User login
Determining cause of skin lesions in COVID-19 patients remains challenging
published in the Journal of the American Academy of Dermatology.
SARS-CoV-2 infection has been associated with a range of skin conditions, wrote Antonio Martinez-Lopez, MD, of Virgen de las Nieves University Hospital, Granada, Spain, and colleagues, who provided an overview of the cutaneous side effects associated with drugs used to treat COVID-19 infection.
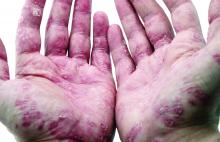
“Cutaneous manifestations have recently been described in patients with the new coronavirus infection, similar to cutaneous involvement occurring in common viral infections,” they said. Infected individuals have experienced maculopapular eruption, pseudo-chilblain lesions, urticaria, monomorphic disseminated vesicular lesions, acral vesicular-pustulous lesions, and livedo or necrosis, they noted.
Diagnosing skin manifestations in patients with COVID-19 remains a challenge, because it is unclear whether the skin lesions are related to the virus, the authors said. “Skin diseases not related to coronavirus, other seasonal viral infections, and drug reactions should be considered in the differential diagnosis, especially in those patients suffering from nonspecific manifestations such as urticaria or maculopapular eruptions,” they wrote.
However, “urticarial lesions and maculopapular eruptions in SARS-CoV-2 infections usually appear at the same time as the systemic symptoms, while drug adverse reactions are likely to arise hours to days after the start of the treatment,” they said.
The reviewers noted several cutaneous side effects associated with several of the often-prescribed drugs for COVID-19 infection. The antimalarials hydroxychloroquine and chloroquine had been authorized for COVID-19 treatment by the Food and Drug Administration, but this emergency authorization was rescinded in June. They noted that up to 11.5% of patients on these drugs may experience cutaneous adverse effects, including some that “can be mistaken for skin manifestations of SARS-CoV-2, especially those with maculopapular rash or exanthematous reactions.” Another side effect is exacerbation of psoriasis, which has been described in patients with COVID-19, the authors said.
The oral antiretroviral combination lopinavir/ritonavir, under investigation in clinical trials for COVID-19, has been associated with skin rashes in as many as 5% of adults in HIV studies. Usually appearing after treatment is started, the maculopapular pruritic rash is “usually well tolerated,” they said, although there have been reports of Stevens-Johnson syndrome. Alopecia areata is among the other side effects reported.
Remdesivir also has been authorized for emergency treatment of COVID-19, and the small amount of data available suggest that cutaneous manifestations may be infrequent, the reviewers said. In a recent study of 53 patients treated with remdesivir for 10 days, approximately 8% developed a rash, but the study did not include any information “about rash morphology, distribution, or timeline in relation to remdesivir that may help clinicians differentiate from cutaneous manifestations of COVID-19,” they said.
Other potential treatments for complications of COVID-19 include imatinib, tocilizumab, anakinra, immunoglobulins, corticosteroids, colchicine, and low molecular weight heparins; all have the potential for association with skin reactions, but data on skin manifestations associated with COVID-19 are limited, the authors wrote.
Notably, data on the use of systemic corticosteroids for COVID-19 patients are controversial, although preliminary data showed some reduced mortality in COVID-19 patients who were on respiratory support, they noted. “With regard to differential diagnosis of cutaneous manifestations of COVID-19, the vascular fragility associated with corticosteroid use, especially in elderly patients, may be similar to the thrombotic complications of COVID-19 infection.”
Knowledge about the virology of COVID-19 continues to evolve rapidly, and the number of drugs being studied as treatments continues to expand, the authors pointed out.
“By considering adverse drug reactions in the differential diagnosis, dermatologists can be useful in assisting in the care of these patients,” they wrote. Drugs, rather than the infection, may be the cause of skin reactions in some COVID-19 patients, and “management is often symptomatic, but it is sometimes necessary to modify or discontinue the treatment, and some conditions can even be life-threatening,” they concluded.
The study received no outside funding. The researchers had no financial conflicts to disclose.
SOURCE: Martinez-Lopez A et al. J Am Acad Dermatol. 2020 doi: 10.1016/j.jaad.2020.08.006.
published in the Journal of the American Academy of Dermatology.
SARS-CoV-2 infection has been associated with a range of skin conditions, wrote Antonio Martinez-Lopez, MD, of Virgen de las Nieves University Hospital, Granada, Spain, and colleagues, who provided an overview of the cutaneous side effects associated with drugs used to treat COVID-19 infection.
“Cutaneous manifestations have recently been described in patients with the new coronavirus infection, similar to cutaneous involvement occurring in common viral infections,” they said. Infected individuals have experienced maculopapular eruption, pseudo-chilblain lesions, urticaria, monomorphic disseminated vesicular lesions, acral vesicular-pustulous lesions, and livedo or necrosis, they noted.
Diagnosing skin manifestations in patients with COVID-19 remains a challenge, because it is unclear whether the skin lesions are related to the virus, the authors said. “Skin diseases not related to coronavirus, other seasonal viral infections, and drug reactions should be considered in the differential diagnosis, especially in those patients suffering from nonspecific manifestations such as urticaria or maculopapular eruptions,” they wrote.
However, “urticarial lesions and maculopapular eruptions in SARS-CoV-2 infections usually appear at the same time as the systemic symptoms, while drug adverse reactions are likely to arise hours to days after the start of the treatment,” they said.
The reviewers noted several cutaneous side effects associated with several of the often-prescribed drugs for COVID-19 infection. The antimalarials hydroxychloroquine and chloroquine had been authorized for COVID-19 treatment by the Food and Drug Administration, but this emergency authorization was rescinded in June. They noted that up to 11.5% of patients on these drugs may experience cutaneous adverse effects, including some that “can be mistaken for skin manifestations of SARS-CoV-2, especially those with maculopapular rash or exanthematous reactions.” Another side effect is exacerbation of psoriasis, which has been described in patients with COVID-19, the authors said.
The oral antiretroviral combination lopinavir/ritonavir, under investigation in clinical trials for COVID-19, has been associated with skin rashes in as many as 5% of adults in HIV studies. Usually appearing after treatment is started, the maculopapular pruritic rash is “usually well tolerated,” they said, although there have been reports of Stevens-Johnson syndrome. Alopecia areata is among the other side effects reported.
Remdesivir also has been authorized for emergency treatment of COVID-19, and the small amount of data available suggest that cutaneous manifestations may be infrequent, the reviewers said. In a recent study of 53 patients treated with remdesivir for 10 days, approximately 8% developed a rash, but the study did not include any information “about rash morphology, distribution, or timeline in relation to remdesivir that may help clinicians differentiate from cutaneous manifestations of COVID-19,” they said.
Other potential treatments for complications of COVID-19 include imatinib, tocilizumab, anakinra, immunoglobulins, corticosteroids, colchicine, and low molecular weight heparins; all have the potential for association with skin reactions, but data on skin manifestations associated with COVID-19 are limited, the authors wrote.
Notably, data on the use of systemic corticosteroids for COVID-19 patients are controversial, although preliminary data showed some reduced mortality in COVID-19 patients who were on respiratory support, they noted. “With regard to differential diagnosis of cutaneous manifestations of COVID-19, the vascular fragility associated with corticosteroid use, especially in elderly patients, may be similar to the thrombotic complications of COVID-19 infection.”
Knowledge about the virology of COVID-19 continues to evolve rapidly, and the number of drugs being studied as treatments continues to expand, the authors pointed out.
“By considering adverse drug reactions in the differential diagnosis, dermatologists can be useful in assisting in the care of these patients,” they wrote. Drugs, rather than the infection, may be the cause of skin reactions in some COVID-19 patients, and “management is often symptomatic, but it is sometimes necessary to modify or discontinue the treatment, and some conditions can even be life-threatening,” they concluded.
The study received no outside funding. The researchers had no financial conflicts to disclose.
SOURCE: Martinez-Lopez A et al. J Am Acad Dermatol. 2020 doi: 10.1016/j.jaad.2020.08.006.
published in the Journal of the American Academy of Dermatology.
SARS-CoV-2 infection has been associated with a range of skin conditions, wrote Antonio Martinez-Lopez, MD, of Virgen de las Nieves University Hospital, Granada, Spain, and colleagues, who provided an overview of the cutaneous side effects associated with drugs used to treat COVID-19 infection.
“Cutaneous manifestations have recently been described in patients with the new coronavirus infection, similar to cutaneous involvement occurring in common viral infections,” they said. Infected individuals have experienced maculopapular eruption, pseudo-chilblain lesions, urticaria, monomorphic disseminated vesicular lesions, acral vesicular-pustulous lesions, and livedo or necrosis, they noted.
Diagnosing skin manifestations in patients with COVID-19 remains a challenge, because it is unclear whether the skin lesions are related to the virus, the authors said. “Skin diseases not related to coronavirus, other seasonal viral infections, and drug reactions should be considered in the differential diagnosis, especially in those patients suffering from nonspecific manifestations such as urticaria or maculopapular eruptions,” they wrote.
However, “urticarial lesions and maculopapular eruptions in SARS-CoV-2 infections usually appear at the same time as the systemic symptoms, while drug adverse reactions are likely to arise hours to days after the start of the treatment,” they said.
The reviewers noted several cutaneous side effects associated with several of the often-prescribed drugs for COVID-19 infection. The antimalarials hydroxychloroquine and chloroquine had been authorized for COVID-19 treatment by the Food and Drug Administration, but this emergency authorization was rescinded in June. They noted that up to 11.5% of patients on these drugs may experience cutaneous adverse effects, including some that “can be mistaken for skin manifestations of SARS-CoV-2, especially those with maculopapular rash or exanthematous reactions.” Another side effect is exacerbation of psoriasis, which has been described in patients with COVID-19, the authors said.
The oral antiretroviral combination lopinavir/ritonavir, under investigation in clinical trials for COVID-19, has been associated with skin rashes in as many as 5% of adults in HIV studies. Usually appearing after treatment is started, the maculopapular pruritic rash is “usually well tolerated,” they said, although there have been reports of Stevens-Johnson syndrome. Alopecia areata is among the other side effects reported.
Remdesivir also has been authorized for emergency treatment of COVID-19, and the small amount of data available suggest that cutaneous manifestations may be infrequent, the reviewers said. In a recent study of 53 patients treated with remdesivir for 10 days, approximately 8% developed a rash, but the study did not include any information “about rash morphology, distribution, or timeline in relation to remdesivir that may help clinicians differentiate from cutaneous manifestations of COVID-19,” they said.
Other potential treatments for complications of COVID-19 include imatinib, tocilizumab, anakinra, immunoglobulins, corticosteroids, colchicine, and low molecular weight heparins; all have the potential for association with skin reactions, but data on skin manifestations associated with COVID-19 are limited, the authors wrote.
Notably, data on the use of systemic corticosteroids for COVID-19 patients are controversial, although preliminary data showed some reduced mortality in COVID-19 patients who were on respiratory support, they noted. “With regard to differential diagnosis of cutaneous manifestations of COVID-19, the vascular fragility associated with corticosteroid use, especially in elderly patients, may be similar to the thrombotic complications of COVID-19 infection.”
Knowledge about the virology of COVID-19 continues to evolve rapidly, and the number of drugs being studied as treatments continues to expand, the authors pointed out.
“By considering adverse drug reactions in the differential diagnosis, dermatologists can be useful in assisting in the care of these patients,” they wrote. Drugs, rather than the infection, may be the cause of skin reactions in some COVID-19 patients, and “management is often symptomatic, but it is sometimes necessary to modify or discontinue the treatment, and some conditions can even be life-threatening,” they concluded.
The study received no outside funding. The researchers had no financial conflicts to disclose.
SOURCE: Martinez-Lopez A et al. J Am Acad Dermatol. 2020 doi: 10.1016/j.jaad.2020.08.006.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
FDA clamps down on compliance for gluten-free products
To retain the label of “gluten free,” manufacturers of foods that are fermented and hydrolyzed, or that contain fermented or hydrolyzed ingredients, must make and keep detailed records of the manufacturing and production process, according to a final rule issued by the Food and Drug Administration.
In an announcement released on Aug. 12, the FDA stated that manufacturers must confirm that food products such as soy sauce, yogurt, sauerkraut, pickles, cheese, and green olives, as well as distilled foods such as vinegar, meet the definition of gluten free before the fermentation or hydrolysis process. In addition, the rule states that “the manufacturer has adequately evaluated the potential for cross-contact with gluten during the manufacturing process; and if necessary, measures are in place to prevent the introduction of gluten into the food during the manufacturing process,” according to the FDA.
Gluten breaks down during fermentation and hydrolysis, and the gluten-free status of products manufactured in this way can’t be confirmed after the process using currently available methods, according to the FDA.
The new rule is designed to ensure that products labeled as gluten-free meet the definition of gluten free, which remains unchanged from the FDA guidance in 2013.
“The FDA continues to work to protect people with celiac disease, which impacts at least 3 million Americans,” FDA Commissioner Stephen M. Hahn, MD, said in a statement.
“The agency has taken a number of steps on this front by first establishing a standardized definition of gluten free, and now by continuing to work to ensure manufacturers are keeping the products that are labeled with this claim gluten free,” he emphasized.
The final rule states that manufacturers will not need to keep such records if and when other analytical methods are developed, but in the meantime products that do not meet the definition will be deemed misbranded, according to the FDA.
To retain the label of “gluten free,” manufacturers of foods that are fermented and hydrolyzed, or that contain fermented or hydrolyzed ingredients, must make and keep detailed records of the manufacturing and production process, according to a final rule issued by the Food and Drug Administration.
In an announcement released on Aug. 12, the FDA stated that manufacturers must confirm that food products such as soy sauce, yogurt, sauerkraut, pickles, cheese, and green olives, as well as distilled foods such as vinegar, meet the definition of gluten free before the fermentation or hydrolysis process. In addition, the rule states that “the manufacturer has adequately evaluated the potential for cross-contact with gluten during the manufacturing process; and if necessary, measures are in place to prevent the introduction of gluten into the food during the manufacturing process,” according to the FDA.
Gluten breaks down during fermentation and hydrolysis, and the gluten-free status of products manufactured in this way can’t be confirmed after the process using currently available methods, according to the FDA.
The new rule is designed to ensure that products labeled as gluten-free meet the definition of gluten free, which remains unchanged from the FDA guidance in 2013.
“The FDA continues to work to protect people with celiac disease, which impacts at least 3 million Americans,” FDA Commissioner Stephen M. Hahn, MD, said in a statement.
“The agency has taken a number of steps on this front by first establishing a standardized definition of gluten free, and now by continuing to work to ensure manufacturers are keeping the products that are labeled with this claim gluten free,” he emphasized.
The final rule states that manufacturers will not need to keep such records if and when other analytical methods are developed, but in the meantime products that do not meet the definition will be deemed misbranded, according to the FDA.
To retain the label of “gluten free,” manufacturers of foods that are fermented and hydrolyzed, or that contain fermented or hydrolyzed ingredients, must make and keep detailed records of the manufacturing and production process, according to a final rule issued by the Food and Drug Administration.
In an announcement released on Aug. 12, the FDA stated that manufacturers must confirm that food products such as soy sauce, yogurt, sauerkraut, pickles, cheese, and green olives, as well as distilled foods such as vinegar, meet the definition of gluten free before the fermentation or hydrolysis process. In addition, the rule states that “the manufacturer has adequately evaluated the potential for cross-contact with gluten during the manufacturing process; and if necessary, measures are in place to prevent the introduction of gluten into the food during the manufacturing process,” according to the FDA.
Gluten breaks down during fermentation and hydrolysis, and the gluten-free status of products manufactured in this way can’t be confirmed after the process using currently available methods, according to the FDA.
The new rule is designed to ensure that products labeled as gluten-free meet the definition of gluten free, which remains unchanged from the FDA guidance in 2013.
“The FDA continues to work to protect people with celiac disease, which impacts at least 3 million Americans,” FDA Commissioner Stephen M. Hahn, MD, said in a statement.
“The agency has taken a number of steps on this front by first establishing a standardized definition of gluten free, and now by continuing to work to ensure manufacturers are keeping the products that are labeled with this claim gluten free,” he emphasized.
The final rule states that manufacturers will not need to keep such records if and when other analytical methods are developed, but in the meantime products that do not meet the definition will be deemed misbranded, according to the FDA.
Cachexia affects more than half of lupus patients
Cachexia developed in 56% of adults with systemic lupus erythematosus over a 5-year period, and 18% did not recover their weight, based on data from more than 2,000 patients.
