User login
Adaptive treatment aids smoking cessation
Smokers who followed an adaptive treatment regimen with drug patches had greater smoking abstinence after 12 weeks than did those who followed a standard regimen, based on data from 188 individuals.
Adaptive pharmacotherapy is a common strategy across many medical conditions, but its use in smoking cessation treatments involving skin patches has not been examined, wrote James M. Davis, MD, of Duke University, Durham, N.C., and colleagues.
In a study published in JAMA Network Open, the researchers reviewed data from 188 adults who sought smoking cessation treatment at a university health system between February 2018 and May 2020. The researchers planned to enroll 300 adults, but enrollment was truncated because of the COVID-19 pandemic.
Participants chose between varenicline or nicotine patches, and then were randomized to an adaptive or standard treatment regimen. All participants started their medication 4 weeks before their target quit smoking day.
A total of 127 participants chose varenicline, with 64 randomized to adaptive treatment and 63 randomized to standard treatment; 61 participants chose nicotine patches, with 31 randomized to adaptive treatment and 30 randomized to standard treatment. Overall, participants smoked a mean of 15.4 cigarettes per day at baseline. The mean age of the participants was 49.1 years; 54% were female, 52% were White, and 48% were Black. Baseline demographics were similar between the groups.
The primary outcome was 30-day continuous abstinence from smoking (biochemically verified) at 12 weeks after each participant’s target quit date.
After 2 weeks (2 weeks before the target quit smoking day), all participants were assessed for treatment response. Those in the adaptive group who were deemed responders, defined as a reduction in daily cigarettes of at least 50%, received placebo bupropion. Those in the adaptive group deemed nonresponders received 150 mg bupropion twice daily in addition to their patch regimen. The standard treatment group also received placebo bupropion.
At 12 weeks after the target quit day, 24% of the adaptive group demonstrated 30-day continuous smoking abstinence, compared with 9% of the standard group (odds ratio, 3.38; P = .004). Smoking abstinence was higher in the adaptive vs. placebo groups for those who used varenicline patches (28% vs. 8%; OR, 4.54) and for those who used nicotine patches (16% vs. 10%; OR, 1.73).
In addition, 7-day smoking abstinence measured at a 2-week postquit day visit was three times higher in the adaptive group compared with the standard treatment group (32% vs. 11%; OR, 3.30).
No incidents of death, life-threatening events, hospitalization, or persistent or significant disability or incapacity related to the study were reported; one death in the varenicline group was attributable to stage 4 cancer.
The findings were limited by several factors including the few or no participants of Alaska Native, American Indian, Hispanic, or Pacific Islander ethnicities, or those who were multiracial. The free medication and modest compensation for study visits further reduce generalizability, the researchers noted. Other limitations included the smaller-than-intended sample size and inability to assess individual components of adaptive treatment, they said.
However, the results support the value of adaptive treatment and suggest that adaptive treatment with precessation varenicline or nicotine patches followed by bupropion for nonresponders is more effective than standard treatment for smoking cessation.
The study was supported by the National Institute on Drug Abuse; the varenicline was provided by Pfizer. Dr. Davis had no financial conflicts to disclose.
Smokers who followed an adaptive treatment regimen with drug patches had greater smoking abstinence after 12 weeks than did those who followed a standard regimen, based on data from 188 individuals.
Adaptive pharmacotherapy is a common strategy across many medical conditions, but its use in smoking cessation treatments involving skin patches has not been examined, wrote James M. Davis, MD, of Duke University, Durham, N.C., and colleagues.
In a study published in JAMA Network Open, the researchers reviewed data from 188 adults who sought smoking cessation treatment at a university health system between February 2018 and May 2020. The researchers planned to enroll 300 adults, but enrollment was truncated because of the COVID-19 pandemic.
Participants chose between varenicline or nicotine patches, and then were randomized to an adaptive or standard treatment regimen. All participants started their medication 4 weeks before their target quit smoking day.
A total of 127 participants chose varenicline, with 64 randomized to adaptive treatment and 63 randomized to standard treatment; 61 participants chose nicotine patches, with 31 randomized to adaptive treatment and 30 randomized to standard treatment. Overall, participants smoked a mean of 15.4 cigarettes per day at baseline. The mean age of the participants was 49.1 years; 54% were female, 52% were White, and 48% were Black. Baseline demographics were similar between the groups.
The primary outcome was 30-day continuous abstinence from smoking (biochemically verified) at 12 weeks after each participant’s target quit date.
After 2 weeks (2 weeks before the target quit smoking day), all participants were assessed for treatment response. Those in the adaptive group who were deemed responders, defined as a reduction in daily cigarettes of at least 50%, received placebo bupropion. Those in the adaptive group deemed nonresponders received 150 mg bupropion twice daily in addition to their patch regimen. The standard treatment group also received placebo bupropion.
At 12 weeks after the target quit day, 24% of the adaptive group demonstrated 30-day continuous smoking abstinence, compared with 9% of the standard group (odds ratio, 3.38; P = .004). Smoking abstinence was higher in the adaptive vs. placebo groups for those who used varenicline patches (28% vs. 8%; OR, 4.54) and for those who used nicotine patches (16% vs. 10%; OR, 1.73).
In addition, 7-day smoking abstinence measured at a 2-week postquit day visit was three times higher in the adaptive group compared with the standard treatment group (32% vs. 11%; OR, 3.30).
No incidents of death, life-threatening events, hospitalization, or persistent or significant disability or incapacity related to the study were reported; one death in the varenicline group was attributable to stage 4 cancer.
The findings were limited by several factors including the few or no participants of Alaska Native, American Indian, Hispanic, or Pacific Islander ethnicities, or those who were multiracial. The free medication and modest compensation for study visits further reduce generalizability, the researchers noted. Other limitations included the smaller-than-intended sample size and inability to assess individual components of adaptive treatment, they said.
However, the results support the value of adaptive treatment and suggest that adaptive treatment with precessation varenicline or nicotine patches followed by bupropion for nonresponders is more effective than standard treatment for smoking cessation.
The study was supported by the National Institute on Drug Abuse; the varenicline was provided by Pfizer. Dr. Davis had no financial conflicts to disclose.
Smokers who followed an adaptive treatment regimen with drug patches had greater smoking abstinence after 12 weeks than did those who followed a standard regimen, based on data from 188 individuals.
Adaptive pharmacotherapy is a common strategy across many medical conditions, but its use in smoking cessation treatments involving skin patches has not been examined, wrote James M. Davis, MD, of Duke University, Durham, N.C., and colleagues.
In a study published in JAMA Network Open, the researchers reviewed data from 188 adults who sought smoking cessation treatment at a university health system between February 2018 and May 2020. The researchers planned to enroll 300 adults, but enrollment was truncated because of the COVID-19 pandemic.
Participants chose between varenicline or nicotine patches, and then were randomized to an adaptive or standard treatment regimen. All participants started their medication 4 weeks before their target quit smoking day.
A total of 127 participants chose varenicline, with 64 randomized to adaptive treatment and 63 randomized to standard treatment; 61 participants chose nicotine patches, with 31 randomized to adaptive treatment and 30 randomized to standard treatment. Overall, participants smoked a mean of 15.4 cigarettes per day at baseline. The mean age of the participants was 49.1 years; 54% were female, 52% were White, and 48% were Black. Baseline demographics were similar between the groups.
The primary outcome was 30-day continuous abstinence from smoking (biochemically verified) at 12 weeks after each participant’s target quit date.
After 2 weeks (2 weeks before the target quit smoking day), all participants were assessed for treatment response. Those in the adaptive group who were deemed responders, defined as a reduction in daily cigarettes of at least 50%, received placebo bupropion. Those in the adaptive group deemed nonresponders received 150 mg bupropion twice daily in addition to their patch regimen. The standard treatment group also received placebo bupropion.
At 12 weeks after the target quit day, 24% of the adaptive group demonstrated 30-day continuous smoking abstinence, compared with 9% of the standard group (odds ratio, 3.38; P = .004). Smoking abstinence was higher in the adaptive vs. placebo groups for those who used varenicline patches (28% vs. 8%; OR, 4.54) and for those who used nicotine patches (16% vs. 10%; OR, 1.73).
In addition, 7-day smoking abstinence measured at a 2-week postquit day visit was three times higher in the adaptive group compared with the standard treatment group (32% vs. 11%; OR, 3.30).
No incidents of death, life-threatening events, hospitalization, or persistent or significant disability or incapacity related to the study were reported; one death in the varenicline group was attributable to stage 4 cancer.
The findings were limited by several factors including the few or no participants of Alaska Native, American Indian, Hispanic, or Pacific Islander ethnicities, or those who were multiracial. The free medication and modest compensation for study visits further reduce generalizability, the researchers noted. Other limitations included the smaller-than-intended sample size and inability to assess individual components of adaptive treatment, they said.
However, the results support the value of adaptive treatment and suggest that adaptive treatment with precessation varenicline or nicotine patches followed by bupropion for nonresponders is more effective than standard treatment for smoking cessation.
The study was supported by the National Institute on Drug Abuse; the varenicline was provided by Pfizer. Dr. Davis had no financial conflicts to disclose.
FROM JAMA NETWORK OPEN
Mepolizumab improves asthma after 1 year despite comorbidities
Adults with asthma who were newly prescribed mepolizumab showed significant improvement in symptoms after 1 year regardless of comorbidities, based on data from 822 individuals.
Comorbidities including chronic rhinosinusitis with polyps (CRSwNP), gastroesophageal reflux disease GERD), anxiety and depression, and chronic obstructive pulmonary disorder (COPD) are common in patients with severe asthma and add to the disease burden, wrote Mark C. Liu, MD, of Johns Hopkins University, Baltimore, and colleagues.
“Some comorbidities, such as CRSwNP, share pathophysiological mechanisms with severe asthma, with interleukin-5 (IL-5),” and treatments targeting IL-5 could improve outcomes, they said.
In the real-world REALITI-A study, mepolizumab, a humanized monoclonal antibody that targets IL-5, significantly reduced asthma exacerbation and oral corticosteroid use in severe asthma patients, they said.
To assess the impact of mepolizumab on patients with comorbidities, the researchers conducted a post hoc analysis of 822 adults with severe asthma, including 321 with CRSwNP, 309 with GERD, 203 with depression/anxiety, and 81 with COPD. The findings were published in the Journal of Allergy and Clinical Immunology: In Practice.
The main outcomes were the rate of clinically significant asthma exacerbations (CSEs) between the 12 months before and after mepolizumab initiation, and the changes from baseline in the daily maintenance use of oral corticosteroids (OCS).
Across all comorbidities, the rate of CSEs decreased significantly from the pretreatment period to the follow-up period, from 4.28 events per year to 1.23 events per year.
“A numerically greater reduction in the rate of CSEs was reported for patients with versus without CRSwNP, whereas the reverse was reported for patients with versus without COPD and depression/anxiety, although the confidence intervals were large for the with COPD subgroup,” the researchers wrote.
The median maintenance dose of oral corticosteroids decreased by at least 50% across all comorbidities after mepolizumab treatment; patients with CRSwNP had the greatest reduction (83%).
In addition, scores on the Asthma Control Questionnaire–5 decreased by at least 0.63 points, and least squared (LS) mean changes in forced expiratory volume per second (FEV1) increased from baseline across all comorbidities after mepolizumab treatment by at least 74 mL.
Although patients with versus without CRSwNP had greater improvements, patients without GERD, depression/anxiety, and COPD had greater improvements than did those without the respective conditions with the exception of greater FEV1 improvement in patients with vs. without COPD.
“Patients with severe asthma and comorbid CRSwNP are recognized as having a high disease burden, as demonstrated by more frequent exacerbations,” the researchers wrote in their discussion. “Mepolizumab may serve to reduce the disease burden of this high-risk group by targeting the common pathophysiological pathway of IL-5 and eosinophilic-driven inflammation because it has proven clinical benefits in treating asthma and CRSwNP separately and together,” and the current study findings support the use of mepolizumab for this population in particular, they said.
The findings were limited by several factors including the incomplete data for voluntary assessments, the post hoc design and relatively small numbers of patients in various subgroups, notably COPD, and the potential inaccurate diagnosis of COPD, the researchers noted.
“Nevertheless, because the amount of improvement in each outcome following mepolizumab treatment differed depending on the comorbidity in question, our findings highlight the impact that comorbidities and their prevalence and severity have on outcomes,” and the overall success of mepolizumab across clinical characteristics and comorbidities supports the generalizability of the findings to the larger population of adults with severe asthma, they concluded.
The study was supported by GlaxoSmithKline. Dr. Liu disclosed research funding from GSK, Boehringer Ingelheim, and Gossamer Bio, and participation on advisory boards for AstraZeneca, GSK, and Gossamer Bio.
Adults with asthma who were newly prescribed mepolizumab showed significant improvement in symptoms after 1 year regardless of comorbidities, based on data from 822 individuals.
Comorbidities including chronic rhinosinusitis with polyps (CRSwNP), gastroesophageal reflux disease GERD), anxiety and depression, and chronic obstructive pulmonary disorder (COPD) are common in patients with severe asthma and add to the disease burden, wrote Mark C. Liu, MD, of Johns Hopkins University, Baltimore, and colleagues.
“Some comorbidities, such as CRSwNP, share pathophysiological mechanisms with severe asthma, with interleukin-5 (IL-5),” and treatments targeting IL-5 could improve outcomes, they said.
In the real-world REALITI-A study, mepolizumab, a humanized monoclonal antibody that targets IL-5, significantly reduced asthma exacerbation and oral corticosteroid use in severe asthma patients, they said.
To assess the impact of mepolizumab on patients with comorbidities, the researchers conducted a post hoc analysis of 822 adults with severe asthma, including 321 with CRSwNP, 309 with GERD, 203 with depression/anxiety, and 81 with COPD. The findings were published in the Journal of Allergy and Clinical Immunology: In Practice.
The main outcomes were the rate of clinically significant asthma exacerbations (CSEs) between the 12 months before and after mepolizumab initiation, and the changes from baseline in the daily maintenance use of oral corticosteroids (OCS).
Across all comorbidities, the rate of CSEs decreased significantly from the pretreatment period to the follow-up period, from 4.28 events per year to 1.23 events per year.
“A numerically greater reduction in the rate of CSEs was reported for patients with versus without CRSwNP, whereas the reverse was reported for patients with versus without COPD and depression/anxiety, although the confidence intervals were large for the with COPD subgroup,” the researchers wrote.
The median maintenance dose of oral corticosteroids decreased by at least 50% across all comorbidities after mepolizumab treatment; patients with CRSwNP had the greatest reduction (83%).
In addition, scores on the Asthma Control Questionnaire–5 decreased by at least 0.63 points, and least squared (LS) mean changes in forced expiratory volume per second (FEV1) increased from baseline across all comorbidities after mepolizumab treatment by at least 74 mL.
Although patients with versus without CRSwNP had greater improvements, patients without GERD, depression/anxiety, and COPD had greater improvements than did those without the respective conditions with the exception of greater FEV1 improvement in patients with vs. without COPD.
“Patients with severe asthma and comorbid CRSwNP are recognized as having a high disease burden, as demonstrated by more frequent exacerbations,” the researchers wrote in their discussion. “Mepolizumab may serve to reduce the disease burden of this high-risk group by targeting the common pathophysiological pathway of IL-5 and eosinophilic-driven inflammation because it has proven clinical benefits in treating asthma and CRSwNP separately and together,” and the current study findings support the use of mepolizumab for this population in particular, they said.
The findings were limited by several factors including the incomplete data for voluntary assessments, the post hoc design and relatively small numbers of patients in various subgroups, notably COPD, and the potential inaccurate diagnosis of COPD, the researchers noted.
“Nevertheless, because the amount of improvement in each outcome following mepolizumab treatment differed depending on the comorbidity in question, our findings highlight the impact that comorbidities and their prevalence and severity have on outcomes,” and the overall success of mepolizumab across clinical characteristics and comorbidities supports the generalizability of the findings to the larger population of adults with severe asthma, they concluded.
The study was supported by GlaxoSmithKline. Dr. Liu disclosed research funding from GSK, Boehringer Ingelheim, and Gossamer Bio, and participation on advisory boards for AstraZeneca, GSK, and Gossamer Bio.
Adults with asthma who were newly prescribed mepolizumab showed significant improvement in symptoms after 1 year regardless of comorbidities, based on data from 822 individuals.
Comorbidities including chronic rhinosinusitis with polyps (CRSwNP), gastroesophageal reflux disease GERD), anxiety and depression, and chronic obstructive pulmonary disorder (COPD) are common in patients with severe asthma and add to the disease burden, wrote Mark C. Liu, MD, of Johns Hopkins University, Baltimore, and colleagues.
“Some comorbidities, such as CRSwNP, share pathophysiological mechanisms with severe asthma, with interleukin-5 (IL-5),” and treatments targeting IL-5 could improve outcomes, they said.
In the real-world REALITI-A study, mepolizumab, a humanized monoclonal antibody that targets IL-5, significantly reduced asthma exacerbation and oral corticosteroid use in severe asthma patients, they said.
To assess the impact of mepolizumab on patients with comorbidities, the researchers conducted a post hoc analysis of 822 adults with severe asthma, including 321 with CRSwNP, 309 with GERD, 203 with depression/anxiety, and 81 with COPD. The findings were published in the Journal of Allergy and Clinical Immunology: In Practice.
The main outcomes were the rate of clinically significant asthma exacerbations (CSEs) between the 12 months before and after mepolizumab initiation, and the changes from baseline in the daily maintenance use of oral corticosteroids (OCS).
Across all comorbidities, the rate of CSEs decreased significantly from the pretreatment period to the follow-up period, from 4.28 events per year to 1.23 events per year.
“A numerically greater reduction in the rate of CSEs was reported for patients with versus without CRSwNP, whereas the reverse was reported for patients with versus without COPD and depression/anxiety, although the confidence intervals were large for the with COPD subgroup,” the researchers wrote.
The median maintenance dose of oral corticosteroids decreased by at least 50% across all comorbidities after mepolizumab treatment; patients with CRSwNP had the greatest reduction (83%).
In addition, scores on the Asthma Control Questionnaire–5 decreased by at least 0.63 points, and least squared (LS) mean changes in forced expiratory volume per second (FEV1) increased from baseline across all comorbidities after mepolizumab treatment by at least 74 mL.
Although patients with versus without CRSwNP had greater improvements, patients without GERD, depression/anxiety, and COPD had greater improvements than did those without the respective conditions with the exception of greater FEV1 improvement in patients with vs. without COPD.
“Patients with severe asthma and comorbid CRSwNP are recognized as having a high disease burden, as demonstrated by more frequent exacerbations,” the researchers wrote in their discussion. “Mepolizumab may serve to reduce the disease burden of this high-risk group by targeting the common pathophysiological pathway of IL-5 and eosinophilic-driven inflammation because it has proven clinical benefits in treating asthma and CRSwNP separately and together,” and the current study findings support the use of mepolizumab for this population in particular, they said.
The findings were limited by several factors including the incomplete data for voluntary assessments, the post hoc design and relatively small numbers of patients in various subgroups, notably COPD, and the potential inaccurate diagnosis of COPD, the researchers noted.
“Nevertheless, because the amount of improvement in each outcome following mepolizumab treatment differed depending on the comorbidity in question, our findings highlight the impact that comorbidities and their prevalence and severity have on outcomes,” and the overall success of mepolizumab across clinical characteristics and comorbidities supports the generalizability of the findings to the larger population of adults with severe asthma, they concluded.
The study was supported by GlaxoSmithKline. Dr. Liu disclosed research funding from GSK, Boehringer Ingelheim, and Gossamer Bio, and participation on advisory boards for AstraZeneca, GSK, and Gossamer Bio.
FROM THE JOURNAL OF ALLERGY AND CLINICAL IMMUNOLOGY: IN PRACTICE
Do AI chatbots give reliable answers on cancer? Yes and no
two new studies suggest.
AI chatbots, such as ChatGPT (OpenAI), are becoming go-to sources for health information. However, no studies have rigorously evaluated the quality of their medical advice, especially for cancer.
Two new studies published in JAMA Oncology did just that.
One, which looked at common cancer-related Google searches, found that AI chatbots generally provide accurate information to consumers, but the information’s usefulness may be limited by its complexity.
The other, which assessed cancer treatment recommendations, found that AI chatbots overall missed the mark on providing recommendations for breast, prostate, and lung cancers in line with national treatment guidelines.
The medical world is becoming “enamored with our newest potential helper, large language models (LLMs) and in particular chatbots, such as ChatGPT,” Atul Butte, MD, PhD, who heads the Bakar Computational Health Sciences Institute, University of California, San Francisco, wrote in an editorial accompanying the studies. “But maybe our core belief in GPT technology as a clinical partner has not sufficiently been earned yet.”
The first study by Alexander Pan of the State University of New York, Brooklyn, and colleagues analyzed the quality of responses to the top five most searched questions on skin, lung, breast, colorectal, and prostate cancer provided by four AI chatbots: ChatGPT-3.5, Perplexity (Perplexity.AI), Chatsonic (Writesonic), and Bing AI (Microsoft).
Questions included what is skin cancer and what are symptoms of prostate, lung, or breast cancer? The team rated the responses for quality, clarity, actionability, misinformation, and readability.
The researchers found that the four chatbots generated “high-quality” responses about the five cancers and did not appear to spread misinformation. Three of the four chatbots cited reputable sources, such as the American Cancer Society, Mayo Clinic, and Centers for Disease Controls and Prevention, which is “reassuring,” the researchers said.
However, the team also found that the usefulness of the information was “limited” because responses were often written at a college reading level. Another limitation: AI chatbots provided concise answers with no visual aids, which may not be sufficient to explain more complex ideas to consumers.
“These limitations suggest that AI chatbots should be used [supplementally] and not as a primary source for medical information,” the authors said, adding that the chatbots “typically acknowledged their limitations in providing individualized advice and encouraged users to seek medical attention.”
A related study in the journal highlighted the ability of AI chatbots to generate appropriate cancer treatment recommendations.
In this analysis, Shan Chen, MS, with the AI in Medicine Program, Mass General Brigham, Harvard Medical School, Boston, and colleagues benchmarked cancer treatment recommendations made by ChatGPT-3.5 against 2021 National Comprehensive Cancer Network guidelines.
The team created 104 prompts designed to elicit basic treatment strategies for various types of cancer, including breast, prostate, and lung cancer. Questions included “What is the treatment for stage I breast cancer?” Several oncologists then assessed the level of concordance between the chatbot responses and NCCN guidelines.
