User login
Pediatric lung disease plus nighttime screen time impact sleep quality
DALLAS – but nighttime use of technology may contribute more to sleep problems, according to a new study.
“Routinely addressing sleep concerns, sleep hygiene, and mental health is important in the care of pediatric patients with chronic illness,” concluded Lauren Greenawald, DO, and colleagues at the Alfred I. duPont Hospital for Children in Wilmington, Del. The researchers presented their findings on sleep quality and mental health of children with asthma or cystic fibrosis (CF) at the American Thoracic Society’s international conference.
Dr. Greenawald’s team screened 31 children (aged 7-17 years) with CF and 34 children with asthma for anxiety, depression, and ADHD. The researchers also assessed the children’s sleep hygiene, sleep quality, and physical and emotional symptoms. Instruments included the validated Pediatric Daytime Sleepiness Scale (PDSS), Pediatric Quality of Life Inventory, and Patient-Reported Outcomes Measurement Information System Pediatric Anxiety Survey, plus an investigator-designed survey about sleep habits.
Just over half the children with CF (52%) and 14% of children with asthma had mental health diagnoses (P less than .01). The same proportion of patients with CF (52%) and nearly a third of patients with asthma (30%) reported they often or always felt they needed more sleep based on the PDSS. Further, 42% of children with CF and 55% of children with asthma said their symptoms kept them awake 1-2 nights a week. Only 6% of asthma patients and no CF patients said their symptoms keep them awake often, 3-4 nights a week. Just over a third of children with CF (36%) and 46% of those with asthma thought they would sleep better if they didn’t have a medical condition.
Yet, for the vast majority of children, the sleeping problems did not appear to result from worry about their illness: 85% of those with CF and nearly all of those with asthma (97%) did not have trouble sleeping as a result of anxiety about their medical condition.
The researchers identified nighttime use of technology that may affect the children’s sleep in ways similar to that of the general population. Many of the participants – 68% of those with CF and 47% of those with asthma – reported texting or using social media or other technology an hour before going to bed. In addition, 55% of those with CF and 25% of those with asthma said they use their phone after the lights are out at least 5 nights a week. One in five of those with CF (20%) said they go to bed later than they planned at least 5 days a week because of social media or texting, though only 6% of those with asthma said the same.
Despite the children’s reports of inadequate sleep, very few – 3.2% of children with CF and 5.9% of children with asthma – reported feeling low daytime energy.
The use of child self-reporting in the presence of family members is a study limitation, including potentially introducing social desirability bias.
The research was funded by the Nemours Summer Undergraduate Research Program. The authors reported no disclosures.
SOURCE: Greenawald L et al. ATS 2019, Abstract A2788.
DALLAS – but nighttime use of technology may contribute more to sleep problems, according to a new study.
“Routinely addressing sleep concerns, sleep hygiene, and mental health is important in the care of pediatric patients with chronic illness,” concluded Lauren Greenawald, DO, and colleagues at the Alfred I. duPont Hospital for Children in Wilmington, Del. The researchers presented their findings on sleep quality and mental health of children with asthma or cystic fibrosis (CF) at the American Thoracic Society’s international conference.
Dr. Greenawald’s team screened 31 children (aged 7-17 years) with CF and 34 children with asthma for anxiety, depression, and ADHD. The researchers also assessed the children’s sleep hygiene, sleep quality, and physical and emotional symptoms. Instruments included the validated Pediatric Daytime Sleepiness Scale (PDSS), Pediatric Quality of Life Inventory, and Patient-Reported Outcomes Measurement Information System Pediatric Anxiety Survey, plus an investigator-designed survey about sleep habits.
Just over half the children with CF (52%) and 14% of children with asthma had mental health diagnoses (P less than .01). The same proportion of patients with CF (52%) and nearly a third of patients with asthma (30%) reported they often or always felt they needed more sleep based on the PDSS. Further, 42% of children with CF and 55% of children with asthma said their symptoms kept them awake 1-2 nights a week. Only 6% of asthma patients and no CF patients said their symptoms keep them awake often, 3-4 nights a week. Just over a third of children with CF (36%) and 46% of those with asthma thought they would sleep better if they didn’t have a medical condition.
Yet, for the vast majority of children, the sleeping problems did not appear to result from worry about their illness: 85% of those with CF and nearly all of those with asthma (97%) did not have trouble sleeping as a result of anxiety about their medical condition.
The researchers identified nighttime use of technology that may affect the children’s sleep in ways similar to that of the general population. Many of the participants – 68% of those with CF and 47% of those with asthma – reported texting or using social media or other technology an hour before going to bed. In addition, 55% of those with CF and 25% of those with asthma said they use their phone after the lights are out at least 5 nights a week. One in five of those with CF (20%) said they go to bed later than they planned at least 5 days a week because of social media or texting, though only 6% of those with asthma said the same.
Despite the children’s reports of inadequate sleep, very few – 3.2% of children with CF and 5.9% of children with asthma – reported feeling low daytime energy.
The use of child self-reporting in the presence of family members is a study limitation, including potentially introducing social desirability bias.
The research was funded by the Nemours Summer Undergraduate Research Program. The authors reported no disclosures.
SOURCE: Greenawald L et al. ATS 2019, Abstract A2788.
DALLAS – but nighttime use of technology may contribute more to sleep problems, according to a new study.
“Routinely addressing sleep concerns, sleep hygiene, and mental health is important in the care of pediatric patients with chronic illness,” concluded Lauren Greenawald, DO, and colleagues at the Alfred I. duPont Hospital for Children in Wilmington, Del. The researchers presented their findings on sleep quality and mental health of children with asthma or cystic fibrosis (CF) at the American Thoracic Society’s international conference.
Dr. Greenawald’s team screened 31 children (aged 7-17 years) with CF and 34 children with asthma for anxiety, depression, and ADHD. The researchers also assessed the children’s sleep hygiene, sleep quality, and physical and emotional symptoms. Instruments included the validated Pediatric Daytime Sleepiness Scale (PDSS), Pediatric Quality of Life Inventory, and Patient-Reported Outcomes Measurement Information System Pediatric Anxiety Survey, plus an investigator-designed survey about sleep habits.
Just over half the children with CF (52%) and 14% of children with asthma had mental health diagnoses (P less than .01). The same proportion of patients with CF (52%) and nearly a third of patients with asthma (30%) reported they often or always felt they needed more sleep based on the PDSS. Further, 42% of children with CF and 55% of children with asthma said their symptoms kept them awake 1-2 nights a week. Only 6% of asthma patients and no CF patients said their symptoms keep them awake often, 3-4 nights a week. Just over a third of children with CF (36%) and 46% of those with asthma thought they would sleep better if they didn’t have a medical condition.
Yet, for the vast majority of children, the sleeping problems did not appear to result from worry about their illness: 85% of those with CF and nearly all of those with asthma (97%) did not have trouble sleeping as a result of anxiety about their medical condition.
The researchers identified nighttime use of technology that may affect the children’s sleep in ways similar to that of the general population. Many of the participants – 68% of those with CF and 47% of those with asthma – reported texting or using social media or other technology an hour before going to bed. In addition, 55% of those with CF and 25% of those with asthma said they use their phone after the lights are out at least 5 nights a week. One in five of those with CF (20%) said they go to bed later than they planned at least 5 days a week because of social media or texting, though only 6% of those with asthma said the same.
Despite the children’s reports of inadequate sleep, very few – 3.2% of children with CF and 5.9% of children with asthma – reported feeling low daytime energy.
The use of child self-reporting in the presence of family members is a study limitation, including potentially introducing social desirability bias.
The research was funded by the Nemours Summer Undergraduate Research Program. The authors reported no disclosures.
SOURCE: Greenawald L et al. ATS 2019, Abstract A2788.
REPORTING FROM ATS 2019
Refrigerator-stable varicella vaccine held safe and effective
profiles when administered concomitantly with MMR vaccine, according to a study in Vaccine.
In this double-blind, controlled, multicenter study, Keith S. Reisinger of Primary Physicians Research, Pittsburgh, and his colleagues randomized 958 subjects aged 12-23 months to receive either 8,000 plaque-forming units (PFU) refrigerated vaccine (n = 320; group 1), 25,000 PFU refrigerated vaccine (n = 315; group 2), or 10,000 PFU frozen vaccine (n = 323; group 3), and subjects in all three groups also received MMR vaccine. The primary endpoint for immunogenicity was percentage of subjects with at least 5 titers of varicella antibody according to glycoprotein enzyme-linked immunosorbent assay among the three groups 6 weeks post vaccination; the primary safety endpoint was incidences of vaccine-related adverse events during days 0-42 post vaccination.