Although weight loss is common in patients with systemic lupus erythematosus (SLE), cachexia, a disorder of involuntary weight loss, is largely undescribed in SLE patients, wrote George Stojan, MD, of Johns Hopkins University, Baltimore, and colleagues. Cachexia has been described in a range of disorders, including heart failure, renal disease, and rheumatoid arthritis, they said. “Cachexia has been shown to lead to progressive functional impairment, treatment-related complications, poor quality of life, and increased mortality,” they added.
In a study published in Arthritis Care & Research, the investigators reviewed data from the Hopkins Lupus Cohort, consisting of all SLE patients seen at a single center who are followed at least quarterly.
The study population included 2,452 SLE patients older than 18 years who had their weight assessed at each clinic visit. The average follow-up period was 7.75 years, and the average number of weight measurements per patient was nearly 24.
Cachexia was defined as a 5% stable weight loss in 6 months without starvation relative to the average weight in all prior cohort visits; and/or weight loss of more than 2% without starvation relative to the average weight in all prior cohort visits in addition to a body mass index less than 20 kg/m2.
Overall, the risk for cachexia within 5 years of entering the study was significantly higher in patients with a BMI less than 20, current steroid use, vasculitis, lupus nephritis, serositis, hematologic lupus, positive anti-double strand DNA (anti-dsDNA), anti-Smith (anti-Sm), and antiribonucleoprotein (anti-RNP), the researchers noted. After adjustment for prednisone use, cachexia remained significantly associated with lupus nephritis, vasculitis, serositis, and hematologic lupus.
Future organ damage including cataracts, retinal change or optic atrophy, cognitive impairment, cerebrovascular accidents, cranial or peripheral neuropathy, pulmonary hypertension, pleural fibrosis, angina or coronary bypass, bowel infarction or resection, osteoporosis, avascular necrosis, and premature gonadal failure were significantly more likely among patients with intermittent cachexia, compared with those with continuous or no cachexia. Patients with continuous cachexia were significantly more likely to experience an estimated glomerular filtration rate less than 50 mL/min/1.73 m2, proteinuria greater than 3.5 g/day, and end-stage renal disease.
The patients who never developed cachexia were significantly less likely to develop malignancies, diabetes, valvular disease, or cardiomyopathy than were those who did have cachexia, the researchers said.
The mechanisms of action for cachexia in SLE remain unclear, but studies in cancer patients may provide some guidance, the researchers noted. “Tumors secrete a range of procachexia factors thought to be unique to cancer-related cachexia, and colloquially termed the ‘tumor secretome,’ ” they said. “Every single proinflammatory cytokine mentioned as part of the tumor secretome has a role in lupus pathogenesis,” suggesting a possible common pathway to cachexia across different diseases, they said.
The study findings were limited by several factors, mainly the use of BMI to measure weight “since BMI is a rather poor indicator of percent of body fat,” the researchers noted. “Ideally, cachexia would be defined as sarcopenia based on body composition evaluation with a dual x-ray absorptiometry,” they wrote.
The study was supported by the National Institutes of Health and the NIH Roadmap for Medical Research. The researchers had no financial conflicts to disclose.
SOURCE: Stojan G et al. Arthritis Care Res. 2020 Aug 2. doi: 10.1002/acr.24395.
Cachexia developed in 56% of adults with systemic lupus erythematosus over a 5-year period, and 18% did not recover their weight, based on data from more than 2,000 patients.
Although weight loss is common in patients with systemic lupus erythematosus (SLE), cachexia, a disorder of involuntary weight loss, is largely undescribed in SLE patients, wrote George Stojan, MD, of Johns Hopkins University, Baltimore, and colleagues. Cachexia has been described in a range of disorders, including heart failure, renal disease, and rheumatoid arthritis, they said. “Cachexia has been shown to lead to progressive functional impairment, treatment-related complications, poor quality of life, and increased mortality,” they added.
In a study published in Arthritis Care & Research, the investigators reviewed data from the Hopkins Lupus Cohort, consisting of all SLE patients seen at a single center who are followed at least quarterly.
The study population included 2,452 SLE patients older than 18 years who had their weight assessed at each clinic visit. The average follow-up period was 7.75 years, and the average number of weight measurements per patient was nearly 24.
Cachexia was defined as a 5% stable weight loss in 6 months without starvation relative to the average weight in all prior cohort visits; and/or weight loss of more than 2% without starvation relative to the average weight in all prior cohort visits in addition to a body mass index less than 20 kg/m2.
Overall, the risk for cachexia within 5 years of entering the study was significantly higher in patients with a BMI less than 20, current steroid use, vasculitis, lupus nephritis, serositis, hematologic lupus, positive anti-double strand DNA (anti-dsDNA), anti-Smith (anti-Sm), and antiribonucleoprotein (anti-RNP), the researchers noted. After adjustment for prednisone use, cachexia remained significantly associated with lupus nephritis, vasculitis, serositis, and hematologic lupus.
Future organ damage including cataracts, retinal change or optic atrophy, cognitive impairment, cerebrovascular accidents, cranial or peripheral neuropathy, pulmonary hypertension, pleural fibrosis, angina or coronary bypass, bowel infarction or resection, osteoporosis, avascular necrosis, and premature gonadal failure were significantly more likely among patients with intermittent cachexia, compared with those with continuous or no cachexia. Patients with continuous cachexia were significantly more likely to experience an estimated glomerular filtration rate less than 50 mL/min/1.73 m2, proteinuria greater than 3.5 g/day, and end-stage renal disease.
The patients who never developed cachexia were significantly less likely to develop malignancies, diabetes, valvular disease, or cardiomyopathy than were those who did have cachexia, the researchers said.
The mechanisms of action for cachexia in SLE remain unclear, but studies in cancer patients may provide some guidance, the researchers noted. “Tumors secrete a range of procachexia factors thought to be unique to cancer-related cachexia, and colloquially termed the ‘tumor secretome,’ ” they said. “Every single proinflammatory cytokine mentioned as part of the tumor secretome has a role in lupus pathogenesis,” suggesting a possible common pathway to cachexia across different diseases, they said.
The study findings were limited by several factors, mainly the use of BMI to measure weight “since BMI is a rather poor indicator of percent of body fat,” the researchers noted. “Ideally, cachexia would be defined as sarcopenia based on body composition evaluation with a dual x-ray absorptiometry,” they wrote.
The study was supported by the National Institutes of Health and the NIH Roadmap for Medical Research. The researchers had no financial conflicts to disclose.
SOURCE: Stojan G et al. Arthritis Care Res. 2020 Aug 2. doi: 10.1002/acr.24395.
Cachexia developed in 56% of adults with systemic lupus erythematosus over a 5-year period, and 18% did not recover their weight, based on data from more than 2,000 patients.
Although weight loss is common in patients with systemic lupus erythematosus (SLE), cachexia, a disorder of involuntary weight loss, is largely undescribed in SLE patients, wrote George Stojan, MD, of Johns Hopkins University, Baltimore, and colleagues. Cachexia has been described in a range of disorders, including heart failure, renal disease, and rheumatoid arthritis, they said. “Cachexia has been shown to lead to progressive functional impairment, treatment-related complications, poor quality of life, and increased mortality,” they added.
In a study published in Arthritis Care & Research, the investigators reviewed data from the Hopkins Lupus Cohort, consisting of all SLE patients seen at a single center who are followed at least quarterly.
The study population included 2,452 SLE patients older than 18 years who had their weight assessed at each clinic visit. The average follow-up period was 7.75 years, and the average number of weight measurements per patient was nearly 24.
Cachexia was defined as a 5% stable weight loss in 6 months without starvation relative to the average weight in all prior cohort visits; and/or weight loss of more than 2% without starvation relative to the average weight in all prior cohort visits in addition to a body mass index less than 20 kg/m2.
Overall, the risk for cachexia within 5 years of entering the study was significantly higher in patients with a BMI less than 20, current steroid use, vasculitis, lupus nephritis, serositis, hematologic lupus, positive anti-double strand DNA (anti-dsDNA), anti-Smith (anti-Sm), and antiribonucleoprotein (anti-RNP), the researchers noted. After adjustment for prednisone use, cachexia remained significantly associated with lupus nephritis, vasculitis, serositis, and hematologic lupus.
Future organ damage including cataracts, retinal change or optic atrophy, cognitive impairment, cerebrovascular accidents, cranial or peripheral neuropathy, pulmonary hypertension, pleural fibrosis, angina or coronary bypass, bowel infarction or resection, osteoporosis, avascular necrosis, and premature gonadal failure were significantly more likely among patients with intermittent cachexia, compared with those with continuous or no cachexia. Patients with continuous cachexia were significantly more likely to experience an estimated glomerular filtration rate less than 50 mL/min/1.73 m2, proteinuria greater than 3.5 g/day, and end-stage renal disease.
The patients who never developed cachexia were significantly less likely to develop malignancies, diabetes, valvular disease, or cardiomyopathy than were those who did have cachexia, the researchers said.
The mechanisms of action for cachexia in SLE remain unclear, but studies in cancer patients may provide some guidance, the researchers noted. “Tumors secrete a range of procachexia factors thought to be unique to cancer-related cachexia, and colloquially termed the ‘tumor secretome,’ ” they said. “Every single proinflammatory cytokine mentioned as part of the tumor secretome has a role in lupus pathogenesis,” suggesting a possible common pathway to cachexia across different diseases, they said.
The study findings were limited by several factors, mainly the use of BMI to measure weight “since BMI is a rather poor indicator of percent of body fat,” the researchers noted. “Ideally, cachexia would be defined as sarcopenia based on body composition evaluation with a dual x-ray absorptiometry,” they wrote.
The study was supported by the National Institutes of Health and the NIH Roadmap for Medical Research. The researchers had no financial conflicts to disclose.
SOURCE: Stojan G et al. Arthritis Care Res. 2020 Aug 2. doi: 10.1002/acr.24395.
FROM ARTHRITIS CARE & RESEARCH
Health disparities training falls short for internal medicine residents
Less than half of internal medicine residency program directors report formal curricula on the topic of health disparities, according to findings of a survey of medical directors and residents across the United States.
Despite recommendations from the Institute of Medicine going back to 2002 calling for increased education on the topic for health care providers, data from a 2012 survey showed that only 17% of internal medicine programs had a health disparities curriculum, wrote Denise M. Dupras, MD, of the Mayo Medical School, Rochester, Minn., and colleagues.
To describe internal medicine residency training programs’ curricula and educational experiences on health disparities and to determine residents’ perceptions of training, the researchers designed a cross-sectional survey study including 227 program directors and 22,723 internal medicine residents. The survey was conducted from August to November 2015.
Overall, 91 program directors (40%) reported a curriculum in health disparities, but only 16 of them described the quality of their education as very good or excellent. In 56% of the programs, outcomes of the curriculum were not measured.
A majority (90%) of the programs included racial/ethnic diversity and socioeconomic status in their curricula, 58% included information about limited English proficiency, and 53% included information about gender identity and sexual orientation.
Reported barriers to curriculum development in 132 programs that did not have a health disparities curriculum included lack of time in the current curriculum, insufficient faculty skill to teach the topic, lack of institutional support, and lack of faculty interest, the researchers noted.
A total of 13,251 residents (70%) reported receiving some training in caring for patients at risk for health disparities over 3 years of training, and 10,494 (80%) of these rated the quality as very good or excellent. “Residents who cared for a larger proportion of underserved patients perceived that they received health disparities training at a higher rate,” the researchers wrote. However, increased care of at-risk populations does not necessarily translate into increased knowledge and skills. “Our finding that residents’ rating of the quality of their training was not associated with the presence of a curriculum in health disparities in their program also raises a concern that perceptions may overestimate the acquisition of needed skills,” they added.
The major limitation of the study was “that residents were not asked directly if they were exposed to a curriculum in health disparities but rather if they received training in the care of patients who would be at risk, which raises the concern that we cannot distinguish between their recognition of a formal and informal curriculum,” the researchers noted. In addition, the survey could not confirm that program directors were aware of all training. “Furthermore, because the survey items were embedded in larger program director survey, we were limited in the ability to ask them to define more specifically the components of their health disparities curricula,” they wrote.
However, the results were strengthened by the large and comprehensive study population, and highlight not only the need for standardized health disparities curricula, but also the need for research to determine the most effective domains for such curricula in graduate medical education, they emphasized.
“There are opportunities to explore partnerships among residencies, institutional clinical practices, and communities for productive collaborations around disparities-related quality improvement projects to address gaps in health care that are specific to the populations they serve,” they concluded.
The surveys were conducted in 2015 and the comparative work in 2018, prior to the COVID-19 pandemic and the subsequent increased concerns about disparities in health care, Dr. Dupras said in an interview.
“We conducted the survey because we recognized that health disparities were still prevalent in our society despite calls to improve the education of our learners to address them. We wanted to determine what our programs were providing for educational curriculum and what our learners were experiencing,” she said.
“We did not know what the surveys would show, so I cannot say that we were surprised by the findings,” said Dr. Dupras. “One of the challenges in interpreting our results is inherent in studies that rely on surveys. We cannot know how those filling out the surveys interpret the questions.” The study results yield several messages.
“First, residency training programs have opportunities to do a better job in developing educational opportunities related to health disparities; second, residents learn in the context of care and we must optimize education around these experiences; third, every patient is different. It is time to move towards cultural humility, since the risk for disparities is not associated with one patient characteristic, but composed of multiple factors,” she said.
“Given that 5 years has passed since our original survey, it would be important to repeat the survey and consider expanding it to include other training programs that provide frontline care, such as family medicine and pediatrics,” Dr. Dupras noted.
Dr. Dupras and colleagues had no financial conflicts to disclose.
SOURCE: Dupras DM et al. JAMA Netw Open. 2020 Aug 10. doi: 10.1001/jamanetworkopen.2020.12757.
Less than half of internal medicine residency program directors report formal curricula on the topic of health disparities, according to findings of a survey of medical directors and residents across the United States.
Despite recommendations from the Institute of Medicine going back to 2002 calling for increased education on the topic for health care providers, data from a 2012 survey showed that only 17% of internal medicine programs had a health disparities curriculum, wrote Denise M. Dupras, MD, of the Mayo Medical School, Rochester, Minn., and colleagues.
To describe internal medicine residency training programs’ curricula and educational experiences on health disparities and to determine residents’ perceptions of training, the researchers designed a cross-sectional survey study including 227 program directors and 22,723 internal medicine residents. The survey was conducted from August to November 2015.
Overall, 91 program directors (40%) reported a curriculum in health disparities, but only 16 of them described the quality of their education as very good or excellent. In 56% of the programs, outcomes of the curriculum were not measured.
A majority (90%) of the programs included racial/ethnic diversity and socioeconomic status in their curricula, 58% included information about limited English proficiency, and 53% included information about gender identity and sexual orientation.
Reported barriers to curriculum development in 132 programs that did not have a health disparities curriculum included lack of time in the current curriculum, insufficient faculty skill to teach the topic, lack of institutional support, and lack of faculty interest, the researchers noted.
A total of 13,251 residents (70%) reported receiving some training in caring for patients at risk for health disparities over 3 years of training, and 10,494 (80%) of these rated the quality as very good or excellent. “Residents who cared for a larger proportion of underserved patients perceived that they received health disparities training at a higher rate,” the researchers wrote. However, increased care of at-risk populations does not necessarily translate into increased knowledge and skills. “Our finding that residents’ rating of the quality of their training was not associated with the presence of a curriculum in health disparities in their program also raises a concern that perceptions may overestimate the acquisition of needed skills,” they added.
The major limitation of the study was “that residents were not asked directly if they were exposed to a curriculum in health disparities but rather if they received training in the care of patients who would be at risk, which raises the concern that we cannot distinguish between their recognition of a formal and informal curriculum,” the researchers noted. In addition, the survey could not confirm that program directors were aware of all training. “Furthermore, because the survey items were embedded in larger program director survey, we were limited in the ability to ask them to define more specifically the components of their health disparities curricula,” they wrote.
However, the results were strengthened by the large and comprehensive study population, and highlight not only the need for standardized health disparities curricula, but also the need for research to determine the most effective domains for such curricula in graduate medical education, they emphasized.
“There are opportunities to explore partnerships among residencies, institutional clinical practices, and communities for productive collaborations around disparities-related quality improvement projects to address gaps in health care that are specific to the populations they serve,” they concluded.
The surveys were conducted in 2015 and the comparative work in 2018, prior to the COVID-19 pandemic and the subsequent increased concerns about disparities in health care, Dr. Dupras said in an interview.
“We conducted the survey because we recognized that health disparities were still prevalent in our society despite calls to improve the education of our learners to address them. We wanted to determine what our programs were providing for educational curriculum and what our learners were experiencing,” she said.
