User login
ID Practitioner is an independent news source that provides infectious disease specialists with timely and relevant news and commentary about clinical developments and the impact of health care policy on the infectious disease specialist’s practice. Specialty focus topics include antimicrobial resistance, emerging infections, global ID, hepatitis, HIV, hospital-acquired infections, immunizations and vaccines, influenza, mycoses, pediatric infections, and STIs. Infectious Diseases News is owned by Frontline Medical Communications.
sofosbuvir
ritonavir with dasabuvir
discount
support path
program
ritonavir
greedy
ledipasvir
assistance
viekira pak
vpak
advocacy
needy
protest
abbvie
paritaprevir
ombitasvir
direct-acting antivirals
dasabuvir
gilead
fake-ovir
support
v pak
oasis
harvoni
section[contains(@class, 'footer-nav-section-wrapper')]
div[contains(@class, 'pane-pub-article-idp')]
div[contains(@class, 'pane-medstat-latest-articles-articles-section')]
div[contains(@class, 'pane-pub-home-idp')]
div[contains(@class, 'pane-pub-topic-idp')]
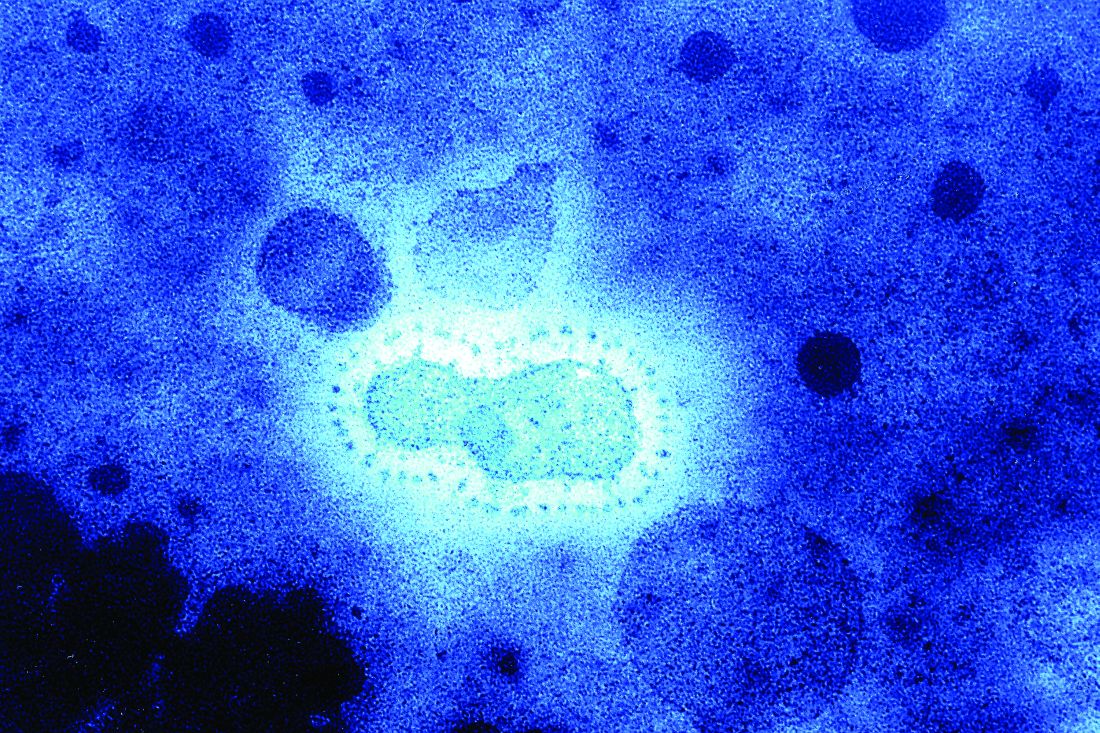
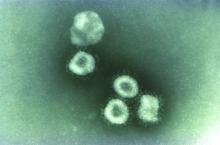
Wuhan coronavirus cluster suggests human-to-human spread
A Chinese man became ill from a novel coronavirus (2019-nCoV) 4 days after arriving in Vietnam to visit his 27-year-old son. Three days later the healthy young man was also stricken, according to a report published online Jan. 28 in the New England Journal of Medicine.
“This family cluster of 2019-nCoV infection that occurred outside China arouses concern regarding human-to-human transmission,” the authors wrote.
The father, age 65 years and with multiple comorbidities including hypertension, type 2 diabetes, coronary heart disease with stent placement, and lung cancer, flew to Hanoi with his wife on January 13; they traveled from the Wuchang district in Wuhan, China, where outbreaks of 2019-nCoV have been occurring.
On Jan. 17, the older man and his wife met their adult son in Ho Chi Minh City, Vietnam, and shared a hotel room with him for 3 days. The father developed a fever that same day and the son developed a dry cough, fever, diarrhea, and vomiting on Jan. 20. Both men went to a hospital ED on Jan. 22.
The authors say the timing of the son’s symptoms suggests the incubation period may have been 3 days or fewer.
Upon admission to the hospital, the father reported that he had not visited a “wet market” where live and dead animals are sold while he was in Wuhan. Throat swabs were positive for 2019-nCoV on real-time reverse-transcription–polymerase-chain-reaction assays.
The man was placed in isolation and “treated empirically with antiviral agents, broad-spectrum antibiotics, and supportive therapies,” wrote Lan T. Phan, PhD, from the Pasteur Institute Ho Chi Minh City and coauthors.
On admission, chest radiographs revealed an infiltrate in the upper lobe of his left lung; he developed worsening dyspnea with hypoxemia on Jan. 25 and required supplemental oxygen at 5 L/min by nasal cannula. Chest radiographs showed a progressive infiltrate and consolidation. His fever resolved on that day and he has progressively improved.
The man’s son had a fever of 39° C (102.2° F) when the two men arrived at the hospital on Jan. 22; hospital staff isolated the son, and chest radiographs and other laboratory tests were normal with the exception of an increased C-reactive protein level.
The son’s throat swab was positive for 2019-nCoV and he is believed to have been exposed from his father; however, the strains have not been ascertained.
“This family had traveled to four cities across Vietnam using various forms of transportation, including planes, trains, and taxis,” the authors wrote. A total of 28 close contacts were identified, none of whom have developed respiratory symptoms. The older man’s wife has been healthy as well.
The authors have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
A Chinese man became ill from a novel coronavirus (2019-nCoV) 4 days after arriving in Vietnam to visit his 27-year-old son. Three days later the healthy young man was also stricken, according to a report published online Jan. 28 in the New England Journal of Medicine.
“This family cluster of 2019-nCoV infection that occurred outside China arouses concern regarding human-to-human transmission,” the authors wrote.
The father, age 65 years and with multiple comorbidities including hypertension, type 2 diabetes, coronary heart disease with stent placement, and lung cancer, flew to Hanoi with his wife on January 13; they traveled from the Wuchang district in Wuhan, China, where outbreaks of 2019-nCoV have been occurring.
On Jan. 17, the older man and his wife met their adult son in Ho Chi Minh City, Vietnam, and shared a hotel room with him for 3 days. The father developed a fever that same day and the son developed a dry cough, fever, diarrhea, and vomiting on Jan. 20. Both men went to a hospital ED on Jan. 22.
The authors say the timing of the son’s symptoms suggests the incubation period may have been 3 days or fewer.
Upon admission to the hospital, the father reported that he had not visited a “wet market” where live and dead animals are sold while he was in Wuhan. Throat swabs were positive for 2019-nCoV on real-time reverse-transcription–polymerase-chain-reaction assays.
The man was placed in isolation and “treated empirically with antiviral agents, broad-spectrum antibiotics, and supportive therapies,” wrote Lan T. Phan, PhD, from the Pasteur Institute Ho Chi Minh City and coauthors.
On admission, chest radiographs revealed an infiltrate in the upper lobe of his left lung; he developed worsening dyspnea with hypoxemia on Jan. 25 and required supplemental oxygen at 5 L/min by nasal cannula. Chest radiographs showed a progressive infiltrate and consolidation. His fever resolved on that day and he has progressively improved.
The man’s son had a fever of 39° C (102.2° F) when the two men arrived at the hospital on Jan. 22; hospital staff isolated the son, and chest radiographs and other laboratory tests were normal with the exception of an increased C-reactive protein level.
The son’s throat swab was positive for 2019-nCoV and he is believed to have been exposed from his father; however, the strains have not been ascertained.
“This family had traveled to four cities across Vietnam using various forms of transportation, including planes, trains, and taxis,” the authors wrote. A total of 28 close contacts were identified, none of whom have developed respiratory symptoms. The older man’s wife has been healthy as well.
The authors have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
A Chinese man became ill from a novel coronavirus (2019-nCoV) 4 days after arriving in Vietnam to visit his 27-year-old son. Three days later the healthy young man was also stricken, according to a report published online Jan. 28 in the New England Journal of Medicine.
“This family cluster of 2019-nCoV infection that occurred outside China arouses concern regarding human-to-human transmission,” the authors wrote.
The father, age 65 years and with multiple comorbidities including hypertension, type 2 diabetes, coronary heart disease with stent placement, and lung cancer, flew to Hanoi with his wife on January 13; they traveled from the Wuchang district in Wuhan, China, where outbreaks of 2019-nCoV have been occurring.
On Jan. 17, the older man and his wife met their adult son in Ho Chi Minh City, Vietnam, and shared a hotel room with him for 3 days. The father developed a fever that same day and the son developed a dry cough, fever, diarrhea, and vomiting on Jan. 20. Both men went to a hospital ED on Jan. 22.
The authors say the timing of the son’s symptoms suggests the incubation period may have been 3 days or fewer.
Upon admission to the hospital, the father reported that he had not visited a “wet market” where live and dead animals are sold while he was in Wuhan. Throat swabs were positive for 2019-nCoV on real-time reverse-transcription–polymerase-chain-reaction assays.
The man was placed in isolation and “treated empirically with antiviral agents, broad-spectrum antibiotics, and supportive therapies,” wrote Lan T. Phan, PhD, from the Pasteur Institute Ho Chi Minh City and coauthors.
On admission, chest radiographs revealed an infiltrate in the upper lobe of his left lung; he developed worsening dyspnea with hypoxemia on Jan. 25 and required supplemental oxygen at 5 L/min by nasal cannula. Chest radiographs showed a progressive infiltrate and consolidation. His fever resolved on that day and he has progressively improved.
The man’s son had a fever of 39° C (102.2° F) when the two men arrived at the hospital on Jan. 22; hospital staff isolated the son, and chest radiographs and other laboratory tests were normal with the exception of an increased C-reactive protein level.
The son’s throat swab was positive for 2019-nCoV and he is believed to have been exposed from his father; however, the strains have not been ascertained.
“This family had traveled to four cities across Vietnam using various forms of transportation, including planes, trains, and taxis,” the authors wrote. A total of 28 close contacts were identified, none of whom have developed respiratory symptoms. The older man’s wife has been healthy as well.
The authors have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Echoes of SARS mark 2019 novel coronavirus outbreak
The current outbreak of severe respiratory infections caused by the 2019 novel coronarvirus (2019-nCoV) has a clinical presentation resembling the Severe Acute Respiratory Syndrome coronavirus (SARS-CoV) outbreak that began in 2002, Chinese investigators caution.

By Jan. 2, 2020, 41 patients with confirmed 2019-nCoV had been admitted to a designated hospital in the city of Wuhan, Hubei Province, in central China. Thirteen required ICU admission and six died, reported Chaolin Huang, MD, from Jin Yin-tan Hospital in Wuhan, and colleagues.
