User login
Evaluating the Malignant Potential of Nevus Spilus
PD-L1 positivity correlated with pembrolizumab response in advanced melanoma
Programmed death ligand–1 expression correlated positively and significantly with pembrolizumab response in advanced melanoma, based on an analysis of 405 patients from the international, multicohort, open-label phase I KEYNOTE-001 trial.
Among patients for whom 33%-65% of tumor cells expressed PD-L1, the objective rate of response was 57%, but the rate was only 8% among patients whose specimens did not express PD-L1, Adil Daud, MD, of the University of California, San Francisco, and associates reported. Taken together, the findings suggest that melanoma is most likely to respond to pembrolizumab when specimens show at least 10% positivity, the investigators reported (J Clin Oncol. 2016 Oct 10. doi: 10.1200/JCO.2016.67.2477).
Among the 405 patients who also were evaluable for tumor response, the overall objective response rate was 33% (95% confidence interval, 28%-37%). Grouping patients based on the melanoma scoring system for PD-L1 expression showed that PD-L1 positivity correlated significantly with the objective response rate (P less than .001). Furthermore, a higher PD-L1 melanoma score correlated significantly with both progression-free survival (hazard ratio, 0.76; 95% CI, 0.7-0.82) and overall survival (HR, 0.76; 95% CI, 0.69-0.83), with P values less than .001 for each association.
Median progression-free survival was 5.6 months in PD-L1–positive patients and 2.8 months in PD-L1–negative patients, while median overall survival was 30 months in PD-L1–positive patients and 12.6 months in PD-L1–negative patients, the researchers reported. “The high prevalence of PD-L1 positivity observed in this study, along with the durable responses observed in PD-L1–negative tumors, suggest that pembrolizumab treatment should not be limited to patients with PD-L1–positive tumors,” they concluded. “Ongoing clinical trials with correlative studies will further delineate the role of PD-L1 expression in melanoma.”
Merck sponsored the trial. Dr. Daud disclosed ties to Merck, OncoSec, Novartis, Genentech, Bristol-Myers Squibb, and Array BioPharma.
Programmed death ligand–1 expression correlated positively and significantly with pembrolizumab response in advanced melanoma, based on an analysis of 405 patients from the international, multicohort, open-label phase I KEYNOTE-001 trial.
Among patients for whom 33%-65% of tumor cells expressed PD-L1, the objective rate of response was 57%, but the rate was only 8% among patients whose specimens did not express PD-L1, Adil Daud, MD, of the University of California, San Francisco, and associates reported. Taken together, the findings suggest that melanoma is most likely to respond to pembrolizumab when specimens show at least 10% positivity, the investigators reported (J Clin Oncol. 2016 Oct 10. doi: 10.1200/JCO.2016.67.2477).
Among the 405 patients who also were evaluable for tumor response, the overall objective response rate was 33% (95% confidence interval, 28%-37%). Grouping patients based on the melanoma scoring system for PD-L1 expression showed that PD-L1 positivity correlated significantly with the objective response rate (P less than .001). Furthermore, a higher PD-L1 melanoma score correlated significantly with both progression-free survival (hazard ratio, 0.76; 95% CI, 0.7-0.82) and overall survival (HR, 0.76; 95% CI, 0.69-0.83), with P values less than .001 for each association.
Median progression-free survival was 5.6 months in PD-L1–positive patients and 2.8 months in PD-L1–negative patients, while median overall survival was 30 months in PD-L1–positive patients and 12.6 months in PD-L1–negative patients, the researchers reported. “The high prevalence of PD-L1 positivity observed in this study, along with the durable responses observed in PD-L1–negative tumors, suggest that pembrolizumab treatment should not be limited to patients with PD-L1–positive tumors,” they concluded. “Ongoing clinical trials with correlative studies will further delineate the role of PD-L1 expression in melanoma.”
Merck sponsored the trial. Dr. Daud disclosed ties to Merck, OncoSec, Novartis, Genentech, Bristol-Myers Squibb, and Array BioPharma.
Programmed death ligand–1 expression correlated positively and significantly with pembrolizumab response in advanced melanoma, based on an analysis of 405 patients from the international, multicohort, open-label phase I KEYNOTE-001 trial.
Among patients for whom 33%-65% of tumor cells expressed PD-L1, the objective rate of response was 57%, but the rate was only 8% among patients whose specimens did not express PD-L1, Adil Daud, MD, of the University of California, San Francisco, and associates reported. Taken together, the findings suggest that melanoma is most likely to respond to pembrolizumab when specimens show at least 10% positivity, the investigators reported (J Clin Oncol. 2016 Oct 10. doi: 10.1200/JCO.2016.67.2477).
Among the 405 patients who also were evaluable for tumor response, the overall objective response rate was 33% (95% confidence interval, 28%-37%). Grouping patients based on the melanoma scoring system for PD-L1 expression showed that PD-L1 positivity correlated significantly with the objective response rate (P less than .001). Furthermore, a higher PD-L1 melanoma score correlated significantly with both progression-free survival (hazard ratio, 0.76; 95% CI, 0.7-0.82) and overall survival (HR, 0.76; 95% CI, 0.69-0.83), with P values less than .001 for each association.
Median progression-free survival was 5.6 months in PD-L1–positive patients and 2.8 months in PD-L1–negative patients, while median overall survival was 30 months in PD-L1–positive patients and 12.6 months in PD-L1–negative patients, the researchers reported. “The high prevalence of PD-L1 positivity observed in this study, along with the durable responses observed in PD-L1–negative tumors, suggest that pembrolizumab treatment should not be limited to patients with PD-L1–positive tumors,” they concluded. “Ongoing clinical trials with correlative studies will further delineate the role of PD-L1 expression in melanoma.”
Merck sponsored the trial. Dr. Daud disclosed ties to Merck, OncoSec, Novartis, Genentech, Bristol-Myers Squibb, and Array BioPharma.
Key clinical point: PD-L1 expression correlates with pembrolizumab response in advanced melanoma.
Major finding: Higher levels of PD-L1 staining correlated positively with tumor response (P less than .001).
Data source: Analyses of 405 patients from KEYNOTE-001, an international, multicohort, open-label phase I trial of pembrolizumab in advanced melanoma.
Disclosures: Merck sponsored the trial. Dr. Daud disclosed ties to Merck, OncoSec, Novartis, Genentech, Bristol-Myers Squibb, and Array BioPharma.
Survival benefit maintained long term with ipilimumab for high-risk melanoma
COPENHAGEN – Five years on, patients with high-risk stage III melanoma treated with the checkpoint inhibitor ipilimumab, following complete resection, continue to have significantly better overall, recurrence-free, and distant metastasis–free survival, compared with patients treated with placebo, reported investigators in a phase III trial.
Five-year overall survival among patients who received a 10-mg/kg dose of ipilimumab (Yervoy) in the EORTC 18071 trial was 65.4%, compared with 54.4% for patients who received placebo. This difference translated into a hazard ratio for death with ipilimumab of 0.72 (P = .001), reported Alexander M.M. Eggermont, MD, from the Gustave Roussy Cancer Center in Villejuif, France.
The survival benefit with ipilimumab “was consistent across all survival endpoints,” he said at a briefing prior to his presentation of the data in a symposium at the European Society for Medical Oncology Congress.
He noted, however, that the 10-mg/kg dose of ipilimumab selected in phase II trials is associated with significant toxicities.
“Ipilimumab is not an easy drug to handle. My recommendation is to keep it in [cancer] centers,” he said.
In the trial, 951 patients with high-risk, stage III, completely resected melanoma were randomly assigned to receive induction therapy with ipilimumab 10 mg/kg every 3 weeks for four cycles or placebo, followed by maintenance with the assigned therapy every 12 weeks for up to 3 years.
The investigators previously reported that, at a median follow-up of 2.7 years, ipilimumab was associated with significantly prolonged overall survival (the primary endpoint), with a hazard ratio vs. placebo of 0.75 (P = .001).
At ESMO 2016, Dr. Eggermont reported final survival results from the trial, at a median follow-up of 5.3 years.
The rate of 5-year overall survival for the 475 patients assigned to ipilimumab was 65.4%, compared with 54.4% among the 476 patients assigned to placebo (HR, 0.72, P = .001).
At 5 years, 41% of patients assigned to ipilimumab were free of recurrences, compared with 30% of patients on placebo, The median recurrence-free survival was 27.6 months vs. 17.1 months, respectively (HR, 0.76, P = .0008).
Similarly, the rate of distant metastasis-free survival at 5 years was 48.3% for patients assigned to ipilimumab, vs. 38.9% in the placebo group (HR for death or distant metastasis, 0.76; P = .002).
The safety analysis showed that twice as many patients assigned to ipilimumab had grade 3 or 4 adverse events (54.1% vs. 26.2%). Grade 3 or 4 adverse immune events occurred in 41.6% vs. 2.7%, respectively.
Five patients assigned to ipilimumab died from immune-related causes: three from colitis (two of whom had intestinal perforations), one from myocarditis, and one from multiorgan failure associated with Guillain-Barré syndrome.
“The final analysis shows, for the first time, that checkpoint blockade is effective in the adjuvant setting,” he said.
The data suggest, however, that the benefit appears to be concentrated in patients with higher-risk features, such as involvement of four or more lymph nodes, microscopic nodal disease, or ulceration, he said.
The discussant also agreed with Dr. Eggermont’s assertions that the decision to treat patients with ipilimumab should factor in toxicity, and that treatment should be administered only in centers experienced in using the drug.
The trial was sponsored by Bristol-Myers Squibb. Dr. Eggermont disclosed serving on an advisory board for Bristol-Myers Squibb and Merck. Dr. Michielin disclosed consulting and/or honoraria from Amgen, Bristol-Myers Squibb, Roche, Merck Sharp & Dohme, Novartis, and GlaxoSmithKline.
COPENHAGEN – Five years on, patients with high-risk stage III melanoma treated with the checkpoint inhibitor ipilimumab, following complete resection, continue to have significantly better overall, recurrence-free, and distant metastasis–free survival, compared with patients treated with placebo, reported investigators in a phase III trial.
Five-year overall survival among patients who received a 10-mg/kg dose of ipilimumab (Yervoy) in the EORTC 18071 trial was 65.4%, compared with 54.4% for patients who received placebo. This difference translated into a hazard ratio for death with ipilimumab of 0.72 (P = .001), reported Alexander M.M. Eggermont, MD, from the Gustave Roussy Cancer Center in Villejuif, France.
The survival benefit with ipilimumab “was consistent across all survival endpoints,” he said at a briefing prior to his presentation of the data in a symposium at the European Society for Medical Oncology Congress.
He noted, however, that the 10-mg/kg dose of ipilimumab selected in phase II trials is associated with significant toxicities.
“Ipilimumab is not an easy drug to handle. My recommendation is to keep it in [cancer] centers,” he said.
In the trial, 951 patients with high-risk, stage III, completely resected melanoma were randomly assigned to receive induction therapy with ipilimumab 10 mg/kg every 3 weeks for four cycles or placebo, followed by maintenance with the assigned therapy every 12 weeks for up to 3 years.
The investigators previously reported that, at a median follow-up of 2.7 years, ipilimumab was associated with significantly prolonged overall survival (the primary endpoint), with a hazard ratio vs. placebo of 0.75 (P = .001).
At ESMO 2016, Dr. Eggermont reported final survival results from the trial, at a median follow-up of 5.3 years.
