User login
Official Newspaper of the American College of Surgeons
Bariatric Surgery Safety Has Increased With Medicare Coverage
CHICAGO – Complication rates for bariatric surgery patients on Medicare declined after Medicare began to cover the procedure in 2006, an analysis of state inpatient data from 12 states has found.
Data from previous studies suggest that bariatric surgery outcomes for Medicare patients improved after the implementation of Medicare’s National Coverage Determination in 2006, said Dr. Justin B. Dimick of the University of Michigan, Ann Arbor. But specific safety data on Medicare patients who have undergone bariatric surgery since the time of the decision are limited, Dr. Dimick said. The decision allowed for Medicare coverage of patients who sought care at facilities certified by the American College of Surgeons or the American Society for Metabolic and Bariatric Surgery.
To assess the impact of the National Coverage Determination on the safety of bariatric surgery, Dr. Dimick and his colleagues reviewed state inpatient data from 12 large, geographically dispersed states. Patients were identified on the basis of ICD-9 codes and diagnosis-related groups for weight loss surgery.
The outcomes were categorized according to any complication, a serious complication, or a reoperation.
The percentage of Medicare patients with any complications dropped from 12% before the determination to 8% afterward. Similarly, the percentage of non-Medicare patients with any complications dropped from 7% before to 5% after the determination.
In a multivariate analysis, the factors contributing to improved outcomes for bariatric surgery in Medicare patients were an increase in the use of laparoscopic gastric banding (lap band surgery), the transfer of patients to better hospitals, and quality improvement within individual hospitals, Dr. Dimick said.
Improvements in the safety of bariatric surgery did not, however, result from patients being redirected to safer hospitals, said Dr. Dimick. In fact, complication rates for procedures performed at Centers of Excellence versus non–Centers of Excellence were not significantly different (odds ratio for any complications, 0.97), he noted.
"CMS should consider dropping the COE [Centers of Excellence] aspect of the coverage decision [that] limits patient access without a beneficial improvement in outcomes," Dr. Dimick said. "Alternatively, CMS could revise the national coverage decision to further encourage participation in a quality improvement registry." Such a registry would need to include measures of long-term effectiveness to identify any unintended consequences of the increase in lap band surgery, he added.
Dr. Dimick is an equity owner and cofounder of ArborMetrix, a health care analytics and software firm.
CHICAGO – Complication rates for bariatric surgery patients on Medicare declined after Medicare began to cover the procedure in 2006, an analysis of state inpatient data from 12 states has found.
Data from previous studies suggest that bariatric surgery outcomes for Medicare patients improved after the implementation of Medicare’s National Coverage Determination in 2006, said Dr. Justin B. Dimick of the University of Michigan, Ann Arbor. But specific safety data on Medicare patients who have undergone bariatric surgery since the time of the decision are limited, Dr. Dimick said. The decision allowed for Medicare coverage of patients who sought care at facilities certified by the American College of Surgeons or the American Society for Metabolic and Bariatric Surgery.
To assess the impact of the National Coverage Determination on the safety of bariatric surgery, Dr. Dimick and his colleagues reviewed state inpatient data from 12 large, geographically dispersed states. Patients were identified on the basis of ICD-9 codes and diagnosis-related groups for weight loss surgery.
The outcomes were categorized according to any complication, a serious complication, or a reoperation.
The percentage of Medicare patients with any complications dropped from 12% before the determination to 8% afterward. Similarly, the percentage of non-Medicare patients with any complications dropped from 7% before to 5% after the determination.
In a multivariate analysis, the factors contributing to improved outcomes for bariatric surgery in Medicare patients were an increase in the use of laparoscopic gastric banding (lap band surgery), the transfer of patients to better hospitals, and quality improvement within individual hospitals, Dr. Dimick said.
Improvements in the safety of bariatric surgery did not, however, result from patients being redirected to safer hospitals, said Dr. Dimick. In fact, complication rates for procedures performed at Centers of Excellence versus non–Centers of Excellence were not significantly different (odds ratio for any complications, 0.97), he noted.
"CMS should consider dropping the COE [Centers of Excellence] aspect of the coverage decision [that] limits patient access without a beneficial improvement in outcomes," Dr. Dimick said. "Alternatively, CMS could revise the national coverage decision to further encourage participation in a quality improvement registry." Such a registry would need to include measures of long-term effectiveness to identify any unintended consequences of the increase in lap band surgery, he added.
Dr. Dimick is an equity owner and cofounder of ArborMetrix, a health care analytics and software firm.
CHICAGO – Complication rates for bariatric surgery patients on Medicare declined after Medicare began to cover the procedure in 2006, an analysis of state inpatient data from 12 states has found.
Data from previous studies suggest that bariatric surgery outcomes for Medicare patients improved after the implementation of Medicare’s National Coverage Determination in 2006, said Dr. Justin B. Dimick of the University of Michigan, Ann Arbor. But specific safety data on Medicare patients who have undergone bariatric surgery since the time of the decision are limited, Dr. Dimick said. The decision allowed for Medicare coverage of patients who sought care at facilities certified by the American College of Surgeons or the American Society for Metabolic and Bariatric Surgery.
To assess the impact of the National Coverage Determination on the safety of bariatric surgery, Dr. Dimick and his colleagues reviewed state inpatient data from 12 large, geographically dispersed states. Patients were identified on the basis of ICD-9 codes and diagnosis-related groups for weight loss surgery.
The outcomes were categorized according to any complication, a serious complication, or a reoperation.
The percentage of Medicare patients with any complications dropped from 12% before the determination to 8% afterward. Similarly, the percentage of non-Medicare patients with any complications dropped from 7% before to 5% after the determination.
In a multivariate analysis, the factors contributing to improved outcomes for bariatric surgery in Medicare patients were an increase in the use of laparoscopic gastric banding (lap band surgery), the transfer of patients to better hospitals, and quality improvement within individual hospitals, Dr. Dimick said.
Improvements in the safety of bariatric surgery did not, however, result from patients being redirected to safer hospitals, said Dr. Dimick. In fact, complication rates for procedures performed at Centers of Excellence versus non–Centers of Excellence were not significantly different (odds ratio for any complications, 0.97), he noted.
"CMS should consider dropping the COE [Centers of Excellence] aspect of the coverage decision [that] limits patient access without a beneficial improvement in outcomes," Dr. Dimick said. "Alternatively, CMS could revise the national coverage decision to further encourage participation in a quality improvement registry." Such a registry would need to include measures of long-term effectiveness to identify any unintended consequences of the increase in lap band surgery, he added.
Dr. Dimick is an equity owner and cofounder of ArborMetrix, a health care analytics and software firm.
AT THE ANNUAL CLINICAL CONGRESS OF THE AMERICAN COLLEGE OF SURGEONS
Major Finding: The percentage of Medicare patients with any complications dropped from 12% before the National Coverage Determination to 8% afterward.
Data Source: The data come from state inpatient data from 12 states.
Disclosures: Dr. Dimick is an equity owner and cofounder of ArborMetrix, a health care analytics and software firm.
The Changing Role of the Physician
As a young doctor, I often dreaded the thought of retirement, since that meant I would be leaving a vibrant field of medicine dedicated to improving the health of patients. At the end of my health care career, however (cut short because of illness), I find that our health care system is not anything I envisioned it would be, and as a patient I consider it nothing short of scary.
A physician’s role in health care should be to promote affordable quality care for the patient. The importance of this role was exemplified in the final Centers for Medicare and Medicaid Services (CMS) regulation that repealed the requirement for local hospital boards but – to help offset concerns regarding "the importance of physician input at the governing body level" – inserted a requirement that at least one physician serve on a facility’s hospital board. It was stated that this requirement "will build in an important element of continuity and ensure regular communication between a hospital’s governing body and its medical staff(s)," and that "a physician who specifically represents medical staff members will hold some measure of enhanced standing within the governing body" (Fed. Reg. 42 CFR Parts 482 and 485, vol. 77, May 16, 2012, 29038).
A month later, the American Hospital Association announced that this regulation had been placed on hold (CMS Reconsidering Board Medical Staff Requirement. AHANews.com, June 18, 2012). Three major concerns arose:
• First, how could CMS, which initially upheld the key role of the physician in the health care delivery system, suddenly reverse course and mitigate that role?
• Second, this was an unbelievable demonstration of the health care industry’s power to almost instantly stop the implementation of a final regulation in its tracks, bringing into question the meaning of the word "final." This power can only be expected to increase as mergers continue to take place (Abelson, R. Hospital Groups Will Get Bigger, Moody's Predicts. New York Times, March 8, 2012), and private equity firms and insurance companies enter the provider market (Niewenhous, M.D.; Private Equity's Interest in Non-Profit Hospitals: What's Next? HealthLeadersMedia; Caramenico, A. Hospital Groups Grow With Insurer, Private Equity Partners. FierceHealthCare. March 8, 2012). As the health care system drifts from being patient centered to cost driven, there may be a concomitant increase in health care expenditures (Gaynor, M.; Town, R. The Impact of Hospital Consolidation - Update. Robert Wood Johnson Foundation. June 2012).
• Finally, physicians having an important voice in governance is a major concern. Most physicians are becoming employees of hospitals and are no longer their customers. As employees, physicians may encounter difficulties maintaining their primary fiduciary responsibility to the patient. As our health care system rapidly changes and as Accountable Care Organizations take hold, safeguarding the physician’s primary fiduciary responsibility is also a concern. Regulations for ACOs offer little protection to the physician, and clearly state that "the governing body members shall have a fiduciary duty to the ACO and must act consistent with that fiduciary duty" (Fed. Reg. 42 CFR Part 425, Vol. 76, Nov. 2, 2011, 67819).
During the comment period for the ACO regulation, consumer advocate organizations (including the Citizen Advocacy Center, Cautious Patient Foundation, Health Watch USA, and Mothers Against Medical Error) and consumer advocates expressed concern about mandating that the actions of the physician be in the best interest of the organization rather than the patient. They suggested the following revision: "A registered nurse, licensed practical nurse, advanced nurse practitioner, doctor of allopathic medicine, doctor of osteopathic medicine, or other health care provider with substantially similar responsibilities shall have their primary fiduciary responsibility to the patient and not to an institution or corporation which employs them, or to an entity which reimburses them for their services."
However, CMS chose not to adopt this proposed revision, thereby missing a vital opportunity to ensure a patient-centered rather than a cost-driven health care system.
Physician organizations have the ability to respond to and halt such occurrences. Supporting patients and having physicians as an important part of a hospital’s governing body are issues that physicians would support, and so would the public.
Both vertical integration by acquiring physician practices (N. Engl. J. Med. 2011;364:1790-3) and horizontal integration through facility mergers have increased both the market and political power of an oligopolistic industry, which now can be governed by a single systemwide hospital board without physician representation. Physician hospital employment, the largest factor in vertical integration, is driven by large Medicare payment discrepancies between freestanding outpatient and hospital outpatient services (Kavanagh, K.T. Health Care Integration: Will Physicians Lose Their Voice? Bull. Am. Coll. Surg.; June 2011). The Medicare Payment Advisory Committee (MedPAC) has recommended that this discrepancy be eliminated for evaluation and management codes, which by the use of facility fees results in an 80% increase in payment for a 15-minute office visit provided by a hospital-owned physician practice (MedPAC, Report to Congress. March 2012, p. xiv).
Physicians must recapture a leadership role in health care. To do so, they should embrace transparency and adopt meaningful health care quality measures that generate data regarding the higher quality of care that they deliver.
As a physician, I believe that health care providers are talented, respected, and caring individuals who want to relieve pain and suffering and practice in a patient-centered health care environment. And as a patient, I would like to be treated in a health care system where providers have significant and meaningful input into its governance and always have the patient as their primary fiduciary responsibility.
Dr. Kavanagh is a retired otolaryngologist and board chairman of Health Watch USA.
As a young doctor, I often dreaded the thought of retirement, since that meant I would be leaving a vibrant field of medicine dedicated to improving the health of patients. At the end of my health care career, however (cut short because of illness), I find that our health care system is not anything I envisioned it would be, and as a patient I consider it nothing short of scary.
A physician’s role in health care should be to promote affordable quality care for the patient. The importance of this role was exemplified in the final Centers for Medicare and Medicaid Services (CMS) regulation that repealed the requirement for local hospital boards but – to help offset concerns regarding "the importance of physician input at the governing body level" – inserted a requirement that at least one physician serve on a facility’s hospital board. It was stated that this requirement "will build in an important element of continuity and ensure regular communication between a hospital’s governing body and its medical staff(s)," and that "a physician who specifically represents medical staff members will hold some measure of enhanced standing within the governing body" (Fed. Reg. 42 CFR Parts 482 and 485, vol. 77, May 16, 2012, 29038).
A month later, the American Hospital Association announced that this regulation had been placed on hold (CMS Reconsidering Board Medical Staff Requirement. AHANews.com, June 18, 2012). Three major concerns arose:
• First, how could CMS, which initially upheld the key role of the physician in the health care delivery system, suddenly reverse course and mitigate that role?
• Second, this was an unbelievable demonstration of the health care industry’s power to almost instantly stop the implementation of a final regulation in its tracks, bringing into question the meaning of the word "final." This power can only be expected to increase as mergers continue to take place (Abelson, R. Hospital Groups Will Get Bigger, Moody's Predicts. New York Times, March 8, 2012), and private equity firms and insurance companies enter the provider market (Niewenhous, M.D.; Private Equity's Interest in Non-Profit Hospitals: What's Next? HealthLeadersMedia; Caramenico, A. Hospital Groups Grow With Insurer, Private Equity Partners. FierceHealthCare. March 8, 2012). As the health care system drifts from being patient centered to cost driven, there may be a concomitant increase in health care expenditures (Gaynor, M.; Town, R. The Impact of Hospital Consolidation - Update. Robert Wood Johnson Foundation. June 2012).
• Finally, physicians having an important voice in governance is a major concern. Most physicians are becoming employees of hospitals and are no longer their customers. As employees, physicians may encounter difficulties maintaining their primary fiduciary responsibility to the patient. As our health care system rapidly changes and as Accountable Care Organizations take hold, safeguarding the physician’s primary fiduciary responsibility is also a concern. Regulations for ACOs offer little protection to the physician, and clearly state that "the governing body members shall have a fiduciary duty to the ACO and must act consistent with that fiduciary duty" (Fed. Reg. 42 CFR Part 425, Vol. 76, Nov. 2, 2011, 67819).
