User login
EULAR recommendations define strategies to improve adherence in RMDs
Clinicians who care for patients with rheumatic and musculoskeletal diseases (RMDs) can now refer to a new set of strategies and points to consider from a European League Against Rheumatism (EULAR) task force in building a patient-centered approach to improve adherence to treatments.
Nonadherence to treatments is concerning given that 30%-80% of patients who have RMDs are thought to not follow a recommended treatment plan according to their physicians’ instructions, according to first author Valentin Ritschl of the Medical University of Vienna and colleagues.
“The problem of poor adherence is addressed in some EULAR recommendations/points to consider on the management of specific health conditions or on the role of professionals,” Mr. Ritschl said in an interview. “However, all these recommendations focus on limited aspects of nonadherence and do not cover the multifaceted nature of this phenomenon.”
Mr. Ritschl and colleagues conducted an extensive systematic literature review, the results of which they presented to a task force consisting of a panel of international experts hailing from 12 different countries. The task force included rheumatologists and other health professionals in rheumatology, as well as patient representatives.
The collaboration resulted in investigators crafting a definition of adherence in addition to drafting four overarching principles and nine points to consider, which were published Dec. 18 in Annals of the Rheumatic Diseases.
They defined adherence as “the extent to which a person’s behavior corresponds with the agreed prescription, of pharmacological or nonpharmacological treatments, by a health care provider.”
The four overarching principles emphasize the following concepts: that adherence affects outcomes in people who have RMDs; the importance of shared decision-making, with the understanding that the adherence describes the patient’s behavior “following an agreed prescription”; that numerous factors can affect adherence; and the notion of adherence being a dynamic process that, consequently, requires continuous evaluation.
Among the nine points to consider, Mr. Ritschl and coauthors encouraged all health care providers involved in caring for RMD patients to assume responsibility for promoting adherence. Practitioners should also strive to create an ongoing, open dialogue to discuss adherence, especially in cases in which the patient’s RMD is not well controlled. The patient-centered recommendations include taking into account the patient’s goals and preferences because these greatly contribute to the patient’s ability to adhere to any medication regimen. Another arm of that exploration also requires the medical professional to evaluate any circumstances that could bear a negative effect on the patient’s adherence – whether it be medication access issues related to cost or availability, or functional challenges such as memory, motivation, or complexity of the medication regimen.
Mr. Ritschl believed the task force’s recommendations will add value and help improve overall outcomes in RMD population management.
“Until today, there are no recommendations or points to consider developed in order to support our patients to be adherent to the agreed treatment plan,” he said. “In our project/initiative, we therefore developed for the first time points to consider to detect, assess, and manage nonadherence in people with RMDs.”
Additionally, the recommendations offer some strategic insights to help improve clinical trials because the deleterious effects of nonadherence also affect study results.
Looking ahead, Mr. Ritschl said randomized, controlled trials are necessary to test strategies that might improve adherence. He strongly emphasized the importance of designing future research studies that are heavily patient centered and effective for shared decision-making.
The project was funded by EULAR. Mr. Ritschl reported having no disclosures, but many of his coauthors reported financial relationships with pharmaceutical companies.
SOURCE: Ritschl V et al. Ann Rheum Dis. 2020 Dec 18. doi: 10.1136/annrheumdis-2020-218986.
Clinicians who care for patients with rheumatic and musculoskeletal diseases (RMDs) can now refer to a new set of strategies and points to consider from a European League Against Rheumatism (EULAR) task force in building a patient-centered approach to improve adherence to treatments.
Nonadherence to treatments is concerning given that 30%-80% of patients who have RMDs are thought to not follow a recommended treatment plan according to their physicians’ instructions, according to first author Valentin Ritschl of the Medical University of Vienna and colleagues.
“The problem of poor adherence is addressed in some EULAR recommendations/points to consider on the management of specific health conditions or on the role of professionals,” Mr. Ritschl said in an interview. “However, all these recommendations focus on limited aspects of nonadherence and do not cover the multifaceted nature of this phenomenon.”
Mr. Ritschl and colleagues conducted an extensive systematic literature review, the results of which they presented to a task force consisting of a panel of international experts hailing from 12 different countries. The task force included rheumatologists and other health professionals in rheumatology, as well as patient representatives.
The collaboration resulted in investigators crafting a definition of adherence in addition to drafting four overarching principles and nine points to consider, which were published Dec. 18 in Annals of the Rheumatic Diseases.
They defined adherence as “the extent to which a person’s behavior corresponds with the agreed prescription, of pharmacological or nonpharmacological treatments, by a health care provider.”
The four overarching principles emphasize the following concepts: that adherence affects outcomes in people who have RMDs; the importance of shared decision-making, with the understanding that the adherence describes the patient’s behavior “following an agreed prescription”; that numerous factors can affect adherence; and the notion of adherence being a dynamic process that, consequently, requires continuous evaluation.
Among the nine points to consider, Mr. Ritschl and coauthors encouraged all health care providers involved in caring for RMD patients to assume responsibility for promoting adherence. Practitioners should also strive to create an ongoing, open dialogue to discuss adherence, especially in cases in which the patient’s RMD is not well controlled. The patient-centered recommendations include taking into account the patient’s goals and preferences because these greatly contribute to the patient’s ability to adhere to any medication regimen. Another arm of that exploration also requires the medical professional to evaluate any circumstances that could bear a negative effect on the patient’s adherence – whether it be medication access issues related to cost or availability, or functional challenges such as memory, motivation, or complexity of the medication regimen.
Mr. Ritschl believed the task force’s recommendations will add value and help improve overall outcomes in RMD population management.
“Until today, there are no recommendations or points to consider developed in order to support our patients to be adherent to the agreed treatment plan,” he said. “In our project/initiative, we therefore developed for the first time points to consider to detect, assess, and manage nonadherence in people with RMDs.”
Additionally, the recommendations offer some strategic insights to help improve clinical trials because the deleterious effects of nonadherence also affect study results.
Looking ahead, Mr. Ritschl said randomized, controlled trials are necessary to test strategies that might improve adherence. He strongly emphasized the importance of designing future research studies that are heavily patient centered and effective for shared decision-making.
The project was funded by EULAR. Mr. Ritschl reported having no disclosures, but many of his coauthors reported financial relationships with pharmaceutical companies.
SOURCE: Ritschl V et al. Ann Rheum Dis. 2020 Dec 18. doi: 10.1136/annrheumdis-2020-218986.
Clinicians who care for patients with rheumatic and musculoskeletal diseases (RMDs) can now refer to a new set of strategies and points to consider from a European League Against Rheumatism (EULAR) task force in building a patient-centered approach to improve adherence to treatments.
Nonadherence to treatments is concerning given that 30%-80% of patients who have RMDs are thought to not follow a recommended treatment plan according to their physicians’ instructions, according to first author Valentin Ritschl of the Medical University of Vienna and colleagues.
“The problem of poor adherence is addressed in some EULAR recommendations/points to consider on the management of specific health conditions or on the role of professionals,” Mr. Ritschl said in an interview. “However, all these recommendations focus on limited aspects of nonadherence and do not cover the multifaceted nature of this phenomenon.”
Mr. Ritschl and colleagues conducted an extensive systematic literature review, the results of which they presented to a task force consisting of a panel of international experts hailing from 12 different countries. The task force included rheumatologists and other health professionals in rheumatology, as well as patient representatives.
The collaboration resulted in investigators crafting a definition of adherence in addition to drafting four overarching principles and nine points to consider, which were published Dec. 18 in Annals of the Rheumatic Diseases.
They defined adherence as “the extent to which a person’s behavior corresponds with the agreed prescription, of pharmacological or nonpharmacological treatments, by a health care provider.”
The four overarching principles emphasize the following concepts: that adherence affects outcomes in people who have RMDs; the importance of shared decision-making, with the understanding that the adherence describes the patient’s behavior “following an agreed prescription”; that numerous factors can affect adherence; and the notion of adherence being a dynamic process that, consequently, requires continuous evaluation.
Among the nine points to consider, Mr. Ritschl and coauthors encouraged all health care providers involved in caring for RMD patients to assume responsibility for promoting adherence. Practitioners should also strive to create an ongoing, open dialogue to discuss adherence, especially in cases in which the patient’s RMD is not well controlled. The patient-centered recommendations include taking into account the patient’s goals and preferences because these greatly contribute to the patient’s ability to adhere to any medication regimen. Another arm of that exploration also requires the medical professional to evaluate any circumstances that could bear a negative effect on the patient’s adherence – whether it be medication access issues related to cost or availability, or functional challenges such as memory, motivation, or complexity of the medication regimen.
Mr. Ritschl believed the task force’s recommendations will add value and help improve overall outcomes in RMD population management.
“Until today, there are no recommendations or points to consider developed in order to support our patients to be adherent to the agreed treatment plan,” he said. “In our project/initiative, we therefore developed for the first time points to consider to detect, assess, and manage nonadherence in people with RMDs.”
Additionally, the recommendations offer some strategic insights to help improve clinical trials because the deleterious effects of nonadherence also affect study results.
Looking ahead, Mr. Ritschl said randomized, controlled trials are necessary to test strategies that might improve adherence. He strongly emphasized the importance of designing future research studies that are heavily patient centered and effective for shared decision-making.
The project was funded by EULAR. Mr. Ritschl reported having no disclosures, but many of his coauthors reported financial relationships with pharmaceutical companies.
SOURCE: Ritschl V et al. Ann Rheum Dis. 2020 Dec 18. doi: 10.1136/annrheumdis-2020-218986.
FROM ANNALS OF THE RHEUMATIC DISEASES
‘Hidden’ danger of type 2 diabetes diagnosis at early age
Those who are found to have type 2 diabetes at a younger age face “hidden” dangers. The issue is becoming more and more important, “since new diagnoses in this younger age group continue to rise,” said the authors of a new study, led by Natalie Nanayakkara, MD.
They believe clinical approaches should be based on age at diagnosis. The results of their new meta-analysis, published online in Diabetologia, reveal the extent of the problem.
Believed to be the first systematic review of its kind, the study showed that the younger the age at diagnosis of type 2 diabetes, the greater the risks of dying and of having either microvascular or macrovascular complications each subsequent year (adjusted for current age).
“This difference in risk between younger and older people in terms of absolute versus lifetime risks of type 2 diabetes complications should perhaps be recognized in diabetes management guidelines,” wrote Dr. Nanayakkara, an endocrinologist at Monash University, Melbourne, and colleagues.
Those diagnosed at younger ages are more likely to develop complications that cause greater disability and lead to loss of productivity compared with people diagnosed at an older age, they stressed.
Hence, they suggested “a greater emphasis on preventive measures for younger people with type 2 diabetes,” with “early intensive multifactorial risk factor intervention ... sustained long term to minimize risks over time.”
Large dataset: Use age at diagnosis to risk stratify patients
Rates of type 2 diabetes have increased in all age groups and virtually all countries over the past 3 decades. Particularly worrying is a trend toward increased rates among adults aged 20-44 years. The increases are associated with higher rates of overweight and obesity, poor diet, and decreasing levels of physical activity, numerous studies have shown.
But few studies have examined the association between age at diagnosis and subsequent complications from type 2 diabetes, the authors noted.
Their review included 26 observational studies involving more than one million individuals from 30 countries in the Asia Pacific, Europe, and North America. The investigators found that each 1-year increase in age at diabetes diagnosis was significantly associated with a 4%, 3%, and 5% decreased risk for all-cause mortality, macrovascular disease, and microvascular disease, respectively, adjusted for current age (all P < .001).
Similar decreases in risk per 1-year increase in age at diabetes diagnosis were seen for coronary heart disease (2%), cerebrovascular disease (2%), peripheral vascular disease (3%), retinopathy (8%), nephropathy (6%), and neuropathy (5%); all associations were significant (P < .001).
Dr. Nanayakkara and colleagues noted that current treatment guidelines are limited in that they’re related to the management of patients with suboptimal blood glucose control, and there is no way to predict which people require intensified treatment.
Therefore, they said, “refined stratification using age at diagnosis may provide a method of identifying, at diagnosis, those at greatest risk of complications who would most benefit from targeted, individualized treatment regimens.”
Awareness of this “hidden” danger to younger adults with type 2 diabetes is becoming more and more important, because such cases continue to rise, they reiterated.
They also advised that “public health measures to delay and/or prevent the onset of type 2 diabetes until older age may yield benefits by reducing the duration of diabetes and the burden of complications.”
Dr. Nanayakkara disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Those who are found to have type 2 diabetes at a younger age face “hidden” dangers. The issue is becoming more and more important, “since new diagnoses in this younger age group continue to rise,” said the authors of a new study, led by Natalie Nanayakkara, MD.
They believe clinical approaches should be based on age at diagnosis. The results of their new meta-analysis, published online in Diabetologia, reveal the extent of the problem.
Believed to be the first systematic review of its kind, the study showed that the younger the age at diagnosis of type 2 diabetes, the greater the risks of dying and of having either microvascular or macrovascular complications each subsequent year (adjusted for current age).
“This difference in risk between younger and older people in terms of absolute versus lifetime risks of type 2 diabetes complications should perhaps be recognized in diabetes management guidelines,” wrote Dr. Nanayakkara, an endocrinologist at Monash University, Melbourne, and colleagues.
Those diagnosed at younger ages are more likely to develop complications that cause greater disability and lead to loss of productivity compared with people diagnosed at an older age, they stressed.
Hence, they suggested “a greater emphasis on preventive measures for younger people with type 2 diabetes,” with “early intensive multifactorial risk factor intervention ... sustained long term to minimize risks over time.”
Large dataset: Use age at diagnosis to risk stratify patients
Rates of type 2 diabetes have increased in all age groups and virtually all countries over the past 3 decades. Particularly worrying is a trend toward increased rates among adults aged 20-44 years. The increases are associated with higher rates of overweight and obesity, poor diet, and decreasing levels of physical activity, numerous studies have shown.
But few studies have examined the association between age at diagnosis and subsequent complications from type 2 diabetes, the authors noted.
Their review included 26 observational studies involving more than one million individuals from 30 countries in the Asia Pacific, Europe, and North America. The investigators found that each 1-year increase in age at diabetes diagnosis was significantly associated with a 4%, 3%, and 5% decreased risk for all-cause mortality, macrovascular disease, and microvascular disease, respectively, adjusted for current age (all P < .001).
Similar decreases in risk per 1-year increase in age at diabetes diagnosis were seen for coronary heart disease (2%), cerebrovascular disease (2%), peripheral vascular disease (3%), retinopathy (8%), nephropathy (6%), and neuropathy (5%); all associations were significant (P < .001).
Dr. Nanayakkara and colleagues noted that current treatment guidelines are limited in that they’re related to the management of patients with suboptimal blood glucose control, and there is no way to predict which people require intensified treatment.
Therefore, they said, “refined stratification using age at diagnosis may provide a method of identifying, at diagnosis, those at greatest risk of complications who would most benefit from targeted, individualized treatment regimens.”
Awareness of this “hidden” danger to younger adults with type 2 diabetes is becoming more and more important, because such cases continue to rise, they reiterated.
They also advised that “public health measures to delay and/or prevent the onset of type 2 diabetes until older age may yield benefits by reducing the duration of diabetes and the burden of complications.”
