User login
High BMI does not complicate postpartum tubal ligation
GRAPEVINE, TEXAS – Higher body mass index is not associated with increased morbidity in women undergoing postpartum tubal ligation, according to a study of more than 1,000 patients.
John J. Byrne, MD, said at the Pregnancy Meeting. Dr. Byrne is affiliated with the department of obstetrics and gynecology at University of Texas Southwestern Medical Center in Dallas.
Physicians may recommend contraception within 6 weeks of delivery, but many patients do not attend postpartum visits. “One option for women who have completed childbearing is bilateral midsegment salpingectomy via minilaparotomy,” Dr. Byrne said at the Pregnancy Meeting, sponsored by the Society for Maternal-Fetal Medicine. “Offering this procedure immediately after delivery makes it available to women who face obstacles to follow-up care.”
The procedure entails the risk of anesthetic complications, bowel injury, and vascular injury. Subsequent pregnancy or ectopic pregnancy also may occur. Some centers will not perform the procedure if a patient’s size affects the surgeon’s ability to feel the relevant anatomy, Dr. Byrne said. “Although operative complications are presumed to be higher among obese women,” prior studies have not examined whether BMI affects rates of procedure completion, complication, or subsequent pregnancy, the researchers said.
To study this question, Dr. Byrne and colleagues examined data from women who requested postpartum sterilization following vaginal delivery at their center in 2018. The center uses the Parkland tubal ligation technique. The researchers assessed complication rates using a composite measure that included surgical complications (that is, blood transfusion, aborted procedure, or extension of incision), anesthetic complications, readmission, superficial or deep wound infection, venous thromboembolism, ileus or small bowel obstruction, incomplete transection, and subsequent pregnancy. The investigators used statistical tests to assess the relationship between BMI and morbidity.
In all, 1,014 patients underwent a postpartum tubal ligation; 17% had undergone prior abdominal surgery. The researchers classified patients’ BMI as normal (7% of the population), overweight (28%), class I obesity (38%), class II obesity (18%), or class III obesity (9%). A composite morbidity event occurred in 2%, and the proportion of patients with a complication did not significantly differ across BMI categories. No morbid events occurred in patients with normal BMI, which indicates “minimal risk” in this population, Dr. Byrne said. One incomplete transection occurred in a patient with class I obesity, and one subsequent pregnancy occurred in a patient with class II obesity. Estimated blood loss ranged from 9 mL in patients with normal BMI to 13 mL in patients with class III obesity, and length of surgery ranged from 32 minutes to 40 minutes. Neither difference is clinically significant, Dr. Byrne said.
“For the woman who desires permanent contraception, BMI should not impede her access to the procedure,” he noted.
The researchers had no relevant disclosures.
SOURCE: Byrne JJ et al. Am J Obstet Gynecol. 2020 Jan;222(1):S290, Abstract 442.
GRAPEVINE, TEXAS – Higher body mass index is not associated with increased morbidity in women undergoing postpartum tubal ligation, according to a study of more than 1,000 patients.
John J. Byrne, MD, said at the Pregnancy Meeting. Dr. Byrne is affiliated with the department of obstetrics and gynecology at University of Texas Southwestern Medical Center in Dallas.
Physicians may recommend contraception within 6 weeks of delivery, but many patients do not attend postpartum visits. “One option for women who have completed childbearing is bilateral midsegment salpingectomy via minilaparotomy,” Dr. Byrne said at the Pregnancy Meeting, sponsored by the Society for Maternal-Fetal Medicine. “Offering this procedure immediately after delivery makes it available to women who face obstacles to follow-up care.”
The procedure entails the risk of anesthetic complications, bowel injury, and vascular injury. Subsequent pregnancy or ectopic pregnancy also may occur. Some centers will not perform the procedure if a patient’s size affects the surgeon’s ability to feel the relevant anatomy, Dr. Byrne said. “Although operative complications are presumed to be higher among obese women,” prior studies have not examined whether BMI affects rates of procedure completion, complication, or subsequent pregnancy, the researchers said.
To study this question, Dr. Byrne and colleagues examined data from women who requested postpartum sterilization following vaginal delivery at their center in 2018. The center uses the Parkland tubal ligation technique. The researchers assessed complication rates using a composite measure that included surgical complications (that is, blood transfusion, aborted procedure, or extension of incision), anesthetic complications, readmission, superficial or deep wound infection, venous thromboembolism, ileus or small bowel obstruction, incomplete transection, and subsequent pregnancy. The investigators used statistical tests to assess the relationship between BMI and morbidity.
In all, 1,014 patients underwent a postpartum tubal ligation; 17% had undergone prior abdominal surgery. The researchers classified patients’ BMI as normal (7% of the population), overweight (28%), class I obesity (38%), class II obesity (18%), or class III obesity (9%). A composite morbidity event occurred in 2%, and the proportion of patients with a complication did not significantly differ across BMI categories. No morbid events occurred in patients with normal BMI, which indicates “minimal risk” in this population, Dr. Byrne said. One incomplete transection occurred in a patient with class I obesity, and one subsequent pregnancy occurred in a patient with class II obesity. Estimated blood loss ranged from 9 mL in patients with normal BMI to 13 mL in patients with class III obesity, and length of surgery ranged from 32 minutes to 40 minutes. Neither difference is clinically significant, Dr. Byrne said.
“For the woman who desires permanent contraception, BMI should not impede her access to the procedure,” he noted.
The researchers had no relevant disclosures.
SOURCE: Byrne JJ et al. Am J Obstet Gynecol. 2020 Jan;222(1):S290, Abstract 442.
GRAPEVINE, TEXAS – Higher body mass index is not associated with increased morbidity in women undergoing postpartum tubal ligation, according to a study of more than 1,000 patients.
John J. Byrne, MD, said at the Pregnancy Meeting. Dr. Byrne is affiliated with the department of obstetrics and gynecology at University of Texas Southwestern Medical Center in Dallas.
Physicians may recommend contraception within 6 weeks of delivery, but many patients do not attend postpartum visits. “One option for women who have completed childbearing is bilateral midsegment salpingectomy via minilaparotomy,” Dr. Byrne said at the Pregnancy Meeting, sponsored by the Society for Maternal-Fetal Medicine. “Offering this procedure immediately after delivery makes it available to women who face obstacles to follow-up care.”
The procedure entails the risk of anesthetic complications, bowel injury, and vascular injury. Subsequent pregnancy or ectopic pregnancy also may occur. Some centers will not perform the procedure if a patient’s size affects the surgeon’s ability to feel the relevant anatomy, Dr. Byrne said. “Although operative complications are presumed to be higher among obese women,” prior studies have not examined whether BMI affects rates of procedure completion, complication, or subsequent pregnancy, the researchers said.
To study this question, Dr. Byrne and colleagues examined data from women who requested postpartum sterilization following vaginal delivery at their center in 2018. The center uses the Parkland tubal ligation technique. The researchers assessed complication rates using a composite measure that included surgical complications (that is, blood transfusion, aborted procedure, or extension of incision), anesthetic complications, readmission, superficial or deep wound infection, venous thromboembolism, ileus or small bowel obstruction, incomplete transection, and subsequent pregnancy. The investigators used statistical tests to assess the relationship between BMI and morbidity.
In all, 1,014 patients underwent a postpartum tubal ligation; 17% had undergone prior abdominal surgery. The researchers classified patients’ BMI as normal (7% of the population), overweight (28%), class I obesity (38%), class II obesity (18%), or class III obesity (9%). A composite morbidity event occurred in 2%, and the proportion of patients with a complication did not significantly differ across BMI categories. No morbid events occurred in patients with normal BMI, which indicates “minimal risk” in this population, Dr. Byrne said. One incomplete transection occurred in a patient with class I obesity, and one subsequent pregnancy occurred in a patient with class II obesity. Estimated blood loss ranged from 9 mL in patients with normal BMI to 13 mL in patients with class III obesity, and length of surgery ranged from 32 minutes to 40 minutes. Neither difference is clinically significant, Dr. Byrne said.
“For the woman who desires permanent contraception, BMI should not impede her access to the procedure,” he noted.
The researchers had no relevant disclosures.
SOURCE: Byrne JJ et al. Am J Obstet Gynecol. 2020 Jan;222(1):S290, Abstract 442.
REPORTING FROM THE PREGNANCY MEETING
Laparoscopic management of bladder fibroids: Surgical tips and tricks
ERAS protocol for cesarean delivery reduces opioid usage
GRAPEVINE, TEX. – An enhanced recovery after surgery (ERAS) pathway for cesarean delivery decreased postoperative opioid usage by 62% in one health care organization, researchers reported at the Pregnancy Meeting. The protocol incorporates a stepwise approach to pain control with no scheduled postoperative opioids.
Abington Jefferson Health, which includes two hospitals in Pennsylvania, implemented an ERAS pathway for all cesarean deliveries in October 2018. Kathryn Ruymann, MD, said at the meeting sponsored by the Society for Maternal-Fetal Medicine. Dr. Ruymann is an obstetrics and gynecology resident at Abington Jefferson Health.
Prior to the ERAS protocol, 99%-100% of patients took an opioid during the postoperative period. “With ERAS, 26% of patients never took an opioid during the postop period,” Dr. Ruymann and her associates reported. “Pain scores decreased with ERAS for postoperative days 1-3 and remained unchanged on day 4.”
One in 300 opioid-naive patients who receives opioids after cesarean delivery becomes a persistent user, one study has shown (Am J Obstet Gynecol. 2016 Sep; 215(3):353.e1-18). “ERAS pathways integrate evidence-based interventions before, during, and after surgery to optimize outcomes, specifically to decrease postoperative opioid use,” the researchers said.
While other surgical fields have adopted ERAS pathways, more research is needed in obstetrics, said Dr. Ruymann. More than 4,500 women deliver at Abington Jefferson Health each year, and about a third undergo cesarean deliveries.
The organization’s ERAS pathway incorporates preoperative education, fasting guidelines, and intraoperative analgesia, nausea prophylaxis, and antimicrobial therapy. Under the new protocol, postoperative analgesia includes scheduled administration of nonopioid medications, including celecoxib and acetaminophen. In addition, patients may take 5-10 mg of oxycodone orally every 4 hours as needed, and hydromorphone 0.4 mg IV as needed may be used for refractory pain. In addition, patients should resume eating as soon as tolerated and be out of bed within 4 hours after surgery, according to the protocol. Postoperative management of pruritus and instructions on how to wean off opioids at home are among the other elements of the enhanced recovery plan.
To examine postoperative opioid usage before and after implementation of the ERAS pathway, the investigators conducted a retrospective cohort study of 316 women who underwent cesarean delivery 3 months before the start of the ERAS pathway and 267 who underwent cesarean delivery 3 months after. The researchers used an application developed in Qlik Sense, a data analytics platform, to calculate opioid usage.
Mean postoperative opioid use decreased by 62%. The reduction in opioid use remained 8 months after starting the ERAS pathway.
“An ERAS pathway for [cesarean delivery] decreases postoperative opioid usage by integrating a multimodal stepwise approach to pain control and recovery,” the researchers said. “Standardized order sets and departmentwide education were crucial in the success of ERAS. Additional research is needed to evaluate the impact of unique components of ERAS in order to optimize this pathway.”
The researchers had no disclosures.
SOURCE: Ruymann K et al. Am J Obstet Gynecol. 2020 Jan;222(1):S212, Abstract 315.
GRAPEVINE, TEX. – An enhanced recovery after surgery (ERAS) pathway for cesarean delivery decreased postoperative opioid usage by 62% in one health care organization, researchers reported at the Pregnancy Meeting. The protocol incorporates a stepwise approach to pain control with no scheduled postoperative opioids.
Abington Jefferson Health, which includes two hospitals in Pennsylvania, implemented an ERAS pathway for all cesarean deliveries in October 2018. Kathryn Ruymann, MD, said at the meeting sponsored by the Society for Maternal-Fetal Medicine. Dr. Ruymann is an obstetrics and gynecology resident at Abington Jefferson Health.
Prior to the ERAS protocol, 99%-100% of patients took an opioid during the postoperative period. “With ERAS, 26% of patients never took an opioid during the postop period,” Dr. Ruymann and her associates reported. “Pain scores decreased with ERAS for postoperative days 1-3 and remained unchanged on day 4.”
One in 300 opioid-naive patients who receives opioids after cesarean delivery becomes a persistent user, one study has shown (Am J Obstet Gynecol. 2016 Sep; 215(3):353.e1-18). “ERAS pathways integrate evidence-based interventions before, during, and after surgery to optimize outcomes, specifically to decrease postoperative opioid use,” the researchers said.
While other surgical fields have adopted ERAS pathways, more research is needed in obstetrics, said Dr. Ruymann. More than 4,500 women deliver at Abington Jefferson Health each year, and about a third undergo cesarean deliveries.
The organization’s ERAS pathway incorporates preoperative education, fasting guidelines, and intraoperative analgesia, nausea prophylaxis, and antimicrobial therapy. Under the new protocol, postoperative analgesia includes scheduled administration of nonopioid medications, including celecoxib and acetaminophen. In addition, patients may take 5-10 mg of oxycodone orally every 4 hours as needed, and hydromorphone 0.4 mg IV as needed may be used for refractory pain. In addition, patients should resume eating as soon as tolerated and be out of bed within 4 hours after surgery, according to the protocol. Postoperative management of pruritus and instructions on how to wean off opioids at home are among the other elements of the enhanced recovery plan.
To examine postoperative opioid usage before and after implementation of the ERAS pathway, the investigators conducted a retrospective cohort study of 316 women who underwent cesarean delivery 3 months before the start of the ERAS pathway and 267 who underwent cesarean delivery 3 months after. The researchers used an application developed in Qlik Sense, a data analytics platform, to calculate opioid usage.
Mean postoperative opioid use decreased by 62%. The reduction in opioid use remained 8 months after starting the ERAS pathway.
“An ERAS pathway for [cesarean delivery] decreases postoperative opioid usage by integrating a multimodal stepwise approach to pain control and recovery,” the researchers said. “Standardized order sets and departmentwide education were crucial in the success of ERAS. Additional research is needed to evaluate the impact of unique components of ERAS in order to optimize this pathway.”
The researchers had no disclosures.
SOURCE: Ruymann K et al. Am J Obstet Gynecol. 2020 Jan;222(1):S212, Abstract 315.
GRAPEVINE, TEX. – An enhanced recovery after surgery (ERAS) pathway for cesarean delivery decreased postoperative opioid usage by 62% in one health care organization, researchers reported at the Pregnancy Meeting. The protocol incorporates a stepwise approach to pain control with no scheduled postoperative opioids.
Abington Jefferson Health, which includes two hospitals in Pennsylvania, implemented an ERAS pathway for all cesarean deliveries in October 2018. Kathryn Ruymann, MD, said at the meeting sponsored by the Society for Maternal-Fetal Medicine. Dr. Ruymann is an obstetrics and gynecology resident at Abington Jefferson Health.
Prior to the ERAS protocol, 99%-100% of patients took an opioid during the postoperative period. “With ERAS, 26% of patients never took an opioid during the postop period,” Dr. Ruymann and her associates reported. “Pain scores decreased with ERAS for postoperative days 1-3 and remained unchanged on day 4.”
One in 300 opioid-naive patients who receives opioids after cesarean delivery becomes a persistent user, one study has shown (Am J Obstet Gynecol. 2016 Sep; 215(3):353.e1-18). “ERAS pathways integrate evidence-based interventions before, during, and after surgery to optimize outcomes, specifically to decrease postoperative opioid use,” the researchers said.
While other surgical fields have adopted ERAS pathways, more research is needed in obstetrics, said Dr. Ruymann. More than 4,500 women deliver at Abington Jefferson Health each year, and about a third undergo cesarean deliveries.
The organization’s ERAS pathway incorporates preoperative education, fasting guidelines, and intraoperative analgesia, nausea prophylaxis, and antimicrobial therapy. Under the new protocol, postoperative analgesia includes scheduled administration of nonopioid medications, including celecoxib and acetaminophen. In addition, patients may take 5-10 mg of oxycodone orally every 4 hours as needed, and hydromorphone 0.4 mg IV as needed may be used for refractory pain. In addition, patients should resume eating as soon as tolerated and be out of bed within 4 hours after surgery, according to the protocol. Postoperative management of pruritus and instructions on how to wean off opioids at home are among the other elements of the enhanced recovery plan.
To examine postoperative opioid usage before and after implementation of the ERAS pathway, the investigators conducted a retrospective cohort study of 316 women who underwent cesarean delivery 3 months before the start of the ERAS pathway and 267 who underwent cesarean delivery 3 months after. The researchers used an application developed in Qlik Sense, a data analytics platform, to calculate opioid usage.
Mean postoperative opioid use decreased by 62%. The reduction in opioid use remained 8 months after starting the ERAS pathway.
“An ERAS pathway for [cesarean delivery] decreases postoperative opioid usage by integrating a multimodal stepwise approach to pain control and recovery,” the researchers said. “Standardized order sets and departmentwide education were crucial in the success of ERAS. Additional research is needed to evaluate the impact of unique components of ERAS in order to optimize this pathway.”
The researchers had no disclosures.
SOURCE: Ruymann K et al. Am J Obstet Gynecol. 2020 Jan;222(1):S212, Abstract 315.
REPORTING FROM THE PREGNANCY MEETING
Salpingectomy adds little time and no complications to cesarean delivery
GRAPEVINE, TEXAS – Performing a total salpingectomy at the time of cesarean delivery added just over 6 minutes of operative time, compared with cesarean delivery and conventional sterilization, according to a recent systematic review and meta-analysis.
Although surgery took a little longer with salpingectomy, there was no increase in surgical complications, Jared Roeckner, MD, said in an interview at the Pregnancy Meeting, sponsored by the Society for Maternal-Fetal Medicine. “Total salpingectomy could provide an effective means of contraception and reduce the risk of future ovarian cancer,” he said.
Dr. Roeckner, a maternal-fetal medicine fellow at the University of South Florida, Tampa, explained in an interview that the systematic review and meta-analysis comprised 11 studies and included 320,443 women who received salpingectomy or standard sterilization methods. Eight cohort studies and three randomized controlled trials were included in the analysis, which was presented in a poster session at the meeting and in a simultaneous publication in Obstetrics & Gynecology.
The review’s results, wrote Dr. Roeckner and colleagues, “suggest total salpingectomy should be offered to women interested in ovarian cancer risk-reduction interventions [who] plan to undergo sterilization at the time of cesarean delivery.”
The eight cohort studies included 7,303 women. In these studies, women who received total salpingectomy at the time of cesarean delivery had operative time – defined as the time from skin incision to skin closure – 6.3 minutes longer than women who received a standard sterilization method (95% confidence interval, 3.5-9.1). The difference in duration of procedure for the three randomized controlled trials was not statistically significant between the two procedures.
Dr. Roeckner and colleagues noted that two of the randomized controlled trials reported times for the sterilization procedures. One study found a duration of 5.6 minutes for salpingectomy with a bipolar device and 6.1 minutes for tubal interruption; the other study compared salpingectomy with suture ligation and tubal interruption, finding operative times of 18.5 and 6.9 minutes, respectively.
In addition to the primary outcome of operative time, Dr. Roeckner and colleagues looked at rates of a variety of complications. These included transfusion, estimated blood loss, change in hemoglobin, wound infection, internal organ damage, readmission, reoperation, and length of stay. Salpingectomy was not associated with higher rates of any of these complications.
“Our main finding was that salpingectomy at the time of cesarean delivery may be associated with a small increase in operative time, but it doesn’t appear to be associated with an increased rate of surgical complications,” the researchers wrote.
One concern that’s been raised about the strategy of salpingectomy is the possibility of reduction of ovarian reserve related to decreased blood supply to the ovaries. However, noted Dr. Roeckner and coinvestigators, other studies have not shown decreases in anti-Müllerian hormone levels or other real-world signals for reduced ovarian reserve.
It’s true, the investigators acknowledged, that there is no possibility for reanastomosis and future fertility with salpingectomy. However, they observed that if the possibility for future fertility exists, conventional tubal ligation should not be performed.
Despite the thoroughness of the review and the investigators’ emphasis on adhering to best practices for systematic reviews and meta-analysis, they acknowledged that there were few studies, which resulted in some difficulties with statistical power. Still, they said, “there does not appear to be a trend toward increased complications among the salpingectomy cohort.”
Dr. Roeckner reported no outside sources of funding and no conflicts of interest.
SOURCE: Roeckner J et al. Pregnancy Meeting, Abstract P180; Obstet Gynecol. 2020 Feb;135:3:550-7.
GRAPEVINE, TEXAS – Performing a total salpingectomy at the time of cesarean delivery added just over 6 minutes of operative time, compared with cesarean delivery and conventional sterilization, according to a recent systematic review and meta-analysis.
Although surgery took a little longer with salpingectomy, there was no increase in surgical complications, Jared Roeckner, MD, said in an interview at the Pregnancy Meeting, sponsored by the Society for Maternal-Fetal Medicine. “Total salpingectomy could provide an effective means of contraception and reduce the risk of future ovarian cancer,” he said.
Dr. Roeckner, a maternal-fetal medicine fellow at the University of South Florida, Tampa, explained in an interview that the systematic review and meta-analysis comprised 11 studies and included 320,443 women who received salpingectomy or standard sterilization methods. Eight cohort studies and three randomized controlled trials were included in the analysis, which was presented in a poster session at the meeting and in a simultaneous publication in Obstetrics & Gynecology.
The review’s results, wrote Dr. Roeckner and colleagues, “suggest total salpingectomy should be offered to women interested in ovarian cancer risk-reduction interventions [who] plan to undergo sterilization at the time of cesarean delivery.”
The eight cohort studies included 7,303 women. In these studies, women who received total salpingectomy at the time of cesarean delivery had operative time – defined as the time from skin incision to skin closure – 6.3 minutes longer than women who received a standard sterilization method (95% confidence interval, 3.5-9.1). The difference in duration of procedure for the three randomized controlled trials was not statistically significant between the two procedures.
Dr. Roeckner and colleagues noted that two of the randomized controlled trials reported times for the sterilization procedures. One study found a duration of 5.6 minutes for salpingectomy with a bipolar device and 6.1 minutes for tubal interruption; the other study compared salpingectomy with suture ligation and tubal interruption, finding operative times of 18.5 and 6.9 minutes, respectively.
In addition to the primary outcome of operative time, Dr. Roeckner and colleagues looked at rates of a variety of complications. These included transfusion, estimated blood loss, change in hemoglobin, wound infection, internal organ damage, readmission, reoperation, and length of stay. Salpingectomy was not associated with higher rates of any of these complications.
“Our main finding was that salpingectomy at the time of cesarean delivery may be associated with a small increase in operative time, but it doesn’t appear to be associated with an increased rate of surgical complications,” the researchers wrote.
One concern that’s been raised about the strategy of salpingectomy is the possibility of reduction of ovarian reserve related to decreased blood supply to the ovaries. However, noted Dr. Roeckner and coinvestigators, other studies have not shown decreases in anti-Müllerian hormone levels or other real-world signals for reduced ovarian reserve.
