User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
nav[contains(@class, 'nav-ce-stack nav-ce-stack__large-screen')]
header[@id='header']
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
Epidiolex plus THC lowers seizures in pediatric epilepsy
the component of cannabis that makes people high in larger quantities, researchers reported.
“THC can contribute to seizure control and mitigation some of the side effects of CBD,” said study coauthor and Austin, Tex., child neurologist Karen Keough, MD, in an interview. Dr. Keough and colleagues presented their findings at the 50th annual meeting of the Child Neurology Society.
In a landmark move, the Food and Drug Administration approved Epidiolex in 2018 for the treatment of seizures in two rare forms of epilepsy, Lennox-Gastaut syndrome and Dravet syndrome. The agency had never before approved a drug with a purified ingredient derived from marijuana.
CBD, the active ingredient in Epidiolex, is nonpsychoactive. The use in medicine of THC, the main driver of marijuana’s ability to make people stoned, is much more controversial.
Dr. Keough said she had treated 60-70 children with CBD, at the same strength as in Epidiolex (100 mg), and 5 mg of THC before the drug was approved. “I was seeing some very impressive results, and some became seizure free who’d always been refractory,” she said.
When the Epidiolex became available, she said, some patients transitioned to it and stopped taking THC. According to her, some patients fared well. But others immediately experienced worse seizures, she said, and some developed side effects to Epidiolex in the absence of THC, such as agitation and appetite suppression.
Combination therapy
For the new study, a retrospective, unblinded cohort analysis, Dr. Keough and colleagues tracked patients who received various doses of CBD, in some cases as Epidiolex, and various doses of THC prescribed by the Texas Original Compassionate Cultivation dispensary, where she serves as chief medical officer.
The initial number of patients was 212; 135 consented to review and 10 were excluded for various reasons leaving a total of 74 subjects in the study. The subjects, whose median age at the start of the study was 12 years (range, 2-25 years), were tracked from 2018 to2021. Just over half (55%) were male, and they remained on the regimen for a median of 805 days (range, 400-1,141).
Of the 74 subjects, 45.9% had a reduction of seizures of more than 75%, and 20.3% had a reduction of 50%-75%. Only 4.1% saw their seizures worsen.
The THC doses varied from none to more than 12 mg/day; CBD doses varied from none to more than 26 mg/kg per day. O the 74 patients, 18 saw their greatest seizure reduction from baseline when they received no THC; 12 saw their greatest seizure reduction from baseline when they received 0-2 mg/kg per day of CBD.
Still controversial
Did the patients get high? In some cases they did, Dr. Keough said. However, “a lot of these patients are either too young or too cognitively limited to describe whether they’re feeling intoxicated. That’s one of the many reasons why this is so controversial. You have to go into this with eyes wide open. We’re working in an environment with limited information as to what an intoxicating dose is for a small kid.”
However, she said, it seems clear that “THC can enhance the effect of CBD in children with epilepsy” and reduce CBD side effects. It’s not surprising that the substances work differently since they interact with brain cells in different ways, she said.
For neurologists, she said, “the challenge is to find a reliable source of THC that you can count on and verify so you aren’t overdosing the patients.”
University of Saskatchewan, Saskatoon, child neurologist and cannabinoid researcher Richard Huntsman, MD, who’s familiar with the study findings, said in an interview that they “provide another strong signal that the addition of THC provides benefit, at least in some patients.”
But it’s still unclear “why some children respond best in regards to seizure reduction and side effect profile with combination CBD:THC therapy, and others seemed to do better with CBD alone,” he said. Also unknown: “the ideal THC:CBD ratio that allows optimal seizure control while preventing the potential harmful effects of THC.”
As for the future, he said, “as we are just scratching the surface of our knowledge about the use of cannabis-based therapies in children with neurological disorders, I suspect that the use of these therapies will expand over time.”
No study funding is reported. Dr. Keough disclosed serving as chief medical officer of Texas Original Compassionate Cultivation. Dr. Huntsman disclosed serving as lead investigator of the Cannabidiol in Children with Refractory Epileptic Encephalopathy study and serving on the boards of the Cannabinoid Research Initiative of Saskatchewan (University of Saskatchewan) and Canadian Childhood Cannabinoid Clinical Trials Consortium. He is also cochair of Health Canada’s Scientific Advisory Committee on Cannabinoids for Health Purposes.
the component of cannabis that makes people high in larger quantities, researchers reported.
“THC can contribute to seizure control and mitigation some of the side effects of CBD,” said study coauthor and Austin, Tex., child neurologist Karen Keough, MD, in an interview. Dr. Keough and colleagues presented their findings at the 50th annual meeting of the Child Neurology Society.
In a landmark move, the Food and Drug Administration approved Epidiolex in 2018 for the treatment of seizures in two rare forms of epilepsy, Lennox-Gastaut syndrome and Dravet syndrome. The agency had never before approved a drug with a purified ingredient derived from marijuana.
CBD, the active ingredient in Epidiolex, is nonpsychoactive. The use in medicine of THC, the main driver of marijuana’s ability to make people stoned, is much more controversial.
Dr. Keough said she had treated 60-70 children with CBD, at the same strength as in Epidiolex (100 mg), and 5 mg of THC before the drug was approved. “I was seeing some very impressive results, and some became seizure free who’d always been refractory,” she said.
When the Epidiolex became available, she said, some patients transitioned to it and stopped taking THC. According to her, some patients fared well. But others immediately experienced worse seizures, she said, and some developed side effects to Epidiolex in the absence of THC, such as agitation and appetite suppression.
Combination therapy
For the new study, a retrospective, unblinded cohort analysis, Dr. Keough and colleagues tracked patients who received various doses of CBD, in some cases as Epidiolex, and various doses of THC prescribed by the Texas Original Compassionate Cultivation dispensary, where she serves as chief medical officer.
The initial number of patients was 212; 135 consented to review and 10 were excluded for various reasons leaving a total of 74 subjects in the study. The subjects, whose median age at the start of the study was 12 years (range, 2-25 years), were tracked from 2018 to2021. Just over half (55%) were male, and they remained on the regimen for a median of 805 days (range, 400-1,141).
Of the 74 subjects, 45.9% had a reduction of seizures of more than 75%, and 20.3% had a reduction of 50%-75%. Only 4.1% saw their seizures worsen.
The THC doses varied from none to more than 12 mg/day; CBD doses varied from none to more than 26 mg/kg per day. O the 74 patients, 18 saw their greatest seizure reduction from baseline when they received no THC; 12 saw their greatest seizure reduction from baseline when they received 0-2 mg/kg per day of CBD.
Still controversial
Did the patients get high? In some cases they did, Dr. Keough said. However, “a lot of these patients are either too young or too cognitively limited to describe whether they’re feeling intoxicated. That’s one of the many reasons why this is so controversial. You have to go into this with eyes wide open. We’re working in an environment with limited information as to what an intoxicating dose is for a small kid.”
However, she said, it seems clear that “THC can enhance the effect of CBD in children with epilepsy” and reduce CBD side effects. It’s not surprising that the substances work differently since they interact with brain cells in different ways, she said.
For neurologists, she said, “the challenge is to find a reliable source of THC that you can count on and verify so you aren’t overdosing the patients.”
University of Saskatchewan, Saskatoon, child neurologist and cannabinoid researcher Richard Huntsman, MD, who’s familiar with the study findings, said in an interview that they “provide another strong signal that the addition of THC provides benefit, at least in some patients.”
But it’s still unclear “why some children respond best in regards to seizure reduction and side effect profile with combination CBD:THC therapy, and others seemed to do better with CBD alone,” he said. Also unknown: “the ideal THC:CBD ratio that allows optimal seizure control while preventing the potential harmful effects of THC.”
As for the future, he said, “as we are just scratching the surface of our knowledge about the use of cannabis-based therapies in children with neurological disorders, I suspect that the use of these therapies will expand over time.”
No study funding is reported. Dr. Keough disclosed serving as chief medical officer of Texas Original Compassionate Cultivation. Dr. Huntsman disclosed serving as lead investigator of the Cannabidiol in Children with Refractory Epileptic Encephalopathy study and serving on the boards of the Cannabinoid Research Initiative of Saskatchewan (University of Saskatchewan) and Canadian Childhood Cannabinoid Clinical Trials Consortium. He is also cochair of Health Canada’s Scientific Advisory Committee on Cannabinoids for Health Purposes.
the component of cannabis that makes people high in larger quantities, researchers reported.
“THC can contribute to seizure control and mitigation some of the side effects of CBD,” said study coauthor and Austin, Tex., child neurologist Karen Keough, MD, in an interview. Dr. Keough and colleagues presented their findings at the 50th annual meeting of the Child Neurology Society.
In a landmark move, the Food and Drug Administration approved Epidiolex in 2018 for the treatment of seizures in two rare forms of epilepsy, Lennox-Gastaut syndrome and Dravet syndrome. The agency had never before approved a drug with a purified ingredient derived from marijuana.
CBD, the active ingredient in Epidiolex, is nonpsychoactive. The use in medicine of THC, the main driver of marijuana’s ability to make people stoned, is much more controversial.
Dr. Keough said she had treated 60-70 children with CBD, at the same strength as in Epidiolex (100 mg), and 5 mg of THC before the drug was approved. “I was seeing some very impressive results, and some became seizure free who’d always been refractory,” she said.
When the Epidiolex became available, she said, some patients transitioned to it and stopped taking THC. According to her, some patients fared well. But others immediately experienced worse seizures, she said, and some developed side effects to Epidiolex in the absence of THC, such as agitation and appetite suppression.
Combination therapy
For the new study, a retrospective, unblinded cohort analysis, Dr. Keough and colleagues tracked patients who received various doses of CBD, in some cases as Epidiolex, and various doses of THC prescribed by the Texas Original Compassionate Cultivation dispensary, where she serves as chief medical officer.
The initial number of patients was 212; 135 consented to review and 10 were excluded for various reasons leaving a total of 74 subjects in the study. The subjects, whose median age at the start of the study was 12 years (range, 2-25 years), were tracked from 2018 to2021. Just over half (55%) were male, and they remained on the regimen for a median of 805 days (range, 400-1,141).
Of the 74 subjects, 45.9% had a reduction of seizures of more than 75%, and 20.3% had a reduction of 50%-75%. Only 4.1% saw their seizures worsen.
The THC doses varied from none to more than 12 mg/day; CBD doses varied from none to more than 26 mg/kg per day. O the 74 patients, 18 saw their greatest seizure reduction from baseline when they received no THC; 12 saw their greatest seizure reduction from baseline when they received 0-2 mg/kg per day of CBD.
Still controversial
Did the patients get high? In some cases they did, Dr. Keough said. However, “a lot of these patients are either too young or too cognitively limited to describe whether they’re feeling intoxicated. That’s one of the many reasons why this is so controversial. You have to go into this with eyes wide open. We’re working in an environment with limited information as to what an intoxicating dose is for a small kid.”
However, she said, it seems clear that “THC can enhance the effect of CBD in children with epilepsy” and reduce CBD side effects. It’s not surprising that the substances work differently since they interact with brain cells in different ways, she said.
For neurologists, she said, “the challenge is to find a reliable source of THC that you can count on and verify so you aren’t overdosing the patients.”
University of Saskatchewan, Saskatoon, child neurologist and cannabinoid researcher Richard Huntsman, MD, who’s familiar with the study findings, said in an interview that they “provide another strong signal that the addition of THC provides benefit, at least in some patients.”
But it’s still unclear “why some children respond best in regards to seizure reduction and side effect profile with combination CBD:THC therapy, and others seemed to do better with CBD alone,” he said. Also unknown: “the ideal THC:CBD ratio that allows optimal seizure control while preventing the potential harmful effects of THC.”
As for the future, he said, “as we are just scratching the surface of our knowledge about the use of cannabis-based therapies in children with neurological disorders, I suspect that the use of these therapies will expand over time.”
No study funding is reported. Dr. Keough disclosed serving as chief medical officer of Texas Original Compassionate Cultivation. Dr. Huntsman disclosed serving as lead investigator of the Cannabidiol in Children with Refractory Epileptic Encephalopathy study and serving on the boards of the Cannabinoid Research Initiative of Saskatchewan (University of Saskatchewan) and Canadian Childhood Cannabinoid Clinical Trials Consortium. He is also cochair of Health Canada’s Scientific Advisory Committee on Cannabinoids for Health Purposes.
FROM CNS 2021
Double antiglutamatergic therapy is ‘promising’ for super-refractory status epilepticus
(SRSE), new research suggests.
In a retrospective cohort study of survivors of cardiac arrest with postanoxic sustained SRSE, resolution of the condition was achieved by 81% of those who received intensive treatment of ketamine plus perampanel, versus 41% of those who received standard care.
The novelty of the new treatment approach is the duration of therapy as well as the dual antiglutamate drugs, researchers note.
“So the logic is to continue treatment until resolution of refractory status epilepticus under continuous EEG [electroencephalographic] monitoring,” reported lead investigator Simone Beretta, MD, San Gerardo University Hospital, Monza, Italy.
Therapy was guided by data on brainstem reflexes, N20 cortical responses, neuronal serum enolase levels, and neuroimaging.
If all or most of these indicators are favorable, “we continue to treat without any time limit,” Dr. Beretta said. However, if the indicators become unfavorable, clinicians should consider lowering the intensity of care, he added.
The findings were presented at the 2021 World Congress of Neurology (WCN).
SUPER-CAT trial
In SRSE, epileptic seizures occur one after another without patients recovering consciousness in between. Standard aggressive therapy for the condition does not include antiglutamatergic drugs, the researchers noted.
In the Super-Refractory Status Epilepticus After Cardiac Arrest: Aggressive Treatment Guided by Multimodal Prognostic Indicators (SUPER-CAT) study, researchers assessed the combination of two such medications.
The first was the anti-NMDA receptor drug ketamine, which was given by intravenous bolus and then continuous infusion for 3 days guided by continuous EEG to reach a ketamine EEG pattern, as evidenced by alpha and beta waves. It was combined with the anti-AMPA receptor antiepileptic perampanel via nasogastric tube for 5 days, followed by slow tapering.
Dr. Beretta noted that in the ongoing TELSTAR trial, which involved a similar patient population, a different drug combination is being used. A major difference between the two trials is that in the TELSTAR trial, aggressive therapy continues for only 2 days if there is no response.
“In the SUPER-CAT study, we continue far beyond 2 days in the majority of patients,” he said. In addition, ketamine and perampanel were not assessed in TELSTAR.
In SUPER-CAT, 489 survivors of cardiac arrest were recruited over 10 years. Of these, 101 had refractory status epilepticus. After excluding those with more than two indicators of poor prognosis (n = 31) or whose status epilepticus resolved (n = 14), 56 patients were determined to have SRSE. All had experienced relapse after undergoing one cycle of anesthetic.
The 56 participants received one of three treatment regimens: double antiglutamate (DAG) therapy of ketamine and perampanel (n = 26), single antiglutamate therapy with either agent (n = 8), or aggressive nonantiglutamate (NAG) therapy with antiepilepsy drugs and anesthetics other than ketamine or perampanel (n = 22).
The single-antiglutamate group was not included in the analysis of patient outcomes.
The DAG and NAG groups were well balanced at baseline. There were no significant differences in median age (60 years vs. 66 years), gender, low cerebral blood flow, presence of bilateral pupillary or corneal reflexes, neuron-specific enolase levels, cortical N20 somatosenory evoked potentials, moderate to severe postanoxic brain injury, and hypothermia/targeted temperature management.
Primary outcome met
More patients in the DAG group (42%) had moderate to severe postanoxic brain injury than in the NAG group (28%). However, the difference was not statistically significant (P = .08), possibly because of the small sample sizes. The number of antiepileptic drugs and the number of cycles of anesthetics did not differ between the groups.
Results showed that efficacy and safety outcomes favored DAG therapy.
The primary efficacy outcome was resolution of status epilepticus within 3 days after initiation of treatments. Status epilepticus resolved for 21 of 26 patients in the DAG group (81%), versus 9 of 22 patients in the NAG group (41%; odds ratio, 6.06; 95% confidence interval, 1.66-22.12; P = .005).
For secondary efficacy outcomes, there was a trend in favor of DAG, but differences from the NAG group were not statistically significant. In the groups, 46% versus 32% awakened and responded to commands before discharge from the intensive care unit, and 32% versus 23% showed good neurologic outcome at 6 months.
The primary safety outcome of all-cause mortality risk in the ICU was 90% lower for patients treated with DAG than for those treated with NAG (15% vs. 64%; OR, 0.1; 95% CI, 0.02-0.41; P < .01). Dr. Beretta explained that the high mortality rate in the NAG group was presumably a result of unresolved status epilepticus.
The secondary safety outcome of a transitory rise of gamma-glutamyl transferase greater than three times the upper limit of normal in the DAG group was expected with high-dose perampanel, the investigators noted. This outcome occurred in 77% of the DAG group versus 27% of the NAG group (OR, 9.88; 95% CI, 2.4-32.9; P < .001).
There was no statistically significant difference in incidence of recurrent cardiac arrest during therapy. This occurred in one member of the DAG group and in none in the NAG group.
Dr. Beretta reported that their investigations are still in a retrospective phase, but the researchers plan to move the work into a prospective phase and possibly a randomized trial soon.
Fascinating, promising
Commenting on the findings, Jaysingh Singh, MD, co-director of the Epilepsy Surgery Center at the Ohio State University Wexner Medical Center, Columbus, said the study provides “fascinating, very helpful data” about a condition that has responded well to current treatment options.
