User login
Effect of Multidisciplinary Transitional Pain Service on Health Care Use and Costs Following Orthopedic Surgery
Opioid use disorder (OUD) is a significant cause of morbidity, mortality, and health care costs in the US.1,2 Surgery can be the inciting cause for exposure to an opioid; as many as 23% of patients develop chronic OUD following surgery.3,4 Patients with a history of substance use, mood disorders, anxiety, or previous chronic opioid use (COU) are at risk for relapse, dose escalation, and poor pain control after high-risk surgery, such as orthopedic joint procedures.5 Recently focus has been on identifying high-risk patients before orthopedic joint surgery and implementing evidence-based strategies that reduce the postoperative incidence of COU.
A transitional pain service (TPS) has been shown to reduce COU for high-risk surgical patients in different health care settings.6-9 The TPS model bundles multiple interventions that can be applied to patients at high risk for COU within a health care system. This includes individually tailored programs for preoperative education or pain management planning, use of multimodal analgesia (including regional or neuraxial techniques or nonopioid systemic medications), application of nonpharmacologic modalities (such as cognitive-based intervention), and a coordinated approach to postdischarge instructions and transitions of care. These interventions are coordinated by a multidisciplinary clinical service consisting of anesthesiologists and advanced practice clinicians with specialization in acute pain management and opioid tapering, nurse care coordinators, and psychologists with expertise in cognitive behavioral therapy.
TPS has been shown to reduce the incidence of COU for patients undergoing orthopedic joint surgery, but its impact on health care use and costs is unknown.6-9 The TPS intervention is resource intensive and increases the use of health care for preoperative education or pain management, which may increase the burden of costs. However, reducing long-term COU may reduce the use of health care for COU- and OUD-related complications, leading to cost savings. This study evaluated whether the TPS intervention influenced health care use and cost for inpatient, outpatient, or pharmacy services during the year following orthopedic joint surgery compared with that of the standard pain management care for procedures that place patients at high risk for COU. We used a difference-in-differences (DID) analysis to estimate this intervention effect, using multivariable regression models that controlled for unobserved time trends and cohort characteristics.
METHODS
This was a quasi-experimental study of patients who underwent orthopedic joint surgery and associated procedures at high risk for COU at the Veterans Affairs Salt Lake City Healthcare System (VASLCHS) between January 2016 through April 2020. The pre-TPS period between January 2016 through December 2017 was compared with the post-TPS period between January 2018 to September 2019. The control patient cohort was selected from 5 geographically diverse VA health care systems throughout the US: Eastern Colorado, Central Plains (Nebraska), White River Junction (Vermont), North Florida/South Georgia, and Portland (Oregon). By sampling health care costs from VA medical centers (VAMCs) across these different regions, our control group was generalizable to veterans receiving orthopedic joint surgery across the US. This study used data from the US Department of Veterans Affairs (VA) Corporate Data Warehouse, a repository of nearly all clinical and administrative data found in electronic health records for VA-provided care and fee-basis care paid for by the VA.10 All data were hosted and analyzed in the VA Informatics and Computing Infrastructure (VINCI) workspace. The University of Utah Institutional Review Board and the VASLCHS Office of Research and Development approved the protocol for this study.
TPS Intervention
The VASLCHS TPS has already been described in detail elsewhere.6,7 Briefly, patients at high risk for COU at the VASLCHS were enrolled in the TPS program before surgery for total knee, hip, or shoulder arthroplasty or rotator cuff procedures. The TPS service consists of an anesthesiologist and advanced practice clinician with specialization in acute pain management and opioid tapering, a psychologist with expertise in cognitive behavioral therapy, and 3 nurse care coordinators. These TPS practitioners work together to provide preoperative education, including setting expectations regarding postoperative pain, recommending nonopioid pain management strategies, and providing guidance regarding the appropriate use of opioids for surgical pain. Individual pain plans were developed and implemented for the perioperative period. After surgery, the TPS provided recommendations and support for nonopioid pain therapies and opioid tapers. Patients were followed by the TPS team for at least 12 months after surgery. At a minimum, the goals set by TPS included cessation of all opioid use for prior nonopioid users (NOU) by 90 days after surgery and the return to baseline opioid use or lower for prior COU patients by 90 days after surgery. The TPS also encouraged and supported opioid tapering among COU patients to reduce or completely stop opioid use after surgery.
Patient Cohorts
Veterans having primary or revision total knee, hip, or shoulder arthroplasty or rotator cuff repair between January 1, 2016, and September 30, 2019, at the aforementioned VAMCs were included in the study. Patients who had any hospitalization within 90 days pre- or postindex surgery or who died within 8 months after surgery were excluded from analysis. Patients who had multiple surgeries during the index inpatient visit or within 90 days after the index surgery also were excluded. Comorbid conditions for mental health and substance use were identified using the International Classification of Diseases, 10th revision Clinical Modification (ICD-10) codes or 9th revision equivalent grouped by Clinical Classifications Software Refined (CCS-R).11 Preoperative exposure to clinically relevant pharmacotherapy (ie, agents associated with prolonged opioid use and nonopioid adjuvants) was captured using VA outpatient prescription records (eAppendix 1).
Outcome Variables
Outcome variables included health care use and costs during 1-year pre- and postperiods from the date of surgery. VA health care costs for outpatient, inpatient, and pharmacy services for direct patient care were collected from the Managerial Cost Accounting System, an activity-based cost allocation system that generates estimates of the cost of individual VA hospital stays, health care encounters, and medications. Health care use was defined as the number of encounters for each visit type in the Managerial Cost Accounting System. All costs were adjusted to 2019 US dollars, using the Personal Consumption Expenditures price index for health care services.15
A set of sociodemographic variables including sex, age at surgery, race and ethnicity, rurality, military branch (Army, Air Force, Marine Corps, Navy, and other), and service connectivity were included as covariates in our regression models.
Statistical Analyses
Descriptive analyses were used to evaluate differences in baseline patient sociodemographic and clinical characteristics between pre- and postperiods for TPS intervention and control cohorts using 2-sample t tests for continuous variables and χ2 tests for categorical variables. We summarized unadjusted health care use and costs for outpatient, inpatient, and pharmacy visits and compared the pre- and postintervention periods using the Mann-Whitney test. Both mean (SD) and median (IQR) were considered, reflecting the skewed distribution of the outcome variables.
We used a DID approach to assess the intervention effect while minimizing confounding from the nonrandom sample. The DID approach controls for unobserved differences between VAMCs that are related to both the intervention and outcomes while controlling for trends over time that could affect outcomes across clinics. To implement the DID approach, we included 3 key independent variables in our regression models: (1) an indicator for whether the observation occurred in the postintervention period; (2) an indicator for whether the patient was exposed to the TPS intervention; and (3) the interaction between these 2 variables.
For cost outcomes, we used multivariable generalized linear models with a log link and a Poisson or Υ family. We analyzed inpatient costs using a 2-part generalized linear model because only 17% to 20% of patients had ≥ 1 inpatient visit. We used multivariable negative binomial regression for health care use outcomes. Demographic and clinical covariates described earlier were included in the regression models to control for differences in the composition of patient groups and clinics that could lead to confounding bias.
RESULTS
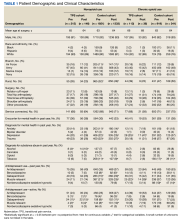
Of the 4954 patients included in our study cohort, 3545 (71.6%) were in the NOU group and 1409 (28.4%) were in the COU group. Among the NOU cohort, 361 patients were in the intervention group and 3184 in the control group. Among the COU cohort, 149 patients were in the intervention group and 1260 in the control group (Table 1). Most patients were male, White race, with a mean (SD) age of 64 (11) years. The most common orthopedic procedure was total knee arthroplasty, followed by total hip arthroplasty. Among both NOU and COU cohorts, patients’ characteristics were similar between the pre- and postintervention period among either TPS or control cohort.
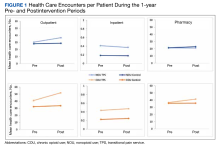
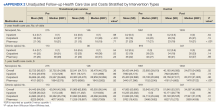
Figures 1 and 2 and eAppendix 2 depict unadjusted per-person average outpatient, inpatient, and pharmacy visits and costs incurred during the 1-year pre- and postintervention periods for the NOU and COU cohorts. Average total health care follow-up costs ranged from $40,000 to $53,000 for NOU and from $47,000 to $82,000 for COU cohort. Cost for outpatient visits accounted for about 70% of the average total costs, followed by costs for inpatient visits of about 20%, and costs for pharmacy for the remaining.
For the NOU cohort, the number of health care encounters remained fairly stable between periods except for the outpatient visits among the TPS group. The TPS group experienced an increase in mean outpatient visits in the postperiod: 30 vs 37 visits (23%) (
Table 2 summarizes the results from the multivariable DID analyses for the outpatient, inpatient, and pharmacy visit and cost outcomes. Here, the estimated effect of the TPS intervention is the coefficient from the interaction between the postintervention and TPS exposure indicator variables. This coefficient was calculated as the difference in the outcome before and after the TPS intervention among the TPS group minus the difference in the outcome before and after the TPS intervention among the control group. For the NOU cohort, TPS was associated with an increase in the use of outpatient health care (mean [SD] increase of 6.9 [2] visits; P < .001) after the surgery with no statistically significant effect on outpatient costs (mean [SD] increase of $2787 [$3749]; P = .55). There was no statistically significant effect of TPS on the use of inpatient visits or pharmacy, but a decrease in costs for inpatient visits among those who had at least 1 inpatient visit (mean [SD] decrease of $12,170 [$6100]; P = .02). For the COU cohort, TPS had no statistically significant impact on the use of outpatient, inpatient, or pharmacy or the corresponding costs.
DISCUSSION
TPS is a multidisciplinary approach to perioperative pain management that has been shown to reduce both the quantity and duration of opioid use among orthopedic surgery patients.6,7 Although the cost burden of providing TPS services to prevent COU is borne by the individual health care system, it is unclear whether this expense is offset by lower long-term medical costs and health care use for COU- and OUD-related complications. In this study focused on a veteran population undergoing orthopedic joint procedures, a DID analysis of cost and health care use showed that TPS, which has been shown to reduce COU for high-risk surgical patients, can be implemented without increasing the overall costs to the VA health care system during the 1 year following surgery, even with increased outpatient visits. For NOU patients, there was no difference in outpatient visit costs or pharmacy costs over 12 months after surgery, although there was a significant reduction in subsequent inpatient costs over the same period. Further, there was no difference in outpatient, inpatient, or pharmacy costs after surgery for COU patients. These findings suggest that TPS can be a cost-effective approach to reduce opioid use among patients undergoing orthopedic joint surgery in VAMCs.
The costs of managing COU after surgery are substantial. Prior reports have shown that adjusted total health care costs are 1.6 to 2.5 times higher for previously NOU patients with new COU after major surgery than those for such patients without persistent use.16 The 1-year costs associated with new COU in this prior study ranged between $7944 and $17,702 after inpatient surgery and between $5598 and $12,834 after outpatient index surgery, depending on the payer, which are in line with the cost differences found in our current study. Another report among patients with COU following orthopedic joint replacement showed that they had higher use of inpatient, emergency department, and ambulance/paramedic services in the 12 months following their surgery than did those without persistent use.17 Although these results highlight the impact that COU plays in driving increased costs after major surgery, there have been limited studies focused on interventions that can neutralize the costs associated with opioid misuse after surgery. To our knowledge, our study is the first analysis to show the impact of using an intervention such as TPS to reduce postoperative opioid use on health care use and cost.
Although a rigorous and comprehensive return on investment analysis was beyond the scope of this analysis, these results may have several implications for other health care systems and hospitals that wish to invest in a multidisciplinary perioperative pain management program such as TPS but may be reluctant due to the upfront investment. First, the increased number of patient follow-up visits needed during TPS seems to be more than offset by the reduction in opioid use and associated complications that may occur after surgery. Second, TPS did not seem to be associated with an increase in overall health care costs during the 1-year follow-up period. Together, these results indicate that the return on investment for a TPS approach to perioperative pain management in which optimal patient-centered outcomes are achieved without increasing long-term costs to a health care system may be positive.
Limitations
This study has several limitations. First, this was a quasi-experimental observational study, and the associations we identified between intervention and outcomes should not be assumed to demonstrate causality. Although our DID analysis controlled for an array of demographic and clinical characteristics, differences in medical costs and health care use between the 2 cohorts might be driven by unobserved confounding variables.
Our study also was limited to veterans who received medical care at the VA, and results may not be generalizable to other non-VA health care systems or to veterans with Medicare insurance who have dual benefits. While our finding on health care use and costs may be incomplete because of the uncaptured health care use outside the VA, our DID analysis helped reduce unobserved bias because the absence of data outside of VA care applies to both TPS and control groups. Further, the total costs of operating a TPS program at any given institution will depend on the size of the hospital and volume of surgical patients who meet criteria for enrollment. However, the relative differences in health care use and costs may be extrapolated to patients undergoing orthopedic surgery in other types of academic and community-based health care systems.
Furthermore, this analysis focused primarily on COU and NOU patients undergoing orthopedic joint surgery. While this represents a high-risk population for OUD, the costs and health care use associated with delivering the TPS intervention to other types of surgical procedures may be significantly different. All costs in this analysis were based on 2019 estimates and do not account for the potential inflation over the past several years. Nonmonetary costs to the patient and per-person average total intervention costs were not included in the study. However, we assumed that costs associated with TPS and standard of care would have increased to an equivalent degree over the same period. Further, the average cost of TPS per patient (approximately $900) is relatively small compared with the average annual costs during 1-year pre- and postoperative periods and was not expected to have a significant effect on the analysis.
Conclusions
We found that the significant reduction in COU seen in previous studies following the implementation of TPS was not accompanied by increased health care costs.6,7 When considering the other costs of long-term opioid use, such as abuse potential, overdose, death, and increased disability, implementation of a TPS service has the potential to improve patient quality of life while reducing other health-related costs. Health care systems should consider the implementation of similar multidisciplinary approaches to perioperative pain management to improve outcomes after orthopedic joint surgery and other high-risk procedures.
1. Rudd RA, Seth P, David F, et al. Increases in drug and opioid-involved overdose deaths—United States, 2010-2015. MMWR Morb Mortal Wkly Rep. 2016;65(50-51):1445-1452. doi:10.15585/mmwr.mm655051e1
2. Florence CS, Zhou C, Luo F, Xu L. The economic burden of prescription opioid overdose, abuse, and dependence in the United States, 2013. Med Care. 2016;54(10):901-906. doi:10.1097/MLR.0000000000000625
3. Jiang X, Orton M, Feng R, et al. Chronic opioid usage in surgical patients in a large academic center. Ann Surg. 2017;265(4):722-727. doi:10.1097/SLA.0000000000001780
4. Johnson SP, Chung KC, Zhong L, et al. Risk of prolonged opioid use among opioid-naive patients following common hand surgery procedures. J Hand Surg Am. 2016;41(10):947-957, e3. doi:10.1016/j.jhsa.2016.07.113
5. Brummett CM, Waljee JF, Goesling J, et al. New persistent opioid use after minor and major surgical procedures in US adults. JAMA Surg. 2017;152(6):e170504. doi:10.1001/jamasurg.2017.0504
6. Buys MJ, Bayless K, Romesser J, et al. Multidisciplinary transitional pain service for the veteran population. Fed Pract. 2020;37(10):472-478. doi:10.12788/fp.0053
7. Buys MJ, Bayless K, Romesser J, et al. Opioid use among veterans undergoing major joint surgery managed by a multidisciplinary transitional pain service. Reg Anesth Pain Med. 2020;45(11):847-852. doi:10.1136/rapm-2020-101797
8. Huang A, Katz J, Clarke H. Ensuring safe prescribing of controlled substances for pain following surgery by developing a transitional pain service. Pain Manag. 2015;5(2):97-105. doi:10.2217/pmt.15.7
9. Katz J, Weinrib A, Fashler SR, et al. The Toronto General Hospital Transitional Pain Service: development and implementation of a multidisciplinary program to prevent chronic postsurgical pain. J Pain Res. 2015;8:695-702. doi:10.2147/JPR.S91924
10. Fihn SD, Francis J, Clancy C, et al. Insights from advanced analytics at the Veterans Health Administration. Health Aff (Millwood). 2014;33(7):1203-1211. doi:10.1377/hlthaff.2014.0054
11. Agency for Healthcare Research and Quality. Clinical Classifications Software Refined (CCSR). Updated December 9, 2022. Accessed October 30, 2023. www.hcup-us.ahrq.gov/toolssoftware/ccsr/ccs_refined.jsp
12. Mosher HJ, Richardson KK, Lund BC. The 1-year treatment course of new opioid recipients in Veterans Health Administration. Pain Med. 2016;17(7):1282-1291. doi:10.1093/pm/pnw058
13. Hadlandsmyth K, Mosher HJ, Vander Weg MW, O’Shea AM, McCoy KD, Lund BC. Utility of accumulated opioid supply days and individual patient factors in predicting probability of transitioning to long-term opioid use: an observational study in the Veterans Health Administration. Pharmacol Res Perspect. 2020;8(2):e00571. doi:10.1002/prp2.571
14. Pagé MG, Kudrina I, Zomahoun HTV, et al. Relative frequency and risk factors for long-term opioid therapy following surgery and trauma among adults: a systematic review protocol. Syst Rev. 2018;7(1):97. doi:10.1186/s13643-018-0760-3
15. US. Bureau of Economic Analysis. Price indexes for personal consumption expenditures by major type of product. Accessed October 30, 2023. https://apps.bea.gov/iTable/?reqid=19&step=3&isuri=1&nipa_table_list=64&categories=survey
16. Brummett CM, Evans-Shields J, England C, et al. Increased health care costs associated with new persistent opioid use after major surgery in opioid-naive patients. J Manag Care Spec Pharm. 2021;27(6):760-771. doi:10.18553/jmcp.2021.20507
17. Gold LS, Strassels SA, Hansen RN. Health care costs and utilization in patients receiving prescriptions for long-acting opioids for acute postsurgical pain. Clin J Pain. 2016;32(9):747-754. doi:10.1097/ajp.0000000000000322
Opioid use disorder (OUD) is a significant cause of morbidity, mortality, and health care costs in the US.1,2 Surgery can be the inciting cause for exposure to an opioid; as many as 23% of patients develop chronic OUD following surgery.3,4 Patients with a history of substance use, mood disorders, anxiety, or previous chronic opioid use (COU) are at risk for relapse, dose escalation, and poor pain control after high-risk surgery, such as orthopedic joint procedures.5 Recently focus has been on identifying high-risk patients before orthopedic joint surgery and implementing evidence-based strategies that reduce the postoperative incidence of COU.
A transitional pain service (TPS) has been shown to reduce COU for high-risk surgical patients in different health care settings.6-9 The TPS model bundles multiple interventions that can be applied to patients at high risk for COU within a health care system. This includes individually tailored programs for preoperative education or pain management planning, use of multimodal analgesia (including regional or neuraxial techniques or nonopioid systemic medications), application of nonpharmacologic modalities (such as cognitive-based intervention), and a coordinated approach to postdischarge instructions and transitions of care. These interventions are coordinated by a multidisciplinary clinical service consisting of anesthesiologists and advanced practice clinicians with specialization in acute pain management and opioid tapering, nurse care coordinators, and psychologists with expertise in cognitive behavioral therapy.
TPS has been shown to reduce the incidence of COU for patients undergoing orthopedic joint surgery, but its impact on health care use and costs is unknown.6-9 The TPS intervention is resource intensive and increases the use of health care for preoperative education or pain management, which may increase the burden of costs. However, reducing long-term COU may reduce the use of health care for COU- and OUD-related complications, leading to cost savings. This study evaluated whether the TPS intervention influenced health care use and cost for inpatient, outpatient, or pharmacy services during the year following orthopedic joint surgery compared with that of the standard pain management care for procedures that place patients at high risk for COU. We used a difference-in-differences (DID) analysis to estimate this intervention effect, using multivariable regression models that controlled for unobserved time trends and cohort characteristics.
METHODS
This was a quasi-experimental study of patients who underwent orthopedic joint surgery and associated procedures at high risk for COU at the Veterans Affairs Salt Lake City Healthcare System (VASLCHS) between January 2016 through April 2020. The pre-TPS period between January 2016 through December 2017 was compared with the post-TPS period between January 2018 to September 2019. The control patient cohort was selected from 5 geographically diverse VA health care systems throughout the US: Eastern Colorado, Central Plains (Nebraska), White River Junction (Vermont), North Florida/South Georgia, and Portland (Oregon). By sampling health care costs from VA medical centers (VAMCs) across these different regions, our control group was generalizable to veterans receiving orthopedic joint surgery across the US. This study used data from the US Department of Veterans Affairs (VA) Corporate Data Warehouse, a repository of nearly all clinical and administrative data found in electronic health records for VA-provided care and fee-basis care paid for by the VA.10 All data were hosted and analyzed in the VA Informatics and Computing Infrastructure (VINCI) workspace. The University of Utah Institutional Review Board and the VASLCHS Office of Research and Development approved the protocol for this study.
TPS Intervention
The VASLCHS TPS has already been described in detail elsewhere.6,7 Briefly, patients at high risk for COU at the VASLCHS were enrolled in the TPS program before surgery for total knee, hip, or shoulder arthroplasty or rotator cuff procedures. The TPS service consists of an anesthesiologist and advanced practice clinician with specialization in acute pain management and opioid tapering, a psychologist with expertise in cognitive behavioral therapy, and 3 nurse care coordinators. These TPS practitioners work together to provide preoperative education, including setting expectations regarding postoperative pain, recommending nonopioid pain management strategies, and providing guidance regarding the appropriate use of opioids for surgical pain. Individual pain plans were developed and implemented for the perioperative period. After surgery, the TPS provided recommendations and support for nonopioid pain therapies and opioid tapers. Patients were followed by the TPS team for at least 12 months after surgery. At a minimum, the goals set by TPS included cessation of all opioid use for prior nonopioid users (NOU) by 90 days after surgery and the return to baseline opioid use or lower for prior COU patients by 90 days after surgery. The TPS also encouraged and supported opioid tapering among COU patients to reduce or completely stop opioid use after surgery.
