User login
Using Telehealth Rehabilitation Therapy to Treat a Finger Flexor Tendon Repair During COVID-19
Telehealth-assisted finger rehabilitat ion therapy demonstrated good functional results following repair of a zone 2 flexor tendon laceration.
In 1948, Sterling Bunnell, MD, used the term no man’s land to describe the area between the A1 pulley at the volar aspect of the metacarpophalangeal joint and the insertion of the flexor digitorum superficialis tendons on the middle phalanx (zone 2).1 Bunnell’s description referenced the area of land in World War I between the trenches of opposing armies, and his goal was to emphasize the heightened risks of performing tendon repair in this area, as these repairs were notorious for poor outcomes. In lieu of tendon repair, Bunnell advocated treatment of tendon lacerations in this area with tendon excision and grafting.
It was not until the 1960s that researchers began to advocate for acute repair of tendons in this area.2,3 Since Verdan’s and Kleinart’s work, fastidious adherence to atraumatic technique and improvements in suture technique and rehabilitation protocols have allowed hand surgeons to repair tendons in this area with some level of success. Over the ensuing decades, acute repair of flexor tendon injuries within zone 2 has become the standard of care. The importance of meticulous technique during flexor tendon repair cannot be overemphasized; however, without appropriate hand therapy, even the most meticulous repair may fail.
COVID-19 has created significant barriers to patient care. Reducing travel and limiting face-to-face patient visits have been emphasized as methods that reduce spread of the virus, but these restrictions also prevent patients from easily accessing hand therapy. Recent adoption of telemedicine and videoconferencing technologies may help to reduce some of these barriers, but few previous studies have described the use of videoconferencing technology to supplant face-to-face hand therapy visits. This case describes the use of videoconferencing technology to provide hand therapy for a patient following repair of an acute flexor tendon laceration in zone 2.
Case Presentation
A patient aged < 50 years presented to a US Department of Veterans Affairs (VA) hand surgery clinic 2 days after sustaining a laceration to the flexor digitorum profundus (FDP) in zone 2 of the small finger while cleaning a knife. During the discussion of their treatment options and the recommended postoperative hand therapy protocol, the patient noted difficulty attending postoperative appointments due to COVID-19 as well as a lack of resources. Given these limitations and following discussion with our hand therapist, we discussed the potential for telehealth follow-up with videoconferencing. Four days following the injury, the patient underwent repair of the FDP. During surgery, the laceration was present at the level of the A3 pulley. The FDP was repaired using a 6-0 polypropylene synthetic suture for the epitendinous repair and 4-strand core suture repair using 3-0 Fiberwire suture in a modified cruciate fashion. The A2 and A4 pulleys were preserved, and venting of the pulleys was not required. At the time of surgery, the flexor digitorum superficialis and radial and ulnar digital neurovascular bundles were intact. Following surgical repair of the tendon, the patient was placed into a dorsal blocking splint with a plan for follow-up within 2 to 3 days.
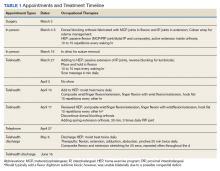
The patient attended the first postoperative visit in person on postoperative day 2. During this visit, the postoperative splint and dressings were removed, and a forearm-based dorsal blocking orthosis was fabricated using thermoplastic. At this visit, the veteran relayed concerns regarding psychosocial and resource barriers in addition to concerns surrounding COVID-19 that would prevent travel to and from hand therapy appointments. Due to these concerns, a passive-motion protocol was initiated using the Indiana manual as a guide.4 The patient returned to the hand clinic at 2 weeks after surgery for evaluation by the operating surgeon and suture removal. All visits after the suture removal were conducted via either telehealth with videoconferencing or by telephone (Table 1).
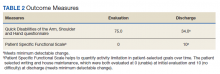
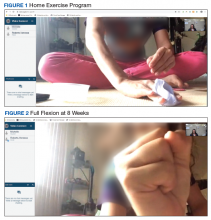
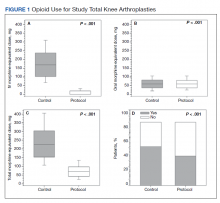
The operative team evaluated the patient 5 times following surgery. Only 2 of these visits were in-person. The patient attended 6 hand therapy sessions with 2 in-person visits to occupational therapy (Figure 1). The remaining 4 visits were conducted using videoconferencing. The patient received therapy supplies by mail as needed, and their use was reviewed in telerehabilitation sessions with videoconferencing as needed. During their postoperative course, the patient experienced little edema or scar tissue formation, and recovery was uncomplicated. The patient developed a mild extensor lag for which a proximal interphalangeal joint spring extension orthosis was provided via mail (Figure 2). The patient admitted only partial adherence with this orthosis, and at discharge, a 10-degree extensor lag remained. The patient was not concerned by this extension deficit and did not experience any associated functional deficits, demonstrated by scores on the Quick Disabilities of the Arm, Shoulder and Hand questionnaire and Patient Specific Functional Scale (Table 2).
Discussion
Few studies have been published that address the efficacy of telerehabilitation after surgical management of traumatic injuries involving the upper extremity. One Australian study performed by Worboys and colleagues concluded that utilization of telehealth services for hand therapy visits may provide accurate patient assessment with favorable patient satisfaction.5 Another study performed in the UK by Gilbert and colleagues demonstrated that videoconferencing is well received by patients, as it may offer shorter wait times, improved convenience, and reduced travel cost.
The authors noted that although videoconferencing may not completely replace in-person therapy, it could act as an adjunct.6 While these in-person visits may be necessary, particularly to establish care, at least one study has demonstrated that patients may prefer follow-up via telehealth if provided the option.7 In a randomized, controlled study performed in Norway, patients were randomized to either an in-person or video consultation with an orthopedic outpatient clinic. Of patients randomized to the in-person clinic visit, 86% preferred to have follow-up via videoconferencing.7
Previous studies have demonstrated that telehealth may produce accurate patient assessment, with relatively high patient satisfaction. Given the COVID-19 pandemic and the limitations that this crisis has placed on in-person outpatient visits, clinics that previously may have been resistant to telehealth are adapting and using the technology to meet the needs of their population.8 The present case demonstrates that videoconferencing is feasible and may lead to successful results, even for cases requiring significant hand therapy follow-up, such as flexor tendon repairs.
Conclusions
Although in-person hand therapy remains the standard of care following flexor tendon repair of the hand, situations may exist in which hand therapy conducted via telehealth is better than no hand therapy at all. The present case study highlights the use of telehealth as an acceptable supplement to in-person postoperative visits.
In our case, use of a standardized protocol with an emphasis on hand function and patient satisfaction as opposed to strict range of motion measurements produced good results. Although a specific telehealth satisfaction measure was not used in this case, commonly used questionnaires may be integrated into future visits to improve telehealth implementation and patient experience. In this specific case, the veteran felt that hand function was regained and expressed general satisfaction with the telemedicine process at the conclusion of care. While telehealth was a useful adjunct in the treatment of the present patient, further study of videoconferencing should be conducted to determine whether hand therapy conducted via telehealth could be implemented more broadly following upper extremity surgery.
1. Hege JJ. History off-hand: Bunnell’s no-man’s land. Hand (NY). 2019;14(4):570-574. doi:10.1177/1558944717744337
2. Verdan C. Primary repair of flexor tendons. J Bone Joint Surg Am. 1960;42-A:647-657.
3. Kleinert HE, Kutz JE, Ashbell TS, et al. Primary repair of lacerated flexor tendon in no man’s land (abstract). J Bone Joint Surg. 1967;49A:577.
4. Cannon NM. Diagnosis and Treatment Manual for Physicians and Therapists: Upper Extremity Rehabilitation. 4th ed. Hand Rehabilitation Center of Indiana; 2001.
5. Worboys T, Brassington M, Ward EC, Cornwell PL. Delivering occupational therapy hand assessment and treatment sessions via telehealth. J Telemed Telecare. 2018;24(3):185-192. doi:10.1177/1357633X17691861
6. Gilbert AW, Jaggi A, May CR. What is the patient acceptability of real time 1:1 videoconferencing in an orthopaedics setting? A systematic review. Physiotherapy. 2018;104(2):178-186. doi:10.1016/j.physio.2017.11.217
7. Buvik A, Bugge E, Knutsen G, Smatresk A, Wilsgaard T. Patient reported outcomes with remote orthopaedic consultations by telemedicine: A randomised controlled trial. J Telemed Telecare. 2019;25(8):451-459. doi:10.1177/1357633X18783921
8. Loeb AE, Rao SS, Ficke JR, Morris CD, Riley LH 3rd, Levin AS. Departmental experience and lessons learned with accelerated introduction of telemedicine during the COVID-19 crisis. J Am Acad Orthop Surg. 2020;28(11):e469-e476. doi:10.5435/JAAOS-D-20-00380
Telehealth-assisted finger rehabilitat ion therapy demonstrated good functional results following repair of a zone 2 flexor tendon laceration.
Telehealth-assisted finger rehabilitat ion therapy demonstrated good functional results following repair of a zone 2 flexor tendon laceration.
In 1948, Sterling Bunnell, MD, used the term no man’s land to describe the area between the A1 pulley at the volar aspect of the metacarpophalangeal joint and the insertion of the flexor digitorum superficialis tendons on the middle phalanx (zone 2).1 Bunnell’s description referenced the area of land in World War I between the trenches of opposing armies, and his goal was to emphasize the heightened risks of performing tendon repair in this area, as these repairs were notorious for poor outcomes. In lieu of tendon repair, Bunnell advocated treatment of tendon lacerations in this area with tendon excision and grafting.
It was not until the 1960s that researchers began to advocate for acute repair of tendons in this area.2,3 Since Verdan’s and Kleinart’s work, fastidious adherence to atraumatic technique and improvements in suture technique and rehabilitation protocols have allowed hand surgeons to repair tendons in this area with some level of success. Over the ensuing decades, acute repair of flexor tendon injuries within zone 2 has become the standard of care. The importance of meticulous technique during flexor tendon repair cannot be overemphasized; however, without appropriate hand therapy, even the most meticulous repair may fail.
COVID-19 has created significant barriers to patient care. Reducing travel and limiting face-to-face patient visits have been emphasized as methods that reduce spread of the virus, but these restrictions also prevent patients from easily accessing hand therapy. Recent adoption of telemedicine and videoconferencing technologies may help to reduce some of these barriers, but few previous studies have described the use of videoconferencing technology to supplant face-to-face hand therapy visits. This case describes the use of videoconferencing technology to provide hand therapy for a patient following repair of an acute flexor tendon laceration in zone 2.
Case Presentation
A patient aged < 50 years presented to a US Department of Veterans Affairs (VA) hand surgery clinic 2 days after sustaining a laceration to the flexor digitorum profundus (FDP) in zone 2 of the small finger while cleaning a knife. During the discussion of their treatment options and the recommended postoperative hand therapy protocol, the patient noted difficulty attending postoperative appointments due to COVID-19 as well as a lack of resources. Given these limitations and following discussion with our hand therapist, we discussed the potential for telehealth follow-up with videoconferencing. Four days following the injury, the patient underwent repair of the FDP. During surgery, the laceration was present at the level of the A3 pulley. The FDP was repaired using a 6-0 polypropylene synthetic suture for the epitendinous repair and 4-strand core suture repair using 3-0 Fiberwire suture in a modified cruciate fashion. The A2 and A4 pulleys were preserved, and venting of the pulleys was not required. At the time of surgery, the flexor digitorum superficialis and radial and ulnar digital neurovascular bundles were intact. Following surgical repair of the tendon, the patient was placed into a dorsal blocking splint with a plan for follow-up within 2 to 3 days.
The patient attended the first postoperative visit in person on postoperative day 2. During this visit, the postoperative splint and dressings were removed, and a forearm-based dorsal blocking orthosis was fabricated using thermoplastic. At this visit, the veteran relayed concerns regarding psychosocial and resource barriers in addition to concerns surrounding COVID-19 that would prevent travel to and from hand therapy appointments. Due to these concerns, a passive-motion protocol was initiated using the Indiana manual as a guide.4 The patient returned to the hand clinic at 2 weeks after surgery for evaluation by the operating surgeon and suture removal. All visits after the suture removal were conducted via either telehealth with videoconferencing or by telephone (Table 1).
The operative team evaluated the patient 5 times following surgery. Only 2 of these visits were in-person. The patient attended 6 hand therapy sessions with 2 in-person visits to occupational therapy (Figure 1). The remaining 4 visits were conducted using videoconferencing. The patient received therapy supplies by mail as needed, and their use was reviewed in telerehabilitation sessions with videoconferencing as needed. During their postoperative course, the patient experienced little edema or scar tissue formation, and recovery was uncomplicated. The patient developed a mild extensor lag for which a proximal interphalangeal joint spring extension orthosis was provided via mail (Figure 2). The patient admitted only partial adherence with this orthosis, and at discharge, a 10-degree extensor lag remained. The patient was not concerned by this extension deficit and did not experience any associated functional deficits, demonstrated by scores on the Quick Disabilities of the Arm, Shoulder and Hand questionnaire and Patient Specific Functional Scale (Table 2).
Discussion
Few studies have been published that address the efficacy of telerehabilitation after surgical management of traumatic injuries involving the upper extremity. One Australian study performed by Worboys and colleagues concluded that utilization of telehealth services for hand therapy visits may provide accurate patient assessment with favorable patient satisfaction.5 Another study performed in the UK by Gilbert and colleagues demonstrated that videoconferencing is well received by patients, as it may offer shorter wait times, improved convenience, and reduced travel cost.
The authors noted that although videoconferencing may not completely replace in-person therapy, it could act as an adjunct.6 While these in-person visits may be necessary, particularly to establish care, at least one study has demonstrated that patients may prefer follow-up via telehealth if provided the option.7 In a randomized, controlled study performed in Norway, patients were randomized to either an in-person or video consultation with an orthopedic outpatient clinic. Of patients randomized to the in-person clinic visit, 86% preferred to have follow-up via videoconferencing.7
Previous studies have demonstrated that telehealth may produce accurate patient assessment, with relatively high patient satisfaction. Given the COVID-19 pandemic and the limitations that this crisis has placed on in-person outpatient visits, clinics that previously may have been resistant to telehealth are adapting and using the technology to meet the needs of their population.8 The present case demonstrates that videoconferencing is feasible and may lead to successful results, even for cases requiring significant hand therapy follow-up, such as flexor tendon repairs.
Conclusions
Although in-person hand therapy remains the standard of care following flexor tendon repair of the hand, situations may exist in which hand therapy conducted via telehealth is better than no hand therapy at all. The present case study highlights the use of telehealth as an acceptable supplement to in-person postoperative visits.
In our case, use of a standardized protocol with an emphasis on hand function and patient satisfaction as opposed to strict range of motion measurements produced good results. Although a specific telehealth satisfaction measure was not used in this case, commonly used questionnaires may be integrated into future visits to improve telehealth implementation and patient experience. In this specific case, the veteran felt that hand function was regained and expressed general satisfaction with the telemedicine process at the conclusion of care. While telehealth was a useful adjunct in the treatment of the present patient, further study of videoconferencing should be conducted to determine whether hand therapy conducted via telehealth could be implemented more broadly following upper extremity surgery.
In 1948, Sterling Bunnell, MD, used the term no man’s land to describe the area between the A1 pulley at the volar aspect of the metacarpophalangeal joint and the insertion of the flexor digitorum superficialis tendons on the middle phalanx (zone 2).1 Bunnell’s description referenced the area of land in World War I between the trenches of opposing armies, and his goal was to emphasize the heightened risks of performing tendon repair in this area, as these repairs were notorious for poor outcomes. In lieu of tendon repair, Bunnell advocated treatment of tendon lacerations in this area with tendon excision and grafting.
It was not until the 1960s that researchers began to advocate for acute repair of tendons in this area.2,3 Since Verdan’s and Kleinart’s work, fastidious adherence to atraumatic technique and improvements in suture technique and rehabilitation protocols have allowed hand surgeons to repair tendons in this area with some level of success. Over the ensuing decades, acute repair of flexor tendon injuries within zone 2 has become the standard of care. The importance of meticulous technique during flexor tendon repair cannot be overemphasized; however, without appropriate hand therapy, even the most meticulous repair may fail.
COVID-19 has created significant barriers to patient care. Reducing travel and limiting face-to-face patient visits have been emphasized as methods that reduce spread of the virus, but these restrictions also prevent patients from easily accessing hand therapy. Recent adoption of telemedicine and videoconferencing technologies may help to reduce some of these barriers, but few previous studies have described the use of videoconferencing technology to supplant face-to-face hand therapy visits. This case describes the use of videoconferencing technology to provide hand therapy for a patient following repair of an acute flexor tendon laceration in zone 2.
Case Presentation
A patient aged < 50 years presented to a US Department of Veterans Affairs (VA) hand surgery clinic 2 days after sustaining a laceration to the flexor digitorum profundus (FDP) in zone 2 of the small finger while cleaning a knife. During the discussion of their treatment options and the recommended postoperative hand therapy protocol, the patient noted difficulty attending postoperative appointments due to COVID-19 as well as a lack of resources. Given these limitations and following discussion with our hand therapist, we discussed the potential for telehealth follow-up with videoconferencing. Four days following the injury, the patient underwent repair of the FDP. During surgery, the laceration was present at the level of the A3 pulley. The FDP was repaired using a 6-0 polypropylene synthetic suture for the epitendinous repair and 4-strand core suture repair using 3-0 Fiberwire suture in a modified cruciate fashion. The A2 and A4 pulleys were preserved, and venting of the pulleys was not required. At the time of surgery, the flexor digitorum superficialis and radial and ulnar digital neurovascular bundles were intact. Following surgical repair of the tendon, the patient was placed into a dorsal blocking splint with a plan for follow-up within 2 to 3 days.
The patient attended the first postoperative visit in person on postoperative day 2. During this visit, the postoperative splint and dressings were removed, and a forearm-based dorsal blocking orthosis was fabricated using thermoplastic. At this visit, the veteran relayed concerns regarding psychosocial and resource barriers in addition to concerns surrounding COVID-19 that would prevent travel to and from hand therapy appointments. Due to these concerns, a passive-motion protocol was initiated using the Indiana manual as a guide.4 The patient returned to the hand clinic at 2 weeks after surgery for evaluation by the operating surgeon and suture removal. All visits after the suture removal were conducted via either telehealth with videoconferencing or by telephone (Table 1).
The operative team evaluated the patient 5 times following surgery. Only 2 of these visits were in-person. The patient attended 6 hand therapy sessions with 2 in-person visits to occupational therapy (Figure 1). The remaining 4 visits were conducted using videoconferencing. The patient received therapy supplies by mail as needed, and their use was reviewed in telerehabilitation sessions with videoconferencing as needed. During their postoperative course, the patient experienced little edema or scar tissue formation, and recovery was uncomplicated. The patient developed a mild extensor lag for which a proximal interphalangeal joint spring extension orthosis was provided via mail (Figure 2). The patient admitted only partial adherence with this orthosis, and at discharge, a 10-degree extensor lag remained. The patient was not concerned by this extension deficit and did not experience any associated functional deficits, demonstrated by scores on the Quick Disabilities of the Arm, Shoulder and Hand questionnaire and Patient Specific Functional Scale (Table 2).
Discussion
Few studies have been published that address the efficacy of telerehabilitation after surgical management of traumatic injuries involving the upper extremity. One Australian study performed by Worboys and colleagues concluded that utilization of telehealth services for hand therapy visits may provide accurate patient assessment with favorable patient satisfaction.5 Another study performed in the UK by Gilbert and colleagues demonstrated that videoconferencing is well received by patients, as it may offer shorter wait times, improved convenience, and reduced travel cost.
The authors noted that although videoconferencing may not completely replace in-person therapy, it could act as an adjunct.6 While these in-person visits may be necessary, particularly to establish care, at least one study has demonstrated that patients may prefer follow-up via telehealth if provided the option.7 In a randomized, controlled study performed in Norway, patients were randomized to either an in-person or video consultation with an orthopedic outpatient clinic. Of patients randomized to the in-person clinic visit, 86% preferred to have follow-up via videoconferencing.7
Previous studies have demonstrated that telehealth may produce accurate patient assessment, with relatively high patient satisfaction. Given the COVID-19 pandemic and the limitations that this crisis has placed on in-person outpatient visits, clinics that previously may have been resistant to telehealth are adapting and using the technology to meet the needs of their population.8 The present case demonstrates that videoconferencing is feasible and may lead to successful results, even for cases requiring significant hand therapy follow-up, such as flexor tendon repairs.
Conclusions
Although in-person hand therapy remains the standard of care following flexor tendon repair of the hand, situations may exist in which hand therapy conducted via telehealth is better than no hand therapy at all. The present case study highlights the use of telehealth as an acceptable supplement to in-person postoperative visits.
In our case, use of a standardized protocol with an emphasis on hand function and patient satisfaction as opposed to strict range of motion measurements produced good results. Although a specific telehealth satisfaction measure was not used in this case, commonly used questionnaires may be integrated into future visits to improve telehealth implementation and patient experience. In this specific case, the veteran felt that hand function was regained and expressed general satisfaction with the telemedicine process at the conclusion of care. While telehealth was a useful adjunct in the treatment of the present patient, further study of videoconferencing should be conducted to determine whether hand therapy conducted via telehealth could be implemented more broadly following upper extremity surgery.
1. Hege JJ. History off-hand: Bunnell’s no-man’s land. Hand (NY). 2019;14(4):570-574. doi:10.1177/1558944717744337
2. Verdan C. Primary repair of flexor tendons. J Bone Joint Surg Am. 1960;42-A:647-657.
3. Kleinert HE, Kutz JE, Ashbell TS, et al. Primary repair of lacerated flexor tendon in no man’s land (abstract). J Bone Joint Surg. 1967;49A:577.
4. Cannon NM. Diagnosis and Treatment Manual for Physicians and Therapists: Upper Extremity Rehabilitation. 4th ed. Hand Rehabilitation Center of Indiana; 2001.
5. Worboys T, Brassington M, Ward EC, Cornwell PL. Delivering occupational therapy hand assessment and treatment sessions via telehealth. J Telemed Telecare. 2018;24(3):185-192. doi:10.1177/1357633X17691861
6. Gilbert AW, Jaggi A, May CR. What is the patient acceptability of real time 1:1 videoconferencing in an orthopaedics setting? A systematic review. Physiotherapy. 2018;104(2):178-186. doi:10.1016/j.physio.2017.11.217
7. Buvik A, Bugge E, Knutsen G, Smatresk A, Wilsgaard T. Patient reported outcomes with remote orthopaedic consultations by telemedicine: A randomised controlled trial. J Telemed Telecare. 2019;25(8):451-459. doi:10.1177/1357633X18783921
8. Loeb AE, Rao SS, Ficke JR, Morris CD, Riley LH 3rd, Levin AS. Departmental experience and lessons learned with accelerated introduction of telemedicine during the COVID-19 crisis. J Am Acad Orthop Surg. 2020;28(11):e469-e476. doi:10.5435/JAAOS-D-20-00380
1. Hege JJ. History off-hand: Bunnell’s no-man’s land. Hand (NY). 2019;14(4):570-574. doi:10.1177/1558944717744337
2. Verdan C. Primary repair of flexor tendons. J Bone Joint Surg Am. 1960;42-A:647-657.
3. Kleinert HE, Kutz JE, Ashbell TS, et al. Primary repair of lacerated flexor tendon in no man’s land (abstract). J Bone Joint Surg. 1967;49A:577.
4. Cannon NM. Diagnosis and Treatment Manual for Physicians and Therapists: Upper Extremity Rehabilitation. 4th ed. Hand Rehabilitation Center of Indiana; 2001.
5. Worboys T, Brassington M, Ward EC, Cornwell PL. Delivering occupational therapy hand assessment and treatment sessions via telehealth. J Telemed Telecare. 2018;24(3):185-192. doi:10.1177/1357633X17691861
6. Gilbert AW, Jaggi A, May CR. What is the patient acceptability of real time 1:1 videoconferencing in an orthopaedics setting? A systematic review. Physiotherapy. 2018;104(2):178-186. doi:10.1016/j.physio.2017.11.217
7. Buvik A, Bugge E, Knutsen G, Smatresk A, Wilsgaard T. Patient reported outcomes with remote orthopaedic consultations by telemedicine: A randomised controlled trial. J Telemed Telecare. 2019;25(8):451-459. doi:10.1177/1357633X18783921
8. Loeb AE, Rao SS, Ficke JR, Morris CD, Riley LH 3rd, Levin AS. Departmental experience and lessons learned with accelerated introduction of telemedicine during the COVID-19 crisis. J Am Acad Orthop Surg. 2020;28(11):e469-e476. doi:10.5435/JAAOS-D-20-00380
Review of Efficacy and Safety of Spinal Cord Stimulation in Veterans
Lower back pain (LBP) affects an estimated 9.4% of the global population and has resulted in more years lived with disability than any other health condition.1 LBP affects a wide range of populations, but US veterans have been shown to have significantly higher rates of back pain than nonveterans. The National Institutes of Health reports that 65.6% of veterans experience chronic pain; 9.1% of veterans experience severe, chronic pain.2 Chronic back pain is treated by a range of methods, including medications, surgery, physical therapy (PT), patient education, and behavioral therapy.3 However, chronic neuropathic back pain has been shown to have limited responsiveness to medication.4
Neuropathic pain is caused by lesions in the somatosensory nervous system, resulting in spontaneous pain and amplified pain responses to both painful and nonpainful stimuli.5 The most common location for neuropathic pain is the back and legs. Between 10% and 40% of people who undergo lumbosacral spine surgery to treat neuropathic radicular pain will experience further neuropathic pain.6 This condition is referred to as failed back surgery syndrome or postlaminectomy syndrome (PLS). While neuropathic back pain has had limited responsiveness to medication and repeated lumbosacral spine surgery, spinal cord stimulation (SCS) has shown promise as an effective form of pain treatment for those experiencing PLS and other spine disorders.7-10 In addition, SCS therapy has had a very low incidence of complications, which may be on the decline with recent technological advancements.11 Patients with a diagnosis of PLS, LBP, or complex regional pain syndrome (CRPS) who have not responded to medications, therapy, and/or injections for ≥ 6 months were eligible for a trial of SCS therapy. Trial leads were placed via the percutaneous route with the battery strapped to the waistline for 3 to 5 days and were removed in clinic. Patients who experienced > 60% pain relief and functional improvement received a SCS implant.
The effectiveness of SCS has been demonstrated in a nonveteran population, but it has not been studied in a veteran population.12 US Department of Veterans Affairs (VA) health care coverage is different from Medicare and private insurance in that it is classified as a benefit and not insurance. The goals of treatment at the VA may include considerations in addition to feeling better, and patient presentations may not align with those in the private sector.
We hypothesize that SCS is both a safe and beneficial treatment option for veterans with chronic intractable spine and/or extremity pain. The purpose of this study was to determine the efficacy and safety of SCS in a veteran population.
Methods
The efficacy and safety of SCS was determined via a retrospective study. Inclusion criteria for the study consisted of any Southeastern Louisiana Veterans Health Care System (SLVHCS) patient who had an SCS trial and/or implant from 2008 to 2020. Eligible veterans must have had chronic pain for at least 6 months and had previously tried multiple medications, PT, transcutaneous nerve stimulation, facet injections, epidural steroid injections, or surgery without success. For medication therapy to be considered unsuccessful, it must have included acetaminophen, nonsteroidal anti-inflammatory drugs, and ≥ 1 adjuvant medication (gabapentin, duloxetine, amitriptyline, lidocaine, and menthol). A diagnosis of chronic LBP, PLS, cervical or lumbar spondylosis with radiculopathy, complex regional pain syndrome, or chronic pain syndrome was required for eligibility. Patients whose pain decreased by > 60% and had functional improvement in a 3- to 5-day trial received SCS implantation with percutaneous leads by a pain physician or paddle lead by a neurosurgeon.
The SLVHCS Institutional Review Board approved this study. Electronic health records were reviewed to determine patient age, anthropometric data, and date of SCS implantation. Patients were then called and interviewed to complete a survey. After obtaining verbal consent to the study, subjects were surveyed regarding whether the patient would recommend the procedure to peers, adverse effects (AEs) or complications, and the ability to decrease opiates if applicable. A verbal Pain Outcome Questionnaire (POQ) assessment of activities of daily living also was given during the phone interview regarding pain levels before SCS and at the time of the phone interview.13 (eAppendix available at doi:10.12788/fp.0204) Following the survey, a chart review was performed to corroborate the given AEs or complications and opiate use information. Before and after results of the POQ were compared via a paired sample t test, and P values < .05 were considered significant. Analyses were performed by IBM SPSS, version 26.
The primary outcome measure for this study was whether veterans would recommend SCS to their peers; in our view, this categorical outcome measure seemed to be more valuable to share with future patients who might be candidates for SCS. Since VA health care coverage and goals of treatment may be different from a nonveteran population, we opted to use this primary measure to decrease the possibility of confounding variables.
Secondary outcome measures included changes in POC scores, improvements in activities of daily living, and decreases in use of opioid pain medications.
POQ responses were recorded during the telephone interviews (0 to 10 scale). A paired sample t test was conducted to compare pain levels before and after SCS implant. Pain levels were gathered in the single phone call. Patient opioid usage, if applicable, was assessed by converting medications to morphine milligram equivalent dosing (MMED). Since patients who were on chronic opioids took multiple formulations, we changed the total daily dose to all morphine; for this study, morphine was considered equivalent to hydrocodone, and oxycodone was 1.5x morphine.
Results
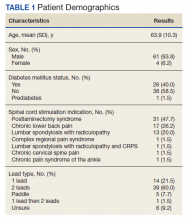
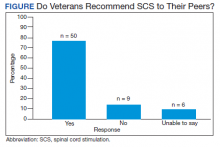
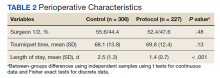
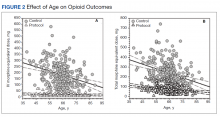
Of the 90 SLVHCS patients who received an SCS implant between 2008 and 2020, 76 were reached by telephone and 65 had their responses recorded in the study. Of the 11 patients who were not included, 5 had the SCS removed; it is unclear whether these veterans would have recommended the treatment. Four were unable to quantify pain and/or SCS effects, and 2 were excluded due to a dementia diagnosis years after the implant. The mean (SD) age of participants was 63.9 (10.3) years. Forty percent of patients had a diabetes mellitus diagnosis and 1 had prediabetes. Patients’ most common qualifying diagnosis for SCS was PLS (47.7%) followed by chronic LBP (26.2%). A percutaneous 2-lead technique was the most common type of SCS type used (60.0%) followed by 1-lead (21.5%). The most common SCS manufacturer was Boston Scientific (87.7%)(Table 1). Most veterans (76.9%) recommended SCS to their peers; 13.8% did not recommend SCS; 9.2% were undecided and stated that they were unable to recommend because they did not want to persuade a peer to get SCS (Figure).
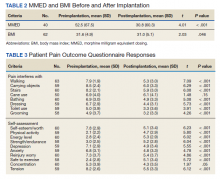
There was a statistically significant decrease in opioid use for the 40 veterans for whom pain medication was converted (P < .001)(Table 2). Six patients reported using opioids at some point but could not remember their dose, and no records were found in their chart review, so they were not included in the MMED analysis. In that group, 4 patients reported using opioids before SCS but discontinued the opioid use after SCS implantation, and 2 patients noted using opioids before SCS and concomitantly. Eighteen subjects reported no opioid use at any point before or after SCS (Table 3).
There were few life-threatening complications of SCS. Three veterans developed skin dehiscence; 2 had dehiscence at the battery/generator site, and 1 had dehiscence at the lead anchor site. Two patients with dehiscence also had morbid obesity, and the third had postoperative malnourishment. The dehiscence occurred 3 and 8 months postoperation. All 3 patients with dehiscence had the SCS explanted, though they were eager to get a new SCS implanted as soon as possible because SCS was their most successful treatment to date.
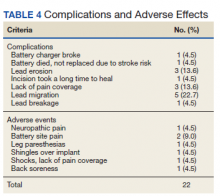
Twenty of the 64 veterans surveyed reported other complications of SCS, including lead migration, lack of pain coverage, paresthesia and numbness, soreness around generator site, SCS shocking patient when performing full thoracic spine flexion, and shingles at the battery site (Table 4). There were 11 explants among the 76 veterans contacted. The primary reason for explant was lack of pain coverage.
Patient concerns included pain with sitting in chairs due to tenderness around the implant, SCS helping with physical pain but not mental pain, SCS only working during the day and not helping with sleep, and patients lacking education regarding possible complications of SCS.
Discussion
In this nonrandomized retrospective review, SCS was shown to be an effective treatment for intractable spine and/or extremity pain. Veterans’ pain levels were significantly reduced following SCS implantation, and more than three-fourths of veterans recommended SCS to their peers. We used the recommendation of SCS to peers as the most important metric regarding the effectiveness of SCS, as this measure was felt to be more valuable to share with future patients; furthermore, categorical analysis has been shown to be more valuable than ordinal pain scales to measure pain.14 In addition to wanting to expand the available research to the general public, we wanted a measure that we could easily relay to our patient population regarding SCS.
The explant rate of 14.5% among surveyed veterans falls at the higher end of the normal ranges found in previous studies of long-term SCS outcomes.15-17 One possible reason for the higher rate is that we did not differentiate based on the reason for the explant (ie, no benefit, further surgery needed for underlying medical condition, or SCS-specific complications). Another possible contributing factor to the higher than expected explant rate is the geographic location in the New Orleans metro area; New Orleans is considered to have one of the highest rates of obesity in the United States and obesity typically has other diseases associated with it such as hypertension and diabetes mellitus.
Limitations
Limitations of the study include the relatively low number of subjects, subjective nature of the interview questions, and the patients’ answers. Typically the POQ has been used as a prospective assessment of pain; whether it is valid in a retrospective analysis is not clear. While there was a statistically significant decrease of opioid use after getting SCS, this study can only show correlation, not causation. During the study period, there has been a drastic change in opioid prescribing patterns and efforts to decrease the amount of opioids prescribed.
Subjects also were asked to rate their pain and quality of life before SCS. Some subjects had SCS implantation up to 10 years prior to the phone interview. The variable amount of time between SCS implantation and interview likely affected subjects’ responses. Chronic pain is a moving target. Patients have good days and bad days that would likely change opinions on SCS benefits on a single phone interview. Some patients needed battery replacements at the time of the interview (battery life averaged about 3 to 5 years in our study population) and were asked to report current levels of pain from the perspective of when their batteries were still functional, further affecting results.
Conclusions
SCS was shown to improve the quality of life of US veterans at SLVHCS across a wide variety of metrics, including activities of daily living, as well as mental and physical health. For veterans with chronic intractable pain who have tried and failed more conservative treatments, SCS is a great treatment.
