User login
EADV: Fresh insights into Gorlin syndrome
COPENHAGEN – A glimpse into the massive burden imposed upon patients with basal cell nevus syndrome (BCNS) is provided by the initial report from the first U.S. registry of patients affected with the disorder to include individuals who’ve tried the Hedgehog signaling pathway inhibitor vismodegib.
The first 94 patients (average age, 56 years) with BCNS to enroll in the prospective registry had a reported lifetime mean of 312 basal cell carcinomas (BCCs), Dr. Marieke Peters reported at the annual congress of the European Academy of Dermatology and Venereology.
The patients had an average of 36 new BCCs or other tumors in the previous 2 years, and 70% were categorized as having moderate to severe BCNS (also known as Gorlin syndrome), as defined by more than 10 new BCCs during that time period.
Most (94%) of tumors were on sun-exposed areas. Patients reported a lifetime mean of 202 surgical excisions, according to Dr. Peters of Catharina Hospital Eindhoven, the Netherlands.
The median age at diagnosis of BCNS was 15 years, and most patients were diagnosed clinically. Only 26% had undergone genetic testing for PTCH1. (The syndrome is caused by mutations in the PTCH1 gene). Sixty-two percent of subjects reported a family history of BCNS.
Other abnormalities associated with BCNS were common: Eighty percent of patients had jaw cysts, 82% had palmer pitting, 50% had various bone abnormalities, 21% reported ovarian fibromas, 4% reported medulloblastomas, and 4% had other tumors.
Fifty-seven percent of patients had tried vismodegib (Erivedge). However, only 15% were currently on the drug at enrollment; the rest had discontinued it, mainly because of side effects; less frequently because of loss of efficacy due to the development of new mutations. The chief side effects were nausea, vomiting, and diarrhea; less frequent side effects were weight loss, fatigue, muscle cramps, and alopecia, according to the dermatologist.
Gorlin syndrome is a rare autosomal dominant disorder caused mainly by mutations in the PTCH1 gene. The U.S. registry is directed by investigators at Children’s Hospital Oakland Research Institute and Stanford (Calif.) University.
The patient questionnaire used in the BCNS registry is available at https://redcap.stanford.edu/surveys/?s=7MWW9E37ND. For more information about the registry, contact Dr. Ervin Epstein, at [email protected].
Dr. Peters reported having no financial conflicts regarding her report. At the time of the EADV meeting, she was a dermatology resident at the Academic Medical Center, Amsterdam.
*This story was updated 12/15/2015.
COPENHAGEN – A glimpse into the massive burden imposed upon patients with basal cell nevus syndrome (BCNS) is provided by the initial report from the first U.S. registry of patients affected with the disorder to include individuals who’ve tried the Hedgehog signaling pathway inhibitor vismodegib.
The first 94 patients (average age, 56 years) with BCNS to enroll in the prospective registry had a reported lifetime mean of 312 basal cell carcinomas (BCCs), Dr. Marieke Peters reported at the annual congress of the European Academy of Dermatology and Venereology.
The patients had an average of 36 new BCCs or other tumors in the previous 2 years, and 70% were categorized as having moderate to severe BCNS (also known as Gorlin syndrome), as defined by more than 10 new BCCs during that time period.
Most (94%) of tumors were on sun-exposed areas. Patients reported a lifetime mean of 202 surgical excisions, according to Dr. Peters of Catharina Hospital Eindhoven, the Netherlands.
The median age at diagnosis of BCNS was 15 years, and most patients were diagnosed clinically. Only 26% had undergone genetic testing for PTCH1. (The syndrome is caused by mutations in the PTCH1 gene). Sixty-two percent of subjects reported a family history of BCNS.
Other abnormalities associated with BCNS were common: Eighty percent of patients had jaw cysts, 82% had palmer pitting, 50% had various bone abnormalities, 21% reported ovarian fibromas, 4% reported medulloblastomas, and 4% had other tumors.
Fifty-seven percent of patients had tried vismodegib (Erivedge). However, only 15% were currently on the drug at enrollment; the rest had discontinued it, mainly because of side effects; less frequently because of loss of efficacy due to the development of new mutations. The chief side effects were nausea, vomiting, and diarrhea; less frequent side effects were weight loss, fatigue, muscle cramps, and alopecia, according to the dermatologist.
Gorlin syndrome is a rare autosomal dominant disorder caused mainly by mutations in the PTCH1 gene. The U.S. registry is directed by investigators at Children’s Hospital Oakland Research Institute and Stanford (Calif.) University.
The patient questionnaire used in the BCNS registry is available at https://redcap.stanford.edu/surveys/?s=7MWW9E37ND. For more information about the registry, contact Dr. Ervin Epstein, at [email protected].
Dr. Peters reported having no financial conflicts regarding her report. At the time of the EADV meeting, she was a dermatology resident at the Academic Medical Center, Amsterdam.
*This story was updated 12/15/2015.
COPENHAGEN – A glimpse into the massive burden imposed upon patients with basal cell nevus syndrome (BCNS) is provided by the initial report from the first U.S. registry of patients affected with the disorder to include individuals who’ve tried the Hedgehog signaling pathway inhibitor vismodegib.
The first 94 patients (average age, 56 years) with BCNS to enroll in the prospective registry had a reported lifetime mean of 312 basal cell carcinomas (BCCs), Dr. Marieke Peters reported at the annual congress of the European Academy of Dermatology and Venereology.
The patients had an average of 36 new BCCs or other tumors in the previous 2 years, and 70% were categorized as having moderate to severe BCNS (also known as Gorlin syndrome), as defined by more than 10 new BCCs during that time period.
Most (94%) of tumors were on sun-exposed areas. Patients reported a lifetime mean of 202 surgical excisions, according to Dr. Peters of Catharina Hospital Eindhoven, the Netherlands.
The median age at diagnosis of BCNS was 15 years, and most patients were diagnosed clinically. Only 26% had undergone genetic testing for PTCH1. (The syndrome is caused by mutations in the PTCH1 gene). Sixty-two percent of subjects reported a family history of BCNS.
Other abnormalities associated with BCNS were common: Eighty percent of patients had jaw cysts, 82% had palmer pitting, 50% had various bone abnormalities, 21% reported ovarian fibromas, 4% reported medulloblastomas, and 4% had other tumors.
Fifty-seven percent of patients had tried vismodegib (Erivedge). However, only 15% were currently on the drug at enrollment; the rest had discontinued it, mainly because of side effects; less frequently because of loss of efficacy due to the development of new mutations. The chief side effects were nausea, vomiting, and diarrhea; less frequent side effects were weight loss, fatigue, muscle cramps, and alopecia, according to the dermatologist.
Gorlin syndrome is a rare autosomal dominant disorder caused mainly by mutations in the PTCH1 gene. The U.S. registry is directed by investigators at Children’s Hospital Oakland Research Institute and Stanford (Calif.) University.
The patient questionnaire used in the BCNS registry is available at https://redcap.stanford.edu/surveys/?s=7MWW9E37ND. For more information about the registry, contact Dr. Ervin Epstein, at [email protected].
Dr. Peters reported having no financial conflicts regarding her report. At the time of the EADV meeting, she was a dermatology resident at the Academic Medical Center, Amsterdam.
*This story was updated 12/15/2015.
AT THE EADV CONGRESS
Key clinical point: Gorlin syndrome imposes an impressively heavy burden on affected patients.
Major finding: Patients with basal cell nevus syndrome reported a lifetime mean of 312 basal cell carcinomas and 202 surgical excisions.
Data source: The initial report from a prospective U.S. registry of patients with basal cell nevus syndrome, which is still enrolling participants.
Disclosures: The presenter reported having no financial conflicts regarding her study.
Nivolumab approved as first-line, single agent for advanced melanoma
Nivolumab, a programmed death receptor–1 (PD-1) blocking antibody, has been approved as a single agent for the first-line treatment of patients with BRAF V600 wild-type unresectable or metastatic melanoma, the manufacturer has announced.
The approval is based on the results of a phase III study, CheckMate 066, which found a significant overall survival (OS) benefit for nivolumab, compared with chemotherapy, as a first-line treatment of patients with BRAF wild-type advanced melanoma, according to a press release issued by Bristol Myers Squibb (BMS) on Nov. 24.
The company markets nivolumab as Opdivo, which was first approved by the Food and Drug Administration in December 2014 for the treatment of patients with unresectable or metastatic melanoma and disease progression following ipilimumab (Yervoy) treatment and, if BRAF V600 mutation positive, a BRAF inhibitor.
OS was the primary endpoint in the CheckMate 066 trial, which compared treatment with nivolumab (3mg/kg administered intravenously every 2 weeks) to dacarbazine (1,000 mg/m2 administered intravenously every 3 weeks) administered in 418 treatment-naive patients with BRAF wild-type unresectable or metastatic melanoma. In an interim analysis, the median OS for those on dacarbazine was 10.8 months, but was not reached for those on nivolumab (hazard ratio, 0.42; P less than .0001), according to BMS.
The study was stopped early in 2014, after the OS results were noted, the company statement said. At the time the study was designed, ipilimumab had not yet been approved, and dacarbazine was chosen as the comparator because “it represented the standard of care in many regions outside of the U.S.,” the statement added.
Other results included a significantly greater median PFS among those on nivolumab vs. dacarbazine (5.1 months vs. 2.2 months).
In the study, 36% of nivolumab-treated patients had serious adverse reactions; and 41% had grade 3 and 4 adverse reactions, the most common included an increase in gamma-glutamyl transferase in almost 4%, and diarrhea in 3.4%. In addition, 7% stopped treatment permanently because of adverse events, and treatment had to be interrupted in 26%.
The most common adverse reactions reported by those on nivolumab included fatigue, in 49% (vs. 39% of those on dacarbazine) and musculoskeletal pain in 32% (vs. 25%). Rash and pruritus were reported by 28% and 23%, respectively, among those on nivolumab vs. 12% for both rash and pruritus among those on dacarbazine, BMS said.
Nivolumab, a programmed death receptor–1 (PD-1) blocking antibody, has been approved as a single agent for the first-line treatment of patients with BRAF V600 wild-type unresectable or metastatic melanoma, the manufacturer has announced.
The approval is based on the results of a phase III study, CheckMate 066, which found a significant overall survival (OS) benefit for nivolumab, compared with chemotherapy, as a first-line treatment of patients with BRAF wild-type advanced melanoma, according to a press release issued by Bristol Myers Squibb (BMS) on Nov. 24.
The company markets nivolumab as Opdivo, which was first approved by the Food and Drug Administration in December 2014 for the treatment of patients with unresectable or metastatic melanoma and disease progression following ipilimumab (Yervoy) treatment and, if BRAF V600 mutation positive, a BRAF inhibitor.
OS was the primary endpoint in the CheckMate 066 trial, which compared treatment with nivolumab (3mg/kg administered intravenously every 2 weeks) to dacarbazine (1,000 mg/m2 administered intravenously every 3 weeks) administered in 418 treatment-naive patients with BRAF wild-type unresectable or metastatic melanoma. In an interim analysis, the median OS for those on dacarbazine was 10.8 months, but was not reached for those on nivolumab (hazard ratio, 0.42; P less than .0001), according to BMS.
The study was stopped early in 2014, after the OS results were noted, the company statement said. At the time the study was designed, ipilimumab had not yet been approved, and dacarbazine was chosen as the comparator because “it represented the standard of care in many regions outside of the U.S.,” the statement added.
Other results included a significantly greater median PFS among those on nivolumab vs. dacarbazine (5.1 months vs. 2.2 months).
In the study, 36% of nivolumab-treated patients had serious adverse reactions; and 41% had grade 3 and 4 adverse reactions, the most common included an increase in gamma-glutamyl transferase in almost 4%, and diarrhea in 3.4%. In addition, 7% stopped treatment permanently because of adverse events, and treatment had to be interrupted in 26%.
The most common adverse reactions reported by those on nivolumab included fatigue, in 49% (vs. 39% of those on dacarbazine) and musculoskeletal pain in 32% (vs. 25%). Rash and pruritus were reported by 28% and 23%, respectively, among those on nivolumab vs. 12% for both rash and pruritus among those on dacarbazine, BMS said.
Nivolumab, a programmed death receptor–1 (PD-1) blocking antibody, has been approved as a single agent for the first-line treatment of patients with BRAF V600 wild-type unresectable or metastatic melanoma, the manufacturer has announced.
The approval is based on the results of a phase III study, CheckMate 066, which found a significant overall survival (OS) benefit for nivolumab, compared with chemotherapy, as a first-line treatment of patients with BRAF wild-type advanced melanoma, according to a press release issued by Bristol Myers Squibb (BMS) on Nov. 24.
The company markets nivolumab as Opdivo, which was first approved by the Food and Drug Administration in December 2014 for the treatment of patients with unresectable or metastatic melanoma and disease progression following ipilimumab (Yervoy) treatment and, if BRAF V600 mutation positive, a BRAF inhibitor.
OS was the primary endpoint in the CheckMate 066 trial, which compared treatment with nivolumab (3mg/kg administered intravenously every 2 weeks) to dacarbazine (1,000 mg/m2 administered intravenously every 3 weeks) administered in 418 treatment-naive patients with BRAF wild-type unresectable or metastatic melanoma. In an interim analysis, the median OS for those on dacarbazine was 10.8 months, but was not reached for those on nivolumab (hazard ratio, 0.42; P less than .0001), according to BMS.
The study was stopped early in 2014, after the OS results were noted, the company statement said. At the time the study was designed, ipilimumab had not yet been approved, and dacarbazine was chosen as the comparator because “it represented the standard of care in many regions outside of the U.S.,” the statement added.
Other results included a significantly greater median PFS among those on nivolumab vs. dacarbazine (5.1 months vs. 2.2 months).
In the study, 36% of nivolumab-treated patients had serious adverse reactions; and 41% had grade 3 and 4 adverse reactions, the most common included an increase in gamma-glutamyl transferase in almost 4%, and diarrhea in 3.4%. In addition, 7% stopped treatment permanently because of adverse events, and treatment had to be interrupted in 26%.
The most common adverse reactions reported by those on nivolumab included fatigue, in 49% (vs. 39% of those on dacarbazine) and musculoskeletal pain in 32% (vs. 25%). Rash and pruritus were reported by 28% and 23%, respectively, among those on nivolumab vs. 12% for both rash and pruritus among those on dacarbazine, BMS said.
Dermatologists’ management of melanoma varies
CHICAGO – Significant variance exists in management of primary cutaneous melanoma, according to a national survey of 510 dermatologists.
Most dermatologists (36%) preferred a shave biopsy for lesions suspected of being melanoma, despite guidelines from the American Academy of Dermatology (AAD) and National Comprehensive Cancer Network (NCCN) guidelines that recommend narrow excision biopsy.
In all, 31% of dermatologists used a narrow local excision (less than 5 mm margin), 13% saucerization/scoop shave biopsy, 11%, punch biopsy, 3% wide local excision, and 7% other.
“The guidelines and academy are all very clear that one of the goals of the biopsy is to obtain tumor depth, so we were surprised that a significant number of providers use shave biopsy or other methods that may leave a risk of not getting the correct depth,” study co-author Dr. Aaron S. Farberg, a melanoma clinical research fellow at the National Society for Cutaneous Medicine in New York City, said in an interview.
Notably, dermatologists in academic and dermatology-based group practices were significantly less likely than those in multispecialty or solo practice to use narrow excision (23% vs. 42%; P < .001).
Although treatment for melanoma evolves continuously, the authors observed that dermatologists remain at the forefront of melanoma management and play a critical role in patient decision making.
“This study suggests that a knowledge gap may exist representing an educational opportunity to more effectively disseminate and implement recommended approaches,” Dr. Farberg and Dr. Darrell Rigel, of New York University School of Medicine, reported in a poster presentation at the annual meeting of the American Society for Dermatologic Surgery.
The survey also revealed that dermatologists are going beyond suggested surgical margins when excising melanoma.
For malignant melanoma in situ (MMIS), 62% used a 5 mm or less margin, 36% a 6 mm to 10 mm margin, and 2% a 1.1 cm to 1.9 cm margin. For these lesions, the AAD recommends a 0.5 cm-1 cm (5 mm-10 mm) margin and the NCCN a 0.5 cm margin, Drs. Farber and Rigel reported.
Academic dermatologists were significantly more likely than all other practice types to refer patients with MMIS out for excision (18% vs. 10%; P < .05).
