User login
Waiting for Therapy? There’s an App for That
TOPLINE:
Smartphone apps, including those using cognitive-behavioral therapy (CBT) and mindfulness techniques, showed comparable efficacy in reducing depression, anxiety, and suicidality in patients with psychiatric conditions waiting for appointments with psychiatrists or therapists.
METHODOLOGY:
- Participants were adults aged 18 years or older seeking outpatient psychiatric services from several mental and behavioral health clinics within the University of Michigan Health System.
- Eligible participants were those with either a scheduled future mental health appointment or an initial appointment completed within the past 60 days and daily access to a smartphone.
- After completing a baseline survey that gathered data on participants’ depression, anxiety, and suicidality scores, 2080 participants were randomly assigned to one of five groups:
- Enhanced personalized feedback (EPF) only (n = 690)
- SilverCloud only (SilverCloud, a mobile application designed to deliver CBT strategies; n = 345)
- SilverCloud plus EPF (n = 346)
- Headspace only (Headspace, a mobile application designed to train users in mindfulness practices; n = 349)
- Headspace plus EPF (n = 349)
TAKEAWAY:
- The mean baseline Patient Health Questionnaire-9 depression score was 12.7 (6.4% patients). Overall, depression scores significantly decreased by 2.5 points from baseline to the 6-week follow-up for all five arms, with marginal mean differences in mean change ranging from −2.1 to −2.9 (P < .001).
- The magnitude of change was not significantly different across the five arms on most measures (P = .31). Additionally, the groups did not differ in decrease of anxiety or substance use symptoms.
- The Headspace arms reported significantly greater improvements on a suicidality measure subscale than the SilverCloud arms (mean difference in mean change, 0.63; P = .004).
IN PRACTICE:
“Having this type of option, especially for people who are motivated enough to seek an appointment and wait for it, could be very valuable when providers have long wait lists,” lead author Adam Horwitz, PhD, University of Michigan, Ann Arbor, said in a press release.
“These individuals want to be doing something about their mental health but don’t yet have access, so this suggests that providing them with some sort of digital option when their motivation is already high, and they are ready to do something, could begin to make a difference.”
SOURCE:
Dr. Horwitz led the study, which was published online in JAMA Network Open.
LIMITATIONS:
There may have been aspects of formal or in-person care that contributed to the improvement in symptoms across groups and diluted the ability to identify differences between applications in effects on symptom reduction.
DISCLOSURES:
This study was funded by a grant from Precision Health, the Eisenberg Family Depression Center, and the National Institute of Mental Health. Disclosures are noted in the original article.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.A version of this article first appeared on Medscape.com.
TOPLINE:
Smartphone apps, including those using cognitive-behavioral therapy (CBT) and mindfulness techniques, showed comparable efficacy in reducing depression, anxiety, and suicidality in patients with psychiatric conditions waiting for appointments with psychiatrists or therapists.
METHODOLOGY:
- Participants were adults aged 18 years or older seeking outpatient psychiatric services from several mental and behavioral health clinics within the University of Michigan Health System.
- Eligible participants were those with either a scheduled future mental health appointment or an initial appointment completed within the past 60 days and daily access to a smartphone.
- After completing a baseline survey that gathered data on participants’ depression, anxiety, and suicidality scores, 2080 participants were randomly assigned to one of five groups:
- Enhanced personalized feedback (EPF) only (n = 690)
- SilverCloud only (SilverCloud, a mobile application designed to deliver CBT strategies; n = 345)
- SilverCloud plus EPF (n = 346)
- Headspace only (Headspace, a mobile application designed to train users in mindfulness practices; n = 349)
- Headspace plus EPF (n = 349)
TAKEAWAY:
- The mean baseline Patient Health Questionnaire-9 depression score was 12.7 (6.4% patients). Overall, depression scores significantly decreased by 2.5 points from baseline to the 6-week follow-up for all five arms, with marginal mean differences in mean change ranging from −2.1 to −2.9 (P < .001).
- The magnitude of change was not significantly different across the five arms on most measures (P = .31). Additionally, the groups did not differ in decrease of anxiety or substance use symptoms.
- The Headspace arms reported significantly greater improvements on a suicidality measure subscale than the SilverCloud arms (mean difference in mean change, 0.63; P = .004).
IN PRACTICE:
“Having this type of option, especially for people who are motivated enough to seek an appointment and wait for it, could be very valuable when providers have long wait lists,” lead author Adam Horwitz, PhD, University of Michigan, Ann Arbor, said in a press release.
“These individuals want to be doing something about their mental health but don’t yet have access, so this suggests that providing them with some sort of digital option when their motivation is already high, and they are ready to do something, could begin to make a difference.”
SOURCE:
Dr. Horwitz led the study, which was published online in JAMA Network Open.
LIMITATIONS:
There may have been aspects of formal or in-person care that contributed to the improvement in symptoms across groups and diluted the ability to identify differences between applications in effects on symptom reduction.
DISCLOSURES:
This study was funded by a grant from Precision Health, the Eisenberg Family Depression Center, and the National Institute of Mental Health. Disclosures are noted in the original article.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.A version of this article first appeared on Medscape.com.
TOPLINE:
Smartphone apps, including those using cognitive-behavioral therapy (CBT) and mindfulness techniques, showed comparable efficacy in reducing depression, anxiety, and suicidality in patients with psychiatric conditions waiting for appointments with psychiatrists or therapists.
METHODOLOGY:
- Participants were adults aged 18 years or older seeking outpatient psychiatric services from several mental and behavioral health clinics within the University of Michigan Health System.
- Eligible participants were those with either a scheduled future mental health appointment or an initial appointment completed within the past 60 days and daily access to a smartphone.
- After completing a baseline survey that gathered data on participants’ depression, anxiety, and suicidality scores, 2080 participants were randomly assigned to one of five groups:
- Enhanced personalized feedback (EPF) only (n = 690)
- SilverCloud only (SilverCloud, a mobile application designed to deliver CBT strategies; n = 345)
- SilverCloud plus EPF (n = 346)
- Headspace only (Headspace, a mobile application designed to train users in mindfulness practices; n = 349)
- Headspace plus EPF (n = 349)
TAKEAWAY:
- The mean baseline Patient Health Questionnaire-9 depression score was 12.7 (6.4% patients). Overall, depression scores significantly decreased by 2.5 points from baseline to the 6-week follow-up for all five arms, with marginal mean differences in mean change ranging from −2.1 to −2.9 (P < .001).
- The magnitude of change was not significantly different across the five arms on most measures (P = .31). Additionally, the groups did not differ in decrease of anxiety or substance use symptoms.
- The Headspace arms reported significantly greater improvements on a suicidality measure subscale than the SilverCloud arms (mean difference in mean change, 0.63; P = .004).
IN PRACTICE:
“Having this type of option, especially for people who are motivated enough to seek an appointment and wait for it, could be very valuable when providers have long wait lists,” lead author Adam Horwitz, PhD, University of Michigan, Ann Arbor, said in a press release.
“These individuals want to be doing something about their mental health but don’t yet have access, so this suggests that providing them with some sort of digital option when their motivation is already high, and they are ready to do something, could begin to make a difference.”
SOURCE:
Dr. Horwitz led the study, which was published online in JAMA Network Open.
LIMITATIONS:
There may have been aspects of formal or in-person care that contributed to the improvement in symptoms across groups and diluted the ability to identify differences between applications in effects on symptom reduction.
DISCLOSURES:
This study was funded by a grant from Precision Health, the Eisenberg Family Depression Center, and the National Institute of Mental Health. Disclosures are noted in the original article.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.A version of this article first appeared on Medscape.com.
Introducing: A New Way to Get Teens Mental Health Care
Lauren Opladen remembers the agonizing wait all too well.
At age 17, struggling with paralyzing depression after losing her brother to suicide and her father to amyotrophic lateral sclerosis, her teacher suggested she seek help.
So, she did. But she had to spend 3 days inside an emergency department at the University of Rochester Medical Center in Rochester, New York, where the Comprehensive Psychiatric Emergency Program (CPEP) provides immediate care for youth and adults experiencing psychiatric emergencies.
“We were sleeping on a couch just waiting for all these services, when that’s precious time wasted,” Ms. Opladen said.
Ms. Opladen made it through that dark period, and 5 years later, she is a registered nurse at the same hospital. Every day she walks past a new facility she wishes had existed during her troubled teenage years: An urgent care center for children and adolescents experiencing mental health crises.
Brighter Days Pediatric Mental Health Urgent Care Center, Rochester, New York, opened in July as a walk-in clinic offering rapid assessment, crisis intervention, and short-term stabilization, provides referrals to counseling or psychiatric care. Children and adolescents at immediate risk of harming themselves or others, or who need inpatient care, are sent to CPEP or another emergency department in the area.
Similar walk-in facilities linking youth to longer-term services are popping up in nearly a dozen states, including New York, Ohio, Massachusetts, and Wisconsin. The emerging model of care may offer a crucial bridge between traditional outpatient services and emergency room (ER) visits for some young people experiencing mental health crises.
“We’ve seen a significant increase in the number of children and adolescents presenting to emergency departments with mental health concerns,” said Michael A. Scharf, MD, chief of the Division of Child and Adolescent Psychiatry at the University of Rochester Medical Center, who oversees operations at Brighter Days. “These urgent care centers provide a more appropriate setting for many of these cases, offering specialized care without the often overwhelming environment of an ER.”
The urgency of addressing youth behavioral health has become increasingly apparent. The most recent data from the US Centers for Disease Control and Prevention showed that over a 6-month period in 2020, during the early months of the COVID-19 pandemic, visits to the emergency department for mental health problems spiked 24% among children aged 5-11 years and 31% among 12-17-year-olds compared with the same period in 2019. Between March 2021 and February 2022, such emergency visits rose by 22% for teen girls, while falling by 15% for boys ages 5-12 years and 9% for older boys. Most visits occur during the school year.
But staffing shortages and limited physical space are taxing the capacity of the healthcare system to screen, diagnose, and manage these patients, according to a 2023 report published in Pediatrics.
Urgent Care: A Misnomer?
Some in the mental health community said the label “urgent” in these centers’ titles is misleading. Brighter Days and similar facilities do not conduct involuntary holds, administer medication, or handle serious cases like psychotic episodes.
David Mathison, MD, senior vice president of clinic operations at PM Pediatrics, a chain of pediatric urgent care clinics in Maryland, said patients and their families may mistakenly believe the centers will address mental health problems quickly.
“It’s really not urgent behavioral health. It’s really just another access point to get behavioral health,” Dr. Mathison said. “Crises in pediatrics are so much more complex” than physical injuries or acute infections, which are the bread and butter of urgent care centers.
“An urgent care center almost implies you’re going to come in for a solution to a simple problem, and it’s going to be done relatively quickly on demand, and it’s just not what the behavioral health centers do,” he said.
Dr. Mathison, who also serves on the executive committee for the section on urgent care at the American Academy of Pediatrics, likened the centers to in-person versions of crisis center hotlines, which offer virtual counseling and talk therapy and may refer individuals to specialists who can provide clinical care over the long term.
Instead, Brighter Days and other centers provide crisis de-escalation for individuals experiencing an exacerbation of a diagnosed mental illness, such a manic episode from bipolar disorder.
“Most places aren’t just going to change their therapy without either contacting their psychiatrist or having psychiatrists on staff,” Dr. Mathison said.
Other challenges at Brighter Days and similar centers include staffing with appropriately trained mental health professionals, given the nationwide shortage of child and adolescent psychiatrists, Dr. Scharf said.
The number of child and adolescent psychiatrists per 100,000 children varies significantly across states. Nationally, the average stands at 14 psychiatrists per 100,000 children, but ranges from as low as 4 to 65, according to the American Academy of Child & Adolescent Psychiatry.
For now, Dr. Scharf said, patients who visit Brighter Days are billed as if they are having a routine pediatric office visit as opposed to a pricier trip to the emergency department. And the center accepts all individuals, regardless of their insurance status.
Ms. Opladen said the urgent care center represents a significant improvement over her experience at the emergency department’s psychiatric triage.
“I saw how awful it was and just the environment,” she said. “The first thing I thought was, what do I need to do to get out of here?”
She said the pediatric mental health urgent care centers are “the complete opposite.” Like Brighter Days, these centers are designed to look more like a pediatrician’s office, with bright welcoming colors and games and toys.
“It’s separated from everything else. There’s a welcome, relaxed space,” she said. “The welcoming feel is just a whole different environment, and that’s really how it should be.”
A version of this article first appeared on Medscape.com.
Lauren Opladen remembers the agonizing wait all too well.
At age 17, struggling with paralyzing depression after losing her brother to suicide and her father to amyotrophic lateral sclerosis, her teacher suggested she seek help.
So, she did. But she had to spend 3 days inside an emergency department at the University of Rochester Medical Center in Rochester, New York, where the Comprehensive Psychiatric Emergency Program (CPEP) provides immediate care for youth and adults experiencing psychiatric emergencies.
“We were sleeping on a couch just waiting for all these services, when that’s precious time wasted,” Ms. Opladen said.
Ms. Opladen made it through that dark period, and 5 years later, she is a registered nurse at the same hospital. Every day she walks past a new facility she wishes had existed during her troubled teenage years: An urgent care center for children and adolescents experiencing mental health crises.
Brighter Days Pediatric Mental Health Urgent Care Center, Rochester, New York, opened in July as a walk-in clinic offering rapid assessment, crisis intervention, and short-term stabilization, provides referrals to counseling or psychiatric care. Children and adolescents at immediate risk of harming themselves or others, or who need inpatient care, are sent to CPEP or another emergency department in the area.
Similar walk-in facilities linking youth to longer-term services are popping up in nearly a dozen states, including New York, Ohio, Massachusetts, and Wisconsin. The emerging model of care may offer a crucial bridge between traditional outpatient services and emergency room (ER) visits for some young people experiencing mental health crises.
“We’ve seen a significant increase in the number of children and adolescents presenting to emergency departments with mental health concerns,” said Michael A. Scharf, MD, chief of the Division of Child and Adolescent Psychiatry at the University of Rochester Medical Center, who oversees operations at Brighter Days. “These urgent care centers provide a more appropriate setting for many of these cases, offering specialized care without the often overwhelming environment of an ER.”
The urgency of addressing youth behavioral health has become increasingly apparent. The most recent data from the US Centers for Disease Control and Prevention showed that over a 6-month period in 2020, during the early months of the COVID-19 pandemic, visits to the emergency department for mental health problems spiked 24% among children aged 5-11 years and 31% among 12-17-year-olds compared with the same period in 2019. Between March 2021 and February 2022, such emergency visits rose by 22% for teen girls, while falling by 15% for boys ages 5-12 years and 9% for older boys. Most visits occur during the school year.
But staffing shortages and limited physical space are taxing the capacity of the healthcare system to screen, diagnose, and manage these patients, according to a 2023 report published in Pediatrics.
Urgent Care: A Misnomer?
Some in the mental health community said the label “urgent” in these centers’ titles is misleading. Brighter Days and similar facilities do not conduct involuntary holds, administer medication, or handle serious cases like psychotic episodes.
David Mathison, MD, senior vice president of clinic operations at PM Pediatrics, a chain of pediatric urgent care clinics in Maryland, said patients and their families may mistakenly believe the centers will address mental health problems quickly.
“It’s really not urgent behavioral health. It’s really just another access point to get behavioral health,” Dr. Mathison said. “Crises in pediatrics are so much more complex” than physical injuries or acute infections, which are the bread and butter of urgent care centers.
“An urgent care center almost implies you’re going to come in for a solution to a simple problem, and it’s going to be done relatively quickly on demand, and it’s just not what the behavioral health centers do,” he said.
Dr. Mathison, who also serves on the executive committee for the section on urgent care at the American Academy of Pediatrics, likened the centers to in-person versions of crisis center hotlines, which offer virtual counseling and talk therapy and may refer individuals to specialists who can provide clinical care over the long term.
Instead, Brighter Days and other centers provide crisis de-escalation for individuals experiencing an exacerbation of a diagnosed mental illness, such a manic episode from bipolar disorder.
“Most places aren’t just going to change their therapy without either contacting their psychiatrist or having psychiatrists on staff,” Dr. Mathison said.
Other challenges at Brighter Days and similar centers include staffing with appropriately trained mental health professionals, given the nationwide shortage of child and adolescent psychiatrists, Dr. Scharf said.
The number of child and adolescent psychiatrists per 100,000 children varies significantly across states. Nationally, the average stands at 14 psychiatrists per 100,000 children, but ranges from as low as 4 to 65, according to the American Academy of Child & Adolescent Psychiatry.
For now, Dr. Scharf said, patients who visit Brighter Days are billed as if they are having a routine pediatric office visit as opposed to a pricier trip to the emergency department. And the center accepts all individuals, regardless of their insurance status.
Ms. Opladen said the urgent care center represents a significant improvement over her experience at the emergency department’s psychiatric triage.
“I saw how awful it was and just the environment,” she said. “The first thing I thought was, what do I need to do to get out of here?”
She said the pediatric mental health urgent care centers are “the complete opposite.” Like Brighter Days, these centers are designed to look more like a pediatrician’s office, with bright welcoming colors and games and toys.
“It’s separated from everything else. There’s a welcome, relaxed space,” she said. “The welcoming feel is just a whole different environment, and that’s really how it should be.”
A version of this article first appeared on Medscape.com.
Lauren Opladen remembers the agonizing wait all too well.
At age 17, struggling with paralyzing depression after losing her brother to suicide and her father to amyotrophic lateral sclerosis, her teacher suggested she seek help.
So, she did. But she had to spend 3 days inside an emergency department at the University of Rochester Medical Center in Rochester, New York, where the Comprehensive Psychiatric Emergency Program (CPEP) provides immediate care for youth and adults experiencing psychiatric emergencies.
“We were sleeping on a couch just waiting for all these services, when that’s precious time wasted,” Ms. Opladen said.
Ms. Opladen made it through that dark period, and 5 years later, she is a registered nurse at the same hospital. Every day she walks past a new facility she wishes had existed during her troubled teenage years: An urgent care center for children and adolescents experiencing mental health crises.
Brighter Days Pediatric Mental Health Urgent Care Center, Rochester, New York, opened in July as a walk-in clinic offering rapid assessment, crisis intervention, and short-term stabilization, provides referrals to counseling or psychiatric care. Children and adolescents at immediate risk of harming themselves or others, or who need inpatient care, are sent to CPEP or another emergency department in the area.
Similar walk-in facilities linking youth to longer-term services are popping up in nearly a dozen states, including New York, Ohio, Massachusetts, and Wisconsin. The emerging model of care may offer a crucial bridge between traditional outpatient services and emergency room (ER) visits for some young people experiencing mental health crises.
“We’ve seen a significant increase in the number of children and adolescents presenting to emergency departments with mental health concerns,” said Michael A. Scharf, MD, chief of the Division of Child and Adolescent Psychiatry at the University of Rochester Medical Center, who oversees operations at Brighter Days. “These urgent care centers provide a more appropriate setting for many of these cases, offering specialized care without the often overwhelming environment of an ER.”
The urgency of addressing youth behavioral health has become increasingly apparent. The most recent data from the US Centers for Disease Control and Prevention showed that over a 6-month period in 2020, during the early months of the COVID-19 pandemic, visits to the emergency department for mental health problems spiked 24% among children aged 5-11 years and 31% among 12-17-year-olds compared with the same period in 2019. Between March 2021 and February 2022, such emergency visits rose by 22% for teen girls, while falling by 15% for boys ages 5-12 years and 9% for older boys. Most visits occur during the school year.
But staffing shortages and limited physical space are taxing the capacity of the healthcare system to screen, diagnose, and manage these patients, according to a 2023 report published in Pediatrics.
Urgent Care: A Misnomer?
Some in the mental health community said the label “urgent” in these centers’ titles is misleading. Brighter Days and similar facilities do not conduct involuntary holds, administer medication, or handle serious cases like psychotic episodes.
David Mathison, MD, senior vice president of clinic operations at PM Pediatrics, a chain of pediatric urgent care clinics in Maryland, said patients and their families may mistakenly believe the centers will address mental health problems quickly.
“It’s really not urgent behavioral health. It’s really just another access point to get behavioral health,” Dr. Mathison said. “Crises in pediatrics are so much more complex” than physical injuries or acute infections, which are the bread and butter of urgent care centers.
“An urgent care center almost implies you’re going to come in for a solution to a simple problem, and it’s going to be done relatively quickly on demand, and it’s just not what the behavioral health centers do,” he said.
Dr. Mathison, who also serves on the executive committee for the section on urgent care at the American Academy of Pediatrics, likened the centers to in-person versions of crisis center hotlines, which offer virtual counseling and talk therapy and may refer individuals to specialists who can provide clinical care over the long term.
Instead, Brighter Days and other centers provide crisis de-escalation for individuals experiencing an exacerbation of a diagnosed mental illness, such a manic episode from bipolar disorder.
“Most places aren’t just going to change their therapy without either contacting their psychiatrist or having psychiatrists on staff,” Dr. Mathison said.
Other challenges at Brighter Days and similar centers include staffing with appropriately trained mental health professionals, given the nationwide shortage of child and adolescent psychiatrists, Dr. Scharf said.
The number of child and adolescent psychiatrists per 100,000 children varies significantly across states. Nationally, the average stands at 14 psychiatrists per 100,000 children, but ranges from as low as 4 to 65, according to the American Academy of Child & Adolescent Psychiatry.
For now, Dr. Scharf said, patients who visit Brighter Days are billed as if they are having a routine pediatric office visit as opposed to a pricier trip to the emergency department. And the center accepts all individuals, regardless of their insurance status.
Ms. Opladen said the urgent care center represents a significant improvement over her experience at the emergency department’s psychiatric triage.
“I saw how awful it was and just the environment,” she said. “The first thing I thought was, what do I need to do to get out of here?”
She said the pediatric mental health urgent care centers are “the complete opposite.” Like Brighter Days, these centers are designed to look more like a pediatrician’s office, with bright welcoming colors and games and toys.
“It’s separated from everything else. There’s a welcome, relaxed space,” she said. “The welcoming feel is just a whole different environment, and that’s really how it should be.”
A version of this article first appeared on Medscape.com.
Study Links Newer Shingles Vaccine to Delayed Dementia Diagnosis
The study builds on previous observations of a reduction in dementia risk with the older live shingles vaccine and reports a delay in dementia diagnosis of 164 days with the newer recombinant version, compared with the live vaccine.
“Given the prevalence of dementia, a delay of 164 days in diagnosis would not be a trivial effect at the public health level. It’s a big enough effect that if there is a causality it feels meaningful,” said senior author Paul Harrison, DM, FRCPsych, professor of psychiatry at the University of Oxford, Oxford, England.
But Dr. Harrison stressed that the study had not proven that the shingles vaccine reduced dementia risk.
“The design of the study allows us to do away with many of the confounding effects we usually see in observational studies, but this is still an observational study, and as such it cannot prove a definite causal effect,” he said.
The study was published online on July 25 in Nature Medicine.
‘Natural Experiment’
Given the risk for deleterious consequences of shingles, vaccination is now recommended for older adults in many countries. The previously used live shingles vaccine (Zostavax) is being replaced in most countries with the new recombinant shingles vaccine (Shingrix), which is more effective at preventing shingles infection.
The current study made use of a “natural experiment” in the United States, which switched over from use of the live vaccine to the recombinant vaccine in October 2017.
Researchers used electronic heath records to compare the incidence of a dementia diagnosis in individuals who received the live shingles vaccine prior to October 2017 with those who received the recombinant version after the United States made the switch.
They also used propensity score matching to further control for confounding factors, comparing 103,837 individuals who received a first dose of the live shingles vaccine between October 2014 and September 2017 with the same number of matched people who received the recombinant vaccine between November 2017 and October 2020.
Results showed that within the 6 years after vaccination, the recombinant vaccine was associated with a delay in the diagnosis of dementia, compared with the live vaccine. Specifically, receiving the recombinant vaccine was associated with a 17% increase in diagnosis-free time, translating to 164 additional days lived without a diagnosis of dementia in those subsequently affected.
As an additional control, the researchers also found significantly lower risks for dementia in individuals receiving the new recombinant shingles vaccine vs two other vaccines commonly used in older people: influenza and tetanus/diphtheria/pertussis vaccines, with increases in diagnosis-free time of 14%-27%.
Reduced Risk or Delayed Diagnosis?
Speaking at a Science Media Centre press conference on the study, lead author Maxime Taquet, PhD, FRCPsych, clinical lecturer in psychiatry at the University of Oxford, noted that the total number of dementia cases were similar in the two shingles vaccine groups by the end of the 6-year follow-up period but there was a difference in the time at which they received a diagnosis of dementia.
“The study suggests that rather than actually reducing dementia risk, the recombinant vaccine delays the onset of dementia compared to the live vaccine in patients who go on to develop the condition,” he explained.
But when comparing the recombinant vaccine with the influenza and tetanus/diphtheria/pertussis vaccines there was a clear reduction in dementia risk itself, Dr. Taquet reported.
“It might well be that the live vaccine has a potential effect on the risk of dementia itself and therefore the recombinant vaccine only shows a delay in dementia compared to the live vaccine, but both of them might decrease the overall risk of dementia,” he suggested.
But the researchers cautioned that this study could not prove causality.
“While the two groups were very carefully matched in terms of factors that might influence the development of dementia, we still have to be cautious before assuming that the vaccine is indeed causally reducing the risk of onset of dementia,” Dr. Harrison warned.
The researchers say the results would need to be confirmed in a randomized trial, which may have to be conducted in a slightly younger age group, as currently shingles vaccine is recommended for all older individuals in the United Kingdom.
Vaccine recommendations vary from country to country, Dr. Harrison added. In the United States, the Centers for Disease Control and Prevention recommends the recombinant shingles vaccine for all adults aged 50 years or older.
In the meantime, it would be interesting to see whether further observational studies in other countries find similar results as this US study, Dr. Harrison said.
Mechanism Uncertain
Speculating on a possible mechanism behind the findings, Dr. Harrison suggested two plausible explanations.
“First, it is thought that the herpes virus could be one of many factors that could promote dementia, so a vaccine that stops reactivation of this virus might therefore be delaying that process,” he noted.
The other possibility is that adjuvants included in the recombinant vaccine to stimulate the immune system might have played a role.
“We don’t have any data on the mechanism, and thus study did not address that, so further studies are needed to look into this,” Dr. Harrison said.
Stronger Effect in Women
Another intriguing finding is that the association with the recombinant vaccine and delayed dementia diagnosis seemed to be stronger in women vs men.
In the original study of the live shingles vaccine, a protective effect against dementia was shown only in women.
In the current study, the delay in dementia diagnosis was seen in both sexes but was stronger in women, showing a 22% increased time without dementia in women versus a 13% increased time in men with the recombinant versus the live vaccine.
As expected, the recombinant vaccine was associated with a lower risk for shingles disease vs the live vaccine (2.5% versus 3.5%), but women did not have a better response than men did in this respect.
“The better protection against shingles with the recombinant vaccine was similar in men and women, an observation that might be one reason to question the possible mechanism behind the dementia effect being better suppression of the herpes zoster virus by the recombinant vaccine,” Dr. Harrison commented.
Though these findings are not likely to lead to any immediate changes in policy regarding the shingles vaccine, Dr. Harrison said it would be interesting to see whether uptake of the vaccine increased after this study.
He estimated that, currently in the United Kingdom, about 60% of older adults choose to have the shingles vaccine. A 2020 study in the United States found that only about one-third of US adults over 60 had received the vaccine.
“It will be interesting to see if that figure increases after these data are publicized, but I am not recommending that people have the vaccine specifically to lower their risk of dementia because of the caveats about the study that we have discussed,” he commented.
Outside Experts Positive
Outside experts, providing comment to the Science Media Centre, welcomed the new research.
“ The study is very well-conducted and adds to previous data indicating that vaccination against shingles is associated with lower dementia risk. More research is needed in future to determine why this vaccine is associated with lower dementia risk,” said Tara Spires-Jones, FMedSci, president of the British Neuroscience Association.
The high number of patients in the study and the adjustments for potential confounders are also strong points, noted Andrew Doig, PhD, professor of biochemistry, University of Manchester, Manchester, England.
“This is a significant result, comparable in effectiveness to the recent antibody drugs for Alzheimer’s disease,” Dr. Doig said. “Administering the recombinant shingles vaccine could well be a simple and cheap way to lower the risk of Alzheimer’s disease.”
Dr. Doig noted that a link between herpes zoster infection and the onset of dementia has been suspected for some time, and a trial of the antiviral drug valacyclovir against Alzheimer’s disease is currently underway.
In regard to the shingles vaccine, he said a placebo-controlled trial would be needed to prove causality.
“We also need to see how many years the effect might last and whether we should vaccinate people at a younger age. We know that the path to Alzheimer’s can start decades before any symptoms are apparent, so the vaccine might be even more effective if given to people in their 40s or 50s,” he said.
Dr. Harrison and Dr. Taquet reported no disclosures. Dr. Doig is a founder, director, and consultant for PharmaKure, which works on Alzheimer’s drugs and diagnostics. Other commentators declared no disclosures.
A version of this article first appeared on Medscape.com.
The study builds on previous observations of a reduction in dementia risk with the older live shingles vaccine and reports a delay in dementia diagnosis of 164 days with the newer recombinant version, compared with the live vaccine.
“Given the prevalence of dementia, a delay of 164 days in diagnosis would not be a trivial effect at the public health level. It’s a big enough effect that if there is a causality it feels meaningful,” said senior author Paul Harrison, DM, FRCPsych, professor of psychiatry at the University of Oxford, Oxford, England.
But Dr. Harrison stressed that the study had not proven that the shingles vaccine reduced dementia risk.
“The design of the study allows us to do away with many of the confounding effects we usually see in observational studies, but this is still an observational study, and as such it cannot prove a definite causal effect,” he said.
The study was published online on July 25 in Nature Medicine.
‘Natural Experiment’
Given the risk for deleterious consequences of shingles, vaccination is now recommended for older adults in many countries. The previously used live shingles vaccine (Zostavax) is being replaced in most countries with the new recombinant shingles vaccine (Shingrix), which is more effective at preventing shingles infection.
The current study made use of a “natural experiment” in the United States, which switched over from use of the live vaccine to the recombinant vaccine in October 2017.
Researchers used electronic heath records to compare the incidence of a dementia diagnosis in individuals who received the live shingles vaccine prior to October 2017 with those who received the recombinant version after the United States made the switch.
They also used propensity score matching to further control for confounding factors, comparing 103,837 individuals who received a first dose of the live shingles vaccine between October 2014 and September 2017 with the same number of matched people who received the recombinant vaccine between November 2017 and October 2020.
Results showed that within the 6 years after vaccination, the recombinant vaccine was associated with a delay in the diagnosis of dementia, compared with the live vaccine. Specifically, receiving the recombinant vaccine was associated with a 17% increase in diagnosis-free time, translating to 164 additional days lived without a diagnosis of dementia in those subsequently affected.
As an additional control, the researchers also found significantly lower risks for dementia in individuals receiving the new recombinant shingles vaccine vs two other vaccines commonly used in older people: influenza and tetanus/diphtheria/pertussis vaccines, with increases in diagnosis-free time of 14%-27%.
Reduced Risk or Delayed Diagnosis?
Speaking at a Science Media Centre press conference on the study, lead author Maxime Taquet, PhD, FRCPsych, clinical lecturer in psychiatry at the University of Oxford, noted that the total number of dementia cases were similar in the two shingles vaccine groups by the end of the 6-year follow-up period but there was a difference in the time at which they received a diagnosis of dementia.
“The study suggests that rather than actually reducing dementia risk, the recombinant vaccine delays the onset of dementia compared to the live vaccine in patients who go on to develop the condition,” he explained.
But when comparing the recombinant vaccine with the influenza and tetanus/diphtheria/pertussis vaccines there was a clear reduction in dementia risk itself, Dr. Taquet reported.
“It might well be that the live vaccine has a potential effect on the risk of dementia itself and therefore the recombinant vaccine only shows a delay in dementia compared to the live vaccine, but both of them might decrease the overall risk of dementia,” he suggested.
But the researchers cautioned that this study could not prove causality.
“While the two groups were very carefully matched in terms of factors that might influence the development of dementia, we still have to be cautious before assuming that the vaccine is indeed causally reducing the risk of onset of dementia,” Dr. Harrison warned.
The researchers say the results would need to be confirmed in a randomized trial, which may have to be conducted in a slightly younger age group, as currently shingles vaccine is recommended for all older individuals in the United Kingdom.
Vaccine recommendations vary from country to country, Dr. Harrison added. In the United States, the Centers for Disease Control and Prevention recommends the recombinant shingles vaccine for all adults aged 50 years or older.
In the meantime, it would be interesting to see whether further observational studies in other countries find similar results as this US study, Dr. Harrison said.
Mechanism Uncertain
Speculating on a possible mechanism behind the findings, Dr. Harrison suggested two plausible explanations.
“First, it is thought that the herpes virus could be one of many factors that could promote dementia, so a vaccine that stops reactivation of this virus might therefore be delaying that process,” he noted.
The other possibility is that adjuvants included in the recombinant vaccine to stimulate the immune system might have played a role.
“We don’t have any data on the mechanism, and thus study did not address that, so further studies are needed to look into this,” Dr. Harrison said.
Stronger Effect in Women
Another intriguing finding is that the association with the recombinant vaccine and delayed dementia diagnosis seemed to be stronger in women vs men.
In the original study of the live shingles vaccine, a protective effect against dementia was shown only in women.
In the current study, the delay in dementia diagnosis was seen in both sexes but was stronger in women, showing a 22% increased time without dementia in women versus a 13% increased time in men with the recombinant versus the live vaccine.
As expected, the recombinant vaccine was associated with a lower risk for shingles disease vs the live vaccine (2.5% versus 3.5%), but women did not have a better response than men did in this respect.
“The better protection against shingles with the recombinant vaccine was similar in men and women, an observation that might be one reason to question the possible mechanism behind the dementia effect being better suppression of the herpes zoster virus by the recombinant vaccine,” Dr. Harrison commented.
Though these findings are not likely to lead to any immediate changes in policy regarding the shingles vaccine, Dr. Harrison said it would be interesting to see whether uptake of the vaccine increased after this study.
He estimated that, currently in the United Kingdom, about 60% of older adults choose to have the shingles vaccine. A 2020 study in the United States found that only about one-third of US adults over 60 had received the vaccine.
“It will be interesting to see if that figure increases after these data are publicized, but I am not recommending that people have the vaccine specifically to lower their risk of dementia because of the caveats about the study that we have discussed,” he commented.
Outside Experts Positive
Outside experts, providing comment to the Science Media Centre, welcomed the new research.
“ The study is very well-conducted and adds to previous data indicating that vaccination against shingles is associated with lower dementia risk. More research is needed in future to determine why this vaccine is associated with lower dementia risk,” said Tara Spires-Jones, FMedSci, president of the British Neuroscience Association.
The high number of patients in the study and the adjustments for potential confounders are also strong points, noted Andrew Doig, PhD, professor of biochemistry, University of Manchester, Manchester, England.
“This is a significant result, comparable in effectiveness to the recent antibody drugs for Alzheimer’s disease,” Dr. Doig said. “Administering the recombinant shingles vaccine could well be a simple and cheap way to lower the risk of Alzheimer’s disease.”
Dr. Doig noted that a link between herpes zoster infection and the onset of dementia has been suspected for some time, and a trial of the antiviral drug valacyclovir against Alzheimer’s disease is currently underway.
In regard to the shingles vaccine, he said a placebo-controlled trial would be needed to prove causality.
“We also need to see how many years the effect might last and whether we should vaccinate people at a younger age. We know that the path to Alzheimer’s can start decades before any symptoms are apparent, so the vaccine might be even more effective if given to people in their 40s or 50s,” he said.
Dr. Harrison and Dr. Taquet reported no disclosures. Dr. Doig is a founder, director, and consultant for PharmaKure, which works on Alzheimer’s drugs and diagnostics. Other commentators declared no disclosures.
A version of this article first appeared on Medscape.com.
The study builds on previous observations of a reduction in dementia risk with the older live shingles vaccine and reports a delay in dementia diagnosis of 164 days with the newer recombinant version, compared with the live vaccine.
“Given the prevalence of dementia, a delay of 164 days in diagnosis would not be a trivial effect at the public health level. It’s a big enough effect that if there is a causality it feels meaningful,” said senior author Paul Harrison, DM, FRCPsych, professor of psychiatry at the University of Oxford, Oxford, England.
But Dr. Harrison stressed that the study had not proven that the shingles vaccine reduced dementia risk.
“The design of the study allows us to do away with many of the confounding effects we usually see in observational studies, but this is still an observational study, and as such it cannot prove a definite causal effect,” he said.
The study was published online on July 25 in Nature Medicine.
‘Natural Experiment’
Given the risk for deleterious consequences of shingles, vaccination is now recommended for older adults in many countries. The previously used live shingles vaccine (Zostavax) is being replaced in most countries with the new recombinant shingles vaccine (Shingrix), which is more effective at preventing shingles infection.
The current study made use of a “natural experiment” in the United States, which switched over from use of the live vaccine to the recombinant vaccine in October 2017.
Researchers used electronic heath records to compare the incidence of a dementia diagnosis in individuals who received the live shingles vaccine prior to October 2017 with those who received the recombinant version after the United States made the switch.
They also used propensity score matching to further control for confounding factors, comparing 103,837 individuals who received a first dose of the live shingles vaccine between October 2014 and September 2017 with the same number of matched people who received the recombinant vaccine between November 2017 and October 2020.
Results showed that within the 6 years after vaccination, the recombinant vaccine was associated with a delay in the diagnosis of dementia, compared with the live vaccine. Specifically, receiving the recombinant vaccine was associated with a 17% increase in diagnosis-free time, translating to 164 additional days lived without a diagnosis of dementia in those subsequently affected.
As an additional control, the researchers also found significantly lower risks for dementia in individuals receiving the new recombinant shingles vaccine vs two other vaccines commonly used in older people: influenza and tetanus/diphtheria/pertussis vaccines, with increases in diagnosis-free time of 14%-27%.
Reduced Risk or Delayed Diagnosis?
Speaking at a Science Media Centre press conference on the study, lead author Maxime Taquet, PhD, FRCPsych, clinical lecturer in psychiatry at the University of Oxford, noted that the total number of dementia cases were similar in the two shingles vaccine groups by the end of the 6-year follow-up period but there was a difference in the time at which they received a diagnosis of dementia.
“The study suggests that rather than actually reducing dementia risk, the recombinant vaccine delays the onset of dementia compared to the live vaccine in patients who go on to develop the condition,” he explained.
But when comparing the recombinant vaccine with the influenza and tetanus/diphtheria/pertussis vaccines there was a clear reduction in dementia risk itself, Dr. Taquet reported.
“It might well be that the live vaccine has a potential effect on the risk of dementia itself and therefore the recombinant vaccine only shows a delay in dementia compared to the live vaccine, but both of them might decrease the overall risk of dementia,” he suggested.
But the researchers cautioned that this study could not prove causality.
“While the two groups were very carefully matched in terms of factors that might influence the development of dementia, we still have to be cautious before assuming that the vaccine is indeed causally reducing the risk of onset of dementia,” Dr. Harrison warned.
The researchers say the results would need to be confirmed in a randomized trial, which may have to be conducted in a slightly younger age group, as currently shingles vaccine is recommended for all older individuals in the United Kingdom.
Vaccine recommendations vary from country to country, Dr. Harrison added. In the United States, the Centers for Disease Control and Prevention recommends the recombinant shingles vaccine for all adults aged 50 years or older.
In the meantime, it would be interesting to see whether further observational studies in other countries find similar results as this US study, Dr. Harrison said.
Mechanism Uncertain
Speculating on a possible mechanism behind the findings, Dr. Harrison suggested two plausible explanations.
“First, it is thought that the herpes virus could be one of many factors that could promote dementia, so a vaccine that stops reactivation of this virus might therefore be delaying that process,” he noted.
The other possibility is that adjuvants included in the recombinant vaccine to stimulate the immune system might have played a role.
“We don’t have any data on the mechanism, and thus study did not address that, so further studies are needed to look into this,” Dr. Harrison said.
Stronger Effect in Women
Another intriguing finding is that the association with the recombinant vaccine and delayed dementia diagnosis seemed to be stronger in women vs men.
In the original study of the live shingles vaccine, a protective effect against dementia was shown only in women.
In the current study, the delay in dementia diagnosis was seen in both sexes but was stronger in women, showing a 22% increased time without dementia in women versus a 13% increased time in men with the recombinant versus the live vaccine.
As expected, the recombinant vaccine was associated with a lower risk for shingles disease vs the live vaccine (2.5% versus 3.5%), but women did not have a better response than men did in this respect.
“The better protection against shingles with the recombinant vaccine was similar in men and women, an observation that might be one reason to question the possible mechanism behind the dementia effect being better suppression of the herpes zoster virus by the recombinant vaccine,” Dr. Harrison commented.
Though these findings are not likely to lead to any immediate changes in policy regarding the shingles vaccine, Dr. Harrison said it would be interesting to see whether uptake of the vaccine increased after this study.
He estimated that, currently in the United Kingdom, about 60% of older adults choose to have the shingles vaccine. A 2020 study in the United States found that only about one-third of US adults over 60 had received the vaccine.
“It will be interesting to see if that figure increases after these data are publicized, but I am not recommending that people have the vaccine specifically to lower their risk of dementia because of the caveats about the study that we have discussed,” he commented.
Outside Experts Positive
Outside experts, providing comment to the Science Media Centre, welcomed the new research.
“ The study is very well-conducted and adds to previous data indicating that vaccination against shingles is associated with lower dementia risk. More research is needed in future to determine why this vaccine is associated with lower dementia risk,” said Tara Spires-Jones, FMedSci, president of the British Neuroscience Association.
The high number of patients in the study and the adjustments for potential confounders are also strong points, noted Andrew Doig, PhD, professor of biochemistry, University of Manchester, Manchester, England.
“This is a significant result, comparable in effectiveness to the recent antibody drugs for Alzheimer’s disease,” Dr. Doig said. “Administering the recombinant shingles vaccine could well be a simple and cheap way to lower the risk of Alzheimer’s disease.”
Dr. Doig noted that a link between herpes zoster infection and the onset of dementia has been suspected for some time, and a trial of the antiviral drug valacyclovir against Alzheimer’s disease is currently underway.
In regard to the shingles vaccine, he said a placebo-controlled trial would be needed to prove causality.
“We also need to see how many years the effect might last and whether we should vaccinate people at a younger age. We know that the path to Alzheimer’s can start decades before any symptoms are apparent, so the vaccine might be even more effective if given to people in their 40s or 50s,” he said.
Dr. Harrison and Dr. Taquet reported no disclosures. Dr. Doig is a founder, director, and consultant for PharmaKure, which works on Alzheimer’s drugs and diagnostics. Other commentators declared no disclosures.
A version of this article first appeared on Medscape.com.
FROM NATURE MEDICINE
Brain Structure Differs in Youth With Conduct Disorder
Youth with conduct disorder (CD) have extensive brain structure differences, new research showed.
“We know very little about this disorder even though it can carry a high burden for families and societies,” co–lead author Yidian Gao, PhD, of the University of Birmingham, Birmingham, England, said in a press release.
“The sample included in our study is 10-20 times larger than previous studies and contains data on children from North America, Europe, and Asia. It provides the most compelling evidence to date that CD is associated with widespread structural brain differences,” he added.
The findings were published online in The Lancet Psychiatry.
An Understudied Disorder
In the largest study of its kind, researchers at the Universities of Bath and Birmingham, both in England, collaborated with research teams across Europe, North America, and Asia, as part of the Enhancing NeuroImaging Genetics through Meta-Analysis–Antisocial Behavior Working Group to learn more about one of the “least researched psychiatric disorders,” they wrote.
The investigators used MRI to examine the brain structure of 1185 children with a clinical diagnosis of CD and 1253 typically developing children from 17-21 across 15 international study cohorts.
After adjusting for total intracranial volume investigators found that youth with CD (29% women; mean age, 13.7 years) had lower total surface area and lower regional surface area in 26 of the 34 cortical regions, spanning all four lobes of the brain, compared with their typically developing counterparts (35.6% women; mean age, 13.5 years).
Youth with CD also showed greater cortical thickness in the caudal anterior cingulate cortex (P = .0001) and lower cortical thickness in the banks of the superior temporal sulcus vs those without CD (P = .0010).
In addition, the CD group also had lower volume in the thalamus (P = .0009), amygdala (P = .0014), hippocampus (P = .0031), and nucleus accumbens (P = .0052).
Most findings remained significant after adjusting for intelligence quotient, psychiatric comorbidities, and psychotropic medication use. Of note, group difference in cortical thickness, 22 of 27 differences in surface area. In addition, three of four subcortical differences remained robust after adjusting for co-occurring attention-deficit/hyperactivity disorder, the most frequent comorbidity.
When the investigators divided individuals with CD into two subgroups — those with high vs low levels of callous-unemotional traits — they found limited overall differences. However, those with high callous-unemotional traits had lower surface area in the superior temporal and superior frontal gyri vs those with low callous-unemotional traits and the typically developing group.
Investigators also found that individuals with childhood-onset CD had greater cortical thickness in the caudal anterior cingulate cortex compared with those with adolescent-onset CD.
Study limitations include comparison of different cohorts with differing protocols that could affect the validity of the findings. In addition, subgroup samples were small and had lower statistical power.
“Our finding of robust brain alterations in conduct disorder — similar to those in more widely recognized and widely treated disorders such as ADHD — emphasize the need for a greater focus on conduct disorder in research, treatment, and public policy,” the authors noted.
Seven study authors reported conflicts of interest with various pharmaceutical companies and other organizations.
A version of this article first appeared on Medscape.com.
Youth with conduct disorder (CD) have extensive brain structure differences, new research showed.
“We know very little about this disorder even though it can carry a high burden for families and societies,” co–lead author Yidian Gao, PhD, of the University of Birmingham, Birmingham, England, said in a press release.
“The sample included in our study is 10-20 times larger than previous studies and contains data on children from North America, Europe, and Asia. It provides the most compelling evidence to date that CD is associated with widespread structural brain differences,” he added.
The findings were published online in The Lancet Psychiatry.
An Understudied Disorder
In the largest study of its kind, researchers at the Universities of Bath and Birmingham, both in England, collaborated with research teams across Europe, North America, and Asia, as part of the Enhancing NeuroImaging Genetics through Meta-Analysis–Antisocial Behavior Working Group to learn more about one of the “least researched psychiatric disorders,” they wrote.
The investigators used MRI to examine the brain structure of 1185 children with a clinical diagnosis of CD and 1253 typically developing children from 17-21 across 15 international study cohorts.
After adjusting for total intracranial volume investigators found that youth with CD (29% women; mean age, 13.7 years) had lower total surface area and lower regional surface area in 26 of the 34 cortical regions, spanning all four lobes of the brain, compared with their typically developing counterparts (35.6% women; mean age, 13.5 years).
Youth with CD also showed greater cortical thickness in the caudal anterior cingulate cortex (P = .0001) and lower cortical thickness in the banks of the superior temporal sulcus vs those without CD (P = .0010).
In addition, the CD group also had lower volume in the thalamus (P = .0009), amygdala (P = .0014), hippocampus (P = .0031), and nucleus accumbens (P = .0052).
Most findings remained significant after adjusting for intelligence quotient, psychiatric comorbidities, and psychotropic medication use. Of note, group difference in cortical thickness, 22 of 27 differences in surface area. In addition, three of four subcortical differences remained robust after adjusting for co-occurring attention-deficit/hyperactivity disorder, the most frequent comorbidity.
When the investigators divided individuals with CD into two subgroups — those with high vs low levels of callous-unemotional traits — they found limited overall differences. However, those with high callous-unemotional traits had lower surface area in the superior temporal and superior frontal gyri vs those with low callous-unemotional traits and the typically developing group.
Investigators also found that individuals with childhood-onset CD had greater cortical thickness in the caudal anterior cingulate cortex compared with those with adolescent-onset CD.
Study limitations include comparison of different cohorts with differing protocols that could affect the validity of the findings. In addition, subgroup samples were small and had lower statistical power.
“Our finding of robust brain alterations in conduct disorder — similar to those in more widely recognized and widely treated disorders such as ADHD — emphasize the need for a greater focus on conduct disorder in research, treatment, and public policy,” the authors noted.
Seven study authors reported conflicts of interest with various pharmaceutical companies and other organizations.
A version of this article first appeared on Medscape.com.
Youth with conduct disorder (CD) have extensive brain structure differences, new research showed.
“We know very little about this disorder even though it can carry a high burden for families and societies,” co–lead author Yidian Gao, PhD, of the University of Birmingham, Birmingham, England, said in a press release.
“The sample included in our study is 10-20 times larger than previous studies and contains data on children from North America, Europe, and Asia. It provides the most compelling evidence to date that CD is associated with widespread structural brain differences,” he added.
The findings were published online in The Lancet Psychiatry.
An Understudied Disorder
In the largest study of its kind, researchers at the Universities of Bath and Birmingham, both in England, collaborated with research teams across Europe, North America, and Asia, as part of the Enhancing NeuroImaging Genetics through Meta-Analysis–Antisocial Behavior Working Group to learn more about one of the “least researched psychiatric disorders,” they wrote.
The investigators used MRI to examine the brain structure of 1185 children with a clinical diagnosis of CD and 1253 typically developing children from 17-21 across 15 international study cohorts.
After adjusting for total intracranial volume investigators found that youth with CD (29% women; mean age, 13.7 years) had lower total surface area and lower regional surface area in 26 of the 34 cortical regions, spanning all four lobes of the brain, compared with their typically developing counterparts (35.6% women; mean age, 13.5 years).
Youth with CD also showed greater cortical thickness in the caudal anterior cingulate cortex (P = .0001) and lower cortical thickness in the banks of the superior temporal sulcus vs those without CD (P = .0010).
In addition, the CD group also had lower volume in the thalamus (P = .0009), amygdala (P = .0014), hippocampus (P = .0031), and nucleus accumbens (P = .0052).
Most findings remained significant after adjusting for intelligence quotient, psychiatric comorbidities, and psychotropic medication use. Of note, group difference in cortical thickness, 22 of 27 differences in surface area. In addition, three of four subcortical differences remained robust after adjusting for co-occurring attention-deficit/hyperactivity disorder, the most frequent comorbidity.
When the investigators divided individuals with CD into two subgroups — those with high vs low levels of callous-unemotional traits — they found limited overall differences. However, those with high callous-unemotional traits had lower surface area in the superior temporal and superior frontal gyri vs those with low callous-unemotional traits and the typically developing group.
Investigators also found that individuals with childhood-onset CD had greater cortical thickness in the caudal anterior cingulate cortex compared with those with adolescent-onset CD.
Study limitations include comparison of different cohorts with differing protocols that could affect the validity of the findings. In addition, subgroup samples were small and had lower statistical power.
“Our finding of robust brain alterations in conduct disorder — similar to those in more widely recognized and widely treated disorders such as ADHD — emphasize the need for a greater focus on conduct disorder in research, treatment, and public policy,” the authors noted.
Seven study authors reported conflicts of interest with various pharmaceutical companies and other organizations.
A version of this article first appeared on Medscape.com.
FROM THE LANCET PSYCHIATRY
Bidirectional Link for Mental Health and Diabetic Complications
TOPLINE:
Mental health disorders increase the likelihood of developing chronic diabetic complications and vice versa across all age groups in patients with type 1 diabetes (T1D) or type 2 diabetes (T2D).
METHODOLOGY:
- Researchers used a US national healthcare claims database (data obtained from 2001 to 2018) to analyze individuals with and without T1D and T2D, who had no prior mental health disorder or chronic diabetic complication.
- The onset and presence of chronic diabetic complications and mental health disorders were identified to determine their possible association.
- Individuals were stratified by age: 0-19, 20-39, 40-59, and ≥ 60 years.
TAKEAWAY:
- Researchers analyzed 44,735 patients with T1D (47.5% women) and 152,187 with T2D (46.0% women), who were matched with 356,630 individuals without diabetes (51.8% women).
- The presence of chronic diabetic complications increased the risk for a mental health disorder across all age groups, with the highest risk seen in patients aged ≥ 60 years (hazard ratio [HR], 2.9).
- Similarly, diagnosis of a mental health disorder increased the risk for chronic diabetic complications across all age groups, with the highest risk seen in patients aged 0-19 years (HR, 2.5).
- Patients with T2D had a significantly higher risk for a mental health disorder and a lower risk for chronic diabetic complications than those with T1D across all age groups, except those aged ≥ 60 years.
- The bidirectional association between mental health disorders and chronic diabetic complications was not affected by the diabetes type (P > .05 for all interactions).
IN PRACTICE:
“Clinicians and healthcare systems likely need to increase their focus on MHDs [mental health disorders], and innovative models of care are required to optimize care for both individuals with type 1 diabetes and those with type 2 diabetes,” the authors wrote.
SOURCE:
The study, led by Maya Watanabe, Department of Biostatistics, University of Michigan, Ann Arbor, Michigan, was published online in Diabetes Care.
LIMITATIONS:
The study relied on International Classification of Diseases 9th and 10th revision codes, which might have led to misclassification of mental health conditions, chronic diabetes complications, and diabetes type. The data did not capture the symptom onset and severity. The findings may not be generalizable to populations outside the United States.
DISCLOSURES:
The study was supported by the Juvenile Diabetes Research Foundation (now Breakthrough T1D). Some authors reported receiving speaker or expert testimony honoraria and research support, and some declared serving on medical or digital advisory boards or as consultants for various pharmaceutical and medical device companies.
A version of this article first appeared on Medscape.com.
TOPLINE:
Mental health disorders increase the likelihood of developing chronic diabetic complications and vice versa across all age groups in patients with type 1 diabetes (T1D) or type 2 diabetes (T2D).
METHODOLOGY:
- Researchers used a US national healthcare claims database (data obtained from 2001 to 2018) to analyze individuals with and without T1D and T2D, who had no prior mental health disorder or chronic diabetic complication.
- The onset and presence of chronic diabetic complications and mental health disorders were identified to determine their possible association.
- Individuals were stratified by age: 0-19, 20-39, 40-59, and ≥ 60 years.
TAKEAWAY:
- Researchers analyzed 44,735 patients with T1D (47.5% women) and 152,187 with T2D (46.0% women), who were matched with 356,630 individuals without diabetes (51.8% women).
- The presence of chronic diabetic complications increased the risk for a mental health disorder across all age groups, with the highest risk seen in patients aged ≥ 60 years (hazard ratio [HR], 2.9).
- Similarly, diagnosis of a mental health disorder increased the risk for chronic diabetic complications across all age groups, with the highest risk seen in patients aged 0-19 years (HR, 2.5).
- Patients with T2D had a significantly higher risk for a mental health disorder and a lower risk for chronic diabetic complications than those with T1D across all age groups, except those aged ≥ 60 years.
- The bidirectional association between mental health disorders and chronic diabetic complications was not affected by the diabetes type (P > .05 for all interactions).
IN PRACTICE:
“Clinicians and healthcare systems likely need to increase their focus on MHDs [mental health disorders], and innovative models of care are required to optimize care for both individuals with type 1 diabetes and those with type 2 diabetes,” the authors wrote.
SOURCE:
The study, led by Maya Watanabe, Department of Biostatistics, University of Michigan, Ann Arbor, Michigan, was published online in Diabetes Care.
LIMITATIONS:
The study relied on International Classification of Diseases 9th and 10th revision codes, which might have led to misclassification of mental health conditions, chronic diabetes complications, and diabetes type. The data did not capture the symptom onset and severity. The findings may not be generalizable to populations outside the United States.
DISCLOSURES:
The study was supported by the Juvenile Diabetes Research Foundation (now Breakthrough T1D). Some authors reported receiving speaker or expert testimony honoraria and research support, and some declared serving on medical or digital advisory boards or as consultants for various pharmaceutical and medical device companies.
A version of this article first appeared on Medscape.com.
TOPLINE:
Mental health disorders increase the likelihood of developing chronic diabetic complications and vice versa across all age groups in patients with type 1 diabetes (T1D) or type 2 diabetes (T2D).
METHODOLOGY:
- Researchers used a US national healthcare claims database (data obtained from 2001 to 2018) to analyze individuals with and without T1D and T2D, who had no prior mental health disorder or chronic diabetic complication.
- The onset and presence of chronic diabetic complications and mental health disorders were identified to determine their possible association.
- Individuals were stratified by age: 0-19, 20-39, 40-59, and ≥ 60 years.
TAKEAWAY:
- Researchers analyzed 44,735 patients with T1D (47.5% women) and 152,187 with T2D (46.0% women), who were matched with 356,630 individuals without diabetes (51.8% women).
- The presence of chronic diabetic complications increased the risk for a mental health disorder across all age groups, with the highest risk seen in patients aged ≥ 60 years (hazard ratio [HR], 2.9).
- Similarly, diagnosis of a mental health disorder increased the risk for chronic diabetic complications across all age groups, with the highest risk seen in patients aged 0-19 years (HR, 2.5).
- Patients with T2D had a significantly higher risk for a mental health disorder and a lower risk for chronic diabetic complications than those with T1D across all age groups, except those aged ≥ 60 years.
- The bidirectional association between mental health disorders and chronic diabetic complications was not affected by the diabetes type (P > .05 for all interactions).
IN PRACTICE:
“Clinicians and healthcare systems likely need to increase their focus on MHDs [mental health disorders], and innovative models of care are required to optimize care for both individuals with type 1 diabetes and those with type 2 diabetes,” the authors wrote.
SOURCE:
The study, led by Maya Watanabe, Department of Biostatistics, University of Michigan, Ann Arbor, Michigan, was published online in Diabetes Care.
LIMITATIONS:
The study relied on International Classification of Diseases 9th and 10th revision codes, which might have led to misclassification of mental health conditions, chronic diabetes complications, and diabetes type. The data did not capture the symptom onset and severity. The findings may not be generalizable to populations outside the United States.
DISCLOSURES:
The study was supported by the Juvenile Diabetes Research Foundation (now Breakthrough T1D). Some authors reported receiving speaker or expert testimony honoraria and research support, and some declared serving on medical or digital advisory boards or as consultants for various pharmaceutical and medical device companies.
A version of this article first appeared on Medscape.com.
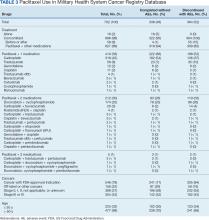
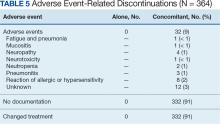
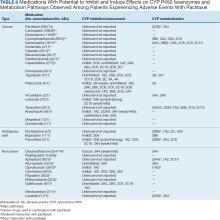
Paclitaxel Drug-Drug Interactions in the Military Health System
Background
Paclitaxel was first derived from the bark of the yew tree (Taxus brevifolia). It was discovered as part of a National Cancer Institute program screen of plants and natural products with putative anticancer activity during the 1960s.1-9 Paclitaxel works by suppressing spindle microtube dynamics, which results in the blockage of the metaphase-anaphase transitions, inhibition of mitosis, and induction of apoptosis in a broad spectrum of cancer cells. Paclitaxel also displayed additional anticancer activities, including the suppression of cell proliferation and antiangiogenic effects. However, since the growth of normal body cells may also be affected, other adverse effects (AEs) will also occur.8-18
Two different chemotherapy drugs contain paclitaxel—paclitaxel and nab-paclitaxel—and the US Food and Drug Administration (FDA) recognizes them as separate entities.19-21 Taxol (paclitaxel) was approved by the FDA in 1992 for treating advanced ovarian cancer.20 It has since been approved for the treatment of metastatic breast cancer, AIDS-related Kaposi sarcoma (as an orphan drug), non-small cell lung cancer (NSCLC), and cervical cancers (in combination withbevacizumab) in 1994, 1997, 1999, and 2014, respectively.21 Since 2002, a generic version of Taxol, known as paclitaxel injectable, has been FDA-approved from different manufacturers. According to the National Cancer Institute, a combination of carboplatin and Taxol is approved to treat carcinoma of unknown primary, cervical, endometrial, NSCLC, ovarian, and thymoma cancers.19 Abraxane (nab-paclitaxel) was FDA-approved to treat metastatic breast cancer in 2005. It was later approved for first-line treatment of advanced NSCLC and late-stage pancreatic cancer in 2012 and 2013, respectively. In 2018 and 2020, both Taxol and Abraxane were approved for first-line treatment of metastatic squamous cell NSCLC in combination with carboplatin and pembrolizumab and metastatic triple-negative breast cancer in combination with pembrolizumab, respectively.22-26 In 2019, Abraxane was approved with atezolizumab to treat metastatic triple-negative breast cancer, but this approval was withdrawn in 2021. In 2022, a generic version of Abraxane, known as paclitaxel protein-bound, was released in the United States. Furthermore, paclitaxel-containing formulations also are being studied in the treatment of other types of cancer.19-32
One of the main limitations of paclitaxel is its low solubility in water, which complicates its drug supply. To distribute this hydrophobic anticancer drug efficiently, paclitaxel is formulated and administered to patients via polyethoxylated castor oil or albumin-bound (nab-paclitaxel). However, polyethoxylated castor oil induces complement activation and is the cause of common hypersensitivity reactions related to paclitaxel use.2,17,33-38 Therefore, many alternatives to polyethoxylated castor oil have been researched.
Since 2000, new paclitaxel formulations have emerged using nanomedicine techniques. The difference between these formulations is the drug vehicle. Different paclitaxel-based nanotechnological vehicles have been developed and approved, such as albumin-based nanoparticles, polymeric lipidic nanoparticles, polymeric micelles, and liposomes, with many others in clinical trial phases.3,37 Albumin-based nanoparticles have a high response rate (33%), whereas the response rate for polyethoxylated castor oil is 25% in patients with metastatic breast cancer.33,39-52 The use of paclitaxel dimer nanoparticles also has been proposed as a method for increasing drug solubility.33,53
Paclitaxel is metabolized by cytochrome P450 (CYP) isoenzymes 2C8 and 3A4. When administering paclitaxel with known inhibitors, inducers, or substrates of CYP2C8 or CYP3A4, caution is required.19-22 Regulations for CYP research were not issued until 2008, so potential interactions between paclitaxel and other drugs have not been extensively evaluated in clinical trials. A study of 12 kinase inhibitors showed strong inhibition of CYP2C8 and/or CYP3A4 pathways by these inhibitors, which could alter the ratio of paclitaxel metabolites in vivo, leading to clinically relevant changes.54 Differential metabolism has been linked to paclitaxel-induced neurotoxicity in patients with cancer.55 Nonetheless, variants in the CYP2C8, CYP3A4, CYP3A5, and ABCB1 genes do not account for significant interindividual variability in paclitaxel pharmacokinetics.56 In liver microsomes, losartan inhibited paclitaxel metabolism when used at concentrations > 50 µmol/L.57 Many drug-drug interaction (DDI) studies of CYP2C8 and CYP3A4 have shown similar results for paclitaxel.58-64
The goals of this study are to investigate prescribed drugs used with paclitaxel and determine patient outcomes through several Military Health System (MHS) databases. The investigation focused on (1) the functions of paclitaxel; (2) identifying AEs that patients experienced; (3) evaluating differences when paclitaxel is used alone vs concomitantly and between the completed vs discontinued treatment groups; (4) identifying all drugs used during paclitaxel treatment; and (5) evaluating DDIs with antidepressants (that have an FDA boxed warning and are known to have DDIs confirmed in previous publications) and other drugs.65-67
The Walter Reed National Military Medical Center in Bethesda, Maryland, institutionalreview board approved the study protocol and ensured compliance with the Health Insurance Portability and Accountability Act as an exempt protocol. The Joint Pathology Center (JPC) of the US Department of Defense (DoD) Cancer Registry Program and MHS data experts from the Comprehensive Ambulatory/Professional Encounter Record (CAPER) and the Pharmacy Data Transaction Service (PDTS) provided data for the analysis.
METHODS
The DoD Cancer Registry Program was established in 1986 and currently contains data from 1998 to 2024. CAPER and PDTS are part of the MHS Data Repository/Management Analysis and Reporting Tool database. Each observation in the CAPER record represents an ambulatory encounter at a military treatment facility (MTF). CAPER includes data from 2003 to 2024.
Each observation in the PDTS record represents a prescription filled for an MHS beneficiary at an MTF through the TRICARE mail-order program or a US retail pharmacy. Missing from this record are prescriptions filled at international civilian pharmacies and inpatient pharmacy prescriptions. The MHS Data Repository PDTS record is available from 2002 to 2024. The legacy Composite Health Care System is being replaced by GENESIS at MTFs.
Data Extraction Design
The study design involved a cross-sectional analysis. We requested data extraction for paclitaxel from 1998 to 2022. Data from the DoD Cancer Registry Program were used to identify patients who received cancer treatment. Once patients were identified, the CAPER database was searched for diagnoses to identify other health conditions, whereas the PDTS database was used to populate a list of prescription medications filled during chemotherapy treatment.
Data collected from the JPC included cancer treatment, cancer information, demographics, and physicians’ comments on AEs. Collected data from the MHS include diagnosis and filled prescription history from initiation to completion of the therapy period (or 2 years after the diagnosis date). For the analysis of the DoD Cancer Registry Program and CAPER databases, we used all collected data without excluding any. When analyzing PDTS data, we excluded patients with PDTS data but without a record of paclitaxel being filled, or medications filled outside the chemotherapy period (by evaluating the dispensed date and day of supply).
Data Extraction Analysis
The Surveillance, Epidemiology, and End Results Program Coding and Staging Manual 2016 and the International Classification of Diseases for Oncology, 3rd edition, 1st revision, were used to decode disease and cancer types.68,69 Data sorting and analysis were performed using Microsoft Excel. The percentage for the total was calculated by using the number of patients or data available within the paclitaxel groups divided by the total number of patients or data variables. The subgroup percentage was calculated by using the number of patients or data available within the subgroup divided by the total number of patients in that subgroup.
In alone vs concomitant and completed vs discontinued treatment groups, a 2-tailed, 2-sample z test was used to statistical significance (P < .05) using a statistics website.70 Concomitant was defined as paclitaxel taken with other antineoplastic agent(s) before, after, or at the same time as cancer therapy. For the retrospective data analysis, physicians’ notes with a period, comma, forward slash, semicolon, or space between medication names were interpreted as concurrent, whereas plus (+), minus/plus (-/+), or “and” between drug names that were dispensed on the same day were interpreted as combined with known common combinations: 2 drugs (DM886 paclitaxel and carboplatin and DM881-TC-1 paclitaxel and cisplatin) or 3 drugs (DM887-ACT doxorubicin, cyclophosphamide, and paclitaxel). Completed treatment was defined as paclitaxel as the last medication the patient took without recorded AEs; switching or experiencing AEs was defined as discontinued treatment.
RESULTS
The JPC provided 702 entries for 687 patients with a mean age of 56 years (range, 2 months to 88 years) who were treated with paclitaxel from March 1996 to October 2021. Fifteen patients had duplicate entries because they had multiple cancer sites or occurrences. There were 623 patients (89%) who received paclitaxel for FDA-approved indications. The most common types of cancer identified were 344 patients with breast cancer (49%), 91 patients with lung cancer (13%), 79 patients with ovarian cancer (11%), and 75 patients with endometrial cancer (11%) (Table 1). Seventy-nine patients (11%) received paclitaxel for cancers that were not for FDA-approved indications, including 19 for cancers of the fallopian tube (3%) and 17 for esophageal cancer (2%) (Table 2).
There were 477 patients (68%) aged > 50 years. A total of 304 patients (43%) had a stage III or IV cancer diagnosis and 398 (57%) had stage II or lower (combination of data for stages 0, I, and II; not applicable; and unknown) cancer diagnosis. For systemic treatment, 16 patients (2%) were treated with paclitaxel alone and 686 patients (98%) received paclitaxel concomitantly with additional chemotherapy: 59 patients (9%) in the before or after group, 410 patients (58%) had a 2-drug combination, 212 patients (30%) had a 3-drug combination, and 5 patients (1%) had a 4-drug combination. In addition, for doublet therapies, paclitaxel combined with carboplatin, trastuzumab, gemcitabine, or cisplatin had more patients (318, 58, 12, and 11, respectively) than other combinations (≤ 4 patients). For triplet therapies, paclitaxel combined withdoxorubicin plus cyclophosphamide or carboplatin plus bevacizumab had more patients (174 and 20, respectively) than other combinations, including quadruplet therapies (≤ 4 patients) (Table 3).
Patients were more likely to discontinue paclitaxel if they received concomitant treatment. None of the 16 patients receiving paclitaxel monotherapy experienced AEs, whereas 364 of 686 patients (53%) treated concomitantly discontinued (P < .001). Comparisons of 1 drug vs combination (2 to 4 drugs) and use for treating cancers that were FDA-approved indications vs off-label use were significant (P < .001), whereas comparisons of stage II or lower vs stage III and IV cancer and of those aged ≤ 50 years vs aged > 50 years were not significant (P = .50 andP = .30, respectively) (Table 4).
Among the 364 patients who had concomitant treatment and had discontinued their treatment, 332 (91%) switched treatments with no AEs documented and 32 (9%) experienced fatigue with pneumonia, mucositis, neuropathy, neurotoxicity, neutropenia, pneumonitis, allergic or hypersensitivity reaction, or an unknown AE. Patients who discontinued treatment because of unknown AEs had a physician’s note that detailed progressive disease, a significant decline in performance status, and another unknown adverse effect due to a previous sinus tract infection and infectious colitis (Table 5).
Management Analysis and Reporting Tool Database
MHS data analysts provided data on diagnoses for 639 patients among 687 submitteddiagnoses, with 294 patients completing and 345 discontinuing paclitaxel treatment. Patients in the completed treatment group had 3 to 258 unique health conditions documented, while patients in the discontinued treatment group had 4 to 181 unique health conditions documented. The MHS reported 3808 unique diagnosis conditions for the completed group and 3714 for the discontinued group (P = .02).
The mean (SD) number of diagnoses was 51 (31) for the completed and 55 (28) for the discontinued treatment groups (Figure). Among 639 patients who received paclitaxel, the top 5 diagnoses were administrative, including encounters for other administrative examinations; antineoplastic chemotherapy; administrative examination for unspecified; other specified counseling; and adjustment and management of vascular access device. The database does not differentiate between administrative and clinically significant diagnoses.
MHS data analysts provided data for 336 of 687 submitted patients who were prescribed paclitaxel; 46 patients had no PDTS data, and 305 patients had PDTS data without paclitaxel, Taxol, or Abraxane dispensed. Medications that were filled outside the chemotherapy period were removed by evaluating the dispensed date and day of supply. Among these 336 patients, 151 completed the treatment and 185 discontinued, with 14 patients experiencing documented AEs. Patients in the completed treatment group filled 9 to 56 prescriptions while patients in the discontinued treatment group filled 6 to 70 prescriptions.Patients in the discontinued group filled more prescriptions than those who completed treatment: 793 vs 591, respectively (P = .34).
The mean (SD) number of filled prescription drugs was 24 (9) for the completed and 34 (12) for the discontinued treatment group. The 5 most filled prescriptions with paclitaxel from 336 patients with PDTS data were dexamethasone (324 prescriptions with 14 recorded AEs), diphenhydramine (296 prescriptions with 12 recorded AEs), ondansetron (277 prescriptions with 11 recorded AEs), prochlorperazine (265 prescriptions with 12 recorded AEs), and sodium chloride (232 prescriptions with 11 recorded AEs).
DISCUSSION
As a retrospective review, this study is more limited in the strength of its conclusions when compared to randomized control trials. The DoD Cancer Registry Program only contains information about cancer types, stages, treatment regimens, and physicians’ notes. Therefore, noncancer drugs are based solely on the PDTS database. In most cases, physicians' notes on AEs were not detailed. There was no distinction between initial vs later lines of therapy and dosage reductions. The change in status or appearance of a new medical condition did not indicate whether paclitaxel caused the changes to develop or directly worsen a pre-existing condition. The PDTS records prescriptions filled, but that may not reflect patients taking prescriptions.
Paclitaxel
Paclitaxel has a long list of both approved and off-label uses in malignancies as a primary agent and in conjunction with other drugs. The FDA prescribing information for Taxol and Abraxane was last updated in April 2011 and September 2020, respectively.20,21 The National Institutes of Health National Library of Medicine has the current update for paclitaxel on July 2023.19,22 Thus, the prescribed information for paclitaxel referenced in the database may not always be up to date. The combinations of paclitaxel with bevacizumab, carboplatin, or carboplatin and pembrolizumab were not in the Taxol prescribing information. Likewise, a combination of nab-paclitaxel with atezolizumab or carboplatin and pembrolizumab is missing in the Abraxane prescribing information.22-27
The generic name is not the same as a generic drug, which may have slight differences from the brand name product.71 The generic drug versions of Taxol and Abraxane have been approved by the FDA as paclitaxel injectable and paclitaxel-protein bound, respectively. There was a global shortage of nab-paclitaxel from October 2021 to June 2022 because of a manufacturing problem.72 During this shortage, data showed similar comments from physician documents that treatment switched to Taxol due to the Abraxane shortage.
Of 336 patients in the PDTS database with dispensed paclitaxel prescriptions, 276 received paclitaxel (year dispensed, 2013-2022), 27 received Abraxane (year dispensed, 2013-2022), 47 received Taxol (year dispensed, 2004-2015), 8 received both Abraxane and paclitaxel, and 6 received both Taxol and paclitaxel. Based on this information, it appears that the distinction between the drugs was not made in the PDTS until after 2015, 10 years after Abraxane received FDA approval. Abraxane was prescribed in the MHS in 2013, 8 years after FDA approval. There were a few comparison studies of Abraxane and Taxol.73-76
Safety and effectiveness in pediatric patients have not been established for paclitaxel. According to the DoD Cancer Registry Program, the youngest patient was aged 2 months. In 2021, this patient was diagnosed with corpus uteri and treated with carboplatin and Taxol in course 1; in course 2, the patient reacted to Taxol; in course 3, Taxol was replaced with Abraxane; in courses 4 to 7, the patient was treated with carboplatin only.
Discontinued Treatment
Ten patients had prescribed Taxol that was changed due to AEs: 1 was switched to Abraxane and atezolizumab, 3 switched to Abraxane, 2 switched to docetaxel, 1 switched to doxorubicin, and 3 switched to pembrolizumab (based on physician’s comments). Of the 10 patients, 7 had Taxol reaction, 2 experienced disease progression, and 1 experienced high programmed death–ligand 1 expression (this patient with breast cancer was switched to Abraxane and atezolizumab during the accelerated FDA approval phase for atezolizumab, which was later revoked). Five patients were treated with carboplatin and Taxol for cancer of the anal canal (changed to pembrolizumab after disease progression), lung not otherwise specified (changed to carboplatin and pembrolizumab due to Taxol reaction), lower inner quadrant of the breast (changed to doxorubicin due to hypersensitivity reaction), corpus uteri (changed to Abraxane due to Taxol reaction), and ovary (changed to docetaxel due to Taxol reaction). Three patients were treated with doxorubicin, cyclophosphamide, and Taxol for breast cancer; 2 patients with breast cancer not otherwise specified switched to Abraxane due to cardiopulmonary hypersensitivity and Taxol reaction and 1 patient with cancer of the upper outer quadrant of the breast changed to docetaxel due to allergic reaction. One patient, who was treated with paclitaxel, ifosfamide, and cisplatin for metastasis of the lower lobe of the lung and kidney cancer, experienced complications due to infectious colitis (treated with ciprofloxacin) and then switched to pembrolizumab after the disease progressed. These AEs are known in paclitaxel medical literature on paclitaxel AEs.19-24,77-81
Combining 2 or more treatments to target cancer-inducing or cell-sustaining pathways is a cornerstone of chemotherapy.82-84 Most combinations are given on the same day, but some are not. For 3- or 4-drug combinations, doxorubicin and cyclophosphamide were given first, followed by paclitaxel with or withouttrastuzumab, carboplatin, or pembrolizumab. Only 16 patients (2%) were treated with paclitaxel alone; therefore, the completed and discontinued treatment groups are mostly concomitant treatment. As a result, the comparisons of the completed and discontinued treatment groups were almost the same for the diagnosis. The PDTS data have a better result because 2 exclusion criteria were applied before narrowing the analysis down to paclitaxel treatment specifically.
Antidepressants and Other Drugs
Drug response can vary from person to person and can lead to treatment failure related to AEs. One major factor in drug metabolism is CYP.85 CYP2C8 is the major pathway for paclitaxel and CYP3A4 is the minor pathway. When evaluating the noncancer drugs, there were no reports of CYP2C8 inhibition or induction. Over the years, many DDI warnings have been issued for paclitaxel with different drugs in various electronic resources.
Oncologists follow guidelines to prevent DDIs, as paclitaxel is known to have severe, moderate, and minor interactions with other drugs. Among 687 patients, 261 (38%) were prescribed any of 14 antidepressants. Eight of these antidepressants (amitriptyline, citalopram, desipramine, doxepin, venlafaxine, escitalopram, nortriptyline, and trazodone) are metabolized, 3 (mirtazapine, sertraline, and fluoxetine) are metabolized and inhibited, 2 (bupropion and duloxetine) are neither metabolized nor inhibited, and 1 (paroxetine) is inhibited by CYP3A4. Duloxetine, venlafaxine, and trazodone were more commonly dispensed (84, 78, and 42 patients, respectively) than others (≤ 33 patients).
Of 32 patients with documented AEs,14 (44%) had 168 dispensed drugs in the PDTS database. Six patients (19%) were treated with doxorubicin and cyclophosphamide followed by paclitaxel for breast cancer; 6 (19%) were treated with carboplatin and paclitaxel for cancer of the lung (n = 3), corpus uteri (n = 2), and ovary (n = 1); 1 patient (3%) was treated with carboplatin and paclitaxel, then switched to carboplatin, bevacizumab, and paclitaxel, and then completed treatment with carboplatin and paclitaxel for an unspecified female genital cancer; and 1 patient (3%) was treated with cisplatin, ifosfamide, and paclitaxel for metastasis of the lower lobe lung and kidney cancer.
The 14 patients with PDTS data had 18 cancer drugs dispensed. Eleven had moderate interaction reports and 7 had no interaction reports. A total of 165 noncancer drugs were dispensed, of which 3 were antidepressants and had no interactions reported, 8 had moderate interactions reported, and 2 had minor interactions with Taxol and Abraxane, respectively (Table 6).86-129
Of 3 patients who were dispensed bupropion, nortriptyline, or paroxetine, 1 patient with breast cancer was treated with doxorubicin andcyclophosphamide, followed by paclitaxel with bupropion, nortriptyline, pegfilgrastim,dexamethasone, and 17 other noncancer drugs that had no interaction report dispensed during paclitaxel treatment. Of 2 patients with lung cancer, 1 patient was treated with carboplatin and paclitaxel with nortriptyline, dexamethasone, and 13 additional medications, and the second patient was treated with paroxetine, cimetidine, dexamethasone, and 12 other medications. Patients were dispensed up to6 noncancer medications on the same day as paclitaxel administration to control the AEs, not including the prodrugs filled before the treatments. Paroxetine and cimetidine have weak inhibition, and dexamethasone has weak induction of CYP3A4. Therefore, while 1:1 DDIs might have little or no effect with weak inhibit/induce CYP3A4 drugs, 1:1:1 or more combinations could have a different outcome (confirmed in previous publications).65-67
Dispensed on the same day may not mean taken at the same time. One patient experienced an AE with dispensed 50 mg losartan, carboplatin plus paclitaxel, dexamethasone, and 6 other noncancer drugs. Losartan inhibits paclitaxel, which can lead to negative AEs.57,66,67 However, there were no blood or plasma samples taken to confirm the losartan was taken at the same time as the paclitaxel given this was not a clinical trial.
Conclusions
This retrospective study discusses the use of paclitaxel in the MHS and the potential DDIs associated with it. The study population consisted mostly of active-duty personnel, who are required to be healthy or have controlled or nonactive medical diagnoses and be physically fit. This group is mixed with dependents and retirees that are more reflective of the average US population. As a result, this patient population is healthier than the general population, with a lower prevalence of common illnesses such as diabetes and obesity. The study aimed to identify drugs used alongside paclitaxel treatment. While further research is needed to identify potential DDIs among patients who experienced AEs, in vitro testing will need to be conducted before confirming causality. The low number of AEs experienced by only 32 of 702 patients (5%), with no deaths during paclitaxel treatment, indicates that the drug is generally well tolerated. Although this study cannot conclude that concomitant use with noncancer drugs led to the discontinuation of paclitaxel, we can conclude that there seems to be no significant DDIsidentified between paclitaxel and antidepressants. This comprehensive overview provides clinicians with a complete picture of paclitaxel use for 27 years (1996-2022), enabling them to make informed decisions about paclitaxel treatment.
Acknowledgments
The Department of Research Program funds at Walter Reed National Military Medical Center supported this protocol. We sincerely appreciate the contribution of data extraction from the Joint Pathology Center teams (Francisco J. Rentas, John D. McGeeney, Beatriz A. Hallo, and Johnny P. Beason) and the MHS database personnel (Maj Ryan Costantino, Brandon E. Jenkins, and Alexander G. Rittel). We gratefully thank you for the protocol support from the Department of Research programs: CDR Martin L. Boese, CDR Wesley R. Campbell, Maj. Abhimanyu Chandel, CDR Ling Ye, Chelsea N. Powers, Yaling Zhou, Elizabeth Schafer, Micah Stretch, Diane Beaner, and Adrienne Woodard.
1. American Chemical Society. Discovery of camptothecin and taxol. acs.org. Accessed June 4, 2024. https://www.acs.org/education/whatischemistry/landmarks/camptothecintaxol.html
2. Bocci G, Di Paolo A, Danesi R. The pharmacological bases of the antiangiogenic activity of paclitaxel. Angiogenesis. 2013;16(3):481-492. doi:10.1007/s10456-013-9334-0.
3. Meštrovic T. Paclitaxel history. News Medical Life Sciences. Updated March 11, 2023. Accessed June 4, 2024. https://www.news-medical.net/health/Paclitaxel-History.aspx
4. Rowinsky EK, Donehower RC. Paclitaxel (taxol). N Engl J Med. 1995;332(15):1004-1014. doi:10.1056/NEJM199504133321507
5. Walsh V, Goodman J. The billion dollar molecule: Taxol in historical and theoretical perspective. Clio Med. 2002;66:245-267. doi:10.1163/9789004333499_013
6. Perdue RE, Jr, Hartwell JL. The search for plant sources of anticancer drugs. Morris Arboretum Bull. 1969;20:35-53.
7. Wall ME, Wani MC. Camptothecin and taxol: discovery to clinic—thirteenth Bruce F. Cain Memorial Award lecture. Cancer Res. 1995;55:753-760.
8. Wani MC, Taylor HL, Wall ME, Coggon P, McPhail AT. Plant antitumor agents. VI. The isolation and structure of taxol, a novel antileukemic and antitumor agent from taxus brevifolia. J Am Chem Soc. 1971;93(9):2325-2327. doi:10.1021/ja00738a045
9. Weaver BA. How taxol/paclitaxel kills cancer cells. Mol Biol Cell. 2014;25(18):2677-2681. doi:10.1091/mbc.E14-04-0916
10. Chen JG, Horwitz SB. Differential mitotic responses to microtubule-stabilizing and-destabilizing drugs. Cancer Res. 2002;62(7):1935-1938.
11. Singh S, Dash AK. Paclitaxel in cancer treatment: perspectives and prospects of its delivery challenges. Crit Rev Ther Drug Carrier Syst. 2009;26(4):333-372. doi:10.1615/critrevtherdrugcarriersyst.v26.i4.10
12. Schiff PB, Fant J, Horwitz SB. Promotion of microtubule assembly in vitro by taxol. Nature. 1979;277(5698):665-667. doi:10.1038/277665a0
13. Fuchs DA, Johnson RK. Cytologic evidence that taxol, an antineoplastic agent from taxus brevifolia, acts as a mitotic spindle poison. Cancer Treat Rep. 1978;62(8):1219-1222.
14. Walsh V, Goodman J. From taxol to taxol: the changing identities and ownership of an anti-cancer drug. Med Anthropol. 2002;21(3-4):307-336. doi:10.1080/01459740214074
15. Walsh V, Goodman J. Cancer chemotherapy, biodiversity, public and private property: the case of the anti-cancer drug taxol. Soc Sci Med. 1999;49(9):1215-1225. doi:10.1016/s0277-9536(99)00161-6
16. Jordan MA, Wendell K, Gardiner S, Derry WB, Copp H, Wilson L. Mitotic block induced in HeLa cells by low concentrations of paclitaxel (taxol) results in abnormal mitotic exit and apoptotic cell death. Cancer Res. 1996;56(4):816-825.
17. Picard M, Castells MC. Re-visiting hypersensitivity reactions to taxanes: a comprehensive review. Clin Rev Allergy Immunol. 2015;49(2):177-191. doi:10.1007/s12016-014-8416-0
18. Zasadil LM, Andersen KA, Yeum D, et al. Cytotoxicity of paclitaxel in breast cancer is due to chromosome missegregation on multipolar spindles. Sci Transl Med. 2014;6:229ra243. doi:10.1126/scitranslmed.3007965
19. National Cancer Institute. Carboplatin-Taxol. Published May 30, 2012. Updated March 22, 2023. Accessed June 4, 2024. https://www.cancer.gov/about-cancer/treatment/drugs/carboplatin-taxol
20. Taxol (paclitaxel). Prescribing information. Bristol-Myers Squibb; 2011. Accessed June 4, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2011/020262s049lbl.pdf
21. Abraxane (paclitaxel). Prescribing information. Celgene Corporation; 2021. Accessed June 4, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2020/021660s047lbl.pdf
22. Awosika AO, Farrar MC, Jacobs TF. Paclitaxel. StatPearls. Updated November 18, 2023. Accessed June 5, 2024. https://www.ncbi.nlm.nih.gov/books/NBK536917/
23. Gerriets V, Kasi A. Bevacizumab. StatPearls. Updated September 1, 2022. Accessed June 5, 2024. https://www.ncbi.nlm.nih.gov/books/NBK482126/
24. American Cancer Society. Chemotherapy for endometrial cancer. Updated March 27, 2019. Accessed June 4, 2024. https://www.cancer.org/cancer/types/endometrial-cancer/treating/chemotherapy.html
25. US Food and Drug Administration. FDA approves pembrolizumab in combination with chemotherapy for first-line treatment of metastatic squamous NSCLC. October 30, 2018. Updated December 14, 2018. Accessed June 4, 2024. https://www.fda.gov/drugs/fda-approves-pembrolizumab-combination-chemotherapy-first-line-treatment-metastatic-squamous-nsclc
26. US Food and Drug Administration. FDA grants accelerated approval to pembrolizumab for locally recurrent unresectable or metastatic triple negative breast cancer. November 13, 2020. Accessed June 4, 2024. https://www.fda.gov/drugs/resources-information-approved-drugs/fda-grants-accelerated-approval-pembrolizumab-locally-recurrent-unresectable-or-metastatic-triple
27. US Food and Drug Administration. FDA approves atezolizumab for PD-L1 positive unresectable locally advanced or metastatic triple-negative breast. March 8, 2019. Updated March 18, 2019. Accessed June 5, 2024. https://www.fda.gov/drugs/drug-approvals-and-databases/fda-approves-atezolizumab-pd-l1-positive-unresectable-locally-advanced-or-metastatic-triple-negative
28. US Food and Drug Administration. FDA issues alert about efficacy and potential safety concerns with atezolizumab in combination with paclitaxel for treatment of breast cancer. September 8, 2020. Accessed June 5, 2024. https://www.fda.gov/drugs/resources-information-approved-drugs/fda-issues-alert-about-efficacy-and-potential-safety-concerns-atezolizumab-combination-paclitaxel
29. Tan AR. Chemoimmunotherapy: still the standard of care for metastatic triple-negative breast cancer. ASCO Daily News. February 23, 2022. Accessed June 5, 2024. https://dailynews.ascopubs.org/do/chemoimmunotherapy-still-standard-care-metastatic-triple-negative-breast-cancer
30. McGuire WP, Rowinsky EK, Rosenshein NB, et al. Taxol: a unique antineoplastic agent with significant activity in advanced ovarian epithelial neoplasms. Ann Intern Med. 1989;111(4):273-279. doi:10.7326/0003-4819-111-4-273
31. Milas L, Hunter NR, Kurdoglu B, et al. Kinetics of mitotic arrest and apoptosis in murine mammary and ovarian tumors treated with taxol. Cancer Chemother Pharmacol. 1995;35(4):297-303. doi:10.1007/BF00689448
32. Searle J, Collins DJ, Harmon B, Kerr JF. The spontaneous occurrence of apoptosis in squamous carcinomas of the uterine cervix. Pathology. 1973;5(2):163-169. doi:10.3109/00313027309060831
33. Gallego-Jara J, Lozano-Terol G, Sola-Martínez RA, Cánovas-Díaz M, de Diego Puente T. A compressive review about taxol®: history and future challenges. Molecules. 2020;25(24):5986. doi:10.3390/molecules25245986
34. Bernabeu E, Cagel M, Lagomarsino E, Moretton M, Chiappetta DA. Paclitaxel: What has been done and the challenges remain ahead. Int J Pharm. 2017;526(1-2):474-495. doi:10.1016/j.ijpharm.2017.05.016
35. Nehate C, Jain S, Saneja A, et al. Paclitaxel formulations: challenges and novel delivery options. Curr Drug Deliv. 2014;11(6):666-686. doi:10.2174/1567201811666140609154949
36. Gelderblom H, Verweij J, Nooter K, Sparreboom A, Cremophor EL. The drawbacks and advantages of vehicle selection for drug formulation. Eur J Cancer. 2001;37(13):1590-1598. doi:10.1016/S0959-8049(01)00171-x
37. Chowdhury MR, Moshikur RM, Wakabayashi R, et al. In vivo biocompatibility, pharmacokinetics, antitumor efficacy, and hypersensitivity evaluation of ionic liquid-mediated paclitaxel formulations. Int J Pharm. 2019;565:219-226. doi:10.1016/j.ijpharm.2019.05.020
38. Borgå O, Henriksson R, Bjermo H, Lilienberg E, Heldring N, Loman N. Maximum tolerated dose and pharmacokinetics of paclitaxel micellar in patients with recurrent malignant solid tumours: a dose-escalation study. Adv Ther. 2019;36(5):1150-1163. doi:10.1007/s12325-019-00909-6
39. Rouzier R, Rajan R, Wagner P, et al. Microtubule-associated protein tau: a marker of paclitaxel sensitivity in breast cancer. Proc Natl Acad Sci USA. 2005;102(23):8315-8320. doi:10.1073/pnas.0408974102
40. Choudhury H, Gorain B, Tekade RK, Pandey M, Karmakar S, Pal TK. Safety against nephrotoxicity in paclitaxel treatment: oral nanocarrier as an effective tool in preclinical evaluation with marked in vivo antitumor activity. Regul Toxicol Pharmacol. 2017;91:179-189. doi:10.1016/j.yrtph.2017.10.023
41. Barkat MA, Beg S, Pottoo FH, Ahmad FJ. Nanopaclitaxel therapy: an evidence based review on the battle for next-generation formulation challenges. Nanomedicine (Lond). 2019;14(10):1323-1341. doi:10.2217/nnm-2018-0313
42. Sofias AM, Dunne M, Storm G, Allen C. The battle of “nano” paclitaxel. Adv Drug Deliv Rev. 2017;122:20-30. doi:10.1016/j.addr.2017.02.003
43. Yang N, Wang C, Wang J, et al. Aurora inase a stabilizes FOXM1 to enhance paclitaxel resistance in triple-negative breast cancer. J Cell Mol Med. 2019;23(9):6442-6453. doi:10.1111/jcmm.14538
44. Chowdhury MR, Moshikur RM, Wakabayashi R, et al. Ionic-liquid-based paclitaxel preparation: a new potential formulation for cancer treatment. Mol Pharm. 2018;15(16):2484-2488. doi:10.1021/acs.molpharmaceut.8b00305
45. Chung HJ, Kim HJ, Hong ST. Tumor-specific delivery of a paclitaxel-loading HSA-haemin nanoparticle for cancer treatment. Nanomedicine. 2020;23:102089. doi:10.1016/j.nano.2019.102089
46. Ye L, He J, Hu Z, et al. Antitumor effect and toxicity of lipusu in rat ovarian cancer xenografts. Food Chem Toxicol. 2013;52:200-206. doi:10.1016/j.fct.2012.11.004
47. Ma WW, Lam ET, Dy GK, et al. A pharmacokinetic and dose-escalating study of paclitaxel injection concentrate for nano-dispersion (PICN) alone and with arboplatin in patients with advanced solid tumors. J Clin Oncol. 2013;31:2557. doi:10.1200/jco.2013.31.15_suppl.2557
48. Micha JP, Goldstein BH, Birk CL, Rettenmaier MA, Brown JV. Abraxane in the treatment of ovarian cancer: the absence of hypersensitivity reactions. Gynecol Oncol. 2006;100(2):437-438. doi:10.1016/j.ygyno.2005.09.012
49. Ingle SG, Pai RV, Monpara JD, Vavia PR. Liposils: an effective strategy for stabilizing paclitaxel loaded liposomes by surface coating with silica. Eur J Pharm Sci. 2018;122:51-63. doi:10.1016/j.ejps.2018.06.025
50. Abriata JP, Turatti RC, Luiz MT, et al. Development, characterization and biological in vitro assays of paclitaxel-loaded PCL polymeric nanoparticles. Mater Sci Eng C Mater Biol Appl. 2019;96:347-355. doi:10.1016/j.msec.2018.11.035
51. Hu J, Fu S, Peng Q, et al. Paclitaxel-loaded polymeric nanoparticles combined with chronomodulated chemotherapy on lung cancer: in vitro and in vivo evaluation. Int J Pharm. 2017;516(1-2):313-322. doi:10.1016/j.ijpharm.2016.11.047
52. Dranitsaris G, Yu B, Wang L, et al. Abraxane® vs Taxol® for patients with advanced breast cancer: a prospective time and motion analysis from a chinese health care perspective. J Oncol Pharm Pract. 2016;22(2):205-211. doi:10.1177/1078155214556008
53. Pei Q, Hu X, Liu S, Li Y, Xie Z, Jing X. Paclitaxel dimers assembling nanomedicines for treatment of cervix carcinoma. J Control Release. 2017;254:23-33. doi:10.1016/j.jconrel.2017.03.391
54. Wang Y, Wang M, Qi H, et al. Pathway-dependent inhibition of paclitaxel hydroxylation by kinase inhibitors and assessment of drug-drug interaction potentials. Drug Metab Dispos. 2014;42(4):782-795. doi:10.1124/dmd.113.053793
55. Shen F, Jiang G, Philips S, et al. Cytochrome P450 oxidoreductase (POR) associated with severe paclitaxel-induced peripheral neuropathy in patients of european ancestry from ECOG-ACRIN E5103. Clin Cancer Res. 2023;29(13):2494-2500. doi:10.1158/1078-0432.CCR-22-2431
56. Henningsson A, Marsh S, Loos WJ, et al. Association of CYP2C8, CYP3A4, CYP3A5, and ABCB1 polymorphisms with the pharmacokinetics of paclitaxel. Clin Cancer Res. 2005;11(22):8097-8104. doi:10.1158/1078-0432.CCR-05-1152
57. Mukai Y, Senda A, Toda T, et al. Drug-drug interaction between losartan and paclitaxel in human liver microsomes with different CYP2C8 genotypes. Basic Clin Pharmacol Toxicol. 2015;116(6):493-498. doi:10.1111/bcpt.12355
58. Kawahara B, Faull KF, Janzen C, Mascharak PK. Carbon monoxide inhibits cytochrome P450 enzymes CYP3A4/2C8 in human breast cancer cells, increasing sensitivity to paclitaxel. J Med Chem. 2021;64(12):8437-8446. doi:10.1021/acs.jmedchem.1c00404
59. Cresteil T, Monsarrat B, Dubois J, Sonnier M, Alvinerie P, Gueritte F. Regioselective metabolism of taxoids by human CYP3A4 and 2C8: structure-activity relationship. Drug Metab Dispos. 2002;30(4):438-445. doi:10.1124/dmd.30.4.438
60. Taniguchi R, Kumai T, Matsumoto N, et al. Utilization of human liver microsomes to explain individual differences in paclitaxel metabolism by CYP2C8 and CYP3A4. J Pharmacol Sci. 2005;97(1):83-90. doi:10.1254/jphs.fp0040603
61. Nakayama A, Tsuchiya K, Xu L, Matsumoto T, Makino T. Drug-interaction between paclitaxel and goshajinkigan extract and its constituents. J Nat Med. 2022;76(1):59-67. doi:10.1007/s11418-021-01552-8
62. Monsarrat B, Chatelut E, Royer I, et al. Modification of paclitaxel metabolism in a cancer patient by induction of cytochrome P450 3A4. Drug Metab Dispos. 1998;26(3):229-233.