“2019-nCoV still needs to be studied deeply in case it becomes a global health threat. Reliable quick pathogen tests and feasible differential diagnosis based on clinical description are crucial for clinicians in their first contact with suspected patients. Because of the pandemic potential of 2019-nCoV, careful surveillance is essential to monitor its future host adaption, viral evolution, infectivity, transmissibility, and pathogenicity,” they wrote in a review published online by The Lancet.
According to the U.S. Centers for Disease Control and Prevention, as of Jan. 28, 2020, the total number of 2019-nCoV cases reported in the United States stood at five, but further cases of the infection – which Chinese health officials have confirmed can be transmitted person-to-person – are expected.
Dr. Huang and colleagues note that although most human coronavirus infections are mild, SARS-CoV and the Middle East respiratory syndrome coronavirus (MERS-CoV) were responsible for more than 10,000 infections, with mortality rates ranging from 10% with SARS to 37% with MERS. To date, 2019-nCoV has “caused clusters of fatal pneumonia greatly resembling SARS-CoV,” they write.
The authors studied the epidemiological, clinical, laboratory, and radiological characteristics as well as treatments and clinical outcomes of 41 patients admitted or transferred to the Jin Yin-tan Hospital with laboratory-confirmed 2019-nCoV infections.
The median patient age was 49 years. Thirty of the 41 patients (73%) were male. Comorbid conditions included diabetes in 13 of the 41 patients (32%), hypertension in 6 (15%), and cardiovascular disease in 6.
In all 27 of the 41 patients had been exposed to the Huanan seafood market in Wuhan, the suspected epicenter of the outbreak that was shut down by health authorities on Jan. 1 of this year.
The most common symptoms at the onset of the illness were fever in all but one of the 41 patients, cough in 31, and myalgia or fatigue in 18. Other, less frequent symptoms included sputum production in 11, headache in three, hemoptysis in two, and diarrhea in one.
“In this cohort, most patients presented with fever, dry cough, dyspnoea, and bilateral ground-glass opacities on chest CT scans. These features of 2019-nCoV infection bear some resemblance to SARS-CoV and MERS-CoV infections. However, few patients with 2019-nCoV infection had prominent upper respiratory tract signs and symptoms (e.g., rhinorrhoea, sneezing, or sore throat), indicating that the target cells might be located in the lower airway. Furthermore, 2019-nCoV patients rarely developed intestinal signs and symptoms (e.g., diarrhoea), whereas about 20%-25% of patients with MERS-CoV or SARS-CoV infection had diarrhoea.”
In all, 22 patients developed dyspnea, with a median time from illness onset to dyspnea of 8 days. The median time from illness onset to admission was 7 days, median time to shortness of breath was 8 days, median time to acute respiratory distress syndrome (ARDS) was 9 days, and median time to both mechanical ventilation and ICU admission was 10.5 days.
All of the patients developed pneumonia with abnormal findings on chest CT scan. In addition, 12 patients developed ARDS, six had RNAaemia, five developed acute cardiac injury, and four developed a secondary infection. As noted before, 13 of the 14 patients were admitted to an ICU, and six died. RNAaemia is a positive result for real-time polymerase chain reaction in plasma samples. Patients admitted to the ICU had higher initial concentrations of multiple inflammatory cytokines than patients who did not need ICU care, “suggesting that the cytokine storm was associated with disease severity.”
All of the patients received empirical antibiotics, 38 were treated with oseltamivir (Tamiflu), and 9 received systemic corticosteroids.
The investigators have initiated a randomized controlled trial of the antiviral agents lopinavir and ritonavir for patients hospitalized with 2019-nCoV infection.
The study was funded by the Chinese Ministry of Science and Technology, Chinese Academy of Medical Sciences, National Natural Science Foundation of China, and Beijing Municipal Science and Technology Commission. All authors declared having no competing interests.
SOURCE: Huang C et al. Lancet. 2020 Jan 24. doi: 10.1016/S0140-6736(20)30183-5.
The current outbreak of severe respiratory infections caused by the 2019 novel coronarvirus (2019-nCoV) has a clinical presentation resembling the Severe Acute Respiratory Syndrome coronavirus (SARS-CoV) outbreak that began in 2002, Chinese investigators caution.

By Jan. 2, 2020, 41 patients with confirmed 2019-nCoV had been admitted to a designated hospital in the city of Wuhan, Hubei Province, in central China. Thirteen required ICU admission and six died, reported Chaolin Huang, MD, from Jin Yin-tan Hospital in Wuhan, and colleagues.
“2019-nCoV still needs to be studied deeply in case it becomes a global health threat. Reliable quick pathogen tests and feasible differential diagnosis based on clinical description are crucial for clinicians in their first contact with suspected patients. Because of the pandemic potential of 2019-nCoV, careful surveillance is essential to monitor its future host adaption, viral evolution, infectivity, transmissibility, and pathogenicity,” they wrote in a review published online by The Lancet.
According to the U.S. Centers for Disease Control and Prevention, as of Jan. 28, 2020, the total number of 2019-nCoV cases reported in the United States stood at five, but further cases of the infection – which Chinese health officials have confirmed can be transmitted person-to-person – are expected.
Dr. Huang and colleagues note that although most human coronavirus infections are mild, SARS-CoV and the Middle East respiratory syndrome coronavirus (MERS-CoV) were responsible for more than 10,000 infections, with mortality rates ranging from 10% with SARS to 37% with MERS. To date, 2019-nCoV has “caused clusters of fatal pneumonia greatly resembling SARS-CoV,” they write.
The authors studied the epidemiological, clinical, laboratory, and radiological characteristics as well as treatments and clinical outcomes of 41 patients admitted or transferred to the Jin Yin-tan Hospital with laboratory-confirmed 2019-nCoV infections.
The median patient age was 49 years. Thirty of the 41 patients (73%) were male. Comorbid conditions included diabetes in 13 of the 41 patients (32%), hypertension in 6 (15%), and cardiovascular disease in 6.
In all 27 of the 41 patients had been exposed to the Huanan seafood market in Wuhan, the suspected epicenter of the outbreak that was shut down by health authorities on Jan. 1 of this year.
The most common symptoms at the onset of the illness were fever in all but one of the 41 patients, cough in 31, and myalgia or fatigue in 18. Other, less frequent symptoms included sputum production in 11, headache in three, hemoptysis in two, and diarrhea in one.
“In this cohort, most patients presented with fever, dry cough, dyspnoea, and bilateral ground-glass opacities on chest CT scans. These features of 2019-nCoV infection bear some resemblance to SARS-CoV and MERS-CoV infections. However, few patients with 2019-nCoV infection had prominent upper respiratory tract signs and symptoms (e.g., rhinorrhoea, sneezing, or sore throat), indicating that the target cells might be located in the lower airway. Furthermore, 2019-nCoV patients rarely developed intestinal signs and symptoms (e.g., diarrhoea), whereas about 20%-25% of patients with MERS-CoV or SARS-CoV infection had diarrhoea.”
In all, 22 patients developed dyspnea, with a median time from illness onset to dyspnea of 8 days. The median time from illness onset to admission was 7 days, median time to shortness of breath was 8 days, median time to acute respiratory distress syndrome (ARDS) was 9 days, and median time to both mechanical ventilation and ICU admission was 10.5 days.
All of the patients developed pneumonia with abnormal findings on chest CT scan. In addition, 12 patients developed ARDS, six had RNAaemia, five developed acute cardiac injury, and four developed a secondary infection. As noted before, 13 of the 14 patients were admitted to an ICU, and six died. RNAaemia is a positive result for real-time polymerase chain reaction in plasma samples. Patients admitted to the ICU had higher initial concentrations of multiple inflammatory cytokines than patients who did not need ICU care, “suggesting that the cytokine storm was associated with disease severity.”
All of the patients received empirical antibiotics, 38 were treated with oseltamivir (Tamiflu), and 9 received systemic corticosteroids.
The investigators have initiated a randomized controlled trial of the antiviral agents lopinavir and ritonavir for patients hospitalized with 2019-nCoV infection.
The study was funded by the Chinese Ministry of Science and Technology, Chinese Academy of Medical Sciences, National Natural Science Foundation of China, and Beijing Municipal Science and Technology Commission. All authors declared having no competing interests.
SOURCE: Huang C et al. Lancet. 2020 Jan 24. doi: 10.1016/S0140-6736(20)30183-5.
The current outbreak of severe respiratory infections caused by the 2019 novel coronarvirus (2019-nCoV) has a clinical presentation resembling the Severe Acute Respiratory Syndrome coronavirus (SARS-CoV) outbreak that began in 2002, Chinese investigators caution.

By Jan. 2, 2020, 41 patients with confirmed 2019-nCoV had been admitted to a designated hospital in the city of Wuhan, Hubei Province, in central China. Thirteen required ICU admission and six died, reported Chaolin Huang, MD, from Jin Yin-tan Hospital in Wuhan, and colleagues.
“2019-nCoV still needs to be studied deeply in case it becomes a global health threat. Reliable quick pathogen tests and feasible differential diagnosis based on clinical description are crucial for clinicians in their first contact with suspected patients. Because of the pandemic potential of 2019-nCoV, careful surveillance is essential to monitor its future host adaption, viral evolution, infectivity, transmissibility, and pathogenicity,” they wrote in a review published online by The Lancet.
According to the U.S. Centers for Disease Control and Prevention, as of Jan. 28, 2020, the total number of 2019-nCoV cases reported in the United States stood at five, but further cases of the infection – which Chinese health officials have confirmed can be transmitted person-to-person – are expected.
Dr. Huang and colleagues note that although most human coronavirus infections are mild, SARS-CoV and the Middle East respiratory syndrome coronavirus (MERS-CoV) were responsible for more than 10,000 infections, with mortality rates ranging from 10% with SARS to 37% with MERS. To date, 2019-nCoV has “caused clusters of fatal pneumonia greatly resembling SARS-CoV,” they write.
The authors studied the epidemiological, clinical, laboratory, and radiological characteristics as well as treatments and clinical outcomes of 41 patients admitted or transferred to the Jin Yin-tan Hospital with laboratory-confirmed 2019-nCoV infections.
The median patient age was 49 years. Thirty of the 41 patients (73%) were male. Comorbid conditions included diabetes in 13 of the 41 patients (32%), hypertension in 6 (15%), and cardiovascular disease in 6.
In all 27 of the 41 patients had been exposed to the Huanan seafood market in Wuhan, the suspected epicenter of the outbreak that was shut down by health authorities on Jan. 1 of this year.
The most common symptoms at the onset of the illness were fever in all but one of the 41 patients, cough in 31, and myalgia or fatigue in 18. Other, less frequent symptoms included sputum production in 11, headache in three, hemoptysis in two, and diarrhea in one.
“In this cohort, most patients presented with fever, dry cough, dyspnoea, and bilateral ground-glass opacities on chest CT scans. These features of 2019-nCoV infection bear some resemblance to SARS-CoV and MERS-CoV infections. However, few patients with 2019-nCoV infection had prominent upper respiratory tract signs and symptoms (e.g., rhinorrhoea, sneezing, or sore throat), indicating that the target cells might be located in the lower airway. Furthermore, 2019-nCoV patients rarely developed intestinal signs and symptoms (e.g., diarrhoea), whereas about 20%-25% of patients with MERS-CoV or SARS-CoV infection had diarrhoea.”
In all, 22 patients developed dyspnea, with a median time from illness onset to dyspnea of 8 days. The median time from illness onset to admission was 7 days, median time to shortness of breath was 8 days, median time to acute respiratory distress syndrome (ARDS) was 9 days, and median time to both mechanical ventilation and ICU admission was 10.5 days.
All of the patients developed pneumonia with abnormal findings on chest CT scan. In addition, 12 patients developed ARDS, six had RNAaemia, five developed acute cardiac injury, and four developed a secondary infection. As noted before, 13 of the 14 patients were admitted to an ICU, and six died. RNAaemia is a positive result for real-time polymerase chain reaction in plasma samples. Patients admitted to the ICU had higher initial concentrations of multiple inflammatory cytokines than patients who did not need ICU care, “suggesting that the cytokine storm was associated with disease severity.”
