User login
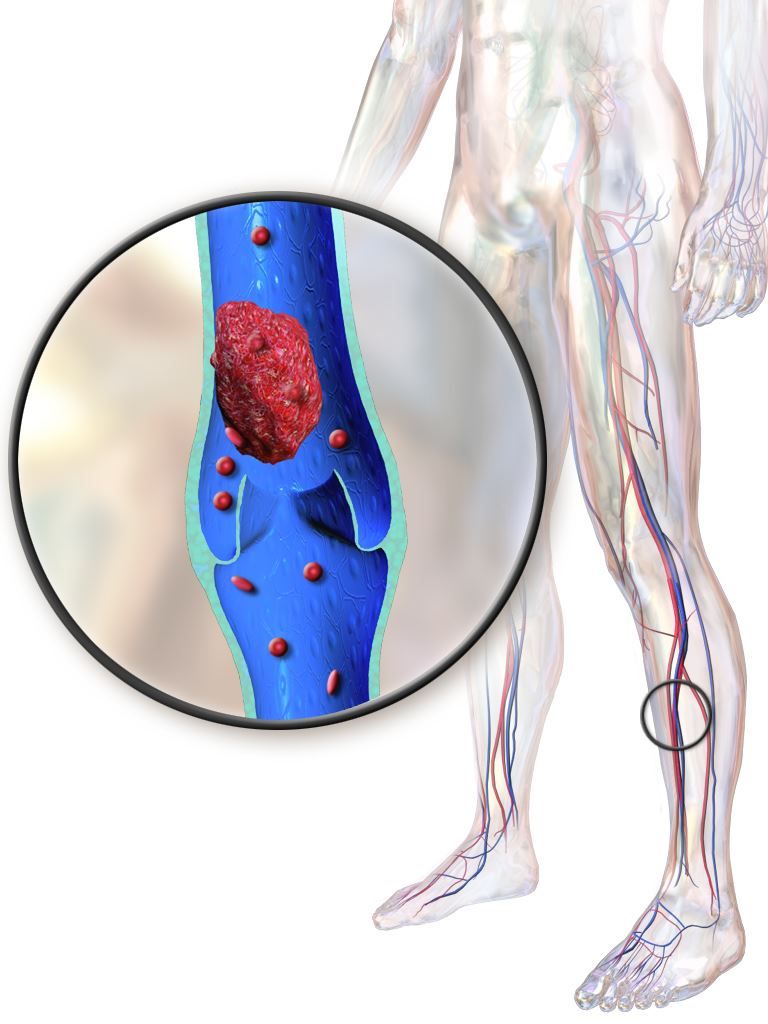
How does alcohol intake affect dementia risk in older adults?
Mild cognitive impairment (MCI) may influence the relationship between alcohol consumption and dementia risk, a study of more than 3,000 adults suggests. In addition, , according to the study, which was published in JAMA Network Open.
“The associations of self-reported alcohol consumption with dementia risk and cognitive decline were more consistently adverse among individuals with MCI than those with normal cognition,” reported Manja Koch, PhD, a researcher in the department of nutrition at Harvard T.H. Chan School of Public Health in Boston and colleagues. “This was particularly true for the subset of individuals [with MCI] who drank more than 14.0 servings per week, whose rate of cognitive decline and risk of dementia were the highest of any subgroup.”
Among older adults with normal cognition, the results generally were consistent with those of a recent meta-analysis that found a U-shaped relationship between drinking and dementia, the researchers said (Eur J Epidemiol. 2017 Jan;32[1]:31-42.).
“Our results did not show significant associations and clearly do not suffice to suggest a clinical benefit from even limited alcohol use,” said Dr. Koch and colleagues. “Nonetheless, our findings provide some reassurance that alcohol consumed within recommended limits was not associated with an increased risk of dementia among older adults with normal baseline cognition.”
GEMS data
To study whether alcohol consumption is associated with the risk of dementia and cognitive decline in older adults with and without MCI, the investigators analyzed data from the Ginkgo Evaluation of Memory Study (GEMS). GEMS was a randomized controlled trial conducted between 2000 and 2008 that found no overall association between ginkgo biloba and dementia prevention. During the trial, participants completed the Modified Mini-Mental State Examination, the Clinical Dementia Rating scale, and the cognitive portion of the Alzheimer’s Disease Assessment Scale.
In the present study, the investigators analyzed data from 3,021 participants aged 72 years and older who were free of dementia at baseline and had provided information about their alcohol intake. Their median age was 78 years, and 46.2% were female. Fifty-eight percent consumed alcohol, including 45% of the participants with MCI at baseline.
During follow-up, 512 cases of dementia occurred. Among the 473 participants with MCI at baseline, the adjusted hazard ratio (HR) for dementia was 1.72 for those who consumed more than 14 drinks per week, compared with light drinkers who consumed less than 1 drink per week. For participants who consumed between 7 and 14 drinks per week, the adjusted HR for dementia was 0.63 among those without MCI and 0.93 among those with MCI, relative to light drinkers who consumed less than 1 drink per week.
Among adults with normal cognition at baseline, daily low-quantity drinking was associated with lower dementia risk, compared with infrequent higher-quantity drinking (HR, 0.45).
Trial excluded adults with excessive alcohol use
Limitations of the study include a lack of data about any changes in alcohol consumption over time. In addition, the original trial excluded people with a known history of excessive alcohol use. Furthermore, it is possible that the “long preclinical phase of dementia” and other health issues affect drinking behavior, the authors said. “At present, our findings cannot be directly translated into clinical recommendations,” the authors said. Nevertheless, the results “suggest that, while caring for older adults, physicians should carefully assess the full dimensions of drinking behavior and cognition when providing guidance to patients about alcohol consumption,” they said.
The study was supported by grants from the National Center for Complementary and Alternative Medicine; the National Institute of Neurological Disorders and Stroke; the Office of Dietary Supplements of the National Institute on Aging; the National Heart, Lung, and Blood Institute; the University of Pittsburgh Alzheimer’s Disease Research Center; the Roena Kulynych Center for Memory and Cognition Research; and Wake Forest University School of Medicine. In addition, the researchers used plasma samples from the National Cell Repository for Alzheimer’s Disease, which receives support from the National Institute on Aging. Dr. Koch had no conflicts of interest. Coauthors disclosed university and government grants and personal fees from pharmaceutical companies outside the study. One author was an employee of Genentech at the time of publication, but Genentech did not contribute to the study.
SOURCE: Koch M et al. JAMA Network Open. 2019 Sep 27. doi: 10.1001/jamanetworkopen.2019.10319.
Mild cognitive impairment (MCI) may influence the relationship between alcohol consumption and dementia risk, a study of more than 3,000 adults suggests. In addition, , according to the study, which was published in JAMA Network Open.
“The associations of self-reported alcohol consumption with dementia risk and cognitive decline were more consistently adverse among individuals with MCI than those with normal cognition,” reported Manja Koch, PhD, a researcher in the department of nutrition at Harvard T.H. Chan School of Public Health in Boston and colleagues. “This was particularly true for the subset of individuals [with MCI] who drank more than 14.0 servings per week, whose rate of cognitive decline and risk of dementia were the highest of any subgroup.”
Among older adults with normal cognition, the results generally were consistent with those of a recent meta-analysis that found a U-shaped relationship between drinking and dementia, the researchers said (Eur J Epidemiol. 2017 Jan;32[1]:31-42.).
“Our results did not show significant associations and clearly do not suffice to suggest a clinical benefit from even limited alcohol use,” said Dr. Koch and colleagues. “Nonetheless, our findings provide some reassurance that alcohol consumed within recommended limits was not associated with an increased risk of dementia among older adults with normal baseline cognition.”
GEMS data
To study whether alcohol consumption is associated with the risk of dementia and cognitive decline in older adults with and without MCI, the investigators analyzed data from the Ginkgo Evaluation of Memory Study (GEMS). GEMS was a randomized controlled trial conducted between 2000 and 2008 that found no overall association between ginkgo biloba and dementia prevention. During the trial, participants completed the Modified Mini-Mental State Examination, the Clinical Dementia Rating scale, and the cognitive portion of the Alzheimer’s Disease Assessment Scale.
In the present study, the investigators analyzed data from 3,021 participants aged 72 years and older who were free of dementia at baseline and had provided information about their alcohol intake. Their median age was 78 years, and 46.2% were female. Fifty-eight percent consumed alcohol, including 45% of the participants with MCI at baseline.
During follow-up, 512 cases of dementia occurred. Among the 473 participants with MCI at baseline, the adjusted hazard ratio (HR) for dementia was 1.72 for those who consumed more than 14 drinks per week, compared with light drinkers who consumed less than 1 drink per week. For participants who consumed between 7 and 14 drinks per week, the adjusted HR for dementia was 0.63 among those without MCI and 0.93 among those with MCI, relative to light drinkers who consumed less than 1 drink per week.
Among adults with normal cognition at baseline, daily low-quantity drinking was associated with lower dementia risk, compared with infrequent higher-quantity drinking (HR, 0.45).
Trial excluded adults with excessive alcohol use
Limitations of the study include a lack of data about any changes in alcohol consumption over time. In addition, the original trial excluded people with a known history of excessive alcohol use. Furthermore, it is possible that the “long preclinical phase of dementia” and other health issues affect drinking behavior, the authors said. “At present, our findings cannot be directly translated into clinical recommendations,” the authors said. Nevertheless, the results “suggest that, while caring for older adults, physicians should carefully assess the full dimensions of drinking behavior and cognition when providing guidance to patients about alcohol consumption,” they said.
The study was supported by grants from the National Center for Complementary and Alternative Medicine; the National Institute of Neurological Disorders and Stroke; the Office of Dietary Supplements of the National Institute on Aging; the National Heart, Lung, and Blood Institute; the University of Pittsburgh Alzheimer’s Disease Research Center; the Roena Kulynych Center for Memory and Cognition Research; and Wake Forest University School of Medicine. In addition, the researchers used plasma samples from the National Cell Repository for Alzheimer’s Disease, which receives support from the National Institute on Aging. Dr. Koch had no conflicts of interest. Coauthors disclosed university and government grants and personal fees from pharmaceutical companies outside the study. One author was an employee of Genentech at the time of publication, but Genentech did not contribute to the study.
SOURCE: Koch M et al. JAMA Network Open. 2019 Sep 27. doi: 10.1001/jamanetworkopen.2019.10319.
Mild cognitive impairment (MCI) may influence the relationship between alcohol consumption and dementia risk, a study of more than 3,000 adults suggests. In addition, , according to the study, which was published in JAMA Network Open.
“The associations of self-reported alcohol consumption with dementia risk and cognitive decline were more consistently adverse among individuals with MCI than those with normal cognition,” reported Manja Koch, PhD, a researcher in the department of nutrition at Harvard T.H. Chan School of Public Health in Boston and colleagues. “This was particularly true for the subset of individuals [with MCI] who drank more than 14.0 servings per week, whose rate of cognitive decline and risk of dementia were the highest of any subgroup.”
Among older adults with normal cognition, the results generally were consistent with those of a recent meta-analysis that found a U-shaped relationship between drinking and dementia, the researchers said (Eur J Epidemiol. 2017 Jan;32[1]:31-42.).
“Our results did not show significant associations and clearly do not suffice to suggest a clinical benefit from even limited alcohol use,” said Dr. Koch and colleagues. “Nonetheless, our findings provide some reassurance that alcohol consumed within recommended limits was not associated with an increased risk of dementia among older adults with normal baseline cognition.”
GEMS data
To study whether alcohol consumption is associated with the risk of dementia and cognitive decline in older adults with and without MCI, the investigators analyzed data from the Ginkgo Evaluation of Memory Study (GEMS). GEMS was a randomized controlled trial conducted between 2000 and 2008 that found no overall association between ginkgo biloba and dementia prevention. During the trial, participants completed the Modified Mini-Mental State Examination, the Clinical Dementia Rating scale, and the cognitive portion of the Alzheimer’s Disease Assessment Scale.
In the present study, the investigators analyzed data from 3,021 participants aged 72 years and older who were free of dementia at baseline and had provided information about their alcohol intake. Their median age was 78 years, and 46.2% were female. Fifty-eight percent consumed alcohol, including 45% of the participants with MCI at baseline.
During follow-up, 512 cases of dementia occurred. Among the 473 participants with MCI at baseline, the adjusted hazard ratio (HR) for dementia was 1.72 for those who consumed more than 14 drinks per week, compared with light drinkers who consumed less than 1 drink per week. For participants who consumed between 7 and 14 drinks per week, the adjusted HR for dementia was 0.63 among those without MCI and 0.93 among those with MCI, relative to light drinkers who consumed less than 1 drink per week.
Among adults with normal cognition at baseline, daily low-quantity drinking was associated with lower dementia risk, compared with infrequent higher-quantity drinking (HR, 0.45).
Trial excluded adults with excessive alcohol use
Limitations of the study include a lack of data about any changes in alcohol consumption over time. In addition, the original trial excluded people with a known history of excessive alcohol use. Furthermore, it is possible that the “long preclinical phase of dementia” and other health issues affect drinking behavior, the authors said. “At present, our findings cannot be directly translated into clinical recommendations,” the authors said. Nevertheless, the results “suggest that, while caring for older adults, physicians should carefully assess the full dimensions of drinking behavior and cognition when providing guidance to patients about alcohol consumption,” they said.
The study was supported by grants from the National Center for Complementary and Alternative Medicine; the National Institute of Neurological Disorders and Stroke; the Office of Dietary Supplements of the National Institute on Aging; the National Heart, Lung, and Blood Institute; the University of Pittsburgh Alzheimer’s Disease Research Center; the Roena Kulynych Center for Memory and Cognition Research; and Wake Forest University School of Medicine. In addition, the researchers used plasma samples from the National Cell Repository for Alzheimer’s Disease, which receives support from the National Institute on Aging. Dr. Koch had no conflicts of interest. Coauthors disclosed university and government grants and personal fees from pharmaceutical companies outside the study. One author was an employee of Genentech at the time of publication, but Genentech did not contribute to the study.
SOURCE: Koch M et al. JAMA Network Open. 2019 Sep 27. doi: 10.1001/jamanetworkopen.2019.10319.
