User login
Official news magazine of the Society of Hospital Medicine
Copyright by Society of Hospital Medicine or related companies. All rights reserved. ISSN 1553-085X
nav[contains(@class, 'nav-ce-stack nav-ce-stack__large-screen')]
header[@id='header']
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
div[contains(@class, 'pane-pub-article-hospitalist')]


Fentanyl analogs nearly double their overdose death toll
, according to preliminary data from 10 states.
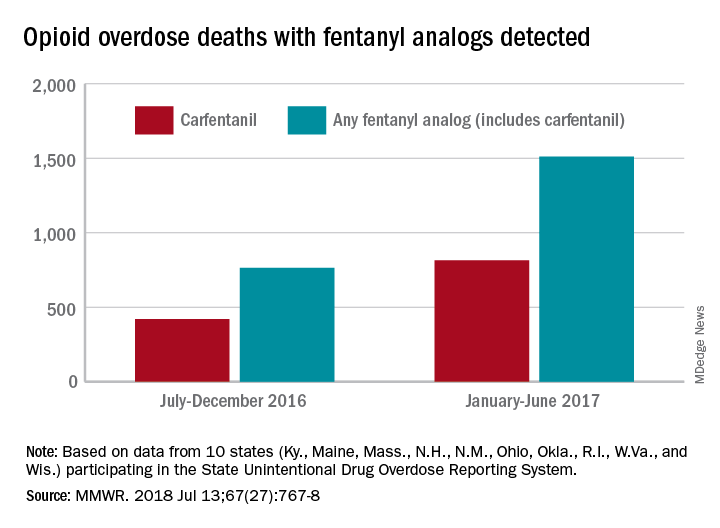
During July 2016 to December 2016, there were 764 opioid overdose deaths that tested positive for any fentanyl analog, with carfentanil being the most common (421 deaths). From January 2017 to June 2017, the respective numbers increased by 98% (1,511) and 94% (815), wrote Julie O’Donnell, PhD, and her associates at the Centers for Disease Control and Prevention’s National Center for Injury Prevention and Control. The report was published in the Morbidity and Mortality Weekly Report.
“The increasing array of fentanyl analogs highlights the need to build forensic toxicological testing capabilities to identify and report emerging threats, and to enhance capacity to rapidly respond to evolving drug trends,” Dr. O’Donnell and her associates said.
Along with carfentanil, 13 other analogs were detected in decedents during the 12-month period: 3-methylfentanyl, 4-fluorobutyrfentanyl, 4-fluorofentanyl, 4-fluoroisobutyrfentanyl, acetylfentanyl, acrylfentanyl, butyrylfentanyl, cyclopropylfentanyl, cyclopentylfentanyl, furanylethylfentanyl, furanylfentanyl, isobutyrylfentanyl, and tetrahydrofuranylfentanyl. Deaths may have involved “more than one analog, as well as ... other opioid and nonopioid substances,” they noted.
The 10 states reporting data to the State Unintentional Drug Overdose Reporting System (SUDORS) were Kentucky, Maine, Massachusetts, New Hampshire, New Mexico, Ohio, Oklahoma, Rhode Island, West Virginia, and Wisconsin. Two other SUDORS-reporting states – Missouri and Pennsylvania – did not have their data ready in time to be included in this analysis.
The increasing availability of fentanyl analogs hit Ohio especially hard: More deaths occurred there than in the other 10 states combined. Of the 421 carfentanil-related deaths in July 2016 to December 2016, nearly 400 were in Ohio, and there were 218 Ohio deaths in April 2017 alone. A look at the bigger picture shows that 3 of the 10 states reported carfentanil-related overdose deaths in the second half of 2016, compared with 7 in the first half of 2017, the investigators said.
Carfentanil, which is the most potent of the 14 fentanyl analogs that have been detected so far, “is intended for sedation of large animals, and is estimated to have 10,000 times the potency of morphine,” Dr. O’Donnell and her associates wrote.
SOURCE: O’Donnell J et al. MMWR. 2018 Jul 13;67(27):767-8.
, according to preliminary data from 10 states.

During July 2016 to December 2016, there were 764 opioid overdose deaths that tested positive for any fentanyl analog, with carfentanil being the most common (421 deaths). From January 2017 to June 2017, the respective numbers increased by 98% (1,511) and 94% (815), wrote Julie O’Donnell, PhD, and her associates at the Centers for Disease Control and Prevention’s National Center for Injury Prevention and Control. The report was published in the Morbidity and Mortality Weekly Report.
“The increasing array of fentanyl analogs highlights the need to build forensic toxicological testing capabilities to identify and report emerging threats, and to enhance capacity to rapidly respond to evolving drug trends,” Dr. O’Donnell and her associates said.
Along with carfentanil, 13 other analogs were detected in decedents during the 12-month period: 3-methylfentanyl, 4-fluorobutyrfentanyl, 4-fluorofentanyl, 4-fluoroisobutyrfentanyl, acetylfentanyl, acrylfentanyl, butyrylfentanyl, cyclopropylfentanyl, cyclopentylfentanyl, furanylethylfentanyl, furanylfentanyl, isobutyrylfentanyl, and tetrahydrofuranylfentanyl. Deaths may have involved “more than one analog, as well as ... other opioid and nonopioid substances,” they noted.
The 10 states reporting data to the State Unintentional Drug Overdose Reporting System (SUDORS) were Kentucky, Maine, Massachusetts, New Hampshire, New Mexico, Ohio, Oklahoma, Rhode Island, West Virginia, and Wisconsin. Two other SUDORS-reporting states – Missouri and Pennsylvania – did not have their data ready in time to be included in this analysis.
The increasing availability of fentanyl analogs hit Ohio especially hard: More deaths occurred there than in the other 10 states combined. Of the 421 carfentanil-related deaths in July 2016 to December 2016, nearly 400 were in Ohio, and there were 218 Ohio deaths in April 2017 alone. A look at the bigger picture shows that 3 of the 10 states reported carfentanil-related overdose deaths in the second half of 2016, compared with 7 in the first half of 2017, the investigators said.
Carfentanil, which is the most potent of the 14 fentanyl analogs that have been detected so far, “is intended for sedation of large animals, and is estimated to have 10,000 times the potency of morphine,” Dr. O’Donnell and her associates wrote.
SOURCE: O’Donnell J et al. MMWR. 2018 Jul 13;67(27):767-8.
, according to preliminary data from 10 states.

During July 2016 to December 2016, there were 764 opioid overdose deaths that tested positive for any fentanyl analog, with carfentanil being the most common (421 deaths). From January 2017 to June 2017, the respective numbers increased by 98% (1,511) and 94% (815), wrote Julie O’Donnell, PhD, and her associates at the Centers for Disease Control and Prevention’s National Center for Injury Prevention and Control. The report was published in the Morbidity and Mortality Weekly Report.
“The increasing array of fentanyl analogs highlights the need to build forensic toxicological testing capabilities to identify and report emerging threats, and to enhance capacity to rapidly respond to evolving drug trends,” Dr. O’Donnell and her associates said.
Along with carfentanil, 13 other analogs were detected in decedents during the 12-month period: 3-methylfentanyl, 4-fluorobutyrfentanyl, 4-fluorofentanyl, 4-fluoroisobutyrfentanyl, acetylfentanyl, acrylfentanyl, butyrylfentanyl, cyclopropylfentanyl, cyclopentylfentanyl, furanylethylfentanyl, furanylfentanyl, isobutyrylfentanyl, and tetrahydrofuranylfentanyl. Deaths may have involved “more than one analog, as well as ... other opioid and nonopioid substances,” they noted.
The 10 states reporting data to the State Unintentional Drug Overdose Reporting System (SUDORS) were Kentucky, Maine, Massachusetts, New Hampshire, New Mexico, Ohio, Oklahoma, Rhode Island, West Virginia, and Wisconsin. Two other SUDORS-reporting states – Missouri and Pennsylvania – did not have their data ready in time to be included in this analysis.
The increasing availability of fentanyl analogs hit Ohio especially hard: More deaths occurred there than in the other 10 states combined. Of the 421 carfentanil-related deaths in July 2016 to December 2016, nearly 400 were in Ohio, and there were 218 Ohio deaths in April 2017 alone. A look at the bigger picture shows that 3 of the 10 states reported carfentanil-related overdose deaths in the second half of 2016, compared with 7 in the first half of 2017, the investigators said.
Carfentanil, which is the most potent of the 14 fentanyl analogs that have been detected so far, “is intended for sedation of large animals, and is estimated to have 10,000 times the potency of morphine,” Dr. O’Donnell and her associates wrote.
SOURCE: O’Donnell J et al. MMWR. 2018 Jul 13;67(27):767-8.
FROM MMWR
Pediatric inpatient seizures treated quickly with new intervention
TORONTO – Researchers at UCSF Benioff Children’s Hospital in San Francisco implemented a novel intervention that leveraged existing in-room technology to expedite antiepileptic drug administration to inpatients having a seizure.
With the quality initiative, they were able to decrease median time from seizure onset to benzodiazepine (BZD) administration from 7 minutes (preintervention) to 2 minutes (post intervention) and reduce the median time from order to administration of second-phase non-BZDs from 28 minutes to 11 minutes.
“Leveraging existing patient room technology to mobilize pharmacy to the bedside expedited non-BZD administration by 60%,” reported principal investigator Arpi Bekmezian, MD, a pediatric hospitalist and medical director of quality and safety at Benioff Children’s Hospital. She presented the findings at the Pediatric Academic Societies annual meeting.
“Furthermore, the rapid-response seizure rescue process may have created an increased sense of urgency helping to expedite initial BZD administration by 70%. ... This may have prevented the need for second-phase therapy and progression to status epilepticus, potentially minimizing the risk of neuronal injury, and all without the additional resources of a Code team.”
Early and rapid escalation of treatment is critical to prevent neuronal injury in patients with status epilepticus. Guidelines recommend initial antiepileptic therapy at 5 minutes, with rapid escalation to second-phase therapy if the seizure persists.
Preintervention baseline data from UCSF Benioff Children’s indicated a 7-minute lag time from seizure onset to BZD therapy and a 28-minute lag from order to administration of non-BZDs (phenobarbital, phenytoin, levetiracetam, valproic acid). Other studies have shown significantly greater delays to antiepileptic treatment.
“That was just too long, and it matched our clinical experience of being at the bedside of a seizing patient and wondering why the medication was taking so long to arrive from the pharmacy.”
The researchers set out to reduce time to BZD administration from 7 minutes to 5 minutes or less and to reduce time to second-phase non-BZD administration to less than 10 minutes. To accomplish this, a multidisciplinary team that included leadership from physicians, pharmacy, and nursing defined primary and secondary drivers of efficiency, with interventions targeting both team communication and medication delivery.
The intervention period lasted 16 months, during which time there were 61 seizure events requiring urgent antiepileptic treatment. Complete data were available for 57 seizures.
Among the interventions they implemented was to stock all medication-dispensing stations with intranasal/buccal BZD available on “nursing override” for easy access and administration.
Because non-BZDs require pharmacy compounding, and the main pharmacy receives many STAT orders with competing priorities, they developed a hospitalwide “seizure rescue” (SR) process by using patient-room staff terminals to activate a dedicated individual from the pharmacy, who would then report to the bedside with a backpack stocked with non-BZDs ready to compound. Nurses were trained to press the SR button for any seizure that may require urgent therapy.
“We didn’t want nurses to waste time on the phone [calling pharmacy], and we considered calling a Code, but we couldn’t really justify the resource utilization as most of these patients didn’t have respiratory compromise, and they didn’t need the whole Code team,” said Dr. Bekmezian. She noted that her hospital strongly discourages bedside compounding by nursing staff.
Instead, they realized they could easily reprogram the patient-room electronic staff terminals to have a dedicated SR button that would directly alert a dedicated pharmacist carrying the SR phone. The pharmacist could then swipe and confirm that they received the alert and let the nurse know they were on the way, “and this would free up the nurse to go ahead and obtain the benzodiazepines and administer them as pharmacy made their way to the room.”
“To our knowledge, this is the first study to report expediting antiepileptic drug delivery to patients in the hospital,” said Dr. Bekmezian. She noted that less than 50% of cases actually required pharmacist response, “but the pharmacy staff chose to be activated earlier in the management algorithm to avoid delays in treatment.”
UCSF Children’s Hospital San Francisco campus is a 183-bed, tertiary care, teaching children’s hospital that has pediatric, neonatal, and cardiac intensive care units and set-down units. They provide liver, bone marrow, kidney, and cardiac transplantation and have more than 10,000 annual admissions.
The investigators reported no conflicts of interest.
SOURCE: Bekmezian A et al. PAS 2018. Abstract 3545.3.
TORONTO – Researchers at UCSF Benioff Children’s Hospital in San Francisco implemented a novel intervention that leveraged existing in-room technology to expedite antiepileptic drug administration to inpatients having a seizure.
With the quality initiative, they were able to decrease median time from seizure onset to benzodiazepine (BZD) administration from 7 minutes (preintervention) to 2 minutes (post intervention) and reduce the median time from order to administration of second-phase non-BZDs from 28 minutes to 11 minutes.
“Leveraging existing patient room technology to mobilize pharmacy to the bedside expedited non-BZD administration by 60%,” reported principal investigator Arpi Bekmezian, MD, a pediatric hospitalist and medical director of quality and safety at Benioff Children’s Hospital. She presented the findings at the Pediatric Academic Societies annual meeting.
“Furthermore, the rapid-response seizure rescue process may have created an increased sense of urgency helping to expedite initial BZD administration by 70%. ... This may have prevented the need for second-phase therapy and progression to status epilepticus, potentially minimizing the risk of neuronal injury, and all without the additional resources of a Code team.”
Early and rapid escalation of treatment is critical to prevent neuronal injury in patients with status epilepticus. Guidelines recommend initial antiepileptic therapy at 5 minutes, with rapid escalation to second-phase therapy if the seizure persists.
Preintervention baseline data from UCSF Benioff Children’s indicated a 7-minute lag time from seizure onset to BZD therapy and a 28-minute lag from order to administration of non-BZDs (phenobarbital, phenytoin, levetiracetam, valproic acid). Other studies have shown significantly greater delays to antiepileptic treatment.
“That was just too long, and it matched our clinical experience of being at the bedside of a seizing patient and wondering why the medication was taking so long to arrive from the pharmacy.”
The researchers set out to reduce time to BZD administration from 7 minutes to 5 minutes or less and to reduce time to second-phase non-BZD administration to less than 10 minutes. To accomplish this, a multidisciplinary team that included leadership from physicians, pharmacy, and nursing defined primary and secondary drivers of efficiency, with interventions targeting both team communication and medication delivery.
The intervention period lasted 16 months, during which time there were 61 seizure events requiring urgent antiepileptic treatment. Complete data were available for 57 seizures.
Among the interventions they implemented was to stock all medication-dispensing stations with intranasal/buccal BZD available on “nursing override” for easy access and administration.
Because non-BZDs require pharmacy compounding, and the main pharmacy receives many STAT orders with competing priorities, they developed a hospitalwide “seizure rescue” (SR) process by using patient-room staff terminals to activate a dedicated individual from the pharmacy, who would then report to the bedside with a backpack stocked with non-BZDs ready to compound. Nurses were trained to press the SR button for any seizure that may require urgent therapy.
“We didn’t want nurses to waste time on the phone [calling pharmacy], and we considered calling a Code, but we couldn’t really justify the resource utilization as most of these patients didn’t have respiratory compromise, and they didn’t need the whole Code team,” said Dr. Bekmezian. She noted that her hospital strongly discourages bedside compounding by nursing staff.
Instead, they realized they could easily reprogram the patient-room electronic staff terminals to have a dedicated SR button that would directly alert a dedicated pharmacist carrying the SR phone. The pharmacist could then swipe and confirm that they received the alert and let the nurse know they were on the way, “and this would free up the nurse to go ahead and obtain the benzodiazepines and administer them as pharmacy made their way to the room.”
“To our knowledge, this is the first study to report expediting antiepileptic drug delivery to patients in the hospital,” said Dr. Bekmezian. She noted that less than 50% of cases actually required pharmacist response, “but the pharmacy staff chose to be activated earlier in the management algorithm to avoid delays in treatment.”
UCSF Children’s Hospital San Francisco campus is a 183-bed, tertiary care, teaching children’s hospital that has pediatric, neonatal, and cardiac intensive care units and set-down units. They provide liver, bone marrow, kidney, and cardiac transplantation and have more than 10,000 annual admissions.
The investigators reported no conflicts of interest.
SOURCE: Bekmezian A et al. PAS 2018. Abstract 3545.3.
TORONTO – Researchers at UCSF Benioff Children’s Hospital in San Francisco implemented a novel intervention that leveraged existing in-room technology to expedite antiepileptic drug administration to inpatients having a seizure.
With the quality initiative, they were able to decrease median time from seizure onset to benzodiazepine (BZD) administration from 7 minutes (preintervention) to 2 minutes (post intervention) and reduce the median time from order to administration of second-phase non-BZDs from 28 minutes to 11 minutes.
“Leveraging existing patient room technology to mobilize pharmacy to the bedside expedited non-BZD administration by 60%,” reported principal investigator Arpi Bekmezian, MD, a pediatric hospitalist and medical director of quality and safety at Benioff Children’s Hospital. She presented the findings at the Pediatric Academic Societies annual meeting.
“Furthermore, the rapid-response seizure rescue process may have created an increased sense of urgency helping to expedite initial BZD administration by 70%. ... This may have prevented the need for second-phase therapy and progression to status epilepticus, potentially minimizing the risk of neuronal injury, and all without the additional resources of a Code team.”
Early and rapid escalation of treatment is critical to prevent neuronal injury in patients with status epilepticus. Guidelines recommend initial antiepileptic therapy at 5 minutes, with rapid escalation to second-phase therapy if the seizure persists.
Preintervention baseline data from UCSF Benioff Children’s indicated a 7-minute lag time from seizure onset to BZD therapy and a 28-minute lag from order to administration of non-BZDs (phenobarbital, phenytoin, levetiracetam, valproic acid). Other studies have shown significantly greater delays to antiepileptic treatment.
“That was just too long, and it matched our clinical experience of being at the bedside of a seizing patient and wondering why the medication was taking so long to arrive from the pharmacy.”
The researchers set out to reduce time to BZD administration from 7 minutes to 5 minutes or less and to reduce time to second-phase non-BZD administration to less than 10 minutes. To accomplish this, a multidisciplinary team that included leadership from physicians, pharmacy, and nursing defined primary and secondary drivers of efficiency, with interventions targeting both team communication and medication delivery.
The intervention period lasted 16 months, during which time there were 61 seizure events requiring urgent antiepileptic treatment. Complete data were available for 57 seizures.