The rate of 5-year overall survival for the 475 patients assigned to ipilimumab was 65.4%, compared with 54.4% among the 476 patients assigned to placebo (HR, 0.72, P = .001).
At 5 years, 41% of patients assigned to ipilimumab were free of recurrences, compared with 30% of patients on placebo, The median recurrence-free survival was 27.6 months vs. 17.1 months, respectively (HR, 0.76, P = .0008).
Similarly, the rate of distant metastasis-free survival at 5 years was 48.3% for patients assigned to ipilimumab, vs. 38.9% in the placebo group (HR for death or distant metastasis, 0.76; P = .002).
The safety analysis showed that twice as many patients assigned to ipilimumab had grade 3 or 4 adverse events (54.1% vs. 26.2%). Grade 3 or 4 adverse immune events occurred in 41.6% vs. 2.7%, respectively.
Five patients assigned to ipilimumab died from immune-related causes: three from colitis (two of whom had intestinal perforations), one from myocarditis, and one from multiorgan failure associated with Guillain-Barré syndrome.
“The final analysis shows, for the first time, that checkpoint blockade is effective in the adjuvant setting,” he said.
The data suggest, however, that the benefit appears to be concentrated in patients with higher-risk features, such as involvement of four or more lymph nodes, microscopic nodal disease, or ulceration, he said.
The discussant also agreed with Dr. Eggermont’s assertions that the decision to treat patients with ipilimumab should factor in toxicity, and that treatment should be administered only in centers experienced in using the drug.
The trial was sponsored by Bristol-Myers Squibb. Dr. Eggermont disclosed serving on an advisory board for Bristol-Myers Squibb and Merck. Dr. Michielin disclosed consulting and/or honoraria from Amgen, Bristol-Myers Squibb, Roche, Merck Sharp & Dohme, Novartis, and GlaxoSmithKline.
COPENHAGEN – Five years on, patients with high-risk stage III melanoma treated with the checkpoint inhibitor ipilimumab, following complete resection, continue to have significantly better overall, recurrence-free, and distant metastasis–free survival, compared with patients treated with placebo, reported investigators in a phase III trial.
Five-year overall survival among patients who received a 10-mg/kg dose of ipilimumab (Yervoy) in the EORTC 18071 trial was 65.4%, compared with 54.4% for patients who received placebo. This difference translated into a hazard ratio for death with ipilimumab of 0.72 (P = .001), reported Alexander M.M. Eggermont, MD, from the Gustave Roussy Cancer Center in Villejuif, France.
The survival benefit with ipilimumab “was consistent across all survival endpoints,” he said at a briefing prior to his presentation of the data in a symposium at the European Society for Medical Oncology Congress.
He noted, however, that the 10-mg/kg dose of ipilimumab selected in phase II trials is associated with significant toxicities.
“Ipilimumab is not an easy drug to handle. My recommendation is to keep it in [cancer] centers,” he said.
In the trial, 951 patients with high-risk, stage III, completely resected melanoma were randomly assigned to receive induction therapy with ipilimumab 10 mg/kg every 3 weeks for four cycles or placebo, followed by maintenance with the assigned therapy every 12 weeks for up to 3 years.
The investigators previously reported that, at a median follow-up of 2.7 years, ipilimumab was associated with significantly prolonged overall survival (the primary endpoint), with a hazard ratio vs. placebo of 0.75 (P = .001).
At ESMO 2016, Dr. Eggermont reported final survival results from the trial, at a median follow-up of 5.3 years.
The rate of 5-year overall survival for the 475 patients assigned to ipilimumab was 65.4%, compared with 54.4% among the 476 patients assigned to placebo (HR, 0.72, P = .001).
At 5 years, 41% of patients assigned to ipilimumab were free of recurrences, compared with 30% of patients on placebo, The median recurrence-free survival was 27.6 months vs. 17.1 months, respectively (HR, 0.76, P = .0008).
Similarly, the rate of distant metastasis-free survival at 5 years was 48.3% for patients assigned to ipilimumab, vs. 38.9% in the placebo group (HR for death or distant metastasis, 0.76; P = .002).
The safety analysis showed that twice as many patients assigned to ipilimumab had grade 3 or 4 adverse events (54.1% vs. 26.2%). Grade 3 or 4 adverse immune events occurred in 41.6% vs. 2.7%, respectively.
Five patients assigned to ipilimumab died from immune-related causes: three from colitis (two of whom had intestinal perforations), one from myocarditis, and one from multiorgan failure associated with Guillain-Barré syndrome.
“The final analysis shows, for the first time, that checkpoint blockade is effective in the adjuvant setting,” he said.
The data suggest, however, that the benefit appears to be concentrated in patients with higher-risk features, such as involvement of four or more lymph nodes, microscopic nodal disease, or ulceration, he said.
The discussant also agreed with Dr. Eggermont’s assertions that the decision to treat patients with ipilimumab should factor in toxicity, and that treatment should be administered only in centers experienced in using the drug.
The trial was sponsored by Bristol-Myers Squibb. Dr. Eggermont disclosed serving on an advisory board for Bristol-Myers Squibb and Merck. Dr. Michielin disclosed consulting and/or honoraria from Amgen, Bristol-Myers Squibb, Roche, Merck Sharp & Dohme, Novartis, and GlaxoSmithKline.
Key clinical point: The CTLA-4 checkpoint inhibitor ipilimumab offers survival benefit, compared with placebo in patients with malignant melanoma.
Major finding: The hazard ratio for death with ipilimumab vs. placebo was 0.72 (P = .001).
Data source: Randomized, controlled, phase III trial in 951 patients with high-risk stage III malignant melanoma following complete resection.
Disclosures: The trial was sponsored by Bristol-Myers Squibb. Dr. Eggermont disclosed serving on an advisory board for Bristol-Myers Squibb and Merck. Dr. Michielin disclosed consulting and/or honoraria from Amgen, Bristol-Myers Squibb, Roche, Merck Sharp & Dohme, Novartis, and GlaxoSmithKline.
Exploration of Modern Military Research Resources
Advances in medical biotechnologies, data-gathering techniques, and -omics technologies have resulted in the broader understanding of disease pathology and treatment and have facilitated the individualization of health care plans to meet the unique needs of each patient. Military medicine often has been on the forefront of medical technology, disease understanding, and clinical care both on and off the battlefield, in large part due to the unique resources available in the military health care system. These resources allow investigators the ability to integrate vast amounts of epidemiologic data with an extensive biological sample database of its service members, which in the modern age has translated into advances in the understanding of melanoma and the treatment of scars.
History of Research in the Military
Starting in the 1950s, the US Department of Defense (DoD) started to collect serum samples of its service members for the purpose of research.1 It was not until 1985 that the DoD implemented a long-term frozen storage system for serum samples obtained through mandatory screening for human immunodeficiency virus (HIV) in service members.2 Subsequently, the Department of Defense Serum Repository (DoDSR) was officially established in 1989 as a central archive for the long-term storage of serum obtained from active-duty and reserve service members in the US Navy, Army, and Marines.2,3 In the mid-1990s, the DoDSR expanded its capabilities to include the storage of serum samples from all military members, including the US Air Force, obtained predeployment and postdeployment.3,4 At that time, a records-keeping system was established, now known as the Defense Medical Surveillance System (DMSS). The creation of the DMSS provided an extensive epidemiologic database that provided valuable information such as demographic data, service records, deployment data, reportable medical events, exposure history, and vaccination records, which could be linked to the serum samples of each service member.2-4 Since 2008, the responsibilities of maintaining the DoDSR and the DMSS were transferred to the Armed Forces Health Surveillance Center (AFHSC).5
There have been several other databases created over the years that provide additional support and resources to military investigators. The Automated Central Tumor Registry and Department of Pathology and Area Laboratory Services both help investigators to track the incidence of specific cancers in the military population and provide them with pathologic specimens. Additionally, electronic medical records including the composite health care system and the Armed Forces Health Longitudinal Technology Application supplemented with insurance claims data accessible from the Military Health System Management and Reporting Tool (M2) database have made it possible to track patient data.
Utilization of Military Research Resources
Today, the DoDSR is a secure facility that maintains more than 56 million serum specimens from more than 11 million individuals in –30°C freezers, making it one of the largest repositories in the world.3,6 Each serum sample is linked with an individual’s DMSS record, providing a way for investigators to study how external factors such as deployment history, occupation, and exposure history relate to an individual’s unique genetic and physiological makeup. Furthermore, these data can be used for seroepidemiologic investigations that contribute to all facets of clinical care. The AFHSC routinely publishes findings related to notifiable diseases, disease outbreaks, and disease trends in a monthly report.7
There are strict guidelines in place that limit access to the DoDSR and service members’ data. Use of the repository for information directly related to a patient’s health care is one reason for access, such as analyzing serum for antibodies and seroconversion to assist in the diagnosis of a disease such as HIV. Another reason would be to obtain information needed for criminal investigations and prosecution. Typically, these types of requests require a judge-issued court order and approval by the Assistant Secretary of Defense for Health Affairs.4 The DoDSR also is used to study force health protection issues, such as infectious disease incidence and disease prevalence in the military population.
Obtaining access to the DoDSR and service members’ data for research purposes requires that the principal investigator be a DoD employee. Each research proposal is reviewed by members of the AFHSC to determine if the DoDSR is able to meet the demands of the project, including having the appropriate number of serum samples and supporting epidemiologic data available. The AFHSC provides a letter of support if it deems the project to be in line with its current resources and capabilities. Each research proposal is then sent to an institutional review board (IRB) to determine if the study is exempt or needs to go through a full IRB review process. A study might be exempt if the investigators are not obtaining data through interaction with living individuals or not having access to any identifiable protected health information associated with the samples.6 Regardless of whether the study is exempt or not exempt, the AFHSC will de-identify each sample before releasing the samples to the investigators by using a coding system to shield the patient’s identity from the investigator.
Resources within the military medical research system provide investigators with access to an extensive biorepository of serum and linked epidemiological data. Samples from the DoDSR have been used in no less than 75 peer-reviewed publications since 1985.8,9 Several of these studies have been influential in expanding knowledge about conditions seen more commonly in the military population such as stress fractures, traumatic brain injuries, posttraumatic stress disorder, and suicide.8 Additionally, DoDSR samples have been used to form military vaccination policies and track both infectious and noninfectious conditions in the military; for example, during the H1N1 influenza virus outbreak of 2009, AFHSC was essential in helping to limit the spread of the virus within the military community by using its data and collaborating with groups such as the Centers for Disease Control and Prevention to develop a plan for disease surveillance and control.5
Several military research resources are currently being used for a melanoma study that aims to assess if specific phenotypic features, melanoma risk alleles, and environmental factors (eg, duty station location, occupation, amount of UV exposure) can be used to develop better screening models to identify individuals who are at risk for developing melanoma. Secondarily, the study aims to determine if recently developed multimarker diagnostic and prognostic assays for melanoma will prove useful in the diagnostic and prognostic assessment of melanocytic neoplasms in the military population. For this study, one of the authors (J.H.M) is utilizing DoDSR serum from 1700 retrospective cases of invasive melanoma and 1700 matched controls. Additionally, the Automated Central Tumor Registry and Department of Pathology and Area Laboratory Services databases are being used to obtain tissue from more than 300 melanoma cases and nevi controls.
