User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Atopic dermatitis: Abrocitinib effective as flexible dose in phase 3
Key clinical point: Most patients with moderate-to-severe atopic dermatitis (AD) who initially respond to abrocitinib maintained response with reduced dosing. Moreover, rescue treatment with abrocitinib and topical therapy recaptured response in patients who flared.
Major finding: At the end of the maintenance period, flare probability was 18.9%, 42.6%, and 80.9% with abrocitinib 200 mg, abrocitinib 100 mg, and placebo, respectively. Overall, 351 patients entered the rescue period, and Investigator’s Global Assessment of 0/1 response was recaptured in 36.6%, 58.8%, and 81.6% of patients in the abrocitinib 200 mg, abrocitinib 100 mg, and placebo maintenance arms, respectively.
Study details: JADE REGIMEN, a phase 3 trial included 1,233 patients with moderate-to-severe AD. Patients (n=798) who responded to 12 weeks of abrocitinib 200 mg were randomly assigned to abrocitinib 200 mg, abrocitinib 100 mg, or placebo for 40 weeks.
Disclosures: This study was funded by Pfizer Inc. Some of the authors declared serving as advisor, investigator, advisory board member, speaker, lecturer, and/or consultant and/or receiving grants and personal fees from various sources including Pfizer and being present/past employees and shareholders of Pfizer.
Source: Blauvelt A et al. J Am Acad Dermatol. 2021 Aug 16. doi: 10.1016/j.jaad.2021.05.075.
Key clinical point: Most patients with moderate-to-severe atopic dermatitis (AD) who initially respond to abrocitinib maintained response with reduced dosing. Moreover, rescue treatment with abrocitinib and topical therapy recaptured response in patients who flared.
Major finding: At the end of the maintenance period, flare probability was 18.9%, 42.6%, and 80.9% with abrocitinib 200 mg, abrocitinib 100 mg, and placebo, respectively. Overall, 351 patients entered the rescue period, and Investigator’s Global Assessment of 0/1 response was recaptured in 36.6%, 58.8%, and 81.6% of patients in the abrocitinib 200 mg, abrocitinib 100 mg, and placebo maintenance arms, respectively.
Study details: JADE REGIMEN, a phase 3 trial included 1,233 patients with moderate-to-severe AD. Patients (n=798) who responded to 12 weeks of abrocitinib 200 mg were randomly assigned to abrocitinib 200 mg, abrocitinib 100 mg, or placebo for 40 weeks.
Disclosures: This study was funded by Pfizer Inc. Some of the authors declared serving as advisor, investigator, advisory board member, speaker, lecturer, and/or consultant and/or receiving grants and personal fees from various sources including Pfizer and being present/past employees and shareholders of Pfizer.
Source: Blauvelt A et al. J Am Acad Dermatol. 2021 Aug 16. doi: 10.1016/j.jaad.2021.05.075.
Key clinical point: Most patients with moderate-to-severe atopic dermatitis (AD) who initially respond to abrocitinib maintained response with reduced dosing. Moreover, rescue treatment with abrocitinib and topical therapy recaptured response in patients who flared.
Major finding: At the end of the maintenance period, flare probability was 18.9%, 42.6%, and 80.9% with abrocitinib 200 mg, abrocitinib 100 mg, and placebo, respectively. Overall, 351 patients entered the rescue period, and Investigator’s Global Assessment of 0/1 response was recaptured in 36.6%, 58.8%, and 81.6% of patients in the abrocitinib 200 mg, abrocitinib 100 mg, and placebo maintenance arms, respectively.
Study details: JADE REGIMEN, a phase 3 trial included 1,233 patients with moderate-to-severe AD. Patients (n=798) who responded to 12 weeks of abrocitinib 200 mg were randomly assigned to abrocitinib 200 mg, abrocitinib 100 mg, or placebo for 40 weeks.
Disclosures: This study was funded by Pfizer Inc. Some of the authors declared serving as advisor, investigator, advisory board member, speaker, lecturer, and/or consultant and/or receiving grants and personal fees from various sources including Pfizer and being present/past employees and shareholders of Pfizer.
Source: Blauvelt A et al. J Am Acad Dermatol. 2021 Aug 16. doi: 10.1016/j.jaad.2021.05.075.
Atopic dermatitis: Upadacitinib and topical corticosteroids combo shows long-term efficacy and safety in phase 3
Key clinical point: The combination of upadacitinib and topical corticosteroids (TCS) showed long-term efficacy and a favorable safety profile in moderate-to-severe atopic dermatitis (AD).
Major finding: Among patients receiving upadacitinib 15 mg or 30 mg each in combination with TCS, 50.8% (95% confidence interval [CI], 45.1%-56.5%) and 69.0% (95% CI, 63.7%-74.3%) achieved 75% or more improvement in Eczema Area and Severity Index, whereas 33.5% (95% CI, 28.1%-38.9%) and 45.2% (95% CI, 39.5%-50.9%) achieved validated Investigator’s Global Assessment for AD 0/1, respectively, at week 52. Both doses of upadacitinib+TCS were well tolerated with no new safety risks or deaths.
Study details: Findings are 52-week results from AD Up, an ongoing phase 3 trial including 901 patients with chronic AD randomly assigned to upadacitinib 15 mg, upadacitinib 30 mg, or placebo, all in combination with TCS.
Disclosures: This study was funded by AbbVie. The authors declared serving as speaker, consultant, advisor, and investigator and/or receiving consulting fees, honoraria, and grants from various sources including AbbVie. Five authors declared being employees and/or shareholders of AbbVie.
Source: Silverberg JI et al. J Allergy Clin Immunol. 2021 Aug 14. doi: 10.1016/j.jaci.2021.07.036.
Key clinical point: The combination of upadacitinib and topical corticosteroids (TCS) showed long-term efficacy and a favorable safety profile in moderate-to-severe atopic dermatitis (AD).
Major finding: Among patients receiving upadacitinib 15 mg or 30 mg each in combination with TCS, 50.8% (95% confidence interval [CI], 45.1%-56.5%) and 69.0% (95% CI, 63.7%-74.3%) achieved 75% or more improvement in Eczema Area and Severity Index, whereas 33.5% (95% CI, 28.1%-38.9%) and 45.2% (95% CI, 39.5%-50.9%) achieved validated Investigator’s Global Assessment for AD 0/1, respectively, at week 52. Both doses of upadacitinib+TCS were well tolerated with no new safety risks or deaths.
Study details: Findings are 52-week results from AD Up, an ongoing phase 3 trial including 901 patients with chronic AD randomly assigned to upadacitinib 15 mg, upadacitinib 30 mg, or placebo, all in combination with TCS.
Disclosures: This study was funded by AbbVie. The authors declared serving as speaker, consultant, advisor, and investigator and/or receiving consulting fees, honoraria, and grants from various sources including AbbVie. Five authors declared being employees and/or shareholders of AbbVie.
Source: Silverberg JI et al. J Allergy Clin Immunol. 2021 Aug 14. doi: 10.1016/j.jaci.2021.07.036.
Key clinical point: The combination of upadacitinib and topical corticosteroids (TCS) showed long-term efficacy and a favorable safety profile in moderate-to-severe atopic dermatitis (AD).
Major finding: Among patients receiving upadacitinib 15 mg or 30 mg each in combination with TCS, 50.8% (95% confidence interval [CI], 45.1%-56.5%) and 69.0% (95% CI, 63.7%-74.3%) achieved 75% or more improvement in Eczema Area and Severity Index, whereas 33.5% (95% CI, 28.1%-38.9%) and 45.2% (95% CI, 39.5%-50.9%) achieved validated Investigator’s Global Assessment for AD 0/1, respectively, at week 52. Both doses of upadacitinib+TCS were well tolerated with no new safety risks or deaths.
Study details: Findings are 52-week results from AD Up, an ongoing phase 3 trial including 901 patients with chronic AD randomly assigned to upadacitinib 15 mg, upadacitinib 30 mg, or placebo, all in combination with TCS.
Disclosures: This study was funded by AbbVie. The authors declared serving as speaker, consultant, advisor, and investigator and/or receiving consulting fees, honoraria, and grants from various sources including AbbVie. Five authors declared being employees and/or shareholders of AbbVie.
Source: Silverberg JI et al. J Allergy Clin Immunol. 2021 Aug 14. doi: 10.1016/j.jaci.2021.07.036.
Atopic dermatitis tied to symptoms of depression and internalizing behavior in early childhood
Key clinical point: Severe atopic dermatitis (AD) is associated with an almost 2-fold higher risk for depression and internalizing behavior in early childhood.
Major finding: Children with vs. without severe AD were more prone to experience depressive (adjusted odds ratio [aOR], 2.38; 95% confidence interval [CI], 1.21-4.72) and internalizing (aOR, 1.90; 95% CI, 1.14-3.16) symptoms.
Study details: Findings are from a longitudinal, population-based birth cohort study including 11,181 children who were followed up from birth for a mean duration of 10 years.
Disclosures: This study was funded by Wellcome Trust, National Institute of Arthritis and Musculoskeletal and Skin Diseases, National Center for Advancing Translational Sciences, and National Institutes of Health. Dr. Wan and Dr. Abuabara declared receiving research funding from Pfizer.
Source: Kern C et al. JAMA Dermatol. 2021 Sep 1. doi: 10.1001/jamadermatol.2021.2657.
Key clinical point: Severe atopic dermatitis (AD) is associated with an almost 2-fold higher risk for depression and internalizing behavior in early childhood.
Major finding: Children with vs. without severe AD were more prone to experience depressive (adjusted odds ratio [aOR], 2.38; 95% confidence interval [CI], 1.21-4.72) and internalizing (aOR, 1.90; 95% CI, 1.14-3.16) symptoms.
Study details: Findings are from a longitudinal, population-based birth cohort study including 11,181 children who were followed up from birth for a mean duration of 10 years.
Disclosures: This study was funded by Wellcome Trust, National Institute of Arthritis and Musculoskeletal and Skin Diseases, National Center for Advancing Translational Sciences, and National Institutes of Health. Dr. Wan and Dr. Abuabara declared receiving research funding from Pfizer.
Source: Kern C et al. JAMA Dermatol. 2021 Sep 1. doi: 10.1001/jamadermatol.2021.2657.
Key clinical point: Severe atopic dermatitis (AD) is associated with an almost 2-fold higher risk for depression and internalizing behavior in early childhood.
Major finding: Children with vs. without severe AD were more prone to experience depressive (adjusted odds ratio [aOR], 2.38; 95% confidence interval [CI], 1.21-4.72) and internalizing (aOR, 1.90; 95% CI, 1.14-3.16) symptoms.
Study details: Findings are from a longitudinal, population-based birth cohort study including 11,181 children who were followed up from birth for a mean duration of 10 years.
Disclosures: This study was funded by Wellcome Trust, National Institute of Arthritis and Musculoskeletal and Skin Diseases, National Center for Advancing Translational Sciences, and National Institutes of Health. Dr. Wan and Dr. Abuabara declared receiving research funding from Pfizer.
Source: Kern C et al. JAMA Dermatol. 2021 Sep 1. doi: 10.1001/jamadermatol.2021.2657.
Oral abrocitinib plus topical therapy effective in adolescents with atopic dermatitis
Key clinical point: The combination of oral abrocitinib and topical therapy was effective and well tolerated in adolescents with moderate-to-severe atopic dermatitis (AD).
Major finding: At week 12, a significantly higher proportion of patients treated with abrocitinib 200 mg or 100 mg vs placebo achieved an Investigator’s Global Assessment response of 0/1 (46.2% and 41.6% vs 24.5%) and 75% or more improvement in Eczema Area and Severity Index (72.0% and 68.5% vs 41.5%; P < .05 for all). Serious adverse events were reported by less than 3% of patients.
Study details: Findings are from JADE TEEN, a phase 3 trial including 285 adolescents with moderate-to-severe AD and an inadequate response to topical medication or in need for systemic therapy, who were randomly assigned to receive once-daily oral abrocitinib, 200 mg or 100 mg, or placebo for 12 weeks in combination with topical therapy.
Disclosures: This study was funded by Pfizer Inc. The authors declared receiving nonfinancial support, grants, and personal fees from several sources including Pfizer. Four authors reported being employees and/or shareholders of Pfizer.
Source: Eichenfield LF et al. JAMA Dermatol. 2021 Aug 18. doi: 10.1001/jamadermatol.2021.2830.
Key clinical point: The combination of oral abrocitinib and topical therapy was effective and well tolerated in adolescents with moderate-to-severe atopic dermatitis (AD).
Major finding: At week 12, a significantly higher proportion of patients treated with abrocitinib 200 mg or 100 mg vs placebo achieved an Investigator’s Global Assessment response of 0/1 (46.2% and 41.6% vs 24.5%) and 75% or more improvement in Eczema Area and Severity Index (72.0% and 68.5% vs 41.5%; P < .05 for all). Serious adverse events were reported by less than 3% of patients.