The percentages of subjects meeting the primary endpoint for immunogenicity were comparable among the groups, at 93%, 94%, and 95% for groups 1, 2, and 3, respectively. Results for the safety endpoints also were similar among the groups; for example, rates of injection-site adverse events or vaccine-related injection-site adverse events were 44%, 40%, and 43%, with no statistically significant between-group differences.
The study authors noted that one of the problems with having only frozen formulations is how that limits availability in parts of the world where only refrigeration of 2°C–8°C is available. “Use of a refrigerator-stable formulation of varicella vaccine will allow for increased availability of the product throughout the world and may help to increase vaccination rates against varicella,” they concluded.
Strengths of the study included its head-to-head design. Limitations included how adverse events were based on parental reporting.
Some authors reported relationships, including employment, with Merck & Co, which developed the vaccine in this study and funded the study.
SOURCE: Reisinger KS et al. Vaccine. 2018 Aug 23. doi: 10.1016/j.vaccine.2018.01.089.
profiles when administered concomitantly with MMR vaccine, according to a study in Vaccine.
In this double-blind, controlled, multicenter study, Keith S. Reisinger of Primary Physicians Research, Pittsburgh, and his colleagues randomized 958 subjects aged 12-23 months to receive either 8,000 plaque-forming units (PFU) refrigerated vaccine (n = 320; group 1), 25,000 PFU refrigerated vaccine (n = 315; group 2), or 10,000 PFU frozen vaccine (n = 323; group 3), and subjects in all three groups also received MMR vaccine. The primary endpoint for immunogenicity was percentage of subjects with at least 5 titers of varicella antibody according to glycoprotein enzyme-linked immunosorbent assay among the three groups 6 weeks post vaccination; the primary safety endpoint was incidences of vaccine-related adverse events during days 0-42 post vaccination.
The percentages of subjects meeting the primary endpoint for immunogenicity were comparable among the groups, at 93%, 94%, and 95% for groups 1, 2, and 3, respectively. Results for the safety endpoints also were similar among the groups; for example, rates of injection-site adverse events or vaccine-related injection-site adverse events were 44%, 40%, and 43%, with no statistically significant between-group differences.
The study authors noted that one of the problems with having only frozen formulations is how that limits availability in parts of the world where only refrigeration of 2°C–8°C is available. “Use of a refrigerator-stable formulation of varicella vaccine will allow for increased availability of the product throughout the world and may help to increase vaccination rates against varicella,” they concluded.
Strengths of the study included its head-to-head design. Limitations included how adverse events were based on parental reporting.
Some authors reported relationships, including employment, with Merck & Co, which developed the vaccine in this study and funded the study.
SOURCE: Reisinger KS et al. Vaccine. 2018 Aug 23. doi: 10.1016/j.vaccine.2018.01.089.
profiles when administered concomitantly with MMR vaccine, according to a study in Vaccine.
In this double-blind, controlled, multicenter study, Keith S. Reisinger of Primary Physicians Research, Pittsburgh, and his colleagues randomized 958 subjects aged 12-23 months to receive either 8,000 plaque-forming units (PFU) refrigerated vaccine (n = 320; group 1), 25,000 PFU refrigerated vaccine (n = 315; group 2), or 10,000 PFU frozen vaccine (n = 323; group 3), and subjects in all three groups also received MMR vaccine. The primary endpoint for immunogenicity was percentage of subjects with at least 5 titers of varicella antibody according to glycoprotein enzyme-linked immunosorbent assay among the three groups 6 weeks post vaccination; the primary safety endpoint was incidences of vaccine-related adverse events during days 0-42 post vaccination.
The percentages of subjects meeting the primary endpoint for immunogenicity were comparable among the groups, at 93%, 94%, and 95% for groups 1, 2, and 3, respectively. Results for the safety endpoints also were similar among the groups; for example, rates of injection-site adverse events or vaccine-related injection-site adverse events were 44%, 40%, and 43%, with no statistically significant between-group differences.
The study authors noted that one of the problems with having only frozen formulations is how that limits availability in parts of the world where only refrigeration of 2°C–8°C is available. “Use of a refrigerator-stable formulation of varicella vaccine will allow for increased availability of the product throughout the world and may help to increase vaccination rates against varicella,” they concluded.
Strengths of the study included its head-to-head design. Limitations included how adverse events were based on parental reporting.
Some authors reported relationships, including employment, with Merck & Co, which developed the vaccine in this study and funded the study.
SOURCE: Reisinger KS et al. Vaccine. 2018 Aug 23. doi: 10.1016/j.vaccine.2018.01.089.
Children’s book effectively assesses literary skills during well-child visits
reported John S. Hutton, MS, MD, of Cincinnati Children’s Hospital Medical Center, and his associates.
The Reading House (TRH) is a 14-page, full-color, board book with a simple, rhyming narrative and illustrated content showing children of various ethnicities and sexes going about their day. For the study, published in Pediatrics, 278 children aged 36-52 months (mean age, 43.1 months) were recruited from seven pediatric primary care clinics, two of which were affiliated with an academic children’s hospital primarily serving families of lower socioeconomic status. The children’s reading comprehension was measured by way of a 9-item TRH assessment, as well as the 25-item Get Ready to Read! (GRTR) validated measure; parent, child, and provider impressions of TRH also were collected.
The mean TRH assessment score was 4.2, and the mean GRTR score was 11.1. The TRH score was positively associated with GRTR score, female sex, private practice, and child age (Pediatrics 2019 May 30. doi: 10.1542/peds.2018-3843).
Of the 72 clinical providers surveyed on the effectiveness of the TRH assessment, most reported that the assessment was not invasive in patient flow (93% not at all, 6% somewhat, 1% very much), that TRH would be feasible to administer (49% yes, 43% not sure, 8% no), would be clinically useful (67% yes, 31% not sure, 2% no), and would be useful for families (85% yes, 14% not sure, 1% no). Similar results on the effectiveness and enjoyability of TRH were reported by parents and children.
“Although psychometric properties are critical, effective screening should be perceived as useful and not burdensome or invasive. Responses to parent, child, and provider surveys were favorable, which suggests that TRH screening may be an enjoyable and valuable addition to well-child visits,” the investigators wrote.
Dr. Hutton conceived, wrote, and edited the children’s book used in the study, and is the founder of the company that published the book, although he receives no salary or compensation for this role. The book’s intended use is as a screening tool, distributed at low cost to clinical practices and organizations. The other study authors did not report any conflicts of interest.
reported John S. Hutton, MS, MD, of Cincinnati Children’s Hospital Medical Center, and his associates.
The Reading House (TRH) is a 14-page, full-color, board book with a simple, rhyming narrative and illustrated content showing children of various ethnicities and sexes going about their day. For the study, published in Pediatrics, 278 children aged 36-52 months (mean age, 43.1 months) were recruited from seven pediatric primary care clinics, two of which were affiliated with an academic children’s hospital primarily serving families of lower socioeconomic status. The children’s reading comprehension was measured by way of a 9-item TRH assessment, as well as the 25-item Get Ready to Read! (GRTR) validated measure; parent, child, and provider impressions of TRH also were collected.
The mean TRH assessment score was 4.2, and the mean GRTR score was 11.1. The TRH score was positively associated with GRTR score, female sex, private practice, and child age (Pediatrics 2019 May 30. doi: 10.1542/peds.2018-3843).
Of the 72 clinical providers surveyed on the effectiveness of the TRH assessment, most reported that the assessment was not invasive in patient flow (93% not at all, 6% somewhat, 1% very much), that TRH would be feasible to administer (49% yes, 43% not sure, 8% no), would be clinically useful (67% yes, 31% not sure, 2% no), and would be useful for families (85% yes, 14% not sure, 1% no). Similar results on the effectiveness and enjoyability of TRH were reported by parents and children.
“Although psychometric properties are critical, effective screening should be perceived as useful and not burdensome or invasive. Responses to parent, child, and provider surveys were favorable, which suggests that TRH screening may be an enjoyable and valuable addition to well-child visits,” the investigators wrote.
Dr. Hutton conceived, wrote, and edited the children’s book used in the study, and is the founder of the company that published the book, although he receives no salary or compensation for this role. The book’s intended use is as a screening tool, distributed at low cost to clinical practices and organizations. The other study authors did not report any conflicts of interest.
reported John S. Hutton, MS, MD, of Cincinnati Children’s Hospital Medical Center, and his associates.