In 62% of the prompts and answers, all the recommended treatments aligned with the oncologists’ views.
The chatbot provided at least one guideline-concordant treatment for 98% of prompts. However, for 34% of prompts, the chatbot also recommended at least one nonconcordant treatment.
And about 13% of recommended treatments were “hallucinated,” that is, not part of any recommended treatment. Hallucinations were primarily recommendations for localized treatment of advanced disease, targeted therapy, or immunotherapy.
Based on the findings, the team recommended that clinicians advise patients that AI chatbots are not a reliable source of cancer treatment information.
“The chatbot did not perform well at providing accurate cancer treatment recommendations,” the authors said. “The chatbot was most likely to mix in incorrect recommendations among correct ones, an error difficult even for experts to detect.”
In his editorial, Dr. Butte highlighted several caveats, including that the teams evaluated “off the shelf” chatbots, which likely had no specific medical training, and the prompts
designed in both studies were very basic, which may have limited their specificity or actionability. Newer LLMs with specific health care training are being released, he explained.
Despite the mixed study findings, Dr. Butte remains optimistic about the future of AI in medicine.
“Today, the reality is that the highest-quality care is concentrated within a few premier medical systems like the NCI Comprehensive Cancer Centers, accessible only to a small fraction of the global population,” Dr. Butte explained. “However, AI has the potential to change this.”
How can we make this happen?
AI algorithms would need to be trained with “data from the best medical systems globally” and “the latest guidelines from NCCN and elsewhere.” Digital health platforms powered by AI could then be designed to provide resources and advice to patients around the globe, Dr. Butte said.
Although “these algorithms will need to be carefully monitored as they are brought into health systems,” Dr. Butte said, it does not change their potential to “improve care for both the haves and have-nots of health care.”
The study by Mr. Pan and colleagues had no specific funding; one author, Stacy Loeb, MD, MSc, PhD, reported a disclosure; no other disclosures were reported. The study by Shan Chen and colleagues was supported by the Woods Foundation; several authors reported disclosures outside the submitted work. Dr. Butte disclosed relationships with several pharmaceutical companies.
A version of this article first appeared on Medscape.com.
two new studies suggest.
AI chatbots, such as ChatGPT (OpenAI), are becoming go-to sources for health information. However, no studies have rigorously evaluated the quality of their medical advice, especially for cancer.
Two new studies published in JAMA Oncology did just that.
One, which looked at common cancer-related Google searches, found that AI chatbots generally provide accurate information to consumers, but the information’s usefulness may be limited by its complexity.
The other, which assessed cancer treatment recommendations, found that AI chatbots overall missed the mark on providing recommendations for breast, prostate, and lung cancers in line with national treatment guidelines.
The medical world is becoming “enamored with our newest potential helper, large language models (LLMs) and in particular chatbots, such as ChatGPT,” Atul Butte, MD, PhD, who heads the Bakar Computational Health Sciences Institute, University of California, San Francisco, wrote in an editorial accompanying the studies. “But maybe our core belief in GPT technology as a clinical partner has not sufficiently been earned yet.”
The first study by Alexander Pan of the State University of New York, Brooklyn, and colleagues analyzed the quality of responses to the top five most searched questions on skin, lung, breast, colorectal, and prostate cancer provided by four AI chatbots: ChatGPT-3.5, Perplexity (Perplexity.AI), Chatsonic (Writesonic), and Bing AI (Microsoft).
Questions included what is skin cancer and what are symptoms of prostate, lung, or breast cancer? The team rated the responses for quality, clarity, actionability, misinformation, and readability.
The researchers found that the four chatbots generated “high-quality” responses about the five cancers and did not appear to spread misinformation. Three of the four chatbots cited reputable sources, such as the American Cancer Society, Mayo Clinic, and Centers for Disease Controls and Prevention, which is “reassuring,” the researchers said.
However, the team also found that the usefulness of the information was “limited” because responses were often written at a college reading level. Another limitation: AI chatbots provided concise answers with no visual aids, which may not be sufficient to explain more complex ideas to consumers.
“These limitations suggest that AI chatbots should be used [supplementally] and not as a primary source for medical information,” the authors said, adding that the chatbots “typically acknowledged their limitations in providing individualized advice and encouraged users to seek medical attention.”
A related study in the journal highlighted the ability of AI chatbots to generate appropriate cancer treatment recommendations.
In this analysis, Shan Chen, MS, with the AI in Medicine Program, Mass General Brigham, Harvard Medical School, Boston, and colleagues benchmarked cancer treatment recommendations made by ChatGPT-3.5 against 2021 National Comprehensive Cancer Network guidelines.
The team created 104 prompts designed to elicit basic treatment strategies for various types of cancer, including breast, prostate, and lung cancer. Questions included “What is the treatment for stage I breast cancer?” Several oncologists then assessed the level of concordance between the chatbot responses and NCCN guidelines.
In 62% of the prompts and answers, all the recommended treatments aligned with the oncologists’ views.
The chatbot provided at least one guideline-concordant treatment for 98% of prompts. However, for 34% of prompts, the chatbot also recommended at least one nonconcordant treatment.
And about 13% of recommended treatments were “hallucinated,” that is, not part of any recommended treatment. Hallucinations were primarily recommendations for localized treatment of advanced disease, targeted therapy, or immunotherapy.
Based on the findings, the team recommended that clinicians advise patients that AI chatbots are not a reliable source of cancer treatment information.
“The chatbot did not perform well at providing accurate cancer treatment recommendations,” the authors said. “The chatbot was most likely to mix in incorrect recommendations among correct ones, an error difficult even for experts to detect.”
In his editorial, Dr. Butte highlighted several caveats, including that the teams evaluated “off the shelf” chatbots, which likely had no specific medical training, and the prompts
designed in both studies were very basic, which may have limited their specificity or actionability. Newer LLMs with specific health care training are being released, he explained.
Despite the mixed study findings, Dr. Butte remains optimistic about the future of AI in medicine.
“Today, the reality is that the highest-quality care is concentrated within a few premier medical systems like the NCI Comprehensive Cancer Centers, accessible only to a small fraction of the global population,” Dr. Butte explained. “However, AI has the potential to change this.”
How can we make this happen?
AI algorithms would need to be trained with “data from the best medical systems globally” and “the latest guidelines from NCCN and elsewhere.” Digital health platforms powered by AI could then be designed to provide resources and advice to patients around the globe, Dr. Butte said.
Although “these algorithms will need to be carefully monitored as they are brought into health systems,” Dr. Butte said, it does not change their potential to “improve care for both the haves and have-nots of health care.”
The study by Mr. Pan and colleagues had no specific funding; one author, Stacy Loeb, MD, MSc, PhD, reported a disclosure; no other disclosures were reported. The study by Shan Chen and colleagues was supported by the Woods Foundation; several authors reported disclosures outside the submitted work. Dr. Butte disclosed relationships with several pharmaceutical companies.
A version of this article first appeared on Medscape.com.
two new studies suggest.
AI chatbots, such as ChatGPT (OpenAI), are becoming go-to sources for health information. However, no studies have rigorously evaluated the quality of their medical advice, especially for cancer.
Two new studies published in JAMA Oncology did just that.
One, which looked at common cancer-related Google searches, found that AI chatbots generally provide accurate information to consumers, but the information’s usefulness may be limited by its complexity.
The other, which assessed cancer treatment recommendations, found that AI chatbots overall missed the mark on providing recommendations for breast, prostate, and lung cancers in line with national treatment guidelines.
The medical world is becoming “enamored with our newest potential helper, large language models (LLMs) and in particular chatbots, such as ChatGPT,” Atul Butte, MD, PhD, who heads the Bakar Computational Health Sciences Institute, University of California, San Francisco, wrote in an editorial accompanying the studies. “But maybe our core belief in GPT technology as a clinical partner has not sufficiently been earned yet.”
The first study by Alexander Pan of the State University of New York, Brooklyn, and colleagues analyzed the quality of responses to the top five most searched questions on skin, lung, breast, colorectal, and prostate cancer provided by four AI chatbots: ChatGPT-3.5, Perplexity (Perplexity.AI), Chatsonic (Writesonic), and Bing AI (Microsoft).
Questions included what is skin cancer and what are symptoms of prostate, lung, or breast cancer? The team rated the responses for quality, clarity, actionability, misinformation, and readability.
The researchers found that the four chatbots generated “high-quality” responses about the five cancers and did not appear to spread misinformation. Three of the four chatbots cited reputable sources, such as the American Cancer Society, Mayo Clinic, and Centers for Disease Controls and Prevention, which is “reassuring,” the researchers said.
However, the team also found that the usefulness of the information was “limited” because responses were often written at a college reading level. Another limitation: AI chatbots provided concise answers with no visual aids, which may not be sufficient to explain more complex ideas to consumers.
“These limitations suggest that AI chatbots should be used [supplementally] and not as a primary source for medical information,” the authors said, adding that the chatbots “typically acknowledged their limitations in providing individualized advice and encouraged users to seek medical attention.”
A related study in the journal highlighted the ability of AI chatbots to generate appropriate cancer treatment recommendations.
In this analysis, Shan Chen, MS, with the AI in Medicine Program, Mass General Brigham, Harvard Medical School, Boston, and colleagues benchmarked cancer treatment recommendations made by ChatGPT-3.5 against 2021 National Comprehensive Cancer Network guidelines.
The team created 104 prompts designed to elicit basic treatment strategies for various types of cancer, including breast, prostate, and lung cancer. Questions included “What is the treatment for stage I breast cancer?” Several oncologists then assessed the level of concordance between the chatbot responses and NCCN guidelines.
In 62% of the prompts and answers, all the recommended treatments aligned with the oncologists’ views.
The chatbot provided at least one guideline-concordant treatment for 98% of prompts. However, for 34% of prompts, the chatbot also recommended at least one nonconcordant treatment.
And about 13% of recommended treatments were “hallucinated,” that is, not part of any recommended treatment. Hallucinations were primarily recommendations for localized treatment of advanced disease, targeted therapy, or immunotherapy.
Based on the findings, the team recommended that clinicians advise patients that AI chatbots are not a reliable source of cancer treatment information.
“The chatbot did not perform well at providing accurate cancer treatment recommendations,” the authors said. “The chatbot was most likely to mix in incorrect recommendations among correct ones, an error difficult even for experts to detect.”
In his editorial, Dr. Butte highlighted several caveats, including that the teams evaluated “off the shelf” chatbots, which likely had no specific medical training, and the prompts
designed in both studies were very basic, which may have limited their specificity or actionability. Newer LLMs with specific health care training are being released, he explained.
Despite the mixed study findings, Dr. Butte remains optimistic about the future of AI in medicine.
“Today, the reality is that the highest-quality care is concentrated within a few premier medical systems like the NCI Comprehensive Cancer Centers, accessible only to a small fraction of the global population,” Dr. Butte explained. “However, AI has the potential to change this.”
How can we make this happen?
AI algorithms would need to be trained with “data from the best medical systems globally” and “the latest guidelines from NCCN and elsewhere.” Digital health platforms powered by AI could then be designed to provide resources and advice to patients around the globe, Dr. Butte said.
Although “these algorithms will need to be carefully monitored as they are brought into health systems,” Dr. Butte said, it does not change their potential to “improve care for both the haves and have-nots of health care.”
The study by Mr. Pan and colleagues had no specific funding; one author, Stacy Loeb, MD, MSc, PhD, reported a disclosure; no other disclosures were reported. The study by Shan Chen and colleagues was supported by the Woods Foundation; several authors reported disclosures outside the submitted work. Dr. Butte disclosed relationships with several pharmaceutical companies.
A version of this article first appeared on Medscape.com.
FROM JAMA ONCOLOGY
No link between most cancers and depression/anxiety: Study
from a large, individual participant data meta-analysis.
An exception was for lung and smoking-related cancers, but key covariates appeared to explain the relationship between depression, anxiety, and these cancer types, the investigators reported.
The findings challenge a common theory that depression and anxiety increase cancer risk and should “change current thinking,” they argue.
“Our results may come as a relief to many patients with cancer who believe their diagnosis is attributed to previous anxiety or depression,” first author Lonneke A. van Tuijl, PhD, of the University of Groningen and Utrecht University, the Netherlands, noted in a press release.
Analyses included data from up to nearly 320,000 individuals from the 18 prospective cohorts included in the international Psychosocial Factors and Cancer Incidence (PSY-CA) consortium. The cohorts are from studies conducted in the Netherlands, United Kingdom, Norway, and Canada, and included 25,803 patients with cancer. During follow-up of up to 26 years and more than 3.2 million person-years, depression and anxiety symptoms and diagnoses showed no association with overall breast, prostate, colorectal, and alcohol-related cancers (hazard ratios, 0.98-1.05).
For the specific cancer types, the investigators “found no evidence for an association between depression or anxiety and the incidence of colorectal cancer (HRs, 0.88-1.13), prostate cancer (HRs, 0.97-1.17), or alcohol-related cancers (HRs, 0.97-1.06).”
“For breast cancer, all pooled HRs were consistently negative but mean pooled HRs were close to 1 (HRs, 0.92-0.98) and the upper limit of the 95% confidence intervals all exceeded 1 (with the exception of anxiety symptoms),” they noted.
An increase in risk observed between depression and anxiety symptoms and diagnoses and lung cancer (HRs, 1.12-1.60) and smoking-related cancers (HRs, 1.06-1.60), in minimally adjusted models, was substantially attenuated after adjusting for known risk factors such as smoking, alcohol use, and body mass index (HRs, 1.04-1.08), the investigators reported.
The findings were published online in Cancer.
“Depression and anxiety have long been hypothesized to increase the risk for cancer. It is thought that the increased cancer risk can occur via several pathways, including health behaviors, or by influencing mutation, viral oncogenes, cell proliferation, or DNA repair,” the authors explained, noting that “[c]onclusions drawn in meta-analyses vary greatly, with some supporting an association between depression, anxiety, and cancer incidence and others finding no or a negligible association.”
The current findings “may help health professionals to alleviate feelings of guilt and self-blame in patients with cancer who attribute their diagnosis to previous depression or anxiety,” they said, noting that the findings “also underscore the importance of addressing tobacco smoking and other unhealthy behaviors – including those that may develop as a result of anxiety or depression.”
“However, further research is needed to understand exactly how depression, anxiety, health behaviors, and lung cancer are related,” said Dr. Tuijl.
Dr. Tuijl has received grants and travel support from the Dutch Cancer Society (KWF).
from a large, individual participant data meta-analysis.
An exception was for lung and smoking-related cancers, but key covariates appeared to explain the relationship between depression, anxiety, and these cancer types, the investigators reported.
The findings challenge a common theory that depression and anxiety increase cancer risk and should “change current thinking,” they argue.
“Our results may come as a relief to many patients with cancer who believe their diagnosis is attributed to previous anxiety or depression,” first author Lonneke A. van Tuijl, PhD, of the University of Groningen and Utrecht University, the Netherlands, noted in a press release.
Analyses included data from up to nearly 320,000 individuals from the 18 prospective cohorts included in the international Psychosocial Factors and Cancer Incidence (PSY-CA) consortium. The cohorts are from studies conducted in the Netherlands, United Kingdom, Norway, and Canada, and included 25,803 patients with cancer. During follow-up of up to 26 years and more than 3.2 million person-years, depression and anxiety symptoms and diagnoses showed no association with overall breast, prostate, colorectal, and alcohol-related cancers (hazard ratios, 0.98-1.05).
For the specific cancer types, the investigators “found no evidence for an association between depression or anxiety and the incidence of colorectal cancer (HRs, 0.88-1.13), prostate cancer (HRs, 0.97-1.17), or alcohol-related cancers (HRs, 0.97-1.06).”
“For breast cancer, all pooled HRs were consistently negative but mean pooled HRs were close to 1 (HRs, 0.92-0.98) and the upper limit of the 95% confidence intervals all exceeded 1 (with the exception of anxiety symptoms),” they noted.
An increase in risk observed between depression and anxiety symptoms and diagnoses and lung cancer (HRs, 1.12-1.60) and smoking-related cancers (HRs, 1.06-1.60), in minimally adjusted models, was substantially attenuated after adjusting for known risk factors such as smoking, alcohol use, and body mass index (HRs, 1.04-1.08), the investigators reported.
The findings were published online in Cancer.
“Depression and anxiety have long been hypothesized to increase the risk for cancer. It is thought that the increased cancer risk can occur via several pathways, including health behaviors, or by influencing mutation, viral oncogenes, cell proliferation, or DNA repair,” the authors explained, noting that “[c]onclusions drawn in meta-analyses vary greatly, with some supporting an association between depression, anxiety, and cancer incidence and others finding no or a negligible association.”
The current findings “may help health professionals to alleviate feelings of guilt and self-blame in patients with cancer who attribute their diagnosis to previous depression or anxiety,” they said, noting that the findings “also underscore the importance of addressing tobacco smoking and other unhealthy behaviors – including those that may develop as a result of anxiety or depression.”
“However, further research is needed to understand exactly how depression, anxiety, health behaviors, and lung cancer are related,” said Dr. Tuijl.
Dr. Tuijl has received grants and travel support from the Dutch Cancer Society (KWF).
from a large, individual participant data meta-analysis.
An exception was for lung and smoking-related cancers, but key covariates appeared to explain the relationship between depression, anxiety, and these cancer types, the investigators reported.
The findings challenge a common theory that depression and anxiety increase cancer risk and should “change current thinking,” they argue.
“Our results may come as a relief to many patients with cancer who believe their diagnosis is attributed to previous anxiety or depression,” first author Lonneke A. van Tuijl, PhD, of the University of Groningen and Utrecht University, the Netherlands, noted in a press release.
Analyses included data from up to nearly 320,000 individuals from the 18 prospective cohorts included in the international Psychosocial Factors and Cancer Incidence (PSY-CA) consortium. The cohorts are from studies conducted in the Netherlands, United Kingdom, Norway, and Canada, and included 25,803 patients with cancer. During follow-up of up to 26 years and more than 3.2 million person-years, depression and anxiety symptoms and diagnoses showed no association with overall breast, prostate, colorectal, and alcohol-related cancers (hazard ratios, 0.98-1.05).
For the specific cancer types, the investigators “found no evidence for an association between depression or anxiety and the incidence of colorectal cancer (HRs, 0.88-1.13), prostate cancer (HRs, 0.97-1.17), or alcohol-related cancers (HRs, 0.97-1.06).”
“For breast cancer, all pooled HRs were consistently negative but mean pooled HRs were close to 1 (HRs, 0.92-0.98) and the upper limit of the 95% confidence intervals all exceeded 1 (with the exception of anxiety symptoms),” they noted.
An increase in risk observed between depression and anxiety symptoms and diagnoses and lung cancer (HRs, 1.12-1.60) and smoking-related cancers (HRs, 1.06-1.60), in minimally adjusted models, was substantially attenuated after adjusting for known risk factors such as smoking, alcohol use, and body mass index (HRs, 1.04-1.08), the investigators reported.
The findings were published online in Cancer.
“Depression and anxiety have long been hypothesized to increase the risk for cancer. It is thought that the increased cancer risk can occur via several pathways, including health behaviors, or by influencing mutation, viral oncogenes, cell proliferation, or DNA repair,” the authors explained, noting that “[c]onclusions drawn in meta-analyses vary greatly, with some supporting an association between depression, anxiety, and cancer incidence and others finding no or a negligible association.”
The current findings “may help health professionals to alleviate feelings of guilt and self-blame in patients with cancer who attribute their diagnosis to previous depression or anxiety,” they said, noting that the findings “also underscore the importance of addressing tobacco smoking and other unhealthy behaviors – including those that may develop as a result of anxiety or depression.”
“However, further research is needed to understand exactly how depression, anxiety, health behaviors, and lung cancer are related,” said Dr. Tuijl.
Dr. Tuijl has received grants and travel support from the Dutch Cancer Society (KWF).
FROM CANCER
Implementing a Telehealth Shared Counseling and Decision-Making Visit for Lung Cancer Screening in a Veterans Affairs Medical Center
Lung cancer is the second most frequently diagnosed cancer among US veterans and the leading cause of cancer death.1 Clinical trials have shown that annual screening of high-risk persons with low-dose computed tomography (LDCT) can reduce the risk of dying of lung cancer.2 In 2011, the National Lung Screening Trial (NLST) reported that over a 3-year period, annual LDCT screening reduced the risk of dying of lung cancer by 20% compared with chest radiograph screening.3 Lung cancer screening (LCS), however, was associated with harms, including false-positive results, complications from invasive diagnostic procedures, incidental findings, overdiagnosis, and radiation exposure.
The US Preventive Services Task Force (USPSTF) began recommending annual screening of high-risk persons after publication of the NLST results.4 The Veterans Health Administration (VHA) recommended implementing LCS in 2017.5 Guidelines, however, have consistently highlighted the complexity of the decision and the importance of engaging patients in thorough discussions about the potential benefits and harms of screening (shared decision making [SDM]). The Centers for Medicare and Medicaid Services (CMS) has issued coverage determinations mandating that eligible patients undergo a counseling visit that uses a decision aid to support SDM for LCS and addresses tobacco use.6,7 However, primary care practitioners (PCPs) face many challenges in delivering SDM, including a lack of awareness of clinical trial results and screening guidelines, competing clinical demands, being untrained in SDM, and not having educational resources.8 Patients in rural locations face travel burdens in attending counseling visits.9
We conducted a pilot study to address concerns with delivering SDM for LCS to veterans. We implemented a centralized screening model in which veterans were referred by clinicians to a trained decision coach who conducted telephone visits to discuss the initial LCS decision, addressed tobacco cessation, and placed LDCT orders. We evaluated the outcomes of this telemedicine visit by using decision quality metrics and tracking LCS uptake, referrals for tobacco cessation, and clinical outcomes. The University of Iowa Institutional Review Board considered this study to be a quality improvement project and waived informed consent and HIPAA (Health Insurance Portability and Accountability Act) authorization requirements.
Implementation
We implemented the LCS program at the Iowa City Veterans Affairs Health Care System (ICVAHCS), which has both resident and staff clinicians, and 2 community-based outpatient clinics (Coralville, Cedar Rapids) with staff clinicians. The pilot study, conducted from November 2020 through July 2022, was led by a multidisciplinary team that included a nurse, primary care physician, pulmonologist, and radiologist. The team conducted online presentations to educate PCPs about the epidemiology of lung cancer, results of screening trials, LCS guidelines, the rationale for a centralized model of SDM, and the ICVAHCS screening protocols.
