User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Ranking seven COVID-19 antigen tests by ease of use: Report
Some COVID-19 rapid antigen home test kits are much easier to use than others, according to an analysis by ECRI, an independent, nonprofit patient safety organization.
None of the tests were rated as “excellent” in terms of usability and some had “noteworthy” usability concerns, the company said.
If a test is hard to use, “chances are that you may miss a step or not follow the right order, or contaminate the testing area and that can definitely influence the accuracy of the test and lead to a wrong test result,” Marcus Schabacker, MD, PhD, president and CEO of ECRI, told this news organization.
To gauge usability, ECRI used the “industry-standard” system usability scale (SUS), which rates products on a scale of 0 to 100 with 100 being the easiest to use.
More than 30 points separated the top and bottom tests analyzed. The top performer was On/Go, followed by CareStart and Flowflex.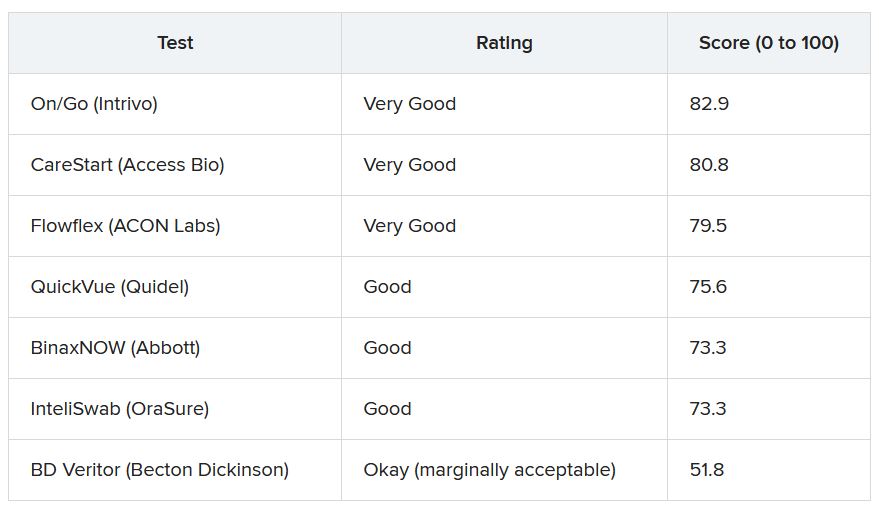
ECRI analysts found that some tests require particularly fine motor skills or have instructions with extremely small font size that may make it hard for older adults or people with complex health conditions to use the tests correctly.
“If you have a tremor from Parkinson’s, for example, or anything which won’t allow you to handle small items, you will have difficulties to do that test by yourself. That is the No. 1 concern we have,” Dr. Schabacker said.
“The second concern is readability, as all of these tests have relatively small instructions. One of them actually has doesn’t even have instructions – you have to download an app,” he noted.
Given demand and supply issues, Dr. Schabacker acknowledged that consumers might not have a choice in which test to use and may have to rely on whatever is available.
These tests are a “hot commodity right now,” he said. “If you have a choice, people should use the ones which are easiest to use, which is the On/Go, the CareStart, or the Flowflex.”
A version of this article first appeared on Medscape.com.
Some COVID-19 rapid antigen home test kits are much easier to use than others, according to an analysis by ECRI, an independent, nonprofit patient safety organization.
None of the tests were rated as “excellent” in terms of usability and some had “noteworthy” usability concerns, the company said.
If a test is hard to use, “chances are that you may miss a step or not follow the right order, or contaminate the testing area and that can definitely influence the accuracy of the test and lead to a wrong test result,” Marcus Schabacker, MD, PhD, president and CEO of ECRI, told this news organization.
To gauge usability, ECRI used the “industry-standard” system usability scale (SUS), which rates products on a scale of 0 to 100 with 100 being the easiest to use.
More than 30 points separated the top and bottom tests analyzed. The top performer was On/Go, followed by CareStart and Flowflex.
ECRI analysts found that some tests require particularly fine motor skills or have instructions with extremely small font size that may make it hard for older adults or people with complex health conditions to use the tests correctly.
“If you have a tremor from Parkinson’s, for example, or anything which won’t allow you to handle small items, you will have difficulties to do that test by yourself. That is the No. 1 concern we have,” Dr. Schabacker said.
“The second concern is readability, as all of these tests have relatively small instructions. One of them actually has doesn’t even have instructions – you have to download an app,” he noted.
Given demand and supply issues, Dr. Schabacker acknowledged that consumers might not have a choice in which test to use and may have to rely on whatever is available.
These tests are a “hot commodity right now,” he said. “If you have a choice, people should use the ones which are easiest to use, which is the On/Go, the CareStart, or the Flowflex.”
A version of this article first appeared on Medscape.com.
Some COVID-19 rapid antigen home test kits are much easier to use than others, according to an analysis by ECRI, an independent, nonprofit patient safety organization.
None of the tests were rated as “excellent” in terms of usability and some had “noteworthy” usability concerns, the company said.
If a test is hard to use, “chances are that you may miss a step or not follow the right order, or contaminate the testing area and that can definitely influence the accuracy of the test and lead to a wrong test result,” Marcus Schabacker, MD, PhD, president and CEO of ECRI, told this news organization.
To gauge usability, ECRI used the “industry-standard” system usability scale (SUS), which rates products on a scale of 0 to 100 with 100 being the easiest to use.
More than 30 points separated the top and bottom tests analyzed. The top performer was On/Go, followed by CareStart and Flowflex.
ECRI analysts found that some tests require particularly fine motor skills or have instructions with extremely small font size that may make it hard for older adults or people with complex health conditions to use the tests correctly.
“If you have a tremor from Parkinson’s, for example, or anything which won’t allow you to handle small items, you will have difficulties to do that test by yourself. That is the No. 1 concern we have,” Dr. Schabacker said.
“The second concern is readability, as all of these tests have relatively small instructions. One of them actually has doesn’t even have instructions – you have to download an app,” he noted.
Given demand and supply issues, Dr. Schabacker acknowledged that consumers might not have a choice in which test to use and may have to rely on whatever is available.
These tests are a “hot commodity right now,” he said. “If you have a choice, people should use the ones which are easiest to use, which is the On/Go, the CareStart, or the Flowflex.”
A version of this article first appeared on Medscape.com.
Physicians react: Should docs lose their licenses for spreading false COVID information?
Doctors providing “fraudulent” COVID-19 information became a hot-button issue for physicians responding to Medscape’s recent article, "Shouldn’t Doctors Who Spread False COVID-19 Information Lose Their Licenses?”
COVID-19 safety recommendations are set by mainstream medical organizations as new information becomes available, but some doctors consistently oppose advice from the Centers for Disease Control and Prevention and other medical authorities. These physicians often promote off-label, unapproved use of medications for COVID-19 and/or contradict mainstream safety guidelines such as vaccines, masks, and social distancing.
Some medical organizations are concerned that these doctors are hampering efforts to control the highly contagious coronavirus and are, at worst, placing lives in danger with their contrarian views that can spread like wildfire on social media sites. Their words are often used by those who refuse to be vaccinated or wear masks.
State licensing boards have mostly refused to discipline these doctors for making false and/or misleading claims, but as the virus spreads, there are calls to take action against them. However, others worry that such actions would violate free speech and critical thought.
Yes, those doctors are doing wrong
Several physicians took a strong stand against their fellow doctors who are spreading misinformation about COVID-19.
One doctor endorsed the idea of removing licenses for spreading misinformation and called for criminal prosecution: “It should certainly be grounds for cancellation of all licensing (after appropriate examination to rule out acute psychotic episodes, dementia, tumor, etc.) and very likely [include] a charge of manslaughter.”
Another health care provider said, “A person who does not accept science should not, of course, be allowed to practice medicine. One who argues publicly that vaccines and masks don’t work should be prosecuted for crimes ranging from reckless endangerment to attempted murder.”
One reader framed COVID-19 misinformers in stark terms: “These men and women are medical prostitutes. Their medical and surgical colleges [should] have a panel to track in-court testimony and the disinformation they spread ...”
“This is malpractice of the worst kind,” said a clinician. “Public health officials and science are quite clear on [the] best practices for safety during a pandemic, which is killing millions. This is a standard of care.”
“Medical Boards should suspend licenses and give the physician a chance to testify [about] the scientific basis for his comments,” added a health care provider. “Boards involve themselves in all kinds of perceived disciplinary infractions. We are in the midst of a lethal pandemic. I would think that would take precedence over many other issues?”
“I do believe that physicians have the responsibility to speak the truth and have scientifically displayed minds,” said a reader. “Not [to] promulgate misleading, false, and/or unverified information.”
“Any physician, who holds a license, should abide [by] government and state regulation,” asserted a doctor. “He should be disciplined by the board for spreading medical/public misinformation since he is creating potential harm to the population.”
One specialist insisted that “state boards do not do enough to restrict/limit the practice of physicians touting questionable therapies.”
“Any doctor who spreads false information about Covid is hurting our country, our individuals, and our economy and leading to needless deaths,” asserted a physician. “However, there are uncertainties, and where those exist, physicians [should] simply say ‘it is unknown.’”
No, those physicians have a right to speak their beliefs
However, many physicians worried that science and controversial thought were being muzzled.
“Absolutely no,” a doctor stated. “Who judges what is misinformation in this age where debate is canceled? Science advances with challenge, and it’s not about an authority dictating the allowable opinion.”
Another clinician claimed the “truth is very difficult to discern from less-than-truth in a country running on a profit-oriented economic ideology.”
One specialist warned that if disinformation doctors are held responsible, then “that means a lot of doctors” will be “gone” because “almost anything that is written or said about COVID can be contested.”
Another physician warned his colleagues about suppressing new ideas: “To condemn what we didn’t try, or purposefully ignore a different approach because [it] doesn’t agree with our opinion is suppression of information.”
Some doctors insisted the issue extended beyond medicine and into Constitutional freedoms. They also expressed their mistrust in the government to regulate physicians.
“There is a First Amendment in this country,” said one reader. “What you think is false may not be so. The people can listen to whoever they want to and make their own medical decisions. We do not need one iota more of politicizing medicine. Having an MD or DO does not mean you relinquish your First Amendment rights.”
“One of the fundamental problems with a system that allows government to ‘license’ physicians, or any other profession, is that politics inevitably turn to cronyism, and big businesses and wealthy people start controlling the government,” argued a doctor.
One clinician suggested enforcement against health food, drug company commercials, and talk shows: “What about all the [misinformation] at the health food stores and the like. Doctors of natural-whatever? Those info-commercials on tv. How many faxes do I get to ‘approve’ because ‘patients request’ braces and pain-treating expensive compounds advertised on TV? We tolerate those ... What about Dr. Oz and the docs on talk shows claiming BS?”
And the debate goes even further
Some physicians questioned the very notion of claiming “truth.”
“Nobody should be certain that they have the ‘absolute truth,’” said one reader. “In fact, the best clinical insights exceed so-called knowledge by at least one step.”
“Who can determine exactly what is truth?” asked another clinician. “For sure, the ‘Federal Government,’ who ‘is here to help you,’ is not qualified to make such determinations, and who are you to make such a suggestion as to remove someone’s license because they disagree with you? Give me a break!”
Another physician echoed that sentiment: “What’s true and false is often and certainly currently debatable. There are well-qualified physicians (with credentials such as the development of mRNA technology), virologists, and biostatisticians that have valid thoughts on this but do not necessarily agree with the drug company-sponsored journals and news channels (most of them). Their voices should be heard, and they should not lose their licenses. They are doing their work in good conscience.”
One reader commented that he wanted his “freedom of speech,” and offered this defiant advice: “You can take this license and shove it.”
Finally, a physician noted that the political climate has influenced medical directives: “If someone in a leadership role knowingly, and with intent, spread false information, that is wrong. However, during this global pandemic the active and the politics have combined. Red state no mandate, blue state mandate – what does that tell you about American leadership?”
A version of this article first appeared on Medscape.com.
Doctors providing “fraudulent” COVID-19 information became a hot-button issue for physicians responding to Medscape’s recent article, "Shouldn’t Doctors Who Spread False COVID-19 Information Lose Their Licenses?”
COVID-19 safety recommendations are set by mainstream medical organizations as new information becomes available, but some doctors consistently oppose advice from the Centers for Disease Control and Prevention and other medical authorities. These physicians often promote off-label, unapproved use of medications for COVID-19 and/or contradict mainstream safety guidelines such as vaccines, masks, and social distancing.
Some medical organizations are concerned that these doctors are hampering efforts to control the highly contagious coronavirus and are, at worst, placing lives in danger with their contrarian views that can spread like wildfire on social media sites. Their words are often used by those who refuse to be vaccinated or wear masks.