Study details: Findings are from JADE TEEN, a phase 3 trial including 285 adolescents with moderate-to-severe AD and an inadequate response to topical medication or in need for systemic therapy, who were randomly assigned to receive once-daily oral abrocitinib, 200 mg or 100 mg, or placebo for 12 weeks in combination with topical therapy.
Disclosures: This study was funded by Pfizer Inc. The authors declared receiving nonfinancial support, grants, and personal fees from several sources including Pfizer. Four authors reported being employees and/or shareholders of Pfizer.
Source: Eichenfield LF et al. JAMA Dermatol. 2021 Aug 18. doi: 10.1001/jamadermatol.2021.2830.
Key clinical point: The combination of oral abrocitinib and topical therapy was effective and well tolerated in adolescents with moderate-to-severe atopic dermatitis (AD).
Major finding: At week 12, a significantly higher proportion of patients treated with abrocitinib 200 mg or 100 mg vs placebo achieved an Investigator’s Global Assessment response of 0/1 (46.2% and 41.6% vs 24.5%) and 75% or more improvement in Eczema Area and Severity Index (72.0% and 68.5% vs 41.5%; P < .05 for all). Serious adverse events were reported by less than 3% of patients.
Study details: Findings are from JADE TEEN, a phase 3 trial including 285 adolescents with moderate-to-severe AD and an inadequate response to topical medication or in need for systemic therapy, who were randomly assigned to receive once-daily oral abrocitinib, 200 mg or 100 mg, or placebo for 12 weeks in combination with topical therapy.
Disclosures: This study was funded by Pfizer Inc. The authors declared receiving nonfinancial support, grants, and personal fees from several sources including Pfizer. Four authors reported being employees and/or shareholders of Pfizer.
Source: Eichenfield LF et al. JAMA Dermatol. 2021 Aug 18. doi: 10.1001/jamadermatol.2021.2830.
FDA panel backs Pfizer's COVID booster for 65 and older, those at high risk
An expert panel that advises the Food and Drug Administration on its regulatory decisions voted Sept. 17 against recommending third doses of Pfizer’s COVID-19 vaccine for younger Americans.
But they didn’t kill the idea of booster shots completely.
In a dramatic, last-minute pivot, the 18 members of the FDA’s Vaccines and Related Biological Products Advisory Committee unanimously voted to recommend the FDA make boosters available for seniors and others at high risk of severe outcomes from COVID-19, including health care workers.
The 16-2 vote was a rebuttal to Pfizer’s initial request. The company had asked the FDA to allow it to offer third doses to all Americans over the age of 16 at least six months after their second shot.
The company requested an amendment to the full approval the FDA granted in August. That is the typical way boosters are authorized in the U.S., but it requires a higher bar of evidence and more regulatory scrutiny than the agency had been able to give since Pfizer filed for the change just days after its vaccine was granted full approval.
The committee’s actions were also a rebuff to the Biden administration, which announced before the FDA approved them that boosters would be rolled out to the general public Sept. 20. The announcement triggered the resignations of two of the agency’s top vaccine reviewers, who both participated in the Sept. 17 meeting.
After initially voting against Pfizer’s request to amend its license, the committee then worked on the fly with FDA officials to craft a strategy that would allow third doses to be offered under an emergency use authorization (EUA).
An EUA requires a lower standard of evidence and is more specific. It will restrict third doses to a more defined population than a change to the license would. It will also require Pfizer to continue to monitor the safety of third doses as they begin to be administered.
“This should demonstrate to the public that the members of this committee are independent of the FDA and that we do, in fact, bring our voices to the table when we are asked to serve on this committee,” said Archana Chattergee, MD, a pediatric infectious disease specialist who is dean of the Chicago Medical School at Rosalind Franklin University in Illinois.
The FDA doesn’t have to follow the committee’s recommendation, but almost certainly will, though regulators said they may still make some changes.
“We are not bound at FDA by your vote, we can tweak this,” said Peter Marks, MD, director of the Center for Biologics Evaluation and Research at the FDA. Dr. Marks participated in the meeting and helped to draft the revised proposal.
If the FDA issues the recommended EUA, a council of independent advisors to the CDC will make specific recommendations about how the third doses should be given. After the CDC director weighs in, boosters will begin rolling out to the public.
Moderna submitted data to the FDA on Sept. 1 in support of adding a booster dose to its regimen. The agency has not yet scheduled a public review of that data.
The Biden administration is prepared to administer shots as soon as they get the green light, Surgeon General Vivek Murthy, MD, said at a White House briefing earlier Sept. 17.
"This process is consistent with what we outlined in August where our goals were to stay ahead of the virus," Dr. Murthy said. "Our goal then and now is to protect the health and well-being of the public. As soon as the FDA and CDC complete their evaluations, we will be ready to move forward accordingly."
He added, "We've used this time since our August announcement to communicate and coordinate with pharmacy partners, nursing homes, states, and localities."
White House COVID-19 Response Coordinator Jeff Zients said vaccine supply is "in good shape for all Americans to get boosters as recommended."
Taking cues from Israel
In considering Pfizer’s original request, the committee overwhelmingly felt that they didn’t have enough information to say that the benefits of an additional dose of vaccine in 16- and 17-year-olds would outweigh its risk. Teens have the highest risk of rare heart inflammation after vaccination, a side effect known as myocarditis. It is not known how the vaccines are causing these cases of heart swelling. Most who have been diagnosed with the condition have recovered, though some have needed hospital care.
Pfizer didn’t include 16- and 17-year-olds in its studies of boosters, which included about 300 people between the ages of 18 and 55. The company acknowledged that gap in its data but pointed to FDA guidance that said evidence from adults could be extrapolated to teens.
“We don’t know that much about risks,” said committee member Eric Rubin, MD, who is editor-in-chief of the New England Journal of Medicine.
Much of the data on the potential benefits and harms of third Pfizer doses comes from Israel, which first began rolling out boosters to older adults in July.
In a highly anticipated presentation, Sharon Alroy-Preis, Israel’s director of public health services, joined the meeting to describe Israel’s experience with boosters.
Israel began to see a third surge of COVID-19 cases in December.
“This was after having two waves and two lockdowns,” Ms. Alroy-Preis said. By the third surge, she said, Israelis were tired.
“We decided on a lockdown, but the compliance of the public wasn’t as it was in the previous two waves,” she said.
Then the vaccine arrived. Israel started vaccinations as soon as the FDA approved it, and they quickly vaccinated a high percentage of their population, about 3 months faster than the rest of the world.
All vaccinations are reported and tracked by the Ministry of Health, so the country is able to keep close tabs on how well the shots are working.
As vaccines rolled out, cases fell dramatically. The pandemic seemed to be behind them. Delta arrived in March. By June, their cases were doubling every 10 days, despite about 80% of their most vulnerable adults being fully vaccinated, she said.
Most concerning was that about 60% of severe cases were breakthrough cases in fully vaccinated individuals.
“We had to stop and figure out, was this a Delta issue,” she said. “Or was this a waning immunity issue.”
“We had some clue that it might not be the Delta variant, at least not alone,” she said.
People who had originally been first in line for the vaccines, seniors and health care workers, were having the highest rates of breakthrough infections. People further away from their second dose were more likely to get a breakthrough infection.
Ms. Alroy-Preis said that if they had not started booster doses in July, their hospitals would have been overwhelmed. They had projected that they would have 2,000 cases in the hospital each day.
Boosters have helped to flatten the curve, though they are still seeing a significant number of infections.
Data from Israel presented at the meeting show boosters are largely safe and effective at reducing severe outcomes in seniors. Israeli experience also showed that third doses, which generate very high levels of neutralizing antibodies—the first and fastest line of the body’s immune defense - -may also slow transmission of the virus.
Key differences in the U.S.
The benefit of slowing down the explosive spread of a highly contagious virus was tantalizing, but many members noted that circumstances in Israel are very different than in the United States. Israel went into its current Delta surge already having high levels of vaccination in its population. They also relied on the Pfizer vaccine almost exclusively for their campaign.
The United States used a different mix of vaccines – Pfizer, Moderna, and Johnson & Johnson -- and doesn’t have the same high level of vaccination coverage of its population.
In the United States, transmission is mainly being driven by unvaccinated people, Dr. Rubin noted.
“That really means the primary benefit is going to be in reducing disease,” he said, “And we know the people who are going to benefit from that … and those are the kinds of people the FDA has already approved a third dose for,” he said, referring to those with underlying health conditions.
But Israel only began vaccinating younger people a few weeks ago. Most are still within a window where rare risks like myocarditis could appear, Rubin noted.
He and other members of the committee said they wished they had more information about the safety of third doses in younger adults.
“We don’t have that right now, and I don’t think I would be comfortable giving it to a 16-year-old,” he said.
At the same time, the primary benefit for third doses would be in preventing severe disease, and overall, data from the United States and other countries show that two doses of the vaccines remain highly effective at preventing hospitalization and death.
Asked why Israel began to see more severe cases in fully vaccinated people, the CDC’s Sara Oliver, MD, a disease detective with the CDC, said it was probably due to a mix of factors including the fact that Israel defines severe cases a little differently.
In the United States, a severe case is generally a person who has to be hospitalized or who has died from the infection. In Israel, a person with a severe case is someone who has an elevated respiratory rate and someone who has a blood oxygen level less than 94%. In the United States, that kind of patient wouldn’t necessarily be hospitalized.
In the end, one of the two committee members who wanted full approval for Pfizer’s third doses said he was satisfied with the outcome.
Mark Sawyer, MD, a professor of pediatrics and infectious disease at the University of California at San Diego, said he voted yes on the first question because he thought full approval was the best way to give doctors the flexibility to prescribe the shots to vulnerable individuals.
“I’m really glad we authorized a vaccine for a third dose, and I plan to go out and get my vaccine this afternoon,” Dr. Sawyer said, noting that he was at high risk as a health care provider.
This article was updated 9/19/21.
A version of this article first appeared on Medscape.com.
An expert panel that advises the Food and Drug Administration on its regulatory decisions voted Sept. 17 against recommending third doses of Pfizer’s COVID-19 vaccine for younger Americans.
But they didn’t kill the idea of booster shots completely.
In a dramatic, last-minute pivot, the 18 members of the FDA’s Vaccines and Related Biological Products Advisory Committee unanimously voted to recommend the FDA make boosters available for seniors and others at high risk of severe outcomes from COVID-19, including health care workers.
The 16-2 vote was a rebuttal to Pfizer’s initial request. The company had asked the FDA to allow it to offer third doses to all Americans over the age of 16 at least six months after their second shot.
The company requested an amendment to the full approval the FDA granted in August. That is the typical way boosters are authorized in the U.S., but it requires a higher bar of evidence and more regulatory scrutiny than the agency had been able to give since Pfizer filed for the change just days after its vaccine was granted full approval.
The committee’s actions were also a rebuff to the Biden administration, which announced before the FDA approved them that boosters would be rolled out to the general public Sept. 20. The announcement triggered the resignations of two of the agency’s top vaccine reviewers, who both participated in the Sept. 17 meeting.
After initially voting against Pfizer’s request to amend its license, the committee then worked on the fly with FDA officials to craft a strategy that would allow third doses to be offered under an emergency use authorization (EUA).
An EUA requires a lower standard of evidence and is more specific. It will restrict third doses to a more defined population than a change to the license would. It will also require Pfizer to continue to monitor the safety of third doses as they begin to be administered.
“This should demonstrate to the public that the members of this committee are independent of the FDA and that we do, in fact, bring our voices to the table when we are asked to serve on this committee,” said Archana Chattergee, MD, a pediatric infectious disease specialist who is dean of the Chicago Medical School at Rosalind Franklin University in Illinois.
The FDA doesn’t have to follow the committee’s recommendation, but almost certainly will, though regulators said they may still make some changes.
“We are not bound at FDA by your vote, we can tweak this,” said Peter Marks, MD, director of the Center for Biologics Evaluation and Research at the FDA. Dr. Marks participated in the meeting and helped to draft the revised proposal.
If the FDA issues the recommended EUA, a council of independent advisors to the CDC will make specific recommendations about how the third doses should be given. After the CDC director weighs in, boosters will begin rolling out to the public.
Moderna submitted data to the FDA on Sept. 1 in support of adding a booster dose to its regimen. The agency has not yet scheduled a public review of that data.
The Biden administration is prepared to administer shots as soon as they get the green light, Surgeon General Vivek Murthy, MD, said at a White House briefing earlier Sept. 17.
