User login
TKA outcomes for age 80+ similar to younger patients
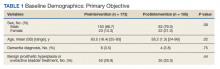
CHICAGO - Patients 80 years or older undergoing primary total knee arthroplasty (TKA) have similar odds of complications, compared with 65- to 79-year-old patients, an analysis of more than 1.7 million cases suggests.
Priscilla Varghese, MBA, MS, and an MD candidate at State University of New York, Brooklyn, led the research, presented at the American Academy of Orthopaedic Surgeons 2022 annual meeting.
Ms. Varghese’s team queried a Medicare claims database for the years 2005-2014 and analyzed information from 295,908 octogenarians and 1.4 million control patients aged 65-79 who received TKA.
Study group patients were randomly matched to controls in a 1:5 ratio according to gender and comorbidities, including chronic obstructive pulmonary disease, congestive heart failure, diabetes, peripheral vascular disease, and kidney failure.
Octogenarians were found to have higher incidence and odds of 90-day readmission rates (10.59% vs. 9.35%; odds ratio, 1.15; 95% confidence interval, 1.13-1.16; P < .0001).
Hospital stays were also longer (3.69 days ± 1.95 vs. 3.23 days ± 1.83; P < .0001), compared with controls.
Reassuring older patients
However, Ms. Varghese told this news organization she was surprised to find that the older group had equal incidence and odds of developing medical complications (1.26% vs. 1.26%; OR, 0.99; 95% CI, 0.96-1.03; P =.99).
“That’s a really important piece of information to have when we are advising 80-year-olds – to be able to say their risk of adverse outcomes is similar to someone who’s 10 years, 15 years younger,” she said. “It’s really reassuring.”
These results offer good news to older patients who might be hesitant to undergo the surgery, and good news in general as life expectancy increases and people stay active long into their later years, forecasting the need for more knee replacements.
The number of total knee replacements is expected to rise dramatically in the United States.
In a 2017 study published in Osteoarthritis Cartilage, the authors write, “the number of TKAs in the U.S., which already has the highest [incidence rate] of knee arthroplasty in the world, is expected to increase 143% by 2050.”
Thomas Fleeter, MD, an orthopedic surgeon practicing in Reston, Virginia, who was not involved in the study, told this news organization this study reinforces that “it’s OK to do knee replacements in elderly people; you just have to pick the right ones.”
He pointed out that the study also showed that the 80-and-older patients don’t have the added risk of loosening their mechanical components after the surgery, likely because they are less inclined than their younger counterparts to follow surgery with strenuous activities.
In a subanalysis, revision rates were also lower for the octogenarians (0.01% vs. 0.02% for controls).
Octogenarians who had TKA were found to have lower incidence and odds (1.6% vs. 1.93%; OR, 0.86; 95% CI, 0.83-0.88, P < .001) of implant-related complications, compared with the younger group.
The increased length of stay would be expected, Dr. Fleeter said, because those 80-plus may need a bit more help getting out of bed and may not have as much support at home.
A total knee replacement can have the substantial benefit of improving octogenarians’ ability to maintain their independence longer by facilitating driving or walking.
“It’s a small and manageable risk if you pick the right patients,” he said.
Demand for TKAs rises as population ages
As patients are living longer and wanting to maintain their mobility and as obesity rates are rising, more older patients will seek total knee replacements, especially since the payoff is high, Ms. Varghese noted.
“People who undergo this operation tend to show remarkable decreases in pain and increases in range of motion,” she said.
This study has the advantage of a more personalized look at risks of TKA because it stratifies age groups.
“The literature tends to look at the elderly population as one big cohort – 65 and older,” Ms. Varghese said. “We were able to provide patients more specific data.”
Ms. Varghese and Dr. Fleeter have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
CHICAGO - Patients 80 years or older undergoing primary total knee arthroplasty (TKA) have similar odds of complications, compared with 65- to 79-year-old patients, an analysis of more than 1.7 million cases suggests.
Priscilla Varghese, MBA, MS, and an MD candidate at State University of New York, Brooklyn, led the research, presented at the American Academy of Orthopaedic Surgeons 2022 annual meeting.
Ms. Varghese’s team queried a Medicare claims database for the years 2005-2014 and analyzed information from 295,908 octogenarians and 1.4 million control patients aged 65-79 who received TKA.
Study group patients were randomly matched to controls in a 1:5 ratio according to gender and comorbidities, including chronic obstructive pulmonary disease, congestive heart failure, diabetes, peripheral vascular disease, and kidney failure.
Octogenarians were found to have higher incidence and odds of 90-day readmission rates (10.59% vs. 9.35%; odds ratio, 1.15; 95% confidence interval, 1.13-1.16; P < .0001).
Hospital stays were also longer (3.69 days ± 1.95 vs. 3.23 days ± 1.83; P < .0001), compared with controls.
Reassuring older patients
However, Ms. Varghese told this news organization she was surprised to find that the older group had equal incidence and odds of developing medical complications (1.26% vs. 1.26%; OR, 0.99; 95% CI, 0.96-1.03; P =.99).
“That’s a really important piece of information to have when we are advising 80-year-olds – to be able to say their risk of adverse outcomes is similar to someone who’s 10 years, 15 years younger,” she said. “It’s really reassuring.”
These results offer good news to older patients who might be hesitant to undergo the surgery, and good news in general as life expectancy increases and people stay active long into their later years, forecasting the need for more knee replacements.
The number of total knee replacements is expected to rise dramatically in the United States.
In a 2017 study published in Osteoarthritis Cartilage, the authors write, “the number of TKAs in the U.S., which already has the highest [incidence rate] of knee arthroplasty in the world, is expected to increase 143% by 2050.”
Thomas Fleeter, MD, an orthopedic surgeon practicing in Reston, Virginia, who was not involved in the study, told this news organization this study reinforces that “it’s OK to do knee replacements in elderly people; you just have to pick the right ones.”
He pointed out that the study also showed that the 80-and-older patients don’t have the added risk of loosening their mechanical components after the surgery, likely because they are less inclined than their younger counterparts to follow surgery with strenuous activities.
In a subanalysis, revision rates were also lower for the octogenarians (0.01% vs. 0.02% for controls).
Octogenarians who had TKA were found to have lower incidence and odds (1.6% vs. 1.93%; OR, 0.86; 95% CI, 0.83-0.88, P < .001) of implant-related complications, compared with the younger group.
The increased length of stay would be expected, Dr. Fleeter said, because those 80-plus may need a bit more help getting out of bed and may not have as much support at home.
A total knee replacement can have the substantial benefit of improving octogenarians’ ability to maintain their independence longer by facilitating driving or walking.
“It’s a small and manageable risk if you pick the right patients,” he said.
Demand for TKAs rises as population ages
As patients are living longer and wanting to maintain their mobility and as obesity rates are rising, more older patients will seek total knee replacements, especially since the payoff is high, Ms. Varghese noted.
“People who undergo this operation tend to show remarkable decreases in pain and increases in range of motion,” she said.
This study has the advantage of a more personalized look at risks of TKA because it stratifies age groups.
“The literature tends to look at the elderly population as one big cohort – 65 and older,” Ms. Varghese said. “We were able to provide patients more specific data.”
Ms. Varghese and Dr. Fleeter have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
CHICAGO - Patients 80 years or older undergoing primary total knee arthroplasty (TKA) have similar odds of complications, compared with 65- to 79-year-old patients, an analysis of more than 1.7 million cases suggests.
Priscilla Varghese, MBA, MS, and an MD candidate at State University of New York, Brooklyn, led the research, presented at the American Academy of Orthopaedic Surgeons 2022 annual meeting.
Ms. Varghese’s team queried a Medicare claims database for the years 2005-2014 and analyzed information from 295,908 octogenarians and 1.4 million control patients aged 65-79 who received TKA.
Study group patients were randomly matched to controls in a 1:5 ratio according to gender and comorbidities, including chronic obstructive pulmonary disease, congestive heart failure, diabetes, peripheral vascular disease, and kidney failure.
Octogenarians were found to have higher incidence and odds of 90-day readmission rates (10.59% vs. 9.35%; odds ratio, 1.15; 95% confidence interval, 1.13-1.16; P < .0001).
Hospital stays were also longer (3.69 days ± 1.95 vs. 3.23 days ± 1.83; P < .0001), compared with controls.
Reassuring older patients
However, Ms. Varghese told this news organization she was surprised to find that the older group had equal incidence and odds of developing medical complications (1.26% vs. 1.26%; OR, 0.99; 95% CI, 0.96-1.03; P =.99).
“That’s a really important piece of information to have when we are advising 80-year-olds – to be able to say their risk of adverse outcomes is similar to someone who’s 10 years, 15 years younger,” she said. “It’s really reassuring.”
These results offer good news to older patients who might be hesitant to undergo the surgery, and good news in general as life expectancy increases and people stay active long into their later years, forecasting the need for more knee replacements.
The number of total knee replacements is expected to rise dramatically in the United States.
In a 2017 study published in Osteoarthritis Cartilage, the authors write, “the number of TKAs in the U.S., which already has the highest [incidence rate] of knee arthroplasty in the world, is expected to increase 143% by 2050.”
Thomas Fleeter, MD, an orthopedic surgeon practicing in Reston, Virginia, who was not involved in the study, told this news organization this study reinforces that “it’s OK to do knee replacements in elderly people; you just have to pick the right ones.”
He pointed out that the study also showed that the 80-and-older patients don’t have the added risk of loosening their mechanical components after the surgery, likely because they are less inclined than their younger counterparts to follow surgery with strenuous activities.
In a subanalysis, revision rates were also lower for the octogenarians (0.01% vs. 0.02% for controls).
Octogenarians who had TKA were found to have lower incidence and odds (1.6% vs. 1.93%; OR, 0.86; 95% CI, 0.83-0.88, P < .001) of implant-related complications, compared with the younger group.
The increased length of stay would be expected, Dr. Fleeter said, because those 80-plus may need a bit more help getting out of bed and may not have as much support at home.
A total knee replacement can have the substantial benefit of improving octogenarians’ ability to maintain their independence longer by facilitating driving or walking.
“It’s a small and manageable risk if you pick the right patients,” he said.
Demand for TKAs rises as population ages
As patients are living longer and wanting to maintain their mobility and as obesity rates are rising, more older patients will seek total knee replacements, especially since the payoff is high, Ms. Varghese noted.
“People who undergo this operation tend to show remarkable decreases in pain and increases in range of motion,” she said.
This study has the advantage of a more personalized look at risks of TKA because it stratifies age groups.
“The literature tends to look at the elderly population as one big cohort – 65 and older,” Ms. Varghese said. “We were able to provide patients more specific data.”
Ms. Varghese and Dr. Fleeter have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Orphenadrine recalled due to possible nitrosamine impurity
Recent tests of 13 lots of the skeletal muscle relaxant Orphenadrine Citrate 100 mg Extended Release (ER) found unacceptably high levels of a nitrosamine impurity in the tablets, leading manufacturer Sandoz (Princeton, N.J.) to announce a voluntary recall of the lots on March 21.
The nitrosamine impurity detected (N-methyl-N-nitroso-2-[(2-methylphenyl)phenylmethoxy]ethanamine [NMOA or Nitroso-Orphenadrine]) may potentially be consumed at a level higher than the Food and Drug Administration’s Acceptable Daily Intake of 26.5 ng/day. Nitrosamines have carcinogenic potency when present above the allowable exposure limits, according to Sandoz, but the company said it “has not received any reports of adverse events related to the presence of a nitrosamine impurity in the lot.”
The Orphenadrine Citrate 100 mg ER Tablets were shipped to customers from August 2019 to April 2021 and have lot numbers of JX6411, JX6413, KC0723, KC3303, KE4348, KE7169, KE4349, KL3199, KM0072, KS3939, LA7704, LA7703, and LA9243.
The lots contain 100- and 1,000-count bottles of Orphenadrine Citrate ER Tablets, which are used as an adjunct to rest, physical therapy, and other measures for the relief of discomfort associated with acute painful musculoskeletal conditions.
The recall does not apply to any other strengths of Sandoz’s Orphenadrine Citrate ER Tablets or to other lot numbers of the product.
Sandoz advises that wholesalers and distributors should “immediately stop distribution of the recalled product and quarantine and return all recalled product in their inventory.” The company advises consumers to stop taking the recalled product and immediately consult with their physicians to obtain another prescription, notifying them of any problems that may be related to taking or using the tablets.
Sandoz says that retailers and consumers should contact Sedgwick directly by phone at 844-491-7869 or email at [email protected] to return the recalled product, and report adverse reactions to Sandoz by phone at (800) 525-8747 or by email at [email protected]. Adverse reactions and quality problems can be reported to the FDA’s MedWatch Adverse Event Reporting program either online, by regular mail, or by fax to 1-800-FDA-0178.
A version of this article first appeared on Medscape.com.
Recent tests of 13 lots of the skeletal muscle relaxant Orphenadrine Citrate 100 mg Extended Release (ER) found unacceptably high levels of a nitrosamine impurity in the tablets, leading manufacturer Sandoz (Princeton, N.J.) to announce a voluntary recall of the lots on March 21.
The nitrosamine impurity detected (N-methyl-N-nitroso-2-[(2-methylphenyl)phenylmethoxy]ethanamine [NMOA or Nitroso-Orphenadrine]) may potentially be consumed at a level higher than the Food and Drug Administration’s Acceptable Daily Intake of 26.5 ng/day. Nitrosamines have carcinogenic potency when present above the allowable exposure limits, according to Sandoz, but the company said it “has not received any reports of adverse events related to the presence of a nitrosamine impurity in the lot.”
The Orphenadrine Citrate 100 mg ER Tablets were shipped to customers from August 2019 to April 2021 and have lot numbers of JX6411, JX6413, KC0723, KC3303, KE4348, KE7169, KE4349, KL3199, KM0072, KS3939, LA7704, LA7703, and LA9243.
The lots contain 100- and 1,000-count bottles of Orphenadrine Citrate ER Tablets, which are used as an adjunct to rest, physical therapy, and other measures for the relief of discomfort associated with acute painful musculoskeletal conditions.
The recall does not apply to any other strengths of Sandoz’s Orphenadrine Citrate ER Tablets or to other lot numbers of the product.
Sandoz advises that wholesalers and distributors should “immediately stop distribution of the recalled product and quarantine and return all recalled product in their inventory.” The company advises consumers to stop taking the recalled product and immediately consult with their physicians to obtain another prescription, notifying them of any problems that may be related to taking or using the tablets.
Sandoz says that retailers and consumers should contact Sedgwick directly by phone at 844-491-7869 or email at [email protected] to return the recalled product, and report adverse reactions to Sandoz by phone at (800) 525-8747 or by email at [email protected]. Adverse reactions and quality problems can be reported to the FDA’s MedWatch Adverse Event Reporting program either online, by regular mail, or by fax to 1-800-FDA-0178.
A version of this article first appeared on Medscape.com.
Recent tests of 13 lots of the skeletal muscle relaxant Orphenadrine Citrate 100 mg Extended Release (ER) found unacceptably high levels of a nitrosamine impurity in the tablets, leading manufacturer Sandoz (Princeton, N.J.) to announce a voluntary recall of the lots on March 21.
The nitrosamine impurity detected (N-methyl-N-nitroso-2-[(2-methylphenyl)phenylmethoxy]ethanamine [NMOA or Nitroso-Orphenadrine]) may potentially be consumed at a level higher than the Food and Drug Administration’s Acceptable Daily Intake of 26.5 ng/day. Nitrosamines have carcinogenic potency when present above the allowable exposure limits, according to Sandoz, but the company said it “has not received any reports of adverse events related to the presence of a nitrosamine impurity in the lot.”
The Orphenadrine Citrate 100 mg ER Tablets were shipped to customers from August 2019 to April 2021 and have lot numbers of JX6411, JX6413, KC0723, KC3303, KE4348, KE7169, KE4349, KL3199, KM0072, KS3939, LA7704, LA7703, and LA9243.
The lots contain 100- and 1,000-count bottles of Orphenadrine Citrate ER Tablets, which are used as an adjunct to rest, physical therapy, and other measures for the relief of discomfort associated with acute painful musculoskeletal conditions.
The recall does not apply to any other strengths of Sandoz’s Orphenadrine Citrate ER Tablets or to other lot numbers of the product.
Sandoz advises that wholesalers and distributors should “immediately stop distribution of the recalled product and quarantine and return all recalled product in their inventory.” The company advises consumers to stop taking the recalled product and immediately consult with their physicians to obtain another prescription, notifying them of any problems that may be related to taking or using the tablets.
Sandoz says that retailers and consumers should contact Sedgwick directly by phone at 844-491-7869 or email at [email protected] to return the recalled product, and report adverse reactions to Sandoz by phone at (800) 525-8747 or by email at [email protected]. Adverse reactions and quality problems can be reported to the FDA’s MedWatch Adverse Event Reporting program either online, by regular mail, or by fax to 1-800-FDA-0178.
A version of this article first appeared on Medscape.com.
Updated perioperative guidance says when to hold antirheumatics
The American College of Rheumatology and the American Association of Hip and Knee Surgeons have released updated guidelines regarding whether to withhold drugs such as biologics and immunosuppressives for patients with inflammatory rheumatic disease who are scheduled to undergo elective total hip or knee replacement surgery.
The guidelines, published in a summary by the societies on Feb. 28, include revised and new recommendations about biologics and Janus kinase (JAK) inhibitors for patients with several types of inflammatory arthritis and systemic lupus erythematosus (SLE). In general, the guidelines recommend that the most powerful medications be withheld prior to surgery except for patients whose SLE is so severe that it threatens organs. They also recommend a shorter period of withholding drugs – 3 days instead of 7 – for JAK inhibitors.
The previous guidelines were published in 2017.
“These recommendations seek to balance flares of disease that are likely when medications are stopped vs. the risk of infection,” Susan M. Goodman, MD, a rheumatologist at the Hospital for Special Surgery, New York, and co–principal investigator of the guideline, told this news organization. “Patients and physicians may want to be either more conservative or more aggressive with their medications, depending on their personal priorities or specific medical history.”
According to Dr. Goodman, patients with inflammatory rheumatic diseases are especially likely to undergo joint replacement surgery because the conditions can damage the joints. “While the introduction of potent biologics has been linked to a decrease in surgery of soft tissues and small joints, there has been little impact on large-joint surgeries,” she said.
The risk of infection in these patients is about 50% higher than in the general population, she said. However, “it is hard to determine the magnitude of the effect of withholding medications, given the low rate of infection. In fact, using pharmaco-epidemiologic methods in large Medicare databases, no difference was seen in patients whose immunosuppressant medication infusions were close to the time of surgery compared to those patients whose medication infusions were months prior to surgery.”
The guidelines add a recommendation for the first time for apremilast (Otezla), saying that when it is administered twice daily it is okay to schedule surgery at any time.
Withholding drugs in patients with SLE
“We now recommend continuing biologics used to treat SLE – rituximab and belimumab – in patients with severe SLE but continue to recommend withholding them in less severe cases where there is little risk of organ damage,” Bryan D. Springer, MD, an orthopedic surgeon in Charlotte, N.C., first vice president of the AAHKS, and co–principal investigator of the new guidelines, told this news organization.
In severe SLE cases, the guidelines recommend timing total joint replacement surgery for 4-6 months after the latest IV dose of rituximab (Rituxan), which is given every 4-6 months. For patients taking belimumab (Benlysta), time surgery anytime when weekly subcutaneous doses are administered or at week 4 when monthly IV doses are given.
The guidelines also make recommendations regarding two new drugs for the treatment of severe SLE:
- Anifrolumab (Saphnelo): Time surgery at week 4 when IV treatment is given every 4 weeks.
- Voclosporin (Lupkynis): Continue doses when they’re given twice daily.
An ACR statement cautions that there are no published, peer-reviewed data regarding the use of these two drugs prior to total joint surgery. “The medications do increase the risk of infection,” the statement says, “and therefore their use in patients with severe SLE would merit review by the treating rheumatologist in consideration of surgery.”
Timing of stopping and restarting medication
The guidelines also recommend that certain drugs be withheld for patients with rheumatoid arthritis, ankylosing spondylitis, or any type of SLE and then “restarting the antirheumatic therapy once the wound shows evidence of healing, any sutures/staples are out, there is no significant swelling, erythema, or drainage, and there is no ongoing nonsurgical site infection, which is typically about 14 days.”
In regard to biologics, “we continue to recommend withholding biologic medications in patients with inflammatory arthritis, withholding the medication for a dosing cycle prior to surgery, and scheduling the surgery after that dose would be due,” Dr. Springer said. “For example, if a patient takes the medication every 4 weeks, the patient would withhold the dose of the medication and schedule surgery in the 5th week.”
The new recommendations for biologics suggest scheduling surgery at week 5 when the interleukin (IL)-17 inhibitor ixekizumab (Taltz) is given once every 4 weeks and at week 9 when the IL-23 inhibitor guselkumab (Tremfya) is given every 8 weeks.
The guidelines also revise the previous recommendation about tofacitinib (Xeljanz): Surgery should be scheduled on day 4 when the drug is given once or twice daily. New recommendations for fellow JAK inhibitors baricitinib (Olumiant, daily) and upadacitinib (Rinvoq, daily) are the same: Withhold for 3 days prior to surgery and perform surgery on the 4th day.
“We shortened the time between the last dose of JAK inhibitors and surgery to 3 days from 7 based on trial data demonstrating early flares when the drug was withheld, suggesting the immunosuppressant effect wears off sooner than we previously thought,” Dr. Springer said.
The guidelines caution that the recommendations for JAK inhibitors are for infection risk but do not consider the risk of cardiac events or venous thromboembolism.
In patients with nonsevere SLE, the guidelines revise the recommendations for mycophenolate mofetil (twice daily), cyclosporine (twice daily), and tacrolimus (twice daily, IV and oral). The new advice is to withhold the drugs for 1 week after last dose prior to surgery. New recommendations offer the same advice for belimumab, both IV and subcutaneous: Withhold for 1 week after last dose prior to surgery.
The board of the ACR approved the guidelines summary; the full manuscript has been submitted for peer review with an eye toward later publication in the journals Arthritis and Rheumatology and Arthritis Care and Research.
The ACR and AAHKS funded the guidelines. Dr. Goodman and Dr. Springer report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The American College of Rheumatology and the American Association of Hip and Knee Surgeons have released updated guidelines regarding whether to withhold drugs such as biologics and immunosuppressives for patients with inflammatory rheumatic disease who are scheduled to undergo elective total hip or knee replacement surgery.
The guidelines, published in a summary by the societies on Feb. 28, include revised and new recommendations about biologics and Janus kinase (JAK) inhibitors for patients with several types of inflammatory arthritis and systemic lupus erythematosus (SLE). In general, the guidelines recommend that the most powerful medications be withheld prior to surgery except for patients whose SLE is so severe that it threatens organs. They also recommend a shorter period of withholding drugs – 3 days instead of 7 – for JAK inhibitors.
The previous guidelines were published in 2017.
“These recommendations seek to balance flares of disease that are likely when medications are stopped vs. the risk of infection,” Susan M. Goodman, MD, a rheumatologist at the Hospital for Special Surgery, New York, and co–principal investigator of the guideline, told this news organization. “Patients and physicians may want to be either more conservative or more aggressive with their medications, depending on their personal priorities or specific medical history.”
According to Dr. Goodman, patients with inflammatory rheumatic diseases are especially likely to undergo joint replacement surgery because the conditions can damage the joints. “While the introduction of potent biologics has been linked to a decrease in surgery of soft tissues and small joints, there has been little impact on large-joint surgeries,” she said.
The risk of infection in these patients is about 50% higher than in the general population, she said. However, “it is hard to determine the magnitude of the effect of withholding medications, given the low rate of infection. In fact, using pharmaco-epidemiologic methods in large Medicare databases, no difference was seen in patients whose immunosuppressant medication infusions were close to the time of surgery compared to those patients whose medication infusions were months prior to surgery.”
The guidelines add a recommendation for the first time for apremilast (Otezla), saying that when it is administered twice daily it is okay to schedule surgery at any time.
Withholding drugs in patients with SLE
“We now recommend continuing biologics used to treat SLE – rituximab and belimumab – in patients with severe SLE but continue to recommend withholding them in less severe cases where there is little risk of organ damage,” Bryan D. Springer, MD, an orthopedic surgeon in Charlotte, N.C., first vice president of the AAHKS, and co–principal investigator of the new guidelines, told this news organization.
In severe SLE cases, the guidelines recommend timing total joint replacement surgery for 4-6 months after the latest IV dose of rituximab (Rituxan), which is given every 4-6 months. For patients taking belimumab (Benlysta), time surgery anytime when weekly subcutaneous doses are administered or at week 4 when monthly IV doses are given.
The guidelines also make recommendations regarding two new drugs for the treatment of severe SLE:
- Anifrolumab (Saphnelo): Time surgery at week 4 when IV treatment is given every 4 weeks.
- Voclosporin (Lupkynis): Continue doses when they’re given twice daily.
An ACR statement cautions that there are no published, peer-reviewed data regarding the use of these two drugs prior to total joint surgery. “The medications do increase the risk of infection,” the statement says, “and therefore their use in patients with severe SLE would merit review by the treating rheumatologist in consideration of surgery.”
Timing of stopping and restarting medication
The guidelines also recommend that certain drugs be withheld for patients with rheumatoid arthritis, ankylosing spondylitis, or any type of SLE and then “restarting the antirheumatic therapy once the wound shows evidence of healing, any sutures/staples are out, there is no significant swelling, erythema, or drainage, and there is no ongoing nonsurgical site infection, which is typically about 14 days.”
In regard to biologics, “we continue to recommend withholding biologic medications in patients with inflammatory arthritis, withholding the medication for a dosing cycle prior to surgery, and scheduling the surgery after that dose would be due,” Dr. Springer said. “For example, if a patient takes the medication every 4 weeks, the patient would withhold the dose of the medication and schedule surgery in the 5th week.”
The new recommendations for biologics suggest scheduling surgery at week 5 when the interleukin (IL)-17 inhibitor ixekizumab (Taltz) is given once every 4 weeks and at week 9 when the IL-23 inhibitor guselkumab (Tremfya) is given every 8 weeks.
The guidelines also revise the previous recommendation about tofacitinib (Xeljanz): Surgery should be scheduled on day 4 when the drug is given once or twice daily. New recommendations for fellow JAK inhibitors baricitinib (Olumiant, daily) and upadacitinib (Rinvoq, daily) are the same: Withhold for 3 days prior to surgery and perform surgery on the 4th day.
“We shortened the time between the last dose of JAK inhibitors and surgery to 3 days from 7 based on trial data demonstrating early flares when the drug was withheld, suggesting the immunosuppressant effect wears off sooner than we previously thought,” Dr. Springer said.
The guidelines caution that the recommendations for JAK inhibitors are for infection risk but do not consider the risk of cardiac events or venous thromboembolism.
In patients with nonsevere SLE, the guidelines revise the recommendations for mycophenolate mofetil (twice daily), cyclosporine (twice daily), and tacrolimus (twice daily, IV and oral). The new advice is to withhold the drugs for 1 week after last dose prior to surgery. New recommendations offer the same advice for belimumab, both IV and subcutaneous: Withhold for 1 week after last dose prior to surgery.
The board of the ACR approved the guidelines summary; the full manuscript has been submitted for peer review with an eye toward later publication in the journals Arthritis and Rheumatology and Arthritis Care and Research.
The ACR and AAHKS funded the guidelines. Dr. Goodman and Dr. Springer report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The American College of Rheumatology and the American Association of Hip and Knee Surgeons have released updated guidelines regarding whether to withhold drugs such as biologics and immunosuppressives for patients with inflammatory rheumatic disease who are scheduled to undergo elective total hip or knee replacement surgery.
The guidelines, published in a summary by the societies on Feb. 28, include revised and new recommendations about biologics and Janus kinase (JAK) inhibitors for patients with several types of inflammatory arthritis and systemic lupus erythematosus (SLE). In general, the guidelines recommend that the most powerful medications be withheld prior to surgery except for patients whose SLE is so severe that it threatens organs. They also recommend a shorter period of withholding drugs – 3 days instead of 7 – for JAK inhibitors.
The previous guidelines were published in 2017.
“These recommendations seek to balance flares of disease that are likely when medications are stopped vs. the risk of infection,” Susan M. Goodman, MD, a rheumatologist at the Hospital for Special Surgery, New York, and co–principal investigator of the guideline, told this news organization. “Patients and physicians may want to be either more conservative or more aggressive with their medications, depending on their personal priorities or specific medical history.”
According to Dr. Goodman, patients with inflammatory rheumatic diseases are especially likely to undergo joint replacement surgery because the conditions can damage the joints. “While the introduction of potent biologics has been linked to a decrease in surgery of soft tissues and small joints, there has been little impact on large-joint surgeries,” she said.
The risk of infection in these patients is about 50% higher than in the general population, she said. However, “it is hard to determine the magnitude of the effect of withholding medications, given the low rate of infection. In fact, using pharmaco-epidemiologic methods in large Medicare databases, no difference was seen in patients whose immunosuppressant medication infusions were close to the time of surgery compared to those patients whose medication infusions were months prior to surgery.”
The guidelines add a recommendation for the first time for apremilast (Otezla), saying that when it is administered twice daily it is okay to schedule surgery at any time.
