User login
Postpartum depression risk increased among sexual minority women
However, with sexual orientation highly underdocumented among women giving birth, understanding of the prevalence is lacking.
“To our knowledge, this cohort study was the first to examine perinatal depression screening and symptom endorsement among sexual minority women in a major medical center in the U.S.,” reported the authors of the study published in JAMA Psychiatry.
The results “highlight the need for investigations that include strategies for measuring sexual orientation because reliance on medical record review has substantial limitations with regard to the research questions and the validity of the data,” they noted.
Clinical guidelines recommend universal perinatal depression screening at obstetric and pediatric well-infant visits; however, there are significant gaps in data on the issue when it comes to sexual minority women.
To assess the prevalence of sexual minority people giving birth and compare perinatal depression screening rates and scores with those of heterosexual cisgender women, the authors conducted a review of medical records of 18,243 female patients who gave birth at a large, diverse, university-based medical center in Chicago between January and December of 2019.
Of the patients, 57.3% of whom were non-Hispanic White, 1.5% (280) had documentation of their sexual orientation, or sexual minority status.
The results show that those identified as being in sexual minorities, including lesbian, bisexual, queer, pansexual or asexual, were more likely than were heterosexual women to be more engaged in their care – they were more likely to have attended at least one prenatal visit (20.0% vs. 13.7%; P = .002) and at least one postpartum care visit (18.6% vs. 12.8%; P = .004), and more likely to be screened for depression during postpartum care (odds ratio, 1.77; P = .002).
Sexual minority women were also significantly more likely to screen positive for depression during the postpartum period than were heterosexual women (odds ratio, 2.38; P = .03); however, all other comparisons were not significantly different.
The finding regarding postpartum depression was consistent with recent literature, including a systematic review indicating that the stress of being in a sexual minority may be heightened during the postpartum period, the authors noted.
Reasons for the heightened stress may include “being perceived as inadequate parents, heteronormativity in perinatal care, such as intake forms asking for information about the child’s father, and lack of familial social support due to nonacceptance of the parents’ sexual orientation,” the researchers explained.
The rate of only 1.5% of people giving birth who identified as a sexual minority was significantly lower than expected, and much lower that the 17% reported in a recent nationally representative sample of women, first author Leiszle Lapping-Carr, PhD, director of the sexual and relationship health program, department of psychiatry and behavioral sciences, Northwestern University, Chicago, said in an interview.
“I did not expect a rate as low at 1.5%,” she said. “I anticipated it would not be as high as the 17%, but this was quite low. I think one primary reason is that women are not interested in disclosing their sexual orientation to their ob.gyns. if they don’t have to.”
Furthermore, Dr. Lapping-Carr said, “most medical systems do not have an easy way to document sexual orientation or gender identity, and even if it exists many physicians are unaware of the process.”
On a broader level, the lower rates may be indicative of a lack of acknowledgment of sexual minorities in the ob.gyn. setting, Dr. Lapping-Carr added.
“There is a heteronormative bias implicit in most obstetrics clinics, in which pregnant people are automatically gendered as women and assumed to be heterosexual, especially if they present with a male partner,” she said.
Because of those factors, even if a pregnant person discloses sexual identity, that person may request that it not be documented in the chart, she noted.
The higher rates of postpartum depression are consistent with higher rates of mental illness that are reported in general among sexual minority women, pregnant or not, including depression, anxiety, higher rates of substance abuse, stressful life events, and intimate partner violence, compared with heterosexual women, the authors noted.
Develop more supportive systems
To address postpartum depression among sexual minority women, Dr. Lapping-Carr suggested that clinicians generally start by avoiding language and behaviors that could suggest the potential bias that sexual minority patients can face.
“The main change [in treatment] that would likely be helpful for postpartum depression treatment is removing heteronormative language, e.g., not referring to partners as ‘fathers,’ ” she said.
Also, patients may benefit from “discussion of issues of relevance to people with sexual minority identities, such as the process of adoption for female non-birthing partners,” Dr. Lapping-Carr added.
“Starting to create spaces that are inclusive and welcoming for people of all identities will go a long way in increasing your patient’s trust in you,” she said.
While there is a lack of published data regarding increases in rates of sexual minority patients who are giving birth, societal trends suggest the rates may likely be on the rise, Dr. Lapping-Carr said.
“We do know that among adolescents, endorsement of sexual and gender minority identities is much higher than in previous generations, so it would follow that the proportion of birthing people with sexual and gender minority identities would also increase,” she said.
Commenting on the study, K. Ashley Brandt, DO, obstetrics section chief and medical director of Gender Affirming Surgery at Reading Hospital, in West Reading, Pa., noted that limitations include a lack of information about the bigger picture of patients’ risk factors.
“There is no documentation of other risks factors, including rates of depression in the antenatal period, which is higher in LGBTQ individuals and also a risk factor for postpartum depression,” Dr. Brandt told this news organization.
She agreed, however, that patients may be reluctant to report their sexual minority status on the record – but such issues are often addressed.
“I believe that obstetricians do ask this question far more than other providers, but it may not be easily captured in medical records, and patients may also hesitate to disclose sexual practices and sexual orientation due to fear of medical discrimination, which is still extremely prevalent,” Dr. Brandt said.
The study underscores, however, that “same-sex parents are a reality that providers will face,” she said. “They have unique social determinants for health that often go undocumented and unaddressed, which could contribute to higher rates of depression in the postpartum period.”
Factors that may be ignored or undocumented, such as sexual minorities’ religious beliefs or social and familial support, can play significant roles in health care outcomes, Dr. Brandt added.
“Providers need to find ways to better educate themselves about LGBTQ individuals and develop more supportive systems to ensure patients feel safe in disclosing their identities.”
The authors and Dr. Brandt had no disclosures to report.
However, with sexual orientation highly underdocumented among women giving birth, understanding of the prevalence is lacking.
“To our knowledge, this cohort study was the first to examine perinatal depression screening and symptom endorsement among sexual minority women in a major medical center in the U.S.,” reported the authors of the study published in JAMA Psychiatry.
The results “highlight the need for investigations that include strategies for measuring sexual orientation because reliance on medical record review has substantial limitations with regard to the research questions and the validity of the data,” they noted.
Clinical guidelines recommend universal perinatal depression screening at obstetric and pediatric well-infant visits; however, there are significant gaps in data on the issue when it comes to sexual minority women.
To assess the prevalence of sexual minority people giving birth and compare perinatal depression screening rates and scores with those of heterosexual cisgender women, the authors conducted a review of medical records of 18,243 female patients who gave birth at a large, diverse, university-based medical center in Chicago between January and December of 2019.
Of the patients, 57.3% of whom were non-Hispanic White, 1.5% (280) had documentation of their sexual orientation, or sexual minority status.
The results show that those identified as being in sexual minorities, including lesbian, bisexual, queer, pansexual or asexual, were more likely than were heterosexual women to be more engaged in their care – they were more likely to have attended at least one prenatal visit (20.0% vs. 13.7%; P = .002) and at least one postpartum care visit (18.6% vs. 12.8%; P = .004), and more likely to be screened for depression during postpartum care (odds ratio, 1.77; P = .002).
Sexual minority women were also significantly more likely to screen positive for depression during the postpartum period than were heterosexual women (odds ratio, 2.38; P = .03); however, all other comparisons were not significantly different.
The finding regarding postpartum depression was consistent with recent literature, including a systematic review indicating that the stress of being in a sexual minority may be heightened during the postpartum period, the authors noted.
Reasons for the heightened stress may include “being perceived as inadequate parents, heteronormativity in perinatal care, such as intake forms asking for information about the child’s father, and lack of familial social support due to nonacceptance of the parents’ sexual orientation,” the researchers explained.
The rate of only 1.5% of people giving birth who identified as a sexual minority was significantly lower than expected, and much lower that the 17% reported in a recent nationally representative sample of women, first author Leiszle Lapping-Carr, PhD, director of the sexual and relationship health program, department of psychiatry and behavioral sciences, Northwestern University, Chicago, said in an interview.
“I did not expect a rate as low at 1.5%,” she said. “I anticipated it would not be as high as the 17%, but this was quite low. I think one primary reason is that women are not interested in disclosing their sexual orientation to their ob.gyns. if they don’t have to.”
Furthermore, Dr. Lapping-Carr said, “most medical systems do not have an easy way to document sexual orientation or gender identity, and even if it exists many physicians are unaware of the process.”
On a broader level, the lower rates may be indicative of a lack of acknowledgment of sexual minorities in the ob.gyn. setting, Dr. Lapping-Carr added.
“There is a heteronormative bias implicit in most obstetrics clinics, in which pregnant people are automatically gendered as women and assumed to be heterosexual, especially if they present with a male partner,” she said.
Because of those factors, even if a pregnant person discloses sexual identity, that person may request that it not be documented in the chart, she noted.
The higher rates of postpartum depression are consistent with higher rates of mental illness that are reported in general among sexual minority women, pregnant or not, including depression, anxiety, higher rates of substance abuse, stressful life events, and intimate partner violence, compared with heterosexual women, the authors noted.
Develop more supportive systems
To address postpartum depression among sexual minority women, Dr. Lapping-Carr suggested that clinicians generally start by avoiding language and behaviors that could suggest the potential bias that sexual minority patients can face.
“The main change [in treatment] that would likely be helpful for postpartum depression treatment is removing heteronormative language, e.g., not referring to partners as ‘fathers,’ ” she said.
Also, patients may benefit from “discussion of issues of relevance to people with sexual minority identities, such as the process of adoption for female non-birthing partners,” Dr. Lapping-Carr added.
“Starting to create spaces that are inclusive and welcoming for people of all identities will go a long way in increasing your patient’s trust in you,” she said.
While there is a lack of published data regarding increases in rates of sexual minority patients who are giving birth, societal trends suggest the rates may likely be on the rise, Dr. Lapping-Carr said.
“We do know that among adolescents, endorsement of sexual and gender minority identities is much higher than in previous generations, so it would follow that the proportion of birthing people with sexual and gender minority identities would also increase,” she said.
Commenting on the study, K. Ashley Brandt, DO, obstetrics section chief and medical director of Gender Affirming Surgery at Reading Hospital, in West Reading, Pa., noted that limitations include a lack of information about the bigger picture of patients’ risk factors.
“There is no documentation of other risks factors, including rates of depression in the antenatal period, which is higher in LGBTQ individuals and also a risk factor for postpartum depression,” Dr. Brandt told this news organization.
She agreed, however, that patients may be reluctant to report their sexual minority status on the record – but such issues are often addressed.
“I believe that obstetricians do ask this question far more than other providers, but it may not be easily captured in medical records, and patients may also hesitate to disclose sexual practices and sexual orientation due to fear of medical discrimination, which is still extremely prevalent,” Dr. Brandt said.
The study underscores, however, that “same-sex parents are a reality that providers will face,” she said. “They have unique social determinants for health that often go undocumented and unaddressed, which could contribute to higher rates of depression in the postpartum period.”
Factors that may be ignored or undocumented, such as sexual minorities’ religious beliefs or social and familial support, can play significant roles in health care outcomes, Dr. Brandt added.
“Providers need to find ways to better educate themselves about LGBTQ individuals and develop more supportive systems to ensure patients feel safe in disclosing their identities.”
The authors and Dr. Brandt had no disclosures to report.
However, with sexual orientation highly underdocumented among women giving birth, understanding of the prevalence is lacking.
“To our knowledge, this cohort study was the first to examine perinatal depression screening and symptom endorsement among sexual minority women in a major medical center in the U.S.,” reported the authors of the study published in JAMA Psychiatry.
The results “highlight the need for investigations that include strategies for measuring sexual orientation because reliance on medical record review has substantial limitations with regard to the research questions and the validity of the data,” they noted.
Clinical guidelines recommend universal perinatal depression screening at obstetric and pediatric well-infant visits; however, there are significant gaps in data on the issue when it comes to sexual minority women.
To assess the prevalence of sexual minority people giving birth and compare perinatal depression screening rates and scores with those of heterosexual cisgender women, the authors conducted a review of medical records of 18,243 female patients who gave birth at a large, diverse, university-based medical center in Chicago between January and December of 2019.
Of the patients, 57.3% of whom were non-Hispanic White, 1.5% (280) had documentation of their sexual orientation, or sexual minority status.
The results show that those identified as being in sexual minorities, including lesbian, bisexual, queer, pansexual or asexual, were more likely than were heterosexual women to be more engaged in their care – they were more likely to have attended at least one prenatal visit (20.0% vs. 13.7%; P = .002) and at least one postpartum care visit (18.6% vs. 12.8%; P = .004), and more likely to be screened for depression during postpartum care (odds ratio, 1.77; P = .002).
Sexual minority women were also significantly more likely to screen positive for depression during the postpartum period than were heterosexual women (odds ratio, 2.38; P = .03); however, all other comparisons were not significantly different.
The finding regarding postpartum depression was consistent with recent literature, including a systematic review indicating that the stress of being in a sexual minority may be heightened during the postpartum period, the authors noted.
Reasons for the heightened stress may include “being perceived as inadequate parents, heteronormativity in perinatal care, such as intake forms asking for information about the child’s father, and lack of familial social support due to nonacceptance of the parents’ sexual orientation,” the researchers explained.
The rate of only 1.5% of people giving birth who identified as a sexual minority was significantly lower than expected, and much lower that the 17% reported in a recent nationally representative sample of women, first author Leiszle Lapping-Carr, PhD, director of the sexual and relationship health program, department of psychiatry and behavioral sciences, Northwestern University, Chicago, said in an interview.
“I did not expect a rate as low at 1.5%,” she said. “I anticipated it would not be as high as the 17%, but this was quite low. I think one primary reason is that women are not interested in disclosing their sexual orientation to their ob.gyns. if they don’t have to.”
Furthermore, Dr. Lapping-Carr said, “most medical systems do not have an easy way to document sexual orientation or gender identity, and even if it exists many physicians are unaware of the process.”
On a broader level, the lower rates may be indicative of a lack of acknowledgment of sexual minorities in the ob.gyn. setting, Dr. Lapping-Carr added.
“There is a heteronormative bias implicit in most obstetrics clinics, in which pregnant people are automatically gendered as women and assumed to be heterosexual, especially if they present with a male partner,” she said.
Because of those factors, even if a pregnant person discloses sexual identity, that person may request that it not be documented in the chart, she noted.
The higher rates of postpartum depression are consistent with higher rates of mental illness that are reported in general among sexual minority women, pregnant or not, including depression, anxiety, higher rates of substance abuse, stressful life events, and intimate partner violence, compared with heterosexual women, the authors noted.
Develop more supportive systems
To address postpartum depression among sexual minority women, Dr. Lapping-Carr suggested that clinicians generally start by avoiding language and behaviors that could suggest the potential bias that sexual minority patients can face.
“The main change [in treatment] that would likely be helpful for postpartum depression treatment is removing heteronormative language, e.g., not referring to partners as ‘fathers,’ ” she said.
Also, patients may benefit from “discussion of issues of relevance to people with sexual minority identities, such as the process of adoption for female non-birthing partners,” Dr. Lapping-Carr added.
“Starting to create spaces that are inclusive and welcoming for people of all identities will go a long way in increasing your patient’s trust in you,” she said.
While there is a lack of published data regarding increases in rates of sexual minority patients who are giving birth, societal trends suggest the rates may likely be on the rise, Dr. Lapping-Carr said.
“We do know that among adolescents, endorsement of sexual and gender minority identities is much higher than in previous generations, so it would follow that the proportion of birthing people with sexual and gender minority identities would also increase,” she said.
Commenting on the study, K. Ashley Brandt, DO, obstetrics section chief and medical director of Gender Affirming Surgery at Reading Hospital, in West Reading, Pa., noted that limitations include a lack of information about the bigger picture of patients’ risk factors.
“There is no documentation of other risks factors, including rates of depression in the antenatal period, which is higher in LGBTQ individuals and also a risk factor for postpartum depression,” Dr. Brandt told this news organization.
She agreed, however, that patients may be reluctant to report their sexual minority status on the record – but such issues are often addressed.
“I believe that obstetricians do ask this question far more than other providers, but it may not be easily captured in medical records, and patients may also hesitate to disclose sexual practices and sexual orientation due to fear of medical discrimination, which is still extremely prevalent,” Dr. Brandt said.
The study underscores, however, that “same-sex parents are a reality that providers will face,” she said. “They have unique social determinants for health that often go undocumented and unaddressed, which could contribute to higher rates of depression in the postpartum period.”
Factors that may be ignored or undocumented, such as sexual minorities’ religious beliefs or social and familial support, can play significant roles in health care outcomes, Dr. Brandt added.
“Providers need to find ways to better educate themselves about LGBTQ individuals and develop more supportive systems to ensure patients feel safe in disclosing their identities.”
The authors and Dr. Brandt had no disclosures to report.
FROM JAMA PSYCHIATRY
Managing intrahepatic cholestasis of pregnancy
CASE Pregnant woman with intense itching
A 28-year-old woman (G1P0) is seen for a routine prenatal visit at 32 3/7 weeks’ gestation. She reports having generalized intense itching, including on her palms and soles, that is most intense at night and has been present for approximately 1 week. Her pregnancy is otherwise uncomplicated to date. Physical exam is within normal limits, with no evidence of a skin rash. Cholestasis of pregnancy is suspected, and laboratory tests are ordered, including bile acids and liver transaminases. Test results show that her aspartate transaminase (AST) and alanine transaminase (ALT) levels are mildly elevated at 55 IU/L and 41 IU/L, respectively, and several days later her bile acid level result is 21 µmol/L.
How should this patient be managed?
Intrahepatic cholestasis of pregnancy (ICP) affects 0.5% to 0.7% of pregnant individuals and results in maternal pruritus and elevated serum bile acid levels.1-3 Pruritus in ICP typically is generalized, including occurrence on the palms of the hands and soles of the feet, and it often is reported to be worse at night.4 Up to 25% of pregnant women will develop pruritus during pregnancy but the majority will not have ICP.2,5 Patients with ICP have no associated rash, but clinicians may note excoriations on exam. ICP typically presents in the third trimester of pregnancy but has been reported to occur earlier in gestation.6
Making a diagnosis of ICP
The presence of maternal pruritus in the absence of a skin condition along with elevated levels of serum bile acids are required for the diagnosis of ICP.7 Thus, a thorough history and physical exam is recommended to rule out another skin condition that could potentially explain the patient’s pruritus.
Some controversy exists regarding the bile acid level cutoff that should be used to make a diagnosis of ICP.8 It has been noted that nonfasting serum bile acid levels in pregnancy are considerably higher than those in in the nonpregnant state, and an upper limit of 18 µmol/L has been proposed as a cutoff in pregnancy.9 However, nonfasting total serum bile acids also have been shown to vary considerably by race, with levels 25.8% higher in Black women compared with those in White women and 24.3% higher in Black women compared with those in south Asian women.9 This raises the question of whether we should be using race-specific bile acid values to make a diagnosis of ICP.
Bile acid levels also vary based on whether a patient is in a fasting or postprandial state.10 Despite this variation, most guidelines do not recommend testing fasting bile acid levels as the postprandial state effect overall is small.7,9,11 The Society for Maternal-Fetal Medicine (SMFM) recommends that if a pregnancy-specific bile acid range is available from the laboratory, then the upper limit of normal for pregnancy should be used when making a diagnosis of ICP.7 The SMFM guidelines also acknowledge, however, that pregnancy-specific values rarely are available, and in this case, levels above the upper limit of normal—often 10 µmol/L should be considered diagnostic for ICP until further evidence regarding optimal bile acid cutoff levels in pregnancy becomes available.7
For patients with suspected ICP, liver transaminase levels should be measured in addition to nonfasting serum bile acid levels.7 A thorough history should include assessment for additional symptoms of liver disease, such as changes in weight, appetite, jaundice, excessive fatigue, malaise, and abdominal pain.7 Elevated transaminases levels may be associated with ICP, but they are not necessary for diagnosis. In the absence of additional clinical symptoms that suggest underlying liver disease or severe early onset ICP, additional evaluation beyond nonfasting serum bile acids and liver transaminase levels, such as liver ultrasonography or evaluation for viral or autoimmune hepatitis, is not recommended.7 Obstetric care clinicians should be aware that there is an increased incidence of preeclampsia among patients with ICP, although no specific guidance regarding further recommendations for screening is provided.7

Continue to: Risks associated with ICP...
Risks associated with ICP
For both patients and clinicians, the greatest concern among patients with ICP is the increased risk of stillbirth. Stillbirth risk in ICP appears to be related to serum bile acid levels and has been reported to be highest in patients with bile acid levels greater than 100 µmol/L. A systematic review and meta-analysis of ICP studies demonstrated no increased risk of stillbirth among patients with bile acid levels less than 100 µmol/L.12 These results, however, must be interpreted with extreme caution as the majority of studies included patients with ICP who were actively managed with attempts to mitigate the risk of stillbirth.7
In the absence of additional pregnancy risk factors, the risk of stillbirth among patients with ICP and serum bile acid levels between 19 and 39 µmol/L does not appear to be elevated above their baseline risk.11 The same is true for pregnant individuals with ICP and no additional pregnancy risk factors with serum bile acid levels between 40 and 99 µmol/L until approximately 38 weeks’ gestation, when the risk of stillbirth is elevated.11 The risk of stillbirth is elevated in ICP with peak bile acid levels greater than 100 µmol/L, with an absolute risk of 3.44%.11
Management of patients with ICP
Laboratory evaluation
There is no consensus on the need for repeat testing of bile acid levels in patients with ICP. SMFM advises that follow-up testing of bile acid levels may help to guide delivery timing, especially in cases of severe ICP, but the society recommends against serial testing.7 By contrast, the Royal College of Obstetricians and Gynaecologists (RCOG) provides a detailed algorithm regarding time intervals between serum bile acid level testing to guide delivery timing.11 The TABLE lists the strategy for reassessment of serum bile acid levels in ICP as recommended by the RCOG.11

In the United States, bile acid testing traditionally takes several days as the testing is commonly performed at reference laboratories. We therefore suggest that clinicians consider repeating bile acid level testing in situations in which the timing of delivery may be altered if further elevations of bile acid levels were noted. This is particularly relevant for patients diagnosed with ICP early in the third trimester when repeat bile acid levels would still allow for an adjustment in delivery timing.
Antepartum fetal surveillance
Unfortunately, antepartum fetal testing for pregnant patients with ICP does not appear to reliably predict or prevent stillbirth as several studies have reported stillbirths within days of normal fetal testing.13-16 It is therefore important to counsel pregnant patients regarding monitoring of fetal movements and advise them to present for evaluation if concerns arise.