1. Hoy DG, Smith E, Cross M, et al. The global burden of musculoskeletal conditions for 2010: an overview of methods. Ann Rheum Dis. 2014;73(6):982-989 doi:10.1136/annrheumdis-2013-204344
2. Nahin RL. Severe pain in veterans: the effect of age and sex, and comparisons with the general population. J Pain. 2017;18(3):247-254. doi:10.1016/j.jpain.2016.10.021
3. Institute of Medicine (US) Committee on Advancing Pain Research, Care, and Education. Relieving Pain in America: A Blueprint for Transforming Prevention, Care, Education, and Research. Washington, DC: National Academies Press; 2011.
4. Finnerup NB, Attal N, Haroutounian S, et al. Pharmacotherapy for neuropathic pain in adults: a systematic review and meta-analysis. Lancet Neurol. 2015;14(2):162-173. doi:10.1016/S1474-4422(14)70251-0
5. Costigan M, Scholz J, Woolf CJ. Neuropathic pain: a maladaptive response of the nervous system to damage. Annu Rev Neurosci. 2009;32:1-32. doi:10.1146/annurev.neuro.051508.135531
6. Wilkinson HA. The Failed Back Syndrome: Etiology and Therapy. 2nd ed. Harper & Row; 1991.
7. Kumar K, Taylor RS, Jacques L, et al. Spinal cord stimulation versus conventional medical management for neuropathic pain: a multicentre randomised controlled trial in patients with failed back surgery syndrome. Pain. 2007;132(1-2):179-188. doi:10.1016/j.pain.2007.07.028
8. North RB, Kidd DH, Farrokhi F, Piantadosi SA. Spinal cord stimulation versus repeated lumbosacral spine surgery for chronic pain: a randomized, controlled trial. Neurosurgery. 2005;56(1):98-107. doi:10.1227/01.neu.0000144839.65524.e0
9. Geurts JW, Smits H, Kemler MA, Brunner F, Kessels AG, van Kleef M. Spinal cord stimulation for complex regional pain syndrome type I: a prospective cohort study with long-term follow-up. Neuromodulation. 2013;16(6):523-529. doi:10.1111/ner.12024
10. Kumar K, Rizvi S, Bnurs SB. Spinal cord stimulation is effective in management of complex regional pain syndrome I: fact or fiction. Neurosurgery. 2011;69(3):566-5580. doi:10.1227/NEU.0b013e3182181e60
11. Mekhail NA, Mathews M, Nageeb F, Guirguis M, Mekhail MN, Cheng J. Retrospective review of 707 cases of spinal cord stimulation: indications and complications. Pain Pract. 2011;11(2):148-153. doi:10.1111/j.1533-2500.2010.00407.x
12. Veizi E, Hayek SM, North J, et al. Spinal cord stimulation (SCS) with anatomically guided (3D) neural targeting shows superior chronic axial low back pain relief compared to traditional SCS-LUMINA Study. Pain Med. 2017;18(8):1534-1548. doi:10.1093/pm/pnw286
13. Gordon DB, Polomano RC, Pellino TA, et al. Revised American Pain Society Patient Outcome Questionnaire (APS-POQ-R) for quality improvement of pain management in hospitalized adults: preliminary psychometric evaluation. J Pain. 2010;11(11):1172-1186. doi:10.1016/j.jpain.2010.02.012
14. Kennedy DJ, Schneider B. Lies, damn lies, and statistic: a commentary. Pain Med. 2020;21(10):2052-2054. doi:10.1093/pm/pnaa287
15. Van Buyten JP, Wille F, Smet I, et al. Therapy-related explants after spinal cord stimulation: results of an international retrospective chart review study. Neuromodulation. 2017;20(7):642-649. doi:10.1111/ner.12642
16. Hayek SM, Veizi E, Hanes M. Treatment-limiting complications of percutaneous spinal cord stimulator implants: a review of eight years of experience from an academic center database. Neuromodulation. 2015;18(7):603-609. doi:10.1111/ner.12312
17. Pope JE, Deer TR, Falowski S, et al. Multicenter retrospective study of neurostimulation with exit of therapy by explant. Neuromodulation. 2017;20(6):543-552. doi:10.1111/ner.12634
Lower back pain (LBP) affects an estimated 9.4% of the global population and has resulted in more years lived with disability than any other health condition.1 LBP affects a wide range of populations, but US veterans have been shown to have significantly higher rates of back pain than nonveterans. The National Institutes of Health reports that 65.6% of veterans experience chronic pain; 9.1% of veterans experience severe, chronic pain.2 Chronic back pain is treated by a range of methods, including medications, surgery, physical therapy (PT), patient education, and behavioral therapy.3 However, chronic neuropathic back pain has been shown to have limited responsiveness to medication.4
Neuropathic pain is caused by lesions in the somatosensory nervous system, resulting in spontaneous pain and amplified pain responses to both painful and nonpainful stimuli.5 The most common location for neuropathic pain is the back and legs. Between 10% and 40% of people who undergo lumbosacral spine surgery to treat neuropathic radicular pain will experience further neuropathic pain.6 This condition is referred to as failed back surgery syndrome or postlaminectomy syndrome (PLS). While neuropathic back pain has had limited responsiveness to medication and repeated lumbosacral spine surgery, spinal cord stimulation (SCS) has shown promise as an effective form of pain treatment for those experiencing PLS and other spine disorders.7-10 In addition, SCS therapy has had a very low incidence of complications, which may be on the decline with recent technological advancements.11 Patients with a diagnosis of PLS, LBP, or complex regional pain syndrome (CRPS) who have not responded to medications, therapy, and/or injections for ≥ 6 months were eligible for a trial of SCS therapy. Trial leads were placed via the percutaneous route with the battery strapped to the waistline for 3 to 5 days and were removed in clinic. Patients who experienced > 60% pain relief and functional improvement received a SCS implant.
The effectiveness of SCS has been demonstrated in a nonveteran population, but it has not been studied in a veteran population.12 US Department of Veterans Affairs (VA) health care coverage is different from Medicare and private insurance in that it is classified as a benefit and not insurance. The goals of treatment at the VA may include considerations in addition to feeling better, and patient presentations may not align with those in the private sector.
We hypothesize that SCS is both a safe and beneficial treatment option for veterans with chronic intractable spine and/or extremity pain. The purpose of this study was to determine the efficacy and safety of SCS in a veteran population.
Methods
The efficacy and safety of SCS was determined via a retrospective study. Inclusion criteria for the study consisted of any Southeastern Louisiana Veterans Health Care System (SLVHCS) patient who had an SCS trial and/or implant from 2008 to 2020. Eligible veterans must have had chronic pain for at least 6 months and had previously tried multiple medications, PT, transcutaneous nerve stimulation, facet injections, epidural steroid injections, or surgery without success. For medication therapy to be considered unsuccessful, it must have included acetaminophen, nonsteroidal anti-inflammatory drugs, and ≥ 1 adjuvant medication (gabapentin, duloxetine, amitriptyline, lidocaine, and menthol). A diagnosis of chronic LBP, PLS, cervical or lumbar spondylosis with radiculopathy, complex regional pain syndrome, or chronic pain syndrome was required for eligibility. Patients whose pain decreased by > 60% and had functional improvement in a 3- to 5-day trial received SCS implantation with percutaneous leads by a pain physician or paddle lead by a neurosurgeon.
The SLVHCS Institutional Review Board approved this study. Electronic health records were reviewed to determine patient age, anthropometric data, and date of SCS implantation. Patients were then called and interviewed to complete a survey. After obtaining verbal consent to the study, subjects were surveyed regarding whether the patient would recommend the procedure to peers, adverse effects (AEs) or complications, and the ability to decrease opiates if applicable. A verbal Pain Outcome Questionnaire (POQ) assessment of activities of daily living also was given during the phone interview regarding pain levels before SCS and at the time of the phone interview.13 (eAppendix available at doi:10.12788/fp.0204) Following the survey, a chart review was performed to corroborate the given AEs or complications and opiate use information. Before and after results of the POQ were compared via a paired sample t test, and P values < .05 were considered significant. Analyses were performed by IBM SPSS, version 26.
The primary outcome measure for this study was whether veterans would recommend SCS to their peers; in our view, this categorical outcome measure seemed to be more valuable to share with future patients who might be candidates for SCS. Since VA health care coverage and goals of treatment may be different from a nonveteran population, we opted to use this primary measure to decrease the possibility of confounding variables.
Secondary outcome measures included changes in POC scores, improvements in activities of daily living, and decreases in use of opioid pain medications.
POQ responses were recorded during the telephone interviews (0 to 10 scale). A paired sample t test was conducted to compare pain levels before and after SCS implant. Pain levels were gathered in the single phone call. Patient opioid usage, if applicable, was assessed by converting medications to morphine milligram equivalent dosing (MMED). Since patients who were on chronic opioids took multiple formulations, we changed the total daily dose to all morphine; for this study, morphine was considered equivalent to hydrocodone, and oxycodone was 1.5x morphine.
Results
Of the 90 SLVHCS patients who received an SCS implant between 2008 and 2020, 76 were reached by telephone and 65 had their responses recorded in the study. Of the 11 patients who were not included, 5 had the SCS removed; it is unclear whether these veterans would have recommended the treatment. Four were unable to quantify pain and/or SCS effects, and 2 were excluded due to a dementia diagnosis years after the implant. The mean (SD) age of participants was 63.9 (10.3) years. Forty percent of patients had a diabetes mellitus diagnosis and 1 had prediabetes. Patients’ most common qualifying diagnosis for SCS was PLS (47.7%) followed by chronic LBP (26.2%). A percutaneous 2-lead technique was the most common type of SCS type used (60.0%) followed by 1-lead (21.5%). The most common SCS manufacturer was Boston Scientific (87.7%)(Table 1). Most veterans (76.9%) recommended SCS to their peers; 13.8% did not recommend SCS; 9.2% were undecided and stated that they were unable to recommend because they did not want to persuade a peer to get SCS (Figure).
There was a statistically significant decrease in opioid use for the 40 veterans for whom pain medication was converted (P < .001)(Table 2). Six patients reported using opioids at some point but could not remember their dose, and no records were found in their chart review, so they were not included in the MMED analysis. In that group, 4 patients reported using opioids before SCS but discontinued the opioid use after SCS implantation, and 2 patients noted using opioids before SCS and concomitantly. Eighteen subjects reported no opioid use at any point before or after SCS (Table 3).
There were few life-threatening complications of SCS. Three veterans developed skin dehiscence; 2 had dehiscence at the battery/generator site, and 1 had dehiscence at the lead anchor site. Two patients with dehiscence also had morbid obesity, and the third had postoperative malnourishment. The dehiscence occurred 3 and 8 months postoperation. All 3 patients with dehiscence had the SCS explanted, though they were eager to get a new SCS implanted as soon as possible because SCS was their most successful treatment to date.
Twenty of the 64 veterans surveyed reported other complications of SCS, including lead migration, lack of pain coverage, paresthesia and numbness, soreness around generator site, SCS shocking patient when performing full thoracic spine flexion, and shingles at the battery site (Table 4). There were 11 explants among the 76 veterans contacted. The primary reason for explant was lack of pain coverage.
Patient concerns included pain with sitting in chairs due to tenderness around the implant, SCS helping with physical pain but not mental pain, SCS only working during the day and not helping with sleep, and patients lacking education regarding possible complications of SCS.
Discussion
In this nonrandomized retrospective review, SCS was shown to be an effective treatment for intractable spine and/or extremity pain. Veterans’ pain levels were significantly reduced following SCS implantation, and more than three-fourths of veterans recommended SCS to their peers. We used the recommendation of SCS to peers as the most important metric regarding the effectiveness of SCS, as this measure was felt to be more valuable to share with future patients; furthermore, categorical analysis has been shown to be more valuable than ordinal pain scales to measure pain.14 In addition to wanting to expand the available research to the general public, we wanted a measure that we could easily relay to our patient population regarding SCS.
The explant rate of 14.5% among surveyed veterans falls at the higher end of the normal ranges found in previous studies of long-term SCS outcomes.15-17 One possible reason for the higher rate is that we did not differentiate based on the reason for the explant (ie, no benefit, further surgery needed for underlying medical condition, or SCS-specific complications). Another possible contributing factor to the higher than expected explant rate is the geographic location in the New Orleans metro area; New Orleans is considered to have one of the highest rates of obesity in the United States and obesity typically has other diseases associated with it such as hypertension and diabetes mellitus.
Limitations
Limitations of the study include the relatively low number of subjects, subjective nature of the interview questions, and the patients’ answers. Typically the POQ has been used as a prospective assessment of pain; whether it is valid in a retrospective analysis is not clear. While there was a statistically significant decrease of opioid use after getting SCS, this study can only show correlation, not causation. During the study period, there has been a drastic change in opioid prescribing patterns and efforts to decrease the amount of opioids prescribed.
Subjects also were asked to rate their pain and quality of life before SCS. Some subjects had SCS implantation up to 10 years prior to the phone interview. The variable amount of time between SCS implantation and interview likely affected subjects’ responses. Chronic pain is a moving target. Patients have good days and bad days that would likely change opinions on SCS benefits on a single phone interview. Some patients needed battery replacements at the time of the interview (battery life averaged about 3 to 5 years in our study population) and were asked to report current levels of pain from the perspective of when their batteries were still functional, further affecting results.
Conclusions
SCS was shown to improve the quality of life of US veterans at SLVHCS across a wide variety of metrics, including activities of daily living, as well as mental and physical health. For veterans with chronic intractable pain who have tried and failed more conservative treatments, SCS is a great treatment.
Lower back pain (LBP) affects an estimated 9.4% of the global population and has resulted in more years lived with disability than any other health condition.1 LBP affects a wide range of populations, but US veterans have been shown to have significantly higher rates of back pain than nonveterans. The National Institutes of Health reports that 65.6% of veterans experience chronic pain; 9.1% of veterans experience severe, chronic pain.2 Chronic back pain is treated by a range of methods, including medications, surgery, physical therapy (PT), patient education, and behavioral therapy.3 However, chronic neuropathic back pain has been shown to have limited responsiveness to medication.4
Neuropathic pain is caused by lesions in the somatosensory nervous system, resulting in spontaneous pain and amplified pain responses to both painful and nonpainful stimuli.5 The most common location for neuropathic pain is the back and legs. Between 10% and 40% of people who undergo lumbosacral spine surgery to treat neuropathic radicular pain will experience further neuropathic pain.6 This condition is referred to as failed back surgery syndrome or postlaminectomy syndrome (PLS). While neuropathic back pain has had limited responsiveness to medication and repeated lumbosacral spine surgery, spinal cord stimulation (SCS) has shown promise as an effective form of pain treatment for those experiencing PLS and other spine disorders.7-10 In addition, SCS therapy has had a very low incidence of complications, which may be on the decline with recent technological advancements.11 Patients with a diagnosis of PLS, LBP, or complex regional pain syndrome (CRPS) who have not responded to medications, therapy, and/or injections for ≥ 6 months were eligible for a trial of SCS therapy. Trial leads were placed via the percutaneous route with the battery strapped to the waistline for 3 to 5 days and were removed in clinic. Patients who experienced > 60% pain relief and functional improvement received a SCS implant.
The effectiveness of SCS has been demonstrated in a nonveteran population, but it has not been studied in a veteran population.12 US Department of Veterans Affairs (VA) health care coverage is different from Medicare and private insurance in that it is classified as a benefit and not insurance. The goals of treatment at the VA may include considerations in addition to feeling better, and patient presentations may not align with those in the private sector.
We hypothesize that SCS is both a safe and beneficial treatment option for veterans with chronic intractable spine and/or extremity pain. The purpose of this study was to determine the efficacy and safety of SCS in a veteran population.
Methods
The efficacy and safety of SCS was determined via a retrospective study. Inclusion criteria for the study consisted of any Southeastern Louisiana Veterans Health Care System (SLVHCS) patient who had an SCS trial and/or implant from 2008 to 2020. Eligible veterans must have had chronic pain for at least 6 months and had previously tried multiple medications, PT, transcutaneous nerve stimulation, facet injections, epidural steroid injections, or surgery without success. For medication therapy to be considered unsuccessful, it must have included acetaminophen, nonsteroidal anti-inflammatory drugs, and ≥ 1 adjuvant medication (gabapentin, duloxetine, amitriptyline, lidocaine, and menthol). A diagnosis of chronic LBP, PLS, cervical or lumbar spondylosis with radiculopathy, complex regional pain syndrome, or chronic pain syndrome was required for eligibility. Patients whose pain decreased by > 60% and had functional improvement in a 3- to 5-day trial received SCS implantation with percutaneous leads by a pain physician or paddle lead by a neurosurgeon.
The SLVHCS Institutional Review Board approved this study. Electronic health records were reviewed to determine patient age, anthropometric data, and date of SCS implantation. Patients were then called and interviewed to complete a survey. After obtaining verbal consent to the study, subjects were surveyed regarding whether the patient would recommend the procedure to peers, adverse effects (AEs) or complications, and the ability to decrease opiates if applicable. A verbal Pain Outcome Questionnaire (POQ) assessment of activities of daily living also was given during the phone interview regarding pain levels before SCS and at the time of the phone interview.13 (eAppendix available at doi:10.12788/fp.0204) Following the survey, a chart review was performed to corroborate the given AEs or complications and opiate use information. Before and after results of the POQ were compared via a paired sample t test, and P values < .05 were considered significant. Analyses were performed by IBM SPSS, version 26.
The primary outcome measure for this study was whether veterans would recommend SCS to their peers; in our view, this categorical outcome measure seemed to be more valuable to share with future patients who might be candidates for SCS. Since VA health care coverage and goals of treatment may be different from a nonveteran population, we opted to use this primary measure to decrease the possibility of confounding variables.
Secondary outcome measures included changes in POC scores, improvements in activities of daily living, and decreases in use of opioid pain medications.
POQ responses were recorded during the telephone interviews (0 to 10 scale). A paired sample t test was conducted to compare pain levels before and after SCS implant. Pain levels were gathered in the single phone call. Patient opioid usage, if applicable, was assessed by converting medications to morphine milligram equivalent dosing (MMED). Since patients who were on chronic opioids took multiple formulations, we changed the total daily dose to all morphine; for this study, morphine was considered equivalent to hydrocodone, and oxycodone was 1.5x morphine.
Results
Of the 90 SLVHCS patients who received an SCS implant between 2008 and 2020, 76 were reached by telephone and 65 had their responses recorded in the study. Of the 11 patients who were not included, 5 had the SCS removed; it is unclear whether these veterans would have recommended the treatment. Four were unable to quantify pain and/or SCS effects, and 2 were excluded due to a dementia diagnosis years after the implant. The mean (SD) age of participants was 63.9 (10.3) years. Forty percent of patients had a diabetes mellitus diagnosis and 1 had prediabetes. Patients’ most common qualifying diagnosis for SCS was PLS (47.7%) followed by chronic LBP (26.2%). A percutaneous 2-lead technique was the most common type of SCS type used (60.0%) followed by 1-lead (21.5%). The most common SCS manufacturer was Boston Scientific (87.7%)(Table 1). Most veterans (76.9%) recommended SCS to their peers; 13.8% did not recommend SCS; 9.2% were undecided and stated that they were unable to recommend because they did not want to persuade a peer to get SCS (Figure).
There was a statistically significant decrease in opioid use for the 40 veterans for whom pain medication was converted (P < .001)(Table 2). Six patients reported using opioids at some point but could not remember their dose, and no records were found in their chart review, so they were not included in the MMED analysis. In that group, 4 patients reported using opioids before SCS but discontinued the opioid use after SCS implantation, and 2 patients noted using opioids before SCS and concomitantly. Eighteen subjects reported no opioid use at any point before or after SCS (Table 3).
There were few life-threatening complications of SCS. Three veterans developed skin dehiscence; 2 had dehiscence at the battery/generator site, and 1 had dehiscence at the lead anchor site. Two patients with dehiscence also had morbid obesity, and the third had postoperative malnourishment. The dehiscence occurred 3 and 8 months postoperation. All 3 patients with dehiscence had the SCS explanted, though they were eager to get a new SCS implanted as soon as possible because SCS was their most successful treatment to date.
Twenty of the 64 veterans surveyed reported other complications of SCS, including lead migration, lack of pain coverage, paresthesia and numbness, soreness around generator site, SCS shocking patient when performing full thoracic spine flexion, and shingles at the battery site (Table 4). There were 11 explants among the 76 veterans contacted. The primary reason for explant was lack of pain coverage.
Patient concerns included pain with sitting in chairs due to tenderness around the implant, SCS helping with physical pain but not mental pain, SCS only working during the day and not helping with sleep, and patients lacking education regarding possible complications of SCS.
Discussion
In this nonrandomized retrospective review, SCS was shown to be an effective treatment for intractable spine and/or extremity pain. Veterans’ pain levels were significantly reduced following SCS implantation, and more than three-fourths of veterans recommended SCS to their peers. We used the recommendation of SCS to peers as the most important metric regarding the effectiveness of SCS, as this measure was felt to be more valuable to share with future patients; furthermore, categorical analysis has been shown to be more valuable than ordinal pain scales to measure pain.14 In addition to wanting to expand the available research to the general public, we wanted a measure that we could easily relay to our patient population regarding SCS.
The explant rate of 14.5% among surveyed veterans falls at the higher end of the normal ranges found in previous studies of long-term SCS outcomes.15-17 One possible reason for the higher rate is that we did not differentiate based on the reason for the explant (ie, no benefit, further surgery needed for underlying medical condition, or SCS-specific complications). Another possible contributing factor to the higher than expected explant rate is the geographic location in the New Orleans metro area; New Orleans is considered to have one of the highest rates of obesity in the United States and obesity typically has other diseases associated with it such as hypertension and diabetes mellitus.
Limitations
Limitations of the study include the relatively low number of subjects, subjective nature of the interview questions, and the patients’ answers. Typically the POQ has been used as a prospective assessment of pain; whether it is valid in a retrospective analysis is not clear. While there was a statistically significant decrease of opioid use after getting SCS, this study can only show correlation, not causation. During the study period, there has been a drastic change in opioid prescribing patterns and efforts to decrease the amount of opioids prescribed.
Subjects also were asked to rate their pain and quality of life before SCS. Some subjects had SCS implantation up to 10 years prior to the phone interview. The variable amount of time between SCS implantation and interview likely affected subjects’ responses. Chronic pain is a moving target. Patients have good days and bad days that would likely change opinions on SCS benefits on a single phone interview. Some patients needed battery replacements at the time of the interview (battery life averaged about 3 to 5 years in our study population) and were asked to report current levels of pain from the perspective of when their batteries were still functional, further affecting results.
Conclusions
SCS was shown to improve the quality of life of US veterans at SLVHCS across a wide variety of metrics, including activities of daily living, as well as mental and physical health. For veterans with chronic intractable pain who have tried and failed more conservative treatments, SCS is a great treatment.
1. Hoy DG, Smith E, Cross M, et al. The global burden of musculoskeletal conditions for 2010: an overview of methods. Ann Rheum Dis. 2014;73(6):982-989 doi:10.1136/annrheumdis-2013-204344
2. Nahin RL. Severe pain in veterans: the effect of age and sex, and comparisons with the general population. J Pain. 2017;18(3):247-254. doi:10.1016/j.jpain.2016.10.021
3. Institute of Medicine (US) Committee on Advancing Pain Research, Care, and Education. Relieving Pain in America: A Blueprint for Transforming Prevention, Care, Education, and Research. Washington, DC: National Academies Press; 2011.
4. Finnerup NB, Attal N, Haroutounian S, et al. Pharmacotherapy for neuropathic pain in adults: a systematic review and meta-analysis. Lancet Neurol. 2015;14(2):162-173. doi:10.1016/S1474-4422(14)70251-0
5. Costigan M, Scholz J, Woolf CJ. Neuropathic pain: a maladaptive response of the nervous system to damage. Annu Rev Neurosci. 2009;32:1-32. doi:10.1146/annurev.neuro.051508.135531
6. Wilkinson HA. The Failed Back Syndrome: Etiology and Therapy. 2nd ed. Harper & Row; 1991.
7. Kumar K, Taylor RS, Jacques L, et al. Spinal cord stimulation versus conventional medical management for neuropathic pain: a multicentre randomised controlled trial in patients with failed back surgery syndrome. Pain. 2007;132(1-2):179-188. doi:10.1016/j.pain.2007.07.028
8. North RB, Kidd DH, Farrokhi F, Piantadosi SA. Spinal cord stimulation versus repeated lumbosacral spine surgery for chronic pain: a randomized, controlled trial. Neurosurgery. 2005;56(1):98-107. doi:10.1227/01.neu.0000144839.65524.e0
9. Geurts JW, Smits H, Kemler MA, Brunner F, Kessels AG, van Kleef M. Spinal cord stimulation for complex regional pain syndrome type I: a prospective cohort study with long-term follow-up. Neuromodulation. 2013;16(6):523-529. doi:10.1111/ner.12024
10. Kumar K, Rizvi S, Bnurs SB. Spinal cord stimulation is effective in management of complex regional pain syndrome I: fact or fiction. Neurosurgery. 2011;69(3):566-5580. doi:10.1227/NEU.0b013e3182181e60
11. Mekhail NA, Mathews M, Nageeb F, Guirguis M, Mekhail MN, Cheng J. Retrospective review of 707 cases of spinal cord stimulation: indications and complications. Pain Pract. 2011;11(2):148-153. doi:10.1111/j.1533-2500.2010.00407.x
12. Veizi E, Hayek SM, North J, et al. Spinal cord stimulation (SCS) with anatomically guided (3D) neural targeting shows superior chronic axial low back pain relief compared to traditional SCS-LUMINA Study. Pain Med. 2017;18(8):1534-1548. doi:10.1093/pm/pnw286
13. Gordon DB, Polomano RC, Pellino TA, et al. Revised American Pain Society Patient Outcome Questionnaire (APS-POQ-R) for quality improvement of pain management in hospitalized adults: preliminary psychometric evaluation. J Pain. 2010;11(11):1172-1186. doi:10.1016/j.jpain.2010.02.012
14. Kennedy DJ, Schneider B. Lies, damn lies, and statistic: a commentary. Pain Med. 2020;21(10):2052-2054. doi:10.1093/pm/pnaa287
15. Van Buyten JP, Wille F, Smet I, et al. Therapy-related explants after spinal cord stimulation: results of an international retrospective chart review study. Neuromodulation. 2017;20(7):642-649. doi:10.1111/ner.12642
16. Hayek SM, Veizi E, Hanes M. Treatment-limiting complications of percutaneous spinal cord stimulator implants: a review of eight years of experience from an academic center database. Neuromodulation. 2015;18(7):603-609. doi:10.1111/ner.12312
17. Pope JE, Deer TR, Falowski S, et al. Multicenter retrospective study of neurostimulation with exit of therapy by explant. Neuromodulation. 2017;20(6):543-552. doi:10.1111/ner.12634
1. Hoy DG, Smith E, Cross M, et al. The global burden of musculoskeletal conditions for 2010: an overview of methods. Ann Rheum Dis. 2014;73(6):982-989 doi:10.1136/annrheumdis-2013-204344
2. Nahin RL. Severe pain in veterans: the effect of age and sex, and comparisons with the general population. J Pain. 2017;18(3):247-254. doi:10.1016/j.jpain.2016.10.021
3. Institute of Medicine (US) Committee on Advancing Pain Research, Care, and Education. Relieving Pain in America: A Blueprint for Transforming Prevention, Care, Education, and Research. Washington, DC: National Academies Press; 2011.
4. Finnerup NB, Attal N, Haroutounian S, et al. Pharmacotherapy for neuropathic pain in adults: a systematic review and meta-analysis. Lancet Neurol. 2015;14(2):162-173. doi:10.1016/S1474-4422(14)70251-0
5. Costigan M, Scholz J, Woolf CJ. Neuropathic pain: a maladaptive response of the nervous system to damage. Annu Rev Neurosci. 2009;32:1-32. doi:10.1146/annurev.neuro.051508.135531
6. Wilkinson HA. The Failed Back Syndrome: Etiology and Therapy. 2nd ed. Harper & Row; 1991.
7. Kumar K, Taylor RS, Jacques L, et al. Spinal cord stimulation versus conventional medical management for neuropathic pain: a multicentre randomised controlled trial in patients with failed back surgery syndrome. Pain. 2007;132(1-2):179-188. doi:10.1016/j.pain.2007.07.028
8. North RB, Kidd DH, Farrokhi F, Piantadosi SA. Spinal cord stimulation versus repeated lumbosacral spine surgery for chronic pain: a randomized, controlled trial. Neurosurgery. 2005;56(1):98-107. doi:10.1227/01.neu.0000144839.65524.e0
9. Geurts JW, Smits H, Kemler MA, Brunner F, Kessels AG, van Kleef M. Spinal cord stimulation for complex regional pain syndrome type I: a prospective cohort study with long-term follow-up. Neuromodulation. 2013;16(6):523-529. doi:10.1111/ner.12024
10. Kumar K, Rizvi S, Bnurs SB. Spinal cord stimulation is effective in management of complex regional pain syndrome I: fact or fiction. Neurosurgery. 2011;69(3):566-5580. doi:10.1227/NEU.0b013e3182181e60
11. Mekhail NA, Mathews M, Nageeb F, Guirguis M, Mekhail MN, Cheng J. Retrospective review of 707 cases of spinal cord stimulation: indications and complications. Pain Pract. 2011;11(2):148-153. doi:10.1111/j.1533-2500.2010.00407.x
12. Veizi E, Hayek SM, North J, et al. Spinal cord stimulation (SCS) with anatomically guided (3D) neural targeting shows superior chronic axial low back pain relief compared to traditional SCS-LUMINA Study. Pain Med. 2017;18(8):1534-1548. doi:10.1093/pm/pnw286
13. Gordon DB, Polomano RC, Pellino TA, et al. Revised American Pain Society Patient Outcome Questionnaire (APS-POQ-R) for quality improvement of pain management in hospitalized adults: preliminary psychometric evaluation. J Pain. 2010;11(11):1172-1186. doi:10.1016/j.jpain.2010.02.012
14. Kennedy DJ, Schneider B. Lies, damn lies, and statistic: a commentary. Pain Med. 2020;21(10):2052-2054. doi:10.1093/pm/pnaa287
15. Van Buyten JP, Wille F, Smet I, et al. Therapy-related explants after spinal cord stimulation: results of an international retrospective chart review study. Neuromodulation. 2017;20(7):642-649. doi:10.1111/ner.12642
16. Hayek SM, Veizi E, Hanes M. Treatment-limiting complications of percutaneous spinal cord stimulator implants: a review of eight years of experience from an academic center database. Neuromodulation. 2015;18(7):603-609. doi:10.1111/ner.12312
17. Pope JE, Deer TR, Falowski S, et al. Multicenter retrospective study of neurostimulation with exit of therapy by explant. Neuromodulation. 2017;20(6):543-552. doi:10.1111/ner.12634
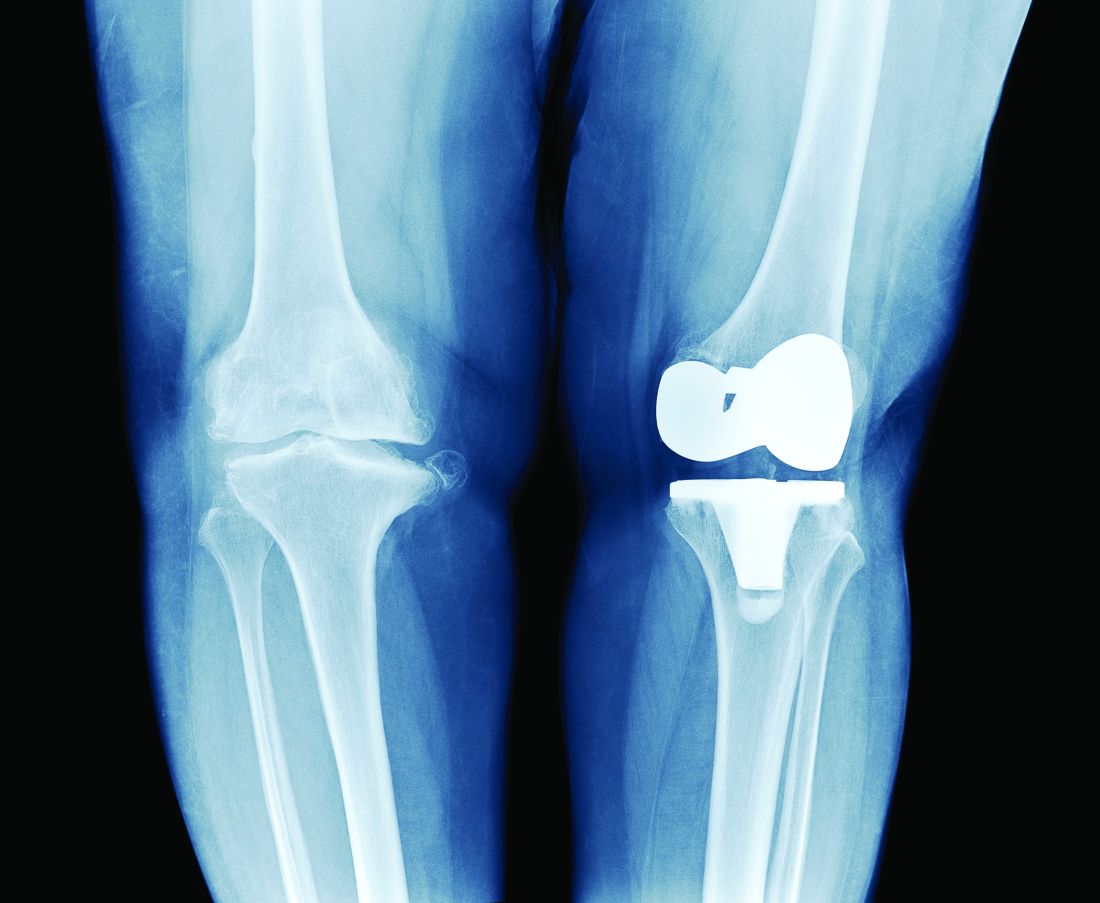
Rotating Hinge Distal Femur Replacement: A Turn for the Worse
Preoperatively periprosthetic joint infection with a postoperative complication of 180° rotation of the press-fit femoral component is a rare event, and knowledge of this possible complication is important for arthroplasty surgeons.
The use of a rotating hinge distal femur replacement (DFR) for significant bone and soft tissue defects in the setting of total knee arthroplasty (TKA) revision has become increasingly more common. Although significant advancements have been made in modern DFR components, complications and failure rates remain high. The unanticipated early failure presented serves as the first reported case in the literature to our knowledge of a 180° rotation of a press-fit DFR.
Originally, DFRs were used primarily for oncology patients with substantial bone loss following large mass excisions. The utility of DFRs has grown to include massive bone loss in the setting of TKA revision, periprosthetic fractures, and periprosthetic joint infections.1-3 DFRs help restore the joint line in the setting of significant bone loss and contain a rotating hinge mechanism that provides functional movement despite the loss of soft tissue constraints around the knee.1-3
DFRs have been associated with early postoperative mobilization and decreased need for ambulatory devices at 1 year in revision TKA and periprosthetic and geriatric distal femur fractures.4-6 Advances in prosthetic design, biomechanics, and fixation technique have led to improved survival rates.3 Despite these improvements, the overall complication rate remains high at 30 to 40%.3-7 Commonly reported complications after DFR include infection, aseptic loosening, soft tissue failure, and structural failure.3,4,7 Recent case studies also have reported on dislocation or disengagement of the rotating hinge.8-11
In this case report, we present a patient who had a DFR as the second stage of a 2-stage TKA revision due to a periprosthetic joint infection with a postoperative complication of 180° rotation of the press-fit femoral component. Although this is a rare event, knowledge of this possible complication is important for arthroplasty surgeons.
