User login
Nail biopsies made simple
CHICAGO – Maral Skelsey, MD, doesn’t get flowers from her patients very often. But, she said, a big bouquet recently landed on her desk after she had performed a nail biopsy on a patient. The note from the patient read, “That wasn’t as bad as I thought it would be!”
The patient’s relief after the procedure highlights the apprehension that both patients and dermatologists can feel when a nail biopsy becomes necessary, said Dr. Skelsey, director of dermatologic surgery at Georgetown University, Washington, D.C.
Speaking at the summer meeting of the American Academy of Dermatology, Dr. Skelsey said that the most important advice she can give about the nail biopsy is, “Do it early and often.”
Dr. Skelsey reminded the audience that the musician Bob Marley died of malignant melanoma; the first sign of his cancer was a longitudinal melanonychia that went unbiopsied. “The biggest mistake we make is not doing it,” she said.
In performing a nail biopsy, said Dr. Skelsey, the goals are, first and foremost, to optimize the pathologic diagnosis. Correct technique can help avoid complications such as bleeding, infection, and nail dystrophy; the right approach can minimize pain and anxiety, she added.
In preparing for a biopsy for melanonychia, “dermoscopy can be very helpful” in assessing the location of the pigment and fine-tuning planning for the biopsy, said Dr. Skelsey. Also, if the streak of melanonychia has reached the distal nail, sending the clipping for pathology can be useful as well.
For dorsal pigmentation, the proximal nail matrix should be biopsied.
“Do not use a punch biopsy on the nail fold to diagnose melanoma – you will get a false negative,” Dr. Skelsey said. It’s not possible to get an accurate diagnosis going through the nail plate to the nail bed, she said.
The preoperative assessment is usually straightforward. Pertinent items in the patient’s history include any medication allergies, current anticoagulation, and any history of prior trauma to the digit to be biopsied. Occasionally, imaging may be helpful, and patients should always be assessed for vascular insufficiency, she noted.
Preoperatively, she asks her patients to remove nail polish and pretreat the area with povidone iodine for 2 days prior to the procedure. Patients need to have a ride home after the procedure, and should be prepared to elevate the affected extremity for 48 hours post procedure. If a toenail is biopsied, they’re advised to come with a postop shoe.
Her patients receive a 5-minute isopropyl alcohol wash of the area to be biopsied just before the procedure, followed by air drying and a 5-minute scrub with 7.5% povidone iodine, which then is wiped off preprocedure.
For hemostasis, a tourniquet can be improvised with a sterile glove finger and a hemostat; there are also dedicated finger cots available that work well for this purpose, she said. In addition to nail nippers and a nail elevator, an English nail splitter can be helpful, said Dr. Skelsey.
For anesthesia, she said she ordinarily uses a 30 gauge needle with buffered lidocaine and epinephrine at room temperature to deliver a wing block. Beginning about 1 cm proximal and lateral to the junction of the proximal and lateral nail fold, the dermatologist can slowly inject about 1.5 cc per side. As the block takes effect, the lateral nail fold will blanch distally in a wing-shaped pattern. This technique, she said, also has the benefit of acting as a volumetric tourniquet.
“To avulse or not to avulse?” asked Dr. Skelsey. “I used to avulse almost everything,” she said, but noted that a complete avulsion is a “pretty traumatic” procedure. Now, unless a full avulsion is required for complete and accurate pathology, she will usually perform a partial nail plate avulsion.
A partial avulsion can reduce pain and morbidity, and can be done by two different methods: the partial proximal avulsion, and the “trap door” avulsion. In a trap door avulsion, she said, the distal matrix is primarily visualized, so this may be a good option for a longitudinal melanonychia arising from the distal matrix. A Freer elevator is used to detach the nail plate from the bed and the matrix, after which the nail plate can be lifted with a hemostat.
In a partial proximal avulsion, the proximal nail fold is reflected, so it’s a better option when the proximal nail matrix needs evaluation, she said.
After the avulsion has been done, “the matrix has been exposed. Now what? Punch or shave?” asked Dr. Skelsey. She noted that she used to perform punch biopsies on “everything,” and that it’s a good option if the pigmented area spans 3 mm or less. One issue, though, is that the specimen can get stuck in the puncher, and extraction can make it difficult to deliver an intact specimen.
Shave biopsies, Dr. Skelsey said, are effective in dealing with nail matrix lesions. They can yield an accurate pathologic diagnosis, and the biopsied digits healed without nail dystrophy in about three quarters of the cases in one study, she said. Potential recurrence of pigmentation is one drawback of the shave technique, she said.
With a shave biopsy, she performs tangential incisions of the proximal and lateral nail folds, and scores and reflects the nail. Then, the band of pigment is shaved tangentially. She cauterizes the area, and sometimes will use a bit of an absorbable gelatin sponge (Gelfoam) as well. Then the proximal nail fold and nail plate are sutured.
Replacing the nail plate results in better cosmesis and is much more comfortable for the patient, she said. An 18-gauge needle can be used to bore a hole through the avulsed nail plate, which may be held in an antiseptic solution soak during the biopsy. The sutures should then be placed from skin to nail plate, so nail fragments aren’t driven into the skin during the suturing process. Finally, specimen margins should be inked, and separate labeled formalin jars are needed for the nail plate, nail bed, and the matrix.
Dr. Skelsey reported that she had no conflicts of interest.
[email protected]
On Twitter @karioakes
CHICAGO – Maral Skelsey, MD, doesn’t get flowers from her patients very often. But, she said, a big bouquet recently landed on her desk after she had performed a nail biopsy on a patient. The note from the patient read, “That wasn’t as bad as I thought it would be!”
The patient’s relief after the procedure highlights the apprehension that both patients and dermatologists can feel when a nail biopsy becomes necessary, said Dr. Skelsey, director of dermatologic surgery at Georgetown University, Washington, D.C.
Speaking at the summer meeting of the American Academy of Dermatology, Dr. Skelsey said that the most important advice she can give about the nail biopsy is, “Do it early and often.”
Dr. Skelsey reminded the audience that the musician Bob Marley died of malignant melanoma; the first sign of his cancer was a longitudinal melanonychia that went unbiopsied. “The biggest mistake we make is not doing it,” she said.
In performing a nail biopsy, said Dr. Skelsey, the goals are, first and foremost, to optimize the pathologic diagnosis. Correct technique can help avoid complications such as bleeding, infection, and nail dystrophy; the right approach can minimize pain and anxiety, she added.
In preparing for a biopsy for melanonychia, “dermoscopy can be very helpful” in assessing the location of the pigment and fine-tuning planning for the biopsy, said Dr. Skelsey. Also, if the streak of melanonychia has reached the distal nail, sending the clipping for pathology can be useful as well.
For dorsal pigmentation, the proximal nail matrix should be biopsied.
“Do not use a punch biopsy on the nail fold to diagnose melanoma – you will get a false negative,” Dr. Skelsey said. It’s not possible to get an accurate diagnosis going through the nail plate to the nail bed, she said.
The preoperative assessment is usually straightforward. Pertinent items in the patient’s history include any medication allergies, current anticoagulation, and any history of prior trauma to the digit to be biopsied. Occasionally, imaging may be helpful, and patients should always be assessed for vascular insufficiency, she noted.
Preoperatively, she asks her patients to remove nail polish and pretreat the area with povidone iodine for 2 days prior to the procedure. Patients need to have a ride home after the procedure, and should be prepared to elevate the affected extremity for 48 hours post procedure. If a toenail is biopsied, they’re advised to come with a postop shoe.
Her patients receive a 5-minute isopropyl alcohol wash of the area to be biopsied just before the procedure, followed by air drying and a 5-minute scrub with 7.5% povidone iodine, which then is wiped off preprocedure.
For hemostasis, a tourniquet can be improvised with a sterile glove finger and a hemostat; there are also dedicated finger cots available that work well for this purpose, she said. In addition to nail nippers and a nail elevator, an English nail splitter can be helpful, said Dr. Skelsey.
For anesthesia, she said she ordinarily uses a 30 gauge needle with buffered lidocaine and epinephrine at room temperature to deliver a wing block. Beginning about 1 cm proximal and lateral to the junction of the proximal and lateral nail fold, the dermatologist can slowly inject about 1.5 cc per side. As the block takes effect, the lateral nail fold will blanch distally in a wing-shaped pattern. This technique, she said, also has the benefit of acting as a volumetric tourniquet.
“To avulse or not to avulse?” asked Dr. Skelsey. “I used to avulse almost everything,” she said, but noted that a complete avulsion is a “pretty traumatic” procedure. Now, unless a full avulsion is required for complete and accurate pathology, she will usually perform a partial nail plate avulsion.
A partial avulsion can reduce pain and morbidity, and can be done by two different methods: the partial proximal avulsion, and the “trap door” avulsion. In a trap door avulsion, she said, the distal matrix is primarily visualized, so this may be a good option for a longitudinal melanonychia arising from the distal matrix. A Freer elevator is used to detach the nail plate from the bed and the matrix, after which the nail plate can be lifted with a hemostat.
In a partial proximal avulsion, the proximal nail fold is reflected, so it’s a better option when the proximal nail matrix needs evaluation, she said.
After the avulsion has been done, “the matrix has been exposed. Now what? Punch or shave?” asked Dr. Skelsey. She noted that she used to perform punch biopsies on “everything,” and that it’s a good option if the pigmented area spans 3 mm or less. One issue, though, is that the specimen can get stuck in the puncher, and extraction can make it difficult to deliver an intact specimen.
Shave biopsies, Dr. Skelsey said, are effective in dealing with nail matrix lesions. They can yield an accurate pathologic diagnosis, and the biopsied digits healed without nail dystrophy in about three quarters of the cases in one study, she said. Potential recurrence of pigmentation is one drawback of the shave technique, she said.
With a shave biopsy, she performs tangential incisions of the proximal and lateral nail folds, and scores and reflects the nail. Then, the band of pigment is shaved tangentially. She cauterizes the area, and sometimes will use a bit of an absorbable gelatin sponge (Gelfoam) as well. Then the proximal nail fold and nail plate are sutured.
Replacing the nail plate results in better cosmesis and is much more comfortable for the patient, she said. An 18-gauge needle can be used to bore a hole through the avulsed nail plate, which may be held in an antiseptic solution soak during the biopsy. The sutures should then be placed from skin to nail plate, so nail fragments aren’t driven into the skin during the suturing process. Finally, specimen margins should be inked, and separate labeled formalin jars are needed for the nail plate, nail bed, and the matrix.
Dr. Skelsey reported that she had no conflicts of interest.
[email protected]
On Twitter @karioakes
CHICAGO – Maral Skelsey, MD, doesn’t get flowers from her patients very often. But, she said, a big bouquet recently landed on her desk after she had performed a nail biopsy on a patient. The note from the patient read, “That wasn’t as bad as I thought it would be!”
The patient’s relief after the procedure highlights the apprehension that both patients and dermatologists can feel when a nail biopsy becomes necessary, said Dr. Skelsey, director of dermatologic surgery at Georgetown University, Washington, D.C.
Speaking at the summer meeting of the American Academy of Dermatology, Dr. Skelsey said that the most important advice she can give about the nail biopsy is, “Do it early and often.”
Dr. Skelsey reminded the audience that the musician Bob Marley died of malignant melanoma; the first sign of his cancer was a longitudinal melanonychia that went unbiopsied. “The biggest mistake we make is not doing it,” she said.
In performing a nail biopsy, said Dr. Skelsey, the goals are, first and foremost, to optimize the pathologic diagnosis. Correct technique can help avoid complications such as bleeding, infection, and nail dystrophy; the right approach can minimize pain and anxiety, she added.
In preparing for a biopsy for melanonychia, “dermoscopy can be very helpful” in assessing the location of the pigment and fine-tuning planning for the biopsy, said Dr. Skelsey. Also, if the streak of melanonychia has reached the distal nail, sending the clipping for pathology can be useful as well.
For dorsal pigmentation, the proximal nail matrix should be biopsied.
“Do not use a punch biopsy on the nail fold to diagnose melanoma – you will get a false negative,” Dr. Skelsey said. It’s not possible to get an accurate diagnosis going through the nail plate to the nail bed, she said.
The preoperative assessment is usually straightforward. Pertinent items in the patient’s history include any medication allergies, current anticoagulation, and any history of prior trauma to the digit to be biopsied. Occasionally, imaging may be helpful, and patients should always be assessed for vascular insufficiency, she noted.
Preoperatively, she asks her patients to remove nail polish and pretreat the area with povidone iodine for 2 days prior to the procedure. Patients need to have a ride home after the procedure, and should be prepared to elevate the affected extremity for 48 hours post procedure. If a toenail is biopsied, they’re advised to come with a postop shoe.
Her patients receive a 5-minute isopropyl alcohol wash of the area to be biopsied just before the procedure, followed by air drying and a 5-minute scrub with 7.5% povidone iodine, which then is wiped off preprocedure.
For hemostasis, a tourniquet can be improvised with a sterile glove finger and a hemostat; there are also dedicated finger cots available that work well for this purpose, she said. In addition to nail nippers and a nail elevator, an English nail splitter can be helpful, said Dr. Skelsey.
For anesthesia, she said she ordinarily uses a 30 gauge needle with buffered lidocaine and epinephrine at room temperature to deliver a wing block. Beginning about 1 cm proximal and lateral to the junction of the proximal and lateral nail fold, the dermatologist can slowly inject about 1.5 cc per side. As the block takes effect, the lateral nail fold will blanch distally in a wing-shaped pattern. This technique, she said, also has the benefit of acting as a volumetric tourniquet.
“To avulse or not to avulse?” asked Dr. Skelsey. “I used to avulse almost everything,” she said, but noted that a complete avulsion is a “pretty traumatic” procedure. Now, unless a full avulsion is required for complete and accurate pathology, she will usually perform a partial nail plate avulsion.
A partial avulsion can reduce pain and morbidity, and can be done by two different methods: the partial proximal avulsion, and the “trap door” avulsion. In a trap door avulsion, she said, the distal matrix is primarily visualized, so this may be a good option for a longitudinal melanonychia arising from the distal matrix. A Freer elevator is used to detach the nail plate from the bed and the matrix, after which the nail plate can be lifted with a hemostat.
In a partial proximal avulsion, the proximal nail fold is reflected, so it’s a better option when the proximal nail matrix needs evaluation, she said.
After the avulsion has been done, “the matrix has been exposed. Now what? Punch or shave?” asked Dr. Skelsey. She noted that she used to perform punch biopsies on “everything,” and that it’s a good option if the pigmented area spans 3 mm or less. One issue, though, is that the specimen can get stuck in the puncher, and extraction can make it difficult to deliver an intact specimen.
Shave biopsies, Dr. Skelsey said, are effective in dealing with nail matrix lesions. They can yield an accurate pathologic diagnosis, and the biopsied digits healed without nail dystrophy in about three quarters of the cases in one study, she said. Potential recurrence of pigmentation is one drawback of the shave technique, she said.
With a shave biopsy, she performs tangential incisions of the proximal and lateral nail folds, and scores and reflects the nail. Then, the band of pigment is shaved tangentially. She cauterizes the area, and sometimes will use a bit of an absorbable gelatin sponge (Gelfoam) as well. Then the proximal nail fold and nail plate are sutured.
Replacing the nail plate results in better cosmesis and is much more comfortable for the patient, she said. An 18-gauge needle can be used to bore a hole through the avulsed nail plate, which may be held in an antiseptic solution soak during the biopsy. The sutures should then be placed from skin to nail plate, so nail fragments aren’t driven into the skin during the suturing process. Finally, specimen margins should be inked, and separate labeled formalin jars are needed for the nail plate, nail bed, and the matrix.
Dr. Skelsey reported that she had no conflicts of interest.
[email protected]
On Twitter @karioakes
AT THE 2017 AAD SUMMER MEETING
Clinical Trial Designs for Topical Antifungal Treatments of Onychomycosis and Implications on Clinical Practice
Onychomycosis is a fungal nail infection primarily caused by dermatophytes.1 If left untreated, the infection can cause nail destruction and deformities,1 resulting in pain and discomfort,2 impaired foot mobility,3 and an overall reduced quality of life.1 Onychomycosis is a chronic condition that requires long treatment periods due to the slow growth rates of toenails.1 To successfully cure the condition, fungal eradication must be achieved.
Prior to the US Food and Drug Administration (FDA) approval of tavaborole and efinaconazole, ciclopirox was the only approved topical treatment for onychomycosis.4 The recent approval of tavaborole and efinaconazole has increased treatment options available to patients and has started to pave the way for future topical treatments. This article discusses the 3 approved topical treatments for onychomycosis and focuses on the design of the phase 3 clinical trials that led to their approval.
Topical Agents Used to Treat Onychomycosis
Tavaborole, efinaconazole, and ciclopirox have undergone extensive clinical investigation to receive FDA approval. Results from pivotal phase 3 studies establishing the efficacy and safety of each agent formed the basis for regulatory submission. Although it may seem intuitive to compare the relative performance of these agents based on their respective phase 3 clinical trial data, there are important differences in study methodology, conduct, and populations that prevent direct comparisons. The FDA provides limited guidance to the pharmaceutical industry on how to conduct clinical trials for potential onychomycosis treatments. Comparative efficacy and safety claims are limited based on cross-study comparisons. The details of the phase 3 trial designs are summarized in the Table.
Tavaborole
Tavaborole is a boron-based treatment with a novel mechanism of action.5 Tavaborole binds to the editing domain of leucyl–transfer ribonucleic acid synthetase via an integrated boron atom and inhibits fungal protein synthesis.6 Two identical randomized, double-blind, vehicle-controlled, parallel-group, phase 3 clinical trials evaluating tavaborole were performed.5 The first study (registered at www.clinicaltrials.gov with the identifier NCT01270971) included 594 participants from27 sites in the United States and Mexico and was conducted between December 2010 and November 2012. The second study (NCT01302119) included 604 participants from 32 sites in the United States and Canada and was conducted between February 2011 and January 2013.