All of the patients received empirical antibiotics, 38 were treated with oseltamivir (Tamiflu), and 9 received systemic corticosteroids.
The investigators have initiated a randomized controlled trial of the antiviral agents lopinavir and ritonavir for patients hospitalized with 2019-nCoV infection.
The study was funded by the Chinese Ministry of Science and Technology, Chinese Academy of Medical Sciences, National Natural Science Foundation of China, and Beijing Municipal Science and Technology Commission. All authors declared having no competing interests.
SOURCE: Huang C et al. Lancet. 2020 Jan 24. doi: 10.1016/S0140-6736(20)30183-5.
FROM THE LANCET
CDC: Five confirmed 2019-nCoV cases in the U.S.
Five cases of the new infectious coronavirus, 2019-nCoV, have been confirmed in the United States, Nancy Messonnier, MD, director of the National Center for Immunization and Respiratory Diseases at the Centers for Disease Control and Prevention, said during a Jan. 27 press briefing.
A total of 110 individuals are under investigation in 26 states, she said. While five cases have been confirmed positive for the virus, 32 cases were confirmed negative. There have been no new cases overnight.
Last week, CDC scientists developed a real-time polymerase chain reaction (PCR) test that can diagnose the virus in respiratory and serum samples from clinical specimens. On Jan. 24, the protocol for this test was publicly posted. “This is essentially a blueprint to make the test,” Dr. Messonnier explained. “Currently, we are refining the use of the test so that it can provide optimal guidance to states and labs on how to use it. We are working on a plan so that priority states get these test kits as soon as possible. In the coming weeks, we will share these tests with domestic and international partners so they can test for this virus themselves.”
The CDC uploaded the entire genome of the virus from the first two cases in the United States to GenBank. It was similar to the one that China had previously posted. “Right now, based on CDC’s analysis of the available data, it doesn’t look like the virus has mutated,” she said. “And we are growing the virus in cell culture, which is necessary for further studies, including the additional genetic characterization.”
As of today, 16 international locations, including the United States, have identified cases of the virus. CDC officials are continuing to screen passengers from Wuhan, China, at five designated airports. “This serves two purposes: first to detect the illness and rapidly respond to [affected] people entering the country,” Dr. Messonnier said. “The second purpose is to educate travelers about the symptoms of this new virus, and what to do if they develop symptoms. I expect that in the coming days, our travel recommendations will change. Risk depends on exposure. Right now, we have an handful of new patients with this new virus here in the U.S. However, at this time in the U.S., this virus is not spreading in the community. For that reason, we believe that the immediate health risk of the new virus to the general American public is low.”
The CDC is asking its clinical lab partners to send virus samples to the CDC to ensure that results are analyzed as accurately as possible.
Five cases of the new infectious coronavirus, 2019-nCoV, have been confirmed in the United States, Nancy Messonnier, MD, director of the National Center for Immunization and Respiratory Diseases at the Centers for Disease Control and Prevention, said during a Jan. 27 press briefing.
A total of 110 individuals are under investigation in 26 states, she said. While five cases have been confirmed positive for the virus, 32 cases were confirmed negative. There have been no new cases overnight.
Last week, CDC scientists developed a real-time polymerase chain reaction (PCR) test that can diagnose the virus in respiratory and serum samples from clinical specimens. On Jan. 24, the protocol for this test was publicly posted. “This is essentially a blueprint to make the test,” Dr. Messonnier explained. “Currently, we are refining the use of the test so that it can provide optimal guidance to states and labs on how to use it. We are working on a plan so that priority states get these test kits as soon as possible. In the coming weeks, we will share these tests with domestic and international partners so they can test for this virus themselves.”
The CDC uploaded the entire genome of the virus from the first two cases in the United States to GenBank. It was similar to the one that China had previously posted. “Right now, based on CDC’s analysis of the available data, it doesn’t look like the virus has mutated,” she said. “And we are growing the virus in cell culture, which is necessary for further studies, including the additional genetic characterization.”
As of today, 16 international locations, including the United States, have identified cases of the virus. CDC officials are continuing to screen passengers from Wuhan, China, at five designated airports. “This serves two purposes: first to detect the illness and rapidly respond to [affected] people entering the country,” Dr. Messonnier said. “The second purpose is to educate travelers about the symptoms of this new virus, and what to do if they develop symptoms. I expect that in the coming days, our travel recommendations will change. Risk depends on exposure. Right now, we have an handful of new patients with this new virus here in the U.S. However, at this time in the U.S., this virus is not spreading in the community. For that reason, we believe that the immediate health risk of the new virus to the general American public is low.”
The CDC is asking its clinical lab partners to send virus samples to the CDC to ensure that results are analyzed as accurately as possible.
Five cases of the new infectious coronavirus, 2019-nCoV, have been confirmed in the United States, Nancy Messonnier, MD, director of the National Center for Immunization and Respiratory Diseases at the Centers for Disease Control and Prevention, said during a Jan. 27 press briefing.
A total of 110 individuals are under investigation in 26 states, she said. While five cases have been confirmed positive for the virus, 32 cases were confirmed negative. There have been no new cases overnight.
Last week, CDC scientists developed a real-time polymerase chain reaction (PCR) test that can diagnose the virus in respiratory and serum samples from clinical specimens. On Jan. 24, the protocol for this test was publicly posted. “This is essentially a blueprint to make the test,” Dr. Messonnier explained. “Currently, we are refining the use of the test so that it can provide optimal guidance to states and labs on how to use it. We are working on a plan so that priority states get these test kits as soon as possible. In the coming weeks, we will share these tests with domestic and international partners so they can test for this virus themselves.”
The CDC uploaded the entire genome of the virus from the first two cases in the United States to GenBank. It was similar to the one that China had previously posted. “Right now, based on CDC’s analysis of the available data, it doesn’t look like the virus has mutated,” she said. “And we are growing the virus in cell culture, which is necessary for further studies, including the additional genetic characterization.”
As of today, 16 international locations, including the United States, have identified cases of the virus. CDC officials are continuing to screen passengers from Wuhan, China, at five designated airports. “This serves two purposes: first to detect the illness and rapidly respond to [affected] people entering the country,” Dr. Messonnier said. “The second purpose is to educate travelers about the symptoms of this new virus, and what to do if they develop symptoms. I expect that in the coming days, our travel recommendations will change. Risk depends on exposure. Right now, we have an handful of new patients with this new virus here in the U.S. However, at this time in the U.S., this virus is not spreading in the community. For that reason, we believe that the immediate health risk of the new virus to the general American public is low.”
The CDC is asking its clinical lab partners to send virus samples to the CDC to ensure that results are analyzed as accurately as possible.
FDA approves fidaxomicin for treatment of C. difficile–associated diarrhea
The Food and Drug Administration has approved fidaxomicin (Dificid) for the treatment of Clostridioides difficile–associated diarrhea in children aged 6 months and older.
Approval was based on results from SUNSHINE, a phase 3, multicenter, investigator-blind, randomized, parallel-group study in 142 pediatric patients aged between 6 months and 18 years with confirmed C. difficile infection who received either fidaxomicin or vancomycin for 10 days. Clinical response 2 days after the conclusion of treatment was similar in both groups (77.6% for fidaxomicin vs. 70.5% for vancomycin), and fidaxomicin had a superior sustained response 30 days after the conclusion of treatment (68.4% vs. 50.0%).
The safety of fidaxomicin was assessed in a pair of clinical trials involving 136 patients; the most common adverse events were pyrexia, abdominal pain, vomiting, diarrhea, constipation, increased aminotransferases, and rash. Four patients discontinued fidaxomicin treatment because of adverse events, and four patients died during the trials, though all deaths were in patients aged younger than 2 years and seemed to be related to other comorbidities.
“C. difficile is an important cause of health care– and community-associated diarrheal illness in children, and sustained cure is difficult to achieve in some patients. The fidaxomicin pediatric trial was the first randomized, controlled trial of C. difficile infection treatment in children,” Larry K. Kociolek, MD, associate medical director of infection prevention and control at Ann & Robert H. Lurie Children’s Hospital of Chicago, said in the press release from Merck, manufacturer of fidaxomicin.
*This story was updated on 1/27/2020.
The Food and Drug Administration has approved fidaxomicin (Dificid) for the treatment of Clostridioides difficile–associated diarrhea in children aged 6 months and older.
Approval was based on results from SUNSHINE, a phase 3, multicenter, investigator-blind, randomized, parallel-group study in 142 pediatric patients aged between 6 months and 18 years with confirmed C. difficile infection who received either fidaxomicin or vancomycin for 10 days. Clinical response 2 days after the conclusion of treatment was similar in both groups (77.6% for fidaxomicin vs. 70.5% for vancomycin), and fidaxomicin had a superior sustained response 30 days after the conclusion of treatment (68.4% vs. 50.0%).
The safety of fidaxomicin was assessed in a pair of clinical trials involving 136 patients; the most common adverse events were pyrexia, abdominal pain, vomiting, diarrhea, constipation, increased aminotransferases, and rash. Four patients discontinued fidaxomicin treatment because of adverse events, and four patients died during the trials, though all deaths were in patients aged younger than 2 years and seemed to be related to other comorbidities.
“C. difficile is an important cause of health care– and community-associated diarrheal illness in children, and sustained cure is difficult to achieve in some patients. The fidaxomicin pediatric trial was the first randomized, controlled trial of C. difficile infection treatment in children,” Larry K. Kociolek, MD, associate medical director of infection prevention and control at Ann & Robert H. Lurie Children’s Hospital of Chicago, said in the press release from Merck, manufacturer of fidaxomicin.
*This story was updated on 1/27/2020.
The Food and Drug Administration has approved fidaxomicin (Dificid) for the treatment of Clostridioides difficile–associated diarrhea in children aged 6 months and older.
Approval was based on results from SUNSHINE, a phase 3, multicenter, investigator-blind, randomized, parallel-group study in 142 pediatric patients aged between 6 months and 18 years with confirmed C. difficile infection who received either fidaxomicin or vancomycin for 10 days. Clinical response 2 days after the conclusion of treatment was similar in both groups (77.6% for fidaxomicin vs. 70.5% for vancomycin), and fidaxomicin had a superior sustained response 30 days after the conclusion of treatment (68.4% vs. 50.0%).
The safety of fidaxomicin was assessed in a pair of clinical trials involving 136 patients; the most common adverse events were pyrexia, abdominal pain, vomiting, diarrhea, constipation, increased aminotransferases, and rash. Four patients discontinued fidaxomicin treatment because of adverse events, and four patients died during the trials, though all deaths were in patients aged younger than 2 years and seemed to be related to other comorbidities.
“C. difficile is an important cause of health care– and community-associated diarrheal illness in children, and sustained cure is difficult to achieve in some patients. The fidaxomicin pediatric trial was the first randomized, controlled trial of C. difficile infection treatment in children,” Larry K. Kociolek, MD, associate medical director of infection prevention and control at Ann & Robert H. Lurie Children’s Hospital of Chicago, said in the press release from Merck, manufacturer of fidaxomicin.
*This story was updated on 1/27/2020.
Zika virus: Birth defects rose fourfold in U.S. hardest-hit areas
according to the Centers for Disease Control and Prevention.