Among the interventions they implemented was to stock all medication-dispensing stations with intranasal/buccal BZD available on “nursing override” for easy access and administration.
Because non-BZDs require pharmacy compounding, and the main pharmacy receives many STAT orders with competing priorities, they developed a hospitalwide “seizure rescue” (SR) process by using patient-room staff terminals to activate a dedicated individual from the pharmacy, who would then report to the bedside with a backpack stocked with non-BZDs ready to compound. Nurses were trained to press the SR button for any seizure that may require urgent therapy.
“We didn’t want nurses to waste time on the phone [calling pharmacy], and we considered calling a Code, but we couldn’t really justify the resource utilization as most of these patients didn’t have respiratory compromise, and they didn’t need the whole Code team,” said Dr. Bekmezian. She noted that her hospital strongly discourages bedside compounding by nursing staff.
Instead, they realized they could easily reprogram the patient-room electronic staff terminals to have a dedicated SR button that would directly alert a dedicated pharmacist carrying the SR phone. The pharmacist could then swipe and confirm that they received the alert and let the nurse know they were on the way, “and this would free up the nurse to go ahead and obtain the benzodiazepines and administer them as pharmacy made their way to the room.”
“To our knowledge, this is the first study to report expediting antiepileptic drug delivery to patients in the hospital,” said Dr. Bekmezian. She noted that less than 50% of cases actually required pharmacist response, “but the pharmacy staff chose to be activated earlier in the management algorithm to avoid delays in treatment.”
UCSF Children’s Hospital San Francisco campus is a 183-bed, tertiary care, teaching children’s hospital that has pediatric, neonatal, and cardiac intensive care units and set-down units. They provide liver, bone marrow, kidney, and cardiac transplantation and have more than 10,000 annual admissions.
The investigators reported no conflicts of interest.
SOURCE: Bekmezian A et al. PAS 2018. Abstract 3545.3.
AT PAS 2018
Key clinical point: An intervention to speed delivery of antiepileptic drugs significantly reduced time to treatment.
Major finding: Median time from seizure onset to benzodiazepine (BZD) administration fell from 7 minutes preintervention to 2 minutes post intervention, and median time from order to administration of non-BZDs dropped from 28 minutes to 11 minutes.
Study details: A prospective, multicenter study of 57 seizure events during a 16-month period.
Disclosures: The investigators reported no conflicts of interest.
Source: Bekmezian A et al. PAS 2018. Abstract 3545.3.
Benefits, drawbacks when hospitalists expand roles
Hospitalists can’t ‘fill all the cracks’ in primary care
As vice chair of the hospital medicine service at Northwell Health, Nick Fitterman, MD, FACP, SFHM, oversees 16 HM groups at 15 hospitals in New York. He says the duties of his hospitalist staff, like those of most U.S. hospitalists, are similar to what they have traditionally been – clinical care on the wards, teaching, comanagement of surgery, quality improvement, committee work, and research. But he has noticed a trend of late: rapid expansion of the hospitalist’s role.
Speaking at an education session at HM18 in Orlando, Dr. Fitterman said the role of the hospitalist is growing to include tasks that might not be as common, but are becoming more familiar all the time: working at infusion centers, caring for patients in skilled nursing facilities, specializing in electronic health record use, colocating in psychiatric hospitals, even being deployed to natural disasters. His list went on, and it was much longer than the list of traditional hospitalist responsibilities.
“Where do we draw the line and say, ‘Wait a minute, our primary site is going to suffer if we continue to get spread this thin. Can we really do it all?” Dr. Fitterman said. As the number of hats hospitalists wear grows ever bigger, he said more thought must be placed into how expansion happens.
The preop clinic
Efren Manjarrez, MD, SFHM, former chief of hospital medicine at the University of Miami, told a cautionary tale about a preoperative clinic staffed by hospitalists that appeared to provide a financial benefit to a hospital – helping to avoid costly last-minute cancellations of surgeries – but that ultimately was shuttered. The hospital, he said, loses $8,000-$10,000 for each case that gets canceled on the same day.
“Think about that just for a minute,” Dr. Manjarrez said. “If 100 cases are canceled during the year at the last minute, that’s a lot of money.”
A preoperative clinic seemed like a worthwhile role for hospitalists – the program was started in Miami by the same doctor who initiated a similar program at Cleveland Clinic. “Surgical cases are what support the hospital [financially], and we’re here to help them along,” Dr. Manjarrez said. “The purpose of hospitalists is to make sure that patients are medically optimized.”
The preop program concept, used in U.S. medicine since the 1990s, was originally started by anesthesiologists, but they may not always be the best fit to staff such programs.
“Anesthesiologists do not manage all beta blockers,” Dr. Manjarrez said. “They don’t manage ACE inhibitors by mouth. They don’t manage all oral diabetes agents, and they sure as heck don’t manage pills that are anticoagulants. That’s the domain of internal medicine. And as patients have become more complex, that’s where hospitalists who [work in] preop clinics have stepped in.”
Studies have found that hospitalists staffing preop clinics have improved quality metrics and some clinical outcomes, including lowering cancellation rates and more appropriate use of beta blockers, he said.
In the Miami program described by Dr. Manjarrez, hospitalists in the preop clinic at first saw only patients who’d been financially cleared as able to pay. But ultimately, a tiered system was developed, and hospitalists saw only patients who were higher risk – those with COPD or stroke patients, for example – without regard to ability to pay.
“The hospital would have to make up any financial deficit at the very end,” Dr. Manjarrez said. This meant there were no longer efficient 5-minute encounters with patients. Instead, visits lasted about 45 minutes, so fewer patients were seen.
The program was successful, in that the same-day cancellation rate for surgeries dropped to less than 0.1% – fewer than 1 in 1,000 – with the preop clinic up and running, Dr. Manjarrez. Still, the hospital decided to end the program. “The hospital no longer wanted to reimburse us,” he said.
A takeaway from this experience for Dr. Manjarrez was that hospitalists need to do a better job of showing the financial benefits in their expanding roles, if they want them to endure.
“At the end of the day, hospitalists do provide value in preoperative clinics,” he said. “But unfortunately, we’re not doing a great job of publishing our data and showing our value.”
At-home care
At Brigham and Women’s Hospital in Boston, hospitalists have demonstrated good results with a program to provide care at home rather than in the hospital.
David Levine, MD, MPH, MA, clinician and investigator at Brigham and Women’s and an instructor in medicine at Harvard Medical School, said that the structure of inpatient care has generally not changed much over decades, despite advances in technology.
“We round on them once a day – if they’re lucky, twice,” he said. “The medicines have changed and imaging has changed, but we really haven’t changed the structure of how we take care of acutely ill adults for almost a hundred years.”
Hospitalizing patients brings unintended consequences. Twenty percent of older adults will become delirious during their stay, 1 out of 3 will lose a level of functional status in the hospital that they’ll never regain, and 1 out of 10 hospitalized patients will experience an adverse event, like an infection or a medication error.
Brigham and Women’s program of at-home care involves “admitting” patients to their homes after being treated in the emergency department. The goal is to reduce costs by 20%, while maintaining quality and safety and improving patients’ quality of life and experience.
Researchers are studying their results. They randomized patients, after the ED determined they required admission, either to admission to the hospital or to their home. The decision on whether to admit was made before the study investigators became involved with the patients, Dr. Levine said.
The program is also intended to improve access to hospitals. Brigham and Women’s is often over 100% capacity in the general medical ward.
Patients in the study needed to live within a 5-mile radius of either Brigham and Women’s Hospital, or Brigham and Women’s Faulkner Hospital, a nearby community hospital. A physician and a registered nurse form the core team; they assess patient needs and ratchet care either up or down, perhaps adding a home health aide or social worker.
The home care team takes advantage of technology: Portable equipment allows a basic metabolic panel to be performed on the spot – for example, a hemoglobin and hematocrit can be produced within 2 minutes. Also, portable ultrasounds and x-rays are used. Doctors keep a “tackle box” of urgent medications such as antibiotics and diuretics.
“We showed a direct cost reduction taking care of patients at home,” Dr. Levine said. There was also a reduction in utilization of care, and an increase in patient activity, with patients taking about 1,800 steps at home, compared with 180 in the hospital. There were no significant changes in safety, quality, or patient experience, he said.
Postdischarge clinics
Lauren Doctoroff, MD, FHM – a hospitalist at Beth Israel Deaconness Medical Center in Boston and assistant professor of medicine at Harvard Medical School – explained another hospitalist-staffed project meant to improve access to care: her center’s postdischarge clinic, which was started in 2009 but is no longer operating.
The clinic tackled the problem of what to do with patients when you discharge them, Dr. Doctoroff said, and its goal was to foster more cooperation between hospitalists and the faculty primary care practice, as well as to improve postdischarge access for patients from that practice.
A dedicated group of hospitalists staffed the clinics, handling medication reconciliation, symptom management, pending tests, and other services the patients were supposed to be getting after discharge, Dr. Doctoroff said.
“We greatly improved access so that when you came to see us you generally saw a hospitalist a week before you would have seen your primary care doctor,” she said. “And that was mostly because we created open access in a clinic that did not have open access. So if a doctor discharging a patient really thought that the patient needed to be seen after discharge, they would often see us.”
Hospitalists considering starting such a clinic have several key questions to consider, Dr. Doctoroff said.
“You need to focus on who the patient population is, the clinic structure, how you plan to staff the clinic, and what your outcomes are – mainly how you will measure performance,” she said.
Dr. Doctoroff said hospitalists are good for this role because “we’re very comfortable with patients who are complicated, and we are very adept at accessing information from the hospitalization. I think, as a hospitalist who spent 5 years seeing patients in a discharge clinic, it greatly enhances my understanding of patients and their challenges at discharge.”
The clinic was closed, she said, in part because it was largely an extension of primary care, and the patient volume wasn’t big enough to justify continuing it.
“Postdischarge clinics are, in a very narrow sense, a bit of a Band-Aid for a really dysfunctional primary care system,” Dr. Doctoroff said. “Ideally, if all you’re doing is providing a postdischarge physician visit, then you really want primary care to be able to do that in order to reengage with their patient. I think this is because postdischarge clinics are construed in a very narrow way to address the simple need to see a patient after discharge. And this may lead to the failure of these clinics, or make them easy to replace. Also, often what patients really need is more than just a physician visit, so a discharge clinic may need to be designed to provide an enhanced array of services.”
Dr. Fitterman said that these stories show that not all role expansion in hospital medicine is good role expansion. The experiences described by Dr. Manjarrez, Dr. Levine, and Dr. Doctoroff demonstrate the challenges hospitalists face as they attempt expansions into new roles, he said.
“We can’t be expected to fill all the cracks in primary care,” Dr. Fitterman said. “As a country we need to really prop up primary care. This all can’t come under the roof of hospital medicine. We need to be part of a patient-centered medical home – but we are not the patient-centered medical home.”
He said the experience with the preop clinic described by Dr. Manjarrez also shows the need for buy-in from hospital or health system administration.
“While most of us are employed by hospitals and want to help meet their needs, we have to be more cautious. We have to look, I think, with a more critical eye, for the value; it may not always be in the dollars coming back in,” he said. “It might be in cost avoidance, such as reducing readmissions, or reducing same-day cancellations in an OR. Unless the C-suite appreciates that value, such programs will be short-lived.”
Hospitalists can’t ‘fill all the cracks’ in primary care
Hospitalists can’t ‘fill all the cracks’ in primary care
As vice chair of the hospital medicine service at Northwell Health, Nick Fitterman, MD, FACP, SFHM, oversees 16 HM groups at 15 hospitals in New York. He says the duties of his hospitalist staff, like those of most U.S. hospitalists, are similar to what they have traditionally been – clinical care on the wards, teaching, comanagement of surgery, quality improvement, committee work, and research. But he has noticed a trend of late: rapid expansion of the hospitalist’s role.
Speaking at an education session at HM18 in Orlando, Dr. Fitterman said the role of the hospitalist is growing to include tasks that might not be as common, but are becoming more familiar all the time: working at infusion centers, caring for patients in skilled nursing facilities, specializing in electronic health record use, colocating in psychiatric hospitals, even being deployed to natural disasters. His list went on, and it was much longer than the list of traditional hospitalist responsibilities.
“Where do we draw the line and say, ‘Wait a minute, our primary site is going to suffer if we continue to get spread this thin. Can we really do it all?” Dr. Fitterman said. As the number of hats hospitalists wear grows ever bigger, he said more thought must be placed into how expansion happens.
The preop clinic
Efren Manjarrez, MD, SFHM, former chief of hospital medicine at the University of Miami, told a cautionary tale about a preoperative clinic staffed by hospitalists that appeared to provide a financial benefit to a hospital – helping to avoid costly last-minute cancellations of surgeries – but that ultimately was shuttered. The hospital, he said, loses $8,000-$10,000 for each case that gets canceled on the same day.
“Think about that just for a minute,” Dr. Manjarrez said. “If 100 cases are canceled during the year at the last minute, that’s a lot of money.”
A preoperative clinic seemed like a worthwhile role for hospitalists – the program was started in Miami by the same doctor who initiated a similar program at Cleveland Clinic. “Surgical cases are what support the hospital [financially], and we’re here to help them along,” Dr. Manjarrez said. “The purpose of hospitalists is to make sure that patients are medically optimized.”
The preop program concept, used in U.S. medicine since the 1990s, was originally started by anesthesiologists, but they may not always be the best fit to staff such programs.
“Anesthesiologists do not manage all beta blockers,” Dr. Manjarrez said. “They don’t manage ACE inhibitors by mouth. They don’t manage all oral diabetes agents, and they sure as heck don’t manage pills that are anticoagulants. That’s the domain of internal medicine. And as patients have become more complex, that’s where hospitalists who [work in] preop clinics have stepped in.”
Studies have found that hospitalists staffing preop clinics have improved quality metrics and some clinical outcomes, including lowering cancellation rates and more appropriate use of beta blockers, he said.
In the Miami program described by Dr. Manjarrez, hospitalists in the preop clinic at first saw only patients who’d been financially cleared as able to pay. But ultimately, a tiered system was developed, and hospitalists saw only patients who were higher risk – those with COPD or stroke patients, for example – without regard to ability to pay.
“The hospital would have to make up any financial deficit at the very end,” Dr. Manjarrez said. This meant there were no longer efficient 5-minute encounters with patients. Instead, visits lasted about 45 minutes, so fewer patients were seen.
The program was successful, in that the same-day cancellation rate for surgeries dropped to less than 0.1% – fewer than 1 in 1,000 – with the preop clinic up and running, Dr. Manjarrez. Still, the hospital decided to end the program. “The hospital no longer wanted to reimburse us,” he said.
A takeaway from this experience for Dr. Manjarrez was that hospitalists need to do a better job of showing the financial benefits in their expanding roles, if they want them to endure.
“At the end of the day, hospitalists do provide value in preoperative clinics,” he said. “But unfortunately, we’re not doing a great job of publishing our data and showing our value.”
At-home care
At Brigham and Women’s Hospital in Boston, hospitalists have demonstrated good results with a program to provide care at home rather than in the hospital.
David Levine, MD, MPH, MA, clinician and investigator at Brigham and Women’s and an instructor in medicine at Harvard Medical School, said that the structure of inpatient care has generally not changed much over decades, despite advances in technology.
“We round on them once a day – if they’re lucky, twice,” he said. “The medicines have changed and imaging has changed, but we really haven’t changed the structure of how we take care of acutely ill adults for almost a hundred years.”
Hospitalizing patients brings unintended consequences. Twenty percent of older adults will become delirious during their stay, 1 out of 3 will lose a level of functional status in the hospital that they’ll never regain, and 1 out of 10 hospitalized patients will experience an adverse event, like an infection or a medication error.
Brigham and Women’s program of at-home care involves “admitting” patients to their homes after being treated in the emergency department. The goal is to reduce costs by 20%, while maintaining quality and safety and improving patients’ quality of life and experience.
Researchers are studying their results. They randomized patients, after the ED determined they required admission, either to admission to the hospital or to their home. The decision on whether to admit was made before the study investigators became involved with the patients, Dr. Levine said.
The program is also intended to improve access to hospitals. Brigham and Women’s is often over 100% capacity in the general medical ward.
Patients in the study needed to live within a 5-mile radius of either Brigham and Women’s Hospital, or Brigham and Women’s Faulkner Hospital, a nearby community hospital. A physician and a registered nurse form the core team; they assess patient needs and ratchet care either up or down, perhaps adding a home health aide or social worker.
The home care team takes advantage of technology: Portable equipment allows a basic metabolic panel to be performed on the spot – for example, a hemoglobin and hematocrit can be produced within 2 minutes. Also, portable ultrasounds and x-rays are used. Doctors keep a “tackle box” of urgent medications such as antibiotics and diuretics.
“We showed a direct cost reduction taking care of patients at home,” Dr. Levine said. There was also a reduction in utilization of care, and an increase in patient activity, with patients taking about 1,800 steps at home, compared with 180 in the hospital. There were no significant changes in safety, quality, or patient experience, he said.
Postdischarge clinics
Lauren Doctoroff, MD, FHM – a hospitalist at Beth Israel Deaconness Medical Center in Boston and assistant professor of medicine at Harvard Medical School – explained another hospitalist-staffed project meant to improve access to care: her center’s postdischarge clinic, which was started in 2009 but is no longer operating.
The clinic tackled the problem of what to do with patients when you discharge them, Dr. Doctoroff said, and its goal was to foster more cooperation between hospitalists and the faculty primary care practice, as well as to improve postdischarge access for patients from that practice.
A dedicated group of hospitalists staffed the clinics, handling medication reconciliation, symptom management, pending tests, and other services the patients were supposed to be getting after discharge, Dr. Doctoroff said.
“We greatly improved access so that when you came to see us you generally saw a hospitalist a week before you would have seen your primary care doctor,” she said. “And that was mostly because we created open access in a clinic that did not have open access. So if a doctor discharging a patient really thought that the patient needed to be seen after discharge, they would often see us.”
Hospitalists considering starting such a clinic have several key questions to consider, Dr. Doctoroff said.
“You need to focus on who the patient population is, the clinic structure, how you plan to staff the clinic, and what your outcomes are – mainly how you will measure performance,” she said.
Dr. Doctoroff said hospitalists are good for this role because “we’re very comfortable with patients who are complicated, and we are very adept at accessing information from the hospitalization. I think, as a hospitalist who spent 5 years seeing patients in a discharge clinic, it greatly enhances my understanding of patients and their challenges at discharge.”
The clinic was closed, she said, in part because it was largely an extension of primary care, and the patient volume wasn’t big enough to justify continuing it.