Eligible participants 18 years and older had distal subungual onychomycosis (DSO) of the toenails affecting 20% to 60% of 1 or more target great toenails (TGTs), tested positive for fungus using potassium hydroxide (KOH) wet mounts and positive for Trichophyton rubrum and Trichophyton mentagrophytes on fungal culture diagnostic tests, had distal TGT thickness of 3 mm or less, and had 3 mm or more of clear nail between the proximal nail fold and the most proximal visible mycotic border.5 Those with active tinea pedis requiring treatment or with a history of chronic moccasin-type tinea pedis were excluded. Participants were randomized to receive either tavaborole or vehicle (2:1). Treatments were applied once daily to all infected toenails for a total of 48 weeks, and nail debridement (defined as partial or complete removal of the toenail) was not permitted. Notably, controlled trimming of the nail was allowed to 1 mm of the leading nail edge. Regular assessments of each toenail for disease involvement, onycholysis, and subungual hyperkeratosis were made at screening, baseline, week 2, week 6, and every 6 weeks thereafter until week 52. Subungual TGT samples were taken at screening and every 12 weeks during the study for examination at a mycology laboratory, which performed KOH and fungal culture tests. A follow-up assessment was made at week 52.5
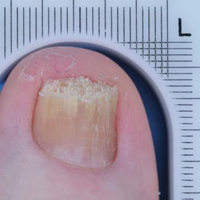
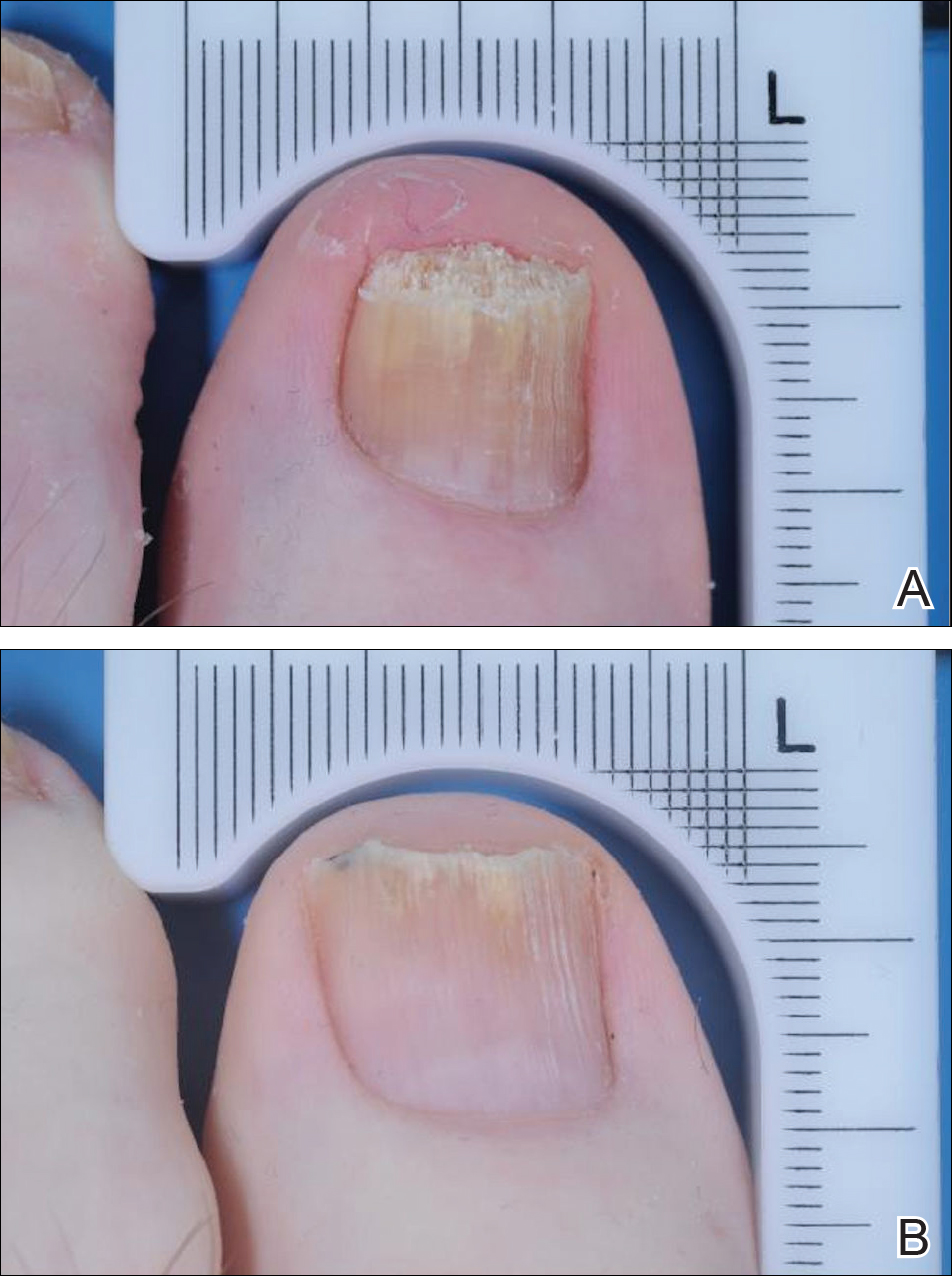
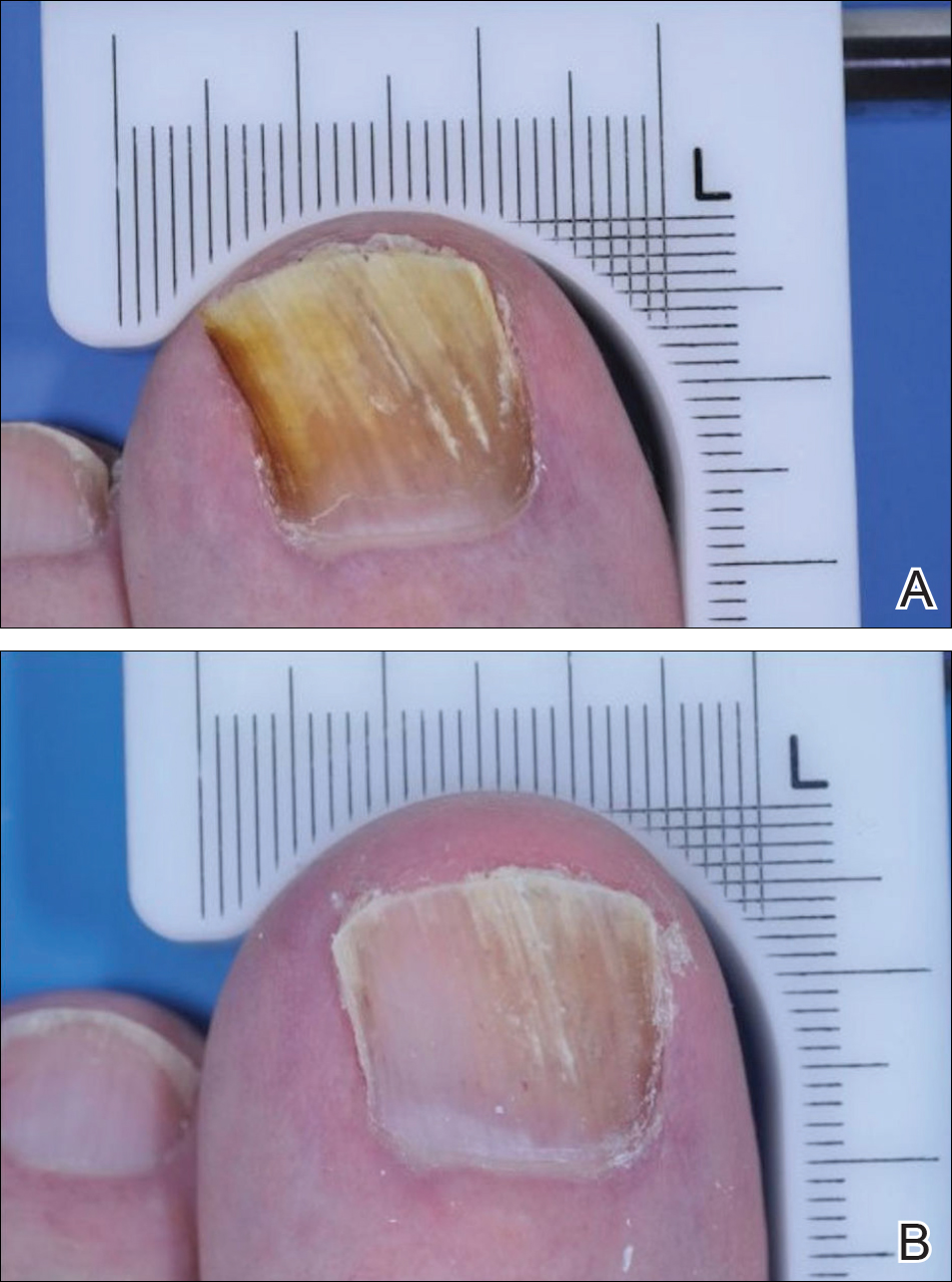
The primary end point was complete cure of the TGT at week 52, with secondary end points of completely or almost clear TGT nail (≤10% dystrophic nail), completely or almost clear TGT nail (≤10% dystrophic nail) plus negative mycology, and negative mycology of TGT.5 Examples of TGTs in participants who achieved complete cure and almost clear nails with negative mycology before and after treatment with tavaborole are shown in Figure 1. An example of a patient considered to have treatment failure is shown in Figure 2. This patient showed marked improvement in nail appearance and had a negative culture result but had a positive KOH test, which demonstrates the stringency in which topical agents are judged in onychomycosis trials.5
Efinaconazole
Efinaconazole is a topical triazole antifungal specifically indicated to treat onychomycosis. Two identical randomized, vehicle-controlled, double-blind, multicenter trials were performed to assess the safety and efficacy of efinaconazole solution 10%.7 The first study (NCT01008033) involved 870 participants and was conducted at a total of 74 sites in Japan (33 sites), Canada (7 sites), and the United States (34 sites) between December 2009 and September 2011. The second study (NCT01007708) had 785 participants and was conducted at 44 sites in Canada (8 sites) and the United States (36 sites) between December 2009 and October 2011.
Participants aged 18 to 70 years with a clinical diagnosis of DSO affecting 1 or more TGT were eligible to participate.7 Other eligibility criteria included an uninfected toenail length 3 mm or more from the proximal nail fold, a maximum toenail thickness of 3 mm, positive KOH wet mounts, and positive dermatophyte or mixed dermatophyte/candida cultures. Dermatophytes included T rubrum and T mentagrophytes. Those with severe moccasin-type tinea pedis were excluded. Participants were randomized to receive efinaconazole or vehicle (3:1). Once-daily treatments were self-applied to nails for 48 weeks. Clinical assessments were made at baseline and every 12 weeks until week 48, with a follow-up assessment at week 52. No nail trimming protocol was provided.7
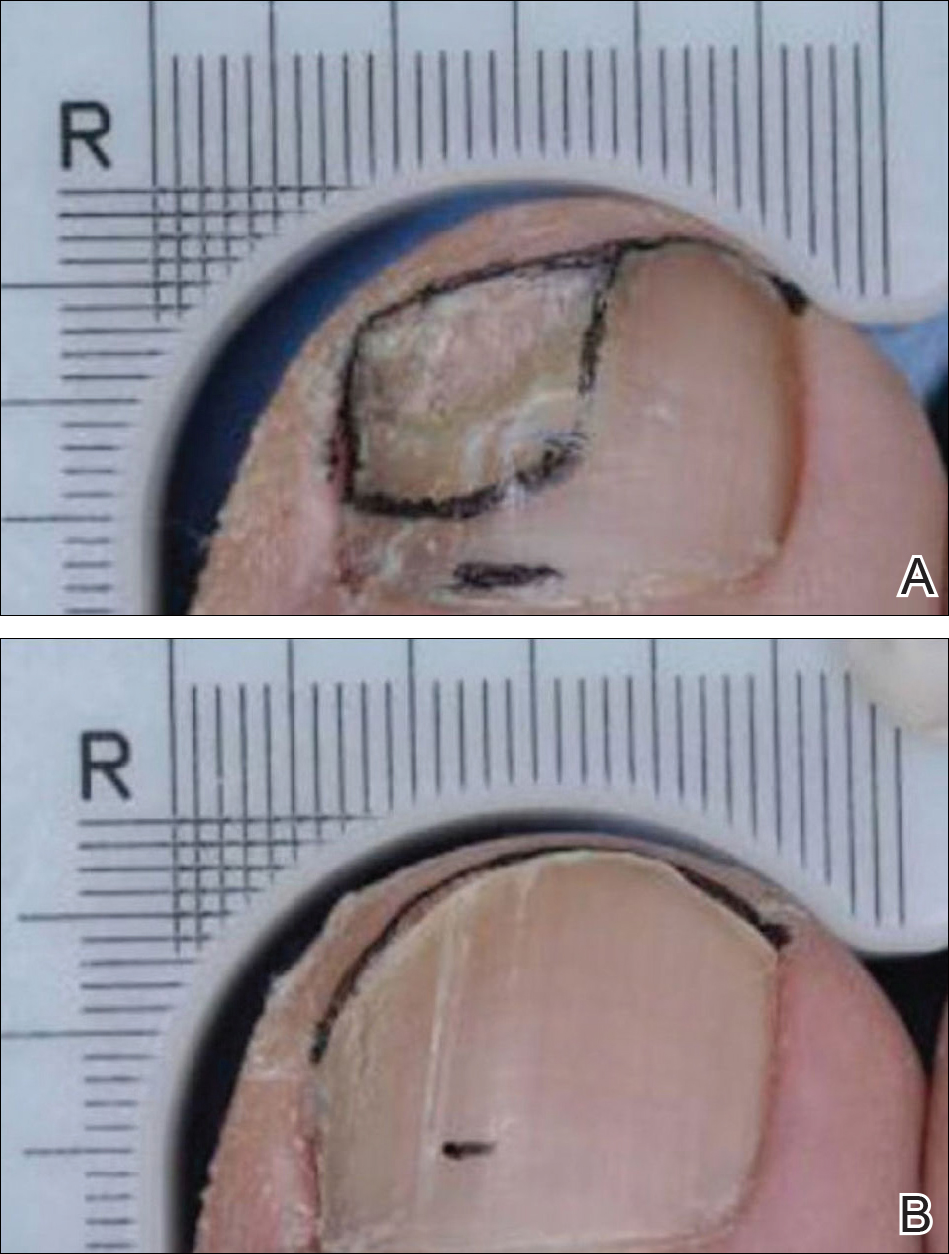
The primary end point of the efinaconazole phase 3 trials was complete cure at week 52, with secondary end points including mycologic cure, treatment success (≤5% mycotic nail), and complete or almost complete cure (negative culture and KOH, ≤5% mycotic nail). An example of a complete cure from baseline to week 52 is shown in Figure 3.7
Ciclopirox
Ciclopirox was the first topical therapy to be approved for the treatment of onychomycosis. Ciclopirox is a broad-spectrum antifungal agent that inhibits metal-dependent enzymes, which are responsible for the degradation of toxic peroxides in fungal cells. The safety and efficacy of ciclopirox nail lacquer topical solution 8% also was investigated in 2 identical phase 3 clinical trials.8 The first study was conducted at 9 sites in the United States between June 1994 and June 1996 and included 223 participants. The second study was conducted at 9 sites in the United States between July 1994 and April 1996 and included 237 participants.
Eligible participants were required to have DSO in at least one TGT, positive KOH wet mount with positive dermatophyte culture, and 20% to 65% nail involvement.8 Those with tinea pedis were not excluded. Participants were randomized to receive once-daily treatment with ciclopirox or vehicle (1:1)(applied to all toenails and affected fingernails) for 48 weeks. The product was to be removed by the patient with alcohol on a weekly basis. Trimming was allowed as necessary, and mechanical debridement by the physician could be performed monthly. Assessments were made every 4 weeks, and mycologic examinations were performed every 12 weeks. Participants who were clinically cured were assessed further in a 12- to 24-week posttreatment follow-up period.8
The primary end point of complete cure and secondary end points of treatment success (negative culture and KOH, ≤10% mycotic nail), mycologic cure, and negative mycologic culture were assessed at week 48.8
Phase 3 Clinical Trial Similarities and Differences
The phase 3 clinical trials used to investigate the safety and efficacy of tavaborole,5 efinaconazole,7 and ciclopirox8 were similar in their overall design. All trials were randomized, double-blind, vehicle-controlled studies in patients with DSO. Each agent was assessed using a once-daily application for a treatment period of 48 weeks.
Primary differences among study designs included the age range of participants, the range of mycotic nail involvement, the presence/absence of tinea pedis, and the nail trimming/debridement protocols used. Differences were observed in the patient eligibility criteria of these trials. Both mycotic area and participant age range were inconsistent for each agent (eTable). Participants with larger mycotic areas usually have a poorer prognosis, as they tend to have a greater fungal load.9 A baseline mycotic area of 20% to 60%,5 20% to 50%,7 and 20% to 65%8 at baseline was required for the tavaborole, efinaconazole, and ciclopirox trials, respectively. Variations in mycotic area between trials can affect treatment efficacy, as clinical cures can be reached quicker by patients with smaller areas of infection. Of note, the average mycotic area of involvement was not reported in the tavaborole studies but was 36% and 40% for the efinaconazole and ciclopirox studies, respectively.5,8 It also is more difficult to achieve complete cure in older patients, as they have poor circulation and reduced nail growth rates.1,10 The participant age range was 18 to 88 years in the tavaborole trials, with 8% of the participants older than 70 years,5 compared to 18 to 71 years in both the efinaconazole and ciclopirox trials.7,8 The average age of participants in each study was approximately 54, 51, and 50 years for tavaborole, efinaconazole, and ciclopirox, respectively. Because factors impacting treatment failure can increase with age, efficacy results can be confounded by differing age distributions across different studies.
Another important feature that differed between the clinical trials was the approach to nail trimming—defined as shortening of the free edge of the nail distal to the hyponychium—which varies from debridement in that the nail plate is removed or reduced in thickness proximal to the hyponychium. In the tavaborole trials, trimming was controlled to within 1 mm of the free edge of the nail,5 whereas the protocol used for the ciclopirox trials allowed nail trimming as necessary as well as moderate debridement before treatment application and on a monthly basis.8 Debridement is an important component in all ciclopirox trials, as it is used to reduce fungal load.11 No trimming control was provided during the efinaconazole trials; however, debridement was prohibited.7 These differences can dramatically affect the study results, as residual fungal elements and portions of infected nails are removed during the trimming process in an uncontrolled manner, which can affect mycologic testing results as well as the clinical efficacy results determined through investigator evaluation. Discrepancies regarding nail trimming approach inevitably makes the trial results difficult to compare, as mycologic cure is not translatable between studies.
Furthermore, somewhat unusually, complete cure rate variations were observed between different study centers in the efinaconazole trials. Japanese centers in the first efinaconazole study (NCT01008033) had higher complete cure rates in both the efinaconazole and vehicle treatment arms, which is notable because approximately 29% of participants in this study were Asian, mostly hailing from 33 Japanese centers. The reason for these confounding results is unknown and requires further analysis.
Lastly, the presence or absence of tinea pedis can affect the response to onychomycosis treatment. In the tavaborole trials, patients with active interdigital tinea pedis or exclusively plantar tinea pedis or chronic moccasin-type tinea pedis requiring treatment were excluded from the studies.5 In contrast, only patients with severe moccasin-type tinea pedis were excluded in efinaconazole trials.7 The ciclopirox studies had no exclusions based on presence of tinea pedis.8 These differences are noteworthy, as tinea pedis can serve as a reservoir for fungal infection if not treated and can lead to recurrence of onychomycosis.12
Conclusion
In recent years, disappointing efficacy has resulted in the failure of several topical agents for onychomycosis during their development; however, there are several aspects to consider when examining efficacy data in onychomycosis studies. Obtaining a complete cure in onychomycosis is difficult. Because patients applying treatments at home are unlikely to undergo mycologic testing to confirm complete cure, visual inspections are helpful to determine treatment efficacy.
Despite similar overall designs, notable differences in the study designs of the phase 3 clinical trials investigating tavaborole, efinaconazole, and ciclopirox are likely to have had an effect on the reported results, making the efficacy of the agents difficult to compare. It is particularly tempting to compare the primary end point results of each trial, especially considering tavaborole and efinaconazole had primary end points with the same parameters; however, there are several other factors (eg, age range of study population, extent of infection, nail trimming, patient demographics) that may have affected the outcomes of the studies and precluded a direct comparison of any end points. Without head-to-head investigations, there is room for prescribing clinicians to interpret results differently.
Acknowledgment
Writing and editorial assistance was provided by ApotheCom Associates, LLC, Yardley, Pennsylvania, and was supported by Sandoz, a Novartis division.
- Elewski BE. Onychomycosis: pathogenesis, diagnosis, and management. Clin Microbiol Rev. 1998;11:415-429.
- Thomas J, Jacobson GA, Narkowicz CK, et al. Toenail onychomycosis: an important global disease burden. J Clin Pharm Ther. 2010;35:497-519.
- Scher RK. Onychomycosis: a significant medical disorder. J Am Acad Dermatol. 1996;35(3, pt 2):S2-S5.
- Del Rosso JQ. The role of topical antifungal therapy for onychomycosis and the emergence of newer agents. J Clin Aesthet Dermatol. 2014;7:10-18.
- Elewski BE, Aly R, Baldwin SL, et al. Efficacy and safety of tavaborole topical solution, 5%, a novel boron-based antifungal agent, for the treatment of toenail onychomycosis: results from 2 randomized phase-III studies. J Am Acad Dermatol. 2015;73:62-69.
- Rock FL, Mao W, Yaremchuk A, et al. An antifungal agent inhibits an aminoacyl-tRNA synthetase by trapping tRNA in the editing site. Science. 2007;316:1759-1761.
- Elewski BE, Rich P, Pollak R, et al. Efinaconazole 10% solution in the treatment of toenail onychomycosis: two phase III multicenter, randomized, double-blind studies. J Am Acad Dermatol. 2013;68:600-608.
- Gupta AK, Joseph WS. Ciclopirox 8% nail lacquer in the treatment of onychomycosis of the toenails in the United States. J Am Pod Med Assoc. 2000;90:495-501.
- Carney C, Tosti A, Daniel R, et al. A new classification system for grading the severity of onychomycosis: Onychomycosis Severity Index. Arch Dermatol. 2011;147:1277-1282.
- Gupta AK. Onychomycosis in the elderly. Drugs Aging. 2000;16:397-407.