He added that his center has used “the innovative treatments” discussed in the study for a few patients.
“More concrete evidence will push us to use it more uniformly across all our patient population [that] has refractory status. So I’m very optimistic, and the data were very promising,” said Dr. Singh, who was not involved with the research.
He cautioned that the study was retrospective, not randomized or controlled, and that it involved a small number of patients but said that the data were “heading in the right direction.”
Although resolution of status epilepticus was better among patients in the DAG group than in the NAG group, the awakenings and neurologic outcomes were “pretty much same as standard medical therapy, which we commonly give to our patients,” said Dr. Singh. “We see this phenomenon all the time in our patients.”
He noted that other factors can determine how patients respond, such as conditions of the heart or kidneys, the presence of sepsis, and multiorgan dysfunction. These factors were not controlled for in the study.
Nonetheless, he said the study achieved its primary endpoint of better resolution of status epilepticus “because that’s the first thing you want to see: whether the treatment is taking care of that.”
The study received no outside funding. Dr. Beretta and Dr. Singh have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
(SRSE), new research suggests.
In a retrospective cohort study of survivors of cardiac arrest with postanoxic sustained SRSE, resolution of the condition was achieved by 81% of those who received intensive treatment of ketamine plus perampanel, versus 41% of those who received standard care.
The novelty of the new treatment approach is the duration of therapy as well as the dual antiglutamate drugs, researchers note.
“So the logic is to continue treatment until resolution of refractory status epilepticus under continuous EEG [electroencephalographic] monitoring,” reported lead investigator Simone Beretta, MD, San Gerardo University Hospital, Monza, Italy.
Therapy was guided by data on brainstem reflexes, N20 cortical responses, neuronal serum enolase levels, and neuroimaging.
If all or most of these indicators are favorable, “we continue to treat without any time limit,” Dr. Beretta said. However, if the indicators become unfavorable, clinicians should consider lowering the intensity of care, he added.
The findings were presented at the 2021 World Congress of Neurology (WCN).
SUPER-CAT trial
In SRSE, epileptic seizures occur one after another without patients recovering consciousness in between. Standard aggressive therapy for the condition does not include antiglutamatergic drugs, the researchers noted.
In the Super-Refractory Status Epilepticus After Cardiac Arrest: Aggressive Treatment Guided by Multimodal Prognostic Indicators (SUPER-CAT) study, researchers assessed the combination of two such medications.
The first was the anti-NMDA receptor drug ketamine, which was given by intravenous bolus and then continuous infusion for 3 days guided by continuous EEG to reach a ketamine EEG pattern, as evidenced by alpha and beta waves. It was combined with the anti-AMPA receptor antiepileptic perampanel via nasogastric tube for 5 days, followed by slow tapering.
Dr. Beretta noted that in the ongoing TELSTAR trial, which involved a similar patient population, a different drug combination is being used. A major difference between the two trials is that in the TELSTAR trial, aggressive therapy continues for only 2 days if there is no response.
“In the SUPER-CAT study, we continue far beyond 2 days in the majority of patients,” he said. In addition, ketamine and perampanel were not assessed in TELSTAR.
In SUPER-CAT, 489 survivors of cardiac arrest were recruited over 10 years. Of these, 101 had refractory status epilepticus. After excluding those with more than two indicators of poor prognosis (n = 31) or whose status epilepticus resolved (n = 14), 56 patients were determined to have SRSE. All had experienced relapse after undergoing one cycle of anesthetic.
The 56 participants received one of three treatment regimens: double antiglutamate (DAG) therapy of ketamine and perampanel (n = 26), single antiglutamate therapy with either agent (n = 8), or aggressive nonantiglutamate (NAG) therapy with antiepilepsy drugs and anesthetics other than ketamine or perampanel (n = 22).
The single-antiglutamate group was not included in the analysis of patient outcomes.
The DAG and NAG groups were well balanced at baseline. There were no significant differences in median age (60 years vs. 66 years), gender, low cerebral blood flow, presence of bilateral pupillary or corneal reflexes, neuron-specific enolase levels, cortical N20 somatosenory evoked potentials, moderate to severe postanoxic brain injury, and hypothermia/targeted temperature management.
Primary outcome met
More patients in the DAG group (42%) had moderate to severe postanoxic brain injury than in the NAG group (28%). However, the difference was not statistically significant (P = .08), possibly because of the small sample sizes. The number of antiepileptic drugs and the number of cycles of anesthetics did not differ between the groups.
Results showed that efficacy and safety outcomes favored DAG therapy.
The primary efficacy outcome was resolution of status epilepticus within 3 days after initiation of treatments. Status epilepticus resolved for 21 of 26 patients in the DAG group (81%), versus 9 of 22 patients in the NAG group (41%; odds ratio, 6.06; 95% confidence interval, 1.66-22.12; P = .005).
For secondary efficacy outcomes, there was a trend in favor of DAG, but differences from the NAG group were not statistically significant. In the groups, 46% versus 32% awakened and responded to commands before discharge from the intensive care unit, and 32% versus 23% showed good neurologic outcome at 6 months.
The primary safety outcome of all-cause mortality risk in the ICU was 90% lower for patients treated with DAG than for those treated with NAG (15% vs. 64%; OR, 0.1; 95% CI, 0.02-0.41; P < .01). Dr. Beretta explained that the high mortality rate in the NAG group was presumably a result of unresolved status epilepticus.
The secondary safety outcome of a transitory rise of gamma-glutamyl transferase greater than three times the upper limit of normal in the DAG group was expected with high-dose perampanel, the investigators noted. This outcome occurred in 77% of the DAG group versus 27% of the NAG group (OR, 9.88; 95% CI, 2.4-32.9; P < .001).
There was no statistically significant difference in incidence of recurrent cardiac arrest during therapy. This occurred in one member of the DAG group and in none in the NAG group.
Dr. Beretta reported that their investigations are still in a retrospective phase, but the researchers plan to move the work into a prospective phase and possibly a randomized trial soon.
Fascinating, promising
Commenting on the findings, Jaysingh Singh, MD, co-director of the Epilepsy Surgery Center at the Ohio State University Wexner Medical Center, Columbus, said the study provides “fascinating, very helpful data” about a condition that has responded well to current treatment options.
He added that his center has used “the innovative treatments” discussed in the study for a few patients.
“More concrete evidence will push us to use it more uniformly across all our patient population [that] has refractory status. So I’m very optimistic, and the data were very promising,” said Dr. Singh, who was not involved with the research.
He cautioned that the study was retrospective, not randomized or controlled, and that it involved a small number of patients but said that the data were “heading in the right direction.”
Although resolution of status epilepticus was better among patients in the DAG group than in the NAG group, the awakenings and neurologic outcomes were “pretty much same as standard medical therapy, which we commonly give to our patients,” said Dr. Singh. “We see this phenomenon all the time in our patients.”
He noted that other factors can determine how patients respond, such as conditions of the heart or kidneys, the presence of sepsis, and multiorgan dysfunction. These factors were not controlled for in the study.
Nonetheless, he said the study achieved its primary endpoint of better resolution of status epilepticus “because that’s the first thing you want to see: whether the treatment is taking care of that.”
The study received no outside funding. Dr. Beretta and Dr. Singh have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
(SRSE), new research suggests.
In a retrospective cohort study of survivors of cardiac arrest with postanoxic sustained SRSE, resolution of the condition was achieved by 81% of those who received intensive treatment of ketamine plus perampanel, versus 41% of those who received standard care.
The novelty of the new treatment approach is the duration of therapy as well as the dual antiglutamate drugs, researchers note.
“So the logic is to continue treatment until resolution of refractory status epilepticus under continuous EEG [electroencephalographic] monitoring,” reported lead investigator Simone Beretta, MD, San Gerardo University Hospital, Monza, Italy.
Therapy was guided by data on brainstem reflexes, N20 cortical responses, neuronal serum enolase levels, and neuroimaging.
If all or most of these indicators are favorable, “we continue to treat without any time limit,” Dr. Beretta said. However, if the indicators become unfavorable, clinicians should consider lowering the intensity of care, he added.
The findings were presented at the 2021 World Congress of Neurology (WCN).
SUPER-CAT trial
In SRSE, epileptic seizures occur one after another without patients recovering consciousness in between. Standard aggressive therapy for the condition does not include antiglutamatergic drugs, the researchers noted.
In the Super-Refractory Status Epilepticus After Cardiac Arrest: Aggressive Treatment Guided by Multimodal Prognostic Indicators (SUPER-CAT) study, researchers assessed the combination of two such medications.
The first was the anti-NMDA receptor drug ketamine, which was given by intravenous bolus and then continuous infusion for 3 days guided by continuous EEG to reach a ketamine EEG pattern, as evidenced by alpha and beta waves. It was combined with the anti-AMPA receptor antiepileptic perampanel via nasogastric tube for 5 days, followed by slow tapering.
Dr. Beretta noted that in the ongoing TELSTAR trial, which involved a similar patient population, a different drug combination is being used. A major difference between the two trials is that in the TELSTAR trial, aggressive therapy continues for only 2 days if there is no response.
“In the SUPER-CAT study, we continue far beyond 2 days in the majority of patients,” he said. In addition, ketamine and perampanel were not assessed in TELSTAR.
In SUPER-CAT, 489 survivors of cardiac arrest were recruited over 10 years. Of these, 101 had refractory status epilepticus. After excluding those with more than two indicators of poor prognosis (n = 31) or whose status epilepticus resolved (n = 14), 56 patients were determined to have SRSE. All had experienced relapse after undergoing one cycle of anesthetic.
The 56 participants received one of three treatment regimens: double antiglutamate (DAG) therapy of ketamine and perampanel (n = 26), single antiglutamate therapy with either agent (n = 8), or aggressive nonantiglutamate (NAG) therapy with antiepilepsy drugs and anesthetics other than ketamine or perampanel (n = 22).
The single-antiglutamate group was not included in the analysis of patient outcomes.
The DAG and NAG groups were well balanced at baseline. There were no significant differences in median age (60 years vs. 66 years), gender, low cerebral blood flow, presence of bilateral pupillary or corneal reflexes, neuron-specific enolase levels, cortical N20 somatosenory evoked potentials, moderate to severe postanoxic brain injury, and hypothermia/targeted temperature management.
Primary outcome met
More patients in the DAG group (42%) had moderate to severe postanoxic brain injury than in the NAG group (28%). However, the difference was not statistically significant (P = .08), possibly because of the small sample sizes. The number of antiepileptic drugs and the number of cycles of anesthetics did not differ between the groups.
Results showed that efficacy and safety outcomes favored DAG therapy.
The primary efficacy outcome was resolution of status epilepticus within 3 days after initiation of treatments. Status epilepticus resolved for 21 of 26 patients in the DAG group (81%), versus 9 of 22 patients in the NAG group (41%; odds ratio, 6.06; 95% confidence interval, 1.66-22.12; P = .005).
For secondary efficacy outcomes, there was a trend in favor of DAG, but differences from the NAG group were not statistically significant. In the groups, 46% versus 32% awakened and responded to commands before discharge from the intensive care unit, and 32% versus 23% showed good neurologic outcome at 6 months.
The primary safety outcome of all-cause mortality risk in the ICU was 90% lower for patients treated with DAG than for those treated with NAG (15% vs. 64%; OR, 0.1; 95% CI, 0.02-0.41; P < .01). Dr. Beretta explained that the high mortality rate in the NAG group was presumably a result of unresolved status epilepticus.
The secondary safety outcome of a transitory rise of gamma-glutamyl transferase greater than three times the upper limit of normal in the DAG group was expected with high-dose perampanel, the investigators noted. This outcome occurred in 77% of the DAG group versus 27% of the NAG group (OR, 9.88; 95% CI, 2.4-32.9; P < .001).
There was no statistically significant difference in incidence of recurrent cardiac arrest during therapy. This occurred in one member of the DAG group and in none in the NAG group.
Dr. Beretta reported that their investigations are still in a retrospective phase, but the researchers plan to move the work into a prospective phase and possibly a randomized trial soon.
Fascinating, promising
Commenting on the findings, Jaysingh Singh, MD, co-director of the Epilepsy Surgery Center at the Ohio State University Wexner Medical Center, Columbus, said the study provides “fascinating, very helpful data” about a condition that has responded well to current treatment options.
He added that his center has used “the innovative treatments” discussed in the study for a few patients.
“More concrete evidence will push us to use it more uniformly across all our patient population [that] has refractory status. So I’m very optimistic, and the data were very promising,” said Dr. Singh, who was not involved with the research.
He cautioned that the study was retrospective, not randomized or controlled, and that it involved a small number of patients but said that the data were “heading in the right direction.”
Although resolution of status epilepticus was better among patients in the DAG group than in the NAG group, the awakenings and neurologic outcomes were “pretty much same as standard medical therapy, which we commonly give to our patients,” said Dr. Singh. “We see this phenomenon all the time in our patients.”
He noted that other factors can determine how patients respond, such as conditions of the heart or kidneys, the presence of sepsis, and multiorgan dysfunction. These factors were not controlled for in the study.
Nonetheless, he said the study achieved its primary endpoint of better resolution of status epilepticus “because that’s the first thing you want to see: whether the treatment is taking care of that.”
The study received no outside funding. Dr. Beretta and Dr. Singh have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
From WCN 2021
Is genetic testing valuable in the clinical management of epilepsy?
, new research shows.
Results of a survey that included more than 400 patients showed that positive findings from genetic testing helped guide clinical management in 50% of cases and improved patient outcomes in 75%. In addition, the findings were applicable to both children and adults.
“Fifty percent of the time the physicians reported that, yes, receiving the genetic diagnosis did change how they managed the patients,” reported co-investigator Dianalee McKnight, PhD, director of medical affairs at Invitae, a medical genetic testing company headquartered in San Francisco. In 81.3% of cases, providers reported they changed clinical management within 3 months of receiving the genetic results, she added.
The findings were presented at the 2021 World Congress of Neurology (WCN).
Test results can be practice-changing
Nearly 50% of positive genetic test results in epilepsy patients can help guide clinical management, Dr. McKnight noted. However, information on how physicians use genetic information in decision-making has been limited, prompting her conduct the survey.
A total of 1,567 physicians with 3,572 patients who had a definitive diagnosis of epilepsy were contacted. A total of 170 (10.8%) clinicians provided completed and eligible surveys on 429 patients with epilepsy.
The patient cohort comprised mostly children, with nearly 50 adults, which Dr. McKnight said is typical of the population receiving genetic testing in clinical practice.
She reported that genetic testing results prompted clinicians to make medication changes about 50% of the time. Other changes included specialist referral or to a clinical trial, monitoring for other neurological disease, and recommendations for dietary change or for surgery.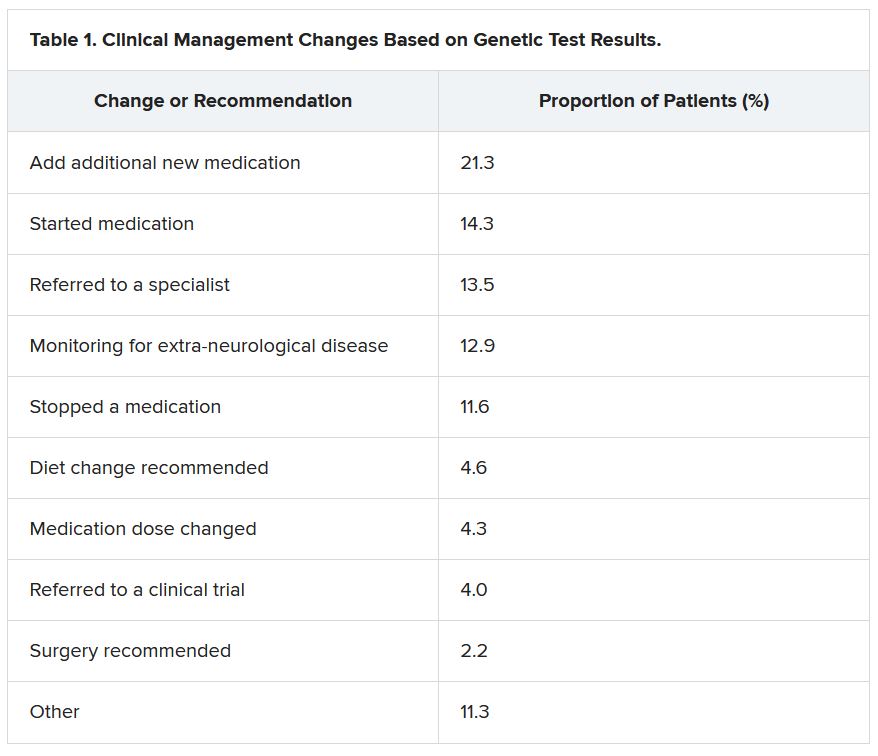
“Of the physicians who changed treatment, 75% reported there were positive outcomes for the patients,” Dr. McKnight told meeting attendees. “Most common was a reduction or a complete elimination of seizures, and that was reported in 65% of the cases.”
In many cases, the changes resulted in clinical improvements.