63. Walle T. Assays of CYP2C8- and CYP3A4-mediated metabolism of taxol in vivo and in vitro. Methods Enzymol. 1996;272:145-151. doi:10.1016/s0076-6879(96)72018-9
64. Hanioka N, Matsumoto K, Saito Y, Narimatsu S. Functional characterization of CYP2C8.13 and CYP2C8.14: catalytic activities toward paclitaxel. Basic Clin Pharmacol Toxicol. 2010;107(1):565-569. doi:10.1111/j.1742-7843.2010.00543.x
65. Luong TT, Powers CN, Reinhardt BJ, Weina PJ. Pre-clinical drug-drug interactions (DDIs) of gefitinib with/without losartan and selective serotonin reuptake inhibitors (SSRIs): citalopram, fluoxetine, fluvoxamine, paroxetine, sertraline, and venlafaxine. Curr Res Pharmacol Drug Discov. 2022;3:100112. doi:10.1016/j.crphar.2022.100112
66. Luong TT, McAnulty MJ, Evers DL, Reinhardt BJ, Weina PJ. Pre-clinical drug-drug interaction (DDI) of gefitinib or erlotinib with Cytochrome P450 (CYP) inhibiting drugs, fluoxetine and/or losartan. Curr Res Toxicol. 2021;2:217-224. doi:10.1016/j.crtox.2021.05.006
67. Luong TT, Powers CN, Reinhardt BJ, et al. Retrospective evaluation of drug-drug interactions with erlotinib and gefitinib use in the military health system. Fed Pract. 2023;40(suppl 3):S24-S34. doi:10.12788/fp.0401
68. Adamo M, Dickie L, Ruhl J. SEER program coding and staging manual 2016. National Cancer Institute. Accessed June 5, 2024. https://seer.cancer.gov/archive/manuals/2016/SPCSM_2016_maindoc.pdf
69. World Health Organization. International classification of diseases for oncology (ICD-O) 3rd ed, 1st revision. World Health Organization; 2013. Accessed June 5, 2024. https://apps.who.int/iris/handle/10665/96612
70. Z score calculator for 2 population proportions. Social science statistics. Accessed June 5, 2024. https://www.socscistatistics.com/tests/ztest/default2.aspx
71. US Food and Drug Administration. Generic drugs: question & answers. FDA.gov. Accessed June 5, 2024. https://www.fda.gov/drugs/frequently-asked-questions-popular-topics/generic-drugs-questions-answers
72. Oura M, Saito H, Nishikawa Y. Shortage of nab-paclitaxel in Japan and around the world: issues in global information sharing. JMA J. 2023;6(2):192-195. doi:10.31662/jmaj.2022-0179
73. Yuan H, Guo H, Luan X, et al. Albumin nanoparticle of paclitaxel (abraxane) decreases while taxol increases breast cancer stem cells in treatment of triple negative breast cancer. Mol Pharm. 2020;17(7):2275-2286. doi:10.1021/acs.molpharmaceut.9b01221
74. Dranitsaris G, Yu B, Wang L, et al. Abraxane® versus Taxol® for patients with advanced breast cancer: a prospective time and motion analysis from a Chinese health care perspective. J Oncol Pharm Pract. 2016;22(2):205-211. doi:10.1177/1078155214556008
75. Gradishar WJ, Tjulandin S, Davidson N, et al. Phase III trial of nanoparticle albumin-bound paclitaxel compared with polyethylated castor oil-based paclitaxel in women with breast cancer. J Clin Oncol. 2005;23(31):7794-7803. doi:10.1200/JCO.2005.04.
76. Liu M, Liu S, Yang L, Wang S. Comparison between nab-paclitaxel and solvent-based taxanes as neoadjuvant therapy in breast cancer: a systematic review and meta-analysis. BMC Cancer. 2021;21(1):118. doi:10.1186/s12885-021-07831-7
77. Rowinsky EK, Eisenhauer EA, Chaudhry V, Arbuck SG, Donehower RC. Clinical toxicities encountered with paclitaxel (taxol). Semin Oncol. 1993;20(4 Suppl 3):1-15.
78. Banerji A, Lax T, Guyer A, Hurwitz S, Camargo CA Jr, Long AA. Management of hypersensitivity reactions to carboplatin and paclitaxel in an outpatient oncology infusion center: a 5-year review. J Allergy Clin Immunol Pract. 2014;2(4):428-433. doi:10.1016/j.jaip.2014.04.010
79. Staff NP, Fehrenbacher JC, Caillaud M, Damaj MI, Segal RA, Rieger S. Pathogenesis of paclitaxel-induced peripheral neuropathy: a current review of in vitro and in vivo findings using rodent and human model systems. Exp Neurol. 2020;324:113121. doi:10.1016/j.expneurol.2019.113121
80. Postma TJ, Vermorken JB, Liefting AJ, Pinedo HM, Heimans JJ. Paclitaxel-induced neuropathy. Ann Oncol. 1995;6(5):489-494. doi:10.1093/oxfordjournals.annonc.a059220
81. Liu JM, Chen YM, Chao Y, et al. Paclitaxel-induced severe neuropathy in patients with previous radiotherapy to the head and neck region. J Natl Cancer Inst. 1996;88(14):1000-1002. doi:10.1093/jnci/88.14.1000-a
82. Bayat Mokhtari R, Homayouni TS, Baluch N, et al. Combination therapy in combating cancer. Oncotarget. 2017;8(23):38022-38043. doi:10.18632/oncotarget.16723
83. Blagosklonny MV. Analysis of FDA approved anticancer drugs reveals the future of cancer therapy. Cell Cycle. 2004;3(8):1035-1042.
84. Yap TA, Omlin A, de Bono JS. Development of therapeutic combinations targeting major cancer signaling pathways. J Clin Oncol. 2013;31(12):1592-1605. doi:10.1200/JCO.2011.37.6418
85. Gilani B, Cassagnol M. Biochemistry, Cytochrome P450. StatPearls. Updated April 24, 2023. Accessed June 5, 2024. https://www.ncbi.nlm.nih.gov/books/NBK557698/
86. LiverTox: clinical and research information on drug-induced liver injury; 2012. Carboplatin. Updated September 15, 2020. Accessed June 5, 2024. https://www.ncbi.nlm.nih.gov/books/NBK548565/
87. Carboplatin. Prescribing information. Teva Parenteral Medicines; 2012. Accessed June 5, 204. https://www.accessdata.fda.gov/drugsatfda_docs/label/2012/077139Orig1s016lbl.pdf
88. Johnson-Arbor K, Dubey R. Doxorubicin. StatPearls. Updated August 8, 2023. Accessed June 5, 2024. https://www.ncbi.nlm.nih.gov/books/NBK459232/
89. Doxorubicin hydrochloride injection. Prescribing information. Pfizer; 2019. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2020/050467s078,050629s030lbl.pdf
90. Gor, PP, Su, HI, Gray, RJ, et al. Cyclophosphamide-metabolizing enzyme polymorphisms and survival outcomes after adjuvant chemotherapy for node-positive breast cancer: a retrospective cohort study. Breast Cancer Res. 2010;12(3):R26. doi:10.1186/bcr2570
91. Cyclophosphamide. Prescribing information. Ingenus Pharmaceuticals; 2020. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2020/212501s000lbl.pdf
92. Gemcitabine. Prescribing information. Hospira; 2019. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2019/200795Orig1s010lbl.pdf
93. Ifex (ifosfamide). Prescribing information. Baxter; 2012. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2012/019763s017lbl.pdf
94. Cisplatin. Prescribing information. WG Critical Care; 2019. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2019/018057s089lbl.pdf
95. Gerriets V, Kasi A. Bevacizumab. StatPearls. Updated August 28, 2023. Accessed June 5, 2024. https://www.ncbi.nlm.nih.gov/books/NBK482126/
96. Avastin (bevacizumab). Prescribing information. Genentech; 2022. Accessed June 5, 2024. https://www.accessdata .fda.gov/drugsatfda_docs/label/2022/125085s340lbl.pdf
 97. Keytruda (pembrolizumab). Prescribing information. Merck; 2021. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2021/125514s096lbl.pdf
97. Keytruda (pembrolizumab). Prescribing information. Merck; 2021. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2021/125514s096lbl.pdf
98. Dean L, Kane M. Capecitabine therapy and DPYD genotype. National Center for Biotechnology Information (US); 2012. Updated November 2, 2020. Accessed June 5, 2024. https://www.ncbi.nlm.nih.gov/books/NBK385155/
99. Xeloda (capecitabine). Prescribing information. Roche; 2000. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2000/20896lbl.pdf
100. Pemetrexed injection. Prescribing information. Fareva Unterach; 2022. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2022/214657s000lbl.pdf
101. Topotecan Injection. Prescribing information. Zydus Hospira Oncology; 2014. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2014/200582s001lbl.pdf
102. Ibrance (palbociclib). Prescribing information. Pfizer; 2019. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2019/207103s008lbl.pdf
103. Navelbine (vinorelbine) injection. Prescribing information. Pierre Fabre Médicament; 2020. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2020/020388s037lbl.pdf
104. LiverTox: clinical and research information on drug-induced liver injury; 2012. Letrozole. Updated July 25, 2017. Accessed June 5, 2024. https://www.ncbi.nlm.nih.gov/books/NBK548381/
105. Femara (letrozole). Prescribing information. Novartis; 2014. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2014/020726s027lbl.pdf
106. Soltamox (tamoxifen citrate). Prescribing information. Rosemont Pharmaceuticals; 2018. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2018/021807s005lbl.pdf
107. LiverTox: clinical and research information on drug-induced liver injury; 2012. Anastrozole. Updated July 25, 2017. Accessed June 5, 2024. https://www.ncbi.nlm.nih.gov/books/NBK548189/
108. Grimm SW, Dyroff MC. Inhibition of human drug metabolizing cytochromes P450 by anastrozole, a potent and selective inhibitor of aromatase. Drug Metab Dispos. 1997;25(5):598-602.
109. Arimidex (anastrozole). Prescribing information. AstraZeneca; 2010. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2011/020541s026lbl.pdf
110. Megace (megestrol acetate). Prescribing information. Endo Pharmaceuticals; 2018. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2018/021778s024lbl.pdf
111. Imfinzi (durvalumab). Prescribing information. AstraZeneca; 2020. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2020/761069s018lbl.pdf
112. Merwar G, Gibbons JR, Hosseini SA, et al. Nortriptyline. StatPearls. Updated June 5, 2023. Accessed June 5, 2024. https://www.ncbi.nlm.nih.gov/books/NBK482214/
113. Pamelor (nortriptyline HCl). Prescribing information. Patheon Inc.; 2012. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2012/018012s029,018013s061lbl.pdf
114. Wellbutrin (bupropion hydrochloride). Prescribing information. GlaxoSmithKline; 2017. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2017/018644s052lbl.pdf
115. Paxil (paroxetine). Prescribing information. Apotex Inc.; 2021. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2021/020031s077lbl.pdf
116. Johnson DB, Lopez MJ, Kelley B. Dexamethasone. StatPearls. Updated May 2, 2023. Accessed June 5, 2024. https://www.ncbi.nlm.nih.gov/books/NBK482130/
117. Hemady (dexamethasone). Prescribing information. Dexcel Pharma; 2019. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2019/211379s000lbl.pdf
118. Parker SD, King N, Jacobs TF. Pegfilgrastim. StatPearls. Updated May 9, 2024. Accessed June 5, 2024. https://www.ncbi.nlm.nih.gov/books/NBK532893/
119. Fylnetra (pegfilgrastim-pbbk). Prescribing information. Kashiv BioSciences; 2022. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2022/761084s000lbl.pdf
120. Emend (aprepitant). Prescribing information. Merck; 2015. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2015/207865lbl.pdf
121. Lipitor (atorvastatin calcium). Prescribing information. Viatris Specialty; 2022. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2022/020702Orig1s079correctedlbl.pdf
122. Cipro (ciprofloxacin hydrochloride). Prescribing information. Bayer HealthCare Pharmaceuticals Inc.; 2020. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2020/019537s090,020780s047lbl.pdf
123. Pino MA, Azer SA. Cimetidine. StatPearls. Updated March 6, 2023. Accessed June 5, 2024. https://www.ncbi.nlm.nih.gov/books/NBK544255/
124. Tagament (Cimetidine). Prescribing information. Mylan; 2020. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2020/020238Orig1s024lbl.pdf
125. Neupogen (filgrastim). Prescribing information. Amgen Inc.; 2015. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2015/103353s5184lbl.pdf
126. Flagyl (metronidazole). Prescribing information. Pfizer; 2013. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2013/020334s008lbl.pdf
127. Zymaxid (gatifloxacin ophthalmic solution). Prescribing information. Allergan; 2016. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2016/022548s002lbl.pdf
128. Macrobid (nitrofurantoin monohydrate). Prescribing information. Procter and Gamble Pharmaceutical Inc.; 2009. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2009/020064s019lbl.pdf
129. Hyzaar (losartan). Prescribing information. Merck; 2020. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2020/020387s067lbl.pdf
Background
Paclitaxel was first derived from the bark of the yew tree (Taxus brevifolia). It was discovered as part of a National Cancer Institute program screen of plants and natural products with putative anticancer activity during the 1960s.1-9 Paclitaxel works by suppressing spindle microtube dynamics, which results in the blockage of the metaphase-anaphase transitions, inhibition of mitosis, and induction of apoptosis in a broad spectrum of cancer cells. Paclitaxel also displayed additional anticancer activities, including the suppression of cell proliferation and antiangiogenic effects. However, since the growth of normal body cells may also be affected, other adverse effects (AEs) will also occur.8-18
Two different chemotherapy drugs contain paclitaxel—paclitaxel and nab-paclitaxel—and the US Food and Drug Administration (FDA) recognizes them as separate entities.19-21 Taxol (paclitaxel) was approved by the FDA in 1992 for treating advanced ovarian cancer.20 It has since been approved for the treatment of metastatic breast cancer, AIDS-related Kaposi sarcoma (as an orphan drug), non-small cell lung cancer (NSCLC), and cervical cancers (in combination withbevacizumab) in 1994, 1997, 1999, and 2014, respectively.21 Since 2002, a generic version of Taxol, known as paclitaxel injectable, has been FDA-approved from different manufacturers. According to the National Cancer Institute, a combination of carboplatin and Taxol is approved to treat carcinoma of unknown primary, cervical, endometrial, NSCLC, ovarian, and thymoma cancers.19 Abraxane (nab-paclitaxel) was FDA-approved to treat metastatic breast cancer in 2005. It was later approved for first-line treatment of advanced NSCLC and late-stage pancreatic cancer in 2012 and 2013, respectively. In 2018 and 2020, both Taxol and Abraxane were approved for first-line treatment of metastatic squamous cell NSCLC in combination with carboplatin and pembrolizumab and metastatic triple-negative breast cancer in combination with pembrolizumab, respectively.22-26 In 2019, Abraxane was approved with atezolizumab to treat metastatic triple-negative breast cancer, but this approval was withdrawn in 2021. In 2022, a generic version of Abraxane, known as paclitaxel protein-bound, was released in the United States. Furthermore, paclitaxel-containing formulations also are being studied in the treatment of other types of cancer.19-32
One of the main limitations of paclitaxel is its low solubility in water, which complicates its drug supply. To distribute this hydrophobic anticancer drug efficiently, paclitaxel is formulated and administered to patients via polyethoxylated castor oil or albumin-bound (nab-paclitaxel). However, polyethoxylated castor oil induces complement activation and is the cause of common hypersensitivity reactions related to paclitaxel use.2,17,33-38 Therefore, many alternatives to polyethoxylated castor oil have been researched.
Since 2000, new paclitaxel formulations have emerged using nanomedicine techniques. The difference between these formulations is the drug vehicle. Different paclitaxel-based nanotechnological vehicles have been developed and approved, such as albumin-based nanoparticles, polymeric lipidic nanoparticles, polymeric micelles, and liposomes, with many others in clinical trial phases.3,37 Albumin-based nanoparticles have a high response rate (33%), whereas the response rate for polyethoxylated castor oil is 25% in patients with metastatic breast cancer.33,39-52 The use of paclitaxel dimer nanoparticles also has been proposed as a method for increasing drug solubility.33,53
Paclitaxel is metabolized by cytochrome P450 (CYP) isoenzymes 2C8 and 3A4. When administering paclitaxel with known inhibitors, inducers, or substrates of CYP2C8 or CYP3A4, caution is required.19-22 Regulations for CYP research were not issued until 2008, so potential interactions between paclitaxel and other drugs have not been extensively evaluated in clinical trials. A study of 12 kinase inhibitors showed strong inhibition of CYP2C8 and/or CYP3A4 pathways by these inhibitors, which could alter the ratio of paclitaxel metabolites in vivo, leading to clinically relevant changes.54 Differential metabolism has been linked to paclitaxel-induced neurotoxicity in patients with cancer.55 Nonetheless, variants in the CYP2C8, CYP3A4, CYP3A5, and ABCB1 genes do not account for significant interindividual variability in paclitaxel pharmacokinetics.56 In liver microsomes, losartan inhibited paclitaxel metabolism when used at concentrations > 50 µmol/L.57 Many drug-drug interaction (DDI) studies of CYP2C8 and CYP3A4 have shown similar results for paclitaxel.58-64
The goals of this study are to investigate prescribed drugs used with paclitaxel and determine patient outcomes through several Military Health System (MHS) databases. The investigation focused on (1) the functions of paclitaxel; (2) identifying AEs that patients experienced; (3) evaluating differences when paclitaxel is used alone vs concomitantly and between the completed vs discontinued treatment groups; (4) identifying all drugs used during paclitaxel treatment; and (5) evaluating DDIs with antidepressants (that have an FDA boxed warning and are known to have DDIs confirmed in previous publications) and other drugs.65-67
The Walter Reed National Military Medical Center in Bethesda, Maryland, institutionalreview board approved the study protocol and ensured compliance with the Health Insurance Portability and Accountability Act as an exempt protocol. The Joint Pathology Center (JPC) of the US Department of Defense (DoD) Cancer Registry Program and MHS data experts from the Comprehensive Ambulatory/Professional Encounter Record (CAPER) and the Pharmacy Data Transaction Service (PDTS) provided data for the analysis.
METHODS
The DoD Cancer Registry Program was established in 1986 and currently contains data from 1998 to 2024. CAPER and PDTS are part of the MHS Data Repository/Management Analysis and Reporting Tool database. Each observation in the CAPER record represents an ambulatory encounter at a military treatment facility (MTF). CAPER includes data from 2003 to 2024.
Each observation in the PDTS record represents a prescription filled for an MHS beneficiary at an MTF through the TRICARE mail-order program or a US retail pharmacy. Missing from this record are prescriptions filled at international civilian pharmacies and inpatient pharmacy prescriptions. The MHS Data Repository PDTS record is available from 2002 to 2024. The legacy Composite Health Care System is being replaced by GENESIS at MTFs.
Data Extraction Design
The study design involved a cross-sectional analysis. We requested data extraction for paclitaxel from 1998 to 2022. Data from the DoD Cancer Registry Program were used to identify patients who received cancer treatment. Once patients were identified, the CAPER database was searched for diagnoses to identify other health conditions, whereas the PDTS database was used to populate a list of prescription medications filled during chemotherapy treatment.
Data collected from the JPC included cancer treatment, cancer information, demographics, and physicians’ comments on AEs. Collected data from the MHS include diagnosis and filled prescription history from initiation to completion of the therapy period (or 2 years after the diagnosis date). For the analysis of the DoD Cancer Registry Program and CAPER databases, we used all collected data without excluding any. When analyzing PDTS data, we excluded patients with PDTS data but without a record of paclitaxel being filled, or medications filled outside the chemotherapy period (by evaluating the dispensed date and day of supply).
Data Extraction Analysis
The Surveillance, Epidemiology, and End Results Program Coding and Staging Manual 2016 and the International Classification of Diseases for Oncology, 3rd edition, 1st revision, were used to decode disease and cancer types.68,69 Data sorting and analysis were performed using Microsoft Excel. The percentage for the total was calculated by using the number of patients or data available within the paclitaxel groups divided by the total number of patients or data variables. The subgroup percentage was calculated by using the number of patients or data available within the subgroup divided by the total number of patients in that subgroup.
In alone vs concomitant and completed vs discontinued treatment groups, a 2-tailed, 2-sample z test was used to statistical significance (P < .05) using a statistics website.70 Concomitant was defined as paclitaxel taken with other antineoplastic agent(s) before, after, or at the same time as cancer therapy. For the retrospective data analysis, physicians’ notes with a period, comma, forward slash, semicolon, or space between medication names were interpreted as concurrent, whereas plus (+), minus/plus (-/+), or “and” between drug names that were dispensed on the same day were interpreted as combined with known common combinations: 2 drugs (DM886 paclitaxel and carboplatin and DM881-TC-1 paclitaxel and cisplatin) or 3 drugs (DM887-ACT doxorubicin, cyclophosphamide, and paclitaxel). Completed treatment was defined as paclitaxel as the last medication the patient took without recorded AEs; switching or experiencing AEs was defined as discontinued treatment.
RESULTS
The JPC provided 702 entries for 687 patients with a mean age of 56 years (range, 2 months to 88 years) who were treated with paclitaxel from March 1996 to October 2021. Fifteen patients had duplicate entries because they had multiple cancer sites or occurrences. There were 623 patients (89%) who received paclitaxel for FDA-approved indications. The most common types of cancer identified were 344 patients with breast cancer (49%), 91 patients with lung cancer (13%), 79 patients with ovarian cancer (11%), and 75 patients with endometrial cancer (11%) (Table 1). Seventy-nine patients (11%) received paclitaxel for cancers that were not for FDA-approved indications, including 19 for cancers of the fallopian tube (3%) and 17 for esophageal cancer (2%) (Table 2).
There were 477 patients (68%) aged > 50 years. A total of 304 patients (43%) had a stage III or IV cancer diagnosis and 398 (57%) had stage II or lower (combination of data for stages 0, I, and II; not applicable; and unknown) cancer diagnosis. For systemic treatment, 16 patients (2%) were treated with paclitaxel alone and 686 patients (98%) received paclitaxel concomitantly with additional chemotherapy: 59 patients (9%) in the before or after group, 410 patients (58%) had a 2-drug combination, 212 patients (30%) had a 3-drug combination, and 5 patients (1%) had a 4-drug combination. In addition, for doublet therapies, paclitaxel combined with carboplatin, trastuzumab, gemcitabine, or cisplatin had more patients (318, 58, 12, and 11, respectively) than other combinations (≤ 4 patients). For triplet therapies, paclitaxel combined withdoxorubicin plus cyclophosphamide or carboplatin plus bevacizumab had more patients (174 and 20, respectively) than other combinations, including quadruplet therapies (≤ 4 patients) (Table 3).
Patients were more likely to discontinue paclitaxel if they received concomitant treatment. None of the 16 patients receiving paclitaxel monotherapy experienced AEs, whereas 364 of 686 patients (53%) treated concomitantly discontinued (P < .001). Comparisons of 1 drug vs combination (2 to 4 drugs) and use for treating cancers that were FDA-approved indications vs off-label use were significant (P < .001), whereas comparisons of stage II or lower vs stage III and IV cancer and of those aged ≤ 50 years vs aged > 50 years were not significant (P = .50 andP = .30, respectively) (Table 4).
Among the 364 patients who had concomitant treatment and had discontinued their treatment, 332 (91%) switched treatments with no AEs documented and 32 (9%) experienced fatigue with pneumonia, mucositis, neuropathy, neurotoxicity, neutropenia, pneumonitis, allergic or hypersensitivity reaction, or an unknown AE. Patients who discontinued treatment because of unknown AEs had a physician’s note that detailed progressive disease, a significant decline in performance status, and another unknown adverse effect due to a previous sinus tract infection and infectious colitis (Table 5).
Management Analysis and Reporting Tool Database
MHS data analysts provided data on diagnoses for 639 patients among 687 submitteddiagnoses, with 294 patients completing and 345 discontinuing paclitaxel treatment. Patients in the completed treatment group had 3 to 258 unique health conditions documented, while patients in the discontinued treatment group had 4 to 181 unique health conditions documented. The MHS reported 3808 unique diagnosis conditions for the completed group and 3714 for the discontinued group (P = .02).
The mean (SD) number of diagnoses was 51 (31) for the completed and 55 (28) for the discontinued treatment groups (Figure). Among 639 patients who received paclitaxel, the top 5 diagnoses were administrative, including encounters for other administrative examinations; antineoplastic chemotherapy; administrative examination for unspecified; other specified counseling; and adjustment and management of vascular access device. The database does not differentiate between administrative and clinically significant diagnoses.
MHS data analysts provided data for 336 of 687 submitted patients who were prescribed paclitaxel; 46 patients had no PDTS data, and 305 patients had PDTS data without paclitaxel, Taxol, or Abraxane dispensed. Medications that were filled outside the chemotherapy period were removed by evaluating the dispensed date and day of supply. Among these 336 patients, 151 completed the treatment and 185 discontinued, with 14 patients experiencing documented AEs. Patients in the completed treatment group filled 9 to 56 prescriptions while patients in the discontinued treatment group filled 6 to 70 prescriptions.Patients in the discontinued group filled more prescriptions than those who completed treatment: 793 vs 591, respectively (P = .34).
The mean (SD) number of filled prescription drugs was 24 (9) for the completed and 34 (12) for the discontinued treatment group. The 5 most filled prescriptions with paclitaxel from 336 patients with PDTS data were dexamethasone (324 prescriptions with 14 recorded AEs), diphenhydramine (296 prescriptions with 12 recorded AEs), ondansetron (277 prescriptions with 11 recorded AEs), prochlorperazine (265 prescriptions with 12 recorded AEs), and sodium chloride (232 prescriptions with 11 recorded AEs).
DISCUSSION
As a retrospective review, this study is more limited in the strength of its conclusions when compared to randomized control trials. The DoD Cancer Registry Program only contains information about cancer types, stages, treatment regimens, and physicians’ notes. Therefore, noncancer drugs are based solely on the PDTS database. In most cases, physicians' notes on AEs were not detailed. There was no distinction between initial vs later lines of therapy and dosage reductions. The change in status or appearance of a new medical condition did not indicate whether paclitaxel caused the changes to develop or directly worsen a pre-existing condition. The PDTS records prescriptions filled, but that may not reflect patients taking prescriptions.
Paclitaxel
Paclitaxel has a long list of both approved and off-label uses in malignancies as a primary agent and in conjunction with other drugs. The FDA prescribing information for Taxol and Abraxane was last updated in April 2011 and September 2020, respectively.20,21 The National Institutes of Health National Library of Medicine has the current update for paclitaxel on July 2023.19,22 Thus, the prescribed information for paclitaxel referenced in the database may not always be up to date. The combinations of paclitaxel with bevacizumab, carboplatin, or carboplatin and pembrolizumab were not in the Taxol prescribing information. Likewise, a combination of nab-paclitaxel with atezolizumab or carboplatin and pembrolizumab is missing in the Abraxane prescribing information.22-27
The generic name is not the same as a generic drug, which may have slight differences from the brand name product.71 The generic drug versions of Taxol and Abraxane have been approved by the FDA as paclitaxel injectable and paclitaxel-protein bound, respectively. There was a global shortage of nab-paclitaxel from October 2021 to June 2022 because of a manufacturing problem.72 During this shortage, data showed similar comments from physician documents that treatment switched to Taxol due to the Abraxane shortage.
Of 336 patients in the PDTS database with dispensed paclitaxel prescriptions, 276 received paclitaxel (year dispensed, 2013-2022), 27 received Abraxane (year dispensed, 2013-2022), 47 received Taxol (year dispensed, 2004-2015), 8 received both Abraxane and paclitaxel, and 6 received both Taxol and paclitaxel. Based on this information, it appears that the distinction between the drugs was not made in the PDTS until after 2015, 10 years after Abraxane received FDA approval. Abraxane was prescribed in the MHS in 2013, 8 years after FDA approval. There were a few comparison studies of Abraxane and Taxol.73-76
Safety and effectiveness in pediatric patients have not been established for paclitaxel. According to the DoD Cancer Registry Program, the youngest patient was aged 2 months. In 2021, this patient was diagnosed with corpus uteri and treated with carboplatin and Taxol in course 1; in course 2, the patient reacted to Taxol; in course 3, Taxol was replaced with Abraxane; in courses 4 to 7, the patient was treated with carboplatin only.
Discontinued Treatment
Ten patients had prescribed Taxol that was changed due to AEs: 1 was switched to Abraxane and atezolizumab, 3 switched to Abraxane, 2 switched to docetaxel, 1 switched to doxorubicin, and 3 switched to pembrolizumab (based on physician’s comments). Of the 10 patients, 7 had Taxol reaction, 2 experienced disease progression, and 1 experienced high programmed death–ligand 1 expression (this patient with breast cancer was switched to Abraxane and atezolizumab during the accelerated FDA approval phase for atezolizumab, which was later revoked). Five patients were treated with carboplatin and Taxol for cancer of the anal canal (changed to pembrolizumab after disease progression), lung not otherwise specified (changed to carboplatin and pembrolizumab due to Taxol reaction), lower inner quadrant of the breast (changed to doxorubicin due to hypersensitivity reaction), corpus uteri (changed to Abraxane due to Taxol reaction), and ovary (changed to docetaxel due to Taxol reaction). Three patients were treated with doxorubicin, cyclophosphamide, and Taxol for breast cancer; 2 patients with breast cancer not otherwise specified switched to Abraxane due to cardiopulmonary hypersensitivity and Taxol reaction and 1 patient with cancer of the upper outer quadrant of the breast changed to docetaxel due to allergic reaction. One patient, who was treated with paclitaxel, ifosfamide, and cisplatin for metastasis of the lower lobe of the lung and kidney cancer, experienced complications due to infectious colitis (treated with ciprofloxacin) and then switched to pembrolizumab after the disease progressed. These AEs are known in paclitaxel medical literature on paclitaxel AEs.19-24,77-81
Combining 2 or more treatments to target cancer-inducing or cell-sustaining pathways is a cornerstone of chemotherapy.82-84 Most combinations are given on the same day, but some are not. For 3- or 4-drug combinations, doxorubicin and cyclophosphamide were given first, followed by paclitaxel with or withouttrastuzumab, carboplatin, or pembrolizumab. Only 16 patients (2%) were treated with paclitaxel alone; therefore, the completed and discontinued treatment groups are mostly concomitant treatment. As a result, the comparisons of the completed and discontinued treatment groups were almost the same for the diagnosis. The PDTS data have a better result because 2 exclusion criteria were applied before narrowing the analysis down to paclitaxel treatment specifically.
Antidepressants and Other Drugs
Drug response can vary from person to person and can lead to treatment failure related to AEs. One major factor in drug metabolism is CYP.85 CYP2C8 is the major pathway for paclitaxel and CYP3A4 is the minor pathway. When evaluating the noncancer drugs, there were no reports of CYP2C8 inhibition or induction. Over the years, many DDI warnings have been issued for paclitaxel with different drugs in various electronic resources.