- Gupta AK, Malkin KF. Ciclopirox nail lacquer and podiatric practice. J Am Podiatr Med Assoc. 2000;90:502-507.
- Scher RK, Baran R. Onychomycosis in clinical practice: factors contributing to recurrence. Br J Dermatol. 2003;149(suppl 65):5-9.
Onychomycosis is a fungal nail infection primarily caused by dermatophytes.1 If left untreated, the infection can cause nail destruction and deformities,1 resulting in pain and discomfort,2 impaired foot mobility,3 and an overall reduced quality of life.1 Onychomycosis is a chronic condition that requires long treatment periods due to the slow growth rates of toenails.1 To successfully cure the condition, fungal eradication must be achieved.
Prior to the US Food and Drug Administration (FDA) approval of tavaborole and efinaconazole, ciclopirox was the only approved topical treatment for onychomycosis.4 The recent approval of tavaborole and efinaconazole has increased treatment options available to patients and has started to pave the way for future topical treatments. This article discusses the 3 approved topical treatments for onychomycosis and focuses on the design of the phase 3 clinical trials that led to their approval.
Topical Agents Used to Treat Onychomycosis
Tavaborole, efinaconazole, and ciclopirox have undergone extensive clinical investigation to receive FDA approval. Results from pivotal phase 3 studies establishing the efficacy and safety of each agent formed the basis for regulatory submission. Although it may seem intuitive to compare the relative performance of these agents based on their respective phase 3 clinical trial data, there are important differences in study methodology, conduct, and populations that prevent direct comparisons. The FDA provides limited guidance to the pharmaceutical industry on how to conduct clinical trials for potential onychomycosis treatments. Comparative efficacy and safety claims are limited based on cross-study comparisons. The details of the phase 3 trial designs are summarized in the Table.

Tavaborole
Tavaborole is a boron-based treatment with a novel mechanism of action.5 Tavaborole binds to the editing domain of leucyl–transfer ribonucleic acid synthetase via an integrated boron atom and inhibits fungal protein synthesis.6 Two identical randomized, double-blind, vehicle-controlled, parallel-group, phase 3 clinical trials evaluating tavaborole were performed.5 The first study (registered at www.clinicaltrials.gov with the identifier NCT01270971) included 594 participants from27 sites in the United States and Mexico and was conducted between December 2010 and November 2012. The second study (NCT01302119) included 604 participants from 32 sites in the United States and Canada and was conducted between February 2011 and January 2013.
Eligible participants 18 years and older had distal subungual onychomycosis (DSO) of the toenails affecting 20% to 60% of 1 or more target great toenails (TGTs), tested positive for fungus using potassium hydroxide (KOH) wet mounts and positive for Trichophyton rubrum and Trichophyton mentagrophytes on fungal culture diagnostic tests, had distal TGT thickness of 3 mm or less, and had 3 mm or more of clear nail between the proximal nail fold and the most proximal visible mycotic border.5 Those with active tinea pedis requiring treatment or with a history of chronic moccasin-type tinea pedis were excluded. Participants were randomized to receive either tavaborole or vehicle (2:1). Treatments were applied once daily to all infected toenails for a total of 48 weeks, and nail debridement (defined as partial or complete removal of the toenail) was not permitted. Notably, controlled trimming of the nail was allowed to 1 mm of the leading nail edge. Regular assessments of each toenail for disease involvement, onycholysis, and subungual hyperkeratosis were made at screening, baseline, week 2, week 6, and every 6 weeks thereafter until week 52. Subungual TGT samples were taken at screening and every 12 weeks during the study for examination at a mycology laboratory, which performed KOH and fungal culture tests. A follow-up assessment was made at week 52.5
The primary end point was complete cure of the TGT at week 52, with secondary end points of completely or almost clear TGT nail (≤10% dystrophic nail), completely or almost clear TGT nail (≤10% dystrophic nail) plus negative mycology, and negative mycology of TGT.5 Examples of TGTs in participants who achieved complete cure and almost clear nails with negative mycology before and after treatment with tavaborole are shown in Figure 1. An example of a patient considered to have treatment failure is shown in Figure 2. This patient showed marked improvement in nail appearance and had a negative culture result but had a positive KOH test, which demonstrates the stringency in which topical agents are judged in onychomycosis trials.5


Efinaconazole
Efinaconazole is a topical triazole antifungal specifically indicated to treat onychomycosis. Two identical randomized, vehicle-controlled, double-blind, multicenter trials were performed to assess the safety and efficacy of efinaconazole solution 10%.7 The first study (NCT01008033) involved 870 participants and was conducted at a total of 74 sites in Japan (33 sites), Canada (7 sites), and the United States (34 sites) between December 2009 and September 2011. The second study (NCT01007708) had 785 participants and was conducted at 44 sites in Canada (8 sites) and the United States (36 sites) between December 2009 and October 2011.
Participants aged 18 to 70 years with a clinical diagnosis of DSO affecting 1 or more TGT were eligible to participate.7 Other eligibility criteria included an uninfected toenail length 3 mm or more from the proximal nail fold, a maximum toenail thickness of 3 mm, positive KOH wet mounts, and positive dermatophyte or mixed dermatophyte/candida cultures. Dermatophytes included T rubrum and T mentagrophytes. Those with severe moccasin-type tinea pedis were excluded. Participants were randomized to receive efinaconazole or vehicle (3:1). Once-daily treatments were self-applied to nails for 48 weeks. Clinical assessments were made at baseline and every 12 weeks until week 48, with a follow-up assessment at week 52. No nail trimming protocol was provided.7
The primary end point of the efinaconazole phase 3 trials was complete cure at week 52, with secondary end points including mycologic cure, treatment success (≤5% mycotic nail), and complete or almost complete cure (negative culture and KOH, ≤5% mycotic nail). An example of a complete cure from baseline to week 52 is shown in Figure 3.7

Ciclopirox
Ciclopirox was the first topical therapy to be approved for the treatment of onychomycosis. Ciclopirox is a broad-spectrum antifungal agent that inhibits metal-dependent enzymes, which are responsible for the degradation of toxic peroxides in fungal cells. The safety and efficacy of ciclopirox nail lacquer topical solution 8% also was investigated in 2 identical phase 3 clinical trials.8 The first study was conducted at 9 sites in the United States between June 1994 and June 1996 and included 223 participants. The second study was conducted at 9 sites in the United States between July 1994 and April 1996 and included 237 participants.
Eligible participants were required to have DSO in at least one TGT, positive KOH wet mount with positive dermatophyte culture, and 20% to 65% nail involvement.8 Those with tinea pedis were not excluded. Participants were randomized to receive once-daily treatment with ciclopirox or vehicle (1:1)(applied to all toenails and affected fingernails) for 48 weeks. The product was to be removed by the patient with alcohol on a weekly basis. Trimming was allowed as necessary, and mechanical debridement by the physician could be performed monthly. Assessments were made every 4 weeks, and mycologic examinations were performed every 12 weeks. Participants who were clinically cured were assessed further in a 12- to 24-week posttreatment follow-up period.8
The primary end point of complete cure and secondary end points of treatment success (negative culture and KOH, ≤10% mycotic nail), mycologic cure, and negative mycologic culture were assessed at week 48.8
Phase 3 Clinical Trial Similarities and Differences
The phase 3 clinical trials used to investigate the safety and efficacy of tavaborole,5 efinaconazole,7 and ciclopirox8 were similar in their overall design. All trials were randomized, double-blind, vehicle-controlled studies in patients with DSO. Each agent was assessed using a once-daily application for a treatment period of 48 weeks.
Primary differences among study designs included the age range of participants, the range of mycotic nail involvement, the presence/absence of tinea pedis, and the nail trimming/debridement protocols used. Differences were observed in the patient eligibility criteria of these trials. Both mycotic area and participant age range were inconsistent for each agent (eTable). Participants with larger mycotic areas usually have a poorer prognosis, as they tend to have a greater fungal load.9 A baseline mycotic area of 20% to 60%,5 20% to 50%,7 and 20% to 65%8 at baseline was required for the tavaborole, efinaconazole, and ciclopirox trials, respectively. Variations in mycotic area between trials can affect treatment efficacy, as clinical cures can be reached quicker by patients with smaller areas of infection. Of note, the average mycotic area of involvement was not reported in the tavaborole studies but was 36% and 40% for the efinaconazole and ciclopirox studies, respectively.5,8 It also is more difficult to achieve complete cure in older patients, as they have poor circulation and reduced nail growth rates.1,10 The participant age range was 18 to 88 years in the tavaborole trials, with 8% of the participants older than 70 years,5 compared to 18 to 71 years in both the efinaconazole and ciclopirox trials.7,8 The average age of participants in each study was approximately 54, 51, and 50 years for tavaborole, efinaconazole, and ciclopirox, respectively. Because factors impacting treatment failure can increase with age, efficacy results can be confounded by differing age distributions across different studies.
Another important feature that differed between the clinical trials was the approach to nail trimming—defined as shortening of the free edge of the nail distal to the hyponychium—which varies from debridement in that the nail plate is removed or reduced in thickness proximal to the hyponychium. In the tavaborole trials, trimming was controlled to within 1 mm of the free edge of the nail,5 whereas the protocol used for the ciclopirox trials allowed nail trimming as necessary as well as moderate debridement before treatment application and on a monthly basis.8 Debridement is an important component in all ciclopirox trials, as it is used to reduce fungal load.11 No trimming control was provided during the efinaconazole trials; however, debridement was prohibited.7 These differences can dramatically affect the study results, as residual fungal elements and portions of infected nails are removed during the trimming process in an uncontrolled manner, which can affect mycologic testing results as well as the clinical efficacy results determined through investigator evaluation. Discrepancies regarding nail trimming approach inevitably makes the trial results difficult to compare, as mycologic cure is not translatable between studies.
Furthermore, somewhat unusually, complete cure rate variations were observed between different study centers in the efinaconazole trials. Japanese centers in the first efinaconazole study (NCT01008033) had higher complete cure rates in both the efinaconazole and vehicle treatment arms, which is notable because approximately 29% of participants in this study were Asian, mostly hailing from 33 Japanese centers. The reason for these confounding results is unknown and requires further analysis.
Lastly, the presence or absence of tinea pedis can affect the response to onychomycosis treatment. In the tavaborole trials, patients with active interdigital tinea pedis or exclusively plantar tinea pedis or chronic moccasin-type tinea pedis requiring treatment were excluded from the studies.5 In contrast, only patients with severe moccasin-type tinea pedis were excluded in efinaconazole trials.7 The ciclopirox studies had no exclusions based on presence of tinea pedis.8 These differences are noteworthy, as tinea pedis can serve as a reservoir for fungal infection if not treated and can lead to recurrence of onychomycosis.12
Conclusion
In recent years, disappointing efficacy has resulted in the failure of several topical agents for onychomycosis during their development; however, there are several aspects to consider when examining efficacy data in onychomycosis studies. Obtaining a complete cure in onychomycosis is difficult. Because patients applying treatments at home are unlikely to undergo mycologic testing to confirm complete cure, visual inspections are helpful to determine treatment efficacy.
Despite similar overall designs, notable differences in the study designs of the phase 3 clinical trials investigating tavaborole, efinaconazole, and ciclopirox are likely to have had an effect on the reported results, making the efficacy of the agents difficult to compare. It is particularly tempting to compare the primary end point results of each trial, especially considering tavaborole and efinaconazole had primary end points with the same parameters; however, there are several other factors (eg, age range of study population, extent of infection, nail trimming, patient demographics) that may have affected the outcomes of the studies and precluded a direct comparison of any end points. Without head-to-head investigations, there is room for prescribing clinicians to interpret results differently.
Acknowledgment
Writing and editorial assistance was provided by ApotheCom Associates, LLC, Yardley, Pennsylvania, and was supported by Sandoz, a Novartis division.
Onychomycosis is a fungal nail infection primarily caused by dermatophytes.1 If left untreated, the infection can cause nail destruction and deformities,1 resulting in pain and discomfort,2 impaired foot mobility,3 and an overall reduced quality of life.1 Onychomycosis is a chronic condition that requires long treatment periods due to the slow growth rates of toenails.1 To successfully cure the condition, fungal eradication must be achieved.
Prior to the US Food and Drug Administration (FDA) approval of tavaborole and efinaconazole, ciclopirox was the only approved topical treatment for onychomycosis.4 The recent approval of tavaborole and efinaconazole has increased treatment options available to patients and has started to pave the way for future topical treatments. This article discusses the 3 approved topical treatments for onychomycosis and focuses on the design of the phase 3 clinical trials that led to their approval.
Topical Agents Used to Treat Onychomycosis
Tavaborole, efinaconazole, and ciclopirox have undergone extensive clinical investigation to receive FDA approval. Results from pivotal phase 3 studies establishing the efficacy and safety of each agent formed the basis for regulatory submission. Although it may seem intuitive to compare the relative performance of these agents based on their respective phase 3 clinical trial data, there are important differences in study methodology, conduct, and populations that prevent direct comparisons. The FDA provides limited guidance to the pharmaceutical industry on how to conduct clinical trials for potential onychomycosis treatments. Comparative efficacy and safety claims are limited based on cross-study comparisons. The details of the phase 3 trial designs are summarized in the Table.

Tavaborole
Tavaborole is a boron-based treatment with a novel mechanism of action.5 Tavaborole binds to the editing domain of leucyl–transfer ribonucleic acid synthetase via an integrated boron atom and inhibits fungal protein synthesis.6 Two identical randomized, double-blind, vehicle-controlled, parallel-group, phase 3 clinical trials evaluating tavaborole were performed.5 The first study (registered at www.clinicaltrials.gov with the identifier NCT01270971) included 594 participants from27 sites in the United States and Mexico and was conducted between December 2010 and November 2012. The second study (NCT01302119) included 604 participants from 32 sites in the United States and Canada and was conducted between February 2011 and January 2013.
Eligible participants 18 years and older had distal subungual onychomycosis (DSO) of the toenails affecting 20% to 60% of 1 or more target great toenails (TGTs), tested positive for fungus using potassium hydroxide (KOH) wet mounts and positive for Trichophyton rubrum and Trichophyton mentagrophytes on fungal culture diagnostic tests, had distal TGT thickness of 3 mm or less, and had 3 mm or more of clear nail between the proximal nail fold and the most proximal visible mycotic border.5 Those with active tinea pedis requiring treatment or with a history of chronic moccasin-type tinea pedis were excluded. Participants were randomized to receive either tavaborole or vehicle (2:1). Treatments were applied once daily to all infected toenails for a total of 48 weeks, and nail debridement (defined as partial or complete removal of the toenail) was not permitted. Notably, controlled trimming of the nail was allowed to 1 mm of the leading nail edge. Regular assessments of each toenail for disease involvement, onycholysis, and subungual hyperkeratosis were made at screening, baseline, week 2, week 6, and every 6 weeks thereafter until week 52. Subungual TGT samples were taken at screening and every 12 weeks during the study for examination at a mycology laboratory, which performed KOH and fungal culture tests. A follow-up assessment was made at week 52.5
The primary end point was complete cure of the TGT at week 52, with secondary end points of completely or almost clear TGT nail (≤10% dystrophic nail), completely or almost clear TGT nail (≤10% dystrophic nail) plus negative mycology, and negative mycology of TGT.5 Examples of TGTs in participants who achieved complete cure and almost clear nails with negative mycology before and after treatment with tavaborole are shown in Figure 1. An example of a patient considered to have treatment failure is shown in Figure 2. This patient showed marked improvement in nail appearance and had a negative culture result but had a positive KOH test, which demonstrates the stringency in which topical agents are judged in onychomycosis trials.5


Efinaconazole
Efinaconazole is a topical triazole antifungal specifically indicated to treat onychomycosis. Two identical randomized, vehicle-controlled, double-blind, multicenter trials were performed to assess the safety and efficacy of efinaconazole solution 10%.7 The first study (NCT01008033) involved 870 participants and was conducted at a total of 74 sites in Japan (33 sites), Canada (7 sites), and the United States (34 sites) between December 2009 and September 2011. The second study (NCT01007708) had 785 participants and was conducted at 44 sites in Canada (8 sites) and the United States (36 sites) between December 2009 and October 2011.
Participants aged 18 to 70 years with a clinical diagnosis of DSO affecting 1 or more TGT were eligible to participate.7 Other eligibility criteria included an uninfected toenail length 3 mm or more from the proximal nail fold, a maximum toenail thickness of 3 mm, positive KOH wet mounts, and positive dermatophyte or mixed dermatophyte/candida cultures. Dermatophytes included T rubrum and T mentagrophytes. Those with severe moccasin-type tinea pedis were excluded. Participants were randomized to receive efinaconazole or vehicle (3:1). Once-daily treatments were self-applied to nails for 48 weeks. Clinical assessments were made at baseline and every 12 weeks until week 48, with a follow-up assessment at week 52. No nail trimming protocol was provided.7
The primary end point of the efinaconazole phase 3 trials was complete cure at week 52, with secondary end points including mycologic cure, treatment success (≤5% mycotic nail), and complete or almost complete cure (negative culture and KOH, ≤5% mycotic nail). An example of a complete cure from baseline to week 52 is shown in Figure 3.7

Ciclopirox
Ciclopirox was the first topical therapy to be approved for the treatment of onychomycosis. Ciclopirox is a broad-spectrum antifungal agent that inhibits metal-dependent enzymes, which are responsible for the degradation of toxic peroxides in fungal cells. The safety and efficacy of ciclopirox nail lacquer topical solution 8% also was investigated in 2 identical phase 3 clinical trials.8 The first study was conducted at 9 sites in the United States between June 1994 and June 1996 and included 223 participants. The second study was conducted at 9 sites in the United States between July 1994 and April 1996 and included 237 participants.
Eligible participants were required to have DSO in at least one TGT, positive KOH wet mount with positive dermatophyte culture, and 20% to 65% nail involvement.8 Those with tinea pedis were not excluded. Participants were randomized to receive once-daily treatment with ciclopirox or vehicle (1:1)(applied to all toenails and affected fingernails) for 48 weeks. The product was to be removed by the patient with alcohol on a weekly basis. Trimming was allowed as necessary, and mechanical debridement by the physician could be performed monthly. Assessments were made every 4 weeks, and mycologic examinations were performed every 12 weeks. Participants who were clinically cured were assessed further in a 12- to 24-week posttreatment follow-up period.8
The primary end point of complete cure and secondary end points of treatment success (negative culture and KOH, ≤10% mycotic nail), mycologic cure, and negative mycologic culture were assessed at week 48.8
Phase 3 Clinical Trial Similarities and Differences
The phase 3 clinical trials used to investigate the safety and efficacy of tavaborole,5 efinaconazole,7 and ciclopirox8 were similar in their overall design. All trials were randomized, double-blind, vehicle-controlled studies in patients with DSO. Each agent was assessed using a once-daily application for a treatment period of 48 weeks.
Primary differences among study designs included the age range of participants, the range of mycotic nail involvement, the presence/absence of tinea pedis, and the nail trimming/debridement protocols used. Differences were observed in the patient eligibility criteria of these trials. Both mycotic area and participant age range were inconsistent for each agent (eTable). Participants with larger mycotic areas usually have a poorer prognosis, as they tend to have a greater fungal load.9 A baseline mycotic area of 20% to 60%,5 20% to 50%,7 and 20% to 65%8 at baseline was required for the tavaborole, efinaconazole, and ciclopirox trials, respectively. Variations in mycotic area between trials can affect treatment efficacy, as clinical cures can be reached quicker by patients with smaller areas of infection. Of note, the average mycotic area of involvement was not reported in the tavaborole studies but was 36% and 40% for the efinaconazole and ciclopirox studies, respectively.5,8 It also is more difficult to achieve complete cure in older patients, as they have poor circulation and reduced nail growth rates.1,10 The participant age range was 18 to 88 years in the tavaborole trials, with 8% of the participants older than 70 years,5 compared to 18 to 71 years in both the efinaconazole and ciclopirox trials.7,8 The average age of participants in each study was approximately 54, 51, and 50 years for tavaborole, efinaconazole, and ciclopirox, respectively. Because factors impacting treatment failure can increase with age, efficacy results can be confounded by differing age distributions across different studies.