Withholding drugs in patients with SLE
“We now recommend continuing biologics used to treat SLE – rituximab and belimumab – in patients with severe SLE but continue to recommend withholding them in less severe cases where there is little risk of organ damage,” Bryan D. Springer, MD, an orthopedic surgeon in Charlotte, N.C., first vice president of the AAHKS, and co–principal investigator of the new guidelines, told this news organization.
In severe SLE cases, the guidelines recommend timing total joint replacement surgery for 4-6 months after the latest IV dose of rituximab (Rituxan), which is given every 4-6 months. For patients taking belimumab (Benlysta), time surgery anytime when weekly subcutaneous doses are administered or at week 4 when monthly IV doses are given.
The guidelines also make recommendations regarding two new drugs for the treatment of severe SLE:
- Anifrolumab (Saphnelo): Time surgery at week 4 when IV treatment is given every 4 weeks.
- Voclosporin (Lupkynis): Continue doses when they’re given twice daily.
An ACR statement cautions that there are no published, peer-reviewed data regarding the use of these two drugs prior to total joint surgery. “The medications do increase the risk of infection,” the statement says, “and therefore their use in patients with severe SLE would merit review by the treating rheumatologist in consideration of surgery.”
Timing of stopping and restarting medication
The guidelines also recommend that certain drugs be withheld for patients with rheumatoid arthritis, ankylosing spondylitis, or any type of SLE and then “restarting the antirheumatic therapy once the wound shows evidence of healing, any sutures/staples are out, there is no significant swelling, erythema, or drainage, and there is no ongoing nonsurgical site infection, which is typically about 14 days.”
In regard to biologics, “we continue to recommend withholding biologic medications in patients with inflammatory arthritis, withholding the medication for a dosing cycle prior to surgery, and scheduling the surgery after that dose would be due,” Dr. Springer said. “For example, if a patient takes the medication every 4 weeks, the patient would withhold the dose of the medication and schedule surgery in the 5th week.”
The new recommendations for biologics suggest scheduling surgery at week 5 when the interleukin (IL)-17 inhibitor ixekizumab (Taltz) is given once every 4 weeks and at week 9 when the IL-23 inhibitor guselkumab (Tremfya) is given every 8 weeks.
The guidelines also revise the previous recommendation about tofacitinib (Xeljanz): Surgery should be scheduled on day 4 when the drug is given once or twice daily. New recommendations for fellow JAK inhibitors baricitinib (Olumiant, daily) and upadacitinib (Rinvoq, daily) are the same: Withhold for 3 days prior to surgery and perform surgery on the 4th day.
“We shortened the time between the last dose of JAK inhibitors and surgery to 3 days from 7 based on trial data demonstrating early flares when the drug was withheld, suggesting the immunosuppressant effect wears off sooner than we previously thought,” Dr. Springer said.
The guidelines caution that the recommendations for JAK inhibitors are for infection risk but do not consider the risk of cardiac events or venous thromboembolism.
In patients with nonsevere SLE, the guidelines revise the recommendations for mycophenolate mofetil (twice daily), cyclosporine (twice daily), and tacrolimus (twice daily, IV and oral). The new advice is to withhold the drugs for 1 week after last dose prior to surgery. New recommendations offer the same advice for belimumab, both IV and subcutaneous: Withhold for 1 week after last dose prior to surgery.
The board of the ACR approved the guidelines summary; the full manuscript has been submitted for peer review with an eye toward later publication in the journals Arthritis and Rheumatology and Arthritis Care and Research.
The ACR and AAHKS funded the guidelines. Dr. Goodman and Dr. Springer report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Mild Grisel Syndrome: Expanding the Differential for Posttonsillectomy Adenoidectomy Symptoms
Tonsillectomy with or without adenoidectomy (T&A) is the second most common pediatric surgical procedure in the United States.1 It is most often performed during childhood between 5 and 8 years of age with a second peak observed between 17 and 21 years of age in the adolescent and young adult populations.2 While recurrent tonsillitis has been traditionally associated with tonsillectomy, sleep disordered breathing with obstructive sleep apnea is now the primary indication for the procedure.1
Up to 97% of T&As are performed as an outpatient same-day surgery not requiring inpatient admission.2 Although largely a safe and routinely performed surgery, several complications have been described. Due to the outpatient nature of the procedure, the complications are often encountered in the emergency department (ED) and sometimes in primary care settings. Common complications (outside of the perioperative time frame) include nausea, vomiting, otalgia, odynophagia, infection of the throat (broadly), and hemorrhage; uncommon complications include subcutaneous emphysema, taste disorders, and Eagle syndrome. Some complications are rarer still and carry significant morbidity and even mortality, including mediastinitis, cervical osteomyelitis, and Grisel syndrome.3 The following case encourages the clinician to expand the differential for a patient presenting after T&A.
Case Presentation
A child aged < 3 years was brought to the ED by their mother. She reported neck pain and stiffness 10 days after T&A with concurrent tympanostomy tube placement at an outside pediatric hospital. At triage, their heart rate was 94 bpm, temperature was 98.2 °F, respiratory rate, 22 breaths per minute, and oxygen saturation, 97% on room air. The mother of the patient (MOP) had been giving the prescribed oral liquid formulations of ibuprofen and acetaminophen with hydrocodone as directed. No drug allergies were reported, and immunizations were up to date for age. Other medical and surgical history included eczema and remote cutaneous hemangioma resection. The patient lived at home with 2 parents and was not exposed to smoke; their family history was noncontributory.
Since the surgery, the MOP had noticed constant and increasing neck stiffness, specifically with looking up and down but not side to side. She also had noticed swelling behind both ears. She reported no substantial decrease in intake by mouth or decrease in urine or bowel frequency. On review of systems, she reported no fever, vomiting, difficulty breathing, bleeding from the mouth or nose, eye or ear drainage, or rash.
On physical examination, the patient was alert and in no acute distress; active and playful on an electronic device but was notably not moving their head, which was held in a forward-looking position without any signs of trauma. When asked, the child would not flex or extend their neck but would rotate a few degrees from neutral to both sides. Even with moving the electronic device up and down in space, no active neck extension or flexion could be elicited. The examination of the head, eyes, ears, nose, and throat was otherwise only remarkable for palpable and mildly tender postauricular lymph nodes and diffuse erythema in the posterior pharynx. Cardiopulmonary, abdominal, skin, and extremity examinations were unremarkable.
With concern for an infectious process, the physician ordered blood chemistry and hematology tests along with neck radiography. While awaiting the results, the patient was given a weight-based bolus of normal saline, and the home pain regimen was administered. An attempt was made to passively flex and extend the neck as the child slept in their mother’s arms, but the patient immediately awoke and began to cry.
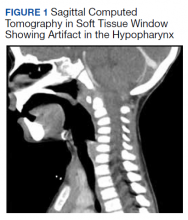
All values of the comprehensive metabolic panel were within normal limits except for a slight elevation in the blood urea nitrogen to 21 mg/dL and glucose to 159 mg/dL. The complete blood count was unrevealing. The computed tomography (CT) scan with contrast of the soft tissues of the neck was limited by motion artifact but showed a head held in axial rotation with soft tissue irregularity in the anterior aspect of the adenoids (Figure 1). There was what appeared to be normal lymphadenopathy in the hypopharynx, but the soft tissues were otherwise unremarkable.
The on-call pediatric otolaryngologist at the hospital where the procedure was performed was paged. On hearing the details of the case, the specialist was concerned for Grisel syndrome and requested to see the patient in their facility. No additional recommendations for care were provided; the mother was updated and agreed to transfer. The patient was comfortable and stable with repeat vitals as follows: heart rate, 86 beats per minute, blood pressure, 99/62, temperature, 98.3 °F, respiratory rate, 20 breaths per minute, and oxygen saturation, 99% on room air.
On arrival at the receiving facility, the emergency team performed a history and physical that revealed no significant changes from the initial evaluation. They then facilitated evaluation by the pediatric otolaryngologist who conducted a more directed physical examination. Decreased active and passive range of motion (ROM) of the neck without rotatory restriction was again noted. They also observed scant fibrinous exudate within the oropharynx and tonsillar fossa, which was normal in the setting of the recent surgery. They recommended additional analgesia with intramuscular ketorolac, weight-based dosing at 1 mg/kg.
With repeat examination after this additional analgesic, ROM of the neck first passive then active had improved. The patient was then discharged to follow up in the coming days with instructions to continue the pain and anti-inflammatory regimen. They were not started on an antibiotic at that time nor were they placed in a cervical collar. At the follow-up, the MOP reported persistence of neck stiffness for a few days initially but then observed slow improvement. By postoperative day 18, the stiffness had resolved. No other follow-up or referrals related to this issue were readily apparent in review of the patient’s health record.
Discussion
Grisel syndrome is the atraumatic rotary subluxation of the atlantoaxial joint, specifically, the atlas (C1 vertebra) rotates to a fixed, nonanatomic position while the axis (C2 vertebra) remains in normal alignment in relation to the remainder of the spinal column. The subluxation occurs in the absence of ligamentous injury but is associated with an increase in ligamentous laxity.4 The atlas is a ring-shaped vertebra with 2 lateral masses connected by anterior and posterior arches; it lacks a spinous process unlike other vertebrae. It articulates with the skull by means of the 2 articular facets on the superior aspect of the lateral masses. Articulation with the axis occurs at 3 sites: 2 articular facets on the inferior portion of the lateral masses of the atlas and a facet for the dens on the posterior portion of the anterior arch. The dens projects superiorly from the body of the axis and is bound posteriorly by the transverse ligament of the atlas.5
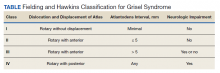
The degree of subluxation seen in Grisel syndrome correlates to the disease severity and is classified by the Fielding and Hawkins (FH) system (Table). This system accounts for the distance from the atlas to the dens (atlantodens interval) and the relative asymmetry of the atlantoaxial joint.6 In a normal adult, the upper limit of normal for the atlantodens interval is 3 mm, whereas this distance increases to 4.5 mm for the pediatric population.7 Type I (FH-I) involves rotary subluxation alone without any increase in the atlantodens interval; in FH-II, that interval has increased from normal but to no more than 5 mm. FH-I and FH-II are the most encountered and are not associated with neurologic impairment. In FH-III, neurologic deficits can be present, and the atlantodens interval is increased to > 5 mm. Different from FH-II and FH-III in which anterior dislocation of the atlas with reference to the dens is observed, FH-IV involves a rotary movement of the atlas with concurrent posterior displacement and often involves spinal cord compression.6
Subluxation and displacement without trauma are key components of Grisel syndrome. The 2-hit hypothesis is often used to explain how this can occur, ie, 2 anomalies must be present simultaneously for this condition to develop. First, the laxity of the transverse ligament, the posterior wall of the dens, and other atlantoaxial ligaments must be increased. Second, an asymmetric contraction of the deep erector muscles of the neck either abruptly or more insidiously rotate and dislocate the atlas.8 The pathophysiology is not exactly understood, but the most commonly held hypothesis describes contiguous spread of infection or inflammatory mediators from the pharynx to the ligaments and muscles described.6
Spread could occur via the venous system. The posterior superior pharyngeal region is drained by the periodontoidal venous plexus; the connections here with the pharyngovertebral veins allow for the embolization of infectious or other proinflammatory material to the prevertebral fascia. These emboli induce fasciitis and subsequent aberrant relaxation of the ligaments. In reaction to the inflammation or increased laxity, contiguous muscles of the deep neck contract and freeze the joint out of anatomic alignment.4
The abnormal alignment is apparent grossly as torticollis. Most broadly, torticollis describes an anomalous head posture due to involuntary muscle contractions of neck muscles and specifically describes chin deviation to the side. The antecollis and retrocollis subtypes of torticollis describe forward flexion and backward extension of the neck, respectively.7 Torticollis (broadly) is the most frequently reported condition of those found to have Grisel syndrome (90.7%); other common presenting conditions include neck pain (81.5%) and neck stiffness (31.5%). Fever is found in only 27.8% of cases. Pediatric patients (aged ≤ 12 years) are the most commonly affected, accounting for 87% of cases with an observed 4:1 male to female predominance.7,8 Symptoms begin most often within the first week from the inciting event in 85% of the cases.8 Head and neck surgery precedes up to 67% of cases, and infectious etiologies largely account for the remaining cases.7 Of the postsurgical cases, 55.6% had undergone T&A.8
Although anomalous head posture or neck stiffness following T&A would be of great clinic concern for Grisel syndrome, radiographic studies play a confirmatory role. CT scan is used to evaluate the bony structures, with 3D reconstruction of the cervical spine being most useful to determine the presence and degree of subluxation.8 Magnetic resonance imaging also aids in diagnosis to evaluate ligamentous structures in the area of concern as well as in the evaluation of spinal cord compression.6 Laboratory tests are largely unhelpful in making or excluding the diagnosis.8
If Grisel syndrome is suspected, both the original surgeon (if preceded by surgery) and the neurosurgical team should be consulted. Although no widely adopted guidelines exist for the management of this rare disease, general practice patterns have emerged with the degree of intervention predictably correlating to disease severity. FH-I is usually treated with nonsteroidal anti-inflammatory drugs and muscle relaxants with or without a soft cervical collar. For FH-II, closed reduction and immobilization in a stiff cervical collar is recommended. If no neurologic defect is present, FH-III is treated with bed rest, a period of inline cervical traction, and subsequent immobilization. FH-III with neurologic sequelae and all FH-IV necessitate emergent neurosurgical consultation.4 Surgical intervention is a last resort but is required in up to 24.1% of cases.8
Antibiotic therapy is not routinely given unless clear infectious etiology is identified. No standard antibiotic regimen exists, but coverage for typical upper respiratory pathogens likely suffices. Empiric antibiotic therapy is not recommended for all causes of Grisel syndrome, ie, when the underlying cause is not yet elucidated.6 One case of Grisel syndrome occurring in the setting of cervical osteomyelitis has been described, though, and required prolonged IV antibiotics.3 Physical therapy is recommended as adjunct with no limitations for range of motion save for that of the patient’s individual pain threshold.4
Possibly attributable to waxing and waning ligamentous laxity and strength of the neck muscle contraction, the atlantodens interval and the degree of subluxation can change, making Grisel syndrome dynamic. As such, the FH classification can change, necessitating more or less aggressive therapy. A neurologic evaluation is recommended at least every 2 weeks after the diagnosis is made. If initial identification or recognition of known disease progression is delayed, serious complications can develop. Acutely, spinal cord compression can lead to quadriplegia and death; more insidious complications include reduced neck mobility, dysphonia, and dysphagia.4 As serious, life-threatening complications can arise from Grisel syndrome while good functional outcomes can be achieved with timely and appropriate treatment, the clinician should be inspired to have a high clinical suspicion for this syndrome given the right context.
Conclusions
The patient experienced a desirable outcome with minimal, conservative treatment. As such, the pathology in this case was likely attributed to the mildest form of Grisel syndrome (FH-I). The follow-up was reassuring as well, revealing no worsening or progression of symptoms. The initial evaluation in this case was limited by the inadequacy of the CT scan. Motion artifact in the pharynx prevented the definite exclusion of deep space infection, while the rotation of the head in combination with motion artifact in the cranial-most portions of the vertebral column made determining alignment difficult. One clear axial image, though, does show rotation of the atlas (Figure 2). The uncertainty at the end of our workup prompted surgical consultation, not, admittedly, concern for Grisel syndrome. Awareness of this disease entity is nevertheless important and clinically relevant. Early identification and treatment is associated with decreased morbidity and improvement in long-term functional outcomes.6 Despite its rarity, the clinician should consider Grisel syndrome in any pediatric patient presenting with neck stiffness following the commonly performed T&A.
1. Ramos SD, Mukerji S, Pine HS. Tonsillectomy and adenoidectomy. Pediatr Clin North Am. 2013;60(4):793-807. doi:10.1016/j.pcl.2013.04.015
2. Stoner MJ, Dulaurier M. Pediatric ENT emergencies. Emerg Med Clin North Am. 2013;31(3):795-808. doi:10.1016/j.emc.2013.04.005
3. Leong SC, Karoos PD, Papouliakos SM, et al. Unusual complications of tonsillectomy: a systematic review. Am J Otolaryngol. 2007;28(6):419-422. doi:10.1016/j.amjoto.2006.10.016
4. Fath L, Cebula H, Santin MN, Cocab A, Debrya C, Proustb F. The Grisel’s syndrome: a non-traumatic subluxation of the atlantoaxial joint. Neurochirurgie. 2018;64(4):327-330. doi:10.1016/j.neuchi.2018.02.001
5. Moore K, Agur A, Dalley A. Essential Clinical Anatomy. 5th ed. Baltimore: Lippincott, Williams, and Wilkins; 2015:282-287.
6. Spennato P, Nicosia G, Rapanà A, et al. Grisel syndrome following adenoidectomy: surgical management in a case with delayed diagnosis. World Neurosurg. 2015;84(5):1494.e7-e12.
7. Anania P, Pavone P, Pacetti M, et al. Grisel syndrome in pediatric age: a single-center Italian experience and review of the literature. World Neurosurg. 2019;125:374-382. doi:10.1016/j.wneu.2019.02.035
8. Aldriweesh T, Altheyab F, Alenezi M, et al. Grisel’s syndrome post otolaryngology procedures: a systematic review. Int J Pediatr Otorhinolaryngol. 2020;137:110-125. doi:10.1016/j.ijporl.2020.110225
Tonsillectomy with or without adenoidectomy (T&A) is the second most common pediatric surgical procedure in the United States.1 It is most often performed during childhood between 5 and 8 years of age with a second peak observed between 17 and 21 years of age in the adolescent and young adult populations.2 While recurrent tonsillitis has been traditionally associated with tonsillectomy, sleep disordered breathing with obstructive sleep apnea is now the primary indication for the procedure.1
Up to 97% of T&As are performed as an outpatient same-day surgery not requiring inpatient admission.2 Although largely a safe and routinely performed surgery, several complications have been described. Due to the outpatient nature of the procedure, the complications are often encountered in the emergency department (ED) and sometimes in primary care settings. Common complications (outside of the perioperative time frame) include nausea, vomiting, otalgia, odynophagia, infection of the throat (broadly), and hemorrhage; uncommon complications include subcutaneous emphysema, taste disorders, and Eagle syndrome. Some complications are rarer still and carry significant morbidity and even mortality, including mediastinitis, cervical osteomyelitis, and Grisel syndrome.3 The following case encourages the clinician to expand the differential for a patient presenting after T&A.
Case Presentation
A child aged < 3 years was brought to the ED by their mother. She reported neck pain and stiffness 10 days after T&A with concurrent tympanostomy tube placement at an outside pediatric hospital. At triage, their heart rate was 94 bpm, temperature was 98.2 °F, respiratory rate, 22 breaths per minute, and oxygen saturation, 97% on room air. The mother of the patient (MOP) had been giving the prescribed oral liquid formulations of ibuprofen and acetaminophen with hydrocodone as directed. No drug allergies were reported, and immunizations were up to date for age. Other medical and surgical history included eczema and remote cutaneous hemangioma resection. The patient lived at home with 2 parents and was not exposed to smoke; their family history was noncontributory.
Since the surgery, the MOP had noticed constant and increasing neck stiffness, specifically with looking up and down but not side to side. She also had noticed swelling behind both ears. She reported no substantial decrease in intake by mouth or decrease in urine or bowel frequency. On review of systems, she reported no fever, vomiting, difficulty breathing, bleeding from the mouth or nose, eye or ear drainage, or rash.
On physical examination, the patient was alert and in no acute distress; active and playful on an electronic device but was notably not moving their head, which was held in a forward-looking position without any signs of trauma. When asked, the child would not flex or extend their neck but would rotate a few degrees from neutral to both sides. Even with moving the electronic device up and down in space, no active neck extension or flexion could be elicited. The examination of the head, eyes, ears, nose, and throat was otherwise only remarkable for palpable and mildly tender postauricular lymph nodes and diffuse erythema in the posterior pharynx. Cardiopulmonary, abdominal, skin, and extremity examinations were unremarkable.
With concern for an infectious process, the physician ordered blood chemistry and hematology tests along with neck radiography. While awaiting the results, the patient was given a weight-based bolus of normal saline, and the home pain regimen was administered. An attempt was made to passively flex and extend the neck as the child slept in their mother’s arms, but the patient immediately awoke and began to cry.
All values of the comprehensive metabolic panel were within normal limits except for a slight elevation in the blood urea nitrogen to 21 mg/dL and glucose to 159 mg/dL. The complete blood count was unrevealing. The computed tomography (CT) scan with contrast of the soft tissues of the neck was limited by motion artifact but showed a head held in axial rotation with soft tissue irregularity in the anterior aspect of the adenoids (Figure 1). There was what appeared to be normal lymphadenopathy in the hypopharynx, but the soft tissues were otherwise unremarkable.
The on-call pediatric otolaryngologist at the hospital where the procedure was performed was paged. On hearing the details of the case, the specialist was concerned for Grisel syndrome and requested to see the patient in their facility. No additional recommendations for care were provided; the mother was updated and agreed to transfer. The patient was comfortable and stable with repeat vitals as follows: heart rate, 86 beats per minute, blood pressure, 99/62, temperature, 98.3 °F, respiratory rate, 20 breaths per minute, and oxygen saturation, 99% on room air.
On arrival at the receiving facility, the emergency team performed a history and physical that revealed no significant changes from the initial evaluation. They then facilitated evaluation by the pediatric otolaryngologist who conducted a more directed physical examination. Decreased active and passive range of motion (ROM) of the neck without rotatory restriction was again noted. They also observed scant fibrinous exudate within the oropharynx and tonsillar fossa, which was normal in the setting of the recent surgery. They recommended additional analgesia with intramuscular ketorolac, weight-based dosing at 1 mg/kg.
With repeat examination after this additional analgesic, ROM of the neck first passive then active had improved. The patient was then discharged to follow up in the coming days with instructions to continue the pain and anti-inflammatory regimen. They were not started on an antibiotic at that time nor were they placed in a cervical collar. At the follow-up, the MOP reported persistence of neck stiffness for a few days initially but then observed slow improvement. By postoperative day 18, the stiffness had resolved. No other follow-up or referrals related to this issue were readily apparent in review of the patient’s health record.
Discussion
Grisel syndrome is the atraumatic rotary subluxation of the atlantoaxial joint, specifically, the atlas (C1 vertebra) rotates to a fixed, nonanatomic position while the axis (C2 vertebra) remains in normal alignment in relation to the remainder of the spinal column. The subluxation occurs in the absence of ligamentous injury but is associated with an increase in ligamentous laxity.4 The atlas is a ring-shaped vertebra with 2 lateral masses connected by anterior and posterior arches; it lacks a spinous process unlike other vertebrae. It articulates with the skull by means of the 2 articular facets on the superior aspect of the lateral masses. Articulation with the axis occurs at 3 sites: 2 articular facets on the inferior portion of the lateral masses of the atlas and a facet for the dens on the posterior portion of the anterior arch. The dens projects superiorly from the body of the axis and is bound posteriorly by the transverse ligament of the atlas.5
The degree of subluxation seen in Grisel syndrome correlates to the disease severity and is classified by the Fielding and Hawkins (FH) system (Table). This system accounts for the distance from the atlas to the dens (atlantodens interval) and the relative asymmetry of the atlantoaxial joint.6 In a normal adult, the upper limit of normal for the atlantodens interval is 3 mm, whereas this distance increases to 4.5 mm for the pediatric population.7 Type I (FH-I) involves rotary subluxation alone without any increase in the atlantodens interval; in FH-II, that interval has increased from normal but to no more than 5 mm. FH-I and FH-II are the most encountered and are not associated with neurologic impairment. In FH-III, neurologic deficits can be present, and the atlantodens interval is increased to > 5 mm. Different from FH-II and FH-III in which anterior dislocation of the atlas with reference to the dens is observed, FH-IV involves a rotary movement of the atlas with concurrent posterior displacement and often involves spinal cord compression.6
Subluxation and displacement without trauma are key components of Grisel syndrome. The 2-hit hypothesis is often used to explain how this can occur, ie, 2 anomalies must be present simultaneously for this condition to develop. First, the laxity of the transverse ligament, the posterior wall of the dens, and other atlantoaxial ligaments must be increased. Second, an asymmetric contraction of the deep erector muscles of the neck either abruptly or more insidiously rotate and dislocate the atlas.8 The pathophysiology is not exactly understood, but the most commonly held hypothesis describes contiguous spread of infection or inflammatory mediators from the pharynx to the ligaments and muscles described.6
Spread could occur via the venous system. The posterior superior pharyngeal region is drained by the periodontoidal venous plexus; the connections here with the pharyngovertebral veins allow for the embolization of infectious or other proinflammatory material to the prevertebral fascia. These emboli induce fasciitis and subsequent aberrant relaxation of the ligaments. In reaction to the inflammation or increased laxity, contiguous muscles of the deep neck contract and freeze the joint out of anatomic alignment.4
The abnormal alignment is apparent grossly as torticollis. Most broadly, torticollis describes an anomalous head posture due to involuntary muscle contractions of neck muscles and specifically describes chin deviation to the side. The antecollis and retrocollis subtypes of torticollis describe forward flexion and backward extension of the neck, respectively.7 Torticollis (broadly) is the most frequently reported condition of those found to have Grisel syndrome (90.7%); other common presenting conditions include neck pain (81.5%) and neck stiffness (31.5%). Fever is found in only 27.8% of cases. Pediatric patients (aged ≤ 12 years) are the most commonly affected, accounting for 87% of cases with an observed 4:1 male to female predominance.7,8 Symptoms begin most often within the first week from the inciting event in 85% of the cases.8 Head and neck surgery precedes up to 67% of cases, and infectious etiologies largely account for the remaining cases.7 Of the postsurgical cases, 55.6% had undergone T&A.8
Although anomalous head posture or neck stiffness following T&A would be of great clinic concern for Grisel syndrome, radiographic studies play a confirmatory role. CT scan is used to evaluate the bony structures, with 3D reconstruction of the cervical spine being most useful to determine the presence and degree of subluxation.8 Magnetic resonance imaging also aids in diagnosis to evaluate ligamentous structures in the area of concern as well as in the evaluation of spinal cord compression.6 Laboratory tests are largely unhelpful in making or excluding the diagnosis.8
If Grisel syndrome is suspected, both the original surgeon (if preceded by surgery) and the neurosurgical team should be consulted. Although no widely adopted guidelines exist for the management of this rare disease, general practice patterns have emerged with the degree of intervention predictably correlating to disease severity. FH-I is usually treated with nonsteroidal anti-inflammatory drugs and muscle relaxants with or without a soft cervical collar. For FH-II, closed reduction and immobilization in a stiff cervical collar is recommended. If no neurologic defect is present, FH-III is treated with bed rest, a period of inline cervical traction, and subsequent immobilization. FH-III with neurologic sequelae and all FH-IV necessitate emergent neurosurgical consultation.4 Surgical intervention is a last resort but is required in up to 24.1% of cases.8
Antibiotic therapy is not routinely given unless clear infectious etiology is identified. No standard antibiotic regimen exists, but coverage for typical upper respiratory pathogens likely suffices. Empiric antibiotic therapy is not recommended for all causes of Grisel syndrome, ie, when the underlying cause is not yet elucidated.6 One case of Grisel syndrome occurring in the setting of cervical osteomyelitis has been described, though, and required prolonged IV antibiotics.3 Physical therapy is recommended as adjunct with no limitations for range of motion save for that of the patient’s individual pain threshold.4
Possibly attributable to waxing and waning ligamentous laxity and strength of the neck muscle contraction, the atlantodens interval and the degree of subluxation can change, making Grisel syndrome dynamic. As such, the FH classification can change, necessitating more or less aggressive therapy. A neurologic evaluation is recommended at least every 2 weeks after the diagnosis is made. If initial identification or recognition of known disease progression is delayed, serious complications can develop. Acutely, spinal cord compression can lead to quadriplegia and death; more insidious complications include reduced neck mobility, dysphonia, and dysphagia.4 As serious, life-threatening complications can arise from Grisel syndrome while good functional outcomes can be achieved with timely and appropriate treatment, the clinician should be inspired to have a high clinical suspicion for this syndrome given the right context.
Conclusions
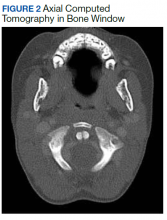
The patient experienced a desirable outcome with minimal, conservative treatment. As such, the pathology in this case was likely attributed to the mildest form of Grisel syndrome (FH-I). The follow-up was reassuring as well, revealing no worsening or progression of symptoms. The initial evaluation in this case was limited by the inadequacy of the CT scan. Motion artifact in the pharynx prevented the definite exclusion of deep space infection, while the rotation of the head in combination with motion artifact in the cranial-most portions of the vertebral column made determining alignment difficult. One clear axial image, though, does show rotation of the atlas (Figure 2). The uncertainty at the end of our workup prompted surgical consultation, not, admittedly, concern for Grisel syndrome. Awareness of this disease entity is nevertheless important and clinically relevant. Early identification and treatment is associated with decreased morbidity and improvement in long-term functional outcomes.6 Despite its rarity, the clinician should consider Grisel syndrome in any pediatric patient presenting with neck stiffness following the commonly performed T&A.