“We did not know what the surveys would show, so I cannot say that we were surprised by the findings,” said Dr. Dupras. “One of the challenges in interpreting our results is inherent in studies that rely on surveys. We cannot know how those filling out the surveys interpret the questions.” The study results yield several messages.
“First, residency training programs have opportunities to do a better job in developing educational opportunities related to health disparities; second, residents learn in the context of care and we must optimize education around these experiences; third, every patient is different. It is time to move towards cultural humility, since the risk for disparities is not associated with one patient characteristic, but composed of multiple factors,” she said.
“Given that 5 years has passed since our original survey, it would be important to repeat the survey and consider expanding it to include other training programs that provide frontline care, such as family medicine and pediatrics,” Dr. Dupras noted.
Dr. Dupras and colleagues had no financial conflicts to disclose.
SOURCE: Dupras DM et al. JAMA Netw Open. 2020 Aug 10. doi: 10.1001/jamanetworkopen.2020.12757.
Less than half of internal medicine residency program directors report formal curricula on the topic of health disparities, according to findings of a survey of medical directors and residents across the United States.
Despite recommendations from the Institute of Medicine going back to 2002 calling for increased education on the topic for health care providers, data from a 2012 survey showed that only 17% of internal medicine programs had a health disparities curriculum, wrote Denise M. Dupras, MD, of the Mayo Medical School, Rochester, Minn., and colleagues.
To describe internal medicine residency training programs’ curricula and educational experiences on health disparities and to determine residents’ perceptions of training, the researchers designed a cross-sectional survey study including 227 program directors and 22,723 internal medicine residents. The survey was conducted from August to November 2015.
Overall, 91 program directors (40%) reported a curriculum in health disparities, but only 16 of them described the quality of their education as very good or excellent. In 56% of the programs, outcomes of the curriculum were not measured.
A majority (90%) of the programs included racial/ethnic diversity and socioeconomic status in their curricula, 58% included information about limited English proficiency, and 53% included information about gender identity and sexual orientation.
Reported barriers to curriculum development in 132 programs that did not have a health disparities curriculum included lack of time in the current curriculum, insufficient faculty skill to teach the topic, lack of institutional support, and lack of faculty interest, the researchers noted.
A total of 13,251 residents (70%) reported receiving some training in caring for patients at risk for health disparities over 3 years of training, and 10,494 (80%) of these rated the quality as very good or excellent. “Residents who cared for a larger proportion of underserved patients perceived that they received health disparities training at a higher rate,” the researchers wrote. However, increased care of at-risk populations does not necessarily translate into increased knowledge and skills. “Our finding that residents’ rating of the quality of their training was not associated with the presence of a curriculum in health disparities in their program also raises a concern that perceptions may overestimate the acquisition of needed skills,” they added.
The major limitation of the study was “that residents were not asked directly if they were exposed to a curriculum in health disparities but rather if they received training in the care of patients who would be at risk, which raises the concern that we cannot distinguish between their recognition of a formal and informal curriculum,” the researchers noted. In addition, the survey could not confirm that program directors were aware of all training. “Furthermore, because the survey items were embedded in larger program director survey, we were limited in the ability to ask them to define more specifically the components of their health disparities curricula,” they wrote.
However, the results were strengthened by the large and comprehensive study population, and highlight not only the need for standardized health disparities curricula, but also the need for research to determine the most effective domains for such curricula in graduate medical education, they emphasized.
“There are opportunities to explore partnerships among residencies, institutional clinical practices, and communities for productive collaborations around disparities-related quality improvement projects to address gaps in health care that are specific to the populations they serve,” they concluded.
The surveys were conducted in 2015 and the comparative work in 2018, prior to the COVID-19 pandemic and the subsequent increased concerns about disparities in health care, Dr. Dupras said in an interview.
“We conducted the survey because we recognized that health disparities were still prevalent in our society despite calls to improve the education of our learners to address them. We wanted to determine what our programs were providing for educational curriculum and what our learners were experiencing,” she said.
“We did not know what the surveys would show, so I cannot say that we were surprised by the findings,” said Dr. Dupras. “One of the challenges in interpreting our results is inherent in studies that rely on surveys. We cannot know how those filling out the surveys interpret the questions.” The study results yield several messages.
“First, residency training programs have opportunities to do a better job in developing educational opportunities related to health disparities; second, residents learn in the context of care and we must optimize education around these experiences; third, every patient is different. It is time to move towards cultural humility, since the risk for disparities is not associated with one patient characteristic, but composed of multiple factors,” she said.
“Given that 5 years has passed since our original survey, it would be important to repeat the survey and consider expanding it to include other training programs that provide frontline care, such as family medicine and pediatrics,” Dr. Dupras noted.
Dr. Dupras and colleagues had no financial conflicts to disclose.
SOURCE: Dupras DM et al. JAMA Netw Open. 2020 Aug 10. doi: 10.1001/jamanetworkopen.2020.12757.
FROM JAMA NETWORK OPEN
Educational intervention curbs use of antibiotics for respiratory infections
A clinician education program significantly reduced overall antibiotic prescribing during pediatric visits for acute respiratory tract infections, according to data from 57 clinicians who participated in an intervention.
In a study published in Pediatrics, Matthew P. Kronman, MD, of the University of Washington, Seattle, and associates randomized 57 clinicians at 19 pediatric practices to a stepped-wedge clinical trial. The study included visits for acute otitis media, bronchitis, pharyngitis, sinusitis, and upper respiratory infections (defined as ARTI visits) for children aged 6 months to less than 11 years, for a total of 72,723 ARTI visits by 29,762 patients. The primary outcome was overall antibiotic prescribing for ARTI visits.
For the intervention, known as the Dialogue Around Respiratory Illness Treatment (DART) quality improvement (QI) program, clinicians received three program modules containing online tutorials and webinars. These professionally-produced modules included a combination of evidence-based communication strategies and antibiotic prescribing, booster video vignettes, and individualized antibiotic prescribing feedback reports over 11 months.
Overall, the probability of antibiotic prescribing for ARTI visits decreased by 7% (adjusted relative risk 0.93) from baseline to a 2- to 8-month postintervention in an adjusted intent-to-treat analysis.
Analysis of secondary outcomes revealed that prescribing any antibiotics for viral ARTI decreased by 40% during the postintervention period compared to baseline (aRR 0.60).
In addition, second-line antibiotic prescribing decreased from baseline by 34% for streptococcal pharyngitis (aRR 0.66), and by 41% for sinusitis (aRR 0.59); however there was no significant change in prescribing for acute otitis media, the researchers said.
The study findings were limited by several factors including the potential for biased results because of the randomization of clinicians from multiple practices and the potential for clinicians to change their prescribing habits after the start of the study, Dr. Kronman and colleagues noted.
In addition, the study did not include complete data on rapid streptococcal antigen testing, which might eliminate some children from the study population, and the relatively short postintervention period “may not represent the true long-term intervention durability may not represent the true long-term intervention durability,” they said.
However, the results support the potential of the DART program. “The 7% reduction in antibiotic prescribing for all ARTIs, if extrapolated to all ambulatory ARTI visits to pediatricians nationally, would represent 1.5 million fewer antibiotic prescriptions for children with ARTI annually,” they wrote.
“Providing online communication training and evidence-based antibiotic prescribing education in combination with individualized antibiotic prescribing feedback reports may help achieve national goals of reducing unnecessary outpatient antibiotic prescribing for children,” Dr. Kronman and associates concluded.
Combining interventions are key to reducing unnecessary antibiotics use in pediatric ambulatory care, Rana F. Hamdy, MD, MPH, of Children’s National Hospital, Washington, , and Sophie E. Katz, MD, of Vanderbilt University, Nashville, Tenn., wrote in an accompanying editorial (Pediatrics. 2020 Aug 3. doi: 10.1542/peds.2020-012922).
The researchers in the current study “seem to recognize that clinicians are adult learners, and they combine interventions to implement these adult learning theory tenets to improve appropriate antibiotic prescribing,” they wrote. The DART intervention combined best practices training, communications training, and individualized antibiotic prescribing feedback reports to improve communication between providers and families “especially when faced with a situation in which a parent or guardian might expect an antibiotic prescription but the provider does not think one is necessary,” Dr. Hamdy and Dr. Katz said.
Overall, the findings suggest that the interventions work best in combination vs. being used alone, although the study did not evaluate the separate contributions of each intervention, the editorialists wrote.
“In the current study, nonengaged physicians had an increase in second-line antibiotic prescribing, whereas the engaged physicians had a decrease in second-line antibiotic prescribing,” they noted. “This suggests that the addition of communications training could mitigate the undesirable effects that may result from solely using feedback reports.”
“Each year, U.S. children are prescribed as many as 10 million unnecessary antibiotic courses for acute respiratory tract infections,” Kristina A. Bryant, MD, of the University of Louisville, Ky., said in an interview. “Some of these prescriptions result in side effects or allergic reactions, and they contribute to growing antibiotic resistance. We need effective interventions to reduce antibiotic prescribing.”
Although the DART modules are free and available online, busy clinicians might struggle to find time to view them consistently, said Dr. Bryant.
“One advantage of the study design was that information was pushed to clinicians along with communication booster videos,” she said. “We know that education and reinforcement over time works better than a one and done approach.
“Study participants also received feedback over time about their prescribing habits, which can be a powerful motivator for change, although not all clinicians may have easy access to these reports,” she noted.
To overcome some of the barriers to using the modules, clinicians who are “interested in improving their prescribing could work with their office managers to develop antibiotic prescribing reports and schedule reminders to review them,” said Dr. Bryant.
“An individual could commit to education and review of his or her own prescribing patterns, but support from one’s partners and shared accountability is likely to be even more effective,” she said. “Sharing data within a practice and exploring differences in prescribing patterns can drive improvement.
“Spaced education and regular feedback about prescribing patterns can improve antibiotic prescribing for pharyngitis and sinusitis, and reduce antibiotic prescriptions for ARTIs,” Dr. Bryant said. The take-home from the study is that it should prompt anyone who prescribes antibiotics for children to ask themselves how they can improve their own prescribing habits.
“In this study, prescribing for viral ARTIs was reduced but not eliminated. We need additional studies to further reduce unnecessary antibiotic use,” Dr. Bryant said.
In addition, areas for future research could include longer-term follow-up. “Study participants were followed for 2 to 8 months after the intervention ended in June 2018. It would be interesting to know about their prescribing practices now, and if the changes observed in the study were durable,” she concluded.
The study was supported by the National Institutes of Health, along with additional infrastructure funding from the American Academy of Pediatrics and the Health Resources and Services Administration of the Department of Health and Human Services. The researchers had no financial conflicts to disclose.
Dr. Hamdy and Dr. Katz had no financial conflicts to disclose, but Dr. Katz disclosed grant support through the Centers for Disease Control and Prevention as a recipient of the Leadership in Epidemiology, Antimicrobial Stewardship, and Public Health fellowship, sponsored by the Society for Healthcare Epidemiology of America, Infectious Diseases Society of America, and Pediatric Infectious Diseases Society.
Dr. Bryant disclosed serving as an investigator on multicenter clinical vaccine trials funded by Pfizer (but not in the last year). She also serves as the current president of the Pediatric Infectious Diseases Society, but the opinions expressed here are her own and do not necessarily reflect the views of PIDS.
SOURCE: Kronman MP et al. Pediatrics. 2020 Aug 3. doi: 10.1542/peds.2020-0038.
A clinician education program significantly reduced overall antibiotic prescribing during pediatric visits for acute respiratory tract infections, according to data from 57 clinicians who participated in an intervention.
In a study published in Pediatrics, Matthew P. Kronman, MD, of the University of Washington, Seattle, and associates randomized 57 clinicians at 19 pediatric practices to a stepped-wedge clinical trial. The study included visits for acute otitis media, bronchitis, pharyngitis, sinusitis, and upper respiratory infections (defined as ARTI visits) for children aged 6 months to less than 11 years, for a total of 72,723 ARTI visits by 29,762 patients. The primary outcome was overall antibiotic prescribing for ARTI visits.
For the intervention, known as the Dialogue Around Respiratory Illness Treatment (DART) quality improvement (QI) program, clinicians received three program modules containing online tutorials and webinars. These professionally-produced modules included a combination of evidence-based communication strategies and antibiotic prescribing, booster video vignettes, and individualized antibiotic prescribing feedback reports over 11 months.
Overall, the probability of antibiotic prescribing for ARTI visits decreased by 7% (adjusted relative risk 0.93) from baseline to a 2- to 8-month postintervention in an adjusted intent-to-treat analysis.
Analysis of secondary outcomes revealed that prescribing any antibiotics for viral ARTI decreased by 40% during the postintervention period compared to baseline (aRR 0.60).
In addition, second-line antibiotic prescribing decreased from baseline by 34% for streptococcal pharyngitis (aRR 0.66), and by 41% for sinusitis (aRR 0.59); however there was no significant change in prescribing for acute otitis media, the researchers said.
The study findings were limited by several factors including the potential for biased results because of the randomization of clinicians from multiple practices and the potential for clinicians to change their prescribing habits after the start of the study, Dr. Kronman and colleagues noted.
In addition, the study did not include complete data on rapid streptococcal antigen testing, which might eliminate some children from the study population, and the relatively short postintervention period “may not represent the true long-term intervention durability may not represent the true long-term intervention durability,” they said.
However, the results support the potential of the DART program. “The 7% reduction in antibiotic prescribing for all ARTIs, if extrapolated to all ambulatory ARTI visits to pediatricians nationally, would represent 1.5 million fewer antibiotic prescriptions for children with ARTI annually,” they wrote.
“Providing online communication training and evidence-based antibiotic prescribing education in combination with individualized antibiotic prescribing feedback reports may help achieve national goals of reducing unnecessary outpatient antibiotic prescribing for children,” Dr. Kronman and associates concluded.
Combining interventions are key to reducing unnecessary antibiotics use in pediatric ambulatory care, Rana F. Hamdy, MD, MPH, of Children’s National Hospital, Washington, , and Sophie E. Katz, MD, of Vanderbilt University, Nashville, Tenn., wrote in an accompanying editorial (Pediatrics. 2020 Aug 3. doi: 10.1542/peds.2020-012922).
The researchers in the current study “seem to recognize that clinicians are adult learners, and they combine interventions to implement these adult learning theory tenets to improve appropriate antibiotic prescribing,” they wrote. The DART intervention combined best practices training, communications training, and individualized antibiotic prescribing feedback reports to improve communication between providers and families “especially when faced with a situation in which a parent or guardian might expect an antibiotic prescription but the provider does not think one is necessary,” Dr. Hamdy and Dr. Katz said.
Overall, the findings suggest that the interventions work best in combination vs. being used alone, although the study did not evaluate the separate contributions of each intervention, the editorialists wrote.
“In the current study, nonengaged physicians had an increase in second-line antibiotic prescribing, whereas the engaged physicians had a decrease in second-line antibiotic prescribing,” they noted. “This suggests that the addition of communications training could mitigate the undesirable effects that may result from solely using feedback reports.”
“Each year, U.S. children are prescribed as many as 10 million unnecessary antibiotic courses for acute respiratory tract infections,” Kristina A. Bryant, MD, of the University of Louisville, Ky., said in an interview. “Some of these prescriptions result in side effects or allergic reactions, and they contribute to growing antibiotic resistance. We need effective interventions to reduce antibiotic prescribing.”
Although the DART modules are free and available online, busy clinicians might struggle to find time to view them consistently, said Dr. Bryant.
“One advantage of the study design was that information was pushed to clinicians along with communication booster videos,” she said. “We know that education and reinforcement over time works better than a one and done approach.
“Study participants also received feedback over time about their prescribing habits, which can be a powerful motivator for change, although not all clinicians may have easy access to these reports,” she noted.
To overcome some of the barriers to using the modules, clinicians who are “interested in improving their prescribing could work with their office managers to develop antibiotic prescribing reports and schedule reminders to review them,” said Dr. Bryant.
“An individual could commit to education and review of his or her own prescribing patterns, but support from one’s partners and shared accountability is likely to be even more effective,” she said. “Sharing data within a practice and exploring differences in prescribing patterns can drive improvement.
“Spaced education and regular feedback about prescribing patterns can improve antibiotic prescribing for pharyngitis and sinusitis, and reduce antibiotic prescriptions for ARTIs,” Dr. Bryant said. The take-home from the study is that it should prompt anyone who prescribes antibiotics for children to ask themselves how they can improve their own prescribing habits.
“In this study, prescribing for viral ARTIs was reduced but not eliminated. We need additional studies to further reduce unnecessary antibiotic use,” Dr. Bryant said.