Another important feature that differed between the clinical trials was the approach to nail trimming—defined as shortening of the free edge of the nail distal to the hyponychium—which varies from debridement in that the nail plate is removed or reduced in thickness proximal to the hyponychium. In the tavaborole trials, trimming was controlled to within 1 mm of the free edge of the nail,5 whereas the protocol used for the ciclopirox trials allowed nail trimming as necessary as well as moderate debridement before treatment application and on a monthly basis.8 Debridement is an important component in all ciclopirox trials, as it is used to reduce fungal load.11 No trimming control was provided during the efinaconazole trials; however, debridement was prohibited.7 These differences can dramatically affect the study results, as residual fungal elements and portions of infected nails are removed during the trimming process in an uncontrolled manner, which can affect mycologic testing results as well as the clinical efficacy results determined through investigator evaluation. Discrepancies regarding nail trimming approach inevitably makes the trial results difficult to compare, as mycologic cure is not translatable between studies.
Furthermore, somewhat unusually, complete cure rate variations were observed between different study centers in the efinaconazole trials. Japanese centers in the first efinaconazole study (NCT01008033) had higher complete cure rates in both the efinaconazole and vehicle treatment arms, which is notable because approximately 29% of participants in this study were Asian, mostly hailing from 33 Japanese centers. The reason for these confounding results is unknown and requires further analysis.
Lastly, the presence or absence of tinea pedis can affect the response to onychomycosis treatment. In the tavaborole trials, patients with active interdigital tinea pedis or exclusively plantar tinea pedis or chronic moccasin-type tinea pedis requiring treatment were excluded from the studies.5 In contrast, only patients with severe moccasin-type tinea pedis were excluded in efinaconazole trials.7 The ciclopirox studies had no exclusions based on presence of tinea pedis.8 These differences are noteworthy, as tinea pedis can serve as a reservoir for fungal infection if not treated and can lead to recurrence of onychomycosis.12
Conclusion
In recent years, disappointing efficacy has resulted in the failure of several topical agents for onychomycosis during their development; however, there are several aspects to consider when examining efficacy data in onychomycosis studies. Obtaining a complete cure in onychomycosis is difficult. Because patients applying treatments at home are unlikely to undergo mycologic testing to confirm complete cure, visual inspections are helpful to determine treatment efficacy.
Despite similar overall designs, notable differences in the study designs of the phase 3 clinical trials investigating tavaborole, efinaconazole, and ciclopirox are likely to have had an effect on the reported results, making the efficacy of the agents difficult to compare. It is particularly tempting to compare the primary end point results of each trial, especially considering tavaborole and efinaconazole had primary end points with the same parameters; however, there are several other factors (eg, age range of study population, extent of infection, nail trimming, patient demographics) that may have affected the outcomes of the studies and precluded a direct comparison of any end points. Without head-to-head investigations, there is room for prescribing clinicians to interpret results differently.
Acknowledgment
Writing and editorial assistance was provided by ApotheCom Associates, LLC, Yardley, Pennsylvania, and was supported by Sandoz, a Novartis division.
- Elewski BE. Onychomycosis: pathogenesis, diagnosis, and management. Clin Microbiol Rev. 1998;11:415-429.
- Thomas J, Jacobson GA, Narkowicz CK, et al. Toenail onychomycosis: an important global disease burden. J Clin Pharm Ther. 2010;35:497-519.
- Scher RK. Onychomycosis: a significant medical disorder. J Am Acad Dermatol. 1996;35(3, pt 2):S2-S5.
- Del Rosso JQ. The role of topical antifungal therapy for onychomycosis and the emergence of newer agents. J Clin Aesthet Dermatol. 2014;7:10-18.
- Elewski BE, Aly R, Baldwin SL, et al. Efficacy and safety of tavaborole topical solution, 5%, a novel boron-based antifungal agent, for the treatment of toenail onychomycosis: results from 2 randomized phase-III studies. J Am Acad Dermatol. 2015;73:62-69.
- Rock FL, Mao W, Yaremchuk A, et al. An antifungal agent inhibits an aminoacyl-tRNA synthetase by trapping tRNA in the editing site. Science. 2007;316:1759-1761.
- Elewski BE, Rich P, Pollak R, et al. Efinaconazole 10% solution in the treatment of toenail onychomycosis: two phase III multicenter, randomized, double-blind studies. J Am Acad Dermatol. 2013;68:600-608.
- Gupta AK, Joseph WS. Ciclopirox 8% nail lacquer in the treatment of onychomycosis of the toenails in the United States. J Am Pod Med Assoc. 2000;90:495-501.
- Carney C, Tosti A, Daniel R, et al. A new classification system for grading the severity of onychomycosis: Onychomycosis Severity Index. Arch Dermatol. 2011;147:1277-1282.
- Gupta AK. Onychomycosis in the elderly. Drugs Aging. 2000;16:397-407.
- Gupta AK, Malkin KF. Ciclopirox nail lacquer and podiatric practice. J Am Podiatr Med Assoc. 2000;90:502-507.
- Scher RK, Baran R. Onychomycosis in clinical practice: factors contributing to recurrence. Br J Dermatol. 2003;149(suppl 65):5-9.
- Elewski BE. Onychomycosis: pathogenesis, diagnosis, and management. Clin Microbiol Rev. 1998;11:415-429.
- Thomas J, Jacobson GA, Narkowicz CK, et al. Toenail onychomycosis: an important global disease burden. J Clin Pharm Ther. 2010;35:497-519.
- Scher RK. Onychomycosis: a significant medical disorder. J Am Acad Dermatol. 1996;35(3, pt 2):S2-S5.
- Del Rosso JQ. The role of topical antifungal therapy for onychomycosis and the emergence of newer agents. J Clin Aesthet Dermatol. 2014;7:10-18.
- Elewski BE, Aly R, Baldwin SL, et al. Efficacy and safety of tavaborole topical solution, 5%, a novel boron-based antifungal agent, for the treatment of toenail onychomycosis: results from 2 randomized phase-III studies. J Am Acad Dermatol. 2015;73:62-69.
- Rock FL, Mao W, Yaremchuk A, et al. An antifungal agent inhibits an aminoacyl-tRNA synthetase by trapping tRNA in the editing site. Science. 2007;316:1759-1761.
- Elewski BE, Rich P, Pollak R, et al. Efinaconazole 10% solution in the treatment of toenail onychomycosis: two phase III multicenter, randomized, double-blind studies. J Am Acad Dermatol. 2013;68:600-608.
- Gupta AK, Joseph WS. Ciclopirox 8% nail lacquer in the treatment of onychomycosis of the toenails in the United States. J Am Pod Med Assoc. 2000;90:495-501.
- Carney C, Tosti A, Daniel R, et al. A new classification system for grading the severity of onychomycosis: Onychomycosis Severity Index. Arch Dermatol. 2011;147:1277-1282.
- Gupta AK. Onychomycosis in the elderly. Drugs Aging. 2000;16:397-407.
- Gupta AK, Malkin KF. Ciclopirox nail lacquer and podiatric practice. J Am Podiatr Med Assoc. 2000;90:502-507.
- Scher RK, Baran R. Onychomycosis in clinical practice: factors contributing to recurrence. Br J Dermatol. 2003;149(suppl 65):5-9.
Practice Points
- Despite similar overall designs, notable differences in the study designs of phase 3 clinical trials investigating tavaborole, efinaconazole, and ciclopirox for the treatment of onychomycosis are likely to have had an effect on the reported results, making the efficacy of these agents difficult to compare.
- The primary difference between studies for tavaborole, efinaconazole, and ciclopirox include the age range of participants, the range of mycotic nail involvement, the presence/absence of tinea pedis, and the nail trimming/debridement protocols used.
- Without head-to-head investigations, there is room for prescribing clinicians to interpret study results for these agents differently.
Atypical Fibroxanthoma Arising Within Erosive Pustular Dermatosis of the Scalp
Atypical fibroxanthoma (AFX) is a low-grade dermal malignancy comprised of atypical spindle cells.1 Classified as a superficial fibrohistiocytic tumor with intermediate malignant potential, AFX has an incidence of approximately 0.24% worldwide.2 The tumor appears mainly on the head and neck in sun-exposed areas but can occur less frequently on the trunk and limbs in non–sun-exposed areas. There is a 70% to 80% predominance in men aged 69 to 77 years, with lesions primarily occurring in sun-exposed areas of the head and neck.3 A median period of 4 months between time of onset and time of diagnosis has been previously established.4
When AFX does occur in non–sun-exposed areas, it tends to be in a younger patient population. Clinically, it presents as a rather nondescript, firm, erythematous papule or nodule less than 2 cm in diameter. Atypical fibroxanthoma most often presents asymptomatically, but the tumor may ulcerate and bleed, though pain and pruritus are uncommon.5 Findings are nonspecific, and the diagnosis must be confirmed with biopsy, as it can resemble other common dermatological lesions. The pathogenesis of AFX has been controversial. Two different studies looked at AFX using electron microscopy and concluded that the tumor most closely resembled a myofibroblast,6,7 which is consistent with current thinking today.
Atypical fibroxanthoma is believed to be associated with p53 mutation and is closely linked with exposure to UV radiation due to its predominance in sun-exposed areas. Other predisposing factors may include prior exposure to UV radiation, history of organ transplantation, immunosuppression, advanced age in men, and xeroderma pigmentosum. The differential diagnosis for AFX encompasses basal cell carcinoma, squamous cell carcinoma, Merkel cell carcinoma, adnexal tumor, and pyogenic granuloma.
Case Report
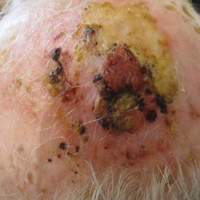
On physical examination, the lesions appeared erosive with crusting and granulation tissue (Figure 1A). The presentation was consistent with erosive pustular dermatosis of the scalp. Biopsy revealed granulation tissue. The patient underwent PDT and prednisone treatment with improvement. Additional biopsies revealed AKs. His condition improved with 2 PDT sessions but never fully cleared. During the PDT sessions, the patient reported intense unilateral headaches without visual changes. The headaches were intermittent and not apparently related to the treatments. He was referred for a temporal artery biopsy and rebiopsy of the remaining lesion on the scalp. The temporal artery biopsy was negative. The lesion that remained was a large nodule on the vertex scalp, and biopsy revealed AFX.
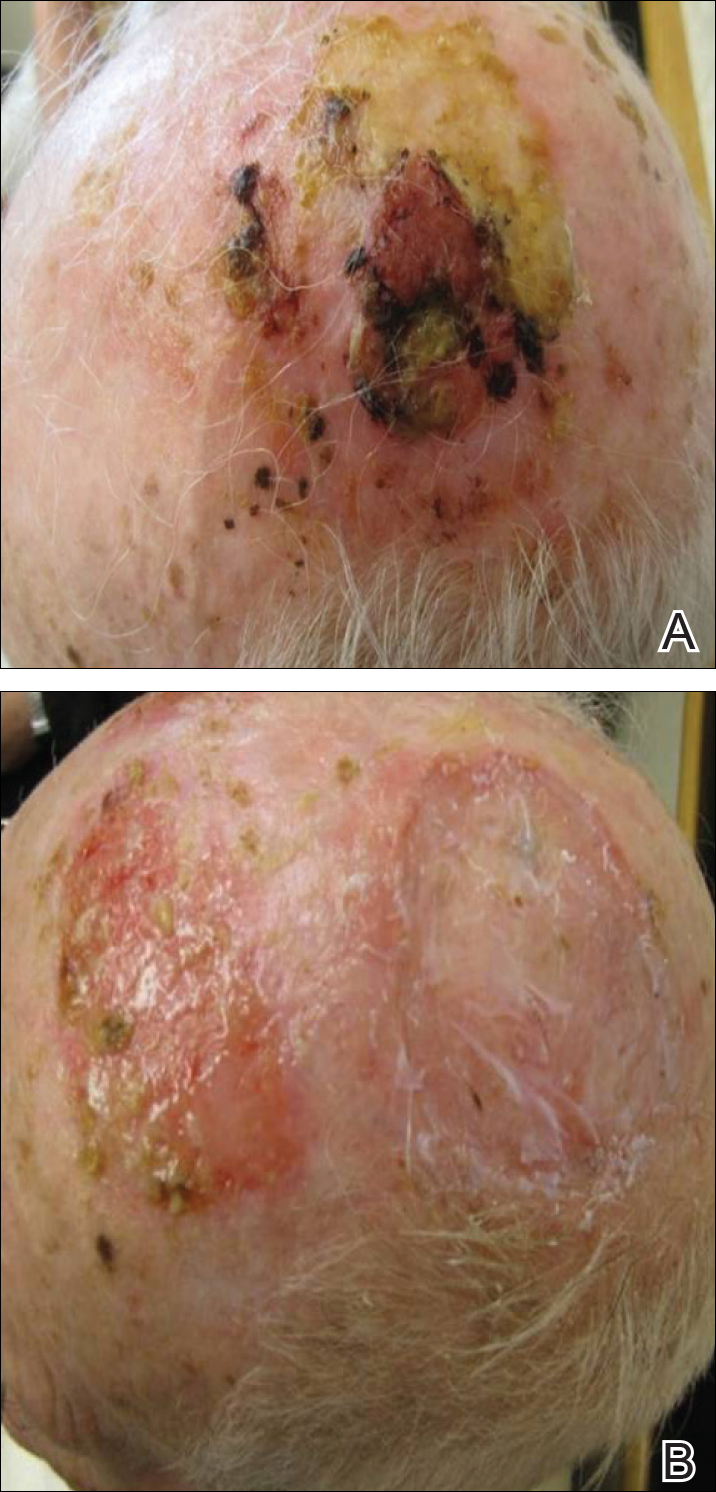
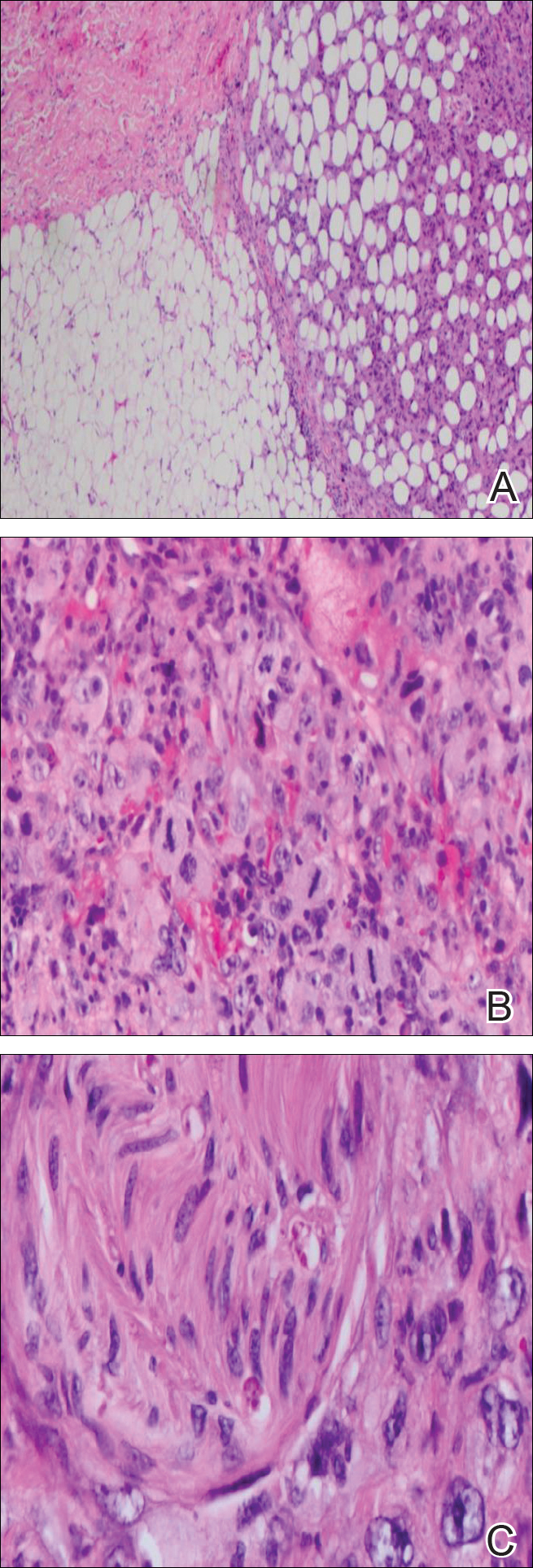
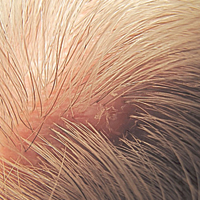
Immunohistochemical marker studies for S-100 and cytokeratin were negative. Invasion into subcutaneous fat was encountered (Figure 2A). Highly atypical spindle cells and mitoses were present (Figure 2B). Neoplastic cells were noted adjacent to nerve (Figure 2C). Excision of the lesion was curative, and his symptoms of pain and erosive pustular dermatosis resolved weeks thereafter (Figure 1B). The area of erosive pustular dermatosis was not excised, but symptoms resolved weeks following excision of the AFX.
Comment
Our case of AFX is unique due to the patient’s atypical presentation of severe pain. Because AFX usually presents asymptomatically, pain is an uncommon symptom. Based on the histologic findings in our case, we suspected that neural involvement of the tumor most likely explained the intense pain that our patient experienced.
The presence of erosive pustular dermatosis of the scalp also is interesting in our case. This elderly man had an extensive history of actinic damage and had reported pustules, scaling, itching, and scabbing of the scalp. It is possible that erosive pustular dermatosis was superimposed over the tumor and could have been the reason that multiple biopsies were needed to eventually arrive at a diagnosis. The coexistence of the 2 entities suggests that the chronic actinic damage played a role in the etiology of both.
Classification
There is a question regarding nomenclature when discussing AFX. Atypical fibroxanthoma has been referred to as a variant of undifferentiated pleomorphic sarcoma, which is a type of soft tissue sarcoma. Atypical fibroxanthoma can be referred to as undifferentiated pleomorphic sarcoma if it is more than 2 cm in diameter, if it involves the fascia or subcutaneous tissue, or if there is evidence of necrosis.3 Atypical fibroxanthoma generally is confined to the head and neck region and usually is less than 2 cm in diameter. In this patient, the presentation was consistent with AFX, as there was evidence of necrosis and invasion into the subcutaneous fat. The fact that the lesion also appeared on the scalp further supported the diagnosis of AFX.
Pathology
Biopsy of AFX typically reveals a spindle cell proliferation that usually arises in the setting of profound actinic damage. The epidermis may or may not be ulcerated, and in most cases, it is seen in close proximity to the overlying epidermis but not arising from it.8 Classic AFX is composed of highly atypical histiocytelike (epithelioid) cells admixed with pleomorphic spindle cells and giant cells, all showing frequent mitoses including atypical ones.9 Several histologic subtypes of AFX have been described, including clear cell, granular cell, pigmented cell, chondroid, osteoid, osteoclastic, and the most common spindle cell subtype.9 Features that indicate potential aggressive behavior include infiltration into the subcutaneous tissue, vascular invasion, and presence of necrosis. A diagnosis of AFX is made by exclusion of other malignant neoplasms with similar morphology, namely spindle cell squamous cell carcinoma, spindle cell melanoma, and leiomyoscarcoma.9 As such, immunohistochemistry plays a critical role in distinguishing these lesions, as they arise as part of the differential diagnosis. A panel of immunohistochemical stains is helpful for diagnosis and commonly includes but is not limited to S-100, Melan-A, smooth muscle actin, desmin, and cytokeratin.
Sampling error is an inherent flaw in any biopsy specimen. The eventual diagnosis of AFX in our case supports the argument for multiple biopsies of an unknown lesion, seeing as the affected area was interpreted as both granulation tissue and AK prior to the eventual diagnosis. Repeat biopsies, especially if a lesion is nonhealing, often can help clinicians arrive at a definitive diagnosis.
Treatment
Different treatment options have been used to manage AFX. Mohs micrographic surgery is most often used because of its tissue-sparing potential, often giving the most cosmetically appealing result. Wide local excision is another surgical technique utilized, generally with fixed margins of at least 1 cm.10 Radiation at the tumor site is used as a treatment method but most often during cases of reoccurrence. Cryotherapy as well as electrodesiccation and curettage are possible treatment options but are not the standard of care.