"This process is consistent with what we outlined in August where our goals were to stay ahead of the virus," Dr. Murthy said. "Our goal then and now is to protect the health and well-being of the public. As soon as the FDA and CDC complete their evaluations, we will be ready to move forward accordingly."
He added, "We've used this time since our August announcement to communicate and coordinate with pharmacy partners, nursing homes, states, and localities."
White House COVID-19 Response Coordinator Jeff Zients said vaccine supply is "in good shape for all Americans to get boosters as recommended."
Taking cues from Israel
In considering Pfizer’s original request, the committee overwhelmingly felt that they didn’t have enough information to say that the benefits of an additional dose of vaccine in 16- and 17-year-olds would outweigh its risk. Teens have the highest risk of rare heart inflammation after vaccination, a side effect known as myocarditis. It is not known how the vaccines are causing these cases of heart swelling. Most who have been diagnosed with the condition have recovered, though some have needed hospital care.
Pfizer didn’t include 16- and 17-year-olds in its studies of boosters, which included about 300 people between the ages of 18 and 55. The company acknowledged that gap in its data but pointed to FDA guidance that said evidence from adults could be extrapolated to teens.
“We don’t know that much about risks,” said committee member Eric Rubin, MD, who is editor-in-chief of the New England Journal of Medicine.
Much of the data on the potential benefits and harms of third Pfizer doses comes from Israel, which first began rolling out boosters to older adults in July.
In a highly anticipated presentation, Sharon Alroy-Preis, Israel’s director of public health services, joined the meeting to describe Israel’s experience with boosters.
Israel began to see a third surge of COVID-19 cases in December.
“This was after having two waves and two lockdowns,” Ms. Alroy-Preis said. By the third surge, she said, Israelis were tired.
“We decided on a lockdown, but the compliance of the public wasn’t as it was in the previous two waves,” she said.
Then the vaccine arrived. Israel started vaccinations as soon as the FDA approved it, and they quickly vaccinated a high percentage of their population, about 3 months faster than the rest of the world.
All vaccinations are reported and tracked by the Ministry of Health, so the country is able to keep close tabs on how well the shots are working.
As vaccines rolled out, cases fell dramatically. The pandemic seemed to be behind them. Delta arrived in March. By June, their cases were doubling every 10 days, despite about 80% of their most vulnerable adults being fully vaccinated, she said.
Most concerning was that about 60% of severe cases were breakthrough cases in fully vaccinated individuals.
“We had to stop and figure out, was this a Delta issue,” she said. “Or was this a waning immunity issue.”
“We had some clue that it might not be the Delta variant, at least not alone,” she said.
People who had originally been first in line for the vaccines, seniors and health care workers, were having the highest rates of breakthrough infections. People further away from their second dose were more likely to get a breakthrough infection.
Ms. Alroy-Preis said that if they had not started booster doses in July, their hospitals would have been overwhelmed. They had projected that they would have 2,000 cases in the hospital each day.
Boosters have helped to flatten the curve, though they are still seeing a significant number of infections.
Data from Israel presented at the meeting show boosters are largely safe and effective at reducing severe outcomes in seniors. Israeli experience also showed that third doses, which generate very high levels of neutralizing antibodies—the first and fastest line of the body’s immune defense - -may also slow transmission of the virus.
Key differences in the U.S.
The benefit of slowing down the explosive spread of a highly contagious virus was tantalizing, but many members noted that circumstances in Israel are very different than in the United States. Israel went into its current Delta surge already having high levels of vaccination in its population. They also relied on the Pfizer vaccine almost exclusively for their campaign.
The United States used a different mix of vaccines – Pfizer, Moderna, and Johnson & Johnson -- and doesn’t have the same high level of vaccination coverage of its population.
In the United States, transmission is mainly being driven by unvaccinated people, Dr. Rubin noted.
“That really means the primary benefit is going to be in reducing disease,” he said, “And we know the people who are going to benefit from that … and those are the kinds of people the FDA has already approved a third dose for,” he said, referring to those with underlying health conditions.
But Israel only began vaccinating younger people a few weeks ago. Most are still within a window where rare risks like myocarditis could appear, Rubin noted.
He and other members of the committee said they wished they had more information about the safety of third doses in younger adults.
“We don’t have that right now, and I don’t think I would be comfortable giving it to a 16-year-old,” he said.
At the same time, the primary benefit for third doses would be in preventing severe disease, and overall, data from the United States and other countries show that two doses of the vaccines remain highly effective at preventing hospitalization and death.
Asked why Israel began to see more severe cases in fully vaccinated people, the CDC’s Sara Oliver, MD, a disease detective with the CDC, said it was probably due to a mix of factors including the fact that Israel defines severe cases a little differently.
In the United States, a severe case is generally a person who has to be hospitalized or who has died from the infection. In Israel, a person with a severe case is someone who has an elevated respiratory rate and someone who has a blood oxygen level less than 94%. In the United States, that kind of patient wouldn’t necessarily be hospitalized.
In the end, one of the two committee members who wanted full approval for Pfizer’s third doses said he was satisfied with the outcome.
Mark Sawyer, MD, a professor of pediatrics and infectious disease at the University of California at San Diego, said he voted yes on the first question because he thought full approval was the best way to give doctors the flexibility to prescribe the shots to vulnerable individuals.
“I’m really glad we authorized a vaccine for a third dose, and I plan to go out and get my vaccine this afternoon,” Dr. Sawyer said, noting that he was at high risk as a health care provider.
This article was updated 9/19/21.
A version of this article first appeared on Medscape.com.
An expert panel that advises the Food and Drug Administration on its regulatory decisions voted Sept. 17 against recommending third doses of Pfizer’s COVID-19 vaccine for younger Americans.
But they didn’t kill the idea of booster shots completely.
In a dramatic, last-minute pivot, the 18 members of the FDA’s Vaccines and Related Biological Products Advisory Committee unanimously voted to recommend the FDA make boosters available for seniors and others at high risk of severe outcomes from COVID-19, including health care workers.
The 16-2 vote was a rebuttal to Pfizer’s initial request. The company had asked the FDA to allow it to offer third doses to all Americans over the age of 16 at least six months after their second shot.
The company requested an amendment to the full approval the FDA granted in August. That is the typical way boosters are authorized in the U.S., but it requires a higher bar of evidence and more regulatory scrutiny than the agency had been able to give since Pfizer filed for the change just days after its vaccine was granted full approval.
The committee’s actions were also a rebuff to the Biden administration, which announced before the FDA approved them that boosters would be rolled out to the general public Sept. 20. The announcement triggered the resignations of two of the agency’s top vaccine reviewers, who both participated in the Sept. 17 meeting.
After initially voting against Pfizer’s request to amend its license, the committee then worked on the fly with FDA officials to craft a strategy that would allow third doses to be offered under an emergency use authorization (EUA).
An EUA requires a lower standard of evidence and is more specific. It will restrict third doses to a more defined population than a change to the license would. It will also require Pfizer to continue to monitor the safety of third doses as they begin to be administered.
“This should demonstrate to the public that the members of this committee are independent of the FDA and that we do, in fact, bring our voices to the table when we are asked to serve on this committee,” said Archana Chattergee, MD, a pediatric infectious disease specialist who is dean of the Chicago Medical School at Rosalind Franklin University in Illinois.
The FDA doesn’t have to follow the committee’s recommendation, but almost certainly will, though regulators said they may still make some changes.
“We are not bound at FDA by your vote, we can tweak this,” said Peter Marks, MD, director of the Center for Biologics Evaluation and Research at the FDA. Dr. Marks participated in the meeting and helped to draft the revised proposal.
If the FDA issues the recommended EUA, a council of independent advisors to the CDC will make specific recommendations about how the third doses should be given. After the CDC director weighs in, boosters will begin rolling out to the public.
Moderna submitted data to the FDA on Sept. 1 in support of adding a booster dose to its regimen. The agency has not yet scheduled a public review of that data.
The Biden administration is prepared to administer shots as soon as they get the green light, Surgeon General Vivek Murthy, MD, said at a White House briefing earlier Sept. 17.
"This process is consistent with what we outlined in August where our goals were to stay ahead of the virus," Dr. Murthy said. "Our goal then and now is to protect the health and well-being of the public. As soon as the FDA and CDC complete their evaluations, we will be ready to move forward accordingly."
He added, "We've used this time since our August announcement to communicate and coordinate with pharmacy partners, nursing homes, states, and localities."
White House COVID-19 Response Coordinator Jeff Zients said vaccine supply is "in good shape for all Americans to get boosters as recommended."
Taking cues from Israel
In considering Pfizer’s original request, the committee overwhelmingly felt that they didn’t have enough information to say that the benefits of an additional dose of vaccine in 16- and 17-year-olds would outweigh its risk. Teens have the highest risk of rare heart inflammation after vaccination, a side effect known as myocarditis. It is not known how the vaccines are causing these cases of heart swelling. Most who have been diagnosed with the condition have recovered, though some have needed hospital care.
Pfizer didn’t include 16- and 17-year-olds in its studies of boosters, which included about 300 people between the ages of 18 and 55. The company acknowledged that gap in its data but pointed to FDA guidance that said evidence from adults could be extrapolated to teens.
“We don’t know that much about risks,” said committee member Eric Rubin, MD, who is editor-in-chief of the New England Journal of Medicine.
Much of the data on the potential benefits and harms of third Pfizer doses comes from Israel, which first began rolling out boosters to older adults in July.
In a highly anticipated presentation, Sharon Alroy-Preis, Israel’s director of public health services, joined the meeting to describe Israel’s experience with boosters.
Israel began to see a third surge of COVID-19 cases in December.
“This was after having two waves and two lockdowns,” Ms. Alroy-Preis said. By the third surge, she said, Israelis were tired.
“We decided on a lockdown, but the compliance of the public wasn’t as it was in the previous two waves,” she said.
Then the vaccine arrived. Israel started vaccinations as soon as the FDA approved it, and they quickly vaccinated a high percentage of their population, about 3 months faster than the rest of the world.
All vaccinations are reported and tracked by the Ministry of Health, so the country is able to keep close tabs on how well the shots are working.
As vaccines rolled out, cases fell dramatically. The pandemic seemed to be behind them. Delta arrived in March. By June, their cases were doubling every 10 days, despite about 80% of their most vulnerable adults being fully vaccinated, she said.
Most concerning was that about 60% of severe cases were breakthrough cases in fully vaccinated individuals.
“We had to stop and figure out, was this a Delta issue,” she said. “Or was this a waning immunity issue.”
“We had some clue that it might not be the Delta variant, at least not alone,” she said.
People who had originally been first in line for the vaccines, seniors and health care workers, were having the highest rates of breakthrough infections. People further away from their second dose were more likely to get a breakthrough infection.
Ms. Alroy-Preis said that if they had not started booster doses in July, their hospitals would have been overwhelmed. They had projected that they would have 2,000 cases in the hospital each day.
Boosters have helped to flatten the curve, though they are still seeing a significant number of infections.
Data from Israel presented at the meeting show boosters are largely safe and effective at reducing severe outcomes in seniors. Israeli experience also showed that third doses, which generate very high levels of neutralizing antibodies—the first and fastest line of the body’s immune defense - -may also slow transmission of the virus.
Key differences in the U.S.
The benefit of slowing down the explosive spread of a highly contagious virus was tantalizing, but many members noted that circumstances in Israel are very different than in the United States. Israel went into its current Delta surge already having high levels of vaccination in its population. They also relied on the Pfizer vaccine almost exclusively for their campaign.
The United States used a different mix of vaccines – Pfizer, Moderna, and Johnson & Johnson -- and doesn’t have the same high level of vaccination coverage of its population.
In the United States, transmission is mainly being driven by unvaccinated people, Dr. Rubin noted.
“That really means the primary benefit is going to be in reducing disease,” he said, “And we know the people who are going to benefit from that … and those are the kinds of people the FDA has already approved a third dose for,” he said, referring to those with underlying health conditions.
But Israel only began vaccinating younger people a few weeks ago. Most are still within a window where rare risks like myocarditis could appear, Rubin noted.
He and other members of the committee said they wished they had more information about the safety of third doses in younger adults.
“We don’t have that right now, and I don’t think I would be comfortable giving it to a 16-year-old,” he said.
At the same time, the primary benefit for third doses would be in preventing severe disease, and overall, data from the United States and other countries show that two doses of the vaccines remain highly effective at preventing hospitalization and death.
Asked why Israel began to see more severe cases in fully vaccinated people, the CDC’s Sara Oliver, MD, a disease detective with the CDC, said it was probably due to a mix of factors including the fact that Israel defines severe cases a little differently.
In the United States, a severe case is generally a person who has to be hospitalized or who has died from the infection. In Israel, a person with a severe case is someone who has an elevated respiratory rate and someone who has a blood oxygen level less than 94%. In the United States, that kind of patient wouldn’t necessarily be hospitalized.
