User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Dracunculiasis – guinea worm disease – is close to eradication. But will we ever reach the finish line?
When in 1988 former U.S. President Jimmy Carter toured Denchira and Elevanyo, two villages near Accra, Ghana, he noticed a young woman who appeared to be cradling a baby. Carter approached her for a chat, but was stopped in his tracks by a disquieting sight.
“It was not a baby. It was her right breast, which was about a foot long, and it had a guinea worm coming out of its nipple,” Mr. Carter later recalled. During his tour of Ghana that year, Mr. Carter saw hundreds of people affected by the guinea worm, an infection known as dracunculiasis – a disease caused by the nematode parasite Dracunculus medinensis. It’s a condition that can cause fever, severe pain, and even permanent damage to affected limbs.
In the late 1980s the country reported as many as 180,000 cases of guinea worm disease per year. Across the globe, that number was a staggering 3.5 million. However, by 2020, the world was down to just 27 cases, all of them in Africa.
This enormous reduction in prevalence is a direct effect of campaigns by endemic countries assisted by organizations such as the Centers for Disease Control and Prevention, the World Health Organization, and the Carter Center (a not-for-profit founded in 1982 by Jimmy Carter), which have strived since the 1980s to eradicate dracunculiasis, hoping to make it the second human disease purposefully wiped off the face of Earth. (Smallpox was the first.)
“That’s an extraordinary public health achievement,” David Molyneux, PhD, parasitologist at the Liverpool School of Tropical Medicine, said in an interview. Yet the eradication goal, currently set for 2030, seems unlikely to be met. What’s more, some experts argue that chasing eradication may be altogether a misguided idea.
Humanity has known dracunculiasis for millennia. Well-preserved specimens of Dracunculus medinensis were discovered in Egyptian mummies, while some researchers claim that the Old Testament’s “fiery serpents” that descended upon the Israelites near the Red Sea were in fact guinea worms, as the parasite was endemic to the area in the past. Even the serpent coiled around the staff of Asclepius, the god of medicine, might have been a guinea worm, according to some historians.
This would make sense considering how the disease is treated. When an adult worm emerges through the skin, a painful and crippling occurrence, it is wound up around a stick or a piece of gauze, a little at a time, to slowly draw it out of the skin. As the worm can be over 3 feet long, this procedure may take weeks. What you end up with is a stick with a long, snake-like animal coiled around it. Asclepius’s staff.
The first step in the infection is when a person drinks water contaminated with copepods, or water fleas, which contain the larvae of Dracunculus medinensis. Next, the larvae are freed in the stomach and start migrating through the body, looking to mate. The fertilized female worm is the one that causes the debilitating symptoms.
About a year after the initial infection, the pregnant female worm looks for exit points from the body, usually through legs or feet, ready to release new larvae. If the unlucky sufferer steps into a pond or a river, the immature larvae escape into the water, where they are eaten by water fleas. “People are fetching water to drink, and they walk into the water thinking they can get cleaner water not along the edge,” Adam Weiss, MPH, director of the Carter Center’s Guinea Worm Eradication Program, said in an interview. The vicious cycle begins anew.
Dracunculiasis may not be a killer disease, but it is painful and disabling. A study on school attendance in Nigeria showed that in 1995 when guinea worm infection prevalence among schoolchildren was as high as 27.7%, it was responsible for almost all school absences. As the result of the infection, children were seen wandering and sitting around the village helplessly. If it was the parents who got infected, children stayed out of school to help around the home. The dracunculiasis’ impact on work and earning capacity is so profound, in fact, that in Mali the infliction is known as “the disease of the empty granary.”
When in 1986 the Carter Center took the reins of the global dracunculiasis eradication campaign, India was the only country with a national program to get rid of the disease. Yet, once other nations joined the struggle, the results rapidly became visible. By 1993, the American Journal of Tropical Medicine and Hygiene published a paper titled, “Dracunculiasis Eradication: Beginning of the End.” The cases plummeted from 3.5 million in 1986 to 221,000 in 1993 and 32,000 in 2003, then to a mere 22 cases in 2015. What worked was a combination of surveillance, education campaigns, safe water provision, and treating potentially contaminated water with a chemical called Abate, a potent larvicide.
Today, many endemic countries, from Chad and Ethiopia to Mali and South Sudan, follow similar procedures. First and foremost is the supply of clean drinking water. However, Mr. Weiss said, this is not a “silver bullet, given how people live.” Those who are seminomadic or otherwise take care of livestock often fetch water outside of the village, from ponds or rivers. This is why dracunculiasis eradication programs include handing out portable water filters, which can be worn around the neck.
But if you don’t know why you should filter water, in all likelihood you won’t do it – cloth filters distributed for home water purification sometimes ended up as decorations or sewn into wedding dresses. That’s why education is key, too. Poster campaigns, comic books, radio broadcasts, instructions by volunteers, even t-shirts with health messages slowly but surely did change behaviors.
Cash rewards for reporting cases of dracunculiasis, which can be as high as $100, also work well to boost surveillance systems. Once a case is identified, patients may be moved to a containment center, both to treat the wound and to prevent patients from spreading the disease. Local water sources, meanwhile, may be sprayed with Abate.
1995 was the first year set as a target date for the eradication of dracunculiasis. Yet the goal wasn’t met – even though the total number of cases did decline by 97%. Next goals followed: 2009, 2020, and now, finally, 2030. For well over a decade now the world has been down to a trickle of cases per year, but the numbers don’t seem to want to budge lower. Mr. Weiss calls it a “limbo period” – we are almost there, but not quite. The final push, it seems, may be the one that’s the most difficult, especially now that we have two further complications: increasing conflicts in some endemic areas and zoonotic transmission.
According to WHO, in places like the Democratic Republic of the Congo, Mali, South Sudan, and Sudan, insecurity “hinders eradication efforts.” Not only does this insecurity make it difficult for health workers to reach endemic areas, but wars and violence also displace people, pushing those infected with guinea worm to walk far distances in search of safety, and spreading the disease during their travels. Case containment and contact tracing become challenging. A recent study by Dr. Molyneux and colleagues showed that, in the 3 years since 2018, conflicts in the endemic areas have increased dramatically.
And then there are the animals. Up until 2012, eradication of guinea worm seemed fairly simple, at least from a biological perspective: Stop infected humans from contaminating drinking water and the parasites won’t be able to continue their life cycle. But in 2012, news came from Chad that a significant number of local dogs were found infected with the Dracunculus medinensis parasite, the very same one that attacks humans. In 2020, close to 1,600 dogs were reported to be infected with guinea worm, most of them in Chad. This left scientists scratching their heads: Dracunculiasis was supposed to be a purely human infliction. How were the dogs getting infected? Did the parasite jump to a new species because we were so efficient at eliminating it from humans?
“I have first seen a guinea worm transmission in dogs back in 2003,” Teshome Gebre, PhD, said in an interview. Dr. Gebre is regional director for Africa at International Trachoma Initiative and has spent more than 40 years fighting to eradicate various diseases, including smallpox and guinea worm. Yet in 2003, Dr. Gebre’s report was dismissed: it couldn’t have been the same species of the parasite, the reasoning went, since Dracunculus medinensis was exclusive to humans.
“I think it’s fair to say that there were infections in dogs before 2012. I find it difficult to believe, logically, that it just came out of nowhere,” Mr. Weiss said. A 2018 genetic study showed that a novel host switch is an unlikely scenario – the parasites must have been infecting dogs in the past, we just haven’t been looking. By 2012, Chad had a very efficient guinea worm surveillance system, with generous cash rewards for human cases, and people started reporting the dogs, too. Soon money was also offered for news on infected animals, and the cases exploded. This was then followed by accounts of afflicted cats and baboons.
To announce the eradication of dracunculiasis in 2030, the requirement will be no more transmission of the parasite for at least 4 years prior anywhere in the world – not only zero human cases, but also no infections in dogs, cats, or baboons. Seven countries remain to be certified as guinea worm free, all of them in Africa. “We have to be a 100% sure that there is no transmission of the parasite in a country,” said Dr. Molyneux, who participated in country certification teams – a rigorous process to validate country reports. He believes that the presence of animal hosts as well as growing insecurities in the region make such certification extremely challenging over the next few years.
“Eradication as it is defined does not seem feasible by 2030 as things stand, [considering] political and resource constraints, the unknowns of the ecology of dogs, and the possible impact of climate change and geopolitical instability and with countries having other health priorities, including COVID,” Dr. Molyneux said.
For Mr. Weiss, dogs are not that much of a problem – since they can be tethered to prevent the spread of the disease. But you can’t tether baboons. “That does raise that more existential threat–related question of: Is this scientifically possible?” he said. Mr. Weiss and colleagues at the Centers for Disease Control and Prevention are currently working on a serologic assay to test whether baboons are important for human transmission.
For some experts, such as Dr. Gebre, the current struggles to bring cases down to zero put a spotlight on a bigger question: is it worthwhile to strive for eradication at all? That last stretch of the eradication campaign can appear a bit like a game of whack-a-mole. “There were times when we’ve achieved zero cases [in Ethiopia]. Zero. And then, it just reemerges,” Dr. Gebre said. Programs aimed at certification are costly, running up to $1.6 million per year in Nigeria. The funds often come from the same donor pockets that pay for the fight against malaria, HIV, polio, as well as other neglected tropical diseases. Dr. Gebre believed it would be more cost and time efficient to switch the effort from total eradication to elimination as a public health care problem.
Of course, there is the risk that the cases would go up again once we ease up on the pressure to eradicate dracunculiasis. “Do we want to be fighting guinea worm in perpetuity?” Mr. Weiss asked. However, Dr. Gebre believed the cases are unlikely to explode anymore.
“The situation in the countries is not the way it was 30 years ago,” Dr. Gebre said, pointing out increased awareness, higher education levels, and better community-based health facilities. “You can cap it around a trickle number of cases a year – 10, 15, 20 maybe.”
The keys, Dr. Gebre and Dr. Molyneux both said, include the provision of safe drinking water and strengthening the healthcare systems of endemic countries in general, so they can deal with whatever cases may come up. “Water, sanitation, surveillance, good public education – and the maintenance of the guinea worm–specific reward system to maintain awareness, as well as continuing research” are all needed, Dr. Molyneux said.
Getting out of the dracunculiasis limbo period won’t be easy. We certainly need more data on animal transmission to better understand what challenges we might be facing. The experts agree that what’s important is to follow the science and stay flexible. “We have made an incredible progress, our investment has been worthwhile,” Dr. Molyneux said. But “you have to adapt to the changing realities.”
Dr. Gebre received no financial support for the review article and has no other conflicts of interest to declare. Dr. Molyneux is a member of the WHO International Commission for the Certification of Dracunculus Eradication, an independent body appointed by the director general of WHO. He acts as a rapporteur for the ICCDE as a paid consultant. He declared he does not receive any financial support for other related activities. Mr. Weiss receives support from the nonprofit Carter Center.
A version of this article first appeared on Medscape.com.
When in 1988 former U.S. President Jimmy Carter toured Denchira and Elevanyo, two villages near Accra, Ghana, he noticed a young woman who appeared to be cradling a baby. Carter approached her for a chat, but was stopped in his tracks by a disquieting sight.
“It was not a baby. It was her right breast, which was about a foot long, and it had a guinea worm coming out of its nipple,” Mr. Carter later recalled. During his tour of Ghana that year, Mr. Carter saw hundreds of people affected by the guinea worm, an infection known as dracunculiasis – a disease caused by the nematode parasite Dracunculus medinensis. It’s a condition that can cause fever, severe pain, and even permanent damage to affected limbs.
In the late 1980s the country reported as many as 180,000 cases of guinea worm disease per year. Across the globe, that number was a staggering 3.5 million. However, by 2020, the world was down to just 27 cases, all of them in Africa.
This enormous reduction in prevalence is a direct effect of campaigns by endemic countries assisted by organizations such as the Centers for Disease Control and Prevention, the World Health Organization, and the Carter Center (a not-for-profit founded in 1982 by Jimmy Carter), which have strived since the 1980s to eradicate dracunculiasis, hoping to make it the second human disease purposefully wiped off the face of Earth. (Smallpox was the first.)
“That’s an extraordinary public health achievement,” David Molyneux, PhD, parasitologist at the Liverpool School of Tropical Medicine, said in an interview. Yet the eradication goal, currently set for 2030, seems unlikely to be met. What’s more, some experts argue that chasing eradication may be altogether a misguided idea.
Humanity has known dracunculiasis for millennia. Well-preserved specimens of Dracunculus medinensis were discovered in Egyptian mummies, while some researchers claim that the Old Testament’s “fiery serpents” that descended upon the Israelites near the Red Sea were in fact guinea worms, as the parasite was endemic to the area in the past. Even the serpent coiled around the staff of Asclepius, the god of medicine, might have been a guinea worm, according to some historians.
This would make sense considering how the disease is treated. When an adult worm emerges through the skin, a painful and crippling occurrence, it is wound up around a stick or a piece of gauze, a little at a time, to slowly draw it out of the skin. As the worm can be over 3 feet long, this procedure may take weeks. What you end up with is a stick with a long, snake-like animal coiled around it. Asclepius’s staff.
The first step in the infection is when a person drinks water contaminated with copepods, or water fleas, which contain the larvae of Dracunculus medinensis. Next, the larvae are freed in the stomach and start migrating through the body, looking to mate. The fertilized female worm is the one that causes the debilitating symptoms.
About a year after the initial infection, the pregnant female worm looks for exit points from the body, usually through legs or feet, ready to release new larvae. If the unlucky sufferer steps into a pond or a river, the immature larvae escape into the water, where they are eaten by water fleas. “People are fetching water to drink, and they walk into the water thinking they can get cleaner water not along the edge,” Adam Weiss, MPH, director of the Carter Center’s Guinea Worm Eradication Program, said in an interview. The vicious cycle begins anew.
Dracunculiasis may not be a killer disease, but it is painful and disabling. A study on school attendance in Nigeria showed that in 1995 when guinea worm infection prevalence among schoolchildren was as high as 27.7%, it was responsible for almost all school absences. As the result of the infection, children were seen wandering and sitting around the village helplessly. If it was the parents who got infected, children stayed out of school to help around the home. The dracunculiasis’ impact on work and earning capacity is so profound, in fact, that in Mali the infliction is known as “the disease of the empty granary.”
When in 1986 the Carter Center took the reins of the global dracunculiasis eradication campaign, India was the only country with a national program to get rid of the disease. Yet, once other nations joined the struggle, the results rapidly became visible. By 1993, the American Journal of Tropical Medicine and Hygiene published a paper titled, “Dracunculiasis Eradication: Beginning of the End.” The cases plummeted from 3.5 million in 1986 to 221,000 in 1993 and 32,000 in 2003, then to a mere 22 cases in 2015. What worked was a combination of surveillance, education campaigns, safe water provision, and treating potentially contaminated water with a chemical called Abate, a potent larvicide.
Today, many endemic countries, from Chad and Ethiopia to Mali and South Sudan, follow similar procedures. First and foremost is the supply of clean drinking water. However, Mr. Weiss said, this is not a “silver bullet, given how people live.” Those who are seminomadic or otherwise take care of livestock often fetch water outside of the village, from ponds or rivers. This is why dracunculiasis eradication programs include handing out portable water filters, which can be worn around the neck.
But if you don’t know why you should filter water, in all likelihood you won’t do it – cloth filters distributed for home water purification sometimes ended up as decorations or sewn into wedding dresses. That’s why education is key, too. Poster campaigns, comic books, radio broadcasts, instructions by volunteers, even t-shirts with health messages slowly but surely did change behaviors.
Cash rewards for reporting cases of dracunculiasis, which can be as high as $100, also work well to boost surveillance systems. Once a case is identified, patients may be moved to a containment center, both to treat the wound and to prevent patients from spreading the disease. Local water sources, meanwhile, may be sprayed with Abate.
1995 was the first year set as a target date for the eradication of dracunculiasis. Yet the goal wasn’t met – even though the total number of cases did decline by 97%. Next goals followed: 2009, 2020, and now, finally, 2030. For well over a decade now the world has been down to a trickle of cases per year, but the numbers don’t seem to want to budge lower. Mr. Weiss calls it a “limbo period” – we are almost there, but not quite. The final push, it seems, may be the one that’s the most difficult, especially now that we have two further complications: increasing conflicts in some endemic areas and zoonotic transmission.
According to WHO, in places like the Democratic Republic of the Congo, Mali, South Sudan, and Sudan, insecurity “hinders eradication efforts.” Not only does this insecurity make it difficult for health workers to reach endemic areas, but wars and violence also displace people, pushing those infected with guinea worm to walk far distances in search of safety, and spreading the disease during their travels. Case containment and contact tracing become challenging. A recent study by Dr. Molyneux and colleagues showed that, in the 3 years since 2018, conflicts in the endemic areas have increased dramatically.
And then there are the animals. Up until 2012, eradication of guinea worm seemed fairly simple, at least from a biological perspective: Stop infected humans from contaminating drinking water and the parasites won’t be able to continue their life cycle. But in 2012, news came from Chad that a significant number of local dogs were found infected with the Dracunculus medinensis parasite, the very same one that attacks humans. In 2020, close to 1,600 dogs were reported to be infected with guinea worm, most of them in Chad. This left scientists scratching their heads: Dracunculiasis was supposed to be a purely human infliction. How were the dogs getting infected? Did the parasite jump to a new species because we were so efficient at eliminating it from humans?
“I have first seen a guinea worm transmission in dogs back in 2003,” Teshome Gebre, PhD, said in an interview. Dr. Gebre is regional director for Africa at International Trachoma Initiative and has spent more than 40 years fighting to eradicate various diseases, including smallpox and guinea worm. Yet in 2003, Dr. Gebre’s report was dismissed: it couldn’t have been the same species of the parasite, the reasoning went, since Dracunculus medinensis was exclusive to humans.