Dr. Nanayakkara disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Those who are found to have type 2 diabetes at a younger age face “hidden” dangers. The issue is becoming more and more important, “since new diagnoses in this younger age group continue to rise,” said the authors of a new study, led by Natalie Nanayakkara, MD.
They believe clinical approaches should be based on age at diagnosis. The results of their new meta-analysis, published online in Diabetologia, reveal the extent of the problem.
Believed to be the first systematic review of its kind, the study showed that the younger the age at diagnosis of type 2 diabetes, the greater the risks of dying and of having either microvascular or macrovascular complications each subsequent year (adjusted for current age).
“This difference in risk between younger and older people in terms of absolute versus lifetime risks of type 2 diabetes complications should perhaps be recognized in diabetes management guidelines,” wrote Dr. Nanayakkara, an endocrinologist at Monash University, Melbourne, and colleagues.
Those diagnosed at younger ages are more likely to develop complications that cause greater disability and lead to loss of productivity compared with people diagnosed at an older age, they stressed.
Hence, they suggested “a greater emphasis on preventive measures for younger people with type 2 diabetes,” with “early intensive multifactorial risk factor intervention ... sustained long term to minimize risks over time.”
Large dataset: Use age at diagnosis to risk stratify patients
Rates of type 2 diabetes have increased in all age groups and virtually all countries over the past 3 decades. Particularly worrying is a trend toward increased rates among adults aged 20-44 years. The increases are associated with higher rates of overweight and obesity, poor diet, and decreasing levels of physical activity, numerous studies have shown.
But few studies have examined the association between age at diagnosis and subsequent complications from type 2 diabetes, the authors noted.
Their review included 26 observational studies involving more than one million individuals from 30 countries in the Asia Pacific, Europe, and North America. The investigators found that each 1-year increase in age at diabetes diagnosis was significantly associated with a 4%, 3%, and 5% decreased risk for all-cause mortality, macrovascular disease, and microvascular disease, respectively, adjusted for current age (all P < .001).
Similar decreases in risk per 1-year increase in age at diabetes diagnosis were seen for coronary heart disease (2%), cerebrovascular disease (2%), peripheral vascular disease (3%), retinopathy (8%), nephropathy (6%), and neuropathy (5%); all associations were significant (P < .001).
Dr. Nanayakkara and colleagues noted that current treatment guidelines are limited in that they’re related to the management of patients with suboptimal blood glucose control, and there is no way to predict which people require intensified treatment.
Therefore, they said, “refined stratification using age at diagnosis may provide a method of identifying, at diagnosis, those at greatest risk of complications who would most benefit from targeted, individualized treatment regimens.”
Awareness of this “hidden” danger to younger adults with type 2 diabetes is becoming more and more important, because such cases continue to rise, they reiterated.
They also advised that “public health measures to delay and/or prevent the onset of type 2 diabetes until older age may yield benefits by reducing the duration of diabetes and the burden of complications.”
Dr. Nanayakkara disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
New pediatric cases down as U.S. tops 2 million children with COVID-19
The United States exceeded 2 million reported cases of COVID-19 in children just 6 weeks after recording its 1 millionth case, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.
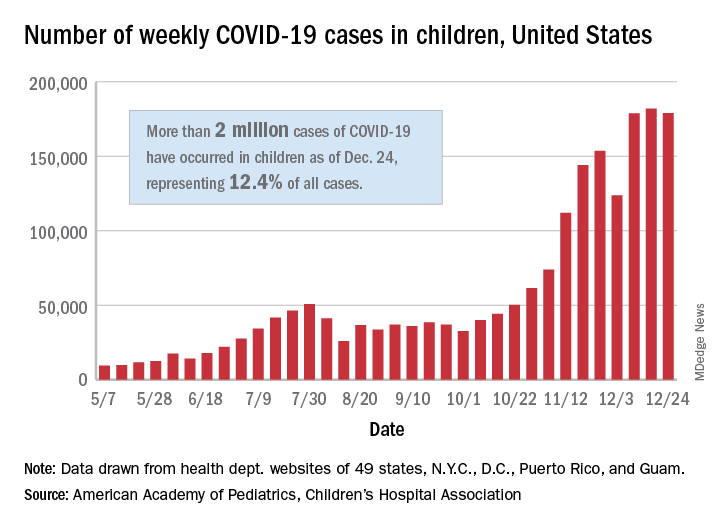
The total number of cases in children was 2,000,681 as of Dec. 24, which represents 12.4% of all cases reported by the health departments of 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam, the AAP and CHA stated Dec. 29.
The case count for just the latest week, 178,935, was actually down 1.7% from the 182,018 reported the week before, marking the second drop since the beginning of December. The first came during the week ending Dec. 3, when the number of cases dropped more than 19% from the previous week, based on data from the AAP/CHA report.
The cumulative national rate of coronavirus infection is now 2,658 cases per 100,000 children, and “13 states have reported more than 4,000 cases per 100,000,” the two groups said.
The highest rate for any state can be found in North Dakota, which has had 7,722 cases of COVID-19 per 100,000 children. Wyoming has the highest proportion of cases in children at 20.5%, and California has reported the most cases overall, 234,174, the report shows.
Data on testing, hospitalization, and mortality were not included in the Dec. 29 report because of the holiday but will be available in the next edition, scheduled for release on Jan. 5, 2021.
The United States exceeded 2 million reported cases of COVID-19 in children just 6 weeks after recording its 1 millionth case, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.

The total number of cases in children was 2,000,681 as of Dec. 24, which represents 12.4% of all cases reported by the health departments of 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam, the AAP and CHA stated Dec. 29.
The case count for just the latest week, 178,935, was actually down 1.7% from the 182,018 reported the week before, marking the second drop since the beginning of December. The first came during the week ending Dec. 3, when the number of cases dropped more than 19% from the previous week, based on data from the AAP/CHA report.
The cumulative national rate of coronavirus infection is now 2,658 cases per 100,000 children, and “13 states have reported more than 4,000 cases per 100,000,” the two groups said.
The highest rate for any state can be found in North Dakota, which has had 7,722 cases of COVID-19 per 100,000 children. Wyoming has the highest proportion of cases in children at 20.5%, and California has reported the most cases overall, 234,174, the report shows.
Data on testing, hospitalization, and mortality were not included in the Dec. 29 report because of the holiday but will be available in the next edition, scheduled for release on Jan. 5, 2021.
The United States exceeded 2 million reported cases of COVID-19 in children just 6 weeks after recording its 1 millionth case, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.

The total number of cases in children was 2,000,681 as of Dec. 24, which represents 12.4% of all cases reported by the health departments of 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam, the AAP and CHA stated Dec. 29.
The case count for just the latest week, 178,935, was actually down 1.7% from the 182,018 reported the week before, marking the second drop since the beginning of December. The first came during the week ending Dec. 3, when the number of cases dropped more than 19% from the previous week, based on data from the AAP/CHA report.
The cumulative national rate of coronavirus infection is now 2,658 cases per 100,000 children, and “13 states have reported more than 4,000 cases per 100,000,” the two groups said.
The highest rate for any state can be found in North Dakota, which has had 7,722 cases of COVID-19 per 100,000 children. Wyoming has the highest proportion of cases in children at 20.5%, and California has reported the most cases overall, 234,174, the report shows.
Data on testing, hospitalization, and mortality were not included in the Dec. 29 report because of the holiday but will be available in the next edition, scheduled for release on Jan. 5, 2021.
New dietary guidelines omit recommended cuts to sugar, alcohol intake
Although the new guidelines were informed by an advisory committee’s scientific report, officials omitted certain recommendations that would have reduced allowances for added sugars and alcohol intake.
The 2020-2025 Dietary Guidelines for Americans “carried forward the committee’s emphasis on limiting these dietary components, but did not include changes to quantitative recommendations, as there was not a preponderance of evidence in the material the committee reviewed to support specific changes, as required by law,” the agencies said in a news release.
The guidelines encourage Americans to “Make Every Bite Count” through four overarching suggestions:
- Follow a healthy dietary pattern at every life stage.
- Customize nutrient-dense food and beverage choices to reflect preferences, cultural traditions, and budgets.
- Focus on meeting dietary needs from five food groups – vegetables, fruits, grains, dairy and fortified soy alternatives, and proteins – and stay within calorie limits.
- Limit foods and beverages that are higher in added sugars, saturated fat, and sodium, and limit alcoholic beverages.
The guidance “can help all Americans lead healthier lives by making every bite count,” Secretary of Agriculture Sonny Perdue said.
Proposed cutoffs rejected
The guidelines omit a recommendation from the advisory committee’s scientific report to reduce intake of added sugars from less than 10% of calories to less than 6% of calories.
It also omits a recommendation that men and women who drink alcohol limit themselves to one drink per day. It maintains guidance from the 2015-2020 edition that allows two drinks per day for men.
The agencies published a document explaining why they omitted the advisory committee›s conclusions.
The American Heart Association in July had praised the suggestion to reduce added sugars. The proposed change would have helped “steer the public toward a more heart-healthy path in their daily diets,” Mitchell S.V. Elkind, MD, president of the AHA, said at the time. The association would “strongly oppose any efforts to weaken these recommendations,” he added.
In its response to the new guidelines, Dr. Elkind praised the emphasis on a healthy diet “at every life stage” but called out a missed opportunity.
“We are disappointed that USDA and HHS did not accept all of the Dietary Guidelines Advisory Committee’s science-based recommendations in the final guidelines for 2020, including the recommendation to lower added sugars consumption to less than 6% of calories,” he said in a prepared statement.
Guidance for infants and toddlers
The guidelines advise that for about the first 6 months of life, infants should exclusively receive breast milk. Infants should continue to receive breast milk through at least the first year of life, and longer if desired. Infants should be fed iron-fortified infant formula during the first year of life when breast milk is unavailable, and infants should receive supplemental vitamin D soon after birth, the guidelines advise.
At about 6 months, infants should be introduced to a variety of nutrient-dense complementary foods, including potentially allergenic foods. Infants should eat foods that are rich in iron and zinc, particularly if they are fed breast milk.
The guidelines also include dietary and caloric advice for pregnant and lactating women with daily or weekly amounts of food from different groups and subgroups.
Dr. Elkind highlighted the significance of these additions.
“We are pleased that for the first time, the guidelines provide recommendations for pregnant and breastfeeding women as well as infants and toddlers, underscoring the importance of maternal health and proper nutrition across the lifespan,” he said.
For all ages
From 12 months through older adulthood, people should follow a healthy dietary pattern to meet nutrient needs, help achieve a healthy body weight, and reduce the risk of chronic disease.
According to the guidelines, core elements of a healthy diet include:
- Vegetables of all types (dark green; red and orange; beans, peas, and lentils; starchy; and other types).
- Fruits (especially whole fruit).
- Grains, at least half of which are whole grain.
- Dairy, including fat-free or low-fat milk, yogurt, and cheese, and lactose-free versions; and fortified soy beverages and yogurt as alternatives.
- Protein foods, including lean meats, poultry, and eggs; seafood; beans, peas, and lentils; and nuts, seeds, and soy products.
- Oils, including vegetable oils and oils in food, such as seafood and nuts.
The guidelines spell out limits to added sugars, sodium, saturated fat, and alcohol. The recommendation to limit added sugars to less than 10% of calories per day starts at age 2 years. Before age 2, foods and beverages with added sugars should be avoided.
Saturated fat should be limited to less than 10% of calories per day starting at age 2. And sodium intake should be limited to 2,300 mg/day for those age 14 and older, but just 1,200 mg/day for toddlers, 1,500 mg/day for children aged 4-8, and 1,800 mg/day for children 9-13.
“Adults of legal drinking age can choose not to drink or to drink in moderation by limiting intake to 2 drinks or less in a day for men and 1 drink or less in a day for women, when alcohol is consumed,” the agencies said. “Drinking less is better for health than drinking more. There are some adults who should not drink alcohol, such as women who are pregnant.”
An appendix includes estimated calorie needs based on a person’s age, sex, height, weight, and level of physical activity. A need to lose, maintain, or gain weight are among the factors that influence how many calories should be consumed, the guidelines note.
The guidelines are designed for use by health care professionals and policymakers. The USDA has launched a new MyPlate website to help consumers incorporate the dietary guidance.
A version of this article first appeared on Medscape.com.
Although the new guidelines were informed by an advisory committee’s scientific report, officials omitted certain recommendations that would have reduced allowances for added sugars and alcohol intake.
The 2020-2025 Dietary Guidelines for Americans “carried forward the committee’s emphasis on limiting these dietary components, but did not include changes to quantitative recommendations, as there was not a preponderance of evidence in the material the committee reviewed to support specific changes, as required by law,” the agencies said in a news release.
The guidelines encourage Americans to “Make Every Bite Count” through four overarching suggestions:
- Follow a healthy dietary pattern at every life stage.
- Customize nutrient-dense food and beverage choices to reflect preferences, cultural traditions, and budgets.
- Focus on meeting dietary needs from five food groups – vegetables, fruits, grains, dairy and fortified soy alternatives, and proteins – and stay within calorie limits.
- Limit foods and beverages that are higher in added sugars, saturated fat, and sodium, and limit alcoholic beverages.
The guidance “can help all Americans lead healthier lives by making every bite count,” Secretary of Agriculture Sonny Perdue said.
Proposed cutoffs rejected
The guidelines omit a recommendation from the advisory committee’s scientific report to reduce intake of added sugars from less than 10% of calories to less than 6% of calories.
It also omits a recommendation that men and women who drink alcohol limit themselves to one drink per day. It maintains guidance from the 2015-2020 edition that allows two drinks per day for men.
The agencies published a document explaining why they omitted the advisory committee›s conclusions.
The American Heart Association in July had praised the suggestion to reduce added sugars. The proposed change would have helped “steer the public toward a more heart-healthy path in their daily diets,” Mitchell S.V. Elkind, MD, president of the AHA, said at the time. The association would “strongly oppose any efforts to weaken these recommendations,” he added.
In its response to the new guidelines, Dr. Elkind praised the emphasis on a healthy diet “at every life stage” but called out a missed opportunity.
“We are disappointed that USDA and HHS did not accept all of the Dietary Guidelines Advisory Committee’s science-based recommendations in the final guidelines for 2020, including the recommendation to lower added sugars consumption to less than 6% of calories,” he said in a prepared statement.
Guidance for infants and toddlers
The guidelines advise that for about the first 6 months of life, infants should exclusively receive breast milk. Infants should continue to receive breast milk through at least the first year of life, and longer if desired. Infants should be fed iron-fortified infant formula during the first year of life when breast milk is unavailable, and infants should receive supplemental vitamin D soon after birth, the guidelines advise.
At about 6 months, infants should be introduced to a variety of nutrient-dense complementary foods, including potentially allergenic foods. Infants should eat foods that are rich in iron and zinc, particularly if they are fed breast milk.
The guidelines also include dietary and caloric advice for pregnant and lactating women with daily or weekly amounts of food from different groups and subgroups.
Dr. Elkind highlighted the significance of these additions.
“We are pleased that for the first time, the guidelines provide recommendations for pregnant and breastfeeding women as well as infants and toddlers, underscoring the importance of maternal health and proper nutrition across the lifespan,” he said.
For all ages
From 12 months through older adulthood, people should follow a healthy dietary pattern to meet nutrient needs, help achieve a healthy body weight, and reduce the risk of chronic disease.