Oncologists follow guidelines to prevent DDIs, as paclitaxel is known to have severe, moderate, and minor interactions with other drugs. Among 687 patients, 261 (38%) were prescribed any of 14 antidepressants. Eight of these antidepressants (amitriptyline, citalopram, desipramine, doxepin, venlafaxine, escitalopram, nortriptyline, and trazodone) are metabolized, 3 (mirtazapine, sertraline, and fluoxetine) are metabolized and inhibited, 2 (bupropion and duloxetine) are neither metabolized nor inhibited, and 1 (paroxetine) is inhibited by CYP3A4. Duloxetine, venlafaxine, and trazodone were more commonly dispensed (84, 78, and 42 patients, respectively) than others (≤ 33 patients).
Of 32 patients with documented AEs,14 (44%) had 168 dispensed drugs in the PDTS database. Six patients (19%) were treated with doxorubicin and cyclophosphamide followed by paclitaxel for breast cancer; 6 (19%) were treated with carboplatin and paclitaxel for cancer of the lung (n = 3), corpus uteri (n = 2), and ovary (n = 1); 1 patient (3%) was treated with carboplatin and paclitaxel, then switched to carboplatin, bevacizumab, and paclitaxel, and then completed treatment with carboplatin and paclitaxel for an unspecified female genital cancer; and 1 patient (3%) was treated with cisplatin, ifosfamide, and paclitaxel for metastasis of the lower lobe lung and kidney cancer.
The 14 patients with PDTS data had 18 cancer drugs dispensed. Eleven had moderate interaction reports and 7 had no interaction reports. A total of 165 noncancer drugs were dispensed, of which 3 were antidepressants and had no interactions reported, 8 had moderate interactions reported, and 2 had minor interactions with Taxol and Abraxane, respectively (Table 6).86-129
Of 3 patients who were dispensed bupropion, nortriptyline, or paroxetine, 1 patient with breast cancer was treated with doxorubicin andcyclophosphamide, followed by paclitaxel with bupropion, nortriptyline, pegfilgrastim,dexamethasone, and 17 other noncancer drugs that had no interaction report dispensed during paclitaxel treatment. Of 2 patients with lung cancer, 1 patient was treated with carboplatin and paclitaxel with nortriptyline, dexamethasone, and 13 additional medications, and the second patient was treated with paroxetine, cimetidine, dexamethasone, and 12 other medications. Patients were dispensed up to6 noncancer medications on the same day as paclitaxel administration to control the AEs, not including the prodrugs filled before the treatments. Paroxetine and cimetidine have weak inhibition, and dexamethasone has weak induction of CYP3A4. Therefore, while 1:1 DDIs might have little or no effect with weak inhibit/induce CYP3A4 drugs, 1:1:1 or more combinations could have a different outcome (confirmed in previous publications).65-67
Dispensed on the same day may not mean taken at the same time. One patient experienced an AE with dispensed 50 mg losartan, carboplatin plus paclitaxel, dexamethasone, and 6 other noncancer drugs. Losartan inhibits paclitaxel, which can lead to negative AEs.57,66,67 However, there were no blood or plasma samples taken to confirm the losartan was taken at the same time as the paclitaxel given this was not a clinical trial.
Conclusions
This retrospective study discusses the use of paclitaxel in the MHS and the potential DDIs associated with it. The study population consisted mostly of active-duty personnel, who are required to be healthy or have controlled or nonactive medical diagnoses and be physically fit. This group is mixed with dependents and retirees that are more reflective of the average US population. As a result, this patient population is healthier than the general population, with a lower prevalence of common illnesses such as diabetes and obesity. The study aimed to identify drugs used alongside paclitaxel treatment. While further research is needed to identify potential DDIs among patients who experienced AEs, in vitro testing will need to be conducted before confirming causality. The low number of AEs experienced by only 32 of 702 patients (5%), with no deaths during paclitaxel treatment, indicates that the drug is generally well tolerated. Although this study cannot conclude that concomitant use with noncancer drugs led to the discontinuation of paclitaxel, we can conclude that there seems to be no significant DDIsidentified between paclitaxel and antidepressants. This comprehensive overview provides clinicians with a complete picture of paclitaxel use for 27 years (1996-2022), enabling them to make informed decisions about paclitaxel treatment.
Acknowledgments
The Department of Research Program funds at Walter Reed National Military Medical Center supported this protocol. We sincerely appreciate the contribution of data extraction from the Joint Pathology Center teams (Francisco J. Rentas, John D. McGeeney, Beatriz A. Hallo, and Johnny P. Beason) and the MHS database personnel (Maj Ryan Costantino, Brandon E. Jenkins, and Alexander G. Rittel). We gratefully thank you for the protocol support from the Department of Research programs: CDR Martin L. Boese, CDR Wesley R. Campbell, Maj. Abhimanyu Chandel, CDR Ling Ye, Chelsea N. Powers, Yaling Zhou, Elizabeth Schafer, Micah Stretch, Diane Beaner, and Adrienne Woodard.
Background
Paclitaxel was first derived from the bark of the yew tree (Taxus brevifolia). It was discovered as part of a National Cancer Institute program screen of plants and natural products with putative anticancer activity during the 1960s.1-9 Paclitaxel works by suppressing spindle microtube dynamics, which results in the blockage of the metaphase-anaphase transitions, inhibition of mitosis, and induction of apoptosis in a broad spectrum of cancer cells. Paclitaxel also displayed additional anticancer activities, including the suppression of cell proliferation and antiangiogenic effects. However, since the growth of normal body cells may also be affected, other adverse effects (AEs) will also occur.8-18
Two different chemotherapy drugs contain paclitaxel—paclitaxel and nab-paclitaxel—and the US Food and Drug Administration (FDA) recognizes them as separate entities.19-21 Taxol (paclitaxel) was approved by the FDA in 1992 for treating advanced ovarian cancer.20 It has since been approved for the treatment of metastatic breast cancer, AIDS-related Kaposi sarcoma (as an orphan drug), non-small cell lung cancer (NSCLC), and cervical cancers (in combination withbevacizumab) in 1994, 1997, 1999, and 2014, respectively.21 Since 2002, a generic version of Taxol, known as paclitaxel injectable, has been FDA-approved from different manufacturers. According to the National Cancer Institute, a combination of carboplatin and Taxol is approved to treat carcinoma of unknown primary, cervical, endometrial, NSCLC, ovarian, and thymoma cancers.19 Abraxane (nab-paclitaxel) was FDA-approved to treat metastatic breast cancer in 2005. It was later approved for first-line treatment of advanced NSCLC and late-stage pancreatic cancer in 2012 and 2013, respectively. In 2018 and 2020, both Taxol and Abraxane were approved for first-line treatment of metastatic squamous cell NSCLC in combination with carboplatin and pembrolizumab and metastatic triple-negative breast cancer in combination with pembrolizumab, respectively.22-26 In 2019, Abraxane was approved with atezolizumab to treat metastatic triple-negative breast cancer, but this approval was withdrawn in 2021. In 2022, a generic version of Abraxane, known as paclitaxel protein-bound, was released in the United States. Furthermore, paclitaxel-containing formulations also are being studied in the treatment of other types of cancer.19-32
One of the main limitations of paclitaxel is its low solubility in water, which complicates its drug supply. To distribute this hydrophobic anticancer drug efficiently, paclitaxel is formulated and administered to patients via polyethoxylated castor oil or albumin-bound (nab-paclitaxel). However, polyethoxylated castor oil induces complement activation and is the cause of common hypersensitivity reactions related to paclitaxel use.2,17,33-38 Therefore, many alternatives to polyethoxylated castor oil have been researched.
Since 2000, new paclitaxel formulations have emerged using nanomedicine techniques. The difference between these formulations is the drug vehicle. Different paclitaxel-based nanotechnological vehicles have been developed and approved, such as albumin-based nanoparticles, polymeric lipidic nanoparticles, polymeric micelles, and liposomes, with many others in clinical trial phases.3,37 Albumin-based nanoparticles have a high response rate (33%), whereas the response rate for polyethoxylated castor oil is 25% in patients with metastatic breast cancer.33,39-52 The use of paclitaxel dimer nanoparticles also has been proposed as a method for increasing drug solubility.33,53
Paclitaxel is metabolized by cytochrome P450 (CYP) isoenzymes 2C8 and 3A4. When administering paclitaxel with known inhibitors, inducers, or substrates of CYP2C8 or CYP3A4, caution is required.19-22 Regulations for CYP research were not issued until 2008, so potential interactions between paclitaxel and other drugs have not been extensively evaluated in clinical trials. A study of 12 kinase inhibitors showed strong inhibition of CYP2C8 and/or CYP3A4 pathways by these inhibitors, which could alter the ratio of paclitaxel metabolites in vivo, leading to clinically relevant changes.54 Differential metabolism has been linked to paclitaxel-induced neurotoxicity in patients with cancer.55 Nonetheless, variants in the CYP2C8, CYP3A4, CYP3A5, and ABCB1 genes do not account for significant interindividual variability in paclitaxel pharmacokinetics.56 In liver microsomes, losartan inhibited paclitaxel metabolism when used at concentrations > 50 µmol/L.57 Many drug-drug interaction (DDI) studies of CYP2C8 and CYP3A4 have shown similar results for paclitaxel.58-64
The goals of this study are to investigate prescribed drugs used with paclitaxel and determine patient outcomes through several Military Health System (MHS) databases. The investigation focused on (1) the functions of paclitaxel; (2) identifying AEs that patients experienced; (3) evaluating differences when paclitaxel is used alone vs concomitantly and between the completed vs discontinued treatment groups; (4) identifying all drugs used during paclitaxel treatment; and (5) evaluating DDIs with antidepressants (that have an FDA boxed warning and are known to have DDIs confirmed in previous publications) and other drugs.65-67
The Walter Reed National Military Medical Center in Bethesda, Maryland, institutionalreview board approved the study protocol and ensured compliance with the Health Insurance Portability and Accountability Act as an exempt protocol. The Joint Pathology Center (JPC) of the US Department of Defense (DoD) Cancer Registry Program and MHS data experts from the Comprehensive Ambulatory/Professional Encounter Record (CAPER) and the Pharmacy Data Transaction Service (PDTS) provided data for the analysis.
METHODS
The DoD Cancer Registry Program was established in 1986 and currently contains data from 1998 to 2024. CAPER and PDTS are part of the MHS Data Repository/Management Analysis and Reporting Tool database. Each observation in the CAPER record represents an ambulatory encounter at a military treatment facility (MTF). CAPER includes data from 2003 to 2024.
Each observation in the PDTS record represents a prescription filled for an MHS beneficiary at an MTF through the TRICARE mail-order program or a US retail pharmacy. Missing from this record are prescriptions filled at international civilian pharmacies and inpatient pharmacy prescriptions. The MHS Data Repository PDTS record is available from 2002 to 2024. The legacy Composite Health Care System is being replaced by GENESIS at MTFs.
Data Extraction Design
The study design involved a cross-sectional analysis. We requested data extraction for paclitaxel from 1998 to 2022. Data from the DoD Cancer Registry Program were used to identify patients who received cancer treatment. Once patients were identified, the CAPER database was searched for diagnoses to identify other health conditions, whereas the PDTS database was used to populate a list of prescription medications filled during chemotherapy treatment.
Data collected from the JPC included cancer treatment, cancer information, demographics, and physicians’ comments on AEs. Collected data from the MHS include diagnosis and filled prescription history from initiation to completion of the therapy period (or 2 years after the diagnosis date). For the analysis of the DoD Cancer Registry Program and CAPER databases, we used all collected data without excluding any. When analyzing PDTS data, we excluded patients with PDTS data but without a record of paclitaxel being filled, or medications filled outside the chemotherapy period (by evaluating the dispensed date and day of supply).
Data Extraction Analysis
The Surveillance, Epidemiology, and End Results Program Coding and Staging Manual 2016 and the International Classification of Diseases for Oncology, 3rd edition, 1st revision, were used to decode disease and cancer types.68,69 Data sorting and analysis were performed using Microsoft Excel. The percentage for the total was calculated by using the number of patients or data available within the paclitaxel groups divided by the total number of patients or data variables. The subgroup percentage was calculated by using the number of patients or data available within the subgroup divided by the total number of patients in that subgroup.
In alone vs concomitant and completed vs discontinued treatment groups, a 2-tailed, 2-sample z test was used to statistical significance (P < .05) using a statistics website.70 Concomitant was defined as paclitaxel taken with other antineoplastic agent(s) before, after, or at the same time as cancer therapy. For the retrospective data analysis, physicians’ notes with a period, comma, forward slash, semicolon, or space between medication names were interpreted as concurrent, whereas plus (+), minus/plus (-/+), or “and” between drug names that were dispensed on the same day were interpreted as combined with known common combinations: 2 drugs (DM886 paclitaxel and carboplatin and DM881-TC-1 paclitaxel and cisplatin) or 3 drugs (DM887-ACT doxorubicin, cyclophosphamide, and paclitaxel). Completed treatment was defined as paclitaxel as the last medication the patient took without recorded AEs; switching or experiencing AEs was defined as discontinued treatment.
RESULTS
The JPC provided 702 entries for 687 patients with a mean age of 56 years (range, 2 months to 88 years) who were treated with paclitaxel from March 1996 to October 2021. Fifteen patients had duplicate entries because they had multiple cancer sites or occurrences. There were 623 patients (89%) who received paclitaxel for FDA-approved indications. The most common types of cancer identified were 344 patients with breast cancer (49%), 91 patients with lung cancer (13%), 79 patients with ovarian cancer (11%), and 75 patients with endometrial cancer (11%) (Table 1). Seventy-nine patients (11%) received paclitaxel for cancers that were not for FDA-approved indications, including 19 for cancers of the fallopian tube (3%) and 17 for esophageal cancer (2%) (Table 2).
There were 477 patients (68%) aged > 50 years. A total of 304 patients (43%) had a stage III or IV cancer diagnosis and 398 (57%) had stage II or lower (combination of data for stages 0, I, and II; not applicable; and unknown) cancer diagnosis. For systemic treatment, 16 patients (2%) were treated with paclitaxel alone and 686 patients (98%) received paclitaxel concomitantly with additional chemotherapy: 59 patients (9%) in the before or after group, 410 patients (58%) had a 2-drug combination, 212 patients (30%) had a 3-drug combination, and 5 patients (1%) had a 4-drug combination. In addition, for doublet therapies, paclitaxel combined with carboplatin, trastuzumab, gemcitabine, or cisplatin had more patients (318, 58, 12, and 11, respectively) than other combinations (≤ 4 patients). For triplet therapies, paclitaxel combined withdoxorubicin plus cyclophosphamide or carboplatin plus bevacizumab had more patients (174 and 20, respectively) than other combinations, including quadruplet therapies (≤ 4 patients) (Table 3).
Patients were more likely to discontinue paclitaxel if they received concomitant treatment. None of the 16 patients receiving paclitaxel monotherapy experienced AEs, whereas 364 of 686 patients (53%) treated concomitantly discontinued (P < .001). Comparisons of 1 drug vs combination (2 to 4 drugs) and use for treating cancers that were FDA-approved indications vs off-label use were significant (P < .001), whereas comparisons of stage II or lower vs stage III and IV cancer and of those aged ≤ 50 years vs aged > 50 years were not significant (P = .50 andP = .30, respectively) (Table 4).
Among the 364 patients who had concomitant treatment and had discontinued their treatment, 332 (91%) switched treatments with no AEs documented and 32 (9%) experienced fatigue with pneumonia, mucositis, neuropathy, neurotoxicity, neutropenia, pneumonitis, allergic or hypersensitivity reaction, or an unknown AE. Patients who discontinued treatment because of unknown AEs had a physician’s note that detailed progressive disease, a significant decline in performance status, and another unknown adverse effect due to a previous sinus tract infection and infectious colitis (Table 5).
Management Analysis and Reporting Tool Database
MHS data analysts provided data on diagnoses for 639 patients among 687 submitteddiagnoses, with 294 patients completing and 345 discontinuing paclitaxel treatment. Patients in the completed treatment group had 3 to 258 unique health conditions documented, while patients in the discontinued treatment group had 4 to 181 unique health conditions documented. The MHS reported 3808 unique diagnosis conditions for the completed group and 3714 for the discontinued group (P = .02).
The mean (SD) number of diagnoses was 51 (31) for the completed and 55 (28) for the discontinued treatment groups (Figure). Among 639 patients who received paclitaxel, the top 5 diagnoses were administrative, including encounters for other administrative examinations; antineoplastic chemotherapy; administrative examination for unspecified; other specified counseling; and adjustment and management of vascular access device. The database does not differentiate between administrative and clinically significant diagnoses.
MHS data analysts provided data for 336 of 687 submitted patients who were prescribed paclitaxel; 46 patients had no PDTS data, and 305 patients had PDTS data without paclitaxel, Taxol, or Abraxane dispensed. Medications that were filled outside the chemotherapy period were removed by evaluating the dispensed date and day of supply. Among these 336 patients, 151 completed the treatment and 185 discontinued, with 14 patients experiencing documented AEs. Patients in the completed treatment group filled 9 to 56 prescriptions while patients in the discontinued treatment group filled 6 to 70 prescriptions.Patients in the discontinued group filled more prescriptions than those who completed treatment: 793 vs 591, respectively (P = .34).
The mean (SD) number of filled prescription drugs was 24 (9) for the completed and 34 (12) for the discontinued treatment group. The 5 most filled prescriptions with paclitaxel from 336 patients with PDTS data were dexamethasone (324 prescriptions with 14 recorded AEs), diphenhydramine (296 prescriptions with 12 recorded AEs), ondansetron (277 prescriptions with 11 recorded AEs), prochlorperazine (265 prescriptions with 12 recorded AEs), and sodium chloride (232 prescriptions with 11 recorded AEs).
DISCUSSION
As a retrospective review, this study is more limited in the strength of its conclusions when compared to randomized control trials. The DoD Cancer Registry Program only contains information about cancer types, stages, treatment regimens, and physicians’ notes. Therefore, noncancer drugs are based solely on the PDTS database. In most cases, physicians' notes on AEs were not detailed. There was no distinction between initial vs later lines of therapy and dosage reductions. The change in status or appearance of a new medical condition did not indicate whether paclitaxel caused the changes to develop or directly worsen a pre-existing condition. The PDTS records prescriptions filled, but that may not reflect patients taking prescriptions.
Paclitaxel
Paclitaxel has a long list of both approved and off-label uses in malignancies as a primary agent and in conjunction with other drugs. The FDA prescribing information for Taxol and Abraxane was last updated in April 2011 and September 2020, respectively.20,21 The National Institutes of Health National Library of Medicine has the current update for paclitaxel on July 2023.19,22 Thus, the prescribed information for paclitaxel referenced in the database may not always be up to date. The combinations of paclitaxel with bevacizumab, carboplatin, or carboplatin and pembrolizumab were not in the Taxol prescribing information. Likewise, a combination of nab-paclitaxel with atezolizumab or carboplatin and pembrolizumab is missing in the Abraxane prescribing information.22-27
The generic name is not the same as a generic drug, which may have slight differences from the brand name product.71 The generic drug versions of Taxol and Abraxane have been approved by the FDA as paclitaxel injectable and paclitaxel-protein bound, respectively. There was a global shortage of nab-paclitaxel from October 2021 to June 2022 because of a manufacturing problem.72 During this shortage, data showed similar comments from physician documents that treatment switched to Taxol due to the Abraxane shortage.
Of 336 patients in the PDTS database with dispensed paclitaxel prescriptions, 276 received paclitaxel (year dispensed, 2013-2022), 27 received Abraxane (year dispensed, 2013-2022), 47 received Taxol (year dispensed, 2004-2015), 8 received both Abraxane and paclitaxel, and 6 received both Taxol and paclitaxel. Based on this information, it appears that the distinction between the drugs was not made in the PDTS until after 2015, 10 years after Abraxane received FDA approval. Abraxane was prescribed in the MHS in 2013, 8 years after FDA approval. There were a few comparison studies of Abraxane and Taxol.73-76
Safety and effectiveness in pediatric patients have not been established for paclitaxel. According to the DoD Cancer Registry Program, the youngest patient was aged 2 months. In 2021, this patient was diagnosed with corpus uteri and treated with carboplatin and Taxol in course 1; in course 2, the patient reacted to Taxol; in course 3, Taxol was replaced with Abraxane; in courses 4 to 7, the patient was treated with carboplatin only.
Discontinued Treatment
Ten patients had prescribed Taxol that was changed due to AEs: 1 was switched to Abraxane and atezolizumab, 3 switched to Abraxane, 2 switched to docetaxel, 1 switched to doxorubicin, and 3 switched to pembrolizumab (based on physician’s comments). Of the 10 patients, 7 had Taxol reaction, 2 experienced disease progression, and 1 experienced high programmed death–ligand 1 expression (this patient with breast cancer was switched to Abraxane and atezolizumab during the accelerated FDA approval phase for atezolizumab, which was later revoked). Five patients were treated with carboplatin and Taxol for cancer of the anal canal (changed to pembrolizumab after disease progression), lung not otherwise specified (changed to carboplatin and pembrolizumab due to Taxol reaction), lower inner quadrant of the breast (changed to doxorubicin due to hypersensitivity reaction), corpus uteri (changed to Abraxane due to Taxol reaction), and ovary (changed to docetaxel due to Taxol reaction). Three patients were treated with doxorubicin, cyclophosphamide, and Taxol for breast cancer; 2 patients with breast cancer not otherwise specified switched to Abraxane due to cardiopulmonary hypersensitivity and Taxol reaction and 1 patient with cancer of the upper outer quadrant of the breast changed to docetaxel due to allergic reaction. One patient, who was treated with paclitaxel, ifosfamide, and cisplatin for metastasis of the lower lobe of the lung and kidney cancer, experienced complications due to infectious colitis (treated with ciprofloxacin) and then switched to pembrolizumab after the disease progressed. These AEs are known in paclitaxel medical literature on paclitaxel AEs.19-24,77-81
Combining 2 or more treatments to target cancer-inducing or cell-sustaining pathways is a cornerstone of chemotherapy.82-84 Most combinations are given on the same day, but some are not. For 3- or 4-drug combinations, doxorubicin and cyclophosphamide were given first, followed by paclitaxel with or withouttrastuzumab, carboplatin, or pembrolizumab. Only 16 patients (2%) were treated with paclitaxel alone; therefore, the completed and discontinued treatment groups are mostly concomitant treatment. As a result, the comparisons of the completed and discontinued treatment groups were almost the same for the diagnosis. The PDTS data have a better result because 2 exclusion criteria were applied before narrowing the analysis down to paclitaxel treatment specifically.
Antidepressants and Other Drugs
Drug response can vary from person to person and can lead to treatment failure related to AEs. One major factor in drug metabolism is CYP.85 CYP2C8 is the major pathway for paclitaxel and CYP3A4 is the minor pathway. When evaluating the noncancer drugs, there were no reports of CYP2C8 inhibition or induction. Over the years, many DDI warnings have been issued for paclitaxel with different drugs in various electronic resources.
Oncologists follow guidelines to prevent DDIs, as paclitaxel is known to have severe, moderate, and minor interactions with other drugs. Among 687 patients, 261 (38%) were prescribed any of 14 antidepressants. Eight of these antidepressants (amitriptyline, citalopram, desipramine, doxepin, venlafaxine, escitalopram, nortriptyline, and trazodone) are metabolized, 3 (mirtazapine, sertraline, and fluoxetine) are metabolized and inhibited, 2 (bupropion and duloxetine) are neither metabolized nor inhibited, and 1 (paroxetine) is inhibited by CYP3A4. Duloxetine, venlafaxine, and trazodone were more commonly dispensed (84, 78, and 42 patients, respectively) than others (≤ 33 patients).
Of 32 patients with documented AEs,14 (44%) had 168 dispensed drugs in the PDTS database. Six patients (19%) were treated with doxorubicin and cyclophosphamide followed by paclitaxel for breast cancer; 6 (19%) were treated with carboplatin and paclitaxel for cancer of the lung (n = 3), corpus uteri (n = 2), and ovary (n = 1); 1 patient (3%) was treated with carboplatin and paclitaxel, then switched to carboplatin, bevacizumab, and paclitaxel, and then completed treatment with carboplatin and paclitaxel for an unspecified female genital cancer; and 1 patient (3%) was treated with cisplatin, ifosfamide, and paclitaxel for metastasis of the lower lobe lung and kidney cancer.
The 14 patients with PDTS data had 18 cancer drugs dispensed. Eleven had moderate interaction reports and 7 had no interaction reports. A total of 165 noncancer drugs were dispensed, of which 3 were antidepressants and had no interactions reported, 8 had moderate interactions reported, and 2 had minor interactions with Taxol and Abraxane, respectively (Table 6).86-129
Of 3 patients who were dispensed bupropion, nortriptyline, or paroxetine, 1 patient with breast cancer was treated with doxorubicin andcyclophosphamide, followed by paclitaxel with bupropion, nortriptyline, pegfilgrastim,dexamethasone, and 17 other noncancer drugs that had no interaction report dispensed during paclitaxel treatment. Of 2 patients with lung cancer, 1 patient was treated with carboplatin and paclitaxel with nortriptyline, dexamethasone, and 13 additional medications, and the second patient was treated with paroxetine, cimetidine, dexamethasone, and 12 other medications. Patients were dispensed up to6 noncancer medications on the same day as paclitaxel administration to control the AEs, not including the prodrugs filled before the treatments. Paroxetine and cimetidine have weak inhibition, and dexamethasone has weak induction of CYP3A4. Therefore, while 1:1 DDIs might have little or no effect with weak inhibit/induce CYP3A4 drugs, 1:1:1 or more combinations could have a different outcome (confirmed in previous publications).65-67
Dispensed on the same day may not mean taken at the same time. One patient experienced an AE with dispensed 50 mg losartan, carboplatin plus paclitaxel, dexamethasone, and 6 other noncancer drugs. Losartan inhibits paclitaxel, which can lead to negative AEs.57,66,67 However, there were no blood or plasma samples taken to confirm the losartan was taken at the same time as the paclitaxel given this was not a clinical trial.
Conclusions
This retrospective study discusses the use of paclitaxel in the MHS and the potential DDIs associated with it. The study population consisted mostly of active-duty personnel, who are required to be healthy or have controlled or nonactive medical diagnoses and be physically fit. This group is mixed with dependents and retirees that are more reflective of the average US population. As a result, this patient population is healthier than the general population, with a lower prevalence of common illnesses such as diabetes and obesity. The study aimed to identify drugs used alongside paclitaxel treatment. While further research is needed to identify potential DDIs among patients who experienced AEs, in vitro testing will need to be conducted before confirming causality. The low number of AEs experienced by only 32 of 702 patients (5%), with no deaths during paclitaxel treatment, indicates that the drug is generally well tolerated. Although this study cannot conclude that concomitant use with noncancer drugs led to the discontinuation of paclitaxel, we can conclude that there seems to be no significant DDIsidentified between paclitaxel and antidepressants. This comprehensive overview provides clinicians with a complete picture of paclitaxel use for 27 years (1996-2022), enabling them to make informed decisions about paclitaxel treatment.
Acknowledgments
The Department of Research Program funds at Walter Reed National Military Medical Center supported this protocol. We sincerely appreciate the contribution of data extraction from the Joint Pathology Center teams (Francisco J. Rentas, John D. McGeeney, Beatriz A. Hallo, and Johnny P. Beason) and the MHS database personnel (Maj Ryan Costantino, Brandon E. Jenkins, and Alexander G. Rittel). We gratefully thank you for the protocol support from the Department of Research programs: CDR Martin L. Boese, CDR Wesley R. Campbell, Maj. Abhimanyu Chandel, CDR Ling Ye, Chelsea N. Powers, Yaling Zhou, Elizabeth Schafer, Micah Stretch, Diane Beaner, and Adrienne Woodard.
1. American Chemical Society. Discovery of camptothecin and taxol. acs.org. Accessed June 4, 2024. https://www.acs.org/education/whatischemistry/landmarks/camptothecintaxol.html
2. Bocci G, Di Paolo A, Danesi R. The pharmacological bases of the antiangiogenic activity of paclitaxel. Angiogenesis. 2013;16(3):481-492. doi:10.1007/s10456-013-9334-0.
3. Meštrovic T. Paclitaxel history. News Medical Life Sciences. Updated March 11, 2023. Accessed June 4, 2024. https://www.news-medical.net/health/Paclitaxel-History.aspx
4. Rowinsky EK, Donehower RC. Paclitaxel (taxol). N Engl J Med. 1995;332(15):1004-1014. doi:10.1056/NEJM199504133321507
5. Walsh V, Goodman J. The billion dollar molecule: Taxol in historical and theoretical perspective. Clio Med. 2002;66:245-267. doi:10.1163/9789004333499_013
6. Perdue RE, Jr, Hartwell JL. The search for plant sources of anticancer drugs. Morris Arboretum Bull. 1969;20:35-53.
7. Wall ME, Wani MC. Camptothecin and taxol: discovery to clinic—thirteenth Bruce F. Cain Memorial Award lecture. Cancer Res. 1995;55:753-760.
8. Wani MC, Taylor HL, Wall ME, Coggon P, McPhail AT. Plant antitumor agents. VI. The isolation and structure of taxol, a novel antileukemic and antitumor agent from taxus brevifolia. J Am Chem Soc. 1971;93(9):2325-2327. doi:10.1021/ja00738a045
9. Weaver BA. How taxol/paclitaxel kills cancer cells. Mol Biol Cell. 2014;25(18):2677-2681. doi:10.1091/mbc.E14-04-0916
10. Chen JG, Horwitz SB. Differential mitotic responses to microtubule-stabilizing and-destabilizing drugs. Cancer Res. 2002;62(7):1935-1938.
11. Singh S, Dash AK. Paclitaxel in cancer treatment: perspectives and prospects of its delivery challenges. Crit Rev Ther Drug Carrier Syst. 2009;26(4):333-372. doi:10.1615/critrevtherdrugcarriersyst.v26.i4.10
12. Schiff PB, Fant J, Horwitz SB. Promotion of microtubule assembly in vitro by taxol. Nature. 1979;277(5698):665-667. doi:10.1038/277665a0
13. Fuchs DA, Johnson RK. Cytologic evidence that taxol, an antineoplastic agent from taxus brevifolia, acts as a mitotic spindle poison. Cancer Treat Rep. 1978;62(8):1219-1222.
14. Walsh V, Goodman J. From taxol to taxol: the changing identities and ownership of an anti-cancer drug. Med Anthropol. 2002;21(3-4):307-336. doi:10.1080/01459740214074
15. Walsh V, Goodman J. Cancer chemotherapy, biodiversity, public and private property: the case of the anti-cancer drug taxol. Soc Sci Med. 1999;49(9):1215-1225. doi:10.1016/s0277-9536(99)00161-6
16. Jordan MA, Wendell K, Gardiner S, Derry WB, Copp H, Wilson L. Mitotic block induced in HeLa cells by low concentrations of paclitaxel (taxol) results in abnormal mitotic exit and apoptotic cell death. Cancer Res. 1996;56(4):816-825.