“I think it’s fair to say that there were infections in dogs before 2012. I find it difficult to believe, logically, that it just came out of nowhere,” Mr. Weiss said. A 2018 genetic study showed that a novel host switch is an unlikely scenario – the parasites must have been infecting dogs in the past, we just haven’t been looking. By 2012, Chad had a very efficient guinea worm surveillance system, with generous cash rewards for human cases, and people started reporting the dogs, too. Soon money was also offered for news on infected animals, and the cases exploded. This was then followed by accounts of afflicted cats and baboons.
To announce the eradication of dracunculiasis in 2030, the requirement will be no more transmission of the parasite for at least 4 years prior anywhere in the world – not only zero human cases, but also no infections in dogs, cats, or baboons. Seven countries remain to be certified as guinea worm free, all of them in Africa. “We have to be a 100% sure that there is no transmission of the parasite in a country,” said Dr. Molyneux, who participated in country certification teams – a rigorous process to validate country reports. He believes that the presence of animal hosts as well as growing insecurities in the region make such certification extremely challenging over the next few years.
“Eradication as it is defined does not seem feasible by 2030 as things stand, [considering] political and resource constraints, the unknowns of the ecology of dogs, and the possible impact of climate change and geopolitical instability and with countries having other health priorities, including COVID,” Dr. Molyneux said.
For Mr. Weiss, dogs are not that much of a problem – since they can be tethered to prevent the spread of the disease. But you can’t tether baboons. “That does raise that more existential threat–related question of: Is this scientifically possible?” he said. Mr. Weiss and colleagues at the Centers for Disease Control and Prevention are currently working on a serologic assay to test whether baboons are important for human transmission.
For some experts, such as Dr. Gebre, the current struggles to bring cases down to zero put a spotlight on a bigger question: is it worthwhile to strive for eradication at all? That last stretch of the eradication campaign can appear a bit like a game of whack-a-mole. “There were times when we’ve achieved zero cases [in Ethiopia]. Zero. And then, it just reemerges,” Dr. Gebre said. Programs aimed at certification are costly, running up to $1.6 million per year in Nigeria. The funds often come from the same donor pockets that pay for the fight against malaria, HIV, polio, as well as other neglected tropical diseases. Dr. Gebre believed it would be more cost and time efficient to switch the effort from total eradication to elimination as a public health care problem.
Of course, there is the risk that the cases would go up again once we ease up on the pressure to eradicate dracunculiasis. “Do we want to be fighting guinea worm in perpetuity?” Mr. Weiss asked. However, Dr. Gebre believed the cases are unlikely to explode anymore.
“The situation in the countries is not the way it was 30 years ago,” Dr. Gebre said, pointing out increased awareness, higher education levels, and better community-based health facilities. “You can cap it around a trickle number of cases a year – 10, 15, 20 maybe.”
The keys, Dr. Gebre and Dr. Molyneux both said, include the provision of safe drinking water and strengthening the healthcare systems of endemic countries in general, so they can deal with whatever cases may come up. “Water, sanitation, surveillance, good public education – and the maintenance of the guinea worm–specific reward system to maintain awareness, as well as continuing research” are all needed, Dr. Molyneux said.
Getting out of the dracunculiasis limbo period won’t be easy. We certainly need more data on animal transmission to better understand what challenges we might be facing. The experts agree that what’s important is to follow the science and stay flexible. “We have made an incredible progress, our investment has been worthwhile,” Dr. Molyneux said. But “you have to adapt to the changing realities.”
Dr. Gebre received no financial support for the review article and has no other conflicts of interest to declare. Dr. Molyneux is a member of the WHO International Commission for the Certification of Dracunculus Eradication, an independent body appointed by the director general of WHO. He acts as a rapporteur for the ICCDE as a paid consultant. He declared he does not receive any financial support for other related activities. Mr. Weiss receives support from the nonprofit Carter Center.
A version of this article first appeared on Medscape.com.
When in 1988 former U.S. President Jimmy Carter toured Denchira and Elevanyo, two villages near Accra, Ghana, he noticed a young woman who appeared to be cradling a baby. Carter approached her for a chat, but was stopped in his tracks by a disquieting sight.
“It was not a baby. It was her right breast, which was about a foot long, and it had a guinea worm coming out of its nipple,” Mr. Carter later recalled. During his tour of Ghana that year, Mr. Carter saw hundreds of people affected by the guinea worm, an infection known as dracunculiasis – a disease caused by the nematode parasite Dracunculus medinensis. It’s a condition that can cause fever, severe pain, and even permanent damage to affected limbs.
In the late 1980s the country reported as many as 180,000 cases of guinea worm disease per year. Across the globe, that number was a staggering 3.5 million. However, by 2020, the world was down to just 27 cases, all of them in Africa.
This enormous reduction in prevalence is a direct effect of campaigns by endemic countries assisted by organizations such as the Centers for Disease Control and Prevention, the World Health Organization, and the Carter Center (a not-for-profit founded in 1982 by Jimmy Carter), which have strived since the 1980s to eradicate dracunculiasis, hoping to make it the second human disease purposefully wiped off the face of Earth. (Smallpox was the first.)
“That’s an extraordinary public health achievement,” David Molyneux, PhD, parasitologist at the Liverpool School of Tropical Medicine, said in an interview. Yet the eradication goal, currently set for 2030, seems unlikely to be met. What’s more, some experts argue that chasing eradication may be altogether a misguided idea.
Humanity has known dracunculiasis for millennia. Well-preserved specimens of Dracunculus medinensis were discovered in Egyptian mummies, while some researchers claim that the Old Testament’s “fiery serpents” that descended upon the Israelites near the Red Sea were in fact guinea worms, as the parasite was endemic to the area in the past. Even the serpent coiled around the staff of Asclepius, the god of medicine, might have been a guinea worm, according to some historians.
This would make sense considering how the disease is treated. When an adult worm emerges through the skin, a painful and crippling occurrence, it is wound up around a stick or a piece of gauze, a little at a time, to slowly draw it out of the skin. As the worm can be over 3 feet long, this procedure may take weeks. What you end up with is a stick with a long, snake-like animal coiled around it. Asclepius’s staff.
The first step in the infection is when a person drinks water contaminated with copepods, or water fleas, which contain the larvae of Dracunculus medinensis. Next, the larvae are freed in the stomach and start migrating through the body, looking to mate. The fertilized female worm is the one that causes the debilitating symptoms.
About a year after the initial infection, the pregnant female worm looks for exit points from the body, usually through legs or feet, ready to release new larvae. If the unlucky sufferer steps into a pond or a river, the immature larvae escape into the water, where they are eaten by water fleas. “People are fetching water to drink, and they walk into the water thinking they can get cleaner water not along the edge,” Adam Weiss, MPH, director of the Carter Center’s Guinea Worm Eradication Program, said in an interview. The vicious cycle begins anew.
Dracunculiasis may not be a killer disease, but it is painful and disabling. A study on school attendance in Nigeria showed that in 1995 when guinea worm infection prevalence among schoolchildren was as high as 27.7%, it was responsible for almost all school absences. As the result of the infection, children were seen wandering and sitting around the village helplessly. If it was the parents who got infected, children stayed out of school to help around the home. The dracunculiasis’ impact on work and earning capacity is so profound, in fact, that in Mali the infliction is known as “the disease of the empty granary.”
When in 1986 the Carter Center took the reins of the global dracunculiasis eradication campaign, India was the only country with a national program to get rid of the disease. Yet, once other nations joined the struggle, the results rapidly became visible. By 1993, the American Journal of Tropical Medicine and Hygiene published a paper titled, “Dracunculiasis Eradication: Beginning of the End.” The cases plummeted from 3.5 million in 1986 to 221,000 in 1993 and 32,000 in 2003, then to a mere 22 cases in 2015. What worked was a combination of surveillance, education campaigns, safe water provision, and treating potentially contaminated water with a chemical called Abate, a potent larvicide.
Today, many endemic countries, from Chad and Ethiopia to Mali and South Sudan, follow similar procedures. First and foremost is the supply of clean drinking water. However, Mr. Weiss said, this is not a “silver bullet, given how people live.” Those who are seminomadic or otherwise take care of livestock often fetch water outside of the village, from ponds or rivers. This is why dracunculiasis eradication programs include handing out portable water filters, which can be worn around the neck.
But if you don’t know why you should filter water, in all likelihood you won’t do it – cloth filters distributed for home water purification sometimes ended up as decorations or sewn into wedding dresses. That’s why education is key, too. Poster campaigns, comic books, radio broadcasts, instructions by volunteers, even t-shirts with health messages slowly but surely did change behaviors.
Cash rewards for reporting cases of dracunculiasis, which can be as high as $100, also work well to boost surveillance systems. Once a case is identified, patients may be moved to a containment center, both to treat the wound and to prevent patients from spreading the disease. Local water sources, meanwhile, may be sprayed with Abate.
1995 was the first year set as a target date for the eradication of dracunculiasis. Yet the goal wasn’t met – even though the total number of cases did decline by 97%. Next goals followed: 2009, 2020, and now, finally, 2030. For well over a decade now the world has been down to a trickle of cases per year, but the numbers don’t seem to want to budge lower. Mr. Weiss calls it a “limbo period” – we are almost there, but not quite. The final push, it seems, may be the one that’s the most difficult, especially now that we have two further complications: increasing conflicts in some endemic areas and zoonotic transmission.
According to WHO, in places like the Democratic Republic of the Congo, Mali, South Sudan, and Sudan, insecurity “hinders eradication efforts.” Not only does this insecurity make it difficult for health workers to reach endemic areas, but wars and violence also displace people, pushing those infected with guinea worm to walk far distances in search of safety, and spreading the disease during their travels. Case containment and contact tracing become challenging. A recent study by Dr. Molyneux and colleagues showed that, in the 3 years since 2018, conflicts in the endemic areas have increased dramatically.
And then there are the animals. Up until 2012, eradication of guinea worm seemed fairly simple, at least from a biological perspective: Stop infected humans from contaminating drinking water and the parasites won’t be able to continue their life cycle. But in 2012, news came from Chad that a significant number of local dogs were found infected with the Dracunculus medinensis parasite, the very same one that attacks humans. In 2020, close to 1,600 dogs were reported to be infected with guinea worm, most of them in Chad. This left scientists scratching their heads: Dracunculiasis was supposed to be a purely human infliction. How were the dogs getting infected? Did the parasite jump to a new species because we were so efficient at eliminating it from humans?
“I have first seen a guinea worm transmission in dogs back in 2003,” Teshome Gebre, PhD, said in an interview. Dr. Gebre is regional director for Africa at International Trachoma Initiative and has spent more than 40 years fighting to eradicate various diseases, including smallpox and guinea worm. Yet in 2003, Dr. Gebre’s report was dismissed: it couldn’t have been the same species of the parasite, the reasoning went, since Dracunculus medinensis was exclusive to humans.
“I think it’s fair to say that there were infections in dogs before 2012. I find it difficult to believe, logically, that it just came out of nowhere,” Mr. Weiss said. A 2018 genetic study showed that a novel host switch is an unlikely scenario – the parasites must have been infecting dogs in the past, we just haven’t been looking. By 2012, Chad had a very efficient guinea worm surveillance system, with generous cash rewards for human cases, and people started reporting the dogs, too. Soon money was also offered for news on infected animals, and the cases exploded. This was then followed by accounts of afflicted cats and baboons.
To announce the eradication of dracunculiasis in 2030, the requirement will be no more transmission of the parasite for at least 4 years prior anywhere in the world – not only zero human cases, but also no infections in dogs, cats, or baboons. Seven countries remain to be certified as guinea worm free, all of them in Africa. “We have to be a 100% sure that there is no transmission of the parasite in a country,” said Dr. Molyneux, who participated in country certification teams – a rigorous process to validate country reports. He believes that the presence of animal hosts as well as growing insecurities in the region make such certification extremely challenging over the next few years.
“Eradication as it is defined does not seem feasible by 2030 as things stand, [considering] political and resource constraints, the unknowns of the ecology of dogs, and the possible impact of climate change and geopolitical instability and with countries having other health priorities, including COVID,” Dr. Molyneux said.
For Mr. Weiss, dogs are not that much of a problem – since they can be tethered to prevent the spread of the disease. But you can’t tether baboons. “That does raise that more existential threat–related question of: Is this scientifically possible?” he said. Mr. Weiss and colleagues at the Centers for Disease Control and Prevention are currently working on a serologic assay to test whether baboons are important for human transmission.
For some experts, such as Dr. Gebre, the current struggles to bring cases down to zero put a spotlight on a bigger question: is it worthwhile to strive for eradication at all? That last stretch of the eradication campaign can appear a bit like a game of whack-a-mole. “There were times when we’ve achieved zero cases [in Ethiopia]. Zero. And then, it just reemerges,” Dr. Gebre said. Programs aimed at certification are costly, running up to $1.6 million per year in Nigeria. The funds often come from the same donor pockets that pay for the fight against malaria, HIV, polio, as well as other neglected tropical diseases. Dr. Gebre believed it would be more cost and time efficient to switch the effort from total eradication to elimination as a public health care problem.
Of course, there is the risk that the cases would go up again once we ease up on the pressure to eradicate dracunculiasis. “Do we want to be fighting guinea worm in perpetuity?” Mr. Weiss asked. However, Dr. Gebre believed the cases are unlikely to explode anymore.
“The situation in the countries is not the way it was 30 years ago,” Dr. Gebre said, pointing out increased awareness, higher education levels, and better community-based health facilities. “You can cap it around a trickle number of cases a year – 10, 15, 20 maybe.”
The keys, Dr. Gebre and Dr. Molyneux both said, include the provision of safe drinking water and strengthening the healthcare systems of endemic countries in general, so they can deal with whatever cases may come up. “Water, sanitation, surveillance, good public education – and the maintenance of the guinea worm–specific reward system to maintain awareness, as well as continuing research” are all needed, Dr. Molyneux said.
Getting out of the dracunculiasis limbo period won’t be easy. We certainly need more data on animal transmission to better understand what challenges we might be facing. The experts agree that what’s important is to follow the science and stay flexible. “We have made an incredible progress, our investment has been worthwhile,” Dr. Molyneux said. But “you have to adapt to the changing realities.”
Dr. Gebre received no financial support for the review article and has no other conflicts of interest to declare. Dr. Molyneux is a member of the WHO International Commission for the Certification of Dracunculus Eradication, an independent body appointed by the director general of WHO. He acts as a rapporteur for the ICCDE as a paid consultant. He declared he does not receive any financial support for other related activities. Mr. Weiss receives support from the nonprofit Carter Center.
A version of this article first appeared on Medscape.com.
A very strange place to find a tooth
A nose for the tooth
Have you ever had a stuffy nose that just wouldn’t go away? Those irritating head colds have nothing on the stuffy nose a man in New York recently had to go through. A stuffy nose to top all stuffy noses. One stuffy nose to rule them all, as it were.
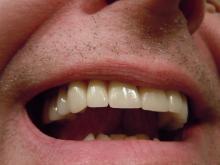
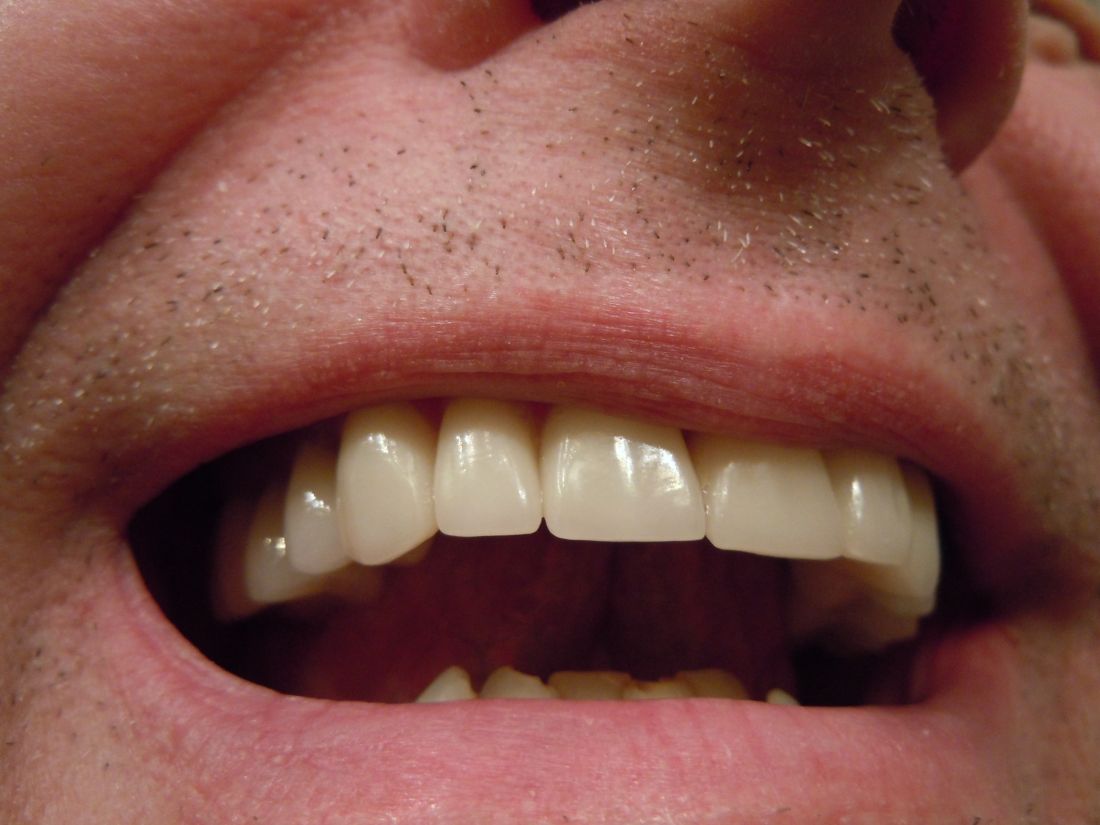
This man went to a Mount Sinai clinic with difficulty breathing through his right nostril, a problem that had been going on for years. Let us repeat that: A stuffy nose that lasted for years. The exam revealed a white mass jutting through the back of the septum and a CT scan confirmed the diagnosis. Perhaps you’ve already guessed, since the headline does give things away. Yes, this man had a tooth growing into his nose.