- Helwig EB. Atypical fibroxanthoma, in tumor seminar. proceedings of 18th Annual Seminar of San Antonio Society of Pathologists, 1961. Tex State J Med. 1963;59:664-667.
- Anderson HL, Joseph AK. A pilot feasibility study of a rare skin tumor database. Dermatol Surg. 2007;33:693-696.
- Iorizzo LJ 3rd, Brown MD. Atypical fibroxanthoma: a review of the literature. Dermatol Surg. 2011;37:146-157.
- Fretzin DF, Helwig EB. Atypical fibroxanthoma of the skin. a clinicopathologic study of 140 cases. Cancer. 1973;31:1541-1552.
- Vandergriff TW, Reed JA, Orengo IF. An unusual presentation of atypical fibroxanthoma. Dermatol Online J. 2008;14:6.
- Weedon D, Kerr JF. Atypical fibroxanthoma of skin: an electron microscope study. Pathology. 1975;7:173-177.
- Woyke S, Domagala W, Olszewski W, et al. Pseudosarcoma of the skin. an electron microscopic study and comparison with the fine structure of spindle-cell variant of squamous carcinoma. Cancer. 1974;33:970-980.
- Edward S, Yung A. Essential Dermatopathology. Philadelphia, PA: Lippincott Williams & Wilkins; 2012.
- Luzar B, Calonje E. Morphologic and immunohistochemical characteristics of atypical fibroxanthoma with a special emphasis on potential diagnostic pitfalls: a review. J Cutan Pathol. 2010;37:301-309.
- González-García R, Nam-Cha SH, Muñoz-Guerra MF, et al. Atypical fibroxanthoma of the head and neck: report of 5 cases. J Oral Maxillofac Surg. 2007;65:526-531.
Atypical fibroxanthoma (AFX) is a low-grade dermal malignancy comprised of atypical spindle cells.1 Classified as a superficial fibrohistiocytic tumor with intermediate malignant potential, AFX has an incidence of approximately 0.24% worldwide.2 The tumor appears mainly on the head and neck in sun-exposed areas but can occur less frequently on the trunk and limbs in non–sun-exposed areas. There is a 70% to 80% predominance in men aged 69 to 77 years, with lesions primarily occurring in sun-exposed areas of the head and neck.3 A median period of 4 months between time of onset and time of diagnosis has been previously established.4
When AFX does occur in non–sun-exposed areas, it tends to be in a younger patient population. Clinically, it presents as a rather nondescript, firm, erythematous papule or nodule less than 2 cm in diameter. Atypical fibroxanthoma most often presents asymptomatically, but the tumor may ulcerate and bleed, though pain and pruritus are uncommon.5 Findings are nonspecific, and the diagnosis must be confirmed with biopsy, as it can resemble other common dermatological lesions. The pathogenesis of AFX has been controversial. Two different studies looked at AFX using electron microscopy and concluded that the tumor most closely resembled a myofibroblast,6,7 which is consistent with current thinking today.
Atypical fibroxanthoma is believed to be associated with p53 mutation and is closely linked with exposure to UV radiation due to its predominance in sun-exposed areas. Other predisposing factors may include prior exposure to UV radiation, history of organ transplantation, immunosuppression, advanced age in men, and xeroderma pigmentosum. The differential diagnosis for AFX encompasses basal cell carcinoma, squamous cell carcinoma, Merkel cell carcinoma, adnexal tumor, and pyogenic granuloma.
Case Report
On physical examination, the lesions appeared erosive with crusting and granulation tissue (Figure 1A). The presentation was consistent with erosive pustular dermatosis of the scalp. Biopsy revealed granulation tissue. The patient underwent PDT and prednisone treatment with improvement. Additional biopsies revealed AKs. His condition improved with 2 PDT sessions but never fully cleared. During the PDT sessions, the patient reported intense unilateral headaches without visual changes. The headaches were intermittent and not apparently related to the treatments. He was referred for a temporal artery biopsy and rebiopsy of the remaining lesion on the scalp. The temporal artery biopsy was negative. The lesion that remained was a large nodule on the vertex scalp, and biopsy revealed AFX.


Immunohistochemical marker studies for S-100 and cytokeratin were negative. Invasion into subcutaneous fat was encountered (Figure 2A). Highly atypical spindle cells and mitoses were present (Figure 2B). Neoplastic cells were noted adjacent to nerve (Figure 2C). Excision of the lesion was curative, and his symptoms of pain and erosive pustular dermatosis resolved weeks thereafter (Figure 1B). The area of erosive pustular dermatosis was not excised, but symptoms resolved weeks following excision of the AFX.
Comment
Our case of AFX is unique due to the patient’s atypical presentation of severe pain. Because AFX usually presents asymptomatically, pain is an uncommon symptom. Based on the histologic findings in our case, we suspected that neural involvement of the tumor most likely explained the intense pain that our patient experienced.
The presence of erosive pustular dermatosis of the scalp also is interesting in our case. This elderly man had an extensive history of actinic damage and had reported pustules, scaling, itching, and scabbing of the scalp. It is possible that erosive pustular dermatosis was superimposed over the tumor and could have been the reason that multiple biopsies were needed to eventually arrive at a diagnosis. The coexistence of the 2 entities suggests that the chronic actinic damage played a role in the etiology of both.
Classification
There is a question regarding nomenclature when discussing AFX. Atypical fibroxanthoma has been referred to as a variant of undifferentiated pleomorphic sarcoma, which is a type of soft tissue sarcoma. Atypical fibroxanthoma can be referred to as undifferentiated pleomorphic sarcoma if it is more than 2 cm in diameter, if it involves the fascia or subcutaneous tissue, or if there is evidence of necrosis.3 Atypical fibroxanthoma generally is confined to the head and neck region and usually is less than 2 cm in diameter. In this patient, the presentation was consistent with AFX, as there was evidence of necrosis and invasion into the subcutaneous fat. The fact that the lesion also appeared on the scalp further supported the diagnosis of AFX.
Pathology
Biopsy of AFX typically reveals a spindle cell proliferation that usually arises in the setting of profound actinic damage. The epidermis may or may not be ulcerated, and in most cases, it is seen in close proximity to the overlying epidermis but not arising from it.8 Classic AFX is composed of highly atypical histiocytelike (epithelioid) cells admixed with pleomorphic spindle cells and giant cells, all showing frequent mitoses including atypical ones.9 Several histologic subtypes of AFX have been described, including clear cell, granular cell, pigmented cell, chondroid, osteoid, osteoclastic, and the most common spindle cell subtype.9 Features that indicate potential aggressive behavior include infiltration into the subcutaneous tissue, vascular invasion, and presence of necrosis. A diagnosis of AFX is made by exclusion of other malignant neoplasms with similar morphology, namely spindle cell squamous cell carcinoma, spindle cell melanoma, and leiomyoscarcoma.9 As such, immunohistochemistry plays a critical role in distinguishing these lesions, as they arise as part of the differential diagnosis. A panel of immunohistochemical stains is helpful for diagnosis and commonly includes but is not limited to S-100, Melan-A, smooth muscle actin, desmin, and cytokeratin.
Sampling error is an inherent flaw in any biopsy specimen. The eventual diagnosis of AFX in our case supports the argument for multiple biopsies of an unknown lesion, seeing as the affected area was interpreted as both granulation tissue and AK prior to the eventual diagnosis. Repeat biopsies, especially if a lesion is nonhealing, often can help clinicians arrive at a definitive diagnosis.
Treatment
Different treatment options have been used to manage AFX. Mohs micrographic surgery is most often used because of its tissue-sparing potential, often giving the most cosmetically appealing result. Wide local excision is another surgical technique utilized, generally with fixed margins of at least 1 cm.10 Radiation at the tumor site is used as a treatment method but most often during cases of reoccurrence. Cryotherapy as well as electrodesiccation and curettage are possible treatment options but are not the standard of care.
Atypical fibroxanthoma (AFX) is a low-grade dermal malignancy comprised of atypical spindle cells.1 Classified as a superficial fibrohistiocytic tumor with intermediate malignant potential, AFX has an incidence of approximately 0.24% worldwide.2 The tumor appears mainly on the head and neck in sun-exposed areas but can occur less frequently on the trunk and limbs in non–sun-exposed areas. There is a 70% to 80% predominance in men aged 69 to 77 years, with lesions primarily occurring in sun-exposed areas of the head and neck.3 A median period of 4 months between time of onset and time of diagnosis has been previously established.4
When AFX does occur in non–sun-exposed areas, it tends to be in a younger patient population. Clinically, it presents as a rather nondescript, firm, erythematous papule or nodule less than 2 cm in diameter. Atypical fibroxanthoma most often presents asymptomatically, but the tumor may ulcerate and bleed, though pain and pruritus are uncommon.5 Findings are nonspecific, and the diagnosis must be confirmed with biopsy, as it can resemble other common dermatological lesions. The pathogenesis of AFX has been controversial. Two different studies looked at AFX using electron microscopy and concluded that the tumor most closely resembled a myofibroblast,6,7 which is consistent with current thinking today.
Atypical fibroxanthoma is believed to be associated with p53 mutation and is closely linked with exposure to UV radiation due to its predominance in sun-exposed areas. Other predisposing factors may include prior exposure to UV radiation, history of organ transplantation, immunosuppression, advanced age in men, and xeroderma pigmentosum. The differential diagnosis for AFX encompasses basal cell carcinoma, squamous cell carcinoma, Merkel cell carcinoma, adnexal tumor, and pyogenic granuloma.
Case Report
On physical examination, the lesions appeared erosive with crusting and granulation tissue (Figure 1A). The presentation was consistent with erosive pustular dermatosis of the scalp. Biopsy revealed granulation tissue. The patient underwent PDT and prednisone treatment with improvement. Additional biopsies revealed AKs. His condition improved with 2 PDT sessions but never fully cleared. During the PDT sessions, the patient reported intense unilateral headaches without visual changes. The headaches were intermittent and not apparently related to the treatments. He was referred for a temporal artery biopsy and rebiopsy of the remaining lesion on the scalp. The temporal artery biopsy was negative. The lesion that remained was a large nodule on the vertex scalp, and biopsy revealed AFX.


Immunohistochemical marker studies for S-100 and cytokeratin were negative. Invasion into subcutaneous fat was encountered (Figure 2A). Highly atypical spindle cells and mitoses were present (Figure 2B). Neoplastic cells were noted adjacent to nerve (Figure 2C). Excision of the lesion was curative, and his symptoms of pain and erosive pustular dermatosis resolved weeks thereafter (Figure 1B). The area of erosive pustular dermatosis was not excised, but symptoms resolved weeks following excision of the AFX.
Comment
Our case of AFX is unique due to the patient’s atypical presentation of severe pain. Because AFX usually presents asymptomatically, pain is an uncommon symptom. Based on the histologic findings in our case, we suspected that neural involvement of the tumor most likely explained the intense pain that our patient experienced.
The presence of erosive pustular dermatosis of the scalp also is interesting in our case. This elderly man had an extensive history of actinic damage and had reported pustules, scaling, itching, and scabbing of the scalp. It is possible that erosive pustular dermatosis was superimposed over the tumor and could have been the reason that multiple biopsies were needed to eventually arrive at a diagnosis. The coexistence of the 2 entities suggests that the chronic actinic damage played a role in the etiology of both.
Classification
There is a question regarding nomenclature when discussing AFX. Atypical fibroxanthoma has been referred to as a variant of undifferentiated pleomorphic sarcoma, which is a type of soft tissue sarcoma. Atypical fibroxanthoma can be referred to as undifferentiated pleomorphic sarcoma if it is more than 2 cm in diameter, if it involves the fascia or subcutaneous tissue, or if there is evidence of necrosis.3 Atypical fibroxanthoma generally is confined to the head and neck region and usually is less than 2 cm in diameter. In this patient, the presentation was consistent with AFX, as there was evidence of necrosis and invasion into the subcutaneous fat. The fact that the lesion also appeared on the scalp further supported the diagnosis of AFX.
Pathology
Biopsy of AFX typically reveals a spindle cell proliferation that usually arises in the setting of profound actinic damage. The epidermis may or may not be ulcerated, and in most cases, it is seen in close proximity to the overlying epidermis but not arising from it.8 Classic AFX is composed of highly atypical histiocytelike (epithelioid) cells admixed with pleomorphic spindle cells and giant cells, all showing frequent mitoses including atypical ones.9 Several histologic subtypes of AFX have been described, including clear cell, granular cell, pigmented cell, chondroid, osteoid, osteoclastic, and the most common spindle cell subtype.9 Features that indicate potential aggressive behavior include infiltration into the subcutaneous tissue, vascular invasion, and presence of necrosis. A diagnosis of AFX is made by exclusion of other malignant neoplasms with similar morphology, namely spindle cell squamous cell carcinoma, spindle cell melanoma, and leiomyoscarcoma.9 As such, immunohistochemistry plays a critical role in distinguishing these lesions, as they arise as part of the differential diagnosis. A panel of immunohistochemical stains is helpful for diagnosis and commonly includes but is not limited to S-100, Melan-A, smooth muscle actin, desmin, and cytokeratin.
Sampling error is an inherent flaw in any biopsy specimen. The eventual diagnosis of AFX in our case supports the argument for multiple biopsies of an unknown lesion, seeing as the affected area was interpreted as both granulation tissue and AK prior to the eventual diagnosis. Repeat biopsies, especially if a lesion is nonhealing, often can help clinicians arrive at a definitive diagnosis.
Treatment
Different treatment options have been used to manage AFX. Mohs micrographic surgery is most often used because of its tissue-sparing potential, often giving the most cosmetically appealing result. Wide local excision is another surgical technique utilized, generally with fixed margins of at least 1 cm.10 Radiation at the tumor site is used as a treatment method but most often during cases of reoccurrence. Cryotherapy as well as electrodesiccation and curettage are possible treatment options but are not the standard of care.
- Helwig EB. Atypical fibroxanthoma, in tumor seminar. proceedings of 18th Annual Seminar of San Antonio Society of Pathologists, 1961. Tex State J Med. 1963;59:664-667.
- Anderson HL, Joseph AK. A pilot feasibility study of a rare skin tumor database. Dermatol Surg. 2007;33:693-696.
- Iorizzo LJ 3rd, Brown MD. Atypical fibroxanthoma: a review of the literature. Dermatol Surg. 2011;37:146-157.
- Fretzin DF, Helwig EB. Atypical fibroxanthoma of the skin. a clinicopathologic study of 140 cases. Cancer. 1973;31:1541-1552.
- Vandergriff TW, Reed JA, Orengo IF. An unusual presentation of atypical fibroxanthoma. Dermatol Online J. 2008;14:6.
- Weedon D, Kerr JF. Atypical fibroxanthoma of skin: an electron microscope study. Pathology. 1975;7:173-177.
- Woyke S, Domagala W, Olszewski W, et al. Pseudosarcoma of the skin. an electron microscopic study and comparison with the fine structure of spindle-cell variant of squamous carcinoma. Cancer. 1974;33:970-980.
- Edward S, Yung A. Essential Dermatopathology. Philadelphia, PA: Lippincott Williams & Wilkins; 2012.
- Luzar B, Calonje E. Morphologic and immunohistochemical characteristics of atypical fibroxanthoma with a special emphasis on potential diagnostic pitfalls: a review. J Cutan Pathol. 2010;37:301-309.
- González-García R, Nam-Cha SH, Muñoz-Guerra MF, et al. Atypical fibroxanthoma of the head and neck: report of 5 cases. J Oral Maxillofac Surg. 2007;65:526-531.
- Helwig EB. Atypical fibroxanthoma, in tumor seminar. proceedings of 18th Annual Seminar of San Antonio Society of Pathologists, 1961. Tex State J Med. 1963;59:664-667.
- Anderson HL, Joseph AK. A pilot feasibility study of a rare skin tumor database. Dermatol Surg. 2007;33:693-696.
- Iorizzo LJ 3rd, Brown MD. Atypical fibroxanthoma: a review of the literature. Dermatol Surg. 2011;37:146-157.
- Fretzin DF, Helwig EB. Atypical fibroxanthoma of the skin. a clinicopathologic study of 140 cases. Cancer. 1973;31:1541-1552.
- Vandergriff TW, Reed JA, Orengo IF. An unusual presentation of atypical fibroxanthoma. Dermatol Online J. 2008;14:6.
- Weedon D, Kerr JF. Atypical fibroxanthoma of skin: an electron microscope study. Pathology. 1975;7:173-177.
- Woyke S, Domagala W, Olszewski W, et al. Pseudosarcoma of the skin. an electron microscopic study and comparison with the fine structure of spindle-cell variant of squamous carcinoma. Cancer. 1974;33:970-980.
- Edward S, Yung A. Essential Dermatopathology. Philadelphia, PA: Lippincott Williams & Wilkins; 2012.
- Luzar B, Calonje E. Morphologic and immunohistochemical characteristics of atypical fibroxanthoma with a special emphasis on potential diagnostic pitfalls: a review. J Cutan Pathol. 2010;37:301-309.
- González-García R, Nam-Cha SH, Muñoz-Guerra MF, et al. Atypical fibroxanthoma of the head and neck: report of 5 cases. J Oral Maxillofac Surg. 2007;65:526-531.
Practice Points
- Atypical fibroxanthoma predominantly occurs in older men on the head and neck.
- Erosive pustular dermatosis may be a benign entity, but if it does not resolve, continue to rebiopsy, as rare tumors may mimic this condition.
Platelet-rich plasma treatment for hair loss continues to be refined
SAN DIEGO – There is currently no standard protocol for injecting autologous platelet-rich plasma to stimulate hair growth, but the technique appears to be about 50% effective, according to Marc R. Avram, MD.
“I tell patients that this is not FDA [Food and Drug Administration] approved, but we think it to be safe,” said Dr. Avram, clinical professor of dermatology at the Cornell University, New York, said at the annual Masters of Aesthetics Symposium. “We don’t know how well it’s going to work. There are a lot of published data on it, but none of [them are] randomized or controlled long-term.”
In Dr. Avram’s experience, he has found that PRP is a good option for patients with difficult hair loss, such as those who had extensive hair loss after chemotherapy but the hair never grew back in the same fashion, or patients who have failed treatment with finasteride and minoxidil.
Currently, there is no standard protocol for using PRP to stimulate hair growth, but the approach Dr. Avram follows is modeled on his experience of injecting thousands of patients with triamcinolone acetonide (Kenalog) for hair loss every 4-6 weeks. After drawing 20 ccs-30 ccs of blood from the patient, the vial is placed in a centrifuge for 10 minutes, a process that separates PRP from red blood cells. Next, the clinician injects PRP into the deep dermis/superficial subcutaneous tissue of the desired treatment area. An average of 4 ccs-8 ccs is injected during each session.
After three monthly treatments, patients follow up at 3 and 6 months after the last treatment to evaluate efficacy. “All patients are told if there is regrowth or thickening of terminal hair, maintenance treatments will be needed every 6-9 months,” he said.
Published clinical trials of PRP include a follow-up period of 3-12 months and most demonstrate an efficacy in the range of 50%-70%. “It seems to be more effective for earlier stages of hair loss, and there are no known side effects to date,” said Dr. Avram, who has authored five textbooks on hair and cosmetic dermatology. “I had one patient call up to say he thought he had an increase in hair loss 2-3 weeks after treatment, but that’s one patient in a couple hundred. This may be similar to the effect minoxidil has on some patients. I’ve had no other issues with side effects.”
In his opinion, future challenges in the use of PRP for restoring hair loss include better defining optimal candidates for the procedure and establishing a better treatment protocol. “How often should maintenance be done?” he asked. “Is this going to be helpful for alopecia areata and scarring alopecia? Also, we need to determine if finasteride, minoxidil, low-level light laser therapy, or any other medications can enhance PRP efficacy in combination. What’s the optimal combination for patients? We don’t know yet. But I think in the future we will.”
Dr. Avram disclosed that he is a consultant for Restoration Robotics.
SAN DIEGO – There is currently no standard protocol for injecting autologous platelet-rich plasma to stimulate hair growth, but the technique appears to be about 50% effective, according to Marc R. Avram, MD.
“I tell patients that this is not FDA [Food and Drug Administration] approved, but we think it to be safe,” said Dr. Avram, clinical professor of dermatology at the Cornell University, New York, said at the annual Masters of Aesthetics Symposium. “We don’t know how well it’s going to work. There are a lot of published data on it, but none of [them are] randomized or controlled long-term.”