Limitations of the Current System
Despite the impressive capabilities of the current system, there are some issues that limit its potential. One such limitation is associated with the way that the serum samples at the DoDSR are utilized. Through 2012, the DoDSR had 54,542,658 serum specimens available, of which only 228,610 (0.42%) had ever been accessed for study.8 With such a wealth of information and relative availability, why are the serum samples not being accessed more frequently for studies? The inherent nature of the DoDSR being a restricted facility and only accessible to DoD-affiliated investigators may contribute, which allows the DoDSR to fulfill its primary purpose of contributing to military-relevant investigations but at the same time limits the number and type of investigations that can be performed. One idea that has been proposed is allowing civilian investigator access to the DoDSR if it can be proven that the research is targeted toward military-relevant issues.8 However, the current AFHSC access guidelines would need revision and would require additional safeguards to ensure that military-protected health information is not compromised. Nonetheless, such a change may result in more extensive use of DoDSR resources in the future.
An ethical issue that needs to be addressed pertains to how the DoDSR permits use of human serum samples for research purposes without getting consent from the individuals being studied. The serum samples are collected as part of mandatory predeployment and postdeployment examinations for HIV screening of all military members. These individuals are not informed of potential use of their serum specimens for research purposes and no consent forms or opt-out options are provided. Although it is true that military members must comply with specific requirements pertaining to military readiness (eg, receiving appropriate vaccinations, drug testing, regular medical screening), it is debated whether they still retain the right as patients to refuse participating in research and clinical trials.10 The AFHSC does have several regulatory steps in place to ensure that military members’ samples are used in an appropriate manner, including requiring a DoD primary investigator, IRB review of every research proposal, and de-identification of samples. At a minimum, giving military members the ability to provide informed consent would ensure that the military system is adhering to evolving human research standards.
The current lack of biological specimens other than serum in the DoDSR is another limitation of the current system. Recent advances in molecular analyses are impacted by expanding -omics techniques, such as epigenomics, transcriptomics, and proteomics. The field of epigenomics is the study of reversible changes to DNA (eg, methylation) associated with specific disease states or following specific environmental exposures.9,11 Transcriptomics, which analyzes messenger RNA transcript levels of expressed genes, and proteomics, which uses expression of proteins, are 2 techniques being used to develop biomarkers associated with specific diseases and environmental exposures.9,11 Serum alone does not provide the high-quality nucleic acids needed for many of these studies to take place. Adding whole-blood specimens or blood spot samples of military service members to the DoDSR would allow researchers to use these techniques to investigate many new biomarkers associated with military-relevant diseases and exposures. These techniques also can be used in the expanding field of personalized medicine so that health care providers are able to tailor all phases of care, including diagnosis and treatment, to an individual’s genetic profile.
Conclusion
The history of research in military medicine has been built on achieving the primary goal of serving those men and women who put their lives in danger to protect this country. In an evolving environment of new technologies that have led to changes in service members’ injuries, exposures, and diseases, military medicine also must adapt. Resources such as the DoDSR and DMSS, which provide investigators with the unique ability to link epidemiological data with serum samples, have been invaluable contributors to this overall mission. As with any large system, there are always improvements that can be made. Improving access to the DoDSR serum samples, educating and obtaining consent from military service members to use their samples in research, and adding specimens to the DoDSR that can be used for -omics techniques are 3 changes that should be considered to maximize
- Liao SJ. Immunity status of military recruits in 1951 in the United States. I. results of Schick tests. Am J Hyg. 1954;59:262-272.
- Rubertone MV, Brundage JF. The defense medical surveillance system and the department of defense serum repository: glimpses of the future of public health surveillance. Am J Public Health. 2002;92:1900-1904.
- Department of Defense Serum Repository. Military Health System and the Defense Health Agency website. http://www.health.mil/Military-Health-Topics/Health-Readiness/Armed-Forces-Health-Surveillance-Branch/Data-Management-and-Technical-Support/Department-of-Defense-Serum-Repository. Accessed August 2, 2016.
- Perdue CL, Eick-Cost AA, Rubertone MV. A brief description of the operation of the DoD serum repository. Mil Med. 2015;180(10 suppl):10-12.
- DeFraites RF. The Armed Forces Health Surveillance Center: enhancing the Military Health System’s public health capabilities. BMC Public Health. 2011;11(suppl 2):S1.
- Pavlin JA, Welch RA. Ethics, human use, and the department of defense serum repository. Mil Med. 2015;180:49-56.
- Defense Medical Surveillance System. Military Health System and the Defense Health Agency website. http://www.health.mil/Military-Health-Topics/Health-Readiness/Armed-Forces-Health-Surveillance-Branch/Data-Management-and-Technical-Support/Defense-Medical-Surveillance-System. Accessed August 2, 2016.
- Perdue CL, Eick-Cost AA, Rubertone MV, et al. Description and utilization of the United States Department of Defense Serum Repository: a review of published studies, 1985-2012. Plos One. 2015;10:1-16.
- Mancuso JD, Mallon TM, Gaydos JC. Maximizing the capabilities of the DoD serum repository to meet current and future needs: report of the needs panel. Mil Med. 2015;180:14-24.
- Department of Defense. Department of Defense Instruction. http://www.dtic.mil/whs/directives/corres/pdf/600014p.pdf. Posted September 26, 2001. Updated October 3, 2013. Accessed August 2, 2016.
- Lindler LE. Building a DoD biorepository for the future: potential benefits and way forward. Mil Med. 2015;180:90-94.
Advances in medical biotechnologies, data-gathering techniques, and -omics technologies have resulted in the broader understanding of disease pathology and treatment and have facilitated the individualization of health care plans to meet the unique needs of each patient. Military medicine often has been on the forefront of medical technology, disease understanding, and clinical care both on and off the battlefield, in large part due to the unique resources available in the military health care system. These resources allow investigators the ability to integrate vast amounts of epidemiologic data with an extensive biological sample database of its service members, which in the modern age has translated into advances in the understanding of melanoma and the treatment of scars.
History of Research in the Military
Starting in the 1950s, the US Department of Defense (DoD) started to collect serum samples of its service members for the purpose of research.1 It was not until 1985 that the DoD implemented a long-term frozen storage system for serum samples obtained through mandatory screening for human immunodeficiency virus (HIV) in service members.2 Subsequently, the Department of Defense Serum Repository (DoDSR) was officially established in 1989 as a central archive for the long-term storage of serum obtained from active-duty and reserve service members in the US Navy, Army, and Marines.2,3 In the mid-1990s, the DoDSR expanded its capabilities to include the storage of serum samples from all military members, including the US Air Force, obtained predeployment and postdeployment.3,4 At that time, a records-keeping system was established, now known as the Defense Medical Surveillance System (DMSS). The creation of the DMSS provided an extensive epidemiologic database that provided valuable information such as demographic data, service records, deployment data, reportable medical events, exposure history, and vaccination records, which could be linked to the serum samples of each service member.2-4 Since 2008, the responsibilities of maintaining the DoDSR and the DMSS were transferred to the Armed Forces Health Surveillance Center (AFHSC).5
There have been several other databases created over the years that provide additional support and resources to military investigators. The Automated Central Tumor Registry and Department of Pathology and Area Laboratory Services both help investigators to track the incidence of specific cancers in the military population and provide them with pathologic specimens. Additionally, electronic medical records including the composite health care system and the Armed Forces Health Longitudinal Technology Application supplemented with insurance claims data accessible from the Military Health System Management and Reporting Tool (M2) database have made it possible to track patient data.
Utilization of Military Research Resources
Today, the DoDSR is a secure facility that maintains more than 56 million serum specimens from more than 11 million individuals in –30°C freezers, making it one of the largest repositories in the world.3,6 Each serum sample is linked with an individual’s DMSS record, providing a way for investigators to study how external factors such as deployment history, occupation, and exposure history relate to an individual’s unique genetic and physiological makeup. Furthermore, these data can be used for seroepidemiologic investigations that contribute to all facets of clinical care. The AFHSC routinely publishes findings related to notifiable diseases, disease outbreaks, and disease trends in a monthly report.7
There are strict guidelines in place that limit access to the DoDSR and service members’ data. Use of the repository for information directly related to a patient’s health care is one reason for access, such as analyzing serum for antibodies and seroconversion to assist in the diagnosis of a disease such as HIV. Another reason would be to obtain information needed for criminal investigations and prosecution. Typically, these types of requests require a judge-issued court order and approval by the Assistant Secretary of Defense for Health Affairs.4 The DoDSR also is used to study force health protection issues, such as infectious disease incidence and disease prevalence in the military population.
Obtaining access to the DoDSR and service members’ data for research purposes requires that the principal investigator be a DoD employee. Each research proposal is reviewed by members of the AFHSC to determine if the DoDSR is able to meet the demands of the project, including having the appropriate number of serum samples and supporting epidemiologic data available. The AFHSC provides a letter of support if it deems the project to be in line with its current resources and capabilities. Each research proposal is then sent to an institutional review board (IRB) to determine if the study is exempt or needs to go through a full IRB review process. A study might be exempt if the investigators are not obtaining data through interaction with living individuals or not having access to any identifiable protected health information associated with the samples.6 Regardless of whether the study is exempt or not exempt, the AFHSC will de-identify each sample before releasing the samples to the investigators by using a coding system to shield the patient’s identity from the investigator.
Resources within the military medical research system provide investigators with access to an extensive biorepository of serum and linked epidemiological data. Samples from the DoDSR have been used in no less than 75 peer-reviewed publications since 1985.8,9 Several of these studies have been influential in expanding knowledge about conditions seen more commonly in the military population such as stress fractures, traumatic brain injuries, posttraumatic stress disorder, and suicide.8 Additionally, DoDSR samples have been used to form military vaccination policies and track both infectious and noninfectious conditions in the military; for example, during the H1N1 influenza virus outbreak of 2009, AFHSC was essential in helping to limit the spread of the virus within the military community by using its data and collaborating with groups such as the Centers for Disease Control and Prevention to develop a plan for disease surveillance and control.5
Several military research resources are currently being used for a melanoma study that aims to assess if specific phenotypic features, melanoma risk alleles, and environmental factors (eg, duty station location, occupation, amount of UV exposure) can be used to develop better screening models to identify individuals who are at risk for developing melanoma. Secondarily, the study aims to determine if recently developed multimarker diagnostic and prognostic assays for melanoma will prove useful in the diagnostic and prognostic assessment of melanocytic neoplasms in the military population. For this study, one of the authors (J.H.M) is utilizing DoDSR serum from 1700 retrospective cases of invasive melanoma and 1700 matched controls. Additionally, the Automated Central Tumor Registry and Department of Pathology and Area Laboratory Services databases are being used to obtain tissue from more than 300 melanoma cases and nevi controls.
Limitations of the Current System
Despite the impressive capabilities of the current system, there are some issues that limit its potential. One such limitation is associated with the way that the serum samples at the DoDSR are utilized. Through 2012, the DoDSR had 54,542,658 serum specimens available, of which only 228,610 (0.42%) had ever been accessed for study.8 With such a wealth of information and relative availability, why are the serum samples not being accessed more frequently for studies? The inherent nature of the DoDSR being a restricted facility and only accessible to DoD-affiliated investigators may contribute, which allows the DoDSR to fulfill its primary purpose of contributing to military-relevant investigations but at the same time limits the number and type of investigations that can be performed. One idea that has been proposed is allowing civilian investigator access to the DoDSR if it can be proven that the research is targeted toward military-relevant issues.8 However, the current AFHSC access guidelines would need revision and would require additional safeguards to ensure that military-protected health information is not compromised. Nonetheless, such a change may result in more extensive use of DoDSR resources in the future.
