User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
New Infant RSV Antibody Treatment Shows Strong Results
The new RSV antibody treatment for babies has been highly effective in its first season, according to a first look at data from four children’s hospitals.
Babies who received the new preventive treatment for RSV shortly after birth were 90% less likely to be severely sickened with the potentially deadly respiratory illness, according to the new estimate published by the Centers for Disease Control and Prevention. It is the first real-world evaluation of Beyfortus (the generic name is nirsevimab), which was approved by the Food and Drug Administration last July.
RSV is a seasonal illness that affects more people — particularly infants and the elderly — in the fall and winter. Symptoms are usually mild in healthy adults, but infants are particularly at risk of getting bronchiolitis, which results in exhausting wheezing and coughing in babies due to swelling in their airways and lungs. Babies who are hospitalized may need fluids and medical devices to help them breathe.
RSV peaked this season from November to January, with more than 10,000 monthly diagnoses reported to the CDC.
The new CDC analysis was conducted among about 700 babies hospitalized for severe respiratory problems from October to the end of February. Among the babies in the study, 407 were diagnosed with RSV and 292 tested negative. The researchers found that 1% of babies in the study who were diagnosed with RSV had received Beyfortus, while the remaining babies who were positive for the virus had not.
Among the babies hospitalized for other severe respiratory problems, 18% had received Beyfortus. Overall, just 59 babies among the nearly 700 in the study received Beyfortus, perhaps reflecting the short supply of the medicine the first season it was available. The report authors noted that babies in the study who did receive Beyfortus also tended to have high-risk medical conditions.
The number of babies nationwide who received Beyfortus during this first season of availability is unclear, but a January CDC survey showed that 4 in 10 parents said their babies under 8 months old had received the treatment. The Wall Street Journal reported recently that a shortage last fall resulted from underestimated demand and from production plans that were set before the CDC decided to recommend that all infants under 8 months old receive Beyfortus if their mothers did not get a maternal vaccine that can protect infants from RSV.
Both the antibody treatment for infants and the maternal vaccine were shown in clinical trials to be about 80% effective at preventing severe illness stemming from RSV.
The authors of the latest CDC report concluded that “this early estimate supports the current nirsevimab recommendation for the prevention of severe RSV disease in infants. Infants should be protected by maternal RSV vaccination or infant receipt of nirsevimab.”
A version of this article appeared on WebMD.com.
The new RSV antibody treatment for babies has been highly effective in its first season, according to a first look at data from four children’s hospitals.
Babies who received the new preventive treatment for RSV shortly after birth were 90% less likely to be severely sickened with the potentially deadly respiratory illness, according to the new estimate published by the Centers for Disease Control and Prevention. It is the first real-world evaluation of Beyfortus (the generic name is nirsevimab), which was approved by the Food and Drug Administration last July.
RSV is a seasonal illness that affects more people — particularly infants and the elderly — in the fall and winter. Symptoms are usually mild in healthy adults, but infants are particularly at risk of getting bronchiolitis, which results in exhausting wheezing and coughing in babies due to swelling in their airways and lungs. Babies who are hospitalized may need fluids and medical devices to help them breathe.
RSV peaked this season from November to January, with more than 10,000 monthly diagnoses reported to the CDC.
The new CDC analysis was conducted among about 700 babies hospitalized for severe respiratory problems from October to the end of February. Among the babies in the study, 407 were diagnosed with RSV and 292 tested negative. The researchers found that 1% of babies in the study who were diagnosed with RSV had received Beyfortus, while the remaining babies who were positive for the virus had not.
Among the babies hospitalized for other severe respiratory problems, 18% had received Beyfortus. Overall, just 59 babies among the nearly 700 in the study received Beyfortus, perhaps reflecting the short supply of the medicine the first season it was available. The report authors noted that babies in the study who did receive Beyfortus also tended to have high-risk medical conditions.
The number of babies nationwide who received Beyfortus during this first season of availability is unclear, but a January CDC survey showed that 4 in 10 parents said their babies under 8 months old had received the treatment. The Wall Street Journal reported recently that a shortage last fall resulted from underestimated demand and from production plans that were set before the CDC decided to recommend that all infants under 8 months old receive Beyfortus if their mothers did not get a maternal vaccine that can protect infants from RSV.
Both the antibody treatment for infants and the maternal vaccine were shown in clinical trials to be about 80% effective at preventing severe illness stemming from RSV.
The authors of the latest CDC report concluded that “this early estimate supports the current nirsevimab recommendation for the prevention of severe RSV disease in infants. Infants should be protected by maternal RSV vaccination or infant receipt of nirsevimab.”
A version of this article appeared on WebMD.com.
The new RSV antibody treatment for babies has been highly effective in its first season, according to a first look at data from four children’s hospitals.
Babies who received the new preventive treatment for RSV shortly after birth were 90% less likely to be severely sickened with the potentially deadly respiratory illness, according to the new estimate published by the Centers for Disease Control and Prevention. It is the first real-world evaluation of Beyfortus (the generic name is nirsevimab), which was approved by the Food and Drug Administration last July.
RSV is a seasonal illness that affects more people — particularly infants and the elderly — in the fall and winter. Symptoms are usually mild in healthy adults, but infants are particularly at risk of getting bronchiolitis, which results in exhausting wheezing and coughing in babies due to swelling in their airways and lungs. Babies who are hospitalized may need fluids and medical devices to help them breathe.
RSV peaked this season from November to January, with more than 10,000 monthly diagnoses reported to the CDC.
The new CDC analysis was conducted among about 700 babies hospitalized for severe respiratory problems from October to the end of February. Among the babies in the study, 407 were diagnosed with RSV and 292 tested negative. The researchers found that 1% of babies in the study who were diagnosed with RSV had received Beyfortus, while the remaining babies who were positive for the virus had not.
Among the babies hospitalized for other severe respiratory problems, 18% had received Beyfortus. Overall, just 59 babies among the nearly 700 in the study received Beyfortus, perhaps reflecting the short supply of the medicine the first season it was available. The report authors noted that babies in the study who did receive Beyfortus also tended to have high-risk medical conditions.
The number of babies nationwide who received Beyfortus during this first season of availability is unclear, but a January CDC survey showed that 4 in 10 parents said their babies under 8 months old had received the treatment. The Wall Street Journal reported recently that a shortage last fall resulted from underestimated demand and from production plans that were set before the CDC decided to recommend that all infants under 8 months old receive Beyfortus if their mothers did not get a maternal vaccine that can protect infants from RSV.
Both the antibody treatment for infants and the maternal vaccine were shown in clinical trials to be about 80% effective at preventing severe illness stemming from RSV.
The authors of the latest CDC report concluded that “this early estimate supports the current nirsevimab recommendation for the prevention of severe RSV disease in infants. Infants should be protected by maternal RSV vaccination or infant receipt of nirsevimab.”
A version of this article appeared on WebMD.com.
Medicare Doc Pay Cut Eased, but When Will Serious Revisions Come?
President Joe Biden on March 9 signed into law a measure that softened — but did not completely eliminate — a 2024 cut in a key rate used to determine how physicians are paid for treating Medicare patients.
While physician groups hailed the move as partial relief, they say they’ll continue to press for broader changes in the Medicare physician fee schedule.
The Medicare provision was tucked into a larger spending package approved by the US House and Senate.
The American Academy of Family Physicians (AAFP), the American Medical Association (AMA), and other groups have lobbied Congress for months to undo a 3.4% cut in the base rate, or conversion factor, in the physician fee schedule for 2024.
The conversion factor is used in calculations to determine reimbursement for myriad other services. Federal Medicare officials said the cut would mean a 1.25% decrease in overall payments in 2024, compared with 2023.
“With the passage of this legislation, Congress has offset 2.93% of that payment cut,” said Steven P. Furr, MD, AAFP’s president in a statement. “We appreciate this temporary measure but continue to urge Congress to advance comprehensive, long-term Medicare payment reform.”
In a statement, Representative Larry Bucshon, MD (R-IN), said the payment cut could not be completely eliminated because of budget constraints.
The Medicare physician fee schedule covers much of the care clinicians provide to people older than 65 and those with disabilities. It covers about 8000 different types of services, ranging from office visits to surgical procedures, imaging, and tests, according to the Medicare Payment Advisory Commission (MedPAC).
Along with physicians, the fee schedule sets payments for nurse practitioners, physician assistants, podiatrists, physical therapists, psychologists, and other clinicians.
In 2021, the Medicare program and its beneficiaries paid $92.8 billion for services provided by almost 1.3 million clinicians, MedPAC said.
Larger Changes Ahead?
Rep. Bucshon is among the physicians serving in the House who are pressing for a permanent revamp of the Medicare physician fee schedule. He cosponsored a bill (HR 2474) that would peg future annual increases in the physician fee schedule to the Medicare Economic Index, which would reflect inflation’s effect.
In April, more than 120 state and national medical groups signed onto an AMA-led letter urging Congress to pass this bill.
The measure is a key priority for the AMA. The organization reached out repeatedly last year to federal officials about it through its own in-house lobbyists, this news organization found through a review of congressional lobbying forms submitted by AMA.
These required disclosure forms reveal how much AMA and other organizations spend each quarter to appeal to members of Congress and federal agencies on specific issues. The disclosure forms do not include a detailed accounting of spending on each issue.
But they do show which issues are priorities for an organization. AMA’s in-house lobbyists reported raising dozens of issues in 2024 within contacts in Congress and federal agencies. These issues included abortion access, maternal health, physician burnout, and potential for bias in clinical use of algorithms, as well as Medicare payment for physicians.
AMA reported spending estimated cost of $20.6 million. (AMA spent $6.7 million in the first quarter, $4.75 million in the second quarter, $3.42 million in the third quarter, and $5.74 million in the fourth quarter.)
In a March 6 statement, Jesse M. Ehrenfeld, MD, MPH, AMA president, urged Congress to turn to more serious consideration of Medicare physician pay beyond short-term tweaks attached to other larger bills.
“As physicians, we are trained to run toward emergencies. We urge Congress to do the same,” Dr. Ehrenfeld said. “We encourage Congress to act if this policy decision is an emergency because — in fact — it is. It is well past time to put an end to stopgap measures that fail to address the underlying causes of the continuing decline in Medicare physician payments.”
There’s bipartisan interest in a revamp of the physician fee schedule amid widespread criticism of the last such overhaul, the Medicare Access and CHIP Reauthorization Act of 2015.
For example, Senate Budget Chairman Sheldon Whitehouse (D-RI) has proposed the creation of a technical advisory committee to improve how Medicare sets the physician fee schedule. The existing fee schedule provides too little money for primary care services and primary care provider pay, contributing to shortages, Sen. Whitehouse said.
Sen. Whitehouse on March 6 held a hearing on ways to beef up US primary care. Among the experts who appeared was Amol Navathe, MD, PhD, of the University of Pennsylvania, Philadelphia, Pennsylvania.
Dr. Navathe said the current Medicare physician fee schedule tilts in favor of procedural services, leading to “underinvestment in cognitive, diagnostic, and supportive services such as primary care.”
In addition, much of what primary care clinicians do, “such as addressing social challenges, is not included in the codes of the fee schedule itself,” said Dr. Navathe, who also serves as the vice chairman of MedPAC.
It’s unclear when Congress will attempt a serious revision to the Medicare physician fee schedule. Lawmakers are unlikely to take on such a major challenge in this election year.
There would be significant opposition and challenges for lawmakers in trying to clear a bill that added an inflation adjustment for what’s already seen as an imperfect physician fee schedule, said Mark E. Miller, PhD, executive vice president of healthcare at the philanthropy Arnold Ventures, which studies how payment decisions affect medical care.
“That bill could cost a lot of money and raise a lot of questions,” Dr. Miller said.
A version of this article appeared on Medscape.com.
President Joe Biden on March 9 signed into law a measure that softened — but did not completely eliminate — a 2024 cut in a key rate used to determine how physicians are paid for treating Medicare patients.
While physician groups hailed the move as partial relief, they say they’ll continue to press for broader changes in the Medicare physician fee schedule.
The Medicare provision was tucked into a larger spending package approved by the US House and Senate.
The American Academy of Family Physicians (AAFP), the American Medical Association (AMA), and other groups have lobbied Congress for months to undo a 3.4% cut in the base rate, or conversion factor, in the physician fee schedule for 2024.
The conversion factor is used in calculations to determine reimbursement for myriad other services. Federal Medicare officials said the cut would mean a 1.25% decrease in overall payments in 2024, compared with 2023.
“With the passage of this legislation, Congress has offset 2.93% of that payment cut,” said Steven P. Furr, MD, AAFP’s president in a statement. “We appreciate this temporary measure but continue to urge Congress to advance comprehensive, long-term Medicare payment reform.”
In a statement, Representative Larry Bucshon, MD (R-IN), said the payment cut could not be completely eliminated because of budget constraints.
The Medicare physician fee schedule covers much of the care clinicians provide to people older than 65 and those with disabilities. It covers about 8000 different types of services, ranging from office visits to surgical procedures, imaging, and tests, according to the Medicare Payment Advisory Commission (MedPAC).
Along with physicians, the fee schedule sets payments for nurse practitioners, physician assistants, podiatrists, physical therapists, psychologists, and other clinicians.
In 2021, the Medicare program and its beneficiaries paid $92.8 billion for services provided by almost 1.3 million clinicians, MedPAC said.
Larger Changes Ahead?
Rep. Bucshon is among the physicians serving in the House who are pressing for a permanent revamp of the Medicare physician fee schedule. He cosponsored a bill (HR 2474) that would peg future annual increases in the physician fee schedule to the Medicare Economic Index, which would reflect inflation’s effect.
In April, more than 120 state and national medical groups signed onto an AMA-led letter urging Congress to pass this bill.
The measure is a key priority for the AMA. The organization reached out repeatedly last year to federal officials about it through its own in-house lobbyists, this news organization found through a review of congressional lobbying forms submitted by AMA.
These required disclosure forms reveal how much AMA and other organizations spend each quarter to appeal to members of Congress and federal agencies on specific issues. The disclosure forms do not include a detailed accounting of spending on each issue.
But they do show which issues are priorities for an organization. AMA’s in-house lobbyists reported raising dozens of issues in 2024 within contacts in Congress and federal agencies. These issues included abortion access, maternal health, physician burnout, and potential for bias in clinical use of algorithms, as well as Medicare payment for physicians.
AMA reported spending estimated cost of $20.6 million. (AMA spent $6.7 million in the first quarter, $4.75 million in the second quarter, $3.42 million in the third quarter, and $5.74 million in the fourth quarter.)
In a March 6 statement, Jesse M. Ehrenfeld, MD, MPH, AMA president, urged Congress to turn to more serious consideration of Medicare physician pay beyond short-term tweaks attached to other larger bills.
“As physicians, we are trained to run toward emergencies. We urge Congress to do the same,” Dr. Ehrenfeld said. “We encourage Congress to act if this policy decision is an emergency because — in fact — it is. It is well past time to put an end to stopgap measures that fail to address the underlying causes of the continuing decline in Medicare physician payments.”
There’s bipartisan interest in a revamp of the physician fee schedule amid widespread criticism of the last such overhaul, the Medicare Access and CHIP Reauthorization Act of 2015.
For example, Senate Budget Chairman Sheldon Whitehouse (D-RI) has proposed the creation of a technical advisory committee to improve how Medicare sets the physician fee schedule. The existing fee schedule provides too little money for primary care services and primary care provider pay, contributing to shortages, Sen. Whitehouse said.
Sen. Whitehouse on March 6 held a hearing on ways to beef up US primary care. Among the experts who appeared was Amol Navathe, MD, PhD, of the University of Pennsylvania, Philadelphia, Pennsylvania.
Dr. Navathe said the current Medicare physician fee schedule tilts in favor of procedural services, leading to “underinvestment in cognitive, diagnostic, and supportive services such as primary care.”
In addition, much of what primary care clinicians do, “such as addressing social challenges, is not included in the codes of the fee schedule itself,” said Dr. Navathe, who also serves as the vice chairman of MedPAC.
It’s unclear when Congress will attempt a serious revision to the Medicare physician fee schedule. Lawmakers are unlikely to take on such a major challenge in this election year.
There would be significant opposition and challenges for lawmakers in trying to clear a bill that added an inflation adjustment for what’s already seen as an imperfect physician fee schedule, said Mark E. Miller, PhD, executive vice president of healthcare at the philanthropy Arnold Ventures, which studies how payment decisions affect medical care.
“That bill could cost a lot of money and raise a lot of questions,” Dr. Miller said.
A version of this article appeared on Medscape.com.
President Joe Biden on March 9 signed into law a measure that softened — but did not completely eliminate — a 2024 cut in a key rate used to determine how physicians are paid for treating Medicare patients.
While physician groups hailed the move as partial relief, they say they’ll continue to press for broader changes in the Medicare physician fee schedule.
The Medicare provision was tucked into a larger spending package approved by the US House and Senate.
The American Academy of Family Physicians (AAFP), the American Medical Association (AMA), and other groups have lobbied Congress for months to undo a 3.4% cut in the base rate, or conversion factor, in the physician fee schedule for 2024.
The conversion factor is used in calculations to determine reimbursement for myriad other services. Federal Medicare officials said the cut would mean a 1.25% decrease in overall payments in 2024, compared with 2023.
“With the passage of this legislation, Congress has offset 2.93% of that payment cut,” said Steven P. Furr, MD, AAFP’s president in a statement. “We appreciate this temporary measure but continue to urge Congress to advance comprehensive, long-term Medicare payment reform.”
In a statement, Representative Larry Bucshon, MD (R-IN), said the payment cut could not be completely eliminated because of budget constraints.
The Medicare physician fee schedule covers much of the care clinicians provide to people older than 65 and those with disabilities. It covers about 8000 different types of services, ranging from office visits to surgical procedures, imaging, and tests, according to the Medicare Payment Advisory Commission (MedPAC).
Along with physicians, the fee schedule sets payments for nurse practitioners, physician assistants, podiatrists, physical therapists, psychologists, and other clinicians.
In 2021, the Medicare program and its beneficiaries paid $92.8 billion for services provided by almost 1.3 million clinicians, MedPAC said.
Larger Changes Ahead?
Rep. Bucshon is among the physicians serving in the House who are pressing for a permanent revamp of the Medicare physician fee schedule. He cosponsored a bill (HR 2474) that would peg future annual increases in the physician fee schedule to the Medicare Economic Index, which would reflect inflation’s effect.
In April, more than 120 state and national medical groups signed onto an AMA-led letter urging Congress to pass this bill.
The measure is a key priority for the AMA. The organization reached out repeatedly last year to federal officials about it through its own in-house lobbyists, this news organization found through a review of congressional lobbying forms submitted by AMA.
