User login
Cancer care ‘transformed in space of a month’ because of pandemic
, the most “revolutionary” being a deep dive into telehealth, predicts Deborah Schrag, MD, MPH, a medical oncologist specializing in gastrointestinal cancers at the Dana Farber Cancer Institute in Boston, Massachusetts.
“In the space of a month, approaches and accepted norms of cancer care delivery have been transformed of necessity,” Schrag and colleagues write in an article published in JAMA on April 13.
“Most of these changes would not have occurred without the pandemic,” they add. They predict that some changes will last after the crisis is over.
“None of us want to be thrown in the deep end.... On the other hand, sometimes it works,” Schrag told Medscape Medical News.
“The in-person visit between patient and physician has been upended,” she said.
“I don’t think there’s any going back to the way it was before because cancer patients won’t stand for it,” she said. “They’re not going to drive in to get the results of a blood test.
“I think that on balance, of course, there are situations where you need eye-to-eye contact. No one wants to have an initial oncology meeting by telehealth – doctors or patients – that’s ridiculous,” she said. “But for follow-up visits, patients are now going to be more demanding, and doctors will be more willing.”
The “essential empathy” of oncologists can still “transcend the new physical barriers presented by masks and telehealth,” Schrag and colleagues comment.
“Doctors are figuring out how to deliver empathy by Zoom,” she told Medscape Medical News. “It’s not the same, but we all convey empathy to our elderly relatives over the phone.”
Pandemic impact on oncology
While the crisis has affected all of medicine – dismantling how care is delivered and forcing clinicians to make difficult decisions regarding triage – the fact that some cancers present an immediate threat to survival means that oncology “provides a lens into the major shifts currently underway in clinical care,” Schrag and colleagues write.
They illustrate the point by highlighting systemic chemotherapy, which is provided to a large proportion of patients with advanced cancer. The pandemic has tipped the risk-benefit ratio away from treatments that have a marginal effect on quality or quantity of life, they note. It has forced an “elimination of low-value treatments that were identified by the Choosing Wisely campaign,” the authors write. Up to now, the uptake of recommendations to eliminate these treatments has been slow.
“For example, for most metastatic solid tumors, chemotherapy beyond the third regimen does not improve survival for more than a few weeks; therefore, oncologists are advising supportive care instead. For patients receiving adjuvant therapy for curable cancers, delaying initiation or abbreviating the number of cycles is appropriate. Oncologists are postponing initiation of adjuvant chemotherapy for some estrogen receptor–negative stage II breast cancers by 8 weeks and administering 6 rather than 12 cycles of adjuvant chemotherapy for stage III colorectal cancers,” Schrag and colleagues write.
On the other hand, even in the epicenters of the pandemic, thus far, oncologists are still delivering cancer treatments that have the potential to cure and cannot safely be delayed, they point out. “This includes most patients with new diagnoses of acute leukemia, high-grade lymphoma, and those with chemotherapy-responsive tumors such as testicular, ovarian, and small cell lung cancer. Despite the risks, oncologists are not modifying such treatments because these cancers are likely more lethal than COVID-19.”
It’s the cancer patients who fall in between these two extremes who pose the biggest treatment challenge during this crisis – the patients for whom a delay would have “moderate clinically important adverse influence on quality of life or survival.” In these cases, oncologists are “prescribing marginally less effective regimens that have lower risk of precipitating hospitalization,” the authors note.
These treatments include the use of “white cell growth factor, more stringent neutrophil counts for proceeding with a next cycle of therapy, and omitting use of steroids to manage nausea.” In addition, where possible, oncologists are substituting oral agents for intravenous agents and “myriad other modifications to minimize visits and hospitalizations.”
Most hospitals and outpatient infusion centers now prohibit visitors from accompanying patients, and oncologists are prioritizing conversations with patients about advance directives, healthcare proxies, and end-of-life care preferences. Yet, even here, telehealth offers a new, enhanced layer to those conversations by enabling families to gather with their loved one and the doctor, she said.
This article first appeared on Medscape.com.
, the most “revolutionary” being a deep dive into telehealth, predicts Deborah Schrag, MD, MPH, a medical oncologist specializing in gastrointestinal cancers at the Dana Farber Cancer Institute in Boston, Massachusetts.
“In the space of a month, approaches and accepted norms of cancer care delivery have been transformed of necessity,” Schrag and colleagues write in an article published in JAMA on April 13.
“Most of these changes would not have occurred without the pandemic,” they add. They predict that some changes will last after the crisis is over.
“None of us want to be thrown in the deep end.... On the other hand, sometimes it works,” Schrag told Medscape Medical News.
“The in-person visit between patient and physician has been upended,” she said.
“I don’t think there’s any going back to the way it was before because cancer patients won’t stand for it,” she said. “They’re not going to drive in to get the results of a blood test.
“I think that on balance, of course, there are situations where you need eye-to-eye contact. No one wants to have an initial oncology meeting by telehealth – doctors or patients – that’s ridiculous,” she said. “But for follow-up visits, patients are now going to be more demanding, and doctors will be more willing.”
The “essential empathy” of oncologists can still “transcend the new physical barriers presented by masks and telehealth,” Schrag and colleagues comment.
“Doctors are figuring out how to deliver empathy by Zoom,” she told Medscape Medical News. “It’s not the same, but we all convey empathy to our elderly relatives over the phone.”
Pandemic impact on oncology
While the crisis has affected all of medicine – dismantling how care is delivered and forcing clinicians to make difficult decisions regarding triage – the fact that some cancers present an immediate threat to survival means that oncology “provides a lens into the major shifts currently underway in clinical care,” Schrag and colleagues write.
They illustrate the point by highlighting systemic chemotherapy, which is provided to a large proportion of patients with advanced cancer. The pandemic has tipped the risk-benefit ratio away from treatments that have a marginal effect on quality or quantity of life, they note. It has forced an “elimination of low-value treatments that were identified by the Choosing Wisely campaign,” the authors write. Up to now, the uptake of recommendations to eliminate these treatments has been slow.
“For example, for most metastatic solid tumors, chemotherapy beyond the third regimen does not improve survival for more than a few weeks; therefore, oncologists are advising supportive care instead. For patients receiving adjuvant therapy for curable cancers, delaying initiation or abbreviating the number of cycles is appropriate. Oncologists are postponing initiation of adjuvant chemotherapy for some estrogen receptor–negative stage II breast cancers by 8 weeks and administering 6 rather than 12 cycles of adjuvant chemotherapy for stage III colorectal cancers,” Schrag and colleagues write.
On the other hand, even in the epicenters of the pandemic, thus far, oncologists are still delivering cancer treatments that have the potential to cure and cannot safely be delayed, they point out. “This includes most patients with new diagnoses of acute leukemia, high-grade lymphoma, and those with chemotherapy-responsive tumors such as testicular, ovarian, and small cell lung cancer. Despite the risks, oncologists are not modifying such treatments because these cancers are likely more lethal than COVID-19.”
It’s the cancer patients who fall in between these two extremes who pose the biggest treatment challenge during this crisis – the patients for whom a delay would have “moderate clinically important adverse influence on quality of life or survival.” In these cases, oncologists are “prescribing marginally less effective regimens that have lower risk of precipitating hospitalization,” the authors note.
These treatments include the use of “white cell growth factor, more stringent neutrophil counts for proceeding with a next cycle of therapy, and omitting use of steroids to manage nausea.” In addition, where possible, oncologists are substituting oral agents for intravenous agents and “myriad other modifications to minimize visits and hospitalizations.”
Most hospitals and outpatient infusion centers now prohibit visitors from accompanying patients, and oncologists are prioritizing conversations with patients about advance directives, healthcare proxies, and end-of-life care preferences. Yet, even here, telehealth offers a new, enhanced layer to those conversations by enabling families to gather with their loved one and the doctor, she said.
This article first appeared on Medscape.com.
, the most “revolutionary” being a deep dive into telehealth, predicts Deborah Schrag, MD, MPH, a medical oncologist specializing in gastrointestinal cancers at the Dana Farber Cancer Institute in Boston, Massachusetts.
“In the space of a month, approaches and accepted norms of cancer care delivery have been transformed of necessity,” Schrag and colleagues write in an article published in JAMA on April 13.
“Most of these changes would not have occurred without the pandemic,” they add. They predict that some changes will last after the crisis is over.
“None of us want to be thrown in the deep end.... On the other hand, sometimes it works,” Schrag told Medscape Medical News.
“The in-person visit between patient and physician has been upended,” she said.
“I don’t think there’s any going back to the way it was before because cancer patients won’t stand for it,” she said. “They’re not going to drive in to get the results of a blood test.
“I think that on balance, of course, there are situations where you need eye-to-eye contact. No one wants to have an initial oncology meeting by telehealth – doctors or patients – that’s ridiculous,” she said. “But for follow-up visits, patients are now going to be more demanding, and doctors will be more willing.”
The “essential empathy” of oncologists can still “transcend the new physical barriers presented by masks and telehealth,” Schrag and colleagues comment.
“Doctors are figuring out how to deliver empathy by Zoom,” she told Medscape Medical News. “It’s not the same, but we all convey empathy to our elderly relatives over the phone.”
Pandemic impact on oncology
While the crisis has affected all of medicine – dismantling how care is delivered and forcing clinicians to make difficult decisions regarding triage – the fact that some cancers present an immediate threat to survival means that oncology “provides a lens into the major shifts currently underway in clinical care,” Schrag and colleagues write.
They illustrate the point by highlighting systemic chemotherapy, which is provided to a large proportion of patients with advanced cancer. The pandemic has tipped the risk-benefit ratio away from treatments that have a marginal effect on quality or quantity of life, they note. It has forced an “elimination of low-value treatments that were identified by the Choosing Wisely campaign,” the authors write. Up to now, the uptake of recommendations to eliminate these treatments has been slow.
“For example, for most metastatic solid tumors, chemotherapy beyond the third regimen does not improve survival for more than a few weeks; therefore, oncologists are advising supportive care instead. For patients receiving adjuvant therapy for curable cancers, delaying initiation or abbreviating the number of cycles is appropriate. Oncologists are postponing initiation of adjuvant chemotherapy for some estrogen receptor–negative stage II breast cancers by 8 weeks and administering 6 rather than 12 cycles of adjuvant chemotherapy for stage III colorectal cancers,” Schrag and colleagues write.
On the other hand, even in the epicenters of the pandemic, thus far, oncologists are still delivering cancer treatments that have the potential to cure and cannot safely be delayed, they point out. “This includes most patients with new diagnoses of acute leukemia, high-grade lymphoma, and those with chemotherapy-responsive tumors such as testicular, ovarian, and small cell lung cancer. Despite the risks, oncologists are not modifying such treatments because these cancers are likely more lethal than COVID-19.”
It’s the cancer patients who fall in between these two extremes who pose the biggest treatment challenge during this crisis – the patients for whom a delay would have “moderate clinically important adverse influence on quality of life or survival.” In these cases, oncologists are “prescribing marginally less effective regimens that have lower risk of precipitating hospitalization,” the authors note.
These treatments include the use of “white cell growth factor, more stringent neutrophil counts for proceeding with a next cycle of therapy, and omitting use of steroids to manage nausea.” In addition, where possible, oncologists are substituting oral agents for intravenous agents and “myriad other modifications to minimize visits and hospitalizations.”
Most hospitals and outpatient infusion centers now prohibit visitors from accompanying patients, and oncologists are prioritizing conversations with patients about advance directives, healthcare proxies, and end-of-life care preferences. Yet, even here, telehealth offers a new, enhanced layer to those conversations by enabling families to gather with their loved one and the doctor, she said.
This article first appeared on Medscape.com.
Pandemic strains blood supply for COVID-19 and noninfected patients
The COVID-19 pandemic is putting a strain on the blood supply and could be putting people – including those who normally get transfusions, such as patients with sickle cell disease and cancer – at risk.
“Around the beginning of March, the hematology community got wind of what was going on because the blood banks were saying think about your patients and begin to restrict blood usage because we are expecting an increase in usage for COVID-positive ICU patients,” Ifeyinwa (Ify) Osunkwo, MD, a specialist in hematology and sickle cell disease at Levine Cancer Institute in Charlotte, N.C., said in an interview.
“I think that was the first call to arms around hematology ... you don’t want to shortchange somebody who is well and who is being sustained by life-giving transfusions and cut out their transfusion therapy because you are hoping to use the blood for people who are coming in with COVID-19,” she continued. “That is an ethical dilemma that no doctor wants to have to go through. But the reality is we have to do something to make it work for everybody.”
