User login
Results vary for pregnancy-related antismoking efforts
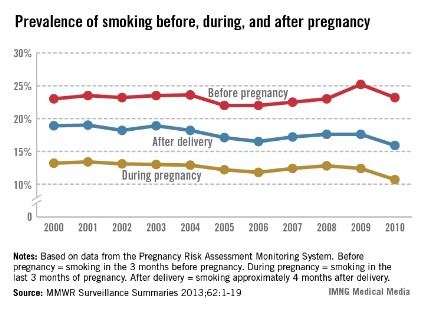
Smoking during pregnancy and after delivery decreased moderately but significantly from 2000 to 2010, while the prevalence of smoking before pregnancy dropped only slightly in that time, the Centers for Disease Control and Prevention reported.
The prevalence of smoking during pregnancy went from 13.2% in 2000 to 10.7% in 2010, and the prevalence of smoking after delivery dropped from 18.9% to 15.9%. In comparison, the prevalence of smoking during the 3 months before pregnancy made a nonsignificant drop from 23% in 2000 to 22.5% in 2007, and rose to 23.2% in 2010, according to data from the Pregnancy Risk Assessment Monitoring System (PRAMS).
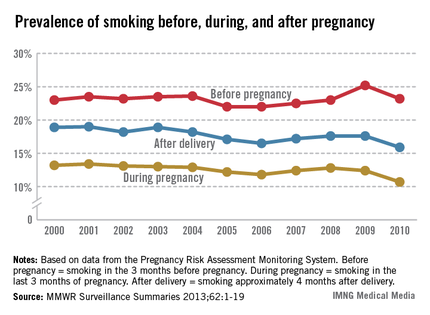
The CDC report noted the Healthy People 2020 goal of reducing the prevalence of prenatal smoking to 1.4% and suggested that, "at the current pace ... it could take another 100 years for the United States to reach this goal" (MMWR Surveillance Summaries 2013;62[SS06]:1-19).
For 2010, the data from 27 PRAMS sites nationwide – 26 states and New York City -– represent approximately 52% of live births.
Smoking during pregnancy and after delivery decreased moderately but significantly from 2000 to 2010, while the prevalence of smoking before pregnancy dropped only slightly in that time, the Centers for Disease Control and Prevention reported.
The prevalence of smoking during pregnancy went from 13.2% in 2000 to 10.7% in 2010, and the prevalence of smoking after delivery dropped from 18.9% to 15.9%. In comparison, the prevalence of smoking during the 3 months before pregnancy made a nonsignificant drop from 23% in 2000 to 22.5% in 2007, and rose to 23.2% in 2010, according to data from the Pregnancy Risk Assessment Monitoring System (PRAMS).

The CDC report noted the Healthy People 2020 goal of reducing the prevalence of prenatal smoking to 1.4% and suggested that, "at the current pace ... it could take another 100 years for the United States to reach this goal" (MMWR Surveillance Summaries 2013;62[SS06]:1-19).
For 2010, the data from 27 PRAMS sites nationwide – 26 states and New York City -– represent approximately 52% of live births.
Smoking during pregnancy and after delivery decreased moderately but significantly from 2000 to 2010, while the prevalence of smoking before pregnancy dropped only slightly in that time, the Centers for Disease Control and Prevention reported.
The prevalence of smoking during pregnancy went from 13.2% in 2000 to 10.7% in 2010, and the prevalence of smoking after delivery dropped from 18.9% to 15.9%. In comparison, the prevalence of smoking during the 3 months before pregnancy made a nonsignificant drop from 23% in 2000 to 22.5% in 2007, and rose to 23.2% in 2010, according to data from the Pregnancy Risk Assessment Monitoring System (PRAMS).

The CDC report noted the Healthy People 2020 goal of reducing the prevalence of prenatal smoking to 1.4% and suggested that, "at the current pace ... it could take another 100 years for the United States to reach this goal" (MMWR Surveillance Summaries 2013;62[SS06]:1-19).
For 2010, the data from 27 PRAMS sites nationwide – 26 states and New York City -– represent approximately 52% of live births.
FROM MMWR
Limited neonatal benefit from later delivery in congenital heart disease
SYDNEY – A shift in practice away from elective late preterm delivery to early term deliveries has shown little effect on neonatal outcomes such as intensive care length of stay, neonatal morbidity, or mortality in a population with major congenital heart disease, a study has found.
Researchers at Columbia University Medical Center and Morgan Stanley Children’s Hospital, both in New York, sought to evaluate the effect of a change in delivery practice that was introduced following the 2009 publication of a study showing improved outcomes in infants delivered after 39 weeks.
A review of the medical records of infants with critical congenital heart disease compared outcomes in 878 infants born at a single tertiary center during 2004-2008 to outcomes in 124 infants born in 2010, after the change in practice had been implemented.
There was a significant increase in the mean gestational age (37.8 weeks vs. 38.4 weeks, respectively; P less than .01) and in mean birth weight (2,975 grams vs. 3,134 grams; P less than .01).
However there were no significant differences between 2004-2008 and 2010 data in 5-minute APGAR scores (8.5 vs. 8.6; P = .11), median length of stay (14 days [1-197] vs. 16 days [1-144]; P = .18) and neonatal mortality (6% vs. 11%; P = .07), according to data presented at the International Society of Ultrasound in Obstetrics and Gynecology world congress.
"After the 2009 paper came out, there was a real shift in practice among obstetricians – and also pushed by neonatologists – to try to not deliver patients before 39 weeks," said researcher and fetal cardiologist Dr. Ismee Williams, of New York–Presbyterian Hospital/Columbia University Medical Center.
"Then our goal, instead of being 38 weeks, was 39 weeks, and we would not schedule anybody for induction or a cesarean section unless they were 39 weeks and change," Dr. Williams said in an interview.
As a result of the change in practice, there were significant decreases in scheduled cesarean delivery from 2004-2008 to 2010 and in scheduled induction, but significant increases in urgent inductions, urgent cesarean deliveries, and cesarean deliveries after labor.
"We were happy to see that the change did affect a consequent increase in gestational age and birth weight, but we were somewhat surprised that it didn’t affect length of stay," Dr. Williams said.
Given that the study was conducted in a population with congenital heart disease, she said a likely explanation was that most of the infants would require surgery before they were discharged from hospital.
Dr. Williams said that the study didn’t look at maternal length of stay in hospital or other indicators of maternal morbidity, but she noted perhaps the next step would be to examine more closely the effect of the change in practice on mothers.
"However, it’s possible that, in the congenital heart disease population, whether or not you deliver at 38 or 39 weeks, we’re not seeing that big of a difference in terms of the neonatal outcomes or a big impact on big markers of morbidity like length of stay," Dr. Williams said.
There were no conflicts of interest declared.
SYDNEY – A shift in practice away from elective late preterm delivery to early term deliveries has shown little effect on neonatal outcomes such as intensive care length of stay, neonatal morbidity, or mortality in a population with major congenital heart disease, a study has found.
Researchers at Columbia University Medical Center and Morgan Stanley Children’s Hospital, both in New York, sought to evaluate the effect of a change in delivery practice that was introduced following the 2009 publication of a study showing improved outcomes in infants delivered after 39 weeks.
A review of the medical records of infants with critical congenital heart disease compared outcomes in 878 infants born at a single tertiary center during 2004-2008 to outcomes in 124 infants born in 2010, after the change in practice had been implemented.
There was a significant increase in the mean gestational age (37.8 weeks vs. 38.4 weeks, respectively; P less than .01) and in mean birth weight (2,975 grams vs. 3,134 grams; P less than .01).
However there were no significant differences between 2004-2008 and 2010 data in 5-minute APGAR scores (8.5 vs. 8.6; P = .11), median length of stay (14 days [1-197] vs. 16 days [1-144]; P = .18) and neonatal mortality (6% vs. 11%; P = .07), according to data presented at the International Society of Ultrasound in Obstetrics and Gynecology world congress.
"After the 2009 paper came out, there was a real shift in practice among obstetricians – and also pushed by neonatologists – to try to not deliver patients before 39 weeks," said researcher and fetal cardiologist Dr. Ismee Williams, of New York–Presbyterian Hospital/Columbia University Medical Center.
"Then our goal, instead of being 38 weeks, was 39 weeks, and we would not schedule anybody for induction or a cesarean section unless they were 39 weeks and change," Dr. Williams said in an interview.
As a result of the change in practice, there were significant decreases in scheduled cesarean delivery from 2004-2008 to 2010 and in scheduled induction, but significant increases in urgent inductions, urgent cesarean deliveries, and cesarean deliveries after labor.
"We were happy to see that the change did affect a consequent increase in gestational age and birth weight, but we were somewhat surprised that it didn’t affect length of stay," Dr. Williams said.
Given that the study was conducted in a population with congenital heart disease, she said a likely explanation was that most of the infants would require surgery before they were discharged from hospital.
Dr. Williams said that the study didn’t look at maternal length of stay in hospital or other indicators of maternal morbidity, but she noted perhaps the next step would be to examine more closely the effect of the change in practice on mothers.
"However, it’s possible that, in the congenital heart disease population, whether or not you deliver at 38 or 39 weeks, we’re not seeing that big of a difference in terms of the neonatal outcomes or a big impact on big markers of morbidity like length of stay," Dr. Williams said.
There were no conflicts of interest declared.
SYDNEY – A shift in practice away from elective late preterm delivery to early term deliveries has shown little effect on neonatal outcomes such as intensive care length of stay, neonatal morbidity, or mortality in a population with major congenital heart disease, a study has found.
Researchers at Columbia University Medical Center and Morgan Stanley Children’s Hospital, both in New York, sought to evaluate the effect of a change in delivery practice that was introduced following the 2009 publication of a study showing improved outcomes in infants delivered after 39 weeks.
A review of the medical records of infants with critical congenital heart disease compared outcomes in 878 infants born at a single tertiary center during 2004-2008 to outcomes in 124 infants born in 2010, after the change in practice had been implemented.
There was a significant increase in the mean gestational age (37.8 weeks vs. 38.4 weeks, respectively; P less than .01) and in mean birth weight (2,975 grams vs. 3,134 grams; P less than .01).
However there were no significant differences between 2004-2008 and 2010 data in 5-minute APGAR scores (8.5 vs. 8.6; P = .11), median length of stay (14 days [1-197] vs. 16 days [1-144]; P = .18) and neonatal mortality (6% vs. 11%; P = .07), according to data presented at the International Society of Ultrasound in Obstetrics and Gynecology world congress.
"After the 2009 paper came out, there was a real shift in practice among obstetricians – and also pushed by neonatologists – to try to not deliver patients before 39 weeks," said researcher and fetal cardiologist Dr. Ismee Williams, of New York–Presbyterian Hospital/Columbia University Medical Center.
"Then our goal, instead of being 38 weeks, was 39 weeks, and we would not schedule anybody for induction or a cesarean section unless they were 39 weeks and change," Dr. Williams said in an interview.
As a result of the change in practice, there were significant decreases in scheduled cesarean delivery from 2004-2008 to 2010 and in scheduled induction, but significant increases in urgent inductions, urgent cesarean deliveries, and cesarean deliveries after labor.
"We were happy to see that the change did affect a consequent increase in gestational age and birth weight, but we were somewhat surprised that it didn’t affect length of stay," Dr. Williams said.
Given that the study was conducted in a population with congenital heart disease, she said a likely explanation was that most of the infants would require surgery before they were discharged from hospital.
Dr. Williams said that the study didn’t look at maternal length of stay in hospital or other indicators of maternal morbidity, but she noted perhaps the next step would be to examine more closely the effect of the change in practice on mothers.
"However, it’s possible that, in the congenital heart disease population, whether or not you deliver at 38 or 39 weeks, we’re not seeing that big of a difference in terms of the neonatal outcomes or a big impact on big markers of morbidity like length of stay," Dr. Williams said.
There were no conflicts of interest declared.
AT THE ISUOG WORLD CONGRESS
Major finding: There were no significant differences between 2004-2008 and 2010 data in 5-minute APGAR scores (8.5 vs. 8.6; P = .11), median length of stay (14 days [1-197] vs. 16 days [1-144]; P = .18), and neonatal mortality (6% vs. 11%; P = .07)
Data source: Single-center retrospective study of medical records of 1,002 infants with congenital heart disease.
Disclosures: There were no conflicts of interest declared.
Genetic anomalies account for majority of miscarriages
BOSTON – A substantial majority of miscarriages appear to be caused by genetic abnormalities rather than the usual suspects of thrombophilias, antiphospholipid syndromes, immunologic problems, or other maternal factors, study results suggest.
A decade of microscopic studies of placental tissues obtained after pregnancy losses showed that of 615 samples, 495 cases (80.5%) were caused by genetic abnormalities, determined by the presence of markers for genetic abnormalities, said Dr. Harvey J. Kliman, director of the reproductive and placental research unit at Yale University in New Haven, Conn.
Data from a second study of the karyotypes of embryos from spontaneous abortions showed that embryonic chromosomal abnormalities accounted for a significant portion of the miscarriages, and that two-thirds of women with prior loss of an aneuploid embryo went on to have a live birth, said Dr. Ruth B. Lathi, director of the recurrent pregnancy loss program at the Stanford (Calif.) Fertility and Reproductive Medicine Center.
The studies were reported at the conjoint meeting of the International Federation of Fertility Societies and the American Society for Reproductive Medicine.
Although standard workups fail to identify the cause of recurrent pregnancy losses in more than half of all cases, the findings should be reassuring to patients, Dr. Lathi said in an interview.
"The vast majority of miscarriages are chromosomally or somehow morphologically or genetically abnormal – even if we can’t prove it, we all believe it. Luckily, these genetic abnormalities for the most part are random, and women can have a combination of abnormal pregnancies and normal pregnancies," Dr. Lathi said.
"Each new pregnancy is a new set of genetics: It’s a new egg, it’s a new sperm, and what was wrong in the last pregnancy may not carry over to the next pregnancy," she added.
Dr. Kliman said that if women know that recurrent miscarriages are unlikely to be related to something they have done, such as having a cocktail or smoking marijuana when they weren’t even aware that they were pregnant, they will be better able to cope emotionally and resume trying.
"I have a lot of families that have six pregnancies: three perfectly normal children and three losses, and the losses always happen at the same time, because it’s a programming error; that’s another hint that it’s genetic," he said.
Dr. Kliman and Kristin M. Milano, director of laboratory services in the reproductive and placental research unit at Yale, reported data on 615 pregnancy losses that occurred from 7 to 20 weeks’ gestational age. The samples were fixed, sectioned, stained, and then examined microscopically for abnormalities.
The primary diagnosis in 495 of the 615 samples examined was dysmorphic trophoblastic invaginations and inclusions. In 55 cases (8.9%) there were no chorionic villi, 53 cases (8.6%) had no pathologic abnormalities, 10 cases (1.6%) were apparently due to thrombosis, 1 (0.2%) was due to abruption, and 1 to chronic villitis.
"This study validates the importance of pathologic examination of all pregnancy losses," Dr. Kliman said in a poster.
Dr. Lathi and her colleague, Dr. Jamie Massie, examined the live birth rate by embryonic karyotype among 95 women with a self-reported history of recurrent pregnancy loss.
They found that 40 of the women (42%) had a miscarriage of a euploid embryo, and 55 (58%) lost a pregnancy with an aneuploid embryo. Of the aneuploid abnormalities, 95% were numeric (for example, monosomies and trisomies), and 5% were structural.
In all, 21 of the 40 women (52.5%) with loss of a euploid embryo had a subsequent live birth, compared with 37 of 55 (67.3%) of those with loss of an aneuploid pregnancy.
The findings, which need to be validated in larger studies, suggest that aneuploidy may be a good prognostic indicator for the success of future live births, the investigators concluded.
Dr. Lathi was coauthor of a separate study of 87 women with two or more recurrent pregnancy losses from either explained or unexplained causes, which found that 59.7% of the patients had a healthy term delivery, with obstetrical outcomes similar to those in women with infertility problems.
Dr. Kliman’s study was supported by the reproductive and placental research unit at Yale. He reported having no relevant financial disclosures. The funding sources for Dr. Lathi’s studies were not disclosed. She reported having no relevant conflicts of interest.
BOSTON – A substantial majority of miscarriages appear to be caused by genetic abnormalities rather than the usual suspects of thrombophilias, antiphospholipid syndromes, immunologic problems, or other maternal factors, study results suggest.