For invasive melanoma less than 1 mm in depth, both the AAD and NCCN recommend a 1 cm margin (10 mm). In all, 61% of dermatologists reported using 6 mm to 10 mm margins, with 34% opting for 1.1 cm to 1.9 cm margins, 3% at least 2 cm margins, and 2% no more than 5 mm margins.
No significant difference was found across provider types for treatment of melanomas less than 1 mm in depth.
For invasive melanoma greater than 1 mm in depth, 54% of respondents used 1.1 cm to 1.9 cm margins, with most (67%) referring the patient to another provider. Both national guidelines recommend 1 cm to 2 cm margins for melanomas 1 mm to 2 mm in depth and 2 cm margins for melanoma greater than 2 mm in depth.
Academic dermatologists were significantly more likely than other dermatologists to treat these lesions rather than to refer the patient out (51% vs. 30%; P < .001).
“This is exciting new data that suggests that there still is a variance in early melanoma management,” Dr. Rigel, past president of AAD and ASDS, said in an interview. “The data suggest more studies need to be done to better access why this is occurring.”
Dr. Hensin Tsao, who served on the 2011 AAD guideline working group and is co-chairing the AAD’s pending guideline update, said in an interview that, “Dr. Rigel is well respected in the field and the project will undoubtedly be submitted for publication and subject to further review. It is worthwhile to wait on the final published results and conclusions.”
He agreed, however, with the authors’ suggestion that there is significant variation in practice. Regarding the finding that 36% of respondents use a shave biopsy for suspicious lesions, the AAD guidelines recommend that the entire lesion be removed with a 1 mm to 3 mm margin, which can be accomplished by an elliptical or punch excision with sutures or shave removal to a depth below the anticipated plane of the lesion, Dr. Tsao, of Massachusetts General Hospital in Boston, said.
“It is quite possible that some of the respondents to the questionnaire interpreted ‘shave biopsy’ as a full shave disk excision,” he said. “That said, intentional and routine partial sampling of suspected melanomas would be at odds with the guidelines.”
It is not inappropriate to remove a suspicious lesion, if small enough, with a punch biopsy, Dr. Tsao said, adding, “Perhaps again, the respondents failed to distinguish between partial punch biopsy and punch excision.”
On occasion, the AAD guidelines also make accommodations for an incisional biopsy “of the clinically or dermatoscopically most atypical portion of the lesion.” In this situation, a smaller incisional punch biopsy may be performed of the highly suspicious area.
“There are certainly areas that the investigators have identified which could represent potential knowledge gaps,” Dr. Tsao said. “For instance, 14% of the respondents used a 0.6 cm to 1.0 cm (6 mm-10 mm) margin for melanoma greater than 1 mm.”
Dermatologists’ surveillance of patients in the survey was somewhat less divergent. The most recommended follow-up interval for patients diagnosed within the last 5 years, was 6 months (49%), followed by a 3-month interval (25%), and other (24%). Follow-up was extended to yearly by 63% of dermatologists for patients diagnosed more than five years earlier, Dr. Farberg and Dr. Rigel reported.
Both the AAD and NCCN recommend follow-up every 3 to 12 months for those diagnosed with melanoma within the previous 5 years, while for those diagnosed more than 5 years prior, the AAD recommends follow-up every 3 to 12 months and the NCCN every 12 months.
“Adherence to evidence-based guidelines should lead to improved patient outcomes and quality of care. However, newer studies and emerging data may also justify deviations from existing guidelines, suggesting review of those guidelines may be indicated,” Dr. Farberg and Dr. Rigel concluded.
For example, a prospective series examining surgical margins in 1,072 patients with 1,120 malignant melanoma in situs showed that only 86% were successfully excised with a 6-mm margin, significantly less than the 98.9% clearance achieved with a 9-mm margin, Dr. Farberg told this publication.
That said, a 9-mm margin may not be appropriate for all lesions, such as those on the face, he observed.
The investigators said they hope the survey results will spur on revision of the guidelines, last updated in 2011.
Limitations of the survey include a lack of information on clinical factors such as patient history or anatomic site, the survey may have led to generalized answers, and access to care and reimbursement also may have impacted management, the authors noted.
The response rate to the survey was also low at 8%, with 6,177 practicing U.S. dermatologists surveyed. The demographics of the respondents, however, strongly reflected the full AAD membership, Dr. Farberg said. No significant geographical differences were observed.
CHICAGO – Significant variance exists in management of primary cutaneous melanoma, according to a national survey of 510 dermatologists.
Most dermatologists (36%) preferred a shave biopsy for lesions suspected of being melanoma, despite guidelines from the American Academy of Dermatology (AAD) and National Comprehensive Cancer Network (NCCN) guidelines that recommend narrow excision biopsy.
In all, 31% of dermatologists used a narrow local excision (less than 5 mm margin), 13% saucerization/scoop shave biopsy, 11%, punch biopsy, 3% wide local excision, and 7% other.
“The guidelines and academy are all very clear that one of the goals of the biopsy is to obtain tumor depth, so we were surprised that a significant number of providers use shave biopsy or other methods that may leave a risk of not getting the correct depth,” study co-author Dr. Aaron S. Farberg, a melanoma clinical research fellow at the National Society for Cutaneous Medicine in New York City, said in an interview.
Notably, dermatologists in academic and dermatology-based group practices were significantly less likely than those in multispecialty or solo practice to use narrow excision (23% vs. 42%; P < .001).
Although treatment for melanoma evolves continuously, the authors observed that dermatologists remain at the forefront of melanoma management and play a critical role in patient decision making.
“This study suggests that a knowledge gap may exist representing an educational opportunity to more effectively disseminate and implement recommended approaches,” Dr. Farberg and Dr. Darrell Rigel, of New York University School of Medicine, reported in a poster presentation at the annual meeting of the American Society for Dermatologic Surgery.
The survey also revealed that dermatologists are going beyond suggested surgical margins when excising melanoma.
For malignant melanoma in situ (MMIS), 62% used a 5 mm or less margin, 36% a 6 mm to 10 mm margin, and 2% a 1.1 cm to 1.9 cm margin. For these lesions, the AAD recommends a 0.5 cm-1 cm (5 mm-10 mm) margin and the NCCN a 0.5 cm margin, Drs. Farber and Rigel reported.
Academic dermatologists were significantly more likely than all other practice types to refer patients with MMIS out for excision (18% vs. 10%; P < .05).
For invasive melanoma less than 1 mm in depth, both the AAD and NCCN recommend a 1 cm margin (10 mm). In all, 61% of dermatologists reported using 6 mm to 10 mm margins, with 34% opting for 1.1 cm to 1.9 cm margins, 3% at least 2 cm margins, and 2% no more than 5 mm margins.
No significant difference was found across provider types for treatment of melanomas less than 1 mm in depth.
For invasive melanoma greater than 1 mm in depth, 54% of respondents used 1.1 cm to 1.9 cm margins, with most (67%) referring the patient to another provider. Both national guidelines recommend 1 cm to 2 cm margins for melanomas 1 mm to 2 mm in depth and 2 cm margins for melanoma greater than 2 mm in depth.
Academic dermatologists were significantly more likely than other dermatologists to treat these lesions rather than to refer the patient out (51% vs. 30%; P < .001).
“This is exciting new data that suggests that there still is a variance in early melanoma management,” Dr. Rigel, past president of AAD and ASDS, said in an interview. “The data suggest more studies need to be done to better access why this is occurring.”
Dr. Hensin Tsao, who served on the 2011 AAD guideline working group and is co-chairing the AAD’s pending guideline update, said in an interview that, “Dr. Rigel is well respected in the field and the project will undoubtedly be submitted for publication and subject to further review. It is worthwhile to wait on the final published results and conclusions.”
He agreed, however, with the authors’ suggestion that there is significant variation in practice. Regarding the finding that 36% of respondents use a shave biopsy for suspicious lesions, the AAD guidelines recommend that the entire lesion be removed with a 1 mm to 3 mm margin, which can be accomplished by an elliptical or punch excision with sutures or shave removal to a depth below the anticipated plane of the lesion, Dr. Tsao, of Massachusetts General Hospital in Boston, said.
“It is quite possible that some of the respondents to the questionnaire interpreted ‘shave biopsy’ as a full shave disk excision,” he said. “That said, intentional and routine partial sampling of suspected melanomas would be at odds with the guidelines.”
It is not inappropriate to remove a suspicious lesion, if small enough, with a punch biopsy, Dr. Tsao said, adding, “Perhaps again, the respondents failed to distinguish between partial punch biopsy and punch excision.”
On occasion, the AAD guidelines also make accommodations for an incisional biopsy “of the clinically or dermatoscopically most atypical portion of the lesion.” In this situation, a smaller incisional punch biopsy may be performed of the highly suspicious area.
“There are certainly areas that the investigators have identified which could represent potential knowledge gaps,” Dr. Tsao said. “For instance, 14% of the respondents used a 0.6 cm to 1.0 cm (6 mm-10 mm) margin for melanoma greater than 1 mm.”
Dermatologists’ surveillance of patients in the survey was somewhat less divergent. The most recommended follow-up interval for patients diagnosed within the last 5 years, was 6 months (49%), followed by a 3-month interval (25%), and other (24%). Follow-up was extended to yearly by 63% of dermatologists for patients diagnosed more than five years earlier, Dr. Farberg and Dr. Rigel reported.
Both the AAD and NCCN recommend follow-up every 3 to 12 months for those diagnosed with melanoma within the previous 5 years, while for those diagnosed more than 5 years prior, the AAD recommends follow-up every 3 to 12 months and the NCCN every 12 months.
“Adherence to evidence-based guidelines should lead to improved patient outcomes and quality of care. However, newer studies and emerging data may also justify deviations from existing guidelines, suggesting review of those guidelines may be indicated,” Dr. Farberg and Dr. Rigel concluded.
For example, a prospective series examining surgical margins in 1,072 patients with 1,120 malignant melanoma in situs showed that only 86% were successfully excised with a 6-mm margin, significantly less than the 98.9% clearance achieved with a 9-mm margin, Dr. Farberg told this publication.
That said, a 9-mm margin may not be appropriate for all lesions, such as those on the face, he observed.
The investigators said they hope the survey results will spur on revision of the guidelines, last updated in 2011.
Limitations of the survey include a lack of information on clinical factors such as patient history or anatomic site, the survey may have led to generalized answers, and access to care and reimbursement also may have impacted management, the authors noted.
The response rate to the survey was also low at 8%, with 6,177 practicing U.S. dermatologists surveyed. The demographics of the respondents, however, strongly reflected the full AAD membership, Dr. Farberg said. No significant geographical differences were observed.
CHICAGO – Significant variance exists in management of primary cutaneous melanoma, according to a national survey of 510 dermatologists.
Most dermatologists (36%) preferred a shave biopsy for lesions suspected of being melanoma, despite guidelines from the American Academy of Dermatology (AAD) and National Comprehensive Cancer Network (NCCN) guidelines that recommend narrow excision biopsy.
In all, 31% of dermatologists used a narrow local excision (less than 5 mm margin), 13% saucerization/scoop shave biopsy, 11%, punch biopsy, 3% wide local excision, and 7% other.
“The guidelines and academy are all very clear that one of the goals of the biopsy is to obtain tumor depth, so we were surprised that a significant number of providers use shave biopsy or other methods that may leave a risk of not getting the correct depth,” study co-author Dr. Aaron S. Farberg, a melanoma clinical research fellow at the National Society for Cutaneous Medicine in New York City, said in an interview.
Notably, dermatologists in academic and dermatology-based group practices were significantly less likely than those in multispecialty or solo practice to use narrow excision (23% vs. 42%; P < .001).
Although treatment for melanoma evolves continuously, the authors observed that dermatologists remain at the forefront of melanoma management and play a critical role in patient decision making.
“This study suggests that a knowledge gap may exist representing an educational opportunity to more effectively disseminate and implement recommended approaches,” Dr. Farberg and Dr. Darrell Rigel, of New York University School of Medicine, reported in a poster presentation at the annual meeting of the American Society for Dermatologic Surgery.
The survey also revealed that dermatologists are going beyond suggested surgical margins when excising melanoma.
For malignant melanoma in situ (MMIS), 62% used a 5 mm or less margin, 36% a 6 mm to 10 mm margin, and 2% a 1.1 cm to 1.9 cm margin. For these lesions, the AAD recommends a 0.5 cm-1 cm (5 mm-10 mm) margin and the NCCN a 0.5 cm margin, Drs. Farber and Rigel reported.
Academic dermatologists were significantly more likely than all other practice types to refer patients with MMIS out for excision (18% vs. 10%; P < .05).
For invasive melanoma less than 1 mm in depth, both the AAD and NCCN recommend a 1 cm margin (10 mm). In all, 61% of dermatologists reported using 6 mm to 10 mm margins, with 34% opting for 1.1 cm to 1.9 cm margins, 3% at least 2 cm margins, and 2% no more than 5 mm margins.
No significant difference was found across provider types for treatment of melanomas less than 1 mm in depth.
For invasive melanoma greater than 1 mm in depth, 54% of respondents used 1.1 cm to 1.9 cm margins, with most (67%) referring the patient to another provider. Both national guidelines recommend 1 cm to 2 cm margins for melanomas 1 mm to 2 mm in depth and 2 cm margins for melanoma greater than 2 mm in depth.
Academic dermatologists were significantly more likely than other dermatologists to treat these lesions rather than to refer the patient out (51% vs. 30%; P < .001).
“This is exciting new data that suggests that there still is a variance in early melanoma management,” Dr. Rigel, past president of AAD and ASDS, said in an interview. “The data suggest more studies need to be done to better access why this is occurring.”
Dr. Hensin Tsao, who served on the 2011 AAD guideline working group and is co-chairing the AAD’s pending guideline update, said in an interview that, “Dr. Rigel is well respected in the field and the project will undoubtedly be submitted for publication and subject to further review. It is worthwhile to wait on the final published results and conclusions.”
He agreed, however, with the authors’ suggestion that there is significant variation in practice. Regarding the finding that 36% of respondents use a shave biopsy for suspicious lesions, the AAD guidelines recommend that the entire lesion be removed with a 1 mm to 3 mm margin, which can be accomplished by an elliptical or punch excision with sutures or shave removal to a depth below the anticipated plane of the lesion, Dr. Tsao, of Massachusetts General Hospital in Boston, said.
“It is quite possible that some of the respondents to the questionnaire interpreted ‘shave biopsy’ as a full shave disk excision,” he said. “That said, intentional and routine partial sampling of suspected melanomas would be at odds with the guidelines.”
It is not inappropriate to remove a suspicious lesion, if small enough, with a punch biopsy, Dr. Tsao said, adding, “Perhaps again, the respondents failed to distinguish between partial punch biopsy and punch excision.”
On occasion, the AAD guidelines also make accommodations for an incisional biopsy “of the clinically or dermatoscopically most atypical portion of the lesion.” In this situation, a smaller incisional punch biopsy may be performed of the highly suspicious area.
“There are certainly areas that the investigators have identified which could represent potential knowledge gaps,” Dr. Tsao said. “For instance, 14% of the respondents used a 0.6 cm to 1.0 cm (6 mm-10 mm) margin for melanoma greater than 1 mm.”
Dermatologists’ surveillance of patients in the survey was somewhat less divergent. The most recommended follow-up interval for patients diagnosed within the last 5 years, was 6 months (49%), followed by a 3-month interval (25%), and other (24%). Follow-up was extended to yearly by 63% of dermatologists for patients diagnosed more than five years earlier, Dr. Farberg and Dr. Rigel reported.
Both the AAD and NCCN recommend follow-up every 3 to 12 months for those diagnosed with melanoma within the previous 5 years, while for those diagnosed more than 5 years prior, the AAD recommends follow-up every 3 to 12 months and the NCCN every 12 months.
“Adherence to evidence-based guidelines should lead to improved patient outcomes and quality of care. However, newer studies and emerging data may also justify deviations from existing guidelines, suggesting review of those guidelines may be indicated,” Dr. Farberg and Dr. Rigel concluded.
For example, a prospective series examining surgical margins in 1,072 patients with 1,120 malignant melanoma in situs showed that only 86% were successfully excised with a 6-mm margin, significantly less than the 98.9% clearance achieved with a 9-mm margin, Dr. Farberg told this publication.
That said, a 9-mm margin may not be appropriate for all lesions, such as those on the face, he observed.