And the timing of the social restrictions due to the pandemic has added additional strain on the blood supply.
“Over the winter, traditionally, blood drives slow down because of the flu and different viruses,” she noted. “The spring and the summer are when we see the biggest recruitment and uptake of blood donation. COVID-19 hit [and] a lot of the blood drives that were traditionally scheduled to supply blood for the country have been canceled because of the new guidance for social distancing.”
Another big source of blood are health care professionals themselves and they may not be able to donate because of the extra hours being worked because of the pandemic.
In speaking about the needs for traditional patients such as those who are dealing with cancer or leukemia or sickle cell diseases as well as those who are being treated for COVID-19 in North Carolina, “we are not at the critical point, but I am a little bit nervous that we may get there because they are not going to up the usual blood drives anytime this summer. We project [sometime] in the fall, but maybe not even then. So there needs to be a significant call-out for people to make every effort to donate blood,” said Dr. Osunkwo. She added that in places such as New York City that are hot spots for the COVID-19 outbreak, the need is likely a lot greater.
She recalled a recent incident at a New York hospital that highlighted how those managing blood supplies are being restrictive and how this could be harming patients.
“A sickle cell patient came in with COVID-19 and the treatment recommendation was do a red blood cell exchange but the blood bank was nervous about getting enough blood to supply for that exchange transfusion,” she said, noting that the doctor still went to bat for that patient to get the needed treatment. “We gave her the supporting evidence that when you are on treatment for sickle cell disease, you tend to do better if you get COVID-19 or any other viral infection. The symptoms of COVID-19 in sickle cell disease is acute chest syndrome, for which the treatment is red blood cell exchange. Not doing that for [these patients] is really not giving them the optimal way of managing their disease, and managing their disease in the setting of COVID-19.”
To that end, Dr. Osunkwo stressed that doctors need to be doing all they can to get the word out that blood is needed and that the American Red Cross and other donation organizations are making it safe for people to donate. She has been using social media to highlight when her fellow doctors and others make donations as a way to motivate individuals.
“Everybody can do something during this pandemic,” she said. “Don’t feel like you are not working, that you are not a frontline worker, that you have nothing to contribute. You can donate blood. Your cousin can donate blood. You can tell your friends, your neighbors, your relatives, your enemies to go donate. We will take every kind of blood we can get because people are needing it more now. Even though we canceled elective surgeries, my patients when they get COVID-19, they need more blood ... than they usually do during their regular sickle cell admission. It is going to be the same for people who have other blood disorders like cancer and leukemia. We can’t stop life-saving treatments just because we have the COVID pandemic.”
Dr. Osunkwo also praised recent actions taken by the Food and Drug Administration to lessen some of the deferral periods for when an individual can donate.
The FDA on April 2 issued three sets of revised recommendations aimed at getting more people eligible to donate blood. All of the revised recommendations will remain in effect after the COVID-19 health emergency is declared over.
The first revised recommendation makes changes to December 2015 guidance.
For male blood donors who would have been deferred for having sex with another male partner, the deferral period has been reduced from 12 months to 3 months. That deferral period change also applies to female donors who had sex with a man who had sex with another man as well as for those with recent tattoos and piercings.
The second recommendation revises guidance from August 2013 and relates to the risk of transfusion-transmitted malaria.
Under the new recommendations, for those who traveled to malaria-endemic areas (and are residents of malaria non-endemic countries), the FDA is lowering the recommended deferral period from 12 months to 3 months, and also provides notices of an alternate procedure that permits donations without a deferral period provided the blood components are pathogen-reduced using an FDA-approved pathogen reduction device.
The third recommendation finalizes draft guidance from January that eliminates the referral period for donors who spent time in certain European countries or were on military bases in Europe and were previously considered to have been exposed to a potential risk of transmission of Creutzfeldt-Jakob Disease or Variant Creutzfeldt-Jakob Disease.
Dr. Osunkwo reports consultancy and being on the speakers bureau and participating in the advisory board for Novartis, and relationships with a variety of other pharmaceutical companies. She is the editor-in-chief for Hematology News.
The COVID-19 pandemic is putting a strain on the blood supply and could be putting people – including those who normally get transfusions, such as patients with sickle cell disease and cancer – at risk.
“Around the beginning of March, the hematology community got wind of what was going on because the blood banks were saying think about your patients and begin to restrict blood usage because we are expecting an increase in usage for COVID-positive ICU patients,” Ifeyinwa (Ify) Osunkwo, MD, a specialist in hematology and sickle cell disease at Levine Cancer Institute in Charlotte, N.C., said in an interview.
“I think that was the first call to arms around hematology ... you don’t want to shortchange somebody who is well and who is being sustained by life-giving transfusions and cut out their transfusion therapy because you are hoping to use the blood for people who are coming in with COVID-19,” she continued. “That is an ethical dilemma that no doctor wants to have to go through. But the reality is we have to do something to make it work for everybody.”
And the timing of the social restrictions due to the pandemic has added additional strain on the blood supply.
“Over the winter, traditionally, blood drives slow down because of the flu and different viruses,” she noted. “The spring and the summer are when we see the biggest recruitment and uptake of blood donation. COVID-19 hit [and] a lot of the blood drives that were traditionally scheduled to supply blood for the country have been canceled because of the new guidance for social distancing.”
Another big source of blood are health care professionals themselves and they may not be able to donate because of the extra hours being worked because of the pandemic.
In speaking about the needs for traditional patients such as those who are dealing with cancer or leukemia or sickle cell diseases as well as those who are being treated for COVID-19 in North Carolina, “we are not at the critical point, but I am a little bit nervous that we may get there because they are not going to up the usual blood drives anytime this summer. We project [sometime] in the fall, but maybe not even then. So there needs to be a significant call-out for people to make every effort to donate blood,” said Dr. Osunkwo. She added that in places such as New York City that are hot spots for the COVID-19 outbreak, the need is likely a lot greater.
She recalled a recent incident at a New York hospital that highlighted how those managing blood supplies are being restrictive and how this could be harming patients.
“A sickle cell patient came in with COVID-19 and the treatment recommendation was do a red blood cell exchange but the blood bank was nervous about getting enough blood to supply for that exchange transfusion,” she said, noting that the doctor still went to bat for that patient to get the needed treatment. “We gave her the supporting evidence that when you are on treatment for sickle cell disease, you tend to do better if you get COVID-19 or any other viral infection. The symptoms of COVID-19 in sickle cell disease is acute chest syndrome, for which the treatment is red blood cell exchange. Not doing that for [these patients] is really not giving them the optimal way of managing their disease, and managing their disease in the setting of COVID-19.”
To that end, Dr. Osunkwo stressed that doctors need to be doing all they can to get the word out that blood is needed and that the American Red Cross and other donation organizations are making it safe for people to donate. She has been using social media to highlight when her fellow doctors and others make donations as a way to motivate individuals.
“Everybody can do something during this pandemic,” she said. “Don’t feel like you are not working, that you are not a frontline worker, that you have nothing to contribute. You can donate blood. Your cousin can donate blood. You can tell your friends, your neighbors, your relatives, your enemies to go donate. We will take every kind of blood we can get because people are needing it more now. Even though we canceled elective surgeries, my patients when they get COVID-19, they need more blood ... than they usually do during their regular sickle cell admission. It is going to be the same for people who have other blood disorders like cancer and leukemia. We can’t stop life-saving treatments just because we have the COVID pandemic.”
Dr. Osunkwo also praised recent actions taken by the Food and Drug Administration to lessen some of the deferral periods for when an individual can donate.
The FDA on April 2 issued three sets of revised recommendations aimed at getting more people eligible to donate blood. All of the revised recommendations will remain in effect after the COVID-19 health emergency is declared over.
The first revised recommendation makes changes to December 2015 guidance.
For male blood donors who would have been deferred for having sex with another male partner, the deferral period has been reduced from 12 months to 3 months. That deferral period change also applies to female donors who had sex with a man who had sex with another man as well as for those with recent tattoos and piercings.
The second recommendation revises guidance from August 2013 and relates to the risk of transfusion-transmitted malaria.
Under the new recommendations, for those who traveled to malaria-endemic areas (and are residents of malaria non-endemic countries), the FDA is lowering the recommended deferral period from 12 months to 3 months, and also provides notices of an alternate procedure that permits donations without a deferral period provided the blood components are pathogen-reduced using an FDA-approved pathogen reduction device.
The third recommendation finalizes draft guidance from January that eliminates the referral period for donors who spent time in certain European countries or were on military bases in Europe and were previously considered to have been exposed to a potential risk of transmission of Creutzfeldt-Jakob Disease or Variant Creutzfeldt-Jakob Disease.
Dr. Osunkwo reports consultancy and being on the speakers bureau and participating in the advisory board for Novartis, and relationships with a variety of other pharmaceutical companies. She is the editor-in-chief for Hematology News.
The COVID-19 pandemic is putting a strain on the blood supply and could be putting people – including those who normally get transfusions, such as patients with sickle cell disease and cancer – at risk.
“Around the beginning of March, the hematology community got wind of what was going on because the blood banks were saying think about your patients and begin to restrict blood usage because we are expecting an increase in usage for COVID-positive ICU patients,” Ifeyinwa (Ify) Osunkwo, MD, a specialist in hematology and sickle cell disease at Levine Cancer Institute in Charlotte, N.C., said in an interview.
“I think that was the first call to arms around hematology ... you don’t want to shortchange somebody who is well and who is being sustained by life-giving transfusions and cut out their transfusion therapy because you are hoping to use the blood for people who are coming in with COVID-19,” she continued. “That is an ethical dilemma that no doctor wants to have to go through. But the reality is we have to do something to make it work for everybody.”
And the timing of the social restrictions due to the pandemic has added additional strain on the blood supply.
“Over the winter, traditionally, blood drives slow down because of the flu and different viruses,” she noted. “The spring and the summer are when we see the biggest recruitment and uptake of blood donation. COVID-19 hit [and] a lot of the blood drives that were traditionally scheduled to supply blood for the country have been canceled because of the new guidance for social distancing.”
Another big source of blood are health care professionals themselves and they may not be able to donate because of the extra hours being worked because of the pandemic.
In speaking about the needs for traditional patients such as those who are dealing with cancer or leukemia or sickle cell diseases as well as those who are being treated for COVID-19 in North Carolina, “we are not at the critical point, but I am a little bit nervous that we may get there because they are not going to up the usual blood drives anytime this summer. We project [sometime] in the fall, but maybe not even then. So there needs to be a significant call-out for people to make every effort to donate blood,” said Dr. Osunkwo. She added that in places such as New York City that are hot spots for the COVID-19 outbreak, the need is likely a lot greater.
She recalled a recent incident at a New York hospital that highlighted how those managing blood supplies are being restrictive and how this could be harming patients.
“A sickle cell patient came in with COVID-19 and the treatment recommendation was do a red blood cell exchange but the blood bank was nervous about getting enough blood to supply for that exchange transfusion,” she said, noting that the doctor still went to bat for that patient to get the needed treatment. “We gave her the supporting evidence that when you are on treatment for sickle cell disease, you tend to do better if you get COVID-19 or any other viral infection. The symptoms of COVID-19 in sickle cell disease is acute chest syndrome, for which the treatment is red blood cell exchange. Not doing that for [these patients] is really not giving them the optimal way of managing their disease, and managing their disease in the setting of COVID-19.”
To that end, Dr. Osunkwo stressed that doctors need to be doing all they can to get the word out that blood is needed and that the American Red Cross and other donation organizations are making it safe for people to donate. She has been using social media to highlight when her fellow doctors and others make donations as a way to motivate individuals.
“Everybody can do something during this pandemic,” she said. “Don’t feel like you are not working, that you are not a frontline worker, that you have nothing to contribute. You can donate blood. Your cousin can donate blood. You can tell your friends, your neighbors, your relatives, your enemies to go donate. We will take every kind of blood we can get because people are needing it more now. Even though we canceled elective surgeries, my patients when they get COVID-19, they need more blood ... than they usually do during their regular sickle cell admission. It is going to be the same for people who have other blood disorders like cancer and leukemia. We can’t stop life-saving treatments just because we have the COVID pandemic.”
Dr. Osunkwo also praised recent actions taken by the Food and Drug Administration to lessen some of the deferral periods for when an individual can donate.
The FDA on April 2 issued three sets of revised recommendations aimed at getting more people eligible to donate blood. All of the revised recommendations will remain in effect after the COVID-19 health emergency is declared over.
The first revised recommendation makes changes to December 2015 guidance.
