, Debajyoti R, et al. Secondary hypertension in adults. Singapore Med J. 2016;57:228-232.
4. Poulter NR, Prabhakaran D, Caulfield M. Hypertension. Lancet. 2015;386:801-812.
5. Faselis C, Doumas M, Papademetriou V. Common secondary causes of resistant hypertension and rational for treatment. Int J Hypertens. 2010;2011: doi: 10.4061/2011/236239.
6. Else T, Hammer GD. Disorders of the Adrenal Cortex. In: Hammer GD, McPhee SJ, eds. Pathophysiology of Disease: An Introduction to Clinical Medicine. 8th ed. New York, NY: McGraw-Hill; 2014.
7. Nieman L, Swearingen B; the Pituitary Society. Cushing’s syndrome and Cushing’s disease: your questions answered. www.pituitarysociety.org/sites/all/pdfs/Pituitary_Society_Cushings_brochure.pdf. Accessed May 20, 2019.
8. Koch, C. Chrousos, G. Overview of endocrine hypertension. In: Feingold KR, Anawalt B, Boyce A, et al, eds. Endotext. South Dartmouth, MA: MDText.com; 2016.
9. Barlow M, Abdel-Latif A. The forgotten cause of hypertension: a case report and literature review of the prevalence, diagnosis and management of primary aldosteronism. Case Rep Intern Med. 2018;5:4-7.
10. Viera A, Neutze D. Diagnosis of secondary hypertension: an age-based approach. Am Fam Physician. 2010;82:1471-1478.
11. Young WF, Calhoun DA, Lenders JWM, et al. Screening for endocrine hypertension: an Endocrine Society scientific statement. Endocr Rev. 2017;38:103-122.
12. Kotchen TA. Hypertensive vascular disease. In: Jameson JL, Fauci AS, Kasper DL, et al, eds. Harrison’s Principles of Internal Medicine. 20th ed. New York, NY: McGraw-Hill Education; 2018.
13. Rossi GP, Bernini G, Caliumi C, et al; PAPY Study Investigators. A prospective study of the prevalence of primary aldosteronism in 1,125 hypertensive patients. J Am Coll Cardiol. 2006;48:2293-2300.
14. Funder JW, Carey RM, Mantero F, et al. The management of primary aldosteronism: case detection, diagnosis, and treatment: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2016;101:1889-1916.
15. Stowasser M, Taylor PJ, Pimenta E, et al. Laboratory investigation of primary aldosteronism. Clin Biochem Rev. 2010;31:39-56.
16. Stowasser M, Gordon RD. The aldosterone-renin ratio for screening for primary aldosteronism. Endocrinologist. 2004;14:267-276.
17. Newell-Price J, Bertagna X, Grossman AB, et al. Cushing’s syndrome. Lancet. 2006;367:1605-1617.
18. Nieman LK, Biller BM, Findling JW, et al. The diagnosis of Cushing’s syndrome: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2008;93: 1526-1540.
19. Rimoldi S, Scherrer U, Messerli F. Secondary arterial hypertension: when, who, and how to screen? Eur Heart J. 2014;35:1245-1254.
20. Kirk L, Hash R, Harold K. Cushing’s syndrome and Cushing’s disease. Am Fam Physician. 2000;62:1133-1134.
21. Thomas RM, Ruel E, Shantavasinkul PC. Endocrine hypertension: an overview on the current etiopathogenesis and management options. World J Hypertens. 2015;5:14-27.
22. Ariton M, Juan CS, AvRuskin TW. Pheochromocytoma: clinical observations from a Brooklyn tertiary hospital. Endocr Pract. 2000;6:249-252.
23. Calhoun DA, Jones D, Textor S, et al. Resistant hypertension: diagnosis, evaluation, and treatment: a scientific statement from the American Heart Association Professional Education Committee of the Council for High Blood Pressure Research. Hypertension. 2008;51(6):1403-1419.
24. Lenders JW, Eisenhofer G, Mannelli M, et al. Pheochromocytoma. Lancet. 2005;366:665-675.
25. Fishbein L, Else T. Disorders of the adrenal medulla. In: Hammer GD, McPhee SJ, eds. Pathophysiology of Disease: An Introduction to Clinical Medicine. 8th ed. New York, NY: McGraw-Hill; 2014.