FROM JAMA NETWORK OPEN
Lumbar spine BMD, bone strength benefits persist after romosozumab-to-alendronate switch
ORLANDO – Patients who took romosozumab for 12 months and then switched to alendronate continued to see benefits in bone mineral density (BMD) of the lumbar spine after 12 months of therapy with alendronate, compared with patients who began taking, and continued to take, alendronate over the same time period, according to findings from a subgroup of the ARCH study presented at the annual meeting of the American Society for Bone and Mineral Research.
“These effects occurred rapidly, as early as month 6, were sustained beyond 12 months after transitioning to alendronate, and are consistent with greater fracture-risk reduction observed in ARCH with romosozumab to alendronate versus alendronate to alendronate,” Jacques P. Brown, MD, FRCPC, of Laval University, Quebec City, said in his presentation.
In the double-blinded ARCH study, 4,093 postmenopausal women with osteoporosis and a previous fracture history were randomized to receive subcutaneous monthly romosozumab 210 mg or oral weekly alendronate 70 mg for 12 months, followed by an open-label period during which romosozumab patients received oral weekly alendronate 70 mg and alendronate patients continued to receive the same dose on the same schedule for an additional 24 months (Saag KG et al. N Eng J Med. 2017 Oct 12. doi: 10.1056/NEJMoa1708322).
Dr. Brown and colleagues performed an imaging substudy of ARCH, which included examining how the romosozumab-to-alendronate and alendronate-only groups improved lumbar spine BMD and lumbar spine bone strength. Lumbar spine BMD was assessed through quantitative CT, and lumbar spine bone strength was measured with finite element analysis. The researchers received quantitative CT images from baseline and at 6 months, 12 months, and 24 months, and determined the percentage change at each of those periods to calculate integral, trabecular, and cortical lumbar spine volumetric BMD (vBMD), and to bone mineral content (BMC). They also measured areal BMD (aBMD) at baseline, 6 months, 12 months, 18 months, and 24 months with dual-energy x-ray absorptiometry.
Overall, 49 romosozumab patients and 41 alendronate patients from the ARCH study were enrolled in the imaging substudy. Of those patients, 76 had vBMD and BMC information available at baseline and one or more time periods post baseline, and 86 patients had finite element analysis data at baseline and one or more postbaseline time periods. Patients in the romosozumab and alendronate groups had similar baseline characteristics with regard to age (73.1 years vs. 72.8 years, respectively), mean lumbar spine BMD T score (–2.82 vs. –3.38), mean total hip BMD T score (–2.65 vs. –2.75), mean femoral neck T score (–2.84 vs. –2.83), mean lumbar spine integral vBMD (130.3 mg/cm3 vs. 120.5 mg/cm3), trabecular vBMD (60.1 mg/cm3 vs. 53.7 mg/cm3) and cortical vBMD (284.6 mg/cm3 vs. 270.9 mg/cm3). Patients in both groups also had similar rates of previous osteoporotic fracture at or after aged 45 years, previous vertebral fracture, and history of hip fracture.
Beginning at 6 months, there were significant least squares mean BMD improvements in both groups, but the romosozumab group had significant improvements in aBMD percentage changes, compared with the alendronate group, which persisted until 24 months (P less than .001 at all time points). Integral, trabecular, and cortical vBMD in the romosozumab group also saw significantly greater increases from baseline, compared with the alendronate group, and those results persisted in the open-label portion of the study for patients in the romosozumab group who transitioned to alendronate and patients in the alendronate to alendronate group (P less than .001 at all time points).
“The rapid and large increases in BMD with romosozumab followed by BMD consolidation where [patients were] transitioning to alendronate, support the important role of romosozumab as a first-line therapy in treating patients who are at very high risk for fracture,” Dr. Brown said.
In regard to BMC, there were larger increases in least squares mean BMC changes from baseline in the cortical compartment than the trabecular compartment, and actual change in bone strength as measured by finite element analysis was highly correlated with integral BMC in the romosozumab group.
Dr. Brown said the study was limited to the small sample size from the imaging substudy of ARCH, and quantitative CT dictated the imaging sites for the substudy, which may have affected patient selection. However, he noted that the characteristics of the ARCH imaging substudy were similar to patients in the overall ARCH study.
Amgen, UCB Pharma, and Astellas Pharma funded the study in part. Amgen and UCB Pharma assisted in the preparation of Dr. Brown’s presentation at ASBMR 2019, including funding costs associated with its development. Dr. Brown and the other coauthors reported relationships with Amgen, UCB Pharma, and other companies in the form of consultancies, grants and research support, speaker’s bureau appointments, paid employment, and stock options.
SOURCE: Brown JP et al. ASBMR 2019, Abstract 1050.
ORLANDO – Patients who took romosozumab for 12 months and then switched to alendronate continued to see benefits in bone mineral density (BMD) of the lumbar spine after 12 months of therapy with alendronate, compared with patients who began taking, and continued to take, alendronate over the same time period, according to findings from a subgroup of the ARCH study presented at the annual meeting of the American Society for Bone and Mineral Research.
“These effects occurred rapidly, as early as month 6, were sustained beyond 12 months after transitioning to alendronate, and are consistent with greater fracture-risk reduction observed in ARCH with romosozumab to alendronate versus alendronate to alendronate,” Jacques P. Brown, MD, FRCPC, of Laval University, Quebec City, said in his presentation.
In the double-blinded ARCH study, 4,093 postmenopausal women with osteoporosis and a previous fracture history were randomized to receive subcutaneous monthly romosozumab 210 mg or oral weekly alendronate 70 mg for 12 months, followed by an open-label period during which romosozumab patients received oral weekly alendronate 70 mg and alendronate patients continued to receive the same dose on the same schedule for an additional 24 months (Saag KG et al. N Eng J Med. 2017 Oct 12. doi: 10.1056/NEJMoa1708322).
Dr. Brown and colleagues performed an imaging substudy of ARCH, which included examining how the romosozumab-to-alendronate and alendronate-only groups improved lumbar spine BMD and lumbar spine bone strength. Lumbar spine BMD was assessed through quantitative CT, and lumbar spine bone strength was measured with finite element analysis. The researchers received quantitative CT images from baseline and at 6 months, 12 months, and 24 months, and determined the percentage change at each of those periods to calculate integral, trabecular, and cortical lumbar spine volumetric BMD (vBMD), and to bone mineral content (BMC). They also measured areal BMD (aBMD) at baseline, 6 months, 12 months, 18 months, and 24 months with dual-energy x-ray absorptiometry.
Overall, 49 romosozumab patients and 41 alendronate patients from the ARCH study were enrolled in the imaging substudy. Of those patients, 76 had vBMD and BMC information available at baseline and one or more time periods post baseline, and 86 patients had finite element analysis data at baseline and one or more postbaseline time periods. Patients in the romosozumab and alendronate groups had similar baseline characteristics with regard to age (73.1 years vs. 72.8 years, respectively), mean lumbar spine BMD T score (–2.82 vs. –3.38), mean total hip BMD T score (–2.65 vs. –2.75), mean femoral neck T score (–2.84 vs. –2.83), mean lumbar spine integral vBMD (130.3 mg/cm3 vs. 120.5 mg/cm3), trabecular vBMD (60.1 mg/cm3 vs. 53.7 mg/cm3) and cortical vBMD (284.6 mg/cm3 vs. 270.9 mg/cm3). Patients in both groups also had similar rates of previous osteoporotic fracture at or after aged 45 years, previous vertebral fracture, and history of hip fracture.
Beginning at 6 months, there were significant least squares mean BMD improvements in both groups, but the romosozumab group had significant improvements in aBMD percentage changes, compared with the alendronate group, which persisted until 24 months (P less than .001 at all time points). Integral, trabecular, and cortical vBMD in the romosozumab group also saw significantly greater increases from baseline, compared with the alendronate group, and those results persisted in the open-label portion of the study for patients in the romosozumab group who transitioned to alendronate and patients in the alendronate to alendronate group (P less than .001 at all time points).
“The rapid and large increases in BMD with romosozumab followed by BMD consolidation where [patients were] transitioning to alendronate, support the important role of romosozumab as a first-line therapy in treating patients who are at very high risk for fracture,” Dr. Brown said.
In regard to BMC, there were larger increases in least squares mean BMC changes from baseline in the cortical compartment than the trabecular compartment, and actual change in bone strength as measured by finite element analysis was highly correlated with integral BMC in the romosozumab group.
Dr. Brown said the study was limited to the small sample size from the imaging substudy of ARCH, and quantitative CT dictated the imaging sites for the substudy, which may have affected patient selection. However, he noted that the characteristics of the ARCH imaging substudy were similar to patients in the overall ARCH study.
Amgen, UCB Pharma, and Astellas Pharma funded the study in part. Amgen and UCB Pharma assisted in the preparation of Dr. Brown’s presentation at ASBMR 2019, including funding costs associated with its development. Dr. Brown and the other coauthors reported relationships with Amgen, UCB Pharma, and other companies in the form of consultancies, grants and research support, speaker’s bureau appointments, paid employment, and stock options.
SOURCE: Brown JP et al. ASBMR 2019, Abstract 1050.
ORLANDO – Patients who took romosozumab for 12 months and then switched to alendronate continued to see benefits in bone mineral density (BMD) of the lumbar spine after 12 months of therapy with alendronate, compared with patients who began taking, and continued to take, alendronate over the same time period, according to findings from a subgroup of the ARCH study presented at the annual meeting of the American Society for Bone and Mineral Research.
“These effects occurred rapidly, as early as month 6, were sustained beyond 12 months after transitioning to alendronate, and are consistent with greater fracture-risk reduction observed in ARCH with romosozumab to alendronate versus alendronate to alendronate,” Jacques P. Brown, MD, FRCPC, of Laval University, Quebec City, said in his presentation.
In the double-blinded ARCH study, 4,093 postmenopausal women with osteoporosis and a previous fracture history were randomized to receive subcutaneous monthly romosozumab 210 mg or oral weekly alendronate 70 mg for 12 months, followed by an open-label period during which romosozumab patients received oral weekly alendronate 70 mg and alendronate patients continued to receive the same dose on the same schedule for an additional 24 months (Saag KG et al. N Eng J Med. 2017 Oct 12. doi: 10.1056/NEJMoa1708322).
Dr. Brown and colleagues performed an imaging substudy of ARCH, which included examining how the romosozumab-to-alendronate and alendronate-only groups improved lumbar spine BMD and lumbar spine bone strength. Lumbar spine BMD was assessed through quantitative CT, and lumbar spine bone strength was measured with finite element analysis. The researchers received quantitative CT images from baseline and at 6 months, 12 months, and 24 months, and determined the percentage change at each of those periods to calculate integral, trabecular, and cortical lumbar spine volumetric BMD (vBMD), and to bone mineral content (BMC). They also measured areal BMD (aBMD) at baseline, 6 months, 12 months, 18 months, and 24 months with dual-energy x-ray absorptiometry.
Overall, 49 romosozumab patients and 41 alendronate patients from the ARCH study were enrolled in the imaging substudy. Of those patients, 76 had vBMD and BMC information available at baseline and one or more time periods post baseline, and 86 patients had finite element analysis data at baseline and one or more postbaseline time periods. Patients in the romosozumab and alendronate groups had similar baseline characteristics with regard to age (73.1 years vs. 72.8 years, respectively), mean lumbar spine BMD T score (–2.82 vs. –3.38), mean total hip BMD T score (–2.65 vs. –2.75), mean femoral neck T score (–2.84 vs. –2.83), mean lumbar spine integral vBMD (130.3 mg/cm3 vs. 120.5 mg/cm3), trabecular vBMD (60.1 mg/cm3 vs. 53.7 mg/cm3) and cortical vBMD (284.6 mg/cm3 vs. 270.9 mg/cm3). Patients in both groups also had similar rates of previous osteoporotic fracture at or after aged 45 years, previous vertebral fracture, and history of hip fracture.
Beginning at 6 months, there were significant least squares mean BMD improvements in both groups, but the romosozumab group had significant improvements in aBMD percentage changes, compared with the alendronate group, which persisted until 24 months (P less than .001 at all time points). Integral, trabecular, and cortical vBMD in the romosozumab group also saw significantly greater increases from baseline, compared with the alendronate group, and those results persisted in the open-label portion of the study for patients in the romosozumab group who transitioned to alendronate and patients in the alendronate to alendronate group (P less than .001 at all time points).
“The rapid and large increases in BMD with romosozumab followed by BMD consolidation where [patients were] transitioning to alendronate, support the important role of romosozumab as a first-line therapy in treating patients who are at very high risk for fracture,” Dr. Brown said.
In regard to BMC, there were larger increases in least squares mean BMC changes from baseline in the cortical compartment than the trabecular compartment, and actual change in bone strength as measured by finite element analysis was highly correlated with integral BMC in the romosozumab group.
Dr. Brown said the study was limited to the small sample size from the imaging substudy of ARCH, and quantitative CT dictated the imaging sites for the substudy, which may have affected patient selection. However, he noted that the characteristics of the ARCH imaging substudy were similar to patients in the overall ARCH study.
Amgen, UCB Pharma, and Astellas Pharma funded the study in part. Amgen and UCB Pharma assisted in the preparation of Dr. Brown’s presentation at ASBMR 2019, including funding costs associated with its development. Dr. Brown and the other coauthors reported relationships with Amgen, UCB Pharma, and other companies in the form of consultancies, grants and research support, speaker’s bureau appointments, paid employment, and stock options.
SOURCE: Brown JP et al. ASBMR 2019, Abstract 1050.
REPORTING FROM ASBMR 2019
Vitamin D does not improve bone density, structure in healthy patients
ORLANDO – after 2 years of daily use, according to data presented at the annual meeting of the American Society for Bone and Mineral Research.
“Participants may have already reached the vitamin D level needed for bone health,” Meryl S. LeBoff, MD, of Brigham and Women’s Hospital in Boston, said in her presentation.
Dr. LeBoff presented results from 771 patients (mean age, 63.8 years) in the Bone Health Subcohort of VITAL (Vitamin D and OmegA-3 TriaL) who were not on any bone active medications and were randomized to receive daily vitamin D3 at a dose of 2,000 IU or placebo. Patients received bone imaging at baseline and at 2 years; areal bone mineral density (aBMD) of the whole body, femoral neck, total hip, and spine was assessed via dual x-ray absorptiometry scan. Total 25-hydroxyvitamin D (25[OH]D) levels were measured via liquid chromatography tandem mass spectrometry, and free 25(OH)D levels were measured via the ELISA assay. The baseline characteristics of the vitamin D3 supplementation and placebo groups were similar. Overall, 52% of patients had osteopenia and 10.4% had osteoporosis.