In Dr. Avram’s experience, he has found that PRP is a good option for patients with difficult hair loss, such as those who had extensive hair loss after chemotherapy but the hair never grew back in the same fashion, or patients who have failed treatment with finasteride and minoxidil.
Currently, there is no standard protocol for using PRP to stimulate hair growth, but the approach Dr. Avram follows is modeled on his experience of injecting thousands of patients with triamcinolone acetonide (Kenalog) for hair loss every 4-6 weeks. After drawing 20 ccs-30 ccs of blood from the patient, the vial is placed in a centrifuge for 10 minutes, a process that separates PRP from red blood cells. Next, the clinician injects PRP into the deep dermis/superficial subcutaneous tissue of the desired treatment area. An average of 4 ccs-8 ccs is injected during each session.
After three monthly treatments, patients follow up at 3 and 6 months after the last treatment to evaluate efficacy. “All patients are told if there is regrowth or thickening of terminal hair, maintenance treatments will be needed every 6-9 months,” he said.
Published clinical trials of PRP include a follow-up period of 3-12 months and most demonstrate an efficacy in the range of 50%-70%. “It seems to be more effective for earlier stages of hair loss, and there are no known side effects to date,” said Dr. Avram, who has authored five textbooks on hair and cosmetic dermatology. “I had one patient call up to say he thought he had an increase in hair loss 2-3 weeks after treatment, but that’s one patient in a couple hundred. This may be similar to the effect minoxidil has on some patients. I’ve had no other issues with side effects.”
In his opinion, future challenges in the use of PRP for restoring hair loss include better defining optimal candidates for the procedure and establishing a better treatment protocol. “How often should maintenance be done?” he asked. “Is this going to be helpful for alopecia areata and scarring alopecia? Also, we need to determine if finasteride, minoxidil, low-level light laser therapy, or any other medications can enhance PRP efficacy in combination. What’s the optimal combination for patients? We don’t know yet. But I think in the future we will.”
Dr. Avram disclosed that he is a consultant for Restoration Robotics.
SAN DIEGO – There is currently no standard protocol for injecting autologous platelet-rich plasma to stimulate hair growth, but the technique appears to be about 50% effective, according to Marc R. Avram, MD.
“I tell patients that this is not FDA [Food and Drug Administration] approved, but we think it to be safe,” said Dr. Avram, clinical professor of dermatology at the Cornell University, New York, said at the annual Masters of Aesthetics Symposium. “We don’t know how well it’s going to work. There are a lot of published data on it, but none of [them are] randomized or controlled long-term.”
In Dr. Avram’s experience, he has found that PRP is a good option for patients with difficult hair loss, such as those who had extensive hair loss after chemotherapy but the hair never grew back in the same fashion, or patients who have failed treatment with finasteride and minoxidil.
Currently, there is no standard protocol for using PRP to stimulate hair growth, but the approach Dr. Avram follows is modeled on his experience of injecting thousands of patients with triamcinolone acetonide (Kenalog) for hair loss every 4-6 weeks. After drawing 20 ccs-30 ccs of blood from the patient, the vial is placed in a centrifuge for 10 minutes, a process that separates PRP from red blood cells. Next, the clinician injects PRP into the deep dermis/superficial subcutaneous tissue of the desired treatment area. An average of 4 ccs-8 ccs is injected during each session.
After three monthly treatments, patients follow up at 3 and 6 months after the last treatment to evaluate efficacy. “All patients are told if there is regrowth or thickening of terminal hair, maintenance treatments will be needed every 6-9 months,” he said.
Published clinical trials of PRP include a follow-up period of 3-12 months and most demonstrate an efficacy in the range of 50%-70%. “It seems to be more effective for earlier stages of hair loss, and there are no known side effects to date,” said Dr. Avram, who has authored five textbooks on hair and cosmetic dermatology. “I had one patient call up to say he thought he had an increase in hair loss 2-3 weeks after treatment, but that’s one patient in a couple hundred. This may be similar to the effect minoxidil has on some patients. I’ve had no other issues with side effects.”
In his opinion, future challenges in the use of PRP for restoring hair loss include better defining optimal candidates for the procedure and establishing a better treatment protocol. “How often should maintenance be done?” he asked. “Is this going to be helpful for alopecia areata and scarring alopecia? Also, we need to determine if finasteride, minoxidil, low-level light laser therapy, or any other medications can enhance PRP efficacy in combination. What’s the optimal combination for patients? We don’t know yet. But I think in the future we will.”
Dr. Avram disclosed that he is a consultant for Restoration Robotics.
AT MOAS 2017
Alopecia patients share their struggles
SILVER SPRING, MD. – Alopecia areata patients struggle as much, if not more so, with the social and emotional challenges of the disease as with the physical challenges, according to patients and others who spoke at a public meeting on alopecia areata patient-focused drug development.
Alopecia areata affects as many as 6.8 million individuals in the United States, according to the National Alopecia Areata Foundation (NAAF). However, the particulars of alopecia can vary widely from one person to another; some patients experience total hair loss (alopecia universalis), while others retain eyebrows, eyelashes, or some body hair.
The FDA meeting, held on Sept. 11, is part of the agency’s patient-focused drug development initiative. “We wanted to hear the broader patient’s voice,” Theresa M. Mullin, PhD, director of the FDA’s Office of Strategic Programs, said in her opening remarks. Gary Sherwood, communications director for NAAF, said that the meeting was the culmination of a 5-year effort, begun in 2012 when alopecia areata was named as one of 39 disease categories under consideration for such a meeting. “It is too early to know what the exact results will be … but if the past is any indication, they may be significant. The meeting held with psoriasis yielded FDA approval of a treatment previously denied,” he added in an interview.
Two panel presentations featured patients who discussed their experiences with alopecia; each was followed by a discussion period where patients and family members in the audience were invited to share their experiences.
The “Health Effects and Daily Impacts” panel allowed several patients and their family members the opportunity to identify specific issues that may surprise clinicians.
“One thing I learned was how much the patients are bothered by sweating of the scalp; this can affect what type of head covering, hair piece, or hat/helmet they are able to wear, and thus limits activities,” Dr. Marathe continued. “This is not something I had focused on previously. I will be more inclined to ask about sweating and offer treatments, such as scalp botulinum toxin or aluminum chloride now that I have been alerted to this concern. Also, the challenges of facial makeup such as pencil for eyebrows was another thing that the FDA session brought home for me; I’m more inclined to suggest things such as microblading for eyebrows, or to try treatments like latanoprost for eyebrows/lashes.”
The second panel, “Current Approaches to Treatment,” included a different group of patients who shared stories of treatments that had been successful and those that had not. “The patients at the FDA meeting expressed very eloquently what our patients feel – different treatments may work temporarily and then stop working, which leads to a roller coaster of emotions of hope and disappointment,” A. Yasmine Kirkorian, MD, also a dermatologist at Children’s National Health System, said in an interview. “Patients and physicians would be interested in a treatment option with a track record for predictable efficacy with durable and sustained hair regrowth and minimal side effects.”
Dr. Marathe noted that in her experience, those who develop alopecia totalis or universalis at a younger age tend to have more recalcitrant disease. “It is still very hard for me to predict which children will regrow their hair spontaneously, or with topical therapies, versus those with more resistant disease. I hope that continued study will allow us to offer a more realistic prognosis for these patients,” she said.
Discussion after the treatment panel included testimonials from patients who reported successful treatment with tofacitinib (Xeljanz), a Janus kinase inhibitor approved for rheumatoid arthritis, which is not approved for treatment of alopecia.
“I absolutely agree with the focus on JAK inhibitors and increasing our understanding of how they work, as well as what some of the long-term effects are,” said Dr. Marathe. “The better we are able to target the pathogenesis of this condition, the more easily we can treat in a more focused fashion and reduce side effects,” but more clinical trials are needed to determine safety and efficacy for children and teens, she noted.
One of her hesitations in prescribing tofacitinib to her patients is that she cannot provide them with a sense of how long they will need to be on the treatment. “Current data show that the hair growth on the medication is usually lost upon stopping it; the question I still struggle with is whether it is realistic to put a 4- or 5-year-old on a medication that has no estimated or anticipated stop date,” she said.
As for what she offers patients in terms of resources for emotional support, Dr. Kirkorian said the psychosocial aspects of alopecia areata are always discussed at patient visits. “Psychosocial needs vary based on age, personality, and personal philosophy. We offer the gamut of outside resources from local support groups, the National Alopecia Areata Foundation, referral to psychology/psychiatry and, very importantly, referral to Camp Discovery. Children have told us across the board how important and meaningful it was to them to be able to just be themselves around other children who look like them.”
Dr. Marathe and Dr. Kirkorian were attendees at the meeting; they had no relevant disclosures. They are members of the Dermatology News Editorial Advisory Board.
SILVER SPRING, MD. – Alopecia areata patients struggle as much, if not more so, with the social and emotional challenges of the disease as with the physical challenges, according to patients and others who spoke at a public meeting on alopecia areata patient-focused drug development.
Alopecia areata affects as many as 6.8 million individuals in the United States, according to the National Alopecia Areata Foundation (NAAF). However, the particulars of alopecia can vary widely from one person to another; some patients experience total hair loss (alopecia universalis), while others retain eyebrows, eyelashes, or some body hair.
The FDA meeting, held on Sept. 11, is part of the agency’s patient-focused drug development initiative. “We wanted to hear the broader patient’s voice,” Theresa M. Mullin, PhD, director of the FDA’s Office of Strategic Programs, said in her opening remarks. Gary Sherwood, communications director for NAAF, said that the meeting was the culmination of a 5-year effort, begun in 2012 when alopecia areata was named as one of 39 disease categories under consideration for such a meeting. “It is too early to know what the exact results will be … but if the past is any indication, they may be significant. The meeting held with psoriasis yielded FDA approval of a treatment previously denied,” he added in an interview.
Two panel presentations featured patients who discussed their experiences with alopecia; each was followed by a discussion period where patients and family members in the audience were invited to share their experiences.
The “Health Effects and Daily Impacts” panel allowed several patients and their family members the opportunity to identify specific issues that may surprise clinicians.
“One thing I learned was how much the patients are bothered by sweating of the scalp; this can affect what type of head covering, hair piece, or hat/helmet they are able to wear, and thus limits activities,” Dr. Marathe continued. “This is not something I had focused on previously. I will be more inclined to ask about sweating and offer treatments, such as scalp botulinum toxin or aluminum chloride now that I have been alerted to this concern. Also, the challenges of facial makeup such as pencil for eyebrows was another thing that the FDA session brought home for me; I’m more inclined to suggest things such as microblading for eyebrows, or to try treatments like latanoprost for eyebrows/lashes.”
The second panel, “Current Approaches to Treatment,” included a different group of patients who shared stories of treatments that had been successful and those that had not. “The patients at the FDA meeting expressed very eloquently what our patients feel – different treatments may work temporarily and then stop working, which leads to a roller coaster of emotions of hope and disappointment,” A. Yasmine Kirkorian, MD, also a dermatologist at Children’s National Health System, said in an interview. “Patients and physicians would be interested in a treatment option with a track record for predictable efficacy with durable and sustained hair regrowth and minimal side effects.”
Dr. Marathe noted that in her experience, those who develop alopecia totalis or universalis at a younger age tend to have more recalcitrant disease. “It is still very hard for me to predict which children will regrow their hair spontaneously, or with topical therapies, versus those with more resistant disease. I hope that continued study will allow us to offer a more realistic prognosis for these patients,” she said.
Discussion after the treatment panel included testimonials from patients who reported successful treatment with tofacitinib (Xeljanz), a Janus kinase inhibitor approved for rheumatoid arthritis, which is not approved for treatment of alopecia.
“I absolutely agree with the focus on JAK inhibitors and increasing our understanding of how they work, as well as what some of the long-term effects are,” said Dr. Marathe. “The better we are able to target the pathogenesis of this condition, the more easily we can treat in a more focused fashion and reduce side effects,” but more clinical trials are needed to determine safety and efficacy for children and teens, she noted.
One of her hesitations in prescribing tofacitinib to her patients is that she cannot provide them with a sense of how long they will need to be on the treatment. “Current data show that the hair growth on the medication is usually lost upon stopping it; the question I still struggle with is whether it is realistic to put a 4- or 5-year-old on a medication that has no estimated or anticipated stop date,” she said.
As for what she offers patients in terms of resources for emotional support, Dr. Kirkorian said the psychosocial aspects of alopecia areata are always discussed at patient visits. “Psychosocial needs vary based on age, personality, and personal philosophy. We offer the gamut of outside resources from local support groups, the National Alopecia Areata Foundation, referral to psychology/psychiatry and, very importantly, referral to Camp Discovery. Children have told us across the board how important and meaningful it was to them to be able to just be themselves around other children who look like them.”
Dr. Marathe and Dr. Kirkorian were attendees at the meeting; they had no relevant disclosures. They are members of the Dermatology News Editorial Advisory Board.
SILVER SPRING, MD. – Alopecia areata patients struggle as much, if not more so, with the social and emotional challenges of the disease as with the physical challenges, according to patients and others who spoke at a public meeting on alopecia areata patient-focused drug development.
Alopecia areata affects as many as 6.8 million individuals in the United States, according to the National Alopecia Areata Foundation (NAAF). However, the particulars of alopecia can vary widely from one person to another; some patients experience total hair loss (alopecia universalis), while others retain eyebrows, eyelashes, or some body hair.
The FDA meeting, held on Sept. 11, is part of the agency’s patient-focused drug development initiative. “We wanted to hear the broader patient’s voice,” Theresa M. Mullin, PhD, director of the FDA’s Office of Strategic Programs, said in her opening remarks. Gary Sherwood, communications director for NAAF, said that the meeting was the culmination of a 5-year effort, begun in 2012 when alopecia areata was named as one of 39 disease categories under consideration for such a meeting. “It is too early to know what the exact results will be … but if the past is any indication, they may be significant. The meeting held with psoriasis yielded FDA approval of a treatment previously denied,” he added in an interview.
Two panel presentations featured patients who discussed their experiences with alopecia; each was followed by a discussion period where patients and family members in the audience were invited to share their experiences.
The “Health Effects and Daily Impacts” panel allowed several patients and their family members the opportunity to identify specific issues that may surprise clinicians.
“One thing I learned was how much the patients are bothered by sweating of the scalp; this can affect what type of head covering, hair piece, or hat/helmet they are able to wear, and thus limits activities,” Dr. Marathe continued. “This is not something I had focused on previously. I will be more inclined to ask about sweating and offer treatments, such as scalp botulinum toxin or aluminum chloride now that I have been alerted to this concern. Also, the challenges of facial makeup such as pencil for eyebrows was another thing that the FDA session brought home for me; I’m more inclined to suggest things such as microblading for eyebrows, or to try treatments like latanoprost for eyebrows/lashes.”
The second panel, “Current Approaches to Treatment,” included a different group of patients who shared stories of treatments that had been successful and those that had not. “The patients at the FDA meeting expressed very eloquently what our patients feel – different treatments may work temporarily and then stop working, which leads to a roller coaster of emotions of hope and disappointment,” A. Yasmine Kirkorian, MD, also a dermatologist at Children’s National Health System, said in an interview. “Patients and physicians would be interested in a treatment option with a track record for predictable efficacy with durable and sustained hair regrowth and minimal side effects.”
Dr. Marathe noted that in her experience, those who develop alopecia totalis or universalis at a younger age tend to have more recalcitrant disease. “It is still very hard for me to predict which children will regrow their hair spontaneously, or with topical therapies, versus those with more resistant disease. I hope that continued study will allow us to offer a more realistic prognosis for these patients,” she said.
Discussion after the treatment panel included testimonials from patients who reported successful treatment with tofacitinib (Xeljanz), a Janus kinase inhibitor approved for rheumatoid arthritis, which is not approved for treatment of alopecia.
“I absolutely agree with the focus on JAK inhibitors and increasing our understanding of how they work, as well as what some of the long-term effects are,” said Dr. Marathe. “The better we are able to target the pathogenesis of this condition, the more easily we can treat in a more focused fashion and reduce side effects,” but more clinical trials are needed to determine safety and efficacy for children and teens, she noted.
One of her hesitations in prescribing tofacitinib to her patients is that she cannot provide them with a sense of how long they will need to be on the treatment. “Current data show that the hair growth on the medication is usually lost upon stopping it; the question I still struggle with is whether it is realistic to put a 4- or 5-year-old on a medication that has no estimated or anticipated stop date,” she said.
As for what she offers patients in terms of resources for emotional support, Dr. Kirkorian said the psychosocial aspects of alopecia areata are always discussed at patient visits. “Psychosocial needs vary based on age, personality, and personal philosophy. We offer the gamut of outside resources from local support groups, the National Alopecia Areata Foundation, referral to psychology/psychiatry and, very importantly, referral to Camp Discovery. Children have told us across the board how important and meaningful it was to them to be able to just be themselves around other children who look like them.”
Dr. Marathe and Dr. Kirkorian were attendees at the meeting; they had no relevant disclosures. They are members of the Dermatology News Editorial Advisory Board.
AT AN FDA PUBLIC MEETING
VIDEO: Alopecia areata patients seek emotional support
SILVER SPRING, MD. – The emotional challenges facing alopecia areata patients are as tough, or tougher, than the physical challenges, according to many patients participating in a public meeting on alopecia areata patient-focused drug development.
A panel of patients shared their experiences of living with alopecia areata, including Elizabeth DeCarlo of Wilmington, Delaware. In a video interview at the meeting, held at FDA headquarters on Sept. 11, Ms. DeCarlo elaborated on what she would like clinicians to understand about alopecia patients that might surprise them, and what matters to her as a patient.
“I would tell them to be more compassionate,” Ms. DeCarlo said. “It’s very emotional.” She also emphasized the value of giving alopecia patients information about local support groups, as well as national organizations such as the National Alopecia Areata Foundation.
Ms. DeCarlo had no financial conflicts to disclose.
SILVER SPRING, MD. – The emotional challenges facing alopecia areata patients are as tough, or tougher, than the physical challenges, according to many patients participating in a public meeting on alopecia areata patient-focused drug development.
A panel of patients shared their experiences of living with alopecia areata, including Elizabeth DeCarlo of Wilmington, Delaware. In a video interview at the meeting, held at FDA headquarters on Sept. 11, Ms. DeCarlo elaborated on what she would like clinicians to understand about alopecia patients that might surprise them, and what matters to her as a patient.
“I would tell them to be more compassionate,” Ms. DeCarlo said. “It’s very emotional.” She also emphasized the value of giving alopecia patients information about local support groups, as well as national organizations such as the National Alopecia Areata Foundation.
Ms. DeCarlo had no financial conflicts to disclose.
SILVER SPRING, MD. – The emotional challenges facing alopecia areata patients are as tough, or tougher, than the physical challenges, according to many patients participating in a public meeting on alopecia areata patient-focused drug development.
A panel of patients shared their experiences of living with alopecia areata, including Elizabeth DeCarlo of Wilmington, Delaware. In a video interview at the meeting, held at FDA headquarters on Sept. 11, Ms. DeCarlo elaborated on what she would like clinicians to understand about alopecia patients that might surprise them, and what matters to her as a patient.
“I would tell them to be more compassionate,” Ms. DeCarlo said. “It’s very emotional.” She also emphasized the value of giving alopecia patients information about local support groups, as well as national organizations such as the National Alopecia Areata Foundation.
Ms. DeCarlo had no financial conflicts to disclose.
AT AN FDA PUBLIC MEETING
Maintenance therapy typically required after laser hair removal
REPORTING FROM MOAS 2017
SAN DIEGO – Hair removal ranks as the most popular laser procedure performed in the United States, but patients with blond, red, or gray hairs are out of luck, since those threadlike strands lack a chromophore for the laser to respond to.
“For now, I recommend that these patients get electrolysis or use eflornithine cream,” Arisa Ortiz, MD, said at the annual Masters of Aesthetics Symposium.
Future treatment options for patients with light-colored hair look promising, however. One emerging technology combines laser hair removal with the insertion of a silver nanoparticle into the unpigmented hair follicle. “These are currently in pivotal trials, so we should be seeing them on the market very soon,” she said.