The investigators said they hope the survey results will spur on revision of the guidelines, last updated in 2011.
Limitations of the survey include a lack of information on clinical factors such as patient history or anatomic site, the survey may have led to generalized answers, and access to care and reimbursement also may have impacted management, the authors noted.
The response rate to the survey was also low at 8%, with 6,177 practicing U.S. dermatologists surveyed. The demographics of the respondents, however, strongly reflected the full AAD membership, Dr. Farberg said. No significant geographical differences were observed.
AT THE ASDS ANNUAL MEETING
Key clinical point: Significant variance exists in dermatologists’ management of melanoma.
Major finding: Only 31% of dermatologists used narrow excision as recommended to biopsy suspicious lesions.
Data source: Survey of 510 U.S. practicing dermatologists.
Disclosures: The authors reported having no financial disclosures.
Immune-related events with checkpoint inhibitors are manageable
BOSTON– Immune-related adverse events associated with checkpoint inhibitor therapy are generally mild to moderate and transient, but some less common side effects can be serious or even fatal, according to an immunotherapy researcher.
“Rapid identification of these side effects and initiation of systemic immunosuppression can improve outcomes without compromising the efficacy of immune-checkpoint inhibition,” said Dr. Antoine Italiano from the Institut Bergonié in Bordeaux, France.
There is also evidence to suggest that immune-related adverse events (irAEs) associated with the programmed-death 1 (PD-1) inhibitors pembrolizumab (Keytruda) and nivolumab (Opdivo) may be predictive of favorable outcomes. In contrast, although there was early clinical evidence to suggest that adverse reactions to immune checkpoint inhibition with cytotoxic T-lymphocyte–associated protein 4 (anti-CTLA-4) antibodies such as ipilimumab (Yervoy) correlate with outcomes, more recent evidence suggests that toxicity with this class of agents is not predictive of efficacy, Dr. Italiano said at the AACR–NCI–EORTC International Conference on Molecular Targets and Cancer Therapeutics.
“Correlation between safety profile and outcome must be confirmed by further studies,” he said, adding that “further studies are also needed to identify patients at high risk of poor tolerability.”
Immune-related adverse events associated with anti-CTLA-4 therapy generally involve organ systems such as the skin, digestive tract, and endocrine system. Rare adverse events reported with these agents include renal injury, sarcoidosis, uveitis, and myelitis, among others.
The events tend to arise around 10 weeks of therapy, following three cycles with either ipilimumab or the investigational agent tremelimumab. Late-occurring events, defined as those that arise more than 70 days after the last infusion, are uncommon, occurring in less than 7% of patients.
Most irAEs seen with anti-CTLA-4 therapy are reversible within about 6 weeks, although some events, such as hypophysitis (autoimmune inflammation of the pituitary gland), can take significantly longer to resolve, Dr. Italiano said.
He cited a recent systematic review and meta-analysis showing that among patients treated with any anti-CTLA-4, the overall incidence of all-grade irAEs was 72%, and the overall incidence of high-grade irAEs was 24%. This study also showed that there was a dose-dependent risk of developing irAEs with ipilimumab, with the incidence of all grades of events at 61% for the 3 mg/kg dose, and 79% for the 10 mg/kg dose.
Two potential biomarkers for gastrointestinal irAEs, the neutrophil-activation markers CD177 and CEACAM1, were identified in a 2013 study. This finding suggests a possible role of neutrophils in ipilimumab-associated GI irAEs, Dr. Italiano noted.
Evidence from early clinical studies of ipilimumab in metastatic melanoma suggested that irAEs correlated with outcomes, but a study published in October 2015 seems to debunk this notion, showing that among 298 patients treated with ipilimumab, neither time to treatment failure nor overall survival were affected by the occurrence of irAEs, he added.
As to whether therapy with anti-CTLA-4 antibodies is safe for treatment of cancer for patients with autoimmune diseases or immunodeficient states, the jury is still out, because these patients were typically excluded from clinical trials.
“But there are a few recent case reports suggesting that treating patients with autoimmune disease with ipilimumab is safe and does not induce exacerbation of the symptoms of the underlying autoimmune disease,” Dr. Italiano said.
PD-1 inhibitors
Adverse events common to the PD-1 inhibitors pembrolizumab and nivolumab and occurring in more than 5% of patients with each include fatigue/asthenia, decreased appetite, diarrhea, rash, pruritus, nausea, and arthralgia.
In clinical trials of the agents for treatment of melanoma, vitiligo was the most common irAE, occurring in 7%-8% of patients. Other events, occurring in similar frequency across the various trials, included hypo- or hyperthyroidism, pneumonitis, colitis, hepatitis, renal failure/nephritis, uveitis/iritis, and hypophysitis.
The time to first occurrence and resolution of irAEs with the PD-1 inhibitors varies by organ system, with skin toxicity occurring within the few weeks of therapy, peaking at about 15 weeks, and resolving by about 25 weeks. Gastrointestinal toxicities crop up at about 10 weeks, but quickly resolve.
Among the less common (less than 10%) irAEs, hepatic and pulmonary events seen to occur around week 8 or 9 and resolve within 2-4 weeks, whereas endocrine events start showing up around week 10, peak at about 25 weeks, and resolve around 40 weeks.
Among patients treated with PD-1 inhibitors for non–small cell lung cancer, the adverse-event profile is similar to that seen in treatment of patients with melanoma, except for the absence of vitiligo, Dr. Italiano noted.
In contrast to the CTLA-4 inhibitors, irAEs seen with the PD-1 inhibitors, especially cutaneous events, appear to be associated with favorable outcomes.
For example, in a prospective, single-center observational study of pembrolizumab in 67 patients with metastatic melanoma, 17 developed vitiligo, and 12 of these patients had an objective response (18% complete and 53% partial responses). The objective response rate in this group was 71%, compared with 28% (14 of 50 patients) for those who did not develop vitiligo.
In a second, retrospective study of 83 patients enrolled in clinical trials of pembrolizumab for melanoma, non–small cell lung cancer, prostate cancer, and Merkel cell carcinoma, patients in each of three pembrolizumab dosing groups who developed cutaneous AEs had significantly longer progression-free intervals than patients who did not develop cutaneous AEs.
A similar correlation between cutaneous events with nivolumab and favorable outcomes was seen in a study of pooled data on 148 patients with resected or unresectable metastatic melanoma. The investigators found that both rash and vitiligo correlated significantly with better overall survival.
BOSTON– Immune-related adverse events associated with checkpoint inhibitor therapy are generally mild to moderate and transient, but some less common side effects can be serious or even fatal, according to an immunotherapy researcher.
“Rapid identification of these side effects and initiation of systemic immunosuppression can improve outcomes without compromising the efficacy of immune-checkpoint inhibition,” said Dr. Antoine Italiano from the Institut Bergonié in Bordeaux, France.
There is also evidence to suggest that immune-related adverse events (irAEs) associated with the programmed-death 1 (PD-1) inhibitors pembrolizumab (Keytruda) and nivolumab (Opdivo) may be predictive of favorable outcomes. In contrast, although there was early clinical evidence to suggest that adverse reactions to immune checkpoint inhibition with cytotoxic T-lymphocyte–associated protein 4 (anti-CTLA-4) antibodies such as ipilimumab (Yervoy) correlate with outcomes, more recent evidence suggests that toxicity with this class of agents is not predictive of efficacy, Dr. Italiano said at the AACR–NCI–EORTC International Conference on Molecular Targets and Cancer Therapeutics.
“Correlation between safety profile and outcome must be confirmed by further studies,” he said, adding that “further studies are also needed to identify patients at high risk of poor tolerability.”
Immune-related adverse events associated with anti-CTLA-4 therapy generally involve organ systems such as the skin, digestive tract, and endocrine system. Rare adverse events reported with these agents include renal injury, sarcoidosis, uveitis, and myelitis, among others.
The events tend to arise around 10 weeks of therapy, following three cycles with either ipilimumab or the investigational agent tremelimumab. Late-occurring events, defined as those that arise more than 70 days after the last infusion, are uncommon, occurring in less than 7% of patients.
Most irAEs seen with anti-CTLA-4 therapy are reversible within about 6 weeks, although some events, such as hypophysitis (autoimmune inflammation of the pituitary gland), can take significantly longer to resolve, Dr. Italiano said.
He cited a recent systematic review and meta-analysis showing that among patients treated with any anti-CTLA-4, the overall incidence of all-grade irAEs was 72%, and the overall incidence of high-grade irAEs was 24%. This study also showed that there was a dose-dependent risk of developing irAEs with ipilimumab, with the incidence of all grades of events at 61% for the 3 mg/kg dose, and 79% for the 10 mg/kg dose.
Two potential biomarkers for gastrointestinal irAEs, the neutrophil-activation markers CD177 and CEACAM1, were identified in a 2013 study. This finding suggests a possible role of neutrophils in ipilimumab-associated GI irAEs, Dr. Italiano noted.
Evidence from early clinical studies of ipilimumab in metastatic melanoma suggested that irAEs correlated with outcomes, but a study published in October 2015 seems to debunk this notion, showing that among 298 patients treated with ipilimumab, neither time to treatment failure nor overall survival were affected by the occurrence of irAEs, he added.
As to whether therapy with anti-CTLA-4 antibodies is safe for treatment of cancer for patients with autoimmune diseases or immunodeficient states, the jury is still out, because these patients were typically excluded from clinical trials.
“But there are a few recent case reports suggesting that treating patients with autoimmune disease with ipilimumab is safe and does not induce exacerbation of the symptoms of the underlying autoimmune disease,” Dr. Italiano said.
PD-1 inhibitors
Adverse events common to the PD-1 inhibitors pembrolizumab and nivolumab and occurring in more than 5% of patients with each include fatigue/asthenia, decreased appetite, diarrhea, rash, pruritus, nausea, and arthralgia.
In clinical trials of the agents for treatment of melanoma, vitiligo was the most common irAE, occurring in 7%-8% of patients. Other events, occurring in similar frequency across the various trials, included hypo- or hyperthyroidism, pneumonitis, colitis, hepatitis, renal failure/nephritis, uveitis/iritis, and hypophysitis.
The time to first occurrence and resolution of irAEs with the PD-1 inhibitors varies by organ system, with skin toxicity occurring within the few weeks of therapy, peaking at about 15 weeks, and resolving by about 25 weeks. Gastrointestinal toxicities crop up at about 10 weeks, but quickly resolve.
Among the less common (less than 10%) irAEs, hepatic and pulmonary events seen to occur around week 8 or 9 and resolve within 2-4 weeks, whereas endocrine events start showing up around week 10, peak at about 25 weeks, and resolve around 40 weeks.
Among patients treated with PD-1 inhibitors for non–small cell lung cancer, the adverse-event profile is similar to that seen in treatment of patients with melanoma, except for the absence of vitiligo, Dr. Italiano noted.
In contrast to the CTLA-4 inhibitors, irAEs seen with the PD-1 inhibitors, especially cutaneous events, appear to be associated with favorable outcomes.
For example, in a prospective, single-center observational study of pembrolizumab in 67 patients with metastatic melanoma, 17 developed vitiligo, and 12 of these patients had an objective response (18% complete and 53% partial responses). The objective response rate in this group was 71%, compared with 28% (14 of 50 patients) for those who did not develop vitiligo.
In a second, retrospective study of 83 patients enrolled in clinical trials of pembrolizumab for melanoma, non–small cell lung cancer, prostate cancer, and Merkel cell carcinoma, patients in each of three pembrolizumab dosing groups who developed cutaneous AEs had significantly longer progression-free intervals than patients who did not develop cutaneous AEs.
A similar correlation between cutaneous events with nivolumab and favorable outcomes was seen in a study of pooled data on 148 patients with resected or unresectable metastatic melanoma. The investigators found that both rash and vitiligo correlated significantly with better overall survival.
BOSTON– Immune-related adverse events associated with checkpoint inhibitor therapy are generally mild to moderate and transient, but some less common side effects can be serious or even fatal, according to an immunotherapy researcher.
“Rapid identification of these side effects and initiation of systemic immunosuppression can improve outcomes without compromising the efficacy of immune-checkpoint inhibition,” said Dr. Antoine Italiano from the Institut Bergonié in Bordeaux, France.
There is also evidence to suggest that immune-related adverse events (irAEs) associated with the programmed-death 1 (PD-1) inhibitors pembrolizumab (Keytruda) and nivolumab (Opdivo) may be predictive of favorable outcomes. In contrast, although there was early clinical evidence to suggest that adverse reactions to immune checkpoint inhibition with cytotoxic T-lymphocyte–associated protein 4 (anti-CTLA-4) antibodies such as ipilimumab (Yervoy) correlate with outcomes, more recent evidence suggests that toxicity with this class of agents is not predictive of efficacy, Dr. Italiano said at the AACR–NCI–EORTC International Conference on Molecular Targets and Cancer Therapeutics.
“Correlation between safety profile and outcome must be confirmed by further studies,” he said, adding that “further studies are also needed to identify patients at high risk of poor tolerability.”
Immune-related adverse events associated with anti-CTLA-4 therapy generally involve organ systems such as the skin, digestive tract, and endocrine system. Rare adverse events reported with these agents include renal injury, sarcoidosis, uveitis, and myelitis, among others.
The events tend to arise around 10 weeks of therapy, following three cycles with either ipilimumab or the investigational agent tremelimumab. Late-occurring events, defined as those that arise more than 70 days after the last infusion, are uncommon, occurring in less than 7% of patients.
Most irAEs seen with anti-CTLA-4 therapy are reversible within about 6 weeks, although some events, such as hypophysitis (autoimmune inflammation of the pituitary gland), can take significantly longer to resolve, Dr. Italiano said.
He cited a recent systematic review and meta-analysis showing that among patients treated with any anti-CTLA-4, the overall incidence of all-grade irAEs was 72%, and the overall incidence of high-grade irAEs was 24%. This study also showed that there was a dose-dependent risk of developing irAEs with ipilimumab, with the incidence of all grades of events at 61% for the 3 mg/kg dose, and 79% for the 10 mg/kg dose.
Two potential biomarkers for gastrointestinal irAEs, the neutrophil-activation markers CD177 and CEACAM1, were identified in a 2013 study. This finding suggests a possible role of neutrophils in ipilimumab-associated GI irAEs, Dr. Italiano noted.
Evidence from early clinical studies of ipilimumab in metastatic melanoma suggested that irAEs correlated with outcomes, but a study published in October 2015 seems to debunk this notion, showing that among 298 patients treated with ipilimumab, neither time to treatment failure nor overall survival were affected by the occurrence of irAEs, he added.
As to whether therapy with anti-CTLA-4 antibodies is safe for treatment of cancer for patients with autoimmune diseases or immunodeficient states, the jury is still out, because these patients were typically excluded from clinical trials.
“But there are a few recent case reports suggesting that treating patients with autoimmune disease with ipilimumab is safe and does not induce exacerbation of the symptoms of the underlying autoimmune disease,” Dr. Italiano said.
PD-1 inhibitors
Adverse events common to the PD-1 inhibitors pembrolizumab and nivolumab and occurring in more than 5% of patients with each include fatigue/asthenia, decreased appetite, diarrhea, rash, pruritus, nausea, and arthralgia.
In clinical trials of the agents for treatment of melanoma, vitiligo was the most common irAE, occurring in 7%-8% of patients. Other events, occurring in similar frequency across the various trials, included hypo- or hyperthyroidism, pneumonitis, colitis, hepatitis, renal failure/nephritis, uveitis/iritis, and hypophysitis.
The time to first occurrence and resolution of irAEs with the PD-1 inhibitors varies by organ system, with skin toxicity occurring within the few weeks of therapy, peaking at about 15 weeks, and resolving by about 25 weeks. Gastrointestinal toxicities crop up at about 10 weeks, but quickly resolve.
Among the less common (less than 10%) irAEs, hepatic and pulmonary events seen to occur around week 8 or 9 and resolve within 2-4 weeks, whereas endocrine events start showing up around week 10, peak at about 25 weeks, and resolve around 40 weeks.
Among patients treated with PD-1 inhibitors for non–small cell lung cancer, the adverse-event profile is similar to that seen in treatment of patients with melanoma, except for the absence of vitiligo, Dr. Italiano noted.
In contrast to the CTLA-4 inhibitors, irAEs seen with the PD-1 inhibitors, especially cutaneous events, appear to be associated with favorable outcomes.