These required disclosure forms reveal how much AMA and other organizations spend each quarter to appeal to members of Congress and federal agencies on specific issues. The disclosure forms do not include a detailed accounting of spending on each issue.
But they do show which issues are priorities for an organization. AMA’s in-house lobbyists reported raising dozens of issues in 2024 within contacts in Congress and federal agencies. These issues included abortion access, maternal health, physician burnout, and potential for bias in clinical use of algorithms, as well as Medicare payment for physicians.
AMA reported spending estimated cost of $20.6 million. (AMA spent $6.7 million in the first quarter, $4.75 million in the second quarter, $3.42 million in the third quarter, and $5.74 million in the fourth quarter.)
In a March 6 statement, Jesse M. Ehrenfeld, MD, MPH, AMA president, urged Congress to turn to more serious consideration of Medicare physician pay beyond short-term tweaks attached to other larger bills.
“As physicians, we are trained to run toward emergencies. We urge Congress to do the same,” Dr. Ehrenfeld said. “We encourage Congress to act if this policy decision is an emergency because — in fact — it is. It is well past time to put an end to stopgap measures that fail to address the underlying causes of the continuing decline in Medicare physician payments.”
There’s bipartisan interest in a revamp of the physician fee schedule amid widespread criticism of the last such overhaul, the Medicare Access and CHIP Reauthorization Act of 2015.
For example, Senate Budget Chairman Sheldon Whitehouse (D-RI) has proposed the creation of a technical advisory committee to improve how Medicare sets the physician fee schedule. The existing fee schedule provides too little money for primary care services and primary care provider pay, contributing to shortages, Sen. Whitehouse said.
Sen. Whitehouse on March 6 held a hearing on ways to beef up US primary care. Among the experts who appeared was Amol Navathe, MD, PhD, of the University of Pennsylvania, Philadelphia, Pennsylvania.
Dr. Navathe said the current Medicare physician fee schedule tilts in favor of procedural services, leading to “underinvestment in cognitive, diagnostic, and supportive services such as primary care.”
In addition, much of what primary care clinicians do, “such as addressing social challenges, is not included in the codes of the fee schedule itself,” said Dr. Navathe, who also serves as the vice chairman of MedPAC.
It’s unclear when Congress will attempt a serious revision to the Medicare physician fee schedule. Lawmakers are unlikely to take on such a major challenge in this election year.
There would be significant opposition and challenges for lawmakers in trying to clear a bill that added an inflation adjustment for what’s already seen as an imperfect physician fee schedule, said Mark E. Miller, PhD, executive vice president of healthcare at the philanthropy Arnold Ventures, which studies how payment decisions affect medical care.
“That bill could cost a lot of money and raise a lot of questions,” Dr. Miller said.
A version of this article appeared on Medscape.com.
Medicare Pay Bump Provision in Federal Bill Falls Short, Doc Groups Say
Lawmakers have added a provision to raise Medicare payments to clinicians to a $460 billion bipartisan package of federal spending bills that passed in the House on March 6 and is expected to be passed in the Senate and signed by President Biden before then end of March 8, but industry groups have criticized it as paltry.
Lawmakers often tweak Medicare policy by adding provisions to other kinds of legislation, including the spending bills Congress must pass to keep the federal government running.
Physicians’ groups and some lawmakers have long pressed Congress to change Medicare payment rules with little success, even as inflation has caused physicians’ expenses to rise. Doctors now face a 3.4% cut to Medicare reimbursements in 2024, which would be only partly mitigated by the recently announced provision.
The Medical Group Management Association (MGMA) said the proposed increase would total 1.68%. The increase, part of a bipartisan package of bills released by the House and Senate Appropriations committees on March 3, would apply to the budget for fiscal 2024, which began on October 1, 2023.
“We are deeply disappointed with Congress’ half-hearted attempt to remedy the devastating blow physician practices were dealt by the 2024 Medicare Physician Fee Schedule,” Anders Gilberg, senior vice president of MGMA, said in a statement. “Anything less than a full reversal of the 3.4% cut is appallingly inadequate.”
The American Medical Association said it was “extremely disappointed” that the boost only eased, but did not fully reverse, a deeper planned cut.
The American Academy of Family Physicians (AAFP) also expressed disappointment with the proposed increase.
“The AAFP has repeatedly told Congress that the 3.4% Medicare payment reduction that went into effect on January 1 is untenable for family physicians and threatens patients’ access to primary care,” the group said in a statement.
“While we appreciate the partial relief, family physicians continue to face an annual threat of payment cuts that are detrimental to practices and patients,” AAFP said.
A version of this article appeared on Medscape.com.
Lawmakers have added a provision to raise Medicare payments to clinicians to a $460 billion bipartisan package of federal spending bills that passed in the House on March 6 and is expected to be passed in the Senate and signed by President Biden before then end of March 8, but industry groups have criticized it as paltry.
Lawmakers often tweak Medicare policy by adding provisions to other kinds of legislation, including the spending bills Congress must pass to keep the federal government running.
Physicians’ groups and some lawmakers have long pressed Congress to change Medicare payment rules with little success, even as inflation has caused physicians’ expenses to rise. Doctors now face a 3.4% cut to Medicare reimbursements in 2024, which would be only partly mitigated by the recently announced provision.
The Medical Group Management Association (MGMA) said the proposed increase would total 1.68%. The increase, part of a bipartisan package of bills released by the House and Senate Appropriations committees on March 3, would apply to the budget for fiscal 2024, which began on October 1, 2023.
“We are deeply disappointed with Congress’ half-hearted attempt to remedy the devastating blow physician practices were dealt by the 2024 Medicare Physician Fee Schedule,” Anders Gilberg, senior vice president of MGMA, said in a statement. “Anything less than a full reversal of the 3.4% cut is appallingly inadequate.”
The American Medical Association said it was “extremely disappointed” that the boost only eased, but did not fully reverse, a deeper planned cut.
The American Academy of Family Physicians (AAFP) also expressed disappointment with the proposed increase.
“The AAFP has repeatedly told Congress that the 3.4% Medicare payment reduction that went into effect on January 1 is untenable for family physicians and threatens patients’ access to primary care,” the group said in a statement.
“While we appreciate the partial relief, family physicians continue to face an annual threat of payment cuts that are detrimental to practices and patients,” AAFP said.
A version of this article appeared on Medscape.com.
Lawmakers have added a provision to raise Medicare payments to clinicians to a $460 billion bipartisan package of federal spending bills that passed in the House on March 6 and is expected to be passed in the Senate and signed by President Biden before then end of March 8, but industry groups have criticized it as paltry.
Lawmakers often tweak Medicare policy by adding provisions to other kinds of legislation, including the spending bills Congress must pass to keep the federal government running.
Physicians’ groups and some lawmakers have long pressed Congress to change Medicare payment rules with little success, even as inflation has caused physicians’ expenses to rise. Doctors now face a 3.4% cut to Medicare reimbursements in 2024, which would be only partly mitigated by the recently announced provision.
The Medical Group Management Association (MGMA) said the proposed increase would total 1.68%. The increase, part of a bipartisan package of bills released by the House and Senate Appropriations committees on March 3, would apply to the budget for fiscal 2024, which began on October 1, 2023.
“We are deeply disappointed with Congress’ half-hearted attempt to remedy the devastating blow physician practices were dealt by the 2024 Medicare Physician Fee Schedule,” Anders Gilberg, senior vice president of MGMA, said in a statement. “Anything less than a full reversal of the 3.4% cut is appallingly inadequate.”
The American Medical Association said it was “extremely disappointed” that the boost only eased, but did not fully reverse, a deeper planned cut.
The American Academy of Family Physicians (AAFP) also expressed disappointment with the proposed increase.
“The AAFP has repeatedly told Congress that the 3.4% Medicare payment reduction that went into effect on January 1 is untenable for family physicians and threatens patients’ access to primary care,” the group said in a statement.
“While we appreciate the partial relief, family physicians continue to face an annual threat of payment cuts that are detrimental to practices and patients,” AAFP said.
A version of this article appeared on Medscape.com.
How These MDs Conquered Imposter Syndrome
Do I deserve to be here? Am I doing what I’m supposed to be doing? Is anyone going to tell me if I’m terrible?
Kerri Palamara McGrath, MD, remembered worrying over these questions as chief resident at Massachusetts General Hospital, Boston, Massachusetts, in 2009. Having graduated from New York Medical College, she felt out of step with her peers from Ivy League medical schools and considered herself lucky to be there. In order to measure up, she felt she had to work twice as hard as everybody else.
But as Dr. McGrath moved through residency and spoke with other trainees, she had a realization. Her constant fears, the nagging voice in her head saying she wasn’t good enough, these issues weren’t exclusive to her; they were pervasive.
Today, Dr. McGrath is the director of the Center for Physician Well-Being at Massachusetts General Hospital. The facility aims to address physician stress and equip doctors with the tools to navigate personal and professional issues. Dr. McGrath is also a physician coach, a growing nonclinical field, helping doctors identify their own stressors, values, and measures of success. This type of internal work, Dr. McGrath feels, can help alleviate imposter syndrome, that inner refrain saying: I’ll never be good enough.
What Is Imposter Syndrome?
While not a formal medical diagnosis, imposter syndrome has been defined as «an internal experience of intellectual phoniness.» It›s considered an inability to internalize success and a tendency to attribute gains to external factors — for example, being in the right place at the right time.
“Many people describe imposter phenomena in medicine as fearing that others are going to realize that they don’t belong somewhere or question why they’re there,” said Dr. McGrath.
It’s a “fear of being found out,” added Jessi Gold, MD, a psychiatrist who treats physicians. “In many ways, imposter syndrome shows up as a conflict between the outer self — the metaphorical mask you’re ‘putting on’ [in order] to achieve, and the inner self — how you feel like you’re not measuring up.”
Dr. McGrath said she experienced imposter syndrome before her medical career even began. She applied to 26 medical schools. Only one accepted her. “The whole time, I was like, ‘This is the only school you got into, so you’re obviously not good enough,’” she recalled. Later, having been chosen by a “coveted” institution like Mass General, “you assume that, at some point, someone will realize that the gig is up, that everybody’s better than you.”
Where Does Imposter Syndrome Come From?
Dr. McGrath felt that in medicine, high expectations are often coupled with low self-compassion. “We are so hard on ourselves, and when we set our expectations so high, we’re constantly disappointed in ourselves,” she said. External markers of success — papers published, promotions, or even social media — can further fuel this.
It can feel like “striving for excellence in a sea of excellence,” Dr. McGrath added, and this can invite comparison.
Ravi Parikh, MD, a medical oncologist and physician-scientist at the University of Pennsylvania, Philadelphia, Pennsylvania, remembered struggling with imposter syndrome early in his career. As a new doctor, he had a ton of questions, and yet those above him seemed able to make weighty decisions on their own. The comparison shook his confidence. “I remember thinking that when I became an attending, I would just magically not have to run decisions by people,” said Dr. Parikh. But even then, the “magical” self-assurance didn’t materialize.
Research found that imposter syndrome is more likely to affect women and groups that are underrepresented in medicine. But overall, the incidence is remarkably high.
A 2023 survey published in the Journal of the American College of Surgeons found that 90% of female surgeons and more than two-thirds of male ones experienced imposter syndrome. In a 2023 study on medical students in JAMA, it was nearly universal; 97% reported feelings of imposter syndrome with women 1.7 times more likely to report it than men and underrepresented groups often three times more likely.
‘I’m Clearly in the Minority Here’
The term “imposter” also suggests a lack of belonging. If medicine doesn’t “look like you,” this can create feelings of pressure, like you’re “representing a whole group with your mere existence,” said Dr. Gold, “and you have to keep proving yourself.”
Chloe Slocum, MD, MPH, an assistant professor of physical medicine and rehabilitation at Harvard Medical School, Boston, Massachusetts, remembered that feeling of conspicuous “otherness.” As a resident, Dr. Slocum began presenting at national meetings and later pursued physician leadership training. Many of her counterparts at these events were older males. “At some programs early on, I’d wonder, ‘I’m clearly in the minority here; did they really make the right decision including me in this?’”
Reactions from those around you can also have an impact. Dr. McGrath — who is 5’ 2” and describes herself as looking “very young” — noted that when she started out, neither patients nor other providers thought she was a doctor.
“I have tried everything in the book to be seen, in somebody else’s eyes, as more consistent with a doctor,” she said. “I’ve dressed down. I’ve dressed up. I’ve worn heels. I’ve worn flats. I’ve worn glasses. I’ve done all the things. When you’re constantly being told you don’t look like a doctor, you start questioning yourself.”
The Emotional Toll
If that sounds mentally exhausting, it is. Research found that imposter syndrome is often linked with burnout, depression, and anxiety.
The need to prove yourself and prevent being “found out” can push some doctors toward traditional measurements of success — promotions or published work, said Dr. Gold. But “if you’re trying to achieve in ways that you don’t value,” she warned, “you’re going to burn out.”
On the other hand, intense self-doubt can also limit advancement. After all, if you don’t think you’re good enough, you may not apply for job opportunities or leadership positions.
This mental burden can persist over years and even decades. A 2020 review of studies on imposter syndrome noted that “it would be reassuring to believe that imposter symptoms decline with age.” Unfortunately, several studies indicated that they do not.
How to Manage Imposter Syndrome
While it can be difficult to overcome imposter syndrome, there are ways to work through it and make it less pervasive or intense. Here are some tips from our experts:
- Prioritize your mental health. This can be difficult for some physicians, but don’t ignore symptoms of depression, anxiety, or burnout. Untreated mental health conditions cloud the ability to reflect on some of the existential questions that will help you navigate imposter syndrome, said Dr. Gold.
- Assess how often you need validation and why. Try to identify what you›re feeling, what needs aren›t being met, and how you can meet those needs. You can then consider where to get that validation either internally or by connecting with a colleague. Dr. McGrath encourages physicians to ask, “What does success look like for me?” and can you make success more personal and meaningful. It might sound shocking, but rather than an unattainable ideal, success should be something that feels good.
- Know the power of teamwork. As Dr. Parikh eventually realized, collaborative care is a common and beneficial part of medicine — not something that makes you a less-than physician. “There’s a lot of opportunity to crowdsource the medical decision-making process in ways that increase your own confidence as a doctor,” he said.
- Practice self-compassion. Critical voices in your head add to an already hard and stressful world. This is where self-compassion comes in. “We don’t have much control over medicine, but we have control over how medicine makes us feel,” Dr. Gold said. Imagine treating yourself how you would treat a friend.
- Consider a physician coach. suggests that physician coaches can help lower rates of burnout and improve well-being, resilience, professional fulfillment, and self-worth. “Coaching looks into the future to help you envision what things would look like if you were feeling differently. It helps you explore what’s in your control and how you want to shape that,” said Dr. McGrath.
- Amplify the good. Apps and web-based tools can remind you to celebrate your own achievements. The “” exercise created by J. Bryan Sexton, PhD, at the Duke Center for Healthcare Safety & Quality for example, was documented in a . When healthcare workers reflected on three good things that happened each day for 2 weeks, they reported significant improvements in depression, burnout, and work-life balance.
- Do a values check. Dr. Gold often suggested that physicians with imposter syndrome ask themselves what they value, what medicine values, and how the two line up. Pausing to consider this can guide you toward useful strategies. If you value family life but feel like medicine doesn’t, for example, you might talk with a colleague who has navigated this conflict.
Dr. Gold added that reminding yourself of the range of options can be freeing. “There’s no ‘one career’ in medicine,” she said. “There are multiple ways to be happy in medicine; there are multiple ways to be happy outside of medicine. And you’re not a failure for the path you choose.”
A version of this article appeared on Medscape.com.
Do I deserve to be here? Am I doing what I’m supposed to be doing? Is anyone going to tell me if I’m terrible?
Kerri Palamara McGrath, MD, remembered worrying over these questions as chief resident at Massachusetts General Hospital, Boston, Massachusetts, in 2009. Having graduated from New York Medical College, she felt out of step with her peers from Ivy League medical schools and considered herself lucky to be there. In order to measure up, she felt she had to work twice as hard as everybody else.
But as Dr. McGrath moved through residency and spoke with other trainees, she had a realization. Her constant fears, the nagging voice in her head saying she wasn’t good enough, these issues weren’t exclusive to her; they were pervasive.
Today, Dr. McGrath is the director of the Center for Physician Well-Being at Massachusetts General Hospital. The facility aims to address physician stress and equip doctors with the tools to navigate personal and professional issues. Dr. McGrath is also a physician coach, a growing nonclinical field, helping doctors identify their own stressors, values, and measures of success. This type of internal work, Dr. McGrath feels, can help alleviate imposter syndrome, that inner refrain saying: I’ll never be good enough.
What Is Imposter Syndrome?
While not a formal medical diagnosis, imposter syndrome has been defined as «an internal experience of intellectual phoniness.» It›s considered an inability to internalize success and a tendency to attribute gains to external factors — for example, being in the right place at the right time.
“Many people describe imposter phenomena in medicine as fearing that others are going to realize that they don’t belong somewhere or question why they’re there,” said Dr. McGrath.
It’s a “fear of being found out,” added Jessi Gold, MD, a psychiatrist who treats physicians. “In many ways, imposter syndrome shows up as a conflict between the outer self — the metaphorical mask you’re ‘putting on’ [in order] to achieve, and the inner self — how you feel like you’re not measuring up.”
Dr. McGrath said she experienced imposter syndrome before her medical career even began. She applied to 26 medical schools. Only one accepted her. “The whole time, I was like, ‘This is the only school you got into, so you’re obviously not good enough,’” she recalled. Later, having been chosen by a “coveted” institution like Mass General, “you assume that, at some point, someone will realize that the gig is up, that everybody’s better than you.”
Where Does Imposter Syndrome Come From?
Dr. McGrath felt that in medicine, high expectations are often coupled with low self-compassion. “We are so hard on ourselves, and when we set our expectations so high, we’re constantly disappointed in ourselves,” she said. External markers of success — papers published, promotions, or even social media — can further fuel this.
It can feel like “striving for excellence in a sea of excellence,” Dr. McGrath added, and this can invite comparison.
Ravi Parikh, MD, a medical oncologist and physician-scientist at the University of Pennsylvania, Philadelphia, Pennsylvania, remembered struggling with imposter syndrome early in his career. As a new doctor, he had a ton of questions, and yet those above him seemed able to make weighty decisions on their own. The comparison shook his confidence. “I remember thinking that when I became an attending, I would just magically not have to run decisions by people,” said Dr. Parikh. But even then, the “magical” self-assurance didn’t materialize.
Research found that imposter syndrome is more likely to affect women and groups that are underrepresented in medicine. But overall, the incidence is remarkably high.