Tonsillectomy with or without adenoidectomy (T&A) is the second most common pediatric surgical procedure in the United States.1 It is most often performed during childhood between 5 and 8 years of age with a second peak observed between 17 and 21 years of age in the adolescent and young adult populations.2 While recurrent tonsillitis has been traditionally associated with tonsillectomy, sleep disordered breathing with obstructive sleep apnea is now the primary indication for the procedure.1
Up to 97% of T&As are performed as an outpatient same-day surgery not requiring inpatient admission.2 Although largely a safe and routinely performed surgery, several complications have been described. Due to the outpatient nature of the procedure, the complications are often encountered in the emergency department (ED) and sometimes in primary care settings. Common complications (outside of the perioperative time frame) include nausea, vomiting, otalgia, odynophagia, infection of the throat (broadly), and hemorrhage; uncommon complications include subcutaneous emphysema, taste disorders, and Eagle syndrome. Some complications are rarer still and carry significant morbidity and even mortality, including mediastinitis, cervical osteomyelitis, and Grisel syndrome.3 The following case encourages the clinician to expand the differential for a patient presenting after T&A.
Case Presentation
A child aged < 3 years was brought to the ED by their mother. She reported neck pain and stiffness 10 days after T&A with concurrent tympanostomy tube placement at an outside pediatric hospital. At triage, their heart rate was 94 bpm, temperature was 98.2 °F, respiratory rate, 22 breaths per minute, and oxygen saturation, 97% on room air. The mother of the patient (MOP) had been giving the prescribed oral liquid formulations of ibuprofen and acetaminophen with hydrocodone as directed. No drug allergies were reported, and immunizations were up to date for age. Other medical and surgical history included eczema and remote cutaneous hemangioma resection. The patient lived at home with 2 parents and was not exposed to smoke; their family history was noncontributory.
Since the surgery, the MOP had noticed constant and increasing neck stiffness, specifically with looking up and down but not side to side. She also had noticed swelling behind both ears. She reported no substantial decrease in intake by mouth or decrease in urine or bowel frequency. On review of systems, she reported no fever, vomiting, difficulty breathing, bleeding from the mouth or nose, eye or ear drainage, or rash.
On physical examination, the patient was alert and in no acute distress; active and playful on an electronic device but was notably not moving their head, which was held in a forward-looking position without any signs of trauma. When asked, the child would not flex or extend their neck but would rotate a few degrees from neutral to both sides. Even with moving the electronic device up and down in space, no active neck extension or flexion could be elicited. The examination of the head, eyes, ears, nose, and throat was otherwise only remarkable for palpable and mildly tender postauricular lymph nodes and diffuse erythema in the posterior pharynx. Cardiopulmonary, abdominal, skin, and extremity examinations were unremarkable.
With concern for an infectious process, the physician ordered blood chemistry and hematology tests along with neck radiography. While awaiting the results, the patient was given a weight-based bolus of normal saline, and the home pain regimen was administered. An attempt was made to passively flex and extend the neck as the child slept in their mother’s arms, but the patient immediately awoke and began to cry.
All values of the comprehensive metabolic panel were within normal limits except for a slight elevation in the blood urea nitrogen to 21 mg/dL and glucose to 159 mg/dL. The complete blood count was unrevealing. The computed tomography (CT) scan with contrast of the soft tissues of the neck was limited by motion artifact but showed a head held in axial rotation with soft tissue irregularity in the anterior aspect of the adenoids (Figure 1). There was what appeared to be normal lymphadenopathy in the hypopharynx, but the soft tissues were otherwise unremarkable.
The on-call pediatric otolaryngologist at the hospital where the procedure was performed was paged. On hearing the details of the case, the specialist was concerned for Grisel syndrome and requested to see the patient in their facility. No additional recommendations for care were provided; the mother was updated and agreed to transfer. The patient was comfortable and stable with repeat vitals as follows: heart rate, 86 beats per minute, blood pressure, 99/62, temperature, 98.3 °F, respiratory rate, 20 breaths per minute, and oxygen saturation, 99% on room air.
On arrival at the receiving facility, the emergency team performed a history and physical that revealed no significant changes from the initial evaluation. They then facilitated evaluation by the pediatric otolaryngologist who conducted a more directed physical examination. Decreased active and passive range of motion (ROM) of the neck without rotatory restriction was again noted. They also observed scant fibrinous exudate within the oropharynx and tonsillar fossa, which was normal in the setting of the recent surgery. They recommended additional analgesia with intramuscular ketorolac, weight-based dosing at 1 mg/kg.
With repeat examination after this additional analgesic, ROM of the neck first passive then active had improved. The patient was then discharged to follow up in the coming days with instructions to continue the pain and anti-inflammatory regimen. They were not started on an antibiotic at that time nor were they placed in a cervical collar. At the follow-up, the MOP reported persistence of neck stiffness for a few days initially but then observed slow improvement. By postoperative day 18, the stiffness had resolved. No other follow-up or referrals related to this issue were readily apparent in review of the patient’s health record.
Discussion
Grisel syndrome is the atraumatic rotary subluxation of the atlantoaxial joint, specifically, the atlas (C1 vertebra) rotates to a fixed, nonanatomic position while the axis (C2 vertebra) remains in normal alignment in relation to the remainder of the spinal column. The subluxation occurs in the absence of ligamentous injury but is associated with an increase in ligamentous laxity.4 The atlas is a ring-shaped vertebra with 2 lateral masses connected by anterior and posterior arches; it lacks a spinous process unlike other vertebrae. It articulates with the skull by means of the 2 articular facets on the superior aspect of the lateral masses. Articulation with the axis occurs at 3 sites: 2 articular facets on the inferior portion of the lateral masses of the atlas and a facet for the dens on the posterior portion of the anterior arch. The dens projects superiorly from the body of the axis and is bound posteriorly by the transverse ligament of the atlas.5
The degree of subluxation seen in Grisel syndrome correlates to the disease severity and is classified by the Fielding and Hawkins (FH) system (Table). This system accounts for the distance from the atlas to the dens (atlantodens interval) and the relative asymmetry of the atlantoaxial joint.6 In a normal adult, the upper limit of normal for the atlantodens interval is 3 mm, whereas this distance increases to 4.5 mm for the pediatric population.7 Type I (FH-I) involves rotary subluxation alone without any increase in the atlantodens interval; in FH-II, that interval has increased from normal but to no more than 5 mm. FH-I and FH-II are the most encountered and are not associated with neurologic impairment. In FH-III, neurologic deficits can be present, and the atlantodens interval is increased to > 5 mm. Different from FH-II and FH-III in which anterior dislocation of the atlas with reference to the dens is observed, FH-IV involves a rotary movement of the atlas with concurrent posterior displacement and often involves spinal cord compression.6
Subluxation and displacement without trauma are key components of Grisel syndrome. The 2-hit hypothesis is often used to explain how this can occur, ie, 2 anomalies must be present simultaneously for this condition to develop. First, the laxity of the transverse ligament, the posterior wall of the dens, and other atlantoaxial ligaments must be increased. Second, an asymmetric contraction of the deep erector muscles of the neck either abruptly or more insidiously rotate and dislocate the atlas.8 The pathophysiology is not exactly understood, but the most commonly held hypothesis describes contiguous spread of infection or inflammatory mediators from the pharynx to the ligaments and muscles described.6
Spread could occur via the venous system. The posterior superior pharyngeal region is drained by the periodontoidal venous plexus; the connections here with the pharyngovertebral veins allow for the embolization of infectious or other proinflammatory material to the prevertebral fascia. These emboli induce fasciitis and subsequent aberrant relaxation of the ligaments. In reaction to the inflammation or increased laxity, contiguous muscles of the deep neck contract and freeze the joint out of anatomic alignment.4
The abnormal alignment is apparent grossly as torticollis. Most broadly, torticollis describes an anomalous head posture due to involuntary muscle contractions of neck muscles and specifically describes chin deviation to the side. The antecollis and retrocollis subtypes of torticollis describe forward flexion and backward extension of the neck, respectively.7 Torticollis (broadly) is the most frequently reported condition of those found to have Grisel syndrome (90.7%); other common presenting conditions include neck pain (81.5%) and neck stiffness (31.5%). Fever is found in only 27.8% of cases. Pediatric patients (aged ≤ 12 years) are the most commonly affected, accounting for 87% of cases with an observed 4:1 male to female predominance.7,8 Symptoms begin most often within the first week from the inciting event in 85% of the cases.8 Head and neck surgery precedes up to 67% of cases, and infectious etiologies largely account for the remaining cases.7 Of the postsurgical cases, 55.6% had undergone T&A.8
Although anomalous head posture or neck stiffness following T&A would be of great clinic concern for Grisel syndrome, radiographic studies play a confirmatory role. CT scan is used to evaluate the bony structures, with 3D reconstruction of the cervical spine being most useful to determine the presence and degree of subluxation.8 Magnetic resonance imaging also aids in diagnosis to evaluate ligamentous structures in the area of concern as well as in the evaluation of spinal cord compression.6 Laboratory tests are largely unhelpful in making or excluding the diagnosis.8
If Grisel syndrome is suspected, both the original surgeon (if preceded by surgery) and the neurosurgical team should be consulted. Although no widely adopted guidelines exist for the management of this rare disease, general practice patterns have emerged with the degree of intervention predictably correlating to disease severity. FH-I is usually treated with nonsteroidal anti-inflammatory drugs and muscle relaxants with or without a soft cervical collar. For FH-II, closed reduction and immobilization in a stiff cervical collar is recommended. If no neurologic defect is present, FH-III is treated with bed rest, a period of inline cervical traction, and subsequent immobilization. FH-III with neurologic sequelae and all FH-IV necessitate emergent neurosurgical consultation.4 Surgical intervention is a last resort but is required in up to 24.1% of cases.8
Antibiotic therapy is not routinely given unless clear infectious etiology is identified. No standard antibiotic regimen exists, but coverage for typical upper respiratory pathogens likely suffices. Empiric antibiotic therapy is not recommended for all causes of Grisel syndrome, ie, when the underlying cause is not yet elucidated.6 One case of Grisel syndrome occurring in the setting of cervical osteomyelitis has been described, though, and required prolonged IV antibiotics.3 Physical therapy is recommended as adjunct with no limitations for range of motion save for that of the patient’s individual pain threshold.4
Possibly attributable to waxing and waning ligamentous laxity and strength of the neck muscle contraction, the atlantodens interval and the degree of subluxation can change, making Grisel syndrome dynamic. As such, the FH classification can change, necessitating more or less aggressive therapy. A neurologic evaluation is recommended at least every 2 weeks after the diagnosis is made. If initial identification or recognition of known disease progression is delayed, serious complications can develop. Acutely, spinal cord compression can lead to quadriplegia and death; more insidious complications include reduced neck mobility, dysphonia, and dysphagia.4 As serious, life-threatening complications can arise from Grisel syndrome while good functional outcomes can be achieved with timely and appropriate treatment, the clinician should be inspired to have a high clinical suspicion for this syndrome given the right context.
Conclusions
The patient experienced a desirable outcome with minimal, conservative treatment. As such, the pathology in this case was likely attributed to the mildest form of Grisel syndrome (FH-I). The follow-up was reassuring as well, revealing no worsening or progression of symptoms. The initial evaluation in this case was limited by the inadequacy of the CT scan. Motion artifact in the pharynx prevented the definite exclusion of deep space infection, while the rotation of the head in combination with motion artifact in the cranial-most portions of the vertebral column made determining alignment difficult. One clear axial image, though, does show rotation of the atlas (Figure 2). The uncertainty at the end of our workup prompted surgical consultation, not, admittedly, concern for Grisel syndrome. Awareness of this disease entity is nevertheless important and clinically relevant. Early identification and treatment is associated with decreased morbidity and improvement in long-term functional outcomes.6 Despite its rarity, the clinician should consider Grisel syndrome in any pediatric patient presenting with neck stiffness following the commonly performed T&A.
1. Ramos SD, Mukerji S, Pine HS. Tonsillectomy and adenoidectomy. Pediatr Clin North Am. 2013;60(4):793-807. doi:10.1016/j.pcl.2013.04.015
2. Stoner MJ, Dulaurier M. Pediatric ENT emergencies. Emerg Med Clin North Am. 2013;31(3):795-808. doi:10.1016/j.emc.2013.04.005
3. Leong SC, Karoos PD, Papouliakos SM, et al. Unusual complications of tonsillectomy: a systematic review. Am J Otolaryngol. 2007;28(6):419-422. doi:10.1016/j.amjoto.2006.10.016
4. Fath L, Cebula H, Santin MN, Cocab A, Debrya C, Proustb F. The Grisel’s syndrome: a non-traumatic subluxation of the atlantoaxial joint. Neurochirurgie. 2018;64(4):327-330. doi:10.1016/j.neuchi.2018.02.001
5. Moore K, Agur A, Dalley A. Essential Clinical Anatomy. 5th ed. Baltimore: Lippincott, Williams, and Wilkins; 2015:282-287.
6. Spennato P, Nicosia G, Rapanà A, et al. Grisel syndrome following adenoidectomy: surgical management in a case with delayed diagnosis. World Neurosurg. 2015;84(5):1494.e7-e12.
7. Anania P, Pavone P, Pacetti M, et al. Grisel syndrome in pediatric age: a single-center Italian experience and review of the literature. World Neurosurg. 2019;125:374-382. doi:10.1016/j.wneu.2019.02.035
8. Aldriweesh T, Altheyab F, Alenezi M, et al. Grisel’s syndrome post otolaryngology procedures: a systematic review. Int J Pediatr Otorhinolaryngol. 2020;137:110-125. doi:10.1016/j.ijporl.2020.110225
1. Ramos SD, Mukerji S, Pine HS. Tonsillectomy and adenoidectomy. Pediatr Clin North Am. 2013;60(4):793-807. doi:10.1016/j.pcl.2013.04.015
2. Stoner MJ, Dulaurier M. Pediatric ENT emergencies. Emerg Med Clin North Am. 2013;31(3):795-808. doi:10.1016/j.emc.2013.04.005
3. Leong SC, Karoos PD, Papouliakos SM, et al. Unusual complications of tonsillectomy: a systematic review. Am J Otolaryngol. 2007;28(6):419-422. doi:10.1016/j.amjoto.2006.10.016
4. Fath L, Cebula H, Santin MN, Cocab A, Debrya C, Proustb F. The Grisel’s syndrome: a non-traumatic subluxation of the atlantoaxial joint. Neurochirurgie. 2018;64(4):327-330. doi:10.1016/j.neuchi.2018.02.001
5. Moore K, Agur A, Dalley A. Essential Clinical Anatomy. 5th ed. Baltimore: Lippincott, Williams, and Wilkins; 2015:282-287.
6. Spennato P, Nicosia G, Rapanà A, et al. Grisel syndrome following adenoidectomy: surgical management in a case with delayed diagnosis. World Neurosurg. 2015;84(5):1494.e7-e12.
7. Anania P, Pavone P, Pacetti M, et al. Grisel syndrome in pediatric age: a single-center Italian experience and review of the literature. World Neurosurg. 2019;125:374-382. doi:10.1016/j.wneu.2019.02.035
8. Aldriweesh T, Altheyab F, Alenezi M, et al. Grisel’s syndrome post otolaryngology procedures: a systematic review. Int J Pediatr Otorhinolaryngol. 2020;137:110-125. doi:10.1016/j.ijporl.2020.110225
Machine Learning: the Future of Total Knee Replacement
Total knee replacement (TKR) is one of the most common surgeries worldwide, with > 1 million performed last year. Many patients have seen tremendous benefit from TKR; however, studies have shown that up to 20% of patients are not satisfied with the results of this procedure.1,2 This equates to about 200,000 patients worldwide every year who are dissatisfied. This is a huge concern to patients, surgeons, implant manufacturers, hospitals, and health care payers.
Many attempts to improve satisfaction in TKR have been tried, including computer navigation, minimally invasive surgery, rotating platform prostheses, gender-specific implants, different materials, changes in pain management, and revised postoperative rehabilitation.3-7 However, these efforts show no significant improvement in satisfaction.
The most common method of TKR today involves using a long rod placed through a drill hole in the femur. Standardized cuts on the femur and tibia are made through metal cutting blocks. Only metal mechanical instruments are used to perform the surgery, and all patients are aligned the same. However, anatomic studies have shown that patient anatomy in 3 dimensions (3D) varies widely from patient to patient.8 Our current technique seems far removed from modern engineering, where we now see extensive use of artificial intelligence (AI) to improve outcomes.
Machine learning (ML) is considered a subset of AI that involves the use of various computer algorithms. ML allows the computer to learn and continually improve analysis of data. Large sets of inputs and outputs are used to train the machine to make autonomous recommendations or decisions.9,10
Seven years ago, our team at the Phoenix Veteran Affairs Medical Center in Arizona published a randomized controlled trial evaluating a new, individualized alignment technique for TKR.11 This method used 3D-printed guides made from an MRI of an individual patient’s knee. Instead of aligning all knee replacements the same, each patient was aligned according to their unique anatomy. Compared with the conventional alignment technique, the newer technique showed significant improvement in all outcome scores and range of motion at 2 years postsurgery. There has been a great deal of interest in individualizing TKR, and many articles and techniques have followed.12
Our surgical technique has evolved since publishing our trial. Currently, knee X-rays are digitally templated for each patient. Understanding the patient’s preoperative alignment can then assist in planning a TKR in 3D. A plastic 3D-printed guide is manufactured in Belgium, shipped to the US, sterilized, and used in surgery. These guides fit accurately on the patient’s anatomy and allow precise angles and depth of resection for each surgical bone cut. Our research has shown that these guides are accurate to within 0.5° and 0.5 mm for the bone cuts performed in surgery. After surgery, we track patient-reported outcomes (PROs), which can then be used in ML or logistic regression analysis to determine alignment factors that contribute to the best outcome.13
Soon, use of a robot will take the place of the templating and preplanning, allowing the 3D plan to be immediately produced in surgery by the software installed in the robot.14-16 Each patient’s preoperative alignment can then be immediately compared with the postoperative result, and smartphone technology can allow a patient to input their PRO after the surgery is healed.17
Collecting all this information in a large database can allow ML analyses of the outcomes and individual alignment.14-17 As the factors contributing to the best clinical results are determined, the computer can be programmed to learn how to make the best recommendations for alignment of each patient, which can be incorporated into the robotic platform for each surgery. Also pre- and postoperative factors can be added to the ML platform so we can identify the best preoperative patient parameters, anticoagulation program postoperative rehabilitation program, etc, to help drive higher PROs and satisfaction.
Multiple surgical robots for TKR are now on the market. Orthopedic literature includes ML algorithms to improve outcomes after total hip arthroplasty.18 The EHR can be used to develop models to predict poor outcomes after TKR. Integrating these models into clinical decision support could improve patient selection, education, and satisfaction.19 AI for adult spinal surgery using predictive analytics can help surgeons better inform patients about outcomes after corrective surgery.20,21
With worldwide TKRs expected to exceed 3 million over the next decade, ML using large databases, robotic surgery, and PROs could be key to improving our TKR outcomes.22 This form of AI may reduce the large number of patients currently not satisfied with their knee replacement.
1. Baker PN, van der Meulen JH, Lewsey J, Gregg PJ; National Joint Registry for England and Wales. The role of pain and function in determining patient satisfaction after total knee replacement. Data from the National Joint Registry for England and Wales. J Bone Joint Surg Br. 2007;89(7):893-900. doi:10.1302/0301-620X.89B7.19091
2. Noble PC, Conditt MA, Cook KF, Mathis KB. The John Insall Award: patient expectations affect satisfaction with total knee arthroplasty. Clin Orthop Relat Res. 2006;452:35-43. doi:10.1097/01.blo.0000238825.63648.1e
3. Matziolis G, Krocker D, Weiss U, Tohtz S, Perka C. A prospective, randomized study of computer-assisted and conventional total knee arthroplasty. Three-dimensional evaluation of implant alignment and rotation. J Bone Joint Surg Am. 2007;89(2):236-243. doi:10.2106/JBJS.F.00386
4. Stulberg SD, Yaffe MA, Koo SS. Computer-assisted surgery versus manual total knee arthroplasty: a case-controlled study. J Bone Joint Surg Am. 2006;88(suppl 4):47-54. doi:10.2106/JBJS.F.00698
5. Kalisvaart MM, Pagnano MW, Trousdale RT, Stuart MJ, Hanssen AD. Randomized clinical trial of rotating-platform and fixed-bearing total knee arthroplasty: no clinically detectable differences at five years. J Bone Joint Surg Am. 2012;94(6):481-489. doi:10.2106/JBJS.K.00315
6. Wülker N, Lambermont JP, Sacchetti L, Lazaró JG, Nardi J. A prospective randomized study of minimally invasive total knee arthroplasty compared with conventional surgery. J Bone Joint Surg Am. 2010;92(7):1584-1590. doi:10.2106/JBJS.H.01070
7. Thomsen MG, Husted H, Bencke J, Curtis D, Holm G, Troelsen A. Do we need a gender-specific total knee replacement? A randomised controlled trial comparing a high-flex and a gender-specific posterior design. J Bone Joint Surg Br. 2012;94(6):787-792. doi:10.1302/0301-620X.94B6.28781
8. Eckhoff D, Hogan C, DiMatteo L, Robinson M, Bach J. Difference between the epicondylar and cylindrical axis of the knee. Clin Orthop Relat Res. 2007;461:238-244. doi:10.1097/BLO.0b013e318112416b
9. Martin RK, Ley C, Pareek A, Groll A, Tischer T, Seil R. Artificial intelligence and machine learning: an introduction for orthopaedic surgeons [published online ahead of print, 2021 Sep 15]. Knee Surg Sports Traumatol Arthrosc. 2021;10.1007/s00167-021-06741-2. doi:10.1007/s00167-021-06741-2
10. Helm JM, Swiergosz AM, Haeberle HS, et al. Machine Learning and Artificial Intelligence: Definitions, Applications, and Future Directions. Curr Rev Musculoskelet Med. 2020;13(1):69-76. doi:10.1007/s12178-020-09600-8
11. Dossett HG, Estrada NA, Swartz GJ, LeFevre GW, Kwasman BG. A randomised controlled trial of kinematically and mechanically aligned total knee replacements: two-year clinical results. Bone Joint J. 2014;96-B(7):907-913. doi:10.1302/0301-620X.96B7.32812
12. Rivière C, Iranpour F, Auvinet E, et al. Alignment options for total knee arthroplasty: a systematic review. Orthop Traumatol Surg Res. 2017;103(7):1047-1056. doi:10.1016/j.otsr.2017.07.010
13. Dossett HG. High reliability in total knee replacement surgery: is it possible? Orthop Proc. 2018;95-B(suppl 34):292-293.
14. Schock J, Truhn D, Abrar DB, et al. Automated analysis of alignment in long-leg radiographs by using a fully automated support system based on artificial intelligence. Radiol: Artif Intell. Dec 23, 2020;3(2). doi:10.1148/ryai.2020200198
15. Cabitza F, Locoro A, Banfi G. Machine learning in orthopedics: a literature review. Front Bioeng Biotechnol. 2018;6:75. Published 2018 Jun 27. doi:10.3389/fbioe.2018.00075
16. von Schacky CE, Wilhelm NJ, Schäfer VS, et al. Multitask deep learning for segmentation and classification of primary bone tumors on radiographs. Radiology. 2021;301(2):398-406. doi:10.1148/radiol.2021204531
17. Myers TG, Ramkumar PN, Ricciardi BF, Urish KL, Kipper J, Ketonis C. Artificial intelligence and orthopaedics: an introduction for clinicians. J Bone Joint Surg Am. 2020;102(9):830-840. doi:10.2106/JBJS.19.01128
18. Kunze KN, Karhade AV, Sadauskas AJ, Schwab JH, Levine BR. Development of machine learning algorithms to predict clinically meaningful improvement for the patient-reported health state after total hip arthroplasty. J Arthroplasty. 2020;35(8):2119-2123. doi:10.1016/j.arth.2020.03.019
19. Harris AHS, Kuo AC, Bowe TR, Manfredi L, Lalani NF, Giori NJ. Can machine learning methods produce accurate and easy-to-use preoperative prediction models of one-year improvements in pain and functioning after knee arthroplasty? J Arthroplasty. 2021;36(1):112-117.e6. doi:10.1016/j.arth.2020.07.026
20. Rasouli JJ, Shao J, Neifert S, et al. Artificial intelligence and robotics in spine surgery. Global Spine J. 2021;11(4):556-564. doi:10.1177/2192568220915718
21. Joshi RS, Haddad AF, Lau D, Ames CP. Artificial intelligence for adult spinal deformity. Neurospine. 2019;16(4):686-694. doi:10.14245/ns.1938414.207
22. Kurtz S, Ong K, Lau E, Mowat F, Halpern M. Projections of primary and revision hip and knee arthroplasty in the United States from 2005 to 2030. J Bone Joint Surg Am. 2007;89(4):780-785. doi:10.2106/JBJS.F.00222
Total knee replacement (TKR) is one of the most common surgeries worldwide, with > 1 million performed last year. Many patients have seen tremendous benefit from TKR; however, studies have shown that up to 20% of patients are not satisfied with the results of this procedure.1,2 This equates to about 200,000 patients worldwide every year who are dissatisfied. This is a huge concern to patients, surgeons, implant manufacturers, hospitals, and health care payers.
Many attempts to improve satisfaction in TKR have been tried, including computer navigation, minimally invasive surgery, rotating platform prostheses, gender-specific implants, different materials, changes in pain management, and revised postoperative rehabilitation.3-7 However, these efforts show no significant improvement in satisfaction.
The most common method of TKR today involves using a long rod placed through a drill hole in the femur. Standardized cuts on the femur and tibia are made through metal cutting blocks. Only metal mechanical instruments are used to perform the surgery, and all patients are aligned the same. However, anatomic studies have shown that patient anatomy in 3 dimensions (3D) varies widely from patient to patient.8 Our current technique seems far removed from modern engineering, where we now see extensive use of artificial intelligence (AI) to improve outcomes.
Machine learning (ML) is considered a subset of AI that involves the use of various computer algorithms. ML allows the computer to learn and continually improve analysis of data. Large sets of inputs and outputs are used to train the machine to make autonomous recommendations or decisions.9,10
Seven years ago, our team at the Phoenix Veteran Affairs Medical Center in Arizona published a randomized controlled trial evaluating a new, individualized alignment technique for TKR.11 This method used 3D-printed guides made from an MRI of an individual patient’s knee. Instead of aligning all knee replacements the same, each patient was aligned according to their unique anatomy. Compared with the conventional alignment technique, the newer technique showed significant improvement in all outcome scores and range of motion at 2 years postsurgery. There has been a great deal of interest in individualizing TKR, and many articles and techniques have followed.12
Our surgical technique has evolved since publishing our trial. Currently, knee X-rays are digitally templated for each patient. Understanding the patient’s preoperative alignment can then assist in planning a TKR in 3D. A plastic 3D-printed guide is manufactured in Belgium, shipped to the US, sterilized, and used in surgery. These guides fit accurately on the patient’s anatomy and allow precise angles and depth of resection for each surgical bone cut. Our research has shown that these guides are accurate to within 0.5° and 0.5 mm for the bone cuts performed in surgery. After surgery, we track patient-reported outcomes (PROs), which can then be used in ML or logistic regression analysis to determine alignment factors that contribute to the best outcome.13
Soon, use of a robot will take the place of the templating and preplanning, allowing the 3D plan to be immediately produced in surgery by the software installed in the robot.14-16 Each patient’s preoperative alignment can then be immediately compared with the postoperative result, and smartphone technology can allow a patient to input their PRO after the surgery is healed.17
Collecting all this information in a large database can allow ML analyses of the outcomes and individual alignment.14-17 As the factors contributing to the best clinical results are determined, the computer can be programmed to learn how to make the best recommendations for alignment of each patient, which can be incorporated into the robotic platform for each surgery. Also pre- and postoperative factors can be added to the ML platform so we can identify the best preoperative patient parameters, anticoagulation program postoperative rehabilitation program, etc, to help drive higher PROs and satisfaction.
Multiple surgical robots for TKR are now on the market. Orthopedic literature includes ML algorithms to improve outcomes after total hip arthroplasty.18 The EHR can be used to develop models to predict poor outcomes after TKR. Integrating these models into clinical decision support could improve patient selection, education, and satisfaction.19 AI for adult spinal surgery using predictive analytics can help surgeons better inform patients about outcomes after corrective surgery.20,21
With worldwide TKRs expected to exceed 3 million over the next decade, ML using large databases, robotic surgery, and PROs could be key to improving our TKR outcomes.22 This form of AI may reduce the large number of patients currently not satisfied with their knee replacement.
Total knee replacement (TKR) is one of the most common surgeries worldwide, with > 1 million performed last year. Many patients have seen tremendous benefit from TKR; however, studies have shown that up to 20% of patients are not satisfied with the results of this procedure.1,2 This equates to about 200,000 patients worldwide every year who are dissatisfied. This is a huge concern to patients, surgeons, implant manufacturers, hospitals, and health care payers.
Many attempts to improve satisfaction in TKR have been tried, including computer navigation, minimally invasive surgery, rotating platform prostheses, gender-specific implants, different materials, changes in pain management, and revised postoperative rehabilitation.3-7 However, these efforts show no significant improvement in satisfaction.
The most common method of TKR today involves using a long rod placed through a drill hole in the femur. Standardized cuts on the femur and tibia are made through metal cutting blocks. Only metal mechanical instruments are used to perform the surgery, and all patients are aligned the same. However, anatomic studies have shown that patient anatomy in 3 dimensions (3D) varies widely from patient to patient.8 Our current technique seems far removed from modern engineering, where we now see extensive use of artificial intelligence (AI) to improve outcomes.
