User login
Tide beginning to turn on vaccine hesitancy
NEW ORLEANS –
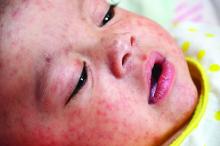
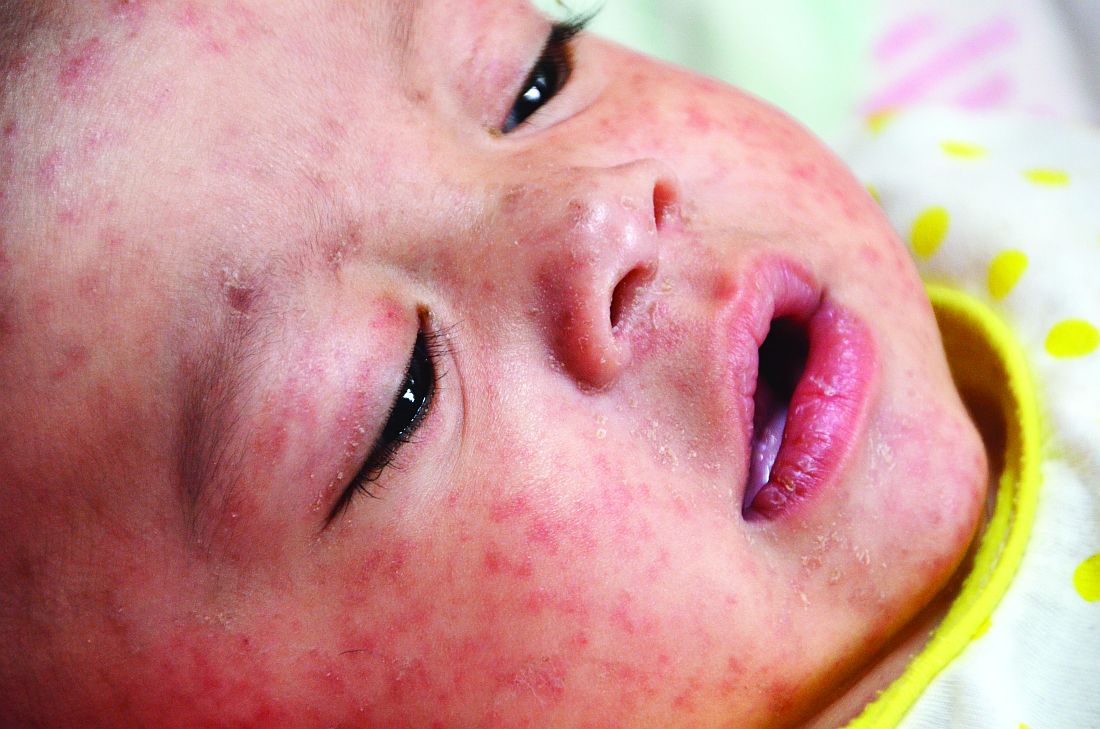
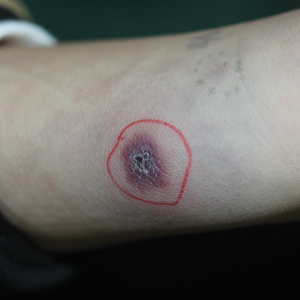
The shift began with the measles outbreak in Southern California in late 2014, he said. According to the Centers for Disease Control and Prevention, 125 measles cases with rash that occurred between Dec. 28, 2014, and Feb. 8, 2015, were confirmed in U.S. residents. Of these, 100 were California residents (MMWR. 2015 Feb 20;64[06];153-4).
“This outbreak spread ultimately to 25 states and involved 189 people,” Dr. Offit said at the annual meeting of the American Academy of Pediatrics. “It was in the news almost every day. As a consequence, there were measles outbreaks in New York, New Jersey, Florida, Oregon, and Texas, and Washington, which began to turn the public sentiment against the antivaccine movement.”
Even longstanding skeptics are changing their tune. Dr. Offit, professor of pediatrics in the division of infectious diseases at the Children’s Hospital of Philadelphia, cited a recent study from the Autism Science Foundation which found that 85% of parents of children with autism spectrum disorder don’t believe that vaccines cause the condition. “Although there will be parents who continue to believe that vaccines cause autism, most parents of children with autism don’t believe that,” he said. “Also, it’s a little hard to make your case that vaccines are dangerous and that you shouldn’t get them in the midst of outbreaks.”
Perhaps the greatest pushback against antivaccination efforts has been made in the legal arena. In 2019 alone, legislators in California banned parents from not vaccinating their kids because of personal beliefs, while lawmakers in New York repealed the religious exemption to vaccinate, those in Maine repealed the religious and philosophical exemption, those in New Jersey required detailed written explanation for religious exemption, and those in Washington State repealed the philosophical exemption for the MMR vaccine.
Pushback also is apparent on various social media platforms. For example, Dr. Offit said, Pinterest restricts vaccine search results to curb the spread of misinformation, YouTube removes ads from antivaccine channels, Amazon Prime has pulled antivaccination documentaries from its video service, and Facebook has taken steps to curb misinformation about vaccines. “With outbreaks and with children suffering, the media and public sentiment has largely turned against those who are vehemently against vaccines,” he said. “I’m talking about an angry, politically connected, lawyer-backed group of people who are conspiracy theorists, [those] who no matter what you say, they’re going to believe there’s a conspiracy theory to hurt their children and not believe you. When that group becomes big enough and you start to see outbreaks like we’ve seen, then it becomes an issue. That’s where it comes down to legislation. Is it your inalienable right as a U.S. citizen to allow your child to catch and transmit a potentially fatal infection? That’s what we’re struggling with now.”
When meeting with parents who are skeptical about vaccines or refuse their children to have them, Dr. Offit advises clinicians to “go down swinging” in favor of vaccination. He shared how his wife, Bonnie, a pediatrician who practices in suburban Philadelphia, counsels parents who raise such concerns. “The way she handled it initially was to do the best she could to eventually get people vaccinated,” he said. “She was successful about one-quarter of the time. Then she drew a line. She started saying to parents, ‘Look; don’t put me in a position where you are asking me to practice substandard care. I can’t send them out of this room knowing that there’s more measles out there, knowing that there’s mumps out there, knowing that there’s whooping cough out there, knowing that there’s pneumococcus and varicella out there. If this child leaves this office and is hurt by any of those viruses or bacteria and I knew I could have done something to prevent it, I couldn’t live with myself. If you’re going to let this child out without being vaccinated I can’t see you anymore because I’m responsible for the health of this child.’ With that [approach], she has been far more successful. Because at some level, if you continue to see that patient, you’re tacitly agreeing that it’s okay to [not vaccinate].”
In 2000, Dr. Offit and colleagues created the Vaccine Education Center at Children’s Hospital of Philadelphia, which provides complete, up-to-date, and reliable information about vaccines to parents and clinicians. It summarizes the purpose of each vaccine, and the relative risks and benefits in easy-to-read language. The CDC also maintains updated information about vaccines and immunizations on its web site. For his part, Dr. Offit tells parents that passing on an opportunity to vaccinate their child is not a risk-free choice. “If you choose not to get a vaccine you probably will get away with it, but you might not,” he said. “You are playing a game of Russian roulette. It may not be five empty chambers and one bullet, but maybe it’s 100,000 empty chambers and one bullet. There’s a bullet there.”
Dr. Offit reported having no relevant financial disclosures.
NEW ORLEANS –
The shift began with the measles outbreak in Southern California in late 2014, he said. According to the Centers for Disease Control and Prevention, 125 measles cases with rash that occurred between Dec. 28, 2014, and Feb. 8, 2015, were confirmed in U.S. residents. Of these, 100 were California residents (MMWR. 2015 Feb 20;64[06];153-4).
“This outbreak spread ultimately to 25 states and involved 189 people,” Dr. Offit said at the annual meeting of the American Academy of Pediatrics. “It was in the news almost every day. As a consequence, there were measles outbreaks in New York, New Jersey, Florida, Oregon, and Texas, and Washington, which began to turn the public sentiment against the antivaccine movement.”
Even longstanding skeptics are changing their tune. Dr. Offit, professor of pediatrics in the division of infectious diseases at the Children’s Hospital of Philadelphia, cited a recent study from the Autism Science Foundation which found that 85% of parents of children with autism spectrum disorder don’t believe that vaccines cause the condition. “Although there will be parents who continue to believe that vaccines cause autism, most parents of children with autism don’t believe that,” he said. “Also, it’s a little hard to make your case that vaccines are dangerous and that you shouldn’t get them in the midst of outbreaks.”
Perhaps the greatest pushback against antivaccination efforts has been made in the legal arena. In 2019 alone, legislators in California banned parents from not vaccinating their kids because of personal beliefs, while lawmakers in New York repealed the religious exemption to vaccinate, those in Maine repealed the religious and philosophical exemption, those in New Jersey required detailed written explanation for religious exemption, and those in Washington State repealed the philosophical exemption for the MMR vaccine.
Pushback also is apparent on various social media platforms. For example, Dr. Offit said, Pinterest restricts vaccine search results to curb the spread of misinformation, YouTube removes ads from antivaccine channels, Amazon Prime has pulled antivaccination documentaries from its video service, and Facebook has taken steps to curb misinformation about vaccines. “With outbreaks and with children suffering, the media and public sentiment has largely turned against those who are vehemently against vaccines,” he said. “I’m talking about an angry, politically connected, lawyer-backed group of people who are conspiracy theorists, [those] who no matter what you say, they’re going to believe there’s a conspiracy theory to hurt their children and not believe you. When that group becomes big enough and you start to see outbreaks like we’ve seen, then it becomes an issue. That’s where it comes down to legislation. Is it your inalienable right as a U.S. citizen to allow your child to catch and transmit a potentially fatal infection? That’s what we’re struggling with now.”
When meeting with parents who are skeptical about vaccines or refuse their children to have them, Dr. Offit advises clinicians to “go down swinging” in favor of vaccination. He shared how his wife, Bonnie, a pediatrician who practices in suburban Philadelphia, counsels parents who raise such concerns. “The way she handled it initially was to do the best she could to eventually get people vaccinated,” he said. “She was successful about one-quarter of the time. Then she drew a line. She started saying to parents, ‘Look; don’t put me in a position where you are asking me to practice substandard care. I can’t send them out of this room knowing that there’s more measles out there, knowing that there’s mumps out there, knowing that there’s whooping cough out there, knowing that there’s pneumococcus and varicella out there. If this child leaves this office and is hurt by any of those viruses or bacteria and I knew I could have done something to prevent it, I couldn’t live with myself. If you’re going to let this child out without being vaccinated I can’t see you anymore because I’m responsible for the health of this child.’ With that [approach], she has been far more successful. Because at some level, if you continue to see that patient, you’re tacitly agreeing that it’s okay to [not vaccinate].”
In 2000, Dr. Offit and colleagues created the Vaccine Education Center at Children’s Hospital of Philadelphia, which provides complete, up-to-date, and reliable information about vaccines to parents and clinicians. It summarizes the purpose of each vaccine, and the relative risks and benefits in easy-to-read language. The CDC also maintains updated information about vaccines and immunizations on its web site. For his part, Dr. Offit tells parents that passing on an opportunity to vaccinate their child is not a risk-free choice. “If you choose not to get a vaccine you probably will get away with it, but you might not,” he said. “You are playing a game of Russian roulette. It may not be five empty chambers and one bullet, but maybe it’s 100,000 empty chambers and one bullet. There’s a bullet there.”
Dr. Offit reported having no relevant financial disclosures.
NEW ORLEANS –
The shift began with the measles outbreak in Southern California in late 2014, he said. According to the Centers for Disease Control and Prevention, 125 measles cases with rash that occurred between Dec. 28, 2014, and Feb. 8, 2015, were confirmed in U.S. residents. Of these, 100 were California residents (MMWR. 2015 Feb 20;64[06];153-4).
“This outbreak spread ultimately to 25 states and involved 189 people,” Dr. Offit said at the annual meeting of the American Academy of Pediatrics. “It was in the news almost every day. As a consequence, there were measles outbreaks in New York, New Jersey, Florida, Oregon, and Texas, and Washington, which began to turn the public sentiment against the antivaccine movement.”
Even longstanding skeptics are changing their tune. Dr. Offit, professor of pediatrics in the division of infectious diseases at the Children’s Hospital of Philadelphia, cited a recent study from the Autism Science Foundation which found that 85% of parents of children with autism spectrum disorder don’t believe that vaccines cause the condition. “Although there will be parents who continue to believe that vaccines cause autism, most parents of children with autism don’t believe that,” he said. “Also, it’s a little hard to make your case that vaccines are dangerous and that you shouldn’t get them in the midst of outbreaks.”
Perhaps the greatest pushback against antivaccination efforts has been made in the legal arena. In 2019 alone, legislators in California banned parents from not vaccinating their kids because of personal beliefs, while lawmakers in New York repealed the religious exemption to vaccinate, those in Maine repealed the religious and philosophical exemption, those in New Jersey required detailed written explanation for religious exemption, and those in Washington State repealed the philosophical exemption for the MMR vaccine.
Pushback also is apparent on various social media platforms. For example, Dr. Offit said, Pinterest restricts vaccine search results to curb the spread of misinformation, YouTube removes ads from antivaccine channels, Amazon Prime has pulled antivaccination documentaries from its video service, and Facebook has taken steps to curb misinformation about vaccines. “With outbreaks and with children suffering, the media and public sentiment has largely turned against those who are vehemently against vaccines,” he said. “I’m talking about an angry, politically connected, lawyer-backed group of people who are conspiracy theorists, [those] who no matter what you say, they’re going to believe there’s a conspiracy theory to hurt their children and not believe you. When that group becomes big enough and you start to see outbreaks like we’ve seen, then it becomes an issue. That’s where it comes down to legislation. Is it your inalienable right as a U.S. citizen to allow your child to catch and transmit a potentially fatal infection? That’s what we’re struggling with now.”
When meeting with parents who are skeptical about vaccines or refuse their children to have them, Dr. Offit advises clinicians to “go down swinging” in favor of vaccination. He shared how his wife, Bonnie, a pediatrician who practices in suburban Philadelphia, counsels parents who raise such concerns. “The way she handled it initially was to do the best she could to eventually get people vaccinated,” he said. “She was successful about one-quarter of the time. Then she drew a line. She started saying to parents, ‘Look; don’t put me in a position where you are asking me to practice substandard care. I can’t send them out of this room knowing that there’s more measles out there, knowing that there’s mumps out there, knowing that there’s whooping cough out there, knowing that there’s pneumococcus and varicella out there. If this child leaves this office and is hurt by any of those viruses or bacteria and I knew I could have done something to prevent it, I couldn’t live with myself. If you’re going to let this child out without being vaccinated I can’t see you anymore because I’m responsible for the health of this child.’ With that [approach], she has been far more successful. Because at some level, if you continue to see that patient, you’re tacitly agreeing that it’s okay to [not vaccinate].”
In 2000, Dr. Offit and colleagues created the Vaccine Education Center at Children’s Hospital of Philadelphia, which provides complete, up-to-date, and reliable information about vaccines to parents and clinicians. It summarizes the purpose of each vaccine, and the relative risks and benefits in easy-to-read language. The CDC also maintains updated information about vaccines and immunizations on its web site. For his part, Dr. Offit tells parents that passing on an opportunity to vaccinate their child is not a risk-free choice. “If you choose not to get a vaccine you probably will get away with it, but you might not,” he said. “You are playing a game of Russian roulette. It may not be five empty chambers and one bullet, but maybe it’s 100,000 empty chambers and one bullet. There’s a bullet there.”
Dr. Offit reported having no relevant financial disclosures.
EXPERT ANALYSIS FROM AAP 2019
AAD-NPF Pediatric psoriasis guideline advises on physical and mental care
Psoriasis management in children involves attention not only to treatment of the physical condition but also psychosocial wellness and quality of life, according to
Psoriasis affects approximately 1% of children, either alone or associated with comorbid conditions such as psoriatic arthritis (PsA), wrote Alan Menter, MD, of Baylor University Medical Center, Dallas, and coauthors of the guideline.
In the guideline, published in the Journal of the American Academy of Dermatology, the multidisciplinary work group identified screening tools to measure disease severity, strategies for management of comorbidities, and the safety and effectiveness of topical, systemic, and phototherapy treatments.
To assess disease severity, the work group recommended not only the use of body surface area (BSA), similar to measurement of severity in adults, but also the use of the Children’s Dermatology Life Quality Index, a 10-question quality of life survey, as BSA alone does not account for the potential negative impact of the disease on quality of life in terms of physical, emotional, social, and psychological function.
“For example, a child with psoriasis limited to the face or the entire scalp does not have severe disease based on BSA definitions, but if this involvement causes shame, social withdrawal, or bullying, it satisfies criteria for severe disease based on impact beyond the skin,” they said.
The work group stated that a variety of conditions may trigger or exacerbate psoriasis in children, including infections, cutaneous trauma, or physiological, emotional, and environmental stressors.
The majority of children with PsA develop joint inflammation before skin disease, the work group wrote. In addition, children with psoriasis are at increased risk for rheumatoid arthritis, so clinicians may need to distinguish between a combination of psoriasis and musculoskeletal issues and cases of either psoriatic or rheumatoid arthritis in young patients.
The cardiovascular risk factors associated with metabolic syndrome are greater in children with psoriasis, compared with children without psoriasis, the work group noted. In addition, pediatric psoriasis patients have a higher prevalence of obesity than children without psoriasis, and they recommended that children with psoriasis be monitored for the development of obesity, and that obese children with psoriasis should be referred for weight management.
The work group noted that data are insufficient in children to support the link between psoriasis and cardiovascular disease that has been documented in adults with psoriasis. However, “patients with pediatric psoriasis should have American Academy of Pediatrics (AAP)–recommended age-related cardiovascular screening regardless of the presence of signs or symptoms,” they said.
The guideline also recommends screening for dyslipidemia and hypertension according to AAP guidelines and educating pediatric psoriasis patients about the risk of diabetes and regularly screening for diabetes and insulin resistance in those who are obese. Overweight children with psoriasis may be screened at the provider’s discretion, they wrote. Patients with signs of inflammatory bowel disease, which also is associated with psoriasis in adults, should be considered for referral to a gastroenterologist, they noted.
Children with psoriasis should be screened regularly for mental health conditions regardless of age, and they should be asked about substance abuse, according to the guideline, and those with concerns should be referred for additional assessment and management.
The guideline divides treatment of psoriasis in children into three categories: topical, phototherapy and photochemotherapy, and systemic treatments (nonbiologic or biologic).
For topicals, the guideline recommendations include corticosteroids as an off-label therapy, as well as ultra-high-potency topical corticosteroids as monotherapy. Overall, “selection of a therapeutic routine (potency, delivery vehicle, frequency of application) should take into account sites of involvement, type and thickness of psoriasis, age of the patient, total BSA of application, anticipated occlusion, and disease acuity, among other patient-, disease-, and drug-related factors,” the authors wrote. Other topical options included in the recommendations: calcineurin inhibitors, topical vitamin D analogues, tazarotene (off label), anthralin, and coal tar.
Phototherapy has a history of use in psoriasis treatment and remains part of the current recommendations, although data in children are limited, and data on the use of phototherapy for pustular psoriasis in children are insufficient to make specific treatment and dosing recommendations, the work group noted. The researchers also noted that in-office phototherapy may not be feasible for many patients, but that in-home ultraviolet light equipment or natural sunlight in moderation could be recommended as an alternative.
The use of systemic, nonbiologic treatments for pediatric psoriasis should be “based on baseline severity of disease, subtype of psoriasis, speed of disease progression, lack of response to more conservative therapies such as topical agents and phototherapy (when appropriate), impaired physical or psychological functioning or [quality of life] due to disease extent, and the presence of comorbidities such as PsA,” the workgroup said.
Options for systemic treatment include methotrexate, cyclosporine (notably for pustular as well as plaque and erythrodermic psoriasis), and systemic retinoids. In addition, fumaric acid esters may be an option for children with moderate to severe psoriasis, with recommended clinical and laboratory monitoring.
The increasing safety and efficacy data on biologics in pediatric psoriasis patients support their consideration among first-line systemic treatments, the work group suggested. “Etanercept and ustekinumab are now [Food and Drug Administration] approved for patients with psoriasis 4 years and older and 12 years and older, respectively,” they said, and infliximab and adalimumab have been used off label in children.
The work group concluded that research and knowledge gaps about pediatric psoriasis persist and include mechanism of disease onset, development of comorbidities, and identification of ideal dosing for various treatments.
Finally, the work group emphasized the importance of collaboration between dermatologists and primary care providers for managing psoriasis in children, as well as the importance of patient education.
“Dermatologists should be mindful of the unique aspects of the emotional development of children and the social dynamics of having a visible difference,” they wrote. “Shared decision making with the patient (if age appropriate) and the caregivers is a useful approach, particularly as related to the use of off-label medications to treat severe disease,” they said.
“This is the first time that pediatric psoriasis has been discussed as an independent topic within the guideline,” said one of the guideline authors, Dawn M.R. Davis, MD, of the Mayo Clinic, Rochester, Minn., in an interview. “Children have unique physiology and psychosocial aspects to their care relative to adults. In addition, psoriasis has some clinical manifestations that are oftentimes distinctly seen in children,” she commented. “Creation of a guideline specific to children allows us to summarize the similarities and differences of disease presentation and management. It also allows an opportunity to clarify what research data (especially therapeutics) have been studied in children and their uses, safety profiles, and dosing,” she noted.
Psoriasis can be a psychosocially debilitating disease, she emphasized. “In children, for example, isolated or prominent facial involvement is common, which can be embarrassing and impact relationships.”
The take-home message for clinicians, Dr. Davis said, is to keep in mind the multisystemic nature of psoriasis. “It is not limited to the skin,” she said. “Treating a patient with psoriasis necessitates practicing whole-person care” and considering the multiple comorbidities that impact quality of life and overall health in children, as well as adults with psoriasis, she commented. “Dermatologists can empower patients and their caregivers by educating them on the multifocal, complex nature of the disease.” She added, “We have much to learn regarding psoriasis in the pediatric population. More research into therapeutics, topical and systemic, is necessary to optimize patient care.”
The guideline was based on studies published in the PubMed and MEDLINE databases from January 2011 through December 31, 2017.