A 2023 survey published in the Journal of the American College of Surgeons found that 90% of female surgeons and more than two-thirds of male ones experienced imposter syndrome. In a 2023 study on medical students in JAMA, it was nearly universal; 97% reported feelings of imposter syndrome with women 1.7 times more likely to report it than men and underrepresented groups often three times more likely.
‘I’m Clearly in the Minority Here’
The term “imposter” also suggests a lack of belonging. If medicine doesn’t “look like you,” this can create feelings of pressure, like you’re “representing a whole group with your mere existence,” said Dr. Gold, “and you have to keep proving yourself.”
Chloe Slocum, MD, MPH, an assistant professor of physical medicine and rehabilitation at Harvard Medical School, Boston, Massachusetts, remembered that feeling of conspicuous “otherness.” As a resident, Dr. Slocum began presenting at national meetings and later pursued physician leadership training. Many of her counterparts at these events were older males. “At some programs early on, I’d wonder, ‘I’m clearly in the minority here; did they really make the right decision including me in this?’”
Reactions from those around you can also have an impact. Dr. McGrath — who is 5’ 2” and describes herself as looking “very young” — noted that when she started out, neither patients nor other providers thought she was a doctor.
“I have tried everything in the book to be seen, in somebody else’s eyes, as more consistent with a doctor,” she said. “I’ve dressed down. I’ve dressed up. I’ve worn heels. I’ve worn flats. I’ve worn glasses. I’ve done all the things. When you’re constantly being told you don’t look like a doctor, you start questioning yourself.”
The Emotional Toll
If that sounds mentally exhausting, it is. Research found that imposter syndrome is often linked with burnout, depression, and anxiety.
The need to prove yourself and prevent being “found out” can push some doctors toward traditional measurements of success — promotions or published work, said Dr. Gold. But “if you’re trying to achieve in ways that you don’t value,” she warned, “you’re going to burn out.”
On the other hand, intense self-doubt can also limit advancement. After all, if you don’t think you’re good enough, you may not apply for job opportunities or leadership positions.
This mental burden can persist over years and even decades. A 2020 review of studies on imposter syndrome noted that “it would be reassuring to believe that imposter symptoms decline with age.” Unfortunately, several studies indicated that they do not.
How to Manage Imposter Syndrome
While it can be difficult to overcome imposter syndrome, there are ways to work through it and make it less pervasive or intense. Here are some tips from our experts:
- Prioritize your mental health. This can be difficult for some physicians, but don’t ignore symptoms of depression, anxiety, or burnout. Untreated mental health conditions cloud the ability to reflect on some of the existential questions that will help you navigate imposter syndrome, said Dr. Gold.
- Assess how often you need validation and why. Try to identify what you›re feeling, what needs aren›t being met, and how you can meet those needs. You can then consider where to get that validation either internally or by connecting with a colleague. Dr. McGrath encourages physicians to ask, “What does success look like for me?” and can you make success more personal and meaningful. It might sound shocking, but rather than an unattainable ideal, success should be something that feels good.
- Know the power of teamwork. As Dr. Parikh eventually realized, collaborative care is a common and beneficial part of medicine — not something that makes you a less-than physician. “There’s a lot of opportunity to crowdsource the medical decision-making process in ways that increase your own confidence as a doctor,” he said.
- Practice self-compassion. Critical voices in your head add to an already hard and stressful world. This is where self-compassion comes in. “We don’t have much control over medicine, but we have control over how medicine makes us feel,” Dr. Gold said. Imagine treating yourself how you would treat a friend.
- Consider a physician coach. suggests that physician coaches can help lower rates of burnout and improve well-being, resilience, professional fulfillment, and self-worth. “Coaching looks into the future to help you envision what things would look like if you were feeling differently. It helps you explore what’s in your control and how you want to shape that,” said Dr. McGrath.
- Amplify the good. Apps and web-based tools can remind you to celebrate your own achievements. The “” exercise created by J. Bryan Sexton, PhD, at the Duke Center for Healthcare Safety & Quality for example, was documented in a . When healthcare workers reflected on three good things that happened each day for 2 weeks, they reported significant improvements in depression, burnout, and work-life balance.
- Do a values check. Dr. Gold often suggested that physicians with imposter syndrome ask themselves what they value, what medicine values, and how the two line up. Pausing to consider this can guide you toward useful strategies. If you value family life but feel like medicine doesn’t, for example, you might talk with a colleague who has navigated this conflict.
Dr. Gold added that reminding yourself of the range of options can be freeing. “There’s no ‘one career’ in medicine,” she said. “There are multiple ways to be happy in medicine; there are multiple ways to be happy outside of medicine. And you’re not a failure for the path you choose.”
A version of this article appeared on Medscape.com.
Do I deserve to be here? Am I doing what I’m supposed to be doing? Is anyone going to tell me if I’m terrible?
Kerri Palamara McGrath, MD, remembered worrying over these questions as chief resident at Massachusetts General Hospital, Boston, Massachusetts, in 2009. Having graduated from New York Medical College, she felt out of step with her peers from Ivy League medical schools and considered herself lucky to be there. In order to measure up, she felt she had to work twice as hard as everybody else.
But as Dr. McGrath moved through residency and spoke with other trainees, she had a realization. Her constant fears, the nagging voice in her head saying she wasn’t good enough, these issues weren’t exclusive to her; they were pervasive.
Today, Dr. McGrath is the director of the Center for Physician Well-Being at Massachusetts General Hospital. The facility aims to address physician stress and equip doctors with the tools to navigate personal and professional issues. Dr. McGrath is also a physician coach, a growing nonclinical field, helping doctors identify their own stressors, values, and measures of success. This type of internal work, Dr. McGrath feels, can help alleviate imposter syndrome, that inner refrain saying: I’ll never be good enough.
What Is Imposter Syndrome?
While not a formal medical diagnosis, imposter syndrome has been defined as «an internal experience of intellectual phoniness.» It›s considered an inability to internalize success and a tendency to attribute gains to external factors — for example, being in the right place at the right time.
“Many people describe imposter phenomena in medicine as fearing that others are going to realize that they don’t belong somewhere or question why they’re there,” said Dr. McGrath.
It’s a “fear of being found out,” added Jessi Gold, MD, a psychiatrist who treats physicians. “In many ways, imposter syndrome shows up as a conflict between the outer self — the metaphorical mask you’re ‘putting on’ [in order] to achieve, and the inner self — how you feel like you’re not measuring up.”
Dr. McGrath said she experienced imposter syndrome before her medical career even began. She applied to 26 medical schools. Only one accepted her. “The whole time, I was like, ‘This is the only school you got into, so you’re obviously not good enough,’” she recalled. Later, having been chosen by a “coveted” institution like Mass General, “you assume that, at some point, someone will realize that the gig is up, that everybody’s better than you.”
Where Does Imposter Syndrome Come From?
Dr. McGrath felt that in medicine, high expectations are often coupled with low self-compassion. “We are so hard on ourselves, and when we set our expectations so high, we’re constantly disappointed in ourselves,” she said. External markers of success — papers published, promotions, or even social media — can further fuel this.
It can feel like “striving for excellence in a sea of excellence,” Dr. McGrath added, and this can invite comparison.
Ravi Parikh, MD, a medical oncologist and physician-scientist at the University of Pennsylvania, Philadelphia, Pennsylvania, remembered struggling with imposter syndrome early in his career. As a new doctor, he had a ton of questions, and yet those above him seemed able to make weighty decisions on their own. The comparison shook his confidence. “I remember thinking that when I became an attending, I would just magically not have to run decisions by people,” said Dr. Parikh. But even then, the “magical” self-assurance didn’t materialize.
Research found that imposter syndrome is more likely to affect women and groups that are underrepresented in medicine. But overall, the incidence is remarkably high.
A 2023 survey published in the Journal of the American College of Surgeons found that 90% of female surgeons and more than two-thirds of male ones experienced imposter syndrome. In a 2023 study on medical students in JAMA, it was nearly universal; 97% reported feelings of imposter syndrome with women 1.7 times more likely to report it than men and underrepresented groups often three times more likely.
‘I’m Clearly in the Minority Here’
The term “imposter” also suggests a lack of belonging. If medicine doesn’t “look like you,” this can create feelings of pressure, like you’re “representing a whole group with your mere existence,” said Dr. Gold, “and you have to keep proving yourself.”
Chloe Slocum, MD, MPH, an assistant professor of physical medicine and rehabilitation at Harvard Medical School, Boston, Massachusetts, remembered that feeling of conspicuous “otherness.” As a resident, Dr. Slocum began presenting at national meetings and later pursued physician leadership training. Many of her counterparts at these events were older males. “At some programs early on, I’d wonder, ‘I’m clearly in the minority here; did they really make the right decision including me in this?’”
Reactions from those around you can also have an impact. Dr. McGrath — who is 5’ 2” and describes herself as looking “very young” — noted that when she started out, neither patients nor other providers thought she was a doctor.
“I have tried everything in the book to be seen, in somebody else’s eyes, as more consistent with a doctor,” she said. “I’ve dressed down. I’ve dressed up. I’ve worn heels. I’ve worn flats. I’ve worn glasses. I’ve done all the things. When you’re constantly being told you don’t look like a doctor, you start questioning yourself.”
The Emotional Toll
If that sounds mentally exhausting, it is. Research found that imposter syndrome is often linked with burnout, depression, and anxiety.
The need to prove yourself and prevent being “found out” can push some doctors toward traditional measurements of success — promotions or published work, said Dr. Gold. But “if you’re trying to achieve in ways that you don’t value,” she warned, “you’re going to burn out.”
On the other hand, intense self-doubt can also limit advancement. After all, if you don’t think you’re good enough, you may not apply for job opportunities or leadership positions.
This mental burden can persist over years and even decades. A 2020 review of studies on imposter syndrome noted that “it would be reassuring to believe that imposter symptoms decline with age.” Unfortunately, several studies indicated that they do not.
How to Manage Imposter Syndrome
While it can be difficult to overcome imposter syndrome, there are ways to work through it and make it less pervasive or intense. Here are some tips from our experts:
- Prioritize your mental health. This can be difficult for some physicians, but don’t ignore symptoms of depression, anxiety, or burnout. Untreated mental health conditions cloud the ability to reflect on some of the existential questions that will help you navigate imposter syndrome, said Dr. Gold.
- Assess how often you need validation and why. Try to identify what you›re feeling, what needs aren›t being met, and how you can meet those needs. You can then consider where to get that validation either internally or by connecting with a colleague. Dr. McGrath encourages physicians to ask, “What does success look like for me?” and can you make success more personal and meaningful. It might sound shocking, but rather than an unattainable ideal, success should be something that feels good.
- Know the power of teamwork. As Dr. Parikh eventually realized, collaborative care is a common and beneficial part of medicine — not something that makes you a less-than physician. “There’s a lot of opportunity to crowdsource the medical decision-making process in ways that increase your own confidence as a doctor,” he said.
- Practice self-compassion. Critical voices in your head add to an already hard and stressful world. This is where self-compassion comes in. “We don’t have much control over medicine, but we have control over how medicine makes us feel,” Dr. Gold said. Imagine treating yourself how you would treat a friend.
- Consider a physician coach. suggests that physician coaches can help lower rates of burnout and improve well-being, resilience, professional fulfillment, and self-worth. “Coaching looks into the future to help you envision what things would look like if you were feeling differently. It helps you explore what’s in your control and how you want to shape that,” said Dr. McGrath.
- Amplify the good. Apps and web-based tools can remind you to celebrate your own achievements. The “” exercise created by J. Bryan Sexton, PhD, at the Duke Center for Healthcare Safety & Quality for example, was documented in a . When healthcare workers reflected on three good things that happened each day for 2 weeks, they reported significant improvements in depression, burnout, and work-life balance.
- Do a values check. Dr. Gold often suggested that physicians with imposter syndrome ask themselves what they value, what medicine values, and how the two line up. Pausing to consider this can guide you toward useful strategies. If you value family life but feel like medicine doesn’t, for example, you might talk with a colleague who has navigated this conflict.
Dr. Gold added that reminding yourself of the range of options can be freeing. “There’s no ‘one career’ in medicine,” she said. “There are multiple ways to be happy in medicine; there are multiple ways to be happy outside of medicine. And you’re not a failure for the path you choose.”
A version of this article appeared on Medscape.com.
COVID-19 Is a Very Weird Virus
This transcript has been edited for clarity.
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr F. Perry Wilson of the Yale School of Medicine.
In the early days of the pandemic, before we really understood what COVID was, two specialties in the hospital had a foreboding sense that something was very strange about this virus. The first was the pulmonologists, who noticed the striking levels of hypoxemia — low oxygen in the blood — and the rapidity with which patients who had previously been stable would crash in the intensive care unit.
The second, and I mark myself among this group, were the nephrologists. The dialysis machines stopped working right. I remember rounding on patients in the hospital who were on dialysis for kidney failure in the setting of severe COVID infection and seeing clots forming on the dialysis filters. Some patients could barely get in a full treatment because the filters would clog so quickly.
We knew it was worse than flu because of the mortality rates, but these oddities made us realize that it was different too — not just a particularly nasty respiratory virus but one that had effects on the body that we hadn’t really seen before.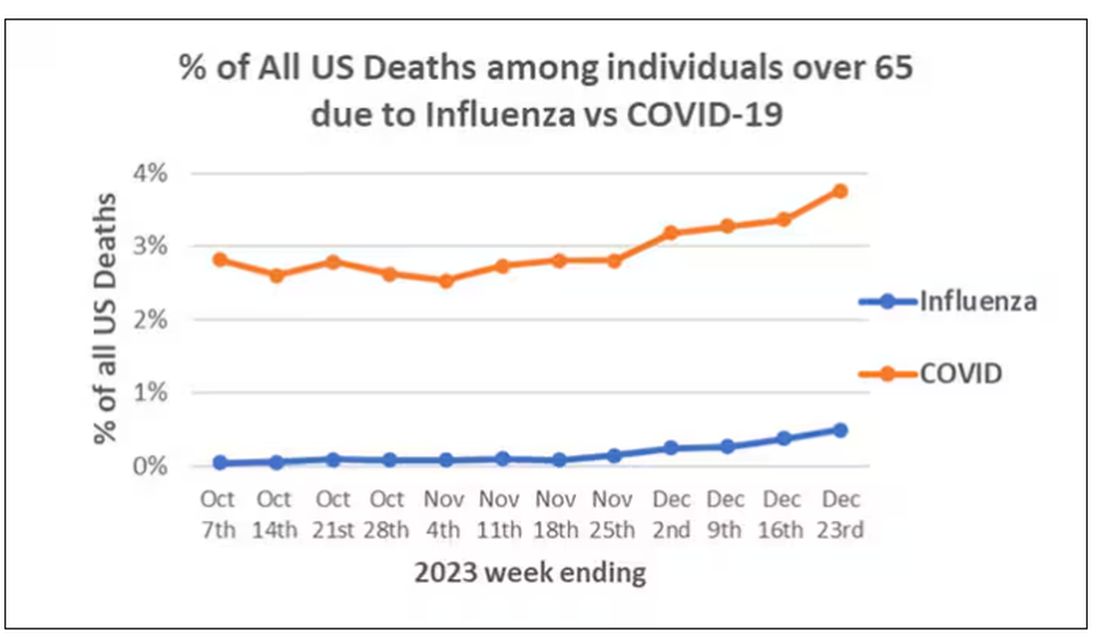
That’s why I’ve always been interested in studies that compare what happens to patients after COVID infection vs what happens to patients after other respiratory infections. This week, we’ll look at an intriguing study that suggests that COVID may lead to autoimmune diseases like rheumatoid arthritis, lupus, and vasculitis.
The study appears in the Annals of Internal Medicine and is made possible by the universal electronic health record systems of South Korea and Japan, who collaborated to create a truly staggering cohort of more than 20 million individuals living in those countries from 2020 to 2021.