17. Picard M, Castells MC. Re-visiting hypersensitivity reactions to taxanes: a comprehensive review. Clin Rev Allergy Immunol. 2015;49(2):177-191. doi:10.1007/s12016-014-8416-0
18. Zasadil LM, Andersen KA, Yeum D, et al. Cytotoxicity of paclitaxel in breast cancer is due to chromosome missegregation on multipolar spindles. Sci Transl Med. 2014;6:229ra243. doi:10.1126/scitranslmed.3007965
19. National Cancer Institute. Carboplatin-Taxol. Published May 30, 2012. Updated March 22, 2023. Accessed June 4, 2024. https://www.cancer.gov/about-cancer/treatment/drugs/carboplatin-taxol
20. Taxol (paclitaxel). Prescribing information. Bristol-Myers Squibb; 2011. Accessed June 4, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2011/020262s049lbl.pdf
21. Abraxane (paclitaxel). Prescribing information. Celgene Corporation; 2021. Accessed June 4, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2020/021660s047lbl.pdf
22. Awosika AO, Farrar MC, Jacobs TF. Paclitaxel. StatPearls. Updated November 18, 2023. Accessed June 5, 2024. https://www.ncbi.nlm.nih.gov/books/NBK536917/
23. Gerriets V, Kasi A. Bevacizumab. StatPearls. Updated September 1, 2022. Accessed June 5, 2024. https://www.ncbi.nlm.nih.gov/books/NBK482126/
24. American Cancer Society. Chemotherapy for endometrial cancer. Updated March 27, 2019. Accessed June 4, 2024. https://www.cancer.org/cancer/types/endometrial-cancer/treating/chemotherapy.html
25. US Food and Drug Administration. FDA approves pembrolizumab in combination with chemotherapy for first-line treatment of metastatic squamous NSCLC. October 30, 2018. Updated December 14, 2018. Accessed June 4, 2024. https://www.fda.gov/drugs/fda-approves-pembrolizumab-combination-chemotherapy-first-line-treatment-metastatic-squamous-nsclc
26. US Food and Drug Administration. FDA grants accelerated approval to pembrolizumab for locally recurrent unresectable or metastatic triple negative breast cancer. November 13, 2020. Accessed June 4, 2024. https://www.fda.gov/drugs/resources-information-approved-drugs/fda-grants-accelerated-approval-pembrolizumab-locally-recurrent-unresectable-or-metastatic-triple
27. US Food and Drug Administration. FDA approves atezolizumab for PD-L1 positive unresectable locally advanced or metastatic triple-negative breast. March 8, 2019. Updated March 18, 2019. Accessed June 5, 2024. https://www.fda.gov/drugs/drug-approvals-and-databases/fda-approves-atezolizumab-pd-l1-positive-unresectable-locally-advanced-or-metastatic-triple-negative
28. US Food and Drug Administration. FDA issues alert about efficacy and potential safety concerns with atezolizumab in combination with paclitaxel for treatment of breast cancer. September 8, 2020. Accessed June 5, 2024. https://www.fda.gov/drugs/resources-information-approved-drugs/fda-issues-alert-about-efficacy-and-potential-safety-concerns-atezolizumab-combination-paclitaxel
29. Tan AR. Chemoimmunotherapy: still the standard of care for metastatic triple-negative breast cancer. ASCO Daily News. February 23, 2022. Accessed June 5, 2024. https://dailynews.ascopubs.org/do/chemoimmunotherapy-still-standard-care-metastatic-triple-negative-breast-cancer
30. McGuire WP, Rowinsky EK, Rosenshein NB, et al. Taxol: a unique antineoplastic agent with significant activity in advanced ovarian epithelial neoplasms. Ann Intern Med. 1989;111(4):273-279. doi:10.7326/0003-4819-111-4-273
31. Milas L, Hunter NR, Kurdoglu B, et al. Kinetics of mitotic arrest and apoptosis in murine mammary and ovarian tumors treated with taxol. Cancer Chemother Pharmacol. 1995;35(4):297-303. doi:10.1007/BF00689448
32. Searle J, Collins DJ, Harmon B, Kerr JF. The spontaneous occurrence of apoptosis in squamous carcinomas of the uterine cervix. Pathology. 1973;5(2):163-169. doi:10.3109/00313027309060831
33. Gallego-Jara J, Lozano-Terol G, Sola-Martínez RA, Cánovas-Díaz M, de Diego Puente T. A compressive review about taxol®: history and future challenges. Molecules. 2020;25(24):5986. doi:10.3390/molecules25245986
34. Bernabeu E, Cagel M, Lagomarsino E, Moretton M, Chiappetta DA. Paclitaxel: What has been done and the challenges remain ahead. Int J Pharm. 2017;526(1-2):474-495. doi:10.1016/j.ijpharm.2017.05.016
35. Nehate C, Jain S, Saneja A, et al. Paclitaxel formulations: challenges and novel delivery options. Curr Drug Deliv. 2014;11(6):666-686. doi:10.2174/1567201811666140609154949
36. Gelderblom H, Verweij J, Nooter K, Sparreboom A, Cremophor EL. The drawbacks and advantages of vehicle selection for drug formulation. Eur J Cancer. 2001;37(13):1590-1598. doi:10.1016/S0959-8049(01)00171-x
37. Chowdhury MR, Moshikur RM, Wakabayashi R, et al. In vivo biocompatibility, pharmacokinetics, antitumor efficacy, and hypersensitivity evaluation of ionic liquid-mediated paclitaxel formulations. Int J Pharm. 2019;565:219-226. doi:10.1016/j.ijpharm.2019.05.020
38. Borgå O, Henriksson R, Bjermo H, Lilienberg E, Heldring N, Loman N. Maximum tolerated dose and pharmacokinetics of paclitaxel micellar in patients with recurrent malignant solid tumours: a dose-escalation study. Adv Ther. 2019;36(5):1150-1163. doi:10.1007/s12325-019-00909-6
39. Rouzier R, Rajan R, Wagner P, et al. Microtubule-associated protein tau: a marker of paclitaxel sensitivity in breast cancer. Proc Natl Acad Sci USA. 2005;102(23):8315-8320. doi:10.1073/pnas.0408974102
40. Choudhury H, Gorain B, Tekade RK, Pandey M, Karmakar S, Pal TK. Safety against nephrotoxicity in paclitaxel treatment: oral nanocarrier as an effective tool in preclinical evaluation with marked in vivo antitumor activity. Regul Toxicol Pharmacol. 2017;91:179-189. doi:10.1016/j.yrtph.2017.10.023
41. Barkat MA, Beg S, Pottoo FH, Ahmad FJ. Nanopaclitaxel therapy: an evidence based review on the battle for next-generation formulation challenges. Nanomedicine (Lond). 2019;14(10):1323-1341. doi:10.2217/nnm-2018-0313
42. Sofias AM, Dunne M, Storm G, Allen C. The battle of “nano” paclitaxel. Adv Drug Deliv Rev. 2017;122:20-30. doi:10.1016/j.addr.2017.02.003
43. Yang N, Wang C, Wang J, et al. Aurora inase a stabilizes FOXM1 to enhance paclitaxel resistance in triple-negative breast cancer. J Cell Mol Med. 2019;23(9):6442-6453. doi:10.1111/jcmm.14538
44. Chowdhury MR, Moshikur RM, Wakabayashi R, et al. Ionic-liquid-based paclitaxel preparation: a new potential formulation for cancer treatment. Mol Pharm. 2018;15(16):2484-2488. doi:10.1021/acs.molpharmaceut.8b00305
45. Chung HJ, Kim HJ, Hong ST. Tumor-specific delivery of a paclitaxel-loading HSA-haemin nanoparticle for cancer treatment. Nanomedicine. 2020;23:102089. doi:10.1016/j.nano.2019.102089
46. Ye L, He J, Hu Z, et al. Antitumor effect and toxicity of lipusu in rat ovarian cancer xenografts. Food Chem Toxicol. 2013;52:200-206. doi:10.1016/j.fct.2012.11.004
47. Ma WW, Lam ET, Dy GK, et al. A pharmacokinetic and dose-escalating study of paclitaxel injection concentrate for nano-dispersion (PICN) alone and with arboplatin in patients with advanced solid tumors. J Clin Oncol. 2013;31:2557. doi:10.1200/jco.2013.31.15_suppl.2557
48. Micha JP, Goldstein BH, Birk CL, Rettenmaier MA, Brown JV. Abraxane in the treatment of ovarian cancer: the absence of hypersensitivity reactions. Gynecol Oncol. 2006;100(2):437-438. doi:10.1016/j.ygyno.2005.09.012
49. Ingle SG, Pai RV, Monpara JD, Vavia PR. Liposils: an effective strategy for stabilizing paclitaxel loaded liposomes by surface coating with silica. Eur J Pharm Sci. 2018;122:51-63. doi:10.1016/j.ejps.2018.06.025
50. Abriata JP, Turatti RC, Luiz MT, et al. Development, characterization and biological in vitro assays of paclitaxel-loaded PCL polymeric nanoparticles. Mater Sci Eng C Mater Biol Appl. 2019;96:347-355. doi:10.1016/j.msec.2018.11.035
51. Hu J, Fu S, Peng Q, et al. Paclitaxel-loaded polymeric nanoparticles combined with chronomodulated chemotherapy on lung cancer: in vitro and in vivo evaluation. Int J Pharm. 2017;516(1-2):313-322. doi:10.1016/j.ijpharm.2016.11.047
52. Dranitsaris G, Yu B, Wang L, et al. Abraxane® vs Taxol® for patients with advanced breast cancer: a prospective time and motion analysis from a chinese health care perspective. J Oncol Pharm Pract. 2016;22(2):205-211. doi:10.1177/1078155214556008
53. Pei Q, Hu X, Liu S, Li Y, Xie Z, Jing X. Paclitaxel dimers assembling nanomedicines for treatment of cervix carcinoma. J Control Release. 2017;254:23-33. doi:10.1016/j.jconrel.2017.03.391
54. Wang Y, Wang M, Qi H, et al. Pathway-dependent inhibition of paclitaxel hydroxylation by kinase inhibitors and assessment of drug-drug interaction potentials. Drug Metab Dispos. 2014;42(4):782-795. doi:10.1124/dmd.113.053793
55. Shen F, Jiang G, Philips S, et al. Cytochrome P450 oxidoreductase (POR) associated with severe paclitaxel-induced peripheral neuropathy in patients of european ancestry from ECOG-ACRIN E5103. Clin Cancer Res. 2023;29(13):2494-2500. doi:10.1158/1078-0432.CCR-22-2431
56. Henningsson A, Marsh S, Loos WJ, et al. Association of CYP2C8, CYP3A4, CYP3A5, and ABCB1 polymorphisms with the pharmacokinetics of paclitaxel. Clin Cancer Res. 2005;11(22):8097-8104. doi:10.1158/1078-0432.CCR-05-1152
57. Mukai Y, Senda A, Toda T, et al. Drug-drug interaction between losartan and paclitaxel in human liver microsomes with different CYP2C8 genotypes. Basic Clin Pharmacol Toxicol. 2015;116(6):493-498. doi:10.1111/bcpt.12355
58. Kawahara B, Faull KF, Janzen C, Mascharak PK. Carbon monoxide inhibits cytochrome P450 enzymes CYP3A4/2C8 in human breast cancer cells, increasing sensitivity to paclitaxel. J Med Chem. 2021;64(12):8437-8446. doi:10.1021/acs.jmedchem.1c00404
59. Cresteil T, Monsarrat B, Dubois J, Sonnier M, Alvinerie P, Gueritte F. Regioselective metabolism of taxoids by human CYP3A4 and 2C8: structure-activity relationship. Drug Metab Dispos. 2002;30(4):438-445. doi:10.1124/dmd.30.4.438
60. Taniguchi R, Kumai T, Matsumoto N, et al. Utilization of human liver microsomes to explain individual differences in paclitaxel metabolism by CYP2C8 and CYP3A4. J Pharmacol Sci. 2005;97(1):83-90. doi:10.1254/jphs.fp0040603
61. Nakayama A, Tsuchiya K, Xu L, Matsumoto T, Makino T. Drug-interaction between paclitaxel and goshajinkigan extract and its constituents. J Nat Med. 2022;76(1):59-67. doi:10.1007/s11418-021-01552-8
62. Monsarrat B, Chatelut E, Royer I, et al. Modification of paclitaxel metabolism in a cancer patient by induction of cytochrome P450 3A4. Drug Metab Dispos. 1998;26(3):229-233.
63. Walle T. Assays of CYP2C8- and CYP3A4-mediated metabolism of taxol in vivo and in vitro. Methods Enzymol. 1996;272:145-151. doi:10.1016/s0076-6879(96)72018-9
64. Hanioka N, Matsumoto K, Saito Y, Narimatsu S. Functional characterization of CYP2C8.13 and CYP2C8.14: catalytic activities toward paclitaxel. Basic Clin Pharmacol Toxicol. 2010;107(1):565-569. doi:10.1111/j.1742-7843.2010.00543.x
65. Luong TT, Powers CN, Reinhardt BJ, Weina PJ. Pre-clinical drug-drug interactions (DDIs) of gefitinib with/without losartan and selective serotonin reuptake inhibitors (SSRIs): citalopram, fluoxetine, fluvoxamine, paroxetine, sertraline, and venlafaxine. Curr Res Pharmacol Drug Discov. 2022;3:100112. doi:10.1016/j.crphar.2022.100112
66. Luong TT, McAnulty MJ, Evers DL, Reinhardt BJ, Weina PJ. Pre-clinical drug-drug interaction (DDI) of gefitinib or erlotinib with Cytochrome P450 (CYP) inhibiting drugs, fluoxetine and/or losartan. Curr Res Toxicol. 2021;2:217-224. doi:10.1016/j.crtox.2021.05.006
67. Luong TT, Powers CN, Reinhardt BJ, et al. Retrospective evaluation of drug-drug interactions with erlotinib and gefitinib use in the military health system. Fed Pract. 2023;40(suppl 3):S24-S34. doi:10.12788/fp.0401
68. Adamo M, Dickie L, Ruhl J. SEER program coding and staging manual 2016. National Cancer Institute. Accessed June 5, 2024. https://seer.cancer.gov/archive/manuals/2016/SPCSM_2016_maindoc.pdf
69. World Health Organization. International classification of diseases for oncology (ICD-O) 3rd ed, 1st revision. World Health Organization; 2013. Accessed June 5, 2024. https://apps.who.int/iris/handle/10665/96612
70. Z score calculator for 2 population proportions. Social science statistics. Accessed June 5, 2024. https://www.socscistatistics.com/tests/ztest/default2.aspx
71. US Food and Drug Administration. Generic drugs: question & answers. FDA.gov. Accessed June 5, 2024. https://www.fda.gov/drugs/frequently-asked-questions-popular-topics/generic-drugs-questions-answers
72. Oura M, Saito H, Nishikawa Y. Shortage of nab-paclitaxel in Japan and around the world: issues in global information sharing. JMA J. 2023;6(2):192-195. doi:10.31662/jmaj.2022-0179
73. Yuan H, Guo H, Luan X, et al. Albumin nanoparticle of paclitaxel (abraxane) decreases while taxol increases breast cancer stem cells in treatment of triple negative breast cancer. Mol Pharm. 2020;17(7):2275-2286. doi:10.1021/acs.molpharmaceut.9b01221
74. Dranitsaris G, Yu B, Wang L, et al. Abraxane® versus Taxol® for patients with advanced breast cancer: a prospective time and motion analysis from a Chinese health care perspective. J Oncol Pharm Pract. 2016;22(2):205-211. doi:10.1177/1078155214556008
75. Gradishar WJ, Tjulandin S, Davidson N, et al. Phase III trial of nanoparticle albumin-bound paclitaxel compared with polyethylated castor oil-based paclitaxel in women with breast cancer. J Clin Oncol. 2005;23(31):7794-7803. doi:10.1200/JCO.2005.04.
76. Liu M, Liu S, Yang L, Wang S. Comparison between nab-paclitaxel and solvent-based taxanes as neoadjuvant therapy in breast cancer: a systematic review and meta-analysis. BMC Cancer. 2021;21(1):118. doi:10.1186/s12885-021-07831-7
77. Rowinsky EK, Eisenhauer EA, Chaudhry V, Arbuck SG, Donehower RC. Clinical toxicities encountered with paclitaxel (taxol). Semin Oncol. 1993;20(4 Suppl 3):1-15.
78. Banerji A, Lax T, Guyer A, Hurwitz S, Camargo CA Jr, Long AA. Management of hypersensitivity reactions to carboplatin and paclitaxel in an outpatient oncology infusion center: a 5-year review. J Allergy Clin Immunol Pract. 2014;2(4):428-433. doi:10.1016/j.jaip.2014.04.010
79. Staff NP, Fehrenbacher JC, Caillaud M, Damaj MI, Segal RA, Rieger S. Pathogenesis of paclitaxel-induced peripheral neuropathy: a current review of in vitro and in vivo findings using rodent and human model systems. Exp Neurol. 2020;324:113121. doi:10.1016/j.expneurol.2019.113121
80. Postma TJ, Vermorken JB, Liefting AJ, Pinedo HM, Heimans JJ. Paclitaxel-induced neuropathy. Ann Oncol. 1995;6(5):489-494. doi:10.1093/oxfordjournals.annonc.a059220
81. Liu JM, Chen YM, Chao Y, et al. Paclitaxel-induced severe neuropathy in patients with previous radiotherapy to the head and neck region. J Natl Cancer Inst. 1996;88(14):1000-1002. doi:10.1093/jnci/88.14.1000-a
82. Bayat Mokhtari R, Homayouni TS, Baluch N, et al. Combination therapy in combating cancer. Oncotarget. 2017;8(23):38022-38043. doi:10.18632/oncotarget.16723
83. Blagosklonny MV. Analysis of FDA approved anticancer drugs reveals the future of cancer therapy. Cell Cycle. 2004;3(8):1035-1042.
84. Yap TA, Omlin A, de Bono JS. Development of therapeutic combinations targeting major cancer signaling pathways. J Clin Oncol. 2013;31(12):1592-1605. doi:10.1200/JCO.2011.37.6418
85. Gilani B, Cassagnol M. Biochemistry, Cytochrome P450. StatPearls. Updated April 24, 2023. Accessed June 5, 2024. https://www.ncbi.nlm.nih.gov/books/NBK557698/
86. LiverTox: clinical and research information on drug-induced liver injury; 2012. Carboplatin. Updated September 15, 2020. Accessed June 5, 2024. https://www.ncbi.nlm.nih.gov/books/NBK548565/
87. Carboplatin. Prescribing information. Teva Parenteral Medicines; 2012. Accessed June 5, 204. https://www.accessdata.fda.gov/drugsatfda_docs/label/2012/077139Orig1s016lbl.pdf
88. Johnson-Arbor K, Dubey R. Doxorubicin. StatPearls. Updated August 8, 2023. Accessed June 5, 2024. https://www.ncbi.nlm.nih.gov/books/NBK459232/
89. Doxorubicin hydrochloride injection. Prescribing information. Pfizer; 2019. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2020/050467s078,050629s030lbl.pdf
90. Gor, PP, Su, HI, Gray, RJ, et al. Cyclophosphamide-metabolizing enzyme polymorphisms and survival outcomes after adjuvant chemotherapy for node-positive breast cancer: a retrospective cohort study. Breast Cancer Res. 2010;12(3):R26. doi:10.1186/bcr2570
91. Cyclophosphamide. Prescribing information. Ingenus Pharmaceuticals; 2020. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2020/212501s000lbl.pdf
92. Gemcitabine. Prescribing information. Hospira; 2019. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2019/200795Orig1s010lbl.pdf
93. Ifex (ifosfamide). Prescribing information. Baxter; 2012. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2012/019763s017lbl.pdf
94. Cisplatin. Prescribing information. WG Critical Care; 2019. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2019/018057s089lbl.pdf
95. Gerriets V, Kasi A. Bevacizumab. StatPearls. Updated August 28, 2023. Accessed June 5, 2024. https://www.ncbi.nlm.nih.gov/books/NBK482126/
96. Avastin (bevacizumab). Prescribing information. Genentech; 2022. Accessed June 5, 2024. https://www.accessdata .fda.gov/drugsatfda_docs/label/2022/125085s340lbl.pdf
 97. Keytruda (pembrolizumab). Prescribing information. Merck; 2021. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2021/125514s096lbl.pdf
97. Keytruda (pembrolizumab). Prescribing information. Merck; 2021. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2021/125514s096lbl.pdf
98. Dean L, Kane M. Capecitabine therapy and DPYD genotype. National Center for Biotechnology Information (US); 2012. Updated November 2, 2020. Accessed June 5, 2024. https://www.ncbi.nlm.nih.gov/books/NBK385155/
99. Xeloda (capecitabine). Prescribing information. Roche; 2000. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2000/20896lbl.pdf
100. Pemetrexed injection. Prescribing information. Fareva Unterach; 2022. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2022/214657s000lbl.pdf
101. Topotecan Injection. Prescribing information. Zydus Hospira Oncology; 2014. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2014/200582s001lbl.pdf
102. Ibrance (palbociclib). Prescribing information. Pfizer; 2019. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2019/207103s008lbl.pdf
103. Navelbine (vinorelbine) injection. Prescribing information. Pierre Fabre Médicament; 2020. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2020/020388s037lbl.pdf
104. LiverTox: clinical and research information on drug-induced liver injury; 2012. Letrozole. Updated July 25, 2017. Accessed June 5, 2024. https://www.ncbi.nlm.nih.gov/books/NBK548381/
105. Femara (letrozole). Prescribing information. Novartis; 2014. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2014/020726s027lbl.pdf
106. Soltamox (tamoxifen citrate). Prescribing information. Rosemont Pharmaceuticals; 2018. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2018/021807s005lbl.pdf
107. LiverTox: clinical and research information on drug-induced liver injury; 2012. Anastrozole. Updated July 25, 2017. Accessed June 5, 2024. https://www.ncbi.nlm.nih.gov/books/NBK548189/
108. Grimm SW, Dyroff MC. Inhibition of human drug metabolizing cytochromes P450 by anastrozole, a potent and selective inhibitor of aromatase. Drug Metab Dispos. 1997;25(5):598-602.
109. Arimidex (anastrozole). Prescribing information. AstraZeneca; 2010. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2011/020541s026lbl.pdf
110. Megace (megestrol acetate). Prescribing information. Endo Pharmaceuticals; 2018. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2018/021778s024lbl.pdf
111. Imfinzi (durvalumab). Prescribing information. AstraZeneca; 2020. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2020/761069s018lbl.pdf
112. Merwar G, Gibbons JR, Hosseini SA, et al. Nortriptyline. StatPearls. Updated June 5, 2023. Accessed June 5, 2024. https://www.ncbi.nlm.nih.gov/books/NBK482214/
113. Pamelor (nortriptyline HCl). Prescribing information. Patheon Inc.; 2012. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2012/018012s029,018013s061lbl.pdf
114. Wellbutrin (bupropion hydrochloride). Prescribing information. GlaxoSmithKline; 2017. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2017/018644s052lbl.pdf
115. Paxil (paroxetine). Prescribing information. Apotex Inc.; 2021. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2021/020031s077lbl.pdf
116. Johnson DB, Lopez MJ, Kelley B. Dexamethasone. StatPearls. Updated May 2, 2023. Accessed June 5, 2024. https://www.ncbi.nlm.nih.gov/books/NBK482130/
117. Hemady (dexamethasone). Prescribing information. Dexcel Pharma; 2019. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2019/211379s000lbl.pdf
118. Parker SD, King N, Jacobs TF. Pegfilgrastim. StatPearls. Updated May 9, 2024. Accessed June 5, 2024. https://www.ncbi.nlm.nih.gov/books/NBK532893/
119. Fylnetra (pegfilgrastim-pbbk). Prescribing information. Kashiv BioSciences; 2022. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2022/761084s000lbl.pdf
120. Emend (aprepitant). Prescribing information. Merck; 2015. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2015/207865lbl.pdf
121. Lipitor (atorvastatin calcium). Prescribing information. Viatris Specialty; 2022. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2022/020702Orig1s079correctedlbl.pdf
122. Cipro (ciprofloxacin hydrochloride). Prescribing information. Bayer HealthCare Pharmaceuticals Inc.; 2020. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2020/019537s090,020780s047lbl.pdf
123. Pino MA, Azer SA. Cimetidine. StatPearls. Updated March 6, 2023. Accessed June 5, 2024. https://www.ncbi.nlm.nih.gov/books/NBK544255/
124. Tagament (Cimetidine). Prescribing information. Mylan; 2020. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2020/020238Orig1s024lbl.pdf
125. Neupogen (filgrastim). Prescribing information. Amgen Inc.; 2015. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2015/103353s5184lbl.pdf
126. Flagyl (metronidazole). Prescribing information. Pfizer; 2013. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2013/020334s008lbl.pdf
127. Zymaxid (gatifloxacin ophthalmic solution). Prescribing information. Allergan; 2016. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2016/022548s002lbl.pdf
128. Macrobid (nitrofurantoin monohydrate). Prescribing information. Procter and Gamble Pharmaceutical Inc.; 2009. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2009/020064s019lbl.pdf
129. Hyzaar (losartan). Prescribing information. Merck; 2020. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2020/020387s067lbl.pdf
1. American Chemical Society. Discovery of camptothecin and taxol. acs.org. Accessed June 4, 2024. https://www.acs.org/education/whatischemistry/landmarks/camptothecintaxol.html
2. Bocci G, Di Paolo A, Danesi R. The pharmacological bases of the antiangiogenic activity of paclitaxel. Angiogenesis. 2013;16(3):481-492. doi:10.1007/s10456-013-9334-0.
3. Meštrovic T. Paclitaxel history. News Medical Life Sciences. Updated March 11, 2023. Accessed June 4, 2024. https://www.news-medical.net/health/Paclitaxel-History.aspx
4. Rowinsky EK, Donehower RC. Paclitaxel (taxol). N Engl J Med. 1995;332(15):1004-1014. doi:10.1056/NEJM199504133321507
5. Walsh V, Goodman J. The billion dollar molecule: Taxol in historical and theoretical perspective. Clio Med. 2002;66:245-267. doi:10.1163/9789004333499_013
6. Perdue RE, Jr, Hartwell JL. The search for plant sources of anticancer drugs. Morris Arboretum Bull. 1969;20:35-53.
7. Wall ME, Wani MC. Camptothecin and taxol: discovery to clinic—thirteenth Bruce F. Cain Memorial Award lecture. Cancer Res. 1995;55:753-760.
8. Wani MC, Taylor HL, Wall ME, Coggon P, McPhail AT. Plant antitumor agents. VI. The isolation and structure of taxol, a novel antileukemic and antitumor agent from taxus brevifolia. J Am Chem Soc. 1971;93(9):2325-2327. doi:10.1021/ja00738a045
9. Weaver BA. How taxol/paclitaxel kills cancer cells. Mol Biol Cell. 2014;25(18):2677-2681. doi:10.1091/mbc.E14-04-0916
10. Chen JG, Horwitz SB. Differential mitotic responses to microtubule-stabilizing and-destabilizing drugs. Cancer Res. 2002;62(7):1935-1938.
11. Singh S, Dash AK. Paclitaxel in cancer treatment: perspectives and prospects of its delivery challenges. Crit Rev Ther Drug Carrier Syst. 2009;26(4):333-372. doi:10.1615/critrevtherdrugcarriersyst.v26.i4.10
12. Schiff PB, Fant J, Horwitz SB. Promotion of microtubule assembly in vitro by taxol. Nature. 1979;277(5698):665-667. doi:10.1038/277665a0
13. Fuchs DA, Johnson RK. Cytologic evidence that taxol, an antineoplastic agent from taxus brevifolia, acts as a mitotic spindle poison. Cancer Treat Rep. 1978;62(8):1219-1222.
14. Walsh V, Goodman J. From taxol to taxol: the changing identities and ownership of an anti-cancer drug. Med Anthropol. 2002;21(3-4):307-336. doi:10.1080/01459740214074
15. Walsh V, Goodman J. Cancer chemotherapy, biodiversity, public and private property: the case of the anti-cancer drug taxol. Soc Sci Med. 1999;49(9):1215-1225. doi:10.1016/s0277-9536(99)00161-6
16. Jordan MA, Wendell K, Gardiner S, Derry WB, Copp H, Wilson L. Mitotic block induced in HeLa cells by low concentrations of paclitaxel (taxol) results in abnormal mitotic exit and apoptotic cell death. Cancer Res. 1996;56(4):816-825.