For male blood donors who would have been deferred for having sex with another male partner, the deferral period has been reduced from 12 months to 3 months. That deferral period change also applies to female donors who had sex with a man who had sex with another man as well as for those with recent tattoos and piercings.
The second recommendation revises guidance from August 2013 and relates to the risk of transfusion-transmitted malaria.
Under the new recommendations, for those who traveled to malaria-endemic areas (and are residents of malaria non-endemic countries), the FDA is lowering the recommended deferral period from 12 months to 3 months, and also provides notices of an alternate procedure that permits donations without a deferral period provided the blood components are pathogen-reduced using an FDA-approved pathogen reduction device.
The third recommendation finalizes draft guidance from January that eliminates the referral period for donors who spent time in certain European countries or were on military bases in Europe and were previously considered to have been exposed to a potential risk of transmission of Creutzfeldt-Jakob Disease or Variant Creutzfeldt-Jakob Disease.
Dr. Osunkwo reports consultancy and being on the speakers bureau and participating in the advisory board for Novartis, and relationships with a variety of other pharmaceutical companies. She is the editor-in-chief for Hematology News.
Severe COVID-19 may lower hemoglobin levels
A meta-analysis of four applicable studies found that the hemoglobin value was significantly lower in COVID-19 patients with severe disease, compared with those with milder forms, according to a letter to the editor of Hematology Transfusion and Cell Therapy by Giuseppe Lippi, MD, of the University of Verona (Italy) and colleague.
The four studies comprised 1,210 COVID-19 patients (224 with severe disease; 18.5%). The primary endpoint was defined as a composite of admission to the ICU, need of mechanical ventilation or death. The heterogeneity among the studies was high.
Overall, the hemoglobin value was found to be significantly lower in COVID-19 patients with severe disease than in those with milder forms, yielding a weighted mean difference of −7.1 g/L, with a 95% confidence interval of −8.3 g/L to −5.9 g/L.
“Initial assessment and longitudinal monitoring of hemoglobin values seems advisable in patients with the SARS-CoV-2 infection, whereby a progressive decrease in the hemoglobin concentration may reflect a worse clinical progression,” the authors stated. They also suggested that studies should be “urgently planned to assess whether transfusion support (e.g., with administration of blood or packed red blood cells) may be helpful in this clinical setting to prevent evolution into severe disease and death.”
The authors declared the had no conflicts of interest.
SOURCE: Lippi G et al. Hematol Transfus Cell Ther. 2020 Apr 11; doi:10.1016/j.htct.2020.03.001.
A meta-analysis of four applicable studies found that the hemoglobin value was significantly lower in COVID-19 patients with severe disease, compared with those with milder forms, according to a letter to the editor of Hematology Transfusion and Cell Therapy by Giuseppe Lippi, MD, of the University of Verona (Italy) and colleague.
The four studies comprised 1,210 COVID-19 patients (224 with severe disease; 18.5%). The primary endpoint was defined as a composite of admission to the ICU, need of mechanical ventilation or death. The heterogeneity among the studies was high.
Overall, the hemoglobin value was found to be significantly lower in COVID-19 patients with severe disease than in those with milder forms, yielding a weighted mean difference of −7.1 g/L, with a 95% confidence interval of −8.3 g/L to −5.9 g/L.
“Initial assessment and longitudinal monitoring of hemoglobin values seems advisable in patients with the SARS-CoV-2 infection, whereby a progressive decrease in the hemoglobin concentration may reflect a worse clinical progression,” the authors stated. They also suggested that studies should be “urgently planned to assess whether transfusion support (e.g., with administration of blood or packed red blood cells) may be helpful in this clinical setting to prevent evolution into severe disease and death.”
The authors declared the had no conflicts of interest.
SOURCE: Lippi G et al. Hematol Transfus Cell Ther. 2020 Apr 11; doi:10.1016/j.htct.2020.03.001.
A meta-analysis of four applicable studies found that the hemoglobin value was significantly lower in COVID-19 patients with severe disease, compared with those with milder forms, according to a letter to the editor of Hematology Transfusion and Cell Therapy by Giuseppe Lippi, MD, of the University of Verona (Italy) and colleague.
The four studies comprised 1,210 COVID-19 patients (224 with severe disease; 18.5%). The primary endpoint was defined as a composite of admission to the ICU, need of mechanical ventilation or death. The heterogeneity among the studies was high.
Overall, the hemoglobin value was found to be significantly lower in COVID-19 patients with severe disease than in those with milder forms, yielding a weighted mean difference of −7.1 g/L, with a 95% confidence interval of −8.3 g/L to −5.9 g/L.
“Initial assessment and longitudinal monitoring of hemoglobin values seems advisable in patients with the SARS-CoV-2 infection, whereby a progressive decrease in the hemoglobin concentration may reflect a worse clinical progression,” the authors stated. They also suggested that studies should be “urgently planned to assess whether transfusion support (e.g., with administration of blood or packed red blood cells) may be helpful in this clinical setting to prevent evolution into severe disease and death.”
The authors declared the had no conflicts of interest.
SOURCE: Lippi G et al. Hematol Transfus Cell Ther. 2020 Apr 11; doi:10.1016/j.htct.2020.03.001.
FROM HEMATOLOGY, TRANSFUSION AND CELL THERAPY
Most e-consults not followed by specialist visit
Studies have shown that e-consults increase access to specialist care and primary care physician (PCP) education, according to research published in the Annals of Internal Medicine (2020. Apr 14. doi: 10.7326/M19-3852) by Salman Ahmed, MD, and colleagues.
These resources are already being frequently used by physicians, but more often by general internists and hospitalists than by subspecialists, according to a recent survey by the American College of Physicians. That survey found that 42% of its respondents are using e-consults and that subspecialists’ use is less common primarily because of the lack of access to e-consult technology.
What hasn’t been widely researched are the effects of large-scale e-consult programs, said Dr. Ahmed, who is associate physician in the renal division at Brigham and Women’s Hospital, Boston, in an interview.
For frontline providers such as PCPs, e-consults are a way to quickly seek out answers to clinical questions from specialists. In turn, the specialist can help a wider pool of participants, he noted.
The findings of Dr. Ahmed’s study, which included several academic centers and hospitals affiliated with Partners HealthCare System, a nonprofit network in eastern Massachusetts that includes Brigham and Women’s Hospital, used several metrics to analyze the appropriateness and utility of e-consults across a range of specialties. An e-consult was considered useful if it resulted in the avoidance of a visit to a specialist, which was defined as the absence of an in-person visit to the type of specialist consulted electronically for 120 days. An e-consult was considered appropriate if it met the following four criteria.
- It could not be answered by referring to society guidelines or widely available, evidence-based summary sources.
- It did not seek logistic information, such as where to have a specific laboratory test done.
- It did not include a question of high urgency.
- The medical complexity of the clinical situation was not substantial enough to warrant an in-person consultation.
The investigators examined e-consult inquiries to mostly physician health care providers in five specialties – hematology, infectious disease, dermatology, rheumatology, and psychiatry – over a year.
High rates of appropriateness
The search spanned 6,512 eligible e-consults from 1,096 referring providers to 121 specialist consultants. Narrowing their search to 741 records with complete data, the investigators found that 70.2% of these consults met the criteria for appropriateness. In an analysis of four reviewers blinded to each other’s results, raters agreed on the appropriateness of 94% of e-consults.
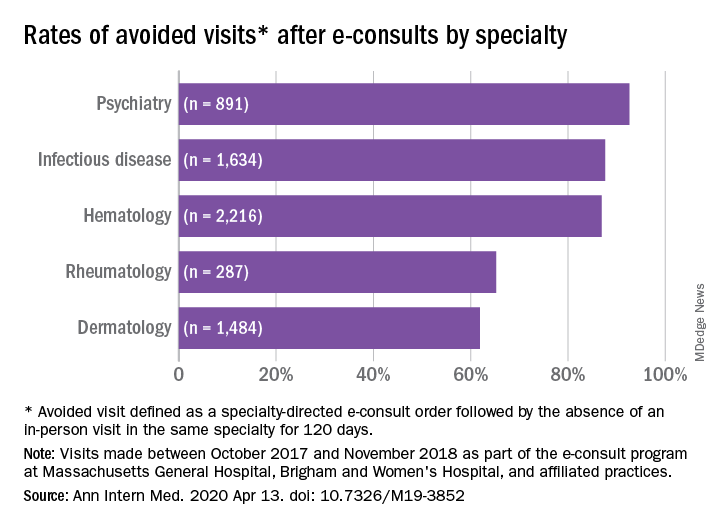
Across specialties, more than 81% of e-consults were associated with avoided in-person visits.
The reasons for most e-consults were to seek answers to questions about diagnosis, therapeutics, or patient inquiries, or to request further education by PCPs.
“Across all specialties, the most common reasons an e-consult was not considered appropriate were failing the point-of-care resource test and asking a question of inappropriately high complexity,” the authors summarized.
Physicians and PCPs from tertiary care practices made up the majority of referring providers, with turnaround time for consults averaging 24 hours across specialties.
Rates of appropriateness, content, patient demographics, and timeliness of e-consult responses varied among the four specialties. Those with high avoidance of visits rates tended to have high appropriateness rates, indicating that some specialties may be more conducive to e-consults than others, the authors noted. Psychiatry and hematology had the highest proportion of appropriate e-consults (77.9% and 73.3% respectively). Rheumatology had the lowest proportion of appropriate e-consults and one of the lowest rates of avoided in-person visits, and dermatology had the lowest rate of avoided in-person visits, at 61.9%.
The majority (93%) of e-consults sought in psychiatry were therapy related, whereas 88.4% of the e-consult questions in rheumatology related to diagnosis.
“Questions about diagnosis were less likely to be answerable via e-consult, which suggests that to provide diagnoses, consultants may wish to engage with the patient directly,” Dr. Ahmed said in an interview.
Infectious disease specialists seemed to be the fastest responders, with nearly 90% of their consultations having been answered within a day. Dermatology specialists had the distinction of having the youngest e-consult patients (mean age, 38.6 years).
PCPs weigh in on results
Physicians said in interviews that the study data reflects their own positive experiences with e-consults.
“Although I don’t always think [an e-consult] is able to fully prevent the specialist visit, it does allow the specialist to provide recommendations for work-up that can be done prior to the specialist visit,” said Santina Wheat MD, a family physician at Erie Family Health Center in Chicago. This reduces the time in which the consult is placed to when effective treatment can take place.
Patients who may have to wait months or even years to see a specialty doctor, benefit from e-consults, said Dr. Wheat, who is also a member of the editorial advisory board of Family Practice News. “As part of an organization that does e-consults to another hospital with a different electronic medical record, the e-consult increases the likelihood that all of the clinical information reaches the specialists and prevents tests from being repeated.”
Starting an e-consult may also increase the likelihood that the patient quickly sees a specialist at the contracted hospital, she added.
Sarah G. Candler, MD, said in an interview that she also sees e-consults as an essential tool. “When patients present with rare, complex, or atypical pictures, I find it helpful to have specialists weigh in. The e-consult helps me ensure that I work to the top of my abilities as an internist,” said Dr. Candler, who is practice medical director and physician director of academic relations at Iora Primary Care, Northside Clinic, Houston. However, she did not agree with the study’s avoided in-person visits metric for assessing utility.
“In some cases, the end result of an e-consult is a referral for an in-person evaluation, and the role of the e-consult is to ensure that I have done my due diligence as a primary care doctor asking the correct questions, getting the appropriate work-up completed, and referring to the appropriate specialty for next steps, when necessary,” noted Dr. Candler, who also serves on the editorial advisory board of Internal Medicine News.
Financial considerations
The study’s authors suggested taking a closer look at standardizing payment for the use of e-consults and developing appropriateness criteria for them.
Health systems could use such criteria to study what makes an e-consult useful and how to best utilize this tool, Dr. Ahmed said in an interview.
“Compensation models that promote high-quality, effective, and efficient e-consults are needed to reinforce the ability of health systems to optimize the mix of e-consults and in-person visits,” Dr. Ahmed and colleagues suggested.
Because not all patient care requires e-consults, the model makes the most sense in practices that already participate in value-based payment programs. In these types of programs, the cost can be shared according to the variable risk and patient need for the service, Dr. Candler explained.
“I have been fortunate to work in two different systems that function in this way, which means that e-consults have been readily available and encouraged-both to improve patient care and decrease overall cost by decreasing unnecessary testing or specialist referral,” she said.
Dr. Wheat said that the managed care organization affiliated with her practice seems to be saving money with e-consults, as it decreases the need to pay for specialist visits in some instances and for repeated work-ups.