The problem was a half-inch-long ectopic tooth. Ectopic teeth are rare, occurring in less than 1% of people, but an ectopic tooth growing backward into the nasal cavity? Well, that’s so uncommon that this man got a case report in the New England Journal of Medicine.
This story does have a happy ending. Not all ectopic teeth need to be treated, but this one really did have to go. The offending tooth was surgically removed and, at a 3-month follow-up, the stuffy nose issue was completely resolved. So our friend gets the best of both worlds: His issue gets cured and he gets a case report in a major medical publication. If that’s not living the dream, we don’t know what is, and that’s the tooth.
Lettuce recommend you a sleep aid
Lettuce is great for many things. The star in a salad? Of course. The fresh element in a BLT? Yep. A sleep aid? According to a TikTok hack with almost 5 million views, the pinch hitter in a sandwich is switching leagues to be used like a tea for faster sleep. But, does it really work? Researchers say yes and no, according to a recent report at Tyla.com.
Studies conducted in 2013 and 2017 pointed toward a compound called lactucin, which is found in the plant’s n-butanol fraction. In the 2013 study, mice that received n-butanol fraction fell asleep faster and stayed asleep longer. In 2017, researchers found that lettuce made mice sleep longer and helped protect against cell inflammation and damage.
OK, so it works on mice. But what about humans? In the TikTok video, user Shapla Hoque pours hot water on a few lettuce leaves in a mug with a peppermint tea bag (for flavor). After 10 minutes, when the leaves are soaked and soggy, she removes them and drinks the lettuce tea. By the end of the video she’s visibly drowsy and ready to crash. Does this hold water?
Here’s the no. Dr. Charlotte Norton of the Slimming Clinic told Tyla.com that yeah, there are some properties in lettuce that will help you fall asleep, such as lactucarium, which is prominent in romaine. But you would need a massive amount of lettuce to get any effect. The TikTok video, she said, is an example of the placebo effect.
Brains get a rise out of Viagra
A lot of medications are used off label. Antidepressants for COVID have taken the cake recently, but here’s a new one: Viagra for Alzheimer’s disease.
Although there’s no definite link yet between the two, neuron models derived from induced pluripotent stem cells from patients with Alzheimer’s suggest that sildenafil increases neurite growth and decreases phospho-tau expression, Jiansong Fang, PhD, of the Cleveland Clinic, and associates said in Nature Aging.
Their research is an attempt to find untapped sources of new treatments among existing drugs. They began the search with 1,600 approved drugs and focused on those that target the buildup of beta amyloid and tau proteins in the brain, according to the Daily Beast.
Since sildenafil is obviously for men, more research will need to be done on how this drug affects women. Don’t start stocking up just yet.
Omicron is not a social-distancing robot
COVID, safe to say, has not been your typical, run-of-the-mill pandemic. People have protested social distancing. People have protested lockdowns. People have protested mask mandates. People have protested vaccine mandates. People have protested people protesting vaccine mandates.
Someone used a fake arm to get a COVID vaccine card. People have tried to reverse their COVID vaccinations. People had COVID contamination parties.
The common denominator? People. Humans. Maybe what we need is a nonhuman intervention. To fight COVID, we need a hero. A robotic hero.
And where can we find such a hero? The University of Maryland, of course, where computer scientists and engineers are working on an autonomous mobile robot to enforce indoor social-distancing rules.
Their robot can detect lapses in social distancing using cameras, both thermal and visual, along with a LiDAR (Light Detection and Ranging) sensor. It then sorts the offenders into various groups depending on whether they are standing still or moving and predicts their future movement using a state-of-the-art hybrid collision avoidance method known as Frozone, Adarsh Jagan Sathyamoorthy and associates explained in PLOS One.
“Once it reaches the breach, the robot encourages people to move apart via text that appears on a mounted display,” ScienceDaily said.
Maybe you were expecting a Terminator-type robot coming to enforce social distancing requirements rather than a simple text message. Let’s just hope that all COVID guidelines are followed, including social distancing, so the pandemic will finally end and won’t “be back.”
A nose for the tooth
Have you ever had a stuffy nose that just wouldn’t go away? Those irritating head colds have nothing on the stuffy nose a man in New York recently had to go through. A stuffy nose to top all stuffy noses. One stuffy nose to rule them all, as it were.
This man went to a Mount Sinai clinic with difficulty breathing through his right nostril, a problem that had been going on for years. Let us repeat that: A stuffy nose that lasted for years. The exam revealed a white mass jutting through the back of the septum and a CT scan confirmed the diagnosis. Perhaps you’ve already guessed, since the headline does give things away. Yes, this man had a tooth growing into his nose.
The problem was a half-inch-long ectopic tooth. Ectopic teeth are rare, occurring in less than 1% of people, but an ectopic tooth growing backward into the nasal cavity? Well, that’s so uncommon that this man got a case report in the New England Journal of Medicine.
This story does have a happy ending. Not all ectopic teeth need to be treated, but this one really did have to go. The offending tooth was surgically removed and, at a 3-month follow-up, the stuffy nose issue was completely resolved. So our friend gets the best of both worlds: His issue gets cured and he gets a case report in a major medical publication. If that’s not living the dream, we don’t know what is, and that’s the tooth.
Lettuce recommend you a sleep aid
Lettuce is great for many things. The star in a salad? Of course. The fresh element in a BLT? Yep. A sleep aid? According to a TikTok hack with almost 5 million views, the pinch hitter in a sandwich is switching leagues to be used like a tea for faster sleep. But, does it really work? Researchers say yes and no, according to a recent report at Tyla.com.
Studies conducted in 2013 and 2017 pointed toward a compound called lactucin, which is found in the plant’s n-butanol fraction. In the 2013 study, mice that received n-butanol fraction fell asleep faster and stayed asleep longer. In 2017, researchers found that lettuce made mice sleep longer and helped protect against cell inflammation and damage.
OK, so it works on mice. But what about humans? In the TikTok video, user Shapla Hoque pours hot water on a few lettuce leaves in a mug with a peppermint tea bag (for flavor). After 10 minutes, when the leaves are soaked and soggy, she removes them and drinks the lettuce tea. By the end of the video she’s visibly drowsy and ready to crash. Does this hold water?
Here’s the no. Dr. Charlotte Norton of the Slimming Clinic told Tyla.com that yeah, there are some properties in lettuce that will help you fall asleep, such as lactucarium, which is prominent in romaine. But you would need a massive amount of lettuce to get any effect. The TikTok video, she said, is an example of the placebo effect.
Brains get a rise out of Viagra
A lot of medications are used off label. Antidepressants for COVID have taken the cake recently, but here’s a new one: Viagra for Alzheimer’s disease.
Although there’s no definite link yet between the two, neuron models derived from induced pluripotent stem cells from patients with Alzheimer’s suggest that sildenafil increases neurite growth and decreases phospho-tau expression, Jiansong Fang, PhD, of the Cleveland Clinic, and associates said in Nature Aging.
Their research is an attempt to find untapped sources of new treatments among existing drugs. They began the search with 1,600 approved drugs and focused on those that target the buildup of beta amyloid and tau proteins in the brain, according to the Daily Beast.
Since sildenafil is obviously for men, more research will need to be done on how this drug affects women. Don’t start stocking up just yet.
Omicron is not a social-distancing robot
COVID, safe to say, has not been your typical, run-of-the-mill pandemic. People have protested social distancing. People have protested lockdowns. People have protested mask mandates. People have protested vaccine mandates. People have protested people protesting vaccine mandates.
Someone used a fake arm to get a COVID vaccine card. People have tried to reverse their COVID vaccinations. People had COVID contamination parties.
The common denominator? People. Humans. Maybe what we need is a nonhuman intervention. To fight COVID, we need a hero. A robotic hero.
And where can we find such a hero? The University of Maryland, of course, where computer scientists and engineers are working on an autonomous mobile robot to enforce indoor social-distancing rules.
Their robot can detect lapses in social distancing using cameras, both thermal and visual, along with a LiDAR (Light Detection and Ranging) sensor. It then sorts the offenders into various groups depending on whether they are standing still or moving and predicts their future movement using a state-of-the-art hybrid collision avoidance method known as Frozone, Adarsh Jagan Sathyamoorthy and associates explained in PLOS One.
“Once it reaches the breach, the robot encourages people to move apart via text that appears on a mounted display,” ScienceDaily said.
Maybe you were expecting a Terminator-type robot coming to enforce social distancing requirements rather than a simple text message. Let’s just hope that all COVID guidelines are followed, including social distancing, so the pandemic will finally end and won’t “be back.”
A nose for the tooth
Have you ever had a stuffy nose that just wouldn’t go away? Those irritating head colds have nothing on the stuffy nose a man in New York recently had to go through. A stuffy nose to top all stuffy noses. One stuffy nose to rule them all, as it were.
This man went to a Mount Sinai clinic with difficulty breathing through his right nostril, a problem that had been going on for years. Let us repeat that: A stuffy nose that lasted for years. The exam revealed a white mass jutting through the back of the septum and a CT scan confirmed the diagnosis. Perhaps you’ve already guessed, since the headline does give things away. Yes, this man had a tooth growing into his nose.
The problem was a half-inch-long ectopic tooth. Ectopic teeth are rare, occurring in less than 1% of people, but an ectopic tooth growing backward into the nasal cavity? Well, that’s so uncommon that this man got a case report in the New England Journal of Medicine.
This story does have a happy ending. Not all ectopic teeth need to be treated, but this one really did have to go. The offending tooth was surgically removed and, at a 3-month follow-up, the stuffy nose issue was completely resolved. So our friend gets the best of both worlds: His issue gets cured and he gets a case report in a major medical publication. If that’s not living the dream, we don’t know what is, and that’s the tooth.
Lettuce recommend you a sleep aid
Lettuce is great for many things. The star in a salad? Of course. The fresh element in a BLT? Yep. A sleep aid? According to a TikTok hack with almost 5 million views, the pinch hitter in a sandwich is switching leagues to be used like a tea for faster sleep. But, does it really work? Researchers say yes and no, according to a recent report at Tyla.com.
Studies conducted in 2013 and 2017 pointed toward a compound called lactucin, which is found in the plant’s n-butanol fraction. In the 2013 study, mice that received n-butanol fraction fell asleep faster and stayed asleep longer. In 2017, researchers found that lettuce made mice sleep longer and helped protect against cell inflammation and damage.
OK, so it works on mice. But what about humans? In the TikTok video, user Shapla Hoque pours hot water on a few lettuce leaves in a mug with a peppermint tea bag (for flavor). After 10 minutes, when the leaves are soaked and soggy, she removes them and drinks the lettuce tea. By the end of the video she’s visibly drowsy and ready to crash. Does this hold water?
Here’s the no. Dr. Charlotte Norton of the Slimming Clinic told Tyla.com that yeah, there are some properties in lettuce that will help you fall asleep, such as lactucarium, which is prominent in romaine. But you would need a massive amount of lettuce to get any effect. The TikTok video, she said, is an example of the placebo effect.
Brains get a rise out of Viagra
A lot of medications are used off label. Antidepressants for COVID have taken the cake recently, but here’s a new one: Viagra for Alzheimer’s disease.
Although there’s no definite link yet between the two, neuron models derived from induced pluripotent stem cells from patients with Alzheimer’s suggest that sildenafil increases neurite growth and decreases phospho-tau expression, Jiansong Fang, PhD, of the Cleveland Clinic, and associates said in Nature Aging.
Their research is an attempt to find untapped sources of new treatments among existing drugs. They began the search with 1,600 approved drugs and focused on those that target the buildup of beta amyloid and tau proteins in the brain, according to the Daily Beast.
Since sildenafil is obviously for men, more research will need to be done on how this drug affects women. Don’t start stocking up just yet.
Omicron is not a social-distancing robot
COVID, safe to say, has not been your typical, run-of-the-mill pandemic. People have protested social distancing. People have protested lockdowns. People have protested mask mandates. People have protested vaccine mandates. People have protested people protesting vaccine mandates.
Someone used a fake arm to get a COVID vaccine card. People have tried to reverse their COVID vaccinations. People had COVID contamination parties.
The common denominator? People. Humans. Maybe what we need is a nonhuman intervention. To fight COVID, we need a hero. A robotic hero.
And where can we find such a hero? The University of Maryland, of course, where computer scientists and engineers are working on an autonomous mobile robot to enforce indoor social-distancing rules.
Their robot can detect lapses in social distancing using cameras, both thermal and visual, along with a LiDAR (Light Detection and Ranging) sensor. It then sorts the offenders into various groups depending on whether they are standing still or moving and predicts their future movement using a state-of-the-art hybrid collision avoidance method known as Frozone, Adarsh Jagan Sathyamoorthy and associates explained in PLOS One.
“Once it reaches the breach, the robot encourages people to move apart via text that appears on a mounted display,” ScienceDaily said.
Maybe you were expecting a Terminator-type robot coming to enforce social distancing requirements rather than a simple text message. Let’s just hope that all COVID guidelines are followed, including social distancing, so the pandemic will finally end and won’t “be back.”
Bulevirtide shows real-world efficacy versus HDV
A real-world analysis of bulevirtide found a safety and efficacy profile similar to what was seen in earlier clinical trials in the treatment of hepatitis delta virus (HDV) infection.
HDV can only infect patients already carrying hepatitis B virus (HBV), but it causes the most severe form of viral hepatitis as it can progress to cirrhosis within 5 years and to hepatocellular carcinoma within 10 years.
Bulevirtide is a first-in-class medication that mimics the hepatitis B surface antigen, binding to its receptor on hepatocytes and preventing HDV viral particles from binding to it. The drug received conditional marketing approval by the European Medicines Agency in 2020 and has received a breakthrough therapy designation from the U.S. Food and Drug Administration.
The study was presented at the annual meeting of the American Association for the Study of Liver Diseases by Victor De Ledinghen, PhD, who is a professor of hepatology and head of the hepatology and liver transplantation unit at Bordeaux (France) University Hospital.
The early-access program launched after the French National Agency for Medicines and Health Products approved bulevirtide in 2019. It was made available to patients with compensated cirrhosis or severe liver fibrosis (F3) or patients with F2 fibrosis and alanine amino transferase levels more than twice the upper limit of normal for 6 months or more. Patients received bulevirtide alone (n = 77) or in combination with peg-interferon (n = 68), as determined by their physician.
The researchers defined virologic efficacy as HDV RNA levels being undetectable, or decreased by at least 2 log10 from baseline. They defined biochemical efficacy as ALT levels below 40 IU/L.
A per-protocol analysis included all patients in the bulevirtide group, but excluded 12 from the combination group who discontinued peg-interferon (n = 56). Nineteen patients in bulevirtide group had a treatment modification, and seven discontinued treatment. Five in the combination group had a treatment modification, and 14 stopped treatment. At 12 months, there was a greater decline in median log10 IU/mL in the combination group (–5.65 versus –3.64), though the study was not powered to compare the two. At 12 months, the combination group had 93.9% virologic efficacy, compared with 68.3% in the bulevirtide group.
The two groups had similar mean ALT levels at 12 months (48.91 and 48.03 IU/mL, respectively), with more patients in the bulevirtide group having normal ALT levels (<40 IU/L; 48.8% versus 36.4%). At 12 months, 39.0% of the bulevirtide group and 30.3% of the combination group had a combined response, defined as either undetectable HDV RNA or ≥2 log10 from baseline plus normal ALT levels.
Twenty-nine patients in the bulevirtide group had an adverse event, compared with 43 in the combination group. The two groups were similar in the frequency of grade 3-4 adverse events (7 versus 6), discontinuation due to adverse events (2 versus 3), deaths (0 in both), injection-site reactions (2 in both), liver-related adverse events (4 versus 2), and elevated bile acid (76 versus 68).
During the Q&A period following the presentation, Dr. De Ledinghen was asked if he has a preferred regimen for HDV patients. “I think it depends on the tolerance of peg-interferon because of all the side effects with this drug. I think we need to have predictive factors of virological response with or without interferon. At this time, I don’t have a preference, but I think at this time we need to work on predictive factors associated with virologic response,” he said.
The EMA’s conditional bulevirtide approval hinged on results from phase 2 clinical trials, while the phase 3 clinical studies are ongoing. “This was a very unusual step for the EMA to provide what is similar to emergency use approval while the phase 3 clinical trials are still ongoing,” said Anna Lok, MD, who was asked to comment on the study. Dr. Lok is a professor of internal medicine, director of clinical hepatology, and assistant dean for clinical research at the University of Michigan, Ann Arbor.
She noted that the phase 2 studies indicated that the combination with peg-interferon seems to have an additive effect on HDV suppression, while monotherapy with bulevirtide has a greater effect on normalizing ALT levels. The real-world experience confirms these findings.
But the real-world data revealed some concerns. “What really worried me is the large number of patients who required dose modifications or discontinuations, and that seems to be the case in both treatment groups. They didn’t really go into a lot of details [about] why patients needed treatment modifications, but one has to assume that this is due to side effects,” said Dr. Lok.