An ethical issue that needs to be addressed pertains to how the DoDSR permits use of human serum samples for research purposes without getting consent from the individuals being studied. The serum samples are collected as part of mandatory predeployment and postdeployment examinations for HIV screening of all military members. These individuals are not informed of potential use of their serum specimens for research purposes and no consent forms or opt-out options are provided. Although it is true that military members must comply with specific requirements pertaining to military readiness (eg, receiving appropriate vaccinations, drug testing, regular medical screening), it is debated whether they still retain the right as patients to refuse participating in research and clinical trials.10 The AFHSC does have several regulatory steps in place to ensure that military members’ samples are used in an appropriate manner, including requiring a DoD primary investigator, IRB review of every research proposal, and de-identification of samples. At a minimum, giving military members the ability to provide informed consent would ensure that the military system is adhering to evolving human research standards.
The current lack of biological specimens other than serum in the DoDSR is another limitation of the current system. Recent advances in molecular analyses are impacted by expanding -omics techniques, such as epigenomics, transcriptomics, and proteomics. The field of epigenomics is the study of reversible changes to DNA (eg, methylation) associated with specific disease states or following specific environmental exposures.9,11 Transcriptomics, which analyzes messenger RNA transcript levels of expressed genes, and proteomics, which uses expression of proteins, are 2 techniques being used to develop biomarkers associated with specific diseases and environmental exposures.9,11 Serum alone does not provide the high-quality nucleic acids needed for many of these studies to take place. Adding whole-blood specimens or blood spot samples of military service members to the DoDSR would allow researchers to use these techniques to investigate many new biomarkers associated with military-relevant diseases and exposures. These techniques also can be used in the expanding field of personalized medicine so that health care providers are able to tailor all phases of care, including diagnosis and treatment, to an individual’s genetic profile.
Conclusion
The history of research in military medicine has been built on achieving the primary goal of serving those men and women who put their lives in danger to protect this country. In an evolving environment of new technologies that have led to changes in service members’ injuries, exposures, and diseases, military medicine also must adapt. Resources such as the DoDSR and DMSS, which provide investigators with the unique ability to link epidemiological data with serum samples, have been invaluable contributors to this overall mission. As with any large system, there are always improvements that can be made. Improving access to the DoDSR serum samples, educating and obtaining consent from military service members to use their samples in research, and adding specimens to the DoDSR that can be used for -omics techniques are 3 changes that should be considered to maximize
Advances in medical biotechnologies, data-gathering techniques, and -omics technologies have resulted in the broader understanding of disease pathology and treatment and have facilitated the individualization of health care plans to meet the unique needs of each patient. Military medicine often has been on the forefront of medical technology, disease understanding, and clinical care both on and off the battlefield, in large part due to the unique resources available in the military health care system. These resources allow investigators the ability to integrate vast amounts of epidemiologic data with an extensive biological sample database of its service members, which in the modern age has translated into advances in the understanding of melanoma and the treatment of scars.
History of Research in the Military
Starting in the 1950s, the US Department of Defense (DoD) started to collect serum samples of its service members for the purpose of research.1 It was not until 1985 that the DoD implemented a long-term frozen storage system for serum samples obtained through mandatory screening for human immunodeficiency virus (HIV) in service members.2 Subsequently, the Department of Defense Serum Repository (DoDSR) was officially established in 1989 as a central archive for the long-term storage of serum obtained from active-duty and reserve service members in the US Navy, Army, and Marines.2,3 In the mid-1990s, the DoDSR expanded its capabilities to include the storage of serum samples from all military members, including the US Air Force, obtained predeployment and postdeployment.3,4 At that time, a records-keeping system was established, now known as the Defense Medical Surveillance System (DMSS). The creation of the DMSS provided an extensive epidemiologic database that provided valuable information such as demographic data, service records, deployment data, reportable medical events, exposure history, and vaccination records, which could be linked to the serum samples of each service member.2-4 Since 2008, the responsibilities of maintaining the DoDSR and the DMSS were transferred to the Armed Forces Health Surveillance Center (AFHSC).5
There have been several other databases created over the years that provide additional support and resources to military investigators. The Automated Central Tumor Registry and Department of Pathology and Area Laboratory Services both help investigators to track the incidence of specific cancers in the military population and provide them with pathologic specimens. Additionally, electronic medical records including the composite health care system and the Armed Forces Health Longitudinal Technology Application supplemented with insurance claims data accessible from the Military Health System Management and Reporting Tool (M2) database have made it possible to track patient data.
Utilization of Military Research Resources
Today, the DoDSR is a secure facility that maintains more than 56 million serum specimens from more than 11 million individuals in –30°C freezers, making it one of the largest repositories in the world.3,6 Each serum sample is linked with an individual’s DMSS record, providing a way for investigators to study how external factors such as deployment history, occupation, and exposure history relate to an individual’s unique genetic and physiological makeup. Furthermore, these data can be used for seroepidemiologic investigations that contribute to all facets of clinical care. The AFHSC routinely publishes findings related to notifiable diseases, disease outbreaks, and disease trends in a monthly report.7
There are strict guidelines in place that limit access to the DoDSR and service members’ data. Use of the repository for information directly related to a patient’s health care is one reason for access, such as analyzing serum for antibodies and seroconversion to assist in the diagnosis of a disease such as HIV. Another reason would be to obtain information needed for criminal investigations and prosecution. Typically, these types of requests require a judge-issued court order and approval by the Assistant Secretary of Defense for Health Affairs.4 The DoDSR also is used to study force health protection issues, such as infectious disease incidence and disease prevalence in the military population.
Obtaining access to the DoDSR and service members’ data for research purposes requires that the principal investigator be a DoD employee. Each research proposal is reviewed by members of the AFHSC to determine if the DoDSR is able to meet the demands of the project, including having the appropriate number of serum samples and supporting epidemiologic data available. The AFHSC provides a letter of support if it deems the project to be in line with its current resources and capabilities. Each research proposal is then sent to an institutional review board (IRB) to determine if the study is exempt or needs to go through a full IRB review process. A study might be exempt if the investigators are not obtaining data through interaction with living individuals or not having access to any identifiable protected health information associated with the samples.6 Regardless of whether the study is exempt or not exempt, the AFHSC will de-identify each sample before releasing the samples to the investigators by using a coding system to shield the patient’s identity from the investigator.
Resources within the military medical research system provide investigators with access to an extensive biorepository of serum and linked epidemiological data. Samples from the DoDSR have been used in no less than 75 peer-reviewed publications since 1985.8,9 Several of these studies have been influential in expanding knowledge about conditions seen more commonly in the military population such as stress fractures, traumatic brain injuries, posttraumatic stress disorder, and suicide.8 Additionally, DoDSR samples have been used to form military vaccination policies and track both infectious and noninfectious conditions in the military; for example, during the H1N1 influenza virus outbreak of 2009, AFHSC was essential in helping to limit the spread of the virus within the military community by using its data and collaborating with groups such as the Centers for Disease Control and Prevention to develop a plan for disease surveillance and control.5
Several military research resources are currently being used for a melanoma study that aims to assess if specific phenotypic features, melanoma risk alleles, and environmental factors (eg, duty station location, occupation, amount of UV exposure) can be used to develop better screening models to identify individuals who are at risk for developing melanoma. Secondarily, the study aims to determine if recently developed multimarker diagnostic and prognostic assays for melanoma will prove useful in the diagnostic and prognostic assessment of melanocytic neoplasms in the military population. For this study, one of the authors (J.H.M) is utilizing DoDSR serum from 1700 retrospective cases of invasive melanoma and 1700 matched controls. Additionally, the Automated Central Tumor Registry and Department of Pathology and Area Laboratory Services databases are being used to obtain tissue from more than 300 melanoma cases and nevi controls.
Limitations of the Current System
Despite the impressive capabilities of the current system, there are some issues that limit its potential. One such limitation is associated with the way that the serum samples at the DoDSR are utilized. Through 2012, the DoDSR had 54,542,658 serum specimens available, of which only 228,610 (0.42%) had ever been accessed for study.8 With such a wealth of information and relative availability, why are the serum samples not being accessed more frequently for studies? The inherent nature of the DoDSR being a restricted facility and only accessible to DoD-affiliated investigators may contribute, which allows the DoDSR to fulfill its primary purpose of contributing to military-relevant investigations but at the same time limits the number and type of investigations that can be performed. One idea that has been proposed is allowing civilian investigator access to the DoDSR if it can be proven that the research is targeted toward military-relevant issues.8 However, the current AFHSC access guidelines would need revision and would require additional safeguards to ensure that military-protected health information is not compromised. Nonetheless, such a change may result in more extensive use of DoDSR resources in the future.
An ethical issue that needs to be addressed pertains to how the DoDSR permits use of human serum samples for research purposes without getting consent from the individuals being studied. The serum samples are collected as part of mandatory predeployment and postdeployment examinations for HIV screening of all military members. These individuals are not informed of potential use of their serum specimens for research purposes and no consent forms or opt-out options are provided. Although it is true that military members must comply with specific requirements pertaining to military readiness (eg, receiving appropriate vaccinations, drug testing, regular medical screening), it is debated whether they still retain the right as patients to refuse participating in research and clinical trials.10 The AFHSC does have several regulatory steps in place to ensure that military members’ samples are used in an appropriate manner, including requiring a DoD primary investigator, IRB review of every research proposal, and de-identification of samples. At a minimum, giving military members the ability to provide informed consent would ensure that the military system is adhering to evolving human research standards.
The current lack of biological specimens other than serum in the DoDSR is another limitation of the current system. Recent advances in molecular analyses are impacted by expanding -omics techniques, such as epigenomics, transcriptomics, and proteomics. The field of epigenomics is the study of reversible changes to DNA (eg, methylation) associated with specific disease states or following specific environmental exposures.9,11 Transcriptomics, which analyzes messenger RNA transcript levels of expressed genes, and proteomics, which uses expression of proteins, are 2 techniques being used to develop biomarkers associated with specific diseases and environmental exposures.9,11 Serum alone does not provide the high-quality nucleic acids needed for many of these studies to take place. Adding whole-blood specimens or blood spot samples of military service members to the DoDSR would allow researchers to use these techniques to investigate many new biomarkers associated with military-relevant diseases and exposures. These techniques also can be used in the expanding field of personalized medicine so that health care providers are able to tailor all phases of care, including diagnosis and treatment, to an individual’s genetic profile.
Conclusion
The history of research in military medicine has been built on achieving the primary goal of serving those men and women who put their lives in danger to protect this country. In an evolving environment of new technologies that have led to changes in service members’ injuries, exposures, and diseases, military medicine also must adapt. Resources such as the DoDSR and DMSS, which provide investigators with the unique ability to link epidemiological data with serum samples, have been invaluable contributors to this overall mission. As with any large system, there are always improvements that can be made. Improving access to the DoDSR serum samples, educating and obtaining consent from military service members to use their samples in research, and adding specimens to the DoDSR that can be used for -omics techniques are 3 changes that should be considered to maximize
- Liao SJ. Immunity status of military recruits in 1951 in the United States. I. results of Schick tests. Am J Hyg. 1954;59:262-272.
- Rubertone MV, Brundage JF. The defense medical surveillance system and the department of defense serum repository: glimpses of the future of public health surveillance. Am J Public Health. 2002;92:1900-1904.