During the comment period for the ACO regulation, consumer advocate organizations (including the Citizen Advocacy Center, Cautious Patient Foundation, Health Watch USA, and Mothers Against Medical Error) and consumer advocates expressed concern about mandating that the actions of the physician be in the best interest of the organization rather than the patient. They suggested the following revision: "A registered nurse, licensed practical nurse, advanced nurse practitioner, doctor of allopathic medicine, doctor of osteopathic medicine, or other health care provider with substantially similar responsibilities shall have their primary fiduciary responsibility to the patient and not to an institution or corporation which employs them, or to an entity which reimburses them for their services."
However, CMS chose not to adopt this proposed revision, thereby missing a vital opportunity to ensure a patient-centered rather than a cost-driven health care system.
Physician organizations have the ability to respond to and halt such occurrences. Supporting patients and having physicians as an important part of a hospital’s governing body are issues that physicians would support, and so would the public.
Both vertical integration by acquiring physician practices (N. Engl. J. Med. 2011;364:1790-3) and horizontal integration through facility mergers have increased both the market and political power of an oligopolistic industry, which now can be governed by a single systemwide hospital board without physician representation. Physician hospital employment, the largest factor in vertical integration, is driven by large Medicare payment discrepancies between freestanding outpatient and hospital outpatient services (Kavanagh, K.T. Health Care Integration: Will Physicians Lose Their Voice? Bull. Am. Coll. Surg.; June 2011). The Medicare Payment Advisory Committee (MedPAC) has recommended that this discrepancy be eliminated for evaluation and management codes, which by the use of facility fees results in an 80% increase in payment for a 15-minute office visit provided by a hospital-owned physician practice (MedPAC, Report to Congress. March 2012, p. xiv).
Physicians must recapture a leadership role in health care. To do so, they should embrace transparency and adopt meaningful health care quality measures that generate data regarding the higher quality of care that they deliver.
As a physician, I believe that health care providers are talented, respected, and caring individuals who want to relieve pain and suffering and practice in a patient-centered health care environment. And as a patient, I would like to be treated in a health care system where providers have significant and meaningful input into its governance and always have the patient as their primary fiduciary responsibility.
Dr. Kavanagh is a retired otolaryngologist and board chairman of Health Watch USA.
As a young doctor, I often dreaded the thought of retirement, since that meant I would be leaving a vibrant field of medicine dedicated to improving the health of patients. At the end of my health care career, however (cut short because of illness), I find that our health care system is not anything I envisioned it would be, and as a patient I consider it nothing short of scary.
A physician’s role in health care should be to promote affordable quality care for the patient. The importance of this role was exemplified in the final Centers for Medicare and Medicaid Services (CMS) regulation that repealed the requirement for local hospital boards but – to help offset concerns regarding "the importance of physician input at the governing body level" – inserted a requirement that at least one physician serve on a facility’s hospital board. It was stated that this requirement "will build in an important element of continuity and ensure regular communication between a hospital’s governing body and its medical staff(s)," and that "a physician who specifically represents medical staff members will hold some measure of enhanced standing within the governing body" (Fed. Reg. 42 CFR Parts 482 and 485, vol. 77, May 16, 2012, 29038).
A month later, the American Hospital Association announced that this regulation had been placed on hold (CMS Reconsidering Board Medical Staff Requirement. AHANews.com, June 18, 2012). Three major concerns arose:
• First, how could CMS, which initially upheld the key role of the physician in the health care delivery system, suddenly reverse course and mitigate that role?
• Second, this was an unbelievable demonstration of the health care industry’s power to almost instantly stop the implementation of a final regulation in its tracks, bringing into question the meaning of the word "final." This power can only be expected to increase as mergers continue to take place (Abelson, R. Hospital Groups Will Get Bigger, Moody's Predicts. New York Times, March 8, 2012), and private equity firms and insurance companies enter the provider market (Niewenhous, M.D.; Private Equity's Interest in Non-Profit Hospitals: What's Next? HealthLeadersMedia; Caramenico, A. Hospital Groups Grow With Insurer, Private Equity Partners. FierceHealthCare. March 8, 2012). As the health care system drifts from being patient centered to cost driven, there may be a concomitant increase in health care expenditures (Gaynor, M.; Town, R. The Impact of Hospital Consolidation - Update. Robert Wood Johnson Foundation. June 2012).
• Finally, physicians having an important voice in governance is a major concern. Most physicians are becoming employees of hospitals and are no longer their customers. As employees, physicians may encounter difficulties maintaining their primary fiduciary responsibility to the patient. As our health care system rapidly changes and as Accountable Care Organizations take hold, safeguarding the physician’s primary fiduciary responsibility is also a concern. Regulations for ACOs offer little protection to the physician, and clearly state that "the governing body members shall have a fiduciary duty to the ACO and must act consistent with that fiduciary duty" (Fed. Reg. 42 CFR Part 425, Vol. 76, Nov. 2, 2011, 67819).
During the comment period for the ACO regulation, consumer advocate organizations (including the Citizen Advocacy Center, Cautious Patient Foundation, Health Watch USA, and Mothers Against Medical Error) and consumer advocates expressed concern about mandating that the actions of the physician be in the best interest of the organization rather than the patient. They suggested the following revision: "A registered nurse, licensed practical nurse, advanced nurse practitioner, doctor of allopathic medicine, doctor of osteopathic medicine, or other health care provider with substantially similar responsibilities shall have their primary fiduciary responsibility to the patient and not to an institution or corporation which employs them, or to an entity which reimburses them for their services."
However, CMS chose not to adopt this proposed revision, thereby missing a vital opportunity to ensure a patient-centered rather than a cost-driven health care system.
Physician organizations have the ability to respond to and halt such occurrences. Supporting patients and having physicians as an important part of a hospital’s governing body are issues that physicians would support, and so would the public.
Both vertical integration by acquiring physician practices (N. Engl. J. Med. 2011;364:1790-3) and horizontal integration through facility mergers have increased both the market and political power of an oligopolistic industry, which now can be governed by a single systemwide hospital board without physician representation. Physician hospital employment, the largest factor in vertical integration, is driven by large Medicare payment discrepancies between freestanding outpatient and hospital outpatient services (Kavanagh, K.T. Health Care Integration: Will Physicians Lose Their Voice? Bull. Am. Coll. Surg.; June 2011). The Medicare Payment Advisory Committee (MedPAC) has recommended that this discrepancy be eliminated for evaluation and management codes, which by the use of facility fees results in an 80% increase in payment for a 15-minute office visit provided by a hospital-owned physician practice (MedPAC, Report to Congress. March 2012, p. xiv).
Physicians must recapture a leadership role in health care. To do so, they should embrace transparency and adopt meaningful health care quality measures that generate data regarding the higher quality of care that they deliver.
As a physician, I believe that health care providers are talented, respected, and caring individuals who want to relieve pain and suffering and practice in a patient-centered health care environment. And as a patient, I would like to be treated in a health care system where providers have significant and meaningful input into its governance and always have the patient as their primary fiduciary responsibility.
Dr. Kavanagh is a retired otolaryngologist and board chairman of Health Watch USA.
Vein Sealed With Superglue-Based Implant
ATLANTA – A cyanoacrylate adhesive–based implant is feasible, safe, and effective for the treatment of great saphenous vein incompetence, according to 1-year follow-up data from the first study of the product in humans.
The treatment requires neither local anesthesia nor use of medical compression stockings, Dr. Thomas Proebstle reported at the annual meeting of the American Society for Dermatologic Surgery.
Of 38 patients with an incompetent great saphenous vein who were treated with a proprietary formulation of cyanoacrylate adhesive (commonly known as superglue), 100% demonstrated complete closure of the vein immediately and at the 48-hour follow-up, as measured using duplex ultrasound and clinical examination; at 1-year follow-up, 92% maintained complete closure of the vein, said Dr. Proebstle of Hirschberg, Germany. One complete recanalization and two partial recanalizations occurred during follow-up – at 1, 3, and 6 months, respectively, he noted.
Most patients (89%) had improvement in leg edema within 48 hours, and all had venous clinical severity score improvement, which changed from a mean of 6.1 at baseline to a mean of 1.1 at 6 months.
The study involved 29 women and 9 men with a median age of 51 years. Treatment was administered by catheter deployment under ultrasound guidance via a repetitive bolus injection algorithm, Dr. Proebstle noted. No tumescent anesthesia or compression stockings were used.
For this study, the mean total volume of endovenous cyanoacrylate adhesive delivered was 1.26 mL. Side effects were mild and self-limited.
The cyanoacrylate adhesive implant, known as the VenaSeal Sapheon closure system, has received European Union regulatory approval. The manufacturer, Sapheon, is currently preparing for U.S. clinical trials and Food and Drug Administration approval, according to the company’s website.
Dr. Proebstle is a consultant for Sapheon. He also owns stock in the company.
ATLANTA – A cyanoacrylate adhesive–based implant is feasible, safe, and effective for the treatment of great saphenous vein incompetence, according to 1-year follow-up data from the first study of the product in humans.
The treatment requires neither local anesthesia nor use of medical compression stockings, Dr. Thomas Proebstle reported at the annual meeting of the American Society for Dermatologic Surgery.
Of 38 patients with an incompetent great saphenous vein who were treated with a proprietary formulation of cyanoacrylate adhesive (commonly known as superglue), 100% demonstrated complete closure of the vein immediately and at the 48-hour follow-up, as measured using duplex ultrasound and clinical examination; at 1-year follow-up, 92% maintained complete closure of the vein, said Dr. Proebstle of Hirschberg, Germany. One complete recanalization and two partial recanalizations occurred during follow-up – at 1, 3, and 6 months, respectively, he noted.
Most patients (89%) had improvement in leg edema within 48 hours, and all had venous clinical severity score improvement, which changed from a mean of 6.1 at baseline to a mean of 1.1 at 6 months.
The study involved 29 women and 9 men with a median age of 51 years. Treatment was administered by catheter deployment under ultrasound guidance via a repetitive bolus injection algorithm, Dr. Proebstle noted. No tumescent anesthesia or compression stockings were used.
For this study, the mean total volume of endovenous cyanoacrylate adhesive delivered was 1.26 mL. Side effects were mild and self-limited.
The cyanoacrylate adhesive implant, known as the VenaSeal Sapheon closure system, has received European Union regulatory approval. The manufacturer, Sapheon, is currently preparing for U.S. clinical trials and Food and Drug Administration approval, according to the company’s website.
Dr. Proebstle is a consultant for Sapheon. He also owns stock in the company.
ATLANTA – A cyanoacrylate adhesive–based implant is feasible, safe, and effective for the treatment of great saphenous vein incompetence, according to 1-year follow-up data from the first study of the product in humans.
The treatment requires neither local anesthesia nor use of medical compression stockings, Dr. Thomas Proebstle reported at the annual meeting of the American Society for Dermatologic Surgery.
Of 38 patients with an incompetent great saphenous vein who were treated with a proprietary formulation of cyanoacrylate adhesive (commonly known as superglue), 100% demonstrated complete closure of the vein immediately and at the 48-hour follow-up, as measured using duplex ultrasound and clinical examination; at 1-year follow-up, 92% maintained complete closure of the vein, said Dr. Proebstle of Hirschberg, Germany. One complete recanalization and two partial recanalizations occurred during follow-up – at 1, 3, and 6 months, respectively, he noted.
Most patients (89%) had improvement in leg edema within 48 hours, and all had venous clinical severity score improvement, which changed from a mean of 6.1 at baseline to a mean of 1.1 at 6 months.
The study involved 29 women and 9 men with a median age of 51 years. Treatment was administered by catheter deployment under ultrasound guidance via a repetitive bolus injection algorithm, Dr. Proebstle noted. No tumescent anesthesia or compression stockings were used.
For this study, the mean total volume of endovenous cyanoacrylate adhesive delivered was 1.26 mL. Side effects were mild and self-limited.
The cyanoacrylate adhesive implant, known as the VenaSeal Sapheon closure system, has received European Union regulatory approval. The manufacturer, Sapheon, is currently preparing for U.S. clinical trials and Food and Drug Administration approval, according to the company’s website.
Dr. Proebstle is a consultant for Sapheon. He also owns stock in the company.
AT THE ANNUAL MEETING OF THE AMERICAN SOCIETY FOR DERMATOLOGIC SURGERY
Major Finding: Of 38 patients with an incompetent great saphenous vein who were treated with a proprietary formulation of cyanoacrylate adhesive, 100% demonstrated complete closure of the vein immediately and at the 48-hour follow-up.
Data Source: Data are from a prospective study involving 38 patients.
Disclosures: Dr. Proebstle is a consultant for Sapheon. He also owns stock in the company.
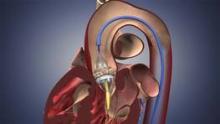
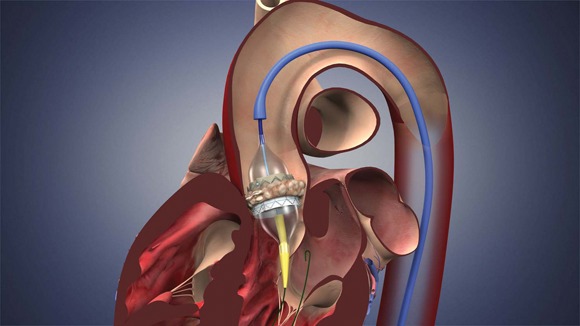
FDA Expands Sapien Approval
The Food and Drug Administration has expanded approval for the Sapien transcatheter heart valve for patients with aortic valve stenosis who are at high risk for serious surgical complications or death.
It is the second approval for the device, which is made by Edwards Lifesciences Corp. of Irvine, Calif. In November 2011, the FDA approved it for severe aortic valve stenosis in inoperable patients.
The indication for high-risk patients was reviewed by an FDA advisory panel in July. The majority of the panel indicated that the device was safe and effective in that group.
Both approvals were based on results from the PARTNER trial (N. Engl. J. Med. 2010;363:1597-607).
"Any procedure to replace the aortic valve carries the risk for serious complications, but for some patients with coexisting conditions or diseases, that risk may be especially high," Christy Foreman, director of the office of device evaluation at FDA’s Center for Devices and Radiological Health, said in a statement.
"The THV serves as an alternative for some very high-risk patients."
Larry L. Wood, Edwards’ corporate vice president, transcatheter heart valves, said in a statement: "It is extremely rewarding for us and our clinical partners to know that high-risk patients suffering from this often-debilitating disease will now have access to this life-saving therapy."
