User login
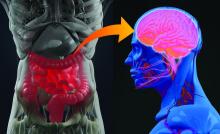
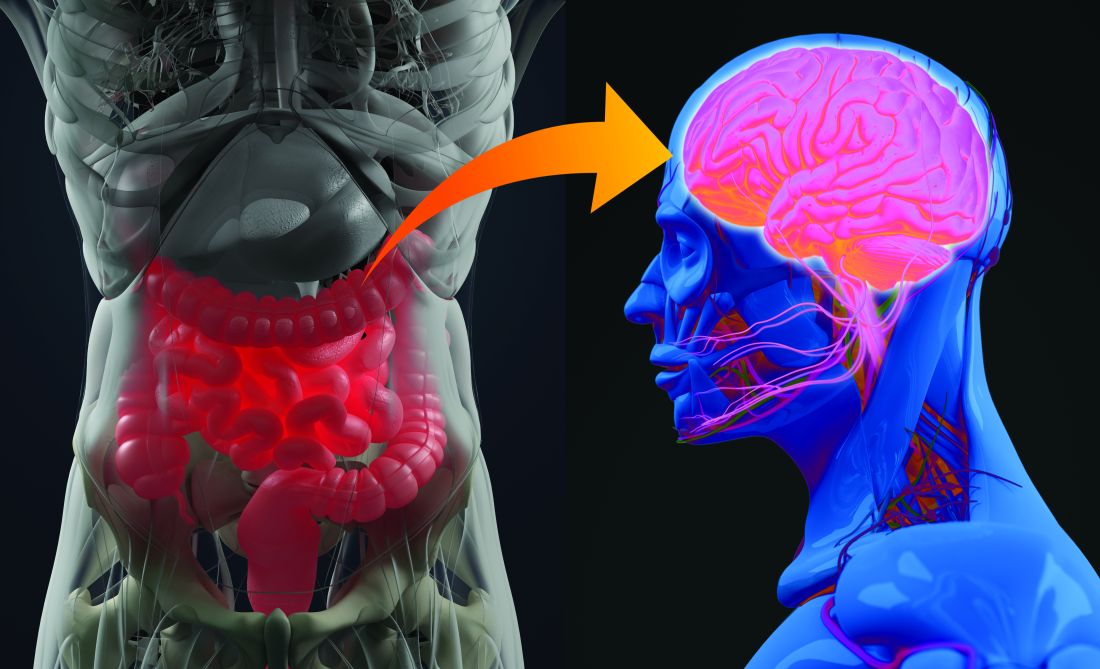
The role of probiotics in mental health
In 1950, at Staten Island’s Sea View Hospital, a group of patients with terminal tuberculosis were given a new antibiotic called isoniazid, which caused some unexpected side effects. The patients reported euphoria, mental stimulation, and improved sleep, and even began socializing with more vigor. The press was all over the case, writing about the sick “dancing in the halls tho’ they had holes in their lungs.” Soon doctors started prescribing isoniazid as the first-ever antidepressant.
The Sea View Hospital experiment was an early hint that changing the composition of the gut microbiome – in this case, via antibiotics – might affect our mental health. Yet only in the last 2 decades has research into connections between what we ingest and psychiatric disorders really taken off. In 2004, a landmark study showed that germ-free mice (born in such sterile conditions that they lacked a microbiome) had an exaggerated stress response. The effects were reversed, however, if the mice were fed a bacterial strain, Bifidobacterium infantis, a probiotic. This sparked academic interest, and thousands of research papers followed.
According to Stephen Ilardi, PhD, a clinical psychologist at the University of Kansas, Lawrence, focusing on the etiology and treatment of depression, now is the “time of exciting discovery” in the field of probiotics and psychiatric disorders, although, admittedly, a lot still remains unknown.
Gut microbiome profiles in mental health disorders
We humans have about 100 trillion microbes residing in our guts. Some of these are archaea, some fungi, some protozoans and even viruses, but most are bacteria. Things like diet, sleep, and stress can all impact the composition of our gut microbiome. When the microbiome differs considerably from the typical, doctors and researchers describe it as dysbiosis, or imbalance. Studies have uncovered dysbiosis in patients with depression, anxiety, schizophrenia, and bipolar disorder.
“I think there is now pretty good evidence that the gut microbiome is actually an important factor in a number of psychiatric disorders,” says Allan Young, MBChB, clinical psychiatrist at King’s College London. The gut microbiome composition does seem to differ between psychiatric patients and the healthy. In depression, for example, a recent review of nine studies found an increase on the genus level in Streptococcus and Oscillibacter and low abundance of Lactobacillus and Coprococcus, among others. In generalized anxiety disorder, meanwhile, there appears to be an increase in Fusobacteria and Escherichia/Shigella .
For Dr. Ilardi, the next important question is whether there are plausible mechanisms that could explain how gut microbiota may influence brain function. And, it appears there are.
“The microbes in the gut can release neurotransmitters into blood that cross into the brain and influence brain function. They can release hormones into the blood that again cross into the brain. They’ve got a lot of tricks up their sleeve,” he says.
One particularly important pathway runs through the vagus nerve – the longest nerve that emerges directly from the brain, connecting it to the gut. Another is the immune pathway. Gut bacteria can interact with immune cells and reduce cytokine production, which in turn can reduce systemic inflammation. Inflammatory processes have been implicated in both depression and bipolar disorder. What’s more, gut microbes can upregulate the expression of a protein called BDNF – brain-derived neurotrophic factor – which helps the development and survival of nerve cells in the brain.
Probiotics’ promise varies for different conditions
As the pathways by which gut dysbiosis may influence psychiatric disorders become clearer, the next logical step is to try to influence the composition of the microbiome to prevent and treat depression, anxiety, or schizophrenia. That’s where probiotics come in.
The evidence for the effects of probiotics – live microorganisms which, when ingested in adequate amounts, confer a health benefit – so far is the strongest for depression, says Viktoriya Nikolova, MRes, MSc, a PhD student and researcher at King’s College London. In their 2021 meta-analysis of seven trials, Mr. Nikolova and colleagues revealed that probiotics can significantly reduce depressive symptoms after just 8 weeks. There was a caveat, however – the probiotics only worked when used in addition to an approved antidepressant. Another meta-analysis, published in 2018, also showed that probiotics, when compared with placebo, improve mood in people with depressive symptoms (here, no antidepressant treatment was necessary).
Roumen Milev, MD, PhD, a neuroscientist at Queen’s University, Kingston, Ont., and coauthor of a review on probiotics and depression published in the Annals of General Psychiatry, warns, however, that the research is still in its infancy. “,” he says.
When it comes to using probiotics to relieve anxiety, “the evidence in the animal literature is really compelling,” says Dr. Ilardi. Human studies are less convincing, however, which Dr. Dr. Ilardi showed in his 2018 review and meta-analysis involving 743 animals and 1,527 humans. “Studies are small for the most part, and some of them aren’t terribly well conducted, and they often use very low doses of probiotics,” he says. One of the larger double-blind and placebo-controlled trials showed that supplementation with Lactobacillus plantarum helps reduce stress and anxiety, while the levels of proinflammatory cytokines go down. Another meta-analysis, published in June, revealed that, when it comes to reducing stress and anxiety in youth, the results are mixed.
Evidence of probiotics’ efficiency in schizophrenia is emerging, yet also limited. A 2019 review concluded that currently available results only “hint” at a possibility that probiotics could make a difference in schizophrenia. Similarly, a 2020 review summed up that the role of probiotics in bipolar disorder “remains unclear and underexplored.”
Better studies, remaining questions
Apart from small samples, one issue with research on probiotics is that they generally tend to use varied doses of different strains of bacteria, or even multistrain mixtures, making it tough to compare results. Although there are hundreds of species of bacteria in the human gut, only a few have been evaluated for their antidepressant or antianxiety effects.
“To make it even worse, it’s almost certainly the case that depending on a person’s actual genetics or maybe their epigenetics, a strain that is helpful for one person may not be helpful for another. There is almost certainly no one-size-fits-all probiotic formulation,” says Dr. Ilardi.