Currently, SMFM recommends that patients with ICP should begin antenatal fetal surveillance at a gestational age when abnormal fetal testing would result in delivery.7 Patients should be counseled, however, regarding the unpredictability of stillbirth with ICP in the setting of a low absolute risk of such.
Medications
While SMFM recommends a starting dose of ursodeoxycholic acid 10 to 15 mg/kg per day divided into 2 or 3 daily doses as first-line therapy for the treatment of maternal symptoms of ICP, it is important to acknowledge that the goal of treatment is to alleviate maternal symptoms as there is no evidence that ursodeoxycholic acid improves either maternal serum bile acid levels or perinatal outcomes.7,17,18 Since publication of the guidelines, ursodeoxycholic acid’s lack of benefit has been further confirmed in a meta-analysis, and thus discontinuation is not unreasonable in the absence of any improvement in maternal symptoms.18
Timing of delivery
The optimal management of ICP remains unknown. SMFM recommends delivery based on peak serum bile acid levels. Delivery is recommended at 36 weeks’ gestation with ICP and total bile acid levels greater than 100 µmol/L as these patients have the greatest risk of stillbirth.7 For patients with ICP and bile acid levels less than 100 µmol/L, delivery is recommended between 36 0/7 and 39 0/7 weeks’ gestation.7 This is a wide gestational age window for clinicians to consider timing of delivery, and certainly the risks of stillbirth should be carefully balanced with the morbidity associated with a preterm or early term delivery.
For patients with ICP who have bile acid levels greater than 40 µmol/L, it is reasonable to consider delivery earlier in the gestational age window, given an evidence of increased risk of stillbirth after 38 weeks.7,12 For patients with ICP who have bile acid levels less than 40 µmol/L, delivery closer to 39 weeks’ gestation is recommended, as the risk of stillbirth does not appear to be increased above the baseline risk.7,12 Clinicians should be aware that the presence of concomitant morbidities, such as preeclampsia and gestational diabetes, are associated with an increased risk of stillbirth and should be considered for delivery planning.19
Postpartum follow-up
Routine laboratory evaluation following delivery is not recommended.7 However, in the presence of persistent pruritus or other signs and symptoms of hepatobiliary disease, liver function tests should be repeated with referral to hepatology if results are persistently abnormal 4 to 6 weeks postpartum.7
CASE Patient follow-up and outcomes
- Abedin P, Weaver JB, Egginton E. Intrahepatic cholestasis of pregnancy: prevalence and ethnic distribution. Ethn Health. 1999;4:35-37.
- Kenyon AP, Tribe RM, Nelson-Piercy C, et al. Pruritus in pregnancy: a study of anatomical distribution and prevalence in relation to the development of obstetric cholestasis. Obstet Med. 2010;3:25-29.
- Wikstrom Shemer E, Marschall HU, Ludvigsson JF, et al. Intrahepatic cholestasis of pregnancy and associated adverse pregnancy and fetal outcomes: a 12-year population-based cohort study. BJOG. 2013;120:717-723.
- Ambros-Rudolph CM, Glatz M, Trauner M, et al. The importance of serum bile acid level analysis and treatment with ursodeoxycholic acid in intrahepatic cholestasis of pregnancy: a case series from central Europe. Arch Dermatol. 2007;143:757-762.
- Szczech J, Wiatrowski A, Hirnle L, et al. Prevalence and relevance of pruritus in pregnancy. Biomed Res Int. 2017;2017:4238139.
- Geenes V, Williamson C. Intrahepatic cholestasis of pregnancy. World J Gastroenterol. 2009;15:2049-2066.
- Society for Maternal-Fetal Medicine; Lee RH, Greenberg M, Metz TD, et al. Society for Maternal-Fetal Medicine Consult Series #53: intrahepatic cholestasis of pregnancy: replaces Consult #13, April 2011. Am J Obstet Gynecol. 2021;224:B2-B9.
- Horgan R, Bitas C, Abuhamad A. Intrahepatic cholestasis of pregnancy: a comparison of Society for Maternal-Fetal Medicine and the Royal College of Obstetricians and Gynaecologists’ guidelines. Am J Obstet Gynecol MFM. 2023;5:100838.
- Mitchell AL, Ovadia C, Syngelaki A, et al. Re-evaluating diagnostic thresholds for intrahepatic cholestasis of pregnancy: case-control and cohort study. BJOG. 2021;128:1635-1644.
- Adams A, Jacobs K, Vogel RI, et al. Bile acid determination after standardized glucose load in pregnant women. AJP Rep. 2015;5:e168-e171.
- Girling J, Knight CL, Chappell L; Royal College of Obstetricians and Gynaecologists. Intrahepatic cholestasis of pregnancy: Green-top guideline no. 43, June 2022. BJOG. 2022;129:e95-e114.
- Ovadia C, Seed PT, Sklavounos A, et al. Association of adverse perinatal outcomes of intrahepatic cholestasis of pregnancy with biochemical markers: results of aggregate and individual patient data meta-analyses. Lancet. 2019;393:899-909.
- Alsulyman OM, Ouzounian JG, Ames-Castro M, et al. Intrahepatic cholestasis of pregnancy: perinatal outcome associated with expectant management. Am J Obstet Gynecol. 1996;175:957-960.
- Herrera CA, Manuck TA, Stoddard GJ, et al. Perinatal outcomes associated with intrahepatic cholestasis of pregnancy. J Matern Fetal Neonatal Med. 2018;31:1913-1920.
- Lee RH, Incerpi MH, Miller DA, et al. Sudden fetal death in intrahepatic cholestasis of pregnancy. Obstet Gynecol. 2009;113:528-531.
- Sentilhes L, Verspyck E, Pia P, et al. Fetal death in a patient with intrahepatic cholestasis of pregnancy. Obstet Gynecol. 2006;107:458-460.
- Chappell LC, Bell JL, Smith A, et al; PITCHES Study Group. Ursodeoxycholic acid versus placebo in women with intrahepatic cholestasis of pregnancy (PITCHES): a randomised controlled trial. Lancet. 2019;394:849-860.
- Ovadia C, Sajous J, Seed PT, et al. Ursodeoxycholic acid in intrahepatic cholestasis of pregnancy: a systematic review and individual participant data meta-analysis. Lancet Gastroenterol Hepatol. 2021;6:547-558.
- Geenes V, Chappell LC, Seed PT, et al. Association of severe intrahepatic cholestasis of pregnancy with adverse pregnancy outcomes: a prospective population-based case-control study. Hepatology. 2014;59:1482-1491.
CASE Pregnant woman with intense itching
A 28-year-old woman (G1P0) is seen for a routine prenatal visit at 32 3/7 weeks’ gestation. She reports having generalized intense itching, including on her palms and soles, that is most intense at night and has been present for approximately 1 week. Her pregnancy is otherwise uncomplicated to date. Physical exam is within normal limits, with no evidence of a skin rash. Cholestasis of pregnancy is suspected, and laboratory tests are ordered, including bile acids and liver transaminases. Test results show that her aspartate transaminase (AST) and alanine transaminase (ALT) levels are mildly elevated at 55 IU/L and 41 IU/L, respectively, and several days later her bile acid level result is 21 µmol/L.
How should this patient be managed?
Intrahepatic cholestasis of pregnancy (ICP) affects 0.5% to 0.7% of pregnant individuals and results in maternal pruritus and elevated serum bile acid levels.1-3 Pruritus in ICP typically is generalized, including occurrence on the palms of the hands and soles of the feet, and it often is reported to be worse at night.4 Up to 25% of pregnant women will develop pruritus during pregnancy but the majority will not have ICP.2,5 Patients with ICP have no associated rash, but clinicians may note excoriations on exam. ICP typically presents in the third trimester of pregnancy but has been reported to occur earlier in gestation.6
Making a diagnosis of ICP
The presence of maternal pruritus in the absence of a skin condition along with elevated levels of serum bile acids are required for the diagnosis of ICP.7 Thus, a thorough history and physical exam is recommended to rule out another skin condition that could potentially explain the patient’s pruritus.
Some controversy exists regarding the bile acid level cutoff that should be used to make a diagnosis of ICP.8 It has been noted that nonfasting serum bile acid levels in pregnancy are considerably higher than those in in the nonpregnant state, and an upper limit of 18 µmol/L has been proposed as a cutoff in pregnancy.9 However, nonfasting total serum bile acids also have been shown to vary considerably by race, with levels 25.8% higher in Black women compared with those in White women and 24.3% higher in Black women compared with those in south Asian women.9 This raises the question of whether we should be using race-specific bile acid values to make a diagnosis of ICP.
Bile acid levels also vary based on whether a patient is in a fasting or postprandial state.10 Despite this variation, most guidelines do not recommend testing fasting bile acid levels as the postprandial state effect overall is small.7,9,11 The Society for Maternal-Fetal Medicine (SMFM) recommends that if a pregnancy-specific bile acid range is available from the laboratory, then the upper limit of normal for pregnancy should be used when making a diagnosis of ICP.7 The SMFM guidelines also acknowledge, however, that pregnancy-specific values rarely are available, and in this case, levels above the upper limit of normal—often 10 µmol/L should be considered diagnostic for ICP until further evidence regarding optimal bile acid cutoff levels in pregnancy becomes available.7
For patients with suspected ICP, liver transaminase levels should be measured in addition to nonfasting serum bile acid levels.7 A thorough history should include assessment for additional symptoms of liver disease, such as changes in weight, appetite, jaundice, excessive fatigue, malaise, and abdominal pain.7 Elevated transaminases levels may be associated with ICP, but they are not necessary for diagnosis. In the absence of additional clinical symptoms that suggest underlying liver disease or severe early onset ICP, additional evaluation beyond nonfasting serum bile acids and liver transaminase levels, such as liver ultrasonography or evaluation for viral or autoimmune hepatitis, is not recommended.7 Obstetric care clinicians should be aware that there is an increased incidence of preeclampsia among patients with ICP, although no specific guidance regarding further recommendations for screening is provided.7

Continue to: Risks associated with ICP...
Risks associated with ICP
For both patients and clinicians, the greatest concern among patients with ICP is the increased risk of stillbirth. Stillbirth risk in ICP appears to be related to serum bile acid levels and has been reported to be highest in patients with bile acid levels greater than 100 µmol/L. A systematic review and meta-analysis of ICP studies demonstrated no increased risk of stillbirth among patients with bile acid levels less than 100 µmol/L.12 These results, however, must be interpreted with extreme caution as the majority of studies included patients with ICP who were actively managed with attempts to mitigate the risk of stillbirth.7
In the absence of additional pregnancy risk factors, the risk of stillbirth among patients with ICP and serum bile acid levels between 19 and 39 µmol/L does not appear to be elevated above their baseline risk.11 The same is true for pregnant individuals with ICP and no additional pregnancy risk factors with serum bile acid levels between 40 and 99 µmol/L until approximately 38 weeks’ gestation, when the risk of stillbirth is elevated.11 The risk of stillbirth is elevated in ICP with peak bile acid levels greater than 100 µmol/L, with an absolute risk of 3.44%.11
Management of patients with ICP
Laboratory evaluation
There is no consensus on the need for repeat testing of bile acid levels in patients with ICP. SMFM advises that follow-up testing of bile acid levels may help to guide delivery timing, especially in cases of severe ICP, but the society recommends against serial testing.7 By contrast, the Royal College of Obstetricians and Gynaecologists (RCOG) provides a detailed algorithm regarding time intervals between serum bile acid level testing to guide delivery timing.11 The TABLE lists the strategy for reassessment of serum bile acid levels in ICP as recommended by the RCOG.11

In the United States, bile acid testing traditionally takes several days as the testing is commonly performed at reference laboratories. We therefore suggest that clinicians consider repeating bile acid level testing in situations in which the timing of delivery may be altered if further elevations of bile acid levels were noted. This is particularly relevant for patients diagnosed with ICP early in the third trimester when repeat bile acid levels would still allow for an adjustment in delivery timing.
Antepartum fetal surveillance
Unfortunately, antepartum fetal testing for pregnant patients with ICP does not appear to reliably predict or prevent stillbirth as several studies have reported stillbirths within days of normal fetal testing.13-16 It is therefore important to counsel pregnant patients regarding monitoring of fetal movements and advise them to present for evaluation if concerns arise.
Currently, SMFM recommends that patients with ICP should begin antenatal fetal surveillance at a gestational age when abnormal fetal testing would result in delivery.7 Patients should be counseled, however, regarding the unpredictability of stillbirth with ICP in the setting of a low absolute risk of such.
Medications
While SMFM recommends a starting dose of ursodeoxycholic acid 10 to 15 mg/kg per day divided into 2 or 3 daily doses as first-line therapy for the treatment of maternal symptoms of ICP, it is important to acknowledge that the goal of treatment is to alleviate maternal symptoms as there is no evidence that ursodeoxycholic acid improves either maternal serum bile acid levels or perinatal outcomes.7,17,18 Since publication of the guidelines, ursodeoxycholic acid’s lack of benefit has been further confirmed in a meta-analysis, and thus discontinuation is not unreasonable in the absence of any improvement in maternal symptoms.18
Timing of delivery
The optimal management of ICP remains unknown. SMFM recommends delivery based on peak serum bile acid levels. Delivery is recommended at 36 weeks’ gestation with ICP and total bile acid levels greater than 100 µmol/L as these patients have the greatest risk of stillbirth.7 For patients with ICP and bile acid levels less than 100 µmol/L, delivery is recommended between 36 0/7 and 39 0/7 weeks’ gestation.7 This is a wide gestational age window for clinicians to consider timing of delivery, and certainly the risks of stillbirth should be carefully balanced with the morbidity associated with a preterm or early term delivery.
For patients with ICP who have bile acid levels greater than 40 µmol/L, it is reasonable to consider delivery earlier in the gestational age window, given an evidence of increased risk of stillbirth after 38 weeks.7,12 For patients with ICP who have bile acid levels less than 40 µmol/L, delivery closer to 39 weeks’ gestation is recommended, as the risk of stillbirth does not appear to be increased above the baseline risk.7,12 Clinicians should be aware that the presence of concomitant morbidities, such as preeclampsia and gestational diabetes, are associated with an increased risk of stillbirth and should be considered for delivery planning.19
Postpartum follow-up
Routine laboratory evaluation following delivery is not recommended.7 However, in the presence of persistent pruritus or other signs and symptoms of hepatobiliary disease, liver function tests should be repeated with referral to hepatology if results are persistently abnormal 4 to 6 weeks postpartum.7
CASE Patient follow-up and outcomes
CASE Pregnant woman with intense itching
A 28-year-old woman (G1P0) is seen for a routine prenatal visit at 32 3/7 weeks’ gestation. She reports having generalized intense itching, including on her palms and soles, that is most intense at night and has been present for approximately 1 week. Her pregnancy is otherwise uncomplicated to date. Physical exam is within normal limits, with no evidence of a skin rash. Cholestasis of pregnancy is suspected, and laboratory tests are ordered, including bile acids and liver transaminases. Test results show that her aspartate transaminase (AST) and alanine transaminase (ALT) levels are mildly elevated at 55 IU/L and 41 IU/L, respectively, and several days later her bile acid level result is 21 µmol/L.
How should this patient be managed?
Intrahepatic cholestasis of pregnancy (ICP) affects 0.5% to 0.7% of pregnant individuals and results in maternal pruritus and elevated serum bile acid levels.1-3 Pruritus in ICP typically is generalized, including occurrence on the palms of the hands and soles of the feet, and it often is reported to be worse at night.4 Up to 25% of pregnant women will develop pruritus during pregnancy but the majority will not have ICP.2,5 Patients with ICP have no associated rash, but clinicians may note excoriations on exam. ICP typically presents in the third trimester of pregnancy but has been reported to occur earlier in gestation.6
Making a diagnosis of ICP
The presence of maternal pruritus in the absence of a skin condition along with elevated levels of serum bile acids are required for the diagnosis of ICP.7 Thus, a thorough history and physical exam is recommended to rule out another skin condition that could potentially explain the patient’s pruritus.
Some controversy exists regarding the bile acid level cutoff that should be used to make a diagnosis of ICP.8 It has been noted that nonfasting serum bile acid levels in pregnancy are considerably higher than those in in the nonpregnant state, and an upper limit of 18 µmol/L has been proposed as a cutoff in pregnancy.9 However, nonfasting total serum bile acids also have been shown to vary considerably by race, with levels 25.8% higher in Black women compared with those in White women and 24.3% higher in Black women compared with those in south Asian women.9 This raises the question of whether we should be using race-specific bile acid values to make a diagnosis of ICP.
Bile acid levels also vary based on whether a patient is in a fasting or postprandial state.10 Despite this variation, most guidelines do not recommend testing fasting bile acid levels as the postprandial state effect overall is small.7,9,11 The Society for Maternal-Fetal Medicine (SMFM) recommends that if a pregnancy-specific bile acid range is available from the laboratory, then the upper limit of normal for pregnancy should be used when making a diagnosis of ICP.7 The SMFM guidelines also acknowledge, however, that pregnancy-specific values rarely are available, and in this case, levels above the upper limit of normal—often 10 µmol/L should be considered diagnostic for ICP until further evidence regarding optimal bile acid cutoff levels in pregnancy becomes available.7
For patients with suspected ICP, liver transaminase levels should be measured in addition to nonfasting serum bile acid levels.7 A thorough history should include assessment for additional symptoms of liver disease, such as changes in weight, appetite, jaundice, excessive fatigue, malaise, and abdominal pain.7 Elevated transaminases levels may be associated with ICP, but they are not necessary for diagnosis. In the absence of additional clinical symptoms that suggest underlying liver disease or severe early onset ICP, additional evaluation beyond nonfasting serum bile acids and liver transaminase levels, such as liver ultrasonography or evaluation for viral or autoimmune hepatitis, is not recommended.7 Obstetric care clinicians should be aware that there is an increased incidence of preeclampsia among patients with ICP, although no specific guidance regarding further recommendations for screening is provided.7

Continue to: Risks associated with ICP...
Risks associated with ICP
For both patients and clinicians, the greatest concern among patients with ICP is the increased risk of stillbirth. Stillbirth risk in ICP appears to be related to serum bile acid levels and has been reported to be highest in patients with bile acid levels greater than 100 µmol/L. A systematic review and meta-analysis of ICP studies demonstrated no increased risk of stillbirth among patients with bile acid levels less than 100 µmol/L.12 These results, however, must be interpreted with extreme caution as the majority of studies included patients with ICP who were actively managed with attempts to mitigate the risk of stillbirth.7
In the absence of additional pregnancy risk factors, the risk of stillbirth among patients with ICP and serum bile acid levels between 19 and 39 µmol/L does not appear to be elevated above their baseline risk.11 The same is true for pregnant individuals with ICP and no additional pregnancy risk factors with serum bile acid levels between 40 and 99 µmol/L until approximately 38 weeks’ gestation, when the risk of stillbirth is elevated.11 The risk of stillbirth is elevated in ICP with peak bile acid levels greater than 100 µmol/L, with an absolute risk of 3.44%.11
Management of patients with ICP
Laboratory evaluation
There is no consensus on the need for repeat testing of bile acid levels in patients with ICP. SMFM advises that follow-up testing of bile acid levels may help to guide delivery timing, especially in cases of severe ICP, but the society recommends against serial testing.7 By contrast, the Royal College of Obstetricians and Gynaecologists (RCOG) provides a detailed algorithm regarding time intervals between serum bile acid level testing to guide delivery timing.11 The TABLE lists the strategy for reassessment of serum bile acid levels in ICP as recommended by the RCOG.11

In the United States, bile acid testing traditionally takes several days as the testing is commonly performed at reference laboratories. We therefore suggest that clinicians consider repeating bile acid level testing in situations in which the timing of delivery may be altered if further elevations of bile acid levels were noted. This is particularly relevant for patients diagnosed with ICP early in the third trimester when repeat bile acid levels would still allow for an adjustment in delivery timing.
Antepartum fetal surveillance
Unfortunately, antepartum fetal testing for pregnant patients with ICP does not appear to reliably predict or prevent stillbirth as several studies have reported stillbirths within days of normal fetal testing.13-16 It is therefore important to counsel pregnant patients regarding monitoring of fetal movements and advise them to present for evaluation if concerns arise.
Currently, SMFM recommends that patients with ICP should begin antenatal fetal surveillance at a gestational age when abnormal fetal testing would result in delivery.7 Patients should be counseled, however, regarding the unpredictability of stillbirth with ICP in the setting of a low absolute risk of such.
Medications
While SMFM recommends a starting dose of ursodeoxycholic acid 10 to 15 mg/kg per day divided into 2 or 3 daily doses as first-line therapy for the treatment of maternal symptoms of ICP, it is important to acknowledge that the goal of treatment is to alleviate maternal symptoms as there is no evidence that ursodeoxycholic acid improves either maternal serum bile acid levels or perinatal outcomes.7,17,18 Since publication of the guidelines, ursodeoxycholic acid’s lack of benefit has been further confirmed in a meta-analysis, and thus discontinuation is not unreasonable in the absence of any improvement in maternal symptoms.18
Timing of delivery
The optimal management of ICP remains unknown. SMFM recommends delivery based on peak serum bile acid levels. Delivery is recommended at 36 weeks’ gestation with ICP and total bile acid levels greater than 100 µmol/L as these patients have the greatest risk of stillbirth.7 For patients with ICP and bile acid levels less than 100 µmol/L, delivery is recommended between 36 0/7 and 39 0/7 weeks’ gestation.7 This is a wide gestational age window for clinicians to consider timing of delivery, and certainly the risks of stillbirth should be carefully balanced with the morbidity associated with a preterm or early term delivery.
For patients with ICP who have bile acid levels greater than 40 µmol/L, it is reasonable to consider delivery earlier in the gestational age window, given an evidence of increased risk of stillbirth after 38 weeks.7,12 For patients with ICP who have bile acid levels less than 40 µmol/L, delivery closer to 39 weeks’ gestation is recommended, as the risk of stillbirth does not appear to be increased above the baseline risk.7,12 Clinicians should be aware that the presence of concomitant morbidities, such as preeclampsia and gestational diabetes, are associated with an increased risk of stillbirth and should be considered for delivery planning.19
Postpartum follow-up
Routine laboratory evaluation following delivery is not recommended.7 However, in the presence of persistent pruritus or other signs and symptoms of hepatobiliary disease, liver function tests should be repeated with referral to hepatology if results are persistently abnormal 4 to 6 weeks postpartum.7
CASE Patient follow-up and outcomes
- Abedin P, Weaver JB, Egginton E. Intrahepatic cholestasis of pregnancy: prevalence and ethnic distribution. Ethn Health. 1999;4:35-37.