The Sapien THV is implanted at the site of the diseased valve using a catheter that is delivered either through a transfemoral or transapical approach. According to the Edwards statement, the transapical approach, which requires insertion through the ribs and myocardium, was not approved outside of clinical trials until now.
Transcatheter aortic valve replacement has become one of the driving forces in the heart valve market, which may hit $1.5 billion by 2016.
The new indication for the Sapien THV is expected to expand that market further. According to the FDA, the approved labeling indicates that a surgeon must determine whether a patient is eligible for the Sapien. But in fact, Edwards, the Centers for Medicare and Medicaid Services, and professional societies such as the American College of Cardiology and the Society of Thoracic Surgeons, have worked together to establish standards and requirements for "Heart Teams" that must be present during the procedure and evaluation of the patient by two cardiac surgeons.
There is an increased risk for major vascular complications and for stroke during the first month post implant. Thus, the device is contraindicated in patients who cannot tolerate anticoagulation/antiplatelet therapy.
Edwards will monitor safety and complication rates through the national Transcatheter Valve Therapy Registry.
The Food and Drug Administration has expanded approval for the Sapien transcatheter heart valve for patients with aortic valve stenosis who are at high risk for serious surgical complications or death.
It is the second approval for the device, which is made by Edwards Lifesciences Corp. of Irvine, Calif. In November 2011, the FDA approved it for severe aortic valve stenosis in inoperable patients.
The indication for high-risk patients was reviewed by an FDA advisory panel in July. The majority of the panel indicated that the device was safe and effective in that group.
Both approvals were based on results from the PARTNER trial (N. Engl. J. Med. 2010;363:1597-607).
"Any procedure to replace the aortic valve carries the risk for serious complications, but for some patients with coexisting conditions or diseases, that risk may be especially high," Christy Foreman, director of the office of device evaluation at FDA’s Center for Devices and Radiological Health, said in a statement.
"The THV serves as an alternative for some very high-risk patients."
Larry L. Wood, Edwards’ corporate vice president, transcatheter heart valves, said in a statement: "It is extremely rewarding for us and our clinical partners to know that high-risk patients suffering from this often-debilitating disease will now have access to this life-saving therapy."
The Sapien THV is implanted at the site of the diseased valve using a catheter that is delivered either through a transfemoral or transapical approach. According to the Edwards statement, the transapical approach, which requires insertion through the ribs and myocardium, was not approved outside of clinical trials until now.
Transcatheter aortic valve replacement has become one of the driving forces in the heart valve market, which may hit $1.5 billion by 2016.
The new indication for the Sapien THV is expected to expand that market further. According to the FDA, the approved labeling indicates that a surgeon must determine whether a patient is eligible for the Sapien. But in fact, Edwards, the Centers for Medicare and Medicaid Services, and professional societies such as the American College of Cardiology and the Society of Thoracic Surgeons, have worked together to establish standards and requirements for "Heart Teams" that must be present during the procedure and evaluation of the patient by two cardiac surgeons.
There is an increased risk for major vascular complications and for stroke during the first month post implant. Thus, the device is contraindicated in patients who cannot tolerate anticoagulation/antiplatelet therapy.
Edwards will monitor safety and complication rates through the national Transcatheter Valve Therapy Registry.
The Food and Drug Administration has expanded approval for the Sapien transcatheter heart valve for patients with aortic valve stenosis who are at high risk for serious surgical complications or death.
It is the second approval for the device, which is made by Edwards Lifesciences Corp. of Irvine, Calif. In November 2011, the FDA approved it for severe aortic valve stenosis in inoperable patients.
The indication for high-risk patients was reviewed by an FDA advisory panel in July. The majority of the panel indicated that the device was safe and effective in that group.
Both approvals were based on results from the PARTNER trial (N. Engl. J. Med. 2010;363:1597-607).
"Any procedure to replace the aortic valve carries the risk for serious complications, but for some patients with coexisting conditions or diseases, that risk may be especially high," Christy Foreman, director of the office of device evaluation at FDA’s Center for Devices and Radiological Health, said in a statement.
"The THV serves as an alternative for some very high-risk patients."
Larry L. Wood, Edwards’ corporate vice president, transcatheter heart valves, said in a statement: "It is extremely rewarding for us and our clinical partners to know that high-risk patients suffering from this often-debilitating disease will now have access to this life-saving therapy."
The Sapien THV is implanted at the site of the diseased valve using a catheter that is delivered either through a transfemoral or transapical approach. According to the Edwards statement, the transapical approach, which requires insertion through the ribs and myocardium, was not approved outside of clinical trials until now.
Transcatheter aortic valve replacement has become one of the driving forces in the heart valve market, which may hit $1.5 billion by 2016.
The new indication for the Sapien THV is expected to expand that market further. According to the FDA, the approved labeling indicates that a surgeon must determine whether a patient is eligible for the Sapien. But in fact, Edwards, the Centers for Medicare and Medicaid Services, and professional societies such as the American College of Cardiology and the Society of Thoracic Surgeons, have worked together to establish standards and requirements for "Heart Teams" that must be present during the procedure and evaluation of the patient by two cardiac surgeons.
There is an increased risk for major vascular complications and for stroke during the first month post implant. Thus, the device is contraindicated in patients who cannot tolerate anticoagulation/antiplatelet therapy.
Edwards will monitor safety and complication rates through the national Transcatheter Valve Therapy Registry.
Poor Coordination Blamed for Flow Disruptions in Trauma Care
CHICAGO – Breakdowns in the coordination of care, rather than communication, were the most common type of flow disruption at a level I trauma center, based on an analysis of data from 86 cases.
The finding contrasts with results from previous studies showing communication breakdowns as the top flow disruptions, and the study "is one of the first and largest to objectively document that flow disruptions in trauma care occur frequently," according to Dr. Daniel Shouhed of Cedars-Sinai Medical Center in Los Angeles.
"Defective systems allow human errors to cause harm to patients," Dr. Shouhed said at the annual clinical congress of the American College of Surgeons. "We believe systems can be better designed to prevent or detect errors before a patient is harmed."
As part of an effort to improve systems, Dr. Shouhed and his colleagues conducted a prospective observational study of flow disruptions over 2 months of 24-hour coverage in a level I trauma center.
Categories of flow disruptions included communication, coordination, environment, equipment, external interruptions, patient factors, technical skills, and training.
Communication breakdowns were defined as "disruptions that involve the verbal transition of information between at least two team members," while coordination breakdowns were defined as "disruptions that involve the interaction with some piece of equipment as well as at least one other team member," Dr. Shouhed said.
Flow disruptions were defined as low or high impact, based on whether they caused a pause in the progression of patient care or significant consequences to patient care. For example, a low-impact communication disruption might be "a scribe nurse unable to hear primary survey from resident," while a high-impact disruption might be "an ED attending did not hear request for STAT blood; order not placed."
Multiple physicians reviewed and scored each disruption. Each case could include three phases of care: trauma bay, imaging, and operating room (or interventional radiology).
The researchers observed 86 cases, 6 of which required immediate surgery. The average case duration was 102 minutes. On average, patients spent 25 minutes in the trauma bay, 30 minutes in the CT scanner, and 148 minutes in the operating room.
A total of 1,757 flow disruptions were observed. Of these, 46% occurred in the trauma bay, 33% in imaging, and 21% in the operating room. The researchers examined the effect of trauma level and trauma phase on the rate of flow disruptions, excluding the six OR cases because of the small sample size.
Flow disruption rates were divided into low-level (trauma 200) or high-level (trauma 100) activations, Dr. Shouhed said.
A comparison of the rate of flow disruptions between the trauma bay and imaging phases of care based on clinical impact scores showed a significantly higher rate of low-impact flow disruptions in the imaging phase, particularly among trauma 100 cases.
In examining the relationship between high clinical impact and low clinical impact flow disruptions, the researchers found a strong correlation between the rate of low-impact and high-impact flow disruptions among all phases of care.
"Most of the flow disruptions were low impact; however, we found that as the rate of low-impact flow disruptions increased, so did the rate of high-impact flow disruptions – thus attesting to the cascade of events that typically transpire when the progression of care is even slightly disrupted," said Dr. Shouhed.
Based on type of disruption and clinical impact, the highest rate of flow disruption involved breakdowns in coordination, which were associated with approximately three low clinical impact and one high clinical impact flow disruptions per hour.
"We believe that prospective observation allows individual hospitals to accurately identify systemic deficiencies and evaluate the impact interventions may have on improving patient safety and efficiency of care," he added.
The study was funded by a Military Operating Room of the Future grant from the Department of Defense.
CHICAGO – Breakdowns in the coordination of care, rather than communication, were the most common type of flow disruption at a level I trauma center, based on an analysis of data from 86 cases.
The finding contrasts with results from previous studies showing communication breakdowns as the top flow disruptions, and the study "is one of the first and largest to objectively document that flow disruptions in trauma care occur frequently," according to Dr. Daniel Shouhed of Cedars-Sinai Medical Center in Los Angeles.
"Defective systems allow human errors to cause harm to patients," Dr. Shouhed said at the annual clinical congress of the American College of Surgeons. "We believe systems can be better designed to prevent or detect errors before a patient is harmed."
As part of an effort to improve systems, Dr. Shouhed and his colleagues conducted a prospective observational study of flow disruptions over 2 months of 24-hour coverage in a level I trauma center.
Categories of flow disruptions included communication, coordination, environment, equipment, external interruptions, patient factors, technical skills, and training.
Communication breakdowns were defined as "disruptions that involve the verbal transition of information between at least two team members," while coordination breakdowns were defined as "disruptions that involve the interaction with some piece of equipment as well as at least one other team member," Dr. Shouhed said.
Flow disruptions were defined as low or high impact, based on whether they caused a pause in the progression of patient care or significant consequences to patient care. For example, a low-impact communication disruption might be "a scribe nurse unable to hear primary survey from resident," while a high-impact disruption might be "an ED attending did not hear request for STAT blood; order not placed."
Multiple physicians reviewed and scored each disruption. Each case could include three phases of care: trauma bay, imaging, and operating room (or interventional radiology).
The researchers observed 86 cases, 6 of which required immediate surgery. The average case duration was 102 minutes. On average, patients spent 25 minutes in the trauma bay, 30 minutes in the CT scanner, and 148 minutes in the operating room.
A total of 1,757 flow disruptions were observed. Of these, 46% occurred in the trauma bay, 33% in imaging, and 21% in the operating room. The researchers examined the effect of trauma level and trauma phase on the rate of flow disruptions, excluding the six OR cases because of the small sample size.
Flow disruption rates were divided into low-level (trauma 200) or high-level (trauma 100) activations, Dr. Shouhed said.
A comparison of the rate of flow disruptions between the trauma bay and imaging phases of care based on clinical impact scores showed a significantly higher rate of low-impact flow disruptions in the imaging phase, particularly among trauma 100 cases.
In examining the relationship between high clinical impact and low clinical impact flow disruptions, the researchers found a strong correlation between the rate of low-impact and high-impact flow disruptions among all phases of care.
"Most of the flow disruptions were low impact; however, we found that as the rate of low-impact flow disruptions increased, so did the rate of high-impact flow disruptions – thus attesting to the cascade of events that typically transpire when the progression of care is even slightly disrupted," said Dr. Shouhed.
Based on type of disruption and clinical impact, the highest rate of flow disruption involved breakdowns in coordination, which were associated with approximately three low clinical impact and one high clinical impact flow disruptions per hour.
"We believe that prospective observation allows individual hospitals to accurately identify systemic deficiencies and evaluate the impact interventions may have on improving patient safety and efficiency of care," he added.
The study was funded by a Military Operating Room of the Future grant from the Department of Defense.
CHICAGO – Breakdowns in the coordination of care, rather than communication, were the most common type of flow disruption at a level I trauma center, based on an analysis of data from 86 cases.
The finding contrasts with results from previous studies showing communication breakdowns as the top flow disruptions, and the study "is one of the first and largest to objectively document that flow disruptions in trauma care occur frequently," according to Dr. Daniel Shouhed of Cedars-Sinai Medical Center in Los Angeles.
"Defective systems allow human errors to cause harm to patients," Dr. Shouhed said at the annual clinical congress of the American College of Surgeons. "We believe systems can be better designed to prevent or detect errors before a patient is harmed."
As part of an effort to improve systems, Dr. Shouhed and his colleagues conducted a prospective observational study of flow disruptions over 2 months of 24-hour coverage in a level I trauma center.
Categories of flow disruptions included communication, coordination, environment, equipment, external interruptions, patient factors, technical skills, and training.
Communication breakdowns were defined as "disruptions that involve the verbal transition of information between at least two team members," while coordination breakdowns were defined as "disruptions that involve the interaction with some piece of equipment as well as at least one other team member," Dr. Shouhed said.
Flow disruptions were defined as low or high impact, based on whether they caused a pause in the progression of patient care or significant consequences to patient care. For example, a low-impact communication disruption might be "a scribe nurse unable to hear primary survey from resident," while a high-impact disruption might be "an ED attending did not hear request for STAT blood; order not placed."
Multiple physicians reviewed and scored each disruption. Each case could include three phases of care: trauma bay, imaging, and operating room (or interventional radiology).
The researchers observed 86 cases, 6 of which required immediate surgery. The average case duration was 102 minutes. On average, patients spent 25 minutes in the trauma bay, 30 minutes in the CT scanner, and 148 minutes in the operating room.
A total of 1,757 flow disruptions were observed. Of these, 46% occurred in the trauma bay, 33% in imaging, and 21% in the operating room. The researchers examined the effect of trauma level and trauma phase on the rate of flow disruptions, excluding the six OR cases because of the small sample size.
Flow disruption rates were divided into low-level (trauma 200) or high-level (trauma 100) activations, Dr. Shouhed said.
A comparison of the rate of flow disruptions between the trauma bay and imaging phases of care based on clinical impact scores showed a significantly higher rate of low-impact flow disruptions in the imaging phase, particularly among trauma 100 cases.
In examining the relationship between high clinical impact and low clinical impact flow disruptions, the researchers found a strong correlation between the rate of low-impact and high-impact flow disruptions among all phases of care.
"Most of the flow disruptions were low impact; however, we found that as the rate of low-impact flow disruptions increased, so did the rate of high-impact flow disruptions – thus attesting to the cascade of events that typically transpire when the progression of care is even slightly disrupted," said Dr. Shouhed.