In addition, areas for future research could include longer-term follow-up. “Study participants were followed for 2 to 8 months after the intervention ended in June 2018. It would be interesting to know about their prescribing practices now, and if the changes observed in the study were durable,” she concluded.
The study was supported by the National Institutes of Health, along with additional infrastructure funding from the American Academy of Pediatrics and the Health Resources and Services Administration of the Department of Health and Human Services. The researchers had no financial conflicts to disclose.
Dr. Hamdy and Dr. Katz had no financial conflicts to disclose, but Dr. Katz disclosed grant support through the Centers for Disease Control and Prevention as a recipient of the Leadership in Epidemiology, Antimicrobial Stewardship, and Public Health fellowship, sponsored by the Society for Healthcare Epidemiology of America, Infectious Diseases Society of America, and Pediatric Infectious Diseases Society.
Dr. Bryant disclosed serving as an investigator on multicenter clinical vaccine trials funded by Pfizer (but not in the last year). She also serves as the current president of the Pediatric Infectious Diseases Society, but the opinions expressed here are her own and do not necessarily reflect the views of PIDS.
SOURCE: Kronman MP et al. Pediatrics. 2020 Aug 3. doi: 10.1542/peds.2020-0038.
A clinician education program significantly reduced overall antibiotic prescribing during pediatric visits for acute respiratory tract infections, according to data from 57 clinicians who participated in an intervention.
In a study published in Pediatrics, Matthew P. Kronman, MD, of the University of Washington, Seattle, and associates randomized 57 clinicians at 19 pediatric practices to a stepped-wedge clinical trial. The study included visits for acute otitis media, bronchitis, pharyngitis, sinusitis, and upper respiratory infections (defined as ARTI visits) for children aged 6 months to less than 11 years, for a total of 72,723 ARTI visits by 29,762 patients. The primary outcome was overall antibiotic prescribing for ARTI visits.
For the intervention, known as the Dialogue Around Respiratory Illness Treatment (DART) quality improvement (QI) program, clinicians received three program modules containing online tutorials and webinars. These professionally-produced modules included a combination of evidence-based communication strategies and antibiotic prescribing, booster video vignettes, and individualized antibiotic prescribing feedback reports over 11 months.
Overall, the probability of antibiotic prescribing for ARTI visits decreased by 7% (adjusted relative risk 0.93) from baseline to a 2- to 8-month postintervention in an adjusted intent-to-treat analysis.
Analysis of secondary outcomes revealed that prescribing any antibiotics for viral ARTI decreased by 40% during the postintervention period compared to baseline (aRR 0.60).
In addition, second-line antibiotic prescribing decreased from baseline by 34% for streptococcal pharyngitis (aRR 0.66), and by 41% for sinusitis (aRR 0.59); however there was no significant change in prescribing for acute otitis media, the researchers said.
The study findings were limited by several factors including the potential for biased results because of the randomization of clinicians from multiple practices and the potential for clinicians to change their prescribing habits after the start of the study, Dr. Kronman and colleagues noted.
In addition, the study did not include complete data on rapid streptococcal antigen testing, which might eliminate some children from the study population, and the relatively short postintervention period “may not represent the true long-term intervention durability may not represent the true long-term intervention durability,” they said.
However, the results support the potential of the DART program. “The 7% reduction in antibiotic prescribing for all ARTIs, if extrapolated to all ambulatory ARTI visits to pediatricians nationally, would represent 1.5 million fewer antibiotic prescriptions for children with ARTI annually,” they wrote.
“Providing online communication training and evidence-based antibiotic prescribing education in combination with individualized antibiotic prescribing feedback reports may help achieve national goals of reducing unnecessary outpatient antibiotic prescribing for children,” Dr. Kronman and associates concluded.
Combining interventions are key to reducing unnecessary antibiotics use in pediatric ambulatory care, Rana F. Hamdy, MD, MPH, of Children’s National Hospital, Washington, , and Sophie E. Katz, MD, of Vanderbilt University, Nashville, Tenn., wrote in an accompanying editorial (Pediatrics. 2020 Aug 3. doi: 10.1542/peds.2020-012922).
The researchers in the current study “seem to recognize that clinicians are adult learners, and they combine interventions to implement these adult learning theory tenets to improve appropriate antibiotic prescribing,” they wrote. The DART intervention combined best practices training, communications training, and individualized antibiotic prescribing feedback reports to improve communication between providers and families “especially when faced with a situation in which a parent or guardian might expect an antibiotic prescription but the provider does not think one is necessary,” Dr. Hamdy and Dr. Katz said.
Overall, the findings suggest that the interventions work best in combination vs. being used alone, although the study did not evaluate the separate contributions of each intervention, the editorialists wrote.
“In the current study, nonengaged physicians had an increase in second-line antibiotic prescribing, whereas the engaged physicians had a decrease in second-line antibiotic prescribing,” they noted. “This suggests that the addition of communications training could mitigate the undesirable effects that may result from solely using feedback reports.”
“Each year, U.S. children are prescribed as many as 10 million unnecessary antibiotic courses for acute respiratory tract infections,” Kristina A. Bryant, MD, of the University of Louisville, Ky., said in an interview. “Some of these prescriptions result in side effects or allergic reactions, and they contribute to growing antibiotic resistance. We need effective interventions to reduce antibiotic prescribing.”
Although the DART modules are free and available online, busy clinicians might struggle to find time to view them consistently, said Dr. Bryant.
“One advantage of the study design was that information was pushed to clinicians along with communication booster videos,” she said. “We know that education and reinforcement over time works better than a one and done approach.
“Study participants also received feedback over time about their prescribing habits, which can be a powerful motivator for change, although not all clinicians may have easy access to these reports,” she noted.
To overcome some of the barriers to using the modules, clinicians who are “interested in improving their prescribing could work with their office managers to develop antibiotic prescribing reports and schedule reminders to review them,” said Dr. Bryant.
“An individual could commit to education and review of his or her own prescribing patterns, but support from one’s partners and shared accountability is likely to be even more effective,” she said. “Sharing data within a practice and exploring differences in prescribing patterns can drive improvement.
“Spaced education and regular feedback about prescribing patterns can improve antibiotic prescribing for pharyngitis and sinusitis, and reduce antibiotic prescriptions for ARTIs,” Dr. Bryant said. The take-home from the study is that it should prompt anyone who prescribes antibiotics for children to ask themselves how they can improve their own prescribing habits.
“In this study, prescribing for viral ARTIs was reduced but not eliminated. We need additional studies to further reduce unnecessary antibiotic use,” Dr. Bryant said.
In addition, areas for future research could include longer-term follow-up. “Study participants were followed for 2 to 8 months after the intervention ended in June 2018. It would be interesting to know about their prescribing practices now, and if the changes observed in the study were durable,” she concluded.
The study was supported by the National Institutes of Health, along with additional infrastructure funding from the American Academy of Pediatrics and the Health Resources and Services Administration of the Department of Health and Human Services. The researchers had no financial conflicts to disclose.
Dr. Hamdy and Dr. Katz had no financial conflicts to disclose, but Dr. Katz disclosed grant support through the Centers for Disease Control and Prevention as a recipient of the Leadership in Epidemiology, Antimicrobial Stewardship, and Public Health fellowship, sponsored by the Society for Healthcare Epidemiology of America, Infectious Diseases Society of America, and Pediatric Infectious Diseases Society.
Dr. Bryant disclosed serving as an investigator on multicenter clinical vaccine trials funded by Pfizer (but not in the last year). She also serves as the current president of the Pediatric Infectious Diseases Society, but the opinions expressed here are her own and do not necessarily reflect the views of PIDS.
SOURCE: Kronman MP et al. Pediatrics. 2020 Aug 3. doi: 10.1542/peds.2020-0038.
FROM PEDIATRICS
Many children with COVID-19 present without classic symptoms
Most children who tested positive for SARS-CoV-2 had no respiratory illness, according to data from a retrospective study of 22 patients at a single center.
To date, children account for less than 5% of COVID-19 cases in the United States, but details of the clinical presentations in children are limited, wrote Rabia Agha, MD, and colleagues of Maimonides Children’s Hospital, Brooklyn, N.Y.
In a study published in Hospital Pediatrics, the researchers reviewed data from 22 children aged 0-18 years who tested positive for SARS-CoV-2 by polymerase chain reaction (PCR) and were admitted to a single hospital over a 4-week period from March 18, 2020, to April 15, 2020.
Of four patients requiring mechanical ventilation, two had underlying pulmonary disease. The other two patients who required intubation were one with cerebral palsy and status epilepticus and one who presented in a state of cardiac arrest.
The study population ranged from 11 days to 18 years of age, but 45% were infants younger than 1 year. None of the children had a travel history that might increase their risk for SARS-CoV-2 infection; 27% had confirmed exposure to the virus.
Most of the children (82%) were hospitalized within 3 days of the onset of symptoms, and no deaths occurred during the study period. The most common symptom was fever without a source in five (23%) otherwise healthy infants aged 11-35 days. All five of these children underwent a sepsis evaluation, received empiric antibiotics, and were discharged home with negative bacterial cultures within 48-72 hours. Another 10 children had fever in combination with other symptoms.
Other presenting symptoms were respiratory (9), fatigue (6), seizures (2), and headache (1).
Most children with respiratory illness were treated with supportive therapy and antibiotics, but three of those on mechanical ventilation also were treated with remdesivir; all three were ultimately extubated.
Neurological abnormalities occurred in two patients: an 11-year-old otherwise healthy boy who presented with fever, headache, confusion, and seizure but ultimately improved without short-term sequelae; and a 12-year-old girl with cerebral palsy who developed new onset seizures and required mechanical ventilation, but ultimately improved to baseline.
Positive PCR results were identified in seven patients (32%) during the second half of the study period who were initially hospitalized for non-COVID related symptoms; four with bacterial infections, two with illnesses of unknown etiology, and one with cardiac arrest. Another two children were completely asymptomatic at the time of admission but then tested positive by PCR; one child had been admitted for routine chemotherapy and the other for social reasons, Dr. Agha and associates said.
The study findings contrast with early data from China in which respiratory illness of varying severity was the major presentation in children with COVID-19, but support a more recent meta-analysis of 551 cases, the researchers noted. The findings also highlight the value of universal testing for children.
“Our initial testing strategy was according to the federal and local guidelines that recommended PCR testing for the symptoms of fever, cough and shortness of breath, or travel to certain countries or close contact with a confirmed case,” Dr. Agha and colleagues said.
“With the implementation of our universal screening strategy of all admitted pediatric patients, we identified 9 (41%) patients with COVID-19 that would have been missed, as they did not meet the then-recommended criteria for testing,” they wrote.
The results suggest the need for broader guidelines to test pediatric patients because children presenting with other illnesses may be positive for SARS-CoV-2 as well, the researchers said.
“Testing of all hospitalized patients will not only identify cases early in the course of their admission process, but will also help prevent inadvertent exposure of other patients and health care workers, assist in cohorting infected patients, and aid in conservation of personal protective equipment,” Dr. Agha and associates concluded.
The current study is important as clinicians continue to learn about how infection with SARS-CoV-2 presents in different populations, Diana Lee, MD, of the Icahn School of Medicine at Mount Sinai, New York, said in an interview.
“Understanding how it can present in the pediatric population is important in identifying children who may have the infection and developing strategies for testing,” she said.
“I was not surprised by the finding that most children did not present with the classic symptoms of COVID-19 in adults based on other published studies and my personal clinical experience taking care of hospitalized children in New York City,” said Dr. Lee. “Studies from the U.S. and other countries have reported that fewer children experience fever, cough, and shortness of breath [compared with] adults, and that most children have a milder clinical course, though there is a small percentage of children who can have severe or critical illness,” she said.
“A multisystem inflammatory syndrome in children associated with COVID-19 has also emerged and appears to be a postinfectious process with a presentation that often differs from classic COVID-19 infection in adults,” she added.
The take-home message for clinicians is the reminder that SARS-CoV-2 infection often presents differently in children than in adults, said Dr. Lee.
“Children who present to the hospital with non-classic COVID-19 symptoms or with other diagnoses may be positive for SARS-CoV-2 on testing. Broadly testing hospitalized children for SARS-CoV-2 and instituting appropriate isolation precautions may help to protect other individuals from being exposed to the virus,” she said.
“Further research is needed to understand which individuals are contagious and how to accurately distinguish those who are infectious versus those who are not,” said Dr. Lee. “There have been individuals who persistently test positive for SARS-CoV-2 RNA (the genetic material of the virus), but were not found to have virus in their bodies that can replicate and thereby infect others,” she emphasized. “Further study is needed regarding the likelihood of household exposures in children with SARS-CoV-2 infection given that this study was done early in the epidemic in New York City when testing and contact tracing was less established,” she said.
The study received no outside funding. The researchers had no financial conflicts to disclose. Dr. Lee had no financial conflicts to disclose.
SOURCE: Agha R et al. Hosp Pediatr. 2020 July. doi: 10.1542/hpeds.2020-000257.
Most children who tested positive for SARS-CoV-2 had no respiratory illness, according to data from a retrospective study of 22 patients at a single center.
To date, children account for less than 5% of COVID-19 cases in the United States, but details of the clinical presentations in children are limited, wrote Rabia Agha, MD, and colleagues of Maimonides Children’s Hospital, Brooklyn, N.Y.
In a study published in Hospital Pediatrics, the researchers reviewed data from 22 children aged 0-18 years who tested positive for SARS-CoV-2 by polymerase chain reaction (PCR) and were admitted to a single hospital over a 4-week period from March 18, 2020, to April 15, 2020.
Of four patients requiring mechanical ventilation, two had underlying pulmonary disease. The other two patients who required intubation were one with cerebral palsy and status epilepticus and one who presented in a state of cardiac arrest.
The study population ranged from 11 days to 18 years of age, but 45% were infants younger than 1 year. None of the children had a travel history that might increase their risk for SARS-CoV-2 infection; 27% had confirmed exposure to the virus.
Most of the children (82%) were hospitalized within 3 days of the onset of symptoms, and no deaths occurred during the study period. The most common symptom was fever without a source in five (23%) otherwise healthy infants aged 11-35 days. All five of these children underwent a sepsis evaluation, received empiric antibiotics, and were discharged home with negative bacterial cultures within 48-72 hours. Another 10 children had fever in combination with other symptoms.
Other presenting symptoms were respiratory (9), fatigue (6), seizures (2), and headache (1).
Most children with respiratory illness were treated with supportive therapy and antibiotics, but three of those on mechanical ventilation also were treated with remdesivir; all three were ultimately extubated.
Neurological abnormalities occurred in two patients: an 11-year-old otherwise healthy boy who presented with fever, headache, confusion, and seizure but ultimately improved without short-term sequelae; and a 12-year-old girl with cerebral palsy who developed new onset seizures and required mechanical ventilation, but ultimately improved to baseline.
Positive PCR results were identified in seven patients (32%) during the second half of the study period who were initially hospitalized for non-COVID related symptoms; four with bacterial infections, two with illnesses of unknown etiology, and one with cardiac arrest. Another two children were completely asymptomatic at the time of admission but then tested positive by PCR; one child had been admitted for routine chemotherapy and the other for social reasons, Dr. Agha and associates said.
The study findings contrast with early data from China in which respiratory illness of varying severity was the major presentation in children with COVID-19, but support a more recent meta-analysis of 551 cases, the researchers noted. The findings also highlight the value of universal testing for children.
“Our initial testing strategy was according to the federal and local guidelines that recommended PCR testing for the symptoms of fever, cough and shortness of breath, or travel to certain countries or close contact with a confirmed case,” Dr. Agha and colleagues said.
“With the implementation of our universal screening strategy of all admitted pediatric patients, we identified 9 (41%) patients with COVID-19 that would have been missed, as they did not meet the then-recommended criteria for testing,” they wrote.
The results suggest the need for broader guidelines to test pediatric patients because children presenting with other illnesses may be positive for SARS-CoV-2 as well, the researchers said.
“Testing of all hospitalized patients will not only identify cases early in the course of their admission process, but will also help prevent inadvertent exposure of other patients and health care workers, assist in cohorting infected patients, and aid in conservation of personal protective equipment,” Dr. Agha and associates concluded.
The current study is important as clinicians continue to learn about how infection with SARS-CoV-2 presents in different populations, Diana Lee, MD, of the Icahn School of Medicine at Mount Sinai, New York, said in an interview.
“Understanding how it can present in the pediatric population is important in identifying children who may have the infection and developing strategies for testing,” she said.