It’s true, the investigators acknowledged, that there is no possibility for reanastomosis and future fertility with salpingectomy. However, they observed that if the possibility for future fertility exists, conventional tubal ligation should not be performed.
Despite the thoroughness of the review and the investigators’ emphasis on adhering to best practices for systematic reviews and meta-analysis, they acknowledged that there were few studies, which resulted in some difficulties with statistical power. Still, they said, “there does not appear to be a trend toward increased complications among the salpingectomy cohort.”
Dr. Roeckner reported no outside sources of funding and no conflicts of interest.
SOURCE: Roeckner J et al. Pregnancy Meeting, Abstract P180; Obstet Gynecol. 2020 Feb;135:3:550-7.
GRAPEVINE, TEXAS – Performing a total salpingectomy at the time of cesarean delivery added just over 6 minutes of operative time, compared with cesarean delivery and conventional sterilization, according to a recent systematic review and meta-analysis.
Although surgery took a little longer with salpingectomy, there was no increase in surgical complications, Jared Roeckner, MD, said in an interview at the Pregnancy Meeting, sponsored by the Society for Maternal-Fetal Medicine. “Total salpingectomy could provide an effective means of contraception and reduce the risk of future ovarian cancer,” he said.
Dr. Roeckner, a maternal-fetal medicine fellow at the University of South Florida, Tampa, explained in an interview that the systematic review and meta-analysis comprised 11 studies and included 320,443 women who received salpingectomy or standard sterilization methods. Eight cohort studies and three randomized controlled trials were included in the analysis, which was presented in a poster session at the meeting and in a simultaneous publication in Obstetrics & Gynecology.
The review’s results, wrote Dr. Roeckner and colleagues, “suggest total salpingectomy should be offered to women interested in ovarian cancer risk-reduction interventions [who] plan to undergo sterilization at the time of cesarean delivery.”
The eight cohort studies included 7,303 women. In these studies, women who received total salpingectomy at the time of cesarean delivery had operative time – defined as the time from skin incision to skin closure – 6.3 minutes longer than women who received a standard sterilization method (95% confidence interval, 3.5-9.1). The difference in duration of procedure for the three randomized controlled trials was not statistically significant between the two procedures.
Dr. Roeckner and colleagues noted that two of the randomized controlled trials reported times for the sterilization procedures. One study found a duration of 5.6 minutes for salpingectomy with a bipolar device and 6.1 minutes for tubal interruption; the other study compared salpingectomy with suture ligation and tubal interruption, finding operative times of 18.5 and 6.9 minutes, respectively.
In addition to the primary outcome of operative time, Dr. Roeckner and colleagues looked at rates of a variety of complications. These included transfusion, estimated blood loss, change in hemoglobin, wound infection, internal organ damage, readmission, reoperation, and length of stay. Salpingectomy was not associated with higher rates of any of these complications.
“Our main finding was that salpingectomy at the time of cesarean delivery may be associated with a small increase in operative time, but it doesn’t appear to be associated with an increased rate of surgical complications,” the researchers wrote.
One concern that’s been raised about the strategy of salpingectomy is the possibility of reduction of ovarian reserve related to decreased blood supply to the ovaries. However, noted Dr. Roeckner and coinvestigators, other studies have not shown decreases in anti-Müllerian hormone levels or other real-world signals for reduced ovarian reserve.
It’s true, the investigators acknowledged, that there is no possibility for reanastomosis and future fertility with salpingectomy. However, they observed that if the possibility for future fertility exists, conventional tubal ligation should not be performed.
Despite the thoroughness of the review and the investigators’ emphasis on adhering to best practices for systematic reviews and meta-analysis, they acknowledged that there were few studies, which resulted in some difficulties with statistical power. Still, they said, “there does not appear to be a trend toward increased complications among the salpingectomy cohort.”
Dr. Roeckner reported no outside sources of funding and no conflicts of interest.
SOURCE: Roeckner J et al. Pregnancy Meeting, Abstract P180; Obstet Gynecol. 2020 Feb;135:3:550-7.
REPORTING FROM THE PREGNANCY MEETING
Conservative care or surgery for rotator cuff tears?
Rotator cuff disease accounts for as many as 65% of shoulder-related visits to physicians’ offices,1 yet the natural course of rotator cuff tears is still not well understood.2 Treatment options are controversial because both conservative and surgical management have been successful. Physical therapy is a durable and reliable treatment option, but there are concerns about long-term progression of the tear.3 Surgical arthroscopic techniques, which result in less morbidity than open surgery, have improved overall surgical care; as such, the rate of rotator cuff procedures has increased significantly.4
Our goal in this article is to provide clinical guidance to the primary care provider. We review management options for rotator cuff injury; summarize considerations for proceeding with conservative or surgical management; and discuss surgical risks and complications.
Conservative management: Who is most likely to benefit?
The choice of treatment for rotator cuff injury depends on a host of variables, including shoulder dominance, duration of symptoms, type of tear (partial or full), age, demands (activity level, occupation, sport), and comorbidities (diabetes, tobacco use). Treatment goals include resolution of pain, normalized range of motion and strength, and restored arm and shoulder function.5
Initial nonoperative management is indicated in patients who
- have a partial-thickness tear (a notable exception is young patients with traumatic injury),6
- have lower functional demands and moderate symptoms, or
- refuse surgery.7
Patients who respond to nonoperative management will, typically, do so within 6 to 12 weeks.5,8
Few randomized, controlled trials have compared conservative and surgical management of rotator cuff tears; furthermore, the findings of these studies have been mixed. Nonoperative management has been shown to be the favored initial treatment for isolated, symptomatic, nontraumatic, supraspinatus tears in older patients.9 In a recent study,10 5-year outcomes were examined in a prospective cohort enrolled in a rotator cuff treatment program: Approximately 75% of patients remained successfully treated with nonoperative management, and clinical outcomes of the operative and nonoperative groups were not significantly different at 5-year follow-up. Investigators concluded that nonoperative treatment is effective for many patients who have a chronic, full-thickness rotator cuff tear.
In a study investigating the treatment of degenerative rotator cuff tear, patients were randomly treated using an operative or nonoperative protocol. No differences in functional outcomes were observed at 1 year after treatment; however, surgical treatment significantly improved subjective parameters of pain and disability.11 A similar study suggested statistically significant improvement in outcomes for patients managed operatively, compared with those treated nonoperatively, but differences in shoulder outcome and the visual analog pain score were small and failed to meet thresholds considered clinically significant. Larger studies, with longer follow-up, are required to determine whether clinical differences between these types of treatment become more evident over time.12
Continue to: A look at nonoperative options and outcomes
A look at nonoperative options and outcomes
Surveillance. Rotator cuff disease of the supraspinatus tendon often results from a degenerative process that progresses to partial and, eventually, full-thickness tearing.8 Once a tear develops, progression is difficult to predict. Many rotator cuff tears grow larger over time; this progression is commonly associated with new or increased pain and weakness, or both. Although asymptomatic progression of a tear is uncommon, many patients—and physicians—are apprehensive about proceeding with nonoperative treatment for a full-thickness tear.8
To diminish such fears, surveillance can include regular assessment of shoulder motion and strength, with consideration of repeat imaging until surgery is performed or the patient is no longer a surgical candidate or interested in surgical treatment.7 Patients and providers need to remain vigilant because tears that are initially graded as repairable can become irreparable if the tendon retracts or there is fatty infiltration of the muscle belly. Results of secondary surgical repair following failed prolonged nonoperative treatment tend to be inferior to results seen in patients who undergo primary tendon repair.7
Analgesics. Simple analgesics, such as acetaminophen, are a low-risk first-line option for pain relief; however, there are limited data on the efficacy of acetaminophen in rotator cuff disease. A topical or oral nonsteroidal anti-inflammatory drug (NSAID), or both, can be considered, but potential contraindications, such as gastrointestinal, renal, and cardiovascular risks, should be monitored.13 Avoid opioids, given the potential for abuse, except during the immediate postoperative period.5
Glucocorticoid injection. Injection of a glucocorticoid drug into the subacromial space should be considered in patients whose pain interferes with sleep, limits activities of daily living, or hinders the ability to participate in physical therapy.5 A recent systematic review demonstrated that NSAIDs and glucocorticoids brought similar pain relief and active abduction at 4 to 6 weeks, but that glucocorticoids were significantly better at achieving remission of symptoms.14 There are no data comparing glucocorticoid preparations (ie, different glucocorticoids or anesthetics, dosages, volumes), and ultrasound guidance does not appear to be necessary for short-term pain relief.15 Note: Repeated injection has been shown to decrease the durability of surgically repaired tendons16; if a patient is a candidate for surgery, repeat injections should be carefully considered—and avoided if possible.
Physical therapy. The goals of physical therapy are activity modification, stretching the shoulder capsule, and strengthening the surrounding musculature (periscapular, rotator cuff, and deltoid). Patients advance through 3 phases of recovery: shoulder mobility, strengthening, and function (ie, joint reactivation to improve shoulder proprioception and coordination).
Continue to: A recent meta-analysis...
A recent meta-analysis17 found comparative evidence on treating rotator cuff tears with physical therapy to be inconclusive. At 1-year follow-up, there was no clinically significant difference between surgery and active physical therapy in either improving the Constant Shoulder Score (an assessment of function) or reducing pain caused by a rotator cuff tear. Therefore, the authors proposed, given the low risk of harm, a conservative approach should be the initial treatment modality for a tear.
A Cochrane review18 examined 60 eligible trials, in which the mean age of patients was 51 years and the mean duration of symptoms, 11 months. Overall, the review concluded that the effects of manual therapy and exercise might be similar to those of glucocorticoid injection and arthroscopic subacromial decompression. The authors noted that this conclusion is based on low-quality evidence, with only 1 study in the review that compared the combination of manual therapy and exercise to placebo.
Other conservative options. Ultrasound, topical nitroglycerin, topical lidocaine, glucocorticoid iontophoresis, transcutaneous electrical nerve stimulation, massage, acupuncture, extracorporeal shockwave therapy, hyaluronic acid, and platelet-rich plasma have been used to treat rotator cuff disease. These modalities require further study, however, to determine their effectiveness for this indication.7,19
Who is a candidate for surgical management?
Although nonoperative treatment is preferred for rotator cuff tendinitis or tendinosis and partial-thickness tears, appropriate management of full-thickness tears is debatable.20 Some surgeons advocate early operative intervention of repairable full-thickness tears to prevent further progression and reduce the risk of long-term dysfunction.
The decision to pursue operative repair depends on
- patient characteristics (age, activity level, comorbidities),
- patient function (amount of disability caused by the tear),
- characteristics of the tear (length, depth, retraction), and
- chronicity of the tear (acuity).
Continue to: TABLE 1...
TABLE 121,22 highlights variables that influence the decision to proceed, or not to proceed, with operative intervention. Because enlargement of a tear usually exacerbates symptoms,23 patients with a tear who are successfully managed nonoperatively should be counseled on the potential of the tear to progress.

What are the surgical options?
Little clinical evidence favors one exposure technique over another. This equivalency has been demonstrated by a systematic review of randomized controlled trials comparing arthroscopic and mini-open rotator cuff repair, which showed no difference in function, pain, or range of motion.24 That conclusion notwithstanding, arthroscopic repair is increasingly popular because it results in less pain, initially, and faster return to work.20
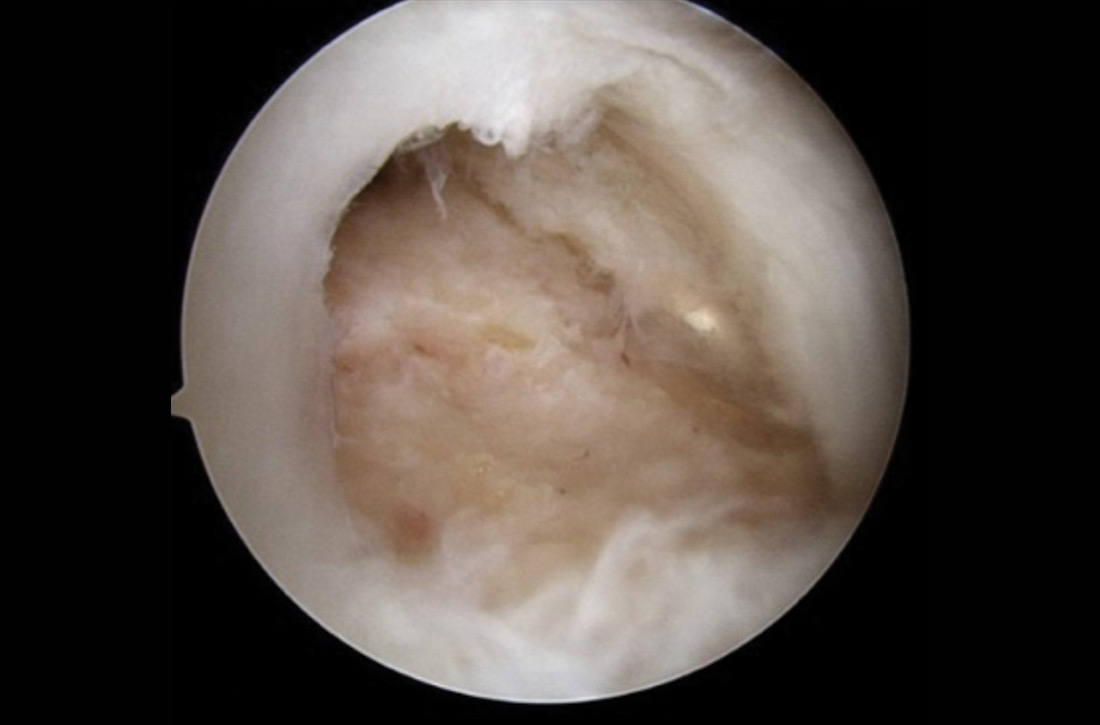
There is controversy among surgeons regarding the choice of fixation technique: Tendons can be secured using 1 or 2 rows of anchors (FIGURE). Advocates of single-row repair cite shorter surgical time, decreased cost, and equivalent outcomes; surgeons who favor double-row, or so-called transosseous-equivalent, repair claim that it provides better restoration of normal anatomy and biomechanical superiority.25,26
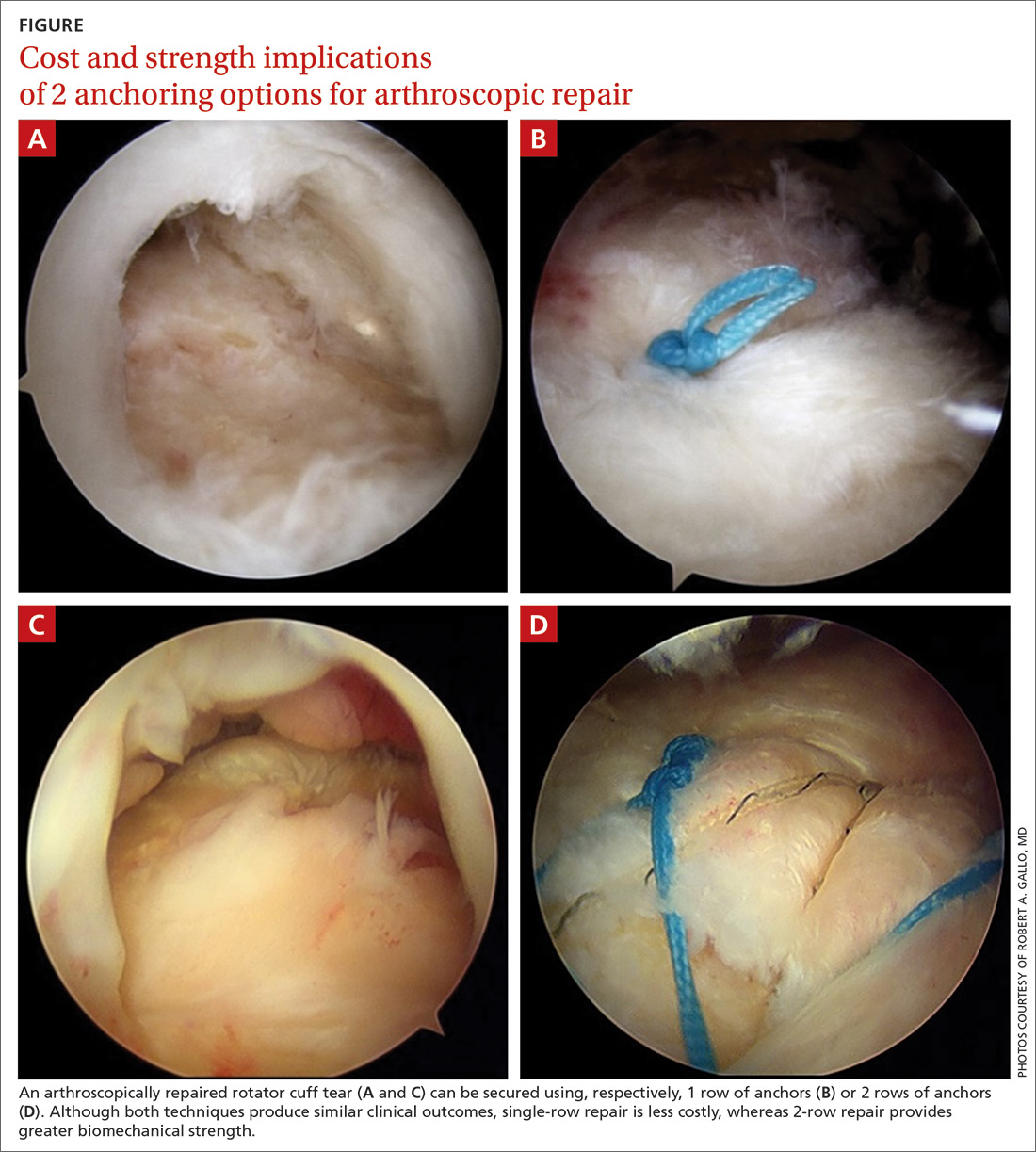
Regardless of technique, most patients are immobilized for 4 to 6 weeks postoperatively.27 Physical therapy usually commences within the first week or 2 postop, limited to passive motion for 6 to 12 weeks. Active motion and strengthening of rotator-cuff muscles often is initiated by 3 months postop, although this phase is sometimes delayed because of concern over slow tendon healing. Typically, patients make a full return to sports and manual work at 6 months postop. Patients experience most symptomatic improvement during the first 6 months following surgery, although functional gains can be realized for as long as 2 years after surgery.28
Most torn rotator cuffs can be fixed back to the greater tuberosity, but some chronic, massive, retracted tears lack the mobility to be repaired, or re-tear shortly after repair. Over time, the humeral head in a rotator cuff–deficient shoulder can migrate superiorly to abut the undersurface of the acromion, which can lead to significant glenohumeral osteoarthritis. To prevent or remedy elevation of the humeral head, salvage procedures—debridement, partial repair, spanning graft, tendon transfer, superior capsule reconstruction, balloon arthroplasty, reverse total shoulder replacement—can be used to alleviate pain and restore function. These procedures have significant limitations, however, and usually provide less favorable outcomes than standard repair.29-35
Continue to: Surgical outcomes
Surgical outcomes
Pain, function, and patient satisfaction outcomes following rotator cuff repair are generally favorable: 90% of patients are “happy” 6 months postop.28 Younger populations often have traumatic rotator cuff tears; they generally are interested in returning to sporting activities following their injury. Nearly 85% of younger patients who undergo rotator cuff repair return to sports, and 65.9% return to an equivalent level of play.36
Variables associated with an unfavorable outcome include increasing age, smoking, increased size of the tear, poor tendon quality, hyperlipidemia, workers’ compensation status, fatty infiltration of muscle, obesity, diabetes, and additional procedures to the biceps tendon and acromioclavicular joint performed at the time of rotator cuff repair.37-39 Interestingly, a study concluded that, if a patient expects a good surgical outcome, they are more likely to go on to report a favorable outcome—suggesting that a patient’s expectations might influence their actual outcome.40
Risks and complications
Although rotator cuff surgery has much lower morbidity than other orthopedic surgeries, it is not without risk of complications. If re-tears are excluded, postop complications have been reported in approximately 10% of patients.41 Common complications and their anticipated rate of occurrence are listed in TABLE 2.42-49
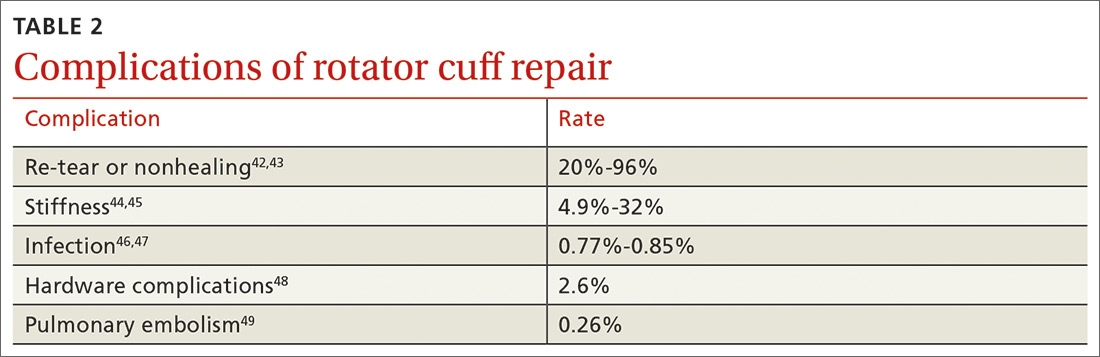
Re-tear of the surgically repaired tendon is the most common postop complication. Published re-tear rates range from 20% to 96%42,43 and generally correlate with initial tear size: A small tear is twice as likely to heal as a massive tear.50 That large range—a span of 76%—results from using a variety of methods to measure re-tear and might not have clinical meaning. A meta-analysis that examined more than 8000 shoulder surgeries reported an overall re-tear rate of 26.6%; however, both patients whose tendons healed and those who re-tore demonstrated clinical improvement.51 In a separate study, patients reported improvement in pain, function, range of motion, and satisfaction regardless of the integrity of the tendon; however, significant improvement in strength was seen only in those whose repair had healed.52
Postop stiffness is more common with arthroscopic repair than with open surgery, and with smaller rather than larger tears.53 Patient variables associated with an increased risk of postop adhesive capsulitis include workers’ compensation insurance, age < 50 years, and preoperative calcific tendonitis or adhesive capsulitis.53 Stiffness generally responds to physical therapy and rarely requires surgical lysis of adhesions or capsular release.
Continue to: Significant injury...
Significant injury to the deltoid muscle has become increasingly uncommon with the advancement of arthroscopic surgery. In traditional open surgery, detachment of the deltoid (and subsequent repair) is required to improve visualization; however, doing so can lead to atrophy and muscle rupture and dehiscence. Deltoid damage occurs in ≤ 60% of open surgeries but is negligible in arthroscopic and mini-open repairs, which involve splitting deltoid fibers to gain exposure of the underlying rotator cuff.54
SIDEBAR
Key takeaways in the management of rotator cuff injury
- Chronic, nontraumatic, and partial-thickness tears respond well to conservative management as first-line treatment. Poor surgical candidates should also be offered a trial of conservative therapy.