According to the guidelines, core elements of a healthy diet include:
- Vegetables of all types (dark green; red and orange; beans, peas, and lentils; starchy; and other types).
- Fruits (especially whole fruit).
- Grains, at least half of which are whole grain.
- Dairy, including fat-free or low-fat milk, yogurt, and cheese, and lactose-free versions; and fortified soy beverages and yogurt as alternatives.
- Protein foods, including lean meats, poultry, and eggs; seafood; beans, peas, and lentils; and nuts, seeds, and soy products.
- Oils, including vegetable oils and oils in food, such as seafood and nuts.
The guidelines spell out limits to added sugars, sodium, saturated fat, and alcohol. The recommendation to limit added sugars to less than 10% of calories per day starts at age 2 years. Before age 2, foods and beverages with added sugars should be avoided.
Saturated fat should be limited to less than 10% of calories per day starting at age 2. And sodium intake should be limited to 2,300 mg/day for those age 14 and older, but just 1,200 mg/day for toddlers, 1,500 mg/day for children aged 4-8, and 1,800 mg/day for children 9-13.
“Adults of legal drinking age can choose not to drink or to drink in moderation by limiting intake to 2 drinks or less in a day for men and 1 drink or less in a day for women, when alcohol is consumed,” the agencies said. “Drinking less is better for health than drinking more. There are some adults who should not drink alcohol, such as women who are pregnant.”
An appendix includes estimated calorie needs based on a person’s age, sex, height, weight, and level of physical activity. A need to lose, maintain, or gain weight are among the factors that influence how many calories should be consumed, the guidelines note.
The guidelines are designed for use by health care professionals and policymakers. The USDA has launched a new MyPlate website to help consumers incorporate the dietary guidance.
A version of this article first appeared on Medscape.com.
Although the new guidelines were informed by an advisory committee’s scientific report, officials omitted certain recommendations that would have reduced allowances for added sugars and alcohol intake.
The 2020-2025 Dietary Guidelines for Americans “carried forward the committee’s emphasis on limiting these dietary components, but did not include changes to quantitative recommendations, as there was not a preponderance of evidence in the material the committee reviewed to support specific changes, as required by law,” the agencies said in a news release.
The guidelines encourage Americans to “Make Every Bite Count” through four overarching suggestions:
- Follow a healthy dietary pattern at every life stage.
- Customize nutrient-dense food and beverage choices to reflect preferences, cultural traditions, and budgets.
- Focus on meeting dietary needs from five food groups – vegetables, fruits, grains, dairy and fortified soy alternatives, and proteins – and stay within calorie limits.
- Limit foods and beverages that are higher in added sugars, saturated fat, and sodium, and limit alcoholic beverages.
The guidance “can help all Americans lead healthier lives by making every bite count,” Secretary of Agriculture Sonny Perdue said.
Proposed cutoffs rejected
The guidelines omit a recommendation from the advisory committee’s scientific report to reduce intake of added sugars from less than 10% of calories to less than 6% of calories.
It also omits a recommendation that men and women who drink alcohol limit themselves to one drink per day. It maintains guidance from the 2015-2020 edition that allows two drinks per day for men.
The agencies published a document explaining why they omitted the advisory committee›s conclusions.
The American Heart Association in July had praised the suggestion to reduce added sugars. The proposed change would have helped “steer the public toward a more heart-healthy path in their daily diets,” Mitchell S.V. Elkind, MD, president of the AHA, said at the time. The association would “strongly oppose any efforts to weaken these recommendations,” he added.
In its response to the new guidelines, Dr. Elkind praised the emphasis on a healthy diet “at every life stage” but called out a missed opportunity.
“We are disappointed that USDA and HHS did not accept all of the Dietary Guidelines Advisory Committee’s science-based recommendations in the final guidelines for 2020, including the recommendation to lower added sugars consumption to less than 6% of calories,” he said in a prepared statement.
Guidance for infants and toddlers
The guidelines advise that for about the first 6 months of life, infants should exclusively receive breast milk. Infants should continue to receive breast milk through at least the first year of life, and longer if desired. Infants should be fed iron-fortified infant formula during the first year of life when breast milk is unavailable, and infants should receive supplemental vitamin D soon after birth, the guidelines advise.
At about 6 months, infants should be introduced to a variety of nutrient-dense complementary foods, including potentially allergenic foods. Infants should eat foods that are rich in iron and zinc, particularly if they are fed breast milk.
The guidelines also include dietary and caloric advice for pregnant and lactating women with daily or weekly amounts of food from different groups and subgroups.
Dr. Elkind highlighted the significance of these additions.
“We are pleased that for the first time, the guidelines provide recommendations for pregnant and breastfeeding women as well as infants and toddlers, underscoring the importance of maternal health and proper nutrition across the lifespan,” he said.
For all ages
From 12 months through older adulthood, people should follow a healthy dietary pattern to meet nutrient needs, help achieve a healthy body weight, and reduce the risk of chronic disease.
According to the guidelines, core elements of a healthy diet include:
- Vegetables of all types (dark green; red and orange; beans, peas, and lentils; starchy; and other types).
- Fruits (especially whole fruit).
- Grains, at least half of which are whole grain.
- Dairy, including fat-free or low-fat milk, yogurt, and cheese, and lactose-free versions; and fortified soy beverages and yogurt as alternatives.
- Protein foods, including lean meats, poultry, and eggs; seafood; beans, peas, and lentils; and nuts, seeds, and soy products.
- Oils, including vegetable oils and oils in food, such as seafood and nuts.
The guidelines spell out limits to added sugars, sodium, saturated fat, and alcohol. The recommendation to limit added sugars to less than 10% of calories per day starts at age 2 years. Before age 2, foods and beverages with added sugars should be avoided.
Saturated fat should be limited to less than 10% of calories per day starting at age 2. And sodium intake should be limited to 2,300 mg/day for those age 14 and older, but just 1,200 mg/day for toddlers, 1,500 mg/day for children aged 4-8, and 1,800 mg/day for children 9-13.
“Adults of legal drinking age can choose not to drink or to drink in moderation by limiting intake to 2 drinks or less in a day for men and 1 drink or less in a day for women, when alcohol is consumed,” the agencies said. “Drinking less is better for health than drinking more. There are some adults who should not drink alcohol, such as women who are pregnant.”
An appendix includes estimated calorie needs based on a person’s age, sex, height, weight, and level of physical activity. A need to lose, maintain, or gain weight are among the factors that influence how many calories should be consumed, the guidelines note.
The guidelines are designed for use by health care professionals and policymakers. The USDA has launched a new MyPlate website to help consumers incorporate the dietary guidance.
A version of this article first appeared on Medscape.com.
Intense intervention may boost addiction program retention
An intense and assertive “won’t take no for an answer” approach is effective for engaging in treatment young adults with substance abuse who have been in and out of various recovery programs for years, new research suggests.
The Youth Opioid Recovery Support (YORS) program is a team effort that includes home delivery of the prescribed medication, family engagement, assertive outreach, and contingency management.
In a new study of 42 patients in recovery for substance use disorder (SUD), those who were treated with extended-release naltrexone or extended-release buprenorphine plus YORS received more outpatient doses of their medication, and rates of opioid relapse at 12 and 24 weeks were lower compared with their peers who received only treatment as usual.
“ coinvestigator Marc Fishman, MD, director of the Maryland Treatment Centers, Johns Hopkins University, Baltimore, said in an interview.
The findings were presented at the virtual American Academy of Addiction Psychiatry 31st Annual Meeting.
Treatment barriers
Young adults with SUD are difficult to reach, which leads to decreased addiction program retention, decreased medication adherence, early drop out, waxing and waning motivation, and worse outcomes, compared with older adults with SUD, Dr. Fishman said.
In July, positive results from a pilot trial conducted by the investigators of YORS were published online in Addiction.
In that study, 41 young adults aged 18-26 years who intended to undergo treatment for SUD with extended-release naltrexone were randomly assigned to also undergo YORS or treatment as usual, which consisted of a standard referral to outpatient care following an inpatient stay.
The primary outcomes were number of medication doses received over 24 weeks and relapse to opioid use, which was defined as 10 or more days of use within 28 days at 24 weeks.
Participants in the YORS group received more doses of extended-release naltrexone (mean, 4.28; standard deviation, 2.3) than participants in the treatment-as-usual group (mean, 0.70; SD, 1.2; P < .01).
In the YORS group, rates of relapse at both 12 and 24 weeks were lower, and there were fewer overall days of opioid use.
For the current study, the investigators wanted to test whether there was a possible effect when patients were given a choice of medication. In the earlier trial, patients did not have a choice – they had to take extended-release naltrexone. In this study, they could opt for it or extended-release buprenorphine.
The researchers recruited 22 young adults (aged 18-26 years) from their inpatient clinic to participate. Half the patients chose to take extended-release naltrexone, and the other half chose extended-release buprenorphine.
The groups were then compared to a historical group of 20 patients who received treatment as usual and served as the control group.
Positive outcomes
As in the first study, outcomes in the new study were better with YORS.
All participants who underwent YORS received more outpatient medication doses at 12 weeks and 24 weeks than those who received treatment as usual (1.91 vs. 0.40 and 3.76 vs. 0.70, respectively; P < .001).
For the YORS group, rates of opioid relapse were lower at 12 weeks (27.3% vs. 75.0%) and at 24 weeks (52.9% vs. 95.0%; P < .01.)
All components of YORS work together to improve retention, Dr. Fishman noted. Patients do much better if a relative such as a mother, father, or grandmother is closely involved, he added.
Also important is drug delivery.
“In some ways, this is similar to the assertive community treatment, or ACT, for schizophrenia. Like substance use disorder, schizophrenia requires long-acting injectable antipsychotics. When that is delivered to the patient through an organized delivery service like YORS, it improves outcomes,” said Dr. Fishman.
SUD is a chronic, relapsing illness in which an individual’s judgment is impaired, he added.
“ACT has become a relatively standard feature of treatment in most communities in this country and internationally and is sustainable under public sector funding, so it’s not an impossible leap to say it could be done. But it will not be cheap,” Dr. Fishman said.
Removing barriers
In a comment, Serra Akyar, MD, a psychiatry resident at Northwell Health’s Staten Island University Hospital, New York, said that the YORS program may appear to be labor intensive.
“However, the combination of medication-assisted treatment and support are essential to the treatment of opioid use disorder, especially for young adults. Developing effective interventions for young adults is particularly important, given the plasticity of their brains,” said Dr. Akyar, who was not involved with the research.
Inability to access medication and a lack of a supportive environment, both in everyday life and in regards to therapy, are barriers to successful treatment, she noted.
“The YORS intervention aims to remove these barriers to further enhance engagement to care through a combination of medication delivery and family engagement and assertive outreach via text messaging, a modality presumed to be well received by youth,” Dr. Akyar said.
Despite having a limited sample size, the study shows how a comprehensive intervention can have a large impact on the maintenance of medication adherence and reduction of relapse in young adults, she added.
“Its early success is encouraging and warrants further study on a larger scale to determine long-term effectiveness, overall costs and feasibility, generalizability, and whether certain independent factors exist that may predict medication adherence and reduction of relapse,” she said.
Wraparound support
The study is also a significant reminder that the opioid crisis has affected the young adult population, who are very vulnerable to OUD, said Jose Vito, MD, child, adolescent, and addiction psychiatrist at New York University.
“The study made me realize the importance of the four components of YORS, which were the outreach, family involvement, home delivery, and monetary incentives,” Dr. Vito said in an interview.
All of these components, in addition to extended-release naltrexone or extended-release buprenorphine, “have contributed to lower rates of opioid relapse, and the relapses are much later in the course of treatment if they do occur,” he said.
Overall, the findings demonstrate the importance of not giving up on these youths, he noted.
“Programs like YORS that provide wraparound support can help alleviate the opioid health care crisis by keeping these young adults in treatment,” Dr. Vito concluded.
The study was funded by the University of Maryland Center for Addiction Research, Education, and Service. Dr. Fishman has a financial relationship with Alkermes.
A version of this article first appeared on Medscape.com.
An intense and assertive “won’t take no for an answer” approach is effective for engaging in treatment young adults with substance abuse who have been in and out of various recovery programs for years, new research suggests.
The Youth Opioid Recovery Support (YORS) program is a team effort that includes home delivery of the prescribed medication, family engagement, assertive outreach, and contingency management.
In a new study of 42 patients in recovery for substance use disorder (SUD), those who were treated with extended-release naltrexone or extended-release buprenorphine plus YORS received more outpatient doses of their medication, and rates of opioid relapse at 12 and 24 weeks were lower compared with their peers who received only treatment as usual.
“ coinvestigator Marc Fishman, MD, director of the Maryland Treatment Centers, Johns Hopkins University, Baltimore, said in an interview.
The findings were presented at the virtual American Academy of Addiction Psychiatry 31st Annual Meeting.
Treatment barriers
Young adults with SUD are difficult to reach, which leads to decreased addiction program retention, decreased medication adherence, early drop out, waxing and waning motivation, and worse outcomes, compared with older adults with SUD, Dr. Fishman said.
In July, positive results from a pilot trial conducted by the investigators of YORS were published online in Addiction.
In that study, 41 young adults aged 18-26 years who intended to undergo treatment for SUD with extended-release naltrexone were randomly assigned to also undergo YORS or treatment as usual, which consisted of a standard referral to outpatient care following an inpatient stay.
The primary outcomes were number of medication doses received over 24 weeks and relapse to opioid use, which was defined as 10 or more days of use within 28 days at 24 weeks.
Participants in the YORS group received more doses of extended-release naltrexone (mean, 4.28; standard deviation, 2.3) than participants in the treatment-as-usual group (mean, 0.70; SD, 1.2; P < .01).
In the YORS group, rates of relapse at both 12 and 24 weeks were lower, and there were fewer overall days of opioid use.
For the current study, the investigators wanted to test whether there was a possible effect when patients were given a choice of medication. In the earlier trial, patients did not have a choice – they had to take extended-release naltrexone. In this study, they could opt for it or extended-release buprenorphine.
The researchers recruited 22 young adults (aged 18-26 years) from their inpatient clinic to participate. Half the patients chose to take extended-release naltrexone, and the other half chose extended-release buprenorphine.
The groups were then compared to a historical group of 20 patients who received treatment as usual and served as the control group.
Positive outcomes
As in the first study, outcomes in the new study were better with YORS.
All participants who underwent YORS received more outpatient medication doses at 12 weeks and 24 weeks than those who received treatment as usual (1.91 vs. 0.40 and 3.76 vs. 0.70, respectively; P < .001).
For the YORS group, rates of opioid relapse were lower at 12 weeks (27.3% vs. 75.0%) and at 24 weeks (52.9% vs. 95.0%; P < .01.)
All components of YORS work together to improve retention, Dr. Fishman noted. Patients do much better if a relative such as a mother, father, or grandmother is closely involved, he added.
Also important is drug delivery.
“In some ways, this is similar to the assertive community treatment, or ACT, for schizophrenia. Like substance use disorder, schizophrenia requires long-acting injectable antipsychotics. When that is delivered to the patient through an organized delivery service like YORS, it improves outcomes,” said Dr. Fishman.
SUD is a chronic, relapsing illness in which an individual’s judgment is impaired, he added.
“ACT has become a relatively standard feature of treatment in most communities in this country and internationally and is sustainable under public sector funding, so it’s not an impossible leap to say it could be done. But it will not be cheap,” Dr. Fishman said.