Another critical question that remains to be answered is that of potential side effects.
“Probiotics are often seen as food supplements, so they don’t follow under the same regulations as drugs would,” says Mr. Nikolova. “They don’t necessarily have to follow the pattern of drug trials in many countries, which means that the monitoring of side effects is not the requirement.”
That’s something that worries King’s College psychiatrist Young too. “If you are giving it to modulate how the brain works, you could potentially induce psychiatric symptoms or a psychiatric disorder. There could be allergic reactions. There could be lots of different things,” he says.
When you search the web for “probiotics,” chances are you will come across sites boasting amazing effects that such products can have on cardiovascular heath, the immune system, and yes, mental well-being. Many also sell various probiotic supplements “formulated” for your gut health or improved moods. However, many such commercially available strains have never been actually tested in clinical trials. What’s more, according to Kathrin Cohen Kadosh, PhD, a neuroscientist at University of Surrey (England), “it is not always clear whether the different strains actually reach the gut intact.”
For now, considering the limited research evidence, a safer bet is to try to improve gut health through consumption of fermented foods that naturally contain probiotics, such as miso, kefir, or sauerkraut. Alternatively, you could reach for prebiotics, such as foods containing fiber (prebiotics enhance the growth of beneficial gut microbes). This, Dr. Kadosh says, could be “a gentler way of improving gut health” than popping a pill. Whether an improved mental well-being might follow still remains to be seen.
A version of this article first appeared on Medscape.com.
In 1950, at Staten Island’s Sea View Hospital, a group of patients with terminal tuberculosis were given a new antibiotic called isoniazid, which caused some unexpected side effects. The patients reported euphoria, mental stimulation, and improved sleep, and even began socializing with more vigor. The press was all over the case, writing about the sick “dancing in the halls tho’ they had holes in their lungs.” Soon doctors started prescribing isoniazid as the first-ever antidepressant.
The Sea View Hospital experiment was an early hint that changing the composition of the gut microbiome – in this case, via antibiotics – might affect our mental health. Yet only in the last 2 decades has research into connections between what we ingest and psychiatric disorders really taken off. In 2004, a landmark study showed that germ-free mice (born in such sterile conditions that they lacked a microbiome) had an exaggerated stress response. The effects were reversed, however, if the mice were fed a bacterial strain, Bifidobacterium infantis, a probiotic. This sparked academic interest, and thousands of research papers followed.
According to Stephen Ilardi, PhD, a clinical psychologist at the University of Kansas, Lawrence, focusing on the etiology and treatment of depression, now is the “time of exciting discovery” in the field of probiotics and psychiatric disorders, although, admittedly, a lot still remains unknown.
Gut microbiome profiles in mental health disorders
We humans have about 100 trillion microbes residing in our guts. Some of these are archaea, some fungi, some protozoans and even viruses, but most are bacteria. Things like diet, sleep, and stress can all impact the composition of our gut microbiome. When the microbiome differs considerably from the typical, doctors and researchers describe it as dysbiosis, or imbalance. Studies have uncovered dysbiosis in patients with depression, anxiety, schizophrenia, and bipolar disorder.
“I think there is now pretty good evidence that the gut microbiome is actually an important factor in a number of psychiatric disorders,” says Allan Young, MBChB, clinical psychiatrist at King’s College London. The gut microbiome composition does seem to differ between psychiatric patients and the healthy. In depression, for example, a recent review of nine studies found an increase on the genus level in Streptococcus and Oscillibacter and low abundance of Lactobacillus and Coprococcus, among others. In generalized anxiety disorder, meanwhile, there appears to be an increase in Fusobacteria and Escherichia/Shigella .
For Dr. Ilardi, the next important question is whether there are plausible mechanisms that could explain how gut microbiota may influence brain function. And, it appears there are.
“The microbes in the gut can release neurotransmitters into blood that cross into the brain and influence brain function. They can release hormones into the blood that again cross into the brain. They’ve got a lot of tricks up their sleeve,” he says.
One particularly important pathway runs through the vagus nerve – the longest nerve that emerges directly from the brain, connecting it to the gut. Another is the immune pathway. Gut bacteria can interact with immune cells and reduce cytokine production, which in turn can reduce systemic inflammation. Inflammatory processes have been implicated in both depression and bipolar disorder. What’s more, gut microbes can upregulate the expression of a protein called BDNF – brain-derived neurotrophic factor – which helps the development and survival of nerve cells in the brain.
Probiotics’ promise varies for different conditions
As the pathways by which gut dysbiosis may influence psychiatric disorders become clearer, the next logical step is to try to influence the composition of the microbiome to prevent and treat depression, anxiety, or schizophrenia. That’s where probiotics come in.
The evidence for the effects of probiotics – live microorganisms which, when ingested in adequate amounts, confer a health benefit – so far is the strongest for depression, says Viktoriya Nikolova, MRes, MSc, a PhD student and researcher at King’s College London. In their 2021 meta-analysis of seven trials, Mr. Nikolova and colleagues revealed that probiotics can significantly reduce depressive symptoms after just 8 weeks. There was a caveat, however – the probiotics only worked when used in addition to an approved antidepressant. Another meta-analysis, published in 2018, also showed that probiotics, when compared with placebo, improve mood in people with depressive symptoms (here, no antidepressant treatment was necessary).
Roumen Milev, MD, PhD, a neuroscientist at Queen’s University, Kingston, Ont., and coauthor of a review on probiotics and depression published in the Annals of General Psychiatry, warns, however, that the research is still in its infancy. “,” he says.
When it comes to using probiotics to relieve anxiety, “the evidence in the animal literature is really compelling,” says Dr. Ilardi. Human studies are less convincing, however, which Dr. Dr. Ilardi showed in his 2018 review and meta-analysis involving 743 animals and 1,527 humans. “Studies are small for the most part, and some of them aren’t terribly well conducted, and they often use very low doses of probiotics,” he says. One of the larger double-blind and placebo-controlled trials showed that supplementation with Lactobacillus plantarum helps reduce stress and anxiety, while the levels of proinflammatory cytokines go down. Another meta-analysis, published in June, revealed that, when it comes to reducing stress and anxiety in youth, the results are mixed.
Evidence of probiotics’ efficiency in schizophrenia is emerging, yet also limited. A 2019 review concluded that currently available results only “hint” at a possibility that probiotics could make a difference in schizophrenia. Similarly, a 2020 review summed up that the role of probiotics in bipolar disorder “remains unclear and underexplored.”
Better studies, remaining questions
Apart from small samples, one issue with research on probiotics is that they generally tend to use varied doses of different strains of bacteria, or even multistrain mixtures, making it tough to compare results. Although there are hundreds of species of bacteria in the human gut, only a few have been evaluated for their antidepressant or antianxiety effects.
“To make it even worse, it’s almost certainly the case that depending on a person’s actual genetics or maybe their epigenetics, a strain that is helpful for one person may not be helpful for another. There is almost certainly no one-size-fits-all probiotic formulation,” says Dr. Ilardi.
Another critical question that remains to be answered is that of potential side effects.
“Probiotics are often seen as food supplements, so they don’t follow under the same regulations as drugs would,” says Mr. Nikolova. “They don’t necessarily have to follow the pattern of drug trials in many countries, which means that the monitoring of side effects is not the requirement.”
That’s something that worries King’s College psychiatrist Young too. “If you are giving it to modulate how the brain works, you could potentially induce psychiatric symptoms or a psychiatric disorder. There could be allergic reactions. There could be lots of different things,” he says.
When you search the web for “probiotics,” chances are you will come across sites boasting amazing effects that such products can have on cardiovascular heath, the immune system, and yes, mental well-being. Many also sell various probiotic supplements “formulated” for your gut health or improved moods. However, many such commercially available strains have never been actually tested in clinical trials. What’s more, according to Kathrin Cohen Kadosh, PhD, a neuroscientist at University of Surrey (England), “it is not always clear whether the different strains actually reach the gut intact.”