State licensing boards have mostly refused to discipline these doctors for making false and/or misleading claims, but as the virus spreads, there are calls to take action against them. However, others worry that such actions would violate free speech and critical thought.
Yes, those doctors are doing wrong
Several physicians took a strong stand against their fellow doctors who are spreading misinformation about COVID-19.
One doctor endorsed the idea of removing licenses for spreading misinformation and called for criminal prosecution: “It should certainly be grounds for cancellation of all licensing (after appropriate examination to rule out acute psychotic episodes, dementia, tumor, etc.) and very likely [include] a charge of manslaughter.”
Another health care provider said, “A person who does not accept science should not, of course, be allowed to practice medicine. One who argues publicly that vaccines and masks don’t work should be prosecuted for crimes ranging from reckless endangerment to attempted murder.”
One reader framed COVID-19 misinformers in stark terms: “These men and women are medical prostitutes. Their medical and surgical colleges [should] have a panel to track in-court testimony and the disinformation they spread ...”
“This is malpractice of the worst kind,” said a clinician. “Public health officials and science are quite clear on [the] best practices for safety during a pandemic, which is killing millions. This is a standard of care.”
“Medical Boards should suspend licenses and give the physician a chance to testify [about] the scientific basis for his comments,” added a health care provider. “Boards involve themselves in all kinds of perceived disciplinary infractions. We are in the midst of a lethal pandemic. I would think that would take precedence over many other issues?”
“I do believe that physicians have the responsibility to speak the truth and have scientifically displayed minds,” said a reader. “Not [to] promulgate misleading, false, and/or unverified information.”
“Any physician, who holds a license, should abide [by] government and state regulation,” asserted a doctor. “He should be disciplined by the board for spreading medical/public misinformation since he is creating potential harm to the population.”
One specialist insisted that “state boards do not do enough to restrict/limit the practice of physicians touting questionable therapies.”
“Any doctor who spreads false information about Covid is hurting our country, our individuals, and our economy and leading to needless deaths,” asserted a physician. “However, there are uncertainties, and where those exist, physicians [should] simply say ‘it is unknown.’”
No, those physicians have a right to speak their beliefs
However, many physicians worried that science and controversial thought were being muzzled.
“Absolutely no,” a doctor stated. “Who judges what is misinformation in this age where debate is canceled? Science advances with challenge, and it’s not about an authority dictating the allowable opinion.”
Another clinician claimed the “truth is very difficult to discern from less-than-truth in a country running on a profit-oriented economic ideology.”
One specialist warned that if disinformation doctors are held responsible, then “that means a lot of doctors” will be “gone” because “almost anything that is written or said about COVID can be contested.”
Another physician warned his colleagues about suppressing new ideas: “To condemn what we didn’t try, or purposefully ignore a different approach because [it] doesn’t agree with our opinion is suppression of information.”
Some doctors insisted the issue extended beyond medicine and into Constitutional freedoms. They also expressed their mistrust in the government to regulate physicians.
“There is a First Amendment in this country,” said one reader. “What you think is false may not be so. The people can listen to whoever they want to and make their own medical decisions. We do not need one iota more of politicizing medicine. Having an MD or DO does not mean you relinquish your First Amendment rights.”
“One of the fundamental problems with a system that allows government to ‘license’ physicians, or any other profession, is that politics inevitably turn to cronyism, and big businesses and wealthy people start controlling the government,” argued a doctor.
One clinician suggested enforcement against health food, drug company commercials, and talk shows: “What about all the [misinformation] at the health food stores and the like. Doctors of natural-whatever? Those info-commercials on tv. How many faxes do I get to ‘approve’ because ‘patients request’ braces and pain-treating expensive compounds advertised on TV? We tolerate those ... What about Dr. Oz and the docs on talk shows claiming BS?”
And the debate goes even further
Some physicians questioned the very notion of claiming “truth.”
“Nobody should be certain that they have the ‘absolute truth,’” said one reader. “In fact, the best clinical insights exceed so-called knowledge by at least one step.”
“Who can determine exactly what is truth?” asked another clinician. “For sure, the ‘Federal Government,’ who ‘is here to help you,’ is not qualified to make such determinations, and who are you to make such a suggestion as to remove someone’s license because they disagree with you? Give me a break!”
Another physician echoed that sentiment: “What’s true and false is often and certainly currently debatable. There are well-qualified physicians (with credentials such as the development of mRNA technology), virologists, and biostatisticians that have valid thoughts on this but do not necessarily agree with the drug company-sponsored journals and news channels (most of them). Their voices should be heard, and they should not lose their licenses. They are doing their work in good conscience.”
One reader commented that he wanted his “freedom of speech,” and offered this defiant advice: “You can take this license and shove it.”
Finally, a physician noted that the political climate has influenced medical directives: “If someone in a leadership role knowingly, and with intent, spread false information, that is wrong. However, during this global pandemic the active and the politics have combined. Red state no mandate, blue state mandate – what does that tell you about American leadership?”
A version of this article first appeared on Medscape.com.
Doctors providing “fraudulent” COVID-19 information became a hot-button issue for physicians responding to Medscape’s recent article, "Shouldn’t Doctors Who Spread False COVID-19 Information Lose Their Licenses?”
COVID-19 safety recommendations are set by mainstream medical organizations as new information becomes available, but some doctors consistently oppose advice from the Centers for Disease Control and Prevention and other medical authorities. These physicians often promote off-label, unapproved use of medications for COVID-19 and/or contradict mainstream safety guidelines such as vaccines, masks, and social distancing.
Some medical organizations are concerned that these doctors are hampering efforts to control the highly contagious coronavirus and are, at worst, placing lives in danger with their contrarian views that can spread like wildfire on social media sites. Their words are often used by those who refuse to be vaccinated or wear masks.
State licensing boards have mostly refused to discipline these doctors for making false and/or misleading claims, but as the virus spreads, there are calls to take action against them. However, others worry that such actions would violate free speech and critical thought.
Yes, those doctors are doing wrong
Several physicians took a strong stand against their fellow doctors who are spreading misinformation about COVID-19.
One doctor endorsed the idea of removing licenses for spreading misinformation and called for criminal prosecution: “It should certainly be grounds for cancellation of all licensing (after appropriate examination to rule out acute psychotic episodes, dementia, tumor, etc.) and very likely [include] a charge of manslaughter.”
Another health care provider said, “A person who does not accept science should not, of course, be allowed to practice medicine. One who argues publicly that vaccines and masks don’t work should be prosecuted for crimes ranging from reckless endangerment to attempted murder.”
One reader framed COVID-19 misinformers in stark terms: “These men and women are medical prostitutes. Their medical and surgical colleges [should] have a panel to track in-court testimony and the disinformation they spread ...”
“This is malpractice of the worst kind,” said a clinician. “Public health officials and science are quite clear on [the] best practices for safety during a pandemic, which is killing millions. This is a standard of care.”
“Medical Boards should suspend licenses and give the physician a chance to testify [about] the scientific basis for his comments,” added a health care provider. “Boards involve themselves in all kinds of perceived disciplinary infractions. We are in the midst of a lethal pandemic. I would think that would take precedence over many other issues?”
“I do believe that physicians have the responsibility to speak the truth and have scientifically displayed minds,” said a reader. “Not [to] promulgate misleading, false, and/or unverified information.”
“Any physician, who holds a license, should abide [by] government and state regulation,” asserted a doctor. “He should be disciplined by the board for spreading medical/public misinformation since he is creating potential harm to the population.”
One specialist insisted that “state boards do not do enough to restrict/limit the practice of physicians touting questionable therapies.”
“Any doctor who spreads false information about Covid is hurting our country, our individuals, and our economy and leading to needless deaths,” asserted a physician. “However, there are uncertainties, and where those exist, physicians [should] simply say ‘it is unknown.’”
No, those physicians have a right to speak their beliefs
However, many physicians worried that science and controversial thought were being muzzled.
“Absolutely no,” a doctor stated. “Who judges what is misinformation in this age where debate is canceled? Science advances with challenge, and it’s not about an authority dictating the allowable opinion.”
Another clinician claimed the “truth is very difficult to discern from less-than-truth in a country running on a profit-oriented economic ideology.”
One specialist warned that if disinformation doctors are held responsible, then “that means a lot of doctors” will be “gone” because “almost anything that is written or said about COVID can be contested.”
Another physician warned his colleagues about suppressing new ideas: “To condemn what we didn’t try, or purposefully ignore a different approach because [it] doesn’t agree with our opinion is suppression of information.”
Some doctors insisted the issue extended beyond medicine and into Constitutional freedoms. They also expressed their mistrust in the government to regulate physicians.
“There is a First Amendment in this country,” said one reader. “What you think is false may not be so. The people can listen to whoever they want to and make their own medical decisions. We do not need one iota more of politicizing medicine. Having an MD or DO does not mean you relinquish your First Amendment rights.”
“One of the fundamental problems with a system that allows government to ‘license’ physicians, or any other profession, is that politics inevitably turn to cronyism, and big businesses and wealthy people start controlling the government,” argued a doctor.
One clinician suggested enforcement against health food, drug company commercials, and talk shows: “What about all the [misinformation] at the health food stores and the like. Doctors of natural-whatever? Those info-commercials on tv. How many faxes do I get to ‘approve’ because ‘patients request’ braces and pain-treating expensive compounds advertised on TV? We tolerate those ... What about Dr. Oz and the docs on talk shows claiming BS?”
And the debate goes even further
Some physicians questioned the very notion of claiming “truth.”
“Nobody should be certain that they have the ‘absolute truth,’” said one reader. “In fact, the best clinical insights exceed so-called knowledge by at least one step.”
“Who can determine exactly what is truth?” asked another clinician. “For sure, the ‘Federal Government,’ who ‘is here to help you,’ is not qualified to make such determinations, and who are you to make such a suggestion as to remove someone’s license because they disagree with you? Give me a break!”
Another physician echoed that sentiment: “What’s true and false is often and certainly currently debatable. There are well-qualified physicians (with credentials such as the development of mRNA technology), virologists, and biostatisticians that have valid thoughts on this but do not necessarily agree with the drug company-sponsored journals and news channels (most of them). Their voices should be heard, and they should not lose their licenses. They are doing their work in good conscience.”
One reader commented that he wanted his “freedom of speech,” and offered this defiant advice: “You can take this license and shove it.”
Finally, a physician noted that the political climate has influenced medical directives: “If someone in a leadership role knowingly, and with intent, spread false information, that is wrong. However, during this global pandemic the active and the politics have combined. Red state no mandate, blue state mandate – what does that tell you about American leadership?”
A version of this article first appeared on Medscape.com.
The etiology of acute otitis media in young children in recent years
Since the COVID-19 pandemic began, pediatricians have been seeing fewer cases of all respiratory illnesses, including acute otitis media (AOM). However, as I prepare this column, an uptick has commenced and likely will continue in an upward trajectory as we emerge from the pandemic into an endemic coronavirus era. Our group in Rochester, N.Y., has continued prospective studies of AOM throughout the pandemic. We found that nasopharyngeal colonization by Streptococcus pneumoniae (pneumococcus), Haemophilus influenzae, and Moraxella catarrhalis remained prevalent in our study cohort of children aged 6-36 months. However, with all the precautions of masking, social distancing, hand washing, and quick exclusion from day care when illness occurred, the frequency of detecting these common otopathogens decreased, as one might expect.1
Leading up to the pandemic, we had an abundance of data to characterize AOM etiology and found that the cause of AOM continues to change following the introduction of the 13-valent pneumococcal conjugate vaccine (PCV13, Prevnar 13). Our most recent report on otopathogen distribution and antibiotic susceptibility covered the years 2015-2019.2 A total of 589 children were enrolled prospectively and we collected 495 middle ear fluid samples (MEF) from 319 AOM cases using tympanocentesis. The frequency of isolates was H. influenzae (34%), pneumococcus (24%), and M. catarrhalis (15%). Beta-lactamase–positive H. influenzae strains were identified among 49% of the isolates, rendering them resistant to amoxicillin. PCV13 serotypes were infrequently isolated. However, we did isolate vaccine types (VTs) in some children from MEF, notably serotypes 19F, 19A, and 3. Non-PCV13 pneumococcus serotypes 35B, 23B, and 15B/C emerged as the most common serotypes. Amoxicillin resistance was identified among 25% of pneumococcal strains. Out of 16 antibiotics tested, 9 (56%) showed a significant increase in nonsusceptibility among pneumococcal isolates. 100% of M. catarrhalis isolates were beta-lactamase producers and therefore resistant to amoxicillin.