A decade of microscopic studies of placental tissues obtained after pregnancy losses showed that of 615 samples, 495 cases (80.5%) were caused by genetic abnormalities, determined by the presence of markers for genetic abnormalities, said Dr. Harvey J. Kliman, director of the reproductive and placental research unit at Yale University in New Haven, Conn.
Data from a second study of the karyotypes of embryos from spontaneous abortions showed that embryonic chromosomal abnormalities accounted for a significant portion of the miscarriages, and that two-thirds of women with prior loss of an aneuploid embryo went on to have a live birth, said Dr. Ruth B. Lathi, director of the recurrent pregnancy loss program at the Stanford (Calif.) Fertility and Reproductive Medicine Center.
The studies were reported at the conjoint meeting of the International Federation of Fertility Societies and the American Society for Reproductive Medicine.
Although standard workups fail to identify the cause of recurrent pregnancy losses in more than half of all cases, the findings should be reassuring to patients, Dr. Lathi said in an interview.
"The vast majority of miscarriages are chromosomally or somehow morphologically or genetically abnormal – even if we can’t prove it, we all believe it. Luckily, these genetic abnormalities for the most part are random, and women can have a combination of abnormal pregnancies and normal pregnancies," Dr. Lathi said.
"Each new pregnancy is a new set of genetics: It’s a new egg, it’s a new sperm, and what was wrong in the last pregnancy may not carry over to the next pregnancy," she added.
Dr. Kliman said that if women know that recurrent miscarriages are unlikely to be related to something they have done, such as having a cocktail or smoking marijuana when they weren’t even aware that they were pregnant, they will be better able to cope emotionally and resume trying.
"I have a lot of families that have six pregnancies: three perfectly normal children and three losses, and the losses always happen at the same time, because it’s a programming error; that’s another hint that it’s genetic," he said.
Dr. Kliman and Kristin M. Milano, director of laboratory services in the reproductive and placental research unit at Yale, reported data on 615 pregnancy losses that occurred from 7 to 20 weeks’ gestational age. The samples were fixed, sectioned, stained, and then examined microscopically for abnormalities.
The primary diagnosis in 495 of the 615 samples examined was dysmorphic trophoblastic invaginations and inclusions. In 55 cases (8.9%) there were no chorionic villi, 53 cases (8.6%) had no pathologic abnormalities, 10 cases (1.6%) were apparently due to thrombosis, 1 (0.2%) was due to abruption, and 1 to chronic villitis.
"This study validates the importance of pathologic examination of all pregnancy losses," Dr. Kliman said in a poster.
Dr. Lathi and her colleague, Dr. Jamie Massie, examined the live birth rate by embryonic karyotype among 95 women with a self-reported history of recurrent pregnancy loss.
They found that 40 of the women (42%) had a miscarriage of a euploid embryo, and 55 (58%) lost a pregnancy with an aneuploid embryo. Of the aneuploid abnormalities, 95% were numeric (for example, monosomies and trisomies), and 5% were structural.
In all, 21 of the 40 women (52.5%) with loss of a euploid embryo had a subsequent live birth, compared with 37 of 55 (67.3%) of those with loss of an aneuploid pregnancy.
The findings, which need to be validated in larger studies, suggest that aneuploidy may be a good prognostic indicator for the success of future live births, the investigators concluded.
Dr. Lathi was coauthor of a separate study of 87 women with two or more recurrent pregnancy losses from either explained or unexplained causes, which found that 59.7% of the patients had a healthy term delivery, with obstetrical outcomes similar to those in women with infertility problems.
Dr. Kliman’s study was supported by the reproductive and placental research unit at Yale. He reported having no relevant financial disclosures. The funding sources for Dr. Lathi’s studies were not disclosed. She reported having no relevant conflicts of interest.
BOSTON – A substantial majority of miscarriages appear to be caused by genetic abnormalities rather than the usual suspects of thrombophilias, antiphospholipid syndromes, immunologic problems, or other maternal factors, study results suggest.
A decade of microscopic studies of placental tissues obtained after pregnancy losses showed that of 615 samples, 495 cases (80.5%) were caused by genetic abnormalities, determined by the presence of markers for genetic abnormalities, said Dr. Harvey J. Kliman, director of the reproductive and placental research unit at Yale University in New Haven, Conn.
Data from a second study of the karyotypes of embryos from spontaneous abortions showed that embryonic chromosomal abnormalities accounted for a significant portion of the miscarriages, and that two-thirds of women with prior loss of an aneuploid embryo went on to have a live birth, said Dr. Ruth B. Lathi, director of the recurrent pregnancy loss program at the Stanford (Calif.) Fertility and Reproductive Medicine Center.
The studies were reported at the conjoint meeting of the International Federation of Fertility Societies and the American Society for Reproductive Medicine.
Although standard workups fail to identify the cause of recurrent pregnancy losses in more than half of all cases, the findings should be reassuring to patients, Dr. Lathi said in an interview.
"The vast majority of miscarriages are chromosomally or somehow morphologically or genetically abnormal – even if we can’t prove it, we all believe it. Luckily, these genetic abnormalities for the most part are random, and women can have a combination of abnormal pregnancies and normal pregnancies," Dr. Lathi said.
"Each new pregnancy is a new set of genetics: It’s a new egg, it’s a new sperm, and what was wrong in the last pregnancy may not carry over to the next pregnancy," she added.
Dr. Kliman said that if women know that recurrent miscarriages are unlikely to be related to something they have done, such as having a cocktail or smoking marijuana when they weren’t even aware that they were pregnant, they will be better able to cope emotionally and resume trying.
"I have a lot of families that have six pregnancies: three perfectly normal children and three losses, and the losses always happen at the same time, because it’s a programming error; that’s another hint that it’s genetic," he said.
Dr. Kliman and Kristin M. Milano, director of laboratory services in the reproductive and placental research unit at Yale, reported data on 615 pregnancy losses that occurred from 7 to 20 weeks’ gestational age. The samples were fixed, sectioned, stained, and then examined microscopically for abnormalities.
The primary diagnosis in 495 of the 615 samples examined was dysmorphic trophoblastic invaginations and inclusions. In 55 cases (8.9%) there were no chorionic villi, 53 cases (8.6%) had no pathologic abnormalities, 10 cases (1.6%) were apparently due to thrombosis, 1 (0.2%) was due to abruption, and 1 to chronic villitis.
"This study validates the importance of pathologic examination of all pregnancy losses," Dr. Kliman said in a poster.
Dr. Lathi and her colleague, Dr. Jamie Massie, examined the live birth rate by embryonic karyotype among 95 women with a self-reported history of recurrent pregnancy loss.
They found that 40 of the women (42%) had a miscarriage of a euploid embryo, and 55 (58%) lost a pregnancy with an aneuploid embryo. Of the aneuploid abnormalities, 95% were numeric (for example, monosomies and trisomies), and 5% were structural.
In all, 21 of the 40 women (52.5%) with loss of a euploid embryo had a subsequent live birth, compared with 37 of 55 (67.3%) of those with loss of an aneuploid pregnancy.
The findings, which need to be validated in larger studies, suggest that aneuploidy may be a good prognostic indicator for the success of future live births, the investigators concluded.
Dr. Lathi was coauthor of a separate study of 87 women with two or more recurrent pregnancy losses from either explained or unexplained causes, which found that 59.7% of the patients had a healthy term delivery, with obstetrical outcomes similar to those in women with infertility problems.
Dr. Kliman’s study was supported by the reproductive and placental research unit at Yale. He reported having no relevant financial disclosures. The funding sources for Dr. Lathi’s studies were not disclosed. She reported having no relevant conflicts of interest.
AT THE JOINT IFFS/ASRM MEETING
Major finding: Of 615 spontaneous abortions, 495 (80.5%) were caused by genetic abnormalities in the embryo rather than maternal factors.
Data source: Prospective single-center studies of the contribution of genetic abnormalities to pregnancy loss.
Disclosures: Dr. Kliman’s study was supported by the reproductive and placental research unit at Yale. He reported having no relevant financial disclosures. The funding sources for Dr. Lathi’s studies were not disclosed. She reported having no relevant conflicts of interest.
Novel predictor of poor outcomes in lupus pregnancies
SAN DIEGO – Alteration in the balance of placentally secreted angiogenic factors early in pregnancy provides a potent new predictor of subsequent preeclampsia and other poor outcomes in pregnant women with systemic lupus erythematosus and/or antiphospholipid antibody syndrome.
Patients with systemic lupus erythematosus (SLE) and/or antiphospholipid antibody syndrome (APS) who had an elevated ratio of a splice variant of vascular endothelial growth factor R1 called sFLT1 to placental growth factor (PlGF) when measured at 16-19 weeks’ gestation were at 13.8-fold increased relative risk of preeclampsia before 34 weeks, compared with patients with an sFLT1/PlGF ratio below that cut-point in the large, multicenter, observational PROMISSE (Predictors of Pregnancy Outcome: Biomarkers in Antiphospholipid Antibody Syndrome and SLE) study, Dr. Jane E. Salmon reported at the annual meeting of the American College of Rheumatology.
"Nearly half of the patients with an SFLT1/PlGF ratio greater than 3.45 when measured at 16-19 weeks’ gestation will develop early preeclampsia. On the other hand, a low ratio, as well as low levels of sFLT1 or high levels of PlGF, can reassure physicians and patients that preterm preeclampsia is unlikely: a 3% chance," said Dr. Salmon, professor of medicine and of ob.gyn. at Cornell University and a rheumatologist at the Hospital for Special Surgery, both in New York.
Pregnancy in patients with lupus is associated with obstetric complications placing both mother and fetus at great risk. Yet, until now it hasn’t been possible to predict which patients will have poor outcomes.
The key to identifying those at high risk lies in a recognition that preeclampsia and other poor outcomes are dramatic manifestations of placental insufficiency, which actually begins, initially silently, early in pregnancy. The maternal hypertension, proteinuria, thrombocytopenia, and other end-organ manifestations of preeclampsia are caused by maternal endothelial dysfunction mediated by placental secretion of antiangiogenic factors. Angiogenic growth factors, such as PIGF and vascular endothelial growth factor (VEGF), are essential to a healthy endothelium. But placentally secreted sFLT1 binds to these two angiogenic growth factors, rendering them unavailable to the endothelium, she explained.
Overexpression of sFLT1 in multiple animal models results in hypertension and proteinuria, the hallmarks of preeclampsia. Moreover, cancer patients treated with VEGF inhibitors often develop these two conditions. Based in part on these observations, Dr. Salmon and her coinvestigators turned to the PROMISSE study to test their hypothesis that elevated levels of antiangiogenic factors early in pregnancy predict poor outcomes in patients with SLE and/or APS. The prospective study involved 503 pregnant women with SLE and/or APS and 204 healthy controls, all with monthly blood draws starting before 12 weeks’ gestation.
The composite outcome of preeclampsia, small for gestational age, indicated preterm delivery, and other adverse events occurred in 37% of SLE patients who also had APS. The rate was 16% in patients with SLE alone, 26% in those with APS alone, and 3% in controls.
Subjects with SLE and/or APS who developed preeclampsia and other pregnancy complications displayed significantly higher levels of sFLT1 beginning at 12 weeks and sustained through 31 weeks’ gestation, compared with those with normal pregnancies. Moreover, PlGF levels were significantly lower during weeks 16-31 in the patients with pregnancy complications. The investigators determined that the best predictor of pregnancy complications was the ratio of antiangiogenic sFLT1 to angiogenic PlGF. And the optimal cut-point was 3.45.
Audience members said that while a predictive test for preeclampsia is most welcome, the fact remains that physicians don’t have a lot to offer in terms of prevention or treatment of this feared pregnancy complication. Dr. Salmon responded that the SFLT1/PlGF ratio can be used to risk-stratify pregnant lupus patients for future interventional trials with new drugs looking at new pathways. Already, for example, other investigators have reported some success using a strategy targeting sFLT1 itself. In a small study, they found that women with severe preeclampsia who had their blood run through a heparin column that binds and removes sFLT1 were able to maintain their pregnancies for up to 2 weeks.
"It’s a tiny, open-label trial involving a device, but I think that will move forward," she predicted.
The PROMISSE study was funded by the National Institutes of Health, the Alliance for Lupus Research, and the Mary Kirkland Center for Lupus Research at the Hospital for Special Surgery. Dr. Salmon reported having received research grants from and/or serving as a consultant to Alexion, Novartis, and Roche.
SLE, APS, vascular endothelial growth factor R1, sFLT1, placental growth factor, PlGF, preeclampsia, PROMISSE study, Predictors of Pregnancy Outcome: Biomarkers in Antiphospholipid Antibody Syndrome and SLE study, Dr. Jane E. Salmon, annual meeting of the American College of Rheumatology, early preeclampsia,
SAN DIEGO – Alteration in the balance of placentally secreted angiogenic factors early in pregnancy provides a potent new predictor of subsequent preeclampsia and other poor outcomes in pregnant women with systemic lupus erythematosus and/or antiphospholipid antibody syndrome.
Patients with systemic lupus erythematosus (SLE) and/or antiphospholipid antibody syndrome (APS) who had an elevated ratio of a splice variant of vascular endothelial growth factor R1 called sFLT1 to placental growth factor (PlGF) when measured at 16-19 weeks’ gestation were at 13.8-fold increased relative risk of preeclampsia before 34 weeks, compared with patients with an sFLT1/PlGF ratio below that cut-point in the large, multicenter, observational PROMISSE (Predictors of Pregnancy Outcome: Biomarkers in Antiphospholipid Antibody Syndrome and SLE) study, Dr. Jane E. Salmon reported at the annual meeting of the American College of Rheumatology.
"Nearly half of the patients with an SFLT1/PlGF ratio greater than 3.45 when measured at 16-19 weeks’ gestation will develop early preeclampsia. On the other hand, a low ratio, as well as low levels of sFLT1 or high levels of PlGF, can reassure physicians and patients that preterm preeclampsia is unlikely: a 3% chance," said Dr. Salmon, professor of medicine and of ob.gyn. at Cornell University and a rheumatologist at the Hospital for Special Surgery, both in New York.
Pregnancy in patients with lupus is associated with obstetric complications placing both mother and fetus at great risk. Yet, until now it hasn’t been possible to predict which patients will have poor outcomes.
The key to identifying those at high risk lies in a recognition that preeclampsia and other poor outcomes are dramatic manifestations of placental insufficiency, which actually begins, initially silently, early in pregnancy. The maternal hypertension, proteinuria, thrombocytopenia, and other end-organ manifestations of preeclampsia are caused by maternal endothelial dysfunction mediated by placental secretion of antiangiogenic factors. Angiogenic growth factors, such as PIGF and vascular endothelial growth factor (VEGF), are essential to a healthy endothelium. But placentally secreted sFLT1 binds to these two angiogenic growth factors, rendering them unavailable to the endothelium, she explained.
Overexpression of sFLT1 in multiple animal models results in hypertension and proteinuria, the hallmarks of preeclampsia. Moreover, cancer patients treated with VEGF inhibitors often develop these two conditions. Based in part on these observations, Dr. Salmon and her coinvestigators turned to the PROMISSE study to test their hypothesis that elevated levels of antiangiogenic factors early in pregnancy predict poor outcomes in patients with SLE and/or APS. The prospective study involved 503 pregnant women with SLE and/or APS and 204 healthy controls, all with monthly blood draws starting before 12 weeks’ gestation.
The composite outcome of preeclampsia, small for gestational age, indicated preterm delivery, and other adverse events occurred in 37% of SLE patients who also had APS. The rate was 16% in patients with SLE alone, 26% in those with APS alone, and 3% in controls.
Subjects with SLE and/or APS who developed preeclampsia and other pregnancy complications displayed significantly higher levels of sFLT1 beginning at 12 weeks and sustained through 31 weeks’ gestation, compared with those with normal pregnancies. Moreover, PlGF levels were significantly lower during weeks 16-31 in the patients with pregnancy complications. The investigators determined that the best predictor of pregnancy complications was the ratio of antiangiogenic sFLT1 to angiogenic PlGF. And the optimal cut-point was 3.45.