Patient Cohorts
Veterans having primary or revision total knee, hip, or shoulder arthroplasty or rotator cuff repair between January 1, 2016, and September 30, 2019, at the aforementioned VAMCs were included in the study. Patients who had any hospitalization within 90 days pre- or postindex surgery or who died within 8 months after surgery were excluded from analysis. Patients who had multiple surgeries during the index inpatient visit or within 90 days after the index surgery also were excluded. Comorbid conditions for mental health and substance use were identified using the International Classification of Diseases, 10th revision Clinical Modification (ICD-10) codes or 9th revision equivalent grouped by Clinical Classifications Software Refined (CCS-R).11 Preoperative exposure to clinically relevant pharmacotherapy (ie, agents associated with prolonged opioid use and nonopioid adjuvants) was captured using VA outpatient prescription records (eAppendix 1).
Outcome Variables
Outcome variables included health care use and costs during 1-year pre- and postperiods from the date of surgery. VA health care costs for outpatient, inpatient, and pharmacy services for direct patient care were collected from the Managerial Cost Accounting System, an activity-based cost allocation system that generates estimates of the cost of individual VA hospital stays, health care encounters, and medications. Health care use was defined as the number of encounters for each visit type in the Managerial Cost Accounting System. All costs were adjusted to 2019 US dollars, using the Personal Consumption Expenditures price index for health care services.15
A set of sociodemographic variables including sex, age at surgery, race and ethnicity, rurality, military branch (Army, Air Force, Marine Corps, Navy, and other), and service connectivity were included as covariates in our regression models.
Statistical Analyses
Descriptive analyses were used to evaluate differences in baseline patient sociodemographic and clinical characteristics between pre- and postperiods for TPS intervention and control cohorts using 2-sample t tests for continuous variables and χ2 tests for categorical variables. We summarized unadjusted health care use and costs for outpatient, inpatient, and pharmacy visits and compared the pre- and postintervention periods using the Mann-Whitney test. Both mean (SD) and median (IQR) were considered, reflecting the skewed distribution of the outcome variables.
We used a DID approach to assess the intervention effect while minimizing confounding from the nonrandom sample. The DID approach controls for unobserved differences between VAMCs that are related to both the intervention and outcomes while controlling for trends over time that could affect outcomes across clinics. To implement the DID approach, we included 3 key independent variables in our regression models: (1) an indicator for whether the observation occurred in the postintervention period; (2) an indicator for whether the patient was exposed to the TPS intervention; and (3) the interaction between these 2 variables.
For cost outcomes, we used multivariable generalized linear models with a log link and a Poisson or Υ family. We analyzed inpatient costs using a 2-part generalized linear model because only 17% to 20% of patients had ≥ 1 inpatient visit. We used multivariable negative binomial regression for health care use outcomes. Demographic and clinical covariates described earlier were included in the regression models to control for differences in the composition of patient groups and clinics that could lead to confounding bias.
RESULTS
Of the 4954 patients included in our study cohort, 3545 (71.6%) were in the NOU group and 1409 (28.4%) were in the COU group. Among the NOU cohort, 361 patients were in the intervention group and 3184 in the control group. Among the COU cohort, 149 patients were in the intervention group and 1260 in the control group (Table 1). Most patients were male, White race, with a mean (SD) age of 64 (11) years. The most common orthopedic procedure was total knee arthroplasty, followed by total hip arthroplasty. Among both NOU and COU cohorts, patients’ characteristics were similar between the pre- and postintervention period among either TPS or control cohort.
Figures 1 and 2 and eAppendix 2 depict unadjusted per-person average outpatient, inpatient, and pharmacy visits and costs incurred during the 1-year pre- and postintervention periods for the NOU and COU cohorts. Average total health care follow-up costs ranged from $40,000 to $53,000 for NOU and from $47,000 to $82,000 for COU cohort. Cost for outpatient visits accounted for about 70% of the average total costs, followed by costs for inpatient visits of about 20%, and costs for pharmacy for the remaining.
For the NOU cohort, the number of health care encounters remained fairly stable between periods except for the outpatient visits among the TPS group. The TPS group experienced an increase in mean outpatient visits in the postperiod: 30 vs 37 visits (23%) (
Table 2 summarizes the results from the multivariable DID analyses for the outpatient, inpatient, and pharmacy visit and cost outcomes. Here, the estimated effect of the TPS intervention is the coefficient from the interaction between the postintervention and TPS exposure indicator variables. This coefficient was calculated as the difference in the outcome before and after the TPS intervention among the TPS group minus the difference in the outcome before and after the TPS intervention among the control group. For the NOU cohort, TPS was associated with an increase in the use of outpatient health care (mean [SD] increase of 6.9 [2] visits; P < .001) after the surgery with no statistically significant effect on outpatient costs (mean [SD] increase of $2787 [$3749]; P = .55). There was no statistically significant effect of TPS on the use of inpatient visits or pharmacy, but a decrease in costs for inpatient visits among those who had at least 1 inpatient visit (mean [SD] decrease of $12,170 [$6100]; P = .02). For the COU cohort, TPS had no statistically significant impact on the use of outpatient, inpatient, or pharmacy or the corresponding costs.
DISCUSSION
TPS is a multidisciplinary approach to perioperative pain management that has been shown to reduce both the quantity and duration of opioid use among orthopedic surgery patients.6,7 Although the cost burden of providing TPS services to prevent COU is borne by the individual health care system, it is unclear whether this expense is offset by lower long-term medical costs and health care use for COU- and OUD-related complications. In this study focused on a veteran population undergoing orthopedic joint procedures, a DID analysis of cost and health care use showed that TPS, which has been shown to reduce COU for high-risk surgical patients, can be implemented without increasing the overall costs to the VA health care system during the 1 year following surgery, even with increased outpatient visits. For NOU patients, there was no difference in outpatient visit costs or pharmacy costs over 12 months after surgery, although there was a significant reduction in subsequent inpatient costs over the same period. Further, there was no difference in outpatient, inpatient, or pharmacy costs after surgery for COU patients. These findings suggest that TPS can be a cost-effective approach to reduce opioid use among patients undergoing orthopedic joint surgery in VAMCs.
The costs of managing COU after surgery are substantial. Prior reports have shown that adjusted total health care costs are 1.6 to 2.5 times higher for previously NOU patients with new COU after major surgery than those for such patients without persistent use.16 The 1-year costs associated with new COU in this prior study ranged between $7944 and $17,702 after inpatient surgery and between $5598 and $12,834 after outpatient index surgery, depending on the payer, which are in line with the cost differences found in our current study. Another report among patients with COU following orthopedic joint replacement showed that they had higher use of inpatient, emergency department, and ambulance/paramedic services in the 12 months following their surgery than did those without persistent use.17 Although these results highlight the impact that COU plays in driving increased costs after major surgery, there have been limited studies focused on interventions that can neutralize the costs associated with opioid misuse after surgery. To our knowledge, our study is the first analysis to show the impact of using an intervention such as TPS to reduce postoperative opioid use on health care use and cost.
Although a rigorous and comprehensive return on investment analysis was beyond the scope of this analysis, these results may have several implications for other health care systems and hospitals that wish to invest in a multidisciplinary perioperative pain management program such as TPS but may be reluctant due to the upfront investment. First, the increased number of patient follow-up visits needed during TPS seems to be more than offset by the reduction in opioid use and associated complications that may occur after surgery. Second, TPS did not seem to be associated with an increase in overall health care costs during the 1-year follow-up period. Together, these results indicate that the return on investment for a TPS approach to perioperative pain management in which optimal patient-centered outcomes are achieved without increasing long-term costs to a health care system may be positive.
Limitations
This study has several limitations. First, this was a quasi-experimental observational study, and the associations we identified between intervention and outcomes should not be assumed to demonstrate causality. Although our DID analysis controlled for an array of demographic and clinical characteristics, differences in medical costs and health care use between the 2 cohorts might be driven by unobserved confounding variables.
Our study also was limited to veterans who received medical care at the VA, and results may not be generalizable to other non-VA health care systems or to veterans with Medicare insurance who have dual benefits. While our finding on health care use and costs may be incomplete because of the uncaptured health care use outside the VA, our DID analysis helped reduce unobserved bias because the absence of data outside of VA care applies to both TPS and control groups. Further, the total costs of operating a TPS program at any given institution will depend on the size of the hospital and volume of surgical patients who meet criteria for enrollment. However, the relative differences in health care use and costs may be extrapolated to patients undergoing orthopedic surgery in other types of academic and community-based health care systems.
Furthermore, this analysis focused primarily on COU and NOU patients undergoing orthopedic joint surgery. While this represents a high-risk population for OUD, the costs and health care use associated with delivering the TPS intervention to other types of surgical procedures may be significantly different. All costs in this analysis were based on 2019 estimates and do not account for the potential inflation over the past several years. Nonmonetary costs to the patient and per-person average total intervention costs were not included in the study. However, we assumed that costs associated with TPS and standard of care would have increased to an equivalent degree over the same period. Further, the average cost of TPS per patient (approximately $900) is relatively small compared with the average annual costs during 1-year pre- and postoperative periods and was not expected to have a significant effect on the analysis.
Conclusions
We found that the significant reduction in COU seen in previous studies following the implementation of TPS was not accompanied by increased health care costs.6,7 When considering the other costs of long-term opioid use, such as abuse potential, overdose, death, and increased disability, implementation of a TPS service has the potential to improve patient quality of life while reducing other health-related costs. Health care systems should consider the implementation of similar multidisciplinary approaches to perioperative pain management to improve outcomes after orthopedic joint surgery and other high-risk procedures.
Opioid use disorder (OUD) is a significant cause of morbidity, mortality, and health care costs in the US.1,2 Surgery can be the inciting cause for exposure to an opioid; as many as 23% of patients develop chronic OUD following surgery.3,4 Patients with a history of substance use, mood disorders, anxiety, or previous chronic opioid use (COU) are at risk for relapse, dose escalation, and poor pain control after high-risk surgery, such as orthopedic joint procedures.5 Recently focus has been on identifying high-risk patients before orthopedic joint surgery and implementing evidence-based strategies that reduce the postoperative incidence of COU.
A transitional pain service (TPS) has been shown to reduce COU for high-risk surgical patients in different health care settings.6-9 The TPS model bundles multiple interventions that can be applied to patients at high risk for COU within a health care system. This includes individually tailored programs for preoperative education or pain management planning, use of multimodal analgesia (including regional or neuraxial techniques or nonopioid systemic medications), application of nonpharmacologic modalities (such as cognitive-based intervention), and a coordinated approach to postdischarge instructions and transitions of care. These interventions are coordinated by a multidisciplinary clinical service consisting of anesthesiologists and advanced practice clinicians with specialization in acute pain management and opioid tapering, nurse care coordinators, and psychologists with expertise in cognitive behavioral therapy.
TPS has been shown to reduce the incidence of COU for patients undergoing orthopedic joint surgery, but its impact on health care use and costs is unknown.6-9 The TPS intervention is resource intensive and increases the use of health care for preoperative education or pain management, which may increase the burden of costs. However, reducing long-term COU may reduce the use of health care for COU- and OUD-related complications, leading to cost savings. This study evaluated whether the TPS intervention influenced health care use and cost for inpatient, outpatient, or pharmacy services during the year following orthopedic joint surgery compared with that of the standard pain management care for procedures that place patients at high risk for COU. We used a difference-in-differences (DID) analysis to estimate this intervention effect, using multivariable regression models that controlled for unobserved time trends and cohort characteristics.
METHODS
This was a quasi-experimental study of patients who underwent orthopedic joint surgery and associated procedures at high risk for COU at the Veterans Affairs Salt Lake City Healthcare System (VASLCHS) between January 2016 through April 2020. The pre-TPS period between January 2016 through December 2017 was compared with the post-TPS period between January 2018 to September 2019. The control patient cohort was selected from 5 geographically diverse VA health care systems throughout the US: Eastern Colorado, Central Plains (Nebraska), White River Junction (Vermont), North Florida/South Georgia, and Portland (Oregon). By sampling health care costs from VA medical centers (VAMCs) across these different regions, our control group was generalizable to veterans receiving orthopedic joint surgery across the US. This study used data from the US Department of Veterans Affairs (VA) Corporate Data Warehouse, a repository of nearly all clinical and administrative data found in electronic health records for VA-provided care and fee-basis care paid for by the VA.10 All data were hosted and analyzed in the VA Informatics and Computing Infrastructure (VINCI) workspace. The University of Utah Institutional Review Board and the VASLCHS Office of Research and Development approved the protocol for this study.
TPS Intervention
The VASLCHS TPS has already been described in detail elsewhere.6,7 Briefly, patients at high risk for COU at the VASLCHS were enrolled in the TPS program before surgery for total knee, hip, or shoulder arthroplasty or rotator cuff procedures. The TPS service consists of an anesthesiologist and advanced practice clinician with specialization in acute pain management and opioid tapering, a psychologist with expertise in cognitive behavioral therapy, and 3 nurse care coordinators. These TPS practitioners work together to provide preoperative education, including setting expectations regarding postoperative pain, recommending nonopioid pain management strategies, and providing guidance regarding the appropriate use of opioids for surgical pain. Individual pain plans were developed and implemented for the perioperative period. After surgery, the TPS provided recommendations and support for nonopioid pain therapies and opioid tapers. Patients were followed by the TPS team for at least 12 months after surgery. At a minimum, the goals set by TPS included cessation of all opioid use for prior nonopioid users (NOU) by 90 days after surgery and the return to baseline opioid use or lower for prior COU patients by 90 days after surgery. The TPS also encouraged and supported opioid tapering among COU patients to reduce or completely stop opioid use after surgery.
Patient Cohorts
Veterans having primary or revision total knee, hip, or shoulder arthroplasty or rotator cuff repair between January 1, 2016, and September 30, 2019, at the aforementioned VAMCs were included in the study. Patients who had any hospitalization within 90 days pre- or postindex surgery or who died within 8 months after surgery were excluded from analysis. Patients who had multiple surgeries during the index inpatient visit or within 90 days after the index surgery also were excluded. Comorbid conditions for mental health and substance use were identified using the International Classification of Diseases, 10th revision Clinical Modification (ICD-10) codes or 9th revision equivalent grouped by Clinical Classifications Software Refined (CCS-R).11 Preoperative exposure to clinically relevant pharmacotherapy (ie, agents associated with prolonged opioid use and nonopioid adjuvants) was captured using VA outpatient prescription records (eAppendix 1).
Outcome Variables
Outcome variables included health care use and costs during 1-year pre- and postperiods from the date of surgery. VA health care costs for outpatient, inpatient, and pharmacy services for direct patient care were collected from the Managerial Cost Accounting System, an activity-based cost allocation system that generates estimates of the cost of individual VA hospital stays, health care encounters, and medications. Health care use was defined as the number of encounters for each visit type in the Managerial Cost Accounting System. All costs were adjusted to 2019 US dollars, using the Personal Consumption Expenditures price index for health care services.15
A set of sociodemographic variables including sex, age at surgery, race and ethnicity, rurality, military branch (Army, Air Force, Marine Corps, Navy, and other), and service connectivity were included as covariates in our regression models.
Statistical Analyses
Descriptive analyses were used to evaluate differences in baseline patient sociodemographic and clinical characteristics between pre- and postperiods for TPS intervention and control cohorts using 2-sample t tests for continuous variables and χ2 tests for categorical variables. We summarized unadjusted health care use and costs for outpatient, inpatient, and pharmacy visits and compared the pre- and postintervention periods using the Mann-Whitney test. Both mean (SD) and median (IQR) were considered, reflecting the skewed distribution of the outcome variables.
We used a DID approach to assess the intervention effect while minimizing confounding from the nonrandom sample. The DID approach controls for unobserved differences between VAMCs that are related to both the intervention and outcomes while controlling for trends over time that could affect outcomes across clinics. To implement the DID approach, we included 3 key independent variables in our regression models: (1) an indicator for whether the observation occurred in the postintervention period; (2) an indicator for whether the patient was exposed to the TPS intervention; and (3) the interaction between these 2 variables.
For cost outcomes, we used multivariable generalized linear models with a log link and a Poisson or Υ family. We analyzed inpatient costs using a 2-part generalized linear model because only 17% to 20% of patients had ≥ 1 inpatient visit. We used multivariable negative binomial regression for health care use outcomes. Demographic and clinical covariates described earlier were included in the regression models to control for differences in the composition of patient groups and clinics that could lead to confounding bias.
RESULTS
Of the 4954 patients included in our study cohort, 3545 (71.6%) were in the NOU group and 1409 (28.4%) were in the COU group. Among the NOU cohort, 361 patients were in the intervention group and 3184 in the control group. Among the COU cohort, 149 patients were in the intervention group and 1260 in the control group (Table 1). Most patients were male, White race, with a mean (SD) age of 64 (11) years. The most common orthopedic procedure was total knee arthroplasty, followed by total hip arthroplasty. Among both NOU and COU cohorts, patients’ characteristics were similar between the pre- and postintervention period among either TPS or control cohort.
Figures 1 and 2 and eAppendix 2 depict unadjusted per-person average outpatient, inpatient, and pharmacy visits and costs incurred during the 1-year pre- and postintervention periods for the NOU and COU cohorts. Average total health care follow-up costs ranged from $40,000 to $53,000 for NOU and from $47,000 to $82,000 for COU cohort. Cost for outpatient visits accounted for about 70% of the average total costs, followed by costs for inpatient visits of about 20%, and costs for pharmacy for the remaining.
For the NOU cohort, the number of health care encounters remained fairly stable between periods except for the outpatient visits among the TPS group. The TPS group experienced an increase in mean outpatient visits in the postperiod: 30 vs 37 visits (23%) (
Table 2 summarizes the results from the multivariable DID analyses for the outpatient, inpatient, and pharmacy visit and cost outcomes. Here, the estimated effect of the TPS intervention is the coefficient from the interaction between the postintervention and TPS exposure indicator variables. This coefficient was calculated as the difference in the outcome before and after the TPS intervention among the TPS group minus the difference in the outcome before and after the TPS intervention among the control group. For the NOU cohort, TPS was associated with an increase in the use of outpatient health care (mean [SD] increase of 6.9 [2] visits; P < .001) after the surgery with no statistically significant effect on outpatient costs (mean [SD] increase of $2787 [$3749]; P = .55). There was no statistically significant effect of TPS on the use of inpatient visits or pharmacy, but a decrease in costs for inpatient visits among those who had at least 1 inpatient visit (mean [SD] decrease of $12,170 [$6100]; P = .02). For the COU cohort, TPS had no statistically significant impact on the use of outpatient, inpatient, or pharmacy or the corresponding costs.
DISCUSSION
TPS is a multidisciplinary approach to perioperative pain management that has been shown to reduce both the quantity and duration of opioid use among orthopedic surgery patients.6,7 Although the cost burden of providing TPS services to prevent COU is borne by the individual health care system, it is unclear whether this expense is offset by lower long-term medical costs and health care use for COU- and OUD-related complications. In this study focused on a veteran population undergoing orthopedic joint procedures, a DID analysis of cost and health care use showed that TPS, which has been shown to reduce COU for high-risk surgical patients, can be implemented without increasing the overall costs to the VA health care system during the 1 year following surgery, even with increased outpatient visits. For NOU patients, there was no difference in outpatient visit costs or pharmacy costs over 12 months after surgery, although there was a significant reduction in subsequent inpatient costs over the same period. Further, there was no difference in outpatient, inpatient, or pharmacy costs after surgery for COU patients. These findings suggest that TPS can be a cost-effective approach to reduce opioid use among patients undergoing orthopedic joint surgery in VAMCs.
The costs of managing COU after surgery are substantial. Prior reports have shown that adjusted total health care costs are 1.6 to 2.5 times higher for previously NOU patients with new COU after major surgery than those for such patients without persistent use.16 The 1-year costs associated with new COU in this prior study ranged between $7944 and $17,702 after inpatient surgery and between $5598 and $12,834 after outpatient index surgery, depending on the payer, which are in line with the cost differences found in our current study. Another report among patients with COU following orthopedic joint replacement showed that they had higher use of inpatient, emergency department, and ambulance/paramedic services in the 12 months following their surgery than did those without persistent use.17 Although these results highlight the impact that COU plays in driving increased costs after major surgery, there have been limited studies focused on interventions that can neutralize the costs associated with opioid misuse after surgery. To our knowledge, our study is the first analysis to show the impact of using an intervention such as TPS to reduce postoperative opioid use on health care use and cost.
Although a rigorous and comprehensive return on investment analysis was beyond the scope of this analysis, these results may have several implications for other health care systems and hospitals that wish to invest in a multidisciplinary perioperative pain management program such as TPS but may be reluctant due to the upfront investment. First, the increased number of patient follow-up visits needed during TPS seems to be more than offset by the reduction in opioid use and associated complications that may occur after surgery. Second, TPS did not seem to be associated with an increase in overall health care costs during the 1-year follow-up period. Together, these results indicate that the return on investment for a TPS approach to perioperative pain management in which optimal patient-centered outcomes are achieved without increasing long-term costs to a health care system may be positive.
Limitations
This study has several limitations. First, this was a quasi-experimental observational study, and the associations we identified between intervention and outcomes should not be assumed to demonstrate causality. Although our DID analysis controlled for an array of demographic and clinical characteristics, differences in medical costs and health care use between the 2 cohorts might be driven by unobserved confounding variables.
Our study also was limited to veterans who received medical care at the VA, and results may not be generalizable to other non-VA health care systems or to veterans with Medicare insurance who have dual benefits. While our finding on health care use and costs may be incomplete because of the uncaptured health care use outside the VA, our DID analysis helped reduce unobserved bias because the absence of data outside of VA care applies to both TPS and control groups. Further, the total costs of operating a TPS program at any given institution will depend on the size of the hospital and volume of surgical patients who meet criteria for enrollment. However, the relative differences in health care use and costs may be extrapolated to patients undergoing orthopedic surgery in other types of academic and community-based health care systems.
Furthermore, this analysis focused primarily on COU and NOU patients undergoing orthopedic joint surgery. While this represents a high-risk population for OUD, the costs and health care use associated with delivering the TPS intervention to other types of surgical procedures may be significantly different. All costs in this analysis were based on 2019 estimates and do not account for the potential inflation over the past several years. Nonmonetary costs to the patient and per-person average total intervention costs were not included in the study. However, we assumed that costs associated with TPS and standard of care would have increased to an equivalent degree over the same period. Further, the average cost of TPS per patient (approximately $900) is relatively small compared with the average annual costs during 1-year pre- and postoperative periods and was not expected to have a significant effect on the analysis.