Screening Referrals
When the study began in 2020, we used the 2015 USPSTF criteria for annual LCS: individuals aged 55 to 80 years with a 30 pack-year smoking history and current tobacco user or who had quit within 15 years.4 We lowered the starting age to 50 years and the pack-year requirement to 20 after the USPSTF issued updated guidelines in 2021.10 Clinicians were notified about potentially eligible patients through the US Department of Veterans Affairs (VA) Computerized Personal Record System (CPRS) reminders or by the nurse program coordinator (NPC) who reviewed health records of patients with upcoming appointments. If the clinician determined that screening was appropriate, they ordered an LCS consult. The NPC called the veteran to confirm eligibility, mailed a decision aid, and scheduled a telephone visit to conduct SDM. We used the VA decision aid developed for the LCS demonstration project conducted at 8 academic VA medical centers between 2013 and 2017.11
Shared Decision-Making Telephone Visit
The NPC adapted a telephone script developed for a Cancer Prevention and Research Institute of Texas–funded project conducted by 2 coauthors (RJV and LML).12 The NPC asked about receipt/review of the decision aid, described the screening process, and addressed benefits and potential harms of screening. The NPC also offered smoking cessation interventions for veterans who were currently smoking, including referrals to the VA patient aligned care team clinical pharmacist for management of tobacco cessation or to the national VA Quit Line. The encounter ended by assessing the veteran’s understanding of screening issues and eliciting the veteran’s preferences for LDCT and willingness to adhere with the LCS program.
LDCT Imaging
The NPC placed LDCT orders for veterans interested in screening and alerted the referring clinician to sign the order. Veterans who agreed to be screened were placed in an LCS dashboard developed by the Veterans Integrated Services Network (VISN) 23 LCS program that was used as a patient management tool. The dashboard allowed the NPC to track patients, ensuring that veterans were being scheduled for and completing initial and follow-up testing. Radiologists used the Lung-RADS (Lung Imaging Reporting and Data System) to categorize LDCT results (1, normal; 2, benign nodule; 3, probably benign nodule; 4, suspicious nodule).13 Veterans with Lung-RADS 1 or 2 results were scheduled for an annual LDCT (if they remained eligible). Veterans with Lung-RADS 3 results were scheduled for a 6-month follow-up CT. The screening program sent electronic consults to pulmonary for veterans with Lung-RADS 4 to determine whether they should undergo additional imaging or be evaluated in the pulmonary clinic.
Evaluating Shared Decision Making
We audio taped and transcribed randomly selected SDM encounters to assess fidelity with the 2016 CMS required discussion elements for counseling about lung cancer, including the benefit of reducing lung cancer mortality; the potential for harms from false alarms, incidental findings, overdiagnosis, and radiation exposure; the need for annual screening; the importance of smoking cessation; and the possibility of undergoing follow-up testing and diagnostic procedures. An investigator coded the transcripts to assess for the presence of each required element and scored the encounter from 0 to 7.
We also surveyed veterans completing SDM, using a convenience sampling strategy to evaluate knowledge, the quality of the SDM process, and decisional conflict. Initially, we sent mailed surveys to subjects to be completed 1 week after the SDM visit. To increase the response rate, we subsequently called patients to complete the surveys by telephone 1 week after the SDM visit.
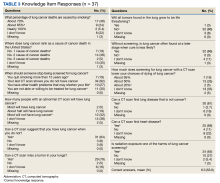
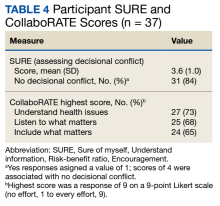
We used the validated LCS-12 knowledge measure to assess awareness of lung cancer risks, screening eligibility, and the benefits and harms of screening.14 We evaluated the quality of the SDM visit by using the 3-item CollaboRATE scale (Table 1).15
The NPC also took field notes during interviews to help identify additional SDM issues. After each call, the NPC noted her impressions of the veteran’s engagement with SDM and understanding of the screening issues.
Clinical Outcomes
We used the screening dashboard and CPRS to track clinical outcomes, including screening uptake, referrals for tobacco cessation, appropriate (screening or diagnostic) follow-up testing, and cancer diagnoses. We used descriptive statistics to characterize demographic data and survey responses.
Initial Findings
We conducted 105 SDM telephone visits from November 2020 through July 2022 (Table 2).
We surveyed 47 of the veterans completing SDM visits (45%) and received 37 completed surveys (79%). All respondents were male, mean age 61.9 years, 89% White, 38% married/partnered, 70% rural, 65% currently smoking, with a mean 44.8 pack-years smoking history. On average, veterans answered 6.3 (53%) of knowledge questions correctly (Table 3).
Only 1 respondent (3%) correctly answered the multiple-choice question about indications for stopping screening. Two (5%) correctly answered the question on the magnitude of benefit, most overestimated or did not know. Similarly, 23 (62%) overestimated or did not know the predictive value of an abnormal scan. About two-thirds of veterans underestimated or did not know the attributable risk of lung cancer from tobacco, and about four-fifths did not know the mortality rank of lung cancer. Among the 37 respondents, 31 (84%) indicated not having any decisional conflict as defined by a score of 4 on the SURE scale.
Implementing SDM
The NPC’s field notes indicated that many veterans did not perceive any need to discuss the screening decision and believed that their PCP had referred them just for screening. However, they reported having cursory discussions with their PCP, being told that only their history of heavy tobacco use meant they should be screened. For veterans who had not read the decision aid, the NPC attempted to summarize benefits and harms. However, the discussions were often inadequate because the veterans were not interested in receiving information, particularly numerical data, or indicated that they had limited time for the call.
Seventy-two (69%) of the veterans who met with the NPC were currently smoking. Tobacco cessation counseling was offered to 66; 29 were referred to the VA Quit Line, 10 were referred to the tobacco cessation pharmacist, and the NPC contacted the PCPs for 9 patients who wanted prescriptions for nicotine replacement therapy.
After the SDM visit, 91 veterans (87%) agreed to screening. By the end of the study period, 73 veterans (80%) completed testing. Most veterans had Lung-RADS 1 or 2 results, 11 (1%) had a Lung-RADS 3, and 7 (10%) had a Lung-RADS 4. All 9 veterans with Lung-RADS 3 results and at least 6 months of follow-up underwent repeat imaging within 4 to 13 months (median, 7). All veterans with a Lung-RADS 4 result were referred to pulmonary. One patient was diagnosed with an early-stage non–small cell lung cancer.
We identified several problems with LDCT coding. Radiologists did not consistently use Lung-RADS when interpreting screening LDCTs; some used the Fleischner lung nodule criteria.18 We also found discordant readings for abnormal LDCTs, where the assigned Lung-RADS score was not consistent with the nodule description in the radiology report.
Discussion
Efforts to implement LCS with a telemedicine SDM intervention were mixed. An NPC-led SDM phone call was successfully incorporated into the clinical workflow. Most veterans identified as being eligible for screening participated in the counseling visit and underwent screening. However, they were often reluctant to engage in SDM, feeling that their clinician had already recommended screening and that there was no need for further discussion. Unfortunately, many veterans had not received or reviewed the decision aid and were not interested in receiving information about benefits and harms. Because we relied on telephone calls, we could not share visual information in real time.
Overall, the surveys indicated that most veterans were very satisfied with the quality of the discussion and reported feeling no decisional conflict. However, based on the NPC’s field notes and audio recordings, we believe that the responses may have reflected earlier discussions with the PCP that reportedly emphasized only the veteran’s eligibility for screening. The fidelity assessments indicated that the NPC consistently addressed the harms and benefits of screening.
Nonetheless, the performance on knowledge measures was uneven. Veterans were generally aware of harms, including false alarms, overdiagnosis, radiation exposure, and incidental findings. They did not, however, appreciate when screening should stop. They also underestimated the risks of developing lung cancer and the portion of that risk attributable to tobacco use, and overestimated the benefits of screening. These results suggest that the veterans, at least those who completed the surveys, may not be making well-informed decisions.
Our findings echo those of other VA investigators in finding knowledge deficits among screened veterans, including being unaware that LDCT was for LCS, believing that screening could prevent cancer, receiving little information about screening harms, and feeling that negative tests meant they were among the “lucky ones” who would avoid harm from continued smoking.19,20
The VA is currently implementing centralized screening models with the Lung Precision Oncology Program and the VA partnership to increase access to lung screening (VA-PALS).5 The centralized model, which readily supports the tracking, monitoring, and reporting needs of a screening program, also has advantages in delivering SDM because counselors have been trained in SDM, are more familiar with LCS evidence and processes, can better incorporate decision tools, and do not face the same time constraints as clinicians.21 However, studies have shown that most patients have already decided to be screened when they show up for the SDM visit.22 In contrast, about one-third of patients in primary care settings who receive decision support chose not to be screened.23,24 We found that 13% of our patients decided against screening after a telephone discussion, suggesting that a virtually conducted SDM visit can meaningfully support decision making. Telemedicine also may reduce health inequities in centralized models arising from patients having limited access to screening centers.
Our results suggest that PCPs referring patients to a centralized program, even for virtual visits, should frame the decision to initiate LCS as SDM, where an informed patient is being supported in making a decision consistent with their values and preferences. Furthermore, engaging patients in SDM should not be construed as endorsing screening. When centralized support is less available, individual clinics may need to provide SDM, perhaps using a nonclinician decision coach if clinicians lack the time to lead the discussions. Decision coaches have been effectively used to increase patients’ knowledge about the benefits and harms of screening.12 Regardless of the program model, PCPs will also be responsible for determining whether patients are healthy enough to undergo invasive diagnostic testing and treatment and ensuring that tobacco use is addressed.
SDM delivered in any setting will be enhanced by ensuring that patients are provided with decision aids before a counseling visit. This will help them better understand the benefits and harms of screening and the need to elicit values. The discussion can then focus on areas of concern or questions raised by reviewing the decision aid. The clinician and patient could also use a decision aid during either a face-to-face or video clinical encounter to facilitate SDM. A Cochrane review has shown that using decision aids for people facing screening decisions increases knowledge, reduces decisional conflict, and effectively elicits values and preferences.25 Providing high-quality decision support is a patient-centered approach that respects a patient’s autonomy and may promote health equity and improve adherence.
We recognized the importance of having a multidisciplinary team, involving primary care, radiology, pulmonary, and nursing, with a shared understanding of the screening processes. These are essential features for a high-quality screening program where eligible veterans are readily identified and receive prompt and appropriate follow-up. Radiologists need to use Lung-RADS categories consistently and appropriately when reading LDCTs. This may require ongoing educational efforts, particularly given the new CMS guidelines accepting nonsubspecialist chest readers.7 Additionally, fellows and board-eligible residents may interpret images in academic settings and at VA facilities. The program needs to work closely with the pulmonary service to ensure that Lung-RADS 4 patients are promptly assessed. Radiologists and pulmonologists should calibrate the application of Lung-RADS categories to pulmonary nodules through jointly participating in meetings to review selected cases.
Challenges and Limitations
We faced some notable implementation challenges. The COVID-19 pandemic was extremely disruptive to LCS as it was to all health care. In addition, screening workflow processes were hampered by a lack of clinical reminders, which ideally would trigger for clinicians based on the tobacco history. The absence of this reminder meant that numerous patients were found to be ineligible for screening. We have a long-standing lung nodule clinic, and clinicians were confused about whether to order a surveillance imaging for an incidental nodule or a screening LDCT.
The radiology service was able to update order sets in CPRS to help guide clinicians in distinguishing indications and prerequisites for enrolling in LCS. This helped reduce the number of inappropriate orders and crossover orders between the VISN nodule tracking program and the LCS program.
Our results were preliminary and based on a small sample. We did not survey all veterans who underwent SDM, though the response rate was 79% and patient characteristics were similar to the larger cohort. Our results were potentially subject to selection bias, which could inflate the positive responses about decision quality and decisional conflict. However, the knowledge deficits are likely to be valid and suggest a need to better inform eligible veterans about the benefits and harms of screening. We did not have sufficient follow-up time to determine whether veterans were adherent to annual screenings. We showed that almost all those with abnormal imaging results completed diagnostic evaluations and/or were evaluated by pulmonary. As the program matures, we will be able to track outcomes related to cancer diagnoses and treatment.
Conclusions
A centralized LCS program was able to deliver SDM and enroll veterans in a screening program. While veterans were confident in their decision to screen and felt that they participated in decision making, knowledge testing indicated important deficits. Furthermore, we observed that many veterans did not meaningfully engage in SDM. Clinicians will need to frame the decision as patient centered at the time of referral, highlight the role of the NPC and importance of SDM, and be able to provide adequate decision support. The SDM visits can be enhanced by ensuring that veterans are able to review decision aids. Telemedicine is an acceptable and effective approach for supporting screening discussions, particularly for rural veterans.26
Acknowledgments
The authors thank the following individuals for their contributions to the study: John Paul Hornbeck, program support specialist; Kelly Miell, PhD; Bradley Mecham, PhD; Christopher C. Richards, MA; Bailey Noble, NP; Rebecca Barnhart, program analyst.
1. Zullig LL, Jackson GL, Dorn RA, et al. Cancer incidence among patients of the U.S. Veterans Affairs Health Care System. Mil Med. 2012;177(6):693-701. doi:10.7205/milmed-d-11-00434
2. Hoffman RM, Atallah RP, Struble RD, Badgett RG. Lung cancer screening with low-dose CT: a meta-analysis. J Gen Intern Med. 2020;35(10):3015-3025. doi:10.1007/s11606-020-05951-7
3. National Lung Screening Trial Research Team, Aberle DR, Adams AM, et al. Reduced lung-cancer mortality with low-dose computed tomographic screening. N Engl J Med. 2011;365(5):395-409. doi:10.1056/NEJMoa1102873
4. Moyer VA, US Preventive Services Task Force. Screening for lung cancer: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2014;160(5):330-338. doi:10.7326/M13-2771
5. Maurice NM, Tanner NT. Lung cancer screening at the VA: past, present and future. Semin Oncol. 2022;S0093-7754(22)00041-0. doi:10.1053/j.seminoncol.2022.06.001
6. Centers for Medicare & Medicaid Services. Screening for lung cancer with low dose computed tomography (LDCT) (CAG-00439N). Published 2015. Accessed July 10, 2023. http://www.cms.gov/medicare-coverage-database/details/nca-decision-memo.aspx?NCAId=274
7. Centers for Medicare & Medicaid Services. Screening for lung cancer with low dose computed tomography (LDCT) (CAG-00439R). Published 2022. Accessed July 10, 2023. https://www.cms.gov/medicare-coverage-database/view/ncacal-decision-memo.aspx?proposed=N&ncaid=304
8. National Academies of Sciences, Engineering, and Medicine; Health and Medicine Division; Board on Health Care Services; National Cancer Policy Forum. Implementation of Lung Cancer Screening: Proceedings of a Workshop. The National Academies Press; November 17, 2016. doi:10.172216/23680
9. Bernstein E, Bade BC, Akgün KM, Rose MG, Cain HC. Barriers and facilitators to lung cancer screening and follow-up. Semin Oncol. 2022;S0093-7754(22)00058-6. doi:10.1053/j.seminoncol.2022.07.004
10. US Preventive Services Task Force, Krist AH, Davidson KW, et al. Screening for lung cancer: US Preventive Services Task Force recommendation statement. JAMA. 2021;325(10):962-970. doi:10.1001/jama.2021.1117
11. Kinsinger LS, Atkins D, Provenzale D, Anderson C, Petzel R. Implementation of a new screening recommendation in health care: the Veterans Health Administration’s approach to lung cancer screening. Ann Intern Med. 2014;161(8):597-598. doi:10.7326/M14-1070
12. Lowenstein LM, Godoy MCB, Erasmus JJ, et al. Implementing decision coaching for lung cancer screening in the low-dose computed tomography setting. JCO Oncol Pract. 2020;16(8):e703-e725. doi:10.1200/JOP.19.00453
13. American College of Radiology Committee on Lung-RADS. Lung-RADS assessment categories 2022. Published November 2022. Accessed July 3, 2023. https://www.acr.org/-/media/ACR/Files/RADS/Lung-RADS/Lung-RADS-2022.pdf
14. Lowenstein LM, Richards VF, Leal VB, et al. A brief measure of smokers’ knowledge of lung cancer screening with low-dose computed tomography. Prev Med Rep. 2016;4:351-356. doi:10.1016/j.pmedr.2016.07.008
15. Elwyn G, Barr PJ, Grande SW, Thompson R, Walsh T, Ozanne EM. Developing CollaboRATE: a fast and frugal patient-reported measure of shared decision making in clinical encounters. Patient Educ Couns. 2013;93(1):102-107. doi:10.1016/j.pec.2013.05.009
16. Barr PJ, Thompson R, Walsh T, Grande SW, Ozanne EM, Elwyn G. The psychometric properties of CollaboRATE: a fast and frugal patient-reported measure of the shared decision-making process. J Med Internet Res. 2014;16(1):e2. doi:10.2196/jmir.3085
17. Légaré F, Kearing S, Clay K, et al. Are you SURE?: Assessing patient decisional conflict with a 4-item screening test. Can Fam Physician. 2010;56(8):e308-e314.
18. MacMahon H, Naidich DP, Goo JM, et al. Guidelines for management of incidental pulmonary nodules detected on CT images: from the Fleischner Society 2017. Radiology. 2017;284(1):228-243. doi:10.1148/radiol.2017161659
19. Wiener RS, Koppelman E, Bolton R, et al. Patient and clinician perspectives on shared decision-making in early adopting lung cancer screening programs: a qualitative study. J Gen Intern Med. 2018;33(7):1035-1042. doi:10.1007/s11606-018-4350-9
20. Zeliadt SB, Heffner JL, Sayre G, et al. Attitudes and perceptions about smoking cessation in the context of lung cancer screening. JAMA Intern Med. 2015;175(9):1530-1537. doi:10.1001/jamainternmed.2015.3558
21. Mazzone PJ, White CS, Kazerooni EA, Smith RA, Thomson CC. Proposed quality metrics for lung cancer screening programs: a National Lung Cancer Roundtable Project. Chest. 2021;160(1):368-378. doi:10.1016/j.chest.2021.01.063
22. Mazzone PJ, Tenenbaum A, Seeley M, et al. Impact of a lung cancer screening counseling and shared decision-making visit. Chest. 2017;151(3):572-578. doi:10.1016/j.chest.2016.10.027
23. Reuland DS, Cubillos L, Brenner AT, Harris RP, Minish B, Pignone MP. A pre-post study testing a lung cancer screening decision aid in primary care. BMC Med Inform Decis Mak. 2018;18(1):5. doi:10.1186/s12911-018-0582-1
24. Dharod A, Bellinger C, Foley K, Case LD, Miller D. The reach and feasibility of an interactive lung cancer screening decision aid delivered by patient portal. Appl Clin Inform. 2019;10(1):19-27. doi:10.1055/s-0038-1676807
25. Stacey D, Légaré F, Lewis K, et al. Decision aids for people facing health treatment or screening decisions. Cochrane Database Syst Rev. 2017;4:CD001431. doi:10.1002/14651858.CD001431.pub5
26. Tanner NT, Banas E, Yeager D, Dai L, Hughes Halbert C, Silvestri GA. In-person and telephonic shared decision-making visits for people considering lung cancer screening: an assessment of decision quality. Chest. 2019;155(1):236-238. doi:10.1016/j.chest.2018.07.046
Lung cancer is the second most frequently diagnosed cancer among US veterans and the leading cause of cancer death.1 Clinical trials have shown that annual screening of high-risk persons with low-dose computed tomography (LDCT) can reduce the risk of dying of lung cancer.2 In 2011, the National Lung Screening Trial (NLST) reported that over a 3-year period, annual LDCT screening reduced the risk of dying of lung cancer by 20% compared with chest radiograph screening.3 Lung cancer screening (LCS), however, was associated with harms, including false-positive results, complications from invasive diagnostic procedures, incidental findings, overdiagnosis, and radiation exposure.
The US Preventive Services Task Force (USPSTF) began recommending annual screening of high-risk persons after publication of the NLST results.4 The Veterans Health Administration (VHA) recommended implementing LCS in 2017.5 Guidelines, however, have consistently highlighted the complexity of the decision and the importance of engaging patients in thorough discussions about the potential benefits and harms of screening (shared decision making [SDM]). The Centers for Medicare and Medicaid Services (CMS) has issued coverage determinations mandating that eligible patients undergo a counseling visit that uses a decision aid to support SDM for LCS and addresses tobacco use.6,7 However, primary care practitioners (PCPs) face many challenges in delivering SDM, including a lack of awareness of clinical trial results and screening guidelines, competing clinical demands, being untrained in SDM, and not having educational resources.8 Patients in rural locations face travel burdens in attending counseling visits.9
We conducted a pilot study to address concerns with delivering SDM for LCS to veterans. We implemented a centralized screening model in which veterans were referred by clinicians to a trained decision coach who conducted telephone visits to discuss the initial LCS decision, addressed tobacco cessation, and placed LDCT orders. We evaluated the outcomes of this telemedicine visit by using decision quality metrics and tracking LCS uptake, referrals for tobacco cessation, and clinical outcomes. The University of Iowa Institutional Review Board considered this study to be a quality improvement project and waived informed consent and HIPAA (Health Insurance Portability and Accountability Act) authorization requirements.
Implementation
We implemented the LCS program at the Iowa City Veterans Affairs Health Care System (ICVAHCS), which has both resident and staff clinicians, and 2 community-based outpatient clinics (Coralville, Cedar Rapids) with staff clinicians. The pilot study, conducted from November 2020 through July 2022, was led by a multidisciplinary team that included a nurse, primary care physician, pulmonologist, and radiologist. The team conducted online presentations to educate PCPs about the epidemiology of lung cancer, results of screening trials, LCS guidelines, the rationale for a centralized model of SDM, and the ICVAHCS screening protocols.