Based on type of disruption and clinical impact, the highest rate of flow disruption involved breakdowns in coordination, which were associated with approximately three low clinical impact and one high clinical impact flow disruptions per hour.
"We believe that prospective observation allows individual hospitals to accurately identify systemic deficiencies and evaluate the impact interventions may have on improving patient safety and efficiency of care," he added.
The study was funded by a Military Operating Room of the Future grant from the Department of Defense.
FROM THE ANNUAL CLINICAL CONGRESS OF THE AMERICAN COLLEGE OF SURGEONS
Major Finding: A total of 1,757 flow disruptions were observed. Of these, 46% occurred in the trauma bay, 33% in imaging, and 21% in the operating room.
Data Source: The data come from a prospective observational study of 24-hour coverage for 2 months at a level I trauma center.
Disclosures: The study was funded by a Military Operating Room of the Future grant from the Department of Defense.
Scrutinizing Health Reform: The Policy & Practice Podcast
With just weeks left before the election, health reform talk has slowed down quite a bit. That said, physician groups called on Congress to put the Sustainable Growth Rate formula back on the political agenda. In a letter to Congress, the American Medical Association and more than 100 other groups renewed their call for an SGR repeal, and outlined the ideal transition model. Their plan is one of several alternatives on the table.
Meanwhile, at their annual Congress of Delegates, the American Academy of Family Physicians called out states that are choosing to forgo Medicaid expansion under the Affordable Care Act, saying that partisan politics is overriding good judgment and the extension of health care coverage to more Americans. So far, Texas, South Carolina, Nebraska, Iowa, Louisiana, Mississippi, and Florida have all said that their states will opt out.
At the same time, the law’s skeptics drew some attention to the ACA's Prevention and Public Health Fund. The Government Accountability Office released a report, requested by GOP senators, detailing exactly where the fund’s $14 billion has gone. For the details on that and more, listen to this week’s Policy & Practice Podcast.
--Frances Correa (@FMCReporting)
With just weeks left before the election, health reform talk has slowed down quite a bit. That said, physician groups called on Congress to put the Sustainable Growth Rate formula back on the political agenda. In a letter to Congress, the American Medical Association and more than 100 other groups renewed their call for an SGR repeal, and outlined the ideal transition model. Their plan is one of several alternatives on the table.
Meanwhile, at their annual Congress of Delegates, the American Academy of Family Physicians called out states that are choosing to forgo Medicaid expansion under the Affordable Care Act, saying that partisan politics is overriding good judgment and the extension of health care coverage to more Americans. So far, Texas, South Carolina, Nebraska, Iowa, Louisiana, Mississippi, and Florida have all said that their states will opt out.
At the same time, the law’s skeptics drew some attention to the ACA's Prevention and Public Health Fund. The Government Accountability Office released a report, requested by GOP senators, detailing exactly where the fund’s $14 billion has gone. For the details on that and more, listen to this week’s Policy & Practice Podcast.
--Frances Correa (@FMCReporting)
With just weeks left before the election, health reform talk has slowed down quite a bit. That said, physician groups called on Congress to put the Sustainable Growth Rate formula back on the political agenda. In a letter to Congress, the American Medical Association and more than 100 other groups renewed their call for an SGR repeal, and outlined the ideal transition model. Their plan is one of several alternatives on the table.
Meanwhile, at their annual Congress of Delegates, the American Academy of Family Physicians called out states that are choosing to forgo Medicaid expansion under the Affordable Care Act, saying that partisan politics is overriding good judgment and the extension of health care coverage to more Americans. So far, Texas, South Carolina, Nebraska, Iowa, Louisiana, Mississippi, and Florida have all said that their states will opt out.
At the same time, the law’s skeptics drew some attention to the ACA's Prevention and Public Health Fund. The Government Accountability Office released a report, requested by GOP senators, detailing exactly where the fund’s $14 billion has gone. For the details on that and more, listen to this week’s Policy & Practice Podcast.
--Frances Correa (@FMCReporting)
Simple Surgical Risk Tool Useful for Resource-Limited Areas
A risk-adjusted tool based on three preoperative variables from the National Surgical Quality Improvement Program had a high rate of efficacy in predicting inpatient mortality, which suggests it may be useful in resource-limited settings such as small rural hospitals or low- and middle-income countries.
"By offering a simplified risk-adjustment tool, we can compare surgical outcomes among hospitals on a global scale, regardless of the spectrum of surgical procedures offered or hospital resources," Jamie E. Anderson and associates in the department of surgery, University of California, San Diego, wrote in Archives of Surgery.
"Although participation in programs such as the NSQIP offers administrative support and comparison of outcomes among participating hospitals, the low-cost options reported can expand the number of hospitals that participate in risk-adjustment outcomes analysis and quality improvement programs," they added.
The American College of Surgeons’ NSQIP risk-adjusted tool uses more than 130 variables plus a 30-day patient follow-up, and thus is not affordable for use in settings where resources are limited, including small, rural hospitals, the authors pointed out (Arch. Surg. 2012;147:798-803).
Using data from more than 600,000 patients in the 2005-2009 NSQIP database, they developed different models to predict inpatient mortality and validated the models based on data on 239 patients from a 110-bed hospital in California with a level IV trauma center. They calculated that the "area under the receiver operator characteristic curve (AUROC)" for each model as a measure of how well the model separated the two groups of interest (survivors vs. nonsurvivors) with a value of 1.0 (or 100%) would mean that the model was able to completely separate the two groups.
The model using three preoperative NSQIP variables – age, American Society of Anesthesiologists (ASA) physical status classification, and functional status – had AUROC values that were more than 0.90, or more than 90%, which was similar to the value achieved for the model that used four or six variables. The model that used 66 variables was about the same as the value achieved with the model that used 4 variables (about 91%).
Considering that an AUROC value of 0.5 indicates that the model cannot distinguish between two groups any better than chance and that an AUROC value of 1.0 indicates that the model completely discriminates between the two groups, the authors said that an AUROC value of greater than 90% is substantial. Therefore, based on their results, "3 or 4 variables may be sufficient for adequate risk adjustment to measure surgical outcomes," they added.
"Future risk-adjustment tools [should] be based on 6 or fewer variables to allow for surgical outcomes to be measured and compared within and among hospitals in resource-limited settings," they concluded.
The authors had no disclosures.
In an accompanying editorial titled "NSQIP Lite," Dr. Diana L. Farmer wrote that, while the statistical methods used were "dense," the significance of this study should not be overlooked and that "the development of a simple and inexpensive tool that could be used in the most resource-poor settings in this country and around the world is of enormous importance." The tool, which needs to be validated further in diverse settings, "stands as a powerful potential mechanism for answering the most compelling question of in-hospital mortality," she added (Arch. Surg. 2012;147:803-4).
Dr. Farmer is chair of the department of surgery at UC Davis Health Systems, Sacramento, Calif. She had no disclosures to report.
In an accompanying editorial titled "NSQIP Lite," Dr. Diana L. Farmer wrote that, while the statistical methods used were "dense," the significance of this study should not be overlooked and that "the development of a simple and inexpensive tool that could be used in the most resource-poor settings in this country and around the world is of enormous importance." The tool, which needs to be validated further in diverse settings, "stands as a powerful potential mechanism for answering the most compelling question of in-hospital mortality," she added (Arch. Surg. 2012;147:803-4).
Dr. Farmer is chair of the department of surgery at UC Davis Health Systems, Sacramento, Calif. She had no disclosures to report.
In an accompanying editorial titled "NSQIP Lite," Dr. Diana L. Farmer wrote that, while the statistical methods used were "dense," the significance of this study should not be overlooked and that "the development of a simple and inexpensive tool that could be used in the most resource-poor settings in this country and around the world is of enormous importance." The tool, which needs to be validated further in diverse settings, "stands as a powerful potential mechanism for answering the most compelling question of in-hospital mortality," she added (Arch. Surg. 2012;147:803-4).
Dr. Farmer is chair of the department of surgery at UC Davis Health Systems, Sacramento, Calif. She had no disclosures to report.
A risk-adjusted tool based on three preoperative variables from the National Surgical Quality Improvement Program had a high rate of efficacy in predicting inpatient mortality, which suggests it may be useful in resource-limited settings such as small rural hospitals or low- and middle-income countries.
"By offering a simplified risk-adjustment tool, we can compare surgical outcomes among hospitals on a global scale, regardless of the spectrum of surgical procedures offered or hospital resources," Jamie E. Anderson and associates in the department of surgery, University of California, San Diego, wrote in Archives of Surgery.
"Although participation in programs such as the NSQIP offers administrative support and comparison of outcomes among participating hospitals, the low-cost options reported can expand the number of hospitals that participate in risk-adjustment outcomes analysis and quality improvement programs," they added.
The American College of Surgeons’ NSQIP risk-adjusted tool uses more than 130 variables plus a 30-day patient follow-up, and thus is not affordable for use in settings where resources are limited, including small, rural hospitals, the authors pointed out (Arch. Surg. 2012;147:798-803).
Using data from more than 600,000 patients in the 2005-2009 NSQIP database, they developed different models to predict inpatient mortality and validated the models based on data on 239 patients from a 110-bed hospital in California with a level IV trauma center. They calculated that the "area under the receiver operator characteristic curve (AUROC)" for each model as a measure of how well the model separated the two groups of interest (survivors vs. nonsurvivors) with a value of 1.0 (or 100%) would mean that the model was able to completely separate the two groups.
The model using three preoperative NSQIP variables – age, American Society of Anesthesiologists (ASA) physical status classification, and functional status – had AUROC values that were more than 0.90, or more than 90%, which was similar to the value achieved for the model that used four or six variables. The model that used 66 variables was about the same as the value achieved with the model that used 4 variables (about 91%).
Considering that an AUROC value of 0.5 indicates that the model cannot distinguish between two groups any better than chance and that an AUROC value of 1.0 indicates that the model completely discriminates between the two groups, the authors said that an AUROC value of greater than 90% is substantial. Therefore, based on their results, "3 or 4 variables may be sufficient for adequate risk adjustment to measure surgical outcomes," they added.
"Future risk-adjustment tools [should] be based on 6 or fewer variables to allow for surgical outcomes to be measured and compared within and among hospitals in resource-limited settings," they concluded.
The authors had no disclosures.
A risk-adjusted tool based on three preoperative variables from the National Surgical Quality Improvement Program had a high rate of efficacy in predicting inpatient mortality, which suggests it may be useful in resource-limited settings such as small rural hospitals or low- and middle-income countries.
"By offering a simplified risk-adjustment tool, we can compare surgical outcomes among hospitals on a global scale, regardless of the spectrum of surgical procedures offered or hospital resources," Jamie E. Anderson and associates in the department of surgery, University of California, San Diego, wrote in Archives of Surgery.
"Although participation in programs such as the NSQIP offers administrative support and comparison of outcomes among participating hospitals, the low-cost options reported can expand the number of hospitals that participate in risk-adjustment outcomes analysis and quality improvement programs," they added.
The American College of Surgeons’ NSQIP risk-adjusted tool uses more than 130 variables plus a 30-day patient follow-up, and thus is not affordable for use in settings where resources are limited, including small, rural hospitals, the authors pointed out (Arch. Surg. 2012;147:798-803).
Using data from more than 600,000 patients in the 2005-2009 NSQIP database, they developed different models to predict inpatient mortality and validated the models based on data on 239 patients from a 110-bed hospital in California with a level IV trauma center. They calculated that the "area under the receiver operator characteristic curve (AUROC)" for each model as a measure of how well the model separated the two groups of interest (survivors vs. nonsurvivors) with a value of 1.0 (or 100%) would mean that the model was able to completely separate the two groups.
The model using three preoperative NSQIP variables – age, American Society of Anesthesiologists (ASA) physical status classification, and functional status – had AUROC values that were more than 0.90, or more than 90%, which was similar to the value achieved for the model that used four or six variables. The model that used 66 variables was about the same as the value achieved with the model that used 4 variables (about 91%).
Considering that an AUROC value of 0.5 indicates that the model cannot distinguish between two groups any better than chance and that an AUROC value of 1.0 indicates that the model completely discriminates between the two groups, the authors said that an AUROC value of greater than 90% is substantial. Therefore, based on their results, "3 or 4 variables may be sufficient for adequate risk adjustment to measure surgical outcomes," they added.
"Future risk-adjustment tools [should] be based on 6 or fewer variables to allow for surgical outcomes to be measured and compared within and among hospitals in resource-limited settings," they concluded.
The authors had no disclosures.
FROM THE ARCHIVES OF SURGERY
Major Finding: A risk-adjusted tool using three preoperative variables from the National Surgical Quality Improvement Program (NSQIP) database was more than 90% effective at predicting inpatient mortality – an efficacy rate comparable with that of tools using many more variables.
Data Source: The study entailed developing the tool with a different number of variables from the NSQIP database between 2005 and 2009 to predict inpatient mortality and validating the tool using patient data from a 110-bed community hospital in California.
Disclosures: The authors had no disclosures.
Simple Falls, Slips Proving Treacherous for Obese
MILWAUKEE – The obesity epidemic in the United States is bringing Americans to their knees, literally.
A growing number of obese patients are presenting to the emergency department with low-energy (LE) knee dislocations (KDs) caused by slips and falls simply from standing or from a single step.
Despite the isolated nature of their injuries, this new cohort of LE patients stayed in the hospital just as long as multisystem trauma patients with KDs resulting from high-energy injuries like car or motorcycle collisions and more than twice as long as nonobese patients with traditional low-energy knee dislocations from sports injuries.
The reason?
Obese patients with low-energy KDs are more likely to have vascular and nerve injuries and to require open arterial procedures than are patients with high-energy trauma or nonobese patients with LE knee dislocations, Dr. Andrew Georgiadis explained at the annual meeting of the Midwestern Vascular Surgical Society.
He noted that knee dislocation involves progressive hyperextension of the knee, and that, at 30 degrees of hyperextension, the posterior knee capsule is rent, and at 50 degrees, the popliteal artery actually fails.
Dr. Georgiadis and his surgical colleagues have been studying this phenomenon at Detroit’s Henry Ford Hospital, where, over a 17-year period, the proportion of low-energy KDs in the obese has risen from 17% in 1995-2000 to 33% in 2001-2006 and now represents the majority (53%) of all KDs in the hospital.
Among 53 KD patients treated between January 1995 and April 2012, 28 had high-energy injuries and 25 had low-energy injuries, of which 18 were obese and 7 nonobese. Five of the obese patients had a BMI of at least 30 kg/m2 or less than 40 kg/m2, while the remaining 13 had a BMI of more than 40 kg/m2.