Conclusions
We found that the significant reduction in COU seen in previous studies following the implementation of TPS was not accompanied by increased health care costs.6,7 When considering the other costs of long-term opioid use, such as abuse potential, overdose, death, and increased disability, implementation of a TPS service has the potential to improve patient quality of life while reducing other health-related costs. Health care systems should consider the implementation of similar multidisciplinary approaches to perioperative pain management to improve outcomes after orthopedic joint surgery and other high-risk procedures.
1. Rudd RA, Seth P, David F, et al. Increases in drug and opioid-involved overdose deaths—United States, 2010-2015. MMWR Morb Mortal Wkly Rep. 2016;65(50-51):1445-1452. doi:10.15585/mmwr.mm655051e1
2. Florence CS, Zhou C, Luo F, Xu L. The economic burden of prescription opioid overdose, abuse, and dependence in the United States, 2013. Med Care. 2016;54(10):901-906. doi:10.1097/MLR.0000000000000625
3. Jiang X, Orton M, Feng R, et al. Chronic opioid usage in surgical patients in a large academic center. Ann Surg. 2017;265(4):722-727. doi:10.1097/SLA.0000000000001780
4. Johnson SP, Chung KC, Zhong L, et al. Risk of prolonged opioid use among opioid-naive patients following common hand surgery procedures. J Hand Surg Am. 2016;41(10):947-957, e3. doi:10.1016/j.jhsa.2016.07.113
5. Brummett CM, Waljee JF, Goesling J, et al. New persistent opioid use after minor and major surgical procedures in US adults. JAMA Surg. 2017;152(6):e170504. doi:10.1001/jamasurg.2017.0504
6. Buys MJ, Bayless K, Romesser J, et al. Multidisciplinary transitional pain service for the veteran population. Fed Pract. 2020;37(10):472-478. doi:10.12788/fp.0053
7. Buys MJ, Bayless K, Romesser J, et al. Opioid use among veterans undergoing major joint surgery managed by a multidisciplinary transitional pain service. Reg Anesth Pain Med. 2020;45(11):847-852. doi:10.1136/rapm-2020-101797
8. Huang A, Katz J, Clarke H. Ensuring safe prescribing of controlled substances for pain following surgery by developing a transitional pain service. Pain Manag. 2015;5(2):97-105. doi:10.2217/pmt.15.7
9. Katz J, Weinrib A, Fashler SR, et al. The Toronto General Hospital Transitional Pain Service: development and implementation of a multidisciplinary program to prevent chronic postsurgical pain. J Pain Res. 2015;8:695-702. doi:10.2147/JPR.S91924
10. Fihn SD, Francis J, Clancy C, et al. Insights from advanced analytics at the Veterans Health Administration. Health Aff (Millwood). 2014;33(7):1203-1211. doi:10.1377/hlthaff.2014.0054
11. Agency for Healthcare Research and Quality. Clinical Classifications Software Refined (CCSR). Updated December 9, 2022. Accessed October 30, 2023. www.hcup-us.ahrq.gov/toolssoftware/ccsr/ccs_refined.jsp
12. Mosher HJ, Richardson KK, Lund BC. The 1-year treatment course of new opioid recipients in Veterans Health Administration. Pain Med. 2016;17(7):1282-1291. doi:10.1093/pm/pnw058
13. Hadlandsmyth K, Mosher HJ, Vander Weg MW, O’Shea AM, McCoy KD, Lund BC. Utility of accumulated opioid supply days and individual patient factors in predicting probability of transitioning to long-term opioid use: an observational study in the Veterans Health Administration. Pharmacol Res Perspect. 2020;8(2):e00571. doi:10.1002/prp2.571
14. Pagé MG, Kudrina I, Zomahoun HTV, et al. Relative frequency and risk factors for long-term opioid therapy following surgery and trauma among adults: a systematic review protocol. Syst Rev. 2018;7(1):97. doi:10.1186/s13643-018-0760-3
15. US. Bureau of Economic Analysis. Price indexes for personal consumption expenditures by major type of product. Accessed October 30, 2023. https://apps.bea.gov/iTable/?reqid=19&step=3&isuri=1&nipa_table_list=64&categories=survey
16. Brummett CM, Evans-Shields J, England C, et al. Increased health care costs associated with new persistent opioid use after major surgery in opioid-naive patients. J Manag Care Spec Pharm. 2021;27(6):760-771. doi:10.18553/jmcp.2021.20507
17. Gold LS, Strassels SA, Hansen RN. Health care costs and utilization in patients receiving prescriptions for long-acting opioids for acute postsurgical pain. Clin J Pain. 2016;32(9):747-754. doi:10.1097/ajp.0000000000000322
1. Rudd RA, Seth P, David F, et al. Increases in drug and opioid-involved overdose deaths—United States, 2010-2015. MMWR Morb Mortal Wkly Rep. 2016;65(50-51):1445-1452. doi:10.15585/mmwr.mm655051e1
2. Florence CS, Zhou C, Luo F, Xu L. The economic burden of prescription opioid overdose, abuse, and dependence in the United States, 2013. Med Care. 2016;54(10):901-906. doi:10.1097/MLR.0000000000000625
3. Jiang X, Orton M, Feng R, et al. Chronic opioid usage in surgical patients in a large academic center. Ann Surg. 2017;265(4):722-727. doi:10.1097/SLA.0000000000001780
4. Johnson SP, Chung KC, Zhong L, et al. Risk of prolonged opioid use among opioid-naive patients following common hand surgery procedures. J Hand Surg Am. 2016;41(10):947-957, e3. doi:10.1016/j.jhsa.2016.07.113
5. Brummett CM, Waljee JF, Goesling J, et al. New persistent opioid use after minor and major surgical procedures in US adults. JAMA Surg. 2017;152(6):e170504. doi:10.1001/jamasurg.2017.0504
6. Buys MJ, Bayless K, Romesser J, et al. Multidisciplinary transitional pain service for the veteran population. Fed Pract. 2020;37(10):472-478. doi:10.12788/fp.0053
7. Buys MJ, Bayless K, Romesser J, et al. Opioid use among veterans undergoing major joint surgery managed by a multidisciplinary transitional pain service. Reg Anesth Pain Med. 2020;45(11):847-852. doi:10.1136/rapm-2020-101797
8. Huang A, Katz J, Clarke H. Ensuring safe prescribing of controlled substances for pain following surgery by developing a transitional pain service. Pain Manag. 2015;5(2):97-105. doi:10.2217/pmt.15.7
9. Katz J, Weinrib A, Fashler SR, et al. The Toronto General Hospital Transitional Pain Service: development and implementation of a multidisciplinary program to prevent chronic postsurgical pain. J Pain Res. 2015;8:695-702. doi:10.2147/JPR.S91924
10. Fihn SD, Francis J, Clancy C, et al. Insights from advanced analytics at the Veterans Health Administration. Health Aff (Millwood). 2014;33(7):1203-1211. doi:10.1377/hlthaff.2014.0054
11. Agency for Healthcare Research and Quality. Clinical Classifications Software Refined (CCSR). Updated December 9, 2022. Accessed October 30, 2023. www.hcup-us.ahrq.gov/toolssoftware/ccsr/ccs_refined.jsp
12. Mosher HJ, Richardson KK, Lund BC. The 1-year treatment course of new opioid recipients in Veterans Health Administration. Pain Med. 2016;17(7):1282-1291. doi:10.1093/pm/pnw058
13. Hadlandsmyth K, Mosher HJ, Vander Weg MW, O’Shea AM, McCoy KD, Lund BC. Utility of accumulated opioid supply days and individual patient factors in predicting probability of transitioning to long-term opioid use: an observational study in the Veterans Health Administration. Pharmacol Res Perspect. 2020;8(2):e00571. doi:10.1002/prp2.571
14. Pagé MG, Kudrina I, Zomahoun HTV, et al. Relative frequency and risk factors for long-term opioid therapy following surgery and trauma among adults: a systematic review protocol. Syst Rev. 2018;7(1):97. doi:10.1186/s13643-018-0760-3
15. US. Bureau of Economic Analysis. Price indexes for personal consumption expenditures by major type of product. Accessed October 30, 2023. https://apps.bea.gov/iTable/?reqid=19&step=3&isuri=1&nipa_table_list=64&categories=survey
16. Brummett CM, Evans-Shields J, England C, et al. Increased health care costs associated with new persistent opioid use after major surgery in opioid-naive patients. J Manag Care Spec Pharm. 2021;27(6):760-771. doi:10.18553/jmcp.2021.20507
17. Gold LS, Strassels SA, Hansen RN. Health care costs and utilization in patients receiving prescriptions for long-acting opioids for acute postsurgical pain. Clin J Pain. 2016;32(9):747-754. doi:10.1097/ajp.0000000000000322
Despite effective therapies, fibroid care still lacking
In 2022, two colleagues from Johns Hopkins University, Baltimore, Bhuchitra Singh, MD, MPH, MS, MBA, and James Segars Jr., MD, reviewed the available literature to evaluate the effectiveness of newer minimally invasive therapies in reducing bleeding and improving the quality of life and control of symptoms linked to uterine fibroids.
Their goal, according to Dr. Segars, a professor of obstetrics and gynecology and director of the division of women’s health research at Johns Hopkins, was to help guide clinicians and patients in making decisions about the use of the newer therapies, including radiofrequency ablation and ultrasound-guided removal of lesions.
But he and Dr. Singh, the director of clinical research at the Howard W. and Georgeanna Seegar Jones Laboratory of Reproductive Sciences and Women’s Health Research, were surprised by their findings. “The outcomes were relatively the same,” Dr. Segars said. “All of the modalities lead to significant reduction in bleeding and other fibroid-related symptoms.”
The data on long-term complications and risk for recurrence are sparse for some of the newer approaches, and not enough high-quality long-term studies have been conducted for the Food and Drug Administration to approve them as fertility-sparing treatments.
But perhaps, the biggest challenge now is to ensure that women can take advantage of these newer therapies, with large gaps in both the diagnosis of fibroids and geographic access to minimally invasive treatments.
A widespread condition widely underdiagnosed
Uterine fibroids occur in most women (the incidence rises with age) and can be found in up to 70% of women by the time they reach menopause. Risk factors include family history, increasing interval since last birth, hypertension, and obesity. Increasing parity and use of oral contraceptives are protective.
But as many as 50% of cases go undiagnosed, and one reason for this is the failure of clinicians to dig deeply enough into women’s menstrual histories to diagnose fibroids.
“The most common cause of anemia is heavy menstrual bleeding,” said Shannon Laughlin-Tommaso, MD, MPH, a professor of obstetrics and gynecology at Mayo Clinic in Rochester, Minn. She frequently sees patients who have already undergone colonoscopy to work-up the source of their anemia before anyone suspects that fibroids are the culprit.
“When women tell us about their periods, what they’ve been told is normal [bleeding] – or what they’ve always had and considered normal – is actually kind of on the heavier spectrum,” she said.
Ideally, treatment for uterine fibroids would fix abnormally prolonged or heavy menstrual bleeding, relieve pain, and ameliorate symptoms associated with an enlarged uterus, such as pelvic pressure, urinary frequency, and constipation. And the fibroids would never recur.
By those measures, hysterectomy fits the bill: Success rates in relieving symptoms are high, and the risk for recurrence is zero. But the procedure carries significant drawbacks: short-term complications of surgery, including infection, bleeding, and injury to the bowels and bladder along with potential long-term risks for cardiovascular disease, cancer, ovarian failure and premature menopause, depression, and decline in cognitive function. Those factors loom even larger for women who still hope to have children.
For that reason, the American College of Obstetricians and Gynecologists recommends myomectomy, or surgical removal of individual fibroids, for women who desire uterine preservation or future pregnancy. And the literature here is solid, according to Dr. Singh, who found that 95% of myomectomy patients achieved control of their bleeding symptoms, whether it was via laparoscopy, hysteroscopy, or laparotomy. Up to 40% of women may develop new fibroids, or leiomyomas, within 3 years, although only 12.2% required a second surgery up to after 5 years.
But myomectomy is invasive, requiring general anesthesia, incisions in the uterus, and stitches to close the organ.
Newer techniques have emerged that can effectively treat symptoms of fibroids without requiring surgery. Uterine artery embolization (UAE), which involves passing a catheter into the femoral artery, or laparoscopic uterine artery occlusion can be used to cut off the blood supply of the fibroid. Other techniques, including focused ultrasound surgery and radiofrequency ablation (RFA), use various forms of energy to heat and ablate fibroids. The latter two can be performed in outpatient settings and often without general anesthesia.
Approved for use in 1994, UAE has the most data available, with reduction in the volume of fibroids and uterine tissue lasting up to 5 years, and rates of reintervention of 19%-38% between 2 and 5 years after the procedure. Dr. Singh’s review found that 79%-98.5% of recipients of the procedure reported declines in bleeding that persisted for several years, which is comparable to myomectomy. Quality of life and pain scores also showed good improvement, with follow-up in the different studies ranging from 12 months to over 5 years, the analysis showed.
UAE does have its drawbacks. In rare cases, embolization can deprive the entire uterus and ovaries of blood, which can cause ovarian dysfunction and potentially result in premature menopause, although this outcome is most common in women who are older than 45 years. The procedure can often also be painful enough that overnight hospitalization is required.
Focused ultrasound surgeries, which include magnetic resonance–guided focused ultrasound surgery (MRgFUS) and high-intensity focused ultrasound (HIFU), were approved by the FDA in 2004. Focused ultrasound waves pass through the abdominal wall and produce significant heating, causing a burn that destroys the targeted tissue without damaging surrounding tissue. As with UAE, improvements in fibroid-associated bleeding and measures of quality of life were similar to those after myomectomy up to 3 years later.
But Dr. Singh noted that both focused ultrasound and RFA can damage the skin or internal organs. “[As] always with the thermal interventions, there is the probability of skin as well as internal organs that might get the thermal energy if it’s not focused correctly on to the fibroid itself,” he said. In addition, MRgFUS is not an option for women who are not good candidates to undergo an MRI, such as those with claustrophobia or pacemakers.
Also, with focus ultrasound and RFA, “we do worry about that fibroid getting blood flow back,” which can lead to recurrence of heavy menstrual bleeding, Dr. Laughlin-Tommaso noted.
Although data on RFA are limited to 12 months of follow-up, most women reported meaningful reductions in bleeding symptoms. Longer follow-up has been reported for bleeding symptoms after MRgFUS, with similar results up to 3 years later.
For Leslie Hansen-Lindner, MD, chief of obstetrics and gynecology at Atrium Health in Charlotte, N.C., choosing the right procedure starts with a patient-centered conversation weighing the pros and cons of the options and the woman’s goals.
“Is their goal to reduce the size and impact of their fibroid, bleed less, and have a better quality of life on their period?” Dr. Hansen-Lindner said. “Or is their goal to have the entire fibroid removed?”
If the former, an RFA is appealing to many women. If the latter, laparoscopic or mini-laparotomy myomectomy might be a better choice. Although fewer than 10% of patients require surgical reintervention at 3 years of follow-up for RFA, myomectomy has more consistent long-term evidence showing that fewer women require re-intervention and preserve their fertility, she added.
Age also plays a role in the decision: The closer a woman is to menopause, the less likely she is to experience a recurrence, so a less-invasive procedure is preferable. But for younger women hoping to become pregnant, the lower risk for recurrence and good prognosis for future fertility might sway the choice toward myomectomy.
The first laparoscopic RFA procedures were approved for uterine fibroids in 2012. Dr. Hansen-Lindner is a proponent of transcervical fibroid ablation (TFA), a newer RFA procedure that the FDA approved in 2018. Performed through the cervix, TFA requires no incisions and can generally be done without general anesthesia. Eligible candidates would be any woman with symptomatic fibroids, such as heavy menstrual bleeding, pain, or bulk symptoms. The contraindications are few.
“It’s going to come down to size and location of fibroids, and whether or not they would be accessible by the TFA,” Dr. Hansen-Lindner said. “I have to make sure that there isn’t a fibroid blocking their cervix and that the fibroids are accessible with this device.”
TFA also is not suitable for removing most submucosal lesions, which typically must be removed by hysteroscopic myomectomy. Dr. Hansen-Lindner said that she often uses TFA in conjunction with hysteroscopic myomectomy for this scenario. Although data on pregnancy after RFA (including TFA), MRgFUS, and HIFU are lacking, Gynesonics, the manufacturer of the Sonata System (the device that delivers radiofrequency energy to shrink the fibroid) has documented 79 pregnancies among the 2,200 women who have undergone TFA in the United States since 2018.
Disparities hampering care
Uterine fibroids are a particular problem for Black women, whose symptoms are more likely to be ignored by clinicians, according to Jodie Katon, PhD, a core investigator at the Veterans Affairs Greater Los Angeles Center for the Study of Healthcare Innovation, Implementation and Policy. Dr. Katon cited studies in which Black women interviewed about their experiences reported a consistent theme: Clinicians dismissed their symptoms, told them these were nothing to worry about, and advised them to lose weight. Those interactions not only delayed diagnosis among Black women but also led many of them to mistrust clinicians and avoid the health care system altogether.
The failure of clinicians to take their complaints seriously is just one of the disparities affecting Black women. In reviewing the literature, Dr. Laughlin-Tommaso, who also serves as the associate dean for Education Diversity, Equity, and Inclusion at the Mayo Clinic, found that African American women experience two to three times the risk for fibroids, compared with White women, as well as earlier onset and more severe disease, as measured by number and size of the lesions.
According to Dr. Katon, the etiology of fibroids is still poorly understood. “What we do know is that Black women are disproportionately exposed to a variety of factors that we have shown through observational studies are associated with increased risk of development of uterine fibroids.”
The list includes factors like stress; interpersonal racism; early age at menarche; various indicators of poor diets, such as vitamin D deficiency; the use of certain beauty products, specifically hair straighteners; as well as exposure to air pollution and other environmental toxins.
Laughlin-Tommaso also pointed to historical disparities in management, citing a doubled risk for hysterectomy for Black women in a study published in 2007 despite survey data suggesting that Black women report being more interested in uterine-preserving therapies rather than a hysterectomy.
Breaking down barriers of access to new treatments
Dr. Laughlin-Tommaso looked at more recent trends in the management of fibroids using data from the multicenter COMPARE-UF study, which enrolled women between 2015 and 2020 undergoing fibroid treatment into a longitudinal registry to track their outcomes. She found that Black women underwent hysterectomies at a lower rate than did White women and were instead more likely to undergo myomectomy or UAE.
Some of the change may reflect lack of approved minimally invasive procedures before 2000. “But now that we have expanded options, I think most women are opting not to have a hysterectomy,” Dr. Laughlin-Tommaso said.
Dr. Katon has research funding from the VA to look more closely at racial disparities in the treatment of fibroids. In a study published in April 2023, she reported some surprising trends.
During the period from 2010 to 2018, she found that Black veterans diagnosed with fibroids were less likely than White veterans were to receive treatment, regardless of their age or the severity of their symptoms. This finding held even among women with anemia, which should have been a clear indication for treatment.
But, as in the COMPARE-UF study, the subset of Black veterans who received an interventional treatment were less likely than their White peers were to undergo hysterectomy in favor of a fertility-sparing treatment as their initial procedure. Dr. Katon called it a “welcome but unexpected finding.”
But another significant barrier remains: The two newest types of procedures, RFA and guided focused ultrasound, are not commonly performed outside of tertiary care facilities. However, studies have found that all these procedures are cost effective (studies for myomectomy, UAE, MRgFUS, and TFA). The implementation of a category 1 billing code for laparoscopic RFA in 2017 has led more insurance companies to cover the service, and a category 1 code will be available for TFA effective January 2024.
Although RFA does require investment in specialized equipment, which limits facilities from offering the procedure, any gynecologist who routinely performs hysteroscopy can easily learn to do TFA. And the VA, which is committed to eliminating disparities in women’s health, established a 2-year advanced fellowship in minimally invasive gynecologic surgery in 2022 to help expand their capacity to offer these procedures.
The VA has been rapidly expanding their gynecology services, and Katon said that she is confident that ultrasound-guided procedures and RFA will become more available within the system. “I would say we’re keeping pace. And in some ways, you know, as a national system we may be positioned to actually outpace the rest of the U.S.”
Dr. Segars reported prior research funding for clinical trials from BioSpecifics Technologies, Bayer, Allergan, AbbVie, and ObsEva and currently receives funding from Myovant Sciences. Dr. Hansen-Lindner reported personal fees from Gynesonics. Dr. Singh, Dr. Laughlin-Tommaso, and Dr. Katon reported no financial conflicts of interest.
A version of this article first appeared on Medscape.com.
In 2022, two colleagues from Johns Hopkins University, Baltimore, Bhuchitra Singh, MD, MPH, MS, MBA, and James Segars Jr., MD, reviewed the available literature to evaluate the effectiveness of newer minimally invasive therapies in reducing bleeding and improving the quality of life and control of symptoms linked to uterine fibroids.
Their goal, according to Dr. Segars, a professor of obstetrics and gynecology and director of the division of women’s health research at Johns Hopkins, was to help guide clinicians and patients in making decisions about the use of the newer therapies, including radiofrequency ablation and ultrasound-guided removal of lesions.
But he and Dr. Singh, the director of clinical research at the Howard W. and Georgeanna Seegar Jones Laboratory of Reproductive Sciences and Women’s Health Research, were surprised by their findings. “The outcomes were relatively the same,” Dr. Segars said. “All of the modalities lead to significant reduction in bleeding and other fibroid-related symptoms.”
The data on long-term complications and risk for recurrence are sparse for some of the newer approaches, and not enough high-quality long-term studies have been conducted for the Food and Drug Administration to approve them as fertility-sparing treatments.
But perhaps, the biggest challenge now is to ensure that women can take advantage of these newer therapies, with large gaps in both the diagnosis of fibroids and geographic access to minimally invasive treatments.
A widespread condition widely underdiagnosed
Uterine fibroids occur in most women (the incidence rises with age) and can be found in up to 70% of women by the time they reach menopause. Risk factors include family history, increasing interval since last birth, hypertension, and obesity. Increasing parity and use of oral contraceptives are protective.
But as many as 50% of cases go undiagnosed, and one reason for this is the failure of clinicians to dig deeply enough into women’s menstrual histories to diagnose fibroids.
“The most common cause of anemia is heavy menstrual bleeding,” said Shannon Laughlin-Tommaso, MD, MPH, a professor of obstetrics and gynecology at Mayo Clinic in Rochester, Minn. She frequently sees patients who have already undergone colonoscopy to work-up the source of their anemia before anyone suspects that fibroids are the culprit.
“When women tell us about their periods, what they’ve been told is normal [bleeding] – or what they’ve always had and considered normal – is actually kind of on the heavier spectrum,” she said.