The Reading House (TRH) is a 14-page, full-color, board book with a simple, rhyming narrative and illustrated content showing children of various ethnicities and sexes going about their day. For the study, published in Pediatrics, 278 children aged 36-52 months (mean age, 43.1 months) were recruited from seven pediatric primary care clinics, two of which were affiliated with an academic children’s hospital primarily serving families of lower socioeconomic status. The children’s reading comprehension was measured by way of a 9-item TRH assessment, as well as the 25-item Get Ready to Read! (GRTR) validated measure; parent, child, and provider impressions of TRH also were collected.
The mean TRH assessment score was 4.2, and the mean GRTR score was 11.1. The TRH score was positively associated with GRTR score, female sex, private practice, and child age (Pediatrics 2019 May 30. doi: 10.1542/peds.2018-3843).
Of the 72 clinical providers surveyed on the effectiveness of the TRH assessment, most reported that the assessment was not invasive in patient flow (93% not at all, 6% somewhat, 1% very much), that TRH would be feasible to administer (49% yes, 43% not sure, 8% no), would be clinically useful (67% yes, 31% not sure, 2% no), and would be useful for families (85% yes, 14% not sure, 1% no). Similar results on the effectiveness and enjoyability of TRH were reported by parents and children.
“Although psychometric properties are critical, effective screening should be perceived as useful and not burdensome or invasive. Responses to parent, child, and provider surveys were favorable, which suggests that TRH screening may be an enjoyable and valuable addition to well-child visits,” the investigators wrote.
Dr. Hutton conceived, wrote, and edited the children’s book used in the study, and is the founder of the company that published the book, although he receives no salary or compensation for this role. The book’s intended use is as a screening tool, distributed at low cost to clinical practices and organizations. The other study authors did not report any conflicts of interest.
FROM PEDIATRICS
HPV vaccine: Is one dose enough?
LJUBLJANA, SLOVENIA – There is good news and bad news about human papillomavirus (HPV) vaccination as a means of preventing cervical cancer.
The bad news is the HPV vaccines are projected to be in short supply, unable to meet global demand until at least 2024. The good news is that – in one study, for 11 years and counting – which would effectively double the existing supply, Aimee R. Kreimer, PhD, said at the annual meeting of the European Society for Paediatric Infectious Diseases.
These data come from post hoc analyses of major phase 3 randomized controlled trials of bivalent HPV vaccine in Costa Rica and quadrivalent vaccine in India. However, these secondary analyses aren’t considered rock solid evidence because the subjects who got a single dose weren’t randomized to that strategy, they simply for one reason or another didn’t receive the recommended additional dose or doses.
“I don’t know if these studies are enough, so several studies have been launched over the past couple of years with an eye toward generating the quality of data that would be sufficient to motivate policy change, if in fact one dose is proven to be effective,” said Dr. Kreimer, a senior scientist at the National Cancer Institute in Bethesda, Md.
The first of these formal randomized, controlled trials – a delayed second-dose study in 9- to 11-year-old U.S. boys and girls – is due to be completed next year. Four other trials ongoing in Africa and Costa Rica, all in females, are expected to report findings in 2022-2025.
Dr. Kreimer is first author of a soon-to-be-published 11-year update from the phase 3 Costa Rica HPV Vaccine Trial, which was launched prior to licensure of the GlaxoSmithKline bivalent HPV vaccine. Previous analyses showed that at both 4 and 7 years of follow-up, a single dose of the vaccine was as effective as two or three in preventing infection with HPV types 16 and 18, which are covered by the vaccine.
“Now the research question has transitioned to, ‘Will one dose be sufficiently durable?’ she explained.
The answer from this study is yes. At 11 years since receipt of the bivalent HPV vaccine, there was no difference in terms of prevalent HPV 16/18 infection between the one-, two-, and three-dose groups. To address the issue of possible selection bias in this post hoc nonrandomized comparison, Dr. Kreimer and her coinvestigators looked at rates of infection with HPV 31 and 45, which aren’t covered by the vaccine. The rates were similar regardless of the number of vaccine doses received 11 years earlier, indicating women in all three dosing groups are at similar risk for acquiring HPV infection, thus bolstering the legitimacy of the conclusion that one dose provides effective long-term protection.
Intriguingly, HPV serum antibody levels in the single-dose group have remained stable for 11 years at a level that’s only about one-quarter of that associated with three doses of the vaccine, albeit an order of magnitude greater than the level induced by natural immunity.
“This really challenges the dogma of the HPV vaccine,” according to Dr. Kreimer. “It suggests that inferior [HPV] antibodies do not necessarily mean inferior protection.”
The explanation for this phenomenon appears to be that HPV subunit vaccine mimics the shell of authentic virions so well that the immune system sees it as dangerous and mounts long-term antibody production. Also, cervical infection by HPV is a relatively slow process, allowing time for vaccine-induced antibodies to interrupt it, she said.
In contrast to the encouraging findings from this post hoc analysis and another from a phase 3 trial of quadrivalent vaccine in India, numerous phase 4 vaccine effectiveness monitoring studies have shown markedly lower vaccine effectiveness for one dose of HPV vaccine. Dr. Kreimer cautioned that this is a flawed conclusion attributable to a methodologic artifact whereby the investigators have lumped together single-dose recipients who were 17 years old or more at the time with those who were younger.
“The problem is that many people who are aged 17-18 years already have HPV infection, so when they are vaccinated it shows up as a vaccine failure. That’s not correct. These are prophylactic HPV vaccines. They’re not meant to help clear an infection,” she noted.
Stepping back, Dr. Kreimer observed that cervical cancer “is really a story of inequality.” Indeed, 90% of cervical cancers occur in low-income countries, where HPV vaccination uptake remains very low even more than a decade after licensure. When modelers project out in the future, they estimate that at current HPV vaccination levels in Sub-Saharan Africa, which has the highest cervical cancer rates in the world, it would take more than 100 years to achieve the World Health Organization goal of eliminating the malignancy.
Asked by an audience member how low a single-dose vaccine effectiveness level she considers acceptable to help reach the goal of eliminating cervical cancer in developing countries, Dr. Kreimer cautioned against the tendency to let ‘perfect’ become the enemy of ‘good.’
“I’ll remind everyone that, in this moment, very few of the target girls in the lower– and upper-lower–income countries are getting any vaccination. So I don’t think it’s a question of whether we should be going from two to one dose, I think it’s really a question of, for those who are at zero doses, how do we get them one dose? And with the HPV vaccine, we’ve even seen suggestions of herd immunity if we have 50% uptake,” she replied.
Dr. Kreimer reported having no financial conflicts regarding her presentation.
LJUBLJANA, SLOVENIA – There is good news and bad news about human papillomavirus (HPV) vaccination as a means of preventing cervical cancer.
The bad news is the HPV vaccines are projected to be in short supply, unable to meet global demand until at least 2024. The good news is that – in one study, for 11 years and counting – which would effectively double the existing supply, Aimee R. Kreimer, PhD, said at the annual meeting of the European Society for Paediatric Infectious Diseases.
These data come from post hoc analyses of major phase 3 randomized controlled trials of bivalent HPV vaccine in Costa Rica and quadrivalent vaccine in India. However, these secondary analyses aren’t considered rock solid evidence because the subjects who got a single dose weren’t randomized to that strategy, they simply for one reason or another didn’t receive the recommended additional dose or doses.
“I don’t know if these studies are enough, so several studies have been launched over the past couple of years with an eye toward generating the quality of data that would be sufficient to motivate policy change, if in fact one dose is proven to be effective,” said Dr. Kreimer, a senior scientist at the National Cancer Institute in Bethesda, Md.
The first of these formal randomized, controlled trials – a delayed second-dose study in 9- to 11-year-old U.S. boys and girls – is due to be completed next year. Four other trials ongoing in Africa and Costa Rica, all in females, are expected to report findings in 2022-2025.
Dr. Kreimer is first author of a soon-to-be-published 11-year update from the phase 3 Costa Rica HPV Vaccine Trial, which was launched prior to licensure of the GlaxoSmithKline bivalent HPV vaccine. Previous analyses showed that at both 4 and 7 years of follow-up, a single dose of the vaccine was as effective as two or three in preventing infection with HPV types 16 and 18, which are covered by the vaccine.
“Now the research question has transitioned to, ‘Will one dose be sufficiently durable?’ she explained.