Dr. Menter and Craig A. Elmets, MD, professor of dermatology, at the University of Alabama, Birmingham, were cochairs of the work group. The pediatric guideline is the latest in a multipart series of AAD-NPF guidelines on psoriasis being published this year in the Journal of the American Academy of Dermatology.
Many of the guideline authors, including lead author Dr. Menter, disclosed relationships with multiple companies; however, a minimum 51% of workgroup members had no relevant conflicts of interest in accordance with AAD policy. There was no funding source. Dr. Davis disclosed serving as an investigator for Regeneron, with no compensation.
SOURCE: Menter et al. J Am Acad Dermatol. 2019. doi: 10.1016/j.jaad.2019.08.049.
Psoriasis management in children involves attention not only to treatment of the physical condition but also psychosocial wellness and quality of life, according to
Psoriasis affects approximately 1% of children, either alone or associated with comorbid conditions such as psoriatic arthritis (PsA), wrote Alan Menter, MD, of Baylor University Medical Center, Dallas, and coauthors of the guideline.
In the guideline, published in the Journal of the American Academy of Dermatology, the multidisciplinary work group identified screening tools to measure disease severity, strategies for management of comorbidities, and the safety and effectiveness of topical, systemic, and phototherapy treatments.
To assess disease severity, the work group recommended not only the use of body surface area (BSA), similar to measurement of severity in adults, but also the use of the Children’s Dermatology Life Quality Index, a 10-question quality of life survey, as BSA alone does not account for the potential negative impact of the disease on quality of life in terms of physical, emotional, social, and psychological function.
“For example, a child with psoriasis limited to the face or the entire scalp does not have severe disease based on BSA definitions, but if this involvement causes shame, social withdrawal, or bullying, it satisfies criteria for severe disease based on impact beyond the skin,” they said.
The work group stated that a variety of conditions may trigger or exacerbate psoriasis in children, including infections, cutaneous trauma, or physiological, emotional, and environmental stressors.
The majority of children with PsA develop joint inflammation before skin disease, the work group wrote. In addition, children with psoriasis are at increased risk for rheumatoid arthritis, so clinicians may need to distinguish between a combination of psoriasis and musculoskeletal issues and cases of either psoriatic or rheumatoid arthritis in young patients.
The cardiovascular risk factors associated with metabolic syndrome are greater in children with psoriasis, compared with children without psoriasis, the work group noted. In addition, pediatric psoriasis patients have a higher prevalence of obesity than children without psoriasis, and they recommended that children with psoriasis be monitored for the development of obesity, and that obese children with psoriasis should be referred for weight management.
The work group noted that data are insufficient in children to support the link between psoriasis and cardiovascular disease that has been documented in adults with psoriasis. However, “patients with pediatric psoriasis should have American Academy of Pediatrics (AAP)–recommended age-related cardiovascular screening regardless of the presence of signs or symptoms,” they said.
The guideline also recommends screening for dyslipidemia and hypertension according to AAP guidelines and educating pediatric psoriasis patients about the risk of diabetes and regularly screening for diabetes and insulin resistance in those who are obese. Overweight children with psoriasis may be screened at the provider’s discretion, they wrote. Patients with signs of inflammatory bowel disease, which also is associated with psoriasis in adults, should be considered for referral to a gastroenterologist, they noted.
Children with psoriasis should be screened regularly for mental health conditions regardless of age, and they should be asked about substance abuse, according to the guideline, and those with concerns should be referred for additional assessment and management.
The guideline divides treatment of psoriasis in children into three categories: topical, phototherapy and photochemotherapy, and systemic treatments (nonbiologic or biologic).
For topicals, the guideline recommendations include corticosteroids as an off-label therapy, as well as ultra-high-potency topical corticosteroids as monotherapy. Overall, “selection of a therapeutic routine (potency, delivery vehicle, frequency of application) should take into account sites of involvement, type and thickness of psoriasis, age of the patient, total BSA of application, anticipated occlusion, and disease acuity, among other patient-, disease-, and drug-related factors,” the authors wrote. Other topical options included in the recommendations: calcineurin inhibitors, topical vitamin D analogues, tazarotene (off label), anthralin, and coal tar.
Phototherapy has a history of use in psoriasis treatment and remains part of the current recommendations, although data in children are limited, and data on the use of phototherapy for pustular psoriasis in children are insufficient to make specific treatment and dosing recommendations, the work group noted. The researchers also noted that in-office phototherapy may not be feasible for many patients, but that in-home ultraviolet light equipment or natural sunlight in moderation could be recommended as an alternative.
The use of systemic, nonbiologic treatments for pediatric psoriasis should be “based on baseline severity of disease, subtype of psoriasis, speed of disease progression, lack of response to more conservative therapies such as topical agents and phototherapy (when appropriate), impaired physical or psychological functioning or [quality of life] due to disease extent, and the presence of comorbidities such as PsA,” the workgroup said.
Options for systemic treatment include methotrexate, cyclosporine (notably for pustular as well as plaque and erythrodermic psoriasis), and systemic retinoids. In addition, fumaric acid esters may be an option for children with moderate to severe psoriasis, with recommended clinical and laboratory monitoring.
The increasing safety and efficacy data on biologics in pediatric psoriasis patients support their consideration among first-line systemic treatments, the work group suggested. “Etanercept and ustekinumab are now [Food and Drug Administration] approved for patients with psoriasis 4 years and older and 12 years and older, respectively,” they said, and infliximab and adalimumab have been used off label in children.
The work group concluded that research and knowledge gaps about pediatric psoriasis persist and include mechanism of disease onset, development of comorbidities, and identification of ideal dosing for various treatments.
Finally, the work group emphasized the importance of collaboration between dermatologists and primary care providers for managing psoriasis in children, as well as the importance of patient education.
“Dermatologists should be mindful of the unique aspects of the emotional development of children and the social dynamics of having a visible difference,” they wrote. “Shared decision making with the patient (if age appropriate) and the caregivers is a useful approach, particularly as related to the use of off-label medications to treat severe disease,” they said.
“This is the first time that pediatric psoriasis has been discussed as an independent topic within the guideline,” said one of the guideline authors, Dawn M.R. Davis, MD, of the Mayo Clinic, Rochester, Minn., in an interview. “Children have unique physiology and psychosocial aspects to their care relative to adults. In addition, psoriasis has some clinical manifestations that are oftentimes distinctly seen in children,” she commented. “Creation of a guideline specific to children allows us to summarize the similarities and differences of disease presentation and management. It also allows an opportunity to clarify what research data (especially therapeutics) have been studied in children and their uses, safety profiles, and dosing,” she noted.
Psoriasis can be a psychosocially debilitating disease, she emphasized. “In children, for example, isolated or prominent facial involvement is common, which can be embarrassing and impact relationships.”
The take-home message for clinicians, Dr. Davis said, is to keep in mind the multisystemic nature of psoriasis. “It is not limited to the skin,” she said. “Treating a patient with psoriasis necessitates practicing whole-person care” and considering the multiple comorbidities that impact quality of life and overall health in children, as well as adults with psoriasis, she commented. “Dermatologists can empower patients and their caregivers by educating them on the multifocal, complex nature of the disease.” She added, “We have much to learn regarding psoriasis in the pediatric population. More research into therapeutics, topical and systemic, is necessary to optimize patient care.”
The guideline was based on studies published in the PubMed and MEDLINE databases from January 2011 through December 31, 2017.
Dr. Menter and Craig A. Elmets, MD, professor of dermatology, at the University of Alabama, Birmingham, were cochairs of the work group. The pediatric guideline is the latest in a multipart series of AAD-NPF guidelines on psoriasis being published this year in the Journal of the American Academy of Dermatology.
Many of the guideline authors, including lead author Dr. Menter, disclosed relationships with multiple companies; however, a minimum 51% of workgroup members had no relevant conflicts of interest in accordance with AAD policy. There was no funding source. Dr. Davis disclosed serving as an investigator for Regeneron, with no compensation.
SOURCE: Menter et al. J Am Acad Dermatol. 2019. doi: 10.1016/j.jaad.2019.08.049.
Psoriasis management in children involves attention not only to treatment of the physical condition but also psychosocial wellness and quality of life, according to
Psoriasis affects approximately 1% of children, either alone or associated with comorbid conditions such as psoriatic arthritis (PsA), wrote Alan Menter, MD, of Baylor University Medical Center, Dallas, and coauthors of the guideline.
In the guideline, published in the Journal of the American Academy of Dermatology, the multidisciplinary work group identified screening tools to measure disease severity, strategies for management of comorbidities, and the safety and effectiveness of topical, systemic, and phototherapy treatments.
To assess disease severity, the work group recommended not only the use of body surface area (BSA), similar to measurement of severity in adults, but also the use of the Children’s Dermatology Life Quality Index, a 10-question quality of life survey, as BSA alone does not account for the potential negative impact of the disease on quality of life in terms of physical, emotional, social, and psychological function.
“For example, a child with psoriasis limited to the face or the entire scalp does not have severe disease based on BSA definitions, but if this involvement causes shame, social withdrawal, or bullying, it satisfies criteria for severe disease based on impact beyond the skin,” they said.
The work group stated that a variety of conditions may trigger or exacerbate psoriasis in children, including infections, cutaneous trauma, or physiological, emotional, and environmental stressors.
The majority of children with PsA develop joint inflammation before skin disease, the work group wrote. In addition, children with psoriasis are at increased risk for rheumatoid arthritis, so clinicians may need to distinguish between a combination of psoriasis and musculoskeletal issues and cases of either psoriatic or rheumatoid arthritis in young patients.
The cardiovascular risk factors associated with metabolic syndrome are greater in children with psoriasis, compared with children without psoriasis, the work group noted. In addition, pediatric psoriasis patients have a higher prevalence of obesity than children without psoriasis, and they recommended that children with psoriasis be monitored for the development of obesity, and that obese children with psoriasis should be referred for weight management.
The work group noted that data are insufficient in children to support the link between psoriasis and cardiovascular disease that has been documented in adults with psoriasis. However, “patients with pediatric psoriasis should have American Academy of Pediatrics (AAP)–recommended age-related cardiovascular screening regardless of the presence of signs or symptoms,” they said.
The guideline also recommends screening for dyslipidemia and hypertension according to AAP guidelines and educating pediatric psoriasis patients about the risk of diabetes and regularly screening for diabetes and insulin resistance in those who are obese. Overweight children with psoriasis may be screened at the provider’s discretion, they wrote. Patients with signs of inflammatory bowel disease, which also is associated with psoriasis in adults, should be considered for referral to a gastroenterologist, they noted.
Children with psoriasis should be screened regularly for mental health conditions regardless of age, and they should be asked about substance abuse, according to the guideline, and those with concerns should be referred for additional assessment and management.
The guideline divides treatment of psoriasis in children into three categories: topical, phototherapy and photochemotherapy, and systemic treatments (nonbiologic or biologic).
For topicals, the guideline recommendations include corticosteroids as an off-label therapy, as well as ultra-high-potency topical corticosteroids as monotherapy. Overall, “selection of a therapeutic routine (potency, delivery vehicle, frequency of application) should take into account sites of involvement, type and thickness of psoriasis, age of the patient, total BSA of application, anticipated occlusion, and disease acuity, among other patient-, disease-, and drug-related factors,” the authors wrote. Other topical options included in the recommendations: calcineurin inhibitors, topical vitamin D analogues, tazarotene (off label), anthralin, and coal tar.
Phototherapy has a history of use in psoriasis treatment and remains part of the current recommendations, although data in children are limited, and data on the use of phototherapy for pustular psoriasis in children are insufficient to make specific treatment and dosing recommendations, the work group noted. The researchers also noted that in-office phototherapy may not be feasible for many patients, but that in-home ultraviolet light equipment or natural sunlight in moderation could be recommended as an alternative.
The use of systemic, nonbiologic treatments for pediatric psoriasis should be “based on baseline severity of disease, subtype of psoriasis, speed of disease progression, lack of response to more conservative therapies such as topical agents and phototherapy (when appropriate), impaired physical or psychological functioning or [quality of life] due to disease extent, and the presence of comorbidities such as PsA,” the workgroup said.
Options for systemic treatment include methotrexate, cyclosporine (notably for pustular as well as plaque and erythrodermic psoriasis), and systemic retinoids. In addition, fumaric acid esters may be an option for children with moderate to severe psoriasis, with recommended clinical and laboratory monitoring.
The increasing safety and efficacy data on biologics in pediatric psoriasis patients support their consideration among first-line systemic treatments, the work group suggested. “Etanercept and ustekinumab are now [Food and Drug Administration] approved for patients with psoriasis 4 years and older and 12 years and older, respectively,” they said, and infliximab and adalimumab have been used off label in children.
The work group concluded that research and knowledge gaps about pediatric psoriasis persist and include mechanism of disease onset, development of comorbidities, and identification of ideal dosing for various treatments.
Finally, the work group emphasized the importance of collaboration between dermatologists and primary care providers for managing psoriasis in children, as well as the importance of patient education.
“Dermatologists should be mindful of the unique aspects of the emotional development of children and the social dynamics of having a visible difference,” they wrote. “Shared decision making with the patient (if age appropriate) and the caregivers is a useful approach, particularly as related to the use of off-label medications to treat severe disease,” they said.
“This is the first time that pediatric psoriasis has been discussed as an independent topic within the guideline,” said one of the guideline authors, Dawn M.R. Davis, MD, of the Mayo Clinic, Rochester, Minn., in an interview. “Children have unique physiology and psychosocial aspects to their care relative to adults. In addition, psoriasis has some clinical manifestations that are oftentimes distinctly seen in children,” she commented. “Creation of a guideline specific to children allows us to summarize the similarities and differences of disease presentation and management. It also allows an opportunity to clarify what research data (especially therapeutics) have been studied in children and their uses, safety profiles, and dosing,” she noted.
Psoriasis can be a psychosocially debilitating disease, she emphasized. “In children, for example, isolated or prominent facial involvement is common, which can be embarrassing and impact relationships.”
The take-home message for clinicians, Dr. Davis said, is to keep in mind the multisystemic nature of psoriasis. “It is not limited to the skin,” she said. “Treating a patient with psoriasis necessitates practicing whole-person care” and considering the multiple comorbidities that impact quality of life and overall health in children, as well as adults with psoriasis, she commented. “Dermatologists can empower patients and their caregivers by educating them on the multifocal, complex nature of the disease.” She added, “We have much to learn regarding psoriasis in the pediatric population. More research into therapeutics, topical and systemic, is necessary to optimize patient care.”
The guideline was based on studies published in the PubMed and MEDLINE databases from January 2011 through December 31, 2017.
Dr. Menter and Craig A. Elmets, MD, professor of dermatology, at the University of Alabama, Birmingham, were cochairs of the work group. The pediatric guideline is the latest in a multipart series of AAD-NPF guidelines on psoriasis being published this year in the Journal of the American Academy of Dermatology.
Many of the guideline authors, including lead author Dr. Menter, disclosed relationships with multiple companies; however, a minimum 51% of workgroup members had no relevant conflicts of interest in accordance with AAD policy. There was no funding source. Dr. Davis disclosed serving as an investigator for Regeneron, with no compensation.
SOURCE: Menter et al. J Am Acad Dermatol. 2019. doi: 10.1016/j.jaad.2019.08.049.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
Measles infection linked to impaired ‘immune memory’
Infection with the measles virus appears to reduce immunity to other pathogens, according to a paper published in Science.
The hypothesis that the measles virus could cause “immunological amnesia” by impairing immune memory is supported by early research showing children with measles had negative cutaneous tuberculin reactions after having previously tested positive.
“Subsequent studies have shown decreased interferon signaling, skewed cytokine responses, lymphopenia, and suppression of lymphocyte proliferation shortly after infection,” wrote Michael Mina, MD, from Brigham and Women’s Hospital in Boston, and coauthors.
“Given the variation in the degree of immune repertoire modulation we observed, we anticipate that future risk of morbidity and mortality after measles would not be homogeneous but would be skewed toward individuals with the most severe elimination of immunological memory,” they wrote. “These findings underscore the crucial need for continued widespread vaccination.”
In this study, researchers compared the levels of around 400 pathogen-specific antibodies in blood samples from 77 unvaccinated children, taken before and 2 months after natural measles infection, with 5 unvaccinated children who did not contract measles. A total of 34 the children experienced mild measles, and 43 had severe measles.
They found that the samples taken after measles infection showed “substantial” reductions in the number of pathogen epitopes, compared with the samples from children who did not get infected with measles.
This amounted to approximately a 20% mean reduction in overall diversity or size of the antibody repertoire. However, in children who experienced severe measles, there was a median loss of 40% (range, 11%-62%) of antibody repertoire, compared with a median of 33% (range, 12%-73%) range in children who experienced mild infection. Meanwhile, the control subjects retained approximately 90% of their antibody repertoire over a similar or longer time period. Some children lost up to 70% of antibodies for specific pathogens.
The study did find increases in measles virus–specific antigens in children both after measles infection and MMR vaccination. However the authors did not detect any changes in total IgG, IgA, or IgM levels.
Dr. Mina and associates wrote.
They also noted that controls who received the MMR vaccine showed a marked increase in overall antibody repertoire.
In a separate investigation reported in Science Immunology, Velislava N. Petrova, PhD, of the Wellcome Sanger Institute in Cambridge, England, and coauthors investigated genetic changes in 26 unvaccinated children from the Netherlands who previously had measles to determine if B-cell impairment can lead to measles-associated immunosuppression. Their antibody genes were sequenced before any symptoms of measles developed and roughly 40 days after rash. Two control groups also were sequenced accordingly: vaccinated adults and three unvaccinated children from the same community who were not infected with measles.
Naive B cells from individuals in the vaccinated and uninfected control groups showed high correlation of immunoglobulin heavy chain (IgVH-J) gene frequencies across time periods (R2 = 0.96 and 0.92, respectively) but no significant differences in gene expression (P greater than .05). At the same time, although B-cell frequencies in measles patients recovered to levels before infection, they had significant changes in IgVH-J gene frequencies (P = .01) and decreased correlation in gene expression (R2 = 0.78).
In addition, individuals in the control groups had “a stable genetic composition of B memory cells” but no significant changes in the third complementarity-determining region (CDR3) lengths or mutational frequency of IgVH-J genes (P greater than .05). B memory cells in measles patients, however, showed increases in mutational frequency (P = .0008) and a reduction in CDR3 length (P = .017) of IgVH genes, Dr. Petrova and associates reported.
The study by Mina et al. was supported by grants from various U.S., European, and Finnish foundations and national organizations. Some of the coauthors had relationships with biotechnology and pharmaceutical companies, and three reported a patent holding related to technology used in the study. The study by Petrova et al. was funded by grants to the investigators from various Indonesian and German organizations and the Wellcome Trust. The authors reported no conflicts of interest.
SOURCES: Mina M et al. Science. 2019 Nov 1;366:599-606; Petrova VN et al. Sci Immunol. 2019 Nov 1. doi: 10.1126/sciimmunol.aay6125.
As a result of reduced vaccination, after decades of decline, the number of worldwide cases of measles has increased by nearly 300% since 2018. Epidemiologic evidence has associated measles infections with increases in morbidity and mortality for as long as 5 years after the infection and suggests that, in the prevaccine era, measles virus may have been associated with up to 50% of all childhood deaths, mostly because of nonmeasles infections. Measles replication in immune cells has been hypothesized to impair immune memory, potentially causing what some scientists call “immunological amnesia.”A measles virus receptor, called CD150/ SLAMF1, is highly expressed on memory T, B, and plasma cells. Measles virus gains entry to these immune memory cells using that receptor and kills the cells.
The scientists stated that it could take months or years to return the immune repertoire back to baseline. During the rebuilding process, children would be at increased risk for infectious diseases they had previously experienced.
In a second outstanding paper, Petrova et al. in Science Immunology studied B cells before and after measles infection, and identified two immunologic consequences: The naive B-cell pool was depleted, leading to a return to immunologic immaturity, and the memory B-cell pool was depleted, resulting in compromised immune memory to previously encountered pathogens.
Thus, the link between measles infections and increased susceptibility to other infections and increased deaths from nonmeasles infectious diseases in the aftermath of measles has been revealed. This information adds new data to share with parents who consider refusing measles vaccination. The risks are far greater than getting measles.
Michael E. Pichichero, MD, is a specialist in pediatric infectious diseases and director of the Research Institute at Rochester (N.Y.) General Hospital. He was asked to comment on the articles. Dr. Pichichero has no conflicts to declare.
As a result of reduced vaccination, after decades of decline, the number of worldwide cases of measles has increased by nearly 300% since 2018. Epidemiologic evidence has associated measles infections with increases in morbidity and mortality for as long as 5 years after the infection and suggests that, in the prevaccine era, measles virus may have been associated with up to 50% of all childhood deaths, mostly because of nonmeasles infections. Measles replication in immune cells has been hypothesized to impair immune memory, potentially causing what some scientists call “immunological amnesia.”A measles virus receptor, called CD150/ SLAMF1, is highly expressed on memory T, B, and plasma cells. Measles virus gains entry to these immune memory cells using that receptor and kills the cells.
The scientists stated that it could take months or years to return the immune repertoire back to baseline. During the rebuilding process, children would be at increased risk for infectious diseases they had previously experienced.
In a second outstanding paper, Petrova et al. in Science Immunology studied B cells before and after measles infection, and identified two immunologic consequences: The naive B-cell pool was depleted, leading to a return to immunologic immaturity, and the memory B-cell pool was depleted, resulting in compromised immune memory to previously encountered pathogens.
Thus, the link between measles infections and increased susceptibility to other infections and increased deaths from nonmeasles infectious diseases in the aftermath of measles has been revealed. This information adds new data to share with parents who consider refusing measles vaccination. The risks are far greater than getting measles.
Michael E. Pichichero, MD, is a specialist in pediatric infectious diseases and director of the Research Institute at Rochester (N.Y.) General Hospital. He was asked to comment on the articles. Dr. Pichichero has no conflicts to declare.
As a result of reduced vaccination, after decades of decline, the number of worldwide cases of measles has increased by nearly 300% since 2018. Epidemiologic evidence has associated measles infections with increases in morbidity and mortality for as long as 5 years after the infection and suggests that, in the prevaccine era, measles virus may have been associated with up to 50% of all childhood deaths, mostly because of nonmeasles infections. Measles replication in immune cells has been hypothesized to impair immune memory, potentially causing what some scientists call “immunological amnesia.”A measles virus receptor, called CD150/ SLAMF1, is highly expressed on memory T, B, and plasma cells. Measles virus gains entry to these immune memory cells using that receptor and kills the cells.
The scientists stated that it could take months or years to return the immune repertoire back to baseline. During the rebuilding process, children would be at increased risk for infectious diseases they had previously experienced.
In a second outstanding paper, Petrova et al. in Science Immunology studied B cells before and after measles infection, and identified two immunologic consequences: The naive B-cell pool was depleted, leading to a return to immunologic immaturity, and the memory B-cell pool was depleted, resulting in compromised immune memory to previously encountered pathogens.
Thus, the link between measles infections and increased susceptibility to other infections and increased deaths from nonmeasles infectious diseases in the aftermath of measles has been revealed. This information adds new data to share with parents who consider refusing measles vaccination. The risks are far greater than getting measles.
Michael E. Pichichero, MD, is a specialist in pediatric infectious diseases and director of the Research Institute at Rochester (N.Y.) General Hospital. He was asked to comment on the articles. Dr. Pichichero has no conflicts to declare.
Infection with the measles virus appears to reduce immunity to other pathogens, according to a paper published in Science.
The hypothesis that the measles virus could cause “immunological amnesia” by impairing immune memory is supported by early research showing children with measles had negative cutaneous tuberculin reactions after having previously tested positive.
“Subsequent studies have shown decreased interferon signaling, skewed cytokine responses, lymphopenia, and suppression of lymphocyte proliferation shortly after infection,” wrote Michael Mina, MD, from Brigham and Women’s Hospital in Boston, and coauthors.
“Given the variation in the degree of immune repertoire modulation we observed, we anticipate that future risk of morbidity and mortality after measles would not be homogeneous but would be skewed toward individuals with the most severe elimination of immunological memory,” they wrote. “These findings underscore the crucial need for continued widespread vaccination.”
In this study, researchers compared the levels of around 400 pathogen-specific antibodies in blood samples from 77 unvaccinated children, taken before and 2 months after natural measles infection, with 5 unvaccinated children who did not contract measles. A total of 34 the children experienced mild measles, and 43 had severe measles.
They found that the samples taken after measles infection showed “substantial” reductions in the number of pathogen epitopes, compared with the samples from children who did not get infected with measles.
This amounted to approximately a 20% mean reduction in overall diversity or size of the antibody repertoire. However, in children who experienced severe measles, there was a median loss of 40% (range, 11%-62%) of antibody repertoire, compared with a median of 33% (range, 12%-73%) range in children who experienced mild infection. Meanwhile, the control subjects retained approximately 90% of their antibody repertoire over a similar or longer time period. Some children lost up to 70% of antibodies for specific pathogens.
The study did find increases in measles virus–specific antigens in children both after measles infection and MMR vaccination. However the authors did not detect any changes in total IgG, IgA, or IgM levels.
Dr. Mina and associates wrote.
They also noted that controls who received the MMR vaccine showed a marked increase in overall antibody repertoire.
In a separate investigation reported in Science Immunology, Velislava N. Petrova, PhD, of the Wellcome Sanger Institute in Cambridge, England, and coauthors investigated genetic changes in 26 unvaccinated children from the Netherlands who previously had measles to determine if B-cell impairment can lead to measles-associated immunosuppression. Their antibody genes were sequenced before any symptoms of measles developed and roughly 40 days after rash. Two control groups also were sequenced accordingly: vaccinated adults and three unvaccinated children from the same community who were not infected with measles.
Naive B cells from individuals in the vaccinated and uninfected control groups showed high correlation of immunoglobulin heavy chain (IgVH-J) gene frequencies across time periods (R2 = 0.96 and 0.92, respectively) but no significant differences in gene expression (P greater than .05). At the same time, although B-cell frequencies in measles patients recovered to levels before infection, they had significant changes in IgVH-J gene frequencies (P = .01) and decreased correlation in gene expression (R2 = 0.78).
In addition, individuals in the control groups had “a stable genetic composition of B memory cells” but no significant changes in the third complementarity-determining region (CDR3) lengths or mutational frequency of IgVH-J genes (P greater than .05). B memory cells in measles patients, however, showed increases in mutational frequency (P = .0008) and a reduction in CDR3 length (P = .017) of IgVH genes, Dr. Petrova and associates reported.
The study by Mina et al. was supported by grants from various U.S., European, and Finnish foundations and national organizations. Some of the coauthors had relationships with biotechnology and pharmaceutical companies, and three reported a patent holding related to technology used in the study. The study by Petrova et al. was funded by grants to the investigators from various Indonesian and German organizations and the Wellcome Trust. The authors reported no conflicts of interest.
SOURCES: Mina M et al. Science. 2019 Nov 1;366:599-606; Petrova VN et al. Sci Immunol. 2019 Nov 1. doi: 10.1126/sciimmunol.aay6125.
Infection with the measles virus appears to reduce immunity to other pathogens, according to a paper published in Science.
The hypothesis that the measles virus could cause “immunological amnesia” by impairing immune memory is supported by early research showing children with measles had negative cutaneous tuberculin reactions after having previously tested positive.
“Subsequent studies have shown decreased interferon signaling, skewed cytokine responses, lymphopenia, and suppression of lymphocyte proliferation shortly after infection,” wrote Michael Mina, MD, from Brigham and Women’s Hospital in Boston, and coauthors.
“Given the variation in the degree of immune repertoire modulation we observed, we anticipate that future risk of morbidity and mortality after measles would not be homogeneous but would be skewed toward individuals with the most severe elimination of immunological memory,” they wrote. “These findings underscore the crucial need for continued widespread vaccination.”
In this study, researchers compared the levels of around 400 pathogen-specific antibodies in blood samples from 77 unvaccinated children, taken before and 2 months after natural measles infection, with 5 unvaccinated children who did not contract measles. A total of 34 the children experienced mild measles, and 43 had severe measles.
They found that the samples taken after measles infection showed “substantial” reductions in the number of pathogen epitopes, compared with the samples from children who did not get infected with measles.
This amounted to approximately a 20% mean reduction in overall diversity or size of the antibody repertoire. However, in children who experienced severe measles, there was a median loss of 40% (range, 11%-62%) of antibody repertoire, compared with a median of 33% (range, 12%-73%) range in children who experienced mild infection. Meanwhile, the control subjects retained approximately 90% of their antibody repertoire over a similar or longer time period. Some children lost up to 70% of antibodies for specific pathogens.
The study did find increases in measles virus–specific antigens in children both after measles infection and MMR vaccination. However the authors did not detect any changes in total IgG, IgA, or IgM levels.
Dr. Mina and associates wrote.
They also noted that controls who received the MMR vaccine showed a marked increase in overall antibody repertoire.
In a separate investigation reported in Science Immunology, Velislava N. Petrova, PhD, of the Wellcome Sanger Institute in Cambridge, England, and coauthors investigated genetic changes in 26 unvaccinated children from the Netherlands who previously had measles to determine if B-cell impairment can lead to measles-associated immunosuppression. Their antibody genes were sequenced before any symptoms of measles developed and roughly 40 days after rash. Two control groups also were sequenced accordingly: vaccinated adults and three unvaccinated children from the same community who were not infected with measles.
Naive B cells from individuals in the vaccinated and uninfected control groups showed high correlation of immunoglobulin heavy chain (IgVH-J) gene frequencies across time periods (R2 = 0.96 and 0.92, respectively) but no significant differences in gene expression (P greater than .05). At the same time, although B-cell frequencies in measles patients recovered to levels before infection, they had significant changes in IgVH-J gene frequencies (P = .01) and decreased correlation in gene expression (R2 = 0.78).
In addition, individuals in the control groups had “a stable genetic composition of B memory cells” but no significant changes in the third complementarity-determining region (CDR3) lengths or mutational frequency of IgVH-J genes (P greater than .05). B memory cells in measles patients, however, showed increases in mutational frequency (P = .0008) and a reduction in CDR3 length (P = .017) of IgVH genes, Dr. Petrova and associates reported.
The study by Mina et al. was supported by grants from various U.S., European, and Finnish foundations and national organizations. Some of the coauthors had relationships with biotechnology and pharmaceutical companies, and three reported a patent holding related to technology used in the study. The study by Petrova et al. was funded by grants to the investigators from various Indonesian and German organizations and the Wellcome Trust. The authors reported no conflicts of interest.
SOURCES: Mina M et al. Science. 2019 Nov 1;366:599-606; Petrova VN et al. Sci Immunol. 2019 Nov 1. doi: 10.1126/sciimmunol.aay6125.
FROM SCIENCE
Adverse childhood experiences increase the risk of poor long-term health
and societal outcomes, according to a new report by the Centers for Disease Control and Prevention.
“Our analysis suggests that preventing or reducing these adverse childhood experiences [ACEs] could potentially reduce the annual number of coronary heart disease cases by up to 13%,” said Ann Schuchat, MD, the CDC’s principal deputy director. “If we apply this analysis to other national disease estimates, preventing ACEs could prevent 1.9 million cases of heart disease, 2.5 million cases of overweight or obesity, 21 million cases of depression, and 1.5 million high-school incompletions.”
The analysis, conducted by Melissa T. Merrick, PhD, and colleagues at the National Center for Injury Prevention and Control at the CDC, Atlanta, is based on data acquired from more than 144,000 adults in 27 states.
It’s the first time the CDC has waded into this territory, Dr. Schuchat said during a press briefing. But a hard look into the data is long overdue. ACEs have been linked to at least 5 of the top 10 leading causes of death in the United States: heart disease, cancer, respiratory disease, diabetes, and suicide.
“It’s been proven that exposure to abuse, violence, and familial substance abuse and mental health problems can lead to health and social problems during the entire lifespan. Multiple exposures can produce toxic stress and chronic activation of the stress response system,” Dr. Schuchat continued. “Our report found that more than half of adults have experienced at least one type of ACE, and one in six adults has been exposed to four or more. The effects add up – the more types of ACE encountered, the higher the risk for negative outcomes that limit their entire lives.”
Dr. Merrick, a behavioral scientist with the CDC, and her team reviewed data collected from the Behavioral Risk Factor Surveillance System (BRFSS), a telephone survey of noninstitutionalized adults administered every year within each state. During the 2015-2017 data collection years, 27 states included questions about ACEs. The experiences included childhood exposure to three types of abuse (physical, emotional, and sexual) and five types of household challenges (household member substance misuse, incarceration, mental illness, parental divorce, or witnessing intimate partner violence) before age 18 years.
In all, 61% of respondents reported experiencing at least one of the events; 16% reported experiencing four or more. Women, Native Americans, Native Alaskans, and blacks were more likely to have these experiences than were men and whites.
A multivariate regression analysis found that adults with the highest level of ACE exposure had significantly elevated risks of several chronic health issues and social challenges, compared with nonexposed subjects. These included increased risk of overweight or obesity (adjusted odds ratio, 1.2), chronic obstructive pulmonary disease (aOR, 2.8), depression (aOR 5.3), smoking (aOR 3.1), heavy drinking (aOR 1.8), and underemployment (aOR 1.7), compared with adults reporting no ACEs.
Reducing ACE exposures could in turn reduce many of these challenges, especially among people with the highest number of exposures. Among this group, preventing all ACE exposure could cut overweight and obesity by up to 1.7%, chronic obstructive pulmonary disease by up to 27%, depression by up to 44%, smoking by up to 33%, and heavy drinking by 24%. Preventing ACE exposure also could reduce lack of health insurance by 4% and unemployment by 15%, the researchers said.
The good news, Dr. Merrick and associates said, is that ACE exposure can be at least partially offset by positive interactions with adults and in social and community settings.
“Prevention of adverse childhood experiences is possible with state and community efforts to build resilient families and communities, provide parental support to develop positive parenting and coping skills, and increase access to, and use of, comprehensive health services,” they said.
The CDC recommends a comprehensive approach to preventing ACEs and mitigating their impact. The data-driven suggestions include:
- Promoting family economic health, including tax credits and family-focused work policy.
- Endorsing programs to mitigate violence and adversity, including public education programs that support parents.
- Promoting early childhood development with high-quality child care and preschool programs.
- Recommending stress reduction skills for parents and young people, and programs that teach safe dating and healthy relationship skills.
- Supporting youth development by connecting youth to adult mentors and after-school programs.
- Encouraging clinicians to identify and address ACE exposure with screening, referral, and support.
“This is important for reducing the consequences of adverse childhood experiences and for helping to protect the next generation of children from exposure to violence and other adverse experiences, such as witnessing substance misuse in their household,” Dr. Merrick and associates said.
The researchers had no relevant financial disclosures.
SOURCE: Merrick M et al. MMWR. 2019 Nov 5. doi: 10.15585/mmwr.mm6844e1.
Adverse childhood experiences (ACEs) trigger pathophysiologic responses that exert real physical and psychological harm. Thus, clinicians can and should address them as part of good medical care, Christopher M. Jones, PharmD, Melissa T. Merrick, PhD, and Debra E. Houry, MD, MPH, said in a JAMA commentary.
“A large and growing body of research indicates that the underlying mechanism by which ACEs are associated with health outcomes is through the development of toxic stress, a chronic activation of the stress response system. Toxic stress results in dysregulation of the limbic-hypothalamic-pituitary-adrenal axis, elevating levels of catecholamines (“fight or flight” response), cortisol, and proinflammatory cytokines, leading to cascading effects on the nervous, endocrine, and immune systems. These changes can affect attention and other executive functioning, impulsive behavior, brain reward systems, decision-making, and response to stress throughout the life span,” they said.
While societies and communities at large must work together to reduce ACE exposure, clinicians also have a role. Research indicates that many don’t routinely ask questions about these issues, in a large part because they lack training in how and when to screen.
“Incorporating components of primary ACEs prevention into everyday clinical practice may be achievable through talking with parents and caregivers about creating safe, stable, nurturing environments and protective relationships, and reinforcing positive parenting techniques and coping skills at routine clinical visits,” the editorialists said. “In addition, clinicians can refer parents to parenting skills classes or refer higher-risk parents to home visitation programs such as Healthy Families America and Nurse-Family Partnership. Home visitation programs have demonstrated significant reductions in rates of child abuse and neglect and have improved substance use, violence, and parenting outcomes.”
Clinicians also may have a role to play in mitigating the harms of ACEs, by incorporating trauma-informed care and services into their daily practice.
“Important elements of trauma-informed care include understanding how trauma affects health, routinely screening for ACEs and trauma, using culturally responsive assessments, promoting resilience and protective factors, addressing trauma-related somatic and mental health issues, and ensuring appropriate linkage to services and supports for identified issues,” the editorialists concluded.
Dr. Jones is associate director in the Office of Strategy and Innovation in the CDC Injury Center. Dr. Merrick is president and CEO of Prevent Childhood Abuse America, Chicago. Dr. Houry is director of the National Center for Injury Prevention and Control at the CDC, Atlanta. They discussed the MMWR analysis in a commentary (JAMA. 2019 Nov 5. doi: 10.1001/jama.2019.18499). They had no relevant financial disclosures.
Adverse childhood experiences (ACEs) trigger pathophysiologic responses that exert real physical and psychological harm. Thus, clinicians can and should address them as part of good medical care, Christopher M. Jones, PharmD, Melissa T. Merrick, PhD, and Debra E. Houry, MD, MPH, said in a JAMA commentary.
“A large and growing body of research indicates that the underlying mechanism by which ACEs are associated with health outcomes is through the development of toxic stress, a chronic activation of the stress response system. Toxic stress results in dysregulation of the limbic-hypothalamic-pituitary-adrenal axis, elevating levels of catecholamines (“fight or flight” response), cortisol, and proinflammatory cytokines, leading to cascading effects on the nervous, endocrine, and immune systems. These changes can affect attention and other executive functioning, impulsive behavior, brain reward systems, decision-making, and response to stress throughout the life span,” they said.
While societies and communities at large must work together to reduce ACE exposure, clinicians also have a role. Research indicates that many don’t routinely ask questions about these issues, in a large part because they lack training in how and when to screen.
“Incorporating components of primary ACEs prevention into everyday clinical practice may be achievable through talking with parents and caregivers about creating safe, stable, nurturing environments and protective relationships, and reinforcing positive parenting techniques and coping skills at routine clinical visits,” the editorialists said. “In addition, clinicians can refer parents to parenting skills classes or refer higher-risk parents to home visitation programs such as Healthy Families America and Nurse-Family Partnership. Home visitation programs have demonstrated significant reductions in rates of child abuse and neglect and have improved substance use, violence, and parenting outcomes.”
Clinicians also may have a role to play in mitigating the harms of ACEs, by incorporating trauma-informed care and services into their daily practice.
“Important elements of trauma-informed care include understanding how trauma affects health, routinely screening for ACEs and trauma, using culturally responsive assessments, promoting resilience and protective factors, addressing trauma-related somatic and mental health issues, and ensuring appropriate linkage to services and supports for identified issues,” the editorialists concluded.
Dr. Jones is associate director in the Office of Strategy and Innovation in the CDC Injury Center. Dr. Merrick is president and CEO of Prevent Childhood Abuse America, Chicago. Dr. Houry is director of the National Center for Injury Prevention and Control at the CDC, Atlanta. They discussed the MMWR analysis in a commentary (JAMA. 2019 Nov 5. doi: 10.1001/jama.2019.18499). They had no relevant financial disclosures.
Adverse childhood experiences (ACEs) trigger pathophysiologic responses that exert real physical and psychological harm. Thus, clinicians can and should address them as part of good medical care, Christopher M. Jones, PharmD, Melissa T. Merrick, PhD, and Debra E. Houry, MD, MPH, said in a JAMA commentary.
“A large and growing body of research indicates that the underlying mechanism by which ACEs are associated with health outcomes is through the development of toxic stress, a chronic activation of the stress response system. Toxic stress results in dysregulation of the limbic-hypothalamic-pituitary-adrenal axis, elevating levels of catecholamines (“fight or flight” response), cortisol, and proinflammatory cytokines, leading to cascading effects on the nervous, endocrine, and immune systems. These changes can affect attention and other executive functioning, impulsive behavior, brain reward systems, decision-making, and response to stress throughout the life span,” they said.
While societies and communities at large must work together to reduce ACE exposure, clinicians also have a role. Research indicates that many don’t routinely ask questions about these issues, in a large part because they lack training in how and when to screen.
“Incorporating components of primary ACEs prevention into everyday clinical practice may be achievable through talking with parents and caregivers about creating safe, stable, nurturing environments and protective relationships, and reinforcing positive parenting techniques and coping skills at routine clinical visits,” the editorialists said. “In addition, clinicians can refer parents to parenting skills classes or refer higher-risk parents to home visitation programs such as Healthy Families America and Nurse-Family Partnership. Home visitation programs have demonstrated significant reductions in rates of child abuse and neglect and have improved substance use, violence, and parenting outcomes.”
Clinicians also may have a role to play in mitigating the harms of ACEs, by incorporating trauma-informed care and services into their daily practice.
“Important elements of trauma-informed care include understanding how trauma affects health, routinely screening for ACEs and trauma, using culturally responsive assessments, promoting resilience and protective factors, addressing trauma-related somatic and mental health issues, and ensuring appropriate linkage to services and supports for identified issues,” the editorialists concluded.
Dr. Jones is associate director in the Office of Strategy and Innovation in the CDC Injury Center. Dr. Merrick is president and CEO of Prevent Childhood Abuse America, Chicago. Dr. Houry is director of the National Center for Injury Prevention and Control at the CDC, Atlanta. They discussed the MMWR analysis in a commentary (JAMA. 2019 Nov 5. doi: 10.1001/jama.2019.18499). They had no relevant financial disclosures.
and societal outcomes, according to a new report by the Centers for Disease Control and Prevention.
“Our analysis suggests that preventing or reducing these adverse childhood experiences [ACEs] could potentially reduce the annual number of coronary heart disease cases by up to 13%,” said Ann Schuchat, MD, the CDC’s principal deputy director. “If we apply this analysis to other national disease estimates, preventing ACEs could prevent 1.9 million cases of heart disease, 2.5 million cases of overweight or obesity, 21 million cases of depression, and 1.5 million high-school incompletions.”
The analysis, conducted by Melissa T. Merrick, PhD, and colleagues at the National Center for Injury Prevention and Control at the CDC, Atlanta, is based on data acquired from more than 144,000 adults in 27 states.
It’s the first time the CDC has waded into this territory, Dr. Schuchat said during a press briefing. But a hard look into the data is long overdue. ACEs have been linked to at least 5 of the top 10 leading causes of death in the United States: heart disease, cancer, respiratory disease, diabetes, and suicide.
“It’s been proven that exposure to abuse, violence, and familial substance abuse and mental health problems can lead to health and social problems during the entire lifespan. Multiple exposures can produce toxic stress and chronic activation of the stress response system,” Dr. Schuchat continued. “Our report found that more than half of adults have experienced at least one type of ACE, and one in six adults has been exposed to four or more. The effects add up – the more types of ACE encountered, the higher the risk for negative outcomes that limit their entire lives.”
Dr. Merrick, a behavioral scientist with the CDC, and her team reviewed data collected from the Behavioral Risk Factor Surveillance System (BRFSS), a telephone survey of noninstitutionalized adults administered every year within each state. During the 2015-2017 data collection years, 27 states included questions about ACEs. The experiences included childhood exposure to three types of abuse (physical, emotional, and sexual) and five types of household challenges (household member substance misuse, incarceration, mental illness, parental divorce, or witnessing intimate partner violence) before age 18 years.
In all, 61% of respondents reported experiencing at least one of the events; 16% reported experiencing four or more. Women, Native Americans, Native Alaskans, and blacks were more likely to have these experiences than were men and whites.
A multivariate regression analysis found that adults with the highest level of ACE exposure had significantly elevated risks of several chronic health issues and social challenges, compared with nonexposed subjects. These included increased risk of overweight or obesity (adjusted odds ratio, 1.2), chronic obstructive pulmonary disease (aOR, 2.8), depression (aOR 5.3), smoking (aOR 3.1), heavy drinking (aOR 1.8), and underemployment (aOR 1.7), compared with adults reporting no ACEs.
Reducing ACE exposures could in turn reduce many of these challenges, especially among people with the highest number of exposures. Among this group, preventing all ACE exposure could cut overweight and obesity by up to 1.7%, chronic obstructive pulmonary disease by up to 27%, depression by up to 44%, smoking by up to 33%, and heavy drinking by 24%. Preventing ACE exposure also could reduce lack of health insurance by 4% and unemployment by 15%, the researchers said.
The good news, Dr. Merrick and associates said, is that ACE exposure can be at least partially offset by positive interactions with adults and in social and community settings.
“Prevention of adverse childhood experiences is possible with state and community efforts to build resilient families and communities, provide parental support to develop positive parenting and coping skills, and increase access to, and use of, comprehensive health services,” they said.
The CDC recommends a comprehensive approach to preventing ACEs and mitigating their impact. The data-driven suggestions include:
- Promoting family economic health, including tax credits and family-focused work policy.
- Endorsing programs to mitigate violence and adversity, including public education programs that support parents.
- Promoting early childhood development with high-quality child care and preschool programs.
- Recommending stress reduction skills for parents and young people, and programs that teach safe dating and healthy relationship skills.
- Supporting youth development by connecting youth to adult mentors and after-school programs.
- Encouraging clinicians to identify and address ACE exposure with screening, referral, and support.
“This is important for reducing the consequences of adverse childhood experiences and for helping to protect the next generation of children from exposure to violence and other adverse experiences, such as witnessing substance misuse in their household,” Dr. Merrick and associates said.
The researchers had no relevant financial disclosures.
SOURCE: Merrick M et al. MMWR. 2019 Nov 5. doi: 10.15585/mmwr.mm6844e1.
and societal outcomes, according to a new report by the Centers for Disease Control and Prevention.
“Our analysis suggests that preventing or reducing these adverse childhood experiences [ACEs] could potentially reduce the annual number of coronary heart disease cases by up to 13%,” said Ann Schuchat, MD, the CDC’s principal deputy director. “If we apply this analysis to other national disease estimates, preventing ACEs could prevent 1.9 million cases of heart disease, 2.5 million cases of overweight or obesity, 21 million cases of depression, and 1.5 million high-school incompletions.”
The analysis, conducted by Melissa T. Merrick, PhD, and colleagues at the National Center for Injury Prevention and Control at the CDC, Atlanta, is based on data acquired from more than 144,000 adults in 27 states.
It’s the first time the CDC has waded into this territory, Dr. Schuchat said during a press briefing. But a hard look into the data is long overdue. ACEs have been linked to at least 5 of the top 10 leading causes of death in the United States: heart disease, cancer, respiratory disease, diabetes, and suicide.
“It’s been proven that exposure to abuse, violence, and familial substance abuse and mental health problems can lead to health and social problems during the entire lifespan. Multiple exposures can produce toxic stress and chronic activation of the stress response system,” Dr. Schuchat continued. “Our report found that more than half of adults have experienced at least one type of ACE, and one in six adults has been exposed to four or more. The effects add up – the more types of ACE encountered, the higher the risk for negative outcomes that limit their entire lives.”
Dr. Merrick, a behavioral scientist with the CDC, and her team reviewed data collected from the Behavioral Risk Factor Surveillance System (BRFSS), a telephone survey of noninstitutionalized adults administered every year within each state. During the 2015-2017 data collection years, 27 states included questions about ACEs. The experiences included childhood exposure to three types of abuse (physical, emotional, and sexual) and five types of household challenges (household member substance misuse, incarceration, mental illness, parental divorce, or witnessing intimate partner violence) before age 18 years.
In all, 61% of respondents reported experiencing at least one of the events; 16% reported experiencing four or more. Women, Native Americans, Native Alaskans, and blacks were more likely to have these experiences than were men and whites.
A multivariate regression analysis found that adults with the highest level of ACE exposure had significantly elevated risks of several chronic health issues and social challenges, compared with nonexposed subjects. These included increased risk of overweight or obesity (adjusted odds ratio, 1.2), chronic obstructive pulmonary disease (aOR, 2.8), depression (aOR 5.3), smoking (aOR 3.1), heavy drinking (aOR 1.8), and underemployment (aOR 1.7), compared with adults reporting no ACEs.
Reducing ACE exposures could in turn reduce many of these challenges, especially among people with the highest number of exposures. Among this group, preventing all ACE exposure could cut overweight and obesity by up to 1.7%, chronic obstructive pulmonary disease by up to 27%, depression by up to 44%, smoking by up to 33%, and heavy drinking by 24%. Preventing ACE exposure also could reduce lack of health insurance by 4% and unemployment by 15%, the researchers said.
The good news, Dr. Merrick and associates said, is that ACE exposure can be at least partially offset by positive interactions with adults and in social and community settings.
“Prevention of adverse childhood experiences is possible with state and community efforts to build resilient families and communities, provide parental support to develop positive parenting and coping skills, and increase access to, and use of, comprehensive health services,” they said.
The CDC recommends a comprehensive approach to preventing ACEs and mitigating their impact. The data-driven suggestions include:
- Promoting family economic health, including tax credits and family-focused work policy.
- Endorsing programs to mitigate violence and adversity, including public education programs that support parents.
- Promoting early childhood development with high-quality child care and preschool programs.
- Recommending stress reduction skills for parents and young people, and programs that teach safe dating and healthy relationship skills.
- Supporting youth development by connecting youth to adult mentors and after-school programs.
- Encouraging clinicians to identify and address ACE exposure with screening, referral, and support.
“This is important for reducing the consequences of adverse childhood experiences and for helping to protect the next generation of children from exposure to violence and other adverse experiences, such as witnessing substance misuse in their household,” Dr. Merrick and associates said.
The researchers had no relevant financial disclosures.
SOURCE: Merrick M et al. MMWR. 2019 Nov 5. doi: 10.15585/mmwr.mm6844e1.
FROM MMWR
Ketoacidosis is on the rise in children with type 1 diabetes
BARCELONA – As many as 40%-60% of children have diabetic ketoacidosis (DKA) at the time of being diagnosed with type 1 diabetes, according to data from two U.S. analyses – and the figures have been rising for the past 10 years.
Between 2010 and 2017, the prevalence of DKA at diagnosis in children who were followed up at the Barbara Davies Cancer Center in Denver (n = 2,429) went from 41% to 59%, with a 7% annual rise, Arleta Rewers, MD, PhD, of Children’s Hospital Colorado, Denver, reported at the annual meeting of the European Association for the Study of Diabetes.
Meanwhile, in another analysis that included multiple U.S. centers and about 7,600 cases of youth-onset type 1 diabetes, the overall prevalence of DKA at diagnosis was 38.5% between 2010 and 2016. However, the prevalence had increased from 35% in 2010 to 40.6% in 2016, according to Elizabeth T. Jensen, MPH, PhD, of Wake Forest University, Winston-Salem, N.C. The annual increase in prevalence of DKA at diagnosis of type 1 disease was 2%, adjusted for sociodemographic factors.
Rising prevalence
“DKA occurs most commonly at the time of type 1 diabetes diagnosis,” observed Dr. Jensen, who noted that “in the United States, among children, it’s younger children, uninsured or underinsured children, and children from minority racial or ethnic groups, who are at greatest risk.”
Dr. Jensen and colleagues had previously shown that the prevalence of DKA at diagnosis was around 30% between 2002 and 2010, with no significant change in its prevalence. However, more recent reports from referral-based, single-center studies had suggested there was an increase, and that led her and her colleagues to take a closer look at the data.
To characterize the risk factors for DKA and the prevalence of DKA over time, Dr. Jensen and her team used the SEARCH for Diabetes in Youth database, which, she said, was “uniquely suited” for this purpose. SEARCH is a population-based, multicenter study conducted in centers in five U.S. states: South Carolina, Ohio, Colorado, California, and Washington.
A diagnosis of DKA was based on blood bicarbonate levels of less than 15 mmol/L, a venous pH of less than 7.25 or arterial or capillary pH of less than 7.3, or if there was any documentation of a DKA diagnosis.
As expected, the prevalence of DKA was highest in the youngest age group (0-4 years), Dr. Jensen said, but the increase in prevalence in that group was no different from the increases seen over time in the other age groups (5-9 years, 10-14 years, and 15 years or older).
There were no differences in the prevalence of DKA between the sexes, although there was a general increase over time. Similar trends were seen in DKA prevalence by race or ethnicity and by season, or time of year.
Of note, higher rates of DKA were seen in children who were covered by public health insurance, than in those covered by private insurance, although there was no difference in the rate of increase in DKA prevalence between the two groups. Dr. Jensen noted that only 64% of this study population had private insurance.
She said that future research in this area would need to look at the economic drivers and the “changing landscape of health insurance coverage in the United States.”
Expansion in health coverage
In presenting the findings of a study showing an increase in the prevalence of DKA at diagnosis of type 1 diabetes in children in Colorado from 2010 to 2017, Dr. Rewers said that the increase “paradoxically occurred” at a time of increasing health insurance coverage, a reference to the expansion of Medicaid during 2008-2012 and implementation in 2013 of the Affordable Care Act.
“Our group in Colorado has followed the frequency of DKA for almost 2 decades,” Dr. Rewers said. It’s important to study DKA as it is linked to worse glycemic control – with children with DKA having an HbA1c level of around 1% higher than those without DKA – and the potential for future, long-term complications.
Dr. Rewers noted that the increase in DKA at diagnosis of type 1 diabetes was more rapid in the children who had private rather than public health insurance. Of 1,187 patients with DKA, 57% had private health insurance, and 37% had public insurance, compared with 66% and 28%, respectively, in those without DKA. In 2010, the prevalence of DKA at diagnosis was 35.3% in those who were privately insured and 52.2% of those with public health insurance, but by 2017, a similar percentage of DKA was seen in the privately and publicly insured children (59.6% and 58.5%, respectively).
She said one possible explanation for that might be that “increased enrollment in high-deductible insurance plans could discourage families with private insurance from seeking timely care.”
Another explanation is that there is a low awareness of type 1 diabetes in the general population, she added. “Educational campaigns and autoimmunity screening have been shown to reduce DKA at diabetes diagnosis, but unfortunately they are not used widely at this point.”
Identifying at-risk children
“Diabetic ketoacidosis is a serious complication of diabetes [and] is difficult to diagnose because of the variability of the symptoms, said Angela Ibald-Mulli, PhD, who presented the findings of a retrospective cohort study in which she and her colleagues used a “discovery algorithm” called Q-Finder to identify the predictive factors for DKA in youth with type 1 diabetes, based on data from the Diabetes Prospective Follow-up Registry (DPV).
“The better we know the risk factors, the better we can care for our patients,” she emphasized.
The investigators obtained data on 108,223 patients with a diagnosis of type 1 disease and with more than two visits related to diabetes. The prevalence of DKA – defined as a pH of less than 7.3 during hospitalization occurring at least 10 days after the onset of type 1 diabetes – was 5.2%, said Dr. Ibald-Mulli, head of Medical Evidence Generation Primary Care at Sanofi, Paris.
A total of 129 different features were considered for their association with DKA – including comorbidities, sociodemographic factors, laboratory values, and concomitant medications – and were then used to identify, test, and the validate likely risk profiles.
After comparing the characteristics of patients with and without DKA, eight significant factors, all of which have been reported previously in the DPV cohort, were seen: younger age, lower body weight, higher HbA1c, younger age at onset of T1D; shorter disease duration; having a migration background; being less active; and having had more medical visits.
The investigators used the algorithm, and found 11 distinct profiles associated with DKA: an HbA1c higher than 8.87%; being aged 6-10 years; being aged 11-15 years; a diagnosis of nephropathy; DKA being present at onset; a prevalence of hypoglycemia with coma; a diagnosis of thyroiditis; a standardized body mass index lower than 16.9; not using short-acting insulin; younger than age 15 years; and not using continuous glucose monitoring.
Almost two-thirds of patients (64.7%) belonged to at least one of these risk profiles, Dr. Ibald-Mulli observed, with 7.1% of them having DKA, compared with 1.6% who belonged to none of the profiles.
Dr. Ibald-Mulli said it was important to note that the DKA risk profiles could overlap. “The more profiles a patient belongs to, the higher is the risk of having DKA,” she emphasized, adding that most patients (88.8%) with DKA belonged to just one profile, and fewer than 5% belonged to three or more profiles.
“Overall, the results of the algorithm confirmed known risk-factor profiles that had been previously identified by conventional statistical methods,” she concluded. It also provided “additional insights that can be further explored.”
SEARCH is funded by the Centers for Disease and Prevention and the National Institute of Diabetes and Digestive and Kidney Diseases. The DPV Registry is funded by multiple sponsors, including the European Federation for the Study of Diabetes and other academic institutions with the support of several commercial partners. Sanofi sponsored the study presented by Dr. Ibald-Mulli. Dr. Rewers made no disclosures, and Dr. Jensen did not have any conflicts of interest to declare. Dr. Ibald-Mulli is an employee of Sanofi.
SOURCE: Rewers A et al. EASD 2019, Abstract 115; Jensen E et al. EASD 2019, Abstract 116; Ibald-Mulli A et al. EASD 2019, Abstract 117.
BARCELONA – As many as 40%-60% of children have diabetic ketoacidosis (DKA) at the time of being diagnosed with type 1 diabetes, according to data from two U.S. analyses – and the figures have been rising for the past 10 years.
Between 2010 and 2017, the prevalence of DKA at diagnosis in children who were followed up at the Barbara Davies Cancer Center in Denver (n = 2,429) went from 41% to 59%, with a 7% annual rise, Arleta Rewers, MD, PhD, of Children’s Hospital Colorado, Denver, reported at the annual meeting of the European Association for the Study of Diabetes.
Meanwhile, in another analysis that included multiple U.S. centers and about 7,600 cases of youth-onset type 1 diabetes, the overall prevalence of DKA at diagnosis was 38.5% between 2010 and 2016. However, the prevalence had increased from 35% in 2010 to 40.6% in 2016, according to Elizabeth T. Jensen, MPH, PhD, of Wake Forest University, Winston-Salem, N.C. The annual increase in prevalence of DKA at diagnosis of type 1 disease was 2%, adjusted for sociodemographic factors.
Rising prevalence
“DKA occurs most commonly at the time of type 1 diabetes diagnosis,” observed Dr. Jensen, who noted that “in the United States, among children, it’s younger children, uninsured or underinsured children, and children from minority racial or ethnic groups, who are at greatest risk.”
Dr. Jensen and colleagues had previously shown that the prevalence of DKA at diagnosis was around 30% between 2002 and 2010, with no significant change in its prevalence. However, more recent reports from referral-based, single-center studies had suggested there was an increase, and that led her and her colleagues to take a closer look at the data.
To characterize the risk factors for DKA and the prevalence of DKA over time, Dr. Jensen and her team used the SEARCH for Diabetes in Youth database, which, she said, was “uniquely suited” for this purpose. SEARCH is a population-based, multicenter study conducted in centers in five U.S. states: South Carolina, Ohio, Colorado, California, and Washington.
A diagnosis of DKA was based on blood bicarbonate levels of less than 15 mmol/L, a venous pH of less than 7.25 or arterial or capillary pH of less than 7.3, or if there was any documentation of a DKA diagnosis.
As expected, the prevalence of DKA was highest in the youngest age group (0-4 years), Dr. Jensen said, but the increase in prevalence in that group was no different from the increases seen over time in the other age groups (5-9 years, 10-14 years, and 15 years or older).
There were no differences in the prevalence of DKA between the sexes, although there was a general increase over time. Similar trends were seen in DKA prevalence by race or ethnicity and by season, or time of year.
Of note, higher rates of DKA were seen in children who were covered by public health insurance, than in those covered by private insurance, although there was no difference in the rate of increase in DKA prevalence between the two groups. Dr. Jensen noted that only 64% of this study population had private insurance.
She said that future research in this area would need to look at the economic drivers and the “changing landscape of health insurance coverage in the United States.”
Expansion in health coverage
In presenting the findings of a study showing an increase in the prevalence of DKA at diagnosis of type 1 diabetes in children in Colorado from 2010 to 2017, Dr. Rewers said that the increase “paradoxically occurred” at a time of increasing health insurance coverage, a reference to the expansion of Medicaid during 2008-2012 and implementation in 2013 of the Affordable Care Act.
“Our group in Colorado has followed the frequency of DKA for almost 2 decades,” Dr. Rewers said. It’s important to study DKA as it is linked to worse glycemic control – with children with DKA having an HbA1c level of around 1% higher than those without DKA – and the potential for future, long-term complications.
Dr. Rewers noted that the increase in DKA at diagnosis of type 1 diabetes was more rapid in the children who had private rather than public health insurance. Of 1,187 patients with DKA, 57% had private health insurance, and 37% had public insurance, compared with 66% and 28%, respectively, in those without DKA. In 2010, the prevalence of DKA at diagnosis was 35.3% in those who were privately insured and 52.2% of those with public health insurance, but by 2017, a similar percentage of DKA was seen in the privately and publicly insured children (59.6% and 58.5%, respectively).
She said one possible explanation for that might be that “increased enrollment in high-deductible insurance plans could discourage families with private insurance from seeking timely care.”
Another explanation is that there is a low awareness of type 1 diabetes in the general population, she added. “Educational campaigns and autoimmunity screening have been shown to reduce DKA at diabetes diagnosis, but unfortunately they are not used widely at this point.”
Identifying at-risk children
“Diabetic ketoacidosis is a serious complication of diabetes [and] is difficult to diagnose because of the variability of the symptoms, said Angela Ibald-Mulli, PhD, who presented the findings of a retrospective cohort study in which she and her colleagues used a “discovery algorithm” called Q-Finder to identify the predictive factors for DKA in youth with type 1 diabetes, based on data from the Diabetes Prospective Follow-up Registry (DPV).
“The better we know the risk factors, the better we can care for our patients,” she emphasized.
The investigators obtained data on 108,223 patients with a diagnosis of type 1 disease and with more than two visits related to diabetes. The prevalence of DKA – defined as a pH of less than 7.3 during hospitalization occurring at least 10 days after the onset of type 1 diabetes – was 5.2%, said Dr. Ibald-Mulli, head of Medical Evidence Generation Primary Care at Sanofi, Paris.
A total of 129 different features were considered for their association with DKA – including comorbidities, sociodemographic factors, laboratory values, and concomitant medications – and were then used to identify, test, and the validate likely risk profiles.
After comparing the characteristics of patients with and without DKA, eight significant factors, all of which have been reported previously in the DPV cohort, were seen: younger age, lower body weight, higher HbA1c, younger age at onset of T1D; shorter disease duration; having a migration background; being less active; and having had more medical visits.
The investigators used the algorithm, and found 11 distinct profiles associated with DKA: an HbA1c higher than 8.87%; being aged 6-10 years; being aged 11-15 years; a diagnosis of nephropathy; DKA being present at onset; a prevalence of hypoglycemia with coma; a diagnosis of thyroiditis; a standardized body mass index lower than 16.9; not using short-acting insulin; younger than age 15 years; and not using continuous glucose monitoring.
Almost two-thirds of patients (64.7%) belonged to at least one of these risk profiles, Dr. Ibald-Mulli observed, with 7.1% of them having DKA, compared with 1.6% who belonged to none of the profiles.
Dr. Ibald-Mulli said it was important to note that the DKA risk profiles could overlap. “The more profiles a patient belongs to, the higher is the risk of having DKA,” she emphasized, adding that most patients (88.8%) with DKA belonged to just one profile, and fewer than 5% belonged to three or more profiles.
“Overall, the results of the algorithm confirmed known risk-factor profiles that had been previously identified by conventional statistical methods,” she concluded. It also provided “additional insights that can be further explored.”
SEARCH is funded by the Centers for Disease and Prevention and the National Institute of Diabetes and Digestive and Kidney Diseases. The DPV Registry is funded by multiple sponsors, including the European Federation for the Study of Diabetes and other academic institutions with the support of several commercial partners. Sanofi sponsored the study presented by Dr. Ibald-Mulli. Dr. Rewers made no disclosures, and Dr. Jensen did not have any conflicts of interest to declare. Dr. Ibald-Mulli is an employee of Sanofi.
SOURCE: Rewers A et al. EASD 2019, Abstract 115; Jensen E et al. EASD 2019, Abstract 116; Ibald-Mulli A et al. EASD 2019, Abstract 117.
BARCELONA – As many as 40%-60% of children have diabetic ketoacidosis (DKA) at the time of being diagnosed with type 1 diabetes, according to data from two U.S. analyses – and the figures have been rising for the past 10 years.
Between 2010 and 2017, the prevalence of DKA at diagnosis in children who were followed up at the Barbara Davies Cancer Center in Denver (n = 2,429) went from 41% to 59%, with a 7% annual rise, Arleta Rewers, MD, PhD, of Children’s Hospital Colorado, Denver, reported at the annual meeting of the European Association for the Study of Diabetes.
Meanwhile, in another analysis that included multiple U.S. centers and about 7,600 cases of youth-onset type 1 diabetes, the overall prevalence of DKA at diagnosis was 38.5% between 2010 and 2016. However, the prevalence had increased from 35% in 2010 to 40.6% in 2016, according to Elizabeth T. Jensen, MPH, PhD, of Wake Forest University, Winston-Salem, N.C. The annual increase in prevalence of DKA at diagnosis of type 1 disease was 2%, adjusted for sociodemographic factors.
Rising prevalence
“DKA occurs most commonly at the time of type 1 diabetes diagnosis,” observed Dr. Jensen, who noted that “in the United States, among children, it’s younger children, uninsured or underinsured children, and children from minority racial or ethnic groups, who are at greatest risk.”
Dr. Jensen and colleagues had previously shown that the prevalence of DKA at diagnosis was around 30% between 2002 and 2010, with no significant change in its prevalence. However, more recent reports from referral-based, single-center studies had suggested there was an increase, and that led her and her colleagues to take a closer look at the data.
To characterize the risk factors for DKA and the prevalence of DKA over time, Dr. Jensen and her team used the SEARCH for Diabetes in Youth database, which, she said, was “uniquely suited” for this purpose. SEARCH is a population-based, multicenter study conducted in centers in five U.S. states: South Carolina, Ohio, Colorado, California, and Washington.
A diagnosis of DKA was based on blood bicarbonate levels of less than 15 mmol/L, a venous pH of less than 7.25 or arterial or capillary pH of less than 7.3, or if there was any documentation of a DKA diagnosis.
As expected, the prevalence of DKA was highest in the youngest age group (0-4 years), Dr. Jensen said, but the increase in prevalence in that group was no different from the increases seen over time in the other age groups (5-9 years, 10-14 years, and 15 years or older).
There were no differences in the prevalence of DKA between the sexes, although there was a general increase over time. Similar trends were seen in DKA prevalence by race or ethnicity and by season, or time of year.
Of note, higher rates of DKA were seen in children who were covered by public health insurance, than in those covered by private insurance, although there was no difference in the rate of increase in DKA prevalence between the two groups. Dr. Jensen noted that only 64% of this study population had private insurance.
She said that future research in this area would need to look at the economic drivers and the “changing landscape of health insurance coverage in the United States.”
Expansion in health coverage
In presenting the findings of a study showing an increase in the prevalence of DKA at diagnosis of type 1 diabetes in children in Colorado from 2010 to 2017, Dr. Rewers said that the increase “paradoxically occurred” at a time of increasing health insurance coverage, a reference to the expansion of Medicaid during 2008-2012 and implementation in 2013 of the Affordable Care Act.
“Our group in Colorado has followed the frequency of DKA for almost 2 decades,” Dr. Rewers said. It’s important to study DKA as it is linked to worse glycemic control – with children with DKA having an HbA1c level of around 1% higher than those without DKA – and the potential for future, long-term complications.
Dr. Rewers noted that the increase in DKA at diagnosis of type 1 diabetes was more rapid in the children who had private rather than public health insurance. Of 1,187 patients with DKA, 57% had private health insurance, and 37% had public insurance, compared with 66% and 28%, respectively, in those without DKA. In 2010, the prevalence of DKA at diagnosis was 35.3% in those who were privately insured and 52.2% of those with public health insurance, but by 2017, a similar percentage of DKA was seen in the privately and publicly insured children (59.6% and 58.5%, respectively).
She said one possible explanation for that might be that “increased enrollment in high-deductible insurance plans could discourage families with private insurance from seeking timely care.”
Another explanation is that there is a low awareness of type 1 diabetes in the general population, she added. “Educational campaigns and autoimmunity screening have been shown to reduce DKA at diabetes diagnosis, but unfortunately they are not used widely at this point.”
Identifying at-risk children
“Diabetic ketoacidosis is a serious complication of diabetes [and] is difficult to diagnose because of the variability of the symptoms, said Angela Ibald-Mulli, PhD, who presented the findings of a retrospective cohort study in which she and her colleagues used a “discovery algorithm” called Q-Finder to identify the predictive factors for DKA in youth with type 1 diabetes, based on data from the Diabetes Prospective Follow-up Registry (DPV).
“The better we know the risk factors, the better we can care for our patients,” she emphasized.
The investigators obtained data on 108,223 patients with a diagnosis of type 1 disease and with more than two visits related to diabetes. The prevalence of DKA – defined as a pH of less than 7.3 during hospitalization occurring at least 10 days after the onset of type 1 diabetes – was 5.2%, said Dr. Ibald-Mulli, head of Medical Evidence Generation Primary Care at Sanofi, Paris.
A total of 129 different features were considered for their association with DKA – including comorbidities, sociodemographic factors, laboratory values, and concomitant medications – and were then used to identify, test, and the validate likely risk profiles.
After comparing the characteristics of patients with and without DKA, eight significant factors, all of which have been reported previously in the DPV cohort, were seen: younger age, lower body weight, higher HbA1c, younger age at onset of T1D; shorter disease duration; having a migration background; being less active; and having had more medical visits.
The investigators used the algorithm, and found 11 distinct profiles associated with DKA: an HbA1c higher than 8.87%; being aged 6-10 years; being aged 11-15 years; a diagnosis of nephropathy; DKA being present at onset; a prevalence of hypoglycemia with coma; a diagnosis of thyroiditis; a standardized body mass index lower than 16.9; not using short-acting insulin; younger than age 15 years; and not using continuous glucose monitoring.
Almost two-thirds of patients (64.7%) belonged to at least one of these risk profiles, Dr. Ibald-Mulli observed, with 7.1% of them having DKA, compared with 1.6% who belonged to none of the profiles.
Dr. Ibald-Mulli said it was important to note that the DKA risk profiles could overlap. “The more profiles a patient belongs to, the higher is the risk of having DKA,” she emphasized, adding that most patients (88.8%) with DKA belonged to just one profile, and fewer than 5% belonged to three or more profiles.
“Overall, the results of the algorithm confirmed known risk-factor profiles that had been previously identified by conventional statistical methods,” she concluded. It also provided “additional insights that can be further explored.”
SEARCH is funded by the Centers for Disease and Prevention and the National Institute of Diabetes and Digestive and Kidney Diseases. The DPV Registry is funded by multiple sponsors, including the European Federation for the Study of Diabetes and other academic institutions with the support of several commercial partners. Sanofi sponsored the study presented by Dr. Ibald-Mulli. Dr. Rewers made no disclosures, and Dr. Jensen did not have any conflicts of interest to declare. Dr. Ibald-Mulli is an employee of Sanofi.
SOURCE: Rewers A et al. EASD 2019, Abstract 115; Jensen E et al. EASD 2019, Abstract 116; Ibald-Mulli A et al. EASD 2019, Abstract 117.
REPORTING FROM EASD 2019
Families face challenges of gene therapy
WASHINGTON– Gene therapy for the treatment of rare diseases continues to develop and new products are entering the pipeline; however, more work is needed to make the gene therapy experience easier on patients and their families, according to members of a panel at the NORD Rare Diseases & Orphan Product Breakthrough Summit, held by the National Organization for Rare Disorders.
Companies developing gene therapy cite their main challenges as identifying patients, developing clinical trials, coordinating treatment and supporting families, managing reimbursement, and manufacturing the treatment, said Mark Rothera, president and CEO of Orchard Therapeutics, developer of ex vivo autologous hematopoietic stem cell gene therapy.
For families of patients with rare diseases who are undergoing gene therapy, challenges include struggles such as language barriers, lack of wifi, and separation from other family members for extended periods, according to Amy Price, mother of a gene therapy recipient, as well as principal consultant to Rarallel and an advocate for metachromatic leukodystrophy.
Ms. Price cited a survey she conducted of families with children who underwent gene therapy. She collected data from 16 families about their initial visit as part of a gene therapy trial; the trials included 14 families in Milan; 1 in Bethesda, Md.; and 1 in Paris. The average age of the patients at the start of the trial was 3 years, with a range of 8 months to 11 years. The trials were conducted between 1990 and 2018.
Families participating in the trials spent an average of 5.5 months in the city where the trial was conducted, and an average of 48 days in an isolation ward with their child at the start of the study.
The five biggest challenges were financial well-being (cited by 60% of survey respondents), social isolation/being away from support system (60%), fear of the unknown/long-term treatment diagnosis (73%), family separation (67%), and caring for other children simultaneous during the trial period (60%).
In addition, patients averaged 12 follow-up visits, and the most common secondary challenges cited in the survey included time spent at the hospital, emotional and physical stress on the patient, fear of test results and outcomes, exhaustion, time away from work and school, and travel logistics.
Other stressors include language barriers and not being in children’s hospital, Ms. Price said.
Ms. Price proposed patient-focused solutions such as addressing cultural challenges, connecting families to local resources, and providing clinical follow-up locally to reduce the burden of travel to the trial site.
WASHINGTON– Gene therapy for the treatment of rare diseases continues to develop and new products are entering the pipeline; however, more work is needed to make the gene therapy experience easier on patients and their families, according to members of a panel at the NORD Rare Diseases & Orphan Product Breakthrough Summit, held by the National Organization for Rare Disorders.
Companies developing gene therapy cite their main challenges as identifying patients, developing clinical trials, coordinating treatment and supporting families, managing reimbursement, and manufacturing the treatment, said Mark Rothera, president and CEO of Orchard Therapeutics, developer of ex vivo autologous hematopoietic stem cell gene therapy.
For families of patients with rare diseases who are undergoing gene therapy, challenges include struggles such as language barriers, lack of wifi, and separation from other family members for extended periods, according to Amy Price, mother of a gene therapy recipient, as well as principal consultant to Rarallel and an advocate for metachromatic leukodystrophy.
Ms. Price cited a survey she conducted of families with children who underwent gene therapy. She collected data from 16 families about their initial visit as part of a gene therapy trial; the trials included 14 families in Milan; 1 in Bethesda, Md.; and 1 in Paris. The average age of the patients at the start of the trial was 3 years, with a range of 8 months to 11 years. The trials were conducted between 1990 and 2018.
Families participating in the trials spent an average of 5.5 months in the city where the trial was conducted, and an average of 48 days in an isolation ward with their child at the start of the study.
The five biggest challenges were financial well-being (cited by 60% of survey respondents), social isolation/being away from support system (60%), fear of the unknown/long-term treatment diagnosis (73%), family separation (67%), and caring for other children simultaneous during the trial period (60%).
In addition, patients averaged 12 follow-up visits, and the most common secondary challenges cited in the survey included time spent at the hospital, emotional and physical stress on the patient, fear of test results and outcomes, exhaustion, time away from work and school, and travel logistics.
Other stressors include language barriers and not being in children’s hospital, Ms. Price said.
Ms. Price proposed patient-focused solutions such as addressing cultural challenges, connecting families to local resources, and providing clinical follow-up locally to reduce the burden of travel to the trial site.
WASHINGTON– Gene therapy for the treatment of rare diseases continues to develop and new products are entering the pipeline; however, more work is needed to make the gene therapy experience easier on patients and their families, according to members of a panel at the NORD Rare Diseases & Orphan Product Breakthrough Summit, held by the National Organization for Rare Disorders.
Companies developing gene therapy cite their main challenges as identifying patients, developing clinical trials, coordinating treatment and supporting families, managing reimbursement, and manufacturing the treatment, said Mark Rothera, president and CEO of Orchard Therapeutics, developer of ex vivo autologous hematopoietic stem cell gene therapy.
For families of patients with rare diseases who are undergoing gene therapy, challenges include struggles such as language barriers, lack of wifi, and separation from other family members for extended periods, according to Amy Price, mother of a gene therapy recipient, as well as principal consultant to Rarallel and an advocate for metachromatic leukodystrophy.
Ms. Price cited a survey she conducted of families with children who underwent gene therapy. She collected data from 16 families about their initial visit as part of a gene therapy trial; the trials included 14 families in Milan; 1 in Bethesda, Md.; and 1 in Paris. The average age of the patients at the start of the trial was 3 years, with a range of 8 months to 11 years. The trials were conducted between 1990 and 2018.
Families participating in the trials spent an average of 5.5 months in the city where the trial was conducted, and an average of 48 days in an isolation ward with their child at the start of the study.
The five biggest challenges were financial well-being (cited by 60% of survey respondents), social isolation/being away from support system (60%), fear of the unknown/long-term treatment diagnosis (73%), family separation (67%), and caring for other children simultaneous during the trial period (60%).
In addition, patients averaged 12 follow-up visits, and the most common secondary challenges cited in the survey included time spent at the hospital, emotional and physical stress on the patient, fear of test results and outcomes, exhaustion, time away from work and school, and travel logistics.
Other stressors include language barriers and not being in children’s hospital, Ms. Price said.
Ms. Price proposed patient-focused solutions such as addressing cultural challenges, connecting families to local resources, and providing clinical follow-up locally to reduce the burden of travel to the trial site.
EXPERT ANALYSIS FROM NORD 2019
Student vapers make mint the most popular Juul flavor
according to data from the 2019 Monitoring the Future study.

Almost half (47.1%) of the 12th graders who had used Juul e-cigarettes in the past 30 days reported that mint was the flavor they most often used, compared with 23.8% for mango and 8.6% for fruit, which is a combination of flavors, Adam M. Leventhal, PhD, of the University of Southern California, Los Angeles, and associates wrote in JAMA.
Mint was also the flavor most often used by 10th graders (43.5%), with mango again second at 27.3%, and fruit third at 10.8%. Eighth-grade students switched mango (33.5%) and mint (29.2%) but had fruit third again at 16.0%, the investigators reported, based on data for 1,739 respondents to the Monitoring the Future survey who had used a vaping product within the past 30 days.
Juul has suspended sales of four – mango, fruit, creme, and cucumber – of its original eight flavors, Dr. Leventhal and associates noted, and e-cigarette flavors other than tobacco, menthol, and mint have been prohibited by some local municipalities.
“The current findings raise uncertainty whether regulations or sales suspensions that exempt mint flavors are optimal strategies for reducing youth e-cigarette use,” they wrote.
As this article was being written, the Wall Street Journal had just reported that the Food and Drug Administration will ban mint and all other e-cigarette flavors except tobacco and menthol.
SOURCE: Leventhal AM et al. JAMA. 2019 Nov 5. doi: 10.1001/jama.2019.17968.
according to data from the 2019 Monitoring the Future study.

Almost half (47.1%) of the 12th graders who had used Juul e-cigarettes in the past 30 days reported that mint was the flavor they most often used, compared with 23.8% for mango and 8.6% for fruit, which is a combination of flavors, Adam M. Leventhal, PhD, of the University of Southern California, Los Angeles, and associates wrote in JAMA.
Mint was also the flavor most often used by 10th graders (43.5%), with mango again second at 27.3%, and fruit third at 10.8%. Eighth-grade students switched mango (33.5%) and mint (29.2%) but had fruit third again at 16.0%, the investigators reported, based on data for 1,739 respondents to the Monitoring the Future survey who had used a vaping product within the past 30 days.
Juul has suspended sales of four – mango, fruit, creme, and cucumber – of its original eight flavors, Dr. Leventhal and associates noted, and e-cigarette flavors other than tobacco, menthol, and mint have been prohibited by some local municipalities.
“The current findings raise uncertainty whether regulations or sales suspensions that exempt mint flavors are optimal strategies for reducing youth e-cigarette use,” they wrote.
As this article was being written, the Wall Street Journal had just reported that the Food and Drug Administration will ban mint and all other e-cigarette flavors except tobacco and menthol.
SOURCE: Leventhal AM et al. JAMA. 2019 Nov 5. doi: 10.1001/jama.2019.17968.
according to data from the 2019 Monitoring the Future study.

Almost half (47.1%) of the 12th graders who had used Juul e-cigarettes in the past 30 days reported that mint was the flavor they most often used, compared with 23.8% for mango and 8.6% for fruit, which is a combination of flavors, Adam M. Leventhal, PhD, of the University of Southern California, Los Angeles, and associates wrote in JAMA.
Mint was also the flavor most often used by 10th graders (43.5%), with mango again second at 27.3%, and fruit third at 10.8%. Eighth-grade students switched mango (33.5%) and mint (29.2%) but had fruit third again at 16.0%, the investigators reported, based on data for 1,739 respondents to the Monitoring the Future survey who had used a vaping product within the past 30 days.
Juul has suspended sales of four – mango, fruit, creme, and cucumber – of its original eight flavors, Dr. Leventhal and associates noted, and e-cigarette flavors other than tobacco, menthol, and mint have been prohibited by some local municipalities.
“The current findings raise uncertainty whether regulations or sales suspensions that exempt mint flavors are optimal strategies for reducing youth e-cigarette use,” they wrote.
As this article was being written, the Wall Street Journal had just reported that the Food and Drug Administration will ban mint and all other e-cigarette flavors except tobacco and menthol.
SOURCE: Leventhal AM et al. JAMA. 2019 Nov 5. doi: 10.1001/jama.2019.17968.
FROM JAMA
Caspofungin bests fluconazole for antifungal prophylaxis in young AML patients
Antifungal prophylaxis with caspofungin led to a lower incidence of invasive fungal disease, compared with fluconazole, in children and young adults with acute myeloid leukemia (AML), according to new study findings.
The results suggest caspofungin may be an appropriate prophylactic strategy to prevent invasive fungal disease in younger patients with newly diagnosed AML, reported Brian T. Fisher, DO, of the University of Pennsylvania, Philadelphia, and colleagues. The study was published in JAMA.
The randomized, open-label study included 517 children, adolescents, and young adults with de novo, relapsed, or secondary AML. Study patients received treatment in 115 centers throughout United States and Canada. The median age of patients in the study was 9 years (range, 0-26 years); 56% were male and approximately 69% were white.
Study subjects were randomly assigned to receive antifungal prophylaxis with 70 mg/m2 (maximum dose 70 mg/day) of intravenous caspofungin on day 1, followed by 50 mg/m2 per day (maximum dose 50 mg/day) thereafter, or age-dosed intravenous or oral fluconazole.
Prophylactic therapy was initiated 24-72 hours after the completion of each chemotherapy cycle, and was maintained until the end of the neutropenic period following each cycle.
At 5-month follow-up, the cumulative incidence rate of probable or proven invasive fungal disease was 3.1% (95% confidence interval, 1.3%-7.0%) in the caspofungin arm, compared with 7.2% (95% CI, 4.4%-11.8%) in the fluconazole arm (overall P = .03).
In addition, the 5-month cumulative incidence rate of probable or proven invasive aspergillosis infection was 0.5% (95%CI, 0.1%-3.5%) in patients who received caspofungin, compared with 3.1% (95% CI, 1.4%-6.9%) in patients who received fluconazole (overall P = .046).
“No statistically significant differences in empirical antifungal therapy or 2-year overall survival were observed,” they reported.
With respect to safety, the most frequently reported adverse events were hypokalemia (22 events in the caspofungin arm vs. 13 in the fluconazole arm), respiratory failure (6 events in the caspofungin arm vs. 9 in the fluconazole arm), and elevated alanine transaminase (4 events in the caspofungin arm vs. 8 in the fluconazole arm).
The researchers acknowledged a key limitation of the study was the short duration of follow-up. As a result, the precision of some comparative measures may have been reduced.
The National Cancer Institute funded the study. The authors reported financial affiliations with Astellas, Celgene, Leadiant Biosciences, Merck, Nabriva Therapeutics, Novartis, Pfizer, Shire, and T2 Biosystems.
SOURCE: Fisher BT et al. JAMA. 2019;322(17):1673-81.
Antifungal prophylaxis with caspofungin led to a lower incidence of invasive fungal disease, compared with fluconazole, in children and young adults with acute myeloid leukemia (AML), according to new study findings.
The results suggest caspofungin may be an appropriate prophylactic strategy to prevent invasive fungal disease in younger patients with newly diagnosed AML, reported Brian T. Fisher, DO, of the University of Pennsylvania, Philadelphia, and colleagues. The study was published in JAMA.
The randomized, open-label study included 517 children, adolescents, and young adults with de novo, relapsed, or secondary AML. Study patients received treatment in 115 centers throughout United States and Canada. The median age of patients in the study was 9 years (range, 0-26 years); 56% were male and approximately 69% were white.
Study subjects were randomly assigned to receive antifungal prophylaxis with 70 mg/m2 (maximum dose 70 mg/day) of intravenous caspofungin on day 1, followed by 50 mg/m2 per day (maximum dose 50 mg/day) thereafter, or age-dosed intravenous or oral fluconazole.
Prophylactic therapy was initiated 24-72 hours after the completion of each chemotherapy cycle, and was maintained until the end of the neutropenic period following each cycle.
At 5-month follow-up, the cumulative incidence rate of probable or proven invasive fungal disease was 3.1% (95% confidence interval, 1.3%-7.0%) in the caspofungin arm, compared with 7.2% (95% CI, 4.4%-11.8%) in the fluconazole arm (overall P = .03).
In addition, the 5-month cumulative incidence rate of probable or proven invasive aspergillosis infection was 0.5% (95%CI, 0.1%-3.5%) in patients who received caspofungin, compared with 3.1% (95% CI, 1.4%-6.9%) in patients who received fluconazole (overall P = .046).
“No statistically significant differences in empirical antifungal therapy or 2-year overall survival were observed,” they reported.
With respect to safety, the most frequently reported adverse events were hypokalemia (22 events in the caspofungin arm vs. 13 in the fluconazole arm), respiratory failure (6 events in the caspofungin arm vs. 9 in the fluconazole arm), and elevated alanine transaminase (4 events in the caspofungin arm vs. 8 in the fluconazole arm).
The researchers acknowledged a key limitation of the study was the short duration of follow-up. As a result, the precision of some comparative measures may have been reduced.
The National Cancer Institute funded the study. The authors reported financial affiliations with Astellas, Celgene, Leadiant Biosciences, Merck, Nabriva Therapeutics, Novartis, Pfizer, Shire, and T2 Biosystems.
SOURCE: Fisher BT et al. JAMA. 2019;322(17):1673-81.
Antifungal prophylaxis with caspofungin led to a lower incidence of invasive fungal disease, compared with fluconazole, in children and young adults with acute myeloid leukemia (AML), according to new study findings.
The results suggest caspofungin may be an appropriate prophylactic strategy to prevent invasive fungal disease in younger patients with newly diagnosed AML, reported Brian T. Fisher, DO, of the University of Pennsylvania, Philadelphia, and colleagues. The study was published in JAMA.
The randomized, open-label study included 517 children, adolescents, and young adults with de novo, relapsed, or secondary AML. Study patients received treatment in 115 centers throughout United States and Canada. The median age of patients in the study was 9 years (range, 0-26 years); 56% were male and approximately 69% were white.
Study subjects were randomly assigned to receive antifungal prophylaxis with 70 mg/m2 (maximum dose 70 mg/day) of intravenous caspofungin on day 1, followed by 50 mg/m2 per day (maximum dose 50 mg/day) thereafter, or age-dosed intravenous or oral fluconazole.
Prophylactic therapy was initiated 24-72 hours after the completion of each chemotherapy cycle, and was maintained until the end of the neutropenic period following each cycle.
At 5-month follow-up, the cumulative incidence rate of probable or proven invasive fungal disease was 3.1% (95% confidence interval, 1.3%-7.0%) in the caspofungin arm, compared with 7.2% (95% CI, 4.4%-11.8%) in the fluconazole arm (overall P = .03).
In addition, the 5-month cumulative incidence rate of probable or proven invasive aspergillosis infection was 0.5% (95%CI, 0.1%-3.5%) in patients who received caspofungin, compared with 3.1% (95% CI, 1.4%-6.9%) in patients who received fluconazole (overall P = .046).
“No statistically significant differences in empirical antifungal therapy or 2-year overall survival were observed,” they reported.
With respect to safety, the most frequently reported adverse events were hypokalemia (22 events in the caspofungin arm vs. 13 in the fluconazole arm), respiratory failure (6 events in the caspofungin arm vs. 9 in the fluconazole arm), and elevated alanine transaminase (4 events in the caspofungin arm vs. 8 in the fluconazole arm).
The researchers acknowledged a key limitation of the study was the short duration of follow-up. As a result, the precision of some comparative measures may have been reduced.
The National Cancer Institute funded the study. The authors reported financial affiliations with Astellas, Celgene, Leadiant Biosciences, Merck, Nabriva Therapeutics, Novartis, Pfizer, Shire, and T2 Biosystems.
SOURCE: Fisher BT et al. JAMA. 2019;322(17):1673-81.
FROM JAMA
Systemic Epstein-Barr Virus–Positive T-cell Lymphoma of Childhood
Case Report
A 7-year-old Chinese boy presented with multiple painful oral and tongue ulcers of 2 weeks’ duration as well as acute onset of moderate to high fever (highest temperature, 39.3°C) for 5 days. The fever was reported to have run a relapsing course, accompanied by rigors but without convulsions or cognitive changes. At times, the patient had nasal congestion, nasal discharge, and cough. He also had a transient eruption on the back and hands as well as an indurated red nodule on the left forearm.
Before the patient was hospitalized, antibiotic therapy was administered by other physicians, but the condition of fever and oral ulcers did not improve. After the patient was hospitalized, new tender nodules emerged on the scalp, buttocks, and lower extremities. New ulcers also appeared on the palate.
History
Two months earlier, the patient had presented with a painful perioral skin ulcer that resolved after being treated as contagious eczema. Another dermatologist previously had considered a diagnosis of hand-foot-and-mouth disease.
The patient was born by normal spontaneous vaginal delivery, without abnormality. He was breastfed; feeding, growth, and the developmental history showed no abnormality. He was the family’s eldest child, with a healthy brother and sister. There was no history of familial illness. He received bacillus Calmette-Guérin and poliomyelitis vaccines after birth; the rest of the vaccine history was unclear. There was no history of immunologic abnormality.
Physical Examination
A 1.5×1.5-cm, warm, red nodule with a central black crust was noted on the left forearm (Figure 1A). Several similar lesions were noted on the buttocks, scalp, and lower extremities. Multiple ulcers, as large as 1 cm, were present on the tongue, palate, and left angle of the mouth (Figure 1B). The pharynx was congested, and the tonsils were mildly enlarged. Multiple enlarged, movable, nontender lymph nodes could be palpated in the cervical basins, axillae, and groin. No purpura or ecchymosis was detected.
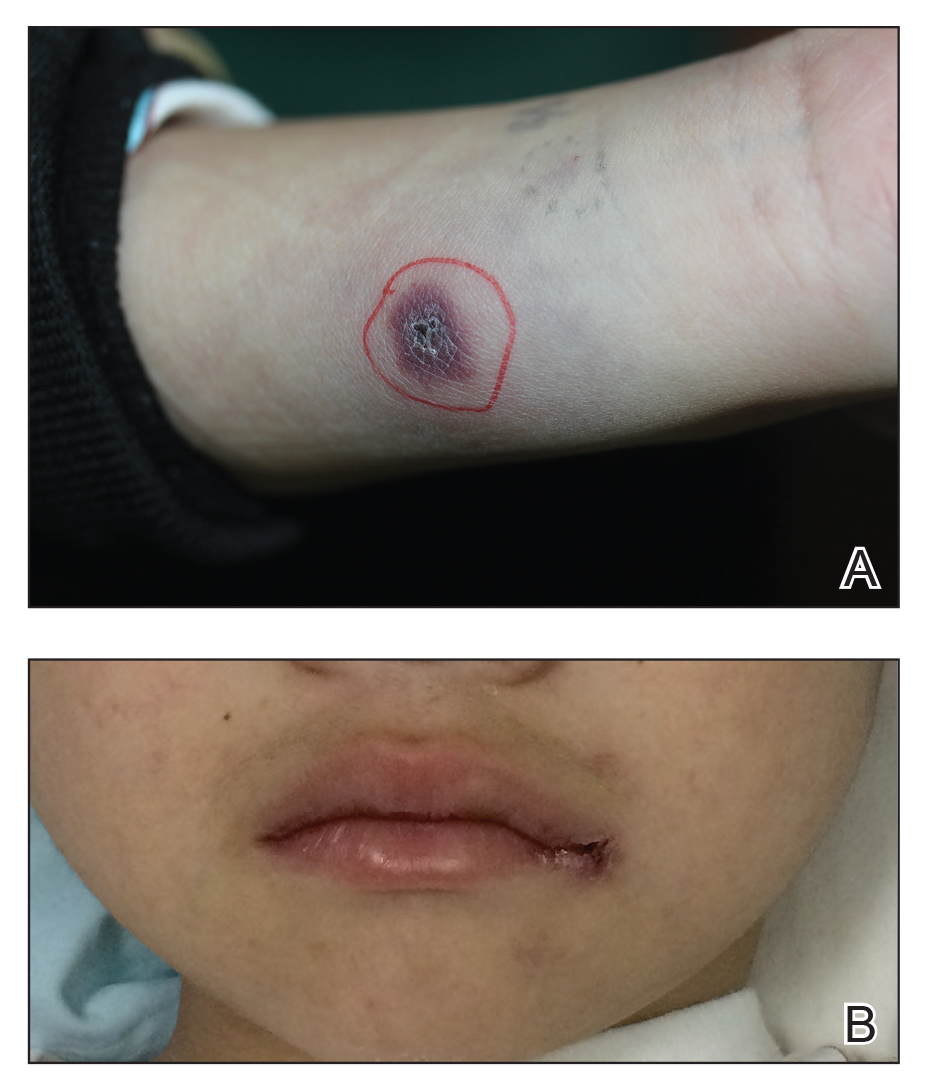
Laboratory Results
Laboratory testing revealed a normal total white blood cell count (4.26×109/L [reference range, 4.0–12.0×109/L]), with normal neutrophils (1.36×109/L [reference range, 1.32–7.90×109/L]), lymphocytes (2.77×109/L [reference range, 1.20–6.00×109/L]), and monocytes (0.13×109/L [reference range, 0.08–0.80×109/L]); a mildly decreased hemoglobin level (115 g/L [reference range, 120–160 g/L]); a normal platelet count (102×109/L [reference range, 100–380×109/L]); an elevated lactate dehydrogenase level (614 U/L [reference range, 110–330 U/L]); an elevated α-hydroxybutyrate dehydrogenase level (483 U/L [reference range, 120–270 U/L]); elevated prothrombin time (15.3 s [reference range, 9–14 s]); elevated activated partial thromboplastin time (59.8 s [reference range, 20.6–39.6 s]); and an elevated D-dimer level (1.51 mg/L [reference range, <0.73 mg/L]). In addition, autoantibody testing revealed a positive antinuclear antibody titer of 1:320 and a strong positive anti–Ro-52 level.
The peripheral blood lymphocyte classification demonstrated a prominent elevated percentage of T lymphocytes, with predominantly CD8+ cells (CD3, 94.87%; CD8, 71.57%; CD4, 24.98%; CD4:CD8 ratio, 0.35) and a diminished percentage of B lymphocytes and natural killer (NK) cells. Epstein-Barr virus (EBV) antibody testing was positive for anti–viral capsid antigen (VCA) IgG and negative for anti-VCA IgM.
Smears of the ulcer on the tongue demonstrated gram-positive cocci, gram-negative bacilli, and diplococci. Culture of sputum showed methicillin-resistant Staphylococcus aureus. Inspection for acid-fast bacilli in sputum yielded negative results 3 times. A purified protein derivative skin test for Mycobacterium tuberculosis infection was negative.
Imaging and Other Studies
Computed tomography of the chest and abdomen demonstrated 2 nodular opacities on the lower right lung; spotted opacities on the upper right lung; floccular opacities on the rest area of the lung; mild pleural effusion; enlargement of lymph nodes on the mediastinum, the bilateral hilum of the lung, and mesentery; and hepatosplenomegaly. Electrocardiography showed sinus tachycardia. Nasal cavity endoscopy showed sinusitis. Fundus examination showed vasculopathy of the left retina. A colonoscopy showed normal mucosa.
Histopathology
Biopsy of the nodule on the left arm showed dense, superficial to deep perivascular, periadnexal, perineural, and panniculitislike lymphoid infiltrates, as well as a sparse interstitial infiltrate with irregular and pleomorphic medium to large nuclei. Lymphoid cells showed mild epidermotropism, with tagging to the basal layer. Some vessel walls were infiltrated by similar cells (Figure 2). Infiltrative atypical lymphoid cells expressed CD3 and CD7 and were mostly CD8+, with a few CD4+ cells and most cells negative for CD5, CD20, CD30, CD56, and anaplastic lymphoma kinase. Cytotoxic markers granzyme B and T-cell intracellular antigen protein 1 were scattered positive. Immunostaining for Ki-67 protein highlighted an increased proliferative rate of 80% in malignant cells. In situ hybridization for EBV-encoded RNA (EBER) demonstrated EBV-positive atypical lymphoid cells (Figure 3). Analysis for T-cell receptor (TCR) γ gene rearrangement revealed a monoclonal pattern. Bone marrow aspirate showed proliferation of the 3 cell lines. The percentage of T lymphocytes was increased (20% of all nucleated cells). No hemophagocytic activity was found.
Diagnosis
A diagnosis of systemic EBV-positive T-cell lymphoma was made. Before the final diagnosis was made, the patient was treated by rheumatologists with antibiotics, antiviral drugs, nonsteroidal anti-inflammatory drugs, and other symptomatic treatments. Following antibiotic therapy, a sputum culture reverted to normal flora, the coagulation index (ie, prothrombin time, activated partial thromboplastin time) returned to normal, and the D-dimer level decreased to 1.19 mg/L.
The patient’s parents refused to accept chemotherapy for him. Instead, they chose herbal therapy only; 5 months later, they reported that all of his symptoms had resolved; however, the disease suddenly relapsed after another 7 months, with multiple skin nodules and fever. The patient died, even with chemotherapy in another hospital.
Comment
Prevalence and Presentation
Epstein-Barr virus is a ubiquitous γ-herpesvirus with tropism for B cells, affecting more than 90% of the adult population worldwide. In addition to infecting B cells, EBV is capable of infecting T and NK cells, leading to various EBV-related lymphoproliferative disorders (LPDs). The frequency and clinical presentation of infection varies based on the type of EBV-infected cells and the state of host immunity.1-3
Primary infection usually is asymptomatic and occurs early in life; when symptomatic, the disease usually presents as infectious mononucleosis (IM), characterized by polyclonal expansion of infected B cells and subsequent cytotoxic T-cell response. A diagnosis of EBV infection can be made by testing for specific IgM and IgG antibodies against VCA, early antigens, and EBV nuclear antigen proteins.3,4
Associated LPDs
Although most symptoms associated with IM resolve within weeks or months, persistent or recurrent IM-like symptoms or even lasting disease occasionally occur, particularly in children and young adults. This complication is known as chronic active EBV infection (CAEBV), frequently associated with EBV-infected T-cell or NK-cell proliferation, especially in East Asian populations.3,5
Epstein-Barr virus–positive T-cell and NK-cell LPDs of childhood include CAEBV infection of T-cell and NK-cell types and systemic EBV-positive T-cell lymphoma of childhood. The former includes hydroa vacciniforme–like LPD and severe mosquito bite allergy.3
Systemic EBV-Positive T-cell Lymphoma of Childhood
This entity occurs not only in children but also in adolescents and young adults. A fulminant illness characterized by clonal proliferation of EBV-infected cytotoxic T cells, it can develop shortly after primary EBV infection or is linked to CAEBV infection. The disorder is rare and has a racial predilection for Asian (ie, Japanese, Chinese, Korean) populations and indigenous populations of Mexico and Central and South America.6-8
Complications
Systemic EBV-positive T-cell lymphoma of childhood is often complicated by hemophagocytic syndrome, coagulopathy, sepsis, and multiorgan failure. Other signs and symptoms include high fever, rash, jaundice, diarrhea, pancytopenia, and hepatosplenomegaly. The liver, spleen, lymph nodes, and bone marrow are commonly involved, and the disease can involve skin, the heart, and the lungs.9,10
Diagnosis
When systemic EBV-positive T-cell lymphoma occurs shortly after IM, serology shows low or absent anti-VCA IgM and positive anti-VCA IgG. Infiltrating T cells usually are small and lack cytologic atypia; however, cases with pleomorphic, medium to large lymphoid cells, irregular nuclei, and frequent mitoses have been described. Hemophagocytosis can be seen in the liver, spleen, and bone marrow.3,11
The most typical phenotype of systemic EBV-positive T-cell lymphoma is CD2+CD3+CD8+CD20−CD56−, with expression of the cytotoxic granules known as T-cell intracellular antigen 1 and granzyme B. Rare cases of CD4+ and mixed CD4+/CD8+ phenotypes have been described, usually in the setting of CAEBV infection.3,12 Neoplastic cells have monoclonally rearranged TCR-γ genes and consistent EBER positivity with in situ hybridization.13 A final diagnosis is based on a comprehensive analysis of clinical, morphological, immunohistochemical, and molecular biological aspects.
Clinical Course and Prognosis
Most patients with systemic EBV-positive T-cell lymphoma have an aggressive clinical course with high mortality. In a few cases, patients were reported to respond to a regimen of etoposide and dexamethasone, followed by allogeneic hematopoietic stem cell transplantation.3
In recognition of the aggressive clinical behavior and desire to clearly distinguish systemic EBV-positive T-cell lymphoma from CAEBV infection, the older term systemic EBV-positive T-cell LPD of childhood, which had been introduced in 2008 to the World Health Organization classification, was changed to systemic EBV-positive T-cell lymphoma of childhood in the revised 2016 World Health Organization classification.6,12 However, Kim et al14 reported a case with excellent response to corticosteroid administration, suggesting that systemic EBV-positive T-cell lymphoma of childhood may be more heterogeneous in terms of prognosis.
Our patient presented with acute IM-like symptoms, including high fever, tonsillar enlargement, lymphadenopathy, and hepatosplenomegaly, as well as uncommon oral ulcers and skin lesions, including indurated nodules. Histopathologic changes in the skin nodule, proliferation in bone marrow, immunohistochemical phenotype, and positivity of EBER and TCR-γ monoclonal rearrangement were all consistent with systemic EBV-positive T-cell lymphoma of childhood. The patient was positive for VCA IgG and negative for VCA IgM, compatible with systemic EBV-positive T-cell lymphoma of childhood occurring shortly after IM. Neither pancytopenia, hemophagocytic syndrome, nor multiorgan failure occurred during the course.
Differential Diagnosis
It is important to distinguish IM from systemic EBV-positive T-cell lymphoma of childhood and CAEBV infection. Detection of anti–VCA IgM in the early stage, its disappearance during the clinical course, and appearance of anti-EBV–determined nuclear antigen is useful to distinguish IM from the neoplasms, as systemic EBV-positive T-cell lymphoma of childhood is negative for anti-EBV–determined nuclear antigen. Carefully following the clinical course also is important.3,15
Epstein-Barr virus–associated hemophagocytic lymphohistiocytosis can occur in association with systemic EBV-positive T-cell lymphoma of childhood and might represent a continuum of disease rather than distinct entities.14 The most useful marker for differentiating EBV-associated hemophagocytic lymphohistiocytosis and systemic EBV-positive T-cell lymphoma of childhood is an abnormal karyotype rather than molecular clonality.16
Outcome
Mortality risk in EBV-associated T-cell and NK-cell LPD is not primarily dependent on whether the lesion has progressed to lymphoma but instead is related to associated complications.17
Conclusion
Although systemic EBV-positive T-cell lymphoma of childhood is a rare disorder and has race predilection, dermatologists should be aware due to the aggressive clinical source and poor prognosis. Histopathology and in situ hybridization for EBER and TCR gene rearrangements are critical for final diagnosis. Although rare cases can show temporary resolution, the final outcome of this disease is not optimistic.
- Ameli F, Ghafourian F, Masir N. Systematic Epstein-Barr virus-positive T-cell lymphoproliferative disease presenting as a persistent fever and cough: a case report. J Med Case Rep. 2014;8:288.
- Kim HJ, Ko YH, Kim JE, et al. Epstein-Barr virus-associated lympho-proliferative disorders: review and update on 2016 WHO classification. J Pathol Transl Med. 2017;51:352-358.
- Dojcinov SD, Fend F, Quintanilla-Martinez L. EBV-positive lymphoproliferations of B- T- and NK-cell derivation in non-immunocompromised hosts [published online March 7, 2018]. Pathogens. doi:10.3390/pathogens7010028.
- Luzuriaga K, Sullivan JL. Infectious mononucleosis. N Engl J Med. 2010;362:1993-2000.
- Cohen JI, Kimura H, Nakamura S, et al. Epstein-Barr virus-associated lymphoproliferative disease in non-immunocompromised hosts: a status report and summary of an international meeting, 8-9 September 2008. Ann Oncol. 2009;20:1472-1482.
- Swerdlow SH, Campo E, Pileri SA, et al. The 2016 revision of the World Health Organization classification of lymphoid neoplasms. Blood. 2016;127:2375-2390.
- Kim WY, Montes-Mojarro IA, Fend F, et al. Epstein-Barr virus-associated T and NK-cell lymphoproliferative diseases. Front Pediatr. 2019;7:71.
- Hong M, Ko YH, Yoo KH, et al. EBV-positive T/NK-cell lymphoproliferative disease of childhood. Korean J Pathol. 2013;47:137-147.
- Quintanilla-Martinez L, Kumar S, Fend F, et al. Fulminant EBV(+) T-cell lymphoproliferative disorder following acute/chronic EBV infection: a distinct clinicopathologic syndrome. Blood. 2000;96:443-451.
- Chen G, Chen L, Qin X, et al. Systemic Epstein-Barr virus positive T-cell lymphoproliferative disease of childhood with hemophagocytic syndrome. Int J Clin Exp Pathol. 2014;7:7110-7113.
- Grywalska E, Rolinski J. Epstein-Barr virus-associated lymphomas. Semin Oncol. 2015;42:291-303.
- Huang W, Lv N, Ying J, et al. Clinicopathological characteristics of four cases of EBV positive T-cell lymphoproliferative disorders of childhood in China. Int J Clin Exp Pathol. 2014;7:4991-4999.
- Tabanelli V, Agostinelli C, Sabattini E, et al. Systemic Epstein-Barr-virus-positive T cell lymphoproliferative childhood disease in a 22-year-old Caucasian man: a case report and review of the literature. J Med Case Rep. 2011;5:218.
- Kim DH, Kim M, Kim Y, et al. Systemic Epstein-Barr virus-positive T-cell lymphoproliferative disease of childhood with good response to steroid therapy. J Pediatr Hematol Oncol. 2017;39:e497-e500.
- Arai A, Yamaguchi T, Komatsu H, et al. Infectious mononucleosis accompanied by clonal proliferation of EBV-infected cells and infection of CD8-positive cells. Int J Hematol. 2014;99:671-675.
- Smith MC, Cohen DN, Greig B, et al. The ambiguous boundary between EBV-related hemophagocytic lymphohistiocytosis and systemic EBV-driven T cell lymphoproliferative disorder. Int J Clin Exp Pathol. 2014;7:5738-5749.
- Paik JH, Choe JY, Kim H, et al. Clinicopathological categorization of Epstein-Barr virus-positive T/NK-cell lymphoproliferative disease: an analysis of 42 cases with an emphasis on prognostic implications. Leuk Lymphoma. 2017;58:53-63.
Case Report
A 7-year-old Chinese boy presented with multiple painful oral and tongue ulcers of 2 weeks’ duration as well as acute onset of moderate to high fever (highest temperature, 39.3°C) for 5 days. The fever was reported to have run a relapsing course, accompanied by rigors but without convulsions or cognitive changes. At times, the patient had nasal congestion, nasal discharge, and cough. He also had a transient eruption on the back and hands as well as an indurated red nodule on the left forearm.
Before the patient was hospitalized, antibiotic therapy was administered by other physicians, but the condition of fever and oral ulcers did not improve. After the patient was hospitalized, new tender nodules emerged on the scalp, buttocks, and lower extremities. New ulcers also appeared on the palate.
History
Two months earlier, the patient had presented with a painful perioral skin ulcer that resolved after being treated as contagious eczema. Another dermatologist previously had considered a diagnosis of hand-foot-and-mouth disease.
The patient was born by normal spontaneous vaginal delivery, without abnormality. He was breastfed; feeding, growth, and the developmental history showed no abnormality. He was the family’s eldest child, with a healthy brother and sister. There was no history of familial illness. He received bacillus Calmette-Guérin and poliomyelitis vaccines after birth; the rest of the vaccine history was unclear. There was no history of immunologic abnormality.
Physical Examination
A 1.5×1.5-cm, warm, red nodule with a central black crust was noted on the left forearm (Figure 1A). Several similar lesions were noted on the buttocks, scalp, and lower extremities. Multiple ulcers, as large as 1 cm, were present on the tongue, palate, and left angle of the mouth (Figure 1B). The pharynx was congested, and the tonsils were mildly enlarged. Multiple enlarged, movable, nontender lymph nodes could be palpated in the cervical basins, axillae, and groin. No purpura or ecchymosis was detected.

Laboratory Results
Laboratory testing revealed a normal total white blood cell count (4.26×109/L [reference range, 4.0–12.0×109/L]), with normal neutrophils (1.36×109/L [reference range, 1.32–7.90×109/L]), lymphocytes (2.77×109/L [reference range, 1.20–6.00×109/L]), and monocytes (0.13×109/L [reference range, 0.08–0.80×109/L]); a mildly decreased hemoglobin level (115 g/L [reference range, 120–160 g/L]); a normal platelet count (102×109/L [reference range, 100–380×109/L]); an elevated lactate dehydrogenase level (614 U/L [reference range, 110–330 U/L]); an elevated α-hydroxybutyrate dehydrogenase level (483 U/L [reference range, 120–270 U/L]); elevated prothrombin time (15.3 s [reference range, 9–14 s]); elevated activated partial thromboplastin time (59.8 s [reference range, 20.6–39.6 s]); and an elevated D-dimer level (1.51 mg/L [reference range, <0.73 mg/L]). In addition, autoantibody testing revealed a positive antinuclear antibody titer of 1:320 and a strong positive anti–Ro-52 level.
The peripheral blood lymphocyte classification demonstrated a prominent elevated percentage of T lymphocytes, with predominantly CD8+ cells (CD3, 94.87%; CD8, 71.57%; CD4, 24.98%; CD4:CD8 ratio, 0.35) and a diminished percentage of B lymphocytes and natural killer (NK) cells. Epstein-Barr virus (EBV) antibody testing was positive for anti–viral capsid antigen (VCA) IgG and negative for anti-VCA IgM.
Smears of the ulcer on the tongue demonstrated gram-positive cocci, gram-negative bacilli, and diplococci. Culture of sputum showed methicillin-resistant Staphylococcus aureus. Inspection for acid-fast bacilli in sputum yielded negative results 3 times. A purified protein derivative skin test for Mycobacterium tuberculosis infection was negative.
Imaging and Other Studies
Computed tomography of the chest and abdomen demonstrated 2 nodular opacities on the lower right lung; spotted opacities on the upper right lung; floccular opacities on the rest area of the lung; mild pleural effusion; enlargement of lymph nodes on the mediastinum, the bilateral hilum of the lung, and mesentery; and hepatosplenomegaly. Electrocardiography showed sinus tachycardia. Nasal cavity endoscopy showed sinusitis. Fundus examination showed vasculopathy of the left retina. A colonoscopy showed normal mucosa.
Histopathology
Biopsy of the nodule on the left arm showed dense, superficial to deep perivascular, periadnexal, perineural, and panniculitislike lymphoid infiltrates, as well as a sparse interstitial infiltrate with irregular and pleomorphic medium to large nuclei. Lymphoid cells showed mild epidermotropism, with tagging to the basal layer. Some vessel walls were infiltrated by similar cells (Figure 2). Infiltrative atypical lymphoid cells expressed CD3 and CD7 and were mostly CD8+, with a few CD4+ cells and most cells negative for CD5, CD20, CD30, CD56, and anaplastic lymphoma kinase. Cytotoxic markers granzyme B and T-cell intracellular antigen protein 1 were scattered positive. Immunostaining for Ki-67 protein highlighted an increased proliferative rate of 80% in malignant cells. In situ hybridization for EBV-encoded RNA (EBER) demonstrated EBV-positive atypical lymphoid cells (Figure 3). Analysis for T-cell receptor (TCR) γ gene rearrangement revealed a monoclonal pattern. Bone marrow aspirate showed proliferation of the 3 cell lines. The percentage of T lymphocytes was increased (20% of all nucleated cells). No hemophagocytic activity was found.


Diagnosis
A diagnosis of systemic EBV-positive T-cell lymphoma was made. Before the final diagnosis was made, the patient was treated by rheumatologists with antibiotics, antiviral drugs, nonsteroidal anti-inflammatory drugs, and other symptomatic treatments. Following antibiotic therapy, a sputum culture reverted to normal flora, the coagulation index (ie, prothrombin time, activated partial thromboplastin time) returned to normal, and the D-dimer level decreased to 1.19 mg/L.
The patient’s parents refused to accept chemotherapy for him. Instead, they chose herbal therapy only; 5 months later, they reported that all of his symptoms had resolved; however, the disease suddenly relapsed after another 7 months, with multiple skin nodules and fever. The patient died, even with chemotherapy in another hospital.
Comment
Prevalence and Presentation
Epstein-Barr virus is a ubiquitous γ-herpesvirus with tropism for B cells, affecting more than 90% of the adult population worldwide. In addition to infecting B cells, EBV is capable of infecting T and NK cells, leading to various EBV-related lymphoproliferative disorders (LPDs). The frequency and clinical presentation of infection varies based on the type of EBV-infected cells and the state of host immunity.1-3
Primary infection usually is asymptomatic and occurs early in life; when symptomatic, the disease usually presents as infectious mononucleosis (IM), characterized by polyclonal expansion of infected B cells and subsequent cytotoxic T-cell response. A diagnosis of EBV infection can be made by testing for specific IgM and IgG antibodies against VCA, early antigens, and EBV nuclear antigen proteins.3,4
Associated LPDs
Although most symptoms associated with IM resolve within weeks or months, persistent or recurrent IM-like symptoms or even lasting disease occasionally occur, particularly in children and young adults. This complication is known as chronic active EBV infection (CAEBV), frequently associated with EBV-infected T-cell or NK-cell proliferation, especially in East Asian populations.3,5
Epstein-Barr virus–positive T-cell and NK-cell LPDs of childhood include CAEBV infection of T-cell and NK-cell types and systemic EBV-positive T-cell lymphoma of childhood. The former includes hydroa vacciniforme–like LPD and severe mosquito bite allergy.3
Systemic EBV-Positive T-cell Lymphoma of Childhood
This entity occurs not only in children but also in adolescents and young adults. A fulminant illness characterized by clonal proliferation of EBV-infected cytotoxic T cells, it can develop shortly after primary EBV infection or is linked to CAEBV infection. The disorder is rare and has a racial predilection for Asian (ie, Japanese, Chinese, Korean) populations and indigenous populations of Mexico and Central and South America.6-8
Complications
Systemic EBV-positive T-cell lymphoma of childhood is often complicated by hemophagocytic syndrome, coagulopathy, sepsis, and multiorgan failure. Other signs and symptoms include high fever, rash, jaundice, diarrhea, pancytopenia, and hepatosplenomegaly. The liver, spleen, lymph nodes, and bone marrow are commonly involved, and the disease can involve skin, the heart, and the lungs.9,10
Diagnosis
When systemic EBV-positive T-cell lymphoma occurs shortly after IM, serology shows low or absent anti-VCA IgM and positive anti-VCA IgG. Infiltrating T cells usually are small and lack cytologic atypia; however, cases with pleomorphic, medium to large lymphoid cells, irregular nuclei, and frequent mitoses have been described. Hemophagocytosis can be seen in the liver, spleen, and bone marrow.3,11
The most typical phenotype of systemic EBV-positive T-cell lymphoma is CD2+CD3+CD8+CD20−CD56−, with expression of the cytotoxic granules known as T-cell intracellular antigen 1 and granzyme B. Rare cases of CD4+ and mixed CD4+/CD8+ phenotypes have been described, usually in the setting of CAEBV infection.3,12 Neoplastic cells have monoclonally rearranged TCR-γ genes and consistent EBER positivity with in situ hybridization.13 A final diagnosis is based on a comprehensive analysis of clinical, morphological, immunohistochemical, and molecular biological aspects.
Clinical Course and Prognosis
Most patients with systemic EBV-positive T-cell lymphoma have an aggressive clinical course with high mortality. In a few cases, patients were reported to respond to a regimen of etoposide and dexamethasone, followed by allogeneic hematopoietic stem cell transplantation.3
In recognition of the aggressive clinical behavior and desire to clearly distinguish systemic EBV-positive T-cell lymphoma from CAEBV infection, the older term systemic EBV-positive T-cell LPD of childhood, which had been introduced in 2008 to the World Health Organization classification, was changed to systemic EBV-positive T-cell lymphoma of childhood in the revised 2016 World Health Organization classification.6,12 However, Kim et al14 reported a case with excellent response to corticosteroid administration, suggesting that systemic EBV-positive T-cell lymphoma of childhood may be more heterogeneous in terms of prognosis.
Our patient presented with acute IM-like symptoms, including high fever, tonsillar enlargement, lymphadenopathy, and hepatosplenomegaly, as well as uncommon oral ulcers and skin lesions, including indurated nodules. Histopathologic changes in the skin nodule, proliferation in bone marrow, immunohistochemical phenotype, and positivity of EBER and TCR-γ monoclonal rearrangement were all consistent with systemic EBV-positive T-cell lymphoma of childhood. The patient was positive for VCA IgG and negative for VCA IgM, compatible with systemic EBV-positive T-cell lymphoma of childhood occurring shortly after IM. Neither pancytopenia, hemophagocytic syndrome, nor multiorgan failure occurred during the course.
Differential Diagnosis
It is important to distinguish IM from systemic EBV-positive T-cell lymphoma of childhood and CAEBV infection. Detection of anti–VCA IgM in the early stage, its disappearance during the clinical course, and appearance of anti-EBV–determined nuclear antigen is useful to distinguish IM from the neoplasms, as systemic EBV-positive T-cell lymphoma of childhood is negative for anti-EBV–determined nuclear antigen. Carefully following the clinical course also is important.3,15
Epstein-Barr virus–associated hemophagocytic lymphohistiocytosis can occur in association with systemic EBV-positive T-cell lymphoma of childhood and might represent a continuum of disease rather than distinct entities.14 The most useful marker for differentiating EBV-associated hemophagocytic lymphohistiocytosis and systemic EBV-positive T-cell lymphoma of childhood is an abnormal karyotype rather than molecular clonality.16
Outcome
Mortality risk in EBV-associated T-cell and NK-cell LPD is not primarily dependent on whether the lesion has progressed to lymphoma but instead is related to associated complications.17
Conclusion
Although systemic EBV-positive T-cell lymphoma of childhood is a rare disorder and has race predilection, dermatologists should be aware due to the aggressive clinical source and poor prognosis. Histopathology and in situ hybridization for EBER and TCR gene rearrangements are critical for final diagnosis. Although rare cases can show temporary resolution, the final outcome of this disease is not optimistic.
Case Report
A 7-year-old Chinese boy presented with multiple painful oral and tongue ulcers of 2 weeks’ duration as well as acute onset of moderate to high fever (highest temperature, 39.3°C) for 5 days. The fever was reported to have run a relapsing course, accompanied by rigors but without convulsions or cognitive changes. At times, the patient had nasal congestion, nasal discharge, and cough. He also had a transient eruption on the back and hands as well as an indurated red nodule on the left forearm.
Before the patient was hospitalized, antibiotic therapy was administered by other physicians, but the condition of fever and oral ulcers did not improve. After the patient was hospitalized, new tender nodules emerged on the scalp, buttocks, and lower extremities. New ulcers also appeared on the palate.
History
Two months earlier, the patient had presented with a painful perioral skin ulcer that resolved after being treated as contagious eczema. Another dermatologist previously had considered a diagnosis of hand-foot-and-mouth disease.
The patient was born by normal spontaneous vaginal delivery, without abnormality. He was breastfed; feeding, growth, and the developmental history showed no abnormality. He was the family’s eldest child, with a healthy brother and sister. There was no history of familial illness. He received bacillus Calmette-Guérin and poliomyelitis vaccines after birth; the rest of the vaccine history was unclear. There was no history of immunologic abnormality.
Physical Examination
A 1.5×1.5-cm, warm, red nodule with a central black crust was noted on the left forearm (Figure 1A). Several similar lesions were noted on the buttocks, scalp, and lower extremities. Multiple ulcers, as large as 1 cm, were present on the tongue, palate, and left angle of the mouth (Figure 1B). The pharynx was congested, and the tonsils were mildly enlarged. Multiple enlarged, movable, nontender lymph nodes could be palpated in the cervical basins, axillae, and groin. No purpura or ecchymosis was detected.

Laboratory Results
Laboratory testing revealed a normal total white blood cell count (4.26×109/L [reference range, 4.0–12.0×109/L]), with normal neutrophils (1.36×109/L [reference range, 1.32–7.90×109/L]), lymphocytes (2.77×109/L [reference range, 1.20–6.00×109/L]), and monocytes (0.13×109/L [reference range, 0.08–0.80×109/L]); a mildly decreased hemoglobin level (115 g/L [reference range, 120–160 g/L]); a normal platelet count (102×109/L [reference range, 100–380×109/L]); an elevated lactate dehydrogenase level (614 U/L [reference range, 110–330 U/L]); an elevated α-hydroxybutyrate dehydrogenase level (483 U/L [reference range, 120–270 U/L]); elevated prothrombin time (15.3 s [reference range, 9–14 s]); elevated activated partial thromboplastin time (59.8 s [reference range, 20.6–39.6 s]); and an elevated D-dimer level (1.51 mg/L [reference range, <0.73 mg/L]). In addition, autoantibody testing revealed a positive antinuclear antibody titer of 1:320 and a strong positive anti–Ro-52 level.
The peripheral blood lymphocyte classification demonstrated a prominent elevated percentage of T lymphocytes, with predominantly CD8+ cells (CD3, 94.87%; CD8, 71.57%; CD4, 24.98%; CD4:CD8 ratio, 0.35) and a diminished percentage of B lymphocytes and natural killer (NK) cells. Epstein-Barr virus (EBV) antibody testing was positive for anti–viral capsid antigen (VCA) IgG and negative for anti-VCA IgM.
Smears of the ulcer on the tongue demonstrated gram-positive cocci, gram-negative bacilli, and diplococci. Culture of sputum showed methicillin-resistant Staphylococcus aureus. Inspection for acid-fast bacilli in sputum yielded negative results 3 times. A purified protein derivative skin test for Mycobacterium tuberculosis infection was negative.
Imaging and Other Studies
Computed tomography of the chest and abdomen demonstrated 2 nodular opacities on the lower right lung; spotted opacities on the upper right lung; floccular opacities on the rest area of the lung; mild pleural effusion; enlargement of lymph nodes on the mediastinum, the bilateral hilum of the lung, and mesentery; and hepatosplenomegaly. Electrocardiography showed sinus tachycardia. Nasal cavity endoscopy showed sinusitis. Fundus examination showed vasculopathy of the left retina. A colonoscopy showed normal mucosa.
Histopathology
Biopsy of the nodule on the left arm showed dense, superficial to deep perivascular, periadnexal, perineural, and panniculitislike lymphoid infiltrates, as well as a sparse interstitial infiltrate with irregular and pleomorphic medium to large nuclei. Lymphoid cells showed mild epidermotropism, with tagging to the basal layer. Some vessel walls were infiltrated by similar cells (Figure 2). Infiltrative atypical lymphoid cells expressed CD3 and CD7 and were mostly CD8+, with a few CD4+ cells and most cells negative for CD5, CD20, CD30, CD56, and anaplastic lymphoma kinase. Cytotoxic markers granzyme B and T-cell intracellular antigen protein 1 were scattered positive. Immunostaining for Ki-67 protein highlighted an increased proliferative rate of 80% in malignant cells. In situ hybridization for EBV-encoded RNA (EBER) demonstrated EBV-positive atypical lymphoid cells (Figure 3). Analysis for T-cell receptor (TCR) γ gene rearrangement revealed a monoclonal pattern. Bone marrow aspirate showed proliferation of the 3 cell lines. The percentage of T lymphocytes was increased (20% of all nucleated cells). No hemophagocytic activity was found.


Diagnosis
A diagnosis of systemic EBV-positive T-cell lymphoma was made. Before the final diagnosis was made, the patient was treated by rheumatologists with antibiotics, antiviral drugs, nonsteroidal anti-inflammatory drugs, and other symptomatic treatments. Following antibiotic therapy, a sputum culture reverted to normal flora, the coagulation index (ie, prothrombin time, activated partial thromboplastin time) returned to normal, and the D-dimer level decreased to 1.19 mg/L.
The patient’s parents refused to accept chemotherapy for him. Instead, they chose herbal therapy only; 5 months later, they reported that all of his symptoms had resolved; however, the disease suddenly relapsed after another 7 months, with multiple skin nodules and fever. The patient died, even with chemotherapy in another hospital.
Comment
Prevalence and Presentation
Epstein-Barr virus is a ubiquitous γ-herpesvirus with tropism for B cells, affecting more than 90% of the adult population worldwide. In addition to infecting B cells, EBV is capable of infecting T and NK cells, leading to various EBV-related lymphoproliferative disorders (LPDs). The frequency and clinical presentation of infection varies based on the type of EBV-infected cells and the state of host immunity.1-3
Primary infection usually is asymptomatic and occurs early in life; when symptomatic, the disease usually presents as infectious mononucleosis (IM), characterized by polyclonal expansion of infected B cells and subsequent cytotoxic T-cell response. A diagnosis of EBV infection can be made by testing for specific IgM and IgG antibodies against VCA, early antigens, and EBV nuclear antigen proteins.3,4
Associated LPDs
Although most symptoms associated with IM resolve within weeks or months, persistent or recurrent IM-like symptoms or even lasting disease occasionally occur, particularly in children and young adults. This complication is known as chronic active EBV infection (CAEBV), frequently associated with EBV-infected T-cell or NK-cell proliferation, especially in East Asian populations.3,5
Epstein-Barr virus–positive T-cell and NK-cell LPDs of childhood include CAEBV infection of T-cell and NK-cell types and systemic EBV-positive T-cell lymphoma of childhood. The former includes hydroa vacciniforme–like LPD and severe mosquito bite allergy.3
Systemic EBV-Positive T-cell Lymphoma of Childhood
This entity occurs not only in children but also in adolescents and young adults. A fulminant illness characterized by clonal proliferation of EBV-infected cytotoxic T cells, it can develop shortly after primary EBV infection or is linked to CAEBV infection. The disorder is rare and has a racial predilection for Asian (ie, Japanese, Chinese, Korean) populations and indigenous populations of Mexico and Central and South America.6-8
Complications
Systemic EBV-positive T-cell lymphoma of childhood is often complicated by hemophagocytic syndrome, coagulopathy, sepsis, and multiorgan failure. Other signs and symptoms include high fever, rash, jaundice, diarrhea, pancytopenia, and hepatosplenomegaly. The liver, spleen, lymph nodes, and bone marrow are commonly involved, and the disease can involve skin, the heart, and the lungs.9,10
Diagnosis
When systemic EBV-positive T-cell lymphoma occurs shortly after IM, serology shows low or absent anti-VCA IgM and positive anti-VCA IgG. Infiltrating T cells usually are small and lack cytologic atypia; however, cases with pleomorphic, medium to large lymphoid cells, irregular nuclei, and frequent mitoses have been described. Hemophagocytosis can be seen in the liver, spleen, and bone marrow.3,11
The most typical phenotype of systemic EBV-positive T-cell lymphoma is CD2+CD3+CD8+CD20−CD56−, with expression of the cytotoxic granules known as T-cell intracellular antigen 1 and granzyme B. Rare cases of CD4+ and mixed CD4+/CD8+ phenotypes have been described, usually in the setting of CAEBV infection.3,12 Neoplastic cells have monoclonally rearranged TCR-γ genes and consistent EBER positivity with in situ hybridization.13 A final diagnosis is based on a comprehensive analysis of clinical, morphological, immunohistochemical, and molecular biological aspects.
Clinical Course and Prognosis
Most patients with systemic EBV-positive T-cell lymphoma have an aggressive clinical course with high mortality. In a few cases, patients were reported to respond to a regimen of etoposide and dexamethasone, followed by allogeneic hematopoietic stem cell transplantation.3
In recognition of the aggressive clinical behavior and desire to clearly distinguish systemic EBV-positive T-cell lymphoma from CAEBV infection, the older term systemic EBV-positive T-cell LPD of childhood, which had been introduced in 2008 to the World Health Organization classification, was changed to systemic EBV-positive T-cell lymphoma of childhood in the revised 2016 World Health Organization classification.6,12 However, Kim et al14 reported a case with excellent response to corticosteroid administration, suggesting that systemic EBV-positive T-cell lymphoma of childhood may be more heterogeneous in terms of prognosis.
Our patient presented with acute IM-like symptoms, including high fever, tonsillar enlargement, lymphadenopathy, and hepatosplenomegaly, as well as uncommon oral ulcers and skin lesions, including indurated nodules. Histopathologic changes in the skin nodule, proliferation in bone marrow, immunohistochemical phenotype, and positivity of EBER and TCR-γ monoclonal rearrangement were all consistent with systemic EBV-positive T-cell lymphoma of childhood. The patient was positive for VCA IgG and negative for VCA IgM, compatible with systemic EBV-positive T-cell lymphoma of childhood occurring shortly after IM. Neither pancytopenia, hemophagocytic syndrome, nor multiorgan failure occurred during the course.
Differential Diagnosis
It is important to distinguish IM from systemic EBV-positive T-cell lymphoma of childhood and CAEBV infection. Detection of anti–VCA IgM in the early stage, its disappearance during the clinical course, and appearance of anti-EBV–determined nuclear antigen is useful to distinguish IM from the neoplasms, as systemic EBV-positive T-cell lymphoma of childhood is negative for anti-EBV–determined nuclear antigen. Carefully following the clinical course also is important.3,15
Epstein-Barr virus–associated hemophagocytic lymphohistiocytosis can occur in association with systemic EBV-positive T-cell lymphoma of childhood and might represent a continuum of disease rather than distinct entities.14 The most useful marker for differentiating EBV-associated hemophagocytic lymphohistiocytosis and systemic EBV-positive T-cell lymphoma of childhood is an abnormal karyotype rather than molecular clonality.16
Outcome
Mortality risk in EBV-associated T-cell and NK-cell LPD is not primarily dependent on whether the lesion has progressed to lymphoma but instead is related to associated complications.17
Conclusion
Although systemic EBV-positive T-cell lymphoma of childhood is a rare disorder and has race predilection, dermatologists should be aware due to the aggressive clinical source and poor prognosis. Histopathology and in situ hybridization for EBER and TCR gene rearrangements are critical for final diagnosis. Although rare cases can show temporary resolution, the final outcome of this disease is not optimistic.
- Ameli F, Ghafourian F, Masir N. Systematic Epstein-Barr virus-positive T-cell lymphoproliferative disease presenting as a persistent fever and cough: a case report. J Med Case Rep. 2014;8:288.
- Kim HJ, Ko YH, Kim JE, et al. Epstein-Barr virus-associated lympho-proliferative disorders: review and update on 2016 WHO classification. J Pathol Transl Med. 2017;51:352-358.
- Dojcinov SD, Fend F, Quintanilla-Martinez L. EBV-positive lymphoproliferations of B- T- and NK-cell derivation in non-immunocompromised hosts [published online March 7, 2018]. Pathogens. doi:10.3390/pathogens7010028.
- Luzuriaga K, Sullivan JL. Infectious mononucleosis. N Engl J Med. 2010;362:1993-2000.
- Cohen JI, Kimura H, Nakamura S, et al. Epstein-Barr virus-associated lymphoproliferative disease in non-immunocompromised hosts: a status report and summary of an international meeting, 8-9 September 2008. Ann Oncol. 2009;20:1472-1482.
- Swerdlow SH, Campo E, Pileri SA, et al. The 2016 revision of the World Health Organization classification of lymphoid neoplasms. Blood. 2016;127:2375-2390.
- Kim WY, Montes-Mojarro IA, Fend F, et al. Epstein-Barr virus-associated T and NK-cell lymphoproliferative diseases. Front Pediatr. 2019;7:71.
- Hong M, Ko YH, Yoo KH, et al. EBV-positive T/NK-cell lymphoproliferative disease of childhood. Korean J Pathol. 2013;47:137-147.
- Quintanilla-Martinez L, Kumar S, Fend F, et al. Fulminant EBV(+) T-cell lymphoproliferative disorder following acute/chronic EBV infection: a distinct clinicopathologic syndrome. Blood. 2000;96:443-451.
- Chen G, Chen L, Qin X, et al. Systemic Epstein-Barr virus positive T-cell lymphoproliferative disease of childhood with hemophagocytic syndrome. Int J Clin Exp Pathol. 2014;7:7110-7113.
- Grywalska E, Rolinski J. Epstein-Barr virus-associated lymphomas. Semin Oncol. 2015;42:291-303.
- Huang W, Lv N, Ying J, et al. Clinicopathological characteristics of four cases of EBV positive T-cell lymphoproliferative disorders of childhood in China. Int J Clin Exp Pathol. 2014;7:4991-4999.
- Tabanelli V, Agostinelli C, Sabattini E, et al. Systemic Epstein-Barr-virus-positive T cell lymphoproliferative childhood disease in a 22-year-old Caucasian man: a case report and review of the literature. J Med Case Rep. 2011;5:218.
- Kim DH, Kim M, Kim Y, et al. Systemic Epstein-Barr virus-positive T-cell lymphoproliferative disease of childhood with good response to steroid therapy. J Pediatr Hematol Oncol. 2017;39:e497-e500.
- Arai A, Yamaguchi T, Komatsu H, et al. Infectious mononucleosis accompanied by clonal proliferation of EBV-infected cells and infection of CD8-positive cells. Int J Hematol. 2014;99:671-675.
- Smith MC, Cohen DN, Greig B, et al. The ambiguous boundary between EBV-related hemophagocytic lymphohistiocytosis and systemic EBV-driven T cell lymphoproliferative disorder. Int J Clin Exp Pathol. 2014;7:5738-5749.
- Paik JH, Choe JY, Kim H, et al. Clinicopathological categorization of Epstein-Barr virus-positive T/NK-cell lymphoproliferative disease: an analysis of 42 cases with an emphasis on prognostic implications. Leuk Lymphoma. 2017;58:53-63.
- Ameli F, Ghafourian F, Masir N. Systematic Epstein-Barr virus-positive T-cell lymphoproliferative disease presenting as a persistent fever and cough: a case report. J Med Case Rep. 2014;8:288.
- Kim HJ, Ko YH, Kim JE, et al. Epstein-Barr virus-associated lympho-proliferative disorders: review and update on 2016 WHO classification. J Pathol Transl Med. 2017;51:352-358.
- Dojcinov SD, Fend F, Quintanilla-Martinez L. EBV-positive lymphoproliferations of B- T- and NK-cell derivation in non-immunocompromised hosts [published online March 7, 2018]. Pathogens. doi:10.3390/pathogens7010028.
- Luzuriaga K, Sullivan JL. Infectious mononucleosis. N Engl J Med. 2010;362:1993-2000.
- Cohen JI, Kimura H, Nakamura S, et al. Epstein-Barr virus-associated lymphoproliferative disease in non-immunocompromised hosts: a status report and summary of an international meeting, 8-9 September 2008. Ann Oncol. 2009;20:1472-1482.
- Swerdlow SH, Campo E, Pileri SA, et al. The 2016 revision of the World Health Organization classification of lymphoid neoplasms. Blood. 2016;127:2375-2390.
- Kim WY, Montes-Mojarro IA, Fend F, et al. Epstein-Barr virus-associated T and NK-cell lymphoproliferative diseases. Front Pediatr. 2019;7:71.
- Hong M, Ko YH, Yoo KH, et al. EBV-positive T/NK-cell lymphoproliferative disease of childhood. Korean J Pathol. 2013;47:137-147.
- Quintanilla-Martinez L, Kumar S, Fend F, et al. Fulminant EBV(+) T-cell lymphoproliferative disorder following acute/chronic EBV infection: a distinct clinicopathologic syndrome. Blood. 2000;96:443-451.
- Chen G, Chen L, Qin X, et al. Systemic Epstein-Barr virus positive T-cell lymphoproliferative disease of childhood with hemophagocytic syndrome. Int J Clin Exp Pathol. 2014;7:7110-7113.
- Grywalska E, Rolinski J. Epstein-Barr virus-associated lymphomas. Semin Oncol. 2015;42:291-303.
- Huang W, Lv N, Ying J, et al. Clinicopathological characteristics of four cases of EBV positive T-cell lymphoproliferative disorders of childhood in China. Int J Clin Exp Pathol. 2014;7:4991-4999.
- Tabanelli V, Agostinelli C, Sabattini E, et al. Systemic Epstein-Barr-virus-positive T cell lymphoproliferative childhood disease in a 22-year-old Caucasian man: a case report and review of the literature. J Med Case Rep. 2011;5:218.
- Kim DH, Kim M, Kim Y, et al. Systemic Epstein-Barr virus-positive T-cell lymphoproliferative disease of childhood with good response to steroid therapy. J Pediatr Hematol Oncol. 2017;39:e497-e500.
- Arai A, Yamaguchi T, Komatsu H, et al. Infectious mononucleosis accompanied by clonal proliferation of EBV-infected cells and infection of CD8-positive cells. Int J Hematol. 2014;99:671-675.
- Smith MC, Cohen DN, Greig B, et al. The ambiguous boundary between EBV-related hemophagocytic lymphohistiocytosis and systemic EBV-driven T cell lymphoproliferative disorder. Int J Clin Exp Pathol. 2014;7:5738-5749.
- Paik JH, Choe JY, Kim H, et al. Clinicopathological categorization of Epstein-Barr virus-positive T/NK-cell lymphoproliferative disease: an analysis of 42 cases with an emphasis on prognostic implications. Leuk Lymphoma. 2017;58:53-63.
Practice Points
- Systemic Epstein-Barr virus (EBV)–positive T-cell lymphoma of childhood is a fulminant illness with a predilection for Asians and indigenous populations from Mexico and Central and South America. In most patients, the disease has an aggressive clinical course with high mortality.
- The disease often is complicated by hemophagocytic syndrome, coagulopathy, sepsis, and multiorgan failure. When these severe complications are absent, the prognosis might be better.
- In situ hybridization for EBV-encoded RNA and for T-cell receptor gene rearrangements is an important tool to establish the diagnosis as well as for treatment options and predicting the prognosis.
Seborrhea Herpeticum: Cutaneous Herpes Simplex Virus Infection Within Infantile Seborrheic Dermatitis
Classically, eczema herpeticum is associated with atopic dermatitis (AD), but it also has been previously reported in the setting of pemphigus vulgaris, Darier disease, ichthyosis vulgaris, burns, psoriasis, and irritant contact dermatitis.1,2 Descriptions of cutaneous herpes simplex virus (HSV) in the setting of seborrheic dermatitis are lacking.
Case Report
A 2-month-old infant boy who was otherwise healthy presented to the emergency department with a new rash on the scalp. Initially there were a few clusters of small fluid-filled lesions that evolved over several days into diffuse clusters covering the scalp and extending onto the forehead and upper chest (Figure). The patient’s medical history was notable for infantile seborrheic dermatitis and a family history of AD. His grandmother, who was his primary caretaker, had a recent history of herpes labialis.
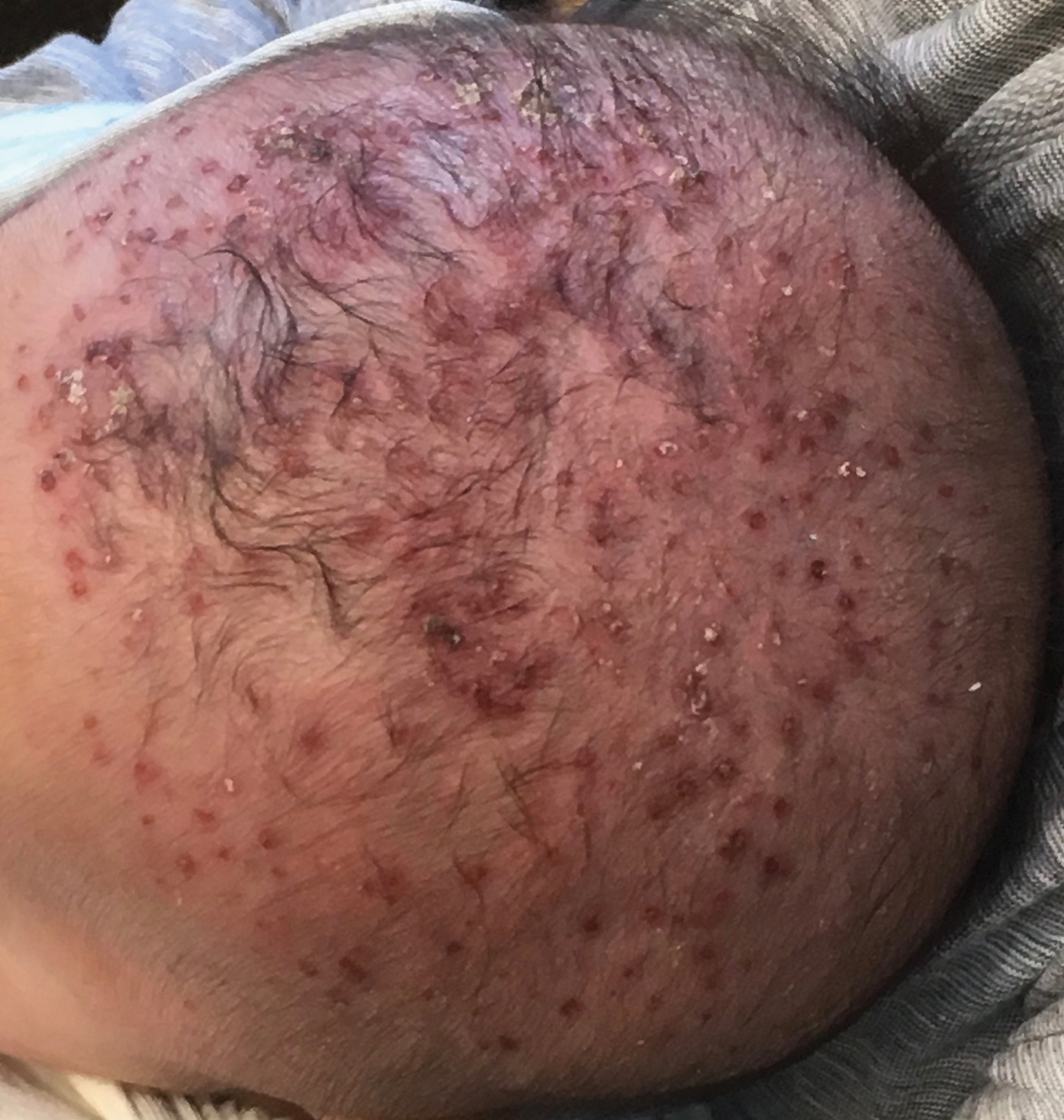
Physical examination revealed numerous discrete, erythematous, and punched-out erosions diffusely on the scalp. There were fewer similar erosions on the forehead and upper chest. There were no oral or periocular lesions. There were no areas of lichenification or eczematous plaques on the remainder of the trunk or extremities. Laboratory testing was positive for HSV type 1 polymerase chain reaction and positive for HSV type 1 viral culture. Liver enzymes were elevated with alanine aminotransferase at 107 U/L (reference range, 7–52 U/L) and aspartate aminotransferase at 94 U/L (reference range, 13–39 U/L).
The patient was admitted to the hospital and was treated by the dermatology and infectious disease services. Intravenous acyclovir 60 mg/kg daily was administered for 3 days until all lesions had crusted over. On the day of discharge, the patient was transitioned to oral valacyclovir 20 mg/kg daily for 7 days with resolution. One month later he developed a recurrence that was within his existing seborrheic dermatitis. After a repeat 7-day course of oral valacyclovir 20 mg/kg daily, he was placed on prophylaxis therapy of oral acyclovir 10 mg/kg daily. Gentle skin care precautions also were recommended.
Comment
Eczema herpeticum refers to disseminated cutaneous infection with HSV types 1 or 2 in the setting of underlying dermatosis.2 Although it is classically associated with AD, it has been reported in a number of other chronic skin disorders and can lead to serious complications, including hepatitis, keratoconjunctivitis, and meningitis. In those with AD who develop HSV, presentation may occur in active dermatitis locations because of skin barrier disruption, which may lead to increased susceptibility to viral infection.3
Herpes simplex virus in a background of seborrheic dermatitis has not been well described. Although the pathogenesis of seborrheic dermatitis has not been fully reported, several gene mutations and protein deficiencies have been identified in patients and animal models that are associated with immune response or epidermal differentiation.4 Therefore, it is possible that, as with AD, a disruption in the skin barrier increases susceptibility to viral infection.
It also has been suggested that infantile seborrheic dermatitis and AD represent the same spectrum of disease.5 Given our patient’s family history of AD, it is possible his presentation represents early underlying AD. Providers should be aware that cutaneous HSV can be confined to a seborrheic distribution and may represent underlying epidermal dysfunction secondary to seborrheic dermatitis.
- Wheeler CE, Abele DC. Eczema herpeticum, primary and recurrent. Arch Dermatol. 1966;93:162-173.
- Santmyire-Rosenberger BR, Nigra TP. Psoriasis herpeticum: three cases of Kaposi’s varicelliform eruption in psoriasis. J Am Acad Dermatol. 2005;53:52-56.
- Wollenberg A, Wetzel S, Burgdorf WH, et al. Viral infections in atopic dermatitis: pathogenic aspects and clinical management. J Allergy Clin Immunol. 2003;112:667-674.
- Karakadze M, Hirt P, Wikramanayake T. The genetic basis of seborrhoeic dermatitis: a review. J Eur Acad Dermatol Venereol. 2017;32:529-536.
- Alexopoulos A, Kakourou T, Orfanou I, et al. Retrospective analysis of the relationship between infantile seborrheic dermatitis and atopic dermatitis. Pediatr Dermatol. 2013;31:125-130.
Classically, eczema herpeticum is associated with atopic dermatitis (AD), but it also has been previously reported in the setting of pemphigus vulgaris, Darier disease, ichthyosis vulgaris, burns, psoriasis, and irritant contact dermatitis.1,2 Descriptions of cutaneous herpes simplex virus (HSV) in the setting of seborrheic dermatitis are lacking.
Case Report
A 2-month-old infant boy who was otherwise healthy presented to the emergency department with a new rash on the scalp. Initially there were a few clusters of small fluid-filled lesions that evolved over several days into diffuse clusters covering the scalp and extending onto the forehead and upper chest (Figure). The patient’s medical history was notable for infantile seborrheic dermatitis and a family history of AD. His grandmother, who was his primary caretaker, had a recent history of herpes labialis.

Physical examination revealed numerous discrete, erythematous, and punched-out erosions diffusely on the scalp. There were fewer similar erosions on the forehead and upper chest. There were no oral or periocular lesions. There were no areas of lichenification or eczematous plaques on the remainder of the trunk or extremities. Laboratory testing was positive for HSV type 1 polymerase chain reaction and positive for HSV type 1 viral culture. Liver enzymes were elevated with alanine aminotransferase at 107 U/L (reference range, 7–52 U/L) and aspartate aminotransferase at 94 U/L (reference range, 13–39 U/L).
The patient was admitted to the hospital and was treated by the dermatology and infectious disease services. Intravenous acyclovir 60 mg/kg daily was administered for 3 days until all lesions had crusted over. On the day of discharge, the patient was transitioned to oral valacyclovir 20 mg/kg daily for 7 days with resolution. One month later he developed a recurrence that was within his existing seborrheic dermatitis. After a repeat 7-day course of oral valacyclovir 20 mg/kg daily, he was placed on prophylaxis therapy of oral acyclovir 10 mg/kg daily. Gentle skin care precautions also were recommended.
Comment
Eczema herpeticum refers to disseminated cutaneous infection with HSV types 1 or 2 in the setting of underlying dermatosis.2 Although it is classically associated with AD, it has been reported in a number of other chronic skin disorders and can lead to serious complications, including hepatitis, keratoconjunctivitis, and meningitis. In those with AD who develop HSV, presentation may occur in active dermatitis locations because of skin barrier disruption, which may lead to increased susceptibility to viral infection.3
Herpes simplex virus in a background of seborrheic dermatitis has not been well described. Although the pathogenesis of seborrheic dermatitis has not been fully reported, several gene mutations and protein deficiencies have been identified in patients and animal models that are associated with immune response or epidermal differentiation.4 Therefore, it is possible that, as with AD, a disruption in the skin barrier increases susceptibility to viral infection.
It also has been suggested that infantile seborrheic dermatitis and AD represent the same spectrum of disease.5 Given our patient’s family history of AD, it is possible his presentation represents early underlying AD. Providers should be aware that cutaneous HSV can be confined to a seborrheic distribution and may represent underlying epidermal dysfunction secondary to seborrheic dermatitis.
Classically, eczema herpeticum is associated with atopic dermatitis (AD), but it also has been previously reported in the setting of pemphigus vulgaris, Darier disease, ichthyosis vulgaris, burns, psoriasis, and irritant contact dermatitis.1,2 Descriptions of cutaneous herpes simplex virus (HSV) in the setting of seborrheic dermatitis are lacking.
Case Report
A 2-month-old infant boy who was otherwise healthy presented to the emergency department with a new rash on the scalp. Initially there were a few clusters of small fluid-filled lesions that evolved over several days into diffuse clusters covering the scalp and extending onto the forehead and upper chest (Figure). The patient’s medical history was notable for infantile seborrheic dermatitis and a family history of AD. His grandmother, who was his primary caretaker, had a recent history of herpes labialis.

Physical examination revealed numerous discrete, erythematous, and punched-out erosions diffusely on the scalp. There were fewer similar erosions on the forehead and upper chest. There were no oral or periocular lesions. There were no areas of lichenification or eczematous plaques on the remainder of the trunk or extremities. Laboratory testing was positive for HSV type 1 polymerase chain reaction and positive for HSV type 1 viral culture. Liver enzymes were elevated with alanine aminotransferase at 107 U/L (reference range, 7–52 U/L) and aspartate aminotransferase at 94 U/L (reference range, 13–39 U/L).
The patient was admitted to the hospital and was treated by the dermatology and infectious disease services. Intravenous acyclovir 60 mg/kg daily was administered for 3 days until all lesions had crusted over. On the day of discharge, the patient was transitioned to oral valacyclovir 20 mg/kg daily for 7 days with resolution. One month later he developed a recurrence that was within his existing seborrheic dermatitis. After a repeat 7-day course of oral valacyclovir 20 mg/kg daily, he was placed on prophylaxis therapy of oral acyclovir 10 mg/kg daily. Gentle skin care precautions also were recommended.
Comment
Eczema herpeticum refers to disseminated cutaneous infection with HSV types 1 or 2 in the setting of underlying dermatosis.2 Although it is classically associated with AD, it has been reported in a number of other chronic skin disorders and can lead to serious complications, including hepatitis, keratoconjunctivitis, and meningitis. In those with AD who develop HSV, presentation may occur in active dermatitis locations because of skin barrier disruption, which may lead to increased susceptibility to viral infection.3
Herpes simplex virus in a background of seborrheic dermatitis has not been well described. Although the pathogenesis of seborrheic dermatitis has not been fully reported, several gene mutations and protein deficiencies have been identified in patients and animal models that are associated with immune response or epidermal differentiation.4 Therefore, it is possible that, as with AD, a disruption in the skin barrier increases susceptibility to viral infection.
It also has been suggested that infantile seborrheic dermatitis and AD represent the same spectrum of disease.5 Given our patient’s family history of AD, it is possible his presentation represents early underlying AD. Providers should be aware that cutaneous HSV can be confined to a seborrheic distribution and may represent underlying epidermal dysfunction secondary to seborrheic dermatitis.
- Wheeler CE, Abele DC. Eczema herpeticum, primary and recurrent. Arch Dermatol. 1966;93:162-173.
- Santmyire-Rosenberger BR, Nigra TP. Psoriasis herpeticum: three cases of Kaposi’s varicelliform eruption in psoriasis. J Am Acad Dermatol. 2005;53:52-56.
- Wollenberg A, Wetzel S, Burgdorf WH, et al. Viral infections in atopic dermatitis: pathogenic aspects and clinical management. J Allergy Clin Immunol. 2003;112:667-674.
- Karakadze M, Hirt P, Wikramanayake T. The genetic basis of seborrhoeic dermatitis: a review. J Eur Acad Dermatol Venereol. 2017;32:529-536.
- Alexopoulos A, Kakourou T, Orfanou I, et al. Retrospective analysis of the relationship between infantile seborrheic dermatitis and atopic dermatitis. Pediatr Dermatol. 2013;31:125-130.
- Wheeler CE, Abele DC. Eczema herpeticum, primary and recurrent. Arch Dermatol. 1966;93:162-173.
- Santmyire-Rosenberger BR, Nigra TP. Psoriasis herpeticum: three cases of Kaposi’s varicelliform eruption in psoriasis. J Am Acad Dermatol. 2005;53:52-56.
- Wollenberg A, Wetzel S, Burgdorf WH, et al. Viral infections in atopic dermatitis: pathogenic aspects and clinical management. J Allergy Clin Immunol. 2003;112:667-674.
- Karakadze M, Hirt P, Wikramanayake T. The genetic basis of seborrhoeic dermatitis: a review. J Eur Acad Dermatol Venereol. 2017;32:529-536.
- Alexopoulos A, Kakourou T, Orfanou I, et al. Retrospective analysis of the relationship between infantile seborrheic dermatitis and atopic dermatitis. Pediatr Dermatol. 2013;31:125-130.
Practice Points
- Cutaneous herpes simplex virus may present in a seborrheic distribution within infantile seborrheic dermatitis, suggesting underlying dysfunction secondary to seborrheic dermatitis.
- Treatment of seborrhea herpeticum involves antiviral therapy to treat the secondary viral infection and gentle skin care precautions for the primary condition.