For now, considering the limited research evidence, a safer bet is to try to improve gut health through consumption of fermented foods that naturally contain probiotics, such as miso, kefir, or sauerkraut. Alternatively, you could reach for prebiotics, such as foods containing fiber (prebiotics enhance the growth of beneficial gut microbes). This, Dr. Kadosh says, could be “a gentler way of improving gut health” than popping a pill. Whether an improved mental well-being might follow still remains to be seen.
A version of this article first appeared on Medscape.com.
In 1950, at Staten Island’s Sea View Hospital, a group of patients with terminal tuberculosis were given a new antibiotic called isoniazid, which caused some unexpected side effects. The patients reported euphoria, mental stimulation, and improved sleep, and even began socializing with more vigor. The press was all over the case, writing about the sick “dancing in the halls tho’ they had holes in their lungs.” Soon doctors started prescribing isoniazid as the first-ever antidepressant.
The Sea View Hospital experiment was an early hint that changing the composition of the gut microbiome – in this case, via antibiotics – might affect our mental health. Yet only in the last 2 decades has research into connections between what we ingest and psychiatric disorders really taken off. In 2004, a landmark study showed that germ-free mice (born in such sterile conditions that they lacked a microbiome) had an exaggerated stress response. The effects were reversed, however, if the mice were fed a bacterial strain, Bifidobacterium infantis, a probiotic. This sparked academic interest, and thousands of research papers followed.
According to Stephen Ilardi, PhD, a clinical psychologist at the University of Kansas, Lawrence, focusing on the etiology and treatment of depression, now is the “time of exciting discovery” in the field of probiotics and psychiatric disorders, although, admittedly, a lot still remains unknown.
Gut microbiome profiles in mental health disorders
We humans have about 100 trillion microbes residing in our guts. Some of these are archaea, some fungi, some protozoans and even viruses, but most are bacteria. Things like diet, sleep, and stress can all impact the composition of our gut microbiome. When the microbiome differs considerably from the typical, doctors and researchers describe it as dysbiosis, or imbalance. Studies have uncovered dysbiosis in patients with depression, anxiety, schizophrenia, and bipolar disorder.
“I think there is now pretty good evidence that the gut microbiome is actually an important factor in a number of psychiatric disorders,” says Allan Young, MBChB, clinical psychiatrist at King’s College London. The gut microbiome composition does seem to differ between psychiatric patients and the healthy. In depression, for example, a recent review of nine studies found an increase on the genus level in Streptococcus and Oscillibacter and low abundance of Lactobacillus and Coprococcus, among others. In generalized anxiety disorder, meanwhile, there appears to be an increase in Fusobacteria and Escherichia/Shigella .
For Dr. Ilardi, the next important question is whether there are plausible mechanisms that could explain how gut microbiota may influence brain function. And, it appears there are.
“The microbes in the gut can release neurotransmitters into blood that cross into the brain and influence brain function. They can release hormones into the blood that again cross into the brain. They’ve got a lot of tricks up their sleeve,” he says.
One particularly important pathway runs through the vagus nerve – the longest nerve that emerges directly from the brain, connecting it to the gut. Another is the immune pathway. Gut bacteria can interact with immune cells and reduce cytokine production, which in turn can reduce systemic inflammation. Inflammatory processes have been implicated in both depression and bipolar disorder. What’s more, gut microbes can upregulate the expression of a protein called BDNF – brain-derived neurotrophic factor – which helps the development and survival of nerve cells in the brain.
Probiotics’ promise varies for different conditions
As the pathways by which gut dysbiosis may influence psychiatric disorders become clearer, the next logical step is to try to influence the composition of the microbiome to prevent and treat depression, anxiety, or schizophrenia. That’s where probiotics come in.
The evidence for the effects of probiotics – live microorganisms which, when ingested in adequate amounts, confer a health benefit – so far is the strongest for depression, says Viktoriya Nikolova, MRes, MSc, a PhD student and researcher at King’s College London. In their 2021 meta-analysis of seven trials, Mr. Nikolova and colleagues revealed that probiotics can significantly reduce depressive symptoms after just 8 weeks. There was a caveat, however – the probiotics only worked when used in addition to an approved antidepressant. Another meta-analysis, published in 2018, also showed that probiotics, when compared with placebo, improve mood in people with depressive symptoms (here, no antidepressant treatment was necessary).
Roumen Milev, MD, PhD, a neuroscientist at Queen’s University, Kingston, Ont., and coauthor of a review on probiotics and depression published in the Annals of General Psychiatry, warns, however, that the research is still in its infancy. “,” he says.
When it comes to using probiotics to relieve anxiety, “the evidence in the animal literature is really compelling,” says Dr. Ilardi. Human studies are less convincing, however, which Dr. Dr. Ilardi showed in his 2018 review and meta-analysis involving 743 animals and 1,527 humans. “Studies are small for the most part, and some of them aren’t terribly well conducted, and they often use very low doses of probiotics,” he says. One of the larger double-blind and placebo-controlled trials showed that supplementation with Lactobacillus plantarum helps reduce stress and anxiety, while the levels of proinflammatory cytokines go down. Another meta-analysis, published in June, revealed that, when it comes to reducing stress and anxiety in youth, the results are mixed.
Evidence of probiotics’ efficiency in schizophrenia is emerging, yet also limited. A 2019 review concluded that currently available results only “hint” at a possibility that probiotics could make a difference in schizophrenia. Similarly, a 2020 review summed up that the role of probiotics in bipolar disorder “remains unclear and underexplored.”
Better studies, remaining questions
Apart from small samples, one issue with research on probiotics is that they generally tend to use varied doses of different strains of bacteria, or even multistrain mixtures, making it tough to compare results. Although there are hundreds of species of bacteria in the human gut, only a few have been evaluated for their antidepressant or antianxiety effects.
“To make it even worse, it’s almost certainly the case that depending on a person’s actual genetics or maybe their epigenetics, a strain that is helpful for one person may not be helpful for another. There is almost certainly no one-size-fits-all probiotic formulation,” says Dr. Ilardi.
Another critical question that remains to be answered is that of potential side effects.
“Probiotics are often seen as food supplements, so they don’t follow under the same regulations as drugs would,” says Mr. Nikolova. “They don’t necessarily have to follow the pattern of drug trials in many countries, which means that the monitoring of side effects is not the requirement.”
That’s something that worries King’s College psychiatrist Young too. “If you are giving it to modulate how the brain works, you could potentially induce psychiatric symptoms or a psychiatric disorder. There could be allergic reactions. There could be lots of different things,” he says.
When you search the web for “probiotics,” chances are you will come across sites boasting amazing effects that such products can have on cardiovascular heath, the immune system, and yes, mental well-being. Many also sell various probiotic supplements “formulated” for your gut health or improved moods. However, many such commercially available strains have never been actually tested in clinical trials. What’s more, according to Kathrin Cohen Kadosh, PhD, a neuroscientist at University of Surrey (England), “it is not always clear whether the different strains actually reach the gut intact.”
For now, considering the limited research evidence, a safer bet is to try to improve gut health through consumption of fermented foods that naturally contain probiotics, such as miso, kefir, or sauerkraut. Alternatively, you could reach for prebiotics, such as foods containing fiber (prebiotics enhance the growth of beneficial gut microbes). This, Dr. Kadosh says, could be “a gentler way of improving gut health” than popping a pill. Whether an improved mental well-being might follow still remains to be seen.
A version of this article first appeared on Medscape.com.
Marijuana and LSD guidance for pediatricians
Seeking novelty is central to adolescence; experimentation is how they explore their identity, exert independence, and establish deep and connected relationships outside of the family. Research over the past 2 decades has demonstrated the neurobiological changes that underpin this increase in sensation seeking. Most adolescents are very good at assessing risk but are willing to tolerate higher levels of risk than adults in the pursuit of novelty.1 If their knowledge base is limited or inaccurate, as is often the case with drugs and alcohol, accepting higher risk becomes more dangerous. Adolescents are more likely to trust their peers than their parents, but their pediatricians still have authority and credibility.