For example, in a prospective, single-center observational study of pembrolizumab in 67 patients with metastatic melanoma, 17 developed vitiligo, and 12 of these patients had an objective response (18% complete and 53% partial responses). The objective response rate in this group was 71%, compared with 28% (14 of 50 patients) for those who did not develop vitiligo.
In a second, retrospective study of 83 patients enrolled in clinical trials of pembrolizumab for melanoma, non–small cell lung cancer, prostate cancer, and Merkel cell carcinoma, patients in each of three pembrolizumab dosing groups who developed cutaneous AEs had significantly longer progression-free intervals than patients who did not develop cutaneous AEs.
A similar correlation between cutaneous events with nivolumab and favorable outcomes was seen in a study of pooled data on 148 patients with resected or unresectable metastatic melanoma. The investigators found that both rash and vitiligo correlated significantly with better overall survival.
AT AACR–NCI–EORTC
Key clinical point: Inhibitors of CTLA-4 and PD-1/PD-L1 are associated with manageable immune-related adverse events.
Major finding: Cutaneous adverse events with PD-1 inhibitor therapy appear to be predictive of favorable outcomes.
Data source: Review of current knowledge of the immune-related adverse events associated with checkpoint inhibitors.
Disclosures: Dr. Italiano reported no conflicts of interest.
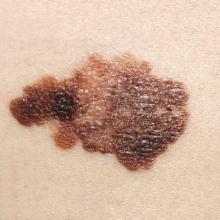
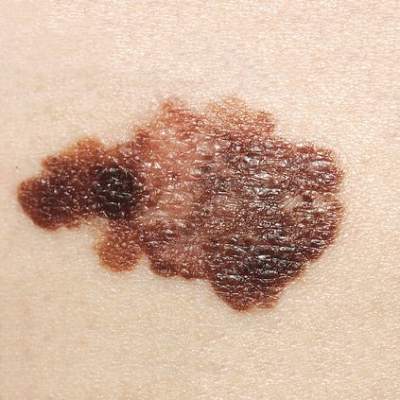
Desmoplastic Melanoma
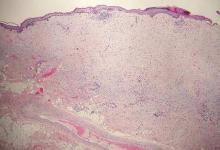
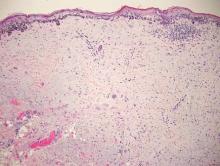
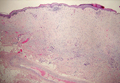
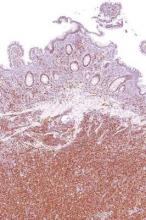
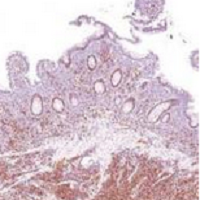
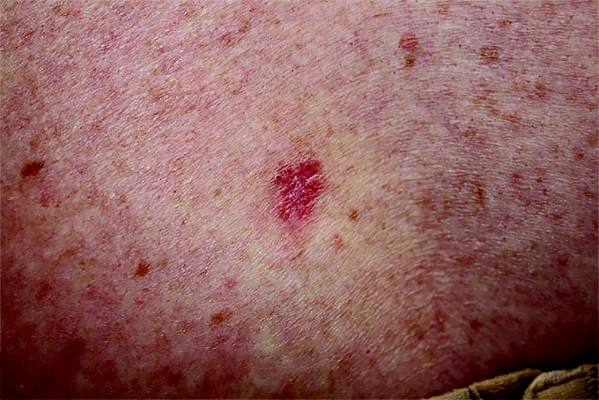
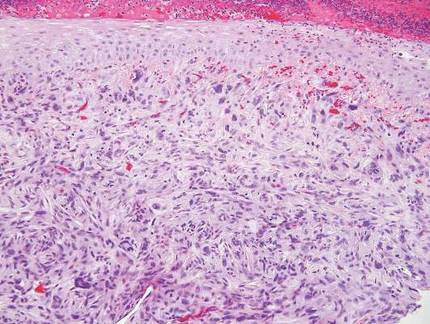
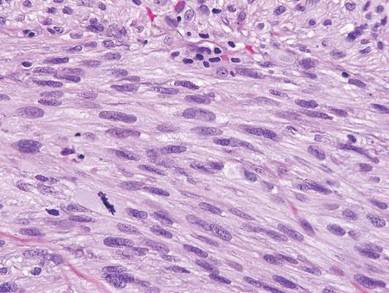
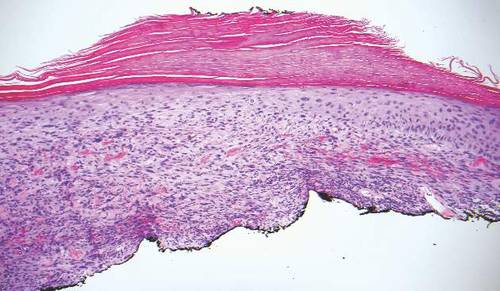
Desmoplastic melanoma, an uncommon variant of melanoma, poses a diagnostic challenge to the clinician because the tumors frequently appear as nonspecific flesh-colored or amelanotic plaques or nodules. They are more common in men than in women and are frequently found on the head and neck.1,2 Their innocuous appearance may lead to a delay in diagnosis and may explain why desmoplastic melanomas often are deeply infiltrative at the time of biopsy. Desmoplastic melanoma arises de novo in approximately one-third of cases.1 In the remainder of cases, it is seen in conjunction with overlying melanoma in situ, most commonly lentigo maligna melanoma.1 Histologically, desmoplastic melanomas are characterized by malignant spindle cells within a densely fibrotic stroma (Figure 1). Adjacent lymphoid aggregates and perineural involvement are common features,2 while pigment and atypical mitoses can be infrequent. Desmoplastic melanoma can be classified as mixed or pure based on the degree of desmoplasia and cellularity. Within mixed desmoplastic melanomas, there are areas that have histologic features of conventional melanomas while others demonstrate more typical desmoplastic characteristics. Pure desmoplastic melanoma has a higher degree of desmoplasia and fewer tumor cells than the mixed type.1 The pure subtype tends to be less aggressive and is less likely to metastasize to the lymph nodes.1 In the absence of an in situ component (Figure 2), desmoplastic melanoma may be indistinguishable from other spindle cell tumors on routine hematoxylin and eosin staining; thus, immunohistochemical staining generally is required. The most reliable stains in confirming a diagnosis of desmoplastic melanoma are S100 and SOX10 (SRY-related HMG-box 10)(Figure 3)(eTable).3
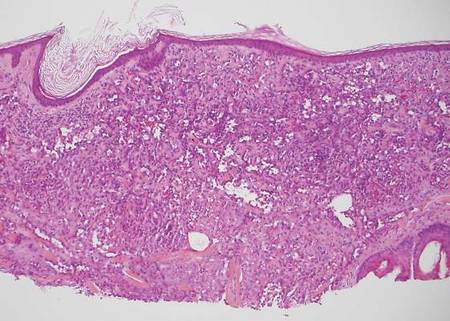
Atypical fibroxathoma typically presents as a nodule in the head and neck region or other sun-exposed areas in elderly individuals and is more commonly seen in men than in women.4 Histologically, atypical fibroxanthomas are composed of pleomorphic spindle, epithelioid, and multinucleated giant cells with numerous and atypical mitoses (Figure 4).5 Atypical fibroxanthoma is considered a diagnosis of exclusion; therefore, other dermal spindle cell tumors need to be ruled out before diagnosis can be made. Atypical fibroxanthomas generally stain negative for cytokeratin, S100, SOX10, and desmin, but in some cases there is positive focal staining for smooth muscle actin.4 Multiple immunohistochemical markers, including CD10, have shown reactivity in atypical fibroxanthomas,4 but none of these markers has a high specificity for this tumor; thus, it remains a diagnosis of exclusion.
Cutaneous angiosarcomas are aggressive tumors associated with a high mortality rate despite appropriate treatment with surgical resection and postoperative radiation treatment. They typically present as ecchymotic macules or nodules on the face or scalp of elderly patients.6,7 Ionizing radiation and chronic lymphedema are risk factors for cutaneous angiosarcoma.6 Histologically, well-differentiated cutaneous angiosarcomas are composed of irregular, anastomosing vascular channels that dissect through the dermis (Figure 5).6,7 Less well-differentiated tumors may contain spindle cells and lack obvious vascular structures; thus immunohistochemistry is essential for making the correct diagnosis in these cases. Cutaneous angiosarcomas typically stain positive for ERG (ETS-related gene) protein, CD31, CD34, and factor VIII.6,8 Unfortunately these tumors may also occasionally stain with cytokeratin, which may lead to the erroneous diagnosis of a carcinoma.6
| 
| |
| Figure 4. Pleomorphic spindle, epithelioid, and multinucleate giant cells with atypical mitoses filling the dermis in atypical fibroxanthoma (H&E, original magnification ×200). | Figure 5. Anastamosing vascular channels dissecting through collagen bundles and consuming the epidermis in cutaneous angiosarcoma (H&E, original magnification ×100). |
Cutaneous leiomyosarcoma is a smooth muscle neoplasm that arises from arrector pili muscles, genital smooth muscles, or vascular smooth muscles. It typically presents as a single plaque or nodule on the arms and legs of individuals older than 50 years of age.9 Cutaneous leiomyosarcomas can be classified as either dermal, in which at least 90% of the tumor is confined to the dermis, or subcutaneous; this distinction is important because the latter type has a higher rate of metastasis and a poorer prognosis.9 Because of this tumor’s smooth muscle derivation, well-differentiated tumors may retain features of typical smooth muscle cells, including cigar-shaped nuclei with adjacent glycogen vacuoles (Figure 6). If fascicle formation is observed, this may be an additional clue to the diagnosis. In poorly differentiated tumors, immunohistochemistry is invaluable. Leiomyosarcoma often stains positive for smooth muscle actin, muscle specific actin, h-caldesmon, desmin, and calponin.9-11
Spindle cell squamous cell carcinomas often present as ulcerated nodules on sun-exposed skin or on sites of prior ionizing radiation.2,12 Like desmoplastic melanoma, spindle cell squamous cell carcinomas are characterized by spindle cells in the dermis. Helpful diagnostic clues may include evidence of squamous differentiation, including keratin pearls or overlying actinic keratosis (Figure 7). However, actinic keratosis is common on sun-damaged skin and cannot be used to definitively confirm this diagnosis. There also may be areas of the tumor with more typical epithelioid cells that are easily identified as squamous cell carcinoma.2 Spindle cell squamous cell carcinoma stains positive for high–molecular weight cytokeratin antibodies and p63,2 which can help to differentiate it from the other spindle cell tumors in the differential.
| 
| |
| Figure 6. Spindle cells of leiomyosarcoma with cigar-shaped nuclei and adjacent glycogen vacuoles (H&E, original magnification ×600). | Figure 7. Spindle cell squamous cell carcinoma with overlying epidermal atypia that blends with the underlying dermal spindle cells (H&E, original magnification ×100). |
1. Chen LL, Jaimes N, Barker CA, et al. Desmoplastic melanoma: a review. J Am Acad Dermatol. 2013;68:825-833.
2. Calonje JE, Brenn T, Lazar AJ, et al. McKee’s Pathology of the Skin. 4th ed. St Louis, MO: Elsevier Saunders; 2012.
3. Elston DM, Ferringer TC, Ko C, et al. Dermatopathology: Requisites in Dermatology. 2nd ed. Philadelphia, PA: Saunders Elsevier; 2014.
4. Luzar B, Calonje E. Morphological and immunohistochemical characteristics of atypical fibroxanthoma with a special emphasis on potential diagnostic pitfalls: a review. J Cutan Pathol. 2010;37:301-309.
5. Iorizzo LJ III, Brown MD. Atypical fibroxanthoma: a review of the literature. Dermatol Surg. 2011;37:146-157.
6. Luca DR. Angiosarcoma, radiation-associated angiosarcoma, and atypical vascular lesion. Arch Pathol Lab Med. 2009;133:1804-1809.
7. Mendenhall WM, Mendenhall CM, Werning JW, et al. Cutaneous angiosarcoma. Am J Oncol. 2006;29:524-528.
8. Thum C, Husain EA, Mulholland K, et al. Atypical fibroxanthoma with pseudoangiomatous features: a histological and immunohistochemical mimic of cutaneous angiosarcoma. Ann Diagn Pathol. 2013;17:502-507.
9. Bolognia JL, Jorizzo JL, Shaffer JV. Dermatology. 3rd ed. Philadelphia, PA: Elsevier; 2012.
10. Hall BJ, Grossmann AH, Webber NP, et al. Atypical intradermal smooth muscle neoplasms (formerly cutaneous leiomyosarcomas): case series, immunohistochemical profile and review of the literature. Appl Immunohistochem Mol Morphol. 2013;21:132-138.
11. Perez-Montiel MD, Plaza JA, Dominguez-Malagon H, et al. Differential expression of smooth muscle myosin, smooth muscle actin, h-caldesmon, and calponin in the diagnosis of myofibroblastic and smooth muscle lesions of skin and soft tissue. Am J Dermatopathol. 2006;28:105-111.
12. Cassarino DS, DeRienzo DP, Barr RJ. Cutaneous squamous cell carcinoma: a comprehensive clinicopathologic classification. part one. J Cutan Pathol. 2006;33:191-205.
Desmoplastic melanoma, an uncommon variant of melanoma, poses a diagnostic challenge to the clinician because the tumors frequently appear as nonspecific flesh-colored or amelanotic plaques or nodules. They are more common in men than in women and are frequently found on the head and neck.1,2 Their innocuous appearance may lead to a delay in diagnosis and may explain why desmoplastic melanomas often are deeply infiltrative at the time of biopsy. Desmoplastic melanoma arises de novo in approximately one-third of cases.1 In the remainder of cases, it is seen in conjunction with overlying melanoma in situ, most commonly lentigo maligna melanoma.1 Histologically, desmoplastic melanomas are characterized by malignant spindle cells within a densely fibrotic stroma (Figure 1). Adjacent lymphoid aggregates and perineural involvement are common features,2 while pigment and atypical mitoses can be infrequent. Desmoplastic melanoma can be classified as mixed or pure based on the degree of desmoplasia and cellularity. Within mixed desmoplastic melanomas, there are areas that have histologic features of conventional melanomas while others demonstrate more typical desmoplastic characteristics. Pure desmoplastic melanoma has a higher degree of desmoplasia and fewer tumor cells than the mixed type.1 The pure subtype tends to be less aggressive and is less likely to metastasize to the lymph nodes.1 In the absence of an in situ component (Figure 2), desmoplastic melanoma may be indistinguishable from other spindle cell tumors on routine hematoxylin and eosin staining; thus, immunohistochemical staining generally is required. The most reliable stains in confirming a diagnosis of desmoplastic melanoma are S100 and SOX10 (SRY-related HMG-box 10)(Figure 3)(eTable).3
Atypical fibroxathoma typically presents as a nodule in the head and neck region or other sun-exposed areas in elderly individuals and is more commonly seen in men than in women.4 Histologically, atypical fibroxanthomas are composed of pleomorphic spindle, epithelioid, and multinucleated giant cells with numerous and atypical mitoses (Figure 4).5 Atypical fibroxanthoma is considered a diagnosis of exclusion; therefore, other dermal spindle cell tumors need to be ruled out before diagnosis can be made. Atypical fibroxanthomas generally stain negative for cytokeratin, S100, SOX10, and desmin, but in some cases there is positive focal staining for smooth muscle actin.4 Multiple immunohistochemical markers, including CD10, have shown reactivity in atypical fibroxanthomas,4 but none of these markers has a high specificity for this tumor; thus, it remains a diagnosis of exclusion.