Between baseline and 2 years, the vitamin D group’s total 25(OH)D levels increased from a mean 27.0 ng/mL to 39.5 ng/mL (46%) and the free 25(OH)D levels increased from 5.8 pg/mL to 9.0 pg/mL (55%), whereas levels in the placebo stayed the same. The researchers found no significant absolute percentage changes over 2 years in aBMD of the whole body (P = .60), femoral neck (P = .16), total hip (P = .23) and spine (P = .55), compared with patients in the placebo group.
In a secondary analysis, Dr. LeBoff and colleagues found no benefit to volumetric BMD (vBMD) of the radius and the tibia at 2 years, and the results persisted after they performed a sensitivity analysis. Adverse events, such as hypercalciuria, kidney stones, and gastrointestinal symptoms, were not significantly different in the vitamin D group, compared with the placebo group.
Dr. LeBoff noted among the limitations of the study that it evaluated one dose level of vitamin D and was not designed to determine whether vitamin D supplementation was effective in people with vitamin D insufficiency, and the results are not generalizable to patients with osteoporosis or osteomalacia. Future studies should also examine whether free 25(OH)D levels can be used to detect which patients can benefit from vitamin D supplementation, she added.
Risk of falls
In a separate abstract, which Dr. LeBoff presented in a different session, 12,927 patients who received vitamin D supplementation for 5 years, were studied for risk of falls, compared with 12,994 individuals in a placebo group. At baseline, 33.3% of patients had fallen at least once in the previous year, and overall 6,605 patients reported 13,235 falls. At 5.3 years of follow-up, there were no significant differences in number of falls between groups, falls leading to injury, and falls leading to a doctor or a hospital visit.
There are ongoing parallel studies examining the incidence of fractures between groups in the total population of the VITAL study (25,871 participants); bone turnover markers; bone microarchitecture measurements through high-resolution peripheral quantitative computed tomography; and examining the connection between free 25(OH)D, parathyroid hormone, and vitamin D binding protein, said Dr. LeBoff.
The study was funded in part by grants from the National Cancer Institute, the National Heart, Lung and Blood Institute, the Office of Dietary Supplements, the National Institute of Neurological Disorders and Stroke, and the National Center for Complementary and Integrative Health. Dr. LeBoff reported receiving grants from the National Institute of Arthritis Musculoskeletal and Skin Diseases. Two authors reported nonfinancial support Pharmavite LLC of Northridge, Calif., Pronova BioPharma of Norway and BASF, and Quest Diagnostics. The remaining authors reported no conflicts of interest.
SOURCE: LeBoff M et al. ASBMR 2019, Abstracts 1046 and 1057.
ORLANDO – after 2 years of daily use, according to data presented at the annual meeting of the American Society for Bone and Mineral Research.
“Participants may have already reached the vitamin D level needed for bone health,” Meryl S. LeBoff, MD, of Brigham and Women’s Hospital in Boston, said in her presentation.
Dr. LeBoff presented results from 771 patients (mean age, 63.8 years) in the Bone Health Subcohort of VITAL (Vitamin D and OmegA-3 TriaL) who were not on any bone active medications and were randomized to receive daily vitamin D3 at a dose of 2,000 IU or placebo. Patients received bone imaging at baseline and at 2 years; areal bone mineral density (aBMD) of the whole body, femoral neck, total hip, and spine was assessed via dual x-ray absorptiometry scan. Total 25-hydroxyvitamin D (25[OH]D) levels were measured via liquid chromatography tandem mass spectrometry, and free 25(OH)D levels were measured via the ELISA assay. The baseline characteristics of the vitamin D3 supplementation and placebo groups were similar. Overall, 52% of patients had osteopenia and 10.4% had osteoporosis.
Between baseline and 2 years, the vitamin D group’s total 25(OH)D levels increased from a mean 27.0 ng/mL to 39.5 ng/mL (46%) and the free 25(OH)D levels increased from 5.8 pg/mL to 9.0 pg/mL (55%), whereas levels in the placebo stayed the same. The researchers found no significant absolute percentage changes over 2 years in aBMD of the whole body (P = .60), femoral neck (P = .16), total hip (P = .23) and spine (P = .55), compared with patients in the placebo group.
In a secondary analysis, Dr. LeBoff and colleagues found no benefit to volumetric BMD (vBMD) of the radius and the tibia at 2 years, and the results persisted after they performed a sensitivity analysis. Adverse events, such as hypercalciuria, kidney stones, and gastrointestinal symptoms, were not significantly different in the vitamin D group, compared with the placebo group.
Dr. LeBoff noted among the limitations of the study that it evaluated one dose level of vitamin D and was not designed to determine whether vitamin D supplementation was effective in people with vitamin D insufficiency, and the results are not generalizable to patients with osteoporosis or osteomalacia. Future studies should also examine whether free 25(OH)D levels can be used to detect which patients can benefit from vitamin D supplementation, she added.
Risk of falls
In a separate abstract, which Dr. LeBoff presented in a different session, 12,927 patients who received vitamin D supplementation for 5 years, were studied for risk of falls, compared with 12,994 individuals in a placebo group. At baseline, 33.3% of patients had fallen at least once in the previous year, and overall 6,605 patients reported 13,235 falls. At 5.3 years of follow-up, there were no significant differences in number of falls between groups, falls leading to injury, and falls leading to a doctor or a hospital visit.
There are ongoing parallel studies examining the incidence of fractures between groups in the total population of the VITAL study (25,871 participants); bone turnover markers; bone microarchitecture measurements through high-resolution peripheral quantitative computed tomography; and examining the connection between free 25(OH)D, parathyroid hormone, and vitamin D binding protein, said Dr. LeBoff.
The study was funded in part by grants from the National Cancer Institute, the National Heart, Lung and Blood Institute, the Office of Dietary Supplements, the National Institute of Neurological Disorders and Stroke, and the National Center for Complementary and Integrative Health. Dr. LeBoff reported receiving grants from the National Institute of Arthritis Musculoskeletal and Skin Diseases. Two authors reported nonfinancial support Pharmavite LLC of Northridge, Calif., Pronova BioPharma of Norway and BASF, and Quest Diagnostics. The remaining authors reported no conflicts of interest.
SOURCE: LeBoff M et al. ASBMR 2019, Abstracts 1046 and 1057.
ORLANDO – after 2 years of daily use, according to data presented at the annual meeting of the American Society for Bone and Mineral Research.
“Participants may have already reached the vitamin D level needed for bone health,” Meryl S. LeBoff, MD, of Brigham and Women’s Hospital in Boston, said in her presentation.
Dr. LeBoff presented results from 771 patients (mean age, 63.8 years) in the Bone Health Subcohort of VITAL (Vitamin D and OmegA-3 TriaL) who were not on any bone active medications and were randomized to receive daily vitamin D3 at a dose of 2,000 IU or placebo. Patients received bone imaging at baseline and at 2 years; areal bone mineral density (aBMD) of the whole body, femoral neck, total hip, and spine was assessed via dual x-ray absorptiometry scan. Total 25-hydroxyvitamin D (25[OH]D) levels were measured via liquid chromatography tandem mass spectrometry, and free 25(OH)D levels were measured via the ELISA assay. The baseline characteristics of the vitamin D3 supplementation and placebo groups were similar. Overall, 52% of patients had osteopenia and 10.4% had osteoporosis.
Between baseline and 2 years, the vitamin D group’s total 25(OH)D levels increased from a mean 27.0 ng/mL to 39.5 ng/mL (46%) and the free 25(OH)D levels increased from 5.8 pg/mL to 9.0 pg/mL (55%), whereas levels in the placebo stayed the same. The researchers found no significant absolute percentage changes over 2 years in aBMD of the whole body (P = .60), femoral neck (P = .16), total hip (P = .23) and spine (P = .55), compared with patients in the placebo group.
In a secondary analysis, Dr. LeBoff and colleagues found no benefit to volumetric BMD (vBMD) of the radius and the tibia at 2 years, and the results persisted after they performed a sensitivity analysis. Adverse events, such as hypercalciuria, kidney stones, and gastrointestinal symptoms, were not significantly different in the vitamin D group, compared with the placebo group.
Dr. LeBoff noted among the limitations of the study that it evaluated one dose level of vitamin D and was not designed to determine whether vitamin D supplementation was effective in people with vitamin D insufficiency, and the results are not generalizable to patients with osteoporosis or osteomalacia. Future studies should also examine whether free 25(OH)D levels can be used to detect which patients can benefit from vitamin D supplementation, she added.
Risk of falls
In a separate abstract, which Dr. LeBoff presented in a different session, 12,927 patients who received vitamin D supplementation for 5 years, were studied for risk of falls, compared with 12,994 individuals in a placebo group. At baseline, 33.3% of patients had fallen at least once in the previous year, and overall 6,605 patients reported 13,235 falls. At 5.3 years of follow-up, there were no significant differences in number of falls between groups, falls leading to injury, and falls leading to a doctor or a hospital visit.
There are ongoing parallel studies examining the incidence of fractures between groups in the total population of the VITAL study (25,871 participants); bone turnover markers; bone microarchitecture measurements through high-resolution peripheral quantitative computed tomography; and examining the connection between free 25(OH)D, parathyroid hormone, and vitamin D binding protein, said Dr. LeBoff.
The study was funded in part by grants from the National Cancer Institute, the National Heart, Lung and Blood Institute, the Office of Dietary Supplements, the National Institute of Neurological Disorders and Stroke, and the National Center for Complementary and Integrative Health. Dr. LeBoff reported receiving grants from the National Institute of Arthritis Musculoskeletal and Skin Diseases. Two authors reported nonfinancial support Pharmavite LLC of Northridge, Calif., Pronova BioPharma of Norway and BASF, and Quest Diagnostics. The remaining authors reported no conflicts of interest.
SOURCE: LeBoff M et al. ASBMR 2019, Abstracts 1046 and 1057.
REPORTING FROM ASBMR 2019
Project ECHO helps osteoporosis specialists connect with PCPs
ORLANDO – The use of a teleconferencing program to share knowledge about osteoporosis has helped health care professionals learn about the disease and may potentially reduce the osteoporosis treatment gap in underserved communities, according to a speaker at the annual meeting of the American Society for Bone and Mineral Research.
The concept, called “technology-enabled collaborative learning,” is intended to address the problem of there being not enough specialists to see patients who need treatment, and the ineffectiveness of educating primary care providers in how to treat complex medical conditions, E. Michael Lewiecki, MD, the director of the New Mexico Clinical Research & Osteoporosis Center said in his presentation.
“Primary care doctors are busy,” said Dr. Lewiecki. “They have limited time taking care of patients. They don’t have the time or often the skills to manage patients who have any questions or concerns about osteoporosis and treatments for osteoporosis.”
One solution, he said, is to find health care professionals in underserved communities who are already interested in and motivated to learn more about osteoporosis, turn them into near-experts on osteoporosis for their patients as well as in their own community.
Dr. Lewiecki proposed the Extension for Community Healthcare Outcomes (ECHO), or Project ECHO, an initiative out of the University of New Mexico School of Medicine, as a potential answer. Project ECHO uses videoconferencing to connect experts in a therapeutic area, with Bone Health TeleECHO focusing on raising knowledge of osteoporosis for its participants. “The idea of ECHO is to be a force multiplier to educate health care professionals, each of whom takes care of many patients, and to have many ECHO programs around the world in convenient time zones and convenient languages for people who are interested in participating,” said Dr. Lewiecki.
The idea began when a gastroenterologist at Dr. Lewiecki’s own center was frustrated that patients were not seeking treatment for hepatitis C because of time or travel issues. In response, a pilot program for Project ECHO was developed through a collaboration between the University of New Mexico Health Sciences Center and the Osteoporosis Foundation of New Mexico where gastroenterologists at University of New Mexico connected with primary care providers across the state, sharing information about hepatitis C and discussing case studies. The results of the pilot program were published in the New England Journal of Medicine and showed a similar rate of sustained viral response between patients treated at the University of New Mexico clinic (84 of 146 patients; 57.5%) and at 21 ECHO clinics (152 of 261 patients; 58.2%) (Arora S et al. N Eng J Med. 2011. doi: 10.1056/NEJMoa1009370).
“ECHO expands the capacity to deliver best practice medical care through collegial, interactive, case-based discussions with minimal disruption to the office routine,” said Dr. Lewiecki. “Patients benefit from better care, closer to home, with greater convenience and lower cost than referral to a medical center. And the potential is to reduce the osteoporosis treatment gap by having many ECHOs starting up in many places in the world.”
Today, the ECHO program is in 37 countries, with 322 ECHO hubs and 677 ECHO programs. The top three specialties are endocrinology, orthopedics, and rheumatology; 51% of ECHO participants are primary care providers, 24% are advanced care providers, and 19% are health care providers such as nutritionists, physical therapists, and other providers that have an interest in bone health.
In survey results adapted from a 2017 study from his own group, Dr. Lewiecki showed that 263 health care professionals who participated in Bone Health TeleECHO rated themselves as more confident in 20 different domains of osteoporosis treatment, such as secondary osteoporosis and anabolic therapy, after 21 months of using the ECHO program (Lewiecki EM et al. J Endocr Soc. 2017. doi: 10.1210/js.2017-00361). However, he admitted that showing fracture prevention outcomes at these ECHO centers has proven more difficult.
“Of course, we’re all interested in outcomes. The ultimate outcome here is preventing fractures, but it is extraordinarily difficult to design a study to actually show that we’re reducing fractures, but certainly self-confidence in managing osteoporosis has improved,” he said.
There have also been some misconceptions of the Project ECHO. The program is not only for beginners or primary care providers, said Dr. Lewiecki. It is also not limited to providers in rural areas, as the program has many participants at urban centers, he added.
“We are a virtual community of practice. It’s a collegial relationship,” he said. “It’s really recapitulating the way that we learned during our postgraduate training: When we see a patient, we present the case to our attending, the attending pontificates a little bit, we bounce things off of one another, and we go back and then we do some different things with our patients. And that’s exactly what we do with Echo. It makes learning fun again.”