According to Dr. Ortiz, director of laser and cosmetic dermatology in the department of dermatology at the University of California, San Diego, there is still a place for nonlaser hair removal, including shaving, waxing, threading, and electrolysis, but laser hair removal is safe, effective in skilled hands, and permanent. Key factors in optimizing treatment include understanding laser safety and laser-tissue interaction, proper patient selection, preoperative preparation, parameter selection, and recognizing complications.
The first-degree target in laser hair removal is eumelanin contained in the bulb of hair follicles, she said, but the heat must diffuse to a secondary target – follicular stem cells in the bulge of the outer root sheath. “Pulse duration is important,” she said. “The thermal relaxation time of a terminal hair follicle is roughly 100 milliseconds. Longer pulse widths are going to be safer for darker skin types, and you want shorter pulse durations for fine hair, and longer pulse durations for thicker hair. Spot size is also important. Larger spot sizes are faster and create less pain and less epidermal damage.”
Indications include unwanted hair, hypertrichosis, and hirsutism/polycystic ovary syndrome (PCOS). “You want to counsel patients with PCOS properly, because they will require multiple treatments as they tend to make new hair follicles,” she said. Other indications include ingrown hairs, pseudofolliculitis barbae, and pilonidal cysts.
The best candidates for laser hair removal are patients who have a light skin color and dark hair, and those who have thick, coarse hair. “Be cautious when treating tanned patients, and adjust your setting to a longer pulse duration and a lower fluence,” she continued. “I tell (patients) they’ll likely need at least six treatments. You want to treat them every 6 to 8 weeks. If you do treatments sooner than that, it’s probably not cost effective for the patient, because of the way hair follicles cycle. It’s also important that they avoid the sun.”
Clinicians can achieve temporary hair removal with Q-switched lasers, which may be suitable for patients with pseudofolliculitis barbae but who may not want permanent hair removal. “This will just vaporize the actual hair follicle, but that heat is not extending to the stem cells, so it’s temporary hair removal, because the hair follicle transitions into the telogen phase,” Dr. Ortiz explained. “The hair will then grow back after a few months.”
Endpoints are the most important factor for laser hair removal. You want to see perifollicular erythema, perifollicular edema, or hair singeing. “Then you know you have an effective treatment setting,” she said. “Sometimes, however, it takes time for this erythema or edema to develop, so you don’t want to keep increasing your fluences to see this end point. If you’re not comfortable with the laser you’re using, I recommend waiting a few minutes after treatment, and looking for the end point. You could always go higher during the next treatment, if you need to.”
Higher fluences have been correlated with greater permanent hair removal, but also with more side effects. “The recommended treatment settings are going to be the highest possible tolerated fluence that yields the desired endpoint without any adverse effects,” Dr. Ortiz said.
The first hair removal laser to hit the market was the Ruby 694-nm laser, which is safe for Fitzpatrick skin types I-III. A long-term follow-up of the seminal study showed permanent posttreatment efficacy of up to 2 years (Arch Dermatol. 1998;134[7]:837-42). The Alexandrite 755-nm laser, meanwhile, penetrates deeper because it’s a longer wavelength, so there’s less melanin absorption, and it’s safer for darker skin types. “With a device like this, you want to make sure that you’re always holding the laser perpendicular to the skin surface so that your cryogen spray is firing at the same area as the laser. [That way] you don’t get a burn injury,” she said.
The diode at 800 nm and 810 nm penetrates even deeper, which results in less melanin absorption. “Originally these devices had smaller spot sizes, but now some of the newer devices have larger hand pieces and use contact cooling,” she said. “Some of the diode lasers cause singeing and char. The carbon actually sticks onto the sapphire window of the device, so you want to make sure you swipe the window after every few pulses so that you’re not putting the char onto the epidermis and causing an epidermal burn,” Dr. Ortiz advised.
She described the Nd:YAG 1,064-nm laser as the safest for skin types V and VI. It has the deepest penetration but the least melanin absorption. Intense pulsed light (IPL) can also be used for hair removal. IPLs “have a larger spot size, and you can use various cutoff filters to make them safer for darker skin types,” she said. “However, in head-to-head studies, usually laser hair removal does better than IPL.”
Potential complications from laser hair removal include paradoxical hypertrichosis; pigmentary alterations such as hyperpigmentation or hypopigmentation; infections/folliculitis, scarring, and eye injury. Dr. Ortiz underscored the importance of counseling patients about the need for maintenance treatments prior to initiating their first hair removal session. Laser hair removal removes about 85-90% of hairs permanently “so that leaves a significant number that remain, and new hairs may grow over time,” she said.
Authors of a recent study found that the plume release during laser hair removal should be considered a potential biohazard that warrants the use of smoke evacuators and good room ventilation (JAMA Dermatol. 2016;152[12]:1320-26). “We are learning that we should be more careful to evacuate the plume from laser hair removal or wear laser protective masks as the plume may contain harmful chemicals that we breathe in on a daily basis,” said Dr. Ortiz, who was not affiliated with the analysis.
She disclosed serving as a consultant to, receiving equipment from, and/or being a member of the scientific board of several device companies, including Alastin, Allergan, BTL, Cutera, InMode, Merz, Revance, Rodan and Fields, Sciton, and Sienna Biopharmaceuticals.
-[email protected]
REPORTING FROM MOAS 2017
SAN DIEGO – Hair removal ranks as the most popular laser procedure performed in the United States, but patients with blond, red, or gray hairs are out of luck, since those threadlike strands lack a chromophore for the laser to respond to.
“For now, I recommend that these patients get electrolysis or use eflornithine cream,” Arisa Ortiz, MD, said at the annual Masters of Aesthetics Symposium.
Future treatment options for patients with light-colored hair look promising, however. One emerging technology combines laser hair removal with the insertion of a silver nanoparticle into the unpigmented hair follicle. “These are currently in pivotal trials, so we should be seeing them on the market very soon,” she said.
According to Dr. Ortiz, director of laser and cosmetic dermatology in the department of dermatology at the University of California, San Diego, there is still a place for nonlaser hair removal, including shaving, waxing, threading, and electrolysis, but laser hair removal is safe, effective in skilled hands, and permanent. Key factors in optimizing treatment include understanding laser safety and laser-tissue interaction, proper patient selection, preoperative preparation, parameter selection, and recognizing complications.
The first-degree target in laser hair removal is eumelanin contained in the bulb of hair follicles, she said, but the heat must diffuse to a secondary target – follicular stem cells in the bulge of the outer root sheath. “Pulse duration is important,” she said. “The thermal relaxation time of a terminal hair follicle is roughly 100 milliseconds. Longer pulse widths are going to be safer for darker skin types, and you want shorter pulse durations for fine hair, and longer pulse durations for thicker hair. Spot size is also important. Larger spot sizes are faster and create less pain and less epidermal damage.”
Indications include unwanted hair, hypertrichosis, and hirsutism/polycystic ovary syndrome (PCOS). “You want to counsel patients with PCOS properly, because they will require multiple treatments as they tend to make new hair follicles,” she said. Other indications include ingrown hairs, pseudofolliculitis barbae, and pilonidal cysts.
The best candidates for laser hair removal are patients who have a light skin color and dark hair, and those who have thick, coarse hair. “Be cautious when treating tanned patients, and adjust your setting to a longer pulse duration and a lower fluence,” she continued. “I tell (patients) they’ll likely need at least six treatments. You want to treat them every 6 to 8 weeks. If you do treatments sooner than that, it’s probably not cost effective for the patient, because of the way hair follicles cycle. It’s also important that they avoid the sun.”
Clinicians can achieve temporary hair removal with Q-switched lasers, which may be suitable for patients with pseudofolliculitis barbae but who may not want permanent hair removal. “This will just vaporize the actual hair follicle, but that heat is not extending to the stem cells, so it’s temporary hair removal, because the hair follicle transitions into the telogen phase,” Dr. Ortiz explained. “The hair will then grow back after a few months.”
Endpoints are the most important factor for laser hair removal. You want to see perifollicular erythema, perifollicular edema, or hair singeing. “Then you know you have an effective treatment setting,” she said. “Sometimes, however, it takes time for this erythema or edema to develop, so you don’t want to keep increasing your fluences to see this end point. If you’re not comfortable with the laser you’re using, I recommend waiting a few minutes after treatment, and looking for the end point. You could always go higher during the next treatment, if you need to.”
Higher fluences have been correlated with greater permanent hair removal, but also with more side effects. “The recommended treatment settings are going to be the highest possible tolerated fluence that yields the desired endpoint without any adverse effects,” Dr. Ortiz said.
The first hair removal laser to hit the market was the Ruby 694-nm laser, which is safe for Fitzpatrick skin types I-III. A long-term follow-up of the seminal study showed permanent posttreatment efficacy of up to 2 years (Arch Dermatol. 1998;134[7]:837-42). The Alexandrite 755-nm laser, meanwhile, penetrates deeper because it’s a longer wavelength, so there’s less melanin absorption, and it’s safer for darker skin types. “With a device like this, you want to make sure that you’re always holding the laser perpendicular to the skin surface so that your cryogen spray is firing at the same area as the laser. [That way] you don’t get a burn injury,” she said.
The diode at 800 nm and 810 nm penetrates even deeper, which results in less melanin absorption. “Originally these devices had smaller spot sizes, but now some of the newer devices have larger hand pieces and use contact cooling,” she said. “Some of the diode lasers cause singeing and char. The carbon actually sticks onto the sapphire window of the device, so you want to make sure you swipe the window after every few pulses so that you’re not putting the char onto the epidermis and causing an epidermal burn,” Dr. Ortiz advised.
She described the Nd:YAG 1,064-nm laser as the safest for skin types V and VI. It has the deepest penetration but the least melanin absorption. Intense pulsed light (IPL) can also be used for hair removal. IPLs “have a larger spot size, and you can use various cutoff filters to make them safer for darker skin types,” she said. “However, in head-to-head studies, usually laser hair removal does better than IPL.”
Potential complications from laser hair removal include paradoxical hypertrichosis; pigmentary alterations such as hyperpigmentation or hypopigmentation; infections/folliculitis, scarring, and eye injury. Dr. Ortiz underscored the importance of counseling patients about the need for maintenance treatments prior to initiating their first hair removal session. Laser hair removal removes about 85-90% of hairs permanently “so that leaves a significant number that remain, and new hairs may grow over time,” she said.
Authors of a recent study found that the plume release during laser hair removal should be considered a potential biohazard that warrants the use of smoke evacuators and good room ventilation (JAMA Dermatol. 2016;152[12]:1320-26). “We are learning that we should be more careful to evacuate the plume from laser hair removal or wear laser protective masks as the plume may contain harmful chemicals that we breathe in on a daily basis,” said Dr. Ortiz, who was not affiliated with the analysis.
She disclosed serving as a consultant to, receiving equipment from, and/or being a member of the scientific board of several device companies, including Alastin, Allergan, BTL, Cutera, InMode, Merz, Revance, Rodan and Fields, Sciton, and Sienna Biopharmaceuticals.
-[email protected]
REPORTING FROM MOAS 2017
SAN DIEGO – Hair removal ranks as the most popular laser procedure performed in the United States, but patients with blond, red, or gray hairs are out of luck, since those threadlike strands lack a chromophore for the laser to respond to.
“For now, I recommend that these patients get electrolysis or use eflornithine cream,” Arisa Ortiz, MD, said at the annual Masters of Aesthetics Symposium.
Future treatment options for patients with light-colored hair look promising, however. One emerging technology combines laser hair removal with the insertion of a silver nanoparticle into the unpigmented hair follicle. “These are currently in pivotal trials, so we should be seeing them on the market very soon,” she said.
According to Dr. Ortiz, director of laser and cosmetic dermatology in the department of dermatology at the University of California, San Diego, there is still a place for nonlaser hair removal, including shaving, waxing, threading, and electrolysis, but laser hair removal is safe, effective in skilled hands, and permanent. Key factors in optimizing treatment include understanding laser safety and laser-tissue interaction, proper patient selection, preoperative preparation, parameter selection, and recognizing complications.
The first-degree target in laser hair removal is eumelanin contained in the bulb of hair follicles, she said, but the heat must diffuse to a secondary target – follicular stem cells in the bulge of the outer root sheath. “Pulse duration is important,” she said. “The thermal relaxation time of a terminal hair follicle is roughly 100 milliseconds. Longer pulse widths are going to be safer for darker skin types, and you want shorter pulse durations for fine hair, and longer pulse durations for thicker hair. Spot size is also important. Larger spot sizes are faster and create less pain and less epidermal damage.”
Indications include unwanted hair, hypertrichosis, and hirsutism/polycystic ovary syndrome (PCOS). “You want to counsel patients with PCOS properly, because they will require multiple treatments as they tend to make new hair follicles,” she said. Other indications include ingrown hairs, pseudofolliculitis barbae, and pilonidal cysts.
The best candidates for laser hair removal are patients who have a light skin color and dark hair, and those who have thick, coarse hair. “Be cautious when treating tanned patients, and adjust your setting to a longer pulse duration and a lower fluence,” she continued. “I tell (patients) they’ll likely need at least six treatments. You want to treat them every 6 to 8 weeks. If you do treatments sooner than that, it’s probably not cost effective for the patient, because of the way hair follicles cycle. It’s also important that they avoid the sun.”
Clinicians can achieve temporary hair removal with Q-switched lasers, which may be suitable for patients with pseudofolliculitis barbae but who may not want permanent hair removal. “This will just vaporize the actual hair follicle, but that heat is not extending to the stem cells, so it’s temporary hair removal, because the hair follicle transitions into the telogen phase,” Dr. Ortiz explained. “The hair will then grow back after a few months.”
Endpoints are the most important factor for laser hair removal. You want to see perifollicular erythema, perifollicular edema, or hair singeing. “Then you know you have an effective treatment setting,” she said. “Sometimes, however, it takes time for this erythema or edema to develop, so you don’t want to keep increasing your fluences to see this end point. If you’re not comfortable with the laser you’re using, I recommend waiting a few minutes after treatment, and looking for the end point. You could always go higher during the next treatment, if you need to.”
Higher fluences have been correlated with greater permanent hair removal, but also with more side effects. “The recommended treatment settings are going to be the highest possible tolerated fluence that yields the desired endpoint without any adverse effects,” Dr. Ortiz said.
The first hair removal laser to hit the market was the Ruby 694-nm laser, which is safe for Fitzpatrick skin types I-III. A long-term follow-up of the seminal study showed permanent posttreatment efficacy of up to 2 years (Arch Dermatol. 1998;134[7]:837-42). The Alexandrite 755-nm laser, meanwhile, penetrates deeper because it’s a longer wavelength, so there’s less melanin absorption, and it’s safer for darker skin types. “With a device like this, you want to make sure that you’re always holding the laser perpendicular to the skin surface so that your cryogen spray is firing at the same area as the laser. [That way] you don’t get a burn injury,” she said.
The diode at 800 nm and 810 nm penetrates even deeper, which results in less melanin absorption. “Originally these devices had smaller spot sizes, but now some of the newer devices have larger hand pieces and use contact cooling,” she said. “Some of the diode lasers cause singeing and char. The carbon actually sticks onto the sapphire window of the device, so you want to make sure you swipe the window after every few pulses so that you’re not putting the char onto the epidermis and causing an epidermal burn,” Dr. Ortiz advised.
She described the Nd:YAG 1,064-nm laser as the safest for skin types V and VI. It has the deepest penetration but the least melanin absorption. Intense pulsed light (IPL) can also be used for hair removal. IPLs “have a larger spot size, and you can use various cutoff filters to make them safer for darker skin types,” she said. “However, in head-to-head studies, usually laser hair removal does better than IPL.”
Potential complications from laser hair removal include paradoxical hypertrichosis; pigmentary alterations such as hyperpigmentation or hypopigmentation; infections/folliculitis, scarring, and eye injury. Dr. Ortiz underscored the importance of counseling patients about the need for maintenance treatments prior to initiating their first hair removal session. Laser hair removal removes about 85-90% of hairs permanently “so that leaves a significant number that remain, and new hairs may grow over time,” she said.
Authors of a recent study found that the plume release during laser hair removal should be considered a potential biohazard that warrants the use of smoke evacuators and good room ventilation (JAMA Dermatol. 2016;152[12]:1320-26). “We are learning that we should be more careful to evacuate the plume from laser hair removal or wear laser protective masks as the plume may contain harmful chemicals that we breathe in on a daily basis,” said Dr. Ortiz, who was not affiliated with the analysis.
She disclosed serving as a consultant to, receiving equipment from, and/or being a member of the scientific board of several device companies, including Alastin, Allergan, BTL, Cutera, InMode, Merz, Revance, Rodan and Fields, Sciton, and Sienna Biopharmaceuticals.
-[email protected]
Cosmetic Corner: Dermatologists Weigh in on Men’s Products
To improve patient care and outcomes, leading dermatologists offered their recommendations on men’s products. Consideration must be given to:
- bareMinerals SPF 30 Natural Sunscreen
Bare Escentuals Beauty, Inc
“I recommend this product to my male patients when they are not wearing a hat. It protects the scalp from UV damage without a heavy greasy finish.”—Shari Lipner, MD, PhD, New York, New York
- Ducray Alopexy 5% For Men
Pierre Fabre Laboratories
“This dermatologist-dispensed product for men addresses chronic hair loss as well as thinning hair. It contains an optimal level of minoxidil 5% in an elegant unscented formulation and is designed to spray on smoothly and evenly.”—Jeannette Graf, MD, Great Neck, New York
- Facial Fuel Energizing Scrub
Kiehl’s
“This product is great for oily skin and enlarged pores. The particles in the product allow one to get a deep-clean feeling.”—Gary Goldenberg, MD, New York, New York
- Physical Matte UV Defense SPF 50
SkinCeuticals
“For men I like to keep things simple. I recommend what I use with the single most important thing being daily sun protection. SkinCeuticals Physical Matte UV Defense SPF 50 is my favorite and I use it after I shave. It goes on smoothly and has a natural tint along with a high SPF.”—Jerome Potozkin, MD, Danville, California
- Ultimate Brushless Shave Cream
Kiehl’s
“I recommend this product for men with frequent irritation from shaving. This cream-based product helps to provide a close shave without as much irritation from other gel-based products. A small amount goes a long way!”—Anthony M. Rossi, MD, New York, New York
Cutis invites readers to send us their recommendations. Athlete’s foot treatments and cleansing devices will be featured in upcoming editions of Cosmetic Corner. Please email your recommendation(s) to the Editorial Office.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.
To improve patient care and outcomes, leading dermatologists offered their recommendations on men’s products. Consideration must be given to:
- bareMinerals SPF 30 Natural Sunscreen
Bare Escentuals Beauty, Inc
“I recommend this product to my male patients when they are not wearing a hat. It protects the scalp from UV damage without a heavy greasy finish.”—Shari Lipner, MD, PhD, New York, New York
- Ducray Alopexy 5% For Men
Pierre Fabre Laboratories
“This dermatologist-dispensed product for men addresses chronic hair loss as well as thinning hair. It contains an optimal level of minoxidil 5% in an elegant unscented formulation and is designed to spray on smoothly and evenly.”—Jeannette Graf, MD, Great Neck, New York
- Facial Fuel Energizing Scrub
Kiehl’s
“This product is great for oily skin and enlarged pores. The particles in the product allow one to get a deep-clean feeling.”—Gary Goldenberg, MD, New York, New York
- Physical Matte UV Defense SPF 50
SkinCeuticals
“For men I like to keep things simple. I recommend what I use with the single most important thing being daily sun protection. SkinCeuticals Physical Matte UV Defense SPF 50 is my favorite and I use it after I shave. It goes on smoothly and has a natural tint along with a high SPF.”—Jerome Potozkin, MD, Danville, California
- Ultimate Brushless Shave Cream
Kiehl’s
“I recommend this product for men with frequent irritation from shaving. This cream-based product helps to provide a close shave without as much irritation from other gel-based products. A small amount goes a long way!”—Anthony M. Rossi, MD, New York, New York
Cutis invites readers to send us their recommendations. Athlete’s foot treatments and cleansing devices will be featured in upcoming editions of Cosmetic Corner. Please email your recommendation(s) to the Editorial Office.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.