In the end, one of the two committee members who wanted full approval for Pfizer’s third doses said he was satisfied with the outcome.
Mark Sawyer, MD, a professor of pediatrics and infectious disease at the University of California at San Diego, said he voted yes on the first question because he thought full approval was the best way to give doctors the flexibility to prescribe the shots to vulnerable individuals.
“I’m really glad we authorized a vaccine for a third dose, and I plan to go out and get my vaccine this afternoon,” Dr. Sawyer said, noting that he was at high risk as a health care provider.
This article was updated 9/19/21.
A version of this article first appeared on Medscape.com.
Acid series: Lactic acid
One of the most commonly used organic acids used on the skin, lactic acid, has been used for over 3 decades. Originally derived from milk or plant-derived sugars, this gentle exfoliating acid can be used in peels, serums, masks, and toners, and has the additional benefit of hydrating the skin. Lactic acid is formulated in concentrations from 2% to 50%; however, because of its large molecular size, it doesn’t penetrate the deeper layers of the dermis to the same extent as the other alpha-hydroxy acids (AHAs), such as glycolic acid. Thus, it is one of the gentler exfoliants and one that can be used in sensitive skin or darker skin types.
Despite its mild peeling effects, lactic acid is best used to treat xerotic skin because of its function as a humectant, drawing moisture into the stratum corneum. Similar to the other AHAs, lactic acid has also been shown to decrease melanogenesis and is a gentle treatment for skin hyperpigmentation, particularly in skin of color. Side effects include peeling, stinging, erythema, photosensitivity, and hyperpigmentation when improperly used.
Very little clinical research has been reported in the last 20 years as to the uses and benefits of lactic acid in skincare. In my clinical experience, daily use of lactic acid is more effective and has more long-term benefits for hydration and rejuvenation of the skin than the other AHAs. Concentrations of 10%-15% used daily on the skin as a mild exfoliant and humectant have shown to improve texture, decrease pigmentation and improve fine lines – without thinning of the skin seen with the deeper dermal penetrating acids.
Confusion in the market has also risen as many over-the-counter brands have included ammonium lactate in their portfolio of moisturizers. Ammonium lactate is a combination of ammonium hydroxide and lactic acid, or the salt of lactic acid. A comparative study evaluating the difference between 5% lactic acid and 12% ammonium lactate for the treatment of xerosis showed that ammonium lactate was significantly more effective at reducing xerosis. It is widely used in the treatment of keratosis pilaris, calluses, xerosis, and ichthyosis.
Widespread use of lactic acid has not gotten as much glory as that of glycolic acid. However, in clinical practice, its functions are more widespread. It is a much safer acid to use, and its added benefit of increasing hydration of the skin is crucial in its long-term use for both photoaging and the prevention of wrinkles. With any acid, the exfoliating properties must be treated with adequate hydration and barrier repair.
The intrinsic moisturizing effect of lactic acid makes it a much more well-rounded acid and that can be used for longer periods of time in a broader spectrum of patients.
Dr. Lily Talakoub and Dr. Naissan O. Wesley are cocontributors to this column. Dr. Talakoub is in private practice in McLean, Va. Dr. Wesley practices dermatology in Beverly Hills, Calif. This month’s column is by Dr. Talakoub. Write to them at [email protected]. They had no relevant disclosures.
One of the most commonly used organic acids used on the skin, lactic acid, has been used for over 3 decades. Originally derived from milk or plant-derived sugars, this gentle exfoliating acid can be used in peels, serums, masks, and toners, and has the additional benefit of hydrating the skin. Lactic acid is formulated in concentrations from 2% to 50%; however, because of its large molecular size, it doesn’t penetrate the deeper layers of the dermis to the same extent as the other alpha-hydroxy acids (AHAs), such as glycolic acid. Thus, it is one of the gentler exfoliants and one that can be used in sensitive skin or darker skin types.
Despite its mild peeling effects, lactic acid is best used to treat xerotic skin because of its function as a humectant, drawing moisture into the stratum corneum. Similar to the other AHAs, lactic acid has also been shown to decrease melanogenesis and is a gentle treatment for skin hyperpigmentation, particularly in skin of color. Side effects include peeling, stinging, erythema, photosensitivity, and hyperpigmentation when improperly used.
Very little clinical research has been reported in the last 20 years as to the uses and benefits of lactic acid in skincare. In my clinical experience, daily use of lactic acid is more effective and has more long-term benefits for hydration and rejuvenation of the skin than the other AHAs. Concentrations of 10%-15% used daily on the skin as a mild exfoliant and humectant have shown to improve texture, decrease pigmentation and improve fine lines – without thinning of the skin seen with the deeper dermal penetrating acids.
Confusion in the market has also risen as many over-the-counter brands have included ammonium lactate in their portfolio of moisturizers. Ammonium lactate is a combination of ammonium hydroxide and lactic acid, or the salt of lactic acid. A comparative study evaluating the difference between 5% lactic acid and 12% ammonium lactate for the treatment of xerosis showed that ammonium lactate was significantly more effective at reducing xerosis. It is widely used in the treatment of keratosis pilaris, calluses, xerosis, and ichthyosis.
Widespread use of lactic acid has not gotten as much glory as that of glycolic acid. However, in clinical practice, its functions are more widespread. It is a much safer acid to use, and its added benefit of increasing hydration of the skin is crucial in its long-term use for both photoaging and the prevention of wrinkles. With any acid, the exfoliating properties must be treated with adequate hydration and barrier repair.
The intrinsic moisturizing effect of lactic acid makes it a much more well-rounded acid and that can be used for longer periods of time in a broader spectrum of patients.
Dr. Lily Talakoub and Dr. Naissan O. Wesley are cocontributors to this column. Dr. Talakoub is in private practice in McLean, Va. Dr. Wesley practices dermatology in Beverly Hills, Calif. This month’s column is by Dr. Talakoub. Write to them at [email protected]. They had no relevant disclosures.
One of the most commonly used organic acids used on the skin, lactic acid, has been used for over 3 decades. Originally derived from milk or plant-derived sugars, this gentle exfoliating acid can be used in peels, serums, masks, and toners, and has the additional benefit of hydrating the skin. Lactic acid is formulated in concentrations from 2% to 50%; however, because of its large molecular size, it doesn’t penetrate the deeper layers of the dermis to the same extent as the other alpha-hydroxy acids (AHAs), such as glycolic acid. Thus, it is one of the gentler exfoliants and one that can be used in sensitive skin or darker skin types.
Despite its mild peeling effects, lactic acid is best used to treat xerotic skin because of its function as a humectant, drawing moisture into the stratum corneum. Similar to the other AHAs, lactic acid has also been shown to decrease melanogenesis and is a gentle treatment for skin hyperpigmentation, particularly in skin of color. Side effects include peeling, stinging, erythema, photosensitivity, and hyperpigmentation when improperly used.
Very little clinical research has been reported in the last 20 years as to the uses and benefits of lactic acid in skincare. In my clinical experience, daily use of lactic acid is more effective and has more long-term benefits for hydration and rejuvenation of the skin than the other AHAs. Concentrations of 10%-15% used daily on the skin as a mild exfoliant and humectant have shown to improve texture, decrease pigmentation and improve fine lines – without thinning of the skin seen with the deeper dermal penetrating acids.
Confusion in the market has also risen as many over-the-counter brands have included ammonium lactate in their portfolio of moisturizers. Ammonium lactate is a combination of ammonium hydroxide and lactic acid, or the salt of lactic acid. A comparative study evaluating the difference between 5% lactic acid and 12% ammonium lactate for the treatment of xerosis showed that ammonium lactate was significantly more effective at reducing xerosis. It is widely used in the treatment of keratosis pilaris, calluses, xerosis, and ichthyosis.
Widespread use of lactic acid has not gotten as much glory as that of glycolic acid. However, in clinical practice, its functions are more widespread. It is a much safer acid to use, and its added benefit of increasing hydration of the skin is crucial in its long-term use for both photoaging and the prevention of wrinkles. With any acid, the exfoliating properties must be treated with adequate hydration and barrier repair.
The intrinsic moisturizing effect of lactic acid makes it a much more well-rounded acid and that can be used for longer periods of time in a broader spectrum of patients.
Dr. Lily Talakoub and Dr. Naissan O. Wesley are cocontributors to this column. Dr. Talakoub is in private practice in McLean, Va. Dr. Wesley practices dermatology in Beverly Hills, Calif. This month’s column is by Dr. Talakoub. Write to them at [email protected]. They had no relevant disclosures.
Acne Vulgaris
THE COMPARISON
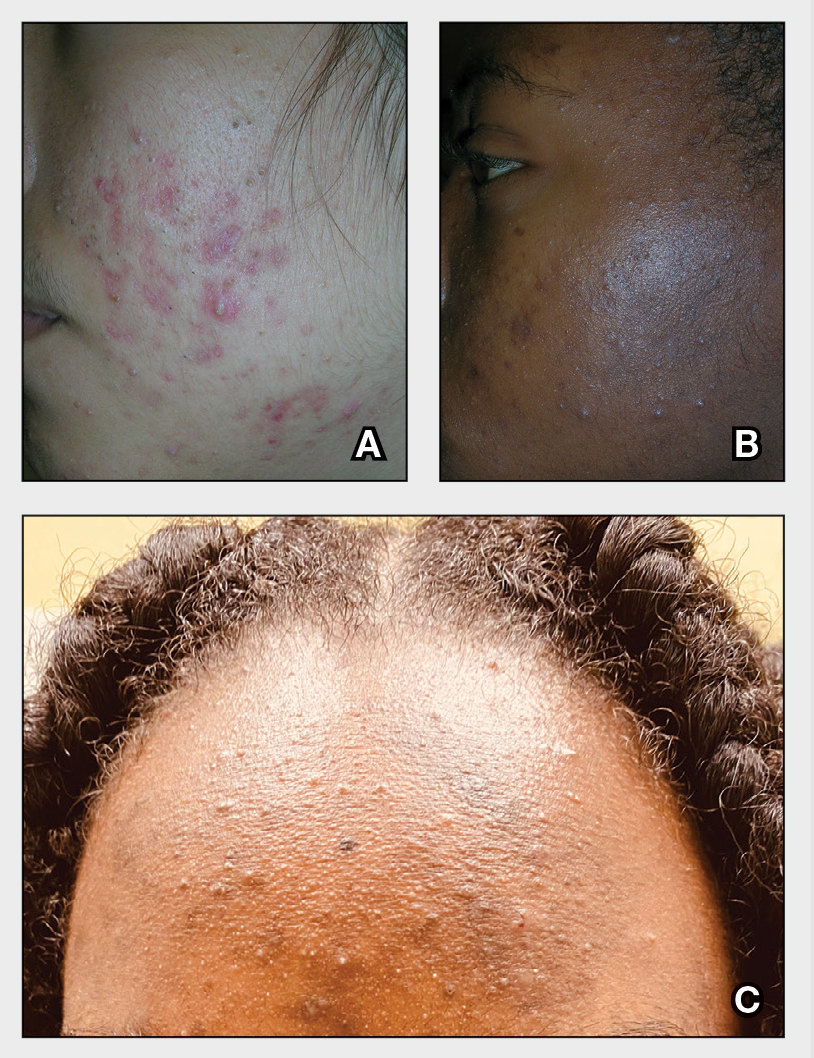
A A 27-year-old Hispanic woman with comedonal and inflammatory acne. Erythema is prominent around the inflammatory lesions. Note the pustule on the cheek surrounded by pink color.
B A teenaged Black boy with acne papules and pustules on the face. There are comedones, hyperpigmented macules, and pustules on the cheek.
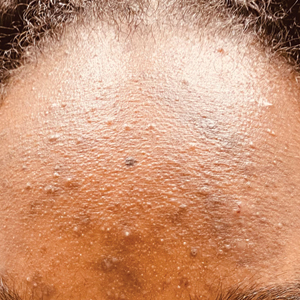
C A teenaged Black girl with pomade acne. The patient used various hair care products, which obstructed the pilosebaceous units on the forehead.
Epidemiology
Acne is a leading dermatologic condition in individuals with skin of color in the United States.1
Key clinical features in people with darker skin tones include:
- erythematous or hyperpigmented papules or comedones
- hyperpigmented macules and postinflammatory hyperpigmentation (PIH)
- increased risk for keloidal scars.2
Worth noting
- Patients with darker skin tones may be more concerned with the dark marks (also referred to as scars or manchas in Spanish) than the acne itself. This PIH may be viewed by patients as the major problem.