- Consider referral for surgical consultation if the patient does not respond to conservative therapy in 6 to 12 weeks; also, patients who have a full-thickness tear and young patients with traumatic injury should be referred for surgical consultation.
- Arthroscopy has become the preferred approach to rotator cuff repair because it is associated with less pain, fewer complications, and faster recovery.
- Patients should be counseled that recovery from surgical repair of a torn rotator cuff takes, on average, 6 months. Some massive or retracted rotator cuff injuries require more extensive procedures that increase healing time.
- Overall, patients are “happy” with rotator cuff repair at 6 months; clinical complications are uncommon, making surgery a suitable option in appropriately selected patients.
CORRESPONDENCE
Cayce Onks, DO, MS, ATC, Penn State Health Milton S. Hershey Medical Center, Penn State College of Medicine, Family and Community Medicine H154, 500 University Drive, PO Box 850, Hershey, PA 17033-0850; [email protected].
1. Vecchio P, Kavanagh R, Hazleman BL, et al. Shoulder pain in a community-based rheumatology clinic. Br J Rheumatol. 1995;34:440-442.
2. Eljabu W, Klinger HM, von Knoch M. The natural history of rotator cuff tears: a systematic review. Arch Orthop Trauma Surg. 2015;135:1055-1061.
3. Dunn WR, Kuhn JE, Sanders R, et al; MOON Shoulder Group. 2013 Neer Award: predictors of failure of nonoperative treatment of chronic, symptomatic, full-thickness rotator cuff tears. J Shoulder Elbow Surg. 2016;25:1303-1311.
4. Colvin AC, Egorova N, Harrison AK, et al. National trends in rotator cuff repair. J Bone Joint Surg Am. 2012;94:227-233.
5. Whittle S, Buchbinder R. In the clinic. Rotator cuff disease. Ann Intern Med. 2015;162:ITC1-ITC15.
6. Lazarides AL, Alentorn-Geli E, Choi JHJ, et al. Rotator cuff tears in young patients: a different disease than rotator cuff tears in elderly patients. J Shoulder Elbow Surg. 2015;24:1834-1843.
7. Petri M, Ettinger M, Brand S, et al. Non-operative management of rotator cuff tears. Open Orthop J. 2016;10:349-356.
8. Schmidt CC, Jarrett CD, Brown BT. Management of rotator cuff tears. J Hand Surg Am. 2015;40:399-408.
9. Kukkonen J, Joukainen A, Lehtinen J, et al. Treatment of nontraumatic rotator cuff tears: a randomized controlled trial with two years of clinical and imaging follow-up. J Bone Joint Surg Am. 2015;97:1729-1737.
10. Boorman RS, More KD, Hollinshead RM, et al. What happens to patients when we do not repair their cuff tears? Five-year rotator cuff quality-of-life index outcomes following nonoperative treatment of patients with full-thickness rotator cuff tears. J Shoulder Elbow Surg. 2018;27:444-448.
11. Lambers Heerspink FO, van Raay JJ, Koorevaar RCT, et al. Comparing surgical repair with conservative treatment for degenerative rotator cuff tears: a randomized controlled trial. J Shoulder Elbow Surg. 2015;24:1274-1281.
12. Piper CC, Hughes AJ, Ma Y, et al. Operative versus nonoperative treatment for the management of full-thickness rotator cuff tears: a systematic review and meta-analysis. J Shoulder Elbow Surg. 2018;27:572-576.
13. Boudreault J, Desmeules F, Roy J-S, et al. The efficacy of oral non-steroidal anti-inflammatory drugs for rotator cuff tendinopathy: a systematic review and meta-analysis. J Rehabil Med. 2014;46:294-306.
14. Zheng X-Q, Li K, Wei Y-D, et al. Nonsteroidal anti-inflammatory drugs versus corticosteroid for treatment of shoulder pain: a systematic review and meta-analysis. Arch Phys Med Rehabil. 2014;95:1824-1831.
15. Bloom JE, Rischin A, Johnston RV, et al. Image-guided versus blind glucocorticoid injection for shoulder pain. Cochrane Database Syst Rev. 2012;(8):CD009147.
16. Wiggins ME, Fadale PD, Ehrlich MG, et al. Effects of local injection of corticosteroids on the healing of ligaments. A follow-up report. J Bone Joint Surg Am. 1995;77:1682-1691.
17. Ryösä A, Laimi K, Äärimaa V, et al. Surgery or conservative treatment for rotator cuff tear: a meta-analysis. Disabil Rehabil. 2017;39:1357-1363.
18. Page MJ, Green S, McBain B, et al. Manual therapy and exercise for rotator cuff disease. Cochrane Database Syst Rev. 2016;(6):CD012224.
19. Page MJ, Green S, Mrocki MA, et al. Electrotherapy modalities for rotator cuff disease. Cochrane Database Syst Rev. 2016;(6):CD012225.
20. Acevedo DC, Paxton ES, Williams GR, et al. A survey of expert opinion regarding rotator cuff repair. J Bone Joint Surg Am. 2014;96:e123.
21. Pedowitz RA, Yamaguchi K, Ahmad CS, et al. American Academy of Orthopaedic Surgeons Clinical Practice Guideline on: optimizing the management of rotator cuff problems. J Bone Joint Surg Am. 2012;94:163-167.
22. Thorpe A, Hurworth M, O’Sullivan P, et al. Rotator cuff disease: opinion regarding surgical criteria and likely outcome. ANZ J Surg. 2017;87:291-295.
23. Mall NA, Kim HM, Keener JD, et al. Symptomatic progression of asymptomatic rotator cuff tears: a prospective study of clinical and sonographic variables. J Bone Joint Surg Am. 2010;92:2623-2633.
24. Ji X, Bi C, Wang F, et al. Arthroscopic versus mini-open rotator cuff repair: an up-to-date meta-analysis of randomized controlled trials. Arthroscopy. 2015;31:118-124.
25. Duquin TR, Buyea C, Bisson LJ. Which method of rotator cuff repair leads to the highest rate of structural healing? A systematic review. Am J Sports Med. 2010;38:835-841.
26. Choi S, Kim MK, Kim GM, et al. Factors associated with clinical and structural outcomes after arthroscopic rotator cuff repair with a suture bridge technique in medium, large, and massive tears. J Shoulder Elbow Surg. 2014;23:1675-1681.
27. Shen C, Tang Z-H, Hu J-Z, et al. Does immobilization after arthroscopic rotator cuff repair increase tendon healing? A systematic review and meta-analysis. Arch Orthop Trauma Surg. 2014;134:1279-1285.
28. Gulotta LV, Nho SJ, Dodson CC, et al; . Prospective evaluation of arthroscopic rotator cuff repairs at 5 years: part I. Functional outcomes and radiographic healing rates. J Shoulder Elbow Surg. 2011;20:934-940.
29. Liem D, Lengers N, Dedy N, et al. Arthroscopic debridement of massive irreparable rotator cuff tears. Arthroscopy. 2008;24:743-748.
30. Weber SC. Partial rotator cuff repair in massive rotator cuff tears: long-term follow-up. J Shoulder Elbow Surg. 2017;26:e171.
31. Lewington MR, Ferguson DP, Smith TD, et al. Graft utilization in the bridging reconstruction of irreparable rotator cuff tears: a systematic review. Am J Sports Med. 2017;45:3149-3157.
32. Longo UG, Franceschetti E, Petrillo S, et al. Latissimus dorsi tendon transfer for massive irreparable rotator cuff tears: a systematic review. Sports Med Arthrosc Rev. 2011;19:428-437.
33. Noyes MP, Denard PJ. Arthroscopic superior capsular reconstruction: indications and outcomes. Oper Tech Sports Med. 2018;26:29-34.
34. Piekaar RSM, Bouman ICE, van Kampen PM, et al. Early promising outcome following arthroscopic implantation of the subacromial balloon spacer for treating massive rotator cuff tear. Musculoskeletal Surg. 2018;102:247-255.
35. Ek ETH, Neukom L, Catanzaro S, et al. Reverse total shoulder arthroplasty for massive irreparable rotator cuff tears in patients younger than 65 years old: results after five to fifteen years. J Shoulder Elbow Surg. 2013;22:1199-1208.
36. Klouche S, Lefevre N, Herman S, et al. Return to sport after rotator cuff tear repair: a systematic review and meta-analysis. Am J Sports Med. 2016;44:1877-1887.
37. Garcia GH, Liu JN, Wong A, et al. Hyperlipidemia increases the risk of retear after arthroscopic rotator cuff repair. J Shoulder Elbow Surg. 2017;26:2086-2090.
38. Khair MM, Lehman J, Tsouris N, et al. A systematic review of preoperative fatty infiltration and rotator cuff outcomes. HSS J. 2016;12:170-176.
39. Lambers Heerspink FO, Dorrestijn O, van Raay JJAM, et al. Specific patient-related prognostic factors for rotator cuff repair: a systematic review. J Shoulder Elbow Surg. 2014;23:1073-1080.
40. Henn RF 3rd, Kang L, Tashjian RZ, et al. Patients’ preoperative expectations predict the outcome of rotator cuff repair. J Bone Joint Surg Am. 2007;89:1913-1919.
41. Mansat P, Cofield RH, Kersten TE, et al. Complications of rotator cuff repair. Orthop Clin North Am. 1997;28:205-213.
42. Boileau P, Brassart N, Watkinson DJ, et al. Arthroscopic repair of full-thickness tears of the supraspinatus: does the tendon really heal? J Bone Joint Surg Am. 2005;87:1229-1240.
43. Galatz LM, Ball CM, Teefey SA, et al. The outcome and repair integrity of completely arthroscopically repaired large and massive rotator cuff tears. J Bone Joint Surg Am. 2004;86:219-224.
44. Aydin N, Kocaoglu B, Guven O. Single-row versus double-row arthroscopic rotator cuff repair in small- to medium-sized tears. J Shoulder Elbow Surg. 2010;19:722-725.
45. Peltz CD, Dourte LM, Kuntz AF, et al. The effect of postoperative passive motion on rotator cuff healing in a rat model. J Bone Joint Surg Am. 2009;91:2421-2429.
46. Vopat BG, Lee BJ, DeStefano S, et al. Risk factors for infection after rotator cuff repair. Arthroscopy. 2016;32:428-434.
47. Pauzenberger L, Grieb A, Hexel M, et al. Infections following arthroscopic rotator cuff repair: incidence, risk factors, and prophylaxis. Knee Surg Sports Traumatol Arthrosc. 2017;25:595-601.
48. Randelli P, Spennacchio P, Ragone V, et al. Complications associated with arthroscopic rotator cuff repair: a literature review. Musculoskelet Surg. 2012;96:9-16.
49. Hoxie SC, Sperling JW, Cofield RH. Pulmonary embolism following rotator cuff repair. Int J Shoulder Surg. 2008;2:49-51.
50. Wu XL, Briggs L, Murrell GAC. Intraoperative determinants of rotator cuff repair integrity: an analysis of 500 consecutive repairs. Am J Sports Med. 2012;40:2771-2776.
51. McElvany MD, McGoldrick E, Gee AO, et al. Rotator cuff repair: published evidence on factors associated with repair integrity and clinical outcome. Am J Sports Med. 2015;43:491-500.
52. Yoo JH, Cho NS, Rhee YG. Effect of postoperative repair integrity on health-related quality of life after rotator cuff repair: healed versus retear group. Am J Sports Med. 2013;41;2637-2644.
53. Huberty DP, Schoolfield JD, Brady PC, et al. Incidence and treatment of postoperative stiffness following arthroscopic rotator cuff repair. Arthroscopy. 2009;25:880-890.
54. Cho NS, Cha SW, Rhee YG. Alterations of the deltoid muscle after open versus arthroscopic rotator cuff repair. Am J Sports Med. 2015;43:2927-2934.
Rotator cuff disease accounts for as many as 65% of shoulder-related visits to physicians’ offices,1 yet the natural course of rotator cuff tears is still not well understood.2 Treatment options are controversial because both conservative and surgical management have been successful. Physical therapy is a durable and reliable treatment option, but there are concerns about long-term progression of the tear.3 Surgical arthroscopic techniques, which result in less morbidity than open surgery, have improved overall surgical care; as such, the rate of rotator cuff procedures has increased significantly.4
Our goal in this article is to provide clinical guidance to the primary care provider. We review management options for rotator cuff injury; summarize considerations for proceeding with conservative or surgical management; and discuss surgical risks and complications.
Conservative management: Who is most likely to benefit?
The choice of treatment for rotator cuff injury depends on a host of variables, including shoulder dominance, duration of symptoms, type of tear (partial or full), age, demands (activity level, occupation, sport), and comorbidities (diabetes, tobacco use). Treatment goals include resolution of pain, normalized range of motion and strength, and restored arm and shoulder function.5
Initial nonoperative management is indicated in patients who
- have a partial-thickness tear (a notable exception is young patients with traumatic injury),6
- have lower functional demands and moderate symptoms, or
- refuse surgery.7
Patients who respond to nonoperative management will, typically, do so within 6 to 12 weeks.5,8
Few randomized, controlled trials have compared conservative and surgical management of rotator cuff tears; furthermore, the findings of these studies have been mixed. Nonoperative management has been shown to be the favored initial treatment for isolated, symptomatic, nontraumatic, supraspinatus tears in older patients.9 In a recent study,10 5-year outcomes were examined in a prospective cohort enrolled in a rotator cuff treatment program: Approximately 75% of patients remained successfully treated with nonoperative management, and clinical outcomes of the operative and nonoperative groups were not significantly different at 5-year follow-up. Investigators concluded that nonoperative treatment is effective for many patients who have a chronic, full-thickness rotator cuff tear.
In a study investigating the treatment of degenerative rotator cuff tear, patients were randomly treated using an operative or nonoperative protocol. No differences in functional outcomes were observed at 1 year after treatment; however, surgical treatment significantly improved subjective parameters of pain and disability.11 A similar study suggested statistically significant improvement in outcomes for patients managed operatively, compared with those treated nonoperatively, but differences in shoulder outcome and the visual analog pain score were small and failed to meet thresholds considered clinically significant. Larger studies, with longer follow-up, are required to determine whether clinical differences between these types of treatment become more evident over time.12
Continue to: A look at nonoperative options and outcomes
A look at nonoperative options and outcomes
Surveillance. Rotator cuff disease of the supraspinatus tendon often results from a degenerative process that progresses to partial and, eventually, full-thickness tearing.8 Once a tear develops, progression is difficult to predict. Many rotator cuff tears grow larger over time; this progression is commonly associated with new or increased pain and weakness, or both. Although asymptomatic progression of a tear is uncommon, many patients—and physicians—are apprehensive about proceeding with nonoperative treatment for a full-thickness tear.8
To diminish such fears, surveillance can include regular assessment of shoulder motion and strength, with consideration of repeat imaging until surgery is performed or the patient is no longer a surgical candidate or interested in surgical treatment.7 Patients and providers need to remain vigilant because tears that are initially graded as repairable can become irreparable if the tendon retracts or there is fatty infiltration of the muscle belly. Results of secondary surgical repair following failed prolonged nonoperative treatment tend to be inferior to results seen in patients who undergo primary tendon repair.7
Analgesics. Simple analgesics, such as acetaminophen, are a low-risk first-line option for pain relief; however, there are limited data on the efficacy of acetaminophen in rotator cuff disease. A topical or oral nonsteroidal anti-inflammatory drug (NSAID), or both, can be considered, but potential contraindications, such as gastrointestinal, renal, and cardiovascular risks, should be monitored.13 Avoid opioids, given the potential for abuse, except during the immediate postoperative period.5
Glucocorticoid injection. Injection of a glucocorticoid drug into the subacromial space should be considered in patients whose pain interferes with sleep, limits activities of daily living, or hinders the ability to participate in physical therapy.5 A recent systematic review demonstrated that NSAIDs and glucocorticoids brought similar pain relief and active abduction at 4 to 6 weeks, but that glucocorticoids were significantly better at achieving remission of symptoms.14 There are no data comparing glucocorticoid preparations (ie, different glucocorticoids or anesthetics, dosages, volumes), and ultrasound guidance does not appear to be necessary for short-term pain relief.15 Note: Repeated injection has been shown to decrease the durability of surgically repaired tendons16; if a patient is a candidate for surgery, repeat injections should be carefully considered—and avoided if possible.
Physical therapy. The goals of physical therapy are activity modification, stretching the shoulder capsule, and strengthening the surrounding musculature (periscapular, rotator cuff, and deltoid). Patients advance through 3 phases of recovery: shoulder mobility, strengthening, and function (ie, joint reactivation to improve shoulder proprioception and coordination).
Continue to: A recent meta-analysis...
A recent meta-analysis17 found comparative evidence on treating rotator cuff tears with physical therapy to be inconclusive. At 1-year follow-up, there was no clinically significant difference between surgery and active physical therapy in either improving the Constant Shoulder Score (an assessment of function) or reducing pain caused by a rotator cuff tear. Therefore, the authors proposed, given the low risk of harm, a conservative approach should be the initial treatment modality for a tear.
A Cochrane review18 examined 60 eligible trials, in which the mean age of patients was 51 years and the mean duration of symptoms, 11 months. Overall, the review concluded that the effects of manual therapy and exercise might be similar to those of glucocorticoid injection and arthroscopic subacromial decompression. The authors noted that this conclusion is based on low-quality evidence, with only 1 study in the review that compared the combination of manual therapy and exercise to placebo.
Other conservative options. Ultrasound, topical nitroglycerin, topical lidocaine, glucocorticoid iontophoresis, transcutaneous electrical nerve stimulation, massage, acupuncture, extracorporeal shockwave therapy, hyaluronic acid, and platelet-rich plasma have been used to treat rotator cuff disease. These modalities require further study, however, to determine their effectiveness for this indication.7,19
Who is a candidate for surgical management?
Although nonoperative treatment is preferred for rotator cuff tendinitis or tendinosis and partial-thickness tears, appropriate management of full-thickness tears is debatable.20 Some surgeons advocate early operative intervention of repairable full-thickness tears to prevent further progression and reduce the risk of long-term dysfunction.
The decision to pursue operative repair depends on
- patient characteristics (age, activity level, comorbidities),
- patient function (amount of disability caused by the tear),
- characteristics of the tear (length, depth, retraction), and
- chronicity of the tear (acuity).
Continue to: TABLE 1...
TABLE 121,22 highlights variables that influence the decision to proceed, or not to proceed, with operative intervention. Because enlargement of a tear usually exacerbates symptoms,23 patients with a tear who are successfully managed nonoperatively should be counseled on the potential of the tear to progress.

What are the surgical options?
Little clinical evidence favors one exposure technique over another. This equivalency has been demonstrated by a systematic review of randomized controlled trials comparing arthroscopic and mini-open rotator cuff repair, which showed no difference in function, pain, or range of motion.24 That conclusion notwithstanding, arthroscopic repair is increasingly popular because it results in less pain, initially, and faster return to work.20
There is controversy among surgeons regarding the choice of fixation technique: Tendons can be secured using 1 or 2 rows of anchors (FIGURE). Advocates of single-row repair cite shorter surgical time, decreased cost, and equivalent outcomes; surgeons who favor double-row, or so-called transosseous-equivalent, repair claim that it provides better restoration of normal anatomy and biomechanical superiority.25,26

Regardless of technique, most patients are immobilized for 4 to 6 weeks postoperatively.27 Physical therapy usually commences within the first week or 2 postop, limited to passive motion for 6 to 12 weeks. Active motion and strengthening of rotator-cuff muscles often is initiated by 3 months postop, although this phase is sometimes delayed because of concern over slow tendon healing. Typically, patients make a full return to sports and manual work at 6 months postop. Patients experience most symptomatic improvement during the first 6 months following surgery, although functional gains can be realized for as long as 2 years after surgery.28
Most torn rotator cuffs can be fixed back to the greater tuberosity, but some chronic, massive, retracted tears lack the mobility to be repaired, or re-tear shortly after repair. Over time, the humeral head in a rotator cuff–deficient shoulder can migrate superiorly to abut the undersurface of the acromion, which can lead to significant glenohumeral osteoarthritis. To prevent or remedy elevation of the humeral head, salvage procedures—debridement, partial repair, spanning graft, tendon transfer, superior capsule reconstruction, balloon arthroplasty, reverse total shoulder replacement—can be used to alleviate pain and restore function. These procedures have significant limitations, however, and usually provide less favorable outcomes than standard repair.29-35
Continue to: Surgical outcomes
Surgical outcomes
Pain, function, and patient satisfaction outcomes following rotator cuff repair are generally favorable: 90% of patients are “happy” 6 months postop.28 Younger populations often have traumatic rotator cuff tears; they generally are interested in returning to sporting activities following their injury. Nearly 85% of younger patients who undergo rotator cuff repair return to sports, and 65.9% return to an equivalent level of play.36
Variables associated with an unfavorable outcome include increasing age, smoking, increased size of the tear, poor tendon quality, hyperlipidemia, workers’ compensation status, fatty infiltration of muscle, obesity, diabetes, and additional procedures to the biceps tendon and acromioclavicular joint performed at the time of rotator cuff repair.37-39 Interestingly, a study concluded that, if a patient expects a good surgical outcome, they are more likely to go on to report a favorable outcome—suggesting that a patient’s expectations might influence their actual outcome.40
Risks and complications
Although rotator cuff surgery has much lower morbidity than other orthopedic surgeries, it is not without risk of complications. If re-tears are excluded, postop complications have been reported in approximately 10% of patients.41 Common complications and their anticipated rate of occurrence are listed in TABLE 2.42-49

Re-tear of the surgically repaired tendon is the most common postop complication. Published re-tear rates range from 20% to 96%42,43 and generally correlate with initial tear size: A small tear is twice as likely to heal as a massive tear.50 That large range—a span of 76%—results from using a variety of methods to measure re-tear and might not have clinical meaning. A meta-analysis that examined more than 8000 shoulder surgeries reported an overall re-tear rate of 26.6%; however, both patients whose tendons healed and those who re-tore demonstrated clinical improvement.51 In a separate study, patients reported improvement in pain, function, range of motion, and satisfaction regardless of the integrity of the tendon; however, significant improvement in strength was seen only in those whose repair had healed.52
Postop stiffness is more common with arthroscopic repair than with open surgery, and with smaller rather than larger tears.53 Patient variables associated with an increased risk of postop adhesive capsulitis include workers’ compensation insurance, age < 50 years, and preoperative calcific tendonitis or adhesive capsulitis.53 Stiffness generally responds to physical therapy and rarely requires surgical lysis of adhesions or capsular release.
Continue to: Significant injury...
Significant injury to the deltoid muscle has become increasingly uncommon with the advancement of arthroscopic surgery. In traditional open surgery, detachment of the deltoid (and subsequent repair) is required to improve visualization; however, doing so can lead to atrophy and muscle rupture and dehiscence. Deltoid damage occurs in ≤ 60% of open surgeries but is negligible in arthroscopic and mini-open repairs, which involve splitting deltoid fibers to gain exposure of the underlying rotator cuff.54
SIDEBAR
Key takeaways in the management of rotator cuff injury
- Chronic, nontraumatic, and partial-thickness tears respond well to conservative management as first-line treatment. Poor surgical candidates should also be offered a trial of conservative therapy.