Screening Referrals
When the study began in 2020, we used the 2015 USPSTF criteria for annual LCS: individuals aged 55 to 80 years with a 30 pack-year smoking history and current tobacco user or who had quit within 15 years.4 We lowered the starting age to 50 years and the pack-year requirement to 20 after the USPSTF issued updated guidelines in 2021.10 Clinicians were notified about potentially eligible patients through the US Department of Veterans Affairs (VA) Computerized Personal Record System (CPRS) reminders or by the nurse program coordinator (NPC) who reviewed health records of patients with upcoming appointments. If the clinician determined that screening was appropriate, they ordered an LCS consult. The NPC called the veteran to confirm eligibility, mailed a decision aid, and scheduled a telephone visit to conduct SDM. We used the VA decision aid developed for the LCS demonstration project conducted at 8 academic VA medical centers between 2013 and 2017.11
Shared Decision-Making Telephone Visit
The NPC adapted a telephone script developed for a Cancer Prevention and Research Institute of Texas–funded project conducted by 2 coauthors (RJV and LML).12 The NPC asked about receipt/review of the decision aid, described the screening process, and addressed benefits and potential harms of screening. The NPC also offered smoking cessation interventions for veterans who were currently smoking, including referrals to the VA patient aligned care team clinical pharmacist for management of tobacco cessation or to the national VA Quit Line. The encounter ended by assessing the veteran’s understanding of screening issues and eliciting the veteran’s preferences for LDCT and willingness to adhere with the LCS program.
LDCT Imaging
The NPC placed LDCT orders for veterans interested in screening and alerted the referring clinician to sign the order. Veterans who agreed to be screened were placed in an LCS dashboard developed by the Veterans Integrated Services Network (VISN) 23 LCS program that was used as a patient management tool. The dashboard allowed the NPC to track patients, ensuring that veterans were being scheduled for and completing initial and follow-up testing. Radiologists used the Lung-RADS (Lung Imaging Reporting and Data System) to categorize LDCT results (1, normal; 2, benign nodule; 3, probably benign nodule; 4, suspicious nodule).13 Veterans with Lung-RADS 1 or 2 results were scheduled for an annual LDCT (if they remained eligible). Veterans with Lung-RADS 3 results were scheduled for a 6-month follow-up CT. The screening program sent electronic consults to pulmonary for veterans with Lung-RADS 4 to determine whether they should undergo additional imaging or be evaluated in the pulmonary clinic.
Evaluating Shared Decision Making
We audio taped and transcribed randomly selected SDM encounters to assess fidelity with the 2016 CMS required discussion elements for counseling about lung cancer, including the benefit of reducing lung cancer mortality; the potential for harms from false alarms, incidental findings, overdiagnosis, and radiation exposure; the need for annual screening; the importance of smoking cessation; and the possibility of undergoing follow-up testing and diagnostic procedures. An investigator coded the transcripts to assess for the presence of each required element and scored the encounter from 0 to 7.
We also surveyed veterans completing SDM, using a convenience sampling strategy to evaluate knowledge, the quality of the SDM process, and decisional conflict. Initially, we sent mailed surveys to subjects to be completed 1 week after the SDM visit. To increase the response rate, we subsequently called patients to complete the surveys by telephone 1 week after the SDM visit.
We used the validated LCS-12 knowledge measure to assess awareness of lung cancer risks, screening eligibility, and the benefits and harms of screening.14 We evaluated the quality of the SDM visit by using the 3-item CollaboRATE scale (Table 1).15
The NPC also took field notes during interviews to help identify additional SDM issues. After each call, the NPC noted her impressions of the veteran’s engagement with SDM and understanding of the screening issues.
Clinical Outcomes
We used the screening dashboard and CPRS to track clinical outcomes, including screening uptake, referrals for tobacco cessation, appropriate (screening or diagnostic) follow-up testing, and cancer diagnoses. We used descriptive statistics to characterize demographic data and survey responses.
Initial Findings
We conducted 105 SDM telephone visits from November 2020 through July 2022 (Table 2).
We surveyed 47 of the veterans completing SDM visits (45%) and received 37 completed surveys (79%). All respondents were male, mean age 61.9 years, 89% White, 38% married/partnered, 70% rural, 65% currently smoking, with a mean 44.8 pack-years smoking history. On average, veterans answered 6.3 (53%) of knowledge questions correctly (Table 3).
Only 1 respondent (3%) correctly answered the multiple-choice question about indications for stopping screening. Two (5%) correctly answered the question on the magnitude of benefit, most overestimated or did not know. Similarly, 23 (62%) overestimated or did not know the predictive value of an abnormal scan. About two-thirds of veterans underestimated or did not know the attributable risk of lung cancer from tobacco, and about four-fifths did not know the mortality rank of lung cancer. Among the 37 respondents, 31 (84%) indicated not having any decisional conflict as defined by a score of 4 on the SURE scale.
Implementing SDM
The NPC’s field notes indicated that many veterans did not perceive any need to discuss the screening decision and believed that their PCP had referred them just for screening. However, they reported having cursory discussions with their PCP, being told that only their history of heavy tobacco use meant they should be screened. For veterans who had not read the decision aid, the NPC attempted to summarize benefits and harms. However, the discussions were often inadequate because the veterans were not interested in receiving information, particularly numerical data, or indicated that they had limited time for the call.
Seventy-two (69%) of the veterans who met with the NPC were currently smoking. Tobacco cessation counseling was offered to 66; 29 were referred to the VA Quit Line, 10 were referred to the tobacco cessation pharmacist, and the NPC contacted the PCPs for 9 patients who wanted prescriptions for nicotine replacement therapy.
After the SDM visit, 91 veterans (87%) agreed to screening. By the end of the study period, 73 veterans (80%) completed testing. Most veterans had Lung-RADS 1 or 2 results, 11 (1%) had a Lung-RADS 3, and 7 (10%) had a Lung-RADS 4. All 9 veterans with Lung-RADS 3 results and at least 6 months of follow-up underwent repeat imaging within 4 to 13 months (median, 7). All veterans with a Lung-RADS 4 result were referred to pulmonary. One patient was diagnosed with an early-stage non–small cell lung cancer.
We identified several problems with LDCT coding. Radiologists did not consistently use Lung-RADS when interpreting screening LDCTs; some used the Fleischner lung nodule criteria.18 We also found discordant readings for abnormal LDCTs, where the assigned Lung-RADS score was not consistent with the nodule description in the radiology report.
Discussion
Efforts to implement LCS with a telemedicine SDM intervention were mixed. An NPC-led SDM phone call was successfully incorporated into the clinical workflow. Most veterans identified as being eligible for screening participated in the counseling visit and underwent screening. However, they were often reluctant to engage in SDM, feeling that their clinician had already recommended screening and that there was no need for further discussion. Unfortunately, many veterans had not received or reviewed the decision aid and were not interested in receiving information about benefits and harms. Because we relied on telephone calls, we could not share visual information in real time.
Overall, the surveys indicated that most veterans were very satisfied with the quality of the discussion and reported feeling no decisional conflict. However, based on the NPC’s field notes and audio recordings, we believe that the responses may have reflected earlier discussions with the PCP that reportedly emphasized only the veteran’s eligibility for screening. The fidelity assessments indicated that the NPC consistently addressed the harms and benefits of screening.
Nonetheless, the performance on knowledge measures was uneven. Veterans were generally aware of harms, including false alarms, overdiagnosis, radiation exposure, and incidental findings. They did not, however, appreciate when screening should stop. They also underestimated the risks of developing lung cancer and the portion of that risk attributable to tobacco use, and overestimated the benefits of screening. These results suggest that the veterans, at least those who completed the surveys, may not be making well-informed decisions.
Our findings echo those of other VA investigators in finding knowledge deficits among screened veterans, including being unaware that LDCT was for LCS, believing that screening could prevent cancer, receiving little information about screening harms, and feeling that negative tests meant they were among the “lucky ones” who would avoid harm from continued smoking.19,20
The VA is currently implementing centralized screening models with the Lung Precision Oncology Program and the VA partnership to increase access to lung screening (VA-PALS).5 The centralized model, which readily supports the tracking, monitoring, and reporting needs of a screening program, also has advantages in delivering SDM because counselors have been trained in SDM, are more familiar with LCS evidence and processes, can better incorporate decision tools, and do not face the same time constraints as clinicians.21 However, studies have shown that most patients have already decided to be screened when they show up for the SDM visit.22 In contrast, about one-third of patients in primary care settings who receive decision support chose not to be screened.23,24 We found that 13% of our patients decided against screening after a telephone discussion, suggesting that a virtually conducted SDM visit can meaningfully support decision making. Telemedicine also may reduce health inequities in centralized models arising from patients having limited access to screening centers.
Our results suggest that PCPs referring patients to a centralized program, even for virtual visits, should frame the decision to initiate LCS as SDM, where an informed patient is being supported in making a decision consistent with their values and preferences. Furthermore, engaging patients in SDM should not be construed as endorsing screening. When centralized support is less available, individual clinics may need to provide SDM, perhaps using a nonclinician decision coach if clinicians lack the time to lead the discussions. Decision coaches have been effectively used to increase patients’ knowledge about the benefits and harms of screening.12 Regardless of the program model, PCPs will also be responsible for determining whether patients are healthy enough to undergo invasive diagnostic testing and treatment and ensuring that tobacco use is addressed.
SDM delivered in any setting will be enhanced by ensuring that patients are provided with decision aids before a counseling visit. This will help them better understand the benefits and harms of screening and the need to elicit values. The discussion can then focus on areas of concern or questions raised by reviewing the decision aid. The clinician and patient could also use a decision aid during either a face-to-face or video clinical encounter to facilitate SDM. A Cochrane review has shown that using decision aids for people facing screening decisions increases knowledge, reduces decisional conflict, and effectively elicits values and preferences.25 Providing high-quality decision support is a patient-centered approach that respects a patient’s autonomy and may promote health equity and improve adherence.
We recognized the importance of having a multidisciplinary team, involving primary care, radiology, pulmonary, and nursing, with a shared understanding of the screening processes. These are essential features for a high-quality screening program where eligible veterans are readily identified and receive prompt and appropriate follow-up. Radiologists need to use Lung-RADS categories consistently and appropriately when reading LDCTs. This may require ongoing educational efforts, particularly given the new CMS guidelines accepting nonsubspecialist chest readers.7 Additionally, fellows and board-eligible residents may interpret images in academic settings and at VA facilities. The program needs to work closely with the pulmonary service to ensure that Lung-RADS 4 patients are promptly assessed. Radiologists and pulmonologists should calibrate the application of Lung-RADS categories to pulmonary nodules through jointly participating in meetings to review selected cases.
Challenges and Limitations
We faced some notable implementation challenges. The COVID-19 pandemic was extremely disruptive to LCS as it was to all health care. In addition, screening workflow processes were hampered by a lack of clinical reminders, which ideally would trigger for clinicians based on the tobacco history. The absence of this reminder meant that numerous patients were found to be ineligible for screening. We have a long-standing lung nodule clinic, and clinicians were confused about whether to order a surveillance imaging for an incidental nodule or a screening LDCT.
The radiology service was able to update order sets in CPRS to help guide clinicians in distinguishing indications and prerequisites for enrolling in LCS. This helped reduce the number of inappropriate orders and crossover orders between the VISN nodule tracking program and the LCS program.
Our results were preliminary and based on a small sample. We did not survey all veterans who underwent SDM, though the response rate was 79% and patient characteristics were similar to the larger cohort. Our results were potentially subject to selection bias, which could inflate the positive responses about decision quality and decisional conflict. However, the knowledge deficits are likely to be valid and suggest a need to better inform eligible veterans about the benefits and harms of screening. We did not have sufficient follow-up time to determine whether veterans were adherent to annual screenings. We showed that almost all those with abnormal imaging results completed diagnostic evaluations and/or were evaluated by pulmonary. As the program matures, we will be able to track outcomes related to cancer diagnoses and treatment.
Conclusions
A centralized LCS program was able to deliver SDM and enroll veterans in a screening program. While veterans were confident in their decision to screen and felt that they participated in decision making, knowledge testing indicated important deficits. Furthermore, we observed that many veterans did not meaningfully engage in SDM. Clinicians will need to frame the decision as patient centered at the time of referral, highlight the role of the NPC and importance of SDM, and be able to provide adequate decision support. The SDM visits can be enhanced by ensuring that veterans are able to review decision aids. Telemedicine is an acceptable and effective approach for supporting screening discussions, particularly for rural veterans.26
Acknowledgments
The authors thank the following individuals for their contributions to the study: John Paul Hornbeck, program support specialist; Kelly Miell, PhD; Bradley Mecham, PhD; Christopher C. Richards, MA; Bailey Noble, NP; Rebecca Barnhart, program analyst.
Lung cancer is the second most frequently diagnosed cancer among US veterans and the leading cause of cancer death.1 Clinical trials have shown that annual screening of high-risk persons with low-dose computed tomography (LDCT) can reduce the risk of dying of lung cancer.2 In 2011, the National Lung Screening Trial (NLST) reported that over a 3-year period, annual LDCT screening reduced the risk of dying of lung cancer by 20% compared with chest radiograph screening.3 Lung cancer screening (LCS), however, was associated with harms, including false-positive results, complications from invasive diagnostic procedures, incidental findings, overdiagnosis, and radiation exposure.
The US Preventive Services Task Force (USPSTF) began recommending annual screening of high-risk persons after publication of the NLST results.4 The Veterans Health Administration (VHA) recommended implementing LCS in 2017.5 Guidelines, however, have consistently highlighted the complexity of the decision and the importance of engaging patients in thorough discussions about the potential benefits and harms of screening (shared decision making [SDM]). The Centers for Medicare and Medicaid Services (CMS) has issued coverage determinations mandating that eligible patients undergo a counseling visit that uses a decision aid to support SDM for LCS and addresses tobacco use.6,7 However, primary care practitioners (PCPs) face many challenges in delivering SDM, including a lack of awareness of clinical trial results and screening guidelines, competing clinical demands, being untrained in SDM, and not having educational resources.8 Patients in rural locations face travel burdens in attending counseling visits.9
We conducted a pilot study to address concerns with delivering SDM for LCS to veterans. We implemented a centralized screening model in which veterans were referred by clinicians to a trained decision coach who conducted telephone visits to discuss the initial LCS decision, addressed tobacco cessation, and placed LDCT orders. We evaluated the outcomes of this telemedicine visit by using decision quality metrics and tracking LCS uptake, referrals for tobacco cessation, and clinical outcomes. The University of Iowa Institutional Review Board considered this study to be a quality improvement project and waived informed consent and HIPAA (Health Insurance Portability and Accountability Act) authorization requirements.
Implementation
We implemented the LCS program at the Iowa City Veterans Affairs Health Care System (ICVAHCS), which has both resident and staff clinicians, and 2 community-based outpatient clinics (Coralville, Cedar Rapids) with staff clinicians. The pilot study, conducted from November 2020 through July 2022, was led by a multidisciplinary team that included a nurse, primary care physician, pulmonologist, and radiologist. The team conducted online presentations to educate PCPs about the epidemiology of lung cancer, results of screening trials, LCS guidelines, the rationale for a centralized model of SDM, and the ICVAHCS screening protocols.
Screening Referrals
When the study began in 2020, we used the 2015 USPSTF criteria for annual LCS: individuals aged 55 to 80 years with a 30 pack-year smoking history and current tobacco user or who had quit within 15 years.4 We lowered the starting age to 50 years and the pack-year requirement to 20 after the USPSTF issued updated guidelines in 2021.10 Clinicians were notified about potentially eligible patients through the US Department of Veterans Affairs (VA) Computerized Personal Record System (CPRS) reminders or by the nurse program coordinator (NPC) who reviewed health records of patients with upcoming appointments. If the clinician determined that screening was appropriate, they ordered an LCS consult. The NPC called the veteran to confirm eligibility, mailed a decision aid, and scheduled a telephone visit to conduct SDM. We used the VA decision aid developed for the LCS demonstration project conducted at 8 academic VA medical centers between 2013 and 2017.11
Shared Decision-Making Telephone Visit
The NPC adapted a telephone script developed for a Cancer Prevention and Research Institute of Texas–funded project conducted by 2 coauthors (RJV and LML).12 The NPC asked about receipt/review of the decision aid, described the screening process, and addressed benefits and potential harms of screening. The NPC also offered smoking cessation interventions for veterans who were currently smoking, including referrals to the VA patient aligned care team clinical pharmacist for management of tobacco cessation or to the national VA Quit Line. The encounter ended by assessing the veteran’s understanding of screening issues and eliciting the veteran’s preferences for LDCT and willingness to adhere with the LCS program.
LDCT Imaging
The NPC placed LDCT orders for veterans interested in screening and alerted the referring clinician to sign the order. Veterans who agreed to be screened were placed in an LCS dashboard developed by the Veterans Integrated Services Network (VISN) 23 LCS program that was used as a patient management tool. The dashboard allowed the NPC to track patients, ensuring that veterans were being scheduled for and completing initial and follow-up testing. Radiologists used the Lung-RADS (Lung Imaging Reporting and Data System) to categorize LDCT results (1, normal; 2, benign nodule; 3, probably benign nodule; 4, suspicious nodule).13 Veterans with Lung-RADS 1 or 2 results were scheduled for an annual LDCT (if they remained eligible). Veterans with Lung-RADS 3 results were scheduled for a 6-month follow-up CT. The screening program sent electronic consults to pulmonary for veterans with Lung-RADS 4 to determine whether they should undergo additional imaging or be evaluated in the pulmonary clinic.
Evaluating Shared Decision Making
We audio taped and transcribed randomly selected SDM encounters to assess fidelity with the 2016 CMS required discussion elements for counseling about lung cancer, including the benefit of reducing lung cancer mortality; the potential for harms from false alarms, incidental findings, overdiagnosis, and radiation exposure; the need for annual screening; the importance of smoking cessation; and the possibility of undergoing follow-up testing and diagnostic procedures. An investigator coded the transcripts to assess for the presence of each required element and scored the encounter from 0 to 7.
We also surveyed veterans completing SDM, using a convenience sampling strategy to evaluate knowledge, the quality of the SDM process, and decisional conflict. Initially, we sent mailed surveys to subjects to be completed 1 week after the SDM visit. To increase the response rate, we subsequently called patients to complete the surveys by telephone 1 week after the SDM visit.
We used the validated LCS-12 knowledge measure to assess awareness of lung cancer risks, screening eligibility, and the benefits and harms of screening.14 We evaluated the quality of the SDM visit by using the 3-item CollaboRATE scale (Table 1).15
The NPC also took field notes during interviews to help identify additional SDM issues. After each call, the NPC noted her impressions of the veteran’s engagement with SDM and understanding of the screening issues.
Clinical Outcomes
We used the screening dashboard and CPRS to track clinical outcomes, including screening uptake, referrals for tobacco cessation, appropriate (screening or diagnostic) follow-up testing, and cancer diagnoses. We used descriptive statistics to characterize demographic data and survey responses.
Initial Findings
We conducted 105 SDM telephone visits from November 2020 through July 2022 (Table 2).
We surveyed 47 of the veterans completing SDM visits (45%) and received 37 completed surveys (79%). All respondents were male, mean age 61.9 years, 89% White, 38% married/partnered, 70% rural, 65% currently smoking, with a mean 44.8 pack-years smoking history. On average, veterans answered 6.3 (53%) of knowledge questions correctly (Table 3).
Only 1 respondent (3%) correctly answered the multiple-choice question about indications for stopping screening. Two (5%) correctly answered the question on the magnitude of benefit, most overestimated or did not know. Similarly, 23 (62%) overestimated or did not know the predictive value of an abnormal scan. About two-thirds of veterans underestimated or did not know the attributable risk of lung cancer from tobacco, and about four-fifths did not know the mortality rank of lung cancer. Among the 37 respondents, 31 (84%) indicated not having any decisional conflict as defined by a score of 4 on the SURE scale.
Implementing SDM
The NPC’s field notes indicated that many veterans did not perceive any need to discuss the screening decision and believed that their PCP had referred them just for screening. However, they reported having cursory discussions with their PCP, being told that only their history of heavy tobacco use meant they should be screened. For veterans who had not read the decision aid, the NPC attempted to summarize benefits and harms. However, the discussions were often inadequate because the veterans were not interested in receiving information, particularly numerical data, or indicated that they had limited time for the call.
Seventy-two (69%) of the veterans who met with the NPC were currently smoking. Tobacco cessation counseling was offered to 66; 29 were referred to the VA Quit Line, 10 were referred to the tobacco cessation pharmacist, and the NPC contacted the PCPs for 9 patients who wanted prescriptions for nicotine replacement therapy.
After the SDM visit, 91 veterans (87%) agreed to screening. By the end of the study period, 73 veterans (80%) completed testing. Most veterans had Lung-RADS 1 or 2 results, 11 (1%) had a Lung-RADS 3, and 7 (10%) had a Lung-RADS 4. All 9 veterans with Lung-RADS 3 results and at least 6 months of follow-up underwent repeat imaging within 4 to 13 months (median, 7). All veterans with a Lung-RADS 4 result were referred to pulmonary. One patient was diagnosed with an early-stage non–small cell lung cancer.
We identified several problems with LDCT coding. Radiologists did not consistently use Lung-RADS when interpreting screening LDCTs; some used the Fleischner lung nodule criteria.18 We also found discordant readings for abnormal LDCTs, where the assigned Lung-RADS score was not consistent with the nodule description in the radiology report.
Discussion
Efforts to implement LCS with a telemedicine SDM intervention were mixed. An NPC-led SDM phone call was successfully incorporated into the clinical workflow. Most veterans identified as being eligible for screening participated in the counseling visit and underwent screening. However, they were often reluctant to engage in SDM, feeling that their clinician had already recommended screening and that there was no need for further discussion. Unfortunately, many veterans had not received or reviewed the decision aid and were not interested in receiving information about benefits and harms. Because we relied on telephone calls, we could not share visual information in real time.
Overall, the surveys indicated that most veterans were very satisfied with the quality of the discussion and reported feeling no decisional conflict. However, based on the NPC’s field notes and audio recordings, we believe that the responses may have reflected earlier discussions with the PCP that reportedly emphasized only the veteran’s eligibility for screening. The fidelity assessments indicated that the NPC consistently addressed the harms and benefits of screening.
Nonetheless, the performance on knowledge measures was uneven. Veterans were generally aware of harms, including false alarms, overdiagnosis, radiation exposure, and incidental findings. They did not, however, appreciate when screening should stop. They also underestimated the risks of developing lung cancer and the portion of that risk attributable to tobacco use, and overestimated the benefits of screening. These results suggest that the veterans, at least those who completed the surveys, may not be making well-informed decisions.