Machine learning (ML) is considered a subset of AI that involves the use of various computer algorithms. ML allows the computer to learn and continually improve analysis of data. Large sets of inputs and outputs are used to train the machine to make autonomous recommendations or decisions.9,10
Seven years ago, our team at the Phoenix Veteran Affairs Medical Center in Arizona published a randomized controlled trial evaluating a new, individualized alignment technique for TKR.11 This method used 3D-printed guides made from an MRI of an individual patient’s knee. Instead of aligning all knee replacements the same, each patient was aligned according to their unique anatomy. Compared with the conventional alignment technique, the newer technique showed significant improvement in all outcome scores and range of motion at 2 years postsurgery. There has been a great deal of interest in individualizing TKR, and many articles and techniques have followed.12
Our surgical technique has evolved since publishing our trial. Currently, knee X-rays are digitally templated for each patient. Understanding the patient’s preoperative alignment can then assist in planning a TKR in 3D. A plastic 3D-printed guide is manufactured in Belgium, shipped to the US, sterilized, and used in surgery. These guides fit accurately on the patient’s anatomy and allow precise angles and depth of resection for each surgical bone cut. Our research has shown that these guides are accurate to within 0.5° and 0.5 mm for the bone cuts performed in surgery. After surgery, we track patient-reported outcomes (PROs), which can then be used in ML or logistic regression analysis to determine alignment factors that contribute to the best outcome.13
Soon, use of a robot will take the place of the templating and preplanning, allowing the 3D plan to be immediately produced in surgery by the software installed in the robot.14-16 Each patient’s preoperative alignment can then be immediately compared with the postoperative result, and smartphone technology can allow a patient to input their PRO after the surgery is healed.17
Collecting all this information in a large database can allow ML analyses of the outcomes and individual alignment.14-17 As the factors contributing to the best clinical results are determined, the computer can be programmed to learn how to make the best recommendations for alignment of each patient, which can be incorporated into the robotic platform for each surgery. Also pre- and postoperative factors can be added to the ML platform so we can identify the best preoperative patient parameters, anticoagulation program postoperative rehabilitation program, etc, to help drive higher PROs and satisfaction.
Multiple surgical robots for TKR are now on the market. Orthopedic literature includes ML algorithms to improve outcomes after total hip arthroplasty.18 The EHR can be used to develop models to predict poor outcomes after TKR. Integrating these models into clinical decision support could improve patient selection, education, and satisfaction.19 AI for adult spinal surgery using predictive analytics can help surgeons better inform patients about outcomes after corrective surgery.20,21
With worldwide TKRs expected to exceed 3 million over the next decade, ML using large databases, robotic surgery, and PROs could be key to improving our TKR outcomes.22 This form of AI may reduce the large number of patients currently not satisfied with their knee replacement.
1. Baker PN, van der Meulen JH, Lewsey J, Gregg PJ; National Joint Registry for England and Wales. The role of pain and function in determining patient satisfaction after total knee replacement. Data from the National Joint Registry for England and Wales. J Bone Joint Surg Br. 2007;89(7):893-900. doi:10.1302/0301-620X.89B7.19091
2. Noble PC, Conditt MA, Cook KF, Mathis KB. The John Insall Award: patient expectations affect satisfaction with total knee arthroplasty. Clin Orthop Relat Res. 2006;452:35-43. doi:10.1097/01.blo.0000238825.63648.1e
3. Matziolis G, Krocker D, Weiss U, Tohtz S, Perka C. A prospective, randomized study of computer-assisted and conventional total knee arthroplasty. Three-dimensional evaluation of implant alignment and rotation. J Bone Joint Surg Am. 2007;89(2):236-243. doi:10.2106/JBJS.F.00386
4. Stulberg SD, Yaffe MA, Koo SS. Computer-assisted surgery versus manual total knee arthroplasty: a case-controlled study. J Bone Joint Surg Am. 2006;88(suppl 4):47-54. doi:10.2106/JBJS.F.00698
5. Kalisvaart MM, Pagnano MW, Trousdale RT, Stuart MJ, Hanssen AD. Randomized clinical trial of rotating-platform and fixed-bearing total knee arthroplasty: no clinically detectable differences at five years. J Bone Joint Surg Am. 2012;94(6):481-489. doi:10.2106/JBJS.K.00315
6. Wülker N, Lambermont JP, Sacchetti L, Lazaró JG, Nardi J. A prospective randomized study of minimally invasive total knee arthroplasty compared with conventional surgery. J Bone Joint Surg Am. 2010;92(7):1584-1590. doi:10.2106/JBJS.H.01070
7. Thomsen MG, Husted H, Bencke J, Curtis D, Holm G, Troelsen A. Do we need a gender-specific total knee replacement? A randomised controlled trial comparing a high-flex and a gender-specific posterior design. J Bone Joint Surg Br. 2012;94(6):787-792. doi:10.1302/0301-620X.94B6.28781
8. Eckhoff D, Hogan C, DiMatteo L, Robinson M, Bach J. Difference between the epicondylar and cylindrical axis of the knee. Clin Orthop Relat Res. 2007;461:238-244. doi:10.1097/BLO.0b013e318112416b
9. Martin RK, Ley C, Pareek A, Groll A, Tischer T, Seil R. Artificial intelligence and machine learning: an introduction for orthopaedic surgeons [published online ahead of print, 2021 Sep 15]. Knee Surg Sports Traumatol Arthrosc. 2021;10.1007/s00167-021-06741-2. doi:10.1007/s00167-021-06741-2
10. Helm JM, Swiergosz AM, Haeberle HS, et al. Machine Learning and Artificial Intelligence: Definitions, Applications, and Future Directions. Curr Rev Musculoskelet Med. 2020;13(1):69-76. doi:10.1007/s12178-020-09600-8
11. Dossett HG, Estrada NA, Swartz GJ, LeFevre GW, Kwasman BG. A randomised controlled trial of kinematically and mechanically aligned total knee replacements: two-year clinical results. Bone Joint J. 2014;96-B(7):907-913. doi:10.1302/0301-620X.96B7.32812
12. Rivière C, Iranpour F, Auvinet E, et al. Alignment options for total knee arthroplasty: a systematic review. Orthop Traumatol Surg Res. 2017;103(7):1047-1056. doi:10.1016/j.otsr.2017.07.010
13. Dossett HG. High reliability in total knee replacement surgery: is it possible? Orthop Proc. 2018;95-B(suppl 34):292-293.
14. Schock J, Truhn D, Abrar DB, et al. Automated analysis of alignment in long-leg radiographs by using a fully automated support system based on artificial intelligence. Radiol: Artif Intell. Dec 23, 2020;3(2). doi:10.1148/ryai.2020200198
15. Cabitza F, Locoro A, Banfi G. Machine learning in orthopedics: a literature review. Front Bioeng Biotechnol. 2018;6:75. Published 2018 Jun 27. doi:10.3389/fbioe.2018.00075
16. von Schacky CE, Wilhelm NJ, Schäfer VS, et al. Multitask deep learning for segmentation and classification of primary bone tumors on radiographs. Radiology. 2021;301(2):398-406. doi:10.1148/radiol.2021204531
17. Myers TG, Ramkumar PN, Ricciardi BF, Urish KL, Kipper J, Ketonis C. Artificial intelligence and orthopaedics: an introduction for clinicians. J Bone Joint Surg Am. 2020;102(9):830-840. doi:10.2106/JBJS.19.01128
18. Kunze KN, Karhade AV, Sadauskas AJ, Schwab JH, Levine BR. Development of machine learning algorithms to predict clinically meaningful improvement for the patient-reported health state after total hip arthroplasty. J Arthroplasty. 2020;35(8):2119-2123. doi:10.1016/j.arth.2020.03.019
19. Harris AHS, Kuo AC, Bowe TR, Manfredi L, Lalani NF, Giori NJ. Can machine learning methods produce accurate and easy-to-use preoperative prediction models of one-year improvements in pain and functioning after knee arthroplasty? J Arthroplasty. 2021;36(1):112-117.e6. doi:10.1016/j.arth.2020.07.026
20. Rasouli JJ, Shao J, Neifert S, et al. Artificial intelligence and robotics in spine surgery. Global Spine J. 2021;11(4):556-564. doi:10.1177/2192568220915718
21. Joshi RS, Haddad AF, Lau D, Ames CP. Artificial intelligence for adult spinal deformity. Neurospine. 2019;16(4):686-694. doi:10.14245/ns.1938414.207
22. Kurtz S, Ong K, Lau E, Mowat F, Halpern M. Projections of primary and revision hip and knee arthroplasty in the United States from 2005 to 2030. J Bone Joint Surg Am. 2007;89(4):780-785. doi:10.2106/JBJS.F.00222
1. Baker PN, van der Meulen JH, Lewsey J, Gregg PJ; National Joint Registry for England and Wales. The role of pain and function in determining patient satisfaction after total knee replacement. Data from the National Joint Registry for England and Wales. J Bone Joint Surg Br. 2007;89(7):893-900. doi:10.1302/0301-620X.89B7.19091
2. Noble PC, Conditt MA, Cook KF, Mathis KB. The John Insall Award: patient expectations affect satisfaction with total knee arthroplasty. Clin Orthop Relat Res. 2006;452:35-43. doi:10.1097/01.blo.0000238825.63648.1e
3. Matziolis G, Krocker D, Weiss U, Tohtz S, Perka C. A prospective, randomized study of computer-assisted and conventional total knee arthroplasty. Three-dimensional evaluation of implant alignment and rotation. J Bone Joint Surg Am. 2007;89(2):236-243. doi:10.2106/JBJS.F.00386
4. Stulberg SD, Yaffe MA, Koo SS. Computer-assisted surgery versus manual total knee arthroplasty: a case-controlled study. J Bone Joint Surg Am. 2006;88(suppl 4):47-54. doi:10.2106/JBJS.F.00698
5. Kalisvaart MM, Pagnano MW, Trousdale RT, Stuart MJ, Hanssen AD. Randomized clinical trial of rotating-platform and fixed-bearing total knee arthroplasty: no clinically detectable differences at five years. J Bone Joint Surg Am. 2012;94(6):481-489. doi:10.2106/JBJS.K.00315
6. Wülker N, Lambermont JP, Sacchetti L, Lazaró JG, Nardi J. A prospective randomized study of minimally invasive total knee arthroplasty compared with conventional surgery. J Bone Joint Surg Am. 2010;92(7):1584-1590. doi:10.2106/JBJS.H.01070
7. Thomsen MG, Husted H, Bencke J, Curtis D, Holm G, Troelsen A. Do we need a gender-specific total knee replacement? A randomised controlled trial comparing a high-flex and a gender-specific posterior design. J Bone Joint Surg Br. 2012;94(6):787-792. doi:10.1302/0301-620X.94B6.28781
8. Eckhoff D, Hogan C, DiMatteo L, Robinson M, Bach J. Difference between the epicondylar and cylindrical axis of the knee. Clin Orthop Relat Res. 2007;461:238-244. doi:10.1097/BLO.0b013e318112416b
9. Martin RK, Ley C, Pareek A, Groll A, Tischer T, Seil R. Artificial intelligence and machine learning: an introduction for orthopaedic surgeons [published online ahead of print, 2021 Sep 15]. Knee Surg Sports Traumatol Arthrosc. 2021;10.1007/s00167-021-06741-2. doi:10.1007/s00167-021-06741-2
10. Helm JM, Swiergosz AM, Haeberle HS, et al. Machine Learning and Artificial Intelligence: Definitions, Applications, and Future Directions. Curr Rev Musculoskelet Med. 2020;13(1):69-76. doi:10.1007/s12178-020-09600-8
11. Dossett HG, Estrada NA, Swartz GJ, LeFevre GW, Kwasman BG. A randomised controlled trial of kinematically and mechanically aligned total knee replacements: two-year clinical results. Bone Joint J. 2014;96-B(7):907-913. doi:10.1302/0301-620X.96B7.32812
12. Rivière C, Iranpour F, Auvinet E, et al. Alignment options for total knee arthroplasty: a systematic review. Orthop Traumatol Surg Res. 2017;103(7):1047-1056. doi:10.1016/j.otsr.2017.07.010
13. Dossett HG. High reliability in total knee replacement surgery: is it possible? Orthop Proc. 2018;95-B(suppl 34):292-293.
14. Schock J, Truhn D, Abrar DB, et al. Automated analysis of alignment in long-leg radiographs by using a fully automated support system based on artificial intelligence. Radiol: Artif Intell. Dec 23, 2020;3(2). doi:10.1148/ryai.2020200198
15. Cabitza F, Locoro A, Banfi G. Machine learning in orthopedics: a literature review. Front Bioeng Biotechnol. 2018;6:75. Published 2018 Jun 27. doi:10.3389/fbioe.2018.00075
16. von Schacky CE, Wilhelm NJ, Schäfer VS, et al. Multitask deep learning for segmentation and classification of primary bone tumors on radiographs. Radiology. 2021;301(2):398-406. doi:10.1148/radiol.2021204531
17. Myers TG, Ramkumar PN, Ricciardi BF, Urish KL, Kipper J, Ketonis C. Artificial intelligence and orthopaedics: an introduction for clinicians. J Bone Joint Surg Am. 2020;102(9):830-840. doi:10.2106/JBJS.19.01128
18. Kunze KN, Karhade AV, Sadauskas AJ, Schwab JH, Levine BR. Development of machine learning algorithms to predict clinically meaningful improvement for the patient-reported health state after total hip arthroplasty. J Arthroplasty. 2020;35(8):2119-2123. doi:10.1016/j.arth.2020.03.019
19. Harris AHS, Kuo AC, Bowe TR, Manfredi L, Lalani NF, Giori NJ. Can machine learning methods produce accurate and easy-to-use preoperative prediction models of one-year improvements in pain and functioning after knee arthroplasty? J Arthroplasty. 2021;36(1):112-117.e6. doi:10.1016/j.arth.2020.07.026
20. Rasouli JJ, Shao J, Neifert S, et al. Artificial intelligence and robotics in spine surgery. Global Spine J. 2021;11(4):556-564. doi:10.1177/2192568220915718
21. Joshi RS, Haddad AF, Lau D, Ames CP. Artificial intelligence for adult spinal deformity. Neurospine. 2019;16(4):686-694. doi:10.14245/ns.1938414.207
22. Kurtz S, Ong K, Lau E, Mowat F, Halpern M. Projections of primary and revision hip and knee arthroplasty in the United States from 2005 to 2030. J Bone Joint Surg Am. 2007;89(4):780-785. doi:10.2106/JBJS.F.00222
Naloxone Dispensing in Patients at Risk for Opioid Overdose After Total Knee Arthroplasty Within the Veterans Health Administration
Opioid overdose is a major public health challenge, with recent reports estimating 41 deaths per day in the United States from prescription opioid overdose.1,2 Prescribing naloxone has increasingly been advocated to reduce the risk of opioid overdose for patients identified as high risk. Naloxone distribution has been shown to decrease the incidence of opioid overdoses in the general population.3,4 The Centers for Disease Control and Prevention (CDC) Guideline for Prescribing Opioids for Chronic Pain recommends considering naloxone prescription for patients with a history of overdose or substance use disorder, opioid dosages ≥ 50 morphine equivalent daily dose (MEDD), and concurrent use of benzodiazepines.5
Although the CDC guidelines are intended for primary care clinicians in outpatient settings, naloxone prescribing is also relevant in the postsurgical setting.5 Many surgical patients are at risk for opioid overdose and data from the Veterans Health Administration (VHA) has shown that risk of opioid overdose is 11-fold higher in the 30 days following discharge from a surgical admission, when compared with the subsequent calendar year.6,7 This likely occurs due to new prescriptions or escalated doses of opioids following surgery. Overdose risk may be particularly relevant to orthopedic surgery as postoperative opioids are commonly prescribed.8 Patients undergoing total knee arthroplasty (TKA) may represent a vulnerable population to overdose as it is one of the most commonly performed surgeries for the treatment of chronic pain, and is frequently performed in older adults with medical comorbidities.9,10
Identifying patients at high risk for opioid overdose is important for targeted naloxone dispensing.5 A risk index for overdose or serious opioid-induced respiratory depression (RIOSORD) tool has been developed and validated in veteran and other populations to identify such patients.11 The RIOSORD tool classifies patients by risk level (1-10) and predicts probability of overdose or serious opioid-induced respiratory depression (OSORD). A patient’s level of risk is based on a weighted combination of the 15 independent risk factors most highly associated with OSORD, including comorbid conditions, prescription drug use, and health care utilization.12 Using the RIOSORD tool, the VHA Opioid Education and Naloxone Distribution (OEND) program is a risk mitigation initiative that aims to decrease opioid-related overdose morbidity and mortality. This is achieved via opioid overdose education for prevention, recognition, and response and includes outpatient naloxone prescription.13,14
Despite the comprehensive OEND program, there exists very little data to guide postsurgical naloxone prescribing. The prevalence of known risk factors for overdose in surgical patients remains unknown, as does the prevalence of perioperative naloxone distribution. Understanding overdose risk factors and naloxone prescribing patterns in surgical patients may identify potential targets for OEND efforts. This study retrospectively estimated RIOSORD scores for TKA patients between 2013 to 2016 and described naloxone distribution based on RIOSORD scores and risk factors.
Methods
We identified patients who had undergone primary TKA at VHA hospitals using Current Procedural Terminology (CPT), International Classification of Diseases, Ninth Revision (ICD-9) procedure codes, and data extracted from the VHA Corporate Data Warehouse (CDW) of electronic health records (EHRs). Our study was granted approval with exemption from informed consent by the Durham Veteran Affairs Healthcare System Institutional Review Board.
This retrospective cohort study included all veterans who underwent elective primary TKA from January 1, 2013 through December 31, 2016. We excluded patients who died before discharge.
Outcomes
Our primary outcome was being dispensed an outpatient naloxone prescription following TKA. Naloxone dispensing was identified by examining CDW outpatient pharmacy records with a final dispense date from 1 year before surgery through 7 days after discharge following TKA. To exclude naloxone administration that may have been given in a clinic, prescription data included only records with an outpatient prescription copay. Naloxone dispensing in the year before surgery was chosen to estimate likely preoperative possession of naloxone which could be available in the postoperative period. Naloxone dispensing until 7 days after discharge was chosen to identify any new dispensing that would be available in the postoperative period. These outcomes were examined over the study time frame on an annual basis.
Patient Factors
Demographic variables included age, sex, and race/ethnicity. Independent risk factors for overdose from RIOSORD were identified for each patient.15 These risk factors included comorbidities (opioid use disorder, schizophrenia, bipolar disorder, liver disease, chronic kidney disease, sleep apnea, or lung disease) and prescription drug use (use of opioids, benzodiazepines, long-acting opioids, ≥ 50 MEDD or ≥ 100 MEDD). ICD-9 and ICD-10 diagnosis codes were used to identify comorbidities. Risk classes on day of surgery were identified using a RIOSORD algorithm code. Consistent with the display of RIOSORD risk classes on the VHA Academic Detailing Service OEND risk report, patients were grouped into 3 groups based on their RIOSORD score: classes 1 to 4 (low risk), 5 to 7 (moderate risk), and 8 to 10 (high risk).
Descriptive statistics were used to summarize data on patient demographics, RIOSORD risk factors, overdose events, and naloxone dispensing over time.
Results
The study cohort included 38,011 veterans who underwent primary TKA in the VHA between January 1, 2013 and December 30, 2016. In this cohort, the mean age was 65 years, 93% were male, and 77% were White patients (Table 1). The most common comorbidities were lung disease in 9170 (24.1%) patients, sleep apnea in 6630 (17.4%) patients, chronic kidney disease in 4036 (10.6%) patients, liver disease in 2822 (7.4%) patients, and bipolar disorder in 1748 (4.6%) patients.
In 2013, 63.1% of patients presenting for surgery were actively prescribed opioids. By 2016, this decreased to 50.5%. Benzodiazepine use decreased from 13.2 to 8.8% and long-acting opioid use decreased from 8.5 to 5.8% over the same period. Patients taking ≥ 50 MEDD decreased from 8.0 to 5.3% and patients taking ≥ 100 MEDD decreased from 3.3 to 2.2%. The prevalence of moderate-risk patients decreased from 2.5 to 1.6% and high-risk patients decreased from 0.8 to 0.6% (Figure 1). Cumulatively, the prevalence of presenting with either moderate or high risk of overdose decreased from 3.3 to 2.2% between 2013 to 2016.
Naloxone Dispensing
In 2013, naloxone was not dispensed to any patients at moderate or high risk for overdose between 365 days prior to surgery until 7 days after discharge (Table 2 and Figure 2). Low-risk group naloxone dispensing increased to 2 (0.0%) in 2014, to 13 (0.1%), in 2015, and to 86 (0.9%) in 2016. Moderate-risk group naloxone dispensing remained at 0 (0.0%) in 2014, but increased to 8 (3.5%) in 2015, and to 18 (10.9%) in 2016. High-risk group naloxone dispensing remained at 0 (0.0%) in 2014, but increased to 5 (5.8%) in 2015, and to 8 (12.7%) in 2016 (Figure 3).
Discussion
Our data demonstrate that patients presenting for TKA between 2013 and 2016 routinely had individual risk factors for overdose related to either prescription drug use or comorbidities. We also show that, although the number of patients at moderate and high risk for opioid overdose is decreasing, 2.2% of TKA patients remain at moderate or high risk for opioid overdose based on a weighted combination of these individual risk factors using RIOSORD. As demand for primary TKA is projected to grow to 3.5 million procedures by 2030, using prevalence from 2016, we estimate that 76,560 patients may present for TKA across the US with moderate or high risk for opioid overdose.9 Following discharge, this risk may be even higher as this estimate does not yet account for postoperative opioid use. We demonstrate that through a VHA OEND initiative, naloxone distribution increased and appeared to be targeted to those most at risk using a simple validated tool like RIOSORD.
Presence of an individual risk factor for overdose was present in as many as 63.1% of patients presenting for TKA, as was seen in 2013 with preoperative opioid use. The 3 highest scoring prescription use–related risk factors in RIOSORD are use of opioids ≥ 100 MEDD (16 points), ≥ 50 MEDD (9 points), and long-acting formulations (9 points). All 3 decreased in prevalence over the study period but by 2016 were still seen in 2.2% for ≥ 100 MEDD, 5.3% for ≥ 50 MEDD, and 5.8% for long-acting opioids. This decrease was not surprising given implementation of a VHA-wide opioid safety initiative and the OEND program, but this could also be related to changes in patient selection for surgery in the context of increased awareness of the opioid epidemic. Despite the trend toward safer opioid prescribing, by 2016 over half of patients (50.5%) who presented for TKA were already taking opioids, with 10.6% (543 of 5127) on doses ≥ 50 MEDD.
We observed a decrease in RIOSORD risk each year, consistent with decreasing prescription-related risk factors over time. This was most obvious in the moderate-risk group. It is unclear why a similar decrease was not as obvious in the high-risk group, but this in part may be due to the already low numbers of patients in the high-risk group. This may also represent the high-risk group being somewhat resistant to the initiatives that shifted moderate-risk patients to the low-risk group. There were proportionately more patients in the moderate- and high-risk groups in the original RIOSORD population than in our surgical population, which may be attributed to the fewer comorbidities seen in our surgical population, as well as the higher opioid-prescribing patterns seen prior to the VA OEND initiative.12
Naloxone prescribing was rare prior to the OEND initiative and increased from 2013 to 2016. Increases were most marked in those in moderate- and high-risk groups, although naloxone prescribing also increased among the low-risk group. Integration of RIOSORD stratification into the OEND initiative likely played a role in targeting increased access to naloxone among those at highest risk of overdose. Naloxone dispensing increased for every group, although a significant proportion of moderate- and high-risk patients, 89.1% and 87.3%, respectively, were still not dispensed naloxone by 2016. Moreover, our estimates of perioperative naloxone access were likely an overestimate by including patients dispensed naloxone up to 1 year before surgery until 7 days after surgery. The aim was to include patients who may not have been prescribed naloxone postoperatively because of an existing naloxone prescription at home. Perioperative naloxone access estimates would have been even lower if a narrower window had been used to approximate perioperative access. This identifies an important gap between those who may benefit from naloxone dispensing and those who received naloxone. This in part may be because OEND has not been implemented as routinely in surgical settings as other settings (eg, primary care). OEND efforts may more effectively increase naloxone prescribing among surgical patients if these efforts were targeted at surgical and anesthesia departments. Given that the Comprehensive Addiction and Recovery Act of 2016 requires an assessment of patient risk prior to opioid prescribing and VHA efforts to increase utilization of tools like the Stratification Tool for Opioid Risk Mitigation (STORM), which estimates patient risk when initiating an opioid prescription and includes naloxone as one of many risk mitigation strategies, we anticipate that rates of naloxone prescribing will increase over time.
Limitations
Our study captures a large number of patients across VHA hospitals of varying size nationwide, including a mix of those with and without academic medical center affiliations. This veteran population may not represent the US commercially insured population (CIP). Zedler and colleagues highlighted the differences in prevalence of individual risk factors: notably, the CIP had a substantially higher proportion of females and younger patients.11 VHA had a greater prevalence of common chronic conditions associated with older age. The frequency of opioid dependence was similar among CIP and VHA. However, substance abuse and nonopioid substance dependence diagnoses were 4-fold more frequent among VHA controls as CIP controls. Prescribing of all opioids, except morphine and methadone, was substantially greater in CIP than in VHA.11 Despite a difference in individual risk factors, a CIP-specific RIOSORD has been validated and can be used outside of the VHA to obviate the limitations of the VHA-specific RIOSORD.11
Other limitations include our estimation of naloxone access. We do not know whether naloxone was administered or have a reliable estimate of overdose incidence in this postoperative TKA population. Also, it is important to note that RIOSORD was not developed for surgical patients. The use of RIOSORD in a postoperative population likely underestimates risk of opioid overdose due to the frequent prescriptions of new opioids or escalation of existing MEDD to the postoperative patient. Our study was also retrospective in nature and reliant on accurate coding of patient risk factors. It is possible that comorbidities were not accurately identified by EHR and therefore subject to inconsistency.
Conclusions
Veterans presenting for TKA routinely have risk factors for opioid overdose. We observed a trend toward decreasing overdose risk which coincided with the Opioid Safety and OEND initiatives within the VHA. We also observed an increase in naloxone prescription for moderate- and high-risk patients undergoing TKA, although most of these patients still did not receive naloxone as of 2016. More research is needed to refine and validate the RIOSORD score for surgical populations. Expanding initiatives such as OEND to include surgical patients presents an opportunity to improve access to naloxone for postoperative patients that may help reduce opioid overdose in this population.