“There were 64 individuals who were having daily seizures before the genetic testing,” Dr. McKnight reported via email. “After receiving the genetic diagnosis and modifying their treatment, their physicians reported that 26% of individuals had complete seizure control and 46% of individuals had reduced seizure frequency to either weekly (20%), monthly (20%) or annually (6%).”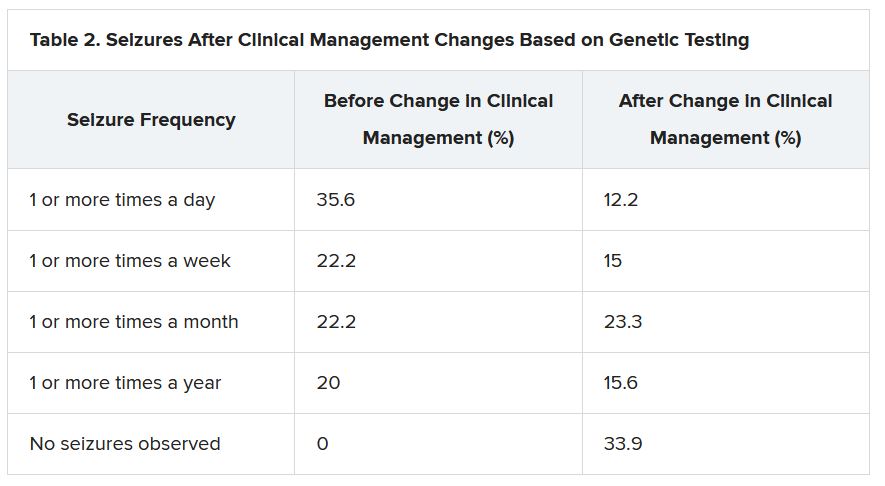
The best seizure control after modifying disease management occurred among children. Although the changes were not as dramatic for adults, they trended toward lower seizure frequency.
“It is still pretty significant that adults can receive genetic testing later in life and still have benefit in controlling their seizures,” Dr. McKnight said.
Twenty-three percent of patients showed improvement in behavior, development, academics, or movement issues, while 6% experienced reduced medication side effects.
Dr. McKnight also explored reasons for physicians not making changes to clinical management of patients based on the genetic results. The most common reason was that management was already consistent with the results (47.3%), followed by the results not being informative (26.1%), the results possibly being useful for future treatments in development (19.0%), or other or unknown reasons (7.6%).
Besides direct health and quality of life benefits from better seizure control, Dr. McKnight cited previous economic studies showing lower health care costs.
“It looked like an individual who has good seizure control will incur about 14,000 U.S. dollars a year compared with an individual with pretty poor seizure control, where it can be closer to 23,000 U.S. dollars a year,” Dr. McKnight said. This is mainly attributed to reduced hospitalizations and emergency department visits.
Dr. McKnight noted that currently there is no cost of genetic testing to the patient, the hospital, or insurers. Pharmaceutical companies, she said, sponsor the testing to potentially gather patients for clinical drug trials in development. However, patients remain completely anonymous.
Physicians who wish to have patient samples tested agree that the companies may contact them to ask if any of their patients with positive genetic test results would like to participate in a trial.
Dr. McKnight noted that genetic testing can be considered actionable in the clinic, helping to guide clinical decision-making and potentially leading to better outcomes. Going forward, she suggested performing large case-controlled studies “of individuals with the same genetic etiology ... to really find a true causation or correlation.”
Growing influence of genetic testing
Commenting on the findings, Jaysingh Singh, MD, co-director of the Epilepsy Surgery Center at the Ohio State University Wexner Medical Center in Columbus, noted that the study highlights the value of gene testing in improving outcomes in patients with epilepsy, particularly the pediatric population.
He said the findings make him optimistic about the potential of genetic testing in adult patients – with at least one caveat.
“The limitation is that if we do find some mutation, we don’t know what to do with that. That’s definitely one challenge. And we see that more often in the adult patient population,” said Dr. Singh, who was not involved with the research.
He noted that there is a small group of genetic mutations when, found in adults, may dramatically alter treatment.
For example, he noted that if there is a gene mutation related to mTOR pathways, that could provide a future target because there are already medications that target this pathway.
Genetic testing may also be useful in cases where patients have normal brain imaging and poor response to standard treatment or in cases where patients have congenital abnormalities such as intellectual impairment or facial dysmorphic features and a co-morbid seizure disorder, he said.
Dr. Singh noted that he has often found genetic testing impractical because “if I order DNA testing right now, it will take 4 months for me to get the results. I cannot wait 4 months for the results to come back” to adjust treatment.
Dr. McKnight is an employee of and a shareholder in Invitae, which funded the study. Dr. Singh has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, new research shows.
Results of a survey that included more than 400 patients showed that positive findings from genetic testing helped guide clinical management in 50% of cases and improved patient outcomes in 75%. In addition, the findings were applicable to both children and adults.
“Fifty percent of the time the physicians reported that, yes, receiving the genetic diagnosis did change how they managed the patients,” reported co-investigator Dianalee McKnight, PhD, director of medical affairs at Invitae, a medical genetic testing company headquartered in San Francisco. In 81.3% of cases, providers reported they changed clinical management within 3 months of receiving the genetic results, she added.
The findings were presented at the 2021 World Congress of Neurology (WCN).
Test results can be practice-changing
Nearly 50% of positive genetic test results in epilepsy patients can help guide clinical management, Dr. McKnight noted. However, information on how physicians use genetic information in decision-making has been limited, prompting her conduct the survey.
A total of 1,567 physicians with 3,572 patients who had a definitive diagnosis of epilepsy were contacted. A total of 170 (10.8%) clinicians provided completed and eligible surveys on 429 patients with epilepsy.
The patient cohort comprised mostly children, with nearly 50 adults, which Dr. McKnight said is typical of the population receiving genetic testing in clinical practice.
She reported that genetic testing results prompted clinicians to make medication changes about 50% of the time. Other changes included specialist referral or to a clinical trial, monitoring for other neurological disease, and recommendations for dietary change or for surgery.
“Of the physicians who changed treatment, 75% reported there were positive outcomes for the patients,” Dr. McKnight told meeting attendees. “Most common was a reduction or a complete elimination of seizures, and that was reported in 65% of the cases.”
In many cases, the changes resulted in clinical improvements.
“There were 64 individuals who were having daily seizures before the genetic testing,” Dr. McKnight reported via email. “After receiving the genetic diagnosis and modifying their treatment, their physicians reported that 26% of individuals had complete seizure control and 46% of individuals had reduced seizure frequency to either weekly (20%), monthly (20%) or annually (6%).”
The best seizure control after modifying disease management occurred among children. Although the changes were not as dramatic for adults, they trended toward lower seizure frequency.
“It is still pretty significant that adults can receive genetic testing later in life and still have benefit in controlling their seizures,” Dr. McKnight said.
Twenty-three percent of patients showed improvement in behavior, development, academics, or movement issues, while 6% experienced reduced medication side effects.
Dr. McKnight also explored reasons for physicians not making changes to clinical management of patients based on the genetic results. The most common reason was that management was already consistent with the results (47.3%), followed by the results not being informative (26.1%), the results possibly being useful for future treatments in development (19.0%), or other or unknown reasons (7.6%).
Besides direct health and quality of life benefits from better seizure control, Dr. McKnight cited previous economic studies showing lower health care costs.
“It looked like an individual who has good seizure control will incur about 14,000 U.S. dollars a year compared with an individual with pretty poor seizure control, where it can be closer to 23,000 U.S. dollars a year,” Dr. McKnight said. This is mainly attributed to reduced hospitalizations and emergency department visits.
Dr. McKnight noted that currently there is no cost of genetic testing to the patient, the hospital, or insurers. Pharmaceutical companies, she said, sponsor the testing to potentially gather patients for clinical drug trials in development. However, patients remain completely anonymous.
Physicians who wish to have patient samples tested agree that the companies may contact them to ask if any of their patients with positive genetic test results would like to participate in a trial.
Dr. McKnight noted that genetic testing can be considered actionable in the clinic, helping to guide clinical decision-making and potentially leading to better outcomes. Going forward, she suggested performing large case-controlled studies “of individuals with the same genetic etiology ... to really find a true causation or correlation.”
Growing influence of genetic testing
Commenting on the findings, Jaysingh Singh, MD, co-director of the Epilepsy Surgery Center at the Ohio State University Wexner Medical Center in Columbus, noted that the study highlights the value of gene testing in improving outcomes in patients with epilepsy, particularly the pediatric population.
He said the findings make him optimistic about the potential of genetic testing in adult patients – with at least one caveat.
“The limitation is that if we do find some mutation, we don’t know what to do with that. That’s definitely one challenge. And we see that more often in the adult patient population,” said Dr. Singh, who was not involved with the research.
He noted that there is a small group of genetic mutations when, found in adults, may dramatically alter treatment.
For example, he noted that if there is a gene mutation related to mTOR pathways, that could provide a future target because there are already medications that target this pathway.
Genetic testing may also be useful in cases where patients have normal brain imaging and poor response to standard treatment or in cases where patients have congenital abnormalities such as intellectual impairment or facial dysmorphic features and a co-morbid seizure disorder, he said.
Dr. Singh noted that he has often found genetic testing impractical because “if I order DNA testing right now, it will take 4 months for me to get the results. I cannot wait 4 months for the results to come back” to adjust treatment.
Dr. McKnight is an employee of and a shareholder in Invitae, which funded the study. Dr. Singh has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, new research shows.
Results of a survey that included more than 400 patients showed that positive findings from genetic testing helped guide clinical management in 50% of cases and improved patient outcomes in 75%. In addition, the findings were applicable to both children and adults.
“Fifty percent of the time the physicians reported that, yes, receiving the genetic diagnosis did change how they managed the patients,” reported co-investigator Dianalee McKnight, PhD, director of medical affairs at Invitae, a medical genetic testing company headquartered in San Francisco. In 81.3% of cases, providers reported they changed clinical management within 3 months of receiving the genetic results, she added.
The findings were presented at the 2021 World Congress of Neurology (WCN).
Test results can be practice-changing
Nearly 50% of positive genetic test results in epilepsy patients can help guide clinical management, Dr. McKnight noted. However, information on how physicians use genetic information in decision-making has been limited, prompting her conduct the survey.
A total of 1,567 physicians with 3,572 patients who had a definitive diagnosis of epilepsy were contacted. A total of 170 (10.8%) clinicians provided completed and eligible surveys on 429 patients with epilepsy.
The patient cohort comprised mostly children, with nearly 50 adults, which Dr. McKnight said is typical of the population receiving genetic testing in clinical practice.
She reported that genetic testing results prompted clinicians to make medication changes about 50% of the time. Other changes included specialist referral or to a clinical trial, monitoring for other neurological disease, and recommendations for dietary change or for surgery.
“Of the physicians who changed treatment, 75% reported there were positive outcomes for the patients,” Dr. McKnight told meeting attendees. “Most common was a reduction or a complete elimination of seizures, and that was reported in 65% of the cases.”
In many cases, the changes resulted in clinical improvements.
“There were 64 individuals who were having daily seizures before the genetic testing,” Dr. McKnight reported via email. “After receiving the genetic diagnosis and modifying their treatment, their physicians reported that 26% of individuals had complete seizure control and 46% of individuals had reduced seizure frequency to either weekly (20%), monthly (20%) or annually (6%).”
The best seizure control after modifying disease management occurred among children. Although the changes were not as dramatic for adults, they trended toward lower seizure frequency.
“It is still pretty significant that adults can receive genetic testing later in life and still have benefit in controlling their seizures,” Dr. McKnight said.
Twenty-three percent of patients showed improvement in behavior, development, academics, or movement issues, while 6% experienced reduced medication side effects.
Dr. McKnight also explored reasons for physicians not making changes to clinical management of patients based on the genetic results. The most common reason was that management was already consistent with the results (47.3%), followed by the results not being informative (26.1%), the results possibly being useful for future treatments in development (19.0%), or other or unknown reasons (7.6%).
Besides direct health and quality of life benefits from better seizure control, Dr. McKnight cited previous economic studies showing lower health care costs.
“It looked like an individual who has good seizure control will incur about 14,000 U.S. dollars a year compared with an individual with pretty poor seizure control, where it can be closer to 23,000 U.S. dollars a year,” Dr. McKnight said. This is mainly attributed to reduced hospitalizations and emergency department visits.
Dr. McKnight noted that currently there is no cost of genetic testing to the patient, the hospital, or insurers. Pharmaceutical companies, she said, sponsor the testing to potentially gather patients for clinical drug trials in development. However, patients remain completely anonymous.
Physicians who wish to have patient samples tested agree that the companies may contact them to ask if any of their patients with positive genetic test results would like to participate in a trial.
Dr. McKnight noted that genetic testing can be considered actionable in the clinic, helping to guide clinical decision-making and potentially leading to better outcomes. Going forward, she suggested performing large case-controlled studies “of individuals with the same genetic etiology ... to really find a true causation or correlation.”
Growing influence of genetic testing
Commenting on the findings, Jaysingh Singh, MD, co-director of the Epilepsy Surgery Center at the Ohio State University Wexner Medical Center in Columbus, noted that the study highlights the value of gene testing in improving outcomes in patients with epilepsy, particularly the pediatric population.
He said the findings make him optimistic about the potential of genetic testing in adult patients – with at least one caveat.
“The limitation is that if we do find some mutation, we don’t know what to do with that. That’s definitely one challenge. And we see that more often in the adult patient population,” said Dr. Singh, who was not involved with the research.
He noted that there is a small group of genetic mutations when, found in adults, may dramatically alter treatment.
For example, he noted that if there is a gene mutation related to mTOR pathways, that could provide a future target because there are already medications that target this pathway.
Genetic testing may also be useful in cases where patients have normal brain imaging and poor response to standard treatment or in cases where patients have congenital abnormalities such as intellectual impairment or facial dysmorphic features and a co-morbid seizure disorder, he said.
Dr. Singh noted that he has often found genetic testing impractical because “if I order DNA testing right now, it will take 4 months for me to get the results. I cannot wait 4 months for the results to come back” to adjust treatment.
Dr. McKnight is an employee of and a shareholder in Invitae, which funded the study. Dr. Singh has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
From WCN 2021
Merck seeks FDA authorization for antiviral COVID-19 pill
, an experimental antiviral COVID-19 treatment.
If the FDA grants authorization, the drug would be the first oral antiviral treatment for COVID-19. The capsule, made by Merck and Ridgeback Biotherapeutics, is intended to treat mild to moderate COVID-19 in adults who are at risk of having severe COVID-19 or hospitalization.
“The extraordinary impact of this pandemic demands that we move with unprecedented urgency, and that is what our teams have done by submitting this application for molnupiravir to the FDA within 10 days of receiving the data,” Robert Davis, CEO and president of Merck, said in a statement. On Oct. 1, Merck and Ridgeback released interim data from its phase III clinical trial, which showed that molnupiravir reduced the risk of hospitalization or death by about 50%. About 7% of patients who received the drug were hospitalized within 30 days in the study, as compared with 14% of patients who took a placebo, the company said.
No deaths were reported in the group that received the drug, as compared with eight deaths in the group that received the placebo. None of the trial participants had been vaccinated.
“Medicines and vaccines are both essential to our collective efforts,” Mr. Davis said. “We look forward to working with the FDA on its review of our application, and to working with other regulatory agencies as we do everything we can to bring molnupiravir to patients around the world as quickly as possible.”
Merck has been producing molnupiravir in anticipation of the clinical trial results and FDA authorization. The company expects to produce 10 million courses of treatment by the end of the year, with more expected for 2022.
In June, Merck signed an agreement with the United States to supply 1.7 million courses of molnupiravir once the FDA authorizes the drug. The company has agreed to advance purchase agreements with other countries as well.
Earlier in the year, Merck also announced voluntary licensing agreements with several generics manufacturers in India to provide molnupiravir to more than 100 low- and middle-income countries after approval from local regulatory agencies.
Data from the company’s late-stage clinical trial has not yet been peer-reviewed or published.
Last week, Anthony Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, said the clinical trial results were “very encouraging” but noted that the FDA should closely scrutinize the drug, CNN reported.
“It is very important that this now must go through the usual process of careful examination of the data by the Food and Drug Administration, both for effectiveness but also for safety, because whenever you introduce a new compound, safety is very important,” Dr. Fauci said, adding that vaccines remain “our best tools against COVID-19.”
A version of this article firsts appeared on WebMD.com.
, an experimental antiviral COVID-19 treatment.
If the FDA grants authorization, the drug would be the first oral antiviral treatment for COVID-19. The capsule, made by Merck and Ridgeback Biotherapeutics, is intended to treat mild to moderate COVID-19 in adults who are at risk of having severe COVID-19 or hospitalization.
“The extraordinary impact of this pandemic demands that we move with unprecedented urgency, and that is what our teams have done by submitting this application for molnupiravir to the FDA within 10 days of receiving the data,” Robert Davis, CEO and president of Merck, said in a statement. On Oct. 1, Merck and Ridgeback released interim data from its phase III clinical trial, which showed that molnupiravir reduced the risk of hospitalization or death by about 50%. About 7% of patients who received the drug were hospitalized within 30 days in the study, as compared with 14% of patients who took a placebo, the company said.
No deaths were reported in the group that received the drug, as compared with eight deaths in the group that received the placebo. None of the trial participants had been vaccinated.
“Medicines and vaccines are both essential to our collective efforts,” Mr. Davis said. “We look forward to working with the FDA on its review of our application, and to working with other regulatory agencies as we do everything we can to bring molnupiravir to patients around the world as quickly as possible.”
Merck has been producing molnupiravir in anticipation of the clinical trial results and FDA authorization. The company expects to produce 10 million courses of treatment by the end of the year, with more expected for 2022.