“Postdischarge clinics are, in a very narrow sense, a bit of a Band-Aid for a really dysfunctional primary care system,” Dr. Doctoroff said. “Ideally, if all you’re doing is providing a postdischarge physician visit, then you really want primary care to be able to do that in order to reengage with their patient. I think this is because postdischarge clinics are construed in a very narrow way to address the simple need to see a patient after discharge. And this may lead to the failure of these clinics, or make them easy to replace. Also, often what patients really need is more than just a physician visit, so a discharge clinic may need to be designed to provide an enhanced array of services.”
Dr. Fitterman said that these stories show that not all role expansion in hospital medicine is good role expansion. The experiences described by Dr. Manjarrez, Dr. Levine, and Dr. Doctoroff demonstrate the challenges hospitalists face as they attempt expansions into new roles, he said.
“We can’t be expected to fill all the cracks in primary care,” Dr. Fitterman said. “As a country we need to really prop up primary care. This all can’t come under the roof of hospital medicine. We need to be part of a patient-centered medical home – but we are not the patient-centered medical home.”
He said the experience with the preop clinic described by Dr. Manjarrez also shows the need for buy-in from hospital or health system administration.
“While most of us are employed by hospitals and want to help meet their needs, we have to be more cautious. We have to look, I think, with a more critical eye, for the value; it may not always be in the dollars coming back in,” he said. “It might be in cost avoidance, such as reducing readmissions, or reducing same-day cancellations in an OR. Unless the C-suite appreciates that value, such programs will be short-lived.”
As vice chair of the hospital medicine service at Northwell Health, Nick Fitterman, MD, FACP, SFHM, oversees 16 HM groups at 15 hospitals in New York. He says the duties of his hospitalist staff, like those of most U.S. hospitalists, are similar to what they have traditionally been – clinical care on the wards, teaching, comanagement of surgery, quality improvement, committee work, and research. But he has noticed a trend of late: rapid expansion of the hospitalist’s role.
Speaking at an education session at HM18 in Orlando, Dr. Fitterman said the role of the hospitalist is growing to include tasks that might not be as common, but are becoming more familiar all the time: working at infusion centers, caring for patients in skilled nursing facilities, specializing in electronic health record use, colocating in psychiatric hospitals, even being deployed to natural disasters. His list went on, and it was much longer than the list of traditional hospitalist responsibilities.
“Where do we draw the line and say, ‘Wait a minute, our primary site is going to suffer if we continue to get spread this thin. Can we really do it all?” Dr. Fitterman said. As the number of hats hospitalists wear grows ever bigger, he said more thought must be placed into how expansion happens.
The preop clinic
Efren Manjarrez, MD, SFHM, former chief of hospital medicine at the University of Miami, told a cautionary tale about a preoperative clinic staffed by hospitalists that appeared to provide a financial benefit to a hospital – helping to avoid costly last-minute cancellations of surgeries – but that ultimately was shuttered. The hospital, he said, loses $8,000-$10,000 for each case that gets canceled on the same day.
“Think about that just for a minute,” Dr. Manjarrez said. “If 100 cases are canceled during the year at the last minute, that’s a lot of money.”
A preoperative clinic seemed like a worthwhile role for hospitalists – the program was started in Miami by the same doctor who initiated a similar program at Cleveland Clinic. “Surgical cases are what support the hospital [financially], and we’re here to help them along,” Dr. Manjarrez said. “The purpose of hospitalists is to make sure that patients are medically optimized.”
The preop program concept, used in U.S. medicine since the 1990s, was originally started by anesthesiologists, but they may not always be the best fit to staff such programs.
“Anesthesiologists do not manage all beta blockers,” Dr. Manjarrez said. “They don’t manage ACE inhibitors by mouth. They don’t manage all oral diabetes agents, and they sure as heck don’t manage pills that are anticoagulants. That’s the domain of internal medicine. And as patients have become more complex, that’s where hospitalists who [work in] preop clinics have stepped in.”
Studies have found that hospitalists staffing preop clinics have improved quality metrics and some clinical outcomes, including lowering cancellation rates and more appropriate use of beta blockers, he said.
In the Miami program described by Dr. Manjarrez, hospitalists in the preop clinic at first saw only patients who’d been financially cleared as able to pay. But ultimately, a tiered system was developed, and hospitalists saw only patients who were higher risk – those with COPD or stroke patients, for example – without regard to ability to pay.
“The hospital would have to make up any financial deficit at the very end,” Dr. Manjarrez said. This meant there were no longer efficient 5-minute encounters with patients. Instead, visits lasted about 45 minutes, so fewer patients were seen.
The program was successful, in that the same-day cancellation rate for surgeries dropped to less than 0.1% – fewer than 1 in 1,000 – with the preop clinic up and running, Dr. Manjarrez. Still, the hospital decided to end the program. “The hospital no longer wanted to reimburse us,” he said.
A takeaway from this experience for Dr. Manjarrez was that hospitalists need to do a better job of showing the financial benefits in their expanding roles, if they want them to endure.
“At the end of the day, hospitalists do provide value in preoperative clinics,” he said. “But unfortunately, we’re not doing a great job of publishing our data and showing our value.”
At-home care
At Brigham and Women’s Hospital in Boston, hospitalists have demonstrated good results with a program to provide care at home rather than in the hospital.
David Levine, MD, MPH, MA, clinician and investigator at Brigham and Women’s and an instructor in medicine at Harvard Medical School, said that the structure of inpatient care has generally not changed much over decades, despite advances in technology.
“We round on them once a day – if they’re lucky, twice,” he said. “The medicines have changed and imaging has changed, but we really haven’t changed the structure of how we take care of acutely ill adults for almost a hundred years.”
Hospitalizing patients brings unintended consequences. Twenty percent of older adults will become delirious during their stay, 1 out of 3 will lose a level of functional status in the hospital that they’ll never regain, and 1 out of 10 hospitalized patients will experience an adverse event, like an infection or a medication error.
Brigham and Women’s program of at-home care involves “admitting” patients to their homes after being treated in the emergency department. The goal is to reduce costs by 20%, while maintaining quality and safety and improving patients’ quality of life and experience.
Researchers are studying their results. They randomized patients, after the ED determined they required admission, either to admission to the hospital or to their home. The decision on whether to admit was made before the study investigators became involved with the patients, Dr. Levine said.
The program is also intended to improve access to hospitals. Brigham and Women’s is often over 100% capacity in the general medical ward.
Patients in the study needed to live within a 5-mile radius of either Brigham and Women’s Hospital, or Brigham and Women’s Faulkner Hospital, a nearby community hospital. A physician and a registered nurse form the core team; they assess patient needs and ratchet care either up or down, perhaps adding a home health aide or social worker.
The home care team takes advantage of technology: Portable equipment allows a basic metabolic panel to be performed on the spot – for example, a hemoglobin and hematocrit can be produced within 2 minutes. Also, portable ultrasounds and x-rays are used. Doctors keep a “tackle box” of urgent medications such as antibiotics and diuretics.
“We showed a direct cost reduction taking care of patients at home,” Dr. Levine said. There was also a reduction in utilization of care, and an increase in patient activity, with patients taking about 1,800 steps at home, compared with 180 in the hospital. There were no significant changes in safety, quality, or patient experience, he said.
Postdischarge clinics
Lauren Doctoroff, MD, FHM – a hospitalist at Beth Israel Deaconness Medical Center in Boston and assistant professor of medicine at Harvard Medical School – explained another hospitalist-staffed project meant to improve access to care: her center’s postdischarge clinic, which was started in 2009 but is no longer operating.
The clinic tackled the problem of what to do with patients when you discharge them, Dr. Doctoroff said, and its goal was to foster more cooperation between hospitalists and the faculty primary care practice, as well as to improve postdischarge access for patients from that practice.
A dedicated group of hospitalists staffed the clinics, handling medication reconciliation, symptom management, pending tests, and other services the patients were supposed to be getting after discharge, Dr. Doctoroff said.
“We greatly improved access so that when you came to see us you generally saw a hospitalist a week before you would have seen your primary care doctor,” she said. “And that was mostly because we created open access in a clinic that did not have open access. So if a doctor discharging a patient really thought that the patient needed to be seen after discharge, they would often see us.”
Hospitalists considering starting such a clinic have several key questions to consider, Dr. Doctoroff said.
“You need to focus on who the patient population is, the clinic structure, how you plan to staff the clinic, and what your outcomes are – mainly how you will measure performance,” she said.
Dr. Doctoroff said hospitalists are good for this role because “we’re very comfortable with patients who are complicated, and we are very adept at accessing information from the hospitalization. I think, as a hospitalist who spent 5 years seeing patients in a discharge clinic, it greatly enhances my understanding of patients and their challenges at discharge.”
The clinic was closed, she said, in part because it was largely an extension of primary care, and the patient volume wasn’t big enough to justify continuing it.
“Postdischarge clinics are, in a very narrow sense, a bit of a Band-Aid for a really dysfunctional primary care system,” Dr. Doctoroff said. “Ideally, if all you’re doing is providing a postdischarge physician visit, then you really want primary care to be able to do that in order to reengage with their patient. I think this is because postdischarge clinics are construed in a very narrow way to address the simple need to see a patient after discharge. And this may lead to the failure of these clinics, or make them easy to replace. Also, often what patients really need is more than just a physician visit, so a discharge clinic may need to be designed to provide an enhanced array of services.”
Dr. Fitterman said that these stories show that not all role expansion in hospital medicine is good role expansion. The experiences described by Dr. Manjarrez, Dr. Levine, and Dr. Doctoroff demonstrate the challenges hospitalists face as they attempt expansions into new roles, he said.
“We can’t be expected to fill all the cracks in primary care,” Dr. Fitterman said. “As a country we need to really prop up primary care. This all can’t come under the roof of hospital medicine. We need to be part of a patient-centered medical home – but we are not the patient-centered medical home.”
He said the experience with the preop clinic described by Dr. Manjarrez also shows the need for buy-in from hospital or health system administration.
“While most of us are employed by hospitals and want to help meet their needs, we have to be more cautious. We have to look, I think, with a more critical eye, for the value; it may not always be in the dollars coming back in,” he said. “It might be in cost avoidance, such as reducing readmissions, or reducing same-day cancellations in an OR. Unless the C-suite appreciates that value, such programs will be short-lived.”
SHM: My home as a pediatric hospitalist
As I began my career in pediatric hospital medicine at the Monroe Carell Jr. Children’s Hospital at Vanderbilt in Nashville, Tenn., I knew that I wanted a way to continue my education and to network with other hospitalists with interests in academics and pediatrics.
In 2010, I decided to attend a pre-course to the Society of Hospital Medicine’s annual conference that focused on academic hospital medicine, and my career has never been the same! I am thrilled to say I have found my professional home in SHM.
Here’s a quick list of the reasons SHM has been such a warm, welcoming home for me. I’ve highlighted the few options that stood out to me, but rest assured there is so much more from which to choose:
- Leadership opportunities in our Pediatrics Special Interest Group.
- Representation on the Annual Conference Committee to select pediatric-specific content as well as workshops on leadership, education, patient experience, and quality improvement.
- The Academic Hospitalist Academy, first as a pre-course before the SHM annual conference, and now as its own amazing meeting for academic pediatric hospital medicine providers.
- SHM’s Leadership Academy, a wonderful opportunity to learn leadership skills and network with other leaders. This year, it is in Vancouver!
- Participation in quality improvement initiatives like Pedi-BOOST, a care transitions program that specializes in pediatric patients.
- Traveling to Abu Dhabi and the Middle East Update in Hospital Medicine this March – being able to spread the latest trends in hospital medicine in the USA is one of the best experiences I have had with SHM!
Another reason SHM truly made me feel welcomed was the opportunity to attend the Pediatric Hospital Medicine (PHM) meeting. Each July, SHM helps to put on the largest gathering of pediatric hospital medicine providers. This year, it will be held in Atlanta from July 19-22.
This meeting is organized and supported by SHM, the American Academy of Pediatrics (AAP), and the Academic Pediatric Association (APA), and offers spectacular content in many tracks, including quality improvement, education, research, and the incredibly popular “Top Articles” presentation at lunch on Saturday. This session provides teaching materials that can span the year for Journal Clubs and resident and student education. The abstracts and poster sessions are top-notch and provide an opportunity for young and experienced providers to share their work.
The fourth annual Knowledge Café will be a highlight for me as well, as it allows collaboration and networking experiences in hot topics for early career hospitalists. How to strive for work/life balance, how to get the most out of your first meeting, and techniques for talking with your boss about difficult issues are some of the topics we plan to cover this year.
On top of this, networking and participation on various committees and work groups afforded me the opportunity to join the SHM Board of Directors in May of 2017. Having completed my first year on the Board, I have an even deeper appreciation for the progressive thinking of our leadership team and the amazing work that the staff of SHM does behind the scenes to help us maximize our memberships. I love the continuous process improvement that is happening with every Board meeting.
As a member of the Board, it’s important to keep tabs on the pulse of SHM members and their evolving needs. One way I have really enjoyed getting to learn about our membership is by attending local chapter meetings. I recently traveled to West Virginia and Connecticut, both of which have active, engaged chapters working to improve care in their local communities – it was so inspiring to have the opportunity to represent the organization, and I look forward to more meetings just like this. For our local chapter in Nashville, I have the honor of picking the venue for our meetings, which keeps me on my toes as I look for the latest hot spots in an incredibly happening city!
Last summer, the benefits of membership in SHM and my career choice of hospital medicine took on a whole new meaning. In July, just before PHM 2017, a meeting that I was lucky enough to chair, my husband started to feel the pain of a recurrent kidney stone as he was traveling with our four sons and their three friends. Can you imagine being on an airplane with seven elementary school–age boys when the worst pain EVER strikes?
I was home in Nashville thinking, “Who can I call to help him in Minneapolis?” My first thought was of fellow members of SHM with whom I’ve developed friendships over the years – other hospitalists like you and me. Many people came to mind, all of whom practice hospital medicine! A huge thank-you to our friend Dr. Shaun Frost, who rescued my husband, drove him to a local ED, AND took the seven boys out for lunch. I truly have never been so grateful!
My task for you is simple: Engage with the Society of Hospital Medicine! Come to a meeting, join a special interest group, connect with your local chapter, and make friends who can support you through your career – and, as evidenced by my husband’s experience – even in your personal life. It’s truly a special organization, and I can’t wait to share some experiences just like these with you.
Dr. Rehm is associate professor, pediatrics, and director, division of pediatric outreach medicine at Vanderbilt University and Monroe Carell Jr. Children’s Hospital at Vanderbilt, both in Nashville, Tenn. She is also a member of the SHM board of directors.
As I began my career in pediatric hospital medicine at the Monroe Carell Jr. Children’s Hospital at Vanderbilt in Nashville, Tenn., I knew that I wanted a way to continue my education and to network with other hospitalists with interests in academics and pediatrics.
In 2010, I decided to attend a pre-course to the Society of Hospital Medicine’s annual conference that focused on academic hospital medicine, and my career has never been the same! I am thrilled to say I have found my professional home in SHM.
Here’s a quick list of the reasons SHM has been such a warm, welcoming home for me. I’ve highlighted the few options that stood out to me, but rest assured there is so much more from which to choose:
- Leadership opportunities in our Pediatrics Special Interest Group.
- Representation on the Annual Conference Committee to select pediatric-specific content as well as workshops on leadership, education, patient experience, and quality improvement.
- The Academic Hospitalist Academy, first as a pre-course before the SHM annual conference, and now as its own amazing meeting for academic pediatric hospital medicine providers.
- SHM’s Leadership Academy, a wonderful opportunity to learn leadership skills and network with other leaders. This year, it is in Vancouver!
- Participation in quality improvement initiatives like Pedi-BOOST, a care transitions program that specializes in pediatric patients.
- Traveling to Abu Dhabi and the Middle East Update in Hospital Medicine this March – being able to spread the latest trends in hospital medicine in the USA is one of the best experiences I have had with SHM!
Another reason SHM truly made me feel welcomed was the opportunity to attend the Pediatric Hospital Medicine (PHM) meeting. Each July, SHM helps to put on the largest gathering of pediatric hospital medicine providers. This year, it will be held in Atlanta from July 19-22.
This meeting is organized and supported by SHM, the American Academy of Pediatrics (AAP), and the Academic Pediatric Association (APA), and offers spectacular content in many tracks, including quality improvement, education, research, and the incredibly popular “Top Articles” presentation at lunch on Saturday. This session provides teaching materials that can span the year for Journal Clubs and resident and student education. The abstracts and poster sessions are top-notch and provide an opportunity for young and experienced providers to share their work.
The fourth annual Knowledge Café will be a highlight for me as well, as it allows collaboration and networking experiences in hot topics for early career hospitalists. How to strive for work/life balance, how to get the most out of your first meeting, and techniques for talking with your boss about difficult issues are some of the topics we plan to cover this year.
On top of this, networking and participation on various committees and work groups afforded me the opportunity to join the SHM Board of Directors in May of 2017. Having completed my first year on the Board, I have an even deeper appreciation for the progressive thinking of our leadership team and the amazing work that the staff of SHM does behind the scenes to help us maximize our memberships. I love the continuous process improvement that is happening with every Board meeting.
As a member of the Board, it’s important to keep tabs on the pulse of SHM members and their evolving needs. One way I have really enjoyed getting to learn about our membership is by attending local chapter meetings. I recently traveled to West Virginia and Connecticut, both of which have active, engaged chapters working to improve care in their local communities – it was so inspiring to have the opportunity to represent the organization, and I look forward to more meetings just like this. For our local chapter in Nashville, I have the honor of picking the venue for our meetings, which keeps me on my toes as I look for the latest hot spots in an incredibly happening city!
Last summer, the benefits of membership in SHM and my career choice of hospital medicine took on a whole new meaning. In July, just before PHM 2017, a meeting that I was lucky enough to chair, my husband started to feel the pain of a recurrent kidney stone as he was traveling with our four sons and their three friends. Can you imagine being on an airplane with seven elementary school–age boys when the worst pain EVER strikes?
I was home in Nashville thinking, “Who can I call to help him in Minneapolis?” My first thought was of fellow members of SHM with whom I’ve developed friendships over the years – other hospitalists like you and me. Many people came to mind, all of whom practice hospital medicine! A huge thank-you to our friend Dr. Shaun Frost, who rescued my husband, drove him to a local ED, AND took the seven boys out for lunch. I truly have never been so grateful!
My task for you is simple: Engage with the Society of Hospital Medicine! Come to a meeting, join a special interest group, connect with your local chapter, and make friends who can support you through your career – and, as evidenced by my husband’s experience – even in your personal life. It’s truly a special organization, and I can’t wait to share some experiences just like these with you.
Dr. Rehm is associate professor, pediatrics, and director, division of pediatric outreach medicine at Vanderbilt University and Monroe Carell Jr. Children’s Hospital at Vanderbilt, both in Nashville, Tenn. She is also a member of the SHM board of directors.
As I began my career in pediatric hospital medicine at the Monroe Carell Jr. Children’s Hospital at Vanderbilt in Nashville, Tenn., I knew that I wanted a way to continue my education and to network with other hospitalists with interests in academics and pediatrics.
In 2010, I decided to attend a pre-course to the Society of Hospital Medicine’s annual conference that focused on academic hospital medicine, and my career has never been the same! I am thrilled to say I have found my professional home in SHM.
Here’s a quick list of the reasons SHM has been such a warm, welcoming home for me. I’ve highlighted the few options that stood out to me, but rest assured there is so much more from which to choose:
- Leadership opportunities in our Pediatrics Special Interest Group.
- Representation on the Annual Conference Committee to select pediatric-specific content as well as workshops on leadership, education, patient experience, and quality improvement.
- The Academic Hospitalist Academy, first as a pre-course before the SHM annual conference, and now as its own amazing meeting for academic pediatric hospital medicine providers.
- SHM’s Leadership Academy, a wonderful opportunity to learn leadership skills and network with other leaders. This year, it is in Vancouver!
- Participation in quality improvement initiatives like Pedi-BOOST, a care transitions program that specializes in pediatric patients.
- Traveling to Abu Dhabi and the Middle East Update in Hospital Medicine this March – being able to spread the latest trends in hospital medicine in the USA is one of the best experiences I have had with SHM!
Another reason SHM truly made me feel welcomed was the opportunity to attend the Pediatric Hospital Medicine (PHM) meeting. Each July, SHM helps to put on the largest gathering of pediatric hospital medicine providers. This year, it will be held in Atlanta from July 19-22.
This meeting is organized and supported by SHM, the American Academy of Pediatrics (AAP), and the Academic Pediatric Association (APA), and offers spectacular content in many tracks, including quality improvement, education, research, and the incredibly popular “Top Articles” presentation at lunch on Saturday. This session provides teaching materials that can span the year for Journal Clubs and resident and student education. The abstracts and poster sessions are top-notch and provide an opportunity for young and experienced providers to share their work.
The fourth annual Knowledge Café will be a highlight for me as well, as it allows collaboration and networking experiences in hot topics for early career hospitalists. How to strive for work/life balance, how to get the most out of your first meeting, and techniques for talking with your boss about difficult issues are some of the topics we plan to cover this year.
On top of this, networking and participation on various committees and work groups afforded me the opportunity to join the SHM Board of Directors in May of 2017. Having completed my first year on the Board, I have an even deeper appreciation for the progressive thinking of our leadership team and the amazing work that the staff of SHM does behind the scenes to help us maximize our memberships. I love the continuous process improvement that is happening with every Board meeting.
As a member of the Board, it’s important to keep tabs on the pulse of SHM members and their evolving needs. One way I have really enjoyed getting to learn about our membership is by attending local chapter meetings. I recently traveled to West Virginia and Connecticut, both of which have active, engaged chapters working to improve care in their local communities – it was so inspiring to have the opportunity to represent the organization, and I look forward to more meetings just like this. For our local chapter in Nashville, I have the honor of picking the venue for our meetings, which keeps me on my toes as I look for the latest hot spots in an incredibly happening city!
Last summer, the benefits of membership in SHM and my career choice of hospital medicine took on a whole new meaning. In July, just before PHM 2017, a meeting that I was lucky enough to chair, my husband started to feel the pain of a recurrent kidney stone as he was traveling with our four sons and their three friends. Can you imagine being on an airplane with seven elementary school–age boys when the worst pain EVER strikes?
I was home in Nashville thinking, “Who can I call to help him in Minneapolis?” My first thought was of fellow members of SHM with whom I’ve developed friendships over the years – other hospitalists like you and me. Many people came to mind, all of whom practice hospital medicine! A huge thank-you to our friend Dr. Shaun Frost, who rescued my husband, drove him to a local ED, AND took the seven boys out for lunch. I truly have never been so grateful!
My task for you is simple: Engage with the Society of Hospital Medicine! Come to a meeting, join a special interest group, connect with your local chapter, and make friends who can support you through your career – and, as evidenced by my husband’s experience – even in your personal life. It’s truly a special organization, and I can’t wait to share some experiences just like these with you.
Dr. Rehm is associate professor, pediatrics, and director, division of pediatric outreach medicine at Vanderbilt University and Monroe Carell Jr. Children’s Hospital at Vanderbilt, both in Nashville, Tenn. She is also a member of the SHM board of directors.
The rapidly disappearing community pediatric inpatient unit
Greed kills babies. Children’s lives matter. Children over profit.
These were the slogans proclaimed by signs carried by protesters outside of MedStar Franklin Square Medical Center in Baltimore in early May of 2018 to protest the closure of the dedicated pediatric emergency department and inpatient pediatric unit.
But administrators at Franklin Square Medical Center had made their decision long before the glue had dried on the signs, and the protests of patients and community officials fell on deaf ears. Eight doctors and 30 other staff had already lost their jobs, including the chair of pediatrics, Scott Krugman, MD.1
And this was just another drop in a slow ooze of pediatric inpatient units based in community hospitals that have seen the ax fall on what was thought to be a vital medical resource for their communities – yet not vital enough to survive its lack of profitability. From Taunton, Mass., to Chicago, Ill., to rural Tennessee, pediatric inpatient units in community hospitals have failed to even flirt with breaking even, let alone show profitability. Many community pediatric inpatient units are saddled with rock-bottom reimbursements offered by state Medicaid programs, the overwhelmingly prevalent payer for pediatric hospitalizations, which is compounded by the seasonality and unpredictability of pediatric inpatient volumes, so many have seen a glowing red bottom line lead to their demise.
What does this mean for pediatric health in underserved and rural communities? The closure of the pediatric inpatient unit at MedStar Franklin Square Medical Center meant the loss of physicians and nurses staffing the child protection team helping to assist the local district attorney in child abuse cases. Sometimes described as “secondary care,” community pediatric hospitalists also serve as a link between primary care providers and tertiary care subspecialists; they can serve as pediatric generalists throughout a hospital and provide newborn nursery care, delivery room resuscitations, ED consultations, procedural sedations, psychiatric unit support, surgical comanagement, and informal or formal outpatient consultations.2 Losing even a small inpatient pediatric unit can have a ripple effect on inpatient and outpatient pediatric services in a health system and community.
For patients and their caregivers, the loss of pediatric inpatient services in their community hospital can erect additional hurdles to appropriate health care. The need to travel longer distances to urban centers or even the other side of town can be challenging given the difficulties posed by long distances, traffic congestion, public transportation, or just parking.3 For patients suffering from longer hospitalizations caused by medical complexity or chronic illnesses, traveling long distances can exacerbate the caregiver stress from attempting to care for a family at home while participating in the care of a hospitalized child. Longer travel times can also worsen family stress by increasing a caregiver’s absence from home and increased nonmedical expenses, not to mention loss of wages.4 Comfort levels with inpatient providers can also suffer because most pediatric units in community hospitals are staffed by either community general pediatricians or very small pediatric hospitalist groups, which breeds familiarity with frequently admitted patients and their caregivers. This familiarity can be lacking in large academic centers, with confusing and ever-rotating teams of academic hospitalists, residents, and medical students.5
What is driving the slow drumbeat of pediatric inpatient unit closures? On a macroeconomic scale, pediatric hospitalizations have been dropping yearly, driven down by immunizations (despite the best efforts of certain celebrities), antibiotic stewardship, and improved access to outpatient care. In 2006, there were 6.6 million hospitalizations for children aged 17 years and younger,6 but by 2012 this had dropped to 5.9 million hospitalizations.7 In the same age group, the rate of hospitalization from the ED dropped from 4.4% in 2006 to 3.2% in 2015.8
On a hospital level, the presence of multiple small pediatric units in a region may not make sense from a cost standpoint, and a larger, merged unit may provide higher quality because of its higher volumes. On a state and local level, alternative payment models have been implemented with the best of intentions but have led administrators at community general hospitals to look at pediatric units as the lowest hanging money-losing fruit in their efforts to survive a brave new world of hospital payment.
The most extreme (or advanced, depending on your viewpoint) model is in Maryland: Since 2014, acute care hospitals have been only able to receive a fixed amount of revenue from all payers, including Medicare, Medicaid, and commercial insurers.9 Known as an all-payer global budget, it incentivizes lowering unnecessary costs of care, such as readmissions, but also encourages cauterization of cost centers hemorrhaging money – such as inpatient pediatrics. Even the venerable Johns Hopkins Children’s Center has seen its profitability pale in comparison to the expansion team Johns Hopkins All Children’s Hospital in St. Petersburg, Fla., which is the second-most profitable hospital in the Hopkins system, only edged out by Sibley Memorial Hospital – which also sports an out-of-state location in the District of Columbia.10
But all hope is not lost for your comfy local pediatric inpatient unit. In other states and regions where a more favorable (to hospitals) payer mix exists, large pediatric hospitals are still engaged in turf battles with other local competitors to grab market share. In these regions, community pediatric units have survived by partnering with large pediatric institutions, either through affiliations or wholesale transplantation of the larger pediatric institution’s providers, nurses, and EHRs into essentially what is a leased floor. In addition, large pediatric institutions that participate in capitated models such as accountable care organizations have paradoxically found it financially favorable to direct “bread-and-butter” pediatric hospitalizations to community pediatric units, which often provide the same care at a lower cost.
Utilizing community inpatient pediatric units was “initially … a means of expanding their market share and ‘downstream’ revenue from transfers, but more commonly now [is] a way of alleviating the costs associated with admitting low to moderate acuity patients to the main tertiary sites,” said Francisco Alvarez, MD, associate chief of Regional Pediatric Hospital Medicine Programs at Lucile Packard Children’s Hospital in Palo Alto, Calif. “The cost of care provided by pediatric hospitals has always been higher than the average cost for nonpediatric hospitals in regard to caring for pediatric patients due to their highly skilled specialties and services. These have become more scrutinized by private and government insurance plans and, in some cases, have led to lower reimbursements and therefore a lower or deficient net revenue for certain patient populations.”
For community pediatric hospitalists, the shifting sands of reimbursement on which pediatric inpatient care is built can be a motion illness–inducing experience. In addition to concerns over community health care, job security, and population health, care provided in community hospitals can often be subtly undercut by tertiary and quaternary care pediatric hospitals.
“The focus of pediatric residency programs in freestanding children’s hospitals has created a situation where new pediatricians have less opportunity to develop respect for community pediatric hospital medicine,” said Beth Natt, MD, director of pediatric hospital medicine in the Regional Programs at Connecticut Children’s Medical Center, Hartford. “We are the nameless ‘OSH,’ the place that gets ‘Monday-morning quarterbacked’ in resident morning reports without having a voice at the table. Add this to residents learning ‘only’ protocolized care as opposed to a spectrum of appropriate care, and we create a culture of ‘wrong and right’ with the backward nonprotocol driven community docs looking like they are practicing medicine in the Wild West.”
What’s a community pediatric hospitalist to do, faced with an uncertain future and diminishing respect? Continuing to partner with local pediatric providers, community leaders, and local health care advocacy groups will help to enmesh inpatient providers in the fabric of a community’s health care. But making the value case to hospital administrators is critical for community pediatric hospitalists, as adult hospitalists realized soon after the inception of the hospitalist field.
Goals valued by hospital administrators are pursued on a daily basis by community pediatric hospitalists, and these successes need to be brought to light. Achieving value and quality metrics, pursuing high-value care, reducing readmission rates, championing EHRs, and improving documentation are goals that community pediatric hospitalists and hospital administrators can work toward together.11 By pursuing and sharing success in meeting these shared goals, perhaps the local community pediatric inpatient unit can survive – and thrive.
As for Dr. Krugman, he has moved on and is soon to be gainfully employed again. But he continues to be focused, as always, on the health of his patients.
“What are we going to do to take care of kids in their own communities?” Dr. Krugman asked. “It’s going to be an increasing challenge over the next decade due to the consolidation of children’s hospitals and low payments, especially for hospitals that are adult-focused. Unless we find a way to pay for pediatric care as a country.”
Dr. Chang is a pediatric hospitalist at Baystate Children’s Hospital in Springfield, Mass., and is the pediatric editor of The Hospitalist.
References
1. McDaniels A. (2018). Protesters denounce reduction in pediatric services at Baltimore’s MedStar Franklin Square hospital. Baltimore Sun. Available at: http://www.baltimoresun.com/health/health-care/bs-hs-franklin-square-hospital-protest-20180508-story.html.
2. Roberts KB. Pediatric hospitalists in community hospitals: Hospital-based generalists with expanded roles. Hosp Pediatr. 2015 May;5(5):290-2.
3. Georgia Health News. (2018). A hospital crisis is killing rural communities. This state is ‘Ground Zero’. Available at: http://www.georgiahealthnews.com/2017/09/hospital-crisis-killing-rural-communities-state-ground-zero/.
4. DiFazio RL et al. Non-medical out-of-pocket expenses incurred by families during their child’s hospitalization. J Child Health Care. 2013 Sep;17(3):230-41.
5. Gunderman R. Hospitalist and the decline of comprehensive care. N Engl J Med. 2016 Sep 15; 375(11):1011-3.
6. Statistical Brief #56. (2018). Retrieved from https://www.hcup-us.ahrq.gov/reports/statbriefs/sb56.jsp.
7. Overview of Hospital Stays for Children in the United States, 2012 #187. (2018). Retrieved from https://www.hcup-us.ahrq.gov/reports/statbriefs/sb187-Hospital-Stays-Children-2012.jsp.
8. Trends in Hospital Inpatient Stays by Age and Payer, 2000-2015 #235. (2018). Retrieved from https://www.hcup-us.ahrq.gov/reports/statbriefs/sb235-Inpatient-Stays-Age-Payer-Trends.jsp.
9. Maryland All-Payer Model | Center for Medicare & Medicaid Innovation. (2018). Retrieved from https://innovation.cms.gov/initiatives/Maryland-All-Payer-Model/.
10. The effects of Maryland’s unique health care system. (2018). Retrieved from https://www.axios.com/johns-hopkins-finances-maryland-1518553853-722c2195-731e-4e02-ab1e-94e4211ba945.html.
11. The Increasing Need for Hospitalist Programs to Demonstrate Value | SCP. (2018). Retrieved from https://www.schumacherclinical.com/providers/blog/the-increasing-need-for-hospitalist-programs-to-demonstrate-value.
Greed kills babies. Children’s lives matter. Children over profit.
These were the slogans proclaimed by signs carried by protesters outside of MedStar Franklin Square Medical Center in Baltimore in early May of 2018 to protest the closure of the dedicated pediatric emergency department and inpatient pediatric unit.
But administrators at Franklin Square Medical Center had made their decision long before the glue had dried on the signs, and the protests of patients and community officials fell on deaf ears. Eight doctors and 30 other staff had already lost their jobs, including the chair of pediatrics, Scott Krugman, MD.1
And this was just another drop in a slow ooze of pediatric inpatient units based in community hospitals that have seen the ax fall on what was thought to be a vital medical resource for their communities – yet not vital enough to survive its lack of profitability. From Taunton, Mass., to Chicago, Ill., to rural Tennessee, pediatric inpatient units in community hospitals have failed to even flirt with breaking even, let alone show profitability. Many community pediatric inpatient units are saddled with rock-bottom reimbursements offered by state Medicaid programs, the overwhelmingly prevalent payer for pediatric hospitalizations, which is compounded by the seasonality and unpredictability of pediatric inpatient volumes, so many have seen a glowing red bottom line lead to their demise.
What does this mean for pediatric health in underserved and rural communities? The closure of the pediatric inpatient unit at MedStar Franklin Square Medical Center meant the loss of physicians and nurses staffing the child protection team helping to assist the local district attorney in child abuse cases. Sometimes described as “secondary care,” community pediatric hospitalists also serve as a link between primary care providers and tertiary care subspecialists; they can serve as pediatric generalists throughout a hospital and provide newborn nursery care, delivery room resuscitations, ED consultations, procedural sedations, psychiatric unit support, surgical comanagement, and informal or formal outpatient consultations.2 Losing even a small inpatient pediatric unit can have a ripple effect on inpatient and outpatient pediatric services in a health system and community.
For patients and their caregivers, the loss of pediatric inpatient services in their community hospital can erect additional hurdles to appropriate health care. The need to travel longer distances to urban centers or even the other side of town can be challenging given the difficulties posed by long distances, traffic congestion, public transportation, or just parking.3 For patients suffering from longer hospitalizations caused by medical complexity or chronic illnesses, traveling long distances can exacerbate the caregiver stress from attempting to care for a family at home while participating in the care of a hospitalized child. Longer travel times can also worsen family stress by increasing a caregiver’s absence from home and increased nonmedical expenses, not to mention loss of wages.4 Comfort levels with inpatient providers can also suffer because most pediatric units in community hospitals are staffed by either community general pediatricians or very small pediatric hospitalist groups, which breeds familiarity with frequently admitted patients and their caregivers. This familiarity can be lacking in large academic centers, with confusing and ever-rotating teams of academic hospitalists, residents, and medical students.5
What is driving the slow drumbeat of pediatric inpatient unit closures? On a macroeconomic scale, pediatric hospitalizations have been dropping yearly, driven down by immunizations (despite the best efforts of certain celebrities), antibiotic stewardship, and improved access to outpatient care. In 2006, there were 6.6 million hospitalizations for children aged 17 years and younger,6 but by 2012 this had dropped to 5.9 million hospitalizations.7 In the same age group, the rate of hospitalization from the ED dropped from 4.4% in 2006 to 3.2% in 2015.8
On a hospital level, the presence of multiple small pediatric units in a region may not make sense from a cost standpoint, and a larger, merged unit may provide higher quality because of its higher volumes. On a state and local level, alternative payment models have been implemented with the best of intentions but have led administrators at community general hospitals to look at pediatric units as the lowest hanging money-losing fruit in their efforts to survive a brave new world of hospital payment.
The most extreme (or advanced, depending on your viewpoint) model is in Maryland: Since 2014, acute care hospitals have been only able to receive a fixed amount of revenue from all payers, including Medicare, Medicaid, and commercial insurers.9 Known as an all-payer global budget, it incentivizes lowering unnecessary costs of care, such as readmissions, but also encourages cauterization of cost centers hemorrhaging money – such as inpatient pediatrics. Even the venerable Johns Hopkins Children’s Center has seen its profitability pale in comparison to the expansion team Johns Hopkins All Children’s Hospital in St. Petersburg, Fla., which is the second-most profitable hospital in the Hopkins system, only edged out by Sibley Memorial Hospital – which also sports an out-of-state location in the District of Columbia.10
But all hope is not lost for your comfy local pediatric inpatient unit. In other states and regions where a more favorable (to hospitals) payer mix exists, large pediatric hospitals are still engaged in turf battles with other local competitors to grab market share. In these regions, community pediatric units have survived by partnering with large pediatric institutions, either through affiliations or wholesale transplantation of the larger pediatric institution’s providers, nurses, and EHRs into essentially what is a leased floor. In addition, large pediatric institutions that participate in capitated models such as accountable care organizations have paradoxically found it financially favorable to direct “bread-and-butter” pediatric hospitalizations to community pediatric units, which often provide the same care at a lower cost.
Utilizing community inpatient pediatric units was “initially … a means of expanding their market share and ‘downstream’ revenue from transfers, but more commonly now [is] a way of alleviating the costs associated with admitting low to moderate acuity patients to the main tertiary sites,” said Francisco Alvarez, MD, associate chief of Regional Pediatric Hospital Medicine Programs at Lucile Packard Children’s Hospital in Palo Alto, Calif. “The cost of care provided by pediatric hospitals has always been higher than the average cost for nonpediatric hospitals in regard to caring for pediatric patients due to their highly skilled specialties and services. These have become more scrutinized by private and government insurance plans and, in some cases, have led to lower reimbursements and therefore a lower or deficient net revenue for certain patient populations.”
For community pediatric hospitalists, the shifting sands of reimbursement on which pediatric inpatient care is built can be a motion illness–inducing experience. In addition to concerns over community health care, job security, and population health, care provided in community hospitals can often be subtly undercut by tertiary and quaternary care pediatric hospitals.
“The focus of pediatric residency programs in freestanding children’s hospitals has created a situation where new pediatricians have less opportunity to develop respect for community pediatric hospital medicine,” said Beth Natt, MD, director of pediatric hospital medicine in the Regional Programs at Connecticut Children’s Medical Center, Hartford. “We are the nameless ‘OSH,’ the place that gets ‘Monday-morning quarterbacked’ in resident morning reports without having a voice at the table. Add this to residents learning ‘only’ protocolized care as opposed to a spectrum of appropriate care, and we create a culture of ‘wrong and right’ with the backward nonprotocol driven community docs looking like they are practicing medicine in the Wild West.”
What’s a community pediatric hospitalist to do, faced with an uncertain future and diminishing respect? Continuing to partner with local pediatric providers, community leaders, and local health care advocacy groups will help to enmesh inpatient providers in the fabric of a community’s health care. But making the value case to hospital administrators is critical for community pediatric hospitalists, as adult hospitalists realized soon after the inception of the hospitalist field.
Goals valued by hospital administrators are pursued on a daily basis by community pediatric hospitalists, and these successes need to be brought to light. Achieving value and quality metrics, pursuing high-value care, reducing readmission rates, championing EHRs, and improving documentation are goals that community pediatric hospitalists and hospital administrators can work toward together.11 By pursuing and sharing success in meeting these shared goals, perhaps the local community pediatric inpatient unit can survive – and thrive.
As for Dr. Krugman, he has moved on and is soon to be gainfully employed again. But he continues to be focused, as always, on the health of his patients.
“What are we going to do to take care of kids in their own communities?” Dr. Krugman asked. “It’s going to be an increasing challenge over the next decade due to the consolidation of children’s hospitals and low payments, especially for hospitals that are adult-focused. Unless we find a way to pay for pediatric care as a country.”
Dr. Chang is a pediatric hospitalist at Baystate Children’s Hospital in Springfield, Mass., and is the pediatric editor of The Hospitalist.
References
1. McDaniels A. (2018). Protesters denounce reduction in pediatric services at Baltimore’s MedStar Franklin Square hospital. Baltimore Sun. Available at: http://www.baltimoresun.com/health/health-care/bs-hs-franklin-square-hospital-protest-20180508-story.html.
2. Roberts KB. Pediatric hospitalists in community hospitals: Hospital-based generalists with expanded roles. Hosp Pediatr. 2015 May;5(5):290-2.
3. Georgia Health News. (2018). A hospital crisis is killing rural communities. This state is ‘Ground Zero’. Available at: http://www.georgiahealthnews.com/2017/09/hospital-crisis-killing-rural-communities-state-ground-zero/.
4. DiFazio RL et al. Non-medical out-of-pocket expenses incurred by families during their child’s hospitalization. J Child Health Care. 2013 Sep;17(3):230-41.
5. Gunderman R. Hospitalist and the decline of comprehensive care. N Engl J Med. 2016 Sep 15; 375(11):1011-3.
6. Statistical Brief #56. (2018). Retrieved from https://www.hcup-us.ahrq.gov/reports/statbriefs/sb56.jsp.
7. Overview of Hospital Stays for Children in the United States, 2012 #187. (2018). Retrieved from https://www.hcup-us.ahrq.gov/reports/statbriefs/sb187-Hospital-Stays-Children-2012.jsp.
8. Trends in Hospital Inpatient Stays by Age and Payer, 2000-2015 #235. (2018). Retrieved from https://www.hcup-us.ahrq.gov/reports/statbriefs/sb235-Inpatient-Stays-Age-Payer-Trends.jsp.
9. Maryland All-Payer Model | Center for Medicare & Medicaid Innovation. (2018). Retrieved from https://innovation.cms.gov/initiatives/Maryland-All-Payer-Model/.
10. The effects of Maryland’s unique health care system. (2018). Retrieved from https://www.axios.com/johns-hopkins-finances-maryland-1518553853-722c2195-731e-4e02-ab1e-94e4211ba945.html.
11. The Increasing Need for Hospitalist Programs to Demonstrate Value | SCP. (2018). Retrieved from https://www.schumacherclinical.com/providers/blog/the-increasing-need-for-hospitalist-programs-to-demonstrate-value.
Greed kills babies. Children’s lives matter. Children over profit.
These were the slogans proclaimed by signs carried by protesters outside of MedStar Franklin Square Medical Center in Baltimore in early May of 2018 to protest the closure of the dedicated pediatric emergency department and inpatient pediatric unit.
But administrators at Franklin Square Medical Center had made their decision long before the glue had dried on the signs, and the protests of patients and community officials fell on deaf ears. Eight doctors and 30 other staff had already lost their jobs, including the chair of pediatrics, Scott Krugman, MD.1
And this was just another drop in a slow ooze of pediatric inpatient units based in community hospitals that have seen the ax fall on what was thought to be a vital medical resource for their communities – yet not vital enough to survive its lack of profitability. From Taunton, Mass., to Chicago, Ill., to rural Tennessee, pediatric inpatient units in community hospitals have failed to even flirt with breaking even, let alone show profitability. Many community pediatric inpatient units are saddled with rock-bottom reimbursements offered by state Medicaid programs, the overwhelmingly prevalent payer for pediatric hospitalizations, which is compounded by the seasonality and unpredictability of pediatric inpatient volumes, so many have seen a glowing red bottom line lead to their demise.
What does this mean for pediatric health in underserved and rural communities? The closure of the pediatric inpatient unit at MedStar Franklin Square Medical Center meant the loss of physicians and nurses staffing the child protection team helping to assist the local district attorney in child abuse cases. Sometimes described as “secondary care,” community pediatric hospitalists also serve as a link between primary care providers and tertiary care subspecialists; they can serve as pediatric generalists throughout a hospital and provide newborn nursery care, delivery room resuscitations, ED consultations, procedural sedations, psychiatric unit support, surgical comanagement, and informal or formal outpatient consultations.2 Losing even a small inpatient pediatric unit can have a ripple effect on inpatient and outpatient pediatric services in a health system and community.
For patients and their caregivers, the loss of pediatric inpatient services in their community hospital can erect additional hurdles to appropriate health care. The need to travel longer distances to urban centers or even the other side of town can be challenging given the difficulties posed by long distances, traffic congestion, public transportation, or just parking.3 For patients suffering from longer hospitalizations caused by medical complexity or chronic illnesses, traveling long distances can exacerbate the caregiver stress from attempting to care for a family at home while participating in the care of a hospitalized child. Longer travel times can also worsen family stress by increasing a caregiver’s absence from home and increased nonmedical expenses, not to mention loss of wages.4 Comfort levels with inpatient providers can also suffer because most pediatric units in community hospitals are staffed by either community general pediatricians or very small pediatric hospitalist groups, which breeds familiarity with frequently admitted patients and their caregivers. This familiarity can be lacking in large academic centers, with confusing and ever-rotating teams of academic hospitalists, residents, and medical students.5
What is driving the slow drumbeat of pediatric inpatient unit closures? On a macroeconomic scale, pediatric hospitalizations have been dropping yearly, driven down by immunizations (despite the best efforts of certain celebrities), antibiotic stewardship, and improved access to outpatient care. In 2006, there were 6.6 million hospitalizations for children aged 17 years and younger,6 but by 2012 this had dropped to 5.9 million hospitalizations.7 In the same age group, the rate of hospitalization from the ED dropped from 4.4% in 2006 to 3.2% in 2015.8
On a hospital level, the presence of multiple small pediatric units in a region may not make sense from a cost standpoint, and a larger, merged unit may provide higher quality because of its higher volumes. On a state and local level, alternative payment models have been implemented with the best of intentions but have led administrators at community general hospitals to look at pediatric units as the lowest hanging money-losing fruit in their efforts to survive a brave new world of hospital payment.
The most extreme (or advanced, depending on your viewpoint) model is in Maryland: Since 2014, acute care hospitals have been only able to receive a fixed amount of revenue from all payers, including Medicare, Medicaid, and commercial insurers.9 Known as an all-payer global budget, it incentivizes lowering unnecessary costs of care, such as readmissions, but also encourages cauterization of cost centers hemorrhaging money – such as inpatient pediatrics. Even the venerable Johns Hopkins Children’s Center has seen its profitability pale in comparison to the expansion team Johns Hopkins All Children’s Hospital in St. Petersburg, Fla., which is the second-most profitable hospital in the Hopkins system, only edged out by Sibley Memorial Hospital – which also sports an out-of-state location in the District of Columbia.10
But all hope is not lost for your comfy local pediatric inpatient unit. In other states and regions where a more favorable (to hospitals) payer mix exists, large pediatric hospitals are still engaged in turf battles with other local competitors to grab market share. In these regions, community pediatric units have survived by partnering with large pediatric institutions, either through affiliations or wholesale transplantation of the larger pediatric institution’s providers, nurses, and EHRs into essentially what is a leased floor. In addition, large pediatric institutions that participate in capitated models such as accountable care organizations have paradoxically found it financially favorable to direct “bread-and-butter” pediatric hospitalizations to community pediatric units, which often provide the same care at a lower cost.
Utilizing community inpatient pediatric units was “initially … a means of expanding their market share and ‘downstream’ revenue from transfers, but more commonly now [is] a way of alleviating the costs associated with admitting low to moderate acuity patients to the main tertiary sites,” said Francisco Alvarez, MD, associate chief of Regional Pediatric Hospital Medicine Programs at Lucile Packard Children’s Hospital in Palo Alto, Calif. “The cost of care provided by pediatric hospitals has always been higher than the average cost for nonpediatric hospitals in regard to caring for pediatric patients due to their highly skilled specialties and services. These have become more scrutinized by private and government insurance plans and, in some cases, have led to lower reimbursements and therefore a lower or deficient net revenue for certain patient populations.”
For community pediatric hospitalists, the shifting sands of reimbursement on which pediatric inpatient care is built can be a motion illness–inducing experience. In addition to concerns over community health care, job security, and population health, care provided in community hospitals can often be subtly undercut by tertiary and quaternary care pediatric hospitals.
“The focus of pediatric residency programs in freestanding children’s hospitals has created a situation where new pediatricians have less opportunity to develop respect for community pediatric hospital medicine,” said Beth Natt, MD, director of pediatric hospital medicine in the Regional Programs at Connecticut Children’s Medical Center, Hartford. “We are the nameless ‘OSH,’ the place that gets ‘Monday-morning quarterbacked’ in resident morning reports without having a voice at the table. Add this to residents learning ‘only’ protocolized care as opposed to a spectrum of appropriate care, and we create a culture of ‘wrong and right’ with the backward nonprotocol driven community docs looking like they are practicing medicine in the Wild West.”
What’s a community pediatric hospitalist to do, faced with an uncertain future and diminishing respect? Continuing to partner with local pediatric providers, community leaders, and local health care advocacy groups will help to enmesh inpatient providers in the fabric of a community’s health care. But making the value case to hospital administrators is critical for community pediatric hospitalists, as adult hospitalists realized soon after the inception of the hospitalist field.
Goals valued by hospital administrators are pursued on a daily basis by community pediatric hospitalists, and these successes need to be brought to light. Achieving value and quality metrics, pursuing high-value care, reducing readmission rates, championing EHRs, and improving documentation are goals that community pediatric hospitalists and hospital administrators can work toward together.11 By pursuing and sharing success in meeting these shared goals, perhaps the local community pediatric inpatient unit can survive – and thrive.
As for Dr. Krugman, he has moved on and is soon to be gainfully employed again. But he continues to be focused, as always, on the health of his patients.
“What are we going to do to take care of kids in their own communities?” Dr. Krugman asked. “It’s going to be an increasing challenge over the next decade due to the consolidation of children’s hospitals and low payments, especially for hospitals that are adult-focused. Unless we find a way to pay for pediatric care as a country.”
Dr. Chang is a pediatric hospitalist at Baystate Children’s Hospital in Springfield, Mass., and is the pediatric editor of The Hospitalist.
References
1. McDaniels A. (2018). Protesters denounce reduction in pediatric services at Baltimore’s MedStar Franklin Square hospital. Baltimore Sun. Available at: http://www.baltimoresun.com/health/health-care/bs-hs-franklin-square-hospital-protest-20180508-story.html.
2. Roberts KB. Pediatric hospitalists in community hospitals: Hospital-based generalists with expanded roles. Hosp Pediatr. 2015 May;5(5):290-2.
3. Georgia Health News. (2018). A hospital crisis is killing rural communities. This state is ‘Ground Zero’. Available at: http://www.georgiahealthnews.com/2017/09/hospital-crisis-killing-rural-communities-state-ground-zero/.
4. DiFazio RL et al. Non-medical out-of-pocket expenses incurred by families during their child’s hospitalization. J Child Health Care. 2013 Sep;17(3):230-41.
5. Gunderman R. Hospitalist and the decline of comprehensive care. N Engl J Med. 2016 Sep 15; 375(11):1011-3.
6. Statistical Brief #56. (2018). Retrieved from https://www.hcup-us.ahrq.gov/reports/statbriefs/sb56.jsp.
7. Overview of Hospital Stays for Children in the United States, 2012 #187. (2018). Retrieved from https://www.hcup-us.ahrq.gov/reports/statbriefs/sb187-Hospital-Stays-Children-2012.jsp.
8. Trends in Hospital Inpatient Stays by Age and Payer, 2000-2015 #235. (2018). Retrieved from https://www.hcup-us.ahrq.gov/reports/statbriefs/sb235-Inpatient-Stays-Age-Payer-Trends.jsp.
9. Maryland All-Payer Model | Center for Medicare & Medicaid Innovation. (2018). Retrieved from https://innovation.cms.gov/initiatives/Maryland-All-Payer-Model/.
10. The effects of Maryland’s unique health care system. (2018). Retrieved from https://www.axios.com/johns-hopkins-finances-maryland-1518553853-722c2195-731e-4e02-ab1e-94e4211ba945.html.
11. The Increasing Need for Hospitalist Programs to Demonstrate Value | SCP. (2018). Retrieved from https://www.schumacherclinical.com/providers/blog/the-increasing-need-for-hospitalist-programs-to-demonstrate-value.
Closed-loop insulin control for T2DM is feasible in hospital setting
ORLANDO – (T2DM).
The findings, released at the annual scientific sessions of the American Diabetes Association and via simultaneous publication in The New England Journal of Medicine, don’t examine cost or clinical outcomes. However, “our results suggest this new technology might be another approach to manage in-patient hypoglycemia in a safe and effective way, lead author Lia Bally, MD, PhD, of the division of endocrinology, diabetes, and clinical nutrition, Bern (Switzerland ) University Hospital, said in an interview.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
For the open-label trial, the researchers recruited 136 adults with T2DM under noncritical care at two hospitals (one in the England and the other in Switzerland). Some patients had undergone surgery, Dr. Bally said, and some others were being treated for systemic infections. Comorbidities were significantly more severe in the closed-loop group, and 43% had sepsis.
All of the subjects required subcutaneous insulin therapy.
From 2016 to 2017, patients were randomly assigned to receive normal subcutaneous insulin therapy (n = 70) or closed-loop insulin delivery (n = 66).
It took about 15 minutes to perform the procedure to implement the closed-loop insulin delivery system, Dr. Bally said. It featured a subcutaneous cannula inserted into the abdomen, a continuous glucose monitor (a device also used in the control group), and a trial insulin pump.
This was not a hybrid system, and it did not include prandial insulin boluses or input of the timing and carbohydrate content of meals. One reason behind the choice to adopt a fully automated system was to relieve the burden on both health care professionals and patients, coauthor Hood Thabit, PhD, of Wellcome Trust–MRC Institute of Metabolic Science, the Manchester Academic Health Science Center, and University of Manchester, said in an interview.
For up to 15 days or until discharge, researchers tracked how much of the time sensor glucose measurements were in a target range of 100 mg/dL to 180 mg/dL.
In the closed-loop group, glucose measurements were in the target range 66 mg/dL ± 17% of the time compared to 42 mg/dL ± 17% in the control group, a difference of 24 mg/dL ± 3% (95% confidence interval, 19-30; P less than .001).
For the closed-loop group, the average glucose level was 154 mg/dL, and it was 188 mg/dL in the control group (P less than .001).
The researchers didn’t find a statistically significant difference between the groups in duration of hypoglycemia or amount of insulin delivered.
None of the patients suffered from severe hypoglycemia or clinically significant hyperglycemia with ketonemia.
There were 18 incidents of clinically significant hyperglycemia events (capillary glucose levels of more than 360 mg/dL) in the closed-loop group, compared with 41 such events in the control group. (P = .03)
Three patients in each group had adverse trial-related device effects.
Of 62 patients in the closed-loop group who completed the trial, 87% reported being pleased by their glucose levels, and all but one reported being happy to have their levels monitored automatically. All 62 patients said they’d recommend the system to others.
Going forward, the researchers hope to launch a multicenter trial that will examine clinical outcomes such as postoperative complications, infections, mortality, and glucose control after hospital discharge, according to Dr. Bally.
The study was supported by Diabetes UK, the Swiss National Science Foundation, the European Foundation for the Study of Diabetes, the JDRF, the National Institute for Health Research Cambridge Biomedical Research Center, and a Wellcome Strategic Award. Abbott Diabetes Care supplied equipment and guidance regarding connectivity, and representatives reviewed the manuscript before submission.