When compared with the high-energy and LE non-obese patients, LE obese patients were significantly more likely to have a vascular injury (33% vs. 9%), vascular repair (28% vs. 6%), and nerve injury (50% vs. 6%), said Dr. Georgiadis, an orthopedic surgery resident at Henry Ford.
These rates were even higher in the morbidly obese (BMI over 40 kg/m2) at 39%, 39%, and 54%.
Although there were only seven arterial repairs in the entire series, five of these patients had "massive BMIs" between 42-69 kg/m2 and "they all had seemingly innocuous trauma, they all had transected arteries, they all had a vein graft bypass, and all of them had some early complication related to their procedure," he said.
Those complications included wound breakdown, early return to the operating room for a fasciotomy, graft occlusion requiring early thrombectomy/revision, and rhabdomyolysis/limb loss in a patient with a prolonged diagnosis.
When asked by the audience why the grafts thrombosed early, Dr. Georgiadis replied, "I think technical difficulty is really at the heart of all these things. And remember, these are patients who are probably being diagnosed later than someone who is crushed between two buses, so there are a lot of factors combining in these cases."
LE obese patients stayed in the hospital for an average of 8.1 days, which was comparable with the 11.4 days in the high-energy KD patients, of whom only 43% had isolated injuries, and significantly longer than the 3.7 days for non-obese LE patients, he said.
Given the increasing prevalence of obese low-energy KD patients, "we should probably have heightened awareness of this injury, especially at tertiary care centers, to avoid the morbidity of neurovascular injury and the consequences of delayed recognition," Dr. Georgiadis said.
A quick glimpse at the literature suggests that the ever-expanding American is not the only one at risk of obesity-related KDs.
Four cases of knee dislocation were recently reported by Morriston Hospital in Swansea, Wales – all in morbidly obese women (BMI range of 35-41) who experienced a simple mechanical fall from standing. The four cases occurred over the course of 1 year, and all had multiple knee ligament rupture on MRI. One case also had peroneal nerve palsy, according to the article, entitled "Dislocation of the Knee: An Epidemic in Waiting?"
Dr. Georgiadis reported no conflicts of interest.
MILWAUKEE – The obesity epidemic in the United States is bringing Americans to their knees, literally.
A growing number of obese patients are presenting to the emergency department with low-energy (LE) knee dislocations (KDs) caused by slips and falls simply from standing or from a single step.
Despite the isolated nature of their injuries, this new cohort of LE patients stayed in the hospital just as long as multisystem trauma patients with KDs resulting from high-energy injuries like car or motorcycle collisions and more than twice as long as nonobese patients with traditional low-energy knee dislocations from sports injuries.
The reason?
Obese patients with low-energy KDs are more likely to have vascular and nerve injuries and to require open arterial procedures than are patients with high-energy trauma or nonobese patients with LE knee dislocations, Dr. Andrew Georgiadis explained at the annual meeting of the Midwestern Vascular Surgical Society.
He noted that knee dislocation involves progressive hyperextension of the knee, and that, at 30 degrees of hyperextension, the posterior knee capsule is rent, and at 50 degrees, the popliteal artery actually fails.
Dr. Georgiadis and his surgical colleagues have been studying this phenomenon at Detroit’s Henry Ford Hospital, where, over a 17-year period, the proportion of low-energy KDs in the obese has risen from 17% in 1995-2000 to 33% in 2001-2006 and now represents the majority (53%) of all KDs in the hospital.
Among 53 KD patients treated between January 1995 and April 2012, 28 had high-energy injuries and 25 had low-energy injuries, of which 18 were obese and 7 nonobese. Five of the obese patients had a BMI of at least 30 kg/m2 or less than 40 kg/m2, while the remaining 13 had a BMI of more than 40 kg/m2.
When compared with the high-energy and LE non-obese patients, LE obese patients were significantly more likely to have a vascular injury (33% vs. 9%), vascular repair (28% vs. 6%), and nerve injury (50% vs. 6%), said Dr. Georgiadis, an orthopedic surgery resident at Henry Ford.
These rates were even higher in the morbidly obese (BMI over 40 kg/m2) at 39%, 39%, and 54%.
Although there were only seven arterial repairs in the entire series, five of these patients had "massive BMIs" between 42-69 kg/m2 and "they all had seemingly innocuous trauma, they all had transected arteries, they all had a vein graft bypass, and all of them had some early complication related to their procedure," he said.
Those complications included wound breakdown, early return to the operating room for a fasciotomy, graft occlusion requiring early thrombectomy/revision, and rhabdomyolysis/limb loss in a patient with a prolonged diagnosis.
When asked by the audience why the grafts thrombosed early, Dr. Georgiadis replied, "I think technical difficulty is really at the heart of all these things. And remember, these are patients who are probably being diagnosed later than someone who is crushed between two buses, so there are a lot of factors combining in these cases."
LE obese patients stayed in the hospital for an average of 8.1 days, which was comparable with the 11.4 days in the high-energy KD patients, of whom only 43% had isolated injuries, and significantly longer than the 3.7 days for non-obese LE patients, he said.
Given the increasing prevalence of obese low-energy KD patients, "we should probably have heightened awareness of this injury, especially at tertiary care centers, to avoid the morbidity of neurovascular injury and the consequences of delayed recognition," Dr. Georgiadis said.
A quick glimpse at the literature suggests that the ever-expanding American is not the only one at risk of obesity-related KDs.
Four cases of knee dislocation were recently reported by Morriston Hospital in Swansea, Wales – all in morbidly obese women (BMI range of 35-41) who experienced a simple mechanical fall from standing. The four cases occurred over the course of 1 year, and all had multiple knee ligament rupture on MRI. One case also had peroneal nerve palsy, according to the article, entitled "Dislocation of the Knee: An Epidemic in Waiting?"
Dr. Georgiadis reported no conflicts of interest.
MILWAUKEE – The obesity epidemic in the United States is bringing Americans to their knees, literally.
A growing number of obese patients are presenting to the emergency department with low-energy (LE) knee dislocations (KDs) caused by slips and falls simply from standing or from a single step.
Despite the isolated nature of their injuries, this new cohort of LE patients stayed in the hospital just as long as multisystem trauma patients with KDs resulting from high-energy injuries like car or motorcycle collisions and more than twice as long as nonobese patients with traditional low-energy knee dislocations from sports injuries.
The reason?
Obese patients with low-energy KDs are more likely to have vascular and nerve injuries and to require open arterial procedures than are patients with high-energy trauma or nonobese patients with LE knee dislocations, Dr. Andrew Georgiadis explained at the annual meeting of the Midwestern Vascular Surgical Society.
He noted that knee dislocation involves progressive hyperextension of the knee, and that, at 30 degrees of hyperextension, the posterior knee capsule is rent, and at 50 degrees, the popliteal artery actually fails.
Dr. Georgiadis and his surgical colleagues have been studying this phenomenon at Detroit’s Henry Ford Hospital, where, over a 17-year period, the proportion of low-energy KDs in the obese has risen from 17% in 1995-2000 to 33% in 2001-2006 and now represents the majority (53%) of all KDs in the hospital.
Among 53 KD patients treated between January 1995 and April 2012, 28 had high-energy injuries and 25 had low-energy injuries, of which 18 were obese and 7 nonobese. Five of the obese patients had a BMI of at least 30 kg/m2 or less than 40 kg/m2, while the remaining 13 had a BMI of more than 40 kg/m2.
When compared with the high-energy and LE non-obese patients, LE obese patients were significantly more likely to have a vascular injury (33% vs. 9%), vascular repair (28% vs. 6%), and nerve injury (50% vs. 6%), said Dr. Georgiadis, an orthopedic surgery resident at Henry Ford.
These rates were even higher in the morbidly obese (BMI over 40 kg/m2) at 39%, 39%, and 54%.
Although there were only seven arterial repairs in the entire series, five of these patients had "massive BMIs" between 42-69 kg/m2 and "they all had seemingly innocuous trauma, they all had transected arteries, they all had a vein graft bypass, and all of them had some early complication related to their procedure," he said.
Those complications included wound breakdown, early return to the operating room for a fasciotomy, graft occlusion requiring early thrombectomy/revision, and rhabdomyolysis/limb loss in a patient with a prolonged diagnosis.
When asked by the audience why the grafts thrombosed early, Dr. Georgiadis replied, "I think technical difficulty is really at the heart of all these things. And remember, these are patients who are probably being diagnosed later than someone who is crushed between two buses, so there are a lot of factors combining in these cases."
LE obese patients stayed in the hospital for an average of 8.1 days, which was comparable with the 11.4 days in the high-energy KD patients, of whom only 43% had isolated injuries, and significantly longer than the 3.7 days for non-obese LE patients, he said.
Given the increasing prevalence of obese low-energy KD patients, "we should probably have heightened awareness of this injury, especially at tertiary care centers, to avoid the morbidity of neurovascular injury and the consequences of delayed recognition," Dr. Georgiadis said.
A quick glimpse at the literature suggests that the ever-expanding American is not the only one at risk of obesity-related KDs.
Four cases of knee dislocation were recently reported by Morriston Hospital in Swansea, Wales – all in morbidly obese women (BMI range of 35-41) who experienced a simple mechanical fall from standing. The four cases occurred over the course of 1 year, and all had multiple knee ligament rupture on MRI. One case also had peroneal nerve palsy, according to the article, entitled "Dislocation of the Knee: An Epidemic in Waiting?"
Dr. Georgiadis reported no conflicts of interest.
EXPERT ANALYSIS FROM THE ANNUAL MEETING OF THE MIDWESTERN VASCULAR SURGICAL SOCIETY
Myelomeningocele Repair Drives Changes in Fetal Surgery
Publication of results from the landmark Management of Myelomeningocele Study last year established fetal surgery as a viable option, and possibly the best option for ameliorating the complications of spina bifida.
But the Management of Myelomeningocele Study (MOMS) also appears to have had farther-reaching effects. In the 18 months since the trial results appeared (N. Engl. J. Med. 2011;364:993-1004), myelomeningocele (MMC) repair in fetuses repositioned from investigational surgery performed at just three U.S. centers to arguably standard of care that could potentially be done at whichever centers gear up to offer it. Beyond that, the possibility of effective and reasonably safe fetal MMC repair that the MOMS results documented is also proving to be a catalyst in the transformation of the still-young field of fetal surgery from an investigational, niche specialty to a more mainstream intervention.
"For many of the other [fetal surgery] interventions, there are so few cases that having more than a few centers doing them did not make sense. To have a fetal center you need a dedicated, multidisciplinary team available 24/7/365 that does a certain number of cases per year to keep up its team’s skills. For fetal surgery to be successful, it will need to be regionalized so that there is access for all patients, but also focused at centers of excellence so there is enough volume to keep skills sharp," said Dr. Mark P. Johnson, obstetrical director of the Center for Fetal Diagnosis and Treatment at the Children’s Hospital of Philadelphia (CHOP).
The MOMS result "has already had a profound effect on the treatment of MMC, on reimbursement standards for fetal intervention, and has defined how fetal surgery centers should be organized and staffed across the United States," wrote Dr. Shinjiro Hirose, a fetal surgeon at the University of California, San Francisco (UCSF), and his associates in an article on maternal-fetal surgery that appeared in June (Clin. Perinatology 2012;39:269-78).
Growing fetal-intervention options also put unprecedented responsibility on the physicians who provide primary obstetrical care to identify pregnancies early that may have an anomaly that’s amenable to surgical intervention and make an appropriate referral so that the disorder can be confirmed and intervention offered if it is possible.
Growing Numbers of MMC Repairs and Other Fetal Surgeries
Although the exact number of fetal MMC repairs done in the United States during the 18 months since publication of the MOMS results is hard to pin down, experts estimate roughly 80 surgeries occurred during April 2011-March 2012. They project as many as 100 or more being done in 2012, with about eight U.S. centers now offering the surgery, including the three centers that participated in MOMS and another five or so that have begun performing the procedure since the results were announced.
The other "high volume" fetal surgery now done is fetoscopic laser ablation of communicating vessels on the placental chorionic plate in monochorionic twin pregnancies that develop Twin-Twin transfusion syndrome (TTTS), which first became established as standard of care in 2004 (N. Engl. J. Med. 2004;351:136-44). It is now widely accepted as the preferred option in these cases, done on upward of 500 U.S. pregnancies a year. When the annualized rate of 600+ MMC repairs and laser ablations for TTTS couples with various low-volume fetal surgeries done for selected, rare anomalies, U.S. fetal surgery is on track this year to treat some 700-1,000 cases, and this number will likely rise substantially in the near future. The current, annual U.S. incidence of spina bifida in fetuses is about 2,500 (with about 1,500 infants born with MMC each year), suggesting that as fetal MMC repair becomes more widely accepted and performed, the number of U.S. surgeries for this indication could eventually run into several hundred a year.
Planning for Fetal MMC Repairs After MOMS
Driven primarily by concerns about how the MOMS results would translate into routine practice, the Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD), the agency that sponsored MOMS, organized an expert panel with representatives from 11 U.S. professional and patient groups to come up with recommendations on which centers should perform fetal MMC repairs and the clinical issues that the teams at each center should address. Participants came from the American College of Obstetricians and Gynecologists, the American Academy of Pediatrics, the American Institute of Ultrasound in Medicine, the Society for Maternal-Fetal Medicine, the Spina Bifida Association, and six other groups.
Formal publication of these recommendations had not occurred yet but was expected before the end of 2012. The article by Dr. Hirose and his coauthors provided a preview of some of the panel’s key recommendations, including a minimum annual volume of at least five MMC repairs as well as at least 30 fetuses with MMC evaluated for surgery; strict adherence to the MOMS protocol until improvements are proven better; comprehensive counseling that includes a reflective period for families of at least 24 hours; and participation in a national registry.
That article was removed from Clinics in Perinatology in early October at the request of the publication’s consulting editor.
According to a note on the publication’s website, "It was learned after publication that the guidelines for fetal repair of myelomeningocele that were presented as being published by the NIH were neither supplied by the NIH nor published by them. To prevent this information from being erroneously cited, the article has been removed."