- Kenyon AP, Tribe RM, Nelson-Piercy C, et al. Pruritus in pregnancy: a study of anatomical distribution and prevalence in relation to the development of obstetric cholestasis. Obstet Med. 2010;3:25-29.
- Wikstrom Shemer E, Marschall HU, Ludvigsson JF, et al. Intrahepatic cholestasis of pregnancy and associated adverse pregnancy and fetal outcomes: a 12-year population-based cohort study. BJOG. 2013;120:717-723.
- Ambros-Rudolph CM, Glatz M, Trauner M, et al. The importance of serum bile acid level analysis and treatment with ursodeoxycholic acid in intrahepatic cholestasis of pregnancy: a case series from central Europe. Arch Dermatol. 2007;143:757-762.
- Szczech J, Wiatrowski A, Hirnle L, et al. Prevalence and relevance of pruritus in pregnancy. Biomed Res Int. 2017;2017:4238139.
- Geenes V, Williamson C. Intrahepatic cholestasis of pregnancy. World J Gastroenterol. 2009;15:2049-2066.
- Society for Maternal-Fetal Medicine; Lee RH, Greenberg M, Metz TD, et al. Society for Maternal-Fetal Medicine Consult Series #53: intrahepatic cholestasis of pregnancy: replaces Consult #13, April 2011. Am J Obstet Gynecol. 2021;224:B2-B9.
- Horgan R, Bitas C, Abuhamad A. Intrahepatic cholestasis of pregnancy: a comparison of Society for Maternal-Fetal Medicine and the Royal College of Obstetricians and Gynaecologists’ guidelines. Am J Obstet Gynecol MFM. 2023;5:100838.
- Mitchell AL, Ovadia C, Syngelaki A, et al. Re-evaluating diagnostic thresholds for intrahepatic cholestasis of pregnancy: case-control and cohort study. BJOG. 2021;128:1635-1644.
- Adams A, Jacobs K, Vogel RI, et al. Bile acid determination after standardized glucose load in pregnant women. AJP Rep. 2015;5:e168-e171.
- Girling J, Knight CL, Chappell L; Royal College of Obstetricians and Gynaecologists. Intrahepatic cholestasis of pregnancy: Green-top guideline no. 43, June 2022. BJOG. 2022;129:e95-e114.
- Ovadia C, Seed PT, Sklavounos A, et al. Association of adverse perinatal outcomes of intrahepatic cholestasis of pregnancy with biochemical markers: results of aggregate and individual patient data meta-analyses. Lancet. 2019;393:899-909.
- Alsulyman OM, Ouzounian JG, Ames-Castro M, et al. Intrahepatic cholestasis of pregnancy: perinatal outcome associated with expectant management. Am J Obstet Gynecol. 1996;175:957-960.
- Herrera CA, Manuck TA, Stoddard GJ, et al. Perinatal outcomes associated with intrahepatic cholestasis of pregnancy. J Matern Fetal Neonatal Med. 2018;31:1913-1920.
- Lee RH, Incerpi MH, Miller DA, et al. Sudden fetal death in intrahepatic cholestasis of pregnancy. Obstet Gynecol. 2009;113:528-531.
- Sentilhes L, Verspyck E, Pia P, et al. Fetal death in a patient with intrahepatic cholestasis of pregnancy. Obstet Gynecol. 2006;107:458-460.
- Chappell LC, Bell JL, Smith A, et al; PITCHES Study Group. Ursodeoxycholic acid versus placebo in women with intrahepatic cholestasis of pregnancy (PITCHES): a randomised controlled trial. Lancet. 2019;394:849-860.
- Ovadia C, Sajous J, Seed PT, et al. Ursodeoxycholic acid in intrahepatic cholestasis of pregnancy: a systematic review and individual participant data meta-analysis. Lancet Gastroenterol Hepatol. 2021;6:547-558.
- Geenes V, Chappell LC, Seed PT, et al. Association of severe intrahepatic cholestasis of pregnancy with adverse pregnancy outcomes: a prospective population-based case-control study. Hepatology. 2014;59:1482-1491.
- Abedin P, Weaver JB, Egginton E. Intrahepatic cholestasis of pregnancy: prevalence and ethnic distribution. Ethn Health. 1999;4:35-37.
- Kenyon AP, Tribe RM, Nelson-Piercy C, et al. Pruritus in pregnancy: a study of anatomical distribution and prevalence in relation to the development of obstetric cholestasis. Obstet Med. 2010;3:25-29.
- Wikstrom Shemer E, Marschall HU, Ludvigsson JF, et al. Intrahepatic cholestasis of pregnancy and associated adverse pregnancy and fetal outcomes: a 12-year population-based cohort study. BJOG. 2013;120:717-723.
- Ambros-Rudolph CM, Glatz M, Trauner M, et al. The importance of serum bile acid level analysis and treatment with ursodeoxycholic acid in intrahepatic cholestasis of pregnancy: a case series from central Europe. Arch Dermatol. 2007;143:757-762.
- Szczech J, Wiatrowski A, Hirnle L, et al. Prevalence and relevance of pruritus in pregnancy. Biomed Res Int. 2017;2017:4238139.
- Geenes V, Williamson C. Intrahepatic cholestasis of pregnancy. World J Gastroenterol. 2009;15:2049-2066.
- Society for Maternal-Fetal Medicine; Lee RH, Greenberg M, Metz TD, et al. Society for Maternal-Fetal Medicine Consult Series #53: intrahepatic cholestasis of pregnancy: replaces Consult #13, April 2011. Am J Obstet Gynecol. 2021;224:B2-B9.
- Horgan R, Bitas C, Abuhamad A. Intrahepatic cholestasis of pregnancy: a comparison of Society for Maternal-Fetal Medicine and the Royal College of Obstetricians and Gynaecologists’ guidelines. Am J Obstet Gynecol MFM. 2023;5:100838.
- Mitchell AL, Ovadia C, Syngelaki A, et al. Re-evaluating diagnostic thresholds for intrahepatic cholestasis of pregnancy: case-control and cohort study. BJOG. 2021;128:1635-1644.
- Adams A, Jacobs K, Vogel RI, et al. Bile acid determination after standardized glucose load in pregnant women. AJP Rep. 2015;5:e168-e171.
- Girling J, Knight CL, Chappell L; Royal College of Obstetricians and Gynaecologists. Intrahepatic cholestasis of pregnancy: Green-top guideline no. 43, June 2022. BJOG. 2022;129:e95-e114.
- Ovadia C, Seed PT, Sklavounos A, et al. Association of adverse perinatal outcomes of intrahepatic cholestasis of pregnancy with biochemical markers: results of aggregate and individual patient data meta-analyses. Lancet. 2019;393:899-909.
- Alsulyman OM, Ouzounian JG, Ames-Castro M, et al. Intrahepatic cholestasis of pregnancy: perinatal outcome associated with expectant management. Am J Obstet Gynecol. 1996;175:957-960.
- Herrera CA, Manuck TA, Stoddard GJ, et al. Perinatal outcomes associated with intrahepatic cholestasis of pregnancy. J Matern Fetal Neonatal Med. 2018;31:1913-1920.
- Lee RH, Incerpi MH, Miller DA, et al. Sudden fetal death in intrahepatic cholestasis of pregnancy. Obstet Gynecol. 2009;113:528-531.
- Sentilhes L, Verspyck E, Pia P, et al. Fetal death in a patient with intrahepatic cholestasis of pregnancy. Obstet Gynecol. 2006;107:458-460.
- Chappell LC, Bell JL, Smith A, et al; PITCHES Study Group. Ursodeoxycholic acid versus placebo in women with intrahepatic cholestasis of pregnancy (PITCHES): a randomised controlled trial. Lancet. 2019;394:849-860.
- Ovadia C, Sajous J, Seed PT, et al. Ursodeoxycholic acid in intrahepatic cholestasis of pregnancy: a systematic review and individual participant data meta-analysis. Lancet Gastroenterol Hepatol. 2021;6:547-558.
- Geenes V, Chappell LC, Seed PT, et al. Association of severe intrahepatic cholestasis of pregnancy with adverse pregnancy outcomes: a prospective population-based case-control study. Hepatology. 2014;59:1482-1491.
Experts highlight benefits and offer caveats for first postpartum depression pill
For the first time, the Food and Drug Administration approved a pill taken once daily for 14 days to help women manage the often strong, sometimes overpowering symptoms of postpartum depression.
for a condition that affects an estimated 1 in 8 women in the United States. What will it mean for easing symptoms such as hopelessness, crankiness, and lack of interest in bonding with the baby or, in the case of multiples, babies – and in some cases, thoughts of death or suicide?
A fast-acting option
“We don’t have many oral medications that are fast-acting antidepressants, so this is incredibly exciting,” said Sarah Oreck, MD, a psychiatrist in private practice in Los Angeles who specializes in reproductive psychiatry. The rapid response is likely because the medication targets the hormonal mechanism underlying postpartum depression, she added.
Zuranolone (Zurzuvae, Biogen/Sage) is different from most other antidepressants – it is designed to be taken for a shorter period. Also, Because zuranolone is a pill, it is more convenient to take than the other FDA-approved treatment, the IV infusion brexanolone (Zulresso, Sage).
“It’s obviously game changing to have something in pill form. The infusion has to be done at an infusion center to monitor people for any complications,” said Kimberly Yonkers, MD, a psychiatrist specializing in women’s health, a Distinguished Life Fellow of the American Psychiatric Association (APA), and the Katz Family Chair of Psychiatry at the University of Massachusetts Chan Medical School/UMass Memorial Medical Center in Worcester.
Women may experience improvement in postpartum depression in as soon as 3 days after starting the medication. In contrast, “typical antidepressants can take up to 2 weeks before patients notice a difference and 4 to 8 weeks to see a full response. A fast-acting pill that can be taken orally could be an ideal option for the 15% to 20% of women who experience postpartum depression,” said Priya Gopalan, MD, a psychiatrist with UPMC Western Psychiatric Hospital and Magee-Womens Hospital in Pittsburgh.
The medical community, and reproductive psychiatrists in particular, has always suspected differences in the biological underpinnings of postpartum depression and major depressive disorder, Dr. Oreck said. “We know that postpartum depression looks different from major depressive disorder and that hormonal shifts during pregnancy and postpartum are a huge risk factor for postpartum depression,” she said.
Although selective serotonin reuptake inhibitors (SSRIs) are helpful and currently the standard of care for treating moderate to severe postpartum depression in combination with therapy, Dr. Oreck added, early studies suggest that zuranolone may work faster and potentially be more effective than SSRIs in treating the condition.
Zuranolone is a version of a naturally occurring hormone called allopregnanolone, a metabolite of progesterone. Concentrations of allopregnanolone rise dramatically during pregnancy and then drop precipitously after childbirth. Zuranolone works through modulating GABA-A, a neurotransmitter implicated in the development of depression.
“It is encouraging that postpartum individuals may now have more options to manage a debilitating condition that affects them and their families,” said Christopher Zahn, MD, interim CEO and chief of clinical practice and health equity and quality for the American College of Obstetricians and Gynecologists (ACOG).
ACOG recommends women be screened for depression at least three times – during early pregnancy, later in pregnancy, and again after delivery. A decision to start this or any other medicine should be individualized and based on shared decision-making between a patient and doctor, Dr. Zahn added.
The cost of zuranolone is not yet known. Dr. Yonkers said cost of the infusion can serve as a cautionary tale for the manufacturer. Some reports put the infusion cost at $34,000. “Cost is going to be an important component to this. The previous intervention was priced so high that it was not affordable to many people and it was difficult to access.”
Beyond ‘baby blues’
The APA has changed the name from “postpartum depression” to “peripartum depression” because evidence suggests feelings and symptoms also can start late in pregnancy. “It means you don’t have to wait until somebody delivers to screen for depression. We have to recognize that depression can occur during pregnancy,” Dr. Yonkers said. “In fact it is not uncommon during the third trimester.”
No matter when it starts, the condition can be “very serious,” particularly if the person already experiences depression, including bipolar disorder, Dr. Yonkers added.
Postpartum depression “is more than just ‘baby blues.’ It is a potentially debilitating illness that causes feelings of intense sadness and worthlessness, making it difficult to care for and bond with your newborn,” Dr. Gopalan said.
Can be a medical emergency
Severe postpartum depression requires immediate attention and treatment.
“One of the things we have to be cautious about is for people with previous predisposition to hurt themselves,” Dr. Yonkers said. “It is therefore important to consider somebody’s medical and behavioral health history as well.
“For an individual with recurring depression or severe episodes of depression, this may not be sufficient, because they are just going to get these 14 days of therapy,” Dr. Yonkers said. “They may need ongoing antidepressants.
“It may not be the right pill for everybody,” Dr. Yonkers added. She recommended everyone be followed closely during and after treatment “to make sure they are responding and to monitor for relapse.”
The science that led to approval
The clinical trials showed early response in patients with severe postpartum depression. Researchers conducted two studies of women who developed a major depressive episode in the third trimester of pregnancy or within 4 weeks of delivery. They found women who took zuranolone once in the evening for 14 days “showed significantly more improvement in their symptoms compared to those in the placebo group.”
The antidepressant effect lasted at least 4 weeks after stopping the medication.
Drowsiness, dizziness, diarrhea, fatigue, nasopharyngitis, and urinary tract infection were the most common side effects. The label has a boxed warning noting that the medication can affect a person’s ability to drive and perform other potentially hazardous activities. Use of zuranolone may also cause suicidal thoughts and behavior, according to an FDA news release announcing the approval.
The start of more help for mothers?
Zuranolone is not a cure-all. As with most psychiatric prescriptions, the medication likely will work best in conjunction with behavioral health treatments such as psychotherapy, use of other medications, behavioral management, support groups, and self-care tools such as meditation, exercise, and yoga, Dr. Gopalan said.
Dr. Oreck said she hopes this first pill approval will lead to more discoveries. “I hope this is the beginning of more innovation and development of novel treatments that can target women’s mental health issues specifically – female reproductive hormones impact mental health in unique ways and it’s exciting to finally see research and development dollars dedicated to them,” she said. “The FDA approval of this pill provides the potential to improve the lives of millions of Americans suffering from postpartum depression.”
Dr. Oreck, Dr. Yonkers, Dr. Gopalan, and Dr. Zahn have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
For the first time, the Food and Drug Administration approved a pill taken once daily for 14 days to help women manage the often strong, sometimes overpowering symptoms of postpartum depression.
for a condition that affects an estimated 1 in 8 women in the United States. What will it mean for easing symptoms such as hopelessness, crankiness, and lack of interest in bonding with the baby or, in the case of multiples, babies – and in some cases, thoughts of death or suicide?
A fast-acting option
“We don’t have many oral medications that are fast-acting antidepressants, so this is incredibly exciting,” said Sarah Oreck, MD, a psychiatrist in private practice in Los Angeles who specializes in reproductive psychiatry. The rapid response is likely because the medication targets the hormonal mechanism underlying postpartum depression, she added.
Zuranolone (Zurzuvae, Biogen/Sage) is different from most other antidepressants – it is designed to be taken for a shorter period. Also, Because zuranolone is a pill, it is more convenient to take than the other FDA-approved treatment, the IV infusion brexanolone (Zulresso, Sage).
“It’s obviously game changing to have something in pill form. The infusion has to be done at an infusion center to monitor people for any complications,” said Kimberly Yonkers, MD, a psychiatrist specializing in women’s health, a Distinguished Life Fellow of the American Psychiatric Association (APA), and the Katz Family Chair of Psychiatry at the University of Massachusetts Chan Medical School/UMass Memorial Medical Center in Worcester.
Women may experience improvement in postpartum depression in as soon as 3 days after starting the medication. In contrast, “typical antidepressants can take up to 2 weeks before patients notice a difference and 4 to 8 weeks to see a full response. A fast-acting pill that can be taken orally could be an ideal option for the 15% to 20% of women who experience postpartum depression,” said Priya Gopalan, MD, a psychiatrist with UPMC Western Psychiatric Hospital and Magee-Womens Hospital in Pittsburgh.
The medical community, and reproductive psychiatrists in particular, has always suspected differences in the biological underpinnings of postpartum depression and major depressive disorder, Dr. Oreck said. “We know that postpartum depression looks different from major depressive disorder and that hormonal shifts during pregnancy and postpartum are a huge risk factor for postpartum depression,” she said.
Although selective serotonin reuptake inhibitors (SSRIs) are helpful and currently the standard of care for treating moderate to severe postpartum depression in combination with therapy, Dr. Oreck added, early studies suggest that zuranolone may work faster and potentially be more effective than SSRIs in treating the condition.
Zuranolone is a version of a naturally occurring hormone called allopregnanolone, a metabolite of progesterone. Concentrations of allopregnanolone rise dramatically during pregnancy and then drop precipitously after childbirth. Zuranolone works through modulating GABA-A, a neurotransmitter implicated in the development of depression.
“It is encouraging that postpartum individuals may now have more options to manage a debilitating condition that affects them and their families,” said Christopher Zahn, MD, interim CEO and chief of clinical practice and health equity and quality for the American College of Obstetricians and Gynecologists (ACOG).
ACOG recommends women be screened for depression at least three times – during early pregnancy, later in pregnancy, and again after delivery. A decision to start this or any other medicine should be individualized and based on shared decision-making between a patient and doctor, Dr. Zahn added.
The cost of zuranolone is not yet known. Dr. Yonkers said cost of the infusion can serve as a cautionary tale for the manufacturer. Some reports put the infusion cost at $34,000. “Cost is going to be an important component to this. The previous intervention was priced so high that it was not affordable to many people and it was difficult to access.”
Beyond ‘baby blues’
The APA has changed the name from “postpartum depression” to “peripartum depression” because evidence suggests feelings and symptoms also can start late in pregnancy. “It means you don’t have to wait until somebody delivers to screen for depression. We have to recognize that depression can occur during pregnancy,” Dr. Yonkers said. “In fact it is not uncommon during the third trimester.”
No matter when it starts, the condition can be “very serious,” particularly if the person already experiences depression, including bipolar disorder, Dr. Yonkers added.
Postpartum depression “is more than just ‘baby blues.’ It is a potentially debilitating illness that causes feelings of intense sadness and worthlessness, making it difficult to care for and bond with your newborn,” Dr. Gopalan said.
Can be a medical emergency
Severe postpartum depression requires immediate attention and treatment.
“One of the things we have to be cautious about is for people with previous predisposition to hurt themselves,” Dr. Yonkers said. “It is therefore important to consider somebody’s medical and behavioral health history as well.
“For an individual with recurring depression or severe episodes of depression, this may not be sufficient, because they are just going to get these 14 days of therapy,” Dr. Yonkers said. “They may need ongoing antidepressants.
“It may not be the right pill for everybody,” Dr. Yonkers added. She recommended everyone be followed closely during and after treatment “to make sure they are responding and to monitor for relapse.”
The science that led to approval
The clinical trials showed early response in patients with severe postpartum depression. Researchers conducted two studies of women who developed a major depressive episode in the third trimester of pregnancy or within 4 weeks of delivery. They found women who took zuranolone once in the evening for 14 days “showed significantly more improvement in their symptoms compared to those in the placebo group.”
The antidepressant effect lasted at least 4 weeks after stopping the medication.
Drowsiness, dizziness, diarrhea, fatigue, nasopharyngitis, and urinary tract infection were the most common side effects. The label has a boxed warning noting that the medication can affect a person’s ability to drive and perform other potentially hazardous activities. Use of zuranolone may also cause suicidal thoughts and behavior, according to an FDA news release announcing the approval.
The start of more help for mothers?
Zuranolone is not a cure-all. As with most psychiatric prescriptions, the medication likely will work best in conjunction with behavioral health treatments such as psychotherapy, use of other medications, behavioral management, support groups, and self-care tools such as meditation, exercise, and yoga, Dr. Gopalan said.
Dr. Oreck said she hopes this first pill approval will lead to more discoveries. “I hope this is the beginning of more innovation and development of novel treatments that can target women’s mental health issues specifically – female reproductive hormones impact mental health in unique ways and it’s exciting to finally see research and development dollars dedicated to them,” she said. “The FDA approval of this pill provides the potential to improve the lives of millions of Americans suffering from postpartum depression.”
Dr. Oreck, Dr. Yonkers, Dr. Gopalan, and Dr. Zahn have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
For the first time, the Food and Drug Administration approved a pill taken once daily for 14 days to help women manage the often strong, sometimes overpowering symptoms of postpartum depression.
for a condition that affects an estimated 1 in 8 women in the United States. What will it mean for easing symptoms such as hopelessness, crankiness, and lack of interest in bonding with the baby or, in the case of multiples, babies – and in some cases, thoughts of death or suicide?
A fast-acting option
“We don’t have many oral medications that are fast-acting antidepressants, so this is incredibly exciting,” said Sarah Oreck, MD, a psychiatrist in private practice in Los Angeles who specializes in reproductive psychiatry. The rapid response is likely because the medication targets the hormonal mechanism underlying postpartum depression, she added.
Zuranolone (Zurzuvae, Biogen/Sage) is different from most other antidepressants – it is designed to be taken for a shorter period. Also, Because zuranolone is a pill, it is more convenient to take than the other FDA-approved treatment, the IV infusion brexanolone (Zulresso, Sage).
“It’s obviously game changing to have something in pill form. The infusion has to be done at an infusion center to monitor people for any complications,” said Kimberly Yonkers, MD, a psychiatrist specializing in women’s health, a Distinguished Life Fellow of the American Psychiatric Association (APA), and the Katz Family Chair of Psychiatry at the University of Massachusetts Chan Medical School/UMass Memorial Medical Center in Worcester.
Women may experience improvement in postpartum depression in as soon as 3 days after starting the medication. In contrast, “typical antidepressants can take up to 2 weeks before patients notice a difference and 4 to 8 weeks to see a full response. A fast-acting pill that can be taken orally could be an ideal option for the 15% to 20% of women who experience postpartum depression,” said Priya Gopalan, MD, a psychiatrist with UPMC Western Psychiatric Hospital and Magee-Womens Hospital in Pittsburgh.
The medical community, and reproductive psychiatrists in particular, has always suspected differences in the biological underpinnings of postpartum depression and major depressive disorder, Dr. Oreck said. “We know that postpartum depression looks different from major depressive disorder and that hormonal shifts during pregnancy and postpartum are a huge risk factor for postpartum depression,” she said.