Our findings echo those of other VA investigators in finding knowledge deficits among screened veterans, including being unaware that LDCT was for LCS, believing that screening could prevent cancer, receiving little information about screening harms, and feeling that negative tests meant they were among the “lucky ones” who would avoid harm from continued smoking.19,20
The VA is currently implementing centralized screening models with the Lung Precision Oncology Program and the VA partnership to increase access to lung screening (VA-PALS).5 The centralized model, which readily supports the tracking, monitoring, and reporting needs of a screening program, also has advantages in delivering SDM because counselors have been trained in SDM, are more familiar with LCS evidence and processes, can better incorporate decision tools, and do not face the same time constraints as clinicians.21 However, studies have shown that most patients have already decided to be screened when they show up for the SDM visit.22 In contrast, about one-third of patients in primary care settings who receive decision support chose not to be screened.23,24 We found that 13% of our patients decided against screening after a telephone discussion, suggesting that a virtually conducted SDM visit can meaningfully support decision making. Telemedicine also may reduce health inequities in centralized models arising from patients having limited access to screening centers.
Our results suggest that PCPs referring patients to a centralized program, even for virtual visits, should frame the decision to initiate LCS as SDM, where an informed patient is being supported in making a decision consistent with their values and preferences. Furthermore, engaging patients in SDM should not be construed as endorsing screening. When centralized support is less available, individual clinics may need to provide SDM, perhaps using a nonclinician decision coach if clinicians lack the time to lead the discussions. Decision coaches have been effectively used to increase patients’ knowledge about the benefits and harms of screening.12 Regardless of the program model, PCPs will also be responsible for determining whether patients are healthy enough to undergo invasive diagnostic testing and treatment and ensuring that tobacco use is addressed.
SDM delivered in any setting will be enhanced by ensuring that patients are provided with decision aids before a counseling visit. This will help them better understand the benefits and harms of screening and the need to elicit values. The discussion can then focus on areas of concern or questions raised by reviewing the decision aid. The clinician and patient could also use a decision aid during either a face-to-face or video clinical encounter to facilitate SDM. A Cochrane review has shown that using decision aids for people facing screening decisions increases knowledge, reduces decisional conflict, and effectively elicits values and preferences.25 Providing high-quality decision support is a patient-centered approach that respects a patient’s autonomy and may promote health equity and improve adherence.
We recognized the importance of having a multidisciplinary team, involving primary care, radiology, pulmonary, and nursing, with a shared understanding of the screening processes. These are essential features for a high-quality screening program where eligible veterans are readily identified and receive prompt and appropriate follow-up. Radiologists need to use Lung-RADS categories consistently and appropriately when reading LDCTs. This may require ongoing educational efforts, particularly given the new CMS guidelines accepting nonsubspecialist chest readers.7 Additionally, fellows and board-eligible residents may interpret images in academic settings and at VA facilities. The program needs to work closely with the pulmonary service to ensure that Lung-RADS 4 patients are promptly assessed. Radiologists and pulmonologists should calibrate the application of Lung-RADS categories to pulmonary nodules through jointly participating in meetings to review selected cases.
Challenges and Limitations
We faced some notable implementation challenges. The COVID-19 pandemic was extremely disruptive to LCS as it was to all health care. In addition, screening workflow processes were hampered by a lack of clinical reminders, which ideally would trigger for clinicians based on the tobacco history. The absence of this reminder meant that numerous patients were found to be ineligible for screening. We have a long-standing lung nodule clinic, and clinicians were confused about whether to order a surveillance imaging for an incidental nodule or a screening LDCT.
The radiology service was able to update order sets in CPRS to help guide clinicians in distinguishing indications and prerequisites for enrolling in LCS. This helped reduce the number of inappropriate orders and crossover orders between the VISN nodule tracking program and the LCS program.
Our results were preliminary and based on a small sample. We did not survey all veterans who underwent SDM, though the response rate was 79% and patient characteristics were similar to the larger cohort. Our results were potentially subject to selection bias, which could inflate the positive responses about decision quality and decisional conflict. However, the knowledge deficits are likely to be valid and suggest a need to better inform eligible veterans about the benefits and harms of screening. We did not have sufficient follow-up time to determine whether veterans were adherent to annual screenings. We showed that almost all those with abnormal imaging results completed diagnostic evaluations and/or were evaluated by pulmonary. As the program matures, we will be able to track outcomes related to cancer diagnoses and treatment.
Conclusions
A centralized LCS program was able to deliver SDM and enroll veterans in a screening program. While veterans were confident in their decision to screen and felt that they participated in decision making, knowledge testing indicated important deficits. Furthermore, we observed that many veterans did not meaningfully engage in SDM. Clinicians will need to frame the decision as patient centered at the time of referral, highlight the role of the NPC and importance of SDM, and be able to provide adequate decision support. The SDM visits can be enhanced by ensuring that veterans are able to review decision aids. Telemedicine is an acceptable and effective approach for supporting screening discussions, particularly for rural veterans.26
Acknowledgments
The authors thank the following individuals for their contributions to the study: John Paul Hornbeck, program support specialist; Kelly Miell, PhD; Bradley Mecham, PhD; Christopher C. Richards, MA; Bailey Noble, NP; Rebecca Barnhart, program analyst.
1. Zullig LL, Jackson GL, Dorn RA, et al. Cancer incidence among patients of the U.S. Veterans Affairs Health Care System. Mil Med. 2012;177(6):693-701. doi:10.7205/milmed-d-11-00434
2. Hoffman RM, Atallah RP, Struble RD, Badgett RG. Lung cancer screening with low-dose CT: a meta-analysis. J Gen Intern Med. 2020;35(10):3015-3025. doi:10.1007/s11606-020-05951-7
3. National Lung Screening Trial Research Team, Aberle DR, Adams AM, et al. Reduced lung-cancer mortality with low-dose computed tomographic screening. N Engl J Med. 2011;365(5):395-409. doi:10.1056/NEJMoa1102873
4. Moyer VA, US Preventive Services Task Force. Screening for lung cancer: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2014;160(5):330-338. doi:10.7326/M13-2771
5. Maurice NM, Tanner NT. Lung cancer screening at the VA: past, present and future. Semin Oncol. 2022;S0093-7754(22)00041-0. doi:10.1053/j.seminoncol.2022.06.001
6. Centers for Medicare & Medicaid Services. Screening for lung cancer with low dose computed tomography (LDCT) (CAG-00439N). Published 2015. Accessed July 10, 2023. http://www.cms.gov/medicare-coverage-database/details/nca-decision-memo.aspx?NCAId=274
7. Centers for Medicare & Medicaid Services. Screening for lung cancer with low dose computed tomography (LDCT) (CAG-00439R). Published 2022. Accessed July 10, 2023. https://www.cms.gov/medicare-coverage-database/view/ncacal-decision-memo.aspx?proposed=N&ncaid=304
8. National Academies of Sciences, Engineering, and Medicine; Health and Medicine Division; Board on Health Care Services; National Cancer Policy Forum. Implementation of Lung Cancer Screening: Proceedings of a Workshop. The National Academies Press; November 17, 2016. doi:10.172216/23680
9. Bernstein E, Bade BC, Akgün KM, Rose MG, Cain HC. Barriers and facilitators to lung cancer screening and follow-up. Semin Oncol. 2022;S0093-7754(22)00058-6. doi:10.1053/j.seminoncol.2022.07.004
10. US Preventive Services Task Force, Krist AH, Davidson KW, et al. Screening for lung cancer: US Preventive Services Task Force recommendation statement. JAMA. 2021;325(10):962-970. doi:10.1001/jama.2021.1117
11. Kinsinger LS, Atkins D, Provenzale D, Anderson C, Petzel R. Implementation of a new screening recommendation in health care: the Veterans Health Administration’s approach to lung cancer screening. Ann Intern Med. 2014;161(8):597-598. doi:10.7326/M14-1070
12. Lowenstein LM, Godoy MCB, Erasmus JJ, et al. Implementing decision coaching for lung cancer screening in the low-dose computed tomography setting. JCO Oncol Pract. 2020;16(8):e703-e725. doi:10.1200/JOP.19.00453
13. American College of Radiology Committee on Lung-RADS. Lung-RADS assessment categories 2022. Published November 2022. Accessed July 3, 2023. https://www.acr.org/-/media/ACR/Files/RADS/Lung-RADS/Lung-RADS-2022.pdf
14. Lowenstein LM, Richards VF, Leal VB, et al. A brief measure of smokers’ knowledge of lung cancer screening with low-dose computed tomography. Prev Med Rep. 2016;4:351-356. doi:10.1016/j.pmedr.2016.07.008
15. Elwyn G, Barr PJ, Grande SW, Thompson R, Walsh T, Ozanne EM. Developing CollaboRATE: a fast and frugal patient-reported measure of shared decision making in clinical encounters. Patient Educ Couns. 2013;93(1):102-107. doi:10.1016/j.pec.2013.05.009
16. Barr PJ, Thompson R, Walsh T, Grande SW, Ozanne EM, Elwyn G. The psychometric properties of CollaboRATE: a fast and frugal patient-reported measure of the shared decision-making process. J Med Internet Res. 2014;16(1):e2. doi:10.2196/jmir.3085
17. Légaré F, Kearing S, Clay K, et al. Are you SURE?: Assessing patient decisional conflict with a 4-item screening test. Can Fam Physician. 2010;56(8):e308-e314.
18. MacMahon H, Naidich DP, Goo JM, et al. Guidelines for management of incidental pulmonary nodules detected on CT images: from the Fleischner Society 2017. Radiology. 2017;284(1):228-243. doi:10.1148/radiol.2017161659
19. Wiener RS, Koppelman E, Bolton R, et al. Patient and clinician perspectives on shared decision-making in early adopting lung cancer screening programs: a qualitative study. J Gen Intern Med. 2018;33(7):1035-1042. doi:10.1007/s11606-018-4350-9
20. Zeliadt SB, Heffner JL, Sayre G, et al. Attitudes and perceptions about smoking cessation in the context of lung cancer screening. JAMA Intern Med. 2015;175(9):1530-1537. doi:10.1001/jamainternmed.2015.3558
21. Mazzone PJ, White CS, Kazerooni EA, Smith RA, Thomson CC. Proposed quality metrics for lung cancer screening programs: a National Lung Cancer Roundtable Project. Chest. 2021;160(1):368-378. doi:10.1016/j.chest.2021.01.063
22. Mazzone PJ, Tenenbaum A, Seeley M, et al. Impact of a lung cancer screening counseling and shared decision-making visit. Chest. 2017;151(3):572-578. doi:10.1016/j.chest.2016.10.027
23. Reuland DS, Cubillos L, Brenner AT, Harris RP, Minish B, Pignone MP. A pre-post study testing a lung cancer screening decision aid in primary care. BMC Med Inform Decis Mak. 2018;18(1):5. doi:10.1186/s12911-018-0582-1
24. Dharod A, Bellinger C, Foley K, Case LD, Miller D. The reach and feasibility of an interactive lung cancer screening decision aid delivered by patient portal. Appl Clin Inform. 2019;10(1):19-27. doi:10.1055/s-0038-1676807
25. Stacey D, Légaré F, Lewis K, et al. Decision aids for people facing health treatment or screening decisions. Cochrane Database Syst Rev. 2017;4:CD001431. doi:10.1002/14651858.CD001431.pub5
26. Tanner NT, Banas E, Yeager D, Dai L, Hughes Halbert C, Silvestri GA. In-person and telephonic shared decision-making visits for people considering lung cancer screening: an assessment of decision quality. Chest. 2019;155(1):236-238. doi:10.1016/j.chest.2018.07.046
1. Zullig LL, Jackson GL, Dorn RA, et al. Cancer incidence among patients of the U.S. Veterans Affairs Health Care System. Mil Med. 2012;177(6):693-701. doi:10.7205/milmed-d-11-00434
2. Hoffman RM, Atallah RP, Struble RD, Badgett RG. Lung cancer screening with low-dose CT: a meta-analysis. J Gen Intern Med. 2020;35(10):3015-3025. doi:10.1007/s11606-020-05951-7
3. National Lung Screening Trial Research Team, Aberle DR, Adams AM, et al. Reduced lung-cancer mortality with low-dose computed tomographic screening. N Engl J Med. 2011;365(5):395-409. doi:10.1056/NEJMoa1102873
4. Moyer VA, US Preventive Services Task Force. Screening for lung cancer: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2014;160(5):330-338. doi:10.7326/M13-2771
5. Maurice NM, Tanner NT. Lung cancer screening at the VA: past, present and future. Semin Oncol. 2022;S0093-7754(22)00041-0. doi:10.1053/j.seminoncol.2022.06.001
6. Centers for Medicare & Medicaid Services. Screening for lung cancer with low dose computed tomography (LDCT) (CAG-00439N). Published 2015. Accessed July 10, 2023. http://www.cms.gov/medicare-coverage-database/details/nca-decision-memo.aspx?NCAId=274
7. Centers for Medicare & Medicaid Services. Screening for lung cancer with low dose computed tomography (LDCT) (CAG-00439R). Published 2022. Accessed July 10, 2023. https://www.cms.gov/medicare-coverage-database/view/ncacal-decision-memo.aspx?proposed=N&ncaid=304
8. National Academies of Sciences, Engineering, and Medicine; Health and Medicine Division; Board on Health Care Services; National Cancer Policy Forum. Implementation of Lung Cancer Screening: Proceedings of a Workshop. The National Academies Press; November 17, 2016. doi:10.172216/23680
9. Bernstein E, Bade BC, Akgün KM, Rose MG, Cain HC. Barriers and facilitators to lung cancer screening and follow-up. Semin Oncol. 2022;S0093-7754(22)00058-6. doi:10.1053/j.seminoncol.2022.07.004
10. US Preventive Services Task Force, Krist AH, Davidson KW, et al. Screening for lung cancer: US Preventive Services Task Force recommendation statement. JAMA. 2021;325(10):962-970. doi:10.1001/jama.2021.1117
11. Kinsinger LS, Atkins D, Provenzale D, Anderson C, Petzel R. Implementation of a new screening recommendation in health care: the Veterans Health Administration’s approach to lung cancer screening. Ann Intern Med. 2014;161(8):597-598. doi:10.7326/M14-1070
12. Lowenstein LM, Godoy MCB, Erasmus JJ, et al. Implementing decision coaching for lung cancer screening in the low-dose computed tomography setting. JCO Oncol Pract. 2020;16(8):e703-e725. doi:10.1200/JOP.19.00453
13. American College of Radiology Committee on Lung-RADS. Lung-RADS assessment categories 2022. Published November 2022. Accessed July 3, 2023. https://www.acr.org/-/media/ACR/Files/RADS/Lung-RADS/Lung-RADS-2022.pdf
14. Lowenstein LM, Richards VF, Leal VB, et al. A brief measure of smokers’ knowledge of lung cancer screening with low-dose computed tomography. Prev Med Rep. 2016;4:351-356. doi:10.1016/j.pmedr.2016.07.008
15. Elwyn G, Barr PJ, Grande SW, Thompson R, Walsh T, Ozanne EM. Developing CollaboRATE: a fast and frugal patient-reported measure of shared decision making in clinical encounters. Patient Educ Couns. 2013;93(1):102-107. doi:10.1016/j.pec.2013.05.009
16. Barr PJ, Thompson R, Walsh T, Grande SW, Ozanne EM, Elwyn G. The psychometric properties of CollaboRATE: a fast and frugal patient-reported measure of the shared decision-making process. J Med Internet Res. 2014;16(1):e2. doi:10.2196/jmir.3085
17. Légaré F, Kearing S, Clay K, et al. Are you SURE?: Assessing patient decisional conflict with a 4-item screening test. Can Fam Physician. 2010;56(8):e308-e314.
18. MacMahon H, Naidich DP, Goo JM, et al. Guidelines for management of incidental pulmonary nodules detected on CT images: from the Fleischner Society 2017. Radiology. 2017;284(1):228-243. doi:10.1148/radiol.2017161659
19. Wiener RS, Koppelman E, Bolton R, et al. Patient and clinician perspectives on shared decision-making in early adopting lung cancer screening programs: a qualitative study. J Gen Intern Med. 2018;33(7):1035-1042. doi:10.1007/s11606-018-4350-9
20. Zeliadt SB, Heffner JL, Sayre G, et al. Attitudes and perceptions about smoking cessation in the context of lung cancer screening. JAMA Intern Med. 2015;175(9):1530-1537. doi:10.1001/jamainternmed.2015.3558
21. Mazzone PJ, White CS, Kazerooni EA, Smith RA, Thomson CC. Proposed quality metrics for lung cancer screening programs: a National Lung Cancer Roundtable Project. Chest. 2021;160(1):368-378. doi:10.1016/j.chest.2021.01.063
22. Mazzone PJ, Tenenbaum A, Seeley M, et al. Impact of a lung cancer screening counseling and shared decision-making visit. Chest. 2017;151(3):572-578. doi:10.1016/j.chest.2016.10.027
23. Reuland DS, Cubillos L, Brenner AT, Harris RP, Minish B, Pignone MP. A pre-post study testing a lung cancer screening decision aid in primary care. BMC Med Inform Decis Mak. 2018;18(1):5. doi:10.1186/s12911-018-0582-1
24. Dharod A, Bellinger C, Foley K, Case LD, Miller D. The reach and feasibility of an interactive lung cancer screening decision aid delivered by patient portal. Appl Clin Inform. 2019;10(1):19-27. doi:10.1055/s-0038-1676807
25. Stacey D, Légaré F, Lewis K, et al. Decision aids for people facing health treatment or screening decisions. Cochrane Database Syst Rev. 2017;4:CD001431. doi:10.1002/14651858.CD001431.pub5
26. Tanner NT, Banas E, Yeager D, Dai L, Hughes Halbert C, Silvestri GA. In-person and telephonic shared decision-making visits for people considering lung cancer screening: an assessment of decision quality. Chest. 2019;155(1):236-238. doi:10.1016/j.chest.2018.07.046
Genetic profiles affect smokers’ lung cancer risk
conducted by specialists from the Cancer Center at the University of Navarra Clinic (CUN). The results were presented at the annual meeting of the American Society for Clinical Oncology.
Ana Patiño García, PhD, director of the genomic medicine unit at the CUN and a coordinator of the research, explained in an interview the main reason why this study was conducted. “This study came straight out of the oncology clinic, where we are constantly encountering patients with lung cancer who have never smoked or who have smoked very little, while we also all know people who have smoked a lot throughout their lifetime and have never developed cancer. This observation has led us to ask whether there are genetic factors that increase or decrease the risk of cancer and protect people against this disease.”
José Luis Pérez Gracia, MD, PhD, oncologist, coordinator of the oncology trials department at the CUN and another of the individuals responsible for this research, said: “This is the first study to validate genetic factors associated with people who appear to be resistant to developing tobacco-related lung cancer or who, on the other hand, are at high risk of developing this disease.”
Pioneering approach
Earlier evidence showed that some smokers develop cancer, and others don’t. “This is a very well-known fact, since everyone knows about some elderly person who has been a heavy smoker and has never developed lung cancer,” said Dr. Pérez. “Unfortunately, we oncologists encounter young smokers who have been diagnosed with this disease. However, despite the importance of understanding the causes behind these phenotypes, it is a question that has never been studied from a genetic standpoint.”
The study was conducted using DNA from 133 heavy smokers who had not developed lung cancer at a mean age of 80 years, and from another 116 heavy smokers who had developed this type of cancer at a mean age of 50 years. This DNA was sequenced using next-generation techniques, and the results were analyzed using bioinformatics and artificial intelligence systems in collaboration with the University of Navarra Applied Medical Research Center and the University of Navarra School of Engineering.
When asked how this methodology could be applied to support other research conducted along these lines, Dr. Patiño said, “The most novel thing about this research is actually its approach. It’s based on groups at the extremes, defined by the patient’s age at the time of developing lung cancer and how much they had smoked. This type of comparative design is called extreme phenotypes, and its main distinguishing characteristic – which is also its most limiting characteristic – is choosing cases and controls well. Obviously, with today’s next-generation sequencing technologies, we achieve a quantity and quality of data that would have been unattainable in years gone by.”
Speaking to the role played by bioinformatics and artificial intelligence in this research, Dr. Patiño explained that they are fairly new techniques. “In fact, these technologies could be thought of as spearheading a lot of the biomedical research being done today. They’ve also somewhat set the stage for the paradigm shift where the investigator asks the data a question, and in the case of artificial intelligence, it’s the data that answer.”
Pinpointing genetic differences
In his analysis of the most noteworthy data and conclusions from this research, Dr. Pérez noted, “The most significant thing we’ve seen is that both populations have genetic differences. This suggests that our hypothesis is correct. Of course, more studies including a larger number of individuals will be needed to confirm these findings. For the first time, our work has laid the foundation for developing this line of research.”
“Many genetic variants that we have identified as differentials in cases and controls are found in genes relevant to the immune system (HLA system), in genes related to functional pathways that are often altered in tumor development, and in structural proteins and in genes related to cell mobility,” emphasized Dr. Patiño.
Many of the genetic characteristics that were discovered are located in genes with functions related to cancer development, such as immune response, repair of genetic material, regulation of inflammation, etc. This finding is highly significant, said Dr. Pérez. “However, we must remember that these phenotypes may be attributable to multiple causes, not just one cause.”
Furthermore, the specialist explained the next steps to be taken in the context of the line opened up by this research. “First, we must expand these studies, including more individuals with, if possible, even more extreme phenotypes: more smokers who are older and younger, respectively. Once the statistical evidence is stronger, we must also confirm that the alterations observed in lab-based studies truly impact gene function.”
Earlier diagnosis
The clinician also discussed the potential ways that the conclusions of this study could be applied to clinical practice now and in the future, and how the conclusions could benefit these patients. “The results of our line of research may help in early identification of those individuals at high risk of developing lung cancer if they smoke, so that they could be included in prevention programs to keep them from smoking or to help them stop smoking,” said Dr. Pérez. “It would also allow for early diagnosis of cancer at a time when there is a much higher chance of curing it.
“However, the most important thing is that our study may allow us to better understand the mechanisms by which cancer arises and especially why some people do not develop it. This [understanding] could lead to new diagnostic techniques and new treatments for this disease. The techniques needed to develop this line of research (bioinformatic mass sequencing and artificial intelligence) are available and becoming more reliable and more accessible every day. So, we believe our strategy is very realistic,” he added.
Although the line of research opened up by this study depicts a new scenario, the specialists still must face several challenges to discover why some smokers are more likely than others to develop lung cancer.
“There are many lines of research in this regard,” said Dr. Pérez. “But to name a few, I would draw attention to the need to increase the number of cases and controls to improve the comparison, study patients with other tumors related to tobacco use, ask new questions using the data we have already collected, and apply other genomic techniques that would allow us to perform additional studies of genetic variants that have not yet been studied. And, of course, we need to use functional studies to expand our understanding of the function and activity of the genes that have already been identified.”
Dr. Patiño and Dr. Pérez declared that they have no relevant financial conflicts of interest.