At press time, the article was still accessible via the National Library of Medicine’s PubMed.gov.*
Having a U.S. agency sponsor a major trial and then organize an expert panel to deliberate on how to best implement its results is unusual, but fetal MMC repair is unusual surgery, said Dr. Julie S. Moldenhauer, a maternal-fetal medicine physician and a representative to the NICHD panel appointed by the North American Fetal Therapy Network. "It is very high-risk surgery that is elective, it is an open procedure, not fetoscopic, and it depends on a very multidisciplinary team," she said in an interview. These are a combination of features that makes it unique, said Dr. Moldenhauer, who is an attending physician at the Center for Fetal Diagnosis and Treatment and the medical director of the special delivery unit, both at CHOP.
One other element of MOMS also adds to its special character: The trial lasted nearly 8 years and randomized just 183 patients, and during that time fetal MMC repair for U.S. patients was limited to three centers – CHOP, UCSF, and Vanderbilt University in Nashville, Tenn. Then, when the researchers reported their positive results last year, NICHD officials and others in this field faced the dilemma of how to make the surgery available elsewhere while ensuring that the results would be as good as in MOMS. "What the NICHD fears is that if MMC repair is offered uncontrolled, the outcomes won’t be what they were in MOMS," said Dr. Johnson.
Making Fetal MMC Repair Better
MMC repair is not easy and still has limitations, most notably the risk for extreme prematurity. "The risk of prematurity is the big obstacle to making it more mainstream," said Dr. Noel B. Tulipan, professor and director of pediatric neurosurgery at Vanderbilt University Medical Center.
Most fetuses treated in MOMS were delivered at 34 weeks’ gestation or later, and "these days, we almost don’t consider 34 weeks premature, because most of these babies do extremely well," Dr. Tulipan said in an interview. "But in MOMS, there was a 13% rate of extreme prematurity" with the infants delivered at less than 30 weeks.
This limitation is improving. The challenge, he said, is the incision and closure of the uterus. Dr. Tulipan and his colleagues have recently given increased attention to suturing the amniotic membranes to prevent them from separating from the uterine wall "With this change, our results have gotten substantially better. We’re getting to a rate that could make it mainstream. If the [extreme] prematurity rate was less than 5%, it would be hard to argue against this surgery."
Fetal MMC repair "will continue to grow, but not by an order of magnitude until there is a new technique that could be offered earlier in gestation and in a minimally-invasive way," said Dr. N. Scott Adzick, surgeon in chief and director of the Center for Fetal Diagnosis and Treatment at CHOP. Currently, MMC repairs are done at 20 weeks’-25 weeks’ 6 days gestation. His group at CHOP, as well as others, is also trying to perfect less-invasive approaches. "We are working on a tissue-engineering approach to seal the MMC defect before birth, and thus prevent exposure of the spinal cord to damaging amniotic fluid, and also prevent leakage of the cerebrospinal fluid from the spina bifida. CSF leakage is the underlying cause of hindbrain herniation and hydrocephalus seen with spina bifida. The goal is for this tissue-engineered component to be introduced through a single fetoscopic port or through an amniocentesis needle under sonographic guidance," Dr. Adzick said.
The risk of prematurity, as well as potential complications for the mother, means that centers offering fetal MMC repair provide extensive counseling for potential parents. "We are proud of the fact that over half of our families decide not to have prenatal surgery after the counseling," said Dr. Hanmin Lee, professor and chief of pediatric surgery at UCSF, and one of the leaders of that center’s MMC fetal repair program.
At CHOP, during the first year after the MOMS publication 238 pregnancies underwent evaluation, with 137 making it to a more extensive stage of assessment. Of these, 40 mothers (29%) underwent fetal MMC repair, Dr. Adzick said.
At Cincinnati Children’s Hospital, which began offering fetal MMC repairs following publication of the MOMS results, the winnowing was about as sharp as at CHOP. Since the program began, the Cincinnati team evaluated 53 pregnancies and performed 10 repairs, said Dr. Foong-Yen Lim, surgical director of the Fetal Care Center there. A handful of these patients who were first seen at Cincinnati decided to travel to Vanderbilt or CHOP instead for the surgery and thereby take advantage of the greater experience those centers offered.
That sentiment underscores a challenge faced by the programs that are trying to establish themselves as new options for fetal MMC repair, as the procedure rolls out post-MOMS.
"There is nothing wrong with patients going to a higher-volume center; they know that their outcome will be close to what is available today, compared with new centers that have not done as many of these procedures," Dr. Lim said in an interview. "Patients should hear the options and pick what’s best for them, not just whether to choose the procedure, but also to decide where to go. The reason why MOMS had such difficulty recruiting patients was that they had to uproot themselves to go to a MMC repair center, and a lot of patients decided not to go.
"Regionalization is the future. Spina bifida is one of the most common birth defects, so there is a large number of cases that could benefit" from fetal repair. But what does it mean to be a fetal surgery center, and what does it require," said CHOP’s Dr. Johnson. "These are the ideas that the fetal surgery community is struggling with and trying to address. The big question is which will be the centers of excellence. The first step toward answering this will be to see whether the new centers can replicate the MOMS results, he added.
Spreading the Word About Fetal Surgery
Another key element in growing fetal MMC repair, and fetal surgeries of all types, is boosting awareness of repair options among primary care obstetrical providers and making referrals more timely.
"The market is controlled by the number of physicians who are aware of surgical interventions," said Dr. Mark I. Evans, a clinical professor of obstetrics, gynecology, and reproductive services at Mount Sinai Medical Center in New York. "The number of anomalies that are potentially amenable to treatment is a small percent of all pregnancies, but with more than 4 million U.S. births per year, it adds up to a fairly substantial number."
Fetal interventions "are accepted therapies; in the fetal community, I don’t think anyone thinks they are still investigational. But the number of cases is small, and a lot of the people who are diagnosing these may not be aware of how successful a lot of the procedures are," said Dr. Johnson.
"Obstetricians and maternal-fetal medicine physicians need to be more aware of what we can do. Our biggest challenge is to disseminate the information. The treatments are safe and effective, and families should at least have the option of going to centers to get more information and make informed decisions."
In addition, "screening ultrasound at between 18 to 20 weeks would be a great thing," he added.
"In the United States, ultrasound screening is becoming more the standard of care, but a lot of screening is not done until 22 or 23 weeks, often too late for patients to be referred to fetal treatment centers for evaluations before 24 weeks so that the option of termination is often no longer available to them. Also, with some anomalies irreversible damage has already occurred by 24 weeks so that fetal therapy would not help. In cases of lower urinary tract obstructions, well over half the cases we see are too late to offer therapy. It is recognized that screening at 19-20 weeks won’t pick up all anomalies, but it would pick up a lot of major anomalies," Dr. Johnson said.
A Fetal Surgery Checklist
Aside from myelomeningocele repair, placental laser ablation in cases of severe twin-twin transfusion syndrome is the fetal surgery with the best evidence base and, for now, it’s also the most frequently performed fetal surgery, although experts say it’s also underused.
"The success story of fetal surgery is laser ablation for TTTS," said Dr. Francois I. Luks, director of the Fetal Treatment Program at Hasbro Children’s Hospital in Providence, R.I. "Left untreated, severe TTTS leads to greater than 80% dual mortality. With laser surgery, survival of at least one twin is in excess of 80%."
First established as a good alternative to standard amnioreduction therapy in a 2004 randomized trial (N. Engl. J. Med. 2004;351:136-44), placental laser ablation has since become standard-of-care for TTTS.
"More and more groups are taking [laser ablation for TTTS] on because they believe they should provide it to patients," and it currently is available at about 30 U.S. centers, said Dr. Lim. "The maternal risk [from placental laser ablation] is acceptable and the outcomes are quite good. But some cases [of TTTS] are still being misdiagnosed," he said.
"Awareness is more widespread, but not at the rate it should be. We still have patients who are not referred until something bad happens," Dr. Lim said in an interview.
In Cincinnati alone, the program does more than 100 placental laser ablations each year, he added.
About 80 per year are performed at CHOP, and roughly 300-400 per year total at the 24 U.S. and Canadian centers that form the North American Fetal Therapy Network (NAFTNet), said Dr. Luks. (CHOP and Cincinnati Children’s are members of NAFTNet.) At least two other U.S. centers not in NAFTNet do more than 100 laser ablations each annually, and several other hospitals outside of NAFTNet do smaller numbers yearly, which means that while the current annual U.S. volume of these cases is uncertain it easily exceeds 500, Dr. Luks said.
All the other fetal surgeries each occur in fewer than about 50 U.S. cases a year, are available at fewer U.S. centers and, in some cases, have a checkered history of success and failure although today several are considered effective, relatively safe, and standard therapy.
The third most-common fetal surgery, based on NAFTNet records, are various types of selective umbilical cord occlusions in twin pregnancies, for reasons such as intrauterine growth retardation, placental insufficiency, and other situations in which problems with one twin puts a healthy twin at risk, said Dr. Johnson.
Dr. Johnson, Dr. Hirose, Dr. Adzick, Dr. Moldenhauer, Dr. Lee, Dr. Tulipan, Dr. Lim, and Dr. Evans all said that they had no relevant financial disclosures.
* This story was updated on 10/3/1012.
Publication of results from the landmark Management of Myelomeningocele Study last year established fetal surgery as a viable option, and possibly the best option for ameliorating the complications of spina bifida.
But the Management of Myelomeningocele Study (MOMS) also appears to have had farther-reaching effects. In the 18 months since the trial results appeared (N. Engl. J. Med. 2011;364:993-1004), myelomeningocele (MMC) repair in fetuses repositioned from investigational surgery performed at just three U.S. centers to arguably standard of care that could potentially be done at whichever centers gear up to offer it. Beyond that, the possibility of effective and reasonably safe fetal MMC repair that the MOMS results documented is also proving to be a catalyst in the transformation of the still-young field of fetal surgery from an investigational, niche specialty to a more mainstream intervention.
"For many of the other [fetal surgery] interventions, there are so few cases that having more than a few centers doing them did not make sense. To have a fetal center you need a dedicated, multidisciplinary team available 24/7/365 that does a certain number of cases per year to keep up its team’s skills. For fetal surgery to be successful, it will need to be regionalized so that there is access for all patients, but also focused at centers of excellence so there is enough volume to keep skills sharp," said Dr. Mark P. Johnson, obstetrical director of the Center for Fetal Diagnosis and Treatment at the Children’s Hospital of Philadelphia (CHOP).
The MOMS result "has already had a profound effect on the treatment of MMC, on reimbursement standards for fetal intervention, and has defined how fetal surgery centers should be organized and staffed across the United States," wrote Dr. Shinjiro Hirose, a fetal surgeon at the University of California, San Francisco (UCSF), and his associates in an article on maternal-fetal surgery that appeared in June (Clin. Perinatology 2012;39:269-78).
Growing fetal-intervention options also put unprecedented responsibility on the physicians who provide primary obstetrical care to identify pregnancies early that may have an anomaly that’s amenable to surgical intervention and make an appropriate referral so that the disorder can be confirmed and intervention offered if it is possible.
Growing Numbers of MMC Repairs and Other Fetal Surgeries
Although the exact number of fetal MMC repairs done in the United States during the 18 months since publication of the MOMS results is hard to pin down, experts estimate roughly 80 surgeries occurred during April 2011-March 2012. They project as many as 100 or more being done in 2012, with about eight U.S. centers now offering the surgery, including the three centers that participated in MOMS and another five or so that have begun performing the procedure since the results were announced.
The other "high volume" fetal surgery now done is fetoscopic laser ablation of communicating vessels on the placental chorionic plate in monochorionic twin pregnancies that develop Twin-Twin transfusion syndrome (TTTS), which first became established as standard of care in 2004 (N. Engl. J. Med. 2004;351:136-44). It is now widely accepted as the preferred option in these cases, done on upward of 500 U.S. pregnancies a year. When the annualized rate of 600+ MMC repairs and laser ablations for TTTS couples with various low-volume fetal surgeries done for selected, rare anomalies, U.S. fetal surgery is on track this year to treat some 700-1,000 cases, and this number will likely rise substantially in the near future. The current, annual U.S. incidence of spina bifida in fetuses is about 2,500 (with about 1,500 infants born with MMC each year), suggesting that as fetal MMC repair becomes more widely accepted and performed, the number of U.S. surgeries for this indication could eventually run into several hundred a year.
Planning for Fetal MMC Repairs After MOMS
Driven primarily by concerns about how the MOMS results would translate into routine practice, the Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD), the agency that sponsored MOMS, organized an expert panel with representatives from 11 U.S. professional and patient groups to come up with recommendations on which centers should perform fetal MMC repairs and the clinical issues that the teams at each center should address. Participants came from the American College of Obstetricians and Gynecologists, the American Academy of Pediatrics, the American Institute of Ultrasound in Medicine, the Society for Maternal-Fetal Medicine, the Spina Bifida Association, and six other groups.
Formal publication of these recommendations had not occurred yet but was expected before the end of 2012. The article by Dr. Hirose and his coauthors provided a preview of some of the panel’s key recommendations, including a minimum annual volume of at least five MMC repairs as well as at least 30 fetuses with MMC evaluated for surgery; strict adherence to the MOMS protocol until improvements are proven better; comprehensive counseling that includes a reflective period for families of at least 24 hours; and participation in a national registry.
That article was removed from Clinics in Perinatology in early October at the request of the publication’s consulting editor.
According to a note on the publication’s website, "It was learned after publication that the guidelines for fetal repair of myelomeningocele that were presented as being published by the NIH were neither supplied by the NIH nor published by them. To prevent this information from being erroneously cited, the article has been removed."
At press time, the article was still accessible via the National Library of Medicine’s PubMed.gov.*
Having a U.S. agency sponsor a major trial and then organize an expert panel to deliberate on how to best implement its results is unusual, but fetal MMC repair is unusual surgery, said Dr. Julie S. Moldenhauer, a maternal-fetal medicine physician and a representative to the NICHD panel appointed by the North American Fetal Therapy Network. "It is very high-risk surgery that is elective, it is an open procedure, not fetoscopic, and it depends on a very multidisciplinary team," she said in an interview. These are a combination of features that makes it unique, said Dr. Moldenhauer, who is an attending physician at the Center for Fetal Diagnosis and Treatment and the medical director of the special delivery unit, both at CHOP.
One other element of MOMS also adds to its special character: The trial lasted nearly 8 years and randomized just 183 patients, and during that time fetal MMC repair for U.S. patients was limited to three centers – CHOP, UCSF, and Vanderbilt University in Nashville, Tenn. Then, when the researchers reported their positive results last year, NICHD officials and others in this field faced the dilemma of how to make the surgery available elsewhere while ensuring that the results would be as good as in MOMS. "What the NICHD fears is that if MMC repair is offered uncontrolled, the outcomes won’t be what they were in MOMS," said Dr. Johnson.