Although selective serotonin reuptake inhibitors (SSRIs) are helpful and currently the standard of care for treating moderate to severe postpartum depression in combination with therapy, Dr. Oreck added, early studies suggest that zuranolone may work faster and potentially be more effective than SSRIs in treating the condition.
Zuranolone is a version of a naturally occurring hormone called allopregnanolone, a metabolite of progesterone. Concentrations of allopregnanolone rise dramatically during pregnancy and then drop precipitously after childbirth. Zuranolone works through modulating GABA-A, a neurotransmitter implicated in the development of depression.
“It is encouraging that postpartum individuals may now have more options to manage a debilitating condition that affects them and their families,” said Christopher Zahn, MD, interim CEO and chief of clinical practice and health equity and quality for the American College of Obstetricians and Gynecologists (ACOG).
ACOG recommends women be screened for depression at least three times – during early pregnancy, later in pregnancy, and again after delivery. A decision to start this or any other medicine should be individualized and based on shared decision-making between a patient and doctor, Dr. Zahn added.
The cost of zuranolone is not yet known. Dr. Yonkers said cost of the infusion can serve as a cautionary tale for the manufacturer. Some reports put the infusion cost at $34,000. “Cost is going to be an important component to this. The previous intervention was priced so high that it was not affordable to many people and it was difficult to access.”
Beyond ‘baby blues’
The APA has changed the name from “postpartum depression” to “peripartum depression” because evidence suggests feelings and symptoms also can start late in pregnancy. “It means you don’t have to wait until somebody delivers to screen for depression. We have to recognize that depression can occur during pregnancy,” Dr. Yonkers said. “In fact it is not uncommon during the third trimester.”
No matter when it starts, the condition can be “very serious,” particularly if the person already experiences depression, including bipolar disorder, Dr. Yonkers added.
Postpartum depression “is more than just ‘baby blues.’ It is a potentially debilitating illness that causes feelings of intense sadness and worthlessness, making it difficult to care for and bond with your newborn,” Dr. Gopalan said.
Can be a medical emergency
Severe postpartum depression requires immediate attention and treatment.
“One of the things we have to be cautious about is for people with previous predisposition to hurt themselves,” Dr. Yonkers said. “It is therefore important to consider somebody’s medical and behavioral health history as well.
“For an individual with recurring depression or severe episodes of depression, this may not be sufficient, because they are just going to get these 14 days of therapy,” Dr. Yonkers said. “They may need ongoing antidepressants.
“It may not be the right pill for everybody,” Dr. Yonkers added. She recommended everyone be followed closely during and after treatment “to make sure they are responding and to monitor for relapse.”
The science that led to approval
The clinical trials showed early response in patients with severe postpartum depression. Researchers conducted two studies of women who developed a major depressive episode in the third trimester of pregnancy or within 4 weeks of delivery. They found women who took zuranolone once in the evening for 14 days “showed significantly more improvement in their symptoms compared to those in the placebo group.”
The antidepressant effect lasted at least 4 weeks after stopping the medication.
Drowsiness, dizziness, diarrhea, fatigue, nasopharyngitis, and urinary tract infection were the most common side effects. The label has a boxed warning noting that the medication can affect a person’s ability to drive and perform other potentially hazardous activities. Use of zuranolone may also cause suicidal thoughts and behavior, according to an FDA news release announcing the approval.
The start of more help for mothers?
Zuranolone is not a cure-all. As with most psychiatric prescriptions, the medication likely will work best in conjunction with behavioral health treatments such as psychotherapy, use of other medications, behavioral management, support groups, and self-care tools such as meditation, exercise, and yoga, Dr. Gopalan said.
Dr. Oreck said she hopes this first pill approval will lead to more discoveries. “I hope this is the beginning of more innovation and development of novel treatments that can target women’s mental health issues specifically – female reproductive hormones impact mental health in unique ways and it’s exciting to finally see research and development dollars dedicated to them,” she said. “The FDA approval of this pill provides the potential to improve the lives of millions of Americans suffering from postpartum depression.”
Dr. Oreck, Dr. Yonkers, Dr. Gopalan, and Dr. Zahn have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Ontario case shows potential supplement risk for consumers
A woman’s quest to become pregnant resulted in lead poisoning from an Ayurvedic treatment. The case triggered the seizure of pills from an Ontario natural-products clinic and the issuance of government warnings about the risks of products from this business, according to a new report.
, including the presence of lead and other metals in Ayurvedic products, according to the report.
“When consumer products may be contaminated with lead, or when lead exposure is linked to sources in the community, involving public health can facilitate broader actions to reduce and prevent exposures to other people at risk,” wrote report author Julian Gitelman, MD, MPH, a resident physician at the University of Toronto Dalla Lana School of Public Health, and colleagues.
Their case study was published in the Canadian Medical Association Journal.
The researchers detailed what happened after a 39-year-old woman sought medical care for abdominal pain, constipation, nausea, and vomiting. The woman underwent a series of tests, including colonoscopy, laparoscopy, and biopsies of bone marrow and ovarian cysts.
Only later did clinicians home in on the cause of her ailments: the Ayurvedic medications that the patient had been taking daily for more than a year for infertility. Her daily regimen had varied, ranging from a few pills to a dozen pills.
Heavy metals are sometimes intentionally added to Ayurvedic supplements for perceived healing properties, wrote the authors. They cited a previous study of a sample of Ayurvedic pills bought on the Internet from manufacturers based in the United States and India that showed that 21% contained lead, mercury, or arsenic.
A case report published last year in German Medical Weekly raised the same issue.
Melatonin gummies
Regulators in many countries struggle to help consumers understand the risks of natural health supplements, and the challenge extends well beyond Ayurvedic products.
There has been a “huge and very troubling increase” in U.S. poison control calls associated with gummy-bear products containing melatonin, said Canadian Senator Stan Kutcher, MD, at a May 11 meeting of Canada’s Standing Senate Committee on Social Affairs, Science, and Technology.
In April, JAMA published a U.S. analysis of melatonin gummy products, Dr. Kutcher noted. In this research letter, investigators reported that one product did not contain detectable levels of melatonin but did contain 31.3 mg of cannabidiol.
In other products, the quantity of melatonin ranged from 74% to 347% of the labeled quantity. A previous Canadian study of 16 melatonin brands found that the actual dose of melatonin ranged from 17% to 478% of the declared quantity, the letter noted.
The May 11 Senate meeting provided a forum for many of the recurring debates about supplements, which also are known as natural health products.
Barry Power, PharmD, editor in chief for the Canadian Pharmacists Association, said that his group was disappointed when Canada excluded natural health products from Vanessa’s Law, which was passed in 2014. This law sought to improve the reporting of adverse reactions to drugs.
“We’re glad this is being revisited now,” Dr. Power told the Senate committee. “Although natural health products are often seen as low risk, we need to keep in mind that ‘low risk’ does not mean ‘no risk,’ and ‘natural’ does not mean ‘safe.’ ”
In contrast, Aaron Skelton, chief executive of the Canadian Health Food Association, spoke against this bid to expand the reach of Vanessa’s Law into natural health products. Canadian lawmakers attached provisions regarding increased oversight of natural health products to a budget package instead of considering them as part of a stand-alone bill.
“Our concern is that the powers that are being discussed have not been reviewed and debated,” Mr. Skelton told Dr. Kutcher. “The potential for overreach and unnecessary regulation is significant, and that deserves debate.”
“Profits should not trump Canadians’ health,” answered Dr. Kutcher, who earlier served as head of the psychiatry department at Dalhousie University in Halifax, N.S.
By June, Vanessa’s Law had been expanded with provisions that address natural health products, including the reporting of products that present a serious risk to consumers.
Educating consumers
Many consumers overestimate the level of government regulation of supplements, said Pieter A. Cohen, MD, leader of the Supplement Research Program at Cambridge Health Alliance in Massachusetts. Dr. Cohen was the lead author of the JAMA research letter about melatonin products.
Supplements often share shelves in pharmacies with medicines that are subject to more strict regulation, which causes confusion.
“It’s really hard to wrap your brain around [the fact] that a health product is being sold in pharmacies in the United States and it’s not being vetted by the FDA [U.S. Food and Drug Administration]”, Dr. Cohen said in an interview
The confusion extends across borders. Many consumers in other countries will assume that the FDA performed premarket screening of U.S.-made supplements, but that is not the case, he said.
People who want to take supplements should look for reputable sources of information about them, such as the website of the National Institutes of Health’s Office of Dietary Supplements, Dr. Cohen said. But patients often forget or fail to do this, which can create medical puzzles, such as the case of the woman in the Ontario case study, said Peter Lurie, MD, MPH, executive director of the nonprofit Center for Science in the Public Interest, which has pressed for increased regulation of supplements.
Clinicians need to keep in mind that patients may need prodding to reveal what supplements they are taking, he said.
“They just think of them as different, somehow not the province of the doctor,” Dr. Lurie said. “For others, they are concerned that the doctors will disapprove. So, they hide it.”
A version of this article first appeared on Medscape.com.
A woman’s quest to become pregnant resulted in lead poisoning from an Ayurvedic treatment. The case triggered the seizure of pills from an Ontario natural-products clinic and the issuance of government warnings about the risks of products from this business, according to a new report.
, including the presence of lead and other metals in Ayurvedic products, according to the report.
“When consumer products may be contaminated with lead, or when lead exposure is linked to sources in the community, involving public health can facilitate broader actions to reduce and prevent exposures to other people at risk,” wrote report author Julian Gitelman, MD, MPH, a resident physician at the University of Toronto Dalla Lana School of Public Health, and colleagues.
Their case study was published in the Canadian Medical Association Journal.
The researchers detailed what happened after a 39-year-old woman sought medical care for abdominal pain, constipation, nausea, and vomiting. The woman underwent a series of tests, including colonoscopy, laparoscopy, and biopsies of bone marrow and ovarian cysts.
Only later did clinicians home in on the cause of her ailments: the Ayurvedic medications that the patient had been taking daily for more than a year for infertility. Her daily regimen had varied, ranging from a few pills to a dozen pills.
Heavy metals are sometimes intentionally added to Ayurvedic supplements for perceived healing properties, wrote the authors. They cited a previous study of a sample of Ayurvedic pills bought on the Internet from manufacturers based in the United States and India that showed that 21% contained lead, mercury, or arsenic.
A case report published last year in German Medical Weekly raised the same issue.
Melatonin gummies
Regulators in many countries struggle to help consumers understand the risks of natural health supplements, and the challenge extends well beyond Ayurvedic products.
There has been a “huge and very troubling increase” in U.S. poison control calls associated with gummy-bear products containing melatonin, said Canadian Senator Stan Kutcher, MD, at a May 11 meeting of Canada’s Standing Senate Committee on Social Affairs, Science, and Technology.
In April, JAMA published a U.S. analysis of melatonin gummy products, Dr. Kutcher noted. In this research letter, investigators reported that one product did not contain detectable levels of melatonin but did contain 31.3 mg of cannabidiol.
In other products, the quantity of melatonin ranged from 74% to 347% of the labeled quantity. A previous Canadian study of 16 melatonin brands found that the actual dose of melatonin ranged from 17% to 478% of the declared quantity, the letter noted.
The May 11 Senate meeting provided a forum for many of the recurring debates about supplements, which also are known as natural health products.
Barry Power, PharmD, editor in chief for the Canadian Pharmacists Association, said that his group was disappointed when Canada excluded natural health products from Vanessa’s Law, which was passed in 2014. This law sought to improve the reporting of adverse reactions to drugs.
“We’re glad this is being revisited now,” Dr. Power told the Senate committee. “Although natural health products are often seen as low risk, we need to keep in mind that ‘low risk’ does not mean ‘no risk,’ and ‘natural’ does not mean ‘safe.’ ”
In contrast, Aaron Skelton, chief executive of the Canadian Health Food Association, spoke against this bid to expand the reach of Vanessa’s Law into natural health products. Canadian lawmakers attached provisions regarding increased oversight of natural health products to a budget package instead of considering them as part of a stand-alone bill.
“Our concern is that the powers that are being discussed have not been reviewed and debated,” Mr. Skelton told Dr. Kutcher. “The potential for overreach and unnecessary regulation is significant, and that deserves debate.”
“Profits should not trump Canadians’ health,” answered Dr. Kutcher, who earlier served as head of the psychiatry department at Dalhousie University in Halifax, N.S.
By June, Vanessa’s Law had been expanded with provisions that address natural health products, including the reporting of products that present a serious risk to consumers.
Educating consumers
Many consumers overestimate the level of government regulation of supplements, said Pieter A. Cohen, MD, leader of the Supplement Research Program at Cambridge Health Alliance in Massachusetts. Dr. Cohen was the lead author of the JAMA research letter about melatonin products.
Supplements often share shelves in pharmacies with medicines that are subject to more strict regulation, which causes confusion.
“It’s really hard to wrap your brain around [the fact] that a health product is being sold in pharmacies in the United States and it’s not being vetted by the FDA [U.S. Food and Drug Administration]”, Dr. Cohen said in an interview
The confusion extends across borders. Many consumers in other countries will assume that the FDA performed premarket screening of U.S.-made supplements, but that is not the case, he said.
People who want to take supplements should look for reputable sources of information about them, such as the website of the National Institutes of Health’s Office of Dietary Supplements, Dr. Cohen said. But patients often forget or fail to do this, which can create medical puzzles, such as the case of the woman in the Ontario case study, said Peter Lurie, MD, MPH, executive director of the nonprofit Center for Science in the Public Interest, which has pressed for increased regulation of supplements.
Clinicians need to keep in mind that patients may need prodding to reveal what supplements they are taking, he said.
“They just think of them as different, somehow not the province of the doctor,” Dr. Lurie said. “For others, they are concerned that the doctors will disapprove. So, they hide it.”
A version of this article first appeared on Medscape.com.
A woman’s quest to become pregnant resulted in lead poisoning from an Ayurvedic treatment. The case triggered the seizure of pills from an Ontario natural-products clinic and the issuance of government warnings about the risks of products from this business, according to a new report.
, including the presence of lead and other metals in Ayurvedic products, according to the report.
“When consumer products may be contaminated with lead, or when lead exposure is linked to sources in the community, involving public health can facilitate broader actions to reduce and prevent exposures to other people at risk,” wrote report author Julian Gitelman, MD, MPH, a resident physician at the University of Toronto Dalla Lana School of Public Health, and colleagues.
Their case study was published in the Canadian Medical Association Journal.
The researchers detailed what happened after a 39-year-old woman sought medical care for abdominal pain, constipation, nausea, and vomiting. The woman underwent a series of tests, including colonoscopy, laparoscopy, and biopsies of bone marrow and ovarian cysts.
Only later did clinicians home in on the cause of her ailments: the Ayurvedic medications that the patient had been taking daily for more than a year for infertility. Her daily regimen had varied, ranging from a few pills to a dozen pills.
Heavy metals are sometimes intentionally added to Ayurvedic supplements for perceived healing properties, wrote the authors. They cited a previous study of a sample of Ayurvedic pills bought on the Internet from manufacturers based in the United States and India that showed that 21% contained lead, mercury, or arsenic.
A case report published last year in German Medical Weekly raised the same issue.
Melatonin gummies
Regulators in many countries struggle to help consumers understand the risks of natural health supplements, and the challenge extends well beyond Ayurvedic products.
There has been a “huge and very troubling increase” in U.S. poison control calls associated with gummy-bear products containing melatonin, said Canadian Senator Stan Kutcher, MD, at a May 11 meeting of Canada’s Standing Senate Committee on Social Affairs, Science, and Technology.
In April, JAMA published a U.S. analysis of melatonin gummy products, Dr. Kutcher noted. In this research letter, investigators reported that one product did not contain detectable levels of melatonin but did contain 31.3 mg of cannabidiol.
In other products, the quantity of melatonin ranged from 74% to 347% of the labeled quantity. A previous Canadian study of 16 melatonin brands found that the actual dose of melatonin ranged from 17% to 478% of the declared quantity, the letter noted.
The May 11 Senate meeting provided a forum for many of the recurring debates about supplements, which also are known as natural health products.
Barry Power, PharmD, editor in chief for the Canadian Pharmacists Association, said that his group was disappointed when Canada excluded natural health products from Vanessa’s Law, which was passed in 2014. This law sought to improve the reporting of adverse reactions to drugs.
“We’re glad this is being revisited now,” Dr. Power told the Senate committee. “Although natural health products are often seen as low risk, we need to keep in mind that ‘low risk’ does not mean ‘no risk,’ and ‘natural’ does not mean ‘safe.’ ”
In contrast, Aaron Skelton, chief executive of the Canadian Health Food Association, spoke against this bid to expand the reach of Vanessa’s Law into natural health products. Canadian lawmakers attached provisions regarding increased oversight of natural health products to a budget package instead of considering them as part of a stand-alone bill.
“Our concern is that the powers that are being discussed have not been reviewed and debated,” Mr. Skelton told Dr. Kutcher. “The potential for overreach and unnecessary regulation is significant, and that deserves debate.”
“Profits should not trump Canadians’ health,” answered Dr. Kutcher, who earlier served as head of the psychiatry department at Dalhousie University in Halifax, N.S.
By June, Vanessa’s Law had been expanded with provisions that address natural health products, including the reporting of products that present a serious risk to consumers.
Educating consumers
Many consumers overestimate the level of government regulation of supplements, said Pieter A. Cohen, MD, leader of the Supplement Research Program at Cambridge Health Alliance in Massachusetts. Dr. Cohen was the lead author of the JAMA research letter about melatonin products.
Supplements often share shelves in pharmacies with medicines that are subject to more strict regulation, which causes confusion.
“It’s really hard to wrap your brain around [the fact] that a health product is being sold in pharmacies in the United States and it’s not being vetted by the FDA [U.S. Food and Drug Administration]”, Dr. Cohen said in an interview
The confusion extends across borders. Many consumers in other countries will assume that the FDA performed premarket screening of U.S.-made supplements, but that is not the case, he said.
People who want to take supplements should look for reputable sources of information about them, such as the website of the National Institutes of Health’s Office of Dietary Supplements, Dr. Cohen said. But patients often forget or fail to do this, which can create medical puzzles, such as the case of the woman in the Ontario case study, said Peter Lurie, MD, MPH, executive director of the nonprofit Center for Science in the Public Interest, which has pressed for increased regulation of supplements.
Clinicians need to keep in mind that patients may need prodding to reveal what supplements they are taking, he said.
“They just think of them as different, somehow not the province of the doctor,” Dr. Lurie said. “For others, they are concerned that the doctors will disapprove. So, they hide it.”
A version of this article first appeared on Medscape.com.
FROM THE CANADIAN MEDICAL ASSOCIATION JOURNAL
Unveiling the potential of prediction models in obstetrics
In the dawn of artificial intelligence’s potential to inform clinical practice, the importance of understanding the intent and interpretation of prediction tools is vital. In medicine, informed decision-making promotes patient autonomy and can lead to improved patient satisfaction and engagement in their own care.
This shared understanding enables patients to make informed choices about their care, reducing anxiety and increasing confidence in medical decision-making.
In obstetric clinical practice, prediction tools have been created to assess risk of primary cesarean delivery in gestational diabetes,1 cesarean delivery in hypertensive disorders of pregnancy,2 and failed induction of labor in nulliparous patients with an unfavorable cervix.3 By assessing a patient’s risk profile, clinicians can identify high-risk individuals who may require closer monitoring, early interventions, or specialized care. This allows for more timely interventions to optimize maternal and fetal health outcomes.
Other prediction tools are created to better elucidate to patients their individual risk of an outcome that may be modifiable, aiding physician counseling on mitigating factors to improve overall results. A relevant example is the American Diabetes Association’s risk of type 2 diabetes calculator used for counseling patients on risk reduction. This model includes both preexisting (ethnicity, family history, age, sex assigned at birth) and modifiable risk factors (body mass index, hypertension, physical activity) to predict risk of type 2 diabetes and is widely used in clinical practice to encourage integration of lifestyle changes to decrease risk.4 This model highlights the utility of prediction tools in counseling, providing quantitative data to clinicians to discuss a patient’s individual risk and how to mitigate that risk.
While predictive models clearly have many advantages and potential to improve personalized medicine, concerns have been raised that their interpretation and application can sometimes have unintended consequences as the complexity of these models can lead to variation in understanding among clinicians that impact decision-making. Different clinicians may assign different levels of importance to the predicted risks, resulting in differences in treatment plans and interventions. This variability can lead to disparities in care and outcomes, as patients with similar risk profiles may receive different management approaches based on the interpreting clinician.
Providers may either overly rely on prediction models or completely disregard them, depending on their level of trust or skepticism. Overreliance on prediction models may lead to the neglect of important clinical information or intuition, while disregarding the models may result in missed opportunities for early intervention or appropriate risk stratification. Achieving a balance between clinical judgment and the use of prediction models is crucial for optimal decision-making.
An example of how misinterpretation of the role of prediction tools in patient counseling can have far reaching consequences is the vaginal birth after cesarean (VBAC) calculator where race and ethnicity naturalized racial differences and likely contributed to cesarean overuse in Black pregnant people as non-White race was associated with a decreased chance of successful VBAC. Although the authors of the study that created the VBAC calculator intended it to be used as an adjunct to counseling, institutions and providers used low calculator scores to discourage or prohibit pregnant people from attempting a trial of labor after cesarean (TOLAC). This highlighted the importance of contextualizing the intent of prediction models within the broader clinical setting and individual patient circumstances and preferences.
This gap between intent and interpretation and subsequent application is influenced by individual clinician experience, training, personal biases, and subjective judgment. These subjective elements can introduce inconsistencies and variability in the utilization of prediction tools, leading to potential discrepancies in patient care. Inadequate understanding of prediction models and their statistical concepts can contribute to misinterpretation. It is this bias that prevents prediction models from serving their true purpose: to inform clinical decision-making, improve patient outcomes, and optimize resource allocation.
Clinicians may struggle with concepts such as predictive accuracy, overfitting, calibration, and external validation. Educational initiatives and enhanced training in statistical literacy can empower clinicians to better comprehend and apply prediction models in their practice. Researchers should make it clear that models should not be used in isolation, but rather integrated with clinical expertise and patient preferences. Understanding the limitations of prediction models and incorporating additional clinical information is essential.
Prediction models in obstetrics should undergo continuous evaluation and improvement to enhance their reliability and applicability. Regular updates, external validation, and recalibration are necessary to account for evolving clinical practices, changes in patient populations, and emerging evidence. Engaging clinicians in the evaluation process can foster ownership and promote a sense of trust in the models.