- Consider referral for surgical consultation if the patient does not respond to conservative therapy in 6 to 12 weeks; also, patients who have a full-thickness tear and young patients with traumatic injury should be referred for surgical consultation.
- Arthroscopy has become the preferred approach to rotator cuff repair because it is associated with less pain, fewer complications, and faster recovery.
- Patients should be counseled that recovery from surgical repair of a torn rotator cuff takes, on average, 6 months. Some massive or retracted rotator cuff injuries require more extensive procedures that increase healing time.
- Overall, patients are “happy” with rotator cuff repair at 6 months; clinical complications are uncommon, making surgery a suitable option in appropriately selected patients.
CORRESPONDENCE
Cayce Onks, DO, MS, ATC, Penn State Health Milton S. Hershey Medical Center, Penn State College of Medicine, Family and Community Medicine H154, 500 University Drive, PO Box 850, Hershey, PA 17033-0850; [email protected].
Rotator cuff disease accounts for as many as 65% of shoulder-related visits to physicians’ offices,1 yet the natural course of rotator cuff tears is still not well understood.2 Treatment options are controversial because both conservative and surgical management have been successful. Physical therapy is a durable and reliable treatment option, but there are concerns about long-term progression of the tear.3 Surgical arthroscopic techniques, which result in less morbidity than open surgery, have improved overall surgical care; as such, the rate of rotator cuff procedures has increased significantly.4
Our goal in this article is to provide clinical guidance to the primary care provider. We review management options for rotator cuff injury; summarize considerations for proceeding with conservative or surgical management; and discuss surgical risks and complications.
Conservative management: Who is most likely to benefit?
The choice of treatment for rotator cuff injury depends on a host of variables, including shoulder dominance, duration of symptoms, type of tear (partial or full), age, demands (activity level, occupation, sport), and comorbidities (diabetes, tobacco use). Treatment goals include resolution of pain, normalized range of motion and strength, and restored arm and shoulder function.5
Initial nonoperative management is indicated in patients who
- have a partial-thickness tear (a notable exception is young patients with traumatic injury),6
- have lower functional demands and moderate symptoms, or
- refuse surgery.7
Patients who respond to nonoperative management will, typically, do so within 6 to 12 weeks.5,8
Few randomized, controlled trials have compared conservative and surgical management of rotator cuff tears; furthermore, the findings of these studies have been mixed. Nonoperative management has been shown to be the favored initial treatment for isolated, symptomatic, nontraumatic, supraspinatus tears in older patients.9 In a recent study,10 5-year outcomes were examined in a prospective cohort enrolled in a rotator cuff treatment program: Approximately 75% of patients remained successfully treated with nonoperative management, and clinical outcomes of the operative and nonoperative groups were not significantly different at 5-year follow-up. Investigators concluded that nonoperative treatment is effective for many patients who have a chronic, full-thickness rotator cuff tear.
In a study investigating the treatment of degenerative rotator cuff tear, patients were randomly treated using an operative or nonoperative protocol. No differences in functional outcomes were observed at 1 year after treatment; however, surgical treatment significantly improved subjective parameters of pain and disability.11 A similar study suggested statistically significant improvement in outcomes for patients managed operatively, compared with those treated nonoperatively, but differences in shoulder outcome and the visual analog pain score were small and failed to meet thresholds considered clinically significant. Larger studies, with longer follow-up, are required to determine whether clinical differences between these types of treatment become more evident over time.12
Continue to: A look at nonoperative options and outcomes
A look at nonoperative options and outcomes
Surveillance. Rotator cuff disease of the supraspinatus tendon often results from a degenerative process that progresses to partial and, eventually, full-thickness tearing.8 Once a tear develops, progression is difficult to predict. Many rotator cuff tears grow larger over time; this progression is commonly associated with new or increased pain and weakness, or both. Although asymptomatic progression of a tear is uncommon, many patients—and physicians—are apprehensive about proceeding with nonoperative treatment for a full-thickness tear.8
To diminish such fears, surveillance can include regular assessment of shoulder motion and strength, with consideration of repeat imaging until surgery is performed or the patient is no longer a surgical candidate or interested in surgical treatment.7 Patients and providers need to remain vigilant because tears that are initially graded as repairable can become irreparable if the tendon retracts or there is fatty infiltration of the muscle belly. Results of secondary surgical repair following failed prolonged nonoperative treatment tend to be inferior to results seen in patients who undergo primary tendon repair.7
Analgesics. Simple analgesics, such as acetaminophen, are a low-risk first-line option for pain relief; however, there are limited data on the efficacy of acetaminophen in rotator cuff disease. A topical or oral nonsteroidal anti-inflammatory drug (NSAID), or both, can be considered, but potential contraindications, such as gastrointestinal, renal, and cardiovascular risks, should be monitored.13 Avoid opioids, given the potential for abuse, except during the immediate postoperative period.5
Glucocorticoid injection. Injection of a glucocorticoid drug into the subacromial space should be considered in patients whose pain interferes with sleep, limits activities of daily living, or hinders the ability to participate in physical therapy.5 A recent systematic review demonstrated that NSAIDs and glucocorticoids brought similar pain relief and active abduction at 4 to 6 weeks, but that glucocorticoids were significantly better at achieving remission of symptoms.14 There are no data comparing glucocorticoid preparations (ie, different glucocorticoids or anesthetics, dosages, volumes), and ultrasound guidance does not appear to be necessary for short-term pain relief.15 Note: Repeated injection has been shown to decrease the durability of surgically repaired tendons16; if a patient is a candidate for surgery, repeat injections should be carefully considered—and avoided if possible.
Physical therapy. The goals of physical therapy are activity modification, stretching the shoulder capsule, and strengthening the surrounding musculature (periscapular, rotator cuff, and deltoid). Patients advance through 3 phases of recovery: shoulder mobility, strengthening, and function (ie, joint reactivation to improve shoulder proprioception and coordination).
Continue to: A recent meta-analysis...
A recent meta-analysis17 found comparative evidence on treating rotator cuff tears with physical therapy to be inconclusive. At 1-year follow-up, there was no clinically significant difference between surgery and active physical therapy in either improving the Constant Shoulder Score (an assessment of function) or reducing pain caused by a rotator cuff tear. Therefore, the authors proposed, given the low risk of harm, a conservative approach should be the initial treatment modality for a tear.
A Cochrane review18 examined 60 eligible trials, in which the mean age of patients was 51 years and the mean duration of symptoms, 11 months. Overall, the review concluded that the effects of manual therapy and exercise might be similar to those of glucocorticoid injection and arthroscopic subacromial decompression. The authors noted that this conclusion is based on low-quality evidence, with only 1 study in the review that compared the combination of manual therapy and exercise to placebo.
Other conservative options. Ultrasound, topical nitroglycerin, topical lidocaine, glucocorticoid iontophoresis, transcutaneous electrical nerve stimulation, massage, acupuncture, extracorporeal shockwave therapy, hyaluronic acid, and platelet-rich plasma have been used to treat rotator cuff disease. These modalities require further study, however, to determine their effectiveness for this indication.7,19
Who is a candidate for surgical management?
Although nonoperative treatment is preferred for rotator cuff tendinitis or tendinosis and partial-thickness tears, appropriate management of full-thickness tears is debatable.20 Some surgeons advocate early operative intervention of repairable full-thickness tears to prevent further progression and reduce the risk of long-term dysfunction.
The decision to pursue operative repair depends on
- patient characteristics (age, activity level, comorbidities),
- patient function (amount of disability caused by the tear),
- characteristics of the tear (length, depth, retraction), and
- chronicity of the tear (acuity).
Continue to: TABLE 1...
TABLE 121,22 highlights variables that influence the decision to proceed, or not to proceed, with operative intervention. Because enlargement of a tear usually exacerbates symptoms,23 patients with a tear who are successfully managed nonoperatively should be counseled on the potential of the tear to progress.

What are the surgical options?
Little clinical evidence favors one exposure technique over another. This equivalency has been demonstrated by a systematic review of randomized controlled trials comparing arthroscopic and mini-open rotator cuff repair, which showed no difference in function, pain, or range of motion.24 That conclusion notwithstanding, arthroscopic repair is increasingly popular because it results in less pain, initially, and faster return to work.20
There is controversy among surgeons regarding the choice of fixation technique: Tendons can be secured using 1 or 2 rows of anchors (FIGURE). Advocates of single-row repair cite shorter surgical time, decreased cost, and equivalent outcomes; surgeons who favor double-row, or so-called transosseous-equivalent, repair claim that it provides better restoration of normal anatomy and biomechanical superiority.25,26

Regardless of technique, most patients are immobilized for 4 to 6 weeks postoperatively.27 Physical therapy usually commences within the first week or 2 postop, limited to passive motion for 6 to 12 weeks. Active motion and strengthening of rotator-cuff muscles often is initiated by 3 months postop, although this phase is sometimes delayed because of concern over slow tendon healing. Typically, patients make a full return to sports and manual work at 6 months postop. Patients experience most symptomatic improvement during the first 6 months following surgery, although functional gains can be realized for as long as 2 years after surgery.28
Most torn rotator cuffs can be fixed back to the greater tuberosity, but some chronic, massive, retracted tears lack the mobility to be repaired, or re-tear shortly after repair. Over time, the humeral head in a rotator cuff–deficient shoulder can migrate superiorly to abut the undersurface of the acromion, which can lead to significant glenohumeral osteoarthritis. To prevent or remedy elevation of the humeral head, salvage procedures—debridement, partial repair, spanning graft, tendon transfer, superior capsule reconstruction, balloon arthroplasty, reverse total shoulder replacement—can be used to alleviate pain and restore function. These procedures have significant limitations, however, and usually provide less favorable outcomes than standard repair.29-35
Continue to: Surgical outcomes
Surgical outcomes
Pain, function, and patient satisfaction outcomes following rotator cuff repair are generally favorable: 90% of patients are “happy” 6 months postop.28 Younger populations often have traumatic rotator cuff tears; they generally are interested in returning to sporting activities following their injury. Nearly 85% of younger patients who undergo rotator cuff repair return to sports, and 65.9% return to an equivalent level of play.36
Variables associated with an unfavorable outcome include increasing age, smoking, increased size of the tear, poor tendon quality, hyperlipidemia, workers’ compensation status, fatty infiltration of muscle, obesity, diabetes, and additional procedures to the biceps tendon and acromioclavicular joint performed at the time of rotator cuff repair.37-39 Interestingly, a study concluded that, if a patient expects a good surgical outcome, they are more likely to go on to report a favorable outcome—suggesting that a patient’s expectations might influence their actual outcome.40
Risks and complications
Although rotator cuff surgery has much lower morbidity than other orthopedic surgeries, it is not without risk of complications. If re-tears are excluded, postop complications have been reported in approximately 10% of patients.41 Common complications and their anticipated rate of occurrence are listed in TABLE 2.42-49

Re-tear of the surgically repaired tendon is the most common postop complication. Published re-tear rates range from 20% to 96%42,43 and generally correlate with initial tear size: A small tear is twice as likely to heal as a massive tear.50 That large range—a span of 76%—results from using a variety of methods to measure re-tear and might not have clinical meaning. A meta-analysis that examined more than 8000 shoulder surgeries reported an overall re-tear rate of 26.6%; however, both patients whose tendons healed and those who re-tore demonstrated clinical improvement.51 In a separate study, patients reported improvement in pain, function, range of motion, and satisfaction regardless of the integrity of the tendon; however, significant improvement in strength was seen only in those whose repair had healed.52
Postop stiffness is more common with arthroscopic repair than with open surgery, and with smaller rather than larger tears.53 Patient variables associated with an increased risk of postop adhesive capsulitis include workers’ compensation insurance, age < 50 years, and preoperative calcific tendonitis or adhesive capsulitis.53 Stiffness generally responds to physical therapy and rarely requires surgical lysis of adhesions or capsular release.
Continue to: Significant injury...
Significant injury to the deltoid muscle has become increasingly uncommon with the advancement of arthroscopic surgery. In traditional open surgery, detachment of the deltoid (and subsequent repair) is required to improve visualization; however, doing so can lead to atrophy and muscle rupture and dehiscence. Deltoid damage occurs in ≤ 60% of open surgeries but is negligible in arthroscopic and mini-open repairs, which involve splitting deltoid fibers to gain exposure of the underlying rotator cuff.54
SIDEBAR
Key takeaways in the management of rotator cuff injury
- Chronic, nontraumatic, and partial-thickness tears respond well to conservative management as first-line treatment. Poor surgical candidates should also be offered a trial of conservative therapy.
- Consider referral for surgical consultation if the patient does not respond to conservative therapy in 6 to 12 weeks; also, patients who have a full-thickness tear and young patients with traumatic injury should be referred for surgical consultation.
- Arthroscopy has become the preferred approach to rotator cuff repair because it is associated with less pain, fewer complications, and faster recovery.
- Patients should be counseled that recovery from surgical repair of a torn rotator cuff takes, on average, 6 months. Some massive or retracted rotator cuff injuries require more extensive procedures that increase healing time.
- Overall, patients are “happy” with rotator cuff repair at 6 months; clinical complications are uncommon, making surgery a suitable option in appropriately selected patients.
CORRESPONDENCE
Cayce Onks, DO, MS, ATC, Penn State Health Milton S. Hershey Medical Center, Penn State College of Medicine, Family and Community Medicine H154, 500 University Drive, PO Box 850, Hershey, PA 17033-0850; [email protected].
1. Vecchio P, Kavanagh R, Hazleman BL, et al. Shoulder pain in a community-based rheumatology clinic. Br J Rheumatol. 1995;34:440-442.
2. Eljabu W, Klinger HM, von Knoch M. The natural history of rotator cuff tears: a systematic review. Arch Orthop Trauma Surg. 2015;135:1055-1061.
3. Dunn WR, Kuhn JE, Sanders R, et al; MOON Shoulder Group. 2013 Neer Award: predictors of failure of nonoperative treatment of chronic, symptomatic, full-thickness rotator cuff tears. J Shoulder Elbow Surg. 2016;25:1303-1311.
4. Colvin AC, Egorova N, Harrison AK, et al. National trends in rotator cuff repair. J Bone Joint Surg Am. 2012;94:227-233.
5. Whittle S, Buchbinder R. In the clinic. Rotator cuff disease. Ann Intern Med. 2015;162:ITC1-ITC15.
6. Lazarides AL, Alentorn-Geli E, Choi JHJ, et al. Rotator cuff tears in young patients: a different disease than rotator cuff tears in elderly patients. J Shoulder Elbow Surg. 2015;24:1834-1843.
7. Petri M, Ettinger M, Brand S, et al. Non-operative management of rotator cuff tears. Open Orthop J. 2016;10:349-356.
8. Schmidt CC, Jarrett CD, Brown BT. Management of rotator cuff tears. J Hand Surg Am. 2015;40:399-408.
9. Kukkonen J, Joukainen A, Lehtinen J, et al. Treatment of nontraumatic rotator cuff tears: a randomized controlled trial with two years of clinical and imaging follow-up. J Bone Joint Surg Am. 2015;97:1729-1737.
10. Boorman RS, More KD, Hollinshead RM, et al. What happens to patients when we do not repair their cuff tears? Five-year rotator cuff quality-of-life index outcomes following nonoperative treatment of patients with full-thickness rotator cuff tears. J Shoulder Elbow Surg. 2018;27:444-448.
11. Lambers Heerspink FO, van Raay JJ, Koorevaar RCT, et al. Comparing surgical repair with conservative treatment for degenerative rotator cuff tears: a randomized controlled trial. J Shoulder Elbow Surg. 2015;24:1274-1281.
12. Piper CC, Hughes AJ, Ma Y, et al. Operative versus nonoperative treatment for the management of full-thickness rotator cuff tears: a systematic review and meta-analysis. J Shoulder Elbow Surg. 2018;27:572-576.
13. Boudreault J, Desmeules F, Roy J-S, et al. The efficacy of oral non-steroidal anti-inflammatory drugs for rotator cuff tendinopathy: a systematic review and meta-analysis. J Rehabil Med. 2014;46:294-306.
14. Zheng X-Q, Li K, Wei Y-D, et al. Nonsteroidal anti-inflammatory drugs versus corticosteroid for treatment of shoulder pain: a systematic review and meta-analysis. Arch Phys Med Rehabil. 2014;95:1824-1831.
15. Bloom JE, Rischin A, Johnston RV, et al. Image-guided versus blind glucocorticoid injection for shoulder pain. Cochrane Database Syst Rev. 2012;(8):CD009147.
16. Wiggins ME, Fadale PD, Ehrlich MG, et al. Effects of local injection of corticosteroids on the healing of ligaments. A follow-up report. J Bone Joint Surg Am. 1995;77:1682-1691.
17. Ryösä A, Laimi K, Äärimaa V, et al. Surgery or conservative treatment for rotator cuff tear: a meta-analysis. Disabil Rehabil. 2017;39:1357-1363.
18. Page MJ, Green S, McBain B, et al. Manual therapy and exercise for rotator cuff disease. Cochrane Database Syst Rev. 2016;(6):CD012224.
19. Page MJ, Green S, Mrocki MA, et al. Electrotherapy modalities for rotator cuff disease. Cochrane Database Syst Rev. 2016;(6):CD012225.
20. Acevedo DC, Paxton ES, Williams GR, et al. A survey of expert opinion regarding rotator cuff repair. J Bone Joint Surg Am. 2014;96:e123.
21. Pedowitz RA, Yamaguchi K, Ahmad CS, et al. American Academy of Orthopaedic Surgeons Clinical Practice Guideline on: optimizing the management of rotator cuff problems. J Bone Joint Surg Am. 2012;94:163-167.
22. Thorpe A, Hurworth M, O’Sullivan P, et al. Rotator cuff disease: opinion regarding surgical criteria and likely outcome. ANZ J Surg. 2017;87:291-295.
23. Mall NA, Kim HM, Keener JD, et al. Symptomatic progression of asymptomatic rotator cuff tears: a prospective study of clinical and sonographic variables. J Bone Joint Surg Am. 2010;92:2623-2633.
24. Ji X, Bi C, Wang F, et al. Arthroscopic versus mini-open rotator cuff repair: an up-to-date meta-analysis of randomized controlled trials. Arthroscopy. 2015;31:118-124.
25. Duquin TR, Buyea C, Bisson LJ. Which method of rotator cuff repair leads to the highest rate of structural healing? A systematic review. Am J Sports Med. 2010;38:835-841.
26. Choi S, Kim MK, Kim GM, et al. Factors associated with clinical and structural outcomes after arthroscopic rotator cuff repair with a suture bridge technique in medium, large, and massive tears. J Shoulder Elbow Surg. 2014;23:1675-1681.
27. Shen C, Tang Z-H, Hu J-Z, et al. Does immobilization after arthroscopic rotator cuff repair increase tendon healing? A systematic review and meta-analysis. Arch Orthop Trauma Surg. 2014;134:1279-1285.
28. Gulotta LV, Nho SJ, Dodson CC, et al; . Prospective evaluation of arthroscopic rotator cuff repairs at 5 years: part I. Functional outcomes and radiographic healing rates. J Shoulder Elbow Surg. 2011;20:934-940.
29. Liem D, Lengers N, Dedy N, et al. Arthroscopic debridement of massive irreparable rotator cuff tears. Arthroscopy. 2008;24:743-748.
30. Weber SC. Partial rotator cuff repair in massive rotator cuff tears: long-term follow-up. J Shoulder Elbow Surg. 2017;26:e171.
31. Lewington MR, Ferguson DP, Smith TD, et al. Graft utilization in the bridging reconstruction of irreparable rotator cuff tears: a systematic review. Am J Sports Med. 2017;45:3149-3157.
32. Longo UG, Franceschetti E, Petrillo S, et al. Latissimus dorsi tendon transfer for massive irreparable rotator cuff tears: a systematic review. Sports Med Arthrosc Rev. 2011;19:428-437.
33. Noyes MP, Denard PJ. Arthroscopic superior capsular reconstruction: indications and outcomes. Oper Tech Sports Med. 2018;26:29-34.
34. Piekaar RSM, Bouman ICE, van Kampen PM, et al. Early promising outcome following arthroscopic implantation of the subacromial balloon spacer for treating massive rotator cuff tear. Musculoskeletal Surg. 2018;102:247-255.
35. Ek ETH, Neukom L, Catanzaro S, et al. Reverse total shoulder arthroplasty for massive irreparable rotator cuff tears in patients younger than 65 years old: results after five to fifteen years. J Shoulder Elbow Surg. 2013;22:1199-1208.
36. Klouche S, Lefevre N, Herman S, et al. Return to sport after rotator cuff tear repair: a systematic review and meta-analysis. Am J Sports Med. 2016;44:1877-1887.
37. Garcia GH, Liu JN, Wong A, et al. Hyperlipidemia increases the risk of retear after arthroscopic rotator cuff repair. J Shoulder Elbow Surg. 2017;26:2086-2090.
38. Khair MM, Lehman J, Tsouris N, et al. A systematic review of preoperative fatty infiltration and rotator cuff outcomes. HSS J. 2016;12:170-176.
39. Lambers Heerspink FO, Dorrestijn O, van Raay JJAM, et al. Specific patient-related prognostic factors for rotator cuff repair: a systematic review. J Shoulder Elbow Surg. 2014;23:1073-1080.
40. Henn RF 3rd, Kang L, Tashjian RZ, et al. Patients’ preoperative expectations predict the outcome of rotator cuff repair. J Bone Joint Surg Am. 2007;89:1913-1919.
41. Mansat P, Cofield RH, Kersten TE, et al. Complications of rotator cuff repair. Orthop Clin North Am. 1997;28:205-213.
42. Boileau P, Brassart N, Watkinson DJ, et al. Arthroscopic repair of full-thickness tears of the supraspinatus: does the tendon really heal? J Bone Joint Surg Am. 2005;87:1229-1240.
43. Galatz LM, Ball CM, Teefey SA, et al. The outcome and repair integrity of completely arthroscopically repaired large and massive rotator cuff tears. J Bone Joint Surg Am. 2004;86:219-224.
44. Aydin N, Kocaoglu B, Guven O. Single-row versus double-row arthroscopic rotator cuff repair in small- to medium-sized tears. J Shoulder Elbow Surg. 2010;19:722-725.
45. Peltz CD, Dourte LM, Kuntz AF, et al. The effect of postoperative passive motion on rotator cuff healing in a rat model. J Bone Joint Surg Am. 2009;91:2421-2429.
46. Vopat BG, Lee BJ, DeStefano S, et al. Risk factors for infection after rotator cuff repair. Arthroscopy. 2016;32:428-434.
47. Pauzenberger L, Grieb A, Hexel M, et al. Infections following arthroscopic rotator cuff repair: incidence, risk factors, and prophylaxis. Knee Surg Sports Traumatol Arthrosc. 2017;25:595-601.
48. Randelli P, Spennacchio P, Ragone V, et al. Complications associated with arthroscopic rotator cuff repair: a literature review. Musculoskelet Surg. 2012;96:9-16.