In June, Merck signed an agreement with the United States to supply 1.7 million courses of molnupiravir once the FDA authorizes the drug. The company has agreed to advance purchase agreements with other countries as well.
Earlier in the year, Merck also announced voluntary licensing agreements with several generics manufacturers in India to provide molnupiravir to more than 100 low- and middle-income countries after approval from local regulatory agencies.
Data from the company’s late-stage clinical trial has not yet been peer-reviewed or published.
Last week, Anthony Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, said the clinical trial results were “very encouraging” but noted that the FDA should closely scrutinize the drug, CNN reported.
“It is very important that this now must go through the usual process of careful examination of the data by the Food and Drug Administration, both for effectiveness but also for safety, because whenever you introduce a new compound, safety is very important,” Dr. Fauci said, adding that vaccines remain “our best tools against COVID-19.”
A version of this article firsts appeared on WebMD.com.
, an experimental antiviral COVID-19 treatment.
If the FDA grants authorization, the drug would be the first oral antiviral treatment for COVID-19. The capsule, made by Merck and Ridgeback Biotherapeutics, is intended to treat mild to moderate COVID-19 in adults who are at risk of having severe COVID-19 or hospitalization.
“The extraordinary impact of this pandemic demands that we move with unprecedented urgency, and that is what our teams have done by submitting this application for molnupiravir to the FDA within 10 days of receiving the data,” Robert Davis, CEO and president of Merck, said in a statement. On Oct. 1, Merck and Ridgeback released interim data from its phase III clinical trial, which showed that molnupiravir reduced the risk of hospitalization or death by about 50%. About 7% of patients who received the drug were hospitalized within 30 days in the study, as compared with 14% of patients who took a placebo, the company said.
No deaths were reported in the group that received the drug, as compared with eight deaths in the group that received the placebo. None of the trial participants had been vaccinated.
“Medicines and vaccines are both essential to our collective efforts,” Mr. Davis said. “We look forward to working with the FDA on its review of our application, and to working with other regulatory agencies as we do everything we can to bring molnupiravir to patients around the world as quickly as possible.”
Merck has been producing molnupiravir in anticipation of the clinical trial results and FDA authorization. The company expects to produce 10 million courses of treatment by the end of the year, with more expected for 2022.
In June, Merck signed an agreement with the United States to supply 1.7 million courses of molnupiravir once the FDA authorizes the drug. The company has agreed to advance purchase agreements with other countries as well.
Earlier in the year, Merck also announced voluntary licensing agreements with several generics manufacturers in India to provide molnupiravir to more than 100 low- and middle-income countries after approval from local regulatory agencies.
Data from the company’s late-stage clinical trial has not yet been peer-reviewed or published.
Last week, Anthony Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, said the clinical trial results were “very encouraging” but noted that the FDA should closely scrutinize the drug, CNN reported.
“It is very important that this now must go through the usual process of careful examination of the data by the Food and Drug Administration, both for effectiveness but also for safety, because whenever you introduce a new compound, safety is very important,” Dr. Fauci said, adding that vaccines remain “our best tools against COVID-19.”
A version of this article firsts appeared on WebMD.com.
HEPA filters may clean SARS-CoV-2 from the air: Study
, researchers report in the preprint server medRxiv.
The journal Nature reported Oct. 6 that the research, which has not been peer-reviewed, suggests the filters may help reduce the risk of hospital-acquired SARS-CoV-2.
Researchers, led by intensivist Andrew Conway-Morris, MBChB, PhD, with the division of anaesthesia in the school of clinical medicine at University of Cambridge, United Kingdom, write that earlier experiments assessed air filters’ ability to remove inactive particles in carefully controlled environments, but it was unknown how they would work in a real-world setting.
Co-author Vilas Navapurkar, MBChB, an ICU physician at Addenbrooke’s Hospital in Cambridge, United Kingdom, said that hospitals have used portable air filters when their isolation facilities are full, but evidence was needed as to whether such filters are effective or whether they provide a false sense of security.
The researchers installed the filters in two fully occupied COVID-19 wards — a general ward and an ICU. They chose HEPA filters because they can catch extremely small particles.
The team collected air samples from the wards during a week when the air filters were on and 2 weeks when they were turned off, then compared results.
According to the study, “airborne SARS-CoV-2 was detected in the ward on all five days before activation of air/UV filtration, but on none of the five days when the air/UV filter was operational; SARS-CoV-2 was again detected on four out of five days when the filter was off.”
Airborne SARS-CoV-2 was not frequently detected in the ICU, even when the filters were off.
Cheap and easy
According to the Nature article, the authors suggest several potential explanations for this, “including slower viral replication at later stages of the disease.” Therefore, the authors say, filtering the virus from the air might be more important in general wards than in ICUs.
The filters significantly reduced the other microbial bioaerosols in both the ward (48 pathogens detected before filtration, 2 after, P = .05) and the ICU (45 pathogens detected before filtration, 5 after P = .05).
National Institute for Occupational Safety and Health (NIOSH) cyclonic aerosol samplers and PCR tests were used to detect airborne SARS-CoV-2 and other microbial bioaerosol.
David Fisman, MD, an epidemiologist at the University of Toronto, who was not involved in the research, said in the Nature article, “This study suggests that HEPA air cleaners, which remain little-used in Canadian hospitals, are a cheap and easy way to reduce risk from airborne pathogens.”This work was supported by a Wellcome senior research fellowship to co-author Stephen Baker. Conway Morris is supported by a Clinician Scientist Fellowship from the Medical Research Council. Dr. Navapurkar is the founder, director, and shareholder of Cambridge Infection Diagnostics Ltd. Dr. Conway-Morris and several co-authors are members of the Scientific Advisory Board of Cambridge Infection Diagnostics Ltd. Co-author Theodore Gouliouris has received a research grant from Shionogi and co-author R. Andres Floto has received research grants and/or consultancy payments from GSK, AstraZeneca, Chiesi, Shionogi, Insmed, and Thirty Technology.
A version of this article first appeared on Medscape.com.
, researchers report in the preprint server medRxiv.
The journal Nature reported Oct. 6 that the research, which has not been peer-reviewed, suggests the filters may help reduce the risk of hospital-acquired SARS-CoV-2.
Researchers, led by intensivist Andrew Conway-Morris, MBChB, PhD, with the division of anaesthesia in the school of clinical medicine at University of Cambridge, United Kingdom, write that earlier experiments assessed air filters’ ability to remove inactive particles in carefully controlled environments, but it was unknown how they would work in a real-world setting.
Co-author Vilas Navapurkar, MBChB, an ICU physician at Addenbrooke’s Hospital in Cambridge, United Kingdom, said that hospitals have used portable air filters when their isolation facilities are full, but evidence was needed as to whether such filters are effective or whether they provide a false sense of security.
The researchers installed the filters in two fully occupied COVID-19 wards — a general ward and an ICU. They chose HEPA filters because they can catch extremely small particles.
The team collected air samples from the wards during a week when the air filters were on and 2 weeks when they were turned off, then compared results.
According to the study, “airborne SARS-CoV-2 was detected in the ward on all five days before activation of air/UV filtration, but on none of the five days when the air/UV filter was operational; SARS-CoV-2 was again detected on four out of five days when the filter was off.”
Airborne SARS-CoV-2 was not frequently detected in the ICU, even when the filters were off.
Cheap and easy
According to the Nature article, the authors suggest several potential explanations for this, “including slower viral replication at later stages of the disease.” Therefore, the authors say, filtering the virus from the air might be more important in general wards than in ICUs.
The filters significantly reduced the other microbial bioaerosols in both the ward (48 pathogens detected before filtration, 2 after, P = .05) and the ICU (45 pathogens detected before filtration, 5 after P = .05).
National Institute for Occupational Safety and Health (NIOSH) cyclonic aerosol samplers and PCR tests were used to detect airborne SARS-CoV-2 and other microbial bioaerosol.
David Fisman, MD, an epidemiologist at the University of Toronto, who was not involved in the research, said in the Nature article, “This study suggests that HEPA air cleaners, which remain little-used in Canadian hospitals, are a cheap and easy way to reduce risk from airborne pathogens.”This work was supported by a Wellcome senior research fellowship to co-author Stephen Baker. Conway Morris is supported by a Clinician Scientist Fellowship from the Medical Research Council. Dr. Navapurkar is the founder, director, and shareholder of Cambridge Infection Diagnostics Ltd. Dr. Conway-Morris and several co-authors are members of the Scientific Advisory Board of Cambridge Infection Diagnostics Ltd. Co-author Theodore Gouliouris has received a research grant from Shionogi and co-author R. Andres Floto has received research grants and/or consultancy payments from GSK, AstraZeneca, Chiesi, Shionogi, Insmed, and Thirty Technology.
A version of this article first appeared on Medscape.com.
, researchers report in the preprint server medRxiv.
The journal Nature reported Oct. 6 that the research, which has not been peer-reviewed, suggests the filters may help reduce the risk of hospital-acquired SARS-CoV-2.
Researchers, led by intensivist Andrew Conway-Morris, MBChB, PhD, with the division of anaesthesia in the school of clinical medicine at University of Cambridge, United Kingdom, write that earlier experiments assessed air filters’ ability to remove inactive particles in carefully controlled environments, but it was unknown how they would work in a real-world setting.
Co-author Vilas Navapurkar, MBChB, an ICU physician at Addenbrooke’s Hospital in Cambridge, United Kingdom, said that hospitals have used portable air filters when their isolation facilities are full, but evidence was needed as to whether such filters are effective or whether they provide a false sense of security.
The researchers installed the filters in two fully occupied COVID-19 wards — a general ward and an ICU. They chose HEPA filters because they can catch extremely small particles.
The team collected air samples from the wards during a week when the air filters were on and 2 weeks when they were turned off, then compared results.
According to the study, “airborne SARS-CoV-2 was detected in the ward on all five days before activation of air/UV filtration, but on none of the five days when the air/UV filter was operational; SARS-CoV-2 was again detected on four out of five days when the filter was off.”
Airborne SARS-CoV-2 was not frequently detected in the ICU, even when the filters were off.
Cheap and easy
According to the Nature article, the authors suggest several potential explanations for this, “including slower viral replication at later stages of the disease.” Therefore, the authors say, filtering the virus from the air might be more important in general wards than in ICUs.
The filters significantly reduced the other microbial bioaerosols in both the ward (48 pathogens detected before filtration, 2 after, P = .05) and the ICU (45 pathogens detected before filtration, 5 after P = .05).
National Institute for Occupational Safety and Health (NIOSH) cyclonic aerosol samplers and PCR tests were used to detect airborne SARS-CoV-2 and other microbial bioaerosol.
David Fisman, MD, an epidemiologist at the University of Toronto, who was not involved in the research, said in the Nature article, “This study suggests that HEPA air cleaners, which remain little-used in Canadian hospitals, are a cheap and easy way to reduce risk from airborne pathogens.”This work was supported by a Wellcome senior research fellowship to co-author Stephen Baker. Conway Morris is supported by a Clinician Scientist Fellowship from the Medical Research Council. Dr. Navapurkar is the founder, director, and shareholder of Cambridge Infection Diagnostics Ltd. Dr. Conway-Morris and several co-authors are members of the Scientific Advisory Board of Cambridge Infection Diagnostics Ltd. Co-author Theodore Gouliouris has received a research grant from Shionogi and co-author R. Andres Floto has received research grants and/or consultancy payments from GSK, AstraZeneca, Chiesi, Shionogi, Insmed, and Thirty Technology.
A version of this article first appeared on Medscape.com.
Adolescents who exercised after a concussion recovered faster in RCT
After a concussion, resuming aerobic exercise relatively early on – at an intensity that does not worsen symptoms – may help young athletes recover sooner, compared with stretching, a randomized controlled trial (RCT) shows.
The study adds to emerging evidence that clinicians should prescribe exercise, rather than strict rest, to facilitate concussion recovery, researchers said.
Tamara McLeod, PhD, ATC, professor and director of athletic training programs at A.T. Still University in Mesa, Ariz., hopes the findings help clinicians see that “this is an approach that should be taken.”
“Too often with concussion, patients are given a laundry list of things they are NOT allowed to do,” including sports, school, and social activities, said Dr. McLeod, who was not involved in the study.
The research, published in The Lancet Child & Adolescent Health, largely replicates the findings of a prior trial while addressing limitations of the previous study’s design, researchers said.
For the trial, John J. Leddy, MD, with the State University of New York at Buffalo and colleagues recruited 118 male and female adolescent athletes aged 13-18 years who had had a sport-related concussion in the past 10 days. Investigators at three community and hospital-affiliated sports medicine concussion centers in the United States randomly assigned the athletes to individualized subsymptom-threshold aerobic exercise (61 participants) or stretching exercise (57 participants) at least 20 minutes per day for up to 4 weeks. Aerobic exercise included walking, jogging, or stationary cycling at home.
“It is important that the general clinician community appreciates that prolonged rest and avoidance of physical activity until spontaneous symptom resolution is no longer an acceptable approach to caring for adolescents with concussion,” Dr. Leddy and coauthors said.
The investigators improved on the “the scientific rigor of their previous RCT by including intention-to-treat and per-protocol analyses, daily symptom reporting, objective exercise adherence measurements, and greater heterogeneity of concussion severity,” said Carolyn A. Emery, PhD, and Jonathan Smirl, PhD, both with the University of Calgary (Alta.), in a related commentary. The new study is the first to show that early targeted heart rate subsymptom-threshold aerobic exercise, relative to stretching, shortened recovery time within 4 weeks after sport-related concussion (hazard ratio, 0.52) when controlling for sex, study site, and average daily exercise time, Dr. Emery and Dr. Smirl said.
A larger proportion of athletes assigned to stretching did not recover by 4 weeks, compared with those assigned to aerobic exercise (32% vs. 21%). The median time to full recovery was longer for the stretching group than for the aerobic exercise group (19 days vs. 14 days).
Among athletes who adhered to their assigned regimens, the differences were more pronounced: The median recovery time was 21 days for the stretching group, compared with 12 days for the aerobic exercise group. The rate of postconcussion symptoms beyond 28 days was 9% in the aerobic exercise group versus 31% in the stretching group, among adherent participants.
More research is needed to establish the efficacy of postconcussion aerobic exercise in adults and for nonsport injury, the researchers noted. Possible mechanisms underlying aerobic exercise’s benefits could include increased parasympathetic autonomic tone, improved cerebral blood flow regulation, or enhanced neuron repair, they suggested.
The right amount and timing of exercise, and doing so at an intensity that does not exacerbate symptoms, may be key. Other research has suggested that too much exercise, too soon may delay recovery, Dr. Emery said in an interview. “But there is now a lot of evidence to support low and moderate levels of physical activity to expedite recovery,” she said.
The study was funded by the American Medical Society for Sports Medicine. The study and commentary authors and Dr. McLeod had no disclosures.
After a concussion, resuming aerobic exercise relatively early on – at an intensity that does not worsen symptoms – may help young athletes recover sooner, compared with stretching, a randomized controlled trial (RCT) shows.
The study adds to emerging evidence that clinicians should prescribe exercise, rather than strict rest, to facilitate concussion recovery, researchers said.
Tamara McLeod, PhD, ATC, professor and director of athletic training programs at A.T. Still University in Mesa, Ariz., hopes the findings help clinicians see that “this is an approach that should be taken.”
“Too often with concussion, patients are given a laundry list of things they are NOT allowed to do,” including sports, school, and social activities, said Dr. McLeod, who was not involved in the study.
The research, published in The Lancet Child & Adolescent Health, largely replicates the findings of a prior trial while addressing limitations of the previous study’s design, researchers said.
For the trial, John J. Leddy, MD, with the State University of New York at Buffalo and colleagues recruited 118 male and female adolescent athletes aged 13-18 years who had had a sport-related concussion in the past 10 days. Investigators at three community and hospital-affiliated sports medicine concussion centers in the United States randomly assigned the athletes to individualized subsymptom-threshold aerobic exercise (61 participants) or stretching exercise (57 participants) at least 20 minutes per day for up to 4 weeks. Aerobic exercise included walking, jogging, or stationary cycling at home.
“It is important that the general clinician community appreciates that prolonged rest and avoidance of physical activity until spontaneous symptom resolution is no longer an acceptable approach to caring for adolescents with concussion,” Dr. Leddy and coauthors said.
The investigators improved on the “the scientific rigor of their previous RCT by including intention-to-treat and per-protocol analyses, daily symptom reporting, objective exercise adherence measurements, and greater heterogeneity of concussion severity,” said Carolyn A. Emery, PhD, and Jonathan Smirl, PhD, both with the University of Calgary (Alta.), in a related commentary. The new study is the first to show that early targeted heart rate subsymptom-threshold aerobic exercise, relative to stretching, shortened recovery time within 4 weeks after sport-related concussion (hazard ratio, 0.52) when controlling for sex, study site, and average daily exercise time, Dr. Emery and Dr. Smirl said.
A larger proportion of athletes assigned to stretching did not recover by 4 weeks, compared with those assigned to aerobic exercise (32% vs. 21%). The median time to full recovery was longer for the stretching group than for the aerobic exercise group (19 days vs. 14 days).
Among athletes who adhered to their assigned regimens, the differences were more pronounced: The median recovery time was 21 days for the stretching group, compared with 12 days for the aerobic exercise group. The rate of postconcussion symptoms beyond 28 days was 9% in the aerobic exercise group versus 31% in the stretching group, among adherent participants.