As machine learning and artificial intelligence improve the accuracy of prediction models, there is potential to revolutionize obstetric care by enabling more accurate individualized risk assessment and decision-making. Machine learning has the potential to significantly enhance prediction models in obstetrics by leveraging complex algorithms and advanced computational techniques. However, the unpredictable nature of clinician interpretation poses challenges to the effective utilization of these models.
By emphasizing communication, collaboration, education, and continuous evaluation, we can bridge the gap between prediction models and clinician interpretation that optimizes their use. This concerted effort will ultimately lead to improved patient care, enhanced clinical outcomes, and a more harmonious integration of these tools into obstetric practice.
Dr. Ramos is assistant professor of maternal fetal medicine and associate principal investigator at the Mother Infant Research Institute, Tufts University and Tufts Medical Center, Boston.
References
1. Ramos SZ et al. Predicting primary cesarean delivery in pregnancies complicated by gestational diabetes mellitus. Am J Obstet Gynecol. 2023 Jun 7;S0002-9378(23)00371-X. doi: 10.1016/j.ajog.2023.06.002.
2. Beninati MJ et al. Prediction model for vaginal birth after induction of labor in women with hypertensive disorders of pregnancy. Obstet Gynecol. 2020 Aug;136(2):402-410. doi: 10.1097/AOG.0000000000003938.
3. Levine LD et al. A validated calculator to estimate risk of cesarean after an induction of labor with an unfavorable cervix. Am J Obstet Gynecol. 2018 Feb;218(2):254.e1-254.e7. doi: 10.1016/j.ajog.2017.11.603.
4. American Diabetes Association. Our 60-Second Type 2 Diabetes Risk Test.
In the dawn of artificial intelligence’s potential to inform clinical practice, the importance of understanding the intent and interpretation of prediction tools is vital. In medicine, informed decision-making promotes patient autonomy and can lead to improved patient satisfaction and engagement in their own care.
This shared understanding enables patients to make informed choices about their care, reducing anxiety and increasing confidence in medical decision-making.
In obstetric clinical practice, prediction tools have been created to assess risk of primary cesarean delivery in gestational diabetes,1 cesarean delivery in hypertensive disorders of pregnancy,2 and failed induction of labor in nulliparous patients with an unfavorable cervix.3 By assessing a patient’s risk profile, clinicians can identify high-risk individuals who may require closer monitoring, early interventions, or specialized care. This allows for more timely interventions to optimize maternal and fetal health outcomes.
Other prediction tools are created to better elucidate to patients their individual risk of an outcome that may be modifiable, aiding physician counseling on mitigating factors to improve overall results. A relevant example is the American Diabetes Association’s risk of type 2 diabetes calculator used for counseling patients on risk reduction. This model includes both preexisting (ethnicity, family history, age, sex assigned at birth) and modifiable risk factors (body mass index, hypertension, physical activity) to predict risk of type 2 diabetes and is widely used in clinical practice to encourage integration of lifestyle changes to decrease risk.4 This model highlights the utility of prediction tools in counseling, providing quantitative data to clinicians to discuss a patient’s individual risk and how to mitigate that risk.
While predictive models clearly have many advantages and potential to improve personalized medicine, concerns have been raised that their interpretation and application can sometimes have unintended consequences as the complexity of these models can lead to variation in understanding among clinicians that impact decision-making. Different clinicians may assign different levels of importance to the predicted risks, resulting in differences in treatment plans and interventions. This variability can lead to disparities in care and outcomes, as patients with similar risk profiles may receive different management approaches based on the interpreting clinician.
Providers may either overly rely on prediction models or completely disregard them, depending on their level of trust or skepticism. Overreliance on prediction models may lead to the neglect of important clinical information or intuition, while disregarding the models may result in missed opportunities for early intervention or appropriate risk stratification. Achieving a balance between clinical judgment and the use of prediction models is crucial for optimal decision-making.
An example of how misinterpretation of the role of prediction tools in patient counseling can have far reaching consequences is the vaginal birth after cesarean (VBAC) calculator where race and ethnicity naturalized racial differences and likely contributed to cesarean overuse in Black pregnant people as non-White race was associated with a decreased chance of successful VBAC. Although the authors of the study that created the VBAC calculator intended it to be used as an adjunct to counseling, institutions and providers used low calculator scores to discourage or prohibit pregnant people from attempting a trial of labor after cesarean (TOLAC). This highlighted the importance of contextualizing the intent of prediction models within the broader clinical setting and individual patient circumstances and preferences.
This gap between intent and interpretation and subsequent application is influenced by individual clinician experience, training, personal biases, and subjective judgment. These subjective elements can introduce inconsistencies and variability in the utilization of prediction tools, leading to potential discrepancies in patient care. Inadequate understanding of prediction models and their statistical concepts can contribute to misinterpretation. It is this bias that prevents prediction models from serving their true purpose: to inform clinical decision-making, improve patient outcomes, and optimize resource allocation.
Clinicians may struggle with concepts such as predictive accuracy, overfitting, calibration, and external validation. Educational initiatives and enhanced training in statistical literacy can empower clinicians to better comprehend and apply prediction models in their practice. Researchers should make it clear that models should not be used in isolation, but rather integrated with clinical expertise and patient preferences. Understanding the limitations of prediction models and incorporating additional clinical information is essential.
Prediction models in obstetrics should undergo continuous evaluation and improvement to enhance their reliability and applicability. Regular updates, external validation, and recalibration are necessary to account for evolving clinical practices, changes in patient populations, and emerging evidence. Engaging clinicians in the evaluation process can foster ownership and promote a sense of trust in the models.
As machine learning and artificial intelligence improve the accuracy of prediction models, there is potential to revolutionize obstetric care by enabling more accurate individualized risk assessment and decision-making. Machine learning has the potential to significantly enhance prediction models in obstetrics by leveraging complex algorithms and advanced computational techniques. However, the unpredictable nature of clinician interpretation poses challenges to the effective utilization of these models.
By emphasizing communication, collaboration, education, and continuous evaluation, we can bridge the gap between prediction models and clinician interpretation that optimizes their use. This concerted effort will ultimately lead to improved patient care, enhanced clinical outcomes, and a more harmonious integration of these tools into obstetric practice.
Dr. Ramos is assistant professor of maternal fetal medicine and associate principal investigator at the Mother Infant Research Institute, Tufts University and Tufts Medical Center, Boston.
References
1. Ramos SZ et al. Predicting primary cesarean delivery in pregnancies complicated by gestational diabetes mellitus. Am J Obstet Gynecol. 2023 Jun 7;S0002-9378(23)00371-X. doi: 10.1016/j.ajog.2023.06.002.
2. Beninati MJ et al. Prediction model for vaginal birth after induction of labor in women with hypertensive disorders of pregnancy. Obstet Gynecol. 2020 Aug;136(2):402-410. doi: 10.1097/AOG.0000000000003938.
3. Levine LD et al. A validated calculator to estimate risk of cesarean after an induction of labor with an unfavorable cervix. Am J Obstet Gynecol. 2018 Feb;218(2):254.e1-254.e7. doi: 10.1016/j.ajog.2017.11.603.
4. American Diabetes Association. Our 60-Second Type 2 Diabetes Risk Test.
In the dawn of artificial intelligence’s potential to inform clinical practice, the importance of understanding the intent and interpretation of prediction tools is vital. In medicine, informed decision-making promotes patient autonomy and can lead to improved patient satisfaction and engagement in their own care.
This shared understanding enables patients to make informed choices about their care, reducing anxiety and increasing confidence in medical decision-making.
In obstetric clinical practice, prediction tools have been created to assess risk of primary cesarean delivery in gestational diabetes,1 cesarean delivery in hypertensive disorders of pregnancy,2 and failed induction of labor in nulliparous patients with an unfavorable cervix.3 By assessing a patient’s risk profile, clinicians can identify high-risk individuals who may require closer monitoring, early interventions, or specialized care. This allows for more timely interventions to optimize maternal and fetal health outcomes.
Other prediction tools are created to better elucidate to patients their individual risk of an outcome that may be modifiable, aiding physician counseling on mitigating factors to improve overall results. A relevant example is the American Diabetes Association’s risk of type 2 diabetes calculator used for counseling patients on risk reduction. This model includes both preexisting (ethnicity, family history, age, sex assigned at birth) and modifiable risk factors (body mass index, hypertension, physical activity) to predict risk of type 2 diabetes and is widely used in clinical practice to encourage integration of lifestyle changes to decrease risk.4 This model highlights the utility of prediction tools in counseling, providing quantitative data to clinicians to discuss a patient’s individual risk and how to mitigate that risk.
While predictive models clearly have many advantages and potential to improve personalized medicine, concerns have been raised that their interpretation and application can sometimes have unintended consequences as the complexity of these models can lead to variation in understanding among clinicians that impact decision-making. Different clinicians may assign different levels of importance to the predicted risks, resulting in differences in treatment plans and interventions. This variability can lead to disparities in care and outcomes, as patients with similar risk profiles may receive different management approaches based on the interpreting clinician.
Providers may either overly rely on prediction models or completely disregard them, depending on their level of trust or skepticism. Overreliance on prediction models may lead to the neglect of important clinical information or intuition, while disregarding the models may result in missed opportunities for early intervention or appropriate risk stratification. Achieving a balance between clinical judgment and the use of prediction models is crucial for optimal decision-making.
An example of how misinterpretation of the role of prediction tools in patient counseling can have far reaching consequences is the vaginal birth after cesarean (VBAC) calculator where race and ethnicity naturalized racial differences and likely contributed to cesarean overuse in Black pregnant people as non-White race was associated with a decreased chance of successful VBAC. Although the authors of the study that created the VBAC calculator intended it to be used as an adjunct to counseling, institutions and providers used low calculator scores to discourage or prohibit pregnant people from attempting a trial of labor after cesarean (TOLAC). This highlighted the importance of contextualizing the intent of prediction models within the broader clinical setting and individual patient circumstances and preferences.
This gap between intent and interpretation and subsequent application is influenced by individual clinician experience, training, personal biases, and subjective judgment. These subjective elements can introduce inconsistencies and variability in the utilization of prediction tools, leading to potential discrepancies in patient care. Inadequate understanding of prediction models and their statistical concepts can contribute to misinterpretation. It is this bias that prevents prediction models from serving their true purpose: to inform clinical decision-making, improve patient outcomes, and optimize resource allocation.
Clinicians may struggle with concepts such as predictive accuracy, overfitting, calibration, and external validation. Educational initiatives and enhanced training in statistical literacy can empower clinicians to better comprehend and apply prediction models in their practice. Researchers should make it clear that models should not be used in isolation, but rather integrated with clinical expertise and patient preferences. Understanding the limitations of prediction models and incorporating additional clinical information is essential.
Prediction models in obstetrics should undergo continuous evaluation and improvement to enhance their reliability and applicability. Regular updates, external validation, and recalibration are necessary to account for evolving clinical practices, changes in patient populations, and emerging evidence. Engaging clinicians in the evaluation process can foster ownership and promote a sense of trust in the models.
As machine learning and artificial intelligence improve the accuracy of prediction models, there is potential to revolutionize obstetric care by enabling more accurate individualized risk assessment and decision-making. Machine learning has the potential to significantly enhance prediction models in obstetrics by leveraging complex algorithms and advanced computational techniques. However, the unpredictable nature of clinician interpretation poses challenges to the effective utilization of these models.
By emphasizing communication, collaboration, education, and continuous evaluation, we can bridge the gap between prediction models and clinician interpretation that optimizes their use. This concerted effort will ultimately lead to improved patient care, enhanced clinical outcomes, and a more harmonious integration of these tools into obstetric practice.
Dr. Ramos is assistant professor of maternal fetal medicine and associate principal investigator at the Mother Infant Research Institute, Tufts University and Tufts Medical Center, Boston.
References
1. Ramos SZ et al. Predicting primary cesarean delivery in pregnancies complicated by gestational diabetes mellitus. Am J Obstet Gynecol. 2023 Jun 7;S0002-9378(23)00371-X. doi: 10.1016/j.ajog.2023.06.002.
2. Beninati MJ et al. Prediction model for vaginal birth after induction of labor in women with hypertensive disorders of pregnancy. Obstet Gynecol. 2020 Aug;136(2):402-410. doi: 10.1097/AOG.0000000000003938.
3. Levine LD et al. A validated calculator to estimate risk of cesarean after an induction of labor with an unfavorable cervix. Am J Obstet Gynecol. 2018 Feb;218(2):254.e1-254.e7. doi: 10.1016/j.ajog.2017.11.603.
4. American Diabetes Association. Our 60-Second Type 2 Diabetes Risk Test.
Dural-puncture epidural drives faster conversion to cesarean anesthesia
DPE, while not new, has become more popular as an option for initiating labor analgesia, but data comparing DPE with standard epidural in conversion to surgical anesthesia for cesarean deliveries are limited, Nadir Sharawi, MD, of the University of Arkansas for Medical Sciences, Little Rock, and colleagues wrote.
DPE involves no injection of intrathecal drugs, and the potential advantages include easier translocation of epidural medications into the intrathecal space for improved analgesia, but the effects of DPE on the onset and reliability of surgical anesthesia remain unknown, they said.
In a study published in JAMA Network Open, the researchers randomized 70 women scheduled for cesarean delivery of singleton pregnancies to DPE and 70 to a standard epidural. The participants were aged 18 years and older, with a mean age of the 30.1 years; the study was conducted between April 2019 and October 2022 at a single center.
The primary outcome was the time to the loss of sharp sensation at T6, defined as “the start of epidural extension anesthesia (time zero on the stopwatch) to when the patient could no longer feel sharp sensation at T6 (assessed bilaterally at the midclavicular line),” the researchers wrote.
The onset time to surgical anesthesia was faster in the DPE group, compared with the standard group, with a median of 422 seconds versus 655 seconds.
A key secondary outcome was a composite measure of the quality of epidural anesthesia that included failure to achieve a T10 bilateral block preoperatively in the delivery room, failure to achieve a surgical block at T6 within 15 minutes of chloroprocaine administration, requirement for intraoperative analgesia, repeat neuraxial procedure, and conversion to general anesthesia. The composite rates of lower quality anesthesia were significantly less in the DPE group, compared with the standard group (15.7% vs. 36.3%; P = .007).
Additional secondary outcomes included maternal satisfaction and pain score during surgery, adverse events, opioid use in the first 24 hours, maternal vasopressor requirements, epidural block assessments, and neonatal outcomes. No significant differences in these outcomes were noted between the groups, and no instances of local anesthetic systemic toxicity or neurological complications were reported.
The findings were limited by several factors including the study population of women scheduled for cesarean delivery and not in labor, and the inability to detect less frequent complications such as post–dural-puncture headache and accidental dural puncture, the researchers noted.
In addition, the results may vary with the use of other combinations of local anesthetics and opioids. “Chloroprocaine was chosen in this study because of its ease of administration without the need for opioids and other additives along with the low risk of systemic toxic effects, which favors rapid administration for emergent cesarean delivery,” they wrote.
However, the results show an association between DPE within an hour of epidural extension for elective cesarean delivery and a faster onset of anesthesia, improved block quality, and a more favorable ratio of risks versus benefits, compared with the use of standard epidural, the researchers concluded.
No need for general anesthesia?
“There is controversy over whether the dural puncture epidural technique improves labor analgesia when compared to a standard epidural,” Dr. Shawari said in an interview. “However, there are limited data on whether the dural puncture epidural technique decreases the onset time to surgical anesthesia when compared to a standard epidural for cesarean delivery. This is important as a pre-existing epidural is commonly used to convert labor analgesia to surgical anesthesia in the setting of urgent cesarean delivery. A faster onset of epidural anesthesia could potentially avoid the need for general anesthesia in an emergency.”
The researchers were not surprised by the findings given their experience with performing dural puncture epidurals for labor analgesia, Dr. Shawari said. In those cases, DPE provided a faster onset when converting cesarean anesthesia, compared with a standard epidural.
The takeaway from the current study is that DPE also provided “a faster onset and improved quality of anesthesia when compared to standard epidural for elective cesarean delivery,” Dr. Shawari said. However, additional research is needed to confirm the findings for intrapartum cesarean delivery.
Progress in improving pain control
“Adequate pain control during cesarean delivery is incredibly important,” Catherine Albright, MD, a maternal-fetal medicine specialist at the University of Washington, Seattle, said in an interview. “Inadequate pain control leads to the need to provide additional intravenous medications or the need to be put under general anesthesia, which changes the birth experience and is more dangerous for the birthing person and the neonate.
“In my clinical experience, there are many times when patients do not have adequate pain control during a cesarean delivery,” said Dr. Albright, who was not involved in the current study. “I am pleased to see that there is research underway about how to best manage pain on labor and delivery, especially in the setting of conversion from labor anesthesia to cesarean anesthesia.”
The findings may have implications for clinical practice, said Dr. Albright. If the dural puncture epidural can improve cesarean anesthesia following an epidural during labor, rather than anesthesia provided for an elective cesarean), “then I believe it would reduce the number of patients who require additional pain medication, have a poor cesarean experience, and/or need to be put under general anesthesia.”
However, “as noted by the authors, additional research is needed to further determine possible risks and side effects from this technique, and also to ensure that it also works in the setting of labor, rather than for an elective cesarean,” Dr. Albright added.
The study received no outside funding. The researchers had no financial conflicts to disclose. Dr. Albright had no financial conflicts to disclose.
DPE, while not new, has become more popular as an option for initiating labor analgesia, but data comparing DPE with standard epidural in conversion to surgical anesthesia for cesarean deliveries are limited, Nadir Sharawi, MD, of the University of Arkansas for Medical Sciences, Little Rock, and colleagues wrote.
DPE involves no injection of intrathecal drugs, and the potential advantages include easier translocation of epidural medications into the intrathecal space for improved analgesia, but the effects of DPE on the onset and reliability of surgical anesthesia remain unknown, they said.
In a study published in JAMA Network Open, the researchers randomized 70 women scheduled for cesarean delivery of singleton pregnancies to DPE and 70 to a standard epidural. The participants were aged 18 years and older, with a mean age of the 30.1 years; the study was conducted between April 2019 and October 2022 at a single center.
The primary outcome was the time to the loss of sharp sensation at T6, defined as “the start of epidural extension anesthesia (time zero on the stopwatch) to when the patient could no longer feel sharp sensation at T6 (assessed bilaterally at the midclavicular line),” the researchers wrote.
The onset time to surgical anesthesia was faster in the DPE group, compared with the standard group, with a median of 422 seconds versus 655 seconds.
A key secondary outcome was a composite measure of the quality of epidural anesthesia that included failure to achieve a T10 bilateral block preoperatively in the delivery room, failure to achieve a surgical block at T6 within 15 minutes of chloroprocaine administration, requirement for intraoperative analgesia, repeat neuraxial procedure, and conversion to general anesthesia. The composite rates of lower quality anesthesia were significantly less in the DPE group, compared with the standard group (15.7% vs. 36.3%; P = .007).
Additional secondary outcomes included maternal satisfaction and pain score during surgery, adverse events, opioid use in the first 24 hours, maternal vasopressor requirements, epidural block assessments, and neonatal outcomes. No significant differences in these outcomes were noted between the groups, and no instances of local anesthetic systemic toxicity or neurological complications were reported.
The findings were limited by several factors including the study population of women scheduled for cesarean delivery and not in labor, and the inability to detect less frequent complications such as post–dural-puncture headache and accidental dural puncture, the researchers noted.
In addition, the results may vary with the use of other combinations of local anesthetics and opioids. “Chloroprocaine was chosen in this study because of its ease of administration without the need for opioids and other additives along with the low risk of systemic toxic effects, which favors rapid administration for emergent cesarean delivery,” they wrote.
However, the results show an association between DPE within an hour of epidural extension for elective cesarean delivery and a faster onset of anesthesia, improved block quality, and a more favorable ratio of risks versus benefits, compared with the use of standard epidural, the researchers concluded.
No need for general anesthesia?
“There is controversy over whether the dural puncture epidural technique improves labor analgesia when compared to a standard epidural,” Dr. Shawari said in an interview. “However, there are limited data on whether the dural puncture epidural technique decreases the onset time to surgical anesthesia when compared to a standard epidural for cesarean delivery. This is important as a pre-existing epidural is commonly used to convert labor analgesia to surgical anesthesia in the setting of urgent cesarean delivery. A faster onset of epidural anesthesia could potentially avoid the need for general anesthesia in an emergency.”
The researchers were not surprised by the findings given their experience with performing dural puncture epidurals for labor analgesia, Dr. Shawari said. In those cases, DPE provided a faster onset when converting cesarean anesthesia, compared with a standard epidural.
The takeaway from the current study is that DPE also provided “a faster onset and improved quality of anesthesia when compared to standard epidural for elective cesarean delivery,” Dr. Shawari said. However, additional research is needed to confirm the findings for intrapartum cesarean delivery.
Progress in improving pain control
“Adequate pain control during cesarean delivery is incredibly important,” Catherine Albright, MD, a maternal-fetal medicine specialist at the University of Washington, Seattle, said in an interview. “Inadequate pain control leads to the need to provide additional intravenous medications or the need to be put under general anesthesia, which changes the birth experience and is more dangerous for the birthing person and the neonate.
“In my clinical experience, there are many times when patients do not have adequate pain control during a cesarean delivery,” said Dr. Albright, who was not involved in the current study. “I am pleased to see that there is research underway about how to best manage pain on labor and delivery, especially in the setting of conversion from labor anesthesia to cesarean anesthesia.”
The findings may have implications for clinical practice, said Dr. Albright. If the dural puncture epidural can improve cesarean anesthesia following an epidural during labor, rather than anesthesia provided for an elective cesarean), “then I believe it would reduce the number of patients who require additional pain medication, have a poor cesarean experience, and/or need to be put under general anesthesia.”
However, “as noted by the authors, additional research is needed to further determine possible risks and side effects from this technique, and also to ensure that it also works in the setting of labor, rather than for an elective cesarean,” Dr. Albright added.
The study received no outside funding. The researchers had no financial conflicts to disclose. Dr. Albright had no financial conflicts to disclose.
DPE, while not new, has become more popular as an option for initiating labor analgesia, but data comparing DPE with standard epidural in conversion to surgical anesthesia for cesarean deliveries are limited, Nadir Sharawi, MD, of the University of Arkansas for Medical Sciences, Little Rock, and colleagues wrote.
DPE involves no injection of intrathecal drugs, and the potential advantages include easier translocation of epidural medications into the intrathecal space for improved analgesia, but the effects of DPE on the onset and reliability of surgical anesthesia remain unknown, they said.
In a study published in JAMA Network Open, the researchers randomized 70 women scheduled for cesarean delivery of singleton pregnancies to DPE and 70 to a standard epidural. The participants were aged 18 years and older, with a mean age of the 30.1 years; the study was conducted between April 2019 and October 2022 at a single center.