49. Hoxie SC, Sperling JW, Cofield RH. Pulmonary embolism following rotator cuff repair. Int J Shoulder Surg. 2008;2:49-51.
50. Wu XL, Briggs L, Murrell GAC. Intraoperative determinants of rotator cuff repair integrity: an analysis of 500 consecutive repairs. Am J Sports Med. 2012;40:2771-2776.
51. McElvany MD, McGoldrick E, Gee AO, et al. Rotator cuff repair: published evidence on factors associated with repair integrity and clinical outcome. Am J Sports Med. 2015;43:491-500.
52. Yoo JH, Cho NS, Rhee YG. Effect of postoperative repair integrity on health-related quality of life after rotator cuff repair: healed versus retear group. Am J Sports Med. 2013;41;2637-2644.
53. Huberty DP, Schoolfield JD, Brady PC, et al. Incidence and treatment of postoperative stiffness following arthroscopic rotator cuff repair. Arthroscopy. 2009;25:880-890.
54. Cho NS, Cha SW, Rhee YG. Alterations of the deltoid muscle after open versus arthroscopic rotator cuff repair. Am J Sports Med. 2015;43:2927-2934.
1. Vecchio P, Kavanagh R, Hazleman BL, et al. Shoulder pain in a community-based rheumatology clinic. Br J Rheumatol. 1995;34:440-442.
2. Eljabu W, Klinger HM, von Knoch M. The natural history of rotator cuff tears: a systematic review. Arch Orthop Trauma Surg. 2015;135:1055-1061.
3. Dunn WR, Kuhn JE, Sanders R, et al; MOON Shoulder Group. 2013 Neer Award: predictors of failure of nonoperative treatment of chronic, symptomatic, full-thickness rotator cuff tears. J Shoulder Elbow Surg. 2016;25:1303-1311.
4. Colvin AC, Egorova N, Harrison AK, et al. National trends in rotator cuff repair. J Bone Joint Surg Am. 2012;94:227-233.
5. Whittle S, Buchbinder R. In the clinic. Rotator cuff disease. Ann Intern Med. 2015;162:ITC1-ITC15.
6. Lazarides AL, Alentorn-Geli E, Choi JHJ, et al. Rotator cuff tears in young patients: a different disease than rotator cuff tears in elderly patients. J Shoulder Elbow Surg. 2015;24:1834-1843.
7. Petri M, Ettinger M, Brand S, et al. Non-operative management of rotator cuff tears. Open Orthop J. 2016;10:349-356.
8. Schmidt CC, Jarrett CD, Brown BT. Management of rotator cuff tears. J Hand Surg Am. 2015;40:399-408.
9. Kukkonen J, Joukainen A, Lehtinen J, et al. Treatment of nontraumatic rotator cuff tears: a randomized controlled trial with two years of clinical and imaging follow-up. J Bone Joint Surg Am. 2015;97:1729-1737.
10. Boorman RS, More KD, Hollinshead RM, et al. What happens to patients when we do not repair their cuff tears? Five-year rotator cuff quality-of-life index outcomes following nonoperative treatment of patients with full-thickness rotator cuff tears. J Shoulder Elbow Surg. 2018;27:444-448.
11. Lambers Heerspink FO, van Raay JJ, Koorevaar RCT, et al. Comparing surgical repair with conservative treatment for degenerative rotator cuff tears: a randomized controlled trial. J Shoulder Elbow Surg. 2015;24:1274-1281.
12. Piper CC, Hughes AJ, Ma Y, et al. Operative versus nonoperative treatment for the management of full-thickness rotator cuff tears: a systematic review and meta-analysis. J Shoulder Elbow Surg. 2018;27:572-576.
13. Boudreault J, Desmeules F, Roy J-S, et al. The efficacy of oral non-steroidal anti-inflammatory drugs for rotator cuff tendinopathy: a systematic review and meta-analysis. J Rehabil Med. 2014;46:294-306.
14. Zheng X-Q, Li K, Wei Y-D, et al. Nonsteroidal anti-inflammatory drugs versus corticosteroid for treatment of shoulder pain: a systematic review and meta-analysis. Arch Phys Med Rehabil. 2014;95:1824-1831.
15. Bloom JE, Rischin A, Johnston RV, et al. Image-guided versus blind glucocorticoid injection for shoulder pain. Cochrane Database Syst Rev. 2012;(8):CD009147.
16. Wiggins ME, Fadale PD, Ehrlich MG, et al. Effects of local injection of corticosteroids on the healing of ligaments. A follow-up report. J Bone Joint Surg Am. 1995;77:1682-1691.
17. Ryösä A, Laimi K, Äärimaa V, et al. Surgery or conservative treatment for rotator cuff tear: a meta-analysis. Disabil Rehabil. 2017;39:1357-1363.
18. Page MJ, Green S, McBain B, et al. Manual therapy and exercise for rotator cuff disease. Cochrane Database Syst Rev. 2016;(6):CD012224.
19. Page MJ, Green S, Mrocki MA, et al. Electrotherapy modalities for rotator cuff disease. Cochrane Database Syst Rev. 2016;(6):CD012225.
20. Acevedo DC, Paxton ES, Williams GR, et al. A survey of expert opinion regarding rotator cuff repair. J Bone Joint Surg Am. 2014;96:e123.
21. Pedowitz RA, Yamaguchi K, Ahmad CS, et al. American Academy of Orthopaedic Surgeons Clinical Practice Guideline on: optimizing the management of rotator cuff problems. J Bone Joint Surg Am. 2012;94:163-167.
22. Thorpe A, Hurworth M, O’Sullivan P, et al. Rotator cuff disease: opinion regarding surgical criteria and likely outcome. ANZ J Surg. 2017;87:291-295.
23. Mall NA, Kim HM, Keener JD, et al. Symptomatic progression of asymptomatic rotator cuff tears: a prospective study of clinical and sonographic variables. J Bone Joint Surg Am. 2010;92:2623-2633.
24. Ji X, Bi C, Wang F, et al. Arthroscopic versus mini-open rotator cuff repair: an up-to-date meta-analysis of randomized controlled trials. Arthroscopy. 2015;31:118-124.
25. Duquin TR, Buyea C, Bisson LJ. Which method of rotator cuff repair leads to the highest rate of structural healing? A systematic review. Am J Sports Med. 2010;38:835-841.
26. Choi S, Kim MK, Kim GM, et al. Factors associated with clinical and structural outcomes after arthroscopic rotator cuff repair with a suture bridge technique in medium, large, and massive tears. J Shoulder Elbow Surg. 2014;23:1675-1681.
27. Shen C, Tang Z-H, Hu J-Z, et al. Does immobilization after arthroscopic rotator cuff repair increase tendon healing? A systematic review and meta-analysis. Arch Orthop Trauma Surg. 2014;134:1279-1285.
28. Gulotta LV, Nho SJ, Dodson CC, et al; . Prospective evaluation of arthroscopic rotator cuff repairs at 5 years: part I. Functional outcomes and radiographic healing rates. J Shoulder Elbow Surg. 2011;20:934-940.
29. Liem D, Lengers N, Dedy N, et al. Arthroscopic debridement of massive irreparable rotator cuff tears. Arthroscopy. 2008;24:743-748.
30. Weber SC. Partial rotator cuff repair in massive rotator cuff tears: long-term follow-up. J Shoulder Elbow Surg. 2017;26:e171.
31. Lewington MR, Ferguson DP, Smith TD, et al. Graft utilization in the bridging reconstruction of irreparable rotator cuff tears: a systematic review. Am J Sports Med. 2017;45:3149-3157.
32. Longo UG, Franceschetti E, Petrillo S, et al. Latissimus dorsi tendon transfer for massive irreparable rotator cuff tears: a systematic review. Sports Med Arthrosc Rev. 2011;19:428-437.
33. Noyes MP, Denard PJ. Arthroscopic superior capsular reconstruction: indications and outcomes. Oper Tech Sports Med. 2018;26:29-34.
34. Piekaar RSM, Bouman ICE, van Kampen PM, et al. Early promising outcome following arthroscopic implantation of the subacromial balloon spacer for treating massive rotator cuff tear. Musculoskeletal Surg. 2018;102:247-255.
35. Ek ETH, Neukom L, Catanzaro S, et al. Reverse total shoulder arthroplasty for massive irreparable rotator cuff tears in patients younger than 65 years old: results after five to fifteen years. J Shoulder Elbow Surg. 2013;22:1199-1208.
36. Klouche S, Lefevre N, Herman S, et al. Return to sport after rotator cuff tear repair: a systematic review and meta-analysis. Am J Sports Med. 2016;44:1877-1887.
37. Garcia GH, Liu JN, Wong A, et al. Hyperlipidemia increases the risk of retear after arthroscopic rotator cuff repair. J Shoulder Elbow Surg. 2017;26:2086-2090.
38. Khair MM, Lehman J, Tsouris N, et al. A systematic review of preoperative fatty infiltration and rotator cuff outcomes. HSS J. 2016;12:170-176.
39. Lambers Heerspink FO, Dorrestijn O, van Raay JJAM, et al. Specific patient-related prognostic factors for rotator cuff repair: a systematic review. J Shoulder Elbow Surg. 2014;23:1073-1080.
40. Henn RF 3rd, Kang L, Tashjian RZ, et al. Patients’ preoperative expectations predict the outcome of rotator cuff repair. J Bone Joint Surg Am. 2007;89:1913-1919.
41. Mansat P, Cofield RH, Kersten TE, et al. Complications of rotator cuff repair. Orthop Clin North Am. 1997;28:205-213.
42. Boileau P, Brassart N, Watkinson DJ, et al. Arthroscopic repair of full-thickness tears of the supraspinatus: does the tendon really heal? J Bone Joint Surg Am. 2005;87:1229-1240.
43. Galatz LM, Ball CM, Teefey SA, et al. The outcome and repair integrity of completely arthroscopically repaired large and massive rotator cuff tears. J Bone Joint Surg Am. 2004;86:219-224.
44. Aydin N, Kocaoglu B, Guven O. Single-row versus double-row arthroscopic rotator cuff repair in small- to medium-sized tears. J Shoulder Elbow Surg. 2010;19:722-725.
45. Peltz CD, Dourte LM, Kuntz AF, et al. The effect of postoperative passive motion on rotator cuff healing in a rat model. J Bone Joint Surg Am. 2009;91:2421-2429.
46. Vopat BG, Lee BJ, DeStefano S, et al. Risk factors for infection after rotator cuff repair. Arthroscopy. 2016;32:428-434.
47. Pauzenberger L, Grieb A, Hexel M, et al. Infections following arthroscopic rotator cuff repair: incidence, risk factors, and prophylaxis. Knee Surg Sports Traumatol Arthrosc. 2017;25:595-601.
48. Randelli P, Spennacchio P, Ragone V, et al. Complications associated with arthroscopic rotator cuff repair: a literature review. Musculoskelet Surg. 2012;96:9-16.
49. Hoxie SC, Sperling JW, Cofield RH. Pulmonary embolism following rotator cuff repair. Int J Shoulder Surg. 2008;2:49-51.
50. Wu XL, Briggs L, Murrell GAC. Intraoperative determinants of rotator cuff repair integrity: an analysis of 500 consecutive repairs. Am J Sports Med. 2012;40:2771-2776.
51. McElvany MD, McGoldrick E, Gee AO, et al. Rotator cuff repair: published evidence on factors associated with repair integrity and clinical outcome. Am J Sports Med. 2015;43:491-500.
52. Yoo JH, Cho NS, Rhee YG. Effect of postoperative repair integrity on health-related quality of life after rotator cuff repair: healed versus retear group. Am J Sports Med. 2013;41;2637-2644.
53. Huberty DP, Schoolfield JD, Brady PC, et al. Incidence and treatment of postoperative stiffness following arthroscopic rotator cuff repair. Arthroscopy. 2009;25:880-890.
54. Cho NS, Cha SW, Rhee YG. Alterations of the deltoid muscle after open versus arthroscopic rotator cuff repair. Am J Sports Med. 2015;43:2927-2934.
PRACTICE RECOMMENDATIONS
› Offer a trial of conservative management to patients with chronic, nontraumatic, or partial-thickness rotator cuff injury and to those who are poor surgical candidates. B
› Counsel patients that the rate of surgical complications is low and outcomes are favorable in properly selected patients for operative repair of rotator cuff tear. B
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
Joint replacement: What’s new in 2020
MAUI, HAWAII – Outpatient total hip and knee replacement is “the latest craze” in orthopedic surgery, and it’s being driven by the might of Medicare, William Bugbee, MD, said at the 2020 Rheumatology Winter Clinical Symposium.
“In 2019, Medicare took total knee replacement off the inpatient-only list, meaning you could do it as an outpatient. And just in January 2020, they took total hips off that list. So I have to designate most of my hip and knee replacements as outpatients, even if I do it in the hospital and keep them for 1 night. And some of the private insurers have already gone to that, so they’ll deny coverage if I say I want a 1-day hospital stay, believe it or not,” according to Dr. Bugbee, chief of joint reconstruction in the department of orthopedics at the Scripps Clinic in La Jolla, Calif.
He provided a behind-the-scenes look at contemporary trends in joint replacement as well as tips on how rheumatologists can best help their patients get through the experience with excellent outcomes.
Joint replacement remains the best treatment for advanced arthritis of the hips and knees, he said. There is a high degree of confidence about the predictability and durability of the results. But joint replacement has become highly commoditized.
“We’re getting pummeled by Medicare to make this as cheap as possible,” the orthopedic surgeon explained. “An implant costs the hospital $3,000-$6,000. A care episode for a primary total joint replacement should cost a hospital $8,000-$15,000, which is about what Medicare pays for the [Diagnosis Related Group], so the margins are small. That’s why we’re being drilled on about how much we spend on every little thing. We hardly do any labs, x-rays, anything.”
As a result of recent advances in pre-, peri-, and postoperative management, outpatient joint replacement has become a safe and comparatively economical option for generally healthy patients.
“We’ve engineered a much better patient experience, so the assault and battery of 5, 10, 15 years ago isn’t so bad anymore,” Dr. Bugbee said.
Rheumatologists can expect to see a growing number of their patients undergoing total knee or hip replacement at outpatient surgery centers. That’s not a bad thing so long as the procedure is being done there because the outpatient center employs best practices in order to provide a highly efficient episode of care supported by excellent outcome data, he continued.
State-of-the-art perioperative management in 2020 includes accelerated-care pathways that allow ambulation within an hour or 2 after surgery along with same-day discharge, regional anesthesia with motor-sparing nerve blocks, and multimodal pain management with avoidance of intravenous narcotics except in opioid-tolerant patients. Tranexamic acid is now widely used in order to reduce operative blood loss.
“When I started practice 25 years ago, 50% of patients got a blood transfusion. I haven’t given a blood transfusion to a patient in probably 2 years. Tranexamic acid reduces blood loss by 500-700 cc with no discernible adverse effects. It’s truly remarkable,” he said.
Another important technical advance has been the routine use of oral dexamethasone. “Decadron is an antiemetic, it has anti-inflammatory effects, and it makes people happy. It’s a simple, cheap drug that has revolutionized care,” the surgeon continued.
Postoperative management has been streamlined. Dr. Bugbee is among many orthopedic surgeons who no longer routinely prescribe therapist-directed formal physical therapy for total hip arthroplasty patients, relying instead upon online tools and apps for self-administered physical therapy. Pedal exercise devices available online for $30 or so have been shown to be as effective as supervised physical therapy for knee rehabilitation.
What patients want to know about joint replacement
The question patients most often ask both their referring physician and the orthopedic surgeon is, “How long will my joint replacement last?” The best available data come from a couple of recent paired meta-analyses. The investigators reported 82% implant survivorship 25 years after primary total knee arthroplasty and 70% after unicondylar knee arthroplasty as well as a 25-year implant survivorship rate of 77% for total hip arthroplasty.
“I expected that hip arthroplasty survivorship rate to be much higher than 77%. The reason it’s not is probably because of the metal-on-metal bearing surface debacle of about 10 years ago. There’ve been lots of revisions because of that. We thought metal-on-metal implants were going to be all that, with microscopically low wear, but they turned out to be a nightmare because of metal ion release,” Dr. Bugbee observed.
The long-term joint survivorship data are based upon older implants. Encouraging albeit still preliminary data suggest contemporary implants may last significantly longer. The “clear winner,” he said, is a 36-mm ceramic head and a highly crosslinked polyethylene liner.
“That’s been a game changer, with a 10- to 20-fold decrease in wear compared to plastics for weight-bearing surfaces,” Dr. Bugbee said.
In terms of functional improvement, by various measures 85%-97% of patients are satisfied with the results of their total hip replacement, and 60% report returning to high-level recreational activities. Patient satisfaction scores are lower – 75%-90% – after total knee arthroplasty.
“The total knee replacement just doesn’t work like a regular joint,” the surgeon observed. “When I think of hip and knee replacements, I think of a hip as a Ferrari – it’s a high-performance joint replacement – and I think of the knee as a Ford – it’s serviceable, it does the job, and it’s okay but not fantastic.”
How referring physicians can optimize preoperative management and long-term follow-up
Orthopedic surgeons would appreciate help from rheumatologists and primary care physicians in preoperatively addressing the known modifiable risk factors for poor outcomes of joint replacement. These include obesity, smoking, depression, a hemoglobin A1c of 7% or more, and being on opioids. These risk factors are incompatible with outpatient hip or knee replacement.
“Let the surgeon know if you think outpatient joint replacement is a bad idea in your patient for medical reasons,” Dr. Bugbee urged.
Also, orthopedic surgeons can generally benefit from rheumatologist input regarding perioperative management of patients on standard disease-modifying antirheumatic drugs, biologics, or Janus kinase inhibitors as recommended in the guidelines published jointly by the American College of Rheumatology and the American Association of Hip and Knee Surgeons.
“I can guarantee you that most orthopedic surgeons don’t know about these guidelines. The evidence base for these recommendations is not great, but these are the best guidelines we have,” Dr. Bugbee said.
After joint replacement surgery a patient should get an x-ray of the replacement every 5 years. And if a patient develops a painful hip after arthroplasty, it’s worthwhile to order blood chromium and cobalt levels.
“The implant weight-bearing surface matters. You can’t necessarily tell on x-ray what’s a metal-on-metal hip and what’s metal-on-plastic or ceramic. You already send people for a lot of labs. If you see a patient with a painful total hip replacement, just add a cobalt and chromium. If they’re elevated, talk to the orthopedist,” he advised.
The road ahead
Hip and knee replacement is an $18 billion market today. And it’s a major growth industry: According to a recent projection, there will be 1 million total hip replacements and 4 million total knee replacements annually 10 years from now, figures four times greater than projected for 2030 in an earlier 2005 estimate. The rapid growth is coming from the expanding elderly population combined with a virtual epidemic of posttraumatic arthritis in young people – but decidedly not from patients with joint failure attributable to rheumatoid arthritis.
“Congratulations! You’ve eradicated rheumatoid arthritis from my practice,” Dr. Bugbee declared. “Most of the rheumatoid arthritis patients who come to me come because they have osteoarthritis in their joint, not because of their rheumatoid arthritis.”
He reported serving as a consultant to Orthalign, Insight Medical, and Arthrex, and receiving royalties from Smith and Nephew and Depuy.
MAUI, HAWAII – Outpatient total hip and knee replacement is “the latest craze” in orthopedic surgery, and it’s being driven by the might of Medicare, William Bugbee, MD, said at the 2020 Rheumatology Winter Clinical Symposium.
“In 2019, Medicare took total knee replacement off the inpatient-only list, meaning you could do it as an outpatient. And just in January 2020, they took total hips off that list. So I have to designate most of my hip and knee replacements as outpatients, even if I do it in the hospital and keep them for 1 night. And some of the private insurers have already gone to that, so they’ll deny coverage if I say I want a 1-day hospital stay, believe it or not,” according to Dr. Bugbee, chief of joint reconstruction in the department of orthopedics at the Scripps Clinic in La Jolla, Calif.
He provided a behind-the-scenes look at contemporary trends in joint replacement as well as tips on how rheumatologists can best help their patients get through the experience with excellent outcomes.
Joint replacement remains the best treatment for advanced arthritis of the hips and knees, he said. There is a high degree of confidence about the predictability and durability of the results. But joint replacement has become highly commoditized.
“We’re getting pummeled by Medicare to make this as cheap as possible,” the orthopedic surgeon explained. “An implant costs the hospital $3,000-$6,000. A care episode for a primary total joint replacement should cost a hospital $8,000-$15,000, which is about what Medicare pays for the [Diagnosis Related Group], so the margins are small. That’s why we’re being drilled on about how much we spend on every little thing. We hardly do any labs, x-rays, anything.”
As a result of recent advances in pre-, peri-, and postoperative management, outpatient joint replacement has become a safe and comparatively economical option for generally healthy patients.
“We’ve engineered a much better patient experience, so the assault and battery of 5, 10, 15 years ago isn’t so bad anymore,” Dr. Bugbee said.
Rheumatologists can expect to see a growing number of their patients undergoing total knee or hip replacement at outpatient surgery centers. That’s not a bad thing so long as the procedure is being done there because the outpatient center employs best practices in order to provide a highly efficient episode of care supported by excellent outcome data, he continued.
State-of-the-art perioperative management in 2020 includes accelerated-care pathways that allow ambulation within an hour or 2 after surgery along with same-day discharge, regional anesthesia with motor-sparing nerve blocks, and multimodal pain management with avoidance of intravenous narcotics except in opioid-tolerant patients. Tranexamic acid is now widely used in order to reduce operative blood loss.
“When I started practice 25 years ago, 50% of patients got a blood transfusion. I haven’t given a blood transfusion to a patient in probably 2 years. Tranexamic acid reduces blood loss by 500-700 cc with no discernible adverse effects. It’s truly remarkable,” he said.
Another important technical advance has been the routine use of oral dexamethasone. “Decadron is an antiemetic, it has anti-inflammatory effects, and it makes people happy. It’s a simple, cheap drug that has revolutionized care,” the surgeon continued.
Postoperative management has been streamlined. Dr. Bugbee is among many orthopedic surgeons who no longer routinely prescribe therapist-directed formal physical therapy for total hip arthroplasty patients, relying instead upon online tools and apps for self-administered physical therapy. Pedal exercise devices available online for $30 or so have been shown to be as effective as supervised physical therapy for knee rehabilitation.
What patients want to know about joint replacement
The question patients most often ask both their referring physician and the orthopedic surgeon is, “How long will my joint replacement last?” The best available data come from a couple of recent paired meta-analyses. The investigators reported 82% implant survivorship 25 years after primary total knee arthroplasty and 70% after unicondylar knee arthroplasty as well as a 25-year implant survivorship rate of 77% for total hip arthroplasty.
“I expected that hip arthroplasty survivorship rate to be much higher than 77%. The reason it’s not is probably because of the metal-on-metal bearing surface debacle of about 10 years ago. There’ve been lots of revisions because of that. We thought metal-on-metal implants were going to be all that, with microscopically low wear, but they turned out to be a nightmare because of metal ion release,” Dr. Bugbee observed.
The long-term joint survivorship data are based upon older implants. Encouraging albeit still preliminary data suggest contemporary implants may last significantly longer. The “clear winner,” he said, is a 36-mm ceramic head and a highly crosslinked polyethylene liner.
“That’s been a game changer, with a 10- to 20-fold decrease in wear compared to plastics for weight-bearing surfaces,” Dr. Bugbee said.
In terms of functional improvement, by various measures 85%-97% of patients are satisfied with the results of their total hip replacement, and 60% report returning to high-level recreational activities. Patient satisfaction scores are lower – 75%-90% – after total knee arthroplasty.
“The total knee replacement just doesn’t work like a regular joint,” the surgeon observed. “When I think of hip and knee replacements, I think of a hip as a Ferrari – it’s a high-performance joint replacement – and I think of the knee as a Ford – it’s serviceable, it does the job, and it’s okay but not fantastic.”
How referring physicians can optimize preoperative management and long-term follow-up
Orthopedic surgeons would appreciate help from rheumatologists and primary care physicians in preoperatively addressing the known modifiable risk factors for poor outcomes of joint replacement. These include obesity, smoking, depression, a hemoglobin A1c of 7% or more, and being on opioids. These risk factors are incompatible with outpatient hip or knee replacement.
“Let the surgeon know if you think outpatient joint replacement is a bad idea in your patient for medical reasons,” Dr. Bugbee urged.
Also, orthopedic surgeons can generally benefit from rheumatologist input regarding perioperative management of patients on standard disease-modifying antirheumatic drugs, biologics, or Janus kinase inhibitors as recommended in the guidelines published jointly by the American College of Rheumatology and the American Association of Hip and Knee Surgeons.
“I can guarantee you that most orthopedic surgeons don’t know about these guidelines. The evidence base for these recommendations is not great, but these are the best guidelines we have,” Dr. Bugbee said.
After joint replacement surgery a patient should get an x-ray of the replacement every 5 years. And if a patient develops a painful hip after arthroplasty, it’s worthwhile to order blood chromium and cobalt levels.
“The implant weight-bearing surface matters. You can’t necessarily tell on x-ray what’s a metal-on-metal hip and what’s metal-on-plastic or ceramic. You already send people for a lot of labs. If you see a patient with a painful total hip replacement, just add a cobalt and chromium. If they’re elevated, talk to the orthopedist,” he advised.
The road ahead
Hip and knee replacement is an $18 billion market today. And it’s a major growth industry: According to a recent projection, there will be 1 million total hip replacements and 4 million total knee replacements annually 10 years from now, figures four times greater than projected for 2030 in an earlier 2005 estimate. The rapid growth is coming from the expanding elderly population combined with a virtual epidemic of posttraumatic arthritis in young people – but decidedly not from patients with joint failure attributable to rheumatoid arthritis.
“Congratulations! You’ve eradicated rheumatoid arthritis from my practice,” Dr. Bugbee declared. “Most of the rheumatoid arthritis patients who come to me come because they have osteoarthritis in their joint, not because of their rheumatoid arthritis.”
He reported serving as a consultant to Orthalign, Insight Medical, and Arthrex, and receiving royalties from Smith and Nephew and Depuy.
MAUI, HAWAII – Outpatient total hip and knee replacement is “the latest craze” in orthopedic surgery, and it’s being driven by the might of Medicare, William Bugbee, MD, said at the 2020 Rheumatology Winter Clinical Symposium.
“In 2019, Medicare took total knee replacement off the inpatient-only list, meaning you could do it as an outpatient. And just in January 2020, they took total hips off that list. So I have to designate most of my hip and knee replacements as outpatients, even if I do it in the hospital and keep them for 1 night. And some of the private insurers have already gone to that, so they’ll deny coverage if I say I want a 1-day hospital stay, believe it or not,” according to Dr. Bugbee, chief of joint reconstruction in the department of orthopedics at the Scripps Clinic in La Jolla, Calif.
He provided a behind-the-scenes look at contemporary trends in joint replacement as well as tips on how rheumatologists can best help their patients get through the experience with excellent outcomes.
Joint replacement remains the best treatment for advanced arthritis of the hips and knees, he said. There is a high degree of confidence about the predictability and durability of the results. But joint replacement has become highly commoditized.
“We’re getting pummeled by Medicare to make this as cheap as possible,” the orthopedic surgeon explained. “An implant costs the hospital $3,000-$6,000. A care episode for a primary total joint replacement should cost a hospital $8,000-$15,000, which is about what Medicare pays for the [Diagnosis Related Group], so the margins are small. That’s why we’re being drilled on about how much we spend on every little thing. We hardly do any labs, x-rays, anything.”
As a result of recent advances in pre-, peri-, and postoperative management, outpatient joint replacement has become a safe and comparatively economical option for generally healthy patients.
“We’ve engineered a much better patient experience, so the assault and battery of 5, 10, 15 years ago isn’t so bad anymore,” Dr. Bugbee said.
Rheumatologists can expect to see a growing number of their patients undergoing total knee or hip replacement at outpatient surgery centers. That’s not a bad thing so long as the procedure is being done there because the outpatient center employs best practices in order to provide a highly efficient episode of care supported by excellent outcome data, he continued.
State-of-the-art perioperative management in 2020 includes accelerated-care pathways that allow ambulation within an hour or 2 after surgery along with same-day discharge, regional anesthesia with motor-sparing nerve blocks, and multimodal pain management with avoidance of intravenous narcotics except in opioid-tolerant patients. Tranexamic acid is now widely used in order to reduce operative blood loss.
“When I started practice 25 years ago, 50% of patients got a blood transfusion. I haven’t given a blood transfusion to a patient in probably 2 years. Tranexamic acid reduces blood loss by 500-700 cc with no discernible adverse effects. It’s truly remarkable,” he said.
Another important technical advance has been the routine use of oral dexamethasone. “Decadron is an antiemetic, it has anti-inflammatory effects, and it makes people happy. It’s a simple, cheap drug that has revolutionized care,” the surgeon continued.
Postoperative management has been streamlined. Dr. Bugbee is among many orthopedic surgeons who no longer routinely prescribe therapist-directed formal physical therapy for total hip arthroplasty patients, relying instead upon online tools and apps for self-administered physical therapy. Pedal exercise devices available online for $30 or so have been shown to be as effective as supervised physical therapy for knee rehabilitation.
What patients want to know about joint replacement
The question patients most often ask both their referring physician and the orthopedic surgeon is, “How long will my joint replacement last?” The best available data come from a couple of recent paired meta-analyses. The investigators reported 82% implant survivorship 25 years after primary total knee arthroplasty and 70% after unicondylar knee arthroplasty as well as a 25-year implant survivorship rate of 77% for total hip arthroplasty.
“I expected that hip arthroplasty survivorship rate to be much higher than 77%. The reason it’s not is probably because of the metal-on-metal bearing surface debacle of about 10 years ago. There’ve been lots of revisions because of that. We thought metal-on-metal implants were going to be all that, with microscopically low wear, but they turned out to be a nightmare because of metal ion release,” Dr. Bugbee observed.
The long-term joint survivorship data are based upon older implants. Encouraging albeit still preliminary data suggest contemporary implants may last significantly longer. The “clear winner,” he said, is a 36-mm ceramic head and a highly crosslinked polyethylene liner.
“That’s been a game changer, with a 10- to 20-fold decrease in wear compared to plastics for weight-bearing surfaces,” Dr. Bugbee said.
In terms of functional improvement, by various measures 85%-97% of patients are satisfied with the results of their total hip replacement, and 60% report returning to high-level recreational activities. Patient satisfaction scores are lower – 75%-90% – after total knee arthroplasty.
“The total knee replacement just doesn’t work like a regular joint,” the surgeon observed. “When I think of hip and knee replacements, I think of a hip as a Ferrari – it’s a high-performance joint replacement – and I think of the knee as a Ford – it’s serviceable, it does the job, and it’s okay but not fantastic.”
How referring physicians can optimize preoperative management and long-term follow-up
Orthopedic surgeons would appreciate help from rheumatologists and primary care physicians in preoperatively addressing the known modifiable risk factors for poor outcomes of joint replacement. These include obesity, smoking, depression, a hemoglobin A1c of 7% or more, and being on opioids. These risk factors are incompatible with outpatient hip or knee replacement.
“Let the surgeon know if you think outpatient joint replacement is a bad idea in your patient for medical reasons,” Dr. Bugbee urged.
Also, orthopedic surgeons can generally benefit from rheumatologist input regarding perioperative management of patients on standard disease-modifying antirheumatic drugs, biologics, or Janus kinase inhibitors as recommended in the guidelines published jointly by the American College of Rheumatology and the American Association of Hip and Knee Surgeons.
“I can guarantee you that most orthopedic surgeons don’t know about these guidelines. The evidence base for these recommendations is not great, but these are the best guidelines we have,” Dr. Bugbee said.
After joint replacement surgery a patient should get an x-ray of the replacement every 5 years. And if a patient develops a painful hip after arthroplasty, it’s worthwhile to order blood chromium and cobalt levels.
“The implant weight-bearing surface matters. You can’t necessarily tell on x-ray what’s a metal-on-metal hip and what’s metal-on-plastic or ceramic. You already send people for a lot of labs. If you see a patient with a painful total hip replacement, just add a cobalt and chromium. If they’re elevated, talk to the orthopedist,” he advised.
The road ahead
Hip and knee replacement is an $18 billion market today. And it’s a major growth industry: According to a recent projection, there will be 1 million total hip replacements and 4 million total knee replacements annually 10 years from now, figures four times greater than projected for 2030 in an earlier 2005 estimate. The rapid growth is coming from the expanding elderly population combined with a virtual epidemic of posttraumatic arthritis in young people – but decidedly not from patients with joint failure attributable to rheumatoid arthritis.
“Congratulations! You’ve eradicated rheumatoid arthritis from my practice,” Dr. Bugbee declared. “Most of the rheumatoid arthritis patients who come to me come because they have osteoarthritis in their joint, not because of their rheumatoid arthritis.”
He reported serving as a consultant to Orthalign, Insight Medical, and Arthrex, and receiving royalties from Smith and Nephew and Depuy.
REPORTING FROM RWCS 2020
Higher endovascular thrombectomy volumes yield better stroke outcomes
LOS ANGELES – Higher case volumes matter for getting better outcomes in acute ischemic stroke patients treated with endovascular thrombectomy, according to data from more than 13,000 Medicare patients treated during 2016 and 2017.
That’s hardly surprising, given that it’s consistent with what’s already been reported for several other types of endovascular and transcatheter procedures: The more cases a center or individual proceduralist performs, the better their patients do. Routine use of endovascular thrombectomy to treat selected acute ischemic stroke patients is a new-enough paradigm that until now few reports have come out that looked at this issue (Stroke. 2019 May;50[5]:1178-83).
The new analysis of Medicare data “is one of the first contemporary studies of the volume-outcome relationship in endovascular thrombectomy,” Laura K. Stein, MD, said at the International Stroke Conference sponsored by the American Heart Association. The analysis showed that, when the researchers adjusted the Medicare data to better reflect overall case volumes (Medicare patients represent just 59% of all endovascular thrombectomies performed on U.S. acute ischemic stroke patients), the minimum case number for a stroke center to have statistically better in-hospital survival than lower volume centers was 24 cases/year, and 29 cases/year to have a statistically significant higher rate of “good” outcomes than lower-volume centers, reported Dr. Stein, a stroke neurologist with the Mount Sinai Health System in New York. For individual proceduralists, the minimum, adjusted case number to have statistically better acute patient survival was 4 cases/year, and 19 cases/year to have a statistically better rate of good outcomes.
For this analysis, good outcomes were defined as cases when patients left the hospital following their acute care and returned home with either self care or a home health care service, and also patients discharged to rehabilitation. “Bad” outcomes for this analysis were discharges to a skilled nursing facility or hospice, as well as patients who died during their acute hospitalization.
The analyses also showed no plateau to the volume effect for any of the four parameters examined: in-hospital mortality by center and by proceduralist, and the rates of good outcomes by center and by proceduralist. For each of these measures, as case volume increased above the minimum number needed to produce statistically better outcomes, the rate of good outcomes continued to steadily rise and acute mortality continued to steadily fall.
The study run by Dr. Stein and associates used data collected by the Center for Medicare & Medicaid Services on 13,311 Medicare patients who underwent endovascular thrombectomy for acute ischemic stroke at any of 641 U.S. hospitals and received treatment from any of 2,754 thrombectomy proceduralists. Outcomes rated as good occurred in 56% of the patients. The statistical adjustments that the researchers applied to calculate the incremental effect of increasing case volume took into account the variables of patient age, sex, and comorbidities measured by the Charlson Comorbidity Index.
The analysis also showed that, during this 2-year period, the average number of endovascular thrombectomy cases among Medicare patients was just under 21 cases per center, with a range of 1-160 cases; for individual proceduralists, the average was just under 5 cases, with a range of 1-82 cases.
The 19 case/year volume minimum that the analysis identified for an individual proceduralist to have a statistically significant higher rate of good outcomes, compared with lower-volume proceduralists, came close to the 15 cases/year minimum set by the Joint Commission in 2019 for individual operators at centers seeking accreditation from the Joint Commission as either a Thrombectomy-Capable Stroke Center or a Comprehensive Stroke Center. The CMS has not yet set thrombectomy case-load requirements for centers or operators to qualify for Medicare reimbursements, although CMS has set such standards for other endovascular procedures, such as transcatheter aortic valve replacement. When setting such standards, CMS has cited its need to balance the better outcomes produced by higher-volume centers against a societal interest in facilitating access to vital medical services, a balance that Dr. Stein also highlighted in her talk.
“We want to optimize access as well as outcomes for every patient,” she said. “These data support certification volume standards,” but they are “in no way an argument for limiting access based on volume.”
Dr. Stein had no disclosures.
SOURCE: Stein LK et al. ISC 2020, Abstract LB11.
The results reported by Dr. Stein raise issues about balancing the access to certain therapies with the outcomes of those therapies. Having procedures like endovascular thrombectomy for acute ischemic stroke done primarily at high-volume centers might improve procedural outcomes, but having more centers offering this treatment across wider geographical areas would make this treatment more broadly available to more people.
For endovascular thrombectomy, center volume and experience may be much more important than proceduralist volume because having a smoothly functioning system in place is so important for rapid stroke assessment and treatment. It’s also important for programs to provide experienced and comprehensive postthrombectomy care. Success in endovascular thrombectomy involves much more than just taking a clot out. It means quickly and smoothly moving patients through the steps that precede thrombectomy and then following the intervention with a range of services that optimize recovery.
Ashutosh P. Jadhav, MD, PhD , is director of the comprehensive stroke center at the University of Pittsburgh. He had no relevant disclosures. He made these comments in an interview.
The results reported by Dr. Stein raise issues about balancing the access to certain therapies with the outcomes of those therapies. Having procedures like endovascular thrombectomy for acute ischemic stroke done primarily at high-volume centers might improve procedural outcomes, but having more centers offering this treatment across wider geographical areas would make this treatment more broadly available to more people.
For endovascular thrombectomy, center volume and experience may be much more important than proceduralist volume because having a smoothly functioning system in place is so important for rapid stroke assessment and treatment. It’s also important for programs to provide experienced and comprehensive postthrombectomy care. Success in endovascular thrombectomy involves much more than just taking a clot out. It means quickly and smoothly moving patients through the steps that precede thrombectomy and then following the intervention with a range of services that optimize recovery.
Ashutosh P. Jadhav, MD, PhD , is director of the comprehensive stroke center at the University of Pittsburgh. He had no relevant disclosures. He made these comments in an interview.
The results reported by Dr. Stein raise issues about balancing the access to certain therapies with the outcomes of those therapies. Having procedures like endovascular thrombectomy for acute ischemic stroke done primarily at high-volume centers might improve procedural outcomes, but having more centers offering this treatment across wider geographical areas would make this treatment more broadly available to more people.
For endovascular thrombectomy, center volume and experience may be much more important than proceduralist volume because having a smoothly functioning system in place is so important for rapid stroke assessment and treatment. It’s also important for programs to provide experienced and comprehensive postthrombectomy care. Success in endovascular thrombectomy involves much more than just taking a clot out. It means quickly and smoothly moving patients through the steps that precede thrombectomy and then following the intervention with a range of services that optimize recovery.
Ashutosh P. Jadhav, MD, PhD , is director of the comprehensive stroke center at the University of Pittsburgh. He had no relevant disclosures. He made these comments in an interview.
LOS ANGELES – Higher case volumes matter for getting better outcomes in acute ischemic stroke patients treated with endovascular thrombectomy, according to data from more than 13,000 Medicare patients treated during 2016 and 2017.
That’s hardly surprising, given that it’s consistent with what’s already been reported for several other types of endovascular and transcatheter procedures: The more cases a center or individual proceduralist performs, the better their patients do. Routine use of endovascular thrombectomy to treat selected acute ischemic stroke patients is a new-enough paradigm that until now few reports have come out that looked at this issue (Stroke. 2019 May;50[5]:1178-83).
The new analysis of Medicare data “is one of the first contemporary studies of the volume-outcome relationship in endovascular thrombectomy,” Laura K. Stein, MD, said at the International Stroke Conference sponsored by the American Heart Association. The analysis showed that, when the researchers adjusted the Medicare data to better reflect overall case volumes (Medicare patients represent just 59% of all endovascular thrombectomies performed on U.S. acute ischemic stroke patients), the minimum case number for a stroke center to have statistically better in-hospital survival than lower volume centers was 24 cases/year, and 29 cases/year to have a statistically significant higher rate of “good” outcomes than lower-volume centers, reported Dr. Stein, a stroke neurologist with the Mount Sinai Health System in New York. For individual proceduralists, the minimum, adjusted case number to have statistically better acute patient survival was 4 cases/year, and 19 cases/year to have a statistically better rate of good outcomes.
For this analysis, good outcomes were defined as cases when patients left the hospital following their acute care and returned home with either self care or a home health care service, and also patients discharged to rehabilitation. “Bad” outcomes for this analysis were discharges to a skilled nursing facility or hospice, as well as patients who died during their acute hospitalization.
The analyses also showed no plateau to the volume effect for any of the four parameters examined: in-hospital mortality by center and by proceduralist, and the rates of good outcomes by center and by proceduralist. For each of these measures, as case volume increased above the minimum number needed to produce statistically better outcomes, the rate of good outcomes continued to steadily rise and acute mortality continued to steadily fall.
The study run by Dr. Stein and associates used data collected by the Center for Medicare & Medicaid Services on 13,311 Medicare patients who underwent endovascular thrombectomy for acute ischemic stroke at any of 641 U.S. hospitals and received treatment from any of 2,754 thrombectomy proceduralists. Outcomes rated as good occurred in 56% of the patients. The statistical adjustments that the researchers applied to calculate the incremental effect of increasing case volume took into account the variables of patient age, sex, and comorbidities measured by the Charlson Comorbidity Index.
The analysis also showed that, during this 2-year period, the average number of endovascular thrombectomy cases among Medicare patients was just under 21 cases per center, with a range of 1-160 cases; for individual proceduralists, the average was just under 5 cases, with a range of 1-82 cases.
The 19 case/year volume minimum that the analysis identified for an individual proceduralist to have a statistically significant higher rate of good outcomes, compared with lower-volume proceduralists, came close to the 15 cases/year minimum set by the Joint Commission in 2019 for individual operators at centers seeking accreditation from the Joint Commission as either a Thrombectomy-Capable Stroke Center or a Comprehensive Stroke Center. The CMS has not yet set thrombectomy case-load requirements for centers or operators to qualify for Medicare reimbursements, although CMS has set such standards for other endovascular procedures, such as transcatheter aortic valve replacement. When setting such standards, CMS has cited its need to balance the better outcomes produced by higher-volume centers against a societal interest in facilitating access to vital medical services, a balance that Dr. Stein also highlighted in her talk.
“We want to optimize access as well as outcomes for every patient,” she said. “These data support certification volume standards,” but they are “in no way an argument for limiting access based on volume.”
Dr. Stein had no disclosures.
SOURCE: Stein LK et al. ISC 2020, Abstract LB11.
LOS ANGELES – Higher case volumes matter for getting better outcomes in acute ischemic stroke patients treated with endovascular thrombectomy, according to data from more than 13,000 Medicare patients treated during 2016 and 2017.
That’s hardly surprising, given that it’s consistent with what’s already been reported for several other types of endovascular and transcatheter procedures: The more cases a center or individual proceduralist performs, the better their patients do. Routine use of endovascular thrombectomy to treat selected acute ischemic stroke patients is a new-enough paradigm that until now few reports have come out that looked at this issue (Stroke. 2019 May;50[5]:1178-83).
The new analysis of Medicare data “is one of the first contemporary studies of the volume-outcome relationship in endovascular thrombectomy,” Laura K. Stein, MD, said at the International Stroke Conference sponsored by the American Heart Association. The analysis showed that, when the researchers adjusted the Medicare data to better reflect overall case volumes (Medicare patients represent just 59% of all endovascular thrombectomies performed on U.S. acute ischemic stroke patients), the minimum case number for a stroke center to have statistically better in-hospital survival than lower volume centers was 24 cases/year, and 29 cases/year to have a statistically significant higher rate of “good” outcomes than lower-volume centers, reported Dr. Stein, a stroke neurologist with the Mount Sinai Health System in New York. For individual proceduralists, the minimum, adjusted case number to have statistically better acute patient survival was 4 cases/year, and 19 cases/year to have a statistically better rate of good outcomes.
For this analysis, good outcomes were defined as cases when patients left the hospital following their acute care and returned home with either self care or a home health care service, and also patients discharged to rehabilitation. “Bad” outcomes for this analysis were discharges to a skilled nursing facility or hospice, as well as patients who died during their acute hospitalization.
The analyses also showed no plateau to the volume effect for any of the four parameters examined: in-hospital mortality by center and by proceduralist, and the rates of good outcomes by center and by proceduralist. For each of these measures, as case volume increased above the minimum number needed to produce statistically better outcomes, the rate of good outcomes continued to steadily rise and acute mortality continued to steadily fall.
The study run by Dr. Stein and associates used data collected by the Center for Medicare & Medicaid Services on 13,311 Medicare patients who underwent endovascular thrombectomy for acute ischemic stroke at any of 641 U.S. hospitals and received treatment from any of 2,754 thrombectomy proceduralists. Outcomes rated as good occurred in 56% of the patients. The statistical adjustments that the researchers applied to calculate the incremental effect of increasing case volume took into account the variables of patient age, sex, and comorbidities measured by the Charlson Comorbidity Index.
The analysis also showed that, during this 2-year period, the average number of endovascular thrombectomy cases among Medicare patients was just under 21 cases per center, with a range of 1-160 cases; for individual proceduralists, the average was just under 5 cases, with a range of 1-82 cases.
The 19 case/year volume minimum that the analysis identified for an individual proceduralist to have a statistically significant higher rate of good outcomes, compared with lower-volume proceduralists, came close to the 15 cases/year minimum set by the Joint Commission in 2019 for individual operators at centers seeking accreditation from the Joint Commission as either a Thrombectomy-Capable Stroke Center or a Comprehensive Stroke Center. The CMS has not yet set thrombectomy case-load requirements for centers or operators to qualify for Medicare reimbursements, although CMS has set such standards for other endovascular procedures, such as transcatheter aortic valve replacement. When setting such standards, CMS has cited its need to balance the better outcomes produced by higher-volume centers against a societal interest in facilitating access to vital medical services, a balance that Dr. Stein also highlighted in her talk.
“We want to optimize access as well as outcomes for every patient,” she said. “These data support certification volume standards,” but they are “in no way an argument for limiting access based on volume.”
Dr. Stein had no disclosures.
SOURCE: Stein LK et al. ISC 2020, Abstract LB11.
REPORTING FROM ISC 2020
Resident experience with hysterectomy is on the decline
The total number of hysterectomies performed during residency training has declined significantly since 2008, despite an increase in laparoscopic hysterectomies performed, according to a new analysis of data from graduating ob.gyn. residents that has implications for the structure of resident education.
The investigators abstracted case log data from the Accreditation Council for Graduate Medical Education (ACGME) database to assess trends in residents’ operative experience and found decreases in abdominal and vaginal cases but an increase in experience with laparoscopic hysterectomy.
(from 85 cases to 37), and the median number of vaginal hysterectomies decreased by 36% (from 31 to 20 cases).
Laparoscopic hysterectomy increased by 115% from a median of 20 procedures in 2008-2009 to 43 in 2017-2018. Even so, the median total number of hysterectomies per resident decreased by 6%, from 112 to 105 procedures during those two time periods. (Data on total hysterectomy and laparoscopic hysterectomy were not collected by ACGME until 2008.)
While the absolute decrease in the total number of hysterectomies is “relatively small,” the trend “raises questions about what the appropriate number of hysterectomies per graduating resident should be,” Gregory M. Gressel, MD, MSc, of the Montefiore Medical Center, New York, and coauthors wrote in Obstetrics & Gynecology.
“These data point,” they wrote, “to the necessity of maximizing surgical exposure in the face of a declining availability of procedures and the importance of reflecting on which (and how many) procedures an obstetrics and gynecology resident needs to complete before entering clinical practice.”
The training numbers parallel an increased use of laparoscopic hysterectomy in the United States and other countries, as well as a well-documented decline in the total number of hysterectomies performed in the United States, the latter of which is driven largely by the availability and increasing use of alternatives to the procedure (such as hormone therapy, endometrial ablation, and uterine artery embolization).
Hysterectomy still is a “core procedure of gynecologic surgery,” however, and is “at the heart of surgical training in obstetrics and gynecology,” as surgical techniques developed from learning hysterectomy “are applied broadly in the pelvis,” Saketh R. Guntupalli, MD, wrote in an accompanying editorial.
Dr. Guntupalli, of the University of Colorado at Aurora, Denver, was involved in a survey of fellowship program directors, published in 2015, that found only 20% of first-year fellows were able to independently perform a vaginal hysterectomy and 46% to independently perform an abdominal hysterectomy (Obstet Gynecol. 2015;126:559-68).
This and other research suggest that fellowship training is “used to address deficiencies in residency training rather than to develop new, specialized surgical skills,” he wrote. Given a dearth of fellowship positions in ob.gyn., “it is impossible to adequately use those avenues to train the number of competent surgeons necessary to address the surgical needs of women’s health in the United States.”
To address such concerns, some residency programs have instituted resident tracking to direct more hysterectomy cases toward those residents who plan to pursue surgical subspecialties. The Cleveland Clinic, Dr. Guntupalli noted, has tried the latter approach “with success.”
An increase in the number of accredited training programs and a decrease in the number of residents per program also might help to improve surgical exposure for residents, Dr. Gressel and associates wrote. Over the 16-year study period, the number of graduating residents increased significantly (by 12 per year) and the number of residency programs decreased significantly (0.52 fewer programs per year).
Additionally, Dr. Guntupalli wrote, regulatory bodies may need to reevaluate how competencies are assessed, and whether minimal numbers of cases “continue to carry the same weight as they did in previous generations.”
In the study, one coauthor is a full-time employee of ACGME, and another receives funds as a director for the American Board of Obstetrics and Gynecology. The remaining authors had no relevant financial disclosures. There was no outside funding for the study. Dr. Guntupalli said he had no conflicts of interest.
SOURCES: Gressel GM et al. Obstet Gynecol. 2020 Feb;135(2):268-73; Guntupalli SR. Obstet Gynecol 2020 Feb;135(2):266-7.
This excellent paper by Dr. Gressel and coauthors shows decreasing numbers of hysterectomies – especially open and vaginal approaches – being performed by ob.gyn. residents. Considering also the 2015 publication by Guntupalli et al. showing the low numbers of incoming fellows able to perform hysterectomy, as well as Dr. Guntupalli’s editorial on this new research, we all must question how our patients will be able to undergo safe and effective surgery in the future.
Furthermore, it would truly be disheartening and disconcerting for a young physician to choose a residency with the desire of a specific track, only to lose that choice to a coresident.
In his presidential address to the AAGL some years ago, Javier Magrina, MD, of the Mayo Clinic in Phoenix, discussed separating the “O from the G” (J Minim Invasive Gynecol. 2014;21[4]:501-3). Among his points: From 1979 to 2006, there was a 46% decrease in the number of gynecologic operations (2,852,000 vs. 1,309,000), a 54% increase in the number of American College of Obstetricians and Gynecologists’ fellows (21,364 vs. 51,123), and an 81% decrease in the number of gynecologic operations performed per ACOG fellow (132 vs. 25).
In 1980, he pointed out, the total number of hysterectomy procedures performed in the United States was 647,000. In 2007, this total was 517,000. The total number of ACOG fellows in 1980 was 22,516, compared with 52,385 in 2007. And the total number of hysterectomies performed per ACOG fellow was 28, compared with 9.8 hysterectomies per fellow in 2007.
Dr. Magrina’s data goes hand in hand with Dr. Gressel’s new study. The surgical experience of the gynecologic surgeon certainly is on the wane. The result of this lack of experience is noted by Dr. Guntupalli in his 2015 publication. To us, it is readily apparent that Dr. Magrina is right: The only true solution is to finally realize that we must separate the O from the G.
Charles E. Miller, MD, is director of minimally invasive gynecologic surgery, and director of the AAGL fellowship in minimally invasive gynecologic surgery, at Advocate Lutheran General Hospital, Park Ridge, Ill. Kirsten Sasaki, MD, is associate director of the AAGL fellowship in minimally invasive gynecologic surgery at Advocate Lutheran. They have no other conflicts of interest.
This excellent paper by Dr. Gressel and coauthors shows decreasing numbers of hysterectomies – especially open and vaginal approaches – being performed by ob.gyn. residents. Considering also the 2015 publication by Guntupalli et al. showing the low numbers of incoming fellows able to perform hysterectomy, as well as Dr. Guntupalli’s editorial on this new research, we all must question how our patients will be able to undergo safe and effective surgery in the future.
Furthermore, it would truly be disheartening and disconcerting for a young physician to choose a residency with the desire of a specific track, only to lose that choice to a coresident.
In his presidential address to the AAGL some years ago, Javier Magrina, MD, of the Mayo Clinic in Phoenix, discussed separating the “O from the G” (J Minim Invasive Gynecol. 2014;21[4]:501-3). Among his points: From 1979 to 2006, there was a 46% decrease in the number of gynecologic operations (2,852,000 vs. 1,309,000), a 54% increase in the number of American College of Obstetricians and Gynecologists’ fellows (21,364 vs. 51,123), and an 81% decrease in the number of gynecologic operations performed per ACOG fellow (132 vs. 25).
In 1980, he pointed out, the total number of hysterectomy procedures performed in the United States was 647,000. In 2007, this total was 517,000. The total number of ACOG fellows in 1980 was 22,516, compared with 52,385 in 2007. And the total number of hysterectomies performed per ACOG fellow was 28, compared with 9.8 hysterectomies per fellow in 2007.
Dr. Magrina’s data goes hand in hand with Dr. Gressel’s new study. The surgical experience of the gynecologic surgeon certainly is on the wane. The result of this lack of experience is noted by Dr. Guntupalli in his 2015 publication. To us, it is readily apparent that Dr. Magrina is right: The only true solution is to finally realize that we must separate the O from the G.
Charles E. Miller, MD, is director of minimally invasive gynecologic surgery, and director of the AAGL fellowship in minimally invasive gynecologic surgery, at Advocate Lutheran General Hospital, Park Ridge, Ill. Kirsten Sasaki, MD, is associate director of the AAGL fellowship in minimally invasive gynecologic surgery at Advocate Lutheran. They have no other conflicts of interest.
This excellent paper by Dr. Gressel and coauthors shows decreasing numbers of hysterectomies – especially open and vaginal approaches – being performed by ob.gyn. residents. Considering also the 2015 publication by Guntupalli et al. showing the low numbers of incoming fellows able to perform hysterectomy, as well as Dr. Guntupalli’s editorial on this new research, we all must question how our patients will be able to undergo safe and effective surgery in the future.
Furthermore, it would truly be disheartening and disconcerting for a young physician to choose a residency with the desire of a specific track, only to lose that choice to a coresident.
In his presidential address to the AAGL some years ago, Javier Magrina, MD, of the Mayo Clinic in Phoenix, discussed separating the “O from the G” (J Minim Invasive Gynecol. 2014;21[4]:501-3). Among his points: From 1979 to 2006, there was a 46% decrease in the number of gynecologic operations (2,852,000 vs. 1,309,000), a 54% increase in the number of American College of Obstetricians and Gynecologists’ fellows (21,364 vs. 51,123), and an 81% decrease in the number of gynecologic operations performed per ACOG fellow (132 vs. 25).
In 1980, he pointed out, the total number of hysterectomy procedures performed in the United States was 647,000. In 2007, this total was 517,000. The total number of ACOG fellows in 1980 was 22,516, compared with 52,385 in 2007. And the total number of hysterectomies performed per ACOG fellow was 28, compared with 9.8 hysterectomies per fellow in 2007.
Dr. Magrina’s data goes hand in hand with Dr. Gressel’s new study. The surgical experience of the gynecologic surgeon certainly is on the wane. The result of this lack of experience is noted by Dr. Guntupalli in his 2015 publication. To us, it is readily apparent that Dr. Magrina is right: The only true solution is to finally realize that we must separate the O from the G.
Charles E. Miller, MD, is director of minimally invasive gynecologic surgery, and director of the AAGL fellowship in minimally invasive gynecologic surgery, at Advocate Lutheran General Hospital, Park Ridge, Ill. Kirsten Sasaki, MD, is associate director of the AAGL fellowship in minimally invasive gynecologic surgery at Advocate Lutheran. They have no other conflicts of interest.
The total number of hysterectomies performed during residency training has declined significantly since 2008, despite an increase in laparoscopic hysterectomies performed, according to a new analysis of data from graduating ob.gyn. residents that has implications for the structure of resident education.
The investigators abstracted case log data from the Accreditation Council for Graduate Medical Education (ACGME) database to assess trends in residents’ operative experience and found decreases in abdominal and vaginal cases but an increase in experience with laparoscopic hysterectomy.
(from 85 cases to 37), and the median number of vaginal hysterectomies decreased by 36% (from 31 to 20 cases).
Laparoscopic hysterectomy increased by 115% from a median of 20 procedures in 2008-2009 to 43 in 2017-2018. Even so, the median total number of hysterectomies per resident decreased by 6%, from 112 to 105 procedures during those two time periods. (Data on total hysterectomy and laparoscopic hysterectomy were not collected by ACGME until 2008.)
While the absolute decrease in the total number of hysterectomies is “relatively small,” the trend “raises questions about what the appropriate number of hysterectomies per graduating resident should be,” Gregory M. Gressel, MD, MSc, of the Montefiore Medical Center, New York, and coauthors wrote in Obstetrics & Gynecology.
“These data point,” they wrote, “to the necessity of maximizing surgical exposure in the face of a declining availability of procedures and the importance of reflecting on which (and how many) procedures an obstetrics and gynecology resident needs to complete before entering clinical practice.”
The training numbers parallel an increased use of laparoscopic hysterectomy in the United States and other countries, as well as a well-documented decline in the total number of hysterectomies performed in the United States, the latter of which is driven largely by the availability and increasing use of alternatives to the procedure (such as hormone therapy, endometrial ablation, and uterine artery embolization).
Hysterectomy still is a “core procedure of gynecologic surgery,” however, and is “at the heart of surgical training in obstetrics and gynecology,” as surgical techniques developed from learning hysterectomy “are applied broadly in the pelvis,” Saketh R. Guntupalli, MD, wrote in an accompanying editorial.
Dr. Guntupalli, of the University of Colorado at Aurora, Denver, was involved in a survey of fellowship program directors, published in 2015, that found only 20% of first-year fellows were able to independently perform a vaginal hysterectomy and 46% to independently perform an abdominal hysterectomy (Obstet Gynecol. 2015;126:559-68).
This and other research suggest that fellowship training is “used to address deficiencies in residency training rather than to develop new, specialized surgical skills,” he wrote. Given a dearth of fellowship positions in ob.gyn., “it is impossible to adequately use those avenues to train the number of competent surgeons necessary to address the surgical needs of women’s health in the United States.”
To address such concerns, some residency programs have instituted resident tracking to direct more hysterectomy cases toward those residents who plan to pursue surgical subspecialties. The Cleveland Clinic, Dr. Guntupalli noted, has tried the latter approach “with success.”
An increase in the number of accredited training programs and a decrease in the number of residents per program also might help to improve surgical exposure for residents, Dr. Gressel and associates wrote. Over the 16-year study period, the number of graduating residents increased significantly (by 12 per year) and the number of residency programs decreased significantly (0.52 fewer programs per year).
Additionally, Dr. Guntupalli wrote, regulatory bodies may need to reevaluate how competencies are assessed, and whether minimal numbers of cases “continue to carry the same weight as they did in previous generations.”
In the study, one coauthor is a full-time employee of ACGME, and another receives funds as a director for the American Board of Obstetrics and Gynecology. The remaining authors had no relevant financial disclosures. There was no outside funding for the study. Dr. Guntupalli said he had no conflicts of interest.
SOURCES: Gressel GM et al. Obstet Gynecol. 2020 Feb;135(2):268-73; Guntupalli SR. Obstet Gynecol 2020 Feb;135(2):266-7.
The total number of hysterectomies performed during residency training has declined significantly since 2008, despite an increase in laparoscopic hysterectomies performed, according to a new analysis of data from graduating ob.gyn. residents that has implications for the structure of resident education.
The investigators abstracted case log data from the Accreditation Council for Graduate Medical Education (ACGME) database to assess trends in residents’ operative experience and found decreases in abdominal and vaginal cases but an increase in experience with laparoscopic hysterectomy.
(from 85 cases to 37), and the median number of vaginal hysterectomies decreased by 36% (from 31 to 20 cases).
Laparoscopic hysterectomy increased by 115% from a median of 20 procedures in 2008-2009 to 43 in 2017-2018. Even so, the median total number of hysterectomies per resident decreased by 6%, from 112 to 105 procedures during those two time periods. (Data on total hysterectomy and laparoscopic hysterectomy were not collected by ACGME until 2008.)
While the absolute decrease in the total number of hysterectomies is “relatively small,” the trend “raises questions about what the appropriate number of hysterectomies per graduating resident should be,” Gregory M. Gressel, MD, MSc, of the Montefiore Medical Center, New York, and coauthors wrote in Obstetrics & Gynecology.
“These data point,” they wrote, “to the necessity of maximizing surgical exposure in the face of a declining availability of procedures and the importance of reflecting on which (and how many) procedures an obstetrics and gynecology resident needs to complete before entering clinical practice.”
The training numbers parallel an increased use of laparoscopic hysterectomy in the United States and other countries, as well as a well-documented decline in the total number of hysterectomies performed in the United States, the latter of which is driven largely by the availability and increasing use of alternatives to the procedure (such as hormone therapy, endometrial ablation, and uterine artery embolization).
Hysterectomy still is a “core procedure of gynecologic surgery,” however, and is “at the heart of surgical training in obstetrics and gynecology,” as surgical techniques developed from learning hysterectomy “are applied broadly in the pelvis,” Saketh R. Guntupalli, MD, wrote in an accompanying editorial.
Dr. Guntupalli, of the University of Colorado at Aurora, Denver, was involved in a survey of fellowship program directors, published in 2015, that found only 20% of first-year fellows were able to independently perform a vaginal hysterectomy and 46% to independently perform an abdominal hysterectomy (Obstet Gynecol. 2015;126:559-68).
This and other research suggest that fellowship training is “used to address deficiencies in residency training rather than to develop new, specialized surgical skills,” he wrote. Given a dearth of fellowship positions in ob.gyn., “it is impossible to adequately use those avenues to train the number of competent surgeons necessary to address the surgical needs of women’s health in the United States.”
To address such concerns, some residency programs have instituted resident tracking to direct more hysterectomy cases toward those residents who plan to pursue surgical subspecialties. The Cleveland Clinic, Dr. Guntupalli noted, has tried the latter approach “with success.”
An increase in the number of accredited training programs and a decrease in the number of residents per program also might help to improve surgical exposure for residents, Dr. Gressel and associates wrote. Over the 16-year study period, the number of graduating residents increased significantly (by 12 per year) and the number of residency programs decreased significantly (0.52 fewer programs per year).
Additionally, Dr. Guntupalli wrote, regulatory bodies may need to reevaluate how competencies are assessed, and whether minimal numbers of cases “continue to carry the same weight as they did in previous generations.”
In the study, one coauthor is a full-time employee of ACGME, and another receives funds as a director for the American Board of Obstetrics and Gynecology. The remaining authors had no relevant financial disclosures. There was no outside funding for the study. Dr. Guntupalli said he had no conflicts of interest.
SOURCES: Gressel GM et al. Obstet Gynecol. 2020 Feb;135(2):268-73; Guntupalli SR. Obstet Gynecol 2020 Feb;135(2):266-7.
FROM OBSTETRICS & GYNECOLOGY
Risk factors found for respiratory AEs in children following OSA surgery
Underlying cardiac disease, airway anomalies, and younger age each independently boosted the risk of severe perioperative respiratory adverse events (PRAE) in children undergoing adenotonsillectomy to treat obstructive sleep apnea, in a review of 374 patients treated at a single Canadian tertiary-referral center.
In contrast, the analysis failed to show independent, significant effects from any assessed polysomnography or oximetry parameters on the rate of postoperative respiratory complications. The utility of preoperative polysomnography or oximetry for risk stratification is questionable for pediatric patients scheduled to adenotonsillectomy to treat obstructive sleep apnea, wrote Sherri L. Katz, MD, of the University of Ottawa, and associates in a recent report published in the Journal of Clinical Sleep Medicine, although they also added that making these assessments may be “unavoidable” because of their need for diagnosing obstructive sleep apnea and determining the need for surgery.
Despite this caveat, “overall our study results highlight the need to better define the complex interaction between comorbidities, age, nocturnal respiratory events, and gas exchange abnormalities in predicting risk for PRAE” after adenotonsillectomy, the researchers wrote. These findings “are consistent with existing clinical care guidelines,” and “cardiac and craniofacial conditions have been associated with risk of postoperative complications in other studies.”
The analysis used data collected from all children aged 0-18 years who underwent polysomnography assessment followed by adenotonsillectomy at one Canadian tertiary-referral center, Children’s Hospital of Eastern Ontario in Ottawa, during 2010-2016. Their median age was just over 6 years, and 39 patients (10%) were younger than 3 years at the time of their surgery. More than three-quarters of the patients, 286, had at least one identified comorbidity, and nearly half had at least two comorbidities. Polysomnography identified sleep-disordered breathing in 344 of the children (92%), and diagnosed obstructive sleep apnea in 256 (68%), including 148 (43% of the full cohort) with a severe apnea-hypopnea index.
Sixty-six of the children (18%) had at least one severe PRAE that required intervention. Specifically these were either oxygen desaturations requiring intervention or need for airway or ventilatory support with interventions such as jaw thrust, oral or nasal airway placement, bag and mask ventilation, or endotracheal intubation.
A multivariate regression analysis of the measured comorbidity, polysomnography, and oximetry parameters, as well as age, identified three factors that independently linked with a statistically significant increase in the rate of severe PRAE: airway anomaly, underlying cardiac disease, and young age. Patients with an airway anomaly had a 219% increased rate of PRAE, compared with those with no anomaly; patients with underlying cardiac disease had a 109% increased rate, compared with those without cardiac disease; and patients aged younger than 3 years had a 310% higher rate of PRAE, compared with the children aged 6 years or older, while children aged 3-5 years had a 121% higher rate of PRAE, compared with older children.
The study received no commercial funding. Dr. Katz has received honoraria for speaking from Biogen that had no relevance to the study.
SOURCE: Katz SL et al. J Clin Sleep Med. 2020 Jan 15;16(1):41-8.
This well-conducted, retrospective, chart-review study adds important information to the published literature about risk stratification for children in a tertiary-referral population undergoing adenotonsillectomy. Their findings indicate that younger children remain at higher risk as well as those children with complex comorbid medical disease. They also show that children with severe sleep apnea or significant oxyhemoglobin desaturation are likewise at higher risk of postoperative respiratory compromise – emphasizing the need for preoperative polysomnography – particularly in a tertiary setting where many patients have medical comorbidities.
Despite the strengths of this study in assessing perioperative risk for respiratory compromise in a referral population with highly prevalent medical comorbidities, this study does not provide significant insight into the management of otherwise healthy children in a community setting who are undergoing adenotonsillectomy. This is important because a large number of adenotonsillectomies are performed outside of a tertiary-referral center and many of these children may not have undergone preoperative polysomnography to stratify risk. The utility of preoperative polysomnography in the evaluation of all children undergoing adenotonsillectomy remains controversial, with diverging recommendations from two major U.S. medical groups.
This study does not address the utility of polysomnography in community-based populations of otherwise healthy children. It is imperative to accurately ascertain risk so perioperative planning can ensure the safety of children at higher risk following adenotonsillectomy; however, there remains a paucity of studies assessing the cost-effectiveness as well as the positive and negative predictive value of polysomnographic findings. This study highlights the need for community-based studies of otherwise healthy children undergoing adenotonsillectomy to ensure that children at risk receive appropriate monitoring in an inpatient setting whereas those at lesser risk are not unnecessarily hospitalized postoperatively.
Heidi V. Connolly, MD, and Laura E. Tomaselli, MD, are pediatric sleep medicine physicians, and Margo K. McKenna Benoit, MD, is an otolaryngologist at the University of Rochester (N.Y.). They made these comments in a commentary that accompanied the published report ( J Clin Sleep Med. 2020 Jan 15;16[1]:3-4 ). They had no disclosures.
This well-conducted, retrospective, chart-review study adds important information to the published literature about risk stratification for children in a tertiary-referral population undergoing adenotonsillectomy. Their findings indicate that younger children remain at higher risk as well as those children with complex comorbid medical disease. They also show that children with severe sleep apnea or significant oxyhemoglobin desaturation are likewise at higher risk of postoperative respiratory compromise – emphasizing the need for preoperative polysomnography – particularly in a tertiary setting where many patients have medical comorbidities.
Despite the strengths of this study in assessing perioperative risk for respiratory compromise in a referral population with highly prevalent medical comorbidities, this study does not provide significant insight into the management of otherwise healthy children in a community setting who are undergoing adenotonsillectomy. This is important because a large number of adenotonsillectomies are performed outside of a tertiary-referral center and many of these children may not have undergone preoperative polysomnography to stratify risk. The utility of preoperative polysomnography in the evaluation of all children undergoing adenotonsillectomy remains controversial, with diverging recommendations from two major U.S. medical groups.
This study does not address the utility of polysomnography in community-based populations of otherwise healthy children. It is imperative to accurately ascertain risk so perioperative planning can ensure the safety of children at higher risk following adenotonsillectomy; however, there remains a paucity of studies assessing the cost-effectiveness as well as the positive and negative predictive value of polysomnographic findings. This study highlights the need for community-based studies of otherwise healthy children undergoing adenotonsillectomy to ensure that children at risk receive appropriate monitoring in an inpatient setting whereas those at lesser risk are not unnecessarily hospitalized postoperatively.
Heidi V. Connolly, MD, and Laura E. Tomaselli, MD, are pediatric sleep medicine physicians, and Margo K. McKenna Benoit, MD, is an otolaryngologist at the University of Rochester (N.Y.). They made these comments in a commentary that accompanied the published report ( J Clin Sleep Med. 2020 Jan 15;16[1]:3-4 ). They had no disclosures.
This well-conducted, retrospective, chart-review study adds important information to the published literature about risk stratification for children in a tertiary-referral population undergoing adenotonsillectomy. Their findings indicate that younger children remain at higher risk as well as those children with complex comorbid medical disease. They also show that children with severe sleep apnea or significant oxyhemoglobin desaturation are likewise at higher risk of postoperative respiratory compromise – emphasizing the need for preoperative polysomnography – particularly in a tertiary setting where many patients have medical comorbidities.
Despite the strengths of this study in assessing perioperative risk for respiratory compromise in a referral population with highly prevalent medical comorbidities, this study does not provide significant insight into the management of otherwise healthy children in a community setting who are undergoing adenotonsillectomy. This is important because a large number of adenotonsillectomies are performed outside of a tertiary-referral center and many of these children may not have undergone preoperative polysomnography to stratify risk. The utility of preoperative polysomnography in the evaluation of all children undergoing adenotonsillectomy remains controversial, with diverging recommendations from two major U.S. medical groups.
This study does not address the utility of polysomnography in community-based populations of otherwise healthy children. It is imperative to accurately ascertain risk so perioperative planning can ensure the safety of children at higher risk following adenotonsillectomy; however, there remains a paucity of studies assessing the cost-effectiveness as well as the positive and negative predictive value of polysomnographic findings. This study highlights the need for community-based studies of otherwise healthy children undergoing adenotonsillectomy to ensure that children at risk receive appropriate monitoring in an inpatient setting whereas those at lesser risk are not unnecessarily hospitalized postoperatively.
Heidi V. Connolly, MD, and Laura E. Tomaselli, MD, are pediatric sleep medicine physicians, and Margo K. McKenna Benoit, MD, is an otolaryngologist at the University of Rochester (N.Y.). They made these comments in a commentary that accompanied the published report ( J Clin Sleep Med. 2020 Jan 15;16[1]:3-4 ). They had no disclosures.
Underlying cardiac disease, airway anomalies, and younger age each independently boosted the risk of severe perioperative respiratory adverse events (PRAE) in children undergoing adenotonsillectomy to treat obstructive sleep apnea, in a review of 374 patients treated at a single Canadian tertiary-referral center.
In contrast, the analysis failed to show independent, significant effects from any assessed polysomnography or oximetry parameters on the rate of postoperative respiratory complications. The utility of preoperative polysomnography or oximetry for risk stratification is questionable for pediatric patients scheduled to adenotonsillectomy to treat obstructive sleep apnea, wrote Sherri L. Katz, MD, of the University of Ottawa, and associates in a recent report published in the Journal of Clinical Sleep Medicine, although they also added that making these assessments may be “unavoidable” because of their need for diagnosing obstructive sleep apnea and determining the need for surgery.
Despite this caveat, “overall our study results highlight the need to better define the complex interaction between comorbidities, age, nocturnal respiratory events, and gas exchange abnormalities in predicting risk for PRAE” after adenotonsillectomy, the researchers wrote. These findings “are consistent with existing clinical care guidelines,” and “cardiac and craniofacial conditions have been associated with risk of postoperative complications in other studies.”
The analysis used data collected from all children aged 0-18 years who underwent polysomnography assessment followed by adenotonsillectomy at one Canadian tertiary-referral center, Children’s Hospital of Eastern Ontario in Ottawa, during 2010-2016. Their median age was just over 6 years, and 39 patients (10%) were younger than 3 years at the time of their surgery. More than three-quarters of the patients, 286, had at least one identified comorbidity, and nearly half had at least two comorbidities. Polysomnography identified sleep-disordered breathing in 344 of the children (92%), and diagnosed obstructive sleep apnea in 256 (68%), including 148 (43% of the full cohort) with a severe apnea-hypopnea index.
Sixty-six of the children (18%) had at least one severe PRAE that required intervention. Specifically these were either oxygen desaturations requiring intervention or need for airway or ventilatory support with interventions such as jaw thrust, oral or nasal airway placement, bag and mask ventilation, or endotracheal intubation.
A multivariate regression analysis of the measured comorbidity, polysomnography, and oximetry parameters, as well as age, identified three factors that independently linked with a statistically significant increase in the rate of severe PRAE: airway anomaly, underlying cardiac disease, and young age. Patients with an airway anomaly had a 219% increased rate of PRAE, compared with those with no anomaly; patients with underlying cardiac disease had a 109% increased rate, compared with those without cardiac disease; and patients aged younger than 3 years had a 310% higher rate of PRAE, compared with the children aged 6 years or older, while children aged 3-5 years had a 121% higher rate of PRAE, compared with older children.
The study received no commercial funding. Dr. Katz has received honoraria for speaking from Biogen that had no relevance to the study.
SOURCE: Katz SL et al. J Clin Sleep Med. 2020 Jan 15;16(1):41-8.
Underlying cardiac disease, airway anomalies, and younger age each independently boosted the risk of severe perioperative respiratory adverse events (PRAE) in children undergoing adenotonsillectomy to treat obstructive sleep apnea, in a review of 374 patients treated at a single Canadian tertiary-referral center.
In contrast, the analysis failed to show independent, significant effects from any assessed polysomnography or oximetry parameters on the rate of postoperative respiratory complications. The utility of preoperative polysomnography or oximetry for risk stratification is questionable for pediatric patients scheduled to adenotonsillectomy to treat obstructive sleep apnea, wrote Sherri L. Katz, MD, of the University of Ottawa, and associates in a recent report published in the Journal of Clinical Sleep Medicine, although they also added that making these assessments may be “unavoidable” because of their need for diagnosing obstructive sleep apnea and determining the need for surgery.
Despite this caveat, “overall our study results highlight the need to better define the complex interaction between comorbidities, age, nocturnal respiratory events, and gas exchange abnormalities in predicting risk for PRAE” after adenotonsillectomy, the researchers wrote. These findings “are consistent with existing clinical care guidelines,” and “cardiac and craniofacial conditions have been associated with risk of postoperative complications in other studies.”
The analysis used data collected from all children aged 0-18 years who underwent polysomnography assessment followed by adenotonsillectomy at one Canadian tertiary-referral center, Children’s Hospital of Eastern Ontario in Ottawa, during 2010-2016. Their median age was just over 6 years, and 39 patients (10%) were younger than 3 years at the time of their surgery. More than three-quarters of the patients, 286, had at least one identified comorbidity, and nearly half had at least two comorbidities. Polysomnography identified sleep-disordered breathing in 344 of the children (92%), and diagnosed obstructive sleep apnea in 256 (68%), including 148 (43% of the full cohort) with a severe apnea-hypopnea index.
Sixty-six of the children (18%) had at least one severe PRAE that required intervention. Specifically these were either oxygen desaturations requiring intervention or need for airway or ventilatory support with interventions such as jaw thrust, oral or nasal airway placement, bag and mask ventilation, or endotracheal intubation.
A multivariate regression analysis of the measured comorbidity, polysomnography, and oximetry parameters, as well as age, identified three factors that independently linked with a statistically significant increase in the rate of severe PRAE: airway anomaly, underlying cardiac disease, and young age. Patients with an airway anomaly had a 219% increased rate of PRAE, compared with those with no anomaly; patients with underlying cardiac disease had a 109% increased rate, compared with those without cardiac disease; and patients aged younger than 3 years had a 310% higher rate of PRAE, compared with the children aged 6 years or older, while children aged 3-5 years had a 121% higher rate of PRAE, compared with older children.
The study received no commercial funding. Dr. Katz has received honoraria for speaking from Biogen that had no relevance to the study.
SOURCE: Katz SL et al. J Clin Sleep Med. 2020 Jan 15;16(1):41-8.
FROM THE JOURNAL OF CLINICAL SLEEP MEDICINE
‘A glimmer of hope’ for stroke/mortality benefit with AFib catheter ablation
SNOWMASS, COLO. – stroke, major bleeding, or cardiac arrest, compared with rhythm and/or rate control drugs in a propensity score–weighted, retrospective, observational study.
Findings of the investigation, which included more than 183,000 real-world patients in routine clinical practice, were reported by Peter S. Noseworthy, MD, during the annual Cardiovascular Conference at Snowmass sponsored by the American College of Cardiology.
The results breathe new life into the controversy created by the previously reported CABANA trial (Catheter Ablation vs. Antiarrhythmic Drug Therapy for Atrial Fibrillation), a 10-country study in which 2,204 patients with atrial fibrillation (AFib) were randomized to catheter ablation or antiarrhythmic and/or rhythm control medications and followed for a mean of about 4 years. CABANA yielded a negative result (JAMA. 2019 Apr 2;321[13]:1261-74), with the prespecified intent-to-treat analysis indicating no significant between-group difference in the primary composite endpoint – the very same one that was positive in the large observational study.
However, CABANA was marred by major problems arising from protocol deviations: Nearly 28% of patients assigned to medical therapy crossed over to catheter ablation, typically because their antiarrhythmic drugs failed, and 10% of patients randomized to catheter ablation never got it. This muddies the waters when trying to identify a true stroke/mortality benefit for catheter ablation, if indeed any such benefit was actually present.
Here’s where the controversy arose: While CABANA must be called a negative trial based upon the disappointing results of the intent-to-treat analysis, a prespecified post hoc analysis of patients as actually treated showed a statistically significant 27% relative risk reduction for the primary composite endpoint in the catheter ablation group. That’s strikingly similar to the 30% relative risk reduction for catheter ablation seen in the huge observational study, where the CABANA-type primary outcome occurred in 22.5% of the medically managed patients and 16.8% of those who underwent catheter ablation, noted Dr. Noseworthy, professor of medicine and director of heart rhythm and physiology at the Mayo Clinic in Rochester, Minn.
He ought to know: He was both an investigator in CABANA and first author of the published observational study (Eur Heart J. 2019 Apr 21;40[16]:1257-64).
In the observational study, Dr. Noseworthy and coinvestigators utilized a huge U.S. administrative health claims database in order to identify a nationally representative group of 183,760 AFib patients, 12,032 of whom were treated with catheter ablation and the rest with antiarrhythmic and/or rhythm control drugs during the same years the CABANA trial was enrolling patients. The two groups were balanced using propensity score weighting to adjust for baseline differences in 90 variables.
The investigators sought to learn if the CABANA study population was representative of real-world AFib patients, and whether the observational experience could help resolve the CABANA controversy. It turned out that most AFib patients seen in daily clinical practice were CABANA like; that is, 74% of them would have been eligible for the clinical trial because they were symptomatic, over age 65, or younger than 65 with at least one CHADS2 stroke risk factor. About 22% of the large real-world sample would have been excluded from CABANA because they’d failed on amiodarone and other antiarrhythmic agents or had previously undergone ablation. About 4% of patients failed to meet the CABANA inclusion criteria.
The risk reduction for the composite endpoint associated with catheter ablation in the large retrospective study was greatest in the CABANA-like patients, at 30%. It was less robust but still statistically significant at 15% in patients who met at least one of the exclusion criteria for the trial.
The sheer size of this study provides greater statistical power than in CABANA. Of course, a nonrandomized, propensity score–based comparison such as this is always susceptible to confounding, even after adjustment for 90 variables. But the observational study does offer “a glimmer of hope” that catheter ablation, done in the right patients, might confer a stroke risk reduction and mortality benefit, he said.
The 33% relative risk reduction in the small group of real-world patients who failed to meet the CABANA inclusion criteria, while numerically impressive, wasn’t close to statistical significance, probably because event rates in that population were so low.
“Even if you could reduce stroke risk with ablation in that low-risk group, it would be a very inefficient way to reduce the population burden of stroke,” Dr. Noseworthy observed.
Putting together the results of CABANA and the large observational study to sum up his view of where catheter ablation for AF[ib] stands today, Dr. Noseworthy commented, “Ablation is reasonable for symptom control in many patients, basically anyone who is either breaking through on drugs or doesn’t want to take the drugs and is highly symptomatic. And there may be a small stroke and/or mortality benefit for people who are in the sweet spot – and those are people who look a lot like the patients enrolled in CABANA.”
Patients who met the exclusion criteria for CABANA are too advanced in their AFib to be likely to derive a stroke or mortality benefit from catheter ablation. “It’s very hard to move the needle in these patients with either a drug or catheter ablation approach. I wouldn’t try to reduce the risk of stroke here with an expensive and invasive procedure,” the electrophysiologist concluded.
He reported having no financial conflicts regarding his presentation.
SNOWMASS, COLO. – stroke, major bleeding, or cardiac arrest, compared with rhythm and/or rate control drugs in a propensity score–weighted, retrospective, observational study.
Findings of the investigation, which included more than 183,000 real-world patients in routine clinical practice, were reported by Peter S. Noseworthy, MD, during the annual Cardiovascular Conference at Snowmass sponsored by the American College of Cardiology.
The results breathe new life into the controversy created by the previously reported CABANA trial (Catheter Ablation vs. Antiarrhythmic Drug Therapy for Atrial Fibrillation), a 10-country study in which 2,204 patients with atrial fibrillation (AFib) were randomized to catheter ablation or antiarrhythmic and/or rhythm control medications and followed for a mean of about 4 years. CABANA yielded a negative result (JAMA. 2019 Apr 2;321[13]:1261-74), with the prespecified intent-to-treat analysis indicating no significant between-group difference in the primary composite endpoint – the very same one that was positive in the large observational study.
However, CABANA was marred by major problems arising from protocol deviations: Nearly 28% of patients assigned to medical therapy crossed over to catheter ablation, typically because their antiarrhythmic drugs failed, and 10% of patients randomized to catheter ablation never got it. This muddies the waters when trying to identify a true stroke/mortality benefit for catheter ablation, if indeed any such benefit was actually present.
Here’s where the controversy arose: While CABANA must be called a negative trial based upon the disappointing results of the intent-to-treat analysis, a prespecified post hoc analysis of patients as actually treated showed a statistically significant 27% relative risk reduction for the primary composite endpoint in the catheter ablation group. That’s strikingly similar to the 30% relative risk reduction for catheter ablation seen in the huge observational study, where the CABANA-type primary outcome occurred in 22.5% of the medically managed patients and 16.8% of those who underwent catheter ablation, noted Dr. Noseworthy, professor of medicine and director of heart rhythm and physiology at the Mayo Clinic in Rochester, Minn.
He ought to know: He was both an investigator in CABANA and first author of the published observational study (Eur Heart J. 2019 Apr 21;40[16]:1257-64).
In the observational study, Dr. Noseworthy and coinvestigators utilized a huge U.S. administrative health claims database in order to identify a nationally representative group of 183,760 AFib patients, 12,032 of whom were treated with catheter ablation and the rest with antiarrhythmic and/or rhythm control drugs during the same years the CABANA trial was enrolling patients. The two groups were balanced using propensity score weighting to adjust for baseline differences in 90 variables.
The investigators sought to learn if the CABANA study population was representative of real-world AFib patients, and whether the observational experience could help resolve the CABANA controversy. It turned out that most AFib patients seen in daily clinical practice were CABANA like; that is, 74% of them would have been eligible for the clinical trial because they were symptomatic, over age 65, or younger than 65 with at least one CHADS2 stroke risk factor. About 22% of the large real-world sample would have been excluded from CABANA because they’d failed on amiodarone and other antiarrhythmic agents or had previously undergone ablation. About 4% of patients failed to meet the CABANA inclusion criteria.
The risk reduction for the composite endpoint associated with catheter ablation in the large retrospective study was greatest in the CABANA-like patients, at 30%. It was less robust but still statistically significant at 15% in patients who met at least one of the exclusion criteria for the trial.
The sheer size of this study provides greater statistical power than in CABANA. Of course, a nonrandomized, propensity score–based comparison such as this is always susceptible to confounding, even after adjustment for 90 variables. But the observational study does offer “a glimmer of hope” that catheter ablation, done in the right patients, might confer a stroke risk reduction and mortality benefit, he said.
The 33% relative risk reduction in the small group of real-world patients who failed to meet the CABANA inclusion criteria, while numerically impressive, wasn’t close to statistical significance, probably because event rates in that population were so low.
“Even if you could reduce stroke risk with ablation in that low-risk group, it would be a very inefficient way to reduce the population burden of stroke,” Dr. Noseworthy observed.
Putting together the results of CABANA and the large observational study to sum up his view of where catheter ablation for AF[ib] stands today, Dr. Noseworthy commented, “Ablation is reasonable for symptom control in many patients, basically anyone who is either breaking through on drugs or doesn’t want to take the drugs and is highly symptomatic. And there may be a small stroke and/or mortality benefit for people who are in the sweet spot – and those are people who look a lot like the patients enrolled in CABANA.”
Patients who met the exclusion criteria for CABANA are too advanced in their AFib to be likely to derive a stroke or mortality benefit from catheter ablation. “It’s very hard to move the needle in these patients with either a drug or catheter ablation approach. I wouldn’t try to reduce the risk of stroke here with an expensive and invasive procedure,” the electrophysiologist concluded.
He reported having no financial conflicts regarding his presentation.
SNOWMASS, COLO. – stroke, major bleeding, or cardiac arrest, compared with rhythm and/or rate control drugs in a propensity score–weighted, retrospective, observational study.
Findings of the investigation, which included more than 183,000 real-world patients in routine clinical practice, were reported by Peter S. Noseworthy, MD, during the annual Cardiovascular Conference at Snowmass sponsored by the American College of Cardiology.
The results breathe new life into the controversy created by the previously reported CABANA trial (Catheter Ablation vs. Antiarrhythmic Drug Therapy for Atrial Fibrillation), a 10-country study in which 2,204 patients with atrial fibrillation (AFib) were randomized to catheter ablation or antiarrhythmic and/or rhythm control medications and followed for a mean of about 4 years. CABANA yielded a negative result (JAMA. 2019 Apr 2;321[13]:1261-74), with the prespecified intent-to-treat analysis indicating no significant between-group difference in the primary composite endpoint – the very same one that was positive in the large observational study.
However, CABANA was marred by major problems arising from protocol deviations: Nearly 28% of patients assigned to medical therapy crossed over to catheter ablation, typically because their antiarrhythmic drugs failed, and 10% of patients randomized to catheter ablation never got it. This muddies the waters when trying to identify a true stroke/mortality benefit for catheter ablation, if indeed any such benefit was actually present.
Here’s where the controversy arose: While CABANA must be called a negative trial based upon the disappointing results of the intent-to-treat analysis, a prespecified post hoc analysis of patients as actually treated showed a statistically significant 27% relative risk reduction for the primary composite endpoint in the catheter ablation group. That’s strikingly similar to the 30% relative risk reduction for catheter ablation seen in the huge observational study, where the CABANA-type primary outcome occurred in 22.5% of the medically managed patients and 16.8% of those who underwent catheter ablation, noted Dr. Noseworthy, professor of medicine and director of heart rhythm and physiology at the Mayo Clinic in Rochester, Minn.
He ought to know: He was both an investigator in CABANA and first author of the published observational study (Eur Heart J. 2019 Apr 21;40[16]:1257-64).
In the observational study, Dr. Noseworthy and coinvestigators utilized a huge U.S. administrative health claims database in order to identify a nationally representative group of 183,760 AFib patients, 12,032 of whom were treated with catheter ablation and the rest with antiarrhythmic and/or rhythm control drugs during the same years the CABANA trial was enrolling patients. The two groups were balanced using propensity score weighting to adjust for baseline differences in 90 variables.
The investigators sought to learn if the CABANA study population was representative of real-world AFib patients, and whether the observational experience could help resolve the CABANA controversy. It turned out that most AFib patients seen in daily clinical practice were CABANA like; that is, 74% of them would have been eligible for the clinical trial because they were symptomatic, over age 65, or younger than 65 with at least one CHADS2 stroke risk factor. About 22% of the large real-world sample would have been excluded from CABANA because they’d failed on amiodarone and other antiarrhythmic agents or had previously undergone ablation. About 4% of patients failed to meet the CABANA inclusion criteria.
The risk reduction for the composite endpoint associated with catheter ablation in the large retrospective study was greatest in the CABANA-like patients, at 30%. It was less robust but still statistically significant at 15% in patients who met at least one of the exclusion criteria for the trial.
The sheer size of this study provides greater statistical power than in CABANA. Of course, a nonrandomized, propensity score–based comparison such as this is always susceptible to confounding, even after adjustment for 90 variables. But the observational study does offer “a glimmer of hope” that catheter ablation, done in the right patients, might confer a stroke risk reduction and mortality benefit, he said.
The 33% relative risk reduction in the small group of real-world patients who failed to meet the CABANA inclusion criteria, while numerically impressive, wasn’t close to statistical significance, probably because event rates in that population were so low.
“Even if you could reduce stroke risk with ablation in that low-risk group, it would be a very inefficient way to reduce the population burden of stroke,” Dr. Noseworthy observed.
Putting together the results of CABANA and the large observational study to sum up his view of where catheter ablation for AF[ib] stands today, Dr. Noseworthy commented, “Ablation is reasonable for symptom control in many patients, basically anyone who is either breaking through on drugs or doesn’t want to take the drugs and is highly symptomatic. And there may be a small stroke and/or mortality benefit for people who are in the sweet spot – and those are people who look a lot like the patients enrolled in CABANA.”
Patients who met the exclusion criteria for CABANA are too advanced in their AFib to be likely to derive a stroke or mortality benefit from catheter ablation. “It’s very hard to move the needle in these patients with either a drug or catheter ablation approach. I wouldn’t try to reduce the risk of stroke here with an expensive and invasive procedure,” the electrophysiologist concluded.
He reported having no financial conflicts regarding his presentation.
REPORTING FROM ACC SNOWMASS 2020