17. Picard M, Castells MC. Re-visiting hypersensitivity reactions to taxanes: a comprehensive review. Clin Rev Allergy Immunol. 2015;49(2):177-191. doi:10.1007/s12016-014-8416-0
18. Zasadil LM, Andersen KA, Yeum D, et al. Cytotoxicity of paclitaxel in breast cancer is due to chromosome missegregation on multipolar spindles. Sci Transl Med. 2014;6:229ra243. doi:10.1126/scitranslmed.3007965
19. National Cancer Institute. Carboplatin-Taxol. Published May 30, 2012. Updated March 22, 2023. Accessed June 4, 2024. https://www.cancer.gov/about-cancer/treatment/drugs/carboplatin-taxol
20. Taxol (paclitaxel). Prescribing information. Bristol-Myers Squibb; 2011. Accessed June 4, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2011/020262s049lbl.pdf
21. Abraxane (paclitaxel). Prescribing information. Celgene Corporation; 2021. Accessed June 4, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2020/021660s047lbl.pdf
22. Awosika AO, Farrar MC, Jacobs TF. Paclitaxel. StatPearls. Updated November 18, 2023. Accessed June 5, 2024. https://www.ncbi.nlm.nih.gov/books/NBK536917/
23. Gerriets V, Kasi A. Bevacizumab. StatPearls. Updated September 1, 2022. Accessed June 5, 2024. https://www.ncbi.nlm.nih.gov/books/NBK482126/
24. American Cancer Society. Chemotherapy for endometrial cancer. Updated March 27, 2019. Accessed June 4, 2024. https://www.cancer.org/cancer/types/endometrial-cancer/treating/chemotherapy.html
25. US Food and Drug Administration. FDA approves pembrolizumab in combination with chemotherapy for first-line treatment of metastatic squamous NSCLC. October 30, 2018. Updated December 14, 2018. Accessed June 4, 2024. https://www.fda.gov/drugs/fda-approves-pembrolizumab-combination-chemotherapy-first-line-treatment-metastatic-squamous-nsclc
26. US Food and Drug Administration. FDA grants accelerated approval to pembrolizumab for locally recurrent unresectable or metastatic triple negative breast cancer. November 13, 2020. Accessed June 4, 2024. https://www.fda.gov/drugs/resources-information-approved-drugs/fda-grants-accelerated-approval-pembrolizumab-locally-recurrent-unresectable-or-metastatic-triple
27. US Food and Drug Administration. FDA approves atezolizumab for PD-L1 positive unresectable locally advanced or metastatic triple-negative breast. March 8, 2019. Updated March 18, 2019. Accessed June 5, 2024. https://www.fda.gov/drugs/drug-approvals-and-databases/fda-approves-atezolizumab-pd-l1-positive-unresectable-locally-advanced-or-metastatic-triple-negative
28. US Food and Drug Administration. FDA issues alert about efficacy and potential safety concerns with atezolizumab in combination with paclitaxel for treatment of breast cancer. September 8, 2020. Accessed June 5, 2024. https://www.fda.gov/drugs/resources-information-approved-drugs/fda-issues-alert-about-efficacy-and-potential-safety-concerns-atezolizumab-combination-paclitaxel
29. Tan AR. Chemoimmunotherapy: still the standard of care for metastatic triple-negative breast cancer. ASCO Daily News. February 23, 2022. Accessed June 5, 2024. https://dailynews.ascopubs.org/do/chemoimmunotherapy-still-standard-care-metastatic-triple-negative-breast-cancer
30. McGuire WP, Rowinsky EK, Rosenshein NB, et al. Taxol: a unique antineoplastic agent with significant activity in advanced ovarian epithelial neoplasms. Ann Intern Med. 1989;111(4):273-279. doi:10.7326/0003-4819-111-4-273
31. Milas L, Hunter NR, Kurdoglu B, et al. Kinetics of mitotic arrest and apoptosis in murine mammary and ovarian tumors treated with taxol. Cancer Chemother Pharmacol. 1995;35(4):297-303. doi:10.1007/BF00689448
32. Searle J, Collins DJ, Harmon B, Kerr JF. The spontaneous occurrence of apoptosis in squamous carcinomas of the uterine cervix. Pathology. 1973;5(2):163-169. doi:10.3109/00313027309060831
33. Gallego-Jara J, Lozano-Terol G, Sola-Martínez RA, Cánovas-Díaz M, de Diego Puente T. A compressive review about taxol®: history and future challenges. Molecules. 2020;25(24):5986. doi:10.3390/molecules25245986
34. Bernabeu E, Cagel M, Lagomarsino E, Moretton M, Chiappetta DA. Paclitaxel: What has been done and the challenges remain ahead. Int J Pharm. 2017;526(1-2):474-495. doi:10.1016/j.ijpharm.2017.05.016
35. Nehate C, Jain S, Saneja A, et al. Paclitaxel formulations: challenges and novel delivery options. Curr Drug Deliv. 2014;11(6):666-686. doi:10.2174/1567201811666140609154949
36. Gelderblom H, Verweij J, Nooter K, Sparreboom A, Cremophor EL. The drawbacks and advantages of vehicle selection for drug formulation. Eur J Cancer. 2001;37(13):1590-1598. doi:10.1016/S0959-8049(01)00171-x
37. Chowdhury MR, Moshikur RM, Wakabayashi R, et al. In vivo biocompatibility, pharmacokinetics, antitumor efficacy, and hypersensitivity evaluation of ionic liquid-mediated paclitaxel formulations. Int J Pharm. 2019;565:219-226. doi:10.1016/j.ijpharm.2019.05.020
38. Borgå O, Henriksson R, Bjermo H, Lilienberg E, Heldring N, Loman N. Maximum tolerated dose and pharmacokinetics of paclitaxel micellar in patients with recurrent malignant solid tumours: a dose-escalation study. Adv Ther. 2019;36(5):1150-1163. doi:10.1007/s12325-019-00909-6
39. Rouzier R, Rajan R, Wagner P, et al. Microtubule-associated protein tau: a marker of paclitaxel sensitivity in breast cancer. Proc Natl Acad Sci USA. 2005;102(23):8315-8320. doi:10.1073/pnas.0408974102
40. Choudhury H, Gorain B, Tekade RK, Pandey M, Karmakar S, Pal TK. Safety against nephrotoxicity in paclitaxel treatment: oral nanocarrier as an effective tool in preclinical evaluation with marked in vivo antitumor activity. Regul Toxicol Pharmacol. 2017;91:179-189. doi:10.1016/j.yrtph.2017.10.023
41. Barkat MA, Beg S, Pottoo FH, Ahmad FJ. Nanopaclitaxel therapy: an evidence based review on the battle for next-generation formulation challenges. Nanomedicine (Lond). 2019;14(10):1323-1341. doi:10.2217/nnm-2018-0313
42. Sofias AM, Dunne M, Storm G, Allen C. The battle of “nano” paclitaxel. Adv Drug Deliv Rev. 2017;122:20-30. doi:10.1016/j.addr.2017.02.003
43. Yang N, Wang C, Wang J, et al. Aurora inase a stabilizes FOXM1 to enhance paclitaxel resistance in triple-negative breast cancer. J Cell Mol Med. 2019;23(9):6442-6453. doi:10.1111/jcmm.14538
44. Chowdhury MR, Moshikur RM, Wakabayashi R, et al. Ionic-liquid-based paclitaxel preparation: a new potential formulation for cancer treatment. Mol Pharm. 2018;15(16):2484-2488. doi:10.1021/acs.molpharmaceut.8b00305
45. Chung HJ, Kim HJ, Hong ST. Tumor-specific delivery of a paclitaxel-loading HSA-haemin nanoparticle for cancer treatment. Nanomedicine. 2020;23:102089. doi:10.1016/j.nano.2019.102089
46. Ye L, He J, Hu Z, et al. Antitumor effect and toxicity of lipusu in rat ovarian cancer xenografts. Food Chem Toxicol. 2013;52:200-206. doi:10.1016/j.fct.2012.11.004
47. Ma WW, Lam ET, Dy GK, et al. A pharmacokinetic and dose-escalating study of paclitaxel injection concentrate for nano-dispersion (PICN) alone and with arboplatin in patients with advanced solid tumors. J Clin Oncol. 2013;31:2557. doi:10.1200/jco.2013.31.15_suppl.2557
48. Micha JP, Goldstein BH, Birk CL, Rettenmaier MA, Brown JV. Abraxane in the treatment of ovarian cancer: the absence of hypersensitivity reactions. Gynecol Oncol. 2006;100(2):437-438. doi:10.1016/j.ygyno.2005.09.012
49. Ingle SG, Pai RV, Monpara JD, Vavia PR. Liposils: an effective strategy for stabilizing paclitaxel loaded liposomes by surface coating with silica. Eur J Pharm Sci. 2018;122:51-63. doi:10.1016/j.ejps.2018.06.025
50. Abriata JP, Turatti RC, Luiz MT, et al. Development, characterization and biological in vitro assays of paclitaxel-loaded PCL polymeric nanoparticles. Mater Sci Eng C Mater Biol Appl. 2019;96:347-355. doi:10.1016/j.msec.2018.11.035
51. Hu J, Fu S, Peng Q, et al. Paclitaxel-loaded polymeric nanoparticles combined with chronomodulated chemotherapy on lung cancer: in vitro and in vivo evaluation. Int J Pharm. 2017;516(1-2):313-322. doi:10.1016/j.ijpharm.2016.11.047
52. Dranitsaris G, Yu B, Wang L, et al. Abraxane® vs Taxol® for patients with advanced breast cancer: a prospective time and motion analysis from a chinese health care perspective. J Oncol Pharm Pract. 2016;22(2):205-211. doi:10.1177/1078155214556008
53. Pei Q, Hu X, Liu S, Li Y, Xie Z, Jing X. Paclitaxel dimers assembling nanomedicines for treatment of cervix carcinoma. J Control Release. 2017;254:23-33. doi:10.1016/j.jconrel.2017.03.391
54. Wang Y, Wang M, Qi H, et al. Pathway-dependent inhibition of paclitaxel hydroxylation by kinase inhibitors and assessment of drug-drug interaction potentials. Drug Metab Dispos. 2014;42(4):782-795. doi:10.1124/dmd.113.053793
55. Shen F, Jiang G, Philips S, et al. Cytochrome P450 oxidoreductase (POR) associated with severe paclitaxel-induced peripheral neuropathy in patients of european ancestry from ECOG-ACRIN E5103. Clin Cancer Res. 2023;29(13):2494-2500. doi:10.1158/1078-0432.CCR-22-2431
56. Henningsson A, Marsh S, Loos WJ, et al. Association of CYP2C8, CYP3A4, CYP3A5, and ABCB1 polymorphisms with the pharmacokinetics of paclitaxel. Clin Cancer Res. 2005;11(22):8097-8104. doi:10.1158/1078-0432.CCR-05-1152
57. Mukai Y, Senda A, Toda T, et al. Drug-drug interaction between losartan and paclitaxel in human liver microsomes with different CYP2C8 genotypes. Basic Clin Pharmacol Toxicol. 2015;116(6):493-498. doi:10.1111/bcpt.12355
58. Kawahara B, Faull KF, Janzen C, Mascharak PK. Carbon monoxide inhibits cytochrome P450 enzymes CYP3A4/2C8 in human breast cancer cells, increasing sensitivity to paclitaxel. J Med Chem. 2021;64(12):8437-8446. doi:10.1021/acs.jmedchem.1c00404
59. Cresteil T, Monsarrat B, Dubois J, Sonnier M, Alvinerie P, Gueritte F. Regioselective metabolism of taxoids by human CYP3A4 and 2C8: structure-activity relationship. Drug Metab Dispos. 2002;30(4):438-445. doi:10.1124/dmd.30.4.438
60. Taniguchi R, Kumai T, Matsumoto N, et al. Utilization of human liver microsomes to explain individual differences in paclitaxel metabolism by CYP2C8 and CYP3A4. J Pharmacol Sci. 2005;97(1):83-90. doi:10.1254/jphs.fp0040603
61. Nakayama A, Tsuchiya K, Xu L, Matsumoto T, Makino T. Drug-interaction between paclitaxel and goshajinkigan extract and its constituents. J Nat Med. 2022;76(1):59-67. doi:10.1007/s11418-021-01552-8
62. Monsarrat B, Chatelut E, Royer I, et al. Modification of paclitaxel metabolism in a cancer patient by induction of cytochrome P450 3A4. Drug Metab Dispos. 1998;26(3):229-233.
63. Walle T. Assays of CYP2C8- and CYP3A4-mediated metabolism of taxol in vivo and in vitro. Methods Enzymol. 1996;272:145-151. doi:10.1016/s0076-6879(96)72018-9
64. Hanioka N, Matsumoto K, Saito Y, Narimatsu S. Functional characterization of CYP2C8.13 and CYP2C8.14: catalytic activities toward paclitaxel. Basic Clin Pharmacol Toxicol. 2010;107(1):565-569. doi:10.1111/j.1742-7843.2010.00543.x
65. Luong TT, Powers CN, Reinhardt BJ, Weina PJ. Pre-clinical drug-drug interactions (DDIs) of gefitinib with/without losartan and selective serotonin reuptake inhibitors (SSRIs): citalopram, fluoxetine, fluvoxamine, paroxetine, sertraline, and venlafaxine. Curr Res Pharmacol Drug Discov. 2022;3:100112. doi:10.1016/j.crphar.2022.100112
66. Luong TT, McAnulty MJ, Evers DL, Reinhardt BJ, Weina PJ. Pre-clinical drug-drug interaction (DDI) of gefitinib or erlotinib with Cytochrome P450 (CYP) inhibiting drugs, fluoxetine and/or losartan. Curr Res Toxicol. 2021;2:217-224. doi:10.1016/j.crtox.2021.05.006
67. Luong TT, Powers CN, Reinhardt BJ, et al. Retrospective evaluation of drug-drug interactions with erlotinib and gefitinib use in the military health system. Fed Pract. 2023;40(suppl 3):S24-S34. doi:10.12788/fp.0401
68. Adamo M, Dickie L, Ruhl J. SEER program coding and staging manual 2016. National Cancer Institute. Accessed June 5, 2024. https://seer.cancer.gov/archive/manuals/2016/SPCSM_2016_maindoc.pdf
69. World Health Organization. International classification of diseases for oncology (ICD-O) 3rd ed, 1st revision. World Health Organization; 2013. Accessed June 5, 2024. https://apps.who.int/iris/handle/10665/96612
70. Z score calculator for 2 population proportions. Social science statistics. Accessed June 5, 2024. https://www.socscistatistics.com/tests/ztest/default2.aspx
71. US Food and Drug Administration. Generic drugs: question & answers. FDA.gov. Accessed June 5, 2024. https://www.fda.gov/drugs/frequently-asked-questions-popular-topics/generic-drugs-questions-answers
72. Oura M, Saito H, Nishikawa Y. Shortage of nab-paclitaxel in Japan and around the world: issues in global information sharing. JMA J. 2023;6(2):192-195. doi:10.31662/jmaj.2022-0179
73. Yuan H, Guo H, Luan X, et al. Albumin nanoparticle of paclitaxel (abraxane) decreases while taxol increases breast cancer stem cells in treatment of triple negative breast cancer. Mol Pharm. 2020;17(7):2275-2286. doi:10.1021/acs.molpharmaceut.9b01221
74. Dranitsaris G, Yu B, Wang L, et al. Abraxane® versus Taxol® for patients with advanced breast cancer: a prospective time and motion analysis from a Chinese health care perspective. J Oncol Pharm Pract. 2016;22(2):205-211. doi:10.1177/1078155214556008
75. Gradishar WJ, Tjulandin S, Davidson N, et al. Phase III trial of nanoparticle albumin-bound paclitaxel compared with polyethylated castor oil-based paclitaxel in women with breast cancer. J Clin Oncol. 2005;23(31):7794-7803. doi:10.1200/JCO.2005.04.
76. Liu M, Liu S, Yang L, Wang S. Comparison between nab-paclitaxel and solvent-based taxanes as neoadjuvant therapy in breast cancer: a systematic review and meta-analysis. BMC Cancer. 2021;21(1):118. doi:10.1186/s12885-021-07831-7
77. Rowinsky EK, Eisenhauer EA, Chaudhry V, Arbuck SG, Donehower RC. Clinical toxicities encountered with paclitaxel (taxol). Semin Oncol. 1993;20(4 Suppl 3):1-15.
78. Banerji A, Lax T, Guyer A, Hurwitz S, Camargo CA Jr, Long AA. Management of hypersensitivity reactions to carboplatin and paclitaxel in an outpatient oncology infusion center: a 5-year review. J Allergy Clin Immunol Pract. 2014;2(4):428-433. doi:10.1016/j.jaip.2014.04.010
79. Staff NP, Fehrenbacher JC, Caillaud M, Damaj MI, Segal RA, Rieger S. Pathogenesis of paclitaxel-induced peripheral neuropathy: a current review of in vitro and in vivo findings using rodent and human model systems. Exp Neurol. 2020;324:113121. doi:10.1016/j.expneurol.2019.113121
80. Postma TJ, Vermorken JB, Liefting AJ, Pinedo HM, Heimans JJ. Paclitaxel-induced neuropathy. Ann Oncol. 1995;6(5):489-494. doi:10.1093/oxfordjournals.annonc.a059220
81. Liu JM, Chen YM, Chao Y, et al. Paclitaxel-induced severe neuropathy in patients with previous radiotherapy to the head and neck region. J Natl Cancer Inst. 1996;88(14):1000-1002. doi:10.1093/jnci/88.14.1000-a
82. Bayat Mokhtari R, Homayouni TS, Baluch N, et al. Combination therapy in combating cancer. Oncotarget. 2017;8(23):38022-38043. doi:10.18632/oncotarget.16723
83. Blagosklonny MV. Analysis of FDA approved anticancer drugs reveals the future of cancer therapy. Cell Cycle. 2004;3(8):1035-1042.
84. Yap TA, Omlin A, de Bono JS. Development of therapeutic combinations targeting major cancer signaling pathways. J Clin Oncol. 2013;31(12):1592-1605. doi:10.1200/JCO.2011.37.6418
85. Gilani B, Cassagnol M. Biochemistry, Cytochrome P450. StatPearls. Updated April 24, 2023. Accessed June 5, 2024. https://www.ncbi.nlm.nih.gov/books/NBK557698/
86. LiverTox: clinical and research information on drug-induced liver injury; 2012. Carboplatin. Updated September 15, 2020. Accessed June 5, 2024. https://www.ncbi.nlm.nih.gov/books/NBK548565/
87. Carboplatin. Prescribing information. Teva Parenteral Medicines; 2012. Accessed June 5, 204. https://www.accessdata.fda.gov/drugsatfda_docs/label/2012/077139Orig1s016lbl.pdf
88. Johnson-Arbor K, Dubey R. Doxorubicin. StatPearls. Updated August 8, 2023. Accessed June 5, 2024. https://www.ncbi.nlm.nih.gov/books/NBK459232/
89. Doxorubicin hydrochloride injection. Prescribing information. Pfizer; 2019. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2020/050467s078,050629s030lbl.pdf
90. Gor, PP, Su, HI, Gray, RJ, et al. Cyclophosphamide-metabolizing enzyme polymorphisms and survival outcomes after adjuvant chemotherapy for node-positive breast cancer: a retrospective cohort study. Breast Cancer Res. 2010;12(3):R26. doi:10.1186/bcr2570
91. Cyclophosphamide. Prescribing information. Ingenus Pharmaceuticals; 2020. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2020/212501s000lbl.pdf
92. Gemcitabine. Prescribing information. Hospira; 2019. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2019/200795Orig1s010lbl.pdf
93. Ifex (ifosfamide). Prescribing information. Baxter; 2012. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2012/019763s017lbl.pdf
94. Cisplatin. Prescribing information. WG Critical Care; 2019. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2019/018057s089lbl.pdf
95. Gerriets V, Kasi A. Bevacizumab. StatPearls. Updated August 28, 2023. Accessed June 5, 2024. https://www.ncbi.nlm.nih.gov/books/NBK482126/
96. Avastin (bevacizumab). Prescribing information. Genentech; 2022. Accessed June 5, 2024. https://www.accessdata .fda.gov/drugsatfda_docs/label/2022/125085s340lbl.pdf
 97. Keytruda (pembrolizumab). Prescribing information. Merck; 2021. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2021/125514s096lbl.pdf
97. Keytruda (pembrolizumab). Prescribing information. Merck; 2021. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2021/125514s096lbl.pdf
98. Dean L, Kane M. Capecitabine therapy and DPYD genotype. National Center for Biotechnology Information (US); 2012. Updated November 2, 2020. Accessed June 5, 2024. https://www.ncbi.nlm.nih.gov/books/NBK385155/
99. Xeloda (capecitabine). Prescribing information. Roche; 2000. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2000/20896lbl.pdf
100. Pemetrexed injection. Prescribing information. Fareva Unterach; 2022. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2022/214657s000lbl.pdf
101. Topotecan Injection. Prescribing information. Zydus Hospira Oncology; 2014. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2014/200582s001lbl.pdf
102. Ibrance (palbociclib). Prescribing information. Pfizer; 2019. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2019/207103s008lbl.pdf
103. Navelbine (vinorelbine) injection. Prescribing information. Pierre Fabre Médicament; 2020. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2020/020388s037lbl.pdf
104. LiverTox: clinical and research information on drug-induced liver injury; 2012. Letrozole. Updated July 25, 2017. Accessed June 5, 2024. https://www.ncbi.nlm.nih.gov/books/NBK548381/
105. Femara (letrozole). Prescribing information. Novartis; 2014. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2014/020726s027lbl.pdf
106. Soltamox (tamoxifen citrate). Prescribing information. Rosemont Pharmaceuticals; 2018. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2018/021807s005lbl.pdf
107. LiverTox: clinical and research information on drug-induced liver injury; 2012. Anastrozole. Updated July 25, 2017. Accessed June 5, 2024. https://www.ncbi.nlm.nih.gov/books/NBK548189/
108. Grimm SW, Dyroff MC. Inhibition of human drug metabolizing cytochromes P450 by anastrozole, a potent and selective inhibitor of aromatase. Drug Metab Dispos. 1997;25(5):598-602.
109. Arimidex (anastrozole). Prescribing information. AstraZeneca; 2010. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2011/020541s026lbl.pdf
110. Megace (megestrol acetate). Prescribing information. Endo Pharmaceuticals; 2018. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2018/021778s024lbl.pdf
111. Imfinzi (durvalumab). Prescribing information. AstraZeneca; 2020. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2020/761069s018lbl.pdf
112. Merwar G, Gibbons JR, Hosseini SA, et al. Nortriptyline. StatPearls. Updated June 5, 2023. Accessed June 5, 2024. https://www.ncbi.nlm.nih.gov/books/NBK482214/
113. Pamelor (nortriptyline HCl). Prescribing information. Patheon Inc.; 2012. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2012/018012s029,018013s061lbl.pdf
114. Wellbutrin (bupropion hydrochloride). Prescribing information. GlaxoSmithKline; 2017. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2017/018644s052lbl.pdf
115. Paxil (paroxetine). Prescribing information. Apotex Inc.; 2021. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2021/020031s077lbl.pdf
116. Johnson DB, Lopez MJ, Kelley B. Dexamethasone. StatPearls. Updated May 2, 2023. Accessed June 5, 2024. https://www.ncbi.nlm.nih.gov/books/NBK482130/
117. Hemady (dexamethasone). Prescribing information. Dexcel Pharma; 2019. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2019/211379s000lbl.pdf
118. Parker SD, King N, Jacobs TF. Pegfilgrastim. StatPearls. Updated May 9, 2024. Accessed June 5, 2024. https://www.ncbi.nlm.nih.gov/books/NBK532893/
119. Fylnetra (pegfilgrastim-pbbk). Prescribing information. Kashiv BioSciences; 2022. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2022/761084s000lbl.pdf
120. Emend (aprepitant). Prescribing information. Merck; 2015. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2015/207865lbl.pdf
121. Lipitor (atorvastatin calcium). Prescribing information. Viatris Specialty; 2022. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2022/020702Orig1s079correctedlbl.pdf
122. Cipro (ciprofloxacin hydrochloride). Prescribing information. Bayer HealthCare Pharmaceuticals Inc.; 2020. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2020/019537s090,020780s047lbl.pdf
123. Pino MA, Azer SA. Cimetidine. StatPearls. Updated March 6, 2023. Accessed June 5, 2024. https://www.ncbi.nlm.nih.gov/books/NBK544255/
124. Tagament (Cimetidine). Prescribing information. Mylan; 2020. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2020/020238Orig1s024lbl.pdf
125. Neupogen (filgrastim). Prescribing information. Amgen Inc.; 2015. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2015/103353s5184lbl.pdf
126. Flagyl (metronidazole). Prescribing information. Pfizer; 2013. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2013/020334s008lbl.pdf
127. Zymaxid (gatifloxacin ophthalmic solution). Prescribing information. Allergan; 2016. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2016/022548s002lbl.pdf
128. Macrobid (nitrofurantoin monohydrate). Prescribing information. Procter and Gamble Pharmaceutical Inc.; 2009. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2009/020064s019lbl.pdf
129. Hyzaar (losartan). Prescribing information. Merck; 2020. Accessed June 5, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2020/020387s067lbl.pdf
‘Alarming’ Rise in Mental Health Hospital Admissions Involving Methamphetamine
new research showed. Investigators found that between 2008 and 2020, such admissions increased by more than 10-fold.
“Overall, our results show an alarming increase in mental health disorder–related hospitalizations with concurrent methamphetamine use from 2008 to 2020,” wrote the investigators, led by Diensn Xing, Department of Medicine, Louisiana State University Health Sciences Center, Shreveport.
“These results are especially concerning because these hospitalizations outpace hospitalizations for methamphetamine use alone or mental health disorders alone,” they added.
The study was published online in Nature Mental Health .
Action Needed
Mental illness and methamphetamine use are both growing health problems. The investigators pointed out that methamphetamine use can cause serious harm to an individual’s mental, emotional, and social well-being and can significantly alter the brain.
They added that long-term methamphetamine users can exhibit “extreme anxiety, confusion, troubled sleep, mood changes, and aggressive behavior.” In addition, use of the drug can cause psychotic side effects such as paranoia, hallucinations, delusions, and suicidality.
The investigators noted that, to date, no studies have examined the combined effects of both diseases or characterized national trends over more than 10 years.
The researchers analyzed US mental health–related trends in methamphetamine users from 2008 to 2020. In particular, they wanted to characterize which demographic and geographic groups might be affected by both of these diseases because people with mental illness and co-occurring methamphetamine use are an “intersectional group” that is “doubly vulnerable to suicide and overdose death due to the synergistic effects of methamphetamine and mental health disorders.”
The investigators evaluated US trends in mental health disorder–related hospital admissions (MHD-HAs) and compared them with mental health admissions that involved concurrent methamphetamine use (MHD-HA-MUs) between 2008 and 2020.
Using data from the largest US inpatient care database, which encompasses more than 7 million hospital stays annually, they examined close to 4 million weighted hospital admissions and found more than a 10-fold increase in MHD-HA-MUs, compared with a 1.4-fold increase in MHD-HAs.
MHD-HA-MUs increased significantly among men (13-fold), non-Hispanic Black patients (39-fold), and those aged 41-64 years (16-fold). In the southern United States, MHD-HA-MUs increased 24-fold, larger than in any other region in the United States.
“Overall, the data suggest that there are synergistic effects with methamphetamine use and mental health disorder, highlighting this patient group’s unique needs, requiring distinct action,” the researchers wrote.
They proposed several interventions, including public education about substance use disorders, mental illness, and the effects of stigma. They also suggested decreasing criminal penalties for those with substance use disorders and improving healthcare delivery for this patient population.
This work was supported by the National Institutes of Health and an award from the National Institute of General Medical Sciences. The study authors declared no relevant financial relationships.
A version of this article first appeared on Medscape.com.
new research showed. Investigators found that between 2008 and 2020, such admissions increased by more than 10-fold.
“Overall, our results show an alarming increase in mental health disorder–related hospitalizations with concurrent methamphetamine use from 2008 to 2020,” wrote the investigators, led by Diensn Xing, Department of Medicine, Louisiana State University Health Sciences Center, Shreveport.
“These results are especially concerning because these hospitalizations outpace hospitalizations for methamphetamine use alone or mental health disorders alone,” they added.
The study was published online in Nature Mental Health .
Action Needed
Mental illness and methamphetamine use are both growing health problems. The investigators pointed out that methamphetamine use can cause serious harm to an individual’s mental, emotional, and social well-being and can significantly alter the brain.
They added that long-term methamphetamine users can exhibit “extreme anxiety, confusion, troubled sleep, mood changes, and aggressive behavior.” In addition, use of the drug can cause psychotic side effects such as paranoia, hallucinations, delusions, and suicidality.
The investigators noted that, to date, no studies have examined the combined effects of both diseases or characterized national trends over more than 10 years.
The researchers analyzed US mental health–related trends in methamphetamine users from 2008 to 2020. In particular, they wanted to characterize which demographic and geographic groups might be affected by both of these diseases because people with mental illness and co-occurring methamphetamine use are an “intersectional group” that is “doubly vulnerable to suicide and overdose death due to the synergistic effects of methamphetamine and mental health disorders.”
The investigators evaluated US trends in mental health disorder–related hospital admissions (MHD-HAs) and compared them with mental health admissions that involved concurrent methamphetamine use (MHD-HA-MUs) between 2008 and 2020.
Using data from the largest US inpatient care database, which encompasses more than 7 million hospital stays annually, they examined close to 4 million weighted hospital admissions and found more than a 10-fold increase in MHD-HA-MUs, compared with a 1.4-fold increase in MHD-HAs.
MHD-HA-MUs increased significantly among men (13-fold), non-Hispanic Black patients (39-fold), and those aged 41-64 years (16-fold). In the southern United States, MHD-HA-MUs increased 24-fold, larger than in any other region in the United States.
“Overall, the data suggest that there are synergistic effects with methamphetamine use and mental health disorder, highlighting this patient group’s unique needs, requiring distinct action,” the researchers wrote.
They proposed several interventions, including public education about substance use disorders, mental illness, and the effects of stigma. They also suggested decreasing criminal penalties for those with substance use disorders and improving healthcare delivery for this patient population.
This work was supported by the National Institutes of Health and an award from the National Institute of General Medical Sciences. The study authors declared no relevant financial relationships.
A version of this article first appeared on Medscape.com.
new research showed. Investigators found that between 2008 and 2020, such admissions increased by more than 10-fold.
“Overall, our results show an alarming increase in mental health disorder–related hospitalizations with concurrent methamphetamine use from 2008 to 2020,” wrote the investigators, led by Diensn Xing, Department of Medicine, Louisiana State University Health Sciences Center, Shreveport.
“These results are especially concerning because these hospitalizations outpace hospitalizations for methamphetamine use alone or mental health disorders alone,” they added.
The study was published online in Nature Mental Health .
Action Needed
Mental illness and methamphetamine use are both growing health problems. The investigators pointed out that methamphetamine use can cause serious harm to an individual’s mental, emotional, and social well-being and can significantly alter the brain.
They added that long-term methamphetamine users can exhibit “extreme anxiety, confusion, troubled sleep, mood changes, and aggressive behavior.” In addition, use of the drug can cause psychotic side effects such as paranoia, hallucinations, delusions, and suicidality.
The investigators noted that, to date, no studies have examined the combined effects of both diseases or characterized national trends over more than 10 years.
The researchers analyzed US mental health–related trends in methamphetamine users from 2008 to 2020. In particular, they wanted to characterize which demographic and geographic groups might be affected by both of these diseases because people with mental illness and co-occurring methamphetamine use are an “intersectional group” that is “doubly vulnerable to suicide and overdose death due to the synergistic effects of methamphetamine and mental health disorders.”
The investigators evaluated US trends in mental health disorder–related hospital admissions (MHD-HAs) and compared them with mental health admissions that involved concurrent methamphetamine use (MHD-HA-MUs) between 2008 and 2020.
Using data from the largest US inpatient care database, which encompasses more than 7 million hospital stays annually, they examined close to 4 million weighted hospital admissions and found more than a 10-fold increase in MHD-HA-MUs, compared with a 1.4-fold increase in MHD-HAs.
MHD-HA-MUs increased significantly among men (13-fold), non-Hispanic Black patients (39-fold), and those aged 41-64 years (16-fold). In the southern United States, MHD-HA-MUs increased 24-fold, larger than in any other region in the United States.
“Overall, the data suggest that there are synergistic effects with methamphetamine use and mental health disorder, highlighting this patient group’s unique needs, requiring distinct action,” the researchers wrote.
They proposed several interventions, including public education about substance use disorders, mental illness, and the effects of stigma. They also suggested decreasing criminal penalties for those with substance use disorders and improving healthcare delivery for this patient population.
This work was supported by the National Institutes of Health and an award from the National Institute of General Medical Sciences. The study authors declared no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM NATURE MENTAL HEALTH
Accelerated Brain Stimulation Rapidly Curbs Resistant Bipolar Depression
the results of a small randomized controlled trial showed.
Investigators found those who received active aiTBS had a substantial decrease in depressive symptoms compared with those who received sham stimulation.
“aiTBS offers a new potential therapy for depressed patients with bipolar disorder who may not respond well to drugs or cannot tolerate their side effects while also significantly shortening the treatment window,” lead researcher Yvette Sheline, MD, director of the Center for Neuromodulation in Depression and Stress at the University of Pennsylvania, Philadelphia, said in a news release.
The study was published online in JAMA Psychiatry.
Remission After 5 Days
The Food and Drug Administration (FDA) cleared aiTBS to treat major depressive disorder. However, the treatment is not yet approved for bipolar depression.
The investigators assessed the effectiveness of aiTBS in 12 men and 12 women (mean age, 43 years) with treatment-resistant bipolar disorder. All participants were on mood stabilizers for at least 4 weeks and had Montgomery-Åsberg Depression Rating Scale (MADRS) scores of 20 or greater.
Of the 24 participants, 22 had a diagnosis of bipolar II disorder. Over 5 days, participants were randomized to receive, on a 1:1 basis, 10 sessions per day of imaging-guided active aiTBS or sham aiTBS over the left dorsolateral prefrontal cortex. Each session lasted for 1 hour. All 24 participants completed the assigned treatment and 1-month follow-up.
Active aiTBS was significantly more effective than sham stimulation in relieving depressive symptoms.
In the active treatment group, MADRS scores dropped from a mean of 30.4 at baseline to 10.5 after treatment. In contrast, the sham group experienced a minor change in MADRS scores, which decreased from a mean of 28.0 at baseline to 25.3 posttreatment.
After 5 days of treatment, half of the participants in the active aiTBS group were in remission, compared with none in the sham group.
The results demonstrate the “clinical efficacy and a short time to achieve improvement in this difficult-to-treat condition. The effect was seen even though the participant sample had high depression severity and treatment resistance, both associated with poor response,” the investigators noted.
Dr. Sheline said there were no differences between active and sham stimulation in the “expected adverse events of headache and dizziness. There were no incidences of manic “flip,” and the Young Mania Rating Scale scores were stable in both active and sham groups with no difference between them.”
The researchers noted that the “large effect size” of active aiTBS could be caused by several factors, including optimized stimulation targeting, accelerated time course, and high pulse number (18,000 per day, 90,000 total).
Future studies are needed to examine the relative contributions of the different protocol components to optimize and personalize treatment and evaluate the durability of the antidepressant effects of aiTBS, they added.
A Revolutionary Approach
For comment on the study, we reached out to Nolan Williams, MD, associate professor of psychiatry and behavioral sciences at Stanford University and director of the Stanford Brain Stimulation Lab, Stanford, California.
His laboratory pioneered the Stanford Accelerated Intelligent Neuromodulation Therapy (SAINT), which was cleared by the FDA in 2022 for treatment-resistant depression.
Dr. Williams noted that the stimulation and targeting approach used in the current study “mirrors most aspects of the SAINT protocol with very similar results.”
“It’s exciting that we see this kind of pseudo-replication essentially of our work and is supportive of the general view that this approach is revolutionary in its ability to treat people quickly and have such a dramatic clinical effect,” said Dr. Williams.
In March 2024, Dr. Williams and colleagues reported the results of a pilot study of SAINT for bipolar depression, which demonstrated antidepressant efficacy akin to what was observed in the unipolar depression population.
Dr. Williams said, in his experience, the accelerated treatment protocol is convenient and well-liked by patients and, in general, is where the field of psychiatric treatment is headed.
“A general theme that we see in depression and psychiatry is that patients no longer accept long time frames for treatment as being the norm. Whether it be ketamine or this or the upcoming psychedelics, rapid-acting treatments that match the level of acuity will be the norm,” Dr. Williams said.