This article was translated from the Medscape Spanish Edition. A version appeared on Medscape.com.
conducted by specialists from the Cancer Center at the University of Navarra Clinic (CUN). The results were presented at the annual meeting of the American Society for Clinical Oncology.
Ana Patiño García, PhD, director of the genomic medicine unit at the CUN and a coordinator of the research, explained in an interview the main reason why this study was conducted. “This study came straight out of the oncology clinic, where we are constantly encountering patients with lung cancer who have never smoked or who have smoked very little, while we also all know people who have smoked a lot throughout their lifetime and have never developed cancer. This observation has led us to ask whether there are genetic factors that increase or decrease the risk of cancer and protect people against this disease.”
José Luis Pérez Gracia, MD, PhD, oncologist, coordinator of the oncology trials department at the CUN and another of the individuals responsible for this research, said: “This is the first study to validate genetic factors associated with people who appear to be resistant to developing tobacco-related lung cancer or who, on the other hand, are at high risk of developing this disease.”
Pioneering approach
Earlier evidence showed that some smokers develop cancer, and others don’t. “This is a very well-known fact, since everyone knows about some elderly person who has been a heavy smoker and has never developed lung cancer,” said Dr. Pérez. “Unfortunately, we oncologists encounter young smokers who have been diagnosed with this disease. However, despite the importance of understanding the causes behind these phenotypes, it is a question that has never been studied from a genetic standpoint.”
The study was conducted using DNA from 133 heavy smokers who had not developed lung cancer at a mean age of 80 years, and from another 116 heavy smokers who had developed this type of cancer at a mean age of 50 years. This DNA was sequenced using next-generation techniques, and the results were analyzed using bioinformatics and artificial intelligence systems in collaboration with the University of Navarra Applied Medical Research Center and the University of Navarra School of Engineering.
When asked how this methodology could be applied to support other research conducted along these lines, Dr. Patiño said, “The most novel thing about this research is actually its approach. It’s based on groups at the extremes, defined by the patient’s age at the time of developing lung cancer and how much they had smoked. This type of comparative design is called extreme phenotypes, and its main distinguishing characteristic – which is also its most limiting characteristic – is choosing cases and controls well. Obviously, with today’s next-generation sequencing technologies, we achieve a quantity and quality of data that would have been unattainable in years gone by.”
Speaking to the role played by bioinformatics and artificial intelligence in this research, Dr. Patiño explained that they are fairly new techniques. “In fact, these technologies could be thought of as spearheading a lot of the biomedical research being done today. They’ve also somewhat set the stage for the paradigm shift where the investigator asks the data a question, and in the case of artificial intelligence, it’s the data that answer.”
Pinpointing genetic differences
In his analysis of the most noteworthy data and conclusions from this research, Dr. Pérez noted, “The most significant thing we’ve seen is that both populations have genetic differences. This suggests that our hypothesis is correct. Of course, more studies including a larger number of individuals will be needed to confirm these findings. For the first time, our work has laid the foundation for developing this line of research.”
“Many genetic variants that we have identified as differentials in cases and controls are found in genes relevant to the immune system (HLA system), in genes related to functional pathways that are often altered in tumor development, and in structural proteins and in genes related to cell mobility,” emphasized Dr. Patiño.
Many of the genetic characteristics that were discovered are located in genes with functions related to cancer development, such as immune response, repair of genetic material, regulation of inflammation, etc. This finding is highly significant, said Dr. Pérez. “However, we must remember that these phenotypes may be attributable to multiple causes, not just one cause.”
Furthermore, the specialist explained the next steps to be taken in the context of the line opened up by this research. “First, we must expand these studies, including more individuals with, if possible, even more extreme phenotypes: more smokers who are older and younger, respectively. Once the statistical evidence is stronger, we must also confirm that the alterations observed in lab-based studies truly impact gene function.”
Earlier diagnosis
The clinician also discussed the potential ways that the conclusions of this study could be applied to clinical practice now and in the future, and how the conclusions could benefit these patients. “The results of our line of research may help in early identification of those individuals at high risk of developing lung cancer if they smoke, so that they could be included in prevention programs to keep them from smoking or to help them stop smoking,” said Dr. Pérez. “It would also allow for early diagnosis of cancer at a time when there is a much higher chance of curing it.
“However, the most important thing is that our study may allow us to better understand the mechanisms by which cancer arises and especially why some people do not develop it. This [understanding] could lead to new diagnostic techniques and new treatments for this disease. The techniques needed to develop this line of research (bioinformatic mass sequencing and artificial intelligence) are available and becoming more reliable and more accessible every day. So, we believe our strategy is very realistic,” he added.
Although the line of research opened up by this study depicts a new scenario, the specialists still must face several challenges to discover why some smokers are more likely than others to develop lung cancer.
“There are many lines of research in this regard,” said Dr. Pérez. “But to name a few, I would draw attention to the need to increase the number of cases and controls to improve the comparison, study patients with other tumors related to tobacco use, ask new questions using the data we have already collected, and apply other genomic techniques that would allow us to perform additional studies of genetic variants that have not yet been studied. And, of course, we need to use functional studies to expand our understanding of the function and activity of the genes that have already been identified.”
Dr. Patiño and Dr. Pérez declared that they have no relevant financial conflicts of interest.
This article was translated from the Medscape Spanish Edition. A version appeared on Medscape.com.
conducted by specialists from the Cancer Center at the University of Navarra Clinic (CUN). The results were presented at the annual meeting of the American Society for Clinical Oncology.
Ana Patiño García, PhD, director of the genomic medicine unit at the CUN and a coordinator of the research, explained in an interview the main reason why this study was conducted. “This study came straight out of the oncology clinic, where we are constantly encountering patients with lung cancer who have never smoked or who have smoked very little, while we also all know people who have smoked a lot throughout their lifetime and have never developed cancer. This observation has led us to ask whether there are genetic factors that increase or decrease the risk of cancer and protect people against this disease.”
José Luis Pérez Gracia, MD, PhD, oncologist, coordinator of the oncology trials department at the CUN and another of the individuals responsible for this research, said: “This is the first study to validate genetic factors associated with people who appear to be resistant to developing tobacco-related lung cancer or who, on the other hand, are at high risk of developing this disease.”
Pioneering approach
Earlier evidence showed that some smokers develop cancer, and others don’t. “This is a very well-known fact, since everyone knows about some elderly person who has been a heavy smoker and has never developed lung cancer,” said Dr. Pérez. “Unfortunately, we oncologists encounter young smokers who have been diagnosed with this disease. However, despite the importance of understanding the causes behind these phenotypes, it is a question that has never been studied from a genetic standpoint.”
The study was conducted using DNA from 133 heavy smokers who had not developed lung cancer at a mean age of 80 years, and from another 116 heavy smokers who had developed this type of cancer at a mean age of 50 years. This DNA was sequenced using next-generation techniques, and the results were analyzed using bioinformatics and artificial intelligence systems in collaboration with the University of Navarra Applied Medical Research Center and the University of Navarra School of Engineering.
When asked how this methodology could be applied to support other research conducted along these lines, Dr. Patiño said, “The most novel thing about this research is actually its approach. It’s based on groups at the extremes, defined by the patient’s age at the time of developing lung cancer and how much they had smoked. This type of comparative design is called extreme phenotypes, and its main distinguishing characteristic – which is also its most limiting characteristic – is choosing cases and controls well. Obviously, with today’s next-generation sequencing technologies, we achieve a quantity and quality of data that would have been unattainable in years gone by.”
Speaking to the role played by bioinformatics and artificial intelligence in this research, Dr. Patiño explained that they are fairly new techniques. “In fact, these technologies could be thought of as spearheading a lot of the biomedical research being done today. They’ve also somewhat set the stage for the paradigm shift where the investigator asks the data a question, and in the case of artificial intelligence, it’s the data that answer.”
Pinpointing genetic differences
In his analysis of the most noteworthy data and conclusions from this research, Dr. Pérez noted, “The most significant thing we’ve seen is that both populations have genetic differences. This suggests that our hypothesis is correct. Of course, more studies including a larger number of individuals will be needed to confirm these findings. For the first time, our work has laid the foundation for developing this line of research.”
“Many genetic variants that we have identified as differentials in cases and controls are found in genes relevant to the immune system (HLA system), in genes related to functional pathways that are often altered in tumor development, and in structural proteins and in genes related to cell mobility,” emphasized Dr. Patiño.
Many of the genetic characteristics that were discovered are located in genes with functions related to cancer development, such as immune response, repair of genetic material, regulation of inflammation, etc. This finding is highly significant, said Dr. Pérez. “However, we must remember that these phenotypes may be attributable to multiple causes, not just one cause.”
Furthermore, the specialist explained the next steps to be taken in the context of the line opened up by this research. “First, we must expand these studies, including more individuals with, if possible, even more extreme phenotypes: more smokers who are older and younger, respectively. Once the statistical evidence is stronger, we must also confirm that the alterations observed in lab-based studies truly impact gene function.”
Earlier diagnosis
The clinician also discussed the potential ways that the conclusions of this study could be applied to clinical practice now and in the future, and how the conclusions could benefit these patients. “The results of our line of research may help in early identification of those individuals at high risk of developing lung cancer if they smoke, so that they could be included in prevention programs to keep them from smoking or to help them stop smoking,” said Dr. Pérez. “It would also allow for early diagnosis of cancer at a time when there is a much higher chance of curing it.
“However, the most important thing is that our study may allow us to better understand the mechanisms by which cancer arises and especially why some people do not develop it. This [understanding] could lead to new diagnostic techniques and new treatments for this disease. The techniques needed to develop this line of research (bioinformatic mass sequencing and artificial intelligence) are available and becoming more reliable and more accessible every day. So, we believe our strategy is very realistic,” he added.
Although the line of research opened up by this study depicts a new scenario, the specialists still must face several challenges to discover why some smokers are more likely than others to develop lung cancer.
“There are many lines of research in this regard,” said Dr. Pérez. “But to name a few, I would draw attention to the need to increase the number of cases and controls to improve the comparison, study patients with other tumors related to tobacco use, ask new questions using the data we have already collected, and apply other genomic techniques that would allow us to perform additional studies of genetic variants that have not yet been studied. And, of course, we need to use functional studies to expand our understanding of the function and activity of the genes that have already been identified.”
Dr. Patiño and Dr. Pérez declared that they have no relevant financial conflicts of interest.
This article was translated from the Medscape Spanish Edition. A version appeared on Medscape.com.
FROM ASCO 2023
How useful are circulating tumor cells for early diagnosis?
Treatment options for patients with cancer that is detected at a late stage are severely limited, which usually leads to an unfavorable prognosis for such patients. Indeed, the options available for patients with metastatic solid cancers are scarcely curative. Therefore, early diagnosis of neoplasia remains a fundamental mainstay for improving outcomes for cancer patients.
Histopathology is the current gold standard for cancer diagnosis. Biopsy is an invasive procedure that provides physicians with further samples to test but that furnishes limited information concerning tumor heterogeneity. Biopsy specimens are usually obtained only when there is clinical evidence of neoplasia, which significantly limits their usefulness in early diagnosis.
Around 20 years ago, it was discovered that the presence of circulating tumor cells (CTC) in patients with metastatic breast cancer who were about to begin a new line of treatment was predictive of overall and progression-free survival. The prognostic value of CTC was independent of the line of treatment (first or second) and was greater than that of the site of metastasis, the type of therapy, and the time to metastasis after complete primary resection. These results support the idea that the presence of CTC could be used to modify the system for staging advanced disease.
Since then,
Liquid vs. tissue
Liquid biopsy is a minimally invasive tool that is easy to use. It is employed to detect cancer, to assess treatment response, or to monitor disease progression. Liquid biopsy produces test material from primary and metastatic (or micrometastatic) sites and provides a more heterogeneous picture of the entire tumor cell population, compared with specimens obtained with tissue biopsy.
Metastasis
The notion that metastatic lesions are formed from cancer cells that have disseminated from advanced primary tumors has been substantially revised following the identification of disseminated tumor cells (DTC) in the bone marrow of patients with early-stage disease. These results have led researchers to no longer view cancer metastasis as a linear cascade of events but rather as a series of concurrent, partially overlapping processes, as metastasizing cells assume new phenotypes while abandoning older behaviors.
The initiation of metastasis is not simply a cell-autonomous event but is heavily influenced by complex tissue microenvironments. Although colonization of distant tissues by DTC is an extremely inefficient process, at times, relatively numerous CTC can be detected in the blood of cancer patients (> 1,000 CTC/mL of blood plasma), whereas the number of clinically detectable metastases is disproportionately low, confirming that tumor cell diffusion can happen at an early stage but usually occurs later on.
Early dissemination
Little is currently known about the preference of cancer subtypes for distinct tissues or about the receptiveness of a tissue as a metastatic site. What endures as one of the most confounding clinical phenomena is that patients may undergo tumor resection and remain apparently disease free for months, years, and even decades, only to experience relapse and be diagnosed with late-stage metastatic disease. This course may be a result of cell seeding from minimal residual disease after resection of the primary tumor or of preexisting clinically undetectable micrometastases. It may also arise from early disseminated cells that remain dormant and resistant to therapy until they suddenly reawaken to initiate proliferation into clinically detectable macrometastases.
Dormant DTC could be the main reason for delayed detection of metastases. It is thought that around 40% of patients with prostate cancer who undergo radical prostatectomy present with biochemical recurrence, suggesting that it is likely that hidden DTC or micrometastases are present at the time of the procedure. The finding is consistent with the detection of DTC many years after tumor resection, suggesting they were released before surgical treatment. Nevertheless, research into tumor cell dormancy is limited, owing to the invasive and technically challenging nature of obtaining DTC samples, which are predominantly taken from the bone marrow.
CTC metastases
Cancer cells can undergo epithelial-to-mesenchymal transition to facilitate their detachment from the primary tumor and intravasation into the blood circulation (step 1). Dissemination of cancer cells from the primary tumor into circulation can involve either single cells or cell clusters containing multiple CTC as well as immune cells and platelets, known as microemboli. CTC that can survive in circulation (step 2) can exit the bloodstream (step 3) and establish metastatic tumors (step 4), or they can enter dormancy and reside in distant organs, such as the bone marrow.
Use in practice
CTC were discovered over a century ago, but only in recent years has technology been sufficiently advanced to study CTC and to assess their usefulness as biomarkers. Recent evidence suggests that not only do the number of CTC increase during sleep and rest phases but also that these CTC are better able to metastasize, compared to those generated during periods of wakefulness or activity.
CTC clusters (microemboli) are defined as groups of two or more CTC. They can consist of CTC alone (homotypic) or can include various stromal cells, such as cancer-associated fibroblasts or platelets and immune cells (heterotypic). CTC clusters (with or without leukocytes) seem to have greater metastatic capacity, compared with individual CTC.
A multitude of characteristics can be measured in CTC, including genetics and epigenetics, as well as protein levels, which might help in understanding many processes involved in the formation of metastases.
Quantitative assessment of CTC could indicate tumor burden in patients with aggressive cancers, as has been seen in patients with primary lung cancer.
Early cancer diagnosis
Early research into CTC didn’t explore their usefulness in diagnosing early-stage tumors because it was thought that CTC were characteristic of advanced-stage disease. This hypothesis was later rejected following evidence of local intravascular invasion of very early cancer cells, even over a period of several hours. This feature may allow CTC to be detected before the clinical diagnosis of cancer.
CTC have been detected in various neoplastic conditions: in breast cancer, seen in 20% of patients with stage I disease, in 26.8% with stage II disease, and 26.7% with stage III disease; in nonmetastatic colorectal cancer, including stage I and II disease; and in prostate cancer, seen in over 50% of patients with localized disease.
The presence of CTC has been proven to be an unfavorable prognostic predictor of overall survival among patients with early-stage non–small cell lung cancer. It distinguishes patients with pancreatic ductal adenocarcinoma from those with noncancerous pancreatic diseases with a sensitivity of 75% and a specificity of 96.3%.
CTC positivity scoring (appropriately defined), combined with serum prostate-specific antigen level, was predictive of a biopsy diagnosis of clinically significant prostate cancer.
All these data support the utility of CTC in early cancer diagnosis. Their link with metastases, and thus with aggressive tumors, gives them an advantage over other (noninvasive or minimally invasive) biomarkers in the early identification of invasive tumors for therapeutic intervention with better cure rates.
This article was translated from Univadis Italy. A version appeared on Medscape.com.
Treatment options for patients with cancer that is detected at a late stage are severely limited, which usually leads to an unfavorable prognosis for such patients. Indeed, the options available for patients with metastatic solid cancers are scarcely curative. Therefore, early diagnosis of neoplasia remains a fundamental mainstay for improving outcomes for cancer patients.
Histopathology is the current gold standard for cancer diagnosis. Biopsy is an invasive procedure that provides physicians with further samples to test but that furnishes limited information concerning tumor heterogeneity. Biopsy specimens are usually obtained only when there is clinical evidence of neoplasia, which significantly limits their usefulness in early diagnosis.
Around 20 years ago, it was discovered that the presence of circulating tumor cells (CTC) in patients with metastatic breast cancer who were about to begin a new line of treatment was predictive of overall and progression-free survival. The prognostic value of CTC was independent of the line of treatment (first or second) and was greater than that of the site of metastasis, the type of therapy, and the time to metastasis after complete primary resection. These results support the idea that the presence of CTC could be used to modify the system for staging advanced disease.
Since then,
Liquid vs. tissue
Liquid biopsy is a minimally invasive tool that is easy to use. It is employed to detect cancer, to assess treatment response, or to monitor disease progression. Liquid biopsy produces test material from primary and metastatic (or micrometastatic) sites and provides a more heterogeneous picture of the entire tumor cell population, compared with specimens obtained with tissue biopsy.
Metastasis
The notion that metastatic lesions are formed from cancer cells that have disseminated from advanced primary tumors has been substantially revised following the identification of disseminated tumor cells (DTC) in the bone marrow of patients with early-stage disease. These results have led researchers to no longer view cancer metastasis as a linear cascade of events but rather as a series of concurrent, partially overlapping processes, as metastasizing cells assume new phenotypes while abandoning older behaviors.
The initiation of metastasis is not simply a cell-autonomous event but is heavily influenced by complex tissue microenvironments. Although colonization of distant tissues by DTC is an extremely inefficient process, at times, relatively numerous CTC can be detected in the blood of cancer patients (> 1,000 CTC/mL of blood plasma), whereas the number of clinically detectable metastases is disproportionately low, confirming that tumor cell diffusion can happen at an early stage but usually occurs later on.
Early dissemination
Little is currently known about the preference of cancer subtypes for distinct tissues or about the receptiveness of a tissue as a metastatic site. What endures as one of the most confounding clinical phenomena is that patients may undergo tumor resection and remain apparently disease free for months, years, and even decades, only to experience relapse and be diagnosed with late-stage metastatic disease. This course may be a result of cell seeding from minimal residual disease after resection of the primary tumor or of preexisting clinically undetectable micrometastases. It may also arise from early disseminated cells that remain dormant and resistant to therapy until they suddenly reawaken to initiate proliferation into clinically detectable macrometastases.
Dormant DTC could be the main reason for delayed detection of metastases. It is thought that around 40% of patients with prostate cancer who undergo radical prostatectomy present with biochemical recurrence, suggesting that it is likely that hidden DTC or micrometastases are present at the time of the procedure. The finding is consistent with the detection of DTC many years after tumor resection, suggesting they were released before surgical treatment. Nevertheless, research into tumor cell dormancy is limited, owing to the invasive and technically challenging nature of obtaining DTC samples, which are predominantly taken from the bone marrow.
CTC metastases
Cancer cells can undergo epithelial-to-mesenchymal transition to facilitate their detachment from the primary tumor and intravasation into the blood circulation (step 1). Dissemination of cancer cells from the primary tumor into circulation can involve either single cells or cell clusters containing multiple CTC as well as immune cells and platelets, known as microemboli. CTC that can survive in circulation (step 2) can exit the bloodstream (step 3) and establish metastatic tumors (step 4), or they can enter dormancy and reside in distant organs, such as the bone marrow.
Use in practice
CTC were discovered over a century ago, but only in recent years has technology been sufficiently advanced to study CTC and to assess their usefulness as biomarkers. Recent evidence suggests that not only do the number of CTC increase during sleep and rest phases but also that these CTC are better able to metastasize, compared to those generated during periods of wakefulness or activity.
CTC clusters (microemboli) are defined as groups of two or more CTC. They can consist of CTC alone (homotypic) or can include various stromal cells, such as cancer-associated fibroblasts or platelets and immune cells (heterotypic). CTC clusters (with or without leukocytes) seem to have greater metastatic capacity, compared with individual CTC.
A multitude of characteristics can be measured in CTC, including genetics and epigenetics, as well as protein levels, which might help in understanding many processes involved in the formation of metastases.
Quantitative assessment of CTC could indicate tumor burden in patients with aggressive cancers, as has been seen in patients with primary lung cancer.
Early cancer diagnosis
Early research into CTC didn’t explore their usefulness in diagnosing early-stage tumors because it was thought that CTC were characteristic of advanced-stage disease. This hypothesis was later rejected following evidence of local intravascular invasion of very early cancer cells, even over a period of several hours. This feature may allow CTC to be detected before the clinical diagnosis of cancer.
CTC have been detected in various neoplastic conditions: in breast cancer, seen in 20% of patients with stage I disease, in 26.8% with stage II disease, and 26.7% with stage III disease; in nonmetastatic colorectal cancer, including stage I and II disease; and in prostate cancer, seen in over 50% of patients with localized disease.
The presence of CTC has been proven to be an unfavorable prognostic predictor of overall survival among patients with early-stage non–small cell lung cancer. It distinguishes patients with pancreatic ductal adenocarcinoma from those with noncancerous pancreatic diseases with a sensitivity of 75% and a specificity of 96.3%.
CTC positivity scoring (appropriately defined), combined with serum prostate-specific antigen level, was predictive of a biopsy diagnosis of clinically significant prostate cancer.
All these data support the utility of CTC in early cancer diagnosis. Their link with metastases, and thus with aggressive tumors, gives them an advantage over other (noninvasive or minimally invasive) biomarkers in the early identification of invasive tumors for therapeutic intervention with better cure rates.
This article was translated from Univadis Italy. A version appeared on Medscape.com.
Treatment options for patients with cancer that is detected at a late stage are severely limited, which usually leads to an unfavorable prognosis for such patients. Indeed, the options available for patients with metastatic solid cancers are scarcely curative. Therefore, early diagnosis of neoplasia remains a fundamental mainstay for improving outcomes for cancer patients.