- Acne medications such as azelaic acid and some retinoids (when applied appropriately) can treat both acne and PIH.3
- Irritation from topical acne medications, including retinoid dermatitis, may lead to more PIH. Using noncomedogenic moisturizers and applying medication appropriately (ie, a pea-sized amount of topical retinoid per application) may help limit irritation.4,5
- One type of acne seen more commonly, although not exclusively, in Black patients is pomade acne, which principally appears on the forehead and is associated with use of hair care and styling products (Figure, C).
Health disparity highlight
Disparities in access to health care exist for those with dermatologic concerns. According to one study, African American (28.5%) and Hispanic patients (23.9%) were less likely to be seen by a dermatologist solely for the diagnosis of a dermatologic condition compared to Asian and Pacific Islander patients (36.7%) or White patients (43.2%).1
Noting that isotretinoin is the most potent systemic therapy for severe cystic acne vulgaris, Bell et al6 reported that Black patients had lower odds of receiving isotretinoin compared to White patients. Hispanic patients had lower odds of receiving a topical retinoid, tretinoin, than non-Hispanic patients.6
- Davis SA, Narahari S, Feldman SR, et al. Top dermatologic conditions in patients of color: an analysis of nationally representative data. J Drugs Dermatol. 2012;11:466-473.
- Alexis AF, Woolery-Lloyd H, Williams K, et al. Racial/ethnic variations in acne: implications for treatment and skin care recommendations for acne patients with skin of color. J Drugs Dermatol. 2021;20:716-725.
- Woolery-Lloyd HC, Keri J, Doig S. Retinoids and azelaic acid to treat acne and hyperpigmentation in skin of color. J Drugs Dermatol. 2013;12:434-437.
- Grayson C, Heath C. Tips for addressing common conditions affecting pediatric and adolescent patients with skin of color [published online March 2, 2021]. Pediatr Dermatol. doi:10.1111/pde.14525
- Alexis AD, Harper JC, Stein Gold L, et al. Treating acne in patients with skin of color. Semin Cutan Med Surg. 2018;37(suppl 3):S71-S73.
- Bell MA, Whang KA, Thomas J, et al. Racial and ethnic disparities in access to emerging and frontline therapies in common dermatological conditions: a cross-sectional study. J Natl Med Assoc. 2020;112:650-653.

THE COMPARISON
A A 27-year-old Hispanic woman with comedonal and inflammatory acne. Erythema is prominent around the inflammatory lesions. Note the pustule on the cheek surrounded by pink color.
B A teenaged Black boy with acne papules and pustules on the face. There are comedones, hyperpigmented macules, and pustules on the cheek.
C A teenaged Black girl with pomade acne. The patient used various hair care products, which obstructed the pilosebaceous units on the forehead.
Epidemiology
Acne is a leading dermatologic condition in individuals with skin of color in the United States.1
Key clinical features in people with darker skin tones include:
- erythematous or hyperpigmented papules or comedones
- hyperpigmented macules and postinflammatory hyperpigmentation (PIH)
- increased risk for keloidal scars.2
Worth noting
- Patients with darker skin tones may be more concerned with the dark marks (also referred to as scars or manchas in Spanish) than the acne itself. This PIH may be viewed by patients as the major problem.
- Acne medications such as azelaic acid and some retinoids (when applied appropriately) can treat both acne and PIH.3
- Irritation from topical acne medications, including retinoid dermatitis, may lead to more PIH. Using noncomedogenic moisturizers and applying medication appropriately (ie, a pea-sized amount of topical retinoid per application) may help limit irritation.4,5
- One type of acne seen more commonly, although not exclusively, in Black patients is pomade acne, which principally appears on the forehead and is associated with use of hair care and styling products (Figure, C).
Health disparity highlight
Disparities in access to health care exist for those with dermatologic concerns. According to one study, African American (28.5%) and Hispanic patients (23.9%) were less likely to be seen by a dermatologist solely for the diagnosis of a dermatologic condition compared to Asian and Pacific Islander patients (36.7%) or White patients (43.2%).1
Noting that isotretinoin is the most potent systemic therapy for severe cystic acne vulgaris, Bell et al6 reported that Black patients had lower odds of receiving isotretinoin compared to White patients. Hispanic patients had lower odds of receiving a topical retinoid, tretinoin, than non-Hispanic patients.6

THE COMPARISON
A A 27-year-old Hispanic woman with comedonal and inflammatory acne. Erythema is prominent around the inflammatory lesions. Note the pustule on the cheek surrounded by pink color.
B A teenaged Black boy with acne papules and pustules on the face. There are comedones, hyperpigmented macules, and pustules on the cheek.
C A teenaged Black girl with pomade acne. The patient used various hair care products, which obstructed the pilosebaceous units on the forehead.
Epidemiology
Acne is a leading dermatologic condition in individuals with skin of color in the United States.1
Key clinical features in people with darker skin tones include:
- erythematous or hyperpigmented papules or comedones
- hyperpigmented macules and postinflammatory hyperpigmentation (PIH)
- increased risk for keloidal scars.2
Worth noting
- Patients with darker skin tones may be more concerned with the dark marks (also referred to as scars or manchas in Spanish) than the acne itself. This PIH may be viewed by patients as the major problem.
- Acne medications such as azelaic acid and some retinoids (when applied appropriately) can treat both acne and PIH.3
- Irritation from topical acne medications, including retinoid dermatitis, may lead to more PIH. Using noncomedogenic moisturizers and applying medication appropriately (ie, a pea-sized amount of topical retinoid per application) may help limit irritation.4,5
- One type of acne seen more commonly, although not exclusively, in Black patients is pomade acne, which principally appears on the forehead and is associated with use of hair care and styling products (Figure, C).
Health disparity highlight
Disparities in access to health care exist for those with dermatologic concerns. According to one study, African American (28.5%) and Hispanic patients (23.9%) were less likely to be seen by a dermatologist solely for the diagnosis of a dermatologic condition compared to Asian and Pacific Islander patients (36.7%) or White patients (43.2%).1
Noting that isotretinoin is the most potent systemic therapy for severe cystic acne vulgaris, Bell et al6 reported that Black patients had lower odds of receiving isotretinoin compared to White patients. Hispanic patients had lower odds of receiving a topical retinoid, tretinoin, than non-Hispanic patients.6
- Davis SA, Narahari S, Feldman SR, et al. Top dermatologic conditions in patients of color: an analysis of nationally representative data. J Drugs Dermatol. 2012;11:466-473.
- Alexis AF, Woolery-Lloyd H, Williams K, et al. Racial/ethnic variations in acne: implications for treatment and skin care recommendations for acne patients with skin of color. J Drugs Dermatol. 2021;20:716-725.
- Woolery-Lloyd HC, Keri J, Doig S. Retinoids and azelaic acid to treat acne and hyperpigmentation in skin of color. J Drugs Dermatol. 2013;12:434-437.
- Grayson C, Heath C. Tips for addressing common conditions affecting pediatric and adolescent patients with skin of color [published online March 2, 2021]. Pediatr Dermatol. doi:10.1111/pde.14525
- Alexis AD, Harper JC, Stein Gold L, et al. Treating acne in patients with skin of color. Semin Cutan Med Surg. 2018;37(suppl 3):S71-S73.
- Bell MA, Whang KA, Thomas J, et al. Racial and ethnic disparities in access to emerging and frontline therapies in common dermatological conditions: a cross-sectional study. J Natl Med Assoc. 2020;112:650-653.
- Davis SA, Narahari S, Feldman SR, et al. Top dermatologic conditions in patients of color: an analysis of nationally representative data. J Drugs Dermatol. 2012;11:466-473.
- Alexis AF, Woolery-Lloyd H, Williams K, et al. Racial/ethnic variations in acne: implications for treatment and skin care recommendations for acne patients with skin of color. J Drugs Dermatol. 2021;20:716-725.
- Woolery-Lloyd HC, Keri J, Doig S. Retinoids and azelaic acid to treat acne and hyperpigmentation in skin of color. J Drugs Dermatol. 2013;12:434-437.
- Grayson C, Heath C. Tips for addressing common conditions affecting pediatric and adolescent patients with skin of color [published online March 2, 2021]. Pediatr Dermatol. doi:10.1111/pde.14525
- Alexis AD, Harper JC, Stein Gold L, et al. Treating acne in patients with skin of color. Semin Cutan Med Surg. 2018;37(suppl 3):S71-S73.
- Bell MA, Whang KA, Thomas J, et al. Racial and ethnic disparities in access to emerging and frontline therapies in common dermatological conditions: a cross-sectional study. J Natl Med Assoc. 2020;112:650-653.
Vetiver: More than a pleasant aroma?
An important ingredient in the contemporary perfume and cosmetics industries, vetiver, is the only grass cultivated throughout the world to retain its essential oil, which contains sesquiterpene alcohols and hydrocarbons.1-3 Field and glasshouse studies have revealed that vetiver grass can tolerate extreme variations in climate well, including protracted drought, floods, submergence, temperature, and soils high in acidity, alkalinity, and various heavy metals. Its heartiness may explain its continuing or even increasing use in fragrances and other products pertinent to skin health as humanity strives to adapt to climate change.4 In a 2017 review of various commercial essential oils as antimicrobial therapy for cutaneous disorders, Orchard and van Vuuren identified vetiver as warranting particular attention for its capacity to confer broad benefits to the skin in addressing acne, cuts, eczema, oiliness, sores, wounds, and aging skin.5 The focus of this column will be the dermatologic potential of vetiver.
Chemical constituents
Vetiver is thought to be one of the most complex of the essential oils owing to the hundreds of sesquiterpene derivatives with large structural diversity that contribute to its composition. 3
In a 2012 analysis of the components of South Indian vetiver oils, Mallavarapu et al. found an abundance of sesquiterpenes and oxygenated sesquiterpenes with cedrane, bisabolane, eudesmane, eremophilane, and zizaane skeletons. The primary constituents identified in the four oils evaluated included eudesma-4,6-diene (delta-selinene) + beta-vetispirene (3.9%-6.1%), beta-vetivenene (0.9%-9.4%), 13-nor-trans-eudesma-4(15),7-dien-11-one + amorph-4-en-10-ol (5.0%-6.4%), trans-eudesma-4(15),7-dien-12-ol (vetiselinenol) + (E)-opposita-4(15),7(11)-dien-12-ol (3.7%-5.9%), eremophila-1 (10),11-dien-2alpha-ol (nootkatol) + ziza-6(13)-en-12-ol (khusimol) (16.1%-19.2%), and eremophila-1(10),7(11)-dien-2alpha-ol (isonootkatol) + (E)-eremophila-1(10),7(11)-12-ol (isovalencenol) (5.6%-6.9%).6
Antimicrobial activity
In 2012, Saikia et al. assessed the antimycobacterial activity of Vetiveria zizanioides against Mycobacterium tuberculosis H(37)Rv and H(37)Ra strains. Their results showed that ethanolic extracts and hexane fractions displayed robust antimycobacterial properties, buttressing the traditional medical uses of the plant, as well as consideration of this agent as a modern antituberculosis agent.7
Two years later, Dos Santos et al. showed that Vetiveria zizanioides roots grown in Brazil exhibited notable antimicrobial effects against various pathogenic organisms.8In 2017, Burger et al. showed that vetiver essential oil primarily contributes its scent to cosmetic formulations but also displayed antimicrobial activity against Gram-positive bacterial strains, as well as one strain of Candida glabrata. They suggest that vetiver should be considered for its antimicrobial capacity as an added bonus to cosmetic formulations.2
In a 2018 study to ascertain the antimicrobial activity of 247 essential oil combinations against five reference strains of wound pathogens, Orchard et al. found that 26 combinations exhibited extensive antimicrobial activity. Sandalwood and vetiver were found to contribute most to antimicrobial function when used in combination. The investigators concluded that such combinations warrant consideration for wound therapy.9
Antiacne activity
In 2018, Orchard et al. conducted another study of the efficacy of commercial essential oil combinations against the two pathogens responsible for acne, Propionibacterium acnes and Staphlyococcus epidermidis. They investigated 408 combinations, of which 167 exhibited notable antimicrobial activity. They observed that the combination with the lowest minimum inhibitory concentration value against P. acnes and S. epidermidis was vetiver and cinnamon bark.10 This usage points to the potential of vetiver use as an antiacne ingredient.
Safety
The Scientific Committee on Consumer Safety (SCCS) offered a final opinion on the safety of the fragrance ingredient acetylated vetiver oil in 2019, declaring its use with 1% alpha-tocopherol in cosmetic leave-on and rinse-off products safe at proposed concentration levels. They noted that acetylated vetiver oil has been used for several years without provoking contact allergies.11
Conclusion
Much more research is necessary to determine just what kind of a role this perfumery powerhouse can play in dermatology.