Future studies
The study’s cohort represented just one large health care system with a shared electronic health record. “Single-system descriptive studies, such as that of Ahmed and colleagues, are particularly useful for local evaluation and quality improvement efforts,” Varsha G. Vimalananda, MD, and B. Graeme Fincke, MD, both of the Center for Healthcare Organization and Implementation Research at Bedford (Mass.) Veterans Affairs Hospital, wrote in a related editorial.
“However, we need innovative approaches to evaluation that estimate the effect of e-consults on quality and cost of care across health care systems and over time. Implementation studies can help to identify key contributors to success,” the editorialists wrote.
One of the study authors, reported receiving personal fees from Bayer outside the submitted work. The other authors of the paper and the authors of the editorial reported no conflicts of interest. Dr. Candler said her employer contracts with an e-consult service, but that she is not compensated for use of the service. She is also a coeditor of Annals of Internal Medicine’s blog, “Fresh Look.”
SOURCE: Ahmed S et al. Ann Intern Med. 2020 Apr 14. doi: 10.7326/M19-3852.

Studies have shown that e-consults increase access to specialist care and primary care physician (PCP) education, according to research published in the Annals of Internal Medicine (2020. Apr 14. doi: 10.7326/M19-3852) by Salman Ahmed, MD, and colleagues.
These resources are already being frequently used by physicians, but more often by general internists and hospitalists than by subspecialists, according to a recent survey by the American College of Physicians. That survey found that 42% of its respondents are using e-consults and that subspecialists’ use is less common primarily because of the lack of access to e-consult technology.
What hasn’t been widely researched are the effects of large-scale e-consult programs, said Dr. Ahmed, who is associate physician in the renal division at Brigham and Women’s Hospital, Boston, in an interview.
For frontline providers such as PCPs, e-consults are a way to quickly seek out answers to clinical questions from specialists. In turn, the specialist can help a wider pool of participants, he noted.
The findings of Dr. Ahmed’s study, which included several academic centers and hospitals affiliated with Partners HealthCare System, a nonprofit network in eastern Massachusetts that includes Brigham and Women’s Hospital, used several metrics to analyze the appropriateness and utility of e-consults across a range of specialties. An e-consult was considered useful if it resulted in the avoidance of a visit to a specialist, which was defined as the absence of an in-person visit to the type of specialist consulted electronically for 120 days. An e-consult was considered appropriate if it met the following four criteria.
- It could not be answered by referring to society guidelines or widely available, evidence-based summary sources.
- It did not seek logistic information, such as where to have a specific laboratory test done.
- It did not include a question of high urgency.
- The medical complexity of the clinical situation was not substantial enough to warrant an in-person consultation.
The investigators examined e-consult inquiries to mostly physician health care providers in five specialties – hematology, infectious disease, dermatology, rheumatology, and psychiatry – over a year.
High rates of appropriateness
The search spanned 6,512 eligible e-consults from 1,096 referring providers to 121 specialist consultants. Narrowing their search to 741 records with complete data, the investigators found that 70.2% of these consults met the criteria for appropriateness. In an analysis of four reviewers blinded to each other’s results, raters agreed on the appropriateness of 94% of e-consults.
Across specialties, more than 81% of e-consults were associated with avoided in-person visits.
The reasons for most e-consults were to seek answers to questions about diagnosis, therapeutics, or patient inquiries, or to request further education by PCPs.
“Across all specialties, the most common reasons an e-consult was not considered appropriate were failing the point-of-care resource test and asking a question of inappropriately high complexity,” the authors summarized.
Physicians and PCPs from tertiary care practices made up the majority of referring providers, with turnaround time for consults averaging 24 hours across specialties.
Rates of appropriateness, content, patient demographics, and timeliness of e-consult responses varied among the four specialties. Those with high avoidance of visits rates tended to have high appropriateness rates, indicating that some specialties may be more conducive to e-consults than others, the authors noted. Psychiatry and hematology had the highest proportion of appropriate e-consults (77.9% and 73.3% respectively). Rheumatology had the lowest proportion of appropriate e-consults and one of the lowest rates of avoided in-person visits, and dermatology had the lowest rate of avoided in-person visits, at 61.9%.
The majority (93%) of e-consults sought in psychiatry were therapy related, whereas 88.4% of the e-consult questions in rheumatology related to diagnosis.
“Questions about diagnosis were less likely to be answerable via e-consult, which suggests that to provide diagnoses, consultants may wish to engage with the patient directly,” Dr. Ahmed said in an interview.
Infectious disease specialists seemed to be the fastest responders, with nearly 90% of their consultations having been answered within a day. Dermatology specialists had the distinction of having the youngest e-consult patients (mean age, 38.6 years).
PCPs weigh in on results
Physicians said in interviews that the study data reflects their own positive experiences with e-consults.
“Although I don’t always think [an e-consult] is able to fully prevent the specialist visit, it does allow the specialist to provide recommendations for work-up that can be done prior to the specialist visit,” said Santina Wheat MD, a family physician at Erie Family Health Center in Chicago. This reduces the time in which the consult is placed to when effective treatment can take place.
Patients who may have to wait months or even years to see a specialty doctor, benefit from e-consults, said Dr. Wheat, who is also a member of the editorial advisory board of Family Practice News. “As part of an organization that does e-consults to another hospital with a different electronic medical record, the e-consult increases the likelihood that all of the clinical information reaches the specialists and prevents tests from being repeated.”
Starting an e-consult may also increase the likelihood that the patient quickly sees a specialist at the contracted hospital, she added.
Sarah G. Candler, MD, said in an interview that she also sees e-consults as an essential tool. “When patients present with rare, complex, or atypical pictures, I find it helpful to have specialists weigh in. The e-consult helps me ensure that I work to the top of my abilities as an internist,” said Dr. Candler, who is practice medical director and physician director of academic relations at Iora Primary Care, Northside Clinic, Houston. However, she did not agree with the study’s avoided in-person visits metric for assessing utility.
“In some cases, the end result of an e-consult is a referral for an in-person evaluation, and the role of the e-consult is to ensure that I have done my due diligence as a primary care doctor asking the correct questions, getting the appropriate work-up completed, and referring to the appropriate specialty for next steps, when necessary,” noted Dr. Candler, who also serves on the editorial advisory board of Internal Medicine News.
Financial considerations
The study’s authors suggested taking a closer look at standardizing payment for the use of e-consults and developing appropriateness criteria for them.
Health systems could use such criteria to study what makes an e-consult useful and how to best utilize this tool, Dr. Ahmed said in an interview.
“Compensation models that promote high-quality, effective, and efficient e-consults are needed to reinforce the ability of health systems to optimize the mix of e-consults and in-person visits,” Dr. Ahmed and colleagues suggested.
Because not all patient care requires e-consults, the model makes the most sense in practices that already participate in value-based payment programs. In these types of programs, the cost can be shared according to the variable risk and patient need for the service, Dr. Candler explained.
“I have been fortunate to work in two different systems that function in this way, which means that e-consults have been readily available and encouraged-both to improve patient care and decrease overall cost by decreasing unnecessary testing or specialist referral,” she said.
Dr. Wheat said that the managed care organization affiliated with her practice seems to be saving money with e-consults, as it decreases the need to pay for specialist visits in some instances and for repeated work-ups.
Future studies
The study’s cohort represented just one large health care system with a shared electronic health record. “Single-system descriptive studies, such as that of Ahmed and colleagues, are particularly useful for local evaluation and quality improvement efforts,” Varsha G. Vimalananda, MD, and B. Graeme Fincke, MD, both of the Center for Healthcare Organization and Implementation Research at Bedford (Mass.) Veterans Affairs Hospital, wrote in a related editorial.
“However, we need innovative approaches to evaluation that estimate the effect of e-consults on quality and cost of care across health care systems and over time. Implementation studies can help to identify key contributors to success,” the editorialists wrote.
One of the study authors, reported receiving personal fees from Bayer outside the submitted work. The other authors of the paper and the authors of the editorial reported no conflicts of interest. Dr. Candler said her employer contracts with an e-consult service, but that she is not compensated for use of the service. She is also a coeditor of Annals of Internal Medicine’s blog, “Fresh Look.”
SOURCE: Ahmed S et al. Ann Intern Med. 2020 Apr 14. doi: 10.7326/M19-3852.

Studies have shown that e-consults increase access to specialist care and primary care physician (PCP) education, according to research published in the Annals of Internal Medicine (2020. Apr 14. doi: 10.7326/M19-3852) by Salman Ahmed, MD, and colleagues.
These resources are already being frequently used by physicians, but more often by general internists and hospitalists than by subspecialists, according to a recent survey by the American College of Physicians. That survey found that 42% of its respondents are using e-consults and that subspecialists’ use is less common primarily because of the lack of access to e-consult technology.
What hasn’t been widely researched are the effects of large-scale e-consult programs, said Dr. Ahmed, who is associate physician in the renal division at Brigham and Women’s Hospital, Boston, in an interview.
For frontline providers such as PCPs, e-consults are a way to quickly seek out answers to clinical questions from specialists. In turn, the specialist can help a wider pool of participants, he noted.
The findings of Dr. Ahmed’s study, which included several academic centers and hospitals affiliated with Partners HealthCare System, a nonprofit network in eastern Massachusetts that includes Brigham and Women’s Hospital, used several metrics to analyze the appropriateness and utility of e-consults across a range of specialties. An e-consult was considered useful if it resulted in the avoidance of a visit to a specialist, which was defined as the absence of an in-person visit to the type of specialist consulted electronically for 120 days. An e-consult was considered appropriate if it met the following four criteria.
- It could not be answered by referring to society guidelines or widely available, evidence-based summary sources.
- It did not seek logistic information, such as where to have a specific laboratory test done.
- It did not include a question of high urgency.
- The medical complexity of the clinical situation was not substantial enough to warrant an in-person consultation.
The investigators examined e-consult inquiries to mostly physician health care providers in five specialties – hematology, infectious disease, dermatology, rheumatology, and psychiatry – over a year.
High rates of appropriateness
The search spanned 6,512 eligible e-consults from 1,096 referring providers to 121 specialist consultants. Narrowing their search to 741 records with complete data, the investigators found that 70.2% of these consults met the criteria for appropriateness. In an analysis of four reviewers blinded to each other’s results, raters agreed on the appropriateness of 94% of e-consults.
Across specialties, more than 81% of e-consults were associated with avoided in-person visits.
The reasons for most e-consults were to seek answers to questions about diagnosis, therapeutics, or patient inquiries, or to request further education by PCPs.
“Across all specialties, the most common reasons an e-consult was not considered appropriate were failing the point-of-care resource test and asking a question of inappropriately high complexity,” the authors summarized.
Physicians and PCPs from tertiary care practices made up the majority of referring providers, with turnaround time for consults averaging 24 hours across specialties.
Rates of appropriateness, content, patient demographics, and timeliness of e-consult responses varied among the four specialties. Those with high avoidance of visits rates tended to have high appropriateness rates, indicating that some specialties may be more conducive to e-consults than others, the authors noted. Psychiatry and hematology had the highest proportion of appropriate e-consults (77.9% and 73.3% respectively). Rheumatology had the lowest proportion of appropriate e-consults and one of the lowest rates of avoided in-person visits, and dermatology had the lowest rate of avoided in-person visits, at 61.9%.
The majority (93%) of e-consults sought in psychiatry were therapy related, whereas 88.4% of the e-consult questions in rheumatology related to diagnosis.
“Questions about diagnosis were less likely to be answerable via e-consult, which suggests that to provide diagnoses, consultants may wish to engage with the patient directly,” Dr. Ahmed said in an interview.
Infectious disease specialists seemed to be the fastest responders, with nearly 90% of their consultations having been answered within a day. Dermatology specialists had the distinction of having the youngest e-consult patients (mean age, 38.6 years).
PCPs weigh in on results
Physicians said in interviews that the study data reflects their own positive experiences with e-consults.
“Although I don’t always think [an e-consult] is able to fully prevent the specialist visit, it does allow the specialist to provide recommendations for work-up that can be done prior to the specialist visit,” said Santina Wheat MD, a family physician at Erie Family Health Center in Chicago. This reduces the time in which the consult is placed to when effective treatment can take place.
Patients who may have to wait months or even years to see a specialty doctor, benefit from e-consults, said Dr. Wheat, who is also a member of the editorial advisory board of Family Practice News. “As part of an organization that does e-consults to another hospital with a different electronic medical record, the e-consult increases the likelihood that all of the clinical information reaches the specialists and prevents tests from being repeated.”