Dr. Lewiecki challenged the attendees in the room who are already experts in osteoporosis to help share their knowledge of the disease to help other health care professionals learn more about how to better care for their patients. “If you have a passion for teaching, if you want to share knowledge and you’re willing to devote a little bit of your time to doing that and reaching out to more people, this is the way that you can do it.”
Dr. Lewiecki reports research grant support from Amgen, consulting fees from Alexion, Amgen, Radius, Shire, and Ultragenyx, speaking fees from Alexion, Radius, and Shire, and is an advisory board member with the National Osteoporosis Foundation, International Society for Clinical Densitometry, and the Osteoporosis Foundation of New Mexico.
SOURCE: Lewiecki ME. ASBMR 2019. Symposia: Cutting Edge Concepts: Novel Approaches to Reducing Fractures. Bone Health TeleECHO.
ORLANDO – The use of a teleconferencing program to share knowledge about osteoporosis has helped health care professionals learn about the disease and may potentially reduce the osteoporosis treatment gap in underserved communities, according to a speaker at the annual meeting of the American Society for Bone and Mineral Research.
The concept, called “technology-enabled collaborative learning,” is intended to address the problem of there being not enough specialists to see patients who need treatment, and the ineffectiveness of educating primary care providers in how to treat complex medical conditions, E. Michael Lewiecki, MD, the director of the New Mexico Clinical Research & Osteoporosis Center said in his presentation.
“Primary care doctors are busy,” said Dr. Lewiecki. “They have limited time taking care of patients. They don’t have the time or often the skills to manage patients who have any questions or concerns about osteoporosis and treatments for osteoporosis.”
One solution, he said, is to find health care professionals in underserved communities who are already interested in and motivated to learn more about osteoporosis, turn them into near-experts on osteoporosis for their patients as well as in their own community.
Dr. Lewiecki proposed the Extension for Community Healthcare Outcomes (ECHO), or Project ECHO, an initiative out of the University of New Mexico School of Medicine, as a potential answer. Project ECHO uses videoconferencing to connect experts in a therapeutic area, with Bone Health TeleECHO focusing on raising knowledge of osteoporosis for its participants. “The idea of ECHO is to be a force multiplier to educate health care professionals, each of whom takes care of many patients, and to have many ECHO programs around the world in convenient time zones and convenient languages for people who are interested in participating,” said Dr. Lewiecki.
The idea began when a gastroenterologist at Dr. Lewiecki’s own center was frustrated that patients were not seeking treatment for hepatitis C because of time or travel issues. In response, a pilot program for Project ECHO was developed through a collaboration between the University of New Mexico Health Sciences Center and the Osteoporosis Foundation of New Mexico where gastroenterologists at University of New Mexico connected with primary care providers across the state, sharing information about hepatitis C and discussing case studies. The results of the pilot program were published in the New England Journal of Medicine and showed a similar rate of sustained viral response between patients treated at the University of New Mexico clinic (84 of 146 patients; 57.5%) and at 21 ECHO clinics (152 of 261 patients; 58.2%) (Arora S et al. N Eng J Med. 2011. doi: 10.1056/NEJMoa1009370).
“ECHO expands the capacity to deliver best practice medical care through collegial, interactive, case-based discussions with minimal disruption to the office routine,” said Dr. Lewiecki. “Patients benefit from better care, closer to home, with greater convenience and lower cost than referral to a medical center. And the potential is to reduce the osteoporosis treatment gap by having many ECHOs starting up in many places in the world.”
Today, the ECHO program is in 37 countries, with 322 ECHO hubs and 677 ECHO programs. The top three specialties are endocrinology, orthopedics, and rheumatology; 51% of ECHO participants are primary care providers, 24% are advanced care providers, and 19% are health care providers such as nutritionists, physical therapists, and other providers that have an interest in bone health.
In survey results adapted from a 2017 study from his own group, Dr. Lewiecki showed that 263 health care professionals who participated in Bone Health TeleECHO rated themselves as more confident in 20 different domains of osteoporosis treatment, such as secondary osteoporosis and anabolic therapy, after 21 months of using the ECHO program (Lewiecki EM et al. J Endocr Soc. 2017. doi: 10.1210/js.2017-00361). However, he admitted that showing fracture prevention outcomes at these ECHO centers has proven more difficult.
“Of course, we’re all interested in outcomes. The ultimate outcome here is preventing fractures, but it is extraordinarily difficult to design a study to actually show that we’re reducing fractures, but certainly self-confidence in managing osteoporosis has improved,” he said.
There have also been some misconceptions of the Project ECHO. The program is not only for beginners or primary care providers, said Dr. Lewiecki. It is also not limited to providers in rural areas, as the program has many participants at urban centers, he added.
“We are a virtual community of practice. It’s a collegial relationship,” he said. “It’s really recapitulating the way that we learned during our postgraduate training: When we see a patient, we present the case to our attending, the attending pontificates a little bit, we bounce things off of one another, and we go back and then we do some different things with our patients. And that’s exactly what we do with Echo. It makes learning fun again.”
Dr. Lewiecki challenged the attendees in the room who are already experts in osteoporosis to help share their knowledge of the disease to help other health care professionals learn more about how to better care for their patients. “If you have a passion for teaching, if you want to share knowledge and you’re willing to devote a little bit of your time to doing that and reaching out to more people, this is the way that you can do it.”
Dr. Lewiecki reports research grant support from Amgen, consulting fees from Alexion, Amgen, Radius, Shire, and Ultragenyx, speaking fees from Alexion, Radius, and Shire, and is an advisory board member with the National Osteoporosis Foundation, International Society for Clinical Densitometry, and the Osteoporosis Foundation of New Mexico.
SOURCE: Lewiecki ME. ASBMR 2019. Symposia: Cutting Edge Concepts: Novel Approaches to Reducing Fractures. Bone Health TeleECHO.
ORLANDO – The use of a teleconferencing program to share knowledge about osteoporosis has helped health care professionals learn about the disease and may potentially reduce the osteoporosis treatment gap in underserved communities, according to a speaker at the annual meeting of the American Society for Bone and Mineral Research.
The concept, called “technology-enabled collaborative learning,” is intended to address the problem of there being not enough specialists to see patients who need treatment, and the ineffectiveness of educating primary care providers in how to treat complex medical conditions, E. Michael Lewiecki, MD, the director of the New Mexico Clinical Research & Osteoporosis Center said in his presentation.
“Primary care doctors are busy,” said Dr. Lewiecki. “They have limited time taking care of patients. They don’t have the time or often the skills to manage patients who have any questions or concerns about osteoporosis and treatments for osteoporosis.”
One solution, he said, is to find health care professionals in underserved communities who are already interested in and motivated to learn more about osteoporosis, turn them into near-experts on osteoporosis for their patients as well as in their own community.
Dr. Lewiecki proposed the Extension for Community Healthcare Outcomes (ECHO), or Project ECHO, an initiative out of the University of New Mexico School of Medicine, as a potential answer. Project ECHO uses videoconferencing to connect experts in a therapeutic area, with Bone Health TeleECHO focusing on raising knowledge of osteoporosis for its participants. “The idea of ECHO is to be a force multiplier to educate health care professionals, each of whom takes care of many patients, and to have many ECHO programs around the world in convenient time zones and convenient languages for people who are interested in participating,” said Dr. Lewiecki.
The idea began when a gastroenterologist at Dr. Lewiecki’s own center was frustrated that patients were not seeking treatment for hepatitis C because of time or travel issues. In response, a pilot program for Project ECHO was developed through a collaboration between the University of New Mexico Health Sciences Center and the Osteoporosis Foundation of New Mexico where gastroenterologists at University of New Mexico connected with primary care providers across the state, sharing information about hepatitis C and discussing case studies. The results of the pilot program were published in the New England Journal of Medicine and showed a similar rate of sustained viral response between patients treated at the University of New Mexico clinic (84 of 146 patients; 57.5%) and at 21 ECHO clinics (152 of 261 patients; 58.2%) (Arora S et al. N Eng J Med. 2011. doi: 10.1056/NEJMoa1009370).
“ECHO expands the capacity to deliver best practice medical care through collegial, interactive, case-based discussions with minimal disruption to the office routine,” said Dr. Lewiecki. “Patients benefit from better care, closer to home, with greater convenience and lower cost than referral to a medical center. And the potential is to reduce the osteoporosis treatment gap by having many ECHOs starting up in many places in the world.”
Today, the ECHO program is in 37 countries, with 322 ECHO hubs and 677 ECHO programs. The top three specialties are endocrinology, orthopedics, and rheumatology; 51% of ECHO participants are primary care providers, 24% are advanced care providers, and 19% are health care providers such as nutritionists, physical therapists, and other providers that have an interest in bone health.
In survey results adapted from a 2017 study from his own group, Dr. Lewiecki showed that 263 health care professionals who participated in Bone Health TeleECHO rated themselves as more confident in 20 different domains of osteoporosis treatment, such as secondary osteoporosis and anabolic therapy, after 21 months of using the ECHO program (Lewiecki EM et al. J Endocr Soc. 2017. doi: 10.1210/js.2017-00361). However, he admitted that showing fracture prevention outcomes at these ECHO centers has proven more difficult.
“Of course, we’re all interested in outcomes. The ultimate outcome here is preventing fractures, but it is extraordinarily difficult to design a study to actually show that we’re reducing fractures, but certainly self-confidence in managing osteoporosis has improved,” he said.
There have also been some misconceptions of the Project ECHO. The program is not only for beginners or primary care providers, said Dr. Lewiecki. It is also not limited to providers in rural areas, as the program has many participants at urban centers, he added.
“We are a virtual community of practice. It’s a collegial relationship,” he said. “It’s really recapitulating the way that we learned during our postgraduate training: When we see a patient, we present the case to our attending, the attending pontificates a little bit, we bounce things off of one another, and we go back and then we do some different things with our patients. And that’s exactly what we do with Echo. It makes learning fun again.”
Dr. Lewiecki challenged the attendees in the room who are already experts in osteoporosis to help share their knowledge of the disease to help other health care professionals learn more about how to better care for their patients. “If you have a passion for teaching, if you want to share knowledge and you’re willing to devote a little bit of your time to doing that and reaching out to more people, this is the way that you can do it.”
Dr. Lewiecki reports research grant support from Amgen, consulting fees from Alexion, Amgen, Radius, Shire, and Ultragenyx, speaking fees from Alexion, Radius, and Shire, and is an advisory board member with the National Osteoporosis Foundation, International Society for Clinical Densitometry, and the Osteoporosis Foundation of New Mexico.
SOURCE: Lewiecki ME. ASBMR 2019. Symposia: Cutting Edge Concepts: Novel Approaches to Reducing Fractures. Bone Health TeleECHO.
FROM ASBMR 2019
High mortality rates trail tracheostomy patients
findings of a large retrospective study suggest.
Current outcome prediction tools to support decision making regarding tracheostomies are limited, wrote Anuj B. Mehta, MD, of National Jewish Health in Denver, and colleagues. “This study provides novel and in-depth insight into mortality and health care utilization following tracheostomy not previously described at the population-level.”
In a study published in Critical Care Medicine, the researchers reviewed data from 8,343 nonsurgical patients seen in California hospitals from 2012 to 2013 who received a tracheostomy for acute respiratory failure.
Overall, the 1-year mortality rate for patients who had tracheostomies (the primary outcome) was 46.5%, with in-hospital mortality of 18.9% and 30-day mortality of 22.1%. Pneumonia was the most common diagnosis for patients with respiratory failure (79%) and some had an additional diagnosis, such as severe sepsis (56%).
Patients aged 65 years and older had significantly higher mortality than those under 65 (54.7% vs. 36.5%). The average age of the patients was 65 years; approximately 46% were women and 48% were white. The median survival for adults aged 65 years and older was 175 days, compared with median survival of more than a year for younger patients.
Secondary outcomes included discharge destination, hospital readmission, and health care utilization. A majority (86%) of patients were discharged to a long-term care facility, while 11% were sent home and approximately 3% were discharged to other destinations.
Nearly two-thirds (60%) of patients were readmitted to the hospital within a year of tracheostomy, and readmission was more common among older adults, compared with younger (66% vs. 55%).
In addition, just over one-third of all patients (36%) spent more than 50% of their days alive in the hospital in short-term acute care, and this rate was significantly higher for patients aged 65 years and older, compared with those under 65 (43% vs. 29%). On average, the total hospital cost for patients who survived the first year after tracheostomy was $215,369, with no significant difference in average cost among age groups.
The study findings were limited by several factors including the use of data from a single state, possible misclassification of billing codes, and inability to measure quality of life, the researchers noted.
However, “our findings of high mortality, low median survival for older patients, high readmission rates, potentially burdensome cost, and informative outcome trajectories provide significant insight into long-term outcomes following tracheostomy,” they concluded.
Dr. Mehta and several colleagues reported receiving funding from the National Institutes of Health. The researchers had no financial conflicts to disclose.
SOURCE: Mehta AB et al. Crit Care Med. 2019 Aug 8. doi: 10.1097/CCM.0000000000003959.
findings of a large retrospective study suggest.
Current outcome prediction tools to support decision making regarding tracheostomies are limited, wrote Anuj B. Mehta, MD, of National Jewish Health in Denver, and colleagues. “This study provides novel and in-depth insight into mortality and health care utilization following tracheostomy not previously described at the population-level.”
In a study published in Critical Care Medicine, the researchers reviewed data from 8,343 nonsurgical patients seen in California hospitals from 2012 to 2013 who received a tracheostomy for acute respiratory failure.
Overall, the 1-year mortality rate for patients who had tracheostomies (the primary outcome) was 46.5%, with in-hospital mortality of 18.9% and 30-day mortality of 22.1%. Pneumonia was the most common diagnosis for patients with respiratory failure (79%) and some had an additional diagnosis, such as severe sepsis (56%).
Patients aged 65 years and older had significantly higher mortality than those under 65 (54.7% vs. 36.5%). The average age of the patients was 65 years; approximately 46% were women and 48% were white. The median survival for adults aged 65 years and older was 175 days, compared with median survival of more than a year for younger patients.
Secondary outcomes included discharge destination, hospital readmission, and health care utilization. A majority (86%) of patients were discharged to a long-term care facility, while 11% were sent home and approximately 3% were discharged to other destinations.