More research is needed to establish the efficacy of postconcussion aerobic exercise in adults and for nonsport injury, the researchers noted. Possible mechanisms underlying aerobic exercise’s benefits could include increased parasympathetic autonomic tone, improved cerebral blood flow regulation, or enhanced neuron repair, they suggested.
The right amount and timing of exercise, and doing so at an intensity that does not exacerbate symptoms, may be key. Other research has suggested that too much exercise, too soon may delay recovery, Dr. Emery said in an interview. “But there is now a lot of evidence to support low and moderate levels of physical activity to expedite recovery,” she said.
The study was funded by the American Medical Society for Sports Medicine. The study and commentary authors and Dr. McLeod had no disclosures.
After a concussion, resuming aerobic exercise relatively early on – at an intensity that does not worsen symptoms – may help young athletes recover sooner, compared with stretching, a randomized controlled trial (RCT) shows.
The study adds to emerging evidence that clinicians should prescribe exercise, rather than strict rest, to facilitate concussion recovery, researchers said.
Tamara McLeod, PhD, ATC, professor and director of athletic training programs at A.T. Still University in Mesa, Ariz., hopes the findings help clinicians see that “this is an approach that should be taken.”
“Too often with concussion, patients are given a laundry list of things they are NOT allowed to do,” including sports, school, and social activities, said Dr. McLeod, who was not involved in the study.
The research, published in The Lancet Child & Adolescent Health, largely replicates the findings of a prior trial while addressing limitations of the previous study’s design, researchers said.
For the trial, John J. Leddy, MD, with the State University of New York at Buffalo and colleagues recruited 118 male and female adolescent athletes aged 13-18 years who had had a sport-related concussion in the past 10 days. Investigators at three community and hospital-affiliated sports medicine concussion centers in the United States randomly assigned the athletes to individualized subsymptom-threshold aerobic exercise (61 participants) or stretching exercise (57 participants) at least 20 minutes per day for up to 4 weeks. Aerobic exercise included walking, jogging, or stationary cycling at home.
“It is important that the general clinician community appreciates that prolonged rest and avoidance of physical activity until spontaneous symptom resolution is no longer an acceptable approach to caring for adolescents with concussion,” Dr. Leddy and coauthors said.
The investigators improved on the “the scientific rigor of their previous RCT by including intention-to-treat and per-protocol analyses, daily symptom reporting, objective exercise adherence measurements, and greater heterogeneity of concussion severity,” said Carolyn A. Emery, PhD, and Jonathan Smirl, PhD, both with the University of Calgary (Alta.), in a related commentary. The new study is the first to show that early targeted heart rate subsymptom-threshold aerobic exercise, relative to stretching, shortened recovery time within 4 weeks after sport-related concussion (hazard ratio, 0.52) when controlling for sex, study site, and average daily exercise time, Dr. Emery and Dr. Smirl said.
A larger proportion of athletes assigned to stretching did not recover by 4 weeks, compared with those assigned to aerobic exercise (32% vs. 21%). The median time to full recovery was longer for the stretching group than for the aerobic exercise group (19 days vs. 14 days).
Among athletes who adhered to their assigned regimens, the differences were more pronounced: The median recovery time was 21 days for the stretching group, compared with 12 days for the aerobic exercise group. The rate of postconcussion symptoms beyond 28 days was 9% in the aerobic exercise group versus 31% in the stretching group, among adherent participants.
More research is needed to establish the efficacy of postconcussion aerobic exercise in adults and for nonsport injury, the researchers noted. Possible mechanisms underlying aerobic exercise’s benefits could include increased parasympathetic autonomic tone, improved cerebral blood flow regulation, or enhanced neuron repair, they suggested.
The right amount and timing of exercise, and doing so at an intensity that does not exacerbate symptoms, may be key. Other research has suggested that too much exercise, too soon may delay recovery, Dr. Emery said in an interview. “But there is now a lot of evidence to support low and moderate levels of physical activity to expedite recovery,” she said.
The study was funded by the American Medical Society for Sports Medicine. The study and commentary authors and Dr. McLeod had no disclosures.
FROM THE LANCET CHILD & ADOLESCENT HEALTH
Retraining the brain may eliminate chronic back pain
Psychological therapy that changes an individual’s beliefs about pain not only provides lasting chronic pain relief but also alters brain regions related to pain generation, new research shows.
In the first randomized controlled test of pain-reprocessing therapy (PRT), two-thirds of patients with chronic back pain (CBP) who received 4 weeks of PRT were pain free or nearly pain free afterward – and for most patients, relief was maintained for 1 year, the researchers found.
“Primary chronic back pain can be dramatically reduced or even eliminated by psychological treatment focused on changing how threatening we perceive the pain to be,” first author Yoni Ashar, PhD, department of psychiatry, Weill Cornell Medicine, New York, said in an interview.
“ given that large reductions in pain have rarely been observed in studies that tested psychological therapies for chronic back pain.
The study was published online Sept. 29, 2021, in JAMA Psychiatry.
Rethinking pain
CBP is a leading cause of disability, and treatment is often ineffective. In about 85% of cases of primary CBP, a definitive cause of the pain can’t be identified. In these cases, fear, avoidance, and beliefs that pain indicates injury may contribute to ongoing CBP.
PRT educates patients about the role of the brain in generating chronic pain; helps them reappraise their pain as they engage in movements that they had been afraid to undertake; and helps them address emotions that may exacerbate pain.
The study included 151 adults (54% women; mean age, 41 years) who had primary CBP of low to moderate severity (mean pain intensity, 4 of 10) for an average of 10 years.
A total of 50 participants were randomly allocated to undergo PRT (one telehealth session with a physician and eight PRT sessions over 4 weeks), 51 to receive placebo (subcutaneous saline injection in the back), and 50 to continue their routine, usual ongoing care.
Large group differences in pain were observed after treatment. The mean pain score was 1.18 in the PRT group, 2.84 in the placebo group, and 3.13 in the usual-care group. Hedges’ g was –1.14 for PRT versus placebo and –1.74 for PRT versus usual care (P < .001).
Two-thirds (66%) of adults in the PRT group were pain free or nearly pain free following treatment (pain-intensity score of 0 or 1 out of 10), compared with 20% of those in the placebo group and 10% of those who received usual care.
Treatment effects were maintained at 1-year follow-up. The mean pain score was 1.51 in the PRT group, 2.79 in the placebo group, and 3.00 in the usual-care group. Neither age nor sex moderated the effect of PRT on pain intensity.
Retraining the brain
The researchers said the effects of PRT on pain were mediated by lessening the belief that pain indicates tissue damage. Of note, PRT also reduced experimentally evoked back pain and spontaneous pain during functional MRI, with large effect sizes.
“The idea is that by thinking about the pain as safe rather than threatening, patients can alter the brain networks reinforcing the pain, and neutralize it,” Dr. Ashar said in a news release.
The authors noted that study participants were relatively well educated and active. The participants reported having longstanding low to moderate pain and disability at baseline.
The physician and therapists were experts in delivering PRT. Future studies should test generalizability to other patient populations, therapists, and treatment contexts.
“Our clinical experience shows that PRT is effective for other primary chronic pain conditions as well,” said Dr. Ashar, including primary knee pain and tension headache.
Restoring function
Commenting on the findings, Shaheen E. Lakhan, MD, PhD, neurologist and pain specialist in Newton, Mass., said he has long experience using psychological approaches to address pain, with good results.
“Imagine telling a person suffering from decades of chronic pain that your pain is all in your head. I’ve done that for years as a board-certified pain physician managing only the most severe and debilitating forms of pain. When used to ground brain retraining, I could ultimately restore function to people living with chronic pain,” Dr. Lakhan said.
“The statement is true – the brain ultimately processes signals from throughout the body, forms the perception of pain, and links it to emotional brain centers, among others. Pain is an important survival mechanism so that when your body is at threat of injury, you protect yourself from further damage and withdraw. The problem lies when pain outlasts its welcome and chronifies,” said Dr. Lakhan, senior vice president of research and development of Click Therapeutics in Boston.
The investigators in this study “eloquently prove” that with 4 weeks of PRT, patients can learn that chronic pain is largely a “brain-generated false alarm and that constantly affirming this truth can actually reduce or eliminate it,” Dr. Lakhan said.
“Further, the brain areas implicated with pain are calmed after going through the therapy to both resting pain and pain induced by extending the back,” he noted.
“Pain-reprocessing therapy can improve the lives of chronic [pain patients] who have low to moderate levels of pain and disability; however, much work needs to be done to make this scalable and universally available and covered by insurers as a treatment modality,” Dr. Lakhan added.
He cautioned that he has not seen therapies such as this work when there is significant depression, withdrawal, or lack of control over one’s situation such that one behaves in a helpless manner – “a terrible state of mind called learned helplessness.”
The study was funded by the National Institutes of Health, the National Center for Advancing Translational Sciences, the Radiological Society of North America, the German Research Foundation, the Psychophysiologic Disorders Association, the Foundation for the Study of the Therapeutic Encounter, and community donations. Dr. Ashar received grants from the National Institutes of Health during the conduct of the study and personal fees from UnitedHealth Group, Lin Health, Pain Reprocessing Therapy Center, and Mental Health Partners of Boulder County outside the submitted work. Dr. Lakhan disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Psychological therapy that changes an individual’s beliefs about pain not only provides lasting chronic pain relief but also alters brain regions related to pain generation, new research shows.
In the first randomized controlled test of pain-reprocessing therapy (PRT), two-thirds of patients with chronic back pain (CBP) who received 4 weeks of PRT were pain free or nearly pain free afterward – and for most patients, relief was maintained for 1 year, the researchers found.
“Primary chronic back pain can be dramatically reduced or even eliminated by psychological treatment focused on changing how threatening we perceive the pain to be,” first author Yoni Ashar, PhD, department of psychiatry, Weill Cornell Medicine, New York, said in an interview.
“ given that large reductions in pain have rarely been observed in studies that tested psychological therapies for chronic back pain.
The study was published online Sept. 29, 2021, in JAMA Psychiatry.
Rethinking pain
CBP is a leading cause of disability, and treatment is often ineffective. In about 85% of cases of primary CBP, a definitive cause of the pain can’t be identified. In these cases, fear, avoidance, and beliefs that pain indicates injury may contribute to ongoing CBP.
PRT educates patients about the role of the brain in generating chronic pain; helps them reappraise their pain as they engage in movements that they had been afraid to undertake; and helps them address emotions that may exacerbate pain.
The study included 151 adults (54% women; mean age, 41 years) who had primary CBP of low to moderate severity (mean pain intensity, 4 of 10) for an average of 10 years.
A total of 50 participants were randomly allocated to undergo PRT (one telehealth session with a physician and eight PRT sessions over 4 weeks), 51 to receive placebo (subcutaneous saline injection in the back), and 50 to continue their routine, usual ongoing care.
Large group differences in pain were observed after treatment. The mean pain score was 1.18 in the PRT group, 2.84 in the placebo group, and 3.13 in the usual-care group. Hedges’ g was –1.14 for PRT versus placebo and –1.74 for PRT versus usual care (P < .001).
Two-thirds (66%) of adults in the PRT group were pain free or nearly pain free following treatment (pain-intensity score of 0 or 1 out of 10), compared with 20% of those in the placebo group and 10% of those who received usual care.
Treatment effects were maintained at 1-year follow-up. The mean pain score was 1.51 in the PRT group, 2.79 in the placebo group, and 3.00 in the usual-care group. Neither age nor sex moderated the effect of PRT on pain intensity.
Retraining the brain
The researchers said the effects of PRT on pain were mediated by lessening the belief that pain indicates tissue damage. Of note, PRT also reduced experimentally evoked back pain and spontaneous pain during functional MRI, with large effect sizes.
“The idea is that by thinking about the pain as safe rather than threatening, patients can alter the brain networks reinforcing the pain, and neutralize it,” Dr. Ashar said in a news release.
The authors noted that study participants were relatively well educated and active. The participants reported having longstanding low to moderate pain and disability at baseline.
The physician and therapists were experts in delivering PRT. Future studies should test generalizability to other patient populations, therapists, and treatment contexts.
“Our clinical experience shows that PRT is effective for other primary chronic pain conditions as well,” said Dr. Ashar, including primary knee pain and tension headache.
Restoring function
Commenting on the findings, Shaheen E. Lakhan, MD, PhD, neurologist and pain specialist in Newton, Mass., said he has long experience using psychological approaches to address pain, with good results.
“Imagine telling a person suffering from decades of chronic pain that your pain is all in your head. I’ve done that for years as a board-certified pain physician managing only the most severe and debilitating forms of pain. When used to ground brain retraining, I could ultimately restore function to people living with chronic pain,” Dr. Lakhan said.
“The statement is true – the brain ultimately processes signals from throughout the body, forms the perception of pain, and links it to emotional brain centers, among others. Pain is an important survival mechanism so that when your body is at threat of injury, you protect yourself from further damage and withdraw. The problem lies when pain outlasts its welcome and chronifies,” said Dr. Lakhan, senior vice president of research and development of Click Therapeutics in Boston.
The investigators in this study “eloquently prove” that with 4 weeks of PRT, patients can learn that chronic pain is largely a “brain-generated false alarm and that constantly affirming this truth can actually reduce or eliminate it,” Dr. Lakhan said.
“Further, the brain areas implicated with pain are calmed after going through the therapy to both resting pain and pain induced by extending the back,” he noted.
“Pain-reprocessing therapy can improve the lives of chronic [pain patients] who have low to moderate levels of pain and disability; however, much work needs to be done to make this scalable and universally available and covered by insurers as a treatment modality,” Dr. Lakhan added.
He cautioned that he has not seen therapies such as this work when there is significant depression, withdrawal, or lack of control over one’s situation such that one behaves in a helpless manner – “a terrible state of mind called learned helplessness.”
The study was funded by the National Institutes of Health, the National Center for Advancing Translational Sciences, the Radiological Society of North America, the German Research Foundation, the Psychophysiologic Disorders Association, the Foundation for the Study of the Therapeutic Encounter, and community donations. Dr. Ashar received grants from the National Institutes of Health during the conduct of the study and personal fees from UnitedHealth Group, Lin Health, Pain Reprocessing Therapy Center, and Mental Health Partners of Boulder County outside the submitted work. Dr. Lakhan disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Psychological therapy that changes an individual’s beliefs about pain not only provides lasting chronic pain relief but also alters brain regions related to pain generation, new research shows.
In the first randomized controlled test of pain-reprocessing therapy (PRT), two-thirds of patients with chronic back pain (CBP) who received 4 weeks of PRT were pain free or nearly pain free afterward – and for most patients, relief was maintained for 1 year, the researchers found.
“Primary chronic back pain can be dramatically reduced or even eliminated by psychological treatment focused on changing how threatening we perceive the pain to be,” first author Yoni Ashar, PhD, department of psychiatry, Weill Cornell Medicine, New York, said in an interview.
“ given that large reductions in pain have rarely been observed in studies that tested psychological therapies for chronic back pain.
The study was published online Sept. 29, 2021, in JAMA Psychiatry.
Rethinking pain
CBP is a leading cause of disability, and treatment is often ineffective. In about 85% of cases of primary CBP, a definitive cause of the pain can’t be identified. In these cases, fear, avoidance, and beliefs that pain indicates injury may contribute to ongoing CBP.
PRT educates patients about the role of the brain in generating chronic pain; helps them reappraise their pain as they engage in movements that they had been afraid to undertake; and helps them address emotions that may exacerbate pain.
The study included 151 adults (54% women; mean age, 41 years) who had primary CBP of low to moderate severity (mean pain intensity, 4 of 10) for an average of 10 years.
A total of 50 participants were randomly allocated to undergo PRT (one telehealth session with a physician and eight PRT sessions over 4 weeks), 51 to receive placebo (subcutaneous saline injection in the back), and 50 to continue their routine, usual ongoing care.
Large group differences in pain were observed after treatment. The mean pain score was 1.18 in the PRT group, 2.84 in the placebo group, and 3.13 in the usual-care group. Hedges’ g was –1.14 for PRT versus placebo and –1.74 for PRT versus usual care (P < .001).
Two-thirds (66%) of adults in the PRT group were pain free or nearly pain free following treatment (pain-intensity score of 0 or 1 out of 10), compared with 20% of those in the placebo group and 10% of those who received usual care.
Treatment effects were maintained at 1-year follow-up. The mean pain score was 1.51 in the PRT group, 2.79 in the placebo group, and 3.00 in the usual-care group. Neither age nor sex moderated the effect of PRT on pain intensity.
Retraining the brain
The researchers said the effects of PRT on pain were mediated by lessening the belief that pain indicates tissue damage. Of note, PRT also reduced experimentally evoked back pain and spontaneous pain during functional MRI, with large effect sizes.
“The idea is that by thinking about the pain as safe rather than threatening, patients can alter the brain networks reinforcing the pain, and neutralize it,” Dr. Ashar said in a news release.
The authors noted that study participants were relatively well educated and active. The participants reported having longstanding low to moderate pain and disability at baseline.
The physician and therapists were experts in delivering PRT. Future studies should test generalizability to other patient populations, therapists, and treatment contexts.
“Our clinical experience shows that PRT is effective for other primary chronic pain conditions as well,” said Dr. Ashar, including primary knee pain and tension headache.
Restoring function
Commenting on the findings, Shaheen E. Lakhan, MD, PhD, neurologist and pain specialist in Newton, Mass., said he has long experience using psychological approaches to address pain, with good results.