Audience members said that while a predictive test for preeclampsia is most welcome, the fact remains that physicians don’t have a lot to offer in terms of prevention or treatment of this feared pregnancy complication. Dr. Salmon responded that the SFLT1/PlGF ratio can be used to risk-stratify pregnant lupus patients for future interventional trials with new drugs looking at new pathways. Already, for example, other investigators have reported some success using a strategy targeting sFLT1 itself. In a small study, they found that women with severe preeclampsia who had their blood run through a heparin column that binds and removes sFLT1 were able to maintain their pregnancies for up to 2 weeks.
"It’s a tiny, open-label trial involving a device, but I think that will move forward," she predicted.
The PROMISSE study was funded by the National Institutes of Health, the Alliance for Lupus Research, and the Mary Kirkland Center for Lupus Research at the Hospital for Special Surgery. Dr. Salmon reported having received research grants from and/or serving as a consultant to Alexion, Novartis, and Roche.
SAN DIEGO – Alteration in the balance of placentally secreted angiogenic factors early in pregnancy provides a potent new predictor of subsequent preeclampsia and other poor outcomes in pregnant women with systemic lupus erythematosus and/or antiphospholipid antibody syndrome.
Patients with systemic lupus erythematosus (SLE) and/or antiphospholipid antibody syndrome (APS) who had an elevated ratio of a splice variant of vascular endothelial growth factor R1 called sFLT1 to placental growth factor (PlGF) when measured at 16-19 weeks’ gestation were at 13.8-fold increased relative risk of preeclampsia before 34 weeks, compared with patients with an sFLT1/PlGF ratio below that cut-point in the large, multicenter, observational PROMISSE (Predictors of Pregnancy Outcome: Biomarkers in Antiphospholipid Antibody Syndrome and SLE) study, Dr. Jane E. Salmon reported at the annual meeting of the American College of Rheumatology.
"Nearly half of the patients with an SFLT1/PlGF ratio greater than 3.45 when measured at 16-19 weeks’ gestation will develop early preeclampsia. On the other hand, a low ratio, as well as low levels of sFLT1 or high levels of PlGF, can reassure physicians and patients that preterm preeclampsia is unlikely: a 3% chance," said Dr. Salmon, professor of medicine and of ob.gyn. at Cornell University and a rheumatologist at the Hospital for Special Surgery, both in New York.
Pregnancy in patients with lupus is associated with obstetric complications placing both mother and fetus at great risk. Yet, until now it hasn’t been possible to predict which patients will have poor outcomes.
The key to identifying those at high risk lies in a recognition that preeclampsia and other poor outcomes are dramatic manifestations of placental insufficiency, which actually begins, initially silently, early in pregnancy. The maternal hypertension, proteinuria, thrombocytopenia, and other end-organ manifestations of preeclampsia are caused by maternal endothelial dysfunction mediated by placental secretion of antiangiogenic factors. Angiogenic growth factors, such as PIGF and vascular endothelial growth factor (VEGF), are essential to a healthy endothelium. But placentally secreted sFLT1 binds to these two angiogenic growth factors, rendering them unavailable to the endothelium, she explained.
Overexpression of sFLT1 in multiple animal models results in hypertension and proteinuria, the hallmarks of preeclampsia. Moreover, cancer patients treated with VEGF inhibitors often develop these two conditions. Based in part on these observations, Dr. Salmon and her coinvestigators turned to the PROMISSE study to test their hypothesis that elevated levels of antiangiogenic factors early in pregnancy predict poor outcomes in patients with SLE and/or APS. The prospective study involved 503 pregnant women with SLE and/or APS and 204 healthy controls, all with monthly blood draws starting before 12 weeks’ gestation.
The composite outcome of preeclampsia, small for gestational age, indicated preterm delivery, and other adverse events occurred in 37% of SLE patients who also had APS. The rate was 16% in patients with SLE alone, 26% in those with APS alone, and 3% in controls.
Subjects with SLE and/or APS who developed preeclampsia and other pregnancy complications displayed significantly higher levels of sFLT1 beginning at 12 weeks and sustained through 31 weeks’ gestation, compared with those with normal pregnancies. Moreover, PlGF levels were significantly lower during weeks 16-31 in the patients with pregnancy complications. The investigators determined that the best predictor of pregnancy complications was the ratio of antiangiogenic sFLT1 to angiogenic PlGF. And the optimal cut-point was 3.45.
Audience members said that while a predictive test for preeclampsia is most welcome, the fact remains that physicians don’t have a lot to offer in terms of prevention or treatment of this feared pregnancy complication. Dr. Salmon responded that the SFLT1/PlGF ratio can be used to risk-stratify pregnant lupus patients for future interventional trials with new drugs looking at new pathways. Already, for example, other investigators have reported some success using a strategy targeting sFLT1 itself. In a small study, they found that women with severe preeclampsia who had their blood run through a heparin column that binds and removes sFLT1 were able to maintain their pregnancies for up to 2 weeks.
"It’s a tiny, open-label trial involving a device, but I think that will move forward," she predicted.
The PROMISSE study was funded by the National Institutes of Health, the Alliance for Lupus Research, and the Mary Kirkland Center for Lupus Research at the Hospital for Special Surgery. Dr. Salmon reported having received research grants from and/or serving as a consultant to Alexion, Novartis, and Roche.
SLE, APS, vascular endothelial growth factor R1, sFLT1, placental growth factor, PlGF, preeclampsia, PROMISSE study, Predictors of Pregnancy Outcome: Biomarkers in Antiphospholipid Antibody Syndrome and SLE study, Dr. Jane E. Salmon, annual meeting of the American College of Rheumatology, early preeclampsia,
SLE, APS, vascular endothelial growth factor R1, sFLT1, placental growth factor, PlGF, preeclampsia, PROMISSE study, Predictors of Pregnancy Outcome: Biomarkers in Antiphospholipid Antibody Syndrome and SLE study, Dr. Jane E. Salmon, annual meeting of the American College of Rheumatology, early preeclampsia,
AT THE ACR ANNUAL MEETING
Major finding: Pregnant patients with SLE and/or APS had nearly a 14-fold increased risk of developing preeclampsia before 34 weeks’ gestation when they displayed an abnormal ratio of two key placentally derived angiogenic factors at 16-19 weeks’ gestation.
Data source: This analysis included 503 pregnant women with SLE and/or APS and 204 pregnant controls participating in the prospective, observational, multicenter PROMISSE study.
Disclosures: The study was funded by the National Institutes of Health, the Alliance for Lupus Research, and the Mary Kirkland Center for Lupus Research at the Hospital for Special Surgery. The presenter reported no relevant financial interests.
Women’s Sexuality Diminished by Psoriasis
LAS VEGAS – Psoriasis has a negative impact on women’s sexual desire, sexual ability, and sexual relationships, according to Dr. Jennifer Cather of the Modern Dermatology-Aesthetics Center in Dallas.
Dr. Cather and her colleagues are developing a clinical tool to assess the disease’s sexual impact; as part of those efforts, they conducted 60 interviews with moderately to severely psoriatic women with a mean age of 41 years.
The survey results showed that sexuality was clearly another reason why it’s important to keep psoriasis in check. Pain and itchiness during sex, self-consciousness and embarrassment, and avoidance of dating and intimate relationships were among the most common problems the women reported.
"We really didn’t appreciate the impact psoriasis had on relationships" before the survey, Dr. Cather said at the Skin Disease Education Foundation’s annual Las Vegas dermatology seminar. "The sexual impact has been underappreciated," she said.
Those problems might help explain why women with psoriasis, especially those aged younger than 35 years, tend to have fewer babies, she added. Psoriatic women also have higher rates of induced and spontaneous abortions and are more likely to have preterm and underweight births.
Some of the women surveyed said that they worried about passing psoriasis to their children. "Maybe there’s voluntary childlessness," Dr. Cather said. But when women in her practice mention they don’t want kids for fear of passing on the disease, "I answer right back, ‘You don’t have to have what you have. I can help you with it,’ " she said.
Methotrexate, acitretin, and psoralen photochemotherapy (PUVA) are contraindicated during pregnancy, but ultraviolet B (UVB) treatments are safe, said Dr. Cather. She also uses tumor necrosis factor inhibitors, which are FDA pregnancy category B agents; she prefers etanercept for its short half-life (just over 4 days) and because ob.gyns. are usually familiar with it. The Organization of Teratology Information Specialists (OTIS) keeps the etanercept and adalimumab pregnancy registries, and "there’s [been] no signal to date" for those drugs, she said.
Psoriatic women should know that the odds are with them for having a normal pregnancy, and that pregnancy is likely to help clear their skin, Dr. Cather said.
But because half of the pregnancies in the United States are unplanned, it’s important to discuss pregnancy – and psoriasis treatment during pregnancy – as part of routine care. "It’s difficult if a psoriasis patient calls you in a panic because they’re not sure their drug is safe, and not sure their doctor will let them continue it,’ she said. Some ob.gyns. are comfortable with letting women stay on their psoriasis therapy, while others want them to quit everything, even topical steroids.
"In my clinic, when a psoriasis patient gets pregnant, usually we’ve planned for it and are excited about it, and we’ve had some dialogue with the ob.gyn.," she explained.
SDEF and this news organization are owned by Frontline Medical Communications. Dr. Cather is a consultant, speaker, or researcher for AbbVie, Novartis, Leo, Janssen, Amgen, Celgene, Merck, and Pfizer.
LAS VEGAS – Psoriasis has a negative impact on women’s sexual desire, sexual ability, and sexual relationships, according to Dr. Jennifer Cather of the Modern Dermatology-Aesthetics Center in Dallas.
Dr. Cather and her colleagues are developing a clinical tool to assess the disease’s sexual impact; as part of those efforts, they conducted 60 interviews with moderately to severely psoriatic women with a mean age of 41 years.
The survey results showed that sexuality was clearly another reason why it’s important to keep psoriasis in check. Pain and itchiness during sex, self-consciousness and embarrassment, and avoidance of dating and intimate relationships were among the most common problems the women reported.
"We really didn’t appreciate the impact psoriasis had on relationships" before the survey, Dr. Cather said at the Skin Disease Education Foundation’s annual Las Vegas dermatology seminar. "The sexual impact has been underappreciated," she said.
Those problems might help explain why women with psoriasis, especially those aged younger than 35 years, tend to have fewer babies, she added. Psoriatic women also have higher rates of induced and spontaneous abortions and are more likely to have preterm and underweight births.
Some of the women surveyed said that they worried about passing psoriasis to their children. "Maybe there’s voluntary childlessness," Dr. Cather said. But when women in her practice mention they don’t want kids for fear of passing on the disease, "I answer right back, ‘You don’t have to have what you have. I can help you with it,’ " she said.
Methotrexate, acitretin, and psoralen photochemotherapy (PUVA) are contraindicated during pregnancy, but ultraviolet B (UVB) treatments are safe, said Dr. Cather. She also uses tumor necrosis factor inhibitors, which are FDA pregnancy category B agents; she prefers etanercept for its short half-life (just over 4 days) and because ob.gyns. are usually familiar with it. The Organization of Teratology Information Specialists (OTIS) keeps the etanercept and adalimumab pregnancy registries, and "there’s [been] no signal to date" for those drugs, she said.
Psoriatic women should know that the odds are with them for having a normal pregnancy, and that pregnancy is likely to help clear their skin, Dr. Cather said.
But because half of the pregnancies in the United States are unplanned, it’s important to discuss pregnancy – and psoriasis treatment during pregnancy – as part of routine care. "It’s difficult if a psoriasis patient calls you in a panic because they’re not sure their drug is safe, and not sure their doctor will let them continue it,’ she said. Some ob.gyns. are comfortable with letting women stay on their psoriasis therapy, while others want them to quit everything, even topical steroids.
"In my clinic, when a psoriasis patient gets pregnant, usually we’ve planned for it and are excited about it, and we’ve had some dialogue with the ob.gyn.," she explained.
SDEF and this news organization are owned by Frontline Medical Communications. Dr. Cather is a consultant, speaker, or researcher for AbbVie, Novartis, Leo, Janssen, Amgen, Celgene, Merck, and Pfizer.
LAS VEGAS – Psoriasis has a negative impact on women’s sexual desire, sexual ability, and sexual relationships, according to Dr. Jennifer Cather of the Modern Dermatology-Aesthetics Center in Dallas.
Dr. Cather and her colleagues are developing a clinical tool to assess the disease’s sexual impact; as part of those efforts, they conducted 60 interviews with moderately to severely psoriatic women with a mean age of 41 years.
The survey results showed that sexuality was clearly another reason why it’s important to keep psoriasis in check. Pain and itchiness during sex, self-consciousness and embarrassment, and avoidance of dating and intimate relationships were among the most common problems the women reported.
"We really didn’t appreciate the impact psoriasis had on relationships" before the survey, Dr. Cather said at the Skin Disease Education Foundation’s annual Las Vegas dermatology seminar. "The sexual impact has been underappreciated," she said.
Those problems might help explain why women with psoriasis, especially those aged younger than 35 years, tend to have fewer babies, she added. Psoriatic women also have higher rates of induced and spontaneous abortions and are more likely to have preterm and underweight births.
Some of the women surveyed said that they worried about passing psoriasis to their children. "Maybe there’s voluntary childlessness," Dr. Cather said. But when women in her practice mention they don’t want kids for fear of passing on the disease, "I answer right back, ‘You don’t have to have what you have. I can help you with it,’ " she said.
Methotrexate, acitretin, and psoralen photochemotherapy (PUVA) are contraindicated during pregnancy, but ultraviolet B (UVB) treatments are safe, said Dr. Cather. She also uses tumor necrosis factor inhibitors, which are FDA pregnancy category B agents; she prefers etanercept for its short half-life (just over 4 days) and because ob.gyns. are usually familiar with it. The Organization of Teratology Information Specialists (OTIS) keeps the etanercept and adalimumab pregnancy registries, and "there’s [been] no signal to date" for those drugs, she said.
Psoriatic women should know that the odds are with them for having a normal pregnancy, and that pregnancy is likely to help clear their skin, Dr. Cather said.
But because half of the pregnancies in the United States are unplanned, it’s important to discuss pregnancy – and psoriasis treatment during pregnancy – as part of routine care. "It’s difficult if a psoriasis patient calls you in a panic because they’re not sure their drug is safe, and not sure their doctor will let them continue it,’ she said. Some ob.gyns. are comfortable with letting women stay on their psoriasis therapy, while others want them to quit everything, even topical steroids.
"In my clinic, when a psoriasis patient gets pregnant, usually we’ve planned for it and are excited about it, and we’ve had some dialogue with the ob.gyn.," she explained.
SDEF and this news organization are owned by Frontline Medical Communications. Dr. Cather is a consultant, speaker, or researcher for AbbVie, Novartis, Leo, Janssen, Amgen, Celgene, Merck, and Pfizer.
EXPERT ANALYSIS FROM SDEF LAS VEGAS DERMATOLOGY SEMINAR
Risk-based screening misses hepatitis C in many pregnant women
WASHINGTON – Programs to screen pregnant women for hepatitis C infection could go far to reduce a host of adverse outcomes, according to an analysis of a large national sample.
"Targeted HCV screening may overlook many pregnant women with chronic HCV infections, and that may contribute to the underdiagnosis of pediatric hepatitis C in the United States," study investigator Dr. Po-Hung Chen said at the annual meeting of the American Association for the Study of Liver Diseases.
"Chronic hepatitis C is associated with adverse maternal/fetal consequences. In light of these findings, there seems to be a need to further evaluate universal hepatitis C screening as a part of antepartum care," he added.
Dr. Chen of the division of gastroenterology at Johns Hopkins University, Baltimore, and colleagues collected data on all births and spontaneous abortions recorded in the National Inpatient Sample between 2003 and 2010.
More than 28,000 of the 32 million deliveries or miscarriages recorded in the National Inpatient Sample were to mothers infected with HCV, he reported.
Of HCV-positive mothers, 72% had no traditional risk factors for the disease.
HCV-positive mothers were significantly more likely to experience obstetric pulmonary embolism (adjusted OR, 3.05) and thyroid dysfunction (aOR, 1.37), and more likely to experience maternal death, but not significantly so (aOR, 2.49). They also were significantly more likely to be white, less affluent, on Medicaid, substance abusers, and have more comorbidities.
Women who were HCV positive were significantly more likely to have labor before 37 weeks’ gestation (aOR, 1.36), antepartum hemorrhage (aOR, 1.44), and poor fetal growth (aOR, 1.61).
Cost of care and length of stay were significantly greater for mothers who were HCV positive, Dr. Chen said.