The exposure of interest? COVID infection, experienced by just under 5% of that cohort over the study period. (Remember, there was a time when COVID infections were relatively controlled, particularly in some countries.)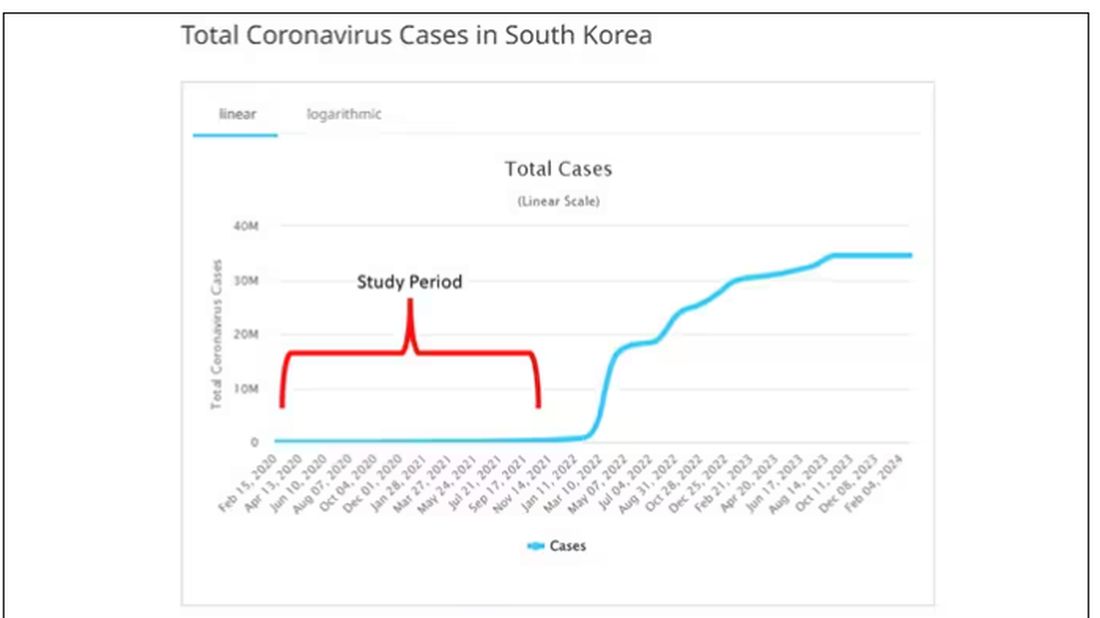
The researchers wanted to compare the risk for autoimmune disease among COVID-infected individuals against two control groups. The first control group was the general population. This is interesting but a difficult analysis, because people who become infected with COVID might be very different from the general population. The second control group was people infected with influenza. I like this a lot better; the risk factors for COVID and influenza are quite similar, and the fact that this group was diagnosed with flu means at least that they are getting medical care and are sort of “in the system,” so to speak.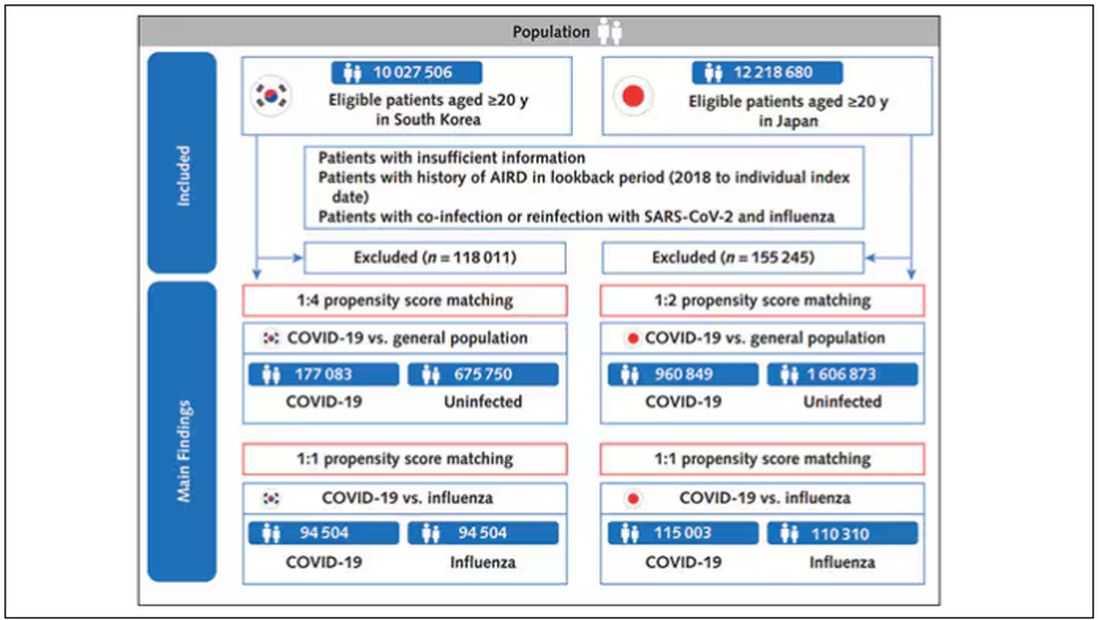
But it’s not enough to simply identify these folks and see who ends up with more autoimmune disease. The authors used propensity score matching to pair individuals infected with COVID with individuals from the control groups who were very similar to them. I’ve talked about this strategy before, but the basic idea is that you build a model predicting the likelihood of infection with COVID, based on a slew of factors — and the slew these authors used is pretty big, as shown below — and then stick people with similar risk for COVID together, with one member of the pair having had COVID and the other having eluded it (at least for the study period).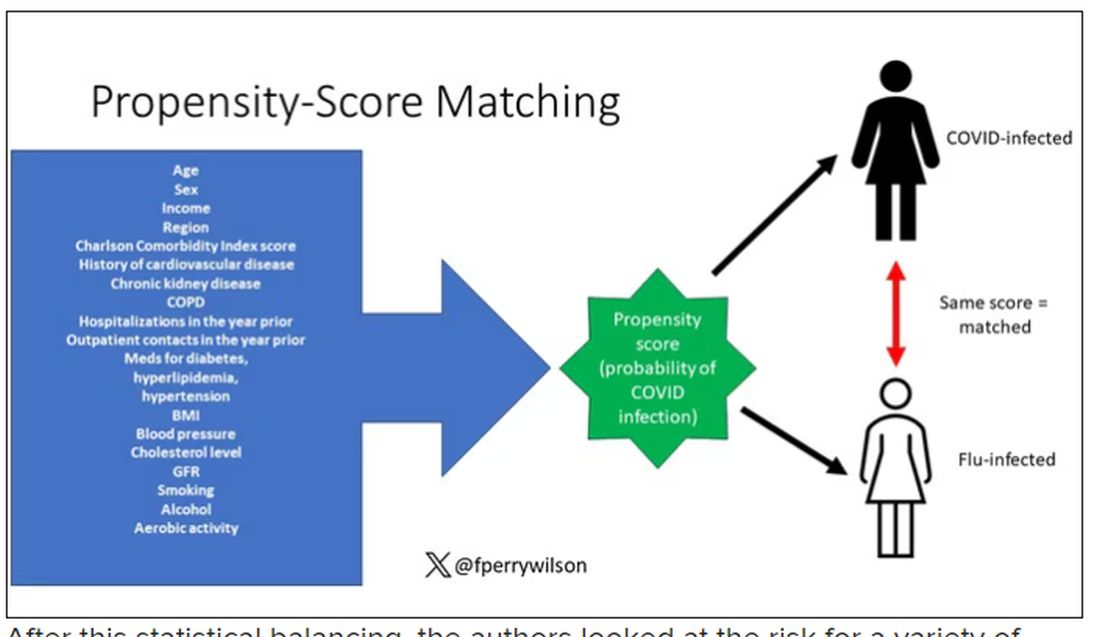
After this statistical balancing, the authors looked at the risk for a variety of autoimmune diseases.
Compared with those infected with flu, those infected with COVID were more likely to be diagnosed with any autoimmune condition, connective tissue disease, and, in Japan at least, inflammatory arthritis.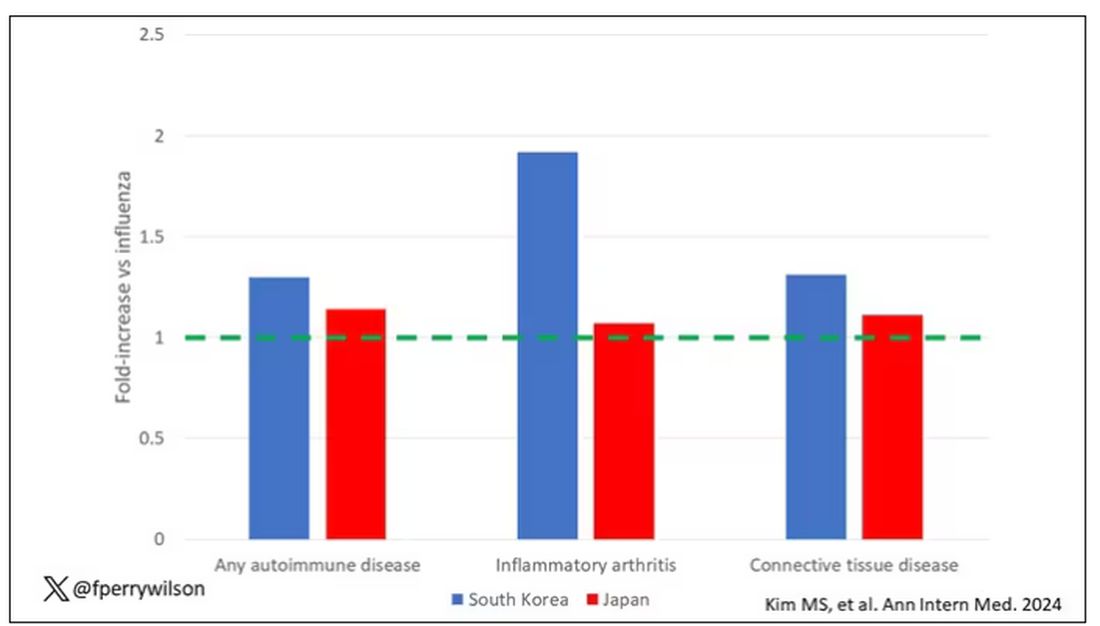
The authors acknowledge that being diagnosed with a disease might not be the same as actually having the disease, so in another analysis they looked only at people who received treatment for the autoimmune conditions, and the signals were even stronger in that group.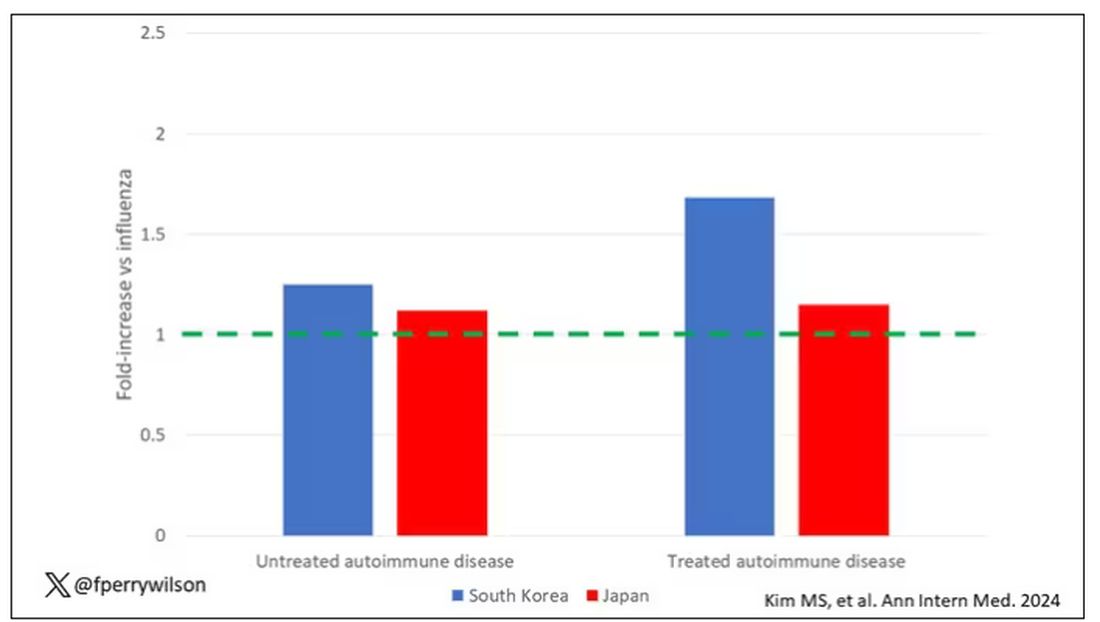
This risk seemed to be highest in the 6 months following the COVID infection, which makes sense biologically if we think that the infection is somehow screwing up the immune system.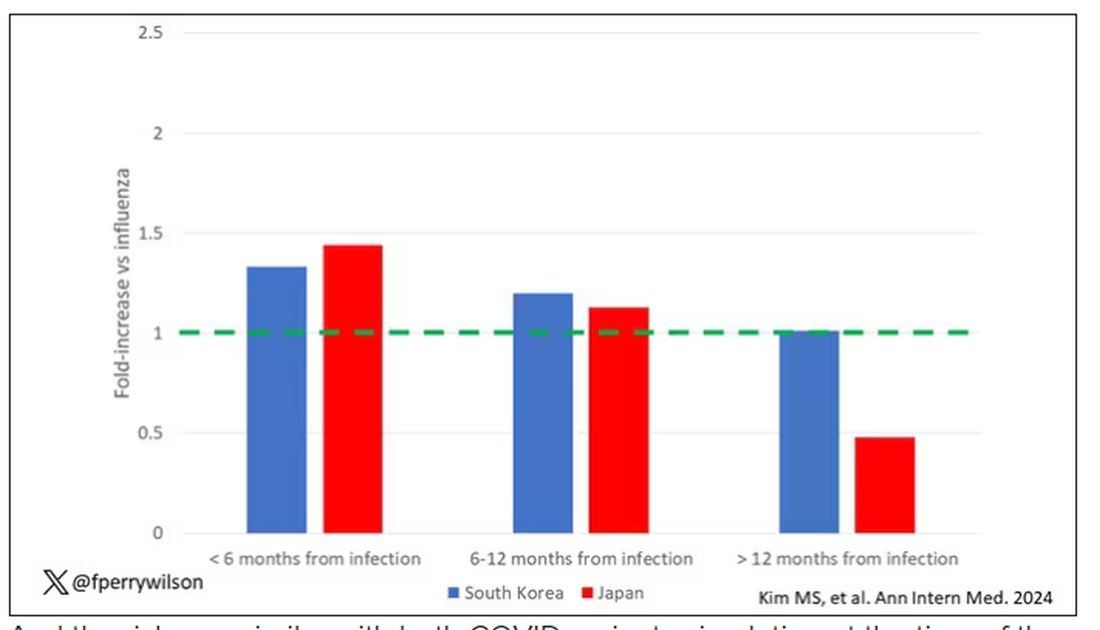
And the risk was similar with both COVID variants circulating at the time of the study.
The only factor that reduced the risk? You guessed it: vaccination. This is a particularly interesting finding because the exposure cohort was defined by having been infected with COVID. Therefore, the mechanism of protection is not prevention of infection; it’s something else. Perhaps vaccination helps to get the immune system in a state to respond to COVID infection more… appropriately?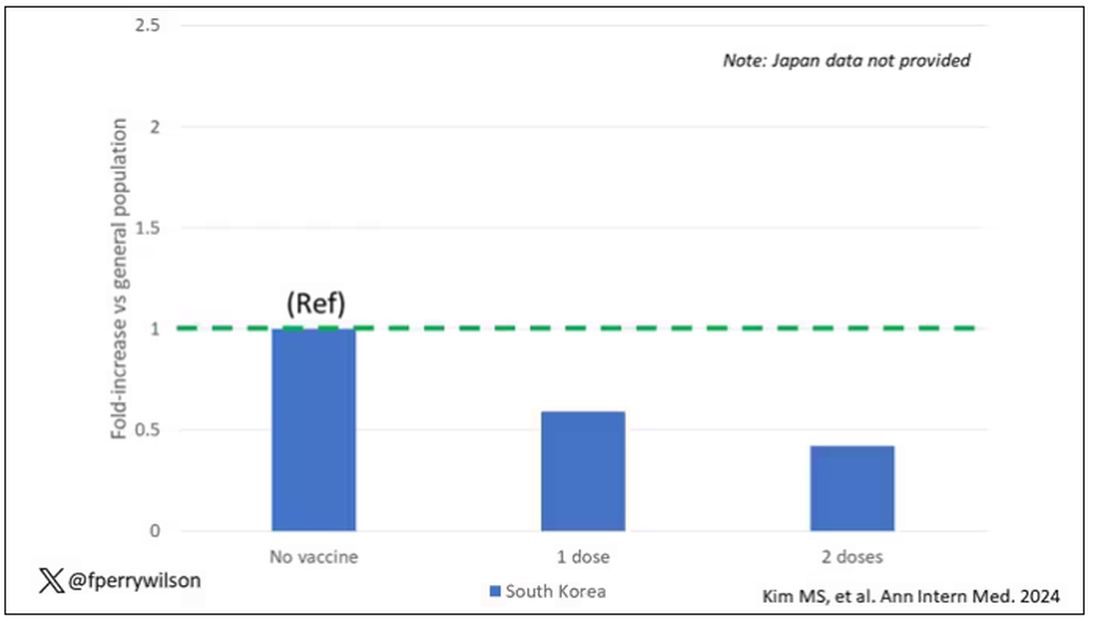
Yes, this study is observational. We can’t draw causal conclusions here. But it does reinforce my long-held belief that COVID is a weird virus, one with effects that are different from the respiratory viruses we are used to. I can’t say for certain whether COVID causes immune system dysfunction that puts someone at risk for autoimmunity — not from this study. But I can say it wouldn’t surprise me.
Dr. F. Perry Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr F. Perry Wilson of the Yale School of Medicine.
In the early days of the pandemic, before we really understood what COVID was, two specialties in the hospital had a foreboding sense that something was very strange about this virus. The first was the pulmonologists, who noticed the striking levels of hypoxemia — low oxygen in the blood — and the rapidity with which patients who had previously been stable would crash in the intensive care unit.
The second, and I mark myself among this group, were the nephrologists. The dialysis machines stopped working right. I remember rounding on patients in the hospital who were on dialysis for kidney failure in the setting of severe COVID infection and seeing clots forming on the dialysis filters. Some patients could barely get in a full treatment because the filters would clog so quickly.
We knew it was worse than flu because of the mortality rates, but these oddities made us realize that it was different too — not just a particularly nasty respiratory virus but one that had effects on the body that we hadn’t really seen before.
That’s why I’ve always been interested in studies that compare what happens to patients after COVID infection vs what happens to patients after other respiratory infections. This week, we’ll look at an intriguing study that suggests that COVID may lead to autoimmune diseases like rheumatoid arthritis, lupus, and vasculitis.
The study appears in the Annals of Internal Medicine and is made possible by the universal electronic health record systems of South Korea and Japan, who collaborated to create a truly staggering cohort of more than 20 million individuals living in those countries from 2020 to 2021.
The exposure of interest? COVID infection, experienced by just under 5% of that cohort over the study period. (Remember, there was a time when COVID infections were relatively controlled, particularly in some countries.)
The researchers wanted to compare the risk for autoimmune disease among COVID-infected individuals against two control groups. The first control group was the general population. This is interesting but a difficult analysis, because people who become infected with COVID might be very different from the general population. The second control group was people infected with influenza. I like this a lot better; the risk factors for COVID and influenza are quite similar, and the fact that this group was diagnosed with flu means at least that they are getting medical care and are sort of “in the system,” so to speak.
But it’s not enough to simply identify these folks and see who ends up with more autoimmune disease. The authors used propensity score matching to pair individuals infected with COVID with individuals from the control groups who were very similar to them. I’ve talked about this strategy before, but the basic idea is that you build a model predicting the likelihood of infection with COVID, based on a slew of factors — and the slew these authors used is pretty big, as shown below — and then stick people with similar risk for COVID together, with one member of the pair having had COVID and the other having eluded it (at least for the study period).
After this statistical balancing, the authors looked at the risk for a variety of autoimmune diseases.
Compared with those infected with flu, those infected with COVID were more likely to be diagnosed with any autoimmune condition, connective tissue disease, and, in Japan at least, inflammatory arthritis.
The authors acknowledge that being diagnosed with a disease might not be the same as actually having the disease, so in another analysis they looked only at people who received treatment for the autoimmune conditions, and the signals were even stronger in that group.
This risk seemed to be highest in the 6 months following the COVID infection, which makes sense biologically if we think that the infection is somehow screwing up the immune system.
And the risk was similar with both COVID variants circulating at the time of the study.
The only factor that reduced the risk? You guessed it: vaccination. This is a particularly interesting finding because the exposure cohort was defined by having been infected with COVID. Therefore, the mechanism of protection is not prevention of infection; it’s something else. Perhaps vaccination helps to get the immune system in a state to respond to COVID infection more… appropriately?
Yes, this study is observational. We can’t draw causal conclusions here. But it does reinforce my long-held belief that COVID is a weird virus, one with effects that are different from the respiratory viruses we are used to. I can’t say for certain whether COVID causes immune system dysfunction that puts someone at risk for autoimmunity — not from this study. But I can say it wouldn’t surprise me.
Dr. F. Perry Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr F. Perry Wilson of the Yale School of Medicine.