She also noted that the per-protocol analysis, instead of an intention-to-treat analysis, is a weakness of the study. Additionally, over time, the number of patients analyzed decreased – as many as 40% of patients didn’t have test results at month 12. “It makes you wonder what happened to those patients. Many probably didn’t respond, in which case your overall response rate will be far lower,” said Dr. Lok.
The study was funded by Gilead. Dr. De Ledinghen has financial relationships with Gilead, AbbVie, Echosens, Hologic, Intercept Pharma, Tillotts, Orphalan, Alfasigma, Bristol Myers Squibb, and Siemens Healthineers. Dr. Lok has no relevant financial disclosures.
A real-world analysis of bulevirtide found a safety and efficacy profile similar to what was seen in earlier clinical trials in the treatment of hepatitis delta virus (HDV) infection.
HDV can only infect patients already carrying hepatitis B virus (HBV), but it causes the most severe form of viral hepatitis as it can progress to cirrhosis within 5 years and to hepatocellular carcinoma within 10 years.
Bulevirtide is a first-in-class medication that mimics the hepatitis B surface antigen, binding to its receptor on hepatocytes and preventing HDV viral particles from binding to it. The drug received conditional marketing approval by the European Medicines Agency in 2020 and has received a breakthrough therapy designation from the U.S. Food and Drug Administration.
The study was presented at the annual meeting of the American Association for the Study of Liver Diseases by Victor De Ledinghen, PhD, who is a professor of hepatology and head of the hepatology and liver transplantation unit at Bordeaux (France) University Hospital.
The early-access program launched after the French National Agency for Medicines and Health Products approved bulevirtide in 2019. It was made available to patients with compensated cirrhosis or severe liver fibrosis (F3) or patients with F2 fibrosis and alanine amino transferase levels more than twice the upper limit of normal for 6 months or more. Patients received bulevirtide alone (n = 77) or in combination with peg-interferon (n = 68), as determined by their physician.
The researchers defined virologic efficacy as HDV RNA levels being undetectable, or decreased by at least 2 log10 from baseline. They defined biochemical efficacy as ALT levels below 40 IU/L.
A per-protocol analysis included all patients in the bulevirtide group, but excluded 12 from the combination group who discontinued peg-interferon (n = 56). Nineteen patients in bulevirtide group had a treatment modification, and seven discontinued treatment. Five in the combination group had a treatment modification, and 14 stopped treatment. At 12 months, there was a greater decline in median log10 IU/mL in the combination group (–5.65 versus –3.64), though the study was not powered to compare the two. At 12 months, the combination group had 93.9% virologic efficacy, compared with 68.3% in the bulevirtide group.
The two groups had similar mean ALT levels at 12 months (48.91 and 48.03 IU/mL, respectively), with more patients in the bulevirtide group having normal ALT levels (<40 IU/L; 48.8% versus 36.4%). At 12 months, 39.0% of the bulevirtide group and 30.3% of the combination group had a combined response, defined as either undetectable HDV RNA or ≥2 log10 from baseline plus normal ALT levels.
Twenty-nine patients in the bulevirtide group had an adverse event, compared with 43 in the combination group. The two groups were similar in the frequency of grade 3-4 adverse events (7 versus 6), discontinuation due to adverse events (2 versus 3), deaths (0 in both), injection-site reactions (2 in both), liver-related adverse events (4 versus 2), and elevated bile acid (76 versus 68).
During the Q&A period following the presentation, Dr. De Ledinghen was asked if he has a preferred regimen for HDV patients. “I think it depends on the tolerance of peg-interferon because of all the side effects with this drug. I think we need to have predictive factors of virological response with or without interferon. At this time, I don’t have a preference, but I think at this time we need to work on predictive factors associated with virologic response,” he said.
The EMA’s conditional bulevirtide approval hinged on results from phase 2 clinical trials, while the phase 3 clinical studies are ongoing. “This was a very unusual step for the EMA to provide what is similar to emergency use approval while the phase 3 clinical trials are still ongoing,” said Anna Lok, MD, who was asked to comment on the study. Dr. Lok is a professor of internal medicine, director of clinical hepatology, and assistant dean for clinical research at the University of Michigan, Ann Arbor.
She noted that the phase 2 studies indicated that the combination with peg-interferon seems to have an additive effect on HDV suppression, while monotherapy with bulevirtide has a greater effect on normalizing ALT levels. The real-world experience confirms these findings.
But the real-world data revealed some concerns. “What really worried me is the large number of patients who required dose modifications or discontinuations, and that seems to be the case in both treatment groups. They didn’t really go into a lot of details [about] why patients needed treatment modifications, but one has to assume that this is due to side effects,” said Dr. Lok.
She also noted that the per-protocol analysis, instead of an intention-to-treat analysis, is a weakness of the study. Additionally, over time, the number of patients analyzed decreased – as many as 40% of patients didn’t have test results at month 12. “It makes you wonder what happened to those patients. Many probably didn’t respond, in which case your overall response rate will be far lower,” said Dr. Lok.
The study was funded by Gilead. Dr. De Ledinghen has financial relationships with Gilead, AbbVie, Echosens, Hologic, Intercept Pharma, Tillotts, Orphalan, Alfasigma, Bristol Myers Squibb, and Siemens Healthineers. Dr. Lok has no relevant financial disclosures.
A real-world analysis of bulevirtide found a safety and efficacy profile similar to what was seen in earlier clinical trials in the treatment of hepatitis delta virus (HDV) infection.
HDV can only infect patients already carrying hepatitis B virus (HBV), but it causes the most severe form of viral hepatitis as it can progress to cirrhosis within 5 years and to hepatocellular carcinoma within 10 years.
Bulevirtide is a first-in-class medication that mimics the hepatitis B surface antigen, binding to its receptor on hepatocytes and preventing HDV viral particles from binding to it. The drug received conditional marketing approval by the European Medicines Agency in 2020 and has received a breakthrough therapy designation from the U.S. Food and Drug Administration.
The study was presented at the annual meeting of the American Association for the Study of Liver Diseases by Victor De Ledinghen, PhD, who is a professor of hepatology and head of the hepatology and liver transplantation unit at Bordeaux (France) University Hospital.
The early-access program launched after the French National Agency for Medicines and Health Products approved bulevirtide in 2019. It was made available to patients with compensated cirrhosis or severe liver fibrosis (F3) or patients with F2 fibrosis and alanine amino transferase levels more than twice the upper limit of normal for 6 months or more. Patients received bulevirtide alone (n = 77) or in combination with peg-interferon (n = 68), as determined by their physician.
The researchers defined virologic efficacy as HDV RNA levels being undetectable, or decreased by at least 2 log10 from baseline. They defined biochemical efficacy as ALT levels below 40 IU/L.
A per-protocol analysis included all patients in the bulevirtide group, but excluded 12 from the combination group who discontinued peg-interferon (n = 56). Nineteen patients in bulevirtide group had a treatment modification, and seven discontinued treatment. Five in the combination group had a treatment modification, and 14 stopped treatment. At 12 months, there was a greater decline in median log10 IU/mL in the combination group (–5.65 versus –3.64), though the study was not powered to compare the two. At 12 months, the combination group had 93.9% virologic efficacy, compared with 68.3% in the bulevirtide group.
The two groups had similar mean ALT levels at 12 months (48.91 and 48.03 IU/mL, respectively), with more patients in the bulevirtide group having normal ALT levels (<40 IU/L; 48.8% versus 36.4%). At 12 months, 39.0% of the bulevirtide group and 30.3% of the combination group had a combined response, defined as either undetectable HDV RNA or ≥2 log10 from baseline plus normal ALT levels.
Twenty-nine patients in the bulevirtide group had an adverse event, compared with 43 in the combination group. The two groups were similar in the frequency of grade 3-4 adverse events (7 versus 6), discontinuation due to adverse events (2 versus 3), deaths (0 in both), injection-site reactions (2 in both), liver-related adverse events (4 versus 2), and elevated bile acid (76 versus 68).
During the Q&A period following the presentation, Dr. De Ledinghen was asked if he has a preferred regimen for HDV patients. “I think it depends on the tolerance of peg-interferon because of all the side effects with this drug. I think we need to have predictive factors of virological response with or without interferon. At this time, I don’t have a preference, but I think at this time we need to work on predictive factors associated with virologic response,” he said.
The EMA’s conditional bulevirtide approval hinged on results from phase 2 clinical trials, while the phase 3 clinical studies are ongoing. “This was a very unusual step for the EMA to provide what is similar to emergency use approval while the phase 3 clinical trials are still ongoing,” said Anna Lok, MD, who was asked to comment on the study. Dr. Lok is a professor of internal medicine, director of clinical hepatology, and assistant dean for clinical research at the University of Michigan, Ann Arbor.
She noted that the phase 2 studies indicated that the combination with peg-interferon seems to have an additive effect on HDV suppression, while monotherapy with bulevirtide has a greater effect on normalizing ALT levels. The real-world experience confirms these findings.
But the real-world data revealed some concerns. “What really worried me is the large number of patients who required dose modifications or discontinuations, and that seems to be the case in both treatment groups. They didn’t really go into a lot of details [about] why patients needed treatment modifications, but one has to assume that this is due to side effects,” said Dr. Lok.
She also noted that the per-protocol analysis, instead of an intention-to-treat analysis, is a weakness of the study. Additionally, over time, the number of patients analyzed decreased – as many as 40% of patients didn’t have test results at month 12. “It makes you wonder what happened to those patients. Many probably didn’t respond, in which case your overall response rate will be far lower,” said Dr. Lok.
The study was funded by Gilead. Dr. De Ledinghen has financial relationships with Gilead, AbbVie, Echosens, Hologic, Intercept Pharma, Tillotts, Orphalan, Alfasigma, Bristol Myers Squibb, and Siemens Healthineers. Dr. Lok has no relevant financial disclosures.
FROM THE LIVER MEETING
New data on rare myocarditis after COVID-19 vaccination
Adolescents and adults younger than age 21 who develop myocarditis after mRNA COVID-19 vaccination frequently have abnormal findings on cardiac MRI (cMRI) but most have a mild clinical course with rapid resolution of symptoms, a new study concludes.
“This study supports what we’ve been seeing. People identified and treated early and appropriately for the rare complication of COVID-19 vaccine-related myocarditis typically experienced only mild cases and short recovery times,” American Heart Association President Donald M. Lloyd-Jones, MD, said in a podcast.
“Overwhelmingly, the data continue to indicate [that] the benefits of COVID-19 vaccine far outweigh any very rare risks of adverse events from the vaccine, including myocarditis,” Dr. Lloyd-Jones added.
The study was published online Dec. 6 in Circulation.
Using data from 26 pediatric medical centers across the United States and Canada, the researchers reviewed the medical records of 139 patients younger than 21 with suspected myocarditis within 1 month of receiving a COVID-19 vaccination.
They made the following key observations:
- Most patients were male (90.6%), White (66.2%) and with a median age of 15.8 years.
- Suspected myocarditis occurred in 136 patients (97.8%) following mRNA vaccine, with 131 (94.2%) following the Pfizer-BioNTech vaccine; 128 cases (91.4%) occurred after the second dose.
- Symptoms started a median of 2 days (range 0 to 22 days) following vaccination administration.
- Chest pain was the most common symptom (99.3%), with fever present in 30.9% of patients and shortness of breath in 27.3%.
- Patients were treated with nonsteroidal anti-inflammatory drugs (81.3%), intravenous immunoglobulin (21.6%), glucocorticoids (21.6%), colchicine (7.9%) or no anti-inflammatory therapies (8.6%).
- Twenty-six patients (18.7%) were admitted to the intensive care unit; 2 received inotropic/vasoactive support; none required extracorporeal membrane oxygenation or died.
- Median time spent in the hospital was 2 days.
- A total of 111 patients had elevated troponin I (8.12 ng/mL) and 28 had elevated troponin T (0.61 ng/mL).
- More than two-thirds (69.8%) had abnormal electrocardiograms and/or arrhythmias (7 with nonsustained ventricular tachycardia).
- Twenty-six patients (18.7%) had left ventricular ejection fraction (LVEF) less than 55% on echocardiogram; LVEF had returned to normal in the 25 who returned for follow-up.
- 75 of 97 patients (77.3%) who underwent cMRI at a median of 5 days from symptom onset had abnormal findings; 74 (76.3%) had late gadolinium enhancement, 54 (55.7%) had myocardial edema, and 49 (50.5%) met Lake Louise criteria for myocarditis.
“These data suggest that most cases of suspected COVID-19 vaccine–related myocarditis in people younger than 21 are mild and resolve quickly,” corresponding author Dongngan Truong, MD, Division of Pediatric Cardiology, University of Utah and Primary Children’s Hospital, Salt Lake City, said in a statement.
“We were very happy to see that type of recovery. However, we are awaiting further studies to better understand the long-term outcomes of patients who have had COVID-19 vaccination-related myocarditis. We also need to study the risk factors and mechanisms for this rare complication,” Dr. Truong added.
Dr. Lloyd-Jones said these findings support the AHA’s position that COVID-19 vaccines are “safe, highly effective, and fundamental to saving lives, protecting our families and communities against COVID-19, and ending the pandemic.”
The study received no funding. Dr. Truong consults for Pfizer on vaccine-associated myocarditis. A complete list of author disclosures is available with the original article.
A version of this article first appeared on Medscape.com.
Adolescents and adults younger than age 21 who develop myocarditis after mRNA COVID-19 vaccination frequently have abnormal findings on cardiac MRI (cMRI) but most have a mild clinical course with rapid resolution of symptoms, a new study concludes.
“This study supports what we’ve been seeing. People identified and treated early and appropriately for the rare complication of COVID-19 vaccine-related myocarditis typically experienced only mild cases and short recovery times,” American Heart Association President Donald M. Lloyd-Jones, MD, said in a podcast.
“Overwhelmingly, the data continue to indicate [that] the benefits of COVID-19 vaccine far outweigh any very rare risks of adverse events from the vaccine, including myocarditis,” Dr. Lloyd-Jones added.
The study was published online Dec. 6 in Circulation.
Using data from 26 pediatric medical centers across the United States and Canada, the researchers reviewed the medical records of 139 patients younger than 21 with suspected myocarditis within 1 month of receiving a COVID-19 vaccination.
They made the following key observations:
- Most patients were male (90.6%), White (66.2%) and with a median age of 15.8 years.
- Suspected myocarditis occurred in 136 patients (97.8%) following mRNA vaccine, with 131 (94.2%) following the Pfizer-BioNTech vaccine; 128 cases (91.4%) occurred after the second dose.
- Symptoms started a median of 2 days (range 0 to 22 days) following vaccination administration.
- Chest pain was the most common symptom (99.3%), with fever present in 30.9% of patients and shortness of breath in 27.3%.
- Patients were treated with nonsteroidal anti-inflammatory drugs (81.3%), intravenous immunoglobulin (21.6%), glucocorticoids (21.6%), colchicine (7.9%) or no anti-inflammatory therapies (8.6%).
- Twenty-six patients (18.7%) were admitted to the intensive care unit; 2 received inotropic/vasoactive support; none required extracorporeal membrane oxygenation or died.
- Median time spent in the hospital was 2 days.
- A total of 111 patients had elevated troponin I (8.12 ng/mL) and 28 had elevated troponin T (0.61 ng/mL).
- More than two-thirds (69.8%) had abnormal electrocardiograms and/or arrhythmias (7 with nonsustained ventricular tachycardia).
- Twenty-six patients (18.7%) had left ventricular ejection fraction (LVEF) less than 55% on echocardiogram; LVEF had returned to normal in the 25 who returned for follow-up.
- 75 of 97 patients (77.3%) who underwent cMRI at a median of 5 days from symptom onset had abnormal findings; 74 (76.3%) had late gadolinium enhancement, 54 (55.7%) had myocardial edema, and 49 (50.5%) met Lake Louise criteria for myocarditis.
“These data suggest that most cases of suspected COVID-19 vaccine–related myocarditis in people younger than 21 are mild and resolve quickly,” corresponding author Dongngan Truong, MD, Division of Pediatric Cardiology, University of Utah and Primary Children’s Hospital, Salt Lake City, said in a statement.
“We were very happy to see that type of recovery. However, we are awaiting further studies to better understand the long-term outcomes of patients who have had COVID-19 vaccination-related myocarditis. We also need to study the risk factors and mechanisms for this rare complication,” Dr. Truong added.
Dr. Lloyd-Jones said these findings support the AHA’s position that COVID-19 vaccines are “safe, highly effective, and fundamental to saving lives, protecting our families and communities against COVID-19, and ending the pandemic.”
The study received no funding. Dr. Truong consults for Pfizer on vaccine-associated myocarditis. A complete list of author disclosures is available with the original article.
A version of this article first appeared on Medscape.com.
Adolescents and adults younger than age 21 who develop myocarditis after mRNA COVID-19 vaccination frequently have abnormal findings on cardiac MRI (cMRI) but most have a mild clinical course with rapid resolution of symptoms, a new study concludes.
“This study supports what we’ve been seeing. People identified and treated early and appropriately for the rare complication of COVID-19 vaccine-related myocarditis typically experienced only mild cases and short recovery times,” American Heart Association President Donald M. Lloyd-Jones, MD, said in a podcast.
“Overwhelmingly, the data continue to indicate [that] the benefits of COVID-19 vaccine far outweigh any very rare risks of adverse events from the vaccine, including myocarditis,” Dr. Lloyd-Jones added.
The study was published online Dec. 6 in Circulation.
Using data from 26 pediatric medical centers across the United States and Canada, the researchers reviewed the medical records of 139 patients younger than 21 with suspected myocarditis within 1 month of receiving a COVID-19 vaccination.
They made the following key observations:
- Most patients were male (90.6%), White (66.2%) and with a median age of 15.8 years.