Previous analysis using data from the National Health and Nutrition Examination Survey (NHANES) showed that the prevalence of hepatitis C virus infection in women aged 20-39 years was 1%-1.6% in the period between 1999 and 2002. Dr. Chen noted that this could be an underestimation because NHANES is based on home health surveys and therefore does not include prison populations and homeless women, who are at high risk for HCV infection.
Currently, no definitive guidelines exist on how to manage chronic HCV in pregnant women, likely because of a dearth of suitable proven management options, Dr. Chen said, adding that ribavirin is contraindicated in pregnancy, interferon is generally not recommended, and the currently approved protease inhibitors are not options for single-drug management.
The American College of Obstetricians and Gynecologists does not recommend routine prenatal screening for HCV; instead, the college recommends screening only in women who are at high risk based on Centers for Disease Control and Prevention criteria.
Dr. Chen did not report financial conflicts of interest.
WASHINGTON – Programs to screen pregnant women for hepatitis C infection could go far to reduce a host of adverse outcomes, according to an analysis of a large national sample.
"Targeted HCV screening may overlook many pregnant women with chronic HCV infections, and that may contribute to the underdiagnosis of pediatric hepatitis C in the United States," study investigator Dr. Po-Hung Chen said at the annual meeting of the American Association for the Study of Liver Diseases.
"Chronic hepatitis C is associated with adverse maternal/fetal consequences. In light of these findings, there seems to be a need to further evaluate universal hepatitis C screening as a part of antepartum care," he added.
Dr. Chen of the division of gastroenterology at Johns Hopkins University, Baltimore, and colleagues collected data on all births and spontaneous abortions recorded in the National Inpatient Sample between 2003 and 2010.
More than 28,000 of the 32 million deliveries or miscarriages recorded in the National Inpatient Sample were to mothers infected with HCV, he reported.
Of HCV-positive mothers, 72% had no traditional risk factors for the disease.
HCV-positive mothers were significantly more likely to experience obstetric pulmonary embolism (adjusted OR, 3.05) and thyroid dysfunction (aOR, 1.37), and more likely to experience maternal death, but not significantly so (aOR, 2.49). They also were significantly more likely to be white, less affluent, on Medicaid, substance abusers, and have more comorbidities.
Women who were HCV positive were significantly more likely to have labor before 37 weeks’ gestation (aOR, 1.36), antepartum hemorrhage (aOR, 1.44), and poor fetal growth (aOR, 1.61).
Cost of care and length of stay were significantly greater for mothers who were HCV positive, Dr. Chen said.
Previous analysis using data from the National Health and Nutrition Examination Survey (NHANES) showed that the prevalence of hepatitis C virus infection in women aged 20-39 years was 1%-1.6% in the period between 1999 and 2002. Dr. Chen noted that this could be an underestimation because NHANES is based on home health surveys and therefore does not include prison populations and homeless women, who are at high risk for HCV infection.
Currently, no definitive guidelines exist on how to manage chronic HCV in pregnant women, likely because of a dearth of suitable proven management options, Dr. Chen said, adding that ribavirin is contraindicated in pregnancy, interferon is generally not recommended, and the currently approved protease inhibitors are not options for single-drug management.
The American College of Obstetricians and Gynecologists does not recommend routine prenatal screening for HCV; instead, the college recommends screening only in women who are at high risk based on Centers for Disease Control and Prevention criteria.
Dr. Chen did not report financial conflicts of interest.
WASHINGTON – Programs to screen pregnant women for hepatitis C infection could go far to reduce a host of adverse outcomes, according to an analysis of a large national sample.
"Targeted HCV screening may overlook many pregnant women with chronic HCV infections, and that may contribute to the underdiagnosis of pediatric hepatitis C in the United States," study investigator Dr. Po-Hung Chen said at the annual meeting of the American Association for the Study of Liver Diseases.
"Chronic hepatitis C is associated with adverse maternal/fetal consequences. In light of these findings, there seems to be a need to further evaluate universal hepatitis C screening as a part of antepartum care," he added.
Dr. Chen of the division of gastroenterology at Johns Hopkins University, Baltimore, and colleagues collected data on all births and spontaneous abortions recorded in the National Inpatient Sample between 2003 and 2010.
More than 28,000 of the 32 million deliveries or miscarriages recorded in the National Inpatient Sample were to mothers infected with HCV, he reported.
Of HCV-positive mothers, 72% had no traditional risk factors for the disease.
HCV-positive mothers were significantly more likely to experience obstetric pulmonary embolism (adjusted OR, 3.05) and thyroid dysfunction (aOR, 1.37), and more likely to experience maternal death, but not significantly so (aOR, 2.49). They also were significantly more likely to be white, less affluent, on Medicaid, substance abusers, and have more comorbidities.
Women who were HCV positive were significantly more likely to have labor before 37 weeks’ gestation (aOR, 1.36), antepartum hemorrhage (aOR, 1.44), and poor fetal growth (aOR, 1.61).
Cost of care and length of stay were significantly greater for mothers who were HCV positive, Dr. Chen said.
Previous analysis using data from the National Health and Nutrition Examination Survey (NHANES) showed that the prevalence of hepatitis C virus infection in women aged 20-39 years was 1%-1.6% in the period between 1999 and 2002. Dr. Chen noted that this could be an underestimation because NHANES is based on home health surveys and therefore does not include prison populations and homeless women, who are at high risk for HCV infection.
Currently, no definitive guidelines exist on how to manage chronic HCV in pregnant women, likely because of a dearth of suitable proven management options, Dr. Chen said, adding that ribavirin is contraindicated in pregnancy, interferon is generally not recommended, and the currently approved protease inhibitors are not options for single-drug management.
The American College of Obstetricians and Gynecologists does not recommend routine prenatal screening for HCV; instead, the college recommends screening only in women who are at high risk based on Centers for Disease Control and Prevention criteria.
Dr. Chen did not report financial conflicts of interest.
AT THE LIVER MEETING 2013
Major finding: Hepatitis C–positive mothers were significantly more likely than HCV-negative mothers to suffer obstetric pulmonary embolism (adjusted OR, 3.05) and maternal death (aOR, 2.49).
Data source: Analysis of 32 million births or miscarriages in the National Inpatient Sample, 2003-2011.
Disclosures: Dr. Chen did not report financial conflicts of interest.
Checklist for perineal lacerations: Readers weigh in
In a recent editorial, OBG Management Editor in Chief Dr. Robert L. Barbieri presented a draft checklist to guide clinicians during repair of third- and fourth-degree perineal lacerations. He also invited the journal’s readers to modify the checklist as they saw fit—and plenty of you responded! Here is a selection of the suggestions we received, with Dr. Barbieri’s reply.
How I adjusted my surgical protocol for the repair of severe perineal lacerations
I appreciate the clinical pearls offered by Dr. Robert Barbieri in his editorial on the repair of severe perineal lacerations. In response, I plan to change my protocol to initiate antibiotics prior to repair.
I would also offer this tip: When addressing the rectal mucosa, try to stay on a submucosal level, and perform a post-repair rectal exam to ensure that the mucosa has not been violated by a suture and to reduce the risk of rectovaginal fistula.
I repair the internal sphincter using an interrupted technique. And I place only three sutures in the external sphincter, at 8, 12, and 4 o’clock.
Martin E. Kanoff, DO
Sewell, New Jersey
A few pearls on checklist design
I am a big proponent of checklists and use them daily in my professional and personal life. However, as Atul Gawande, MD, MPH, mentioned in his book The Checklist Manifesto, checklists can’t teach a pilot how to fly a plane—that is best achieved through simulation. Dr. Gawande also noted that checklists should be short and to the point, usually containing no more than 10 items, and should highlight things more likely to be overlooked.
For the repair of perineal lacerations, a checklist might include:
- Call for help
- Consider repair in an OR
- Administer preoperative antibiotics
- Ensure adequate exposure and visualization
- Repair in layers (?)
- Postprocedure laxative
- Prophylaxis for deep venous thrombosis (?)
- Return to office in 1 week (?).
The items followed by a question mark are of uncertain value.
Ideally, each checklist should undergo validation via dry runs and team training. In my experience, it is not as difficult to design a checklist as to design a system where checklists get executed entirely and correctly.
Alex Shilkrut, MD
Safety Director, Department of Obstetrics and Gynecology, Metropolitan Hospital, New York, New York
A question about sphincters
I am grateful for Dr. Barbieri’s excellent editorial on using a checklist. I was taught that three sphincters require attention in 4th-degree lacerations: the internal anal sphincter, the external anal sphincter, and the superficial transverse perineal muscle. Would Dr. Barbieri agree?
John Lavin, MD
Kalispell, Montana
QUICK POLL RESULTS
Ensure adequate anesthesia
I appreciate the checklist. I would add to it an explicit statement that the patient must be well-anesthetized, up to and including any readministration of epidural anesthesia or intravenous conscious sedation.
Marcie Richardson, MD
Boston, Massachusetts
My experience repairing severe perineal lacerations
Dr. Barbieri’s column provided an excellent and timely summary! I have been repairing severe perineal lacerations consistently since I was taught a technique during my first year of residency in 1976. Since 1980, I have been at a community hospital with an average of 400 deliveries per year. In more than 33 years, I have had only one repair fail (knock wood!). It was readdressed without problems 1 month later after a low-residue diet, bowel prep, and other preparatory strategies. Interestingly, the patient delivered another 10-lb infant vaginally several years later with no problems, after a cesarean had been advised and declined.
Here is my rationale for the repair of severe lacerations:
- Identification of the extent of the laceration is the first and most important step. In many cases, a mucosal tear may be present in the setting of an intact transverse perineal muscle and even perineum.
- I have never had to move a patient to the OR, but sometimes I ask a nurse to hold retractors, as needed, especially if the patient is large. I use the Gelpi retractor for repair of every fourth-degree laceration.
- I have never given prophylactic antibiotics prior to repair of a third- or fourth-degree laceration. Known or suspected amnionitis or endometritis may be appropriate indications for such prophylaxis, however.
- The apex of the repair is critical, but I start a few millimeters above the laceration rather than 1 cm. Certainly more is better. The suture I use for the rectal mucosa is 3-0 GI chromic suture on a tapered needle in a running fashion, carried to the nadir of the tear. I tag that stitch to mark the lower limit of the repair. I then use the same type of suture to bring the internal anal sphincter and serosa together over the first layer in a running, interlocking fashion, knowing where to stop by virtue of the tagged stitch.
- I use 2-0 chromic figure-of-eight stitches for the external sphincter/transverse perineal muscle capsule at the 3, 6, 12, and 9 o’clock positions as suggested, which brings the clamps together in the midline. (They are removed prior to tying of the 9 o’clock stitch.) The position of the knots is not important, and I have always performed an end-to-end repair rather than overlapping. I always repair the vaginal portion of the tear or episiotomy before attending to the muscular portion of the perineal repair, as access to the vagina is more difficult to achieve afterward. If significant open space is noted behind or superior to the muscle repair, then interrupted stitches of 2-0 chromic suture can be used to approximate tissue and close dead space. The perineal skin is then closed in the usual fashion, down to the tagged initial stitch, or upward from it, if you prefer. Always perform a rectal exam at the conclusion to ensure that the repair is intact.
- I give stool softeners rather than laxatives twice daily for 2 weeks. After the initial 24 to 48 hours, I recommend use of a nonsteroidal anti-inflammatory drug for discomfort rather than medications that slow bowel transit time. I also recommend sitz baths four times daily for at least 1 week, after which they can be given as needed. Ice packs can be applied to the swollen perineum initially for relief of symptoms while the patient is on the postpartum service.
- Rarely has a patient needed to be seen before the normally scheduled postpartum visit. However, with reports of anal incontinence becoming more frequent in the literature, I make it a point to ask about this problem. I have had to refer only two patients for anal manometry, and neither patient had a fourth-degree laceration. Both patients delivered elsewhere.
I am not convinced that another checklist is needed, as we may end up with a cookbook approach if we aren’t careful. Just do the job correctly with a well-thought-out rationale for your approach.
Donald R. Wilson, MD
Maysville, Kentucky
A few questions about bowel preparation
I appreciate the clear-cut guidelines presented in this editorial. I have taught gynecology for 25 years at an Egyptian university. The addition of critical details to our surgical protocols will be of great help in improving outcomes. However, I have one major concern that was not mentioned in the article, and it involves bowel preparation.
Not all women undergo colon preparation and enema before coming to the labor room. Some are transferred from the emergency room or come to the hospital after home delivery. These women may have a full rectum and colon, sometimes even constipation with hard fecal matter, or they may have eaten recently.
How should these women be managed when a severe perineal laceration is present? Should the contaminated area be addressed first, followed by repair? Also, it seems to me that a full rectum and colon at the time of repair will place the sutures under undue stress, risking rupture with the patient’s first defecation.
In the past, we cleaned all areas of contamination with a povidone-iodine solution and administered a constipating regimen for 5 to 7 days postoperatively, with fluids and parenteral treatment, followed by laxatives and lactulose for 2 days before gradually incorporating a regular diet. Some physicians have proposed administering lactulose or a similar enema in the delivery room to evacuate bowel contents, followed by antisepsis, repair, and antibiotics.
I wonder how Dr. Barbieri would address this scenario?
Magdy Hassan Balaha, MD
Ahsaa, Saudi Arabia
More on the need for analgesia and anesthesia
I have a suggestion for checklists for the repair of severe perineal lacerations: Add an item mentioning the need to ensure adequate analgesia/anesthesia. Besides being kind to the patient, this step may facilitate exposure.
Also important is an environment that encourages consultation by any provider confronted with a perineal laceration of uncertain extent or unclear anatomy.
Nancy Kerr, MD, MPH
Telluride, Colorado, and Albuquerque, New Mexico
Fluids are important
When I repair fourth-degree lacerations, I use at least 1 L of normal saline in 100- to 200-cc aliquots throughout the repair.
Paul G. Crawford, MD
Shreveport, Louisiana
DR. BARBIERI RESPONDS:
We thank Dr. Kanoff, Dr. Shilkrut, Dr. Richardson, Dr. Wilson, Dr. Hassan Balaha, Dr. Kerr, and Dr. Crawford for their excellent additions to the perineal laceration checklist, all of which I support.
Dr. Richardson and Dr. Kerr specifically mention adequate analgesia/anesthesia, which is critically important to facilitate an optimal repair. Dr. Lavin mentions the repair of three sphincters: internal and external rectal sphincters and the superficial transverse perineal muscle. I am not aware that the superficial transverse perineal muscle is a classic “sphincter,” but it is important to include it in the repair of second-degree perineal lacerations.
Dr. Hassan Balaha asks about the handling of a contaminated field during repair. There are no randomized studies addressing this problem; most obstetricians will cleanse the area as well as possible and proceed with the repair. Dr. Hassan Balaha also recommends a two-step bowel regimen, involving a constipating regimen for 5 to 7 days, followed by laxatives and lactulose for 2 days BEFORE initiating a regular diet—a very interesting idea, which I will share with my colleagues. As noted in the editorial, the only clinical trial of this situation randomized women to a narcotic (constipating regimen) or a laxative regimen. The women in the laxative group reported earlier and less painful bowel movements than the women in the narcotic group.1
We want to hear from you! Tell us what you think.
Reference
- Mahony R, Behan M, O’Herlihy C, O’Connell PR. Randomised clinical trial of bowel confinement vs. laxative use after primary repair of a third degree obstetric anal sphincter tear. Dis Colon Rectum. 2004;47(1):12–17.
In a recent editorial, OBG Management Editor in Chief Dr. Robert L. Barbieri presented a draft checklist to guide clinicians during repair of third- and fourth-degree perineal lacerations. He also invited the journal’s readers to modify the checklist as they saw fit—and plenty of you responded! Here is a selection of the suggestions we received, with Dr. Barbieri’s reply.
How I adjusted my surgical protocol for the repair of severe perineal lacerations
I appreciate the clinical pearls offered by Dr. Robert Barbieri in his editorial on the repair of severe perineal lacerations. In response, I plan to change my protocol to initiate antibiotics prior to repair.
I would also offer this tip: When addressing the rectal mucosa, try to stay on a submucosal level, and perform a post-repair rectal exam to ensure that the mucosa has not been violated by a suture and to reduce the risk of rectovaginal fistula.
I repair the internal sphincter using an interrupted technique. And I place only three sutures in the external sphincter, at 8, 12, and 4 o’clock.