“I was not surprised by the finding that most children did not present with the classic symptoms of COVID-19 in adults based on other published studies and my personal clinical experience taking care of hospitalized children in New York City,” said Dr. Lee. “Studies from the U.S. and other countries have reported that fewer children experience fever, cough, and shortness of breath [compared with] adults, and that most children have a milder clinical course, though there is a small percentage of children who can have severe or critical illness,” she said.
“A multisystem inflammatory syndrome in children associated with COVID-19 has also emerged and appears to be a postinfectious process with a presentation that often differs from classic COVID-19 infection in adults,” she added.
The take-home message for clinicians is the reminder that SARS-CoV-2 infection often presents differently in children than in adults, said Dr. Lee.
“Children who present to the hospital with non-classic COVID-19 symptoms or with other diagnoses may be positive for SARS-CoV-2 on testing. Broadly testing hospitalized children for SARS-CoV-2 and instituting appropriate isolation precautions may help to protect other individuals from being exposed to the virus,” she said.
“Further research is needed to understand which individuals are contagious and how to accurately distinguish those who are infectious versus those who are not,” said Dr. Lee. “There have been individuals who persistently test positive for SARS-CoV-2 RNA (the genetic material of the virus), but were not found to have virus in their bodies that can replicate and thereby infect others,” she emphasized. “Further study is needed regarding the likelihood of household exposures in children with SARS-CoV-2 infection given that this study was done early in the epidemic in New York City when testing and contact tracing was less established,” she said.
The study received no outside funding. The researchers had no financial conflicts to disclose. Dr. Lee had no financial conflicts to disclose.
SOURCE: Agha R et al. Hosp Pediatr. 2020 July. doi: 10.1542/hpeds.2020-000257.
Most children who tested positive for SARS-CoV-2 had no respiratory illness, according to data from a retrospective study of 22 patients at a single center.
To date, children account for less than 5% of COVID-19 cases in the United States, but details of the clinical presentations in children are limited, wrote Rabia Agha, MD, and colleagues of Maimonides Children’s Hospital, Brooklyn, N.Y.
In a study published in Hospital Pediatrics, the researchers reviewed data from 22 children aged 0-18 years who tested positive for SARS-CoV-2 by polymerase chain reaction (PCR) and were admitted to a single hospital over a 4-week period from March 18, 2020, to April 15, 2020.
Of four patients requiring mechanical ventilation, two had underlying pulmonary disease. The other two patients who required intubation were one with cerebral palsy and status epilepticus and one who presented in a state of cardiac arrest.
The study population ranged from 11 days to 18 years of age, but 45% were infants younger than 1 year. None of the children had a travel history that might increase their risk for SARS-CoV-2 infection; 27% had confirmed exposure to the virus.
Most of the children (82%) were hospitalized within 3 days of the onset of symptoms, and no deaths occurred during the study period. The most common symptom was fever without a source in five (23%) otherwise healthy infants aged 11-35 days. All five of these children underwent a sepsis evaluation, received empiric antibiotics, and were discharged home with negative bacterial cultures within 48-72 hours. Another 10 children had fever in combination with other symptoms.
Other presenting symptoms were respiratory (9), fatigue (6), seizures (2), and headache (1).
Most children with respiratory illness were treated with supportive therapy and antibiotics, but three of those on mechanical ventilation also were treated with remdesivir; all three were ultimately extubated.
Neurological abnormalities occurred in two patients: an 11-year-old otherwise healthy boy who presented with fever, headache, confusion, and seizure but ultimately improved without short-term sequelae; and a 12-year-old girl with cerebral palsy who developed new onset seizures and required mechanical ventilation, but ultimately improved to baseline.
Positive PCR results were identified in seven patients (32%) during the second half of the study period who were initially hospitalized for non-COVID related symptoms; four with bacterial infections, two with illnesses of unknown etiology, and one with cardiac arrest. Another two children were completely asymptomatic at the time of admission but then tested positive by PCR; one child had been admitted for routine chemotherapy and the other for social reasons, Dr. Agha and associates said.
The study findings contrast with early data from China in which respiratory illness of varying severity was the major presentation in children with COVID-19, but support a more recent meta-analysis of 551 cases, the researchers noted. The findings also highlight the value of universal testing for children.
“Our initial testing strategy was according to the federal and local guidelines that recommended PCR testing for the symptoms of fever, cough and shortness of breath, or travel to certain countries or close contact with a confirmed case,” Dr. Agha and colleagues said.
“With the implementation of our universal screening strategy of all admitted pediatric patients, we identified 9 (41%) patients with COVID-19 that would have been missed, as they did not meet the then-recommended criteria for testing,” they wrote.
The results suggest the need for broader guidelines to test pediatric patients because children presenting with other illnesses may be positive for SARS-CoV-2 as well, the researchers said.
“Testing of all hospitalized patients will not only identify cases early in the course of their admission process, but will also help prevent inadvertent exposure of other patients and health care workers, assist in cohorting infected patients, and aid in conservation of personal protective equipment,” Dr. Agha and associates concluded.
The current study is important as clinicians continue to learn about how infection with SARS-CoV-2 presents in different populations, Diana Lee, MD, of the Icahn School of Medicine at Mount Sinai, New York, said in an interview.
“Understanding how it can present in the pediatric population is important in identifying children who may have the infection and developing strategies for testing,” she said.
“I was not surprised by the finding that most children did not present with the classic symptoms of COVID-19 in adults based on other published studies and my personal clinical experience taking care of hospitalized children in New York City,” said Dr. Lee. “Studies from the U.S. and other countries have reported that fewer children experience fever, cough, and shortness of breath [compared with] adults, and that most children have a milder clinical course, though there is a small percentage of children who can have severe or critical illness,” she said.
“A multisystem inflammatory syndrome in children associated with COVID-19 has also emerged and appears to be a postinfectious process with a presentation that often differs from classic COVID-19 infection in adults,” she added.
The take-home message for clinicians is the reminder that SARS-CoV-2 infection often presents differently in children than in adults, said Dr. Lee.
“Children who present to the hospital with non-classic COVID-19 symptoms or with other diagnoses may be positive for SARS-CoV-2 on testing. Broadly testing hospitalized children for SARS-CoV-2 and instituting appropriate isolation precautions may help to protect other individuals from being exposed to the virus,” she said.
“Further research is needed to understand which individuals are contagious and how to accurately distinguish those who are infectious versus those who are not,” said Dr. Lee. “There have been individuals who persistently test positive for SARS-CoV-2 RNA (the genetic material of the virus), but were not found to have virus in their bodies that can replicate and thereby infect others,” she emphasized. “Further study is needed regarding the likelihood of household exposures in children with SARS-CoV-2 infection given that this study was done early in the epidemic in New York City when testing and contact tracing was less established,” she said.
The study received no outside funding. The researchers had no financial conflicts to disclose. Dr. Lee had no financial conflicts to disclose.
SOURCE: Agha R et al. Hosp Pediatr. 2020 July. doi: 10.1542/hpeds.2020-000257.
FROM HOSPITAL PEDIATRICS
Don’t discount discharge planning during pandemic
The COVID-19 pandemic continues to disrupt all aspects of hospital care, and has altered nearly all fundamental practices, including discharge protocols. A session presented at the Society of Hospitalist Medicine’s 2020 Virtual Annual Conference will focus on discharge issues in the COVID-19 era.
“Discharge planning is an integral part of a hospitalist’s clinical care. On a daily basis, we think carefully about how to help our patients safely transition back into life outside of the hospital,” said Maralyssa Bann, MD, of the University of Washington, Seattle, a copresenter at the session.
“Patients need up-to-date information about how to keep themselves and those around them safe,” she said. “They need resources and supports to help them recover from illness.”
These supports include access to appropriate follow-up with primary care doctors or other specialists and being discharged to the right location, such as home or a skilled nursing facility, Dr. Bann noted.
In response to COVID-19, “within an exceptionally short time frame, hospitals have had to rapidly adapt their discharge planning protocols and have had to continue to adapt as new information comes out,” Dr. Bann said.
“In many ways, the COVID-19 pandemic has highlighted the importance of discharge planning for patient care and has added a new element of public health in that we have to take all possible precautions to ensure that patients are not spreading the virus after they leave the hospital,” said Ryan Greysen, MD, of the University of Pennsylvania, Philadelphia, and Dr. Bann’s copresenter.
Many elements go into creating a good discharge plan, but there are often many unknowns, Dr. Greysen said. “I think there is an opportunity to improve the process by improving follow-up as well.”
“For example, one program at our hospital focused on vulnerable older adults includes an in-home visit by a visiting nurse on the day of discharge to verify the patient has everything they need when they arrive home,” However, now with more telemedicine and social distancing, there should be creative approaches to tying up loose ends and monitoring for things that can go wrong in order to give additional guidance, he said.
“In a previous study of 12 U.S. academic medical centers, my colleagues and I interviewed over 1,000 patients who were discharged and then readmitted to ask them what they thought went wrong,” said Dr. Greysen. “Overwhelmingly, patients indicated that they understood their discharge instructions and the plan of care at the time they left the hospital, but then when there were breakdowns or unanticipated challenges in the plan, they were uncertain what to do.”
In the HM20 Virtual session, Dr. Greysen and Dr. Bann will present additional data from the same network that Dr. Greysen used in his study, the Hospital Medicine Reengineering Network or HOMERuN, but expanded to include 22 sites.
The specific areas will include clinical and nonclinical criteria for patients to be discharged home, how criteria differed for discharge destinations other than home, discharge logistics, discharge instructions for patients and caregivers, and postdischarge follow-up.
“Developing a discharge protocol during a pandemic is a major challenge. There are new barriers and challenges to finding the right discharge location, as information about illness course and outcome is incomplete or evolving,” Dr. Bann said. “The safety of patients and their loved ones, health care workers and staff, as well as the public at large is always top of mind. Decisions have to be made in a timely way and communicated clearly. This is a huge task in addition to all of the other competing work in the midst of a pandemic, which is why learning from each other and collectively creating our shared best practices is tremendously helpful. If I can take example approaches from other hospitals and update them for use at my site, this saves a lot of time and effort.”
“There is great urgency to understand when it is safe to discharge these patients from the hospital,” Dr. Greysen said. “Many COVID patients can have worsening of their symptoms after a period of initial improvement so sending them home too soon is a major concern. On the other hand, we can’t keep COVID patients in the hospital until they have fully recovered; we would increase their risk of iatrogenic events and we could risk using up capacity of the health care system to care for other patients, both COVID and non-COVID.”
Unfortunately, no evidence base yet exists to guide the creation of discharge guidelines for COVID patients, said Dr. Greysen. “Therefore, we conducted a survey of HOMERuN sites to synthesize practices across sites and provide some guidance for hospitals based on themes or concordance between these sites.
“One area of clear concordance among sites in our study was around the use of [Centers for Disease Control and Prevention] guidelines to address patient isolation procedures as well as strategies to mitigate transmission, such as providing patients with protective gear like masks or requiring the driver who picks up the patient wear a mask for transportation,” Dr. Greysen said. “We also found that many sites used certain clinical criteria – for example, temperature, oxygen saturation or supplementation, and improvement of presenting symptoms – but there was wide variation in the details for these criteria.”
In addition, “some sites required that a patient be afebrile for a certain period of time before discharge whereas others only required that patients be afebrile at the time of discharge. There was also relatively strong consensus around assessing the level of social support and ability to perform activities of daily living prior to discharge,” since social support and ability to function are often interrelated and can be difficult to assess without visiting the home, he said.
“Further development the evidence around which discharge criteria are associated with adverse outcomes such as readmission or death is urgently needed. At this moment, we really don’t know which clinical criteria such as oxygen supplementation or nonclinical criteria are associated with better outcomes in COVID patients,” Dr. Greysen said, but he and his team plan to study this using EMR data in HOMERuN.
Dr. Bann said that clinical criteria for discharge will likely provoke lively discussions during the interactive part of the virtual session. “Also, I have heard a lot of discussion and interest in learning about how different sites are handling postdischarge monitoring and follow-up, such as how we ensure that patients are recovering well after discharge, and whether there are new or different needs for this patient population,” she added.
“Attendees should come away from this session with an understanding of how hospitals across the country have augmented their discharge planning responses during the COVID-19 pandemic,” Dr. Bann said. “This session is all about learning from each other and creating shared best practices,” she said.
“I hope that those who attend our session are able to see some areas of consensus in our study that could be applied to their discharge criteria,” Dr. Greysen added.
Dr. Bann and Dr. Greysen had no relevant financial conflicts to disclose.
Discharge Planning for COVID-19: Collected Practices from Across the U.S.
The COVID-19 pandemic continues to disrupt all aspects of hospital care, and has altered nearly all fundamental practices, including discharge protocols. A session presented at the Society of Hospitalist Medicine’s 2020 Virtual Annual Conference will focus on discharge issues in the COVID-19 era.
“Discharge planning is an integral part of a hospitalist’s clinical care. On a daily basis, we think carefully about how to help our patients safely transition back into life outside of the hospital,” said Maralyssa Bann, MD, of the University of Washington, Seattle, a copresenter at the session.
“Patients need up-to-date information about how to keep themselves and those around them safe,” she said. “They need resources and supports to help them recover from illness.”
These supports include access to appropriate follow-up with primary care doctors or other specialists and being discharged to the right location, such as home or a skilled nursing facility, Dr. Bann noted.
In response to COVID-19, “within an exceptionally short time frame, hospitals have had to rapidly adapt their discharge planning protocols and have had to continue to adapt as new information comes out,” Dr. Bann said.
“In many ways, the COVID-19 pandemic has highlighted the importance of discharge planning for patient care and has added a new element of public health in that we have to take all possible precautions to ensure that patients are not spreading the virus after they leave the hospital,” said Ryan Greysen, MD, of the University of Pennsylvania, Philadelphia, and Dr. Bann’s copresenter.
Many elements go into creating a good discharge plan, but there are often many unknowns, Dr. Greysen said. “I think there is an opportunity to improve the process by improving follow-up as well.”
“For example, one program at our hospital focused on vulnerable older adults includes an in-home visit by a visiting nurse on the day of discharge to verify the patient has everything they need when they arrive home,” However, now with more telemedicine and social distancing, there should be creative approaches to tying up loose ends and monitoring for things that can go wrong in order to give additional guidance, he said.
“In a previous study of 12 U.S. academic medical centers, my colleagues and I interviewed over 1,000 patients who were discharged and then readmitted to ask them what they thought went wrong,” said Dr. Greysen. “Overwhelmingly, patients indicated that they understood their discharge instructions and the plan of care at the time they left the hospital, but then when there were breakdowns or unanticipated challenges in the plan, they were uncertain what to do.”
In the HM20 Virtual session, Dr. Greysen and Dr. Bann will present additional data from the same network that Dr. Greysen used in his study, the Hospital Medicine Reengineering Network or HOMERuN, but expanded to include 22 sites.
The specific areas will include clinical and nonclinical criteria for patients to be discharged home, how criteria differed for discharge destinations other than home, discharge logistics, discharge instructions for patients and caregivers, and postdischarge follow-up.
“Developing a discharge protocol during a pandemic is a major challenge. There are new barriers and challenges to finding the right discharge location, as information about illness course and outcome is incomplete or evolving,” Dr. Bann said. “The safety of patients and their loved ones, health care workers and staff, as well as the public at large is always top of mind. Decisions have to be made in a timely way and communicated clearly. This is a huge task in addition to all of the other competing work in the midst of a pandemic, which is why learning from each other and collectively creating our shared best practices is tremendously helpful. If I can take example approaches from other hospitals and update them for use at my site, this saves a lot of time and effort.”
“There is great urgency to understand when it is safe to discharge these patients from the hospital,” Dr. Greysen said. “Many COVID patients can have worsening of their symptoms after a period of initial improvement so sending them home too soon is a major concern. On the other hand, we can’t keep COVID patients in the hospital until they have fully recovered; we would increase their risk of iatrogenic events and we could risk using up capacity of the health care system to care for other patients, both COVID and non-COVID.”
Unfortunately, no evidence base yet exists to guide the creation of discharge guidelines for COVID patients, said Dr. Greysen. “Therefore, we conducted a survey of HOMERuN sites to synthesize practices across sites and provide some guidance for hospitals based on themes or concordance between these sites.
“One area of clear concordance among sites in our study was around the use of [Centers for Disease Control and Prevention] guidelines to address patient isolation procedures as well as strategies to mitigate transmission, such as providing patients with protective gear like masks or requiring the driver who picks up the patient wear a mask for transportation,” Dr. Greysen said. “We also found that many sites used certain clinical criteria – for example, temperature, oxygen saturation or supplementation, and improvement of presenting symptoms – but there was wide variation in the details for these criteria.”