Histopathology is the current gold standard for cancer diagnosis. Biopsy is an invasive procedure that provides physicians with further samples to test but that furnishes limited information concerning tumor heterogeneity. Biopsy specimens are usually obtained only when there is clinical evidence of neoplasia, which significantly limits their usefulness in early diagnosis.
Around 20 years ago, it was discovered that the presence of circulating tumor cells (CTC) in patients with metastatic breast cancer who were about to begin a new line of treatment was predictive of overall and progression-free survival. The prognostic value of CTC was independent of the line of treatment (first or second) and was greater than that of the site of metastasis, the type of therapy, and the time to metastasis after complete primary resection. These results support the idea that the presence of CTC could be used to modify the system for staging advanced disease.
Since then,
Liquid vs. tissue
Liquid biopsy is a minimally invasive tool that is easy to use. It is employed to detect cancer, to assess treatment response, or to monitor disease progression. Liquid biopsy produces test material from primary and metastatic (or micrometastatic) sites and provides a more heterogeneous picture of the entire tumor cell population, compared with specimens obtained with tissue biopsy.
Metastasis
The notion that metastatic lesions are formed from cancer cells that have disseminated from advanced primary tumors has been substantially revised following the identification of disseminated tumor cells (DTC) in the bone marrow of patients with early-stage disease. These results have led researchers to no longer view cancer metastasis as a linear cascade of events but rather as a series of concurrent, partially overlapping processes, as metastasizing cells assume new phenotypes while abandoning older behaviors.
The initiation of metastasis is not simply a cell-autonomous event but is heavily influenced by complex tissue microenvironments. Although colonization of distant tissues by DTC is an extremely inefficient process, at times, relatively numerous CTC can be detected in the blood of cancer patients (> 1,000 CTC/mL of blood plasma), whereas the number of clinically detectable metastases is disproportionately low, confirming that tumor cell diffusion can happen at an early stage but usually occurs later on.
Early dissemination
Little is currently known about the preference of cancer subtypes for distinct tissues or about the receptiveness of a tissue as a metastatic site. What endures as one of the most confounding clinical phenomena is that patients may undergo tumor resection and remain apparently disease free for months, years, and even decades, only to experience relapse and be diagnosed with late-stage metastatic disease. This course may be a result of cell seeding from minimal residual disease after resection of the primary tumor or of preexisting clinically undetectable micrometastases. It may also arise from early disseminated cells that remain dormant and resistant to therapy until they suddenly reawaken to initiate proliferation into clinically detectable macrometastases.
Dormant DTC could be the main reason for delayed detection of metastases. It is thought that around 40% of patients with prostate cancer who undergo radical prostatectomy present with biochemical recurrence, suggesting that it is likely that hidden DTC or micrometastases are present at the time of the procedure. The finding is consistent with the detection of DTC many years after tumor resection, suggesting they were released before surgical treatment. Nevertheless, research into tumor cell dormancy is limited, owing to the invasive and technically challenging nature of obtaining DTC samples, which are predominantly taken from the bone marrow.
CTC metastases
Cancer cells can undergo epithelial-to-mesenchymal transition to facilitate their detachment from the primary tumor and intravasation into the blood circulation (step 1). Dissemination of cancer cells from the primary tumor into circulation can involve either single cells or cell clusters containing multiple CTC as well as immune cells and platelets, known as microemboli. CTC that can survive in circulation (step 2) can exit the bloodstream (step 3) and establish metastatic tumors (step 4), or they can enter dormancy and reside in distant organs, such as the bone marrow.
Use in practice
CTC were discovered over a century ago, but only in recent years has technology been sufficiently advanced to study CTC and to assess their usefulness as biomarkers. Recent evidence suggests that not only do the number of CTC increase during sleep and rest phases but also that these CTC are better able to metastasize, compared to those generated during periods of wakefulness or activity.
CTC clusters (microemboli) are defined as groups of two or more CTC. They can consist of CTC alone (homotypic) or can include various stromal cells, such as cancer-associated fibroblasts or platelets and immune cells (heterotypic). CTC clusters (with or without leukocytes) seem to have greater metastatic capacity, compared with individual CTC.
A multitude of characteristics can be measured in CTC, including genetics and epigenetics, as well as protein levels, which might help in understanding many processes involved in the formation of metastases.
Quantitative assessment of CTC could indicate tumor burden in patients with aggressive cancers, as has been seen in patients with primary lung cancer.
Early cancer diagnosis
Early research into CTC didn’t explore their usefulness in diagnosing early-stage tumors because it was thought that CTC were characteristic of advanced-stage disease. This hypothesis was later rejected following evidence of local intravascular invasion of very early cancer cells, even over a period of several hours. This feature may allow CTC to be detected before the clinical diagnosis of cancer.
CTC have been detected in various neoplastic conditions: in breast cancer, seen in 20% of patients with stage I disease, in 26.8% with stage II disease, and 26.7% with stage III disease; in nonmetastatic colorectal cancer, including stage I and II disease; and in prostate cancer, seen in over 50% of patients with localized disease.
The presence of CTC has been proven to be an unfavorable prognostic predictor of overall survival among patients with early-stage non–small cell lung cancer. It distinguishes patients with pancreatic ductal adenocarcinoma from those with noncancerous pancreatic diseases with a sensitivity of 75% and a specificity of 96.3%.
CTC positivity scoring (appropriately defined), combined with serum prostate-specific antigen level, was predictive of a biopsy diagnosis of clinically significant prostate cancer.
All these data support the utility of CTC in early cancer diagnosis. Their link with metastases, and thus with aggressive tumors, gives them an advantage over other (noninvasive or minimally invasive) biomarkers in the early identification of invasive tumors for therapeutic intervention with better cure rates.
This article was translated from Univadis Italy. A version appeared on Medscape.com.
For NSCLC, neoadjuvant, adjuvant, or both?
This transcript has been edited for clarity.
Dr. West: Here at ASCO 2023 [American Society of Clinical Oncology] in Chicago, we’ve seen some blockbuster presentations in thoracic oncology. Many of these have brought up some important questions about the clinical implications that we need to discuss further.
At ASCO, as well as in the couple or 3 months preceding ASCO, we’ve gotten more and more data on perioperative approaches. Of course, over the past couple of years, we’ve had some new options of postoperative immunotherapy for a year, say, after chemotherapy or possibly after chemotherapy.
We have also had very influential data, such as the CheckMate 816 trial that gave three cycles of chemotherapy with nivolumab vs. chemotherapy alone to patients with stage IB to IIIA disease, but largely, nearly two thirds, with IIIA disease. That showed a very clear improvement in the pathologic complete response (pCR) rate with nivolumab added to chemotherapy and also a highly significant improvement in event-free survival and a strong trend toward improved overall survival. This is FDA approved and has been increasingly adopted, I would say, maybe with some variability by geography and center, but really a good amount of enthusiasm.
Now, we have a bunch of trials that give chemotherapy with immunotherapy. We’ve got the AEGEAN trial with durvalumab. We have Neotorch with chemotherapy and toripalimab. At ASCO 2023, we had a highly prominent presentation of KEYNOTE-671, giving four cycles of chemotherapy with pembrolizumab vs. chemotherapy and placebo.
Then there’s the built-in postoperative component of a year of immunotherapy as well, in all these trials. The data for KEYNOTE-671 look quite good. Of course, the other trials also were significant. I would say the comparator now is not nothing or chemotherapy alone anymore; it’s really against what is the best current standard of care.
It certainly adds some cost, it adds some risk for toxicity, and it adds a year of a patient coming into the clinic and getting IV infusions all this time to get a treatment that the patient has already had for four cycles in most of these trials.
If your cancer is resistant, is there going to be an incremental benefit to giving more of it? What are your thoughts about the risk and benefit? Going to a patient in your own clinic, how are you going to counsel your patients? Will anything change after the presentation of all these data and how you approach preoperatively?
Dr. Rotow: I agree. In some sense, it’s an embarrassment of riches, right?
Dr. West: Yes.
Dr. Rotow: We have so many positive studies looking at perioperative immunotherapy for our patients. They all show improved outcomes, but of course, they all compare with the old control arm of chemotherapy alone in some form, and this is no longer a useful control in this space. The open question is, do you use neoadjuvant, do you use adjuvant, or do you use both?
My high-level takeaway from these data is that the neoadjuvant component appears to be important. I think the overall trend, comparing across studies, of course, is that outcomes seem to be better with the neoadjuvant component. You also get the advantage of potential downstaging and potential greater ease of surgical resection. We know they have lower morbidity resection and shorter surgeries. You can comment on that. You also get your pathologic response data.
Dr. West: You get the feedback.
Dr. Rotow: Exactly.
Dr. West: The deliverability is also a big issue. You know you can much more reliably deliver your intended treatment by doing neoadjuvant followed by surgery.
Dr. Rotow: Exactly.
Dr. West: We know there’s major drop-off if patients have surgery, and in the recovery room they hear you got it all, and then they need to come back and maybe get chemotherapy and immunotherapy for a year. They’d ask, “What for? I can’t see anything.”
Dr. Rotow: Exactly. I think there are many advantages to that neoadjuvant component. I think all or many of us now have integrated this into our routine practice. Now the question is, do you need the adjuvant element or not on top? That is challenging because no trial has compared adjuvant to nonadjuvant. I think we all advocate for the need for this trial to answer this in a more randomized, prospective fashion. Of course, that doesn’t help our clinic practice tomorrow when we see a patient.
Dr. West: Or for the next 4 years.
Dr. Rotow: Or for the next 4 years – exactly. There’s going to be the open question of who really needs this? In some sense, we may be guided by the path response during the surgery itself. I think there may be those who claim that if you have a pCR, do you really need additional therapy? We don’t know the answer, but it’s tempting to say we know the outcomes in event-free survival are extremely good with a pCR.
Dr. West: Which is only 20% or 25% of patients, so it’s not most.
Dr. Rotow: It’s not most, but it’s better than the 2% or so with chemotherapy alone. That’s real progress, and it’s nice to have that readout. For that 80% without a pCR, what to do? I suspect there will be variation from provider to provider and from patient to patient, depending on tolerability to prior therapy, the patient’s wishes around the goals of care, and the patient’s risk for autoimmune toxicities.
Maybe there’s a patient with underlying autoimmune disease who’s gotten their neoadjuvant therapy and done well. You don’t want to risk that ongoing risk of exposure. Perhaps a patient with no risk factors who desires very aggressive treatment might be interested in more treatment.
In KEYNOTE-671, I was interested in the PD-L1 subgroups. These did trend the way you expect, with better responses in PD-L1 high, but there were also good outcomes and benefit to immunotherapy with the perioperative strategy in PD-L1–negative patients.
Dr. West: That didn’t really exclude anybody.
Dr. Rotow: It didn’t exclude anybody. In CheckMate 816, everyone benefited, but the benefit was less with those PD-L1–negative patients.
Dr. West: True.
Dr. Rotow: Absent further data to guide me or any prospective data here comparing these strategies, I might lean toward a longer course of immunotherapy in that population in hopes of triggering a response. I suspect that there will be variation from clinician to clinician in that space.
Dr. West: This is a setting where I feel like I have equipoise. I really feel that the incremental benefit is pretty small.
Dr. Rotow: Small. I agree.
Dr. West: It’s, frankly, somewhat dubious. On the other hand, you’re in a situation where if you know that three of four patients will experience a relapse and less-than-amazing outcomes, it’s hard to leave something that’s FDA approved and studied and a well-sanctioned option on the table if this patient may have relapse later.
In the end, I feel like I’d like to offer this and discuss it with all my patients. I think it’s a great place for shared decision-making because if a patient hears about that and decides they’re not interested, I’ll be fine with that. I think that’s a very sensible approach, but I don’t want to make it unilaterally. Other patients may say they want every opportunity, and if it comes back, at least I’ll know I did everything we could.
Dr. Rotow: Exactly. I agree with your statement about equipoise. I truly think that this is present here in the situation, and that there’s room for discussion in both directions with patients.
Now, one caveat I’d like to add to all these data is that the data should not apply to patients with some of our classic nonsmoking-associated driver mutations. This is another piece to the neoadjuvant data that I think is worth commenting on – the need to get appropriate testing before initiation of therapy and the pitfalls of starting this kind of treatment without knowing full biomarker testing. I think that’s something we have to watch for in our clinical practice as well.
Dr. West: Perhaps especially if we’re talking about doing a year of postoperative and someone has an ALK rearrangement or an EGFR mutation and we didn’t know it. That is a group where we’re worried about a rapid transition and potentially prohibitive, even life-threatening, toxicities from not planning in advance for this. This is something you don’t want to give concurrently or one right on top of the other. You don’t want to give immunotherapy and then transition right to targeted therapy. It’s dangerous.
Dr. Rotow: Exactly. The stakes were already high with neoadjuvant alone, but at least you had that gap of the presurgical period, surgical recovery, and then initiation of adjuvant therapy, if needed, or at relapse. With a postoperative long adjuvant period, those stakes are elevated because the immunotherapy exposure continues, so it’s something to be mindful of.
Dr. West: We have a general sense that many, but not all, of the targets that we’re talking about are associated with low benefit from immunotherapy. It’s not that well studied. I think this is another place for individualized discussion of the pros and cons. They were included in the trial, but they probably benefit less.
Dr. Rotow: Exactly. I think with the best established, EGFR and ALK probably are not benefiting much. They were actually included in the trial. Many of the neoadjuvant studies do not allow them to enroll if they’re known. On the other end of that spectrum, I think KRAS is just fine to treat with immunotherapy.
Dr. West: Sure.
Dr. Rotow: It’s an actionable driver. It’s not a traditional nonsmoking-associated driver, and those do just fine.
Dr. West: The studies show that these patients benefit just as much, at least, as the other patients.
Dr. Rotow: Exactly. I would never withhold this form of therapy for a KRAS driver mutation. The others, I think, are still in a gray zone. Depending on the patient demographics and tobacco use, I may elicit more or less caution in that space.
Dr. West: Well, I think we’re going to have much to still tease apart, with room for judgment here without a strong sense of the data telling us exactly what to do.
Dr. Rotow: Exactly.
Dr. West: There’s a large amount of excitement and interest in these new data, but there are still many open questions. I hope we continue to mull it over as we get more data and more insight to shape our plans.
Dr. West is an associate professor at City of Hope Comprehensive Cancer Center in Duarte, Calif., and vice president of network strategy at AccessHope in Los Angeles. Dr. Rotow is the clinical director of the Lowe Center for Thoracic Oncology at the Dana-Farber Cancer Institute in Boston. Dr. West reported conflicts of interest with Ariad/Takeda, Bristol Myers Squibb, Boehringer Ingelheim, Spectrum, AstraZeneca, Celgene, Genentech/Roche, Pfizer, Merck, and Eli Lilly. Dr. Rotow reported conflicts of interest with Genentech, AstraZeneca,Guardant, and Janssen.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Dr. West: Here at ASCO 2023 [American Society of Clinical Oncology] in Chicago, we’ve seen some blockbuster presentations in thoracic oncology. Many of these have brought up some important questions about the clinical implications that we need to discuss further.
At ASCO, as well as in the couple or 3 months preceding ASCO, we’ve gotten more and more data on perioperative approaches. Of course, over the past couple of years, we’ve had some new options of postoperative immunotherapy for a year, say, after chemotherapy or possibly after chemotherapy.
We have also had very influential data, such as the CheckMate 816 trial that gave three cycles of chemotherapy with nivolumab vs. chemotherapy alone to patients with stage IB to IIIA disease, but largely, nearly two thirds, with IIIA disease. That showed a very clear improvement in the pathologic complete response (pCR) rate with nivolumab added to chemotherapy and also a highly significant improvement in event-free survival and a strong trend toward improved overall survival. This is FDA approved and has been increasingly adopted, I would say, maybe with some variability by geography and center, but really a good amount of enthusiasm.
Now, we have a bunch of trials that give chemotherapy with immunotherapy. We’ve got the AEGEAN trial with durvalumab. We have Neotorch with chemotherapy and toripalimab. At ASCO 2023, we had a highly prominent presentation of KEYNOTE-671, giving four cycles of chemotherapy with pembrolizumab vs. chemotherapy and placebo.
Then there’s the built-in postoperative component of a year of immunotherapy as well, in all these trials. The data for KEYNOTE-671 look quite good. Of course, the other trials also were significant. I would say the comparator now is not nothing or chemotherapy alone anymore; it’s really against what is the best current standard of care.
It certainly adds some cost, it adds some risk for toxicity, and it adds a year of a patient coming into the clinic and getting IV infusions all this time to get a treatment that the patient has already had for four cycles in most of these trials.
If your cancer is resistant, is there going to be an incremental benefit to giving more of it? What are your thoughts about the risk and benefit? Going to a patient in your own clinic, how are you going to counsel your patients? Will anything change after the presentation of all these data and how you approach preoperatively?
Dr. Rotow: I agree. In some sense, it’s an embarrassment of riches, right?
Dr. West: Yes.
Dr. Rotow: We have so many positive studies looking at perioperative immunotherapy for our patients. They all show improved outcomes, but of course, they all compare with the old control arm of chemotherapy alone in some form, and this is no longer a useful control in this space. The open question is, do you use neoadjuvant, do you use adjuvant, or do you use both?
My high-level takeaway from these data is that the neoadjuvant component appears to be important. I think the overall trend, comparing across studies, of course, is that outcomes seem to be better with the neoadjuvant component. You also get the advantage of potential downstaging and potential greater ease of surgical resection. We know they have lower morbidity resection and shorter surgeries. You can comment on that. You also get your pathologic response data.
Dr. West: You get the feedback.
Dr. Rotow: Exactly.
Dr. West: The deliverability is also a big issue. You know you can much more reliably deliver your intended treatment by doing neoadjuvant followed by surgery.
Dr. Rotow: Exactly.
Dr. West: We know there’s major drop-off if patients have surgery, and in the recovery room they hear you got it all, and then they need to come back and maybe get chemotherapy and immunotherapy for a year. They’d ask, “What for? I can’t see anything.”
Dr. Rotow: Exactly. I think there are many advantages to that neoadjuvant component. I think all or many of us now have integrated this into our routine practice. Now the question is, do you need the adjuvant element or not on top? That is challenging because no trial has compared adjuvant to nonadjuvant. I think we all advocate for the need for this trial to answer this in a more randomized, prospective fashion. Of course, that doesn’t help our clinic practice tomorrow when we see a patient.
Dr. West: Or for the next 4 years.
Dr. Rotow: Or for the next 4 years – exactly. There’s going to be the open question of who really needs this? In some sense, we may be guided by the path response during the surgery itself. I think there may be those who claim that if you have a pCR, do you really need additional therapy? We don’t know the answer, but it’s tempting to say we know the outcomes in event-free survival are extremely good with a pCR.
Dr. West: Which is only 20% or 25% of patients, so it’s not most.
Dr. Rotow: It’s not most, but it’s better than the 2% or so with chemotherapy alone. That’s real progress, and it’s nice to have that readout. For that 80% without a pCR, what to do? I suspect there will be variation from provider to provider and from patient to patient, depending on tolerability to prior therapy, the patient’s wishes around the goals of care, and the patient’s risk for autoimmune toxicities.
Maybe there’s a patient with underlying autoimmune disease who’s gotten their neoadjuvant therapy and done well. You don’t want to risk that ongoing risk of exposure. Perhaps a patient with no risk factors who desires very aggressive treatment might be interested in more treatment.
In KEYNOTE-671, I was interested in the PD-L1 subgroups. These did trend the way you expect, with better responses in PD-L1 high, but there were also good outcomes and benefit to immunotherapy with the perioperative strategy in PD-L1–negative patients.
Dr. West: That didn’t really exclude anybody.
Dr. Rotow: It didn’t exclude anybody. In CheckMate 816, everyone benefited, but the benefit was less with those PD-L1–negative patients.
Dr. West: True.
Dr. Rotow: Absent further data to guide me or any prospective data here comparing these strategies, I might lean toward a longer course of immunotherapy in that population in hopes of triggering a response. I suspect that there will be variation from clinician to clinician in that space.
Dr. West: This is a setting where I feel like I have equipoise. I really feel that the incremental benefit is pretty small.
Dr. Rotow: Small. I agree.
Dr. West: It’s, frankly, somewhat dubious. On the other hand, you’re in a situation where if you know that three of four patients will experience a relapse and less-than-amazing outcomes, it’s hard to leave something that’s FDA approved and studied and a well-sanctioned option on the table if this patient may have relapse later.
In the end, I feel like I’d like to offer this and discuss it with all my patients. I think it’s a great place for shared decision-making because if a patient hears about that and decides they’re not interested, I’ll be fine with that. I think that’s a very sensible approach, but I don’t want to make it unilaterally. Other patients may say they want every opportunity, and if it comes back, at least I’ll know I did everything we could.
Dr. Rotow: Exactly. I agree with your statement about equipoise. I truly think that this is present here in the situation, and that there’s room for discussion in both directions with patients.
Now, one caveat I’d like to add to all these data is that the data should not apply to patients with some of our classic nonsmoking-associated driver mutations. This is another piece to the neoadjuvant data that I think is worth commenting on – the need to get appropriate testing before initiation of therapy and the pitfalls of starting this kind of treatment without knowing full biomarker testing. I think that’s something we have to watch for in our clinical practice as well.
Dr. West: Perhaps especially if we’re talking about doing a year of postoperative and someone has an ALK rearrangement or an EGFR mutation and we didn’t know it. That is a group where we’re worried about a rapid transition and potentially prohibitive, even life-threatening, toxicities from not planning in advance for this. This is something you don’t want to give concurrently or one right on top of the other. You don’t want to give immunotherapy and then transition right to targeted therapy. It’s dangerous.
Dr. Rotow: Exactly. The stakes were already high with neoadjuvant alone, but at least you had that gap of the presurgical period, surgical recovery, and then initiation of adjuvant therapy, if needed, or at relapse. With a postoperative long adjuvant period, those stakes are elevated because the immunotherapy exposure continues, so it’s something to be mindful of.
Dr. West: We have a general sense that many, but not all, of the targets that we’re talking about are associated with low benefit from immunotherapy. It’s not that well studied. I think this is another place for individualized discussion of the pros and cons. They were included in the trial, but they probably benefit less.
Dr. Rotow: Exactly. I think with the best established, EGFR and ALK probably are not benefiting much. They were actually included in the trial. Many of the neoadjuvant studies do not allow them to enroll if they’re known. On the other end of that spectrum, I think KRAS is just fine to treat with immunotherapy.