Dr. Baumann is a private practice dermatologist, researcher, author, and entrepreneur who practices in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann has written two textbooks and a New York Times Best Sellers book for consumers. Dr. Baumann has received funding for advisory boards and/or clinical research trials from Allergan, Galderma, Revance, Evolus, and Burt’s Bees. She is the CEO of Skin Type Solutions Inc., a company that independently tests skin care products and makes recommendations to physicians on which skin care technologies are best. Write to her at [email protected].
References
1. Del Giudice L et al. Environ Microbiol. 2008 Oct;10(10):2824-41.
2. Burger P et al. Medicines (Basel). 2017 Jun 16;4(2):41.
3. Belhassen E et al. Chem Biodivers. 2014 Nov;11(11):1821–42.
4. Danh LT et al. Int J Phytoremediation. 2009 Oct-Dec;11(8):664–91.
5. Orchard A and van Vuuren S. Evid Based Complement Alternat Med. 2017;2017:4517971.
6. Mallavarapu GR et al. Nat Prod Commun. 2012 Feb;7(2):223–5.
7. Saikia D et al. Complement Ther Med. 2012 Dec;20(6):434–6.
8. Dos Santos DS et al. Acta Pharm. 2014 Dec;64(4):495-501.
9. Orchard A et al. Chem Biodivers. 2018 Dec;15(12):e1800405.
10. Orchard A et al. Int J Cosmet Sci. 2018 Mar 24. [Epub ahead of print].
11. SCCS members & External experts. Regul Toxicol Pharmacol. 2019 Oct;107:104389.
An important ingredient in the contemporary perfume and cosmetics industries, vetiver, is the only grass cultivated throughout the world to retain its essential oil, which contains sesquiterpene alcohols and hydrocarbons.1-3 Field and glasshouse studies have revealed that vetiver grass can tolerate extreme variations in climate well, including protracted drought, floods, submergence, temperature, and soils high in acidity, alkalinity, and various heavy metals. Its heartiness may explain its continuing or even increasing use in fragrances and other products pertinent to skin health as humanity strives to adapt to climate change.4 In a 2017 review of various commercial essential oils as antimicrobial therapy for cutaneous disorders, Orchard and van Vuuren identified vetiver as warranting particular attention for its capacity to confer broad benefits to the skin in addressing acne, cuts, eczema, oiliness, sores, wounds, and aging skin.5 The focus of this column will be the dermatologic potential of vetiver.
Chemical constituents
Vetiver is thought to be one of the most complex of the essential oils owing to the hundreds of sesquiterpene derivatives with large structural diversity that contribute to its composition. 3
In a 2012 analysis of the components of South Indian vetiver oils, Mallavarapu et al. found an abundance of sesquiterpenes and oxygenated sesquiterpenes with cedrane, bisabolane, eudesmane, eremophilane, and zizaane skeletons. The primary constituents identified in the four oils evaluated included eudesma-4,6-diene (delta-selinene) + beta-vetispirene (3.9%-6.1%), beta-vetivenene (0.9%-9.4%), 13-nor-trans-eudesma-4(15),7-dien-11-one + amorph-4-en-10-ol (5.0%-6.4%), trans-eudesma-4(15),7-dien-12-ol (vetiselinenol) + (E)-opposita-4(15),7(11)-dien-12-ol (3.7%-5.9%), eremophila-1 (10),11-dien-2alpha-ol (nootkatol) + ziza-6(13)-en-12-ol (khusimol) (16.1%-19.2%), and eremophila-1(10),7(11)-dien-2alpha-ol (isonootkatol) + (E)-eremophila-1(10),7(11)-12-ol (isovalencenol) (5.6%-6.9%).6
Antimicrobial activity
In 2012, Saikia et al. assessed the antimycobacterial activity of Vetiveria zizanioides against Mycobacterium tuberculosis H(37)Rv and H(37)Ra strains. Their results showed that ethanolic extracts and hexane fractions displayed robust antimycobacterial properties, buttressing the traditional medical uses of the plant, as well as consideration of this agent as a modern antituberculosis agent.7
Two years later, Dos Santos et al. showed that Vetiveria zizanioides roots grown in Brazil exhibited notable antimicrobial effects against various pathogenic organisms.8In 2017, Burger et al. showed that vetiver essential oil primarily contributes its scent to cosmetic formulations but also displayed antimicrobial activity against Gram-positive bacterial strains, as well as one strain of Candida glabrata. They suggest that vetiver should be considered for its antimicrobial capacity as an added bonus to cosmetic formulations.2
In a 2018 study to ascertain the antimicrobial activity of 247 essential oil combinations against five reference strains of wound pathogens, Orchard et al. found that 26 combinations exhibited extensive antimicrobial activity. Sandalwood and vetiver were found to contribute most to antimicrobial function when used in combination. The investigators concluded that such combinations warrant consideration for wound therapy.9
Antiacne activity
In 2018, Orchard et al. conducted another study of the efficacy of commercial essential oil combinations against the two pathogens responsible for acne, Propionibacterium acnes and Staphlyococcus epidermidis. They investigated 408 combinations, of which 167 exhibited notable antimicrobial activity. They observed that the combination with the lowest minimum inhibitory concentration value against P. acnes and S. epidermidis was vetiver and cinnamon bark.10 This usage points to the potential of vetiver use as an antiacne ingredient.
Safety
The Scientific Committee on Consumer Safety (SCCS) offered a final opinion on the safety of the fragrance ingredient acetylated vetiver oil in 2019, declaring its use with 1% alpha-tocopherol in cosmetic leave-on and rinse-off products safe at proposed concentration levels. They noted that acetylated vetiver oil has been used for several years without provoking contact allergies.11
Conclusion
Much more research is necessary to determine just what kind of a role this perfumery powerhouse can play in dermatology.
Dr. Baumann is a private practice dermatologist, researcher, author, and entrepreneur who practices in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann has written two textbooks and a New York Times Best Sellers book for consumers. Dr. Baumann has received funding for advisory boards and/or clinical research trials from Allergan, Galderma, Revance, Evolus, and Burt’s Bees. She is the CEO of Skin Type Solutions Inc., a company that independently tests skin care products and makes recommendations to physicians on which skin care technologies are best. Write to her at [email protected].
References
1. Del Giudice L et al. Environ Microbiol. 2008 Oct;10(10):2824-41.
2. Burger P et al. Medicines (Basel). 2017 Jun 16;4(2):41.
3. Belhassen E et al. Chem Biodivers. 2014 Nov;11(11):1821–42.
4. Danh LT et al. Int J Phytoremediation. 2009 Oct-Dec;11(8):664–91.
5. Orchard A and van Vuuren S. Evid Based Complement Alternat Med. 2017;2017:4517971.
6. Mallavarapu GR et al. Nat Prod Commun. 2012 Feb;7(2):223–5.
7. Saikia D et al. Complement Ther Med. 2012 Dec;20(6):434–6.
8. Dos Santos DS et al. Acta Pharm. 2014 Dec;64(4):495-501.
9. Orchard A et al. Chem Biodivers. 2018 Dec;15(12):e1800405.
10. Orchard A et al. Int J Cosmet Sci. 2018 Mar 24. [Epub ahead of print].
11. SCCS members & External experts. Regul Toxicol Pharmacol. 2019 Oct;107:104389.
An important ingredient in the contemporary perfume and cosmetics industries, vetiver, is the only grass cultivated throughout the world to retain its essential oil, which contains sesquiterpene alcohols and hydrocarbons.1-3 Field and glasshouse studies have revealed that vetiver grass can tolerate extreme variations in climate well, including protracted drought, floods, submergence, temperature, and soils high in acidity, alkalinity, and various heavy metals. Its heartiness may explain its continuing or even increasing use in fragrances and other products pertinent to skin health as humanity strives to adapt to climate change.4 In a 2017 review of various commercial essential oils as antimicrobial therapy for cutaneous disorders, Orchard and van Vuuren identified vetiver as warranting particular attention for its capacity to confer broad benefits to the skin in addressing acne, cuts, eczema, oiliness, sores, wounds, and aging skin.5 The focus of this column will be the dermatologic potential of vetiver.
Chemical constituents
Vetiver is thought to be one of the most complex of the essential oils owing to the hundreds of sesquiterpene derivatives with large structural diversity that contribute to its composition. 3
In a 2012 analysis of the components of South Indian vetiver oils, Mallavarapu et al. found an abundance of sesquiterpenes and oxygenated sesquiterpenes with cedrane, bisabolane, eudesmane, eremophilane, and zizaane skeletons. The primary constituents identified in the four oils evaluated included eudesma-4,6-diene (delta-selinene) + beta-vetispirene (3.9%-6.1%), beta-vetivenene (0.9%-9.4%), 13-nor-trans-eudesma-4(15),7-dien-11-one + amorph-4-en-10-ol (5.0%-6.4%), trans-eudesma-4(15),7-dien-12-ol (vetiselinenol) + (E)-opposita-4(15),7(11)-dien-12-ol (3.7%-5.9%), eremophila-1 (10),11-dien-2alpha-ol (nootkatol) + ziza-6(13)-en-12-ol (khusimol) (16.1%-19.2%), and eremophila-1(10),7(11)-dien-2alpha-ol (isonootkatol) + (E)-eremophila-1(10),7(11)-12-ol (isovalencenol) (5.6%-6.9%).6
Antimicrobial activity
In 2012, Saikia et al. assessed the antimycobacterial activity of Vetiveria zizanioides against Mycobacterium tuberculosis H(37)Rv and H(37)Ra strains. Their results showed that ethanolic extracts and hexane fractions displayed robust antimycobacterial properties, buttressing the traditional medical uses of the plant, as well as consideration of this agent as a modern antituberculosis agent.7
Two years later, Dos Santos et al. showed that Vetiveria zizanioides roots grown in Brazil exhibited notable antimicrobial effects against various pathogenic organisms.8In 2017, Burger et al. showed that vetiver essential oil primarily contributes its scent to cosmetic formulations but also displayed antimicrobial activity against Gram-positive bacterial strains, as well as one strain of Candida glabrata. They suggest that vetiver should be considered for its antimicrobial capacity as an added bonus to cosmetic formulations.2
In a 2018 study to ascertain the antimicrobial activity of 247 essential oil combinations against five reference strains of wound pathogens, Orchard et al. found that 26 combinations exhibited extensive antimicrobial activity. Sandalwood and vetiver were found to contribute most to antimicrobial function when used in combination. The investigators concluded that such combinations warrant consideration for wound therapy.9
Antiacne activity
In 2018, Orchard et al. conducted another study of the efficacy of commercial essential oil combinations against the two pathogens responsible for acne, Propionibacterium acnes and Staphlyococcus epidermidis. They investigated 408 combinations, of which 167 exhibited notable antimicrobial activity. They observed that the combination with the lowest minimum inhibitory concentration value against P. acnes and S. epidermidis was vetiver and cinnamon bark.10 This usage points to the potential of vetiver use as an antiacne ingredient.
Safety
The Scientific Committee on Consumer Safety (SCCS) offered a final opinion on the safety of the fragrance ingredient acetylated vetiver oil in 2019, declaring its use with 1% alpha-tocopherol in cosmetic leave-on and rinse-off products safe at proposed concentration levels. They noted that acetylated vetiver oil has been used for several years without provoking contact allergies.11
Conclusion
Much more research is necessary to determine just what kind of a role this perfumery powerhouse can play in dermatology.
Dr. Baumann is a private practice dermatologist, researcher, author, and entrepreneur who practices in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann has written two textbooks and a New York Times Best Sellers book for consumers. Dr. Baumann has received funding for advisory boards and/or clinical research trials from Allergan, Galderma, Revance, Evolus, and Burt’s Bees. She is the CEO of Skin Type Solutions Inc., a company that independently tests skin care products and makes recommendations to physicians on which skin care technologies are best. Write to her at [email protected].
References
1. Del Giudice L et al. Environ Microbiol. 2008 Oct;10(10):2824-41.
2. Burger P et al. Medicines (Basel). 2017 Jun 16;4(2):41.
3. Belhassen E et al. Chem Biodivers. 2014 Nov;11(11):1821–42.
4. Danh LT et al. Int J Phytoremediation. 2009 Oct-Dec;11(8):664–91.
5. Orchard A and van Vuuren S. Evid Based Complement Alternat Med. 2017;2017:4517971.
6. Mallavarapu GR et al. Nat Prod Commun. 2012 Feb;7(2):223–5.
7. Saikia D et al. Complement Ther Med. 2012 Dec;20(6):434–6.
8. Dos Santos DS et al. Acta Pharm. 2014 Dec;64(4):495-501.
9. Orchard A et al. Chem Biodivers. 2018 Dec;15(12):e1800405.
10. Orchard A et al. Int J Cosmet Sci. 2018 Mar 24. [Epub ahead of print].
11. SCCS members & External experts. Regul Toxicol Pharmacol. 2019 Oct;107:104389.
‘Empathy fatigue’ in clinicians rises with latest COVID-19 surge
Heidi Erickson, MD, is tired. As a pulmonary and critical care physician at Hennepin Healthcare in Minneapolis, she has been providing care for patients with COVID-19 since the start of the pandemic.