Case Presentation
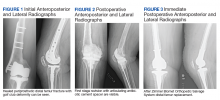
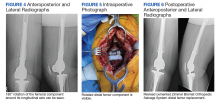
A patient with a history of hypertension, osteopenia, and rheumatoid arthritis underwent a primary right TKA in 2007. Ten weeks postoperatively, the patient had a ground-level fall that resulted in a right periprosthetic supracondylar distal femur fracture that was treated with a distal femur locking plate. The patient healed, however, with a significant golf club deformity (Figure 1). The patient did well for more than a decade but in 2019 was admitted with pelvic inflammatory disease and adnexal abscess that was treated with broad-spectrum IV antibiotics. Shortly after this admission, the patient developed a right knee periprosthetic infection with cultures positive for Ureaplasma parvum.
The patient then underwent a 2-stage revision of the infected TKA. Stage 1 consisted of explant of the TKA components as well as removal of the distal femur plate and screws and placement of an articulating antibiotic cement spacer (Figure 2). The patient completed 6 weeks of IV antibiotics. Following completion of the antibiotic course, we obtained a serum erythrocyte sedimentation rate, C-reactive protein level, and white blood cell count, which were all within normal limits. A knee aspiration was performed and did not show signs of residual infection. Frozen histopathology was sent during the second stage of the revision and did not show infection. After the results of the frozen histopathology returned, the antibiotic spacer was removed, and the femoral canal was thoroughly debrided. Cement and fibrous tissue in the femoral canal were carefully removed. In the setting of significant bone loss and soft tissue compromise due to the previous infection and distal femur fracture, the Zimmer Biomet Orthopedic Salvage System (OSS) with porous coated press-fit elliptical femoral stem was utilized.
The femoral canal was reamed until good cortical chatter was obtained at 16 mm. Per the Biomet OSS guide, “For bowed (curved) long and short press-fit stems, the final flexible reamer shaft diameter may need to be larger than the definitive trial and implant diameter.” After trialing, size 15.5 mm was selected for implantation. Intraoperatively the final stem was noted to have good interference fit after insertion and was stable throughout knee range of motion and varus/valgus stress testing. The patient did well with mobilization while in the hospital postoperatively and was discharged home (Figure 3).
Five days after discharge, the patient kicked the repaired knee onto a chair for rest and elevation and experienced extreme pain and was unable to flex the knee. On presentation to the emergency department, the X-rays showed 180° rotation around the longitudinal axis of the femoral component without any other obvious component failure or fracture (Figure 4). The patient was taken back to surgery the following day. Intraoperatively, the femoral stem was found to be loose and rotated 180° (Figure 5). No failure or dislocation of the tibial or rotating hinge components were identified. The press-fit femoral stem was removed and replaced with a cemented stem (Figure 6).
The postoperative course after the revision surgery was uneventful, and the patient is doing well clinically with no pain, functional range of motion of 5 to 105°, and has returned to regular activities without difficulty.
Discussion
Despite advancements in DFRs and increasing use in the setting of revision TKA, the procedure remains high risk with respect to postoperative complications.3-7 Vertesich and colleagues demonstrated that 43.3% of patients who underwent DFR for failed TKA developed at least 1 postoperative complication that required a return to the operating room.7Physicians need to be aware of the high rate of complications and counsel patients appropriately preoperatively.
Complications after DFR include infection, aseptic loosening, soft tissue failure, and structural failure.4,7 Soft tissue failures include insufficiency or rupture of the extensor mechanism and patella dislocation.4,7 Structural failures include fracture of the hinge mechanism, dissociation of the component from the stem, rotating hinge-bushing failure, and dislocation of the hinge.4,7 In the acute postoperative period, the most common complications are infection and rotating-hinge dislocation/failure.3,12 There are various component options available for DFRs, including straight vs curved, cemented vs cementless/press-fit, and long vs short stems.13 Studies have sought to elucidate the ideal implant to decrease the rate of complications. Lu and colleagues demonstrated that curved press-fit short stems provided a stable interface without loosening over the short term (2 years) in 42 patients.13 No implant failures or incidences of aseptic loosening occurred in their study.13
The implant used in this case was a curved press-fit short-stem DFR. It was thought that this patient was young and with good enough bone quality that a press-fit short stem would be best in preserving bone stock. Both the technique guide and literature support reaming 0 to 2 mm greater than the planned stem size to accommodate the implant curvature.13 In this case, the intramedullary canal was reamed 0.5 mm larger than the curved stem that was implanted (16 mm and 15.5 mm, respectively). Intraoperatively during the index DFR, the component was stable and seemed to have a good press-fit interface. Despite this, obvious loosening of the component occurred with a relatively low-energy mechanism when the patient kicked the leg onto a chair, causing just enough force and femoral rotation to result in 180° rotation of the component.
Conclusions
We present this case report to make surgeons aware of this rare but serious complication. Although the final implant is a porous and curved stem, careful attention should be made during trialing to use the best-fitting implant to prevent this complication. If an adequate interference fit cannot be obtained, cementing the component may be required to prevent its loosening and catastrophic failure.
1. Sculco PK, Abdel MP, Hanssen AD, Lewallen DG. The management of bone loss in revision total knee arthroplasty: rebuild, reinforce, and augment. Bone Joint J. 2016;98-B(1 suppl A):120-124. doi:10.1302/0301-620X.98B1.36345
2. Harrison RJ Jr, Thacker MM, Pitcher JD, Temple HT, Scully SP. Distal femur replacement is useful in complex total knee arthroplasty revisions. Clin Orthop Relat Res. 2006;446:113-120. doi:10.1097/01.blo.0000214433.64774.1b
3. Smith EL, Shah A, Son SJ, et al. Survivorship of megaprostheses in revision hip and knee arthroplasty for septic and aseptic indications: a retrospective, multicenter study with minimum 2-year follow-up. Arthroplast Today. 2020;6(3):475-479. Published 2020 Jun 29. doi:10.1016/j.artd.2020.05.004
4. Wyles CC, Tibbo ME, Yuan BJ, Trousdale RT, Berry DJ, Abdel MP. Long-term results of total knee arthroplasty with contemporary distal femoral replacement. J Bone Joint Surg Am. 2020;102(1):45-51. doi:10.2106/JBJS.19.00489
5. Haidukewych GJ. Role of distal femoral replacement for periprosthetic fractures above a total knee arthroplasty: when and how?, J Orthop Trauma. 2019;33(suppl 6):S33-S35. doi:10.1097/BOT.0000000000001566
6. Hart GP, Kneisl JS, Springer BD, Patt JC, Karunakar MA. Open reduction vs distal femoral replacement arthroplasty for comminuted distal femur fractures in the patients 70 years and older: J Arthroplasty. 2017;32(1):202-206. doi:10.1016/j.arth.2016.06.006
7. Vertesich K, Puchner SE, Staats K, et al. Distal femoral reconstruction following failed total knee arthroplasty is accompanied with risk for complication and reduced joint function. BMC Musculoskelet Disord. 2019 Jan 31;20(1):47-54. doi:10.1186/s12891-019-2432-4
8. Biswas D, Haughom B, Mayle RE Jr, Della Valle CJ. Case report: Failure of rotating-hinge total knee prosthesis by disengagement of the hinge-post extension. Clin Orthop Relat Res. 2013;471(4):1389-1392. doi:10.1007/s11999-012-2736-2
9. Ward WG, Haight D, Ritchie P, Gordon S, Eckardt JJ. Dislocation of rotating hinge knee prostheses. A report of four cases. J Bone Joint Surg Am. 2005;87(5):1108-1112. doi:10.2106/JBJS.00837pp
10. Pacha-Vicente D, Malik A, Castellet-Feliu E, Nardi-Vilardaga J. Dislocation of rotating-hinge knee prostheses with antidislocation mechanism. J Arthroplasty. 2008;23(2):299-303. doi:10.1016/j.arth.2006.11.020
11. Manzano G, Schwarzkopf R. Posterior dislocation of the hinge-post extension in a rotating hinge total knee prosthesis. Case Rep Orthop. 2013;2013:756538. doi:10.1155/2013/756538
12. Vaishya R., Thapa, SS, Vaish A. Non-neoplastic indications and outcomes of the proximal and distal femur megaprosthesis: a critical review. Knee Surg Relat Res. 2020;32(1):18. Published 2020 Apr 9. doi:10.1186/s43019-020-00034-7
13. Lu M, Wang J, Xiao C, et al. Uncemented, curved, short endoprosthesis stem for distal femoral reconstruction: early follow-up outcomes. World J Surg Onc. 2018;16(1):183. doi:10.1186/s12957-018-1486-3
Preoperatively periprosthetic joint infection with a postoperative complication of 180° rotation of the press-fit femoral component is a rare event, and knowledge of this possible complication is important for arthroplasty surgeons.
Preoperatively periprosthetic joint infection with a postoperative complication of 180° rotation of the press-fit femoral component is a rare event, and knowledge of this possible complication is important for arthroplasty surgeons.
The use of a rotating hinge distal femur replacement (DFR) for significant bone and soft tissue defects in the setting of total knee arthroplasty (TKA) revision has become increasingly more common. Although significant advancements have been made in modern DFR components, complications and failure rates remain high. The unanticipated early failure presented serves as the first reported case in the literature to our knowledge of a 180° rotation of a press-fit DFR.
Originally, DFRs were used primarily for oncology patients with substantial bone loss following large mass excisions. The utility of DFRs has grown to include massive bone loss in the setting of TKA revision, periprosthetic fractures, and periprosthetic joint infections.1-3 DFRs help restore the joint line in the setting of significant bone loss and contain a rotating hinge mechanism that provides functional movement despite the loss of soft tissue constraints around the knee.1-3
DFRs have been associated with early postoperative mobilization and decreased need for ambulatory devices at 1 year in revision TKA and periprosthetic and geriatric distal femur fractures.4-6 Advances in prosthetic design, biomechanics, and fixation technique have led to improved survival rates.3 Despite these improvements, the overall complication rate remains high at 30 to 40%.3-7 Commonly reported complications after DFR include infection, aseptic loosening, soft tissue failure, and structural failure.3,4,7 Recent case studies also have reported on dislocation or disengagement of the rotating hinge.8-11
In this case report, we present a patient who had a DFR as the second stage of a 2-stage TKA revision due to a periprosthetic joint infection with a postoperative complication of 180° rotation of the press-fit femoral component. Although this is a rare event, knowledge of this possible complication is important for arthroplasty surgeons.
Case Presentation
A patient with a history of hypertension, osteopenia, and rheumatoid arthritis underwent a primary right TKA in 2007. Ten weeks postoperatively, the patient had a ground-level fall that resulted in a right periprosthetic supracondylar distal femur fracture that was treated with a distal femur locking plate. The patient healed, however, with a significant golf club deformity (Figure 1). The patient did well for more than a decade but in 2019 was admitted with pelvic inflammatory disease and adnexal abscess that was treated with broad-spectrum IV antibiotics. Shortly after this admission, the patient developed a right knee periprosthetic infection with cultures positive for Ureaplasma parvum.
The patient then underwent a 2-stage revision of the infected TKA. Stage 1 consisted of explant of the TKA components as well as removal of the distal femur plate and screws and placement of an articulating antibiotic cement spacer (Figure 2). The patient completed 6 weeks of IV antibiotics. Following completion of the antibiotic course, we obtained a serum erythrocyte sedimentation rate, C-reactive protein level, and white blood cell count, which were all within normal limits. A knee aspiration was performed and did not show signs of residual infection. Frozen histopathology was sent during the second stage of the revision and did not show infection. After the results of the frozen histopathology returned, the antibiotic spacer was removed, and the femoral canal was thoroughly debrided. Cement and fibrous tissue in the femoral canal were carefully removed. In the setting of significant bone loss and soft tissue compromise due to the previous infection and distal femur fracture, the Zimmer Biomet Orthopedic Salvage System (OSS) with porous coated press-fit elliptical femoral stem was utilized.
The femoral canal was reamed until good cortical chatter was obtained at 16 mm. Per the Biomet OSS guide, “For bowed (curved) long and short press-fit stems, the final flexible reamer shaft diameter may need to be larger than the definitive trial and implant diameter.” After trialing, size 15.5 mm was selected for implantation. Intraoperatively the final stem was noted to have good interference fit after insertion and was stable throughout knee range of motion and varus/valgus stress testing. The patient did well with mobilization while in the hospital postoperatively and was discharged home (Figure 3).
Five days after discharge, the patient kicked the repaired knee onto a chair for rest and elevation and experienced extreme pain and was unable to flex the knee. On presentation to the emergency department, the X-rays showed 180° rotation around the longitudinal axis of the femoral component without any other obvious component failure or fracture (Figure 4). The patient was taken back to surgery the following day. Intraoperatively, the femoral stem was found to be loose and rotated 180° (Figure 5). No failure or dislocation of the tibial or rotating hinge components were identified. The press-fit femoral stem was removed and replaced with a cemented stem (Figure 6).
The postoperative course after the revision surgery was uneventful, and the patient is doing well clinically with no pain, functional range of motion of 5 to 105°, and has returned to regular activities without difficulty.
Discussion
Despite advancements in DFRs and increasing use in the setting of revision TKA, the procedure remains high risk with respect to postoperative complications.3-7 Vertesich and colleagues demonstrated that 43.3% of patients who underwent DFR for failed TKA developed at least 1 postoperative complication that required a return to the operating room.7Physicians need to be aware of the high rate of complications and counsel patients appropriately preoperatively.
Complications after DFR include infection, aseptic loosening, soft tissue failure, and structural failure.4,7 Soft tissue failures include insufficiency or rupture of the extensor mechanism and patella dislocation.4,7 Structural failures include fracture of the hinge mechanism, dissociation of the component from the stem, rotating hinge-bushing failure, and dislocation of the hinge.4,7 In the acute postoperative period, the most common complications are infection and rotating-hinge dislocation/failure.3,12 There are various component options available for DFRs, including straight vs curved, cemented vs cementless/press-fit, and long vs short stems.13 Studies have sought to elucidate the ideal implant to decrease the rate of complications. Lu and colleagues demonstrated that curved press-fit short stems provided a stable interface without loosening over the short term (2 years) in 42 patients.13 No implant failures or incidences of aseptic loosening occurred in their study.13
The implant used in this case was a curved press-fit short-stem DFR. It was thought that this patient was young and with good enough bone quality that a press-fit short stem would be best in preserving bone stock. Both the technique guide and literature support reaming 0 to 2 mm greater than the planned stem size to accommodate the implant curvature.13 In this case, the intramedullary canal was reamed 0.5 mm larger than the curved stem that was implanted (16 mm and 15.5 mm, respectively). Intraoperatively during the index DFR, the component was stable and seemed to have a good press-fit interface. Despite this, obvious loosening of the component occurred with a relatively low-energy mechanism when the patient kicked the leg onto a chair, causing just enough force and femoral rotation to result in 180° rotation of the component.
Conclusions
We present this case report to make surgeons aware of this rare but serious complication. Although the final implant is a porous and curved stem, careful attention should be made during trialing to use the best-fitting implant to prevent this complication. If an adequate interference fit cannot be obtained, cementing the component may be required to prevent its loosening and catastrophic failure.
The use of a rotating hinge distal femur replacement (DFR) for significant bone and soft tissue defects in the setting of total knee arthroplasty (TKA) revision has become increasingly more common. Although significant advancements have been made in modern DFR components, complications and failure rates remain high. The unanticipated early failure presented serves as the first reported case in the literature to our knowledge of a 180° rotation of a press-fit DFR.
Originally, DFRs were used primarily for oncology patients with substantial bone loss following large mass excisions. The utility of DFRs has grown to include massive bone loss in the setting of TKA revision, periprosthetic fractures, and periprosthetic joint infections.1-3 DFRs help restore the joint line in the setting of significant bone loss and contain a rotating hinge mechanism that provides functional movement despite the loss of soft tissue constraints around the knee.1-3
DFRs have been associated with early postoperative mobilization and decreased need for ambulatory devices at 1 year in revision TKA and periprosthetic and geriatric distal femur fractures.4-6 Advances in prosthetic design, biomechanics, and fixation technique have led to improved survival rates.3 Despite these improvements, the overall complication rate remains high at 30 to 40%.3-7 Commonly reported complications after DFR include infection, aseptic loosening, soft tissue failure, and structural failure.3,4,7 Recent case studies also have reported on dislocation or disengagement of the rotating hinge.8-11
In this case report, we present a patient who had a DFR as the second stage of a 2-stage TKA revision due to a periprosthetic joint infection with a postoperative complication of 180° rotation of the press-fit femoral component. Although this is a rare event, knowledge of this possible complication is important for arthroplasty surgeons.
Case Presentation
A patient with a history of hypertension, osteopenia, and rheumatoid arthritis underwent a primary right TKA in 2007. Ten weeks postoperatively, the patient had a ground-level fall that resulted in a right periprosthetic supracondylar distal femur fracture that was treated with a distal femur locking plate. The patient healed, however, with a significant golf club deformity (Figure 1). The patient did well for more than a decade but in 2019 was admitted with pelvic inflammatory disease and adnexal abscess that was treated with broad-spectrum IV antibiotics. Shortly after this admission, the patient developed a right knee periprosthetic infection with cultures positive for Ureaplasma parvum.
The patient then underwent a 2-stage revision of the infected TKA. Stage 1 consisted of explant of the TKA components as well as removal of the distal femur plate and screws and placement of an articulating antibiotic cement spacer (Figure 2). The patient completed 6 weeks of IV antibiotics. Following completion of the antibiotic course, we obtained a serum erythrocyte sedimentation rate, C-reactive protein level, and white blood cell count, which were all within normal limits. A knee aspiration was performed and did not show signs of residual infection. Frozen histopathology was sent during the second stage of the revision and did not show infection. After the results of the frozen histopathology returned, the antibiotic spacer was removed, and the femoral canal was thoroughly debrided. Cement and fibrous tissue in the femoral canal were carefully removed. In the setting of significant bone loss and soft tissue compromise due to the previous infection and distal femur fracture, the Zimmer Biomet Orthopedic Salvage System (OSS) with porous coated press-fit elliptical femoral stem was utilized.
The femoral canal was reamed until good cortical chatter was obtained at 16 mm. Per the Biomet OSS guide, “For bowed (curved) long and short press-fit stems, the final flexible reamer shaft diameter may need to be larger than the definitive trial and implant diameter.” After trialing, size 15.5 mm was selected for implantation. Intraoperatively the final stem was noted to have good interference fit after insertion and was stable throughout knee range of motion and varus/valgus stress testing. The patient did well with mobilization while in the hospital postoperatively and was discharged home (Figure 3).
Five days after discharge, the patient kicked the repaired knee onto a chair for rest and elevation and experienced extreme pain and was unable to flex the knee. On presentation to the emergency department, the X-rays showed 180° rotation around the longitudinal axis of the femoral component without any other obvious component failure or fracture (Figure 4). The patient was taken back to surgery the following day. Intraoperatively, the femoral stem was found to be loose and rotated 180° (Figure 5). No failure or dislocation of the tibial or rotating hinge components were identified. The press-fit femoral stem was removed and replaced with a cemented stem (Figure 6).
The postoperative course after the revision surgery was uneventful, and the patient is doing well clinically with no pain, functional range of motion of 5 to 105°, and has returned to regular activities without difficulty.
Discussion
Despite advancements in DFRs and increasing use in the setting of revision TKA, the procedure remains high risk with respect to postoperative complications.3-7 Vertesich and colleagues demonstrated that 43.3% of patients who underwent DFR for failed TKA developed at least 1 postoperative complication that required a return to the operating room.7Physicians need to be aware of the high rate of complications and counsel patients appropriately preoperatively.
Complications after DFR include infection, aseptic loosening, soft tissue failure, and structural failure.4,7 Soft tissue failures include insufficiency or rupture of the extensor mechanism and patella dislocation.4,7 Structural failures include fracture of the hinge mechanism, dissociation of the component from the stem, rotating hinge-bushing failure, and dislocation of the hinge.4,7 In the acute postoperative period, the most common complications are infection and rotating-hinge dislocation/failure.3,12 There are various component options available for DFRs, including straight vs curved, cemented vs cementless/press-fit, and long vs short stems.13 Studies have sought to elucidate the ideal implant to decrease the rate of complications. Lu and colleagues demonstrated that curved press-fit short stems provided a stable interface without loosening over the short term (2 years) in 42 patients.13 No implant failures or incidences of aseptic loosening occurred in their study.13
The implant used in this case was a curved press-fit short-stem DFR. It was thought that this patient was young and with good enough bone quality that a press-fit short stem would be best in preserving bone stock. Both the technique guide and literature support reaming 0 to 2 mm greater than the planned stem size to accommodate the implant curvature.13 In this case, the intramedullary canal was reamed 0.5 mm larger than the curved stem that was implanted (16 mm and 15.5 mm, respectively). Intraoperatively during the index DFR, the component was stable and seemed to have a good press-fit interface. Despite this, obvious loosening of the component occurred with a relatively low-energy mechanism when the patient kicked the leg onto a chair, causing just enough force and femoral rotation to result in 180° rotation of the component.
Conclusions
We present this case report to make surgeons aware of this rare but serious complication. Although the final implant is a porous and curved stem, careful attention should be made during trialing to use the best-fitting implant to prevent this complication. If an adequate interference fit cannot be obtained, cementing the component may be required to prevent its loosening and catastrophic failure.
1. Sculco PK, Abdel MP, Hanssen AD, Lewallen DG. The management of bone loss in revision total knee arthroplasty: rebuild, reinforce, and augment. Bone Joint J. 2016;98-B(1 suppl A):120-124. doi:10.1302/0301-620X.98B1.36345
2. Harrison RJ Jr, Thacker MM, Pitcher JD, Temple HT, Scully SP. Distal femur replacement is useful in complex total knee arthroplasty revisions. Clin Orthop Relat Res. 2006;446:113-120. doi:10.1097/01.blo.0000214433.64774.1b
3. Smith EL, Shah A, Son SJ, et al. Survivorship of megaprostheses in revision hip and knee arthroplasty for septic and aseptic indications: a retrospective, multicenter study with minimum 2-year follow-up. Arthroplast Today. 2020;6(3):475-479. Published 2020 Jun 29. doi:10.1016/j.artd.2020.05.004
4. Wyles CC, Tibbo ME, Yuan BJ, Trousdale RT, Berry DJ, Abdel MP. Long-term results of total knee arthroplasty with contemporary distal femoral replacement. J Bone Joint Surg Am. 2020;102(1):45-51. doi:10.2106/JBJS.19.00489
5. Haidukewych GJ. Role of distal femoral replacement for periprosthetic fractures above a total knee arthroplasty: when and how?, J Orthop Trauma. 2019;33(suppl 6):S33-S35. doi:10.1097/BOT.0000000000001566
6. Hart GP, Kneisl JS, Springer BD, Patt JC, Karunakar MA. Open reduction vs distal femoral replacement arthroplasty for comminuted distal femur fractures in the patients 70 years and older: J Arthroplasty. 2017;32(1):202-206. doi:10.1016/j.arth.2016.06.006
7. Vertesich K, Puchner SE, Staats K, et al. Distal femoral reconstruction following failed total knee arthroplasty is accompanied with risk for complication and reduced joint function. BMC Musculoskelet Disord. 2019 Jan 31;20(1):47-54. doi:10.1186/s12891-019-2432-4
8. Biswas D, Haughom B, Mayle RE Jr, Della Valle CJ. Case report: Failure of rotating-hinge total knee prosthesis by disengagement of the hinge-post extension. Clin Orthop Relat Res. 2013;471(4):1389-1392. doi:10.1007/s11999-012-2736-2
9. Ward WG, Haight D, Ritchie P, Gordon S, Eckardt JJ. Dislocation of rotating hinge knee prostheses. A report of four cases. J Bone Joint Surg Am. 2005;87(5):1108-1112. doi:10.2106/JBJS.00837pp
10. Pacha-Vicente D, Malik A, Castellet-Feliu E, Nardi-Vilardaga J. Dislocation of rotating-hinge knee prostheses with antidislocation mechanism. J Arthroplasty. 2008;23(2):299-303. doi:10.1016/j.arth.2006.11.020
11. Manzano G, Schwarzkopf R. Posterior dislocation of the hinge-post extension in a rotating hinge total knee prosthesis. Case Rep Orthop. 2013;2013:756538. doi:10.1155/2013/756538
12. Vaishya R., Thapa, SS, Vaish A. Non-neoplastic indications and outcomes of the proximal and distal femur megaprosthesis: a critical review. Knee Surg Relat Res. 2020;32(1):18. Published 2020 Apr 9. doi:10.1186/s43019-020-00034-7
13. Lu M, Wang J, Xiao C, et al. Uncemented, curved, short endoprosthesis stem for distal femoral reconstruction: early follow-up outcomes. World J Surg Onc. 2018;16(1):183. doi:10.1186/s12957-018-1486-3
1. Sculco PK, Abdel MP, Hanssen AD, Lewallen DG. The management of bone loss in revision total knee arthroplasty: rebuild, reinforce, and augment. Bone Joint J. 2016;98-B(1 suppl A):120-124. doi:10.1302/0301-620X.98B1.36345
2. Harrison RJ Jr, Thacker MM, Pitcher JD, Temple HT, Scully SP. Distal femur replacement is useful in complex total knee arthroplasty revisions. Clin Orthop Relat Res. 2006;446:113-120. doi:10.1097/01.blo.0000214433.64774.1b
3. Smith EL, Shah A, Son SJ, et al. Survivorship of megaprostheses in revision hip and knee arthroplasty for septic and aseptic indications: a retrospective, multicenter study with minimum 2-year follow-up. Arthroplast Today. 2020;6(3):475-479. Published 2020 Jun 29. doi:10.1016/j.artd.2020.05.004
4. Wyles CC, Tibbo ME, Yuan BJ, Trousdale RT, Berry DJ, Abdel MP. Long-term results of total knee arthroplasty with contemporary distal femoral replacement. J Bone Joint Surg Am. 2020;102(1):45-51. doi:10.2106/JBJS.19.00489
5. Haidukewych GJ. Role of distal femoral replacement for periprosthetic fractures above a total knee arthroplasty: when and how?, J Orthop Trauma. 2019;33(suppl 6):S33-S35. doi:10.1097/BOT.0000000000001566
6. Hart GP, Kneisl JS, Springer BD, Patt JC, Karunakar MA. Open reduction vs distal femoral replacement arthroplasty for comminuted distal femur fractures in the patients 70 years and older: J Arthroplasty. 2017;32(1):202-206. doi:10.1016/j.arth.2016.06.006
7. Vertesich K, Puchner SE, Staats K, et al. Distal femoral reconstruction following failed total knee arthroplasty is accompanied with risk for complication and reduced joint function. BMC Musculoskelet Disord. 2019 Jan 31;20(1):47-54. doi:10.1186/s12891-019-2432-4
8. Biswas D, Haughom B, Mayle RE Jr, Della Valle CJ. Case report: Failure of rotating-hinge total knee prosthesis by disengagement of the hinge-post extension. Clin Orthop Relat Res. 2013;471(4):1389-1392. doi:10.1007/s11999-012-2736-2
9. Ward WG, Haight D, Ritchie P, Gordon S, Eckardt JJ. Dislocation of rotating hinge knee prostheses. A report of four cases. J Bone Joint Surg Am. 2005;87(5):1108-1112. doi:10.2106/JBJS.00837pp
10. Pacha-Vicente D, Malik A, Castellet-Feliu E, Nardi-Vilardaga J. Dislocation of rotating-hinge knee prostheses with antidislocation mechanism. J Arthroplasty. 2008;23(2):299-303. doi:10.1016/j.arth.2006.11.020
11. Manzano G, Schwarzkopf R. Posterior dislocation of the hinge-post extension in a rotating hinge total knee prosthesis. Case Rep Orthop. 2013;2013:756538. doi:10.1155/2013/756538
12. Vaishya R., Thapa, SS, Vaish A. Non-neoplastic indications and outcomes of the proximal and distal femur megaprosthesis: a critical review. Knee Surg Relat Res. 2020;32(1):18. Published 2020 Apr 9. doi:10.1186/s43019-020-00034-7
13. Lu M, Wang J, Xiao C, et al. Uncemented, curved, short endoprosthesis stem for distal femoral reconstruction: early follow-up outcomes. World J Surg Onc. 2018;16(1):183. doi:10.1186/s12957-018-1486-3
Therapeutic aquatic exercise superior to physical therapy for back pain in study
“This is the first study to compare the efficacy of therapeutic aquatic exercise and physical therapy modalities in the treatment of chronic low back pain,” senior coauthors Pei-Jie Chen, PhD and Xue-Qiang Wang, PhD, both of the department of sport rehabilitation, Shanghai (China) University of Sport, wrote in JAMA Network Open. “Therapeutic aquatic exercise is a safe treatment for chronic low back pain and most participants who received it were willing to recommend it to other patients with chronic low back pain.”
As compared with individuals in the physical therapy modalities arm, the therapeutic aquatic exercise experienced greater relief of disability at all time points assessed: after the 3-month intervention, at the 6-month follow-up, and at the 12-month follow-up.
Commenting on the study, Linda Girgis, MD, FAAFP, a family physician in private practice in South River, N.J., agreed that aquatic therapy is a great tool for many chronic low back patients. “It helps them get active for one and do things that may exacerbate their symptoms doing the same exercises on land,” noted Dr. Girgis, who also is a clinical assistant professor at Robert Wood Johnson Medical School, New Brunswick.
She pointed out that access to a pool can be a problem. “But I have found a few physical therapy places in my area that do have access to a pool, and I refer appropriate patients there,” added Dr. Girgis, who was not involved with the study. “I have also found it works well for other types of pain, such as knee and hip pain. It is not for everyone but I have seen some patients get great benefit from it when they didn’t get any with traditional physical therapy.”
Aquatic therapy was more beneficial
Low back pain is a common condition, and clinical practice guidelines currently recommend therapeutic exercise and physical therapy modalities. Among the modalities that are available, therapeutic aquatic exercise is often prescribed for chronic low back pain, and it is becoming increasingly popular for treatment of chronic low back pain, the authors stated in their paper. The authors noted that water is an ideal environment for conducting an exercise program given its various properties, including buoyancy pressure, density, thermal capacity, and conductivity.
Two previously published systematic reviews have suggested that therapeutic aquatic exercise may be able to reduce the intensity of back pain and improve function in this population. But to date, evidence regarding long-term benefits in patients with chronic low back pain is very limited and there haven’t been any studies comparing the efficacy of therapeutic aquatic exercise and physical therapy modalities for chronic low back pain, according to the authors.
In this study, 113 individuals with chronic low back pain were randomized to either therapeutic aquatic exercise or to physical therapy, with an endpoint of efficacy regarding disability. This was measured using the Roland-Morris Disability Questionnaire.
Scores ranged from 0 to 24, with higher scores indicating more severe disability. Secondary endpoints included pain intensity, quality of life, sleep quality, and recommendation of intervention, and these were rated using various standardized tools.
Those randomized to the therapeutic aquatic exercise group had about an hour of therapy, beginning with a 10-minute active warm-up session to enhance neuromuscular activation, then an exercise session for 40 minutes followed by a 10-minute cooldown.
The physical therapy group received transcutaneous electrical nerve stimulation and infrared ray thermal therapy, also for 60 minutes. Both groups received these interventions twice a week for 3 months.
The overall mean age of the cohort was 31.0 years, and they were almost evenly divided by gender; 54 were men (47.8%), and 59 were women (52.2%).
As compared with the physical therapy group, individuals participating in therapeutic aquatic exercise group showed improvement in disability by an additional −1.77 points (95% confidence interval, −3.02 to −0.51) at the end of the 3-month intervention; at 6 months it was −2.42 points (95% CI, −4.13 to −0.70) and −3.61 points (95% CI, −5.63 to −1.58) at the 12-month follow-up (P < .001 for overall group x time interaction).
Functional improvement did not appear to be significantly affected by confounders that included age, sex, body mass index, low back pain duration, educational level, or pain level.
For secondary outcomes, those in the therapeutic aquatic exercise group demonstrated improvement in the most severe pain by an additional −0.79 points (95% CI, −1.31 to −0.27) after the 3-month intervention, −1.34 points (95% CI, −2.06 to −0.62) at 6 months, and −2.04 points (95% CI, −2.75 to −1.34) at the 12-month follow-up (P < .001 for overall group x time interaction), as compared with the physical therapy group. All pain scores differed significantly between the two groups at every time point.
In addition, individuals in the therapeutic aquatic exercise group showed more improvements on the 36-item Short-form Health Survey (overall group x time interaction, P = .003), Pittsburgh Sleep Quality Index (overall group x time interaction, P = .02), Tampa Scale for Kinesiophobia (overall group x time interaction, P < .001), and Fear-Avoidance Beliefs Questionnaire (physical activity subscale overall group x time interaction, P = .04), as compared with the physical therapy group. These improvements were also not influenced by confounders.
Finally, at the 12-month follow-up point, those in the aquatic therapy group had significantly greater improvements in the number of participants who met the minimal clinically important difference in pain (at least a 2-point improvement on the numeric rating scale).
More outside experts’ takes
“The current research evidence does suggest indeed that aquatic exercise therapy is suitable and often better than land exercise, passive relaxation, or other treatments for many people with low back pain,” commented Stelios Psycharakis PhD, senior lecturer in biomechanics, Institute for Sport, Physical Education and Health Sciences, University of Edinburgh.
He also noted that since low back pain is an issue affecting about 80% of all people at some stage of their life, it is “improbable that one could identify a single type of treatment or exercise therapy that would be suitable for every person with this problem.”
Dr. Psycharakis pointed out that there are also some contraindications for aquatic therapy, such as incontinence and skin conditions. “Other than that though, clinicians should definitely consider aquatic exercise therapy when advising people with chronic low back pain,” he said.
Justin M. Lantz, DPT, agreed that the study showed therapeutic aquatic exercise appears to be safe and beneficial in some patients with chronic low back pain, but he also shared limitations of the new research.
“The study has notable limitations as it did not include patients above 65 years old, pain levels were generally low for the subjects involved, and it did not include a treatment group with land therapeutic exercise – so it is difficult to determine if the beneficial effects reported were due to active exercise or because the exercises were performed in water,” said Dr. Lantz, director of the spine physical therapy fellowship program at the University of Southern California, Los Angeles, and an assistant professor of clinical physical therapy.
He also pointed out that, since active exercise has been shown to be beneficial and is advocated in multiple clinical practice guidelines for chronic low back pain, “it would be helpful to determine if the true effects on pain and disability were due to the water environment or the effect of active exercise itself.”
“Due to the significant positive long-term effects and limited adverse events reported, I believe this study supports the use of therapeutic aquatic exercise in select patient populations with chronic low back pain and should be considered as a part of a rehabilitation treatment plan if accessibility is feasible,” Dr. Lantz said.