Dr. Bally reported funding from the University Hospital Bern, University of Bern and the Swiss Diabetes Foundation. Dr. Thabit reported no disclosures. Other authors report no disclosures or various disclosures.
SOURCE: Bally L et al. ADA 2018 Abstract 350-OR. Published simultaneously in The New England Journal of Medicine. June 25, 2018
ORLANDO – (T2DM).
The findings, released at the annual scientific sessions of the American Diabetes Association and via simultaneous publication in The New England Journal of Medicine, don’t examine cost or clinical outcomes. However, “our results suggest this new technology might be another approach to manage in-patient hypoglycemia in a safe and effective way, lead author Lia Bally, MD, PhD, of the division of endocrinology, diabetes, and clinical nutrition, Bern (Switzerland ) University Hospital, said in an interview.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
For the open-label trial, the researchers recruited 136 adults with T2DM under noncritical care at two hospitals (one in the England and the other in Switzerland). Some patients had undergone surgery, Dr. Bally said, and some others were being treated for systemic infections. Comorbidities were significantly more severe in the closed-loop group, and 43% had sepsis.
All of the subjects required subcutaneous insulin therapy.
From 2016 to 2017, patients were randomly assigned to receive normal subcutaneous insulin therapy (n = 70) or closed-loop insulin delivery (n = 66).
It took about 15 minutes to perform the procedure to implement the closed-loop insulin delivery system, Dr. Bally said. It featured a subcutaneous cannula inserted into the abdomen, a continuous glucose monitor (a device also used in the control group), and a trial insulin pump.
This was not a hybrid system, and it did not include prandial insulin boluses or input of the timing and carbohydrate content of meals. One reason behind the choice to adopt a fully automated system was to relieve the burden on both health care professionals and patients, coauthor Hood Thabit, PhD, of Wellcome Trust–MRC Institute of Metabolic Science, the Manchester Academic Health Science Center, and University of Manchester, said in an interview.
For up to 15 days or until discharge, researchers tracked how much of the time sensor glucose measurements were in a target range of 100 mg/dL to 180 mg/dL.
In the closed-loop group, glucose measurements were in the target range 66 mg/dL ± 17% of the time compared to 42 mg/dL ± 17% in the control group, a difference of 24 mg/dL ± 3% (95% confidence interval, 19-30; P less than .001).
For the closed-loop group, the average glucose level was 154 mg/dL, and it was 188 mg/dL in the control group (P less than .001).
The researchers didn’t find a statistically significant difference between the groups in duration of hypoglycemia or amount of insulin delivered.
None of the patients suffered from severe hypoglycemia or clinically significant hyperglycemia with ketonemia.
There were 18 incidents of clinically significant hyperglycemia events (capillary glucose levels of more than 360 mg/dL) in the closed-loop group, compared with 41 such events in the control group. (P = .03)
Three patients in each group had adverse trial-related device effects.
Of 62 patients in the closed-loop group who completed the trial, 87% reported being pleased by their glucose levels, and all but one reported being happy to have their levels monitored automatically. All 62 patients said they’d recommend the system to others.
Going forward, the researchers hope to launch a multicenter trial that will examine clinical outcomes such as postoperative complications, infections, mortality, and glucose control after hospital discharge, according to Dr. Bally.
The study was supported by Diabetes UK, the Swiss National Science Foundation, the European Foundation for the Study of Diabetes, the JDRF, the National Institute for Health Research Cambridge Biomedical Research Center, and a Wellcome Strategic Award. Abbott Diabetes Care supplied equipment and guidance regarding connectivity, and representatives reviewed the manuscript before submission.
Dr. Bally reported funding from the University Hospital Bern, University of Bern and the Swiss Diabetes Foundation. Dr. Thabit reported no disclosures. Other authors report no disclosures or various disclosures.
SOURCE: Bally L et al. ADA 2018 Abstract 350-OR. Published simultaneously in The New England Journal of Medicine. June 25, 2018
ORLANDO – (T2DM).
The findings, released at the annual scientific sessions of the American Diabetes Association and via simultaneous publication in The New England Journal of Medicine, don’t examine cost or clinical outcomes. However, “our results suggest this new technology might be another approach to manage in-patient hypoglycemia in a safe and effective way, lead author Lia Bally, MD, PhD, of the division of endocrinology, diabetes, and clinical nutrition, Bern (Switzerland ) University Hospital, said in an interview.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
For the open-label trial, the researchers recruited 136 adults with T2DM under noncritical care at two hospitals (one in the England and the other in Switzerland). Some patients had undergone surgery, Dr. Bally said, and some others were being treated for systemic infections. Comorbidities were significantly more severe in the closed-loop group, and 43% had sepsis.
All of the subjects required subcutaneous insulin therapy.
From 2016 to 2017, patients were randomly assigned to receive normal subcutaneous insulin therapy (n = 70) or closed-loop insulin delivery (n = 66).
It took about 15 minutes to perform the procedure to implement the closed-loop insulin delivery system, Dr. Bally said. It featured a subcutaneous cannula inserted into the abdomen, a continuous glucose monitor (a device also used in the control group), and a trial insulin pump.
This was not a hybrid system, and it did not include prandial insulin boluses or input of the timing and carbohydrate content of meals. One reason behind the choice to adopt a fully automated system was to relieve the burden on both health care professionals and patients, coauthor Hood Thabit, PhD, of Wellcome Trust–MRC Institute of Metabolic Science, the Manchester Academic Health Science Center, and University of Manchester, said in an interview.
For up to 15 days or until discharge, researchers tracked how much of the time sensor glucose measurements were in a target range of 100 mg/dL to 180 mg/dL.
In the closed-loop group, glucose measurements were in the target range 66 mg/dL ± 17% of the time compared to 42 mg/dL ± 17% in the control group, a difference of 24 mg/dL ± 3% (95% confidence interval, 19-30; P less than .001).
For the closed-loop group, the average glucose level was 154 mg/dL, and it was 188 mg/dL in the control group (P less than .001).
The researchers didn’t find a statistically significant difference between the groups in duration of hypoglycemia or amount of insulin delivered.
None of the patients suffered from severe hypoglycemia or clinically significant hyperglycemia with ketonemia.
There were 18 incidents of clinically significant hyperglycemia events (capillary glucose levels of more than 360 mg/dL) in the closed-loop group, compared with 41 such events in the control group. (P = .03)
Three patients in each group had adverse trial-related device effects.
Of 62 patients in the closed-loop group who completed the trial, 87% reported being pleased by their glucose levels, and all but one reported being happy to have their levels monitored automatically. All 62 patients said they’d recommend the system to others.
Going forward, the researchers hope to launch a multicenter trial that will examine clinical outcomes such as postoperative complications, infections, mortality, and glucose control after hospital discharge, according to Dr. Bally.
The study was supported by Diabetes UK, the Swiss National Science Foundation, the European Foundation for the Study of Diabetes, the JDRF, the National Institute for Health Research Cambridge Biomedical Research Center, and a Wellcome Strategic Award. Abbott Diabetes Care supplied equipment and guidance regarding connectivity, and representatives reviewed the manuscript before submission.
Dr. Bally reported funding from the University Hospital Bern, University of Bern and the Swiss Diabetes Foundation. Dr. Thabit reported no disclosures. Other authors report no disclosures or various disclosures.
SOURCE: Bally L et al. ADA 2018 Abstract 350-OR. Published simultaneously in The New England Journal of Medicine. June 25, 2018
REPORTING FROM ADA 2018
Key clinical point: Use of an automated closed-loop insulin delivery system may be feasible in the noncritical hospital setting.
Major finding: In the closed-loop group, glucose measurements were in the target range 66 mg/dL ± 17% of the time compared with 42 mg/dL ± 17% in the control group, a difference of 24 mg/dL ± 3% (95% CI, 19-30; P less than .001).
Study details: Randomized, open-label, two-center trial of 136 inpatients with type 2 diabetes mellitus assigned to either standard subcutaneous insulin therapy or closed-loop insulin delivery for 15 days or until discharge.
Disclosures: The study was supported by Diabetes UK, the Swiss National Science Foundation, the European Foundation for the Study of Diabetes, the JDRF, the National Institute for Health Research Cambridge Biomedical Research Center, and a Wellcome Strategic Award. Abbott Diabetes Care supplied equipment and guidance regarding connectivity, and representatives reviewed the manuscript before submission. The researchers reported no disclosures or various disclosures.
Source: Bally L et al. ADA 2018 Abstract 350-OR.
New hypertension guidelines would add 15.6 million new diagnoses
A new analysis estimates that adopting the 2017 ACC/AHA hypertension guidelines would add 15.6 million Americans to the ranks of the hypertensives, and half of those would be candidates for treatment.
Similar increases would occur in other countries, according to study authors, who analyzed two large datasets from the United States and China.
That happened by resetting the definition of adult hypertension from the long-standing threshold of 140/90 mm Hg to a blood pressure at or above 130/80 mm Hg, meaning more than half of people aged 45-75 years in both countries would be classified as having hypertension, according to the researchers, led by Harlan M. Krumholz, MD, of the Center for Outcomes Research and Evaluation at Yale–New Haven (Conn.) Hospital and the section of cardiovascular medicine at Yale
An additional 7.5 million Americans would be recommended for treatment under the new lower treatment thresholds, with a correspondingly large increase in the Chinese population, according to results published in the BMJ.
The guideline changes are “not firmly rooted in evidence” and could have health policy implications that include strain on public health programs, Dr. Krumholz and his colleagues said in their report on the study.
“The change occurs at a time when both countries have substantial numbers of people who are not aware of having hypertension, and who have hypertension that is not controlled, even according to the previous standards,” they wrote.
The analysis by Dr. Krumholz and his colleagues was based on the two most recent cycles of the U.S. National Health and Nutrition Examination Survey (NHANES), representing 2013-2014 and 2015-2016 periods, as well as the China Health and Retirement Longitudinal Study (CHARLS) in 2011-2012.
Under the new ACC/AHA guidelines, they found, 70.1 million Americans aged 45-65 years would be classified as hypertensive, representing 63% of that age group. That’s a 27% relative increase over the 55.3 million individuals, or 49.7%, with hypertension as defined in the JNC-8 guidelines.
In addition, 15.6 million persons would be classified as eligible for treatment but not receiving it, up from 8.1 million under the JNC-8 guidance.
Previous estimates projected a far greater jump in new hypertension classifications, including one that used data from the National Health and Nutrition Examination Survey, antihypertensive clinical trials, and population-based cohort studies. That study estimated that 31 million people would newly carry the label (JAMA Cardiol. 2018 May 23; doi: 10.1001/jamacardio.2018.1240.)
In the current analysis, in China, 267 million aged 45-65 years (55% of that age group) would be classified with hypertension under the ACC/AHA guidelines, a relative increase of 45% over the JNC-8 guidelines, while the number of candidates for treatment would be 129 million, up from 74.5 million under the earlier guidelines.
Dr. Krumholz noted that the ACC/AHA guideline changes were prompted by results from the SPRINT trial. However, the improvements in outcomes seen in SPRINT, which included patients at high risk for cardiovascular events but without diabetes, have not been observed in individuals at low or intermediate risk, or in those with diabetes, they said.
“Expanding the pool of patients who merit treatment to include those at low risk could potentially render public health programs less efficient and viable,” they wrote in a discussion of health policy implications.
The new guidelines also put millions at risk of the “psychological morbidity” that comes with the label of a chronic disease, and at risk for more adverse events caused by inappropriate use of drug therapy, they added.
Dr. Krumholz reported research agreements from Medtronic and from Johnson and Johnson (Janssen) through Yale University, and a grant from the Food and Drug Administration and Medtronic. He reported other disclosures related to UnitedHealth, the IBM Watson Health Life Sciences Board, Element Science, Aetna, and Hugo, a personal health information platform he founded. First author Rohan Khera, MD, reported support from the National Institutes of Health.
SOURCE: Khera R et al. BMJ. 2018 Jul 11;362:k2357
This article was updated 7/19/18.
This study addressing hypertension guideline changes is unique because it was initially published on a public preprint server.
Preprints are common in some scientific areas, but uncommon in major medical journals. They allow investigators to share research, quickly and openly, for critique and feedback before standard peer review and publication.
In the case of this study, researchers analyzed the public health implications of the anticipated changes to the 2017 ACC/AHA hypertension guidelines in two nationally representative data sets from the United States and China.
The authors quickly finalized their manuscript right after the revised hypertension guidelines were released. They chose the preprint approach because they realized their research would be immediately relevant to the discussion that followed, first author Rohan Khera, MD, recounted on BMJ Blogs.
“The traditional approach of submitting to a medical journal would mean being out of the public eye for several months,” Dr. Khera said in his post. “The preprint platform offered us an excellent opportunity of ensuring early dissemination of our research study in its entirety, while we sought its evaluation by peer reviewers and the refinement by a medical journal.”
The manuscript was submitted via a Web-based system and was publicly available 2 hours later on the same day the guidelines were published. The researchers received comments and suggestions on the preprint, some of which were incorporated into the final manuscript they submitted for peer review.
Then the manuscript went through the usual iterative peer review process; however, the preprint was still available online to guide other investigators and limit duplication of effort, Dr. Khera said in his blog post.
That contrasts with another recent experience in which Dr. Khera and his colleagues performed work that “failed to inform” ongoing policy discussions, and other research efforts, while they waited for eventual publication.
“We hope that more journals will accept the benefits of science that is publicly available while journal editors and peer reviewers carry out their critical role of improving both the quality and the impact of these scientific contributions,” Dr. Khera wrote.
Rohan Khera, MD, a cardiology fellow at the University of Texas (Dallas) Southwestern Medical Center in, wrote about his experience with preprints for BMJ Blogs . Dr. Khera had no conflicts of interest to disclose.
This study addressing hypertension guideline changes is unique because it was initially published on a public preprint server.
Preprints are common in some scientific areas, but uncommon in major medical journals. They allow investigators to share research, quickly and openly, for critique and feedback before standard peer review and publication.
In the case of this study, researchers analyzed the public health implications of the anticipated changes to the 2017 ACC/AHA hypertension guidelines in two nationally representative data sets from the United States and China.
The authors quickly finalized their manuscript right after the revised hypertension guidelines were released. They chose the preprint approach because they realized their research would be immediately relevant to the discussion that followed, first author Rohan Khera, MD, recounted on BMJ Blogs.
“The traditional approach of submitting to a medical journal would mean being out of the public eye for several months,” Dr. Khera said in his post. “The preprint platform offered us an excellent opportunity of ensuring early dissemination of our research study in its entirety, while we sought its evaluation by peer reviewers and the refinement by a medical journal.”
The manuscript was submitted via a Web-based system and was publicly available 2 hours later on the same day the guidelines were published. The researchers received comments and suggestions on the preprint, some of which were incorporated into the final manuscript they submitted for peer review.
Then the manuscript went through the usual iterative peer review process; however, the preprint was still available online to guide other investigators and limit duplication of effort, Dr. Khera said in his blog post.
That contrasts with another recent experience in which Dr. Khera and his colleagues performed work that “failed to inform” ongoing policy discussions, and other research efforts, while they waited for eventual publication.
“We hope that more journals will accept the benefits of science that is publicly available while journal editors and peer reviewers carry out their critical role of improving both the quality and the impact of these scientific contributions,” Dr. Khera wrote.
Rohan Khera, MD, a cardiology fellow at the University of Texas (Dallas) Southwestern Medical Center in, wrote about his experience with preprints for BMJ Blogs . Dr. Khera had no conflicts of interest to disclose.
This study addressing hypertension guideline changes is unique because it was initially published on a public preprint server.
Preprints are common in some scientific areas, but uncommon in major medical journals. They allow investigators to share research, quickly and openly, for critique and feedback before standard peer review and publication.
In the case of this study, researchers analyzed the public health implications of the anticipated changes to the 2017 ACC/AHA hypertension guidelines in two nationally representative data sets from the United States and China.
The authors quickly finalized their manuscript right after the revised hypertension guidelines were released. They chose the preprint approach because they realized their research would be immediately relevant to the discussion that followed, first author Rohan Khera, MD, recounted on BMJ Blogs.
“The traditional approach of submitting to a medical journal would mean being out of the public eye for several months,” Dr. Khera said in his post. “The preprint platform offered us an excellent opportunity of ensuring early dissemination of our research study in its entirety, while we sought its evaluation by peer reviewers and the refinement by a medical journal.”
The manuscript was submitted via a Web-based system and was publicly available 2 hours later on the same day the guidelines were published. The researchers received comments and suggestions on the preprint, some of which were incorporated into the final manuscript they submitted for peer review.
Then the manuscript went through the usual iterative peer review process; however, the preprint was still available online to guide other investigators and limit duplication of effort, Dr. Khera said in his blog post.
That contrasts with another recent experience in which Dr. Khera and his colleagues performed work that “failed to inform” ongoing policy discussions, and other research efforts, while they waited for eventual publication.
“We hope that more journals will accept the benefits of science that is publicly available while journal editors and peer reviewers carry out their critical role of improving both the quality and the impact of these scientific contributions,” Dr. Khera wrote.
Rohan Khera, MD, a cardiology fellow at the University of Texas (Dallas) Southwestern Medical Center in, wrote about his experience with preprints for BMJ Blogs . Dr. Khera had no conflicts of interest to disclose.
A new analysis estimates that adopting the 2017 ACC/AHA hypertension guidelines would add 15.6 million Americans to the ranks of the hypertensives, and half of those would be candidates for treatment.
Similar increases would occur in other countries, according to study authors, who analyzed two large datasets from the United States and China.
That happened by resetting the definition of adult hypertension from the long-standing threshold of 140/90 mm Hg to a blood pressure at or above 130/80 mm Hg, meaning more than half of people aged 45-75 years in both countries would be classified as having hypertension, according to the researchers, led by Harlan M. Krumholz, MD, of the Center for Outcomes Research and Evaluation at Yale–New Haven (Conn.) Hospital and the section of cardiovascular medicine at Yale
An additional 7.5 million Americans would be recommended for treatment under the new lower treatment thresholds, with a correspondingly large increase in the Chinese population, according to results published in the BMJ.
The guideline changes are “not firmly rooted in evidence” and could have health policy implications that include strain on public health programs, Dr. Krumholz and his colleagues said in their report on the study.
“The change occurs at a time when both countries have substantial numbers of people who are not aware of having hypertension, and who have hypertension that is not controlled, even according to the previous standards,” they wrote.
The analysis by Dr. Krumholz and his colleagues was based on the two most recent cycles of the U.S. National Health and Nutrition Examination Survey (NHANES), representing 2013-2014 and 2015-2016 periods, as well as the China Health and Retirement Longitudinal Study (CHARLS) in 2011-2012.