Ideally, treatment for uterine fibroids would fix abnormally prolonged or heavy menstrual bleeding, relieve pain, and ameliorate symptoms associated with an enlarged uterus, such as pelvic pressure, urinary frequency, and constipation. And the fibroids would never recur.
By those measures, hysterectomy fits the bill: Success rates in relieving symptoms are high, and the risk for recurrence is zero. But the procedure carries significant drawbacks: short-term complications of surgery, including infection, bleeding, and injury to the bowels and bladder along with potential long-term risks for cardiovascular disease, cancer, ovarian failure and premature menopause, depression, and decline in cognitive function. Those factors loom even larger for women who still hope to have children.
For that reason, the American College of Obstetricians and Gynecologists recommends myomectomy, or surgical removal of individual fibroids, for women who desire uterine preservation or future pregnancy. And the literature here is solid, according to Dr. Singh, who found that 95% of myomectomy patients achieved control of their bleeding symptoms, whether it was via laparoscopy, hysteroscopy, or laparotomy. Up to 40% of women may develop new fibroids, or leiomyomas, within 3 years, although only 12.2% required a second surgery up to after 5 years.
But myomectomy is invasive, requiring general anesthesia, incisions in the uterus, and stitches to close the organ.
Newer techniques have emerged that can effectively treat symptoms of fibroids without requiring surgery. Uterine artery embolization (UAE), which involves passing a catheter into the femoral artery, or laparoscopic uterine artery occlusion can be used to cut off the blood supply of the fibroid. Other techniques, including focused ultrasound surgery and radiofrequency ablation (RFA), use various forms of energy to heat and ablate fibroids. The latter two can be performed in outpatient settings and often without general anesthesia.
Approved for use in 1994, UAE has the most data available, with reduction in the volume of fibroids and uterine tissue lasting up to 5 years, and rates of reintervention of 19%-38% between 2 and 5 years after the procedure. Dr. Singh’s review found that 79%-98.5% of recipients of the procedure reported declines in bleeding that persisted for several years, which is comparable to myomectomy. Quality of life and pain scores also showed good improvement, with follow-up in the different studies ranging from 12 months to over 5 years, the analysis showed.
UAE does have its drawbacks. In rare cases, embolization can deprive the entire uterus and ovaries of blood, which can cause ovarian dysfunction and potentially result in premature menopause, although this outcome is most common in women who are older than 45 years. The procedure can often also be painful enough that overnight hospitalization is required.
Focused ultrasound surgeries, which include magnetic resonance–guided focused ultrasound surgery (MRgFUS) and high-intensity focused ultrasound (HIFU), were approved by the FDA in 2004. Focused ultrasound waves pass through the abdominal wall and produce significant heating, causing a burn that destroys the targeted tissue without damaging surrounding tissue. As with UAE, improvements in fibroid-associated bleeding and measures of quality of life were similar to those after myomectomy up to 3 years later.
But Dr. Singh noted that both focused ultrasound and RFA can damage the skin or internal organs. “[As] always with the thermal interventions, there is the probability of skin as well as internal organs that might get the thermal energy if it’s not focused correctly on to the fibroid itself,” he said. In addition, MRgFUS is not an option for women who are not good candidates to undergo an MRI, such as those with claustrophobia or pacemakers.
Also, with focus ultrasound and RFA, “we do worry about that fibroid getting blood flow back,” which can lead to recurrence of heavy menstrual bleeding, Dr. Laughlin-Tommaso noted.
Although data on RFA are limited to 12 months of follow-up, most women reported meaningful reductions in bleeding symptoms. Longer follow-up has been reported for bleeding symptoms after MRgFUS, with similar results up to 3 years later.
For Leslie Hansen-Lindner, MD, chief of obstetrics and gynecology at Atrium Health in Charlotte, N.C., choosing the right procedure starts with a patient-centered conversation weighing the pros and cons of the options and the woman’s goals.
“Is their goal to reduce the size and impact of their fibroid, bleed less, and have a better quality of life on their period?” Dr. Hansen-Lindner said. “Or is their goal to have the entire fibroid removed?”
If the former, an RFA is appealing to many women. If the latter, laparoscopic or mini-laparotomy myomectomy might be a better choice. Although fewer than 10% of patients require surgical reintervention at 3 years of follow-up for RFA, myomectomy has more consistent long-term evidence showing that fewer women require re-intervention and preserve their fertility, she added.
Age also plays a role in the decision: The closer a woman is to menopause, the less likely she is to experience a recurrence, so a less-invasive procedure is preferable. But for younger women hoping to become pregnant, the lower risk for recurrence and good prognosis for future fertility might sway the choice toward myomectomy.
The first laparoscopic RFA procedures were approved for uterine fibroids in 2012. Dr. Hansen-Lindner is a proponent of transcervical fibroid ablation (TFA), a newer RFA procedure that the FDA approved in 2018. Performed through the cervix, TFA requires no incisions and can generally be done without general anesthesia. Eligible candidates would be any woman with symptomatic fibroids, such as heavy menstrual bleeding, pain, or bulk symptoms. The contraindications are few.
“It’s going to come down to size and location of fibroids, and whether or not they would be accessible by the TFA,” Dr. Hansen-Lindner said. “I have to make sure that there isn’t a fibroid blocking their cervix and that the fibroids are accessible with this device.”
TFA also is not suitable for removing most submucosal lesions, which typically must be removed by hysteroscopic myomectomy. Dr. Hansen-Lindner said that she often uses TFA in conjunction with hysteroscopic myomectomy for this scenario. Although data on pregnancy after RFA (including TFA), MRgFUS, and HIFU are lacking, Gynesonics, the manufacturer of the Sonata System (the device that delivers radiofrequency energy to shrink the fibroid) has documented 79 pregnancies among the 2,200 women who have undergone TFA in the United States since 2018.
Disparities hampering care
Uterine fibroids are a particular problem for Black women, whose symptoms are more likely to be ignored by clinicians, according to Jodie Katon, PhD, a core investigator at the Veterans Affairs Greater Los Angeles Center for the Study of Healthcare Innovation, Implementation and Policy. Dr. Katon cited studies in which Black women interviewed about their experiences reported a consistent theme: Clinicians dismissed their symptoms, told them these were nothing to worry about, and advised them to lose weight. Those interactions not only delayed diagnosis among Black women but also led many of them to mistrust clinicians and avoid the health care system altogether.
The failure of clinicians to take their complaints seriously is just one of the disparities affecting Black women. In reviewing the literature, Dr. Laughlin-Tommaso, who also serves as the associate dean for Education Diversity, Equity, and Inclusion at the Mayo Clinic, found that African American women experience two to three times the risk for fibroids, compared with White women, as well as earlier onset and more severe disease, as measured by number and size of the lesions.
According to Dr. Katon, the etiology of fibroids is still poorly understood. “What we do know is that Black women are disproportionately exposed to a variety of factors that we have shown through observational studies are associated with increased risk of development of uterine fibroids.”
The list includes factors like stress; interpersonal racism; early age at menarche; various indicators of poor diets, such as vitamin D deficiency; the use of certain beauty products, specifically hair straighteners; as well as exposure to air pollution and other environmental toxins.
Laughlin-Tommaso also pointed to historical disparities in management, citing a doubled risk for hysterectomy for Black women in a study published in 2007 despite survey data suggesting that Black women report being more interested in uterine-preserving therapies rather than a hysterectomy.
Breaking down barriers of access to new treatments
Dr. Laughlin-Tommaso looked at more recent trends in the management of fibroids using data from the multicenter COMPARE-UF study, which enrolled women between 2015 and 2020 undergoing fibroid treatment into a longitudinal registry to track their outcomes. She found that Black women underwent hysterectomies at a lower rate than did White women and were instead more likely to undergo myomectomy or UAE.
Some of the change may reflect lack of approved minimally invasive procedures before 2000. “But now that we have expanded options, I think most women are opting not to have a hysterectomy,” Dr. Laughlin-Tommaso said.
Dr. Katon has research funding from the VA to look more closely at racial disparities in the treatment of fibroids. In a study published in April 2023, she reported some surprising trends.
During the period from 2010 to 2018, she found that Black veterans diagnosed with fibroids were less likely than White veterans were to receive treatment, regardless of their age or the severity of their symptoms. This finding held even among women with anemia, which should have been a clear indication for treatment.
But, as in the COMPARE-UF study, the subset of Black veterans who received an interventional treatment were less likely than their White peers were to undergo hysterectomy in favor of a fertility-sparing treatment as their initial procedure. Dr. Katon called it a “welcome but unexpected finding.”
But another significant barrier remains: The two newest types of procedures, RFA and guided focused ultrasound, are not commonly performed outside of tertiary care facilities. However, studies have found that all these procedures are cost effective (studies for myomectomy, UAE, MRgFUS, and TFA). The implementation of a category 1 billing code for laparoscopic RFA in 2017 has led more insurance companies to cover the service, and a category 1 code will be available for TFA effective January 2024.
Although RFA does require investment in specialized equipment, which limits facilities from offering the procedure, any gynecologist who routinely performs hysteroscopy can easily learn to do TFA. And the VA, which is committed to eliminating disparities in women’s health, established a 2-year advanced fellowship in minimally invasive gynecologic surgery in 2022 to help expand their capacity to offer these procedures.
The VA has been rapidly expanding their gynecology services, and Katon said that she is confident that ultrasound-guided procedures and RFA will become more available within the system. “I would say we’re keeping pace. And in some ways, you know, as a national system we may be positioned to actually outpace the rest of the U.S.”
Dr. Segars reported prior research funding for clinical trials from BioSpecifics Technologies, Bayer, Allergan, AbbVie, and ObsEva and currently receives funding from Myovant Sciences. Dr. Hansen-Lindner reported personal fees from Gynesonics. Dr. Singh, Dr. Laughlin-Tommaso, and Dr. Katon reported no financial conflicts of interest.
A version of this article first appeared on Medscape.com.
In 2022, two colleagues from Johns Hopkins University, Baltimore, Bhuchitra Singh, MD, MPH, MS, MBA, and James Segars Jr., MD, reviewed the available literature to evaluate the effectiveness of newer minimally invasive therapies in reducing bleeding and improving the quality of life and control of symptoms linked to uterine fibroids.
Their goal, according to Dr. Segars, a professor of obstetrics and gynecology and director of the division of women’s health research at Johns Hopkins, was to help guide clinicians and patients in making decisions about the use of the newer therapies, including radiofrequency ablation and ultrasound-guided removal of lesions.
But he and Dr. Singh, the director of clinical research at the Howard W. and Georgeanna Seegar Jones Laboratory of Reproductive Sciences and Women’s Health Research, were surprised by their findings. “The outcomes were relatively the same,” Dr. Segars said. “All of the modalities lead to significant reduction in bleeding and other fibroid-related symptoms.”
The data on long-term complications and risk for recurrence are sparse for some of the newer approaches, and not enough high-quality long-term studies have been conducted for the Food and Drug Administration to approve them as fertility-sparing treatments.
But perhaps, the biggest challenge now is to ensure that women can take advantage of these newer therapies, with large gaps in both the diagnosis of fibroids and geographic access to minimally invasive treatments.
A widespread condition widely underdiagnosed
Uterine fibroids occur in most women (the incidence rises with age) and can be found in up to 70% of women by the time they reach menopause. Risk factors include family history, increasing interval since last birth, hypertension, and obesity. Increasing parity and use of oral contraceptives are protective.
But as many as 50% of cases go undiagnosed, and one reason for this is the failure of clinicians to dig deeply enough into women’s menstrual histories to diagnose fibroids.
“The most common cause of anemia is heavy menstrual bleeding,” said Shannon Laughlin-Tommaso, MD, MPH, a professor of obstetrics and gynecology at Mayo Clinic in Rochester, Minn. She frequently sees patients who have already undergone colonoscopy to work-up the source of their anemia before anyone suspects that fibroids are the culprit.
“When women tell us about their periods, what they’ve been told is normal [bleeding] – or what they’ve always had and considered normal – is actually kind of on the heavier spectrum,” she said.
Ideally, treatment for uterine fibroids would fix abnormally prolonged or heavy menstrual bleeding, relieve pain, and ameliorate symptoms associated with an enlarged uterus, such as pelvic pressure, urinary frequency, and constipation. And the fibroids would never recur.
By those measures, hysterectomy fits the bill: Success rates in relieving symptoms are high, and the risk for recurrence is zero. But the procedure carries significant drawbacks: short-term complications of surgery, including infection, bleeding, and injury to the bowels and bladder along with potential long-term risks for cardiovascular disease, cancer, ovarian failure and premature menopause, depression, and decline in cognitive function. Those factors loom even larger for women who still hope to have children.
For that reason, the American College of Obstetricians and Gynecologists recommends myomectomy, or surgical removal of individual fibroids, for women who desire uterine preservation or future pregnancy. And the literature here is solid, according to Dr. Singh, who found that 95% of myomectomy patients achieved control of their bleeding symptoms, whether it was via laparoscopy, hysteroscopy, or laparotomy. Up to 40% of women may develop new fibroids, or leiomyomas, within 3 years, although only 12.2% required a second surgery up to after 5 years.
But myomectomy is invasive, requiring general anesthesia, incisions in the uterus, and stitches to close the organ.
Newer techniques have emerged that can effectively treat symptoms of fibroids without requiring surgery. Uterine artery embolization (UAE), which involves passing a catheter into the femoral artery, or laparoscopic uterine artery occlusion can be used to cut off the blood supply of the fibroid. Other techniques, including focused ultrasound surgery and radiofrequency ablation (RFA), use various forms of energy to heat and ablate fibroids. The latter two can be performed in outpatient settings and often without general anesthesia.
Approved for use in 1994, UAE has the most data available, with reduction in the volume of fibroids and uterine tissue lasting up to 5 years, and rates of reintervention of 19%-38% between 2 and 5 years after the procedure. Dr. Singh’s review found that 79%-98.5% of recipients of the procedure reported declines in bleeding that persisted for several years, which is comparable to myomectomy. Quality of life and pain scores also showed good improvement, with follow-up in the different studies ranging from 12 months to over 5 years, the analysis showed.
UAE does have its drawbacks. In rare cases, embolization can deprive the entire uterus and ovaries of blood, which can cause ovarian dysfunction and potentially result in premature menopause, although this outcome is most common in women who are older than 45 years. The procedure can often also be painful enough that overnight hospitalization is required.
Focused ultrasound surgeries, which include magnetic resonance–guided focused ultrasound surgery (MRgFUS) and high-intensity focused ultrasound (HIFU), were approved by the FDA in 2004. Focused ultrasound waves pass through the abdominal wall and produce significant heating, causing a burn that destroys the targeted tissue without damaging surrounding tissue. As with UAE, improvements in fibroid-associated bleeding and measures of quality of life were similar to those after myomectomy up to 3 years later.
But Dr. Singh noted that both focused ultrasound and RFA can damage the skin or internal organs. “[As] always with the thermal interventions, there is the probability of skin as well as internal organs that might get the thermal energy if it’s not focused correctly on to the fibroid itself,” he said. In addition, MRgFUS is not an option for women who are not good candidates to undergo an MRI, such as those with claustrophobia or pacemakers.
Also, with focus ultrasound and RFA, “we do worry about that fibroid getting blood flow back,” which can lead to recurrence of heavy menstrual bleeding, Dr. Laughlin-Tommaso noted.
Although data on RFA are limited to 12 months of follow-up, most women reported meaningful reductions in bleeding symptoms. Longer follow-up has been reported for bleeding symptoms after MRgFUS, with similar results up to 3 years later.
For Leslie Hansen-Lindner, MD, chief of obstetrics and gynecology at Atrium Health in Charlotte, N.C., choosing the right procedure starts with a patient-centered conversation weighing the pros and cons of the options and the woman’s goals.
“Is their goal to reduce the size and impact of their fibroid, bleed less, and have a better quality of life on their period?” Dr. Hansen-Lindner said. “Or is their goal to have the entire fibroid removed?”
If the former, an RFA is appealing to many women. If the latter, laparoscopic or mini-laparotomy myomectomy might be a better choice. Although fewer than 10% of patients require surgical reintervention at 3 years of follow-up for RFA, myomectomy has more consistent long-term evidence showing that fewer women require re-intervention and preserve their fertility, she added.
Age also plays a role in the decision: The closer a woman is to menopause, the less likely she is to experience a recurrence, so a less-invasive procedure is preferable. But for younger women hoping to become pregnant, the lower risk for recurrence and good prognosis for future fertility might sway the choice toward myomectomy.
The first laparoscopic RFA procedures were approved for uterine fibroids in 2012. Dr. Hansen-Lindner is a proponent of transcervical fibroid ablation (TFA), a newer RFA procedure that the FDA approved in 2018. Performed through the cervix, TFA requires no incisions and can generally be done without general anesthesia. Eligible candidates would be any woman with symptomatic fibroids, such as heavy menstrual bleeding, pain, or bulk symptoms. The contraindications are few.
“It’s going to come down to size and location of fibroids, and whether or not they would be accessible by the TFA,” Dr. Hansen-Lindner said. “I have to make sure that there isn’t a fibroid blocking their cervix and that the fibroids are accessible with this device.”
TFA also is not suitable for removing most submucosal lesions, which typically must be removed by hysteroscopic myomectomy. Dr. Hansen-Lindner said that she often uses TFA in conjunction with hysteroscopic myomectomy for this scenario. Although data on pregnancy after RFA (including TFA), MRgFUS, and HIFU are lacking, Gynesonics, the manufacturer of the Sonata System (the device that delivers radiofrequency energy to shrink the fibroid) has documented 79 pregnancies among the 2,200 women who have undergone TFA in the United States since 2018.
Disparities hampering care
Uterine fibroids are a particular problem for Black women, whose symptoms are more likely to be ignored by clinicians, according to Jodie Katon, PhD, a core investigator at the Veterans Affairs Greater Los Angeles Center for the Study of Healthcare Innovation, Implementation and Policy. Dr. Katon cited studies in which Black women interviewed about their experiences reported a consistent theme: Clinicians dismissed their symptoms, told them these were nothing to worry about, and advised them to lose weight. Those interactions not only delayed diagnosis among Black women but also led many of them to mistrust clinicians and avoid the health care system altogether.
The failure of clinicians to take their complaints seriously is just one of the disparities affecting Black women. In reviewing the literature, Dr. Laughlin-Tommaso, who also serves as the associate dean for Education Diversity, Equity, and Inclusion at the Mayo Clinic, found that African American women experience two to three times the risk for fibroids, compared with White women, as well as earlier onset and more severe disease, as measured by number and size of the lesions.
According to Dr. Katon, the etiology of fibroids is still poorly understood. “What we do know is that Black women are disproportionately exposed to a variety of factors that we have shown through observational studies are associated with increased risk of development of uterine fibroids.”
The list includes factors like stress; interpersonal racism; early age at menarche; various indicators of poor diets, such as vitamin D deficiency; the use of certain beauty products, specifically hair straighteners; as well as exposure to air pollution and other environmental toxins.
Laughlin-Tommaso also pointed to historical disparities in management, citing a doubled risk for hysterectomy for Black women in a study published in 2007 despite survey data suggesting that Black women report being more interested in uterine-preserving therapies rather than a hysterectomy.
Breaking down barriers of access to new treatments
Dr. Laughlin-Tommaso looked at more recent trends in the management of fibroids using data from the multicenter COMPARE-UF study, which enrolled women between 2015 and 2020 undergoing fibroid treatment into a longitudinal registry to track their outcomes. She found that Black women underwent hysterectomies at a lower rate than did White women and were instead more likely to undergo myomectomy or UAE.
Some of the change may reflect lack of approved minimally invasive procedures before 2000. “But now that we have expanded options, I think most women are opting not to have a hysterectomy,” Dr. Laughlin-Tommaso said.
Dr. Katon has research funding from the VA to look more closely at racial disparities in the treatment of fibroids. In a study published in April 2023, she reported some surprising trends.
During the period from 2010 to 2018, she found that Black veterans diagnosed with fibroids were less likely than White veterans were to receive treatment, regardless of their age or the severity of their symptoms. This finding held even among women with anemia, which should have been a clear indication for treatment.
But, as in the COMPARE-UF study, the subset of Black veterans who received an interventional treatment were less likely than their White peers were to undergo hysterectomy in favor of a fertility-sparing treatment as their initial procedure. Dr. Katon called it a “welcome but unexpected finding.”
But another significant barrier remains: The two newest types of procedures, RFA and guided focused ultrasound, are not commonly performed outside of tertiary care facilities. However, studies have found that all these procedures are cost effective (studies for myomectomy, UAE, MRgFUS, and TFA). The implementation of a category 1 billing code for laparoscopic RFA in 2017 has led more insurance companies to cover the service, and a category 1 code will be available for TFA effective January 2024.
Although RFA does require investment in specialized equipment, which limits facilities from offering the procedure, any gynecologist who routinely performs hysteroscopy can easily learn to do TFA. And the VA, which is committed to eliminating disparities in women’s health, established a 2-year advanced fellowship in minimally invasive gynecologic surgery in 2022 to help expand their capacity to offer these procedures.
The VA has been rapidly expanding their gynecology services, and Katon said that she is confident that ultrasound-guided procedures and RFA will become more available within the system. “I would say we’re keeping pace. And in some ways, you know, as a national system we may be positioned to actually outpace the rest of the U.S.”
Dr. Segars reported prior research funding for clinical trials from BioSpecifics Technologies, Bayer, Allergan, AbbVie, and ObsEva and currently receives funding from Myovant Sciences. Dr. Hansen-Lindner reported personal fees from Gynesonics. Dr. Singh, Dr. Laughlin-Tommaso, and Dr. Katon reported no financial conflicts of interest.
A version of this article first appeared on Medscape.com.
Chronic pain in the United States: New data
data from the Centers for Disease Control and Prevention show.
Results from the annual National Health Interview Survey (NHIS) show that over-the-counter (OTC) pain relievers were the most commonly used pharmacologic treatment and exercise was the most common choice among nonpharmacologic options.
The results also revealed that prescription opioid use for chronic pain decreased from 15.2% in 2019 to 13.5% in 2020. However, there was no corresponding increase in nonpharmacologic therapies, despite current CDC guidelines that recommend maximizing the use of medication alternatives.
“Public health efforts may reduce health inequities by increasing access to pain management therapies so that all persons with chronic pain can receive safe and effective care,” S. Michaela Rikard, PhD, and colleagues wrote.
The findings were published online in a research letter in Annals of Internal Medicine.