1. Rudd RA, Seth P, David F, Scholl L. Increases in drug and opioid-involved overdose deaths - United States, 2010-2015. MMWR Morb Mortal Wkly Rep. 2016;65(50-51):1445-1452. Published 2016 Dec 30. doi:10.15585/mmwr.mm655051e1
2. Wilson N, Kariisa M, Seth P, Smith H, Davis NL. Drug and opioid-involved overdose deaths - United States, 2017-2018. MMWR Morb Mortal Wkly Rep. 2020;69(11):290-297. doi:10.15585/mmwr.mm6911a4
3. Walley AY, Xuan Z, Hackman HH, et al. Opioid overdose rates and implementation of overdose education and nasal naloxone distribution in Massachusetts: interrupted time series analysis. BMJ. Jan 30 2013;346:f174. doi:10.1136/bmj.f174
4. McClellan C, Lambdin BH, Ali MM, et al. Opioid-overdose laws association with opioid use and overdose mortality. Addict Behav. 2018;86:90-95. doi:10.1016/j.addbeh.2018.03.014
5. Dowell D, Haegerich TM, Chou R. CDC Guideline for prescribing opioids for chronic pain--United States, 2016. JAMA. 2016;315(15):1624-1645. doi:10.1001/jama.2016.1464
6. Brat GA, Agniel D, Beam A, et al. Postsurgical prescriptions for opioid naive patients and association with overdose and misuse: retrospective cohort study. BMJ. 2018;360:j5790. Published 2018 Jan 17. doi:10.1136/bmj.j5790
7. Mudumbai SC, Lewis ET, Oliva EM, et al. Overdose risk associated with opioid use upon hospital discharge in Veterans Health Administration surgical patients. Pain Med. 2019;20(5):1020-1031. doi:10.1093/pm/pny150
8. Hsia HL, Takemoto S, van de Ven T, et al. Acute pain is associated with chronic opioid use after total knee arthroplasty. Reg Anesth Pain Med. 2018;43(7):705-711. doi:10.1097/AAP.0000000000000831
9. Kurtz S, Ong K, Lau E, Mowat F, Halpern M. Projections of primary and revision hip and knee arthroplasty in the United States from 2005 to 2030. J Bone Joint Surg Am. 2007;89(4):780-785. doi:10.2106/JBJS.F.00222
10. Kurtz SM, Ong KL, Lau E, Bozic KJ. Impact of the economic downturn on total joint replacement demand in the United States: updated projections to 2021. J Bone Joint Surg Am. 2014;96(8):624-630. doi:10.2106/JBJS.M.00285
11. Zedler BK, Saunders WB, Joyce AR, Vick CC, Murrelle EL. Validation of a screening risk index for serious prescription opioid-induced respiratory depression or overdose in a US commercial health plan claims database. Pain Med. 2018;19(1):68-78. doi:10.1093/pm/pnx009
12. Zedler B, Xie L, Wang L, et al. Development of a risk index for serious prescription opioid-induced respiratory depression or overdose in Veterans Health Administration patients. Pain Med. 2015;16(8):1566-79. doi:10.1111/pme.12777
13. Oliva EM, Bowe T, Tavakoli S, et al. Development and applications of the Veterans Health Administration’s Stratification Tool for Opioid Risk Mitigation (STORM) to improve opioid safety and prevent overdose and suicide. Psychol Serv. 2017;14(1):34-49. doi:10.1037/ser0000099
14. Oliva EM, Christopher MLD, Wells D, et al. Opioid overdose education and naloxone distribution: development of the Veterans Health Administration’s national program. J Am Pharm Assoc (2003). 2017;57(2S):S168-S179.e4. doi:10.1016/j.japh.2017.01.022
15. Noël PH, Copeland LA, Perrin RA, et al. VHA Corporate Data Warehouse height and weight data: opportunities and challenges for health services research. J Rehabil Res Dev. 2010;47(8):739-750. doi:10.1682/jrrd.2009.08.0110
Opioid overdose is a major public health challenge, with recent reports estimating 41 deaths per day in the United States from prescription opioid overdose.1,2 Prescribing naloxone has increasingly been advocated to reduce the risk of opioid overdose for patients identified as high risk. Naloxone distribution has been shown to decrease the incidence of opioid overdoses in the general population.3,4 The Centers for Disease Control and Prevention (CDC) Guideline for Prescribing Opioids for Chronic Pain recommends considering naloxone prescription for patients with a history of overdose or substance use disorder, opioid dosages ≥ 50 morphine equivalent daily dose (MEDD), and concurrent use of benzodiazepines.5
Although the CDC guidelines are intended for primary care clinicians in outpatient settings, naloxone prescribing is also relevant in the postsurgical setting.5 Many surgical patients are at risk for opioid overdose and data from the Veterans Health Administration (VHA) has shown that risk of opioid overdose is 11-fold higher in the 30 days following discharge from a surgical admission, when compared with the subsequent calendar year.6,7 This likely occurs due to new prescriptions or escalated doses of opioids following surgery. Overdose risk may be particularly relevant to orthopedic surgery as postoperative opioids are commonly prescribed.8 Patients undergoing total knee arthroplasty (TKA) may represent a vulnerable population to overdose as it is one of the most commonly performed surgeries for the treatment of chronic pain, and is frequently performed in older adults with medical comorbidities.9,10
Identifying patients at high risk for opioid overdose is important for targeted naloxone dispensing.5 A risk index for overdose or serious opioid-induced respiratory depression (RIOSORD) tool has been developed and validated in veteran and other populations to identify such patients.11 The RIOSORD tool classifies patients by risk level (1-10) and predicts probability of overdose or serious opioid-induced respiratory depression (OSORD). A patient’s level of risk is based on a weighted combination of the 15 independent risk factors most highly associated with OSORD, including comorbid conditions, prescription drug use, and health care utilization.12 Using the RIOSORD tool, the VHA Opioid Education and Naloxone Distribution (OEND) program is a risk mitigation initiative that aims to decrease opioid-related overdose morbidity and mortality. This is achieved via opioid overdose education for prevention, recognition, and response and includes outpatient naloxone prescription.13,14
Despite the comprehensive OEND program, there exists very little data to guide postsurgical naloxone prescribing. The prevalence of known risk factors for overdose in surgical patients remains unknown, as does the prevalence of perioperative naloxone distribution. Understanding overdose risk factors and naloxone prescribing patterns in surgical patients may identify potential targets for OEND efforts. This study retrospectively estimated RIOSORD scores for TKA patients between 2013 to 2016 and described naloxone distribution based on RIOSORD scores and risk factors.
Methods
We identified patients who had undergone primary TKA at VHA hospitals using Current Procedural Terminology (CPT), International Classification of Diseases, Ninth Revision (ICD-9) procedure codes, and data extracted from the VHA Corporate Data Warehouse (CDW) of electronic health records (EHRs). Our study was granted approval with exemption from informed consent by the Durham Veteran Affairs Healthcare System Institutional Review Board.
This retrospective cohort study included all veterans who underwent elective primary TKA from January 1, 2013 through December 31, 2016. We excluded patients who died before discharge.
Outcomes
Our primary outcome was being dispensed an outpatient naloxone prescription following TKA. Naloxone dispensing was identified by examining CDW outpatient pharmacy records with a final dispense date from 1 year before surgery through 7 days after discharge following TKA. To exclude naloxone administration that may have been given in a clinic, prescription data included only records with an outpatient prescription copay. Naloxone dispensing in the year before surgery was chosen to estimate likely preoperative possession of naloxone which could be available in the postoperative period. Naloxone dispensing until 7 days after discharge was chosen to identify any new dispensing that would be available in the postoperative period. These outcomes were examined over the study time frame on an annual basis.
Patient Factors
Demographic variables included age, sex, and race/ethnicity. Independent risk factors for overdose from RIOSORD were identified for each patient.15 These risk factors included comorbidities (opioid use disorder, schizophrenia, bipolar disorder, liver disease, chronic kidney disease, sleep apnea, or lung disease) and prescription drug use (use of opioids, benzodiazepines, long-acting opioids, ≥ 50 MEDD or ≥ 100 MEDD). ICD-9 and ICD-10 diagnosis codes were used to identify comorbidities. Risk classes on day of surgery were identified using a RIOSORD algorithm code. Consistent with the display of RIOSORD risk classes on the VHA Academic Detailing Service OEND risk report, patients were grouped into 3 groups based on their RIOSORD score: classes 1 to 4 (low risk), 5 to 7 (moderate risk), and 8 to 10 (high risk).
Descriptive statistics were used to summarize data on patient demographics, RIOSORD risk factors, overdose events, and naloxone dispensing over time.
Results
The study cohort included 38,011 veterans who underwent primary TKA in the VHA between January 1, 2013 and December 30, 2016. In this cohort, the mean age was 65 years, 93% were male, and 77% were White patients (Table 1). The most common comorbidities were lung disease in 9170 (24.1%) patients, sleep apnea in 6630 (17.4%) patients, chronic kidney disease in 4036 (10.6%) patients, liver disease in 2822 (7.4%) patients, and bipolar disorder in 1748 (4.6%) patients.
In 2013, 63.1% of patients presenting for surgery were actively prescribed opioids. By 2016, this decreased to 50.5%. Benzodiazepine use decreased from 13.2 to 8.8% and long-acting opioid use decreased from 8.5 to 5.8% over the same period. Patients taking ≥ 50 MEDD decreased from 8.0 to 5.3% and patients taking ≥ 100 MEDD decreased from 3.3 to 2.2%. The prevalence of moderate-risk patients decreased from 2.5 to 1.6% and high-risk patients decreased from 0.8 to 0.6% (Figure 1). Cumulatively, the prevalence of presenting with either moderate or high risk of overdose decreased from 3.3 to 2.2% between 2013 to 2016.
Naloxone Dispensing
In 2013, naloxone was not dispensed to any patients at moderate or high risk for overdose between 365 days prior to surgery until 7 days after discharge (Table 2 and Figure 2). Low-risk group naloxone dispensing increased to 2 (0.0%) in 2014, to 13 (0.1%), in 2015, and to 86 (0.9%) in 2016. Moderate-risk group naloxone dispensing remained at 0 (0.0%) in 2014, but increased to 8 (3.5%) in 2015, and to 18 (10.9%) in 2016. High-risk group naloxone dispensing remained at 0 (0.0%) in 2014, but increased to 5 (5.8%) in 2015, and to 8 (12.7%) in 2016 (Figure 3).
Discussion
Our data demonstrate that patients presenting for TKA between 2013 and 2016 routinely had individual risk factors for overdose related to either prescription drug use or comorbidities. We also show that, although the number of patients at moderate and high risk for opioid overdose is decreasing, 2.2% of TKA patients remain at moderate or high risk for opioid overdose based on a weighted combination of these individual risk factors using RIOSORD. As demand for primary TKA is projected to grow to 3.5 million procedures by 2030, using prevalence from 2016, we estimate that 76,560 patients may present for TKA across the US with moderate or high risk for opioid overdose.9 Following discharge, this risk may be even higher as this estimate does not yet account for postoperative opioid use. We demonstrate that through a VHA OEND initiative, naloxone distribution increased and appeared to be targeted to those most at risk using a simple validated tool like RIOSORD.
Presence of an individual risk factor for overdose was present in as many as 63.1% of patients presenting for TKA, as was seen in 2013 with preoperative opioid use. The 3 highest scoring prescription use–related risk factors in RIOSORD are use of opioids ≥ 100 MEDD (16 points), ≥ 50 MEDD (9 points), and long-acting formulations (9 points). All 3 decreased in prevalence over the study period but by 2016 were still seen in 2.2% for ≥ 100 MEDD, 5.3% for ≥ 50 MEDD, and 5.8% for long-acting opioids. This decrease was not surprising given implementation of a VHA-wide opioid safety initiative and the OEND program, but this could also be related to changes in patient selection for surgery in the context of increased awareness of the opioid epidemic. Despite the trend toward safer opioid prescribing, by 2016 over half of patients (50.5%) who presented for TKA were already taking opioids, with 10.6% (543 of 5127) on doses ≥ 50 MEDD.
We observed a decrease in RIOSORD risk each year, consistent with decreasing prescription-related risk factors over time. This was most obvious in the moderate-risk group. It is unclear why a similar decrease was not as obvious in the high-risk group, but this in part may be due to the already low numbers of patients in the high-risk group. This may also represent the high-risk group being somewhat resistant to the initiatives that shifted moderate-risk patients to the low-risk group. There were proportionately more patients in the moderate- and high-risk groups in the original RIOSORD population than in our surgical population, which may be attributed to the fewer comorbidities seen in our surgical population, as well as the higher opioid-prescribing patterns seen prior to the VA OEND initiative.12
Naloxone prescribing was rare prior to the OEND initiative and increased from 2013 to 2016. Increases were most marked in those in moderate- and high-risk groups, although naloxone prescribing also increased among the low-risk group. Integration of RIOSORD stratification into the OEND initiative likely played a role in targeting increased access to naloxone among those at highest risk of overdose. Naloxone dispensing increased for every group, although a significant proportion of moderate- and high-risk patients, 89.1% and 87.3%, respectively, were still not dispensed naloxone by 2016. Moreover, our estimates of perioperative naloxone access were likely an overestimate by including patients dispensed naloxone up to 1 year before surgery until 7 days after surgery. The aim was to include patients who may not have been prescribed naloxone postoperatively because of an existing naloxone prescription at home. Perioperative naloxone access estimates would have been even lower if a narrower window had been used to approximate perioperative access. This identifies an important gap between those who may benefit from naloxone dispensing and those who received naloxone. This in part may be because OEND has not been implemented as routinely in surgical settings as other settings (eg, primary care). OEND efforts may more effectively increase naloxone prescribing among surgical patients if these efforts were targeted at surgical and anesthesia departments. Given that the Comprehensive Addiction and Recovery Act of 2016 requires an assessment of patient risk prior to opioid prescribing and VHA efforts to increase utilization of tools like the Stratification Tool for Opioid Risk Mitigation (STORM), which estimates patient risk when initiating an opioid prescription and includes naloxone as one of many risk mitigation strategies, we anticipate that rates of naloxone prescribing will increase over time.
Limitations
Our study captures a large number of patients across VHA hospitals of varying size nationwide, including a mix of those with and without academic medical center affiliations. This veteran population may not represent the US commercially insured population (CIP). Zedler and colleagues highlighted the differences in prevalence of individual risk factors: notably, the CIP had a substantially higher proportion of females and younger patients.11 VHA had a greater prevalence of common chronic conditions associated with older age. The frequency of opioid dependence was similar among CIP and VHA. However, substance abuse and nonopioid substance dependence diagnoses were 4-fold more frequent among VHA controls as CIP controls. Prescribing of all opioids, except morphine and methadone, was substantially greater in CIP than in VHA.11 Despite a difference in individual risk factors, a CIP-specific RIOSORD has been validated and can be used outside of the VHA to obviate the limitations of the VHA-specific RIOSORD.11
Other limitations include our estimation of naloxone access. We do not know whether naloxone was administered or have a reliable estimate of overdose incidence in this postoperative TKA population. Also, it is important to note that RIOSORD was not developed for surgical patients. The use of RIOSORD in a postoperative population likely underestimates risk of opioid overdose due to the frequent prescriptions of new opioids or escalation of existing MEDD to the postoperative patient. Our study was also retrospective in nature and reliant on accurate coding of patient risk factors. It is possible that comorbidities were not accurately identified by EHR and therefore subject to inconsistency.
Conclusions
Veterans presenting for TKA routinely have risk factors for opioid overdose. We observed a trend toward decreasing overdose risk which coincided with the Opioid Safety and OEND initiatives within the VHA. We also observed an increase in naloxone prescription for moderate- and high-risk patients undergoing TKA, although most of these patients still did not receive naloxone as of 2016. More research is needed to refine and validate the RIOSORD score for surgical populations. Expanding initiatives such as OEND to include surgical patients presents an opportunity to improve access to naloxone for postoperative patients that may help reduce opioid overdose in this population.
Opioid overdose is a major public health challenge, with recent reports estimating 41 deaths per day in the United States from prescription opioid overdose.1,2 Prescribing naloxone has increasingly been advocated to reduce the risk of opioid overdose for patients identified as high risk. Naloxone distribution has been shown to decrease the incidence of opioid overdoses in the general population.3,4 The Centers for Disease Control and Prevention (CDC) Guideline for Prescribing Opioids for Chronic Pain recommends considering naloxone prescription for patients with a history of overdose or substance use disorder, opioid dosages ≥ 50 morphine equivalent daily dose (MEDD), and concurrent use of benzodiazepines.5
Although the CDC guidelines are intended for primary care clinicians in outpatient settings, naloxone prescribing is also relevant in the postsurgical setting.5 Many surgical patients are at risk for opioid overdose and data from the Veterans Health Administration (VHA) has shown that risk of opioid overdose is 11-fold higher in the 30 days following discharge from a surgical admission, when compared with the subsequent calendar year.6,7 This likely occurs due to new prescriptions or escalated doses of opioids following surgery. Overdose risk may be particularly relevant to orthopedic surgery as postoperative opioids are commonly prescribed.8 Patients undergoing total knee arthroplasty (TKA) may represent a vulnerable population to overdose as it is one of the most commonly performed surgeries for the treatment of chronic pain, and is frequently performed in older adults with medical comorbidities.9,10
Identifying patients at high risk for opioid overdose is important for targeted naloxone dispensing.5 A risk index for overdose or serious opioid-induced respiratory depression (RIOSORD) tool has been developed and validated in veteran and other populations to identify such patients.11 The RIOSORD tool classifies patients by risk level (1-10) and predicts probability of overdose or serious opioid-induced respiratory depression (OSORD). A patient’s level of risk is based on a weighted combination of the 15 independent risk factors most highly associated with OSORD, including comorbid conditions, prescription drug use, and health care utilization.12 Using the RIOSORD tool, the VHA Opioid Education and Naloxone Distribution (OEND) program is a risk mitigation initiative that aims to decrease opioid-related overdose morbidity and mortality. This is achieved via opioid overdose education for prevention, recognition, and response and includes outpatient naloxone prescription.13,14
Despite the comprehensive OEND program, there exists very little data to guide postsurgical naloxone prescribing. The prevalence of known risk factors for overdose in surgical patients remains unknown, as does the prevalence of perioperative naloxone distribution. Understanding overdose risk factors and naloxone prescribing patterns in surgical patients may identify potential targets for OEND efforts. This study retrospectively estimated RIOSORD scores for TKA patients between 2013 to 2016 and described naloxone distribution based on RIOSORD scores and risk factors.
Methods
We identified patients who had undergone primary TKA at VHA hospitals using Current Procedural Terminology (CPT), International Classification of Diseases, Ninth Revision (ICD-9) procedure codes, and data extracted from the VHA Corporate Data Warehouse (CDW) of electronic health records (EHRs). Our study was granted approval with exemption from informed consent by the Durham Veteran Affairs Healthcare System Institutional Review Board.
This retrospective cohort study included all veterans who underwent elective primary TKA from January 1, 2013 through December 31, 2016. We excluded patients who died before discharge.
Outcomes
Our primary outcome was being dispensed an outpatient naloxone prescription following TKA. Naloxone dispensing was identified by examining CDW outpatient pharmacy records with a final dispense date from 1 year before surgery through 7 days after discharge following TKA. To exclude naloxone administration that may have been given in a clinic, prescription data included only records with an outpatient prescription copay. Naloxone dispensing in the year before surgery was chosen to estimate likely preoperative possession of naloxone which could be available in the postoperative period. Naloxone dispensing until 7 days after discharge was chosen to identify any new dispensing that would be available in the postoperative period. These outcomes were examined over the study time frame on an annual basis.
Patient Factors
Demographic variables included age, sex, and race/ethnicity. Independent risk factors for overdose from RIOSORD were identified for each patient.15 These risk factors included comorbidities (opioid use disorder, schizophrenia, bipolar disorder, liver disease, chronic kidney disease, sleep apnea, or lung disease) and prescription drug use (use of opioids, benzodiazepines, long-acting opioids, ≥ 50 MEDD or ≥ 100 MEDD). ICD-9 and ICD-10 diagnosis codes were used to identify comorbidities. Risk classes on day of surgery were identified using a RIOSORD algorithm code. Consistent with the display of RIOSORD risk classes on the VHA Academic Detailing Service OEND risk report, patients were grouped into 3 groups based on their RIOSORD score: classes 1 to 4 (low risk), 5 to 7 (moderate risk), and 8 to 10 (high risk).
Descriptive statistics were used to summarize data on patient demographics, RIOSORD risk factors, overdose events, and naloxone dispensing over time.
Results
The study cohort included 38,011 veterans who underwent primary TKA in the VHA between January 1, 2013 and December 30, 2016. In this cohort, the mean age was 65 years, 93% were male, and 77% were White patients (Table 1). The most common comorbidities were lung disease in 9170 (24.1%) patients, sleep apnea in 6630 (17.4%) patients, chronic kidney disease in 4036 (10.6%) patients, liver disease in 2822 (7.4%) patients, and bipolar disorder in 1748 (4.6%) patients.
In 2013, 63.1% of patients presenting for surgery were actively prescribed opioids. By 2016, this decreased to 50.5%. Benzodiazepine use decreased from 13.2 to 8.8% and long-acting opioid use decreased from 8.5 to 5.8% over the same period. Patients taking ≥ 50 MEDD decreased from 8.0 to 5.3% and patients taking ≥ 100 MEDD decreased from 3.3 to 2.2%. The prevalence of moderate-risk patients decreased from 2.5 to 1.6% and high-risk patients decreased from 0.8 to 0.6% (Figure 1). Cumulatively, the prevalence of presenting with either moderate or high risk of overdose decreased from 3.3 to 2.2% between 2013 to 2016.
Naloxone Dispensing
In 2013, naloxone was not dispensed to any patients at moderate or high risk for overdose between 365 days prior to surgery until 7 days after discharge (Table 2 and Figure 2). Low-risk group naloxone dispensing increased to 2 (0.0%) in 2014, to 13 (0.1%), in 2015, and to 86 (0.9%) in 2016. Moderate-risk group naloxone dispensing remained at 0 (0.0%) in 2014, but increased to 8 (3.5%) in 2015, and to 18 (10.9%) in 2016. High-risk group naloxone dispensing remained at 0 (0.0%) in 2014, but increased to 5 (5.8%) in 2015, and to 8 (12.7%) in 2016 (Figure 3).
Discussion
Our data demonstrate that patients presenting for TKA between 2013 and 2016 routinely had individual risk factors for overdose related to either prescription drug use or comorbidities. We also show that, although the number of patients at moderate and high risk for opioid overdose is decreasing, 2.2% of TKA patients remain at moderate or high risk for opioid overdose based on a weighted combination of these individual risk factors using RIOSORD. As demand for primary TKA is projected to grow to 3.5 million procedures by 2030, using prevalence from 2016, we estimate that 76,560 patients may present for TKA across the US with moderate or high risk for opioid overdose.9 Following discharge, this risk may be even higher as this estimate does not yet account for postoperative opioid use. We demonstrate that through a VHA OEND initiative, naloxone distribution increased and appeared to be targeted to those most at risk using a simple validated tool like RIOSORD.
Presence of an individual risk factor for overdose was present in as many as 63.1% of patients presenting for TKA, as was seen in 2013 with preoperative opioid use. The 3 highest scoring prescription use–related risk factors in RIOSORD are use of opioids ≥ 100 MEDD (16 points), ≥ 50 MEDD (9 points), and long-acting formulations (9 points). All 3 decreased in prevalence over the study period but by 2016 were still seen in 2.2% for ≥ 100 MEDD, 5.3% for ≥ 50 MEDD, and 5.8% for long-acting opioids. This decrease was not surprising given implementation of a VHA-wide opioid safety initiative and the OEND program, but this could also be related to changes in patient selection for surgery in the context of increased awareness of the opioid epidemic. Despite the trend toward safer opioid prescribing, by 2016 over half of patients (50.5%) who presented for TKA were already taking opioids, with 10.6% (543 of 5127) on doses ≥ 50 MEDD.
We observed a decrease in RIOSORD risk each year, consistent with decreasing prescription-related risk factors over time. This was most obvious in the moderate-risk group. It is unclear why a similar decrease was not as obvious in the high-risk group, but this in part may be due to the already low numbers of patients in the high-risk group. This may also represent the high-risk group being somewhat resistant to the initiatives that shifted moderate-risk patients to the low-risk group. There were proportionately more patients in the moderate- and high-risk groups in the original RIOSORD population than in our surgical population, which may be attributed to the fewer comorbidities seen in our surgical population, as well as the higher opioid-prescribing patterns seen prior to the VA OEND initiative.12
Naloxone prescribing was rare prior to the OEND initiative and increased from 2013 to 2016. Increases were most marked in those in moderate- and high-risk groups, although naloxone prescribing also increased among the low-risk group. Integration of RIOSORD stratification into the OEND initiative likely played a role in targeting increased access to naloxone among those at highest risk of overdose. Naloxone dispensing increased for every group, although a significant proportion of moderate- and high-risk patients, 89.1% and 87.3%, respectively, were still not dispensed naloxone by 2016. Moreover, our estimates of perioperative naloxone access were likely an overestimate by including patients dispensed naloxone up to 1 year before surgery until 7 days after surgery. The aim was to include patients who may not have been prescribed naloxone postoperatively because of an existing naloxone prescription at home. Perioperative naloxone access estimates would have been even lower if a narrower window had been used to approximate perioperative access. This identifies an important gap between those who may benefit from naloxone dispensing and those who received naloxone. This in part may be because OEND has not been implemented as routinely in surgical settings as other settings (eg, primary care). OEND efforts may more effectively increase naloxone prescribing among surgical patients if these efforts were targeted at surgical and anesthesia departments. Given that the Comprehensive Addiction and Recovery Act of 2016 requires an assessment of patient risk prior to opioid prescribing and VHA efforts to increase utilization of tools like the Stratification Tool for Opioid Risk Mitigation (STORM), which estimates patient risk when initiating an opioid prescription and includes naloxone as one of many risk mitigation strategies, we anticipate that rates of naloxone prescribing will increase over time.
Limitations
Our study captures a large number of patients across VHA hospitals of varying size nationwide, including a mix of those with and without academic medical center affiliations. This veteran population may not represent the US commercially insured population (CIP). Zedler and colleagues highlighted the differences in prevalence of individual risk factors: notably, the CIP had a substantially higher proportion of females and younger patients.11 VHA had a greater prevalence of common chronic conditions associated with older age. The frequency of opioid dependence was similar among CIP and VHA. However, substance abuse and nonopioid substance dependence diagnoses were 4-fold more frequent among VHA controls as CIP controls. Prescribing of all opioids, except morphine and methadone, was substantially greater in CIP than in VHA.11 Despite a difference in individual risk factors, a CIP-specific RIOSORD has been validated and can be used outside of the VHA to obviate the limitations of the VHA-specific RIOSORD.11
Other limitations include our estimation of naloxone access. We do not know whether naloxone was administered or have a reliable estimate of overdose incidence in this postoperative TKA population. Also, it is important to note that RIOSORD was not developed for surgical patients. The use of RIOSORD in a postoperative population likely underestimates risk of opioid overdose due to the frequent prescriptions of new opioids or escalation of existing MEDD to the postoperative patient. Our study was also retrospective in nature and reliant on accurate coding of patient risk factors. It is possible that comorbidities were not accurately identified by EHR and therefore subject to inconsistency.
Conclusions
Veterans presenting for TKA routinely have risk factors for opioid overdose. We observed a trend toward decreasing overdose risk which coincided with the Opioid Safety and OEND initiatives within the VHA. We also observed an increase in naloxone prescription for moderate- and high-risk patients undergoing TKA, although most of these patients still did not receive naloxone as of 2016. More research is needed to refine and validate the RIOSORD score for surgical populations. Expanding initiatives such as OEND to include surgical patients presents an opportunity to improve access to naloxone for postoperative patients that may help reduce opioid overdose in this population.
1. Rudd RA, Seth P, David F, Scholl L. Increases in drug and opioid-involved overdose deaths - United States, 2010-2015. MMWR Morb Mortal Wkly Rep. 2016;65(50-51):1445-1452. Published 2016 Dec 30. doi:10.15585/mmwr.mm655051e1
2. Wilson N, Kariisa M, Seth P, Smith H, Davis NL. Drug and opioid-involved overdose deaths - United States, 2017-2018. MMWR Morb Mortal Wkly Rep. 2020;69(11):290-297. doi:10.15585/mmwr.mm6911a4
3. Walley AY, Xuan Z, Hackman HH, et al. Opioid overdose rates and implementation of overdose education and nasal naloxone distribution in Massachusetts: interrupted time series analysis. BMJ. Jan 30 2013;346:f174. doi:10.1136/bmj.f174
4. McClellan C, Lambdin BH, Ali MM, et al. Opioid-overdose laws association with opioid use and overdose mortality. Addict Behav. 2018;86:90-95. doi:10.1016/j.addbeh.2018.03.014
5. Dowell D, Haegerich TM, Chou R. CDC Guideline for prescribing opioids for chronic pain--United States, 2016. JAMA. 2016;315(15):1624-1645. doi:10.1001/jama.2016.1464
6. Brat GA, Agniel D, Beam A, et al. Postsurgical prescriptions for opioid naive patients and association with overdose and misuse: retrospective cohort study. BMJ. 2018;360:j5790. Published 2018 Jan 17. doi:10.1136/bmj.j5790
7. Mudumbai SC, Lewis ET, Oliva EM, et al. Overdose risk associated with opioid use upon hospital discharge in Veterans Health Administration surgical patients. Pain Med. 2019;20(5):1020-1031. doi:10.1093/pm/pny150
8. Hsia HL, Takemoto S, van de Ven T, et al. Acute pain is associated with chronic opioid use after total knee arthroplasty. Reg Anesth Pain Med. 2018;43(7):705-711. doi:10.1097/AAP.0000000000000831
9. Kurtz S, Ong K, Lau E, Mowat F, Halpern M. Projections of primary and revision hip and knee arthroplasty in the United States from 2005 to 2030. J Bone Joint Surg Am. 2007;89(4):780-785. doi:10.2106/JBJS.F.00222
10. Kurtz SM, Ong KL, Lau E, Bozic KJ. Impact of the economic downturn on total joint replacement demand in the United States: updated projections to 2021. J Bone Joint Surg Am. 2014;96(8):624-630. doi:10.2106/JBJS.M.00285
11. Zedler BK, Saunders WB, Joyce AR, Vick CC, Murrelle EL. Validation of a screening risk index for serious prescription opioid-induced respiratory depression or overdose in a US commercial health plan claims database. Pain Med. 2018;19(1):68-78. doi:10.1093/pm/pnx009
12. Zedler B, Xie L, Wang L, et al. Development of a risk index for serious prescription opioid-induced respiratory depression or overdose in Veterans Health Administration patients. Pain Med. 2015;16(8):1566-79. doi:10.1111/pme.12777
13. Oliva EM, Bowe T, Tavakoli S, et al. Development and applications of the Veterans Health Administration’s Stratification Tool for Opioid Risk Mitigation (STORM) to improve opioid safety and prevent overdose and suicide. Psychol Serv. 2017;14(1):34-49. doi:10.1037/ser0000099
14. Oliva EM, Christopher MLD, Wells D, et al. Opioid overdose education and naloxone distribution: development of the Veterans Health Administration’s national program. J Am Pharm Assoc (2003). 2017;57(2S):S168-S179.e4. doi:10.1016/j.japh.2017.01.022
15. Noël PH, Copeland LA, Perrin RA, et al. VHA Corporate Data Warehouse height and weight data: opportunities and challenges for health services research. J Rehabil Res Dev. 2010;47(8):739-750. doi:10.1682/jrrd.2009.08.0110
1. Rudd RA, Seth P, David F, Scholl L. Increases in drug and opioid-involved overdose deaths - United States, 2010-2015. MMWR Morb Mortal Wkly Rep. 2016;65(50-51):1445-1452. Published 2016 Dec 30. doi:10.15585/mmwr.mm655051e1
2. Wilson N, Kariisa M, Seth P, Smith H, Davis NL. Drug and opioid-involved overdose deaths - United States, 2017-2018. MMWR Morb Mortal Wkly Rep. 2020;69(11):290-297. doi:10.15585/mmwr.mm6911a4
3. Walley AY, Xuan Z, Hackman HH, et al. Opioid overdose rates and implementation of overdose education and nasal naloxone distribution in Massachusetts: interrupted time series analysis. BMJ. Jan 30 2013;346:f174. doi:10.1136/bmj.f174
4. McClellan C, Lambdin BH, Ali MM, et al. Opioid-overdose laws association with opioid use and overdose mortality. Addict Behav. 2018;86:90-95. doi:10.1016/j.addbeh.2018.03.014
5. Dowell D, Haegerich TM, Chou R. CDC Guideline for prescribing opioids for chronic pain--United States, 2016. JAMA. 2016;315(15):1624-1645. doi:10.1001/jama.2016.1464
6. Brat GA, Agniel D, Beam A, et al. Postsurgical prescriptions for opioid naive patients and association with overdose and misuse: retrospective cohort study. BMJ. 2018;360:j5790. Published 2018 Jan 17. doi:10.1136/bmj.j5790
7. Mudumbai SC, Lewis ET, Oliva EM, et al. Overdose risk associated with opioid use upon hospital discharge in Veterans Health Administration surgical patients. Pain Med. 2019;20(5):1020-1031. doi:10.1093/pm/pny150
8. Hsia HL, Takemoto S, van de Ven T, et al. Acute pain is associated with chronic opioid use after total knee arthroplasty. Reg Anesth Pain Med. 2018;43(7):705-711. doi:10.1097/AAP.0000000000000831
9. Kurtz S, Ong K, Lau E, Mowat F, Halpern M. Projections of primary and revision hip and knee arthroplasty in the United States from 2005 to 2030. J Bone Joint Surg Am. 2007;89(4):780-785. doi:10.2106/JBJS.F.00222
10. Kurtz SM, Ong KL, Lau E, Bozic KJ. Impact of the economic downturn on total joint replacement demand in the United States: updated projections to 2021. J Bone Joint Surg Am. 2014;96(8):624-630. doi:10.2106/JBJS.M.00285
11. Zedler BK, Saunders WB, Joyce AR, Vick CC, Murrelle EL. Validation of a screening risk index for serious prescription opioid-induced respiratory depression or overdose in a US commercial health plan claims database. Pain Med. 2018;19(1):68-78. doi:10.1093/pm/pnx009
12. Zedler B, Xie L, Wang L, et al. Development of a risk index for serious prescription opioid-induced respiratory depression or overdose in Veterans Health Administration patients. Pain Med. 2015;16(8):1566-79. doi:10.1111/pme.12777
13. Oliva EM, Bowe T, Tavakoli S, et al. Development and applications of the Veterans Health Administration’s Stratification Tool for Opioid Risk Mitigation (STORM) to improve opioid safety and prevent overdose and suicide. Psychol Serv. 2017;14(1):34-49. doi:10.1037/ser0000099
14. Oliva EM, Christopher MLD, Wells D, et al. Opioid overdose education and naloxone distribution: development of the Veterans Health Administration’s national program. J Am Pharm Assoc (2003). 2017;57(2S):S168-S179.e4. doi:10.1016/j.japh.2017.01.022
15. Noël PH, Copeland LA, Perrin RA, et al. VHA Corporate Data Warehouse height and weight data: opportunities and challenges for health services research. J Rehabil Res Dev. 2010;47(8):739-750. doi:10.1682/jrrd.2009.08.0110
Evaluating the Impact of a Urinalysis to Reflex Culture Process Change in the Emergency Department at a Veterans Affairs Hospital
Automated urine cultures (UCs) following urinalysis (UA) are often used in emergency departments (EDs) to identify urinary tract infections (UTIs). The fast-paced environment of the ED makes this method of proactive collection and facilitation of UC favorable. However, results are often reported as no organism growth or the growth of clinically insignificant organisms, leading to the overdetection and overtreatment of asymptomatic bacteriuria (ASB).1-3 An estimated 30 to 60% of patients with ASB receive unwarranted antibiotic treatment, which is associated with an increased risk of developing Clostridioides difficile infection and contributes to the development of antimicrobial resistance.4-10 The costs associated with UC are an important consideration given the use of resources, the time and effort required to collect and process large numbers of negative cultures, and further efforts devoted to the follow-up of ED culture results.