Starting an e-consult may also increase the likelihood that the patient quickly sees a specialist at the contracted hospital, she added.
Sarah G. Candler, MD, said in an interview that she also sees e-consults as an essential tool. “When patients present with rare, complex, or atypical pictures, I find it helpful to have specialists weigh in. The e-consult helps me ensure that I work to the top of my abilities as an internist,” said Dr. Candler, who is practice medical director and physician director of academic relations at Iora Primary Care, Northside Clinic, Houston. However, she did not agree with the study’s avoided in-person visits metric for assessing utility.
“In some cases, the end result of an e-consult is a referral for an in-person evaluation, and the role of the e-consult is to ensure that I have done my due diligence as a primary care doctor asking the correct questions, getting the appropriate work-up completed, and referring to the appropriate specialty for next steps, when necessary,” noted Dr. Candler, who also serves on the editorial advisory board of Internal Medicine News.
Financial considerations
The study’s authors suggested taking a closer look at standardizing payment for the use of e-consults and developing appropriateness criteria for them.
Health systems could use such criteria to study what makes an e-consult useful and how to best utilize this tool, Dr. Ahmed said in an interview.
“Compensation models that promote high-quality, effective, and efficient e-consults are needed to reinforce the ability of health systems to optimize the mix of e-consults and in-person visits,” Dr. Ahmed and colleagues suggested.
Because not all patient care requires e-consults, the model makes the most sense in practices that already participate in value-based payment programs. In these types of programs, the cost can be shared according to the variable risk and patient need for the service, Dr. Candler explained.
“I have been fortunate to work in two different systems that function in this way, which means that e-consults have been readily available and encouraged-both to improve patient care and decrease overall cost by decreasing unnecessary testing or specialist referral,” she said.
Dr. Wheat said that the managed care organization affiliated with her practice seems to be saving money with e-consults, as it decreases the need to pay for specialist visits in some instances and for repeated work-ups.
Future studies
The study’s cohort represented just one large health care system with a shared electronic health record. “Single-system descriptive studies, such as that of Ahmed and colleagues, are particularly useful for local evaluation and quality improvement efforts,” Varsha G. Vimalananda, MD, and B. Graeme Fincke, MD, both of the Center for Healthcare Organization and Implementation Research at Bedford (Mass.) Veterans Affairs Hospital, wrote in a related editorial.
“However, we need innovative approaches to evaluation that estimate the effect of e-consults on quality and cost of care across health care systems and over time. Implementation studies can help to identify key contributors to success,” the editorialists wrote.
One of the study authors, reported receiving personal fees from Bayer outside the submitted work. The other authors of the paper and the authors of the editorial reported no conflicts of interest. Dr. Candler said her employer contracts with an e-consult service, but that she is not compensated for use of the service. She is also a coeditor of Annals of Internal Medicine’s blog, “Fresh Look.”
SOURCE: Ahmed S et al. Ann Intern Med. 2020 Apr 14. doi: 10.7326/M19-3852.
FROM ANNALS OF INTERNAL MEDICINE
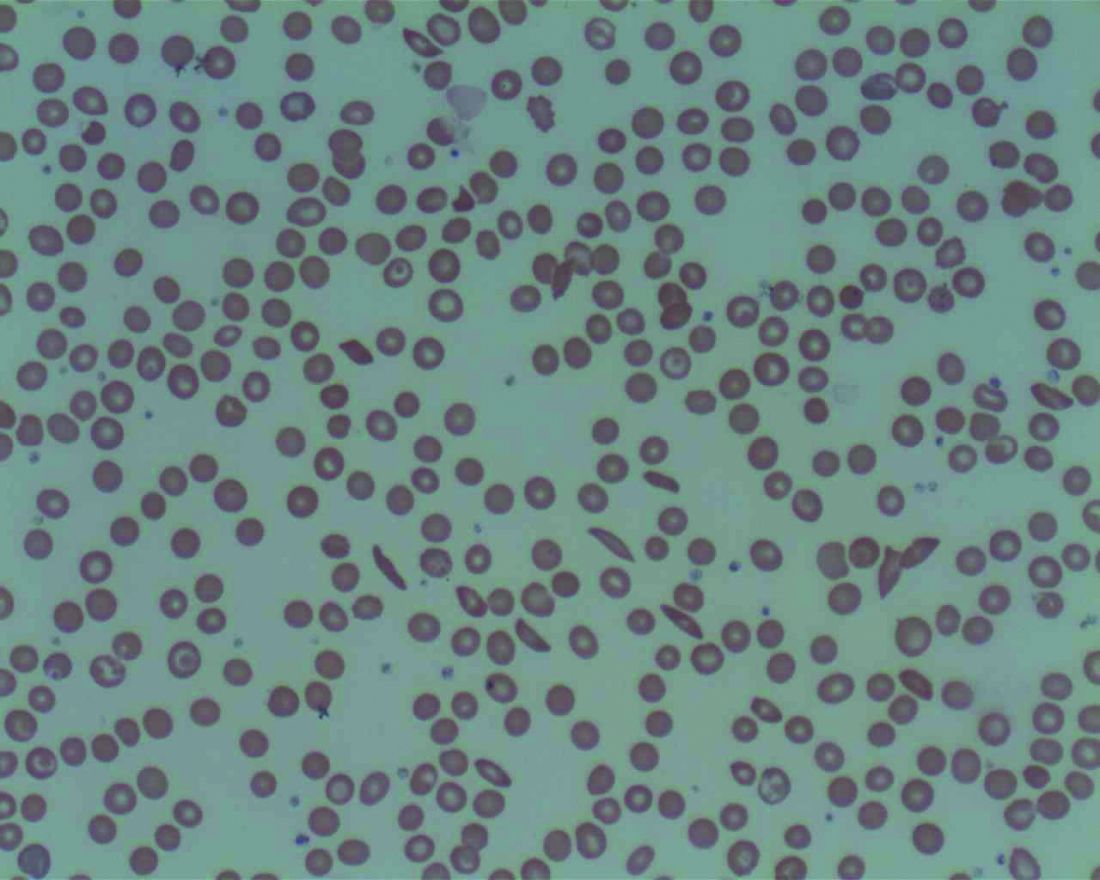
Low plasma sodium levels can predict complications in acute painful episodes of SCD
Although a low blood sodium level has been shown to be a prognostic factor for a number of disorders, it has not been reported on for sickle cell disease, according to French researchers Jean-Simon Rech, MD, and colleagues.
They found that hyponatremia at hospital admission was predictive of complications in initially uncomplicated episodes of painful episodes of sickle cell disease (SCD), according to their study published online in the American Journal of Medicine. Dr. Rech is with the department of internal medicine, Sickle Cell Disease Reference Center, Tenon Hospital, Assistance Publique-Hôpitaux de Paris.
The study assessed 1,218 stays (406 patients) admitted to a single center and the analyses were adjusted for age, sex, hemoglobin genotype and concentration, LDH concentration, and white blood cell count.
The researchers found that hyponatremia (defined as plasma sodium ≤ 135 mmol/L) was significantly associated with the primary endpoint of a composite criterion including acute chest syndrome, intensive care unit transfer, red blood cell transfusion, or inpatient death (P = .001).
With regard to the components of the primary endpoint, hyponatremia was significantly associated with acute chest syndrome (P = .008), as well as with receiving a red blood cell transfusion (P < .001) However, hyponatremia at admission was not significantly associated with intensive care unit transfer (P = .074) and there were no patient deaths.
In addition, hyponatremia was significantly associated with longer stays: 1.1 days adjusted mean length of stay (P < .001).
“Hyponatremia may lead to direct clinical consequences in the management of sickle cell disease patients. Indeed, a plasma sodium concentration ≤ 135mmol/L at admission or a decreasing natremia over the first days of an acute painful episode could be regarded as early signs of incipient acute chest syndrome, prompting clinicians to closely monitor the clinical status of their patients,” the researchers concluded.
The authors declared that they had no conflicts and that there was no funding source.
SOURCE: Rech JS et al. Am J Med. 2020 Mar 18. doi.org/10.1016/j.amjmed.2020.02.017.
Although a low blood sodium level has been shown to be a prognostic factor for a number of disorders, it has not been reported on for sickle cell disease, according to French researchers Jean-Simon Rech, MD, and colleagues.
They found that hyponatremia at hospital admission was predictive of complications in initially uncomplicated episodes of painful episodes of sickle cell disease (SCD), according to their study published online in the American Journal of Medicine. Dr. Rech is with the department of internal medicine, Sickle Cell Disease Reference Center, Tenon Hospital, Assistance Publique-Hôpitaux de Paris.
The study assessed 1,218 stays (406 patients) admitted to a single center and the analyses were adjusted for age, sex, hemoglobin genotype and concentration, LDH concentration, and white blood cell count.
The researchers found that hyponatremia (defined as plasma sodium ≤ 135 mmol/L) was significantly associated with the primary endpoint of a composite criterion including acute chest syndrome, intensive care unit transfer, red blood cell transfusion, or inpatient death (P = .001).
With regard to the components of the primary endpoint, hyponatremia was significantly associated with acute chest syndrome (P = .008), as well as with receiving a red blood cell transfusion (P < .001) However, hyponatremia at admission was not significantly associated with intensive care unit transfer (P = .074) and there were no patient deaths.
In addition, hyponatremia was significantly associated with longer stays: 1.1 days adjusted mean length of stay (P < .001).
“Hyponatremia may lead to direct clinical consequences in the management of sickle cell disease patients. Indeed, a plasma sodium concentration ≤ 135mmol/L at admission or a decreasing natremia over the first days of an acute painful episode could be regarded as early signs of incipient acute chest syndrome, prompting clinicians to closely monitor the clinical status of their patients,” the researchers concluded.
The authors declared that they had no conflicts and that there was no funding source.
SOURCE: Rech JS et al. Am J Med. 2020 Mar 18. doi.org/10.1016/j.amjmed.2020.02.017.
Although a low blood sodium level has been shown to be a prognostic factor for a number of disorders, it has not been reported on for sickle cell disease, according to French researchers Jean-Simon Rech, MD, and colleagues.
They found that hyponatremia at hospital admission was predictive of complications in initially uncomplicated episodes of painful episodes of sickle cell disease (SCD), according to their study published online in the American Journal of Medicine. Dr. Rech is with the department of internal medicine, Sickle Cell Disease Reference Center, Tenon Hospital, Assistance Publique-Hôpitaux de Paris.
The study assessed 1,218 stays (406 patients) admitted to a single center and the analyses were adjusted for age, sex, hemoglobin genotype and concentration, LDH concentration, and white blood cell count.
The researchers found that hyponatremia (defined as plasma sodium ≤ 135 mmol/L) was significantly associated with the primary endpoint of a composite criterion including acute chest syndrome, intensive care unit transfer, red blood cell transfusion, or inpatient death (P = .001).
With regard to the components of the primary endpoint, hyponatremia was significantly associated with acute chest syndrome (P = .008), as well as with receiving a red blood cell transfusion (P < .001) However, hyponatremia at admission was not significantly associated with intensive care unit transfer (P = .074) and there were no patient deaths.
In addition, hyponatremia was significantly associated with longer stays: 1.1 days adjusted mean length of stay (P < .001).
“Hyponatremia may lead to direct clinical consequences in the management of sickle cell disease patients. Indeed, a plasma sodium concentration ≤ 135mmol/L at admission or a decreasing natremia over the first days of an acute painful episode could be regarded as early signs of incipient acute chest syndrome, prompting clinicians to closely monitor the clinical status of their patients,” the researchers concluded.
The authors declared that they had no conflicts and that there was no funding source.
SOURCE: Rech JS et al. Am J Med. 2020 Mar 18. doi.org/10.1016/j.amjmed.2020.02.017.
FROM THE AMERICAN JOURNAL OF MEDICINE
Cardiovascular problems already apparent in children with hemophilia A
Negative cardiovascular health indicators were found to be higher in children with hemophilia A, compared with healthy children, according to a small research study.
Biochemical, imaging, and metabolic analyses were performed to compare 17 boys with severe hemophilia A to 23 healthy boys designated as controls.
The myocardial performance index (MPI) was evaluated using tissue Doppler echocardiography. In addition, peripheral and central blood pressure and arterial stiffness were assessed, as were carotid intima–media thicknesses (CIMTs), serum glucose, insulin, insulin resistance, and lipoprotein levels.
Increased MPI is considered an indicator of global deterioration in myocardial functions.
There were no differences between the two groups in terms of age and biochemical parameters, according to the researchers. There were also no significant differences found between the groups in terms of CIMT, peripheral blood pressure, and central systolic blood pressure.