- Suspected myocarditis occurred in 136 patients (97.8%) following mRNA vaccine, with 131 (94.2%) following the Pfizer-BioNTech vaccine; 128 cases (91.4%) occurred after the second dose.
- Symptoms started a median of 2 days (range 0 to 22 days) following vaccination administration.
- Chest pain was the most common symptom (99.3%), with fever present in 30.9% of patients and shortness of breath in 27.3%.
- Patients were treated with nonsteroidal anti-inflammatory drugs (81.3%), intravenous immunoglobulin (21.6%), glucocorticoids (21.6%), colchicine (7.9%) or no anti-inflammatory therapies (8.6%).
- Twenty-six patients (18.7%) were admitted to the intensive care unit; 2 received inotropic/vasoactive support; none required extracorporeal membrane oxygenation or died.
- Median time spent in the hospital was 2 days.
- A total of 111 patients had elevated troponin I (8.12 ng/mL) and 28 had elevated troponin T (0.61 ng/mL).
- More than two-thirds (69.8%) had abnormal electrocardiograms and/or arrhythmias (7 with nonsustained ventricular tachycardia).
- Twenty-six patients (18.7%) had left ventricular ejection fraction (LVEF) less than 55% on echocardiogram; LVEF had returned to normal in the 25 who returned for follow-up.
- 75 of 97 patients (77.3%) who underwent cMRI at a median of 5 days from symptom onset had abnormal findings; 74 (76.3%) had late gadolinium enhancement, 54 (55.7%) had myocardial edema, and 49 (50.5%) met Lake Louise criteria for myocarditis.
“These data suggest that most cases of suspected COVID-19 vaccine–related myocarditis in people younger than 21 are mild and resolve quickly,” corresponding author Dongngan Truong, MD, Division of Pediatric Cardiology, University of Utah and Primary Children’s Hospital, Salt Lake City, said in a statement.
“We were very happy to see that type of recovery. However, we are awaiting further studies to better understand the long-term outcomes of patients who have had COVID-19 vaccination-related myocarditis. We also need to study the risk factors and mechanisms for this rare complication,” Dr. Truong added.
Dr. Lloyd-Jones said these findings support the AHA’s position that COVID-19 vaccines are “safe, highly effective, and fundamental to saving lives, protecting our families and communities against COVID-19, and ending the pandemic.”
The study received no funding. Dr. Truong consults for Pfizer on vaccine-associated myocarditis. A complete list of author disclosures is available with the original article.
A version of this article first appeared on Medscape.com.
AHA challenges diet doctor’s study alleging COVID vax risks
An abstract and poster presentation questioning the safety of mRNA-based COVID-19 vaccines, embraced by some and lambasted by others, has drawn an “expression of concern” from the American Heart Association, along with a bid for correction.
The abstract in question concludes that COVID vaccines “dramatically increase” levels of certain inflammatory biomarkers, and therefore, the 5-year risk of acute coronary syndromes (ACS), based on pre- and post-vaccination results of an obscure blood panel called the PULS Cardiac Test (GD Biosciences). The findings were presented at the AHA’s 2021 Scientific Sessionsas, an uncontrolled observational study of 566 patients in a preventive cardiology practice.
Some on social media have seized on the abstract as evidence of serious potential harm from the two available mRNA-based SARS-CoV-2 vaccines, BNT162b2 (Pfizer-BioNTech) and mRNA-1273 (Moderna). But others contend that the study’s described design and findings are specious and its conclusions overstated.
They also point to the notoriety of its one listed author, Steven R. Gundry, MD, who promotes his diet books and supplements as well as fringe, highly criticized theories about diet and disease on several websites, including drgundry.com. Dr. Gundry has not responded to requests for an interview.
Dr. Gundry’s abstract from the AHA Scientific Sessions 2021, available on the meeting’s program planner, was marked with an “expression of concern” by the AHA that is to stand “until a suitable correction is published, to indicate that the abstract in its current version may not be reliable.”
The expression of concern statement, also published online Nov. 24 in Circulation, says “potential errors in the abstract” were brought to the attention of the meeting planners. “Specifically, there are several typographical errors, there is no data in the abstract regarding myocardial T-cell infiltration, there are no statistical analyses for significance provided, and the author is not clear that only anecdotal data was used.”
The biomarker elevations on which the abstract’s conclusions are based included hepatocyte growth factor, “which serves as a marker for chemotaxis of T-cells into epithelium and cardiac tissue,” it states.
“The expression of concern about the abstract will remain in place until a correction is accepted and published” in Circulation, AHA spokesperson Suzanne Grant told this news organization by email.
“The specific data needed will be up to the abstract author to determine and supply,” she said, noting that Dr. Gundry “has been in communication with the journal throughout this process.”
Submitting researchers “must always attest to the validity of the abstract,” Ms. Grant said. “Abstracts are then curated by independent review panels, blinded to the identities of the abstract authors, and are considered based on the potential to add to the diversity of scientific issues and views discussed at the meeting.”
Regarding the AHA’s system for vetting abstracts vying for acceptance to the scientific sessions, she said it is not primarily intended to “evaluate scientific validity” and that the organization is “currently reviewing its existing abstract submission processes.”
A recent Reuters report reviews the controversy and provides links to criticisms of the study on social media.
A version of this article first appeared on Medscape.com.
An abstract and poster presentation questioning the safety of mRNA-based COVID-19 vaccines, embraced by some and lambasted by others, has drawn an “expression of concern” from the American Heart Association, along with a bid for correction.
The abstract in question concludes that COVID vaccines “dramatically increase” levels of certain inflammatory biomarkers, and therefore, the 5-year risk of acute coronary syndromes (ACS), based on pre- and post-vaccination results of an obscure blood panel called the PULS Cardiac Test (GD Biosciences). The findings were presented at the AHA’s 2021 Scientific Sessionsas, an uncontrolled observational study of 566 patients in a preventive cardiology practice.
Some on social media have seized on the abstract as evidence of serious potential harm from the two available mRNA-based SARS-CoV-2 vaccines, BNT162b2 (Pfizer-BioNTech) and mRNA-1273 (Moderna). But others contend that the study’s described design and findings are specious and its conclusions overstated.
They also point to the notoriety of its one listed author, Steven R. Gundry, MD, who promotes his diet books and supplements as well as fringe, highly criticized theories about diet and disease on several websites, including drgundry.com. Dr. Gundry has not responded to requests for an interview.
Dr. Gundry’s abstract from the AHA Scientific Sessions 2021, available on the meeting’s program planner, was marked with an “expression of concern” by the AHA that is to stand “until a suitable correction is published, to indicate that the abstract in its current version may not be reliable.”
The expression of concern statement, also published online Nov. 24 in Circulation, says “potential errors in the abstract” were brought to the attention of the meeting planners. “Specifically, there are several typographical errors, there is no data in the abstract regarding myocardial T-cell infiltration, there are no statistical analyses for significance provided, and the author is not clear that only anecdotal data was used.”
The biomarker elevations on which the abstract’s conclusions are based included hepatocyte growth factor, “which serves as a marker for chemotaxis of T-cells into epithelium and cardiac tissue,” it states.
“The expression of concern about the abstract will remain in place until a correction is accepted and published” in Circulation, AHA spokesperson Suzanne Grant told this news organization by email.
“The specific data needed will be up to the abstract author to determine and supply,” she said, noting that Dr. Gundry “has been in communication with the journal throughout this process.”
Submitting researchers “must always attest to the validity of the abstract,” Ms. Grant said. “Abstracts are then curated by independent review panels, blinded to the identities of the abstract authors, and are considered based on the potential to add to the diversity of scientific issues and views discussed at the meeting.”
Regarding the AHA’s system for vetting abstracts vying for acceptance to the scientific sessions, she said it is not primarily intended to “evaluate scientific validity” and that the organization is “currently reviewing its existing abstract submission processes.”
A recent Reuters report reviews the controversy and provides links to criticisms of the study on social media.
A version of this article first appeared on Medscape.com.
An abstract and poster presentation questioning the safety of mRNA-based COVID-19 vaccines, embraced by some and lambasted by others, has drawn an “expression of concern” from the American Heart Association, along with a bid for correction.
The abstract in question concludes that COVID vaccines “dramatically increase” levels of certain inflammatory biomarkers, and therefore, the 5-year risk of acute coronary syndromes (ACS), based on pre- and post-vaccination results of an obscure blood panel called the PULS Cardiac Test (GD Biosciences). The findings were presented at the AHA’s 2021 Scientific Sessionsas, an uncontrolled observational study of 566 patients in a preventive cardiology practice.
Some on social media have seized on the abstract as evidence of serious potential harm from the two available mRNA-based SARS-CoV-2 vaccines, BNT162b2 (Pfizer-BioNTech) and mRNA-1273 (Moderna). But others contend that the study’s described design and findings are specious and its conclusions overstated.
They also point to the notoriety of its one listed author, Steven R. Gundry, MD, who promotes his diet books and supplements as well as fringe, highly criticized theories about diet and disease on several websites, including drgundry.com. Dr. Gundry has not responded to requests for an interview.
Dr. Gundry’s abstract from the AHA Scientific Sessions 2021, available on the meeting’s program planner, was marked with an “expression of concern” by the AHA that is to stand “until a suitable correction is published, to indicate that the abstract in its current version may not be reliable.”
The expression of concern statement, also published online Nov. 24 in Circulation, says “potential errors in the abstract” were brought to the attention of the meeting planners. “Specifically, there are several typographical errors, there is no data in the abstract regarding myocardial T-cell infiltration, there are no statistical analyses for significance provided, and the author is not clear that only anecdotal data was used.”
The biomarker elevations on which the abstract’s conclusions are based included hepatocyte growth factor, “which serves as a marker for chemotaxis of T-cells into epithelium and cardiac tissue,” it states.
“The expression of concern about the abstract will remain in place until a correction is accepted and published” in Circulation, AHA spokesperson Suzanne Grant told this news organization by email.
“The specific data needed will be up to the abstract author to determine and supply,” she said, noting that Dr. Gundry “has been in communication with the journal throughout this process.”
Submitting researchers “must always attest to the validity of the abstract,” Ms. Grant said. “Abstracts are then curated by independent review panels, blinded to the identities of the abstract authors, and are considered based on the potential to add to the diversity of scientific issues and views discussed at the meeting.”
Regarding the AHA’s system for vetting abstracts vying for acceptance to the scientific sessions, she said it is not primarily intended to “evaluate scientific validity” and that the organization is “currently reviewing its existing abstract submission processes.”
A recent Reuters report reviews the controversy and provides links to criticisms of the study on social media.
A version of this article first appeared on Medscape.com.
Louisiana to require the COVID-19 vaccine for students
Louisiana Gov. John Bel Edwards says the state government plans to make the COVID-19 vaccine a required immunization for students 16 and older in the state’s public school system.
“I just think it’s really, really important to embrace the science and really it’s also important to not engage in misinformation,” said Gov. Edwards, a Democrat, according to The Advocate. “Absent some compelling reason, which I at present have not seen, I fully expect that we will be adding the vaccine to the schedule.”
Parents could opt out their children from the requirement with a letter from a medical provider or a simple signature in dissent, The Advocate reported. The new rule would go into effect at the start of the 2022 school year and at first would apply to students aged 16 and older.
Republican legislators voiced their opposition to the COVID-19 vaccine requirement at a hearing on Dec. 6, calling it unneeded and an example of governmental overreach.
“I believe the vaccine should be highly recommended but not mandated,” state Rep. Laurie Schlegel said, according to TV station WDSU.
State Sen. Cameron Henry of Metairie said he received “hundreds of emails” from parents asking him to prevent the rule from going into effect, WDSU said.
WDSU said the governor can overrule the committee if it rejects the proposed vaccine rule.
Louisiana State Health Officer Joseph Kanter, MD, testified on Dec. 6 that 18 children had died of COVID-19 in Louisiana and many others had become sick because of it.
“I can’t think of another disease on that childhood schedule that we’ve lost that many kids from. In my mind, it’s very much in the public interest. But it’s the family and the parents’ decision,” Dr. Kanter said.
The addition of the vaccine is being proposed by the Louisiana Department of Health, which has added other vaccines to the required list over the years. In 2015, the legislature added meningitis as a required shot with no controversy, The Advocate said.
A version of this article first appeared on WebMD.com.
Louisiana Gov. John Bel Edwards says the state government plans to make the COVID-19 vaccine a required immunization for students 16 and older in the state’s public school system.
“I just think it’s really, really important to embrace the science and really it’s also important to not engage in misinformation,” said Gov. Edwards, a Democrat, according to The Advocate. “Absent some compelling reason, which I at present have not seen, I fully expect that we will be adding the vaccine to the schedule.”
Parents could opt out their children from the requirement with a letter from a medical provider or a simple signature in dissent, The Advocate reported. The new rule would go into effect at the start of the 2022 school year and at first would apply to students aged 16 and older.
Republican legislators voiced their opposition to the COVID-19 vaccine requirement at a hearing on Dec. 6, calling it unneeded and an example of governmental overreach.
“I believe the vaccine should be highly recommended but not mandated,” state Rep. Laurie Schlegel said, according to TV station WDSU.
State Sen. Cameron Henry of Metairie said he received “hundreds of emails” from parents asking him to prevent the rule from going into effect, WDSU said.
WDSU said the governor can overrule the committee if it rejects the proposed vaccine rule.
Louisiana State Health Officer Joseph Kanter, MD, testified on Dec. 6 that 18 children had died of COVID-19 in Louisiana and many others had become sick because of it.
“I can’t think of another disease on that childhood schedule that we’ve lost that many kids from. In my mind, it’s very much in the public interest. But it’s the family and the parents’ decision,” Dr. Kanter said.
The addition of the vaccine is being proposed by the Louisiana Department of Health, which has added other vaccines to the required list over the years. In 2015, the legislature added meningitis as a required shot with no controversy, The Advocate said.
A version of this article first appeared on WebMD.com.
Louisiana Gov. John Bel Edwards says the state government plans to make the COVID-19 vaccine a required immunization for students 16 and older in the state’s public school system.
“I just think it’s really, really important to embrace the science and really it’s also important to not engage in misinformation,” said Gov. Edwards, a Democrat, according to The Advocate. “Absent some compelling reason, which I at present have not seen, I fully expect that we will be adding the vaccine to the schedule.”
Parents could opt out their children from the requirement with a letter from a medical provider or a simple signature in dissent, The Advocate reported. The new rule would go into effect at the start of the 2022 school year and at first would apply to students aged 16 and older.
Republican legislators voiced their opposition to the COVID-19 vaccine requirement at a hearing on Dec. 6, calling it unneeded and an example of governmental overreach.
“I believe the vaccine should be highly recommended but not mandated,” state Rep. Laurie Schlegel said, according to TV station WDSU.
State Sen. Cameron Henry of Metairie said he received “hundreds of emails” from parents asking him to prevent the rule from going into effect, WDSU said.
WDSU said the governor can overrule the committee if it rejects the proposed vaccine rule.
Louisiana State Health Officer Joseph Kanter, MD, testified on Dec. 6 that 18 children had died of COVID-19 in Louisiana and many others had become sick because of it.
“I can’t think of another disease on that childhood schedule that we’ve lost that many kids from. In my mind, it’s very much in the public interest. But it’s the family and the parents’ decision,” Dr. Kanter said.
The addition of the vaccine is being proposed by the Louisiana Department of Health, which has added other vaccines to the required list over the years. In 2015, the legislature added meningitis as a required shot with no controversy, The Advocate said.
A version of this article first appeared on WebMD.com.
Intent to vaccinate kids against COVID higher among vaccinated parents
“Parental vaccine hesitancy is a major issue for schools resuming in-person instruction, potentially requiring regular testing, strict mask wearing, and physical distancing for safe operation,” wrote lead author Madhura S. Rane, PhD, from the City University of New York in New York City, and colleagues in their paper, published online in JAMA Pediatrics.
The survey was conducted in June 2021 of 1,162 parents with children ranging in age from 2 to 17 years. The majority of parents (74.4%) were already vaccinated/vaccine-willing ,while 25.6% were vaccine hesitant. The study cohort, including both 1,652 children and their parents, was part of the nationwide CHASING COVID.
Vaccinated parents overall were more willing to vaccinate or had already vaccinated their eligible children when compared with vaccine-hesitant parents: 64.9% vs. 8.3% for children 2-4 years of age; 77.6% vs. 12.1% for children 5-11 years of age; 81.3% vs. 13.9% for children 12-15 years of age; and 86.4% vs. 12.7% for children 16-17 years of age; P < .001.
The researchers found greater hesitancy among Black and Hispanic parents, compared with parents who were non-Hispanic White, women, younger, and did not have a college education. Parents of children who were currently attending school remotely or only partially, were found to be more willing to vaccinate their children when compared to parents of children who were attending school fully in person.
The authors also found that parents who knew someone who had died of COVID-19 or had experienced a prior COVID-19 infection, were more willing to vaccinate their children.
Hesitance in vaccinated parents
Interestingly, 10% of COVID-vaccinated parents said they were still hesitant to vaccinate their kids because of concern for long-term adverse effects of the vaccine.
“These data point out that vaccine concerns may exist even among vaccinated or vaccine-favorable parents, so we should ask any parent who has not vaccinated their child whether we can discuss their concerns and perhaps move their opinions,” said William T. Basco Jr, MD, MS, a professor of pediatrics at the Medical University of South Carolina, Charleston, and director of the division of general pediatrics.
In an interview, when asked whether recent approval of the vaccine for children aged 5-11 will likely aid in overcoming parental hesitancy, Dr. Basco replied: “Absolutely. As more children get the vaccine and people know a neighbor or nephew or cousin, etc., who received the vaccine and did fine, it will engender greater comfort and allow parents to feel better about having their own child receive the vaccine.”