Changes in traditional testing involving testing of both a UA and UC to reflex testing where urine specimens undergo culture only if they meet certain criteria have been described.11-14 This change in traditional testing aims to reduce the number of potentially unnecessary cultures performed without compromising clinical care. Leukocyte quantity in the UA has been shown to be a reliable predictor of true infection.11,15 Fok and colleagues demonstrated that reflex urine testing in ambulatory male urology patients in which cultures were done on only urine specimens with > 5 white blood cells per high-power field (WBC/HPF) would have missed only 7% of positive UCs, while avoiding 69% of cultures.11
At the Edward Hines, Jr Veterans Affairs Hospital (Hines VA), inappropriate UC ordering and treatment for ASB has been identified as an area needing improvement. An evaluation was conducted at the facility to determine the population of inpatient veterans with a positive UC who were appropriately managed. Of the 113 study patients with a positive UC included in this review, 77 (68%) had a diagnosis of ASB, with > 80% of patients with ASB (and no other suspected infections) receiving antimicrobial therapy.8 A subsequent evaluation was conducted at the Hines VA ED to evaluate UTI treatment and follow-up. Of the 173 ED patients included, 23% received antibiotic therapy for an ASB and 60% had a UA and UC collected but did not report symptoms.9 Finally, a review by the Hines VA laboratory showed that in May 2017, of 359 UCs sent from various locations of the hospital, 38% were obtained in the setting of a negative UA.
A multidisciplinary group with representation from primary care, infectious diseases, pharmacy, nursing, laboratory, and informatics was created with a goal to improve the workup and management of UTIs. In addition to periodic education for the clinicians regarding appropriate use and interpretation of UA and UC along with judicious use of antimicrobials especially in the setting of ASB, a UA to reflex culture process change was implemented. This allowed for automatic cancellation of a UC in the setting of a negative UA, which was designed to help facilitate appropriate UC ordering.
Methods
The primary objective of this study was to compare the frequency of inappropriate UC use and inappropriate antibiotic prescribing pre- and postimplementation of this UA to reflex culture process change. An inappropriate UC was defined as a UC ordered despite a negative UA in asymptomatic patients. Inappropriate antibiotic prescribing was defined as treatment of patients with ASB. The secondary objective evaluated postintervention data to assess the frequency of outpatient, ED, and hospital visits for UTI-related symptoms in the group of patients that had a UC cancelled as a result of the new process change (within a 7-day period of the initial UA) to determine whether patients with true infections were missed due to the process change.
Study Design and Setting
This pre-post quality improvement (QI) study analyzed the UC-ordering practices for UTIs sent from the ED at the Hines VA. This VA is a 483-bed tertiary care hospital in Chicago, Illinois, and serves > 57,000 veterans and about 23,000 ED visits annually. This study was approved by the Edward Hines, Jr VA Institutional Review Board as a quality assurance/QI proposal prior to data collection.
Patient Selection
All patients who
When comparing postintervention data with preintervention data for the primary study objective, the same exclusion criteria from the 2015 study were applied to the present study, which excluded ED patients who were admitted for inpatient care, concurrent antibiotic therapy for a non-UTI indication, duplicate cultures, and use of chronic bladder management devices. All patients identified as receiving a UA during the specified postintervention study period were included for evaluation of the secondary study objective.
Interventions
After physician education, an ED process change was implemented on October 3, 2017. This process change involved the creation of new order sets in the EHR that allowed clinicians to order a UA only, a UA with culture that would be cancelled by laboratory personnel if the UA did not result in > 5 WBC/HPF, and a UA with culture designated as do not cancel, where the UC was processed regardless of the UA results. The scenarios in which the latter option was considered appropriate were listed on the ordering screen and included pregnancy, a genitourinary procedure with necessary preoperative culture, and neutropenia.
Measurements
Postimplementation, all UAs were reviewed and grouped as follows: (1) positive UA with subsequent UC; (2) negative UA, culture cancelled; (3) only UA ordered (no culture); or (4) do not cancel UC ordered. Of the UAs that were analyzed, the following data were collected: demographics, comorbidities, concurrent medications for benign prostatic hyperplasia (BPH) and/or overactive bladder (OAB), documented allergies/adverse drug reactions to antibiotics, date of ED visit, documented UTI signs/symptoms (defined as frequency, urgency, dysuria, fever, suprapubic pain, or altered mental status in patients unable to verbalize urinary symptoms), UC results and susceptibilities, number of UCs repeated within 7 days after initial UA, requirement of antibiotic for UTI within 7 days of initial UA, antibiotic prescribed, duration of antibiotic therapy, and outpatient visits, ED visits, or need for hospital admission within 7 days of the initial UA for UTI-related symptoms. Other relevant UA and UC data that could not be obtained from the EHR were collected by generating a report using the Veterans Information Systems and Technology Architecture (VistA).
Analysis
Statistical analysis was performed using SAS v9.4. Independent t tests and Fisher exact tests were used to describe difference pre- and postintervention. Statistical significance was considered for P < .05. Based on results from the previous study conducted at this facility in addition to a literature review, it was determined that 92 patients in each group (pre- and postintervention) would be necessary to detect a 15% increase in percentage of patients appropriately treated for a UTI.
Results
There were 684 UAs evaluated from ED visits, 429 preintervention and 255 postintervention. The 255 patients were evaluated for the secondary objective of the study. Of the 255 patients with UAs identified postintervention, 150 were excluded based on the predefined exclusion criteria, and the remaining 105 were compared with the 173 patients from the preintervention group and were included in the analysis for the primary objective (Figure 1).
Patients in the postintervention group were younger than those in the preintervention group (P < .02): otherwise the groups were similar (Table 1). Inappropriate antibiotics for ASB decreased from 10.2% preintervention to 1.9% postintervention (odds ratio, 0.17; P = .01) (Table 2). UC processing despite a negative UA significantly decreased from 100% preintervention to 38.6% postintervention (P < .001) (Table 3). In patients with a negative UA, antibiotic prescribing decreased by 25.3% postintervention, but this difference was not statistically significant.
Postintervention, of 255 UAs evaluated, 95 (37.3%) were positive with a processed UC and 95 (37.3%) were negative with UC cancelled, 43 (16.9%) were ordered as DNC, and 22 (8.6%) were ordered without a UC (Figure 2). Twenty-eight of the 95 (29.5%) UAs with processed UCs did not meet the criteria for a positive UA and were not designated as DNC. When the UCs of this subgroup of patients were further analyzed, we found that 2 of the cultures were positive of which 1 patient was symptomatic and required antibiotic therapy.
Of the 95 patients with a negative UA, 69 (72.6%) presented without any UTI-related symptoms. In this group, there were no reports of outpatient visits, ED visits, or hospital admissions within 7 days of initial UA for UTI-related symptoms. None of the UCs ordered as DNC had a supporting reason identified. Nonetheless, the UC results from this patient subgroup also were analyzed further and resulted in 4 patients with negative UA and positive subsequent UC, 1 was symptomatic and required antibiotic therapy.
Discussion
A simple process change at the Hines VA resulted in benefits related to antimicrobial stewardship without conferring adverse outcomes on patient safety. Both UC processing despite a negative UA and inappropriate antibiotic prescribing for ASB were reduced significantly postintervention. This process change was piloted in the ED where UCs are often included as part of the initial diagnostic testing in patients who may not report UTI-related symptoms but for whom a UC is often bundled with other infectious workup, depending on the patient presentation.
Reflex testing of urine specimens has been described in the literature, both in an exploratory nature where impact of a reflex UC cancellation protocol based on certain UA criteria is measured by percent reduction of UCs processed as well as results of such interventions implemented into clinical practice.11-13 A retrospective study performed at the University of North Carolina Medical Center evaluated patients who presented to the ED during a 6-month period and had both an automated UA and UC collected. UC processing was restricted to UA that was positive for nitrites, leukocyte esterase, bacteria, or > 10 WBC/HPF. Use of this reflex culture cancellation protocol could have eliminated 604 of the 1546 (39.1%) cultures processed. However, 11 of the 314 (3.5%) positive cultures could have been missed.13 This same protocol was externally validated at another large academic ED setting, where similar results were found.14
In clinical practice, there is a natural tendency to reflexively prescribe antibiotics based on the results of a positive UC due to the hesitancy in ignoring these results, despite lack of a suspicion for a true infection. Leis and colleagues explored this in a proof-of-concept study evaluating the impact of discontinuing the routine reporting of positive UC results from noncatheterized inpatients and requesting clinicians to call the laboratory for results if a UTI was suspected.16 This intervention resulted in a statistically significant reduction in treatment of ASB in noncatheterized patients from 48 to 12% pre- and postintervention. Clinicians requested culture results only 14% of the time, and there were no adverse outcomes among untreated noncatheterized patients. More recently, a QI study conducted at a large community hospital in Toronto, Ontario, Canada, implemented a 2-step model of care for urine collection.17 UC was collected but only processed by the microbiology laboratory if the ED physicians deemed it necessary after clinical assessment.
After implementation, there was a decrease in the proportion of ED visits associated with processed UC (from 6.0% to 4.7% of visits per week; P < .001), ED visits associated with callbacks for processing UC (1.8% to 1.1% of visits per month; P < .001), and antimicrobial prescriptions for urinary symptoms among hospitalized patients (from 20.6% to 10.9%; P < .001). Equally important, despite the 937 cases in which urine was collected but cultures were not processed, no evidence of untreated UTIs was identified.17
The results from the present study similarly demonstrate minimal concern for potentially undertreating these patients. As seen in the subgroup of patients included in the positive UA group, which did not meet criteria for positive UA per protocol (n = 29), only 2 of the subsequent cultures were positive, of which only 1 patient required antibiotic therapy based on the clinical presentation. In addition, in the group of negative UAs with subsequent cancellation of the UC, there were no found reports of outpatient visits, ED visits, or hospital admissions within 7 days of the initial UA for UTI-related symptoms.
Limitations
This single-center, pre-post QI study was not without limitations. Manual chart reviews were required, and accuracy of information was dependent on clinician documentation and assessment of UTI-related symptoms. The population studied was predominately older males; thus, results may not be applicable to females or young adults. Additionally, recognition of a negative UA and subsequent cancellation of the UC was dependent on laboratory personnel. As noted in the patient group with a positive UA, some of these UAs were negative and may have been overlooked; therefore, subsequent UCs were inappropriately processed. However, this occurred infrequently and confirmed the low probability of true UTI in the setting of a negative UA. Follow-up for UTI-related symptoms may not have been captured if a patient had presented to an outside facility. Last, definitions of a positive UA differed slightly between the pre- and postintervention groups. The preintervention study defined a positive UA as a WBC count > 5 WBC/HPF and positive leukocyte esterase, whereas the present study defined a positive UA with a WBC count > 5. This may have resulted in an overestimation of positive UA in the postintervention group.
Conclusions
Better selective use of UC testing may improve stewardship resources and reduce costs impacting both ED and clinical laboratories. Furthermore, benefits can include a reduction in the use of time and resources required to collect samples for culture, use of test supplies, the time and effort required to process the large number of negative cultures, and resources devoted to the follow-up of these ED culture results. The described UA to reflex culture process change demonstrated a significant reduction in the processing of inappropriate UC and unnecessary antibiotics for ASB. There were no missed UTIs or other adverse patient outcomes noted. This process change has been implemented in all departments at the Hines VA and additional data will be collected to ensure consistent outcomes.
1. Chironda B, Clancy S, Powis JE. Optimizing urine culture collection in the emergency department using frontline ownership interventions. Clin Infect Dis. 2014;59(7):1038-1039. doi:10.1093/cid/ciu412
2. Nagurney JT, Brown DF, Chang Y, Sane S, Wang AC, Weiner JB. Use of diagnostic testing in the emergency department for patients presenting with non-traumatic abdominal pain. J Emerg Med. 2003;25(4):363-371. doi:10.1016/s0736-4679(03)00237-3
3. Lammers RL, Gibson S, Kovacs D, Sears W, Strachan G. Comparison of test characteristics of urine dipstick and urinalysis at various test cutoff points. Ann Emerg Med. 2001;38(5):505-512. doi:10.1067/mem.2001.119427
4. Nicolle LE, Gupta K, Bradley SF, et al. Clinical practice guideline for the management of asymptomatic bacteriuria: 2019 update by the Infectious Diseases Society of America. Clin Infect Dis. 2019;68(10):1611-1615. doi:10.1093/cid/ciy1121
5. Trautner BW, Grigoryan L, Petersen NJ, et al. Effectiveness of an antimicrobial stewardship approach for urinary catheter-associated asymptomatic bacteriuria. JAMA Intern Med. 2015;175(7):1120-1127. doi:10.1001/jamainternmed.2015.1878
6. Hartley S, Valley S, Kuhn L, et al. Overtreatment of asymptomatic bacteriuria: identifying targets for improvement. Infect Control Hosp Epidemiol. 2015;36(4):470-473. doi:10.1017/ice.2014.73
7. Bader MS, Loeb M, Brooks AA. An update on the management of urinary tract infections in the era of antimicrobial resistance. Postgrad Med. 2017;129(2):242-258. doi:10.1080/00325481.2017.1246055
8. Spivak ES, Burk M, Zhang R, et al. Management of bacteriuria in Veterans Affairs hospitals. Clin Infect Dis. 2017;65(6):910-917. doi:10.1093/cid/cix474
9. Kim EY, Patel U, Patel B, Suda KJ. Evaluation of bacteriuria treatment and follow-up initiated in the emergency department at a Veterans Affairs hospital. J Pharm Technol. 2017;33(5):183-188. doi:10.1177/8755122517718214
10. Brown E, Talbot GH, Axelrod P, Provencher M, Hoegg C. Risk factors for Clostridium difficile toxin-associated diarrhea. Infect Control Hosp Epidemiol. 1990;11(6):283-290. doi:10.1086/646173
11. Fok C, Fitzgerald MP, Turk T, Mueller E, Dalaza L, Schreckenberger P. Reflex testing of male urine specimens misses few positive cultures may reduce unnecessary testing of normal specimens. Urology. 2010;75(1):74-76. doi:10.1016/j.urology.2009.08.071
12. Munigala S, Jackups RR Jr, Poirier RF, et al. Impact of order set design on urine culturing practices at an academic medical centre emergency department. BMJ Qual Saf. 2018;27(8):587-592. doi:10.1136/bmjqs-2017-006899
13. Jones CW, Culbreath KD, Mehrotra A, Gilligan PH. Reflect urine culture cancellation in the emergency department. J Emerg Med. 2014;46(1):71-76. doi:10.1016/j.jemermed.2013.08.042
14. Hertz JT, Lescallette RD, Barrett TW, Ward MJ, Self WH. External validation of an ED protocol for reflex urine culture cancelation. Am J Emerg Med. 2015;33(12):1838-1839. doi:10.1016/j.ajem.2015.09.026
15. Stamm WE. Measurement of pyuria and its relation to bacteriuria. Am J Med. 1983;75(1B):53-58. doi:10.1016/0002-9343(83)90073-6
16. Leis JA, Rebick GW, Daneman N, et al. Reducing antimicrobial therapy for asymptomatic bacteriuria among noncatheterized inpatients: a proof-of-concept study. Clin Infect Dis. 2014;58(7):980-983. doi:10.1093/cid/ciu010
17. Stagg A, Lutz H, Kirpalaney S, et al. Impact of two-step urine culture ordering in the emergency department: a time series analysis. BMJ Qual Saf. 2017;27:140-147. doi:10.1136/bmjqs-2016-006250
Automated urine cultures (UCs) following urinalysis (UA) are often used in emergency departments (EDs) to identify urinary tract infections (UTIs). The fast-paced environment of the ED makes this method of proactive collection and facilitation of UC favorable. However, results are often reported as no organism growth or the growth of clinically insignificant organisms, leading to the overdetection and overtreatment of asymptomatic bacteriuria (ASB).1-3 An estimated 30 to 60% of patients with ASB receive unwarranted antibiotic treatment, which is associated with an increased risk of developing Clostridioides difficile infection and contributes to the development of antimicrobial resistance.4-10 The costs associated with UC are an important consideration given the use of resources, the time and effort required to collect and process large numbers of negative cultures, and further efforts devoted to the follow-up of ED culture results.
Changes in traditional testing involving testing of both a UA and UC to reflex testing where urine specimens undergo culture only if they meet certain criteria have been described.11-14 This change in traditional testing aims to reduce the number of potentially unnecessary cultures performed without compromising clinical care. Leukocyte quantity in the UA has been shown to be a reliable predictor of true infection.11,15 Fok and colleagues demonstrated that reflex urine testing in ambulatory male urology patients in which cultures were done on only urine specimens with > 5 white blood cells per high-power field (WBC/HPF) would have missed only 7% of positive UCs, while avoiding 69% of cultures.11
At the Edward Hines, Jr Veterans Affairs Hospital (Hines VA), inappropriate UC ordering and treatment for ASB has been identified as an area needing improvement. An evaluation was conducted at the facility to determine the population of inpatient veterans with a positive UC who were appropriately managed. Of the 113 study patients with a positive UC included in this review, 77 (68%) had a diagnosis of ASB, with > 80% of patients with ASB (and no other suspected infections) receiving antimicrobial therapy.8 A subsequent evaluation was conducted at the Hines VA ED to evaluate UTI treatment and follow-up. Of the 173 ED patients included, 23% received antibiotic therapy for an ASB and 60% had a UA and UC collected but did not report symptoms.9 Finally, a review by the Hines VA laboratory showed that in May 2017, of 359 UCs sent from various locations of the hospital, 38% were obtained in the setting of a negative UA.
A multidisciplinary group with representation from primary care, infectious diseases, pharmacy, nursing, laboratory, and informatics was created with a goal to improve the workup and management of UTIs. In addition to periodic education for the clinicians regarding appropriate use and interpretation of UA and UC along with judicious use of antimicrobials especially in the setting of ASB, a UA to reflex culture process change was implemented. This allowed for automatic cancellation of a UC in the setting of a negative UA, which was designed to help facilitate appropriate UC ordering.
Methods
The primary objective of this study was to compare the frequency of inappropriate UC use and inappropriate antibiotic prescribing pre- and postimplementation of this UA to reflex culture process change. An inappropriate UC was defined as a UC ordered despite a negative UA in asymptomatic patients. Inappropriate antibiotic prescribing was defined as treatment of patients with ASB. The secondary objective evaluated postintervention data to assess the frequency of outpatient, ED, and hospital visits for UTI-related symptoms in the group of patients that had a UC cancelled as a result of the new process change (within a 7-day period of the initial UA) to determine whether patients with true infections were missed due to the process change.
Study Design and Setting
This pre-post quality improvement (QI) study analyzed the UC-ordering practices for UTIs sent from the ED at the Hines VA. This VA is a 483-bed tertiary care hospital in Chicago, Illinois, and serves > 57,000 veterans and about 23,000 ED visits annually. This study was approved by the Edward Hines, Jr VA Institutional Review Board as a quality assurance/QI proposal prior to data collection.
Patient Selection
All patients who
When comparing postintervention data with preintervention data for the primary study objective, the same exclusion criteria from the 2015 study were applied to the present study, which excluded ED patients who were admitted for inpatient care, concurrent antibiotic therapy for a non-UTI indication, duplicate cultures, and use of chronic bladder management devices. All patients identified as receiving a UA during the specified postintervention study period were included for evaluation of the secondary study objective.
Interventions
After physician education, an ED process change was implemented on October 3, 2017. This process change involved the creation of new order sets in the EHR that allowed clinicians to order a UA only, a UA with culture that would be cancelled by laboratory personnel if the UA did not result in > 5 WBC/HPF, and a UA with culture designated as do not cancel, where the UC was processed regardless of the UA results. The scenarios in which the latter option was considered appropriate were listed on the ordering screen and included pregnancy, a genitourinary procedure with necessary preoperative culture, and neutropenia.
Measurements
Postimplementation, all UAs were reviewed and grouped as follows: (1) positive UA with subsequent UC; (2) negative UA, culture cancelled; (3) only UA ordered (no culture); or (4) do not cancel UC ordered. Of the UAs that were analyzed, the following data were collected: demographics, comorbidities, concurrent medications for benign prostatic hyperplasia (BPH) and/or overactive bladder (OAB), documented allergies/adverse drug reactions to antibiotics, date of ED visit, documented UTI signs/symptoms (defined as frequency, urgency, dysuria, fever, suprapubic pain, or altered mental status in patients unable to verbalize urinary symptoms), UC results and susceptibilities, number of UCs repeated within 7 days after initial UA, requirement of antibiotic for UTI within 7 days of initial UA, antibiotic prescribed, duration of antibiotic therapy, and outpatient visits, ED visits, or need for hospital admission within 7 days of the initial UA for UTI-related symptoms. Other relevant UA and UC data that could not be obtained from the EHR were collected by generating a report using the Veterans Information Systems and Technology Architecture (VistA).
Analysis
Statistical analysis was performed using SAS v9.4. Independent t tests and Fisher exact tests were used to describe difference pre- and postintervention. Statistical significance was considered for P < .05. Based on results from the previous study conducted at this facility in addition to a literature review, it was determined that 92 patients in each group (pre- and postintervention) would be necessary to detect a 15% increase in percentage of patients appropriately treated for a UTI.
Results
There were 684 UAs evaluated from ED visits, 429 preintervention and 255 postintervention. The 255 patients were evaluated for the secondary objective of the study. Of the 255 patients with UAs identified postintervention, 150 were excluded based on the predefined exclusion criteria, and the remaining 105 were compared with the 173 patients from the preintervention group and were included in the analysis for the primary objective (Figure 1).
Patients in the postintervention group were younger than those in the preintervention group (P < .02): otherwise the groups were similar (Table 1). Inappropriate antibiotics for ASB decreased from 10.2% preintervention to 1.9% postintervention (odds ratio, 0.17; P = .01) (Table 2). UC processing despite a negative UA significantly decreased from 100% preintervention to 38.6% postintervention (P < .001) (Table 3). In patients with a negative UA, antibiotic prescribing decreased by 25.3% postintervention, but this difference was not statistically significant.
Postintervention, of 255 UAs evaluated, 95 (37.3%) were positive with a processed UC and 95 (37.3%) were negative with UC cancelled, 43 (16.9%) were ordered as DNC, and 22 (8.6%) were ordered without a UC (Figure 2). Twenty-eight of the 95 (29.5%) UAs with processed UCs did not meet the criteria for a positive UA and were not designated as DNC. When the UCs of this subgroup of patients were further analyzed, we found that 2 of the cultures were positive of which 1 patient was symptomatic and required antibiotic therapy.
Of the 95 patients with a negative UA, 69 (72.6%) presented without any UTI-related symptoms. In this group, there were no reports of outpatient visits, ED visits, or hospital admissions within 7 days of initial UA for UTI-related symptoms. None of the UCs ordered as DNC had a supporting reason identified. Nonetheless, the UC results from this patient subgroup also were analyzed further and resulted in 4 patients with negative UA and positive subsequent UC, 1 was symptomatic and required antibiotic therapy.
Discussion
A simple process change at the Hines VA resulted in benefits related to antimicrobial stewardship without conferring adverse outcomes on patient safety. Both UC processing despite a negative UA and inappropriate antibiotic prescribing for ASB were reduced significantly postintervention. This process change was piloted in the ED where UCs are often included as part of the initial diagnostic testing in patients who may not report UTI-related symptoms but for whom a UC is often bundled with other infectious workup, depending on the patient presentation.
Reflex testing of urine specimens has been described in the literature, both in an exploratory nature where impact of a reflex UC cancellation protocol based on certain UA criteria is measured by percent reduction of UCs processed as well as results of such interventions implemented into clinical practice.11-13 A retrospective study performed at the University of North Carolina Medical Center evaluated patients who presented to the ED during a 6-month period and had both an automated UA and UC collected. UC processing was restricted to UA that was positive for nitrites, leukocyte esterase, bacteria, or > 10 WBC/HPF. Use of this reflex culture cancellation protocol could have eliminated 604 of the 1546 (39.1%) cultures processed. However, 11 of the 314 (3.5%) positive cultures could have been missed.13 This same protocol was externally validated at another large academic ED setting, where similar results were found.14
In clinical practice, there is a natural tendency to reflexively prescribe antibiotics based on the results of a positive UC due to the hesitancy in ignoring these results, despite lack of a suspicion for a true infection. Leis and colleagues explored this in a proof-of-concept study evaluating the impact of discontinuing the routine reporting of positive UC results from noncatheterized inpatients and requesting clinicians to call the laboratory for results if a UTI was suspected.16 This intervention resulted in a statistically significant reduction in treatment of ASB in noncatheterized patients from 48 to 12% pre- and postintervention. Clinicians requested culture results only 14% of the time, and there were no adverse outcomes among untreated noncatheterized patients. More recently, a QI study conducted at a large community hospital in Toronto, Ontario, Canada, implemented a 2-step model of care for urine collection.17 UC was collected but only processed by the microbiology laboratory if the ED physicians deemed it necessary after clinical assessment.
After implementation, there was a decrease in the proportion of ED visits associated with processed UC (from 6.0% to 4.7% of visits per week; P < .001), ED visits associated with callbacks for processing UC (1.8% to 1.1% of visits per month; P < .001), and antimicrobial prescriptions for urinary symptoms among hospitalized patients (from 20.6% to 10.9%; P < .001). Equally important, despite the 937 cases in which urine was collected but cultures were not processed, no evidence of untreated UTIs was identified.17
The results from the present study similarly demonstrate minimal concern for potentially undertreating these patients. As seen in the subgroup of patients included in the positive UA group, which did not meet criteria for positive UA per protocol (n = 29), only 2 of the subsequent cultures were positive, of which only 1 patient required antibiotic therapy based on the clinical presentation. In addition, in the group of negative UAs with subsequent cancellation of the UC, there were no found reports of outpatient visits, ED visits, or hospital admissions within 7 days of the initial UA for UTI-related symptoms.