Under the new ACC/AHA guidelines, they found, 70.1 million Americans aged 45-65 years would be classified as hypertensive, representing 63% of that age group. That’s a 27% relative increase over the 55.3 million individuals, or 49.7%, with hypertension as defined in the JNC-8 guidelines.
In addition, 15.6 million persons would be classified as eligible for treatment but not receiving it, up from 8.1 million under the JNC-8 guidance.
Previous estimates projected a far greater jump in new hypertension classifications, including one that used data from the National Health and Nutrition Examination Survey, antihypertensive clinical trials, and population-based cohort studies. That study estimated that 31 million people would newly carry the label (JAMA Cardiol. 2018 May 23; doi: 10.1001/jamacardio.2018.1240.)
In the current analysis, in China, 267 million aged 45-65 years (55% of that age group) would be classified with hypertension under the ACC/AHA guidelines, a relative increase of 45% over the JNC-8 guidelines, while the number of candidates for treatment would be 129 million, up from 74.5 million under the earlier guidelines.
Dr. Krumholz noted that the ACC/AHA guideline changes were prompted by results from the SPRINT trial. However, the improvements in outcomes seen in SPRINT, which included patients at high risk for cardiovascular events but without diabetes, have not been observed in individuals at low or intermediate risk, or in those with diabetes, they said.
“Expanding the pool of patients who merit treatment to include those at low risk could potentially render public health programs less efficient and viable,” they wrote in a discussion of health policy implications.
The new guidelines also put millions at risk of the “psychological morbidity” that comes with the label of a chronic disease, and at risk for more adverse events caused by inappropriate use of drug therapy, they added.
Dr. Krumholz reported research agreements from Medtronic and from Johnson and Johnson (Janssen) through Yale University, and a grant from the Food and Drug Administration and Medtronic. He reported other disclosures related to UnitedHealth, the IBM Watson Health Life Sciences Board, Element Science, Aetna, and Hugo, a personal health information platform he founded. First author Rohan Khera, MD, reported support from the National Institutes of Health.
SOURCE: Khera R et al. BMJ. 2018 Jul 11;362:k2357
This article was updated 7/19/18.
A new analysis estimates that adopting the 2017 ACC/AHA hypertension guidelines would add 15.6 million Americans to the ranks of the hypertensives, and half of those would be candidates for treatment.
Similar increases would occur in other countries, according to study authors, who analyzed two large datasets from the United States and China.
That happened by resetting the definition of adult hypertension from the long-standing threshold of 140/90 mm Hg to a blood pressure at or above 130/80 mm Hg, meaning more than half of people aged 45-75 years in both countries would be classified as having hypertension, according to the researchers, led by Harlan M. Krumholz, MD, of the Center for Outcomes Research and Evaluation at Yale–New Haven (Conn.) Hospital and the section of cardiovascular medicine at Yale
An additional 7.5 million Americans would be recommended for treatment under the new lower treatment thresholds, with a correspondingly large increase in the Chinese population, according to results published in the BMJ.
The guideline changes are “not firmly rooted in evidence” and could have health policy implications that include strain on public health programs, Dr. Krumholz and his colleagues said in their report on the study.
“The change occurs at a time when both countries have substantial numbers of people who are not aware of having hypertension, and who have hypertension that is not controlled, even according to the previous standards,” they wrote.
The analysis by Dr. Krumholz and his colleagues was based on the two most recent cycles of the U.S. National Health and Nutrition Examination Survey (NHANES), representing 2013-2014 and 2015-2016 periods, as well as the China Health and Retirement Longitudinal Study (CHARLS) in 2011-2012.
Under the new ACC/AHA guidelines, they found, 70.1 million Americans aged 45-65 years would be classified as hypertensive, representing 63% of that age group. That’s a 27% relative increase over the 55.3 million individuals, or 49.7%, with hypertension as defined in the JNC-8 guidelines.
In addition, 15.6 million persons would be classified as eligible for treatment but not receiving it, up from 8.1 million under the JNC-8 guidance.
Previous estimates projected a far greater jump in new hypertension classifications, including one that used data from the National Health and Nutrition Examination Survey, antihypertensive clinical trials, and population-based cohort studies. That study estimated that 31 million people would newly carry the label (JAMA Cardiol. 2018 May 23; doi: 10.1001/jamacardio.2018.1240.)
In the current analysis, in China, 267 million aged 45-65 years (55% of that age group) would be classified with hypertension under the ACC/AHA guidelines, a relative increase of 45% over the JNC-8 guidelines, while the number of candidates for treatment would be 129 million, up from 74.5 million under the earlier guidelines.
Dr. Krumholz noted that the ACC/AHA guideline changes were prompted by results from the SPRINT trial. However, the improvements in outcomes seen in SPRINT, which included patients at high risk for cardiovascular events but without diabetes, have not been observed in individuals at low or intermediate risk, or in those with diabetes, they said.
“Expanding the pool of patients who merit treatment to include those at low risk could potentially render public health programs less efficient and viable,” they wrote in a discussion of health policy implications.
The new guidelines also put millions at risk of the “psychological morbidity” that comes with the label of a chronic disease, and at risk for more adverse events caused by inappropriate use of drug therapy, they added.
Dr. Krumholz reported research agreements from Medtronic and from Johnson and Johnson (Janssen) through Yale University, and a grant from the Food and Drug Administration and Medtronic. He reported other disclosures related to UnitedHealth, the IBM Watson Health Life Sciences Board, Element Science, Aetna, and Hugo, a personal health information platform he founded. First author Rohan Khera, MD, reported support from the National Institutes of Health.
SOURCE: Khera R et al. BMJ. 2018 Jul 11;362:k2357
This article was updated 7/19/18.
FROM THE BMJ
Key clinical point: The 2017 ACC/AHA hypertension guidelines could dramatically increase the number of individuals with hypertension and candidates for treatment.
Major finding: The number of individuals with untreated hypertension increased from 8.1 million to 15.6 million.
Study details: A cross-sectional study of adults in nationally representative databases in the United States (NHANES) and China (CHARLS).
Disclosures: Authors reported disclosures related to Medtronic, Johnson and Johnson (Janssen), the Food and Drug Administration, UnitedHealth, the IBM Watson Health Life Sciences Board, Element Science, Aetna, and Hugo.
Source: Khera R et al. BMJ 2018;362:k2357.
Fluoroquinolones can cause fatal hypoglycemia, FDA warns
Fluoroquinolones have caused at least 67 cases of life-threatening hypoglycemic coma, including 13 deaths and 9 permanent and disabling injuries, according to an internal safety review by the Food and Drug Administration. Most cases (44) were associated with levofloxacin.
The review also found new neuropsychiatric side effects associated with fluoroquinolones, including disturbances in attention, memory impairment, and delirium.
Considering these findings, the agency will strengthen warning labels on all fluoroquinolones, which already warn that the antibiotics may cause hypoglycemia and mental health issues, especially in older people, the FDA said in a press statement.
“Health care professionals should be aware of the potential risk of hypoglycemia, sometimes resulting in coma, occurring more frequently in the elderly and those with diabetes taking an oral hypoglycemic medicine or insulin,” the statement said. “Alert patients of the symptoms of hypoglycemia and carefully monitor blood glucose levels in these patients and discuss with them how to treat themselves if they have symptoms of hypoglycemia. Inform patients about the risk of psychiatric adverse reactions that can occur after just one dose. Stop fluoroquinolone treatment immediately if a patient reports any central nervous system side effects, including psychiatric adverse reactions, or blood glucose disturbances and switch to a non–fluoroquinolone antibiotic if possible. Stop fluoroquinolone treatment immediately if a patient reports serious side effects involving the tendons, muscles, joints, or nerves, and switch to a non–fluoroquinolone antibiotic to complete the patient’s treatment course.”
The statement also warned not to prescribe fluoroquinolones to patients who have other treatment options for acute bacterial sinusitis, acute bacterial exacerbation of chronic bronchitis, and uncomplicated urinary tract infections because the risks outweigh the benefits in these patients.
The FDA conducted the postmarketing review on all five of the fluoroquinolones (ciprofloxacin, gemifloxacin, levofloxacin, moxifloxacin, and ofloxacin). The newest fluoroquinolone, delafloxacin, approved a year ago, was not included in the class review. However, the agency expects that similar adverse events will be associated with delafloxacin and labeling on that drug will include the new warnings.
The agency reviewed cases in the FDA Adverse Event Reporting System, and in published medical literature, during 1987-2017. Most of the incidents (56) were in the system; 11 additional cases were published. Levofloxacin caused most of the incidents (44), followed by ciprofloxacin (12), moxifloxacin (9), and ofloxacin (2). Four of the fluoroquinolones have a labeled drug interaction with sulfonylurea agents, which can cause hypoglycemia.
Some of those who died were getting the antibiotics for complicated infections, including urinary tract and upper respiratory tract infections, and postoperative antibiotic prophylaxis. Others had renal insufficiency – a risk factor for hypoglycemia.
Of the 54 patients who survived, 9 never fully recovered and had permanent disabilities. Four patients remained in a coma for at least 1 month, despite blood sugar normalization. Five experienced some type of neurologic injury.
The new label changes will also fortify the existing warning about mental health side effects, after the review found new reactions that are not listed in the current warning, including the new reports of disturbance in attention, memory impairment, and delirium.
The FDA statement did not include the number of cases found or the associated drugs. Again, the safety review was based on reports in the FAERS database and published medical literature.
“We found that psychiatric adverse reactions were not consistent in the drug labels. The labels of fluoroquinolones currently include many psychiatric adverse reactions in the Warnings and Precautions section, for example, hallucination, psychoses, confusion, depression, anxiety, and paranoia. In an effort to harmonize the psychiatric adverse reactions described in the drug labels across the class of fluoroquinolones, we are requiring that all fluoroquinolones include six psychiatric adverse reactions (disturbance in attention, memory impairment, delirium, nervousness, agitation, and disorientation) in the Central Nervous System Effects of the Warnings and Precautions section of the labels. Disturbance in attention, memory impairment, and delirium are new adverse reactions to be added to the labels of the entire class of fluoroquinolones. Nervousness, agitation, and disorientation had been previously listed in the fluoroquinolone drug labels and will now be added to the Warnings and Precautions section of each drug label to harmonize labels across the fluoroquinolone drug class. The new label changes will make the psychiatric adverse reactions more prominent and more consistent.”
The FDA has previously warned about other adverse events associated with fluoroquinolones in May 2016, restricting use for certain uncomplicated infections; July 2016, for disabling side effects; August 2013, for peripheral neuropathy, and July 2008, for tendinitis and tendon rupture.
Fluoroquinolones have caused at least 67 cases of life-threatening hypoglycemic coma, including 13 deaths and 9 permanent and disabling injuries, according to an internal safety review by the Food and Drug Administration. Most cases (44) were associated with levofloxacin.
The review also found new neuropsychiatric side effects associated with fluoroquinolones, including disturbances in attention, memory impairment, and delirium.
Considering these findings, the agency will strengthen warning labels on all fluoroquinolones, which already warn that the antibiotics may cause hypoglycemia and mental health issues, especially in older people, the FDA said in a press statement.
“Health care professionals should be aware of the potential risk of hypoglycemia, sometimes resulting in coma, occurring more frequently in the elderly and those with diabetes taking an oral hypoglycemic medicine or insulin,” the statement said. “Alert patients of the symptoms of hypoglycemia and carefully monitor blood glucose levels in these patients and discuss with them how to treat themselves if they have symptoms of hypoglycemia. Inform patients about the risk of psychiatric adverse reactions that can occur after just one dose. Stop fluoroquinolone treatment immediately if a patient reports any central nervous system side effects, including psychiatric adverse reactions, or blood glucose disturbances and switch to a non–fluoroquinolone antibiotic if possible. Stop fluoroquinolone treatment immediately if a patient reports serious side effects involving the tendons, muscles, joints, or nerves, and switch to a non–fluoroquinolone antibiotic to complete the patient’s treatment course.”
The statement also warned not to prescribe fluoroquinolones to patients who have other treatment options for acute bacterial sinusitis, acute bacterial exacerbation of chronic bronchitis, and uncomplicated urinary tract infections because the risks outweigh the benefits in these patients.
The FDA conducted the postmarketing review on all five of the fluoroquinolones (ciprofloxacin, gemifloxacin, levofloxacin, moxifloxacin, and ofloxacin). The newest fluoroquinolone, delafloxacin, approved a year ago, was not included in the class review. However, the agency expects that similar adverse events will be associated with delafloxacin and labeling on that drug will include the new warnings.
The agency reviewed cases in the FDA Adverse Event Reporting System, and in published medical literature, during 1987-2017. Most of the incidents (56) were in the system; 11 additional cases were published. Levofloxacin caused most of the incidents (44), followed by ciprofloxacin (12), moxifloxacin (9), and ofloxacin (2). Four of the fluoroquinolones have a labeled drug interaction with sulfonylurea agents, which can cause hypoglycemia.
Some of those who died were getting the antibiotics for complicated infections, including urinary tract and upper respiratory tract infections, and postoperative antibiotic prophylaxis. Others had renal insufficiency – a risk factor for hypoglycemia.
Of the 54 patients who survived, 9 never fully recovered and had permanent disabilities. Four patients remained in a coma for at least 1 month, despite blood sugar normalization. Five experienced some type of neurologic injury.
The new label changes will also fortify the existing warning about mental health side effects, after the review found new reactions that are not listed in the current warning, including the new reports of disturbance in attention, memory impairment, and delirium.
The FDA statement did not include the number of cases found or the associated drugs. Again, the safety review was based on reports in the FAERS database and published medical literature.
“We found that psychiatric adverse reactions were not consistent in the drug labels. The labels of fluoroquinolones currently include many psychiatric adverse reactions in the Warnings and Precautions section, for example, hallucination, psychoses, confusion, depression, anxiety, and paranoia. In an effort to harmonize the psychiatric adverse reactions described in the drug labels across the class of fluoroquinolones, we are requiring that all fluoroquinolones include six psychiatric adverse reactions (disturbance in attention, memory impairment, delirium, nervousness, agitation, and disorientation) in the Central Nervous System Effects of the Warnings and Precautions section of the labels. Disturbance in attention, memory impairment, and delirium are new adverse reactions to be added to the labels of the entire class of fluoroquinolones. Nervousness, agitation, and disorientation had been previously listed in the fluoroquinolone drug labels and will now be added to the Warnings and Precautions section of each drug label to harmonize labels across the fluoroquinolone drug class. The new label changes will make the psychiatric adverse reactions more prominent and more consistent.”
The FDA has previously warned about other adverse events associated with fluoroquinolones in May 2016, restricting use for certain uncomplicated infections; July 2016, for disabling side effects; August 2013, for peripheral neuropathy, and July 2008, for tendinitis and tendon rupture.
Fluoroquinolones have caused at least 67 cases of life-threatening hypoglycemic coma, including 13 deaths and 9 permanent and disabling injuries, according to an internal safety review by the Food and Drug Administration. Most cases (44) were associated with levofloxacin.
The review also found new neuropsychiatric side effects associated with fluoroquinolones, including disturbances in attention, memory impairment, and delirium.
Considering these findings, the agency will strengthen warning labels on all fluoroquinolones, which already warn that the antibiotics may cause hypoglycemia and mental health issues, especially in older people, the FDA said in a press statement.
“Health care professionals should be aware of the potential risk of hypoglycemia, sometimes resulting in coma, occurring more frequently in the elderly and those with diabetes taking an oral hypoglycemic medicine or insulin,” the statement said. “Alert patients of the symptoms of hypoglycemia and carefully monitor blood glucose levels in these patients and discuss with them how to treat themselves if they have symptoms of hypoglycemia. Inform patients about the risk of psychiatric adverse reactions that can occur after just one dose. Stop fluoroquinolone treatment immediately if a patient reports any central nervous system side effects, including psychiatric adverse reactions, or blood glucose disturbances and switch to a non–fluoroquinolone antibiotic if possible. Stop fluoroquinolone treatment immediately if a patient reports serious side effects involving the tendons, muscles, joints, or nerves, and switch to a non–fluoroquinolone antibiotic to complete the patient’s treatment course.”
The statement also warned not to prescribe fluoroquinolones to patients who have other treatment options for acute bacterial sinusitis, acute bacterial exacerbation of chronic bronchitis, and uncomplicated urinary tract infections because the risks outweigh the benefits in these patients.
The FDA conducted the postmarketing review on all five of the fluoroquinolones (ciprofloxacin, gemifloxacin, levofloxacin, moxifloxacin, and ofloxacin). The newest fluoroquinolone, delafloxacin, approved a year ago, was not included in the class review. However, the agency expects that similar adverse events will be associated with delafloxacin and labeling on that drug will include the new warnings.
The agency reviewed cases in the FDA Adverse Event Reporting System, and in published medical literature, during 1987-2017. Most of the incidents (56) were in the system; 11 additional cases were published. Levofloxacin caused most of the incidents (44), followed by ciprofloxacin (12), moxifloxacin (9), and ofloxacin (2). Four of the fluoroquinolones have a labeled drug interaction with sulfonylurea agents, which can cause hypoglycemia.
Some of those who died were getting the antibiotics for complicated infections, including urinary tract and upper respiratory tract infections, and postoperative antibiotic prophylaxis. Others had renal insufficiency – a risk factor for hypoglycemia.
Of the 54 patients who survived, 9 never fully recovered and had permanent disabilities. Four patients remained in a coma for at least 1 month, despite blood sugar normalization. Five experienced some type of neurologic injury.
The new label changes will also fortify the existing warning about mental health side effects, after the review found new reactions that are not listed in the current warning, including the new reports of disturbance in attention, memory impairment, and delirium.
The FDA statement did not include the number of cases found or the associated drugs. Again, the safety review was based on reports in the FAERS database and published medical literature.
“We found that psychiatric adverse reactions were not consistent in the drug labels. The labels of fluoroquinolones currently include many psychiatric adverse reactions in the Warnings and Precautions section, for example, hallucination, psychoses, confusion, depression, anxiety, and paranoia. In an effort to harmonize the psychiatric adverse reactions described in the drug labels across the class of fluoroquinolones, we are requiring that all fluoroquinolones include six psychiatric adverse reactions (disturbance in attention, memory impairment, delirium, nervousness, agitation, and disorientation) in the Central Nervous System Effects of the Warnings and Precautions section of the labels. Disturbance in attention, memory impairment, and delirium are new adverse reactions to be added to the labels of the entire class of fluoroquinolones. Nervousness, agitation, and disorientation had been previously listed in the fluoroquinolone drug labels and will now be added to the Warnings and Precautions section of each drug label to harmonize labels across the fluoroquinolone drug class. The new label changes will make the psychiatric adverse reactions more prominent and more consistent.”