- Department of Defense Serum Repository. Military Health System and the Defense Health Agency website. http://www.health.mil/Military-Health-Topics/Health-Readiness/Armed-Forces-Health-Surveillance-Branch/Data-Management-and-Technical-Support/Department-of-Defense-Serum-Repository. Accessed August 2, 2016.
- Perdue CL, Eick-Cost AA, Rubertone MV. A brief description of the operation of the DoD serum repository. Mil Med. 2015;180(10 suppl):10-12.
- DeFraites RF. The Armed Forces Health Surveillance Center: enhancing the Military Health System’s public health capabilities. BMC Public Health. 2011;11(suppl 2):S1.
- Pavlin JA, Welch RA. Ethics, human use, and the department of defense serum repository. Mil Med. 2015;180:49-56.
- Defense Medical Surveillance System. Military Health System and the Defense Health Agency website. http://www.health.mil/Military-Health-Topics/Health-Readiness/Armed-Forces-Health-Surveillance-Branch/Data-Management-and-Technical-Support/Defense-Medical-Surveillance-System. Accessed August 2, 2016.
- Perdue CL, Eick-Cost AA, Rubertone MV, et al. Description and utilization of the United States Department of Defense Serum Repository: a review of published studies, 1985-2012. Plos One. 2015;10:1-16.
- Mancuso JD, Mallon TM, Gaydos JC. Maximizing the capabilities of the DoD serum repository to meet current and future needs: report of the needs panel. Mil Med. 2015;180:14-24.
- Department of Defense. Department of Defense Instruction. http://www.dtic.mil/whs/directives/corres/pdf/600014p.pdf. Posted September 26, 2001. Updated October 3, 2013. Accessed August 2, 2016.
- Lindler LE. Building a DoD biorepository for the future: potential benefits and way forward. Mil Med. 2015;180:90-94.
- Liao SJ. Immunity status of military recruits in 1951 in the United States. I. results of Schick tests. Am J Hyg. 1954;59:262-272.
- Rubertone MV, Brundage JF. The defense medical surveillance system and the department of defense serum repository: glimpses of the future of public health surveillance. Am J Public Health. 2002;92:1900-1904.
- Department of Defense Serum Repository. Military Health System and the Defense Health Agency website. http://www.health.mil/Military-Health-Topics/Health-Readiness/Armed-Forces-Health-Surveillance-Branch/Data-Management-and-Technical-Support/Department-of-Defense-Serum-Repository. Accessed August 2, 2016.
- Perdue CL, Eick-Cost AA, Rubertone MV. A brief description of the operation of the DoD serum repository. Mil Med. 2015;180(10 suppl):10-12.
- DeFraites RF. The Armed Forces Health Surveillance Center: enhancing the Military Health System’s public health capabilities. BMC Public Health. 2011;11(suppl 2):S1.
- Pavlin JA, Welch RA. Ethics, human use, and the department of defense serum repository. Mil Med. 2015;180:49-56.
- Defense Medical Surveillance System. Military Health System and the Defense Health Agency website. http://www.health.mil/Military-Health-Topics/Health-Readiness/Armed-Forces-Health-Surveillance-Branch/Data-Management-and-Technical-Support/Defense-Medical-Surveillance-System. Accessed August 2, 2016.
- Perdue CL, Eick-Cost AA, Rubertone MV, et al. Description and utilization of the United States Department of Defense Serum Repository: a review of published studies, 1985-2012. Plos One. 2015;10:1-16.
- Mancuso JD, Mallon TM, Gaydos JC. Maximizing the capabilities of the DoD serum repository to meet current and future needs: report of the needs panel. Mil Med. 2015;180:14-24.
- Department of Defense. Department of Defense Instruction. http://www.dtic.mil/whs/directives/corres/pdf/600014p.pdf. Posted September 26, 2001. Updated October 3, 2013. Accessed August 2, 2016.
- Lindler LE. Building a DoD biorepository for the future: potential benefits and way forward. Mil Med. 2015;180:90-94.
Practice Points
- Large patient databases and tissue repositories are increasingly being used to improve patient care through the use of clinical data, genomics, proteinomics, and metabolomics.
- The US Military has an established electronic medical record as well as tissue and serum repositories that can be leveraged to study melanoma and other dermatologic diseases.
Evaluating PD-1 Inhibitors
PD-1 antibodies are being touted as a promising immunotherapeutic approach for treating malignant melanoma among other cancers. The antibodies target proteins that promote programmed cell death and activate the immune system to attack tumors.
To find out more about the efficacy and safety of PD-1 antibody treatment, researchers from Xiamen University, Chinese Academy of Medical Sciences, and Peking Union Medical College, China, reviewed data from 5 multicenter, randomized clinical trials involving 2,828 patients. In 2 trials, patients were previously untreated; in the other 3, patients had progression after anti-CTLA-4 treatment or had received no more than 1 previous systemic therapy. Patients in the experimental groups received nivolumab or pembrolizumab; patients in the control groups received ipilimumab or chemotherapy.
In all 5 trials, the researchers noted significant differences between the anti-PD-1 groups and the control groups. The PD-1 antibody treatment was associated with a significantly better overall response rate (ORR): 40.0% in patients on nivolumab 3 mg/kg IV every 2 weeks as front-line therapy and 31.6% in those who received nivolumab at the same dosage after progression from anti-CTLA-4 treatment. Response was improved whether the drug was used as first-line treatment or for refractory/relapsed melanoma.
Related: Nivolumab Approved for Expanded Indication
Patients in the PD-1 groups also had a significantly greater rate of progression-free survival (PFS) compared with those who received other treatments, such as chemotherapy and ipilimumab. The median PFS was > 4.7 months in the nivolumab group and > 3.7 months in the pembrolizumab group. In 2 trials, the ORR among patients receiving pembrolizumab was between 23.3% and 33.2%; different dosages improved the overall response rate in both untreated and relapsed/refractory patients.
The most common adverse events (AEs) were fatigue, diarrhea, pruritus, rash, and nausea. Patients treated with nivolumab reported significantly fewer AEs. Although a high dosage or short intermission of pembrolizumab extended the median PFS, a subgroup analysis of different doses revealed a significant dose-dependent increase in AEs.
Source:
Lin Z, Chen X, Li Z, et al. PLoS One. 2016;11(8):e0160485.
doi: 10.1371/journal.pone.0160485.
PD-1 antibodies are being touted as a promising immunotherapeutic approach for treating malignant melanoma among other cancers. The antibodies target proteins that promote programmed cell death and activate the immune system to attack tumors.
To find out more about the efficacy and safety of PD-1 antibody treatment, researchers from Xiamen University, Chinese Academy of Medical Sciences, and Peking Union Medical College, China, reviewed data from 5 multicenter, randomized clinical trials involving 2,828 patients. In 2 trials, patients were previously untreated; in the other 3, patients had progression after anti-CTLA-4 treatment or had received no more than 1 previous systemic therapy. Patients in the experimental groups received nivolumab or pembrolizumab; patients in the control groups received ipilimumab or chemotherapy.
In all 5 trials, the researchers noted significant differences between the anti-PD-1 groups and the control groups. The PD-1 antibody treatment was associated with a significantly better overall response rate (ORR): 40.0% in patients on nivolumab 3 mg/kg IV every 2 weeks as front-line therapy and 31.6% in those who received nivolumab at the same dosage after progression from anti-CTLA-4 treatment. Response was improved whether the drug was used as first-line treatment or for refractory/relapsed melanoma.
Related: Nivolumab Approved for Expanded Indication
Patients in the PD-1 groups also had a significantly greater rate of progression-free survival (PFS) compared with those who received other treatments, such as chemotherapy and ipilimumab. The median PFS was > 4.7 months in the nivolumab group and > 3.7 months in the pembrolizumab group. In 2 trials, the ORR among patients receiving pembrolizumab was between 23.3% and 33.2%; different dosages improved the overall response rate in both untreated and relapsed/refractory patients.
The most common adverse events (AEs) were fatigue, diarrhea, pruritus, rash, and nausea. Patients treated with nivolumab reported significantly fewer AEs. Although a high dosage or short intermission of pembrolizumab extended the median PFS, a subgroup analysis of different doses revealed a significant dose-dependent increase in AEs.
Source:
Lin Z, Chen X, Li Z, et al. PLoS One. 2016;11(8):e0160485.
doi: 10.1371/journal.pone.0160485.
PD-1 antibodies are being touted as a promising immunotherapeutic approach for treating malignant melanoma among other cancers. The antibodies target proteins that promote programmed cell death and activate the immune system to attack tumors.
To find out more about the efficacy and safety of PD-1 antibody treatment, researchers from Xiamen University, Chinese Academy of Medical Sciences, and Peking Union Medical College, China, reviewed data from 5 multicenter, randomized clinical trials involving 2,828 patients. In 2 trials, patients were previously untreated; in the other 3, patients had progression after anti-CTLA-4 treatment or had received no more than 1 previous systemic therapy. Patients in the experimental groups received nivolumab or pembrolizumab; patients in the control groups received ipilimumab or chemotherapy.
In all 5 trials, the researchers noted significant differences between the anti-PD-1 groups and the control groups. The PD-1 antibody treatment was associated with a significantly better overall response rate (ORR): 40.0% in patients on nivolumab 3 mg/kg IV every 2 weeks as front-line therapy and 31.6% in those who received nivolumab at the same dosage after progression from anti-CTLA-4 treatment. Response was improved whether the drug was used as first-line treatment or for refractory/relapsed melanoma.
Related: Nivolumab Approved for Expanded Indication
Patients in the PD-1 groups also had a significantly greater rate of progression-free survival (PFS) compared with those who received other treatments, such as chemotherapy and ipilimumab. The median PFS was > 4.7 months in the nivolumab group and > 3.7 months in the pembrolizumab group. In 2 trials, the ORR among patients receiving pembrolizumab was between 23.3% and 33.2%; different dosages improved the overall response rate in both untreated and relapsed/refractory patients.
The most common adverse events (AEs) were fatigue, diarrhea, pruritus, rash, and nausea. Patients treated with nivolumab reported significantly fewer AEs. Although a high dosage or short intermission of pembrolizumab extended the median PFS, a subgroup analysis of different doses revealed a significant dose-dependent increase in AEs.
Source:
Lin Z, Chen X, Li Z, et al. PLoS One. 2016;11(8):e0160485.
doi: 10.1371/journal.pone.0160485.
FDA modifies dosage regimen for nivolumab
The Food and Drug Administration has modified the dosage regimen for nivolumab for indications of renal cell carcinoma, metastatic melanoma, and non–small cell lung cancer.
The single-dose regimen of nivolumab (3 mg/kg IV every 2 weeks) is replaced with the new recommended regimen of 240 mg IV every 2 weeks until disease progression or intolerable toxicity, the FDA said in a written statement.
The nivolumab (Opdivo) dosing regimen in combination with ipilimumab for melanoma will stay the same (nivolumab 1 mg/kg IV, followed by ipilimumab on the same day, every 3 weeks for four doses); however, after completion of ipilimumab, the recommended nivolumab dose is modified to 240 mg every 2 weeks until disease progression or intolerable toxicity. The recommended dose for classical Hodgkin lymphoma remains at 3 mg/kg IV every 2 weeks until disease progression or intolerable toxicity.
The change was made based on analyses demonstrating the comparability of the pharmacokinetics exposure, safety, and efficacy of the proposed new dosing regimen with the previously approved regimen. “Based on simulations by the population pharmacokinetics model, [the] FDA determined that the overall exposure at 240 mg every 2 weeks flat dose is similar (less than 6% difference) to 3 mg/kg every 2 weeks. These differences in exposure are not likely to have a clinically meaningful effect on safety and efficacy, since dose/exposure response relationships appear to be relatively flat in these three indications,” the FDA said.