Limitations
This single-center, pre-post QI study was not without limitations. Manual chart reviews were required, and accuracy of information was dependent on clinician documentation and assessment of UTI-related symptoms. The population studied was predominately older males; thus, results may not be applicable to females or young adults. Additionally, recognition of a negative UA and subsequent cancellation of the UC was dependent on laboratory personnel. As noted in the patient group with a positive UA, some of these UAs were negative and may have been overlooked; therefore, subsequent UCs were inappropriately processed. However, this occurred infrequently and confirmed the low probability of true UTI in the setting of a negative UA. Follow-up for UTI-related symptoms may not have been captured if a patient had presented to an outside facility. Last, definitions of a positive UA differed slightly between the pre- and postintervention groups. The preintervention study defined a positive UA as a WBC count > 5 WBC/HPF and positive leukocyte esterase, whereas the present study defined a positive UA with a WBC count > 5. This may have resulted in an overestimation of positive UA in the postintervention group.
Conclusions
Better selective use of UC testing may improve stewardship resources and reduce costs impacting both ED and clinical laboratories. Furthermore, benefits can include a reduction in the use of time and resources required to collect samples for culture, use of test supplies, the time and effort required to process the large number of negative cultures, and resources devoted to the follow-up of these ED culture results. The described UA to reflex culture process change demonstrated a significant reduction in the processing of inappropriate UC and unnecessary antibiotics for ASB. There were no missed UTIs or other adverse patient outcomes noted. This process change has been implemented in all departments at the Hines VA and additional data will be collected to ensure consistent outcomes.
Automated urine cultures (UCs) following urinalysis (UA) are often used in emergency departments (EDs) to identify urinary tract infections (UTIs). The fast-paced environment of the ED makes this method of proactive collection and facilitation of UC favorable. However, results are often reported as no organism growth or the growth of clinically insignificant organisms, leading to the overdetection and overtreatment of asymptomatic bacteriuria (ASB).1-3 An estimated 30 to 60% of patients with ASB receive unwarranted antibiotic treatment, which is associated with an increased risk of developing Clostridioides difficile infection and contributes to the development of antimicrobial resistance.4-10 The costs associated with UC are an important consideration given the use of resources, the time and effort required to collect and process large numbers of negative cultures, and further efforts devoted to the follow-up of ED culture results.
Changes in traditional testing involving testing of both a UA and UC to reflex testing where urine specimens undergo culture only if they meet certain criteria have been described.11-14 This change in traditional testing aims to reduce the number of potentially unnecessary cultures performed without compromising clinical care. Leukocyte quantity in the UA has been shown to be a reliable predictor of true infection.11,15 Fok and colleagues demonstrated that reflex urine testing in ambulatory male urology patients in which cultures were done on only urine specimens with > 5 white blood cells per high-power field (WBC/HPF) would have missed only 7% of positive UCs, while avoiding 69% of cultures.11
At the Edward Hines, Jr Veterans Affairs Hospital (Hines VA), inappropriate UC ordering and treatment for ASB has been identified as an area needing improvement. An evaluation was conducted at the facility to determine the population of inpatient veterans with a positive UC who were appropriately managed. Of the 113 study patients with a positive UC included in this review, 77 (68%) had a diagnosis of ASB, with > 80% of patients with ASB (and no other suspected infections) receiving antimicrobial therapy.8 A subsequent evaluation was conducted at the Hines VA ED to evaluate UTI treatment and follow-up. Of the 173 ED patients included, 23% received antibiotic therapy for an ASB and 60% had a UA and UC collected but did not report symptoms.9 Finally, a review by the Hines VA laboratory showed that in May 2017, of 359 UCs sent from various locations of the hospital, 38% were obtained in the setting of a negative UA.
A multidisciplinary group with representation from primary care, infectious diseases, pharmacy, nursing, laboratory, and informatics was created with a goal to improve the workup and management of UTIs. In addition to periodic education for the clinicians regarding appropriate use and interpretation of UA and UC along with judicious use of antimicrobials especially in the setting of ASB, a UA to reflex culture process change was implemented. This allowed for automatic cancellation of a UC in the setting of a negative UA, which was designed to help facilitate appropriate UC ordering.
Methods
The primary objective of this study was to compare the frequency of inappropriate UC use and inappropriate antibiotic prescribing pre- and postimplementation of this UA to reflex culture process change. An inappropriate UC was defined as a UC ordered despite a negative UA in asymptomatic patients. Inappropriate antibiotic prescribing was defined as treatment of patients with ASB. The secondary objective evaluated postintervention data to assess the frequency of outpatient, ED, and hospital visits for UTI-related symptoms in the group of patients that had a UC cancelled as a result of the new process change (within a 7-day period of the initial UA) to determine whether patients with true infections were missed due to the process change.
Study Design and Setting
This pre-post quality improvement (QI) study analyzed the UC-ordering practices for UTIs sent from the ED at the Hines VA. This VA is a 483-bed tertiary care hospital in Chicago, Illinois, and serves > 57,000 veterans and about 23,000 ED visits annually. This study was approved by the Edward Hines, Jr VA Institutional Review Board as a quality assurance/QI proposal prior to data collection.
Patient Selection
All patients who
When comparing postintervention data with preintervention data for the primary study objective, the same exclusion criteria from the 2015 study were applied to the present study, which excluded ED patients who were admitted for inpatient care, concurrent antibiotic therapy for a non-UTI indication, duplicate cultures, and use of chronic bladder management devices. All patients identified as receiving a UA during the specified postintervention study period were included for evaluation of the secondary study objective.
Interventions
After physician education, an ED process change was implemented on October 3, 2017. This process change involved the creation of new order sets in the EHR that allowed clinicians to order a UA only, a UA with culture that would be cancelled by laboratory personnel if the UA did not result in > 5 WBC/HPF, and a UA with culture designated as do not cancel, where the UC was processed regardless of the UA results. The scenarios in which the latter option was considered appropriate were listed on the ordering screen and included pregnancy, a genitourinary procedure with necessary preoperative culture, and neutropenia.
Measurements
Postimplementation, all UAs were reviewed and grouped as follows: (1) positive UA with subsequent UC; (2) negative UA, culture cancelled; (3) only UA ordered (no culture); or (4) do not cancel UC ordered. Of the UAs that were analyzed, the following data were collected: demographics, comorbidities, concurrent medications for benign prostatic hyperplasia (BPH) and/or overactive bladder (OAB), documented allergies/adverse drug reactions to antibiotics, date of ED visit, documented UTI signs/symptoms (defined as frequency, urgency, dysuria, fever, suprapubic pain, or altered mental status in patients unable to verbalize urinary symptoms), UC results and susceptibilities, number of UCs repeated within 7 days after initial UA, requirement of antibiotic for UTI within 7 days of initial UA, antibiotic prescribed, duration of antibiotic therapy, and outpatient visits, ED visits, or need for hospital admission within 7 days of the initial UA for UTI-related symptoms. Other relevant UA and UC data that could not be obtained from the EHR were collected by generating a report using the Veterans Information Systems and Technology Architecture (VistA).
Analysis
Statistical analysis was performed using SAS v9.4. Independent t tests and Fisher exact tests were used to describe difference pre- and postintervention. Statistical significance was considered for P < .05. Based on results from the previous study conducted at this facility in addition to a literature review, it was determined that 92 patients in each group (pre- and postintervention) would be necessary to detect a 15% increase in percentage of patients appropriately treated for a UTI.
Results
There were 684 UAs evaluated from ED visits, 429 preintervention and 255 postintervention. The 255 patients were evaluated for the secondary objective of the study. Of the 255 patients with UAs identified postintervention, 150 were excluded based on the predefined exclusion criteria, and the remaining 105 were compared with the 173 patients from the preintervention group and were included in the analysis for the primary objective (Figure 1).
Patients in the postintervention group were younger than those in the preintervention group (P < .02): otherwise the groups were similar (Table 1). Inappropriate antibiotics for ASB decreased from 10.2% preintervention to 1.9% postintervention (odds ratio, 0.17; P = .01) (Table 2). UC processing despite a negative UA significantly decreased from 100% preintervention to 38.6% postintervention (P < .001) (Table 3). In patients with a negative UA, antibiotic prescribing decreased by 25.3% postintervention, but this difference was not statistically significant.
Postintervention, of 255 UAs evaluated, 95 (37.3%) were positive with a processed UC and 95 (37.3%) were negative with UC cancelled, 43 (16.9%) were ordered as DNC, and 22 (8.6%) were ordered without a UC (Figure 2). Twenty-eight of the 95 (29.5%) UAs with processed UCs did not meet the criteria for a positive UA and were not designated as DNC. When the UCs of this subgroup of patients were further analyzed, we found that 2 of the cultures were positive of which 1 patient was symptomatic and required antibiotic therapy.
Of the 95 patients with a negative UA, 69 (72.6%) presented without any UTI-related symptoms. In this group, there were no reports of outpatient visits, ED visits, or hospital admissions within 7 days of initial UA for UTI-related symptoms. None of the UCs ordered as DNC had a supporting reason identified. Nonetheless, the UC results from this patient subgroup also were analyzed further and resulted in 4 patients with negative UA and positive subsequent UC, 1 was symptomatic and required antibiotic therapy.
Discussion
A simple process change at the Hines VA resulted in benefits related to antimicrobial stewardship without conferring adverse outcomes on patient safety. Both UC processing despite a negative UA and inappropriate antibiotic prescribing for ASB were reduced significantly postintervention. This process change was piloted in the ED where UCs are often included as part of the initial diagnostic testing in patients who may not report UTI-related symptoms but for whom a UC is often bundled with other infectious workup, depending on the patient presentation.
Reflex testing of urine specimens has been described in the literature, both in an exploratory nature where impact of a reflex UC cancellation protocol based on certain UA criteria is measured by percent reduction of UCs processed as well as results of such interventions implemented into clinical practice.11-13 A retrospective study performed at the University of North Carolina Medical Center evaluated patients who presented to the ED during a 6-month period and had both an automated UA and UC collected. UC processing was restricted to UA that was positive for nitrites, leukocyte esterase, bacteria, or > 10 WBC/HPF. Use of this reflex culture cancellation protocol could have eliminated 604 of the 1546 (39.1%) cultures processed. However, 11 of the 314 (3.5%) positive cultures could have been missed.13 This same protocol was externally validated at another large academic ED setting, where similar results were found.14
In clinical practice, there is a natural tendency to reflexively prescribe antibiotics based on the results of a positive UC due to the hesitancy in ignoring these results, despite lack of a suspicion for a true infection. Leis and colleagues explored this in a proof-of-concept study evaluating the impact of discontinuing the routine reporting of positive UC results from noncatheterized inpatients and requesting clinicians to call the laboratory for results if a UTI was suspected.16 This intervention resulted in a statistically significant reduction in treatment of ASB in noncatheterized patients from 48 to 12% pre- and postintervention. Clinicians requested culture results only 14% of the time, and there were no adverse outcomes among untreated noncatheterized patients. More recently, a QI study conducted at a large community hospital in Toronto, Ontario, Canada, implemented a 2-step model of care for urine collection.17 UC was collected but only processed by the microbiology laboratory if the ED physicians deemed it necessary after clinical assessment.
After implementation, there was a decrease in the proportion of ED visits associated with processed UC (from 6.0% to 4.7% of visits per week; P < .001), ED visits associated with callbacks for processing UC (1.8% to 1.1% of visits per month; P < .001), and antimicrobial prescriptions for urinary symptoms among hospitalized patients (from 20.6% to 10.9%; P < .001). Equally important, despite the 937 cases in which urine was collected but cultures were not processed, no evidence of untreated UTIs was identified.17
The results from the present study similarly demonstrate minimal concern for potentially undertreating these patients. As seen in the subgroup of patients included in the positive UA group, which did not meet criteria for positive UA per protocol (n = 29), only 2 of the subsequent cultures were positive, of which only 1 patient required antibiotic therapy based on the clinical presentation. In addition, in the group of negative UAs with subsequent cancellation of the UC, there were no found reports of outpatient visits, ED visits, or hospital admissions within 7 days of the initial UA for UTI-related symptoms.
Limitations
This single-center, pre-post QI study was not without limitations. Manual chart reviews were required, and accuracy of information was dependent on clinician documentation and assessment of UTI-related symptoms. The population studied was predominately older males; thus, results may not be applicable to females or young adults. Additionally, recognition of a negative UA and subsequent cancellation of the UC was dependent on laboratory personnel. As noted in the patient group with a positive UA, some of these UAs were negative and may have been overlooked; therefore, subsequent UCs were inappropriately processed. However, this occurred infrequently and confirmed the low probability of true UTI in the setting of a negative UA. Follow-up for UTI-related symptoms may not have been captured if a patient had presented to an outside facility. Last, definitions of a positive UA differed slightly between the pre- and postintervention groups. The preintervention study defined a positive UA as a WBC count > 5 WBC/HPF and positive leukocyte esterase, whereas the present study defined a positive UA with a WBC count > 5. This may have resulted in an overestimation of positive UA in the postintervention group.
Conclusions
Better selective use of UC testing may improve stewardship resources and reduce costs impacting both ED and clinical laboratories. Furthermore, benefits can include a reduction in the use of time and resources required to collect samples for culture, use of test supplies, the time and effort required to process the large number of negative cultures, and resources devoted to the follow-up of these ED culture results. The described UA to reflex culture process change demonstrated a significant reduction in the processing of inappropriate UC and unnecessary antibiotics for ASB. There were no missed UTIs or other adverse patient outcomes noted. This process change has been implemented in all departments at the Hines VA and additional data will be collected to ensure consistent outcomes.
1. Chironda B, Clancy S, Powis JE. Optimizing urine culture collection in the emergency department using frontline ownership interventions. Clin Infect Dis. 2014;59(7):1038-1039. doi:10.1093/cid/ciu412
2. Nagurney JT, Brown DF, Chang Y, Sane S, Wang AC, Weiner JB. Use of diagnostic testing in the emergency department for patients presenting with non-traumatic abdominal pain. J Emerg Med. 2003;25(4):363-371. doi:10.1016/s0736-4679(03)00237-3
3. Lammers RL, Gibson S, Kovacs D, Sears W, Strachan G. Comparison of test characteristics of urine dipstick and urinalysis at various test cutoff points. Ann Emerg Med. 2001;38(5):505-512. doi:10.1067/mem.2001.119427
4. Nicolle LE, Gupta K, Bradley SF, et al. Clinical practice guideline for the management of asymptomatic bacteriuria: 2019 update by the Infectious Diseases Society of America. Clin Infect Dis. 2019;68(10):1611-1615. doi:10.1093/cid/ciy1121
5. Trautner BW, Grigoryan L, Petersen NJ, et al. Effectiveness of an antimicrobial stewardship approach for urinary catheter-associated asymptomatic bacteriuria. JAMA Intern Med. 2015;175(7):1120-1127. doi:10.1001/jamainternmed.2015.1878
6. Hartley S, Valley S, Kuhn L, et al. Overtreatment of asymptomatic bacteriuria: identifying targets for improvement. Infect Control Hosp Epidemiol. 2015;36(4):470-473. doi:10.1017/ice.2014.73
7. Bader MS, Loeb M, Brooks AA. An update on the management of urinary tract infections in the era of antimicrobial resistance. Postgrad Med. 2017;129(2):242-258. doi:10.1080/00325481.2017.1246055
8. Spivak ES, Burk M, Zhang R, et al. Management of bacteriuria in Veterans Affairs hospitals. Clin Infect Dis. 2017;65(6):910-917. doi:10.1093/cid/cix474
9. Kim EY, Patel U, Patel B, Suda KJ. Evaluation of bacteriuria treatment and follow-up initiated in the emergency department at a Veterans Affairs hospital. J Pharm Technol. 2017;33(5):183-188. doi:10.1177/8755122517718214
10. Brown E, Talbot GH, Axelrod P, Provencher M, Hoegg C. Risk factors for Clostridium difficile toxin-associated diarrhea. Infect Control Hosp Epidemiol. 1990;11(6):283-290. doi:10.1086/646173
11. Fok C, Fitzgerald MP, Turk T, Mueller E, Dalaza L, Schreckenberger P. Reflex testing of male urine specimens misses few positive cultures may reduce unnecessary testing of normal specimens. Urology. 2010;75(1):74-76. doi:10.1016/j.urology.2009.08.071
12. Munigala S, Jackups RR Jr, Poirier RF, et al. Impact of order set design on urine culturing practices at an academic medical centre emergency department. BMJ Qual Saf. 2018;27(8):587-592. doi:10.1136/bmjqs-2017-006899
13. Jones CW, Culbreath KD, Mehrotra A, Gilligan PH. Reflect urine culture cancellation in the emergency department. J Emerg Med. 2014;46(1):71-76. doi:10.1016/j.jemermed.2013.08.042
14. Hertz JT, Lescallette RD, Barrett TW, Ward MJ, Self WH. External validation of an ED protocol for reflex urine culture cancelation. Am J Emerg Med. 2015;33(12):1838-1839. doi:10.1016/j.ajem.2015.09.026
15. Stamm WE. Measurement of pyuria and its relation to bacteriuria. Am J Med. 1983;75(1B):53-58. doi:10.1016/0002-9343(83)90073-6
16. Leis JA, Rebick GW, Daneman N, et al. Reducing antimicrobial therapy for asymptomatic bacteriuria among noncatheterized inpatients: a proof-of-concept study. Clin Infect Dis. 2014;58(7):980-983. doi:10.1093/cid/ciu010
17. Stagg A, Lutz H, Kirpalaney S, et al. Impact of two-step urine culture ordering in the emergency department: a time series analysis. BMJ Qual Saf. 2017;27:140-147. doi:10.1136/bmjqs-2016-006250
1. Chironda B, Clancy S, Powis JE. Optimizing urine culture collection in the emergency department using frontline ownership interventions. Clin Infect Dis. 2014;59(7):1038-1039. doi:10.1093/cid/ciu412
2. Nagurney JT, Brown DF, Chang Y, Sane S, Wang AC, Weiner JB. Use of diagnostic testing in the emergency department for patients presenting with non-traumatic abdominal pain. J Emerg Med. 2003;25(4):363-371. doi:10.1016/s0736-4679(03)00237-3
3. Lammers RL, Gibson S, Kovacs D, Sears W, Strachan G. Comparison of test characteristics of urine dipstick and urinalysis at various test cutoff points. Ann Emerg Med. 2001;38(5):505-512. doi:10.1067/mem.2001.119427
4. Nicolle LE, Gupta K, Bradley SF, et al. Clinical practice guideline for the management of asymptomatic bacteriuria: 2019 update by the Infectious Diseases Society of America. Clin Infect Dis. 2019;68(10):1611-1615. doi:10.1093/cid/ciy1121
5. Trautner BW, Grigoryan L, Petersen NJ, et al. Effectiveness of an antimicrobial stewardship approach for urinary catheter-associated asymptomatic bacteriuria. JAMA Intern Med. 2015;175(7):1120-1127. doi:10.1001/jamainternmed.2015.1878
6. Hartley S, Valley S, Kuhn L, et al. Overtreatment of asymptomatic bacteriuria: identifying targets for improvement. Infect Control Hosp Epidemiol. 2015;36(4):470-473. doi:10.1017/ice.2014.73
7. Bader MS, Loeb M, Brooks AA. An update on the management of urinary tract infections in the era of antimicrobial resistance. Postgrad Med. 2017;129(2):242-258. doi:10.1080/00325481.2017.1246055
8. Spivak ES, Burk M, Zhang R, et al. Management of bacteriuria in Veterans Affairs hospitals. Clin Infect Dis. 2017;65(6):910-917. doi:10.1093/cid/cix474
9. Kim EY, Patel U, Patel B, Suda KJ. Evaluation of bacteriuria treatment and follow-up initiated in the emergency department at a Veterans Affairs hospital. J Pharm Technol. 2017;33(5):183-188. doi:10.1177/8755122517718214
10. Brown E, Talbot GH, Axelrod P, Provencher M, Hoegg C. Risk factors for Clostridium difficile toxin-associated diarrhea. Infect Control Hosp Epidemiol. 1990;11(6):283-290. doi:10.1086/646173
11. Fok C, Fitzgerald MP, Turk T, Mueller E, Dalaza L, Schreckenberger P. Reflex testing of male urine specimens misses few positive cultures may reduce unnecessary testing of normal specimens. Urology. 2010;75(1):74-76. doi:10.1016/j.urology.2009.08.071
12. Munigala S, Jackups RR Jr, Poirier RF, et al. Impact of order set design on urine culturing practices at an academic medical centre emergency department. BMJ Qual Saf. 2018;27(8):587-592. doi:10.1136/bmjqs-2017-006899
13. Jones CW, Culbreath KD, Mehrotra A, Gilligan PH. Reflect urine culture cancellation in the emergency department. J Emerg Med. 2014;46(1):71-76. doi:10.1016/j.jemermed.2013.08.042
14. Hertz JT, Lescallette RD, Barrett TW, Ward MJ, Self WH. External validation of an ED protocol for reflex urine culture cancelation. Am J Emerg Med. 2015;33(12):1838-1839. doi:10.1016/j.ajem.2015.09.026
15. Stamm WE. Measurement of pyuria and its relation to bacteriuria. Am J Med. 1983;75(1B):53-58. doi:10.1016/0002-9343(83)90073-6
16. Leis JA, Rebick GW, Daneman N, et al. Reducing antimicrobial therapy for asymptomatic bacteriuria among noncatheterized inpatients: a proof-of-concept study. Clin Infect Dis. 2014;58(7):980-983. doi:10.1093/cid/ciu010
17. Stagg A, Lutz H, Kirpalaney S, et al. Impact of two-step urine culture ordering in the emergency department: a time series analysis. BMJ Qual Saf. 2017;27:140-147. doi:10.1136/bmjqs-2016-006250
Analysis: Surgery may not be better than casting for some wrist fractures
There are around 100,000 adult distal radius fractures in the United Kingdom each year. Current National Health Service guidelines in the United Kingdom recommend using K-wires to stabilize wrist fractures when closed reduction is possible or there is no involvement of the articular surface. This is in contrast to fractures that require open reduction and internal fixation with a plate and screws to align the joint articular surface.
As a result, the use of K-wires for surgical fixation has been increasing since 2010 with a comparable decrease of the use of plates and screws.
Even though fixation with wires can provide reliable functional outcomes for patients after reduction of a displaced wrist fracture, surgery still carries risks for the patient and adds an additional expense. A well-molded plaster cast is a safer and cheaper intervention, but it is unclear if it could provide the same functional outcome as pinning.
Therefore, researchers in the United Kingdom conducted a multicenter, randomized trial among 36 hospitals within the NHS as part of the Distal Radius Acute Fracture Fixation Trial 2 (DRAFFT2). Investigators randomly assigned 500 patients aged 16 years and older with dorsally displaced distal radius fractures to manipulation followed by a molded cast or manipulation followed by surgical fixation with K-wires plus a cast.
The study was published online in BMJ.
At 1 year, there were no significant differences between the groups in Patient-Rated Wrist Evaluation (PRWE) scores centered on pain and function.
In an interview, Matthew Costa, PhD, professor of orthopaedic trauma at the University of Oxford (England) and the study’s lead author, said, “If a closed reduction of the fracture can be achieved, clinicians may consider the application of a molded plaster cast as a safe and cost-effective alternative to surgical fixation.”
However, in referencing the data his group published, he did find one thing surprising: “One in eight patients treated with a molded cast required later surgery for loss of fracture position in the first 6 weeks after their injury.”
Dr. Costa added, “This was indeed the key bit of information that patients need when making their decision about surgery. Initial feedback from our patient and public involvement group is that they would be happy to take this chance given that seven out of eight patients didn’t need any form of surgical fixation.”
Philip Blazar, MD, chief of the hand and upper extremity service, Brigham and Women’s Faulkner Hospital in Boston, commended the U.K. authors on completing a challenging randomized controlled trial.
Speaking to this news organization, Dr. Blazer observed a critical difference between U.K. and U.S. guidelines. “It is important to remember that a sizable number of these patients had surgery,” said Dr. Blazar, who was not involved with the study. “They had pins inserted under an anesthetic, and would not have [had] surgery compared to current practice as recommended by many authorities, including the American Academy of Orthopedic Surgeon’s Clinical Practice Guidelines on Distal Radius Fractures.”
Like Dr. Costa, Dr. Blazar expressed concerns about the secondary surgeries in the study group: “27% of patients had a second surgery: 13% in the first 6 weeks after manipulation for loss of reduction, and the remaining 14% had carpal tunnel releases, tendon transfers, tenolysis, and/or capsulectomy for limited range of motion.”
In addition, Dr. Blazar is worried that, although recovery is generally considered to be only 12 months for these type of injuries – the duration of follow-up time in the DRAFFT2 study – “the probable outcome is that in the second 12 months after the injury, there will continue to be more of these types of surgeries.”
Dr. Costa agreed that close follow-up is warranted, “It does suggest that patients treated in a molded cast do need to be followed up carefully to spot those that do need later surgery.”
Still, for Dr. Blazar, the largest takeaway of the study is that, “At 12 months, disability scores between these two groups are not different, but the group treated nonsurgically had 10 times the number of secondary surgeries (27% vs. 2%-3%).”
Moving forward, Dr. Blazar would like to see more specific indications for who would benefit from pinning. He told this news organization, “The greatest limitation is that this study provides no information on which patients with distal radius fractures where reduction is indicated would benefit from surgery. Looking at the details of this study, all patients with displaced fractures from age 16 to the elderly were treated as one indication. My impression is that most surgeons operate on patients taking into account radiographic and patient factors such as age, hand dominance, occupation, overall medical health, and activity level.”
The DRAFFT2 study was funded by the U.K. National Institute for Health Research Health Technology Assessment Programme and was supported by NIHR Oxford Biomedical Research Centre. Dr. Blazar and Dr. Costa have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
There are around 100,000 adult distal radius fractures in the United Kingdom each year. Current National Health Service guidelines in the United Kingdom recommend using K-wires to stabilize wrist fractures when closed reduction is possible or there is no involvement of the articular surface. This is in contrast to fractures that require open reduction and internal fixation with a plate and screws to align the joint articular surface.
As a result, the use of K-wires for surgical fixation has been increasing since 2010 with a comparable decrease of the use of plates and screws.
Even though fixation with wires can provide reliable functional outcomes for patients after reduction of a displaced wrist fracture, surgery still carries risks for the patient and adds an additional expense. A well-molded plaster cast is a safer and cheaper intervention, but it is unclear if it could provide the same functional outcome as pinning.
Therefore, researchers in the United Kingdom conducted a multicenter, randomized trial among 36 hospitals within the NHS as part of the Distal Radius Acute Fracture Fixation Trial 2 (DRAFFT2). Investigators randomly assigned 500 patients aged 16 years and older with dorsally displaced distal radius fractures to manipulation followed by a molded cast or manipulation followed by surgical fixation with K-wires plus a cast.
The study was published online in BMJ.
At 1 year, there were no significant differences between the groups in Patient-Rated Wrist Evaluation (PRWE) scores centered on pain and function.
In an interview, Matthew Costa, PhD, professor of orthopaedic trauma at the University of Oxford (England) and the study’s lead author, said, “If a closed reduction of the fracture can be achieved, clinicians may consider the application of a molded plaster cast as a safe and cost-effective alternative to surgical fixation.”
However, in referencing the data his group published, he did find one thing surprising: “One in eight patients treated with a molded cast required later surgery for loss of fracture position in the first 6 weeks after their injury.”
Dr. Costa added, “This was indeed the key bit of information that patients need when making their decision about surgery. Initial feedback from our patient and public involvement group is that they would be happy to take this chance given that seven out of eight patients didn’t need any form of surgical fixation.”
Philip Blazar, MD, chief of the hand and upper extremity service, Brigham and Women’s Faulkner Hospital in Boston, commended the U.K. authors on completing a challenging randomized controlled trial.
Speaking to this news organization, Dr. Blazer observed a critical difference between U.K. and U.S. guidelines. “It is important to remember that a sizable number of these patients had surgery,” said Dr. Blazar, who was not involved with the study. “They had pins inserted under an anesthetic, and would not have [had] surgery compared to current practice as recommended by many authorities, including the American Academy of Orthopedic Surgeon’s Clinical Practice Guidelines on Distal Radius Fractures.”
Like Dr. Costa, Dr. Blazar expressed concerns about the secondary surgeries in the study group: “27% of patients had a second surgery: 13% in the first 6 weeks after manipulation for loss of reduction, and the remaining 14% had carpal tunnel releases, tendon transfers, tenolysis, and/or capsulectomy for limited range of motion.”
In addition, Dr. Blazar is worried that, although recovery is generally considered to be only 12 months for these type of injuries – the duration of follow-up time in the DRAFFT2 study – “the probable outcome is that in the second 12 months after the injury, there will continue to be more of these types of surgeries.”
Dr. Costa agreed that close follow-up is warranted, “It does suggest that patients treated in a molded cast do need to be followed up carefully to spot those that do need later surgery.”
Still, for Dr. Blazar, the largest takeaway of the study is that, “At 12 months, disability scores between these two groups are not different, but the group treated nonsurgically had 10 times the number of secondary surgeries (27% vs. 2%-3%).”