Among 31,500 survey respondents, 7,400 indicated that they had pain on most days or every day for the past 3 months.
The survey collected data on self-reported opioid prescriptions in the past 3 months, as well as prescription and nonprescription opiate use during the same time period.
Among adult respondents, 60% used a combination of pharmacologic and nonpharmacologic treatments for pain and almost 27% used medications alone. Older adults, those with low incomes, uninsured individuals, and those living in the South were among those least likely to turn to nonpharmacologic treatment for pain.
After exercise, complementary therapies were the most commonly used nonpharmacologic options, including massage, meditation, or guided imagery, and spinal manipulation or other forms of chiropractic care.
For those taking medications, 76% self-reported using OTC pain relievers for pain, followed by prescription nonopioids (31%) and prescription opioids (13.5%).
Of those who used both pharmacologic and nonpharmacologic therapies, about half reported nonopioid and nonpharmacologic therapy use and 8% reported combined use of opioids, nonopioids, and nonpharmacologic therapy.
After adjustment for multiple factors, investigators found those who were older, had public insurance, or had more severe pain were more likely to use prescription opioids. They also reported severe pain (22%), but 4% reported only mild pain.
Study limitations included generalizability only to noninstitutionalized civilian adults, potential recall bias, and cross-sectional results that do not include patient or treatment history.
“Despite its limitations, this study identifies opportunities to improve guideline-concordant use of pharmacologic and nonpharmacologic therapies among adults with chronic pain,” the authors wrote.
There was no specific funding source for the study. The authors have reported no relevant financial disclosures.
A version of this article first appeared on Medscape.com.
data from the Centers for Disease Control and Prevention show.
Results from the annual National Health Interview Survey (NHIS) show that over-the-counter (OTC) pain relievers were the most commonly used pharmacologic treatment and exercise was the most common choice among nonpharmacologic options.
The results also revealed that prescription opioid use for chronic pain decreased from 15.2% in 2019 to 13.5% in 2020. However, there was no corresponding increase in nonpharmacologic therapies, despite current CDC guidelines that recommend maximizing the use of medication alternatives.
“Public health efforts may reduce health inequities by increasing access to pain management therapies so that all persons with chronic pain can receive safe and effective care,” S. Michaela Rikard, PhD, and colleagues wrote.
The findings were published online in a research letter in Annals of Internal Medicine.
Among 31,500 survey respondents, 7,400 indicated that they had pain on most days or every day for the past 3 months.
The survey collected data on self-reported opioid prescriptions in the past 3 months, as well as prescription and nonprescription opiate use during the same time period.
Among adult respondents, 60% used a combination of pharmacologic and nonpharmacologic treatments for pain and almost 27% used medications alone. Older adults, those with low incomes, uninsured individuals, and those living in the South were among those least likely to turn to nonpharmacologic treatment for pain.
After exercise, complementary therapies were the most commonly used nonpharmacologic options, including massage, meditation, or guided imagery, and spinal manipulation or other forms of chiropractic care.
For those taking medications, 76% self-reported using OTC pain relievers for pain, followed by prescription nonopioids (31%) and prescription opioids (13.5%).
Of those who used both pharmacologic and nonpharmacologic therapies, about half reported nonopioid and nonpharmacologic therapy use and 8% reported combined use of opioids, nonopioids, and nonpharmacologic therapy.
After adjustment for multiple factors, investigators found those who were older, had public insurance, or had more severe pain were more likely to use prescription opioids. They also reported severe pain (22%), but 4% reported only mild pain.
Study limitations included generalizability only to noninstitutionalized civilian adults, potential recall bias, and cross-sectional results that do not include patient or treatment history.
“Despite its limitations, this study identifies opportunities to improve guideline-concordant use of pharmacologic and nonpharmacologic therapies among adults with chronic pain,” the authors wrote.
There was no specific funding source for the study. The authors have reported no relevant financial disclosures.
A version of this article first appeared on Medscape.com.
data from the Centers for Disease Control and Prevention show.
Results from the annual National Health Interview Survey (NHIS) show that over-the-counter (OTC) pain relievers were the most commonly used pharmacologic treatment and exercise was the most common choice among nonpharmacologic options.
The results also revealed that prescription opioid use for chronic pain decreased from 15.2% in 2019 to 13.5% in 2020. However, there was no corresponding increase in nonpharmacologic therapies, despite current CDC guidelines that recommend maximizing the use of medication alternatives.
“Public health efforts may reduce health inequities by increasing access to pain management therapies so that all persons with chronic pain can receive safe and effective care,” S. Michaela Rikard, PhD, and colleagues wrote.
The findings were published online in a research letter in Annals of Internal Medicine.
Among 31,500 survey respondents, 7,400 indicated that they had pain on most days or every day for the past 3 months.
The survey collected data on self-reported opioid prescriptions in the past 3 months, as well as prescription and nonprescription opiate use during the same time period.
Among adult respondents, 60% used a combination of pharmacologic and nonpharmacologic treatments for pain and almost 27% used medications alone. Older adults, those with low incomes, uninsured individuals, and those living in the South were among those least likely to turn to nonpharmacologic treatment for pain.
After exercise, complementary therapies were the most commonly used nonpharmacologic options, including massage, meditation, or guided imagery, and spinal manipulation or other forms of chiropractic care.
For those taking medications, 76% self-reported using OTC pain relievers for pain, followed by prescription nonopioids (31%) and prescription opioids (13.5%).
Of those who used both pharmacologic and nonpharmacologic therapies, about half reported nonopioid and nonpharmacologic therapy use and 8% reported combined use of opioids, nonopioids, and nonpharmacologic therapy.
After adjustment for multiple factors, investigators found those who were older, had public insurance, or had more severe pain were more likely to use prescription opioids. They also reported severe pain (22%), but 4% reported only mild pain.
Study limitations included generalizability only to noninstitutionalized civilian adults, potential recall bias, and cross-sectional results that do not include patient or treatment history.
“Despite its limitations, this study identifies opportunities to improve guideline-concordant use of pharmacologic and nonpharmacologic therapies among adults with chronic pain,” the authors wrote.
There was no specific funding source for the study. The authors have reported no relevant financial disclosures.
A version of this article first appeared on Medscape.com.
FROM ANNALS OF INTERNAL MEDICINE
Adolescents with migraine need smooth handoff to adult care
, according to a headache specialist who treats adults and children and spoke at the 2023 Scottsdale Headache Symposium.
“I would start at about the age of 15 or 16,” said Hope L. O’Brien, MD, Headache Center of Hope, University of Cincinnati.
Describing the steps that she thinks should be included in an effective transition, Dr. O’Brien maintained, “you will have a greater chance of successful transition and lessen the likelihood of the chronicity and the poor outcomes that we see in adults.”
Dr. O’Brien, who developed a headache clinic that serves individuals between the ages of 15 and 27, has substantial experience with headache patients in this age range. She acknowledged that there are no guideline recommendations for how best to guide the transition from pediatric to adult care, but she has developed some strategies at her own institution, including a tool for determining when the transition should be considered.
“Transition readiness is something that you need to think about,” she said. “You don’t just do it [automatically] at the age of 18.”
TRAQ questionnaire is helpful
The Transition Readiness Assessment Questionnaire (TRAQ) is one tool that can be helpful, according to Dr. O’Brien, This tool, which can be used to evaluate whether young patients feel prepared to describe their own health status and needs and advocate on their own behalf, is not specific to headache, but the principle is particularly important in headache because of the importance of the patient’s history. Dr. O’Brien said that a fellow in her program, Allyson Bazarsky, MD, who is now affiliated with the University of Vermont Medical Center, Burlington, validated TRAQ for headache about 6 years ago.
“TRAQ is available online. It’s free. You can download it as a PDF,” Dr. O’Brien said. In fact, several age-specific versions can now be found readily on a web search for TRAQ questionnaire.
Ultimately, TRAQ helps the clinician to gauge what patients know about their disease, the medications they are taking, and the relevance of any comorbidities, such as mood disorders. It also provides insight about the ability to understand their health issues and to communicate well with caregivers.
Dr. O’Brien sees this as a process over time, rather than something to be implemented a few months before the transition.
“It is important to start making the shift during childhood and talking directly to the child,” Dr. O’Brien said. If education about the disease and its triggers are started relatively early in adolescence, the transition will not only be easier, but patients might have a chance to understand and control their disease at an earlier age.
With this kind of approach, most children are at least in the preparation stage by age 18 years. However, the age at which patients are suitable for transition varies substantially. Many patients 18 years of age or older are in the “action phase,” meaning it is time to take steps to transition.
Again, based on the interrelationship between headache and comorbidities, particularly mood disorders, such as depression and anxiety, the goal should not be limited to headache. Young adults should be educated about taking responsibility for their overall health.
In addition to educating the patient, Dr. O’Brien recommended preparing a transfer packet, such as the one described in an article published in Headache. Geared for communicating with the clinician who will take over care, the contents should include a detailed medical history along with the current treatment plan and list of medications that have been effective and those that have failed, according to Dr. O’Brien.
“An emergency plan in the form of an emergency department letter in case the patient needs to seek emergent care at an outside facility” is also appropriate, Dr. O’Brien said.
The patient should be aware of what is in the transfer pack in order to participate in an informed discussion of health care with the adult neurologist.
Poor transition linked to poor outcomes
A substantial proportion of adolescents with migraine continue to experience episodes as an adult, particularly those with a delayed diagnosis of migraine, those with a first degree relative who has migraine, and those with poor health habits, but this is not inevitable. Dr. O’Brien noted that “unsuccessful transition of care” into adulthood is a factor associated with poorer outcomes, making it an appropriate target for optimizing outcomes.
“Have that discussion on transfer of care with an action plan and do that early, especially in those with chronic or persistent disability headaches,” Dr. O’Brien emphasized.
This is pertinent advice, according to Amy A. Gelfand, MD, director of the child and adolescent headache program at Benioff Children’s Hospitals, University of California, San Francisco. Senior author of a comprehensive review article on pediatric migraine in Neurologic Clinics, Dr. Gelfand said the practical value of young adults learning what medications they are taking, and why, can place them in a better position to monitor their disease and to understand when a clinical visit is appropriate.
“I agree that it is important to help young adults (i.e., 18- or 19-year-olds) to prepare for the transition from the pediatric health care environment to the adult one,” said Dr. Gelfand, who has written frequently on this and related topics, such as the impact of comorbidities on outcome.
Dr. O’Brien reports financial relationships with AbbVie, Eli Lilly, Guidepoint, Pfizer, and Vector Psychometric Group. Dr. Gelfand reports financial relationships with Allergan, Eli Lilly, EMKinetics, eNeura, Teva and Zosano.
, according to a headache specialist who treats adults and children and spoke at the 2023 Scottsdale Headache Symposium.
“I would start at about the age of 15 or 16,” said Hope L. O’Brien, MD, Headache Center of Hope, University of Cincinnati.
Describing the steps that she thinks should be included in an effective transition, Dr. O’Brien maintained, “you will have a greater chance of successful transition and lessen the likelihood of the chronicity and the poor outcomes that we see in adults.”
Dr. O’Brien, who developed a headache clinic that serves individuals between the ages of 15 and 27, has substantial experience with headache patients in this age range. She acknowledged that there are no guideline recommendations for how best to guide the transition from pediatric to adult care, but she has developed some strategies at her own institution, including a tool for determining when the transition should be considered.
“Transition readiness is something that you need to think about,” she said. “You don’t just do it [automatically] at the age of 18.”
TRAQ questionnaire is helpful
The Transition Readiness Assessment Questionnaire (TRAQ) is one tool that can be helpful, according to Dr. O’Brien, This tool, which can be used to evaluate whether young patients feel prepared to describe their own health status and needs and advocate on their own behalf, is not specific to headache, but the principle is particularly important in headache because of the importance of the patient’s history. Dr. O’Brien said that a fellow in her program, Allyson Bazarsky, MD, who is now affiliated with the University of Vermont Medical Center, Burlington, validated TRAQ for headache about 6 years ago.
“TRAQ is available online. It’s free. You can download it as a PDF,” Dr. O’Brien said. In fact, several age-specific versions can now be found readily on a web search for TRAQ questionnaire.
Ultimately, TRAQ helps the clinician to gauge what patients know about their disease, the medications they are taking, and the relevance of any comorbidities, such as mood disorders. It also provides insight about the ability to understand their health issues and to communicate well with caregivers.
Dr. O’Brien sees this as a process over time, rather than something to be implemented a few months before the transition.
“It is important to start making the shift during childhood and talking directly to the child,” Dr. O’Brien said. If education about the disease and its triggers are started relatively early in adolescence, the transition will not only be easier, but patients might have a chance to understand and control their disease at an earlier age.
With this kind of approach, most children are at least in the preparation stage by age 18 years. However, the age at which patients are suitable for transition varies substantially. Many patients 18 years of age or older are in the “action phase,” meaning it is time to take steps to transition.
Again, based on the interrelationship between headache and comorbidities, particularly mood disorders, such as depression and anxiety, the goal should not be limited to headache. Young adults should be educated about taking responsibility for their overall health.
In addition to educating the patient, Dr. O’Brien recommended preparing a transfer packet, such as the one described in an article published in Headache. Geared for communicating with the clinician who will take over care, the contents should include a detailed medical history along with the current treatment plan and list of medications that have been effective and those that have failed, according to Dr. O’Brien.
“An emergency plan in the form of an emergency department letter in case the patient needs to seek emergent care at an outside facility” is also appropriate, Dr. O’Brien said.
The patient should be aware of what is in the transfer pack in order to participate in an informed discussion of health care with the adult neurologist.
Poor transition linked to poor outcomes
A substantial proportion of adolescents with migraine continue to experience episodes as an adult, particularly those with a delayed diagnosis of migraine, those with a first degree relative who has migraine, and those with poor health habits, but this is not inevitable. Dr. O’Brien noted that “unsuccessful transition of care” into adulthood is a factor associated with poorer outcomes, making it an appropriate target for optimizing outcomes.
“Have that discussion on transfer of care with an action plan and do that early, especially in those with chronic or persistent disability headaches,” Dr. O’Brien emphasized.
This is pertinent advice, according to Amy A. Gelfand, MD, director of the child and adolescent headache program at Benioff Children’s Hospitals, University of California, San Francisco. Senior author of a comprehensive review article on pediatric migraine in Neurologic Clinics, Dr. Gelfand said the practical value of young adults learning what medications they are taking, and why, can place them in a better position to monitor their disease and to understand when a clinical visit is appropriate.
“I agree that it is important to help young adults (i.e., 18- or 19-year-olds) to prepare for the transition from the pediatric health care environment to the adult one,” said Dr. Gelfand, who has written frequently on this and related topics, such as the impact of comorbidities on outcome.
Dr. O’Brien reports financial relationships with AbbVie, Eli Lilly, Guidepoint, Pfizer, and Vector Psychometric Group. Dr. Gelfand reports financial relationships with Allergan, Eli Lilly, EMKinetics, eNeura, Teva and Zosano.
, according to a headache specialist who treats adults and children and spoke at the 2023 Scottsdale Headache Symposium.
“I would start at about the age of 15 or 16,” said Hope L. O’Brien, MD, Headache Center of Hope, University of Cincinnati.
Describing the steps that she thinks should be included in an effective transition, Dr. O’Brien maintained, “you will have a greater chance of successful transition and lessen the likelihood of the chronicity and the poor outcomes that we see in adults.”
Dr. O’Brien, who developed a headache clinic that serves individuals between the ages of 15 and 27, has substantial experience with headache patients in this age range. She acknowledged that there are no guideline recommendations for how best to guide the transition from pediatric to adult care, but she has developed some strategies at her own institution, including a tool for determining when the transition should be considered.
“Transition readiness is something that you need to think about,” she said. “You don’t just do it [automatically] at the age of 18.”
TRAQ questionnaire is helpful
The Transition Readiness Assessment Questionnaire (TRAQ) is one tool that can be helpful, according to Dr. O’Brien, This tool, which can be used to evaluate whether young patients feel prepared to describe their own health status and needs and advocate on their own behalf, is not specific to headache, but the principle is particularly important in headache because of the importance of the patient’s history. Dr. O’Brien said that a fellow in her program, Allyson Bazarsky, MD, who is now affiliated with the University of Vermont Medical Center, Burlington, validated TRAQ for headache about 6 years ago.
“TRAQ is available online. It’s free. You can download it as a PDF,” Dr. O’Brien said. In fact, several age-specific versions can now be found readily on a web search for TRAQ questionnaire.
Ultimately, TRAQ helps the clinician to gauge what patients know about their disease, the medications they are taking, and the relevance of any comorbidities, such as mood disorders. It also provides insight about the ability to understand their health issues and to communicate well with caregivers.
Dr. O’Brien sees this as a process over time, rather than something to be implemented a few months before the transition.
“It is important to start making the shift during childhood and talking directly to the child,” Dr. O’Brien said. If education about the disease and its triggers are started relatively early in adolescence, the transition will not only be easier, but patients might have a chance to understand and control their disease at an earlier age.
With this kind of approach, most children are at least in the preparation stage by age 18 years. However, the age at which patients are suitable for transition varies substantially. Many patients 18 years of age or older are in the “action phase,” meaning it is time to take steps to transition.
Again, based on the interrelationship between headache and comorbidities, particularly mood disorders, such as depression and anxiety, the goal should not be limited to headache. Young adults should be educated about taking responsibility for their overall health.
In addition to educating the patient, Dr. O’Brien recommended preparing a transfer packet, such as the one described in an article published in Headache. Geared for communicating with the clinician who will take over care, the contents should include a detailed medical history along with the current treatment plan and list of medications that have been effective and those that have failed, according to Dr. O’Brien.
“An emergency plan in the form of an emergency department letter in case the patient needs to seek emergent care at an outside facility” is also appropriate, Dr. O’Brien said.
The patient should be aware of what is in the transfer pack in order to participate in an informed discussion of health care with the adult neurologist.
Poor transition linked to poor outcomes
A substantial proportion of adolescents with migraine continue to experience episodes as an adult, particularly those with a delayed diagnosis of migraine, those with a first degree relative who has migraine, and those with poor health habits, but this is not inevitable. Dr. O’Brien noted that “unsuccessful transition of care” into adulthood is a factor associated with poorer outcomes, making it an appropriate target for optimizing outcomes.
“Have that discussion on transfer of care with an action plan and do that early, especially in those with chronic or persistent disability headaches,” Dr. O’Brien emphasized.
This is pertinent advice, according to Amy A. Gelfand, MD, director of the child and adolescent headache program at Benioff Children’s Hospitals, University of California, San Francisco. Senior author of a comprehensive review article on pediatric migraine in Neurologic Clinics, Dr. Gelfand said the practical value of young adults learning what medications they are taking, and why, can place them in a better position to monitor their disease and to understand when a clinical visit is appropriate.
“I agree that it is important to help young adults (i.e., 18- or 19-year-olds) to prepare for the transition from the pediatric health care environment to the adult one,” said Dr. Gelfand, who has written frequently on this and related topics, such as the impact of comorbidities on outcome.
Dr. O’Brien reports financial relationships with AbbVie, Eli Lilly, Guidepoint, Pfizer, and Vector Psychometric Group. Dr. Gelfand reports financial relationships with Allergan, Eli Lilly, EMKinetics, eNeura, Teva and Zosano.
FROM THE 2023 SCOTTSDALE HEADACHE SYMPOSIUM
Avoid adding to minority stress when treating headache in LGBTQIA+ patients
It is “important not to assume that just because someone is a member of the LGBTQ+ community they will need psychiatric or behavioral health support,” said Maya A. Marzouk, PhD, division of behavioral medicine and clinical psychology, Cincinnati Children’s Hospital Medical Center.
Instead, it is useful not to make any assumptions. There is a potential association between minority status and headache susceptibility, but it is more reasonable initially to address the diagnosis and treatment of headache in LGBTQIA+ patients the same way it is addressed in any other patient, Dr. Marzouk said at the 2023 Scottsdale Headache Symposium.
The acronym to describe individuals with gender identities different from male and female and sexual orientations not limited to heterosexuality has been in almost constant evolution over several decades. An addition sign that accompanies LGBTQIA refers to those who do not identify with any letters in the acronym (lesbian, gay, bisexual, transsexual, queer/questioning, intersex, and asexual).
Take steps to normalize the interaction
Although many clinicians have been acclimated to these diverse identifies, not all have risen above preconceptions that become obstacles to effective care, according to Dr. Marzouk. In the context of headache management, Dr. Marzouk emphasized the need to be respectful of the range of gender identities and sexual orientations and to take steps to normalize the interaction.
For example, Dr. Marzouk advised using gender-neutral language at the start of each patient encounter and ask open-ended questions about gender, sexual identify, and pronouns to avoid patient discomfort from misidentification. In turn, the clinicians can establish their own gender identification and preferred pronouns to reinforce the idea that doing so is normal behavior.
This change in approach should be made “for all patients. Do not try to guess who needs them,” she said.
Intake forms and office atmosphere, such as signs and images, should also be welcoming to all patients, she added. Rather than trying to make adjustments for a LGBTQIA+ visit, Dr. Marzouk said a uniform approach helps normalize the experience of LGBTQIA+ patients without singling them out.
Despite the effort to provide an open and welcoming environment, Dr. Marzouk acknowledged that mistakes are difficult to avoid for those with limited experience serving the LGBTQIA+ community. When mistakes are made, she advised clinicians to immediately acknowledge the mistake and ask for guidance from the patient.
The potential offense is making the patient feel “other” or abnormal.
A higher rate of migraine
The interactions that LBGTQIA+ patients have with others outside their community is a possible explanation for the substantial rate of headache as well as headache with comorbid psychiatric disorders in this population.
In a survey published in 2020, the rate of migraine was 19.7% in heterosexual women, 26.7% in lesbians, and 36.8% in bisexual women. Among men, it rose from 9.8% in heterosexuals to 14.8% in gays and then to 22.8% in bisexuals.
Migraine relative to headache is also associated with more mood disorders among LGBTQIA+ individuals. In a study published in 2022, LGBTQIA+ patients with migraine relative to those with headache were more likely to have depression (46.4% vs. 22.3%; P < .001), anxiety (72.1% vs. 51.6%; P < .001), and posttraumatic stress disorder (37.5% vs. 21.4%; P < .001).
A vicious cycle of underdiagnosis and undertreatment
These associations are consistent with minority stress theory, according to Dr. Marzouk. This theory postulates that the associated stress of discrimination, rejection, and microaggressions, such as explicit efforts to make LGBTQIA+ individuals to feel “other,” produces epigenetic changes and dysregulation of the hypothalamic-pituitary-adrenal axis. In turn, this plays a role in the pathogenesis of migraine.