While there is ample credible information online (from the National Institute on Drug Abuse and the Substance Abuse and Mental Health Services Administration’s excellent websites, which can be recommended to teens), marijuana and hallucinogens (LSD and psilocybin) bear special discussion here because of changing legality and their potential medical utility. There is an emerging impression of safety with both; however, policy changes and for-profit marketing may not reflect the actual scientific evidence. You have the opportunity and authority to complicate your patient’s thinking by discussing the evidence supporting their medical utility, and the emerging evidence that both types of drugs may pose special risks for their developing brains.
By June 2021, marijuana was legal for recreational use in 19 states; Washington, D.C.; and Guam, and for “medical use” in 36 states and four territories. Entrepreneurs and activists have made spectacular claims that marijuana is effective for the treatment of everything from insomnia to PTSD, but the reality is less impressive. Of course, marijuana remains a schedule I drug under the federal Controlled Substances Act (1970), which has made it difficult for researchers to perform randomized controlled studies concerning treatment or risks.
However, there are a growing number of randomized controlled trials with synthetic cannabinoids (dronabinol and nabilone) and a (legal) drug derived from cannabis (cannabidiol or CBD, as distinct from the other active ingredient, tetrahydrocannabinol). There is Food and Drug Administration approval for CBD for the treatment of epilepsy in Lennox-Gastaut or Dravet syndrome in patients aged 2 years or younger, and for the synthetic agents for the treatment of chemotherapy-related nausea and vomiting in cancer patients and for the treatment of weight loss and muscle wasting related to HIV/AIDS. That’s it. There is some evidence that these agents may be effective for the treatment of muscle spasticity in multiple sclerosis, chronic pain of many etiologies, Tourette syndrome, insomnia related to multiple sclerosis and chronic pain, and possibly PTSD. But there have been multiple studies that have failed to demonstrate efficacy (or have demonstrated exacerbation) for a host of other medical and psychiatric problems.
While the evidence for marijuana’s medicinal uses is modest, there is substantial evidence that its use in adolescence carries risks. It is an addictive substance and regular use is associated with sustained modest cognitive impairment (a loss of up to eight IQ points in the clinically dependent) and higher rates of anxiety and depressive disorders. As with other substances, use before the age of 18 substantially raises the risk (as much as sevenfold) of developing addiction than the same rate of use in adulthood. The rate of schizophrenia in adolescents with heavy marijuana use is between six and seven times greater than in the general population, whereas similar adult use does not have this association.2,3 Studies in rats have demonstrated that use during adolescence delays and permanently changes the maturation of the prefrontal cortex, an area of the brain that is essential for complex decision-making, sustaining attention, abstract reasoning, and impulse control.4 While we do not fully understand the exact nature of these changes, there is good reason to believe that regular marijuana use in adolescence leads to disruption of critical brain development and cognitive or even psychotic consequences. It is worth noting that the potency of many commercially available marijuana products is much higher than those that were studied, raising the risk and uncertainty further.
Hallucinogens, or “psychedelics” (from Greek for “mind manifesting”) are a class that includes LSD and psilocybin (a chemical found in over 200 species of mushrooms). They precipitate visual and auditory “hallucinations,” a loss of sense of self, and a sense of awe that may be transcendent or frightening. While psilocybin was used by many indigenous cultures in religious ceremonies, LSD was synthesized by a chemist at Sandoz in 1938 and made widely available for study until it was classified as a schedule I drug by the 1970 Controlled Substances Act. They are not addictive. Early research demonstrated promise in the treatment of alcohol dependence and several psychiatric conditions (including other addictions and treatment-resistant depression). Research resumed in 2018, demonstrating promise in the treatment of depression related to terminal illness. Research has also concerned the nature of consciousness and spiritual experiences. Hallucinogens have become popular in certain fields (high tech) as a means of optimizing creativity and performance (“microdosing”). There is modest evidence that use in people with a family history of psychotic illness may precipitate sustained psychotic symptoms. Regular use may further increase the risk of persistent psychosis and adolescent users of multiple substances are at high risk for regular hallucinogen use. Adolescents may think that ketamine, phencyclidine , and 3,4-methylenedioxymethamphetamine are also in this category, although they are different and considerably more risky drugs. Overall, these agents show therapeutic promise, but unless your young patients are facing depression related to a terminal illness and until we learn more from studies, the potential risk to their developing brains outweighs any potential benefits.
Aware of this information, you are ready to ask your adolescent patients about their drug and alcohol use and knowledge. Using phrases like “when did you first try ...” can increase the likelihood that your patients will be forthright with you. Or start by asking about what their friends are trying and talking about. Be curious about any drug and alcohol use at home. Find out what they are curious about, whom they trust, and where they get their information. Then you can offer your information about the dramatic changes happening in their brains (just like the rest of their bodies) and the special risks of drug use during this window of brain development. Acknowledge that the risks of marijuana use in adults may very well be lower than the risks of regular alcohol use but remind them about how their brains are different than those of adults. Delaying use until they are 18 (or ideally in their mid-20s when most brain development is complete), can dramatically lower these risks. For adolescents with a family history of addiction, psychosis, or mood and anxiety disorders, discuss the additional risks that drugs may present to them. And for those adolescents who acknowledge very early (before 13) or heavy use, be curious with them about whether they might be trying to “feel better” and not just “feel good.” Screen them for depression, suicidality, and anxiety disorders. Those underlying problems are treatable, but their course will only worsen with drug and alcohol use. You are in a unique position to help your adolescent patients make wise and well-informed choices and to get them assistance if they need it.
Dr. Swick is physician in chief at Ohana, Center for Child and Adolescent Behavioral Health, Community Hospital of the Monterey (Calif.) Peninsula. Dr. Jellinek is professor emeritus of psychiatry and pediatrics, Harvard Medical School, Boston. Email them at [email protected].
References
1. Romer D. Dev Psychobiol. 2010 Apr;52(3):263-76.
2. Szczepanski SM and Knight TR. Neuron. 2014;83:1002-18.
3. Renard J et al. Front Psychiatry. 2018;9:281.
4. Shen H. Proc Natl Acad Sci U S A. 2020 Jan 7;117(1):7-11.
Seeking novelty is central to adolescence; experimentation is how they explore their identity, exert independence, and establish deep and connected relationships outside of the family. Research over the past 2 decades has demonstrated the neurobiological changes that underpin this increase in sensation seeking. Most adolescents are very good at assessing risk but are willing to tolerate higher levels of risk than adults in the pursuit of novelty.1 If their knowledge base is limited or inaccurate, as is often the case with drugs and alcohol, accepting higher risk becomes more dangerous. Adolescents are more likely to trust their peers than their parents, but their pediatricians still have authority and credibility.
While there is ample credible information online (from the National Institute on Drug Abuse and the Substance Abuse and Mental Health Services Administration’s excellent websites, which can be recommended to teens), marijuana and hallucinogens (LSD and psilocybin) bear special discussion here because of changing legality and their potential medical utility. There is an emerging impression of safety with both; however, policy changes and for-profit marketing may not reflect the actual scientific evidence. You have the opportunity and authority to complicate your patient’s thinking by discussing the evidence supporting their medical utility, and the emerging evidence that both types of drugs may pose special risks for their developing brains.
By June 2021, marijuana was legal for recreational use in 19 states; Washington, D.C.; and Guam, and for “medical use” in 36 states and four territories. Entrepreneurs and activists have made spectacular claims that marijuana is effective for the treatment of everything from insomnia to PTSD, but the reality is less impressive. Of course, marijuana remains a schedule I drug under the federal Controlled Substances Act (1970), which has made it difficult for researchers to perform randomized controlled studies concerning treatment or risks.
However, there are a growing number of randomized controlled trials with synthetic cannabinoids (dronabinol and nabilone) and a (legal) drug derived from cannabis (cannabidiol or CBD, as distinct from the other active ingredient, tetrahydrocannabinol). There is Food and Drug Administration approval for CBD for the treatment of epilepsy in Lennox-Gastaut or Dravet syndrome in patients aged 2 years or younger, and for the synthetic agents for the treatment of chemotherapy-related nausea and vomiting in cancer patients and for the treatment of weight loss and muscle wasting related to HIV/AIDS. That’s it. There is some evidence that these agents may be effective for the treatment of muscle spasticity in multiple sclerosis, chronic pain of many etiologies, Tourette syndrome, insomnia related to multiple sclerosis and chronic pain, and possibly PTSD. But there have been multiple studies that have failed to demonstrate efficacy (or have demonstrated exacerbation) for a host of other medical and psychiatric problems.