Nearly two-thirds (60%) of patients were readmitted to the hospital within a year of tracheostomy, and readmission was more common among older adults, compared with younger (66% vs. 55%).
In addition, just over one-third of all patients (36%) spent more than 50% of their days alive in the hospital in short-term acute care, and this rate was significantly higher for patients aged 65 years and older, compared with those under 65 (43% vs. 29%). On average, the total hospital cost for patients who survived the first year after tracheostomy was $215,369, with no significant difference in average cost among age groups.
The study findings were limited by several factors including the use of data from a single state, possible misclassification of billing codes, and inability to measure quality of life, the researchers noted.
However, “our findings of high mortality, low median survival for older patients, high readmission rates, potentially burdensome cost, and informative outcome trajectories provide significant insight into long-term outcomes following tracheostomy,” they concluded.
Dr. Mehta and several colleagues reported receiving funding from the National Institutes of Health. The researchers had no financial conflicts to disclose.
SOURCE: Mehta AB et al. Crit Care Med. 2019 Aug 8. doi: 10.1097/CCM.0000000000003959.
findings of a large retrospective study suggest.
Current outcome prediction tools to support decision making regarding tracheostomies are limited, wrote Anuj B. Mehta, MD, of National Jewish Health in Denver, and colleagues. “This study provides novel and in-depth insight into mortality and health care utilization following tracheostomy not previously described at the population-level.”
In a study published in Critical Care Medicine, the researchers reviewed data from 8,343 nonsurgical patients seen in California hospitals from 2012 to 2013 who received a tracheostomy for acute respiratory failure.
Overall, the 1-year mortality rate for patients who had tracheostomies (the primary outcome) was 46.5%, with in-hospital mortality of 18.9% and 30-day mortality of 22.1%. Pneumonia was the most common diagnosis for patients with respiratory failure (79%) and some had an additional diagnosis, such as severe sepsis (56%).
Patients aged 65 years and older had significantly higher mortality than those under 65 (54.7% vs. 36.5%). The average age of the patients was 65 years; approximately 46% were women and 48% were white. The median survival for adults aged 65 years and older was 175 days, compared with median survival of more than a year for younger patients.
Secondary outcomes included discharge destination, hospital readmission, and health care utilization. A majority (86%) of patients were discharged to a long-term care facility, while 11% were sent home and approximately 3% were discharged to other destinations.
Nearly two-thirds (60%) of patients were readmitted to the hospital within a year of tracheostomy, and readmission was more common among older adults, compared with younger (66% vs. 55%).
In addition, just over one-third of all patients (36%) spent more than 50% of their days alive in the hospital in short-term acute care, and this rate was significantly higher for patients aged 65 years and older, compared with those under 65 (43% vs. 29%). On average, the total hospital cost for patients who survived the first year after tracheostomy was $215,369, with no significant difference in average cost among age groups.
The study findings were limited by several factors including the use of data from a single state, possible misclassification of billing codes, and inability to measure quality of life, the researchers noted.
However, “our findings of high mortality, low median survival for older patients, high readmission rates, potentially burdensome cost, and informative outcome trajectories provide significant insight into long-term outcomes following tracheostomy,” they concluded.
Dr. Mehta and several colleagues reported receiving funding from the National Institutes of Health. The researchers had no financial conflicts to disclose.
SOURCE: Mehta AB et al. Crit Care Med. 2019 Aug 8. doi: 10.1097/CCM.0000000000003959.
FROM CRITICAL CARE MEDICINE
Daily polypill lowers BP, cholesterol in underserved population
A daily polypill regimen improved cardiovascular risk factors in a socioeconomically vulnerable minority population, in a randomized controlled trial.
Patients at a federally qualified community health center in Alabama who received treatment with a combination pill for 1 year had greater reductions in systolic blood pressure and LDL cholesterol than did patients who received usual care, according to results published online on Sept. 19 in the New England Journal of Medicine.
“The simplicity and low cost of the polypill regimen make this approach attractive” when barriers such as lack of income, underinsurance, and difficulty attending clinic visits are common, said first author Daniel Muñoz, MD, of Vanderbilt University in Nashville, and coinvestigators. The investigators obtained the pills at a cost of $26 per month per participant.
People with low socioeconomic status and those who are nonwhite have high cardiovascular mortality, and the southeastern United States and rural areas have disproportionately high levels of cardiovascular disease burden, according to the investigators. The rates at which people with low socioeconomic status receive treatment for hypertension and hypercholesterolemia – leading cardiovascular disease risk factors – “are strikingly low,” Dr. Muñoz and colleagues said.
To assess the effectiveness of a polypill-based strategy in an underserved population with low socioeconomic status, the researchers conducted the randomized trial.
They enrolled 303 adults without cardiovascular disease, and 148 of the patients were randomized to receive the polypill, which contained generic versions of atorvastatin (10 mg), amlodipine (2.5 mg), losartan (25 mg), and hydrochlorothiazide (12.5 mg). The remaining 155 patients received usual care. All participants scheduled 2-month and 12-month follow-up visits.
The participants had an average age of 56 years, 60% were women, and more than 95% were black. More than 70% had an annual household income of less than $15,000. Baseline characteristics of the treatment groups did not significantly differ.
At baseline, the average BP was 140/83 mm Hg, and the average LDL cholesterol level was 113 mg/dL.
In all, 91% of the participants completed the 12-month trial visit. Average systolic BP decreased by 9 mm Hg in the group that received the polypill, compared with 2 mm Hg in the group that received usual care. Average LDL cholesterol level decreased by 15 mg/dL in the polypill group, versus 4 mg/dL in the usual-care group.
Changes in other medications
Clinicians discontinued or reduced doses of other antihypertensive or lipid-lowering medications in 44% of the patients in the polypill group and none in the usual-care group. Clinicians escalated therapy in 2% of the participants in the polypill group and in 10% of the usual-care group.
Side effects in participants who received the polypill included a 1% incidence of myalgias and a 1% incidence of hypotension or light-headedness. Liver function test results were normal.
Five serious adverse events that occurred during the trial – two in the polypill group and three in the usual-care group – were judged to be unrelated to the trial by a data and safety monitoring board.
The authors noted that limitations of the trial include its open-label design and that it was conducted at a single center.
“It is important to emphasize that use of the polypill does not preclude individualized, add-on therapies for residual elevations in blood-pressure or cholesterol levels, as judged by a patient’s physician,” said Dr. Muñoz and colleagues. “We recognize that a ‘one size fits all’ approach to cardiovascular disease prevention runs counter to current trends in precision medicine, in which clinical, genomic, and lifestyle factors are used for the development of individualized treatment strategies. Although the precision approach has clear virtues, a broader approach may benefit patients who face barriers to accessing the full advantages of precision medicine.”
The study was supported by grants from the American Heart Association Strategically Focused Prevention Research Network and the National Institutes of Health. One author disclosed personal fees from Novartis outside the study.
SOURCE: Muñoz D et al. N Engl J Med. 2019 Sep 18;381(12):1114-23. doi: 10.1056/NEJMoa1815359.
A daily polypill regimen improved cardiovascular risk factors in a socioeconomically vulnerable minority population, in a randomized controlled trial.
Patients at a federally qualified community health center in Alabama who received treatment with a combination pill for 1 year had greater reductions in systolic blood pressure and LDL cholesterol than did patients who received usual care, according to results published online on Sept. 19 in the New England Journal of Medicine.
“The simplicity and low cost of the polypill regimen make this approach attractive” when barriers such as lack of income, underinsurance, and difficulty attending clinic visits are common, said first author Daniel Muñoz, MD, of Vanderbilt University in Nashville, and coinvestigators. The investigators obtained the pills at a cost of $26 per month per participant.
People with low socioeconomic status and those who are nonwhite have high cardiovascular mortality, and the southeastern United States and rural areas have disproportionately high levels of cardiovascular disease burden, according to the investigators. The rates at which people with low socioeconomic status receive treatment for hypertension and hypercholesterolemia – leading cardiovascular disease risk factors – “are strikingly low,” Dr. Muñoz and colleagues said.
To assess the effectiveness of a polypill-based strategy in an underserved population with low socioeconomic status, the researchers conducted the randomized trial.
They enrolled 303 adults without cardiovascular disease, and 148 of the patients were randomized to receive the polypill, which contained generic versions of atorvastatin (10 mg), amlodipine (2.5 mg), losartan (25 mg), and hydrochlorothiazide (12.5 mg). The remaining 155 patients received usual care. All participants scheduled 2-month and 12-month follow-up visits.
The participants had an average age of 56 years, 60% were women, and more than 95% were black. More than 70% had an annual household income of less than $15,000. Baseline characteristics of the treatment groups did not significantly differ.
At baseline, the average BP was 140/83 mm Hg, and the average LDL cholesterol level was 113 mg/dL.
In all, 91% of the participants completed the 12-month trial visit. Average systolic BP decreased by 9 mm Hg in the group that received the polypill, compared with 2 mm Hg in the group that received usual care. Average LDL cholesterol level decreased by 15 mg/dL in the polypill group, versus 4 mg/dL in the usual-care group.
Changes in other medications
Clinicians discontinued or reduced doses of other antihypertensive or lipid-lowering medications in 44% of the patients in the polypill group and none in the usual-care group. Clinicians escalated therapy in 2% of the participants in the polypill group and in 10% of the usual-care group.
Side effects in participants who received the polypill included a 1% incidence of myalgias and a 1% incidence of hypotension or light-headedness. Liver function test results were normal.
Five serious adverse events that occurred during the trial – two in the polypill group and three in the usual-care group – were judged to be unrelated to the trial by a data and safety monitoring board.
The authors noted that limitations of the trial include its open-label design and that it was conducted at a single center.
“It is important to emphasize that use of the polypill does not preclude individualized, add-on therapies for residual elevations in blood-pressure or cholesterol levels, as judged by a patient’s physician,” said Dr. Muñoz and colleagues. “We recognize that a ‘one size fits all’ approach to cardiovascular disease prevention runs counter to current trends in precision medicine, in which clinical, genomic, and lifestyle factors are used for the development of individualized treatment strategies. Although the precision approach has clear virtues, a broader approach may benefit patients who face barriers to accessing the full advantages of precision medicine.”
The study was supported by grants from the American Heart Association Strategically Focused Prevention Research Network and the National Institutes of Health. One author disclosed personal fees from Novartis outside the study.
SOURCE: Muñoz D et al. N Engl J Med. 2019 Sep 18;381(12):1114-23. doi: 10.1056/NEJMoa1815359.
A daily polypill regimen improved cardiovascular risk factors in a socioeconomically vulnerable minority population, in a randomized controlled trial.
Patients at a federally qualified community health center in Alabama who received treatment with a combination pill for 1 year had greater reductions in systolic blood pressure and LDL cholesterol than did patients who received usual care, according to results published online on Sept. 19 in the New England Journal of Medicine.
“The simplicity and low cost of the polypill regimen make this approach attractive” when barriers such as lack of income, underinsurance, and difficulty attending clinic visits are common, said first author Daniel Muñoz, MD, of Vanderbilt University in Nashville, and coinvestigators. The investigators obtained the pills at a cost of $26 per month per participant.
People with low socioeconomic status and those who are nonwhite have high cardiovascular mortality, and the southeastern United States and rural areas have disproportionately high levels of cardiovascular disease burden, according to the investigators. The rates at which people with low socioeconomic status receive treatment for hypertension and hypercholesterolemia – leading cardiovascular disease risk factors – “are strikingly low,” Dr. Muñoz and colleagues said.
To assess the effectiveness of a polypill-based strategy in an underserved population with low socioeconomic status, the researchers conducted the randomized trial.
They enrolled 303 adults without cardiovascular disease, and 148 of the patients were randomized to receive the polypill, which contained generic versions of atorvastatin (10 mg), amlodipine (2.5 mg), losartan (25 mg), and hydrochlorothiazide (12.5 mg). The remaining 155 patients received usual care. All participants scheduled 2-month and 12-month follow-up visits.
The participants had an average age of 56 years, 60% were women, and more than 95% were black. More than 70% had an annual household income of less than $15,000. Baseline characteristics of the treatment groups did not significantly differ.
At baseline, the average BP was 140/83 mm Hg, and the average LDL cholesterol level was 113 mg/dL.
In all, 91% of the participants completed the 12-month trial visit. Average systolic BP decreased by 9 mm Hg in the group that received the polypill, compared with 2 mm Hg in the group that received usual care. Average LDL cholesterol level decreased by 15 mg/dL in the polypill group, versus 4 mg/dL in the usual-care group.
Changes in other medications
Clinicians discontinued or reduced doses of other antihypertensive or lipid-lowering medications in 44% of the patients in the polypill group and none in the usual-care group. Clinicians escalated therapy in 2% of the participants in the polypill group and in 10% of the usual-care group.
Side effects in participants who received the polypill included a 1% incidence of myalgias and a 1% incidence of hypotension or light-headedness. Liver function test results were normal.
Five serious adverse events that occurred during the trial – two in the polypill group and three in the usual-care group – were judged to be unrelated to the trial by a data and safety monitoring board.
The authors noted that limitations of the trial include its open-label design and that it was conducted at a single center.
“It is important to emphasize that use of the polypill does not preclude individualized, add-on therapies for residual elevations in blood-pressure or cholesterol levels, as judged by a patient’s physician,” said Dr. Muñoz and colleagues. “We recognize that a ‘one size fits all’ approach to cardiovascular disease prevention runs counter to current trends in precision medicine, in which clinical, genomic, and lifestyle factors are used for the development of individualized treatment strategies. Although the precision approach has clear virtues, a broader approach may benefit patients who face barriers to accessing the full advantages of precision medicine.”
The study was supported by grants from the American Heart Association Strategically Focused Prevention Research Network and the National Institutes of Health. One author disclosed personal fees from Novartis outside the study.
SOURCE: Muñoz D et al. N Engl J Med. 2019 Sep 18;381(12):1114-23. doi: 10.1056/NEJMoa1815359.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Key clinical point: A daily polypill regimen may improve cardiovascular disease prevention in underserved populations.
Major finding: Mean LDL cholesterol levels decreased by 15 mg/dL in the polypill group, vs. 4 mg/dL in the usual-care group.
Study details: An open-label, randomized trial that enrolled 303 adults without cardiovascular disease at a federally qualified community health center in Alabama.