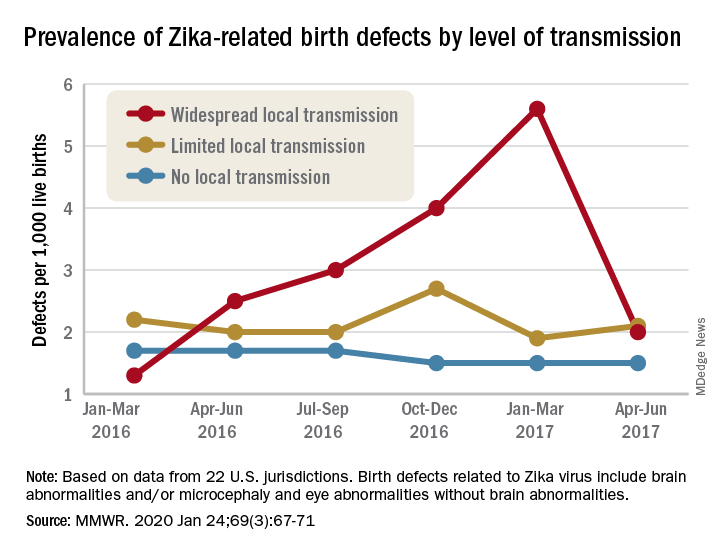
That spike in the prevalence of brain abnormalities and/or microcephaly or eye abnormalities without brain abnormalities came during January through March 2017, about 6 months after the Zika outbreak’s reported peak in the jurisdictions with widespread local transmission, Puerto Rico and the U.S. Virgin Islands, wrote Ashley N. Smoots, MPH, of the CDC’s National Center on Birth Defects and Developmental Disabilities and associates in the Morbidity and Mortality Weekly Report.
In those two territories, the prevalence of birth defects potentially related to Zika virus infection was 5.6 per 1,000 live births during January through March 2017, compared with 1.3 per 1,000 in January through March 2016, they reported.
In the southern areas of Florida and Texas, where there was limited local Zika transmission, the highest prevalence of birth defects, 2.7 per 1,000, occurred during October through December 2016, and was only slightly greater than the baseline rate of 2.2 per 1,000 in January through March 2016, the investigators reported.
Among the other 19 jurisdictions (including Illinois, Louisiana, New Jersey, South Carolina, and Virginia) involved in the analysis, the rate of Zika virus–related birth defects never reached any higher than the 1.7 per 1,000 recorded at the start of the study period in January through March 2016, they said.
“Population-based birth defects surveillance is critical for identifying infants and fetuses with birth defects potentially related to Zika virus regardless of whether Zika virus testing was conducted, especially given the high prevalence of asymptomatic disease. These data can be used to inform follow-up care and services as well as strengthen surveillance,” the investigators wrote.
SOURCE: Smoots AN et al. MMWR. 2020 Jan 24;69(3):67-71.
according to the Centers for Disease Control and Prevention.

That spike in the prevalence of brain abnormalities and/or microcephaly or eye abnormalities without brain abnormalities came during January through March 2017, about 6 months after the Zika outbreak’s reported peak in the jurisdictions with widespread local transmission, Puerto Rico and the U.S. Virgin Islands, wrote Ashley N. Smoots, MPH, of the CDC’s National Center on Birth Defects and Developmental Disabilities and associates in the Morbidity and Mortality Weekly Report.
In those two territories, the prevalence of birth defects potentially related to Zika virus infection was 5.6 per 1,000 live births during January through March 2017, compared with 1.3 per 1,000 in January through March 2016, they reported.
In the southern areas of Florida and Texas, where there was limited local Zika transmission, the highest prevalence of birth defects, 2.7 per 1,000, occurred during October through December 2016, and was only slightly greater than the baseline rate of 2.2 per 1,000 in January through March 2016, the investigators reported.
Among the other 19 jurisdictions (including Illinois, Louisiana, New Jersey, South Carolina, and Virginia) involved in the analysis, the rate of Zika virus–related birth defects never reached any higher than the 1.7 per 1,000 recorded at the start of the study period in January through March 2016, they said.
“Population-based birth defects surveillance is critical for identifying infants and fetuses with birth defects potentially related to Zika virus regardless of whether Zika virus testing was conducted, especially given the high prevalence of asymptomatic disease. These data can be used to inform follow-up care and services as well as strengthen surveillance,” the investigators wrote.
SOURCE: Smoots AN et al. MMWR. 2020 Jan 24;69(3):67-71.
according to the Centers for Disease Control and Prevention.

That spike in the prevalence of brain abnormalities and/or microcephaly or eye abnormalities without brain abnormalities came during January through March 2017, about 6 months after the Zika outbreak’s reported peak in the jurisdictions with widespread local transmission, Puerto Rico and the U.S. Virgin Islands, wrote Ashley N. Smoots, MPH, of the CDC’s National Center on Birth Defects and Developmental Disabilities and associates in the Morbidity and Mortality Weekly Report.
In those two territories, the prevalence of birth defects potentially related to Zika virus infection was 5.6 per 1,000 live births during January through March 2017, compared with 1.3 per 1,000 in January through March 2016, they reported.
In the southern areas of Florida and Texas, where there was limited local Zika transmission, the highest prevalence of birth defects, 2.7 per 1,000, occurred during October through December 2016, and was only slightly greater than the baseline rate of 2.2 per 1,000 in January through March 2016, the investigators reported.
Among the other 19 jurisdictions (including Illinois, Louisiana, New Jersey, South Carolina, and Virginia) involved in the analysis, the rate of Zika virus–related birth defects never reached any higher than the 1.7 per 1,000 recorded at the start of the study period in January through March 2016, they said.
“Population-based birth defects surveillance is critical for identifying infants and fetuses with birth defects potentially related to Zika virus regardless of whether Zika virus testing was conducted, especially given the high prevalence of asymptomatic disease. These data can be used to inform follow-up care and services as well as strengthen surveillance,” the investigators wrote.
SOURCE: Smoots AN et al. MMWR. 2020 Jan 24;69(3):67-71.
FROM MMWR
Wuhan virus: What clinicians need to know
As the Wuhan coronavirus story unfolds, , according to infectious disease experts.

“We are asking that of everyone with fever and respiratory symptoms who comes to our clinics, hospital, or emergency room. It’s a powerful screening tool,” said William Schaffner, MD, professor of preventive medicine and infectious diseases at Vanderbilt University Medical Center, Nashville, Tenn.
In addition to fever, common signs of infection include cough, shortness of breath, and breathing difficulties. Some patients have had diarrhea, vomiting, and other gastrointestinal symptoms. In more severe cases, infection can cause pneumonia, severe acute respiratory syndrome, kidney failure, and death. The incubation period appears to be up to 2 weeks, according to the World Health Organization (WHO).
If patients exhibit symptoms and either they or a close contact has returned from China recently, take standard airborne precautions and send specimens – a serum sample, oral and nasal pharyngeal swabs, and lower respiratory tract specimens if available – to the local health department, which will forward them to the Centers for Disease Control and Prevention (CDC) for testing. Turnaround time is 24-48 hours.
The 2019 Novel Coronavirus (2019-nCoV), identified as the cause of an outbreak of respiratory illness first detected in December in association with a live animal market in Wuhan, China, has been implicated in almost 2,000 cases and 56 deaths in that country. Cases have been reported in 13 countries besides China. Five cases of 2019-nCoV infection have been confirmed in the United States, all in people recently returned from Wuhan. As the virus spreads in China, however, it’s almost certain more cases will show up in the United States. Travel history is key, Dr. Schaffner and others said.
Plan and rehearse
The first step to prepare is to use the CDC’s Interim Guidance for Healthcare Professionals to make a written plan specific to your practice to respond to a potential case. The plan must include notifying the local health department, the CDC liaison for testing, and tracking down patient contacts.
“It’s not good enough to just download CDC’s guidance; use it to make your own local plan and know what to do 24/7,” said Daniel Lucey, MD, an infectious disease expert at Georgetown University Medical Center, Washington, D.C.
“Know who is on call at the health department on weekends and nights,” he said. Know where the patient is going to be isolated; figure out what to do if there’s more than one, and tests come back positive. Have masks on hand, and rehearse the response. “Make a coronavirus team, and absolutely have the nurses involved,” as well as other providers who may come into contact with a case, he added.
“You want to be able to do as well as your counterparts in Washington state and Chicago,” where the first two U.S. cases emerged. “They were prepared. They knew what to do,” Dr. Lucey said.
Those first two U.S. patients – a man in Everett, Wash., and a Chicago woman – developed symptoms after returning from Wuhan, a city of 11 million just over 400 miles inland from the port city of Shanghai. On Jan. 26 three more cases were confirmed by the CDC, two in California and one in Arizona, and each had recently traveled to Wuhan. All five patients remain hospitalized, and there’s no evidence they spread the infection further. There is also no evidence of human-to-human transmission of other cases exported from China to any other countries, according to the WHO.
WHO declined to declare a global health emergency – a Public Health Emergency of International Concern, in its parlance – on Jan. 23. The step would have triggered travel and trade restrictions in member states, including the United States. For now, at least, the group said it wasn’t warranted at this point.
Fatality rates
The focus right now is China. The outbreak has spread beyond Wuhan to other parts of the country, and there’s evidence of fourth-generation spread.
Transportation into and out of Wuhan and other cities has been curtailed, Lunar New Year festivals have been canceled, and the Shanghai Disneyland has been closed, among other measures taken by Chinese officials.
The government could be taking drastic measures in part to prevent the public criticism it took in the early 2000’s for the delayed response and lack of transparency during the global outbreak of another wildlife market coronavirus epidemic, severe acute respiratory syndrome (SARS). In a press conference Jan. 22, WHO officials commended the government’s containment efforts but did not say they recommended them.
According to WHO, serious cases in China have mostly been in people over 40 years old with significant comorbidities and have skewed towards men. Spread seems to be limited to family members, health care providers, and other close contacts, probably by respiratory droplets. If that pattern holds, WHO officials said, the outbreak is containable.
The fatality rate appears to be around 3%, a good deal lower than the 10% reported for SARS and much lower than the nearly 40% reported for Middle East respiratory syndrome (MERS), another recent coronavirus mutation from the animal trade.
The Wuhan virus fatality rate might drop as milder cases are detected and added to the denominator. “It definitely appears to be less severe than SARS and MERS,” said Amesh Adalja, MD, an infectious disease physician in Pittsburgh and emerging infectious disease researcher at Johns Hopkins University, Baltimore.
SARS: Lessons learned
In general, the world is much better equipped for coronavirus outbreaks than when SARS, in particular, emerged in 2003.
WHO officials in their press conference lauded China for it openness with the current outbreak, and for isolating and sequencing the virus immediately, which gave the world a diagnostic test in the first days of the outbreak, something that wasn’t available for SARS. China and other countries also are cooperating and working closely to contain the Wuhan virus.
“What we know today might change tomorrow, so we have to keep tuned in to new information, but we learned a lot from SARS,” Dr. Shaffner said. Overall, it’s likely “the impact on the United States of this new coronavirus is going to be trivial,” he predicted.
Dr. Lucey, however, recalled that the SARS outbreak in Toronto in 2003 started with one missed case. A woman returned asymptomatic from Hong Kong and spread the infection to her family members before she died. Her cause of death wasn’t immediately recognized, nor was the reason her family members were sick, since they hadn’t been to Hong Kong recently.
The infection ultimately spread to more than 200 people, about half of them health care workers. A few people died.
If a virus is sufficiently contagious, “it just takes one. You don’t want to be the one who misses that first patient,” Dr. Lucey said.
Currently, there are no antivirals or vaccines for coronaviruses; researchers are working on both, but for now, care is supportive.
This article was updated with new case numbers on 1/26/20.
As the Wuhan coronavirus story unfolds, , according to infectious disease experts.

“We are asking that of everyone with fever and respiratory symptoms who comes to our clinics, hospital, or emergency room. It’s a powerful screening tool,” said William Schaffner, MD, professor of preventive medicine and infectious diseases at Vanderbilt University Medical Center, Nashville, Tenn.