The authors of the paper, Dr. Girgis, and Dr. Psycharakis had no conflicts of interest. Justin Lantz is a physical therapy consultant to SI-Bone.
“This is the first study to compare the efficacy of therapeutic aquatic exercise and physical therapy modalities in the treatment of chronic low back pain,” senior coauthors Pei-Jie Chen, PhD and Xue-Qiang Wang, PhD, both of the department of sport rehabilitation, Shanghai (China) University of Sport, wrote in JAMA Network Open. “Therapeutic aquatic exercise is a safe treatment for chronic low back pain and most participants who received it were willing to recommend it to other patients with chronic low back pain.”
As compared with individuals in the physical therapy modalities arm, the therapeutic aquatic exercise experienced greater relief of disability at all time points assessed: after the 3-month intervention, at the 6-month follow-up, and at the 12-month follow-up.
Commenting on the study, Linda Girgis, MD, FAAFP, a family physician in private practice in South River, N.J., agreed that aquatic therapy is a great tool for many chronic low back patients. “It helps them get active for one and do things that may exacerbate their symptoms doing the same exercises on land,” noted Dr. Girgis, who also is a clinical assistant professor at Robert Wood Johnson Medical School, New Brunswick.
She pointed out that access to a pool can be a problem. “But I have found a few physical therapy places in my area that do have access to a pool, and I refer appropriate patients there,” added Dr. Girgis, who was not involved with the study. “I have also found it works well for other types of pain, such as knee and hip pain. It is not for everyone but I have seen some patients get great benefit from it when they didn’t get any with traditional physical therapy.”
Aquatic therapy was more beneficial
Low back pain is a common condition, and clinical practice guidelines currently recommend therapeutic exercise and physical therapy modalities. Among the modalities that are available, therapeutic aquatic exercise is often prescribed for chronic low back pain, and it is becoming increasingly popular for treatment of chronic low back pain, the authors stated in their paper. The authors noted that water is an ideal environment for conducting an exercise program given its various properties, including buoyancy pressure, density, thermal capacity, and conductivity.
Two previously published systematic reviews have suggested that therapeutic aquatic exercise may be able to reduce the intensity of back pain and improve function in this population. But to date, evidence regarding long-term benefits in patients with chronic low back pain is very limited and there haven’t been any studies comparing the efficacy of therapeutic aquatic exercise and physical therapy modalities for chronic low back pain, according to the authors.
In this study, 113 individuals with chronic low back pain were randomized to either therapeutic aquatic exercise or to physical therapy, with an endpoint of efficacy regarding disability. This was measured using the Roland-Morris Disability Questionnaire.
Scores ranged from 0 to 24, with higher scores indicating more severe disability. Secondary endpoints included pain intensity, quality of life, sleep quality, and recommendation of intervention, and these were rated using various standardized tools.
Those randomized to the therapeutic aquatic exercise group had about an hour of therapy, beginning with a 10-minute active warm-up session to enhance neuromuscular activation, then an exercise session for 40 minutes followed by a 10-minute cooldown.
The physical therapy group received transcutaneous electrical nerve stimulation and infrared ray thermal therapy, also for 60 minutes. Both groups received these interventions twice a week for 3 months.
The overall mean age of the cohort was 31.0 years, and they were almost evenly divided by gender; 54 were men (47.8%), and 59 were women (52.2%).
As compared with the physical therapy group, individuals participating in therapeutic aquatic exercise group showed improvement in disability by an additional −1.77 points (95% confidence interval, −3.02 to −0.51) at the end of the 3-month intervention; at 6 months it was −2.42 points (95% CI, −4.13 to −0.70) and −3.61 points (95% CI, −5.63 to −1.58) at the 12-month follow-up (P < .001 for overall group x time interaction).
Functional improvement did not appear to be significantly affected by confounders that included age, sex, body mass index, low back pain duration, educational level, or pain level.
For secondary outcomes, those in the therapeutic aquatic exercise group demonstrated improvement in the most severe pain by an additional −0.79 points (95% CI, −1.31 to −0.27) after the 3-month intervention, −1.34 points (95% CI, −2.06 to −0.62) at 6 months, and −2.04 points (95% CI, −2.75 to −1.34) at the 12-month follow-up (P < .001 for overall group x time interaction), as compared with the physical therapy group. All pain scores differed significantly between the two groups at every time point.
In addition, individuals in the therapeutic aquatic exercise group showed more improvements on the 36-item Short-form Health Survey (overall group x time interaction, P = .003), Pittsburgh Sleep Quality Index (overall group x time interaction, P = .02), Tampa Scale for Kinesiophobia (overall group x time interaction, P < .001), and Fear-Avoidance Beliefs Questionnaire (physical activity subscale overall group x time interaction, P = .04), as compared with the physical therapy group. These improvements were also not influenced by confounders.
Finally, at the 12-month follow-up point, those in the aquatic therapy group had significantly greater improvements in the number of participants who met the minimal clinically important difference in pain (at least a 2-point improvement on the numeric rating scale).
More outside experts’ takes
“The current research evidence does suggest indeed that aquatic exercise therapy is suitable and often better than land exercise, passive relaxation, or other treatments for many people with low back pain,” commented Stelios Psycharakis PhD, senior lecturer in biomechanics, Institute for Sport, Physical Education and Health Sciences, University of Edinburgh.
He also noted that since low back pain is an issue affecting about 80% of all people at some stage of their life, it is “improbable that one could identify a single type of treatment or exercise therapy that would be suitable for every person with this problem.”
Dr. Psycharakis pointed out that there are also some contraindications for aquatic therapy, such as incontinence and skin conditions. “Other than that though, clinicians should definitely consider aquatic exercise therapy when advising people with chronic low back pain,” he said.
Justin M. Lantz, DPT, agreed that the study showed therapeutic aquatic exercise appears to be safe and beneficial in some patients with chronic low back pain, but he also shared limitations of the new research.
“The study has notable limitations as it did not include patients above 65 years old, pain levels were generally low for the subjects involved, and it did not include a treatment group with land therapeutic exercise – so it is difficult to determine if the beneficial effects reported were due to active exercise or because the exercises were performed in water,” said Dr. Lantz, director of the spine physical therapy fellowship program at the University of Southern California, Los Angeles, and an assistant professor of clinical physical therapy.
He also pointed out that, since active exercise has been shown to be beneficial and is advocated in multiple clinical practice guidelines for chronic low back pain, “it would be helpful to determine if the true effects on pain and disability were due to the water environment or the effect of active exercise itself.”
“Due to the significant positive long-term effects and limited adverse events reported, I believe this study supports the use of therapeutic aquatic exercise in select patient populations with chronic low back pain and should be considered as a part of a rehabilitation treatment plan if accessibility is feasible,” Dr. Lantz said.
The authors of the paper, Dr. Girgis, and Dr. Psycharakis had no conflicts of interest. Justin Lantz is a physical therapy consultant to SI-Bone.
“This is the first study to compare the efficacy of therapeutic aquatic exercise and physical therapy modalities in the treatment of chronic low back pain,” senior coauthors Pei-Jie Chen, PhD and Xue-Qiang Wang, PhD, both of the department of sport rehabilitation, Shanghai (China) University of Sport, wrote in JAMA Network Open. “Therapeutic aquatic exercise is a safe treatment for chronic low back pain and most participants who received it were willing to recommend it to other patients with chronic low back pain.”
As compared with individuals in the physical therapy modalities arm, the therapeutic aquatic exercise experienced greater relief of disability at all time points assessed: after the 3-month intervention, at the 6-month follow-up, and at the 12-month follow-up.
Commenting on the study, Linda Girgis, MD, FAAFP, a family physician in private practice in South River, N.J., agreed that aquatic therapy is a great tool for many chronic low back patients. “It helps them get active for one and do things that may exacerbate their symptoms doing the same exercises on land,” noted Dr. Girgis, who also is a clinical assistant professor at Robert Wood Johnson Medical School, New Brunswick.
She pointed out that access to a pool can be a problem. “But I have found a few physical therapy places in my area that do have access to a pool, and I refer appropriate patients there,” added Dr. Girgis, who was not involved with the study. “I have also found it works well for other types of pain, such as knee and hip pain. It is not for everyone but I have seen some patients get great benefit from it when they didn’t get any with traditional physical therapy.”
Aquatic therapy was more beneficial
Low back pain is a common condition, and clinical practice guidelines currently recommend therapeutic exercise and physical therapy modalities. Among the modalities that are available, therapeutic aquatic exercise is often prescribed for chronic low back pain, and it is becoming increasingly popular for treatment of chronic low back pain, the authors stated in their paper. The authors noted that water is an ideal environment for conducting an exercise program given its various properties, including buoyancy pressure, density, thermal capacity, and conductivity.
Two previously published systematic reviews have suggested that therapeutic aquatic exercise may be able to reduce the intensity of back pain and improve function in this population. But to date, evidence regarding long-term benefits in patients with chronic low back pain is very limited and there haven’t been any studies comparing the efficacy of therapeutic aquatic exercise and physical therapy modalities for chronic low back pain, according to the authors.
In this study, 113 individuals with chronic low back pain were randomized to either therapeutic aquatic exercise or to physical therapy, with an endpoint of efficacy regarding disability. This was measured using the Roland-Morris Disability Questionnaire.
Scores ranged from 0 to 24, with higher scores indicating more severe disability. Secondary endpoints included pain intensity, quality of life, sleep quality, and recommendation of intervention, and these were rated using various standardized tools.
Those randomized to the therapeutic aquatic exercise group had about an hour of therapy, beginning with a 10-minute active warm-up session to enhance neuromuscular activation, then an exercise session for 40 minutes followed by a 10-minute cooldown.
The physical therapy group received transcutaneous electrical nerve stimulation and infrared ray thermal therapy, also for 60 minutes. Both groups received these interventions twice a week for 3 months.
The overall mean age of the cohort was 31.0 years, and they were almost evenly divided by gender; 54 were men (47.8%), and 59 were women (52.2%).
As compared with the physical therapy group, individuals participating in therapeutic aquatic exercise group showed improvement in disability by an additional −1.77 points (95% confidence interval, −3.02 to −0.51) at the end of the 3-month intervention; at 6 months it was −2.42 points (95% CI, −4.13 to −0.70) and −3.61 points (95% CI, −5.63 to −1.58) at the 12-month follow-up (P < .001 for overall group x time interaction).
Functional improvement did not appear to be significantly affected by confounders that included age, sex, body mass index, low back pain duration, educational level, or pain level.
For secondary outcomes, those in the therapeutic aquatic exercise group demonstrated improvement in the most severe pain by an additional −0.79 points (95% CI, −1.31 to −0.27) after the 3-month intervention, −1.34 points (95% CI, −2.06 to −0.62) at 6 months, and −2.04 points (95% CI, −2.75 to −1.34) at the 12-month follow-up (P < .001 for overall group x time interaction), as compared with the physical therapy group. All pain scores differed significantly between the two groups at every time point.
In addition, individuals in the therapeutic aquatic exercise group showed more improvements on the 36-item Short-form Health Survey (overall group x time interaction, P = .003), Pittsburgh Sleep Quality Index (overall group x time interaction, P = .02), Tampa Scale for Kinesiophobia (overall group x time interaction, P < .001), and Fear-Avoidance Beliefs Questionnaire (physical activity subscale overall group x time interaction, P = .04), as compared with the physical therapy group. These improvements were also not influenced by confounders.
Finally, at the 12-month follow-up point, those in the aquatic therapy group had significantly greater improvements in the number of participants who met the minimal clinically important difference in pain (at least a 2-point improvement on the numeric rating scale).
More outside experts’ takes
“The current research evidence does suggest indeed that aquatic exercise therapy is suitable and often better than land exercise, passive relaxation, or other treatments for many people with low back pain,” commented Stelios Psycharakis PhD, senior lecturer in biomechanics, Institute for Sport, Physical Education and Health Sciences, University of Edinburgh.
He also noted that since low back pain is an issue affecting about 80% of all people at some stage of their life, it is “improbable that one could identify a single type of treatment or exercise therapy that would be suitable for every person with this problem.”
Dr. Psycharakis pointed out that there are also some contraindications for aquatic therapy, such as incontinence and skin conditions. “Other than that though, clinicians should definitely consider aquatic exercise therapy when advising people with chronic low back pain,” he said.
Justin M. Lantz, DPT, agreed that the study showed therapeutic aquatic exercise appears to be safe and beneficial in some patients with chronic low back pain, but he also shared limitations of the new research.
“The study has notable limitations as it did not include patients above 65 years old, pain levels were generally low for the subjects involved, and it did not include a treatment group with land therapeutic exercise – so it is difficult to determine if the beneficial effects reported were due to active exercise or because the exercises were performed in water,” said Dr. Lantz, director of the spine physical therapy fellowship program at the University of Southern California, Los Angeles, and an assistant professor of clinical physical therapy.
He also pointed out that, since active exercise has been shown to be beneficial and is advocated in multiple clinical practice guidelines for chronic low back pain, “it would be helpful to determine if the true effects on pain and disability were due to the water environment or the effect of active exercise itself.”
“Due to the significant positive long-term effects and limited adverse events reported, I believe this study supports the use of therapeutic aquatic exercise in select patient populations with chronic low back pain and should be considered as a part of a rehabilitation treatment plan if accessibility is feasible,” Dr. Lantz said.
The authors of the paper, Dr. Girgis, and Dr. Psycharakis had no conflicts of interest. Justin Lantz is a physical therapy consultant to SI-Bone.
FROM JAMA NETWORK OPEN
Multimodal Pain Management With Adductor Canal Block Decreases Opioid Consumption Following Total Knee Arthroplasty
Ease of access to opioids in the perioperative period is a risk factor for opioid misuse and has been identified as a strong risk factor for heroin use.1,2 Three-quarters of today’s heroin users were introduced to opioids through prescription medications.2 The United States accounts for about 80% of the global opioid supply consumption, and deaths from opioid overdose are increasing: 70,630 deaths in 2019 alone.3,4
The Centers for Disease Control and Prevention (CDC) has called for changes in opioid prescribing. The American Academy of Orthopaedic Surgeons (AAOS) also has published an information statement with strategies to decrease opioid misuse and abuse.5,6 Arthroplasty surgeons have recently focused on decreasing use of opioids in total knee arthroplasty (TKA), a procedure traditionally associated with high levels of opioid consumption and historical reliance on opioid monotherapy for postoperative analgesia.7,8 From a clinical perspective, prolonged postoperative opioid use contributes to poorer surgical outcomes due to increased risk of complications, including stiffness, infection, and revision TKA.9
Multimodal pain regimens are increasingly being used to control postoperative pain as data supports their efficacy.10,11 Previous studies have found that simultaneous modulation of multiple pain pathways decreases narcotics consumption and improves patient outcomes.12,13 Along with other adjuvant therapies, peripheral nerve blocks, such as adductor canal block (ACB) and femoral nerve block (FNB), have been used to decrease postoperative pain.14 Studies have shown that ACB has fewer complications and shorter functional recovery times compared with FNB.15,16 The distribution of the ACB excludes the femoral nerve, thus preserving greater quadriceps strength while providing equivalent levels of analgesia compared with FNB.15,17,18 The ACB has shown decreased near-fall events and improved balance scores in the immediate postoperative period.19
Our study analyzed opioid consumption patterns of TKA patients from a US Department of Veterans Affairs (VA) medical center before and after the institution of a multimodal analgesic protocol using ACB. The primary purpose of this study was to determine whether a protocol that included intraoperative spinal anesthesia with a postoperative multimodal analgesic regimen and ACB was associated with a decreased postoperative opioid requirement when compared with patients who received intraoperative general anesthesia and a traditional opioid regimen. Secondary outcomes included the effect of opioid consumption on range of motion on postoperative day (POD) 1 and number of opioid prescriptions written at the first postoperative clinic visit.
Methods
Approval for the study was obtained from the institutional review board at the Dayton Veterans Affairs Medical Center (DVAMC) in Ohio. A retrospective chart review was performed to collect data from all patients undergoing TKA at DVAMC from June 1, 2011, through December 31, 2015. Exclusion criteria included multiple surgeries in the study time frame, documented chronic pain, allergy to local anesthetics, daily preoperative use of opioids, and incomplete data in the health record.
All surgeries were performed by 2 staff arthroplasty surgeons at a single VAMC. All patients attended a preoperative visit where a history, physical, and anesthesia evaluation were performed, and watched an educational video detailing surgical indications and postoperative rehabilitation. All surgeries were performed with tourniquets and a periarticular injection was performed at the conclusion of each case. Surgeon 1 treatment of choice was 10 mL 0.5% bupivacaine, whereas surgeon 2 performed a posterior capsular injection of 30 mL 0.25% bupivacaine and a periarticular injection of 30 mg ketorolac in 10 mL 0.25% bupivacaine with epinephrine.
Prior to August 2014, general endotracheal anesthesia was used intraoperatively. A patient-controlled analgesia (PCA) pump of morphine or hydromorphone and additional oral oxycodone or hydrocodone was used for postoperative pain. PCA pumps were patient dependent. In the control group, 245 patients received the morphine PCA while 61 received the hydromorphone PCA. Morphine PCA dosing consisted of 1-mg doses every 10 minutes with potential baseline infusion rates of 0.5 to 1.0 mg/h and a 4-hour limit of 20 mg. Hydromorphone PCA dosing consisted of 0.2 to 0.4-mg doses with a potential continuous dose of 0.2 to 0.4 mg/h and a 4-hour limit of 4 mg.
In August 2014, a new analgesic protocol was adopted for TKA consisting of intraoperative spinal anesthesia (0.75% bupivacaine) with IV sedation (propofol), a postoperative multimodal analgesic regimen, an ACB performed in the postanesthesia care unit (PACU), and opioids as needed (protocol group). The ACB catheter was a 0.5% ropivo caine hydrochloride injection. It was attached to a local anesthetic fixed flow rate pump that administers 0.5% ropivacaine without epinephrine at 8 mL/h and was removed on POD 5 by the patient. The multimodal medication regimen included IV ketorolac 15 mg every 6 hours for 3 doses, gabapentin 300 mg every 8 hours, acetaminophen 975 mg every 8 hours, meloxicam 7.5 mg daily, tramadol 50 mg every 6 hours, oxycodone 5 mg 1 to 2 tabs every 4 hours as needed, and IV hydromorphone 0.5 mg every 4 hours as needed for breakthrough pain.
Preoperative demographic characteristics were collected (Table 1). Data on all IV and oral opioid requirements were collected for both groups, converted to morphine milligram equivalents (MME), and a total morphine equivalent dose (MED) was calculated.20,21
In April 2015, a separate protocol change occurred at the DVAMC with the goal of discharge on POD 1. To standardize outcomes before and after this change, data collection regarding opioid requirements was concluded at midnight on POD 1. If a patient was discharged before midnight on POD 1, opioid requirement through the time of discharge was collected. All surgeries were performed in the morning to early afternoon; however, specific surgical times were not collected. Patients were also evaluated by a physical therapist on POD 0, and maximal knee flexion and extension were measured on POD 1. Patients were discharged with prescriptions for oxycodone/acetaminophen and tramadol and were seen 3 weeks later for their first postoperative visit. Opioid refills at the first postoperative visit were recorded. All statistical analyses were performed in SAS 9.4 with significance set to α = 0.05. Between-groups differences in preoperative and perioperative characteristics as well as postoperative outcomes were analyzed using independent samples t tests for continuous variables and Fisher exact tests for dichotomous discrete variables. Where groups differed for a pre- or perioperative variable, linear mixed models analysis was used to determine whether IV, oral, and total MEDs were significantly affected by the interaction between the pre- or perioperative variable with analgesia group. For refills at the postoperative visit, the effects of pre- or perioperative differences were tested using χ2 tests. Effect sizes for outcome variables were estimated using Cohen d and probability of superiority (Δ) for continuous variables, and relative risk (RR) in the case of discrete variables.22
Results
During the study period from June 1, 2011, through December 31, 2015, 533 eligible TKAs were performed, 306 in the control group and 227 in the protocol group. The groups had similar sex distribution; body mass index; knee range of motion; diagnoses of diabetes mellitus, coronary artery disease, and chronic kidney disease; and history of deep vein thrombosis (DVT) or pulmonary embolism (P ≥ .05). The protocol group was significantly older (P = .04) and had a significantly higher rate of chronic obstructive pulmonary disease (COPD) (P = .002). There were no significant differences between number of procedures performed by surgeon (P = .48) or total tourniquet time (P = .13) (Table 2). Mean (SD) length of stay was significantly greater in the control group compared with the protocol group (2.5 [1.3] vs 1.4 [0.7] days, P < .001).
Figure 1 shows the distributions of each type of opioid used. Compared with the control group, the protocol group had a significantly lower mean (SD) IV opioid use: 178.2 (98.0) MED vs 12.0 (24.6) MED (P < .001; d = 2.19; Δ = 0.94) and mean (SD) total opioid use: 241.7 (120.1) MED vs 74.8 (42.7) MED (P < .001; d = 1.76; Δ = 0.89). Mean (SD) oral opioid use did not differ between groups (control, 63.6 [45.4] MED; protocol, 62.9 [31.4] MED; P = .85; d = 0.02; Δ = 0.51). A significantly lower percentage of patients in the protocol group received additional opioids at the 3-week follow-up when compared to the control group: 46.7% vs 61.3%, respectively (P < .001; RR, 0.76; 95% CI, 0.65-0.90).
There were no significant differences in postoperative mean (SD) maximum knee flexion (control, 67.2 [15.7]°; protocol, 67.8 [19.2]°; P = .72; d = 0.03; Δ = 0.51) or mean (SD) total flexion/extension arc (control, 66.2 [15.9]°; protocol, 67.9 [19.4]°; P = .32; d = 0.10; Δ = 0.53). Mean (SD) postoperative maximum knee extension was significantly higher in the protocol group compared with the control group (-0.1 [2.1]° vs 1.0 [3.7]°; P < .001; d = 0.35; Δ = 0.60). More patients in the protocol group (92.5%) were discharged to home compared with the control group (86.6%) (P = .02; RR, 1.07; 95% CI, 1.01-1.13).
Because age and rates of COPD differed between groups, sensitivity analyses were conducted to determine whether these variables influenced postoperative opioid use. The relationship between age and group was significant for IV (P < .001) and total opioid use (P < .001). Younger patients received higher MED doses than older patients within the control group, while dosages were fairly consistent regardless of age in the protocol group (Figure 2). There was no significance in age interaction effect with regard to oral opioids (P = .83) nor opioid refills at 3-week follow-up (P = .24).
The sensitivity analysis for COPD found that a diagnosis of COPD did not significantly influence utilization of IV opioids (P = .10), or total opioids (P = .68). There was a significant interaction effect for oral opioids (Figure 3). Patients in the control group with COPD required significantly higher mean (SD) oral opioids than patients without COPD (91.5 [123.9] MED and 62.0 [36.0] MED, respectively; P = .03). In the control group, the χ2 test was significant regarding opioid prescription refills at the 3-week visit (P = .004) with 62.4% of patients with COPD requiring refills vs 44.4% without COPD (P = .004). There was no difference in refills in the protocol group (46.4% vs 48.4%).
Finally, 2-sided independent samples t test evaluated total MED use between the 2 surgeons. There was no difference in total MED per patient for the surgeons. In the control group, mean (SD) total MED for surgeon 1 was 232.9 (118.7) MED vs 252.8 (121.5) MED for surgeon 2 (P = .18). In the protocol group, the mean (SD) total MED was 72.5 (43.2) and 77.4 (42.1) for surgeon 1 and surgeon 2, respectively (P = .39).
Discussion
Coordinated efforts with major medical organizations are being made to decrease opioid prescriptions and exposure.5,6 To our knowledge, no study has quantified a decrease in opioid requirement in a VA population after implementation of a protocol that includes intraoperative spinal anesthesia and a postoperative multimodal analgesic regimen including ACB after TKA. The analgesic protocol described in this study aligns with recommendations from both the CDC and the AAOS to decrease opioid use and misuse by maximizing nonopioid medications and limiting the size and number of opioid prescriptions. However, public and medical opinion of opioids as well as prescribing practices have changed over time with a trend toward lower opioid use. The interventions, as part of the described protocol, are a result of these changes and attempt to minimize opioid use while maximizing postoperative analgesia.
Our data showed a significant decrease in total opioid use through POD 1, IV opioid use, and opioid prescriptions provided at the first postoperative visit. The protocol group used only 6.7% of the IV opioids and 30.9% of the total opioids that were used by the control group. The substantial difference in IV opioid requirement, 166.2 MED, is equivalent to 8 mg of IV hydromorphone or 55 mg of IV morphine. The difference in total opioid requirement was similar at 166.9 MED, equivalent to 111 mg of oral oxycodone.
Decreasing opioid use has the additional benefit of improving outcomes, as higher doses of opioids have been associated with increased length of stay, greater rates of DVT, and postoperative infection.23 These complications occurred in a stepwise manner, suggesting a dose-response gradient that makes the sizable decrease noted in our data of greater relevance.23 While the adverse effects (AEs) of opioids are well known, there are limited data on opioid dosing and its effect on perioperative outcomes.23
A significant decrease in the percentage of patients receiving an opioid prescription at the first postoperative visit suggests a decrease in the number of patients on prolonged opioids after TKA with implementation of modern analgesic modalities. The duration of postoperative opioid use has been found to be the strongest predictor of misuse, and each postoperative refill increases the probability of misuse by 44%.24 In addition, opioid use for > 3 months after TKA is associated with increased risk of periprosthetic infection, increased overall revision rate, and stiffness at 1 year postoperatively.9 While not entirely under the control of the surgeon, measures to decrease the number of postoperative opioid refills may lead to a decrease in opioid misuse.
In the control group, older patients tended to receive less opioids. This is likely due to physiologic changes in opioid metabolism associated with aging, including decreased renal and hepatic opioid metabolism and alterations in overall body composition that increase relative potency and duration of action of opioids in a geriatric population.25,26 No difference in opioid use by age was found for the protocol group.
Patients in the protocol group demonstrated significantly greater maximal knee extension on POD 1 compared with the control group. No difference in maximal flexion was found. This difference in extension may partially be explained by the use of an ACB. One benefit of ACB is greater quadriceps strength and fewer near-fall events when compared with FNB.15,19
Our results corroborate the findings of similar studies. A randomized controlled trial comparing a multimodal analgesic regimen with a periarticular injection without a postoperative ACB to a hydromorphone PCA revealed a significant decrease in opioid use in the multimodal analgesic group.27 Along with lower opioid requirements, the multimodal analgesic group had lower visual analog scale pain scores, fewer AEs, faster progression to physical therapy milestones, and higher satisfaction.27 Recent guidelines from the French Society of Anaesthesia and Intensive Care Medicine recommend against the use of gabapentin as a method of postoperative pain control. However, this specifically refers to the preoperative administration of gabapentin. This same set of guidelines later cites a high level of evidence suggesting patients undergoing arthroplasty benefit more from gabapentinoids.28 Multiple analgesic protocols that include gabapentin as a part of a multimodal approach have been shown to have positive results.13,29
In our study, patients receiving the multimodal analgesic regimen were significantly more likely to be discharged home rather than to postacute care facilities, which have been associated with increased rates of major complications, 30-day readmission, and 30-day reoperation.30,31 In addition, discharge to an inpatient rehabilitation or skilled nursing facility has not been found to result in higher functional outcomes, despite $3.2 billion spent yearly on rehabilitation services after primary TKA.32,33
A component of our described analgesic protocol included spinal anesthesia intraoperatively. The differences between groups regarding anesthesia type can be attributed to this protocol change. A significantly greater percentage of patients in the protocol group received spinal anesthesia, while more patients in the control group received general anesthesia. While patients who received spinal anesthesia may have enhanced analgesia in the immediate postoperative period, no differences in opioid outcomes were seen based on anesthesia type. Known benefits of intraoperative spinal anesthesia include decreased perioperative blood loss and a smaller decrease in hemoglobin postoperatively, as well as lower rates of in-hospital complications, including pulmonary embolism, pneumonia, cerebrovascular events, and acute renal failure.34
Limitations
A number of limitations of this study should be noted. One was a protocol change regarding length of stay, which occurred during the study period and resulted in a significantly shorter length of stay in the protocol group. As a result, opioid use data were analyzed only through midnight at the end of POD 1. Patients who were discharged on POD 1 did not have opioid use data available for the full duration of the first POD, which may exaggerate the decrease in opioid requirements, as opioids used after discharge but prior to midnight on POD 1 were not recorded. However, opioids taken at home are oral with a low MME compared with IV opioids received by hospitalized patients in the control group. In addition, if taken as prescribed, patients at home would only have enough time to take a few doses of opioids prior to the midnight cutoff. We do not believe this difference in time of opioid use meaningfully affected the data. An additional limitation includes the variability between periarticular injections between surgeons. While the percentage of patients that received injections from surgeon 1 vs surgeon 2 were similar, it cannot be ruled out as a potential confounding factor. Other limitations include a lack of pain scores to compare subjective pain ratings, the retrospective nature of the study, and a largely homogenous male VA population.
Conclusions
Ease of access to opioids is a risk factor for opioid abuse, which itself is a risk factor for subsequent heroin use.1,2 The CDC and AAOS have thus published recommendations regarding opioid prescribing practices to decrease opioid use and abuse.5,6 Our described protocol, which aligns with these recommendations, resulted in a significant decrease in IV opioid requirement, total opioid requirement, and lower rates of opioid prescriptions provided at the first postoperative visit. These promising findings demonstrate a lower percentage of patients on long-term opioids after TKA and a significantly decreased cumulative opioid exposure.
1. Lankenau SE, Teti M, Silva K, Jackson Bloom J, Harocopos A, Treese M. Initiation into prescription opioid misuse amongst young injection drug users. Int J Drug Policy. 2012;23(1):37-44. doi:10.1016/j.drugpo.2011.05.014
2. Jones CM. Heroin use and heroin use risk behaviors among nonmedical users of prescription opioid pain relievers - United States, 2002-2004 and 2008-2010. Drug Alcohol Depend. 2013;132(1-2):95-100. doi:10.1016/j.drugalcdep.2013.01.007
3. Manchikanti L, Singh A. Therapeutic opioids: a ten-year perspective on the complexities and complications of the escalating use, abuse, and nonmedical use of opioids. Pain Physician. 2008;11(suppl 2):S63-S88.
4. Seth P, Scholl L, Rudd RA, Bacon S. Overdose deaths involving opioids, cocaine, and psychostimulants - United States, 2015-2016. MMWR Morb Mortal Wkly Rep. 2018;67(12):349-358. Published 2018 Mar 30. doi:10.15585/mmwr.mm6712a1
5. Dowell D, Haegerich TM, Chou R. CDC Guideline for Prescribing Opioids for Chronic Pain-United States, 2016. JAMA. 2016;315(15):1624-1645. doi:10.1001/jama.2016.1464
6. American Academy of Orthopaedic Surgeons. Information statement: opioid use, misuse, and abuse in orthopaedic practice. Published October 2015. Accessed November 12, 2021. https://aaos.org/globalassets/about /bylaws-library/information-statements/1045-opioid-use -misuse-and-abuse-in-practice.pdf
7. Hernandez NM, Parry JA, Taunton MJ. Patients at risk: large opioid prescriptions after total knee arthroplasty. J Arthroplasty. 2017;32(8):2395-2398. doi:10.1016/j.arth.2017.02.060
8. Gerner P, Poeran J, Cozowicz C, Mörwald EE, Zubizarreta N, Mazumdar M, Memtsoudis SG, Multimodal pain management in total hip and knee arthroplasty: trends over the last 10 years. Abstract presented at: American Society of Anesthesiologists Annual Meeting; October 21, 2017; Boston, MA.
9. Cancienne JM, Patel KJ, Browne JA, Werner BC. Narcotic use and total knee arthroplasty. J Arthroplasty. 2018;33(1):113-118. doi:10.1016/j.arth.2017.08.006
10. Moucha CS, Weiser MC, Levin EJ. Current strategies in anesthesia and analgesia for total knee arthroplasty. J Am Acad Orthop Surg. 2016;24(2):60-73. doi:10.5435/JAAOS-D-14-00259
11. Wick EC, Grant MC, Wu CL. Postoperative multimodal analgesia pain management with nonopioid analgesics and techniques: a review. JAMA Surg. 2017;152(7):691-697.doi:10.1001/jamasurg.2017.0898
12. Lamplot JD, Wagner ER, Manning DW. Multimodal pain management in total knee arthroplasty: a prospective randomized controlled trial. J Arthoplasty. 2014;29(2):329-334. doi:10.1016/j.arth.2013.06.005
13. Golladay GJ, Balch KR, Dalury DF, Satpathy J, Jiranek WA. Oral multimodal analgesia for total joint arthroplasty. J Arthroplasty. 2017;32(9S):S69-S73. doi:10.1016/j.arth.2017.05.002
14. Ardon AE, Clendenen SR, Porter SB, Robards CB, Greengrass RA. Opioid consumption in total knee arthroplasty patients: a retrospective comparison of adductor canal and femoral nerve continuous infusions in the presence of a sciatic nerve catheter. J Clin Anesth. 2016;31:19-26. doi:10.1016/j.jclinane.2015.12.014
15. Li D, Ma GG. Analgesic efficacy and quadriceps strength of adductor canal block versus femoral nerve block following total knee arthroplasty. Knee Surg Sports Traumatol Arthrosc. 2016;24(8):2614-2619. doi:10.1007/s00167-015-3874-3
16. Li D, Yang Z, Xie X, Zhao J, Kang P. Adductor canal block provides better performance after total knee arthroplasty compared with femoral nerve block: a systematic review and meta-analysis. Int Orthop. 2016;40(5):925-933. doi:10.1007/s00264-015-2998-x
17. Horner G, Dellon AL. Innervation of the human knee joint and implications for surgery. Clin Orthop Relat Res. 1994;(301):221-226.