PCV13 has resulted in a decline in both invasive and noninvasive pneumococcal infections caused by strains expressing the 13 capsular serotypes included in the vaccine. However, the emergence of replacement serotypes occurred after introduction of PCV73,4 and continues to occur during the PCV13 era, as shown from the results presented here. Non-PCV13 serotypes accounted for more than 90% of MEF isolates during 2015-2019, with 35B, 21 and 23B being the most commonly isolated. Other emergent serotypes of potential importance were nonvaccine serotypes 15A, 15B, 15C, 23A and 11A. This is highly relevant because forthcoming higher-valency PCVs – PCV15 (manufactured by Merck) and PCV20 (manufactured by Pfizer) will not include many of the dominant capsular serotypes of pneumococcus strains causing AOM. Consequently, the impact of higher-valency PCVs on AOM will not be as great as was observed with the introduction of PCV7 or PCV13.
Of special interest, 22% of pneumococcus isolates from MEF were serotype 35B, making it the most prevalent. Recently we reported a significant rise in antibiotic nonsusceptibility in Spn isolates, contributed mainly by serotype 35B5 and we have been studying how 35B strains transitioned from commensal to otopathogen in children.6 Because serotype 35B strains are increasingly prevalent and often antibiotic resistant, absence of this serotype from PCV15 and PCV20 is cause for concern.
The frequency of isolation of H. influenzae and M. catarrhalis has remained stable across the PCV13 era as the No. 1 and No. 3 pathogens. Similarly, the production of beta-lactamase among strains causing AOM has remained stable at close to 50% and 100%, respectively. Use of amoxicillin, either high dose or standard dose, would not be expected to kill these bacteria.
Our study design has limitations. The population is derived from a predominantly middle-class, suburban population of children in upstate New York and may not be representative of other types of populations in the United States. The children are 6-36 months old, the age when most AOM occurs. MEF samples that were culture negative for bacteria were not further tested by polymerase chain reaction methods.
Dr. Pichichero is a specialist in pediatric infectious diseases, Center for Infectious Diseases and Immunology, and director of the Research Institute, at Rochester (N.Y.) General Hospital. He has no conflicts of interest to declare.
References
1. Kaur R et al. Front Pediatr. 2021;9:722483.
2. Kaur R et al. Euro J Clin Microbiol Infect Dis. 2021;41:37-44
3. Pelton SI et al. Pediatr Infect Disease J. 2004;23:1015-22.
4. Farrell DJ et al. Pediatr Infect Disease J. 2007;26:123-8..
5. Kaur R et al. Clin Infect Dis 2021;72(5):797-805.
6. Fuji N et al. Front Cell Infect Microbiol. 2021;11:744742.
Since the COVID-19 pandemic began, pediatricians have been seeing fewer cases of all respiratory illnesses, including acute otitis media (AOM). However, as I prepare this column, an uptick has commenced and likely will continue in an upward trajectory as we emerge from the pandemic into an endemic coronavirus era. Our group in Rochester, N.Y., has continued prospective studies of AOM throughout the pandemic. We found that nasopharyngeal colonization by Streptococcus pneumoniae (pneumococcus), Haemophilus influenzae, and Moraxella catarrhalis remained prevalent in our study cohort of children aged 6-36 months. However, with all the precautions of masking, social distancing, hand washing, and quick exclusion from day care when illness occurred, the frequency of detecting these common otopathogens decreased, as one might expect.1
Leading up to the pandemic, we had an abundance of data to characterize AOM etiology and found that the cause of AOM continues to change following the introduction of the 13-valent pneumococcal conjugate vaccine (PCV13, Prevnar 13). Our most recent report on otopathogen distribution and antibiotic susceptibility covered the years 2015-2019.2 A total of 589 children were enrolled prospectively and we collected 495 middle ear fluid samples (MEF) from 319 AOM cases using tympanocentesis. The frequency of isolates was H. influenzae (34%), pneumococcus (24%), and M. catarrhalis (15%). Beta-lactamase–positive H. influenzae strains were identified among 49% of the isolates, rendering them resistant to amoxicillin. PCV13 serotypes were infrequently isolated. However, we did isolate vaccine types (VTs) in some children from MEF, notably serotypes 19F, 19A, and 3. Non-PCV13 pneumococcus serotypes 35B, 23B, and 15B/C emerged as the most common serotypes. Amoxicillin resistance was identified among 25% of pneumococcal strains. Out of 16 antibiotics tested, 9 (56%) showed a significant increase in nonsusceptibility among pneumococcal isolates. 100% of M. catarrhalis isolates were beta-lactamase producers and therefore resistant to amoxicillin.
PCV13 has resulted in a decline in both invasive and noninvasive pneumococcal infections caused by strains expressing the 13 capsular serotypes included in the vaccine. However, the emergence of replacement serotypes occurred after introduction of PCV73,4 and continues to occur during the PCV13 era, as shown from the results presented here. Non-PCV13 serotypes accounted for more than 90% of MEF isolates during 2015-2019, with 35B, 21 and 23B being the most commonly isolated. Other emergent serotypes of potential importance were nonvaccine serotypes 15A, 15B, 15C, 23A and 11A. This is highly relevant because forthcoming higher-valency PCVs – PCV15 (manufactured by Merck) and PCV20 (manufactured by Pfizer) will not include many of the dominant capsular serotypes of pneumococcus strains causing AOM. Consequently, the impact of higher-valency PCVs on AOM will not be as great as was observed with the introduction of PCV7 or PCV13.
Of special interest, 22% of pneumococcus isolates from MEF were serotype 35B, making it the most prevalent. Recently we reported a significant rise in antibiotic nonsusceptibility in Spn isolates, contributed mainly by serotype 35B5 and we have been studying how 35B strains transitioned from commensal to otopathogen in children.6 Because serotype 35B strains are increasingly prevalent and often antibiotic resistant, absence of this serotype from PCV15 and PCV20 is cause for concern.
The frequency of isolation of H. influenzae and M. catarrhalis has remained stable across the PCV13 era as the No. 1 and No. 3 pathogens. Similarly, the production of beta-lactamase among strains causing AOM has remained stable at close to 50% and 100%, respectively. Use of amoxicillin, either high dose or standard dose, would not be expected to kill these bacteria.
Our study design has limitations. The population is derived from a predominantly middle-class, suburban population of children in upstate New York and may not be representative of other types of populations in the United States. The children are 6-36 months old, the age when most AOM occurs. MEF samples that were culture negative for bacteria were not further tested by polymerase chain reaction methods.
Dr. Pichichero is a specialist in pediatric infectious diseases, Center for Infectious Diseases and Immunology, and director of the Research Institute, at Rochester (N.Y.) General Hospital. He has no conflicts of interest to declare.
References
1. Kaur R et al. Front Pediatr. 2021;9:722483.
2. Kaur R et al. Euro J Clin Microbiol Infect Dis. 2021;41:37-44
3. Pelton SI et al. Pediatr Infect Disease J. 2004;23:1015-22.
4. Farrell DJ et al. Pediatr Infect Disease J. 2007;26:123-8..
5. Kaur R et al. Clin Infect Dis 2021;72(5):797-805.
6. Fuji N et al. Front Cell Infect Microbiol. 2021;11:744742.
Since the COVID-19 pandemic began, pediatricians have been seeing fewer cases of all respiratory illnesses, including acute otitis media (AOM). However, as I prepare this column, an uptick has commenced and likely will continue in an upward trajectory as we emerge from the pandemic into an endemic coronavirus era. Our group in Rochester, N.Y., has continued prospective studies of AOM throughout the pandemic. We found that nasopharyngeal colonization by Streptococcus pneumoniae (pneumococcus), Haemophilus influenzae, and Moraxella catarrhalis remained prevalent in our study cohort of children aged 6-36 months. However, with all the precautions of masking, social distancing, hand washing, and quick exclusion from day care when illness occurred, the frequency of detecting these common otopathogens decreased, as one might expect.1
Leading up to the pandemic, we had an abundance of data to characterize AOM etiology and found that the cause of AOM continues to change following the introduction of the 13-valent pneumococcal conjugate vaccine (PCV13, Prevnar 13). Our most recent report on otopathogen distribution and antibiotic susceptibility covered the years 2015-2019.2 A total of 589 children were enrolled prospectively and we collected 495 middle ear fluid samples (MEF) from 319 AOM cases using tympanocentesis. The frequency of isolates was H. influenzae (34%), pneumococcus (24%), and M. catarrhalis (15%). Beta-lactamase–positive H. influenzae strains were identified among 49% of the isolates, rendering them resistant to amoxicillin. PCV13 serotypes were infrequently isolated. However, we did isolate vaccine types (VTs) in some children from MEF, notably serotypes 19F, 19A, and 3. Non-PCV13 pneumococcus serotypes 35B, 23B, and 15B/C emerged as the most common serotypes. Amoxicillin resistance was identified among 25% of pneumococcal strains. Out of 16 antibiotics tested, 9 (56%) showed a significant increase in nonsusceptibility among pneumococcal isolates. 100% of M. catarrhalis isolates were beta-lactamase producers and therefore resistant to amoxicillin.
PCV13 has resulted in a decline in both invasive and noninvasive pneumococcal infections caused by strains expressing the 13 capsular serotypes included in the vaccine. However, the emergence of replacement serotypes occurred after introduction of PCV73,4 and continues to occur during the PCV13 era, as shown from the results presented here. Non-PCV13 serotypes accounted for more than 90% of MEF isolates during 2015-2019, with 35B, 21 and 23B being the most commonly isolated. Other emergent serotypes of potential importance were nonvaccine serotypes 15A, 15B, 15C, 23A and 11A. This is highly relevant because forthcoming higher-valency PCVs – PCV15 (manufactured by Merck) and PCV20 (manufactured by Pfizer) will not include many of the dominant capsular serotypes of pneumococcus strains causing AOM. Consequently, the impact of higher-valency PCVs on AOM will not be as great as was observed with the introduction of PCV7 or PCV13.
Of special interest, 22% of pneumococcus isolates from MEF were serotype 35B, making it the most prevalent. Recently we reported a significant rise in antibiotic nonsusceptibility in Spn isolates, contributed mainly by serotype 35B5 and we have been studying how 35B strains transitioned from commensal to otopathogen in children.6 Because serotype 35B strains are increasingly prevalent and often antibiotic resistant, absence of this serotype from PCV15 and PCV20 is cause for concern.
The frequency of isolation of H. influenzae and M. catarrhalis has remained stable across the PCV13 era as the No. 1 and No. 3 pathogens. Similarly, the production of beta-lactamase among strains causing AOM has remained stable at close to 50% and 100%, respectively. Use of amoxicillin, either high dose or standard dose, would not be expected to kill these bacteria.
Our study design has limitations. The population is derived from a predominantly middle-class, suburban population of children in upstate New York and may not be representative of other types of populations in the United States. The children are 6-36 months old, the age when most AOM occurs. MEF samples that were culture negative for bacteria were not further tested by polymerase chain reaction methods.
Dr. Pichichero is a specialist in pediatric infectious diseases, Center for Infectious Diseases and Immunology, and director of the Research Institute, at Rochester (N.Y.) General Hospital. He has no conflicts of interest to declare.
References
1. Kaur R et al. Front Pediatr. 2021;9:722483.
2. Kaur R et al. Euro J Clin Microbiol Infect Dis. 2021;41:37-44
3. Pelton SI et al. Pediatr Infect Disease J. 2004;23:1015-22.
4. Farrell DJ et al. Pediatr Infect Disease J. 2007;26:123-8..
5. Kaur R et al. Clin Infect Dis 2021;72(5):797-805.
6. Fuji N et al. Front Cell Infect Microbiol. 2021;11:744742.
Children and COVID: New cases and hospital admissions skyrocket
, according to the American Academy of Pediatrics and the Children’s Hospital Association.
The total for the week of Dec. 31 to Jan. 6 – the highest since the pandemic began – was an increase of 78% over the previous week (325,000) and 192% higher than just 2 weeks before (199,000), the AAP and CHA said in their weekly COVID-19 report. No region of the country was spared, as all four saw at least 50,000 more cases than the week before, but the increase was largest in the West and smallest in the Midwest.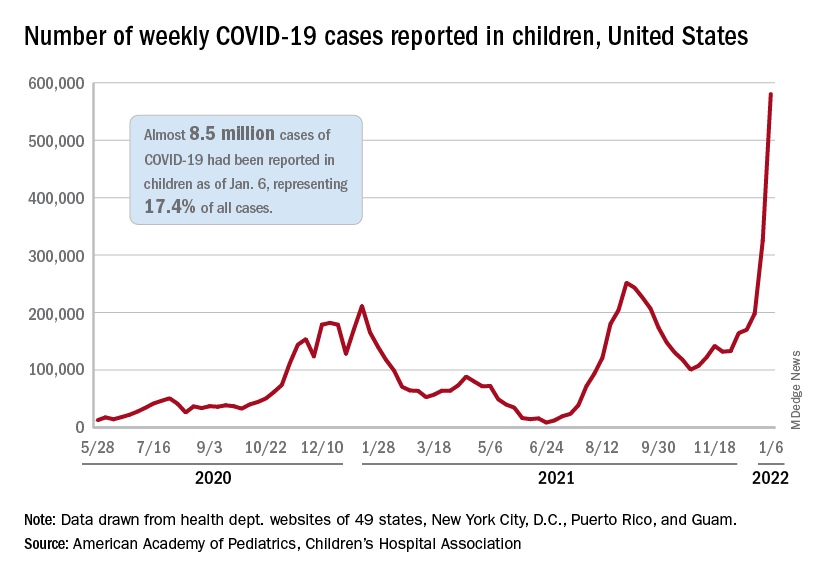
“Nearly 8.5 million children have tested positive for COVID-19 since the onset of the pandemic; nearly 11% of these cases have been added in the past 2 weeks,” the AAP said.