Martin E. Kanoff, DO
Sewell, New Jersey
A few pearls on checklist design
I am a big proponent of checklists and use them daily in my professional and personal life. However, as Atul Gawande, MD, MPH, mentioned in his book The Checklist Manifesto, checklists can’t teach a pilot how to fly a plane—that is best achieved through simulation. Dr. Gawande also noted that checklists should be short and to the point, usually containing no more than 10 items, and should highlight things more likely to be overlooked.
For the repair of perineal lacerations, a checklist might include:
- Call for help
- Consider repair in an OR
- Administer preoperative antibiotics
- Ensure adequate exposure and visualization
- Repair in layers (?)
- Postprocedure laxative
- Prophylaxis for deep venous thrombosis (?)
- Return to office in 1 week (?).
The items followed by a question mark are of uncertain value.
Ideally, each checklist should undergo validation via dry runs and team training. In my experience, it is not as difficult to design a checklist as to design a system where checklists get executed entirely and correctly.
Alex Shilkrut, MD
Safety Director, Department of Obstetrics and Gynecology, Metropolitan Hospital, New York, New York
A question about sphincters
I am grateful for Dr. Barbieri’s excellent editorial on using a checklist. I was taught that three sphincters require attention in 4th-degree lacerations: the internal anal sphincter, the external anal sphincter, and the superficial transverse perineal muscle. Would Dr. Barbieri agree?
John Lavin, MD
Kalispell, Montana
QUICK POLL RESULTS
Ensure adequate anesthesia
I appreciate the checklist. I would add to it an explicit statement that the patient must be well-anesthetized, up to and including any readministration of epidural anesthesia or intravenous conscious sedation.
Marcie Richardson, MD
Boston, Massachusetts
My experience repairing severe perineal lacerations
Dr. Barbieri’s column provided an excellent and timely summary! I have been repairing severe perineal lacerations consistently since I was taught a technique during my first year of residency in 1976. Since 1980, I have been at a community hospital with an average of 400 deliveries per year. In more than 33 years, I have had only one repair fail (knock wood!). It was readdressed without problems 1 month later after a low-residue diet, bowel prep, and other preparatory strategies. Interestingly, the patient delivered another 10-lb infant vaginally several years later with no problems, after a cesarean had been advised and declined.
Here is my rationale for the repair of severe lacerations:
- Identification of the extent of the laceration is the first and most important step. In many cases, a mucosal tear may be present in the setting of an intact transverse perineal muscle and even perineum.
- I have never had to move a patient to the OR, but sometimes I ask a nurse to hold retractors, as needed, especially if the patient is large. I use the Gelpi retractor for repair of every fourth-degree laceration.
- I have never given prophylactic antibiotics prior to repair of a third- or fourth-degree laceration. Known or suspected amnionitis or endometritis may be appropriate indications for such prophylaxis, however.
- The apex of the repair is critical, but I start a few millimeters above the laceration rather than 1 cm. Certainly more is better. The suture I use for the rectal mucosa is 3-0 GI chromic suture on a tapered needle in a running fashion, carried to the nadir of the tear. I tag that stitch to mark the lower limit of the repair. I then use the same type of suture to bring the internal anal sphincter and serosa together over the first layer in a running, interlocking fashion, knowing where to stop by virtue of the tagged stitch.
- I use 2-0 chromic figure-of-eight stitches for the external sphincter/transverse perineal muscle capsule at the 3, 6, 12, and 9 o’clock positions as suggested, which brings the clamps together in the midline. (They are removed prior to tying of the 9 o’clock stitch.) The position of the knots is not important, and I have always performed an end-to-end repair rather than overlapping. I always repair the vaginal portion of the tear or episiotomy before attending to the muscular portion of the perineal repair, as access to the vagina is more difficult to achieve afterward. If significant open space is noted behind or superior to the muscle repair, then interrupted stitches of 2-0 chromic suture can be used to approximate tissue and close dead space. The perineal skin is then closed in the usual fashion, down to the tagged initial stitch, or upward from it, if you prefer. Always perform a rectal exam at the conclusion to ensure that the repair is intact.
- I give stool softeners rather than laxatives twice daily for 2 weeks. After the initial 24 to 48 hours, I recommend use of a nonsteroidal anti-inflammatory drug for discomfort rather than medications that slow bowel transit time. I also recommend sitz baths four times daily for at least 1 week, after which they can be given as needed. Ice packs can be applied to the swollen perineum initially for relief of symptoms while the patient is on the postpartum service.
- Rarely has a patient needed to be seen before the normally scheduled postpartum visit. However, with reports of anal incontinence becoming more frequent in the literature, I make it a point to ask about this problem. I have had to refer only two patients for anal manometry, and neither patient had a fourth-degree laceration. Both patients delivered elsewhere.
I am not convinced that another checklist is needed, as we may end up with a cookbook approach if we aren’t careful. Just do the job correctly with a well-thought-out rationale for your approach.
Donald R. Wilson, MD
Maysville, Kentucky
A few questions about bowel preparation
I appreciate the clear-cut guidelines presented in this editorial. I have taught gynecology for 25 years at an Egyptian university. The addition of critical details to our surgical protocols will be of great help in improving outcomes. However, I have one major concern that was not mentioned in the article, and it involves bowel preparation.
Not all women undergo colon preparation and enema before coming to the labor room. Some are transferred from the emergency room or come to the hospital after home delivery. These women may have a full rectum and colon, sometimes even constipation with hard fecal matter, or they may have eaten recently.
How should these women be managed when a severe perineal laceration is present? Should the contaminated area be addressed first, followed by repair? Also, it seems to me that a full rectum and colon at the time of repair will place the sutures under undue stress, risking rupture with the patient’s first defecation.
In the past, we cleaned all areas of contamination with a povidone-iodine solution and administered a constipating regimen for 5 to 7 days postoperatively, with fluids and parenteral treatment, followed by laxatives and lactulose for 2 days before gradually incorporating a regular diet. Some physicians have proposed administering lactulose or a similar enema in the delivery room to evacuate bowel contents, followed by antisepsis, repair, and antibiotics.
I wonder how Dr. Barbieri would address this scenario?
Magdy Hassan Balaha, MD
Ahsaa, Saudi Arabia
More on the need for analgesia and anesthesia
I have a suggestion for checklists for the repair of severe perineal lacerations: Add an item mentioning the need to ensure adequate analgesia/anesthesia. Besides being kind to the patient, this step may facilitate exposure.
Also important is an environment that encourages consultation by any provider confronted with a perineal laceration of uncertain extent or unclear anatomy.
Nancy Kerr, MD, MPH
Telluride, Colorado, and Albuquerque, New Mexico
Fluids are important
When I repair fourth-degree lacerations, I use at least 1 L of normal saline in 100- to 200-cc aliquots throughout the repair.
Paul G. Crawford, MD
Shreveport, Louisiana
DR. BARBIERI RESPONDS:
We thank Dr. Kanoff, Dr. Shilkrut, Dr. Richardson, Dr. Wilson, Dr. Hassan Balaha, Dr. Kerr, and Dr. Crawford for their excellent additions to the perineal laceration checklist, all of which I support.
Dr. Richardson and Dr. Kerr specifically mention adequate analgesia/anesthesia, which is critically important to facilitate an optimal repair. Dr. Lavin mentions the repair of three sphincters: internal and external rectal sphincters and the superficial transverse perineal muscle. I am not aware that the superficial transverse perineal muscle is a classic “sphincter,” but it is important to include it in the repair of second-degree perineal lacerations.
Dr. Hassan Balaha asks about the handling of a contaminated field during repair. There are no randomized studies addressing this problem; most obstetricians will cleanse the area as well as possible and proceed with the repair. Dr. Hassan Balaha also recommends a two-step bowel regimen, involving a constipating regimen for 5 to 7 days, followed by laxatives and lactulose for 2 days BEFORE initiating a regular diet—a very interesting idea, which I will share with my colleagues. As noted in the editorial, the only clinical trial of this situation randomized women to a narcotic (constipating regimen) or a laxative regimen. The women in the laxative group reported earlier and less painful bowel movements than the women in the narcotic group.1
We want to hear from you! Tell us what you think.
In a recent editorial, OBG Management Editor in Chief Dr. Robert L. Barbieri presented a draft checklist to guide clinicians during repair of third- and fourth-degree perineal lacerations. He also invited the journal’s readers to modify the checklist as they saw fit—and plenty of you responded! Here is a selection of the suggestions we received, with Dr. Barbieri’s reply.
How I adjusted my surgical protocol for the repair of severe perineal lacerations
I appreciate the clinical pearls offered by Dr. Robert Barbieri in his editorial on the repair of severe perineal lacerations. In response, I plan to change my protocol to initiate antibiotics prior to repair.
I would also offer this tip: When addressing the rectal mucosa, try to stay on a submucosal level, and perform a post-repair rectal exam to ensure that the mucosa has not been violated by a suture and to reduce the risk of rectovaginal fistula.
I repair the internal sphincter using an interrupted technique. And I place only three sutures in the external sphincter, at 8, 12, and 4 o’clock.
Martin E. Kanoff, DO
Sewell, New Jersey
A few pearls on checklist design
I am a big proponent of checklists and use them daily in my professional and personal life. However, as Atul Gawande, MD, MPH, mentioned in his book The Checklist Manifesto, checklists can’t teach a pilot how to fly a plane—that is best achieved through simulation. Dr. Gawande also noted that checklists should be short and to the point, usually containing no more than 10 items, and should highlight things more likely to be overlooked.
For the repair of perineal lacerations, a checklist might include:
- Call for help
- Consider repair in an OR
- Administer preoperative antibiotics
- Ensure adequate exposure and visualization
- Repair in layers (?)
- Postprocedure laxative
- Prophylaxis for deep venous thrombosis (?)
- Return to office in 1 week (?).
The items followed by a question mark are of uncertain value.
Ideally, each checklist should undergo validation via dry runs and team training. In my experience, it is not as difficult to design a checklist as to design a system where checklists get executed entirely and correctly.
Alex Shilkrut, MD
Safety Director, Department of Obstetrics and Gynecology, Metropolitan Hospital, New York, New York
A question about sphincters
I am grateful for Dr. Barbieri’s excellent editorial on using a checklist. I was taught that three sphincters require attention in 4th-degree lacerations: the internal anal sphincter, the external anal sphincter, and the superficial transverse perineal muscle. Would Dr. Barbieri agree?
John Lavin, MD
Kalispell, Montana
QUICK POLL RESULTS
Ensure adequate anesthesia
I appreciate the checklist. I would add to it an explicit statement that the patient must be well-anesthetized, up to and including any readministration of epidural anesthesia or intravenous conscious sedation.
Marcie Richardson, MD
Boston, Massachusetts
My experience repairing severe perineal lacerations
Dr. Barbieri’s column provided an excellent and timely summary! I have been repairing severe perineal lacerations consistently since I was taught a technique during my first year of residency in 1976. Since 1980, I have been at a community hospital with an average of 400 deliveries per year. In more than 33 years, I have had only one repair fail (knock wood!). It was readdressed without problems 1 month later after a low-residue diet, bowel prep, and other preparatory strategies. Interestingly, the patient delivered another 10-lb infant vaginally several years later with no problems, after a cesarean had been advised and declined.
Here is my rationale for the repair of severe lacerations:
- Identification of the extent of the laceration is the first and most important step. In many cases, a mucosal tear may be present in the setting of an intact transverse perineal muscle and even perineum.
- I have never had to move a patient to the OR, but sometimes I ask a nurse to hold retractors, as needed, especially if the patient is large. I use the Gelpi retractor for repair of every fourth-degree laceration.
- I have never given prophylactic antibiotics prior to repair of a third- or fourth-degree laceration. Known or suspected amnionitis or endometritis may be appropriate indications for such prophylaxis, however.
- The apex of the repair is critical, but I start a few millimeters above the laceration rather than 1 cm. Certainly more is better. The suture I use for the rectal mucosa is 3-0 GI chromic suture on a tapered needle in a running fashion, carried to the nadir of the tear. I tag that stitch to mark the lower limit of the repair. I then use the same type of suture to bring the internal anal sphincter and serosa together over the first layer in a running, interlocking fashion, knowing where to stop by virtue of the tagged stitch.
- I use 2-0 chromic figure-of-eight stitches for the external sphincter/transverse perineal muscle capsule at the 3, 6, 12, and 9 o’clock positions as suggested, which brings the clamps together in the midline. (They are removed prior to tying of the 9 o’clock stitch.) The position of the knots is not important, and I have always performed an end-to-end repair rather than overlapping. I always repair the vaginal portion of the tear or episiotomy before attending to the muscular portion of the perineal repair, as access to the vagina is more difficult to achieve afterward. If significant open space is noted behind or superior to the muscle repair, then interrupted stitches of 2-0 chromic suture can be used to approximate tissue and close dead space. The perineal skin is then closed in the usual fashion, down to the tagged initial stitch, or upward from it, if you prefer. Always perform a rectal exam at the conclusion to ensure that the repair is intact.
- I give stool softeners rather than laxatives twice daily for 2 weeks. After the initial 24 to 48 hours, I recommend use of a nonsteroidal anti-inflammatory drug for discomfort rather than medications that slow bowel transit time. I also recommend sitz baths four times daily for at least 1 week, after which they can be given as needed. Ice packs can be applied to the swollen perineum initially for relief of symptoms while the patient is on the postpartum service.
- Rarely has a patient needed to be seen before the normally scheduled postpartum visit. However, with reports of anal incontinence becoming more frequent in the literature, I make it a point to ask about this problem. I have had to refer only two patients for anal manometry, and neither patient had a fourth-degree laceration. Both patients delivered elsewhere.
I am not convinced that another checklist is needed, as we may end up with a cookbook approach if we aren’t careful. Just do the job correctly with a well-thought-out rationale for your approach.
Donald R. Wilson, MD
Maysville, Kentucky
A few questions about bowel preparation
I appreciate the clear-cut guidelines presented in this editorial. I have taught gynecology for 25 years at an Egyptian university. The addition of critical details to our surgical protocols will be of great help in improving outcomes. However, I have one major concern that was not mentioned in the article, and it involves bowel preparation.
Not all women undergo colon preparation and enema before coming to the labor room. Some are transferred from the emergency room or come to the hospital after home delivery. These women may have a full rectum and colon, sometimes even constipation with hard fecal matter, or they may have eaten recently.
How should these women be managed when a severe perineal laceration is present? Should the contaminated area be addressed first, followed by repair? Also, it seems to me that a full rectum and colon at the time of repair will place the sutures under undue stress, risking rupture with the patient’s first defecation.
In the past, we cleaned all areas of contamination with a povidone-iodine solution and administered a constipating regimen for 5 to 7 days postoperatively, with fluids and parenteral treatment, followed by laxatives and lactulose for 2 days before gradually incorporating a regular diet. Some physicians have proposed administering lactulose or a similar enema in the delivery room to evacuate bowel contents, followed by antisepsis, repair, and antibiotics.
I wonder how Dr. Barbieri would address this scenario?
Magdy Hassan Balaha, MD
Ahsaa, Saudi Arabia
More on the need for analgesia and anesthesia
I have a suggestion for checklists for the repair of severe perineal lacerations: Add an item mentioning the need to ensure adequate analgesia/anesthesia. Besides being kind to the patient, this step may facilitate exposure.
Also important is an environment that encourages consultation by any provider confronted with a perineal laceration of uncertain extent or unclear anatomy.
Nancy Kerr, MD, MPH
Telluride, Colorado, and Albuquerque, New Mexico
Fluids are important
When I repair fourth-degree lacerations, I use at least 1 L of normal saline in 100- to 200-cc aliquots throughout the repair.
Paul G. Crawford, MD
Shreveport, Louisiana
DR. BARBIERI RESPONDS:
We thank Dr. Kanoff, Dr. Shilkrut, Dr. Richardson, Dr. Wilson, Dr. Hassan Balaha, Dr. Kerr, and Dr. Crawford for their excellent additions to the perineal laceration checklist, all of which I support.
Dr. Richardson and Dr. Kerr specifically mention adequate analgesia/anesthesia, which is critically important to facilitate an optimal repair. Dr. Lavin mentions the repair of three sphincters: internal and external rectal sphincters and the superficial transverse perineal muscle. I am not aware that the superficial transverse perineal muscle is a classic “sphincter,” but it is important to include it in the repair of second-degree perineal lacerations.