Cutaneous angiosarcomas are aggressive tumors associated with a high mortality rate despite appropriate treatment with surgical resection and postoperative radiation treatment. They typically present as ecchymotic macules or nodules on the face or scalp of elderly patients.6,7 Ionizing radiation and chronic lymphedema are risk factors for cutaneous angiosarcoma.6 Histologically, well-differentiated cutaneous angiosarcomas are composed of irregular, anastomosing vascular channels that dissect through the dermis (Figure 5).6,7 Less well-differentiated tumors may contain spindle cells and lack obvious vascular structures; thus immunohistochemistry is essential for making the correct diagnosis in these cases. Cutaneous angiosarcomas typically stain positive for ERG (ETS-related gene) protein, CD31, CD34, and factor VIII.6,8 Unfortunately these tumors may also occasionally stain with cytokeratin, which may lead to the erroneous diagnosis of a carcinoma.6

| 
| |
| Figure 4. Pleomorphic spindle, epithelioid, and multinucleate giant cells with atypical mitoses filling the dermis in atypical fibroxanthoma (H&E, original magnification ×200). | Figure 5. Anastamosing vascular channels dissecting through collagen bundles and consuming the epidermis in cutaneous angiosarcoma (H&E, original magnification ×100). |
Cutaneous leiomyosarcoma is a smooth muscle neoplasm that arises from arrector pili muscles, genital smooth muscles, or vascular smooth muscles. It typically presents as a single plaque or nodule on the arms and legs of individuals older than 50 years of age.9 Cutaneous leiomyosarcomas can be classified as either dermal, in which at least 90% of the tumor is confined to the dermis, or subcutaneous; this distinction is important because the latter type has a higher rate of metastasis and a poorer prognosis.9 Because of this tumor’s smooth muscle derivation, well-differentiated tumors may retain features of typical smooth muscle cells, including cigar-shaped nuclei with adjacent glycogen vacuoles (Figure 6). If fascicle formation is observed, this may be an additional clue to the diagnosis. In poorly differentiated tumors, immunohistochemistry is invaluable. Leiomyosarcoma often stains positive for smooth muscle actin, muscle specific actin, h-caldesmon, desmin, and calponin.9-11
Spindle cell squamous cell carcinomas often present as ulcerated nodules on sun-exposed skin or on sites of prior ionizing radiation.2,12 Like desmoplastic melanoma, spindle cell squamous cell carcinomas are characterized by spindle cells in the dermis. Helpful diagnostic clues may include evidence of squamous differentiation, including keratin pearls or overlying actinic keratosis (Figure 7). However, actinic keratosis is common on sun-damaged skin and cannot be used to definitively confirm this diagnosis. There also may be areas of the tumor with more typical epithelioid cells that are easily identified as squamous cell carcinoma.2 Spindle cell squamous cell carcinoma stains positive for high–molecular weight cytokeratin antibodies and p63,2 which can help to differentiate it from the other spindle cell tumors in the differential.

| 
| |
| Figure 6. Spindle cells of leiomyosarcoma with cigar-shaped nuclei and adjacent glycogen vacuoles (H&E, original magnification ×600). | Figure 7. Spindle cell squamous cell carcinoma with overlying epidermal atypia that blends with the underlying dermal spindle cells (H&E, original magnification ×100). |
Desmoplastic melanoma, an uncommon variant of melanoma, poses a diagnostic challenge to the clinician because the tumors frequently appear as nonspecific flesh-colored or amelanotic plaques or nodules. They are more common in men than in women and are frequently found on the head and neck.1,2 Their innocuous appearance may lead to a delay in diagnosis and may explain why desmoplastic melanomas often are deeply infiltrative at the time of biopsy. Desmoplastic melanoma arises de novo in approximately one-third of cases.1 In the remainder of cases, it is seen in conjunction with overlying melanoma in situ, most commonly lentigo maligna melanoma.1 Histologically, desmoplastic melanomas are characterized by malignant spindle cells within a densely fibrotic stroma (Figure 1). Adjacent lymphoid aggregates and perineural involvement are common features,2 while pigment and atypical mitoses can be infrequent. Desmoplastic melanoma can be classified as mixed or pure based on the degree of desmoplasia and cellularity. Within mixed desmoplastic melanomas, there are areas that have histologic features of conventional melanomas while others demonstrate more typical desmoplastic characteristics. Pure desmoplastic melanoma has a higher degree of desmoplasia and fewer tumor cells than the mixed type.1 The pure subtype tends to be less aggressive and is less likely to metastasize to the lymph nodes.1 In the absence of an in situ component (Figure 2), desmoplastic melanoma may be indistinguishable from other spindle cell tumors on routine hematoxylin and eosin staining; thus, immunohistochemical staining generally is required. The most reliable stains in confirming a diagnosis of desmoplastic melanoma are S100 and SOX10 (SRY-related HMG-box 10)(Figure 3)(eTable).3
Atypical fibroxathoma typically presents as a nodule in the head and neck region or other sun-exposed areas in elderly individuals and is more commonly seen in men than in women.4 Histologically, atypical fibroxanthomas are composed of pleomorphic spindle, epithelioid, and multinucleated giant cells with numerous and atypical mitoses (Figure 4).5 Atypical fibroxanthoma is considered a diagnosis of exclusion; therefore, other dermal spindle cell tumors need to be ruled out before diagnosis can be made. Atypical fibroxanthomas generally stain negative for cytokeratin, S100, SOX10, and desmin, but in some cases there is positive focal staining for smooth muscle actin.4 Multiple immunohistochemical markers, including CD10, have shown reactivity in atypical fibroxanthomas,4 but none of these markers has a high specificity for this tumor; thus, it remains a diagnosis of exclusion.
Cutaneous angiosarcomas are aggressive tumors associated with a high mortality rate despite appropriate treatment with surgical resection and postoperative radiation treatment. They typically present as ecchymotic macules or nodules on the face or scalp of elderly patients.6,7 Ionizing radiation and chronic lymphedema are risk factors for cutaneous angiosarcoma.6 Histologically, well-differentiated cutaneous angiosarcomas are composed of irregular, anastomosing vascular channels that dissect through the dermis (Figure 5).6,7 Less well-differentiated tumors may contain spindle cells and lack obvious vascular structures; thus immunohistochemistry is essential for making the correct diagnosis in these cases. Cutaneous angiosarcomas typically stain positive for ERG (ETS-related gene) protein, CD31, CD34, and factor VIII.6,8 Unfortunately these tumors may also occasionally stain with cytokeratin, which may lead to the erroneous diagnosis of a carcinoma.6

| 
| |
| Figure 4. Pleomorphic spindle, epithelioid, and multinucleate giant cells with atypical mitoses filling the dermis in atypical fibroxanthoma (H&E, original magnification ×200). | Figure 5. Anastamosing vascular channels dissecting through collagen bundles and consuming the epidermis in cutaneous angiosarcoma (H&E, original magnification ×100). |
Cutaneous leiomyosarcoma is a smooth muscle neoplasm that arises from arrector pili muscles, genital smooth muscles, or vascular smooth muscles. It typically presents as a single plaque or nodule on the arms and legs of individuals older than 50 years of age.9 Cutaneous leiomyosarcomas can be classified as either dermal, in which at least 90% of the tumor is confined to the dermis, or subcutaneous; this distinction is important because the latter type has a higher rate of metastasis and a poorer prognosis.9 Because of this tumor’s smooth muscle derivation, well-differentiated tumors may retain features of typical smooth muscle cells, including cigar-shaped nuclei with adjacent glycogen vacuoles (Figure 6). If fascicle formation is observed, this may be an additional clue to the diagnosis. In poorly differentiated tumors, immunohistochemistry is invaluable. Leiomyosarcoma often stains positive for smooth muscle actin, muscle specific actin, h-caldesmon, desmin, and calponin.9-11
Spindle cell squamous cell carcinomas often present as ulcerated nodules on sun-exposed skin or on sites of prior ionizing radiation.2,12 Like desmoplastic melanoma, spindle cell squamous cell carcinomas are characterized by spindle cells in the dermis. Helpful diagnostic clues may include evidence of squamous differentiation, including keratin pearls or overlying actinic keratosis (Figure 7). However, actinic keratosis is common on sun-damaged skin and cannot be used to definitively confirm this diagnosis. There also may be areas of the tumor with more typical epithelioid cells that are easily identified as squamous cell carcinoma.2 Spindle cell squamous cell carcinoma stains positive for high–molecular weight cytokeratin antibodies and p63,2 which can help to differentiate it from the other spindle cell tumors in the differential.

| 
| |
| Figure 6. Spindle cells of leiomyosarcoma with cigar-shaped nuclei and adjacent glycogen vacuoles (H&E, original magnification ×600). | Figure 7. Spindle cell squamous cell carcinoma with overlying epidermal atypia that blends with the underlying dermal spindle cells (H&E, original magnification ×100). |
1. Chen LL, Jaimes N, Barker CA, et al. Desmoplastic melanoma: a review. J Am Acad Dermatol. 2013;68:825-833.
2. Calonje JE, Brenn T, Lazar AJ, et al. McKee’s Pathology of the Skin. 4th ed. St Louis, MO: Elsevier Saunders; 2012.
3. Elston DM, Ferringer TC, Ko C, et al. Dermatopathology: Requisites in Dermatology. 2nd ed. Philadelphia, PA: Saunders Elsevier; 2014.
4. Luzar B, Calonje E. Morphological and immunohistochemical characteristics of atypical fibroxanthoma with a special emphasis on potential diagnostic pitfalls: a review. J Cutan Pathol. 2010;37:301-309.
5. Iorizzo LJ III, Brown MD. Atypical fibroxanthoma: a review of the literature. Dermatol Surg. 2011;37:146-157.
6. Luca DR. Angiosarcoma, radiation-associated angiosarcoma, and atypical vascular lesion. Arch Pathol Lab Med. 2009;133:1804-1809.
7. Mendenhall WM, Mendenhall CM, Werning JW, et al. Cutaneous angiosarcoma. Am J Oncol. 2006;29:524-528.
8. Thum C, Husain EA, Mulholland K, et al. Atypical fibroxanthoma with pseudoangiomatous features: a histological and immunohistochemical mimic of cutaneous angiosarcoma. Ann Diagn Pathol. 2013;17:502-507.
9. Bolognia JL, Jorizzo JL, Shaffer JV. Dermatology. 3rd ed. Philadelphia, PA: Elsevier; 2012.
10. Hall BJ, Grossmann AH, Webber NP, et al. Atypical intradermal smooth muscle neoplasms (formerly cutaneous leiomyosarcomas): case series, immunohistochemical profile and review of the literature. Appl Immunohistochem Mol Morphol. 2013;21:132-138.
11. Perez-Montiel MD, Plaza JA, Dominguez-Malagon H, et al. Differential expression of smooth muscle myosin, smooth muscle actin, h-caldesmon, and calponin in the diagnosis of myofibroblastic and smooth muscle lesions of skin and soft tissue. Am J Dermatopathol. 2006;28:105-111.
12. Cassarino DS, DeRienzo DP, Barr RJ. Cutaneous squamous cell carcinoma: a comprehensive clinicopathologic classification. part one. J Cutan Pathol. 2006;33:191-205.
1. Chen LL, Jaimes N, Barker CA, et al. Desmoplastic melanoma: a review. J Am Acad Dermatol. 2013;68:825-833.
2. Calonje JE, Brenn T, Lazar AJ, et al. McKee’s Pathology of the Skin. 4th ed. St Louis, MO: Elsevier Saunders; 2012.
3. Elston DM, Ferringer TC, Ko C, et al. Dermatopathology: Requisites in Dermatology. 2nd ed. Philadelphia, PA: Saunders Elsevier; 2014.
4. Luzar B, Calonje E. Morphological and immunohistochemical characteristics of atypical fibroxanthoma with a special emphasis on potential diagnostic pitfalls: a review. J Cutan Pathol. 2010;37:301-309.
5. Iorizzo LJ III, Brown MD. Atypical fibroxanthoma: a review of the literature. Dermatol Surg. 2011;37:146-157.
6. Luca DR. Angiosarcoma, radiation-associated angiosarcoma, and atypical vascular lesion. Arch Pathol Lab Med. 2009;133:1804-1809.
7. Mendenhall WM, Mendenhall CM, Werning JW, et al. Cutaneous angiosarcoma. Am J Oncol. 2006;29:524-528.
8. Thum C, Husain EA, Mulholland K, et al. Atypical fibroxanthoma with pseudoangiomatous features: a histological and immunohistochemical mimic of cutaneous angiosarcoma. Ann Diagn Pathol. 2013;17:502-507.
9. Bolognia JL, Jorizzo JL, Shaffer JV. Dermatology. 3rd ed. Philadelphia, PA: Elsevier; 2012.
10. Hall BJ, Grossmann AH, Webber NP, et al. Atypical intradermal smooth muscle neoplasms (formerly cutaneous leiomyosarcomas): case series, immunohistochemical profile and review of the literature. Appl Immunohistochem Mol Morphol. 2013;21:132-138.
11. Perez-Montiel MD, Plaza JA, Dominguez-Malagon H, et al. Differential expression of smooth muscle myosin, smooth muscle actin, h-caldesmon, and calponin in the diagnosis of myofibroblastic and smooth muscle lesions of skin and soft tissue. Am J Dermatopathol. 2006;28:105-111.
12. Cassarino DS, DeRienzo DP, Barr RJ. Cutaneous squamous cell carcinoma: a comprehensive clinicopathologic classification. part one. J Cutan Pathol. 2006;33:191-205.
Successive gene mutations causing melanoma progression identified
Several successive genetic alterations have been identified that underlie the progression of melanoma from benign nevus to invasive malignancy, with different melanoma subtypes showing distinct evolutionary trajectories, according to a report published online Nov. 12 in the New England Journal of Medicine.
This discovery can serve as the basis for formulating refined criteria for diagnosing melanoma and predicting its clinical course. It also “revealed an intermediate category of melanocytic neoplasia, characterized by the presence of more than one pathogenic genetic alteration – a finding that helps to resolve the decades-long controversy about dysplastic nevi,” wrote A. Hunter Shain, Ph.D., of the departments of dermatology and pathology and the Helen Diller Family Comprehensive Cancer Center, University of California, San Francisco, and his associates.
Many of the pathogenic mutations in melanoma have already been catalogued, but the order in which they occur has never been defined before. “An individual tumor cannot be readily studied as it progresses from benign to malignant,” the investigators explained.
They approached the problem by comparing tissue samples from individual melanomas and melanoma precursor lesions across a spectrum of pathologic levels in different patients. They analyzed 37 melanocytic neoplasms archived at UCSF, St.John’s Hospital in London, and University Hospital Zurich, microdissecting 150 distinct areas of these samples for genetic sequencing. Eight dermatopathologists independently categorized the samples histologically as probably benign, intermediate but probably benign, intermediate but probably malignant, or malignant melanoma.
The investigators discovered “a more or less consistent pattern of genetic changes.” In every case, a single mutation was identified as the putative initiating oncogene. These were always mutations already known to activate the MAPK signaling pathway, usually in the BRAF or NRAS genes.
More advanced lesions harbored a broader spectrum of additional mutations that contributed to progression. Often these mutations affected the TERT promoter region, the CDKN2A gene and similar genes encoding particular protein subunits, or the tumor-suppressor ARID1A gene. A total of 77% of the areas examined in these intermediate tumors and melanomas in situ harbored TERT promoter mutations, which indicates that these mutations occur “at an unexpectedly early stage of the neoplastic progression,” Dr. Shain and his associates said (N Engl J Med. 2015 Nov 12. doi: 10.1056/NEJMoa1502583).
Only invasive melanomas were characterized by loss of both copies of the CDKN2A gene. Similarly, mutations in the SWI/SNF chromatin remodeling genes emerged primarily at the invasive stage. And losses in the PTEN and TP53 genes occurred exclusively in the thickest invasive melanomas, implying that these mutations occur late in the process of tumor progression. In addition, melanocytic neoplasms transitioned from linear to branched evolution only at later stages of progression, they said.
The investigators noted that “the existence of a category of lesions residing between clearly benign and clearly malignant states has long been proposed, but has remained controversial.” This study demonstrates that these intermediate neoplasms not only exist, but also carry discrete genetic alterations. “Detailed follow-up studies will be necessary to specifically delineate their histopathological characteristics and determine whether genetic or morphologic features can be identified that determine the risk of their progression to melanoma,” they added.
This study was supported by the National Institutes of Health and the Gerson and Barbara Bass Bakar Distinguished Professorship in Cancer Research. Dr. Shain reported having no relevant financial disclosures; one of his associates reported receiving grant support and personal fees from Novartis, Bristol-Myers Squibb, Roche, GlaxoSmithKline, Merck Sharp & Dohme, and Amgen.
Clinical observations and genetic studies like the one by Dr. Shain and his associates have illuminated cancer pathogenesis to a degree that was unimaginable just a short time ago, offering unprecedented opportunities for improved prevention and treatment.