The situation is the same for hospitalizations. On Dec. 15, the daily rate of new admissions for children aged 0-17 years was 0.26 per 100,000, and by Jan. 7 it had more than quadrupled to 1.15 per 100,000, the Centers for Disease Control and Prevention reported. Before Omicron, the highest rate was 0.47 per 100,000 on Sept. 4, 2021.
The number of children occupying inpatient beds who had laboratory-confirmed COVID-19 went from 2,343 on Jan. 2 to 3,476 on Jan. 9, a jump of more than 48% in just 1 week. Texas had more hospitalized children (392) than any other state on Jan. 9, with California (339) and New York (313) the only other states over 300, according to data from the Department of Health & Human Services.
For vaccinations. however, the situation is definitely not the same. The number of children added to the ranks of those with at least one dose of COVID-19 vaccine was down in early 2022 (Jan. 3-9) for both 5- to 11-year-olds (–8.2%) and 16- to 17-year-olds (–12.2%) but higher among those aged 12-15 (12.2%), compared with the previous week (Dec. 27 to Jan. 2), the CDC said on its COVID Data Tracker.
Cumulative figures show that 26.3% of all children aged 5-11 had received at least one dose of vaccine and 17.2% were fully vaccinated as of Jan. 10, compared with 62.2% and 52.0% of 12- to 15-year-olds and 68.5% and 58.1% of those aged 16-17. Altogether, over 23.8 million children in those three age groups have received at least one dose and almost 18.6 million are fully vaccinated, the CDC said.
, according to the American Academy of Pediatrics and the Children’s Hospital Association.
The total for the week of Dec. 31 to Jan. 6 – the highest since the pandemic began – was an increase of 78% over the previous week (325,000) and 192% higher than just 2 weeks before (199,000), the AAP and CHA said in their weekly COVID-19 report. No region of the country was spared, as all four saw at least 50,000 more cases than the week before, but the increase was largest in the West and smallest in the Midwest.
“Nearly 8.5 million children have tested positive for COVID-19 since the onset of the pandemic; nearly 11% of these cases have been added in the past 2 weeks,” the AAP said.
The situation is the same for hospitalizations. On Dec. 15, the daily rate of new admissions for children aged 0-17 years was 0.26 per 100,000, and by Jan. 7 it had more than quadrupled to 1.15 per 100,000, the Centers for Disease Control and Prevention reported. Before Omicron, the highest rate was 0.47 per 100,000 on Sept. 4, 2021.
The number of children occupying inpatient beds who had laboratory-confirmed COVID-19 went from 2,343 on Jan. 2 to 3,476 on Jan. 9, a jump of more than 48% in just 1 week. Texas had more hospitalized children (392) than any other state on Jan. 9, with California (339) and New York (313) the only other states over 300, according to data from the Department of Health & Human Services.
For vaccinations. however, the situation is definitely not the same. The number of children added to the ranks of those with at least one dose of COVID-19 vaccine was down in early 2022 (Jan. 3-9) for both 5- to 11-year-olds (–8.2%) and 16- to 17-year-olds (–12.2%) but higher among those aged 12-15 (12.2%), compared with the previous week (Dec. 27 to Jan. 2), the CDC said on its COVID Data Tracker.
Cumulative figures show that 26.3% of all children aged 5-11 had received at least one dose of vaccine and 17.2% were fully vaccinated as of Jan. 10, compared with 62.2% and 52.0% of 12- to 15-year-olds and 68.5% and 58.1% of those aged 16-17. Altogether, over 23.8 million children in those three age groups have received at least one dose and almost 18.6 million are fully vaccinated, the CDC said.
, according to the American Academy of Pediatrics and the Children’s Hospital Association.
The total for the week of Dec. 31 to Jan. 6 – the highest since the pandemic began – was an increase of 78% over the previous week (325,000) and 192% higher than just 2 weeks before (199,000), the AAP and CHA said in their weekly COVID-19 report. No region of the country was spared, as all four saw at least 50,000 more cases than the week before, but the increase was largest in the West and smallest in the Midwest.
“Nearly 8.5 million children have tested positive for COVID-19 since the onset of the pandemic; nearly 11% of these cases have been added in the past 2 weeks,” the AAP said.
The situation is the same for hospitalizations. On Dec. 15, the daily rate of new admissions for children aged 0-17 years was 0.26 per 100,000, and by Jan. 7 it had more than quadrupled to 1.15 per 100,000, the Centers for Disease Control and Prevention reported. Before Omicron, the highest rate was 0.47 per 100,000 on Sept. 4, 2021.
The number of children occupying inpatient beds who had laboratory-confirmed COVID-19 went from 2,343 on Jan. 2 to 3,476 on Jan. 9, a jump of more than 48% in just 1 week. Texas had more hospitalized children (392) than any other state on Jan. 9, with California (339) and New York (313) the only other states over 300, according to data from the Department of Health & Human Services.
For vaccinations. however, the situation is definitely not the same. The number of children added to the ranks of those with at least one dose of COVID-19 vaccine was down in early 2022 (Jan. 3-9) for both 5- to 11-year-olds (–8.2%) and 16- to 17-year-olds (–12.2%) but higher among those aged 12-15 (12.2%), compared with the previous week (Dec. 27 to Jan. 2), the CDC said on its COVID Data Tracker.
Cumulative figures show that 26.3% of all children aged 5-11 had received at least one dose of vaccine and 17.2% were fully vaccinated as of Jan. 10, compared with 62.2% and 52.0% of 12- to 15-year-olds and 68.5% and 58.1% of those aged 16-17. Altogether, over 23.8 million children in those three age groups have received at least one dose and almost 18.6 million are fully vaccinated, the CDC said.
U.S. reports record-breaking 1.35 million new COVID cases in a day
The United States reported 1.35 million new COVID-19 cases on Jan. 10, logging the highest daily total for any country in the world during the pandemic.
The United States set the previous record of 1 million cases on Jan. 3. (A large number of cases are reported on Mondays, since many states don’t provide updates over the weekend, according to Reuters.)
Still, the 7-day average for new cases has surpassed 700,000, tripling in 2 weeks as the contagious Omicron variant continues to spread across the country.
The daily record of new cases came a day after the United States crossed the grim milestone of 60 million COVID-19 cases during the pandemic, according to the latest data from Johns Hopkins University. More than 11 million new cases were reported in the past 28 days, with 5 million reported since Jan. 2.
Globally, more than 310 million cases have been reported, resulting in nearly 5.5 million COVID-19 deaths. Almost 40 million cases have been confirmed worldwide during the past month, with the United States accounting for 28% of those.
Texas became the second state to report more than 5 million cases since the pandemic began, behind California’s total of 6 million cases. Florida has reported more than 4.6 million, while New York has reported more than 4.1 million.
The United States has also hit an all-time high for hospitalizations, with nearly 146,000 COVID-19 patients in hospitals across the country, according to the latest data from the U.S. Department of Health and Human Services. The previous record was 142,000 hospitalizations in January 2021.
Jan. 11’s hospitalizations are more than twice as many as 2 weeks ago, according to CNN. About 78% of inpatient beds are in use nationwide, and 21% are being used for COVID-19 patients.
Deaths are averaging about 1,700 per day, Reuters reported, which is up from 1,400 in recent days but not much higher than earlier this winter. The peak average was 3,400 daily deaths in mid-January 2021.
The surging numbers of cases and hospitalizations across the country are straining hospitals. On Jan. 10, Virginia Gov. Ralph Northam declared a state of emergency after the number of intensive care unit hospitalizations more than doubled since Dec. 1, CNN reported. The order allows hospitals to expand bed capacity, use telehealth options, and be more flexible with staffing.
Texas is hiring at least 2,700 medical staff to help with the surge, CNN reported, and Kentucky has mobilized the National Guard to provide support.
“Omicron continues to burn through the commonwealth, growing at levels we have never seen before. Omicron is significantly more contagious than even the Delta variant,” Kentucky Gov. Andy Beshear said during a news briefing Jan. 10.
Kentucky reported its highest weekly total of cases last week and has its highest rate of positive tests, at 26%. Mr. Beshear said the state is down to 134 available adult ICU beds.
“If it spreads at the rate we are seeing, it is certainly going to fill up our hospitals,” he said.
A version of this article first appeared on WebMD.com.
The United States reported 1.35 million new COVID-19 cases on Jan. 10, logging the highest daily total for any country in the world during the pandemic.
The United States set the previous record of 1 million cases on Jan. 3. (A large number of cases are reported on Mondays, since many states don’t provide updates over the weekend, according to Reuters.)
Still, the 7-day average for new cases has surpassed 700,000, tripling in 2 weeks as the contagious Omicron variant continues to spread across the country.
The daily record of new cases came a day after the United States crossed the grim milestone of 60 million COVID-19 cases during the pandemic, according to the latest data from Johns Hopkins University. More than 11 million new cases were reported in the past 28 days, with 5 million reported since Jan. 2.
Globally, more than 310 million cases have been reported, resulting in nearly 5.5 million COVID-19 deaths. Almost 40 million cases have been confirmed worldwide during the past month, with the United States accounting for 28% of those.
Texas became the second state to report more than 5 million cases since the pandemic began, behind California’s total of 6 million cases. Florida has reported more than 4.6 million, while New York has reported more than 4.1 million.
The United States has also hit an all-time high for hospitalizations, with nearly 146,000 COVID-19 patients in hospitals across the country, according to the latest data from the U.S. Department of Health and Human Services. The previous record was 142,000 hospitalizations in January 2021.
Jan. 11’s hospitalizations are more than twice as many as 2 weeks ago, according to CNN. About 78% of inpatient beds are in use nationwide, and 21% are being used for COVID-19 patients.
Deaths are averaging about 1,700 per day, Reuters reported, which is up from 1,400 in recent days but not much higher than earlier this winter. The peak average was 3,400 daily deaths in mid-January 2021.
The surging numbers of cases and hospitalizations across the country are straining hospitals. On Jan. 10, Virginia Gov. Ralph Northam declared a state of emergency after the number of intensive care unit hospitalizations more than doubled since Dec. 1, CNN reported. The order allows hospitals to expand bed capacity, use telehealth options, and be more flexible with staffing.
Texas is hiring at least 2,700 medical staff to help with the surge, CNN reported, and Kentucky has mobilized the National Guard to provide support.
“Omicron continues to burn through the commonwealth, growing at levels we have never seen before. Omicron is significantly more contagious than even the Delta variant,” Kentucky Gov. Andy Beshear said during a news briefing Jan. 10.
Kentucky reported its highest weekly total of cases last week and has its highest rate of positive tests, at 26%. Mr. Beshear said the state is down to 134 available adult ICU beds.
“If it spreads at the rate we are seeing, it is certainly going to fill up our hospitals,” he said.
A version of this article first appeared on WebMD.com.
The United States reported 1.35 million new COVID-19 cases on Jan. 10, logging the highest daily total for any country in the world during the pandemic.
The United States set the previous record of 1 million cases on Jan. 3. (A large number of cases are reported on Mondays, since many states don’t provide updates over the weekend, according to Reuters.)
Still, the 7-day average for new cases has surpassed 700,000, tripling in 2 weeks as the contagious Omicron variant continues to spread across the country.
The daily record of new cases came a day after the United States crossed the grim milestone of 60 million COVID-19 cases during the pandemic, according to the latest data from Johns Hopkins University. More than 11 million new cases were reported in the past 28 days, with 5 million reported since Jan. 2.
Globally, more than 310 million cases have been reported, resulting in nearly 5.5 million COVID-19 deaths. Almost 40 million cases have been confirmed worldwide during the past month, with the United States accounting for 28% of those.
Texas became the second state to report more than 5 million cases since the pandemic began, behind California’s total of 6 million cases. Florida has reported more than 4.6 million, while New York has reported more than 4.1 million.
The United States has also hit an all-time high for hospitalizations, with nearly 146,000 COVID-19 patients in hospitals across the country, according to the latest data from the U.S. Department of Health and Human Services. The previous record was 142,000 hospitalizations in January 2021.
Jan. 11’s hospitalizations are more than twice as many as 2 weeks ago, according to CNN. About 78% of inpatient beds are in use nationwide, and 21% are being used for COVID-19 patients.
Deaths are averaging about 1,700 per day, Reuters reported, which is up from 1,400 in recent days but not much higher than earlier this winter. The peak average was 3,400 daily deaths in mid-January 2021.
The surging numbers of cases and hospitalizations across the country are straining hospitals. On Jan. 10, Virginia Gov. Ralph Northam declared a state of emergency after the number of intensive care unit hospitalizations more than doubled since Dec. 1, CNN reported. The order allows hospitals to expand bed capacity, use telehealth options, and be more flexible with staffing.
Texas is hiring at least 2,700 medical staff to help with the surge, CNN reported, and Kentucky has mobilized the National Guard to provide support.
“Omicron continues to burn through the commonwealth, growing at levels we have never seen before. Omicron is significantly more contagious than even the Delta variant,” Kentucky Gov. Andy Beshear said during a news briefing Jan. 10.
Kentucky reported its highest weekly total of cases last week and has its highest rate of positive tests, at 26%. Mr. Beshear said the state is down to 134 available adult ICU beds.
“If it spreads at the rate we are seeing, it is certainly going to fill up our hospitals,” he said.
A version of this article first appeared on WebMD.com.
What if the National Guard Can’t Help?
What if the National Guard Can’t Help?
In early January, Ohio not only set a state record for COVID-19 hospitalizations—it had the fourth highest rate in the country, with 6,747 hospitalized coronavirus patients on January 10, a 40% increase over the previous 21 days. Most were unvaccinated. To help overwhelmed hospitals cope, Ohio Gov. Mike DeWine turned to the National Guard. Unfortunately, nearly half of the Ohio National Guard also were unvaccinated.
By US Department of Defense (DoD) directive, National Guard members must have a COVID-19 vaccination to be deployed on hospital missions. Thus, in COVID hotspots across the nation, governors are on the horns of a dilemma. They want and need to deploy the National Guard to give medical and nonclinical support but aren’t sure whether they will be able to or, indeed, whether they should.
So far, vaccinated teams are already on the ground in a number of states. In Indiana, where hospitalizations jumped 50% over 2 weeks in December, the National Guard sent 6-person teams, all fully vaccinated. In New Hampshire, 70 guards are being deployed to help hospitals with food service, clerical work, and other nonmedical functions. New York Governor Kathy Hochul has deployed guard members for help to ease the strain on nursing homes. Massachusetts Governor Charlie Baker has activated up to 500 guard members; some will be supporting 55 acute care hospital and 12 ambulance services. In Maine, where cases have peaked, Governor Janet Mills activated guard members to support nursing facilities and administer monoclonal antibodies. The Louisiana National Guard has administered more than 542,000 COVID-19 tests and 206,300 vaccines. As many as 1,000 Maryland Air and Army National Guardsmen are being activated to help with testing and other missions.
However, as in Ohio, other states are facing problematic scenarios. For instance, about 40% of the more than 20,000 Texas National Guard are refusing to get vaccinated, challenging the Biden Administration vaccine requirement for all military.
And a court showdown over federal vaccine mandates, started by Governor Kevin Stitt of Oklahoma and joined by the Republican governors of Wyoming, Iowa, Alaska, Nebraska, and Mississippi, came to a head in December. Last November, Stitt asked Defense Secretary Lloyd Austin to exempt Oklahoma’s National Guard from the vaccine mandate. He claimed the requirement violated the personal freedoms of many Oklahomans and could cause them to “potentially sacrifice their personal beliefs.” But in a memo to the Joint Chiefs chairmen, the service secretaries and the head of the National Guard Bureau, Austin wrote that Pentagon funds could not be used to pay for duties performed under Title 32 for members of the Guard who do not comply with the military’s vaccine requirement. (Title 32 refers to Guard operations under state orders.) Austin also said National Guard members must be vaccinated to participate in drills, training, and other duty conducted under Title 32.
Stitt, maintaining that he is commander in chief of the Oklahoma National Guard as long as it operates under Title 32 orders, put out his own memo stipulating that no Guard member was required to get vaccinated. He also ordered Brig. Gen. Thomas Mancino, newly appointed commander of the Oklahoma National Guard, to not enforce the mandate. Subsequently, Mancino issued a statement pointing out that current state law is limited in protecting troops who opt out of the shot. Moreover, if the Guard were called up under federal orders, he said, he would enforce the mandate. Training events, schools, and mobilizations were going to “eventually force you out of that safe harbor,” he wrote, “…This is reality.”
In late December, a federal judge denied Oklahoma’s motion to enjoin the mandate. The Oklahoma Attorney General’s office responded, “We will not be surprised if the President’s vaccine mandate actually reduces the nation’s military readiness instead of promoting it.”
In a press briefing, Pentagon press secretary John Kirby said, “The Secretary has the authorities he needs to require this vaccine across the force, including the National Guard. …[E]ven when they’re in a Title 32 status.” He added, “It is a lawful order for National Guardsmen to receive the COVID vaccine. It’s a lawful order, and refusing to do that, absent of an improved exemption, puts them in the same potential [position] as active-duty members who refuse the vaccine.” That could mean, for instance, loss of pay and membership in the National Guard.
A core rationale for the mandate, according to Secretary Austin, is the need for military readiness—meaning Guard members must be healthy and fit for duty. And that extends to being healthy and fit for missions like transporting at-risk patients. Ohio National Guard Adjutant General Major General John Harris Jr. said, “I would never put a soldier or airman in harm’s way without the best protection we could put on them—body armor, helmets. And this medical readiness is the exact same thing. We’re putting folks into harm’s way.” He has moved the deadline from the Pentagon’s June 30 date to March 31—a move that boosted the vaccination rate from 53% to 56% in one week.
Ohio Governor DeWine has expressed frustration that almost half of the Ohio Army National Guard personnel can’t be deployed on this mission because they’re unvaccinated. “In some of our testing places, 40 to 50% of the people are testing positive,” he said. “So this is a high-risk operation. You need to be protected. The best way for you to be protected is to get the vaccination.”
As of December 2021, according to the National Guard Bureau, the National Guard as a whole was 66% fully vaccinated. The percentages vary according to service; for instance, nearly 90% of airmen have been vaccinated, compared with only 40% of Army Guardsmen. Among the states challenging the mandate, the vaccinated rates have been moving upward: In Alaska, about 92% of the Air National Guard have been vaccinated—leaving roughly 11,000 troops who had not met the December 2 deadline. In Iowa, as of Nov. 30, 91% of Air National Guard and 80% of Army National Guard members had been vaccinated, but about 9,000 soldiers had been directed to get the vaccination or risk disciplinary action. Almost 2,200 of the more than 2,800-strong Wyoming National Guard (77%) have received at least 1 dose. Nebraska Air National Guard’s force of 1,000 was 94% fully vaccinated as of December 1. (Maj Scott Ingalsbe, public affairs officer, said, “Vaccinations are tied to individual medical readiness. They provide service members with the best protection available so they can perform missions across the globe.”).
In most states, Army National Guard members have until June 30, 2022, to comply. “Our soldiers …have until [the DoD’s deadline], and some of them are just going to wait close to the deadline,” John Goheen of the National Guard Association of the United States said in a discussion on NPR. “That’s human nature.”
Earlier this month, Texas Governor Greg Abbott told National Guard members they can ignore the Pentagon’s COVID-19 vaccine mandate: “President Biden is not your commander-in-chief.” He has also sued the Biden administration over the requirement.
In the meantime, the hospitals at breaking point must hope for the best and take as much help as they can get.
In early January, Ohio not only set a state record for COVID-19 hospitalizations—it had the fourth highest rate in the country, with 6,747 hospitalized coronavirus patients on January 10, a 40% increase over the previous 21 days. Most were unvaccinated. To help overwhelmed hospitals cope, Ohio Gov. Mike DeWine turned to the National Guard. Unfortunately, nearly half of the Ohio National Guard also were unvaccinated.
By US Department of Defense (DoD) directive, National Guard members must have a COVID-19 vaccination to be deployed on hospital missions. Thus, in COVID hotspots across the nation, governors are on the horns of a dilemma. They want and need to deploy the National Guard to give medical and nonclinical support but aren’t sure whether they will be able to or, indeed, whether they should.
So far, vaccinated teams are already on the ground in a number of states. In Indiana, where hospitalizations jumped 50% over 2 weeks in December, the National Guard sent 6-person teams, all fully vaccinated. In New Hampshire, 70 guards are being deployed to help hospitals with food service, clerical work, and other nonmedical functions. New York Governor Kathy Hochul has deployed guard members for help to ease the strain on nursing homes. Massachusetts Governor Charlie Baker has activated up to 500 guard members; some will be supporting 55 acute care hospital and 12 ambulance services. In Maine, where cases have peaked, Governor Janet Mills activated guard members to support nursing facilities and administer monoclonal antibodies. The Louisiana National Guard has administered more than 542,000 COVID-19 tests and 206,300 vaccines. As many as 1,000 Maryland Air and Army National Guardsmen are being activated to help with testing and other missions.
However, as in Ohio, other states are facing problematic scenarios. For instance, about 40% of the more than 20,000 Texas National Guard are refusing to get vaccinated, challenging the Biden Administration vaccine requirement for all military.
And a court showdown over federal vaccine mandates, started by Governor Kevin Stitt of Oklahoma and joined by the Republican governors of Wyoming, Iowa, Alaska, Nebraska, and Mississippi, came to a head in December. Last November, Stitt asked Defense Secretary Lloyd Austin to exempt Oklahoma’s National Guard from the vaccine mandate. He claimed the requirement violated the personal freedoms of many Oklahomans and could cause them to “potentially sacrifice their personal beliefs.” But in a memo to the Joint Chiefs chairmen, the service secretaries and the head of the National Guard Bureau, Austin wrote that Pentagon funds could not be used to pay for duties performed under Title 32 for members of the Guard who do not comply with the military’s vaccine requirement. (Title 32 refers to Guard operations under state orders.) Austin also said National Guard members must be vaccinated to participate in drills, training, and other duty conducted under Title 32.
Stitt, maintaining that he is commander in chief of the Oklahoma National Guard as long as it operates under Title 32 orders, put out his own memo stipulating that no Guard member was required to get vaccinated. He also ordered Brig. Gen. Thomas Mancino, newly appointed commander of the Oklahoma National Guard, to not enforce the mandate. Subsequently, Mancino issued a statement pointing out that current state law is limited in protecting troops who opt out of the shot. Moreover, if the Guard were called up under federal orders, he said, he would enforce the mandate. Training events, schools, and mobilizations were going to “eventually force you out of that safe harbor,” he wrote, “…This is reality.”
In late December, a federal judge denied Oklahoma’s motion to enjoin the mandate. The Oklahoma Attorney General’s office responded, “We will not be surprised if the President’s vaccine mandate actually reduces the nation’s military readiness instead of promoting it.”
In a press briefing, Pentagon press secretary John Kirby said, “The Secretary has the authorities he needs to require this vaccine across the force, including the National Guard. …[E]ven when they’re in a Title 32 status.” He added, “It is a lawful order for National Guardsmen to receive the COVID vaccine. It’s a lawful order, and refusing to do that, absent of an improved exemption, puts them in the same potential [position] as active-duty members who refuse the vaccine.” That could mean, for instance, loss of pay and membership in the National Guard.
A core rationale for the mandate, according to Secretary Austin, is the need for military readiness—meaning Guard members must be healthy and fit for duty. And that extends to being healthy and fit for missions like transporting at-risk patients. Ohio National Guard Adjutant General Major General John Harris Jr. said, “I would never put a soldier or airman in harm’s way without the best protection we could put on them—body armor, helmets. And this medical readiness is the exact same thing. We’re putting folks into harm’s way.” He has moved the deadline from the Pentagon’s June 30 date to March 31—a move that boosted the vaccination rate from 53% to 56% in one week.
Ohio Governor DeWine has expressed frustration that almost half of the Ohio Army National Guard personnel can’t be deployed on this mission because they’re unvaccinated. “In some of our testing places, 40 to 50% of the people are testing positive,” he said. “So this is a high-risk operation. You need to be protected. The best way for you to be protected is to get the vaccination.”