To improve patient care and outcomes, leading dermatologists offered their recommendations on men’s products. Consideration must be given to:
- bareMinerals SPF 30 Natural Sunscreen
Bare Escentuals Beauty, Inc
“I recommend this product to my male patients when they are not wearing a hat. It protects the scalp from UV damage without a heavy greasy finish.”—Shari Lipner, MD, PhD, New York, New York
- Ducray Alopexy 5% For Men
Pierre Fabre Laboratories
“This dermatologist-dispensed product for men addresses chronic hair loss as well as thinning hair. It contains an optimal level of minoxidil 5% in an elegant unscented formulation and is designed to spray on smoothly and evenly.”—Jeannette Graf, MD, Great Neck, New York
- Facial Fuel Energizing Scrub
Kiehl’s
“This product is great for oily skin and enlarged pores. The particles in the product allow one to get a deep-clean feeling.”—Gary Goldenberg, MD, New York, New York
- Physical Matte UV Defense SPF 50
SkinCeuticals
“For men I like to keep things simple. I recommend what I use with the single most important thing being daily sun protection. SkinCeuticals Physical Matte UV Defense SPF 50 is my favorite and I use it after I shave. It goes on smoothly and has a natural tint along with a high SPF.”—Jerome Potozkin, MD, Danville, California
- Ultimate Brushless Shave Cream
Kiehl’s
“I recommend this product for men with frequent irritation from shaving. This cream-based product helps to provide a close shave without as much irritation from other gel-based products. A small amount goes a long way!”—Anthony M. Rossi, MD, New York, New York
Cutis invites readers to send us their recommendations. Athlete’s foot treatments and cleansing devices will be featured in upcoming editions of Cosmetic Corner. Please email your recommendation(s) to the Editorial Office.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.
Solitary Nodule With White Hairs
The Diagnosis: Trichofolliculoma
Microscopic examination revealed a dilated cystic follicle that communicated with the skin surface (Figure). The follicle was lined with squamous epithelium and surrounded by numerous secondary follicles, many of which contained a hair shaft. A diagnosis of trichofolliculoma was made.
Clinically, the differential diagnosis of a flesh-colored papule on the scalp with prominent follicle includes dilated pore of Winer, epidermoid cyst, pilar sheath acanthoma, and trichoepithelioma.1,2 Multiple hair shafts present in a single follicle may be seen in pili multigemini, tufted folliculitis, trichostasis spinulosa, and trichofolliculoma. On histopathologic examination, a dilated central follicle surrounded with smaller secondary follicles was identified, consistent with trichofolliculoma.
Trichofolliculoma is a rare follicular hamartoma typically occurring on the face, scalp, or trunk as a solitary papule or nodule due to the proliferation of abnormal hair follicle stem cells.3,4 It may present as a flesh-colored nodule with a central pore that may drain sebum or contain white vellus hairs. Trichofolliculoma is considered a benign entity, despite one case report of malignant transformation.5 Biopsy is diagnostic and no further treatment is needed. Recurrence rarely occurs at the primary site after surgical excision, which may be performed for cosmetic purposes or to alleviate functional impairment.
- Ghosh SK, Bandyopadhyay D, Barma KD. Perifollicular nodule on the face of a young man. Indian J Dermatol Venereol Leprol. 2011;77:531-533.
- Gokalp H, Gurer MA, Alan S. Trichofolliculoma: a rare variant of hair follicle hamartoma. Dermatol Online J. 2013;19:19264.
- Choi CM, Lew BL, Sim WY. Multiple trichofolliculomas on unusual sites: a case report and review of the literature. Int J Dermatol. 2013;52:87-89.
- Misago N, Kimura T, Toda S, et al. A revaluation of trichofolliculoma: the histopathological and immunohistochemical features. Am J Dermatopathol. 2010;32:35-43.
- Stem JB, Stout DA. Trichofolliculoma showing perineural invasion. trichofolliculocarcinoma? Arch Dermatol. 1979;115:1003-1004.
The Diagnosis: Trichofolliculoma
Microscopic examination revealed a dilated cystic follicle that communicated with the skin surface (Figure). The follicle was lined with squamous epithelium and surrounded by numerous secondary follicles, many of which contained a hair shaft. A diagnosis of trichofolliculoma was made.

Clinically, the differential diagnosis of a flesh-colored papule on the scalp with prominent follicle includes dilated pore of Winer, epidermoid cyst, pilar sheath acanthoma, and trichoepithelioma.1,2 Multiple hair shafts present in a single follicle may be seen in pili multigemini, tufted folliculitis, trichostasis spinulosa, and trichofolliculoma. On histopathologic examination, a dilated central follicle surrounded with smaller secondary follicles was identified, consistent with trichofolliculoma.
Trichofolliculoma is a rare follicular hamartoma typically occurring on the face, scalp, or trunk as a solitary papule or nodule due to the proliferation of abnormal hair follicle stem cells.3,4 It may present as a flesh-colored nodule with a central pore that may drain sebum or contain white vellus hairs. Trichofolliculoma is considered a benign entity, despite one case report of malignant transformation.5 Biopsy is diagnostic and no further treatment is needed. Recurrence rarely occurs at the primary site after surgical excision, which may be performed for cosmetic purposes or to alleviate functional impairment.
The Diagnosis: Trichofolliculoma
Microscopic examination revealed a dilated cystic follicle that communicated with the skin surface (Figure). The follicle was lined with squamous epithelium and surrounded by numerous secondary follicles, many of which contained a hair shaft. A diagnosis of trichofolliculoma was made.

Clinically, the differential diagnosis of a flesh-colored papule on the scalp with prominent follicle includes dilated pore of Winer, epidermoid cyst, pilar sheath acanthoma, and trichoepithelioma.1,2 Multiple hair shafts present in a single follicle may be seen in pili multigemini, tufted folliculitis, trichostasis spinulosa, and trichofolliculoma. On histopathologic examination, a dilated central follicle surrounded with smaller secondary follicles was identified, consistent with trichofolliculoma.
Trichofolliculoma is a rare follicular hamartoma typically occurring on the face, scalp, or trunk as a solitary papule or nodule due to the proliferation of abnormal hair follicle stem cells.3,4 It may present as a flesh-colored nodule with a central pore that may drain sebum or contain white vellus hairs. Trichofolliculoma is considered a benign entity, despite one case report of malignant transformation.5 Biopsy is diagnostic and no further treatment is needed. Recurrence rarely occurs at the primary site after surgical excision, which may be performed for cosmetic purposes or to alleviate functional impairment.
- Ghosh SK, Bandyopadhyay D, Barma KD. Perifollicular nodule on the face of a young man. Indian J Dermatol Venereol Leprol. 2011;77:531-533.
- Gokalp H, Gurer MA, Alan S. Trichofolliculoma: a rare variant of hair follicle hamartoma. Dermatol Online J. 2013;19:19264.
- Choi CM, Lew BL, Sim WY. Multiple trichofolliculomas on unusual sites: a case report and review of the literature. Int J Dermatol. 2013;52:87-89.
- Misago N, Kimura T, Toda S, et al. A revaluation of trichofolliculoma: the histopathological and immunohistochemical features. Am J Dermatopathol. 2010;32:35-43.
- Stem JB, Stout DA. Trichofolliculoma showing perineural invasion. trichofolliculocarcinoma? Arch Dermatol. 1979;115:1003-1004.
- Ghosh SK, Bandyopadhyay D, Barma KD. Perifollicular nodule on the face of a young man. Indian J Dermatol Venereol Leprol. 2011;77:531-533.
- Gokalp H, Gurer MA, Alan S. Trichofolliculoma: a rare variant of hair follicle hamartoma. Dermatol Online J. 2013;19:19264.
- Choi CM, Lew BL, Sim WY. Multiple trichofolliculomas on unusual sites: a case report and review of the literature. Int J Dermatol. 2013;52:87-89.
- Misago N, Kimura T, Toda S, et al. A revaluation of trichofolliculoma: the histopathological and immunohistochemical features. Am J Dermatopathol. 2010;32:35-43.
- Stem JB, Stout DA. Trichofolliculoma showing perineural invasion. trichofolliculocarcinoma? Arch Dermatol. 1979;115:1003-1004.
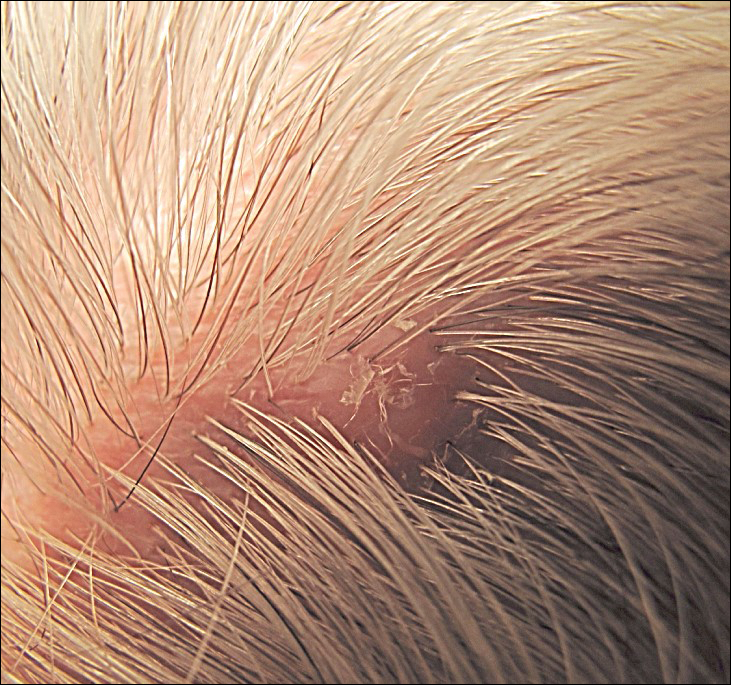
A 72-year-old man presented with a new asymptomatic 0.7-cm flesh-colored papule with a central tuft of white hairs on the posterior scalp. The remainder of the physical examination was unremarkable. Biopsy for histopathologic examination was performed to confirm diagnosis.
Alopecia may be permanent in one in four pediatric HSCT patients
CHICAGO – Late dermatologic manifestations in children who have received hematopoietic stem cell transplants may be more common than previously thought, according to results of a new study.
Johanna Song, MD, and her collaborators reported that, in their prospective pediatric study, 25% of patients had permanent alopecia and 16% had psoriasis, noting that late nonmalignant skin effects of hematopoietic stem cell transplantation (HSCT) have been studied primarily retrospectively, and in adults. Vitiligo and nail changes were also seen.
In a poster presentation at the World Congress of Dermatology, Dr. Song and her colleagues noted that these figures are higher than the previously reported pediatric rates of 1.7% for vitiligo and 15.6% for permanent alopecia. “Early recognition of these late effects can facilitate prompt and appropriate treatment, if desired,” they said.
The single-center, cross-sectional cohort study tracked pediatric patients over an 18-month period and included patients who were at least 1 year post allogeneic HSCT and had not relapsed. Patients who were not English speaking were excluded.
The median age of the 85 patients enrolled in the study was 13.8 years, and participants were a median of 3.6 years post transplant at the time of enrollment. The study’s analysis attempted to determine which patient, transplant, and disease factors might be associated with the late nonmalignant skin changes, according to Dr. Song, a resident dermatologist at Harvard University, Boston, and her colleagues.
Most – 52– of the patients (61.2%) had hematologic malignancies; 12 patients (14.1%) received their transplant for bone marrow failure, and 11 patients (12.5%) had immunodeficiency. Three patients (3.5%) received HSCT for other malignancies, and seven (8.2%) for nonmalignant diseases.
Diffuse hair thinning was seen in 13 (62%) of the 21 patients who had alopecia, while 11 (52%) of the patients with alopecia had an androgenetic hair loss pattern. Chronic graft versus host disease (GVHD), skin chronic GVHD, a HSCT regimen that included busulfan conditioning, and a family history of early male-pattern alopecia were all significantly associated with post-HSCT permanent alopecia (P less than .05 for all).
The patients with androgenetic alopecia may be experiencing an accelerated time course of a condition to which they are already genetically disposed, noted Dr. Song and her colleagues.
Psoriasis was commonly seen on the scalp, affecting 11 of the 14 patients with psoriasis (79%), and involved the face in five of the patients (36%). Just one patient had psoriasis elsewhere on the body. There was a nonsignificant trend towards human leukocyte antigen mismatch among patients who had psoriasis. Although “psoriasis may be a marker of persistent immune dysregulation,” the investigators said, they did not identify any associated risk factors that would point toward this mechanism in their analysis.
Twelve patients (14%) had vitiligo, with halo nevi seen in four of these patients. Children who were younger than age 10 years and those who received their transplant for primary immunodeficiency were significantly more likely to have vitiligo (P less than .05 for both). Specific possible mechanisms triggering vitiligo could include thymic dysfunction resulting in loss of self-tolerance, and donor alloreactivity against the patient’s host antigens, according to the authors.
Nail changes such as pterygium and nail pitting, ridging, or thickening were seen in just five patients, all of whom had chronic GVHD of the skin. “Nail changes are likely a result of persistent inflammation and immune dysregulation from chronic GVHD,” said Dr. Song.
These late effects “can significantly impact patients’ quality of life,” according to the authors, who called for larger studies that follow pediatric HSCT patients longitudinally, beginning before the transplant – and for more investigation into the pathogenesis of specific late effects.
Dr. Song reported no relevant conflicts of interest.
[email protected]
On Twitter @karioakes
CHICAGO – Late dermatologic manifestations in children who have received hematopoietic stem cell transplants may be more common than previously thought, according to results of a new study.
Johanna Song, MD, and her collaborators reported that, in their prospective pediatric study, 25% of patients had permanent alopecia and 16% had psoriasis, noting that late nonmalignant skin effects of hematopoietic stem cell transplantation (HSCT) have been studied primarily retrospectively, and in adults. Vitiligo and nail changes were also seen.
In a poster presentation at the World Congress of Dermatology, Dr. Song and her colleagues noted that these figures are higher than the previously reported pediatric rates of 1.7% for vitiligo and 15.6% for permanent alopecia. “Early recognition of these late effects can facilitate prompt and appropriate treatment, if desired,” they said.
The single-center, cross-sectional cohort study tracked pediatric patients over an 18-month period and included patients who were at least 1 year post allogeneic HSCT and had not relapsed. Patients who were not English speaking were excluded.
The median age of the 85 patients enrolled in the study was 13.8 years, and participants were a median of 3.6 years post transplant at the time of enrollment. The study’s analysis attempted to determine which patient, transplant, and disease factors might be associated with the late nonmalignant skin changes, according to Dr. Song, a resident dermatologist at Harvard University, Boston, and her colleagues.
Most – 52– of the patients (61.2%) had hematologic malignancies; 12 patients (14.1%) received their transplant for bone marrow failure, and 11 patients (12.5%) had immunodeficiency. Three patients (3.5%) received HSCT for other malignancies, and seven (8.2%) for nonmalignant diseases.
Diffuse hair thinning was seen in 13 (62%) of the 21 patients who had alopecia, while 11 (52%) of the patients with alopecia had an androgenetic hair loss pattern. Chronic graft versus host disease (GVHD), skin chronic GVHD, a HSCT regimen that included busulfan conditioning, and a family history of early male-pattern alopecia were all significantly associated with post-HSCT permanent alopecia (P less than .05 for all).
The patients with androgenetic alopecia may be experiencing an accelerated time course of a condition to which they are already genetically disposed, noted Dr. Song and her colleagues.
Psoriasis was commonly seen on the scalp, affecting 11 of the 14 patients with psoriasis (79%), and involved the face in five of the patients (36%). Just one patient had psoriasis elsewhere on the body. There was a nonsignificant trend towards human leukocyte antigen mismatch among patients who had psoriasis. Although “psoriasis may be a marker of persistent immune dysregulation,” the investigators said, they did not identify any associated risk factors that would point toward this mechanism in their analysis.
Twelve patients (14%) had vitiligo, with halo nevi seen in four of these patients. Children who were younger than age 10 years and those who received their transplant for primary immunodeficiency were significantly more likely to have vitiligo (P less than .05 for both). Specific possible mechanisms triggering vitiligo could include thymic dysfunction resulting in loss of self-tolerance, and donor alloreactivity against the patient’s host antigens, according to the authors.
Nail changes such as pterygium and nail pitting, ridging, or thickening were seen in just five patients, all of whom had chronic GVHD of the skin. “Nail changes are likely a result of persistent inflammation and immune dysregulation from chronic GVHD,” said Dr. Song.
These late effects “can significantly impact patients’ quality of life,” according to the authors, who called for larger studies that follow pediatric HSCT patients longitudinally, beginning before the transplant – and for more investigation into the pathogenesis of specific late effects.
Dr. Song reported no relevant conflicts of interest.
[email protected]
On Twitter @karioakes
CHICAGO – Late dermatologic manifestations in children who have received hematopoietic stem cell transplants may be more common than previously thought, according to results of a new study.
Johanna Song, MD, and her collaborators reported that, in their prospective pediatric study, 25% of patients had permanent alopecia and 16% had psoriasis, noting that late nonmalignant skin effects of hematopoietic stem cell transplantation (HSCT) have been studied primarily retrospectively, and in adults. Vitiligo and nail changes were also seen.
In a poster presentation at the World Congress of Dermatology, Dr. Song and her colleagues noted that these figures are higher than the previously reported pediatric rates of 1.7% for vitiligo and 15.6% for permanent alopecia. “Early recognition of these late effects can facilitate prompt and appropriate treatment, if desired,” they said.
The single-center, cross-sectional cohort study tracked pediatric patients over an 18-month period and included patients who were at least 1 year post allogeneic HSCT and had not relapsed. Patients who were not English speaking were excluded.
The median age of the 85 patients enrolled in the study was 13.8 years, and participants were a median of 3.6 years post transplant at the time of enrollment. The study’s analysis attempted to determine which patient, transplant, and disease factors might be associated with the late nonmalignant skin changes, according to Dr. Song, a resident dermatologist at Harvard University, Boston, and her colleagues.
Most – 52– of the patients (61.2%) had hematologic malignancies; 12 patients (14.1%) received their transplant for bone marrow failure, and 11 patients (12.5%) had immunodeficiency. Three patients (3.5%) received HSCT for other malignancies, and seven (8.2%) for nonmalignant diseases.
Diffuse hair thinning was seen in 13 (62%) of the 21 patients who had alopecia, while 11 (52%) of the patients with alopecia had an androgenetic hair loss pattern. Chronic graft versus host disease (GVHD), skin chronic GVHD, a HSCT regimen that included busulfan conditioning, and a family history of early male-pattern alopecia were all significantly associated with post-HSCT permanent alopecia (P less than .05 for all).
The patients with androgenetic alopecia may be experiencing an accelerated time course of a condition to which they are already genetically disposed, noted Dr. Song and her colleagues.
Psoriasis was commonly seen on the scalp, affecting 11 of the 14 patients with psoriasis (79%), and involved the face in five of the patients (36%). Just one patient had psoriasis elsewhere on the body. There was a nonsignificant trend towards human leukocyte antigen mismatch among patients who had psoriasis. Although “psoriasis may be a marker of persistent immune dysregulation,” the investigators said, they did not identify any associated risk factors that would point toward this mechanism in their analysis.
Twelve patients (14%) had vitiligo, with halo nevi seen in four of these patients. Children who were younger than age 10 years and those who received their transplant for primary immunodeficiency were significantly more likely to have vitiligo (P less than .05 for both). Specific possible mechanisms triggering vitiligo could include thymic dysfunction resulting in loss of self-tolerance, and donor alloreactivity against the patient’s host antigens, according to the authors.
Nail changes such as pterygium and nail pitting, ridging, or thickening were seen in just five patients, all of whom had chronic GVHD of the skin. “Nail changes are likely a result of persistent inflammation and immune dysregulation from chronic GVHD,” said Dr. Song.
These late effects “can significantly impact patients’ quality of life,” according to the authors, who called for larger studies that follow pediatric HSCT patients longitudinally, beginning before the transplant – and for more investigation into the pathogenesis of specific late effects.
Dr. Song reported no relevant conflicts of interest.
[email protected]
On Twitter @karioakes
AT WCPD 2017
Key clinical point:
Major finding: Permanent alopecia was seen in 25% of patients, and 16% had psoriasis a median of 3.6 years after transplant.
Data source: A single-center prospective study of 85 children in routine post-HSCT follow-up.
Disclosures: Dr. Song reported no conflicts of interest.