Removing barriers
In a comment, Serra Akyar, MD, a psychiatry resident at Northwell Health’s Staten Island University Hospital, New York, said that the YORS program may appear to be labor intensive.
“However, the combination of medication-assisted treatment and support are essential to the treatment of opioid use disorder, especially for young adults. Developing effective interventions for young adults is particularly important, given the plasticity of their brains,” said Dr. Akyar, who was not involved with the research.
Inability to access medication and a lack of a supportive environment, both in everyday life and in regards to therapy, are barriers to successful treatment, she noted.
“The YORS intervention aims to remove these barriers to further enhance engagement to care through a combination of medication delivery and family engagement and assertive outreach via text messaging, a modality presumed to be well received by youth,” Dr. Akyar said.
Despite having a limited sample size, the study shows how a comprehensive intervention can have a large impact on the maintenance of medication adherence and reduction of relapse in young adults, she added.
“Its early success is encouraging and warrants further study on a larger scale to determine long-term effectiveness, overall costs and feasibility, generalizability, and whether certain independent factors exist that may predict medication adherence and reduction of relapse,” she said.
Wraparound support
The study is also a significant reminder that the opioid crisis has affected the young adult population, who are very vulnerable to OUD, said Jose Vito, MD, child, adolescent, and addiction psychiatrist at New York University.
“The study made me realize the importance of the four components of YORS, which were the outreach, family involvement, home delivery, and monetary incentives,” Dr. Vito said in an interview.
All of these components, in addition to extended-release naltrexone or extended-release buprenorphine, “have contributed to lower rates of opioid relapse, and the relapses are much later in the course of treatment if they do occur,” he said.
Overall, the findings demonstrate the importance of not giving up on these youths, he noted.
“Programs like YORS that provide wraparound support can help alleviate the opioid health care crisis by keeping these young adults in treatment,” Dr. Vito concluded.
The study was funded by the University of Maryland Center for Addiction Research, Education, and Service. Dr. Fishman has a financial relationship with Alkermes.
A version of this article first appeared on Medscape.com.
An intense and assertive “won’t take no for an answer” approach is effective for engaging in treatment young adults with substance abuse who have been in and out of various recovery programs for years, new research suggests.
The Youth Opioid Recovery Support (YORS) program is a team effort that includes home delivery of the prescribed medication, family engagement, assertive outreach, and contingency management.
In a new study of 42 patients in recovery for substance use disorder (SUD), those who were treated with extended-release naltrexone or extended-release buprenorphine plus YORS received more outpatient doses of their medication, and rates of opioid relapse at 12 and 24 weeks were lower compared with their peers who received only treatment as usual.
“ coinvestigator Marc Fishman, MD, director of the Maryland Treatment Centers, Johns Hopkins University, Baltimore, said in an interview.
The findings were presented at the virtual American Academy of Addiction Psychiatry 31st Annual Meeting.
Treatment barriers
Young adults with SUD are difficult to reach, which leads to decreased addiction program retention, decreased medication adherence, early drop out, waxing and waning motivation, and worse outcomes, compared with older adults with SUD, Dr. Fishman said.
In July, positive results from a pilot trial conducted by the investigators of YORS were published online in Addiction.
In that study, 41 young adults aged 18-26 years who intended to undergo treatment for SUD with extended-release naltrexone were randomly assigned to also undergo YORS or treatment as usual, which consisted of a standard referral to outpatient care following an inpatient stay.
The primary outcomes were number of medication doses received over 24 weeks and relapse to opioid use, which was defined as 10 or more days of use within 28 days at 24 weeks.
Participants in the YORS group received more doses of extended-release naltrexone (mean, 4.28; standard deviation, 2.3) than participants in the treatment-as-usual group (mean, 0.70; SD, 1.2; P < .01).
In the YORS group, rates of relapse at both 12 and 24 weeks were lower, and there were fewer overall days of opioid use.
For the current study, the investigators wanted to test whether there was a possible effect when patients were given a choice of medication. In the earlier trial, patients did not have a choice – they had to take extended-release naltrexone. In this study, they could opt for it or extended-release buprenorphine.
The researchers recruited 22 young adults (aged 18-26 years) from their inpatient clinic to participate. Half the patients chose to take extended-release naltrexone, and the other half chose extended-release buprenorphine.
The groups were then compared to a historical group of 20 patients who received treatment as usual and served as the control group.
Positive outcomes
As in the first study, outcomes in the new study were better with YORS.
All participants who underwent YORS received more outpatient medication doses at 12 weeks and 24 weeks than those who received treatment as usual (1.91 vs. 0.40 and 3.76 vs. 0.70, respectively; P < .001).
For the YORS group, rates of opioid relapse were lower at 12 weeks (27.3% vs. 75.0%) and at 24 weeks (52.9% vs. 95.0%; P < .01.)
All components of YORS work together to improve retention, Dr. Fishman noted. Patients do much better if a relative such as a mother, father, or grandmother is closely involved, he added.
Also important is drug delivery.
“In some ways, this is similar to the assertive community treatment, or ACT, for schizophrenia. Like substance use disorder, schizophrenia requires long-acting injectable antipsychotics. When that is delivered to the patient through an organized delivery service like YORS, it improves outcomes,” said Dr. Fishman.
SUD is a chronic, relapsing illness in which an individual’s judgment is impaired, he added.
“ACT has become a relatively standard feature of treatment in most communities in this country and internationally and is sustainable under public sector funding, so it’s not an impossible leap to say it could be done. But it will not be cheap,” Dr. Fishman said.
Removing barriers
In a comment, Serra Akyar, MD, a psychiatry resident at Northwell Health’s Staten Island University Hospital, New York, said that the YORS program may appear to be labor intensive.
“However, the combination of medication-assisted treatment and support are essential to the treatment of opioid use disorder, especially for young adults. Developing effective interventions for young adults is particularly important, given the plasticity of their brains,” said Dr. Akyar, who was not involved with the research.
Inability to access medication and a lack of a supportive environment, both in everyday life and in regards to therapy, are barriers to successful treatment, she noted.
“The YORS intervention aims to remove these barriers to further enhance engagement to care through a combination of medication delivery and family engagement and assertive outreach via text messaging, a modality presumed to be well received by youth,” Dr. Akyar said.
Despite having a limited sample size, the study shows how a comprehensive intervention can have a large impact on the maintenance of medication adherence and reduction of relapse in young adults, she added.
“Its early success is encouraging and warrants further study on a larger scale to determine long-term effectiveness, overall costs and feasibility, generalizability, and whether certain independent factors exist that may predict medication adherence and reduction of relapse,” she said.
Wraparound support
The study is also a significant reminder that the opioid crisis has affected the young adult population, who are very vulnerable to OUD, said Jose Vito, MD, child, adolescent, and addiction psychiatrist at New York University.
“The study made me realize the importance of the four components of YORS, which were the outreach, family involvement, home delivery, and monetary incentives,” Dr. Vito said in an interview.
All of these components, in addition to extended-release naltrexone or extended-release buprenorphine, “have contributed to lower rates of opioid relapse, and the relapses are much later in the course of treatment if they do occur,” he said.
Overall, the findings demonstrate the importance of not giving up on these youths, he noted.
“Programs like YORS that provide wraparound support can help alleviate the opioid health care crisis by keeping these young adults in treatment,” Dr. Vito concluded.
The study was funded by the University of Maryland Center for Addiction Research, Education, and Service. Dr. Fishman has a financial relationship with Alkermes.
A version of this article first appeared on Medscape.com.
Does daily inhaler monitoring improve asthma outcomes in children?
Among children with moderate or severe persistent asthma, a randomized trial suggests.
But the intervention also may lead to more ED visits and increased hospitalization rates.
“We improved asthma symptom control but did not reduce health care use,” Ruchi S. Gupta, MD, MPH, and colleagues, wrote in a study published in Pediatrics.
The monitoring system alerted clinicians when a patient used a short-acting beta-agonist more than four times in a day. It could be that the “alerts enabled providers to detect asthma exacerbation virtually and refer for clinically appropriate care that included directing children to the ED,” the authors suggested. It also is possible that the intervention led caregivers to be more vigilant about symptoms and more empowered to seek care.
Adherence to preventive regimens
Many patients with asthma need to use preventive medications such as daily inhaled corticosteroids to control symptoms. Researchers have developed sensor-based inhaler monitoring interventions to improve treatment adherence, but the effectiveness of these interventions in improving asthma outcomes in urban and minority populations is unclear.
To assess the effectiveness of a clinically integrated, sensor-based inhaler monitoring intervention on improving asthma symptom control and related outcomes in children, Dr. Gupta, of Northwestern University and Ann & Robert H. Lurie Children’s Hospital of Chicago, and colleagues conducted a randomized, unblinded study, known as the Improving Technology-Assisted Recording of Asthma Control in Children (iTRACC) trial. They included 252 children: 127 in the control group and 125 in the intervention group.
Patients in the intervention group received Propeller Health’s Food and Drug Administration–cleared inhaler sensors for inhaled corticosteroids and short-acting beta-agonists. Caregivers could use a mobile application and clinicians could use a Web portal to track patients’ medication use. The app featured personalized insights, educational content, encouragement, surveys, and care team services.
Researchers recruited caregivers and children from five Chicago clinics for the study, which was conducted between 2016 and 2018. They included children aged 4-17 years who had a prescription for daily inhaled corticosteroids for at least 1 year before enrollment. In addition, participants had at least 1 exacerbation requiring oral corticosteroids in the previous year. They excluded children with other respiratory conditions. They also excluded participants who did not speak English because the app was available only in English.
“Sensors monitored inhaled medication use, capturing the date, time, and number of uses, and transmitted this information via Bluetooth to a paired smartphone and the provider portal in real-time,” the authors said.
Clinicians were alerted to call participants if a patient missed inhaled corticosteroid doses for 4 continuous days or used more than 4 short-acting beta-agonist doses per day. Clinicians could help guide asthma management, schedule an appointment, refill medications, and address technical difficulties with the sensors.
The intervention and control groups had similar baseline characteristics. About one-third of the patients were female, and the mean age was 9.3 years. In the control group, 28% identified as Hispanic, and 33% identified as non-Hispanic Black. In the intervention group, 40% identified as Hispanic, and 23% identified as non-Hispanic Black. About 59% reported Medicaid insurance. The intervention and control arms completed electronic surveys at 1, 3, 6, 9, and 12 months.
Average Asthma Control Test score increased from 19 to 22 in the intervention group, compared with an increase from 19 to 20 in the control group. Adjusted rates of emergency department visits and hospitalizations were greater in the intervention group (incidence rate ratios, 2.2 and 3.4, respectively). A measure of caregiver quality of life was greater in the intervention group, although the difference was not significant.
During the trial, more caregivers in the intervention group reported asthma attacks for which steroids were prescribed by a medical office (73% vs. 35%).
Some participants had to manually enter the number of daily puffs into the app because their inhalers were incompatible with the sensors. In addition, some data were missing because of incomplete or missing survey responses and sensor failure over time. “The number of intervention participants with actively transmitting sensors decreased from 102 at baseline to 56 at 12 months,” Dr. Gupta and associates noted.
Important area of research
“One interesting finding of this study is the increase in health care use in the intervention group to nearly twice as many emergency department (ED) visits and three times as many hospitalizations as the control group over 12 months,” Rachelle R. Ramsey, PhD, and Theresa W. Guilbert, MD, MS, of the University of Cincinnati, wrote in a related commentary. “Although it is plausible that, as the authors suggest, greater asthma knowledge and monitoring may have led to increased vigilance of asthma symptoms, it seems that this would have only led to an increase in ED visits but not hospitalizations.”
The mixture of objective electronic monitoring and subjective self-reported adherence may complicate interpretation of the results, they added.
“Overall, this article underscores the feasibility and importance of sensor-based electronic monitoring of adherence in pediatric asthma and encourages future research in this area,” Dr. Ramsey and Dr. Guilbert said.
The trial was supported by the UnitedHealth Group. Dr. Gupta has received grants from the National Institutes of Health, Rho, and other organizations, and has served as a medical consultant and adviser for a variety of companies. Dr. Ramsey is supported by the NIH. Dr. Guilbert reported fees from the American Board of Pediatrics, the Pediatric Pulmonary Subboard, and some pharmaceutical companies, plus grants from the NIH, grants and personal fees from Sanofi, Regeneron, and AstraZeneca, and royalties from UpToDate.
SOURCE: Gupta RS et al. Pediatrics. 2020 Dec 22. doi: 10.1542/peds.2020-1330.
Among children with moderate or severe persistent asthma, a randomized trial suggests.
But the intervention also may lead to more ED visits and increased hospitalization rates.
“We improved asthma symptom control but did not reduce health care use,” Ruchi S. Gupta, MD, MPH, and colleagues, wrote in a study published in Pediatrics.
The monitoring system alerted clinicians when a patient used a short-acting beta-agonist more than four times in a day. It could be that the “alerts enabled providers to detect asthma exacerbation virtually and refer for clinically appropriate care that included directing children to the ED,” the authors suggested. It also is possible that the intervention led caregivers to be more vigilant about symptoms and more empowered to seek care.
Adherence to preventive regimens
Many patients with asthma need to use preventive medications such as daily inhaled corticosteroids to control symptoms. Researchers have developed sensor-based inhaler monitoring interventions to improve treatment adherence, but the effectiveness of these interventions in improving asthma outcomes in urban and minority populations is unclear.
To assess the effectiveness of a clinically integrated, sensor-based inhaler monitoring intervention on improving asthma symptom control and related outcomes in children, Dr. Gupta, of Northwestern University and Ann & Robert H. Lurie Children’s Hospital of Chicago, and colleagues conducted a randomized, unblinded study, known as the Improving Technology-Assisted Recording of Asthma Control in Children (iTRACC) trial. They included 252 children: 127 in the control group and 125 in the intervention group.
Patients in the intervention group received Propeller Health’s Food and Drug Administration–cleared inhaler sensors for inhaled corticosteroids and short-acting beta-agonists. Caregivers could use a mobile application and clinicians could use a Web portal to track patients’ medication use. The app featured personalized insights, educational content, encouragement, surveys, and care team services.
Researchers recruited caregivers and children from five Chicago clinics for the study, which was conducted between 2016 and 2018. They included children aged 4-17 years who had a prescription for daily inhaled corticosteroids for at least 1 year before enrollment. In addition, participants had at least 1 exacerbation requiring oral corticosteroids in the previous year. They excluded children with other respiratory conditions. They also excluded participants who did not speak English because the app was available only in English.
“Sensors monitored inhaled medication use, capturing the date, time, and number of uses, and transmitted this information via Bluetooth to a paired smartphone and the provider portal in real-time,” the authors said.
Clinicians were alerted to call participants if a patient missed inhaled corticosteroid doses for 4 continuous days or used more than 4 short-acting beta-agonist doses per day. Clinicians could help guide asthma management, schedule an appointment, refill medications, and address technical difficulties with the sensors.
The intervention and control groups had similar baseline characteristics. About one-third of the patients were female, and the mean age was 9.3 years. In the control group, 28% identified as Hispanic, and 33% identified as non-Hispanic Black. In the intervention group, 40% identified as Hispanic, and 23% identified as non-Hispanic Black. About 59% reported Medicaid insurance. The intervention and control arms completed electronic surveys at 1, 3, 6, 9, and 12 months.