In addition to fever, common signs of infection include cough, shortness of breath, and breathing difficulties. Some patients have had diarrhea, vomiting, and other gastrointestinal symptoms. In more severe cases, infection can cause pneumonia, severe acute respiratory syndrome, kidney failure, and death. The incubation period appears to be up to 2 weeks, according to the World Health Organization (WHO).
If patients exhibit symptoms and either they or a close contact has returned from China recently, take standard airborne precautions and send specimens – a serum sample, oral and nasal pharyngeal swabs, and lower respiratory tract specimens if available – to the local health department, which will forward them to the Centers for Disease Control and Prevention (CDC) for testing. Turnaround time is 24-48 hours.
The 2019 Novel Coronavirus (2019-nCoV), identified as the cause of an outbreak of respiratory illness first detected in December in association with a live animal market in Wuhan, China, has been implicated in almost 2,000 cases and 56 deaths in that country. Cases have been reported in 13 countries besides China. Five cases of 2019-nCoV infection have been confirmed in the United States, all in people recently returned from Wuhan. As the virus spreads in China, however, it’s almost certain more cases will show up in the United States. Travel history is key, Dr. Schaffner and others said.
Plan and rehearse
The first step to prepare is to use the CDC’s Interim Guidance for Healthcare Professionals to make a written plan specific to your practice to respond to a potential case. The plan must include notifying the local health department, the CDC liaison for testing, and tracking down patient contacts.
“It’s not good enough to just download CDC’s guidance; use it to make your own local plan and know what to do 24/7,” said Daniel Lucey, MD, an infectious disease expert at Georgetown University Medical Center, Washington, D.C.
“Know who is on call at the health department on weekends and nights,” he said. Know where the patient is going to be isolated; figure out what to do if there’s more than one, and tests come back positive. Have masks on hand, and rehearse the response. “Make a coronavirus team, and absolutely have the nurses involved,” as well as other providers who may come into contact with a case, he added.
“You want to be able to do as well as your counterparts in Washington state and Chicago,” where the first two U.S. cases emerged. “They were prepared. They knew what to do,” Dr. Lucey said.
Those first two U.S. patients – a man in Everett, Wash., and a Chicago woman – developed symptoms after returning from Wuhan, a city of 11 million just over 400 miles inland from the port city of Shanghai. On Jan. 26 three more cases were confirmed by the CDC, two in California and one in Arizona, and each had recently traveled to Wuhan. All five patients remain hospitalized, and there’s no evidence they spread the infection further. There is also no evidence of human-to-human transmission of other cases exported from China to any other countries, according to the WHO.
WHO declined to declare a global health emergency – a Public Health Emergency of International Concern, in its parlance – on Jan. 23. The step would have triggered travel and trade restrictions in member states, including the United States. For now, at least, the group said it wasn’t warranted at this point.
Fatality rates
The focus right now is China. The outbreak has spread beyond Wuhan to other parts of the country, and there’s evidence of fourth-generation spread.
Transportation into and out of Wuhan and other cities has been curtailed, Lunar New Year festivals have been canceled, and the Shanghai Disneyland has been closed, among other measures taken by Chinese officials.
The government could be taking drastic measures in part to prevent the public criticism it took in the early 2000’s for the delayed response and lack of transparency during the global outbreak of another wildlife market coronavirus epidemic, severe acute respiratory syndrome (SARS). In a press conference Jan. 22, WHO officials commended the government’s containment efforts but did not say they recommended them.
According to WHO, serious cases in China have mostly been in people over 40 years old with significant comorbidities and have skewed towards men. Spread seems to be limited to family members, health care providers, and other close contacts, probably by respiratory droplets. If that pattern holds, WHO officials said, the outbreak is containable.
The fatality rate appears to be around 3%, a good deal lower than the 10% reported for SARS and much lower than the nearly 40% reported for Middle East respiratory syndrome (MERS), another recent coronavirus mutation from the animal trade.
The Wuhan virus fatality rate might drop as milder cases are detected and added to the denominator. “It definitely appears to be less severe than SARS and MERS,” said Amesh Adalja, MD, an infectious disease physician in Pittsburgh and emerging infectious disease researcher at Johns Hopkins University, Baltimore.
SARS: Lessons learned
In general, the world is much better equipped for coronavirus outbreaks than when SARS, in particular, emerged in 2003.
WHO officials in their press conference lauded China for it openness with the current outbreak, and for isolating and sequencing the virus immediately, which gave the world a diagnostic test in the first days of the outbreak, something that wasn’t available for SARS. China and other countries also are cooperating and working closely to contain the Wuhan virus.
“What we know today might change tomorrow, so we have to keep tuned in to new information, but we learned a lot from SARS,” Dr. Shaffner said. Overall, it’s likely “the impact on the United States of this new coronavirus is going to be trivial,” he predicted.
Dr. Lucey, however, recalled that the SARS outbreak in Toronto in 2003 started with one missed case. A woman returned asymptomatic from Hong Kong and spread the infection to her family members before she died. Her cause of death wasn’t immediately recognized, nor was the reason her family members were sick, since they hadn’t been to Hong Kong recently.
The infection ultimately spread to more than 200 people, about half of them health care workers. A few people died.
If a virus is sufficiently contagious, “it just takes one. You don’t want to be the one who misses that first patient,” Dr. Lucey said.
Currently, there are no antivirals or vaccines for coronaviruses; researchers are working on both, but for now, care is supportive.
This article was updated with new case numbers on 1/26/20.
As the Wuhan coronavirus story unfolds, , according to infectious disease experts.

“We are asking that of everyone with fever and respiratory symptoms who comes to our clinics, hospital, or emergency room. It’s a powerful screening tool,” said William Schaffner, MD, professor of preventive medicine and infectious diseases at Vanderbilt University Medical Center, Nashville, Tenn.
In addition to fever, common signs of infection include cough, shortness of breath, and breathing difficulties. Some patients have had diarrhea, vomiting, and other gastrointestinal symptoms. In more severe cases, infection can cause pneumonia, severe acute respiratory syndrome, kidney failure, and death. The incubation period appears to be up to 2 weeks, according to the World Health Organization (WHO).
If patients exhibit symptoms and either they or a close contact has returned from China recently, take standard airborne precautions and send specimens – a serum sample, oral and nasal pharyngeal swabs, and lower respiratory tract specimens if available – to the local health department, which will forward them to the Centers for Disease Control and Prevention (CDC) for testing. Turnaround time is 24-48 hours.
The 2019 Novel Coronavirus (2019-nCoV), identified as the cause of an outbreak of respiratory illness first detected in December in association with a live animal market in Wuhan, China, has been implicated in almost 2,000 cases and 56 deaths in that country. Cases have been reported in 13 countries besides China. Five cases of 2019-nCoV infection have been confirmed in the United States, all in people recently returned from Wuhan. As the virus spreads in China, however, it’s almost certain more cases will show up in the United States. Travel history is key, Dr. Schaffner and others said.
Plan and rehearse
The first step to prepare is to use the CDC’s Interim Guidance for Healthcare Professionals to make a written plan specific to your practice to respond to a potential case. The plan must include notifying the local health department, the CDC liaison for testing, and tracking down patient contacts.
“It’s not good enough to just download CDC’s guidance; use it to make your own local plan and know what to do 24/7,” said Daniel Lucey, MD, an infectious disease expert at Georgetown University Medical Center, Washington, D.C.
“Know who is on call at the health department on weekends and nights,” he said. Know where the patient is going to be isolated; figure out what to do if there’s more than one, and tests come back positive. Have masks on hand, and rehearse the response. “Make a coronavirus team, and absolutely have the nurses involved,” as well as other providers who may come into contact with a case, he added.
“You want to be able to do as well as your counterparts in Washington state and Chicago,” where the first two U.S. cases emerged. “They were prepared. They knew what to do,” Dr. Lucey said.
Those first two U.S. patients – a man in Everett, Wash., and a Chicago woman – developed symptoms after returning from Wuhan, a city of 11 million just over 400 miles inland from the port city of Shanghai. On Jan. 26 three more cases were confirmed by the CDC, two in California and one in Arizona, and each had recently traveled to Wuhan. All five patients remain hospitalized, and there’s no evidence they spread the infection further. There is also no evidence of human-to-human transmission of other cases exported from China to any other countries, according to the WHO.
WHO declined to declare a global health emergency – a Public Health Emergency of International Concern, in its parlance – on Jan. 23. The step would have triggered travel and trade restrictions in member states, including the United States. For now, at least, the group said it wasn’t warranted at this point.
Fatality rates
The focus right now is China. The outbreak has spread beyond Wuhan to other parts of the country, and there’s evidence of fourth-generation spread.
Transportation into and out of Wuhan and other cities has been curtailed, Lunar New Year festivals have been canceled, and the Shanghai Disneyland has been closed, among other measures taken by Chinese officials.
The government could be taking drastic measures in part to prevent the public criticism it took in the early 2000’s for the delayed response and lack of transparency during the global outbreak of another wildlife market coronavirus epidemic, severe acute respiratory syndrome (SARS). In a press conference Jan. 22, WHO officials commended the government’s containment efforts but did not say they recommended them.
According to WHO, serious cases in China have mostly been in people over 40 years old with significant comorbidities and have skewed towards men. Spread seems to be limited to family members, health care providers, and other close contacts, probably by respiratory droplets. If that pattern holds, WHO officials said, the outbreak is containable.
The fatality rate appears to be around 3%, a good deal lower than the 10% reported for SARS and much lower than the nearly 40% reported for Middle East respiratory syndrome (MERS), another recent coronavirus mutation from the animal trade.
The Wuhan virus fatality rate might drop as milder cases are detected and added to the denominator. “It definitely appears to be less severe than SARS and MERS,” said Amesh Adalja, MD, an infectious disease physician in Pittsburgh and emerging infectious disease researcher at Johns Hopkins University, Baltimore.
SARS: Lessons learned
In general, the world is much better equipped for coronavirus outbreaks than when SARS, in particular, emerged in 2003.
WHO officials in their press conference lauded China for it openness with the current outbreak, and for isolating and sequencing the virus immediately, which gave the world a diagnostic test in the first days of the outbreak, something that wasn’t available for SARS. China and other countries also are cooperating and working closely to contain the Wuhan virus.
“What we know today might change tomorrow, so we have to keep tuned in to new information, but we learned a lot from SARS,” Dr. Shaffner said. Overall, it’s likely “the impact on the United States of this new coronavirus is going to be trivial,” he predicted.
Dr. Lucey, however, recalled that the SARS outbreak in Toronto in 2003 started with one missed case. A woman returned asymptomatic from Hong Kong and spread the infection to her family members before she died. Her cause of death wasn’t immediately recognized, nor was the reason her family members were sick, since they hadn’t been to Hong Kong recently.
The infection ultimately spread to more than 200 people, about half of them health care workers. A few people died.
If a virus is sufficiently contagious, “it just takes one. You don’t want to be the one who misses that first patient,” Dr. Lucey said.
Currently, there are no antivirals or vaccines for coronaviruses; researchers are working on both, but for now, care is supportive.
This article was updated with new case numbers on 1/26/20.
Second U.S. coronavirus patient confirmed
at a Jan. 24, 2020, press briefing.
The first U.S. case, a traveler who entered the United States at Seattle-Tacoma International Airport, was confirmed on Jan. 20.
A Chicago resident returning from Wuhan, China, on Jan. 13, 2020, developed symptoms of the disease and contacted her health care clinician and is currently being treated in isolation at an unnamed hospital, according to Nancy Messonnier, MD, director of the National Center for Immunization and Respiratory Diseases at the CDC. The patient, a woman in her 60s, is in stable condition and remains hospitalized. She was not symptomatic on her flight to Chicago but developed symptoms in the following days after her return from Wuhan. She had limited contacts after her return, and all potential contacts are being tracked.