As of December 2021, according to the National Guard Bureau, the National Guard as a whole was 66% fully vaccinated. The percentages vary according to service; for instance, nearly 90% of airmen have been vaccinated, compared with only 40% of Army Guardsmen. Among the states challenging the mandate, the vaccinated rates have been moving upward: In Alaska, about 92% of the Air National Guard have been vaccinated—leaving roughly 11,000 troops who had not met the December 2 deadline. In Iowa, as of Nov. 30, 91% of Air National Guard and 80% of Army National Guard members had been vaccinated, but about 9,000 soldiers had been directed to get the vaccination or risk disciplinary action. Almost 2,200 of the more than 2,800-strong Wyoming National Guard (77%) have received at least 1 dose. Nebraska Air National Guard’s force of 1,000 was 94% fully vaccinated as of December 1. (Maj Scott Ingalsbe, public affairs officer, said, “Vaccinations are tied to individual medical readiness. They provide service members with the best protection available so they can perform missions across the globe.”).
In most states, Army National Guard members have until June 30, 2022, to comply. “Our soldiers …have until [the DoD’s deadline], and some of them are just going to wait close to the deadline,” John Goheen of the National Guard Association of the United States said in a discussion on NPR. “That’s human nature.”
Earlier this month, Texas Governor Greg Abbott told National Guard members they can ignore the Pentagon’s COVID-19 vaccine mandate: “President Biden is not your commander-in-chief.” He has also sued the Biden administration over the requirement.
In the meantime, the hospitals at breaking point must hope for the best and take as much help as they can get.
In early January, Ohio not only set a state record for COVID-19 hospitalizations—it had the fourth highest rate in the country, with 6,747 hospitalized coronavirus patients on January 10, a 40% increase over the previous 21 days. Most were unvaccinated. To help overwhelmed hospitals cope, Ohio Gov. Mike DeWine turned to the National Guard. Unfortunately, nearly half of the Ohio National Guard also were unvaccinated.
By US Department of Defense (DoD) directive, National Guard members must have a COVID-19 vaccination to be deployed on hospital missions. Thus, in COVID hotspots across the nation, governors are on the horns of a dilemma. They want and need to deploy the National Guard to give medical and nonclinical support but aren’t sure whether they will be able to or, indeed, whether they should.
So far, vaccinated teams are already on the ground in a number of states. In Indiana, where hospitalizations jumped 50% over 2 weeks in December, the National Guard sent 6-person teams, all fully vaccinated. In New Hampshire, 70 guards are being deployed to help hospitals with food service, clerical work, and other nonmedical functions. New York Governor Kathy Hochul has deployed guard members for help to ease the strain on nursing homes. Massachusetts Governor Charlie Baker has activated up to 500 guard members; some will be supporting 55 acute care hospital and 12 ambulance services. In Maine, where cases have peaked, Governor Janet Mills activated guard members to support nursing facilities and administer monoclonal antibodies. The Louisiana National Guard has administered more than 542,000 COVID-19 tests and 206,300 vaccines. As many as 1,000 Maryland Air and Army National Guardsmen are being activated to help with testing and other missions.
However, as in Ohio, other states are facing problematic scenarios. For instance, about 40% of the more than 20,000 Texas National Guard are refusing to get vaccinated, challenging the Biden Administration vaccine requirement for all military.
And a court showdown over federal vaccine mandates, started by Governor Kevin Stitt of Oklahoma and joined by the Republican governors of Wyoming, Iowa, Alaska, Nebraska, and Mississippi, came to a head in December. Last November, Stitt asked Defense Secretary Lloyd Austin to exempt Oklahoma’s National Guard from the vaccine mandate. He claimed the requirement violated the personal freedoms of many Oklahomans and could cause them to “potentially sacrifice their personal beliefs.” But in a memo to the Joint Chiefs chairmen, the service secretaries and the head of the National Guard Bureau, Austin wrote that Pentagon funds could not be used to pay for duties performed under Title 32 for members of the Guard who do not comply with the military’s vaccine requirement. (Title 32 refers to Guard operations under state orders.) Austin also said National Guard members must be vaccinated to participate in drills, training, and other duty conducted under Title 32.
Stitt, maintaining that he is commander in chief of the Oklahoma National Guard as long as it operates under Title 32 orders, put out his own memo stipulating that no Guard member was required to get vaccinated. He also ordered Brig. Gen. Thomas Mancino, newly appointed commander of the Oklahoma National Guard, to not enforce the mandate. Subsequently, Mancino issued a statement pointing out that current state law is limited in protecting troops who opt out of the shot. Moreover, if the Guard were called up under federal orders, he said, he would enforce the mandate. Training events, schools, and mobilizations were going to “eventually force you out of that safe harbor,” he wrote, “…This is reality.”
In late December, a federal judge denied Oklahoma’s motion to enjoin the mandate. The Oklahoma Attorney General’s office responded, “We will not be surprised if the President’s vaccine mandate actually reduces the nation’s military readiness instead of promoting it.”
In a press briefing, Pentagon press secretary John Kirby said, “The Secretary has the authorities he needs to require this vaccine across the force, including the National Guard. …[E]ven when they’re in a Title 32 status.” He added, “It is a lawful order for National Guardsmen to receive the COVID vaccine. It’s a lawful order, and refusing to do that, absent of an improved exemption, puts them in the same potential [position] as active-duty members who refuse the vaccine.” That could mean, for instance, loss of pay and membership in the National Guard.
A core rationale for the mandate, according to Secretary Austin, is the need for military readiness—meaning Guard members must be healthy and fit for duty. And that extends to being healthy and fit for missions like transporting at-risk patients. Ohio National Guard Adjutant General Major General John Harris Jr. said, “I would never put a soldier or airman in harm’s way without the best protection we could put on them—body armor, helmets. And this medical readiness is the exact same thing. We’re putting folks into harm’s way.” He has moved the deadline from the Pentagon’s June 30 date to March 31—a move that boosted the vaccination rate from 53% to 56% in one week.
Ohio Governor DeWine has expressed frustration that almost half of the Ohio Army National Guard personnel can’t be deployed on this mission because they’re unvaccinated. “In some of our testing places, 40 to 50% of the people are testing positive,” he said. “So this is a high-risk operation. You need to be protected. The best way for you to be protected is to get the vaccination.”
As of December 2021, according to the National Guard Bureau, the National Guard as a whole was 66% fully vaccinated. The percentages vary according to service; for instance, nearly 90% of airmen have been vaccinated, compared with only 40% of Army Guardsmen. Among the states challenging the mandate, the vaccinated rates have been moving upward: In Alaska, about 92% of the Air National Guard have been vaccinated—leaving roughly 11,000 troops who had not met the December 2 deadline. In Iowa, as of Nov. 30, 91% of Air National Guard and 80% of Army National Guard members had been vaccinated, but about 9,000 soldiers had been directed to get the vaccination or risk disciplinary action. Almost 2,200 of the more than 2,800-strong Wyoming National Guard (77%) have received at least 1 dose. Nebraska Air National Guard’s force of 1,000 was 94% fully vaccinated as of December 1. (Maj Scott Ingalsbe, public affairs officer, said, “Vaccinations are tied to individual medical readiness. They provide service members with the best protection available so they can perform missions across the globe.”).
In most states, Army National Guard members have until June 30, 2022, to comply. “Our soldiers …have until [the DoD’s deadline], and some of them are just going to wait close to the deadline,” John Goheen of the National Guard Association of the United States said in a discussion on NPR. “That’s human nature.”
Earlier this month, Texas Governor Greg Abbott told National Guard members they can ignore the Pentagon’s COVID-19 vaccine mandate: “President Biden is not your commander-in-chief.” He has also sued the Biden administration over the requirement.
In the meantime, the hospitals at breaking point must hope for the best and take as much help as they can get.
What if the National Guard Can’t Help?
What if the National Guard Can’t Help?
HPV testing plus cytology catches two times more cervical lesions
, according to a new study.
The study, which analyzed data from Mexico’s population-based hrHPV screening program over 6 years, confirms the importance of HPV screening for catching high-grade cervical lesions early.
“Our results provide evidence that hrHPV testing is the best strategy for a timely diagnosis of CIN2+ lesions while avoiding overtreatment of young women,” the study authors write. “Many countries now use hrHPV testing as the primary screening method, given it has higher sensitivity and detects more cervical cancer precursor lesions, such as CIN2+.”
According to Erik Jansen, MSc, the analysis supports recent updates to U.S. screening standards and confirms findings from previous trials, which show that HPV testing significantly improves prevention of cervical cancer.
“The significance of this paper is that the data reported is from a long follow-up in a country that implemented HPV screening on a large scale,” Mr. Jansen, PhD candidate in the Department of Public Health, Erasmus University Medical Center, Rotterdam, the Netherlands, told this news organization.
The study, conducted by Mexico’s National Institute of Public Health, analyzed screening data from the country’s public cervical cancer prevention program from 2010 to 2015. More than 2 million women aged 34 to 65 who had hrHPV-based screening tests followed by cytologic triage if they were HPV positive were included, as were 2.8 million women of the same age who received cytologic testing alone.
In the hrHPV group, 1.2% of women (n = 24,276) received referrals to colposcopy versus 3.1% of women (n = 90,980) in the cytology group. And among all women, only 0.8% who had abnormal results (n = 16,459) in the HPV went for a colposcopy versus 1.5% (n = 43,638) in the cytology group.
Overall, the authors found that 13.3 colposcopies were required to detect a single CIN2+ case in the cytology group compared to 5.7 colposcopies in the hrHPV with cytologic triage group.
The authors also note that the cost of colposcopies was three times lower in the HPV testing group and that the positive predictive value of hrHPV testing with cytologic triage was 17.5% versus 7.5% for cytology alone.
“The positive predictive value did not change for either screening strategy whether or not women lost to follow-up were taken into account,” the authors write.
Although Mr. Jansen noted that the findings are important, he also pointed to several limitations – namely, the significant loss to follow-up in the HPV group.
The HPV testing and cytologic triage happened in separate visits, and under the two-visit protocol, more than 50% of women who tested positive for HPV didn’t return for cytology. Such a significant loss to follow-up may call some of the findings into question, Mr. Jansen noted.
For instance, the rate of colposcopy referrals does not account for HPV-positive women who skipped their cytology screening. Assuming the same HPV risk for women who received cytology and those who did not, Mr. Jansen calculated that without any loss to follow-up, the colposcopy referral rate would have increased from the reported 1.2% to 2.6%, which is much closer to the 3.1% of the women referred in the cytology arm.
The lower colposcopy costs in the HPV group were also likely due, in part, to the loss to follow-up, which is not necessarily a good thing, Mr. Jansen said.
Still, “this study does confirm the finding that a primary HPV screening program is more effective than cytology [alone],” Mr. Jansen said.
Co-author Eduardo Franco reported receiving grants and personal fees from MSD and has a pending patent, “Methylation Markers in Cervical Cancer.” All other authors reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
, according to a new study.
The study, which analyzed data from Mexico’s population-based hrHPV screening program over 6 years, confirms the importance of HPV screening for catching high-grade cervical lesions early.
“Our results provide evidence that hrHPV testing is the best strategy for a timely diagnosis of CIN2+ lesions while avoiding overtreatment of young women,” the study authors write. “Many countries now use hrHPV testing as the primary screening method, given it has higher sensitivity and detects more cervical cancer precursor lesions, such as CIN2+.”
According to Erik Jansen, MSc, the analysis supports recent updates to U.S. screening standards and confirms findings from previous trials, which show that HPV testing significantly improves prevention of cervical cancer.
“The significance of this paper is that the data reported is from a long follow-up in a country that implemented HPV screening on a large scale,” Mr. Jansen, PhD candidate in the Department of Public Health, Erasmus University Medical Center, Rotterdam, the Netherlands, told this news organization.
The study, conducted by Mexico’s National Institute of Public Health, analyzed screening data from the country’s public cervical cancer prevention program from 2010 to 2015. More than 2 million women aged 34 to 65 who had hrHPV-based screening tests followed by cytologic triage if they were HPV positive were included, as were 2.8 million women of the same age who received cytologic testing alone.
In the hrHPV group, 1.2% of women (n = 24,276) received referrals to colposcopy versus 3.1% of women (n = 90,980) in the cytology group. And among all women, only 0.8% who had abnormal results (n = 16,459) in the HPV went for a colposcopy versus 1.5% (n = 43,638) in the cytology group.
Overall, the authors found that 13.3 colposcopies were required to detect a single CIN2+ case in the cytology group compared to 5.7 colposcopies in the hrHPV with cytologic triage group.
The authors also note that the cost of colposcopies was three times lower in the HPV testing group and that the positive predictive value of hrHPV testing with cytologic triage was 17.5% versus 7.5% for cytology alone.
“The positive predictive value did not change for either screening strategy whether or not women lost to follow-up were taken into account,” the authors write.
Although Mr. Jansen noted that the findings are important, he also pointed to several limitations – namely, the significant loss to follow-up in the HPV group.
The HPV testing and cytologic triage happened in separate visits, and under the two-visit protocol, more than 50% of women who tested positive for HPV didn’t return for cytology. Such a significant loss to follow-up may call some of the findings into question, Mr. Jansen noted.