The study was funded by the Milken Institute and the Baszucki Brain Research Fund. The authors have disclosed no conflicts of interest. Dr. Williams is a named inventor on Stanford-owned intellectual property relating to accelerated TMS pulse pattern sequences and neuroimaging-based TMS targeting. He disclosed ties with Otsuka, NeuraWell, Magnus Medical, and Nooma.
A version of this article first appeared on Medscape.com.
the results of a small randomized controlled trial showed.
Investigators found those who received active aiTBS had a substantial decrease in depressive symptoms compared with those who received sham stimulation.
“aiTBS offers a new potential therapy for depressed patients with bipolar disorder who may not respond well to drugs or cannot tolerate their side effects while also significantly shortening the treatment window,” lead researcher Yvette Sheline, MD, director of the Center for Neuromodulation in Depression and Stress at the University of Pennsylvania, Philadelphia, said in a news release.
The study was published online in JAMA Psychiatry.
Remission After 5 Days
The Food and Drug Administration (FDA) cleared aiTBS to treat major depressive disorder. However, the treatment is not yet approved for bipolar depression.
The investigators assessed the effectiveness of aiTBS in 12 men and 12 women (mean age, 43 years) with treatment-resistant bipolar disorder. All participants were on mood stabilizers for at least 4 weeks and had Montgomery-Åsberg Depression Rating Scale (MADRS) scores of 20 or greater.
Of the 24 participants, 22 had a diagnosis of bipolar II disorder. Over 5 days, participants were randomized to receive, on a 1:1 basis, 10 sessions per day of imaging-guided active aiTBS or sham aiTBS over the left dorsolateral prefrontal cortex. Each session lasted for 1 hour. All 24 participants completed the assigned treatment and 1-month follow-up.
Active aiTBS was significantly more effective than sham stimulation in relieving depressive symptoms.
In the active treatment group, MADRS scores dropped from a mean of 30.4 at baseline to 10.5 after treatment. In contrast, the sham group experienced a minor change in MADRS scores, which decreased from a mean of 28.0 at baseline to 25.3 posttreatment.
After 5 days of treatment, half of the participants in the active aiTBS group were in remission, compared with none in the sham group.
The results demonstrate the “clinical efficacy and a short time to achieve improvement in this difficult-to-treat condition. The effect was seen even though the participant sample had high depression severity and treatment resistance, both associated with poor response,” the investigators noted.
Dr. Sheline said there were no differences between active and sham stimulation in the “expected adverse events of headache and dizziness. There were no incidences of manic “flip,” and the Young Mania Rating Scale scores were stable in both active and sham groups with no difference between them.”
The researchers noted that the “large effect size” of active aiTBS could be caused by several factors, including optimized stimulation targeting, accelerated time course, and high pulse number (18,000 per day, 90,000 total).
Future studies are needed to examine the relative contributions of the different protocol components to optimize and personalize treatment and evaluate the durability of the antidepressant effects of aiTBS, they added.
A Revolutionary Approach
For comment on the study, we reached out to Nolan Williams, MD, associate professor of psychiatry and behavioral sciences at Stanford University and director of the Stanford Brain Stimulation Lab, Stanford, California.
His laboratory pioneered the Stanford Accelerated Intelligent Neuromodulation Therapy (SAINT), which was cleared by the FDA in 2022 for treatment-resistant depression.
Dr. Williams noted that the stimulation and targeting approach used in the current study “mirrors most aspects of the SAINT protocol with very similar results.”
“It’s exciting that we see this kind of pseudo-replication essentially of our work and is supportive of the general view that this approach is revolutionary in its ability to treat people quickly and have such a dramatic clinical effect,” said Dr. Williams.
In March 2024, Dr. Williams and colleagues reported the results of a pilot study of SAINT for bipolar depression, which demonstrated antidepressant efficacy akin to what was observed in the unipolar depression population.
Dr. Williams said, in his experience, the accelerated treatment protocol is convenient and well-liked by patients and, in general, is where the field of psychiatric treatment is headed.
“A general theme that we see in depression and psychiatry is that patients no longer accept long time frames for treatment as being the norm. Whether it be ketamine or this or the upcoming psychedelics, rapid-acting treatments that match the level of acuity will be the norm,” Dr. Williams said.
The study was funded by the Milken Institute and the Baszucki Brain Research Fund. The authors have disclosed no conflicts of interest. Dr. Williams is a named inventor on Stanford-owned intellectual property relating to accelerated TMS pulse pattern sequences and neuroimaging-based TMS targeting. He disclosed ties with Otsuka, NeuraWell, Magnus Medical, and Nooma.
A version of this article first appeared on Medscape.com.
the results of a small randomized controlled trial showed.
Investigators found those who received active aiTBS had a substantial decrease in depressive symptoms compared with those who received sham stimulation.
“aiTBS offers a new potential therapy for depressed patients with bipolar disorder who may not respond well to drugs or cannot tolerate their side effects while also significantly shortening the treatment window,” lead researcher Yvette Sheline, MD, director of the Center for Neuromodulation in Depression and Stress at the University of Pennsylvania, Philadelphia, said in a news release.
The study was published online in JAMA Psychiatry.
Remission After 5 Days
The Food and Drug Administration (FDA) cleared aiTBS to treat major depressive disorder. However, the treatment is not yet approved for bipolar depression.
The investigators assessed the effectiveness of aiTBS in 12 men and 12 women (mean age, 43 years) with treatment-resistant bipolar disorder. All participants were on mood stabilizers for at least 4 weeks and had Montgomery-Åsberg Depression Rating Scale (MADRS) scores of 20 or greater.
Of the 24 participants, 22 had a diagnosis of bipolar II disorder. Over 5 days, participants were randomized to receive, on a 1:1 basis, 10 sessions per day of imaging-guided active aiTBS or sham aiTBS over the left dorsolateral prefrontal cortex. Each session lasted for 1 hour. All 24 participants completed the assigned treatment and 1-month follow-up.
Active aiTBS was significantly more effective than sham stimulation in relieving depressive symptoms.
In the active treatment group, MADRS scores dropped from a mean of 30.4 at baseline to 10.5 after treatment. In contrast, the sham group experienced a minor change in MADRS scores, which decreased from a mean of 28.0 at baseline to 25.3 posttreatment.
After 5 days of treatment, half of the participants in the active aiTBS group were in remission, compared with none in the sham group.
The results demonstrate the “clinical efficacy and a short time to achieve improvement in this difficult-to-treat condition. The effect was seen even though the participant sample had high depression severity and treatment resistance, both associated with poor response,” the investigators noted.
Dr. Sheline said there were no differences between active and sham stimulation in the “expected adverse events of headache and dizziness. There were no incidences of manic “flip,” and the Young Mania Rating Scale scores were stable in both active and sham groups with no difference between them.”
The researchers noted that the “large effect size” of active aiTBS could be caused by several factors, including optimized stimulation targeting, accelerated time course, and high pulse number (18,000 per day, 90,000 total).
Future studies are needed to examine the relative contributions of the different protocol components to optimize and personalize treatment and evaluate the durability of the antidepressant effects of aiTBS, they added.
A Revolutionary Approach
For comment on the study, we reached out to Nolan Williams, MD, associate professor of psychiatry and behavioral sciences at Stanford University and director of the Stanford Brain Stimulation Lab, Stanford, California.
His laboratory pioneered the Stanford Accelerated Intelligent Neuromodulation Therapy (SAINT), which was cleared by the FDA in 2022 for treatment-resistant depression.
Dr. Williams noted that the stimulation and targeting approach used in the current study “mirrors most aspects of the SAINT protocol with very similar results.”
“It’s exciting that we see this kind of pseudo-replication essentially of our work and is supportive of the general view that this approach is revolutionary in its ability to treat people quickly and have such a dramatic clinical effect,” said Dr. Williams.
In March 2024, Dr. Williams and colleagues reported the results of a pilot study of SAINT for bipolar depression, which demonstrated antidepressant efficacy akin to what was observed in the unipolar depression population.
Dr. Williams said, in his experience, the accelerated treatment protocol is convenient and well-liked by patients and, in general, is where the field of psychiatric treatment is headed.
“A general theme that we see in depression and psychiatry is that patients no longer accept long time frames for treatment as being the norm. Whether it be ketamine or this or the upcoming psychedelics, rapid-acting treatments that match the level of acuity will be the norm,” Dr. Williams said.
The study was funded by the Milken Institute and the Baszucki Brain Research Fund. The authors have disclosed no conflicts of interest. Dr. Williams is a named inventor on Stanford-owned intellectual property relating to accelerated TMS pulse pattern sequences and neuroimaging-based TMS targeting. He disclosed ties with Otsuka, NeuraWell, Magnus Medical, and Nooma.
A version of this article first appeared on Medscape.com.
FROM JAMA PSYCHIATRY
Munchausen Syndrome by Proxy: Be Aware of Cutaneous Signs
TORONTO — Be suspicious if a child with a severe dermatologic condition is unresponsive to treatment, especially if their parent or caregiver exhibits deceptive behavior.
These could be red flags for Munchausen syndrome by proxy (MSBP), also known as factitious disorder.
“The No. 1 thing dermatologists can do in situations like this is be open to thinking outside the box and ask themselves the difficult question: Could this be something the parent is inflicting on the child,” Kelly Frasier, DO, a dermatology clinical trials and epidemiology research fellow at Northwell Health, Poughkeepsie, New York, said in an interview.
She provided a review on advancing the understanding of the dermatologic manifestations of MSBP during a poster session at the annual meeting of the Society for Pediatric Dermatology (SPD). Dr. Frasier has a particular interest in psychodermatology — she was a mental health therapist before going to medical school.
MSBP is a type of abuse intentionally inflicted by a caregiver typically on their child “for some ulterior motive,” usually to seek attention or sympathy and not for material or financial gain, explained Dr. Frasier. People with MSBP seek medical help for exaggerated or fabricated symptoms in their child. They may alter medical tests, falsify medical records, or induce symptoms in their child.
To do this, these abusers may apply any number of caustic household products, including glue, directly to the child’s skin or even in formula. Dr. Frasier shared a picture of a baby whose formula had been doctored with a caustic substance that had dripped onto his neck and face, causing a rash with blisters.
In addition to blistering, cutaneous manifestations of MSBP can include severe bruising. Or the child may present with signs similar to those of granuloma annulare (a benign condition characterized by small, raised bumps) or cicatricial pemphigoid (a rare, chronic autoimmune blistering disorder) or may have recurrent nail avulsion, purpura, or coagulopathy, said Dr. Frasier.
In almost all cases of MSBP (an estimated 96%), the abuse is inflicted by the mother, who may have a preexisting mental illness. “Usually, a psychological disorder is at play, such as depression or anxiety,” said Dr. Frasier.
Some evidence suggests that, in cases of MSBP, the caregiver may have a personality disorder such as borderline or histrionic personality disorder — or may have suffered abuse or neglect as a child or is experiencing major stress, which some evidence suggests can trigger MSPB, she added.
This type of abuse is rarely seen in children older than 6 years, likely because they get wise to what’s going on and are better able to fight back or resist as they get older, Dr. Fraser noted.
High Mortality Rate
It’s critical that cases of MSBP are identified early. While a small proportion of child abuse cases involve MSBP, the mortality rate is extremely high, about 10%, research suggests, said Dr. Frasier.
Dermatologists should be skeptical if the child’s condition hasn’t improved despite trying numerous treatments that normally would have some effect. “If you’re doing everything you can to treat something that’s usually pretty simple in terms of what you normally see clinically and how you treat it, and you’re not seeing any improvement or things continue to get worse, that’s definitely a sign something else may be going on,” Dr. Frasier said.
Another suspicious sign is inflammation that continues “for weeks or months” and “doesn’t match up with actual lab markers and lab values,” said Dr. Frasier.
Other signs of possible MSBP include evidence of chemicals in the child’s blood, stool, or urine, or the child’s condition improves while in the hospital, but symptoms return after returning home.
Also be aware of the interaction between the parent and child, said Dr. Frasier. “See if you can pick up that something else might be going on, especially if the symptoms aren’t lining up very well with what you’re physically seeing and what your clinical impression is.”
And be suspicious of a parent’s inappropriate behavior; for example, they seem to be deliberately making symptoms worse or appear overly distraught. The seemingly caring parent could be overcompensating for what she’s doing at home, “and she wants to make sure it doesn’t appear that way,” said Dr. Frasier.
To help determine if some sort of trauma is occurring at home, the child would ideally be separated from the caregiver, perhaps with a nurse or other member of the interdisciplinary medical team, Dr. Frasier said.
It appears that pediatric dermatologists are already aware of the importance of protecting children from abuse. During a presentation at the meeting on child abuse and maltreatment in dermatology, not specifically on MSBP, Romy Cho, MD, assistant professor, Department of Pediatrics, University of Toronto, who is involved with the SCAN Program at The Hospital for Sick Children, Toronto, Canada, polled the audience on whether they had ever contacted child protective services (CPS). Almost 80% said they had.
That’s good news for Dr. Frasier. “We have to be willing to contact CPS if we think there’s something going on, and be more open to that because it’s better to be safe than sorry, especially in cases involving children.”
Dr. Frasier and Dr. Cho had no relevant disclosures.
A version of this article first appeared on Medscape.com.
TORONTO — Be suspicious if a child with a severe dermatologic condition is unresponsive to treatment, especially if their parent or caregiver exhibits deceptive behavior.
These could be red flags for Munchausen syndrome by proxy (MSBP), also known as factitious disorder.
“The No. 1 thing dermatologists can do in situations like this is be open to thinking outside the box and ask themselves the difficult question: Could this be something the parent is inflicting on the child,” Kelly Frasier, DO, a dermatology clinical trials and epidemiology research fellow at Northwell Health, Poughkeepsie, New York, said in an interview.
She provided a review on advancing the understanding of the dermatologic manifestations of MSBP during a poster session at the annual meeting of the Society for Pediatric Dermatology (SPD). Dr. Frasier has a particular interest in psychodermatology — she was a mental health therapist before going to medical school.
MSBP is a type of abuse intentionally inflicted by a caregiver typically on their child “for some ulterior motive,” usually to seek attention or sympathy and not for material or financial gain, explained Dr. Frasier. People with MSBP seek medical help for exaggerated or fabricated symptoms in their child. They may alter medical tests, falsify medical records, or induce symptoms in their child.
To do this, these abusers may apply any number of caustic household products, including glue, directly to the child’s skin or even in formula. Dr. Frasier shared a picture of a baby whose formula had been doctored with a caustic substance that had dripped onto his neck and face, causing a rash with blisters.
In addition to blistering, cutaneous manifestations of MSBP can include severe bruising. Or the child may present with signs similar to those of granuloma annulare (a benign condition characterized by small, raised bumps) or cicatricial pemphigoid (a rare, chronic autoimmune blistering disorder) or may have recurrent nail avulsion, purpura, or coagulopathy, said Dr. Frasier.
In almost all cases of MSBP (an estimated 96%), the abuse is inflicted by the mother, who may have a preexisting mental illness. “Usually, a psychological disorder is at play, such as depression or anxiety,” said Dr. Frasier.
Some evidence suggests that, in cases of MSBP, the caregiver may have a personality disorder such as borderline or histrionic personality disorder — or may have suffered abuse or neglect as a child or is experiencing major stress, which some evidence suggests can trigger MSPB, she added.
This type of abuse is rarely seen in children older than 6 years, likely because they get wise to what’s going on and are better able to fight back or resist as they get older, Dr. Fraser noted.
High Mortality Rate
It’s critical that cases of MSBP are identified early. While a small proportion of child abuse cases involve MSBP, the mortality rate is extremely high, about 10%, research suggests, said Dr. Frasier.
Dermatologists should be skeptical if the child’s condition hasn’t improved despite trying numerous treatments that normally would have some effect. “If you’re doing everything you can to treat something that’s usually pretty simple in terms of what you normally see clinically and how you treat it, and you’re not seeing any improvement or things continue to get worse, that’s definitely a sign something else may be going on,” Dr. Frasier said.
Another suspicious sign is inflammation that continues “for weeks or months” and “doesn’t match up with actual lab markers and lab values,” said Dr. Frasier.
Other signs of possible MSBP include evidence of chemicals in the child’s blood, stool, or urine, or the child’s condition improves while in the hospital, but symptoms return after returning home.
Also be aware of the interaction between the parent and child, said Dr. Frasier. “See if you can pick up that something else might be going on, especially if the symptoms aren’t lining up very well with what you’re physically seeing and what your clinical impression is.”
And be suspicious of a parent’s inappropriate behavior; for example, they seem to be deliberately making symptoms worse or appear overly distraught. The seemingly caring parent could be overcompensating for what she’s doing at home, “and she wants to make sure it doesn’t appear that way,” said Dr. Frasier.
To help determine if some sort of trauma is occurring at home, the child would ideally be separated from the caregiver, perhaps with a nurse or other member of the interdisciplinary medical team, Dr. Frasier said.
It appears that pediatric dermatologists are already aware of the importance of protecting children from abuse. During a presentation at the meeting on child abuse and maltreatment in dermatology, not specifically on MSBP, Romy Cho, MD, assistant professor, Department of Pediatrics, University of Toronto, who is involved with the SCAN Program at The Hospital for Sick Children, Toronto, Canada, polled the audience on whether they had ever contacted child protective services (CPS). Almost 80% said they had.
That’s good news for Dr. Frasier. “We have to be willing to contact CPS if we think there’s something going on, and be more open to that because it’s better to be safe than sorry, especially in cases involving children.”
Dr. Frasier and Dr. Cho had no relevant disclosures.
A version of this article first appeared on Medscape.com.
TORONTO — Be suspicious if a child with a severe dermatologic condition is unresponsive to treatment, especially if their parent or caregiver exhibits deceptive behavior.
These could be red flags for Munchausen syndrome by proxy (MSBP), also known as factitious disorder.
“The No. 1 thing dermatologists can do in situations like this is be open to thinking outside the box and ask themselves the difficult question: Could this be something the parent is inflicting on the child,” Kelly Frasier, DO, a dermatology clinical trials and epidemiology research fellow at Northwell Health, Poughkeepsie, New York, said in an interview.
She provided a review on advancing the understanding of the dermatologic manifestations of MSBP during a poster session at the annual meeting of the Society for Pediatric Dermatology (SPD). Dr. Frasier has a particular interest in psychodermatology — she was a mental health therapist before going to medical school.
MSBP is a type of abuse intentionally inflicted by a caregiver typically on their child “for some ulterior motive,” usually to seek attention or sympathy and not for material or financial gain, explained Dr. Frasier. People with MSBP seek medical help for exaggerated or fabricated symptoms in their child. They may alter medical tests, falsify medical records, or induce symptoms in their child.
To do this, these abusers may apply any number of caustic household products, including glue, directly to the child’s skin or even in formula. Dr. Frasier shared a picture of a baby whose formula had been doctored with a caustic substance that had dripped onto his neck and face, causing a rash with blisters.
In addition to blistering, cutaneous manifestations of MSBP can include severe bruising. Or the child may present with signs similar to those of granuloma annulare (a benign condition characterized by small, raised bumps) or cicatricial pemphigoid (a rare, chronic autoimmune blistering disorder) or may have recurrent nail avulsion, purpura, or coagulopathy, said Dr. Frasier.
In almost all cases of MSBP (an estimated 96%), the abuse is inflicted by the mother, who may have a preexisting mental illness. “Usually, a psychological disorder is at play, such as depression or anxiety,” said Dr. Frasier.
Some evidence suggests that, in cases of MSBP, the caregiver may have a personality disorder such as borderline or histrionic personality disorder — or may have suffered abuse or neglect as a child or is experiencing major stress, which some evidence suggests can trigger MSPB, she added.
This type of abuse is rarely seen in children older than 6 years, likely because they get wise to what’s going on and are better able to fight back or resist as they get older, Dr. Fraser noted.
High Mortality Rate
It’s critical that cases of MSBP are identified early. While a small proportion of child abuse cases involve MSBP, the mortality rate is extremely high, about 10%, research suggests, said Dr. Frasier.
Dermatologists should be skeptical if the child’s condition hasn’t improved despite trying numerous treatments that normally would have some effect. “If you’re doing everything you can to treat something that’s usually pretty simple in terms of what you normally see clinically and how you treat it, and you’re not seeing any improvement or things continue to get worse, that’s definitely a sign something else may be going on,” Dr. Frasier said.
Another suspicious sign is inflammation that continues “for weeks or months” and “doesn’t match up with actual lab markers and lab values,” said Dr. Frasier.
Other signs of possible MSBP include evidence of chemicals in the child’s blood, stool, or urine, or the child’s condition improves while in the hospital, but symptoms return after returning home.
Also be aware of the interaction between the parent and child, said Dr. Frasier. “See if you can pick up that something else might be going on, especially if the symptoms aren’t lining up very well with what you’re physically seeing and what your clinical impression is.”
And be suspicious of a parent’s inappropriate behavior; for example, they seem to be deliberately making symptoms worse or appear overly distraught. The seemingly caring parent could be overcompensating for what she’s doing at home, “and she wants to make sure it doesn’t appear that way,” said Dr. Frasier.
To help determine if some sort of trauma is occurring at home, the child would ideally be separated from the caregiver, perhaps with a nurse or other member of the interdisciplinary medical team, Dr. Frasier said.
It appears that pediatric dermatologists are already aware of the importance of protecting children from abuse. During a presentation at the meeting on child abuse and maltreatment in dermatology, not specifically on MSBP, Romy Cho, MD, assistant professor, Department of Pediatrics, University of Toronto, who is involved with the SCAN Program at The Hospital for Sick Children, Toronto, Canada, polled the audience on whether they had ever contacted child protective services (CPS). Almost 80% said they had.
That’s good news for Dr. Frasier. “We have to be willing to contact CPS if we think there’s something going on, and be more open to that because it’s better to be safe than sorry, especially in cases involving children.”
Dr. Frasier and Dr. Cho had no relevant disclosures.
A version of this article first appeared on Medscape.com.
FROM SPD 2024
Strong Sibling Link With Autism Spectrum Disorder
according to a study published in Pediatrics.
When a baby had more than one older sibling with autism, the family recurrence rate rose to 36.9%, the study found.
The researchers, led by Sally Ozonoff, PhD, Department of Psychiatry and Behavioral Sciences at University of California Davis Health in Sacramento, analyzed data from 1,605 infants who had an older sibling with ASD using data from the global Baby Siblings Research Consortium.
They calculated that the rate of autism recurrence is seven times higher in families who already have one autistic child than in the general population, which points to the importance of close developmental observance in infants born in families with autistic children, particularly male infants in those families. This study replicated a 2011 study, also led by Dr. Ozonoff, which found a similar rate of familial recurrence.
Differences by Sex and Race
Dr. Ozonoff’s team found that sex and race played a part in likelihood of recurrence. Younger siblings of females with ASD were much more likely to develop the disorder (34.7%) than siblings of boys (22.5%). And male younger siblings were more likely to have ASD than girls (25.3% vs. 13.1%).
Additionally, ASD recurrence in White families was 17.8% while across other races collectively the recurrence rate was 25%.
Links with Maternal Education
Differences by maternal education were also striking. Recurrence was 32.6% when mothers had a high school or less education; 25.5% with some college; 19.7 with a college degree; and 16.9% with a graduate degree. The parental education revealed a significant effect only for mothers (P < .01); paternal education was not significant (P = .09).
Suzanne Rybczynski, MD, chief medical officer at East Tennessee Children’s Hospital in Knoxville, who was not part of the study, praised the study for following babies over time, “doing serial evaluation using two very standard tools in diagnosing autism and developmental delay.”
The babies were evaluated as early as 6 months of age, for up to seven visits. A final assessment was made at 36 months.
Dr. Rybczynski said it was interesting to see that, although ASD prevalence has increased substantially from the 2011 study (0.9%-2.8%), the findings regarding the sibling link have been consistent (18.7% in the 2011 study to 20.2% now).
Eliminating Biases
Dr. Rybczynski noted the current study also used diagnoses only from autism experts, which strengthened the findings, noting the potential for overdiagnosis when interviews are with the parents. “This really eliminates those biases.”
The authors explained the factors driving the need to update recurrence rate studies, including the growth in the prevalence of ASD in the last decade to 1 in 36. That may be caused partly by “greater awareness and identification of autistic females and cognitively able, verbal children.”
Also, new diagnostic criteria have been published, with different diagnostic thresholds since the last study. This study, they noted, had a sample size twice as large and more diverse than the 2011 sample.
The size and the diversity are particularly important, Dr. Rybczynski said, as it helps support more recent findings that ASD is not as heavily centered in White males as previously thought.
“We need to make sure we’re monitoring all children, especially from groups where there’s at least one older sibling or multiple siblings with autism or a sister with autism,” she said. The findings of this study are important not just for pediatricians but for families and all who have professional interactions with children.
Dr. Ozonoff reports travel reimbursements and honoraria from Autism Speaks and the Autism Science Foundation and book royalties from Guilford Press. One coauthor has served as a paid consultant to F. Hoffmann–La Roche and Servier and has received royalties from Sage Publications and Guilford Publications. Another is supported by the Stollery Children’s Hospital Foundation Chair in Autism. One coauthor reported a consulting agreement with EarliTec Diagnostics and book royalties from Wiley. A fourth coauthor has received funding from the Simons Foundation and consults for the Beasley Law Firm and Linus Technology. Dr. Rybczynski reported no relevant financial relationships.
according to a study published in Pediatrics.
When a baby had more than one older sibling with autism, the family recurrence rate rose to 36.9%, the study found.
The researchers, led by Sally Ozonoff, PhD, Department of Psychiatry and Behavioral Sciences at University of California Davis Health in Sacramento, analyzed data from 1,605 infants who had an older sibling with ASD using data from the global Baby Siblings Research Consortium.
They calculated that the rate of autism recurrence is seven times higher in families who already have one autistic child than in the general population, which points to the importance of close developmental observance in infants born in families with autistic children, particularly male infants in those families. This study replicated a 2011 study, also led by Dr. Ozonoff, which found a similar rate of familial recurrence.
Differences by Sex and Race
Dr. Ozonoff’s team found that sex and race played a part in likelihood of recurrence. Younger siblings of females with ASD were much more likely to develop the disorder (34.7%) than siblings of boys (22.5%). And male younger siblings were more likely to have ASD than girls (25.3% vs. 13.1%).
Additionally, ASD recurrence in White families was 17.8% while across other races collectively the recurrence rate was 25%.
Links with Maternal Education
Differences by maternal education were also striking. Recurrence was 32.6% when mothers had a high school or less education; 25.5% with some college; 19.7 with a college degree; and 16.9% with a graduate degree. The parental education revealed a significant effect only for mothers (P < .01); paternal education was not significant (P = .09).
Suzanne Rybczynski, MD, chief medical officer at East Tennessee Children’s Hospital in Knoxville, who was not part of the study, praised the study for following babies over time, “doing serial evaluation using two very standard tools in diagnosing autism and developmental delay.”
The babies were evaluated as early as 6 months of age, for up to seven visits. A final assessment was made at 36 months.
Dr. Rybczynski said it was interesting to see that, although ASD prevalence has increased substantially from the 2011 study (0.9%-2.8%), the findings regarding the sibling link have been consistent (18.7% in the 2011 study to 20.2% now).
Eliminating Biases
Dr. Rybczynski noted the current study also used diagnoses only from autism experts, which strengthened the findings, noting the potential for overdiagnosis when interviews are with the parents. “This really eliminates those biases.”
The authors explained the factors driving the need to update recurrence rate studies, including the growth in the prevalence of ASD in the last decade to 1 in 36. That may be caused partly by “greater awareness and identification of autistic females and cognitively able, verbal children.”
Also, new diagnostic criteria have been published, with different diagnostic thresholds since the last study. This study, they noted, had a sample size twice as large and more diverse than the 2011 sample.
The size and the diversity are particularly important, Dr. Rybczynski said, as it helps support more recent findings that ASD is not as heavily centered in White males as previously thought.
“We need to make sure we’re monitoring all children, especially from groups where there’s at least one older sibling or multiple siblings with autism or a sister with autism,” she said. The findings of this study are important not just for pediatricians but for families and all who have professional interactions with children.
Dr. Ozonoff reports travel reimbursements and honoraria from Autism Speaks and the Autism Science Foundation and book royalties from Guilford Press. One coauthor has served as a paid consultant to F. Hoffmann–La Roche and Servier and has received royalties from Sage Publications and Guilford Publications. Another is supported by the Stollery Children’s Hospital Foundation Chair in Autism. One coauthor reported a consulting agreement with EarliTec Diagnostics and book royalties from Wiley. A fourth coauthor has received funding from the Simons Foundation and consults for the Beasley Law Firm and Linus Technology. Dr. Rybczynski reported no relevant financial relationships.
according to a study published in Pediatrics.
When a baby had more than one older sibling with autism, the family recurrence rate rose to 36.9%, the study found.
The researchers, led by Sally Ozonoff, PhD, Department of Psychiatry and Behavioral Sciences at University of California Davis Health in Sacramento, analyzed data from 1,605 infants who had an older sibling with ASD using data from the global Baby Siblings Research Consortium.
They calculated that the rate of autism recurrence is seven times higher in families who already have one autistic child than in the general population, which points to the importance of close developmental observance in infants born in families with autistic children, particularly male infants in those families. This study replicated a 2011 study, also led by Dr. Ozonoff, which found a similar rate of familial recurrence.
Differences by Sex and Race
Dr. Ozonoff’s team found that sex and race played a part in likelihood of recurrence. Younger siblings of females with ASD were much more likely to develop the disorder (34.7%) than siblings of boys (22.5%). And male younger siblings were more likely to have ASD than girls (25.3% vs. 13.1%).
Additionally, ASD recurrence in White families was 17.8% while across other races collectively the recurrence rate was 25%.
Links with Maternal Education
Differences by maternal education were also striking. Recurrence was 32.6% when mothers had a high school or less education; 25.5% with some college; 19.7 with a college degree; and 16.9% with a graduate degree. The parental education revealed a significant effect only for mothers (P < .01); paternal education was not significant (P = .09).
Suzanne Rybczynski, MD, chief medical officer at East Tennessee Children’s Hospital in Knoxville, who was not part of the study, praised the study for following babies over time, “doing serial evaluation using two very standard tools in diagnosing autism and developmental delay.”
The babies were evaluated as early as 6 months of age, for up to seven visits. A final assessment was made at 36 months.
Dr. Rybczynski said it was interesting to see that, although ASD prevalence has increased substantially from the 2011 study (0.9%-2.8%), the findings regarding the sibling link have been consistent (18.7% in the 2011 study to 20.2% now).
Eliminating Biases
Dr. Rybczynski noted the current study also used diagnoses only from autism experts, which strengthened the findings, noting the potential for overdiagnosis when interviews are with the parents. “This really eliminates those biases.”
The authors explained the factors driving the need to update recurrence rate studies, including the growth in the prevalence of ASD in the last decade to 1 in 36. That may be caused partly by “greater awareness and identification of autistic females and cognitively able, verbal children.”
Also, new diagnostic criteria have been published, with different diagnostic thresholds since the last study. This study, they noted, had a sample size twice as large and more diverse than the 2011 sample.
The size and the diversity are particularly important, Dr. Rybczynski said, as it helps support more recent findings that ASD is not as heavily centered in White males as previously thought.
“We need to make sure we’re monitoring all children, especially from groups where there’s at least one older sibling or multiple siblings with autism or a sister with autism,” she said. The findings of this study are important not just for pediatricians but for families and all who have professional interactions with children.
Dr. Ozonoff reports travel reimbursements and honoraria from Autism Speaks and the Autism Science Foundation and book royalties from Guilford Press. One coauthor has served as a paid consultant to F. Hoffmann–La Roche and Servier and has received royalties from Sage Publications and Guilford Publications. Another is supported by the Stollery Children’s Hospital Foundation Chair in Autism. One coauthor reported a consulting agreement with EarliTec Diagnostics and book royalties from Wiley. A fourth coauthor has received funding from the Simons Foundation and consults for the Beasley Law Firm and Linus Technology. Dr. Rybczynski reported no relevant financial relationships.
FROM PEDIATRICS