Advice for clinicians from outside expert
“We can always start by asking parents if we can help them understand the vaccine and the need for it. The tidal wave of disinformation is huge, but we can, on a daily basis, offer to help families navigate this decision,” concluded Dr. Basco, who was not involved with the new paper.
Funding for this study was provided through grants from the National Institute of Allergy and Infectious Diseases, the CUNY Institute of Implementation Science in Population Health, and the COVID-19 Grant Program of the CUNY Graduate School of Public Health and Health Policy. The authors and Dr. Basco have disclosed no relevant financial relationships.
“Parental vaccine hesitancy is a major issue for schools resuming in-person instruction, potentially requiring regular testing, strict mask wearing, and physical distancing for safe operation,” wrote lead author Madhura S. Rane, PhD, from the City University of New York in New York City, and colleagues in their paper, published online in JAMA Pediatrics.
The survey was conducted in June 2021 of 1,162 parents with children ranging in age from 2 to 17 years. The majority of parents (74.4%) were already vaccinated/vaccine-willing ,while 25.6% were vaccine hesitant. The study cohort, including both 1,652 children and their parents, was part of the nationwide CHASING COVID.
Vaccinated parents overall were more willing to vaccinate or had already vaccinated their eligible children when compared with vaccine-hesitant parents: 64.9% vs. 8.3% for children 2-4 years of age; 77.6% vs. 12.1% for children 5-11 years of age; 81.3% vs. 13.9% for children 12-15 years of age; and 86.4% vs. 12.7% for children 16-17 years of age; P < .001.
The researchers found greater hesitancy among Black and Hispanic parents, compared with parents who were non-Hispanic White, women, younger, and did not have a college education. Parents of children who were currently attending school remotely or only partially, were found to be more willing to vaccinate their children when compared to parents of children who were attending school fully in person.
The authors also found that parents who knew someone who had died of COVID-19 or had experienced a prior COVID-19 infection, were more willing to vaccinate their children.
Hesitance in vaccinated parents
Interestingly, 10% of COVID-vaccinated parents said they were still hesitant to vaccinate their kids because of concern for long-term adverse effects of the vaccine.
“These data point out that vaccine concerns may exist even among vaccinated or vaccine-favorable parents, so we should ask any parent who has not vaccinated their child whether we can discuss their concerns and perhaps move their opinions,” said William T. Basco Jr, MD, MS, a professor of pediatrics at the Medical University of South Carolina, Charleston, and director of the division of general pediatrics.
In an interview, when asked whether recent approval of the vaccine for children aged 5-11 will likely aid in overcoming parental hesitancy, Dr. Basco replied: “Absolutely. As more children get the vaccine and people know a neighbor or nephew or cousin, etc., who received the vaccine and did fine, it will engender greater comfort and allow parents to feel better about having their own child receive the vaccine.”
Advice for clinicians from outside expert
“We can always start by asking parents if we can help them understand the vaccine and the need for it. The tidal wave of disinformation is huge, but we can, on a daily basis, offer to help families navigate this decision,” concluded Dr. Basco, who was not involved with the new paper.
Funding for this study was provided through grants from the National Institute of Allergy and Infectious Diseases, the CUNY Institute of Implementation Science in Population Health, and the COVID-19 Grant Program of the CUNY Graduate School of Public Health and Health Policy. The authors and Dr. Basco have disclosed no relevant financial relationships.
“Parental vaccine hesitancy is a major issue for schools resuming in-person instruction, potentially requiring regular testing, strict mask wearing, and physical distancing for safe operation,” wrote lead author Madhura S. Rane, PhD, from the City University of New York in New York City, and colleagues in their paper, published online in JAMA Pediatrics.
The survey was conducted in June 2021 of 1,162 parents with children ranging in age from 2 to 17 years. The majority of parents (74.4%) were already vaccinated/vaccine-willing ,while 25.6% were vaccine hesitant. The study cohort, including both 1,652 children and their parents, was part of the nationwide CHASING COVID.
Vaccinated parents overall were more willing to vaccinate or had already vaccinated their eligible children when compared with vaccine-hesitant parents: 64.9% vs. 8.3% for children 2-4 years of age; 77.6% vs. 12.1% for children 5-11 years of age; 81.3% vs. 13.9% for children 12-15 years of age; and 86.4% vs. 12.7% for children 16-17 years of age; P < .001.
The researchers found greater hesitancy among Black and Hispanic parents, compared with parents who were non-Hispanic White, women, younger, and did not have a college education. Parents of children who were currently attending school remotely or only partially, were found to be more willing to vaccinate their children when compared to parents of children who were attending school fully in person.
The authors also found that parents who knew someone who had died of COVID-19 or had experienced a prior COVID-19 infection, were more willing to vaccinate their children.
Hesitance in vaccinated parents
Interestingly, 10% of COVID-vaccinated parents said they were still hesitant to vaccinate their kids because of concern for long-term adverse effects of the vaccine.
“These data point out that vaccine concerns may exist even among vaccinated or vaccine-favorable parents, so we should ask any parent who has not vaccinated their child whether we can discuss their concerns and perhaps move their opinions,” said William T. Basco Jr, MD, MS, a professor of pediatrics at the Medical University of South Carolina, Charleston, and director of the division of general pediatrics.
In an interview, when asked whether recent approval of the vaccine for children aged 5-11 will likely aid in overcoming parental hesitancy, Dr. Basco replied: “Absolutely. As more children get the vaccine and people know a neighbor or nephew or cousin, etc., who received the vaccine and did fine, it will engender greater comfort and allow parents to feel better about having their own child receive the vaccine.”
Advice for clinicians from outside expert
“We can always start by asking parents if we can help them understand the vaccine and the need for it. The tidal wave of disinformation is huge, but we can, on a daily basis, offer to help families navigate this decision,” concluded Dr. Basco, who was not involved with the new paper.
Funding for this study was provided through grants from the National Institute of Allergy and Infectious Diseases, the CUNY Institute of Implementation Science in Population Health, and the COVID-19 Grant Program of the CUNY Graduate School of Public Health and Health Policy. The authors and Dr. Basco have disclosed no relevant financial relationships.
FROM JAMA PEDIATRICS
Are physician-owned large groups better than flying solo?
Large, physician-owned group practices are gaining ground as a popular form of practice, even as the number of physicians in solo and small practices declines, and employment maintains its appeal.
As physicians shift from owning private practices to employment in hospital systems, this countertrend is also taking place. Large group practices are growing in number, even as solo and small practices are in decline.
Do large, physician-owned groups bring benefits that beat employment? And how do large groups compare with smaller practices and new opportunities, such as private equity? You’ll find some answers here.
Working in large group practices
Large group practices with 50 or more physicians are enjoying a renaissance, even though physicians are still streaming into hospital systems. The share of physicians in large practices increased from 14.7% in 2018 to 17.2% in 2020, the largest 2-year change for this group, according to the American Medical Association.
“Physicians expect that large groups will treat them better than hospitals do,” says Robert Pearl, MD, former CEO of Permanente Medical Group, the nation’s largest physicians’ group.
Compared with hospitals, “doctors would prefer working in a group practice, if all other things are equal,” says Dr. Pearl, who is now a professor at Stanford (Calif.) University Medical School.
Large group practices can include both multispecialty groups and single-specialty groups. Groups in specialties like urology, orthopedics, and oncology have been growing in recent years, according to Gregory Mertz, managing director of Physician Strategies Group in Virginia Beach, Va.
A group practice could also be an independent physicians association – a federation of small practices that share functions like negotiations with insurers and management. Physicians can also form larger groups for single purposes like running an accountable care organization.
Some large group practices can have a mix of partners and employees. In these groups, “some doctors either don’t want a partnership or aren’t offered one,” says Nathan Miller, CEO of the Medicus Firm, a physician recruitment company in Dallas. The AMA reports that about 10% of physicians are employees of large practices.
“Large groups like the Permanente Medical Group are not partnerships,” Dr. Pearl says. “They tend to be a corporation with a board of directors, and all the physicians are employees, but it’s a physician-led organization.”
Doctors in these groups can enjoy a great deal of control. While Permanente Medical Group is exclusively affiliated with Kaiser, which runs hospitals and an HMO, the group is an independent corporation run by its doctors, who are both shareholders and employees, Dr. Pearl says.
The Cleveland Clinic and Mayo Clinic are not medical groups in the strict sense of the word. They describe themselves as academic medical centers, but Dr. Pearl says, “Doctors have a tremendous amount of control there, particularly those in the most remunerative specialties.”
Pros of large groups
Group practices are able to focus more on the physician participants’ needs and priorities, says Mr. Mertz. “In a hospital-based organization, physicians’ needs have to compete with the needs of the hospital. … In a large group, it can be easier to get policies changed and order equipment.”
However, for many physicians, their primary reason for joining a large group is having negotiating leverage with health insurance plans, and this leverage seems even more important today. It typically results in higher reimbursements, which could translate into higher pay. The higher practice income, however, could be negated by higher administrative overhead, which is endemic in large organizations.
Mr. Mertz says large groups also have the resources to recruit new doctors. Small practices, in contrast, often decide not to grow. The practice would at first need to guarantee the salary of a new partner, which could require existing partners to take a pay cut, which they often don’t want to do. “They’ll decide to ride the practice into the ground,” which means closing it down when they retire, he says.
Cons of large groups
One individual doctor may have relatively little input in decision-making in a large group, and strong leadership may be lacking. One study examining the pros and cons of large group practices found that lack of physician cooperation, investment, and leadership were the most frequently cited barriers in large groups.
Physicians in large groups can also divide into competing factions. Mr. Mertz says rifts are more likely to take place in multispecialty groups, where higher-reimbursed proceduralists resent having to financially support lower-reimbursed primary care physicians. But it’s rare that such rifts actually break up the practice, he says.
Private practice vs. employment
Even as more physicians enter large groups, physicians continue to flee private practice in general. In 2020, the AMA found that the number of physicians in private practices had dropped nearly 5 percentage points since 2018, the largest 2-year drop recorded by the AMA.
The hardest hit are small groups of 10 physicians or fewer, once the backbone of U.S. medicine. A 2020 survey found that 53.7% of physicians still work in small practices of 10 or fewer physicians, compared with 61.4% in 2012.
Private practices tend to be partnerships, but younger physicians, for their part, often don’t want to become a partner. In a 2016 survey, only 22% of medical residents surveyed said they anticipate owning a stake in a practice someday.
What’s good about private practice?
The obvious advantage of private practice is having control. Physician-owners can choose staff, oversee finances, and decide on the direction the practice should take. They don’t have to worry about being fired, because the partnership agreement virtually guarantees each doctor’s place in the group.
The atmosphere in a small practice is often more relaxed. “Private practices tend to offer a family-like environment,” Mr. Miller says. Owners of small practices tend to have lower burnout than large practices, a 2018 study found.
Unlike hospital-employed doctors, private practitioners get to keep their ancillary income. “Physicians own the equipment and receive income generated from ancillary services, not just professional fees,” says Mr. Miller.
What’s negative about private practice?
Since small groups have little negotiating power with private payers, they can’t get favorable reimbursement rates. And while partners are protected from being fired, the practice could still go bankrupt.
Running a private practice means putting on an entrepreneurial hat. To develop a strong practice, you need to learn about marketing, finance, IT, contract negotiations, and facility management. “Most young doctors have no interest in this work,” Mr. Mertz says.
Value-based contracting has added another disadvantage for small practices. “It can be harder for small, independent groups to compete,” says Mike Belkin, JD, a divisional vice president at Merritt Hawkins, a physician recruitment company based in Dallas. “They don’t have the data and integration of services that are necessary for this.”
Employment in hospital systems
More than one-third of all physicians worked for hospitals in 2018, and hospitals’ share has been growing since then. In 2020, for the first time, the AMA found that more than half of all physicians were employed, and employment is mainly a hospital phenomenon.
The trend shows no signs of stopping. In 2019 and 2020, hospitals and other corporate entities acquired 20,900 physician practices, representing 29,800 doctors. “This trend will continue,” Dr. Pearl says. “The bigger will get bigger. It’s all about market control. Everyone wants to be wider, more vertical, and more powerful.”
Pros of hospital employment
“The advantages of hospital employment are mostly financial,” Mr. Mertz says. Unlike a private practice, “there’s no financial risk to hospital employment because you don’t own it. You won’t be on the tab for any losses.”
“Hospitals usually offer a highly competitive salary with less emphasis on production than in a private practice,” he says. New physicians are typically paid a guaranteed salary in the first 1-3 years of employment.
“You don’t have any management responsibilities, as you would in a practice,” Mr. Mertz says. “The hospital has a professional management team to handle the business side. Most young doctors have no interest in this work.”
“Employed physicians have a built-in referral network at a hospital,” Mr. Miller says. This is especially an advantage for new physicians, who don’t yet have a referral network of their own.”
Cons of hospital employment
Physicians employed by a hospital lack control. “You don’t decide the hours you work, the schedules you follow, and the physical facility you work in, and, for the most part, you don’t pick your staff,” Mr. Mertz says.
Like any big organization, hospitals are bureaucratic. “If you want to purchase a new piece of equipment, your request goes up the chain of command,” Mr. Mertz says. “Your purchase has to fit into the budget.” (This can be the case with large groups, too.)
Many employed doctors chafe under this lack of control. In an earlier survey by Medscape, 45% of employed respondents didn’t like having limited influence in decision-making, and 32% said they had less control over their work or schedule.
It’s no wonder that a large percentage of physicians would rather work in practices than hospitals. According to a 2021 Medicus Firm survey, 23% of physicians are interested in working in hospitals, while 40% would rather work either in multispecialty or single-specialty groups, Mr. Miller reports.
Doctors have differing views of hospital employment
New physicians are apt to dismiss any negatives about hospitals. “Lack of autonomy often matters less to younger physicians, who were trained in team-based models,” Mr. Belkin says.
Many young doctors actually like working in a large organization. “Young doctors out of residency are used to having everything at their fingertips – labs and testing is in-house,” Mr. Mertz says.
On the other hand, doctors who were previously self-employed – a group that makes up almost one-third of all hospital-employed doctors – can often be dissatisfied with employment. In a 2014 Medscape survey, 26% of previously self-employed doctors said job satisfaction had not improved with employment.
Mr. Mertz says these doctors remember what it was like to be in charge of a practice. “If you once owned a practice, you can always compare what’s going on now with that experience, and that can make you frustrated.”
Hospitals have higher turnover
It’s much easier to leave an organization when you don’t have an ownership stake. The annual physician turnover rate at hospitals is 28%, compared with 7% at medical groups, according to a 2019 report.
Mr. Belkin says changing jobs has become a way of life for many doctors. “Staying at a job for only a few years is no longer a red flag,” he says. “Physicians are exploring different options. They might try group practice and switch to hospitals or vice versa.”
Physicians are now part of a high-turnover culture: Once in a new job, many are already thinking about the next one. A 2018 survey found that 46% of doctors planned to leave their position within 3 years.
Private equity ownership of practice
Selling majority control of your practice to a private equity firm is a relatively new phenomenon and accounts for a small share of physicians – just 4% in 2020. This trend was originally limited to certain specialties, such as anesthesiology, emergency medicine, and dermatology, but now many others are courted.
The deals work like this: Physicians sell majority control of their practice to investors in return for shares in the private equity practice, and they become employees of that practice. The private equity firm then adds more physicians to the practice and invests in infrastructure with the intention of selling the practice at a large profit, which is then shared with the original physicians.
Pros of private equity
The original owners of the practice stand to make a substantial profit if they are willing to wait several years for the practice to be built up and sold. “If they are patient, they could earn a bonanza,” Mr. Belkin says.
Private equity investment helps the practice expand. “It’s an alternative to going to the bank and borrowing money,” Mr. Mertz says.
Cons of private equity
Physicians lose control of their practice. A client of Mr. Mertz’s briefly considered a private equity offer and turned it down. “The private equity firm would have veto power over what the doctors wanted to do,” he says.
Mr. Belkin says the selling physicians typically lose income after the sale. “Money they earned from ancillary services now goes to the practice,” Mr. Belkin says. The selling doctors could potentially take up to a 30% cut in their compensation, according to Coker Capital Advisors.
A version of this article first appeared on Medscape.com.
Large, physician-owned group practices are gaining ground as a popular form of practice, even as the number of physicians in solo and small practices declines, and employment maintains its appeal.
As physicians shift from owning private practices to employment in hospital systems, this countertrend is also taking place. Large group practices are growing in number, even as solo and small practices are in decline.
Do large, physician-owned groups bring benefits that beat employment? And how do large groups compare with smaller practices and new opportunities, such as private equity? You’ll find some answers here.
Working in large group practices
Large group practices with 50 or more physicians are enjoying a renaissance, even though physicians are still streaming into hospital systems. The share of physicians in large practices increased from 14.7% in 2018 to 17.2% in 2020, the largest 2-year change for this group, according to the American Medical Association.
“Physicians expect that large groups will treat them better than hospitals do,” says Robert Pearl, MD, former CEO of Permanente Medical Group, the nation’s largest physicians’ group.
Compared with hospitals, “doctors would prefer working in a group practice, if all other things are equal,” says Dr. Pearl, who is now a professor at Stanford (Calif.) University Medical School.
Large group practices can include both multispecialty groups and single-specialty groups. Groups in specialties like urology, orthopedics, and oncology have been growing in recent years, according to Gregory Mertz, managing director of Physician Strategies Group in Virginia Beach, Va.
A group practice could also be an independent physicians association – a federation of small practices that share functions like negotiations with insurers and management. Physicians can also form larger groups for single purposes like running an accountable care organization.