For instance, the rate of colposcopy referrals does not account for HPV-positive women who skipped their cytology screening. Assuming the same HPV risk for women who received cytology and those who did not, Mr. Jansen calculated that without any loss to follow-up, the colposcopy referral rate would have increased from the reported 1.2% to 2.6%, which is much closer to the 3.1% of the women referred in the cytology arm.
The lower colposcopy costs in the HPV group were also likely due, in part, to the loss to follow-up, which is not necessarily a good thing, Mr. Jansen said.
Still, “this study does confirm the finding that a primary HPV screening program is more effective than cytology [alone],” Mr. Jansen said.
Co-author Eduardo Franco reported receiving grants and personal fees from MSD and has a pending patent, “Methylation Markers in Cervical Cancer.” All other authors reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
, according to a new study.
The study, which analyzed data from Mexico’s population-based hrHPV screening program over 6 years, confirms the importance of HPV screening for catching high-grade cervical lesions early.
“Our results provide evidence that hrHPV testing is the best strategy for a timely diagnosis of CIN2+ lesions while avoiding overtreatment of young women,” the study authors write. “Many countries now use hrHPV testing as the primary screening method, given it has higher sensitivity and detects more cervical cancer precursor lesions, such as CIN2+.”
According to Erik Jansen, MSc, the analysis supports recent updates to U.S. screening standards and confirms findings from previous trials, which show that HPV testing significantly improves prevention of cervical cancer.
“The significance of this paper is that the data reported is from a long follow-up in a country that implemented HPV screening on a large scale,” Mr. Jansen, PhD candidate in the Department of Public Health, Erasmus University Medical Center, Rotterdam, the Netherlands, told this news organization.
The study, conducted by Mexico’s National Institute of Public Health, analyzed screening data from the country’s public cervical cancer prevention program from 2010 to 2015. More than 2 million women aged 34 to 65 who had hrHPV-based screening tests followed by cytologic triage if they were HPV positive were included, as were 2.8 million women of the same age who received cytologic testing alone.
In the hrHPV group, 1.2% of women (n = 24,276) received referrals to colposcopy versus 3.1% of women (n = 90,980) in the cytology group. And among all women, only 0.8% who had abnormal results (n = 16,459) in the HPV went for a colposcopy versus 1.5% (n = 43,638) in the cytology group.
Overall, the authors found that 13.3 colposcopies were required to detect a single CIN2+ case in the cytology group compared to 5.7 colposcopies in the hrHPV with cytologic triage group.
The authors also note that the cost of colposcopies was three times lower in the HPV testing group and that the positive predictive value of hrHPV testing with cytologic triage was 17.5% versus 7.5% for cytology alone.
“The positive predictive value did not change for either screening strategy whether or not women lost to follow-up were taken into account,” the authors write.
Although Mr. Jansen noted that the findings are important, he also pointed to several limitations – namely, the significant loss to follow-up in the HPV group.
The HPV testing and cytologic triage happened in separate visits, and under the two-visit protocol, more than 50% of women who tested positive for HPV didn’t return for cytology. Such a significant loss to follow-up may call some of the findings into question, Mr. Jansen noted.
For instance, the rate of colposcopy referrals does not account for HPV-positive women who skipped their cytology screening. Assuming the same HPV risk for women who received cytology and those who did not, Mr. Jansen calculated that without any loss to follow-up, the colposcopy referral rate would have increased from the reported 1.2% to 2.6%, which is much closer to the 3.1% of the women referred in the cytology arm.
The lower colposcopy costs in the HPV group were also likely due, in part, to the loss to follow-up, which is not necessarily a good thing, Mr. Jansen said.
Still, “this study does confirm the finding that a primary HPV screening program is more effective than cytology [alone],” Mr. Jansen said.
Co-author Eduardo Franco reported receiving grants and personal fees from MSD and has a pending patent, “Methylation Markers in Cervical Cancer.” All other authors reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
CDC: More kids hospitalized with COVID since pandemic began
Hospital admissions of U.S. children younger than 5 – the only group ineligible for vaccination – have reached their peak since the start of the pandemic, according to new data from the Centers for Disease Control and Prevention.
CDC Director Rochelle Walensky, MD, said the higher numbers show the importance of vaccination for all eligible groups.
“This is the highest number of pediatric hospitalizations we’ve seen throughout the pandemic, which we said about Delta until now,” she said at a CDC briefing Friday. “This very well may be that there are just more cases out there, and our children are more vulnerable when they have more cases surrounding them.”
Despite the skyrocketing admissions, hospitalizations are still relatively low for children, she said. The hospitalization rate for children under 5 is 4 in 100,000, and it’s about 1 in 100,000 in children 5-17.
Dr. Walensky said not all children are being hospitalized for COVID-19 – some are admitted for unrelated issues and test positive but don’t have symptoms.
“We are still learning more about the severity of Omicron in children,” she said, noting that just over 50% of children 12-18 are fully vaccinated, while only 16% of those ages 5-11 are fully vaccinated.
Friday’s teleconference was the first CDC briefing in several months and comes on the heels of recent guideline updates for testing and isolation that have left the American public dumbfounded. When asked why the briefing was held, Dr. Walensky said there had been interest in hearing more from the CDC, saying, “I anticipate this will be the first of many briefings.”
She also defended the confusing guideline changes, saying, “We’re in an unprecedented time with the speed of Omicron cases rising. … This is hard, and I am committed to continuing to improve as we learn more about the science and communicate that to you.”
A version of this article first appeared on WebMD.com.
Hospital admissions of U.S. children younger than 5 – the only group ineligible for vaccination – have reached their peak since the start of the pandemic, according to new data from the Centers for Disease Control and Prevention.
CDC Director Rochelle Walensky, MD, said the higher numbers show the importance of vaccination for all eligible groups.
“This is the highest number of pediatric hospitalizations we’ve seen throughout the pandemic, which we said about Delta until now,” she said at a CDC briefing Friday. “This very well may be that there are just more cases out there, and our children are more vulnerable when they have more cases surrounding them.”
Despite the skyrocketing admissions, hospitalizations are still relatively low for children, she said. The hospitalization rate for children under 5 is 4 in 100,000, and it’s about 1 in 100,000 in children 5-17.
Dr. Walensky said not all children are being hospitalized for COVID-19 – some are admitted for unrelated issues and test positive but don’t have symptoms.
“We are still learning more about the severity of Omicron in children,” she said, noting that just over 50% of children 12-18 are fully vaccinated, while only 16% of those ages 5-11 are fully vaccinated.
Friday’s teleconference was the first CDC briefing in several months and comes on the heels of recent guideline updates for testing and isolation that have left the American public dumbfounded. When asked why the briefing was held, Dr. Walensky said there had been interest in hearing more from the CDC, saying, “I anticipate this will be the first of many briefings.”
She also defended the confusing guideline changes, saying, “We’re in an unprecedented time with the speed of Omicron cases rising. … This is hard, and I am committed to continuing to improve as we learn more about the science and communicate that to you.”
A version of this article first appeared on WebMD.com.
Hospital admissions of U.S. children younger than 5 – the only group ineligible for vaccination – have reached their peak since the start of the pandemic, according to new data from the Centers for Disease Control and Prevention.
CDC Director Rochelle Walensky, MD, said the higher numbers show the importance of vaccination for all eligible groups.
“This is the highest number of pediatric hospitalizations we’ve seen throughout the pandemic, which we said about Delta until now,” she said at a CDC briefing Friday. “This very well may be that there are just more cases out there, and our children are more vulnerable when they have more cases surrounding them.”
Despite the skyrocketing admissions, hospitalizations are still relatively low for children, she said. The hospitalization rate for children under 5 is 4 in 100,000, and it’s about 1 in 100,000 in children 5-17.
Dr. Walensky said not all children are being hospitalized for COVID-19 – some are admitted for unrelated issues and test positive but don’t have symptoms.
“We are still learning more about the severity of Omicron in children,” she said, noting that just over 50% of children 12-18 are fully vaccinated, while only 16% of those ages 5-11 are fully vaccinated.
Friday’s teleconference was the first CDC briefing in several months and comes on the heels of recent guideline updates for testing and isolation that have left the American public dumbfounded. When asked why the briefing was held, Dr. Walensky said there had been interest in hearing more from the CDC, saying, “I anticipate this will be the first of many briefings.”
She also defended the confusing guideline changes, saying, “We’re in an unprecedented time with the speed of Omicron cases rising. … This is hard, and I am committed to continuing to improve as we learn more about the science and communicate that to you.”
A version of this article first appeared on WebMD.com.
Pediatric antibiotic prescriptions plummeted in pandemic
Antibiotic prescribing in pediatric primary care decreased dramatically when the COVID-19 pandemic hit, and new research indicates that drop was sustained through June of 2021.
Lauren Dutcher, MD, with the division of infectious diseases at Hospital of the University of Pennsylvania in Philadelphia, led a study of 27 pediatric primary care practices in the United States. Encounters from Jan. 1, 2018, through June 30, 2021, were included.
Researchers found a 72.7% drop in antibiotic prescriptions when they compared prepandemic April 2019 through December 2019 with the same period in 2020.
Prescriptions remained at the lower levels, primarily driven by reductions in respiratory tract infection (RTI) encounters, and began to rise only in April of 2021, the authors write.
Findings were published online Jan. 11 in Pediatrics.
Researchers report there were 69,327 antibiotic prescriptions from April through December in 2019 and 18,935 antibiotic prescriptions during the same months in 2020.
“The reduction in prescriptions at visits for respiratory tract infection (RTI) accounted for 87.3% of this decrease,” the authors write.
Both prescribing and acute non–COVID-19 respiratory tract infection diagnoses decreased.
Researchers conclude reductions in viral RTI transmission likely played a large role in reduced RTI pediatric visits and antibiotic prescriptions.
Dr. Dutcher told this publication the reduction was likely caused by a combination of less viral transmission of respiratory infections, helped in part by masking and distancing, but also avoidance of health care in the pandemic.
She said the data reinforce the need for appropriate prescribing.
“Antibiotic prescribing is really heavily driven by respiratory infections so this should continue to clue providers in on how frequently that can be unnecessary,“ she said.
Dr. Dutcher said there was probably a reduction in secondary bacterial infections as well as the viral infections.
The research is more comprehensive than some other previous studies, the authors write.
“Although other studies demonstrated early reductions in RTIs and antibiotic prescribing during the COVID-19 pandemic, to our knowledge, this is the first study to demonstrate a sustained decrease in antibiotic prescribing in pediatric primary care throughout 2020 and early 2021,” they write.
The findings also suggest benefits of preventive measures during the pandemic, the authors say.
“Our data suggest that reducing community viral RTI transmission through social distancing and masking corresponds with a reduction in antibiotic prescribing,” they write.
Kao-Ping Chua, MD, a pediatrician and an assistant professor of pediatrics at the University of Michigan in Ann Arbor, said the reductions indicate one of two things is happening: either children aren’t getting sick as often during the pandemic or they are getting sick, but not coming in.
But if they were sick and not coming in, the expectation would be that they would show up in large numbers in emergency departments from untreated infections, he said.
“We just haven’t seen that,” he said.
He said one of the main points the authors make is that masks, distancing, and hand washing may be keeping kids from diseases beyond COVID-19.
He said longer-term data will be needed to show if the trend highlighted in this paper lasts, given children have now returned to school and pediatricians started to see lots of respiratory syncytial virus (RSV) cases this summer.
Anecdotally, he said, he has been prescribing more antibiotics of late for presentations such as ear infections.
Dr. Dutcher said that, though her team doesn’t have data yet since the end of the study period, she agreed that anecdotally it is likely that the prescriptions have been on the rise since June.
Dr. Chua said the reduction in visits also reduces the chance that a physician will be tempted to give in to families’ demands to prescribe an antibiotic.
“Every visit for a sick child represents an opportunity to inappropriately prescribe antibiotics,” Dr. Chua said. Dr. Chua’s own research has found that up to one-quarter of pediatric and adult antibiotic prescriptions are unnecessary.
This work was supported by a Centers for Disease Control and Prevention cooperative agreement, Epicenters for the Prevention of Healthcare Associated Infections. Dr. Dutcher and Dr. Chua had no relevant financial disclosures.
This article was updated 1/11/22.
Antibiotic prescribing in pediatric primary care decreased dramatically when the COVID-19 pandemic hit, and new research indicates that drop was sustained through June of 2021.
Lauren Dutcher, MD, with the division of infectious diseases at Hospital of the University of Pennsylvania in Philadelphia, led a study of 27 pediatric primary care practices in the United States. Encounters from Jan. 1, 2018, through June 30, 2021, were included.
Researchers found a 72.7% drop in antibiotic prescriptions when they compared prepandemic April 2019 through December 2019 with the same period in 2020.