Making Fetal MMC Repair Better
MMC repair is not easy and still has limitations, most notably the risk for extreme prematurity. "The risk of prematurity is the big obstacle to making it more mainstream," said Dr. Noel B. Tulipan, professor and director of pediatric neurosurgery at Vanderbilt University Medical Center.
Most fetuses treated in MOMS were delivered at 34 weeks’ gestation or later, and "these days, we almost don’t consider 34 weeks premature, because most of these babies do extremely well," Dr. Tulipan said in an interview. "But in MOMS, there was a 13% rate of extreme prematurity" with the infants delivered at less than 30 weeks.
This limitation is improving. The challenge, he said, is the incision and closure of the uterus. Dr. Tulipan and his colleagues have recently given increased attention to suturing the amniotic membranes to prevent them from separating from the uterine wall "With this change, our results have gotten substantially better. We’re getting to a rate that could make it mainstream. If the [extreme] prematurity rate was less than 5%, it would be hard to argue against this surgery."
Fetal MMC repair "will continue to grow, but not by an order of magnitude until there is a new technique that could be offered earlier in gestation and in a minimally-invasive way," said Dr. N. Scott Adzick, surgeon in chief and director of the Center for Fetal Diagnosis and Treatment at CHOP. Currently, MMC repairs are done at 20 weeks’-25 weeks’ 6 days gestation. His group at CHOP, as well as others, is also trying to perfect less-invasive approaches. "We are working on a tissue-engineering approach to seal the MMC defect before birth, and thus prevent exposure of the spinal cord to damaging amniotic fluid, and also prevent leakage of the cerebrospinal fluid from the spina bifida. CSF leakage is the underlying cause of hindbrain herniation and hydrocephalus seen with spina bifida. The goal is for this tissue-engineered component to be introduced through a single fetoscopic port or through an amniocentesis needle under sonographic guidance," Dr. Adzick said.
The risk of prematurity, as well as potential complications for the mother, means that centers offering fetal MMC repair provide extensive counseling for potential parents. "We are proud of the fact that over half of our families decide not to have prenatal surgery after the counseling," said Dr. Hanmin Lee, professor and chief of pediatric surgery at UCSF, and one of the leaders of that center’s MMC fetal repair program.
At CHOP, during the first year after the MOMS publication 238 pregnancies underwent evaluation, with 137 making it to a more extensive stage of assessment. Of these, 40 mothers (29%) underwent fetal MMC repair, Dr. Adzick said.
At Cincinnati Children’s Hospital, which began offering fetal MMC repairs following publication of the MOMS results, the winnowing was about as sharp as at CHOP. Since the program began, the Cincinnati team evaluated 53 pregnancies and performed 10 repairs, said Dr. Foong-Yen Lim, surgical director of the Fetal Care Center there. A handful of these patients who were first seen at Cincinnati decided to travel to Vanderbilt or CHOP instead for the surgery and thereby take advantage of the greater experience those centers offered.
That sentiment underscores a challenge faced by the programs that are trying to establish themselves as new options for fetal MMC repair, as the procedure rolls out post-MOMS.
"There is nothing wrong with patients going to a higher-volume center; they know that their outcome will be close to what is available today, compared with new centers that have not done as many of these procedures," Dr. Lim said in an interview. "Patients should hear the options and pick what’s best for them, not just whether to choose the procedure, but also to decide where to go. The reason why MOMS had such difficulty recruiting patients was that they had to uproot themselves to go to a MMC repair center, and a lot of patients decided not to go.
"Regionalization is the future. Spina bifida is one of the most common birth defects, so there is a large number of cases that could benefit" from fetal repair. But what does it mean to be a fetal surgery center, and what does it require," said CHOP’s Dr. Johnson. "These are the ideas that the fetal surgery community is struggling with and trying to address. The big question is which will be the centers of excellence. The first step toward answering this will be to see whether the new centers can replicate the MOMS results, he added.
Spreading the Word About Fetal Surgery
Another key element in growing fetal MMC repair, and fetal surgeries of all types, is boosting awareness of repair options among primary care obstetrical providers and making referrals more timely.
"The market is controlled by the number of physicians who are aware of surgical interventions," said Dr. Mark I. Evans, a clinical professor of obstetrics, gynecology, and reproductive services at Mount Sinai Medical Center in New York. "The number of anomalies that are potentially amenable to treatment is a small percent of all pregnancies, but with more than 4 million U.S. births per year, it adds up to a fairly substantial number."
Fetal interventions "are accepted therapies; in the fetal community, I don’t think anyone thinks they are still investigational. But the number of cases is small, and a lot of the people who are diagnosing these may not be aware of how successful a lot of the procedures are," said Dr. Johnson.
"Obstetricians and maternal-fetal medicine physicians need to be more aware of what we can do. Our biggest challenge is to disseminate the information. The treatments are safe and effective, and families should at least have the option of going to centers to get more information and make informed decisions."
In addition, "screening ultrasound at between 18 to 20 weeks would be a great thing," he added.
"In the United States, ultrasound screening is becoming more the standard of care, but a lot of screening is not done until 22 or 23 weeks, often too late for patients to be referred to fetal treatment centers for evaluations before 24 weeks so that the option of termination is often no longer available to them. Also, with some anomalies irreversible damage has already occurred by 24 weeks so that fetal therapy would not help. In cases of lower urinary tract obstructions, well over half the cases we see are too late to offer therapy. It is recognized that screening at 19-20 weeks won’t pick up all anomalies, but it would pick up a lot of major anomalies," Dr. Johnson said.
A Fetal Surgery Checklist
Aside from myelomeningocele repair, placental laser ablation in cases of severe twin-twin transfusion syndrome is the fetal surgery with the best evidence base and, for now, it’s also the most frequently performed fetal surgery, although experts say it’s also underused.
"The success story of fetal surgery is laser ablation for TTTS," said Dr. Francois I. Luks, director of the Fetal Treatment Program at Hasbro Children’s Hospital in Providence, R.I. "Left untreated, severe TTTS leads to greater than 80% dual mortality. With laser surgery, survival of at least one twin is in excess of 80%."
First established as a good alternative to standard amnioreduction therapy in a 2004 randomized trial (N. Engl. J. Med. 2004;351:136-44), placental laser ablation has since become standard-of-care for TTTS.
"More and more groups are taking [laser ablation for TTTS] on because they believe they should provide it to patients," and it currently is available at about 30 U.S. centers, said Dr. Lim. "The maternal risk [from placental laser ablation] is acceptable and the outcomes are quite good. But some cases [of TTTS] are still being misdiagnosed," he said.
"Awareness is more widespread, but not at the rate it should be. We still have patients who are not referred until something bad happens," Dr. Lim said in an interview.
In Cincinnati alone, the program does more than 100 placental laser ablations each year, he added.
About 80 per year are performed at CHOP, and roughly 300-400 per year total at the 24 U.S. and Canadian centers that form the North American Fetal Therapy Network (NAFTNet), said Dr. Luks. (CHOP and Cincinnati Children’s are members of NAFTNet.) At least two other U.S. centers not in NAFTNet do more than 100 laser ablations each annually, and several other hospitals outside of NAFTNet do smaller numbers yearly, which means that while the current annual U.S. volume of these cases is uncertain it easily exceeds 500, Dr. Luks said.
All the other fetal surgeries each occur in fewer than about 50 U.S. cases a year, are available at fewer U.S. centers and, in some cases, have a checkered history of success and failure although today several are considered effective, relatively safe, and standard therapy.
The third most-common fetal surgery, based on NAFTNet records, are various types of selective umbilical cord occlusions in twin pregnancies, for reasons such as intrauterine growth retardation, placental insufficiency, and other situations in which problems with one twin puts a healthy twin at risk, said Dr. Johnson.
Dr. Johnson, Dr. Hirose, Dr. Adzick, Dr. Moldenhauer, Dr. Lee, Dr. Tulipan, Dr. Lim, and Dr. Evans all said that they had no relevant financial disclosures.
* This story was updated on 10/3/1012.
Publication of results from the landmark Management of Myelomeningocele Study last year established fetal surgery as a viable option, and possibly the best option for ameliorating the complications of spina bifida.
But the Management of Myelomeningocele Study (MOMS) also appears to have had farther-reaching effects. In the 18 months since the trial results appeared (N. Engl. J. Med. 2011;364:993-1004), myelomeningocele (MMC) repair in fetuses repositioned from investigational surgery performed at just three U.S. centers to arguably standard of care that could potentially be done at whichever centers gear up to offer it. Beyond that, the possibility of effective and reasonably safe fetal MMC repair that the MOMS results documented is also proving to be a catalyst in the transformation of the still-young field of fetal surgery from an investigational, niche specialty to a more mainstream intervention.
"For many of the other [fetal surgery] interventions, there are so few cases that having more than a few centers doing them did not make sense. To have a fetal center you need a dedicated, multidisciplinary team available 24/7/365 that does a certain number of cases per year to keep up its team’s skills. For fetal surgery to be successful, it will need to be regionalized so that there is access for all patients, but also focused at centers of excellence so there is enough volume to keep skills sharp," said Dr. Mark P. Johnson, obstetrical director of the Center for Fetal Diagnosis and Treatment at the Children’s Hospital of Philadelphia (CHOP).
The MOMS result "has already had a profound effect on the treatment of MMC, on reimbursement standards for fetal intervention, and has defined how fetal surgery centers should be organized and staffed across the United States," wrote Dr. Shinjiro Hirose, a fetal surgeon at the University of California, San Francisco (UCSF), and his associates in an article on maternal-fetal surgery that appeared in June (Clin. Perinatology 2012;39:269-78).
Growing fetal-intervention options also put unprecedented responsibility on the physicians who provide primary obstetrical care to identify pregnancies early that may have an anomaly that’s amenable to surgical intervention and make an appropriate referral so that the disorder can be confirmed and intervention offered if it is possible.
Growing Numbers of MMC Repairs and Other Fetal Surgeries
Although the exact number of fetal MMC repairs done in the United States during the 18 months since publication of the MOMS results is hard to pin down, experts estimate roughly 80 surgeries occurred during April 2011-March 2012. They project as many as 100 or more being done in 2012, with about eight U.S. centers now offering the surgery, including the three centers that participated in MOMS and another five or so that have begun performing the procedure since the results were announced.
The other "high volume" fetal surgery now done is fetoscopic laser ablation of communicating vessels on the placental chorionic plate in monochorionic twin pregnancies that develop Twin-Twin transfusion syndrome (TTTS), which first became established as standard of care in 2004 (N. Engl. J. Med. 2004;351:136-44). It is now widely accepted as the preferred option in these cases, done on upward of 500 U.S. pregnancies a year. When the annualized rate of 600+ MMC repairs and laser ablations for TTTS couples with various low-volume fetal surgeries done for selected, rare anomalies, U.S. fetal surgery is on track this year to treat some 700-1,000 cases, and this number will likely rise substantially in the near future. The current, annual U.S. incidence of spina bifida in fetuses is about 2,500 (with about 1,500 infants born with MMC each year), suggesting that as fetal MMC repair becomes more widely accepted and performed, the number of U.S. surgeries for this indication could eventually run into several hundred a year.
Planning for Fetal MMC Repairs After MOMS
Driven primarily by concerns about how the MOMS results would translate into routine practice, the Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD), the agency that sponsored MOMS, organized an expert panel with representatives from 11 U.S. professional and patient groups to come up with recommendations on which centers should perform fetal MMC repairs and the clinical issues that the teams at each center should address. Participants came from the American College of Obstetricians and Gynecologists, the American Academy of Pediatrics, the American Institute of Ultrasound in Medicine, the Society for Maternal-Fetal Medicine, the Spina Bifida Association, and six other groups.
Formal publication of these recommendations had not occurred yet but was expected before the end of 2012. The article by Dr. Hirose and his coauthors provided a preview of some of the panel’s key recommendations, including a minimum annual volume of at least five MMC repairs as well as at least 30 fetuses with MMC evaluated for surgery; strict adherence to the MOMS protocol until improvements are proven better; comprehensive counseling that includes a reflective period for families of at least 24 hours; and participation in a national registry.
That article was removed from Clinics in Perinatology in early October at the request of the publication’s consulting editor.
According to a note on the publication’s website, "It was learned after publication that the guidelines for fetal repair of myelomeningocele that were presented as being published by the NIH were neither supplied by the NIH nor published by them. To prevent this information from being erroneously cited, the article has been removed."
At press time, the article was still accessible via the National Library of Medicine’s PubMed.gov.*
Having a U.S. agency sponsor a major trial and then organize an expert panel to deliberate on how to best implement its results is unusual, but fetal MMC repair is unusual surgery, said Dr. Julie S. Moldenhauer, a maternal-fetal medicine physician and a representative to the NICHD panel appointed by the North American Fetal Therapy Network. "It is very high-risk surgery that is elective, it is an open procedure, not fetoscopic, and it depends on a very multidisciplinary team," she said in an interview. These are a combination of features that makes it unique, said Dr. Moldenhauer, who is an attending physician at the Center for Fetal Diagnosis and Treatment and the medical director of the special delivery unit, both at CHOP.
One other element of MOMS also adds to its special character: The trial lasted nearly 8 years and randomized just 183 patients, and during that time fetal MMC repair for U.S. patients was limited to three centers – CHOP, UCSF, and Vanderbilt University in Nashville, Tenn. Then, when the researchers reported their positive results last year, NICHD officials and others in this field faced the dilemma of how to make the surgery available elsewhere while ensuring that the results would be as good as in MOMS. "What the NICHD fears is that if MMC repair is offered uncontrolled, the outcomes won’t be what they were in MOMS," said Dr. Johnson.
Making Fetal MMC Repair Better
MMC repair is not easy and still has limitations, most notably the risk for extreme prematurity. "The risk of prematurity is the big obstacle to making it more mainstream," said Dr. Noel B. Tulipan, professor and director of pediatric neurosurgery at Vanderbilt University Medical Center.
Most fetuses treated in MOMS were delivered at 34 weeks’ gestation or later, and "these days, we almost don’t consider 34 weeks premature, because most of these babies do extremely well," Dr. Tulipan said in an interview. "But in MOMS, there was a 13% rate of extreme prematurity" with the infants delivered at less than 30 weeks.