Disclosures: The study was supported by grants from the American Heart Association Strategically Focused Prevention Research Network and the National Institutes of Health. One author disclosed personal fees from Novartis outside the study.
Source: Muñoz D et al. N Engl J Med. 2019;381(12):1114-23. doi: 10.1056/NEJMoa1815359.
Older IBD patients are most at risk of postdischarge VTE
Hospitalized patients with inflammatory bowel diseases (IBD) are most likely to be readmitted for venous thromboembolism (VTE) within 60 days of discharge, according to a new study that analyzed 5 years of U.S. readmissions data.
“Given increased thrombotic risk postdischarge, as well as overall safety of VTE prophylaxis, extending prophylaxis for those at highest risk may have significant benefits,” wrote Adam S. Faye, MD, of Columbia University, and coauthors. The study was published in Clinical Gastroenterology and Hepatology.
To determine which IBD patients would be most in need of postdischarge VTE prophylaxis, as well as when to administer it, the researchers analyzed 2010-2014 data from the Nationwide Readmissions Database (NRD). They found a total of 872,122 index admissions for IBD patients; 4% of those patients had a prior VTE. Of the index admissions, 1,160 led to a VTE readmission within 90 days. Readmitted patients had a relatively equal proportion of ulcerative colitis (n = 522) and Crohn’s disease (n = 638).
More than 90% of VTE readmissions occurred within 60 days of discharge; the risk was highest over the first 10 days and then decreased in each ensuing 10-day period until a slight increase at the 81- to 90-day period. All patients over age 30 had higher rates of readmission than those of patients under age 18, with the highest risk in patients between the ages of 66 and 80 years (risk ratio 4.04; 95% confidence interval, 2.54-6.44, P less than .01). Women were at lower risk (RR 0.82; 95% CI, 0.73-0.92, P less than .01). Higher risks of readmission were also associated with being on Medicare (RR 1.39; 95% CI, 1.23-1.58, P less than .01) compared with being on private insurance and being cared for at a large hospital (RR 1.26; 95% CI, 1.04-1.52, P = .02) compared with a small hospital.
The highest risk of VTE readmission was associated with a prior history of VTE (RR 2.89; 95% CI, 2.40-3.48, P less than .01), having two or more comorbidities (RR 2.57; 95% CI, 2.11-3.12, P less than .01) and having a Clostridioides difficile infection as of index admission (RR 1.90; 95% CI, 1.51-2.38, P less than .01). In addition, increased risk was associated with being discharged to a nursing or care facility (RR 1.85; 95% CI, 1.56-2.20, P less than .01) or home with health services (RR 2.05; 95% CI, 1.78-2.38, P less than .01) compared with a routine discharge.
In their multivariable analysis, similar factors such as a history of VTE (adjusted RR 2.41; 95% CI, 1.99-2.90, P less than .01), two or more comorbidities (aRR 1.78; 95% CI, 1.44-2.20, P less than .01) and C. difficile infection (aRR 1.47; 95% CI, 1.17-1.85, P less than.01) continued to be associated with higher risk of VTE readmission.
Though they emphasized that the use of NRD data offered the impressive ability to “review over 15 million discharges across the U.S. annually,” Dr. Faye and coauthors acknowledged that their study did have limitations. These included the inability to verify via chart review the study’s outcomes and covariates. In addition, they were unable to assess potential contributing risk factors such as medication use, use of VTE prophylaxis during hospitalization, disease severity, and family history. Finally, though unlikely, they admitted the possibility that patients could be counted more than once if they were readmitted with a VTE each year of the study.
The authors reported being supported by grants from the National Institutes of Health and various pharmaceutical companies, as well as receiving honoraria and serving as consultants.
SOURCE: Faye AS et al. Clin Gastroenterol Hepatol. 2019 July 20. doi: 10.1016/j.cgh.2019.07.028.
Hospitalized patients with inflammatory bowel diseases (IBD) are most likely to be readmitted for venous thromboembolism (VTE) within 60 days of discharge, according to a new study that analyzed 5 years of U.S. readmissions data.
“Given increased thrombotic risk postdischarge, as well as overall safety of VTE prophylaxis, extending prophylaxis for those at highest risk may have significant benefits,” wrote Adam S. Faye, MD, of Columbia University, and coauthors. The study was published in Clinical Gastroenterology and Hepatology.
To determine which IBD patients would be most in need of postdischarge VTE prophylaxis, as well as when to administer it, the researchers analyzed 2010-2014 data from the Nationwide Readmissions Database (NRD). They found a total of 872,122 index admissions for IBD patients; 4% of those patients had a prior VTE. Of the index admissions, 1,160 led to a VTE readmission within 90 days. Readmitted patients had a relatively equal proportion of ulcerative colitis (n = 522) and Crohn’s disease (n = 638).
More than 90% of VTE readmissions occurred within 60 days of discharge; the risk was highest over the first 10 days and then decreased in each ensuing 10-day period until a slight increase at the 81- to 90-day period. All patients over age 30 had higher rates of readmission than those of patients under age 18, with the highest risk in patients between the ages of 66 and 80 years (risk ratio 4.04; 95% confidence interval, 2.54-6.44, P less than .01). Women were at lower risk (RR 0.82; 95% CI, 0.73-0.92, P less than .01). Higher risks of readmission were also associated with being on Medicare (RR 1.39; 95% CI, 1.23-1.58, P less than .01) compared with being on private insurance and being cared for at a large hospital (RR 1.26; 95% CI, 1.04-1.52, P = .02) compared with a small hospital.
The highest risk of VTE readmission was associated with a prior history of VTE (RR 2.89; 95% CI, 2.40-3.48, P less than .01), having two or more comorbidities (RR 2.57; 95% CI, 2.11-3.12, P less than .01) and having a Clostridioides difficile infection as of index admission (RR 1.90; 95% CI, 1.51-2.38, P less than .01). In addition, increased risk was associated with being discharged to a nursing or care facility (RR 1.85; 95% CI, 1.56-2.20, P less than .01) or home with health services (RR 2.05; 95% CI, 1.78-2.38, P less than .01) compared with a routine discharge.
In their multivariable analysis, similar factors such as a history of VTE (adjusted RR 2.41; 95% CI, 1.99-2.90, P less than .01), two or more comorbidities (aRR 1.78; 95% CI, 1.44-2.20, P less than .01) and C. difficile infection (aRR 1.47; 95% CI, 1.17-1.85, P less than.01) continued to be associated with higher risk of VTE readmission.
Though they emphasized that the use of NRD data offered the impressive ability to “review over 15 million discharges across the U.S. annually,” Dr. Faye and coauthors acknowledged that their study did have limitations. These included the inability to verify via chart review the study’s outcomes and covariates. In addition, they were unable to assess potential contributing risk factors such as medication use, use of VTE prophylaxis during hospitalization, disease severity, and family history. Finally, though unlikely, they admitted the possibility that patients could be counted more than once if they were readmitted with a VTE each year of the study.
The authors reported being supported by grants from the National Institutes of Health and various pharmaceutical companies, as well as receiving honoraria and serving as consultants.
SOURCE: Faye AS et al. Clin Gastroenterol Hepatol. 2019 July 20. doi: 10.1016/j.cgh.2019.07.028.
Hospitalized patients with inflammatory bowel diseases (IBD) are most likely to be readmitted for venous thromboembolism (VTE) within 60 days of discharge, according to a new study that analyzed 5 years of U.S. readmissions data.
“Given increased thrombotic risk postdischarge, as well as overall safety of VTE prophylaxis, extending prophylaxis for those at highest risk may have significant benefits,” wrote Adam S. Faye, MD, of Columbia University, and coauthors. The study was published in Clinical Gastroenterology and Hepatology.
To determine which IBD patients would be most in need of postdischarge VTE prophylaxis, as well as when to administer it, the researchers analyzed 2010-2014 data from the Nationwide Readmissions Database (NRD). They found a total of 872,122 index admissions for IBD patients; 4% of those patients had a prior VTE. Of the index admissions, 1,160 led to a VTE readmission within 90 days. Readmitted patients had a relatively equal proportion of ulcerative colitis (n = 522) and Crohn’s disease (n = 638).
More than 90% of VTE readmissions occurred within 60 days of discharge; the risk was highest over the first 10 days and then decreased in each ensuing 10-day period until a slight increase at the 81- to 90-day period. All patients over age 30 had higher rates of readmission than those of patients under age 18, with the highest risk in patients between the ages of 66 and 80 years (risk ratio 4.04; 95% confidence interval, 2.54-6.44, P less than .01). Women were at lower risk (RR 0.82; 95% CI, 0.73-0.92, P less than .01). Higher risks of readmission were also associated with being on Medicare (RR 1.39; 95% CI, 1.23-1.58, P less than .01) compared with being on private insurance and being cared for at a large hospital (RR 1.26; 95% CI, 1.04-1.52, P = .02) compared with a small hospital.
The highest risk of VTE readmission was associated with a prior history of VTE (RR 2.89; 95% CI, 2.40-3.48, P less than .01), having two or more comorbidities (RR 2.57; 95% CI, 2.11-3.12, P less than .01) and having a Clostridioides difficile infection as of index admission (RR 1.90; 95% CI, 1.51-2.38, P less than .01). In addition, increased risk was associated with being discharged to a nursing or care facility (RR 1.85; 95% CI, 1.56-2.20, P less than .01) or home with health services (RR 2.05; 95% CI, 1.78-2.38, P less than .01) compared with a routine discharge.
In their multivariable analysis, similar factors such as a history of VTE (adjusted RR 2.41; 95% CI, 1.99-2.90, P less than .01), two or more comorbidities (aRR 1.78; 95% CI, 1.44-2.20, P less than .01) and C. difficile infection (aRR 1.47; 95% CI, 1.17-1.85, P less than.01) continued to be associated with higher risk of VTE readmission.
Though they emphasized that the use of NRD data offered the impressive ability to “review over 15 million discharges across the U.S. annually,” Dr. Faye and coauthors acknowledged that their study did have limitations. These included the inability to verify via chart review the study’s outcomes and covariates. In addition, they were unable to assess potential contributing risk factors such as medication use, use of VTE prophylaxis during hospitalization, disease severity, and family history. Finally, though unlikely, they admitted the possibility that patients could be counted more than once if they were readmitted with a VTE each year of the study.
The authors reported being supported by grants from the National Institutes of Health and various pharmaceutical companies, as well as receiving honoraria and serving as consultants.
SOURCE: Faye AS et al. Clin Gastroenterol Hepatol. 2019 July 20. doi: 10.1016/j.cgh.2019.07.028.
FROM CLINICAL GASTROENTEROLOGY AND HEPATOLOGY
Key clinical point: Readmission for VTE in patients with inflammatory bowel diseases most often occurs within 60 days of discharge.
Major finding: The highest readmission risk was in patients between the ages of 66 and 80 (risk ratio 4.04; 95% confidence interval, 2.54-6.44, P less than .01).
Study details: A retrospective cohort study of 1,160 IBD patients who had VTE readmissions via 2010-2014 data from the Nationwide Readmissions Database.
Disclosures: The authors reported being supported by grants from the National Institutes of Health and various pharmaceutical companies, as well as receiving honoraria and serving as consultants.
Source: Faye AS et al. Clin Gastroenterol Hepatol. 2019 July 20. doi: 10.1016/j.cgh.2019.07.028.
Poll: How much has the price of insulin risen in the past 15 years?
Choose your answer in the poll below. To check the accuracy of your answer, see Endocrine Consult: 10 (Safe) Ways to Reduce Patients’ Insulin Costs.
[polldaddy:10400221]
Click on page 2 below to find out what the correct answer is...
The correct answer is d.) 500%
To learn more, see this month's Endocrine Consult: 10 (Safe) Ways to Reduce Patients’ Insulin Costs.
Choose your answer in the poll below. To check the accuracy of your answer, see Endocrine Consult: 10 (Safe) Ways to Reduce Patients’ Insulin Costs.
[polldaddy:10400221]
Click on page 2 below to find out what the correct answer is...
The correct answer is d.) 500%
To learn more, see this month's Endocrine Consult: 10 (Safe) Ways to Reduce Patients’ Insulin Costs.
Choose your answer in the poll below. To check the accuracy of your answer, see Endocrine Consult: 10 (Safe) Ways to Reduce Patients’ Insulin Costs.
[polldaddy:10400221]
Click on page 2 below to find out what the correct answer is...
The correct answer is d.) 500%
To learn more, see this month's Endocrine Consult: 10 (Safe) Ways to Reduce Patients’ Insulin Costs.
10 (Safe) Ways to Reduce Patients’ Insulin Costs
Almost a century after its discovery, insulin remains a life-saving yet costly medication: In the past 15 years, prices have risen more than 500%.1 Patients may ask you why the insulin you prescribe is so expensive, and the complex process for determining drug costs makes it difficult to answer. But the bottom line is, patients need their insulin—and they want it without breaking the bank.
Thankfully, there are several strategies for reducing the cost of insulin. First and foremost, patients must be advised that not taking their prescribed insulin, or taking less insulin than prescribed, is not a safe alternative. An individualized cost-benefit analysis between patient and provider can help to determine the best option for each patient. After working in endocrinology for 5 years, I have learned the following 10 ways to help patients whose financial situations limit their access to insulin.
1 Try older insulins, including mixed insulin 70/30 or 50/50, insulin NPH, or regular insulin. Because the beneficial effects may not be as long lasting with these as with newer insulins on the market, your patient may need to test glucose levels more frequently. Also, insulin NPH and any mixed insulins are suspensions, not solutions, so patients will need to gently roll older insulins prior to use. Those in pen form may also have a shorter shelf life.
2 Switch to a syringe and vial. Although dosing can be less precise, this could be a viable option for patients with good vision and dexterity. This method helps patients save in 3 ways: (1) the insulin is less expensive; (2) syringes generally cost less (about $30 for 100) than pen needle tips (about $50 for 100); and (3) vials of NPH are longer-lasting suspensions that are stable for about 28 days once opened, compared to 14 days for pens.2-4
3 Switch from a 30- to a 90-day supply of refills. This helps to lower copays. For example, a mail-order program (eg, Express Scripts) that ships from a warehouse typically offers lower pricing than a brick-and-mortar pharmacy with greater overhead. Many of these programs provide 2-pharmacist verification for accuracy and free home delivery of medications at a 10% discount, as well as 24-hour pharmacist access.5 The ease of obtaining prescriptions by this method also can help with medication adherence.