The FDA has previously warned about other adverse events associated with fluoroquinolones in May 2016, restricting use for certain uncomplicated infections; July 2016, for disabling side effects; August 2013, for peripheral neuropathy, and July 2008, for tendinitis and tendon rupture.
Rebleeding and mortality after lower-GI bleeding in patients taking antiplatelets or anticoagulants
Clinical question: Is there a difference in lower GI rebleeding risk in patients on antiplatelet medications versus those on anticoagulation medications?
Background: It is estimated that 29%-37% of patient with GI bleeds are also on antiplatelet or anticoagulation medications. Minimal research has looked at outcomes for these populations and the comparative risk of rebleeding.
Study design: A retrospective study.
Setting: Multicenter study in the United Kingdom.
Synopsis: The study followed 2,528 patients with lower GI bleeds, 917 of whom were on antiplatelet or anticoagulation medications. Of these, 504 were on single-antiplatelet therapy, 79 on dual-antiplatelet therapy, 232 on warfarin, and 102 on direct-acting oral anticoagulants (DOACs). Patients on single-antiplatelet agents had a threefold increased risk of rebleeding (hazard ratio, 3.57), those on dual-antiplatelet agents had a fivefold increased risk of rebleeding (HR, 5.38), and patients taking warfarin or DOACs had no increased risk of rebleeding.
In addition, the authors concluded that there was no significant difference in rebleeding risk if antiplatelet medications were held for less than 5 days during hospitalization versus if they were continued. The risk of rebleeding with antiplatelet agents is likely caused by the relatively long half-lives of these therapies. In contrast, warfarin and DOACs have available reversal agents, and DOACs have comparatively shorter half-lives.
Bottom line: The risk of rebleeding from a lower-GI bleed is higher in patients on antiplatelet medications than it is in patients on warfarin or DOACs.
Citation: Oakland K et al. Rebleeding and mortality after lower gastrointestinal bleeding in patients taking antiplatelets or anticoagulants. Clin Gastroent Hepatol. 2017 Dec 23. doi: 10.1016/j.cgh.2017.12.032.
Dr. Thota is a hospitalist at UC San Diego Health and an assistant clinical professor at the University of California, San Diego.
Clinical question: Is there a difference in lower GI rebleeding risk in patients on antiplatelet medications versus those on anticoagulation medications?
Background: It is estimated that 29%-37% of patient with GI bleeds are also on antiplatelet or anticoagulation medications. Minimal research has looked at outcomes for these populations and the comparative risk of rebleeding.
Study design: A retrospective study.
Setting: Multicenter study in the United Kingdom.
Synopsis: The study followed 2,528 patients with lower GI bleeds, 917 of whom were on antiplatelet or anticoagulation medications. Of these, 504 were on single-antiplatelet therapy, 79 on dual-antiplatelet therapy, 232 on warfarin, and 102 on direct-acting oral anticoagulants (DOACs). Patients on single-antiplatelet agents had a threefold increased risk of rebleeding (hazard ratio, 3.57), those on dual-antiplatelet agents had a fivefold increased risk of rebleeding (HR, 5.38), and patients taking warfarin or DOACs had no increased risk of rebleeding.
In addition, the authors concluded that there was no significant difference in rebleeding risk if antiplatelet medications were held for less than 5 days during hospitalization versus if they were continued. The risk of rebleeding with antiplatelet agents is likely caused by the relatively long half-lives of these therapies. In contrast, warfarin and DOACs have available reversal agents, and DOACs have comparatively shorter half-lives.
Bottom line: The risk of rebleeding from a lower-GI bleed is higher in patients on antiplatelet medications than it is in patients on warfarin or DOACs.
Citation: Oakland K et al. Rebleeding and mortality after lower gastrointestinal bleeding in patients taking antiplatelets or anticoagulants. Clin Gastroent Hepatol. 2017 Dec 23. doi: 10.1016/j.cgh.2017.12.032.
Dr. Thota is a hospitalist at UC San Diego Health and an assistant clinical professor at the University of California, San Diego.
Clinical question: Is there a difference in lower GI rebleeding risk in patients on antiplatelet medications versus those on anticoagulation medications?
Background: It is estimated that 29%-37% of patient with GI bleeds are also on antiplatelet or anticoagulation medications. Minimal research has looked at outcomes for these populations and the comparative risk of rebleeding.
Study design: A retrospective study.
Setting: Multicenter study in the United Kingdom.
Synopsis: The study followed 2,528 patients with lower GI bleeds, 917 of whom were on antiplatelet or anticoagulation medications. Of these, 504 were on single-antiplatelet therapy, 79 on dual-antiplatelet therapy, 232 on warfarin, and 102 on direct-acting oral anticoagulants (DOACs). Patients on single-antiplatelet agents had a threefold increased risk of rebleeding (hazard ratio, 3.57), those on dual-antiplatelet agents had a fivefold increased risk of rebleeding (HR, 5.38), and patients taking warfarin or DOACs had no increased risk of rebleeding.
In addition, the authors concluded that there was no significant difference in rebleeding risk if antiplatelet medications were held for less than 5 days during hospitalization versus if they were continued. The risk of rebleeding with antiplatelet agents is likely caused by the relatively long half-lives of these therapies. In contrast, warfarin and DOACs have available reversal agents, and DOACs have comparatively shorter half-lives.
Bottom line: The risk of rebleeding from a lower-GI bleed is higher in patients on antiplatelet medications than it is in patients on warfarin or DOACs.
Citation: Oakland K et al. Rebleeding and mortality after lower gastrointestinal bleeding in patients taking antiplatelets or anticoagulants. Clin Gastroent Hepatol. 2017 Dec 23. doi: 10.1016/j.cgh.2017.12.032.
Dr. Thota is a hospitalist at UC San Diego Health and an assistant clinical professor at the University of California, San Diego.
Alteplase, aspirin provide similar functional outcomes after nondisabling stroke
Treatment with alteplase vs. aspirin did not improve functional outcomes at 90 days in patients with minor nondisabling acute ischemic stroke in the randomized phase 3b PRISMS trial.
At 90 days following a minor stroke judged to be nondisabling, a favorable functional outcome – defined as modified Rankin Scale (mRS) score of 0 or 1 – occurred in 122 (78.2%) of 156 patients who received alteplase and in 128 (81.5%) of 157 who received aspirin (adjusted risk difference, –1.1%), wrote Pooja Khatri, MD, of the University of Cincinnati, and her colleagues. The report was published in the July 10 issue of JAMA.
The PRISMS (Potential of rtPA for Ischemic Strokes with Mild Symptoms) trial was intended as a 948-patient, double-blind, placebo-controlled U.S. trial comparing alteplase and aspirin for emergent stroke in patients with National Institutes of Health Stroke Scale (NIHSS) scores of 0-5 at presentation whose stroke-related neurologic deficits were not clearly disabling and in whom the study treatment could be initiated within 3 hours. However, the trial was terminated early by the sponsor – prior to unblinding or interim analyses – because of below-target enrollment. An original plan to measure the difference in favorable functional outcome in the treatment and placebo groups by Cochran-Mantel-Haenszel hypothesis test with stratification by pretreatment NIHSS score, age, and time from onset to treatment was therefore revised to examine the risk difference of the primary outcome by a linear model adjusted for those factors, the authors explained.
Patients in the study had a mean age of 62 years and mean NIHSS score of 2, and were enrolled between May 30, 2014, and Dec. 20, 2016. They received either intravenous alteplase at a standard dose of 0.9 mg/kg with oral placebo, or oral aspirin at a dose of 325 mg with intravenous placebo, and were followed until March 22, 2017.
The primary safety endpoint of the analysis was symptomatic intracranial hemorrhage (sICH) within 36 hours of intravenous alteplase; this occurred in 5 (3.2%) of the alteplase-treated patients and in none of aspirin-treated patients (risk difference, 3.3%).
“Secondary outcomes, including the ordinal analysis of mRS scores (odds ratio, 0.81) and global favorable recovery (OR, 0.86), did not significantly favor either group,” the investigators added.
The findings are noteworthy because more than half of all patients with acute ischemic stroke have minor neurologic deficits, the investigators said, and while prior studies of alteplase included patients with low NIHSS score, few included patients who had no clearly disabling deficits. They added that while “alteplase is the standard of care for patients with ischemic stroke and disabling deficits regardless of severity judged by NIHSS scores, the optimal management of patients with not clearly disabling deficits is unclear.
“The study results raise the hypothesis that even a 6% treatment effect might be unlikely. However, the very early study termination precludes any definitive conclusions,” they wrote, noting that the study has several other limitations, including possible selection bias, relatively high loss to follow-up, and the subjective nature of the definition of “not clearly disabling.”
Additional research may be warranted, they said.
In an editorial, William J. Powers, MD, of the University of North Carolina at Chapel Hill, wrote that despite the limitations of the PRISMS trial, the findings help define the role for intravenous alteplase in the management of acute ischemic stroke.
“Even with early study termination and resultant wide 95% confidence intervals, the excellent outcome in the aspirin group and the numerically similar outcomes between the two groups render it unlikely that intravenous alteplase treatment meaningfully improves functional outcome in patients with initial NIHSS scores of 5 or lower with nondisabling deficits,” he wrote.
He noted, however – as did the study authors – that these conclusions do not apply to all patients with mild stroke. Rather, the findings provide “more certain, but not definitive, evidence” of a lack of benefit with alteplase over aspirin in this patient population (JAMA. 2018;320[2]:141-3).
The findings do suggest that “for these patients, treatment with aspirin along with close monitoring may be an appropriate course of action.”
The PRISMS trial was sponsored by Genentech. Dr. Khatri and many of her colleagues reported receiving personal fees from Genentech, some of which were for serving on the steering committee of the trial. Many investigators reported various financial ties to companies involved in cerebrovascular disease treatment.
Dr. Powers reported having no disclosures.
SOURCE: Khatri P et al. JAMA. 2018;320[2]:156-66.
Treatment with alteplase vs. aspirin did not improve functional outcomes at 90 days in patients with minor nondisabling acute ischemic stroke in the randomized phase 3b PRISMS trial.
At 90 days following a minor stroke judged to be nondisabling, a favorable functional outcome – defined as modified Rankin Scale (mRS) score of 0 or 1 – occurred in 122 (78.2%) of 156 patients who received alteplase and in 128 (81.5%) of 157 who received aspirin (adjusted risk difference, –1.1%), wrote Pooja Khatri, MD, of the University of Cincinnati, and her colleagues. The report was published in the July 10 issue of JAMA.
The PRISMS (Potential of rtPA for Ischemic Strokes with Mild Symptoms) trial was intended as a 948-patient, double-blind, placebo-controlled U.S. trial comparing alteplase and aspirin for emergent stroke in patients with National Institutes of Health Stroke Scale (NIHSS) scores of 0-5 at presentation whose stroke-related neurologic deficits were not clearly disabling and in whom the study treatment could be initiated within 3 hours. However, the trial was terminated early by the sponsor – prior to unblinding or interim analyses – because of below-target enrollment. An original plan to measure the difference in favorable functional outcome in the treatment and placebo groups by Cochran-Mantel-Haenszel hypothesis test with stratification by pretreatment NIHSS score, age, and time from onset to treatment was therefore revised to examine the risk difference of the primary outcome by a linear model adjusted for those factors, the authors explained.
Patients in the study had a mean age of 62 years and mean NIHSS score of 2, and were enrolled between May 30, 2014, and Dec. 20, 2016. They received either intravenous alteplase at a standard dose of 0.9 mg/kg with oral placebo, or oral aspirin at a dose of 325 mg with intravenous placebo, and were followed until March 22, 2017.
The primary safety endpoint of the analysis was symptomatic intracranial hemorrhage (sICH) within 36 hours of intravenous alteplase; this occurred in 5 (3.2%) of the alteplase-treated patients and in none of aspirin-treated patients (risk difference, 3.3%).
“Secondary outcomes, including the ordinal analysis of mRS scores (odds ratio, 0.81) and global favorable recovery (OR, 0.86), did not significantly favor either group,” the investigators added.
The findings are noteworthy because more than half of all patients with acute ischemic stroke have minor neurologic deficits, the investigators said, and while prior studies of alteplase included patients with low NIHSS score, few included patients who had no clearly disabling deficits. They added that while “alteplase is the standard of care for patients with ischemic stroke and disabling deficits regardless of severity judged by NIHSS scores, the optimal management of patients with not clearly disabling deficits is unclear.
“The study results raise the hypothesis that even a 6% treatment effect might be unlikely. However, the very early study termination precludes any definitive conclusions,” they wrote, noting that the study has several other limitations, including possible selection bias, relatively high loss to follow-up, and the subjective nature of the definition of “not clearly disabling.”
Additional research may be warranted, they said.
In an editorial, William J. Powers, MD, of the University of North Carolina at Chapel Hill, wrote that despite the limitations of the PRISMS trial, the findings help define the role for intravenous alteplase in the management of acute ischemic stroke.
“Even with early study termination and resultant wide 95% confidence intervals, the excellent outcome in the aspirin group and the numerically similar outcomes between the two groups render it unlikely that intravenous alteplase treatment meaningfully improves functional outcome in patients with initial NIHSS scores of 5 or lower with nondisabling deficits,” he wrote.
He noted, however – as did the study authors – that these conclusions do not apply to all patients with mild stroke. Rather, the findings provide “more certain, but not definitive, evidence” of a lack of benefit with alteplase over aspirin in this patient population (JAMA. 2018;320[2]:141-3).
The findings do suggest that “for these patients, treatment with aspirin along with close monitoring may be an appropriate course of action.”
The PRISMS trial was sponsored by Genentech. Dr. Khatri and many of her colleagues reported receiving personal fees from Genentech, some of which were for serving on the steering committee of the trial. Many investigators reported various financial ties to companies involved in cerebrovascular disease treatment.
Dr. Powers reported having no disclosures.
SOURCE: Khatri P et al. JAMA. 2018;320[2]:156-66.
Treatment with alteplase vs. aspirin did not improve functional outcomes at 90 days in patients with minor nondisabling acute ischemic stroke in the randomized phase 3b PRISMS trial.
At 90 days following a minor stroke judged to be nondisabling, a favorable functional outcome – defined as modified Rankin Scale (mRS) score of 0 or 1 – occurred in 122 (78.2%) of 156 patients who received alteplase and in 128 (81.5%) of 157 who received aspirin (adjusted risk difference, –1.1%), wrote Pooja Khatri, MD, of the University of Cincinnati, and her colleagues. The report was published in the July 10 issue of JAMA.
The PRISMS (Potential of rtPA for Ischemic Strokes with Mild Symptoms) trial was intended as a 948-patient, double-blind, placebo-controlled U.S. trial comparing alteplase and aspirin for emergent stroke in patients with National Institutes of Health Stroke Scale (NIHSS) scores of 0-5 at presentation whose stroke-related neurologic deficits were not clearly disabling and in whom the study treatment could be initiated within 3 hours. However, the trial was terminated early by the sponsor – prior to unblinding or interim analyses – because of below-target enrollment. An original plan to measure the difference in favorable functional outcome in the treatment and placebo groups by Cochran-Mantel-Haenszel hypothesis test with stratification by pretreatment NIHSS score, age, and time from onset to treatment was therefore revised to examine the risk difference of the primary outcome by a linear model adjusted for those factors, the authors explained.
Patients in the study had a mean age of 62 years and mean NIHSS score of 2, and were enrolled between May 30, 2014, and Dec. 20, 2016. They received either intravenous alteplase at a standard dose of 0.9 mg/kg with oral placebo, or oral aspirin at a dose of 325 mg with intravenous placebo, and were followed until March 22, 2017.
The primary safety endpoint of the analysis was symptomatic intracranial hemorrhage (sICH) within 36 hours of intravenous alteplase; this occurred in 5 (3.2%) of the alteplase-treated patients and in none of aspirin-treated patients (risk difference, 3.3%).
“Secondary outcomes, including the ordinal analysis of mRS scores (odds ratio, 0.81) and global favorable recovery (OR, 0.86), did not significantly favor either group,” the investigators added.
The findings are noteworthy because more than half of all patients with acute ischemic stroke have minor neurologic deficits, the investigators said, and while prior studies of alteplase included patients with low NIHSS score, few included patients who had no clearly disabling deficits. They added that while “alteplase is the standard of care for patients with ischemic stroke and disabling deficits regardless of severity judged by NIHSS scores, the optimal management of patients with not clearly disabling deficits is unclear.
“The study results raise the hypothesis that even a 6% treatment effect might be unlikely. However, the very early study termination precludes any definitive conclusions,” they wrote, noting that the study has several other limitations, including possible selection bias, relatively high loss to follow-up, and the subjective nature of the definition of “not clearly disabling.”
Additional research may be warranted, they said.
In an editorial, William J. Powers, MD, of the University of North Carolina at Chapel Hill, wrote that despite the limitations of the PRISMS trial, the findings help define the role for intravenous alteplase in the management of acute ischemic stroke.
“Even with early study termination and resultant wide 95% confidence intervals, the excellent outcome in the aspirin group and the numerically similar outcomes between the two groups render it unlikely that intravenous alteplase treatment meaningfully improves functional outcome in patients with initial NIHSS scores of 5 or lower with nondisabling deficits,” he wrote.
He noted, however – as did the study authors – that these conclusions do not apply to all patients with mild stroke. Rather, the findings provide “more certain, but not definitive, evidence” of a lack of benefit with alteplase over aspirin in this patient population (JAMA. 2018;320[2]:141-3).
The findings do suggest that “for these patients, treatment with aspirin along with close monitoring may be an appropriate course of action.”
The PRISMS trial was sponsored by Genentech. Dr. Khatri and many of her colleagues reported receiving personal fees from Genentech, some of which were for serving on the steering committee of the trial. Many investigators reported various financial ties to companies involved in cerebrovascular disease treatment.
Dr. Powers reported having no disclosures.
SOURCE: Khatri P et al. JAMA. 2018;320[2]:156-66.
FROM JAMA
Key clinical point: Alteplase does not appear to offer benefit over aspirin in terms of functional outcomes after nondisabling acute ischemic stroke.
Major finding: No significant difference was seen in functional outcomes with alteplase vs. aspirin at 90 days (adjusted risk difference, –1.1%).
Study details: The phase 3b PRISMS trial, involving 313 patients.
Disclosures: Genentech sponsored the trial. Dr. Khatri and many of her colleagues reported receiving personal fees from Genentech, some of which were for serving on the steering committee of the trial. Many investigators reported various financial ties to companies involved in cerebrovascular disease treatment. Dr. Powers reported having no disclosures.
Source: Khatri P et al. JAMA. 2018; 320[2]:156-66.
