Dr. Messonnier said the CDC expects more cases in the United States but stressed that, although this is a serious public health threat, the risk to the American public is low. She noted that the situation is evolving rapidly and that the CDC is following the developments hour by hour.
Jennifer Layden, MD, PhD, chief medical officer and state epidemiologist with the Illinois Department of Public Health, said public health preparations made it possible to quickly identify and arrange appropriate hospitalization for this patient. Allison Arwady, MD, Chicago Department of Health commissioner, said the Illinois Department of Health partnered with the CDC to test specimens quickly, which led to the diagnosis in this patient.
So far, 63 U.S. patients have been investigated for possible infection with the 2019-nCoV; 11 so far have tested negative and 2 have tested positive. Testing of the remaining potential cases and others is ongoing.
Currently, samples from patients with suspected 2010-nCoV infections are being sent to the CDC for testing, Dr. Messonnier said. The turnaround for testing is currently 4-6 hours. Respiratory samples and some blood samples are being tested by the CDC labs.
The CDC is developing diagnostic kits for public health authorities in the United States for local testing and will work with the World Health Organization to make these kits available to the international community when possible.
Dr. Messonnier said that, at present, the incubation period for this disease appears to be about 14 days, but she suggested that further study will be required to identify the range of time for contagion. She also said it is premature to compare the 2019-nCoV with previous coronavirus outbreaks, such as severe acute respiratory syndrome (SARS) or Middle East respiratory syndrome (MERS), in terms of contagion or fatality rates.
Meanwhile, Andrew D. Mesecar, PhD, the Walther Professor in Cancer Structural Biology and head of the department of biochemistry at Purdue University, West Lafayette, Ind., said on Jan. 24 in a news release that 2019-nCoV is genetically similar to the SARS variant. “MERS virus and the SARS virus are more different genetically,” noted Dr. Mesecar, whose team received the genome of 2019-nCoV on Jan. 17 and analyzed it the next day. “But the Wuhan virus is genetically almost identical to the SARS virus and, therefore, it is expected to look and act nearly the same. In another week or two, we’ll be able to begin to see if the virus is mutating.”
Dr. Messonnier said that nonessential travel to Wuhan is not recommended. In addition, she said, and all other visitors to China need to take appropriate precautions, such as handwashing and avoiding other individuals with respiratory illness.
Screenings at five U.S. airports will continue. So far, approximately 200 flights and 2,000 travelers have been screened as of Jan. 23. No cases were reported, but one traveler has been identified for further for evaluation. Possible contacts with those suspected of infection have been identified and alerted in 22 states.
The CDC will continue to update the public and will post information on the CDC newsroom website.
at a Jan. 24, 2020, press briefing.
The first U.S. case, a traveler who entered the United States at Seattle-Tacoma International Airport, was confirmed on Jan. 20.
A Chicago resident returning from Wuhan, China, on Jan. 13, 2020, developed symptoms of the disease and contacted her health care clinician and is currently being treated in isolation at an unnamed hospital, according to Nancy Messonnier, MD, director of the National Center for Immunization and Respiratory Diseases at the CDC. The patient, a woman in her 60s, is in stable condition and remains hospitalized. She was not symptomatic on her flight to Chicago but developed symptoms in the following days after her return from Wuhan. She had limited contacts after her return, and all potential contacts are being tracked.
Dr. Messonnier said the CDC expects more cases in the United States but stressed that, although this is a serious public health threat, the risk to the American public is low. She noted that the situation is evolving rapidly and that the CDC is following the developments hour by hour.
Jennifer Layden, MD, PhD, chief medical officer and state epidemiologist with the Illinois Department of Public Health, said public health preparations made it possible to quickly identify and arrange appropriate hospitalization for this patient. Allison Arwady, MD, Chicago Department of Health commissioner, said the Illinois Department of Health partnered with the CDC to test specimens quickly, which led to the diagnosis in this patient.
So far, 63 U.S. patients have been investigated for possible infection with the 2019-nCoV; 11 so far have tested negative and 2 have tested positive. Testing of the remaining potential cases and others is ongoing.
Currently, samples from patients with suspected 2010-nCoV infections are being sent to the CDC for testing, Dr. Messonnier said. The turnaround for testing is currently 4-6 hours. Respiratory samples and some blood samples are being tested by the CDC labs.
The CDC is developing diagnostic kits for public health authorities in the United States for local testing and will work with the World Health Organization to make these kits available to the international community when possible.
Dr. Messonnier said that, at present, the incubation period for this disease appears to be about 14 days, but she suggested that further study will be required to identify the range of time for contagion. She also said it is premature to compare the 2019-nCoV with previous coronavirus outbreaks, such as severe acute respiratory syndrome (SARS) or Middle East respiratory syndrome (MERS), in terms of contagion or fatality rates.
Meanwhile, Andrew D. Mesecar, PhD, the Walther Professor in Cancer Structural Biology and head of the department of biochemistry at Purdue University, West Lafayette, Ind., said on Jan. 24 in a news release that 2019-nCoV is genetically similar to the SARS variant. “MERS virus and the SARS virus are more different genetically,” noted Dr. Mesecar, whose team received the genome of 2019-nCoV on Jan. 17 and analyzed it the next day. “But the Wuhan virus is genetically almost identical to the SARS virus and, therefore, it is expected to look and act nearly the same. In another week or two, we’ll be able to begin to see if the virus is mutating.”
Dr. Messonnier said that nonessential travel to Wuhan is not recommended. In addition, she said, and all other visitors to China need to take appropriate precautions, such as handwashing and avoiding other individuals with respiratory illness.
Screenings at five U.S. airports will continue. So far, approximately 200 flights and 2,000 travelers have been screened as of Jan. 23. No cases were reported, but one traveler has been identified for further for evaluation. Possible contacts with those suspected of infection have been identified and alerted in 22 states.
The CDC will continue to update the public and will post information on the CDC newsroom website.
at a Jan. 24, 2020, press briefing.
The first U.S. case, a traveler who entered the United States at Seattle-Tacoma International Airport, was confirmed on Jan. 20.
A Chicago resident returning from Wuhan, China, on Jan. 13, 2020, developed symptoms of the disease and contacted her health care clinician and is currently being treated in isolation at an unnamed hospital, according to Nancy Messonnier, MD, director of the National Center for Immunization and Respiratory Diseases at the CDC. The patient, a woman in her 60s, is in stable condition and remains hospitalized. She was not symptomatic on her flight to Chicago but developed symptoms in the following days after her return from Wuhan. She had limited contacts after her return, and all potential contacts are being tracked.
Dr. Messonnier said the CDC expects more cases in the United States but stressed that, although this is a serious public health threat, the risk to the American public is low. She noted that the situation is evolving rapidly and that the CDC is following the developments hour by hour.
Jennifer Layden, MD, PhD, chief medical officer and state epidemiologist with the Illinois Department of Public Health, said public health preparations made it possible to quickly identify and arrange appropriate hospitalization for this patient. Allison Arwady, MD, Chicago Department of Health commissioner, said the Illinois Department of Health partnered with the CDC to test specimens quickly, which led to the diagnosis in this patient.
So far, 63 U.S. patients have been investigated for possible infection with the 2019-nCoV; 11 so far have tested negative and 2 have tested positive. Testing of the remaining potential cases and others is ongoing.
Currently, samples from patients with suspected 2010-nCoV infections are being sent to the CDC for testing, Dr. Messonnier said. The turnaround for testing is currently 4-6 hours. Respiratory samples and some blood samples are being tested by the CDC labs.
The CDC is developing diagnostic kits for public health authorities in the United States for local testing and will work with the World Health Organization to make these kits available to the international community when possible.
Dr. Messonnier said that, at present, the incubation period for this disease appears to be about 14 days, but she suggested that further study will be required to identify the range of time for contagion. She also said it is premature to compare the 2019-nCoV with previous coronavirus outbreaks, such as severe acute respiratory syndrome (SARS) or Middle East respiratory syndrome (MERS), in terms of contagion or fatality rates.
Meanwhile, Andrew D. Mesecar, PhD, the Walther Professor in Cancer Structural Biology and head of the department of biochemistry at Purdue University, West Lafayette, Ind., said on Jan. 24 in a news release that 2019-nCoV is genetically similar to the SARS variant. “MERS virus and the SARS virus are more different genetically,” noted Dr. Mesecar, whose team received the genome of 2019-nCoV on Jan. 17 and analyzed it the next day. “But the Wuhan virus is genetically almost identical to the SARS virus and, therefore, it is expected to look and act nearly the same. In another week or two, we’ll be able to begin to see if the virus is mutating.”
Dr. Messonnier said that nonessential travel to Wuhan is not recommended. In addition, she said, and all other visitors to China need to take appropriate precautions, such as handwashing and avoiding other individuals with respiratory illness.
Screenings at five U.S. airports will continue. So far, approximately 200 flights and 2,000 travelers have been screened as of Jan. 23. No cases were reported, but one traveler has been identified for further for evaluation. Possible contacts with those suspected of infection have been identified and alerted in 22 states.
The CDC will continue to update the public and will post information on the CDC newsroom website.
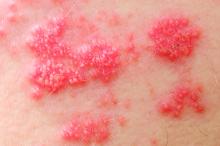
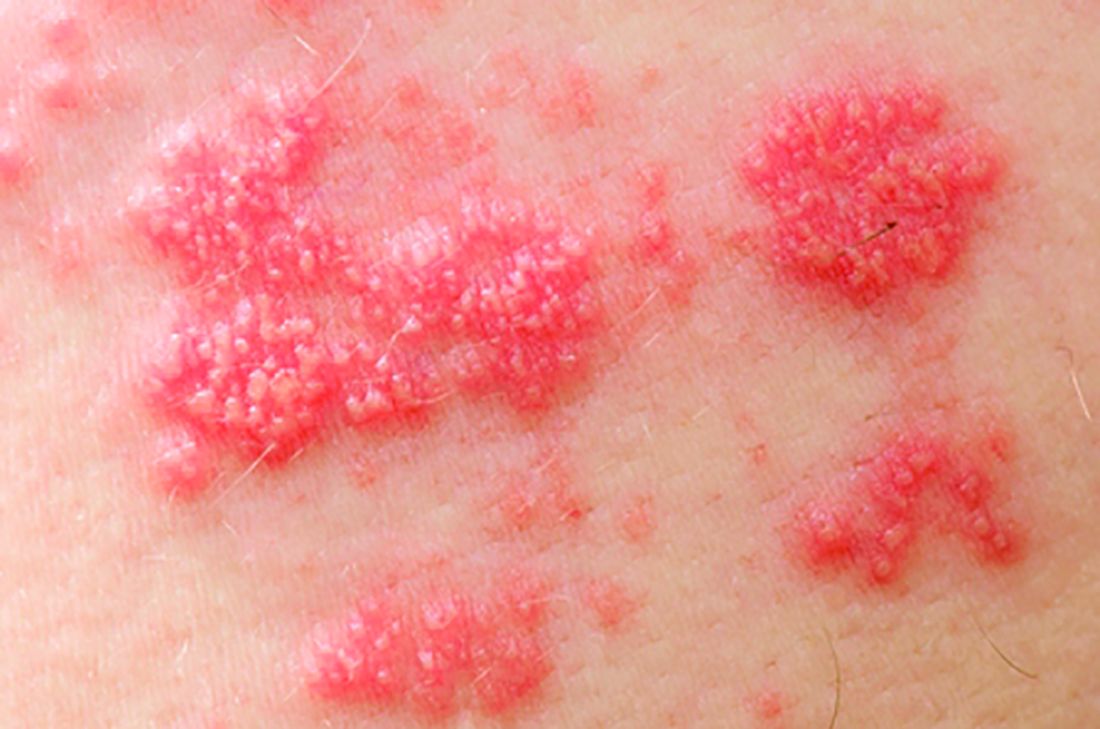
Exogenous boosting against shingles not as robust as thought
Exposure to children with chickenpox reduces the incidence of shingles in adults 33% over 2 years, and 27% out to 20 years, according to British investigators.