18. Kim DH, Lin Y, Goytizolo EA, et al. Adductor canal block versus femoral nerve block for total knee arthroplasty: a prospective, randomized, controlled trial. Anesthesiology. 2014;120(3):540-550. doi:10.1097/ALN.0000000000000119
19. Thacher RR, Hickernell TR, Grosso MJ, et al. Decreased risk of knee buckling with adductor canal block versus femoral nerve block in total knee arthroplasty: a retrospective cohort study. Arthroplasty Today. 2017;3(4):281-285. Published 2017 Apr 15. doi:10.1016/j.artd.2017.02.008
20. Von Korff M, Saunders K, Thomas Ray G, et al. De facto long-term opioid therapy for noncancer pain [published correction appears in Clin J Pain. 2014 Sep;30(9):830. Korff, Michael Von [corrected to Von Korff, Michael]]. Clin J Pain. 2008;24(6):521-527. doi:10.1097/AJP.0b013e318169d03b
21. Kishner S. Opioid equivalents and conversions: overview. Published January 29, 2018. Accessed November 12, 2021. https://emedicine.medscape.com/article/2138678 -overview#a1
22. Ruscio J, Mullen T. Confidence intervals for the probability of superiority effect size measure and the area under a receiver operating characteristic curve. Multivariate Behav Res. 2012;47(2):201-223. doi:10.1080/00273171.2012.658329
23. Cozowicz C, Olson A, Poeran J, et al. Opioid prescription levels and postoperative outcomes in orthopedic orthopedic surgery. Pain. 2017;158(12):2422-2430. doi:10.1097/j.pain.0000000000001047
24. Brat GA, Agniel D, Beam A, et al. Postsurgical prescriptions for opioid naive patients and association with overdose and misuse: retrospective cohort study. BMJ. 2018;360:j5790. Published 2018 Jan 17. doi:10.1136/bmj.j5790
25. Tegeder I, Lötsch J, Geisslinger G. Pharmacokinetics of opioids in liver disease. Clin Pharmacokinet. 1999;37(1):17- 40. doi:10.2165/00003088-199937010-00002
26. Linnebur SA, O’Connell MB, Wessell AM, et al. Pharmacy practice, research, education, and advocacy for older adults. Pharmacotherapy. 2005;25(10):1396-1430. doi:10.1592/phco.2005.25.10.1396
27. Lamplot JD, Wagner ER, Manning DW. Multimodal pain management in total knee arthroplasty: a prospective randomized controlled trial. J Arthroplasty. 2014;29(2):329- 334. doi:10.1016/j.arth.2013.06.005
28. Aubrun F, Nouette-Gaulain K, Fletcher D, et al. Revision of expert panel’s guidelines on postoperative pain management. Anaesth Crit Care Pain Med. 2019;38(4):405-411. doi:10.1016/j.accpm.2019.02.011
29. Han C, Li XD, Jiang HQ, Ma JX, Ma XL. The use of gabapentin in the management of postoperative pain after total knee arthroplasty: A PRISMA-compliant metaanalysis of randomized controlled trials [published correction appears in Medicine (Baltimore). 2016 Jul 18;95(28):e0916]. Medicine (Baltimore). 2016;95(23):e3883. doi:10.1097/MD.0000000000003883
30. McLawhorn AS, Fu MC, Schairer WW, Sculco PK, MacLean CH, Padgett DE. Continued inpatient care after primary total knee arthroplasty increases 30-day postdischarge complications: a propensity score-adjusted analysis. J Arthroplasty. 2017;32(9S):S113-S118. doi:10.1016/j.arth.2017.01.039
31. Pelt CE, Gililland JM, Erickson JA, Trimble DE, Anderson MB, Peters CL. Improving value in total joint arthroplasty: a comprehensive patient education and management program decreases discharge to post-acute care facilities and post-operative complications. J Arthroplasty. 2018;33(1):14-18. doi:10.1016/j.arth.2017.08.003
32. Padgett DE, Christ AB, Joseph AD, Lee YY, Haas SB, Lyman S. Discharge to inpatient rehab does not result in improved functional outcomes following primary total knee arthroplasty. J Arthroplasty. 2018;33(6):1663-1667. doi:10.1016/j.arth.2017.12.033
33. Lavernia CJ, D’Apuzzo MR, Hernandez VH, Lee DJ, Rossi MD. Postdischarge costs in arthroplasty surgery. J Arthroplasty. 2006;21(6 Suppl 2):144-150. doi:10.1016/j.arth.2006.05.003
Ease of access to opioids in the perioperative period is a risk factor for opioid misuse and has been identified as a strong risk factor for heroin use.1,2 Three-quarters of today’s heroin users were introduced to opioids through prescription medications.2 The United States accounts for about 80% of the global opioid supply consumption, and deaths from opioid overdose are increasing: 70,630 deaths in 2019 alone.3,4
The Centers for Disease Control and Prevention (CDC) has called for changes in opioid prescribing. The American Academy of Orthopaedic Surgeons (AAOS) also has published an information statement with strategies to decrease opioid misuse and abuse.5,6 Arthroplasty surgeons have recently focused on decreasing use of opioids in total knee arthroplasty (TKA), a procedure traditionally associated with high levels of opioid consumption and historical reliance on opioid monotherapy for postoperative analgesia.7,8 From a clinical perspective, prolonged postoperative opioid use contributes to poorer surgical outcomes due to increased risk of complications, including stiffness, infection, and revision TKA.9
Multimodal pain regimens are increasingly being used to control postoperative pain as data supports their efficacy.10,11 Previous studies have found that simultaneous modulation of multiple pain pathways decreases narcotics consumption and improves patient outcomes.12,13 Along with other adjuvant therapies, peripheral nerve blocks, such as adductor canal block (ACB) and femoral nerve block (FNB), have been used to decrease postoperative pain.14 Studies have shown that ACB has fewer complications and shorter functional recovery times compared with FNB.15,16 The distribution of the ACB excludes the femoral nerve, thus preserving greater quadriceps strength while providing equivalent levels of analgesia compared with FNB.15,17,18 The ACB has shown decreased near-fall events and improved balance scores in the immediate postoperative period.19
Our study analyzed opioid consumption patterns of TKA patients from a US Department of Veterans Affairs (VA) medical center before and after the institution of a multimodal analgesic protocol using ACB. The primary purpose of this study was to determine whether a protocol that included intraoperative spinal anesthesia with a postoperative multimodal analgesic regimen and ACB was associated with a decreased postoperative opioid requirement when compared with patients who received intraoperative general anesthesia and a traditional opioid regimen. Secondary outcomes included the effect of opioid consumption on range of motion on postoperative day (POD) 1 and number of opioid prescriptions written at the first postoperative clinic visit.
Methods
Approval for the study was obtained from the institutional review board at the Dayton Veterans Affairs Medical Center (DVAMC) in Ohio. A retrospective chart review was performed to collect data from all patients undergoing TKA at DVAMC from June 1, 2011, through December 31, 2015. Exclusion criteria included multiple surgeries in the study time frame, documented chronic pain, allergy to local anesthetics, daily preoperative use of opioids, and incomplete data in the health record.
All surgeries were performed by 2 staff arthroplasty surgeons at a single VAMC. All patients attended a preoperative visit where a history, physical, and anesthesia evaluation were performed, and watched an educational video detailing surgical indications and postoperative rehabilitation. All surgeries were performed with tourniquets and a periarticular injection was performed at the conclusion of each case. Surgeon 1 treatment of choice was 10 mL 0.5% bupivacaine, whereas surgeon 2 performed a posterior capsular injection of 30 mL 0.25% bupivacaine and a periarticular injection of 30 mg ketorolac in 10 mL 0.25% bupivacaine with epinephrine.
Prior to August 2014, general endotracheal anesthesia was used intraoperatively. A patient-controlled analgesia (PCA) pump of morphine or hydromorphone and additional oral oxycodone or hydrocodone was used for postoperative pain. PCA pumps were patient dependent. In the control group, 245 patients received the morphine PCA while 61 received the hydromorphone PCA. Morphine PCA dosing consisted of 1-mg doses every 10 minutes with potential baseline infusion rates of 0.5 to 1.0 mg/h and a 4-hour limit of 20 mg. Hydromorphone PCA dosing consisted of 0.2 to 0.4-mg doses with a potential continuous dose of 0.2 to 0.4 mg/h and a 4-hour limit of 4 mg.
In August 2014, a new analgesic protocol was adopted for TKA consisting of intraoperative spinal anesthesia (0.75% bupivacaine) with IV sedation (propofol), a postoperative multimodal analgesic regimen, an ACB performed in the postanesthesia care unit (PACU), and opioids as needed (protocol group). The ACB catheter was a 0.5% ropivo caine hydrochloride injection. It was attached to a local anesthetic fixed flow rate pump that administers 0.5% ropivacaine without epinephrine at 8 mL/h and was removed on POD 5 by the patient. The multimodal medication regimen included IV ketorolac 15 mg every 6 hours for 3 doses, gabapentin 300 mg every 8 hours, acetaminophen 975 mg every 8 hours, meloxicam 7.5 mg daily, tramadol 50 mg every 6 hours, oxycodone 5 mg 1 to 2 tabs every 4 hours as needed, and IV hydromorphone 0.5 mg every 4 hours as needed for breakthrough pain.
Preoperative demographic characteristics were collected (Table 1). Data on all IV and oral opioid requirements were collected for both groups, converted to morphine milligram equivalents (MME), and a total morphine equivalent dose (MED) was calculated.20,21
In April 2015, a separate protocol change occurred at the DVAMC with the goal of discharge on POD 1. To standardize outcomes before and after this change, data collection regarding opioid requirements was concluded at midnight on POD 1. If a patient was discharged before midnight on POD 1, opioid requirement through the time of discharge was collected. All surgeries were performed in the morning to early afternoon; however, specific surgical times were not collected. Patients were also evaluated by a physical therapist on POD 0, and maximal knee flexion and extension were measured on POD 1. Patients were discharged with prescriptions for oxycodone/acetaminophen and tramadol and were seen 3 weeks later for their first postoperative visit. Opioid refills at the first postoperative visit were recorded. All statistical analyses were performed in SAS 9.4 with significance set to α = 0.05. Between-groups differences in preoperative and perioperative characteristics as well as postoperative outcomes were analyzed using independent samples t tests for continuous variables and Fisher exact tests for dichotomous discrete variables. Where groups differed for a pre- or perioperative variable, linear mixed models analysis was used to determine whether IV, oral, and total MEDs were significantly affected by the interaction between the pre- or perioperative variable with analgesia group. For refills at the postoperative visit, the effects of pre- or perioperative differences were tested using χ2 tests. Effect sizes for outcome variables were estimated using Cohen d and probability of superiority (Δ) for continuous variables, and relative risk (RR) in the case of discrete variables.22
Results
During the study period from June 1, 2011, through December 31, 2015, 533 eligible TKAs were performed, 306 in the control group and 227 in the protocol group. The groups had similar sex distribution; body mass index; knee range of motion; diagnoses of diabetes mellitus, coronary artery disease, and chronic kidney disease; and history of deep vein thrombosis (DVT) or pulmonary embolism (P ≥ .05). The protocol group was significantly older (P = .04) and had a significantly higher rate of chronic obstructive pulmonary disease (COPD) (P = .002). There were no significant differences between number of procedures performed by surgeon (P = .48) or total tourniquet time (P = .13) (Table 2). Mean (SD) length of stay was significantly greater in the control group compared with the protocol group (2.5 [1.3] vs 1.4 [0.7] days, P < .001).
Figure 1 shows the distributions of each type of opioid used. Compared with the control group, the protocol group had a significantly lower mean (SD) IV opioid use: 178.2 (98.0) MED vs 12.0 (24.6) MED (P < .001; d = 2.19; Δ = 0.94) and mean (SD) total opioid use: 241.7 (120.1) MED vs 74.8 (42.7) MED (P < .001; d = 1.76; Δ = 0.89). Mean (SD) oral opioid use did not differ between groups (control, 63.6 [45.4] MED; protocol, 62.9 [31.4] MED; P = .85; d = 0.02; Δ = 0.51). A significantly lower percentage of patients in the protocol group received additional opioids at the 3-week follow-up when compared to the control group: 46.7% vs 61.3%, respectively (P < .001; RR, 0.76; 95% CI, 0.65-0.90).
There were no significant differences in postoperative mean (SD) maximum knee flexion (control, 67.2 [15.7]°; protocol, 67.8 [19.2]°; P = .72; d = 0.03; Δ = 0.51) or mean (SD) total flexion/extension arc (control, 66.2 [15.9]°; protocol, 67.9 [19.4]°; P = .32; d = 0.10; Δ = 0.53). Mean (SD) postoperative maximum knee extension was significantly higher in the protocol group compared with the control group (-0.1 [2.1]° vs 1.0 [3.7]°; P < .001; d = 0.35; Δ = 0.60). More patients in the protocol group (92.5%) were discharged to home compared with the control group (86.6%) (P = .02; RR, 1.07; 95% CI, 1.01-1.13).
Because age and rates of COPD differed between groups, sensitivity analyses were conducted to determine whether these variables influenced postoperative opioid use. The relationship between age and group was significant for IV (P < .001) and total opioid use (P < .001). Younger patients received higher MED doses than older patients within the control group, while dosages were fairly consistent regardless of age in the protocol group (Figure 2). There was no significance in age interaction effect with regard to oral opioids (P = .83) nor opioid refills at 3-week follow-up (P = .24).
The sensitivity analysis for COPD found that a diagnosis of COPD did not significantly influence utilization of IV opioids (P = .10), or total opioids (P = .68). There was a significant interaction effect for oral opioids (Figure 3). Patients in the control group with COPD required significantly higher mean (SD) oral opioids than patients without COPD (91.5 [123.9] MED and 62.0 [36.0] MED, respectively; P = .03). In the control group, the χ2 test was significant regarding opioid prescription refills at the 3-week visit (P = .004) with 62.4% of patients with COPD requiring refills vs 44.4% without COPD (P = .004). There was no difference in refills in the protocol group (46.4% vs 48.4%).
Finally, 2-sided independent samples t test evaluated total MED use between the 2 surgeons. There was no difference in total MED per patient for the surgeons. In the control group, mean (SD) total MED for surgeon 1 was 232.9 (118.7) MED vs 252.8 (121.5) MED for surgeon 2 (P = .18). In the protocol group, the mean (SD) total MED was 72.5 (43.2) and 77.4 (42.1) for surgeon 1 and surgeon 2, respectively (P = .39).
Discussion
Coordinated efforts with major medical organizations are being made to decrease opioid prescriptions and exposure.5,6 To our knowledge, no study has quantified a decrease in opioid requirement in a VA population after implementation of a protocol that includes intraoperative spinal anesthesia and a postoperative multimodal analgesic regimen including ACB after TKA. The analgesic protocol described in this study aligns with recommendations from both the CDC and the AAOS to decrease opioid use and misuse by maximizing nonopioid medications and limiting the size and number of opioid prescriptions. However, public and medical opinion of opioids as well as prescribing practices have changed over time with a trend toward lower opioid use. The interventions, as part of the described protocol, are a result of these changes and attempt to minimize opioid use while maximizing postoperative analgesia.
Our data showed a significant decrease in total opioid use through POD 1, IV opioid use, and opioid prescriptions provided at the first postoperative visit. The protocol group used only 6.7% of the IV opioids and 30.9% of the total opioids that were used by the control group. The substantial difference in IV opioid requirement, 166.2 MED, is equivalent to 8 mg of IV hydromorphone or 55 mg of IV morphine. The difference in total opioid requirement was similar at 166.9 MED, equivalent to 111 mg of oral oxycodone.
Decreasing opioid use has the additional benefit of improving outcomes, as higher doses of opioids have been associated with increased length of stay, greater rates of DVT, and postoperative infection.23 These complications occurred in a stepwise manner, suggesting a dose-response gradient that makes the sizable decrease noted in our data of greater relevance.23 While the adverse effects (AEs) of opioids are well known, there are limited data on opioid dosing and its effect on perioperative outcomes.23
A significant decrease in the percentage of patients receiving an opioid prescription at the first postoperative visit suggests a decrease in the number of patients on prolonged opioids after TKA with implementation of modern analgesic modalities. The duration of postoperative opioid use has been found to be the strongest predictor of misuse, and each postoperative refill increases the probability of misuse by 44%.24 In addition, opioid use for > 3 months after TKA is associated with increased risk of periprosthetic infection, increased overall revision rate, and stiffness at 1 year postoperatively.9 While not entirely under the control of the surgeon, measures to decrease the number of postoperative opioid refills may lead to a decrease in opioid misuse.
In the control group, older patients tended to receive less opioids. This is likely due to physiologic changes in opioid metabolism associated with aging, including decreased renal and hepatic opioid metabolism and alterations in overall body composition that increase relative potency and duration of action of opioids in a geriatric population.25,26 No difference in opioid use by age was found for the protocol group.
Patients in the protocol group demonstrated significantly greater maximal knee extension on POD 1 compared with the control group. No difference in maximal flexion was found. This difference in extension may partially be explained by the use of an ACB. One benefit of ACB is greater quadriceps strength and fewer near-fall events when compared with FNB.15,19
Our results corroborate the findings of similar studies. A randomized controlled trial comparing a multimodal analgesic regimen with a periarticular injection without a postoperative ACB to a hydromorphone PCA revealed a significant decrease in opioid use in the multimodal analgesic group.27 Along with lower opioid requirements, the multimodal analgesic group had lower visual analog scale pain scores, fewer AEs, faster progression to physical therapy milestones, and higher satisfaction.27 Recent guidelines from the French Society of Anaesthesia and Intensive Care Medicine recommend against the use of gabapentin as a method of postoperative pain control. However, this specifically refers to the preoperative administration of gabapentin. This same set of guidelines later cites a high level of evidence suggesting patients undergoing arthroplasty benefit more from gabapentinoids.28 Multiple analgesic protocols that include gabapentin as a part of a multimodal approach have been shown to have positive results.13,29
In our study, patients receiving the multimodal analgesic regimen were significantly more likely to be discharged home rather than to postacute care facilities, which have been associated with increased rates of major complications, 30-day readmission, and 30-day reoperation.30,31 In addition, discharge to an inpatient rehabilitation or skilled nursing facility has not been found to result in higher functional outcomes, despite $3.2 billion spent yearly on rehabilitation services after primary TKA.32,33
A component of our described analgesic protocol included spinal anesthesia intraoperatively. The differences between groups regarding anesthesia type can be attributed to this protocol change. A significantly greater percentage of patients in the protocol group received spinal anesthesia, while more patients in the control group received general anesthesia. While patients who received spinal anesthesia may have enhanced analgesia in the immediate postoperative period, no differences in opioid outcomes were seen based on anesthesia type. Known benefits of intraoperative spinal anesthesia include decreased perioperative blood loss and a smaller decrease in hemoglobin postoperatively, as well as lower rates of in-hospital complications, including pulmonary embolism, pneumonia, cerebrovascular events, and acute renal failure.34
Limitations
A number of limitations of this study should be noted. One was a protocol change regarding length of stay, which occurred during the study period and resulted in a significantly shorter length of stay in the protocol group. As a result, opioid use data were analyzed only through midnight at the end of POD 1. Patients who were discharged on POD 1 did not have opioid use data available for the full duration of the first POD, which may exaggerate the decrease in opioid requirements, as opioids used after discharge but prior to midnight on POD 1 were not recorded. However, opioids taken at home are oral with a low MME compared with IV opioids received by hospitalized patients in the control group. In addition, if taken as prescribed, patients at home would only have enough time to take a few doses of opioids prior to the midnight cutoff. We do not believe this difference in time of opioid use meaningfully affected the data. An additional limitation includes the variability between periarticular injections between surgeons. While the percentage of patients that received injections from surgeon 1 vs surgeon 2 were similar, it cannot be ruled out as a potential confounding factor. Other limitations include a lack of pain scores to compare subjective pain ratings, the retrospective nature of the study, and a largely homogenous male VA population.
Conclusions
Ease of access to opioids is a risk factor for opioid abuse, which itself is a risk factor for subsequent heroin use.1,2 The CDC and AAOS have thus published recommendations regarding opioid prescribing practices to decrease opioid use and abuse.5,6 Our described protocol, which aligns with these recommendations, resulted in a significant decrease in IV opioid requirement, total opioid requirement, and lower rates of opioid prescriptions provided at the first postoperative visit. These promising findings demonstrate a lower percentage of patients on long-term opioids after TKA and a significantly decreased cumulative opioid exposure.
Ease of access to opioids in the perioperative period is a risk factor for opioid misuse and has been identified as a strong risk factor for heroin use.1,2 Three-quarters of today’s heroin users were introduced to opioids through prescription medications.2 The United States accounts for about 80% of the global opioid supply consumption, and deaths from opioid overdose are increasing: 70,630 deaths in 2019 alone.3,4
The Centers for Disease Control and Prevention (CDC) has called for changes in opioid prescribing. The American Academy of Orthopaedic Surgeons (AAOS) also has published an information statement with strategies to decrease opioid misuse and abuse.5,6 Arthroplasty surgeons have recently focused on decreasing use of opioids in total knee arthroplasty (TKA), a procedure traditionally associated with high levels of opioid consumption and historical reliance on opioid monotherapy for postoperative analgesia.7,8 From a clinical perspective, prolonged postoperative opioid use contributes to poorer surgical outcomes due to increased risk of complications, including stiffness, infection, and revision TKA.9
Multimodal pain regimens are increasingly being used to control postoperative pain as data supports their efficacy.10,11 Previous studies have found that simultaneous modulation of multiple pain pathways decreases narcotics consumption and improves patient outcomes.12,13 Along with other adjuvant therapies, peripheral nerve blocks, such as adductor canal block (ACB) and femoral nerve block (FNB), have been used to decrease postoperative pain.14 Studies have shown that ACB has fewer complications and shorter functional recovery times compared with FNB.15,16 The distribution of the ACB excludes the femoral nerve, thus preserving greater quadriceps strength while providing equivalent levels of analgesia compared with FNB.15,17,18 The ACB has shown decreased near-fall events and improved balance scores in the immediate postoperative period.19
Our study analyzed opioid consumption patterns of TKA patients from a US Department of Veterans Affairs (VA) medical center before and after the institution of a multimodal analgesic protocol using ACB. The primary purpose of this study was to determine whether a protocol that included intraoperative spinal anesthesia with a postoperative multimodal analgesic regimen and ACB was associated with a decreased postoperative opioid requirement when compared with patients who received intraoperative general anesthesia and a traditional opioid regimen. Secondary outcomes included the effect of opioid consumption on range of motion on postoperative day (POD) 1 and number of opioid prescriptions written at the first postoperative clinic visit.
Methods
Approval for the study was obtained from the institutional review board at the Dayton Veterans Affairs Medical Center (DVAMC) in Ohio. A retrospective chart review was performed to collect data from all patients undergoing TKA at DVAMC from June 1, 2011, through December 31, 2015. Exclusion criteria included multiple surgeries in the study time frame, documented chronic pain, allergy to local anesthetics, daily preoperative use of opioids, and incomplete data in the health record.
All surgeries were performed by 2 staff arthroplasty surgeons at a single VAMC. All patients attended a preoperative visit where a history, physical, and anesthesia evaluation were performed, and watched an educational video detailing surgical indications and postoperative rehabilitation. All surgeries were performed with tourniquets and a periarticular injection was performed at the conclusion of each case. Surgeon 1 treatment of choice was 10 mL 0.5% bupivacaine, whereas surgeon 2 performed a posterior capsular injection of 30 mL 0.25% bupivacaine and a periarticular injection of 30 mg ketorolac in 10 mL 0.25% bupivacaine with epinephrine.
Prior to August 2014, general endotracheal anesthesia was used intraoperatively. A patient-controlled analgesia (PCA) pump of morphine or hydromorphone and additional oral oxycodone or hydrocodone was used for postoperative pain. PCA pumps were patient dependent. In the control group, 245 patients received the morphine PCA while 61 received the hydromorphone PCA. Morphine PCA dosing consisted of 1-mg doses every 10 minutes with potential baseline infusion rates of 0.5 to 1.0 mg/h and a 4-hour limit of 20 mg. Hydromorphone PCA dosing consisted of 0.2 to 0.4-mg doses with a potential continuous dose of 0.2 to 0.4 mg/h and a 4-hour limit of 4 mg.
In August 2014, a new analgesic protocol was adopted for TKA consisting of intraoperative spinal anesthesia (0.75% bupivacaine) with IV sedation (propofol), a postoperative multimodal analgesic regimen, an ACB performed in the postanesthesia care unit (PACU), and opioids as needed (protocol group). The ACB catheter was a 0.5% ropivo caine hydrochloride injection. It was attached to a local anesthetic fixed flow rate pump that administers 0.5% ropivacaine without epinephrine at 8 mL/h and was removed on POD 5 by the patient. The multimodal medication regimen included IV ketorolac 15 mg every 6 hours for 3 doses, gabapentin 300 mg every 8 hours, acetaminophen 975 mg every 8 hours, meloxicam 7.5 mg daily, tramadol 50 mg every 6 hours, oxycodone 5 mg 1 to 2 tabs every 4 hours as needed, and IV hydromorphone 0.5 mg every 4 hours as needed for breakthrough pain.
Preoperative demographic characteristics were collected (Table 1). Data on all IV and oral opioid requirements were collected for both groups, converted to morphine milligram equivalents (MME), and a total morphine equivalent dose (MED) was calculated.20,21
In April 2015, a separate protocol change occurred at the DVAMC with the goal of discharge on POD 1. To standardize outcomes before and after this change, data collection regarding opioid requirements was concluded at midnight on POD 1. If a patient was discharged before midnight on POD 1, opioid requirement through the time of discharge was collected. All surgeries were performed in the morning to early afternoon; however, specific surgical times were not collected. Patients were also evaluated by a physical therapist on POD 0, and maximal knee flexion and extension were measured on POD 1. Patients were discharged with prescriptions for oxycodone/acetaminophen and tramadol and were seen 3 weeks later for their first postoperative visit. Opioid refills at the first postoperative visit were recorded. All statistical analyses were performed in SAS 9.4 with significance set to α = 0.05. Between-groups differences in preoperative and perioperative characteristics as well as postoperative outcomes were analyzed using independent samples t tests for continuous variables and Fisher exact tests for dichotomous discrete variables. Where groups differed for a pre- or perioperative variable, linear mixed models analysis was used to determine whether IV, oral, and total MEDs were significantly affected by the interaction between the pre- or perioperative variable with analgesia group. For refills at the postoperative visit, the effects of pre- or perioperative differences were tested using χ2 tests. Effect sizes for outcome variables were estimated using Cohen d and probability of superiority (Δ) for continuous variables, and relative risk (RR) in the case of discrete variables.22
Results
During the study period from June 1, 2011, through December 31, 2015, 533 eligible TKAs were performed, 306 in the control group and 227 in the protocol group. The groups had similar sex distribution; body mass index; knee range of motion; diagnoses of diabetes mellitus, coronary artery disease, and chronic kidney disease; and history of deep vein thrombosis (DVT) or pulmonary embolism (P ≥ .05). The protocol group was significantly older (P = .04) and had a significantly higher rate of chronic obstructive pulmonary disease (COPD) (P = .002). There were no significant differences between number of procedures performed by surgeon (P = .48) or total tourniquet time (P = .13) (Table 2). Mean (SD) length of stay was significantly greater in the control group compared with the protocol group (2.5 [1.3] vs 1.4 [0.7] days, P < .001).
Figure 1 shows the distributions of each type of opioid used. Compared with the control group, the protocol group had a significantly lower mean (SD) IV opioid use: 178.2 (98.0) MED vs 12.0 (24.6) MED (P < .001; d = 2.19; Δ = 0.94) and mean (SD) total opioid use: 241.7 (120.1) MED vs 74.8 (42.7) MED (P < .001; d = 1.76; Δ = 0.89). Mean (SD) oral opioid use did not differ between groups (control, 63.6 [45.4] MED; protocol, 62.9 [31.4] MED; P = .85; d = 0.02; Δ = 0.51). A significantly lower percentage of patients in the protocol group received additional opioids at the 3-week follow-up when compared to the control group: 46.7% vs 61.3%, respectively (P < .001; RR, 0.76; 95% CI, 0.65-0.90).
There were no significant differences in postoperative mean (SD) maximum knee flexion (control, 67.2 [15.7]°; protocol, 67.8 [19.2]°; P = .72; d = 0.03; Δ = 0.51) or mean (SD) total flexion/extension arc (control, 66.2 [15.9]°; protocol, 67.9 [19.4]°; P = .32; d = 0.10; Δ = 0.53). Mean (SD) postoperative maximum knee extension was significantly higher in the protocol group compared with the control group (-0.1 [2.1]° vs 1.0 [3.7]°; P < .001; d = 0.35; Δ = 0.60). More patients in the protocol group (92.5%) were discharged to home compared with the control group (86.6%) (P = .02; RR, 1.07; 95% CI, 1.01-1.13).
Because age and rates of COPD differed between groups, sensitivity analyses were conducted to determine whether these variables influenced postoperative opioid use. The relationship between age and group was significant for IV (P < .001) and total opioid use (P < .001). Younger patients received higher MED doses than older patients within the control group, while dosages were fairly consistent regardless of age in the protocol group (Figure 2). There was no significance in age interaction effect with regard to oral opioids (P = .83) nor opioid refills at 3-week follow-up (P = .24).
The sensitivity analysis for COPD found that a diagnosis of COPD did not significantly influence utilization of IV opioids (P = .10), or total opioids (P = .68). There was a significant interaction effect for oral opioids (Figure 3). Patients in the control group with COPD required significantly higher mean (SD) oral opioids than patients without COPD (91.5 [123.9] MED and 62.0 [36.0] MED, respectively; P = .03). In the control group, the χ2 test was significant regarding opioid prescription refills at the 3-week visit (P = .004) with 62.4% of patients with COPD requiring refills vs 44.4% without COPD (P = .004). There was no difference in refills in the protocol group (46.4% vs 48.4%).
Finally, 2-sided independent samples t test evaluated total MED use between the 2 surgeons. There was no difference in total MED per patient for the surgeons. In the control group, mean (SD) total MED for surgeon 1 was 232.9 (118.7) MED vs 252.8 (121.5) MED for surgeon 2 (P = .18). In the protocol group, the mean (SD) total MED was 72.5 (43.2) and 77.4 (42.1) for surgeon 1 and surgeon 2, respectively (P = .39).
Discussion
Coordinated efforts with major medical organizations are being made to decrease opioid prescriptions and exposure.5,6 To our knowledge, no study has quantified a decrease in opioid requirement in a VA population after implementation of a protocol that includes intraoperative spinal anesthesia and a postoperative multimodal analgesic regimen including ACB after TKA. The analgesic protocol described in this study aligns with recommendations from both the CDC and the AAOS to decrease opioid use and misuse by maximizing nonopioid medications and limiting the size and number of opioid prescriptions. However, public and medical opinion of opioids as well as prescribing practices have changed over time with a trend toward lower opioid use. The interventions, as part of the described protocol, are a result of these changes and attempt to minimize opioid use while maximizing postoperative analgesia.
Our data showed a significant decrease in total opioid use through POD 1, IV opioid use, and opioid prescriptions provided at the first postoperative visit. The protocol group used only 6.7% of the IV opioids and 30.9% of the total opioids that were used by the control group. The substantial difference in IV opioid requirement, 166.2 MED, is equivalent to 8 mg of IV hydromorphone or 55 mg of IV morphine. The difference in total opioid requirement was similar at 166.9 MED, equivalent to 111 mg of oral oxycodone.
Decreasing opioid use has the additional benefit of improving outcomes, as higher doses of opioids have been associated with increased length of stay, greater rates of DVT, and postoperative infection.23 These complications occurred in a stepwise manner, suggesting a dose-response gradient that makes the sizable decrease noted in our data of greater relevance.23 While the adverse effects (AEs) of opioids are well known, there are limited data on opioid dosing and its effect on perioperative outcomes.23
A significant decrease in the percentage of patients receiving an opioid prescription at the first postoperative visit suggests a decrease in the number of patients on prolonged opioids after TKA with implementation of modern analgesic modalities. The duration of postoperative opioid use has been found to be the strongest predictor of misuse, and each postoperative refill increases the probability of misuse by 44%.24 In addition, opioid use for > 3 months after TKA is associated with increased risk of periprosthetic infection, increased overall revision rate, and stiffness at 1 year postoperatively.9 While not entirely under the control of the surgeon, measures to decrease the number of postoperative opioid refills may lead to a decrease in opioid misuse.
In the control group, older patients tended to receive less opioids. This is likely due to physiologic changes in opioid metabolism associated with aging, including decreased renal and hepatic opioid metabolism and alterations in overall body composition that increase relative potency and duration of action of opioids in a geriatric population.25,26 No difference in opioid use by age was found for the protocol group.
Patients in the protocol group demonstrated significantly greater maximal knee extension on POD 1 compared with the control group. No difference in maximal flexion was found. This difference in extension may partially be explained by the use of an ACB. One benefit of ACB is greater quadriceps strength and fewer near-fall events when compared with FNB.15,19
Our results corroborate the findings of similar studies. A randomized controlled trial comparing a multimodal analgesic regimen with a periarticular injection without a postoperative ACB to a hydromorphone PCA revealed a significant decrease in opioid use in the multimodal analgesic group.27 Along with lower opioid requirements, the multimodal analgesic group had lower visual analog scale pain scores, fewer AEs, faster progression to physical therapy milestones, and higher satisfaction.27 Recent guidelines from the French Society of Anaesthesia and Intensive Care Medicine recommend against the use of gabapentin as a method of postoperative pain control. However, this specifically refers to the preoperative administration of gabapentin. This same set of guidelines later cites a high level of evidence suggesting patients undergoing arthroplasty benefit more from gabapentinoids.28 Multiple analgesic protocols that include gabapentin as a part of a multimodal approach have been shown to have positive results.13,29
In our study, patients receiving the multimodal analgesic regimen were significantly more likely to be discharged home rather than to postacute care facilities, which have been associated with increased rates of major complications, 30-day readmission, and 30-day reoperation.30,31 In addition, discharge to an inpatient rehabilitation or skilled nursing facility has not been found to result in higher functional outcomes, despite $3.2 billion spent yearly on rehabilitation services after primary TKA.32,33
A component of our described analgesic protocol included spinal anesthesia intraoperatively. The differences between groups regarding anesthesia type can be attributed to this protocol change. A significantly greater percentage of patients in the protocol group received spinal anesthesia, while more patients in the control group received general anesthesia. While patients who received spinal anesthesia may have enhanced analgesia in the immediate postoperative period, no differences in opioid outcomes were seen based on anesthesia type. Known benefits of intraoperative spinal anesthesia include decreased perioperative blood loss and a smaller decrease in hemoglobin postoperatively, as well as lower rates of in-hospital complications, including pulmonary embolism, pneumonia, cerebrovascular events, and acute renal failure.34
Limitations
A number of limitations of this study should be noted. One was a protocol change regarding length of stay, which occurred during the study period and resulted in a significantly shorter length of stay in the protocol group. As a result, opioid use data were analyzed only through midnight at the end of POD 1. Patients who were discharged on POD 1 did not have opioid use data available for the full duration of the first POD, which may exaggerate the decrease in opioid requirements, as opioids used after discharge but prior to midnight on POD 1 were not recorded. However, opioids taken at home are oral with a low MME compared with IV opioids received by hospitalized patients in the control group. In addition, if taken as prescribed, patients at home would only have enough time to take a few doses of opioids prior to the midnight cutoff. We do not believe this difference in time of opioid use meaningfully affected the data. An additional limitation includes the variability between periarticular injections between surgeons. While the percentage of patients that received injections from surgeon 1 vs surgeon 2 were similar, it cannot be ruled out as a potential confounding factor. Other limitations include a lack of pain scores to compare subjective pain ratings, the retrospective nature of the study, and a largely homogenous male VA population.
Conclusions
Ease of access to opioids is a risk factor for opioid abuse, which itself is a risk factor for subsequent heroin use.1,2 The CDC and AAOS have thus published recommendations regarding opioid prescribing practices to decrease opioid use and abuse.5,6 Our described protocol, which aligns with these recommendations, resulted in a significant decrease in IV opioid requirement, total opioid requirement, and lower rates of opioid prescriptions provided at the first postoperative visit. These promising findings demonstrate a lower percentage of patients on long-term opioids after TKA and a significantly decreased cumulative opioid exposure.