In the early days of the pandemic, before we really understood what COVID was, two specialties in the hospital had a foreboding sense that something was very strange about this virus. The first was the pulmonologists, who noticed the striking levels of hypoxemia — low oxygen in the blood — and the rapidity with which patients who had previously been stable would crash in the intensive care unit.
The second, and I mark myself among this group, were the nephrologists. The dialysis machines stopped working right. I remember rounding on patients in the hospital who were on dialysis for kidney failure in the setting of severe COVID infection and seeing clots forming on the dialysis filters. Some patients could barely get in a full treatment because the filters would clog so quickly.
We knew it was worse than flu because of the mortality rates, but these oddities made us realize that it was different too — not just a particularly nasty respiratory virus but one that had effects on the body that we hadn’t really seen before.
That’s why I’ve always been interested in studies that compare what happens to patients after COVID infection vs what happens to patients after other respiratory infections. This week, we’ll look at an intriguing study that suggests that COVID may lead to autoimmune diseases like rheumatoid arthritis, lupus, and vasculitis.
The study appears in the Annals of Internal Medicine and is made possible by the universal electronic health record systems of South Korea and Japan, who collaborated to create a truly staggering cohort of more than 20 million individuals living in those countries from 2020 to 2021.
The exposure of interest? COVID infection, experienced by just under 5% of that cohort over the study period. (Remember, there was a time when COVID infections were relatively controlled, particularly in some countries.)
The researchers wanted to compare the risk for autoimmune disease among COVID-infected individuals against two control groups. The first control group was the general population. This is interesting but a difficult analysis, because people who become infected with COVID might be very different from the general population. The second control group was people infected with influenza. I like this a lot better; the risk factors for COVID and influenza are quite similar, and the fact that this group was diagnosed with flu means at least that they are getting medical care and are sort of “in the system,” so to speak.
But it’s not enough to simply identify these folks and see who ends up with more autoimmune disease. The authors used propensity score matching to pair individuals infected with COVID with individuals from the control groups who were very similar to them. I’ve talked about this strategy before, but the basic idea is that you build a model predicting the likelihood of infection with COVID, based on a slew of factors — and the slew these authors used is pretty big, as shown below — and then stick people with similar risk for COVID together, with one member of the pair having had COVID and the other having eluded it (at least for the study period).
After this statistical balancing, the authors looked at the risk for a variety of autoimmune diseases.
Compared with those infected with flu, those infected with COVID were more likely to be diagnosed with any autoimmune condition, connective tissue disease, and, in Japan at least, inflammatory arthritis.
The authors acknowledge that being diagnosed with a disease might not be the same as actually having the disease, so in another analysis they looked only at people who received treatment for the autoimmune conditions, and the signals were even stronger in that group.
This risk seemed to be highest in the 6 months following the COVID infection, which makes sense biologically if we think that the infection is somehow screwing up the immune system.
And the risk was similar with both COVID variants circulating at the time of the study.
The only factor that reduced the risk? You guessed it: vaccination. This is a particularly interesting finding because the exposure cohort was defined by having been infected with COVID. Therefore, the mechanism of protection is not prevention of infection; it’s something else. Perhaps vaccination helps to get the immune system in a state to respond to COVID infection more… appropriately?
Yes, this study is observational. We can’t draw causal conclusions here. But it does reinforce my long-held belief that COVID is a weird virus, one with effects that are different from the respiratory viruses we are used to. I can’t say for certain whether COVID causes immune system dysfunction that puts someone at risk for autoimmunity — not from this study. But I can say it wouldn’t surprise me.
Dr. F. Perry Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
Psilocybin Poison Control Calls Spike in Teens, Young Adults
Calls to US poison centers related to psilocybin more than tripled among teens and more than doubled in young adults between 2019 and 2022, new research suggests. Investigators say the increase may be linked to decriminalization efforts in US cities and states.
METHODOLOGY:
- Investigators used data from the National Poison Data System (NPDS) to identify calls involving psilocybin between January 2013 and December 2022.
- Researchers focused on calls about individuals between the ages of 13 and 25 years.
- Exposures to psilocybin were examined based on demographics, clinical effects, level of care, and medical outcome.
TAKEAWAY:
- During the entire 10-year study period, 4055 psilocybin-involved exposures were reported in the age groups studied, with 66% being single-substance exposures and close to three quarters receiving medical attention.
- Psilocybin’s most common effects were hallucinations or delusions (37% of calls), agitation (28%), tachycardia (20%), and confusion (16%).
- The number of psilocybin-related calls to poison control centers for youth were largely unchanged from 2013 to 2018 but more than tripled among adolescents (aged 13-19 years) from 2019 and 2022 and more than doubled among young adults (aged 20-25 years) between 2018 and 2022 (P < .0001).
IN PRACTICE:
The increase in poison center calls coincides with psilocybin decriminalization efforts in several states in 2019, the authors noted. However, because those efforts only legalized use in adults aged 21 years and older, the rise among younger people is concerning, they added. “As psilocybin may become more widely available, it is important for parents to be aware that psilocybin is also available in edible forms such as chocolate and gummies. And we learned from our experience with edible cannabis that young children can mistake edibles for candy,” lead author Rita Farah, PharmD, MPH, PhD, Blue Ridge Poison Center epidemiologist, said in a news release.
SOURCE:
Christopher Holstege, MD, director of UVA Health’s Blue Ridge Poison Center and chief of the Division of Medical Toxicology at the UVA School of Medicine was the senior and corresponding author of the study. It was published online on February 26 in the Journal of Adolescent Health.
LIMITATIONS:
NPDS data are not designed to assess potential risk factors leading to increases in psilocybin-related cases. Moreover, because reports to poison control centers are voluntary and don’t capture all exposures, NPDS data likely under-represent cases of hallucinogenic mushroom poisonings. Lastly, NPDS data are susceptible to reporting and misclassification biases.
DISCLOSURES:
Funding source was not disclosed. The authors reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Calls to US poison centers related to psilocybin more than tripled among teens and more than doubled in young adults between 2019 and 2022, new research suggests. Investigators say the increase may be linked to decriminalization efforts in US cities and states.
METHODOLOGY:
- Investigators used data from the National Poison Data System (NPDS) to identify calls involving psilocybin between January 2013 and December 2022.
- Researchers focused on calls about individuals between the ages of 13 and 25 years.
- Exposures to psilocybin were examined based on demographics, clinical effects, level of care, and medical outcome.
TAKEAWAY:
- During the entire 10-year study period, 4055 psilocybin-involved exposures were reported in the age groups studied, with 66% being single-substance exposures and close to three quarters receiving medical attention.
- Psilocybin’s most common effects were hallucinations or delusions (37% of calls), agitation (28%), tachycardia (20%), and confusion (16%).
- The number of psilocybin-related calls to poison control centers for youth were largely unchanged from 2013 to 2018 but more than tripled among adolescents (aged 13-19 years) from 2019 and 2022 and more than doubled among young adults (aged 20-25 years) between 2018 and 2022 (P < .0001).
IN PRACTICE:
The increase in poison center calls coincides with psilocybin decriminalization efforts in several states in 2019, the authors noted. However, because those efforts only legalized use in adults aged 21 years and older, the rise among younger people is concerning, they added. “As psilocybin may become more widely available, it is important for parents to be aware that psilocybin is also available in edible forms such as chocolate and gummies. And we learned from our experience with edible cannabis that young children can mistake edibles for candy,” lead author Rita Farah, PharmD, MPH, PhD, Blue Ridge Poison Center epidemiologist, said in a news release.
SOURCE:
Christopher Holstege, MD, director of UVA Health’s Blue Ridge Poison Center and chief of the Division of Medical Toxicology at the UVA School of Medicine was the senior and corresponding author of the study. It was published online on February 26 in the Journal of Adolescent Health.
LIMITATIONS:
NPDS data are not designed to assess potential risk factors leading to increases in psilocybin-related cases. Moreover, because reports to poison control centers are voluntary and don’t capture all exposures, NPDS data likely under-represent cases of hallucinogenic mushroom poisonings. Lastly, NPDS data are susceptible to reporting and misclassification biases.
DISCLOSURES:
Funding source was not disclosed. The authors reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Calls to US poison centers related to psilocybin more than tripled among teens and more than doubled in young adults between 2019 and 2022, new research suggests. Investigators say the increase may be linked to decriminalization efforts in US cities and states.
METHODOLOGY:
- Investigators used data from the National Poison Data System (NPDS) to identify calls involving psilocybin between January 2013 and December 2022.
- Researchers focused on calls about individuals between the ages of 13 and 25 years.
- Exposures to psilocybin were examined based on demographics, clinical effects, level of care, and medical outcome.
TAKEAWAY:
- During the entire 10-year study period, 4055 psilocybin-involved exposures were reported in the age groups studied, with 66% being single-substance exposures and close to three quarters receiving medical attention.
- Psilocybin’s most common effects were hallucinations or delusions (37% of calls), agitation (28%), tachycardia (20%), and confusion (16%).
- The number of psilocybin-related calls to poison control centers for youth were largely unchanged from 2013 to 2018 but more than tripled among adolescents (aged 13-19 years) from 2019 and 2022 and more than doubled among young adults (aged 20-25 years) between 2018 and 2022 (P < .0001).
IN PRACTICE:
The increase in poison center calls coincides with psilocybin decriminalization efforts in several states in 2019, the authors noted. However, because those efforts only legalized use in adults aged 21 years and older, the rise among younger people is concerning, they added. “As psilocybin may become more widely available, it is important for parents to be aware that psilocybin is also available in edible forms such as chocolate and gummies. And we learned from our experience with edible cannabis that young children can mistake edibles for candy,” lead author Rita Farah, PharmD, MPH, PhD, Blue Ridge Poison Center epidemiologist, said in a news release.
SOURCE:
Christopher Holstege, MD, director of UVA Health’s Blue Ridge Poison Center and chief of the Division of Medical Toxicology at the UVA School of Medicine was the senior and corresponding author of the study. It was published online on February 26 in the Journal of Adolescent Health.
LIMITATIONS:
NPDS data are not designed to assess potential risk factors leading to increases in psilocybin-related cases. Moreover, because reports to poison control centers are voluntary and don’t capture all exposures, NPDS data likely under-represent cases of hallucinogenic mushroom poisonings. Lastly, NPDS data are susceptible to reporting and misclassification biases.
DISCLOSURES:
Funding source was not disclosed. The authors reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Omalizumab for Food Allergies: What PCPs Should Know
Sandra Hong, MD, chair of allergy and immunology and director of the Food Allergy Center of Excellence at Cleveland Clinic, in Ohio, sees firsthand how situations that feel ordinary to most people strike fear in the hearts of her patients with food allergies.
Not only do some experience reactions to milk when they eat a cheese pizza — they can’t be in the same room with someone enjoying a slice nearby. “That would be terrifying,” Dr. Hong said.
Omalizumab (Xolair), recently approved by the US Food and Drug Administration as monotherapy for the treatment of food allergies, may now bring peace of mind to these patients and their families by reducing their risk of dangerous allergic reactions to accidental exposure.
While the drug does not cure food allergies, a phase 3, placebo-controlled trial found that after 16 weeks of treatment, two thirds of participants were able to tolerate at least 600 mg of peanut protein — equal to about 2.5 peanuts — without experiencing moderate to severe reactions.
An open-label extension trial also found the monoclonal antibody reduced the likelihood of serious reactions to eggs by 67%, milk by 66%, and cashews by 42%. The results of the study were published in The New England Journal of Medicine.
The treatment is approved for children as young as the age of 1 year and is the only treatment approved for multiple food allergies. It does not treat anaphylaxis or other emergency situations.
Patient Selection Key
While 8% of children and 10% of adults in the United States have a true food allergy, Brian Vickery, MD, chief of allergy and immunology and director of the Food Allergy Center at Children’s Healthcare of Atlanta, noted that a significantly higher proportion of the population restricts their diet based on perceived food intolerances.
“Most important for family doctors prior to prescribing the medication will be to be sure that the diagnosis is correct,” Kim said. “We know that allergy blood and skin testing is good but not perfect, and false positive results can occur,” said Edwin Kim, MD, chief of the Division of Pediatric Allergy and Immunology and director of the University of North Carolina Food Allergy Initiative at the University of North Carolina School of Medicine, Chapel Hill, who was a coauthor on the study in the New England Journal of Medicine. “ An allergist can conduct food challenges to confirm the diagnosis if results are unclear.”
Even for patients with confirmed IgE-mediated allergies, Dr. Hong said selecting patients who are good candidates for the therapy has “nuances.”
Patients must be willing and able to commit to injections every 2-4 weeks. Dosing depends on body weight and the total IgE levels of each patient. Patients with IgE levels > 1850 UI/mL likely will be disqualified from treatment since the clinical trial did not enroll patients with total IgE above this level and the appropriate dose in those patients is unknown.
“My recommendation for family physicians who are counseling food-allergic patients interested in omalizumab treatment is to partner with an allergist-immunologist, if at all possible,” Dr. Vickery said. He added that patients should have a comprehensive workup before beginning treatment because starting omalizumab would reduce reactivity and alter the outcome a diagnostic oral food challenge.
Two populations Dr. Hong thinks might particularly benefit from the therapy are college students and preschoolers, who may be unable to completely avoid allergens because of poor impulse control and food sharing in group settings.
“The concerns we have about this age group are whether or not there might be other factors involved that may impede their ability to make good decisions.”
Less control of the environment in dorms or other group living situations also could increase the risk of accidental exposure to a food allergen.
For the right patients, the treatment regimen has significant advantages over oral immunotherapy treatment (OIT), including the fact that it’s not a daily medication and it has the potential to treat allergic asthma at the same time.
“The biggest pro for omalizumab is that it can treat all of your food allergies, whether you have one or many, and do it all in one medication,” Dr. Kim said.
Managing Potential Harms
Omalizumab carries risks both primary care providers and patients must consider. First among them is that the drug carries a “black box” warning for an increased risk of anaphylaxis, Dr. Hong said.
Although patients with multiple food allergies typically already have prescriptions for epinephrine, primary care physicians (PCPs) considering offering omalizumab must be comfortable treating severe systemic reactions and their offices capable of post-dose monitoring, Dr. Hong said.
Anaphylaxis “can occur after the first dose or it can be delayed,” she said. “Typically, allergists will give these in our offices and we’ll actually have people wait for delayed amounts of time, for hours.”
The drug has been available since 2003 as a treatment for allergic asthma and urticaria. In addition to the warning for anaphylaxis, common reactions include joint pain and injection-site reactions. It also increases the risk for parasitic infection, and some studies show an increase in the risk for cancer.
Still, Dr. Kim said omalizumab’s safety profile is reassuring and noted it has advantages over OIT. “Since the patient is not exposing themselves to the food they are allergic to like in OIT, the safety is expected to be far better,” he said.
Lifelong Treatment
Dr. Vickery, Dr. Hong, and Dr. Kim all cautioned that patients should understand that, while omalizumab offers protection against accidental exposure and can meaningfully improve quality of life, it won’t allow them to loosen their allergen-avoidant diets.
Further, maintaining protection requires receiving injections every 2-4 weeks for life. For those without insurance, or whose insurance does not cover the treatment, costs could reach thousands of dollars each month, Dr. Hong said.
Omalizumab “has been well covered by insurance for asthma and chronic hives, but we will have to see what it looks like for food allergy. The range of plans and out-of-pocket deductibles available to patients will also play a big role,” Dr. Kim said.
Other novel approaches to food allergies are currently in clinical trials, and both Dr. Hong and Dr. Vickery are optimistic about potential options in the pipeline.
“We’re just on the brink of really exciting therapies coming forward in the future,” Dr. Hong said.
The study was supported by the National Institute of Allergy and Infectious Diseases and the National Center for Advancing Translational Sciences, both part of the National Institutes of Health; the Claudia and Steve Stange Family Fund; Genentech; and Novartis. Dr. Hong, Dr. Kim, and Dr. Vickery reported no relevant financial relationships.
A version of this article appeared on Medscape.com.
Sandra Hong, MD, chair of allergy and immunology and director of the Food Allergy Center of Excellence at Cleveland Clinic, in Ohio, sees firsthand how situations that feel ordinary to most people strike fear in the hearts of her patients with food allergies.
Not only do some experience reactions to milk when they eat a cheese pizza — they can’t be in the same room with someone enjoying a slice nearby. “That would be terrifying,” Dr. Hong said.
Omalizumab (Xolair), recently approved by the US Food and Drug Administration as monotherapy for the treatment of food allergies, may now bring peace of mind to these patients and their families by reducing their risk of dangerous allergic reactions to accidental exposure.
While the drug does not cure food allergies, a phase 3, placebo-controlled trial found that after 16 weeks of treatment, two thirds of participants were able to tolerate at least 600 mg of peanut protein — equal to about 2.5 peanuts — without experiencing moderate to severe reactions.
An open-label extension trial also found the monoclonal antibody reduced the likelihood of serious reactions to eggs by 67%, milk by 66%, and cashews by 42%. The results of the study were published in The New England Journal of Medicine.
The treatment is approved for children as young as the age of 1 year and is the only treatment approved for multiple food allergies. It does not treat anaphylaxis or other emergency situations.
Patient Selection Key
While 8% of children and 10% of adults in the United States have a true food allergy, Brian Vickery, MD, chief of allergy and immunology and director of the Food Allergy Center at Children’s Healthcare of Atlanta, noted that a significantly higher proportion of the population restricts their diet based on perceived food intolerances.
“Most important for family doctors prior to prescribing the medication will be to be sure that the diagnosis is correct,” Kim said. “We know that allergy blood and skin testing is good but not perfect, and false positive results can occur,” said Edwin Kim, MD, chief of the Division of Pediatric Allergy and Immunology and director of the University of North Carolina Food Allergy Initiative at the University of North Carolina School of Medicine, Chapel Hill, who was a coauthor on the study in the New England Journal of Medicine. “ An allergist can conduct food challenges to confirm the diagnosis if results are unclear.”
Even for patients with confirmed IgE-mediated allergies, Dr. Hong said selecting patients who are good candidates for the therapy has “nuances.”
Patients must be willing and able to commit to injections every 2-4 weeks. Dosing depends on body weight and the total IgE levels of each patient. Patients with IgE levels > 1850 UI/mL likely will be disqualified from treatment since the clinical trial did not enroll patients with total IgE above this level and the appropriate dose in those patients is unknown.
“My recommendation for family physicians who are counseling food-allergic patients interested in omalizumab treatment is to partner with an allergist-immunologist, if at all possible,” Dr. Vickery said. He added that patients should have a comprehensive workup before beginning treatment because starting omalizumab would reduce reactivity and alter the outcome a diagnostic oral food challenge.