The inconsistency with which minority stress affects LGBTQIA+ patients might be due to relative differences in social support, coping skills, an innate resilience to these effects, Dr. Marzouk explained.
Dr. Marzouk characterized the LGBTQIA+ community as “underserved” for treatment of headache. She suggested that medical mistrust and self-blame among LGBTQIA+ individuals might be factors contributing to a vicious cycle of underdiagnosis and undertreatment. Efforts by the medical community to reach out to the LGBTQIA+ community are appropriate to address an unmet need.
“Individuals with psychiatric comorbidities may experience even more benefit from migraine care,” she said.
Clinical studies should be more inclusive
While agreeing in principle with these remarks, Eric A. Kaiser, MD, PhD, department of neurology, University of Pennsylvania, Philadelphia, said that this area would be better advanced if studies routinely included patients with diverse-gender identities and sexual orientations. Speaking about how to organize these studies, Dr. Kaiser suggested that enrollment criteria should explicitly seek these individuals and that these differences should be captured in the baseline characteristics.
“For example, gender options could include man, woman, non-binary, gender diverse, gender nonconforming, or gender nonspecified,” he said.
To close “the significant knowledge gap that exists in managing headache disorders in sexually- and gender- diverse people,” Dr. Kaiser said that clinical research studies, like patient treatment of diverse populations, “should be conducted with welcoming and affirming practices.”
Dr. Marzouk reported no potential conflicts of interest. Dr. Kaiser reported financial relationships with Amgen and Lundbeck.
It is “important not to assume that just because someone is a member of the LGBTQ+ community they will need psychiatric or behavioral health support,” said Maya A. Marzouk, PhD, division of behavioral medicine and clinical psychology, Cincinnati Children’s Hospital Medical Center.
Instead, it is useful not to make any assumptions. There is a potential association between minority status and headache susceptibility, but it is more reasonable initially to address the diagnosis and treatment of headache in LGBTQIA+ patients the same way it is addressed in any other patient, Dr. Marzouk said at the 2023 Scottsdale Headache Symposium.
The acronym to describe individuals with gender identities different from male and female and sexual orientations not limited to heterosexuality has been in almost constant evolution over several decades. An addition sign that accompanies LGBTQIA refers to those who do not identify with any letters in the acronym (lesbian, gay, bisexual, transsexual, queer/questioning, intersex, and asexual).
Take steps to normalize the interaction
Although many clinicians have been acclimated to these diverse identifies, not all have risen above preconceptions that become obstacles to effective care, according to Dr. Marzouk. In the context of headache management, Dr. Marzouk emphasized the need to be respectful of the range of gender identities and sexual orientations and to take steps to normalize the interaction.
For example, Dr. Marzouk advised using gender-neutral language at the start of each patient encounter and ask open-ended questions about gender, sexual identify, and pronouns to avoid patient discomfort from misidentification. In turn, the clinicians can establish their own gender identification and preferred pronouns to reinforce the idea that doing so is normal behavior.
This change in approach should be made “for all patients. Do not try to guess who needs them,” she said.
Intake forms and office atmosphere, such as signs and images, should also be welcoming to all patients, she added. Rather than trying to make adjustments for a LGBTQIA+ visit, Dr. Marzouk said a uniform approach helps normalize the experience of LGBTQIA+ patients without singling them out.
Despite the effort to provide an open and welcoming environment, Dr. Marzouk acknowledged that mistakes are difficult to avoid for those with limited experience serving the LGBTQIA+ community. When mistakes are made, she advised clinicians to immediately acknowledge the mistake and ask for guidance from the patient.
The potential offense is making the patient feel “other” or abnormal.
A higher rate of migraine
The interactions that LBGTQIA+ patients have with others outside their community is a possible explanation for the substantial rate of headache as well as headache with comorbid psychiatric disorders in this population.
In a survey published in 2020, the rate of migraine was 19.7% in heterosexual women, 26.7% in lesbians, and 36.8% in bisexual women. Among men, it rose from 9.8% in heterosexuals to 14.8% in gays and then to 22.8% in bisexuals.
Migraine relative to headache is also associated with more mood disorders among LGBTQIA+ individuals. In a study published in 2022, LGBTQIA+ patients with migraine relative to those with headache were more likely to have depression (46.4% vs. 22.3%; P < .001), anxiety (72.1% vs. 51.6%; P < .001), and posttraumatic stress disorder (37.5% vs. 21.4%; P < .001).
A vicious cycle of underdiagnosis and undertreatment
These associations are consistent with minority stress theory, according to Dr. Marzouk. This theory postulates that the associated stress of discrimination, rejection, and microaggressions, such as explicit efforts to make LGBTQIA+ individuals to feel “other,” produces epigenetic changes and dysregulation of the hypothalamic-pituitary-adrenal axis. In turn, this plays a role in the pathogenesis of migraine.
The inconsistency with which minority stress affects LGBTQIA+ patients might be due to relative differences in social support, coping skills, an innate resilience to these effects, Dr. Marzouk explained.
Dr. Marzouk characterized the LGBTQIA+ community as “underserved” for treatment of headache. She suggested that medical mistrust and self-blame among LGBTQIA+ individuals might be factors contributing to a vicious cycle of underdiagnosis and undertreatment. Efforts by the medical community to reach out to the LGBTQIA+ community are appropriate to address an unmet need.
“Individuals with psychiatric comorbidities may experience even more benefit from migraine care,” she said.
Clinical studies should be more inclusive
While agreeing in principle with these remarks, Eric A. Kaiser, MD, PhD, department of neurology, University of Pennsylvania, Philadelphia, said that this area would be better advanced if studies routinely included patients with diverse-gender identities and sexual orientations. Speaking about how to organize these studies, Dr. Kaiser suggested that enrollment criteria should explicitly seek these individuals and that these differences should be captured in the baseline characteristics.
“For example, gender options could include man, woman, non-binary, gender diverse, gender nonconforming, or gender nonspecified,” he said.
To close “the significant knowledge gap that exists in managing headache disorders in sexually- and gender- diverse people,” Dr. Kaiser said that clinical research studies, like patient treatment of diverse populations, “should be conducted with welcoming and affirming practices.”
Dr. Marzouk reported no potential conflicts of interest. Dr. Kaiser reported financial relationships with Amgen and Lundbeck.
It is “important not to assume that just because someone is a member of the LGBTQ+ community they will need psychiatric or behavioral health support,” said Maya A. Marzouk, PhD, division of behavioral medicine and clinical psychology, Cincinnati Children’s Hospital Medical Center.
Instead, it is useful not to make any assumptions. There is a potential association between minority status and headache susceptibility, but it is more reasonable initially to address the diagnosis and treatment of headache in LGBTQIA+ patients the same way it is addressed in any other patient, Dr. Marzouk said at the 2023 Scottsdale Headache Symposium.
The acronym to describe individuals with gender identities different from male and female and sexual orientations not limited to heterosexuality has been in almost constant evolution over several decades. An addition sign that accompanies LGBTQIA refers to those who do not identify with any letters in the acronym (lesbian, gay, bisexual, transsexual, queer/questioning, intersex, and asexual).
Take steps to normalize the interaction
Although many clinicians have been acclimated to these diverse identifies, not all have risen above preconceptions that become obstacles to effective care, according to Dr. Marzouk. In the context of headache management, Dr. Marzouk emphasized the need to be respectful of the range of gender identities and sexual orientations and to take steps to normalize the interaction.
For example, Dr. Marzouk advised using gender-neutral language at the start of each patient encounter and ask open-ended questions about gender, sexual identify, and pronouns to avoid patient discomfort from misidentification. In turn, the clinicians can establish their own gender identification and preferred pronouns to reinforce the idea that doing so is normal behavior.
This change in approach should be made “for all patients. Do not try to guess who needs them,” she said.
Intake forms and office atmosphere, such as signs and images, should also be welcoming to all patients, she added. Rather than trying to make adjustments for a LGBTQIA+ visit, Dr. Marzouk said a uniform approach helps normalize the experience of LGBTQIA+ patients without singling them out.
Despite the effort to provide an open and welcoming environment, Dr. Marzouk acknowledged that mistakes are difficult to avoid for those with limited experience serving the LGBTQIA+ community. When mistakes are made, she advised clinicians to immediately acknowledge the mistake and ask for guidance from the patient.
The potential offense is making the patient feel “other” or abnormal.
A higher rate of migraine
The interactions that LBGTQIA+ patients have with others outside their community is a possible explanation for the substantial rate of headache as well as headache with comorbid psychiatric disorders in this population.
In a survey published in 2020, the rate of migraine was 19.7% in heterosexual women, 26.7% in lesbians, and 36.8% in bisexual women. Among men, it rose from 9.8% in heterosexuals to 14.8% in gays and then to 22.8% in bisexuals.
Migraine relative to headache is also associated with more mood disorders among LGBTQIA+ individuals. In a study published in 2022, LGBTQIA+ patients with migraine relative to those with headache were more likely to have depression (46.4% vs. 22.3%; P < .001), anxiety (72.1% vs. 51.6%; P < .001), and posttraumatic stress disorder (37.5% vs. 21.4%; P < .001).
A vicious cycle of underdiagnosis and undertreatment
These associations are consistent with minority stress theory, according to Dr. Marzouk. This theory postulates that the associated stress of discrimination, rejection, and microaggressions, such as explicit efforts to make LGBTQIA+ individuals to feel “other,” produces epigenetic changes and dysregulation of the hypothalamic-pituitary-adrenal axis. In turn, this plays a role in the pathogenesis of migraine.
The inconsistency with which minority stress affects LGBTQIA+ patients might be due to relative differences in social support, coping skills, an innate resilience to these effects, Dr. Marzouk explained.
Dr. Marzouk characterized the LGBTQIA+ community as “underserved” for treatment of headache. She suggested that medical mistrust and self-blame among LGBTQIA+ individuals might be factors contributing to a vicious cycle of underdiagnosis and undertreatment. Efforts by the medical community to reach out to the LGBTQIA+ community are appropriate to address an unmet need.
“Individuals with psychiatric comorbidities may experience even more benefit from migraine care,” she said.
Clinical studies should be more inclusive
While agreeing in principle with these remarks, Eric A. Kaiser, MD, PhD, department of neurology, University of Pennsylvania, Philadelphia, said that this area would be better advanced if studies routinely included patients with diverse-gender identities and sexual orientations. Speaking about how to organize these studies, Dr. Kaiser suggested that enrollment criteria should explicitly seek these individuals and that these differences should be captured in the baseline characteristics.
“For example, gender options could include man, woman, non-binary, gender diverse, gender nonconforming, or gender nonspecified,” he said.
To close “the significant knowledge gap that exists in managing headache disorders in sexually- and gender- diverse people,” Dr. Kaiser said that clinical research studies, like patient treatment of diverse populations, “should be conducted with welcoming and affirming practices.”
Dr. Marzouk reported no potential conflicts of interest. Dr. Kaiser reported financial relationships with Amgen and Lundbeck.
FROM THE 2023 SCOTTSDALE HEADACHE SYMPOSIUM
Why aren’t doctors managing pain during gynecologic procedures?
During a fellowship rotation in gynecology, Rebekah D. Fenton, MD, asked the attending physicians what pain management options they could offer patients for insertion of an intrauterine device (IUD). Their answer surprised her: None.
The research on the effectiveness of pain management techniques during the procedure were not strong enough to warrant providing potential relief.
But Dr. Fenton knew the attending physician was wrong: She’d received the drug lidocaine during a recent visit to her own ob.gyn. to get an IUD placed. The local anesthetic enabled her to avoid the experiences of many patients who often withstand debilitating cramping and pain during insertion, side effects that can last for hours after the procedure has ended.
By not teaching her how to administer pain treatment options such as lidocaine gel or injection, “they made the decision for me, whether I could give patients this option,” said Dr. Fenton, now an adolescent medicine specialist at Alivio Medical Center in Chicago.
As a result, patients undergoing IUD placements, biopsies, hysteroscopies, and pelvic exams are often subject to pain that could be mitigated.
Some research suggests simple numbing agents, including lidocaine, may induce less pain without the need for full anesthesia. But clinicians don’t always present these options.
During gynecologic procedures, the amount of pain a patient can expect is often downplayed by clinicians. Because every patient experiences the sensation differently, discussing options for pain management and the range of possible pain is paramount in building patient-clinician trust, and ultimately providing the best care for patients in the long run, according to Megan Wasson, DO, chair of the department of medical and surgical gynecology at Mayo Clinic Arizona in Phoenix.
“It comes down to shared decision-making so the patient is aware of the pain that should be expected and what avenue they want to go down,” Dr. Wasson said. “It’s not a one-size-fits-all.”
Lack of uniform protocols
The American College of Obstetricians and Gynecologists (ACOG) has clear guidelines for pain management during pregnancy and delivery but not for many routine gynecologic procedures. Some experts say not offering options for pain management based on lack of efficacy evidence can undermine a patient’s experience.
ACOG does have recommendations for reducing dilation pain during a hysteroscopy, including providing intravaginal misoprostol and estrogen. The organization also recommends performing a vaginoscopy instead if possible because the procedure is typically less painful than is a hysteroscopy.
For an IUD placement, ACOG states that the procedure “may cause temporary discomfort” and recommends that patients take over-the-counter pain relief before a procedure. The most recent clinical bulletin on the topic, published in 2016, states routine misoprostol is not recommended for IUD placement, although it may be considered with difficult insertions for management of pain.
A clinical inquiry published in 2020 outlined the efficacy of several pain options that practitioners can weigh with patients. The inquiry cited a 2019 meta-analysis of 38 studies that found lidocaine-prilocaine cream to be the most effective option for pain management during IUD placement, reducing insertion pain by nearly 30%. The inquiry concluded that a combination of 600 mcg of misoprostol and 4% lidocaine gel may be effective, while lower dosages of both drugs were not effective. A 2018 clinical trial cited in the analysis found that though a 20-cc 1% lidocaine paracervical block on its own did not reduce pain, the block mixed with sodium bicarbonate reduced pain during IUD insertion by 22%.
Some doctors make the decision to not use lidocaine without offering it to patients first, according to Dr. Fenton. Instead, clinicians should discuss any potential drawbacks, such as pain from administering the numbing agent with a needle or the procedure taking extra time while the patient waits for the lidocaine to kick in.
“That always felt unfair, to make that decision for [the patient],” Dr. Fenton said.
Often clinicians won’t know how a patient will respond to a procedure: A 2014 secondary analysis of a clinical trial compared how patients rated their pain after an IUD procedure to the amount of pain physicians perceived the procedure to cause. They found that the average pain scores patients reported were nearly twice as high as clinician expectations were.
ACOG’s guideline states that the evidence backing paracervical blocks and lidocaine to IUD insertion pain is controversial. The American College of Physicians also cites “low-quality evidence” to support patient reports of pain and discomfort during pelvic exams. Some studies have found up to 60% of women report these negative experiences.
The varying evidence highlights the need for a personalized approach – one that includes patients – to pain management for routine gynecological procedures.
“Usually patients are pretty good predictors,” said Lisa Bayer, MD, MPH, associate professor of obstetrics and gynecology at Oregon Health & Science University in Portland. “They can anticipate what different things are going to feel like based on previous experiences.”
Making patients part of the discussion
Clinicians should have open discussions with patients about their past experiences and current anxieties about a gynecologic procedure, according to Dr. Bayer.
“Part of it is just creating a really safe environment of trust as a medical provider,” she said.
A study published in 2016 of more than 800 patients undergoing oocyte retrieval, which has clear protocols for pain management, found that previous negative gynecologic experiences were significantly correlated to greater amounts of pain reported during the procedure.
If pain isn’t properly managed, patients may avoid care in the future, putting them at risk for unplanned pregnancies, skipped cancer screenings, and complications from undiagnosed conditions and infections, Dr. Bayer added. Clinician offices will not always have access to all pain management options, so making referrals to another physician who has access to the appropriate technique may be the best thing for the patient, Dr. Bayer said.
Downplaying the experience
Informing a patient that she will feel only a little discomfort during a procedure – when a clinician doesn’t know how exactly the patient will react – can also result in distrust.
When a clinician says, “ ’It’s only going to be a little cramp, it’s only going to be a little pinch,’ we know extreme pain is a possibility, we’ve seen it,” Dr. Fenton said. “But if we choose to disregard that [possibility], it feels invalidating for patients.”
Failing to fully explain the possible pain scale can also directly interfere with the procedure at hand.
“My first concern is if they aren’t anticipating the amount of pain they are going to experience, they may move; For biopsies and IUD insertions, we need them to be still,” Dr. Wasson said. “If they are unable to tolerate the procedure, we’ve put them through pain and not been able to accomplish the primary goal.”
Managing both pain and what patients can expect is even more crucial for adolescent and teenage patients who are often having their first gynecologic experience.
“We’re framing what these experiences look like,” Dr. Fenton said. “That means there are opportunities for creating a space that builds trust and security for the patients moving forward.”
A version of this article first appeared on Medscape.com.
During a fellowship rotation in gynecology, Rebekah D. Fenton, MD, asked the attending physicians what pain management options they could offer patients for insertion of an intrauterine device (IUD). Their answer surprised her: None.
The research on the effectiveness of pain management techniques during the procedure were not strong enough to warrant providing potential relief.
But Dr. Fenton knew the attending physician was wrong: She’d received the drug lidocaine during a recent visit to her own ob.gyn. to get an IUD placed. The local anesthetic enabled her to avoid the experiences of many patients who often withstand debilitating cramping and pain during insertion, side effects that can last for hours after the procedure has ended.
By not teaching her how to administer pain treatment options such as lidocaine gel or injection, “they made the decision for me, whether I could give patients this option,” said Dr. Fenton, now an adolescent medicine specialist at Alivio Medical Center in Chicago.
As a result, patients undergoing IUD placements, biopsies, hysteroscopies, and pelvic exams are often subject to pain that could be mitigated.
Some research suggests simple numbing agents, including lidocaine, may induce less pain without the need for full anesthesia. But clinicians don’t always present these options.
During gynecologic procedures, the amount of pain a patient can expect is often downplayed by clinicians. Because every patient experiences the sensation differently, discussing options for pain management and the range of possible pain is paramount in building patient-clinician trust, and ultimately providing the best care for patients in the long run, according to Megan Wasson, DO, chair of the department of medical and surgical gynecology at Mayo Clinic Arizona in Phoenix.
“It comes down to shared decision-making so the patient is aware of the pain that should be expected and what avenue they want to go down,” Dr. Wasson said. “It’s not a one-size-fits-all.”
Lack of uniform protocols
The American College of Obstetricians and Gynecologists (ACOG) has clear guidelines for pain management during pregnancy and delivery but not for many routine gynecologic procedures. Some experts say not offering options for pain management based on lack of efficacy evidence can undermine a patient’s experience.
ACOG does have recommendations for reducing dilation pain during a hysteroscopy, including providing intravaginal misoprostol and estrogen. The organization also recommends performing a vaginoscopy instead if possible because the procedure is typically less painful than is a hysteroscopy.
For an IUD placement, ACOG states that the procedure “may cause temporary discomfort” and recommends that patients take over-the-counter pain relief before a procedure. The most recent clinical bulletin on the topic, published in 2016, states routine misoprostol is not recommended for IUD placement, although it may be considered with difficult insertions for management of pain.
A clinical inquiry published in 2020 outlined the efficacy of several pain options that practitioners can weigh with patients. The inquiry cited a 2019 meta-analysis of 38 studies that found lidocaine-prilocaine cream to be the most effective option for pain management during IUD placement, reducing insertion pain by nearly 30%. The inquiry concluded that a combination of 600 mcg of misoprostol and 4% lidocaine gel may be effective, while lower dosages of both drugs were not effective. A 2018 clinical trial cited in the analysis found that though a 20-cc 1% lidocaine paracervical block on its own did not reduce pain, the block mixed with sodium bicarbonate reduced pain during IUD insertion by 22%.
Some doctors make the decision to not use lidocaine without offering it to patients first, according to Dr. Fenton. Instead, clinicians should discuss any potential drawbacks, such as pain from administering the numbing agent with a needle or the procedure taking extra time while the patient waits for the lidocaine to kick in.
“That always felt unfair, to make that decision for [the patient],” Dr. Fenton said.
Often clinicians won’t know how a patient will respond to a procedure: A 2014 secondary analysis of a clinical trial compared how patients rated their pain after an IUD procedure to the amount of pain physicians perceived the procedure to cause. They found that the average pain scores patients reported were nearly twice as high as clinician expectations were.
ACOG’s guideline states that the evidence backing paracervical blocks and lidocaine to IUD insertion pain is controversial. The American College of Physicians also cites “low-quality evidence” to support patient reports of pain and discomfort during pelvic exams. Some studies have found up to 60% of women report these negative experiences.
The varying evidence highlights the need for a personalized approach – one that includes patients – to pain management for routine gynecological procedures.
“Usually patients are pretty good predictors,” said Lisa Bayer, MD, MPH, associate professor of obstetrics and gynecology at Oregon Health & Science University in Portland. “They can anticipate what different things are going to feel like based on previous experiences.”
Making patients part of the discussion
Clinicians should have open discussions with patients about their past experiences and current anxieties about a gynecologic procedure, according to Dr. Bayer.
“Part of it is just creating a really safe environment of trust as a medical provider,” she said.
A study published in 2016 of more than 800 patients undergoing oocyte retrieval, which has clear protocols for pain management, found that previous negative gynecologic experiences were significantly correlated to greater amounts of pain reported during the procedure.
If pain isn’t properly managed, patients may avoid care in the future, putting them at risk for unplanned pregnancies, skipped cancer screenings, and complications from undiagnosed conditions and infections, Dr. Bayer added. Clinician offices will not always have access to all pain management options, so making referrals to another physician who has access to the appropriate technique may be the best thing for the patient, Dr. Bayer said.
Downplaying the experience
Informing a patient that she will feel only a little discomfort during a procedure – when a clinician doesn’t know how exactly the patient will react – can also result in distrust.
When a clinician says, “ ’It’s only going to be a little cramp, it’s only going to be a little pinch,’ we know extreme pain is a possibility, we’ve seen it,” Dr. Fenton said. “But if we choose to disregard that [possibility], it feels invalidating for patients.”
Failing to fully explain the possible pain scale can also directly interfere with the procedure at hand.