“Imagine telling a person suffering from decades of chronic pain that your pain is all in your head. I’ve done that for years as a board-certified pain physician managing only the most severe and debilitating forms of pain. When used to ground brain retraining, I could ultimately restore function to people living with chronic pain,” Dr. Lakhan said.
“The statement is true – the brain ultimately processes signals from throughout the body, forms the perception of pain, and links it to emotional brain centers, among others. Pain is an important survival mechanism so that when your body is at threat of injury, you protect yourself from further damage and withdraw. The problem lies when pain outlasts its welcome and chronifies,” said Dr. Lakhan, senior vice president of research and development of Click Therapeutics in Boston.
The investigators in this study “eloquently prove” that with 4 weeks of PRT, patients can learn that chronic pain is largely a “brain-generated false alarm and that constantly affirming this truth can actually reduce or eliminate it,” Dr. Lakhan said.
“Further, the brain areas implicated with pain are calmed after going through the therapy to both resting pain and pain induced by extending the back,” he noted.
“Pain-reprocessing therapy can improve the lives of chronic [pain patients] who have low to moderate levels of pain and disability; however, much work needs to be done to make this scalable and universally available and covered by insurers as a treatment modality,” Dr. Lakhan added.
He cautioned that he has not seen therapies such as this work when there is significant depression, withdrawal, or lack of control over one’s situation such that one behaves in a helpless manner – “a terrible state of mind called learned helplessness.”
The study was funded by the National Institutes of Health, the National Center for Advancing Translational Sciences, the Radiological Society of North America, the German Research Foundation, the Psychophysiologic Disorders Association, the Foundation for the Study of the Therapeutic Encounter, and community donations. Dr. Ashar received grants from the National Institutes of Health during the conduct of the study and personal fees from UnitedHealth Group, Lin Health, Pain Reprocessing Therapy Center, and Mental Health Partners of Boulder County outside the submitted work. Dr. Lakhan disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Major insurers running billions of dollars behind on payments to hospitals and doctors
Anthem Blue Cross, the country’s second-biggest health insurance company, is behind on billions of dollars in payments owed to hospitals and doctors because of onerous new reimbursement rules, computer problems and mishandled claims, say hospital officials in multiple states.
Anthem, like other big insurers, is using the COVID-19 crisis as cover to institute “egregious” policies that harm patients and pinch hospital finances, said Molly Smith, group vice president at the American Hospital Association. “There’s this sense of ‘Everyone’s distracted. We can get this through.’ ”
Hospitals are also dealing with a spike in retroactive claims denials by UnitedHealthcare, the biggest health insurer, for ED care, the AHA said.
Hospitals say it is hurting their finances as many cope with COVID surges – even after the industry has received tens of billions of dollars in emergency assistance from the federal government.
“We recognize there have been some challenges” to prompt payments caused by claims-processing changes and “a new set of dynamics” amid the pandemic, Anthem spokesperson Colin Manning said in an email. “We apologize for any delays or inconvenience this may have caused.”
Virginia law requires insurers to pay claims within 40 days. In a Sept. 24 letter to state insurance regulators, VCU Health, a system that operates a large teaching hospital in Richmond associated with Virginia Commonwealth University, said Anthem owes it $385 million. More than 40% of the claims are more than 90 days old, VCU said.
For all Virginia hospitals, Anthem’s late, unpaid claims amount to “hundreds of millions of dollars,” the Virginia Hospital and Healthcare Association said in a June 23 letter to state regulators.
Nationwide, the payment delays “are creating an untenable situation,” the American Hospital Association said in a Sept. 9 letter to Anthem CEO Gail Boudreaux. “Patients are facing greater hurdles to accessing care; clinicians are burning out on unnecessary administrative tasks; and the system is straining to finance the personnel and supplies” needed to fight Covid.
Complaints about Anthem extend “from sea to shining sea, from New Hampshire to California,” AHA CEO Rick Pollack told KHN.
Substantial payment delays can be seen on Anthem’s books. On June 30, 2019, before the pandemic, 43% of the insurer’s medical bills for that quarter were unpaid, according to regulatory filings. Two years later that figure had risen to 53% – a difference of $2.5 billion.
Anthem profits were $4.6 billion in 2020 and $3.5 billion in the first half of 2021.
Alexis Thurber, who lives near Seattle, was insured by Anthem when she got an $18,192 hospital bill in May for radiation therapy that doctors said was essential to treat her breast cancer.
The treatments were “experimental” and “not medically necessary,” Anthem said, according to Ms. Thurber. She spent much of the summer trying to get the insurer to pay up – placing two dozen phone calls, spending hours on hold, sending multiple emails and enduring unmeasurable stress and worry. It finally covered the claim months later.
“It’s so egregious. It’s a game they’re playing,” said Ms. Thurber, 51, whose cancer was diagnosed in November. “Trying to get true help was impossible.”
Privacy rules prevent Anthem from commenting on Ms. Thurber’s case, said Anthem spokesperson Colin Manning.
When insurers fail to promptly pay medical bills, patients are left in the lurch. They might first get a notice saying payment is pending or denied. A hospital might bill them for treatment they thought would be covered. Hospitals and doctors often sue patients whose insurance didn’t pay up.
Hospitals point to a variety of Anthem practices contributing to payment delays or denials, including new layers of document requirements, prior-authorization hurdles for routine procedures and requirements that doctors themselves – not support staffers – speak to insurance gatekeepers. “This requires providers to literally leave the patient[’s] bedside to get on the phone with Anthem,” AHA said in its letter.
Anthem often hinders coverage for outpatient surgery, specialty pharmacy and other services in health systems listed as in network, amounting to a “bait and switch” on Anthem members, AHA officials said.
“Demanding that patients be treated outside of the hospital setting, against the advice of the patient’s in-network treating physician, appears to be motivated by a desire to drive up Empire’s profits,” the Greater New York Hospital Association wrote in an April letter to Empire Blue Cross, which is owned by Anthem.
Anthem officials pushed back in a recent letter to the AHA, saying the insurer’s changing rules are intended partly to control excessive prices charged by hospitals for specialty drugs and nonemergency surgery, screening and diagnostic procedures.
Severe problems with Anthem’s new claims management system surfaced months ago and “persist without meaningful improvement,” AHA said in its letter.
Claims have gotten lost in Anthem’s computers, and in some cases VCU Health has had to print medical records and mail them to get paid, VCU said in its letter. The cash slowdown imposes “an unmanageable disruption that threatens to undermine our financial footing,” VCU said.
United denied $31,557 in claims for Emily Long’s care after she was struck in June by a motorcycle in New York City. She needed surgery to repair a fractured cheekbone. United said there was a lack of documentation for “medical necessity” – an “incredibly aggravating” response on top of the distress of the accident, Ms. Long said.
The Brooklyn hospital that treated Ms. Long was “paid appropriately under her plan and within the required time frame,” said United spokesperson Maria Gordon Shydlo. “The facility has the right to appeal the decision.”
United’s unpaid claims came to 54% as of June 30, about the same level as 2 years previously.
When Erin Conlisk initially had trouble gaining approval for a piece of medical equipment for her elderly father this summer, United employees told her the insurer’s entire prior-authorization database had gone down for weeks, said Ms. Conlisk, who lives in California.
“There was a brief issue with our prior-authorization process in mid-July, which was resolved quickly,” Gordon Shydlo said.
When asked by Wall Street analysts about the payment backups, Anthem executives said it partly reflects their decision to increase financial reserves amid the health crisis.
“Really a ton of uncertainty associated with this environment,” John Gallina, the company’s chief financial officer, said on a conference call in July. “We’ve tried to be extremely prudent and conservative in our approach.”
During the pandemic, hospitals have benefited from two extraordinary cash infusions. They and other medical providers have received more than $100 billion through the CARES Act of 2020 and the American Rescue Plan of 2021. Last year United, Anthem and other insurers accelerated billions in hospital reimbursements.
The federal payments enriched many of the biggest, wealthiest systems while poorer hospitals serving low-income patients and rural areas struggled.
Those are the systems most hurt now by insurer payment delays, hospital officials said. Federal relief funds “have been a lifeline, but they don’t make people whole in terms of the losses from increased expenses and lost revenue as a result of the COVID experience,” Mr. Pollack said.
Several health systems declined to comment about claims payment delays or didn’t respond to a reporter’s queries. Among individual hospitals “there is a deep fear of talking on the record about your largest business partner,” AHA’s Ms. Smith said.
Alexis Thurber worried she might have to pay her $18,192 radiation bill herself, and she’s not confident her Anthem policy will do a better job next time of covering the cost of her care.
“It makes me not want to go to the doctor anymore,” she said. “I’m scared to get another mammogram because you can’t rely on it.”
KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation.
Anthem Blue Cross, the country’s second-biggest health insurance company, is behind on billions of dollars in payments owed to hospitals and doctors because of onerous new reimbursement rules, computer problems and mishandled claims, say hospital officials in multiple states.
Anthem, like other big insurers, is using the COVID-19 crisis as cover to institute “egregious” policies that harm patients and pinch hospital finances, said Molly Smith, group vice president at the American Hospital Association. “There’s this sense of ‘Everyone’s distracted. We can get this through.’ ”
Hospitals are also dealing with a spike in retroactive claims denials by UnitedHealthcare, the biggest health insurer, for ED care, the AHA said.
Hospitals say it is hurting their finances as many cope with COVID surges – even after the industry has received tens of billions of dollars in emergency assistance from the federal government.
“We recognize there have been some challenges” to prompt payments caused by claims-processing changes and “a new set of dynamics” amid the pandemic, Anthem spokesperson Colin Manning said in an email. “We apologize for any delays or inconvenience this may have caused.”
Virginia law requires insurers to pay claims within 40 days. In a Sept. 24 letter to state insurance regulators, VCU Health, a system that operates a large teaching hospital in Richmond associated with Virginia Commonwealth University, said Anthem owes it $385 million. More than 40% of the claims are more than 90 days old, VCU said.
For all Virginia hospitals, Anthem’s late, unpaid claims amount to “hundreds of millions of dollars,” the Virginia Hospital and Healthcare Association said in a June 23 letter to state regulators.
Nationwide, the payment delays “are creating an untenable situation,” the American Hospital Association said in a Sept. 9 letter to Anthem CEO Gail Boudreaux. “Patients are facing greater hurdles to accessing care; clinicians are burning out on unnecessary administrative tasks; and the system is straining to finance the personnel and supplies” needed to fight Covid.
Complaints about Anthem extend “from sea to shining sea, from New Hampshire to California,” AHA CEO Rick Pollack told KHN.
Substantial payment delays can be seen on Anthem’s books. On June 30, 2019, before the pandemic, 43% of the insurer’s medical bills for that quarter were unpaid, according to regulatory filings. Two years later that figure had risen to 53% – a difference of $2.5 billion.
Anthem profits were $4.6 billion in 2020 and $3.5 billion in the first half of 2021.
Alexis Thurber, who lives near Seattle, was insured by Anthem when she got an $18,192 hospital bill in May for radiation therapy that doctors said was essential to treat her breast cancer.
The treatments were “experimental” and “not medically necessary,” Anthem said, according to Ms. Thurber. She spent much of the summer trying to get the insurer to pay up – placing two dozen phone calls, spending hours on hold, sending multiple emails and enduring unmeasurable stress and worry. It finally covered the claim months later.
“It’s so egregious. It’s a game they’re playing,” said Ms. Thurber, 51, whose cancer was diagnosed in November. “Trying to get true help was impossible.”
Privacy rules prevent Anthem from commenting on Ms. Thurber’s case, said Anthem spokesperson Colin Manning.
When insurers fail to promptly pay medical bills, patients are left in the lurch. They might first get a notice saying payment is pending or denied. A hospital might bill them for treatment they thought would be covered. Hospitals and doctors often sue patients whose insurance didn’t pay up.
Hospitals point to a variety of Anthem practices contributing to payment delays or denials, including new layers of document requirements, prior-authorization hurdles for routine procedures and requirements that doctors themselves – not support staffers – speak to insurance gatekeepers. “This requires providers to literally leave the patient[’s] bedside to get on the phone with Anthem,” AHA said in its letter.
Anthem often hinders coverage for outpatient surgery, specialty pharmacy and other services in health systems listed as in network, amounting to a “bait and switch” on Anthem members, AHA officials said.
“Demanding that patients be treated outside of the hospital setting, against the advice of the patient’s in-network treating physician, appears to be motivated by a desire to drive up Empire’s profits,” the Greater New York Hospital Association wrote in an April letter to Empire Blue Cross, which is owned by Anthem.
Anthem officials pushed back in a recent letter to the AHA, saying the insurer’s changing rules are intended partly to control excessive prices charged by hospitals for specialty drugs and nonemergency surgery, screening and diagnostic procedures.
Severe problems with Anthem’s new claims management system surfaced months ago and “persist without meaningful improvement,” AHA said in its letter.
Claims have gotten lost in Anthem’s computers, and in some cases VCU Health has had to print medical records and mail them to get paid, VCU said in its letter. The cash slowdown imposes “an unmanageable disruption that threatens to undermine our financial footing,” VCU said.
United denied $31,557 in claims for Emily Long’s care after she was struck in June by a motorcycle in New York City. She needed surgery to repair a fractured cheekbone. United said there was a lack of documentation for “medical necessity” – an “incredibly aggravating” response on top of the distress of the accident, Ms. Long said.
The Brooklyn hospital that treated Ms. Long was “paid appropriately under her plan and within the required time frame,” said United spokesperson Maria Gordon Shydlo. “The facility has the right to appeal the decision.”
United’s unpaid claims came to 54% as of June 30, about the same level as 2 years previously.
When Erin Conlisk initially had trouble gaining approval for a piece of medical equipment for her elderly father this summer, United employees told her the insurer’s entire prior-authorization database had gone down for weeks, said Ms. Conlisk, who lives in California.
“There was a brief issue with our prior-authorization process in mid-July, which was resolved quickly,” Gordon Shydlo said.
When asked by Wall Street analysts about the payment backups, Anthem executives said it partly reflects their decision to increase financial reserves amid the health crisis.
“Really a ton of uncertainty associated with this environment,” John Gallina, the company’s chief financial officer, said on a conference call in July. “We’ve tried to be extremely prudent and conservative in our approach.”
During the pandemic, hospitals have benefited from two extraordinary cash infusions. They and other medical providers have received more than $100 billion through the CARES Act of 2020 and the American Rescue Plan of 2021. Last year United, Anthem and other insurers accelerated billions in hospital reimbursements.
The federal payments enriched many of the biggest, wealthiest systems while poorer hospitals serving low-income patients and rural areas struggled.
Those are the systems most hurt now by insurer payment delays, hospital officials said. Federal relief funds “have been a lifeline, but they don’t make people whole in terms of the losses from increased expenses and lost revenue as a result of the COVID experience,” Mr. Pollack said.
Several health systems declined to comment about claims payment delays or didn’t respond to a reporter’s queries. Among individual hospitals “there is a deep fear of talking on the record about your largest business partner,” AHA’s Ms. Smith said.
Alexis Thurber worried she might have to pay her $18,192 radiation bill herself, and she’s not confident her Anthem policy will do a better job next time of covering the cost of her care.
“It makes me not want to go to the doctor anymore,” she said. “I’m scared to get another mammogram because you can’t rely on it.”
KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation.
Anthem Blue Cross, the country’s second-biggest health insurance company, is behind on billions of dollars in payments owed to hospitals and doctors because of onerous new reimbursement rules, computer problems and mishandled claims, say hospital officials in multiple states.
Anthem, like other big insurers, is using the COVID-19 crisis as cover to institute “egregious” policies that harm patients and pinch hospital finances, said Molly Smith, group vice president at the American Hospital Association. “There’s this sense of ‘Everyone’s distracted. We can get this through.’ ”
Hospitals are also dealing with a spike in retroactive claims denials by UnitedHealthcare, the biggest health insurer, for ED care, the AHA said.
Hospitals say it is hurting their finances as many cope with COVID surges – even after the industry has received tens of billions of dollars in emergency assistance from the federal government.
“We recognize there have been some challenges” to prompt payments caused by claims-processing changes and “a new set of dynamics” amid the pandemic, Anthem spokesperson Colin Manning said in an email. “We apologize for any delays or inconvenience this may have caused.”
Virginia law requires insurers to pay claims within 40 days. In a Sept. 24 letter to state insurance regulators, VCU Health, a system that operates a large teaching hospital in Richmond associated with Virginia Commonwealth University, said Anthem owes it $385 million. More than 40% of the claims are more than 90 days old, VCU said.
For all Virginia hospitals, Anthem’s late, unpaid claims amount to “hundreds of millions of dollars,” the Virginia Hospital and Healthcare Association said in a June 23 letter to state regulators.
Nationwide, the payment delays “are creating an untenable situation,” the American Hospital Association said in a Sept. 9 letter to Anthem CEO Gail Boudreaux. “Patients are facing greater hurdles to accessing care; clinicians are burning out on unnecessary administrative tasks; and the system is straining to finance the personnel and supplies” needed to fight Covid.
Complaints about Anthem extend “from sea to shining sea, from New Hampshire to California,” AHA CEO Rick Pollack told KHN.
Substantial payment delays can be seen on Anthem’s books. On June 30, 2019, before the pandemic, 43% of the insurer’s medical bills for that quarter were unpaid, according to regulatory filings. Two years later that figure had risen to 53% – a difference of $2.5 billion.
Anthem profits were $4.6 billion in 2020 and $3.5 billion in the first half of 2021.
Alexis Thurber, who lives near Seattle, was insured by Anthem when she got an $18,192 hospital bill in May for radiation therapy that doctors said was essential to treat her breast cancer.
The treatments were “experimental” and “not medically necessary,” Anthem said, according to Ms. Thurber. She spent much of the summer trying to get the insurer to pay up – placing two dozen phone calls, spending hours on hold, sending multiple emails and enduring unmeasurable stress and worry. It finally covered the claim months later.
“It’s so egregious. It’s a game they’re playing,” said Ms. Thurber, 51, whose cancer was diagnosed in November. “Trying to get true help was impossible.”
Privacy rules prevent Anthem from commenting on Ms. Thurber’s case, said Anthem spokesperson Colin Manning.
When insurers fail to promptly pay medical bills, patients are left in the lurch. They might first get a notice saying payment is pending or denied. A hospital might bill them for treatment they thought would be covered. Hospitals and doctors often sue patients whose insurance didn’t pay up.
Hospitals point to a variety of Anthem practices contributing to payment delays or denials, including new layers of document requirements, prior-authorization hurdles for routine procedures and requirements that doctors themselves – not support staffers – speak to insurance gatekeepers. “This requires providers to literally leave the patient[’s] bedside to get on the phone with Anthem,” AHA said in its letter.
Anthem often hinders coverage for outpatient surgery, specialty pharmacy and other services in health systems listed as in network, amounting to a “bait and switch” on Anthem members, AHA officials said.
“Demanding that patients be treated outside of the hospital setting, against the advice of the patient’s in-network treating physician, appears to be motivated by a desire to drive up Empire’s profits,” the Greater New York Hospital Association wrote in an April letter to Empire Blue Cross, which is owned by Anthem.
Anthem officials pushed back in a recent letter to the AHA, saying the insurer’s changing rules are intended partly to control excessive prices charged by hospitals for specialty drugs and nonemergency surgery, screening and diagnostic procedures.
Severe problems with Anthem’s new claims management system surfaced months ago and “persist without meaningful improvement,” AHA said in its letter.
Claims have gotten lost in Anthem’s computers, and in some cases VCU Health has had to print medical records and mail them to get paid, VCU said in its letter. The cash slowdown imposes “an unmanageable disruption that threatens to undermine our financial footing,” VCU said.
United denied $31,557 in claims for Emily Long’s care after she was struck in June by a motorcycle in New York City. She needed surgery to repair a fractured cheekbone. United said there was a lack of documentation for “medical necessity” – an “incredibly aggravating” response on top of the distress of the accident, Ms. Long said.
The Brooklyn hospital that treated Ms. Long was “paid appropriately under her plan and within the required time frame,” said United spokesperson Maria Gordon Shydlo. “The facility has the right to appeal the decision.”
United’s unpaid claims came to 54% as of June 30, about the same level as 2 years previously.
When Erin Conlisk initially had trouble gaining approval for a piece of medical equipment for her elderly father this summer, United employees told her the insurer’s entire prior-authorization database had gone down for weeks, said Ms. Conlisk, who lives in California.
“There was a brief issue with our prior-authorization process in mid-July, which was resolved quickly,” Gordon Shydlo said.
When asked by Wall Street analysts about the payment backups, Anthem executives said it partly reflects their decision to increase financial reserves amid the health crisis.
“Really a ton of uncertainty associated with this environment,” John Gallina, the company’s chief financial officer, said on a conference call in July. “We’ve tried to be extremely prudent and conservative in our approach.”
During the pandemic, hospitals have benefited from two extraordinary cash infusions. They and other medical providers have received more than $100 billion through the CARES Act of 2020 and the American Rescue Plan of 2021. Last year United, Anthem and other insurers accelerated billions in hospital reimbursements.
The federal payments enriched many of the biggest, wealthiest systems while poorer hospitals serving low-income patients and rural areas struggled.
Those are the systems most hurt now by insurer payment delays, hospital officials said. Federal relief funds “have been a lifeline, but they don’t make people whole in terms of the losses from increased expenses and lost revenue as a result of the COVID experience,” Mr. Pollack said.
Several health systems declined to comment about claims payment delays or didn’t respond to a reporter’s queries. Among individual hospitals “there is a deep fear of talking on the record about your largest business partner,” AHA’s Ms. Smith said.
Alexis Thurber worried she might have to pay her $18,192 radiation bill herself, and she’s not confident her Anthem policy will do a better job next time of covering the cost of her care.
“It makes me not want to go to the doctor anymore,” she said. “I’m scared to get another mammogram because you can’t rely on it.”
KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation.
Cement found in man’s heart after spinal surgery
, according to a new report published in the New England Journal of Medicine.
The 56-year-old man, who was not identified in the report, went to the emergency room after experiencing 2 days of chest pain and shortness of breath. Imaging scans showed that the chest pain was caused by a foreign object, and he was rushed to surgery.
Surgeons then located and removed a thin, sharp, cylindrical piece of cement and repaired the damage to the patient’s heart. The cement had pierced the upper right chamber of his heart and his right lung, according to the report authors from the Yale University School of Medicine.
A week before, the man had undergone a spinal surgery known as kyphoplasty. The procedure treats spine injuries by injecting a special type of medical cement into damaged vertebrae, according to USA Today. The cement had leaked into the patient’s body, hardened, and traveled to his heart.
The man has now “nearly recovered” since the heart surgery and cement removal, which occurred about a month ago, the journal report stated. He experienced no additional complications.
Cement leakage after kyphoplasty can happen but is an extremely rare complication. Less than 2% of patients who undergo the procedure for osteoporosis or brittle bones have complications, according to patient information from the American Association of Neurological Surgeons.
A version of this article first appeared on WebMD.com.
, according to a new report published in the New England Journal of Medicine.
The 56-year-old man, who was not identified in the report, went to the emergency room after experiencing 2 days of chest pain and shortness of breath. Imaging scans showed that the chest pain was caused by a foreign object, and he was rushed to surgery.
Surgeons then located and removed a thin, sharp, cylindrical piece of cement and repaired the damage to the patient’s heart. The cement had pierced the upper right chamber of his heart and his right lung, according to the report authors from the Yale University School of Medicine.
A week before, the man had undergone a spinal surgery known as kyphoplasty. The procedure treats spine injuries by injecting a special type of medical cement into damaged vertebrae, according to USA Today. The cement had leaked into the patient’s body, hardened, and traveled to his heart.
The man has now “nearly recovered” since the heart surgery and cement removal, which occurred about a month ago, the journal report stated. He experienced no additional complications.
Cement leakage after kyphoplasty can happen but is an extremely rare complication. Less than 2% of patients who undergo the procedure for osteoporosis or brittle bones have complications, according to patient information from the American Association of Neurological Surgeons.
A version of this article first appeared on WebMD.com.
, according to a new report published in the New England Journal of Medicine.
The 56-year-old man, who was not identified in the report, went to the emergency room after experiencing 2 days of chest pain and shortness of breath. Imaging scans showed that the chest pain was caused by a foreign object, and he was rushed to surgery.
Surgeons then located and removed a thin, sharp, cylindrical piece of cement and repaired the damage to the patient’s heart. The cement had pierced the upper right chamber of his heart and his right lung, according to the report authors from the Yale University School of Medicine.
A week before, the man had undergone a spinal surgery known as kyphoplasty. The procedure treats spine injuries by injecting a special type of medical cement into damaged vertebrae, according to USA Today. The cement had leaked into the patient’s body, hardened, and traveled to his heart.
The man has now “nearly recovered” since the heart surgery and cement removal, which occurred about a month ago, the journal report stated. He experienced no additional complications.
Cement leakage after kyphoplasty can happen but is an extremely rare complication. Less than 2% of patients who undergo the procedure for osteoporosis or brittle bones have complications, according to patient information from the American Association of Neurological Surgeons.
A version of this article first appeared on WebMD.com.
Quality of Life, Diagnosis, and/or Therapeutics in Pediatric MS
Vikram Bhise, MD, Is an Associate Professor at Rutgers – Robert Wood Johnson Medical School. He specializes in Epilepsy and Pediatric Neuroimmunology, and runs the pediatric demyelinating diseases program, evaluating children with multiple sclerosis, autoimmune encephalopathy, and related diseases. He trained in Pediatrics and Pediatric Neurology, at Maimonides Medical Center and Montefiore Medical Center, respectively. He subsequently received additional training in Clinical Neurophysiology with a focus on Epilepsy at SUNY Downstate Medical Center, and in Pediatric Multiple Sclerosis at SUNY Stony Brook Medical Center. Dr. Bhise conducts clinical research focused on biomarkers and quality of life in pediatric multiple sclerosis, as well as studies in epilepsy and neurogenetics.
Q1. As a specialist who focuses on neuroimmunology, what forms of measurement do you use to make an evaluation or diagnosis for children with multiple sclerosis?
A1. There's a lot that goes into evaluating or diagnosing children with MS. Usually we start off with the story that the family brings to us. We look at what the child is experiencing and what the parents are seeing. Then we do a dedicated examination trying to substantiate the findings that they're describing and look for others they may not even be aware of. If they are having some blurred vision in their eye, can we tell if there's some abnormalities there that are correlating with what they see?
We try to get a good sense of the time-course of things, observing whether this is the first time something's happened or if this has this been going on for a while. Have there been multiple things going on, multiple episodes? We're primarily looking for events called relapses, which are neurologic attacks that are not quick. They don’t last for seconds or hours; they can last for days to weeks, sometimes even months. Individuals will have episodes that tend to get worse and then tend to get better. This is the type of description we’re looking to come from the families.
Once that assessment is complete, we've found that the MRI is one of the best tools in helping us confirm the diagnosis. It's not just diagnostic but the MRI also has some prognostic potential and we're looking specifically for patterns in the MRIs. For children, that pattern can be a little bit more challenging. Their patterns can often overlap with patterns of other inflammatory diseases of the brain like ADEM for example and make it much more difficult for us to characterize someone as truly having MS.
There are also some other diseases which have been discovered in the past decade or elaborated upon like neuromyelitis optica and MOG antibody disorder, which can look exactly like MS in the early stages. Sometimes, that's just not all the information we need. Sometimes it's more difficult to make those distinctions and in these cases, we will look at a spinal tap, a lumbar puncture, and for specific studies from those procedures to help us get a better understanding. There may be other ancillary tests that we use, such as evoked potentials, for example.
Evoked potential testing has kind of fallen to the wayside over the past decade because of the MRI studies becoming a much more useful tool, but we may still use the visual evoked potential to see if there are subtle lesions that can't be seen on the MRI. Other methods might include optical coherence tomography, which is another test looking at the eye that gives you a specific look at the retinal nerve fiber layer, which gets thinned after attacks on the eye on the optic nerve.
We may do neuropsychological testing, which is a battery of tests looking at different cognitive domains and trying to get a sense of a person's cognitive profile to see if that matches what we would expect in somebody with MS. This test could be more challenging for a teen and a child, particularly a younger child. When it comes to pediatric neuropsychology, it's a little bit harder sometimes to get good data, particularly from younger kids.
In addition, we have a battery of tests that we do on the serum. Some disorders, like NMO and MOG, have antibodies that help us identify them. We don't have a specific test that says, yes, you have MS and no, you don't have MS. It's really the combination of all the tools.
We do these tests often to look for things that mimic MS. We look for other neuroinflammatory or neurobiological diseases that can look a lot like MS and fool us. Most of the time they don't look exactly like MS, but every now and then you get a case that's virtually indistinguishable. There are other tools which may be in less use, but we put the combination of all these things together to help us make the most informed judgment.
The goal is to be able to have honest discussions with families that these tools are just tools that we're trying to play catch up with a disease and try to make a decision as fast as possible to prevent someone from going untreated.
Q2. How does a diagnosis of multiple sclerosis affect the overall quality of life for a child/teen, and how does it affect their overall psychosocial health? Education? Transition needs? Etc.
A2. It can be quite profound, just hearing the diagnosis can be truly life changing for most folks. It would really depend on the family the first time that we meet them. If they have no suspicion that this is what's going on, that can be a shock. Other families may be more aware of what’s going on. Perhaps another physician has already suggested it, or they came from the ER which had already done some of the baseline tests like the MRI, and they had some kind of suspicion. Maybe they googled it and they saw something to be worried about, so they may be prepared. But even then, once you confirm the diagnosis, it's really like the sky falling at that point.
Past the diagnosis stage, there's really an adjustment phase that we see, and we've been doing some work in this. We started doing some work looking at quality of life. We've interviewed a large number of families and asked them some key questions such as, “What's important to you?” It is key for them to tell us rather than us telling them. By doing this, we’re finding out things that may not have been on the forefront of our minds, although it was certainly in the forefront of their minds, so it's a good learning opportunity.
These may be things that we've seen in other quality of life studies in other diseases, but you also have to consider each disease unique and make sure you're looking at this from the perspective of the people that are really being affected. One of the great examples was that the teens really cared more about visible symptoms. For example, an adult with MS may have fatigue, severe fatigue. They may be unable to perform well in their job and that could be a game changer for them. Yet if they had a mild limp, they'd say, yeah, it's kind of embarrassing but I can keep going forward. I can hang in there and my colleagues at work might even support me; but for a teenager, they may care less about the fatigue and way more about having this limp that all their peers can now notice. The symptoms that are important to them can be totally different depending on the age group.
What we found is that teenagers look at things quite differently in trying to optimize their outcomes, and we don't just want them to be medically well. We want them to succeed in school, we want them to succeed in getting into college, or going into the workforce. So, we asked a lot about what it takes to get you there. We asked a lot of the young adults who had pediatric onset MS if they were successful? And if you were, what got you there; and the ones who hadn't reached that yet we asked-- what do you need?
When it came to transition needs, by far, we’ve found almost complete silence on the teenager’s part, which was a little surprising for us. We thought that there would be a little bit of discussion. They didn’t understand what a 504 is. We don't expect the average individual to know, but we thought that they might understand what the tools were, yet they really had no language for discussing that with us. We realized that the start of our transition talks had to be focused on the things that we use for that language.
For example, if I wanted to get a ding in my car fixed. I had to spend 20 minutes explaining to the dealership what I wanted. It was a regular car dealership, so it was integrated. But I had to find the right words to say. I want “auto body.” If I said vehicle repair, they said, oh, you want your car tires replaced? No, no, no. so, it’s very important to speak the right language just to get the process started. Those are some of the things that we found.
Q3. In what ways do environmental and genetic risk factors influence therapeutic decisions in pediatric patients with MS?
A3. They really play a big role in terms of the risk. We find that the more risk factors you have, likely we're dealing with a more severe disease. It doesn't necessarily always work that way, but you may be prepared to use a more potent therapy for individuals that are hitting more of the categories of concern.
But in addition to just the main disease modifying medications and MS, we look at vitamin D. And the data is yet to come out on that. There are some big studies that are trying to confirm or refute if vitamin D really has a therapeutic role, but we find that our teens and our kids have lower than average low vitamin D levels. We know that kids have low vitamin D levels nationwide in this country, but our patients are even lower than that. And that's one thing that we try to supplement and hope that by supplementing it, that it's going to be helpful. Maybe it's not as potent to therapy as the main medications, but we're hoping that's something to add on.
Q4. Overall, what are some advances, trends or recent studies regarding therapies that might support positive outcomes in children with MS?
A4 Interestingly, we just don't have a lot of that research in kids. There's been tons and tons of great research in adults. Like many other fields, you take what you learn from that and you apply it to the teens and kids. But we've learned time and time again they're not just little adults. They're truly a separate group, and we must consider them as such, and we really need those studies in kids.
The first study that came out confirmed that fingolimod was a good and effective therapy in children. But does that mean that you're only limited to using the only FDA approved option, or do you really want to try to offer families the litany of choices that you do for an adult with MS? When I meet families for the first time, we’re spending a good hour just talking about the different treatment choices with them and looking at the risks, the benefits, why one option might be chosen over another, how it's going to affect their lifestyle, and how it might fit into their life.
We want to still be able to make those decisions. I think we can make a more informed decision with fingolimod, but we don't want to just jump to conclusions with all those other therapies. We're a little bit behind the mark when it comes to therapies with kids, and we really need all those studies. They are active, and they are being done now; we're really waiting for those results to come out. That's going to be a huge change. Basically, that's the real trend. We're now going to see those studies in adults being replicated in kids one by one. Every time a new therapy comes out for adults, it must be validated in children as well.
Part of the regulations now do stipulate that these studies must be done. If you do a study in the adult population, you must see if you can do it in the pediatric population. You can't just say, hey, you know we're done. That's really what we're looking for in terms of getting the big therapeutic outcomes.
Chitnis, T., et al. 2021. Trial of Fingolimod versus Interferon Beta-1a in Pediatric Multiple Sclerosis | NEJM. [online] New England Journal of Medicine. Available at: <https://www.nejm.org/doi/
Vikram Bhise, MD, Is an Associate Professor at Rutgers – Robert Wood Johnson Medical School. He specializes in Epilepsy and Pediatric Neuroimmunology, and runs the pediatric demyelinating diseases program, evaluating children with multiple sclerosis, autoimmune encephalopathy, and related diseases. He trained in Pediatrics and Pediatric Neurology, at Maimonides Medical Center and Montefiore Medical Center, respectively. He subsequently received additional training in Clinical Neurophysiology with a focus on Epilepsy at SUNY Downstate Medical Center, and in Pediatric Multiple Sclerosis at SUNY Stony Brook Medical Center. Dr. Bhise conducts clinical research focused on biomarkers and quality of life in pediatric multiple sclerosis, as well as studies in epilepsy and neurogenetics.
Q1. As a specialist who focuses on neuroimmunology, what forms of measurement do you use to make an evaluation or diagnosis for children with multiple sclerosis?
A1. There's a lot that goes into evaluating or diagnosing children with MS. Usually we start off with the story that the family brings to us. We look at what the child is experiencing and what the parents are seeing. Then we do a dedicated examination trying to substantiate the findings that they're describing and look for others they may not even be aware of. If they are having some blurred vision in their eye, can we tell if there's some abnormalities there that are correlating with what they see?
We try to get a good sense of the time-course of things, observing whether this is the first time something's happened or if this has this been going on for a while. Have there been multiple things going on, multiple episodes? We're primarily looking for events called relapses, which are neurologic attacks that are not quick. They don’t last for seconds or hours; they can last for days to weeks, sometimes even months. Individuals will have episodes that tend to get worse and then tend to get better. This is the type of description we’re looking to come from the families.
Once that assessment is complete, we've found that the MRI is one of the best tools in helping us confirm the diagnosis. It's not just diagnostic but the MRI also has some prognostic potential and we're looking specifically for patterns in the MRIs. For children, that pattern can be a little bit more challenging. Their patterns can often overlap with patterns of other inflammatory diseases of the brain like ADEM for example and make it much more difficult for us to characterize someone as truly having MS.
There are also some other diseases which have been discovered in the past decade or elaborated upon like neuromyelitis optica and MOG antibody disorder, which can look exactly like MS in the early stages. Sometimes, that's just not all the information we need. Sometimes it's more difficult to make those distinctions and in these cases, we will look at a spinal tap, a lumbar puncture, and for specific studies from those procedures to help us get a better understanding. There may be other ancillary tests that we use, such as evoked potentials, for example.
Evoked potential testing has kind of fallen to the wayside over the past decade because of the MRI studies becoming a much more useful tool, but we may still use the visual evoked potential to see if there are subtle lesions that can't be seen on the MRI. Other methods might include optical coherence tomography, which is another test looking at the eye that gives you a specific look at the retinal nerve fiber layer, which gets thinned after attacks on the eye on the optic nerve.
We may do neuropsychological testing, which is a battery of tests looking at different cognitive domains and trying to get a sense of a person's cognitive profile to see if that matches what we would expect in somebody with MS. This test could be more challenging for a teen and a child, particularly a younger child. When it comes to pediatric neuropsychology, it's a little bit harder sometimes to get good data, particularly from younger kids.
In addition, we have a battery of tests that we do on the serum. Some disorders, like NMO and MOG, have antibodies that help us identify them. We don't have a specific test that says, yes, you have MS and no, you don't have MS. It's really the combination of all the tools.
We do these tests often to look for things that mimic MS. We look for other neuroinflammatory or neurobiological diseases that can look a lot like MS and fool us. Most of the time they don't look exactly like MS, but every now and then you get a case that's virtually indistinguishable. There are other tools which may be in less use, but we put the combination of all these things together to help us make the most informed judgment.
The goal is to be able to have honest discussions with families that these tools are just tools that we're trying to play catch up with a disease and try to make a decision as fast as possible to prevent someone from going untreated.
Q2. How does a diagnosis of multiple sclerosis affect the overall quality of life for a child/teen, and how does it affect their overall psychosocial health? Education? Transition needs? Etc.
A2. It can be quite profound, just hearing the diagnosis can be truly life changing for most folks. It would really depend on the family the first time that we meet them. If they have no suspicion that this is what's going on, that can be a shock. Other families may be more aware of what’s going on. Perhaps another physician has already suggested it, or they came from the ER which had already done some of the baseline tests like the MRI, and they had some kind of suspicion. Maybe they googled it and they saw something to be worried about, so they may be prepared. But even then, once you confirm the diagnosis, it's really like the sky falling at that point.
Past the diagnosis stage, there's really an adjustment phase that we see, and we've been doing some work in this. We started doing some work looking at quality of life. We've interviewed a large number of families and asked them some key questions such as, “What's important to you?” It is key for them to tell us rather than us telling them. By doing this, we’re finding out things that may not have been on the forefront of our minds, although it was certainly in the forefront of their minds, so it's a good learning opportunity.
These may be things that we've seen in other quality of life studies in other diseases, but you also have to consider each disease unique and make sure you're looking at this from the perspective of the people that are really being affected. One of the great examples was that the teens really cared more about visible symptoms. For example, an adult with MS may have fatigue, severe fatigue. They may be unable to perform well in their job and that could be a game changer for them. Yet if they had a mild limp, they'd say, yeah, it's kind of embarrassing but I can keep going forward. I can hang in there and my colleagues at work might even support me; but for a teenager, they may care less about the fatigue and way more about having this limp that all their peers can now notice. The symptoms that are important to them can be totally different depending on the age group.
What we found is that teenagers look at things quite differently in trying to optimize their outcomes, and we don't just want them to be medically well. We want them to succeed in school, we want them to succeed in getting into college, or going into the workforce. So, we asked a lot about what it takes to get you there. We asked a lot of the young adults who had pediatric onset MS if they were successful? And if you were, what got you there; and the ones who hadn't reached that yet we asked-- what do you need?
When it came to transition needs, by far, we’ve found almost complete silence on the teenager’s part, which was a little surprising for us. We thought that there would be a little bit of discussion. They didn’t understand what a 504 is. We don't expect the average individual to know, but we thought that they might understand what the tools were, yet they really had no language for discussing that with us. We realized that the start of our transition talks had to be focused on the things that we use for that language.
For example, if I wanted to get a ding in my car fixed. I had to spend 20 minutes explaining to the dealership what I wanted. It was a regular car dealership, so it was integrated. But I had to find the right words to say. I want “auto body.” If I said vehicle repair, they said, oh, you want your car tires replaced? No, no, no. so, it’s very important to speak the right language just to get the process started. Those are some of the things that we found.
Q3. In what ways do environmental and genetic risk factors influence therapeutic decisions in pediatric patients with MS?
A3. They really play a big role in terms of the risk. We find that the more risk factors you have, likely we're dealing with a more severe disease. It doesn't necessarily always work that way, but you may be prepared to use a more potent therapy for individuals that are hitting more of the categories of concern.
But in addition to just the main disease modifying medications and MS, we look at vitamin D. And the data is yet to come out on that. There are some big studies that are trying to confirm or refute if vitamin D really has a therapeutic role, but we find that our teens and our kids have lower than average low vitamin D levels. We know that kids have low vitamin D levels nationwide in this country, but our patients are even lower than that. And that's one thing that we try to supplement and hope that by supplementing it, that it's going to be helpful. Maybe it's not as potent to therapy as the main medications, but we're hoping that's something to add on.
Q4. Overall, what are some advances, trends or recent studies regarding therapies that might support positive outcomes in children with MS?
A4 Interestingly, we just don't have a lot of that research in kids. There's been tons and tons of great research in adults. Like many other fields, you take what you learn from that and you apply it to the teens and kids. But we've learned time and time again they're not just little adults. They're truly a separate group, and we must consider them as such, and we really need those studies in kids.
The first study that came out confirmed that fingolimod was a good and effective therapy in children. But does that mean that you're only limited to using the only FDA approved option, or do you really want to try to offer families the litany of choices that you do for an adult with MS? When I meet families for the first time, we’re spending a good hour just talking about the different treatment choices with them and looking at the risks, the benefits, why one option might be chosen over another, how it's going to affect their lifestyle, and how it might fit into their life.
We want to still be able to make those decisions. I think we can make a more informed decision with fingolimod, but we don't want to just jump to conclusions with all those other therapies. We're a little bit behind the mark when it comes to therapies with kids, and we really need all those studies. They are active, and they are being done now; we're really waiting for those results to come out. That's going to be a huge change. Basically, that's the real trend. We're now going to see those studies in adults being replicated in kids one by one. Every time a new therapy comes out for adults, it must be validated in children as well.
Part of the regulations now do stipulate that these studies must be done. If you do a study in the adult population, you must see if you can do it in the pediatric population. You can't just say, hey, you know we're done. That's really what we're looking for in terms of getting the big therapeutic outcomes.
Vikram Bhise, MD, Is an Associate Professor at Rutgers – Robert Wood Johnson Medical School. He specializes in Epilepsy and Pediatric Neuroimmunology, and runs the pediatric demyelinating diseases program, evaluating children with multiple sclerosis, autoimmune encephalopathy, and related diseases. He trained in Pediatrics and Pediatric Neurology, at Maimonides Medical Center and Montefiore Medical Center, respectively. He subsequently received additional training in Clinical Neurophysiology with a focus on Epilepsy at SUNY Downstate Medical Center, and in Pediatric Multiple Sclerosis at SUNY Stony Brook Medical Center. Dr. Bhise conducts clinical research focused on biomarkers and quality of life in pediatric multiple sclerosis, as well as studies in epilepsy and neurogenetics.
Q1. As a specialist who focuses on neuroimmunology, what forms of measurement do you use to make an evaluation or diagnosis for children with multiple sclerosis?
A1. There's a lot that goes into evaluating or diagnosing children with MS. Usually we start off with the story that the family brings to us. We look at what the child is experiencing and what the parents are seeing. Then we do a dedicated examination trying to substantiate the findings that they're describing and look for others they may not even be aware of. If they are having some blurred vision in their eye, can we tell if there's some abnormalities there that are correlating with what they see?
We try to get a good sense of the time-course of things, observing whether this is the first time something's happened or if this has this been going on for a while. Have there been multiple things going on, multiple episodes? We're primarily looking for events called relapses, which are neurologic attacks that are not quick. They don’t last for seconds or hours; they can last for days to weeks, sometimes even months. Individuals will have episodes that tend to get worse and then tend to get better. This is the type of description we’re looking to come from the families.
Once that assessment is complete, we've found that the MRI is one of the best tools in helping us confirm the diagnosis. It's not just diagnostic but the MRI also has some prognostic potential and we're looking specifically for patterns in the MRIs. For children, that pattern can be a little bit more challenging. Their patterns can often overlap with patterns of other inflammatory diseases of the brain like ADEM for example and make it much more difficult for us to characterize someone as truly having MS.
There are also some other diseases which have been discovered in the past decade or elaborated upon like neuromyelitis optica and MOG antibody disorder, which can look exactly like MS in the early stages. Sometimes, that's just not all the information we need. Sometimes it's more difficult to make those distinctions and in these cases, we will look at a spinal tap, a lumbar puncture, and for specific studies from those procedures to help us get a better understanding. There may be other ancillary tests that we use, such as evoked potentials, for example.
Evoked potential testing has kind of fallen to the wayside over the past decade because of the MRI studies becoming a much more useful tool, but we may still use the visual evoked potential to see if there are subtle lesions that can't be seen on the MRI. Other methods might include optical coherence tomography, which is another test looking at the eye that gives you a specific look at the retinal nerve fiber layer, which gets thinned after attacks on the eye on the optic nerve.
We may do neuropsychological testing, which is a battery of tests looking at different cognitive domains and trying to get a sense of a person's cognitive profile to see if that matches what we would expect in somebody with MS. This test could be more challenging for a teen and a child, particularly a younger child. When it comes to pediatric neuropsychology, it's a little bit harder sometimes to get good data, particularly from younger kids.
In addition, we have a battery of tests that we do on the serum. Some disorders, like NMO and MOG, have antibodies that help us identify them. We don't have a specific test that says, yes, you have MS and no, you don't have MS. It's really the combination of all the tools.
We do these tests often to look for things that mimic MS. We look for other neuroinflammatory or neurobiological diseases that can look a lot like MS and fool us. Most of the time they don't look exactly like MS, but every now and then you get a case that's virtually indistinguishable. There are other tools which may be in less use, but we put the combination of all these things together to help us make the most informed judgment.
The goal is to be able to have honest discussions with families that these tools are just tools that we're trying to play catch up with a disease and try to make a decision as fast as possible to prevent someone from going untreated.
Q2. How does a diagnosis of multiple sclerosis affect the overall quality of life for a child/teen, and how does it affect their overall psychosocial health? Education? Transition needs? Etc.
A2. It can be quite profound, just hearing the diagnosis can be truly life changing for most folks. It would really depend on the family the first time that we meet them. If they have no suspicion that this is what's going on, that can be a shock. Other families may be more aware of what’s going on. Perhaps another physician has already suggested it, or they came from the ER which had already done some of the baseline tests like the MRI, and they had some kind of suspicion. Maybe they googled it and they saw something to be worried about, so they may be prepared. But even then, once you confirm the diagnosis, it's really like the sky falling at that point.
Past the diagnosis stage, there's really an adjustment phase that we see, and we've been doing some work in this. We started doing some work looking at quality of life. We've interviewed a large number of families and asked them some key questions such as, “What's important to you?” It is key for them to tell us rather than us telling them. By doing this, we’re finding out things that may not have been on the forefront of our minds, although it was certainly in the forefront of their minds, so it's a good learning opportunity.
These may be things that we've seen in other quality of life studies in other diseases, but you also have to consider each disease unique and make sure you're looking at this from the perspective of the people that are really being affected. One of the great examples was that the teens really cared more about visible symptoms. For example, an adult with MS may have fatigue, severe fatigue. They may be unable to perform well in their job and that could be a game changer for them. Yet if they had a mild limp, they'd say, yeah, it's kind of embarrassing but I can keep going forward. I can hang in there and my colleagues at work might even support me; but for a teenager, they may care less about the fatigue and way more about having this limp that all their peers can now notice. The symptoms that are important to them can be totally different depending on the age group.
What we found is that teenagers look at things quite differently in trying to optimize their outcomes, and we don't just want them to be medically well. We want them to succeed in school, we want them to succeed in getting into college, or going into the workforce. So, we asked a lot about what it takes to get you there. We asked a lot of the young adults who had pediatric onset MS if they were successful? And if you were, what got you there; and the ones who hadn't reached that yet we asked-- what do you need?
When it came to transition needs, by far, we’ve found almost complete silence on the teenager’s part, which was a little surprising for us. We thought that there would be a little bit of discussion. They didn’t understand what a 504 is. We don't expect the average individual to know, but we thought that they might understand what the tools were, yet they really had no language for discussing that with us. We realized that the start of our transition talks had to be focused on the things that we use for that language.
For example, if I wanted to get a ding in my car fixed. I had to spend 20 minutes explaining to the dealership what I wanted. It was a regular car dealership, so it was integrated. But I had to find the right words to say. I want “auto body.” If I said vehicle repair, they said, oh, you want your car tires replaced? No, no, no. so, it’s very important to speak the right language just to get the process started. Those are some of the things that we found.
Q3. In what ways do environmental and genetic risk factors influence therapeutic decisions in pediatric patients with MS?
A3. They really play a big role in terms of the risk. We find that the more risk factors you have, likely we're dealing with a more severe disease. It doesn't necessarily always work that way, but you may be prepared to use a more potent therapy for individuals that are hitting more of the categories of concern.
But in addition to just the main disease modifying medications and MS, we look at vitamin D. And the data is yet to come out on that. There are some big studies that are trying to confirm or refute if vitamin D really has a therapeutic role, but we find that our teens and our kids have lower than average low vitamin D levels. We know that kids have low vitamin D levels nationwide in this country, but our patients are even lower than that. And that's one thing that we try to supplement and hope that by supplementing it, that it's going to be helpful. Maybe it's not as potent to therapy as the main medications, but we're hoping that's something to add on.
Q4. Overall, what are some advances, trends or recent studies regarding therapies that might support positive outcomes in children with MS?
A4 Interestingly, we just don't have a lot of that research in kids. There's been tons and tons of great research in adults. Like many other fields, you take what you learn from that and you apply it to the teens and kids. But we've learned time and time again they're not just little adults. They're truly a separate group, and we must consider them as such, and we really need those studies in kids.
The first study that came out confirmed that fingolimod was a good and effective therapy in children. But does that mean that you're only limited to using the only FDA approved option, or do you really want to try to offer families the litany of choices that you do for an adult with MS? When I meet families for the first time, we’re spending a good hour just talking about the different treatment choices with them and looking at the risks, the benefits, why one option might be chosen over another, how it's going to affect their lifestyle, and how it might fit into their life.
We want to still be able to make those decisions. I think we can make a more informed decision with fingolimod, but we don't want to just jump to conclusions with all those other therapies. We're a little bit behind the mark when it comes to therapies with kids, and we really need all those studies. They are active, and they are being done now; we're really waiting for those results to come out. That's going to be a huge change. Basically, that's the real trend. We're now going to see those studies in adults being replicated in kids one by one. Every time a new therapy comes out for adults, it must be validated in children as well.
Part of the regulations now do stipulate that these studies must be done. If you do a study in the adult population, you must see if you can do it in the pediatric population. You can't just say, hey, you know we're done. That's really what we're looking for in terms of getting the big therapeutic outcomes.
Chitnis, T., et al. 2021. Trial of Fingolimod versus Interferon Beta-1a in Pediatric Multiple Sclerosis | NEJM. [online] New England Journal of Medicine. Available at: <https://www.nejm.org/doi/
Chitnis, T., et al. 2021. Trial of Fingolimod versus Interferon Beta-1a in Pediatric Multiple Sclerosis | NEJM. [online] New England Journal of Medicine. Available at: <https://www.nejm.org/doi/