The primary outcome was the time to the loss of sharp sensation at T6, defined as “the start of epidural extension anesthesia (time zero on the stopwatch) to when the patient could no longer feel sharp sensation at T6 (assessed bilaterally at the midclavicular line),” the researchers wrote.
The onset time to surgical anesthesia was faster in the DPE group, compared with the standard group, with a median of 422 seconds versus 655 seconds.
A key secondary outcome was a composite measure of the quality of epidural anesthesia that included failure to achieve a T10 bilateral block preoperatively in the delivery room, failure to achieve a surgical block at T6 within 15 minutes of chloroprocaine administration, requirement for intraoperative analgesia, repeat neuraxial procedure, and conversion to general anesthesia. The composite rates of lower quality anesthesia were significantly less in the DPE group, compared with the standard group (15.7% vs. 36.3%; P = .007).
Additional secondary outcomes included maternal satisfaction and pain score during surgery, adverse events, opioid use in the first 24 hours, maternal vasopressor requirements, epidural block assessments, and neonatal outcomes. No significant differences in these outcomes were noted between the groups, and no instances of local anesthetic systemic toxicity or neurological complications were reported.
The findings were limited by several factors including the study population of women scheduled for cesarean delivery and not in labor, and the inability to detect less frequent complications such as post–dural-puncture headache and accidental dural puncture, the researchers noted.
In addition, the results may vary with the use of other combinations of local anesthetics and opioids. “Chloroprocaine was chosen in this study because of its ease of administration without the need for opioids and other additives along with the low risk of systemic toxic effects, which favors rapid administration for emergent cesarean delivery,” they wrote.
However, the results show an association between DPE within an hour of epidural extension for elective cesarean delivery and a faster onset of anesthesia, improved block quality, and a more favorable ratio of risks versus benefits, compared with the use of standard epidural, the researchers concluded.
No need for general anesthesia?
“There is controversy over whether the dural puncture epidural technique improves labor analgesia when compared to a standard epidural,” Dr. Shawari said in an interview. “However, there are limited data on whether the dural puncture epidural technique decreases the onset time to surgical anesthesia when compared to a standard epidural for cesarean delivery. This is important as a pre-existing epidural is commonly used to convert labor analgesia to surgical anesthesia in the setting of urgent cesarean delivery. A faster onset of epidural anesthesia could potentially avoid the need for general anesthesia in an emergency.”
The researchers were not surprised by the findings given their experience with performing dural puncture epidurals for labor analgesia, Dr. Shawari said. In those cases, DPE provided a faster onset when converting cesarean anesthesia, compared with a standard epidural.
The takeaway from the current study is that DPE also provided “a faster onset and improved quality of anesthesia when compared to standard epidural for elective cesarean delivery,” Dr. Shawari said. However, additional research is needed to confirm the findings for intrapartum cesarean delivery.
Progress in improving pain control
“Adequate pain control during cesarean delivery is incredibly important,” Catherine Albright, MD, a maternal-fetal medicine specialist at the University of Washington, Seattle, said in an interview. “Inadequate pain control leads to the need to provide additional intravenous medications or the need to be put under general anesthesia, which changes the birth experience and is more dangerous for the birthing person and the neonate.
“In my clinical experience, there are many times when patients do not have adequate pain control during a cesarean delivery,” said Dr. Albright, who was not involved in the current study. “I am pleased to see that there is research underway about how to best manage pain on labor and delivery, especially in the setting of conversion from labor anesthesia to cesarean anesthesia.”
The findings may have implications for clinical practice, said Dr. Albright. If the dural puncture epidural can improve cesarean anesthesia following an epidural during labor, rather than anesthesia provided for an elective cesarean), “then I believe it would reduce the number of patients who require additional pain medication, have a poor cesarean experience, and/or need to be put under general anesthesia.”
However, “as noted by the authors, additional research is needed to further determine possible risks and side effects from this technique, and also to ensure that it also works in the setting of labor, rather than for an elective cesarean,” Dr. Albright added.
The study received no outside funding. The researchers had no financial conflicts to disclose. Dr. Albright had no financial conflicts to disclose.
FROM JAMA NETWORK OPEN
FDA approves first pill for postpartum depression
a condition that affects an estimated one in seven mothers in the United States.
The pill, zuranolone (Zurzuvae), is a neuroactive steroid that acts on GABAA receptors in the brain responsible for regulating mood, arousal, behavior, and cognition, according to Biogen, which, along with Sage Therapeutics, developed the product. The recommended dose for Zurzuvae is 50 mg taken once daily for 14 days, in the evening with a fatty meal, according to the FDA.
Postpartum depression often goes undiagnosed and untreated. Many mothers are hesitant to reveal their symptoms to family and clinicians, fearing they’ll be judged on their parenting. A 2017 study found that suicide accounted for roughly 5% of perinatal deaths among women in Canada, with most of those deaths occurring in the first 3 months in the year after giving birth.
“Postpartum depression is a serious and potentially life-threatening condition in which women experience sadness, guilt, worthlessness – even, in severe cases, thoughts of harming themselves or their child. And, because postpartum depression can disrupt the maternal-infant bond, it can also have consequences for the child’s physical and emotional development,” Tiffany R. Farchione, MD, director of the division of psychiatry at the FDA’s Center for Drug Evaluation and Research, said in a statement about the approval. “Having access to an oral medication will be a beneficial option for many of these women coping with extreme, and sometimes life-threatening, feelings.”
The other approved therapy for postpartum depression is the intravenous agent brexanolone (Zulresso; Sage). But the product requires prolonged infusions in hospital settings and costs $34,000.
FDA approval of Zurzuvae was based in part on data reported in a 2023 study in the American Journal of Psychiatry, which showed that the drug led to significantly greater improvement in depressive symptoms at 15 days compared with the placebo group. Improvements were observed on day 3, the earliest assessment, and were sustained at all subsequent visits during the treatment and follow-up period (through day 42).
Patients with anxiety who received the active drug experienced improvement in related symptoms compared with the patients who received a placebo.
The most common adverse events reported in the trial were somnolence and headaches. Weight gain, sexual dysfunction, withdrawal symptoms, and increased suicidal ideation or behavior were not observed.
The packaging for Zurzuvae will include a boxed warning noting that the drug can affect a user’s ability to drive and perform other potentially hazardous activities, possibly without their knowledge of the impairment, the FDA said. As a result, people who use Zurzuvae should not drive or operate heavy machinery for at least 12 hours after taking the pill.
A version of this article first appeared on Medscape.com.
a condition that affects an estimated one in seven mothers in the United States.
The pill, zuranolone (Zurzuvae), is a neuroactive steroid that acts on GABAA receptors in the brain responsible for regulating mood, arousal, behavior, and cognition, according to Biogen, which, along with Sage Therapeutics, developed the product. The recommended dose for Zurzuvae is 50 mg taken once daily for 14 days, in the evening with a fatty meal, according to the FDA.
Postpartum depression often goes undiagnosed and untreated. Many mothers are hesitant to reveal their symptoms to family and clinicians, fearing they’ll be judged on their parenting. A 2017 study found that suicide accounted for roughly 5% of perinatal deaths among women in Canada, with most of those deaths occurring in the first 3 months in the year after giving birth.
“Postpartum depression is a serious and potentially life-threatening condition in which women experience sadness, guilt, worthlessness – even, in severe cases, thoughts of harming themselves or their child. And, because postpartum depression can disrupt the maternal-infant bond, it can also have consequences for the child’s physical and emotional development,” Tiffany R. Farchione, MD, director of the division of psychiatry at the FDA’s Center for Drug Evaluation and Research, said in a statement about the approval. “Having access to an oral medication will be a beneficial option for many of these women coping with extreme, and sometimes life-threatening, feelings.”
The other approved therapy for postpartum depression is the intravenous agent brexanolone (Zulresso; Sage). But the product requires prolonged infusions in hospital settings and costs $34,000.
FDA approval of Zurzuvae was based in part on data reported in a 2023 study in the American Journal of Psychiatry, which showed that the drug led to significantly greater improvement in depressive symptoms at 15 days compared with the placebo group. Improvements were observed on day 3, the earliest assessment, and were sustained at all subsequent visits during the treatment and follow-up period (through day 42).
Patients with anxiety who received the active drug experienced improvement in related symptoms compared with the patients who received a placebo.
The most common adverse events reported in the trial were somnolence and headaches. Weight gain, sexual dysfunction, withdrawal symptoms, and increased suicidal ideation or behavior were not observed.
The packaging for Zurzuvae will include a boxed warning noting that the drug can affect a user’s ability to drive and perform other potentially hazardous activities, possibly without their knowledge of the impairment, the FDA said. As a result, people who use Zurzuvae should not drive or operate heavy machinery for at least 12 hours after taking the pill.
A version of this article first appeared on Medscape.com.
a condition that affects an estimated one in seven mothers in the United States.
The pill, zuranolone (Zurzuvae), is a neuroactive steroid that acts on GABAA receptors in the brain responsible for regulating mood, arousal, behavior, and cognition, according to Biogen, which, along with Sage Therapeutics, developed the product. The recommended dose for Zurzuvae is 50 mg taken once daily for 14 days, in the evening with a fatty meal, according to the FDA.
Postpartum depression often goes undiagnosed and untreated. Many mothers are hesitant to reveal their symptoms to family and clinicians, fearing they’ll be judged on their parenting. A 2017 study found that suicide accounted for roughly 5% of perinatal deaths among women in Canada, with most of those deaths occurring in the first 3 months in the year after giving birth.
“Postpartum depression is a serious and potentially life-threatening condition in which women experience sadness, guilt, worthlessness – even, in severe cases, thoughts of harming themselves or their child. And, because postpartum depression can disrupt the maternal-infant bond, it can also have consequences for the child’s physical and emotional development,” Tiffany R. Farchione, MD, director of the division of psychiatry at the FDA’s Center for Drug Evaluation and Research, said in a statement about the approval. “Having access to an oral medication will be a beneficial option for many of these women coping with extreme, and sometimes life-threatening, feelings.”
The other approved therapy for postpartum depression is the intravenous agent brexanolone (Zulresso; Sage). But the product requires prolonged infusions in hospital settings and costs $34,000.
FDA approval of Zurzuvae was based in part on data reported in a 2023 study in the American Journal of Psychiatry, which showed that the drug led to significantly greater improvement in depressive symptoms at 15 days compared with the placebo group. Improvements were observed on day 3, the earliest assessment, and were sustained at all subsequent visits during the treatment and follow-up period (through day 42).
Patients with anxiety who received the active drug experienced improvement in related symptoms compared with the patients who received a placebo.
The most common adverse events reported in the trial were somnolence and headaches. Weight gain, sexual dysfunction, withdrawal symptoms, and increased suicidal ideation or behavior were not observed.
The packaging for Zurzuvae will include a boxed warning noting that the drug can affect a user’s ability to drive and perform other potentially hazardous activities, possibly without their knowledge of the impairment, the FDA said. As a result, people who use Zurzuvae should not drive or operate heavy machinery for at least 12 hours after taking the pill.
A version of this article first appeared on Medscape.com.
Trends in prepregnancy diabetes rates in the United States, 2016 -2021
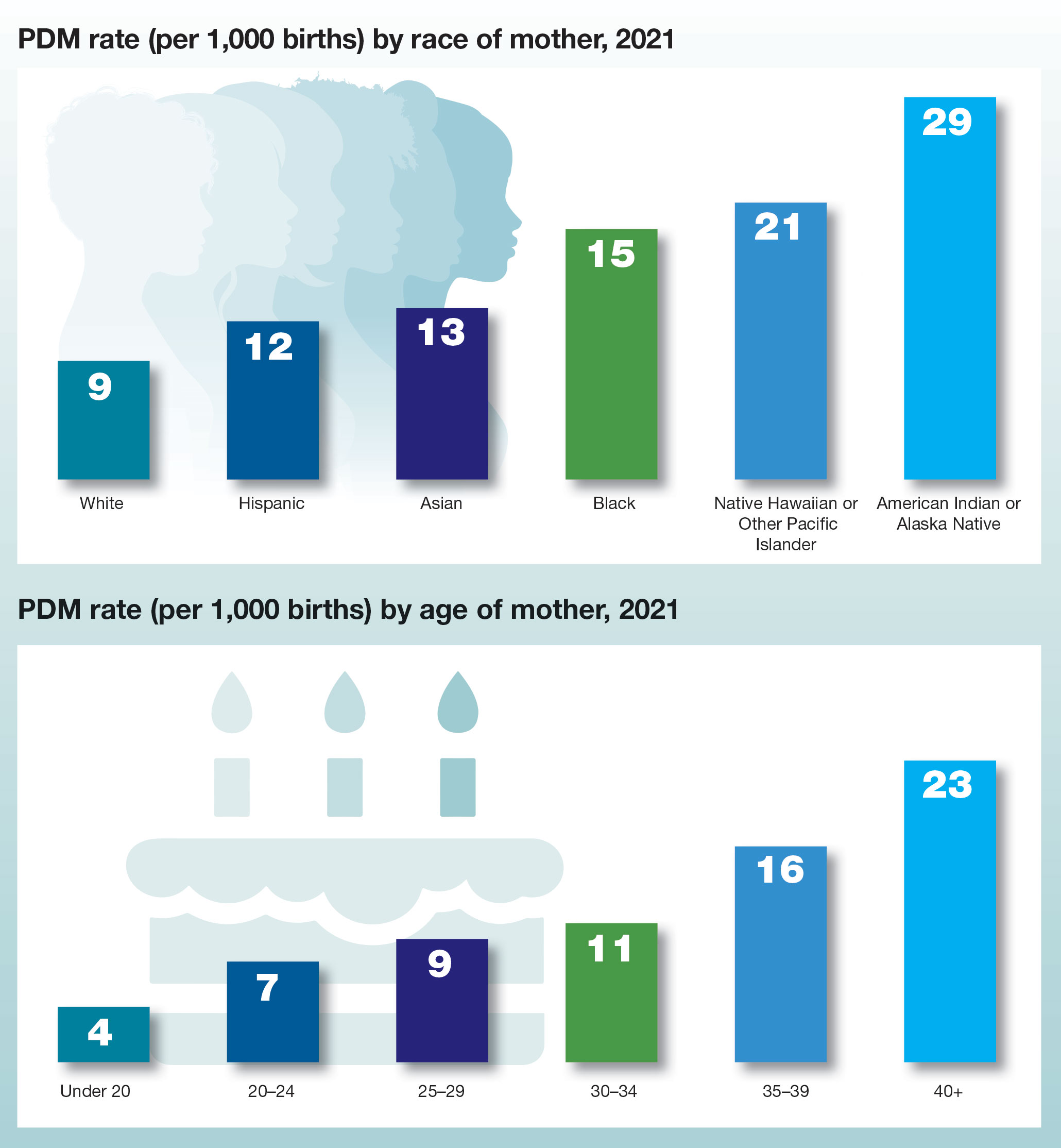


Recurrent pregnancy loss and inherited thrombophilias: Does low molecular weight heparin improve the live birth rate?
Quenby S, Booth K, Hiller L, et al; ALIFE2 Block Writing Committee and ALIFE2 Investigators. Heparin for women with recurrent miscarriage and inherited thrombophilia (ALIFE2): an international open-label, randomised controlled trial. Lancet. 2023;402:54-61. doi:10.1016/S0140-6736(23)00693-1.
EXPERT COMMENTARY
“Follow the evidence to where it leads, even if the conclusion is uncomfortable.”
—Steven James, author
Women with RPL have endured overzealous evaluations and management despite a lack of proven efficacy. From alloimmune testing that results in paternal leukocyte immunization1 and the long-entrusted metroplasty for a septate uterus recently put under fire2 to the “hammer and nail” approach of preimplantation genetic testing for embryo aneuploid screening,3 patients have been subjected to unsubstantiated treatments.
While the evaluation of RPL has evolved, guidelines from the American Society for Reproductive Medicine (ASRM), American College of Obstetricians and Gynecologists (ACOG), and Royal College of Obstetricians and Gynaecologists (RCOG) do not recommend testing for inherited thrombophilias outside of a history for venous thromboembolism.4-6 These 3 societies support treating acquired thrombophilias that represent the antiphospholipid antibody syndrome.
Citing insufficient evidence for reducing adverse pregnancy outcomes, ACOG recommends the use of prophylactic- or intermediate-dose LMWH or unfractionated heparin (UFH) for patients with “high-risk” thrombophilias only to prevent venous thromboembolism during pregnancy and continuing postpartum.4 (High-risk thrombophilias are defined as factor V Leiden homozygosity, prothrombin gene G20210A mutation homozygosity, heterozygosity for both factor V Leiden homozygosity and prothrombin gene G20210A mutation, or an antithrombin deficiency.4)
To determine the impact of LMWH treatment versus no treatment on live birth rate, Quenby and colleagues conducted a prospective randomized controlled trial of women with RPL and inherited thrombophilias (the ALIFE2 trial). This was a follow-up to their 2010 randomized controlled trial that demonstrated no effect of LMWH with low-dose aspirin versus low-dose aspirin alone compared with placebo in women with unexplained RPL.7
 PHOTO: BETAVERSO/SHUTTERSTOCK
PHOTO: BETAVERSO/SHUTTERSTOCK

Continue to: Details of the study...
Details of the study
The ALIFE2 study took place over 8 years and involved 5 countries, including the United States, with the 2 main centers in the Netherlands and the United Kingdom. Women eligible for the study were aged 18 to 42 years, had an inherited thrombophilia (confirmed by 2 tests), experienced recurrent miscarriages (2 or more consecutive miscarriages, nonconsecutive miscarriages, or intrauterine fetal deaths, irrespective of gestational age), and were less than 7 weeks’ estimated gestational age. Study patients were randomly allocated with a positive pregnancy test to either surveillance or LMWH treatment, which was continued throughout pregnancy.
The primary outcome was live birth rate, and secondary outcomes were a history of miscarriage, ectopic pregnancy, and obstetric complications. A total of 164 women were allocated to LMWH plus standard care, and 162 women to standard care alone. LMWH was shown to be safe without major/minor bleeding or maternal heparin-induced thrombocytopenia.
The statistical calculation was by “intention to treat,” which considers all enrolled participants, including those who dropped out of the study, as opposed to a “per protocol” analysis in which only patients who completed the study were analyzed.
Results. Primary outcome data were available for 320 participants. Of the 162 women in the LMWH-treated group, 116 (72%) had live birth rates, as did 112 (71%) of 158 in the standard care group. There was no significant difference between groups (OR, 1.04; 95% CI, 0.64–1.68).
Study strengths and limitations
The outcome of the ALIFE2 study is consistent with that of a Cochrane review that found insufficient evidence for improved live birth rate in patients with RPL and inherited thrombophilias treated with LMWH versus low-dose aspirin. Of their review of the studies at low risk of bias, only 1 was placebo controlled.8
This study by Quenby and colleagues was well designed and ensured a sufficient number of enrolled participants to comply with their power analysis. However, by beginning LMWH at 7 weeks’ gestation, patients may not have received a therapeutic benefit as opposed to initiation of treatment with a positive pregnancy test. The authors did not describe when testing for thrombophilias occurred or explain the protocol and reason for repeat testing.
Study limitations included a deviation from protocol in the standard care group, which was the initiation of LMWH after 7 weeks’ gestation. In the standard care group, 30 participants received LMWH, 18 of whom started heparin treatment before 12 weeks of gestation. The other 12 participants received LMWH after 12 weeks’ gestation, and 6 of those 12 started after 28 weeks’ gestation, since they were determined to need LMWH for thromboprophylaxis according to RCOG guidelines. While this had the potential to influence outcomes, only 18 of 162 (11%) patients were involved.
The authors did not define RPL based on a clinical versus a biochemical pregnancy loss as the latter is more common and is without agreed upon criteria for testing. Additionally, a lack of patient masking to medication could play an undetermined role in affecting the outcome. ●
This elegant, and vital, randomized controlled trial provides double take-home messages: There is no value in testing for inherited thrombophilias in RPL, as they occur in a similar prevalence in the general population, and there is no significant difference in live birth rate from LMWH treatment in women with RPL and inherited thrombophilias compared with surveillance. Consequently, the increased cost of medication and testing can be averted.
MARK P. TROLICE, MD, MBA
- Wong LF, Porter TF, Scott JR. Immunotherapy for recurrent miscarriage. Cochrane Database Syst Rev. 2014; CD000112. doi:10.1002/14651858.CD000112
- Trolice MP. The septate uterus and metroplasty—another dogma under siege. Fertil Steril. 2021;116:693-694. doi:10.1016/j.fertnstert.2021.06.063
- Dahdouh EM, Balayla J, Garcia-Velasco JA, et al. PGT-A for recurrent pregnancy loss: evidence is growing but the issue is not resolved. Hum Reprod. 2021;36:2805-2806. doi:10.1093 /humrep/deab194
- American College of Obstetricians and Gynecologists Committee on Practice Bulletins–Obstetrics. ACOG practice bulletin no. 197: inherited thrombophilias in pregnancy. Obstet Gynecol. 2018;132:e18-e34. doi:10.1097 /AOG.0000000000002703
- Practice Committee of the American Society for Reproductive Medicine. Evaluation and treatment of recurrent pregnancy loss: a committee opinion. Fertil Steril. 2012;98:1103-1111. doi:10.1016/j.fertnstert.2012.06.048
- Regan L, Rai R, Saravelos S, et al; Royal College of Obstetricians and Gynaecologists. Recurrent Miscarriage Green‐top Guideline No. 17. BJOG. June 19, 2023. doi:10.1111/1471 -0528.17515
- Kaandorp SP, Goddijn M, van der Post JA, et al. Aspirin plus heparin or aspirin alone in women with recurrent miscarriage. N Engl J Med. 2010;362:1586-1596. doi:10.1056 /NEJMoa1000641
- de Jong PG, Kaandorp S, Di Nisio M, et al. Aspirin and/or heparin for women with unexplained recurrent miscarriage with or without inherited thrombophilia. Cochrane Database Syst Rev. 2014;CD004734. doi:10.1002/14651858.CD004734 .pub4
Quenby S, Booth K, Hiller L, et al; ALIFE2 Block Writing Committee and ALIFE2 Investigators. Heparin for women with recurrent miscarriage and inherited thrombophilia (ALIFE2): an international open-label, randomised controlled trial. Lancet. 2023;402:54-61. doi:10.1016/S0140-6736(23)00693-1.
EXPERT COMMENTARY
“Follow the evidence to where it leads, even if the conclusion is uncomfortable.”