Being exposed to children with illness due to varicella infection acts as an “exogenous booster” in adults who had chickenpox themselves as children, making shingles less likely, they explained in a BMJ article.
Although that’s good news, it’s been reported previously that exposure to children with chickenpox confers complete protection against shingles in adults for years afterward.
The finding matters in the United Kingdom because varicella vaccine is not part of the pediatric immunization schedule. The United States is the only country that mandates two shots as a requirement for children to attend school.
The United Kingdom, however, is reconsidering its policy. In the past, the exogenous booster idea has been one of the arguments used against mandating the vaccine for children; the concern is that preventing chickenpox in children – and subsequent reexposure to herpes zoster in adults – would kick off a costly wave of shingles in adults.
The study results “are themselves unable to justify for or against specific vaccination schedules, but they do suggest that revised mathematical models are required to estimate the impact of varicella vaccination, with the updated assumption that exogenous boosting is incomplete and only reduces the risk of zoster by about 30%,” noted the investigators, led by Harriet Forbes of the London School of Hygiene and Tropical Medicine.
The researchers identified 9,604 adults with a shingles diagnosis during 1997-2018 who at some point lived with a child who had chickenpox. Data came from the U.K. Clinical Practice Research Datalink, a general practice database.
They then looked at the incidence of shingles within 20 years of exposure to the sick child and compared it with the incidence before exposure and after 20 years, by which time the exogenous booster is thought to wear off. It was a self-controlled case series analysis, “a relatively novel epidemiological study design where individuals act as their own controls. Comparisons are made within individuals rather than between individuals as in a cohort or case control study,” Ms. Forbes and colleagues explained.
After adjustment for age, calendar time, and season, they found that in the 2 years after household exposure to a child with varicella, adults were 33% less likely to develop zoster (incidence ratio 0.67, 95% confidence interval 0.62-0.73), and 27% less likely from 10 to 20 years (IR 0.73, CI 0.62-0.87). The boosting effect appeared to be stronger in men.
“Exogenous boosting provides some protection from the risk of herpes zoster, but not complete immunity, as assumed by previous cost effectiveness estimates of varicella immunization,” the researchers said.
More than two-thirds of the adults with shingles were women, which fits with previous reports. Median age of exposure to a child with varicella was 38 years.
Ms. Forbes and colleagues noted that “the study design required patients with zoster to be living with a child with varicella, therefore the study cohort is younger than a general population with zoster. ... However, when we restricted our analysis to adults aged 50 and older at exposure to varicella, a similar pattern of association was observed, with no evidence of effect modification by age. This suggests that although the median age of our study cohort ... was low, the findings can be generalized to older people.”
There was no external funding for the work, and the lead investigator had no relevant financial disclosures. One investigator reported research grants from GSK and Merck, both makers of chickenpox and shingles vaccines.
SOURCE: Forbes H et al. BMJ. 2020 Jan 22;368:l6987.
Exposure to children with chickenpox reduces the incidence of shingles in adults 33% over 2 years, and 27% out to 20 years, according to British investigators.
Being exposed to children with illness due to varicella infection acts as an “exogenous booster” in adults who had chickenpox themselves as children, making shingles less likely, they explained in a BMJ article.
Although that’s good news, it’s been reported previously that exposure to children with chickenpox confers complete protection against shingles in adults for years afterward.
The finding matters in the United Kingdom because varicella vaccine is not part of the pediatric immunization schedule. The United States is the only country that mandates two shots as a requirement for children to attend school.
The United Kingdom, however, is reconsidering its policy. In the past, the exogenous booster idea has been one of the arguments used against mandating the vaccine for children; the concern is that preventing chickenpox in children – and subsequent reexposure to herpes zoster in adults – would kick off a costly wave of shingles in adults.
The study results “are themselves unable to justify for or against specific vaccination schedules, but they do suggest that revised mathematical models are required to estimate the impact of varicella vaccination, with the updated assumption that exogenous boosting is incomplete and only reduces the risk of zoster by about 30%,” noted the investigators, led by Harriet Forbes of the London School of Hygiene and Tropical Medicine.
The researchers identified 9,604 adults with a shingles diagnosis during 1997-2018 who at some point lived with a child who had chickenpox. Data came from the U.K. Clinical Practice Research Datalink, a general practice database.
They then looked at the incidence of shingles within 20 years of exposure to the sick child and compared it with the incidence before exposure and after 20 years, by which time the exogenous booster is thought to wear off. It was a self-controlled case series analysis, “a relatively novel epidemiological study design where individuals act as their own controls. Comparisons are made within individuals rather than between individuals as in a cohort or case control study,” Ms. Forbes and colleagues explained.
After adjustment for age, calendar time, and season, they found that in the 2 years after household exposure to a child with varicella, adults were 33% less likely to develop zoster (incidence ratio 0.67, 95% confidence interval 0.62-0.73), and 27% less likely from 10 to 20 years (IR 0.73, CI 0.62-0.87). The boosting effect appeared to be stronger in men.
“Exogenous boosting provides some protection from the risk of herpes zoster, but not complete immunity, as assumed by previous cost effectiveness estimates of varicella immunization,” the researchers said.
More than two-thirds of the adults with shingles were women, which fits with previous reports. Median age of exposure to a child with varicella was 38 years.
Ms. Forbes and colleagues noted that “the study design required patients with zoster to be living with a child with varicella, therefore the study cohort is younger than a general population with zoster. ... However, when we restricted our analysis to adults aged 50 and older at exposure to varicella, a similar pattern of association was observed, with no evidence of effect modification by age. This suggests that although the median age of our study cohort ... was low, the findings can be generalized to older people.”
There was no external funding for the work, and the lead investigator had no relevant financial disclosures. One investigator reported research grants from GSK and Merck, both makers of chickenpox and shingles vaccines.
SOURCE: Forbes H et al. BMJ. 2020 Jan 22;368:l6987.
Exposure to children with chickenpox reduces the incidence of shingles in adults 33% over 2 years, and 27% out to 20 years, according to British investigators.
Being exposed to children with illness due to varicella infection acts as an “exogenous booster” in adults who had chickenpox themselves as children, making shingles less likely, they explained in a BMJ article.
Although that’s good news, it’s been reported previously that exposure to children with chickenpox confers complete protection against shingles in adults for years afterward.
The finding matters in the United Kingdom because varicella vaccine is not part of the pediatric immunization schedule. The United States is the only country that mandates two shots as a requirement for children to attend school.
The United Kingdom, however, is reconsidering its policy. In the past, the exogenous booster idea has been one of the arguments used against mandating the vaccine for children; the concern is that preventing chickenpox in children – and subsequent reexposure to herpes zoster in adults – would kick off a costly wave of shingles in adults.
The study results “are themselves unable to justify for or against specific vaccination schedules, but they do suggest that revised mathematical models are required to estimate the impact of varicella vaccination, with the updated assumption that exogenous boosting is incomplete and only reduces the risk of zoster by about 30%,” noted the investigators, led by Harriet Forbes of the London School of Hygiene and Tropical Medicine.
The researchers identified 9,604 adults with a shingles diagnosis during 1997-2018 who at some point lived with a child who had chickenpox. Data came from the U.K. Clinical Practice Research Datalink, a general practice database.
They then looked at the incidence of shingles within 20 years of exposure to the sick child and compared it with the incidence before exposure and after 20 years, by which time the exogenous booster is thought to wear off. It was a self-controlled case series analysis, “a relatively novel epidemiological study design where individuals act as their own controls. Comparisons are made within individuals rather than between individuals as in a cohort or case control study,” Ms. Forbes and colleagues explained.
After adjustment for age, calendar time, and season, they found that in the 2 years after household exposure to a child with varicella, adults were 33% less likely to develop zoster (incidence ratio 0.67, 95% confidence interval 0.62-0.73), and 27% less likely from 10 to 20 years (IR 0.73, CI 0.62-0.87). The boosting effect appeared to be stronger in men.
“Exogenous boosting provides some protection from the risk of herpes zoster, but not complete immunity, as assumed by previous cost effectiveness estimates of varicella immunization,” the researchers said.
More than two-thirds of the adults with shingles were women, which fits with previous reports. Median age of exposure to a child with varicella was 38 years.
Ms. Forbes and colleagues noted that “the study design required patients with zoster to be living with a child with varicella, therefore the study cohort is younger than a general population with zoster. ... However, when we restricted our analysis to adults aged 50 and older at exposure to varicella, a similar pattern of association was observed, with no evidence of effect modification by age. This suggests that although the median age of our study cohort ... was low, the findings can be generalized to older people.”
There was no external funding for the work, and the lead investigator had no relevant financial disclosures. One investigator reported research grants from GSK and Merck, both makers of chickenpox and shingles vaccines.
SOURCE: Forbes H et al. BMJ. 2020 Jan 22;368:l6987.
FROM BMJ
Washington state patient is first U.S. case of novel coronavirus
The first case of the novel coronavirus, named 2019-nCoV, in the United States has been diagnosed in a traveler from China who came through Seattle-Tacoma International Airport on Jan 15, the Centers for Disease Control and Prevention announced today at a press briefing.
The outbreak began at a animal and meat market in China and now has spread to at least three other countries, including Thailand, Japan and South Korea. While originally thought to be spreading from animal to person, it appears that limited person-to-person transmission is occurring, although it is currently unknown how easily this virus spreads between people.
More than 300 cases have been reported and six deaths have occurred. Fourteen health care workers have been infected.
Scott Lindquist, MD, MPH, Washington state epidemiologist, said at the briefing that the patient, a man who had been in Wuhan, arrived at Sea-Tac on Jan. 15, 2 days before airport screening had been initiated. He was symptom free at the time of his arrival and probably would not have been identified as infected with 2019-nCoV. The patient had been aware of the public health and news media coverage of 2019-nCoV and, after developing symptoms, contacted his health care provider on Jan. 19. The patient did not fly directly from Wuhan, but Dr. Lindquist said that he has been fully cooperative and has been helpful to authorities in tracing his route and contacts. The man is being treated at Providence Regional Medical Center, Everett, Wash.
The CDC obtained a specimen from the patient immediately and identified the 2019-nCoV within 24 hours.
Screening at airports is part of a multipart strategy to address this type of infection that includes public health information dissemination, patient education, as well as hospital preparation and training exercises. Currently, a strategy referred to as “funneling” is being implemented wherein travelers from China are rerouted and reticketed to one of the five airports conducting screening. At present, JFK in New York, San Francisco International, Los Angeles International, Hartsfield-Jackson Atlanta International Airport, and Chicago O’Hare International Airport are conducting inbound traveler screening.
The CDC is working in close cooperation with the Department of Homeland Security and the Federal Aviation Administration to coordinate travel screenings and reroutings. In addition, the CDC is working with the World Health Organization and the international global health community to share information about this outbreak. The CDC also has staff on site in Wuhan and is communicating with local health authorities. The CDC has activated its Emergency Operations Center to better provide ongoing support to the 2019-nCoV response. Currently, the focus is on tracing contacts and the means of transmission of this virus.
Updates on the outbreak will be posted on the CDC coronavirus website.
CORRECTION: 1/21/2020: The name of the medical center where the 2019-nCoV patient is being treated was corrected.
The first case of the novel coronavirus, named 2019-nCoV, in the United States has been diagnosed in a traveler from China who came through Seattle-Tacoma International Airport on Jan 15, the Centers for Disease Control and Prevention announced today at a press briefing.