However, the HDL cholesterol levels in the hemophilia group were significantly lower than those in the control group (P < .05). Five of the hemophilia patients had insulin resistance (29.4%), whereas four had low HDL cholesterol levels (23.5%).
The researchers found that the MPI values in the hemophilia group were higher than those in the control group (0.41 vs. 0.34; P = .004). In addition, left ventricle ejection time (ET), which is a predictor of mortality in heart failure and ischemic heart disease, was shorter in the hemophilia group than it was in the control group (266.6 vs. 284.4; P = .014).
As arterial stiffness increases, ejection time decreases owing to the deteriorating myocardial systolic functions, and it has also been reported in the literature that arterial stiffness affects left ventricular systolic functions, according to the authors.
“Arterial stiffness and high [central diastolic blood pressure] developing in patients with severe hemophilia A since their childhood are important risk factors for coronary artery diseases. Predisposition to dyslipidemia and [insulin resistance] noted in the hemophilia group also negatively contributes to this process. Adolescent patients with hemophilia should be monitored for hypertension, obesity, dyslipidemia, and [insulin resistance],” the authors concluded.
The study received no external funding. The authors did not report disclosures.
SOURCE: Özdemir ZC et al. Thrombosis Res. 2020;189:102-7.
Negative cardiovascular health indicators were found to be higher in children with hemophilia A, compared with healthy children, according to a small research study.
Biochemical, imaging, and metabolic analyses were performed to compare 17 boys with severe hemophilia A to 23 healthy boys designated as controls.
The myocardial performance index (MPI) was evaluated using tissue Doppler echocardiography. In addition, peripheral and central blood pressure and arterial stiffness were assessed, as were carotid intima–media thicknesses (CIMTs), serum glucose, insulin, insulin resistance, and lipoprotein levels.
Increased MPI is considered an indicator of global deterioration in myocardial functions.
There were no differences between the two groups in terms of age and biochemical parameters, according to the researchers. There were also no significant differences found between the groups in terms of CIMT, peripheral blood pressure, and central systolic blood pressure.
However, the HDL cholesterol levels in the hemophilia group were significantly lower than those in the control group (P < .05). Five of the hemophilia patients had insulin resistance (29.4%), whereas four had low HDL cholesterol levels (23.5%).
The researchers found that the MPI values in the hemophilia group were higher than those in the control group (0.41 vs. 0.34; P = .004). In addition, left ventricle ejection time (ET), which is a predictor of mortality in heart failure and ischemic heart disease, was shorter in the hemophilia group than it was in the control group (266.6 vs. 284.4; P = .014).
As arterial stiffness increases, ejection time decreases owing to the deteriorating myocardial systolic functions, and it has also been reported in the literature that arterial stiffness affects left ventricular systolic functions, according to the authors.
“Arterial stiffness and high [central diastolic blood pressure] developing in patients with severe hemophilia A since their childhood are important risk factors for coronary artery diseases. Predisposition to dyslipidemia and [insulin resistance] noted in the hemophilia group also negatively contributes to this process. Adolescent patients with hemophilia should be monitored for hypertension, obesity, dyslipidemia, and [insulin resistance],” the authors concluded.
The study received no external funding. The authors did not report disclosures.
SOURCE: Özdemir ZC et al. Thrombosis Res. 2020;189:102-7.
Negative cardiovascular health indicators were found to be higher in children with hemophilia A, compared with healthy children, according to a small research study.
Biochemical, imaging, and metabolic analyses were performed to compare 17 boys with severe hemophilia A to 23 healthy boys designated as controls.
The myocardial performance index (MPI) was evaluated using tissue Doppler echocardiography. In addition, peripheral and central blood pressure and arterial stiffness were assessed, as were carotid intima–media thicknesses (CIMTs), serum glucose, insulin, insulin resistance, and lipoprotein levels.
Increased MPI is considered an indicator of global deterioration in myocardial functions.
There were no differences between the two groups in terms of age and biochemical parameters, according to the researchers. There were also no significant differences found between the groups in terms of CIMT, peripheral blood pressure, and central systolic blood pressure.
However, the HDL cholesterol levels in the hemophilia group were significantly lower than those in the control group (P < .05). Five of the hemophilia patients had insulin resistance (29.4%), whereas four had low HDL cholesterol levels (23.5%).
The researchers found that the MPI values in the hemophilia group were higher than those in the control group (0.41 vs. 0.34; P = .004). In addition, left ventricle ejection time (ET), which is a predictor of mortality in heart failure and ischemic heart disease, was shorter in the hemophilia group than it was in the control group (266.6 vs. 284.4; P = .014).
As arterial stiffness increases, ejection time decreases owing to the deteriorating myocardial systolic functions, and it has also been reported in the literature that arterial stiffness affects left ventricular systolic functions, according to the authors.
“Arterial stiffness and high [central diastolic blood pressure] developing in patients with severe hemophilia A since their childhood are important risk factors for coronary artery diseases. Predisposition to dyslipidemia and [insulin resistance] noted in the hemophilia group also negatively contributes to this process. Adolescent patients with hemophilia should be monitored for hypertension, obesity, dyslipidemia, and [insulin resistance],” the authors concluded.
The study received no external funding. The authors did not report disclosures.
SOURCE: Özdemir ZC et al. Thrombosis Res. 2020;189:102-7.
FROM THROMBOSIS RESEARCH
First advance in MDS for decade: Luspatercept for anemia
The US Food and Drug Administration has approved luspatercept (Reblozyl, Bristol-Myers Squibb/Acceleron) for the treatment of anemia in patients with myelodysplastic syndromes (MDS).
The green light represents the first treatment advancement in MDS in more than a decade, says an expert in the field.
Luspatercept is the first and so far only erythroid maturation agent (EMA), and was launched last year when it was approved for the treatment of anemia in adults with beta thalassemia, who require regular red blood cell transfusions.
The new approval is for the treatment of anemia in adult patients with very low- to intermediate-risk MDS with ring sideroblasts and patients with myelodysplastic/myeloproliferative neoplasm with ring sideroblasts and thrombocytosis, after they have progressed on treatment with an erythropoiesis-stimulating agent and who require two or more red blood cell (RBC) units over 8 weeks.
Luspatercept is not a substitute for RBC transfusions in patients who require immediate correction of anemia.
The FDA approval in MDS is based on results from the pivotal, placebo-controlled, phase 3 MEDALIST trial, conducted in 229 patients with very-low–, low- and intermediate-risk non-del(5q) MDS with ring sideroblasts. All patients were RBC transfusion-dependent and had disease that was refractory to, or unlikely to respond to, erythropoiesis-stimulating agents. Results were published in January in the New England Journal of Medicine. The study was funded by Acceleron Pharma and Celgene, which was later acquired by Bristol-Myers Squibb.
These results were first presented at the 2018 annual meeting of the American Society of Hematology (ASH), as reported by Medscape Medical News. At the time, ASH President Alexis Thompson, MD, said it appears that luspatercept can improve the production of endogenous RBCs by enhancing the maturation of these cells in the bone marrow. The drug significantly reduced the need for RBC transfusions, and “this is a very exciting advance for patients who would have few other treatment options,” she said.
“Anemia and the chronic need for transfusions is a very big issue for these patients,” commented lead study author Pierre Fenaux, MD, PhD, from Hôpital Saint-Louis in Paris, France. “With low hemoglobin levels, patients are tired all the time and have an increased risk of falls and cardiovascular events. When you can improve hemoglobin levels, you really see a difference in quality of life.”
The MEDALIST trial is an important milestone for patients with lower-risk, transfusion-dependent MDS, commented Elizabeth Griffiths, MD, associate professor of oncology and director of MDS, Roswell Park Comprehensive Cancer Center, Buffalo, New York.
“No new agents have been approved for MDS in the last 10 years, highlighting this development as a substantial step forward for the MDS community,” she told Medscape Medical News. “Current therapies are time-intensive and only modestly beneficial.”
“The availability of a new, effective drug — particularly relevant to those harboring SF3B1 mutations — is an exciting development and is likely to offer meaningful improvements in quality of life,” Griffiths said. “Since these patients tend to live longer than others with MDS, there are many patients in my clinical practice who would have fit the enrollment criteria for this study. Such patients are eagerly awaiting the opportunity for a decrease in transfusion burden.”
Study Details
In the trial, luspatercept reduced the severity of anemia — 38% of the 153 patients who received luspatercept achieved transfusion independence for 8 weeks or longer compared with 13% of the 76 patients receiving placebo (P < .001).
In the study, patients received luspatercept at a starting dose of 1.0 mg/kg with titration up to 1.75 mg/kg, if needed, or placebo, subcutaneously every 3 weeks for at least 24 weeks.
During the 16 weeks before the initiation of treatment, study patients had received a median of 5 RBC units transfusions during an 8-week period (43.2% of patients had ≥ 6 RBC units, 27.9% had ≥ 4 to < 6 RBC units, and 28.8% had < 4 RBC units). At baseline, 138 (60.3%), 58 (25.3%), and 32 (14%) patients had serum erythropoietin levels less than 200 IU/L, 200-500 IU/L, and greater than 500 IU/L, respectively.
The most common luspatercept-associated adverse events (any grade) in the trial were fatigue, diarrhea, asthenia, nausea, and dizziness. Grade 3 or 4 treatment-emergent adverse events were reported in 42.5% of patients who received luspatercept and 44.7% of patients who received placebo. The incidence of adverse events decreased over time, according to the study authors.
This article first appeared on Medscape.com.
The US Food and Drug Administration has approved luspatercept (Reblozyl, Bristol-Myers Squibb/Acceleron) for the treatment of anemia in patients with myelodysplastic syndromes (MDS).
The green light represents the first treatment advancement in MDS in more than a decade, says an expert in the field.
Luspatercept is the first and so far only erythroid maturation agent (EMA), and was launched last year when it was approved for the treatment of anemia in adults with beta thalassemia, who require regular red blood cell transfusions.
The new approval is for the treatment of anemia in adult patients with very low- to intermediate-risk MDS with ring sideroblasts and patients with myelodysplastic/myeloproliferative neoplasm with ring sideroblasts and thrombocytosis, after they have progressed on treatment with an erythropoiesis-stimulating agent and who require two or more red blood cell (RBC) units over 8 weeks.
Luspatercept is not a substitute for RBC transfusions in patients who require immediate correction of anemia.
The FDA approval in MDS is based on results from the pivotal, placebo-controlled, phase 3 MEDALIST trial, conducted in 229 patients with very-low–, low- and intermediate-risk non-del(5q) MDS with ring sideroblasts. All patients were RBC transfusion-dependent and had disease that was refractory to, or unlikely to respond to, erythropoiesis-stimulating agents. Results were published in January in the New England Journal of Medicine. The study was funded by Acceleron Pharma and Celgene, which was later acquired by Bristol-Myers Squibb.
These results were first presented at the 2018 annual meeting of the American Society of Hematology (ASH), as reported by Medscape Medical News. At the time, ASH President Alexis Thompson, MD, said it appears that luspatercept can improve the production of endogenous RBCs by enhancing the maturation of these cells in the bone marrow. The drug significantly reduced the need for RBC transfusions, and “this is a very exciting advance for patients who would have few other treatment options,” she said.
“Anemia and the chronic need for transfusions is a very big issue for these patients,” commented lead study author Pierre Fenaux, MD, PhD, from Hôpital Saint-Louis in Paris, France. “With low hemoglobin levels, patients are tired all the time and have an increased risk of falls and cardiovascular events. When you can improve hemoglobin levels, you really see a difference in quality of life.”
The MEDALIST trial is an important milestone for patients with lower-risk, transfusion-dependent MDS, commented Elizabeth Griffiths, MD, associate professor of oncology and director of MDS, Roswell Park Comprehensive Cancer Center, Buffalo, New York.
“No new agents have been approved for MDS in the last 10 years, highlighting this development as a substantial step forward for the MDS community,” she told Medscape Medical News. “Current therapies are time-intensive and only modestly beneficial.”
“The availability of a new, effective drug — particularly relevant to those harboring SF3B1 mutations — is an exciting development and is likely to offer meaningful improvements in quality of life,” Griffiths said. “Since these patients tend to live longer than others with MDS, there are many patients in my clinical practice who would have fit the enrollment criteria for this study. Such patients are eagerly awaiting the opportunity for a decrease in transfusion burden.”
Study Details
In the trial, luspatercept reduced the severity of anemia — 38% of the 153 patients who received luspatercept achieved transfusion independence for 8 weeks or longer compared with 13% of the 76 patients receiving placebo (P < .001).