—Steven James, author
Women with RPL have endured overzealous evaluations and management despite a lack of proven efficacy. From alloimmune testing that results in paternal leukocyte immunization1 and the long-entrusted metroplasty for a septate uterus recently put under fire2 to the “hammer and nail” approach of preimplantation genetic testing for embryo aneuploid screening,3 patients have been subjected to unsubstantiated treatments.
While the evaluation of RPL has evolved, guidelines from the American Society for Reproductive Medicine (ASRM), American College of Obstetricians and Gynecologists (ACOG), and Royal College of Obstetricians and Gynaecologists (RCOG) do not recommend testing for inherited thrombophilias outside of a history for venous thromboembolism.4-6 These 3 societies support treating acquired thrombophilias that represent the antiphospholipid antibody syndrome.
Citing insufficient evidence for reducing adverse pregnancy outcomes, ACOG recommends the use of prophylactic- or intermediate-dose LMWH or unfractionated heparin (UFH) for patients with “high-risk” thrombophilias only to prevent venous thromboembolism during pregnancy and continuing postpartum.4 (High-risk thrombophilias are defined as factor V Leiden homozygosity, prothrombin gene G20210A mutation homozygosity, heterozygosity for both factor V Leiden homozygosity and prothrombin gene G20210A mutation, or an antithrombin deficiency.4)
To determine the impact of LMWH treatment versus no treatment on live birth rate, Quenby and colleagues conducted a prospective randomized controlled trial of women with RPL and inherited thrombophilias (the ALIFE2 trial). This was a follow-up to their 2010 randomized controlled trial that demonstrated no effect of LMWH with low-dose aspirin versus low-dose aspirin alone compared with placebo in women with unexplained RPL.7
 PHOTO: BETAVERSO/SHUTTERSTOCK
PHOTO: BETAVERSO/SHUTTERSTOCK

Continue to: Details of the study...
Details of the study
The ALIFE2 study took place over 8 years and involved 5 countries, including the United States, with the 2 main centers in the Netherlands and the United Kingdom. Women eligible for the study were aged 18 to 42 years, had an inherited thrombophilia (confirmed by 2 tests), experienced recurrent miscarriages (2 or more consecutive miscarriages, nonconsecutive miscarriages, or intrauterine fetal deaths, irrespective of gestational age), and were less than 7 weeks’ estimated gestational age. Study patients were randomly allocated with a positive pregnancy test to either surveillance or LMWH treatment, which was continued throughout pregnancy.
The primary outcome was live birth rate, and secondary outcomes were a history of miscarriage, ectopic pregnancy, and obstetric complications. A total of 164 women were allocated to LMWH plus standard care, and 162 women to standard care alone. LMWH was shown to be safe without major/minor bleeding or maternal heparin-induced thrombocytopenia.
The statistical calculation was by “intention to treat,” which considers all enrolled participants, including those who dropped out of the study, as opposed to a “per protocol” analysis in which only patients who completed the study were analyzed.
Results. Primary outcome data were available for 320 participants. Of the 162 women in the LMWH-treated group, 116 (72%) had live birth rates, as did 112 (71%) of 158 in the standard care group. There was no significant difference between groups (OR, 1.04; 95% CI, 0.64–1.68).
Study strengths and limitations
The outcome of the ALIFE2 study is consistent with that of a Cochrane review that found insufficient evidence for improved live birth rate in patients with RPL and inherited thrombophilias treated with LMWH versus low-dose aspirin. Of their review of the studies at low risk of bias, only 1 was placebo controlled.8
This study by Quenby and colleagues was well designed and ensured a sufficient number of enrolled participants to comply with their power analysis. However, by beginning LMWH at 7 weeks’ gestation, patients may not have received a therapeutic benefit as opposed to initiation of treatment with a positive pregnancy test. The authors did not describe when testing for thrombophilias occurred or explain the protocol and reason for repeat testing.
Study limitations included a deviation from protocol in the standard care group, which was the initiation of LMWH after 7 weeks’ gestation. In the standard care group, 30 participants received LMWH, 18 of whom started heparin treatment before 12 weeks of gestation. The other 12 participants received LMWH after 12 weeks’ gestation, and 6 of those 12 started after 28 weeks’ gestation, since they were determined to need LMWH for thromboprophylaxis according to RCOG guidelines. While this had the potential to influence outcomes, only 18 of 162 (11%) patients were involved.
The authors did not define RPL based on a clinical versus a biochemical pregnancy loss as the latter is more common and is without agreed upon criteria for testing. Additionally, a lack of patient masking to medication could play an undetermined role in affecting the outcome. ●
This elegant, and vital, randomized controlled trial provides double take-home messages: There is no value in testing for inherited thrombophilias in RPL, as they occur in a similar prevalence in the general population, and there is no significant difference in live birth rate from LMWH treatment in women with RPL and inherited thrombophilias compared with surveillance. Consequently, the increased cost of medication and testing can be averted.
MARK P. TROLICE, MD, MBA
Quenby S, Booth K, Hiller L, et al; ALIFE2 Block Writing Committee and ALIFE2 Investigators. Heparin for women with recurrent miscarriage and inherited thrombophilia (ALIFE2): an international open-label, randomised controlled trial. Lancet. 2023;402:54-61. doi:10.1016/S0140-6736(23)00693-1.
EXPERT COMMENTARY
“Follow the evidence to where it leads, even if the conclusion is uncomfortable.”
—Steven James, author
Women with RPL have endured overzealous evaluations and management despite a lack of proven efficacy. From alloimmune testing that results in paternal leukocyte immunization1 and the long-entrusted metroplasty for a septate uterus recently put under fire2 to the “hammer and nail” approach of preimplantation genetic testing for embryo aneuploid screening,3 patients have been subjected to unsubstantiated treatments.
While the evaluation of RPL has evolved, guidelines from the American Society for Reproductive Medicine (ASRM), American College of Obstetricians and Gynecologists (ACOG), and Royal College of Obstetricians and Gynaecologists (RCOG) do not recommend testing for inherited thrombophilias outside of a history for venous thromboembolism.4-6 These 3 societies support treating acquired thrombophilias that represent the antiphospholipid antibody syndrome.
Citing insufficient evidence for reducing adverse pregnancy outcomes, ACOG recommends the use of prophylactic- or intermediate-dose LMWH or unfractionated heparin (UFH) for patients with “high-risk” thrombophilias only to prevent venous thromboembolism during pregnancy and continuing postpartum.4 (High-risk thrombophilias are defined as factor V Leiden homozygosity, prothrombin gene G20210A mutation homozygosity, heterozygosity for both factor V Leiden homozygosity and prothrombin gene G20210A mutation, or an antithrombin deficiency.4)
To determine the impact of LMWH treatment versus no treatment on live birth rate, Quenby and colleagues conducted a prospective randomized controlled trial of women with RPL and inherited thrombophilias (the ALIFE2 trial). This was a follow-up to their 2010 randomized controlled trial that demonstrated no effect of LMWH with low-dose aspirin versus low-dose aspirin alone compared with placebo in women with unexplained RPL.7
 PHOTO: BETAVERSO/SHUTTERSTOCK
PHOTO: BETAVERSO/SHUTTERSTOCK

Continue to: Details of the study...
Details of the study
The ALIFE2 study took place over 8 years and involved 5 countries, including the United States, with the 2 main centers in the Netherlands and the United Kingdom. Women eligible for the study were aged 18 to 42 years, had an inherited thrombophilia (confirmed by 2 tests), experienced recurrent miscarriages (2 or more consecutive miscarriages, nonconsecutive miscarriages, or intrauterine fetal deaths, irrespective of gestational age), and were less than 7 weeks’ estimated gestational age. Study patients were randomly allocated with a positive pregnancy test to either surveillance or LMWH treatment, which was continued throughout pregnancy.
The primary outcome was live birth rate, and secondary outcomes were a history of miscarriage, ectopic pregnancy, and obstetric complications. A total of 164 women were allocated to LMWH plus standard care, and 162 women to standard care alone. LMWH was shown to be safe without major/minor bleeding or maternal heparin-induced thrombocytopenia.
The statistical calculation was by “intention to treat,” which considers all enrolled participants, including those who dropped out of the study, as opposed to a “per protocol” analysis in which only patients who completed the study were analyzed.
Results. Primary outcome data were available for 320 participants. Of the 162 women in the LMWH-treated group, 116 (72%) had live birth rates, as did 112 (71%) of 158 in the standard care group. There was no significant difference between groups (OR, 1.04; 95% CI, 0.64–1.68).
Study strengths and limitations
The outcome of the ALIFE2 study is consistent with that of a Cochrane review that found insufficient evidence for improved live birth rate in patients with RPL and inherited thrombophilias treated with LMWH versus low-dose aspirin. Of their review of the studies at low risk of bias, only 1 was placebo controlled.8
This study by Quenby and colleagues was well designed and ensured a sufficient number of enrolled participants to comply with their power analysis. However, by beginning LMWH at 7 weeks’ gestation, patients may not have received a therapeutic benefit as opposed to initiation of treatment with a positive pregnancy test. The authors did not describe when testing for thrombophilias occurred or explain the protocol and reason for repeat testing.
Study limitations included a deviation from protocol in the standard care group, which was the initiation of LMWH after 7 weeks’ gestation. In the standard care group, 30 participants received LMWH, 18 of whom started heparin treatment before 12 weeks of gestation. The other 12 participants received LMWH after 12 weeks’ gestation, and 6 of those 12 started after 28 weeks’ gestation, since they were determined to need LMWH for thromboprophylaxis according to RCOG guidelines. While this had the potential to influence outcomes, only 18 of 162 (11%) patients were involved.
The authors did not define RPL based on a clinical versus a biochemical pregnancy loss as the latter is more common and is without agreed upon criteria for testing. Additionally, a lack of patient masking to medication could play an undetermined role in affecting the outcome. ●
This elegant, and vital, randomized controlled trial provides double take-home messages: There is no value in testing for inherited thrombophilias in RPL, as they occur in a similar prevalence in the general population, and there is no significant difference in live birth rate from LMWH treatment in women with RPL and inherited thrombophilias compared with surveillance. Consequently, the increased cost of medication and testing can be averted.
MARK P. TROLICE, MD, MBA
- Wong LF, Porter TF, Scott JR. Immunotherapy for recurrent miscarriage. Cochrane Database Syst Rev. 2014; CD000112. doi:10.1002/14651858.CD000112
- Trolice MP. The septate uterus and metroplasty—another dogma under siege. Fertil Steril. 2021;116:693-694. doi:10.1016/j.fertnstert.2021.06.063
- Dahdouh EM, Balayla J, Garcia-Velasco JA, et al. PGT-A for recurrent pregnancy loss: evidence is growing but the issue is not resolved. Hum Reprod. 2021;36:2805-2806. doi:10.1093 /humrep/deab194
- American College of Obstetricians and Gynecologists Committee on Practice Bulletins–Obstetrics. ACOG practice bulletin no. 197: inherited thrombophilias in pregnancy. Obstet Gynecol. 2018;132:e18-e34. doi:10.1097 /AOG.0000000000002703
- Practice Committee of the American Society for Reproductive Medicine. Evaluation and treatment of recurrent pregnancy loss: a committee opinion. Fertil Steril. 2012;98:1103-1111. doi:10.1016/j.fertnstert.2012.06.048
- Regan L, Rai R, Saravelos S, et al; Royal College of Obstetricians and Gynaecologists. Recurrent Miscarriage Green‐top Guideline No. 17. BJOG. June 19, 2023. doi:10.1111/1471 -0528.17515
- Kaandorp SP, Goddijn M, van der Post JA, et al. Aspirin plus heparin or aspirin alone in women with recurrent miscarriage. N Engl J Med. 2010;362:1586-1596. doi:10.1056 /NEJMoa1000641
- de Jong PG, Kaandorp S, Di Nisio M, et al. Aspirin and/or heparin for women with unexplained recurrent miscarriage with or without inherited thrombophilia. Cochrane Database Syst Rev. 2014;CD004734. doi:10.1002/14651858.CD004734 .pub4
- Wong LF, Porter TF, Scott JR. Immunotherapy for recurrent miscarriage. Cochrane Database Syst Rev. 2014; CD000112. doi:10.1002/14651858.CD000112
- Trolice MP. The septate uterus and metroplasty—another dogma under siege. Fertil Steril. 2021;116:693-694. doi:10.1016/j.fertnstert.2021.06.063
- Dahdouh EM, Balayla J, Garcia-Velasco JA, et al. PGT-A for recurrent pregnancy loss: evidence is growing but the issue is not resolved. Hum Reprod. 2021;36:2805-2806. doi:10.1093 /humrep/deab194
- American College of Obstetricians and Gynecologists Committee on Practice Bulletins–Obstetrics. ACOG practice bulletin no. 197: inherited thrombophilias in pregnancy. Obstet Gynecol. 2018;132:e18-e34. doi:10.1097 /AOG.0000000000002703
- Practice Committee of the American Society for Reproductive Medicine. Evaluation and treatment of recurrent pregnancy loss: a committee opinion. Fertil Steril. 2012;98:1103-1111. doi:10.1016/j.fertnstert.2012.06.048
- Regan L, Rai R, Saravelos S, et al; Royal College of Obstetricians and Gynaecologists. Recurrent Miscarriage Green‐top Guideline No. 17. BJOG. June 19, 2023. doi:10.1111/1471 -0528.17515
- Kaandorp SP, Goddijn M, van der Post JA, et al. Aspirin plus heparin or aspirin alone in women with recurrent miscarriage. N Engl J Med. 2010;362:1586-1596. doi:10.1056 /NEJMoa1000641
- de Jong PG, Kaandorp S, Di Nisio M, et al. Aspirin and/or heparin for women with unexplained recurrent miscarriage with or without inherited thrombophilia. Cochrane Database Syst Rev. 2014;CD004734. doi:10.1002/14651858.CD004734 .pub4
U.S. maternal mortality crisis grows, yet deaths seem preventable
On June 2, 2019, 35-year-old Anne Hutchinson gave birth to her first child, Lillian. There were no problems with the pregnancy or the birth at Fairview Hospital, which is part of the Cleveland Clinic system.
But 2 days after the birth, she had shortness of breath and couldn’t lie down and breathe.
“My mom’s a nurse, and she was like, ‘You need to go to the hospital immediately,’” Ms. Hutchinson said. When she was admitted to the hospital, there were suddenly “10 doctors in the room.”
Ms. Hutchinson was diagnosed with peripartum cardiomyopathy, a weakness of the heart muscle. She had heart failure. The seriousness of heart failure is measured by the ejection fraction, or the percentage of blood the heart pumps out. Normal is 50%-70%. Ms. Hutchinson’s ejection fraction was 20%.
She was put on medication, left the hospital after 5 days, and her ejection fraction eventually rose to 35%. But she was still at risk for sudden cardiac death.
“The cardiologist said to me, ‘You probably can’t have any more children.’ My heart did not bounce back,” Ms. Hutchinson said.
By the end of 2019, her cardiologist determined that she needed an internal cardiac defibrillator, which monitors the heartbeat and delivers electric shocks to restore the heart’s normal rhythm when needed.
By 2020, when Ms. Hutchinson’s ejection fraction was near normal, she decided that she wanted another child.
“I had a daughter. She was beautiful and amazing. But I felt like I wanted to have a sibling for her,” she says. Yet when her cardiologist at Fairview Hospital heard the plan, she told her getting pregnant again “would be like Russian roulette.”
Ms. Hutchinson is one of a growing number of women whose medical condition puts them at high risk of death during and after giving birth. An estimated 30% of maternal deaths in the United States result from cardiovascular disease – a problem that has become more common with increases in diabetes and obesity.
And, in some women, hypertension can develop suddenly during pregnancy. This is called preeclampsia and is increasing in the United States, particularly in Black women. In rare cases, it can become the life-threatening condition eclampsia, with seizures and death.
Three-time Olympic medalist and world champion sprinter Tori Bowie was found dead in June of apparent complications of pregnancy. The medical examiner’s office in Orange County, Fla., said she was believed to have been in her 8th month of pregnancy and may have died of eclampsia.
Heart conditions in pregnant women are one of a long list of reasons why the United States has the highest maternal mortality rate of any developed country. But the risk is marked by significant racial differences, with death rates three times higher in Black women, compared with White women.
What troubles many experts is that it is estimated that 80% of these deaths are preventable.
“That is a ridiculous number,” said Melissa A. Simon, MD, MPH, director of the Center for Health Equity Transformation at Northwestern Medicine in Evanston, Ill.. “For a health care system in a country that is so high-resourced and high-income, for 8 out of 10 deaths for moms who are pregnant [to be preventable], that’s absolutely unacceptable.”
Pregnant women are not only at risk of death from cardiovascular complications, but other types of problems, including hemorrhage, thrombotic embolism, and infection.
But experts now are focusing attention on nonmedical reasons for maternal mortality, such as racial disparities and the fundamental issue of whether women are telling doctors about their symptoms but are not being heard.
The government has acknowledged the depth of this problem with the Centers for Disease Control and Prevention’s “Hear Her” campaign, which includes videos of women who describe how their health professionals did not take their concerns seriously.
In one such video, a woman named Sanari says 2 days after the birth of her second child, she started developing soreness.
“By day 3, it just didn’t feel right. I asked the nurses, explained my symptoms and that I was having crazy pains, and they assured me it was just gas,” she says on the video.
Sanari described how she started to have odorous discharge and ended up in an emergency room at a different hospital. Health care providers found a large abscess on her uterus.
“I’m glad I didn’t stop at no, and I’m glad someone finally heard me – someone finally listened to me,” she said.
“Hear Her” featured another woman named Lindsay, who had preeclampsia in her first pregnancy and began to get symptoms during her second pregnancy.
She describes how she voiced her concerns to her doctors, saying, “sometimes it would be, ‘Oh, you’re pregnant and your feet are supposed to swell. … It’s just fine.’ But I didn’t feel fine.”
The campaign aims to raise awareness of warning signs that require fast medical attention to prevent pregnancy-related deaths.
But Shanna Cox, associate director of the CDC’s Division of Reproductive Health, said the agency has collected many stories of women who died or nearly died because their concerns were not being addressed properly.
Ms. Cox says another part of the campaign “is really focused on health care providers and listening … to their patients, providing that respectful patient-centered care to be sure that all their concerns are addressed.”
And some experts believe the thinking has shifted even more dramatically.
“We’ve moved from beyond the days of blaming the individual, the birth person, or the woman, to say you haven’t done this, you haven’t come into health care, you are not taking care of yourself, you aren’t keeping your appointments,” says Laurie Zephyrin, MD, MPH, vice president of the Commonwealth Fund, a private foundation in New York dedicated to improving health care.
Dr. Zephyrin says the health care system falls short of providing equitable, quality care. “There’s data that shows Black people receive worse care than White people for about 40% of quality measures,” she said.
These disparities have led to the formation of organizations like National Birth Equity Collaborative, an advocacy group in New Orleans working to improve maternal care for Black patients.
Carmen Green, vice president of research and strategy, said institutional racism has been embedded into some health care providers.
“They have this hierarchy that teaches them they have to manage, they have to control, they have to direct the medical experience, and that is just not how birthing works,” she said.
She used the example of the birth experience as a car ride, where the mothers have been in the backseat with the doctor driving. “We want the birthing person in the driving seat and want [them] to be respected as a person who is deciding where that destination is going,” Ms. Green said.
She says health providers often “blame the mamas” based on assumptions, stereotypes, and biases against low-income people.
So how is American medicine responding to the medical and social causes of maternal mortality?
This news organization surveyed 10 medical centers ranked by U.S. News & World Report as the country’s top facilities for obstetrics. They were asked what programs they had and studies they had done to try to reduce maternal mortality, improve racial disparities, and target cardiovascular causes of maternal mortality.
One of the most extensive programs was founded at the Stanford School of Medicine in Stanford, Calif., in 2006. The California Maternal Quality Care Collaborative includes 200 hospitals in the state committed to ending preventable maternal mortality and racial disparities.
Nine hospitals in the collaborative have started programs to reduce hemorrhages, manage high blood pressure disorders, and reduce the rate of cesarean deliveries. All are important reasons for maternal mortality.
These programs helped bring about a 62% reduction in California’s maternal mortality rate from 2006 to 2016. And 2023 figures show that California has the lowest maternal mortality rate of any state.
Alabama has the sixth highest rate of maternal mortality in the nation. The University of Alabama at Birmingham wants to address the racial disparities in maternal mortality with a cooperative called the P3 EQUATE Network.
The network is part of a $20 million program by the American Heart Association to gain greater understanding of the disproportionate effect of maternal mortality on Black and Native American people.
The program works with pregnant and postpartum women “to discover ways to reduce racism and social problems that contribute to poor health outcomes.”
In addition to collaborative efforts, the survey found maternal mortality programs at all the top medical centers.
NewYork-Presbyterian Hospital has a Mothers Center that provides specialized care to pregnant women with complications.
The University of Chicago Medical Center established a program called “Systematic Treatment and Management of Postpartum Hypertension” that includes patient and staff education, standardized hospital discharge instructions, and a follow-up in a postpartum hypertension clinic.
A 2021 study found that the program had helped increase the number of postpartum women who correctly follow blood pressure control guidance.
A program called MOMS Navigation at Northwell Health in Long Island, N.Y., provides support to high-risk mothers. The program decreased 30-day readmission rates for all patients by 50% and for Black birthing patients by 60%. Reducing readmission is an important measure for reducing complications.
Vanderbilt University Medical Center in Nashville has what it calls the first-of-its-kind educational podcasts Healthy Mom Healthy Baby, where 30% of the content is devoted to health disparities.
And several centers, including Brigham and Women’s Hospital in Boston and NewYork-Presbyterian, make sure mothers have access to doulas – professional support people trained in the needs of the family during pregnancy and childbirth.
The survey found that 9 of the 10 centers have obstetric programs devoted to cardiac care, including the University of Chicago, Stanford Medicine, UCLA, and the Cleveland Clinic.
But the survey results raise the question: How can we have these programs and research at our best obstetrics centers devoted to reducing maternal mortality and have the highest rate of all developed countries?
“Maternal mortality largely falls on pregnant and birthing persons who do not intersect with, nor are touched, by the best obstetrical care centers in the country,” Dr. Simon said.
Unfortunately, she said, the pregnant people who face “high maternal mortality rates … face all the access-to-care barriers and do not have the privilege of birthing or accessing care at top centers.”
Anne Hutchinson believed going to a top center – the Cleveland Clinic – would give her a good chance of safely delivering a second child.