Prescriptions remained at the lower levels, primarily driven by reductions in respiratory tract infection (RTI) encounters, and began to rise only in April of 2021, the authors write.
Findings were published online Jan. 11 in Pediatrics.
Researchers report there were 69,327 antibiotic prescriptions from April through December in 2019 and 18,935 antibiotic prescriptions during the same months in 2020.
“The reduction in prescriptions at visits for respiratory tract infection (RTI) accounted for 87.3% of this decrease,” the authors write.
Both prescribing and acute non–COVID-19 respiratory tract infection diagnoses decreased.
Researchers conclude reductions in viral RTI transmission likely played a large role in reduced RTI pediatric visits and antibiotic prescriptions.
Dr. Dutcher told this publication the reduction was likely caused by a combination of less viral transmission of respiratory infections, helped in part by masking and distancing, but also avoidance of health care in the pandemic.
She said the data reinforce the need for appropriate prescribing.
“Antibiotic prescribing is really heavily driven by respiratory infections so this should continue to clue providers in on how frequently that can be unnecessary,“ she said.
Dr. Dutcher said there was probably a reduction in secondary bacterial infections as well as the viral infections.
The research is more comprehensive than some other previous studies, the authors write.
“Although other studies demonstrated early reductions in RTIs and antibiotic prescribing during the COVID-19 pandemic, to our knowledge, this is the first study to demonstrate a sustained decrease in antibiotic prescribing in pediatric primary care throughout 2020 and early 2021,” they write.
The findings also suggest benefits of preventive measures during the pandemic, the authors say.
“Our data suggest that reducing community viral RTI transmission through social distancing and masking corresponds with a reduction in antibiotic prescribing,” they write.
Kao-Ping Chua, MD, a pediatrician and an assistant professor of pediatrics at the University of Michigan in Ann Arbor, said the reductions indicate one of two things is happening: either children aren’t getting sick as often during the pandemic or they are getting sick, but not coming in.
But if they were sick and not coming in, the expectation would be that they would show up in large numbers in emergency departments from untreated infections, he said.
“We just haven’t seen that,” he said.
He said one of the main points the authors make is that masks, distancing, and hand washing may be keeping kids from diseases beyond COVID-19.
He said longer-term data will be needed to show if the trend highlighted in this paper lasts, given children have now returned to school and pediatricians started to see lots of respiratory syncytial virus (RSV) cases this summer.
Anecdotally, he said, he has been prescribing more antibiotics of late for presentations such as ear infections.
Dr. Dutcher said that, though her team doesn’t have data yet since the end of the study period, she agreed that anecdotally it is likely that the prescriptions have been on the rise since June.
Dr. Chua said the reduction in visits also reduces the chance that a physician will be tempted to give in to families’ demands to prescribe an antibiotic.
“Every visit for a sick child represents an opportunity to inappropriately prescribe antibiotics,” Dr. Chua said. Dr. Chua’s own research has found that up to one-quarter of pediatric and adult antibiotic prescriptions are unnecessary.
This work was supported by a Centers for Disease Control and Prevention cooperative agreement, Epicenters for the Prevention of Healthcare Associated Infections. Dr. Dutcher and Dr. Chua had no relevant financial disclosures.
This article was updated 1/11/22.
Antibiotic prescribing in pediatric primary care decreased dramatically when the COVID-19 pandemic hit, and new research indicates that drop was sustained through June of 2021.
Lauren Dutcher, MD, with the division of infectious diseases at Hospital of the University of Pennsylvania in Philadelphia, led a study of 27 pediatric primary care practices in the United States. Encounters from Jan. 1, 2018, through June 30, 2021, were included.
Researchers found a 72.7% drop in antibiotic prescriptions when they compared prepandemic April 2019 through December 2019 with the same period in 2020.
Prescriptions remained at the lower levels, primarily driven by reductions in respiratory tract infection (RTI) encounters, and began to rise only in April of 2021, the authors write.
Findings were published online Jan. 11 in Pediatrics.
Researchers report there were 69,327 antibiotic prescriptions from April through December in 2019 and 18,935 antibiotic prescriptions during the same months in 2020.
“The reduction in prescriptions at visits for respiratory tract infection (RTI) accounted for 87.3% of this decrease,” the authors write.
Both prescribing and acute non–COVID-19 respiratory tract infection diagnoses decreased.
Researchers conclude reductions in viral RTI transmission likely played a large role in reduced RTI pediatric visits and antibiotic prescriptions.
Dr. Dutcher told this publication the reduction was likely caused by a combination of less viral transmission of respiratory infections, helped in part by masking and distancing, but also avoidance of health care in the pandemic.
She said the data reinforce the need for appropriate prescribing.
“Antibiotic prescribing is really heavily driven by respiratory infections so this should continue to clue providers in on how frequently that can be unnecessary,“ she said.
Dr. Dutcher said there was probably a reduction in secondary bacterial infections as well as the viral infections.
The research is more comprehensive than some other previous studies, the authors write.
“Although other studies demonstrated early reductions in RTIs and antibiotic prescribing during the COVID-19 pandemic, to our knowledge, this is the first study to demonstrate a sustained decrease in antibiotic prescribing in pediatric primary care throughout 2020 and early 2021,” they write.
The findings also suggest benefits of preventive measures during the pandemic, the authors say.
“Our data suggest that reducing community viral RTI transmission through social distancing and masking corresponds with a reduction in antibiotic prescribing,” they write.
Kao-Ping Chua, MD, a pediatrician and an assistant professor of pediatrics at the University of Michigan in Ann Arbor, said the reductions indicate one of two things is happening: either children aren’t getting sick as often during the pandemic or they are getting sick, but not coming in.
But if they were sick and not coming in, the expectation would be that they would show up in large numbers in emergency departments from untreated infections, he said.
“We just haven’t seen that,” he said.
He said one of the main points the authors make is that masks, distancing, and hand washing may be keeping kids from diseases beyond COVID-19.
He said longer-term data will be needed to show if the trend highlighted in this paper lasts, given children have now returned to school and pediatricians started to see lots of respiratory syncytial virus (RSV) cases this summer.
Anecdotally, he said, he has been prescribing more antibiotics of late for presentations such as ear infections.
Dr. Dutcher said that, though her team doesn’t have data yet since the end of the study period, she agreed that anecdotally it is likely that the prescriptions have been on the rise since June.
Dr. Chua said the reduction in visits also reduces the chance that a physician will be tempted to give in to families’ demands to prescribe an antibiotic.
“Every visit for a sick child represents an opportunity to inappropriately prescribe antibiotics,” Dr. Chua said. Dr. Chua’s own research has found that up to one-quarter of pediatric and adult antibiotic prescriptions are unnecessary.
This work was supported by a Centers for Disease Control and Prevention cooperative agreement, Epicenters for the Prevention of Healthcare Associated Infections. Dr. Dutcher and Dr. Chua had no relevant financial disclosures.
This article was updated 1/11/22.
FROM PEDIATRICS
At-risk Americans become eligible for fourth COVID shot this week
The Centers for Disease Control and Prevention endorsed a third dose of the Pfizer or Moderna vaccines for moderately and severely immunocompromised people on Aug. 13, which is considered part of their first immunization series rather than a booster shot.
In October, the CDC said moderately and severely immunocompromised people could receive a booster shot, or a fourth dose of the vaccine , 6 months after their third dose.
But the CDC last week shortened the timeline to 5 months for a booster shot of the Pfizer or Moderna vaccines. That means immunocompromised people could begin signing up for a fourth shot later this week, the New York Times reported.
About 2.7% of U.S. adults, or about 7 million adults, are considered immunocompromised, according to the CDC. They’re more likely to contract severe COVID-19, have a higher risk for long COVID, have lower antibody levels after vaccination, and develop serious breakthrough infections. About 40% of hospitalized breakthrough cases are among immunocompromised people.
According to CDC guidance, people are considered to be “moderately or severely immunocompromised” if they have:
- Active cancer treatment for tumors or cancers of the blood
- Had an organ transplant and are taking medicine to suppress the immune system
- Had a stem cell transplant in the last 2 years and are taking medicine to suppress the immune system
- Advanced or untreated HIV infection
- Moderate or severe primary immunodeficiency, such as DiGeorge syndrome or Wiskott-Aldrich syndrome
- Active treatment with high-dose corticosteroids or other drugs that suppress the immune response
So far, only moderately and severely immunocompromised Americans are eligible for a fourth shot. Israel has begun offering fourth doses to high-risk groups, including older adults, but the Biden administration hasn’t yet said whether the United States will follow, the Times reported.
Overall, the focus remains on getting third shots to Americans who are eligible for boosters, Rochelle Walensky, MD, the CDC director, told reporters Jan. 7. U.S. officials will remain in touch with Israel to follow their data on fourth shots.
“We will be following our own data carefully as well, to see how these boosters are working in terms of waning effectiveness, not just for infection but, importantly, for severe disease,” she said.
A version of this article first appeared on WebMD.com .
The Centers for Disease Control and Prevention endorsed a third dose of the Pfizer or Moderna vaccines for moderately and severely immunocompromised people on Aug. 13, which is considered part of their first immunization series rather than a booster shot.
In October, the CDC said moderately and severely immunocompromised people could receive a booster shot, or a fourth dose of the vaccine , 6 months after their third dose.
But the CDC last week shortened the timeline to 5 months for a booster shot of the Pfizer or Moderna vaccines. That means immunocompromised people could begin signing up for a fourth shot later this week, the New York Times reported.
About 2.7% of U.S. adults, or about 7 million adults, are considered immunocompromised, according to the CDC. They’re more likely to contract severe COVID-19, have a higher risk for long COVID, have lower antibody levels after vaccination, and develop serious breakthrough infections. About 40% of hospitalized breakthrough cases are among immunocompromised people.
According to CDC guidance, people are considered to be “moderately or severely immunocompromised” if they have:
- Active cancer treatment for tumors or cancers of the blood
- Had an organ transplant and are taking medicine to suppress the immune system
- Had a stem cell transplant in the last 2 years and are taking medicine to suppress the immune system
- Advanced or untreated HIV infection
- Moderate or severe primary immunodeficiency, such as DiGeorge syndrome or Wiskott-Aldrich syndrome
- Active treatment with high-dose corticosteroids or other drugs that suppress the immune response
So far, only moderately and severely immunocompromised Americans are eligible for a fourth shot. Israel has begun offering fourth doses to high-risk groups, including older adults, but the Biden administration hasn’t yet said whether the United States will follow, the Times reported.
Overall, the focus remains on getting third shots to Americans who are eligible for boosters, Rochelle Walensky, MD, the CDC director, told reporters Jan. 7. U.S. officials will remain in touch with Israel to follow their data on fourth shots.
“We will be following our own data carefully as well, to see how these boosters are working in terms of waning effectiveness, not just for infection but, importantly, for severe disease,” she said.
A version of this article first appeared on WebMD.com .
The Centers for Disease Control and Prevention endorsed a third dose of the Pfizer or Moderna vaccines for moderately and severely immunocompromised people on Aug. 13, which is considered part of their first immunization series rather than a booster shot.
In October, the CDC said moderately and severely immunocompromised people could receive a booster shot, or a fourth dose of the vaccine , 6 months after their third dose.
But the CDC last week shortened the timeline to 5 months for a booster shot of the Pfizer or Moderna vaccines. That means immunocompromised people could begin signing up for a fourth shot later this week, the New York Times reported.
About 2.7% of U.S. adults, or about 7 million adults, are considered immunocompromised, according to the CDC. They’re more likely to contract severe COVID-19, have a higher risk for long COVID, have lower antibody levels after vaccination, and develop serious breakthrough infections. About 40% of hospitalized breakthrough cases are among immunocompromised people.
According to CDC guidance, people are considered to be “moderately or severely immunocompromised” if they have:
- Active cancer treatment for tumors or cancers of the blood
- Had an organ transplant and are taking medicine to suppress the immune system
- Had a stem cell transplant in the last 2 years and are taking medicine to suppress the immune system
- Advanced or untreated HIV infection
- Moderate or severe primary immunodeficiency, such as DiGeorge syndrome or Wiskott-Aldrich syndrome
- Active treatment with high-dose corticosteroids or other drugs that suppress the immune response
So far, only moderately and severely immunocompromised Americans are eligible for a fourth shot. Israel has begun offering fourth doses to high-risk groups, including older adults, but the Biden administration hasn’t yet said whether the United States will follow, the Times reported.
Overall, the focus remains on getting third shots to Americans who are eligible for boosters, Rochelle Walensky, MD, the CDC director, told reporters Jan. 7. U.S. officials will remain in touch with Israel to follow their data on fourth shots.
“We will be following our own data carefully as well, to see how these boosters are working in terms of waning effectiveness, not just for infection but, importantly, for severe disease,” she said.
A version of this article first appeared on WebMD.com .