In addition, “some sites required that a patient be afebrile for a certain period of time before discharge whereas others only required that patients be afebrile at the time of discharge. There was also relatively strong consensus around assessing the level of social support and ability to perform activities of daily living prior to discharge,” since social support and ability to function are often interrelated and can be difficult to assess without visiting the home, he said.
“Further development the evidence around which discharge criteria are associated with adverse outcomes such as readmission or death is urgently needed. At this moment, we really don’t know which clinical criteria such as oxygen supplementation or nonclinical criteria are associated with better outcomes in COVID patients,” Dr. Greysen said, but he and his team plan to study this using EMR data in HOMERuN.
Dr. Bann said that clinical criteria for discharge will likely provoke lively discussions during the interactive part of the virtual session. “Also, I have heard a lot of discussion and interest in learning about how different sites are handling postdischarge monitoring and follow-up, such as how we ensure that patients are recovering well after discharge, and whether there are new or different needs for this patient population,” she added.
“Attendees should come away from this session with an understanding of how hospitals across the country have augmented their discharge planning responses during the COVID-19 pandemic,” Dr. Bann said. “This session is all about learning from each other and creating shared best practices,” she said.
“I hope that those who attend our session are able to see some areas of consensus in our study that could be applied to their discharge criteria,” Dr. Greysen added.
Dr. Bann and Dr. Greysen had no relevant financial conflicts to disclose.
Discharge Planning for COVID-19: Collected Practices from Across the U.S.
The COVID-19 pandemic continues to disrupt all aspects of hospital care, and has altered nearly all fundamental practices, including discharge protocols. A session presented at the Society of Hospitalist Medicine’s 2020 Virtual Annual Conference will focus on discharge issues in the COVID-19 era.
“Discharge planning is an integral part of a hospitalist’s clinical care. On a daily basis, we think carefully about how to help our patients safely transition back into life outside of the hospital,” said Maralyssa Bann, MD, of the University of Washington, Seattle, a copresenter at the session.
“Patients need up-to-date information about how to keep themselves and those around them safe,” she said. “They need resources and supports to help them recover from illness.”
These supports include access to appropriate follow-up with primary care doctors or other specialists and being discharged to the right location, such as home or a skilled nursing facility, Dr. Bann noted.
In response to COVID-19, “within an exceptionally short time frame, hospitals have had to rapidly adapt their discharge planning protocols and have had to continue to adapt as new information comes out,” Dr. Bann said.
“In many ways, the COVID-19 pandemic has highlighted the importance of discharge planning for patient care and has added a new element of public health in that we have to take all possible precautions to ensure that patients are not spreading the virus after they leave the hospital,” said Ryan Greysen, MD, of the University of Pennsylvania, Philadelphia, and Dr. Bann’s copresenter.
Many elements go into creating a good discharge plan, but there are often many unknowns, Dr. Greysen said. “I think there is an opportunity to improve the process by improving follow-up as well.”
“For example, one program at our hospital focused on vulnerable older adults includes an in-home visit by a visiting nurse on the day of discharge to verify the patient has everything they need when they arrive home,” However, now with more telemedicine and social distancing, there should be creative approaches to tying up loose ends and monitoring for things that can go wrong in order to give additional guidance, he said.
“In a previous study of 12 U.S. academic medical centers, my colleagues and I interviewed over 1,000 patients who were discharged and then readmitted to ask them what they thought went wrong,” said Dr. Greysen. “Overwhelmingly, patients indicated that they understood their discharge instructions and the plan of care at the time they left the hospital, but then when there were breakdowns or unanticipated challenges in the plan, they were uncertain what to do.”
In the HM20 Virtual session, Dr. Greysen and Dr. Bann will present additional data from the same network that Dr. Greysen used in his study, the Hospital Medicine Reengineering Network or HOMERuN, but expanded to include 22 sites.
The specific areas will include clinical and nonclinical criteria for patients to be discharged home, how criteria differed for discharge destinations other than home, discharge logistics, discharge instructions for patients and caregivers, and postdischarge follow-up.
“Developing a discharge protocol during a pandemic is a major challenge. There are new barriers and challenges to finding the right discharge location, as information about illness course and outcome is incomplete or evolving,” Dr. Bann said. “The safety of patients and their loved ones, health care workers and staff, as well as the public at large is always top of mind. Decisions have to be made in a timely way and communicated clearly. This is a huge task in addition to all of the other competing work in the midst of a pandemic, which is why learning from each other and collectively creating our shared best practices is tremendously helpful. If I can take example approaches from other hospitals and update them for use at my site, this saves a lot of time and effort.”
“There is great urgency to understand when it is safe to discharge these patients from the hospital,” Dr. Greysen said. “Many COVID patients can have worsening of their symptoms after a period of initial improvement so sending them home too soon is a major concern. On the other hand, we can’t keep COVID patients in the hospital until they have fully recovered; we would increase their risk of iatrogenic events and we could risk using up capacity of the health care system to care for other patients, both COVID and non-COVID.”
Unfortunately, no evidence base yet exists to guide the creation of discharge guidelines for COVID patients, said Dr. Greysen. “Therefore, we conducted a survey of HOMERuN sites to synthesize practices across sites and provide some guidance for hospitals based on themes or concordance between these sites.
“One area of clear concordance among sites in our study was around the use of [Centers for Disease Control and Prevention] guidelines to address patient isolation procedures as well as strategies to mitigate transmission, such as providing patients with protective gear like masks or requiring the driver who picks up the patient wear a mask for transportation,” Dr. Greysen said. “We also found that many sites used certain clinical criteria – for example, temperature, oxygen saturation or supplementation, and improvement of presenting symptoms – but there was wide variation in the details for these criteria.”
In addition, “some sites required that a patient be afebrile for a certain period of time before discharge whereas others only required that patients be afebrile at the time of discharge. There was also relatively strong consensus around assessing the level of social support and ability to perform activities of daily living prior to discharge,” since social support and ability to function are often interrelated and can be difficult to assess without visiting the home, he said.
“Further development the evidence around which discharge criteria are associated with adverse outcomes such as readmission or death is urgently needed. At this moment, we really don’t know which clinical criteria such as oxygen supplementation or nonclinical criteria are associated with better outcomes in COVID patients,” Dr. Greysen said, but he and his team plan to study this using EMR data in HOMERuN.
Dr. Bann said that clinical criteria for discharge will likely provoke lively discussions during the interactive part of the virtual session. “Also, I have heard a lot of discussion and interest in learning about how different sites are handling postdischarge monitoring and follow-up, such as how we ensure that patients are recovering well after discharge, and whether there are new or different needs for this patient population,” she added.
“Attendees should come away from this session with an understanding of how hospitals across the country have augmented their discharge planning responses during the COVID-19 pandemic,” Dr. Bann said. “This session is all about learning from each other and creating shared best practices,” she said.
“I hope that those who attend our session are able to see some areas of consensus in our study that could be applied to their discharge criteria,” Dr. Greysen added.
Dr. Bann and Dr. Greysen had no relevant financial conflicts to disclose.
Discharge Planning for COVID-19: Collected Practices from Across the U.S.
Telemedicine meets menopause in customized patient care service
Women facing issues related to perimenopause and menopause can consult their primary care physicians or ob.gyns. through telemedicine visits, but
The Cusp doesn’t claim to replace routine gynecologic care. Rather, it focuses on perimenopause and menopause symptoms specifically, and states that its physicians, some of whom are certified by the North American Menopause Society, provide expertise in menopause beyond what patients might receive as part of a typical ob.gyn. visit.
The Cusp is a for-profit organization, a group of physicians, nurse practitioners, and technologists who focus on integrated care for women in perimenopause and beyond. The aim is to leverage technology as a way to connect women to the care platform to book physician and nurse practitioner visits virtually and to have all of the information about their care centralized in one place.
According to the website, most patients who sign up for a care plan check in with their providers at least once a month to monitor their symptoms and tweak treatment strategies. Patients who sign up are prompted to download an app, which then becomes the main tool for scheduling future visits, tracking symptoms, and communicating with providers.
The Cusp launched in early 2019, before the advent of the COVID-19 pandemic, but the pandemic has accelerated the acceptance across medical specialties, suggesting that telemedicine is here to stay, according to Mindy Goldman, MD, professor of gynecology and gynecologic surgery at the University of California, San Francisco, and director of the Gynecology Center for Cancer Survivors and At-Risk Women at UCSF, who also serves as a medical adviser to the Cusp.
Partnering with technology companies allows opportunities to provide care in areas where there are gaps, such as menopause management, she said. Many clinicians in primary care and ob.gyn. care don’t have the time or training to discuss menopause management in depth with patients, and patient interviews conducted by the Cusp before launching the site showed that this was an area of need.
“One thing that is really unique about the Cusp is that we brought together experts to provide care in both in an evidence-based and holistic fashion,” Dr. Goldman emphasized.
The Cusp’s medical team includes physician and nurse practitioner menopause experts with backgrounds including not only ob.gyn. but also psychiatry, integrative medicine, and naturopathic medicine, with plans to add endocrinology and dermatology as well. This holistic approach allows the Cusp to tailor care based on what each woman is looking for, with evidence-based expertise to support treatment decisions, said Dr. Goldman, whose advisory role includes helping to develop patient treatment protocols and services.
If a woman wants to begin treating symptoms with a naturopathic approach, the team will provide protocols that take current guidelines into account. Regular visits, approximately once a month or as needed, allow for collaboration with the Cusp’s specialists to provide consistent care that is very comprehensive, she said.
One of the benefits of the Cusp is the opportunity for “frequent touchpoints” in which providers reach out to patients via text, email, or video. Although a traditional medical visit may include some initial discussion of menopause and treatment plans, the Cusp offers “a more seamless way to address needs on an ongoing basis,” to provide more complete patient care, Dr. Goldman said.
“We are constantly asking women what they are looking for in menopause care,” and a recurring question was about hormone testing, she said. Nontraditional practitioners may offer hormone testing as a way of individualizing care that also involves compounded formulations, and other treatments that are not standard of care. “In all of our protocols we follow what is recommended by standard organizations such as ACOG [American College of Obstetricians and Gynecologists] and NAMS.”
The Cusp’s newest service is an at-home hormone test currently for women in New York and California, but the company plans to expand this service. The hormone test, while not essential, is another tool to guide menopause management, and having a sense of when menopause will occur “gives us a chance to talk to people about behavioral changes and time to personalize a treatment protocol,” Dr. Goldman said.
The test is based in part on the anti-Müllerian hormone, which recent studies have shown is useful in predicting time to menopause. This, in combination with other hormone tests and other clinical information, will allow the Cusp’s menopause specialists to help women in perimenopause gain perspective on their symptoms and design a treatment plan that can evolve as their needs change, she explained.
“The more information you know about when menopause is going to be happening, you can tailor your treatment plan,” Dr. Goldman said. For example, a woman who may be 2 years away from menopause might consider a naturopathic approach at first, and switch to a different therapy as menopause occurs. “We know that the risks of cardiovascular disease and bone loss increase after menopause, and knowing the time to menopause gives us more guidance when educating patients about healthy lifestyle habits such as exercise and dietary changes that can help reduce these risks.”
The Cusp allows patients to use money in flexible spending accounts or health savings accounts to pay for the program. If doctors require lab tests or other procedures, these are covered through the patients’ regular health insurance as they would be if requested by a primary care physician or other health care professional.
Lubna Pal, MBBS, director of the menopause program at Yale University, New Haven, Conn., commented that part of the value in a telehealth site such as the Cusp is to serve as “a resource for reproductively aging women to understand what is happening to them.”
Any way to improve education on the topic of menopause is empowering to women, said Dr. Pal, professor of obstetrics, gynecology, and reproductive sciences at Yale. “This is an opportunity for patients to have access to a directed evaluation” of menopause-related symptoms. Then, when women visit their regular health care provider in person, they are well-equipped with knowledge to ask more informed questions and discuss a wide range of treatment options.
Dr. Pal noted that the hormone test is less valuable than the interaction between physicians and patients, whether online or in person.
“Menopause is a Monday morning quarterback diagnosis,” she said, emphasizing that, not only is a year without menses part of the diagnosis of menopause, many women in perimenopause can have wide fluctuations in hormone levels, so a test is more of a snapshot than a diagnostic tool, and that the results might cause unnecessary angst and concerns for patients.
However, part of the value of a telehealth site that focuses on menopause is that it gives women a place to learn more about their biology and to clarify their questions about symptoms and become aware of a range of treatment options. Telehealth consultations also can help women recognize how other factors such as lifestyle modifications can play a role in menopause symptoms, and how modifying these factors may provide some relief, she said.
Dr. Pal said she would be cautious about the idea of prescribing without seeing the patient in person, but noted that telehealth sites such as the Cusp can be a win-win to enhance women’s health when used in combination with regular in-person visits to an ob.gyn. The added value in patients’ being able to discuss their concerns and to learn more about their symptoms means that they will be better informed to develop a menopause management strategy in partnership with their providers, said Dr. Pal, who is not associated with the Cusp.
Dr. Goldman disclosed receiving compensation from the Cusp for her advisory work. She also holds stock options in the company. Dr. Pal, who is a member of the Ob.Gyn. News editorial advisory board, had no financial conflicts to disclose.
Women facing issues related to perimenopause and menopause can consult their primary care physicians or ob.gyns. through telemedicine visits, but
The Cusp doesn’t claim to replace routine gynecologic care. Rather, it focuses on perimenopause and menopause symptoms specifically, and states that its physicians, some of whom are certified by the North American Menopause Society, provide expertise in menopause beyond what patients might receive as part of a typical ob.gyn. visit.
The Cusp is a for-profit organization, a group of physicians, nurse practitioners, and technologists who focus on integrated care for women in perimenopause and beyond. The aim is to leverage technology as a way to connect women to the care platform to book physician and nurse practitioner visits virtually and to have all of the information about their care centralized in one place.
According to the website, most patients who sign up for a care plan check in with their providers at least once a month to monitor their symptoms and tweak treatment strategies. Patients who sign up are prompted to download an app, which then becomes the main tool for scheduling future visits, tracking symptoms, and communicating with providers.
The Cusp launched in early 2019, before the advent of the COVID-19 pandemic, but the pandemic has accelerated the acceptance across medical specialties, suggesting that telemedicine is here to stay, according to Mindy Goldman, MD, professor of gynecology and gynecologic surgery at the University of California, San Francisco, and director of the Gynecology Center for Cancer Survivors and At-Risk Women at UCSF, who also serves as a medical adviser to the Cusp.
Partnering with technology companies allows opportunities to provide care in areas where there are gaps, such as menopause management, she said. Many clinicians in primary care and ob.gyn. care don’t have the time or training to discuss menopause management in depth with patients, and patient interviews conducted by the Cusp before launching the site showed that this was an area of need.
“One thing that is really unique about the Cusp is that we brought together experts to provide care in both in an evidence-based and holistic fashion,” Dr. Goldman emphasized.
The Cusp’s medical team includes physician and nurse practitioner menopause experts with backgrounds including not only ob.gyn. but also psychiatry, integrative medicine, and naturopathic medicine, with plans to add endocrinology and dermatology as well. This holistic approach allows the Cusp to tailor care based on what each woman is looking for, with evidence-based expertise to support treatment decisions, said Dr. Goldman, whose advisory role includes helping to develop patient treatment protocols and services.
If a woman wants to begin treating symptoms with a naturopathic approach, the team will provide protocols that take current guidelines into account. Regular visits, approximately once a month or as needed, allow for collaboration with the Cusp’s specialists to provide consistent care that is very comprehensive, she said.
One of the benefits of the Cusp is the opportunity for “frequent touchpoints” in which providers reach out to patients via text, email, or video. Although a traditional medical visit may include some initial discussion of menopause and treatment plans, the Cusp offers “a more seamless way to address needs on an ongoing basis,” to provide more complete patient care, Dr. Goldman said.
“We are constantly asking women what they are looking for in menopause care,” and a recurring question was about hormone testing, she said. Nontraditional practitioners may offer hormone testing as a way of individualizing care that also involves compounded formulations, and other treatments that are not standard of care. “In all of our protocols we follow what is recommended by standard organizations such as ACOG [American College of Obstetricians and Gynecologists] and NAMS.”
The Cusp’s newest service is an at-home hormone test currently for women in New York and California, but the company plans to expand this service. The hormone test, while not essential, is another tool to guide menopause management, and having a sense of when menopause will occur “gives us a chance to talk to people about behavioral changes and time to personalize a treatment protocol,” Dr. Goldman said.
The test is based in part on the anti-Müllerian hormone, which recent studies have shown is useful in predicting time to menopause. This, in combination with other hormone tests and other clinical information, will allow the Cusp’s menopause specialists to help women in perimenopause gain perspective on their symptoms and design a treatment plan that can evolve as their needs change, she explained.