Karlee Hoffman, DO, a cardiologist in the hospital’s cardio-obstetric high-risk clinic, said Ms. Hutchinson “came to me, she was determined to have another child, and she said, ‘Please help me do this. I’m doing it regardless. So, I would really like your support in moving forward,’” Dr. Hoffman recalls.
Ms. Hutchinson said Cleveland Clinic doctors told her she had a 20%-30% chance of peripartum cardiomyopathy again if she had a second child. If that happened, the risks “ranged from mild decompensation of my heart function to death,“ she said.
Ms. Hutchinson and her husband decided to go ahead with the pregnancy. Her parents cried when they found out. But Ms. Hutchinson says she was confident in the cardio obstetric team at Cleveland Clinic.
Her fertility medicine raised the possibility of multiple births, which would be a definite threat to her life. Her heart failure medicine, Entresto, could not be used during pregnancy, so her doctors put her on older medicines.
She got pregnant in June 2022 and developed gestational diabetes, which can affect pregnancy because of raised blood sugar. Another potential risk. She was carefully monitored by the specialists and hospitalized once.
At 37 weeks, she was induced and had a forceps delivery. On Feb. 15, 2023, her second daughter, Charlotte, was born.
Ms. Hutchinson was asked to write about how she felt when she delivered Charlotte:
“I am not sure how to put into words the love, joy, and elation that I felt holding Charlotte for the first time. As I write this, I have tears of joy in my eyes thinking of that moment. I had prayed for her for so long and after being told I couldn’t or shouldn’t have any more children.”
“I felt that Charlotte and I were forever bonded in triumph from that moment on. We did it and made it out alive! And our family was now complete. I have so much joy watching the love that is growing between Charlotte and Lillian. Life is truly amazing, and I am forever grateful to have them.”
A version of this article first appeared on WebMD.com.
On June 2, 2019, 35-year-old Anne Hutchinson gave birth to her first child, Lillian. There were no problems with the pregnancy or the birth at Fairview Hospital, which is part of the Cleveland Clinic system.
But 2 days after the birth, she had shortness of breath and couldn’t lie down and breathe.
“My mom’s a nurse, and she was like, ‘You need to go to the hospital immediately,’” Ms. Hutchinson said. When she was admitted to the hospital, there were suddenly “10 doctors in the room.”
Ms. Hutchinson was diagnosed with peripartum cardiomyopathy, a weakness of the heart muscle. She had heart failure. The seriousness of heart failure is measured by the ejection fraction, or the percentage of blood the heart pumps out. Normal is 50%-70%. Ms. Hutchinson’s ejection fraction was 20%.
She was put on medication, left the hospital after 5 days, and her ejection fraction eventually rose to 35%. But she was still at risk for sudden cardiac death.
“The cardiologist said to me, ‘You probably can’t have any more children.’ My heart did not bounce back,” Ms. Hutchinson said.
By the end of 2019, her cardiologist determined that she needed an internal cardiac defibrillator, which monitors the heartbeat and delivers electric shocks to restore the heart’s normal rhythm when needed.
By 2020, when Ms. Hutchinson’s ejection fraction was near normal, she decided that she wanted another child.
“I had a daughter. She was beautiful and amazing. But I felt like I wanted to have a sibling for her,” she says. Yet when her cardiologist at Fairview Hospital heard the plan, she told her getting pregnant again “would be like Russian roulette.”
Ms. Hutchinson is one of a growing number of women whose medical condition puts them at high risk of death during and after giving birth. An estimated 30% of maternal deaths in the United States result from cardiovascular disease – a problem that has become more common with increases in diabetes and obesity.
And, in some women, hypertension can develop suddenly during pregnancy. This is called preeclampsia and is increasing in the United States, particularly in Black women. In rare cases, it can become the life-threatening condition eclampsia, with seizures and death.
Three-time Olympic medalist and world champion sprinter Tori Bowie was found dead in June of apparent complications of pregnancy. The medical examiner’s office in Orange County, Fla., said she was believed to have been in her 8th month of pregnancy and may have died of eclampsia.
Heart conditions in pregnant women are one of a long list of reasons why the United States has the highest maternal mortality rate of any developed country. But the risk is marked by significant racial differences, with death rates three times higher in Black women, compared with White women.
What troubles many experts is that it is estimated that 80% of these deaths are preventable.
“That is a ridiculous number,” said Melissa A. Simon, MD, MPH, director of the Center for Health Equity Transformation at Northwestern Medicine in Evanston, Ill.. “For a health care system in a country that is so high-resourced and high-income, for 8 out of 10 deaths for moms who are pregnant [to be preventable], that’s absolutely unacceptable.”
Pregnant women are not only at risk of death from cardiovascular complications, but other types of problems, including hemorrhage, thrombotic embolism, and infection.
But experts now are focusing attention on nonmedical reasons for maternal mortality, such as racial disparities and the fundamental issue of whether women are telling doctors about their symptoms but are not being heard.
The government has acknowledged the depth of this problem with the Centers for Disease Control and Prevention’s “Hear Her” campaign, which includes videos of women who describe how their health professionals did not take their concerns seriously.
In one such video, a woman named Sanari says 2 days after the birth of her second child, she started developing soreness.
“By day 3, it just didn’t feel right. I asked the nurses, explained my symptoms and that I was having crazy pains, and they assured me it was just gas,” she says on the video.
Sanari described how she started to have odorous discharge and ended up in an emergency room at a different hospital. Health care providers found a large abscess on her uterus.
“I’m glad I didn’t stop at no, and I’m glad someone finally heard me – someone finally listened to me,” she said.
“Hear Her” featured another woman named Lindsay, who had preeclampsia in her first pregnancy and began to get symptoms during her second pregnancy.
She describes how she voiced her concerns to her doctors, saying, “sometimes it would be, ‘Oh, you’re pregnant and your feet are supposed to swell. … It’s just fine.’ But I didn’t feel fine.”
The campaign aims to raise awareness of warning signs that require fast medical attention to prevent pregnancy-related deaths.
But Shanna Cox, associate director of the CDC’s Division of Reproductive Health, said the agency has collected many stories of women who died or nearly died because their concerns were not being addressed properly.
Ms. Cox says another part of the campaign “is really focused on health care providers and listening … to their patients, providing that respectful patient-centered care to be sure that all their concerns are addressed.”
And some experts believe the thinking has shifted even more dramatically.
“We’ve moved from beyond the days of blaming the individual, the birth person, or the woman, to say you haven’t done this, you haven’t come into health care, you are not taking care of yourself, you aren’t keeping your appointments,” says Laurie Zephyrin, MD, MPH, vice president of the Commonwealth Fund, a private foundation in New York dedicated to improving health care.
Dr. Zephyrin says the health care system falls short of providing equitable, quality care. “There’s data that shows Black people receive worse care than White people for about 40% of quality measures,” she said.
These disparities have led to the formation of organizations like National Birth Equity Collaborative, an advocacy group in New Orleans working to improve maternal care for Black patients.
Carmen Green, vice president of research and strategy, said institutional racism has been embedded into some health care providers.
“They have this hierarchy that teaches them they have to manage, they have to control, they have to direct the medical experience, and that is just not how birthing works,” she said.
She used the example of the birth experience as a car ride, where the mothers have been in the backseat with the doctor driving. “We want the birthing person in the driving seat and want [them] to be respected as a person who is deciding where that destination is going,” Ms. Green said.
She says health providers often “blame the mamas” based on assumptions, stereotypes, and biases against low-income people.
So how is American medicine responding to the medical and social causes of maternal mortality?
This news organization surveyed 10 medical centers ranked by U.S. News & World Report as the country’s top facilities for obstetrics. They were asked what programs they had and studies they had done to try to reduce maternal mortality, improve racial disparities, and target cardiovascular causes of maternal mortality.
One of the most extensive programs was founded at the Stanford School of Medicine in Stanford, Calif., in 2006. The California Maternal Quality Care Collaborative includes 200 hospitals in the state committed to ending preventable maternal mortality and racial disparities.
Nine hospitals in the collaborative have started programs to reduce hemorrhages, manage high blood pressure disorders, and reduce the rate of cesarean deliveries. All are important reasons for maternal mortality.
These programs helped bring about a 62% reduction in California’s maternal mortality rate from 2006 to 2016. And 2023 figures show that California has the lowest maternal mortality rate of any state.
Alabama has the sixth highest rate of maternal mortality in the nation. The University of Alabama at Birmingham wants to address the racial disparities in maternal mortality with a cooperative called the P3 EQUATE Network.
The network is part of a $20 million program by the American Heart Association to gain greater understanding of the disproportionate effect of maternal mortality on Black and Native American people.
The program works with pregnant and postpartum women “to discover ways to reduce racism and social problems that contribute to poor health outcomes.”
In addition to collaborative efforts, the survey found maternal mortality programs at all the top medical centers.
NewYork-Presbyterian Hospital has a Mothers Center that provides specialized care to pregnant women with complications.
The University of Chicago Medical Center established a program called “Systematic Treatment and Management of Postpartum Hypertension” that includes patient and staff education, standardized hospital discharge instructions, and a follow-up in a postpartum hypertension clinic.
A 2021 study found that the program had helped increase the number of postpartum women who correctly follow blood pressure control guidance.
A program called MOMS Navigation at Northwell Health in Long Island, N.Y., provides support to high-risk mothers. The program decreased 30-day readmission rates for all patients by 50% and for Black birthing patients by 60%. Reducing readmission is an important measure for reducing complications.
Vanderbilt University Medical Center in Nashville has what it calls the first-of-its-kind educational podcasts Healthy Mom Healthy Baby, where 30% of the content is devoted to health disparities.
And several centers, including Brigham and Women’s Hospital in Boston and NewYork-Presbyterian, make sure mothers have access to doulas – professional support people trained in the needs of the family during pregnancy and childbirth.
The survey found that 9 of the 10 centers have obstetric programs devoted to cardiac care, including the University of Chicago, Stanford Medicine, UCLA, and the Cleveland Clinic.
But the survey results raise the question: How can we have these programs and research at our best obstetrics centers devoted to reducing maternal mortality and have the highest rate of all developed countries?
“Maternal mortality largely falls on pregnant and birthing persons who do not intersect with, nor are touched, by the best obstetrical care centers in the country,” Dr. Simon said.
Unfortunately, she said, the pregnant people who face “high maternal mortality rates … face all the access-to-care barriers and do not have the privilege of birthing or accessing care at top centers.”
Anne Hutchinson believed going to a top center – the Cleveland Clinic – would give her a good chance of safely delivering a second child.
Karlee Hoffman, DO, a cardiologist in the hospital’s cardio-obstetric high-risk clinic, said Ms. Hutchinson “came to me, she was determined to have another child, and she said, ‘Please help me do this. I’m doing it regardless. So, I would really like your support in moving forward,’” Dr. Hoffman recalls.
Ms. Hutchinson said Cleveland Clinic doctors told her she had a 20%-30% chance of peripartum cardiomyopathy again if she had a second child. If that happened, the risks “ranged from mild decompensation of my heart function to death,“ she said.
Ms. Hutchinson and her husband decided to go ahead with the pregnancy. Her parents cried when they found out. But Ms. Hutchinson says she was confident in the cardio obstetric team at Cleveland Clinic.
Her fertility medicine raised the possibility of multiple births, which would be a definite threat to her life. Her heart failure medicine, Entresto, could not be used during pregnancy, so her doctors put her on older medicines.
She got pregnant in June 2022 and developed gestational diabetes, which can affect pregnancy because of raised blood sugar. Another potential risk. She was carefully monitored by the specialists and hospitalized once.
At 37 weeks, she was induced and had a forceps delivery. On Feb. 15, 2023, her second daughter, Charlotte, was born.
Ms. Hutchinson was asked to write about how she felt when she delivered Charlotte:
“I am not sure how to put into words the love, joy, and elation that I felt holding Charlotte for the first time. As I write this, I have tears of joy in my eyes thinking of that moment. I had prayed for her for so long and after being told I couldn’t or shouldn’t have any more children.”
“I felt that Charlotte and I were forever bonded in triumph from that moment on. We did it and made it out alive! And our family was now complete. I have so much joy watching the love that is growing between Charlotte and Lillian. Life is truly amazing, and I am forever grateful to have them.”
A version of this article first appeared on WebMD.com.
On June 2, 2019, 35-year-old Anne Hutchinson gave birth to her first child, Lillian. There were no problems with the pregnancy or the birth at Fairview Hospital, which is part of the Cleveland Clinic system.
But 2 days after the birth, she had shortness of breath and couldn’t lie down and breathe.
“My mom’s a nurse, and she was like, ‘You need to go to the hospital immediately,’” Ms. Hutchinson said. When she was admitted to the hospital, there were suddenly “10 doctors in the room.”
Ms. Hutchinson was diagnosed with peripartum cardiomyopathy, a weakness of the heart muscle. She had heart failure. The seriousness of heart failure is measured by the ejection fraction, or the percentage of blood the heart pumps out. Normal is 50%-70%. Ms. Hutchinson’s ejection fraction was 20%.
She was put on medication, left the hospital after 5 days, and her ejection fraction eventually rose to 35%. But she was still at risk for sudden cardiac death.
“The cardiologist said to me, ‘You probably can’t have any more children.’ My heart did not bounce back,” Ms. Hutchinson said.
By the end of 2019, her cardiologist determined that she needed an internal cardiac defibrillator, which monitors the heartbeat and delivers electric shocks to restore the heart’s normal rhythm when needed.
By 2020, when Ms. Hutchinson’s ejection fraction was near normal, she decided that she wanted another child.
“I had a daughter. She was beautiful and amazing. But I felt like I wanted to have a sibling for her,” she says. Yet when her cardiologist at Fairview Hospital heard the plan, she told her getting pregnant again “would be like Russian roulette.”
Ms. Hutchinson is one of a growing number of women whose medical condition puts them at high risk of death during and after giving birth. An estimated 30% of maternal deaths in the United States result from cardiovascular disease – a problem that has become more common with increases in diabetes and obesity.
And, in some women, hypertension can develop suddenly during pregnancy. This is called preeclampsia and is increasing in the United States, particularly in Black women. In rare cases, it can become the life-threatening condition eclampsia, with seizures and death.
Three-time Olympic medalist and world champion sprinter Tori Bowie was found dead in June of apparent complications of pregnancy. The medical examiner’s office in Orange County, Fla., said she was believed to have been in her 8th month of pregnancy and may have died of eclampsia.
Heart conditions in pregnant women are one of a long list of reasons why the United States has the highest maternal mortality rate of any developed country. But the risk is marked by significant racial differences, with death rates three times higher in Black women, compared with White women.
What troubles many experts is that it is estimated that 80% of these deaths are preventable.
“That is a ridiculous number,” said Melissa A. Simon, MD, MPH, director of the Center for Health Equity Transformation at Northwestern Medicine in Evanston, Ill.. “For a health care system in a country that is so high-resourced and high-income, for 8 out of 10 deaths for moms who are pregnant [to be preventable], that’s absolutely unacceptable.”
Pregnant women are not only at risk of death from cardiovascular complications, but other types of problems, including hemorrhage, thrombotic embolism, and infection.
But experts now are focusing attention on nonmedical reasons for maternal mortality, such as racial disparities and the fundamental issue of whether women are telling doctors about their symptoms but are not being heard.
The government has acknowledged the depth of this problem with the Centers for Disease Control and Prevention’s “Hear Her” campaign, which includes videos of women who describe how their health professionals did not take their concerns seriously.
In one such video, a woman named Sanari says 2 days after the birth of her second child, she started developing soreness.
“By day 3, it just didn’t feel right. I asked the nurses, explained my symptoms and that I was having crazy pains, and they assured me it was just gas,” she says on the video.
Sanari described how she started to have odorous discharge and ended up in an emergency room at a different hospital. Health care providers found a large abscess on her uterus.
“I’m glad I didn’t stop at no, and I’m glad someone finally heard me – someone finally listened to me,” she said.
“Hear Her” featured another woman named Lindsay, who had preeclampsia in her first pregnancy and began to get symptoms during her second pregnancy.
She describes how she voiced her concerns to her doctors, saying, “sometimes it would be, ‘Oh, you’re pregnant and your feet are supposed to swell. … It’s just fine.’ But I didn’t feel fine.”
The campaign aims to raise awareness of warning signs that require fast medical attention to prevent pregnancy-related deaths.
But Shanna Cox, associate director of the CDC’s Division of Reproductive Health, said the agency has collected many stories of women who died or nearly died because their concerns were not being addressed properly.
Ms. Cox says another part of the campaign “is really focused on health care providers and listening … to their patients, providing that respectful patient-centered care to be sure that all their concerns are addressed.”
And some experts believe the thinking has shifted even more dramatically.
“We’ve moved from beyond the days of blaming the individual, the birth person, or the woman, to say you haven’t done this, you haven’t come into health care, you are not taking care of yourself, you aren’t keeping your appointments,” says Laurie Zephyrin, MD, MPH, vice president of the Commonwealth Fund, a private foundation in New York dedicated to improving health care.
Dr. Zephyrin says the health care system falls short of providing equitable, quality care. “There’s data that shows Black people receive worse care than White people for about 40% of quality measures,” she said.
These disparities have led to the formation of organizations like National Birth Equity Collaborative, an advocacy group in New Orleans working to improve maternal care for Black patients.
Carmen Green, vice president of research and strategy, said institutional racism has been embedded into some health care providers.
“They have this hierarchy that teaches them they have to manage, they have to control, they have to direct the medical experience, and that is just not how birthing works,” she said.
She used the example of the birth experience as a car ride, where the mothers have been in the backseat with the doctor driving. “We want the birthing person in the driving seat and want [them] to be respected as a person who is deciding where that destination is going,” Ms. Green said.
She says health providers often “blame the mamas” based on assumptions, stereotypes, and biases against low-income people.
So how is American medicine responding to the medical and social causes of maternal mortality?
This news organization surveyed 10 medical centers ranked by U.S. News & World Report as the country’s top facilities for obstetrics. They were asked what programs they had and studies they had done to try to reduce maternal mortality, improve racial disparities, and target cardiovascular causes of maternal mortality.
One of the most extensive programs was founded at the Stanford School of Medicine in Stanford, Calif., in 2006. The California Maternal Quality Care Collaborative includes 200 hospitals in the state committed to ending preventable maternal mortality and racial disparities.
Nine hospitals in the collaborative have started programs to reduce hemorrhages, manage high blood pressure disorders, and reduce the rate of cesarean deliveries. All are important reasons for maternal mortality.
These programs helped bring about a 62% reduction in California’s maternal mortality rate from 2006 to 2016. And 2023 figures show that California has the lowest maternal mortality rate of any state.
Alabama has the sixth highest rate of maternal mortality in the nation. The University of Alabama at Birmingham wants to address the racial disparities in maternal mortality with a cooperative called the P3 EQUATE Network.
The network is part of a $20 million program by the American Heart Association to gain greater understanding of the disproportionate effect of maternal mortality on Black and Native American people.
The program works with pregnant and postpartum women “to discover ways to reduce racism and social problems that contribute to poor health outcomes.”
In addition to collaborative efforts, the survey found maternal mortality programs at all the top medical centers.
NewYork-Presbyterian Hospital has a Mothers Center that provides specialized care to pregnant women with complications.
The University of Chicago Medical Center established a program called “Systematic Treatment and Management of Postpartum Hypertension” that includes patient and staff education, standardized hospital discharge instructions, and a follow-up in a postpartum hypertension clinic.
A 2021 study found that the program had helped increase the number of postpartum women who correctly follow blood pressure control guidance.
A program called MOMS Navigation at Northwell Health in Long Island, N.Y., provides support to high-risk mothers. The program decreased 30-day readmission rates for all patients by 50% and for Black birthing patients by 60%. Reducing readmission is an important measure for reducing complications.
Vanderbilt University Medical Center in Nashville has what it calls the first-of-its-kind educational podcasts Healthy Mom Healthy Baby, where 30% of the content is devoted to health disparities.
And several centers, including Brigham and Women’s Hospital in Boston and NewYork-Presbyterian, make sure mothers have access to doulas – professional support people trained in the needs of the family during pregnancy and childbirth.
The survey found that 9 of the 10 centers have obstetric programs devoted to cardiac care, including the University of Chicago, Stanford Medicine, UCLA, and the Cleveland Clinic.
But the survey results raise the question: How can we have these programs and research at our best obstetrics centers devoted to reducing maternal mortality and have the highest rate of all developed countries?
“Maternal mortality largely falls on pregnant and birthing persons who do not intersect with, nor are touched, by the best obstetrical care centers in the country,” Dr. Simon said.
Unfortunately, she said, the pregnant people who face “high maternal mortality rates … face all the access-to-care barriers and do not have the privilege of birthing or accessing care at top centers.”
Anne Hutchinson believed going to a top center – the Cleveland Clinic – would give her a good chance of safely delivering a second child.
Karlee Hoffman, DO, a cardiologist in the hospital’s cardio-obstetric high-risk clinic, said Ms. Hutchinson “came to me, she was determined to have another child, and she said, ‘Please help me do this. I’m doing it regardless. So, I would really like your support in moving forward,’” Dr. Hoffman recalls.
Ms. Hutchinson said Cleveland Clinic doctors told her she had a 20%-30% chance of peripartum cardiomyopathy again if she had a second child. If that happened, the risks “ranged from mild decompensation of my heart function to death,“ she said.
Ms. Hutchinson and her husband decided to go ahead with the pregnancy. Her parents cried when they found out. But Ms. Hutchinson says she was confident in the cardio obstetric team at Cleveland Clinic.
Her fertility medicine raised the possibility of multiple births, which would be a definite threat to her life. Her heart failure medicine, Entresto, could not be used during pregnancy, so her doctors put her on older medicines.
She got pregnant in June 2022 and developed gestational diabetes, which can affect pregnancy because of raised blood sugar. Another potential risk. She was carefully monitored by the specialists and hospitalized once.
At 37 weeks, she was induced and had a forceps delivery. On Feb. 15, 2023, her second daughter, Charlotte, was born.
Ms. Hutchinson was asked to write about how she felt when she delivered Charlotte:
“I am not sure how to put into words the love, joy, and elation that I felt holding Charlotte for the first time. As I write this, I have tears of joy in my eyes thinking of that moment. I had prayed for her for so long and after being told I couldn’t or shouldn’t have any more children.”
“I felt that Charlotte and I were forever bonded in triumph from that moment on. We did it and made it out alive! And our family was now complete. I have so much joy watching the love that is growing between Charlotte and Lillian. Life is truly amazing, and I am forever grateful to have them.”
A version of this article first appeared on WebMD.com.