The Food and Drug Administration has modified the dosage regimen for nivolumab for indications of renal cell carcinoma, metastatic melanoma, and non–small cell lung cancer.
The single-dose regimen of nivolumab (3 mg/kg IV every 2 weeks) is replaced with the new recommended regimen of 240 mg IV every 2 weeks until disease progression or intolerable toxicity, the FDA said in a written statement.
The nivolumab (Opdivo) dosing regimen in combination with ipilimumab for melanoma will stay the same (nivolumab 1 mg/kg IV, followed by ipilimumab on the same day, every 3 weeks for four doses); however, after completion of ipilimumab, the recommended nivolumab dose is modified to 240 mg every 2 weeks until disease progression or intolerable toxicity. The recommended dose for classical Hodgkin lymphoma remains at 3 mg/kg IV every 2 weeks until disease progression or intolerable toxicity.
The change was made based on analyses demonstrating the comparability of the pharmacokinetics exposure, safety, and efficacy of the proposed new dosing regimen with the previously approved regimen. “Based on simulations by the population pharmacokinetics model, [the] FDA determined that the overall exposure at 240 mg every 2 weeks flat dose is similar (less than 6% difference) to 3 mg/kg every 2 weeks. These differences in exposure are not likely to have a clinically meaningful effect on safety and efficacy, since dose/exposure response relationships appear to be relatively flat in these three indications,” the FDA said.
The Food and Drug Administration has modified the dosage regimen for nivolumab for indications of renal cell carcinoma, metastatic melanoma, and non–small cell lung cancer.
The single-dose regimen of nivolumab (3 mg/kg IV every 2 weeks) is replaced with the new recommended regimen of 240 mg IV every 2 weeks until disease progression or intolerable toxicity, the FDA said in a written statement.
The nivolumab (Opdivo) dosing regimen in combination with ipilimumab for melanoma will stay the same (nivolumab 1 mg/kg IV, followed by ipilimumab on the same day, every 3 weeks for four doses); however, after completion of ipilimumab, the recommended nivolumab dose is modified to 240 mg every 2 weeks until disease progression or intolerable toxicity. The recommended dose for classical Hodgkin lymphoma remains at 3 mg/kg IV every 2 weeks until disease progression or intolerable toxicity.
The change was made based on analyses demonstrating the comparability of the pharmacokinetics exposure, safety, and efficacy of the proposed new dosing regimen with the previously approved regimen. “Based on simulations by the population pharmacokinetics model, [the] FDA determined that the overall exposure at 240 mg every 2 weeks flat dose is similar (less than 6% difference) to 3 mg/kg every 2 weeks. These differences in exposure are not likely to have a clinically meaningful effect on safety and efficacy, since dose/exposure response relationships appear to be relatively flat in these three indications,” the FDA said.
Study suggests link between commonly used antihypertensive drug and skin cancer
Despite its widespread use as a guideline-recommended, first-line agent for the treatment of hypertension, hydrochlorothiazide (HCTZ) may contribute to an increased risk of skin cancer, according to Armand B. Cognetta Jr., MD, and his associates in the division of dermatology, Mohs Micrographic Surgery Unit, Florida State University, Tallahassee.
The common, long-term use of HCTZ for treatment of hypertension, combined with its known photosensitizing effects, makes it a potential candidate for increasing the risk of squamous cell carcinoma (SCC) and other skin cancers.
To further elucidate the association between longterm HCTZ exposure and skin cancer risk, Dr. Cognetta and his associates screened medication lists of 75 patients seen in their Mohs practice over a period of 5 days for lifetime SCCs and HCTZ exposure. For this study, patients with more than 20 lifetime SCCs were considered high risk. They also conducted a literature review of previous studies exploring this relationship, from 1966 to 2015.
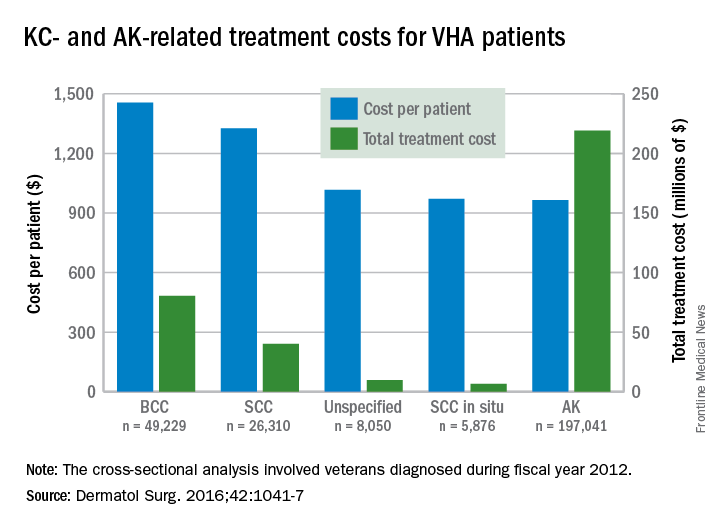
Of the 75 patients screened, 4 met the criteria for inclusion in the high risk category. These four patients had a combined lifetime total of 288 SCC and 98 basal cell carcinomas (BCCs), including 10 that were lip SCC. All four patients were non-Hispanic white males and had been taking HCTZ alone or in combination for 3-15 years (Dermatol Surg. 2016 Sep;42[9]:1107-9).
The literature search produced three relevant studies, all of which had large patient populations, published between 2008 and 2012, “demonstrating an increased risk of SCC or lip cancer” associated with HCTZ use, with the highest risk associated with over 5 years of use, the researchers wrote.
“As cutaneous oncologists, it is our duty to look for ‘correctable’ causes of skin cancer,” they noted. “Hydrochlorothiazide, a known photosensitizer, when taken by white non-Hispanic patients with a history of multiple SCC, skin cancers may represent a ‘correctable’ cause.”
In their practice, they screen their high-risk SCC non-Hispanic patients for HCTZ use, “and send a standard letter explaining the association to the primary care provider with a request to change to a different antihypertensive if possible,” they wrote. “Many primary care providers are unaware of the association between HCTZ use and skin cancer,” they observed.
The authors had no disclosures.
Despite its widespread use as a guideline-recommended, first-line agent for the treatment of hypertension, hydrochlorothiazide (HCTZ) may contribute to an increased risk of skin cancer, according to Armand B. Cognetta Jr., MD, and his associates in the division of dermatology, Mohs Micrographic Surgery Unit, Florida State University, Tallahassee.
The common, long-term use of HCTZ for treatment of hypertension, combined with its known photosensitizing effects, makes it a potential candidate for increasing the risk of squamous cell carcinoma (SCC) and other skin cancers.
To further elucidate the association between longterm HCTZ exposure and skin cancer risk, Dr. Cognetta and his associates screened medication lists of 75 patients seen in their Mohs practice over a period of 5 days for lifetime SCCs and HCTZ exposure. For this study, patients with more than 20 lifetime SCCs were considered high risk. They also conducted a literature review of previous studies exploring this relationship, from 1966 to 2015.
Of the 75 patients screened, 4 met the criteria for inclusion in the high risk category. These four patients had a combined lifetime total of 288 SCC and 98 basal cell carcinomas (BCCs), including 10 that were lip SCC. All four patients were non-Hispanic white males and had been taking HCTZ alone or in combination for 3-15 years (Dermatol Surg. 2016 Sep;42[9]:1107-9).
The literature search produced three relevant studies, all of which had large patient populations, published between 2008 and 2012, “demonstrating an increased risk of SCC or lip cancer” associated with HCTZ use, with the highest risk associated with over 5 years of use, the researchers wrote.
“As cutaneous oncologists, it is our duty to look for ‘correctable’ causes of skin cancer,” they noted. “Hydrochlorothiazide, a known photosensitizer, when taken by white non-Hispanic patients with a history of multiple SCC, skin cancers may represent a ‘correctable’ cause.”
In their practice, they screen their high-risk SCC non-Hispanic patients for HCTZ use, “and send a standard letter explaining the association to the primary care provider with a request to change to a different antihypertensive if possible,” they wrote. “Many primary care providers are unaware of the association between HCTZ use and skin cancer,” they observed.
The authors had no disclosures.
Despite its widespread use as a guideline-recommended, first-line agent for the treatment of hypertension, hydrochlorothiazide (HCTZ) may contribute to an increased risk of skin cancer, according to Armand B. Cognetta Jr., MD, and his associates in the division of dermatology, Mohs Micrographic Surgery Unit, Florida State University, Tallahassee.
The common, long-term use of HCTZ for treatment of hypertension, combined with its known photosensitizing effects, makes it a potential candidate for increasing the risk of squamous cell carcinoma (SCC) and other skin cancers.
To further elucidate the association between longterm HCTZ exposure and skin cancer risk, Dr. Cognetta and his associates screened medication lists of 75 patients seen in their Mohs practice over a period of 5 days for lifetime SCCs and HCTZ exposure. For this study, patients with more than 20 lifetime SCCs were considered high risk. They also conducted a literature review of previous studies exploring this relationship, from 1966 to 2015.
Of the 75 patients screened, 4 met the criteria for inclusion in the high risk category. These four patients had a combined lifetime total of 288 SCC and 98 basal cell carcinomas (BCCs), including 10 that were lip SCC. All four patients were non-Hispanic white males and had been taking HCTZ alone or in combination for 3-15 years (Dermatol Surg. 2016 Sep;42[9]:1107-9).
The literature search produced three relevant studies, all of which had large patient populations, published between 2008 and 2012, “demonstrating an increased risk of SCC or lip cancer” associated with HCTZ use, with the highest risk associated with over 5 years of use, the researchers wrote.
“As cutaneous oncologists, it is our duty to look for ‘correctable’ causes of skin cancer,” they noted. “Hydrochlorothiazide, a known photosensitizer, when taken by white non-Hispanic patients with a history of multiple SCC, skin cancers may represent a ‘correctable’ cause.”
In their practice, they screen their high-risk SCC non-Hispanic patients for HCTZ use, “and send a standard letter explaining the association to the primary care provider with a request to change to a different antihypertensive if possible,” they wrote. “Many primary care providers are unaware of the association between HCTZ use and skin cancer,” they observed.
The authors had no disclosures.
Key clinical point: The antihypertensive agent hydrochlorothiazide (HCTZ ) is photosensitizing and may be associated with an increased risk for skin cancers in non-Hispanic white males with a history of squamous cell carcinoma (SCC).
Major finding: All four patients considered high risk based on lifetime SCC history were non-Hispanic white males with a history of HCTZ exposure, an association supported by three previously published studies.
Data sources: Medical records from 75 patients seen in a single dermatology practice over a 5-day period, and a literature search as well as relevant publications detected through a literature search spanning the years 1966-2015.
Disclosures: The authors had no disclosures. A funding source was not identified.
Sequential protocol benefits patients with field cancerization
BOSTON – Field cancerization poses unique treatment challenges, but useful approaches to managing patients with such extreme dermatoheliosis are emerging, according to Anokhi Jambusaria-Pahlajani, MD.
In patients with extensive sun damage to the head and neck, she said she performs initial curettage of any hyperkeratotic actinic keratoses, followed by topical 0.5% 5-fluorouracil applied twice daily on postcurettage days 1 through 5. On day 6, she performs aminolevulinic acid photodynamic therapy (PDT) with 1 hour of incubation.
“Basically, on day zero they come into my office, and I look for anything that’s hyperkeratotic, and I numb it up and curette it. The goal of that is to get rid of the scale so that when I do the topical 5-fluorouracil or I do the photodynamic therapy, the medicine is actually getting to where we want it to go,” Dr. Jambusaria-Pahlajani, a dermatologist in Round Rock, Tex., said at the American Academy of Dermatology summer meeting.
She discussed one such patient – a lung transplant recipient – in whom this approach worked particularly well. At 6 months’ follow-up, the patient had had a significant response, and during a follow-up of at least 2 years, he did not develop a single actinic keratosis.
Early results with this combination approach were so encouraging that she and her colleagues published a case series of four solid organ transplant recipients. “All four patients tolerated the approach well, demonstrated excellent response to treatment with complete or near complete resolution of actinic keratosis and squamous cell carcinoma in situ lesions,” she said. At 1-6 months following treatment, “basically patients were clear,” she added (Dermatol Surg. 2016 Jan;42:S66-72).
Dr. Jambusaria-Pahlajani also said she has seen “great results” in most of the 30-40 other patients she has treated using this approach. “I’ve followed up patients for years, and they really haven’t had a problem like what they had prior to getting this treatment,” she said.
She noted that, anecdotally, many patients who had been treated with multiple courses of topical 5-fluorouracil monotherapy or photodynamic therapy actually preferred the combination approach. They felt that they were out of commission for a shorter period of time, and most of the patients “tended to be kind of red and inflamed for about 7-10 days total from start to finish, which was a lot better than with, for example, 5-fluorouracil,” she said.
One patient – another lung transplant recipient – initially responded to combination therapy, but returned in 3 months with numerous recurrent lesions. Based on findings from a 2010 study, a cyclic approach to PDT was tried in this patient. In that study of 12 solid organ transplant recipients, cyclic PDT reduced the incidence of squamous cell carcinoma (Dermatol Surg. 2010 May;36[5]:652-8).
The study subjects had a high burden of squamous cell carcinoma in situ. The cyclic PDT they received included blue light PDT with 1 hour of incubation every 4-8 weeks for 2 years. The median number of new invasive and in situ squamous cell carcinomas declined from about 20 to 4 in the first year (a 79% reduction vs. 1 month prior to first treatment) and to 1 at 2 years (a 95% reduction).
“This patient happened to be a transplant patient as well, but I think you could also use [this approach] in nontransplant patients,” Dr. Jambusaria-Pahlajani said. His response was “pretty significant,” and after 18 months of treatment he was doing well.
“The main thing that you have to remember, though, is that these patients have to be willing to come in every 4-8 weeks to get the PDT,” which, she added, “is not for every patient.” It’s also not for every provider. Aside from staff or resource access limitations, these treatments can be very time consuming and costly, she noted. “Treating these patients can be a challenge, but it can be rewarding, as well,” she commented.
She reported having no relevant financial disclosures.
BOSTON – Field cancerization poses unique treatment challenges, but useful approaches to managing patients with such extreme dermatoheliosis are emerging, according to Anokhi Jambusaria-Pahlajani, MD.
In patients with extensive sun damage to the head and neck, she said she performs initial curettage of any hyperkeratotic actinic keratoses, followed by topical 0.5% 5-fluorouracil applied twice daily on postcurettage days 1 through 5. On day 6, she performs aminolevulinic acid photodynamic therapy (PDT) with 1 hour of incubation.
“Basically, on day zero they come into my office, and I look for anything that’s hyperkeratotic, and I numb it up and curette it. The goal of that is to get rid of the scale so that when I do the topical 5-fluorouracil or I do the photodynamic therapy, the medicine is actually getting to where we want it to go,” Dr. Jambusaria-Pahlajani, a dermatologist in Round Rock, Tex., said at the American Academy of Dermatology summer meeting.
She discussed one such patient – a lung transplant recipient – in whom this approach worked particularly well. At 6 months’ follow-up, the patient had had a significant response, and during a follow-up of at least 2 years, he did not develop a single actinic keratosis.
Early results with this combination approach were so encouraging that she and her colleagues published a case series of four solid organ transplant recipients. “All four patients tolerated the approach well, demonstrated excellent response to treatment with complete or near complete resolution of actinic keratosis and squamous cell carcinoma in situ lesions,” she said. At 1-6 months following treatment, “basically patients were clear,” she added (Dermatol Surg. 2016 Jan;42:S66-72).
Dr. Jambusaria-Pahlajani also said she has seen “great results” in most of the 30-40 other patients she has treated using this approach. “I’ve followed up patients for years, and they really haven’t had a problem like what they had prior to getting this treatment,” she said.
She noted that, anecdotally, many patients who had been treated with multiple courses of topical 5-fluorouracil monotherapy or photodynamic therapy actually preferred the combination approach. They felt that they were out of commission for a shorter period of time, and most of the patients “tended to be kind of red and inflamed for about 7-10 days total from start to finish, which was a lot better than with, for example, 5-fluorouracil,” she said.
One patient – another lung transplant recipient – initially responded to combination therapy, but returned in 3 months with numerous recurrent lesions. Based on findings from a 2010 study, a cyclic approach to PDT was tried in this patient. In that study of 12 solid organ transplant recipients, cyclic PDT reduced the incidence of squamous cell carcinoma (Dermatol Surg. 2010 May;36[5]:652-8).
The study subjects had a high burden of squamous cell carcinoma in situ. The cyclic PDT they received included blue light PDT with 1 hour of incubation every 4-8 weeks for 2 years. The median number of new invasive and in situ squamous cell carcinomas declined from about 20 to 4 in the first year (a 79% reduction vs. 1 month prior to first treatment) and to 1 at 2 years (a 95% reduction).
“This patient happened to be a transplant patient as well, but I think you could also use [this approach] in nontransplant patients,” Dr. Jambusaria-Pahlajani said. His response was “pretty significant,” and after 18 months of treatment he was doing well.
“The main thing that you have to remember, though, is that these patients have to be willing to come in every 4-8 weeks to get the PDT,” which, she added, “is not for every patient.” It’s also not for every provider. Aside from staff or resource access limitations, these treatments can be very time consuming and costly, she noted. “Treating these patients can be a challenge, but it can be rewarding, as well,” she commented.
She reported having no relevant financial disclosures.
BOSTON – Field cancerization poses unique treatment challenges, but useful approaches to managing patients with such extreme dermatoheliosis are emerging, according to Anokhi Jambusaria-Pahlajani, MD.
In patients with extensive sun damage to the head and neck, she said she performs initial curettage of any hyperkeratotic actinic keratoses, followed by topical 0.5% 5-fluorouracil applied twice daily on postcurettage days 1 through 5. On day 6, she performs aminolevulinic acid photodynamic therapy (PDT) with 1 hour of incubation.
“Basically, on day zero they come into my office, and I look for anything that’s hyperkeratotic, and I numb it up and curette it. The goal of that is to get rid of the scale so that when I do the topical 5-fluorouracil or I do the photodynamic therapy, the medicine is actually getting to where we want it to go,” Dr. Jambusaria-Pahlajani, a dermatologist in Round Rock, Tex., said at the American Academy of Dermatology summer meeting.
She discussed one such patient – a lung transplant recipient – in whom this approach worked particularly well. At 6 months’ follow-up, the patient had had a significant response, and during a follow-up of at least 2 years, he did not develop a single actinic keratosis.
Early results with this combination approach were so encouraging that she and her colleagues published a case series of four solid organ transplant recipients. “All four patients tolerated the approach well, demonstrated excellent response to treatment with complete or near complete resolution of actinic keratosis and squamous cell carcinoma in situ lesions,” she said. At 1-6 months following treatment, “basically patients were clear,” she added (Dermatol Surg. 2016 Jan;42:S66-72).
Dr. Jambusaria-Pahlajani also said she has seen “great results” in most of the 30-40 other patients she has treated using this approach. “I’ve followed up patients for years, and they really haven’t had a problem like what they had prior to getting this treatment,” she said.
She noted that, anecdotally, many patients who had been treated with multiple courses of topical 5-fluorouracil monotherapy or photodynamic therapy actually preferred the combination approach. They felt that they were out of commission for a shorter period of time, and most of the patients “tended to be kind of red and inflamed for about 7-10 days total from start to finish, which was a lot better than with, for example, 5-fluorouracil,” she said.
One patient – another lung transplant recipient – initially responded to combination therapy, but returned in 3 months with numerous recurrent lesions. Based on findings from a 2010 study, a cyclic approach to PDT was tried in this patient. In that study of 12 solid organ transplant recipients, cyclic PDT reduced the incidence of squamous cell carcinoma (Dermatol Surg. 2010 May;36[5]:652-8).
The study subjects had a high burden of squamous cell carcinoma in situ. The cyclic PDT they received included blue light PDT with 1 hour of incubation every 4-8 weeks for 2 years. The median number of new invasive and in situ squamous cell carcinomas declined from about 20 to 4 in the first year (a 79% reduction vs. 1 month prior to first treatment) and to 1 at 2 years (a 95% reduction).
“This patient happened to be a transplant patient as well, but I think you could also use [this approach] in nontransplant patients,” Dr. Jambusaria-Pahlajani said. His response was “pretty significant,” and after 18 months of treatment he was doing well.
“The main thing that you have to remember, though, is that these patients have to be willing to come in every 4-8 weeks to get the PDT,” which, she added, “is not for every patient.” It’s also not for every provider. Aside from staff or resource access limitations, these treatments can be very time consuming and costly, she noted. “Treating these patients can be a challenge, but it can be rewarding, as well,” she commented.
She reported having no relevant financial disclosures.
EXPERT ANALYSIS FROM AAD SUMMER ACADEMY 2016
Veterans’ keratinocyte carcinoma, actinic keratosis care cost $356 million in 2012
Almost 4% of the 5.9 million veterans treated by the Veterans Health Administration in 2012 had a diagnosis of keratinocyte carcinoma (KC) or actinic keratosis (AK), and their treatment cost $356 million, according to an analysis of VHA-provided or -contracted outpatient encounters.
Treatment costs per patient for KCs, also known as nonmelanoma skin cancers, were $1,456 for basal cell carcinoma (BCC), $1,326 for squamous cell carcinoma (SCC), $1,016 for unspecified, nongenital invasive KCs, $971 for squamous cell carcinoma in situ, and $906 for genital skin cancer (not shown on graph), reported Jean Yoon, PhD, of Veterans Affairs Palo Alto Health Care System, Menlo Park, Calif., and her associates (Dermatol Surg. 2016;42:1041-7).
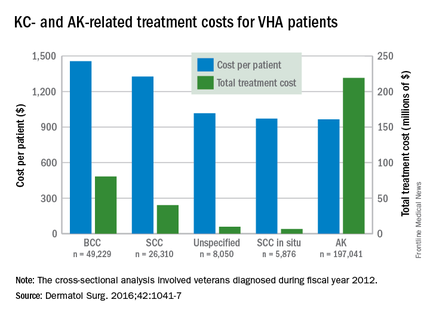
The VHA’s cost per patient for AK was relatively low – $965 per patient in 2012 – but the number of patients – 197,041 – was more than four times higher than any of the KCs. There were 49,229 veterans with BCC, 26,310 veterans with SCC, 8,050 veterans with unspecified KC, 5,876 veterans with SCC in situ, and 512 veterans with genital skin cancer, according to the analysis of administrative data on outpatient care and prescription drugs provided or paid by the VHA in fiscal year 2012. The total number of patients was 227,601, as some patients had more than one of the study diagnoses.