While the evidence for marijuana’s medicinal uses is modest, there is substantial evidence that its use in adolescence carries risks. It is an addictive substance and regular use is associated with sustained modest cognitive impairment (a loss of up to eight IQ points in the clinically dependent) and higher rates of anxiety and depressive disorders. As with other substances, use before the age of 18 substantially raises the risk (as much as sevenfold) of developing addiction than the same rate of use in adulthood. The rate of schizophrenia in adolescents with heavy marijuana use is between six and seven times greater than in the general population, whereas similar adult use does not have this association.2,3 Studies in rats have demonstrated that use during adolescence delays and permanently changes the maturation of the prefrontal cortex, an area of the brain that is essential for complex decision-making, sustaining attention, abstract reasoning, and impulse control.4 While we do not fully understand the exact nature of these changes, there is good reason to believe that regular marijuana use in adolescence leads to disruption of critical brain development and cognitive or even psychotic consequences. It is worth noting that the potency of many commercially available marijuana products is much higher than those that were studied, raising the risk and uncertainty further.
Hallucinogens, or “psychedelics” (from Greek for “mind manifesting”) are a class that includes LSD and psilocybin (a chemical found in over 200 species of mushrooms). They precipitate visual and auditory “hallucinations,” a loss of sense of self, and a sense of awe that may be transcendent or frightening. While psilocybin was used by many indigenous cultures in religious ceremonies, LSD was synthesized by a chemist at Sandoz in 1938 and made widely available for study until it was classified as a schedule I drug by the 1970 Controlled Substances Act. They are not addictive. Early research demonstrated promise in the treatment of alcohol dependence and several psychiatric conditions (including other addictions and treatment-resistant depression). Research resumed in 2018, demonstrating promise in the treatment of depression related to terminal illness. Research has also concerned the nature of consciousness and spiritual experiences. Hallucinogens have become popular in certain fields (high tech) as a means of optimizing creativity and performance (“microdosing”). There is modest evidence that use in people with a family history of psychotic illness may precipitate sustained psychotic symptoms. Regular use may further increase the risk of persistent psychosis and adolescent users of multiple substances are at high risk for regular hallucinogen use. Adolescents may think that ketamine, phencyclidine , and 3,4-methylenedioxymethamphetamine are also in this category, although they are different and considerably more risky drugs. Overall, these agents show therapeutic promise, but unless your young patients are facing depression related to a terminal illness and until we learn more from studies, the potential risk to their developing brains outweighs any potential benefits.
Aware of this information, you are ready to ask your adolescent patients about their drug and alcohol use and knowledge. Using phrases like “when did you first try ...” can increase the likelihood that your patients will be forthright with you. Or start by asking about what their friends are trying and talking about. Be curious about any drug and alcohol use at home. Find out what they are curious about, whom they trust, and where they get their information. Then you can offer your information about the dramatic changes happening in their brains (just like the rest of their bodies) and the special risks of drug use during this window of brain development. Acknowledge that the risks of marijuana use in adults may very well be lower than the risks of regular alcohol use but remind them about how their brains are different than those of adults. Delaying use until they are 18 (or ideally in their mid-20s when most brain development is complete), can dramatically lower these risks. For adolescents with a family history of addiction, psychosis, or mood and anxiety disorders, discuss the additional risks that drugs may present to them. And for those adolescents who acknowledge very early (before 13) or heavy use, be curious with them about whether they might be trying to “feel better” and not just “feel good.” Screen them for depression, suicidality, and anxiety disorders. Those underlying problems are treatable, but their course will only worsen with drug and alcohol use. You are in a unique position to help your adolescent patients make wise and well-informed choices and to get them assistance if they need it.
Dr. Swick is physician in chief at Ohana, Center for Child and Adolescent Behavioral Health, Community Hospital of the Monterey (Calif.) Peninsula. Dr. Jellinek is professor emeritus of psychiatry and pediatrics, Harvard Medical School, Boston. Email them at [email protected].
References
1. Romer D. Dev Psychobiol. 2010 Apr;52(3):263-76.
2. Szczepanski SM and Knight TR. Neuron. 2014;83:1002-18.
3. Renard J et al. Front Psychiatry. 2018;9:281.
4. Shen H. Proc Natl Acad Sci U S A. 2020 Jan 7;117(1):7-11.
Seeking novelty is central to adolescence; experimentation is how they explore their identity, exert independence, and establish deep and connected relationships outside of the family. Research over the past 2 decades has demonstrated the neurobiological changes that underpin this increase in sensation seeking. Most adolescents are very good at assessing risk but are willing to tolerate higher levels of risk than adults in the pursuit of novelty.1 If their knowledge base is limited or inaccurate, as is often the case with drugs and alcohol, accepting higher risk becomes more dangerous. Adolescents are more likely to trust their peers than their parents, but their pediatricians still have authority and credibility.
While there is ample credible information online (from the National Institute on Drug Abuse and the Substance Abuse and Mental Health Services Administration’s excellent websites, which can be recommended to teens), marijuana and hallucinogens (LSD and psilocybin) bear special discussion here because of changing legality and their potential medical utility. There is an emerging impression of safety with both; however, policy changes and for-profit marketing may not reflect the actual scientific evidence. You have the opportunity and authority to complicate your patient’s thinking by discussing the evidence supporting their medical utility, and the emerging evidence that both types of drugs may pose special risks for their developing brains.
By June 2021, marijuana was legal for recreational use in 19 states; Washington, D.C.; and Guam, and for “medical use” in 36 states and four territories. Entrepreneurs and activists have made spectacular claims that marijuana is effective for the treatment of everything from insomnia to PTSD, but the reality is less impressive. Of course, marijuana remains a schedule I drug under the federal Controlled Substances Act (1970), which has made it difficult for researchers to perform randomized controlled studies concerning treatment or risks.
However, there are a growing number of randomized controlled trials with synthetic cannabinoids (dronabinol and nabilone) and a (legal) drug derived from cannabis (cannabidiol or CBD, as distinct from the other active ingredient, tetrahydrocannabinol). There is Food and Drug Administration approval for CBD for the treatment of epilepsy in Lennox-Gastaut or Dravet syndrome in patients aged 2 years or younger, and for the synthetic agents for the treatment of chemotherapy-related nausea and vomiting in cancer patients and for the treatment of weight loss and muscle wasting related to HIV/AIDS. That’s it. There is some evidence that these agents may be effective for the treatment of muscle spasticity in multiple sclerosis, chronic pain of many etiologies, Tourette syndrome, insomnia related to multiple sclerosis and chronic pain, and possibly PTSD. But there have been multiple studies that have failed to demonstrate efficacy (or have demonstrated exacerbation) for a host of other medical and psychiatric problems.
While the evidence for marijuana’s medicinal uses is modest, there is substantial evidence that its use in adolescence carries risks. It is an addictive substance and regular use is associated with sustained modest cognitive impairment (a loss of up to eight IQ points in the clinically dependent) and higher rates of anxiety and depressive disorders. As with other substances, use before the age of 18 substantially raises the risk (as much as sevenfold) of developing addiction than the same rate of use in adulthood. The rate of schizophrenia in adolescents with heavy marijuana use is between six and seven times greater than in the general population, whereas similar adult use does not have this association.2,3 Studies in rats have demonstrated that use during adolescence delays and permanently changes the maturation of the prefrontal cortex, an area of the brain that is essential for complex decision-making, sustaining attention, abstract reasoning, and impulse control.4 While we do not fully understand the exact nature of these changes, there is good reason to believe that regular marijuana use in adolescence leads to disruption of critical brain development and cognitive or even psychotic consequences. It is worth noting that the potency of many commercially available marijuana products is much higher than those that were studied, raising the risk and uncertainty further.