The answer from this study is yes. At 11 years since receipt of the bivalent HPV vaccine, there was no difference in terms of prevalent HPV 16/18 infection between the one-, two-, and three-dose groups. To address the issue of possible selection bias in this post hoc nonrandomized comparison, Dr. Kreimer and her coinvestigators looked at rates of infection with HPV 31 and 45, which aren’t covered by the vaccine. The rates were similar regardless of the number of vaccine doses received 11 years earlier, indicating women in all three dosing groups are at similar risk for acquiring HPV infection, thus bolstering the legitimacy of the conclusion that one dose provides effective long-term protection.
Intriguingly, HPV serum antibody levels in the single-dose group have remained stable for 11 years at a level that’s only about one-quarter of that associated with three doses of the vaccine, albeit an order of magnitude greater than the level induced by natural immunity.
“This really challenges the dogma of the HPV vaccine,” according to Dr. Kreimer. “It suggests that inferior [HPV] antibodies do not necessarily mean inferior protection.”
The explanation for this phenomenon appears to be that HPV subunit vaccine mimics the shell of authentic virions so well that the immune system sees it as dangerous and mounts long-term antibody production. Also, cervical infection by HPV is a relatively slow process, allowing time for vaccine-induced antibodies to interrupt it, she said.
In contrast to the encouraging findings from this post hoc analysis and another from a phase 3 trial of quadrivalent vaccine in India, numerous phase 4 vaccine effectiveness monitoring studies have shown markedly lower vaccine effectiveness for one dose of HPV vaccine. Dr. Kreimer cautioned that this is a flawed conclusion attributable to a methodologic artifact whereby the investigators have lumped together single-dose recipients who were 17 years old or more at the time with those who were younger.
“The problem is that many people who are aged 17-18 years already have HPV infection, so when they are vaccinated it shows up as a vaccine failure. That’s not correct. These are prophylactic HPV vaccines. They’re not meant to help clear an infection,” she noted.
Stepping back, Dr. Kreimer observed that cervical cancer “is really a story of inequality.” Indeed, 90% of cervical cancers occur in low-income countries, where HPV vaccination uptake remains very low even more than a decade after licensure. When modelers project out in the future, they estimate that at current HPV vaccination levels in Sub-Saharan Africa, which has the highest cervical cancer rates in the world, it would take more than 100 years to achieve the World Health Organization goal of eliminating the malignancy.
Asked by an audience member how low a single-dose vaccine effectiveness level she considers acceptable to help reach the goal of eliminating cervical cancer in developing countries, Dr. Kreimer cautioned against the tendency to let ‘perfect’ become the enemy of ‘good.’
“I’ll remind everyone that, in this moment, very few of the target girls in the lower– and upper-lower–income countries are getting any vaccination. So I don’t think it’s a question of whether we should be going from two to one dose, I think it’s really a question of, for those who are at zero doses, how do we get them one dose? And with the HPV vaccine, we’ve even seen suggestions of herd immunity if we have 50% uptake,” she replied.
Dr. Kreimer reported having no financial conflicts regarding her presentation.
LJUBLJANA, SLOVENIA – There is good news and bad news about human papillomavirus (HPV) vaccination as a means of preventing cervical cancer.
The bad news is the HPV vaccines are projected to be in short supply, unable to meet global demand until at least 2024. The good news is that – in one study, for 11 years and counting – which would effectively double the existing supply, Aimee R. Kreimer, PhD, said at the annual meeting of the European Society for Paediatric Infectious Diseases.
These data come from post hoc analyses of major phase 3 randomized controlled trials of bivalent HPV vaccine in Costa Rica and quadrivalent vaccine in India. However, these secondary analyses aren’t considered rock solid evidence because the subjects who got a single dose weren’t randomized to that strategy, they simply for one reason or another didn’t receive the recommended additional dose or doses.
“I don’t know if these studies are enough, so several studies have been launched over the past couple of years with an eye toward generating the quality of data that would be sufficient to motivate policy change, if in fact one dose is proven to be effective,” said Dr. Kreimer, a senior scientist at the National Cancer Institute in Bethesda, Md.
The first of these formal randomized, controlled trials – a delayed second-dose study in 9- to 11-year-old U.S. boys and girls – is due to be completed next year. Four other trials ongoing in Africa and Costa Rica, all in females, are expected to report findings in 2022-2025.
Dr. Kreimer is first author of a soon-to-be-published 11-year update from the phase 3 Costa Rica HPV Vaccine Trial, which was launched prior to licensure of the GlaxoSmithKline bivalent HPV vaccine. Previous analyses showed that at both 4 and 7 years of follow-up, a single dose of the vaccine was as effective as two or three in preventing infection with HPV types 16 and 18, which are covered by the vaccine.
“Now the research question has transitioned to, ‘Will one dose be sufficiently durable?’ she explained.
The answer from this study is yes. At 11 years since receipt of the bivalent HPV vaccine, there was no difference in terms of prevalent HPV 16/18 infection between the one-, two-, and three-dose groups. To address the issue of possible selection bias in this post hoc nonrandomized comparison, Dr. Kreimer and her coinvestigators looked at rates of infection with HPV 31 and 45, which aren’t covered by the vaccine. The rates were similar regardless of the number of vaccine doses received 11 years earlier, indicating women in all three dosing groups are at similar risk for acquiring HPV infection, thus bolstering the legitimacy of the conclusion that one dose provides effective long-term protection.
Intriguingly, HPV serum antibody levels in the single-dose group have remained stable for 11 years at a level that’s only about one-quarter of that associated with three doses of the vaccine, albeit an order of magnitude greater than the level induced by natural immunity.
“This really challenges the dogma of the HPV vaccine,” according to Dr. Kreimer. “It suggests that inferior [HPV] antibodies do not necessarily mean inferior protection.”
The explanation for this phenomenon appears to be that HPV subunit vaccine mimics the shell of authentic virions so well that the immune system sees it as dangerous and mounts long-term antibody production. Also, cervical infection by HPV is a relatively slow process, allowing time for vaccine-induced antibodies to interrupt it, she said.
In contrast to the encouraging findings from this post hoc analysis and another from a phase 3 trial of quadrivalent vaccine in India, numerous phase 4 vaccine effectiveness monitoring studies have shown markedly lower vaccine effectiveness for one dose of HPV vaccine. Dr. Kreimer cautioned that this is a flawed conclusion attributable to a methodologic artifact whereby the investigators have lumped together single-dose recipients who were 17 years old or more at the time with those who were younger.
“The problem is that many people who are aged 17-18 years already have HPV infection, so when they are vaccinated it shows up as a vaccine failure. That’s not correct. These are prophylactic HPV vaccines. They’re not meant to help clear an infection,” she noted.
Stepping back, Dr. Kreimer observed that cervical cancer “is really a story of inequality.” Indeed, 90% of cervical cancers occur in low-income countries, where HPV vaccination uptake remains very low even more than a decade after licensure. When modelers project out in the future, they estimate that at current HPV vaccination levels in Sub-Saharan Africa, which has the highest cervical cancer rates in the world, it would take more than 100 years to achieve the World Health Organization goal of eliminating the malignancy.
Asked by an audience member how low a single-dose vaccine effectiveness level she considers acceptable to help reach the goal of eliminating cervical cancer in developing countries, Dr. Kreimer cautioned against the tendency to let ‘perfect’ become the enemy of ‘good.’
“I’ll remind everyone that, in this moment, very few of the target girls in the lower– and upper-lower–income countries are getting any vaccination. So I don’t think it’s a question of whether we should be going from two to one dose, I think it’s really a question of, for those who are at zero doses, how do we get them one dose? And with the HPV vaccine, we’ve even seen suggestions of herd immunity if we have 50% uptake,” she replied.
Dr. Kreimer reported having no financial conflicts regarding her presentation.
EXPERT ANALYSIS FROM ESPID 2019
10-valent pneumococcal conjugate vaccine confers similar protection to boys and girls
according to Heta Nieminen, MD, of the National Institute for Health and Welfare in Tampere, Finland, and associates.
For the study, published in Vaccine, the investigators conducted a post hoc analysis of the phase III/IV, cluster-randomized, double-blind FinIP trial, in which more than 30,000 infants received the PHiD-CV10 vaccine or a placebo. Patients were aged less than 7 months when they received their first vaccination, and received two or three primary doses, plus a booster shot after the age of 11 months (Vaccine. 2019 May 20. doi: 10.1016/j.vaccine.2019.05.033).