Some large group practices can have a mix of partners and employees. In these groups, “some doctors either don’t want a partnership or aren’t offered one,” says Nathan Miller, CEO of the Medicus Firm, a physician recruitment company in Dallas. The AMA reports that about 10% of physicians are employees of large practices.
“Large groups like the Permanente Medical Group are not partnerships,” Dr. Pearl says. “They tend to be a corporation with a board of directors, and all the physicians are employees, but it’s a physician-led organization.”
Doctors in these groups can enjoy a great deal of control. While Permanente Medical Group is exclusively affiliated with Kaiser, which runs hospitals and an HMO, the group is an independent corporation run by its doctors, who are both shareholders and employees, Dr. Pearl says.
The Cleveland Clinic and Mayo Clinic are not medical groups in the strict sense of the word. They describe themselves as academic medical centers, but Dr. Pearl says, “Doctors have a tremendous amount of control there, particularly those in the most remunerative specialties.”
Pros of large groups
Group practices are able to focus more on the physician participants’ needs and priorities, says Mr. Mertz. “In a hospital-based organization, physicians’ needs have to compete with the needs of the hospital. … In a large group, it can be easier to get policies changed and order equipment.”
However, for many physicians, their primary reason for joining a large group is having negotiating leverage with health insurance plans, and this leverage seems even more important today. It typically results in higher reimbursements, which could translate into higher pay. The higher practice income, however, could be negated by higher administrative overhead, which is endemic in large organizations.
Mr. Mertz says large groups also have the resources to recruit new doctors. Small practices, in contrast, often decide not to grow. The practice would at first need to guarantee the salary of a new partner, which could require existing partners to take a pay cut, which they often don’t want to do. “They’ll decide to ride the practice into the ground,” which means closing it down when they retire, he says.
Cons of large groups
One individual doctor may have relatively little input in decision-making in a large group, and strong leadership may be lacking. One study examining the pros and cons of large group practices found that lack of physician cooperation, investment, and leadership were the most frequently cited barriers in large groups.
Physicians in large groups can also divide into competing factions. Mr. Mertz says rifts are more likely to take place in multispecialty groups, where higher-reimbursed proceduralists resent having to financially support lower-reimbursed primary care physicians. But it’s rare that such rifts actually break up the practice, he says.
Private practice vs. employment
Even as more physicians enter large groups, physicians continue to flee private practice in general. In 2020, the AMA found that the number of physicians in private practices had dropped nearly 5 percentage points since 2018, the largest 2-year drop recorded by the AMA.
The hardest hit are small groups of 10 physicians or fewer, once the backbone of U.S. medicine. A 2020 survey found that 53.7% of physicians still work in small practices of 10 or fewer physicians, compared with 61.4% in 2012.
Private practices tend to be partnerships, but younger physicians, for their part, often don’t want to become a partner. In a 2016 survey, only 22% of medical residents surveyed said they anticipate owning a stake in a practice someday.
What’s good about private practice?
The obvious advantage of private practice is having control. Physician-owners can choose staff, oversee finances, and decide on the direction the practice should take. They don’t have to worry about being fired, because the partnership agreement virtually guarantees each doctor’s place in the group.
The atmosphere in a small practice is often more relaxed. “Private practices tend to offer a family-like environment,” Mr. Miller says. Owners of small practices tend to have lower burnout than large practices, a 2018 study found.
Unlike hospital-employed doctors, private practitioners get to keep their ancillary income. “Physicians own the equipment and receive income generated from ancillary services, not just professional fees,” says Mr. Miller.
What’s negative about private practice?
Since small groups have little negotiating power with private payers, they can’t get favorable reimbursement rates. And while partners are protected from being fired, the practice could still go bankrupt.
Running a private practice means putting on an entrepreneurial hat. To develop a strong practice, you need to learn about marketing, finance, IT, contract negotiations, and facility management. “Most young doctors have no interest in this work,” Mr. Mertz says.
Value-based contracting has added another disadvantage for small practices. “It can be harder for small, independent groups to compete,” says Mike Belkin, JD, a divisional vice president at Merritt Hawkins, a physician recruitment company based in Dallas. “They don’t have the data and integration of services that are necessary for this.”
Employment in hospital systems
More than one-third of all physicians worked for hospitals in 2018, and hospitals’ share has been growing since then. In 2020, for the first time, the AMA found that more than half of all physicians were employed, and employment is mainly a hospital phenomenon.
The trend shows no signs of stopping. In 2019 and 2020, hospitals and other corporate entities acquired 20,900 physician practices, representing 29,800 doctors. “This trend will continue,” Dr. Pearl says. “The bigger will get bigger. It’s all about market control. Everyone wants to be wider, more vertical, and more powerful.”
Pros of hospital employment
“The advantages of hospital employment are mostly financial,” Mr. Mertz says. Unlike a private practice, “there’s no financial risk to hospital employment because you don’t own it. You won’t be on the tab for any losses.”
“Hospitals usually offer a highly competitive salary with less emphasis on production than in a private practice,” he says. New physicians are typically paid a guaranteed salary in the first 1-3 years of employment.
“You don’t have any management responsibilities, as you would in a practice,” Mr. Mertz says. “The hospital has a professional management team to handle the business side. Most young doctors have no interest in this work.”
“Employed physicians have a built-in referral network at a hospital,” Mr. Miller says. This is especially an advantage for new physicians, who don’t yet have a referral network of their own.”
Cons of hospital employment
Physicians employed by a hospital lack control. “You don’t decide the hours you work, the schedules you follow, and the physical facility you work in, and, for the most part, you don’t pick your staff,” Mr. Mertz says.
Like any big organization, hospitals are bureaucratic. “If you want to purchase a new piece of equipment, your request goes up the chain of command,” Mr. Mertz says. “Your purchase has to fit into the budget.” (This can be the case with large groups, too.)
Many employed doctors chafe under this lack of control. In an earlier survey by Medscape, 45% of employed respondents didn’t like having limited influence in decision-making, and 32% said they had less control over their work or schedule.
It’s no wonder that a large percentage of physicians would rather work in practices than hospitals. According to a 2021 Medicus Firm survey, 23% of physicians are interested in working in hospitals, while 40% would rather work either in multispecialty or single-specialty groups, Mr. Miller reports.
Doctors have differing views of hospital employment
New physicians are apt to dismiss any negatives about hospitals. “Lack of autonomy often matters less to younger physicians, who were trained in team-based models,” Mr. Belkin says.
Many young doctors actually like working in a large organization. “Young doctors out of residency are used to having everything at their fingertips – labs and testing is in-house,” Mr. Mertz says.
On the other hand, doctors who were previously self-employed – a group that makes up almost one-third of all hospital-employed doctors – can often be dissatisfied with employment. In a 2014 Medscape survey, 26% of previously self-employed doctors said job satisfaction had not improved with employment.
Mr. Mertz says these doctors remember what it was like to be in charge of a practice. “If you once owned a practice, you can always compare what’s going on now with that experience, and that can make you frustrated.”
Hospitals have higher turnover
It’s much easier to leave an organization when you don’t have an ownership stake. The annual physician turnover rate at hospitals is 28%, compared with 7% at medical groups, according to a 2019 report.
Mr. Belkin says changing jobs has become a way of life for many doctors. “Staying at a job for only a few years is no longer a red flag,” he says. “Physicians are exploring different options. They might try group practice and switch to hospitals or vice versa.”
Physicians are now part of a high-turnover culture: Once in a new job, many are already thinking about the next one. A 2018 survey found that 46% of doctors planned to leave their position within 3 years.
Private equity ownership of practice
Selling majority control of your practice to a private equity firm is a relatively new phenomenon and accounts for a small share of physicians – just 4% in 2020. This trend was originally limited to certain specialties, such as anesthesiology, emergency medicine, and dermatology, but now many others are courted.
The deals work like this: Physicians sell majority control of their practice to investors in return for shares in the private equity practice, and they become employees of that practice. The private equity firm then adds more physicians to the practice and invests in infrastructure with the intention of selling the practice at a large profit, which is then shared with the original physicians.
Pros of private equity
The original owners of the practice stand to make a substantial profit if they are willing to wait several years for the practice to be built up and sold. “If they are patient, they could earn a bonanza,” Mr. Belkin says.
Private equity investment helps the practice expand. “It’s an alternative to going to the bank and borrowing money,” Mr. Mertz says.
Cons of private equity
Physicians lose control of their practice. A client of Mr. Mertz’s briefly considered a private equity offer and turned it down. “The private equity firm would have veto power over what the doctors wanted to do,” he says.
Mr. Belkin says the selling physicians typically lose income after the sale. “Money they earned from ancillary services now goes to the practice,” Mr. Belkin says. The selling doctors could potentially take up to a 30% cut in their compensation, according to Coker Capital Advisors.
A version of this article first appeared on Medscape.com.
Large, physician-owned group practices are gaining ground as a popular form of practice, even as the number of physicians in solo and small practices declines, and employment maintains its appeal.
As physicians shift from owning private practices to employment in hospital systems, this countertrend is also taking place. Large group practices are growing in number, even as solo and small practices are in decline.
Do large, physician-owned groups bring benefits that beat employment? And how do large groups compare with smaller practices and new opportunities, such as private equity? You’ll find some answers here.
Working in large group practices
Large group practices with 50 or more physicians are enjoying a renaissance, even though physicians are still streaming into hospital systems. The share of physicians in large practices increased from 14.7% in 2018 to 17.2% in 2020, the largest 2-year change for this group, according to the American Medical Association.
“Physicians expect that large groups will treat them better than hospitals do,” says Robert Pearl, MD, former CEO of Permanente Medical Group, the nation’s largest physicians’ group.
Compared with hospitals, “doctors would prefer working in a group practice, if all other things are equal,” says Dr. Pearl, who is now a professor at Stanford (Calif.) University Medical School.
Large group practices can include both multispecialty groups and single-specialty groups. Groups in specialties like urology, orthopedics, and oncology have been growing in recent years, according to Gregory Mertz, managing director of Physician Strategies Group in Virginia Beach, Va.
A group practice could also be an independent physicians association – a federation of small practices that share functions like negotiations with insurers and management. Physicians can also form larger groups for single purposes like running an accountable care organization.
Some large group practices can have a mix of partners and employees. In these groups, “some doctors either don’t want a partnership or aren’t offered one,” says Nathan Miller, CEO of the Medicus Firm, a physician recruitment company in Dallas. The AMA reports that about 10% of physicians are employees of large practices.
“Large groups like the Permanente Medical Group are not partnerships,” Dr. Pearl says. “They tend to be a corporation with a board of directors, and all the physicians are employees, but it’s a physician-led organization.”
Doctors in these groups can enjoy a great deal of control. While Permanente Medical Group is exclusively affiliated with Kaiser, which runs hospitals and an HMO, the group is an independent corporation run by its doctors, who are both shareholders and employees, Dr. Pearl says.
The Cleveland Clinic and Mayo Clinic are not medical groups in the strict sense of the word. They describe themselves as academic medical centers, but Dr. Pearl says, “Doctors have a tremendous amount of control there, particularly those in the most remunerative specialties.”
Pros of large groups
Group practices are able to focus more on the physician participants’ needs and priorities, says Mr. Mertz. “In a hospital-based organization, physicians’ needs have to compete with the needs of the hospital. … In a large group, it can be easier to get policies changed and order equipment.”
However, for many physicians, their primary reason for joining a large group is having negotiating leverage with health insurance plans, and this leverage seems even more important today. It typically results in higher reimbursements, which could translate into higher pay. The higher practice income, however, could be negated by higher administrative overhead, which is endemic in large organizations.
Mr. Mertz says large groups also have the resources to recruit new doctors. Small practices, in contrast, often decide not to grow. The practice would at first need to guarantee the salary of a new partner, which could require existing partners to take a pay cut, which they often don’t want to do. “They’ll decide to ride the practice into the ground,” which means closing it down when they retire, he says.
Cons of large groups
One individual doctor may have relatively little input in decision-making in a large group, and strong leadership may be lacking. One study examining the pros and cons of large group practices found that lack of physician cooperation, investment, and leadership were the most frequently cited barriers in large groups.
Physicians in large groups can also divide into competing factions. Mr. Mertz says rifts are more likely to take place in multispecialty groups, where higher-reimbursed proceduralists resent having to financially support lower-reimbursed primary care physicians. But it’s rare that such rifts actually break up the practice, he says.
Private practice vs. employment
Even as more physicians enter large groups, physicians continue to flee private practice in general. In 2020, the AMA found that the number of physicians in private practices had dropped nearly 5 percentage points since 2018, the largest 2-year drop recorded by the AMA.
The hardest hit are small groups of 10 physicians or fewer, once the backbone of U.S. medicine. A 2020 survey found that 53.7% of physicians still work in small practices of 10 or fewer physicians, compared with 61.4% in 2012.
Private practices tend to be partnerships, but younger physicians, for their part, often don’t want to become a partner. In a 2016 survey, only 22% of medical residents surveyed said they anticipate owning a stake in a practice someday.
What’s good about private practice?
The obvious advantage of private practice is having control. Physician-owners can choose staff, oversee finances, and decide on the direction the practice should take. They don’t have to worry about being fired, because the partnership agreement virtually guarantees each doctor’s place in the group.
The atmosphere in a small practice is often more relaxed. “Private practices tend to offer a family-like environment,” Mr. Miller says. Owners of small practices tend to have lower burnout than large practices, a 2018 study found.
Unlike hospital-employed doctors, private practitioners get to keep their ancillary income. “Physicians own the equipment and receive income generated from ancillary services, not just professional fees,” says Mr. Miller.
What’s negative about private practice?
Since small groups have little negotiating power with private payers, they can’t get favorable reimbursement rates. And while partners are protected from being fired, the practice could still go bankrupt.
Running a private practice means putting on an entrepreneurial hat. To develop a strong practice, you need to learn about marketing, finance, IT, contract negotiations, and facility management. “Most young doctors have no interest in this work,” Mr. Mertz says.
Value-based contracting has added another disadvantage for small practices. “It can be harder for small, independent groups to compete,” says Mike Belkin, JD, a divisional vice president at Merritt Hawkins, a physician recruitment company based in Dallas. “They don’t have the data and integration of services that are necessary for this.”
Employment in hospital systems
More than one-third of all physicians worked for hospitals in 2018, and hospitals’ share has been growing since then. In 2020, for the first time, the AMA found that more than half of all physicians were employed, and employment is mainly a hospital phenomenon.
The trend shows no signs of stopping. In 2019 and 2020, hospitals and other corporate entities acquired 20,900 physician practices, representing 29,800 doctors. “This trend will continue,” Dr. Pearl says. “The bigger will get bigger. It’s all about market control. Everyone wants to be wider, more vertical, and more powerful.”
Pros of hospital employment
“The advantages of hospital employment are mostly financial,” Mr. Mertz says. Unlike a private practice, “there’s no financial risk to hospital employment because you don’t own it. You won’t be on the tab for any losses.”
“Hospitals usually offer a highly competitive salary with less emphasis on production than in a private practice,” he says. New physicians are typically paid a guaranteed salary in the first 1-3 years of employment.
“You don’t have any management responsibilities, as you would in a practice,” Mr. Mertz says. “The hospital has a professional management team to handle the business side. Most young doctors have no interest in this work.”
“Employed physicians have a built-in referral network at a hospital,” Mr. Miller says. This is especially an advantage for new physicians, who don’t yet have a referral network of their own.”
Cons of hospital employment
Physicians employed by a hospital lack control. “You don’t decide the hours you work, the schedules you follow, and the physical facility you work in, and, for the most part, you don’t pick your staff,” Mr. Mertz says.
Like any big organization, hospitals are bureaucratic. “If you want to purchase a new piece of equipment, your request goes up the chain of command,” Mr. Mertz says. “Your purchase has to fit into the budget.” (This can be the case with large groups, too.)
Many employed doctors chafe under this lack of control. In an earlier survey by Medscape, 45% of employed respondents didn’t like having limited influence in decision-making, and 32% said they had less control over their work or schedule.
It’s no wonder that a large percentage of physicians would rather work in practices than hospitals. According to a 2021 Medicus Firm survey, 23% of physicians are interested in working in hospitals, while 40% would rather work either in multispecialty or single-specialty groups, Mr. Miller reports.
Doctors have differing views of hospital employment
New physicians are apt to dismiss any negatives about hospitals. “Lack of autonomy often matters less to younger physicians, who were trained in team-based models,” Mr. Belkin says.
Many young doctors actually like working in a large organization. “Young doctors out of residency are used to having everything at their fingertips – labs and testing is in-house,” Mr. Mertz says.
On the other hand, doctors who were previously self-employed – a group that makes up almost one-third of all hospital-employed doctors – can often be dissatisfied with employment. In a 2014 Medscape survey, 26% of previously self-employed doctors said job satisfaction had not improved with employment.
Mr. Mertz says these doctors remember what it was like to be in charge of a practice. “If you once owned a practice, you can always compare what’s going on now with that experience, and that can make you frustrated.”
Hospitals have higher turnover
It’s much easier to leave an organization when you don’t have an ownership stake. The annual physician turnover rate at hospitals is 28%, compared with 7% at medical groups, according to a 2019 report.
Mr. Belkin says changing jobs has become a way of life for many doctors. “Staying at a job for only a few years is no longer a red flag,” he says. “Physicians are exploring different options. They might try group practice and switch to hospitals or vice versa.”
Physicians are now part of a high-turnover culture: Once in a new job, many are already thinking about the next one. A 2018 survey found that 46% of doctors planned to leave their position within 3 years.
Private equity ownership of practice
Selling majority control of your practice to a private equity firm is a relatively new phenomenon and accounts for a small share of physicians – just 4% in 2020. This trend was originally limited to certain specialties, such as anesthesiology, emergency medicine, and dermatology, but now many others are courted.