Hallucinogens, or “psychedelics” (from Greek for “mind manifesting”) are a class that includes LSD and psilocybin (a chemical found in over 200 species of mushrooms). They precipitate visual and auditory “hallucinations,” a loss of sense of self, and a sense of awe that may be transcendent or frightening. While psilocybin was used by many indigenous cultures in religious ceremonies, LSD was synthesized by a chemist at Sandoz in 1938 and made widely available for study until it was classified as a schedule I drug by the 1970 Controlled Substances Act. They are not addictive. Early research demonstrated promise in the treatment of alcohol dependence and several psychiatric conditions (including other addictions and treatment-resistant depression). Research resumed in 2018, demonstrating promise in the treatment of depression related to terminal illness. Research has also concerned the nature of consciousness and spiritual experiences. Hallucinogens have become popular in certain fields (high tech) as a means of optimizing creativity and performance (“microdosing”). There is modest evidence that use in people with a family history of psychotic illness may precipitate sustained psychotic symptoms. Regular use may further increase the risk of persistent psychosis and adolescent users of multiple substances are at high risk for regular hallucinogen use. Adolescents may think that ketamine, phencyclidine , and 3,4-methylenedioxymethamphetamine are also in this category, although they are different and considerably more risky drugs. Overall, these agents show therapeutic promise, but unless your young patients are facing depression related to a terminal illness and until we learn more from studies, the potential risk to their developing brains outweighs any potential benefits.
Aware of this information, you are ready to ask your adolescent patients about their drug and alcohol use and knowledge. Using phrases like “when did you first try ...” can increase the likelihood that your patients will be forthright with you. Or start by asking about what their friends are trying and talking about. Be curious about any drug and alcohol use at home. Find out what they are curious about, whom they trust, and where they get their information. Then you can offer your information about the dramatic changes happening in their brains (just like the rest of their bodies) and the special risks of drug use during this window of brain development. Acknowledge that the risks of marijuana use in adults may very well be lower than the risks of regular alcohol use but remind them about how their brains are different than those of adults. Delaying use until they are 18 (or ideally in their mid-20s when most brain development is complete), can dramatically lower these risks. For adolescents with a family history of addiction, psychosis, or mood and anxiety disorders, discuss the additional risks that drugs may present to them. And for those adolescents who acknowledge very early (before 13) or heavy use, be curious with them about whether they might be trying to “feel better” and not just “feel good.” Screen them for depression, suicidality, and anxiety disorders. Those underlying problems are treatable, but their course will only worsen with drug and alcohol use. You are in a unique position to help your adolescent patients make wise and well-informed choices and to get them assistance if they need it.
Dr. Swick is physician in chief at Ohana, Center for Child and Adolescent Behavioral Health, Community Hospital of the Monterey (Calif.) Peninsula. Dr. Jellinek is professor emeritus of psychiatry and pediatrics, Harvard Medical School, Boston. Email them at [email protected].
References
1. Romer D. Dev Psychobiol. 2010 Apr;52(3):263-76.
2. Szczepanski SM and Knight TR. Neuron. 2014;83:1002-18.
3. Renard J et al. Front Psychiatry. 2018;9:281.
4. Shen H. Proc Natl Acad Sci U S A. 2020 Jan 7;117(1):7-11.
Premature menopause a ‘warning sign’ for greater ASCVD risk
Premature menopause is well known to be linked to cardiovascular disease in women, but it may not carry as much weight as more traditional cardiovascular risk factors in determining a patient’s 10-year risk of having a heart attack or stroke in this population, a cohort study that evaluated the veracity of premature menopause found.
Premature menopause can serve as a “marker or warning sign” that cardiologists should pay closer attention to traditional atherosclerotic cardiovascular disease (ASCVD) risk factors, lead study author Sadiya S. Khan, MD, MS, said in an interview. “When we looked at the addition of premature menopause into the risk-prediction equation, we did not see that it meaningfully improved the ability of the risk predictions of pooled cohort equations [PCEs] to identify who developed cardiovascular disease,” said Dr. Khan, a cardiologist at Northwestern University, Chicago.
The cohort study included 5,466 Black women and 10,584 White women from seven U.S. population-based cohorts, including the Women’s Health Initiative, of whom 951 and 1,039, respectively, self-reported early menopause. The cohort study researchers noted that the 2019 American College of Cardiology/American Heart Association guideline for prevention of CVD acknowledged premature menopause as risk-enhancing factor in the CVD assessment in women younger than 40.
The cohort study found that Black women had almost twice the rate of premature menopause than White women, 17.4% and 9.8%, respectively. And it found that premature menopause was significantly linked with ASCVD in both populations independent of traditional risk factors – a 24% greater risk for Black women and 28% greater risk for White women.
‘Surprising’ finding
However, when premature menopause was added to the pooled cohort equations per the 2013 ACC/AHA guideline, the researchers found no incremental benefit, a finding Dr. Khan called “really surprising to us.”
She added, “If we look at the differences in the characteristics of women who have premature menopause, compared with those who didn’t, there were slight differences in terms of higher blood pressure, higher body mass index, and slightly higher glucose. So maybe what we’re seeing – and this is more speculative – is that risk factors are developing after early menopause, and the focus should be earlier in the patient’s life course to try to prevent hypertension, diabetes, and obesity.”
Dr. Khan emphasized that the findings don’t obviate the value of premature menopause in assessing ASCVD risk in women. “We still know that this is an important marker for women and their risk for heart disease, and it should be a warning sign to pay close attention to those other risk factors and what other preventive measures can be taken,” she said.
Christie Ballantyne, MD, said it’s important to note that the study did not dismiss the relevance of premature menopause in shared decision-making for postmenopausal women. “It certainly doesn’t mean that premature menopause is not a risk,” Dr. Ballantyne said in an interview. “Premature menopause may cause a worsening of traditional CVD risk factors, so that’s one possible explanation for it. The other possible explanation is that women with worse ASCVD risk factors – who are more overweight, have higher blood pressure, and have more diabetes and insulin resistance – are more likely to have earlier menopause.” Dr. Ballantyne is chief of cardiology at Baylor College of Medicine and director of cardiovascular disease prevention at Methodist DeBakey Heart Center, both in Houston.
“You should still look very carefully at the patient’s risk factors, calculate the pooled cohort equations, and make sure you get a recommendation,” he said. “If their risks are up, give recommendations on how to improve diet and exercise. Consider if you need to treat lipids or treat blood pressure with more than diet and exercise because there’s nothing magical about 7.5%”, the threshold for lipid-lowering therapy in the ASCVD risk calculator.
Dr. Khan and coauthors disclosed receiving grants from the National Institutes of Health and the American Heart Association. One coauthor reported a financial relationship with HGM Biopharmaceuticals. Dr. Ballantyne is a lead investigator of the Atherosclerosis Risk in Communities study, one of the population-based cohorts used in the cohort study. He has no other relevant relationships to disclose.
Premature menopause is well known to be linked to cardiovascular disease in women, but it may not carry as much weight as more traditional cardiovascular risk factors in determining a patient’s 10-year risk of having a heart attack or stroke in this population, a cohort study that evaluated the veracity of premature menopause found.
Premature menopause can serve as a “marker or warning sign” that cardiologists should pay closer attention to traditional atherosclerotic cardiovascular disease (ASCVD) risk factors, lead study author Sadiya S. Khan, MD, MS, said in an interview. “When we looked at the addition of premature menopause into the risk-prediction equation, we did not see that it meaningfully improved the ability of the risk predictions of pooled cohort equations [PCEs] to identify who developed cardiovascular disease,” said Dr. Khan, a cardiologist at Northwestern University, Chicago.
The cohort study included 5,466 Black women and 10,584 White women from seven U.S. population-based cohorts, including the Women’s Health Initiative, of whom 951 and 1,039, respectively, self-reported early menopause. The cohort study researchers noted that the 2019 American College of Cardiology/American Heart Association guideline for prevention of CVD acknowledged premature menopause as risk-enhancing factor in the CVD assessment in women younger than 40.
The cohort study found that Black women had almost twice the rate of premature menopause than White women, 17.4% and 9.8%, respectively. And it found that premature menopause was significantly linked with ASCVD in both populations independent of traditional risk factors – a 24% greater risk for Black women and 28% greater risk for White women.