4 Patient assistance programs (PAPs) offered by insulin manufacturers can help lower costs for patients who find it difficult to afford their medication. Information on these programs is available on the respective company’s websites, usually in multiple languages (although some are limited to English and Spanish). Patients applying for a PAP must provide a proof of income and adhere to the program’s specific criteria. Renewal is typically required each year.6-8
5 Copay cards are available to many patients with private insurance and may help make insulin more affordable. Patients may be able to receive a $25 monthly supply of insulin for up to 1 year (specific terms vary). Maximum contributions and contributions toward deductibles also vary by program, so patients need to familiarize themselves with what their particular copay card allows. Generally, copay cards are not a sustainable long-term solution; for one thing, they expire, and for another, emphasis should be placed on affordable medications rather than affording expensive medications.
[polldaddy:10400221]
Continue to: 6 External PAPs for patients on Medicare...
6 External PAPs for patients on Medicare can help lower the costs of prescription medications.9 A database of pharmaceutical PAPs is available on the Medicare website.10 Some PAPs may help patients on Medicare pay through the $5,100 coverage gap or “donut hole”—a term referring to a gap in prescription drug coverage once patients have met their prescription limit (all Medicare part D plans have a donut hole).11,12 Patients and providers will need to read the fine print when applying for an external PAP, because some have a monthly or one-time start-up fee for processing the paperwork (and note, there is often paperwork for the relief program in addition to the PAP paperwork through the pharmaceutical company).
7 A Program of All-Inclusive Care for the Elderly (PACE) is available in many states; check medicare.gov to see if your state is eligible. For patients 55 and older on Medicare or Medicaid who do not opt for care at a nursing home facility, PACE may be able to provide care and coverage in the patient’s home or at a PACE facility. Services include primary care, hospital care, laboratory and x-ray services, medical specialty services, and prescription drugs. To be eligible for PACE services, the patient must live in the service area of a PACE organization and have a requirement for a nursing home-level of care (as certified by your state).
8 Shop around for the best deal. Encourage your patients to comparison shop for the best prices rather than accepting the first or only option at their usual pharmacy. Different pharmacies offer drugs at lower prices than competitors. Also, continually compare prices at GoodRx or HealthWarehouse.com. The latter—a fully licensed Internet-based pharmacy—sells FDA-approved medications at affordable prices in all 50 states, without the requirement for insurance coverage.
9 Use of a patch pump may be less expensive for patients with type 2 diabetes who are taking basal-bolus regimens. Patches slowly deliver single short-acting insulin (usually insulin aspart or lispro) that acts as a basal insulin, with an additional reservoir for prandial insulin at mealtime and for snacks. As there is a catheter in the patch, patients would not require the use of needles.13
10 Try removing mealtime insulin for patients with type 2 diabetes who need minimal mealtime insulin. Clinicians can initiate a safe trial of this removal by encouraging the patient to consume a low-carbohydrate diet, increase exercise, and/or use other noninsulin medications that are more affordable.
Continue to: The affordability of insulins...
The affordability of insulins is a potentially uncomfortable but necessary conversation to have with your patient. Providers are one of the best resources for patients who seek relief from financial difficulties. The recommendations discussed here can help providers and patients design a cost-conscious plan for insulin treatment. Although each recommendation is viable, the pros and cons must be weighed on a case-by-case basis. Providers and patients should also pay attention to the Senate Finance Committee’s ongoing discussions and possible resolutions that could result in lower insulin costs. Until legislation that lowers the prices of insulin comes to fruition, however, providers should continue to plan with their patients on how to best get their insulin at the lowest cost.
Test yourself with the poll here.
1. Grassley, Wyden launch bipartisan investigation into insulin prices. United States Senate Committee on Finance website. www.finance.senate.gov/chairmans-news/grassley-wyden-launch-bipartisan-investigation-into-insulin-prices. Published February 22, 2019. Accessed August 16, 2019.
2. BD Ultra-Fine. Syringe. GoodRx website. www.goodrx.com/bd-ultra-fine?dosage=31-gauge-5-16%22-of-1-cc&form=syringe&label_override=BD+Ultra-Fine&quantity=100. Accessed August 16, 2019.
3. BD Ultra-Fine. Pen needle. GoodRx website. www.goodrx.com/bd-ultra-fine?dosage=5-32%22-of-32-gauge&form=pen-needle&label_override=BD+Ultra-Fine&quantity=100. Accessed August 16, 2019.
4. Joffee D. Stability of common insulins in pens and vials. Diabetes in Control website. www.diabetesincontrol.com/wp-content/uploads/PDF/se_insulin_stability_chart.pdf. Published September 2011. Accessed August 16, 2019.
5. Frequently asked questions. Preferred home delivery program for maintenance medications. Express Scripts website. www.express-scripts.com/art/pdf/SST-custom-preferred-faq.pdf. Accessed August 16, 2019.
6. Patient Connection. Sanofi Patient Connection website. www.sanofipatientconnection.com/. Accessed August 16, 2019.
7. The Lilly Cares Foundation Patient Assistance Program. Lilly website. www.lillycares.com/assistanceprograms.aspx. Accessed August 16, 2019.
8. Novo Nordisk Patient Assistance Program. NovoCare website. www.novocare.com/psp/PAP.html. Accessed August 16, 2019.
9. 6 ways to get help with prescription costs. Medicare website. www.medicare.gov/drug-coverage-part-d/costs-for-medicare-drug-coverage/costs-in-the-coverage-gap/6-ways-to-get-help-with-prescription-costs. Accessed August 16, 2019.
10. Pharmaceutical assistance program. Medicare website. www.medicare.gov/pharmaceutical-assistance-program/Index.aspx. Accessed August 16, 2019.
11. Catastrophic coverage. Medicare website. www.medicare.gov/drug-coverage-part-d/costs-for-medicare-drug-coverage/catastrophic-coverage. Accessed August 16, 2019.
12. Costs in the coverage gap. Medicare website. www.medicare.gov/drug-coverage-part-d/costs-for-medicare-drug-coverage/costs-in-the-coverage-gap. Accessed August 16, 2019.
13. V-Go Reimbursement Assistance Program. V-Go website. www.go-vgo.com/coverage-savings/overview/. Accessed August 16, 2019.
Almost a century after its discovery, insulin remains a life-saving yet costly medication: In the past 15 years, prices have risen more than 500%.1 Patients may ask you why the insulin you prescribe is so expensive, and the complex process for determining drug costs makes it difficult to answer. But the bottom line is, patients need their insulin—and they want it without breaking the bank.
Thankfully, there are several strategies for reducing the cost of insulin. First and foremost, patients must be advised that not taking their prescribed insulin, or taking less insulin than prescribed, is not a safe alternative. An individualized cost-benefit analysis between patient and provider can help to determine the best option for each patient. After working in endocrinology for 5 years, I have learned the following 10 ways to help patients whose financial situations limit their access to insulin.
1 Try older insulins, including mixed insulin 70/30 or 50/50, insulin NPH, or regular insulin. Because the beneficial effects may not be as long lasting with these as with newer insulins on the market, your patient may need to test glucose levels more frequently. Also, insulin NPH and any mixed insulins are suspensions, not solutions, so patients will need to gently roll older insulins prior to use. Those in pen form may also have a shorter shelf life.
2 Switch to a syringe and vial. Although dosing can be less precise, this could be a viable option for patients with good vision and dexterity. This method helps patients save in 3 ways: (1) the insulin is less expensive; (2) syringes generally cost less (about $30 for 100) than pen needle tips (about $50 for 100); and (3) vials of NPH are longer-lasting suspensions that are stable for about 28 days once opened, compared to 14 days for pens.2-4
3 Switch from a 30- to a 90-day supply of refills. This helps to lower copays. For example, a mail-order program (eg, Express Scripts) that ships from a warehouse typically offers lower pricing than a brick-and-mortar pharmacy with greater overhead. Many of these programs provide 2-pharmacist verification for accuracy and free home delivery of medications at a 10% discount, as well as 24-hour pharmacist access.5 The ease of obtaining prescriptions by this method also can help with medication adherence.
4 Patient assistance programs (PAPs) offered by insulin manufacturers can help lower costs for patients who find it difficult to afford their medication. Information on these programs is available on the respective company’s websites, usually in multiple languages (although some are limited to English and Spanish). Patients applying for a PAP must provide a proof of income and adhere to the program’s specific criteria. Renewal is typically required each year.6-8
5 Copay cards are available to many patients with private insurance and may help make insulin more affordable. Patients may be able to receive a $25 monthly supply of insulin for up to 1 year (specific terms vary). Maximum contributions and contributions toward deductibles also vary by program, so patients need to familiarize themselves with what their particular copay card allows. Generally, copay cards are not a sustainable long-term solution; for one thing, they expire, and for another, emphasis should be placed on affordable medications rather than affording expensive medications.
[polldaddy:10400221]
Continue to: 6 External PAPs for patients on Medicare...
6 External PAPs for patients on Medicare can help lower the costs of prescription medications.9 A database of pharmaceutical PAPs is available on the Medicare website.10 Some PAPs may help patients on Medicare pay through the $5,100 coverage gap or “donut hole”—a term referring to a gap in prescription drug coverage once patients have met their prescription limit (all Medicare part D plans have a donut hole).11,12 Patients and providers will need to read the fine print when applying for an external PAP, because some have a monthly or one-time start-up fee for processing the paperwork (and note, there is often paperwork for the relief program in addition to the PAP paperwork through the pharmaceutical company).
7 A Program of All-Inclusive Care for the Elderly (PACE) is available in many states; check medicare.gov to see if your state is eligible. For patients 55 and older on Medicare or Medicaid who do not opt for care at a nursing home facility, PACE may be able to provide care and coverage in the patient’s home or at a PACE facility. Services include primary care, hospital care, laboratory and x-ray services, medical specialty services, and prescription drugs. To be eligible for PACE services, the patient must live in the service area of a PACE organization and have a requirement for a nursing home-level of care (as certified by your state).
8 Shop around for the best deal. Encourage your patients to comparison shop for the best prices rather than accepting the first or only option at their usual pharmacy. Different pharmacies offer drugs at lower prices than competitors. Also, continually compare prices at GoodRx or HealthWarehouse.com. The latter—a fully licensed Internet-based pharmacy—sells FDA-approved medications at affordable prices in all 50 states, without the requirement for insurance coverage.
9 Use of a patch pump may be less expensive for patients with type 2 diabetes who are taking basal-bolus regimens. Patches slowly deliver single short-acting insulin (usually insulin aspart or lispro) that acts as a basal insulin, with an additional reservoir for prandial insulin at mealtime and for snacks. As there is a catheter in the patch, patients would not require the use of needles.13
10 Try removing mealtime insulin for patients with type 2 diabetes who need minimal mealtime insulin. Clinicians can initiate a safe trial of this removal by encouraging the patient to consume a low-carbohydrate diet, increase exercise, and/or use other noninsulin medications that are more affordable.
Continue to: The affordability of insulins...
The affordability of insulins is a potentially uncomfortable but necessary conversation to have with your patient. Providers are one of the best resources for patients who seek relief from financial difficulties. The recommendations discussed here can help providers and patients design a cost-conscious plan for insulin treatment. Although each recommendation is viable, the pros and cons must be weighed on a case-by-case basis. Providers and patients should also pay attention to the Senate Finance Committee’s ongoing discussions and possible resolutions that could result in lower insulin costs. Until legislation that lowers the prices of insulin comes to fruition, however, providers should continue to plan with their patients on how to best get their insulin at the lowest cost.
Test yourself with the poll here.
Almost a century after its discovery, insulin remains a life-saving yet costly medication: In the past 15 years, prices have risen more than 500%.1 Patients may ask you why the insulin you prescribe is so expensive, and the complex process for determining drug costs makes it difficult to answer. But the bottom line is, patients need their insulin—and they want it without breaking the bank.
Thankfully, there are several strategies for reducing the cost of insulin. First and foremost, patients must be advised that not taking their prescribed insulin, or taking less insulin than prescribed, is not a safe alternative. An individualized cost-benefit analysis between patient and provider can help to determine the best option for each patient. After working in endocrinology for 5 years, I have learned the following 10 ways to help patients whose financial situations limit their access to insulin.
1 Try older insulins, including mixed insulin 70/30 or 50/50, insulin NPH, or regular insulin. Because the beneficial effects may not be as long lasting with these as with newer insulins on the market, your patient may need to test glucose levels more frequently. Also, insulin NPH and any mixed insulins are suspensions, not solutions, so patients will need to gently roll older insulins prior to use. Those in pen form may also have a shorter shelf life.
2 Switch to a syringe and vial. Although dosing can be less precise, this could be a viable option for patients with good vision and dexterity. This method helps patients save in 3 ways: (1) the insulin is less expensive; (2) syringes generally cost less (about $30 for 100) than pen needle tips (about $50 for 100); and (3) vials of NPH are longer-lasting suspensions that are stable for about 28 days once opened, compared to 14 days for pens.2-4
3 Switch from a 30- to a 90-day supply of refills. This helps to lower copays. For example, a mail-order program (eg, Express Scripts) that ships from a warehouse typically offers lower pricing than a brick-and-mortar pharmacy with greater overhead. Many of these programs provide 2-pharmacist verification for accuracy and free home delivery of medications at a 10% discount, as well as 24-hour pharmacist access.5 The ease of obtaining prescriptions by this method also can help with medication adherence.
4 Patient assistance programs (PAPs) offered by insulin manufacturers can help lower costs for patients who find it difficult to afford their medication. Information on these programs is available on the respective company’s websites, usually in multiple languages (although some are limited to English and Spanish). Patients applying for a PAP must provide a proof of income and adhere to the program’s specific criteria. Renewal is typically required each year.6-8
5 Copay cards are available to many patients with private insurance and may help make insulin more affordable. Patients may be able to receive a $25 monthly supply of insulin for up to 1 year (specific terms vary). Maximum contributions and contributions toward deductibles also vary by program, so patients need to familiarize themselves with what their particular copay card allows. Generally, copay cards are not a sustainable long-term solution; for one thing, they expire, and for another, emphasis should be placed on affordable medications rather than affording expensive medications.