1. Lankenau SE, Teti M, Silva K, Jackson Bloom J, Harocopos A, Treese M. Initiation into prescription opioid misuse amongst young injection drug users. Int J Drug Policy. 2012;23(1):37-44. doi:10.1016/j.drugpo.2011.05.014
2. Jones CM. Heroin use and heroin use risk behaviors among nonmedical users of prescription opioid pain relievers - United States, 2002-2004 and 2008-2010. Drug Alcohol Depend. 2013;132(1-2):95-100. doi:10.1016/j.drugalcdep.2013.01.007
3. Manchikanti L, Singh A. Therapeutic opioids: a ten-year perspective on the complexities and complications of the escalating use, abuse, and nonmedical use of opioids. Pain Physician. 2008;11(suppl 2):S63-S88.
4. Seth P, Scholl L, Rudd RA, Bacon S. Overdose deaths involving opioids, cocaine, and psychostimulants - United States, 2015-2016. MMWR Morb Mortal Wkly Rep. 2018;67(12):349-358. Published 2018 Mar 30. doi:10.15585/mmwr.mm6712a1
5. Dowell D, Haegerich TM, Chou R. CDC Guideline for Prescribing Opioids for Chronic Pain-United States, 2016. JAMA. 2016;315(15):1624-1645. doi:10.1001/jama.2016.1464
6. American Academy of Orthopaedic Surgeons. Information statement: opioid use, misuse, and abuse in orthopaedic practice. Published October 2015. Accessed November 12, 2021. https://aaos.org/globalassets/about /bylaws-library/information-statements/1045-opioid-use -misuse-and-abuse-in-practice.pdf
7. Hernandez NM, Parry JA, Taunton MJ. Patients at risk: large opioid prescriptions after total knee arthroplasty. J Arthroplasty. 2017;32(8):2395-2398. doi:10.1016/j.arth.2017.02.060
8. Gerner P, Poeran J, Cozowicz C, Mörwald EE, Zubizarreta N, Mazumdar M, Memtsoudis SG, Multimodal pain management in total hip and knee arthroplasty: trends over the last 10 years. Abstract presented at: American Society of Anesthesiologists Annual Meeting; October 21, 2017; Boston, MA.
9. Cancienne JM, Patel KJ, Browne JA, Werner BC. Narcotic use and total knee arthroplasty. J Arthroplasty. 2018;33(1):113-118. doi:10.1016/j.arth.2017.08.006
10. Moucha CS, Weiser MC, Levin EJ. Current strategies in anesthesia and analgesia for total knee arthroplasty. J Am Acad Orthop Surg. 2016;24(2):60-73. doi:10.5435/JAAOS-D-14-00259
11. Wick EC, Grant MC, Wu CL. Postoperative multimodal analgesia pain management with nonopioid analgesics and techniques: a review. JAMA Surg. 2017;152(7):691-697.doi:10.1001/jamasurg.2017.0898
12. Lamplot JD, Wagner ER, Manning DW. Multimodal pain management in total knee arthroplasty: a prospective randomized controlled trial. J Arthoplasty. 2014;29(2):329-334. doi:10.1016/j.arth.2013.06.005
13. Golladay GJ, Balch KR, Dalury DF, Satpathy J, Jiranek WA. Oral multimodal analgesia for total joint arthroplasty. J Arthroplasty. 2017;32(9S):S69-S73. doi:10.1016/j.arth.2017.05.002
14. Ardon AE, Clendenen SR, Porter SB, Robards CB, Greengrass RA. Opioid consumption in total knee arthroplasty patients: a retrospective comparison of adductor canal and femoral nerve continuous infusions in the presence of a sciatic nerve catheter. J Clin Anesth. 2016;31:19-26. doi:10.1016/j.jclinane.2015.12.014
15. Li D, Ma GG. Analgesic efficacy and quadriceps strength of adductor canal block versus femoral nerve block following total knee arthroplasty. Knee Surg Sports Traumatol Arthrosc. 2016;24(8):2614-2619. doi:10.1007/s00167-015-3874-3
16. Li D, Yang Z, Xie X, Zhao J, Kang P. Adductor canal block provides better performance after total knee arthroplasty compared with femoral nerve block: a systematic review and meta-analysis. Int Orthop. 2016;40(5):925-933. doi:10.1007/s00264-015-2998-x
17. Horner G, Dellon AL. Innervation of the human knee joint and implications for surgery. Clin Orthop Relat Res. 1994;(301):221-226.
18. Kim DH, Lin Y, Goytizolo EA, et al. Adductor canal block versus femoral nerve block for total knee arthroplasty: a prospective, randomized, controlled trial. Anesthesiology. 2014;120(3):540-550. doi:10.1097/ALN.0000000000000119
19. Thacher RR, Hickernell TR, Grosso MJ, et al. Decreased risk of knee buckling with adductor canal block versus femoral nerve block in total knee arthroplasty: a retrospective cohort study. Arthroplasty Today. 2017;3(4):281-285. Published 2017 Apr 15. doi:10.1016/j.artd.2017.02.008
20. Von Korff M, Saunders K, Thomas Ray G, et al. De facto long-term opioid therapy for noncancer pain [published correction appears in Clin J Pain. 2014 Sep;30(9):830. Korff, Michael Von [corrected to Von Korff, Michael]]. Clin J Pain. 2008;24(6):521-527. doi:10.1097/AJP.0b013e318169d03b
21. Kishner S. Opioid equivalents and conversions: overview. Published January 29, 2018. Accessed November 12, 2021. https://emedicine.medscape.com/article/2138678 -overview#a1
22. Ruscio J, Mullen T. Confidence intervals for the probability of superiority effect size measure and the area under a receiver operating characteristic curve. Multivariate Behav Res. 2012;47(2):201-223. doi:10.1080/00273171.2012.658329
23. Cozowicz C, Olson A, Poeran J, et al. Opioid prescription levels and postoperative outcomes in orthopedic orthopedic surgery. Pain. 2017;158(12):2422-2430. doi:10.1097/j.pain.0000000000001047
24. Brat GA, Agniel D, Beam A, et al. Postsurgical prescriptions for opioid naive patients and association with overdose and misuse: retrospective cohort study. BMJ. 2018;360:j5790. Published 2018 Jan 17. doi:10.1136/bmj.j5790
25. Tegeder I, Lötsch J, Geisslinger G. Pharmacokinetics of opioids in liver disease. Clin Pharmacokinet. 1999;37(1):17- 40. doi:10.2165/00003088-199937010-00002
26. Linnebur SA, O’Connell MB, Wessell AM, et al. Pharmacy practice, research, education, and advocacy for older adults. Pharmacotherapy. 2005;25(10):1396-1430. doi:10.1592/phco.2005.25.10.1396
27. Lamplot JD, Wagner ER, Manning DW. Multimodal pain management in total knee arthroplasty: a prospective randomized controlled trial. J Arthroplasty. 2014;29(2):329- 334. doi:10.1016/j.arth.2013.06.005
28. Aubrun F, Nouette-Gaulain K, Fletcher D, et al. Revision of expert panel’s guidelines on postoperative pain management. Anaesth Crit Care Pain Med. 2019;38(4):405-411. doi:10.1016/j.accpm.2019.02.011
29. Han C, Li XD, Jiang HQ, Ma JX, Ma XL. The use of gabapentin in the management of postoperative pain after total knee arthroplasty: A PRISMA-compliant metaanalysis of randomized controlled trials [published correction appears in Medicine (Baltimore). 2016 Jul 18;95(28):e0916]. Medicine (Baltimore). 2016;95(23):e3883. doi:10.1097/MD.0000000000003883
30. McLawhorn AS, Fu MC, Schairer WW, Sculco PK, MacLean CH, Padgett DE. Continued inpatient care after primary total knee arthroplasty increases 30-day postdischarge complications: a propensity score-adjusted analysis. J Arthroplasty. 2017;32(9S):S113-S118. doi:10.1016/j.arth.2017.01.039
31. Pelt CE, Gililland JM, Erickson JA, Trimble DE, Anderson MB, Peters CL. Improving value in total joint arthroplasty: a comprehensive patient education and management program decreases discharge to post-acute care facilities and post-operative complications. J Arthroplasty. 2018;33(1):14-18. doi:10.1016/j.arth.2017.08.003
32. Padgett DE, Christ AB, Joseph AD, Lee YY, Haas SB, Lyman S. Discharge to inpatient rehab does not result in improved functional outcomes following primary total knee arthroplasty. J Arthroplasty. 2018;33(6):1663-1667. doi:10.1016/j.arth.2017.12.033
33. Lavernia CJ, D’Apuzzo MR, Hernandez VH, Lee DJ, Rossi MD. Postdischarge costs in arthroplasty surgery. J Arthroplasty. 2006;21(6 Suppl 2):144-150. doi:10.1016/j.arth.2006.05.003
1. Lankenau SE, Teti M, Silva K, Jackson Bloom J, Harocopos A, Treese M. Initiation into prescription opioid misuse amongst young injection drug users. Int J Drug Policy. 2012;23(1):37-44. doi:10.1016/j.drugpo.2011.05.014
2. Jones CM. Heroin use and heroin use risk behaviors among nonmedical users of prescription opioid pain relievers - United States, 2002-2004 and 2008-2010. Drug Alcohol Depend. 2013;132(1-2):95-100. doi:10.1016/j.drugalcdep.2013.01.007
3. Manchikanti L, Singh A. Therapeutic opioids: a ten-year perspective on the complexities and complications of the escalating use, abuse, and nonmedical use of opioids. Pain Physician. 2008;11(suppl 2):S63-S88.
4. Seth P, Scholl L, Rudd RA, Bacon S. Overdose deaths involving opioids, cocaine, and psychostimulants - United States, 2015-2016. MMWR Morb Mortal Wkly Rep. 2018;67(12):349-358. Published 2018 Mar 30. doi:10.15585/mmwr.mm6712a1
5. Dowell D, Haegerich TM, Chou R. CDC Guideline for Prescribing Opioids for Chronic Pain-United States, 2016. JAMA. 2016;315(15):1624-1645. doi:10.1001/jama.2016.1464
6. American Academy of Orthopaedic Surgeons. Information statement: opioid use, misuse, and abuse in orthopaedic practice. Published October 2015. Accessed November 12, 2021. https://aaos.org/globalassets/about /bylaws-library/information-statements/1045-opioid-use -misuse-and-abuse-in-practice.pdf
7. Hernandez NM, Parry JA, Taunton MJ. Patients at risk: large opioid prescriptions after total knee arthroplasty. J Arthroplasty. 2017;32(8):2395-2398. doi:10.1016/j.arth.2017.02.060
8. Gerner P, Poeran J, Cozowicz C, Mörwald EE, Zubizarreta N, Mazumdar M, Memtsoudis SG, Multimodal pain management in total hip and knee arthroplasty: trends over the last 10 years. Abstract presented at: American Society of Anesthesiologists Annual Meeting; October 21, 2017; Boston, MA.
9. Cancienne JM, Patel KJ, Browne JA, Werner BC. Narcotic use and total knee arthroplasty. J Arthroplasty. 2018;33(1):113-118. doi:10.1016/j.arth.2017.08.006
10. Moucha CS, Weiser MC, Levin EJ. Current strategies in anesthesia and analgesia for total knee arthroplasty. J Am Acad Orthop Surg. 2016;24(2):60-73. doi:10.5435/JAAOS-D-14-00259
11. Wick EC, Grant MC, Wu CL. Postoperative multimodal analgesia pain management with nonopioid analgesics and techniques: a review. JAMA Surg. 2017;152(7):691-697.doi:10.1001/jamasurg.2017.0898
12. Lamplot JD, Wagner ER, Manning DW. Multimodal pain management in total knee arthroplasty: a prospective randomized controlled trial. J Arthoplasty. 2014;29(2):329-334. doi:10.1016/j.arth.2013.06.005
13. Golladay GJ, Balch KR, Dalury DF, Satpathy J, Jiranek WA. Oral multimodal analgesia for total joint arthroplasty. J Arthroplasty. 2017;32(9S):S69-S73. doi:10.1016/j.arth.2017.05.002
14. Ardon AE, Clendenen SR, Porter SB, Robards CB, Greengrass RA. Opioid consumption in total knee arthroplasty patients: a retrospective comparison of adductor canal and femoral nerve continuous infusions in the presence of a sciatic nerve catheter. J Clin Anesth. 2016;31:19-26. doi:10.1016/j.jclinane.2015.12.014
15. Li D, Ma GG. Analgesic efficacy and quadriceps strength of adductor canal block versus femoral nerve block following total knee arthroplasty. Knee Surg Sports Traumatol Arthrosc. 2016;24(8):2614-2619. doi:10.1007/s00167-015-3874-3
16. Li D, Yang Z, Xie X, Zhao J, Kang P. Adductor canal block provides better performance after total knee arthroplasty compared with femoral nerve block: a systematic review and meta-analysis. Int Orthop. 2016;40(5):925-933. doi:10.1007/s00264-015-2998-x
17. Horner G, Dellon AL. Innervation of the human knee joint and implications for surgery. Clin Orthop Relat Res. 1994;(301):221-226.
18. Kim DH, Lin Y, Goytizolo EA, et al. Adductor canal block versus femoral nerve block for total knee arthroplasty: a prospective, randomized, controlled trial. Anesthesiology. 2014;120(3):540-550. doi:10.1097/ALN.0000000000000119
19. Thacher RR, Hickernell TR, Grosso MJ, et al. Decreased risk of knee buckling with adductor canal block versus femoral nerve block in total knee arthroplasty: a retrospective cohort study. Arthroplasty Today. 2017;3(4):281-285. Published 2017 Apr 15. doi:10.1016/j.artd.2017.02.008
20. Von Korff M, Saunders K, Thomas Ray G, et al. De facto long-term opioid therapy for noncancer pain [published correction appears in Clin J Pain. 2014 Sep;30(9):830. Korff, Michael Von [corrected to Von Korff, Michael]]. Clin J Pain. 2008;24(6):521-527. doi:10.1097/AJP.0b013e318169d03b
21. Kishner S. Opioid equivalents and conversions: overview. Published January 29, 2018. Accessed November 12, 2021. https://emedicine.medscape.com/article/2138678 -overview#a1
22. Ruscio J, Mullen T. Confidence intervals for the probability of superiority effect size measure and the area under a receiver operating characteristic curve. Multivariate Behav Res. 2012;47(2):201-223. doi:10.1080/00273171.2012.658329
23. Cozowicz C, Olson A, Poeran J, et al. Opioid prescription levels and postoperative outcomes in orthopedic orthopedic surgery. Pain. 2017;158(12):2422-2430. doi:10.1097/j.pain.0000000000001047
24. Brat GA, Agniel D, Beam A, et al. Postsurgical prescriptions for opioid naive patients and association with overdose and misuse: retrospective cohort study. BMJ. 2018;360:j5790. Published 2018 Jan 17. doi:10.1136/bmj.j5790
25. Tegeder I, Lötsch J, Geisslinger G. Pharmacokinetics of opioids in liver disease. Clin Pharmacokinet. 1999;37(1):17- 40. doi:10.2165/00003088-199937010-00002
26. Linnebur SA, O’Connell MB, Wessell AM, et al. Pharmacy practice, research, education, and advocacy for older adults. Pharmacotherapy. 2005;25(10):1396-1430. doi:10.1592/phco.2005.25.10.1396
27. Lamplot JD, Wagner ER, Manning DW. Multimodal pain management in total knee arthroplasty: a prospective randomized controlled trial. J Arthroplasty. 2014;29(2):329- 334. doi:10.1016/j.arth.2013.06.005
28. Aubrun F, Nouette-Gaulain K, Fletcher D, et al. Revision of expert panel’s guidelines on postoperative pain management. Anaesth Crit Care Pain Med. 2019;38(4):405-411. doi:10.1016/j.accpm.2019.02.011
29. Han C, Li XD, Jiang HQ, Ma JX, Ma XL. The use of gabapentin in the management of postoperative pain after total knee arthroplasty: A PRISMA-compliant metaanalysis of randomized controlled trials [published correction appears in Medicine (Baltimore). 2016 Jul 18;95(28):e0916]. Medicine (Baltimore). 2016;95(23):e3883. doi:10.1097/MD.0000000000003883
30. McLawhorn AS, Fu MC, Schairer WW, Sculco PK, MacLean CH, Padgett DE. Continued inpatient care after primary total knee arthroplasty increases 30-day postdischarge complications: a propensity score-adjusted analysis. J Arthroplasty. 2017;32(9S):S113-S118. doi:10.1016/j.arth.2017.01.039
31. Pelt CE, Gililland JM, Erickson JA, Trimble DE, Anderson MB, Peters CL. Improving value in total joint arthroplasty: a comprehensive patient education and management program decreases discharge to post-acute care facilities and post-operative complications. J Arthroplasty. 2018;33(1):14-18. doi:10.1016/j.arth.2017.08.003
32. Padgett DE, Christ AB, Joseph AD, Lee YY, Haas SB, Lyman S. Discharge to inpatient rehab does not result in improved functional outcomes following primary total knee arthroplasty. J Arthroplasty. 2018;33(6):1663-1667. doi:10.1016/j.arth.2017.12.033
33. Lavernia CJ, D’Apuzzo MR, Hernandez VH, Lee DJ, Rossi MD. Postdischarge costs in arthroplasty surgery. J Arthroplasty. 2006;21(6 Suppl 2):144-150. doi:10.1016/j.arth.2006.05.003
Platelet-rich plasma injections show no benefit in knee OA in placebo-controlled trial
A large randomized, placebo-controlled trial of platelet-rich plasma injections for knee osteoarthritis has found almost no symptomatic or structural benefit from the treatment, giving some clarity to an evidence base that has seen both positive and negative trials for the treatment modality.
Given the need for better disease-modifying treatments for osteoarthritis, there has been a lot of interest in biological therapies such as platelet-rich plasma and stem cells, the lead author of the study, Kim Bennell, PhD, told this news organization. “People have started to use it to treat osteoarthritis, but the evidence to support it was limited in terms of its quality, and there’s been very little work looking at effects on structure,” said Dr. Bennell, a research physiotherapist and chair of physiotherapy at the University of Melbourne.
Platelet-rich plasma contains a range of growth factors and cytokines that are thought to be beneficial in building cartilage and reducing inflammation. There have been several clinical trials of the treatment in knee osteoarthritis, but the current study’s authors said these were limited by factors such as a lack of blinding and were at high risk of bias. “That was the impetus to do a large, high-quality study and to look at joint structure,” Dr. Bennell said.
Study details
For the study, which was published Nov. 23 in JAMA, the researchers enrolled 288 adults older than 50 with knee osteoarthritis who had experienced knee pain on most days of the past month and had radiographic evidence of mild to moderate osteoarthritis of the tibiofemoral joint.
After having stopped all nonsteroidal anti-inflammatory and pain-relief drugs 2 weeks prior – except acetaminophen – participants were randomly assigned to receive three weekly intra-articular knee injections of either a commercially available leukocyte-poor platelet-rich plasma or saline placebo. They were then followed for 12 months.
Among the 288 participants in the study, researchers saw no statistically significant difference in the change in pain scores between the treatment and placebo groups at 12 months, although there was a nonsignificantly greater reduction in pain scores among those given platelet-rich plasma. The study also found no statistically significant difference between the two groups in the change in medial tibial cartilage volume.
The researchers also looked at a large number of secondary outcomes, including the effects of treatment on pain and function at 2 months, change in Knee Injury and Osteoarthritis Outcome (KOOS) scores, and change in quality of life scores. There were no indications of any benefits from the treatment at the 2-month follow-up, and at 12 months, the study showed no significant improvements in knee pain while walking or in pain scores, KOOS scores, or quality of life measures.
However, significantly more participants in the treatment group than in the placebo group reported overall improvement at the 2-month point – 48.2% of those in the treatment arm, compared with 36.2% of the placebo group (risk ratio, 1.37; 95% confidence interval, 1.05-1.80; P = .02). At 12 months, 42.8% of those who received platelet-rich plasma reported improved function, compared with 32.1% of those in the placebo group (risk ratio, 1.36; 95% CI, 1.00-1.86, P = .05).
The study also found that significantly more people in the platelet-rich plasma group had three or more areas of cartilage thinning at 12 months (17.1% vs. 6.8%; risk ratio, 2.71; 95% CI, 1.16-6.34; P = .02).
Even when researchers looked for treatment effects in subgroups – for example, based on disease severity, body mass index, or knee alignment – they found no significant differences from placebo.
Dr. Bennell said the results were disappointing but not surprising. “Anecdotally, people do report that they get better, but we know that there is a very large placebo effect with treatment of pain,” she said.
Results emphasize importance of placebo controls
In an accompanying editorial by Jeffrey N. Katz, MD, director of the Orthopaedic and Arthritis Center for Outcomes Research at Brigham and Women’s Hospital, professor of medicine and orthopedic surgery at Harvard Medical School, and professor of epidemiology and environmental health at the Harvard T.H. Chan School of Public Health, all in Boston, draws parallels between this study and two earlier studies of platelet-rich plasma for ankle osteoarthritis and Achilles tendinopathy, both published in JAMA in 2021. None of the three studies showed any significant improvements over and above placebo.
“These findings emphasize the importance of comparing interventions with placebos in trials of injection therapies,” Dr. Katz writes. However, he notes that these studies do suggest possible benefits in secondary outcomes, such as self-reported pain and function, and that earlier studies of the treatment had had more positive outcomes.
Dr. Katz said it was premature to dismiss platelet-rich plasma as a treatment for knee osteoarthritis, but “until a new generation of trials using standardized approaches to PRP [platelet-rich plasma] therapy provides evidence of efficacy, it would be prudent to pause the use of PRP for OA and Achilles tendinitis.”
Not ready to stop using platelet-rich plasma?
When asked for comment, sports medicine physician Maarten Moen, MD, from the Bergman Clinics Naarden (the Netherlands) said the study was the largest yet of the use of platelet-rich plasma for knee osteoarthritis and that it was a well-designed, double-blind, placebo-controlled trial.
However, he also pointed out that at least six earlier randomized, placebo-controlled studies of this treatment approach have been conducted, and of those six, all but two found positive benefits for patients. “It’s a very well-performed study, but for me, it would be a bridge too far to say, ‘Now we have this study, let’s stop doing it,’ ” Dr. Moen said.
Dr. Moen said he would like to see what effect this study had on meta-analyses and systematic reviews of the treatment, as that would give the clearest indication of the overall picture of its effectiveness.
Dr. Moen’s own experience of treating patients with platelet-rich plasma also suggested that, among those who do benefit from the treatment, that benefit would most likely show between 2 and 12 months afterward. He said it would have been useful to see outcomes at 3- and 6-month intervals.
“What I tell people is that, on average, around 9 months’ effect is to be expected,” he said.
Dr. Bennell said the research group chose the 12-month follow-up because they wanted to see if there were long-term improvements in joint structure which they hoped for, given the cost of treatment.
The study was funded by the Australian National Health and Medical Research Council, and Regen Lab SA provided platelet-rich plasma kits free of charge. Two authors reported using platelet-rich plasma injections in clinical practice, one reported scientific advisory board fees from Biobone, Novartis, Tissuegene, Pfizer, and Lilly; two reported fees for contributing to UpToDate clinical guidelines, and two reported grants from the National Health and Medical Research Council outside the submitted work. No other conflicts of interest were declared.
A version of this article first appeared on Medscape.com.
A large randomized, placebo-controlled trial of platelet-rich plasma injections for knee osteoarthritis has found almost no symptomatic or structural benefit from the treatment, giving some clarity to an evidence base that has seen both positive and negative trials for the treatment modality.
Given the need for better disease-modifying treatments for osteoarthritis, there has been a lot of interest in biological therapies such as platelet-rich plasma and stem cells, the lead author of the study, Kim Bennell, PhD, told this news organization. “People have started to use it to treat osteoarthritis, but the evidence to support it was limited in terms of its quality, and there’s been very little work looking at effects on structure,” said Dr. Bennell, a research physiotherapist and chair of physiotherapy at the University of Melbourne.
Platelet-rich plasma contains a range of growth factors and cytokines that are thought to be beneficial in building cartilage and reducing inflammation. There have been several clinical trials of the treatment in knee osteoarthritis, but the current study’s authors said these were limited by factors such as a lack of blinding and were at high risk of bias. “That was the impetus to do a large, high-quality study and to look at joint structure,” Dr. Bennell said.
Study details
For the study, which was published Nov. 23 in JAMA, the researchers enrolled 288 adults older than 50 with knee osteoarthritis who had experienced knee pain on most days of the past month and had radiographic evidence of mild to moderate osteoarthritis of the tibiofemoral joint.
After having stopped all nonsteroidal anti-inflammatory and pain-relief drugs 2 weeks prior – except acetaminophen – participants were randomly assigned to receive three weekly intra-articular knee injections of either a commercially available leukocyte-poor platelet-rich plasma or saline placebo. They were then followed for 12 months.
Among the 288 participants in the study, researchers saw no statistically significant difference in the change in pain scores between the treatment and placebo groups at 12 months, although there was a nonsignificantly greater reduction in pain scores among those given platelet-rich plasma. The study also found no statistically significant difference between the two groups in the change in medial tibial cartilage volume.
The researchers also looked at a large number of secondary outcomes, including the effects of treatment on pain and function at 2 months, change in Knee Injury and Osteoarthritis Outcome (KOOS) scores, and change in quality of life scores. There were no indications of any benefits from the treatment at the 2-month follow-up, and at 12 months, the study showed no significant improvements in knee pain while walking or in pain scores, KOOS scores, or quality of life measures.
However, significantly more participants in the treatment group than in the placebo group reported overall improvement at the 2-month point – 48.2% of those in the treatment arm, compared with 36.2% of the placebo group (risk ratio, 1.37; 95% confidence interval, 1.05-1.80; P = .02). At 12 months, 42.8% of those who received platelet-rich plasma reported improved function, compared with 32.1% of those in the placebo group (risk ratio, 1.36; 95% CI, 1.00-1.86, P = .05).
The study also found that significantly more people in the platelet-rich plasma group had three or more areas of cartilage thinning at 12 months (17.1% vs. 6.8%; risk ratio, 2.71; 95% CI, 1.16-6.34; P = .02).
Even when researchers looked for treatment effects in subgroups – for example, based on disease severity, body mass index, or knee alignment – they found no significant differences from placebo.
Dr. Bennell said the results were disappointing but not surprising. “Anecdotally, people do report that they get better, but we know that there is a very large placebo effect with treatment of pain,” she said.
Results emphasize importance of placebo controls
In an accompanying editorial by Jeffrey N. Katz, MD, director of the Orthopaedic and Arthritis Center for Outcomes Research at Brigham and Women’s Hospital, professor of medicine and orthopedic surgery at Harvard Medical School, and professor of epidemiology and environmental health at the Harvard T.H. Chan School of Public Health, all in Boston, draws parallels between this study and two earlier studies of platelet-rich plasma for ankle osteoarthritis and Achilles tendinopathy, both published in JAMA in 2021. None of the three studies showed any significant improvements over and above placebo.
“These findings emphasize the importance of comparing interventions with placebos in trials of injection therapies,” Dr. Katz writes. However, he notes that these studies do suggest possible benefits in secondary outcomes, such as self-reported pain and function, and that earlier studies of the treatment had had more positive outcomes.
Dr. Katz said it was premature to dismiss platelet-rich plasma as a treatment for knee osteoarthritis, but “until a new generation of trials using standardized approaches to PRP [platelet-rich plasma] therapy provides evidence of efficacy, it would be prudent to pause the use of PRP for OA and Achilles tendinitis.”
Not ready to stop using platelet-rich plasma?
When asked for comment, sports medicine physician Maarten Moen, MD, from the Bergman Clinics Naarden (the Netherlands) said the study was the largest yet of the use of platelet-rich plasma for knee osteoarthritis and that it was a well-designed, double-blind, placebo-controlled trial.
However, he also pointed out that at least six earlier randomized, placebo-controlled studies of this treatment approach have been conducted, and of those six, all but two found positive benefits for patients. “It’s a very well-performed study, but for me, it would be a bridge too far to say, ‘Now we have this study, let’s stop doing it,’ ” Dr. Moen said.
Dr. Moen said he would like to see what effect this study had on meta-analyses and systematic reviews of the treatment, as that would give the clearest indication of the overall picture of its effectiveness.
Dr. Moen’s own experience of treating patients with platelet-rich plasma also suggested that, among those who do benefit from the treatment, that benefit would most likely show between 2 and 12 months afterward. He said it would have been useful to see outcomes at 3- and 6-month intervals.
“What I tell people is that, on average, around 9 months’ effect is to be expected,” he said.
Dr. Bennell said the research group chose the 12-month follow-up because they wanted to see if there were long-term improvements in joint structure which they hoped for, given the cost of treatment.
The study was funded by the Australian National Health and Medical Research Council, and Regen Lab SA provided platelet-rich plasma kits free of charge. Two authors reported using platelet-rich plasma injections in clinical practice, one reported scientific advisory board fees from Biobone, Novartis, Tissuegene, Pfizer, and Lilly; two reported fees for contributing to UpToDate clinical guidelines, and two reported grants from the National Health and Medical Research Council outside the submitted work. No other conflicts of interest were declared.
A version of this article first appeared on Medscape.com.
A large randomized, placebo-controlled trial of platelet-rich plasma injections for knee osteoarthritis has found almost no symptomatic or structural benefit from the treatment, giving some clarity to an evidence base that has seen both positive and negative trials for the treatment modality.
Given the need for better disease-modifying treatments for osteoarthritis, there has been a lot of interest in biological therapies such as platelet-rich plasma and stem cells, the lead author of the study, Kim Bennell, PhD, told this news organization. “People have started to use it to treat osteoarthritis, but the evidence to support it was limited in terms of its quality, and there’s been very little work looking at effects on structure,” said Dr. Bennell, a research physiotherapist and chair of physiotherapy at the University of Melbourne.
Platelet-rich plasma contains a range of growth factors and cytokines that are thought to be beneficial in building cartilage and reducing inflammation. There have been several clinical trials of the treatment in knee osteoarthritis, but the current study’s authors said these were limited by factors such as a lack of blinding and were at high risk of bias. “That was the impetus to do a large, high-quality study and to look at joint structure,” Dr. Bennell said.
Study details
For the study, which was published Nov. 23 in JAMA, the researchers enrolled 288 adults older than 50 with knee osteoarthritis who had experienced knee pain on most days of the past month and had radiographic evidence of mild to moderate osteoarthritis of the tibiofemoral joint.
After having stopped all nonsteroidal anti-inflammatory and pain-relief drugs 2 weeks prior – except acetaminophen – participants were randomly assigned to receive three weekly intra-articular knee injections of either a commercially available leukocyte-poor platelet-rich plasma or saline placebo. They were then followed for 12 months.
Among the 288 participants in the study, researchers saw no statistically significant difference in the change in pain scores between the treatment and placebo groups at 12 months, although there was a nonsignificantly greater reduction in pain scores among those given platelet-rich plasma. The study also found no statistically significant difference between the two groups in the change in medial tibial cartilage volume.
The researchers also looked at a large number of secondary outcomes, including the effects of treatment on pain and function at 2 months, change in Knee Injury and Osteoarthritis Outcome (KOOS) scores, and change in quality of life scores. There were no indications of any benefits from the treatment at the 2-month follow-up, and at 12 months, the study showed no significant improvements in knee pain while walking or in pain scores, KOOS scores, or quality of life measures.
However, significantly more participants in the treatment group than in the placebo group reported overall improvement at the 2-month point – 48.2% of those in the treatment arm, compared with 36.2% of the placebo group (risk ratio, 1.37; 95% confidence interval, 1.05-1.80; P = .02). At 12 months, 42.8% of those who received platelet-rich plasma reported improved function, compared with 32.1% of those in the placebo group (risk ratio, 1.36; 95% CI, 1.00-1.86, P = .05).
The study also found that significantly more people in the platelet-rich plasma group had three or more areas of cartilage thinning at 12 months (17.1% vs. 6.8%; risk ratio, 2.71; 95% CI, 1.16-6.34; P = .02).
Even when researchers looked for treatment effects in subgroups – for example, based on disease severity, body mass index, or knee alignment – they found no significant differences from placebo.
Dr. Bennell said the results were disappointing but not surprising. “Anecdotally, people do report that they get better, but we know that there is a very large placebo effect with treatment of pain,” she said.
Results emphasize importance of placebo controls
In an accompanying editorial by Jeffrey N. Katz, MD, director of the Orthopaedic and Arthritis Center for Outcomes Research at Brigham and Women’s Hospital, professor of medicine and orthopedic surgery at Harvard Medical School, and professor of epidemiology and environmental health at the Harvard T.H. Chan School of Public Health, all in Boston, draws parallels between this study and two earlier studies of platelet-rich plasma for ankle osteoarthritis and Achilles tendinopathy, both published in JAMA in 2021. None of the three studies showed any significant improvements over and above placebo.
“These findings emphasize the importance of comparing interventions with placebos in trials of injection therapies,” Dr. Katz writes. However, he notes that these studies do suggest possible benefits in secondary outcomes, such as self-reported pain and function, and that earlier studies of the treatment had had more positive outcomes.
Dr. Katz said it was premature to dismiss platelet-rich plasma as a treatment for knee osteoarthritis, but “until a new generation of trials using standardized approaches to PRP [platelet-rich plasma] therapy provides evidence of efficacy, it would be prudent to pause the use of PRP for OA and Achilles tendinitis.”
Not ready to stop using platelet-rich plasma?
When asked for comment, sports medicine physician Maarten Moen, MD, from the Bergman Clinics Naarden (the Netherlands) said the study was the largest yet of the use of platelet-rich plasma for knee osteoarthritis and that it was a well-designed, double-blind, placebo-controlled trial.
However, he also pointed out that at least six earlier randomized, placebo-controlled studies of this treatment approach have been conducted, and of those six, all but two found positive benefits for patients. “It’s a very well-performed study, but for me, it would be a bridge too far to say, ‘Now we have this study, let’s stop doing it,’ ” Dr. Moen said.
Dr. Moen said he would like to see what effect this study had on meta-analyses and systematic reviews of the treatment, as that would give the clearest indication of the overall picture of its effectiveness.
Dr. Moen’s own experience of treating patients with platelet-rich plasma also suggested that, among those who do benefit from the treatment, that benefit would most likely show between 2 and 12 months afterward. He said it would have been useful to see outcomes at 3- and 6-month intervals.
“What I tell people is that, on average, around 9 months’ effect is to be expected,” he said.
Dr. Bennell said the research group chose the 12-month follow-up because they wanted to see if there were long-term improvements in joint structure which they hoped for, given the cost of treatment.
The study was funded by the Australian National Health and Medical Research Council, and Regen Lab SA provided platelet-rich plasma kits free of charge. Two authors reported using platelet-rich plasma injections in clinical practice, one reported scientific advisory board fees from Biobone, Novartis, Tissuegene, Pfizer, and Lilly; two reported fees for contributing to UpToDate clinical guidelines, and two reported grants from the National Health and Medical Research Council outside the submitted work. No other conflicts of interest were declared.