Dr. West: Sure.
Dr. Rotow: It’s an actionable driver. It’s not a traditional nonsmoking-associated driver, and those do just fine.
Dr. West: The studies show that these patients benefit just as much, at least, as the other patients.
Dr. Rotow: Exactly. I would never withhold this form of therapy for a KRAS driver mutation. The others, I think, are still in a gray zone. Depending on the patient demographics and tobacco use, I may elicit more or less caution in that space.
Dr. West: Well, I think we’re going to have much to still tease apart, with room for judgment here without a strong sense of the data telling us exactly what to do.
Dr. Rotow: Exactly.
Dr. West: There’s a large amount of excitement and interest in these new data, but there are still many open questions. I hope we continue to mull it over as we get more data and more insight to shape our plans.
Dr. West is an associate professor at City of Hope Comprehensive Cancer Center in Duarte, Calif., and vice president of network strategy at AccessHope in Los Angeles. Dr. Rotow is the clinical director of the Lowe Center for Thoracic Oncology at the Dana-Farber Cancer Institute in Boston. Dr. West reported conflicts of interest with Ariad/Takeda, Bristol Myers Squibb, Boehringer Ingelheim, Spectrum, AstraZeneca, Celgene, Genentech/Roche, Pfizer, Merck, and Eli Lilly. Dr. Rotow reported conflicts of interest with Genentech, AstraZeneca,Guardant, and Janssen.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Dr. West: Here at ASCO 2023 [American Society of Clinical Oncology] in Chicago, we’ve seen some blockbuster presentations in thoracic oncology. Many of these have brought up some important questions about the clinical implications that we need to discuss further.
At ASCO, as well as in the couple or 3 months preceding ASCO, we’ve gotten more and more data on perioperative approaches. Of course, over the past couple of years, we’ve had some new options of postoperative immunotherapy for a year, say, after chemotherapy or possibly after chemotherapy.
We have also had very influential data, such as the CheckMate 816 trial that gave three cycles of chemotherapy with nivolumab vs. chemotherapy alone to patients with stage IB to IIIA disease, but largely, nearly two thirds, with IIIA disease. That showed a very clear improvement in the pathologic complete response (pCR) rate with nivolumab added to chemotherapy and also a highly significant improvement in event-free survival and a strong trend toward improved overall survival. This is FDA approved and has been increasingly adopted, I would say, maybe with some variability by geography and center, but really a good amount of enthusiasm.
Now, we have a bunch of trials that give chemotherapy with immunotherapy. We’ve got the AEGEAN trial with durvalumab. We have Neotorch with chemotherapy and toripalimab. At ASCO 2023, we had a highly prominent presentation of KEYNOTE-671, giving four cycles of chemotherapy with pembrolizumab vs. chemotherapy and placebo.
Then there’s the built-in postoperative component of a year of immunotherapy as well, in all these trials. The data for KEYNOTE-671 look quite good. Of course, the other trials also were significant. I would say the comparator now is not nothing or chemotherapy alone anymore; it’s really against what is the best current standard of care.
It certainly adds some cost, it adds some risk for toxicity, and it adds a year of a patient coming into the clinic and getting IV infusions all this time to get a treatment that the patient has already had for four cycles in most of these trials.
If your cancer is resistant, is there going to be an incremental benefit to giving more of it? What are your thoughts about the risk and benefit? Going to a patient in your own clinic, how are you going to counsel your patients? Will anything change after the presentation of all these data and how you approach preoperatively?
Dr. Rotow: I agree. In some sense, it’s an embarrassment of riches, right?
Dr. West: Yes.
Dr. Rotow: We have so many positive studies looking at perioperative immunotherapy for our patients. They all show improved outcomes, but of course, they all compare with the old control arm of chemotherapy alone in some form, and this is no longer a useful control in this space. The open question is, do you use neoadjuvant, do you use adjuvant, or do you use both?
My high-level takeaway from these data is that the neoadjuvant component appears to be important. I think the overall trend, comparing across studies, of course, is that outcomes seem to be better with the neoadjuvant component. You also get the advantage of potential downstaging and potential greater ease of surgical resection. We know they have lower morbidity resection and shorter surgeries. You can comment on that. You also get your pathologic response data.
Dr. West: You get the feedback.
Dr. Rotow: Exactly.
Dr. West: The deliverability is also a big issue. You know you can much more reliably deliver your intended treatment by doing neoadjuvant followed by surgery.
Dr. Rotow: Exactly.
Dr. West: We know there’s major drop-off if patients have surgery, and in the recovery room they hear you got it all, and then they need to come back and maybe get chemotherapy and immunotherapy for a year. They’d ask, “What for? I can’t see anything.”
Dr. Rotow: Exactly. I think there are many advantages to that neoadjuvant component. I think all or many of us now have integrated this into our routine practice. Now the question is, do you need the adjuvant element or not on top? That is challenging because no trial has compared adjuvant to nonadjuvant. I think we all advocate for the need for this trial to answer this in a more randomized, prospective fashion. Of course, that doesn’t help our clinic practice tomorrow when we see a patient.
Dr. West: Or for the next 4 years.
Dr. Rotow: Or for the next 4 years – exactly. There’s going to be the open question of who really needs this? In some sense, we may be guided by the path response during the surgery itself. I think there may be those who claim that if you have a pCR, do you really need additional therapy? We don’t know the answer, but it’s tempting to say we know the outcomes in event-free survival are extremely good with a pCR.
Dr. West: Which is only 20% or 25% of patients, so it’s not most.
Dr. Rotow: It’s not most, but it’s better than the 2% or so with chemotherapy alone. That’s real progress, and it’s nice to have that readout. For that 80% without a pCR, what to do? I suspect there will be variation from provider to provider and from patient to patient, depending on tolerability to prior therapy, the patient’s wishes around the goals of care, and the patient’s risk for autoimmune toxicities.
Maybe there’s a patient with underlying autoimmune disease who’s gotten their neoadjuvant therapy and done well. You don’t want to risk that ongoing risk of exposure. Perhaps a patient with no risk factors who desires very aggressive treatment might be interested in more treatment.
In KEYNOTE-671, I was interested in the PD-L1 subgroups. These did trend the way you expect, with better responses in PD-L1 high, but there were also good outcomes and benefit to immunotherapy with the perioperative strategy in PD-L1–negative patients.
Dr. West: That didn’t really exclude anybody.
Dr. Rotow: It didn’t exclude anybody. In CheckMate 816, everyone benefited, but the benefit was less with those PD-L1–negative patients.
Dr. West: True.
Dr. Rotow: Absent further data to guide me or any prospective data here comparing these strategies, I might lean toward a longer course of immunotherapy in that population in hopes of triggering a response. I suspect that there will be variation from clinician to clinician in that space.
Dr. West: This is a setting where I feel like I have equipoise. I really feel that the incremental benefit is pretty small.
Dr. Rotow: Small. I agree.
Dr. West: It’s, frankly, somewhat dubious. On the other hand, you’re in a situation where if you know that three of four patients will experience a relapse and less-than-amazing outcomes, it’s hard to leave something that’s FDA approved and studied and a well-sanctioned option on the table if this patient may have relapse later.
In the end, I feel like I’d like to offer this and discuss it with all my patients. I think it’s a great place for shared decision-making because if a patient hears about that and decides they’re not interested, I’ll be fine with that. I think that’s a very sensible approach, but I don’t want to make it unilaterally. Other patients may say they want every opportunity, and if it comes back, at least I’ll know I did everything we could.
Dr. Rotow: Exactly. I agree with your statement about equipoise. I truly think that this is present here in the situation, and that there’s room for discussion in both directions with patients.
Now, one caveat I’d like to add to all these data is that the data should not apply to patients with some of our classic nonsmoking-associated driver mutations. This is another piece to the neoadjuvant data that I think is worth commenting on – the need to get appropriate testing before initiation of therapy and the pitfalls of starting this kind of treatment without knowing full biomarker testing. I think that’s something we have to watch for in our clinical practice as well.
Dr. West: Perhaps especially if we’re talking about doing a year of postoperative and someone has an ALK rearrangement or an EGFR mutation and we didn’t know it. That is a group where we’re worried about a rapid transition and potentially prohibitive, even life-threatening, toxicities from not planning in advance for this. This is something you don’t want to give concurrently or one right on top of the other. You don’t want to give immunotherapy and then transition right to targeted therapy. It’s dangerous.
Dr. Rotow: Exactly. The stakes were already high with neoadjuvant alone, but at least you had that gap of the presurgical period, surgical recovery, and then initiation of adjuvant therapy, if needed, or at relapse. With a postoperative long adjuvant period, those stakes are elevated because the immunotherapy exposure continues, so it’s something to be mindful of.
Dr. West: We have a general sense that many, but not all, of the targets that we’re talking about are associated with low benefit from immunotherapy. It’s not that well studied. I think this is another place for individualized discussion of the pros and cons. They were included in the trial, but they probably benefit less.
Dr. Rotow: Exactly. I think with the best established, EGFR and ALK probably are not benefiting much. They were actually included in the trial. Many of the neoadjuvant studies do not allow them to enroll if they’re known. On the other end of that spectrum, I think KRAS is just fine to treat with immunotherapy.
Dr. West: Sure.
Dr. Rotow: It’s an actionable driver. It’s not a traditional nonsmoking-associated driver, and those do just fine.
Dr. West: The studies show that these patients benefit just as much, at least, as the other patients.
Dr. Rotow: Exactly. I would never withhold this form of therapy for a KRAS driver mutation. The others, I think, are still in a gray zone. Depending on the patient demographics and tobacco use, I may elicit more or less caution in that space.
Dr. West: Well, I think we’re going to have much to still tease apart, with room for judgment here without a strong sense of the data telling us exactly what to do.
Dr. Rotow: Exactly.
Dr. West: There’s a large amount of excitement and interest in these new data, but there are still many open questions. I hope we continue to mull it over as we get more data and more insight to shape our plans.
Dr. West is an associate professor at City of Hope Comprehensive Cancer Center in Duarte, Calif., and vice president of network strategy at AccessHope in Los Angeles. Dr. Rotow is the clinical director of the Lowe Center for Thoracic Oncology at the Dana-Farber Cancer Institute in Boston. Dr. West reported conflicts of interest with Ariad/Takeda, Bristol Myers Squibb, Boehringer Ingelheim, Spectrum, AstraZeneca, Celgene, Genentech/Roche, Pfizer, Merck, and Eli Lilly. Dr. Rotow reported conflicts of interest with Genentech, AstraZeneca,Guardant, and Janssen.
A version of this article first appeared on Medscape.com.
FROM ASCO 2023
RFS failed as endpoint in adjuvant immunotherapy trials
TOPLINE:
METHODOLOGY:
- FDA approvals in the adjuvant setting for cancer immunotherapy are increasingly based on trials that use RFS as a surrogate endpoint for overall survival, largely because such a design allows for smaller, speedier trials.
- To test the validity of using RFS as a surrogate for overall survival in this setting, investigators conducted a meta-analysis of 15 phase 2 and 3 randomized controlled trials (RCTs) of adjuvant CTLA4 and anti–PD-1/PD-L1 blockers for melanoma, non–small cell lung cancer, renal cell cancer, and other tumors.
- The team used weighted regression at the arm and trial levels to assess the efficacy of RFS as a surrogate for overall survival.
- The strength of the association was quantified by weighted coefficients of determination (R2)12Dante MT Stdplz make sure all mentions of R’2’ are superscript, with a strong correlation considered to be R2 of 0.7 or higher.
- If there were strong correlations at both the arm and trial levels, RFS would be considered a robust surrogate endpoint for overall survival; however, if one of the correlations at the arm or trial level was not strong, RFS would not be considered a surrogate endpoint for overall survival.
TAKEAWAY:
- At the arm level, moderate and strong associations were observed between 2-year RFS and 3-year overall survival (R2, 0.58) and between 3-year RFS and 5-year overall survival (R2, 0.72; 95% confidence interval, 0.38-.00).
- At the trial level, a moderate association was observed between effect of treatment on RFS and overall survival (R2, 0.63).
- The findings were confirmed in several sensitivity analyses that were based on different trial phases, experimental arms, cancer types, and treatment strategies.
IN PRACTICE:
“Our meta-analysis failed to find a significantly strong association between RFS and OS in RCTs of adjuvant immunotherapy,” the authors concluded. “RFS should not be used as a surrogate endpoint for OS in this clinical context.” Instead, the finding indicates that overall survival is “the ideal primary endpoint” in this setting.
SOURCE:
The study, led by Yuanfang Li, PhD, of Sun Yat-sen University Cancer Center in Guangzhou, China, was published in the Journal of the National Cancer Institute.
LIMITATIONS:
- Correlations were calculated from a relatively limited number of RCTs that involved different types of cancer, and overall survival data were not fully mature in some of the trials.
- The analysis did not include patient-level data.
DISCLOSURES:
- The work was funded by the National Natural Science Foundation of China and others.
- The investigators had no disclosures.
A version of this article first appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- FDA approvals in the adjuvant setting for cancer immunotherapy are increasingly based on trials that use RFS as a surrogate endpoint for overall survival, largely because such a design allows for smaller, speedier trials.
- To test the validity of using RFS as a surrogate for overall survival in this setting, investigators conducted a meta-analysis of 15 phase 2 and 3 randomized controlled trials (RCTs) of adjuvant CTLA4 and anti–PD-1/PD-L1 blockers for melanoma, non–small cell lung cancer, renal cell cancer, and other tumors.
- The team used weighted regression at the arm and trial levels to assess the efficacy of RFS as a surrogate for overall survival.
- The strength of the association was quantified by weighted coefficients of determination (R2)12Dante MT Stdplz make sure all mentions of R’2’ are superscript, with a strong correlation considered to be R2 of 0.7 or higher.
- If there were strong correlations at both the arm and trial levels, RFS would be considered a robust surrogate endpoint for overall survival; however, if one of the correlations at the arm or trial level was not strong, RFS would not be considered a surrogate endpoint for overall survival.
TAKEAWAY:
- At the arm level, moderate and strong associations were observed between 2-year RFS and 3-year overall survival (R2, 0.58) and between 3-year RFS and 5-year overall survival (R2, 0.72; 95% confidence interval, 0.38-.00).
- At the trial level, a moderate association was observed between effect of treatment on RFS and overall survival (R2, 0.63).
- The findings were confirmed in several sensitivity analyses that were based on different trial phases, experimental arms, cancer types, and treatment strategies.
IN PRACTICE:
“Our meta-analysis failed to find a significantly strong association between RFS and OS in RCTs of adjuvant immunotherapy,” the authors concluded. “RFS should not be used as a surrogate endpoint for OS in this clinical context.” Instead, the finding indicates that overall survival is “the ideal primary endpoint” in this setting.
SOURCE:
The study, led by Yuanfang Li, PhD, of Sun Yat-sen University Cancer Center in Guangzhou, China, was published in the Journal of the National Cancer Institute.
LIMITATIONS:
- Correlations were calculated from a relatively limited number of RCTs that involved different types of cancer, and overall survival data were not fully mature in some of the trials.
- The analysis did not include patient-level data.
DISCLOSURES:
- The work was funded by the National Natural Science Foundation of China and others.
- The investigators had no disclosures.
A version of this article first appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- FDA approvals in the adjuvant setting for cancer immunotherapy are increasingly based on trials that use RFS as a surrogate endpoint for overall survival, largely because such a design allows for smaller, speedier trials.
- To test the validity of using RFS as a surrogate for overall survival in this setting, investigators conducted a meta-analysis of 15 phase 2 and 3 randomized controlled trials (RCTs) of adjuvant CTLA4 and anti–PD-1/PD-L1 blockers for melanoma, non–small cell lung cancer, renal cell cancer, and other tumors.
- The team used weighted regression at the arm and trial levels to assess the efficacy of RFS as a surrogate for overall survival.
- The strength of the association was quantified by weighted coefficients of determination (R2)12Dante MT Stdplz make sure all mentions of R’2’ are superscript, with a strong correlation considered to be R2 of 0.7 or higher.
- If there were strong correlations at both the arm and trial levels, RFS would be considered a robust surrogate endpoint for overall survival; however, if one of the correlations at the arm or trial level was not strong, RFS would not be considered a surrogate endpoint for overall survival.
TAKEAWAY:
- At the arm level, moderate and strong associations were observed between 2-year RFS and 3-year overall survival (R2, 0.58) and between 3-year RFS and 5-year overall survival (R2, 0.72; 95% confidence interval, 0.38-.00).
- At the trial level, a moderate association was observed between effect of treatment on RFS and overall survival (R2, 0.63).
- The findings were confirmed in several sensitivity analyses that were based on different trial phases, experimental arms, cancer types, and treatment strategies.
IN PRACTICE:
“Our meta-analysis failed to find a significantly strong association between RFS and OS in RCTs of adjuvant immunotherapy,” the authors concluded. “RFS should not be used as a surrogate endpoint for OS in this clinical context.” Instead, the finding indicates that overall survival is “the ideal primary endpoint” in this setting.
SOURCE:
The study, led by Yuanfang Li, PhD, of Sun Yat-sen University Cancer Center in Guangzhou, China, was published in the Journal of the National Cancer Institute.
LIMITATIONS:
- Correlations were calculated from a relatively limited number of RCTs that involved different types of cancer, and overall survival data were not fully mature in some of the trials.
- The analysis did not include patient-level data.
DISCLOSURES:
- The work was funded by the National Natural Science Foundation of China and others.
- The investigators had no disclosures.
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF THE NATIONAL CANCER INSTITUTE
Short bursts of activity may cut cancer risk
, a new study published in JAMA Oncology says.
Researchers at the University of Sydney studied data from wearable fitness devices worn by more than 22,000 “non-exercisers,” then examined their health records for 6 or 7 years.
The scientists found that people who did 4-5 minutes of “vigorous intermittent lifestyle physical activity” (VILPA) had a “substantially” lower cancer risk than people who did no VILPA.
Examples of VILPA are vigorous housework, carrying heavy shopping bags around the grocery store, bursts of power walking, and playing high-energy games with children. The activities could occur in 1-minute bursts, instead of all at once.

The study found that a minimum of around 3.5 minutes of daily VILPA was linked to an 18% reduction in cancer rates, compared with no VILPA. The study said 4.5 minutes of daily VILPA was linked to a 32% reduction in cancers related to physical activity, including lung, kidney, bladder, and stomach cancers.
“We know the majority of middle-aged people don’t regularly exercise, which puts them at increased cancer risk, but it’s only through the advent of wearable technology like activity trackers that we are able to look at the impact of short bursts of incidental physical activity done as part of daily living,” Emmanuel Stamatakis, PhD, the lead author of the study and a professor at the University of Sydney’s Charles Perkins Centre, said in a news release.
Study participants had an average age of 62 and reported that they didn’t exercise in their spare time. VILPA, a concept coined by researchers at the university, was measured by wrist accelerometers that people in the study wore over 7 days at the start of the study, the news release said.
“We are just starting to glimpse the potential of wearable technology to track physical activity and understand how unexplored aspects of our lives affect our long-term health – the potential impact on cancer prevention and a host of other health outcomes is enormous,” Dr. Stamatakis said.
A version of this article first appeared on WebMD.com.
, a new study published in JAMA Oncology says.
Researchers at the University of Sydney studied data from wearable fitness devices worn by more than 22,000 “non-exercisers,” then examined their health records for 6 or 7 years.
The scientists found that people who did 4-5 minutes of “vigorous intermittent lifestyle physical activity” (VILPA) had a “substantially” lower cancer risk than people who did no VILPA.
Examples of VILPA are vigorous housework, carrying heavy shopping bags around the grocery store, bursts of power walking, and playing high-energy games with children. The activities could occur in 1-minute bursts, instead of all at once.

The study found that a minimum of around 3.5 minutes of daily VILPA was linked to an 18% reduction in cancer rates, compared with no VILPA. The study said 4.5 minutes of daily VILPA was linked to a 32% reduction in cancers related to physical activity, including lung, kidney, bladder, and stomach cancers.
“We know the majority of middle-aged people don’t regularly exercise, which puts them at increased cancer risk, but it’s only through the advent of wearable technology like activity trackers that we are able to look at the impact of short bursts of incidental physical activity done as part of daily living,” Emmanuel Stamatakis, PhD, the lead author of the study and a professor at the University of Sydney’s Charles Perkins Centre, said in a news release.
Study participants had an average age of 62 and reported that they didn’t exercise in their spare time. VILPA, a concept coined by researchers at the university, was measured by wrist accelerometers that people in the study wore over 7 days at the start of the study, the news release said.
“We are just starting to glimpse the potential of wearable technology to track physical activity and understand how unexplored aspects of our lives affect our long-term health – the potential impact on cancer prevention and a host of other health outcomes is enormous,” Dr. Stamatakis said.
A version of this article first appeared on WebMD.com.
, a new study published in JAMA Oncology says.
Researchers at the University of Sydney studied data from wearable fitness devices worn by more than 22,000 “non-exercisers,” then examined their health records for 6 or 7 years.
The scientists found that people who did 4-5 minutes of “vigorous intermittent lifestyle physical activity” (VILPA) had a “substantially” lower cancer risk than people who did no VILPA.
Examples of VILPA are vigorous housework, carrying heavy shopping bags around the grocery store, bursts of power walking, and playing high-energy games with children. The activities could occur in 1-minute bursts, instead of all at once.

The study found that a minimum of around 3.5 minutes of daily VILPA was linked to an 18% reduction in cancer rates, compared with no VILPA. The study said 4.5 minutes of daily VILPA was linked to a 32% reduction in cancers related to physical activity, including lung, kidney, bladder, and stomach cancers.
“We know the majority of middle-aged people don’t regularly exercise, which puts them at increased cancer risk, but it’s only through the advent of wearable technology like activity trackers that we are able to look at the impact of short bursts of incidental physical activity done as part of daily living,” Emmanuel Stamatakis, PhD, the lead author of the study and a professor at the University of Sydney’s Charles Perkins Centre, said in a news release.
Study participants had an average age of 62 and reported that they didn’t exercise in their spare time. VILPA, a concept coined by researchers at the university, was measured by wrist accelerometers that people in the study wore over 7 days at the start of the study, the news release said.
“We are just starting to glimpse the potential of wearable technology to track physical activity and understand how unexplored aspects of our lives affect our long-term health – the potential impact on cancer prevention and a host of other health outcomes is enormous,” Dr. Stamatakis said.
A version of this article first appeared on WebMD.com.
FROM JAMA ONCOLOGY