It was exhausting from the beginning, as she and her colleagues scrambled to understand how to deal with this new disease. But lately, she has noticed a different kind of exhaustion arising from the knowledge that with vaccines widely available, the latest surge was preventable.
Her intensive care unit is currently as full as it has ever been with COVID-19 patients, many of them young adults and most of them unvaccinated. After the recent death of one patient, an unvaccinated man with teenage children, she had to face his family’s questions about why ivermectin, an antiparasitic medication that was falsely promoted as a COVID-19 treatment, was not administered.
“I’m fatigued because I’m working more than ever, but more people don’t have to die,” Dr. Erickson said in an interview . “It’s been very hard physically, mentally, emotionally.”
Amid yet another surge in COVID-19 cases around the United States, clinicians are speaking out about their growing frustration with this preventable crisis.
Some are using the terms “empathy fatigue” and “compassion fatigue” – a sense that they are losing empathy for unvaccinated individuals who are fueling the pandemic.
Dr. Erickson says she is frustrated not by individual patients but by a system that has allowed disinformation to proliferate. Experts say these types of feelings fit into a widespread pattern of physician burnout that has taken a new turn at this stage of the pandemic.
Paradoxical choices
Empathy is a cornerstone of what clinicians do, and the ability to understand and share a patient’s feelings is an essential skill for providing effective care, says Kaz Nelson, MD, a psychiatrist at the University of Minnesota, Minneapolis.
Practitioners face paradoxical situations all the time, she notes. These include individuals who break bones and go skydiving again, people who have high cholesterol but continue to eat fried foods, and those with advanced lung cancer who continue to smoke.
To treat patients with compassion, practitioners learn to set aside judgment by acknowledging the complexity of human behavior. They may lament the addictive nature of nicotine and advertising that targets children, for example, while still listening and caring.
Empathy requires high-level brain function, but as stress levels rise, brain function that drives empathy tends to shut down. It’s a survival mechanism, Dr. Nelson says.
When health care workers feel overwhelmed, trapped, or threatened by patients demanding unproven treatments or by ICUs with more patients than ventilators, they may experience a fight-or-flight response that makes them defensive, frustrated, angry, or uncaring, notes Mona Masood, DO, a Philadelphia-area psychiatrist and founder of Physician Support Line, a free mental health hotline for doctors.
Some clinicians have taken to Twitter and other social media platforms to post about these types of experiences.
These feelings, which have been brewing for months, have been exacerbated by the complexity of the current situation. Clinicians see a disconnect between what is and what could be, Dr. Nelson notes.
“Prior to vaccines, there weren’t other options, and so we had toxic stress and we had fatigue, but we could still maintain little bits of empathy by saying, ‘You know, people didn’t choose to get infected, and we are in a pandemic.’ We could kind of hate the virus. Now with access to vaccines, that last connection to empathy is removed for many people,” she says.
Self-preservation vs. empathy
Compassion fatigue or empathy fatigue is just one reaction to feeling completely maxed out and overstressed, Dr. Nelson says. Anger at society, such as what Dr. Erickson experienced, is another response.
Practitioners may also feel as if they are just going through the motions of their job, or they might disassociate, ceasing to feel that their patients are human. Plenty of doctors and nurses have cried in their cars after shifts and have posted tearful videos on social media.
Early in the pandemic, Dr. Masood says, physicians who called the support hotline expressed sadness and grief. Now, she had her colleagues hear frustration and anger, along with guilt and shame for having feelings they believe they shouldn’t be having, especially toward patients. They may feel unprofessional or worse – unworthy of being physicians, she says.
One recent caller to the hotline was a long-time ICU physician who had been told so many times by patients that ivermectin was the only medicine that would cure them that he began to doubt himself, says Dr. Masood. This caller needed to be reassured by another physician that he was doing the right thing.
Another emergency department physician told Dr. Masood about a young child who had arrived at the hospital with COVID-19 symptoms. When asked whether the family had been exposed to anyone with COVID-19, the child’s parent lied so that they could be triaged faster.
The physician, who needed to step away from the situation, reached out to Dr. Masood to express her frustration so that she wouldn’t “let it out” on the patient.
“It’s hard to have empathy for people who, for all intents and purposes, are very self-centered,” Dr. Masood says. “We’re at a place where we’re having to choose between self-preservation and empathy.”
How to cope
To help practitioners cope, Dr. Masood offers words that describe what they’re experiencing. She often hears clinicians say things such as, “This is a type of burnout that I feel to my bones,” or “This makes me want to quit,” or “I feel like I’m at the end of my rope.”
She encourages them to consider the terms “empathy fatigue,” and “moral injury” in order to reconcile how their sense of responsibility to take care of people is compromised by factors outside of their control.
It is not shameful to acknowledge that they experience emotions, including difficult ones such as frustration, anger, sadness, and anxiety, Dr. Masood adds.
Being frustrated with a patient doesn’t make someone a bad doctor, and admitting those emotions is the first step toward dealing with them, she says.
before they cause a sense of callousness or other consequences that become harder to heal from as time goes on.
“We’re trained to just go, go, go and sometimes not pause and check in,” she says. Clinicians who open up are likely to find they are not the only ones feeling tired or frustrated right now, she adds.
“Connect with peers and colleagues, because chances are, they can relate,” Dr. Nelson says.
A version of this article first appeared on Medscape.com.
Heidi Erickson, MD, is tired. As a pulmonary and critical care physician at Hennepin Healthcare in Minneapolis, she has been providing care for patients with COVID-19 since the start of the pandemic.
It was exhausting from the beginning, as she and her colleagues scrambled to understand how to deal with this new disease. But lately, she has noticed a different kind of exhaustion arising from the knowledge that with vaccines widely available, the latest surge was preventable.
Her intensive care unit is currently as full as it has ever been with COVID-19 patients, many of them young adults and most of them unvaccinated. After the recent death of one patient, an unvaccinated man with teenage children, she had to face his family’s questions about why ivermectin, an antiparasitic medication that was falsely promoted as a COVID-19 treatment, was not administered.
“I’m fatigued because I’m working more than ever, but more people don’t have to die,” Dr. Erickson said in an interview . “It’s been very hard physically, mentally, emotionally.”
Amid yet another surge in COVID-19 cases around the United States, clinicians are speaking out about their growing frustration with this preventable crisis.
Some are using the terms “empathy fatigue” and “compassion fatigue” – a sense that they are losing empathy for unvaccinated individuals who are fueling the pandemic.
Dr. Erickson says she is frustrated not by individual patients but by a system that has allowed disinformation to proliferate. Experts say these types of feelings fit into a widespread pattern of physician burnout that has taken a new turn at this stage of the pandemic.
Paradoxical choices
Empathy is a cornerstone of what clinicians do, and the ability to understand and share a patient’s feelings is an essential skill for providing effective care, says Kaz Nelson, MD, a psychiatrist at the University of Minnesota, Minneapolis.
Practitioners face paradoxical situations all the time, she notes. These include individuals who break bones and go skydiving again, people who have high cholesterol but continue to eat fried foods, and those with advanced lung cancer who continue to smoke.
To treat patients with compassion, practitioners learn to set aside judgment by acknowledging the complexity of human behavior. They may lament the addictive nature of nicotine and advertising that targets children, for example, while still listening and caring.
Empathy requires high-level brain function, but as stress levels rise, brain function that drives empathy tends to shut down. It’s a survival mechanism, Dr. Nelson says.
When health care workers feel overwhelmed, trapped, or threatened by patients demanding unproven treatments or by ICUs with more patients than ventilators, they may experience a fight-or-flight response that makes them defensive, frustrated, angry, or uncaring, notes Mona Masood, DO, a Philadelphia-area psychiatrist and founder of Physician Support Line, a free mental health hotline for doctors.
Some clinicians have taken to Twitter and other social media platforms to post about these types of experiences.
These feelings, which have been brewing for months, have been exacerbated by the complexity of the current situation. Clinicians see a disconnect between what is and what could be, Dr. Nelson notes.
“Prior to vaccines, there weren’t other options, and so we had toxic stress and we had fatigue, but we could still maintain little bits of empathy by saying, ‘You know, people didn’t choose to get infected, and we are in a pandemic.’ We could kind of hate the virus. Now with access to vaccines, that last connection to empathy is removed for many people,” she says.
Self-preservation vs. empathy
Compassion fatigue or empathy fatigue is just one reaction to feeling completely maxed out and overstressed, Dr. Nelson says. Anger at society, such as what Dr. Erickson experienced, is another response.
Practitioners may also feel as if they are just going through the motions of their job, or they might disassociate, ceasing to feel that their patients are human. Plenty of doctors and nurses have cried in their cars after shifts and have posted tearful videos on social media.
Early in the pandemic, Dr. Masood says, physicians who called the support hotline expressed sadness and grief. Now, she had her colleagues hear frustration and anger, along with guilt and shame for having feelings they believe they shouldn’t be having, especially toward patients. They may feel unprofessional or worse – unworthy of being physicians, she says.
One recent caller to the hotline was a long-time ICU physician who had been told so many times by patients that ivermectin was the only medicine that would cure them that he began to doubt himself, says Dr. Masood. This caller needed to be reassured by another physician that he was doing the right thing.
Another emergency department physician told Dr. Masood about a young child who had arrived at the hospital with COVID-19 symptoms. When asked whether the family had been exposed to anyone with COVID-19, the child’s parent lied so that they could be triaged faster.
The physician, who needed to step away from the situation, reached out to Dr. Masood to express her frustration so that she wouldn’t “let it out” on the patient.
“It’s hard to have empathy for people who, for all intents and purposes, are very self-centered,” Dr. Masood says. “We’re at a place where we’re having to choose between self-preservation and empathy.”
How to cope
To help practitioners cope, Dr. Masood offers words that describe what they’re experiencing. She often hears clinicians say things such as, “This is a type of burnout that I feel to my bones,” or “This makes me want to quit,” or “I feel like I’m at the end of my rope.”
She encourages them to consider the terms “empathy fatigue,” and “moral injury” in order to reconcile how their sense of responsibility to take care of people is compromised by factors outside of their control.
It is not shameful to acknowledge that they experience emotions, including difficult ones such as frustration, anger, sadness, and anxiety, Dr. Masood adds.
Being frustrated with a patient doesn’t make someone a bad doctor, and admitting those emotions is the first step toward dealing with them, she says.
before they cause a sense of callousness or other consequences that become harder to heal from as time goes on.
“We’re trained to just go, go, go and sometimes not pause and check in,” she says. Clinicians who open up are likely to find they are not the only ones feeling tired or frustrated right now, she adds.
“Connect with peers and colleagues, because chances are, they can relate,” Dr. Nelson says.
A version of this article first appeared on Medscape.com.
Heidi Erickson, MD, is tired. As a pulmonary and critical care physician at Hennepin Healthcare in Minneapolis, she has been providing care for patients with COVID-19 since the start of the pandemic.
It was exhausting from the beginning, as she and her colleagues scrambled to understand how to deal with this new disease. But lately, she has noticed a different kind of exhaustion arising from the knowledge that with vaccines widely available, the latest surge was preventable.
Her intensive care unit is currently as full as it has ever been with COVID-19 patients, many of them young adults and most of them unvaccinated. After the recent death of one patient, an unvaccinated man with teenage children, she had to face his family’s questions about why ivermectin, an antiparasitic medication that was falsely promoted as a COVID-19 treatment, was not administered.
“I’m fatigued because I’m working more than ever, but more people don’t have to die,” Dr. Erickson said in an interview . “It’s been very hard physically, mentally, emotionally.”
Amid yet another surge in COVID-19 cases around the United States, clinicians are speaking out about their growing frustration with this preventable crisis.
Some are using the terms “empathy fatigue” and “compassion fatigue” – a sense that they are losing empathy for unvaccinated individuals who are fueling the pandemic.
Dr. Erickson says she is frustrated not by individual patients but by a system that has allowed disinformation to proliferate. Experts say these types of feelings fit into a widespread pattern of physician burnout that has taken a new turn at this stage of the pandemic.
Paradoxical choices
Empathy is a cornerstone of what clinicians do, and the ability to understand and share a patient’s feelings is an essential skill for providing effective care, says Kaz Nelson, MD, a psychiatrist at the University of Minnesota, Minneapolis.
Practitioners face paradoxical situations all the time, she notes. These include individuals who break bones and go skydiving again, people who have high cholesterol but continue to eat fried foods, and those with advanced lung cancer who continue to smoke.
To treat patients with compassion, practitioners learn to set aside judgment by acknowledging the complexity of human behavior. They may lament the addictive nature of nicotine and advertising that targets children, for example, while still listening and caring.