This limitation is improving. The challenge, he said, is the incision and closure of the uterus. Dr. Tulipan and his colleagues have recently given increased attention to suturing the amniotic membranes to prevent them from separating from the uterine wall "With this change, our results have gotten substantially better. We’re getting to a rate that could make it mainstream. If the [extreme] prematurity rate was less than 5%, it would be hard to argue against this surgery."
Fetal MMC repair "will continue to grow, but not by an order of magnitude until there is a new technique that could be offered earlier in gestation and in a minimally-invasive way," said Dr. N. Scott Adzick, surgeon in chief and director of the Center for Fetal Diagnosis and Treatment at CHOP. Currently, MMC repairs are done at 20 weeks’-25 weeks’ 6 days gestation. His group at CHOP, as well as others, is also trying to perfect less-invasive approaches. "We are working on a tissue-engineering approach to seal the MMC defect before birth, and thus prevent exposure of the spinal cord to damaging amniotic fluid, and also prevent leakage of the cerebrospinal fluid from the spina bifida. CSF leakage is the underlying cause of hindbrain herniation and hydrocephalus seen with spina bifida. The goal is for this tissue-engineered component to be introduced through a single fetoscopic port or through an amniocentesis needle under sonographic guidance," Dr. Adzick said.
The risk of prematurity, as well as potential complications for the mother, means that centers offering fetal MMC repair provide extensive counseling for potential parents. "We are proud of the fact that over half of our families decide not to have prenatal surgery after the counseling," said Dr. Hanmin Lee, professor and chief of pediatric surgery at UCSF, and one of the leaders of that center’s MMC fetal repair program.
At CHOP, during the first year after the MOMS publication 238 pregnancies underwent evaluation, with 137 making it to a more extensive stage of assessment. Of these, 40 mothers (29%) underwent fetal MMC repair, Dr. Adzick said.
At Cincinnati Children’s Hospital, which began offering fetal MMC repairs following publication of the MOMS results, the winnowing was about as sharp as at CHOP. Since the program began, the Cincinnati team evaluated 53 pregnancies and performed 10 repairs, said Dr. Foong-Yen Lim, surgical director of the Fetal Care Center there. A handful of these patients who were first seen at Cincinnati decided to travel to Vanderbilt or CHOP instead for the surgery and thereby take advantage of the greater experience those centers offered.
That sentiment underscores a challenge faced by the programs that are trying to establish themselves as new options for fetal MMC repair, as the procedure rolls out post-MOMS.
"There is nothing wrong with patients going to a higher-volume center; they know that their outcome will be close to what is available today, compared with new centers that have not done as many of these procedures," Dr. Lim said in an interview. "Patients should hear the options and pick what’s best for them, not just whether to choose the procedure, but also to decide where to go. The reason why MOMS had such difficulty recruiting patients was that they had to uproot themselves to go to a MMC repair center, and a lot of patients decided not to go.
"Regionalization is the future. Spina bifida is one of the most common birth defects, so there is a large number of cases that could benefit" from fetal repair. But what does it mean to be a fetal surgery center, and what does it require," said CHOP’s Dr. Johnson. "These are the ideas that the fetal surgery community is struggling with and trying to address. The big question is which will be the centers of excellence. The first step toward answering this will be to see whether the new centers can replicate the MOMS results, he added.
Spreading the Word About Fetal Surgery
Another key element in growing fetal MMC repair, and fetal surgeries of all types, is boosting awareness of repair options among primary care obstetrical providers and making referrals more timely.
"The market is controlled by the number of physicians who are aware of surgical interventions," said Dr. Mark I. Evans, a clinical professor of obstetrics, gynecology, and reproductive services at Mount Sinai Medical Center in New York. "The number of anomalies that are potentially amenable to treatment is a small percent of all pregnancies, but with more than 4 million U.S. births per year, it adds up to a fairly substantial number."
Fetal interventions "are accepted therapies; in the fetal community, I don’t think anyone thinks they are still investigational. But the number of cases is small, and a lot of the people who are diagnosing these may not be aware of how successful a lot of the procedures are," said Dr. Johnson.
"Obstetricians and maternal-fetal medicine physicians need to be more aware of what we can do. Our biggest challenge is to disseminate the information. The treatments are safe and effective, and families should at least have the option of going to centers to get more information and make informed decisions."
In addition, "screening ultrasound at between 18 to 20 weeks would be a great thing," he added.
"In the United States, ultrasound screening is becoming more the standard of care, but a lot of screening is not done until 22 or 23 weeks, often too late for patients to be referred to fetal treatment centers for evaluations before 24 weeks so that the option of termination is often no longer available to them. Also, with some anomalies irreversible damage has already occurred by 24 weeks so that fetal therapy would not help. In cases of lower urinary tract obstructions, well over half the cases we see are too late to offer therapy. It is recognized that screening at 19-20 weeks won’t pick up all anomalies, but it would pick up a lot of major anomalies," Dr. Johnson said.
A Fetal Surgery Checklist
Aside from myelomeningocele repair, placental laser ablation in cases of severe twin-twin transfusion syndrome is the fetal surgery with the best evidence base and, for now, it’s also the most frequently performed fetal surgery, although experts say it’s also underused.
"The success story of fetal surgery is laser ablation for TTTS," said Dr. Francois I. Luks, director of the Fetal Treatment Program at Hasbro Children’s Hospital in Providence, R.I. "Left untreated, severe TTTS leads to greater than 80% dual mortality. With laser surgery, survival of at least one twin is in excess of 80%."
First established as a good alternative to standard amnioreduction therapy in a 2004 randomized trial (N. Engl. J. Med. 2004;351:136-44), placental laser ablation has since become standard-of-care for TTTS.
"More and more groups are taking [laser ablation for TTTS] on because they believe they should provide it to patients," and it currently is available at about 30 U.S. centers, said Dr. Lim. "The maternal risk [from placental laser ablation] is acceptable and the outcomes are quite good. But some cases [of TTTS] are still being misdiagnosed," he said.
"Awareness is more widespread, but not at the rate it should be. We still have patients who are not referred until something bad happens," Dr. Lim said in an interview.
In Cincinnati alone, the program does more than 100 placental laser ablations each year, he added.
About 80 per year are performed at CHOP, and roughly 300-400 per year total at the 24 U.S. and Canadian centers that form the North American Fetal Therapy Network (NAFTNet), said Dr. Luks. (CHOP and Cincinnati Children’s are members of NAFTNet.) At least two other U.S. centers not in NAFTNet do more than 100 laser ablations each annually, and several other hospitals outside of NAFTNet do smaller numbers yearly, which means that while the current annual U.S. volume of these cases is uncertain it easily exceeds 500, Dr. Luks said.
All the other fetal surgeries each occur in fewer than about 50 U.S. cases a year, are available at fewer U.S. centers and, in some cases, have a checkered history of success and failure although today several are considered effective, relatively safe, and standard therapy.
The third most-common fetal surgery, based on NAFTNet records, are various types of selective umbilical cord occlusions in twin pregnancies, for reasons such as intrauterine growth retardation, placental insufficiency, and other situations in which problems with one twin puts a healthy twin at risk, said Dr. Johnson.
Dr. Johnson, Dr. Hirose, Dr. Adzick, Dr. Moldenhauer, Dr. Lee, Dr. Tulipan, Dr. Lim, and Dr. Evans all said that they had no relevant financial disclosures.
* This story was updated on 10/3/1012.
Panel Advises Approving Short Bowel Syndrome Drug
SILVER SPRING, MD. – A Food and Drug Administration advisory panel unanimously supported the approval of teduglutide, a recombinant analogue of human glucagon-like peptide-2 (GLP-2) administered subcutaneously, as a treatment to improve the intestinal absorption of fluid and nutrients in adults with short bowel syndrome, at a meeting on Oct. 16.
The FDA’s Gastrointestinal Drugs Advisory Committee voted 12 to 0 that the benefits of teduglutide outweighed the potential risks in patients with short bowel syndrome (SBS). Clinical studies of adults with SBS, who are highly dependent on parenteral nutrition, showed that teduglutide significantly reduced the volume of parenteral nutrition and IV fluids they needed after 6 months of treatment, compared with placebo. While the panelists felt comfortable with the drug’s safety profile, they recommended that more safety data are needed. They also advised following the long-term safety and potential risks of the treatment after approval, including determining whether the risk of colorectal cancer is increased with treatment.
Produced in the small intestine and the proximal large intestine, GLP-2 is "an intestinotrophic peptide that stimulates mucosal epithelium," increasing absorption of fluids and nutrients, according to the manufacturer, NPS Pharmaceuticals. In studies, patients treated with teduglutide had evidence of increased villus height after 21 days of treatment, according to the company.
In a phase III study of 86 adults with SBS, 63% of those treated with 0.05 mg/kg daily had at least a 20% reduction in the volume of parenteral nutrition and IV fluids (the primary end point) required after 24 weeks of treatment, compared with 30% of those on placebo; this was a statistically significant difference. An extension study indicated that this effect is maintained through 1 year of treatment.
If teduglutide is approved, the company will market it as Gattex. About 10,000-15,000 adults in the United States with SBS are dependent on parenteral nutrition and IV fluids for fluid and nutrient replacement, according to NPS. Teduglutide was recently approved in Europe for the same indication. The FDA is expected to make a decision on approval by Dec. 30; if the drug is approved, the company plans to pursue studies in pediatric patients with SBS.
The two drugs currently approved by the FDA for SBS are somatropin rhGH (Zorbtive), a growth hormone approved in 2003, and L-glutamine powder for oral solution (Nutrestore), an adjunctive treatment approved in 2004.
The FDA usually follows the recommendations of its advisory panels, which are not binding. Panelists have been cleared of potential conflicts of interest related to the topic of the meeting. Occasionally, a panelist may be given a waiver, but not at this meeting.
SILVER SPRING, MD. – A Food and Drug Administration advisory panel unanimously supported the approval of teduglutide, a recombinant analogue of human glucagon-like peptide-2 (GLP-2) administered subcutaneously, as a treatment to improve the intestinal absorption of fluid and nutrients in adults with short bowel syndrome, at a meeting on Oct. 16.
The FDA’s Gastrointestinal Drugs Advisory Committee voted 12 to 0 that the benefits of teduglutide outweighed the potential risks in patients with short bowel syndrome (SBS). Clinical studies of adults with SBS, who are highly dependent on parenteral nutrition, showed that teduglutide significantly reduced the volume of parenteral nutrition and IV fluids they needed after 6 months of treatment, compared with placebo. While the panelists felt comfortable with the drug’s safety profile, they recommended that more safety data are needed. They also advised following the long-term safety and potential risks of the treatment after approval, including determining whether the risk of colorectal cancer is increased with treatment.
Produced in the small intestine and the proximal large intestine, GLP-2 is "an intestinotrophic peptide that stimulates mucosal epithelium," increasing absorption of fluids and nutrients, according to the manufacturer, NPS Pharmaceuticals. In studies, patients treated with teduglutide had evidence of increased villus height after 21 days of treatment, according to the company.
In a phase III study of 86 adults with SBS, 63% of those treated with 0.05 mg/kg daily had at least a 20% reduction in the volume of parenteral nutrition and IV fluids (the primary end point) required after 24 weeks of treatment, compared with 30% of those on placebo; this was a statistically significant difference. An extension study indicated that this effect is maintained through 1 year of treatment.
If teduglutide is approved, the company will market it as Gattex. About 10,000-15,000 adults in the United States with SBS are dependent on parenteral nutrition and IV fluids for fluid and nutrient replacement, according to NPS. Teduglutide was recently approved in Europe for the same indication. The FDA is expected to make a decision on approval by Dec. 30; if the drug is approved, the company plans to pursue studies in pediatric patients with SBS.
The two drugs currently approved by the FDA for SBS are somatropin rhGH (Zorbtive), a growth hormone approved in 2003, and L-glutamine powder for oral solution (Nutrestore), an adjunctive treatment approved in 2004.
The FDA usually follows the recommendations of its advisory panels, which are not binding. Panelists have been cleared of potential conflicts of interest related to the topic of the meeting. Occasionally, a panelist may be given a waiver, but not at this meeting.
SILVER SPRING, MD. – A Food and Drug Administration advisory panel unanimously supported the approval of teduglutide, a recombinant analogue of human glucagon-like peptide-2 (GLP-2) administered subcutaneously, as a treatment to improve the intestinal absorption of fluid and nutrients in adults with short bowel syndrome, at a meeting on Oct. 16.
The FDA’s Gastrointestinal Drugs Advisory Committee voted 12 to 0 that the benefits of teduglutide outweighed the potential risks in patients with short bowel syndrome (SBS). Clinical studies of adults with SBS, who are highly dependent on parenteral nutrition, showed that teduglutide significantly reduced the volume of parenteral nutrition and IV fluids they needed after 6 months of treatment, compared with placebo. While the panelists felt comfortable with the drug’s safety profile, they recommended that more safety data are needed. They also advised following the long-term safety and potential risks of the treatment after approval, including determining whether the risk of colorectal cancer is increased with treatment.
Produced in the small intestine and the proximal large intestine, GLP-2 is "an intestinotrophic peptide that stimulates mucosal epithelium," increasing absorption of fluids and nutrients, according to the manufacturer, NPS Pharmaceuticals. In studies, patients treated with teduglutide had evidence of increased villus height after 21 days of treatment, according to the company.
In a phase III study of 86 adults with SBS, 63% of those treated with 0.05 mg/kg daily had at least a 20% reduction in the volume of parenteral nutrition and IV fluids (the primary end point) required after 24 weeks of treatment, compared with 30% of those on placebo; this was a statistically significant difference. An extension study indicated that this effect is maintained through 1 year of treatment.
If teduglutide is approved, the company will market it as Gattex. About 10,000-15,000 adults in the United States with SBS are dependent on parenteral nutrition and IV fluids for fluid and nutrient replacement, according to NPS. Teduglutide was recently approved in Europe for the same indication. The FDA is expected to make a decision on approval by Dec. 30; if the drug is approved, the company plans to pursue studies in pediatric patients with SBS.
The two drugs currently approved by the FDA for SBS are somatropin rhGH (Zorbtive), a growth hormone approved in 2003, and L-glutamine powder for oral solution (Nutrestore), an adjunctive treatment approved in 2004.
The FDA usually follows the recommendations of its advisory panels, which are not binding. Panelists have been cleared of potential conflicts of interest related to the topic of the meeting. Occasionally, a panelist may be given a waiver, but not at this meeting.
AT A MEETING OF THE GASTROINTESTINAL DRUGS ADVISORY COMMITTEE