As a result of the high number of patients, actinic keratosis care totaled $219 million, compared with $80 million for BCC, $40 million for SCC, $9.6 million for nonspecified KC, $6.6 million for SCC in situ, and $582,000 for genital skin cancer, the investigators wrote.
The study was supported by a grant from the Department of Veterans Affairs. One of Dr. Yoon’s associates served as a consultant to several companies, but the remaining investigators had no conflicts to report.
Almost 4% of the 5.9 million veterans treated by the Veterans Health Administration in 2012 had a diagnosis of keratinocyte carcinoma (KC) or actinic keratosis (AK), and their treatment cost $356 million, according to an analysis of VHA-provided or -contracted outpatient encounters.
Treatment costs per patient for KCs, also known as nonmelanoma skin cancers, were $1,456 for basal cell carcinoma (BCC), $1,326 for squamous cell carcinoma (SCC), $1,016 for unspecified, nongenital invasive KCs, $971 for squamous cell carcinoma in situ, and $906 for genital skin cancer (not shown on graph), reported Jean Yoon, PhD, of Veterans Affairs Palo Alto Health Care System, Menlo Park, Calif., and her associates (Dermatol Surg. 2016;42:1041-7).

The VHA’s cost per patient for AK was relatively low – $965 per patient in 2012 – but the number of patients – 197,041 – was more than four times higher than any of the KCs. There were 49,229 veterans with BCC, 26,310 veterans with SCC, 8,050 veterans with unspecified KC, 5,876 veterans with SCC in situ, and 512 veterans with genital skin cancer, according to the analysis of administrative data on outpatient care and prescription drugs provided or paid by the VHA in fiscal year 2012. The total number of patients was 227,601, as some patients had more than one of the study diagnoses.
As a result of the high number of patients, actinic keratosis care totaled $219 million, compared with $80 million for BCC, $40 million for SCC, $9.6 million for nonspecified KC, $6.6 million for SCC in situ, and $582,000 for genital skin cancer, the investigators wrote.
The study was supported by a grant from the Department of Veterans Affairs. One of Dr. Yoon’s associates served as a consultant to several companies, but the remaining investigators had no conflicts to report.
Almost 4% of the 5.9 million veterans treated by the Veterans Health Administration in 2012 had a diagnosis of keratinocyte carcinoma (KC) or actinic keratosis (AK), and their treatment cost $356 million, according to an analysis of VHA-provided or -contracted outpatient encounters.
Treatment costs per patient for KCs, also known as nonmelanoma skin cancers, were $1,456 for basal cell carcinoma (BCC), $1,326 for squamous cell carcinoma (SCC), $1,016 for unspecified, nongenital invasive KCs, $971 for squamous cell carcinoma in situ, and $906 for genital skin cancer (not shown on graph), reported Jean Yoon, PhD, of Veterans Affairs Palo Alto Health Care System, Menlo Park, Calif., and her associates (Dermatol Surg. 2016;42:1041-7).

The VHA’s cost per patient for AK was relatively low – $965 per patient in 2012 – but the number of patients – 197,041 – was more than four times higher than any of the KCs. There were 49,229 veterans with BCC, 26,310 veterans with SCC, 8,050 veterans with unspecified KC, 5,876 veterans with SCC in situ, and 512 veterans with genital skin cancer, according to the analysis of administrative data on outpatient care and prescription drugs provided or paid by the VHA in fiscal year 2012. The total number of patients was 227,601, as some patients had more than one of the study diagnoses.
As a result of the high number of patients, actinic keratosis care totaled $219 million, compared with $80 million for BCC, $40 million for SCC, $9.6 million for nonspecified KC, $6.6 million for SCC in situ, and $582,000 for genital skin cancer, the investigators wrote.
The study was supported by a grant from the Department of Veterans Affairs. One of Dr. Yoon’s associates served as a consultant to several companies, but the remaining investigators had no conflicts to report.
FROM DERMATOLOGIC SURGERY
Pneumonitis with nivolumab treatment shows common radiographic patterns
A study of cancer patients enrolled in trials of the programmed cell death-1 inhibiting medicine nivolumab found that among a minority who developed pneumonitis during treatment, distinct radiographic patterns were significantly associated with the level of pneumonitis severity.
Investigators found that cryptic organizing pneumonia pattern (COP) was the most common, though not the most severe. Led by Mizuki Nishino, MD, of Brigham and Women’s Hospital, Boston, the researchers looked at the 20 patients out of a cohort of 170 (11.8%) who had developed pneumonitis, and found that radiologic patterns indicating acute interstitial pneumonia/acute respiratory distress syndrome (n = 2) had the highest severity grade on a scale of 1-5 (median 3), followed by those with COP pattern (n = 13, median grade 2), hypersensitivity pneumonitis (n = 2, median grade 1), and nonspecific interstitial pneumonia (n = 3, median grade 1). The pattern was significantly associated with severity (P = .0006).
The study cohort included patients being treated with nivolumab for lung cancer, melanoma, and lymphoma; the COP patten was the most common across tumor types and observed in patients receiving monotherapy and combination therapy alike. Therapy with nivolumab was suspended for all 20 pneumonitis patients, and most (n = 17) received treatment for pneumonitis with corticosteroids with or without infliximab, for a median treatment time of 6 weeks. Seven patients were able to restart nivolumab, though pneumonitis recurred in two, the investigators reported (Clin Cancer Res. 2016 Aug 17. doi: 10.1158/1078-0432.CCR-16-1320).
“Time from initiation of therapy to the development of pneumonitis had a wide range (0.5-11.5 months), indicating an importance of careful observation and follow-up for signs and symptoms of pneumonitis throughout treatment,” Dr. Nishino and colleagues wrote in their analysis, adding that shorter times were observed for lung cancer patients, possibly because of their higher pulmonary burden, a lower threshold for performing chest scans in these patients, or both. “In most patients, clinical and radiographic improvements were noted after treatment, indicating that [PD-1 inhibitor-related pneumonitis], although potentially serious, is treatable if diagnosed and managed appropriately. The observation emphasizes the importance of timely recognition, accurate diagnosis, and early intervention.”
The lead author and several coauthors disclosed funding from Bristol-Myers Squibb, which sponsored the trial, as well as from other manufacturers.
A study of cancer patients enrolled in trials of the programmed cell death-1 inhibiting medicine nivolumab found that among a minority who developed pneumonitis during treatment, distinct radiographic patterns were significantly associated with the level of pneumonitis severity.
Investigators found that cryptic organizing pneumonia pattern (COP) was the most common, though not the most severe. Led by Mizuki Nishino, MD, of Brigham and Women’s Hospital, Boston, the researchers looked at the 20 patients out of a cohort of 170 (11.8%) who had developed pneumonitis, and found that radiologic patterns indicating acute interstitial pneumonia/acute respiratory distress syndrome (n = 2) had the highest severity grade on a scale of 1-5 (median 3), followed by those with COP pattern (n = 13, median grade 2), hypersensitivity pneumonitis (n = 2, median grade 1), and nonspecific interstitial pneumonia (n = 3, median grade 1). The pattern was significantly associated with severity (P = .0006).
The study cohort included patients being treated with nivolumab for lung cancer, melanoma, and lymphoma; the COP patten was the most common across tumor types and observed in patients receiving monotherapy and combination therapy alike. Therapy with nivolumab was suspended for all 20 pneumonitis patients, and most (n = 17) received treatment for pneumonitis with corticosteroids with or without infliximab, for a median treatment time of 6 weeks. Seven patients were able to restart nivolumab, though pneumonitis recurred in two, the investigators reported (Clin Cancer Res. 2016 Aug 17. doi: 10.1158/1078-0432.CCR-16-1320).
“Time from initiation of therapy to the development of pneumonitis had a wide range (0.5-11.5 months), indicating an importance of careful observation and follow-up for signs and symptoms of pneumonitis throughout treatment,” Dr. Nishino and colleagues wrote in their analysis, adding that shorter times were observed for lung cancer patients, possibly because of their higher pulmonary burden, a lower threshold for performing chest scans in these patients, or both. “In most patients, clinical and radiographic improvements were noted after treatment, indicating that [PD-1 inhibitor-related pneumonitis], although potentially serious, is treatable if diagnosed and managed appropriately. The observation emphasizes the importance of timely recognition, accurate diagnosis, and early intervention.”
The lead author and several coauthors disclosed funding from Bristol-Myers Squibb, which sponsored the trial, as well as from other manufacturers.
A study of cancer patients enrolled in trials of the programmed cell death-1 inhibiting medicine nivolumab found that among a minority who developed pneumonitis during treatment, distinct radiographic patterns were significantly associated with the level of pneumonitis severity.
Investigators found that cryptic organizing pneumonia pattern (COP) was the most common, though not the most severe. Led by Mizuki Nishino, MD, of Brigham and Women’s Hospital, Boston, the researchers looked at the 20 patients out of a cohort of 170 (11.8%) who had developed pneumonitis, and found that radiologic patterns indicating acute interstitial pneumonia/acute respiratory distress syndrome (n = 2) had the highest severity grade on a scale of 1-5 (median 3), followed by those with COP pattern (n = 13, median grade 2), hypersensitivity pneumonitis (n = 2, median grade 1), and nonspecific interstitial pneumonia (n = 3, median grade 1). The pattern was significantly associated with severity (P = .0006).
The study cohort included patients being treated with nivolumab for lung cancer, melanoma, and lymphoma; the COP patten was the most common across tumor types and observed in patients receiving monotherapy and combination therapy alike. Therapy with nivolumab was suspended for all 20 pneumonitis patients, and most (n = 17) received treatment for pneumonitis with corticosteroids with or without infliximab, for a median treatment time of 6 weeks. Seven patients were able to restart nivolumab, though pneumonitis recurred in two, the investigators reported (Clin Cancer Res. 2016 Aug 17. doi: 10.1158/1078-0432.CCR-16-1320).
“Time from initiation of therapy to the development of pneumonitis had a wide range (0.5-11.5 months), indicating an importance of careful observation and follow-up for signs and symptoms of pneumonitis throughout treatment,” Dr. Nishino and colleagues wrote in their analysis, adding that shorter times were observed for lung cancer patients, possibly because of their higher pulmonary burden, a lower threshold for performing chest scans in these patients, or both. “In most patients, clinical and radiographic improvements were noted after treatment, indicating that [PD-1 inhibitor-related pneumonitis], although potentially serious, is treatable if diagnosed and managed appropriately. The observation emphasizes the importance of timely recognition, accurate diagnosis, and early intervention.”
The lead author and several coauthors disclosed funding from Bristol-Myers Squibb, which sponsored the trial, as well as from other manufacturers.
FROM CLINICAL CANCER RESEARCH
Key clinical point: Pneumonitis related to treatment with PD-1 inhibitors showed distinct radiographic patterns associated with severity; most cases resolved with corticosteroid treatment.
Major finding: Of 20 patients in nivolumab trials who developed pneumonitis, a COP pattern was seen in 13, and other patterns in 7; different patterns were significantly associated with pneumonitis severity (P = .006).
Data source: 170 patients with melanoma, lung cancer or lymphoma enrolled in single-site open-label clinical trial of nivolumab.
Disclosures: The lead author and several coauthors disclosed funding from Bristol-Myers Squibb, which sponsored the trial, as well as from other manufacturers.