In the study, patients received luspatercept at a starting dose of 1.0 mg/kg with titration up to 1.75 mg/kg, if needed, or placebo, subcutaneously every 3 weeks for at least 24 weeks.
During the 16 weeks before the initiation of treatment, study patients had received a median of 5 RBC units transfusions during an 8-week period (43.2% of patients had ≥ 6 RBC units, 27.9% had ≥ 4 to < 6 RBC units, and 28.8% had < 4 RBC units). At baseline, 138 (60.3%), 58 (25.3%), and 32 (14%) patients had serum erythropoietin levels less than 200 IU/L, 200-500 IU/L, and greater than 500 IU/L, respectively.
The most common luspatercept-associated adverse events (any grade) in the trial were fatigue, diarrhea, asthenia, nausea, and dizziness. Grade 3 or 4 treatment-emergent adverse events were reported in 42.5% of patients who received luspatercept and 44.7% of patients who received placebo. The incidence of adverse events decreased over time, according to the study authors.
This article first appeared on Medscape.com.
The US Food and Drug Administration has approved luspatercept (Reblozyl, Bristol-Myers Squibb/Acceleron) for the treatment of anemia in patients with myelodysplastic syndromes (MDS).
The green light represents the first treatment advancement in MDS in more than a decade, says an expert in the field.
Luspatercept is the first and so far only erythroid maturation agent (EMA), and was launched last year when it was approved for the treatment of anemia in adults with beta thalassemia, who require regular red blood cell transfusions.
The new approval is for the treatment of anemia in adult patients with very low- to intermediate-risk MDS with ring sideroblasts and patients with myelodysplastic/myeloproliferative neoplasm with ring sideroblasts and thrombocytosis, after they have progressed on treatment with an erythropoiesis-stimulating agent and who require two or more red blood cell (RBC) units over 8 weeks.
Luspatercept is not a substitute for RBC transfusions in patients who require immediate correction of anemia.
The FDA approval in MDS is based on results from the pivotal, placebo-controlled, phase 3 MEDALIST trial, conducted in 229 patients with very-low–, low- and intermediate-risk non-del(5q) MDS with ring sideroblasts. All patients were RBC transfusion-dependent and had disease that was refractory to, or unlikely to respond to, erythropoiesis-stimulating agents. Results were published in January in the New England Journal of Medicine. The study was funded by Acceleron Pharma and Celgene, which was later acquired by Bristol-Myers Squibb.
These results were first presented at the 2018 annual meeting of the American Society of Hematology (ASH), as reported by Medscape Medical News. At the time, ASH President Alexis Thompson, MD, said it appears that luspatercept can improve the production of endogenous RBCs by enhancing the maturation of these cells in the bone marrow. The drug significantly reduced the need for RBC transfusions, and “this is a very exciting advance for patients who would have few other treatment options,” she said.
“Anemia and the chronic need for transfusions is a very big issue for these patients,” commented lead study author Pierre Fenaux, MD, PhD, from Hôpital Saint-Louis in Paris, France. “With low hemoglobin levels, patients are tired all the time and have an increased risk of falls and cardiovascular events. When you can improve hemoglobin levels, you really see a difference in quality of life.”
The MEDALIST trial is an important milestone for patients with lower-risk, transfusion-dependent MDS, commented Elizabeth Griffiths, MD, associate professor of oncology and director of MDS, Roswell Park Comprehensive Cancer Center, Buffalo, New York.
“No new agents have been approved for MDS in the last 10 years, highlighting this development as a substantial step forward for the MDS community,” she told Medscape Medical News. “Current therapies are time-intensive and only modestly beneficial.”
“The availability of a new, effective drug — particularly relevant to those harboring SF3B1 mutations — is an exciting development and is likely to offer meaningful improvements in quality of life,” Griffiths said. “Since these patients tend to live longer than others with MDS, there are many patients in my clinical practice who would have fit the enrollment criteria for this study. Such patients are eagerly awaiting the opportunity for a decrease in transfusion burden.”
Study Details
In the trial, luspatercept reduced the severity of anemia — 38% of the 153 patients who received luspatercept achieved transfusion independence for 8 weeks or longer compared with 13% of the 76 patients receiving placebo (P < .001).
In the study, patients received luspatercept at a starting dose of 1.0 mg/kg with titration up to 1.75 mg/kg, if needed, or placebo, subcutaneously every 3 weeks for at least 24 weeks.
During the 16 weeks before the initiation of treatment, study patients had received a median of 5 RBC units transfusions during an 8-week period (43.2% of patients had ≥ 6 RBC units, 27.9% had ≥ 4 to < 6 RBC units, and 28.8% had < 4 RBC units). At baseline, 138 (60.3%), 58 (25.3%), and 32 (14%) patients had serum erythropoietin levels less than 200 IU/L, 200-500 IU/L, and greater than 500 IU/L, respectively.
The most common luspatercept-associated adverse events (any grade) in the trial were fatigue, diarrhea, asthenia, nausea, and dizziness. Grade 3 or 4 treatment-emergent adverse events were reported in 42.5% of patients who received luspatercept and 44.7% of patients who received placebo. The incidence of adverse events decreased over time, according to the study authors.
This article first appeared on Medscape.com.
FDA approves recombinant treatment for hemophilia A or B with inhibitors
The Food and Drug Administration has approved a new rabbit-derived recombinant analog of human coagulation factor VII (Sevenfact) for the treatment of hemophilia A or B, according to a release from the agency.
The treatment’s approval is for use in patients aged 12 years and older who’ve developed inhibitors (neutralizing antibodies). This factor VII (FVII) analog bypasses the FVIII and FIX reactions, which are no longer effective pathways for treatment in these patients because of the inhibitors they’ve developed.
According to the release, “the active ingredient of Sevenfact is a recombinant analog of human FVII, which is expressed in the mammary gland of genetically engineered rabbits and secreted into the rabbits’ milk. During purification and processing of the milk, FVII is converted into activated FVII.” The FDA’s Center for Veterinary Medicine has, after “comprehensive analysis of the scientific evidence,” determined that the process is safe for both the rabbits and handlers and that it is an effective means of producing the coagulation factor.
The approval is based on a clinical study that evaluated 27 patients with hemophilia A or B and included treatment for 465 mild to moderate events and 3 severe ones. The low (75 mcg/kg) or high (225 mcg/kg) dose was able to successfully treat 86% of the mild to moderate episodes, and all three of the severe ones were successfully treated with the high dose.
The most common side effects were headache, dizziness, infusion-site discomfort, infusion-related reaction, infusion-site hematoma, and fever. This product is contraindicated in patients with known allergies or hypersensitivities to rabbits or rabbit proteins. There is potential for increased risk of serious arterial or venous thrombotic events among patients with other risk factors for blood clots. More safety information can be found in the prescribing information, and more information about the approval can be found in the full release.
The Food and Drug Administration has approved a new rabbit-derived recombinant analog of human coagulation factor VII (Sevenfact) for the treatment of hemophilia A or B, according to a release from the agency.
The treatment’s approval is for use in patients aged 12 years and older who’ve developed inhibitors (neutralizing antibodies). This factor VII (FVII) analog bypasses the FVIII and FIX reactions, which are no longer effective pathways for treatment in these patients because of the inhibitors they’ve developed.
According to the release, “the active ingredient of Sevenfact is a recombinant analog of human FVII, which is expressed in the mammary gland of genetically engineered rabbits and secreted into the rabbits’ milk. During purification and processing of the milk, FVII is converted into activated FVII.” The FDA’s Center for Veterinary Medicine has, after “comprehensive analysis of the scientific evidence,” determined that the process is safe for both the rabbits and handlers and that it is an effective means of producing the coagulation factor.
The approval is based on a clinical study that evaluated 27 patients with hemophilia A or B and included treatment for 465 mild to moderate events and 3 severe ones. The low (75 mcg/kg) or high (225 mcg/kg) dose was able to successfully treat 86% of the mild to moderate episodes, and all three of the severe ones were successfully treated with the high dose.
The most common side effects were headache, dizziness, infusion-site discomfort, infusion-related reaction, infusion-site hematoma, and fever. This product is contraindicated in patients with known allergies or hypersensitivities to rabbits or rabbit proteins. There is potential for increased risk of serious arterial or venous thrombotic events among patients with other risk factors for blood clots. More safety information can be found in the prescribing information, and more information about the approval can be found in the full release.
The Food and Drug Administration has approved a new rabbit-derived recombinant analog of human coagulation factor VII (Sevenfact) for the treatment of hemophilia A or B, according to a release from the agency.
The treatment’s approval is for use in patients aged 12 years and older who’ve developed inhibitors (neutralizing antibodies). This factor VII (FVII) analog bypasses the FVIII and FIX reactions, which are no longer effective pathways for treatment in these patients because of the inhibitors they’ve developed.
According to the release, “the active ingredient of Sevenfact is a recombinant analog of human FVII, which is expressed in the mammary gland of genetically engineered rabbits and secreted into the rabbits’ milk. During purification and processing of the milk, FVII is converted into activated FVII.” The FDA’s Center for Veterinary Medicine has, after “comprehensive analysis of the scientific evidence,” determined that the process is safe for both the rabbits and handlers and that it is an effective means of producing the coagulation factor.
The approval is based on a clinical study that evaluated 27 patients with hemophilia A or B and included treatment for 465 mild to moderate events and 3 severe ones. The low (75 mcg/kg) or high (225 mcg/kg) dose was able to successfully treat 86% of the mild to moderate episodes, and all three of the severe ones were successfully treated with the high dose.
The most common side effects were headache, dizziness, infusion-site discomfort, infusion-related reaction, infusion-site hematoma, and fever. This product is contraindicated in patients with known allergies or hypersensitivities to rabbits or rabbit proteins. There is potential for increased risk of serious arterial or venous thrombotic events among patients with other risk factors for blood clots. More safety information can be found in the prescribing information, and more information about the approval can be found in the full release.
COVID-19: Adjusting practice in acute leukemia care
The SARS-CoV-2 pandemic poses significant risks to leukemia patients and their providers, impacting every aspect of care from diagnosis through therapy, according to an editorial letter published online in Leukemia Research.
One key concern to be considered is the risk of missed or delayed diagnosis due to the pandemic conditions. An estimated 50%-75% of patients with acute leukemia are febrile at diagnosis and this puts them at high risk of a misdiagnosis of COVID-19 upon initial evaluation. As with other oncological conditions (primary mediastinal lymphoma or lung cancer, for example), which often present with a cough with or without fever, their symptoms “are likely to be considered trivial after a negative SARS-CoV-2 test,” with patients then being sent home without further assessment. In a rapidly progressing disease such as acute leukemia, this could lead to critical delays in therapeutic intervention.
The authors, from the Service and Central Laboratory of Hematology, Lausanne (Switzerland) University Hospital, also discussed the problems that might occur with regard to most standard forms of therapy. In particular, they addressed potential impacts of the pandemic on chemotherapy, bone marrow transplantation, maintenance treatments, supportive measures, and targeted therapies.
Of particular concern, “most patients may suffer from postponed chemotherapy, due to a shortage of isolation beds and blood products or the wish to avoid immunosuppressive treatments,” the authors noted, warning that “delay in chemotherapy initiation may negatively affect prognosis, [particularly in patients under age 60] with favorable- or intermediate-risk disease.”
With regard to stem cell transplantation, the authors detail the many potential difficulties with regard to procedures involving both donors and recipients, and warn that in some cases, delay in transplant could result in the reappearance of a significant minimal residual disease, which has a well-established negative impact on survival.
The authors also noted that blood product shortages have already begun in most affected countries, and how, in response, transfusion societies have called for conservative transfusion policies in strict adherence to evidence-based guidelines for patient’s blood management.
“COVID-19 will result in numerous casualties. Acute leukemia patients are at a higher risk of severe complications,” the authors stated. In particular, physicians should especially be aware of how treatment for acute leukemia may have “interactions with other drugs used to treat SARS-CoV-2–related infections/complications such as antibiotics, antiviral drugs, and various other drugs that prolong QTc or impact targeted-therapy pharmacokinetics,” they concluded.
The authors reported that they received no government or private funding for this research, and that they had no conflicts of interest.
SOURCE: Gavillet M et al. Leuk. Res. 2020. doi.org/10.1016/j.leukres.2020.106353.
The SARS-CoV-2 pandemic poses significant risks to leukemia patients and their providers, impacting every aspect of care from diagnosis through therapy, according to an editorial letter published online in Leukemia Research.