Dr. Hassan Balaha asks about the handling of a contaminated field during repair. There are no randomized studies addressing this problem; most obstetricians will cleanse the area as well as possible and proceed with the repair. Dr. Hassan Balaha also recommends a two-step bowel regimen, involving a constipating regimen for 5 to 7 days, followed by laxatives and lactulose for 2 days BEFORE initiating a regular diet—a very interesting idea, which I will share with my colleagues. As noted in the editorial, the only clinical trial of this situation randomized women to a narcotic (constipating regimen) or a laxative regimen. The women in the laxative group reported earlier and less painful bowel movements than the women in the narcotic group.1
We want to hear from you! Tell us what you think.
Reference
- Mahony R, Behan M, O’Herlihy C, O’Connell PR. Randomised clinical trial of bowel confinement vs. laxative use after primary repair of a third degree obstetric anal sphincter tear. Dis Colon Rectum. 2004;47(1):12–17.
Reference
- Mahony R, Behan M, O’Herlihy C, O’Connell PR. Randomised clinical trial of bowel confinement vs. laxative use after primary repair of a third degree obstetric anal sphincter tear. Dis Colon Rectum. 2004;47(1):12–17.
What do the latest data reveal about the safety of home birth in the United States?
Every morning before I leave for work, I kiss my three children goodbye and tell them, “I love you. Make good choices today.”
This has become my mantra—so much so that, on her way out the door to join her friends at the movies recently, my daughter turned to me and said, “I know, Dad. I know. I’ll make good decisions tonight.”
And what decision is more important than where to deliver your child and who to have in attendance at the birth?
It is said that the passage from the uterus to the outside world that each one of us was forced to negotiate at birth is the most treacherous journey we will ever undertake. Any unnecessary delay or complication can have profound, lifelong consequences.
There is no question that the past few centuries have seen a significant “medicalization” of childbirth, including the relocation of deliveries from the community to a hospital setting, the introduction of male obstetricians, the unfortunate marginalization of midwives and support personnel (doulas), the development of uterotonic drugs, and the evolution of operative vaginal (forceps, vacuum) and cesarean deliveries.
Many of the practices initially introduced by obstetric care providers (including multiple vaginal examinations in labor, induction of labor for a large baby, and active management of labor protocols) have since been shown to be unhelpful in improving pregnancy outcomes, and some practices (such as episiotomy) have even been shown to be harmful.
Related article: Difficult fetal extraction at cesarean delivery: What should you do? Robert L. Barbieri, MD (Editorial, January 2012)
In the midst of this confusion, the one voice that has been lost is that of the patient herself.
Whose birth is it anyway?
The American College of Obstetricians and Gynecologists (ACOG), the American Academy of Pediatrics (AAP), and the American College of Nurse-Midwives (ACNM) all agree that patient autonomy is paramount, and that the final decision of where to deliver and who to have in attendance should be made by the patient herself, ideally in conjunction with her family and her obstetric care provider.1–3 But an informed decision is only as good as the available data. Regrettably, the literature on how planned home birth compares with hospital delivery in terms of pregnancy outcomes in the United States are sparse.
Related article: Lay midwives the the ObGyn: Is collaboration risky? Lucia DiVenere, MA (May 2012)
How safe is home birth in the United States?
Cheng and colleagues attempt to answer this question by reviewing newborn and maternal outcomes among planned home births versus hospital deliveries in a contemporary low-risk birth cohort. Their retrospective study included low-risk women at term with a singleton vertex live birth in 2008 in 27 of the 50 states using information from the Vital Statistics Natality Data provided by the Centers for Disease Control and Prevention.
Of these 2,081,753 women, 0.58% (n = 12,039) had planned home births, and the remainder delivered in a hospital setting. Women who had an “accidental” (unintended) home birth or who delivered in a freestanding birthing center were excluded. The primary outcome was the risk of a 5-minute Apgar score less than 4. Secondary outcomes included the risk of a 5-minute Apgar score less than 7, assisted ventilation for more than 6 hours, neonatal seizures, admission to the NICU, and a series of maternal outcome measures.
Besides the outcomes listed previously (top of page 24), women with a planned home birth had fewer obstetric interventions, including operative vaginal delivery and labor induction or augmentation. They also were less likely to be given antibiotics during labor (although the authors did not distinguish between antibiotics administered for prophylaxis against group B strep or surgical-site infection versus antibiotics to treat infections such as urinary tract infections or chorioamnionitis).
Of special interest is the fact that neither a prior vaginal delivery (multiparity) nor the absence of a prior cesarean delivery was protective against these adverse events.
The women at highest risk of an adverse event were those who delivered at home under the supervision of “other midwives.” Although these providers were not well defined, this term typically refers to community-based lay midwives whose only “training” consists of an unofficial apprenticeship of variable length. Despite the absence of formal training, the lack of certification and standardization of care, and the existence of legislation in many states banning their activity, such lay midwives continue to encourage and support home birth for both low- and high-risk women in the United States.
Related article: Update on Obstetrics John T. Repke, MD, and Jaimey M. Pauli, MD (January 2012)
Limitations of the study design
Although this dataset contains more than 2 million births, it includes only low-risk women at term and, therefore, is underpowered to measure outcomes such as fetal or neonatal death or birth injuries.
No data were presented on a number of important variables and outcome measures, such as the rate of or indications for cesarean delivery, the mode and frequency of intrapartum fetal monitoring, birth weight, intrapartum complications (uterine rupture, postpartum hemorrhage), blood transfusions, and infectious morbidity. The study also lacks long-term follow-up data on the infants.
That said, the study was well designed and very well written, and many of the limitations listed above are inherent in all retrospective cohort studies.
Putting these findings in context
These data are not novel, but they are remarkably consistent with other publications that have explored pregnancy outcomes in planned home birth versus hospital delivery from the Netherlands, the United Kingdom, Australia, and the United States, all of which show a higher rate of neonatal complications with planned home birth [see Reference 4 for review].4
Moreover, it is likely that the data in the current report significantly underestimate the risks of planned home birth for two reasons:
- Attempted home births that ended in transfer and, ultimately, delivery in a hospital setting (presumably for some unforeseen event such as excessive hemorrhage or uterine rupture or cord prolapse or nonreassuring fetal testing) were classified as hospital births.
- Apgar scores at 5 minutes are assigned by the attending care provider, and there is no way to independently verify their accuracy. Because of their limited training and/or concern about efforts to limit the scope of their practice, “other midwives” may be inclined to assign more favorable Apgar scores.
Who is choosing to deliver at home?
The proportion of US women who delivered outside the hospital setting increased by 29% between 2004 and 2009,5 although home births still constitute a minority of low-risk births (0.58% in the current study).
Related Article: Why are well-educated women more likely to choose home birth? Errol R. Norwitz, MD, PhD (Audiocast, November 2013)
One of the more interesting questions raised by this publication is the issue of who is choosing to deliver at home. In this cohort, women who planned home birth were more likely to be older, married, multiparous, white, and well educated. These aren’t exactly the women you would expect to gamble with the lives of their unborn offspring. So why are they choosing to deliver at home?
It could be that they are not well informed about the risks. Alternatively, they may have concluded that, although the relative risk of an adverse event is significantly higher with home birth, the absolute risk is low and acceptable to them. Or it could be that they are frustrated by the lack of autonomy afforded to them in the decisions surrounding antenatal care and the birthing process.
In recent years, more women are asking for minimally invasive births that are physically, emotionally, and socially supported. As hospital-based obstetric care providers, we do not always respect or meet these expectations. We can and should do better.
Women should not have to choose between a good birth experience and medical safety, between social support and hospital resources, between a sense of autonomy and access to life-saving interventions. Although every effort should be taken to make the birthing experience a positive one for the mother and her family as a whole, it should not be done at the expense of safety. I have yet to hear an asphyxiated and brain-damaged child thank his mother’s obstetric care provider for allowing a wonderful birth experience.
What this evidence means for practice
Even in countries where home births are integrated fully into the medical care system and attended by trained and certified nurse-midwives, they are associated with increased risks, including a twofold to threefold increase in the odds of neonatal death.4 In the US, where no such integration exists, home births are dangerous.
Maternity care has come a long way since the 17th Century, when a woman had a 1 in 6 chance of dying in childbirth and only one of every five children lived to enjoy a first birthday. It is appropriate in this era of Obamacare and cost containment that we explore alternative methods. The option of a safe home delivery may well be part of the solution, as it is for many European countries--but until we can be assured that such an approach is safe for both mothers and infants, let's keep home delivery where it belongs...for pizza!
--Errol R. Norwitz, MD, PHD
We want to hear from you! Tell us what you think.
- Committee on Obstetric Practice; American College of Obstetricians and Gynecologists. Committee Opinion #476: Planned home birth. Obstet Gynecol. 2011;117(2 Pt 1):425–428.
- American Academy of Pediatrics. Committee on Fetus and Newborn. Planned home birth. Pediatrics. 2013;131(5):1016–1020.
- American College of Nurse-Midwives. Division of Standards and Practice. Position statement: Home birth. Approved by the ACNM Board of Directors, May 2011. http://midwife.org/ACNM/files/ACNMLibraryData/UPLOADFILENAME/000000000251/Home%20Birth%20Aug%202011.pdf. Accessed October 21, 2013
- Wax JR, Lucas FL, Lamont M, et al. Maternal and newborn outcomes in planned home birth vs planned hospital births: A meta-analysis. Am J Obstet Gynecol. 2012;203(3):243.e1–e8.
- Martin JA, Hamilton BE, Ventura SJ, et al; Division of Vital Statistics. Births: Final data for 2009. Natl Vital Stat Rep. 2011;60(1):1–70.
Every morning before I leave for work, I kiss my three children goodbye and tell them, “I love you. Make good choices today.”
This has become my mantra—so much so that, on her way out the door to join her friends at the movies recently, my daughter turned to me and said, “I know, Dad. I know. I’ll make good decisions tonight.”
And what decision is more important than where to deliver your child and who to have in attendance at the birth?
It is said that the passage from the uterus to the outside world that each one of us was forced to negotiate at birth is the most treacherous journey we will ever undertake. Any unnecessary delay or complication can have profound, lifelong consequences.
There is no question that the past few centuries have seen a significant “medicalization” of childbirth, including the relocation of deliveries from the community to a hospital setting, the introduction of male obstetricians, the unfortunate marginalization of midwives and support personnel (doulas), the development of uterotonic drugs, and the evolution of operative vaginal (forceps, vacuum) and cesarean deliveries.
Many of the practices initially introduced by obstetric care providers (including multiple vaginal examinations in labor, induction of labor for a large baby, and active management of labor protocols) have since been shown to be unhelpful in improving pregnancy outcomes, and some practices (such as episiotomy) have even been shown to be harmful.
Related article: Difficult fetal extraction at cesarean delivery: What should you do? Robert L. Barbieri, MD (Editorial, January 2012)
In the midst of this confusion, the one voice that has been lost is that of the patient herself.
Whose birth is it anyway?
The American College of Obstetricians and Gynecologists (ACOG), the American Academy of Pediatrics (AAP), and the American College of Nurse-Midwives (ACNM) all agree that patient autonomy is paramount, and that the final decision of where to deliver and who to have in attendance should be made by the patient herself, ideally in conjunction with her family and her obstetric care provider.1–3 But an informed decision is only as good as the available data. Regrettably, the literature on how planned home birth compares with hospital delivery in terms of pregnancy outcomes in the United States are sparse.
Related article: Lay midwives the the ObGyn: Is collaboration risky? Lucia DiVenere, MA (May 2012)
How safe is home birth in the United States?
Cheng and colleagues attempt to answer this question by reviewing newborn and maternal outcomes among planned home births versus hospital deliveries in a contemporary low-risk birth cohort. Their retrospective study included low-risk women at term with a singleton vertex live birth in 2008 in 27 of the 50 states using information from the Vital Statistics Natality Data provided by the Centers for Disease Control and Prevention.
Of these 2,081,753 women, 0.58% (n = 12,039) had planned home births, and the remainder delivered in a hospital setting. Women who had an “accidental” (unintended) home birth or who delivered in a freestanding birthing center were excluded. The primary outcome was the risk of a 5-minute Apgar score less than 4. Secondary outcomes included the risk of a 5-minute Apgar score less than 7, assisted ventilation for more than 6 hours, neonatal seizures, admission to the NICU, and a series of maternal outcome measures.
Besides the outcomes listed previously (top of page 24), women with a planned home birth had fewer obstetric interventions, including operative vaginal delivery and labor induction or augmentation. They also were less likely to be given antibiotics during labor (although the authors did not distinguish between antibiotics administered for prophylaxis against group B strep or surgical-site infection versus antibiotics to treat infections such as urinary tract infections or chorioamnionitis).
Of special interest is the fact that neither a prior vaginal delivery (multiparity) nor the absence of a prior cesarean delivery was protective against these adverse events.
The women at highest risk of an adverse event were those who delivered at home under the supervision of “other midwives.” Although these providers were not well defined, this term typically refers to community-based lay midwives whose only “training” consists of an unofficial apprenticeship of variable length. Despite the absence of formal training, the lack of certification and standardization of care, and the existence of legislation in many states banning their activity, such lay midwives continue to encourage and support home birth for both low- and high-risk women in the United States.
Related article: Update on Obstetrics John T. Repke, MD, and Jaimey M. Pauli, MD (January 2012)
Limitations of the study design
Although this dataset contains more than 2 million births, it includes only low-risk women at term and, therefore, is underpowered to measure outcomes such as fetal or neonatal death or birth injuries.
No data were presented on a number of important variables and outcome measures, such as the rate of or indications for cesarean delivery, the mode and frequency of intrapartum fetal monitoring, birth weight, intrapartum complications (uterine rupture, postpartum hemorrhage), blood transfusions, and infectious morbidity. The study also lacks long-term follow-up data on the infants.
That said, the study was well designed and very well written, and many of the limitations listed above are inherent in all retrospective cohort studies.
Putting these findings in context
These data are not novel, but they are remarkably consistent with other publications that have explored pregnancy outcomes in planned home birth versus hospital delivery from the Netherlands, the United Kingdom, Australia, and the United States, all of which show a higher rate of neonatal complications with planned home birth [see Reference 4 for review].4
Moreover, it is likely that the data in the current report significantly underestimate the risks of planned home birth for two reasons:
- Attempted home births that ended in transfer and, ultimately, delivery in a hospital setting (presumably for some unforeseen event such as excessive hemorrhage or uterine rupture or cord prolapse or nonreassuring fetal testing) were classified as hospital births.
- Apgar scores at 5 minutes are assigned by the attending care provider, and there is no way to independently verify their accuracy. Because of their limited training and/or concern about efforts to limit the scope of their practice, “other midwives” may be inclined to assign more favorable Apgar scores.
Who is choosing to deliver at home?
The proportion of US women who delivered outside the hospital setting increased by 29% between 2004 and 2009,5 although home births still constitute a minority of low-risk births (0.58% in the current study).
Related Article: Why are well-educated women more likely to choose home birth? Errol R. Norwitz, MD, PhD (Audiocast, November 2013)
One of the more interesting questions raised by this publication is the issue of who is choosing to deliver at home. In this cohort, women who planned home birth were more likely to be older, married, multiparous, white, and well educated. These aren’t exactly the women you would expect to gamble with the lives of their unborn offspring. So why are they choosing to deliver at home?
It could be that they are not well informed about the risks. Alternatively, they may have concluded that, although the relative risk of an adverse event is significantly higher with home birth, the absolute risk is low and acceptable to them. Or it could be that they are frustrated by the lack of autonomy afforded to them in the decisions surrounding antenatal care and the birthing process.
In recent years, more women are asking for minimally invasive births that are physically, emotionally, and socially supported. As hospital-based obstetric care providers, we do not always respect or meet these expectations. We can and should do better.
Women should not have to choose between a good birth experience and medical safety, between social support and hospital resources, between a sense of autonomy and access to life-saving interventions. Although every effort should be taken to make the birthing experience a positive one for the mother and her family as a whole, it should not be done at the expense of safety. I have yet to hear an asphyxiated and brain-damaged child thank his mother’s obstetric care provider for allowing a wonderful birth experience.
What this evidence means for practice
Even in countries where home births are integrated fully into the medical care system and attended by trained and certified nurse-midwives, they are associated with increased risks, including a twofold to threefold increase in the odds of neonatal death.4 In the US, where no such integration exists, home births are dangerous.