“The more information you know about when menopause is going to be happening, you can tailor your treatment plan,” Dr. Goldman said. For example, a woman who may be 2 years away from menopause might consider a naturopathic approach at first, and switch to a different therapy as menopause occurs. “We know that the risks of cardiovascular disease and bone loss increase after menopause, and knowing the time to menopause gives us more guidance when educating patients about healthy lifestyle habits such as exercise and dietary changes that can help reduce these risks.”
The Cusp allows patients to use money in flexible spending accounts or health savings accounts to pay for the program. If doctors require lab tests or other procedures, these are covered through the patients’ regular health insurance as they would be if requested by a primary care physician or other health care professional.
Lubna Pal, MBBS, director of the menopause program at Yale University, New Haven, Conn., commented that part of the value in a telehealth site such as the Cusp is to serve as “a resource for reproductively aging women to understand what is happening to them.”
Any way to improve education on the topic of menopause is empowering to women, said Dr. Pal, professor of obstetrics, gynecology, and reproductive sciences at Yale. “This is an opportunity for patients to have access to a directed evaluation” of menopause-related symptoms. Then, when women visit their regular health care provider in person, they are well-equipped with knowledge to ask more informed questions and discuss a wide range of treatment options.
Dr. Pal noted that the hormone test is less valuable than the interaction between physicians and patients, whether online or in person.
“Menopause is a Monday morning quarterback diagnosis,” she said, emphasizing that, not only is a year without menses part of the diagnosis of menopause, many women in perimenopause can have wide fluctuations in hormone levels, so a test is more of a snapshot than a diagnostic tool, and that the results might cause unnecessary angst and concerns for patients.
However, part of the value of a telehealth site that focuses on menopause is that it gives women a place to learn more about their biology and to clarify their questions about symptoms and become aware of a range of treatment options. Telehealth consultations also can help women recognize how other factors such as lifestyle modifications can play a role in menopause symptoms, and how modifying these factors may provide some relief, she said.
Dr. Pal said she would be cautious about the idea of prescribing without seeing the patient in person, but noted that telehealth sites such as the Cusp can be a win-win to enhance women’s health when used in combination with regular in-person visits to an ob.gyn. The added value in patients’ being able to discuss their concerns and to learn more about their symptoms means that they will be better informed to develop a menopause management strategy in partnership with their providers, said Dr. Pal, who is not associated with the Cusp.
Dr. Goldman disclosed receiving compensation from the Cusp for her advisory work. She also holds stock options in the company. Dr. Pal, who is a member of the Ob.Gyn. News editorial advisory board, had no financial conflicts to disclose.
Women facing issues related to perimenopause and menopause can consult their primary care physicians or ob.gyns. through telemedicine visits, but
The Cusp doesn’t claim to replace routine gynecologic care. Rather, it focuses on perimenopause and menopause symptoms specifically, and states that its physicians, some of whom are certified by the North American Menopause Society, provide expertise in menopause beyond what patients might receive as part of a typical ob.gyn. visit.
The Cusp is a for-profit organization, a group of physicians, nurse practitioners, and technologists who focus on integrated care for women in perimenopause and beyond. The aim is to leverage technology as a way to connect women to the care platform to book physician and nurse practitioner visits virtually and to have all of the information about their care centralized in one place.
According to the website, most patients who sign up for a care plan check in with their providers at least once a month to monitor their symptoms and tweak treatment strategies. Patients who sign up are prompted to download an app, which then becomes the main tool for scheduling future visits, tracking symptoms, and communicating with providers.
The Cusp launched in early 2019, before the advent of the COVID-19 pandemic, but the pandemic has accelerated the acceptance across medical specialties, suggesting that telemedicine is here to stay, according to Mindy Goldman, MD, professor of gynecology and gynecologic surgery at the University of California, San Francisco, and director of the Gynecology Center for Cancer Survivors and At-Risk Women at UCSF, who also serves as a medical adviser to the Cusp.
Partnering with technology companies allows opportunities to provide care in areas where there are gaps, such as menopause management, she said. Many clinicians in primary care and ob.gyn. care don’t have the time or training to discuss menopause management in depth with patients, and patient interviews conducted by the Cusp before launching the site showed that this was an area of need.
“One thing that is really unique about the Cusp is that we brought together experts to provide care in both in an evidence-based and holistic fashion,” Dr. Goldman emphasized.
The Cusp’s medical team includes physician and nurse practitioner menopause experts with backgrounds including not only ob.gyn. but also psychiatry, integrative medicine, and naturopathic medicine, with plans to add endocrinology and dermatology as well. This holistic approach allows the Cusp to tailor care based on what each woman is looking for, with evidence-based expertise to support treatment decisions, said Dr. Goldman, whose advisory role includes helping to develop patient treatment protocols and services.
If a woman wants to begin treating symptoms with a naturopathic approach, the team will provide protocols that take current guidelines into account. Regular visits, approximately once a month or as needed, allow for collaboration with the Cusp’s specialists to provide consistent care that is very comprehensive, she said.
One of the benefits of the Cusp is the opportunity for “frequent touchpoints” in which providers reach out to patients via text, email, or video. Although a traditional medical visit may include some initial discussion of menopause and treatment plans, the Cusp offers “a more seamless way to address needs on an ongoing basis,” to provide more complete patient care, Dr. Goldman said.
“We are constantly asking women what they are looking for in menopause care,” and a recurring question was about hormone testing, she said. Nontraditional practitioners may offer hormone testing as a way of individualizing care that also involves compounded formulations, and other treatments that are not standard of care. “In all of our protocols we follow what is recommended by standard organizations such as ACOG [American College of Obstetricians and Gynecologists] and NAMS.”
The Cusp’s newest service is an at-home hormone test currently for women in New York and California, but the company plans to expand this service. The hormone test, while not essential, is another tool to guide menopause management, and having a sense of when menopause will occur “gives us a chance to talk to people about behavioral changes and time to personalize a treatment protocol,” Dr. Goldman said.
The test is based in part on the anti-Müllerian hormone, which recent studies have shown is useful in predicting time to menopause. This, in combination with other hormone tests and other clinical information, will allow the Cusp’s menopause specialists to help women in perimenopause gain perspective on their symptoms and design a treatment plan that can evolve as their needs change, she explained.
“The more information you know about when menopause is going to be happening, you can tailor your treatment plan,” Dr. Goldman said. For example, a woman who may be 2 years away from menopause might consider a naturopathic approach at first, and switch to a different therapy as menopause occurs. “We know that the risks of cardiovascular disease and bone loss increase after menopause, and knowing the time to menopause gives us more guidance when educating patients about healthy lifestyle habits such as exercise and dietary changes that can help reduce these risks.”
The Cusp allows patients to use money in flexible spending accounts or health savings accounts to pay for the program. If doctors require lab tests or other procedures, these are covered through the patients’ regular health insurance as they would be if requested by a primary care physician or other health care professional.
Lubna Pal, MBBS, director of the menopause program at Yale University, New Haven, Conn., commented that part of the value in a telehealth site such as the Cusp is to serve as “a resource for reproductively aging women to understand what is happening to them.”
Any way to improve education on the topic of menopause is empowering to women, said Dr. Pal, professor of obstetrics, gynecology, and reproductive sciences at Yale. “This is an opportunity for patients to have access to a directed evaluation” of menopause-related symptoms. Then, when women visit their regular health care provider in person, they are well-equipped with knowledge to ask more informed questions and discuss a wide range of treatment options.
Dr. Pal noted that the hormone test is less valuable than the interaction between physicians and patients, whether online or in person.
“Menopause is a Monday morning quarterback diagnosis,” she said, emphasizing that, not only is a year without menses part of the diagnosis of menopause, many women in perimenopause can have wide fluctuations in hormone levels, so a test is more of a snapshot than a diagnostic tool, and that the results might cause unnecessary angst and concerns for patients.
However, part of the value of a telehealth site that focuses on menopause is that it gives women a place to learn more about their biology and to clarify their questions about symptoms and become aware of a range of treatment options. Telehealth consultations also can help women recognize how other factors such as lifestyle modifications can play a role in menopause symptoms, and how modifying these factors may provide some relief, she said.
Dr. Pal said she would be cautious about the idea of prescribing without seeing the patient in person, but noted that telehealth sites such as the Cusp can be a win-win to enhance women’s health when used in combination with regular in-person visits to an ob.gyn. The added value in patients’ being able to discuss their concerns and to learn more about their symptoms means that they will be better informed to develop a menopause management strategy in partnership with their providers, said Dr. Pal, who is not associated with the Cusp.
Dr. Goldman disclosed receiving compensation from the Cusp for her advisory work. She also holds stock options in the company. Dr. Pal, who is a member of the Ob.Gyn. News editorial advisory board, had no financial conflicts to disclose.
AHA on cannabis: No evidence of heart benefits, but potential harms
Evidence for a link between cannabis use and cardiovascular health remains unsupported, and the potential risks outweigh any potential benefits, according to a scientific statement from the American Heart Association.
The increased legalization of cannabis and cannabis products in the United States has driven medical professionals to evaluate the safety and efficacy of cannabis in relation to health conditions, wrote Robert L. Page II, PharmD, of the University of Colorado, Aurora, and colleagues.
In a statement published in Circulation, the researchers noted that although cannabis has been shown to relieve pain and other symptoms in certain conditions, clinicians in the United States have been limited from studying its health effects because of federal law restrictions. “Cannabis remains a schedule I controlled substance, deeming no accepted medical use, a high potential for abuse, and an unacceptable safety profile,” the researchers wrote.
The statement addresses issues with the use of cannabis by individuals with cardiovascular disease or those at increased risk. Observational studies have shown no cardiovascular benefits associated with cannabis, the writers noted. The most common chemicals in cannabis include THC (tetrahydrocannabinolic acid) and CBD (cannabidiol).
Some research has shown associations between CBD cardiovascular features including lower blood pressure and reduced inflammation, the writers noted. However, THC, the component of cannabis associated with a “high” or intoxication, has been associated with heart rhythm abnormalities. The writers cited data suggesting an increased risk of heart attacks, atrial fibrillation and heart failure, although more research is needed.
The statement outlines common cannabis formulations including plant-based, extracts, crystalline forms, edible products, and tinctures. In addition, the statement notes that synthetic cannabis products are marketed and used in the United States without subject to regulation.
“Over the past 5 years, we have seen a surge in cannabis use, particularly during the COVID-19 pandemic here in Colorado, especially among adolescents and young adults,” Dr. Page said in an interview. Because of the surge, health care practitioners need to familiarize themselves with not only the benefits, but risks associated with cannabis use regardless of the formulation,” he said. As heart disease remains a leading cause of death in the United States, understanding the cardiovascular risks associated with cannabis is crucial at this time.
Dr. Page noted that popular attitudes about cannabis could pose risks to users’ cardiovascular health. “One leading misconception about cannabis is because it is ‘natural’ it must be safe,” Dr. Page said. “As with all medications, cannabis has side effects, some of which can be cardiovascular in nature,” he said. “Significant drug-drug interactions can occur as CBD and THC, both found in cannabis, inhibit CYP3A4, which metabolizes a large number of medications used to treat many cardiovascular conditions,” he noted.
“Unfortunately, much of the published data is observational in nature due to the federal restrictions on cannabis as a schedule I drug,” said Dr. Page. “Nonetheless, safety signals have emerged regarding cannabis use and adverse cardiovascular outcomes, including myocardial infarction, heart failure, and atrial fibrillation. Carefully designed prospective short- and long-term studies regarding cannabis use and cardiovascular safety are needed,” he emphasized.
Areas in particular need of additional research include the cardiovascular effects of cannabis in several vulnerable populations such as adolescents, older adults, pregnant women, transplant recipients, and those with underlying cardiovascular disease, said Dr. Page.
“Nonetheless, based on the safety signals described within this Clinical Science Statement, an open discussion regarding the risks of using cannabis needs to occur between patient and health care providers,” he said. “Furthermore, patients must be transparent regarding their cannabis use with their cardiologist and primary care provider. The cannabis story will continue to evolve and is a rapidly moving/changing target,” he said.
“Whether cannabis use is a definitive risk factor for cardiovascular disease as with tobacco use is still unknown, and both acute and long-term studies are desperately needed to address this issue,” he said.
Dr. Page had no relevant financial conflicts to disclose.
SOURCE: Page et al. Circulation. 2020 Aug 5. doi: 10.1161/CIR.0000000000000883.
Evidence for a link between cannabis use and cardiovascular health remains unsupported, and the potential risks outweigh any potential benefits, according to a scientific statement from the American Heart Association.
The increased legalization of cannabis and cannabis products in the United States has driven medical professionals to evaluate the safety and efficacy of cannabis in relation to health conditions, wrote Robert L. Page II, PharmD, of the University of Colorado, Aurora, and colleagues.
In a statement published in Circulation, the researchers noted that although cannabis has been shown to relieve pain and other symptoms in certain conditions, clinicians in the United States have been limited from studying its health effects because of federal law restrictions. “Cannabis remains a schedule I controlled substance, deeming no accepted medical use, a high potential for abuse, and an unacceptable safety profile,” the researchers wrote.
The statement addresses issues with the use of cannabis by individuals with cardiovascular disease or those at increased risk. Observational studies have shown no cardiovascular benefits associated with cannabis, the writers noted. The most common chemicals in cannabis include THC (tetrahydrocannabinolic acid) and CBD (cannabidiol).
Some research has shown associations between CBD cardiovascular features including lower blood pressure and reduced inflammation, the writers noted. However, THC, the component of cannabis associated with a “high” or intoxication, has been associated with heart rhythm abnormalities. The writers cited data suggesting an increased risk of heart attacks, atrial fibrillation and heart failure, although more research is needed.
The statement outlines common cannabis formulations including plant-based, extracts, crystalline forms, edible products, and tinctures. In addition, the statement notes that synthetic cannabis products are marketed and used in the United States without subject to regulation.
“Over the past 5 years, we have seen a surge in cannabis use, particularly during the COVID-19 pandemic here in Colorado, especially among adolescents and young adults,” Dr. Page said in an interview. Because of the surge, health care practitioners need to familiarize themselves with not only the benefits, but risks associated with cannabis use regardless of the formulation,” he said. As heart disease remains a leading cause of death in the United States, understanding the cardiovascular risks associated with cannabis is crucial at this time.
Dr. Page noted that popular attitudes about cannabis could pose risks to users’ cardiovascular health. “One leading misconception about cannabis is because it is ‘natural’ it must be safe,” Dr. Page said. “As with all medications, cannabis has side effects, some of which can be cardiovascular in nature,” he said. “Significant drug-drug interactions can occur as CBD and THC, both found in cannabis, inhibit CYP3A4, which metabolizes a large number of medications used to treat many cardiovascular conditions,” he noted.
“Unfortunately, much of the published data is observational in nature due to the federal restrictions on cannabis as a schedule I drug,” said Dr. Page. “Nonetheless, safety signals have emerged regarding cannabis use and adverse cardiovascular outcomes, including myocardial infarction, heart failure, and atrial fibrillation. Carefully designed prospective short- and long-term studies regarding cannabis use and cardiovascular safety are needed,” he emphasized.
Areas in particular need of additional research include the cardiovascular effects of cannabis in several vulnerable populations such as adolescents, older adults, pregnant women, transplant recipients, and those with underlying cardiovascular disease, said Dr. Page.
“Nonetheless, based on the safety signals described within this Clinical Science Statement, an open discussion regarding the risks of using cannabis needs to occur between patient and health care providers,” he said. “Furthermore, patients must be transparent regarding their cannabis use with their cardiologist and primary care provider. The cannabis story will continue to evolve and is a rapidly moving/changing target,” he said.
“Whether cannabis use is a definitive risk factor for cardiovascular disease as with tobacco use is still unknown, and both acute and long-term studies are desperately needed to address this issue,” he said.
Dr. Page had no relevant financial conflicts to disclose.
SOURCE: Page et al. Circulation. 2020 Aug 5. doi: 10.1161/CIR.0000000000000883.
Evidence for a link between cannabis use and cardiovascular health remains unsupported, and the potential risks outweigh any potential benefits, according to a scientific statement from the American Heart Association.
The increased legalization of cannabis and cannabis products in the United States has driven medical professionals to evaluate the safety and efficacy of cannabis in relation to health conditions, wrote Robert L. Page II, PharmD, of the University of Colorado, Aurora, and colleagues.