It now appears that most cancers evolve in three broad phases. In the breakthrough phase, a cell acquires a mutation in a specific so-called driver gene and begins to proliferate abnormally. A lesion may or may not be detected at this phase, becoming apparent only after many cell divisions occur over many years. In the expansion phase, a spectrum of new driver-gene mutations arises and enables the cell to thrive in its local environment despite low levels of growth factors, nutrients, and oxygen. This constitutes the abnormal proliferation and disordered cellular architecture characteristic of benign tumors. In the invasive phase, subsequent mutations prompt cells to become malignant, invading normal tissues and growing in otherwise hostile environments. The more invasive-phase cells that are present, the more likely that one of them will succeed in moving to another organ and grow into a clinically important lesion there (metastasizing).
The clinical implications of this process are threefold. First, therapies for advanced cancer will work only if all of the patient’s metastatic lesions harbor a mutation targeted by those therapies. Second, it may be more effective to treat cancers on the basis of their genetic alterations rather than the cell type in which they arose. And third, the progeny of a cell that initiates the breakthrough phase can take decades to progress through the other phases and begin to metastasize because mutations are uncommon, even in tumors. This gives us a large window of time to derail the pathogenetic process.
Dr. Bert Vogelstein and Kenneth W. Kinzler, Ph.D., are with the Ludwig Center and the Howard Hughes Medical Institute at Sidney Kimmel Comprehensive Cancer Center, Johns Hopkins Medical Institutions, Baltimore. Dr. Vogelstein and Dr. Kinzler both reported receiving personal fees and other support from Personal Genome Diagnostics, PapGene, Sysmex Ionostics, and Morphotek. They made these remarks in a Perspective essay accompanying Dr. Shain’s report (N Engl J Med. 2015 Nov 12. doi: 10.1056/NEJMp1508811).
Clinical observations and genetic studies like the one by Dr. Shain and his associates have illuminated cancer pathogenesis to a degree that was unimaginable just a short time ago, offering unprecedented opportunities for improved prevention and treatment.
It now appears that most cancers evolve in three broad phases. In the breakthrough phase, a cell acquires a mutation in a specific so-called driver gene and begins to proliferate abnormally. A lesion may or may not be detected at this phase, becoming apparent only after many cell divisions occur over many years. In the expansion phase, a spectrum of new driver-gene mutations arises and enables the cell to thrive in its local environment despite low levels of growth factors, nutrients, and oxygen. This constitutes the abnormal proliferation and disordered cellular architecture characteristic of benign tumors. In the invasive phase, subsequent mutations prompt cells to become malignant, invading normal tissues and growing in otherwise hostile environments. The more invasive-phase cells that are present, the more likely that one of them will succeed in moving to another organ and grow into a clinically important lesion there (metastasizing).
The clinical implications of this process are threefold. First, therapies for advanced cancer will work only if all of the patient’s metastatic lesions harbor a mutation targeted by those therapies. Second, it may be more effective to treat cancers on the basis of their genetic alterations rather than the cell type in which they arose. And third, the progeny of a cell that initiates the breakthrough phase can take decades to progress through the other phases and begin to metastasize because mutations are uncommon, even in tumors. This gives us a large window of time to derail the pathogenetic process.
Dr. Bert Vogelstein and Kenneth W. Kinzler, Ph.D., are with the Ludwig Center and the Howard Hughes Medical Institute at Sidney Kimmel Comprehensive Cancer Center, Johns Hopkins Medical Institutions, Baltimore. Dr. Vogelstein and Dr. Kinzler both reported receiving personal fees and other support from Personal Genome Diagnostics, PapGene, Sysmex Ionostics, and Morphotek. They made these remarks in a Perspective essay accompanying Dr. Shain’s report (N Engl J Med. 2015 Nov 12. doi: 10.1056/NEJMp1508811).
Clinical observations and genetic studies like the one by Dr. Shain and his associates have illuminated cancer pathogenesis to a degree that was unimaginable just a short time ago, offering unprecedented opportunities for improved prevention and treatment.
It now appears that most cancers evolve in three broad phases. In the breakthrough phase, a cell acquires a mutation in a specific so-called driver gene and begins to proliferate abnormally. A lesion may or may not be detected at this phase, becoming apparent only after many cell divisions occur over many years. In the expansion phase, a spectrum of new driver-gene mutations arises and enables the cell to thrive in its local environment despite low levels of growth factors, nutrients, and oxygen. This constitutes the abnormal proliferation and disordered cellular architecture characteristic of benign tumors. In the invasive phase, subsequent mutations prompt cells to become malignant, invading normal tissues and growing in otherwise hostile environments. The more invasive-phase cells that are present, the more likely that one of them will succeed in moving to another organ and grow into a clinically important lesion there (metastasizing).
The clinical implications of this process are threefold. First, therapies for advanced cancer will work only if all of the patient’s metastatic lesions harbor a mutation targeted by those therapies. Second, it may be more effective to treat cancers on the basis of their genetic alterations rather than the cell type in which they arose. And third, the progeny of a cell that initiates the breakthrough phase can take decades to progress through the other phases and begin to metastasize because mutations are uncommon, even in tumors. This gives us a large window of time to derail the pathogenetic process.
Dr. Bert Vogelstein and Kenneth W. Kinzler, Ph.D., are with the Ludwig Center and the Howard Hughes Medical Institute at Sidney Kimmel Comprehensive Cancer Center, Johns Hopkins Medical Institutions, Baltimore. Dr. Vogelstein and Dr. Kinzler both reported receiving personal fees and other support from Personal Genome Diagnostics, PapGene, Sysmex Ionostics, and Morphotek. They made these remarks in a Perspective essay accompanying Dr. Shain’s report (N Engl J Med. 2015 Nov 12. doi: 10.1056/NEJMp1508811).
Several successive genetic alterations have been identified that underlie the progression of melanoma from benign nevus to invasive malignancy, with different melanoma subtypes showing distinct evolutionary trajectories, according to a report published online Nov. 12 in the New England Journal of Medicine.
This discovery can serve as the basis for formulating refined criteria for diagnosing melanoma and predicting its clinical course. It also “revealed an intermediate category of melanocytic neoplasia, characterized by the presence of more than one pathogenic genetic alteration – a finding that helps to resolve the decades-long controversy about dysplastic nevi,” wrote A. Hunter Shain, Ph.D., of the departments of dermatology and pathology and the Helen Diller Family Comprehensive Cancer Center, University of California, San Francisco, and his associates.
Many of the pathogenic mutations in melanoma have already been catalogued, but the order in which they occur has never been defined before. “An individual tumor cannot be readily studied as it progresses from benign to malignant,” the investigators explained.
They approached the problem by comparing tissue samples from individual melanomas and melanoma precursor lesions across a spectrum of pathologic levels in different patients. They analyzed 37 melanocytic neoplasms archived at UCSF, St.John’s Hospital in London, and University Hospital Zurich, microdissecting 150 distinct areas of these samples for genetic sequencing. Eight dermatopathologists independently categorized the samples histologically as probably benign, intermediate but probably benign, intermediate but probably malignant, or malignant melanoma.
The investigators discovered “a more or less consistent pattern of genetic changes.” In every case, a single mutation was identified as the putative initiating oncogene. These were always mutations already known to activate the MAPK signaling pathway, usually in the BRAF or NRAS genes.
More advanced lesions harbored a broader spectrum of additional mutations that contributed to progression. Often these mutations affected the TERT promoter region, the CDKN2A gene and similar genes encoding particular protein subunits, or the tumor-suppressor ARID1A gene. A total of 77% of the areas examined in these intermediate tumors and melanomas in situ harbored TERT promoter mutations, which indicates that these mutations occur “at an unexpectedly early stage of the neoplastic progression,” Dr. Shain and his associates said (N Engl J Med. 2015 Nov 12. doi: 10.1056/NEJMoa1502583).
Only invasive melanomas were characterized by loss of both copies of the CDKN2A gene. Similarly, mutations in the SWI/SNF chromatin remodeling genes emerged primarily at the invasive stage. And losses in the PTEN and TP53 genes occurred exclusively in the thickest invasive melanomas, implying that these mutations occur late in the process of tumor progression. In addition, melanocytic neoplasms transitioned from linear to branched evolution only at later stages of progression, they said.
The investigators noted that “the existence of a category of lesions residing between clearly benign and clearly malignant states has long been proposed, but has remained controversial.” This study demonstrates that these intermediate neoplasms not only exist, but also carry discrete genetic alterations. “Detailed follow-up studies will be necessary to specifically delineate their histopathological characteristics and determine whether genetic or morphologic features can be identified that determine the risk of their progression to melanoma,” they added.
This study was supported by the National Institutes of Health and the Gerson and Barbara Bass Bakar Distinguished Professorship in Cancer Research. Dr. Shain reported having no relevant financial disclosures; one of his associates reported receiving grant support and personal fees from Novartis, Bristol-Myers Squibb, Roche, GlaxoSmithKline, Merck Sharp & Dohme, and Amgen.
Several successive genetic alterations have been identified that underlie the progression of melanoma from benign nevus to invasive malignancy, with different melanoma subtypes showing distinct evolutionary trajectories, according to a report published online Nov. 12 in the New England Journal of Medicine.
This discovery can serve as the basis for formulating refined criteria for diagnosing melanoma and predicting its clinical course. It also “revealed an intermediate category of melanocytic neoplasia, characterized by the presence of more than one pathogenic genetic alteration – a finding that helps to resolve the decades-long controversy about dysplastic nevi,” wrote A. Hunter Shain, Ph.D., of the departments of dermatology and pathology and the Helen Diller Family Comprehensive Cancer Center, University of California, San Francisco, and his associates.
Many of the pathogenic mutations in melanoma have already been catalogued, but the order in which they occur has never been defined before. “An individual tumor cannot be readily studied as it progresses from benign to malignant,” the investigators explained.
They approached the problem by comparing tissue samples from individual melanomas and melanoma precursor lesions across a spectrum of pathologic levels in different patients. They analyzed 37 melanocytic neoplasms archived at UCSF, St.John’s Hospital in London, and University Hospital Zurich, microdissecting 150 distinct areas of these samples for genetic sequencing. Eight dermatopathologists independently categorized the samples histologically as probably benign, intermediate but probably benign, intermediate but probably malignant, or malignant melanoma.
The investigators discovered “a more or less consistent pattern of genetic changes.” In every case, a single mutation was identified as the putative initiating oncogene. These were always mutations already known to activate the MAPK signaling pathway, usually in the BRAF or NRAS genes.
More advanced lesions harbored a broader spectrum of additional mutations that contributed to progression. Often these mutations affected the TERT promoter region, the CDKN2A gene and similar genes encoding particular protein subunits, or the tumor-suppressor ARID1A gene. A total of 77% of the areas examined in these intermediate tumors and melanomas in situ harbored TERT promoter mutations, which indicates that these mutations occur “at an unexpectedly early stage of the neoplastic progression,” Dr. Shain and his associates said (N Engl J Med. 2015 Nov 12. doi: 10.1056/NEJMoa1502583).
Only invasive melanomas were characterized by loss of both copies of the CDKN2A gene. Similarly, mutations in the SWI/SNF chromatin remodeling genes emerged primarily at the invasive stage. And losses in the PTEN and TP53 genes occurred exclusively in the thickest invasive melanomas, implying that these mutations occur late in the process of tumor progression. In addition, melanocytic neoplasms transitioned from linear to branched evolution only at later stages of progression, they said.
The investigators noted that “the existence of a category of lesions residing between clearly benign and clearly malignant states has long been proposed, but has remained controversial.” This study demonstrates that these intermediate neoplasms not only exist, but also carry discrete genetic alterations. “Detailed follow-up studies will be necessary to specifically delineate their histopathological characteristics and determine whether genetic or morphologic features can be identified that determine the risk of their progression to melanoma,” they added.
This study was supported by the National Institutes of Health and the Gerson and Barbara Bass Bakar Distinguished Professorship in Cancer Research. Dr. Shain reported having no relevant financial disclosures; one of his associates reported receiving grant support and personal fees from Novartis, Bristol-Myers Squibb, Roche, GlaxoSmithKline, Merck Sharp & Dohme, and Amgen.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Key clinical point: Several successive genetic alterations have been identified that underlie the progression of melanoma from benign nevus to invasive malignancy.
Major finding: Every melanocytic neoplasia in this series originated with a single genetic mutation, progressed as a wide spectrum of other specific mutations emerged, and progressed further only in the presence of yet other specific mutations.
Data source: Genetic sequencing of 293 cancer-relevant genes in 150 areas of samples from 37 primary melanomas and their adjacent precursor lesions.
Disclosures: This study was supported by the National Institutes of Health and the Gerson and Barbara Bass Bakar Distinguished Professorship in Cancer Research. Dr. Shain reported having no relevant financial disclosures; one of his associates reported receiving grant support and personal fees from Novartis, Bristol-Myers Squibb, Roche, GlaxoSmithKline, Merck Sharp & Dohme, and Amgen.
Cobimetinib approved as add-on to vemurafenib for advanced melanoma
The Food and Drug Administration has approved the once-daily oral MEK inhibitor cobimetinib in combination with vemurafenib to treat patients with advanced melanoma whose tumors express the BRAF V600E or V600K mutation.
“Today’s approval provides a new targeted treatment that, when added to vemurafenib, demonstrates greater benefit than vemurafenib alone in patients with BRAF mutation–positive melanoma,” said Dr. Richard Pazdur, director of the Office of Hematology and Oncology Products in the FDA’s Center for Drug Evaluation and Research, in a press release announcing the approval.
In a pivotal clinical trial, treatment-naive individuals with unresectable, locally advanced or metastatic melanoma whose tumors were positive for BRAF V600E or V600K mutations were randomized to receive the BRAF inhibitor vemurafenib plus placebo (n = 248) or to receive vemurafenib with cobimetinib (n = 247). At the time of data cut-off in May, 2014, median progression-free survival (PFS) was significantly longer for the cobimetinib group (9.9 vs. 6.2 months, hazard ratio, 0.51; P less than .001); PFS was the primary outcome measure.
Of the secondary outcome measures, objective response was seen in two of three patients on cobimetinib, compared with fewer than half of the placebo arm patients (68% vs. 45%; P less than .001). Another secondary outcome measure, overall survival, could not be calculated because too few events had occurred at the end of data collection.
More grade 3 or higher adverse events occurred for patients in the cobimetinib arm than the placebo arm (65% vs. 59%), but the difference was not statistically significant. Patients taking cobimetinib were no more likely than those taking placebo to discontinue taking the study drug.
Side effects most commonly associated with the combination of vemurafenib and cobimetinib included diarrhea, photosensitivity, nausea, fever, and vomiting.
Individuals taking both a BRAF inhibitor and a MEK inhibitor were less likely to develop nonmelanoma secondary skin cancers, a complication that affects about 25% of those taking vemurafenib alone.
Vemurafenib blocks BRAF, part of a molecular signaling pathway implicated in melanoma tumor cell growth and division. Cobimetinib can help delay tumor resistance to vemurafenib by targeting MEK, a gene in the same signaling pathway.
The senior investigator of the pivotal clinical trial, Dr. Antoni Ribas, said in a press release, “Today’s approval is a significant advance in the treatment of metastatic melanoma.
“For patients with a BRAF-mutated melanoma, the combination has higher activity to shrink their tumors, and with less side effects than the drugs on their own,” said Dr. Ribas, a researcher at the University of California, Los Angeles, Jonsson Comprehensive Cancer Center.
The FDA reviewed cobimetinib under its priority review program and gave the drug an orphan drug designation.
Cobimetinib (Cotellic) and vemurafinib (Zelboraf) are both marketed by Genentech.
On Twitter @karioakes
This article was updated November 16, 2015.
The Food and Drug Administration has approved the once-daily oral MEK inhibitor cobimetinib in combination with vemurafenib to treat patients with advanced melanoma whose tumors express the BRAF V600E or V600K mutation.
“Today’s approval provides a new targeted treatment that, when added to vemurafenib, demonstrates greater benefit than vemurafenib alone in patients with BRAF mutation–positive melanoma,” said Dr. Richard Pazdur, director of the Office of Hematology and Oncology Products in the FDA’s Center for Drug Evaluation and Research, in a press release announcing the approval.
In a pivotal clinical trial, treatment-naive individuals with unresectable, locally advanced or metastatic melanoma whose tumors were positive for BRAF V600E or V600K mutations were randomized to receive the BRAF inhibitor vemurafenib plus placebo (n = 248) or to receive vemurafenib with cobimetinib (n = 247). At the time of data cut-off in May, 2014, median progression-free survival (PFS) was significantly longer for the cobimetinib group (9.9 vs. 6.2 months, hazard ratio, 0.51; P less than .001); PFS was the primary outcome measure.
Of the secondary outcome measures, objective response was seen in two of three patients on cobimetinib, compared with fewer than half of the placebo arm patients (68% vs. 45%; P less than .001). Another secondary outcome measure, overall survival, could not be calculated because too few events had occurred at the end of data collection.
More grade 3 or higher adverse events occurred for patients in the cobimetinib arm than the placebo arm (65% vs. 59%), but the difference was not statistically significant. Patients taking cobimetinib were no more likely than those taking placebo to discontinue taking the study drug.
Side effects most commonly associated with the combination of vemurafenib and cobimetinib included diarrhea, photosensitivity, nausea, fever, and vomiting.
Individuals taking both a BRAF inhibitor and a MEK inhibitor were less likely to develop nonmelanoma secondary skin cancers, a complication that affects about 25% of those taking vemurafenib alone.
Vemurafenib blocks BRAF, part of a molecular signaling pathway implicated in melanoma tumor cell growth and division. Cobimetinib can help delay tumor resistance to vemurafenib by targeting MEK, a gene in the same signaling pathway.
The senior investigator of the pivotal clinical trial, Dr. Antoni Ribas, said in a press release, “Today’s approval is a significant advance in the treatment of metastatic melanoma.
“For patients with a BRAF-mutated melanoma, the combination has higher activity to shrink their tumors, and with less side effects than the drugs on their own,” said Dr. Ribas, a researcher at the University of California, Los Angeles, Jonsson Comprehensive Cancer Center.
The FDA reviewed cobimetinib under its priority review program and gave the drug an orphan drug designation.
Cobimetinib (Cotellic) and vemurafinib (Zelboraf) are both marketed by Genentech.
On Twitter @karioakes
This article was updated November 16, 2015.
The Food and Drug Administration has approved the once-daily oral MEK inhibitor cobimetinib in combination with vemurafenib to treat patients with advanced melanoma whose tumors express the BRAF V600E or V600K mutation.
“Today’s approval provides a new targeted treatment that, when added to vemurafenib, demonstrates greater benefit than vemurafenib alone in patients with BRAF mutation–positive melanoma,” said Dr. Richard Pazdur, director of the Office of Hematology and Oncology Products in the FDA’s Center for Drug Evaluation and Research, in a press release announcing the approval.
In a pivotal clinical trial, treatment-naive individuals with unresectable, locally advanced or metastatic melanoma whose tumors were positive for BRAF V600E or V600K mutations were randomized to receive the BRAF inhibitor vemurafenib plus placebo (n = 248) or to receive vemurafenib with cobimetinib (n = 247). At the time of data cut-off in May, 2014, median progression-free survival (PFS) was significantly longer for the cobimetinib group (9.9 vs. 6.2 months, hazard ratio, 0.51; P less than .001); PFS was the primary outcome measure.
Of the secondary outcome measures, objective response was seen in two of three patients on cobimetinib, compared with fewer than half of the placebo arm patients (68% vs. 45%; P less than .001). Another secondary outcome measure, overall survival, could not be calculated because too few events had occurred at the end of data collection.
More grade 3 or higher adverse events occurred for patients in the cobimetinib arm than the placebo arm (65% vs. 59%), but the difference was not statistically significant. Patients taking cobimetinib were no more likely than those taking placebo to discontinue taking the study drug.
Side effects most commonly associated with the combination of vemurafenib and cobimetinib included diarrhea, photosensitivity, nausea, fever, and vomiting.
Individuals taking both a BRAF inhibitor and a MEK inhibitor were less likely to develop nonmelanoma secondary skin cancers, a complication that affects about 25% of those taking vemurafenib alone.
Vemurafenib blocks BRAF, part of a molecular signaling pathway implicated in melanoma tumor cell growth and division. Cobimetinib can help delay tumor resistance to vemurafenib by targeting MEK, a gene in the same signaling pathway.
The senior investigator of the pivotal clinical trial, Dr. Antoni Ribas, said in a press release, “Today’s approval is a significant advance in the treatment of metastatic melanoma.
“For patients with a BRAF-mutated melanoma, the combination has higher activity to shrink their tumors, and with less side effects than the drugs on their own,” said Dr. Ribas, a researcher at the University of California, Los Angeles, Jonsson Comprehensive Cancer Center.
The FDA reviewed cobimetinib under its priority review program and gave the drug an orphan drug designation.
Cobimetinib (Cotellic) and vemurafinib (Zelboraf) are both marketed by Genentech.
On Twitter @karioakes
This article was updated November 16, 2015.
Lenalidomide plus rituximab achieves 87% response rate
First-line combination biologic therapy with lenalidomide plus rituximab produced an 87% overall response rate in stage 3-4 mantle cell lymphoma, in an industry-sponsored, phase II clinical trial reported online Nov. 5 in the New England Journal of Medicine.
Mantle cell lymphoma is generally incurable, and patients have a median survival of 4-5 years. Initial therapy is usually very intensive, involving high-dose chemotherapy and hematopoietic cell transplantation. Since the malignancy primarily affects older adults who aren’t suitable candidates for intensive regimens, treatment “remains a clinical challenge,” said Dr. Jia Ruan of the Meyer Cancer Center and the division of biostatistics and epidemiology, Weill Cornell Medical College and New York-Presbyterian Hospital, New York, and her associates.
Reasoning that biologic therapy might offer effective disease control with fewer and less intense adverse effects, the investigators performed the open-label, single-group trial over a 3-year period. They treated 38 patients whose mean age was 65 years (range, 42-86 years), most of whom were at intermediate or high risk for imminent progression. These participants received a 12-cycle induction phase of lenalidomide plus rituximab, followed by a maintenance phase until disease progressed, unacceptable adverse effects developed, or patients withdrew from the study. The median follow-up was 30 months (range, 10-42 months).
The primary endpoint – overall response rate – was 87% in the intention-to-treat population, and the complete response rate was 61%. The number of complete responses increased over time with continuing treatment: the median time to a partial response was 3 months, and the median time to a complete response was 11 months. Two-year progression-free survival was 85%, and 2-year overall survival was 97%, the investigators said (New Engl. J. Med. 2015 Nov 5. doi: 10.1056/NEJMoa1505237).
Only eight patients showed progression of mantle cell lymphoma while taking lenalidomide plus rituximab, two of whom died from their disease. The other six patients responded to second-line therapy and remain alive, indicating that this first-line combination biologic therapy doesn’t compromise outcomes after subsequent treatments, Dr. Ruan and her associates said.
Almost as important as these favorable survival results were the findings concerning adverse effects. Scores on several quality-of-life measures either remained stable or improved throughout the induction and maintenance phases of treatment. As expected, grade 3 or 4 hematologic adverse effects included neutropenia (50% of patients), thrombocytopenia (13%), and anemia (11%), all of which resolved; grade 3 or 4 nonhematologic adverse effects included rash (29%), tumor flare (11%), serum sickness (8%), and fatigue (8%), all of which also resolved. All the serious infections that developed during the maintenance phase of treatment, which included pneumonia, cholangitis, and West Nile viral encephalitis, also resolved with antibiotics and supportive care.
Secondary cancers that developed during follow-up included two squamous cell skin cancers, one basal cell skin cancer, two cases of melanoma in situ, one Merkel cell carcinoma, and one pancreatic cancer.
“Our data show that a lower-intensity approach for initial therapy than that usually used in the case of patients with this cancer can be highly active, with durable responses observed in most patients,” Dr. Ruan and her associates said.
This study was supported by Celgene, maker of lenalidomide, and a Weill Cornell Medical College Clinical Translational Science Center grant. Dr. Ruan reported ties to Celgene, and her associates reported ties to numerous industry sources.
First-line combination biologic therapy with lenalidomide plus rituximab produced an 87% overall response rate in stage 3-4 mantle cell lymphoma, in an industry-sponsored, phase II clinical trial reported online Nov. 5 in the New England Journal of Medicine.
Mantle cell lymphoma is generally incurable, and patients have a median survival of 4-5 years. Initial therapy is usually very intensive, involving high-dose chemotherapy and hematopoietic cell transplantation. Since the malignancy primarily affects older adults who aren’t suitable candidates for intensive regimens, treatment “remains a clinical challenge,” said Dr. Jia Ruan of the Meyer Cancer Center and the division of biostatistics and epidemiology, Weill Cornell Medical College and New York-Presbyterian Hospital, New York, and her associates.
Reasoning that biologic therapy might offer effective disease control with fewer and less intense adverse effects, the investigators performed the open-label, single-group trial over a 3-year period. They treated 38 patients whose mean age was 65 years (range, 42-86 years), most of whom were at intermediate or high risk for imminent progression. These participants received a 12-cycle induction phase of lenalidomide plus rituximab, followed by a maintenance phase until disease progressed, unacceptable adverse effects developed, or patients withdrew from the study. The median follow-up was 30 months (range, 10-42 months).
The primary endpoint – overall response rate – was 87% in the intention-to-treat population, and the complete response rate was 61%. The number of complete responses increased over time with continuing treatment: the median time to a partial response was 3 months, and the median time to a complete response was 11 months. Two-year progression-free survival was 85%, and 2-year overall survival was 97%, the investigators said (New Engl. J. Med. 2015 Nov 5. doi: 10.1056/NEJMoa1505237).
Only eight patients showed progression of mantle cell lymphoma while taking lenalidomide plus rituximab, two of whom died from their disease. The other six patients responded to second-line therapy and remain alive, indicating that this first-line combination biologic therapy doesn’t compromise outcomes after subsequent treatments, Dr. Ruan and her associates said.
Almost as important as these favorable survival results were the findings concerning adverse effects. Scores on several quality-of-life measures either remained stable or improved throughout the induction and maintenance phases of treatment. As expected, grade 3 or 4 hematologic adverse effects included neutropenia (50% of patients), thrombocytopenia (13%), and anemia (11%), all of which resolved; grade 3 or 4 nonhematologic adverse effects included rash (29%), tumor flare (11%), serum sickness (8%), and fatigue (8%), all of which also resolved. All the serious infections that developed during the maintenance phase of treatment, which included pneumonia, cholangitis, and West Nile viral encephalitis, also resolved with antibiotics and supportive care.
Secondary cancers that developed during follow-up included two squamous cell skin cancers, one basal cell skin cancer, two cases of melanoma in situ, one Merkel cell carcinoma, and one pancreatic cancer.
“Our data show that a lower-intensity approach for initial therapy than that usually used in the case of patients with this cancer can be highly active, with durable responses observed in most patients,” Dr. Ruan and her associates said.
This study was supported by Celgene, maker of lenalidomide, and a Weill Cornell Medical College Clinical Translational Science Center grant. Dr. Ruan reported ties to Celgene, and her associates reported ties to numerous industry sources.
First-line combination biologic therapy with lenalidomide plus rituximab produced an 87% overall response rate in stage 3-4 mantle cell lymphoma, in an industry-sponsored, phase II clinical trial reported online Nov. 5 in the New England Journal of Medicine.
Mantle cell lymphoma is generally incurable, and patients have a median survival of 4-5 years. Initial therapy is usually very intensive, involving high-dose chemotherapy and hematopoietic cell transplantation. Since the malignancy primarily affects older adults who aren’t suitable candidates for intensive regimens, treatment “remains a clinical challenge,” said Dr. Jia Ruan of the Meyer Cancer Center and the division of biostatistics and epidemiology, Weill Cornell Medical College and New York-Presbyterian Hospital, New York, and her associates.
Reasoning that biologic therapy might offer effective disease control with fewer and less intense adverse effects, the investigators performed the open-label, single-group trial over a 3-year period. They treated 38 patients whose mean age was 65 years (range, 42-86 years), most of whom were at intermediate or high risk for imminent progression. These participants received a 12-cycle induction phase of lenalidomide plus rituximab, followed by a maintenance phase until disease progressed, unacceptable adverse effects developed, or patients withdrew from the study. The median follow-up was 30 months (range, 10-42 months).
The primary endpoint – overall response rate – was 87% in the intention-to-treat population, and the complete response rate was 61%. The number of complete responses increased over time with continuing treatment: the median time to a partial response was 3 months, and the median time to a complete response was 11 months. Two-year progression-free survival was 85%, and 2-year overall survival was 97%, the investigators said (New Engl. J. Med. 2015 Nov 5. doi: 10.1056/NEJMoa1505237).
Only eight patients showed progression of mantle cell lymphoma while taking lenalidomide plus rituximab, two of whom died from their disease. The other six patients responded to second-line therapy and remain alive, indicating that this first-line combination biologic therapy doesn’t compromise outcomes after subsequent treatments, Dr. Ruan and her associates said.
Almost as important as these favorable survival results were the findings concerning adverse effects. Scores on several quality-of-life measures either remained stable or improved throughout the induction and maintenance phases of treatment. As expected, grade 3 or 4 hematologic adverse effects included neutropenia (50% of patients), thrombocytopenia (13%), and anemia (11%), all of which resolved; grade 3 or 4 nonhematologic adverse effects included rash (29%), tumor flare (11%), serum sickness (8%), and fatigue (8%), all of which also resolved. All the serious infections that developed during the maintenance phase of treatment, which included pneumonia, cholangitis, and West Nile viral encephalitis, also resolved with antibiotics and supportive care.
Secondary cancers that developed during follow-up included two squamous cell skin cancers, one basal cell skin cancer, two cases of melanoma in situ, one Merkel cell carcinoma, and one pancreatic cancer.
“Our data show that a lower-intensity approach for initial therapy than that usually used in the case of patients with this cancer can be highly active, with durable responses observed in most patients,” Dr. Ruan and her associates said.
This study was supported by Celgene, maker of lenalidomide, and a Weill Cornell Medical College Clinical Translational Science Center grant. Dr. Ruan reported ties to Celgene, and her associates reported ties to numerous industry sources.
FROM NEW ENGLAND JOURNAL OF MEDICINE
Key clinical point: First-line combination biologic therapy with lenalidomide plus rituximab produced an 87% overall response rate in stage 3 or 4 mantle cell lymphoma.
Major finding: The primary endpoint – overall response rate – was 87%, and the complete response rate was 61%.
Data source: A multicenter, industry-sponsored, open-label, phase II study involving 38 patients with mantle cell lymphoma followed for a median of 30 months.
Disclosures: This study was supported by Celgene, maker of lenalidomide, and a Weill Cornell Medical College Clinical Translational Science Center grant. Dr. Ruan reported ties to Celgene, and her associates reported ties to numerous industry sources.
Higher risk of basal cell carcinoma linked to menopausal hormone therapy
Late age of natural menopause and use of menopausal hormone therapy were associated with significantly increased risk of basal cell carcinoma (BCC), according to results of a large, nationwide cohort study.
Women who experienced menopause at age 55 years or later, compared with age 50-54 years, had higher BCC risk (hazard ratio, 1.50; 95% confidence interval, 1.04-2.17; P for trend = .017). Among all women, those who had used menopausal hormone therapy had a higher risk (HR, 1.16; 95% confidence interval, 1.03-1.30) (J Clin Oncol. 2015 Nov 2. doi: 10.1200/JCO.2015.62.0625).
The findings are consistent with the hypothesis that exposure to endogenous and exogenous estrogens is photocarcinogenic, according to Dr. Elizabeth Cahoon, research fellow in the Radiation Epidemiology Branch of the National Cancer Institute, Bethesda, Md., and colleagues. Women with the highest observed BCC risk were those who experienced natural menopause and used menopausal hormone therapy for 10 or more years (HR, 1.97; 95% CI, 1.35-2.87; P for trend less than .001).
“In contrast to surgical menopause, which is associated with an immediate and dramatic drop in estrogen levels, women undergoing natural menopause may be exposed to a nontrivial amount of total and unopposed estrogen during the menopausal transition,” they wrote. “In this group, the combination of endogenous and exogenous estrogens may have increased the cumulative estrogen exposure (and, thus, the cumulative phototoxic effects).
The prospective study included 46,100 female, white participants of the US Radiologic Technologists Study, an occupational cohort composed of radiologic technologists. All participants were cancer free at baseline, and 4% had an incident BCC during the approximately 10-year follow-up.
Results showed no association between oral contraceptives and BCC, which may be due a to lower susceptibility in young adults to photosensitizing medications or to the lower doses typical of oral contraceptives. The ethinyl estradiol level in oral contraceptives typically ranges from 20 to 50 mcg, compared with a typical equine estrogen dose in menopausal therapy of 625 mcg.
Most reproductive factors evaluated, including age at menarche, number of live births, and age at first birth, showed no association with BCC.
Dr. Cahoon and coauthors reported having no disclosures.
Late age of natural menopause and use of menopausal hormone therapy were associated with significantly increased risk of basal cell carcinoma (BCC), according to results of a large, nationwide cohort study.
Women who experienced menopause at age 55 years or later, compared with age 50-54 years, had higher BCC risk (hazard ratio, 1.50; 95% confidence interval, 1.04-2.17; P for trend = .017). Among all women, those who had used menopausal hormone therapy had a higher risk (HR, 1.16; 95% confidence interval, 1.03-1.30) (J Clin Oncol. 2015 Nov 2. doi: 10.1200/JCO.2015.62.0625).
The findings are consistent with the hypothesis that exposure to endogenous and exogenous estrogens is photocarcinogenic, according to Dr. Elizabeth Cahoon, research fellow in the Radiation Epidemiology Branch of the National Cancer Institute, Bethesda, Md., and colleagues. Women with the highest observed BCC risk were those who experienced natural menopause and used menopausal hormone therapy for 10 or more years (HR, 1.97; 95% CI, 1.35-2.87; P for trend less than .001).
“In contrast to surgical menopause, which is associated with an immediate and dramatic drop in estrogen levels, women undergoing natural menopause may be exposed to a nontrivial amount of total and unopposed estrogen during the menopausal transition,” they wrote. “In this group, the combination of endogenous and exogenous estrogens may have increased the cumulative estrogen exposure (and, thus, the cumulative phototoxic effects).
The prospective study included 46,100 female, white participants of the US Radiologic Technologists Study, an occupational cohort composed of radiologic technologists. All participants were cancer free at baseline, and 4% had an incident BCC during the approximately 10-year follow-up.
Results showed no association between oral contraceptives and BCC, which may be due a to lower susceptibility in young adults to photosensitizing medications or to the lower doses typical of oral contraceptives. The ethinyl estradiol level in oral contraceptives typically ranges from 20 to 50 mcg, compared with a typical equine estrogen dose in menopausal therapy of 625 mcg.
Most reproductive factors evaluated, including age at menarche, number of live births, and age at first birth, showed no association with BCC.
Dr. Cahoon and coauthors reported having no disclosures.
Late age of natural menopause and use of menopausal hormone therapy were associated with significantly increased risk of basal cell carcinoma (BCC), according to results of a large, nationwide cohort study.
Women who experienced menopause at age 55 years or later, compared with age 50-54 years, had higher BCC risk (hazard ratio, 1.50; 95% confidence interval, 1.04-2.17; P for trend = .017). Among all women, those who had used menopausal hormone therapy had a higher risk (HR, 1.16; 95% confidence interval, 1.03-1.30) (J Clin Oncol. 2015 Nov 2. doi: 10.1200/JCO.2015.62.0625).
The findings are consistent with the hypothesis that exposure to endogenous and exogenous estrogens is photocarcinogenic, according to Dr. Elizabeth Cahoon, research fellow in the Radiation Epidemiology Branch of the National Cancer Institute, Bethesda, Md., and colleagues. Women with the highest observed BCC risk were those who experienced natural menopause and used menopausal hormone therapy for 10 or more years (HR, 1.97; 95% CI, 1.35-2.87; P for trend less than .001).
“In contrast to surgical menopause, which is associated with an immediate and dramatic drop in estrogen levels, women undergoing natural menopause may be exposed to a nontrivial amount of total and unopposed estrogen during the menopausal transition,” they wrote. “In this group, the combination of endogenous and exogenous estrogens may have increased the cumulative estrogen exposure (and, thus, the cumulative phototoxic effects).
The prospective study included 46,100 female, white participants of the US Radiologic Technologists Study, an occupational cohort composed of radiologic technologists. All participants were cancer free at baseline, and 4% had an incident BCC during the approximately 10-year follow-up.
Results showed no association between oral contraceptives and BCC, which may be due a to lower susceptibility in young adults to photosensitizing medications or to the lower doses typical of oral contraceptives. The ethinyl estradiol level in oral contraceptives typically ranges from 20 to 50 mcg, compared with a typical equine estrogen dose in menopausal therapy of 625 mcg.
Most reproductive factors evaluated, including age at menarche, number of live births, and age at first birth, showed no association with BCC.
Dr. Cahoon and coauthors reported having no disclosures.
Key clinical point: Risk of basal cell carcinoma was higher in women with late age of natural menopause and in women who used menopausal hormone therapy.
Major finding: Women who experienced menopause at a late age (55 years or older vs. 50-54 years) had higher BCC risk (HR, 1.50; 95% CI, 1.04-2.17; P for trend = .017), as did women who had ever used menopausal hormone therapy (HR, 1.16; 95% CI, 1.03-1.30).
Data source: The prospective cohort included 46,100 female, white participants of the US Radiologic Technologists Study who were cancer free at baseline; 4% had an incident BCC during the approximately 10-year follow-up.
Disclosures: Dr. Cahoon and coauthors reported having no disclosures.
ASTRO: Combined radioimmunotherapy proves promising in metastatic melanoma
SAN ANTONIO – The combination of the checkpoint inhibitor ipilimumab with palliative doses of radiation therapy was not associated with any unexpected toxicities, and half of the patients with metastatic melanoma who were treated showed a promising clinical response in a phase II study.
The trial included 22 patients with stage IV disease aged a median of 62 years, of whom 11 (50%) responded to the immunotherapy.
So far 3 (14%) of the 11 patients who responded have achieved a complete response (CR) at a median 55 weeks’ follow-up, and 3 (14%) had a partial response at a median 40 weeks’ follow-up, reported the lead study investigator, Dr. Susan Hiniker of Stanford University, during a press briefing at the annual meeting of the American Society for Radiation Oncology. The remaining five (23%) patients who responded had stable disease at a median 39 weeks’ follow-up.
The trial is one of the first prospective studies to report on the combined use of radiation with systemic immunotherapy in this patient population, she observed, noting that the idea behind the combination was that radiation might enhance the immunogenicity of tumors, that may then in turn make systemic immunotherapy more potent.
“Multiple trials have shown that there is still a minority of patients who respond to ipilimumab, with response rates of approximately 15%”, Dr. Hiniker said.
“As such, new methods of potentiating ipilimumab-induced responses are needed,” she added. “Local irradiation can modulate the local tumor environment such as to promote immune responses in a number of ways,” and other preliminary data have suggested that it may be able to increase overall response rates to ipilimumab.
Patients were recruited for the trial if they had stage IV melanoma and were aged between 18 and 65 years. Treatment consisted of palliative radiation delivered to one or two metastatic sites and at least one nonmetastatic site within 5 days of initial immunotherapy treatment with ipilimumab at a dose of 3 mg/kg every 3 weeks for a total of four treatment cycles. The dose and fractionation schedule was at the discretion of the treating physician.
Tumors were assessed for response using Response Evaluation Criteria in Solid Tumors (RECIST) and Immune Response Criteria (IRC) at baseline, 2-4 weeks after the last dose of ipilimumab was given, and then every 3 months thereafter until disease progression.
This being a phase II trial, the primary objective was to look at the safety of the approach; no additional or unexpected toxicities were seen.
“We saw approximately a 14% rate of grade 3-4 toxicity, which is really on par with what has been reported for ipilimumab monotherapy – on the order of 20%,” Dr. Hiniker said in an interview. “But that being said, we don’t believe that radiation itself caused any additional toxicity.”
One of the patients did have a perforated colon, she acknowledged, and for this reason future research may look at the combination of radiation therapy with the PD-1 (programmed T-cell death) inhibitors.
“I think right now everyone is so excited about the PD-1 inhibitors, and think that they’re safer,” Dr. Hiniker suggested. “It’s hard to give someone ipilimumab alone as the first line at this point; although many patients may not experience toxicity, [in] the ones that do, it can be severe.”
Dr. Hiniker also reported that IRC (independent review committee) analysis results suggested that several proinflammatory cytokines were elevated in some patients who responded to treatment. Higher levels of interleukin 2 at baseline, and at two time points during the study, for example, were associated with better response rates than lower levels. The findings also suggested that there might be a relationship between an increased number of elevated CD8-positive T cells and response.
“I think that we will find biomarkers, but we are not there yet, and it would need to be confirmed in a longer, larger study,” Dr. Hiniker said.
From a practical perspective, if a patient is already being treated with ipilimumab, and a physician is considering radiotherapy, then these results suggest that there will be no additional toxicity, she suggested.
“This combination approach does appear to be promising,” she said. “So for a subset of carefully selected patients – and the problem is we don’t totally know how to select them yet – this can be extremely promising.”
Dr. Hiniker had no disclosures to report.
SAN ANTONIO – The combination of the checkpoint inhibitor ipilimumab with palliative doses of radiation therapy was not associated with any unexpected toxicities, and half of the patients with metastatic melanoma who were treated showed a promising clinical response in a phase II study.
The trial included 22 patients with stage IV disease aged a median of 62 years, of whom 11 (50%) responded to the immunotherapy.
So far 3 (14%) of the 11 patients who responded have achieved a complete response (CR) at a median 55 weeks’ follow-up, and 3 (14%) had a partial response at a median 40 weeks’ follow-up, reported the lead study investigator, Dr. Susan Hiniker of Stanford University, during a press briefing at the annual meeting of the American Society for Radiation Oncology. The remaining five (23%) patients who responded had stable disease at a median 39 weeks’ follow-up.
The trial is one of the first prospective studies to report on the combined use of radiation with systemic immunotherapy in this patient population, she observed, noting that the idea behind the combination was that radiation might enhance the immunogenicity of tumors, that may then in turn make systemic immunotherapy more potent.
“Multiple trials have shown that there is still a minority of patients who respond to ipilimumab, with response rates of approximately 15%”, Dr. Hiniker said.
“As such, new methods of potentiating ipilimumab-induced responses are needed,” she added. “Local irradiation can modulate the local tumor environment such as to promote immune responses in a number of ways,” and other preliminary data have suggested that it may be able to increase overall response rates to ipilimumab.
Patients were recruited for the trial if they had stage IV melanoma and were aged between 18 and 65 years. Treatment consisted of palliative radiation delivered to one or two metastatic sites and at least one nonmetastatic site within 5 days of initial immunotherapy treatment with ipilimumab at a dose of 3 mg/kg every 3 weeks for a total of four treatment cycles. The dose and fractionation schedule was at the discretion of the treating physician.
Tumors were assessed for response using Response Evaluation Criteria in Solid Tumors (RECIST) and Immune Response Criteria (IRC) at baseline, 2-4 weeks after the last dose of ipilimumab was given, and then every 3 months thereafter until disease progression.
This being a phase II trial, the primary objective was to look at the safety of the approach; no additional or unexpected toxicities were seen.
“We saw approximately a 14% rate of grade 3-4 toxicity, which is really on par with what has been reported for ipilimumab monotherapy – on the order of 20%,” Dr. Hiniker said in an interview. “But that being said, we don’t believe that radiation itself caused any additional toxicity.”
One of the patients did have a perforated colon, she acknowledged, and for this reason future research may look at the combination of radiation therapy with the PD-1 (programmed T-cell death) inhibitors.
“I think right now everyone is so excited about the PD-1 inhibitors, and think that they’re safer,” Dr. Hiniker suggested. “It’s hard to give someone ipilimumab alone as the first line at this point; although many patients may not experience toxicity, [in] the ones that do, it can be severe.”
Dr. Hiniker also reported that IRC (independent review committee) analysis results suggested that several proinflammatory cytokines were elevated in some patients who responded to treatment. Higher levels of interleukin 2 at baseline, and at two time points during the study, for example, were associated with better response rates than lower levels. The findings also suggested that there might be a relationship between an increased number of elevated CD8-positive T cells and response.
“I think that we will find biomarkers, but we are not there yet, and it would need to be confirmed in a longer, larger study,” Dr. Hiniker said.
From a practical perspective, if a patient is already being treated with ipilimumab, and a physician is considering radiotherapy, then these results suggest that there will be no additional toxicity, she suggested.
“This combination approach does appear to be promising,” she said. “So for a subset of carefully selected patients – and the problem is we don’t totally know how to select them yet – this can be extremely promising.”
Dr. Hiniker had no disclosures to report.
SAN ANTONIO – The combination of the checkpoint inhibitor ipilimumab with palliative doses of radiation therapy was not associated with any unexpected toxicities, and half of the patients with metastatic melanoma who were treated showed a promising clinical response in a phase II study.
The trial included 22 patients with stage IV disease aged a median of 62 years, of whom 11 (50%) responded to the immunotherapy.
So far 3 (14%) of the 11 patients who responded have achieved a complete response (CR) at a median 55 weeks’ follow-up, and 3 (14%) had a partial response at a median 40 weeks’ follow-up, reported the lead study investigator, Dr. Susan Hiniker of Stanford University, during a press briefing at the annual meeting of the American Society for Radiation Oncology. The remaining five (23%) patients who responded had stable disease at a median 39 weeks’ follow-up.
The trial is one of the first prospective studies to report on the combined use of radiation with systemic immunotherapy in this patient population, she observed, noting that the idea behind the combination was that radiation might enhance the immunogenicity of tumors, that may then in turn make systemic immunotherapy more potent.
“Multiple trials have shown that there is still a minority of patients who respond to ipilimumab, with response rates of approximately 15%”, Dr. Hiniker said.
“As such, new methods of potentiating ipilimumab-induced responses are needed,” she added. “Local irradiation can modulate the local tumor environment such as to promote immune responses in a number of ways,” and other preliminary data have suggested that it may be able to increase overall response rates to ipilimumab.
Patients were recruited for the trial if they had stage IV melanoma and were aged between 18 and 65 years. Treatment consisted of palliative radiation delivered to one or two metastatic sites and at least one nonmetastatic site within 5 days of initial immunotherapy treatment with ipilimumab at a dose of 3 mg/kg every 3 weeks for a total of four treatment cycles. The dose and fractionation schedule was at the discretion of the treating physician.
Tumors were assessed for response using Response Evaluation Criteria in Solid Tumors (RECIST) and Immune Response Criteria (IRC) at baseline, 2-4 weeks after the last dose of ipilimumab was given, and then every 3 months thereafter until disease progression.
This being a phase II trial, the primary objective was to look at the safety of the approach; no additional or unexpected toxicities were seen.
“We saw approximately a 14% rate of grade 3-4 toxicity, which is really on par with what has been reported for ipilimumab monotherapy – on the order of 20%,” Dr. Hiniker said in an interview. “But that being said, we don’t believe that radiation itself caused any additional toxicity.”
One of the patients did have a perforated colon, she acknowledged, and for this reason future research may look at the combination of radiation therapy with the PD-1 (programmed T-cell death) inhibitors.
“I think right now everyone is so excited about the PD-1 inhibitors, and think that they’re safer,” Dr. Hiniker suggested. “It’s hard to give someone ipilimumab alone as the first line at this point; although many patients may not experience toxicity, [in] the ones that do, it can be severe.”
Dr. Hiniker also reported that IRC (independent review committee) analysis results suggested that several proinflammatory cytokines were elevated in some patients who responded to treatment. Higher levels of interleukin 2 at baseline, and at two time points during the study, for example, were associated with better response rates than lower levels. The findings also suggested that there might be a relationship between an increased number of elevated CD8-positive T cells and response.
“I think that we will find biomarkers, but we are not there yet, and it would need to be confirmed in a longer, larger study,” Dr. Hiniker said.
From a practical perspective, if a patient is already being treated with ipilimumab, and a physician is considering radiotherapy, then these results suggest that there will be no additional toxicity, she suggested.
“This combination approach does appear to be promising,” she said. “So for a subset of carefully selected patients – and the problem is we don’t totally know how to select them yet – this can be extremely promising.”
Dr. Hiniker had no disclosures to report.
AT THE ASTRO ANNUAL MEETING
Key clinical point: Some patients with metastatic melanoma may benefit from combined use of novel immunotherapies and radiation treatment.
Major finding: No additional toxicity was seen and 50% of patients had a complete or partial response to the treatment regimen of combined radioimmunotherapy.
Data source: Prospective phase II clinical trial of 22 patients with melanoma treated with ipilimumab and palliative radiation therapy.
Disclosures: Dr. Hiniker had no disclosures to report.