Average Asthma Control Test score increased from 19 to 22 in the intervention group, compared with an increase from 19 to 20 in the control group. Adjusted rates of emergency department visits and hospitalizations were greater in the intervention group (incidence rate ratios, 2.2 and 3.4, respectively). A measure of caregiver quality of life was greater in the intervention group, although the difference was not significant.
During the trial, more caregivers in the intervention group reported asthma attacks for which steroids were prescribed by a medical office (73% vs. 35%).
Some participants had to manually enter the number of daily puffs into the app because their inhalers were incompatible with the sensors. In addition, some data were missing because of incomplete or missing survey responses and sensor failure over time. “The number of intervention participants with actively transmitting sensors decreased from 102 at baseline to 56 at 12 months,” Dr. Gupta and associates noted.
Important area of research
“One interesting finding of this study is the increase in health care use in the intervention group to nearly twice as many emergency department (ED) visits and three times as many hospitalizations as the control group over 12 months,” Rachelle R. Ramsey, PhD, and Theresa W. Guilbert, MD, MS, of the University of Cincinnati, wrote in a related commentary. “Although it is plausible that, as the authors suggest, greater asthma knowledge and monitoring may have led to increased vigilance of asthma symptoms, it seems that this would have only led to an increase in ED visits but not hospitalizations.”
The mixture of objective electronic monitoring and subjective self-reported adherence may complicate interpretation of the results, they added.
“Overall, this article underscores the feasibility and importance of sensor-based electronic monitoring of adherence in pediatric asthma and encourages future research in this area,” Dr. Ramsey and Dr. Guilbert said.
The trial was supported by the UnitedHealth Group. Dr. Gupta has received grants from the National Institutes of Health, Rho, and other organizations, and has served as a medical consultant and adviser for a variety of companies. Dr. Ramsey is supported by the NIH. Dr. Guilbert reported fees from the American Board of Pediatrics, the Pediatric Pulmonary Subboard, and some pharmaceutical companies, plus grants from the NIH, grants and personal fees from Sanofi, Regeneron, and AstraZeneca, and royalties from UpToDate.
SOURCE: Gupta RS et al. Pediatrics. 2020 Dec 22. doi: 10.1542/peds.2020-1330.
Among children with moderate or severe persistent asthma, a randomized trial suggests.
But the intervention also may lead to more ED visits and increased hospitalization rates.
“We improved asthma symptom control but did not reduce health care use,” Ruchi S. Gupta, MD, MPH, and colleagues, wrote in a study published in Pediatrics.
The monitoring system alerted clinicians when a patient used a short-acting beta-agonist more than four times in a day. It could be that the “alerts enabled providers to detect asthma exacerbation virtually and refer for clinically appropriate care that included directing children to the ED,” the authors suggested. It also is possible that the intervention led caregivers to be more vigilant about symptoms and more empowered to seek care.
Adherence to preventive regimens
Many patients with asthma need to use preventive medications such as daily inhaled corticosteroids to control symptoms. Researchers have developed sensor-based inhaler monitoring interventions to improve treatment adherence, but the effectiveness of these interventions in improving asthma outcomes in urban and minority populations is unclear.
To assess the effectiveness of a clinically integrated, sensor-based inhaler monitoring intervention on improving asthma symptom control and related outcomes in children, Dr. Gupta, of Northwestern University and Ann & Robert H. Lurie Children’s Hospital of Chicago, and colleagues conducted a randomized, unblinded study, known as the Improving Technology-Assisted Recording of Asthma Control in Children (iTRACC) trial. They included 252 children: 127 in the control group and 125 in the intervention group.
Patients in the intervention group received Propeller Health’s Food and Drug Administration–cleared inhaler sensors for inhaled corticosteroids and short-acting beta-agonists. Caregivers could use a mobile application and clinicians could use a Web portal to track patients’ medication use. The app featured personalized insights, educational content, encouragement, surveys, and care team services.
Researchers recruited caregivers and children from five Chicago clinics for the study, which was conducted between 2016 and 2018. They included children aged 4-17 years who had a prescription for daily inhaled corticosteroids for at least 1 year before enrollment. In addition, participants had at least 1 exacerbation requiring oral corticosteroids in the previous year. They excluded children with other respiratory conditions. They also excluded participants who did not speak English because the app was available only in English.
“Sensors monitored inhaled medication use, capturing the date, time, and number of uses, and transmitted this information via Bluetooth to a paired smartphone and the provider portal in real-time,” the authors said.
Clinicians were alerted to call participants if a patient missed inhaled corticosteroid doses for 4 continuous days or used more than 4 short-acting beta-agonist doses per day. Clinicians could help guide asthma management, schedule an appointment, refill medications, and address technical difficulties with the sensors.
The intervention and control groups had similar baseline characteristics. About one-third of the patients were female, and the mean age was 9.3 years. In the control group, 28% identified as Hispanic, and 33% identified as non-Hispanic Black. In the intervention group, 40% identified as Hispanic, and 23% identified as non-Hispanic Black. About 59% reported Medicaid insurance. The intervention and control arms completed electronic surveys at 1, 3, 6, 9, and 12 months.
Average Asthma Control Test score increased from 19 to 22 in the intervention group, compared with an increase from 19 to 20 in the control group. Adjusted rates of emergency department visits and hospitalizations were greater in the intervention group (incidence rate ratios, 2.2 and 3.4, respectively). A measure of caregiver quality of life was greater in the intervention group, although the difference was not significant.
During the trial, more caregivers in the intervention group reported asthma attacks for which steroids were prescribed by a medical office (73% vs. 35%).
Some participants had to manually enter the number of daily puffs into the app because their inhalers were incompatible with the sensors. In addition, some data were missing because of incomplete or missing survey responses and sensor failure over time. “The number of intervention participants with actively transmitting sensors decreased from 102 at baseline to 56 at 12 months,” Dr. Gupta and associates noted.
Important area of research
“One interesting finding of this study is the increase in health care use in the intervention group to nearly twice as many emergency department (ED) visits and three times as many hospitalizations as the control group over 12 months,” Rachelle R. Ramsey, PhD, and Theresa W. Guilbert, MD, MS, of the University of Cincinnati, wrote in a related commentary. “Although it is plausible that, as the authors suggest, greater asthma knowledge and monitoring may have led to increased vigilance of asthma symptoms, it seems that this would have only led to an increase in ED visits but not hospitalizations.”
The mixture of objective electronic monitoring and subjective self-reported adherence may complicate interpretation of the results, they added.
“Overall, this article underscores the feasibility and importance of sensor-based electronic monitoring of adherence in pediatric asthma and encourages future research in this area,” Dr. Ramsey and Dr. Guilbert said.
The trial was supported by the UnitedHealth Group. Dr. Gupta has received grants from the National Institutes of Health, Rho, and other organizations, and has served as a medical consultant and adviser for a variety of companies. Dr. Ramsey is supported by the NIH. Dr. Guilbert reported fees from the American Board of Pediatrics, the Pediatric Pulmonary Subboard, and some pharmaceutical companies, plus grants from the NIH, grants and personal fees from Sanofi, Regeneron, and AstraZeneca, and royalties from UpToDate.
SOURCE: Gupta RS et al. Pediatrics. 2020 Dec 22. doi: 10.1542/peds.2020-1330.
FROM PEDIATRICS
HPV vaccine appears effective for treating warts, particularly in children
The human papillomavirus (HPV) vaccine, recommended by the Centers for Disease Control and Prevention for the prevention of HPV-associated genital warts and neoplasia, appears to be an effective and perhaps underappreciated treatment of existing cutaneous warts, according to expert speaking at the annual Coastal Dermatology symposium, held virtually.
, but a recently published review provides strong evidence that this is a practical clinical strategy, according to Theodore Rosen, MD, professor of dermatology at Baylor College of Medicine, Houston.
“Clearly, if you have someone, particularly a youngster, and you’re having trouble getting rid of their warts and they are age 9 years or above – and they need the vaccine anyhow – that’s a win-win proposition,” Dr. Rosen said.
The current nonavalent HPV vaccine is approved for individuals from age 9 to age 45. Although the CDC recommends routine vaccination at age 11 or 12 years, it allows earlier vaccination within the label.
The recently published and updated evidence of a benefit from treatment comes from a systematic literature review. For the review, 63 articles were drawn from the PubMed and Cochrane databases. The studies yielded 4,439 patients with cutaneous warts at the time they received the HPV vaccine or who specifically received vaccine as a treatment strategy.
As has been suggested previously in the case series and in a limited number of prospective studies, the majority of warts, including cutaneous warts and anogenital warts, resolved following vaccine administration.
“Mostly these were common warts, plantar warts, and flat warts,” Dr. Rosen said, but the paper also reported successful treatment of recurrent respiratory papillomatosis, squamous cell carcinomas, and basal cell carcinomas.
Case reports and small studies associating HPV vaccine with successful resolution of warts are easy to find in the literature. For example, 60% of patients achieved a complete response and 30% a partial response to HPV vaccine in one small prospective study of 26 patients with genital warts. Following vaccination, no recurrences were observed after a median follow-up of more than 8 months.
In the review paper, most of the cases involved patients who received the quadrivalent HPV vaccine, Dr. Rosen noted. Only one received the updated nonavalent vaccine, which, in addition to protection against the 6, 11, 16, and 18 subtypes extends protection to subtypes 31, 33, 45, 52, and 58.
“You would expect the nonavalent vaccine to provide the same protection. It is the same vaccine. It just offers activity against more subtypes,” Dr. Rosen said at the meeting, jointly presented by the University of Louisville and Global Academy for Medical Education. He reported that he personally has used the nonavalent vaccine successfully to treat a cutaneous wart.
The nonavalent vaccine can be administered in just two doses for those who receive the first dose before age 15. In others, it is given in three doses at 1- to 2-month intervals, according to Dr. Rosen. He said the efficacy for preventing genital warts and most HPV-related neoplasia exceeds 90%, although it is lower for penile and anal cancer. The protection extends for at least 10 years, but he said that he believes that it is likely to be longer.
“The HPV vaccine is really, really safe,” Dr. Rosen said. Besides injection-site reactions, the most common adverse event is syncope. For this reason, patients are advised to stay seated for 30 minutes after administration.
There is some evidence for cross-immunity for HPV subtypes not covered by the vaccine, particularly among children, Dr. Rosen commented. Citing the review article, he said that, although almost all HPV-associated warts resolve in children when treated with the vaccine, response is somewhat lower in adolescents and further reduced in adults.
In an interview, the senior author of the recent literature review, Natasha A. Mesinkovska, MD, PhD, associate professor of dermatology, University of California, Irvine, agreed with Dr. Rosen about the value of HPV vaccine for patients not responding to conventional therapies for HPV-related cutaneous warts.
“I think HPV vaccine is an excellent option for those patients, even older ones at 45 years of age if cost is not an issue,” she said. She did offer a caveat. In a recent statement from the International Papillomavirus Society (IPVS) on a world shortage of HPV vaccine, it was estimated that supplies might be limited for the next 3-5 years.
Given this shortage, “obtaining them currently may prove to be difficult,” she cautioned.
This publication and Global Academy for Medical Education are owned by the same parent company.
The human papillomavirus (HPV) vaccine, recommended by the Centers for Disease Control and Prevention for the prevention of HPV-associated genital warts and neoplasia, appears to be an effective and perhaps underappreciated treatment of existing cutaneous warts, according to expert speaking at the annual Coastal Dermatology symposium, held virtually.
, but a recently published review provides strong evidence that this is a practical clinical strategy, according to Theodore Rosen, MD, professor of dermatology at Baylor College of Medicine, Houston.
“Clearly, if you have someone, particularly a youngster, and you’re having trouble getting rid of their warts and they are age 9 years or above – and they need the vaccine anyhow – that’s a win-win proposition,” Dr. Rosen said.
The current nonavalent HPV vaccine is approved for individuals from age 9 to age 45. Although the CDC recommends routine vaccination at age 11 or 12 years, it allows earlier vaccination within the label.
The recently published and updated evidence of a benefit from treatment comes from a systematic literature review. For the review, 63 articles were drawn from the PubMed and Cochrane databases. The studies yielded 4,439 patients with cutaneous warts at the time they received the HPV vaccine or who specifically received vaccine as a treatment strategy.
As has been suggested previously in the case series and in a limited number of prospective studies, the majority of warts, including cutaneous warts and anogenital warts, resolved following vaccine administration.
“Mostly these were common warts, plantar warts, and flat warts,” Dr. Rosen said, but the paper also reported successful treatment of recurrent respiratory papillomatosis, squamous cell carcinomas, and basal cell carcinomas.
Case reports and small studies associating HPV vaccine with successful resolution of warts are easy to find in the literature. For example, 60% of patients achieved a complete response and 30% a partial response to HPV vaccine in one small prospective study of 26 patients with genital warts. Following vaccination, no recurrences were observed after a median follow-up of more than 8 months.
In the review paper, most of the cases involved patients who received the quadrivalent HPV vaccine, Dr. Rosen noted. Only one received the updated nonavalent vaccine, which, in addition to protection against the 6, 11, 16, and 18 subtypes extends protection to subtypes 31, 33, 45, 52, and 58.
“You would expect the nonavalent vaccine to provide the same protection. It is the same vaccine. It just offers activity against more subtypes,” Dr. Rosen said at the meeting, jointly presented by the University of Louisville and Global Academy for Medical Education. He reported that he personally has used the nonavalent vaccine successfully to treat a cutaneous wart.
The nonavalent vaccine can be administered in just two doses for those who receive the first dose before age 15. In others, it is given in three doses at 1- to 2-month intervals, according to Dr. Rosen. He said the efficacy for preventing genital warts and most HPV-related neoplasia exceeds 90%, although it is lower for penile and anal cancer. The protection extends for at least 10 years, but he said that he believes that it is likely to be longer.
“The HPV vaccine is really, really safe,” Dr. Rosen said. Besides injection-site reactions, the most common adverse event is syncope. For this reason, patients are advised to stay seated for 30 minutes after administration.
There is some evidence for cross-immunity for HPV subtypes not covered by the vaccine, particularly among children, Dr. Rosen commented. Citing the review article, he said that, although almost all HPV-associated warts resolve in children when treated with the vaccine, response is somewhat lower in adolescents and further reduced in adults.
In an interview, the senior author of the recent literature review, Natasha A. Mesinkovska, MD, PhD, associate professor of dermatology, University of California, Irvine, agreed with Dr. Rosen about the value of HPV vaccine for patients not responding to conventional therapies for HPV-related cutaneous warts.
“I think HPV vaccine is an excellent option for those patients, even older ones at 45 years of age if cost is not an issue,” she said. She did offer a caveat. In a recent statement from the International Papillomavirus Society (IPVS) on a world shortage of HPV vaccine, it was estimated that supplies might be limited for the next 3-5 years.
Given this shortage, “obtaining them currently may prove to be difficult,” she cautioned.
This publication and Global Academy for Medical Education are owned by the same parent company.
The human papillomavirus (HPV) vaccine, recommended by the Centers for Disease Control and Prevention for the prevention of HPV-associated genital warts and neoplasia, appears to be an effective and perhaps underappreciated treatment of existing cutaneous warts, according to expert speaking at the annual Coastal Dermatology symposium, held virtually.
