User login
Stroke caution on thalidomide for cutaneous LE
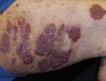
PRAGUE – Low-dose thalidomide for refractory cutaneous lupus erythematosus is best used together with hydroxychloroquine or another antimalarial agent rather than as monotherapy, Dr. Victoria P. Werth asserted at the annual congress of the European Academy of Dermatology and Venereology.
When prescribing thalidomide for a patient with refractory cutaneous lupus erythematosus (CLE), many physicians discontinue antimalarial therapy, reasoning that since the patient wasn’t responsive to monotherapy, there’s no point in continued exposure to the potential risks. But that’s probably a mistake. Combination therapy acting through different mechanisms may boost the likelihood of a good response; plus, the antiplatelet action of hydroxychloroquine or another antimalarial agent will help counteract thalidomide’s prothrombotic effects, said Dr. Werth, professor of dermatology at the University of Pennsylvania, Philadelphia.
Thalidomide is unquestionably an effective therapy in patients with refractory CLE. But it’s also a drug with big problems, including perhaps an increased stroke risk, as highlighted in a recent Spanish study, she noted.
The Spanish study included 60 consecutive patients with refractory CLE who were treated with thalidomide at 100 mg/day and followed for up to 8 years. One dropped out due to side effects. Fifty-eight of the remaining 59 experienced significant clinical improvement, including 49 (85%) with a complete response as defined by a CLASI (Cutaneous Lupus Erythematosus Disease Area and Severity Index) activity score of 0.
Relapse occurred in most patients, usually about 5 months after thalidomide dose reduction or withdrawal. Patients with subacute CLE were 30-fold more likely to remain in remission after drug discontinuation; those with discoid LE were at increased risk for relapse (Br. J. Dermatol. 2012;166:616-23).
Of particular concern to Dr. Werth was the finding that two patients had a stroke while on the drug. Neither had antiphospholipid antibodies, and one was quite young to have had a stroke, although both were heavy smokers.
Prescribing a drug such as thalidomide that promotes a hypercoagulable state to patients with refractory CLE is problematic because they often already have multiple risk factors for thrombosis. For one thing, treatment-refractory CLE patients tend to be smokers. Many of them are women on oral contraceptives. And there is an increased prevalence of antiphospholipid antibodies in patients with CLE, according to Dr. Werth.
Neuropathy is another major issue with thalidomide. In the Spanish study, 11 of 60 patients (18%) developed paresthesias; nerve conduction studies confirmed sensory polyneuropathy in 5 of the 11. Fortunately, the neurologic symptoms resolved in an average of 12 months after drug withdrawal.
Of course, thalidomide is a notorious teratogen. It can also cause premature ovarian failure, although this is usually reversible upon drug discontinuation.
"Obviously we need better therapies than thalidomide," Dr. Werth concluded.
Toward that end, interest is growing in thalidomide analogues as a novel potential therapy for refractory CLE. These analogues are up to 50,000 times more active than thalidomide, and are potentially less neurotoxic. One of them, lenalidomide (Revlimid), is marketed as a treatment for multiple myeloma and myelodysplastic syndrome. Others are in the development pipeline.
Several small observational studies have recently reported favorable results with lenalidomide in patients with refractory CLE. For example, investigators at Vall d’Hebron University Hospital in Barcelona reported on 15 patients treated open label with lenalidomide at 5-10 mg/day, with a follow-up of 15 months. One patient dropped out early due to side effects, but the other 14 saw clinical improvement within the first 2 weeks. Twelve patients, or 86%, achieved a CLASI score of 0. However, 9 of 12 complete responders experienced clinical relapse, usually 2-8 weeks after the drug was tapered and discontinued. Side effects were mild and infrequent, with no thrombosis or polyneuropathy (Arthritis Res. Ther. 2012;14:R265).
In another series, 4 of 5 lenalidomide-treated patients showed significant skin improvement, although one eventually developed symptoms of SLE (J. Am. Acad. Dermatol. 2012;66:571-82).
Based upon these and other promising reports, Celgene, which markets lenalidomide, recently launched the first-ever phase II study of a thalidomide analogue for the treatment of CLE.
Dr. Werth reported having no financial conflicts.
PRAGUE – Low-dose thalidomide for refractory cutaneous lupus erythematosus is best used together with hydroxychloroquine or another antimalarial agent rather than as monotherapy, Dr. Victoria P. Werth asserted at the annual congress of the European Academy of Dermatology and Venereology.
When prescribing thalidomide for a patient with refractory cutaneous lupus erythematosus (CLE), many physicians discontinue antimalarial therapy, reasoning that since the patient wasn’t responsive to monotherapy, there’s no point in continued exposure to the potential risks. But that’s probably a mistake. Combination therapy acting through different mechanisms may boost the likelihood of a good response; plus, the antiplatelet action of hydroxychloroquine or another antimalarial agent will help counteract thalidomide’s prothrombotic effects, said Dr. Werth, professor of dermatology at the University of Pennsylvania, Philadelphia.
Thalidomide is unquestionably an effective therapy in patients with refractory CLE. But it’s also a drug with big problems, including perhaps an increased stroke risk, as highlighted in a recent Spanish study, she noted.
The Spanish study included 60 consecutive patients with refractory CLE who were treated with thalidomide at 100 mg/day and followed for up to 8 years. One dropped out due to side effects. Fifty-eight of the remaining 59 experienced significant clinical improvement, including 49 (85%) with a complete response as defined by a CLASI (Cutaneous Lupus Erythematosus Disease Area and Severity Index) activity score of 0.
Relapse occurred in most patients, usually about 5 months after thalidomide dose reduction or withdrawal. Patients with subacute CLE were 30-fold more likely to remain in remission after drug discontinuation; those with discoid LE were at increased risk for relapse (Br. J. Dermatol. 2012;166:616-23).
Of particular concern to Dr. Werth was the finding that two patients had a stroke while on the drug. Neither had antiphospholipid antibodies, and one was quite young to have had a stroke, although both were heavy smokers.
Prescribing a drug such as thalidomide that promotes a hypercoagulable state to patients with refractory CLE is problematic because they often already have multiple risk factors for thrombosis. For one thing, treatment-refractory CLE patients tend to be smokers. Many of them are women on oral contraceptives. And there is an increased prevalence of antiphospholipid antibodies in patients with CLE, according to Dr. Werth.
Neuropathy is another major issue with thalidomide. In the Spanish study, 11 of 60 patients (18%) developed paresthesias; nerve conduction studies confirmed sensory polyneuropathy in 5 of the 11. Fortunately, the neurologic symptoms resolved in an average of 12 months after drug withdrawal.
Of course, thalidomide is a notorious teratogen. It can also cause premature ovarian failure, although this is usually reversible upon drug discontinuation.
"Obviously we need better therapies than thalidomide," Dr. Werth concluded.
Toward that end, interest is growing in thalidomide analogues as a novel potential therapy for refractory CLE. These analogues are up to 50,000 times more active than thalidomide, and are potentially less neurotoxic. One of them, lenalidomide (Revlimid), is marketed as a treatment for multiple myeloma and myelodysplastic syndrome. Others are in the development pipeline.
Several small observational studies have recently reported favorable results with lenalidomide in patients with refractory CLE. For example, investigators at Vall d’Hebron University Hospital in Barcelona reported on 15 patients treated open label with lenalidomide at 5-10 mg/day, with a follow-up of 15 months. One patient dropped out early due to side effects, but the other 14 saw clinical improvement within the first 2 weeks. Twelve patients, or 86%, achieved a CLASI score of 0. However, 9 of 12 complete responders experienced clinical relapse, usually 2-8 weeks after the drug was tapered and discontinued. Side effects were mild and infrequent, with no thrombosis or polyneuropathy (Arthritis Res. Ther. 2012;14:R265).
In another series, 4 of 5 lenalidomide-treated patients showed significant skin improvement, although one eventually developed symptoms of SLE (J. Am. Acad. Dermatol. 2012;66:571-82).
Based upon these and other promising reports, Celgene, which markets lenalidomide, recently launched the first-ever phase II study of a thalidomide analogue for the treatment of CLE.
Dr. Werth reported having no financial conflicts.
PRAGUE – Low-dose thalidomide for refractory cutaneous lupus erythematosus is best used together with hydroxychloroquine or another antimalarial agent rather than as monotherapy, Dr. Victoria P. Werth asserted at the annual congress of the European Academy of Dermatology and Venereology.
When prescribing thalidomide for a patient with refractory cutaneous lupus erythematosus (CLE), many physicians discontinue antimalarial therapy, reasoning that since the patient wasn’t responsive to monotherapy, there’s no point in continued exposure to the potential risks. But that’s probably a mistake. Combination therapy acting through different mechanisms may boost the likelihood of a good response; plus, the antiplatelet action of hydroxychloroquine or another antimalarial agent will help counteract thalidomide’s prothrombotic effects, said Dr. Werth, professor of dermatology at the University of Pennsylvania, Philadelphia.
Thalidomide is unquestionably an effective therapy in patients with refractory CLE. But it’s also a drug with big problems, including perhaps an increased stroke risk, as highlighted in a recent Spanish study, she noted.
The Spanish study included 60 consecutive patients with refractory CLE who were treated with thalidomide at 100 mg/day and followed for up to 8 years. One dropped out due to side effects. Fifty-eight of the remaining 59 experienced significant clinical improvement, including 49 (85%) with a complete response as defined by a CLASI (Cutaneous Lupus Erythematosus Disease Area and Severity Index) activity score of 0.
Relapse occurred in most patients, usually about 5 months after thalidomide dose reduction or withdrawal. Patients with subacute CLE were 30-fold more likely to remain in remission after drug discontinuation; those with discoid LE were at increased risk for relapse (Br. J. Dermatol. 2012;166:616-23).
Of particular concern to Dr. Werth was the finding that two patients had a stroke while on the drug. Neither had antiphospholipid antibodies, and one was quite young to have had a stroke, although both were heavy smokers.
Prescribing a drug such as thalidomide that promotes a hypercoagulable state to patients with refractory CLE is problematic because they often already have multiple risk factors for thrombosis. For one thing, treatment-refractory CLE patients tend to be smokers. Many of them are women on oral contraceptives. And there is an increased prevalence of antiphospholipid antibodies in patients with CLE, according to Dr. Werth.
Neuropathy is another major issue with thalidomide. In the Spanish study, 11 of 60 patients (18%) developed paresthesias; nerve conduction studies confirmed sensory polyneuropathy in 5 of the 11. Fortunately, the neurologic symptoms resolved in an average of 12 months after drug withdrawal.
Of course, thalidomide is a notorious teratogen. It can also cause premature ovarian failure, although this is usually reversible upon drug discontinuation.
"Obviously we need better therapies than thalidomide," Dr. Werth concluded.
Toward that end, interest is growing in thalidomide analogues as a novel potential therapy for refractory CLE. These analogues are up to 50,000 times more active than thalidomide, and are potentially less neurotoxic. One of them, lenalidomide (Revlimid), is marketed as a treatment for multiple myeloma and myelodysplastic syndrome. Others are in the development pipeline.
Several small observational studies have recently reported favorable results with lenalidomide in patients with refractory CLE. For example, investigators at Vall d’Hebron University Hospital in Barcelona reported on 15 patients treated open label with lenalidomide at 5-10 mg/day, with a follow-up of 15 months. One patient dropped out early due to side effects, but the other 14 saw clinical improvement within the first 2 weeks. Twelve patients, or 86%, achieved a CLASI score of 0. However, 9 of 12 complete responders experienced clinical relapse, usually 2-8 weeks after the drug was tapered and discontinued. Side effects were mild and infrequent, with no thrombosis or polyneuropathy (Arthritis Res. Ther. 2012;14:R265).
In another series, 4 of 5 lenalidomide-treated patients showed significant skin improvement, although one eventually developed symptoms of SLE (J. Am. Acad. Dermatol. 2012;66:571-82).
Based upon these and other promising reports, Celgene, which markets lenalidomide, recently launched the first-ever phase II study of a thalidomide analogue for the treatment of CLE.
Dr. Werth reported having no financial conflicts.
EXPERT ANALYSIS FROM THE ANNUAL CONGRESS OF THE EUROPEAN ACADEMY OF DERMATOLOGY AND VENEREOLOGY
Superficial Pyoderma Gangrenosum Treated With Infliximab: A Case Report
Psoriasis Patients Have Low Rates of Common Cancers
PRAGUE – Rates of three common malignancies – breast cancer, prostate cancer, and colorectal cancer – appear to be lower in psoriasis patients than in the general population of the Canadian maritime provinces of Newfoundland and Labrador, a study has shown.
In addition, only a single case of lymphoma occurred in 3,289 psoriasis patients followed for an average of 10 years, Dr. Wayne Gulliver said in presenting preliminary results of the ongoing study at the annual congress of the European Academy of Dermatology and Venereology.
The issue of a possible elevated risk of lymphoma in psoriasis patients has been controversial. Findings have been inconsistent. Some studies have reported a significantly increased risk. Among these, notably, was a population-based cohort study of more than 153,000 psoriasis patients and close to 800,000 controls in the U.K. General Practice Research Database. Investigators found an age- and sex-adjusted 35% increased risk of lymphoma in the group with psoriasis (J. Invest. Dermatol. 2006;126:2194-201).
However, lymphoma is an uncommon disease and a 35% increased risk is, from an epidemiologic perspective, only modest. Some studies have not found a significant association between psoriasis and lymphoma. The Canadian maritime study can be added to their ranks, according to Dr. Gulliver, professor of medicine and chairman of the division of dermatology at Memorial University of Newfoundland, St. John’s.
He matched records from a comprehensive research database of psoriasis patients in Newfoundland and Labrador for 1989-2010 with Canadian national and provincial cancer statistics. The purpose of the study was straightforward: "Our patients want to know if their psoriasis affects their risk of cancer, especially with the treatments we’re offering them these days," Dr. Gulliver said.
There were 232 cancer cases in 3,289 psoriasis patients, for a cumulative incidence rate of 588/100,000 population with psoriasis. Although the psoriasis population was divided nearly equally between men and women, 145 men developed cancer compared with 87 women.
Cancer rates were similar regardless of whether patients had mild psoriasis – meaning they received no systemic therapies – or moderate to severe psoriasis.
The cumulative incidence rate of breast cancer among psoriasis patients was 52/100,000 patients with mild psoriasis and 64/100,000 with moderate to severe disease compared with 96/100,000 in the general Canadian population and 84/100,000 among residents of Newfoundland and Labrador. The prostate cancer rate was 103/100,000 patients with mild psoriasis and 75/100,000 with moderate to severe psoriasis as opposed to 121/100,000 nationwide and 129/100,000 men in the general population of the two maritime provinces. The cumulative incidence rate for colorectal cancer followed the same pattern: lower in the psoriasis patients than in the general population.
Nonmelanoma skin cancer rates were relatively high: 142/100,000 patients with mild psoriasis and 122/100,000 with moderate to severe disease. That’s consistent with findings from other studies. It comes as no surprise given the common use of UV therapy and sunlight exposure in psoriasis patients.
Dr. Gulliver noted the relatively small number of cancers to date is a significant study limitation. But that number will grow as the psoriasis patients age; plus, more psoriasis patients are being added to the provincial database. He added that the database provides fertile ground for planned future studies. He said he plans soon to compare cancer rates in psoriasis patients on immunosuppressive therapy with biologic agents or cyclosporine to rates in patients on other agents.
One audience member observed that it seems more than coincidental that increased rates of breast, prostate, and colorectal cancers in the general population have previously been linked to low vitamin D levels. Perhaps the same factor that drives the increase in nonmelanoma skin cancer in psoriasis patients – that is, UV exposure – is responsible for their lower risks of those three common internal malignancies through a mechanism involving enhanced serum vitamin D levels, he speculated.
Dr. Gulliver called that a fascinating hypothesis and one he is now eager to investigate, since he has access to blood samples for the study population.
The study was free of commercial involvement, and the presenter reported having no relevant financial conflicts.
PRAGUE – Rates of three common malignancies – breast cancer, prostate cancer, and colorectal cancer – appear to be lower in psoriasis patients than in the general population of the Canadian maritime provinces of Newfoundland and Labrador, a study has shown.
In addition, only a single case of lymphoma occurred in 3,289 psoriasis patients followed for an average of 10 years, Dr. Wayne Gulliver said in presenting preliminary results of the ongoing study at the annual congress of the European Academy of Dermatology and Venereology.
The issue of a possible elevated risk of lymphoma in psoriasis patients has been controversial. Findings have been inconsistent. Some studies have reported a significantly increased risk. Among these, notably, was a population-based cohort study of more than 153,000 psoriasis patients and close to 800,000 controls in the U.K. General Practice Research Database. Investigators found an age- and sex-adjusted 35% increased risk of lymphoma in the group with psoriasis (J. Invest. Dermatol. 2006;126:2194-201).
However, lymphoma is an uncommon disease and a 35% increased risk is, from an epidemiologic perspective, only modest. Some studies have not found a significant association between psoriasis and lymphoma. The Canadian maritime study can be added to their ranks, according to Dr. Gulliver, professor of medicine and chairman of the division of dermatology at Memorial University of Newfoundland, St. John’s.
He matched records from a comprehensive research database of psoriasis patients in Newfoundland and Labrador for 1989-2010 with Canadian national and provincial cancer statistics. The purpose of the study was straightforward: "Our patients want to know if their psoriasis affects their risk of cancer, especially with the treatments we’re offering them these days," Dr. Gulliver said.
There were 232 cancer cases in 3,289 psoriasis patients, for a cumulative incidence rate of 588/100,000 population with psoriasis. Although the psoriasis population was divided nearly equally between men and women, 145 men developed cancer compared with 87 women.
Cancer rates were similar regardless of whether patients had mild psoriasis – meaning they received no systemic therapies – or moderate to severe psoriasis.
The cumulative incidence rate of breast cancer among psoriasis patients was 52/100,000 patients with mild psoriasis and 64/100,000 with moderate to severe disease compared with 96/100,000 in the general Canadian population and 84/100,000 among residents of Newfoundland and Labrador. The prostate cancer rate was 103/100,000 patients with mild psoriasis and 75/100,000 with moderate to severe psoriasis as opposed to 121/100,000 nationwide and 129/100,000 men in the general population of the two maritime provinces. The cumulative incidence rate for colorectal cancer followed the same pattern: lower in the psoriasis patients than in the general population.
Nonmelanoma skin cancer rates were relatively high: 142/100,000 patients with mild psoriasis and 122/100,000 with moderate to severe disease. That’s consistent with findings from other studies. It comes as no surprise given the common use of UV therapy and sunlight exposure in psoriasis patients.
Dr. Gulliver noted the relatively small number of cancers to date is a significant study limitation. But that number will grow as the psoriasis patients age; plus, more psoriasis patients are being added to the provincial database. He added that the database provides fertile ground for planned future studies. He said he plans soon to compare cancer rates in psoriasis patients on immunosuppressive therapy with biologic agents or cyclosporine to rates in patients on other agents.
One audience member observed that it seems more than coincidental that increased rates of breast, prostate, and colorectal cancers in the general population have previously been linked to low vitamin D levels. Perhaps the same factor that drives the increase in nonmelanoma skin cancer in psoriasis patients – that is, UV exposure – is responsible for their lower risks of those three common internal malignancies through a mechanism involving enhanced serum vitamin D levels, he speculated.
Dr. Gulliver called that a fascinating hypothesis and one he is now eager to investigate, since he has access to blood samples for the study population.
The study was free of commercial involvement, and the presenter reported having no relevant financial conflicts.
PRAGUE – Rates of three common malignancies – breast cancer, prostate cancer, and colorectal cancer – appear to be lower in psoriasis patients than in the general population of the Canadian maritime provinces of Newfoundland and Labrador, a study has shown.
In addition, only a single case of lymphoma occurred in 3,289 psoriasis patients followed for an average of 10 years, Dr. Wayne Gulliver said in presenting preliminary results of the ongoing study at the annual congress of the European Academy of Dermatology and Venereology.
The issue of a possible elevated risk of lymphoma in psoriasis patients has been controversial. Findings have been inconsistent. Some studies have reported a significantly increased risk. Among these, notably, was a population-based cohort study of more than 153,000 psoriasis patients and close to 800,000 controls in the U.K. General Practice Research Database. Investigators found an age- and sex-adjusted 35% increased risk of lymphoma in the group with psoriasis (J. Invest. Dermatol. 2006;126:2194-201).
However, lymphoma is an uncommon disease and a 35% increased risk is, from an epidemiologic perspective, only modest. Some studies have not found a significant association between psoriasis and lymphoma. The Canadian maritime study can be added to their ranks, according to Dr. Gulliver, professor of medicine and chairman of the division of dermatology at Memorial University of Newfoundland, St. John’s.
He matched records from a comprehensive research database of psoriasis patients in Newfoundland and Labrador for 1989-2010 with Canadian national and provincial cancer statistics. The purpose of the study was straightforward: "Our patients want to know if their psoriasis affects their risk of cancer, especially with the treatments we’re offering them these days," Dr. Gulliver said.
There were 232 cancer cases in 3,289 psoriasis patients, for a cumulative incidence rate of 588/100,000 population with psoriasis. Although the psoriasis population was divided nearly equally between men and women, 145 men developed cancer compared with 87 women.
Cancer rates were similar regardless of whether patients had mild psoriasis – meaning they received no systemic therapies – or moderate to severe psoriasis.
The cumulative incidence rate of breast cancer among psoriasis patients was 52/100,000 patients with mild psoriasis and 64/100,000 with moderate to severe disease compared with 96/100,000 in the general Canadian population and 84/100,000 among residents of Newfoundland and Labrador. The prostate cancer rate was 103/100,000 patients with mild psoriasis and 75/100,000 with moderate to severe psoriasis as opposed to 121/100,000 nationwide and 129/100,000 men in the general population of the two maritime provinces. The cumulative incidence rate for colorectal cancer followed the same pattern: lower in the psoriasis patients than in the general population.
Nonmelanoma skin cancer rates were relatively high: 142/100,000 patients with mild psoriasis and 122/100,000 with moderate to severe disease. That’s consistent with findings from other studies. It comes as no surprise given the common use of UV therapy and sunlight exposure in psoriasis patients.
Dr. Gulliver noted the relatively small number of cancers to date is a significant study limitation. But that number will grow as the psoriasis patients age; plus, more psoriasis patients are being added to the provincial database. He added that the database provides fertile ground for planned future studies. He said he plans soon to compare cancer rates in psoriasis patients on immunosuppressive therapy with biologic agents or cyclosporine to rates in patients on other agents.
One audience member observed that it seems more than coincidental that increased rates of breast, prostate, and colorectal cancers in the general population have previously been linked to low vitamin D levels. Perhaps the same factor that drives the increase in nonmelanoma skin cancer in psoriasis patients – that is, UV exposure – is responsible for their lower risks of those three common internal malignancies through a mechanism involving enhanced serum vitamin D levels, he speculated.
Dr. Gulliver called that a fascinating hypothesis and one he is now eager to investigate, since he has access to blood samples for the study population.
The study was free of commercial involvement, and the presenter reported having no relevant financial conflicts.
AT THE ANNUAL CONGRESS OF THE EUROPEAN ACADEMY OF DERMATOLOGY AND VENEREOLOGY
Major Finding: There were 232 cancer cases in 3,289 psoriasis patients, for a cumulative incidence rate of 588/100,000 population with psoriasis, a finding that suggests these patients have lower-than-average risks of prostate, breast, and colorectal cancers.
Data Source: This was a population-based cohort study involving 3,289 psoriasis patients in Newfoundland and Labrador. Their risk of various cancers during roughly 32,000 person-years of follow-up was determined by analysis of the comprehensive provincial and national cancer registries.
Disclosures: The study was free of commercial involvement, and the presenter reported having no relevant financial conflicts.
Anti-Jo1 Predicts Improvement With Rituximab
WASHINGTON – The presence of the antisynthetase antibody Jo-1, as well as Mi-2 autoantibodies, strongly predicted improvement in rituximab-treated myositis patients.
On the other hand, global damage is a poor indicator of good response to rituximab in multivariate analysis, meaning that further research into these biomarkers – especially Jo-1 – could have a major impact on treatment in this population, reported Dr. Rohit Aggarwal at the annual meeting of the American College of Rheumatology.
Dr. Aggarwal of the myositis center of the University of Pittsburgh and his colleagues looked at data from the RIM study (Rituximab in Myositis), which evaluated 200 myositis patients treated with the B-cell depleting agent, 83% of whom met the "definition of improvement" (DOI).
The DOI included a 20% or greater improvement in three of six measures: manual muscle testing, muscle enzymes, health assessment questionnaire, patient/parent global assessment, physician global disease activity assessment, and extramuscular disease activity, with not more than 25% worsening of greater than two measures.
These improvements also had to be noted at two consecutive visits to count as meeting the DOI.
Overall, 76 of the 200 myositis patients were more specifically classified as having adult polymyositis; 76 had adult dermatomyositis; and the remaining 48 patients had juvenile dermatomyositis.
In univariate analysis, several factors were associated with more rapid achievement of the DOI. These included lower baseline muscle damage, a higher white blood cell count, lower global damage, and the absence of muscle atrophy.
However, "the primary and strongest predictor of improvement on univariate analysis was autoantibodies," said Dr. Aggarwal – specifically, Jo-1 and Mi-2 – but also other myositis-associated antibodies, including SRP.
Dr. Aggarwal then conducted a multivariate analysis looking only at those variables with P value less than .1 in univariate assessment.
This time, the only significant predictors of improvement were the presence of the antisynthetase autoantibody Jo-1, but only in adults (hazard ratio, 3.08; 95% confidence interval, 1.80-5.28) as well as Mi-2, but only in juvenile and adult dermatomyositis – not adult polymyositis (HR 2.49; 95% CI, 1.42-4.41).
Other antibodies, including those to the signal recognition particle, did not confer a significantly increased association with improvement.
On the other hand, having less global damage at baseline was associated with improvement at 8 weeks, but the effect was washed out by week 20.
"Our findings regarding anti–Jo-1 are even more interesting when these results are coupled with other data that were presented in a concurrent session yesterday," said Dr. Aggarwal, citing a study he also led looking at the relation between anti–Jo-1 serum levels and the six improvement measures assessed in the DOI.
In that study, "After start of the treatment [with rituximab], autoantibody levels in Jo-1 subjects decreased by approximately nine units per week," he wrote.
"Anti–Jo-1 levels longitudinally correlated with all core set measures [of improvement] univariately and after adjusting for IgG levels."
He added, "These data suggest that anti–Jo-1 is also a disease biomarker."
"Further study of B-cell depletion in autoantibody positive, low-damage and juvenile dermatomyositis is warranted."
The RIM study was funded by Genentech, maker of rituximab; several investigators reported relationships to Genentech as well as other pharmaceutical companies.
WASHINGTON – The presence of the antisynthetase antibody Jo-1, as well as Mi-2 autoantibodies, strongly predicted improvement in rituximab-treated myositis patients.
On the other hand, global damage is a poor indicator of good response to rituximab in multivariate analysis, meaning that further research into these biomarkers – especially Jo-1 – could have a major impact on treatment in this population, reported Dr. Rohit Aggarwal at the annual meeting of the American College of Rheumatology.
Dr. Aggarwal of the myositis center of the University of Pittsburgh and his colleagues looked at data from the RIM study (Rituximab in Myositis), which evaluated 200 myositis patients treated with the B-cell depleting agent, 83% of whom met the "definition of improvement" (DOI).
The DOI included a 20% or greater improvement in three of six measures: manual muscle testing, muscle enzymes, health assessment questionnaire, patient/parent global assessment, physician global disease activity assessment, and extramuscular disease activity, with not more than 25% worsening of greater than two measures.
These improvements also had to be noted at two consecutive visits to count as meeting the DOI.
Overall, 76 of the 200 myositis patients were more specifically classified as having adult polymyositis; 76 had adult dermatomyositis; and the remaining 48 patients had juvenile dermatomyositis.
In univariate analysis, several factors were associated with more rapid achievement of the DOI. These included lower baseline muscle damage, a higher white blood cell count, lower global damage, and the absence of muscle atrophy.
However, "the primary and strongest predictor of improvement on univariate analysis was autoantibodies," said Dr. Aggarwal – specifically, Jo-1 and Mi-2 – but also other myositis-associated antibodies, including SRP.
Dr. Aggarwal then conducted a multivariate analysis looking only at those variables with P value less than .1 in univariate assessment.
This time, the only significant predictors of improvement were the presence of the antisynthetase autoantibody Jo-1, but only in adults (hazard ratio, 3.08; 95% confidence interval, 1.80-5.28) as well as Mi-2, but only in juvenile and adult dermatomyositis – not adult polymyositis (HR 2.49; 95% CI, 1.42-4.41).
Other antibodies, including those to the signal recognition particle, did not confer a significantly increased association with improvement.
On the other hand, having less global damage at baseline was associated with improvement at 8 weeks, but the effect was washed out by week 20.
"Our findings regarding anti–Jo-1 are even more interesting when these results are coupled with other data that were presented in a concurrent session yesterday," said Dr. Aggarwal, citing a study he also led looking at the relation between anti–Jo-1 serum levels and the six improvement measures assessed in the DOI.
In that study, "After start of the treatment [with rituximab], autoantibody levels in Jo-1 subjects decreased by approximately nine units per week," he wrote.
"Anti–Jo-1 levels longitudinally correlated with all core set measures [of improvement] univariately and after adjusting for IgG levels."
He added, "These data suggest that anti–Jo-1 is also a disease biomarker."
"Further study of B-cell depletion in autoantibody positive, low-damage and juvenile dermatomyositis is warranted."
The RIM study was funded by Genentech, maker of rituximab; several investigators reported relationships to Genentech as well as other pharmaceutical companies.
WASHINGTON – The presence of the antisynthetase antibody Jo-1, as well as Mi-2 autoantibodies, strongly predicted improvement in rituximab-treated myositis patients.
On the other hand, global damage is a poor indicator of good response to rituximab in multivariate analysis, meaning that further research into these biomarkers – especially Jo-1 – could have a major impact on treatment in this population, reported Dr. Rohit Aggarwal at the annual meeting of the American College of Rheumatology.
Dr. Aggarwal of the myositis center of the University of Pittsburgh and his colleagues looked at data from the RIM study (Rituximab in Myositis), which evaluated 200 myositis patients treated with the B-cell depleting agent, 83% of whom met the "definition of improvement" (DOI).
The DOI included a 20% or greater improvement in three of six measures: manual muscle testing, muscle enzymes, health assessment questionnaire, patient/parent global assessment, physician global disease activity assessment, and extramuscular disease activity, with not more than 25% worsening of greater than two measures.
These improvements also had to be noted at two consecutive visits to count as meeting the DOI.
Overall, 76 of the 200 myositis patients were more specifically classified as having adult polymyositis; 76 had adult dermatomyositis; and the remaining 48 patients had juvenile dermatomyositis.
In univariate analysis, several factors were associated with more rapid achievement of the DOI. These included lower baseline muscle damage, a higher white blood cell count, lower global damage, and the absence of muscle atrophy.
However, "the primary and strongest predictor of improvement on univariate analysis was autoantibodies," said Dr. Aggarwal – specifically, Jo-1 and Mi-2 – but also other myositis-associated antibodies, including SRP.
Dr. Aggarwal then conducted a multivariate analysis looking only at those variables with P value less than .1 in univariate assessment.
This time, the only significant predictors of improvement were the presence of the antisynthetase autoantibody Jo-1, but only in adults (hazard ratio, 3.08; 95% confidence interval, 1.80-5.28) as well as Mi-2, but only in juvenile and adult dermatomyositis – not adult polymyositis (HR 2.49; 95% CI, 1.42-4.41).
Other antibodies, including those to the signal recognition particle, did not confer a significantly increased association with improvement.
On the other hand, having less global damage at baseline was associated with improvement at 8 weeks, but the effect was washed out by week 20.
"Our findings regarding anti–Jo-1 are even more interesting when these results are coupled with other data that were presented in a concurrent session yesterday," said Dr. Aggarwal, citing a study he also led looking at the relation between anti–Jo-1 serum levels and the six improvement measures assessed in the DOI.
In that study, "After start of the treatment [with rituximab], autoantibody levels in Jo-1 subjects decreased by approximately nine units per week," he wrote.
"Anti–Jo-1 levels longitudinally correlated with all core set measures [of improvement] univariately and after adjusting for IgG levels."
He added, "These data suggest that anti–Jo-1 is also a disease biomarker."
"Further study of B-cell depletion in autoantibody positive, low-damage and juvenile dermatomyositis is warranted."
The RIM study was funded by Genentech, maker of rituximab; several investigators reported relationships to Genentech as well as other pharmaceutical companies.
AT THE ANNUAL MEETING OF THE AMERICAN COLLEGE OF RHEUMATOLOGY
Major Finding: Presence of the anti-synthetase autoantibody Jo-1 at baseline strongly predicted improvement in myositis patients taking rituximab (hazard ratio 3.08; 95% confidence interval, 1.80-5.28).
Data Source: A This findings is based on data from the RIM (Rituximab in Myositis) study.
Disclosures: The RIM study was funded by Genentech, maker of rituximab; several investigators reported relationships to Genentech as well as other pharmaceutical companies.
Subcutaneous Nodules Linked to Cardiovascular Disease Risk in RA
WASHINGTON – Subcutaneous nodules in patients with rheumatoid arthritis signal a significantly increased risk of cardiovascular disease, based on findings from the CORRONA database.
Although subcutaneous nodules are associated with more severe rheumatoid arthritis (RA), and RA is known to be associated with an increased risk of cardiovascular disease, the findings are the first to demonstrate a link between these "conspicuous, accessible, and assessable clinical markers" and cardiovascular morbidity in RA, Dr. Prashant Kaushik reported at the annual meeting of the American College of Rheumatology.
In 23,327 RA patients included in the CORRONA (Consortium of Rheumatology Researchers of North America) database who were evaluated at 182,201 individual visits between October 2001 and September 2011, the presence of subcutaneous nodules was found to confer a 44% increase in the risk of cardiovascular disease (hazard ratio, 1.44). The association remained significant after the investigators adjusted for age, sex, age of onset of RA, presence of diabetes mellitus, hypertension, smoking, alcohol consumption, and the use of a lipid-lowering agent (adjusted hazard ratio, 1.25), said Dr. Kaushik, who is rheumatology section chief at the Stratton VA Medical Center, Albany, N.Y.
Additionally, an age-sex interaction was found for women, who had a steeper increase in risk with age, compared with men. The hazard ratios per year of age for men and women were 1.01 and 1.03, but the hazard ratio for women vs. men at age 40 was 0.39, and at age 70 was 0.59, he noted.
Patients included in the CORRONA database had an average follow-up of 3 years and 70,455 patient-years. Nearly a third had subcutaneous nodules, and 795 had a cardiovascular event including a myocardial infarction, stroke or transient ischemic attack, heart failure, and/or cardiovascular death.
While the precise mechanism of the biologic association still needs to be ascertained, these findings indicate that subcutaneous nodules, which are the most common extra-articular manifestation of RA, should serve as a red flag for clinicians, Dr. Kaushik said.
"Subcutaneous nodules may be thought of as a clinical indicator of cardiovascular disease in RA," he concluded.
Dr. Kaushik reported having no relevant financial disclosures.
WASHINGTON – Subcutaneous nodules in patients with rheumatoid arthritis signal a significantly increased risk of cardiovascular disease, based on findings from the CORRONA database.
Although subcutaneous nodules are associated with more severe rheumatoid arthritis (RA), and RA is known to be associated with an increased risk of cardiovascular disease, the findings are the first to demonstrate a link between these "conspicuous, accessible, and assessable clinical markers" and cardiovascular morbidity in RA, Dr. Prashant Kaushik reported at the annual meeting of the American College of Rheumatology.
In 23,327 RA patients included in the CORRONA (Consortium of Rheumatology Researchers of North America) database who were evaluated at 182,201 individual visits between October 2001 and September 2011, the presence of subcutaneous nodules was found to confer a 44% increase in the risk of cardiovascular disease (hazard ratio, 1.44). The association remained significant after the investigators adjusted for age, sex, age of onset of RA, presence of diabetes mellitus, hypertension, smoking, alcohol consumption, and the use of a lipid-lowering agent (adjusted hazard ratio, 1.25), said Dr. Kaushik, who is rheumatology section chief at the Stratton VA Medical Center, Albany, N.Y.
Additionally, an age-sex interaction was found for women, who had a steeper increase in risk with age, compared with men. The hazard ratios per year of age for men and women were 1.01 and 1.03, but the hazard ratio for women vs. men at age 40 was 0.39, and at age 70 was 0.59, he noted.
Patients included in the CORRONA database had an average follow-up of 3 years and 70,455 patient-years. Nearly a third had subcutaneous nodules, and 795 had a cardiovascular event including a myocardial infarction, stroke or transient ischemic attack, heart failure, and/or cardiovascular death.
While the precise mechanism of the biologic association still needs to be ascertained, these findings indicate that subcutaneous nodules, which are the most common extra-articular manifestation of RA, should serve as a red flag for clinicians, Dr. Kaushik said.
"Subcutaneous nodules may be thought of as a clinical indicator of cardiovascular disease in RA," he concluded.
Dr. Kaushik reported having no relevant financial disclosures.
WASHINGTON – Subcutaneous nodules in patients with rheumatoid arthritis signal a significantly increased risk of cardiovascular disease, based on findings from the CORRONA database.
Although subcutaneous nodules are associated with more severe rheumatoid arthritis (RA), and RA is known to be associated with an increased risk of cardiovascular disease, the findings are the first to demonstrate a link between these "conspicuous, accessible, and assessable clinical markers" and cardiovascular morbidity in RA, Dr. Prashant Kaushik reported at the annual meeting of the American College of Rheumatology.
In 23,327 RA patients included in the CORRONA (Consortium of Rheumatology Researchers of North America) database who were evaluated at 182,201 individual visits between October 2001 and September 2011, the presence of subcutaneous nodules was found to confer a 44% increase in the risk of cardiovascular disease (hazard ratio, 1.44). The association remained significant after the investigators adjusted for age, sex, age of onset of RA, presence of diabetes mellitus, hypertension, smoking, alcohol consumption, and the use of a lipid-lowering agent (adjusted hazard ratio, 1.25), said Dr. Kaushik, who is rheumatology section chief at the Stratton VA Medical Center, Albany, N.Y.
Additionally, an age-sex interaction was found for women, who had a steeper increase in risk with age, compared with men. The hazard ratios per year of age for men and women were 1.01 and 1.03, but the hazard ratio for women vs. men at age 40 was 0.39, and at age 70 was 0.59, he noted.
Patients included in the CORRONA database had an average follow-up of 3 years and 70,455 patient-years. Nearly a third had subcutaneous nodules, and 795 had a cardiovascular event including a myocardial infarction, stroke or transient ischemic attack, heart failure, and/or cardiovascular death.
While the precise mechanism of the biologic association still needs to be ascertained, these findings indicate that subcutaneous nodules, which are the most common extra-articular manifestation of RA, should serve as a red flag for clinicians, Dr. Kaushik said.
"Subcutaneous nodules may be thought of as a clinical indicator of cardiovascular disease in RA," he concluded.
Dr. Kaushik reported having no relevant financial disclosures.
AT THE ANNUAL MEETING OF THE AMERICAN COLLEGE OF RHEUMATOLOGY
Major Finding: In 23,327 patients with RA, the presence of subcutaneous nodules was found to confer a 44% increase in the risk of cardiovascular disease (hazard ratio, 1.44). The association remained significant after adjusting for age, sex, age of onset of RA, presence of diabetes mellitus, hypertension, smoking, alcohol consumption, and use of a lipid-lowering agent (adjusted hazard ratio, 1.25).
Data Source: The findings come from the CORRONA database.
Disclosures: Dr. Kaushik said he had no relevant financial disclosures.
Free Pharma Pens: The Skinny Podcast
In this month's program, Dr. Craig Leonardi discusses the link between psoriasis and diabetes, and what it means for your patients.
Reporter Mary Ellen Schneider talks to Dr. Henry Lim about the American Academy of Dermatology's top research priorities.
We also bring you an interview with a physician that believes flu vaccination is an ethical responsibility.
In this month's Cosmetic Counter segment, Dr. Lily Talakoub explains why she believes paramedical micropigmentation should be part of every dermatologist's practice.
And to wrap it up, Dr. Alan Rockoff jots down a few notes with his free drug company pens.
In this month's program, Dr. Craig Leonardi discusses the link between psoriasis and diabetes, and what it means for your patients.
Reporter Mary Ellen Schneider talks to Dr. Henry Lim about the American Academy of Dermatology's top research priorities.
We also bring you an interview with a physician that believes flu vaccination is an ethical responsibility.
In this month's Cosmetic Counter segment, Dr. Lily Talakoub explains why she believes paramedical micropigmentation should be part of every dermatologist's practice.
And to wrap it up, Dr. Alan Rockoff jots down a few notes with his free drug company pens.
In this month's program, Dr. Craig Leonardi discusses the link between psoriasis and diabetes, and what it means for your patients.
Reporter Mary Ellen Schneider talks to Dr. Henry Lim about the American Academy of Dermatology's top research priorities.
We also bring you an interview with a physician that believes flu vaccination is an ethical responsibility.
In this month's Cosmetic Counter segment, Dr. Lily Talakoub explains why she believes paramedical micropigmentation should be part of every dermatologist's practice.
And to wrap it up, Dr. Alan Rockoff jots down a few notes with his free drug company pens.
Ustekinumab's Cardiovascular Event Rates Stable at 5 Years
LAS VEGAS – Cardiovascular event rates in patients treated with ustekinumab have been found to remain stable over time, according to 5-year follow-up data from the manufacturer.
"The overall safety profile of ustekinumab [Stelara] remained stable in adults with moderate to severe plaque psoriasis receiving up to 5 years of ustekinumab treatment," noted the manufacturer of Stelara, Janssen Pharmaceuticals, in a statement. "There was no dose-response or cumulative effect of increasing duration of exposure to ustekinumab on the rates of overall and targeted adverse events after up to 5 years of treatment."
The cardiovascular safety of the plaque psoriasis treatment has been a concern because early testing detected a slightly higher incidence of events in patients treated with ustekinumab, compared with placebo.
But among 3,117 patients – 838 of whom were on the biologic for 5 years – and after a cumulative total of 8,998 patient-years, the rate of major adverse cardiovascular events (MACEs) held largely steady at 0.47 per 100 patient-years at year 1, 0.44 at year 5, and comparable numbers in between. The data were pooled from one phase II and three phase III trials, with MACE defined as stroke, myocardial infarction, or cardiovascular death.
The MACE rate at 5-year follow-up was 0.56 per 100 patient-years (95% confidence interval, 0.35-0.85) among patients receiving the 45-mg dose of the injectable and 0.36 per 100 patient-years (95% CI, 0.22-0.57) in patients receiving 90 mg.
In comparable 4-year follow-up data, researchers found that MACE and other serious adverse event rates among ustekinumab users "were consistent with those in the general and psoriasis populations" (J. Drugs Dermatol. 2012;11:300-12).
"The cardiovascular events over time seem to be stable," said Dr. Craig Leonardi, of the dermatology department at St. Louis University, who summarized the 5-year findings during a talk about the drug.
Even so, Dr. Leonardi, who presented the study results at a seminar sponsored by Skin Disease Education Foundation (SDEF), said that he remains concerned, noting that follow-up studies select for patients who do well on a drug and have minimal adverse events.
It’s possible that patients susceptible to MACEs – those with preexisting cardiac risk factors or, perhaps, an as-yet unrecognized genetic predisposition – dropped out early due to angina or other problems.
Dr. Leonardi also noted a recent meta-analysis that found a higher risk of MACEs in patients treated with ustekinumab or briakinumab – another interleukin-12/23 antibody no longer under development – compared with those on placebo (J. Eur. Acad. Dermatol. Venereol. 2012 March 8 [doi: 10.1111/j.1468-3083.2012.04500]).
It followed an earlier meta-analysis, conducted by Dr. Leonardi and his colleagues, of 22 trials that found 10 MACEs among 3,179 patients on ustekinumab or briakinumab but none among 1,474 placebo patients (JAMA 2011;306:864-71). Even though the results were not statistically significant, "there was a trend, and we are concerned about it. It seems to correspond to peak levels of the drug. We are awaiting further analysis," Dr. Leonardi said.
There’s "no question [ustekinumab] is a high-performance drug with a durable response," but in the meantime, "I am putting all my ustekinumab patients on aspirin, 81 mg/day," he said.
Patients with multiple cardiovascular risk factors should start with a tumor necrosis factor antagonist instead, according to Dr. Leonardi. "These are drugs with proven cardiovascular friendliness," he added. It is also probably wise to start out with the lower 45-mg dose of ustekinumab when initiating treatment.
Dr. Leonardi is a consultant, investigator, and speaker for Abbott, Amgen, and other companies. SDEF and this news organization are owned by Frontline Medical Communications.
LAS VEGAS – Cardiovascular event rates in patients treated with ustekinumab have been found to remain stable over time, according to 5-year follow-up data from the manufacturer.
"The overall safety profile of ustekinumab [Stelara] remained stable in adults with moderate to severe plaque psoriasis receiving up to 5 years of ustekinumab treatment," noted the manufacturer of Stelara, Janssen Pharmaceuticals, in a statement. "There was no dose-response or cumulative effect of increasing duration of exposure to ustekinumab on the rates of overall and targeted adverse events after up to 5 years of treatment."
The cardiovascular safety of the plaque psoriasis treatment has been a concern because early testing detected a slightly higher incidence of events in patients treated with ustekinumab, compared with placebo.
But among 3,117 patients – 838 of whom were on the biologic for 5 years – and after a cumulative total of 8,998 patient-years, the rate of major adverse cardiovascular events (MACEs) held largely steady at 0.47 per 100 patient-years at year 1, 0.44 at year 5, and comparable numbers in between. The data were pooled from one phase II and three phase III trials, with MACE defined as stroke, myocardial infarction, or cardiovascular death.
The MACE rate at 5-year follow-up was 0.56 per 100 patient-years (95% confidence interval, 0.35-0.85) among patients receiving the 45-mg dose of the injectable and 0.36 per 100 patient-years (95% CI, 0.22-0.57) in patients receiving 90 mg.
In comparable 4-year follow-up data, researchers found that MACE and other serious adverse event rates among ustekinumab users "were consistent with those in the general and psoriasis populations" (J. Drugs Dermatol. 2012;11:300-12).
"The cardiovascular events over time seem to be stable," said Dr. Craig Leonardi, of the dermatology department at St. Louis University, who summarized the 5-year findings during a talk about the drug.
Even so, Dr. Leonardi, who presented the study results at a seminar sponsored by Skin Disease Education Foundation (SDEF), said that he remains concerned, noting that follow-up studies select for patients who do well on a drug and have minimal adverse events.
It’s possible that patients susceptible to MACEs – those with preexisting cardiac risk factors or, perhaps, an as-yet unrecognized genetic predisposition – dropped out early due to angina or other problems.
Dr. Leonardi also noted a recent meta-analysis that found a higher risk of MACEs in patients treated with ustekinumab or briakinumab – another interleukin-12/23 antibody no longer under development – compared with those on placebo (J. Eur. Acad. Dermatol. Venereol. 2012 March 8 [doi: 10.1111/j.1468-3083.2012.04500]).
It followed an earlier meta-analysis, conducted by Dr. Leonardi and his colleagues, of 22 trials that found 10 MACEs among 3,179 patients on ustekinumab or briakinumab but none among 1,474 placebo patients (JAMA 2011;306:864-71). Even though the results were not statistically significant, "there was a trend, and we are concerned about it. It seems to correspond to peak levels of the drug. We are awaiting further analysis," Dr. Leonardi said.
There’s "no question [ustekinumab] is a high-performance drug with a durable response," but in the meantime, "I am putting all my ustekinumab patients on aspirin, 81 mg/day," he said.
Patients with multiple cardiovascular risk factors should start with a tumor necrosis factor antagonist instead, according to Dr. Leonardi. "These are drugs with proven cardiovascular friendliness," he added. It is also probably wise to start out with the lower 45-mg dose of ustekinumab when initiating treatment.
Dr. Leonardi is a consultant, investigator, and speaker for Abbott, Amgen, and other companies. SDEF and this news organization are owned by Frontline Medical Communications.
LAS VEGAS – Cardiovascular event rates in patients treated with ustekinumab have been found to remain stable over time, according to 5-year follow-up data from the manufacturer.
"The overall safety profile of ustekinumab [Stelara] remained stable in adults with moderate to severe plaque psoriasis receiving up to 5 years of ustekinumab treatment," noted the manufacturer of Stelara, Janssen Pharmaceuticals, in a statement. "There was no dose-response or cumulative effect of increasing duration of exposure to ustekinumab on the rates of overall and targeted adverse events after up to 5 years of treatment."
The cardiovascular safety of the plaque psoriasis treatment has been a concern because early testing detected a slightly higher incidence of events in patients treated with ustekinumab, compared with placebo.
But among 3,117 patients – 838 of whom were on the biologic for 5 years – and after a cumulative total of 8,998 patient-years, the rate of major adverse cardiovascular events (MACEs) held largely steady at 0.47 per 100 patient-years at year 1, 0.44 at year 5, and comparable numbers in between. The data were pooled from one phase II and three phase III trials, with MACE defined as stroke, myocardial infarction, or cardiovascular death.
The MACE rate at 5-year follow-up was 0.56 per 100 patient-years (95% confidence interval, 0.35-0.85) among patients receiving the 45-mg dose of the injectable and 0.36 per 100 patient-years (95% CI, 0.22-0.57) in patients receiving 90 mg.
In comparable 4-year follow-up data, researchers found that MACE and other serious adverse event rates among ustekinumab users "were consistent with those in the general and psoriasis populations" (J. Drugs Dermatol. 2012;11:300-12).
"The cardiovascular events over time seem to be stable," said Dr. Craig Leonardi, of the dermatology department at St. Louis University, who summarized the 5-year findings during a talk about the drug.
Even so, Dr. Leonardi, who presented the study results at a seminar sponsored by Skin Disease Education Foundation (SDEF), said that he remains concerned, noting that follow-up studies select for patients who do well on a drug and have minimal adverse events.
It’s possible that patients susceptible to MACEs – those with preexisting cardiac risk factors or, perhaps, an as-yet unrecognized genetic predisposition – dropped out early due to angina or other problems.
Dr. Leonardi also noted a recent meta-analysis that found a higher risk of MACEs in patients treated with ustekinumab or briakinumab – another interleukin-12/23 antibody no longer under development – compared with those on placebo (J. Eur. Acad. Dermatol. Venereol. 2012 March 8 [doi: 10.1111/j.1468-3083.2012.04500]).
It followed an earlier meta-analysis, conducted by Dr. Leonardi and his colleagues, of 22 trials that found 10 MACEs among 3,179 patients on ustekinumab or briakinumab but none among 1,474 placebo patients (JAMA 2011;306:864-71). Even though the results were not statistically significant, "there was a trend, and we are concerned about it. It seems to correspond to peak levels of the drug. We are awaiting further analysis," Dr. Leonardi said.
There’s "no question [ustekinumab] is a high-performance drug with a durable response," but in the meantime, "I am putting all my ustekinumab patients on aspirin, 81 mg/day," he said.
Patients with multiple cardiovascular risk factors should start with a tumor necrosis factor antagonist instead, according to Dr. Leonardi. "These are drugs with proven cardiovascular friendliness," he added. It is also probably wise to start out with the lower 45-mg dose of ustekinumab when initiating treatment.
Dr. Leonardi is a consultant, investigator, and speaker for Abbott, Amgen, and other companies. SDEF and this news organization are owned by Frontline Medical Communications.
AT THE SDEF LAS VEGAS DERMATOLOGY SEMINAR
Major Finding: Among 3,117 patients – 838 of whom were on the biologic for 5 years – and after a cumulative total of 8,998 patient-years, the rate of MACEs was 0.47 per 100 patient-years at year 1 and 0.44 at year 5.
Data Source: The results were based on 5-year follow-up data from the manufacturer.
Disclosures: Dr. Leonardi is a consultant, investigator, and speaker for Abbott, Amgen, and other companies. SDEF and this news organization are owned by Frontline Medical Communications.
Biologics May Reduce RA Patients' Risk of Early Death
WASHINGTON – Biologics use by rheumatoid arthritis patients was associated with a 25% reduction in the risk of premature death, compared with patients without exposure to biologics, based on data from a population-based study of more than 4,000 patients. The findings were presented at the annual meeting of the American College of Rheumatology.
Rheumatoid arthritis (RA) is associated with a twofold increase in premature mortality risk, primarily caused by cardiovascular disease, said Dr. Diane Lacaille of the Arthritis Research Centre of Canada, in Vancouver. Previous research suggests that the increased risk is linked to inflammation that affects other organs beyond the RA-affected joints, she said. Biologic agents have shown effectiveness in controlling the inflammation associated with joint damage in RA, but the drugs’ impact on reducing early mortality has not been well studied, Dr. Lacaille said.
Dr. Lacaille and her colleagues reviewed data from the Canadian Ministry of Health to identify all RA cases that used a biologic agent (anti–tumor necrosis factor (anti-TNF), rituximab, anakinra, or abatacept) during follow-up. The study population included 2,156 patients who used biologics and 2,156 controls matched for age, sex, and calendar year. The average age of the patients was 56 years, and 75% were women. The researchers obtained data on all health services used by the study participants – including medications, lab tests, and hospitalizations – between January 1996 and March 2006, with follow-up to March 2010.
"Exposure to biologics was associated with a reduced risk of death, with a hazard ratio of 0.26 [(95% confidence interval, 0.18-0.36), P less than or equal to .0001)], which means that the risk of death in the biologics users was a quarter that of the nonbiologic users," Dr. Lacaille said. Approximately 90% of the biologics users were using anti-TNF therapy, but the findings were consistent for all biologics in a multivariate analysis, she noted.
Overall, 573 deaths were noted during the study period, including 247 in the biologics group and 326 in the control group. The use of three previous disease-modifying antirheumatic drugs or a change in DMARD did not affect the results, Dr. Lacaille noted.
"I think it wouldn’t be accurate from an epidemiological point of view to say that biologics lead to a 75% reduction" in the risk of premature death, she said.
"But we can be confident that there was an association with a reduction in the reduced risk of death," she added.
The study was limited by the lack of randomization, which may have led to some selection bias, said Dr. Lacaille. But given the increased risk of early mortality associated with RA, the data have "important implications for health policy makers, health care providers, and people with RA," she said.
Approximately 28% of the patients had used more than one biologic, but the number or course of prior biologics had no apparent impact on the risk of death, she added.
Dr. Lacaille had no financial conflicts to disclose.
WASHINGTON – Biologics use by rheumatoid arthritis patients was associated with a 25% reduction in the risk of premature death, compared with patients without exposure to biologics, based on data from a population-based study of more than 4,000 patients. The findings were presented at the annual meeting of the American College of Rheumatology.
Rheumatoid arthritis (RA) is associated with a twofold increase in premature mortality risk, primarily caused by cardiovascular disease, said Dr. Diane Lacaille of the Arthritis Research Centre of Canada, in Vancouver. Previous research suggests that the increased risk is linked to inflammation that affects other organs beyond the RA-affected joints, she said. Biologic agents have shown effectiveness in controlling the inflammation associated with joint damage in RA, but the drugs’ impact on reducing early mortality has not been well studied, Dr. Lacaille said.
Dr. Lacaille and her colleagues reviewed data from the Canadian Ministry of Health to identify all RA cases that used a biologic agent (anti–tumor necrosis factor (anti-TNF), rituximab, anakinra, or abatacept) during follow-up. The study population included 2,156 patients who used biologics and 2,156 controls matched for age, sex, and calendar year. The average age of the patients was 56 years, and 75% were women. The researchers obtained data on all health services used by the study participants – including medications, lab tests, and hospitalizations – between January 1996 and March 2006, with follow-up to March 2010.
"Exposure to biologics was associated with a reduced risk of death, with a hazard ratio of 0.26 [(95% confidence interval, 0.18-0.36), P less than or equal to .0001)], which means that the risk of death in the biologics users was a quarter that of the nonbiologic users," Dr. Lacaille said. Approximately 90% of the biologics users were using anti-TNF therapy, but the findings were consistent for all biologics in a multivariate analysis, she noted.
Overall, 573 deaths were noted during the study period, including 247 in the biologics group and 326 in the control group. The use of three previous disease-modifying antirheumatic drugs or a change in DMARD did not affect the results, Dr. Lacaille noted.
"I think it wouldn’t be accurate from an epidemiological point of view to say that biologics lead to a 75% reduction" in the risk of premature death, she said.
"But we can be confident that there was an association with a reduction in the reduced risk of death," she added.
The study was limited by the lack of randomization, which may have led to some selection bias, said Dr. Lacaille. But given the increased risk of early mortality associated with RA, the data have "important implications for health policy makers, health care providers, and people with RA," she said.
Approximately 28% of the patients had used more than one biologic, but the number or course of prior biologics had no apparent impact on the risk of death, she added.
Dr. Lacaille had no financial conflicts to disclose.
WASHINGTON – Biologics use by rheumatoid arthritis patients was associated with a 25% reduction in the risk of premature death, compared with patients without exposure to biologics, based on data from a population-based study of more than 4,000 patients. The findings were presented at the annual meeting of the American College of Rheumatology.
Rheumatoid arthritis (RA) is associated with a twofold increase in premature mortality risk, primarily caused by cardiovascular disease, said Dr. Diane Lacaille of the Arthritis Research Centre of Canada, in Vancouver. Previous research suggests that the increased risk is linked to inflammation that affects other organs beyond the RA-affected joints, she said. Biologic agents have shown effectiveness in controlling the inflammation associated with joint damage in RA, but the drugs’ impact on reducing early mortality has not been well studied, Dr. Lacaille said.
Dr. Lacaille and her colleagues reviewed data from the Canadian Ministry of Health to identify all RA cases that used a biologic agent (anti–tumor necrosis factor (anti-TNF), rituximab, anakinra, or abatacept) during follow-up. The study population included 2,156 patients who used biologics and 2,156 controls matched for age, sex, and calendar year. The average age of the patients was 56 years, and 75% were women. The researchers obtained data on all health services used by the study participants – including medications, lab tests, and hospitalizations – between January 1996 and March 2006, with follow-up to March 2010.
"Exposure to biologics was associated with a reduced risk of death, with a hazard ratio of 0.26 [(95% confidence interval, 0.18-0.36), P less than or equal to .0001)], which means that the risk of death in the biologics users was a quarter that of the nonbiologic users," Dr. Lacaille said. Approximately 90% of the biologics users were using anti-TNF therapy, but the findings were consistent for all biologics in a multivariate analysis, she noted.
Overall, 573 deaths were noted during the study period, including 247 in the biologics group and 326 in the control group. The use of three previous disease-modifying antirheumatic drugs or a change in DMARD did not affect the results, Dr. Lacaille noted.
"I think it wouldn’t be accurate from an epidemiological point of view to say that biologics lead to a 75% reduction" in the risk of premature death, she said.
"But we can be confident that there was an association with a reduction in the reduced risk of death," she added.
The study was limited by the lack of randomization, which may have led to some selection bias, said Dr. Lacaille. But given the increased risk of early mortality associated with RA, the data have "important implications for health policy makers, health care providers, and people with RA," she said.
Approximately 28% of the patients had used more than one biologic, but the number or course of prior biologics had no apparent impact on the risk of death, she added.
Dr. Lacaille had no financial conflicts to disclose.
AT THE ANNUAL MEETING OF THE AMERICAN COLLEGE OF RHEUMATOLOGY
Major Finding: Exposure to biologics was associated with a 25% reduction in the risk of early death in rheumatoid arthritis patients.
Data Source: The data come from a population-based study of 4,312 adults that used administrative billing data from the Canadian Ministry of Health.
Disclosures: Dr. Lacaille had no financial conflicts to disclose.
ACE Inhibitors Up Mortality Risk When Given Prior to Scleroderma Renal Crisis
WASHINGTON – Exposure to angiotensin-converting enzyme inhibitors prior to the onset of renal crisis in patients with scleroderma increases the risk of death, according to 1-year findings from the prospective observational International Scleroderma Renal Crisis Survey.
The findings, which contrast with those from a preliminary analysis reported last year, suggest that clinicians caring for patients with systemic scleroderma should exercise caution when prescribing angiotensin-converting enzyme (ACE) inhibitors, Dr. Marie Hudson said at the annual meeting of the American College of Rheumatology, where she reported the survey results.
Of 75 patients who experienced scleroderma renal crisis (SRC) in the course of the study, 16 were taking an ACE inhibitor prior to onset of SRC. At the 1-year follow-up, 27 (36%) of the patients had died and 25% remained on dialysis. After adjusting for differences in prednisone exposure and history of systemic hypertension, ACE inhibitor exposure prior to SRC, compared with no such exposure, was associated with significantly increased risk of death (adjusted hazard ratio, 2.52), said Dr. Hudson of the division of rheumatology at McGill University, Montreal.
SRC is a rare but life-threatening complication of systemic sclerosis that typically presents with malignant hypertension and acute renal failure.
"Prior to the advent of ACE inhibitors, it was almost a universally deadly complication of scleroderma, but since the advent of ACE inhibitors, the outcomes of patients with scleroderma renal crisis have improved tremendously," Dr. Hudson said, noting also that some evidence suggest that the incidence of SRC has decreased over the past two decades as well – due, perhaps, to more liberal use of ACE inhibitors.
"So, given the benefits of ACE inhibitors to treat SRC along with this perceived decrease in the incidence of SRC, the prophylactic use of ACE inhibitors to prevent SRC has been considered. However, some would argue that there’s no clear physiologic rationale for this," she said, explaining that most patients with SRC are not hyperreninemic prior to renal crisis and that prophylactic treatment could mask hypertension and delay diagnosis, thus leading to worse outcomes in those who develop SRC.
In fact, recent retrospective data support the idea that those exposed prior to SRC may have worse outcomes, she noted.
The findings of this survey, which are important given the widespread availability of ACE inhibitors, confirm that, she said.
Patients included in the study were identified by physicians from numerous practices around the world who had agreed to participate in the survey. A total of 589 physicians were asked biweekly if they had made a diagnosis of SRC, and if so, they filled out a short case report form at that time and then submitted another 1 year later.
The patients had a mean age of 52 years, and 67% were women. Most (76%) had diffuse systemic scleroderma with a median disease duration of 1.5 years. SRC was hypertensive in 71 patients; only 5 patients had normotensive SRC, she noted.
The rate of prednisone use was surprisingly high at nearly 50% overall, but, more importantly, the mean daily dose was twice as high in the group of patients who were not exposed to ACE inhibitors prior to SRC (mean dose of 18 mg/day vs. 9 mg/day in the exposed group), Dr. Hudson said.
However, even after adjusting for prednisone use, the risk of death in exposed patients was more than twice that of nonexposed patients, and the difference was statistically significant, she said.
Although the precise risk of death after SRC remains uncertain, it does appear that caution when using ACE inhibitors in these patients is warranted, especially early in disease when the risk of SRC is the greatest, she said.
Dr. Hudson had no disclosures to report.
WASHINGTON – Exposure to angiotensin-converting enzyme inhibitors prior to the onset of renal crisis in patients with scleroderma increases the risk of death, according to 1-year findings from the prospective observational International Scleroderma Renal Crisis Survey.
The findings, which contrast with those from a preliminary analysis reported last year, suggest that clinicians caring for patients with systemic scleroderma should exercise caution when prescribing angiotensin-converting enzyme (ACE) inhibitors, Dr. Marie Hudson said at the annual meeting of the American College of Rheumatology, where she reported the survey results.
Of 75 patients who experienced scleroderma renal crisis (SRC) in the course of the study, 16 were taking an ACE inhibitor prior to onset of SRC. At the 1-year follow-up, 27 (36%) of the patients had died and 25% remained on dialysis. After adjusting for differences in prednisone exposure and history of systemic hypertension, ACE inhibitor exposure prior to SRC, compared with no such exposure, was associated with significantly increased risk of death (adjusted hazard ratio, 2.52), said Dr. Hudson of the division of rheumatology at McGill University, Montreal.
SRC is a rare but life-threatening complication of systemic sclerosis that typically presents with malignant hypertension and acute renal failure.
"Prior to the advent of ACE inhibitors, it was almost a universally deadly complication of scleroderma, but since the advent of ACE inhibitors, the outcomes of patients with scleroderma renal crisis have improved tremendously," Dr. Hudson said, noting also that some evidence suggest that the incidence of SRC has decreased over the past two decades as well – due, perhaps, to more liberal use of ACE inhibitors.
"So, given the benefits of ACE inhibitors to treat SRC along with this perceived decrease in the incidence of SRC, the prophylactic use of ACE inhibitors to prevent SRC has been considered. However, some would argue that there’s no clear physiologic rationale for this," she said, explaining that most patients with SRC are not hyperreninemic prior to renal crisis and that prophylactic treatment could mask hypertension and delay diagnosis, thus leading to worse outcomes in those who develop SRC.
In fact, recent retrospective data support the idea that those exposed prior to SRC may have worse outcomes, she noted.
The findings of this survey, which are important given the widespread availability of ACE inhibitors, confirm that, she said.
Patients included in the study were identified by physicians from numerous practices around the world who had agreed to participate in the survey. A total of 589 physicians were asked biweekly if they had made a diagnosis of SRC, and if so, they filled out a short case report form at that time and then submitted another 1 year later.
The patients had a mean age of 52 years, and 67% were women. Most (76%) had diffuse systemic scleroderma with a median disease duration of 1.5 years. SRC was hypertensive in 71 patients; only 5 patients had normotensive SRC, she noted.
The rate of prednisone use was surprisingly high at nearly 50% overall, but, more importantly, the mean daily dose was twice as high in the group of patients who were not exposed to ACE inhibitors prior to SRC (mean dose of 18 mg/day vs. 9 mg/day in the exposed group), Dr. Hudson said.
However, even after adjusting for prednisone use, the risk of death in exposed patients was more than twice that of nonexposed patients, and the difference was statistically significant, she said.
Although the precise risk of death after SRC remains uncertain, it does appear that caution when using ACE inhibitors in these patients is warranted, especially early in disease when the risk of SRC is the greatest, she said.
Dr. Hudson had no disclosures to report.
WASHINGTON – Exposure to angiotensin-converting enzyme inhibitors prior to the onset of renal crisis in patients with scleroderma increases the risk of death, according to 1-year findings from the prospective observational International Scleroderma Renal Crisis Survey.
The findings, which contrast with those from a preliminary analysis reported last year, suggest that clinicians caring for patients with systemic scleroderma should exercise caution when prescribing angiotensin-converting enzyme (ACE) inhibitors, Dr. Marie Hudson said at the annual meeting of the American College of Rheumatology, where she reported the survey results.
Of 75 patients who experienced scleroderma renal crisis (SRC) in the course of the study, 16 were taking an ACE inhibitor prior to onset of SRC. At the 1-year follow-up, 27 (36%) of the patients had died and 25% remained on dialysis. After adjusting for differences in prednisone exposure and history of systemic hypertension, ACE inhibitor exposure prior to SRC, compared with no such exposure, was associated with significantly increased risk of death (adjusted hazard ratio, 2.52), said Dr. Hudson of the division of rheumatology at McGill University, Montreal.
SRC is a rare but life-threatening complication of systemic sclerosis that typically presents with malignant hypertension and acute renal failure.
"Prior to the advent of ACE inhibitors, it was almost a universally deadly complication of scleroderma, but since the advent of ACE inhibitors, the outcomes of patients with scleroderma renal crisis have improved tremendously," Dr. Hudson said, noting also that some evidence suggest that the incidence of SRC has decreased over the past two decades as well – due, perhaps, to more liberal use of ACE inhibitors.
"So, given the benefits of ACE inhibitors to treat SRC along with this perceived decrease in the incidence of SRC, the prophylactic use of ACE inhibitors to prevent SRC has been considered. However, some would argue that there’s no clear physiologic rationale for this," she said, explaining that most patients with SRC are not hyperreninemic prior to renal crisis and that prophylactic treatment could mask hypertension and delay diagnosis, thus leading to worse outcomes in those who develop SRC.
In fact, recent retrospective data support the idea that those exposed prior to SRC may have worse outcomes, she noted.
The findings of this survey, which are important given the widespread availability of ACE inhibitors, confirm that, she said.
Patients included in the study were identified by physicians from numerous practices around the world who had agreed to participate in the survey. A total of 589 physicians were asked biweekly if they had made a diagnosis of SRC, and if so, they filled out a short case report form at that time and then submitted another 1 year later.
The patients had a mean age of 52 years, and 67% were women. Most (76%) had diffuse systemic scleroderma with a median disease duration of 1.5 years. SRC was hypertensive in 71 patients; only 5 patients had normotensive SRC, she noted.
The rate of prednisone use was surprisingly high at nearly 50% overall, but, more importantly, the mean daily dose was twice as high in the group of patients who were not exposed to ACE inhibitors prior to SRC (mean dose of 18 mg/day vs. 9 mg/day in the exposed group), Dr. Hudson said.
However, even after adjusting for prednisone use, the risk of death in exposed patients was more than twice that of nonexposed patients, and the difference was statistically significant, she said.
Although the precise risk of death after SRC remains uncertain, it does appear that caution when using ACE inhibitors in these patients is warranted, especially early in disease when the risk of SRC is the greatest, she said.
Dr. Hudson had no disclosures to report.
AT THE ANNUAL MEETING OF THE AMERICAN COLLEGE OF RHEUMATOLOGY
Major Finding: ACE inhibitor exposure prior to SRC, compared with no such exposure, was found to be associated with significantly increased risk of death (adjusted hazard ratio, 2.52).
Data Source: This finding comes from a prospective international cohort study (International Scleroderma Renal Crisis Survey).
Disclosures: Dr. Hudson had no disclosures to report.