“My first concern is if they aren’t anticipating the amount of pain they are going to experience, they may move; For biopsies and IUD insertions, we need them to be still,” Dr. Wasson said. “If they are unable to tolerate the procedure, we’ve put them through pain and not been able to accomplish the primary goal.”
Managing both pain and what patients can expect is even more crucial for adolescent and teenage patients who are often having their first gynecologic experience.
“We’re framing what these experiences look like,” Dr. Fenton said. “That means there are opportunities for creating a space that builds trust and security for the patients moving forward.”
A version of this article first appeared on Medscape.com.
During a fellowship rotation in gynecology, Rebekah D. Fenton, MD, asked the attending physicians what pain management options they could offer patients for insertion of an intrauterine device (IUD). Their answer surprised her: None.
The research on the effectiveness of pain management techniques during the procedure were not strong enough to warrant providing potential relief.
But Dr. Fenton knew the attending physician was wrong: She’d received the drug lidocaine during a recent visit to her own ob.gyn. to get an IUD placed. The local anesthetic enabled her to avoid the experiences of many patients who often withstand debilitating cramping and pain during insertion, side effects that can last for hours after the procedure has ended.
By not teaching her how to administer pain treatment options such as lidocaine gel or injection, “they made the decision for me, whether I could give patients this option,” said Dr. Fenton, now an adolescent medicine specialist at Alivio Medical Center in Chicago.
As a result, patients undergoing IUD placements, biopsies, hysteroscopies, and pelvic exams are often subject to pain that could be mitigated.
Some research suggests simple numbing agents, including lidocaine, may induce less pain without the need for full anesthesia. But clinicians don’t always present these options.
During gynecologic procedures, the amount of pain a patient can expect is often downplayed by clinicians. Because every patient experiences the sensation differently, discussing options for pain management and the range of possible pain is paramount in building patient-clinician trust, and ultimately providing the best care for patients in the long run, according to Megan Wasson, DO, chair of the department of medical and surgical gynecology at Mayo Clinic Arizona in Phoenix.
“It comes down to shared decision-making so the patient is aware of the pain that should be expected and what avenue they want to go down,” Dr. Wasson said. “It’s not a one-size-fits-all.”
Lack of uniform protocols
The American College of Obstetricians and Gynecologists (ACOG) has clear guidelines for pain management during pregnancy and delivery but not for many routine gynecologic procedures. Some experts say not offering options for pain management based on lack of efficacy evidence can undermine a patient’s experience.
ACOG does have recommendations for reducing dilation pain during a hysteroscopy, including providing intravaginal misoprostol and estrogen. The organization also recommends performing a vaginoscopy instead if possible because the procedure is typically less painful than is a hysteroscopy.
For an IUD placement, ACOG states that the procedure “may cause temporary discomfort” and recommends that patients take over-the-counter pain relief before a procedure. The most recent clinical bulletin on the topic, published in 2016, states routine misoprostol is not recommended for IUD placement, although it may be considered with difficult insertions for management of pain.
A clinical inquiry published in 2020 outlined the efficacy of several pain options that practitioners can weigh with patients. The inquiry cited a 2019 meta-analysis of 38 studies that found lidocaine-prilocaine cream to be the most effective option for pain management during IUD placement, reducing insertion pain by nearly 30%. The inquiry concluded that a combination of 600 mcg of misoprostol and 4% lidocaine gel may be effective, while lower dosages of both drugs were not effective. A 2018 clinical trial cited in the analysis found that though a 20-cc 1% lidocaine paracervical block on its own did not reduce pain, the block mixed with sodium bicarbonate reduced pain during IUD insertion by 22%.
Some doctors make the decision to not use lidocaine without offering it to patients first, according to Dr. Fenton. Instead, clinicians should discuss any potential drawbacks, such as pain from administering the numbing agent with a needle or the procedure taking extra time while the patient waits for the lidocaine to kick in.
“That always felt unfair, to make that decision for [the patient],” Dr. Fenton said.
Often clinicians won’t know how a patient will respond to a procedure: A 2014 secondary analysis of a clinical trial compared how patients rated their pain after an IUD procedure to the amount of pain physicians perceived the procedure to cause. They found that the average pain scores patients reported were nearly twice as high as clinician expectations were.
ACOG’s guideline states that the evidence backing paracervical blocks and lidocaine to IUD insertion pain is controversial. The American College of Physicians also cites “low-quality evidence” to support patient reports of pain and discomfort during pelvic exams. Some studies have found up to 60% of women report these negative experiences.
The varying evidence highlights the need for a personalized approach – one that includes patients – to pain management for routine gynecological procedures.
“Usually patients are pretty good predictors,” said Lisa Bayer, MD, MPH, associate professor of obstetrics and gynecology at Oregon Health & Science University in Portland. “They can anticipate what different things are going to feel like based on previous experiences.”
Making patients part of the discussion
Clinicians should have open discussions with patients about their past experiences and current anxieties about a gynecologic procedure, according to Dr. Bayer.
“Part of it is just creating a really safe environment of trust as a medical provider,” she said.
A study published in 2016 of more than 800 patients undergoing oocyte retrieval, which has clear protocols for pain management, found that previous negative gynecologic experiences were significantly correlated to greater amounts of pain reported during the procedure.
If pain isn’t properly managed, patients may avoid care in the future, putting them at risk for unplanned pregnancies, skipped cancer screenings, and complications from undiagnosed conditions and infections, Dr. Bayer added. Clinician offices will not always have access to all pain management options, so making referrals to another physician who has access to the appropriate technique may be the best thing for the patient, Dr. Bayer said.
Downplaying the experience
Informing a patient that she will feel only a little discomfort during a procedure – when a clinician doesn’t know how exactly the patient will react – can also result in distrust.
When a clinician says, “ ’It’s only going to be a little cramp, it’s only going to be a little pinch,’ we know extreme pain is a possibility, we’ve seen it,” Dr. Fenton said. “But if we choose to disregard that [possibility], it feels invalidating for patients.”
Failing to fully explain the possible pain scale can also directly interfere with the procedure at hand.
“My first concern is if they aren’t anticipating the amount of pain they are going to experience, they may move; For biopsies and IUD insertions, we need them to be still,” Dr. Wasson said. “If they are unable to tolerate the procedure, we’ve put them through pain and not been able to accomplish the primary goal.”
Managing both pain and what patients can expect is even more crucial for adolescent and teenage patients who are often having their first gynecologic experience.
“We’re framing what these experiences look like,” Dr. Fenton said. “That means there are opportunities for creating a space that builds trust and security for the patients moving forward.”
A version of this article first appeared on Medscape.com.
Can thermal biofeedback reduce pain and anxiety?
TOPLINE:
A novel handheld tool may hold promise for reducing anxiety and pain and improving sleep quality, according to research presented at Lifestyle Medicine 2023, the annual meeting of the American College of Lifestyle Medicine.
METHODOLOGY:
- Franklin Somchith Ly, a PhD candidate in mechanical engineering at the University of California, Santa Barbara, developed , a product that assesses blood flow to the hand with an infrared temperature sensor and changes color as blood vessels expand during relaxation.
- Exercises such as intentional breathwork, visualization, and muscle relaxation change the color displayed by the device.
- Mr. Ly examined how measures of anxiety, sleep quality, and chronic pain changed after participants used the instrument. Ten participants completed a study assessing anxiety. Eight participants were enrolled in a sleep study where they completed biofeedback sessions before bed for 2 weeks, and 15 participants performed biofeedback twice daily and reported their levels of anxiety and pain.
TAKEAWAY:
- Anxiety scores decreased by about 22% on average (P < .001).
- Seven of the eight participants in the sleep study had improved scores on the Pittsburgh Sleep Quality Index, with an average improvement of nearly 30% (P < .05). Daytime dysfunction improved by 58% (P < .01).
- In the chronic pain study, about 60% of the 350 biofeedback sessions led to reduced pain.
IN PRACTICE:
“These portable devices may aid lifestyle management by alleviating anxiety, chronic pain, and enhancing daytime energy,” Mr. Ly said. “The results support their integration into lifestyle medicine and integrative medicine.”
SOURCE:
Mr. Ly presented the findings as a poster at Lifestyle Medicine 2023, which took place Oct. 29 to Nov. 1 in Denver and online.
LIMITATIONS:
The studies were open label and did not include control groups.
DISCLOSURES:
Mr. Ly is the founder of CalmStone, which markets a thermal biofeedback device. The research was supported by the Bill and Melinda Gates Foundation and the U.S. Army Research Office and Institute for Collaborative Biotechnologies.
A version of this article first appeared on Medscape.com.
TOPLINE:
A novel handheld tool may hold promise for reducing anxiety and pain and improving sleep quality, according to research presented at Lifestyle Medicine 2023, the annual meeting of the American College of Lifestyle Medicine.
METHODOLOGY:
- Franklin Somchith Ly, a PhD candidate in mechanical engineering at the University of California, Santa Barbara, developed , a product that assesses blood flow to the hand with an infrared temperature sensor and changes color as blood vessels expand during relaxation.
- Exercises such as intentional breathwork, visualization, and muscle relaxation change the color displayed by the device.
- Mr. Ly examined how measures of anxiety, sleep quality, and chronic pain changed after participants used the instrument. Ten participants completed a study assessing anxiety. Eight participants were enrolled in a sleep study where they completed biofeedback sessions before bed for 2 weeks, and 15 participants performed biofeedback twice daily and reported their levels of anxiety and pain.
TAKEAWAY:
- Anxiety scores decreased by about 22% on average (P < .001).
- Seven of the eight participants in the sleep study had improved scores on the Pittsburgh Sleep Quality Index, with an average improvement of nearly 30% (P < .05). Daytime dysfunction improved by 58% (P < .01).
- In the chronic pain study, about 60% of the 350 biofeedback sessions led to reduced pain.
IN PRACTICE:
“These portable devices may aid lifestyle management by alleviating anxiety, chronic pain, and enhancing daytime energy,” Mr. Ly said. “The results support their integration into lifestyle medicine and integrative medicine.”
SOURCE:
Mr. Ly presented the findings as a poster at Lifestyle Medicine 2023, which took place Oct. 29 to Nov. 1 in Denver and online.
LIMITATIONS:
The studies were open label and did not include control groups.
DISCLOSURES:
Mr. Ly is the founder of CalmStone, which markets a thermal biofeedback device. The research was supported by the Bill and Melinda Gates Foundation and the U.S. Army Research Office and Institute for Collaborative Biotechnologies.
A version of this article first appeared on Medscape.com.
TOPLINE:
A novel handheld tool may hold promise for reducing anxiety and pain and improving sleep quality, according to research presented at Lifestyle Medicine 2023, the annual meeting of the American College of Lifestyle Medicine.
METHODOLOGY:
- Franklin Somchith Ly, a PhD candidate in mechanical engineering at the University of California, Santa Barbara, developed , a product that assesses blood flow to the hand with an infrared temperature sensor and changes color as blood vessels expand during relaxation.
- Exercises such as intentional breathwork, visualization, and muscle relaxation change the color displayed by the device.
- Mr. Ly examined how measures of anxiety, sleep quality, and chronic pain changed after participants used the instrument. Ten participants completed a study assessing anxiety. Eight participants were enrolled in a sleep study where they completed biofeedback sessions before bed for 2 weeks, and 15 participants performed biofeedback twice daily and reported their levels of anxiety and pain.
TAKEAWAY:
- Anxiety scores decreased by about 22% on average (P < .001).
- Seven of the eight participants in the sleep study had improved scores on the Pittsburgh Sleep Quality Index, with an average improvement of nearly 30% (P < .05). Daytime dysfunction improved by 58% (P < .01).
- In the chronic pain study, about 60% of the 350 biofeedback sessions led to reduced pain.
IN PRACTICE:
“These portable devices may aid lifestyle management by alleviating anxiety, chronic pain, and enhancing daytime energy,” Mr. Ly said. “The results support their integration into lifestyle medicine and integrative medicine.”
SOURCE:
Mr. Ly presented the findings as a poster at Lifestyle Medicine 2023, which took place Oct. 29 to Nov. 1 in Denver and online.
LIMITATIONS:
The studies were open label and did not include control groups.
DISCLOSURES:
Mr. Ly is the founder of CalmStone, which markets a thermal biofeedback device. The research was supported by the Bill and Melinda Gates Foundation and the U.S. Army Research Office and Institute for Collaborative Biotechnologies.
A version of this article first appeared on Medscape.com.
Childhood trauma linked to adult headache
TOPLINE:
with more early adverse experiences raising the risk even more, a new study found.
METHODOLOGY:
- The meta-analysis included 28 observational studies with 154,739 persons in 19 countries that assessed the relationship between at least one adverse childhood experience (ACE) and primary headache (including migraine, tension-type headache, cluster headache, and chronic/severe headache) at age 21 years or older.
- From each study, researchers extracted outcome point estimates and corresponding 95% confidence intervals, number of events in each group, and covariates included in the model. They subcategorized ACEs according to those involving threat (for example, physical, emotional, or sexual abuse) and deprivation (for example, neglect, household substance misuse).
- For the primary analysis, the researchers calculated the odds ratios and hazard ratios of headache among persons with at least one ACE, compared with those with no ACEs.
- They also tested an underlying biological theory that threat and deprivation ACEs may manifest differently in neurodevelopment, with distinct impacts on primary headaches.
TAKEAWAY:
- The most commonly reported ACEs were physical abuse (77%), sexual abuse (73%), and exposure to family violence (38%).
- Compared with having experienced no ACEs, experiencing at least one was associated with primary headaches (pooled OR, 1.48; 95% confidence interval, 1.36-1.61).
- As the number of ACEs increased, the strength of the association with primary headaches increased in a dose-response relationship (P for trend < .0001).
- Both threat and deprivation were independently associated with primary headaches; the pooled main effect was consistent for threat (OR, 1.46; 95% CI, 1.32-1.60) and for deprivation (OR, 1.35; 95% CI, 1.23-1.49), suggesting possible distinct pathways of early adversity.
IN PRACTICE:
Clinicians who treat primary headaches in adults “should routinely screen for ACEs, educate patients on the connection between ACEs and health, and provide referrals for treatment strategies,” the investigators write. Strategies such as trauma-informed or attachment-based therapy may help rewire parts of the brain that have been dysregulated, they add.
SOURCE:
The study was led by Claudia Sikorski, department of health research methods, evidence, and impact, McMaster University, Hamilton, Ont. It was published online in Neurology.
LIMITATIONS:
The findings reflect a conservative estimate of the true impact of ACEs on primary headaches, because ACEs are commonly underreported. The analysis could not statistically disentangle younger adults with developing brains (age 21-26 years) from older adults. Not all included studies adjusted for age and sex, which are known risk factors for headaches. The study did not explore the relationship between ACEs and primary headache disorders in childhood and adolescence. Owing to the inherent nature of studies investigating ACEs, causation cannot be inferred.
DISCLOSURES:
The authors report no targeted funding and no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
TOPLINE:
with more early adverse experiences raising the risk even more, a new study found.
METHODOLOGY:
- The meta-analysis included 28 observational studies with 154,739 persons in 19 countries that assessed the relationship between at least one adverse childhood experience (ACE) and primary headache (including migraine, tension-type headache, cluster headache, and chronic/severe headache) at age 21 years or older.
- From each study, researchers extracted outcome point estimates and corresponding 95% confidence intervals, number of events in each group, and covariates included in the model. They subcategorized ACEs according to those involving threat (for example, physical, emotional, or sexual abuse) and deprivation (for example, neglect, household substance misuse).
- For the primary analysis, the researchers calculated the odds ratios and hazard ratios of headache among persons with at least one ACE, compared with those with no ACEs.
- They also tested an underlying biological theory that threat and deprivation ACEs may manifest differently in neurodevelopment, with distinct impacts on primary headaches.
TAKEAWAY:
- The most commonly reported ACEs were physical abuse (77%), sexual abuse (73%), and exposure to family violence (38%).
- Compared with having experienced no ACEs, experiencing at least one was associated with primary headaches (pooled OR, 1.48; 95% confidence interval, 1.36-1.61).
- As the number of ACEs increased, the strength of the association with primary headaches increased in a dose-response relationship (P for trend < .0001).
- Both threat and deprivation were independently associated with primary headaches; the pooled main effect was consistent for threat (OR, 1.46; 95% CI, 1.32-1.60) and for deprivation (OR, 1.35; 95% CI, 1.23-1.49), suggesting possible distinct pathways of early adversity.
IN PRACTICE:
Clinicians who treat primary headaches in adults “should routinely screen for ACEs, educate patients on the connection between ACEs and health, and provide referrals for treatment strategies,” the investigators write. Strategies such as trauma-informed or attachment-based therapy may help rewire parts of the brain that have been dysregulated, they add.
SOURCE:
The study was led by Claudia Sikorski, department of health research methods, evidence, and impact, McMaster University, Hamilton, Ont. It was published online in Neurology.
LIMITATIONS:
The findings reflect a conservative estimate of the true impact of ACEs on primary headaches, because ACEs are commonly underreported. The analysis could not statistically disentangle younger adults with developing brains (age 21-26 years) from older adults. Not all included studies adjusted for age and sex, which are known risk factors for headaches. The study did not explore the relationship between ACEs and primary headache disorders in childhood and adolescence. Owing to the inherent nature of studies investigating ACEs, causation cannot be inferred.
DISCLOSURES:
The authors report no targeted funding and no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
TOPLINE:
with more early adverse experiences raising the risk even more, a new study found.
METHODOLOGY:
- The meta-analysis included 28 observational studies with 154,739 persons in 19 countries that assessed the relationship between at least one adverse childhood experience (ACE) and primary headache (including migraine, tension-type headache, cluster headache, and chronic/severe headache) at age 21 years or older.
- From each study, researchers extracted outcome point estimates and corresponding 95% confidence intervals, number of events in each group, and covariates included in the model. They subcategorized ACEs according to those involving threat (for example, physical, emotional, or sexual abuse) and deprivation (for example, neglect, household substance misuse).
- For the primary analysis, the researchers calculated the odds ratios and hazard ratios of headache among persons with at least one ACE, compared with those with no ACEs.
- They also tested an underlying biological theory that threat and deprivation ACEs may manifest differently in neurodevelopment, with distinct impacts on primary headaches.
TAKEAWAY:
- The most commonly reported ACEs were physical abuse (77%), sexual abuse (73%), and exposure to family violence (38%).
- Compared with having experienced no ACEs, experiencing at least one was associated with primary headaches (pooled OR, 1.48; 95% confidence interval, 1.36-1.61).
- As the number of ACEs increased, the strength of the association with primary headaches increased in a dose-response relationship (P for trend < .0001).
- Both threat and deprivation were independently associated with primary headaches; the pooled main effect was consistent for threat (OR, 1.46; 95% CI, 1.32-1.60) and for deprivation (OR, 1.35; 95% CI, 1.23-1.49), suggesting possible distinct pathways of early adversity.
IN PRACTICE:
Clinicians who treat primary headaches in adults “should routinely screen for ACEs, educate patients on the connection between ACEs and health, and provide referrals for treatment strategies,” the investigators write. Strategies such as trauma-informed or attachment-based therapy may help rewire parts of the brain that have been dysregulated, they add.
SOURCE:
The study was led by Claudia Sikorski, department of health research methods, evidence, and impact, McMaster University, Hamilton, Ont. It was published online in Neurology.
LIMITATIONS:
The findings reflect a conservative estimate of the true impact of ACEs on primary headaches, because ACEs are commonly underreported. The analysis could not statistically disentangle younger adults with developing brains (age 21-26 years) from older adults. Not all included studies adjusted for age and sex, which are known risk factors for headaches. The study did not explore the relationship between ACEs and primary headache disorders in childhood and adolescence. Owing to the inherent nature of studies investigating ACEs, causation cannot be inferred.
DISCLOSURES:
The authors report no targeted funding and no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
FDA warns of hidden ingredients in arthritis, pain products
Some of these products contain active ingredients found in anti-inflammatory prescription medication.
“These products may cause potentially serious side effects and may interact with medications or dietary supplements a consumer is taking,” the FDA said in a statement. “It is clear from the results of our decade of testing that retailers and distributors, including online marketplaces, do not effectively prevent these types of potentially harmful products from being sold to consumers.”
Unlike prescription medication and over-the-counter drugs such as loratadine (Claritin) or acetaminophen (Tylenol), supplements do not need FDA approval before they can be sold. Only after a complaint is made or FDA testing reveals illegal or unsafe ingredients can the FDA get involved.
From August 2013 to September 2023, the FDA identified 22 arthritis and pain products with active ingredients not disclosed on the product label. The most common hidden ingredients detected in these supplements were prescription-only corticosteroids, nonsteroidal anti-inflammatory drugs (NSAIDs), and muscle relaxants, said Candy Tsourounis, PharmD, a professor in the department of clinical pharmacy at the University of California, San Francisco.
Kuka Flex Forte and Reumo Flex, both promoted for joint pain and arthritis, both contain the NSAID diclofenac. Tapee Tea, a product promoted for pain relief, contains dexamethasone and piroxicam. AK Forte, also sold for joint pain and arthritis, contains diclofenac, dexamethasone, and methocarbamol not disclosed on the label.
“It is interesting that these products have hidden ingredients that are used to reduce swelling and inflammation,” Dr. Tsourounis said. “I don’t know if this was intentional, but it seems suspicious that a product marketed to reduce joint pain and inflammation contains prescription-only ingredients that are used for this purpose.”
Certain products also contained antihistamines including cyproheptadine and chlorpheniramine.
These types of products are likely targeted toward underserved and immigrant communities, added Pieter Cohen, MD, a primary care physician and an assistant professor of medicine at Harvard Medical School, Boston, who studies dietary supplements. They might be sold in mom-and-pop shops or gas stations to individuals with limited access to health care or insurance, he noted.
The FDA warned that this list included “only a small fraction of the potentially dangerous products marketed to consumers online and in stores. Even if a product is not included in this list, consumers should exercise caution before using these types of arthritis and pain management products.”
Advising patients
Research suggests that most patients do not tell doctors about the supplements they are taking, and often, clinicians do not ask, said Dr. Cohen. “Most of the time it’s a total black box – we don’t know what’s going on,” he added.
He advised raising the subject of supplements in a very nonjudgmental way, particularly when treating patients in marginalized and immigrant communities. One approach he suggested was first mentioning that other patients in your care dealing with joint pain have bought remedies locally or have tried treatments that friends recommend. You can then ask a patient about their own use, framing it as a way to better help with treatment decisions.