‘Surprising’ finding
However, when premature menopause was added to the pooled cohort equations per the 2013 ACC/AHA guideline, the researchers found no incremental benefit, a finding Dr. Khan called “really surprising to us.”
She added, “If we look at the differences in the characteristics of women who have premature menopause, compared with those who didn’t, there were slight differences in terms of higher blood pressure, higher body mass index, and slightly higher glucose. So maybe what we’re seeing – and this is more speculative – is that risk factors are developing after early menopause, and the focus should be earlier in the patient’s life course to try to prevent hypertension, diabetes, and obesity.”
Dr. Khan emphasized that the findings don’t obviate the value of premature menopause in assessing ASCVD risk in women. “We still know that this is an important marker for women and their risk for heart disease, and it should be a warning sign to pay close attention to those other risk factors and what other preventive measures can be taken,” she said.
Christie Ballantyne, MD, said it’s important to note that the study did not dismiss the relevance of premature menopause in shared decision-making for postmenopausal women. “It certainly doesn’t mean that premature menopause is not a risk,” Dr. Ballantyne said in an interview. “Premature menopause may cause a worsening of traditional CVD risk factors, so that’s one possible explanation for it. The other possible explanation is that women with worse ASCVD risk factors – who are more overweight, have higher blood pressure, and have more diabetes and insulin resistance – are more likely to have earlier menopause.” Dr. Ballantyne is chief of cardiology at Baylor College of Medicine and director of cardiovascular disease prevention at Methodist DeBakey Heart Center, both in Houston.
“You should still look very carefully at the patient’s risk factors, calculate the pooled cohort equations, and make sure you get a recommendation,” he said. “If their risks are up, give recommendations on how to improve diet and exercise. Consider if you need to treat lipids or treat blood pressure with more than diet and exercise because there’s nothing magical about 7.5%”, the threshold for lipid-lowering therapy in the ASCVD risk calculator.
Dr. Khan and coauthors disclosed receiving grants from the National Institutes of Health and the American Heart Association. One coauthor reported a financial relationship with HGM Biopharmaceuticals. Dr. Ballantyne is a lead investigator of the Atherosclerosis Risk in Communities study, one of the population-based cohorts used in the cohort study. He has no other relevant relationships to disclose.
Premature menopause is well known to be linked to cardiovascular disease in women, but it may not carry as much weight as more traditional cardiovascular risk factors in determining a patient’s 10-year risk of having a heart attack or stroke in this population, a cohort study that evaluated the veracity of premature menopause found.
Premature menopause can serve as a “marker or warning sign” that cardiologists should pay closer attention to traditional atherosclerotic cardiovascular disease (ASCVD) risk factors, lead study author Sadiya S. Khan, MD, MS, said in an interview. “When we looked at the addition of premature menopause into the risk-prediction equation, we did not see that it meaningfully improved the ability of the risk predictions of pooled cohort equations [PCEs] to identify who developed cardiovascular disease,” said Dr. Khan, a cardiologist at Northwestern University, Chicago.
The cohort study included 5,466 Black women and 10,584 White women from seven U.S. population-based cohorts, including the Women’s Health Initiative, of whom 951 and 1,039, respectively, self-reported early menopause. The cohort study researchers noted that the 2019 American College of Cardiology/American Heart Association guideline for prevention of CVD acknowledged premature menopause as risk-enhancing factor in the CVD assessment in women younger than 40.
The cohort study found that Black women had almost twice the rate of premature menopause than White women, 17.4% and 9.8%, respectively. And it found that premature menopause was significantly linked with ASCVD in both populations independent of traditional risk factors – a 24% greater risk for Black women and 28% greater risk for White women.
‘Surprising’ finding
However, when premature menopause was added to the pooled cohort equations per the 2013 ACC/AHA guideline, the researchers found no incremental benefit, a finding Dr. Khan called “really surprising to us.”
She added, “If we look at the differences in the characteristics of women who have premature menopause, compared with those who didn’t, there were slight differences in terms of higher blood pressure, higher body mass index, and slightly higher glucose. So maybe what we’re seeing – and this is more speculative – is that risk factors are developing after early menopause, and the focus should be earlier in the patient’s life course to try to prevent hypertension, diabetes, and obesity.”
Dr. Khan emphasized that the findings don’t obviate the value of premature menopause in assessing ASCVD risk in women. “We still know that this is an important marker for women and their risk for heart disease, and it should be a warning sign to pay close attention to those other risk factors and what other preventive measures can be taken,” she said.
Christie Ballantyne, MD, said it’s important to note that the study did not dismiss the relevance of premature menopause in shared decision-making for postmenopausal women. “It certainly doesn’t mean that premature menopause is not a risk,” Dr. Ballantyne said in an interview. “Premature menopause may cause a worsening of traditional CVD risk factors, so that’s one possible explanation for it. The other possible explanation is that women with worse ASCVD risk factors – who are more overweight, have higher blood pressure, and have more diabetes and insulin resistance – are more likely to have earlier menopause.” Dr. Ballantyne is chief of cardiology at Baylor College of Medicine and director of cardiovascular disease prevention at Methodist DeBakey Heart Center, both in Houston.
“You should still look very carefully at the patient’s risk factors, calculate the pooled cohort equations, and make sure you get a recommendation,” he said. “If their risks are up, give recommendations on how to improve diet and exercise. Consider if you need to treat lipids or treat blood pressure with more than diet and exercise because there’s nothing magical about 7.5%”, the threshold for lipid-lowering therapy in the ASCVD risk calculator.
Dr. Khan and coauthors disclosed receiving grants from the National Institutes of Health and the American Heart Association. One coauthor reported a financial relationship with HGM Biopharmaceuticals. Dr. Ballantyne is a lead investigator of the Atherosclerosis Risk in Communities study, one of the population-based cohorts used in the cohort study. He has no other relevant relationships to disclose.
FROM JAMA CARDIOLOGY
A pediatrician notices empty fields
The high school football team here in Brunswick has had winning years and losing years but the school has always fielded a competitive team. It has been state champion on several occasions and has weathered the challenge when soccer became the new and more popular sport shortly after it arrived in town several decades ago. But this year, on the heels of a strong winning season last year, the numbers are down significantly. The school is in jeopardy of not having enough players to field a junior varsity team.
This dearth of student athletes is a problem not just here in Brunswick. Schools across the state of Maine are being forced to shift to an eight man football format. Nor is it unique to football here in vacationland. A recent article in a Hudson Valley, N.Y., newspaper chronicles a broad-based decline in participation in high school sports including field hockey, tennis, and cross country (‘Covid,’ The Journal News, Nancy Haggerty, Sept. 5, 2021). In many situations the school may have enough players to field a varsity team but too few to play a junior varsity schedule. Without a supply of young talent coming up from the junior varsity, the future of any varsity program is on a shaky legs. Some of the coaches are referring to the decline in participation as a “COVID hangover” triggered in part by season disruptions, cancellations, and fluctuating remote learning formats.
I and some other coaches argue that the participation drought predates the pandemic and is the result of a wide range of unfortunate trends. First, is the general malaise and don’t-give-a-damn-about-anything attitude that has settled on the young people of this country, the causes of which are difficult to define. It may be that after years of sitting in front of a video screen, too many children have settled into the role of being spectators and find the energy it takes to participate just isn’t worth the effort.
Another contributor to the decline in participation is the heavy of emphasis on early specialization. Driven in many cases by unrealistic parental dreams, children are shepherded into elite travel teams with seasons that often stretch to lengths that make it difficult if not impossible for a child to participate in other sports. The child who may simply be a late bloomer or whose family can’t afford the time or money to buy into the travel team ethic quickly finds himself losing ground. Without the additional opportunities for skill development, many of the children noon travel teams eventually wonder if it is worth trying to catch up. Ironically, the trend toward early specialization is short-sighted because many college and professional coaches report that their best athletes shunned becoming one-trick ponies and played a variety of sports growing up.