Moving forward, Dr. Blazar would like to see more specific indications for who would benefit from pinning. He told this news organization, “The greatest limitation is that this study provides no information on which patients with distal radius fractures where reduction is indicated would benefit from surgery. Looking at the details of this study, all patients with displaced fractures from age 16 to the elderly were treated as one indication. My impression is that most surgeons operate on patients taking into account radiographic and patient factors such as age, hand dominance, occupation, overall medical health, and activity level.”
The DRAFFT2 study was funded by the U.K. National Institute for Health Research Health Technology Assessment Programme and was supported by NIHR Oxford Biomedical Research Centre. Dr. Blazar and Dr. Costa have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
There are around 100,000 adult distal radius fractures in the United Kingdom each year. Current National Health Service guidelines in the United Kingdom recommend using K-wires to stabilize wrist fractures when closed reduction is possible or there is no involvement of the articular surface. This is in contrast to fractures that require open reduction and internal fixation with a plate and screws to align the joint articular surface.
As a result, the use of K-wires for surgical fixation has been increasing since 2010 with a comparable decrease of the use of plates and screws.
Even though fixation with wires can provide reliable functional outcomes for patients after reduction of a displaced wrist fracture, surgery still carries risks for the patient and adds an additional expense. A well-molded plaster cast is a safer and cheaper intervention, but it is unclear if it could provide the same functional outcome as pinning.
Therefore, researchers in the United Kingdom conducted a multicenter, randomized trial among 36 hospitals within the NHS as part of the Distal Radius Acute Fracture Fixation Trial 2 (DRAFFT2). Investigators randomly assigned 500 patients aged 16 years and older with dorsally displaced distal radius fractures to manipulation followed by a molded cast or manipulation followed by surgical fixation with K-wires plus a cast.
The study was published online in BMJ.
At 1 year, there were no significant differences between the groups in Patient-Rated Wrist Evaluation (PRWE) scores centered on pain and function.
In an interview, Matthew Costa, PhD, professor of orthopaedic trauma at the University of Oxford (England) and the study’s lead author, said, “If a closed reduction of the fracture can be achieved, clinicians may consider the application of a molded plaster cast as a safe and cost-effective alternative to surgical fixation.”
However, in referencing the data his group published, he did find one thing surprising: “One in eight patients treated with a molded cast required later surgery for loss of fracture position in the first 6 weeks after their injury.”
Dr. Costa added, “This was indeed the key bit of information that patients need when making their decision about surgery. Initial feedback from our patient and public involvement group is that they would be happy to take this chance given that seven out of eight patients didn’t need any form of surgical fixation.”
Philip Blazar, MD, chief of the hand and upper extremity service, Brigham and Women’s Faulkner Hospital in Boston, commended the U.K. authors on completing a challenging randomized controlled trial.
Speaking to this news organization, Dr. Blazer observed a critical difference between U.K. and U.S. guidelines. “It is important to remember that a sizable number of these patients had surgery,” said Dr. Blazar, who was not involved with the study. “They had pins inserted under an anesthetic, and would not have [had] surgery compared to current practice as recommended by many authorities, including the American Academy of Orthopedic Surgeon’s Clinical Practice Guidelines on Distal Radius Fractures.”
Like Dr. Costa, Dr. Blazar expressed concerns about the secondary surgeries in the study group: “27% of patients had a second surgery: 13% in the first 6 weeks after manipulation for loss of reduction, and the remaining 14% had carpal tunnel releases, tendon transfers, tenolysis, and/or capsulectomy for limited range of motion.”
In addition, Dr. Blazar is worried that, although recovery is generally considered to be only 12 months for these type of injuries – the duration of follow-up time in the DRAFFT2 study – “the probable outcome is that in the second 12 months after the injury, there will continue to be more of these types of surgeries.”
Dr. Costa agreed that close follow-up is warranted, “It does suggest that patients treated in a molded cast do need to be followed up carefully to spot those that do need later surgery.”
Still, for Dr. Blazar, the largest takeaway of the study is that, “At 12 months, disability scores between these two groups are not different, but the group treated nonsurgically had 10 times the number of secondary surgeries (27% vs. 2%-3%).”
Moving forward, Dr. Blazar would like to see more specific indications for who would benefit from pinning. He told this news organization, “The greatest limitation is that this study provides no information on which patients with distal radius fractures where reduction is indicated would benefit from surgery. Looking at the details of this study, all patients with displaced fractures from age 16 to the elderly were treated as one indication. My impression is that most surgeons operate on patients taking into account radiographic and patient factors such as age, hand dominance, occupation, overall medical health, and activity level.”
The DRAFFT2 study was funded by the U.K. National Institute for Health Research Health Technology Assessment Programme and was supported by NIHR Oxford Biomedical Research Centre. Dr. Blazar and Dr. Costa have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM BMJ
Orthopedists rank third in malpractice suits, survey finds
according to the Medscape Orthopedist Malpractice Report 2021.
Orthopedists ranked third among specialists most likely to be sued, surpassed only by plastic surgeons and general surgeons (both 83%). In comparison, just over half of physicians across all specialties (51%) reported being named in lawsuit. More than one-third of orthopedists (34%) said they had been individually named in a suit, whereas just 14% of all specialists were named individually.
More than half (54%) of orthopedists said they were sued over complications from treatment or surgery. The second-most common reason orthopedists were sued was poor outcome/disease progression (30%), followed by failure to diagnose/delayed diagnosis (21%), failure to treat/delayed treatment (13%), and abnormal injury (9%).
This new report was compiled from an online survey including more than 4,300 physicians from 29 specialties. The survey was available from May 21 to Aug. 28, 2021, and included 250 orthopedists and orthopedic surgeons. Most respondents (62%) had practiced orthopedics for more than 25 years and 60% were aged 60 years or older.
Orthopedists tended to pay more for malpractice insurance than do other specialists. Less than one-third of orthopedists (31%) reported a premium under $20,000 per year, compared with 52% of all specialists. The most common premium for orthopedists was $30,000 or more (29%), whereas only 11% of all specialists reported paying a similar premium.
Nearly 9 out of 10 (89%) of orthopedists said they were “very surprised” or “somewhat surprised” by the malpractice suit. In some of these cases, the physician never personally treated the patient. Wrote one respondent: “I was part of a group of physicians and got dragged into the suit.” The vast majority of orthopedists (82%) said the suit was not warranted, which was similar to responses for physicians as a whole (83%).
Most commonly, orthopedists said lawsuits were settled before trial (34%). The second-most common outcome was the judge and jury deciding in the respondent’s favor (16%), followed by the plaintiff voluntarily dismissing the suit prior to trial (8%), and the respondent being dismissed from the suit in the first few months (8%). Very few (2%) said the judge or jury ruled in the patient’s favor, and 9% of respondents said the case was ongoing.
Most orthopedists reported that cases lasted between 1 and 2 years (41%) and 29% said a lawsuit took 3-5 years. If the plaintiff did receive a monetary award, 42% of physicians reported paying under $100,000, and 30% paid less than $500,000. This is similar to reports from other specialties, though more patients in orthopedic cases received payments under $1 million, compared with other specialties (21% vs. 15%).
More than three-quarters of orthopedists (76%) said that the lawsuit did not negatively affect their career, and more than half (52%) said they did not undergo any attitude or career changes after the suit. More orthopedists than other specialists (31% vs. 24%) did say that they trusted patients less.
When asked if they would do anything differently, one-third (33%) of orthopedists said their actions would remain the same, compared with 43% of the general physician pool. One-quarter of orthopedists said they would have not taken on the patient in the first place, and 14% noted they would have referred to another physician.
A version of this article first appeared on Medscape.com.
according to the Medscape Orthopedist Malpractice Report 2021.
Orthopedists ranked third among specialists most likely to be sued, surpassed only by plastic surgeons and general surgeons (both 83%). In comparison, just over half of physicians across all specialties (51%) reported being named in lawsuit. More than one-third of orthopedists (34%) said they had been individually named in a suit, whereas just 14% of all specialists were named individually.
More than half (54%) of orthopedists said they were sued over complications from treatment or surgery. The second-most common reason orthopedists were sued was poor outcome/disease progression (30%), followed by failure to diagnose/delayed diagnosis (21%), failure to treat/delayed treatment (13%), and abnormal injury (9%).
This new report was compiled from an online survey including more than 4,300 physicians from 29 specialties. The survey was available from May 21 to Aug. 28, 2021, and included 250 orthopedists and orthopedic surgeons. Most respondents (62%) had practiced orthopedics for more than 25 years and 60% were aged 60 years or older.
Orthopedists tended to pay more for malpractice insurance than do other specialists. Less than one-third of orthopedists (31%) reported a premium under $20,000 per year, compared with 52% of all specialists. The most common premium for orthopedists was $30,000 or more (29%), whereas only 11% of all specialists reported paying a similar premium.
Nearly 9 out of 10 (89%) of orthopedists said they were “very surprised” or “somewhat surprised” by the malpractice suit. In some of these cases, the physician never personally treated the patient. Wrote one respondent: “I was part of a group of physicians and got dragged into the suit.” The vast majority of orthopedists (82%) said the suit was not warranted, which was similar to responses for physicians as a whole (83%).
Most commonly, orthopedists said lawsuits were settled before trial (34%). The second-most common outcome was the judge and jury deciding in the respondent’s favor (16%), followed by the plaintiff voluntarily dismissing the suit prior to trial (8%), and the respondent being dismissed from the suit in the first few months (8%). Very few (2%) said the judge or jury ruled in the patient’s favor, and 9% of respondents said the case was ongoing.
Most orthopedists reported that cases lasted between 1 and 2 years (41%) and 29% said a lawsuit took 3-5 years. If the plaintiff did receive a monetary award, 42% of physicians reported paying under $100,000, and 30% paid less than $500,000. This is similar to reports from other specialties, though more patients in orthopedic cases received payments under $1 million, compared with other specialties (21% vs. 15%).
More than three-quarters of orthopedists (76%) said that the lawsuit did not negatively affect their career, and more than half (52%) said they did not undergo any attitude or career changes after the suit. More orthopedists than other specialists (31% vs. 24%) did say that they trusted patients less.
When asked if they would do anything differently, one-third (33%) of orthopedists said their actions would remain the same, compared with 43% of the general physician pool. One-quarter of orthopedists said they would have not taken on the patient in the first place, and 14% noted they would have referred to another physician.
A version of this article first appeared on Medscape.com.
according to the Medscape Orthopedist Malpractice Report 2021.
Orthopedists ranked third among specialists most likely to be sued, surpassed only by plastic surgeons and general surgeons (both 83%). In comparison, just over half of physicians across all specialties (51%) reported being named in lawsuit. More than one-third of orthopedists (34%) said they had been individually named in a suit, whereas just 14% of all specialists were named individually.
More than half (54%) of orthopedists said they were sued over complications from treatment or surgery. The second-most common reason orthopedists were sued was poor outcome/disease progression (30%), followed by failure to diagnose/delayed diagnosis (21%), failure to treat/delayed treatment (13%), and abnormal injury (9%).
This new report was compiled from an online survey including more than 4,300 physicians from 29 specialties. The survey was available from May 21 to Aug. 28, 2021, and included 250 orthopedists and orthopedic surgeons. Most respondents (62%) had practiced orthopedics for more than 25 years and 60% were aged 60 years or older.
Orthopedists tended to pay more for malpractice insurance than do other specialists. Less than one-third of orthopedists (31%) reported a premium under $20,000 per year, compared with 52% of all specialists. The most common premium for orthopedists was $30,000 or more (29%), whereas only 11% of all specialists reported paying a similar premium.
Nearly 9 out of 10 (89%) of orthopedists said they were “very surprised” or “somewhat surprised” by the malpractice suit. In some of these cases, the physician never personally treated the patient. Wrote one respondent: “I was part of a group of physicians and got dragged into the suit.” The vast majority of orthopedists (82%) said the suit was not warranted, which was similar to responses for physicians as a whole (83%).
Most commonly, orthopedists said lawsuits were settled before trial (34%). The second-most common outcome was the judge and jury deciding in the respondent’s favor (16%), followed by the plaintiff voluntarily dismissing the suit prior to trial (8%), and the respondent being dismissed from the suit in the first few months (8%). Very few (2%) said the judge or jury ruled in the patient’s favor, and 9% of respondents said the case was ongoing.
Most orthopedists reported that cases lasted between 1 and 2 years (41%) and 29% said a lawsuit took 3-5 years. If the plaintiff did receive a monetary award, 42% of physicians reported paying under $100,000, and 30% paid less than $500,000. This is similar to reports from other specialties, though more patients in orthopedic cases received payments under $1 million, compared with other specialties (21% vs. 15%).
More than three-quarters of orthopedists (76%) said that the lawsuit did not negatively affect their career, and more than half (52%) said they did not undergo any attitude or career changes after the suit. More orthopedists than other specialists (31% vs. 24%) did say that they trusted patients less.
When asked if they would do anything differently, one-third (33%) of orthopedists said their actions would remain the same, compared with 43% of the general physician pool. One-quarter of orthopedists said they would have not taken on the patient in the first place, and 14% noted they would have referred to another physician.
A version of this article first appeared on Medscape.com.
We’re dying to tell you about fatigability
Are you tired? Or are you death tired?
When we’re feeling that burnout monster creep in we sometimes say that we’re being worked to death or that we’re dead tired, but what if that feeling could predict when it’s your actual time to go?
In a recent study published in the Journals of Gerontology: Series A, epidemiologists from the University of Pittsburgh were able to associate a level of “physical fatigability” with mortality.
The researchers administered the Pittsburgh Fatigability Scale to almost 3,000 participants aged ≥ 60 years, who ranked from 0 to 5 on how tired they thought they would be after doing activities like light housework or a leisurely 30-minute walk. After accounting for factors such as preexisting conditions and mental health, the researchers found that people who scored 25 or more points were 2.3 times more likely to die in the next 2.7 years, compared with those who scored under 25.
So what does that tell us about the importance of being continuously active? It’s pretty important.
“Previous research indicates that getting more physical activity can reduce a person’s fatigability. Our study is the first to link more severe physical fatigability to an earlier death,” lead author Nancy W. Glynn, PhD, said in a separate statement. The best way to keep physically active, she suggested, is to set manageable goals and a routine.
A nice walk around the neighborhood during golden hour or a little bit of yoga before breakfast could be a great way to keep the body moving, because you know what they say: Use it or lose it.
This work is NFT protected: Do not screenshot
If you’ve been following the nonmedical news, you’ve likely heard the term “NFT” explode in the past few months. Standing for nonfungible token, NFTs are, at least theoretically, a proof of ownership for digital creations that prevents anyone other than the buyer from reselling the artwork. Sounds like a great idea: It protects artists and buyers alike.
Much like its cousin cryptocurrency, however, the NFT world is rife with speculation, scams, misunderstanding, and drawings of bored monkeys. It’s the Wild West out there in the digital art universe: One poor unfortunate accidentally sold a $300k NFT image for $3,000, a group of investors spent $3 million buying an NFT for a rare version of Dune believing it gave them the copyright (it did not), and an Indonesian engineering student’s 5-year series of expressionless selfies is now worth a million dollars.
This is a column detailing weird medical news, however, so with our setup complete (though our understanding of NFTs is very much not), we move to France and meet our hero (?), Emmanuel Masmejean, an orthopedic surgeon who apparently wasn’t making enough money in his lucrative medical career.
In a move of apocalyptic madness, he threw ethics out the window, delved into his archive, and found an x-ray of a young woman with a bullet lodged in her arm. The woman was a survivor of the Bataclan mass shooting and bombing in 2015, and don’t you worry, our intrepid entrepreneur made sure to identify her as such when he tried selling the x-ray as an NFT on an online art website for $2,776. Yes, this is very much a violation of doctor-patient confidentiality, and no, that’s not a lot of money to risk your medical career on.
Naturally, the woman was horrified and shocked to learn that the image was being sold, her lawyer told the Guardian. When the doctor called her, he merely attempted to justify his action, rather than apologizing or showing any remorse. Dr. Masmejean is now facing legal action and a disciplinary charge for his attempted entry into the NFT world for publishing the image without permission, and the NFT has been removed from the website. Should have stuck with the bored monkeys.
Avatars could be the future
Zoom, FaceTime, and Skype are great when people can’t be together in the same room, state, or country. Not the same as being somewhere in person, but a pretty good replacement during a global pandemic. But what if you had a robot that could be present for you?
Seven-year-old Joshua Martinangeli of Berlin has a severe lung disease and needs to wear a tube in his neck, so he cannot attend school. A robot avatar, donated to Joshua through a private initiative, sits in his seat in the classroom and is able to interact with the students and teacher, according to Reuters. A light on the avatar blinks when Joshua wants to speak and the children can talk with him too. Joshua and his classmates agree that it’s not the same as him really being there to talk and learn, but it’s a great way to keep him included.
“We are the only district in Berlin that has bought four avatars for its schools. The impetus was COVID-19, but I think this will be the future well beyond the pandemic,” Torsten Kuehne, district education councilor, told Reuters.
So where do we get an avatar to go out and run errands? Can we send it to the office instead of Zooming the next meeting? Or maybe our avatar could go to the gym for us. But how do we get the results to show up on our bodies? C’mon science, figure this out.
Futility, thy name is Kiribati
Before we get to the rest of our regularly scheduled hilarity, a brief geography lesson is in order: Kiribati is an island nation – actually 32 atolls and one coral island – in the central Pacific Ocean. Those atolls are spread out across 1.4 million square miles around the intersection of the equator and the International Date Line, so Kiribati is the only country in the world located in all four hemispheres.
Now, back to the news.
Kiribati closed its borders early in the COVID-19 pandemic and recorded only two cases in almost 2 years. Things were going so well that the authorities recently decided to reopen the country to international travelers. Silly authorities.
The first plane was set to arrive on Jan. 14 from Fiji. This being the age of COVID, plans were made and precautions were taken. All 54 passengers quarantined for 2 weeks before the flight and underwent regular testing, the Guardian noted, and “they were only allowed on the flight after returning negative tests.”
You guessed it. Two-thirds of those 54 people tested positive for COVID-19 after landing in Kiribati.
All of the passengers were quarantined, but since then a security guard at the quarantine center has tested positive, as has someone who was not involved in the quarantine. According to NPR, the government said that “there is now an assumption that COVID-19 is now spreading in the community on South Tarawa and Betio.”
Moral of the story? You can’t beat COVID, so never try.
[EDITOR: Is that really the message we want to send to our readers?]
If you can’t beat them, join them.
[EDITOR: Nope. Try again.]
Resistance is futile?
[EDITOR: Sigh. Close enough.]
Are you tired? Or are you death tired?
When we’re feeling that burnout monster creep in we sometimes say that we’re being worked to death or that we’re dead tired, but what if that feeling could predict when it’s your actual time to go?
In a recent study published in the Journals of Gerontology: Series A, epidemiologists from the University of Pittsburgh were able to associate a level of “physical fatigability” with mortality.
The researchers administered the Pittsburgh Fatigability Scale to almost 3,000 participants aged ≥ 60 years, who ranked from 0 to 5 on how tired they thought they would be after doing activities like light housework or a leisurely 30-minute walk. After accounting for factors such as preexisting conditions and mental health, the researchers found that people who scored 25 or more points were 2.3 times more likely to die in the next 2.7 years, compared with those who scored under 25.
So what does that tell us about the importance of being continuously active? It’s pretty important.
“Previous research indicates that getting more physical activity can reduce a person’s fatigability. Our study is the first to link more severe physical fatigability to an earlier death,” lead author Nancy W. Glynn, PhD, said in a separate statement. The best way to keep physically active, she suggested, is to set manageable goals and a routine.
A nice walk around the neighborhood during golden hour or a little bit of yoga before breakfast could be a great way to keep the body moving, because you know what they say: Use it or lose it.
This work is NFT protected: Do not screenshot
If you’ve been following the nonmedical news, you’ve likely heard the term “NFT” explode in the past few months. Standing for nonfungible token, NFTs are, at least theoretically, a proof of ownership for digital creations that prevents anyone other than the buyer from reselling the artwork. Sounds like a great idea: It protects artists and buyers alike.
Much like its cousin cryptocurrency, however, the NFT world is rife with speculation, scams, misunderstanding, and drawings of bored monkeys. It’s the Wild West out there in the digital art universe: One poor unfortunate accidentally sold a $300k NFT image for $3,000, a group of investors spent $3 million buying an NFT for a rare version of Dune believing it gave them the copyright (it did not), and an Indonesian engineering student’s 5-year series of expressionless selfies is now worth a million dollars.
This is a column detailing weird medical news, however, so with our setup complete (though our understanding of NFTs is very much not), we move to France and meet our hero (?), Emmanuel Masmejean, an orthopedic surgeon who apparently wasn’t making enough money in his lucrative medical career.
In a move of apocalyptic madness, he threw ethics out the window, delved into his archive, and found an x-ray of a young woman with a bullet lodged in her arm. The woman was a survivor of the Bataclan mass shooting and bombing in 2015, and don’t you worry, our intrepid entrepreneur made sure to identify her as such when he tried selling the x-ray as an NFT on an online art website for $2,776. Yes, this is very much a violation of doctor-patient confidentiality, and no, that’s not a lot of money to risk your medical career on.
Naturally, the woman was horrified and shocked to learn that the image was being sold, her lawyer told the Guardian. When the doctor called her, he merely attempted to justify his action, rather than apologizing or showing any remorse. Dr. Masmejean is now facing legal action and a disciplinary charge for his attempted entry into the NFT world for publishing the image without permission, and the NFT has been removed from the website. Should have stuck with the bored monkeys.
Avatars could be the future
Zoom, FaceTime, and Skype are great when people can’t be together in the same room, state, or country. Not the same as being somewhere in person, but a pretty good replacement during a global pandemic. But what if you had a robot that could be present for you?
Seven-year-old Joshua Martinangeli of Berlin has a severe lung disease and needs to wear a tube in his neck, so he cannot attend school. A robot avatar, donated to Joshua through a private initiative, sits in his seat in the classroom and is able to interact with the students and teacher, according to Reuters. A light on the avatar blinks when Joshua wants to speak and the children can talk with him too. Joshua and his classmates agree that it’s not the same as him really being there to talk and learn, but it’s a great way to keep him included.
“We are the only district in Berlin that has bought four avatars for its schools. The impetus was COVID-19, but I think this will be the future well beyond the pandemic,” Torsten Kuehne, district education councilor, told Reuters.
So where do we get an avatar to go out and run errands? Can we send it to the office instead of Zooming the next meeting? Or maybe our avatar could go to the gym for us. But how do we get the results to show up on our bodies? C’mon science, figure this out.
Futility, thy name is Kiribati
Before we get to the rest of our regularly scheduled hilarity, a brief geography lesson is in order: Kiribati is an island nation – actually 32 atolls and one coral island – in the central Pacific Ocean. Those atolls are spread out across 1.4 million square miles around the intersection of the equator and the International Date Line, so Kiribati is the only country in the world located in all four hemispheres.
Now, back to the news.
Kiribati closed its borders early in the COVID-19 pandemic and recorded only two cases in almost 2 years. Things were going so well that the authorities recently decided to reopen the country to international travelers. Silly authorities.
The first plane was set to arrive on Jan. 14 from Fiji. This being the age of COVID, plans were made and precautions were taken. All 54 passengers quarantined for 2 weeks before the flight and underwent regular testing, the Guardian noted, and “they were only allowed on the flight after returning negative tests.”
You guessed it. Two-thirds of those 54 people tested positive for COVID-19 after landing in Kiribati.
All of the passengers were quarantined, but since then a security guard at the quarantine center has tested positive, as has someone who was not involved in the quarantine. According to NPR, the government said that “there is now an assumption that COVID-19 is now spreading in the community on South Tarawa and Betio.”
Moral of the story? You can’t beat COVID, so never try.
[EDITOR: Is that really the message we want to send to our readers?]
If you can’t beat them, join them.
[EDITOR: Nope. Try again.]
Resistance is futile?
[EDITOR: Sigh. Close enough.]
Are you tired? Or are you death tired?
When we’re feeling that burnout monster creep in we sometimes say that we’re being worked to death or that we’re dead tired, but what if that feeling could predict when it’s your actual time to go?
In a recent study published in the Journals of Gerontology: Series A, epidemiologists from the University of Pittsburgh were able to associate a level of “physical fatigability” with mortality.
The researchers administered the Pittsburgh Fatigability Scale to almost 3,000 participants aged ≥ 60 years, who ranked from 0 to 5 on how tired they thought they would be after doing activities like light housework or a leisurely 30-minute walk. After accounting for factors such as preexisting conditions and mental health, the researchers found that people who scored 25 or more points were 2.3 times more likely to die in the next 2.7 years, compared with those who scored under 25.
So what does that tell us about the importance of being continuously active? It’s pretty important.
“Previous research indicates that getting more physical activity can reduce a person’s fatigability. Our study is the first to link more severe physical fatigability to an earlier death,” lead author Nancy W. Glynn, PhD, said in a separate statement. The best way to keep physically active, she suggested, is to set manageable goals and a routine.
A nice walk around the neighborhood during golden hour or a little bit of yoga before breakfast could be a great way to keep the body moving, because you know what they say: Use it or lose it.
This work is NFT protected: Do not screenshot
If you’ve been following the nonmedical news, you’ve likely heard the term “NFT” explode in the past few months. Standing for nonfungible token, NFTs are, at least theoretically, a proof of ownership for digital creations that prevents anyone other than the buyer from reselling the artwork. Sounds like a great idea: It protects artists and buyers alike.
Much like its cousin cryptocurrency, however, the NFT world is rife with speculation, scams, misunderstanding, and drawings of bored monkeys. It’s the Wild West out there in the digital art universe: One poor unfortunate accidentally sold a $300k NFT image for $3,000, a group of investors spent $3 million buying an NFT for a rare version of Dune believing it gave them the copyright (it did not), and an Indonesian engineering student’s 5-year series of expressionless selfies is now worth a million dollars.
This is a column detailing weird medical news, however, so with our setup complete (though our understanding of NFTs is very much not), we move to France and meet our hero (?), Emmanuel Masmejean, an orthopedic surgeon who apparently wasn’t making enough money in his lucrative medical career.
In a move of apocalyptic madness, he threw ethics out the window, delved into his archive, and found an x-ray of a young woman with a bullet lodged in her arm. The woman was a survivor of the Bataclan mass shooting and bombing in 2015, and don’t you worry, our intrepid entrepreneur made sure to identify her as such when he tried selling the x-ray as an NFT on an online art website for $2,776. Yes, this is very much a violation of doctor-patient confidentiality, and no, that’s not a lot of money to risk your medical career on.
Naturally, the woman was horrified and shocked to learn that the image was being sold, her lawyer told the Guardian. When the doctor called her, he merely attempted to justify his action, rather than apologizing or showing any remorse. Dr. Masmejean is now facing legal action and a disciplinary charge for his attempted entry into the NFT world for publishing the image without permission, and the NFT has been removed from the website. Should have stuck with the bored monkeys.
Avatars could be the future
Zoom, FaceTime, and Skype are great when people can’t be together in the same room, state, or country. Not the same as being somewhere in person, but a pretty good replacement during a global pandemic. But what if you had a robot that could be present for you?
Seven-year-old Joshua Martinangeli of Berlin has a severe lung disease and needs to wear a tube in his neck, so he cannot attend school. A robot avatar, donated to Joshua through a private initiative, sits in his seat in the classroom and is able to interact with the students and teacher, according to Reuters. A light on the avatar blinks when Joshua wants to speak and the children can talk with him too. Joshua and his classmates agree that it’s not the same as him really being there to talk and learn, but it’s a great way to keep him included.
“We are the only district in Berlin that has bought four avatars for its schools. The impetus was COVID-19, but I think this will be the future well beyond the pandemic,” Torsten Kuehne, district education councilor, told Reuters.
So where do we get an avatar to go out and run errands? Can we send it to the office instead of Zooming the next meeting? Or maybe our avatar could go to the gym for us. But how do we get the results to show up on our bodies? C’mon science, figure this out.
Futility, thy name is Kiribati
Before we get to the rest of our regularly scheduled hilarity, a brief geography lesson is in order: Kiribati is an island nation – actually 32 atolls and one coral island – in the central Pacific Ocean. Those atolls are spread out across 1.4 million square miles around the intersection of the equator and the International Date Line, so Kiribati is the only country in the world located in all four hemispheres.
Now, back to the news.
Kiribati closed its borders early in the COVID-19 pandemic and recorded only two cases in almost 2 years. Things were going so well that the authorities recently decided to reopen the country to international travelers. Silly authorities.
The first plane was set to arrive on Jan. 14 from Fiji. This being the age of COVID, plans were made and precautions were taken. All 54 passengers quarantined for 2 weeks before the flight and underwent regular testing, the Guardian noted, and “they were only allowed on the flight after returning negative tests.”
You guessed it. Two-thirds of those 54 people tested positive for COVID-19 after landing in Kiribati.
All of the passengers were quarantined, but since then a security guard at the quarantine center has tested positive, as has someone who was not involved in the quarantine. According to NPR, the government said that “there is now an assumption that COVID-19 is now spreading in the community on South Tarawa and Betio.”
Moral of the story? You can’t beat COVID, so never try.
[EDITOR: Is that really the message we want to send to our readers?]
If you can’t beat them, join them.
[EDITOR: Nope. Try again.]
Resistance is futile?
[EDITOR: Sigh. Close enough.]