The deals work like this: Physicians sell majority control of their practice to investors in return for shares in the private equity practice, and they become employees of that practice. The private equity firm then adds more physicians to the practice and invests in infrastructure with the intention of selling the practice at a large profit, which is then shared with the original physicians.
Pros of private equity
The original owners of the practice stand to make a substantial profit if they are willing to wait several years for the practice to be built up and sold. “If they are patient, they could earn a bonanza,” Mr. Belkin says.
Private equity investment helps the practice expand. “It’s an alternative to going to the bank and borrowing money,” Mr. Mertz says.
Cons of private equity
Physicians lose control of their practice. A client of Mr. Mertz’s briefly considered a private equity offer and turned it down. “The private equity firm would have veto power over what the doctors wanted to do,” he says.
Mr. Belkin says the selling physicians typically lose income after the sale. “Money they earned from ancillary services now goes to the practice,” Mr. Belkin says. The selling doctors could potentially take up to a 30% cut in their compensation, according to Coker Capital Advisors.
A version of this article first appeared on Medscape.com.
AMA president calls on Congress to stabilize Medicare payments to physicians
Physician practices around the country took an unprecedented financial hit with the arrival of the COVID-19 pandemic in March 2020. Recent research from the American Medical Association reveals an estimated pandemic-related shortfall in Medicare physician fee spending of $13.9 billion, or a 14% reduction, across all states and all major specialties in 2020.
While the report pointed to a “strong recovery” in May and June, that recovery stalled in the second half of 2020, and spending never returned to pre–COVID-19 levels.
“Physicians experienced a significant and sustained drop in Medicare revenue during the first 10 months of the pandemic,” said AMA President Gerald Harmon, MD, in a statement. “Medical practices that have not buckled under financial strain continue to be stretched clinically, emotionally, and fiscally as the pandemic persists. Yet physicians face an array of planned cuts that would reduce Medicare physician payments by nearly 10% for 2022.”
The reduction in the Medicare physician fee schedule payments means providers may face payment cuts of more than 9% starting Jan. 1, 2022, when the cuts take effect. That is, unless Congress makes changes.
Medicare physician fee schedule spending on telehealth stood at $4.1 billion, or 5% of the total Medicare spent in 2020. From March 16 to June 30, $1.8 billion of this amount was on telehealth, while $1.1 billion came in during third and fourth quarters of 2020, respectively, per the report.
According to AMA’s research:
- Medicare physician fee schedule spending for 2020, relative to expected 2020 spending, dipped 32% between March 16 and June 30; spending was down during the last 6 months of the year by between 9% and 10%.
- The care settings hit the worst were ambulatory surgical centers, outpatient hospitals, and physician offices; the next worst off were hospital emergency departments, inpatient hospitals, and skilled nursing facilities.
- The specialties that fared worst included physical therapists (-28%), opthamologists (-19%), podiatrists (-18%), and dermatologists (-18%).
- Cumulative spending was down the most in Minnesota (-22%), Maine (-19%), and New York (-19%); less affected states included Idaho (-9%), Oklahoma (-9%), and South Carolina (9%).
AMA: Budget neutrality hurting physicians’ financial stability
Dr. Harmon is calling for financial stability in Medicare spending. In particular, the AMA is “strongly urging Congress to avert the planned payment cuts,” he said in a statement.
The challenge: The Medicare physician fee schedule is currently “budget neutral,” meaning that the budget is fixed, Dr. Harmon, a family medicine specialist in South Carolina, told this news organization.
“If you rob from Peter to pay Paul, Paul is going to be less efficient or less rewarded. It continues to be that there’s always a ‘pay for’ in these things. So budget neutrality is probably one of the first things we need to address,” he said.
Lack of routine care expected to affect health outcomes
The result of reduced screening and treatment during the pandemic could be as many as 10,000 excess deaths due to cancers of the breast and colon during the next 10 years, wrote Norman Sharpless, MD, director of the National Cancer Institute, in Science in June. Combined, breast cancer and colon cancer account for one-sixth of all cancers in the U.S., he wrote.
In addition, blood pressure control has gotten worse since the start of the pandemic, said Michael Rakotz, MD, FAHA, FAAFP, vice president of improving health outcomes at the AMA, in an AMA blog post.
Dr. Harmon’s advice for physician practices on getting patients in for routine care:
- Educate the area’s largest employers to encourage their employees.
- Engage with hospital employees, since hospitals are often the largest employers in many communities.
- Partner with health insurers.
- Show up at athletic events, which is a particularly good fit for “small town America,” said Dr. Harmon.
The AMA’s research doesn’t consider reimbursement from other public and private payers. It also doesn’t account for funding sources such as Provider Relief Fund grants, Paycheck Protection Program loans, and the temporary suspension of the Medicare sequester, per the report.
A version of this article first appeared on Medscape.com.
Physician practices around the country took an unprecedented financial hit with the arrival of the COVID-19 pandemic in March 2020. Recent research from the American Medical Association reveals an estimated pandemic-related shortfall in Medicare physician fee spending of $13.9 billion, or a 14% reduction, across all states and all major specialties in 2020.
While the report pointed to a “strong recovery” in May and June, that recovery stalled in the second half of 2020, and spending never returned to pre–COVID-19 levels.
“Physicians experienced a significant and sustained drop in Medicare revenue during the first 10 months of the pandemic,” said AMA President Gerald Harmon, MD, in a statement. “Medical practices that have not buckled under financial strain continue to be stretched clinically, emotionally, and fiscally as the pandemic persists. Yet physicians face an array of planned cuts that would reduce Medicare physician payments by nearly 10% for 2022.”
The reduction in the Medicare physician fee schedule payments means providers may face payment cuts of more than 9% starting Jan. 1, 2022, when the cuts take effect. That is, unless Congress makes changes.
Medicare physician fee schedule spending on telehealth stood at $4.1 billion, or 5% of the total Medicare spent in 2020. From March 16 to June 30, $1.8 billion of this amount was on telehealth, while $1.1 billion came in during third and fourth quarters of 2020, respectively, per the report.
According to AMA’s research:
- Medicare physician fee schedule spending for 2020, relative to expected 2020 spending, dipped 32% between March 16 and June 30; spending was down during the last 6 months of the year by between 9% and 10%.
- The care settings hit the worst were ambulatory surgical centers, outpatient hospitals, and physician offices; the next worst off were hospital emergency departments, inpatient hospitals, and skilled nursing facilities.
- The specialties that fared worst included physical therapists (-28%), opthamologists (-19%), podiatrists (-18%), and dermatologists (-18%).
- Cumulative spending was down the most in Minnesota (-22%), Maine (-19%), and New York (-19%); less affected states included Idaho (-9%), Oklahoma (-9%), and South Carolina (9%).
AMA: Budget neutrality hurting physicians’ financial stability
Dr. Harmon is calling for financial stability in Medicare spending. In particular, the AMA is “strongly urging Congress to avert the planned payment cuts,” he said in a statement.
The challenge: The Medicare physician fee schedule is currently “budget neutral,” meaning that the budget is fixed, Dr. Harmon, a family medicine specialist in South Carolina, told this news organization.
“If you rob from Peter to pay Paul, Paul is going to be less efficient or less rewarded. It continues to be that there’s always a ‘pay for’ in these things. So budget neutrality is probably one of the first things we need to address,” he said.
Lack of routine care expected to affect health outcomes
The result of reduced screening and treatment during the pandemic could be as many as 10,000 excess deaths due to cancers of the breast and colon during the next 10 years, wrote Norman Sharpless, MD, director of the National Cancer Institute, in Science in June. Combined, breast cancer and colon cancer account for one-sixth of all cancers in the U.S., he wrote.
In addition, blood pressure control has gotten worse since the start of the pandemic, said Michael Rakotz, MD, FAHA, FAAFP, vice president of improving health outcomes at the AMA, in an AMA blog post.
Dr. Harmon’s advice for physician practices on getting patients in for routine care:
- Educate the area’s largest employers to encourage their employees.
- Engage with hospital employees, since hospitals are often the largest employers in many communities.
- Partner with health insurers.
- Show up at athletic events, which is a particularly good fit for “small town America,” said Dr. Harmon.
The AMA’s research doesn’t consider reimbursement from other public and private payers. It also doesn’t account for funding sources such as Provider Relief Fund grants, Paycheck Protection Program loans, and the temporary suspension of the Medicare sequester, per the report.
A version of this article first appeared on Medscape.com.
Physician practices around the country took an unprecedented financial hit with the arrival of the COVID-19 pandemic in March 2020. Recent research from the American Medical Association reveals an estimated pandemic-related shortfall in Medicare physician fee spending of $13.9 billion, or a 14% reduction, across all states and all major specialties in 2020.
While the report pointed to a “strong recovery” in May and June, that recovery stalled in the second half of 2020, and spending never returned to pre–COVID-19 levels.
“Physicians experienced a significant and sustained drop in Medicare revenue during the first 10 months of the pandemic,” said AMA President Gerald Harmon, MD, in a statement. “Medical practices that have not buckled under financial strain continue to be stretched clinically, emotionally, and fiscally as the pandemic persists. Yet physicians face an array of planned cuts that would reduce Medicare physician payments by nearly 10% for 2022.”
The reduction in the Medicare physician fee schedule payments means providers may face payment cuts of more than 9% starting Jan. 1, 2022, when the cuts take effect. That is, unless Congress makes changes.
Medicare physician fee schedule spending on telehealth stood at $4.1 billion, or 5% of the total Medicare spent in 2020. From March 16 to June 30, $1.8 billion of this amount was on telehealth, while $1.1 billion came in during third and fourth quarters of 2020, respectively, per the report.
According to AMA’s research:
- Medicare physician fee schedule spending for 2020, relative to expected 2020 spending, dipped 32% between March 16 and June 30; spending was down during the last 6 months of the year by between 9% and 10%.
- The care settings hit the worst were ambulatory surgical centers, outpatient hospitals, and physician offices; the next worst off were hospital emergency departments, inpatient hospitals, and skilled nursing facilities.
- The specialties that fared worst included physical therapists (-28%), opthamologists (-19%), podiatrists (-18%), and dermatologists (-18%).
- Cumulative spending was down the most in Minnesota (-22%), Maine (-19%), and New York (-19%); less affected states included Idaho (-9%), Oklahoma (-9%), and South Carolina (9%).
AMA: Budget neutrality hurting physicians’ financial stability
Dr. Harmon is calling for financial stability in Medicare spending. In particular, the AMA is “strongly urging Congress to avert the planned payment cuts,” he said in a statement.
The challenge: The Medicare physician fee schedule is currently “budget neutral,” meaning that the budget is fixed, Dr. Harmon, a family medicine specialist in South Carolina, told this news organization.
“If you rob from Peter to pay Paul, Paul is going to be less efficient or less rewarded. It continues to be that there’s always a ‘pay for’ in these things. So budget neutrality is probably one of the first things we need to address,” he said.
Lack of routine care expected to affect health outcomes
The result of reduced screening and treatment during the pandemic could be as many as 10,000 excess deaths due to cancers of the breast and colon during the next 10 years, wrote Norman Sharpless, MD, director of the National Cancer Institute, in Science in June. Combined, breast cancer and colon cancer account for one-sixth of all cancers in the U.S., he wrote.
In addition, blood pressure control has gotten worse since the start of the pandemic, said Michael Rakotz, MD, FAHA, FAAFP, vice president of improving health outcomes at the AMA, in an AMA blog post.
Dr. Harmon’s advice for physician practices on getting patients in for routine care:
- Educate the area’s largest employers to encourage their employees.
- Engage with hospital employees, since hospitals are often the largest employers in many communities.
- Partner with health insurers.
- Show up at athletic events, which is a particularly good fit for “small town America,” said Dr. Harmon.
The AMA’s research doesn’t consider reimbursement from other public and private payers. It also doesn’t account for funding sources such as Provider Relief Fund grants, Paycheck Protection Program loans, and the temporary suspension of the Medicare sequester, per the report.
A version of this article first appeared on Medscape.com.
Children and COVID-19: 7 million cases and still counting
Total COVID-19 cases in children surpassed the 7-million mark as new cases rose slightly after the previous week’s decline, according to the American Academy of Pediatrics and the Children’s Hospital Association.
, the AAP and CHA said in their weekly COVID-19 report. New cases had dropped the previous week after 3 straight weeks of increases since late October.
The Centers for Disease Control and Prevention puts the total number of child COVID-19 cases at 6.2 million, but both estimates are based on all-age totals – 40 million for the CDC and 41 million for the AAP/CHA – that are well short of the CDC’s latest cumulative figure, which is now just over 49 million, so the actual figures are undoubtedly higher.
Meanwhile, the 1-month anniversary of 5- to 11-year-olds’ vaccine eligibility brought many completions: 923,000 received their second dose during the week ending Dec. 6, compared with 405,000 the previous week. About 16.9% (4.9 million) of children aged 5-11 have gotten at least one dose of the COVID-19 vaccine thus far, of whom almost 1.5 million children (5.1% of the age group) are now fully vaccinated, the CDC said on its COVID-19 Data Tracker.
The pace of vaccinations, however, is much lower for older children. Weekly numbers for all COVID-19 vaccinations, both first and second doses, dropped from 84,000 (Nov. 23-29) to 70,000 (Nov. 30 to Dec. 6), for those aged 12-17 years. In that group, 61.6% have received at least one dose and 51.8% are fully vaccinated, the CDC said.
The pace of vaccinations varies for younger children as well, when geography is considered. The AAP analyzed the CDC’s data and found that 42% of all 5- to 11-year-olds in Vermont had received at least one dose as of Dec. 1, followed by Massachusetts (33%), Maine (30%), and Rhode Island (28%). At the other end of the vaccination scale are Alabama, Louisiana, Mississippi, and West Virginia, all with 4%, the AAP reported.
As the United States puts 7 million children infected with COVID-19 in its rear view mirror, another milestone is looming ahead: The CDC’s current count of deaths in children is 974.
Total COVID-19 cases in children surpassed the 7-million mark as new cases rose slightly after the previous week’s decline, according to the American Academy of Pediatrics and the Children’s Hospital Association.

, the AAP and CHA said in their weekly COVID-19 report. New cases had dropped the previous week after 3 straight weeks of increases since late October.
The Centers for Disease Control and Prevention puts the total number of child COVID-19 cases at 6.2 million, but both estimates are based on all-age totals – 40 million for the CDC and 41 million for the AAP/CHA – that are well short of the CDC’s latest cumulative figure, which is now just over 49 million, so the actual figures are undoubtedly higher.
Meanwhile, the 1-month anniversary of 5- to 11-year-olds’ vaccine eligibility brought many completions: 923,000 received their second dose during the week ending Dec. 6, compared with 405,000 the previous week. About 16.9% (4.9 million) of children aged 5-11 have gotten at least one dose of the COVID-19 vaccine thus far, of whom almost 1.5 million children (5.1% of the age group) are now fully vaccinated, the CDC said on its COVID-19 Data Tracker.
The pace of vaccinations, however, is much lower for older children. Weekly numbers for all COVID-19 vaccinations, both first and second doses, dropped from 84,000 (Nov. 23-29) to 70,000 (Nov. 30 to Dec. 6), for those aged 12-17 years. In that group, 61.6% have received at least one dose and 51.8% are fully vaccinated, the CDC said.
The pace of vaccinations varies for younger children as well, when geography is considered. The AAP analyzed the CDC’s data and found that 42% of all 5- to 11-year-olds in Vermont had received at least one dose as of Dec. 1, followed by Massachusetts (33%), Maine (30%), and Rhode Island (28%). At the other end of the vaccination scale are Alabama, Louisiana, Mississippi, and West Virginia, all with 4%, the AAP reported.
As the United States puts 7 million children infected with COVID-19 in its rear view mirror, another milestone is looming ahead: The CDC’s current count of deaths in children is 974.
Total COVID-19 cases in children surpassed the 7-million mark as new cases rose slightly after the previous week’s decline, according to the American Academy of Pediatrics and the Children’s Hospital Association.

, the AAP and CHA said in their weekly COVID-19 report. New cases had dropped the previous week after 3 straight weeks of increases since late October.
The Centers for Disease Control and Prevention puts the total number of child COVID-19 cases at 6.2 million, but both estimates are based on all-age totals – 40 million for the CDC and 41 million for the AAP/CHA – that are well short of the CDC’s latest cumulative figure, which is now just over 49 million, so the actual figures are undoubtedly higher.
Meanwhile, the 1-month anniversary of 5- to 11-year-olds’ vaccine eligibility brought many completions: 923,000 received their second dose during the week ending Dec. 6, compared with 405,000 the previous week. About 16.9% (4.9 million) of children aged 5-11 have gotten at least one dose of the COVID-19 vaccine thus far, of whom almost 1.5 million children (5.1% of the age group) are now fully vaccinated, the CDC said on its COVID-19 Data Tracker.
The pace of vaccinations, however, is much lower for older children. Weekly numbers for all COVID-19 vaccinations, both first and second doses, dropped from 84,000 (Nov. 23-29) to 70,000 (Nov. 30 to Dec. 6), for those aged 12-17 years. In that group, 61.6% have received at least one dose and 51.8% are fully vaccinated, the CDC said.
The pace of vaccinations varies for younger children as well, when geography is considered. The AAP analyzed the CDC’s data and found that 42% of all 5- to 11-year-olds in Vermont had received at least one dose as of Dec. 1, followed by Massachusetts (33%), Maine (30%), and Rhode Island (28%). At the other end of the vaccination scale are Alabama, Louisiana, Mississippi, and West Virginia, all with 4%, the AAP reported.
As the United States puts 7 million children infected with COVID-19 in its rear view mirror, another milestone is looming ahead: The CDC’s current count of deaths in children is 974.