A version of this article first appeared on Medscape.com.
FROM JAMA
PT may lower risk of long-term opioid use after knee replacement
A new study has found that physical therapy may lead to a reduced risk of long-term opioid use in patients who have undergone total knee replacement (TKR).
“Greater number of PT intervention sessions and earlier initiation of outpatient PT care after TKR were associated with lower odds of long-term opioid use,” authors from Boston University wrote in their report on the study, which was published online Oct. 27 in JAMA Network Open.
“In previous large studies, we’ve seen that physical therapy can reduce pain in people with knee osteoarthritis, which is usually the primary indication for TKR,” study coauthor Deepak Kumar, PT, PhD, said in an interview. “But the association of physical therapy with opioid use in people with knee replacement has not yet been explored.
“The reason we focused on opioid use in these patients is because the number of knee replacement surgeries is going up exponentially,” Dr. Kumar said. “And, depending on which data you look at, from one-third to up to half of people who undergo knee replacement and have used opioids before end up becoming long-term users. Even in people who have not used them before, 5%-8% become long-term users after the surgery.
“Given how many surgeries are happening – and that number is expected to keep going up – the number of people who are becoming long-term opioid users is not trivial,” he said.
Study details
To assess the value of PT in reducing opioid use in this subset of patients, the authors reviewed records from the OptumLabs Data Warehouse insurance claims database to identify 67,322 eligible participants aged 40 or older who underwent TKR from Jan. 1, 2001, to Dec. 31, 2016. Of those patients, 38,408 were opioid naive and 28,914 had taken opioids before. The authors evaluated long-term opioid use – defined as 90 days or more of filled prescriptions – during a 12-month outcome assessment period that varied depending on differences in post-TKR PT start date and duration.
The researchers found a significantly lower likelihood of long-term opioid use associated with receipt of any PT before TKR among patients who had not taken opioids before (adjusted odds ratio [aOR], 0.75; 95% confidence interval, 0.60-0.95) and those who had taken opioids in the past (aOR, 0.75; 95% CI, 0.70-0.80).
Investigators found that 2.2% of participants in the opioid-naive group and 32.5% of those in the opioid-experienced group used opioids long-term after TKR. Approximately 76% of participants overall received outpatient PT within the 90 days after surgery, and the receipt of post-TKR PT at any point was associated with lower odds of long-term opioid use in the opioid-experienced group (aOR, 0.75; 95% CI, 0.70-0.79).
Among the opioid-experienced group, receiving between 6 and 12 PT sessions (aOR, 0.82; 95% CI, 0.75-0.90) or ≥ 13 sessions (aOR, 0.71; 95% CI, 0.65-0.77) were both associated with lower odds of long-term opioid use, compared with those who received 1-5 sessions. Beginning PT 31-60 days or 61-90 days after surgery was associated with greater odds of long-term opioid use across both cohorts, compared with those who initiated therapy within 30 days of TKR.
Physical therapy: Underexplored option for pain in knee replacement
One finding caught the researchers slightly off guard: There was no association between active physical therapy and reduced odds of long-term opioid use. “From prior studies, at least in people with knee osteoarthritis, we know that active interventions were more useful than passive interventions,” Dr. Kumar said.
That said, he added that there is still some professional uncertainty regarding “the right type or the right components of physical therapy for managing pain in this population.” Regardless, he believes their study emphasizes the benefits of PT as a pain alleviator in these patients, especially those who have previously used opioids.
“Pharmaceuticals have side effects. Injections are not super effective,” he said. “The idea behind focusing on physical therapy interventions is that it’s widely available, it does you no harm, and it could potentially be lower cost to both the payers and the providers.”
The authors acknowledged their study’s limitations, including not adjusting for opioid use within the 90 days after surgery as well as the different outcome assessment periods for pre-TKR and post-TKR PT exposures. In addition, they admitted that some of the patients who received PT could have been among those less likely to be treated with opioids, and vice versa. “A randomized clinical trial,” they wrote, “would be required to disentangle these issues.”
The study was supported by grants from the National Institutes of Health and the National Institute of Arthritis and Musculoskeletal and Skin Diseases. Dr. Kumar reported receiving grants from the National Institutes of Health during the conduct of the study and grants from Pfizer for unrelated projects outside the submitted work. The full list of author disclosures can be found with the original article.
A version of this article first appeared on Medscape.com.
A new study has found that physical therapy may lead to a reduced risk of long-term opioid use in patients who have undergone total knee replacement (TKR).
“Greater number of PT intervention sessions and earlier initiation of outpatient PT care after TKR were associated with lower odds of long-term opioid use,” authors from Boston University wrote in their report on the study, which was published online Oct. 27 in JAMA Network Open.
“In previous large studies, we’ve seen that physical therapy can reduce pain in people with knee osteoarthritis, which is usually the primary indication for TKR,” study coauthor Deepak Kumar, PT, PhD, said in an interview. “But the association of physical therapy with opioid use in people with knee replacement has not yet been explored.
“The reason we focused on opioid use in these patients is because the number of knee replacement surgeries is going up exponentially,” Dr. Kumar said. “And, depending on which data you look at, from one-third to up to half of people who undergo knee replacement and have used opioids before end up becoming long-term users. Even in people who have not used them before, 5%-8% become long-term users after the surgery.
“Given how many surgeries are happening – and that number is expected to keep going up – the number of people who are becoming long-term opioid users is not trivial,” he said.
Study details
To assess the value of PT in reducing opioid use in this subset of patients, the authors reviewed records from the OptumLabs Data Warehouse insurance claims database to identify 67,322 eligible participants aged 40 or older who underwent TKR from Jan. 1, 2001, to Dec. 31, 2016. Of those patients, 38,408 were opioid naive and 28,914 had taken opioids before. The authors evaluated long-term opioid use – defined as 90 days or more of filled prescriptions – during a 12-month outcome assessment period that varied depending on differences in post-TKR PT start date and duration.
The researchers found a significantly lower likelihood of long-term opioid use associated with receipt of any PT before TKR among patients who had not taken opioids before (adjusted odds ratio [aOR], 0.75; 95% confidence interval, 0.60-0.95) and those who had taken opioids in the past (aOR, 0.75; 95% CI, 0.70-0.80).
Investigators found that 2.2% of participants in the opioid-naive group and 32.5% of those in the opioid-experienced group used opioids long-term after TKR. Approximately 76% of participants overall received outpatient PT within the 90 days after surgery, and the receipt of post-TKR PT at any point was associated with lower odds of long-term opioid use in the opioid-experienced group (aOR, 0.75; 95% CI, 0.70-0.79).
Among the opioid-experienced group, receiving between 6 and 12 PT sessions (aOR, 0.82; 95% CI, 0.75-0.90) or ≥ 13 sessions (aOR, 0.71; 95% CI, 0.65-0.77) were both associated with lower odds of long-term opioid use, compared with those who received 1-5 sessions. Beginning PT 31-60 days or 61-90 days after surgery was associated with greater odds of long-term opioid use across both cohorts, compared with those who initiated therapy within 30 days of TKR.
Physical therapy: Underexplored option for pain in knee replacement
One finding caught the researchers slightly off guard: There was no association between active physical therapy and reduced odds of long-term opioid use. “From prior studies, at least in people with knee osteoarthritis, we know that active interventions were more useful than passive interventions,” Dr. Kumar said.
That said, he added that there is still some professional uncertainty regarding “the right type or the right components of physical therapy for managing pain in this population.” Regardless, he believes their study emphasizes the benefits of PT as a pain alleviator in these patients, especially those who have previously used opioids.
“Pharmaceuticals have side effects. Injections are not super effective,” he said. “The idea behind focusing on physical therapy interventions is that it’s widely available, it does you no harm, and it could potentially be lower cost to both the payers and the providers.”
The authors acknowledged their study’s limitations, including not adjusting for opioid use within the 90 days after surgery as well as the different outcome assessment periods for pre-TKR and post-TKR PT exposures. In addition, they admitted that some of the patients who received PT could have been among those less likely to be treated with opioids, and vice versa. “A randomized clinical trial,” they wrote, “would be required to disentangle these issues.”
The study was supported by grants from the National Institutes of Health and the National Institute of Arthritis and Musculoskeletal and Skin Diseases. Dr. Kumar reported receiving grants from the National Institutes of Health during the conduct of the study and grants from Pfizer for unrelated projects outside the submitted work. The full list of author disclosures can be found with the original article.
A version of this article first appeared on Medscape.com.
A new study has found that physical therapy may lead to a reduced risk of long-term opioid use in patients who have undergone total knee replacement (TKR).
“Greater number of PT intervention sessions and earlier initiation of outpatient PT care after TKR were associated with lower odds of long-term opioid use,” authors from Boston University wrote in their report on the study, which was published online Oct. 27 in JAMA Network Open.
“In previous large studies, we’ve seen that physical therapy can reduce pain in people with knee osteoarthritis, which is usually the primary indication for TKR,” study coauthor Deepak Kumar, PT, PhD, said in an interview. “But the association of physical therapy with opioid use in people with knee replacement has not yet been explored.
“The reason we focused on opioid use in these patients is because the number of knee replacement surgeries is going up exponentially,” Dr. Kumar said. “And, depending on which data you look at, from one-third to up to half of people who undergo knee replacement and have used opioids before end up becoming long-term users. Even in people who have not used them before, 5%-8% become long-term users after the surgery.
“Given how many surgeries are happening – and that number is expected to keep going up – the number of people who are becoming long-term opioid users is not trivial,” he said.
Study details
To assess the value of PT in reducing opioid use in this subset of patients, the authors reviewed records from the OptumLabs Data Warehouse insurance claims database to identify 67,322 eligible participants aged 40 or older who underwent TKR from Jan. 1, 2001, to Dec. 31, 2016. Of those patients, 38,408 were opioid naive and 28,914 had taken opioids before. The authors evaluated long-term opioid use – defined as 90 days or more of filled prescriptions – during a 12-month outcome assessment period that varied depending on differences in post-TKR PT start date and duration.
The researchers found a significantly lower likelihood of long-term opioid use associated with receipt of any PT before TKR among patients who had not taken opioids before (adjusted odds ratio [aOR], 0.75; 95% confidence interval, 0.60-0.95) and those who had taken opioids in the past (aOR, 0.75; 95% CI, 0.70-0.80).
Investigators found that 2.2% of participants in the opioid-naive group and 32.5% of those in the opioid-experienced group used opioids long-term after TKR. Approximately 76% of participants overall received outpatient PT within the 90 days after surgery, and the receipt of post-TKR PT at any point was associated with lower odds of long-term opioid use in the opioid-experienced group (aOR, 0.75; 95% CI, 0.70-0.79).
Among the opioid-experienced group, receiving between 6 and 12 PT sessions (aOR, 0.82; 95% CI, 0.75-0.90) or ≥ 13 sessions (aOR, 0.71; 95% CI, 0.65-0.77) were both associated with lower odds of long-term opioid use, compared with those who received 1-5 sessions. Beginning PT 31-60 days or 61-90 days after surgery was associated with greater odds of long-term opioid use across both cohorts, compared with those who initiated therapy within 30 days of TKR.
Physical therapy: Underexplored option for pain in knee replacement
One finding caught the researchers slightly off guard: There was no association between active physical therapy and reduced odds of long-term opioid use. “From prior studies, at least in people with knee osteoarthritis, we know that active interventions were more useful than passive interventions,” Dr. Kumar said.
That said, he added that there is still some professional uncertainty regarding “the right type or the right components of physical therapy for managing pain in this population.” Regardless, he believes their study emphasizes the benefits of PT as a pain alleviator in these patients, especially those who have previously used opioids.
“Pharmaceuticals have side effects. Injections are not super effective,” he said. “The idea behind focusing on physical therapy interventions is that it’s widely available, it does you no harm, and it could potentially be lower cost to both the payers and the providers.”
The authors acknowledged their study’s limitations, including not adjusting for opioid use within the 90 days after surgery as well as the different outcome assessment periods for pre-TKR and post-TKR PT exposures. In addition, they admitted that some of the patients who received PT could have been among those less likely to be treated with opioids, and vice versa. “A randomized clinical trial,” they wrote, “would be required to disentangle these issues.”
The study was supported by grants from the National Institutes of Health and the National Institute of Arthritis and Musculoskeletal and Skin Diseases. Dr. Kumar reported receiving grants from the National Institutes of Health during the conduct of the study and grants from Pfizer for unrelated projects outside the submitted work. The full list of author disclosures can be found with the original article.
A version of this article first appeared on Medscape.com.
PRP injections don’t top placebo for ankle osteoarthritis
Platelet-rich plasma (PRP) injections did not significantly improve pain or function when compared with placebo injections in patients with ankle osteoarthritis (OA), a new study has found.
“Previous evidence for PRP injections in ankle osteoarthritis was limited to 4 small case series with methodological flaws,” wrote Liam D. A. Paget, MD, of the University of Amsterdam, and coauthors. The study was published online Oct. 26 in JAMA.
To assess the value of PRP injections as a treatment for ankle OA, the researchers launched a double-blind, randomized clinical trial of Dutch patients with notable ankle pain and tibiotalar joint space narrowing. From six sites in the Netherlands, 100 patients (45% women, mean age 56 years) were split into two groups: one that received two intra-articular injections of PRP 6 weeks apart (n = 48) and one that received two injections of saline placebo (n = 52).
At baseline, mean American Orthopaedic Foot and Ankle Society (AOFAS) scores were 63 in the PRP group and 64 in the placebo group (range 0-100, with higher scores indicating less pain and more function). At 26-week follow-up, the mean AOFAS score improved by 10 points in the PRP group (95% confidence interval, 6-14; P < .001) and by 11 points in the placebo group (95% CI, 7-15; P < .001). The adjusted between-group difference for AOFAS improvement over 26 weeks was –1 point (95% CI, –6 to 3; P = .56).
There was one serious adverse event in the placebo group – a transient ischemic attack 3 weeks after the first injection – but it was deemed unrelated.
Searching for answers regarding PRP and osteoarthritis
“From my standpoint, this study is a great step forward to where the field needs to be, which is honing in on longer-term studies that are standardizing PRP and teasing out its effects,” Prathap Jayaram, MD, director of regenerative sports medicine at the Baylor College of Medicine in Houston, said in an interview.
He highlighted the authors’ acknowledgment of previous studies in which PRP injections appeared effective in treating knee OA, including their statement that the “results reported here for ankle osteoarthritis were not consistent with these potentially beneficial effects in knee osteoarthritis.”
“They’re acknowledging that this does have some benefit in knees,” he said. “Could that translate toward the ankle?”
“PRP did lead to an improvement,” he added. “There just wasn’t a big enough difference to say one was superior to the other.”
Citing his team’s recent preclinical study that was published in Osteoarthritis and Cartilage, Dr. Jayaram emphasized the possibility that PRP could have much-needed disease-modifying effects in osteoarthritis. More work is needed to pin down the details.
“We need more mechanistic studies to be done so we can actually identify the therapeutic properties in PRP and leverage them to track reproducible outcomes,” he said, adding that “simply put, your blood and my blood might be different. There is going to be heterogeneity there. The analogy I give my patients is, when they take an antibiotic, we have a specific dose, a specific drug, and a specific duration. It’s very standardized. We’re just not there yet with PRP.”
The authors acknowledged their study’s limitations, including a likely inability to generalize their results to other platelet-rich blood products as well as a lack of composition analysis of the PRP they used. That said, they added that this particular PRP has been “analyzed previously” for another trial and noted that such analysis is not typically performed in a clinical setting.
The study was supported by a grant from the Dutch Arthritis Society. Its authors reported several potential conflicts of interest, including receiving their own grants from the Dutch Arthritis Society and other organizations, as well as accepting loaned Hettich centrifuges from a medical device company for the study.
A version of this article first appeared on Medscape.com.
Platelet-rich plasma (PRP) injections did not significantly improve pain or function when compared with placebo injections in patients with ankle osteoarthritis (OA), a new study has found.
“Previous evidence for PRP injections in ankle osteoarthritis was limited to 4 small case series with methodological flaws,” wrote Liam D. A. Paget, MD, of the University of Amsterdam, and coauthors. The study was published online Oct. 26 in JAMA.
To assess the value of PRP injections as a treatment for ankle OA, the researchers launched a double-blind, randomized clinical trial of Dutch patients with notable ankle pain and tibiotalar joint space narrowing. From six sites in the Netherlands, 100 patients (45% women, mean age 56 years) were split into two groups: one that received two intra-articular injections of PRP 6 weeks apart (n = 48) and one that received two injections of saline placebo (n = 52).
At baseline, mean American Orthopaedic Foot and Ankle Society (AOFAS) scores were 63 in the PRP group and 64 in the placebo group (range 0-100, with higher scores indicating less pain and more function). At 26-week follow-up, the mean AOFAS score improved by 10 points in the PRP group (95% confidence interval, 6-14; P < .001) and by 11 points in the placebo group (95% CI, 7-15; P < .001). The adjusted between-group difference for AOFAS improvement over 26 weeks was –1 point (95% CI, –6 to 3; P = .56).
There was one serious adverse event in the placebo group – a transient ischemic attack 3 weeks after the first injection – but it was deemed unrelated.
Searching for answers regarding PRP and osteoarthritis
“From my standpoint, this study is a great step forward to where the field needs to be, which is honing in on longer-term studies that are standardizing PRP and teasing out its effects,” Prathap Jayaram, MD, director of regenerative sports medicine at the Baylor College of Medicine in Houston, said in an interview.
He highlighted the authors’ acknowledgment of previous studies in which PRP injections appeared effective in treating knee OA, including their statement that the “results reported here for ankle osteoarthritis were not consistent with these potentially beneficial effects in knee osteoarthritis.”
“They’re acknowledging that this does have some benefit in knees,” he said. “Could that translate toward the ankle?”
“PRP did lead to an improvement,” he added. “There just wasn’t a big enough difference to say one was superior to the other.”
Citing his team’s recent preclinical study that was published in Osteoarthritis and Cartilage, Dr. Jayaram emphasized the possibility that PRP could have much-needed disease-modifying effects in osteoarthritis. More work is needed to pin down the details.
“We need more mechanistic studies to be done so we can actually identify the therapeutic properties in PRP and leverage them to track reproducible outcomes,” he said, adding that “simply put, your blood and my blood might be different. There is going to be heterogeneity there. The analogy I give my patients is, when they take an antibiotic, we have a specific dose, a specific drug, and a specific duration. It’s very standardized. We’re just not there yet with PRP.”
The authors acknowledged their study’s limitations, including a likely inability to generalize their results to other platelet-rich blood products as well as a lack of composition analysis of the PRP they used. That said, they added that this particular PRP has been “analyzed previously” for another trial and noted that such analysis is not typically performed in a clinical setting.
The study was supported by a grant from the Dutch Arthritis Society. Its authors reported several potential conflicts of interest, including receiving their own grants from the Dutch Arthritis Society and other organizations, as well as accepting loaned Hettich centrifuges from a medical device company for the study.
A version of this article first appeared on Medscape.com.
Platelet-rich plasma (PRP) injections did not significantly improve pain or function when compared with placebo injections in patients with ankle osteoarthritis (OA), a new study has found.
“Previous evidence for PRP injections in ankle osteoarthritis was limited to 4 small case series with methodological flaws,” wrote Liam D. A. Paget, MD, of the University of Amsterdam, and coauthors. The study was published online Oct. 26 in JAMA.
To assess the value of PRP injections as a treatment for ankle OA, the researchers launched a double-blind, randomized clinical trial of Dutch patients with notable ankle pain and tibiotalar joint space narrowing. From six sites in the Netherlands, 100 patients (45% women, mean age 56 years) were split into two groups: one that received two intra-articular injections of PRP 6 weeks apart (n = 48) and one that received two injections of saline placebo (n = 52).
At baseline, mean American Orthopaedic Foot and Ankle Society (AOFAS) scores were 63 in the PRP group and 64 in the placebo group (range 0-100, with higher scores indicating less pain and more function). At 26-week follow-up, the mean AOFAS score improved by 10 points in the PRP group (95% confidence interval, 6-14; P < .001) and by 11 points in the placebo group (95% CI, 7-15; P < .001). The adjusted between-group difference for AOFAS improvement over 26 weeks was –1 point (95% CI, –6 to 3; P = .56).
There was one serious adverse event in the placebo group – a transient ischemic attack 3 weeks after the first injection – but it was deemed unrelated.
Searching for answers regarding PRP and osteoarthritis
“From my standpoint, this study is a great step forward to where the field needs to be, which is honing in on longer-term studies that are standardizing PRP and teasing out its effects,” Prathap Jayaram, MD, director of regenerative sports medicine at the Baylor College of Medicine in Houston, said in an interview.
He highlighted the authors’ acknowledgment of previous studies in which PRP injections appeared effective in treating knee OA, including their statement that the “results reported here for ankle osteoarthritis were not consistent with these potentially beneficial effects in knee osteoarthritis.”
“They’re acknowledging that this does have some benefit in knees,” he said. “Could that translate toward the ankle?”
“PRP did lead to an improvement,” he added. “There just wasn’t a big enough difference to say one was superior to the other.”
Citing his team’s recent preclinical study that was published in Osteoarthritis and Cartilage, Dr. Jayaram emphasized the possibility that PRP could have much-needed disease-modifying effects in osteoarthritis. More work is needed to pin down the details.
“We need more mechanistic studies to be done so we can actually identify the therapeutic properties in PRP and leverage them to track reproducible outcomes,” he said, adding that “simply put, your blood and my blood might be different. There is going to be heterogeneity there. The analogy I give my patients is, when they take an antibiotic, we have a specific dose, a specific drug, and a specific duration. It’s very standardized. We’re just not there yet with PRP.”
The authors acknowledged their study’s limitations, including a likely inability to generalize their results to other platelet-rich blood products as well as a lack of composition analysis of the PRP they used. That said, they added that this particular PRP has been “analyzed previously” for another trial and noted that such analysis is not typically performed in a clinical setting.
The study was supported by a grant from the Dutch Arthritis Society. Its authors reported several potential conflicts of interest, including receiving their own grants from the Dutch Arthritis Society and other organizations, as well as accepting loaned Hettich centrifuges from a medical device company for the study.
A version of this article first appeared on Medscape.com.
MDs doing wrong-site surgery: Why is it still happening?
In July 2021, University Hospitals, in Cleveland, announced that its staff had transplanted a kidney into the wrong patient. Although the patient who received the kidney was recovering well, the patient who was supposed to have received the kidney was skipped over. As a result of the error, two employees were placed on administrative leave and the incident was being investigated, the hospital announced.
In April 2020, an interventional radiologist at Boca Raton Regional Hospital, in Boca Raton, Fla., was sued for allegedly placing a stent into the wrong kidney of an 80-year-old patient. Using fluoroscopic guidance, the doctor removed an old stent from the right side but incorrectly replaced it with a new stent on the left side, according to an interview conducted by this news organization with the patient’s lawyers at Searcy Law, in West Palm Beach.
“The problem is that it is so rare that doctors don’t focus on it,” says Mary R. Kwaan, MD, a colorectal surgeon at UCLA Medical Center, Los Angeles.
A 2006 study in which Kwaan was the lead author concluded that there was one wrong-site surgery for every 112,994 surgeries. Those mistakes can add up. A 2006 study estimated that 25 to 52 wrong-site surgeries were performed each week in the United States.
“Many surgeons don’t think it can happen to them, so they don’t take extra precautions,” says David Mayer, MD, executive director of the MedStar Institute for Quality and Safety, in Washington, DC. “When they make a wrong-site error, usually the first thing they say is, ‘I never thought this would happen to me,’ ” he says.
Wrong-site surgeries are considered sentinel events -- the worst kinds of medical errors. The Sullivan Group, a patient safety consultancy based in Colorado, reports that in 2013, 2.7% of patients who were involved in wrong-site surgeries died and 41% experienced some type of permanent injury. The mean malpractice payment was $127,000.
Some malpractice payments are much higher. In 2013, a Maryland ob.gyn paid a $1.42 million malpractice award for removing the wrong ovary from a woman in 2009. In 2017, a Pennsylvania urologist paid $870,000 for removing the wrong testicle from a man in 2013.
Wrong-site surgery often involves experienced surgeons
One might think that wrong-site surgeries usually involve younger or less-experienced surgeons, but that’s not the case; two thirds of the surgeons who perform wrong-site surgeries are in their 40s and 50s, compared with fewer than 25% younger than 40.
In a rather chilling statistic, in a 2013 survey, 12.4% of doctors who were involved in sentinel events in general had claims for more than one event.
These errors are more common in certain specialties. In a study reported in the Journal of Neurology, Neurosurgery and Spine, 25% of orthopedic surgeons reported performing at least one wrong-site surgery during their career.
Within orthopedics, spine surgery is ground zero for wrong-site surgery. “Finding the site in spine surgery can be more difficult than in common left-right orthopedic procedures,” says Joseph A. Bosco III, a New York City orthopedist.
A 2007 study found that 25% of neurosurgeons had performed wrong-site surgeries. In Missouri in 2013, for example, a 53-year-old patient who was scheduled to undergo a left-sided craniotomy bypass allegedly underwent a right-sided craniotomy and was unable to speak after surgery.
Wrong-site surgeries are also performed by general surgeons, urologists, cardiologists, otolaryngologists, and ophthalmologists. A 2021 lawsuit accused a Tampa urologist of removing the patient’s wrong testicle. And a 2019 lawsuit accused a Chicago ophthalmologist of operating on the wrong eye to remove a cyst.
It’s not just the surgeon’s mistake
Mistakes are not only made by the surgeon in the operating room (OR). They can be made by staff when scheduling a surgery, radiologists and pathologists when writing their reports for surgery, and by team members in the OR.
Many people are prone to confusing left and right. A 2020 study found that 14.9% of people had difficulty distinguishing left from right; other studies have shown higher rates. Distractions increase the likelihood of mistakes. In a 2015 study, background noise in a hospital ward made it more difficult for medical students to make left-right judgments.
OR personnel can be confused when patients are turned around. “To operate on the back of someone’s leg, the surgeon may turn the patient from supine to prone, and so left becomes right,” says Samuel C. Seiden, MD, an anesthesiologist in Roseville, Calif., who has studied wrong-site surgery.
Operative site markings that are drawn on the skin can be rubbed off when surgical prep is applied, and markings aren’t usually possible for procedures such as spine surgeries. Surgical draping can make it harder to distinguish the patient’s left and right, and a busy surgeon relying on memory may confuse cases and perform wrong-patient surgery.
A push to eliminate wrong-site surgery
In 2004, the Joint Commission, which accredits hospitals and many surgery centers, decided to do something about wrong-site surgery and related surgical errors. It released a universal protocol, which requires hospitals to take three steps to prevent errors: perform preoperative verification that is based on patient care documents; mark the operative site; and take a time-out just before surgery, during which the team should consider whether a mistake is about to be made.
Two years after the Joint Commission published its protocol, Dr. Seiden led a study to determine what effect it had had. The investigators found that wrong-site cases had decreased by only about one third. Preventing wrong-site surgery “turns out to be more complicated to eradicate than anybody thought,” Mark Chassin, MD, president of the Joint Commission, stated a few years later.
Why did the protocol have only a limited effect? Dr. Seiden says that it has been hard to change doctors’ traditional attitudes against standardization. “Some have had an attitude that checklists are for dummies, but that is changing,” he says.
For instance, some surgical teams were not paying attention during time-outs. “The time-out should be like the invocation of the National Anthem,” an orthopedic surgeon from Iowa wrote. “All other activities should stop.”
Even had surgeons followed the universal protocol, about one third of wrong-site surgeries would not have been identified, according to Dr. Kwaan’s study, which was published in the same year as Dr. Seiden’s. As an example, when the wrong kidney was removed at Methodist Hospital, in St. Louis Park, Minn., the hospital said it was following a protocol set by the Minnesota Hospital Association.
Redoubling efforts
In 2009, the Joint Commission decided to take another tack. It encouraged hospitals to make root-cause analyses not only of wrong-site surgeries but also of near misses, which are much more plentiful. It used the insights gained to change surgical routines and protocols.
The Safe Surgery Project, a collaboration between the Joint Commission’s Center for Transforming Healthcare and eight hospitals and surgery centers, reduced the number of errors and near misses by 46% in the scheduling area, 63% in pre-op, and 51% in the OR area.
From that project, the center developed the Targeted Solutions Tool, which basically uses the same methodology that the project used. The center told this news organization that 79 healthcare organizations have used the tool and have reduced the number of errors and near misses by 56% in scheduling, 24% in pre-op, and 48% in the OR.
For this approach to work, however, surgical teams must report their errors to the hospital, which had not been done before. A 2008 study by the Office of the Inspector General of the U.S. Department of Health and Human Services found that surgical staff did not report 86% of adverse events to their hospitals. Reasons given included lack of time, fear of punitive action, and skepticism that reporting would do any good.
Unlike some other adverse events, it’s hard to keep wrong-site surgeries secret from patients, because they can usually see the scars from it, but some surgeons invent ways to cover it up from patients, too, Dr. Mayer says. One wrong-side hernia repair was corrected in mid operation. Afterward, the surgeon told the patient that he had found another hernia on the other side and had fixed that one, too.
Changing the culture
Reformers argue that wrong-site surgeries can be prevented by changing the culture of the hospital or surgery center. “We have to think of wrong-site surgeries as a failure of the system, not of the individual,” says Ron Savrin, MD, a general surgeon in Chagrin Falls, Ohio, who is a surgery subject matter expert for the Sullivan Group. “It should never be only up to one individual to stop an error from occurring.”
Seeing oneself as part of a team can reduce errors. Although other people can introduce errors that make a person look bad, they can also stop the errors that might otherwise have occurred. Punishing individuals for making errors does little good in stopping errors.
“It’s human nature to want to punish somebody for making a mistake, and it’s hard to change that mentality,” Dr. Savrin says. He recalls that when he was a resident, “the morbidity and mortality conferences could be very difficult for anyone who made a mistake, but I think that attitude is changing.”
Studies have found wide variation in the number of wrong-site surgeries among hospitals. A recent Pennsylvania study found an average of one wrong-site surgery or near miss per hospital per year, but about one third of hospitals did not report any.
Wrong-site surgeries are often concentrated in certain hospitals -- even prestigious teaching hospitals are not immune. A decade ago, Rhode Island Hospital had five wrong-site surgeries in 2 years, and Boston’s Beth Israel Deaconess Medical Center had three wrong-spine surgeries within 2 months.
Other ways to reduce errors
Dr. Seiden thinks reform efforts should take a page from his own specialty. Anesthesiology has developed a variety of forcing functions, which are simple changes in technology that can stop errors. An example is the use of a valve that will not deliver a drug unless certain steps are followed.
The StartBox System, a new way to prevent surgical errors, delivers the surgery blade only after all safety information has been provided. Tested by 11 orthopedic surgeons performing 487 procedures, the system identified 17 near misses.
Another approach is to film time-outs so as to enforce compliance with protocols and help with root-cause analyses. NYU-Langone Medical Center, in New York City, not only films the time-out but also grades OR teams on compliance, says Dr. Bosco, who is vice chair of clinical affairs in the department of orthopedic surgery at the hospital.
In addition, more states are requiring hospitals to report adverse events, including wrong-site surgeries. According to the National Academy for State Health Policy, 28 states require the reporting of adverse events. However, only six states identify facilities in public reports; 16 states publish only aggregate data; and five states do not report error data to the public.
The goal is zero errors
Are there fewer wrong-site surgeries now? “My sense is that surgeons, hospitals, and surgery centers are taking wrong-site errors more seriously,” Dr. Savrin says.
Because reported information is spotty and no major studies on incidence have been conducted in recent years, “we don’t have a clear idea,” he says, “but my best guess is that the rate is declining.
“Absolute zero preventable errors has to be our goal,” Dr. Savrin says “We might not get there, but we can’t stop trying.”
A version of this article first appeared on Medscape.com.
In July 2021, University Hospitals, in Cleveland, announced that its staff had transplanted a kidney into the wrong patient. Although the patient who received the kidney was recovering well, the patient who was supposed to have received the kidney was skipped over. As a result of the error, two employees were placed on administrative leave and the incident was being investigated, the hospital announced.
In April 2020, an interventional radiologist at Boca Raton Regional Hospital, in Boca Raton, Fla., was sued for allegedly placing a stent into the wrong kidney of an 80-year-old patient. Using fluoroscopic guidance, the doctor removed an old stent from the right side but incorrectly replaced it with a new stent on the left side, according to an interview conducted by this news organization with the patient’s lawyers at Searcy Law, in West Palm Beach.
“The problem is that it is so rare that doctors don’t focus on it,” says Mary R. Kwaan, MD, a colorectal surgeon at UCLA Medical Center, Los Angeles.
A 2006 study in which Kwaan was the lead author concluded that there was one wrong-site surgery for every 112,994 surgeries. Those mistakes can add up. A 2006 study estimated that 25 to 52 wrong-site surgeries were performed each week in the United States.
“Many surgeons don’t think it can happen to them, so they don’t take extra precautions,” says David Mayer, MD, executive director of the MedStar Institute for Quality and Safety, in Washington, DC. “When they make a wrong-site error, usually the first thing they say is, ‘I never thought this would happen to me,’ ” he says.
Wrong-site surgeries are considered sentinel events -- the worst kinds of medical errors. The Sullivan Group, a patient safety consultancy based in Colorado, reports that in 2013, 2.7% of patients who were involved in wrong-site surgeries died and 41% experienced some type of permanent injury. The mean malpractice payment was $127,000.
Some malpractice payments are much higher. In 2013, a Maryland ob.gyn paid a $1.42 million malpractice award for removing the wrong ovary from a woman in 2009. In 2017, a Pennsylvania urologist paid $870,000 for removing the wrong testicle from a man in 2013.
Wrong-site surgery often involves experienced surgeons
One might think that wrong-site surgeries usually involve younger or less-experienced surgeons, but that’s not the case; two thirds of the surgeons who perform wrong-site surgeries are in their 40s and 50s, compared with fewer than 25% younger than 40.
In a rather chilling statistic, in a 2013 survey, 12.4% of doctors who were involved in sentinel events in general had claims for more than one event.
These errors are more common in certain specialties. In a study reported in the Journal of Neurology, Neurosurgery and Spine, 25% of orthopedic surgeons reported performing at least one wrong-site surgery during their career.
Within orthopedics, spine surgery is ground zero for wrong-site surgery. “Finding the site in spine surgery can be more difficult than in common left-right orthopedic procedures,” says Joseph A. Bosco III, a New York City orthopedist.
A 2007 study found that 25% of neurosurgeons had performed wrong-site surgeries. In Missouri in 2013, for example, a 53-year-old patient who was scheduled to undergo a left-sided craniotomy bypass allegedly underwent a right-sided craniotomy and was unable to speak after surgery.
Wrong-site surgeries are also performed by general surgeons, urologists, cardiologists, otolaryngologists, and ophthalmologists. A 2021 lawsuit accused a Tampa urologist of removing the patient’s wrong testicle. And a 2019 lawsuit accused a Chicago ophthalmologist of operating on the wrong eye to remove a cyst.