Maternity care has come a long way since the 17th Century, when a woman had a 1 in 6 chance of dying in childbirth and only one of every five children lived to enjoy a first birthday. It is appropriate in this era of Obamacare and cost containment that we explore alternative methods. The option of a safe home delivery may well be part of the solution, as it is for many European countries--but until we can be assured that such an approach is safe for both mothers and infants, let's keep home delivery where it belongs...for pizza!
--Errol R. Norwitz, MD, PHD
We want to hear from you! Tell us what you think.
Every morning before I leave for work, I kiss my three children goodbye and tell them, “I love you. Make good choices today.”
This has become my mantra—so much so that, on her way out the door to join her friends at the movies recently, my daughter turned to me and said, “I know, Dad. I know. I’ll make good decisions tonight.”
And what decision is more important than where to deliver your child and who to have in attendance at the birth?
It is said that the passage from the uterus to the outside world that each one of us was forced to negotiate at birth is the most treacherous journey we will ever undertake. Any unnecessary delay or complication can have profound, lifelong consequences.
There is no question that the past few centuries have seen a significant “medicalization” of childbirth, including the relocation of deliveries from the community to a hospital setting, the introduction of male obstetricians, the unfortunate marginalization of midwives and support personnel (doulas), the development of uterotonic drugs, and the evolution of operative vaginal (forceps, vacuum) and cesarean deliveries.
Many of the practices initially introduced by obstetric care providers (including multiple vaginal examinations in labor, induction of labor for a large baby, and active management of labor protocols) have since been shown to be unhelpful in improving pregnancy outcomes, and some practices (such as episiotomy) have even been shown to be harmful.
Related article: Difficult fetal extraction at cesarean delivery: What should you do? Robert L. Barbieri, MD (Editorial, January 2012)
In the midst of this confusion, the one voice that has been lost is that of the patient herself.
Whose birth is it anyway?
The American College of Obstetricians and Gynecologists (ACOG), the American Academy of Pediatrics (AAP), and the American College of Nurse-Midwives (ACNM) all agree that patient autonomy is paramount, and that the final decision of where to deliver and who to have in attendance should be made by the patient herself, ideally in conjunction with her family and her obstetric care provider.1–3 But an informed decision is only as good as the available data. Regrettably, the literature on how planned home birth compares with hospital delivery in terms of pregnancy outcomes in the United States are sparse.
Related article: Lay midwives the the ObGyn: Is collaboration risky? Lucia DiVenere, MA (May 2012)
How safe is home birth in the United States?
Cheng and colleagues attempt to answer this question by reviewing newborn and maternal outcomes among planned home births versus hospital deliveries in a contemporary low-risk birth cohort. Their retrospective study included low-risk women at term with a singleton vertex live birth in 2008 in 27 of the 50 states using information from the Vital Statistics Natality Data provided by the Centers for Disease Control and Prevention.
Of these 2,081,753 women, 0.58% (n = 12,039) had planned home births, and the remainder delivered in a hospital setting. Women who had an “accidental” (unintended) home birth or who delivered in a freestanding birthing center were excluded. The primary outcome was the risk of a 5-minute Apgar score less than 4. Secondary outcomes included the risk of a 5-minute Apgar score less than 7, assisted ventilation for more than 6 hours, neonatal seizures, admission to the NICU, and a series of maternal outcome measures.
Besides the outcomes listed previously (top of page 24), women with a planned home birth had fewer obstetric interventions, including operative vaginal delivery and labor induction or augmentation. They also were less likely to be given antibiotics during labor (although the authors did not distinguish between antibiotics administered for prophylaxis against group B strep or surgical-site infection versus antibiotics to treat infections such as urinary tract infections or chorioamnionitis).
Of special interest is the fact that neither a prior vaginal delivery (multiparity) nor the absence of a prior cesarean delivery was protective against these adverse events.
The women at highest risk of an adverse event were those who delivered at home under the supervision of “other midwives.” Although these providers were not well defined, this term typically refers to community-based lay midwives whose only “training” consists of an unofficial apprenticeship of variable length. Despite the absence of formal training, the lack of certification and standardization of care, and the existence of legislation in many states banning their activity, such lay midwives continue to encourage and support home birth for both low- and high-risk women in the United States.
Related article: Update on Obstetrics John T. Repke, MD, and Jaimey M. Pauli, MD (January 2012)
Limitations of the study design
Although this dataset contains more than 2 million births, it includes only low-risk women at term and, therefore, is underpowered to measure outcomes such as fetal or neonatal death or birth injuries.
No data were presented on a number of important variables and outcome measures, such as the rate of or indications for cesarean delivery, the mode and frequency of intrapartum fetal monitoring, birth weight, intrapartum complications (uterine rupture, postpartum hemorrhage), blood transfusions, and infectious morbidity. The study also lacks long-term follow-up data on the infants.
That said, the study was well designed and very well written, and many of the limitations listed above are inherent in all retrospective cohort studies.
Putting these findings in context
These data are not novel, but they are remarkably consistent with other publications that have explored pregnancy outcomes in planned home birth versus hospital delivery from the Netherlands, the United Kingdom, Australia, and the United States, all of which show a higher rate of neonatal complications with planned home birth [see Reference 4 for review].4
Moreover, it is likely that the data in the current report significantly underestimate the risks of planned home birth for two reasons:
- Attempted home births that ended in transfer and, ultimately, delivery in a hospital setting (presumably for some unforeseen event such as excessive hemorrhage or uterine rupture or cord prolapse or nonreassuring fetal testing) were classified as hospital births.
- Apgar scores at 5 minutes are assigned by the attending care provider, and there is no way to independently verify their accuracy. Because of their limited training and/or concern about efforts to limit the scope of their practice, “other midwives” may be inclined to assign more favorable Apgar scores.
Who is choosing to deliver at home?
The proportion of US women who delivered outside the hospital setting increased by 29% between 2004 and 2009,5 although home births still constitute a minority of low-risk births (0.58% in the current study).
Related Article: Why are well-educated women more likely to choose home birth? Errol R. Norwitz, MD, PhD (Audiocast, November 2013)
One of the more interesting questions raised by this publication is the issue of who is choosing to deliver at home. In this cohort, women who planned home birth were more likely to be older, married, multiparous, white, and well educated. These aren’t exactly the women you would expect to gamble with the lives of their unborn offspring. So why are they choosing to deliver at home?
It could be that they are not well informed about the risks. Alternatively, they may have concluded that, although the relative risk of an adverse event is significantly higher with home birth, the absolute risk is low and acceptable to them. Or it could be that they are frustrated by the lack of autonomy afforded to them in the decisions surrounding antenatal care and the birthing process.
In recent years, more women are asking for minimally invasive births that are physically, emotionally, and socially supported. As hospital-based obstetric care providers, we do not always respect or meet these expectations. We can and should do better.
Women should not have to choose between a good birth experience and medical safety, between social support and hospital resources, between a sense of autonomy and access to life-saving interventions. Although every effort should be taken to make the birthing experience a positive one for the mother and her family as a whole, it should not be done at the expense of safety. I have yet to hear an asphyxiated and brain-damaged child thank his mother’s obstetric care provider for allowing a wonderful birth experience.
What this evidence means for practice
Even in countries where home births are integrated fully into the medical care system and attended by trained and certified nurse-midwives, they are associated with increased risks, including a twofold to threefold increase in the odds of neonatal death.4 In the US, where no such integration exists, home births are dangerous.
Maternity care has come a long way since the 17th Century, when a woman had a 1 in 6 chance of dying in childbirth and only one of every five children lived to enjoy a first birthday. It is appropriate in this era of Obamacare and cost containment that we explore alternative methods. The option of a safe home delivery may well be part of the solution, as it is for many European countries--but until we can be assured that such an approach is safe for both mothers and infants, let's keep home delivery where it belongs...for pizza!
--Errol R. Norwitz, MD, PHD
We want to hear from you! Tell us what you think.
- Committee on Obstetric Practice; American College of Obstetricians and Gynecologists. Committee Opinion #476: Planned home birth. Obstet Gynecol. 2011;117(2 Pt 1):425–428.
- American Academy of Pediatrics. Committee on Fetus and Newborn. Planned home birth. Pediatrics. 2013;131(5):1016–1020.
- American College of Nurse-Midwives. Division of Standards and Practice. Position statement: Home birth. Approved by the ACNM Board of Directors, May 2011. http://midwife.org/ACNM/files/ACNMLibraryData/UPLOADFILENAME/000000000251/Home%20Birth%20Aug%202011.pdf. Accessed October 21, 2013
- Wax JR, Lucas FL, Lamont M, et al. Maternal and newborn outcomes in planned home birth vs planned hospital births: A meta-analysis. Am J Obstet Gynecol. 2012;203(3):243.e1–e8.
- Martin JA, Hamilton BE, Ventura SJ, et al; Division of Vital Statistics. Births: Final data for 2009. Natl Vital Stat Rep. 2011;60(1):1–70.
- Committee on Obstetric Practice; American College of Obstetricians and Gynecologists. Committee Opinion #476: Planned home birth. Obstet Gynecol. 2011;117(2 Pt 1):425–428.
- American Academy of Pediatrics. Committee on Fetus and Newborn. Planned home birth. Pediatrics. 2013;131(5):1016–1020.
- American College of Nurse-Midwives. Division of Standards and Practice. Position statement: Home birth. Approved by the ACNM Board of Directors, May 2011. http://midwife.org/ACNM/files/ACNMLibraryData/UPLOADFILENAME/000000000251/Home%20Birth%20Aug%202011.pdf. Accessed October 21, 2013
- Wax JR, Lucas FL, Lamont M, et al. Maternal and newborn outcomes in planned home birth vs planned hospital births: A meta-analysis. Am J Obstet Gynecol. 2012;203(3):243.e1–e8.
- Martin JA, Hamilton BE, Ventura SJ, et al; Division of Vital Statistics. Births: Final data for 2009. Natl Vital Stat Rep. 2011;60(1):1–70.

Errol R. Norwitz, MD, PhD (November 2013)
Studies link maternal smoking with broad range of infant infections
ORLANDO – Maternal smoking has been associated with a number of serious respiratory and nonrespiratory infectious outcomes in infants, according to findings from two large case-control analyses.
Among 47,404 infants hospitalized for an infectious disease within 1 year of birth and 48,233 controls, and among 627 infants who died within 1 year of birth and 2,730 controls, maternal smoking was associated with both hospitalization because of to any infectious disease and mortality from any infectious disease (adjusted odds ratios, 1.52 and 1.51, respectively), Dr. Susanne Tanski of Dartmouth-Hitchcock Medical Center, Lebanon, N.H., reported at the annual meeting of the American Academy of Pediatrics.
Maternal smoking was associated with hospitalization due to a broad range of infectious diseases, including both respiratory and nonrespiratory diseases (adjusted OR, 1.69 and 1.27, respectively), said Dr. Tanski, who reported the findings of the subgroup analyses on behalf of lead study author Michael Metzger, Ph.D., and coauthor Dr. Abigail Halperin, both of the University of Washington, Seattle.
Stratification by birth weight and gestational age did not appreciably affect the findings with respect to morbidity risk in the infants of smoking mothers, which was found to be independent of both birth weight and gestational age. Mortality risk was found to be independent of gestational age, but the association among low birth weight infants was attenuated.
Even so, if an infant whose mother smoked was of normal weight and gestational age, exposure to maternal smoking appeared to carry an increased risk of infection as well as infectious disease mortality, Dr. Halperin said in an interview.
Included in the study were infants born in Washington from 1987 to 2004. The investigators reviewed linked birth certificate, death certificate, and hospital discharge records.
"We’ve known for a long time that babies born to mothers who smoke during pregnancy are at high risk for serious medical problems relating to low birth weight, premature delivery, and poor lung development, and while we have recognized respiratory infections as a common cause of these sometimes life-threatening infections, this study demonstrates that in utero exposure to smoke increases the risk of hospitalization and death from a much broader range of infections than previously documented," Dr. Halperin said.
She noted that the study was published earlier this year (Pediatr. Infect. Dis. 2013;32:e1-7 [doi:10.1097/INF.0b013e3182704bb5]) but received little notice. When the AAP invited her to submit an abstract for presentation at the conference, she said she was pleased to find that the paper was "selected for this honor."
"I’m very happy it’s receiving some attention now, because it is important to get this information out there so we can try to do something about it. We found that there was a dose-response effect related to number of cigarettes smoked per day, whereby smoking fewer cigarettes significantly lowered the risk for hospitalization," she said.
Thus, counseling pregnant women to reduce their smoking, even if they are not able to quit completely, may help reduce the impact of maternal smoking on infant outcomes, she said.
"Clinicians can help their patients stop smoking by providing nonjudgmental, positively framed advice to quit, and should strongly encourage pregnant women to quit smoking, both for their own health and for the health of their babies," she said, noting that women also should be counseled about the importance of protecting their children from secondhand smoke after birth, as secondhand smoke exposure increases the risk for many of the same infections seen in babies exposed in utero.
Evidence-based treatment, including counseling and approved cessation medication, should be offered to all patients who want to quit, regardless of whether or not they are pregnant, she added.
Dr. Tanski reported a having a patent pending and receiving royalties for a smoke sensor. Dr. Halperin and Mr. Metzger reported having no disclosures.
ORLANDO – Maternal smoking has been associated with a number of serious respiratory and nonrespiratory infectious outcomes in infants, according to findings from two large case-control analyses.
Among 47,404 infants hospitalized for an infectious disease within 1 year of birth and 48,233 controls, and among 627 infants who died within 1 year of birth and 2,730 controls, maternal smoking was associated with both hospitalization because of to any infectious disease and mortality from any infectious disease (adjusted odds ratios, 1.52 and 1.51, respectively), Dr. Susanne Tanski of Dartmouth-Hitchcock Medical Center, Lebanon, N.H., reported at the annual meeting of the American Academy of Pediatrics.
Maternal smoking was associated with hospitalization due to a broad range of infectious diseases, including both respiratory and nonrespiratory diseases (adjusted OR, 1.69 and 1.27, respectively), said Dr. Tanski, who reported the findings of the subgroup analyses on behalf of lead study author Michael Metzger, Ph.D., and coauthor Dr. Abigail Halperin, both of the University of Washington, Seattle.
Stratification by birth weight and gestational age did not appreciably affect the findings with respect to morbidity risk in the infants of smoking mothers, which was found to be independent of both birth weight and gestational age. Mortality risk was found to be independent of gestational age, but the association among low birth weight infants was attenuated.
Even so, if an infant whose mother smoked was of normal weight and gestational age, exposure to maternal smoking appeared to carry an increased risk of infection as well as infectious disease mortality, Dr. Halperin said in an interview.
Included in the study were infants born in Washington from 1987 to 2004. The investigators reviewed linked birth certificate, death certificate, and hospital discharge records.
"We’ve known for a long time that babies born to mothers who smoke during pregnancy are at high risk for serious medical problems relating to low birth weight, premature delivery, and poor lung development, and while we have recognized respiratory infections as a common cause of these sometimes life-threatening infections, this study demonstrates that in utero exposure to smoke increases the risk of hospitalization and death from a much broader range of infections than previously documented," Dr. Halperin said.
She noted that the study was published earlier this year (Pediatr. Infect. Dis. 2013;32:e1-7 [doi:10.1097/INF.0b013e3182704bb5]) but received little notice. When the AAP invited her to submit an abstract for presentation at the conference, she said she was pleased to find that the paper was "selected for this honor."
"I’m very happy it’s receiving some attention now, because it is important to get this information out there so we can try to do something about it. We found that there was a dose-response effect related to number of cigarettes smoked per day, whereby smoking fewer cigarettes significantly lowered the risk for hospitalization," she said.
Thus, counseling pregnant women to reduce their smoking, even if they are not able to quit completely, may help reduce the impact of maternal smoking on infant outcomes, she said.
"Clinicians can help their patients stop smoking by providing nonjudgmental, positively framed advice to quit, and should strongly encourage pregnant women to quit smoking, both for their own health and for the health of their babies," she said, noting that women also should be counseled about the importance of protecting their children from secondhand smoke after birth, as secondhand smoke exposure increases the risk for many of the same infections seen in babies exposed in utero.
Evidence-based treatment, including counseling and approved cessation medication, should be offered to all patients who want to quit, regardless of whether or not they are pregnant, she added.
Dr. Tanski reported a having a patent pending and receiving royalties for a smoke sensor. Dr. Halperin and Mr. Metzger reported having no disclosures.