One key concern to be considered is the risk of missed or delayed diagnosis due to the pandemic conditions. An estimated 50%-75% of patients with acute leukemia are febrile at diagnosis and this puts them at high risk of a misdiagnosis of COVID-19 upon initial evaluation. As with other oncological conditions (primary mediastinal lymphoma or lung cancer, for example), which often present with a cough with or without fever, their symptoms “are likely to be considered trivial after a negative SARS-CoV-2 test,” with patients then being sent home without further assessment. In a rapidly progressing disease such as acute leukemia, this could lead to critical delays in therapeutic intervention.
The authors, from the Service and Central Laboratory of Hematology, Lausanne (Switzerland) University Hospital, also discussed the problems that might occur with regard to most standard forms of therapy. In particular, they addressed potential impacts of the pandemic on chemotherapy, bone marrow transplantation, maintenance treatments, supportive measures, and targeted therapies.
Of particular concern, “most patients may suffer from postponed chemotherapy, due to a shortage of isolation beds and blood products or the wish to avoid immunosuppressive treatments,” the authors noted, warning that “delay in chemotherapy initiation may negatively affect prognosis, [particularly in patients under age 60] with favorable- or intermediate-risk disease.”
With regard to stem cell transplantation, the authors detail the many potential difficulties with regard to procedures involving both donors and recipients, and warn that in some cases, delay in transplant could result in the reappearance of a significant minimal residual disease, which has a well-established negative impact on survival.
The authors also noted that blood product shortages have already begun in most affected countries, and how, in response, transfusion societies have called for conservative transfusion policies in strict adherence to evidence-based guidelines for patient’s blood management.
“COVID-19 will result in numerous casualties. Acute leukemia patients are at a higher risk of severe complications,” the authors stated. In particular, physicians should especially be aware of how treatment for acute leukemia may have “interactions with other drugs used to treat SARS-CoV-2–related infections/complications such as antibiotics, antiviral drugs, and various other drugs that prolong QTc or impact targeted-therapy pharmacokinetics,” they concluded.
The authors reported that they received no government or private funding for this research, and that they had no conflicts of interest.
SOURCE: Gavillet M et al. Leuk. Res. 2020. doi.org/10.1016/j.leukres.2020.106353.
The SARS-CoV-2 pandemic poses significant risks to leukemia patients and their providers, impacting every aspect of care from diagnosis through therapy, according to an editorial letter published online in Leukemia Research.
One key concern to be considered is the risk of missed or delayed diagnosis due to the pandemic conditions. An estimated 50%-75% of patients with acute leukemia are febrile at diagnosis and this puts them at high risk of a misdiagnosis of COVID-19 upon initial evaluation. As with other oncological conditions (primary mediastinal lymphoma or lung cancer, for example), which often present with a cough with or without fever, their symptoms “are likely to be considered trivial after a negative SARS-CoV-2 test,” with patients then being sent home without further assessment. In a rapidly progressing disease such as acute leukemia, this could lead to critical delays in therapeutic intervention.
The authors, from the Service and Central Laboratory of Hematology, Lausanne (Switzerland) University Hospital, also discussed the problems that might occur with regard to most standard forms of therapy. In particular, they addressed potential impacts of the pandemic on chemotherapy, bone marrow transplantation, maintenance treatments, supportive measures, and targeted therapies.
Of particular concern, “most patients may suffer from postponed chemotherapy, due to a shortage of isolation beds and blood products or the wish to avoid immunosuppressive treatments,” the authors noted, warning that “delay in chemotherapy initiation may negatively affect prognosis, [particularly in patients under age 60] with favorable- or intermediate-risk disease.”
With regard to stem cell transplantation, the authors detail the many potential difficulties with regard to procedures involving both donors and recipients, and warn that in some cases, delay in transplant could result in the reappearance of a significant minimal residual disease, which has a well-established negative impact on survival.
The authors also noted that blood product shortages have already begun in most affected countries, and how, in response, transfusion societies have called for conservative transfusion policies in strict adherence to evidence-based guidelines for patient’s blood management.
“COVID-19 will result in numerous casualties. Acute leukemia patients are at a higher risk of severe complications,” the authors stated. In particular, physicians should especially be aware of how treatment for acute leukemia may have “interactions with other drugs used to treat SARS-CoV-2–related infections/complications such as antibiotics, antiviral drugs, and various other drugs that prolong QTc or impact targeted-therapy pharmacokinetics,” they concluded.
The authors reported that they received no government or private funding for this research, and that they had no conflicts of interest.
SOURCE: Gavillet M et al. Leuk. Res. 2020. doi.org/10.1016/j.leukres.2020.106353.
FROM LEUKEMIA RESEARCH
Blood test might detect multiple cancer types, study suggests
Investigators led by Minetta C. Liu, MD, a medical oncologist with the Mayo Clinic, Rochester, Minn., studied 6,689 participants – 2,482 with cancers of more than 50 types and 4,207 without cancer – drawn from the Circulating Cell-free Genome Atlas Study and the STRIVE Study populations.
The investigators performed bisulfite sequencing that targeted informative methylation regions of plasma cell-free DNA (cfDNA), and developed and validated a molecular classifier using methylation patterns to detect cancer and determine its tissue of origin.
Test performance was assessed both for cancer overall and for a prespecified set of 12 cancers (anus, bladder, colon/rectum, esophagus, head and neck, liver/bile duct, lung, lymphoma, ovary, pancreas, plasma cell neoplasm, stomach) that account for about 63% of U.S. cancer deaths annually.
Results reported this week in the Annals of Oncology showed that the test had a specificity of 99.3% in the validation cohort, corresponding to a false-positive rate of just 0.7%.
Sensitivity for detecting stage I-III disease was 43.9% for cancer overall and 67.3% for the prespecified set of cancers accounting for the majority of U.S. cancer deaths.
Test sensitivity increased with stage both for cancer overall (18%, 43%, 81%, and 93% for stage I, II, III, and IV disease, respectively) and for the prespecified set of cancers (39%, 69%, 83%, and 92%, respectively).
The test was able to predict a tissue of origin in 96% of samples in which a cancerlike signal was detected, and in 93% of cases, that prediction was accurate.
Some of the patients who had cancer were symptomatic and therefore would not be considered a screening population, Dr. Liu and coinvestigators acknowledged. Also, the test’s potential for reducing mortality remains unknown, and 1-year follow-up to verify cancer-free status was not yet available for all of the individuals without cancer.
“Together, these data provide compelling evidence that targeted methylation analysis of cfDNA can detect and localize a broad range of nonmetastatic and metastatic cancer types including many common and deadly cancers that lack effective screening strategies,” they maintained. The test’s “specificity and sensitivity performance approach ... the goal for population-level screening.”
“Considering the potential value of early detection in deadly malignancies, further evaluation of this test is justified in prospective population-level studies,” the investigators conclude. “Clinical validation in intended use populations is ongoing ... and a study has been initiated that is returning results to health care providers and patients ....”
Dr. Liu disclosed that the Mayo Clinic was compensated for her advisory board activities for GRAIL Inc. The study was supported by GRAIL, and by Princess Margaret Cancer Centre’s McCain Genitourinary BioBank in the department of surgical oncology.
SOURCE: Liu MC et al. Ann Oncol. 2020 Mar 31. doi: 10.1016/j.annonc.2020.02.011.
Investigators led by Minetta C. Liu, MD, a medical oncologist with the Mayo Clinic, Rochester, Minn., studied 6,689 participants – 2,482 with cancers of more than 50 types and 4,207 without cancer – drawn from the Circulating Cell-free Genome Atlas Study and the STRIVE Study populations.
The investigators performed bisulfite sequencing that targeted informative methylation regions of plasma cell-free DNA (cfDNA), and developed and validated a molecular classifier using methylation patterns to detect cancer and determine its tissue of origin.
Test performance was assessed both for cancer overall and for a prespecified set of 12 cancers (anus, bladder, colon/rectum, esophagus, head and neck, liver/bile duct, lung, lymphoma, ovary, pancreas, plasma cell neoplasm, stomach) that account for about 63% of U.S. cancer deaths annually.
Results reported this week in the Annals of Oncology showed that the test had a specificity of 99.3% in the validation cohort, corresponding to a false-positive rate of just 0.7%.
Sensitivity for detecting stage I-III disease was 43.9% for cancer overall and 67.3% for the prespecified set of cancers accounting for the majority of U.S. cancer deaths.
Test sensitivity increased with stage both for cancer overall (18%, 43%, 81%, and 93% for stage I, II, III, and IV disease, respectively) and for the prespecified set of cancers (39%, 69%, 83%, and 92%, respectively).
The test was able to predict a tissue of origin in 96% of samples in which a cancerlike signal was detected, and in 93% of cases, that prediction was accurate.
Some of the patients who had cancer were symptomatic and therefore would not be considered a screening population, Dr. Liu and coinvestigators acknowledged. Also, the test’s potential for reducing mortality remains unknown, and 1-year follow-up to verify cancer-free status was not yet available for all of the individuals without cancer.
“Together, these data provide compelling evidence that targeted methylation analysis of cfDNA can detect and localize a broad range of nonmetastatic and metastatic cancer types including many common and deadly cancers that lack effective screening strategies,” they maintained. The test’s “specificity and sensitivity performance approach ... the goal for population-level screening.”
“Considering the potential value of early detection in deadly malignancies, further evaluation of this test is justified in prospective population-level studies,” the investigators conclude. “Clinical validation in intended use populations is ongoing ... and a study has been initiated that is returning results to health care providers and patients ....”
Dr. Liu disclosed that the Mayo Clinic was compensated for her advisory board activities for GRAIL Inc. The study was supported by GRAIL, and by Princess Margaret Cancer Centre’s McCain Genitourinary BioBank in the department of surgical oncology.
SOURCE: Liu MC et al. Ann Oncol. 2020 Mar 31. doi: 10.1016/j.annonc.2020.02.011.
Investigators led by Minetta C. Liu, MD, a medical oncologist with the Mayo Clinic, Rochester, Minn., studied 6,689 participants – 2,482 with cancers of more than 50 types and 4,207 without cancer – drawn from the Circulating Cell-free Genome Atlas Study and the STRIVE Study populations.
The investigators performed bisulfite sequencing that targeted informative methylation regions of plasma cell-free DNA (cfDNA), and developed and validated a molecular classifier using methylation patterns to detect cancer and determine its tissue of origin.
Test performance was assessed both for cancer overall and for a prespecified set of 12 cancers (anus, bladder, colon/rectum, esophagus, head and neck, liver/bile duct, lung, lymphoma, ovary, pancreas, plasma cell neoplasm, stomach) that account for about 63% of U.S. cancer deaths annually.
Results reported this week in the Annals of Oncology showed that the test had a specificity of 99.3% in the validation cohort, corresponding to a false-positive rate of just 0.7%.
Sensitivity for detecting stage I-III disease was 43.9% for cancer overall and 67.3% for the prespecified set of cancers accounting for the majority of U.S. cancer deaths.
Test sensitivity increased with stage both for cancer overall (18%, 43%, 81%, and 93% for stage I, II, III, and IV disease, respectively) and for the prespecified set of cancers (39%, 69%, 83%, and 92%, respectively).
The test was able to predict a tissue of origin in 96% of samples in which a cancerlike signal was detected, and in 93% of cases, that prediction was accurate.
Some of the patients who had cancer were symptomatic and therefore would not be considered a screening population, Dr. Liu and coinvestigators acknowledged. Also, the test’s potential for reducing mortality remains unknown, and 1-year follow-up to verify cancer-free status was not yet available for all of the individuals without cancer.
“Together, these data provide compelling evidence that targeted methylation analysis of cfDNA can detect and localize a broad range of nonmetastatic and metastatic cancer types including many common and deadly cancers that lack effective screening strategies,” they maintained. The test’s “specificity and sensitivity performance approach ... the goal for population-level screening.”
“Considering the potential value of early detection in deadly malignancies, further evaluation of this test is justified in prospective population-level studies,” the investigators conclude. “Clinical validation in intended use populations is ongoing ... and a study has been initiated that is returning results to health care providers and patients ....”
Dr. Liu disclosed that the Mayo Clinic was compensated for her advisory board activities for GRAIL Inc. The study was supported by GRAIL, and by Princess Margaret Cancer Centre’s McCain Genitourinary BioBank in the department of surgical oncology.
SOURCE: Liu MC et al. Ann Oncol. 2020 Mar 31. doi: 10.1016/j.annonc.2020.02.011.
FROM ANNALS OF ONCOLOGY