, but a recently published review provides strong evidence that this is a practical clinical strategy, according to Theodore Rosen, MD, professor of dermatology at Baylor College of Medicine, Houston.
“Clearly, if you have someone, particularly a youngster, and you’re having trouble getting rid of their warts and they are age 9 years or above – and they need the vaccine anyhow – that’s a win-win proposition,” Dr. Rosen said.
The current nonavalent HPV vaccine is approved for individuals from age 9 to age 45. Although the CDC recommends routine vaccination at age 11 or 12 years, it allows earlier vaccination within the label.
The recently published and updated evidence of a benefit from treatment comes from a systematic literature review. For the review, 63 articles were drawn from the PubMed and Cochrane databases. The studies yielded 4,439 patients with cutaneous warts at the time they received the HPV vaccine or who specifically received vaccine as a treatment strategy.
As has been suggested previously in the case series and in a limited number of prospective studies, the majority of warts, including cutaneous warts and anogenital warts, resolved following vaccine administration.
“Mostly these were common warts, plantar warts, and flat warts,” Dr. Rosen said, but the paper also reported successful treatment of recurrent respiratory papillomatosis, squamous cell carcinomas, and basal cell carcinomas.
Case reports and small studies associating HPV vaccine with successful resolution of warts are easy to find in the literature. For example, 60% of patients achieved a complete response and 30% a partial response to HPV vaccine in one small prospective study of 26 patients with genital warts. Following vaccination, no recurrences were observed after a median follow-up of more than 8 months.
In the review paper, most of the cases involved patients who received the quadrivalent HPV vaccine, Dr. Rosen noted. Only one received the updated nonavalent vaccine, which, in addition to protection against the 6, 11, 16, and 18 subtypes extends protection to subtypes 31, 33, 45, 52, and 58.
“You would expect the nonavalent vaccine to provide the same protection. It is the same vaccine. It just offers activity against more subtypes,” Dr. Rosen said at the meeting, jointly presented by the University of Louisville and Global Academy for Medical Education. He reported that he personally has used the nonavalent vaccine successfully to treat a cutaneous wart.
The nonavalent vaccine can be administered in just two doses for those who receive the first dose before age 15. In others, it is given in three doses at 1- to 2-month intervals, according to Dr. Rosen. He said the efficacy for preventing genital warts and most HPV-related neoplasia exceeds 90%, although it is lower for penile and anal cancer. The protection extends for at least 10 years, but he said that he believes that it is likely to be longer.
“The HPV vaccine is really, really safe,” Dr. Rosen said. Besides injection-site reactions, the most common adverse event is syncope. For this reason, patients are advised to stay seated for 30 minutes after administration.
There is some evidence for cross-immunity for HPV subtypes not covered by the vaccine, particularly among children, Dr. Rosen commented. Citing the review article, he said that, although almost all HPV-associated warts resolve in children when treated with the vaccine, response is somewhat lower in adolescents and further reduced in adults.
In an interview, the senior author of the recent literature review, Natasha A. Mesinkovska, MD, PhD, associate professor of dermatology, University of California, Irvine, agreed with Dr. Rosen about the value of HPV vaccine for patients not responding to conventional therapies for HPV-related cutaneous warts.
“I think HPV vaccine is an excellent option for those patients, even older ones at 45 years of age if cost is not an issue,” she said. She did offer a caveat. In a recent statement from the International Papillomavirus Society (IPVS) on a world shortage of HPV vaccine, it was estimated that supplies might be limited for the next 3-5 years.
Given this shortage, “obtaining them currently may prove to be difficult,” she cautioned.
This publication and Global Academy for Medical Education are owned by the same parent company.
FROM COASTAL DERM
Expert picks top pediatric dermatology studies of 2020
Lawrence F. Eichenfield, MD, said at the annual Coastal Dermatology Symposium, held virtually.
Dr. Eichenfield, professor of dermatology and pediatrics, at the University of California, San Diego, presented a list of studies, some of which resulted in approvals of pediatric indications in 2020, that he believes deserve attention.
Crisaborole
Crisaborole ointment, 2% is now approved for topical treatment of children aged as young as 3 months, based on the results of the CrisADe CARE1 phase 4 study. In this open-label study of infants aged from 3 months to under 2 years with mild to moderate AD, treated with crisaborole twice a day for 28 days, the mean reduction from baseline in the Eczema Area and Severity Index (EASI) score was 49.6% on day 15 and 57.5% on day 29. The most common side effects were erythema and application-site pain, but neither occurred in more than 4% of patients. The discontinuation rate was less than 3%.
When the indication for treatment of young children down to age 3 months (from 24 months) was granted by the Food and Drug Administration in March 2020, crisaborole, a phosphodiesterase-4 inhibitor, became the only nonsteroidal approved for treatment of AD in children aged younger than 2 years, Dr. Eichenfield pointed out.
Tacrolimus
The topical calcineurin inhibitor tacrolimus (Protopic) poses no detectable risk of cancer in children treated for AD, according to a prospective, multinational study that followed nearly 8,000 children with AD who used topical tacrolimus for at least 6 weeks over 10 years. With 44,469 person-years of follow-up in a population with at least 6 weeks of exposure to tacrolimus, there were six confirmed cancers, a rate not different than background rates, and no lymphomas.
“I have always tried to educate my patients about the potential use of the topical calcineurin inhibitors while reassuring them that the data did not support significant risk,” Dr. Eichenfield said. However, a large set of data reconfirming a low risk of cancer, although not definitive, “are really nice to have.”
Ruxolitinib
For treatment of AD in children aged as young as 12 years, a cream formulation of ruxolitinib, a Janus kinase 1/JAK2 inhibitor, met its primary outcomes in the phase 3 TRuE AD1 and TRuE AD2 trials. (These data are not yet published but were presented at the Revolutionizing Atopic Dermatitis virtual symposium in April 2020.) The primary endpoint of 75% EASI clearance (EASI-75) was achieved in approximately 62% of patients treated with the 1.5% dose of ruxolitinib twice daily. This was a highly significant advantage over vehicle in both studies (P < .0001).
The EASI-75 rates at 8 weeks for the 0.75% formulation, at 56% and 51.5% for the TRuE AD1 and TRuE AD2 trials, respectively, were lower but also superior (P < .0001) to the 24.6% and 14.4% response rates on vehicle, respectively.
Emphasizing a consistent benefit on multiple secondary endpoints, including the “really early itch decrease,” Dr. Eichenfield described the phase 3 data as “really excellent results.” The data have not yet led to FDA approval of ruxolitinib for AD, but approval seems likely. Dr. Eichenfield noted that other drugs in the same class, such as abrocitinib and upadacitinib, have also demonstrated promising efficacy in children aged 12 years or older.
Dupilumab
Dupilumab, an interleukin-4 receptor alpha antagonist, was approved in May, 2020, for the treatment of AD in children ages 6-11 years, on the basis of a recently published phase 3, randomized, placebo-controlled trial that enrolled children aged between 6 and 11 years, comparing dupilumab and topical corticosteroids and placebo plus topical corticosteroids. Severe involvement was an entry criterion.
At 16 weeks, an EASI-75 response was achieved by 67% of the group randomized to 200 mg of dupilumab administered every 2 weeks and 70% of the group randomized to 300 mg every 4 weeks versus 27% of those randomized to placebo. More patients in the dupilumab arms developed conjunctivitis (10.8% vs. 4.7%) and had injection-site reactions (8.5% vs. 3.5%), but the monoclonal antibody was otherwise well tolerated and safe.
These data suggest that younger patients with severe disease “do, if anything, better than adults,” Dr. Eichenfield said at the meeting, jointly presented by the University of Louisville and Global Academy for Medical Education. He cautioned that avoiding live vaccines, which is recommended in patients on dupilumab, “is likely more of an issue in children.”
Ixekizumab
Ixekizumab has been approved for pediatric patients aged as young as 6 years who are eligible for systemic therapy on the basis of a phase 3 trial. For the primary endpoint of 75% clearance on the Psoriasis Area and Severity Index, the response rates were 89% for the IL-17 inhibitor administered every 4 weeks and 25% for placebo. The study also associated ixekizumab with a significant improvement in quality of life.
The availability of more targeted therapies for children are likely. In Europe, secukinumab, another IL-17 inhibitor, was approved for treatment in pediatric patients this past summer, Dr. Eichenfield noted. These data are not yet published, but he expects targeted therapies to join a growing list of biologics already approved in children.
For drugs with established efficacy and safety, he advised, “look at your pediatric psoriasis patients and don’t be wimpy.” In children with poorly controlled psoriasis, he concluded these drugs have been associated with improved quality of life.
In November 2019, the American Academy of Dermatology and National Psoriasis Foundation published psoriasis management guidelines for children. Not all of the most recently approved therapies are included in these guidelines, which are the first to provide specific recommendations for children, but Dr. Eichenfield also included this publication among his top picks for important contributions to the pediatric dermatology literature in 2020.
Dr. Eichenfield reported financial relationships with 20 pharmaceutical companies that manufacture dermatologic products, including those for the diseases he discussed.
This publication and Global Academy for Medical Education are owned by the same parent company.
Lawrence F. Eichenfield, MD, said at the annual Coastal Dermatology Symposium, held virtually.
Dr. Eichenfield, professor of dermatology and pediatrics, at the University of California, San Diego, presented a list of studies, some of which resulted in approvals of pediatric indications in 2020, that he believes deserve attention.
Crisaborole
Crisaborole ointment, 2% is now approved for topical treatment of children aged as young as 3 months, based on the results of the CrisADe CARE1 phase 4 study. In this open-label study of infants aged from 3 months to under 2 years with mild to moderate AD, treated with crisaborole twice a day for 28 days, the mean reduction from baseline in the Eczema Area and Severity Index (EASI) score was 49.6% on day 15 and 57.5% on day 29. The most common side effects were erythema and application-site pain, but neither occurred in more than 4% of patients. The discontinuation rate was less than 3%.
When the indication for treatment of young children down to age 3 months (from 24 months) was granted by the Food and Drug Administration in March 2020, crisaborole, a phosphodiesterase-4 inhibitor, became the only nonsteroidal approved for treatment of AD in children aged younger than 2 years, Dr. Eichenfield pointed out.
Tacrolimus
The topical calcineurin inhibitor tacrolimus (Protopic) poses no detectable risk of cancer in children treated for AD, according to a prospective, multinational study that followed nearly 8,000 children with AD who used topical tacrolimus for at least 6 weeks over 10 years. With 44,469 person-years of follow-up in a population with at least 6 weeks of exposure to tacrolimus, there were six confirmed cancers, a rate not different than background rates, and no lymphomas.
“I have always tried to educate my patients about the potential use of the topical calcineurin inhibitors while reassuring them that the data did not support significant risk,” Dr. Eichenfield said. However, a large set of data reconfirming a low risk of cancer, although not definitive, “are really nice to have.”
Ruxolitinib
For treatment of AD in children aged as young as 12 years, a cream formulation of ruxolitinib, a Janus kinase 1/JAK2 inhibitor, met its primary outcomes in the phase 3 TRuE AD1 and TRuE AD2 trials. (These data are not yet published but were presented at the Revolutionizing Atopic Dermatitis virtual symposium in April 2020.) The primary endpoint of 75% EASI clearance (EASI-75) was achieved in approximately 62% of patients treated with the 1.5% dose of ruxolitinib twice daily. This was a highly significant advantage over vehicle in both studies (P < .0001).
The EASI-75 rates at 8 weeks for the 0.75% formulation, at 56% and 51.5% for the TRuE AD1 and TRuE AD2 trials, respectively, were lower but also superior (P < .0001) to the 24.6% and 14.4% response rates on vehicle, respectively.
Emphasizing a consistent benefit on multiple secondary endpoints, including the “really early itch decrease,” Dr. Eichenfield described the phase 3 data as “really excellent results.” The data have not yet led to FDA approval of ruxolitinib for AD, but approval seems likely. Dr. Eichenfield noted that other drugs in the same class, such as abrocitinib and upadacitinib, have also demonstrated promising efficacy in children aged 12 years or older.
Dupilumab
Dupilumab, an interleukin-4 receptor alpha antagonist, was approved in May, 2020, for the treatment of AD in children ages 6-11 years, on the basis of a recently published phase 3, randomized, placebo-controlled trial that enrolled children aged between 6 and 11 years, comparing dupilumab and topical corticosteroids and placebo plus topical corticosteroids. Severe involvement was an entry criterion.
At 16 weeks, an EASI-75 response was achieved by 67% of the group randomized to 200 mg of dupilumab administered every 2 weeks and 70% of the group randomized to 300 mg every 4 weeks versus 27% of those randomized to placebo. More patients in the dupilumab arms developed conjunctivitis (10.8% vs. 4.7%) and had injection-site reactions (8.5% vs. 3.5%), but the monoclonal antibody was otherwise well tolerated and safe.
These data suggest that younger patients with severe disease “do, if anything, better than adults,” Dr. Eichenfield said at the meeting, jointly presented by the University of Louisville and Global Academy for Medical Education. He cautioned that avoiding live vaccines, which is recommended in patients on dupilumab, “is likely more of an issue in children.”
Ixekizumab
Ixekizumab has been approved for pediatric patients aged as young as 6 years who are eligible for systemic therapy on the basis of a phase 3 trial. For the primary endpoint of 75% clearance on the Psoriasis Area and Severity Index, the response rates were 89% for the IL-17 inhibitor administered every 4 weeks and 25% for placebo. The study also associated ixekizumab with a significant improvement in quality of life.
The availability of more targeted therapies for children are likely. In Europe, secukinumab, another IL-17 inhibitor, was approved for treatment in pediatric patients this past summer, Dr. Eichenfield noted. These data are not yet published, but he expects targeted therapies to join a growing list of biologics already approved in children.
For drugs with established efficacy and safety, he advised, “look at your pediatric psoriasis patients and don’t be wimpy.” In children with poorly controlled psoriasis, he concluded these drugs have been associated with improved quality of life.
In November 2019, the American Academy of Dermatology and National Psoriasis Foundation published psoriasis management guidelines for children. Not all of the most recently approved therapies are included in these guidelines, which are the first to provide specific recommendations for children, but Dr. Eichenfield also included this publication among his top picks for important contributions to the pediatric dermatology literature in 2020.
Dr. Eichenfield reported financial relationships with 20 pharmaceutical companies that manufacture dermatologic products, including those for the diseases he discussed.
This publication and Global Academy for Medical Education are owned by the same parent company.
Lawrence F. Eichenfield, MD, said at the annual Coastal Dermatology Symposium, held virtually.
Dr. Eichenfield, professor of dermatology and pediatrics, at the University of California, San Diego, presented a list of studies, some of which resulted in approvals of pediatric indications in 2020, that he believes deserve attention.
Crisaborole
Crisaborole ointment, 2% is now approved for topical treatment of children aged as young as 3 months, based on the results of the CrisADe CARE1 phase 4 study. In this open-label study of infants aged from 3 months to under 2 years with mild to moderate AD, treated with crisaborole twice a day for 28 days, the mean reduction from baseline in the Eczema Area and Severity Index (EASI) score was 49.6% on day 15 and 57.5% on day 29. The most common side effects were erythema and application-site pain, but neither occurred in more than 4% of patients. The discontinuation rate was less than 3%.