[polldaddy:10400221]
Continue to: 6 External PAPs for patients on Medicare...
6 External PAPs for patients on Medicare can help lower the costs of prescription medications.9 A database of pharmaceutical PAPs is available on the Medicare website.10 Some PAPs may help patients on Medicare pay through the $5,100 coverage gap or “donut hole”—a term referring to a gap in prescription drug coverage once patients have met their prescription limit (all Medicare part D plans have a donut hole).11,12 Patients and providers will need to read the fine print when applying for an external PAP, because some have a monthly or one-time start-up fee for processing the paperwork (and note, there is often paperwork for the relief program in addition to the PAP paperwork through the pharmaceutical company).
7 A Program of All-Inclusive Care for the Elderly (PACE) is available in many states; check medicare.gov to see if your state is eligible. For patients 55 and older on Medicare or Medicaid who do not opt for care at a nursing home facility, PACE may be able to provide care and coverage in the patient’s home or at a PACE facility. Services include primary care, hospital care, laboratory and x-ray services, medical specialty services, and prescription drugs. To be eligible for PACE services, the patient must live in the service area of a PACE organization and have a requirement for a nursing home-level of care (as certified by your state).
8 Shop around for the best deal. Encourage your patients to comparison shop for the best prices rather than accepting the first or only option at their usual pharmacy. Different pharmacies offer drugs at lower prices than competitors. Also, continually compare prices at GoodRx or HealthWarehouse.com. The latter—a fully licensed Internet-based pharmacy—sells FDA-approved medications at affordable prices in all 50 states, without the requirement for insurance coverage.
9 Use of a patch pump may be less expensive for patients with type 2 diabetes who are taking basal-bolus regimens. Patches slowly deliver single short-acting insulin (usually insulin aspart or lispro) that acts as a basal insulin, with an additional reservoir for prandial insulin at mealtime and for snacks. As there is a catheter in the patch, patients would not require the use of needles.13
10 Try removing mealtime insulin for patients with type 2 diabetes who need minimal mealtime insulin. Clinicians can initiate a safe trial of this removal by encouraging the patient to consume a low-carbohydrate diet, increase exercise, and/or use other noninsulin medications that are more affordable.
Continue to: The affordability of insulins...
The affordability of insulins is a potentially uncomfortable but necessary conversation to have with your patient. Providers are one of the best resources for patients who seek relief from financial difficulties. The recommendations discussed here can help providers and patients design a cost-conscious plan for insulin treatment. Although each recommendation is viable, the pros and cons must be weighed on a case-by-case basis. Providers and patients should also pay attention to the Senate Finance Committee’s ongoing discussions and possible resolutions that could result in lower insulin costs. Until legislation that lowers the prices of insulin comes to fruition, however, providers should continue to plan with their patients on how to best get their insulin at the lowest cost.
Test yourself with the poll here.
1. Grassley, Wyden launch bipartisan investigation into insulin prices. United States Senate Committee on Finance website. www.finance.senate.gov/chairmans-news/grassley-wyden-launch-bipartisan-investigation-into-insulin-prices. Published February 22, 2019. Accessed August 16, 2019.
2. BD Ultra-Fine. Syringe. GoodRx website. www.goodrx.com/bd-ultra-fine?dosage=31-gauge-5-16%22-of-1-cc&form=syringe&label_override=BD+Ultra-Fine&quantity=100. Accessed August 16, 2019.
3. BD Ultra-Fine. Pen needle. GoodRx website. www.goodrx.com/bd-ultra-fine?dosage=5-32%22-of-32-gauge&form=pen-needle&label_override=BD+Ultra-Fine&quantity=100. Accessed August 16, 2019.
4. Joffee D. Stability of common insulins in pens and vials. Diabetes in Control website. www.diabetesincontrol.com/wp-content/uploads/PDF/se_insulin_stability_chart.pdf. Published September 2011. Accessed August 16, 2019.
5. Frequently asked questions. Preferred home delivery program for maintenance medications. Express Scripts website. www.express-scripts.com/art/pdf/SST-custom-preferred-faq.pdf. Accessed August 16, 2019.
6. Patient Connection. Sanofi Patient Connection website. www.sanofipatientconnection.com/. Accessed August 16, 2019.
7. The Lilly Cares Foundation Patient Assistance Program. Lilly website. www.lillycares.com/assistanceprograms.aspx. Accessed August 16, 2019.
8. Novo Nordisk Patient Assistance Program. NovoCare website. www.novocare.com/psp/PAP.html. Accessed August 16, 2019.
9. 6 ways to get help with prescription costs. Medicare website. www.medicare.gov/drug-coverage-part-d/costs-for-medicare-drug-coverage/costs-in-the-coverage-gap/6-ways-to-get-help-with-prescription-costs. Accessed August 16, 2019.
10. Pharmaceutical assistance program. Medicare website. www.medicare.gov/pharmaceutical-assistance-program/Index.aspx. Accessed August 16, 2019.
11. Catastrophic coverage. Medicare website. www.medicare.gov/drug-coverage-part-d/costs-for-medicare-drug-coverage/catastrophic-coverage. Accessed August 16, 2019.
12. Costs in the coverage gap. Medicare website. www.medicare.gov/drug-coverage-part-d/costs-for-medicare-drug-coverage/costs-in-the-coverage-gap. Accessed August 16, 2019.
13. V-Go Reimbursement Assistance Program. V-Go website. www.go-vgo.com/coverage-savings/overview/. Accessed August 16, 2019.
1. Grassley, Wyden launch bipartisan investigation into insulin prices. United States Senate Committee on Finance website. www.finance.senate.gov/chairmans-news/grassley-wyden-launch-bipartisan-investigation-into-insulin-prices. Published February 22, 2019. Accessed August 16, 2019.
2. BD Ultra-Fine. Syringe. GoodRx website. www.goodrx.com/bd-ultra-fine?dosage=31-gauge-5-16%22-of-1-cc&form=syringe&label_override=BD+Ultra-Fine&quantity=100. Accessed August 16, 2019.
3. BD Ultra-Fine. Pen needle. GoodRx website. www.goodrx.com/bd-ultra-fine?dosage=5-32%22-of-32-gauge&form=pen-needle&label_override=BD+Ultra-Fine&quantity=100. Accessed August 16, 2019.
4. Joffee D. Stability of common insulins in pens and vials. Diabetes in Control website. www.diabetesincontrol.com/wp-content/uploads/PDF/se_insulin_stability_chart.pdf. Published September 2011. Accessed August 16, 2019.
5. Frequently asked questions. Preferred home delivery program for maintenance medications. Express Scripts website. www.express-scripts.com/art/pdf/SST-custom-preferred-faq.pdf. Accessed August 16, 2019.
6. Patient Connection. Sanofi Patient Connection website. www.sanofipatientconnection.com/. Accessed August 16, 2019.
7. The Lilly Cares Foundation Patient Assistance Program. Lilly website. www.lillycares.com/assistanceprograms.aspx. Accessed August 16, 2019.
8. Novo Nordisk Patient Assistance Program. NovoCare website. www.novocare.com/psp/PAP.html. Accessed August 16, 2019.
9. 6 ways to get help with prescription costs. Medicare website. www.medicare.gov/drug-coverage-part-d/costs-for-medicare-drug-coverage/costs-in-the-coverage-gap/6-ways-to-get-help-with-prescription-costs. Accessed August 16, 2019.
10. Pharmaceutical assistance program. Medicare website. www.medicare.gov/pharmaceutical-assistance-program/Index.aspx. Accessed August 16, 2019.
11. Catastrophic coverage. Medicare website. www.medicare.gov/drug-coverage-part-d/costs-for-medicare-drug-coverage/catastrophic-coverage. Accessed August 16, 2019.
12. Costs in the coverage gap. Medicare website. www.medicare.gov/drug-coverage-part-d/costs-for-medicare-drug-coverage/costs-in-the-coverage-gap. Accessed August 16, 2019.
13. V-Go Reimbursement Assistance Program. V-Go website. www.go-vgo.com/coverage-savings/overview/. Accessed August 16, 2019.
Pseudo-Ludwig angina
An 83-year-old woman with hypertension, hypothyroidism, and a history of depression presented to the emergency department with acute shortness of breath and hypoxia. She was found to have submassive pulmonary embolism, and a heparin infusion was started immediately.
Urgent nasopharyngeal laryngoscopy revealed a hematoma at the base of her tongue that extended into the vallecula, piriform sinuses, and aryepiglottic fold, causing acute airway obstruction. These features combined with the supratherapeutic aPTT led to the diagnosis of pseudo-Ludwig angina.
DANGER OF RAPID AIRWAY COMPROMISE
Pseudo-Ludwig angina is a rare condition in which over-anticoagulation causes sublingual swelling leading to airway obstruction, whereas true Ludwig angina is an infectious regional suppuration of the neck.
Most reported cases of pseudo-Ludwig angina have resulted from overanticogulation with warfarin or warfarin-like substances (rodenticides), or from coagulopathy due to liver disease.1–3 Early recognition is essential to avoid airway compromise.
In our patient, all anticoagulation was discontinued, and she was intubated until the hematoma began to resolve, the aPTT returned to normal, and respiratory compromise improved. At follow-up 2 months later, the sublingual hematoma had completely resolved (Figure 1). And at a 6-month follow-up visit, the pulmonary embolism had resolved, and pulmonary pressures by 2-dimensional echocardiography were normal.
- Lovallo E, Patterson S, Erickson M, Chin C, Blanc P, Durrani TS. When is “pseudo-Ludwig’s angina” associated with coagulopathy also a “pseudo” hemorrhage? J Investig Med High Impact Case Rep 2013; 1(2):2324709613492503. doi:10.1177/2324709613492503
- Smith RG, Parker TJ, Anderson TA. Noninfectious acute upper airway obstruction (pseudo-Ludwig phenomenon): report of a case. J Oral Maxillofac Surg 1987; 45(8):701–704. pmid:3475442
- Zacharia GS, Kandiyil S, Thomas V. Pseudo-Ludwig's phenomenon: a rare clinical manifestation in liver cirrhosis. ACG Case Rep J 2014; 2(1):53–54. doi:10.14309/crj.2014.83
An 83-year-old woman with hypertension, hypothyroidism, and a history of depression presented to the emergency department with acute shortness of breath and hypoxia. She was found to have submassive pulmonary embolism, and a heparin infusion was started immediately.
Urgent nasopharyngeal laryngoscopy revealed a hematoma at the base of her tongue that extended into the vallecula, piriform sinuses, and aryepiglottic fold, causing acute airway obstruction. These features combined with the supratherapeutic aPTT led to the diagnosis of pseudo-Ludwig angina.
DANGER OF RAPID AIRWAY COMPROMISE
Pseudo-Ludwig angina is a rare condition in which over-anticoagulation causes sublingual swelling leading to airway obstruction, whereas true Ludwig angina is an infectious regional suppuration of the neck.
Most reported cases of pseudo-Ludwig angina have resulted from overanticogulation with warfarin or warfarin-like substances (rodenticides), or from coagulopathy due to liver disease.1–3 Early recognition is essential to avoid airway compromise.
In our patient, all anticoagulation was discontinued, and she was intubated until the hematoma began to resolve, the aPTT returned to normal, and respiratory compromise improved. At follow-up 2 months later, the sublingual hematoma had completely resolved (Figure 1). And at a 6-month follow-up visit, the pulmonary embolism had resolved, and pulmonary pressures by 2-dimensional echocardiography were normal.
An 83-year-old woman with hypertension, hypothyroidism, and a history of depression presented to the emergency department with acute shortness of breath and hypoxia. She was found to have submassive pulmonary embolism, and a heparin infusion was started immediately.
Urgent nasopharyngeal laryngoscopy revealed a hematoma at the base of her tongue that extended into the vallecula, piriform sinuses, and aryepiglottic fold, causing acute airway obstruction. These features combined with the supratherapeutic aPTT led to the diagnosis of pseudo-Ludwig angina.
DANGER OF RAPID AIRWAY COMPROMISE
Pseudo-Ludwig angina is a rare condition in which over-anticoagulation causes sublingual swelling leading to airway obstruction, whereas true Ludwig angina is an infectious regional suppuration of the neck.
Most reported cases of pseudo-Ludwig angina have resulted from overanticogulation with warfarin or warfarin-like substances (rodenticides), or from coagulopathy due to liver disease.1–3 Early recognition is essential to avoid airway compromise.
In our patient, all anticoagulation was discontinued, and she was intubated until the hematoma began to resolve, the aPTT returned to normal, and respiratory compromise improved. At follow-up 2 months later, the sublingual hematoma had completely resolved (Figure 1). And at a 6-month follow-up visit, the pulmonary embolism had resolved, and pulmonary pressures by 2-dimensional echocardiography were normal.
- Lovallo E, Patterson S, Erickson M, Chin C, Blanc P, Durrani TS. When is “pseudo-Ludwig’s angina” associated with coagulopathy also a “pseudo” hemorrhage? J Investig Med High Impact Case Rep 2013; 1(2):2324709613492503. doi:10.1177/2324709613492503
- Smith RG, Parker TJ, Anderson TA. Noninfectious acute upper airway obstruction (pseudo-Ludwig phenomenon): report of a case. J Oral Maxillofac Surg 1987; 45(8):701–704. pmid:3475442
- Zacharia GS, Kandiyil S, Thomas V. Pseudo-Ludwig's phenomenon: a rare clinical manifestation in liver cirrhosis. ACG Case Rep J 2014; 2(1):53–54. doi:10.14309/crj.2014.83
- Lovallo E, Patterson S, Erickson M, Chin C, Blanc P, Durrani TS. When is “pseudo-Ludwig’s angina” associated with coagulopathy also a “pseudo” hemorrhage? J Investig Med High Impact Case Rep 2013; 1(2):2324709613492503. doi:10.1177/2324709613492503
- Smith RG, Parker TJ, Anderson TA. Noninfectious acute upper airway obstruction (pseudo-Ludwig phenomenon): report of a case. J Oral Maxillofac Surg 1987; 45(8):701–704. pmid:3475442
- Zacharia GS, Kandiyil S, Thomas V. Pseudo-Ludwig's phenomenon: a rare clinical manifestation in liver cirrhosis. ACG Case Rep J 2014; 2(1):53–54. doi:10.14309/crj.2014.83