In term infants, vaccine effectiveness was similar in boys and girls; while the vaccine worked marginally better in girls, the difference was not significant. Infants who received the 2 + 1 schedule had vaccine effectiveness similar to that of those who received the 3 + 1 schedule. In a smaller subanalysis of 1,519 preterm infants, outcomes of pneumonia were more common, but the vaccine seemed to confer protection, although the sample size was not large enough for statistical significance to be reached.
“The point estimates of vaccine effectiveness suggest protection in both sexes, and also among the preterm and low-birth-weight infants. ... There were no significant differences between the 2 + 1 and 3 + 1 schedules in any of the subgroups analyzed. Based on this study, the 2 + 1 or “Nordic” schedule is sufficient also for the risk groups such as the preterm or low-birth-weight infants,” the investigators concluded.
Five study authors are employees of the National Institute for Health and Welfare, which received funding for the study from GlaxoSmithKline. Four coauthors are employees of GlaxoSmithKline; three of them own shares in the company.
according to Heta Nieminen, MD, of the National Institute for Health and Welfare in Tampere, Finland, and associates.
For the study, published in Vaccine, the investigators conducted a post hoc analysis of the phase III/IV, cluster-randomized, double-blind FinIP trial, in which more than 30,000 infants received the PHiD-CV10 vaccine or a placebo. Patients were aged less than 7 months when they received their first vaccination, and received two or three primary doses, plus a booster shot after the age of 11 months (Vaccine. 2019 May 20. doi: 10.1016/j.vaccine.2019.05.033).
In term infants, vaccine effectiveness was similar in boys and girls; while the vaccine worked marginally better in girls, the difference was not significant. Infants who received the 2 + 1 schedule had vaccine effectiveness similar to that of those who received the 3 + 1 schedule. In a smaller subanalysis of 1,519 preterm infants, outcomes of pneumonia were more common, but the vaccine seemed to confer protection, although the sample size was not large enough for statistical significance to be reached.
“The point estimates of vaccine effectiveness suggest protection in both sexes, and also among the preterm and low-birth-weight infants. ... There were no significant differences between the 2 + 1 and 3 + 1 schedules in any of the subgroups analyzed. Based on this study, the 2 + 1 or “Nordic” schedule is sufficient also for the risk groups such as the preterm or low-birth-weight infants,” the investigators concluded.
Five study authors are employees of the National Institute for Health and Welfare, which received funding for the study from GlaxoSmithKline. Four coauthors are employees of GlaxoSmithKline; three of them own shares in the company.
according to Heta Nieminen, MD, of the National Institute for Health and Welfare in Tampere, Finland, and associates.
For the study, published in Vaccine, the investigators conducted a post hoc analysis of the phase III/IV, cluster-randomized, double-blind FinIP trial, in which more than 30,000 infants received the PHiD-CV10 vaccine or a placebo. Patients were aged less than 7 months when they received their first vaccination, and received two or three primary doses, plus a booster shot after the age of 11 months (Vaccine. 2019 May 20. doi: 10.1016/j.vaccine.2019.05.033).
In term infants, vaccine effectiveness was similar in boys and girls; while the vaccine worked marginally better in girls, the difference was not significant. Infants who received the 2 + 1 schedule had vaccine effectiveness similar to that of those who received the 3 + 1 schedule. In a smaller subanalysis of 1,519 preterm infants, outcomes of pneumonia were more common, but the vaccine seemed to confer protection, although the sample size was not large enough for statistical significance to be reached.
“The point estimates of vaccine effectiveness suggest protection in both sexes, and also among the preterm and low-birth-weight infants. ... There were no significant differences between the 2 + 1 and 3 + 1 schedules in any of the subgroups analyzed. Based on this study, the 2 + 1 or “Nordic” schedule is sufficient also for the risk groups such as the preterm or low-birth-weight infants,” the investigators concluded.
Five study authors are employees of the National Institute for Health and Welfare, which received funding for the study from GlaxoSmithKline. Four coauthors are employees of GlaxoSmithKline; three of them own shares in the company.
FROM VACCINE
Guidelines recommend adding trained personnel for pediatric sedation during dental procedures
at a dental facility or hospital setting, according to new guidelines released by the American Academy of Pediatrics and American Academy of Pediatric Dentistry.
“Sedation for dental procedures in children and teenagers is generally safe,” Charles J. Coté, MD, lead author of the clinical report who is a pediatrician and a pediatric anesthesiologist, stated in a press release from the American Academy of Pediatrics. “However, we are aware of adverse outcomes when a single dental provider simultaneously performs the procedure and administers deep sedation or general anesthesia for dental procedures. These guidelines ensure the safety of patients who undergo these procedures.”
The full report, which will be published in the June issue of Pediatrics, otherwise remains largely the same as the 2016 guidelines published by AAP and the American Academy of Pediatric Dentistry.
While the previous guidelines had recommended at least one individual with Pediatric Advanced Life Support (PALS) certification be present during a procedure, the new guidelines specify at least two individuals with appropriate certification and training in patient rescue should be in the room during a procedure, regardless of setting, and have the PALS certification as well as be trained in drug administration.
One individual also should be dedicated to monitoring the patient during sedation and capable of performing rescue care, such as administering rescue medications and rescuing a child who is not breathing because of airway obstruction, anaphylaxis, hypotension, or cardiorespiratory arrest. The individual observer should be a physician anesthesiologist, dental anesthesiologist, oral surgeon, or a certified registered nurse anesthetist. “This individual’s sole responsibility is to administer drugs and constantly observe the patient’s vital signs, depth of sedation, airway patency, and adequacy of ventilation,” the authors said.
In addition, the guidelines state sedation must be administered by a qualified anesthesia provider with “sedation training and advanced airway skills,” such as a physician anesthesiologist, dentist anesthesiologist, oral surgeon, or “other medical specialists with the requisite licensure, training, and competencies; a certified registered nurse anesthetist or certified anesthesiology assistant; or a nurse with advanced emergency management skills,” they said.
The operating dentist and the independent observer both must be PALS certified.
The authors reported no relevant conflicts of interest.
SOURCE: Coté CJ et al. Pediatrics. 2019. doi: 10.1542/peds.2019-1000.
at a dental facility or hospital setting, according to new guidelines released by the American Academy of Pediatrics and American Academy of Pediatric Dentistry.
“Sedation for dental procedures in children and teenagers is generally safe,” Charles J. Coté, MD, lead author of the clinical report who is a pediatrician and a pediatric anesthesiologist, stated in a press release from the American Academy of Pediatrics. “However, we are aware of adverse outcomes when a single dental provider simultaneously performs the procedure and administers deep sedation or general anesthesia for dental procedures. These guidelines ensure the safety of patients who undergo these procedures.”
The full report, which will be published in the June issue of Pediatrics, otherwise remains largely the same as the 2016 guidelines published by AAP and the American Academy of Pediatric Dentistry.
While the previous guidelines had recommended at least one individual with Pediatric Advanced Life Support (PALS) certification be present during a procedure, the new guidelines specify at least two individuals with appropriate certification and training in patient rescue should be in the room during a procedure, regardless of setting, and have the PALS certification as well as be trained in drug administration.
One individual also should be dedicated to monitoring the patient during sedation and capable of performing rescue care, such as administering rescue medications and rescuing a child who is not breathing because of airway obstruction, anaphylaxis, hypotension, or cardiorespiratory arrest. The individual observer should be a physician anesthesiologist, dental anesthesiologist, oral surgeon, or a certified registered nurse anesthetist. “This individual’s sole responsibility is to administer drugs and constantly observe the patient’s vital signs, depth of sedation, airway patency, and adequacy of ventilation,” the authors said.
In addition, the guidelines state sedation must be administered by a qualified anesthesia provider with “sedation training and advanced airway skills,” such as a physician anesthesiologist, dentist anesthesiologist, oral surgeon, or “other medical specialists with the requisite licensure, training, and competencies; a certified registered nurse anesthetist or certified anesthesiology assistant; or a nurse with advanced emergency management skills,” they said.
The operating dentist and the independent observer both must be PALS certified.
The authors reported no relevant conflicts of interest.
SOURCE: Coté CJ et al. Pediatrics. 2019. doi: 10.1542/peds.2019-1000.
at a dental facility or hospital setting, according to new guidelines released by the American Academy of Pediatrics and American Academy of Pediatric Dentistry.
“Sedation for dental procedures in children and teenagers is generally safe,” Charles J. Coté, MD, lead author of the clinical report who is a pediatrician and a pediatric anesthesiologist, stated in a press release from the American Academy of Pediatrics. “However, we are aware of adverse outcomes when a single dental provider simultaneously performs the procedure and administers deep sedation or general anesthesia for dental procedures. These guidelines ensure the safety of patients who undergo these procedures.”