When the indication for treatment of young children down to age 3 months (from 24 months) was granted by the Food and Drug Administration in March 2020, crisaborole, a phosphodiesterase-4 inhibitor, became the only nonsteroidal approved for treatment of AD in children aged younger than 2 years, Dr. Eichenfield pointed out.
Tacrolimus
The topical calcineurin inhibitor tacrolimus (Protopic) poses no detectable risk of cancer in children treated for AD, according to a prospective, multinational study that followed nearly 8,000 children with AD who used topical tacrolimus for at least 6 weeks over 10 years. With 44,469 person-years of follow-up in a population with at least 6 weeks of exposure to tacrolimus, there were six confirmed cancers, a rate not different than background rates, and no lymphomas.
“I have always tried to educate my patients about the potential use of the topical calcineurin inhibitors while reassuring them that the data did not support significant risk,” Dr. Eichenfield said. However, a large set of data reconfirming a low risk of cancer, although not definitive, “are really nice to have.”
Ruxolitinib
For treatment of AD in children aged as young as 12 years, a cream formulation of ruxolitinib, a Janus kinase 1/JAK2 inhibitor, met its primary outcomes in the phase 3 TRuE AD1 and TRuE AD2 trials. (These data are not yet published but were presented at the Revolutionizing Atopic Dermatitis virtual symposium in April 2020.) The primary endpoint of 75% EASI clearance (EASI-75) was achieved in approximately 62% of patients treated with the 1.5% dose of ruxolitinib twice daily. This was a highly significant advantage over vehicle in both studies (P < .0001).
The EASI-75 rates at 8 weeks for the 0.75% formulation, at 56% and 51.5% for the TRuE AD1 and TRuE AD2 trials, respectively, were lower but also superior (P < .0001) to the 24.6% and 14.4% response rates on vehicle, respectively.
Emphasizing a consistent benefit on multiple secondary endpoints, including the “really early itch decrease,” Dr. Eichenfield described the phase 3 data as “really excellent results.” The data have not yet led to FDA approval of ruxolitinib for AD, but approval seems likely. Dr. Eichenfield noted that other drugs in the same class, such as abrocitinib and upadacitinib, have also demonstrated promising efficacy in children aged 12 years or older.
Dupilumab
Dupilumab, an interleukin-4 receptor alpha antagonist, was approved in May, 2020, for the treatment of AD in children ages 6-11 years, on the basis of a recently published phase 3, randomized, placebo-controlled trial that enrolled children aged between 6 and 11 years, comparing dupilumab and topical corticosteroids and placebo plus topical corticosteroids. Severe involvement was an entry criterion.
At 16 weeks, an EASI-75 response was achieved by 67% of the group randomized to 200 mg of dupilumab administered every 2 weeks and 70% of the group randomized to 300 mg every 4 weeks versus 27% of those randomized to placebo. More patients in the dupilumab arms developed conjunctivitis (10.8% vs. 4.7%) and had injection-site reactions (8.5% vs. 3.5%), but the monoclonal antibody was otherwise well tolerated and safe.
These data suggest that younger patients with severe disease “do, if anything, better than adults,” Dr. Eichenfield said at the meeting, jointly presented by the University of Louisville and Global Academy for Medical Education. He cautioned that avoiding live vaccines, which is recommended in patients on dupilumab, “is likely more of an issue in children.”
Ixekizumab
Ixekizumab has been approved for pediatric patients aged as young as 6 years who are eligible for systemic therapy on the basis of a phase 3 trial. For the primary endpoint of 75% clearance on the Psoriasis Area and Severity Index, the response rates were 89% for the IL-17 inhibitor administered every 4 weeks and 25% for placebo. The study also associated ixekizumab with a significant improvement in quality of life.
The availability of more targeted therapies for children are likely. In Europe, secukinumab, another IL-17 inhibitor, was approved for treatment in pediatric patients this past summer, Dr. Eichenfield noted. These data are not yet published, but he expects targeted therapies to join a growing list of biologics already approved in children.
For drugs with established efficacy and safety, he advised, “look at your pediatric psoriasis patients and don’t be wimpy.” In children with poorly controlled psoriasis, he concluded these drugs have been associated with improved quality of life.
In November 2019, the American Academy of Dermatology and National Psoriasis Foundation published psoriasis management guidelines for children. Not all of the most recently approved therapies are included in these guidelines, which are the first to provide specific recommendations for children, but Dr. Eichenfield also included this publication among his top picks for important contributions to the pediatric dermatology literature in 2020.
Dr. Eichenfield reported financial relationships with 20 pharmaceutical companies that manufacture dermatologic products, including those for the diseases he discussed.
This publication and Global Academy for Medical Education are owned by the same parent company.
FROM COASTAL DERM
Getting closer to a lifesaving RSV vaccine
Louis Bont, MD, PhD, provided an overview of the most recent developments in the complex respiratory syncytial virus (RSV) vaccine landscape at the annual meeting of the European Society for Paediatric Infectious Diseases, held virtually this year.
RSV imposes significant burden worldwide, with 33 million patients, 3 million hospitalizations, and at least 120,000 deaths, reported Dr. Bont of the Wilhelmina Children’s Hospital, University Medical Centre, Utrecht, the Netherlands. Of those deaths, more than 50% are in infants younger than 5 months, and “about 99% of the children dying from RSV live in low- and middle-income countries.”
“There are high-risk populations, such as children with prematurity, congenital heart disease, lung disease, and Down syndrome, but about 73% of all children who are hospitalized for RSV infection were previously healthy children,” Dr. Bont explained. “So, we need to find a solution for all children to prevent RSV infection.”
As observed by Nienke Scheltema in a Lancet Global Health article, population distributions of RSV infection mortality show that, regardless of whether children have comorbidities or they are previously healthy, most children die at a very young age, Dr. Bont explained. These data suggest “that a maternal vaccine or an antibody prophylaxis approach from birth onwards or during the first RSV season is the solution for the problem.”
The path to developing an RSV vaccine has now narrowed its focus onto a structural element of RSV, the prefusion F protein. This shift started with the discovery by Jason McLellan (Science, 2013 [two papers]) that there are two variants of the RSV F-fusion protein: the very stable postfusion conformation and the prefusion active conformation, a metastable protein that exists for a “fraction of a second,” Dr. Bont said.
“The interesting thing is that epitopes that are visible at the prefusion, metastable state … induce highly neutralizing antibodies, whereas epitopes at the postfusion conformation do not,” Dr. Bont explained. “So, by stabilizing the prefusion state, we start inducing neutralizing antibodies that will protect against severe RSV infection, and this is the basic concept of all the vaccine developments currently ongoing.”
These RSV vaccine developments fall into five approach types: live-attenuated or chimeric vaccines, vector-based vaccines, monoclonal antibodies, particle-based vaccines, and subunit or protein-based vaccines.
One breakthrough, which was presented at last year’s ESPID meeting, is the monoclonal antibody nirsevimab. In addition to being nine times more potent than the broadly used antibody palivizumab, it is also more stable; whereas many antibodies have a half-life of 3 weeks, nirsevimab has a half-life of 100 days. “The idea is that a single injection at the start of the RSV season protects children in the first RSV season of their life, a dangerous episode for them.” Dr. Bont explained. The originators, AstraZeneca and Sanofi Pasteur, have “the vision that every child on this planet should receive a single injection with this antibody in the first season,” he explained.
Studies of nanoparticle-based maternal vaccines have also revealed interesting results: Although a phase 3 trial investigating such vaccines didn’t achieve its primary endpoint, “interestingly, 15% of all RSV infections were mild, and only 2% were very severe and leading to hypoxemia,” Dr. Bont noted. “But if we look at vaccine efficacy, we see the opposite – the vaccine was not very efficacious to prevent mild disease, but very efficacious to prevent severe hypoxemia; actually, this is exactly what you would like to see in a vaccine.”
Investigations into live-attenuated and vector-based vaccines have been promising as well, Dr. Bont shared. Studies of live-attenuated vaccines suggest they have a future and that we can move onto their next phase of clinical development, and a study investigating adenoviral vector-based vaccines has demonstrated safety, efficacy, and immunogenicity, though it has also shown that we should anticipate some side effects when using them.
Simple subunit vaccines for RSV are also being explored – a study of DS-Cav1, a stabilized prefusion F subunit protein candidate vaccine, has shown that it has a superior functional profile, compared with previous pre-F subunit vaccines. However, it seemed to be more efficacious against strains of RSV A than strains of RSV B, the dominant strain.
Dr. Bont also discussed exciting work by Sesterhenn et al., in which they used a computer-based program to develop their own vaccine. Using their in-depth knowledge of the RSV prefusion F protein and a computer program, Sesterhenn et al. developed a trivalent vaccine, produced it, and showed – both in vitro and in monkeys – that such vaccines can work up to the level of preclinical in vivo experiments.
“We can now make vaccines behind our computer,” Dr. Bont declared. “And the system doesn’t only work for RSV vaccines, but also for other pathogens – as long as you have an in-depth molecular knowledge of the target epitope,” he added.
Joanne Wildenbeest, MD, PhD, at the Utrecht University, the Netherlands commented: “Lower respiratory tract infections due to RSV are among the leading causes of death worldwide in children under the age of 5, especially young infants. The recent advances in the development of a vaccine and passive immunization are important steps towards the goal to reduce childhood mortality due to RSV worldwide. Since RSV-related mortality is mainly seen in developing countries it is important that, once a vaccine has been approved, it will also be made easily available to these countries.”
Dr. Bont reported the following disclosures: ReSViNET (a nonprofit foundation); investigator-initiated studies with the Bill & Melinda Gates Foundation, AbbVie, MedImmune, and MeMed; participation with Pfizer, Regeneron, and Janssen; and consultancy with GlaxoSmithKline, Ablynx, Novavax, and Janssen.
Louis Bont, MD, PhD, provided an overview of the most recent developments in the complex respiratory syncytial virus (RSV) vaccine landscape at the annual meeting of the European Society for Paediatric Infectious Diseases, held virtually this year.
RSV imposes significant burden worldwide, with 33 million patients, 3 million hospitalizations, and at least 120,000 deaths, reported Dr. Bont of the Wilhelmina Children’s Hospital, University Medical Centre, Utrecht, the Netherlands. Of those deaths, more than 50% are in infants younger than 5 months, and “about 99% of the children dying from RSV live in low- and middle-income countries.”
“There are high-risk populations, such as children with prematurity, congenital heart disease, lung disease, and Down syndrome, but about 73% of all children who are hospitalized for RSV infection were previously healthy children,” Dr. Bont explained. “So, we need to find a solution for all children to prevent RSV infection.”
As observed by Nienke Scheltema in a Lancet Global Health article, population distributions of RSV infection mortality show that, regardless of whether children have comorbidities or they are previously healthy, most children die at a very young age, Dr. Bont explained. These data suggest “that a maternal vaccine or an antibody prophylaxis approach from birth onwards or during the first RSV season is the solution for the problem.”
The path to developing an RSV vaccine has now narrowed its focus onto a structural element of RSV, the prefusion F protein. This shift started with the discovery by Jason McLellan (Science, 2013 [two papers]) that there are two variants of the RSV F-fusion protein: the very stable postfusion conformation and the prefusion active conformation, a metastable protein that exists for a “fraction of a second,” Dr. Bont said.
“The interesting thing is that epitopes that are visible at the prefusion, metastable state … induce highly neutralizing antibodies, whereas epitopes at the postfusion conformation do not,” Dr. Bont explained. “So, by stabilizing the prefusion state, we start inducing neutralizing antibodies that will protect against severe RSV infection, and this is the basic concept of all the vaccine developments currently ongoing.”
These RSV vaccine developments fall into five approach types: live-attenuated or chimeric vaccines, vector-based vaccines, monoclonal antibodies, particle-based vaccines, and subunit or protein-based vaccines.
One breakthrough, which was presented at last year’s ESPID meeting, is the monoclonal antibody nirsevimab. In addition to being nine times more potent than the broadly used antibody palivizumab, it is also more stable; whereas many antibodies have a half-life of 3 weeks, nirsevimab has a half-life of 100 days. “The idea is that a single injection at the start of the RSV season protects children in the first RSV season of their life, a dangerous episode for them.” Dr. Bont explained. The originators, AstraZeneca and Sanofi Pasteur, have “the vision that every child on this planet should receive a single injection with this antibody in the first season,” he explained.
Studies of nanoparticle-based maternal vaccines have also revealed interesting results: Although a phase 3 trial investigating such vaccines didn’t achieve its primary endpoint, “interestingly, 15% of all RSV infections were mild, and only 2% were very severe and leading to hypoxemia,” Dr. Bont noted. “But if we look at vaccine efficacy, we see the opposite – the vaccine was not very efficacious to prevent mild disease, but very efficacious to prevent severe hypoxemia; actually, this is exactly what you would like to see in a vaccine.”
Investigations into live-attenuated and vector-based vaccines have been promising as well, Dr. Bont shared. Studies of live-attenuated vaccines suggest they have a future and that we can move onto their next phase of clinical development, and a study investigating adenoviral vector-based vaccines has demonstrated safety, efficacy, and immunogenicity, though it has also shown that we should anticipate some side effects when using them.
Simple subunit vaccines for RSV are also being explored – a study of DS-Cav1, a stabilized prefusion F subunit protein candidate vaccine, has shown that it has a superior functional profile, compared with previous pre-F subunit vaccines. However, it seemed to be more efficacious against strains of RSV A than strains of RSV B, the dominant strain.
Dr. Bont also discussed exciting work by Sesterhenn et al., in which they used a computer-based program to develop their own vaccine. Using their in-depth knowledge of the RSV prefusion F protein and a computer program, Sesterhenn et al. developed a trivalent vaccine, produced it, and showed – both in vitro and in monkeys – that such vaccines can work up to the level of preclinical in vivo experiments.
“We can now make vaccines behind our computer,” Dr. Bont declared. “And the system doesn’t only work for RSV vaccines, but also for other pathogens – as long as you have an in-depth molecular knowledge of the target epitope,” he added.
Joanne Wildenbeest, MD, PhD, at the Utrecht University, the Netherlands commented: “Lower respiratory tract infections due to RSV are among the leading causes of death worldwide in children under the age of 5, especially young infants. The recent advances in the development of a vaccine and passive immunization are important steps towards the goal to reduce childhood mortality due to RSV worldwide. Since RSV-related mortality is mainly seen in developing countries it is important that, once a vaccine has been approved, it will also be made easily available to these countries.”
Dr. Bont reported the following disclosures: ReSViNET (a nonprofit foundation); investigator-initiated studies with the Bill & Melinda Gates Foundation, AbbVie, MedImmune, and MeMed; participation with Pfizer, Regeneron, and Janssen; and consultancy with GlaxoSmithKline, Ablynx, Novavax, and Janssen.
Louis Bont, MD, PhD, provided an overview of the most recent developments in the complex respiratory syncytial virus (RSV) vaccine landscape at the annual meeting of the European Society for Paediatric Infectious Diseases, held virtually this year.
RSV imposes significant burden worldwide, with 33 million patients, 3 million hospitalizations, and at least 120,000 deaths, reported Dr. Bont of the Wilhelmina Children’s Hospital, University Medical Centre, Utrecht, the Netherlands. Of those deaths, more than 50% are in infants younger than 5 months, and “about 99% of the children dying from RSV live in low- and middle-income countries.”
“There are high-risk populations, such as children with prematurity, congenital heart disease, lung disease, and Down syndrome, but about 73% of all children who are hospitalized for RSV infection were previously healthy children,” Dr. Bont explained. “So, we need to find a solution for all children to prevent RSV infection.”
As observed by Nienke Scheltema in a Lancet Global Health article, population distributions of RSV infection mortality show that, regardless of whether children have comorbidities or they are previously healthy, most children die at a very young age, Dr. Bont explained. These data suggest “that a maternal vaccine or an antibody prophylaxis approach from birth onwards or during the first RSV season is the solution for the problem.”
The path to developing an RSV vaccine has now narrowed its focus onto a structural element of RSV, the prefusion F protein. This shift started with the discovery by Jason McLellan (Science, 2013 [two papers]) that there are two variants of the RSV F-fusion protein: the very stable postfusion conformation and the prefusion active conformation, a metastable protein that exists for a “fraction of a second,” Dr. Bont said.
“The interesting thing is that epitopes that are visible at the prefusion, metastable state … induce highly neutralizing antibodies, whereas epitopes at the postfusion conformation do not,” Dr. Bont explained. “So, by stabilizing the prefusion state, we start inducing neutralizing antibodies that will protect against severe RSV infection, and this is the basic concept of all the vaccine developments currently ongoing.”
These RSV vaccine developments fall into five approach types: live-attenuated or chimeric vaccines, vector-based vaccines, monoclonal antibodies, particle-based vaccines, and subunit or protein-based vaccines.
One breakthrough, which was presented at last year’s ESPID meeting, is the monoclonal antibody nirsevimab. In addition to being nine times more potent than the broadly used antibody palivizumab, it is also more stable; whereas many antibodies have a half-life of 3 weeks, nirsevimab has a half-life of 100 days. “The idea is that a single injection at the start of the RSV season protects children in the first RSV season of their life, a dangerous episode for them.” Dr. Bont explained. The originators, AstraZeneca and Sanofi Pasteur, have “the vision that every child on this planet should receive a single injection with this antibody in the first season,” he explained.
Studies of nanoparticle-based maternal vaccines have also revealed interesting results: Although a phase 3 trial investigating such vaccines didn’t achieve its primary endpoint, “interestingly, 15% of all RSV infections were mild, and only 2% were very severe and leading to hypoxemia,” Dr. Bont noted. “But if we look at vaccine efficacy, we see the opposite – the vaccine was not very efficacious to prevent mild disease, but very efficacious to prevent severe hypoxemia; actually, this is exactly what you would like to see in a vaccine.”
Investigations into live-attenuated and vector-based vaccines have been promising as well, Dr. Bont shared. Studies of live-attenuated vaccines suggest they have a future and that we can move onto their next phase of clinical development, and a study investigating adenoviral vector-based vaccines has demonstrated safety, efficacy, and immunogenicity, though it has also shown that we should anticipate some side effects when using them.
Simple subunit vaccines for RSV are also being explored – a study of DS-Cav1, a stabilized prefusion F subunit protein candidate vaccine, has shown that it has a superior functional profile, compared with previous pre-F subunit vaccines. However, it seemed to be more efficacious against strains of RSV A than strains of RSV B, the dominant strain.
Dr. Bont also discussed exciting work by Sesterhenn et al., in which they used a computer-based program to develop their own vaccine. Using their in-depth knowledge of the RSV prefusion F protein and a computer program, Sesterhenn et al. developed a trivalent vaccine, produced it, and showed – both in vitro and in monkeys – that such vaccines can work up to the level of preclinical in vivo experiments.
“We can now make vaccines behind our computer,” Dr. Bont declared. “And the system doesn’t only work for RSV vaccines, but also for other pathogens – as long as you have an in-depth molecular knowledge of the target epitope,” he added.
Joanne Wildenbeest, MD, PhD, at the Utrecht University, the Netherlands commented: “Lower respiratory tract infections due to RSV are among the leading causes of death worldwide in children under the age of 5, especially young infants. The recent advances in the development of a vaccine and passive immunization are important steps towards the goal to reduce childhood mortality due to RSV worldwide. Since RSV-related mortality is mainly seen in developing countries it is important that, once a vaccine has been approved, it will also be made easily available to these countries.”
Dr. Bont reported the following disclosures: ReSViNET (a nonprofit foundation); investigator-initiated studies with the Bill & Melinda Gates Foundation, AbbVie, MedImmune, and MeMed; participation with Pfizer, Regeneron, and Janssen; and consultancy with GlaxoSmithKline, Ablynx, Novavax, and Janssen.
FROM ESPID 2020
Analysis characterizes common wound microbes in epidermolysis bullosa
– in a retrospective analysis of over 700 wound cultures from 158 patients across the United States and Canada.
The findings from the EB Clinical Characterization and Outcomes Database speak to the value of surveillance cultures with routine testing for microbial resistance – including mupirocin resistance – and to the importance of antibiotic stewardship not only for oral antibiotics but for topicals as well, according to Laura E. Levin, MD, and Kimberly D. Morel, MD, of the departments of dermatology and pediatrics, Columbia University Irving Medical Center, New York, the lead and senior authors, respectively, of the paper recently published in Pediatric Dermatology.
Almost all of the 158 patients with at least one wound culture recorded in the database from the period of 2001-2018 had one or more positive culture results. Of 152 patients with positive cultures, 131 (86%) were positive for SA and 56 (37%) and 34 (22%) were positive for PA and GAS, respectively. Other bacteria isolated included Corynebacterium spp and Proteus spp. Nearly half (47%) of patients with SA-positive cultures had methicillin-resistant SA, and 68% had methicillin-susceptible SA. (Some patients grew both MSSA and MRSA at different points in time.)
Mupirocin-susceptibility testing was performed at only some of the 13 participating centers. Of 15 patients whose cultures had recorded SA mupirocin-susceptibility testing, 11 had cultures positive for mupirocin-susceptible SA and 6 (40%) had mupirocin-resistant SA isolates (2 patients grew both). Of these six patients, half had isolates that were also methicillin-resistant.
Mupirocin, a topical antibiotic, has been a cornerstone of decolonization regimens for MSSA and MRSA, but resistance has been demonstrated in other research as well and is not specific to EB, wrote Dr. Levin, Dr. Morel, and coauthors.
“Pediatric dermatologists often rely on topical antimicrobials in the treatment of patients’ open wounds to both prevent and treat infection, depending on the clinical scenario,” and surveillance cultures with routine testing for mupirocin resistance can help guide antibiotic choice and management strategies, Dr. Levin said in an interview.
More broadly, she added, “it’s helpful to know what bacteria are routinely colonizing wounds, not causing infection, versus those that are more likely to be associated with infection, chronic wounds, or the risk of developing skin cancer ... [to know] which wounds need to be treated more aggressively.”
A subset of patients with EB have been known to be at risk for squamous cell carcinoma, and research is implicating certain bacteria “as contributing to wound inflammation,” Dr. Morel said in an interview.
SCC was reported in 23 out of 717 patients in the database – but fewer than half of the patients with SCC had recorded wound cultures. The small numbers precluded the identification of microbes that may confer significant risk.
Correlating particular microbes with clinical features also will take more research. About half (57%) of the patients with recorded wound cultures had wounds with purulent exudate or other features of clinical infection. However, the presence or absence of clinical signs of infection was not temporally correlated with culture results in the database.
The 158 patients with recorded wound cultures had a mean age of 12.8 years and represented a range of EB subtypes.
PA was present in the wounds of patients as young as 1 month old, the authors noted. Investigators are “looking to further study PA and characterize clinical features ... to understand more about this microbe and its impact on patients with EB,” Dr. Morel said.
In the meantime, the analysis reaffirms the importance of antibiotic stewardship. Mupirocin is labeled to be used three times a day for a short period of time, but “tends to be prescribed and used less judiciously than intended,” Dr. Morel said. “It’s important [not to overuse it]. We have seen that patients’ culture results become sensitive to mupirocin again in the future when they avoid it for a period of time.”
The work was supported by the EB Research Partnership and EB Medical Research Foundation, as well as an NIH/NCATS grant. No investigator disclosures were listed.
SOURCE: Pediatr Dermatol. 2020 Nov 28. doi: 10.1111/pde.14444.
– in a retrospective analysis of over 700 wound cultures from 158 patients across the United States and Canada.
The findings from the EB Clinical Characterization and Outcomes Database speak to the value of surveillance cultures with routine testing for microbial resistance – including mupirocin resistance – and to the importance of antibiotic stewardship not only for oral antibiotics but for topicals as well, according to Laura E. Levin, MD, and Kimberly D. Morel, MD, of the departments of dermatology and pediatrics, Columbia University Irving Medical Center, New York, the lead and senior authors, respectively, of the paper recently published in Pediatric Dermatology.
Almost all of the 158 patients with at least one wound culture recorded in the database from the period of 2001-2018 had one or more positive culture results. Of 152 patients with positive cultures, 131 (86%) were positive for SA and 56 (37%) and 34 (22%) were positive for PA and GAS, respectively. Other bacteria isolated included Corynebacterium spp and Proteus spp. Nearly half (47%) of patients with SA-positive cultures had methicillin-resistant SA, and 68% had methicillin-susceptible SA. (Some patients grew both MSSA and MRSA at different points in time.)
Mupirocin-susceptibility testing was performed at only some of the 13 participating centers. Of 15 patients whose cultures had recorded SA mupirocin-susceptibility testing, 11 had cultures positive for mupirocin-susceptible SA and 6 (40%) had mupirocin-resistant SA isolates (2 patients grew both). Of these six patients, half had isolates that were also methicillin-resistant.
Mupirocin, a topical antibiotic, has been a cornerstone of decolonization regimens for MSSA and MRSA, but resistance has been demonstrated in other research as well and is not specific to EB, wrote Dr. Levin, Dr. Morel, and coauthors.
“Pediatric dermatologists often rely on topical antimicrobials in the treatment of patients’ open wounds to both prevent and treat infection, depending on the clinical scenario,” and surveillance cultures with routine testing for mupirocin resistance can help guide antibiotic choice and management strategies, Dr. Levin said in an interview.
More broadly, she added, “it’s helpful to know what bacteria are routinely colonizing wounds, not causing infection, versus those that are more likely to be associated with infection, chronic wounds, or the risk of developing skin cancer ... [to know] which wounds need to be treated more aggressively.”
A subset of patients with EB have been known to be at risk for squamous cell carcinoma, and research is implicating certain bacteria “as contributing to wound inflammation,” Dr. Morel said in an interview.
SCC was reported in 23 out of 717 patients in the database – but fewer than half of the patients with SCC had recorded wound cultures. The small numbers precluded the identification of microbes that may confer significant risk.
Correlating particular microbes with clinical features also will take more research. About half (57%) of the patients with recorded wound cultures had wounds with purulent exudate or other features of clinical infection. However, the presence or absence of clinical signs of infection was not temporally correlated with culture results in the database.
The 158 patients with recorded wound cultures had a mean age of 12.8 years and represented a range of EB subtypes.
PA was present in the wounds of patients as young as 1 month old, the authors noted. Investigators are “looking to further study PA and characterize clinical features ... to understand more about this microbe and its impact on patients with EB,” Dr. Morel said.
In the meantime, the analysis reaffirms the importance of antibiotic stewardship. Mupirocin is labeled to be used three times a day for a short period of time, but “tends to be prescribed and used less judiciously than intended,” Dr. Morel said. “It’s important [not to overuse it]. We have seen that patients’ culture results become sensitive to mupirocin again in the future when they avoid it for a period of time.”
The work was supported by the EB Research Partnership and EB Medical Research Foundation, as well as an NIH/NCATS grant. No investigator disclosures were listed.
SOURCE: Pediatr Dermatol. 2020 Nov 28. doi: 10.1111/pde.14444.
– in a retrospective analysis of over 700 wound cultures from 158 patients across the United States and Canada.
The findings from the EB Clinical Characterization and Outcomes Database speak to the value of surveillance cultures with routine testing for microbial resistance – including mupirocin resistance – and to the importance of antibiotic stewardship not only for oral antibiotics but for topicals as well, according to Laura E. Levin, MD, and Kimberly D. Morel, MD, of the departments of dermatology and pediatrics, Columbia University Irving Medical Center, New York, the lead and senior authors, respectively, of the paper recently published in Pediatric Dermatology.
Almost all of the 158 patients with at least one wound culture recorded in the database from the period of 2001-2018 had one or more positive culture results. Of 152 patients with positive cultures, 131 (86%) were positive for SA and 56 (37%) and 34 (22%) were positive for PA and GAS, respectively. Other bacteria isolated included Corynebacterium spp and Proteus spp. Nearly half (47%) of patients with SA-positive cultures had methicillin-resistant SA, and 68% had methicillin-susceptible SA. (Some patients grew both MSSA and MRSA at different points in time.)
Mupirocin-susceptibility testing was performed at only some of the 13 participating centers. Of 15 patients whose cultures had recorded SA mupirocin-susceptibility testing, 11 had cultures positive for mupirocin-susceptible SA and 6 (40%) had mupirocin-resistant SA isolates (2 patients grew both). Of these six patients, half had isolates that were also methicillin-resistant.
Mupirocin, a topical antibiotic, has been a cornerstone of decolonization regimens for MSSA and MRSA, but resistance has been demonstrated in other research as well and is not specific to EB, wrote Dr. Levin, Dr. Morel, and coauthors.
“Pediatric dermatologists often rely on topical antimicrobials in the treatment of patients’ open wounds to both prevent and treat infection, depending on the clinical scenario,” and surveillance cultures with routine testing for mupirocin resistance can help guide antibiotic choice and management strategies, Dr. Levin said in an interview.
More broadly, she added, “it’s helpful to know what bacteria are routinely colonizing wounds, not causing infection, versus those that are more likely to be associated with infection, chronic wounds, or the risk of developing skin cancer ... [to know] which wounds need to be treated more aggressively.”
A subset of patients with EB have been known to be at risk for squamous cell carcinoma, and research is implicating certain bacteria “as contributing to wound inflammation,” Dr. Morel said in an interview.
SCC was reported in 23 out of 717 patients in the database – but fewer than half of the patients with SCC had recorded wound cultures. The small numbers precluded the identification of microbes that may confer significant risk.
Correlating particular microbes with clinical features also will take more research. About half (57%) of the patients with recorded wound cultures had wounds with purulent exudate or other features of clinical infection. However, the presence or absence of clinical signs of infection was not temporally correlated with culture results in the database.
The 158 patients with recorded wound cultures had a mean age of 12.8 years and represented a range of EB subtypes.
PA was present in the wounds of patients as young as 1 month old, the authors noted. Investigators are “looking to further study PA and characterize clinical features ... to understand more about this microbe and its impact on patients with EB,” Dr. Morel said.
In the meantime, the analysis reaffirms the importance of antibiotic stewardship. Mupirocin is labeled to be used three times a day for a short period of time, but “tends to be prescribed and used less judiciously than intended,” Dr. Morel said. “It’s important [not to overuse it]. We have seen that patients’ culture results become sensitive to mupirocin again in the future when they avoid it for a period of time.”
The work was supported by the EB Research Partnership and EB Medical Research Foundation, as well as an NIH/NCATS grant. No investigator disclosures were listed.
SOURCE: Pediatr Dermatol. 2020 Nov 28. doi: 10.1111/pde.14444.
FROM PEDIATRIC DERMATOLOGY