It’s not just the surgeon’s mistake
Mistakes are not only made by the surgeon in the operating room (OR). They can be made by staff when scheduling a surgery, radiologists and pathologists when writing their reports for surgery, and by team members in the OR.
Many people are prone to confusing left and right. A 2020 study found that 14.9% of people had difficulty distinguishing left from right; other studies have shown higher rates. Distractions increase the likelihood of mistakes. In a 2015 study, background noise in a hospital ward made it more difficult for medical students to make left-right judgments.
OR personnel can be confused when patients are turned around. “To operate on the back of someone’s leg, the surgeon may turn the patient from supine to prone, and so left becomes right,” says Samuel C. Seiden, MD, an anesthesiologist in Roseville, Calif., who has studied wrong-site surgery.
Operative site markings that are drawn on the skin can be rubbed off when surgical prep is applied, and markings aren’t usually possible for procedures such as spine surgeries. Surgical draping can make it harder to distinguish the patient’s left and right, and a busy surgeon relying on memory may confuse cases and perform wrong-patient surgery.
A push to eliminate wrong-site surgery
In 2004, the Joint Commission, which accredits hospitals and many surgery centers, decided to do something about wrong-site surgery and related surgical errors. It released a universal protocol, which requires hospitals to take three steps to prevent errors: perform preoperative verification that is based on patient care documents; mark the operative site; and take a time-out just before surgery, during which the team should consider whether a mistake is about to be made.
Two years after the Joint Commission published its protocol, Dr. Seiden led a study to determine what effect it had had. The investigators found that wrong-site cases had decreased by only about one third. Preventing wrong-site surgery “turns out to be more complicated to eradicate than anybody thought,” Mark Chassin, MD, president of the Joint Commission, stated a few years later.
Why did the protocol have only a limited effect? Dr. Seiden says that it has been hard to change doctors’ traditional attitudes against standardization. “Some have had an attitude that checklists are for dummies, but that is changing,” he says.
For instance, some surgical teams were not paying attention during time-outs. “The time-out should be like the invocation of the National Anthem,” an orthopedic surgeon from Iowa wrote. “All other activities should stop.”
Even had surgeons followed the universal protocol, about one third of wrong-site surgeries would not have been identified, according to Dr. Kwaan’s study, which was published in the same year as Dr. Seiden’s. As an example, when the wrong kidney was removed at Methodist Hospital, in St. Louis Park, Minn., the hospital said it was following a protocol set by the Minnesota Hospital Association.
Redoubling efforts
In 2009, the Joint Commission decided to take another tack. It encouraged hospitals to make root-cause analyses not only of wrong-site surgeries but also of near misses, which are much more plentiful. It used the insights gained to change surgical routines and protocols.
The Safe Surgery Project, a collaboration between the Joint Commission’s Center for Transforming Healthcare and eight hospitals and surgery centers, reduced the number of errors and near misses by 46% in the scheduling area, 63% in pre-op, and 51% in the OR area.
From that project, the center developed the Targeted Solutions Tool, which basically uses the same methodology that the project used. The center told this news organization that 79 healthcare organizations have used the tool and have reduced the number of errors and near misses by 56% in scheduling, 24% in pre-op, and 48% in the OR.
For this approach to work, however, surgical teams must report their errors to the hospital, which had not been done before. A 2008 study by the Office of the Inspector General of the U.S. Department of Health and Human Services found that surgical staff did not report 86% of adverse events to their hospitals. Reasons given included lack of time, fear of punitive action, and skepticism that reporting would do any good.
Unlike some other adverse events, it’s hard to keep wrong-site surgeries secret from patients, because they can usually see the scars from it, but some surgeons invent ways to cover it up from patients, too, Dr. Mayer says. One wrong-side hernia repair was corrected in mid operation. Afterward, the surgeon told the patient that he had found another hernia on the other side and had fixed that one, too.
Changing the culture
Reformers argue that wrong-site surgeries can be prevented by changing the culture of the hospital or surgery center. “We have to think of wrong-site surgeries as a failure of the system, not of the individual,” says Ron Savrin, MD, a general surgeon in Chagrin Falls, Ohio, who is a surgery subject matter expert for the Sullivan Group. “It should never be only up to one individual to stop an error from occurring.”
Seeing oneself as part of a team can reduce errors. Although other people can introduce errors that make a person look bad, they can also stop the errors that might otherwise have occurred. Punishing individuals for making errors does little good in stopping errors.
“It’s human nature to want to punish somebody for making a mistake, and it’s hard to change that mentality,” Dr. Savrin says. He recalls that when he was a resident, “the morbidity and mortality conferences could be very difficult for anyone who made a mistake, but I think that attitude is changing.”
Studies have found wide variation in the number of wrong-site surgeries among hospitals. A recent Pennsylvania study found an average of one wrong-site surgery or near miss per hospital per year, but about one third of hospitals did not report any.
Wrong-site surgeries are often concentrated in certain hospitals -- even prestigious teaching hospitals are not immune. A decade ago, Rhode Island Hospital had five wrong-site surgeries in 2 years, and Boston’s Beth Israel Deaconess Medical Center had three wrong-spine surgeries within 2 months.
Other ways to reduce errors
Dr. Seiden thinks reform efforts should take a page from his own specialty. Anesthesiology has developed a variety of forcing functions, which are simple changes in technology that can stop errors. An example is the use of a valve that will not deliver a drug unless certain steps are followed.
The StartBox System, a new way to prevent surgical errors, delivers the surgery blade only after all safety information has been provided. Tested by 11 orthopedic surgeons performing 487 procedures, the system identified 17 near misses.
Another approach is to film time-outs so as to enforce compliance with protocols and help with root-cause analyses. NYU-Langone Medical Center, in New York City, not only films the time-out but also grades OR teams on compliance, says Dr. Bosco, who is vice chair of clinical affairs in the department of orthopedic surgery at the hospital.
In addition, more states are requiring hospitals to report adverse events, including wrong-site surgeries. According to the National Academy for State Health Policy, 28 states require the reporting of adverse events. However, only six states identify facilities in public reports; 16 states publish only aggregate data; and five states do not report error data to the public.
The goal is zero errors
Are there fewer wrong-site surgeries now? “My sense is that surgeons, hospitals, and surgery centers are taking wrong-site errors more seriously,” Dr. Savrin says.
Because reported information is spotty and no major studies on incidence have been conducted in recent years, “we don’t have a clear idea,” he says, “but my best guess is that the rate is declining.
“Absolute zero preventable errors has to be our goal,” Dr. Savrin says “We might not get there, but we can’t stop trying.”
A version of this article first appeared on Medscape.com.
In July 2021, University Hospitals, in Cleveland, announced that its staff had transplanted a kidney into the wrong patient. Although the patient who received the kidney was recovering well, the patient who was supposed to have received the kidney was skipped over. As a result of the error, two employees were placed on administrative leave and the incident was being investigated, the hospital announced.
In April 2020, an interventional radiologist at Boca Raton Regional Hospital, in Boca Raton, Fla., was sued for allegedly placing a stent into the wrong kidney of an 80-year-old patient. Using fluoroscopic guidance, the doctor removed an old stent from the right side but incorrectly replaced it with a new stent on the left side, according to an interview conducted by this news organization with the patient’s lawyers at Searcy Law, in West Palm Beach.
“The problem is that it is so rare that doctors don’t focus on it,” says Mary R. Kwaan, MD, a colorectal surgeon at UCLA Medical Center, Los Angeles.
A 2006 study in which Kwaan was the lead author concluded that there was one wrong-site surgery for every 112,994 surgeries. Those mistakes can add up. A 2006 study estimated that 25 to 52 wrong-site surgeries were performed each week in the United States.
“Many surgeons don’t think it can happen to them, so they don’t take extra precautions,” says David Mayer, MD, executive director of the MedStar Institute for Quality and Safety, in Washington, DC. “When they make a wrong-site error, usually the first thing they say is, ‘I never thought this would happen to me,’ ” he says.
Wrong-site surgeries are considered sentinel events -- the worst kinds of medical errors. The Sullivan Group, a patient safety consultancy based in Colorado, reports that in 2013, 2.7% of patients who were involved in wrong-site surgeries died and 41% experienced some type of permanent injury. The mean malpractice payment was $127,000.
Some malpractice payments are much higher. In 2013, a Maryland ob.gyn paid a $1.42 million malpractice award for removing the wrong ovary from a woman in 2009. In 2017, a Pennsylvania urologist paid $870,000 for removing the wrong testicle from a man in 2013.
Wrong-site surgery often involves experienced surgeons
One might think that wrong-site surgeries usually involve younger or less-experienced surgeons, but that’s not the case; two thirds of the surgeons who perform wrong-site surgeries are in their 40s and 50s, compared with fewer than 25% younger than 40.
In a rather chilling statistic, in a 2013 survey, 12.4% of doctors who were involved in sentinel events in general had claims for more than one event.
These errors are more common in certain specialties. In a study reported in the Journal of Neurology, Neurosurgery and Spine, 25% of orthopedic surgeons reported performing at least one wrong-site surgery during their career.
Within orthopedics, spine surgery is ground zero for wrong-site surgery. “Finding the site in spine surgery can be more difficult than in common left-right orthopedic procedures,” says Joseph A. Bosco III, a New York City orthopedist.
A 2007 study found that 25% of neurosurgeons had performed wrong-site surgeries. In Missouri in 2013, for example, a 53-year-old patient who was scheduled to undergo a left-sided craniotomy bypass allegedly underwent a right-sided craniotomy and was unable to speak after surgery.
Wrong-site surgeries are also performed by general surgeons, urologists, cardiologists, otolaryngologists, and ophthalmologists. A 2021 lawsuit accused a Tampa urologist of removing the patient’s wrong testicle. And a 2019 lawsuit accused a Chicago ophthalmologist of operating on the wrong eye to remove a cyst.
It’s not just the surgeon’s mistake
Mistakes are not only made by the surgeon in the operating room (OR). They can be made by staff when scheduling a surgery, radiologists and pathologists when writing their reports for surgery, and by team members in the OR.
Many people are prone to confusing left and right. A 2020 study found that 14.9% of people had difficulty distinguishing left from right; other studies have shown higher rates. Distractions increase the likelihood of mistakes. In a 2015 study, background noise in a hospital ward made it more difficult for medical students to make left-right judgments.
OR personnel can be confused when patients are turned around. “To operate on the back of someone’s leg, the surgeon may turn the patient from supine to prone, and so left becomes right,” says Samuel C. Seiden, MD, an anesthesiologist in Roseville, Calif., who has studied wrong-site surgery.
Operative site markings that are drawn on the skin can be rubbed off when surgical prep is applied, and markings aren’t usually possible for procedures such as spine surgeries. Surgical draping can make it harder to distinguish the patient’s left and right, and a busy surgeon relying on memory may confuse cases and perform wrong-patient surgery.
A push to eliminate wrong-site surgery
In 2004, the Joint Commission, which accredits hospitals and many surgery centers, decided to do something about wrong-site surgery and related surgical errors. It released a universal protocol, which requires hospitals to take three steps to prevent errors: perform preoperative verification that is based on patient care documents; mark the operative site; and take a time-out just before surgery, during which the team should consider whether a mistake is about to be made.
Two years after the Joint Commission published its protocol, Dr. Seiden led a study to determine what effect it had had. The investigators found that wrong-site cases had decreased by only about one third. Preventing wrong-site surgery “turns out to be more complicated to eradicate than anybody thought,” Mark Chassin, MD, president of the Joint Commission, stated a few years later.
Why did the protocol have only a limited effect? Dr. Seiden says that it has been hard to change doctors’ traditional attitudes against standardization. “Some have had an attitude that checklists are for dummies, but that is changing,” he says.
For instance, some surgical teams were not paying attention during time-outs. “The time-out should be like the invocation of the National Anthem,” an orthopedic surgeon from Iowa wrote. “All other activities should stop.”
Even had surgeons followed the universal protocol, about one third of wrong-site surgeries would not have been identified, according to Dr. Kwaan’s study, which was published in the same year as Dr. Seiden’s. As an example, when the wrong kidney was removed at Methodist Hospital, in St. Louis Park, Minn., the hospital said it was following a protocol set by the Minnesota Hospital Association.
Redoubling efforts
In 2009, the Joint Commission decided to take another tack. It encouraged hospitals to make root-cause analyses not only of wrong-site surgeries but also of near misses, which are much more plentiful. It used the insights gained to change surgical routines and protocols.
The Safe Surgery Project, a collaboration between the Joint Commission’s Center for Transforming Healthcare and eight hospitals and surgery centers, reduced the number of errors and near misses by 46% in the scheduling area, 63% in pre-op, and 51% in the OR area.
From that project, the center developed the Targeted Solutions Tool, which basically uses the same methodology that the project used. The center told this news organization that 79 healthcare organizations have used the tool and have reduced the number of errors and near misses by 56% in scheduling, 24% in pre-op, and 48% in the OR.
For this approach to work, however, surgical teams must report their errors to the hospital, which had not been done before. A 2008 study by the Office of the Inspector General of the U.S. Department of Health and Human Services found that surgical staff did not report 86% of adverse events to their hospitals. Reasons given included lack of time, fear of punitive action, and skepticism that reporting would do any good.
Unlike some other adverse events, it’s hard to keep wrong-site surgeries secret from patients, because they can usually see the scars from it, but some surgeons invent ways to cover it up from patients, too, Dr. Mayer says. One wrong-side hernia repair was corrected in mid operation. Afterward, the surgeon told the patient that he had found another hernia on the other side and had fixed that one, too.
Changing the culture
Reformers argue that wrong-site surgeries can be prevented by changing the culture of the hospital or surgery center. “We have to think of wrong-site surgeries as a failure of the system, not of the individual,” says Ron Savrin, MD, a general surgeon in Chagrin Falls, Ohio, who is a surgery subject matter expert for the Sullivan Group. “It should never be only up to one individual to stop an error from occurring.”
Seeing oneself as part of a team can reduce errors. Although other people can introduce errors that make a person look bad, they can also stop the errors that might otherwise have occurred. Punishing individuals for making errors does little good in stopping errors.
“It’s human nature to want to punish somebody for making a mistake, and it’s hard to change that mentality,” Dr. Savrin says. He recalls that when he was a resident, “the morbidity and mortality conferences could be very difficult for anyone who made a mistake, but I think that attitude is changing.”
Studies have found wide variation in the number of wrong-site surgeries among hospitals. A recent Pennsylvania study found an average of one wrong-site surgery or near miss per hospital per year, but about one third of hospitals did not report any.
Wrong-site surgeries are often concentrated in certain hospitals -- even prestigious teaching hospitals are not immune. A decade ago, Rhode Island Hospital had five wrong-site surgeries in 2 years, and Boston’s Beth Israel Deaconess Medical Center had three wrong-spine surgeries within 2 months.
Other ways to reduce errors
Dr. Seiden thinks reform efforts should take a page from his own specialty. Anesthesiology has developed a variety of forcing functions, which are simple changes in technology that can stop errors. An example is the use of a valve that will not deliver a drug unless certain steps are followed.
The StartBox System, a new way to prevent surgical errors, delivers the surgery blade only after all safety information has been provided. Tested by 11 orthopedic surgeons performing 487 procedures, the system identified 17 near misses.
Another approach is to film time-outs so as to enforce compliance with protocols and help with root-cause analyses. NYU-Langone Medical Center, in New York City, not only films the time-out but also grades OR teams on compliance, says Dr. Bosco, who is vice chair of clinical affairs in the department of orthopedic surgery at the hospital.
In addition, more states are requiring hospitals to report adverse events, including wrong-site surgeries. According to the National Academy for State Health Policy, 28 states require the reporting of adverse events. However, only six states identify facilities in public reports; 16 states publish only aggregate data; and five states do not report error data to the public.
The goal is zero errors
Are there fewer wrong-site surgeries now? “My sense is that surgeons, hospitals, and surgery centers are taking wrong-site errors more seriously,” Dr. Savrin says.
Because reported information is spotty and no major studies on incidence have been conducted in recent years, “we don’t have a clear idea,” he says, “but my best guess is that the rate is declining.
“Absolute zero preventable errors has to be our goal,” Dr. Savrin says “We might not get there, but we can’t stop trying.”
A version of this article first appeared on Medscape.com.
AAOS updates guidelines for nonoperative knee OA treatment
After nearly a decade, the American Academy of Orthopaedic Surgeons has updated its guidance on nonoperative treatment for knee osteoarthritis.
The clinical practice guidelines, released Sept. 13, 2021, is the third edition of the orthopedic society’s clinical practice recommendations.
According to Robert Brophy, MD, FAAOS, an orthopedic surgeon at Washington University, St. Louis, and cochair of the AAOS clinical practice guideline work group, the AAOS guidelines are a “living document” that needs periodic updating as new research comes to light.
“The methodology for maintaining the AAOS guidelines aims to update guideline documents at least every 10 years,” Dr. Brophy said in an interview. “Since the last edition was from 2013, it was time to provide an updated guideline on this very important topic that affects such a high percentage of our patients and providers.”
The guidelines work group, composed of 12 medical doctors and 1 physical therapist, evaluated the evidence for 29 areas of treatment.
A rating scale based on available evidence and the strength of related medical studies labeled each treatment area as demonstrating strong, moderate, or limited evidence.
Eight treatment modalities weighed in with strong evidence for or against their use: lateral wedge insoles, topical or oral NSAIDs, exercise (supervised or unsupervised), self-management programs, patient education programs, oral acetaminophen, and oral opioids.
According to Dr. Brophy, many of the recommendations assigned a strong evidence base were similar to the prior edition of the guidelines.
Oral medications
NSAIDs and acetaminophen still remain steadfast options for the treatment of knee pain secondary to OA.
The most notable change was that opioids, which have a long history of being used to treat pain, are strongly recommended not to be used for arthritis.
“Reflecting the growing awareness of and emphasis on the opioid epidemic, one of the strongest changes between the current and prior guidelines centers on the use of opioid medications,” Dr. Brophy said. “In the prior guideline, a strong recommendation was made in favor of tramadol with an inconclusive recommendation made regarding other opioid medications. The updated guideline demonstrates clearly the evidence does not support the use of opioid medications – including tramadol – to treat knee osteoarthritis.”
This may require some education for both patients and doctors to buy in that knee pain can be treated adequately with NSAIDs and acetaminophen.
Patients may not understand that anti-inflammatory drugs treat the pain they are experiencing. They may equate an opioid with a “pain pill” and may need education from their doctor that NSAIDs and acetaminophen not only can relieve their pain, but also avoid potential adverse events prior to or after surgery should they progress to knee replacement surgery.
Furthermore, primary care physicians may not be looking at the long-term picture. Solving a short-term pain problem with opioids may limit the medication’s ability to provide pain relief after surgery should a patient develop a tolerance to the medication’s effects.
Recommendations on hip and foot alignment interventions
When it comes to alignment and joint stresses, the knee is sometimes considered the innocent bystander of hip and foot alignment.
Insoles. How the hip and foot align with each can determine the amount of weight that passes through the medial (inner) or lateral (outer) compartment of the knee. To that end, lateral foot insoles have been used in the past for unloading parts of the knee.
Nevertheless, recent evidence has failed to demonstrate a significant benefit for insoles in the setting of OA knee pain, earning the practice a strong recommendation against its use.
High-tibial osteotomy (HTO). The weight-bearing axis of the lower-extremity axis can also be realigned with HTO. The procedure shifts the body’s weight slightly to the opposite side of the knee.
Newer research has led the practice to be downgraded one level in the new guideline, from moderate to limited, despite its widespread use.
It will, however, likely continue to be used as an alternative to total knee replacement in younger patients and to shift weight away from an area of the knee where cartilage is being restored with a concomitant surgical procedure, according to the work group. They noted that additional research studies on the long-term efficacy of the procedure are still needed.
Topical treatments. The guideline authors gave these a strong recommendation. Gels with anti-inflammatory medication have long been available but were prescription only or of considerable cost. Now several affordable over-the-counter options with the same prescription strength can be found in pharmacies and supermarkets.
What makes these medications unique is that they have an NSAID medication in the formulation, which the vast majority of topical treatments found on shelves do not. They also benefit patients who are unable to tolerate oral NSAIDs because of gastrointestinal side effects.
Comparison with 2019 OARSI recommendations
In 2019, the Osteoarthritis Research Society International also published guidelines for the management of OA of the hand, hip, and knee.
Thomas Trojian, MD, a family medicine physician with expertise in sports medicine in York, Pa., and member of both the AAOS and OARSI recommendation committees, noted that the OARSI guidelines are meant to be practical guidelines of stepwise nonoperative treatment.
He said in an interview that “the OARSI guidelines recommend dietary weight management, education, and land-based [exercise] therapy, next topical NSAIDs, then injection therapy.”
Intra-articular steroids and viscosupplementation injection therapy in the form of hyaluronic acid derivatives continue to be a mainstay of treatment for both groups.
The AAOS group notably gave a moderate strength recommendation for intra-articular steroid injections with the caveat that the effects typically only last for 3 months. They also included newer extended-release steroid injections in the recommendation, stating that the evidence moderately suggests they provide more benefit than traditional short-acting steroid injections.
Methodology differs between guidelines
In the areas where the guidelines don’t fully line up, it is important to remember the methodology of each group often drives the guidelines and recommendations.
According to Yale Fillingham, MD, an orthopedic surgeon in group private practice in the greater Philadelphia area and the other cochair of the AAOS guidelines committee, the biggest difference between the AAOS and OARSI guidelines is that, although the OARSI guidelines are also grounded in the literature, the recommendation level was based on voting among panel members.
“The AAOS methodology requires the recommendation and strength of the recommendation to be dictated primarily by the best available evidence in the literature and much less on the expertise and opinion of the voting panel,” Dr. Fillingham said in an interview.
He pointed out that the AAOS voting panel can alter the guideline by adjusting the strength of the recommendation but noted it was only in very clearly defined situations. Therefore, the differences in methodology between the groups make it difficult to directly compare the two guidelines.
Multiple guidelines do, however, point to the importance of the issue. Dr. Fillingham commented: “The numerous organizations that have produced guidelines on the treatment of knee osteoarthritis are a testament to the widespread and profound impact of knee osteoarthritis on our health care system and society.”
As a member of both recommendation groups, Dr. Trojian finds both guidelines reveal the importance of understanding that knee OA is a chronic illness. “There are ways we can manage knee OA and reduce the morbidity. ... The core skills of motivational interviewing are important. Open-ended questions, affirmation, reflection, and summarizing are needed to help patients find and remove roadblocks to promote lifestyle changes.”
Dr. Brophy, Dr. Trojian, and Dr. Fillingham have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
After nearly a decade, the American Academy of Orthopaedic Surgeons has updated its guidance on nonoperative treatment for knee osteoarthritis.
The clinical practice guidelines, released Sept. 13, 2021, is the third edition of the orthopedic society’s clinical practice recommendations.
According to Robert Brophy, MD, FAAOS, an orthopedic surgeon at Washington University, St. Louis, and cochair of the AAOS clinical practice guideline work group, the AAOS guidelines are a “living document” that needs periodic updating as new research comes to light.
“The methodology for maintaining the AAOS guidelines aims to update guideline documents at least every 10 years,” Dr. Brophy said in an interview. “Since the last edition was from 2013, it was time to provide an updated guideline on this very important topic that affects such a high percentage of our patients and providers.”
The guidelines work group, composed of 12 medical doctors and 1 physical therapist, evaluated the evidence for 29 areas of treatment.
A rating scale based on available evidence and the strength of related medical studies labeled each treatment area as demonstrating strong, moderate, or limited evidence.
Eight treatment modalities weighed in with strong evidence for or against their use: lateral wedge insoles, topical or oral NSAIDs, exercise (supervised or unsupervised), self-management programs, patient education programs, oral acetaminophen, and oral opioids.
According to Dr. Brophy, many of the recommendations assigned a strong evidence base were similar to the prior edition of the guidelines.
Oral medications
NSAIDs and acetaminophen still remain steadfast options for the treatment of knee pain secondary to OA.
The most notable change was that opioids, which have a long history of being used to treat pain, are strongly recommended not to be used for arthritis.
“Reflecting the growing awareness of and emphasis on the opioid epidemic, one of the strongest changes between the current and prior guidelines centers on the use of opioid medications,” Dr. Brophy said. “In the prior guideline, a strong recommendation was made in favor of tramadol with an inconclusive recommendation made regarding other opioid medications. The updated guideline demonstrates clearly the evidence does not support the use of opioid medications – including tramadol – to treat knee osteoarthritis.”
This may require some education for both patients and doctors to buy in that knee pain can be treated adequately with NSAIDs and acetaminophen.
Patients may not understand that anti-inflammatory drugs treat the pain they are experiencing. They may equate an opioid with a “pain pill” and may need education from their doctor that NSAIDs and acetaminophen not only can relieve their pain, but also avoid potential adverse events prior to or after surgery should they progress to knee replacement surgery.
Furthermore, primary care physicians may not be looking at the long-term picture. Solving a short-term pain problem with opioids may limit the medication’s ability to provide pain relief after surgery should a patient develop a tolerance to the medication’s effects.
Recommendations on hip and foot alignment interventions
When it comes to alignment and joint stresses, the knee is sometimes considered the innocent bystander of hip and foot alignment.
Insoles. How the hip and foot align with each can determine the amount of weight that passes through the medial (inner) or lateral (outer) compartment of the knee. To that end, lateral foot insoles have been used in the past for unloading parts of the knee.
Nevertheless, recent evidence has failed to demonstrate a significant benefit for insoles in the setting of OA knee pain, earning the practice a strong recommendation against its use.
High-tibial osteotomy (HTO). The weight-bearing axis of the lower-extremity axis can also be realigned with HTO. The procedure shifts the body’s weight slightly to the opposite side of the knee.
Newer research has led the practice to be downgraded one level in the new guideline, from moderate to limited, despite its widespread use.
It will, however, likely continue to be used as an alternative to total knee replacement in younger patients and to shift weight away from an area of the knee where cartilage is being restored with a concomitant surgical procedure, according to the work group. They noted that additional research studies on the long-term efficacy of the procedure are still needed.
Topical treatments. The guideline authors gave these a strong recommendation. Gels with anti-inflammatory medication have long been available but were prescription only or of considerable cost. Now several affordable over-the-counter options with the same prescription strength can be found in pharmacies and supermarkets.
What makes these medications unique is that they have an NSAID medication in the formulation, which the vast majority of topical treatments found on shelves do not. They also benefit patients who are unable to tolerate oral NSAIDs because of gastrointestinal side effects.
Comparison with 2019 OARSI recommendations
In 2019, the Osteoarthritis Research Society International also published guidelines for the management of OA of the hand, hip, and knee.
Thomas Trojian, MD, a family medicine physician with expertise in sports medicine in York, Pa., and member of both the AAOS and OARSI recommendation committees, noted that the OARSI guidelines are meant to be practical guidelines of stepwise nonoperative treatment.
He said in an interview that “the OARSI guidelines recommend dietary weight management, education, and land-based [exercise] therapy, next topical NSAIDs, then injection therapy.”
Intra-articular steroids and viscosupplementation injection therapy in the form of hyaluronic acid derivatives continue to be a mainstay of treatment for both groups.
The AAOS group notably gave a moderate strength recommendation for intra-articular steroid injections with the caveat that the effects typically only last for 3 months. They also included newer extended-release steroid injections in the recommendation, stating that the evidence moderately suggests they provide more benefit than traditional short-acting steroid injections.
Methodology differs between guidelines
In the areas where the guidelines don’t fully line up, it is important to remember the methodology of each group often drives the guidelines and recommendations.
According to Yale Fillingham, MD, an orthopedic surgeon in group private practice in the greater Philadelphia area and the other cochair of the AAOS guidelines committee, the biggest difference between the AAOS and OARSI guidelines is that, although the OARSI guidelines are also grounded in the literature, the recommendation level was based on voting among panel members.
“The AAOS methodology requires the recommendation and strength of the recommendation to be dictated primarily by the best available evidence in the literature and much less on the expertise and opinion of the voting panel,” Dr. Fillingham said in an interview.
He pointed out that the AAOS voting panel can alter the guideline by adjusting the strength of the recommendation but noted it was only in very clearly defined situations. Therefore, the differences in methodology between the groups make it difficult to directly compare the two guidelines.
Multiple guidelines do, however, point to the importance of the issue. Dr. Fillingham commented: “The numerous organizations that have produced guidelines on the treatment of knee osteoarthritis are a testament to the widespread and profound impact of knee osteoarthritis on our health care system and society.”
As a member of both recommendation groups, Dr. Trojian finds both guidelines reveal the importance of understanding that knee OA is a chronic illness. “There are ways we can manage knee OA and reduce the morbidity. ... The core skills of motivational interviewing are important. Open-ended questions, affirmation, reflection, and summarizing are needed to help patients find and remove roadblocks to promote lifestyle changes.”
Dr. Brophy, Dr. Trojian, and Dr. Fillingham have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
After nearly a decade, the American Academy of Orthopaedic Surgeons has updated its guidance on nonoperative treatment for knee osteoarthritis.
The clinical practice guidelines, released Sept. 13, 2021, is the third edition of the orthopedic society’s clinical practice recommendations.
According to Robert Brophy, MD, FAAOS, an orthopedic surgeon at Washington University, St. Louis, and cochair of the AAOS clinical practice guideline work group, the AAOS guidelines are a “living document” that needs periodic updating as new research comes to light.
“The methodology for maintaining the AAOS guidelines aims to update guideline documents at least every 10 years,” Dr. Brophy said in an interview. “Since the last edition was from 2013, it was time to provide an updated guideline on this very important topic that affects such a high percentage of our patients and providers.”
The guidelines work group, composed of 12 medical doctors and 1 physical therapist, evaluated the evidence for 29 areas of treatment.
A rating scale based on available evidence and the strength of related medical studies labeled each treatment area as demonstrating strong, moderate, or limited evidence.
Eight treatment modalities weighed in with strong evidence for or against their use: lateral wedge insoles, topical or oral NSAIDs, exercise (supervised or unsupervised), self-management programs, patient education programs, oral acetaminophen, and oral opioids.
According to Dr. Brophy, many of the recommendations assigned a strong evidence base were similar to the prior edition of the guidelines.
Oral medications
NSAIDs and acetaminophen still remain steadfast options for the treatment of knee pain secondary to OA.
The most notable change was that opioids, which have a long history of being used to treat pain, are strongly recommended not to be used for arthritis.
“Reflecting the growing awareness of and emphasis on the opioid epidemic, one of the strongest changes between the current and prior guidelines centers on the use of opioid medications,” Dr. Brophy said. “In the prior guideline, a strong recommendation was made in favor of tramadol with an inconclusive recommendation made regarding other opioid medications. The updated guideline demonstrates clearly the evidence does not support the use of opioid medications – including tramadol – to treat knee osteoarthritis.”
This may require some education for both patients and doctors to buy in that knee pain can be treated adequately with NSAIDs and acetaminophen.
Patients may not understand that anti-inflammatory drugs treat the pain they are experiencing. They may equate an opioid with a “pain pill” and may need education from their doctor that NSAIDs and acetaminophen not only can relieve their pain, but also avoid potential adverse events prior to or after surgery should they progress to knee replacement surgery.
Furthermore, primary care physicians may not be looking at the long-term picture. Solving a short-term pain problem with opioids may limit the medication’s ability to provide pain relief after surgery should a patient develop a tolerance to the medication’s effects.
Recommendations on hip and foot alignment interventions
When it comes to alignment and joint stresses, the knee is sometimes considered the innocent bystander of hip and foot alignment.
Insoles. How the hip and foot align with each can determine the amount of weight that passes through the medial (inner) or lateral (outer) compartment of the knee. To that end, lateral foot insoles have been used in the past for unloading parts of the knee.
Nevertheless, recent evidence has failed to demonstrate a significant benefit for insoles in the setting of OA knee pain, earning the practice a strong recommendation against its use.
High-tibial osteotomy (HTO). The weight-bearing axis of the lower-extremity axis can also be realigned with HTO. The procedure shifts the body’s weight slightly to the opposite side of the knee.
Newer research has led the practice to be downgraded one level in the new guideline, from moderate to limited, despite its widespread use.
It will, however, likely continue to be used as an alternative to total knee replacement in younger patients and to shift weight away from an area of the knee where cartilage is being restored with a concomitant surgical procedure, according to the work group. They noted that additional research studies on the long-term efficacy of the procedure are still needed.
Topical treatments. The guideline authors gave these a strong recommendation. Gels with anti-inflammatory medication have long been available but were prescription only or of considerable cost. Now several affordable over-the-counter options with the same prescription strength can be found in pharmacies and supermarkets.
What makes these medications unique is that they have an NSAID medication in the formulation, which the vast majority of topical treatments found on shelves do not. They also benefit patients who are unable to tolerate oral NSAIDs because of gastrointestinal side effects.
Comparison with 2019 OARSI recommendations
In 2019, the Osteoarthritis Research Society International also published guidelines for the management of OA of the hand, hip, and knee.
Thomas Trojian, MD, a family medicine physician with expertise in sports medicine in York, Pa., and member of both the AAOS and OARSI recommendation committees, noted that the OARSI guidelines are meant to be practical guidelines of stepwise nonoperative treatment.
He said in an interview that “the OARSI guidelines recommend dietary weight management, education, and land-based [exercise] therapy, next topical NSAIDs, then injection therapy.”
Intra-articular steroids and viscosupplementation injection therapy in the form of hyaluronic acid derivatives continue to be a mainstay of treatment for both groups.
The AAOS group notably gave a moderate strength recommendation for intra-articular steroid injections with the caveat that the effects typically only last for 3 months. They also included newer extended-release steroid injections in the recommendation, stating that the evidence moderately suggests they provide more benefit than traditional short-acting steroid injections.
Methodology differs between guidelines
In the areas where the guidelines don’t fully line up, it is important to remember the methodology of each group often drives the guidelines and recommendations.
According to Yale Fillingham, MD, an orthopedic surgeon in group private practice in the greater Philadelphia area and the other cochair of the AAOS guidelines committee, the biggest difference between the AAOS and OARSI guidelines is that, although the OARSI guidelines are also grounded in the literature, the recommendation level was based on voting among panel members.
“The AAOS methodology requires the recommendation and strength of the recommendation to be dictated primarily by the best available evidence in the literature and much less on the expertise and opinion of the voting panel,” Dr. Fillingham said in an interview.
He pointed out that the AAOS voting panel can alter the guideline by adjusting the strength of the recommendation but noted it was only in very clearly defined situations. Therefore, the differences in methodology between the groups make it difficult to directly compare the two guidelines.
Multiple guidelines do, however, point to the importance of the issue. Dr. Fillingham commented: “The numerous organizations that have produced guidelines on the treatment of knee osteoarthritis are a testament to the widespread and profound impact of knee osteoarthritis on our health care system and society.”
As a member of both recommendation groups, Dr. Trojian finds both guidelines reveal the importance of understanding that knee OA is a chronic illness. “There are ways we can manage knee OA and reduce the morbidity. ... The core skills of motivational interviewing are important. Open-ended questions, affirmation, reflection, and summarizing are needed to help patients find and remove roadblocks to promote lifestyle changes.”
Dr. Brophy, Dr. Trojian, and Dr. Fillingham have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.