Empathy requires high-level brain function, but as stress levels rise, brain function that drives empathy tends to shut down. It’s a survival mechanism, Dr. Nelson says.
When health care workers feel overwhelmed, trapped, or threatened by patients demanding unproven treatments or by ICUs with more patients than ventilators, they may experience a fight-or-flight response that makes them defensive, frustrated, angry, or uncaring, notes Mona Masood, DO, a Philadelphia-area psychiatrist and founder of Physician Support Line, a free mental health hotline for doctors.
Some clinicians have taken to Twitter and other social media platforms to post about these types of experiences.
These feelings, which have been brewing for months, have been exacerbated by the complexity of the current situation. Clinicians see a disconnect between what is and what could be, Dr. Nelson notes.
“Prior to vaccines, there weren’t other options, and so we had toxic stress and we had fatigue, but we could still maintain little bits of empathy by saying, ‘You know, people didn’t choose to get infected, and we are in a pandemic.’ We could kind of hate the virus. Now with access to vaccines, that last connection to empathy is removed for many people,” she says.
Self-preservation vs. empathy
Compassion fatigue or empathy fatigue is just one reaction to feeling completely maxed out and overstressed, Dr. Nelson says. Anger at society, such as what Dr. Erickson experienced, is another response.
Practitioners may also feel as if they are just going through the motions of their job, or they might disassociate, ceasing to feel that their patients are human. Plenty of doctors and nurses have cried in their cars after shifts and have posted tearful videos on social media.
Early in the pandemic, Dr. Masood says, physicians who called the support hotline expressed sadness and grief. Now, she had her colleagues hear frustration and anger, along with guilt and shame for having feelings they believe they shouldn’t be having, especially toward patients. They may feel unprofessional or worse – unworthy of being physicians, she says.
One recent caller to the hotline was a long-time ICU physician who had been told so many times by patients that ivermectin was the only medicine that would cure them that he began to doubt himself, says Dr. Masood. This caller needed to be reassured by another physician that he was doing the right thing.
Another emergency department physician told Dr. Masood about a young child who had arrived at the hospital with COVID-19 symptoms. When asked whether the family had been exposed to anyone with COVID-19, the child’s parent lied so that they could be triaged faster.
The physician, who needed to step away from the situation, reached out to Dr. Masood to express her frustration so that she wouldn’t “let it out” on the patient.
“It’s hard to have empathy for people who, for all intents and purposes, are very self-centered,” Dr. Masood says. “We’re at a place where we’re having to choose between self-preservation and empathy.”
How to cope
To help practitioners cope, Dr. Masood offers words that describe what they’re experiencing. She often hears clinicians say things such as, “This is a type of burnout that I feel to my bones,” or “This makes me want to quit,” or “I feel like I’m at the end of my rope.”
She encourages them to consider the terms “empathy fatigue,” and “moral injury” in order to reconcile how their sense of responsibility to take care of people is compromised by factors outside of their control.
It is not shameful to acknowledge that they experience emotions, including difficult ones such as frustration, anger, sadness, and anxiety, Dr. Masood adds.
Being frustrated with a patient doesn’t make someone a bad doctor, and admitting those emotions is the first step toward dealing with them, she says.
before they cause a sense of callousness or other consequences that become harder to heal from as time goes on.
“We’re trained to just go, go, go and sometimes not pause and check in,” she says. Clinicians who open up are likely to find they are not the only ones feeling tired or frustrated right now, she adds.
“Connect with peers and colleagues, because chances are, they can relate,” Dr. Nelson says.
A version of this article first appeared on Medscape.com.
Finding employees during a pandemic
.
My own office is prime example: I have had job listings for both front- and back-office positions posted on all the major job boards and other employment portals for months, with a disappointing response. Of the few who do respond, many, incredibly, do not show up for their interviews!
It turns out that this is a widespread problem, and not just in medicine. A recent survey by the National Federation of Independent Business found that 42% of business owners, in all walks of life, had job openings that could not be filled, a record high. Over 90% of those hiring reported few or no qualified applicants and an increase in interview no-shows.
Clearly, this is a huge obstacle to growth – and even to conducting normal operations – for my practice and many others.
Reasons for the situation vary, but a big one has been the unfortunate fact that many open job positions actually pay less than the expanded unemployment benefits that many people have received under the March 2020 CARES Act. By one estimate, almost 70% of unemployed workers have been collecting more on unemployment than they earned while working. The CARES benefits expired in early September, but many potential workers continue to receive payments through a newer FEMA program, and some states have their own ongoing benefit programs.
Other reasons have been offered: Some candidates are unvaccinated (an immediate deal-breaker in my office), and some working parents continue to face a lack of childcare or in-person schooling for their children. Some applicants – regardless of vaccination status – have said they are hesitant to work in a medical office setting and risk getting COVID-19, despite all the precautions we have in place. Others have said they are waiting until the job market improves.
There are no easy solutions to this complicated problem, but here are a few suggestions culled from my research and conversations with HR professionals and others.
One obvious option is to offer higher wages, and perhaps even signing bonuses. “Whenever anyone says they can’t find the workers they need,” a consultant told me, “they are really saying they can’t find them at the wages they want to pay.” There are limits to the wages and benefits a private office with a very finite salary budget can offer, of course – but a few higher-paid employees may be preferable to no new workers at all.
For job candidates who fear COVID-19 exposure, assure them that their health and safety is a priority by spelling out the procedures your office is following (social distancing, reduced patient capacity, interaction barriers, face masks, avoidance of handshakes, enhanced cleaning procedures, symptom questionnaires, temperature checks, etc.) to minimize the risk of exposure.
You also may need to rework your interview process. In the Zoom era, most preliminary interviews can be conducted remotely. For on-site interviews, explain how you’re maintaining a safe interview environment by applying the same office safety policies to interactions with interviewees.
If a promising candidate doesn’t show up for an interview, the applicant could be making a token effort to obtain a job in order to perpetuate unemployment payments, but don’t jump to that conclusion. There may be extenuating circumstances, such as an emergency, illness, or traffic issues. Also, consider the possibility that it was your fault. If you waited too long to schedule the interview, another office could have lured them away. Or you may not have adequately explained your COVID-19 exposure safeguards. At the very least, a drawn-out process or a lack of transparency can make applicants apprehensive about accepting a job with you, particularly if other employers are pursuing them.
To counter the shortsighted appeal of collecting unemployment benefits, it may help to highlight the long-term growth opportunities available at your office. Consider outlining typical career tracks, or providing specific examples of how people have advanced their careers at your facility. I frequently cite the example of my current office manager, who began as an assistant receptionist almost 30 years ago.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News. Write to him at [email protected].
.
My own office is prime example: I have had job listings for both front- and back-office positions posted on all the major job boards and other employment portals for months, with a disappointing response. Of the few who do respond, many, incredibly, do not show up for their interviews!
It turns out that this is a widespread problem, and not just in medicine. A recent survey by the National Federation of Independent Business found that 42% of business owners, in all walks of life, had job openings that could not be filled, a record high. Over 90% of those hiring reported few or no qualified applicants and an increase in interview no-shows.
Clearly, this is a huge obstacle to growth – and even to conducting normal operations – for my practice and many others.
Reasons for the situation vary, but a big one has been the unfortunate fact that many open job positions actually pay less than the expanded unemployment benefits that many people have received under the March 2020 CARES Act. By one estimate, almost 70% of unemployed workers have been collecting more on unemployment than they earned while working. The CARES benefits expired in early September, but many potential workers continue to receive payments through a newer FEMA program, and some states have their own ongoing benefit programs.
Other reasons have been offered: Some candidates are unvaccinated (an immediate deal-breaker in my office), and some working parents continue to face a lack of childcare or in-person schooling for their children. Some applicants – regardless of vaccination status – have said they are hesitant to work in a medical office setting and risk getting COVID-19, despite all the precautions we have in place. Others have said they are waiting until the job market improves.
There are no easy solutions to this complicated problem, but here are a few suggestions culled from my research and conversations with HR professionals and others.
One obvious option is to offer higher wages, and perhaps even signing bonuses. “Whenever anyone says they can’t find the workers they need,” a consultant told me, “they are really saying they can’t find them at the wages they want to pay.” There are limits to the wages and benefits a private office with a very finite salary budget can offer, of course – but a few higher-paid employees may be preferable to no new workers at all.
For job candidates who fear COVID-19 exposure, assure them that their health and safety is a priority by spelling out the procedures your office is following (social distancing, reduced patient capacity, interaction barriers, face masks, avoidance of handshakes, enhanced cleaning procedures, symptom questionnaires, temperature checks, etc.) to minimize the risk of exposure.
You also may need to rework your interview process. In the Zoom era, most preliminary interviews can be conducted remotely. For on-site interviews, explain how you’re maintaining a safe interview environment by applying the same office safety policies to interactions with interviewees.
If a promising candidate doesn’t show up for an interview, the applicant could be making a token effort to obtain a job in order to perpetuate unemployment payments, but don’t jump to that conclusion. There may be extenuating circumstances, such as an emergency, illness, or traffic issues. Also, consider the possibility that it was your fault. If you waited too long to schedule the interview, another office could have lured them away. Or you may not have adequately explained your COVID-19 exposure safeguards. At the very least, a drawn-out process or a lack of transparency can make applicants apprehensive about accepting a job with you, particularly if other employers are pursuing them.
To counter the shortsighted appeal of collecting unemployment benefits, it may help to highlight the long-term growth opportunities available at your office. Consider outlining typical career tracks, or providing specific examples of how people have advanced their careers at your facility. I frequently cite the example of my current office manager, who began as an assistant receptionist almost 30 years ago.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News. Write to him at [email protected].
.
My own office is prime example: I have had job listings for both front- and back-office positions posted on all the major job boards and other employment portals for months, with a disappointing response. Of the few who do respond, many, incredibly, do not show up for their interviews!
It turns out that this is a widespread problem, and not just in medicine. A recent survey by the National Federation of Independent Business found that 42% of business owners, in all walks of life, had job openings that could not be filled, a record high. Over 90% of those hiring reported few or no qualified applicants and an increase in interview no-shows.
Clearly, this is a huge obstacle to growth – and even to conducting normal operations – for my practice and many others.
Reasons for the situation vary, but a big one has been the unfortunate fact that many open job positions actually pay less than the expanded unemployment benefits that many people have received under the March 2020 CARES Act. By one estimate, almost 70% of unemployed workers have been collecting more on unemployment than they earned while working. The CARES benefits expired in early September, but many potential workers continue to receive payments through a newer FEMA program, and some states have their own ongoing benefit programs.
Other reasons have been offered: Some candidates are unvaccinated (an immediate deal-breaker in my office), and some working parents continue to face a lack of childcare or in-person schooling for their children. Some applicants – regardless of vaccination status – have said they are hesitant to work in a medical office setting and risk getting COVID-19, despite all the precautions we have in place. Others have said they are waiting until the job market improves.
There are no easy solutions to this complicated problem, but here are a few suggestions culled from my research and conversations with HR professionals and others.
One obvious option is to offer higher wages, and perhaps even signing bonuses. “Whenever anyone says they can’t find the workers they need,” a consultant told me, “they are really saying they can’t find them at the wages they want to pay.” There are limits to the wages and benefits a private office with a very finite salary budget can offer, of course – but a few higher-paid employees may be preferable to no new workers at all.
For job candidates who fear COVID-19 exposure, assure them that their health and safety is a priority by spelling out the procedures your office is following (social distancing, reduced patient capacity, interaction barriers, face masks, avoidance of handshakes, enhanced cleaning procedures, symptom questionnaires, temperature checks, etc.) to minimize the risk of exposure.
You also may need to rework your interview process. In the Zoom era, most preliminary interviews can be conducted remotely. For on-site interviews, explain how you’re maintaining a safe interview environment by applying the same office safety policies to interactions with interviewees.
If a promising candidate doesn’t show up for an interview, the applicant could be making a token effort to obtain a job in order to perpetuate unemployment payments, but don’t jump to that conclusion. There may be extenuating circumstances, such as an emergency, illness, or traffic issues. Also, consider the possibility that it was your fault. If you waited too long to schedule the interview, another office could have lured them away. Or you may not have adequately explained your COVID-19 exposure safeguards. At the very least, a drawn-out process or a lack of transparency can make applicants apprehensive about accepting a job with you, particularly if other employers are pursuing them.
To counter the shortsighted appeal of collecting unemployment benefits, it may help to highlight the long-term growth opportunities available at your office. Consider outlining typical career tracks, or providing specific examples of how people have advanced their careers at your facility. I frequently cite the example of my current office manager, who began as an assistant receptionist almost 30 years ago.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News. Write to him at [email protected].