The full report, which will be published in the June issue of Pediatrics, otherwise remains largely the same as the 2016 guidelines published by AAP and the American Academy of Pediatric Dentistry.
While the previous guidelines had recommended at least one individual with Pediatric Advanced Life Support (PALS) certification be present during a procedure, the new guidelines specify at least two individuals with appropriate certification and training in patient rescue should be in the room during a procedure, regardless of setting, and have the PALS certification as well as be trained in drug administration.
One individual also should be dedicated to monitoring the patient during sedation and capable of performing rescue care, such as administering rescue medications and rescuing a child who is not breathing because of airway obstruction, anaphylaxis, hypotension, or cardiorespiratory arrest. The individual observer should be a physician anesthesiologist, dental anesthesiologist, oral surgeon, or a certified registered nurse anesthetist. “This individual’s sole responsibility is to administer drugs and constantly observe the patient’s vital signs, depth of sedation, airway patency, and adequacy of ventilation,” the authors said.
In addition, the guidelines state sedation must be administered by a qualified anesthesia provider with “sedation training and advanced airway skills,” such as a physician anesthesiologist, dentist anesthesiologist, oral surgeon, or “other medical specialists with the requisite licensure, training, and competencies; a certified registered nurse anesthetist or certified anesthesiology assistant; or a nurse with advanced emergency management skills,” they said.
The operating dentist and the independent observer both must be PALS certified.
The authors reported no relevant conflicts of interest.
SOURCE: Coté CJ et al. Pediatrics. 2019. doi: 10.1542/peds.2019-1000.
FROM PEDIATRICS
Severe respiratory failure strikes healthy teens on trimethoprim-sulfamethoxazole
TMP-SMX, a frequently prescribed antibiotic, has been associated with “idiosyncratic adverse drug reactions, including cutaneous reactions and hypersensitivity syndromes,” but pulmonary complications are rare, especially in children, wrote Jenna O. Miller, MD, of the University of Missouri–Kansas City and colleagues.
In a case series published in Pediatrics, the researchers described the patients, who were aged 13-18 years; the 18-year-old was male, the others were female. Four of the patients (three females, one male) were taking TMP-SMX for acne vulgaris. One of these patients, a 13-year-old girl, underwent a bilateral lung and heart transplant after developing interstitial lung disease and died as a result of solid organ transplant complications. The other death occurred in a 15-year-old girl who was taking TMP-SMX to treat a urinary tract infection. This patient developed interstitial lung disease and died of complications from the disease while awaiting a lung transplant.
“In all cases, patients were transferred to academic medical facilities, and pediatric pulmonologists and infectious diseases specialists performed extensive evaluations,” the researchers wrote. The patients did not improve when the drug was discontinued, and four of the five were considered or listed for organ transplants. The spectrum of disease was varied among the patients, and the pathophysiology remains poorly understood.
Although no clinical test could confirm causality between TMP-SMX and ARDS in the five teens, “the extensive negative workup, paired with recent TMP-SMX exposure and similarity among these cases, raises the possibility that the observed ARDS was TMP-SMX triggered,” they wrote.
The researchers had no financial conflicts to disclose.
SOURCE: Miller JO et al. Pediatrics. 2019 May 29. doi: 10.1542/peds.2018.3242.
TMP-SMX, a frequently prescribed antibiotic, has been associated with “idiosyncratic adverse drug reactions, including cutaneous reactions and hypersensitivity syndromes,” but pulmonary complications are rare, especially in children, wrote Jenna O. Miller, MD, of the University of Missouri–Kansas City and colleagues.
In a case series published in Pediatrics, the researchers described the patients, who were aged 13-18 years; the 18-year-old was male, the others were female. Four of the patients (three females, one male) were taking TMP-SMX for acne vulgaris. One of these patients, a 13-year-old girl, underwent a bilateral lung and heart transplant after developing interstitial lung disease and died as a result of solid organ transplant complications. The other death occurred in a 15-year-old girl who was taking TMP-SMX to treat a urinary tract infection. This patient developed interstitial lung disease and died of complications from the disease while awaiting a lung transplant.
“In all cases, patients were transferred to academic medical facilities, and pediatric pulmonologists and infectious diseases specialists performed extensive evaluations,” the researchers wrote. The patients did not improve when the drug was discontinued, and four of the five were considered or listed for organ transplants. The spectrum of disease was varied among the patients, and the pathophysiology remains poorly understood.
Although no clinical test could confirm causality between TMP-SMX and ARDS in the five teens, “the extensive negative workup, paired with recent TMP-SMX exposure and similarity among these cases, raises the possibility that the observed ARDS was TMP-SMX triggered,” they wrote.
The researchers had no financial conflicts to disclose.
SOURCE: Miller JO et al. Pediatrics. 2019 May 29. doi: 10.1542/peds.2018.3242.
TMP-SMX, a frequently prescribed antibiotic, has been associated with “idiosyncratic adverse drug reactions, including cutaneous reactions and hypersensitivity syndromes,” but pulmonary complications are rare, especially in children, wrote Jenna O. Miller, MD, of the University of Missouri–Kansas City and colleagues.
In a case series published in Pediatrics, the researchers described the patients, who were aged 13-18 years; the 18-year-old was male, the others were female. Four of the patients (three females, one male) were taking TMP-SMX for acne vulgaris. One of these patients, a 13-year-old girl, underwent a bilateral lung and heart transplant after developing interstitial lung disease and died as a result of solid organ transplant complications. The other death occurred in a 15-year-old girl who was taking TMP-SMX to treat a urinary tract infection. This patient developed interstitial lung disease and died of complications from the disease while awaiting a lung transplant.
“In all cases, patients were transferred to academic medical facilities, and pediatric pulmonologists and infectious diseases specialists performed extensive evaluations,” the researchers wrote. The patients did not improve when the drug was discontinued, and four of the five were considered or listed for organ transplants. The spectrum of disease was varied among the patients, and the pathophysiology remains poorly understood.
Although no clinical test could confirm causality between TMP-SMX and ARDS in the five teens, “the extensive negative workup, paired with recent TMP-SMX exposure and similarity among these cases, raises the possibility that the observed ARDS was TMP-SMX triggered,” they wrote.
The researchers had no financial conflicts to disclose.
SOURCE: Miller JO et al. Pediatrics. 2019 May 29. doi: 10.1542/peds.2018.3242.
FROM PEDIATRICS
Measles count for 2019 now over 900 cases
according to the Centers for Disease Control and Prevention.
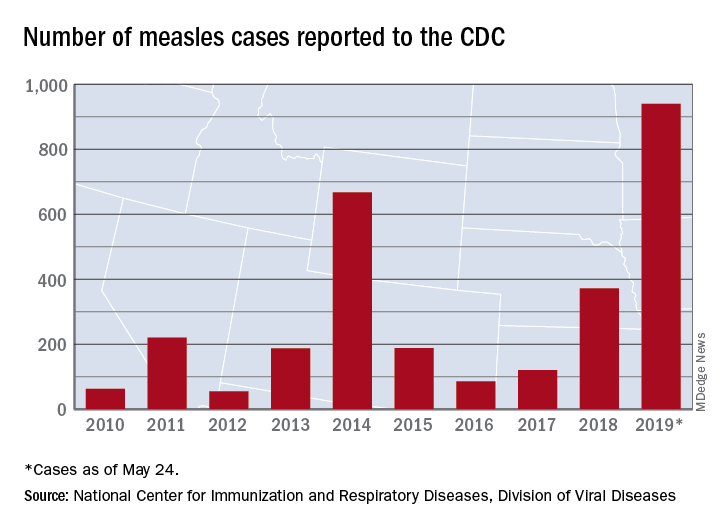
The CDC received reports of 60 new measles cases last week – up from 41 the previous week – bringing the U.S. total to 940 for the year as of May 24. The CDC is currently tracking 10 outbreaks in seven states: California (3), Georgia, Maryland, Michigan, New York (2), Pennsylvania, and Washington.
The Maine Center for Disease Control and Prevention confirmed the state’s first case on May 20. The school-aged child from Somerset County had been vaccinated and is fully recovered from the disease. It’s not yet known where the child was exposed to measles, but sporadic cases are not unexpected, the Maine CDC said.
New Mexico’s first measles case of the year, a 1-year-old in Sierra County, has at least one state lawmaker considering changes to the state’s immunization exemption laws, the Farmington Daily Times reported.
according to the Centers for Disease Control and Prevention.

The CDC received reports of 60 new measles cases last week – up from 41 the previous week – bringing the U.S. total to 940 for the year as of May 24. The CDC is currently tracking 10 outbreaks in seven states: California (3), Georgia, Maryland, Michigan, New York (2), Pennsylvania, and Washington.
The Maine Center for Disease Control and Prevention confirmed the state’s first case on May 20. The school-aged child from Somerset County had been vaccinated and is fully recovered from the disease. It’s not yet known where the child was exposed to measles, but sporadic cases are not unexpected, the Maine CDC said.
New Mexico’s first measles case of the year, a 1-year-old in Sierra County, has at least one state lawmaker considering changes to the state’s immunization exemption laws, the Farmington Daily Times reported.
according to the Centers for Disease Control and Prevention.

The CDC received reports of 60 new measles cases last week – up from 41 the previous week – bringing the U.S. total to 940 for the year as of May 24. The CDC is currently tracking 10 outbreaks in seven states: California (3), Georgia, Maryland, Michigan, New York (2), Pennsylvania, and Washington.
The Maine Center for Disease Control and Prevention confirmed the state’s first case on May 20. The school-aged child from Somerset County had been vaccinated and is fully recovered from the disease. It’s not yet known where the child was exposed to measles, but sporadic cases are not unexpected, the Maine CDC said.
New Mexico’s first measles case of the year, a 1-year-old in Sierra County, has at least one state lawmaker considering changes to the state’s immunization exemption laws, the Farmington Daily Times reported.
Team sports may mitigate tough childhoods
Individuals who experienced adverse childhood experiences but also played team sports as teens were less likely to have mental health problems in adulthood than those with childhood challenges who did not play sports, based on data from nearly 5,000 individuals.
Physical and mental health problems are more prominent throughout life among those exposed to adverse childhood experiences (ACEs), and physical activity in general and team sports in particular have been shown to improve mental health, wrote Molly C. Easterlin, MD, of the University of California, Los Angeles, and colleagues.
In a study published in JAMA Pediatrics, the researchers used data from the National Longitudinal Study of Adolescent to Adult Health to compare the development of depression, anxiety, or depressive symptoms among those with childhood ACEs who did and did not participate in team sports in adolescence.
Overall, team sports participation was significantly associated with reduced odds of depression (adjusted odds ratio, 0.76), anxiety (aOR, 0.70), and depressive symptoms (aOR, 0.85) in young adulthood for individuals with ACEs, compared with those with ACEs who did not play team sports.
Of 9,668 adolescents in the study, 4,888 individuals reported one or more ACEs and 2,084 reported two or more ACEs. The researchers compared data from the 1994-1995 school year when participants were in grades 7-12 and in 2008 to assess their mental health as young adults (aged 24-32 years).
No significant differences in associations appeared between sports participation and mental health between males and females.
The results were limited by several factors including the study design that did not allow for causality and the potential social desirability bias that might lead to underreporting ACEs, Dr. Easterlin and associates noted.
Nonetheless, “given that participation in team sports was associated with improved adult mental health among those with ACEs, and parents might consider enrolling their children with ACEs in team sports,” they wrote.
Dr. Easterlin is supported by the Cedars-Sinai Medical Center via the UCLA National Clinician Scholars Program. The authors reported no conflicts of interest.
SOURCE: Easterlin MC et al. JAMA Pediatr. 2019 May 28. doi: 10.1001/jamapediatrics.2019.1212.
Approximately half of children suffer an adverse childhood experience (ACE) that can negatively affect their mental health throughout life, and “team sports can be an avenue to interrupt these negative sequelae and address the important public health burden of depression,” wrote Amanda E. Paluch, PhD; Nia Heard-Garris, MD, MSc; and Mercedes R. Carnethon, PhD.
However, a significant socioeconomic disparity in team sports for children continues to grow in the United States, driven in part by a youth sports industry and culture that caters to high-income families looking to improve their children’s performance. “Although unintentional, these expenses leave behind lower-income children,” many of whom may be at increased risk for ACEs, the editorialists noted. Many inexpensive, community-based recreation leagues, especially in low-income areas, are often underfunded and unable to update facilities and attract more participants.
The benefits of team sports appear to go beyond the physical, as the study by Easterlin et al. suggests that feeling accepted and connected as part of a team has an impact on mental health. Also, the winning and losing of sports helps build emotional resilience that carries over to other areas of life, the editorialists added.
“Optimizing the opportunities for sports during adolescence requires relatively few resources and is a low-cost way to improve quality of life and reduce the population burden of mental health disorders, especially for adolescents and young adults with histories of ACEs,” they concluded.
Dr. Paluch and Dr. Carnethon are affiliated with the department of preventive medicine and Dr. Heard-Garris is affiliated with the department of pediatrics at Northwestern University, Chicago. They commented on the study by Easterlin et al (JAMA Pediatr. 2019 May 28. doi:10.1001/jamapediatrics.2019.1209). They reported no conflicts of interest.
Approximately half of children suffer an adverse childhood experience (ACE) that can negatively affect their mental health throughout life, and “team sports can be an avenue to interrupt these negative sequelae and address the important public health burden of depression,” wrote Amanda E. Paluch, PhD; Nia Heard-Garris, MD, MSc; and Mercedes R. Carnethon, PhD.
However, a significant socioeconomic disparity in team sports for children continues to grow in the United States, driven in part by a youth sports industry and culture that caters to high-income families looking to improve their children’s performance. “Although unintentional, these expenses leave behind lower-income children,” many of whom may be at increased risk for ACEs, the editorialists noted. Many inexpensive, community-based recreation leagues, especially in low-income areas, are often underfunded and unable to update facilities and attract more participants.
The benefits of team sports appear to go beyond the physical, as the study by Easterlin et al. suggests that feeling accepted and connected as part of a team has an impact on mental health. Also, the winning and losing of sports helps build emotional resilience that carries over to other areas of life, the editorialists added.
“Optimizing the opportunities for sports during adolescence requires relatively few resources and is a low-cost way to improve quality of life and reduce the population burden of mental health disorders, especially for adolescents and young adults with histories of ACEs,” they concluded.
Dr. Paluch and Dr. Carnethon are affiliated with the department of preventive medicine and Dr. Heard-Garris is affiliated with the department of pediatrics at Northwestern University, Chicago. They commented on the study by Easterlin et al (JAMA Pediatr. 2019 May 28. doi:10.1001/jamapediatrics.2019.1209). They reported no conflicts of interest.
Approximately half of children suffer an adverse childhood experience (ACE) that can negatively affect their mental health throughout life, and “team sports can be an avenue to interrupt these negative sequelae and address the important public health burden of depression,” wrote Amanda E. Paluch, PhD; Nia Heard-Garris, MD, MSc; and Mercedes R. Carnethon, PhD.
However, a significant socioeconomic disparity in team sports for children continues to grow in the United States, driven in part by a youth sports industry and culture that caters to high-income families looking to improve their children’s performance. “Although unintentional, these expenses leave behind lower-income children,” many of whom may be at increased risk for ACEs, the editorialists noted. Many inexpensive, community-based recreation leagues, especially in low-income areas, are often underfunded and unable to update facilities and attract more participants.
The benefits of team sports appear to go beyond the physical, as the study by Easterlin et al. suggests that feeling accepted and connected as part of a team has an impact on mental health. Also, the winning and losing of sports helps build emotional resilience that carries over to other areas of life, the editorialists added.
“Optimizing the opportunities for sports during adolescence requires relatively few resources and is a low-cost way to improve quality of life and reduce the population burden of mental health disorders, especially for adolescents and young adults with histories of ACEs,” they concluded.
Dr. Paluch and Dr. Carnethon are affiliated with the department of preventive medicine and Dr. Heard-Garris is affiliated with the department of pediatrics at Northwestern University, Chicago. They commented on the study by Easterlin et al (JAMA Pediatr. 2019 May 28. doi:10.1001/jamapediatrics.2019.1209). They reported no conflicts of interest.
Individuals who experienced adverse childhood experiences but also played team sports as teens were less likely to have mental health problems in adulthood than those with childhood challenges who did not play sports, based on data from nearly 5,000 individuals.
Physical and mental health problems are more prominent throughout life among those exposed to adverse childhood experiences (ACEs), and physical activity in general and team sports in particular have been shown to improve mental health, wrote Molly C. Easterlin, MD, of the University of California, Los Angeles, and colleagues.