Once a clinician understands what their patient is taking, they can then give advice and discuss if a product is safe to combine with prescription drugs, Dr. Cohen said. “If they come down too hard, I think the patients will just clam up and not talk about it anymore,” he said.
If a patient begins to experience side effects or gets sick, a clinician will already be informed of what their patient is taking and can ask that patient to bring the product or supplement in, so they can look over the product together, Dr. Cohen noted. Any side effects or other adverse events potentially related to the use of these products should then be reported to FDA’s MedWatch Safety Information and Adverse Event Reporting Program.
Tips for safe shopping
To make sure supplements and other over-the-counter products are safe to use, Dr. Tsourounis recommends that consumers:
- Buy products from well-known retailers like Target or large pharmacies like CVS or Walgreens.
- Avoid buying products with labels in another language that you cannot read or products with no drug label.
- Be cautious of buying products online or from other countries.
- Look up suspicious products on the FDA’s health fraud database.
- Be wary of any product that offers miracle cures or relies on personal testimonies without evidence.
In general, do not base purchasing decisions on any health claims on a product label because companies selling supplements making these claims “don’t have to have any clinical data to back them up,” Dr. Cohen said.
Dr. Cohen also recommends sticking with individual ingredients. “If you want echinacea, buy echinacea. Don’t buy a complicated mix that is supposed to be good for arthritis with 10 different botanical [ingredients]. That’s more likely to run [you] into trouble,” he said.
Last, Dr. Cohen recommended buying supplements that are certified by NSF International or United States Pharmacopeia, both respected third-party testing organizations. “If it has an NSF International or USP stamp, that gives us more certainty that what’s in the bottle is going to be what’s listed on label,” he said.
Dr. Tsourounis noted that if you are skeptical of a product, you can also try calling the manufacturer number on the product label.
“I always encourage people to call that number to see if somebody answers,” she said. “Sometimes, you can tell a lot about that company just by calling that number.”
Dr. Cohen has received research support from the Consumers Union and PEW Charitable Trusts and royalties from UpToDate. He has collaborated in research with NSF International. Dr. Tsourounis disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Some of these products contain active ingredients found in anti-inflammatory prescription medication.
“These products may cause potentially serious side effects and may interact with medications or dietary supplements a consumer is taking,” the FDA said in a statement. “It is clear from the results of our decade of testing that retailers and distributors, including online marketplaces, do not effectively prevent these types of potentially harmful products from being sold to consumers.”
Unlike prescription medication and over-the-counter drugs such as loratadine (Claritin) or acetaminophen (Tylenol), supplements do not need FDA approval before they can be sold. Only after a complaint is made or FDA testing reveals illegal or unsafe ingredients can the FDA get involved.
From August 2013 to September 2023, the FDA identified 22 arthritis and pain products with active ingredients not disclosed on the product label. The most common hidden ingredients detected in these supplements were prescription-only corticosteroids, nonsteroidal anti-inflammatory drugs (NSAIDs), and muscle relaxants, said Candy Tsourounis, PharmD, a professor in the department of clinical pharmacy at the University of California, San Francisco.
Kuka Flex Forte and Reumo Flex, both promoted for joint pain and arthritis, both contain the NSAID diclofenac. Tapee Tea, a product promoted for pain relief, contains dexamethasone and piroxicam. AK Forte, also sold for joint pain and arthritis, contains diclofenac, dexamethasone, and methocarbamol not disclosed on the label.
“It is interesting that these products have hidden ingredients that are used to reduce swelling and inflammation,” Dr. Tsourounis said. “I don’t know if this was intentional, but it seems suspicious that a product marketed to reduce joint pain and inflammation contains prescription-only ingredients that are used for this purpose.”
Certain products also contained antihistamines including cyproheptadine and chlorpheniramine.
These types of products are likely targeted toward underserved and immigrant communities, added Pieter Cohen, MD, a primary care physician and an assistant professor of medicine at Harvard Medical School, Boston, who studies dietary supplements. They might be sold in mom-and-pop shops or gas stations to individuals with limited access to health care or insurance, he noted.
The FDA warned that this list included “only a small fraction of the potentially dangerous products marketed to consumers online and in stores. Even if a product is not included in this list, consumers should exercise caution before using these types of arthritis and pain management products.”
Advising patients
Research suggests that most patients do not tell doctors about the supplements they are taking, and often, clinicians do not ask, said Dr. Cohen. “Most of the time it’s a total black box – we don’t know what’s going on,” he added.
He advised raising the subject of supplements in a very nonjudgmental way, particularly when treating patients in marginalized and immigrant communities. One approach he suggested was first mentioning that other patients in your care dealing with joint pain have bought remedies locally or have tried treatments that friends recommend. You can then ask a patient about their own use, framing it as a way to better help with treatment decisions.
Once a clinician understands what their patient is taking, they can then give advice and discuss if a product is safe to combine with prescription drugs, Dr. Cohen said. “If they come down too hard, I think the patients will just clam up and not talk about it anymore,” he said.
If a patient begins to experience side effects or gets sick, a clinician will already be informed of what their patient is taking and can ask that patient to bring the product or supplement in, so they can look over the product together, Dr. Cohen noted. Any side effects or other adverse events potentially related to the use of these products should then be reported to FDA’s MedWatch Safety Information and Adverse Event Reporting Program.
Tips for safe shopping
To make sure supplements and other over-the-counter products are safe to use, Dr. Tsourounis recommends that consumers:
- Buy products from well-known retailers like Target or large pharmacies like CVS or Walgreens.
- Avoid buying products with labels in another language that you cannot read or products with no drug label.
- Be cautious of buying products online or from other countries.
- Look up suspicious products on the FDA’s health fraud database.
- Be wary of any product that offers miracle cures or relies on personal testimonies without evidence.
In general, do not base purchasing decisions on any health claims on a product label because companies selling supplements making these claims “don’t have to have any clinical data to back them up,” Dr. Cohen said.
Dr. Cohen also recommends sticking with individual ingredients. “If you want echinacea, buy echinacea. Don’t buy a complicated mix that is supposed to be good for arthritis with 10 different botanical [ingredients]. That’s more likely to run [you] into trouble,” he said.
Last, Dr. Cohen recommended buying supplements that are certified by NSF International or United States Pharmacopeia, both respected third-party testing organizations. “If it has an NSF International or USP stamp, that gives us more certainty that what’s in the bottle is going to be what’s listed on label,” he said.
Dr. Tsourounis noted that if you are skeptical of a product, you can also try calling the manufacturer number on the product label.
“I always encourage people to call that number to see if somebody answers,” she said. “Sometimes, you can tell a lot about that company just by calling that number.”
Dr. Cohen has received research support from the Consumers Union and PEW Charitable Trusts and royalties from UpToDate. He has collaborated in research with NSF International. Dr. Tsourounis disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Some of these products contain active ingredients found in anti-inflammatory prescription medication.
“These products may cause potentially serious side effects and may interact with medications or dietary supplements a consumer is taking,” the FDA said in a statement. “It is clear from the results of our decade of testing that retailers and distributors, including online marketplaces, do not effectively prevent these types of potentially harmful products from being sold to consumers.”
Unlike prescription medication and over-the-counter drugs such as loratadine (Claritin) or acetaminophen (Tylenol), supplements do not need FDA approval before they can be sold. Only after a complaint is made or FDA testing reveals illegal or unsafe ingredients can the FDA get involved.
From August 2013 to September 2023, the FDA identified 22 arthritis and pain products with active ingredients not disclosed on the product label. The most common hidden ingredients detected in these supplements were prescription-only corticosteroids, nonsteroidal anti-inflammatory drugs (NSAIDs), and muscle relaxants, said Candy Tsourounis, PharmD, a professor in the department of clinical pharmacy at the University of California, San Francisco.
Kuka Flex Forte and Reumo Flex, both promoted for joint pain and arthritis, both contain the NSAID diclofenac. Tapee Tea, a product promoted for pain relief, contains dexamethasone and piroxicam. AK Forte, also sold for joint pain and arthritis, contains diclofenac, dexamethasone, and methocarbamol not disclosed on the label.
“It is interesting that these products have hidden ingredients that are used to reduce swelling and inflammation,” Dr. Tsourounis said. “I don’t know if this was intentional, but it seems suspicious that a product marketed to reduce joint pain and inflammation contains prescription-only ingredients that are used for this purpose.”
Certain products also contained antihistamines including cyproheptadine and chlorpheniramine.
These types of products are likely targeted toward underserved and immigrant communities, added Pieter Cohen, MD, a primary care physician and an assistant professor of medicine at Harvard Medical School, Boston, who studies dietary supplements. They might be sold in mom-and-pop shops or gas stations to individuals with limited access to health care or insurance, he noted.
The FDA warned that this list included “only a small fraction of the potentially dangerous products marketed to consumers online and in stores. Even if a product is not included in this list, consumers should exercise caution before using these types of arthritis and pain management products.”
Advising patients
Research suggests that most patients do not tell doctors about the supplements they are taking, and often, clinicians do not ask, said Dr. Cohen. “Most of the time it’s a total black box – we don’t know what’s going on,” he added.
He advised raising the subject of supplements in a very nonjudgmental way, particularly when treating patients in marginalized and immigrant communities. One approach he suggested was first mentioning that other patients in your care dealing with joint pain have bought remedies locally or have tried treatments that friends recommend. You can then ask a patient about their own use, framing it as a way to better help with treatment decisions.
Once a clinician understands what their patient is taking, they can then give advice and discuss if a product is safe to combine with prescription drugs, Dr. Cohen said. “If they come down too hard, I think the patients will just clam up and not talk about it anymore,” he said.
If a patient begins to experience side effects or gets sick, a clinician will already be informed of what their patient is taking and can ask that patient to bring the product or supplement in, so they can look over the product together, Dr. Cohen noted. Any side effects or other adverse events potentially related to the use of these products should then be reported to FDA’s MedWatch Safety Information and Adverse Event Reporting Program.
Tips for safe shopping
To make sure supplements and other over-the-counter products are safe to use, Dr. Tsourounis recommends that consumers:
- Buy products from well-known retailers like Target or large pharmacies like CVS or Walgreens.
- Avoid buying products with labels in another language that you cannot read or products with no drug label.
- Be cautious of buying products online or from other countries.
- Look up suspicious products on the FDA’s health fraud database.
- Be wary of any product that offers miracle cures or relies on personal testimonies without evidence.
In general, do not base purchasing decisions on any health claims on a product label because companies selling supplements making these claims “don’t have to have any clinical data to back them up,” Dr. Cohen said.
Dr. Cohen also recommends sticking with individual ingredients. “If you want echinacea, buy echinacea. Don’t buy a complicated mix that is supposed to be good for arthritis with 10 different botanical [ingredients]. That’s more likely to run [you] into trouble,” he said.
Last, Dr. Cohen recommended buying supplements that are certified by NSF International or United States Pharmacopeia, both respected third-party testing organizations. “If it has an NSF International or USP stamp, that gives us more certainty that what’s in the bottle is going to be what’s listed on label,” he said.
Dr. Tsourounis noted that if you are skeptical of a product, you can also try calling the manufacturer number on the product label.
“I always encourage people to call that number to see if somebody answers,” she said. “Sometimes, you can tell a lot about that company just by calling that number.”
Dr. Cohen has received research support from the Consumers Union and PEW Charitable Trusts and royalties from UpToDate. He has collaborated in research with NSF International. Dr. Tsourounis disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Predictors of prescription opioid overdose
A Canadian systematic review of 28 observational studies has identified 10 strong predictors of fatal and nonfatal prescription opioid overdose.
Published in CMAJ, the analysis found the risk of fatal and nonfatal opioid overdose was notably tied to such factors as high-dose and fentanyl prescriptions, multiple opioid prescribers or pharmacies, and several mental health issues. High-certainty evidence from 14 studies involving more than a million patients showed a linear dose-response relationship with opioid overdose.
“Our findings suggest that awareness of, and attention to, several patient and prescription characteristics may help reduce the risk of opioid overdose among people living with chronic pain,” wrote a research team led by Li Wang, PhD, a researcher at the Michael G. DeGroote Institute for Pain Research and Care and the department of anesthesia at McMaster University, Hamilton, Ont.
Predictors of fatal and nonfatal overdose
Reporting on studies of 103 possible predictors in a pooled cohort of almost 24 million patients, the review found moderate- to high-certainty evidence for large relative associations with the following 10 criteria:
- A history of overdose (odds ratio, 5.85; 95% confidence interval, 3.78-9.04).
- A higher opioid dosage (OR, 2.57; 95% CI, 2.08-3.18 per 90-mg increment).
- Three or more prescribers (OR, 4.68; 95% CI, 3.57-6.12).
- Four or more dispensing pharmacies (OR, 4.92; 95% CI, 4.35-5.57).
- Prescription for fentanyl (OR, 2.80; 95% CI, 2.30-3.41).
- Current substance use disorder (OR, 2.62; 95% CI, 2.09-3.27).
- Any mental health diagnosis (OR, 2.12; 95% CI, 1.73-2.61).
- Depression (OR, 2.22; 95% CI, 1.57-314).
- Bipolar disorder (OR, 2.07; 95% CI, 1.77-2.41).
- Pancreatitis (OR, 2.00; 95% CI,1.52-2.64).
Absolute risks in patients with the predictor ranged from 2 to 6 per 1,000 for fatal overdose and 4 to 12 per 1,000 for nonfatal overdose.
The authors noted that chronic pain affects 20% of the world’s population worldwide, and a 2021 meta-analysis of 60 observational studies revealed that opioids are prescribed for 27% of adults living with chronic pain, with a higher prevalence of prescribing in North America than in Europe.
International review
A total of 28 observational studies comprising 23,963,716 patients (52% female) with mean age of 52 years were enrolled. All used administrative databases. Twenty-four studies were conducted in the United States, three in Canada, and one in the United Kingdom. Twenty-one studies included only patients with noncancer chronic pain, while seven included patients with either cancer-related or noncancer chronic pain. Twenty-two studies accepted patients with previous or current substance use disorder and three excluded patients with comorbid substance use disorder. Twenty-three studies included patients with comorbid mental illness and five exclusively recruited veterans.
The median sample size was 43,885. As a limitation, 25 studies (89%) were at high risk of bias for at least one criterion, the authors acknowledged. Moderate-certainty evidence showed the pooled prevalence of fatal opioid overdose after prescription for chronic pain was 1.3 per 1,000 (95% CI, 0.6-2.3 per 1,000) for fatal overdose and 3.2 per 1,000 (95% CI, 2.0-4.7 per 1,000) for nonfatal overdose.
“Awareness of these predictors may facilitate shared decision-making regarding prescribing opioids for chronic pain and may inform harm-reduction strategies,” Dr. Wang and associates wrote.
This study was supported by a grant from Health Canada’s Substance Use and Addictions Program. Coauthor Dr. Corey Hayes was supported by Veterans Affairs Health Services Research and Development and the National Institute on Drug Abuse Clinical Trials Network. Dr. Jason Busse is supported by the Canadian Institutes of Health Research. Dr. David Juurlink has received travel support for presentations from the CIHR, Stanford University, and Texas Tech University Health Sciences Center.
A Canadian systematic review of 28 observational studies has identified 10 strong predictors of fatal and nonfatal prescription opioid overdose.
Published in CMAJ, the analysis found the risk of fatal and nonfatal opioid overdose was notably tied to such factors as high-dose and fentanyl prescriptions, multiple opioid prescribers or pharmacies, and several mental health issues. High-certainty evidence from 14 studies involving more than a million patients showed a linear dose-response relationship with opioid overdose.
“Our findings suggest that awareness of, and attention to, several patient and prescription characteristics may help reduce the risk of opioid overdose among people living with chronic pain,” wrote a research team led by Li Wang, PhD, a researcher at the Michael G. DeGroote Institute for Pain Research and Care and the department of anesthesia at McMaster University, Hamilton, Ont.
Predictors of fatal and nonfatal overdose
Reporting on studies of 103 possible predictors in a pooled cohort of almost 24 million patients, the review found moderate- to high-certainty evidence for large relative associations with the following 10 criteria:
- A history of overdose (odds ratio, 5.85; 95% confidence interval, 3.78-9.04).
- A higher opioid dosage (OR, 2.57; 95% CI, 2.08-3.18 per 90-mg increment).
- Three or more prescribers (OR, 4.68; 95% CI, 3.57-6.12).
- Four or more dispensing pharmacies (OR, 4.92; 95% CI, 4.35-5.57).
- Prescription for fentanyl (OR, 2.80; 95% CI, 2.30-3.41).
- Current substance use disorder (OR, 2.62; 95% CI, 2.09-3.27).
- Any mental health diagnosis (OR, 2.12; 95% CI, 1.73-2.61).
- Depression (OR, 2.22; 95% CI, 1.57-314).
- Bipolar disorder (OR, 2.07; 95% CI, 1.77-2.41).
- Pancreatitis (OR, 2.00; 95% CI,1.52-2.64).
Absolute risks in patients with the predictor ranged from 2 to 6 per 1,000 for fatal overdose and 4 to 12 per 1,000 for nonfatal overdose.
The authors noted that chronic pain affects 20% of the world’s population worldwide, and a 2021 meta-analysis of 60 observational studies revealed that opioids are prescribed for 27% of adults living with chronic pain, with a higher prevalence of prescribing in North America than in Europe.
International review
A total of 28 observational studies comprising 23,963,716 patients (52% female) with mean age of 52 years were enrolled. All used administrative databases. Twenty-four studies were conducted in the United States, three in Canada, and one in the United Kingdom. Twenty-one studies included only patients with noncancer chronic pain, while seven included patients with either cancer-related or noncancer chronic pain. Twenty-two studies accepted patients with previous or current substance use disorder and three excluded patients with comorbid substance use disorder. Twenty-three studies included patients with comorbid mental illness and five exclusively recruited veterans.
The median sample size was 43,885. As a limitation, 25 studies (89%) were at high risk of bias for at least one criterion, the authors acknowledged. Moderate-certainty evidence showed the pooled prevalence of fatal opioid overdose after prescription for chronic pain was 1.3 per 1,000 (95% CI, 0.6-2.3 per 1,000) for fatal overdose and 3.2 per 1,000 (95% CI, 2.0-4.7 per 1,000) for nonfatal overdose.
“Awareness of these predictors may facilitate shared decision-making regarding prescribing opioids for chronic pain and may inform harm-reduction strategies,” Dr. Wang and associates wrote.
This study was supported by a grant from Health Canada’s Substance Use and Addictions Program. Coauthor Dr. Corey Hayes was supported by Veterans Affairs Health Services Research and Development and the National Institute on Drug Abuse Clinical Trials Network. Dr. Jason Busse is supported by the Canadian Institutes of Health Research. Dr. David Juurlink has received travel support for presentations from the CIHR, Stanford University, and Texas Tech University Health Sciences Center.
A Canadian systematic review of 28 observational studies has identified 10 strong predictors of fatal and nonfatal prescription opioid overdose.
Published in CMAJ, the analysis found the risk of fatal and nonfatal opioid overdose was notably tied to such factors as high-dose and fentanyl prescriptions, multiple opioid prescribers or pharmacies, and several mental health issues. High-certainty evidence from 14 studies involving more than a million patients showed a linear dose-response relationship with opioid overdose.
“Our findings suggest that awareness of, and attention to, several patient and prescription characteristics may help reduce the risk of opioid overdose among people living with chronic pain,” wrote a research team led by Li Wang, PhD, a researcher at the Michael G. DeGroote Institute for Pain Research and Care and the department of anesthesia at McMaster University, Hamilton, Ont.
Predictors of fatal and nonfatal overdose
Reporting on studies of 103 possible predictors in a pooled cohort of almost 24 million patients, the review found moderate- to high-certainty evidence for large relative associations with the following 10 criteria:
- A history of overdose (odds ratio, 5.85; 95% confidence interval, 3.78-9.04).
- A higher opioid dosage (OR, 2.57; 95% CI, 2.08-3.18 per 90-mg increment).
- Three or more prescribers (OR, 4.68; 95% CI, 3.57-6.12).
- Four or more dispensing pharmacies (OR, 4.92; 95% CI, 4.35-5.57).
- Prescription for fentanyl (OR, 2.80; 95% CI, 2.30-3.41).
- Current substance use disorder (OR, 2.62; 95% CI, 2.09-3.27).
- Any mental health diagnosis (OR, 2.12; 95% CI, 1.73-2.61).
- Depression (OR, 2.22; 95% CI, 1.57-314).
- Bipolar disorder (OR, 2.07; 95% CI, 1.77-2.41).
- Pancreatitis (OR, 2.00; 95% CI,1.52-2.64).
Absolute risks in patients with the predictor ranged from 2 to 6 per 1,000 for fatal overdose and 4 to 12 per 1,000 for nonfatal overdose.
The authors noted that chronic pain affects 20% of the world’s population worldwide, and a 2021 meta-analysis of 60 observational studies revealed that opioids are prescribed for 27% of adults living with chronic pain, with a higher prevalence of prescribing in North America than in Europe.
International review
A total of 28 observational studies comprising 23,963,716 patients (52% female) with mean age of 52 years were enrolled. All used administrative databases. Twenty-four studies were conducted in the United States, three in Canada, and one in the United Kingdom. Twenty-one studies included only patients with noncancer chronic pain, while seven included patients with either cancer-related or noncancer chronic pain. Twenty-two studies accepted patients with previous or current substance use disorder and three excluded patients with comorbid substance use disorder. Twenty-three studies included patients with comorbid mental illness and five exclusively recruited veterans.
The median sample size was 43,885. As a limitation, 25 studies (89%) were at high risk of bias for at least one criterion, the authors acknowledged. Moderate-certainty evidence showed the pooled prevalence of fatal opioid overdose after prescription for chronic pain was 1.3 per 1,000 (95% CI, 0.6-2.3 per 1,000) for fatal overdose and 3.2 per 1,000 (95% CI, 2.0-4.7 per 1,000) for nonfatal overdose.
“Awareness of these predictors may facilitate shared decision-making regarding prescribing opioids for chronic pain and may inform harm-reduction strategies,” Dr. Wang and associates wrote.
This study was supported by a grant from Health Canada’s Substance Use and Addictions Program. Coauthor Dr. Corey Hayes was supported by Veterans Affairs Health Services Research and Development and the National Institute on Drug Abuse Clinical Trials Network. Dr. Jason Busse is supported by the Canadian Institutes of Health Research. Dr. David Juurlink has received travel support for presentations from the CIHR, Stanford University, and Texas Tech University Health Sciences Center.
FROM CMAJ