ORLANDO – Maternal smoking has been associated with a number of serious respiratory and nonrespiratory infectious outcomes in infants, according to findings from two large case-control analyses.
Among 47,404 infants hospitalized for an infectious disease within 1 year of birth and 48,233 controls, and among 627 infants who died within 1 year of birth and 2,730 controls, maternal smoking was associated with both hospitalization because of to any infectious disease and mortality from any infectious disease (adjusted odds ratios, 1.52 and 1.51, respectively), Dr. Susanne Tanski of Dartmouth-Hitchcock Medical Center, Lebanon, N.H., reported at the annual meeting of the American Academy of Pediatrics.
Maternal smoking was associated with hospitalization due to a broad range of infectious diseases, including both respiratory and nonrespiratory diseases (adjusted OR, 1.69 and 1.27, respectively), said Dr. Tanski, who reported the findings of the subgroup analyses on behalf of lead study author Michael Metzger, Ph.D., and coauthor Dr. Abigail Halperin, both of the University of Washington, Seattle.
Stratification by birth weight and gestational age did not appreciably affect the findings with respect to morbidity risk in the infants of smoking mothers, which was found to be independent of both birth weight and gestational age. Mortality risk was found to be independent of gestational age, but the association among low birth weight infants was attenuated.
Even so, if an infant whose mother smoked was of normal weight and gestational age, exposure to maternal smoking appeared to carry an increased risk of infection as well as infectious disease mortality, Dr. Halperin said in an interview.
Included in the study were infants born in Washington from 1987 to 2004. The investigators reviewed linked birth certificate, death certificate, and hospital discharge records.
"We’ve known for a long time that babies born to mothers who smoke during pregnancy are at high risk for serious medical problems relating to low birth weight, premature delivery, and poor lung development, and while we have recognized respiratory infections as a common cause of these sometimes life-threatening infections, this study demonstrates that in utero exposure to smoke increases the risk of hospitalization and death from a much broader range of infections than previously documented," Dr. Halperin said.
She noted that the study was published earlier this year (Pediatr. Infect. Dis. 2013;32:e1-7 [doi:10.1097/INF.0b013e3182704bb5]) but received little notice. When the AAP invited her to submit an abstract for presentation at the conference, she said she was pleased to find that the paper was "selected for this honor."
"I’m very happy it’s receiving some attention now, because it is important to get this information out there so we can try to do something about it. We found that there was a dose-response effect related to number of cigarettes smoked per day, whereby smoking fewer cigarettes significantly lowered the risk for hospitalization," she said.
Thus, counseling pregnant women to reduce their smoking, even if they are not able to quit completely, may help reduce the impact of maternal smoking on infant outcomes, she said.
"Clinicians can help their patients stop smoking by providing nonjudgmental, positively framed advice to quit, and should strongly encourage pregnant women to quit smoking, both for their own health and for the health of their babies," she said, noting that women also should be counseled about the importance of protecting their children from secondhand smoke after birth, as secondhand smoke exposure increases the risk for many of the same infections seen in babies exposed in utero.
Evidence-based treatment, including counseling and approved cessation medication, should be offered to all patients who want to quit, regardless of whether or not they are pregnant, she added.
Dr. Tanski reported a having a patent pending and receiving royalties for a smoke sensor. Dr. Halperin and Mr. Metzger reported having no disclosures.
AT THE AAP NATIONAL CONFERENCE
Major finding: Maternal smoking was associated with both hospitalization due to any infectious disease and mortality due to any infectious disease (adjusted odds ratios, 1.52 and 1.51, respectively).
Data source: Two case-control analyses – one involving 47,404 infants hospitalized for an infectious disease within 1 year of birth and 48,233 controls and the other involving 627 infants who died within 1 year of birth and 2,730 controls.
Disclosures: Dr. Tanski reported having a patent pending and receiving royalties for a smoke sensor. Dr. Halperin and Dr. Metzger reported having no disclosures.
Hormone Therapy’s Protection Against Endometrial Cancer
Earn 0.25 hours AMA PRA Category 1 credit: Read this article, and click the link at the end to take the post-test.
AMSTERDAM – The continuous use of estrogen and progestin protects against the development of endometrial cancer in postmenopausal women, according to extended follow-up findings from the seminal Women’s Health Initiative randomized, placebo-controlled trial.
After a median of 13.2 years’ follow-up, there were 35% fewer endometrial cancers among women given combined estrogen and progestin vs. placebo (hazard ratio, 0.65; P = .007). A total of 66 women treated with the hormone therapy (HT) and 95 given placebo had developed endometrial cancer, yielding annual incidences of 0.06% and 0.10%, respectively.
"We do not feel that this effect on endometrial cancer changes the overall balance of risk and benefit of estrogen plus progestin," Dr. Rowan T. Chlebowski stated at the multidisciplinary European cancer congresses.
Dr. Chlebowski of the Los Angeles Biomedical Research Institute at Harbor-UCLA Medical Center in Torrance, added that the original conclusion of the WHI study remains: "Estrogen plus progestin should not be used for chronic disease risk reduction."
Between 1993 and 1998, 16,608 women aged 50-79 years with intact uteri were enrolled into the Women’s Health Initiative randomized, controlled clinical trial of hormone therapy at 40 clinical centers in the United States. The study was halted early as it found an excess of cardiovascular diseases and breast cancer among women given the HT vs. placebo. However, there was a 17% decrease in the risk for endometrial cancer in women given HT, as well as reductions in colorectal cancer and hip fracture (JAMA 2002;288:321-33).
Women who had participated in the study were recontacted to obtain their consent to look at the impact of the HT on their risk of endometrial cancer, with 12,788 (83%) surviving women giving their consent. Of these women, 6,545 had received continuous oral treatment with conjugated equine estrogen (0.625 mg/day) and medroxyprogesterone acetate (2.5 mg/day), and the remaining 6,243 had received placebo.
The findings now reported represented a median of 5.6 years of treatment data and a further 8.2 years of additional follow-up. Analysis revealed that the major difference emerged after the treatment was stopped, with 41 vs. 65 cases reported cases postintervention (HR, 0.59; P = .008).
Dr. Chlebowski reported that in the endometrial cancers that did occur, there were fewer poorly differentiated or anaplastic tumors (HR, 0.51) and fewer cases of regional or distant disease (HR, 0.43) in the HT than placebo group.
Subgroup analyses found that there was a similar effect of the estrogen plus progestin influence on endometrial cancer risk generally, even when body mass index was taken into account. BMI is a known risk factor for endometrial cancer, with risk increasing with increasing body weight.
There were 5 deaths from endometrial cancer in the HT group and 11 in the placebo group, but this difference was not statistically significant (HR, 0.42).
Dr. Marcia Hall, a consultant medical oncologist at the Mount Vernon Cancer Centre in greater London, provided independent comment on the results. "The combination of estrogen plus progestin does indeed protect against endometrial cancer," she said, noting that the results are in line with those of observational studies, such as the U.K.’s Million Women Study (Lancet 2007;369:1703-10).
There are women who may still benefit greatly from HT, Dr. Hall maintained. This includes premenopausal women who have had a hysterectomies, oophorectomies, or chemical castrations for other conditions; women experiencing menopausal symptoms, and those who may be at higher risk for bone diseases, such as osteoporosis and enter the menopause early. "Continuous estrogen, perhaps at the lowest dose possible, is a moderately safe drug in these conditions and situations," Dr. Hall said. She suggested that a low estrogen dose could perhaps be combined with a different progestin and mode of administration, such as levonorgestrel delivered by the intrauterine system (Mirena).
The latest WHI findings "should provoke further exploration of the role of progestins in the prevention of endometrial and possibly colorectal cancers," Dr. Hall concluded. "I hope it may also allow us to rethink the management of endometrial cancer in a population with high-risk comorbidities."
The U.S. National Institutes of Health sponsored the WHI. Dr. Chlebowski has acted as a consultant to Pfizer, Novartis, and Amgen, and has received honorarium from Novartis. Dr. Hall had no relevant conflicts of interest.
To earn 0.25 hours AMA PRA Category 1 credit after reading this article, take the post-test here.
Earn 0.25 hours AMA PRA Category 1 credit: Read this article, and click the link at the end to take the post-test.
AMSTERDAM – The continuous use of estrogen and progestin protects against the development of endometrial cancer in postmenopausal women, according to extended follow-up findings from the seminal Women’s Health Initiative randomized, placebo-controlled trial.
After a median of 13.2 years’ follow-up, there were 35% fewer endometrial cancers among women given combined estrogen and progestin vs. placebo (hazard ratio, 0.65; P = .007). A total of 66 women treated with the hormone therapy (HT) and 95 given placebo had developed endometrial cancer, yielding annual incidences of 0.06% and 0.10%, respectively.
"We do not feel that this effect on endometrial cancer changes the overall balance of risk and benefit of estrogen plus progestin," Dr. Rowan T. Chlebowski stated at the multidisciplinary European cancer congresses.
Dr. Chlebowski of the Los Angeles Biomedical Research Institute at Harbor-UCLA Medical Center in Torrance, added that the original conclusion of the WHI study remains: "Estrogen plus progestin should not be used for chronic disease risk reduction."
Between 1993 and 1998, 16,608 women aged 50-79 years with intact uteri were enrolled into the Women’s Health Initiative randomized, controlled clinical trial of hormone therapy at 40 clinical centers in the United States. The study was halted early as it found an excess of cardiovascular diseases and breast cancer among women given the HT vs. placebo. However, there was a 17% decrease in the risk for endometrial cancer in women given HT, as well as reductions in colorectal cancer and hip fracture (JAMA 2002;288:321-33).
Women who had participated in the study were recontacted to obtain their consent to look at the impact of the HT on their risk of endometrial cancer, with 12,788 (83%) surviving women giving their consent. Of these women, 6,545 had received continuous oral treatment with conjugated equine estrogen (0.625 mg/day) and medroxyprogesterone acetate (2.5 mg/day), and the remaining 6,243 had received placebo.
The findings now reported represented a median of 5.6 years of treatment data and a further 8.2 years of additional follow-up. Analysis revealed that the major difference emerged after the treatment was stopped, with 41 vs. 65 cases reported cases postintervention (HR, 0.59; P = .008).
Dr. Chlebowski reported that in the endometrial cancers that did occur, there were fewer poorly differentiated or anaplastic tumors (HR, 0.51) and fewer cases of regional or distant disease (HR, 0.43) in the HT than placebo group.
Subgroup analyses found that there was a similar effect of the estrogen plus progestin influence on endometrial cancer risk generally, even when body mass index was taken into account. BMI is a known risk factor for endometrial cancer, with risk increasing with increasing body weight.
There were 5 deaths from endometrial cancer in the HT group and 11 in the placebo group, but this difference was not statistically significant (HR, 0.42).
Dr. Marcia Hall, a consultant medical oncologist at the Mount Vernon Cancer Centre in greater London, provided independent comment on the results. "The combination of estrogen plus progestin does indeed protect against endometrial cancer," she said, noting that the results are in line with those of observational studies, such as the U.K.’s Million Women Study (Lancet 2007;369:1703-10).
There are women who may still benefit greatly from HT, Dr. Hall maintained. This includes premenopausal women who have had a hysterectomies, oophorectomies, or chemical castrations for other conditions; women experiencing menopausal symptoms, and those who may be at higher risk for bone diseases, such as osteoporosis and enter the menopause early. "Continuous estrogen, perhaps at the lowest dose possible, is a moderately safe drug in these conditions and situations," Dr. Hall said. She suggested that a low estrogen dose could perhaps be combined with a different progestin and mode of administration, such as levonorgestrel delivered by the intrauterine system (Mirena).
The latest WHI findings "should provoke further exploration of the role of progestins in the prevention of endometrial and possibly colorectal cancers," Dr. Hall concluded. "I hope it may also allow us to rethink the management of endometrial cancer in a population with high-risk comorbidities."
The U.S. National Institutes of Health sponsored the WHI. Dr. Chlebowski has acted as a consultant to Pfizer, Novartis, and Amgen, and has received honorarium from Novartis. Dr. Hall had no relevant conflicts of interest.
To earn 0.25 hours AMA PRA Category 1 credit after reading this article, take the post-test here.
Earn 0.25 hours AMA PRA Category 1 credit: Read this article, and click the link at the end to take the post-test.
AMSTERDAM – The continuous use of estrogen and progestin protects against the development of endometrial cancer in postmenopausal women, according to extended follow-up findings from the seminal Women’s Health Initiative randomized, placebo-controlled trial.
After a median of 13.2 years’ follow-up, there were 35% fewer endometrial cancers among women given combined estrogen and progestin vs. placebo (hazard ratio, 0.65; P = .007). A total of 66 women treated with the hormone therapy (HT) and 95 given placebo had developed endometrial cancer, yielding annual incidences of 0.06% and 0.10%, respectively.
"We do not feel that this effect on endometrial cancer changes the overall balance of risk and benefit of estrogen plus progestin," Dr. Rowan T. Chlebowski stated at the multidisciplinary European cancer congresses.
Dr. Chlebowski of the Los Angeles Biomedical Research Institute at Harbor-UCLA Medical Center in Torrance, added that the original conclusion of the WHI study remains: "Estrogen plus progestin should not be used for chronic disease risk reduction."
Between 1993 and 1998, 16,608 women aged 50-79 years with intact uteri were enrolled into the Women’s Health Initiative randomized, controlled clinical trial of hormone therapy at 40 clinical centers in the United States. The study was halted early as it found an excess of cardiovascular diseases and breast cancer among women given the HT vs. placebo. However, there was a 17% decrease in the risk for endometrial cancer in women given HT, as well as reductions in colorectal cancer and hip fracture (JAMA 2002;288:321-33).
Women who had participated in the study were recontacted to obtain their consent to look at the impact of the HT on their risk of endometrial cancer, with 12,788 (83%) surviving women giving their consent. Of these women, 6,545 had received continuous oral treatment with conjugated equine estrogen (0.625 mg/day) and medroxyprogesterone acetate (2.5 mg/day), and the remaining 6,243 had received placebo.
The findings now reported represented a median of 5.6 years of treatment data and a further 8.2 years of additional follow-up. Analysis revealed that the major difference emerged after the treatment was stopped, with 41 vs. 65 cases reported cases postintervention (HR, 0.59; P = .008).
Dr. Chlebowski reported that in the endometrial cancers that did occur, there were fewer poorly differentiated or anaplastic tumors (HR, 0.51) and fewer cases of regional or distant disease (HR, 0.43) in the HT than placebo group.
Subgroup analyses found that there was a similar effect of the estrogen plus progestin influence on endometrial cancer risk generally, even when body mass index was taken into account. BMI is a known risk factor for endometrial cancer, with risk increasing with increasing body weight.
There were 5 deaths from endometrial cancer in the HT group and 11 in the placebo group, but this difference was not statistically significant (HR, 0.42).
Dr. Marcia Hall, a consultant medical oncologist at the Mount Vernon Cancer Centre in greater London, provided independent comment on the results. "The combination of estrogen plus progestin does indeed protect against endometrial cancer," she said, noting that the results are in line with those of observational studies, such as the U.K.’s Million Women Study (Lancet 2007;369:1703-10).
There are women who may still benefit greatly from HT, Dr. Hall maintained. This includes premenopausal women who have had a hysterectomies, oophorectomies, or chemical castrations for other conditions; women experiencing menopausal symptoms, and those who may be at higher risk for bone diseases, such as osteoporosis and enter the menopause early. "Continuous estrogen, perhaps at the lowest dose possible, is a moderately safe drug in these conditions and situations," Dr. Hall said. She suggested that a low estrogen dose could perhaps be combined with a different progestin and mode of administration, such as levonorgestrel delivered by the intrauterine system (Mirena).
The latest WHI findings "should provoke further exploration of the role of progestins in the prevention of endometrial and possibly colorectal cancers," Dr. Hall concluded. "I hope it may also allow us to rethink the management of endometrial cancer in a population with high-risk comorbidities."
The U.S. National Institutes of Health sponsored the WHI. Dr. Chlebowski has acted as a consultant to Pfizer, Novartis, and Amgen, and has received honorarium from Novartis. Dr. Hall had no relevant conflicts of interest.
To earn 0.25 hours AMA PRA Category 1 credit after reading this article, take the post-test here.
AT THE EUROPEAN CANCER CONGRESS 2013