Two populations Dr. Hong thinks might particularly benefit from the therapy are college students and preschoolers, who may be unable to completely avoid allergens because of poor impulse control and food sharing in group settings.
“The concerns we have about this age group are whether or not there might be other factors involved that may impede their ability to make good decisions.”
Less control of the environment in dorms or other group living situations also could increase the risk of accidental exposure to a food allergen.
For the right patients, the treatment regimen has significant advantages over oral immunotherapy treatment (OIT), including the fact that it’s not a daily medication and it has the potential to treat allergic asthma at the same time.
“The biggest pro for omalizumab is that it can treat all of your food allergies, whether you have one or many, and do it all in one medication,” Dr. Kim said.
Managing Potential Harms
Omalizumab carries risks both primary care providers and patients must consider. First among them is that the drug carries a “black box” warning for an increased risk of anaphylaxis, Dr. Hong said.
Although patients with multiple food allergies typically already have prescriptions for epinephrine, primary care physicians (PCPs) considering offering omalizumab must be comfortable treating severe systemic reactions and their offices capable of post-dose monitoring, Dr. Hong said.
Anaphylaxis “can occur after the first dose or it can be delayed,” she said. “Typically, allergists will give these in our offices and we’ll actually have people wait for delayed amounts of time, for hours.”
The drug has been available since 2003 as a treatment for allergic asthma and urticaria. In addition to the warning for anaphylaxis, common reactions include joint pain and injection-site reactions. It also increases the risk for parasitic infection, and some studies show an increase in the risk for cancer.
Still, Dr. Kim said omalizumab’s safety profile is reassuring and noted it has advantages over OIT. “Since the patient is not exposing themselves to the food they are allergic to like in OIT, the safety is expected to be far better,” he said.
Lifelong Treatment
Dr. Vickery, Dr. Hong, and Dr. Kim all cautioned that patients should understand that, while omalizumab offers protection against accidental exposure and can meaningfully improve quality of life, it won’t allow them to loosen their allergen-avoidant diets.
Further, maintaining protection requires receiving injections every 2-4 weeks for life. For those without insurance, or whose insurance does not cover the treatment, costs could reach thousands of dollars each month, Dr. Hong said.
Omalizumab “has been well covered by insurance for asthma and chronic hives, but we will have to see what it looks like for food allergy. The range of plans and out-of-pocket deductibles available to patients will also play a big role,” Dr. Kim said.
Other novel approaches to food allergies are currently in clinical trials, and both Dr. Hong and Dr. Vickery are optimistic about potential options in the pipeline.
“We’re just on the brink of really exciting therapies coming forward in the future,” Dr. Hong said.
The study was supported by the National Institute of Allergy and Infectious Diseases and the National Center for Advancing Translational Sciences, both part of the National Institutes of Health; the Claudia and Steve Stange Family Fund; Genentech; and Novartis. Dr. Hong, Dr. Kim, and Dr. Vickery reported no relevant financial relationships.
A version of this article appeared on Medscape.com.
Sandra Hong, MD, chair of allergy and immunology and director of the Food Allergy Center of Excellence at Cleveland Clinic, in Ohio, sees firsthand how situations that feel ordinary to most people strike fear in the hearts of her patients with food allergies.
Not only do some experience reactions to milk when they eat a cheese pizza — they can’t be in the same room with someone enjoying a slice nearby. “That would be terrifying,” Dr. Hong said.
Omalizumab (Xolair), recently approved by the US Food and Drug Administration as monotherapy for the treatment of food allergies, may now bring peace of mind to these patients and their families by reducing their risk of dangerous allergic reactions to accidental exposure.
While the drug does not cure food allergies, a phase 3, placebo-controlled trial found that after 16 weeks of treatment, two thirds of participants were able to tolerate at least 600 mg of peanut protein — equal to about 2.5 peanuts — without experiencing moderate to severe reactions.
An open-label extension trial also found the monoclonal antibody reduced the likelihood of serious reactions to eggs by 67%, milk by 66%, and cashews by 42%. The results of the study were published in The New England Journal of Medicine.
The treatment is approved for children as young as the age of 1 year and is the only treatment approved for multiple food allergies. It does not treat anaphylaxis or other emergency situations.
Patient Selection Key
While 8% of children and 10% of adults in the United States have a true food allergy, Brian Vickery, MD, chief of allergy and immunology and director of the Food Allergy Center at Children’s Healthcare of Atlanta, noted that a significantly higher proportion of the population restricts their diet based on perceived food intolerances.
“Most important for family doctors prior to prescribing the medication will be to be sure that the diagnosis is correct,” Kim said. “We know that allergy blood and skin testing is good but not perfect, and false positive results can occur,” said Edwin Kim, MD, chief of the Division of Pediatric Allergy and Immunology and director of the University of North Carolina Food Allergy Initiative at the University of North Carolina School of Medicine, Chapel Hill, who was a coauthor on the study in the New England Journal of Medicine. “ An allergist can conduct food challenges to confirm the diagnosis if results are unclear.”
Even for patients with confirmed IgE-mediated allergies, Dr. Hong said selecting patients who are good candidates for the therapy has “nuances.”
Patients must be willing and able to commit to injections every 2-4 weeks. Dosing depends on body weight and the total IgE levels of each patient. Patients with IgE levels > 1850 UI/mL likely will be disqualified from treatment since the clinical trial did not enroll patients with total IgE above this level and the appropriate dose in those patients is unknown.
“My recommendation for family physicians who are counseling food-allergic patients interested in omalizumab treatment is to partner with an allergist-immunologist, if at all possible,” Dr. Vickery said. He added that patients should have a comprehensive workup before beginning treatment because starting omalizumab would reduce reactivity and alter the outcome a diagnostic oral food challenge.
Two populations Dr. Hong thinks might particularly benefit from the therapy are college students and preschoolers, who may be unable to completely avoid allergens because of poor impulse control and food sharing in group settings.
“The concerns we have about this age group are whether or not there might be other factors involved that may impede their ability to make good decisions.”
Less control of the environment in dorms or other group living situations also could increase the risk of accidental exposure to a food allergen.
For the right patients, the treatment regimen has significant advantages over oral immunotherapy treatment (OIT), including the fact that it’s not a daily medication and it has the potential to treat allergic asthma at the same time.
“The biggest pro for omalizumab is that it can treat all of your food allergies, whether you have one or many, and do it all in one medication,” Dr. Kim said.
Managing Potential Harms
Omalizumab carries risks both primary care providers and patients must consider. First among them is that the drug carries a “black box” warning for an increased risk of anaphylaxis, Dr. Hong said.
Although patients with multiple food allergies typically already have prescriptions for epinephrine, primary care physicians (PCPs) considering offering omalizumab must be comfortable treating severe systemic reactions and their offices capable of post-dose monitoring, Dr. Hong said.
Anaphylaxis “can occur after the first dose or it can be delayed,” she said. “Typically, allergists will give these in our offices and we’ll actually have people wait for delayed amounts of time, for hours.”
The drug has been available since 2003 as a treatment for allergic asthma and urticaria. In addition to the warning for anaphylaxis, common reactions include joint pain and injection-site reactions. It also increases the risk for parasitic infection, and some studies show an increase in the risk for cancer.
Still, Dr. Kim said omalizumab’s safety profile is reassuring and noted it has advantages over OIT. “Since the patient is not exposing themselves to the food they are allergic to like in OIT, the safety is expected to be far better,” he said.
Lifelong Treatment
Dr. Vickery, Dr. Hong, and Dr. Kim all cautioned that patients should understand that, while omalizumab offers protection against accidental exposure and can meaningfully improve quality of life, it won’t allow them to loosen their allergen-avoidant diets.
Further, maintaining protection requires receiving injections every 2-4 weeks for life. For those without insurance, or whose insurance does not cover the treatment, costs could reach thousands of dollars each month, Dr. Hong said.
Omalizumab “has been well covered by insurance for asthma and chronic hives, but we will have to see what it looks like for food allergy. The range of plans and out-of-pocket deductibles available to patients will also play a big role,” Dr. Kim said.
Other novel approaches to food allergies are currently in clinical trials, and both Dr. Hong and Dr. Vickery are optimistic about potential options in the pipeline.
“We’re just on the brink of really exciting therapies coming forward in the future,” Dr. Hong said.
The study was supported by the National Institute of Allergy and Infectious Diseases and the National Center for Advancing Translational Sciences, both part of the National Institutes of Health; the Claudia and Steve Stange Family Fund; Genentech; and Novartis. Dr. Hong, Dr. Kim, and Dr. Vickery reported no relevant financial relationships.
A version of this article appeared on Medscape.com.
Adding Antithrombotic to tPA Does Not Improve Stroke Outcome
PHOENIX — , results of new research show.
“Ultimately, we found no benefit for either medication added to standard-of-care thrombolysis in terms of improving stroke outcomes,” said lead study author Opeolu M. Adeoye, MD, professor of emergency medicine and department chair, Washington University School of Medicine, St. Louis, Missouri.
The results were surprising and disappointing for Dr. Adeoye. “We went into the trial hopeful and thinking we would be able to benefit patients in reducing disability from stroke,” he said.
The Multi-Arm Optimization of Stroke Thrombolysis (MOST) trial was stopped early because of futility following recommendations from the data and safety monitoring board.
The findings were presented at the International Stroke Conference presented by the American Stroke Association, a division of the American Heart Association.
A thrombolytic drug alone doesn’t help all patients, particularly those with larger clots. “Clots can open; they can reform; they can re-occlude, etc.,” said another author, Andrew D. Barreto, MD, associate professor, Department of Neurology, University of Texas Health Science Center, Houston. “The thought was that adding additional medications that thin the blood, like argatroban or eptifibatide, would amplify the effects of the clot-busting drug.”
Indeed, this approach has had success in cardiology in terms of blood vessel opening, said Dr. Adeoye, adding that some preclinical data suggest that antithrombotic drugs may be neuroprotective.
Six phase 2 studies going back over a dozen years suggested that these drugs are safe in stroke patients. Although these studies weren’t powered for efficacy, “we did see a signal that adding them would be better than just the clot-busting drug alone.” These findings prompted the current phase 3 trial, said Dr. Barreto.
The three-arm, single-blind MOST trial included 514 adult patients with acute ischemic stroke and a National Institutes of Health Stroke Scale (NIHSS) score of 6 or greater at 57 US centers. In the study cohort the mean age was about 68 years, 70% White/25% Black, and with about equal numbers of female and male patients.
All received standard stroke care including thrombolysis within 3 hours of symptom onset. Initially, researchers used intravenous alteplase (0.9 mg/kg), but as the standard of care changed over time, they began using tenecteplase (0.25 mg/kg).
Study patients were also randomly assigned to receive placebo or either argatroban (100 mcg/kg bolus followed by 3 mcg/kg per minute for 12 hours) or eptifibatide (135 mcg/kg bolus followed by 0.75 mcg/kg/min infusion for 2 hours). These treatments were initiated within 75 minutes of thrombolysis.
Two Different Mechanisms
The drugs have different mechanisms of action. Argatroban is an anticoagulant, a direct inhibitor of thrombin, while the antiplatelet eptifibatide blocks the glycoprotein IIb/IIIa receptor and was specifically developed to ensure rapid inhibition of platelet aggregation.
Patients could also receive endovascular thrombectomy as part of their usual care. In this study, about 44% of patients received this treatment.
The primary endpoint was 90-day utility weighted modified Rankin Scale (uw-mRS) scores, where the worst outcome is 0 and the best outcome is 10.
The study used a response-adaptive randomization design, where the randomization switches from a drug that doesn’t appear to have a chance of working to the arm more likely to be beneficial.
Of the 514 patients, the analysis included 228 in the placebo, 59 in the argatroban, and 227 in eptifibatide groups. Of the total, 421 completed the study.
The mean 90-day uw-mRS was 6.8 in the placebo group, 5.2 in the argatroban group, and 6.3 in the eptifibatide group.
The probability of argatroban being better than placebo was 0.2%; the probability of eptifibatide being better than placebo was 0.9%. The futility threshold was enrollment of 500 and less than a 20% chance of benefit, thus the decision to stop the trial.
In all subgroup analyses, which looked at age, stroke severity, the two thrombolytic drugs, and use of endovascular therapy, “we didn’t really see much of a signal that would suggest that’s the group we would need to be testing further,” said Dr. Barreto.
No Increased ICH Risk
The primary safety outcome was symptomatic intracranial hemorrhage within 36 hours of randomization. The researchers found no significant increase in rates of this outcome.
The argatroban cohort had significantly lower odds of favorable outcomes compared with placebo, noted Dr. Adeoye. For example, it had more all-cause deaths, although none were related to the study drug.
Speculating on why the intervention didn’t work, Dr. Barreto pointed to changes in standard of care between the earlier trials and the current one, including the incorporation of endovascular therapy and switch to tenecteplase.
Although the results were disappointing, Dr. Adeoye sees a bright side. “What we’re very proud of, and excited about, is the fact that we have a definitive answer on these two drugs, and we did it in one trial as opposed to sequential, separate ongoing trials.”
But he stressed that more work needs to be done, especially given that even with endovascular therapy, half of stroke patients don’t achieve independence.
“In this trial, we established that argatroban and eptifibatide added to thrombolysis did not work, but that doesn’t address the fact that we need to continue to see what we can do to improve the total proportion of stroke patients who, after our treatments, are functionally independent 90 days after the stroke.”
Down the Rabbit Hole
Commenting on the research, Larry B. Goldstein, MD, professor and chair, Department of Neurology, University of Kentucky, Lexington, praised the study’s adaptive design, noted that the hypothesis the study was based on was “reasonable” given the concern about additional thromboses, and found the results useful.
“The goal is not only to see what works but also what doesn’t work so we don’t go down that rabbit hole.”
He also pointed out that because the two blood-thinning drugs studied have very different mechanisms of action, it’s unlikely that another antithrombotic would add benefit to thrombolysis, “but you never say never.”
Dr. Adeoye and Dr. Barreto report research funding from the National Institutes of Health/National Institute of Neurological Disorders and Stroke. Dr. Adeoye also reports an executive role, receiving royalties/being a patent beneficiary, Sense Diagnostics, Inc. Dr. Goldstein has no relevant conflicts of interest.
A version of this article appeared on Medscape.com.
PHOENIX — , results of new research show.
“Ultimately, we found no benefit for either medication added to standard-of-care thrombolysis in terms of improving stroke outcomes,” said lead study author Opeolu M. Adeoye, MD, professor of emergency medicine and department chair, Washington University School of Medicine, St. Louis, Missouri.
The results were surprising and disappointing for Dr. Adeoye. “We went into the trial hopeful and thinking we would be able to benefit patients in reducing disability from stroke,” he said.
The Multi-Arm Optimization of Stroke Thrombolysis (MOST) trial was stopped early because of futility following recommendations from the data and safety monitoring board.
The findings were presented at the International Stroke Conference presented by the American Stroke Association, a division of the American Heart Association.
A thrombolytic drug alone doesn’t help all patients, particularly those with larger clots. “Clots can open; they can reform; they can re-occlude, etc.,” said another author, Andrew D. Barreto, MD, associate professor, Department of Neurology, University of Texas Health Science Center, Houston. “The thought was that adding additional medications that thin the blood, like argatroban or eptifibatide, would amplify the effects of the clot-busting drug.”
Indeed, this approach has had success in cardiology in terms of blood vessel opening, said Dr. Adeoye, adding that some preclinical data suggest that antithrombotic drugs may be neuroprotective.
Six phase 2 studies going back over a dozen years suggested that these drugs are safe in stroke patients. Although these studies weren’t powered for efficacy, “we did see a signal that adding them would be better than just the clot-busting drug alone.” These findings prompted the current phase 3 trial, said Dr. Barreto.
The three-arm, single-blind MOST trial included 514 adult patients with acute ischemic stroke and a National Institutes of Health Stroke Scale (NIHSS) score of 6 or greater at 57 US centers. In the study cohort the mean age was about 68 years, 70% White/25% Black, and with about equal numbers of female and male patients.
All received standard stroke care including thrombolysis within 3 hours of symptom onset. Initially, researchers used intravenous alteplase (0.9 mg/kg), but as the standard of care changed over time, they began using tenecteplase (0.25 mg/kg).
Study patients were also randomly assigned to receive placebo or either argatroban (100 mcg/kg bolus followed by 3 mcg/kg per minute for 12 hours) or eptifibatide (135 mcg/kg bolus followed by 0.75 mcg/kg/min infusion for 2 hours). These treatments were initiated within 75 minutes of thrombolysis.
Two Different Mechanisms
The drugs have different mechanisms of action. Argatroban is an anticoagulant, a direct inhibitor of thrombin, while the antiplatelet eptifibatide blocks the glycoprotein IIb/IIIa receptor and was specifically developed to ensure rapid inhibition of platelet aggregation.
Patients could also receive endovascular thrombectomy as part of their usual care. In this study, about 44% of patients received this treatment.
The primary endpoint was 90-day utility weighted modified Rankin Scale (uw-mRS) scores, where the worst outcome is 0 and the best outcome is 10.
The study used a response-adaptive randomization design, where the randomization switches from a drug that doesn’t appear to have a chance of working to the arm more likely to be beneficial.
Of the 514 patients, the analysis included 228 in the placebo, 59 in the argatroban, and 227 in eptifibatide groups. Of the total, 421 completed the study.
The mean 90-day uw-mRS was 6.8 in the placebo group, 5.2 in the argatroban group, and 6.3 in the eptifibatide group.
The probability of argatroban being better than placebo was 0.2%; the probability of eptifibatide being better than placebo was 0.9%. The futility threshold was enrollment of 500 and less than a 20% chance of benefit, thus the decision to stop the trial.
In all subgroup analyses, which looked at age, stroke severity, the two thrombolytic drugs, and use of endovascular therapy, “we didn’t really see much of a signal that would suggest that’s the group we would need to be testing further,” said Dr. Barreto.
No Increased ICH Risk
The primary safety outcome was symptomatic intracranial hemorrhage within 36 hours of randomization. The researchers found no significant increase in rates of this outcome.
The argatroban cohort had significantly lower odds of favorable outcomes compared with placebo, noted Dr. Adeoye. For example, it had more all-cause deaths, although none were related to the study drug.
Speculating on why the intervention didn’t work, Dr. Barreto pointed to changes in standard of care between the earlier trials and the current one, including the incorporation of endovascular therapy and switch to tenecteplase.
Although the results were disappointing, Dr. Adeoye sees a bright side. “What we’re very proud of, and excited about, is the fact that we have a definitive answer on these two drugs, and we did it in one trial as opposed to sequential, separate ongoing trials.”
But he stressed that more work needs to be done, especially given that even with endovascular therapy, half of stroke patients don’t achieve independence.
“In this trial, we established that argatroban and eptifibatide added to thrombolysis did not work, but that doesn’t address the fact that we need to continue to see what we can do to improve the total proportion of stroke patients who, after our treatments, are functionally independent 90 days after the stroke.”
Down the Rabbit Hole
Commenting on the research, Larry B. Goldstein, MD, professor and chair, Department of Neurology, University of Kentucky, Lexington, praised the study’s adaptive design, noted that the hypothesis the study was based on was “reasonable” given the concern about additional thromboses, and found the results useful.
“The goal is not only to see what works but also what doesn’t work so we don’t go down that rabbit hole.”
He also pointed out that because the two blood-thinning drugs studied have very different mechanisms of action, it’s unlikely that another antithrombotic would add benefit to thrombolysis, “but you never say never.”
Dr. Adeoye and Dr. Barreto report research funding from the National Institutes of Health/National Institute of Neurological Disorders and Stroke. Dr. Adeoye also reports an executive role, receiving royalties/being a patent beneficiary, Sense Diagnostics, Inc. Dr. Goldstein has no relevant conflicts of interest.
A version of this article appeared on Medscape.com.
PHOENIX — , results of new research show.
“Ultimately, we found no benefit for either medication added to standard-of-care thrombolysis in terms of improving stroke outcomes,” said lead study author Opeolu M. Adeoye, MD, professor of emergency medicine and department chair, Washington University School of Medicine, St. Louis, Missouri.
The results were surprising and disappointing for Dr. Adeoye. “We went into the trial hopeful and thinking we would be able to benefit patients in reducing disability from stroke,” he said.
The Multi-Arm Optimization of Stroke Thrombolysis (MOST) trial was stopped early because of futility following recommendations from the data and safety monitoring board.
The findings were presented at the International Stroke Conference presented by the American Stroke Association, a division of the American Heart Association.
A thrombolytic drug alone doesn’t help all patients, particularly those with larger clots. “Clots can open; they can reform; they can re-occlude, etc.,” said another author, Andrew D. Barreto, MD, associate professor, Department of Neurology, University of Texas Health Science Center, Houston. “The thought was that adding additional medications that thin the blood, like argatroban or eptifibatide, would amplify the effects of the clot-busting drug.”
Indeed, this approach has had success in cardiology in terms of blood vessel opening, said Dr. Adeoye, adding that some preclinical data suggest that antithrombotic drugs may be neuroprotective.
Six phase 2 studies going back over a dozen years suggested that these drugs are safe in stroke patients. Although these studies weren’t powered for efficacy, “we did see a signal that adding them would be better than just the clot-busting drug alone.” These findings prompted the current phase 3 trial, said Dr. Barreto.
The three-arm, single-blind MOST trial included 514 adult patients with acute ischemic stroke and a National Institutes of Health Stroke Scale (NIHSS) score of 6 or greater at 57 US centers. In the study cohort the mean age was about 68 years, 70% White/25% Black, and with about equal numbers of female and male patients.
All received standard stroke care including thrombolysis within 3 hours of symptom onset. Initially, researchers used intravenous alteplase (0.9 mg/kg), but as the standard of care changed over time, they began using tenecteplase (0.25 mg/kg).
Study patients were also randomly assigned to receive placebo or either argatroban (100 mcg/kg bolus followed by 3 mcg/kg per minute for 12 hours) or eptifibatide (135 mcg/kg bolus followed by 0.75 mcg/kg/min infusion for 2 hours). These treatments were initiated within 75 minutes of thrombolysis.
Two Different Mechanisms
The drugs have different mechanisms of action. Argatroban is an anticoagulant, a direct inhibitor of thrombin, while the antiplatelet eptifibatide blocks the glycoprotein IIb/IIIa receptor and was specifically developed to ensure rapid inhibition of platelet aggregation.
Patients could also receive endovascular thrombectomy as part of their usual care. In this study, about 44% of patients received this treatment.
The primary endpoint was 90-day utility weighted modified Rankin Scale (uw-mRS) scores, where the worst outcome is 0 and the best outcome is 10.
The study used a response-adaptive randomization design, where the randomization switches from a drug that doesn’t appear to have a chance of working to the arm more likely to be beneficial.
Of the 514 patients, the analysis included 228 in the placebo, 59 in the argatroban, and 227 in eptifibatide groups. Of the total, 421 completed the study.
The mean 90-day uw-mRS was 6.8 in the placebo group, 5.2 in the argatroban group, and 6.3 in the eptifibatide group.
The probability of argatroban being better than placebo was 0.2%; the probability of eptifibatide being better than placebo was 0.9%. The futility threshold was enrollment of 500 and less than a 20% chance of benefit, thus the decision to stop the trial.
In all subgroup analyses, which looked at age, stroke severity, the two thrombolytic drugs, and use of endovascular therapy, “we didn’t really see much of a signal that would suggest that’s the group we would need to be testing further,” said Dr. Barreto.
No Increased ICH Risk
The primary safety outcome was symptomatic intracranial hemorrhage within 36 hours of randomization. The researchers found no significant increase in rates of this outcome.
The argatroban cohort had significantly lower odds of favorable outcomes compared with placebo, noted Dr. Adeoye. For example, it had more all-cause deaths, although none were related to the study drug.
Speculating on why the intervention didn’t work, Dr. Barreto pointed to changes in standard of care between the earlier trials and the current one, including the incorporation of endovascular therapy and switch to tenecteplase.
Although the results were disappointing, Dr. Adeoye sees a bright side. “What we’re very proud of, and excited about, is the fact that we have a definitive answer on these two drugs, and we did it in one trial as opposed to sequential, separate ongoing trials.”
But he stressed that more work needs to be done, especially given that even with endovascular therapy, half of stroke patients don’t achieve independence.
“In this trial, we established that argatroban and eptifibatide added to thrombolysis did not work, but that doesn’t address the fact that we need to continue to see what we can do to improve the total proportion of stroke patients who, after our treatments, are functionally independent 90 days after the stroke.”
Down the Rabbit Hole
Commenting on the research, Larry B. Goldstein, MD, professor and chair, Department of Neurology, University of Kentucky, Lexington, praised the study’s adaptive design, noted that the hypothesis the study was based on was “reasonable” given the concern about additional thromboses, and found the results useful.
“The goal is not only to see what works but also what doesn’t work so we don’t go down that rabbit hole.”
He also pointed out that because the two blood-thinning drugs studied have very different mechanisms of action, it’s unlikely that another antithrombotic would add benefit to thrombolysis, “but you never say never.”
Dr. Adeoye and Dr. Barreto report research funding from the National Institutes of Health/National Institute of Neurological Disorders and Stroke. Dr. Adeoye also reports an executive role, receiving royalties/being a patent beneficiary, Sense Diagnostics, Inc. Dr. Goldstein has no relevant conflicts of interest.
A version of this article appeared on Medscape.com.
FROM ISC 2024
Expanded Window of Stroke Thrombectomy With Simpler Imaging
PHOENIX — Thrombectomy is generally beneficial for patients from a low-income population who have a large vessel occlusion stroke presenting in the later time window and who can be identified as suitable for treatment without the need for advanced and costly imaging, a new Brazilian trial has shown.
“The RESILIENT-Extend trial is the first major study of thrombectomy in the late time window (8-24 h) conducted outside first-world countries and shows the procedure also has benefit in a lower socioeconomic status population without the need for costly imaging equipment,” said lead investigator Raul G. Nogueira, MD.
“The trial expands the treatment window for thrombectomy globally with simplified selection criteria based on non-contrast CT, potentially altering current guidelines,” Dr. Nogueira said.
However, there were some caveats that need to be considered; in particular, a lack of benefit with thrombectomy in older patients (over 68 years of age), which Dr. Nogueira believes is a reflection of the particular population enrolled in this study. Specifically, he suggested that older age in this low socioeconomic status population is a surrogate for frailty, and the study may have identified frailty as a factor that correlates with reduced or lack of benefit of thrombectomy.
Dr. Nogueira, who is a professor of neurology and neurosurgery at the University of Pittsburgh, and Sheila Martins, MD, a professor of neurology at Hospital de Clinicas Porto Alegre in Brazil, presented the RESILIENT-Extend results at the International Stroke Conference presented by the American Stroke Association, a division of the American Heart Association.
Dr. Nogueira explained that the lack of available advanced imaging techniques is a major challenge for implementing endovascular therapy in an extended time window, especially in lower-income countries.
“Our main objective was to see if we could remove the need for advanced imaging to select patients with large vessel occlusion stroke in the late time window (8-24 h) for thrombectomy,” he said. “In this way, our trial overlaps somewhat with the MR CLEAN-LATE Trial conducted in the Netherlands, although the two trials were conducted in very different socioeconomic populations.”
The RESILIENT-Extend trial was conducted in the public health service of Brazil and involved a different population of people than have been included in other thrombectomy trials, which have mostly been conducted in first-world countries.
“The public health system in Brazil is not well-resourced and tends to care for patients at lower socioeconomic levels. These patients are fundamentally different from the average patients in the first-world recruited into most other thrombectomy trials,” Dr. Nogueira noted.
The trial enrolled 245 patients with a large vessel occlusion stroke within 8-24 hours of last known well. Patients were included who had a mismatch between the clinical severity as shown by the National Institutes of Health Stroke Scale (NIHSS) score and the stroke burden on imaging as measured by ASPECTS scores.
They had to have relatively high NIHSS scores (8 or more) showing more severe strokes but also a high ASPECTS score (5-10) excluding patients with large areas of ischemic brain. There was also a sliding scale that adjusted for age to avoid enrolling elderly patients with large strokes.
These patients were identified exclusively using non-contrast CT and CT angiography imaging.
The median age of patients included was 62-63 years. Dr. Nogueira pointed out that patients were slightly younger than seen in other thrombectomy trials, perhaps because in lower-middle-income countries strokes occur at a younger age. They also have a higher case fatality rate.
The median baseline NIHSS score was 16, and the median ASPECTS score was 7-8.
The median time to treatment was 12.5 hours, which is similar to other late window thrombectomy trials.
Conflicting Results on Shift Analysis
The primary outcome was a shift analysis of the modified Rankin Scale (mRS) disability score at 90 days.
This showed a bidirectional result, with thrombectomy increasing the chances of a good or excellent outcome (mRS, 0-3), but there was also a nonsignificant increased risk for a bad outcome (mRS, 5-6).
“This bidirectional result prevents a common odds ratio from being calculated, so the primary endpoint is not applicable,” Dr. Nogueira reported.
The researchers therefore used the secondary outcomes as the main results of the study.
These showed that the number of patients achieving a good outcome (mRS, 0-2) was significantly increased with thrombectomy (25% vs. 14%, adjusted odds ratio, 2.56; P = .012).
The number of patients achieving an excellent outcome (mRS, 0-1) was also significantly increased.
But these increases in good outcomes came at the cost of some patients having an increased risk for severe disability or death (mRS, 5-6).
The odds ratio for an mRS of 0-4 versus 5-6 was 0.71, and for an mRS of 0-5 versus 6, the odds ratio was 0.58. Both these results were nonsignificant.
Another anomaly in the RESILIENT-Extend trial was the observation of no benefit of thrombectomy seen in older patients.
“In general, trials of thrombectomy in the first world have shown a greater treatment effect in older patients, but this was not seen in our trial, where older patients (over 68 years) did not derive any benefit from the procedure,” Dr. Nogueira noted.
A similar observation was also seen in the first RESILIENT trial in patients treated within 8 hours of stroke onset, which was also conducted in Brazil, leading to the suggestion that it is related to the patient population included.
“In the Brazilian public health service, older patients are very vulnerable and frail. They are different to older patients in first world countries. It appears they may be too fragile to withstand the thrombectomy process,” Dr. Nogueira said.
Frailty: A Ceiling Effect?
Results from the two RESILIENT trials give a word of caution to the thrombectomy field, Dr. Nogueira said.
“This procedure was initially thought suitable only for patients with small core strokes, but we now have a series of trials showing benefit of thrombectomy in large core strokes as well,” Dr. Nogueira said. “We have started to believe that this intervention will benefit almost all patients with large vessel occlusion stroke everywhere around the world, but our data suggest that we have to consider the specific populations that we are serving and that factors such as socioeconomic status and frailty have to be taken into account.
“Both the RESILIENT trials have shown that thrombectomy does not appear to be suitable for older patients, over 68-70 years of age, in the public health service in Brazil,” he noted. “In this population, a patient aged 70 can be quite different to a patient of the same age in a first-world country. I think in our population, an age of over 68-70 is a surrogate for frailty, which will not be the case in first-world countries. In this regard, I think we have found a ceiling effect for benefit of thrombectomy, which is frailty.”
Dr. Nogueira speculated that the bidirectional effect on the mRS shift analysis may also have been caused by the frailty of some of the patients.
“What the results may be showing is that for most of the population, there is a benefit of thrombectomy, but for some patients, possibly the most frail, then the procedure can be too overwhelming for them. But the suggestion of harm was not significant, so this observation could have also just been the play of chance,” he added.
Interpreting the Findings
Commenting on the RESILIENT-Extend study results, Michael Hill, MD, professor of neurology at the University of Calgary, Canada, pointed out that there was an absolute benefit of 11.1% on the mRS of 0-2 outcome but a similar signal of harm, with a 10.2% increase in mortality in the thrombectomy group, although that was not statistically significant.
“This signal of harm appears not to be due to an increase in intracranial hemorrhage or procedural mishap,” he said. “It is unclear why there were more deaths; the overall trial numbers are small enough that this could be a chance finding.”
Dr. Hill also noted that the absolute proportion of patients achieving an independent functional outcome was 50% less than in the DAWN trial of thrombectomy in the extended window. “This tells us that the patients selected for inclusion into RESILIENT-Extend were physiologically different from those in DAWN,” he said.
Also commenting on the study, Amrou Sarraj, MD, professor of neurology at University Hospitals Cleveland Medical Center–Case Western Reserve University in Cleveland, said: “The RESILIENT-Extend investigators should be congratulated for the successful conduct of the trial and providing evidence of benefit of thrombectomy procedure with simplified neuroimaging protocol using CT and CTA in resource-limited settings. These findings will help support extending the access to thrombectomy in areas without availability of advanced imaging.”
He said the bidirectional effect on the primary endpoint and the positive interaction between age and thrombectomy treatment effect warranted further investigation.
The RESILIENT-Extend trial was sponsored by the Brazilian Ministry of Health.
A version of this article appeared on Medscape.com.
PHOENIX — Thrombectomy is generally beneficial for patients from a low-income population who have a large vessel occlusion stroke presenting in the later time window and who can be identified as suitable for treatment without the need for advanced and costly imaging, a new Brazilian trial has shown.
“The RESILIENT-Extend trial is the first major study of thrombectomy in the late time window (8-24 h) conducted outside first-world countries and shows the procedure also has benefit in a lower socioeconomic status population without the need for costly imaging equipment,” said lead investigator Raul G. Nogueira, MD.
“The trial expands the treatment window for thrombectomy globally with simplified selection criteria based on non-contrast CT, potentially altering current guidelines,” Dr. Nogueira said.
However, there were some caveats that need to be considered; in particular, a lack of benefit with thrombectomy in older patients (over 68 years of age), which Dr. Nogueira believes is a reflection of the particular population enrolled in this study. Specifically, he suggested that older age in this low socioeconomic status population is a surrogate for frailty, and the study may have identified frailty as a factor that correlates with reduced or lack of benefit of thrombectomy.
Dr. Nogueira, who is a professor of neurology and neurosurgery at the University of Pittsburgh, and Sheila Martins, MD, a professor of neurology at Hospital de Clinicas Porto Alegre in Brazil, presented the RESILIENT-Extend results at the International Stroke Conference presented by the American Stroke Association, a division of the American Heart Association.
Dr. Nogueira explained that the lack of available advanced imaging techniques is a major challenge for implementing endovascular therapy in an extended time window, especially in lower-income countries.
“Our main objective was to see if we could remove the need for advanced imaging to select patients with large vessel occlusion stroke in the late time window (8-24 h) for thrombectomy,” he said. “In this way, our trial overlaps somewhat with the MR CLEAN-LATE Trial conducted in the Netherlands, although the two trials were conducted in very different socioeconomic populations.”
The RESILIENT-Extend trial was conducted in the public health service of Brazil and involved a different population of people than have been included in other thrombectomy trials, which have mostly been conducted in first-world countries.
“The public health system in Brazil is not well-resourced and tends to care for patients at lower socioeconomic levels. These patients are fundamentally different from the average patients in the first-world recruited into most other thrombectomy trials,” Dr. Nogueira noted.
The trial enrolled 245 patients with a large vessel occlusion stroke within 8-24 hours of last known well. Patients were included who had a mismatch between the clinical severity as shown by the National Institutes of Health Stroke Scale (NIHSS) score and the stroke burden on imaging as measured by ASPECTS scores.
They had to have relatively high NIHSS scores (8 or more) showing more severe strokes but also a high ASPECTS score (5-10) excluding patients with large areas of ischemic brain. There was also a sliding scale that adjusted for age to avoid enrolling elderly patients with large strokes.
These patients were identified exclusively using non-contrast CT and CT angiography imaging.
The median age of patients included was 62-63 years. Dr. Nogueira pointed out that patients were slightly younger than seen in other thrombectomy trials, perhaps because in lower-middle-income countries strokes occur at a younger age. They also have a higher case fatality rate.
The median baseline NIHSS score was 16, and the median ASPECTS score was 7-8.
The median time to treatment was 12.5 hours, which is similar to other late window thrombectomy trials.
Conflicting Results on Shift Analysis
The primary outcome was a shift analysis of the modified Rankin Scale (mRS) disability score at 90 days.
This showed a bidirectional result, with thrombectomy increasing the chances of a good or excellent outcome (mRS, 0-3), but there was also a nonsignificant increased risk for a bad outcome (mRS, 5-6).
“This bidirectional result prevents a common odds ratio from being calculated, so the primary endpoint is not applicable,” Dr. Nogueira reported.
The researchers therefore used the secondary outcomes as the main results of the study.
These showed that the number of patients achieving a good outcome (mRS, 0-2) was significantly increased with thrombectomy (25% vs. 14%, adjusted odds ratio, 2.56; P = .012).
The number of patients achieving an excellent outcome (mRS, 0-1) was also significantly increased.
But these increases in good outcomes came at the cost of some patients having an increased risk for severe disability or death (mRS, 5-6).
The odds ratio for an mRS of 0-4 versus 5-6 was 0.71, and for an mRS of 0-5 versus 6, the odds ratio was 0.58. Both these results were nonsignificant.
Another anomaly in the RESILIENT-Extend trial was the observation of no benefit of thrombectomy seen in older patients.
“In general, trials of thrombectomy in the first world have shown a greater treatment effect in older patients, but this was not seen in our trial, where older patients (over 68 years) did not derive any benefit from the procedure,” Dr. Nogueira noted.
A similar observation was also seen in the first RESILIENT trial in patients treated within 8 hours of stroke onset, which was also conducted in Brazil, leading to the suggestion that it is related to the patient population included.
“In the Brazilian public health service, older patients are very vulnerable and frail. They are different to older patients in first world countries. It appears they may be too fragile to withstand the thrombectomy process,” Dr. Nogueira said.
Frailty: A Ceiling Effect?
Results from the two RESILIENT trials give a word of caution to the thrombectomy field, Dr. Nogueira said.
“This procedure was initially thought suitable only for patients with small core strokes, but we now have a series of trials showing benefit of thrombectomy in large core strokes as well,” Dr. Nogueira said. “We have started to believe that this intervention will benefit almost all patients with large vessel occlusion stroke everywhere around the world, but our data suggest that we have to consider the specific populations that we are serving and that factors such as socioeconomic status and frailty have to be taken into account.
“Both the RESILIENT trials have shown that thrombectomy does not appear to be suitable for older patients, over 68-70 years of age, in the public health service in Brazil,” he noted. “In this population, a patient aged 70 can be quite different to a patient of the same age in a first-world country. I think in our population, an age of over 68-70 is a surrogate for frailty, which will not be the case in first-world countries. In this regard, I think we have found a ceiling effect for benefit of thrombectomy, which is frailty.”
Dr. Nogueira speculated that the bidirectional effect on the mRS shift analysis may also have been caused by the frailty of some of the patients.
“What the results may be showing is that for most of the population, there is a benefit of thrombectomy, but for some patients, possibly the most frail, then the procedure can be too overwhelming for them. But the suggestion of harm was not significant, so this observation could have also just been the play of chance,” he added.
Interpreting the Findings
Commenting on the RESILIENT-Extend study results, Michael Hill, MD, professor of neurology at the University of Calgary, Canada, pointed out that there was an absolute benefit of 11.1% on the mRS of 0-2 outcome but a similar signal of harm, with a 10.2% increase in mortality in the thrombectomy group, although that was not statistically significant.
“This signal of harm appears not to be due to an increase in intracranial hemorrhage or procedural mishap,” he said. “It is unclear why there were more deaths; the overall trial numbers are small enough that this could be a chance finding.”
Dr. Hill also noted that the absolute proportion of patients achieving an independent functional outcome was 50% less than in the DAWN trial of thrombectomy in the extended window. “This tells us that the patients selected for inclusion into RESILIENT-Extend were physiologically different from those in DAWN,” he said.
Also commenting on the study, Amrou Sarraj, MD, professor of neurology at University Hospitals Cleveland Medical Center–Case Western Reserve University in Cleveland, said: “The RESILIENT-Extend investigators should be congratulated for the successful conduct of the trial and providing evidence of benefit of thrombectomy procedure with simplified neuroimaging protocol using CT and CTA in resource-limited settings. These findings will help support extending the access to thrombectomy in areas without availability of advanced imaging.”
He said the bidirectional effect on the primary endpoint and the positive interaction between age and thrombectomy treatment effect warranted further investigation.
The RESILIENT-Extend trial was sponsored by the Brazilian Ministry of Health.
A version of this article appeared on Medscape.com.
PHOENIX — Thrombectomy is generally beneficial for patients from a low-income population who have a large vessel occlusion stroke presenting in the later time window and who can be identified as suitable for treatment without the need for advanced and costly imaging, a new Brazilian trial has shown.
“The RESILIENT-Extend trial is the first major study of thrombectomy in the late time window (8-24 h) conducted outside first-world countries and shows the procedure also has benefit in a lower socioeconomic status population without the need for costly imaging equipment,” said lead investigator Raul G. Nogueira, MD.
“The trial expands the treatment window for thrombectomy globally with simplified selection criteria based on non-contrast CT, potentially altering current guidelines,” Dr. Nogueira said.
However, there were some caveats that need to be considered; in particular, a lack of benefit with thrombectomy in older patients (over 68 years of age), which Dr. Nogueira believes is a reflection of the particular population enrolled in this study. Specifically, he suggested that older age in this low socioeconomic status population is a surrogate for frailty, and the study may have identified frailty as a factor that correlates with reduced or lack of benefit of thrombectomy.
Dr. Nogueira, who is a professor of neurology and neurosurgery at the University of Pittsburgh, and Sheila Martins, MD, a professor of neurology at Hospital de Clinicas Porto Alegre in Brazil, presented the RESILIENT-Extend results at the International Stroke Conference presented by the American Stroke Association, a division of the American Heart Association.
Dr. Nogueira explained that the lack of available advanced imaging techniques is a major challenge for implementing endovascular therapy in an extended time window, especially in lower-income countries.
“Our main objective was to see if we could remove the need for advanced imaging to select patients with large vessel occlusion stroke in the late time window (8-24 h) for thrombectomy,” he said. “In this way, our trial overlaps somewhat with the MR CLEAN-LATE Trial conducted in the Netherlands, although the two trials were conducted in very different socioeconomic populations.”
The RESILIENT-Extend trial was conducted in the public health service of Brazil and involved a different population of people than have been included in other thrombectomy trials, which have mostly been conducted in first-world countries.
“The public health system in Brazil is not well-resourced and tends to care for patients at lower socioeconomic levels. These patients are fundamentally different from the average patients in the first-world recruited into most other thrombectomy trials,” Dr. Nogueira noted.
The trial enrolled 245 patients with a large vessel occlusion stroke within 8-24 hours of last known well. Patients were included who had a mismatch between the clinical severity as shown by the National Institutes of Health Stroke Scale (NIHSS) score and the stroke burden on imaging as measured by ASPECTS scores.
They had to have relatively high NIHSS scores (8 or more) showing more severe strokes but also a high ASPECTS score (5-10) excluding patients with large areas of ischemic brain. There was also a sliding scale that adjusted for age to avoid enrolling elderly patients with large strokes.
These patients were identified exclusively using non-contrast CT and CT angiography imaging.
The median age of patients included was 62-63 years. Dr. Nogueira pointed out that patients were slightly younger than seen in other thrombectomy trials, perhaps because in lower-middle-income countries strokes occur at a younger age. They also have a higher case fatality rate.
The median baseline NIHSS score was 16, and the median ASPECTS score was 7-8.
The median time to treatment was 12.5 hours, which is similar to other late window thrombectomy trials.
Conflicting Results on Shift Analysis
The primary outcome was a shift analysis of the modified Rankin Scale (mRS) disability score at 90 days.
This showed a bidirectional result, with thrombectomy increasing the chances of a good or excellent outcome (mRS, 0-3), but there was also a nonsignificant increased risk for a bad outcome (mRS, 5-6).
“This bidirectional result prevents a common odds ratio from being calculated, so the primary endpoint is not applicable,” Dr. Nogueira reported.
The researchers therefore used the secondary outcomes as the main results of the study.
These showed that the number of patients achieving a good outcome (mRS, 0-2) was significantly increased with thrombectomy (25% vs. 14%, adjusted odds ratio, 2.56; P = .012).
The number of patients achieving an excellent outcome (mRS, 0-1) was also significantly increased.
But these increases in good outcomes came at the cost of some patients having an increased risk for severe disability or death (mRS, 5-6).
The odds ratio for an mRS of 0-4 versus 5-6 was 0.71, and for an mRS of 0-5 versus 6, the odds ratio was 0.58. Both these results were nonsignificant.
Another anomaly in the RESILIENT-Extend trial was the observation of no benefit of thrombectomy seen in older patients.
“In general, trials of thrombectomy in the first world have shown a greater treatment effect in older patients, but this was not seen in our trial, where older patients (over 68 years) did not derive any benefit from the procedure,” Dr. Nogueira noted.
A similar observation was also seen in the first RESILIENT trial in patients treated within 8 hours of stroke onset, which was also conducted in Brazil, leading to the suggestion that it is related to the patient population included.
“In the Brazilian public health service, older patients are very vulnerable and frail. They are different to older patients in first world countries. It appears they may be too fragile to withstand the thrombectomy process,” Dr. Nogueira said.
Frailty: A Ceiling Effect?
Results from the two RESILIENT trials give a word of caution to the thrombectomy field, Dr. Nogueira said.
“This procedure was initially thought suitable only for patients with small core strokes, but we now have a series of trials showing benefit of thrombectomy in large core strokes as well,” Dr. Nogueira said. “We have started to believe that this intervention will benefit almost all patients with large vessel occlusion stroke everywhere around the world, but our data suggest that we have to consider the specific populations that we are serving and that factors such as socioeconomic status and frailty have to be taken into account.
“Both the RESILIENT trials have shown that thrombectomy does not appear to be suitable for older patients, over 68-70 years of age, in the public health service in Brazil,” he noted. “In this population, a patient aged 70 can be quite different to a patient of the same age in a first-world country. I think in our population, an age of over 68-70 is a surrogate for frailty, which will not be the case in first-world countries. In this regard, I think we have found a ceiling effect for benefit of thrombectomy, which is frailty.”
Dr. Nogueira speculated that the bidirectional effect on the mRS shift analysis may also have been caused by the frailty of some of the patients.
“What the results may be showing is that for most of the population, there is a benefit of thrombectomy, but for some patients, possibly the most frail, then the procedure can be too overwhelming for them. But the suggestion of harm was not significant, so this observation could have also just been the play of chance,” he added.
Interpreting the Findings
Commenting on the RESILIENT-Extend study results, Michael Hill, MD, professor of neurology at the University of Calgary, Canada, pointed out that there was an absolute benefit of 11.1% on the mRS of 0-2 outcome but a similar signal of harm, with a 10.2% increase in mortality in the thrombectomy group, although that was not statistically significant.
“This signal of harm appears not to be due to an increase in intracranial hemorrhage or procedural mishap,” he said. “It is unclear why there were more deaths; the overall trial numbers are small enough that this could be a chance finding.”
Dr. Hill also noted that the absolute proportion of patients achieving an independent functional outcome was 50% less than in the DAWN trial of thrombectomy in the extended window. “This tells us that the patients selected for inclusion into RESILIENT-Extend were physiologically different from those in DAWN,” he said.
Also commenting on the study, Amrou Sarraj, MD, professor of neurology at University Hospitals Cleveland Medical Center–Case Western Reserve University in Cleveland, said: “The RESILIENT-Extend investigators should be congratulated for the successful conduct of the trial and providing evidence of benefit of thrombectomy procedure with simplified neuroimaging protocol using CT and CTA in resource-limited settings. These findings will help support extending the access to thrombectomy in areas without availability of advanced imaging.”
He said the bidirectional effect on the primary endpoint and the positive interaction between age and thrombectomy treatment effect warranted further investigation.
The RESILIENT-Extend trial was sponsored by the Brazilian Ministry of Health.
A version of this article appeared on Medscape.com.
From ISC 2004
Mixing Paxlovid With Specific Immunosuppressants Risks Serious Adverse Reactions
The Pharmacovigilance Risk Assessment Committee (PRAC) of the European Medicines Agency (EMA) has issued a reminder to healthcare professionals regarding the potential serious adverse reactions associated with Paxlovid when administered in combination with specific immunosuppressants.
These immunosuppressants, encompassing calcineurin inhibitors (tacrolimus and ciclosporin) and mTOR inhibitors (everolimus and sirolimus), possess a narrow safe dosage range. They are recognized for their role in diminishing the activity of the immune system and are typically prescribed for autoimmune conditions and organ transplant recipients.
The highlighted risk arises due to drug-drug interactions, which can compromise the body’s ability to eliminate these medicines effectively.
Paxlovid, also known as nirmatrelvir with ritonavir, is an antiviral medication used to treat COVID-19 in adults who do not require supplemental oxygen and who are at an increased risk of progressing to severe COVID-19. It should be administered as soon as possible after a diagnosis of COVID-19 has been made and within 5 days of symptom onset.
Conditional marketing authorization for Paxlovid was granted across the European Union (EU) on January 28, 2022, and subsequently transitioned to full marketing authorization on February 24, 2023.
Developed by Pfizer, Paxlovid exhibited an 89% reduction in the risk for hospitalization or death among unvaccinated individuals in a phase 2-3 clinical trial. This led the National Institutes of Health to prioritize Paxlovid over other COVID-19 treatments. Subsequent real-world studies have affirmed its effectiveness, even among the vaccinated.
When combining Paxlovid with tacrolimus, ciclosporin, everolimus, or sirolimus, healthcare professionals need to actively monitor their blood levels. This proactive approach is essential to mitigate the risk for drug-drug interactions and potential serious reactions. They should collaborate with a multidisciplinary team of specialists to navigate the complexities of administering these medications concurrently.
Further, Paxlovid must not be coadministered with medications highly reliant on CYP3A liver enzymes for elimination, such as the immunosuppressant voclosporin. When administered together, there is a risk for these drugs interfering with each other’s metabolism, potentially leading to altered blood levels, reduced effectiveness, or an increased risk for adverse reactions.
After a thorough review, PRAC has highlighted potential serious adverse reactions, including fatal cases, due to drug interactions between Paxlovid and specified immunosuppressants. Thus, it issued a direct healthcare professional communication (DHPC) to emphasize the recognized risk for these interactions, as previously outlined in Paxlovid’s product information.
The DHPC for Paxlovid will undergo further evaluation by EMA’s Committee for Medicinal Products for Human Use and, upon adoption, will be disseminated to healthcare professionals. The communication plan will include publication on the DHPCs page and in national registers across EU Member States.
A version of this article appeared on Medscape.com.
The Pharmacovigilance Risk Assessment Committee (PRAC) of the European Medicines Agency (EMA) has issued a reminder to healthcare professionals regarding the potential serious adverse reactions associated with Paxlovid when administered in combination with specific immunosuppressants.
These immunosuppressants, encompassing calcineurin inhibitors (tacrolimus and ciclosporin) and mTOR inhibitors (everolimus and sirolimus), possess a narrow safe dosage range. They are recognized for their role in diminishing the activity of the immune system and are typically prescribed for autoimmune conditions and organ transplant recipients.
The highlighted risk arises due to drug-drug interactions, which can compromise the body’s ability to eliminate these medicines effectively.
Paxlovid, also known as nirmatrelvir with ritonavir, is an antiviral medication used to treat COVID-19 in adults who do not require supplemental oxygen and who are at an increased risk of progressing to severe COVID-19. It should be administered as soon as possible after a diagnosis of COVID-19 has been made and within 5 days of symptom onset.
Conditional marketing authorization for Paxlovid was granted across the European Union (EU) on January 28, 2022, and subsequently transitioned to full marketing authorization on February 24, 2023.
Developed by Pfizer, Paxlovid exhibited an 89% reduction in the risk for hospitalization or death among unvaccinated individuals in a phase 2-3 clinical trial. This led the National Institutes of Health to prioritize Paxlovid over other COVID-19 treatments. Subsequent real-world studies have affirmed its effectiveness, even among the vaccinated.
When combining Paxlovid with tacrolimus, ciclosporin, everolimus, or sirolimus, healthcare professionals need to actively monitor their blood levels. This proactive approach is essential to mitigate the risk for drug-drug interactions and potential serious reactions. They should collaborate with a multidisciplinary team of specialists to navigate the complexities of administering these medications concurrently.
Further, Paxlovid must not be coadministered with medications highly reliant on CYP3A liver enzymes for elimination, such as the immunosuppressant voclosporin. When administered together, there is a risk for these drugs interfering with each other’s metabolism, potentially leading to altered blood levels, reduced effectiveness, or an increased risk for adverse reactions.
After a thorough review, PRAC has highlighted potential serious adverse reactions, including fatal cases, due to drug interactions between Paxlovid and specified immunosuppressants. Thus, it issued a direct healthcare professional communication (DHPC) to emphasize the recognized risk for these interactions, as previously outlined in Paxlovid’s product information.
The DHPC for Paxlovid will undergo further evaluation by EMA’s Committee for Medicinal Products for Human Use and, upon adoption, will be disseminated to healthcare professionals. The communication plan will include publication on the DHPCs page and in national registers across EU Member States.
A version of this article appeared on Medscape.com.
The Pharmacovigilance Risk Assessment Committee (PRAC) of the European Medicines Agency (EMA) has issued a reminder to healthcare professionals regarding the potential serious adverse reactions associated with Paxlovid when administered in combination with specific immunosuppressants.
These immunosuppressants, encompassing calcineurin inhibitors (tacrolimus and ciclosporin) and mTOR inhibitors (everolimus and sirolimus), possess a narrow safe dosage range. They are recognized for their role in diminishing the activity of the immune system and are typically prescribed for autoimmune conditions and organ transplant recipients.
The highlighted risk arises due to drug-drug interactions, which can compromise the body’s ability to eliminate these medicines effectively.
Paxlovid, also known as nirmatrelvir with ritonavir, is an antiviral medication used to treat COVID-19 in adults who do not require supplemental oxygen and who are at an increased risk of progressing to severe COVID-19. It should be administered as soon as possible after a diagnosis of COVID-19 has been made and within 5 days of symptom onset.
Conditional marketing authorization for Paxlovid was granted across the European Union (EU) on January 28, 2022, and subsequently transitioned to full marketing authorization on February 24, 2023.
Developed by Pfizer, Paxlovid exhibited an 89% reduction in the risk for hospitalization or death among unvaccinated individuals in a phase 2-3 clinical trial. This led the National Institutes of Health to prioritize Paxlovid over other COVID-19 treatments. Subsequent real-world studies have affirmed its effectiveness, even among the vaccinated.
When combining Paxlovid with tacrolimus, ciclosporin, everolimus, or sirolimus, healthcare professionals need to actively monitor their blood levels. This proactive approach is essential to mitigate the risk for drug-drug interactions and potential serious reactions. They should collaborate with a multidisciplinary team of specialists to navigate the complexities of administering these medications concurrently.
Further, Paxlovid must not be coadministered with medications highly reliant on CYP3A liver enzymes for elimination, such as the immunosuppressant voclosporin. When administered together, there is a risk for these drugs interfering with each other’s metabolism, potentially leading to altered blood levels, reduced effectiveness, or an increased risk for adverse reactions.
After a thorough review, PRAC has highlighted potential serious adverse reactions, including fatal cases, due to drug interactions between Paxlovid and specified immunosuppressants. Thus, it issued a direct healthcare professional communication (DHPC) to emphasize the recognized risk for these interactions, as previously outlined in Paxlovid’s product information.
The DHPC for Paxlovid will undergo further evaluation by EMA’s Committee for Medicinal Products for Human Use and, upon adoption, will be disseminated to healthcare professionals. The communication plan will include publication on the DHPCs page and in national registers across EU Member States.
A version of this article appeared on Medscape.com.