In a statement published in Circulation, the researchers noted that although cannabis has been shown to relieve pain and other symptoms in certain conditions, clinicians in the United States have been limited from studying its health effects because of federal law restrictions. “Cannabis remains a schedule I controlled substance, deeming no accepted medical use, a high potential for abuse, and an unacceptable safety profile,” the researchers wrote.
The statement addresses issues with the use of cannabis by individuals with cardiovascular disease or those at increased risk. Observational studies have shown no cardiovascular benefits associated with cannabis, the writers noted. The most common chemicals in cannabis include THC (tetrahydrocannabinolic acid) and CBD (cannabidiol).
Some research has shown associations between CBD cardiovascular features including lower blood pressure and reduced inflammation, the writers noted. However, THC, the component of cannabis associated with a “high” or intoxication, has been associated with heart rhythm abnormalities. The writers cited data suggesting an increased risk of heart attacks, atrial fibrillation and heart failure, although more research is needed.
The statement outlines common cannabis formulations including plant-based, extracts, crystalline forms, edible products, and tinctures. In addition, the statement notes that synthetic cannabis products are marketed and used in the United States without subject to regulation.
“Over the past 5 years, we have seen a surge in cannabis use, particularly during the COVID-19 pandemic here in Colorado, especially among adolescents and young adults,” Dr. Page said in an interview. Because of the surge, health care practitioners need to familiarize themselves with not only the benefits, but risks associated with cannabis use regardless of the formulation,” he said. As heart disease remains a leading cause of death in the United States, understanding the cardiovascular risks associated with cannabis is crucial at this time.
Dr. Page noted that popular attitudes about cannabis could pose risks to users’ cardiovascular health. “One leading misconception about cannabis is because it is ‘natural’ it must be safe,” Dr. Page said. “As with all medications, cannabis has side effects, some of which can be cardiovascular in nature,” he said. “Significant drug-drug interactions can occur as CBD and THC, both found in cannabis, inhibit CYP3A4, which metabolizes a large number of medications used to treat many cardiovascular conditions,” he noted.
“Unfortunately, much of the published data is observational in nature due to the federal restrictions on cannabis as a schedule I drug,” said Dr. Page. “Nonetheless, safety signals have emerged regarding cannabis use and adverse cardiovascular outcomes, including myocardial infarction, heart failure, and atrial fibrillation. Carefully designed prospective short- and long-term studies regarding cannabis use and cardiovascular safety are needed,” he emphasized.
Areas in particular need of additional research include the cardiovascular effects of cannabis in several vulnerable populations such as adolescents, older adults, pregnant women, transplant recipients, and those with underlying cardiovascular disease, said Dr. Page.
“Nonetheless, based on the safety signals described within this Clinical Science Statement, an open discussion regarding the risks of using cannabis needs to occur between patient and health care providers,” he said. “Furthermore, patients must be transparent regarding their cannabis use with their cardiologist and primary care provider. The cannabis story will continue to evolve and is a rapidly moving/changing target,” he said.
“Whether cannabis use is a definitive risk factor for cardiovascular disease as with tobacco use is still unknown, and both acute and long-term studies are desperately needed to address this issue,” he said.
Dr. Page had no relevant financial conflicts to disclose.
SOURCE: Page et al. Circulation. 2020 Aug 5. doi: 10.1161/CIR.0000000000000883.
FROM CIRCULATION
New psoriasis guidelines focus on topical and alternative treatments, and severity measures
and the National Psoriasis Foundation.
The guidelines, published in the Journal of the American Academy of Dermatology, focus on treatment for adults, and follow the release of other AAD-NPF guidelines on biologics for psoriasis, psoriasis-related comorbidities, pediatric psoriasis, and phototherapy in 2019, and earlier this year, guidelines for systemic nonbiologic treatments. The latest guidelines’ section on topical treatment outlines evidence for the efficacy, effectiveness, and adverse events related to topical steroids, topical tacrolimus and pimecrolimus, vitamin D analogues, tazarotene, moisturizers, salicylic acid, anthralin, coal tar, combinations with biologic agents, and combinations with nonbiologic treatments (methotrexate, cyclosporine, acitretin, and apremilast).
The guidelines noted the “key role” of topical corticosteroids in treating psoriasis “especially for localized disease,” and include a review of the data on low-, moderate-, high-, and ultrahigh-potency topical steroids for psoriasis.
In general, all topical steroids can be used in combination with biologics, according to the guidelines, but the strongest recommendations based on the latest evidence include the addition of an ultra-high potency topical corticosteroid to standard dose etanercept for 12 weeks. Currently, 11 biologics are approved by the Food and Drug Administration for the treatment of psoriasis.
In addition, “while not FDA approved for psoriasis, the topical calcineurin inhibitors tacrolimus and pimecrolimus are often employed in the treatment of psoriasis,” can be helpful for “thinner skin such as facial and intertriginous areas,” and can be steroid sparing when used for more than 4 weeks, according to the guidelines.
Don’t discount the role of patient preferences when choosing topical treatments, the authors noted. “The optimal vehicle choice is the one the patient is mostly likely to use.”
The guidelines also address the evidence for effectiveness, and adverse events in the use of several alternative medicines for psoriasis including traditional Chinese medicine, and the herbal therapies aloe vera and St. John’s wort, as well as the potential role of dietary supplements including fish oil, vitamin D, turmeric, and zinc in managing psoriasis, and the potential role of a gluten-free diet.
In general, research on the efficacy, effectiveness, and potential adverse effects of these strategies are limited, according to the guidelines, although many patients express interest in supplements and herbal products. For example, “Many patients ask about the overall role of vitamin D in skin health. Rather than adding oral vitamin D supplementation, topical therapy with vitamin D agents is effective for the treatment of psoriasis,” the authors noted.
In addition, they noted that mind/body strategies, namely hypnosis and stress reduction or meditation techniques, have been shown to improve symptoms and can be helpful for some patients, but clinical evidence is limited.
The guidelines also addressed methods for assessing disease severity in psoriasis. They recommended using body surface area (BSA) to assess psoriasis severity and patient response to treatment in the clinical setting. However, BSA is a provider assessment tool that “does not take into account location on the body, clinical characteristics of the plaques, symptoms, or quality of life issues,” the authors noted. The Psoriasis Area and Severity Index (PASI) measures erythema, induration, and scaling and is more suited to assessing psoriasis severity and response to treatment in clinical trials rather than in practice, they said.
Prior AAD guidelines on psoriasis were published more than 10 years ago, and major developments including the availability of new biologic drugs and new data on comorbidities have been recognized in the past decade, working group cochair and author of the guidelines Alan Menter, MD, said in an interview.
The key game-changers from previous guidelines include the full section published on comorbidities plus the development of two new important cytokine classes: three IL-17 drugs and three new IL-23 drugs now available for moderate to severe psoriasis, said Dr. Menter, chairman of the division of dermatology at Baylor University Medical Center, Dallas.
Barriers to implementing the guidelines in practice may occur when “third party payers make the decision on which of the 11 biologic drugs now approved for moderate to severe psoriasis should be used,” he noted.
As for next steps in psoriasis studies, “new biomarker research is currently underway,” Dr. Menter said. With 11 biologic agents new formally approved by the FDA for moderate to severe psoriasis, the next steps are to determine which drug is likely to be the most appropriate for each individual patient.
Dr. Menter disclosed relationships with multiple companies that develop and manufacture psoriasis therapies, including Abbott Labs, AbbVie, Amgen, Eli Lilly and Company, Galderma USA, Janssen Pharmaceuticals, LEO Pharma US, Menlo Therapeutics, and Novartis. The updated guidelines were designed by a multidisciplinary work group of psoriasis experts including dermatologists, a rheumatologist, a cardiologist, and representatives from a patient advocacy organization.
SOURCE: Elmets CA et al. J Am Acad Dermatol. 2020 Jul 29. doi: 10.1016/j.jaad.2020.07.087.
and the National Psoriasis Foundation.
The guidelines, published in the Journal of the American Academy of Dermatology, focus on treatment for adults, and follow the release of other AAD-NPF guidelines on biologics for psoriasis, psoriasis-related comorbidities, pediatric psoriasis, and phototherapy in 2019, and earlier this year, guidelines for systemic nonbiologic treatments. The latest guidelines’ section on topical treatment outlines evidence for the efficacy, effectiveness, and adverse events related to topical steroids, topical tacrolimus and pimecrolimus, vitamin D analogues, tazarotene, moisturizers, salicylic acid, anthralin, coal tar, combinations with biologic agents, and combinations with nonbiologic treatments (methotrexate, cyclosporine, acitretin, and apremilast).
The guidelines noted the “key role” of topical corticosteroids in treating psoriasis “especially for localized disease,” and include a review of the data on low-, moderate-, high-, and ultrahigh-potency topical steroids for psoriasis.
In general, all topical steroids can be used in combination with biologics, according to the guidelines, but the strongest recommendations based on the latest evidence include the addition of an ultra-high potency topical corticosteroid to standard dose etanercept for 12 weeks. Currently, 11 biologics are approved by the Food and Drug Administration for the treatment of psoriasis.
In addition, “while not FDA approved for psoriasis, the topical calcineurin inhibitors tacrolimus and pimecrolimus are often employed in the treatment of psoriasis,” can be helpful for “thinner skin such as facial and intertriginous areas,” and can be steroid sparing when used for more than 4 weeks, according to the guidelines.
Don’t discount the role of patient preferences when choosing topical treatments, the authors noted. “The optimal vehicle choice is the one the patient is mostly likely to use.”
The guidelines also address the evidence for effectiveness, and adverse events in the use of several alternative medicines for psoriasis including traditional Chinese medicine, and the herbal therapies aloe vera and St. John’s wort, as well as the potential role of dietary supplements including fish oil, vitamin D, turmeric, and zinc in managing psoriasis, and the potential role of a gluten-free diet.
In general, research on the efficacy, effectiveness, and potential adverse effects of these strategies are limited, according to the guidelines, although many patients express interest in supplements and herbal products. For example, “Many patients ask about the overall role of vitamin D in skin health. Rather than adding oral vitamin D supplementation, topical therapy with vitamin D agents is effective for the treatment of psoriasis,” the authors noted.
In addition, they noted that mind/body strategies, namely hypnosis and stress reduction or meditation techniques, have been shown to improve symptoms and can be helpful for some patients, but clinical evidence is limited.
The guidelines also addressed methods for assessing disease severity in psoriasis. They recommended using body surface area (BSA) to assess psoriasis severity and patient response to treatment in the clinical setting. However, BSA is a provider assessment tool that “does not take into account location on the body, clinical characteristics of the plaques, symptoms, or quality of life issues,” the authors noted. The Psoriasis Area and Severity Index (PASI) measures erythema, induration, and scaling and is more suited to assessing psoriasis severity and response to treatment in clinical trials rather than in practice, they said.
Prior AAD guidelines on psoriasis were published more than 10 years ago, and major developments including the availability of new biologic drugs and new data on comorbidities have been recognized in the past decade, working group cochair and author of the guidelines Alan Menter, MD, said in an interview.
The key game-changers from previous guidelines include the full section published on comorbidities plus the development of two new important cytokine classes: three IL-17 drugs and three new IL-23 drugs now available for moderate to severe psoriasis, said Dr. Menter, chairman of the division of dermatology at Baylor University Medical Center, Dallas.
Barriers to implementing the guidelines in practice may occur when “third party payers make the decision on which of the 11 biologic drugs now approved for moderate to severe psoriasis should be used,” he noted.
As for next steps in psoriasis studies, “new biomarker research is currently underway,” Dr. Menter said. With 11 biologic agents new formally approved by the FDA for moderate to severe psoriasis, the next steps are to determine which drug is likely to be the most appropriate for each individual patient.
Dr. Menter disclosed relationships with multiple companies that develop and manufacture psoriasis therapies, including Abbott Labs, AbbVie, Amgen, Eli Lilly and Company, Galderma USA, Janssen Pharmaceuticals, LEO Pharma US, Menlo Therapeutics, and Novartis. The updated guidelines were designed by a multidisciplinary work group of psoriasis experts including dermatologists, a rheumatologist, a cardiologist, and representatives from a patient advocacy organization.
SOURCE: Elmets CA et al. J Am Acad Dermatol. 2020 Jul 29. doi: 10.1016/j.jaad.2020.07.087.
and the National Psoriasis Foundation.
The guidelines, published in the Journal of the American Academy of Dermatology, focus on treatment for adults, and follow the release of other AAD-NPF guidelines on biologics for psoriasis, psoriasis-related comorbidities, pediatric psoriasis, and phototherapy in 2019, and earlier this year, guidelines for systemic nonbiologic treatments. The latest guidelines’ section on topical treatment outlines evidence for the efficacy, effectiveness, and adverse events related to topical steroids, topical tacrolimus and pimecrolimus, vitamin D analogues, tazarotene, moisturizers, salicylic acid, anthralin, coal tar, combinations with biologic agents, and combinations with nonbiologic treatments (methotrexate, cyclosporine, acitretin, and apremilast).
The guidelines noted the “key role” of topical corticosteroids in treating psoriasis “especially for localized disease,” and include a review of the data on low-, moderate-, high-, and ultrahigh-potency topical steroids for psoriasis.
In general, all topical steroids can be used in combination with biologics, according to the guidelines, but the strongest recommendations based on the latest evidence include the addition of an ultra-high potency topical corticosteroid to standard dose etanercept for 12 weeks. Currently, 11 biologics are approved by the Food and Drug Administration for the treatment of psoriasis.
In addition, “while not FDA approved for psoriasis, the topical calcineurin inhibitors tacrolimus and pimecrolimus are often employed in the treatment of psoriasis,” can be helpful for “thinner skin such as facial and intertriginous areas,” and can be steroid sparing when used for more than 4 weeks, according to the guidelines.
Don’t discount the role of patient preferences when choosing topical treatments, the authors noted. “The optimal vehicle choice is the one the patient is mostly likely to use.”
The guidelines also address the evidence for effectiveness, and adverse events in the use of several alternative medicines for psoriasis including traditional Chinese medicine, and the herbal therapies aloe vera and St. John’s wort, as well as the potential role of dietary supplements including fish oil, vitamin D, turmeric, and zinc in managing psoriasis, and the potential role of a gluten-free diet.
In general, research on the efficacy, effectiveness, and potential adverse effects of these strategies are limited, according to the guidelines, although many patients express interest in supplements and herbal products. For example, “Many patients ask about the overall role of vitamin D in skin health. Rather than adding oral vitamin D supplementation, topical therapy with vitamin D agents is effective for the treatment of psoriasis,” the authors noted.
In addition, they noted that mind/body strategies, namely hypnosis and stress reduction or meditation techniques, have been shown to improve symptoms and can be helpful for some patients, but clinical evidence is limited.
The guidelines also addressed methods for assessing disease severity in psoriasis. They recommended using body surface area (BSA) to assess psoriasis severity and patient response to treatment in the clinical setting. However, BSA is a provider assessment tool that “does not take into account location on the body, clinical characteristics of the plaques, symptoms, or quality of life issues,” the authors noted. The Psoriasis Area and Severity Index (PASI) measures erythema, induration, and scaling and is more suited to assessing psoriasis severity and response to treatment in clinical trials rather than in practice, they said.
Prior AAD guidelines on psoriasis were published more than 10 years ago, and major developments including the availability of new biologic drugs and new data on comorbidities have been recognized in the past decade, working group cochair and author of the guidelines Alan Menter, MD, said in an interview.
The key game-changers from previous guidelines include the full section published on comorbidities plus the development of two new important cytokine classes: three IL-17 drugs and three new IL-23 drugs now available for moderate to severe psoriasis, said Dr. Menter, chairman of the division of dermatology at Baylor University Medical Center, Dallas.
Barriers to implementing the guidelines in practice may occur when “third party payers make the decision on which of the 11 biologic drugs now approved for moderate to severe psoriasis should be used,” he noted.
As for next steps in psoriasis studies, “new biomarker research is currently underway,” Dr. Menter said. With 11 biologic agents new formally approved by the FDA for moderate to severe psoriasis, the next steps are to determine which drug is likely to be the most appropriate for each individual patient.
Dr. Menter disclosed relationships with multiple companies that develop and manufacture psoriasis therapies, including Abbott Labs, AbbVie, Amgen, Eli Lilly and Company, Galderma USA, Janssen Pharmaceuticals, LEO Pharma US, Menlo Therapeutics, and Novartis. The updated guidelines were designed by a multidisciplinary work group of psoriasis experts including dermatologists, a rheumatologist, a cardiologist, and representatives from a patient advocacy organization.
SOURCE: Elmets CA et al. J Am Acad Dermatol. 2020 Jul 29. doi: 10.1016/j.jaad.2020.07.087.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY