User login
Neurology Reviews covers innovative and emerging news in neurology and neuroscience every month, with a focus on practical approaches to treating Parkinson's disease, epilepsy, headache, stroke, multiple sclerosis, Alzheimer's disease, and other neurologic disorders.
PML
Progressive multifocal leukoencephalopathy
Rituxan
The leading independent newspaper covering neurology news and commentary.
The top tax breaks that physicians use
Plenty of perks come along with earning a physician’s salary, but a low tax rate isn’t among them. Medscape’s Physicians and Taxes Report 2023 shows that last year, doctors paid an average of nearly $100,000 in state and federal taxes, and three-quarters of them thought that they were paying too much to Uncle Sam. In most cases, it’s impossible to eliminate that tax bill, but physicians told us they have found ways to minimize it.
“The percentage you have to pay in taxes escalates as you earn more money, and most doctors are at the maximum rate,” says Paul Joseph, a certified public accountant and founder of Joseph & Joseph Tax & Payroll in Williamston, Mich. “So every dollar you can deduct from your income is worth more.”
To claim most of these options, you’ll need to itemize your deductions when filing your taxes.
Contribute to charity
Claimed by 70% of physicians in 2022.
Who’s eligible: Anyone.
How it works: If you itemize your taxes, you can deduct the value of cash, securities, or property donations to 501(c)(3) organizations. You’ll need a receipt from the charity and a third-party appraisal for any property donations worth more than $5,000.
Pro tip: Donating stocks that have appreciated in value can deliver additional tax benefits: You get to write off both the value of the contribution and avoid capital gains taxes that you’d face for selling the security.
Contribute to a pre-tax 401(k) account
Claimed by 60% of physicians in 2022.
Who’s eligible: Those who work for a company that sponsors a 401(k) plan.
How it works: Contributions to a 401(k) or 403(b) account come directly out of your paycheck, pre-tax, and grow tax-free until you withdraw them in retirement. Many companies offer a match on contributions. In 2023, you can contribute up to $22,500 ($30,000 if you’re age 50 or older) to a 401(k) account.
Pro tip: If you’re maxing out your 401(k) account, you can stash money in other tax-advantaged accounts such as a health savings account (if you have a high-deductible health plan) or an individual retirement account (IRA). Although employees with access to a 401(k) may not get the pre-tax advantage of the IRA contributions, the money will grow tax-free through retirement, and you may have access to additional investment options unavailable in your workplace plan.
“You want to maximize your retirement contributions,” says Mark Steber, the chief tax information officer for Jackson Hewitt Tax Services. “If you’re not taking full advantage of them, you’re probably leaving some tax dollars on the table.”
If you’re self-employed and don’t have access to a workplace plan, there are several options for tax-advantaged retirement savings, including a SEP IRA and a solo 401(k).
Deduct interest on a home mortgage
Claimed by 52% of physicians.
Who’s eligible: Most homeowners who have a mortgage.
How it works: Homeowners can deduct the interest paid on the first $750,000 of their mortgage. (Those who have had the same mortgage since before December 16, 2007, can deduct interest on the first $1 million of their loan.)
Pro tip: If you purchased a home this year and bought points to reduce the rate, you may be able to deduct the cost of those points on your taxes.
Physicians might also be eligible for other home-related tax benefits, such as for green home improvements under the Inflation Reduction Act or for home equity loans used to improve the value of your home.
Write off eligible business expenses
Claimed by 46% of physicians.
Who’s eligible: Physicians who own all or a portion of their practice, as well as those who work as consultants or contractors paid with a 1099.
How it works: Doctors who run their business using an LLC or S corporation can itemize the deductions on their Schedule C. There are dozens of deductions that might qualify, including for office space and supplies, medical equipment, uniforms, staff wages and benefits, and state and local tax payments. Physicians who work as consultants can deduct home office expenses, travel costs, and the price of supplies purchased for the job.
“For business expenses, you want to make sure that you’re tracking those expenses on an ongoing basis, rather than trying to reconstruct something at the end of the year from 8 months ago,” Mr. Joseph says. “You want to have a system in place that’s calculating those expenses every single day.”
Pro tip: The Tax Cuts and Jobs Act of 2017 also allows owners of pass-through businesses to deduct up to 20% of their business income.
“Not all physicians will qualify for that, because they are in a service-based business and many of them make too much money, but it’s always a good idea to look at whether that’s something they’re eligible for and make sure that they claim it,” says Eric Bronnenkant, head of tax at New York–based investment company Betterment.
Contribute to a 529 college savings plan
Claimed by 27% of physicians.
Who’s eligible: Those who live in the 37 states that offer a credit or deduction for 529 plan contributions.
How it works: The rules and amounts that qualify vary significantly by state. Most states offer benefits for contributions to in-state accounts only, whereas others offer a tax break for contributions to any 529 account.
Although there is no federal income tax benefit for contributions to a 529 plan, the money grows tax-free until tapped for qualified education expenses, which include both private primary and high school tuition and college costs. Starting in 2024, up to $35,000 in unused funds can roll over into a Roth IRA for the beneficiary.
“It’s not just about the immediate deduction with a 529 account,” says Brian Copeland, partner and director of financial planning with Hightower Wealth Advisors in St. Louis. “It’s not saving you a lot on day one; it’s more about as that account grows, you don’t have to pay taxes on it along the way, so you’re sheltering it from taxes for the 18 years you’re saving for your kids’ college.”
Pro tip: Even if you live in a state without a state income tax or without a tax break for 529 contributions, opening an account can be a smart financial move. Because you don’t need to choose an in-state plan for the tax breaks, look for one that offers low fees and investment options that you like.
Sell investments at a loss
Claimed by 22% of physicians.
Who’s eligible: Anyone who has sold stocks, mutual funds, or other investments at a loss.
How it works: After selling a security that has lost value, you can deduct the value of that loss on your taxes to offset capital gains in the same year. If you have more losses than gains, you can use the losses to offset up to $3,000 in ordinary income per year. If you have more than $3,000 in losses, you can carry those losses forward to offset future income or capital gains.
Pro tip: In years with a lot of market volatility, such as this one, there’s potential to engage in “tax loss harvesting” in which you intentionally sell securities that have lost value to realize the losses for the tax benefits. Keep in mind that if you sell a security at a loss, you cannot repurchase the same security within 30 days – the IRS sees that as a “wash sale,” which does not qualify for a capital loss for tax purposes.
Contribute to a backdoor Roth IRA
Claimed by 20% of physicians.
Who’s eligible: Anyone who wishes to contribute to a Roth IRA but is not allowed to do so because their income is too high.
How it works: High earners typically don’t qualify for contributions to a Roth IRA, in which contributions go in after taxes but grow tax-free and distributions in retirement are also tax-free. But there are no income requirements for making after-tax contributions to a traditional and then converting it to a Roth IRA.
There are, however, complex tax rules for those who also have a traditional IRA that’s funded with pre-tax dollars. If that’s the case, work with a tax pro or financial advisor to determine whether a backdoor Roth conversion is the most tax-efficient approach for your situation.
Pro tip: A growing number of workplace retirement plans now include an option for Roth contributions. There are no income limits on a Roth 401(k), so contributing to that type of an account could be a smart route for taxpayers for whom a backdoor conversion doesn’t make sense.
A version of this article appeared on Medscape.com.
Plenty of perks come along with earning a physician’s salary, but a low tax rate isn’t among them. Medscape’s Physicians and Taxes Report 2023 shows that last year, doctors paid an average of nearly $100,000 in state and federal taxes, and three-quarters of them thought that they were paying too much to Uncle Sam. In most cases, it’s impossible to eliminate that tax bill, but physicians told us they have found ways to minimize it.
“The percentage you have to pay in taxes escalates as you earn more money, and most doctors are at the maximum rate,” says Paul Joseph, a certified public accountant and founder of Joseph & Joseph Tax & Payroll in Williamston, Mich. “So every dollar you can deduct from your income is worth more.”
To claim most of these options, you’ll need to itemize your deductions when filing your taxes.
Contribute to charity
Claimed by 70% of physicians in 2022.
Who’s eligible: Anyone.
How it works: If you itemize your taxes, you can deduct the value of cash, securities, or property donations to 501(c)(3) organizations. You’ll need a receipt from the charity and a third-party appraisal for any property donations worth more than $5,000.
Pro tip: Donating stocks that have appreciated in value can deliver additional tax benefits: You get to write off both the value of the contribution and avoid capital gains taxes that you’d face for selling the security.
Contribute to a pre-tax 401(k) account
Claimed by 60% of physicians in 2022.
Who’s eligible: Those who work for a company that sponsors a 401(k) plan.
How it works: Contributions to a 401(k) or 403(b) account come directly out of your paycheck, pre-tax, and grow tax-free until you withdraw them in retirement. Many companies offer a match on contributions. In 2023, you can contribute up to $22,500 ($30,000 if you’re age 50 or older) to a 401(k) account.
Pro tip: If you’re maxing out your 401(k) account, you can stash money in other tax-advantaged accounts such as a health savings account (if you have a high-deductible health plan) or an individual retirement account (IRA). Although employees with access to a 401(k) may not get the pre-tax advantage of the IRA contributions, the money will grow tax-free through retirement, and you may have access to additional investment options unavailable in your workplace plan.
“You want to maximize your retirement contributions,” says Mark Steber, the chief tax information officer for Jackson Hewitt Tax Services. “If you’re not taking full advantage of them, you’re probably leaving some tax dollars on the table.”
If you’re self-employed and don’t have access to a workplace plan, there are several options for tax-advantaged retirement savings, including a SEP IRA and a solo 401(k).
Deduct interest on a home mortgage
Claimed by 52% of physicians.
Who’s eligible: Most homeowners who have a mortgage.
How it works: Homeowners can deduct the interest paid on the first $750,000 of their mortgage. (Those who have had the same mortgage since before December 16, 2007, can deduct interest on the first $1 million of their loan.)
Pro tip: If you purchased a home this year and bought points to reduce the rate, you may be able to deduct the cost of those points on your taxes.
Physicians might also be eligible for other home-related tax benefits, such as for green home improvements under the Inflation Reduction Act or for home equity loans used to improve the value of your home.
Write off eligible business expenses
Claimed by 46% of physicians.
Who’s eligible: Physicians who own all or a portion of their practice, as well as those who work as consultants or contractors paid with a 1099.
How it works: Doctors who run their business using an LLC or S corporation can itemize the deductions on their Schedule C. There are dozens of deductions that might qualify, including for office space and supplies, medical equipment, uniforms, staff wages and benefits, and state and local tax payments. Physicians who work as consultants can deduct home office expenses, travel costs, and the price of supplies purchased for the job.
“For business expenses, you want to make sure that you’re tracking those expenses on an ongoing basis, rather than trying to reconstruct something at the end of the year from 8 months ago,” Mr. Joseph says. “You want to have a system in place that’s calculating those expenses every single day.”
Pro tip: The Tax Cuts and Jobs Act of 2017 also allows owners of pass-through businesses to deduct up to 20% of their business income.
“Not all physicians will qualify for that, because they are in a service-based business and many of them make too much money, but it’s always a good idea to look at whether that’s something they’re eligible for and make sure that they claim it,” says Eric Bronnenkant, head of tax at New York–based investment company Betterment.
Contribute to a 529 college savings plan
Claimed by 27% of physicians.
Who’s eligible: Those who live in the 37 states that offer a credit or deduction for 529 plan contributions.
How it works: The rules and amounts that qualify vary significantly by state. Most states offer benefits for contributions to in-state accounts only, whereas others offer a tax break for contributions to any 529 account.
Although there is no federal income tax benefit for contributions to a 529 plan, the money grows tax-free until tapped for qualified education expenses, which include both private primary and high school tuition and college costs. Starting in 2024, up to $35,000 in unused funds can roll over into a Roth IRA for the beneficiary.
“It’s not just about the immediate deduction with a 529 account,” says Brian Copeland, partner and director of financial planning with Hightower Wealth Advisors in St. Louis. “It’s not saving you a lot on day one; it’s more about as that account grows, you don’t have to pay taxes on it along the way, so you’re sheltering it from taxes for the 18 years you’re saving for your kids’ college.”
Pro tip: Even if you live in a state without a state income tax or without a tax break for 529 contributions, opening an account can be a smart financial move. Because you don’t need to choose an in-state plan for the tax breaks, look for one that offers low fees and investment options that you like.
Sell investments at a loss
Claimed by 22% of physicians.
Who’s eligible: Anyone who has sold stocks, mutual funds, or other investments at a loss.
How it works: After selling a security that has lost value, you can deduct the value of that loss on your taxes to offset capital gains in the same year. If you have more losses than gains, you can use the losses to offset up to $3,000 in ordinary income per year. If you have more than $3,000 in losses, you can carry those losses forward to offset future income or capital gains.
Pro tip: In years with a lot of market volatility, such as this one, there’s potential to engage in “tax loss harvesting” in which you intentionally sell securities that have lost value to realize the losses for the tax benefits. Keep in mind that if you sell a security at a loss, you cannot repurchase the same security within 30 days – the IRS sees that as a “wash sale,” which does not qualify for a capital loss for tax purposes.
Contribute to a backdoor Roth IRA
Claimed by 20% of physicians.
Who’s eligible: Anyone who wishes to contribute to a Roth IRA but is not allowed to do so because their income is too high.
How it works: High earners typically don’t qualify for contributions to a Roth IRA, in which contributions go in after taxes but grow tax-free and distributions in retirement are also tax-free. But there are no income requirements for making after-tax contributions to a traditional and then converting it to a Roth IRA.
There are, however, complex tax rules for those who also have a traditional IRA that’s funded with pre-tax dollars. If that’s the case, work with a tax pro or financial advisor to determine whether a backdoor Roth conversion is the most tax-efficient approach for your situation.
Pro tip: A growing number of workplace retirement plans now include an option for Roth contributions. There are no income limits on a Roth 401(k), so contributing to that type of an account could be a smart route for taxpayers for whom a backdoor conversion doesn’t make sense.
A version of this article appeared on Medscape.com.
Plenty of perks come along with earning a physician’s salary, but a low tax rate isn’t among them. Medscape’s Physicians and Taxes Report 2023 shows that last year, doctors paid an average of nearly $100,000 in state and federal taxes, and three-quarters of them thought that they were paying too much to Uncle Sam. In most cases, it’s impossible to eliminate that tax bill, but physicians told us they have found ways to minimize it.
“The percentage you have to pay in taxes escalates as you earn more money, and most doctors are at the maximum rate,” says Paul Joseph, a certified public accountant and founder of Joseph & Joseph Tax & Payroll in Williamston, Mich. “So every dollar you can deduct from your income is worth more.”
To claim most of these options, you’ll need to itemize your deductions when filing your taxes.
Contribute to charity
Claimed by 70% of physicians in 2022.
Who’s eligible: Anyone.
How it works: If you itemize your taxes, you can deduct the value of cash, securities, or property donations to 501(c)(3) organizations. You’ll need a receipt from the charity and a third-party appraisal for any property donations worth more than $5,000.
Pro tip: Donating stocks that have appreciated in value can deliver additional tax benefits: You get to write off both the value of the contribution and avoid capital gains taxes that you’d face for selling the security.
Contribute to a pre-tax 401(k) account
Claimed by 60% of physicians in 2022.
Who’s eligible: Those who work for a company that sponsors a 401(k) plan.
How it works: Contributions to a 401(k) or 403(b) account come directly out of your paycheck, pre-tax, and grow tax-free until you withdraw them in retirement. Many companies offer a match on contributions. In 2023, you can contribute up to $22,500 ($30,000 if you’re age 50 or older) to a 401(k) account.
Pro tip: If you’re maxing out your 401(k) account, you can stash money in other tax-advantaged accounts such as a health savings account (if you have a high-deductible health plan) or an individual retirement account (IRA). Although employees with access to a 401(k) may not get the pre-tax advantage of the IRA contributions, the money will grow tax-free through retirement, and you may have access to additional investment options unavailable in your workplace plan.
“You want to maximize your retirement contributions,” says Mark Steber, the chief tax information officer for Jackson Hewitt Tax Services. “If you’re not taking full advantage of them, you’re probably leaving some tax dollars on the table.”
If you’re self-employed and don’t have access to a workplace plan, there are several options for tax-advantaged retirement savings, including a SEP IRA and a solo 401(k).
Deduct interest on a home mortgage
Claimed by 52% of physicians.
Who’s eligible: Most homeowners who have a mortgage.
How it works: Homeowners can deduct the interest paid on the first $750,000 of their mortgage. (Those who have had the same mortgage since before December 16, 2007, can deduct interest on the first $1 million of their loan.)
Pro tip: If you purchased a home this year and bought points to reduce the rate, you may be able to deduct the cost of those points on your taxes.
Physicians might also be eligible for other home-related tax benefits, such as for green home improvements under the Inflation Reduction Act or for home equity loans used to improve the value of your home.
Write off eligible business expenses
Claimed by 46% of physicians.
Who’s eligible: Physicians who own all or a portion of their practice, as well as those who work as consultants or contractors paid with a 1099.
How it works: Doctors who run their business using an LLC or S corporation can itemize the deductions on their Schedule C. There are dozens of deductions that might qualify, including for office space and supplies, medical equipment, uniforms, staff wages and benefits, and state and local tax payments. Physicians who work as consultants can deduct home office expenses, travel costs, and the price of supplies purchased for the job.
“For business expenses, you want to make sure that you’re tracking those expenses on an ongoing basis, rather than trying to reconstruct something at the end of the year from 8 months ago,” Mr. Joseph says. “You want to have a system in place that’s calculating those expenses every single day.”
Pro tip: The Tax Cuts and Jobs Act of 2017 also allows owners of pass-through businesses to deduct up to 20% of their business income.
“Not all physicians will qualify for that, because they are in a service-based business and many of them make too much money, but it’s always a good idea to look at whether that’s something they’re eligible for and make sure that they claim it,” says Eric Bronnenkant, head of tax at New York–based investment company Betterment.
Contribute to a 529 college savings plan
Claimed by 27% of physicians.
Who’s eligible: Those who live in the 37 states that offer a credit or deduction for 529 plan contributions.
How it works: The rules and amounts that qualify vary significantly by state. Most states offer benefits for contributions to in-state accounts only, whereas others offer a tax break for contributions to any 529 account.
Although there is no federal income tax benefit for contributions to a 529 plan, the money grows tax-free until tapped for qualified education expenses, which include both private primary and high school tuition and college costs. Starting in 2024, up to $35,000 in unused funds can roll over into a Roth IRA for the beneficiary.
“It’s not just about the immediate deduction with a 529 account,” says Brian Copeland, partner and director of financial planning with Hightower Wealth Advisors in St. Louis. “It’s not saving you a lot on day one; it’s more about as that account grows, you don’t have to pay taxes on it along the way, so you’re sheltering it from taxes for the 18 years you’re saving for your kids’ college.”
Pro tip: Even if you live in a state without a state income tax or without a tax break for 529 contributions, opening an account can be a smart financial move. Because you don’t need to choose an in-state plan for the tax breaks, look for one that offers low fees and investment options that you like.
Sell investments at a loss
Claimed by 22% of physicians.
Who’s eligible: Anyone who has sold stocks, mutual funds, or other investments at a loss.
How it works: After selling a security that has lost value, you can deduct the value of that loss on your taxes to offset capital gains in the same year. If you have more losses than gains, you can use the losses to offset up to $3,000 in ordinary income per year. If you have more than $3,000 in losses, you can carry those losses forward to offset future income or capital gains.
Pro tip: In years with a lot of market volatility, such as this one, there’s potential to engage in “tax loss harvesting” in which you intentionally sell securities that have lost value to realize the losses for the tax benefits. Keep in mind that if you sell a security at a loss, you cannot repurchase the same security within 30 days – the IRS sees that as a “wash sale,” which does not qualify for a capital loss for tax purposes.
Contribute to a backdoor Roth IRA
Claimed by 20% of physicians.
Who’s eligible: Anyone who wishes to contribute to a Roth IRA but is not allowed to do so because their income is too high.
How it works: High earners typically don’t qualify for contributions to a Roth IRA, in which contributions go in after taxes but grow tax-free and distributions in retirement are also tax-free. But there are no income requirements for making after-tax contributions to a traditional and then converting it to a Roth IRA.
There are, however, complex tax rules for those who also have a traditional IRA that’s funded with pre-tax dollars. If that’s the case, work with a tax pro or financial advisor to determine whether a backdoor Roth conversion is the most tax-efficient approach for your situation.
Pro tip: A growing number of workplace retirement plans now include an option for Roth contributions. There are no income limits on a Roth 401(k), so contributing to that type of an account could be a smart route for taxpayers for whom a backdoor conversion doesn’t make sense.
A version of this article appeared on Medscape.com.
Hunt, gather, and turn on the Keurig
I’m a creature of habit. I suspect most of us are.
One can of Diet Coke on the drive to my office. Turn on the WiFi and air conditioning. Fire up the computer and unload my briefcase. Then do online refills, check the Astronomy Picture of the Day, look over the day’s schedule, turn on the Keurig, and make one cup of coffee. And so on.
I’m sure most of us have similar routines. Our brains are probably wired that way for survival, though the reasons aren’t the same anymore. Once it was get up, look outside the cave for predators, make sure the tribe is all accounted for, go to the stream for water, look for berries.
The fact is that automatic habits are critical for everything we do. Driving a car is really a series of repetitive tasks. Being able to put most of the ride on our brain’s autopilot allows us to move our attention to scanning the surroundings for changes, and to think about other items such as wonder what to do for dinner and if I remembered to turn off theWiFi and Keurig.
The practice of medicine is similar. Some things are internalized. Watching patients walk back to my office, looking at their hands as they fill out forms, hearing them introduce themselves, and other things that we subconsciously process as part of the exam before we’ve even officially begun the appointment. I quietly file such things away to be used later in the visit.
It certainly wasn’t always that way. In training we learn to filter out signal from noise, because the information available is huge. We all read tests of some sort. When I began reading EEGs, the images and lines were overwhelming, but with time and experience I became skilled at whittling down the mass of information into the things that really needed to be noted so I could turn pages faster (yes, youngsters, EEGs used to be on paper). Now, scanning the screen becomes a background habit, with the brain focusing more on things that stand out (or going back to thinking about what to do for dinner).
The brain in this way is the ultimate Swiss Army Knife – many tools available, but how we adapt and use them for our individual needs is variable.
Which is pretty impressive, actually. In the era of AI and computers, we each come with a (roughly) 2.5-petabyte hard drive that’s not only capable of storing all that information, but figuring out how to use it when we need to. The process is so smooth that we’re rarely aware of it. But what a marvel it is.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
I’m a creature of habit. I suspect most of us are.
One can of Diet Coke on the drive to my office. Turn on the WiFi and air conditioning. Fire up the computer and unload my briefcase. Then do online refills, check the Astronomy Picture of the Day, look over the day’s schedule, turn on the Keurig, and make one cup of coffee. And so on.
I’m sure most of us have similar routines. Our brains are probably wired that way for survival, though the reasons aren’t the same anymore. Once it was get up, look outside the cave for predators, make sure the tribe is all accounted for, go to the stream for water, look for berries.
The fact is that automatic habits are critical for everything we do. Driving a car is really a series of repetitive tasks. Being able to put most of the ride on our brain’s autopilot allows us to move our attention to scanning the surroundings for changes, and to think about other items such as wonder what to do for dinner and if I remembered to turn off theWiFi and Keurig.
The practice of medicine is similar. Some things are internalized. Watching patients walk back to my office, looking at their hands as they fill out forms, hearing them introduce themselves, and other things that we subconsciously process as part of the exam before we’ve even officially begun the appointment. I quietly file such things away to be used later in the visit.
It certainly wasn’t always that way. In training we learn to filter out signal from noise, because the information available is huge. We all read tests of some sort. When I began reading EEGs, the images and lines were overwhelming, but with time and experience I became skilled at whittling down the mass of information into the things that really needed to be noted so I could turn pages faster (yes, youngsters, EEGs used to be on paper). Now, scanning the screen becomes a background habit, with the brain focusing more on things that stand out (or going back to thinking about what to do for dinner).
The brain in this way is the ultimate Swiss Army Knife – many tools available, but how we adapt and use them for our individual needs is variable.
Which is pretty impressive, actually. In the era of AI and computers, we each come with a (roughly) 2.5-petabyte hard drive that’s not only capable of storing all that information, but figuring out how to use it when we need to. The process is so smooth that we’re rarely aware of it. But what a marvel it is.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
I’m a creature of habit. I suspect most of us are.
One can of Diet Coke on the drive to my office. Turn on the WiFi and air conditioning. Fire up the computer and unload my briefcase. Then do online refills, check the Astronomy Picture of the Day, look over the day’s schedule, turn on the Keurig, and make one cup of coffee. And so on.
I’m sure most of us have similar routines. Our brains are probably wired that way for survival, though the reasons aren’t the same anymore. Once it was get up, look outside the cave for predators, make sure the tribe is all accounted for, go to the stream for water, look for berries.
The fact is that automatic habits are critical for everything we do. Driving a car is really a series of repetitive tasks. Being able to put most of the ride on our brain’s autopilot allows us to move our attention to scanning the surroundings for changes, and to think about other items such as wonder what to do for dinner and if I remembered to turn off theWiFi and Keurig.
The practice of medicine is similar. Some things are internalized. Watching patients walk back to my office, looking at their hands as they fill out forms, hearing them introduce themselves, and other things that we subconsciously process as part of the exam before we’ve even officially begun the appointment. I quietly file such things away to be used later in the visit.
It certainly wasn’t always that way. In training we learn to filter out signal from noise, because the information available is huge. We all read tests of some sort. When I began reading EEGs, the images and lines were overwhelming, but with time and experience I became skilled at whittling down the mass of information into the things that really needed to be noted so I could turn pages faster (yes, youngsters, EEGs used to be on paper). Now, scanning the screen becomes a background habit, with the brain focusing more on things that stand out (or going back to thinking about what to do for dinner).
The brain in this way is the ultimate Swiss Army Knife – many tools available, but how we adapt and use them for our individual needs is variable.
Which is pretty impressive, actually. In the era of AI and computers, we each come with a (roughly) 2.5-petabyte hard drive that’s not only capable of storing all that information, but figuring out how to use it when we need to. The process is so smooth that we’re rarely aware of it. But what a marvel it is.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
How does lecanemab work in Alzheimer’s?
Lecanemab (Lequembi, Esai), an amyloid-beta–directed antibody therapy, is approved by the Food and Drug Administration for the treatment of Alzheimer’s disease (AD). But exactly how the drug clears amyloid-beta wasn’t clear.
The investigators tested the effectiveness of various forms of amyloid-beta in activating the plasma contact system and found that amyloid-beta protofibrils, known to be the most toxic form of amyloid-beta, promoted the activation of this molecular cascade and that lecanemab inhibited pathway activation.
“In our study, we looked at lecanemab and found it can block the activation of the contact system, which could be one of the reasons that it works so well for AD,” study coinvestigator Erin Norris, PhD, research associate professor, Rockefeller University, New York, said in an interview.
The study was published online in the Proceedings of the National Academy of Science.
Unknown mechanism
“Many years ago, we started looking at the involvement of vascular dysfunction in AD,” Dr. Norris said. “We wanted to see whether or not irregular blood clotting or problems with blood flow was problematic in Alzheimer’s patients.”
The researchers found that fibrin, a major component involved in blood clotting, can extravasate into the brain.
“The blood-brain barrier can break down in Alzheimer’s, so things from the blood can move into the brain and deposit there,” she added. Fibrin then interacts with amyloid-beta, the major pathogenic protein in AD.
Dr. Norris explained that fibrin clots can form in two different ways. One is through the normal process that occurs when there’s an injury and bleeding. The second is through intrinsic clotting, which takes place through the contact system.
“We started looking into this system and found that the plasma of Alzheimer’s patients showed irregular levels of these enzymes and proteins that are part of the intrinsic clotting system compared to those of normal controls,” said Dr. Norris.
“This paper was an extension of years studying this pathway and these mechanisms. It was also inspired by the approval of lecanemab and its release for use in Alzheimer’s patients,” she added.
In previous research, the same researchers found that amyloid-beta has different forms. “It’s normally soluble, and it’s a very tiny molecule,” Dr. Norris said. “But over time, and in different situations, it can start to aggregate, becoming bigger and bigger.”
Implications beyond Alzheimer’s
Postmortem tissue analysis has found fibrillar plaques that are “clumped together.” These are insoluble and hard to get rid of, she said. “Protofibrils are the step before amyloid-beta forms fibrils and are considered to be the most toxic form, although the mechanism behind why it’s so toxic is not understood.”
Previous research has already shown that amyloid-beta can activate the contact system. The contact system has two “arms,” the first of which is involved with clotting, and the second with inflammation, Dr. Norris said. In fact, it’s the plasma contact system that links vascular and inflammatory pathways.
The plasma contact system leads to the clotting of fibrin, Dr. Norris continued. It activates factor XII, which leads to blood clotting by binding to coagulation factor XI.
The contact system also causes inflammation – the second “arm.” Bradykinin, a potent inflammatory molecule, is released by binding to high-molecular-weight kininogen (HK). In addition to inflammation, bradykinin can cause edema and blood-brain barrier permeability.
Although it’s been known that amyloid-beta can activate the contact system, the particular form of amyloid-beta implicated in this cascade has not been identified. And so, the researchers incubated amyloid-beta42 with human plasma, testing various types of amyloid-beta – monomers, oligomers, protofibrils, and fibrils – to see which would activate the contact system.
Amyloid-beta protofibrils promoted the activation of the contact system, as evidenced by several reactions, including activation of factor XII, while other forms of amyloid-beta did not. HK also “bound tightly” to amyloid-beta protofibrils, with “weaker” binding to other amyloid-beta species, the authors reported, confirming that amyloid-beta protofibrils bind to HK and factor XII.
Bradykinin levels were increased by amyloid-beta protofibrils, which also induced faster clotting, compared with other forms of amyloid-beta.
The researchers introduced lecanemab into the picture and found it “dramatically inhibited” contact system activation induced by amyloid-beta protofibrils. For example, it blocked the binding of factor XII to amyloid-beta. By contrast, human IgG (which the researchers used as a control) had no effect.
Additionally, lecanemab also prevented accelerated intrinsic coagulation in normal human plasma mediated by amyloid-beta protofibril.
Senior author Sidney Strickland, PhD, the Zachary and Elizabeth M. Fisher professor in Alzheimer’s and neurodegenerative disease, Rockefeller University, said in an interview: “One of the strong motivators for conducting this study was the fact that this drug, which is effective in AD, targets this specific form of amyloid-beta; but no one knows why it›s more toxic. We thought we could see if we could tie it to what we›re working on, and we found it ties in beautifully.”
The findings have implications that go beyond AD, Dr. Strickland said. “The contact system is implicated in lots of different pathologies, including sickle cell anemia, sepsis, inflammatory bowel disease, and so on.” Blocking the contact system might be a helpful approach in these conditions too.
Innovative, plausible, but still preliminary
In a comment, Heather M. Snyder, PhD, vice president of medical and scientific relations at the Alzheimer’s Association, called the investigation “innovative,” with ideas that are “certainly plausible.” However, “at this time, the work is preliminary and not conclusive.”
The hypothesized mechanisms for why amyloid (lecanemab’s target) is toxic to the brain “does incorporate important AD-related brain changes that have been observed in other studies, including inflammatory/immune changes and vascular-related changes,” said Dr. Snyder, who was not involved with the current study.
However, “additional studies that look both in model systems and in humans are needed to further illuminate these relationships,” Dr. Snyder said.
The study was supported by grants from the National Institutes of Health as well as the Robertson Therapeutic Development Fund, Samuel Newhouse Foundation, John A. Herrmann, and the May and Samuel Rudin Family Foundation. Dr. Norris, Dr. Strickland, and Dr. Snyder declared no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Lecanemab (Lequembi, Esai), an amyloid-beta–directed antibody therapy, is approved by the Food and Drug Administration for the treatment of Alzheimer’s disease (AD). But exactly how the drug clears amyloid-beta wasn’t clear.
The investigators tested the effectiveness of various forms of amyloid-beta in activating the plasma contact system and found that amyloid-beta protofibrils, known to be the most toxic form of amyloid-beta, promoted the activation of this molecular cascade and that lecanemab inhibited pathway activation.
“In our study, we looked at lecanemab and found it can block the activation of the contact system, which could be one of the reasons that it works so well for AD,” study coinvestigator Erin Norris, PhD, research associate professor, Rockefeller University, New York, said in an interview.
The study was published online in the Proceedings of the National Academy of Science.
Unknown mechanism
“Many years ago, we started looking at the involvement of vascular dysfunction in AD,” Dr. Norris said. “We wanted to see whether or not irregular blood clotting or problems with blood flow was problematic in Alzheimer’s patients.”
The researchers found that fibrin, a major component involved in blood clotting, can extravasate into the brain.
“The blood-brain barrier can break down in Alzheimer’s, so things from the blood can move into the brain and deposit there,” she added. Fibrin then interacts with amyloid-beta, the major pathogenic protein in AD.
Dr. Norris explained that fibrin clots can form in two different ways. One is through the normal process that occurs when there’s an injury and bleeding. The second is through intrinsic clotting, which takes place through the contact system.
“We started looking into this system and found that the plasma of Alzheimer’s patients showed irregular levels of these enzymes and proteins that are part of the intrinsic clotting system compared to those of normal controls,” said Dr. Norris.
“This paper was an extension of years studying this pathway and these mechanisms. It was also inspired by the approval of lecanemab and its release for use in Alzheimer’s patients,” she added.
In previous research, the same researchers found that amyloid-beta has different forms. “It’s normally soluble, and it’s a very tiny molecule,” Dr. Norris said. “But over time, and in different situations, it can start to aggregate, becoming bigger and bigger.”
Implications beyond Alzheimer’s
Postmortem tissue analysis has found fibrillar plaques that are “clumped together.” These are insoluble and hard to get rid of, she said. “Protofibrils are the step before amyloid-beta forms fibrils and are considered to be the most toxic form, although the mechanism behind why it’s so toxic is not understood.”
Previous research has already shown that amyloid-beta can activate the contact system. The contact system has two “arms,” the first of which is involved with clotting, and the second with inflammation, Dr. Norris said. In fact, it’s the plasma contact system that links vascular and inflammatory pathways.
The plasma contact system leads to the clotting of fibrin, Dr. Norris continued. It activates factor XII, which leads to blood clotting by binding to coagulation factor XI.
The contact system also causes inflammation – the second “arm.” Bradykinin, a potent inflammatory molecule, is released by binding to high-molecular-weight kininogen (HK). In addition to inflammation, bradykinin can cause edema and blood-brain barrier permeability.
Although it’s been known that amyloid-beta can activate the contact system, the particular form of amyloid-beta implicated in this cascade has not been identified. And so, the researchers incubated amyloid-beta42 with human plasma, testing various types of amyloid-beta – monomers, oligomers, protofibrils, and fibrils – to see which would activate the contact system.
Amyloid-beta protofibrils promoted the activation of the contact system, as evidenced by several reactions, including activation of factor XII, while other forms of amyloid-beta did not. HK also “bound tightly” to amyloid-beta protofibrils, with “weaker” binding to other amyloid-beta species, the authors reported, confirming that amyloid-beta protofibrils bind to HK and factor XII.
Bradykinin levels were increased by amyloid-beta protofibrils, which also induced faster clotting, compared with other forms of amyloid-beta.
The researchers introduced lecanemab into the picture and found it “dramatically inhibited” contact system activation induced by amyloid-beta protofibrils. For example, it blocked the binding of factor XII to amyloid-beta. By contrast, human IgG (which the researchers used as a control) had no effect.
Additionally, lecanemab also prevented accelerated intrinsic coagulation in normal human plasma mediated by amyloid-beta protofibril.
Senior author Sidney Strickland, PhD, the Zachary and Elizabeth M. Fisher professor in Alzheimer’s and neurodegenerative disease, Rockefeller University, said in an interview: “One of the strong motivators for conducting this study was the fact that this drug, which is effective in AD, targets this specific form of amyloid-beta; but no one knows why it›s more toxic. We thought we could see if we could tie it to what we›re working on, and we found it ties in beautifully.”
The findings have implications that go beyond AD, Dr. Strickland said. “The contact system is implicated in lots of different pathologies, including sickle cell anemia, sepsis, inflammatory bowel disease, and so on.” Blocking the contact system might be a helpful approach in these conditions too.
Innovative, plausible, but still preliminary
In a comment, Heather M. Snyder, PhD, vice president of medical and scientific relations at the Alzheimer’s Association, called the investigation “innovative,” with ideas that are “certainly plausible.” However, “at this time, the work is preliminary and not conclusive.”
The hypothesized mechanisms for why amyloid (lecanemab’s target) is toxic to the brain “does incorporate important AD-related brain changes that have been observed in other studies, including inflammatory/immune changes and vascular-related changes,” said Dr. Snyder, who was not involved with the current study.
However, “additional studies that look both in model systems and in humans are needed to further illuminate these relationships,” Dr. Snyder said.
The study was supported by grants from the National Institutes of Health as well as the Robertson Therapeutic Development Fund, Samuel Newhouse Foundation, John A. Herrmann, and the May and Samuel Rudin Family Foundation. Dr. Norris, Dr. Strickland, and Dr. Snyder declared no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Lecanemab (Lequembi, Esai), an amyloid-beta–directed antibody therapy, is approved by the Food and Drug Administration for the treatment of Alzheimer’s disease (AD). But exactly how the drug clears amyloid-beta wasn’t clear.
The investigators tested the effectiveness of various forms of amyloid-beta in activating the plasma contact system and found that amyloid-beta protofibrils, known to be the most toxic form of amyloid-beta, promoted the activation of this molecular cascade and that lecanemab inhibited pathway activation.
“In our study, we looked at lecanemab and found it can block the activation of the contact system, which could be one of the reasons that it works so well for AD,” study coinvestigator Erin Norris, PhD, research associate professor, Rockefeller University, New York, said in an interview.
The study was published online in the Proceedings of the National Academy of Science.
Unknown mechanism
“Many years ago, we started looking at the involvement of vascular dysfunction in AD,” Dr. Norris said. “We wanted to see whether or not irregular blood clotting or problems with blood flow was problematic in Alzheimer’s patients.”
The researchers found that fibrin, a major component involved in blood clotting, can extravasate into the brain.
“The blood-brain barrier can break down in Alzheimer’s, so things from the blood can move into the brain and deposit there,” she added. Fibrin then interacts with amyloid-beta, the major pathogenic protein in AD.
Dr. Norris explained that fibrin clots can form in two different ways. One is through the normal process that occurs when there’s an injury and bleeding. The second is through intrinsic clotting, which takes place through the contact system.
“We started looking into this system and found that the plasma of Alzheimer’s patients showed irregular levels of these enzymes and proteins that are part of the intrinsic clotting system compared to those of normal controls,” said Dr. Norris.
“This paper was an extension of years studying this pathway and these mechanisms. It was also inspired by the approval of lecanemab and its release for use in Alzheimer’s patients,” she added.
In previous research, the same researchers found that amyloid-beta has different forms. “It’s normally soluble, and it’s a very tiny molecule,” Dr. Norris said. “But over time, and in different situations, it can start to aggregate, becoming bigger and bigger.”
Implications beyond Alzheimer’s
Postmortem tissue analysis has found fibrillar plaques that are “clumped together.” These are insoluble and hard to get rid of, she said. “Protofibrils are the step before amyloid-beta forms fibrils and are considered to be the most toxic form, although the mechanism behind why it’s so toxic is not understood.”
Previous research has already shown that amyloid-beta can activate the contact system. The contact system has two “arms,” the first of which is involved with clotting, and the second with inflammation, Dr. Norris said. In fact, it’s the plasma contact system that links vascular and inflammatory pathways.
The plasma contact system leads to the clotting of fibrin, Dr. Norris continued. It activates factor XII, which leads to blood clotting by binding to coagulation factor XI.
The contact system also causes inflammation – the second “arm.” Bradykinin, a potent inflammatory molecule, is released by binding to high-molecular-weight kininogen (HK). In addition to inflammation, bradykinin can cause edema and blood-brain barrier permeability.
Although it’s been known that amyloid-beta can activate the contact system, the particular form of amyloid-beta implicated in this cascade has not been identified. And so, the researchers incubated amyloid-beta42 with human plasma, testing various types of amyloid-beta – monomers, oligomers, protofibrils, and fibrils – to see which would activate the contact system.
Amyloid-beta protofibrils promoted the activation of the contact system, as evidenced by several reactions, including activation of factor XII, while other forms of amyloid-beta did not. HK also “bound tightly” to amyloid-beta protofibrils, with “weaker” binding to other amyloid-beta species, the authors reported, confirming that amyloid-beta protofibrils bind to HK and factor XII.
Bradykinin levels were increased by amyloid-beta protofibrils, which also induced faster clotting, compared with other forms of amyloid-beta.
The researchers introduced lecanemab into the picture and found it “dramatically inhibited” contact system activation induced by amyloid-beta protofibrils. For example, it blocked the binding of factor XII to amyloid-beta. By contrast, human IgG (which the researchers used as a control) had no effect.
Additionally, lecanemab also prevented accelerated intrinsic coagulation in normal human plasma mediated by amyloid-beta protofibril.
Senior author Sidney Strickland, PhD, the Zachary and Elizabeth M. Fisher professor in Alzheimer’s and neurodegenerative disease, Rockefeller University, said in an interview: “One of the strong motivators for conducting this study was the fact that this drug, which is effective in AD, targets this specific form of amyloid-beta; but no one knows why it›s more toxic. We thought we could see if we could tie it to what we›re working on, and we found it ties in beautifully.”
The findings have implications that go beyond AD, Dr. Strickland said. “The contact system is implicated in lots of different pathologies, including sickle cell anemia, sepsis, inflammatory bowel disease, and so on.” Blocking the contact system might be a helpful approach in these conditions too.
Innovative, plausible, but still preliminary
In a comment, Heather M. Snyder, PhD, vice president of medical and scientific relations at the Alzheimer’s Association, called the investigation “innovative,” with ideas that are “certainly plausible.” However, “at this time, the work is preliminary and not conclusive.”
The hypothesized mechanisms for why amyloid (lecanemab’s target) is toxic to the brain “does incorporate important AD-related brain changes that have been observed in other studies, including inflammatory/immune changes and vascular-related changes,” said Dr. Snyder, who was not involved with the current study.
However, “additional studies that look both in model systems and in humans are needed to further illuminate these relationships,” Dr. Snyder said.
The study was supported by grants from the National Institutes of Health as well as the Robertson Therapeutic Development Fund, Samuel Newhouse Foundation, John A. Herrmann, and the May and Samuel Rudin Family Foundation. Dr. Norris, Dr. Strickland, and Dr. Snyder declared no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM PROCEEDINGS OF THE NATIONAL ACADEMY OF SCIENCE
New evidence early treatment improves preclinical MS outcomes
TOPLINE:
new research shows.
METHODOLOGY:
Early use of DMTs is typically recommended for patients with established MS, but mounting evidence, including the ARISE trial, which assessed Tecfidera, suggests these agents benefit patients with RIS, the earliest detectable preclinical MS stage.
The new study, known as Teriflunomide in Radiologically Isolated Syndrome (TERIS), included 89 adult patients with RIS (mean age, 37.8 years) from centers in France, Switzerland, and Turkey. Participants were randomly assigned to receive placebo or teriflunomide 14 mg daily. Teriflunomide is an oral immunomodulator approved for treating relapsing remitting MS.
Investigators performed MRI at baseline and at weeks 48, 96, and 144 and at any time during the study if warranted.
Researchers adjusted for potential confounders, including sex, age at RIS diagnosis, MS family history, brain T2-weighted hyperintense lesion volume, and presence of Gd+/− lesions.
The primary outcome was time to a first acute or progressive neurologic event resulting from central nervous system demyelination, expressed as a rate of conversion to clinical MS.
TAKEAWAY:
Eighteen participants – nine in each group – discontinued the study, resulting in a dropout rate of 20%.
The risk of a first clinical event was significantly reduced in the teriflunomide arm (mean time to event, 128.2 weeks) with 8 clinical events (6 acute, 2 progressive) in comparison with the placebo arm (mean time to event, 109.6 weeks) with 20 clinical events (18 acute, 2 progressive) and an adjusted hazard ratio of 0.28 (95% CI, 0.11-0.71; P = .007).
All secondary MRI measures, including the cumulative number of new and/or newly enlarging T2 lesions and the cumulative number of Gd+ lesions, did not reach statistical significance, although these were numerically lower in the teriflunomide arm, possibly because participants with early events switched to the treatment arm.
The most common adverse events that occurred more often in patients treated with teriflunomide were gastrointestinal disorders (11.4%), dysmenorrhea (9.1%), benign respiratory infections (6.8%), general disorders/conditions (6.8%), and transient increase of transaminases (4.5%).
IN PRACTICE:
“These results suggest that for the first time, we may have an opportunity to better identify those at risk for a primary progressive clinical course at this preclinical stage and prevent or delay clinical progression from the onset, which is a clear unmet need in MS clinical practice,” wrote the authors.
SOURCE:
The study was carried out by Christine Lebrun-Frénay MD, PhD, head of the inflammatory neurological disorders clinical research unit and MS center at the University of Nice (France). It was published online in JAMA Neurology.
LIMITATIONS:
The investigators could not stratify at-risk subgroups according to risk factors for developing MS, mainly because of power issues. The study was prematurely discontinued by its financial sponsor (Sanofi), owing primarily to slow enrollment that resulted from national regulations on activating recruitment sites and the impact of the COVID-19 pandemic. Another challenge for the study was that some individuals with RIS had already been exposed to a DMT or hesitated to participate in a clinical trial. The financial sponsor, which provided the study drug and placebo tablets, terminated their availability, given the anticipated release of generic teriflunomide.
DISCLOSURES:
The study was supported by Sanofi, the University Hospital of Nice, University Cote d’Azur, and the Radiologically Isolated Syndrome Consortium. Lebrun-Frénay has no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
TOPLINE:
new research shows.
METHODOLOGY:
Early use of DMTs is typically recommended for patients with established MS, but mounting evidence, including the ARISE trial, which assessed Tecfidera, suggests these agents benefit patients with RIS, the earliest detectable preclinical MS stage.
The new study, known as Teriflunomide in Radiologically Isolated Syndrome (TERIS), included 89 adult patients with RIS (mean age, 37.8 years) from centers in France, Switzerland, and Turkey. Participants were randomly assigned to receive placebo or teriflunomide 14 mg daily. Teriflunomide is an oral immunomodulator approved for treating relapsing remitting MS.
Investigators performed MRI at baseline and at weeks 48, 96, and 144 and at any time during the study if warranted.
Researchers adjusted for potential confounders, including sex, age at RIS diagnosis, MS family history, brain T2-weighted hyperintense lesion volume, and presence of Gd+/− lesions.
The primary outcome was time to a first acute or progressive neurologic event resulting from central nervous system demyelination, expressed as a rate of conversion to clinical MS.
TAKEAWAY:
Eighteen participants – nine in each group – discontinued the study, resulting in a dropout rate of 20%.
The risk of a first clinical event was significantly reduced in the teriflunomide arm (mean time to event, 128.2 weeks) with 8 clinical events (6 acute, 2 progressive) in comparison with the placebo arm (mean time to event, 109.6 weeks) with 20 clinical events (18 acute, 2 progressive) and an adjusted hazard ratio of 0.28 (95% CI, 0.11-0.71; P = .007).
All secondary MRI measures, including the cumulative number of new and/or newly enlarging T2 lesions and the cumulative number of Gd+ lesions, did not reach statistical significance, although these were numerically lower in the teriflunomide arm, possibly because participants with early events switched to the treatment arm.
The most common adverse events that occurred more often in patients treated with teriflunomide were gastrointestinal disorders (11.4%), dysmenorrhea (9.1%), benign respiratory infections (6.8%), general disorders/conditions (6.8%), and transient increase of transaminases (4.5%).
IN PRACTICE:
“These results suggest that for the first time, we may have an opportunity to better identify those at risk for a primary progressive clinical course at this preclinical stage and prevent or delay clinical progression from the onset, which is a clear unmet need in MS clinical practice,” wrote the authors.
SOURCE:
The study was carried out by Christine Lebrun-Frénay MD, PhD, head of the inflammatory neurological disorders clinical research unit and MS center at the University of Nice (France). It was published online in JAMA Neurology.
LIMITATIONS:
The investigators could not stratify at-risk subgroups according to risk factors for developing MS, mainly because of power issues. The study was prematurely discontinued by its financial sponsor (Sanofi), owing primarily to slow enrollment that resulted from national regulations on activating recruitment sites and the impact of the COVID-19 pandemic. Another challenge for the study was that some individuals with RIS had already been exposed to a DMT or hesitated to participate in a clinical trial. The financial sponsor, which provided the study drug and placebo tablets, terminated their availability, given the anticipated release of generic teriflunomide.
DISCLOSURES:
The study was supported by Sanofi, the University Hospital of Nice, University Cote d’Azur, and the Radiologically Isolated Syndrome Consortium. Lebrun-Frénay has no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
TOPLINE:
new research shows.
METHODOLOGY:
Early use of DMTs is typically recommended for patients with established MS, but mounting evidence, including the ARISE trial, which assessed Tecfidera, suggests these agents benefit patients with RIS, the earliest detectable preclinical MS stage.
The new study, known as Teriflunomide in Radiologically Isolated Syndrome (TERIS), included 89 adult patients with RIS (mean age, 37.8 years) from centers in France, Switzerland, and Turkey. Participants were randomly assigned to receive placebo or teriflunomide 14 mg daily. Teriflunomide is an oral immunomodulator approved for treating relapsing remitting MS.
Investigators performed MRI at baseline and at weeks 48, 96, and 144 and at any time during the study if warranted.
Researchers adjusted for potential confounders, including sex, age at RIS diagnosis, MS family history, brain T2-weighted hyperintense lesion volume, and presence of Gd+/− lesions.
The primary outcome was time to a first acute or progressive neurologic event resulting from central nervous system demyelination, expressed as a rate of conversion to clinical MS.
TAKEAWAY:
Eighteen participants – nine in each group – discontinued the study, resulting in a dropout rate of 20%.
The risk of a first clinical event was significantly reduced in the teriflunomide arm (mean time to event, 128.2 weeks) with 8 clinical events (6 acute, 2 progressive) in comparison with the placebo arm (mean time to event, 109.6 weeks) with 20 clinical events (18 acute, 2 progressive) and an adjusted hazard ratio of 0.28 (95% CI, 0.11-0.71; P = .007).
All secondary MRI measures, including the cumulative number of new and/or newly enlarging T2 lesions and the cumulative number of Gd+ lesions, did not reach statistical significance, although these were numerically lower in the teriflunomide arm, possibly because participants with early events switched to the treatment arm.
The most common adverse events that occurred more often in patients treated with teriflunomide were gastrointestinal disorders (11.4%), dysmenorrhea (9.1%), benign respiratory infections (6.8%), general disorders/conditions (6.8%), and transient increase of transaminases (4.5%).
IN PRACTICE:
“These results suggest that for the first time, we may have an opportunity to better identify those at risk for a primary progressive clinical course at this preclinical stage and prevent or delay clinical progression from the onset, which is a clear unmet need in MS clinical practice,” wrote the authors.
SOURCE:
The study was carried out by Christine Lebrun-Frénay MD, PhD, head of the inflammatory neurological disorders clinical research unit and MS center at the University of Nice (France). It was published online in JAMA Neurology.
LIMITATIONS:
The investigators could not stratify at-risk subgroups according to risk factors for developing MS, mainly because of power issues. The study was prematurely discontinued by its financial sponsor (Sanofi), owing primarily to slow enrollment that resulted from national regulations on activating recruitment sites and the impact of the COVID-19 pandemic. Another challenge for the study was that some individuals with RIS had already been exposed to a DMT or hesitated to participate in a clinical trial. The financial sponsor, which provided the study drug and placebo tablets, terminated their availability, given the anticipated release of generic teriflunomide.
DISCLOSURES:
The study was supported by Sanofi, the University Hospital of Nice, University Cote d’Azur, and the Radiologically Isolated Syndrome Consortium. Lebrun-Frénay has no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
FROM JAMA NEUROLOGY
Blood transfusions linked to intracerebral hemorrhage risk
In an exploratory analysis, patients receiving red blood cell transfusions from donors who later developed multiple spontaneous ICHs, and were assumed to have CAA, were at a significantly increased risk of developing spontaneous ICH themselves.
“This may suggest a transfusion-transmissible agent associated with some types of spontaneous ICH, although the findings may be susceptible to selection bias and residual confounding, and further research is needed to investigate if transfusion transmission of CAA might explain this association,” the investigators noted.
“We do not think that the findings motivate a change in practice, and we should not let these results discourage otherwise indicated blood transfusion,” said lead author Jingcheng Zhao, MD, PhD, with Karolinska University Hospital Solna, Stockholm.
The study was published online in the Journal of the American Medical Association.
Novel finding
Recent evidence suggests that CAA exhibits “prion-like” transmissivity, with reports of transmission through cadaveric pituitary hormone contaminated with amyloid-beta and tau protein, dura mater grafts, and possibly neurosurgical instruments.
CAA, which is characterized by the deposition of amyloid protein in the brain, is the second most common cause of spontaneous ICH.
The researchers hypothesized that transfusion transmission of CAA may manifest through an increased risk for spontaneous ICH among transfusion recipients given blood from a donor with spontaneous ICH. To explore this hypothesis, they analyzed national registry data from Sweden and Denmark for ICH in recipients of red blood cell transfusion from donors who themselves had ICH over the years after their blood donations, with the assumption that donors with two or more ICHs would likely have CAA.
The cohort included nearly 760,000 individuals in Sweden (median age, 65 years; 59% women) and 330,000 in Denmark (median age, 64 years; 58% women), with a median follow-up of 5.8 and 6.1 years, respectively.
Receiving red blood cell transfusions from donors who later developed multiple spontaneous ICHs was associated with a greater than twofold increased risk of developing spontaneous ICH, compared with receiving a transfusion from donors without subsequent ICH (hazard ratio, 2.73; P < .001 in the Swedish cohort and HR, 2.32; P = .04 in the Danish cohort).
“The observed increased risk of spontaneous ICH associated with receiving a red blood cell transfusion from a donor who later developed multiple spontaneous ICHs, corresponding to a 30-year cumulative incidence difference of 2.3%, is a novel finding,” the researchers wrote.
There was no increase in post-transfusion ICH risk among recipients whose donors had a single post–blood-donation ICH.
The findings were robust to several of the sensitivity analyses.
A “negative” control analysis of post-transfusion ischemic stroke (instead of ICH) found no increased risk among recipients of blood from donors who had single or multiple ICHs.
This study provides “exploratory evidence of possible transfusion-transmission of a factor that causes ICHs, but more research is needed to confirm and to understand the mechanism,” said Dr. Zhao.
The researchers noted that they did not directly assess CAA but expect it would be more common among donors who develop multiple spontaneous ICHs, “as CAA-related ICH has been reported to have a 7-fold increase for recurrent ICHs, compared with non–CAA-related ICH.”
Worrisome finding or false alarm?
In an accompanying editorial, Steven Greenberg, MD, PhD, with the department of neurology, Harvard Medical School, Boston, said there are “good reasons to treat the possibility of CAA transmission via blood transfusion seriously – and good reasons to remain skeptical, at least for the present.”
“Powerful” arguments in support of the findings include the robust study methodology and the “striking” similarity in results from the two registries, which argues against a chance finding. Another is the negative control with ischemic stroke as the outcome, which argues against unsuspected confounding-causing associations with all types of stroke, Dr. Greenberg noted.
Arguments for remaining “unconvinced” of the association center on the weakness of evidence for a plausible biological mechanism for the finding, he points out. Another is the short-time course of ICHs after blood transfusion, which is “quite challenging to explain,” Dr. Greenberg said. Nearly half of the ICHs among blood recipients occurred within 5 years of transfusion, which is “dramatically” faster than the 30- to 40-year interval reported between neurosurgical exposure to cadaveric tissue and first ICH, he added.
Another related “mechanistic reservation” is the plausibility that a transmissible species of amyloid-beta could travel from blood to brain in sufficient quantities to trigger advanced CAA or Alzheimer disease pathology, he wrote.
He added the current study leaves him “squarely at the corner of anxiety and skepticism.”
With more than 10 million units of blood transfused in the United States each year, even a modest increase in risk for future brain hemorrhages or dementia conferred by “an uncommon – but as of now undetectable – donor trait would represent a substantial public health concern,” Dr. Greenberg wrote.
“From the standpoint of scientific plausibility, however, even this well-conducted analysis is at risk of representing a false alarm,” he cautioned.
Looking ahead, Dr. Greenberg said one clear direction is independent replication, ideally with datasets in which donor and recipient dementia can be reliably ascertained to assess the possibility of Alzheimer’s disease as well as CAA transmissibility.
“The other challenge is for experimental biologists to consider the alternative possibility of transfusion-related acceleration of downstream steps in the CAA-ICH pathway, such as the vessel remodeling by which amyloid beta–laden vessels proceed to rupture and bleed.”
“The current study is not yet a reason for alarm, certainly not a reason to avoid otherwise indicated blood transfusion, but it is a strong call for more scientific digging,” Dr. Greenberg concluded.
The study was funded by grants from the Karolinska Institute, the Swedish Research Council, and Region Stockholm. Dr. Zhao and Dr. Greenberg report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In an exploratory analysis, patients receiving red blood cell transfusions from donors who later developed multiple spontaneous ICHs, and were assumed to have CAA, were at a significantly increased risk of developing spontaneous ICH themselves.
“This may suggest a transfusion-transmissible agent associated with some types of spontaneous ICH, although the findings may be susceptible to selection bias and residual confounding, and further research is needed to investigate if transfusion transmission of CAA might explain this association,” the investigators noted.
“We do not think that the findings motivate a change in practice, and we should not let these results discourage otherwise indicated blood transfusion,” said lead author Jingcheng Zhao, MD, PhD, with Karolinska University Hospital Solna, Stockholm.
The study was published online in the Journal of the American Medical Association.
Novel finding
Recent evidence suggests that CAA exhibits “prion-like” transmissivity, with reports of transmission through cadaveric pituitary hormone contaminated with amyloid-beta and tau protein, dura mater grafts, and possibly neurosurgical instruments.
CAA, which is characterized by the deposition of amyloid protein in the brain, is the second most common cause of spontaneous ICH.
The researchers hypothesized that transfusion transmission of CAA may manifest through an increased risk for spontaneous ICH among transfusion recipients given blood from a donor with spontaneous ICH. To explore this hypothesis, they analyzed national registry data from Sweden and Denmark for ICH in recipients of red blood cell transfusion from donors who themselves had ICH over the years after their blood donations, with the assumption that donors with two or more ICHs would likely have CAA.
The cohort included nearly 760,000 individuals in Sweden (median age, 65 years; 59% women) and 330,000 in Denmark (median age, 64 years; 58% women), with a median follow-up of 5.8 and 6.1 years, respectively.
Receiving red blood cell transfusions from donors who later developed multiple spontaneous ICHs was associated with a greater than twofold increased risk of developing spontaneous ICH, compared with receiving a transfusion from donors without subsequent ICH (hazard ratio, 2.73; P < .001 in the Swedish cohort and HR, 2.32; P = .04 in the Danish cohort).
“The observed increased risk of spontaneous ICH associated with receiving a red blood cell transfusion from a donor who later developed multiple spontaneous ICHs, corresponding to a 30-year cumulative incidence difference of 2.3%, is a novel finding,” the researchers wrote.
There was no increase in post-transfusion ICH risk among recipients whose donors had a single post–blood-donation ICH.
The findings were robust to several of the sensitivity analyses.
A “negative” control analysis of post-transfusion ischemic stroke (instead of ICH) found no increased risk among recipients of blood from donors who had single or multiple ICHs.
This study provides “exploratory evidence of possible transfusion-transmission of a factor that causes ICHs, but more research is needed to confirm and to understand the mechanism,” said Dr. Zhao.
The researchers noted that they did not directly assess CAA but expect it would be more common among donors who develop multiple spontaneous ICHs, “as CAA-related ICH has been reported to have a 7-fold increase for recurrent ICHs, compared with non–CAA-related ICH.”
Worrisome finding or false alarm?
In an accompanying editorial, Steven Greenberg, MD, PhD, with the department of neurology, Harvard Medical School, Boston, said there are “good reasons to treat the possibility of CAA transmission via blood transfusion seriously – and good reasons to remain skeptical, at least for the present.”
“Powerful” arguments in support of the findings include the robust study methodology and the “striking” similarity in results from the two registries, which argues against a chance finding. Another is the negative control with ischemic stroke as the outcome, which argues against unsuspected confounding-causing associations with all types of stroke, Dr. Greenberg noted.
Arguments for remaining “unconvinced” of the association center on the weakness of evidence for a plausible biological mechanism for the finding, he points out. Another is the short-time course of ICHs after blood transfusion, which is “quite challenging to explain,” Dr. Greenberg said. Nearly half of the ICHs among blood recipients occurred within 5 years of transfusion, which is “dramatically” faster than the 30- to 40-year interval reported between neurosurgical exposure to cadaveric tissue and first ICH, he added.
Another related “mechanistic reservation” is the plausibility that a transmissible species of amyloid-beta could travel from blood to brain in sufficient quantities to trigger advanced CAA or Alzheimer disease pathology, he wrote.
He added the current study leaves him “squarely at the corner of anxiety and skepticism.”
With more than 10 million units of blood transfused in the United States each year, even a modest increase in risk for future brain hemorrhages or dementia conferred by “an uncommon – but as of now undetectable – donor trait would represent a substantial public health concern,” Dr. Greenberg wrote.
“From the standpoint of scientific plausibility, however, even this well-conducted analysis is at risk of representing a false alarm,” he cautioned.
Looking ahead, Dr. Greenberg said one clear direction is independent replication, ideally with datasets in which donor and recipient dementia can be reliably ascertained to assess the possibility of Alzheimer’s disease as well as CAA transmissibility.
“The other challenge is for experimental biologists to consider the alternative possibility of transfusion-related acceleration of downstream steps in the CAA-ICH pathway, such as the vessel remodeling by which amyloid beta–laden vessels proceed to rupture and bleed.”
“The current study is not yet a reason for alarm, certainly not a reason to avoid otherwise indicated blood transfusion, but it is a strong call for more scientific digging,” Dr. Greenberg concluded.
The study was funded by grants from the Karolinska Institute, the Swedish Research Council, and Region Stockholm. Dr. Zhao and Dr. Greenberg report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In an exploratory analysis, patients receiving red blood cell transfusions from donors who later developed multiple spontaneous ICHs, and were assumed to have CAA, were at a significantly increased risk of developing spontaneous ICH themselves.
“This may suggest a transfusion-transmissible agent associated with some types of spontaneous ICH, although the findings may be susceptible to selection bias and residual confounding, and further research is needed to investigate if transfusion transmission of CAA might explain this association,” the investigators noted.
“We do not think that the findings motivate a change in practice, and we should not let these results discourage otherwise indicated blood transfusion,” said lead author Jingcheng Zhao, MD, PhD, with Karolinska University Hospital Solna, Stockholm.
The study was published online in the Journal of the American Medical Association.
Novel finding
Recent evidence suggests that CAA exhibits “prion-like” transmissivity, with reports of transmission through cadaveric pituitary hormone contaminated with amyloid-beta and tau protein, dura mater grafts, and possibly neurosurgical instruments.
CAA, which is characterized by the deposition of amyloid protein in the brain, is the second most common cause of spontaneous ICH.
The researchers hypothesized that transfusion transmission of CAA may manifest through an increased risk for spontaneous ICH among transfusion recipients given blood from a donor with spontaneous ICH. To explore this hypothesis, they analyzed national registry data from Sweden and Denmark for ICH in recipients of red blood cell transfusion from donors who themselves had ICH over the years after their blood donations, with the assumption that donors with two or more ICHs would likely have CAA.
The cohort included nearly 760,000 individuals in Sweden (median age, 65 years; 59% women) and 330,000 in Denmark (median age, 64 years; 58% women), with a median follow-up of 5.8 and 6.1 years, respectively.
Receiving red blood cell transfusions from donors who later developed multiple spontaneous ICHs was associated with a greater than twofold increased risk of developing spontaneous ICH, compared with receiving a transfusion from donors without subsequent ICH (hazard ratio, 2.73; P < .001 in the Swedish cohort and HR, 2.32; P = .04 in the Danish cohort).
“The observed increased risk of spontaneous ICH associated with receiving a red blood cell transfusion from a donor who later developed multiple spontaneous ICHs, corresponding to a 30-year cumulative incidence difference of 2.3%, is a novel finding,” the researchers wrote.
There was no increase in post-transfusion ICH risk among recipients whose donors had a single post–blood-donation ICH.
The findings were robust to several of the sensitivity analyses.
A “negative” control analysis of post-transfusion ischemic stroke (instead of ICH) found no increased risk among recipients of blood from donors who had single or multiple ICHs.
This study provides “exploratory evidence of possible transfusion-transmission of a factor that causes ICHs, but more research is needed to confirm and to understand the mechanism,” said Dr. Zhao.
The researchers noted that they did not directly assess CAA but expect it would be more common among donors who develop multiple spontaneous ICHs, “as CAA-related ICH has been reported to have a 7-fold increase for recurrent ICHs, compared with non–CAA-related ICH.”
Worrisome finding or false alarm?
In an accompanying editorial, Steven Greenberg, MD, PhD, with the department of neurology, Harvard Medical School, Boston, said there are “good reasons to treat the possibility of CAA transmission via blood transfusion seriously – and good reasons to remain skeptical, at least for the present.”
“Powerful” arguments in support of the findings include the robust study methodology and the “striking” similarity in results from the two registries, which argues against a chance finding. Another is the negative control with ischemic stroke as the outcome, which argues against unsuspected confounding-causing associations with all types of stroke, Dr. Greenberg noted.
Arguments for remaining “unconvinced” of the association center on the weakness of evidence for a plausible biological mechanism for the finding, he points out. Another is the short-time course of ICHs after blood transfusion, which is “quite challenging to explain,” Dr. Greenberg said. Nearly half of the ICHs among blood recipients occurred within 5 years of transfusion, which is “dramatically” faster than the 30- to 40-year interval reported between neurosurgical exposure to cadaveric tissue and first ICH, he added.
Another related “mechanistic reservation” is the plausibility that a transmissible species of amyloid-beta could travel from blood to brain in sufficient quantities to trigger advanced CAA or Alzheimer disease pathology, he wrote.
He added the current study leaves him “squarely at the corner of anxiety and skepticism.”
With more than 10 million units of blood transfused in the United States each year, even a modest increase in risk for future brain hemorrhages or dementia conferred by “an uncommon – but as of now undetectable – donor trait would represent a substantial public health concern,” Dr. Greenberg wrote.
“From the standpoint of scientific plausibility, however, even this well-conducted analysis is at risk of representing a false alarm,” he cautioned.
Looking ahead, Dr. Greenberg said one clear direction is independent replication, ideally with datasets in which donor and recipient dementia can be reliably ascertained to assess the possibility of Alzheimer’s disease as well as CAA transmissibility.
“The other challenge is for experimental biologists to consider the alternative possibility of transfusion-related acceleration of downstream steps in the CAA-ICH pathway, such as the vessel remodeling by which amyloid beta–laden vessels proceed to rupture and bleed.”
“The current study is not yet a reason for alarm, certainly not a reason to avoid otherwise indicated blood transfusion, but it is a strong call for more scientific digging,” Dr. Greenberg concluded.
The study was funded by grants from the Karolinska Institute, the Swedish Research Council, and Region Stockholm. Dr. Zhao and Dr. Greenberg report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
From JAMA
Artificial intelligence in your office
It is difficult to go through any publication or website these days without finding an article about artificial intelligence (AI). Many discuss its current status, while others speculate on potential future applications. Often, AI is described as an “existential threat to human health” by commentators who aren’t even aware of the definition of that term as Kierkegaard conceived it, the role of the individual to breathe meaning into life. Others characterize such cataclysmic predictions as “overblown and misdirected”.
The long-term potential for abuse of AI requires discussion, and should be addressed by policy makers, but that is beyond the scope of this column.
Meanwhile, with no “existential” threat to anybody.
The most popular current AI-based medical applications are automated scribes. They transcribe live consultations between physician and patient automatically and create a searchable report, plus notes for charts and billing.
I’ve written about AI scribes before, but the quality and user-friendliness of these products have improved dramatically in recent years. Language processing capabilities now permit you to speak naturally, without having to memorize specific commands. Some scribes can mimic your writing style based on sample notes that you enter into the system. Others allow you to integrate your own knowledge base, or a bibliography of research studies. With some systems, you can dictate notes directly into most EHR software, ask questions regarding medication dosages, or access a patient’s medical history from hospitals or other offices.
Current popular medical scribe products include DeepCura, DeepScribe, Nuance, Suki, Augmedix, Tali AI, Iodine Software, and ScribeLink. Amazon Web Services recently launched its own product, HealthScribe, as well. (As always, I have no financial interest in any product or service mentioned in this column.)
AI scribes aren’t entirely autonomous, of course; you need to read the output and check for potential inaccuracies. Still, users claim that they substantially reduce documentation and charting time, permitting more patient visits and less after-hours work.
AI can also be used to provide useful content for your patients. If you are not particularly good at writing, or don’t have the time for it, generative algorithms like the much-vaunted ChatGPT can generate posts, FAQs, and other informational content for your website, blog, or social media pages. You can ask for ideas about timely health topics and write general information articles, or create content specific to your location or specialty. You can use it to write emails informing your patients about upcoming office events or educate them on a range of topics, from getting their annual flu shots to scheduling regular screening skin exams.
With some of the same techniques and additional software, you can create entire videos for your website at a fraction of the cost of hiring a video production team. After using ChatGPT to write the content – for example, a 5-minute script on the importance of sunscreen in preventing skin cancer – you can employ a text-to-speech algorithm such as Revoicer to transform the script into audio content, and then a preproduction algorithm like Yepic or Synthesia to generate a video with a synthetic human.
If you are unhappy with your current online presence, you can use AI to create an entire website. Through a series of questions, AI website builders such as Wix ADI, Jimdo, Hostinger, and 10Web gather all the information needed to set up a website draft that is already personalized with medical-specific content. Most offer the option to connect to Instagram, Facebook, Google My Business, and similar sites, to which they can import your office’s logo, images, and descriptive texts.
Some of them are capable of pulling up responsive site pages that automatically adjust to the device – mobile or computer – that the visitor is using. This is important, as I’ve written before, because more than half of all searches for doctors are now made on smartphones, so the more “mobile friendly” your site is, the higher it will be ranked. You can test how easily a visitor can use your website on a mobile device with Google’s free Mobile-Friendly Test.
If you give talks at medical meetings, you know how cumbersome and time-consuming it can be to create Powerpoint presentations. Once again, AI can save you time and trouble. Presentation designers such as Presentations.AI, Deck Robot, iA Presenter, and Beautiful.AI can assemble very acceptable presentations from your primary inputs. You typically choose a template, input your basic data, and AI will format the slides and offer you visuals, animations, voice-overs, and other fancy features. You will also have flexibility in changing segments or images or sizes you don’t like.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News. Write to him at [email protected].
It is difficult to go through any publication or website these days without finding an article about artificial intelligence (AI). Many discuss its current status, while others speculate on potential future applications. Often, AI is described as an “existential threat to human health” by commentators who aren’t even aware of the definition of that term as Kierkegaard conceived it, the role of the individual to breathe meaning into life. Others characterize such cataclysmic predictions as “overblown and misdirected”.
The long-term potential for abuse of AI requires discussion, and should be addressed by policy makers, but that is beyond the scope of this column.
Meanwhile, with no “existential” threat to anybody.
The most popular current AI-based medical applications are automated scribes. They transcribe live consultations between physician and patient automatically and create a searchable report, plus notes for charts and billing.
I’ve written about AI scribes before, but the quality and user-friendliness of these products have improved dramatically in recent years. Language processing capabilities now permit you to speak naturally, without having to memorize specific commands. Some scribes can mimic your writing style based on sample notes that you enter into the system. Others allow you to integrate your own knowledge base, or a bibliography of research studies. With some systems, you can dictate notes directly into most EHR software, ask questions regarding medication dosages, or access a patient’s medical history from hospitals or other offices.
Current popular medical scribe products include DeepCura, DeepScribe, Nuance, Suki, Augmedix, Tali AI, Iodine Software, and ScribeLink. Amazon Web Services recently launched its own product, HealthScribe, as well. (As always, I have no financial interest in any product or service mentioned in this column.)
AI scribes aren’t entirely autonomous, of course; you need to read the output and check for potential inaccuracies. Still, users claim that they substantially reduce documentation and charting time, permitting more patient visits and less after-hours work.
AI can also be used to provide useful content for your patients. If you are not particularly good at writing, or don’t have the time for it, generative algorithms like the much-vaunted ChatGPT can generate posts, FAQs, and other informational content for your website, blog, or social media pages. You can ask for ideas about timely health topics and write general information articles, or create content specific to your location or specialty. You can use it to write emails informing your patients about upcoming office events or educate them on a range of topics, from getting their annual flu shots to scheduling regular screening skin exams.
With some of the same techniques and additional software, you can create entire videos for your website at a fraction of the cost of hiring a video production team. After using ChatGPT to write the content – for example, a 5-minute script on the importance of sunscreen in preventing skin cancer – you can employ a text-to-speech algorithm such as Revoicer to transform the script into audio content, and then a preproduction algorithm like Yepic or Synthesia to generate a video with a synthetic human.
If you are unhappy with your current online presence, you can use AI to create an entire website. Through a series of questions, AI website builders such as Wix ADI, Jimdo, Hostinger, and 10Web gather all the information needed to set up a website draft that is already personalized with medical-specific content. Most offer the option to connect to Instagram, Facebook, Google My Business, and similar sites, to which they can import your office’s logo, images, and descriptive texts.
Some of them are capable of pulling up responsive site pages that automatically adjust to the device – mobile or computer – that the visitor is using. This is important, as I’ve written before, because more than half of all searches for doctors are now made on smartphones, so the more “mobile friendly” your site is, the higher it will be ranked. You can test how easily a visitor can use your website on a mobile device with Google’s free Mobile-Friendly Test.
If you give talks at medical meetings, you know how cumbersome and time-consuming it can be to create Powerpoint presentations. Once again, AI can save you time and trouble. Presentation designers such as Presentations.AI, Deck Robot, iA Presenter, and Beautiful.AI can assemble very acceptable presentations from your primary inputs. You typically choose a template, input your basic data, and AI will format the slides and offer you visuals, animations, voice-overs, and other fancy features. You will also have flexibility in changing segments or images or sizes you don’t like.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News. Write to him at [email protected].
It is difficult to go through any publication or website these days without finding an article about artificial intelligence (AI). Many discuss its current status, while others speculate on potential future applications. Often, AI is described as an “existential threat to human health” by commentators who aren’t even aware of the definition of that term as Kierkegaard conceived it, the role of the individual to breathe meaning into life. Others characterize such cataclysmic predictions as “overblown and misdirected”.
The long-term potential for abuse of AI requires discussion, and should be addressed by policy makers, but that is beyond the scope of this column.
Meanwhile, with no “existential” threat to anybody.
The most popular current AI-based medical applications are automated scribes. They transcribe live consultations between physician and patient automatically and create a searchable report, plus notes for charts and billing.
I’ve written about AI scribes before, but the quality and user-friendliness of these products have improved dramatically in recent years. Language processing capabilities now permit you to speak naturally, without having to memorize specific commands. Some scribes can mimic your writing style based on sample notes that you enter into the system. Others allow you to integrate your own knowledge base, or a bibliography of research studies. With some systems, you can dictate notes directly into most EHR software, ask questions regarding medication dosages, or access a patient’s medical history from hospitals or other offices.
Current popular medical scribe products include DeepCura, DeepScribe, Nuance, Suki, Augmedix, Tali AI, Iodine Software, and ScribeLink. Amazon Web Services recently launched its own product, HealthScribe, as well. (As always, I have no financial interest in any product or service mentioned in this column.)
AI scribes aren’t entirely autonomous, of course; you need to read the output and check for potential inaccuracies. Still, users claim that they substantially reduce documentation and charting time, permitting more patient visits and less after-hours work.
AI can also be used to provide useful content for your patients. If you are not particularly good at writing, or don’t have the time for it, generative algorithms like the much-vaunted ChatGPT can generate posts, FAQs, and other informational content for your website, blog, or social media pages. You can ask for ideas about timely health topics and write general information articles, or create content specific to your location or specialty. You can use it to write emails informing your patients about upcoming office events or educate them on a range of topics, from getting their annual flu shots to scheduling regular screening skin exams.
With some of the same techniques and additional software, you can create entire videos for your website at a fraction of the cost of hiring a video production team. After using ChatGPT to write the content – for example, a 5-minute script on the importance of sunscreen in preventing skin cancer – you can employ a text-to-speech algorithm such as Revoicer to transform the script into audio content, and then a preproduction algorithm like Yepic or Synthesia to generate a video with a synthetic human.
If you are unhappy with your current online presence, you can use AI to create an entire website. Through a series of questions, AI website builders such as Wix ADI, Jimdo, Hostinger, and 10Web gather all the information needed to set up a website draft that is already personalized with medical-specific content. Most offer the option to connect to Instagram, Facebook, Google My Business, and similar sites, to which they can import your office’s logo, images, and descriptive texts.
Some of them are capable of pulling up responsive site pages that automatically adjust to the device – mobile or computer – that the visitor is using. This is important, as I’ve written before, because more than half of all searches for doctors are now made on smartphones, so the more “mobile friendly” your site is, the higher it will be ranked. You can test how easily a visitor can use your website on a mobile device with Google’s free Mobile-Friendly Test.
If you give talks at medical meetings, you know how cumbersome and time-consuming it can be to create Powerpoint presentations. Once again, AI can save you time and trouble. Presentation designers such as Presentations.AI, Deck Robot, iA Presenter, and Beautiful.AI can assemble very acceptable presentations from your primary inputs. You typically choose a template, input your basic data, and AI will format the slides and offer you visuals, animations, voice-overs, and other fancy features. You will also have flexibility in changing segments or images or sizes you don’t like.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News. Write to him at [email protected].
Your workplace is toxic: Can you make it better?
A physician in your office is hot-tempered, critical, and upsets both the physicians and staff. Two of your partners are arguing over a software vendor and refuse to compromise. One doctor’s spouse is the office manager and snipes at everyone; the lead partner micromanages and second-guesses other doctors’ treatment plans, and no one will stand up to her.
If your practice has similar scenarios, you’re likely dealing with your own anger, irritation, and dread at work. You’re struggling with a toxic practice atmosphere, and you must make changes – fast.
However, this isn’t easy, given that what goes on in a doctor’s office is “high consequence,” says Leonard J. Marcus, PhD, founding director of the program for health care negotiation and conflict resolution at the Harvard School of Public Health in Boston.
The two things that tend to plague medical practices most: A culture of fear and someone who is letting ego run the day-to-day, he says.
“Fear overwhelms any chance for good morale among colleagues,” says Dr. Marcus, who is also the coauthor of “Renegotiating Health Care: Resolving Conflict to Build Collaboration.” “In a work environment where the fear is overwhelming, the ego can take over, and someone at the practice becomes overly concerned about getting credit, taking control, ordering other people around, and deciding who is on top and who is on the bottom.”
Tension, stress, back-biting, and rudeness are also symptoms of a more significant problem, says Jes Montgomery, MD, a psychiatrist and medical director of APN Dallas, a mental health–focused practice.
“If you don’t get toxicity under control, it will blow the office apart,” Dr. Montgomery says.
1. Recognize the signs
Part of the problem with a toxic medical practice is that, culturally, we don’t treat mental health and burnout as real illnesses. “A physician who is depressed is not going to be melancholy or bursting into tears with patients,” Dr. Montgomery says. “They’ll get behind on paperwork, skip meals, or find that it’s difficult to sleep at night. Next, they’ll yell at the partners and staff, always be in a foul mood, and gripe about inconsequential things. Their behavior affects everyone.”
Dr. Montgomery says that physicians aren’t taught to ask for help, making it difficult to see what’s really going on when someone displays toxic behavior in the practice. If it’s a partner, take time to ask what’s going on. If it’s yourself, step back and see if you can ask someone for the help you need.
2. Have difficult conversations
This is tough for most of us, says Jeremy Pollack, PhD, CEO and founder of Pollack Peacebuilding Systems, a conflict resolution consulting firm. If a team member is hot-tempered, disrespectful, or talking to patients in an unproductive manner, see if you can have an effective conversation with that person. The tricky part is critiquing in a way that doesn’t make them feel defensive – and wanting to push back.
For a micromanaging office manager, for example, you could say something like,”You’re doing a great job with the inventory, but I need you to let the staff have some autonomy and not hover over every supply they use in the break room, so that people won’t feel resentful toward us.” Make it clear you’re a team, and this is a team challenge. “However, if a doctor feels like they’ve tried to communicate to that colleague and are still walking on eggshells, it’s time to try to get help from someone – perhaps a practice management organization,” says Dr. Pollack.
3. Open lines of communication
It’s critical to create a comfortable space to speak with your colleagues, says Marisa Garshick, MD, a dermatologist in private practice in New York. “Creating an environment where there is an open line of communication, whether it’s directly to somebody in charge or having a system where you can give feedback more privately or anonymously, is important so that tension doesn’t build.”
“Being a doctor is a social enterprise,” Dr. Marcus says. “The science of medicine is critically important, but patients and the other health care workers on your team are also critically important. In the long run, the most successful physicians pay attention to both. It’s a full package.”
4. Emphasize the positive
Instead of discussing things only when they go wrong, try optimism, Dr. Garshick said. When positive things happen, whether it’s an excellent patient encounter or the office did something really well together, highlight it so everyone has a sense of accomplishment. If a patient compliments a medical assistant or raves about a nurse, share those compliments with the employees so that not every encounter you have calls out problems and staff missteps.
Suppose partners have a conflict with one another or are arguing over something. In that case, you may need to mediate or invest in a meaningful intervention so people can reflect on the narrative they’re contributing to the culture.
5. Practice self-care
Finally, the work of a physician is exhausting, so it’s crucial to practice personal TLC. That may mean taking micro breaks, getting adequate sleep, maintaining a healthy diet, and exercising well and managing stress to maintain energy levels and patience.
“Sometimes, when I’m fed up with the office, I need to get away,” Dr. Montgomery says. “I’ll take a day to go fishing, golfing, and not think about the office.” Just a small break can shift the lens that you see through when you return to the office and put problems in perspective.
A version of this article first appeared on Medscape.com.
A physician in your office is hot-tempered, critical, and upsets both the physicians and staff. Two of your partners are arguing over a software vendor and refuse to compromise. One doctor’s spouse is the office manager and snipes at everyone; the lead partner micromanages and second-guesses other doctors’ treatment plans, and no one will stand up to her.
If your practice has similar scenarios, you’re likely dealing with your own anger, irritation, and dread at work. You’re struggling with a toxic practice atmosphere, and you must make changes – fast.
However, this isn’t easy, given that what goes on in a doctor’s office is “high consequence,” says Leonard J. Marcus, PhD, founding director of the program for health care negotiation and conflict resolution at the Harvard School of Public Health in Boston.
The two things that tend to plague medical practices most: A culture of fear and someone who is letting ego run the day-to-day, he says.
“Fear overwhelms any chance for good morale among colleagues,” says Dr. Marcus, who is also the coauthor of “Renegotiating Health Care: Resolving Conflict to Build Collaboration.” “In a work environment where the fear is overwhelming, the ego can take over, and someone at the practice becomes overly concerned about getting credit, taking control, ordering other people around, and deciding who is on top and who is on the bottom.”
Tension, stress, back-biting, and rudeness are also symptoms of a more significant problem, says Jes Montgomery, MD, a psychiatrist and medical director of APN Dallas, a mental health–focused practice.
“If you don’t get toxicity under control, it will blow the office apart,” Dr. Montgomery says.
1. Recognize the signs
Part of the problem with a toxic medical practice is that, culturally, we don’t treat mental health and burnout as real illnesses. “A physician who is depressed is not going to be melancholy or bursting into tears with patients,” Dr. Montgomery says. “They’ll get behind on paperwork, skip meals, or find that it’s difficult to sleep at night. Next, they’ll yell at the partners and staff, always be in a foul mood, and gripe about inconsequential things. Their behavior affects everyone.”
Dr. Montgomery says that physicians aren’t taught to ask for help, making it difficult to see what’s really going on when someone displays toxic behavior in the practice. If it’s a partner, take time to ask what’s going on. If it’s yourself, step back and see if you can ask someone for the help you need.
2. Have difficult conversations
This is tough for most of us, says Jeremy Pollack, PhD, CEO and founder of Pollack Peacebuilding Systems, a conflict resolution consulting firm. If a team member is hot-tempered, disrespectful, or talking to patients in an unproductive manner, see if you can have an effective conversation with that person. The tricky part is critiquing in a way that doesn’t make them feel defensive – and wanting to push back.
For a micromanaging office manager, for example, you could say something like,”You’re doing a great job with the inventory, but I need you to let the staff have some autonomy and not hover over every supply they use in the break room, so that people won’t feel resentful toward us.” Make it clear you’re a team, and this is a team challenge. “However, if a doctor feels like they’ve tried to communicate to that colleague and are still walking on eggshells, it’s time to try to get help from someone – perhaps a practice management organization,” says Dr. Pollack.
3. Open lines of communication
It’s critical to create a comfortable space to speak with your colleagues, says Marisa Garshick, MD, a dermatologist in private practice in New York. “Creating an environment where there is an open line of communication, whether it’s directly to somebody in charge or having a system where you can give feedback more privately or anonymously, is important so that tension doesn’t build.”
“Being a doctor is a social enterprise,” Dr. Marcus says. “The science of medicine is critically important, but patients and the other health care workers on your team are also critically important. In the long run, the most successful physicians pay attention to both. It’s a full package.”
4. Emphasize the positive
Instead of discussing things only when they go wrong, try optimism, Dr. Garshick said. When positive things happen, whether it’s an excellent patient encounter or the office did something really well together, highlight it so everyone has a sense of accomplishment. If a patient compliments a medical assistant or raves about a nurse, share those compliments with the employees so that not every encounter you have calls out problems and staff missteps.
Suppose partners have a conflict with one another or are arguing over something. In that case, you may need to mediate or invest in a meaningful intervention so people can reflect on the narrative they’re contributing to the culture.
5. Practice self-care
Finally, the work of a physician is exhausting, so it’s crucial to practice personal TLC. That may mean taking micro breaks, getting adequate sleep, maintaining a healthy diet, and exercising well and managing stress to maintain energy levels and patience.
“Sometimes, when I’m fed up with the office, I need to get away,” Dr. Montgomery says. “I’ll take a day to go fishing, golfing, and not think about the office.” Just a small break can shift the lens that you see through when you return to the office and put problems in perspective.
A version of this article first appeared on Medscape.com.
A physician in your office is hot-tempered, critical, and upsets both the physicians and staff. Two of your partners are arguing over a software vendor and refuse to compromise. One doctor’s spouse is the office manager and snipes at everyone; the lead partner micromanages and second-guesses other doctors’ treatment plans, and no one will stand up to her.
If your practice has similar scenarios, you’re likely dealing with your own anger, irritation, and dread at work. You’re struggling with a toxic practice atmosphere, and you must make changes – fast.
However, this isn’t easy, given that what goes on in a doctor’s office is “high consequence,” says Leonard J. Marcus, PhD, founding director of the program for health care negotiation and conflict resolution at the Harvard School of Public Health in Boston.
The two things that tend to plague medical practices most: A culture of fear and someone who is letting ego run the day-to-day, he says.
“Fear overwhelms any chance for good morale among colleagues,” says Dr. Marcus, who is also the coauthor of “Renegotiating Health Care: Resolving Conflict to Build Collaboration.” “In a work environment where the fear is overwhelming, the ego can take over, and someone at the practice becomes overly concerned about getting credit, taking control, ordering other people around, and deciding who is on top and who is on the bottom.”
Tension, stress, back-biting, and rudeness are also symptoms of a more significant problem, says Jes Montgomery, MD, a psychiatrist and medical director of APN Dallas, a mental health–focused practice.
“If you don’t get toxicity under control, it will blow the office apart,” Dr. Montgomery says.
1. Recognize the signs
Part of the problem with a toxic medical practice is that, culturally, we don’t treat mental health and burnout as real illnesses. “A physician who is depressed is not going to be melancholy or bursting into tears with patients,” Dr. Montgomery says. “They’ll get behind on paperwork, skip meals, or find that it’s difficult to sleep at night. Next, they’ll yell at the partners and staff, always be in a foul mood, and gripe about inconsequential things. Their behavior affects everyone.”
Dr. Montgomery says that physicians aren’t taught to ask for help, making it difficult to see what’s really going on when someone displays toxic behavior in the practice. If it’s a partner, take time to ask what’s going on. If it’s yourself, step back and see if you can ask someone for the help you need.
2. Have difficult conversations
This is tough for most of us, says Jeremy Pollack, PhD, CEO and founder of Pollack Peacebuilding Systems, a conflict resolution consulting firm. If a team member is hot-tempered, disrespectful, or talking to patients in an unproductive manner, see if you can have an effective conversation with that person. The tricky part is critiquing in a way that doesn’t make them feel defensive – and wanting to push back.
For a micromanaging office manager, for example, you could say something like,”You’re doing a great job with the inventory, but I need you to let the staff have some autonomy and not hover over every supply they use in the break room, so that people won’t feel resentful toward us.” Make it clear you’re a team, and this is a team challenge. “However, if a doctor feels like they’ve tried to communicate to that colleague and are still walking on eggshells, it’s time to try to get help from someone – perhaps a practice management organization,” says Dr. Pollack.
3. Open lines of communication
It’s critical to create a comfortable space to speak with your colleagues, says Marisa Garshick, MD, a dermatologist in private practice in New York. “Creating an environment where there is an open line of communication, whether it’s directly to somebody in charge or having a system where you can give feedback more privately or anonymously, is important so that tension doesn’t build.”
“Being a doctor is a social enterprise,” Dr. Marcus says. “The science of medicine is critically important, but patients and the other health care workers on your team are also critically important. In the long run, the most successful physicians pay attention to both. It’s a full package.”
4. Emphasize the positive
Instead of discussing things only when they go wrong, try optimism, Dr. Garshick said. When positive things happen, whether it’s an excellent patient encounter or the office did something really well together, highlight it so everyone has a sense of accomplishment. If a patient compliments a medical assistant or raves about a nurse, share those compliments with the employees so that not every encounter you have calls out problems and staff missteps.
Suppose partners have a conflict with one another or are arguing over something. In that case, you may need to mediate or invest in a meaningful intervention so people can reflect on the narrative they’re contributing to the culture.
5. Practice self-care
Finally, the work of a physician is exhausting, so it’s crucial to practice personal TLC. That may mean taking micro breaks, getting adequate sleep, maintaining a healthy diet, and exercising well and managing stress to maintain energy levels and patience.
“Sometimes, when I’m fed up with the office, I need to get away,” Dr. Montgomery says. “I’ll take a day to go fishing, golfing, and not think about the office.” Just a small break can shift the lens that you see through when you return to the office and put problems in perspective.
A version of this article first appeared on Medscape.com.
12 steps to closing your practice without problems
Whether you’ve decided to retire, relocate, or work for your local hospital, unwinding your practice will take time.
“Doctors shouldn’t assume everything takes care of itself. Many don’t think about compliance issues, patient abandonment, or accounts receivable that they need to keep open to collect from billing, which can occur months after the dates of service,” said David Zetter, president of Zetter HealthCare management consultants in Pennsylvania.
Debra Phairas, president of Practice and Liability Consultants, LLC, in California, suggests doctors start planning for the closing of their practice at least 90-120 days from their closing date.
“Many people and entities need to be notified,” said Ms. Phairas. The list includes patients, payers, vendors, employees, licensing boards, and federal and state agencies.
Medical societies may have specific bylaws that apply; malpractice carriers have rules about how long you should retain medical records; and some state laws require that you communicate that you’re closing in a newspaper, Mr. Zetter added.
Ms. Phairas recommends that physicians decide first whether they will sell their practice or if they’ll just shut it down. If they sell and the buyer is a doctor, they may want to provide transition assistance such as introducing patients and staff, she said. Otherwise, doctors may need to terminate their staff.
After doctors make that decision, Mr. Zetter and Ms. Phairas recommend taking these 12 steps to ensure that the process goes smoothly.
What to do 60-90 days out
1. Check your insurance contracts. The Centers for Medicare & Medicaid Services requires physicians to notify them 90 days after deciding to retire or withdraw from Medicare or Medicaid. Other payers may also require 90 days’ notice to terminate their contracts.
You’ll also need to provide payers with a forwarding address for sending payments after the office closes, and notify your malpractice insurance carrier and any other contracted insurance carriers such as workers’ compensation or employee benefit plans.
2. Buy “tail” coverage. Doctors can be sued for malpractice years after they close their practice so this provides coverage against claims reported after the liability policy expires.
3. Check your hospital contracts. Most hospitals where you have privileges require 90 days’ notice that you are closing the practice.
4. Arrange for safe storage of medical records. If you are selling your practice to another physician, that doctor can take charge of them, as long as you obtain a patient’s consent to transfer the medical records, said Ms. Phairas. Otherwise, the practice is required to make someone the guardian of the records after the practice closes, said Mr. Zetter. This allows patients at a later date to obtain copies of their records at a cost.
“This usually means printing all the records to PDF to be retained; otherwise, doctors have to continue to pay the license fee for the EMR software to access the records, and no practice is going to continue to pay this indefinitely,” said Mr. Zetter.
Check with your malpractice insurance carrier for how long they require medical records to be retained, which may vary for adult and pediatric records.
Ms. Phairas also advises doctors to keep their original records. “The biggest mistake doctors can make is to give patients all their records. Your chart is your best defense weapon in a liability claim.”
What to do 30-60 days out
5. Tell your staff. They should not hear that you’re retiring or leaving the practice from other people, said Ms. Phairas. But timing is important. “If you notify them too soon, they may look for another job. I recommend telling them about 45 days out and just before you notify patients, although you may want to tell the office manager sooner.”
Doctors may need help closing the practice and should consider offering the employees a severance bonus to stay until the end, said Ms. Phairas. If they do leave sooner, then you can hire temporary staff.
6. Notify patients to avoid any claims of abandonment. You should notify all active patients, which, depending on your state, can be any patient the physician has treated sometime in the past 12-36 months.
Some state laws require the notice to be published as an advertisement in the local newspaper and will say how far in advance it needs to be published and how long the ad needs to run. Notification also should be posted throughout the practice, and patients who call or visit should be given oral reminders.
“Your biggest expense will be mailing a letter to all patients,” said Mr. Zetter. The letter should include:
- The date of closing.
- The name(s) of the physicians taking over the practice (if applicable).
- Local physicians who would be willing to accept new patients.
- Instructions for how patients can obtain or transfer medical records (with a deadline for submitting record requests).
- How to contact the practice if patients and families have any concerns about the closing.
7. Notify your professional associations. These include your state medical board, credentialing organizations, and professional memberships. It’s critical to renew your license even if you plan to practice in other states. He recalled that one doctor let his license lapse and the medical board notified Medicaid that he was no longer licensed. “CMS went after him because he didn’t notify them that he was no longer operating in Washington. CMS shut him down in every state/territory. This interventional radiologist spent 3 years with two attorneys to get it resolved,” said Mr. Zetter.
8. Terminate any leases with landlords or try to negotiate renting the office space on a month-to-month basis until you close or sell, suggests Ms. Phairas. If the practice owns the space, the partners will need to decide if the space will be sold or leased to a new business.
What to do 30 days out
9. Notify referring physicians of when you plan to close your practice so they don’t send new patients after that date.
10. Send a letter to the Drug Enforcement Agency to deactivate your license if you plan not to write another prescription and after you have safely disposed of prescription drugs following the federal guidelines. Destroy all prescription pads and contact drug representatives to determine what to do with unused samples, if needed.
11. Notify all vendors. Inform medical suppliers, office suppliers, collection agencies, laundry services, housekeeping services, hazardous waste disposal services, and any other vendors. Make sure to request a final statement from them so you can close out your accounts.
12. Process your accounts receivable to collect money owed to you. Consider employing a collection agency or staff member to reconcile accounts after the practice has closed.
Mr. Zetter also suggested retaining a certified accountant to handle the expenses for shutting down the business and to handle your future tax returns. “If you shut down the practice in 2023, you will still have to file a tax return for that year in 2024,” he said.
A version of this article first appeared on Medscape.com.
Whether you’ve decided to retire, relocate, or work for your local hospital, unwinding your practice will take time.
“Doctors shouldn’t assume everything takes care of itself. Many don’t think about compliance issues, patient abandonment, or accounts receivable that they need to keep open to collect from billing, which can occur months after the dates of service,” said David Zetter, president of Zetter HealthCare management consultants in Pennsylvania.
Debra Phairas, president of Practice and Liability Consultants, LLC, in California, suggests doctors start planning for the closing of their practice at least 90-120 days from their closing date.
“Many people and entities need to be notified,” said Ms. Phairas. The list includes patients, payers, vendors, employees, licensing boards, and federal and state agencies.
Medical societies may have specific bylaws that apply; malpractice carriers have rules about how long you should retain medical records; and some state laws require that you communicate that you’re closing in a newspaper, Mr. Zetter added.
Ms. Phairas recommends that physicians decide first whether they will sell their practice or if they’ll just shut it down. If they sell and the buyer is a doctor, they may want to provide transition assistance such as introducing patients and staff, she said. Otherwise, doctors may need to terminate their staff.
After doctors make that decision, Mr. Zetter and Ms. Phairas recommend taking these 12 steps to ensure that the process goes smoothly.
What to do 60-90 days out
1. Check your insurance contracts. The Centers for Medicare & Medicaid Services requires physicians to notify them 90 days after deciding to retire or withdraw from Medicare or Medicaid. Other payers may also require 90 days’ notice to terminate their contracts.
You’ll also need to provide payers with a forwarding address for sending payments after the office closes, and notify your malpractice insurance carrier and any other contracted insurance carriers such as workers’ compensation or employee benefit plans.
2. Buy “tail” coverage. Doctors can be sued for malpractice years after they close their practice so this provides coverage against claims reported after the liability policy expires.
3. Check your hospital contracts. Most hospitals where you have privileges require 90 days’ notice that you are closing the practice.
4. Arrange for safe storage of medical records. If you are selling your practice to another physician, that doctor can take charge of them, as long as you obtain a patient’s consent to transfer the medical records, said Ms. Phairas. Otherwise, the practice is required to make someone the guardian of the records after the practice closes, said Mr. Zetter. This allows patients at a later date to obtain copies of their records at a cost.
“This usually means printing all the records to PDF to be retained; otherwise, doctors have to continue to pay the license fee for the EMR software to access the records, and no practice is going to continue to pay this indefinitely,” said Mr. Zetter.
Check with your malpractice insurance carrier for how long they require medical records to be retained, which may vary for adult and pediatric records.
Ms. Phairas also advises doctors to keep their original records. “The biggest mistake doctors can make is to give patients all their records. Your chart is your best defense weapon in a liability claim.”
What to do 30-60 days out
5. Tell your staff. They should not hear that you’re retiring or leaving the practice from other people, said Ms. Phairas. But timing is important. “If you notify them too soon, they may look for another job. I recommend telling them about 45 days out and just before you notify patients, although you may want to tell the office manager sooner.”
Doctors may need help closing the practice and should consider offering the employees a severance bonus to stay until the end, said Ms. Phairas. If they do leave sooner, then you can hire temporary staff.
6. Notify patients to avoid any claims of abandonment. You should notify all active patients, which, depending on your state, can be any patient the physician has treated sometime in the past 12-36 months.
Some state laws require the notice to be published as an advertisement in the local newspaper and will say how far in advance it needs to be published and how long the ad needs to run. Notification also should be posted throughout the practice, and patients who call or visit should be given oral reminders.
“Your biggest expense will be mailing a letter to all patients,” said Mr. Zetter. The letter should include:
- The date of closing.
- The name(s) of the physicians taking over the practice (if applicable).
- Local physicians who would be willing to accept new patients.
- Instructions for how patients can obtain or transfer medical records (with a deadline for submitting record requests).
- How to contact the practice if patients and families have any concerns about the closing.
7. Notify your professional associations. These include your state medical board, credentialing organizations, and professional memberships. It’s critical to renew your license even if you plan to practice in other states. He recalled that one doctor let his license lapse and the medical board notified Medicaid that he was no longer licensed. “CMS went after him because he didn’t notify them that he was no longer operating in Washington. CMS shut him down in every state/territory. This interventional radiologist spent 3 years with two attorneys to get it resolved,” said Mr. Zetter.
8. Terminate any leases with landlords or try to negotiate renting the office space on a month-to-month basis until you close or sell, suggests Ms. Phairas. If the practice owns the space, the partners will need to decide if the space will be sold or leased to a new business.
What to do 30 days out
9. Notify referring physicians of when you plan to close your practice so they don’t send new patients after that date.
10. Send a letter to the Drug Enforcement Agency to deactivate your license if you plan not to write another prescription and after you have safely disposed of prescription drugs following the federal guidelines. Destroy all prescription pads and contact drug representatives to determine what to do with unused samples, if needed.
11. Notify all vendors. Inform medical suppliers, office suppliers, collection agencies, laundry services, housekeeping services, hazardous waste disposal services, and any other vendors. Make sure to request a final statement from them so you can close out your accounts.
12. Process your accounts receivable to collect money owed to you. Consider employing a collection agency or staff member to reconcile accounts after the practice has closed.
Mr. Zetter also suggested retaining a certified accountant to handle the expenses for shutting down the business and to handle your future tax returns. “If you shut down the practice in 2023, you will still have to file a tax return for that year in 2024,” he said.
A version of this article first appeared on Medscape.com.
Whether you’ve decided to retire, relocate, or work for your local hospital, unwinding your practice will take time.
“Doctors shouldn’t assume everything takes care of itself. Many don’t think about compliance issues, patient abandonment, or accounts receivable that they need to keep open to collect from billing, which can occur months after the dates of service,” said David Zetter, president of Zetter HealthCare management consultants in Pennsylvania.
Debra Phairas, president of Practice and Liability Consultants, LLC, in California, suggests doctors start planning for the closing of their practice at least 90-120 days from their closing date.
“Many people and entities need to be notified,” said Ms. Phairas. The list includes patients, payers, vendors, employees, licensing boards, and federal and state agencies.
Medical societies may have specific bylaws that apply; malpractice carriers have rules about how long you should retain medical records; and some state laws require that you communicate that you’re closing in a newspaper, Mr. Zetter added.
Ms. Phairas recommends that physicians decide first whether they will sell their practice or if they’ll just shut it down. If they sell and the buyer is a doctor, they may want to provide transition assistance such as introducing patients and staff, she said. Otherwise, doctors may need to terminate their staff.
After doctors make that decision, Mr. Zetter and Ms. Phairas recommend taking these 12 steps to ensure that the process goes smoothly.
What to do 60-90 days out
1. Check your insurance contracts. The Centers for Medicare & Medicaid Services requires physicians to notify them 90 days after deciding to retire or withdraw from Medicare or Medicaid. Other payers may also require 90 days’ notice to terminate their contracts.
You’ll also need to provide payers with a forwarding address for sending payments after the office closes, and notify your malpractice insurance carrier and any other contracted insurance carriers such as workers’ compensation or employee benefit plans.
2. Buy “tail” coverage. Doctors can be sued for malpractice years after they close their practice so this provides coverage against claims reported after the liability policy expires.
3. Check your hospital contracts. Most hospitals where you have privileges require 90 days’ notice that you are closing the practice.
4. Arrange for safe storage of medical records. If you are selling your practice to another physician, that doctor can take charge of them, as long as you obtain a patient’s consent to transfer the medical records, said Ms. Phairas. Otherwise, the practice is required to make someone the guardian of the records after the practice closes, said Mr. Zetter. This allows patients at a later date to obtain copies of their records at a cost.
“This usually means printing all the records to PDF to be retained; otherwise, doctors have to continue to pay the license fee for the EMR software to access the records, and no practice is going to continue to pay this indefinitely,” said Mr. Zetter.
Check with your malpractice insurance carrier for how long they require medical records to be retained, which may vary for adult and pediatric records.
Ms. Phairas also advises doctors to keep their original records. “The biggest mistake doctors can make is to give patients all their records. Your chart is your best defense weapon in a liability claim.”
What to do 30-60 days out
5. Tell your staff. They should not hear that you’re retiring or leaving the practice from other people, said Ms. Phairas. But timing is important. “If you notify them too soon, they may look for another job. I recommend telling them about 45 days out and just before you notify patients, although you may want to tell the office manager sooner.”
Doctors may need help closing the practice and should consider offering the employees a severance bonus to stay until the end, said Ms. Phairas. If they do leave sooner, then you can hire temporary staff.
6. Notify patients to avoid any claims of abandonment. You should notify all active patients, which, depending on your state, can be any patient the physician has treated sometime in the past 12-36 months.
Some state laws require the notice to be published as an advertisement in the local newspaper and will say how far in advance it needs to be published and how long the ad needs to run. Notification also should be posted throughout the practice, and patients who call or visit should be given oral reminders.
“Your biggest expense will be mailing a letter to all patients,” said Mr. Zetter. The letter should include:
- The date of closing.
- The name(s) of the physicians taking over the practice (if applicable).
- Local physicians who would be willing to accept new patients.
- Instructions for how patients can obtain or transfer medical records (with a deadline for submitting record requests).
- How to contact the practice if patients and families have any concerns about the closing.
7. Notify your professional associations. These include your state medical board, credentialing organizations, and professional memberships. It’s critical to renew your license even if you plan to practice in other states. He recalled that one doctor let his license lapse and the medical board notified Medicaid that he was no longer licensed. “CMS went after him because he didn’t notify them that he was no longer operating in Washington. CMS shut him down in every state/territory. This interventional radiologist spent 3 years with two attorneys to get it resolved,” said Mr. Zetter.
8. Terminate any leases with landlords or try to negotiate renting the office space on a month-to-month basis until you close or sell, suggests Ms. Phairas. If the practice owns the space, the partners will need to decide if the space will be sold or leased to a new business.
What to do 30 days out
9. Notify referring physicians of when you plan to close your practice so they don’t send new patients after that date.
10. Send a letter to the Drug Enforcement Agency to deactivate your license if you plan not to write another prescription and after you have safely disposed of prescription drugs following the federal guidelines. Destroy all prescription pads and contact drug representatives to determine what to do with unused samples, if needed.
11. Notify all vendors. Inform medical suppliers, office suppliers, collection agencies, laundry services, housekeeping services, hazardous waste disposal services, and any other vendors. Make sure to request a final statement from them so you can close out your accounts.
12. Process your accounts receivable to collect money owed to you. Consider employing a collection agency or staff member to reconcile accounts after the practice has closed.
Mr. Zetter also suggested retaining a certified accountant to handle the expenses for shutting down the business and to handle your future tax returns. “If you shut down the practice in 2023, you will still have to file a tax return for that year in 2024,” he said.
A version of this article first appeared on Medscape.com.
Bad blood: Could brain bleeds be contagious?
This transcript has been edited for clarity.
How do you tell if a condition is caused by an infection?
It seems like an obvious question, right? In the post–van Leeuwenhoek era we can look at whatever part of the body is diseased under a microscope and see microbes – you know, the usual suspects.
Except when we can’t. And there are plenty of cases where we can’t: where the microbe is too small to be seen without more advanced imaging techniques, like with viruses; or when the pathogen is sparsely populated or hard to culture, like Mycobacterium.
Finding out that a condition is the result of an infection is not only an exercise for 19th century physicians. After all, it was 2008 when Barry Marshall and Robin Warren won their Nobel Prize for proving that stomach ulcers, long thought to be due to “stress,” were actually caused by a tiny microbe called Helicobacter pylori.
And this week, we are looking at a study which, once again, begins to suggest that a condition thought to be more or less random – cerebral amyloid angiopathy – may actually be the result of an infectious disease.
We’re talking about this paper, appearing in JAMA, which is just a great example of old-fashioned shoe-leather epidemiology. But let’s get up to speed on cerebral amyloid angiopathy (CAA) first.
CAA is characterized by the deposition of amyloid protein in the brain. While there are some genetic causes, they are quite rare, and most cases are thought to be idiopathic. Recent analyses suggest that somewhere between 5% and 7% of cognitively normal older adults have CAA, but the rate is much higher among those with intracerebral hemorrhage – brain bleeds. In fact, CAA is the second-most common cause of bleeding in the brain, second only to severe hypertension.
An article in Nature highlights cases that seemed to develop after the administration of cadaveric pituitary hormone.
Other studies have shown potential transmission via dura mater grafts and neurosurgical instruments. But despite those clues, no infectious organism has been identified. Some have suggested that the long latent period and difficulty of finding a responsible microbe points to a prion-like disease not yet known. But these studies are more or less case series. The new JAMA paper gives us, if not a smoking gun, a pretty decent set of fingerprints.
Here’s the idea: If CAA is caused by some infectious agent, it may be transmitted in the blood. We know that a decent percentage of people who have spontaneous brain bleeds have CAA. If those people donated blood in the past, maybe the people who received that blood would be at risk for brain bleeds too.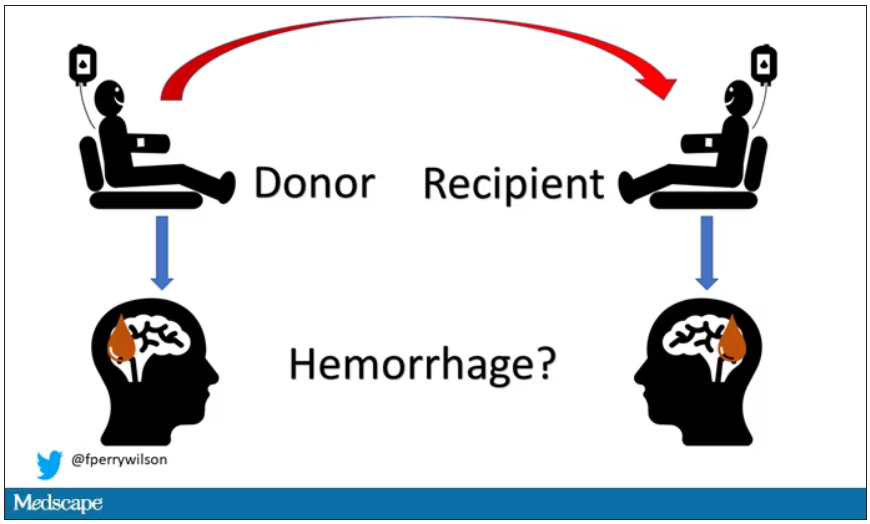
Of course, to really test that hypothesis, you’d need to know who every blood donor in a country was and every person who received that blood and all their subsequent diagnoses for basically their entire lives. No one has that kind of data, right?
Well, if you’ve been watching this space, you’ll know that a few countries do. Enter Sweden and Denmark, with their national electronic health record that captures all of this information, and much more, on every single person who lives or has lived in those countries since before 1970. Unbelievable.
So that’s exactly what the researchers, led by Jingchen Zhao at Karolinska (Sweden) University, did. They identified roughly 760,000 individuals in Sweden and 330,000 people in Denmark who had received a blood transfusion between 1970 and 2017.
Of course, most of those blood donors – 99% of them, actually – never went on to have any bleeding in the brain. It is a rare thing, fortunately.
But some of the donors did, on average within about 5 years of the time they donated blood. The researchers characterized each donor as either never having a brain bleed, having a single bleed, or having multiple bleeds. The latter is most strongly associated with CAA.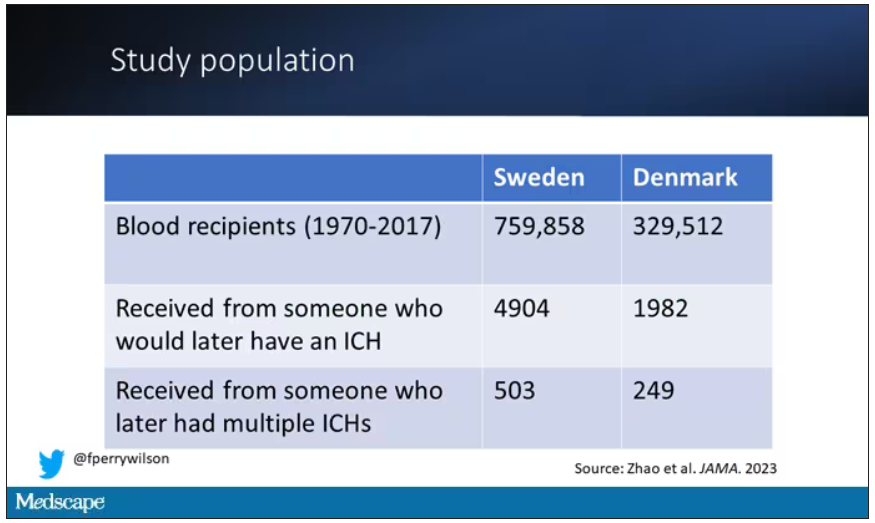
The big question: Would recipients who got blood from individuals who later on had brain bleeds, have brain bleeds themselves?
The answer is yes, though with an asterisk. You can see the results here. The risk of recipients having a brain bleed was lowest if the blood they received was from people who never had a brain bleed, higher if the individual had a single brain bleed, and highest if they got blood from a donor who would go on to have multiple brain bleeds.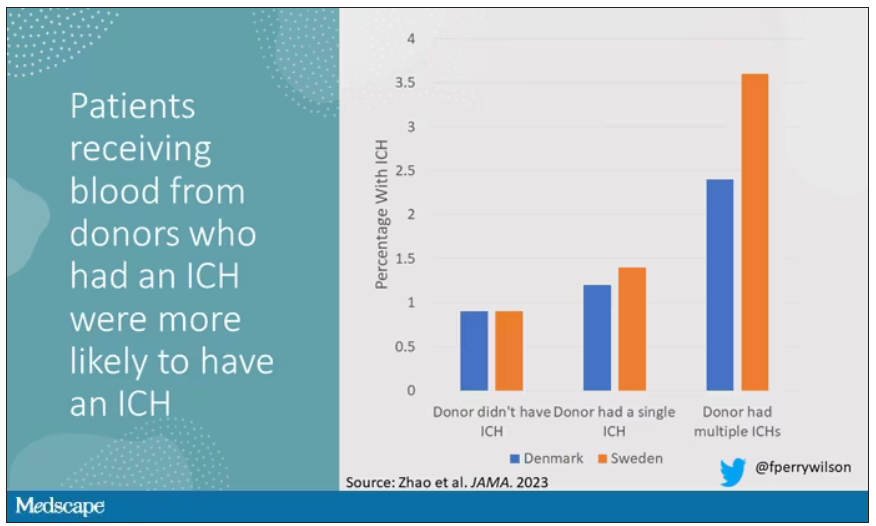
All in all, individuals who received blood from someone who would later have multiple hemorrhages were three times more likely to themselves develop bleeds themselves. It’s fairly compelling evidence of a transmissible agent.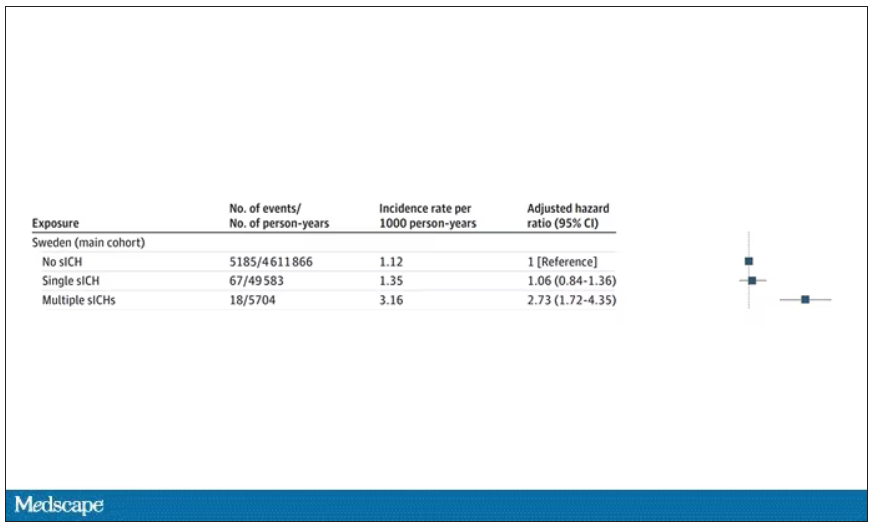
Of course, there are some potential confounders to consider here. Whose blood you get is not totally random. If, for example, people with type O blood are just more likely to have brain bleeds, then you could get results like this, as type O tends to donate to type O and both groups would have higher risk after donation. But the authors adjusted for blood type. They also adjusted for number of transfusions, calendar year, age, sex, and indication for transfusion.
Perhaps most compelling, and most clever, is that they used ischemic stroke as a negative control. Would people who received blood from someone who later had an ischemic stroke themselves be more likely to go on to have an ischemic stroke? No signal at all. It does not appear that there is a transmissible agent associated with ischemic stroke – only the brain bleeds.
I know what you’re thinking. What’s the agent? What’s the microbe, or virus, or prion, or toxin? The study gives us no insight there. These nationwide databases are awesome but they can only do so much. Because of the vagaries of medical coding and the difficulty of making the CAA diagnosis, the authors are using brain bleeds as a proxy here; we don’t even know for sure whether these were CAA-associated brain bleeds.
It’s also worth noting that there’s little we can do about this. None of the blood donors in this study had a brain bleed prior to donation; it’s not like we could screen people out of donating in the future. We have no test for whatever this agent is, if it even exists, nor do we have a potential treatment. Fortunately, whatever it is, it is extremely rare.
Still, this paper feels like a shot across the bow. At this point, the probability has shifted strongly away from CAA being a purely random disease and toward it being an infectious one. It may be time to round up some of the unusual suspects.
Dr. F. Perry Wilson is an associate professor of medicine and public health and director of Yale University’s Clinical and Translational Research Accelerator in New Haven, Conn. He reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
How do you tell if a condition is caused by an infection?
It seems like an obvious question, right? In the post–van Leeuwenhoek era we can look at whatever part of the body is diseased under a microscope and see microbes – you know, the usual suspects.
Except when we can’t. And there are plenty of cases where we can’t: where the microbe is too small to be seen without more advanced imaging techniques, like with viruses; or when the pathogen is sparsely populated or hard to culture, like Mycobacterium.
Finding out that a condition is the result of an infection is not only an exercise for 19th century physicians. After all, it was 2008 when Barry Marshall and Robin Warren won their Nobel Prize for proving that stomach ulcers, long thought to be due to “stress,” were actually caused by a tiny microbe called Helicobacter pylori.
And this week, we are looking at a study which, once again, begins to suggest that a condition thought to be more or less random – cerebral amyloid angiopathy – may actually be the result of an infectious disease.
We’re talking about this paper, appearing in JAMA, which is just a great example of old-fashioned shoe-leather epidemiology. But let’s get up to speed on cerebral amyloid angiopathy (CAA) first.
CAA is characterized by the deposition of amyloid protein in the brain. While there are some genetic causes, they are quite rare, and most cases are thought to be idiopathic. Recent analyses suggest that somewhere between 5% and 7% of cognitively normal older adults have CAA, but the rate is much higher among those with intracerebral hemorrhage – brain bleeds. In fact, CAA is the second-most common cause of bleeding in the brain, second only to severe hypertension.
An article in Nature highlights cases that seemed to develop after the administration of cadaveric pituitary hormone.
Other studies have shown potential transmission via dura mater grafts and neurosurgical instruments. But despite those clues, no infectious organism has been identified. Some have suggested that the long latent period and difficulty of finding a responsible microbe points to a prion-like disease not yet known. But these studies are more or less case series. The new JAMA paper gives us, if not a smoking gun, a pretty decent set of fingerprints.
Here’s the idea: If CAA is caused by some infectious agent, it may be transmitted in the blood. We know that a decent percentage of people who have spontaneous brain bleeds have CAA. If those people donated blood in the past, maybe the people who received that blood would be at risk for brain bleeds too.
Of course, to really test that hypothesis, you’d need to know who every blood donor in a country was and every person who received that blood and all their subsequent diagnoses for basically their entire lives. No one has that kind of data, right?
Well, if you’ve been watching this space, you’ll know that a few countries do. Enter Sweden and Denmark, with their national electronic health record that captures all of this information, and much more, on every single person who lives or has lived in those countries since before 1970. Unbelievable.
So that’s exactly what the researchers, led by Jingchen Zhao at Karolinska (Sweden) University, did. They identified roughly 760,000 individuals in Sweden and 330,000 people in Denmark who had received a blood transfusion between 1970 and 2017.
Of course, most of those blood donors – 99% of them, actually – never went on to have any bleeding in the brain. It is a rare thing, fortunately.
But some of the donors did, on average within about 5 years of the time they donated blood. The researchers characterized each donor as either never having a brain bleed, having a single bleed, or having multiple bleeds. The latter is most strongly associated with CAA.
The big question: Would recipients who got blood from individuals who later on had brain bleeds, have brain bleeds themselves?
The answer is yes, though with an asterisk. You can see the results here. The risk of recipients having a brain bleed was lowest if the blood they received was from people who never had a brain bleed, higher if the individual had a single brain bleed, and highest if they got blood from a donor who would go on to have multiple brain bleeds.
All in all, individuals who received blood from someone who would later have multiple hemorrhages were three times more likely to themselves develop bleeds themselves. It’s fairly compelling evidence of a transmissible agent.
Of course, there are some potential confounders to consider here. Whose blood you get is not totally random. If, for example, people with type O blood are just more likely to have brain bleeds, then you could get results like this, as type O tends to donate to type O and both groups would have higher risk after donation. But the authors adjusted for blood type. They also adjusted for number of transfusions, calendar year, age, sex, and indication for transfusion.
Perhaps most compelling, and most clever, is that they used ischemic stroke as a negative control. Would people who received blood from someone who later had an ischemic stroke themselves be more likely to go on to have an ischemic stroke? No signal at all. It does not appear that there is a transmissible agent associated with ischemic stroke – only the brain bleeds.
I know what you’re thinking. What’s the agent? What’s the microbe, or virus, or prion, or toxin? The study gives us no insight there. These nationwide databases are awesome but they can only do so much. Because of the vagaries of medical coding and the difficulty of making the CAA diagnosis, the authors are using brain bleeds as a proxy here; we don’t even know for sure whether these were CAA-associated brain bleeds.
It’s also worth noting that there’s little we can do about this. None of the blood donors in this study had a brain bleed prior to donation; it’s not like we could screen people out of donating in the future. We have no test for whatever this agent is, if it even exists, nor do we have a potential treatment. Fortunately, whatever it is, it is extremely rare.
Still, this paper feels like a shot across the bow. At this point, the probability has shifted strongly away from CAA being a purely random disease and toward it being an infectious one. It may be time to round up some of the unusual suspects.
Dr. F. Perry Wilson is an associate professor of medicine and public health and director of Yale University’s Clinical and Translational Research Accelerator in New Haven, Conn. He reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
How do you tell if a condition is caused by an infection?
It seems like an obvious question, right? In the post–van Leeuwenhoek era we can look at whatever part of the body is diseased under a microscope and see microbes – you know, the usual suspects.
Except when we can’t. And there are plenty of cases where we can’t: where the microbe is too small to be seen without more advanced imaging techniques, like with viruses; or when the pathogen is sparsely populated or hard to culture, like Mycobacterium.
Finding out that a condition is the result of an infection is not only an exercise for 19th century physicians. After all, it was 2008 when Barry Marshall and Robin Warren won their Nobel Prize for proving that stomach ulcers, long thought to be due to “stress,” were actually caused by a tiny microbe called Helicobacter pylori.
And this week, we are looking at a study which, once again, begins to suggest that a condition thought to be more or less random – cerebral amyloid angiopathy – may actually be the result of an infectious disease.
We’re talking about this paper, appearing in JAMA, which is just a great example of old-fashioned shoe-leather epidemiology. But let’s get up to speed on cerebral amyloid angiopathy (CAA) first.
CAA is characterized by the deposition of amyloid protein in the brain. While there are some genetic causes, they are quite rare, and most cases are thought to be idiopathic. Recent analyses suggest that somewhere between 5% and 7% of cognitively normal older adults have CAA, but the rate is much higher among those with intracerebral hemorrhage – brain bleeds. In fact, CAA is the second-most common cause of bleeding in the brain, second only to severe hypertension.
An article in Nature highlights cases that seemed to develop after the administration of cadaveric pituitary hormone.
Other studies have shown potential transmission via dura mater grafts and neurosurgical instruments. But despite those clues, no infectious organism has been identified. Some have suggested that the long latent period and difficulty of finding a responsible microbe points to a prion-like disease not yet known. But these studies are more or less case series. The new JAMA paper gives us, if not a smoking gun, a pretty decent set of fingerprints.
Here’s the idea: If CAA is caused by some infectious agent, it may be transmitted in the blood. We know that a decent percentage of people who have spontaneous brain bleeds have CAA. If those people donated blood in the past, maybe the people who received that blood would be at risk for brain bleeds too.
Of course, to really test that hypothesis, you’d need to know who every blood donor in a country was and every person who received that blood and all their subsequent diagnoses for basically their entire lives. No one has that kind of data, right?
Well, if you’ve been watching this space, you’ll know that a few countries do. Enter Sweden and Denmark, with their national electronic health record that captures all of this information, and much more, on every single person who lives or has lived in those countries since before 1970. Unbelievable.
So that’s exactly what the researchers, led by Jingchen Zhao at Karolinska (Sweden) University, did. They identified roughly 760,000 individuals in Sweden and 330,000 people in Denmark who had received a blood transfusion between 1970 and 2017.
Of course, most of those blood donors – 99% of them, actually – never went on to have any bleeding in the brain. It is a rare thing, fortunately.
But some of the donors did, on average within about 5 years of the time they donated blood. The researchers characterized each donor as either never having a brain bleed, having a single bleed, or having multiple bleeds. The latter is most strongly associated with CAA.
The big question: Would recipients who got blood from individuals who later on had brain bleeds, have brain bleeds themselves?
The answer is yes, though with an asterisk. You can see the results here. The risk of recipients having a brain bleed was lowest if the blood they received was from people who never had a brain bleed, higher if the individual had a single brain bleed, and highest if they got blood from a donor who would go on to have multiple brain bleeds.
All in all, individuals who received blood from someone who would later have multiple hemorrhages were three times more likely to themselves develop bleeds themselves. It’s fairly compelling evidence of a transmissible agent.
Of course, there are some potential confounders to consider here. Whose blood you get is not totally random. If, for example, people with type O blood are just more likely to have brain bleeds, then you could get results like this, as type O tends to donate to type O and both groups would have higher risk after donation. But the authors adjusted for blood type. They also adjusted for number of transfusions, calendar year, age, sex, and indication for transfusion.
Perhaps most compelling, and most clever, is that they used ischemic stroke as a negative control. Would people who received blood from someone who later had an ischemic stroke themselves be more likely to go on to have an ischemic stroke? No signal at all. It does not appear that there is a transmissible agent associated with ischemic stroke – only the brain bleeds.
I know what you’re thinking. What’s the agent? What’s the microbe, or virus, or prion, or toxin? The study gives us no insight there. These nationwide databases are awesome but they can only do so much. Because of the vagaries of medical coding and the difficulty of making the CAA diagnosis, the authors are using brain bleeds as a proxy here; we don’t even know for sure whether these were CAA-associated brain bleeds.
It’s also worth noting that there’s little we can do about this. None of the blood donors in this study had a brain bleed prior to donation; it’s not like we could screen people out of donating in the future. We have no test for whatever this agent is, if it even exists, nor do we have a potential treatment. Fortunately, whatever it is, it is extremely rare.
Still, this paper feels like a shot across the bow. At this point, the probability has shifted strongly away from CAA being a purely random disease and toward it being an infectious one. It may be time to round up some of the unusual suspects.
Dr. F. Perry Wilson is an associate professor of medicine and public health and director of Yale University’s Clinical and Translational Research Accelerator in New Haven, Conn. He reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
Disenfranchised grief: What it looks like, where it goes
What happens to grief when those around you don’t understand it? Where does it go? How do you process it?
Disenfranchised grief, when someone or society more generally doesn’t see a loss as worthy of mourning, can deprive people of experiencing or processing their sadness. This grief, which may be triggered by the death of an ex-spouse, a pet, a failed adoption, can be painful and long-lasting.
Suzanne Cole, MD: ‘I didn’t feel the right to grieve’
During the COVID-19 pandemic, my little sister unexpectedly died. Though she was not one of the nearly 7 million people who died of the virus, in 2021 she became another type of statistic: one of the 109,699 people in the United State who died from a drug overdose. Hers was from fentanyl laced with methamphetamines.
Her death unraveled me. I felt deep guilt that I could not pull her from the sweeping current that had wrenched her from mainstream society into the underbelly of sex work and toward the solace of mind-altering drugs.
But I did not feel the right to grieve for her as I have grieved for other loved ones who were not blamed for their exit from this world. My sister was living a sordid life on the fringes of society. My grief felt invalid, undeserved. Yet, in the eyes of other “upstanding citizens,” her life was not as worth grieving – or so I thought. I tucked my sorrow into a small corner of my soul so no one would see, and I carried on.
To this day, the shame I feel robbed me of the ability to freely talk about her or share the searing pain I feel. Tears still prick my eyes when I think of her, but I have become adept at swallowing them, shaking off the waves of grief as though nothing happened. Even now, I cannot shake the pervasive feeling that my silent tears don’t deserve to be wept.
Don S. Dizon, MD: Working through tragedy
As a medical student, I worked with an outpatient physician as part of a third-year rotation. When we met, the first thing that struck me was how disheveled he looked. His clothes were wrinkled, and his pants were baggy. He took cigarette breaks, which I found disturbing.
But I quickly came to admire him. Despite my first impression, he was the type of doctor I aspired to be. He didn’t need to look at a patient’s chart to recall who they were. He just knew them. He greeted patients warmly, asked about their family. He even remembered the special occasions his patients had mentioned since their past visit. He epitomized empathy and connectedness.
Spending one day in clinic brought to light the challenges of forming such bonds with patients. A man came into the cancer clinic reporting chest pain and was triaged to an exam room. Soon after, the patient was found unresponsive on the floor. Nurses were yelling for help, and the doctor ran in and started CPR while minutes ticked by waiting for an ambulance that could take him to the ED.
By the time help arrived, the patient was blue.
He had died in the clinic in the middle of the day, as the waiting room filled. After the body was taken away, the doctor went into the bathroom. About 20 minutes later, he came out, eyes bloodshot, and continued with the rest of his day, ensuring each patient was seen and cared for.
As a medical student, it hit me how hard it must be to see something so tragic like the end of a life and then continue with your day as if nothing had happened. This is an experience of grief I later came to know well after nearly 30 years treating patients with advanced cancers: compartmentalizing it and carrying on.
A space for grieving: The Schwartz Center Rounds
Disenfranchised grief, the grief that is hard to share and often seems wrong to feel in the first place, can be triggered in many situations. Losing a person others don’t believe deserve to be grieved, such as an abusive partner or someone who committed a crime; losing someone you cared for in a professional role; a loss experienced in a breakup or same-sex partnership, if that relationship was not accepted by one’s family; loss from infertility, miscarriage, stillbirth, or failed adoption; loss that may be taboo or stigmatized, such as deaths via suicide or abortion; and loss of a job, home, or possession that you treasure.
Many of us have had similar situations or will, and the feeling that no one understands the need to mourn can be paralyzing and alienating. In the early days, intense, crushing feelings can cause intrusive, distracting thoughts, and over time, that grief can linger and find a permanent place in our minds.
More and more, though, we are being given opportunities to reflect on these sad moments.
The Schwartz Rounds are an example of such an opportunity. In these rounds, we gather to talk about the experience of caring for people, not the science of medicine.
During one particularly powerful rounds, I spoke to my colleagues about my initial meeting with a patient who was very sick. I detailed the experience of telling her children and her at that initial consult how I thought she was dying and that I did not recommend therapy. I remember how they cried. And I remembered how powerless I felt.
As I recalled that memory during Schwartz Rounds, I could not stop from crying. The unfairness of being a physician meeting someone for the first time and having to tell them such bad news overwhelmed me.
Even more poignant, I had the chance to reconnect with this woman’s children, who were present that day, not as audience members but as participants. Their presence may have brought my emotions to the surface more strongly. In that moment, I could show them the feelings I had bottled up for the sake of professionalism. Ultimately, I felt relieved, freer somehow, as if this burden my soul was carrying had been lifted.
Although we are both grateful for forums like this, these opportunities to share and express the grief we may have hidden away are not as common as they should be.
As physicians, we may express grief by shedding tears at the bedside of a patient nearing the end of life or through the anxiety we feel when our patient suffers a severe reaction to treatment. But we tend to put it away, to go on with our day, because there are others to be seen and cared for and more work to be done. Somehow, we move forward, shedding tears in one room and celebrating victories in another.
We need to create more spaces to express and feel grief, so we don’t get lost in it. Because understanding how grief impacts us, as people and as providers, is one of the most important realizations we can make as we go about our time-honored profession as healers.
Dr. Dizon is the director of women’s cancers at Lifespan Cancer Institute, director of medical oncology at Rhode Island Hospital, and a professor of medicine at Brown University, all in Providence. He reported conflicts of interest with Regeneron, AstraZeneca, Clovis, Bristol-Myers Squibb, and Kazia.
A version of this article first appeared on Medscape.com.
What happens to grief when those around you don’t understand it? Where does it go? How do you process it?
Disenfranchised grief, when someone or society more generally doesn’t see a loss as worthy of mourning, can deprive people of experiencing or processing their sadness. This grief, which may be triggered by the death of an ex-spouse, a pet, a failed adoption, can be painful and long-lasting.
Suzanne Cole, MD: ‘I didn’t feel the right to grieve’
During the COVID-19 pandemic, my little sister unexpectedly died. Though she was not one of the nearly 7 million people who died of the virus, in 2021 she became another type of statistic: one of the 109,699 people in the United State who died from a drug overdose. Hers was from fentanyl laced with methamphetamines.
Her death unraveled me. I felt deep guilt that I could not pull her from the sweeping current that had wrenched her from mainstream society into the underbelly of sex work and toward the solace of mind-altering drugs.
But I did not feel the right to grieve for her as I have grieved for other loved ones who were not blamed for their exit from this world. My sister was living a sordid life on the fringes of society. My grief felt invalid, undeserved. Yet, in the eyes of other “upstanding citizens,” her life was not as worth grieving – or so I thought. I tucked my sorrow into a small corner of my soul so no one would see, and I carried on.
To this day, the shame I feel robbed me of the ability to freely talk about her or share the searing pain I feel. Tears still prick my eyes when I think of her, but I have become adept at swallowing them, shaking off the waves of grief as though nothing happened. Even now, I cannot shake the pervasive feeling that my silent tears don’t deserve to be wept.
Don S. Dizon, MD: Working through tragedy
As a medical student, I worked with an outpatient physician as part of a third-year rotation. When we met, the first thing that struck me was how disheveled he looked. His clothes were wrinkled, and his pants were baggy. He took cigarette breaks, which I found disturbing.
But I quickly came to admire him. Despite my first impression, he was the type of doctor I aspired to be. He didn’t need to look at a patient’s chart to recall who they were. He just knew them. He greeted patients warmly, asked about their family. He even remembered the special occasions his patients had mentioned since their past visit. He epitomized empathy and connectedness.
Spending one day in clinic brought to light the challenges of forming such bonds with patients. A man came into the cancer clinic reporting chest pain and was triaged to an exam room. Soon after, the patient was found unresponsive on the floor. Nurses were yelling for help, and the doctor ran in and started CPR while minutes ticked by waiting for an ambulance that could take him to the ED.
By the time help arrived, the patient was blue.
He had died in the clinic in the middle of the day, as the waiting room filled. After the body was taken away, the doctor went into the bathroom. About 20 minutes later, he came out, eyes bloodshot, and continued with the rest of his day, ensuring each patient was seen and cared for.
As a medical student, it hit me how hard it must be to see something so tragic like the end of a life and then continue with your day as if nothing had happened. This is an experience of grief I later came to know well after nearly 30 years treating patients with advanced cancers: compartmentalizing it and carrying on.
A space for grieving: The Schwartz Center Rounds
Disenfranchised grief, the grief that is hard to share and often seems wrong to feel in the first place, can be triggered in many situations. Losing a person others don’t believe deserve to be grieved, such as an abusive partner or someone who committed a crime; losing someone you cared for in a professional role; a loss experienced in a breakup or same-sex partnership, if that relationship was not accepted by one’s family; loss from infertility, miscarriage, stillbirth, or failed adoption; loss that may be taboo or stigmatized, such as deaths via suicide or abortion; and loss of a job, home, or possession that you treasure.
Many of us have had similar situations or will, and the feeling that no one understands the need to mourn can be paralyzing and alienating. In the early days, intense, crushing feelings can cause intrusive, distracting thoughts, and over time, that grief can linger and find a permanent place in our minds.
More and more, though, we are being given opportunities to reflect on these sad moments.
The Schwartz Rounds are an example of such an opportunity. In these rounds, we gather to talk about the experience of caring for people, not the science of medicine.
During one particularly powerful rounds, I spoke to my colleagues about my initial meeting with a patient who was very sick. I detailed the experience of telling her children and her at that initial consult how I thought she was dying and that I did not recommend therapy. I remember how they cried. And I remembered how powerless I felt.
As I recalled that memory during Schwartz Rounds, I could not stop from crying. The unfairness of being a physician meeting someone for the first time and having to tell them such bad news overwhelmed me.
Even more poignant, I had the chance to reconnect with this woman’s children, who were present that day, not as audience members but as participants. Their presence may have brought my emotions to the surface more strongly. In that moment, I could show them the feelings I had bottled up for the sake of professionalism. Ultimately, I felt relieved, freer somehow, as if this burden my soul was carrying had been lifted.
Although we are both grateful for forums like this, these opportunities to share and express the grief we may have hidden away are not as common as they should be.
As physicians, we may express grief by shedding tears at the bedside of a patient nearing the end of life or through the anxiety we feel when our patient suffers a severe reaction to treatment. But we tend to put it away, to go on with our day, because there are others to be seen and cared for and more work to be done. Somehow, we move forward, shedding tears in one room and celebrating victories in another.
We need to create more spaces to express and feel grief, so we don’t get lost in it. Because understanding how grief impacts us, as people and as providers, is one of the most important realizations we can make as we go about our time-honored profession as healers.
Dr. Dizon is the director of women’s cancers at Lifespan Cancer Institute, director of medical oncology at Rhode Island Hospital, and a professor of medicine at Brown University, all in Providence. He reported conflicts of interest with Regeneron, AstraZeneca, Clovis, Bristol-Myers Squibb, and Kazia.
A version of this article first appeared on Medscape.com.
What happens to grief when those around you don’t understand it? Where does it go? How do you process it?
Disenfranchised grief, when someone or society more generally doesn’t see a loss as worthy of mourning, can deprive people of experiencing or processing their sadness. This grief, which may be triggered by the death of an ex-spouse, a pet, a failed adoption, can be painful and long-lasting.
Suzanne Cole, MD: ‘I didn’t feel the right to grieve’
During the COVID-19 pandemic, my little sister unexpectedly died. Though she was not one of the nearly 7 million people who died of the virus, in 2021 she became another type of statistic: one of the 109,699 people in the United State who died from a drug overdose. Hers was from fentanyl laced with methamphetamines.
Her death unraveled me. I felt deep guilt that I could not pull her from the sweeping current that had wrenched her from mainstream society into the underbelly of sex work and toward the solace of mind-altering drugs.
But I did not feel the right to grieve for her as I have grieved for other loved ones who were not blamed for their exit from this world. My sister was living a sordid life on the fringes of society. My grief felt invalid, undeserved. Yet, in the eyes of other “upstanding citizens,” her life was not as worth grieving – or so I thought. I tucked my sorrow into a small corner of my soul so no one would see, and I carried on.
To this day, the shame I feel robbed me of the ability to freely talk about her or share the searing pain I feel. Tears still prick my eyes when I think of her, but I have become adept at swallowing them, shaking off the waves of grief as though nothing happened. Even now, I cannot shake the pervasive feeling that my silent tears don’t deserve to be wept.
Don S. Dizon, MD: Working through tragedy
As a medical student, I worked with an outpatient physician as part of a third-year rotation. When we met, the first thing that struck me was how disheveled he looked. His clothes were wrinkled, and his pants were baggy. He took cigarette breaks, which I found disturbing.
But I quickly came to admire him. Despite my first impression, he was the type of doctor I aspired to be. He didn’t need to look at a patient’s chart to recall who they were. He just knew them. He greeted patients warmly, asked about their family. He even remembered the special occasions his patients had mentioned since their past visit. He epitomized empathy and connectedness.
Spending one day in clinic brought to light the challenges of forming such bonds with patients. A man came into the cancer clinic reporting chest pain and was triaged to an exam room. Soon after, the patient was found unresponsive on the floor. Nurses were yelling for help, and the doctor ran in and started CPR while minutes ticked by waiting for an ambulance that could take him to the ED.
By the time help arrived, the patient was blue.
He had died in the clinic in the middle of the day, as the waiting room filled. After the body was taken away, the doctor went into the bathroom. About 20 minutes later, he came out, eyes bloodshot, and continued with the rest of his day, ensuring each patient was seen and cared for.
As a medical student, it hit me how hard it must be to see something so tragic like the end of a life and then continue with your day as if nothing had happened. This is an experience of grief I later came to know well after nearly 30 years treating patients with advanced cancers: compartmentalizing it and carrying on.
A space for grieving: The Schwartz Center Rounds
Disenfranchised grief, the grief that is hard to share and often seems wrong to feel in the first place, can be triggered in many situations. Losing a person others don’t believe deserve to be grieved, such as an abusive partner or someone who committed a crime; losing someone you cared for in a professional role; a loss experienced in a breakup or same-sex partnership, if that relationship was not accepted by one’s family; loss from infertility, miscarriage, stillbirth, or failed adoption; loss that may be taboo or stigmatized, such as deaths via suicide or abortion; and loss of a job, home, or possession that you treasure.
Many of us have had similar situations or will, and the feeling that no one understands the need to mourn can be paralyzing and alienating. In the early days, intense, crushing feelings can cause intrusive, distracting thoughts, and over time, that grief can linger and find a permanent place in our minds.
More and more, though, we are being given opportunities to reflect on these sad moments.
The Schwartz Rounds are an example of such an opportunity. In these rounds, we gather to talk about the experience of caring for people, not the science of medicine.
During one particularly powerful rounds, I spoke to my colleagues about my initial meeting with a patient who was very sick. I detailed the experience of telling her children and her at that initial consult how I thought she was dying and that I did not recommend therapy. I remember how they cried. And I remembered how powerless I felt.
As I recalled that memory during Schwartz Rounds, I could not stop from crying. The unfairness of being a physician meeting someone for the first time and having to tell them such bad news overwhelmed me.
Even more poignant, I had the chance to reconnect with this woman’s children, who were present that day, not as audience members but as participants. Their presence may have brought my emotions to the surface more strongly. In that moment, I could show them the feelings I had bottled up for the sake of professionalism. Ultimately, I felt relieved, freer somehow, as if this burden my soul was carrying had been lifted.
Although we are both grateful for forums like this, these opportunities to share and express the grief we may have hidden away are not as common as they should be.
As physicians, we may express grief by shedding tears at the bedside of a patient nearing the end of life or through the anxiety we feel when our patient suffers a severe reaction to treatment. But we tend to put it away, to go on with our day, because there are others to be seen and cared for and more work to be done. Somehow, we move forward, shedding tears in one room and celebrating victories in another.
We need to create more spaces to express and feel grief, so we don’t get lost in it. Because understanding how grief impacts us, as people and as providers, is one of the most important realizations we can make as we go about our time-honored profession as healers.
Dr. Dizon is the director of women’s cancers at Lifespan Cancer Institute, director of medical oncology at Rhode Island Hospital, and a professor of medicine at Brown University, all in Providence. He reported conflicts of interest with Regeneron, AstraZeneca, Clovis, Bristol-Myers Squibb, and Kazia.
A version of this article first appeared on Medscape.com.