The outbreak began at a animal and meat market in China and now has spread to at least three other countries, including Thailand, Japan and South Korea. While originally thought to be spreading from animal to person, it appears that limited person-to-person transmission is occurring, although it is currently unknown how easily this virus spreads between people.
More than 300 cases have been reported and six deaths have occurred. Fourteen health care workers have been infected.
Scott Lindquist, MD, MPH, Washington state epidemiologist, said at the briefing that the patient, a man who had been in Wuhan, arrived at Sea-Tac on Jan. 15, 2 days before airport screening had been initiated. He was symptom free at the time of his arrival and probably would not have been identified as infected with 2019-nCoV. The patient had been aware of the public health and news media coverage of 2019-nCoV and, after developing symptoms, contacted his health care provider on Jan. 19. The patient did not fly directly from Wuhan, but Dr. Lindquist said that he has been fully cooperative and has been helpful to authorities in tracing his route and contacts. The man is being treated at Providence Regional Medical Center, Everett, Wash.
The CDC obtained a specimen from the patient immediately and identified the 2019-nCoV within 24 hours.
Screening at airports is part of a multipart strategy to address this type of infection that includes public health information dissemination, patient education, as well as hospital preparation and training exercises. Currently, a strategy referred to as “funneling” is being implemented wherein travelers from China are rerouted and reticketed to one of the five airports conducting screening. At present, JFK in New York, San Francisco International, Los Angeles International, Hartsfield-Jackson Atlanta International Airport, and Chicago O’Hare International Airport are conducting inbound traveler screening.
The CDC is working in close cooperation with the Department of Homeland Security and the Federal Aviation Administration to coordinate travel screenings and reroutings. In addition, the CDC is working with the World Health Organization and the international global health community to share information about this outbreak. The CDC also has staff on site in Wuhan and is communicating with local health authorities. The CDC has activated its Emergency Operations Center to better provide ongoing support to the 2019-nCoV response. Currently, the focus is on tracing contacts and the means of transmission of this virus.
Updates on the outbreak will be posted on the CDC coronavirus website.
CORRECTION: 1/21/2020: The name of the medical center where the 2019-nCoV patient is being treated was corrected.
The first case of the novel coronavirus, named 2019-nCoV, in the United States has been diagnosed in a traveler from China who came through Seattle-Tacoma International Airport on Jan 15, the Centers for Disease Control and Prevention announced today at a press briefing.
The outbreak began at a animal and meat market in China and now has spread to at least three other countries, including Thailand, Japan and South Korea. While originally thought to be spreading from animal to person, it appears that limited person-to-person transmission is occurring, although it is currently unknown how easily this virus spreads between people.
More than 300 cases have been reported and six deaths have occurred. Fourteen health care workers have been infected.
Scott Lindquist, MD, MPH, Washington state epidemiologist, said at the briefing that the patient, a man who had been in Wuhan, arrived at Sea-Tac on Jan. 15, 2 days before airport screening had been initiated. He was symptom free at the time of his arrival and probably would not have been identified as infected with 2019-nCoV. The patient had been aware of the public health and news media coverage of 2019-nCoV and, after developing symptoms, contacted his health care provider on Jan. 19. The patient did not fly directly from Wuhan, but Dr. Lindquist said that he has been fully cooperative and has been helpful to authorities in tracing his route and contacts. The man is being treated at Providence Regional Medical Center, Everett, Wash.
The CDC obtained a specimen from the patient immediately and identified the 2019-nCoV within 24 hours.
Screening at airports is part of a multipart strategy to address this type of infection that includes public health information dissemination, patient education, as well as hospital preparation and training exercises. Currently, a strategy referred to as “funneling” is being implemented wherein travelers from China are rerouted and reticketed to one of the five airports conducting screening. At present, JFK in New York, San Francisco International, Los Angeles International, Hartsfield-Jackson Atlanta International Airport, and Chicago O’Hare International Airport are conducting inbound traveler screening.
The CDC is working in close cooperation with the Department of Homeland Security and the Federal Aviation Administration to coordinate travel screenings and reroutings. In addition, the CDC is working with the World Health Organization and the international global health community to share information about this outbreak. The CDC also has staff on site in Wuhan and is communicating with local health authorities. The CDC has activated its Emergency Operations Center to better provide ongoing support to the 2019-nCoV response. Currently, the focus is on tracing contacts and the means of transmission of this virus.
Updates on the outbreak will be posted on the CDC coronavirus website.
CORRECTION: 1/21/2020: The name of the medical center where the 2019-nCoV patient is being treated was corrected.
REPORTING FROM CDC
Nontuberculous mycobacterial lung disease cases on the rise across U.S.
To assess the NTM lung disease burden on a national level, Kevin L. Winthrop, MD, of Oregon Health & Science University, Portland, and associates analyzed patient data from a U.S. managed care claims database between 2008 and 2015. Their findings were published in the Annals of the American Thoracic Society.
A case of NTM lung disease was defined as a patient with at least two medical claims with the disease’s diagnostic codes – 031.0 and A31.0 – that were at least 30 days apart. Of the 74,984,596 beneficiaries in the database, 9,476 met the case definition for NTM lung disease; 69% (n = 6,530) were women.
From 2008 to 2015, the annual incidence of NTM lung disease increased from 3.13 (95% confidence interval, 2.88-3.40) to 4.73 (95% CI, 4.43-5.05) per 100,000 person-years, with the average rate of yearly change being +5.2% (95% CI, 4.0%-6.4%; P less than .01).The annual prevalence increased from 6.78 (95% CI, 6.45-7.14) to 11.70 (95% CI, 11.26-12.16) per 100,000 persons, with the average rate of yearly change being +7.5% (95% CI, 6.7-8.2%; P less than .01).
The majority of NTM lung disease in the United States is caused by Mycobacterium avium complex (17), although other species such as M. abscessus, M. kansasii, M. xenopi, and others contribute to this disease burden.
“It’s a classic chicken-or-egg scenario,” said Sachin Gupta, MD, a pulmonologist in San Francisco, in regard to the rising numbers. “Increased awareness of NTM lung disease is, in part, why we’re seeing prevalence and incidence go up. And yet the disease itself may also be growing in clusters and pockets, as the data show, in various places across the nation.
“The worrisome aspect here,” he added, “is that future studies will likely show that, as incidence is increasing, mortality is increasing as well. That speaks to the challenges with these bugs: Very hard to diagnose, very hard to treat.”
The authors acknowledged their study’s limitations, including the lack of microbiologic or radiographic confirmation of the NTM infection and the inherent shortcomings of claims data–based studies overall. They did note a previous report, however, that “claims-based case identification has a high positive predictive value of approximately 82% for NTM lung disease.”
The study was funded by Insmed; the Intramural Research Programs of the National Institute of Allergy and Infectious Diseases; and the National Heart, Lung, and Blood Institute. The authors reported no conflicts of interest.
SOURCE: Winthrop KL et al. Ann Am Thorac Soc. 2019 Dec 13. doi: 10.1513/AnnalsATS.201804-236OC.
To assess the NTM lung disease burden on a national level, Kevin L. Winthrop, MD, of Oregon Health & Science University, Portland, and associates analyzed patient data from a U.S. managed care claims database between 2008 and 2015. Their findings were published in the Annals of the American Thoracic Society.
A case of NTM lung disease was defined as a patient with at least two medical claims with the disease’s diagnostic codes – 031.0 and A31.0 – that were at least 30 days apart. Of the 74,984,596 beneficiaries in the database, 9,476 met the case definition for NTM lung disease; 69% (n = 6,530) were women.
From 2008 to 2015, the annual incidence of NTM lung disease increased from 3.13 (95% confidence interval, 2.88-3.40) to 4.73 (95% CI, 4.43-5.05) per 100,000 person-years, with the average rate of yearly change being +5.2% (95% CI, 4.0%-6.4%; P less than .01).The annual prevalence increased from 6.78 (95% CI, 6.45-7.14) to 11.70 (95% CI, 11.26-12.16) per 100,000 persons, with the average rate of yearly change being +7.5% (95% CI, 6.7-8.2%; P less than .01).
The majority of NTM lung disease in the United States is caused by Mycobacterium avium complex (17), although other species such as M. abscessus, M. kansasii, M. xenopi, and others contribute to this disease burden.
“It’s a classic chicken-or-egg scenario,” said Sachin Gupta, MD, a pulmonologist in San Francisco, in regard to the rising numbers. “Increased awareness of NTM lung disease is, in part, why we’re seeing prevalence and incidence go up. And yet the disease itself may also be growing in clusters and pockets, as the data show, in various places across the nation.
“The worrisome aspect here,” he added, “is that future studies will likely show that, as incidence is increasing, mortality is increasing as well. That speaks to the challenges with these bugs: Very hard to diagnose, very hard to treat.”
The authors acknowledged their study’s limitations, including the lack of microbiologic or radiographic confirmation of the NTM infection and the inherent shortcomings of claims data–based studies overall. They did note a previous report, however, that “claims-based case identification has a high positive predictive value of approximately 82% for NTM lung disease.”
The study was funded by Insmed; the Intramural Research Programs of the National Institute of Allergy and Infectious Diseases; and the National Heart, Lung, and Blood Institute. The authors reported no conflicts of interest.
SOURCE: Winthrop KL et al. Ann Am Thorac Soc. 2019 Dec 13. doi: 10.1513/AnnalsATS.201804-236OC.
To assess the NTM lung disease burden on a national level, Kevin L. Winthrop, MD, of Oregon Health & Science University, Portland, and associates analyzed patient data from a U.S. managed care claims database between 2008 and 2015. Their findings were published in the Annals of the American Thoracic Society.
A case of NTM lung disease was defined as a patient with at least two medical claims with the disease’s diagnostic codes – 031.0 and A31.0 – that were at least 30 days apart. Of the 74,984,596 beneficiaries in the database, 9,476 met the case definition for NTM lung disease; 69% (n = 6,530) were women.
From 2008 to 2015, the annual incidence of NTM lung disease increased from 3.13 (95% confidence interval, 2.88-3.40) to 4.73 (95% CI, 4.43-5.05) per 100,000 person-years, with the average rate of yearly change being +5.2% (95% CI, 4.0%-6.4%; P less than .01).The annual prevalence increased from 6.78 (95% CI, 6.45-7.14) to 11.70 (95% CI, 11.26-12.16) per 100,000 persons, with the average rate of yearly change being +7.5% (95% CI, 6.7-8.2%; P less than .01).
The majority of NTM lung disease in the United States is caused by Mycobacterium avium complex (17), although other species such as M. abscessus, M. kansasii, M. xenopi, and others contribute to this disease burden.
“It’s a classic chicken-or-egg scenario,” said Sachin Gupta, MD, a pulmonologist in San Francisco, in regard to the rising numbers. “Increased awareness of NTM lung disease is, in part, why we’re seeing prevalence and incidence go up. And yet the disease itself may also be growing in clusters and pockets, as the data show, in various places across the nation.
“The worrisome aspect here,” he added, “is that future studies will likely show that, as incidence is increasing, mortality is increasing as well. That speaks to the challenges with these bugs: Very hard to diagnose, very hard to treat.”
The authors acknowledged their study’s limitations, including the lack of microbiologic or radiographic confirmation of the NTM infection and the inherent shortcomings of claims data–based studies overall. They did note a previous report, however, that “claims-based case identification has a high positive predictive value of approximately 82% for NTM lung disease.”
The study was funded by Insmed; the Intramural Research Programs of the National Institute of Allergy and Infectious Diseases; and the National Heart, Lung, and Blood Institute. The authors reported no conflicts of interest.
SOURCE: Winthrop KL et al. Ann Am Thorac Soc. 2019 Dec 13. doi: 10.1513/AnnalsATS.201804-236OC.
FROM ANNALS OF THE AMERICAN THORACIC SOCIETY