In a study published in JAMA Pediatrics, the researchers used data from the National Longitudinal Study of Adolescent to Adult Health to compare the development of depression, anxiety, or depressive symptoms among those with childhood ACEs who did and did not participate in team sports in adolescence.
Overall, team sports participation was significantly associated with reduced odds of depression (adjusted odds ratio, 0.76), anxiety (aOR, 0.70), and depressive symptoms (aOR, 0.85) in young adulthood for individuals with ACEs, compared with those with ACEs who did not play team sports.
Of 9,668 adolescents in the study, 4,888 individuals reported one or more ACEs and 2,084 reported two or more ACEs. The researchers compared data from the 1994-1995 school year when participants were in grades 7-12 and in 2008 to assess their mental health as young adults (aged 24-32 years).
No significant differences in associations appeared between sports participation and mental health between males and females.
The results were limited by several factors including the study design that did not allow for causality and the potential social desirability bias that might lead to underreporting ACEs, Dr. Easterlin and associates noted.
Nonetheless, “given that participation in team sports was associated with improved adult mental health among those with ACEs, and parents might consider enrolling their children with ACEs in team sports,” they wrote.
Dr. Easterlin is supported by the Cedars-Sinai Medical Center via the UCLA National Clinician Scholars Program. The authors reported no conflicts of interest.
SOURCE: Easterlin MC et al. JAMA Pediatr. 2019 May 28. doi: 10.1001/jamapediatrics.2019.1212.
Individuals who experienced adverse childhood experiences but also played team sports as teens were less likely to have mental health problems in adulthood than those with childhood challenges who did not play sports, based on data from nearly 5,000 individuals.
Physical and mental health problems are more prominent throughout life among those exposed to adverse childhood experiences (ACEs), and physical activity in general and team sports in particular have been shown to improve mental health, wrote Molly C. Easterlin, MD, of the University of California, Los Angeles, and colleagues.
In a study published in JAMA Pediatrics, the researchers used data from the National Longitudinal Study of Adolescent to Adult Health to compare the development of depression, anxiety, or depressive symptoms among those with childhood ACEs who did and did not participate in team sports in adolescence.
Overall, team sports participation was significantly associated with reduced odds of depression (adjusted odds ratio, 0.76), anxiety (aOR, 0.70), and depressive symptoms (aOR, 0.85) in young adulthood for individuals with ACEs, compared with those with ACEs who did not play team sports.
Of 9,668 adolescents in the study, 4,888 individuals reported one or more ACEs and 2,084 reported two or more ACEs. The researchers compared data from the 1994-1995 school year when participants were in grades 7-12 and in 2008 to assess their mental health as young adults (aged 24-32 years).
No significant differences in associations appeared between sports participation and mental health between males and females.
The results were limited by several factors including the study design that did not allow for causality and the potential social desirability bias that might lead to underreporting ACEs, Dr. Easterlin and associates noted.
Nonetheless, “given that participation in team sports was associated with improved adult mental health among those with ACEs, and parents might consider enrolling their children with ACEs in team sports,” they wrote.
Dr. Easterlin is supported by the Cedars-Sinai Medical Center via the UCLA National Clinician Scholars Program. The authors reported no conflicts of interest.
SOURCE: Easterlin MC et al. JAMA Pediatr. 2019 May 28. doi: 10.1001/jamapediatrics.2019.1212.
FROM JAMA PEDIATRICS
HM19: Things we do for no reason
The pediatric version
Presenters
Amit K. Pahwa, MD, FAAP; Nicola Orlov, MD, MPH, FAAP
Workshop title
Things we do for no reason (pediatrics)
Session summary
As he began by stating the Institute of Medicine definition of high-value care (HVC), Amit K. Pahwa, MD, of Johns Hopkins Medicine, Baltimore, described HVC as the best care for the patient, with the optimal result for the circumstances, at the right price. But few pediatric residency programs provide education regarding HVC, with only 11% providing a formal HVC curriculum, as found by a survey of pediatric program directors and chief residents in a study published in 2017.
Dr. Pahwa then provided examples of cases in which HVC could be optimized, including reducing rebound bilirubin levels in neonatal hyperbilirubinemia, giving nasogastric feeds instead of IV hydration in bronchiolitis, reducing unnecessary vital sign checks, and providing apple juice supplemented with liquids of choice instead of more expensive oral electrolyte solutions.
Nicola Orlov, MD, of the University of Chicago presented another illustrative case which highlighted the need to reduce vital sign frequency when appropriate. This was linked to her work at Comer Children’s Hospital on reducing nighttime sleep disruptions in hospitalized children, as part of the SIESTA (Sleep for Inpatients: Empowering Staff to Act) study. This led to a significant reduction in nurse/physician interruptions during the study period.
Key takeaways for HM
- High-value care (HVC) is a key focus of systems improvement in the field of pediatric hospital medicine.
- Educational efforts for all levels of learners is inadequate currently, and needs to be augmented.
- Quality improvement projects to promote HVC can lead not only to reduced costs, but improved quality and patient experience.
Dr. Chang is a pediatric hospitalist at Baystate Children’s Hospital in Springfield, Mass., and is the pediatric editor of The Hospitalist.
The pediatric version
The pediatric version
Presenters
Amit K. Pahwa, MD, FAAP; Nicola Orlov, MD, MPH, FAAP
Workshop title
Things we do for no reason (pediatrics)
Session summary
As he began by stating the Institute of Medicine definition of high-value care (HVC), Amit K. Pahwa, MD, of Johns Hopkins Medicine, Baltimore, described HVC as the best care for the patient, with the optimal result for the circumstances, at the right price. But few pediatric residency programs provide education regarding HVC, with only 11% providing a formal HVC curriculum, as found by a survey of pediatric program directors and chief residents in a study published in 2017.
Dr. Pahwa then provided examples of cases in which HVC could be optimized, including reducing rebound bilirubin levels in neonatal hyperbilirubinemia, giving nasogastric feeds instead of IV hydration in bronchiolitis, reducing unnecessary vital sign checks, and providing apple juice supplemented with liquids of choice instead of more expensive oral electrolyte solutions.
Nicola Orlov, MD, of the University of Chicago presented another illustrative case which highlighted the need to reduce vital sign frequency when appropriate. This was linked to her work at Comer Children’s Hospital on reducing nighttime sleep disruptions in hospitalized children, as part of the SIESTA (Sleep for Inpatients: Empowering Staff to Act) study. This led to a significant reduction in nurse/physician interruptions during the study period.
Key takeaways for HM
- High-value care (HVC) is a key focus of systems improvement in the field of pediatric hospital medicine.
- Educational efforts for all levels of learners is inadequate currently, and needs to be augmented.
- Quality improvement projects to promote HVC can lead not only to reduced costs, but improved quality and patient experience.
Dr. Chang is a pediatric hospitalist at Baystate Children’s Hospital in Springfield, Mass., and is the pediatric editor of The Hospitalist.
Presenters
Amit K. Pahwa, MD, FAAP; Nicola Orlov, MD, MPH, FAAP
Workshop title
Things we do for no reason (pediatrics)
Session summary
As he began by stating the Institute of Medicine definition of high-value care (HVC), Amit K. Pahwa, MD, of Johns Hopkins Medicine, Baltimore, described HVC as the best care for the patient, with the optimal result for the circumstances, at the right price. But few pediatric residency programs provide education regarding HVC, with only 11% providing a formal HVC curriculum, as found by a survey of pediatric program directors and chief residents in a study published in 2017.
Dr. Pahwa then provided examples of cases in which HVC could be optimized, including reducing rebound bilirubin levels in neonatal hyperbilirubinemia, giving nasogastric feeds instead of IV hydration in bronchiolitis, reducing unnecessary vital sign checks, and providing apple juice supplemented with liquids of choice instead of more expensive oral electrolyte solutions.
Nicola Orlov, MD, of the University of Chicago presented another illustrative case which highlighted the need to reduce vital sign frequency when appropriate. This was linked to her work at Comer Children’s Hospital on reducing nighttime sleep disruptions in hospitalized children, as part of the SIESTA (Sleep for Inpatients: Empowering Staff to Act) study. This led to a significant reduction in nurse/physician interruptions during the study period.
Key takeaways for HM
- High-value care (HVC) is a key focus of systems improvement in the field of pediatric hospital medicine.
- Educational efforts for all levels of learners is inadequate currently, and needs to be augmented.
- Quality improvement projects to promote HVC can lead not only to reduced costs, but improved quality and patient experience.
Dr. Chang is a pediatric hospitalist at Baystate Children’s Hospital in Springfield, Mass., and is the pediatric editor of The Hospitalist.