Parental concerns about injury, particularly concussion, probably play a role in the trend of falling participation in sports, even those with minimal risk of head injury. Certainly our new awareness of the long-term effects of multiple concussions is long overdue. However, we as pediatricians must take some of the blame for often emphasizing the injury risk inherent in sports in general while neglecting to highlight the positive benefits of competitive sports such as fitness and team building. Are there situations where our emphasis on preparticipation physicals is acting as a deterrent?
There are exceptions to the general trend of falling participation, lacrosse being the most obvious example. However, as lacrosse becomes more popular across the country there are signs that it is already drifting into the larger and counterproductive elite travel team model. There have always been communities in which an individual coach or parent has created a team culture that is both inclusive and competitive. The two are not mutually exclusive.
Sadly, these exceptional programs are few and far between. I’m not sure where we can start to turn things around so that more children choose to be players rather than observers. But, we pediatricians certainly can play a more positive role in emphasizing the benefits of team play.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected].
The high school football team here in Brunswick has had winning years and losing years but the school has always fielded a competitive team. It has been state champion on several occasions and has weathered the challenge when soccer became the new and more popular sport shortly after it arrived in town several decades ago. But this year, on the heels of a strong winning season last year, the numbers are down significantly. The school is in jeopardy of not having enough players to field a junior varsity team.
This dearth of student athletes is a problem not just here in Brunswick. Schools across the state of Maine are being forced to shift to an eight man football format. Nor is it unique to football here in vacationland. A recent article in a Hudson Valley, N.Y., newspaper chronicles a broad-based decline in participation in high school sports including field hockey, tennis, and cross country (‘Covid,’ The Journal News, Nancy Haggerty, Sept. 5, 2021). In many situations the school may have enough players to field a varsity team but too few to play a junior varsity schedule. Without a supply of young talent coming up from the junior varsity, the future of any varsity program is on a shaky legs. Some of the coaches are referring to the decline in participation as a “COVID hangover” triggered in part by season disruptions, cancellations, and fluctuating remote learning formats.
I and some other coaches argue that the participation drought predates the pandemic and is the result of a wide range of unfortunate trends. First, is the general malaise and don’t-give-a-damn-about-anything attitude that has settled on the young people of this country, the causes of which are difficult to define. It may be that after years of sitting in front of a video screen, too many children have settled into the role of being spectators and find the energy it takes to participate just isn’t worth the effort.
Another contributor to the decline in participation is the heavy of emphasis on early specialization. Driven in many cases by unrealistic parental dreams, children are shepherded into elite travel teams with seasons that often stretch to lengths that make it difficult if not impossible for a child to participate in other sports. The child who may simply be a late bloomer or whose family can’t afford the time or money to buy into the travel team ethic quickly finds himself losing ground. Without the additional opportunities for skill development, many of the children noon travel teams eventually wonder if it is worth trying to catch up. Ironically, the trend toward early specialization is short-sighted because many college and professional coaches report that their best athletes shunned becoming one-trick ponies and played a variety of sports growing up.
Parental concerns about injury, particularly concussion, probably play a role in the trend of falling participation in sports, even those with minimal risk of head injury. Certainly our new awareness of the long-term effects of multiple concussions is long overdue. However, we as pediatricians must take some of the blame for often emphasizing the injury risk inherent in sports in general while neglecting to highlight the positive benefits of competitive sports such as fitness and team building. Are there situations where our emphasis on preparticipation physicals is acting as a deterrent?
There are exceptions to the general trend of falling participation, lacrosse being the most obvious example. However, as lacrosse becomes more popular across the country there are signs that it is already drifting into the larger and counterproductive elite travel team model. There have always been communities in which an individual coach or parent has created a team culture that is both inclusive and competitive. The two are not mutually exclusive.
Sadly, these exceptional programs are few and far between. I’m not sure where we can start to turn things around so that more children choose to be players rather than observers. But, we pediatricians certainly can play a more positive role in emphasizing the benefits of team play.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected].
The high school football team here in Brunswick has had winning years and losing years but the school has always fielded a competitive team. It has been state champion on several occasions and has weathered the challenge when soccer became the new and more popular sport shortly after it arrived in town several decades ago. But this year, on the heels of a strong winning season last year, the numbers are down significantly. The school is in jeopardy of not having enough players to field a junior varsity team.
This dearth of student athletes is a problem not just here in Brunswick. Schools across the state of Maine are being forced to shift to an eight man football format. Nor is it unique to football here in vacationland. A recent article in a Hudson Valley, N.Y., newspaper chronicles a broad-based decline in participation in high school sports including field hockey, tennis, and cross country (‘Covid,’ The Journal News, Nancy Haggerty, Sept. 5, 2021). In many situations the school may have enough players to field a varsity team but too few to play a junior varsity schedule. Without a supply of young talent coming up from the junior varsity, the future of any varsity program is on a shaky legs. Some of the coaches are referring to the decline in participation as a “COVID hangover” triggered in part by season disruptions, cancellations, and fluctuating remote learning formats.
I and some other coaches argue that the participation drought predates the pandemic and is the result of a wide range of unfortunate trends. First, is the general malaise and don’t-give-a-damn-about-anything attitude that has settled on the young people of this country, the causes of which are difficult to define. It may be that after years of sitting in front of a video screen, too many children have settled into the role of being spectators and find the energy it takes to participate just isn’t worth the effort.
Another contributor to the decline in participation is the heavy of emphasis on early specialization. Driven in many cases by unrealistic parental dreams, children are shepherded into elite travel teams with seasons that often stretch to lengths that make it difficult if not impossible for a child to participate in other sports. The child who may simply be a late bloomer or whose family can’t afford the time or money to buy into the travel team ethic quickly finds himself losing ground. Without the additional opportunities for skill development, many of the children noon travel teams eventually wonder if it is worth trying to catch up. Ironically, the trend toward early specialization is short-sighted because many college and professional coaches report that their best athletes shunned becoming one-trick ponies and played a variety of sports growing up.
Parental concerns about injury, particularly concussion, probably play a role in the trend of falling participation in sports, even those with minimal risk of head injury. Certainly our new awareness of the long-term effects of multiple concussions is long overdue. However, we as pediatricians must take some of the blame for often emphasizing the injury risk inherent in sports in general while neglecting to highlight the positive benefits of competitive sports such as fitness and team building. Are there situations where our emphasis on preparticipation physicals is acting as a deterrent?
There are exceptions to the general trend of falling participation, lacrosse being the most obvious example. However, as lacrosse becomes more popular across the country there are signs that it is already drifting into the larger and counterproductive elite travel team model. There have always been communities in which an individual coach or parent has created a team culture that is both inclusive and competitive. The two are not mutually exclusive.
Sadly, these exceptional programs are few and far between. I’m not sure where we can start to turn things around so that more children choose to be players rather than observers. But, we pediatricians certainly can play a more positive role in emphasizing the benefits of team play.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected].
Assessing headache severity via migraine symptoms can help predict outcomes
according to an analysis of data from thousands of headache sufferers who recorded variables like pain and duration in a daily digital diary.
“Our hope is that this work serves as foundational basis for better understanding the complexity of headache as a symptom-based condition,” James S. McGinley, PhD, of Vector Psychometric Group in Chapel Hill, N.C., and coauthors wrote. The study was published in Cephalalgia.
To evaluate whether keeping track of daily headache features can produce a useful, predictive score, the researchers reviewed data from migraine patients that were collected via N1‑Headache, a commercial digital health platform. Ultimately, information from 4,380 adults with a self-reported migraine diagnosis was analyzed; the sample was 90% female and their mean age was 37 years. Study participants reported an average of 33 headaches per month over the last 3 months. Nine patient-reported variables were initially considered in calculating the Headache Day Severity (HDS) score: pain intensity, headache duration, aura, pulsating/throbbing pain, unilateral pain, pain aggravation by activity, nausea/vomiting, photophobia, and phonophobia.
After determining that unilateral pain was not a meaningful variable, the researchers’ model found that, for every 1 standard deviation increase in HDS, the patient’s odds of physician visit increased by 71% (odds ratio, 1.71; 95% confidence interval, 1.32-2.21) and the odds of an ED visit increased by 342% (OR, 4.42; 95% CI, 2.23-7.60). They also found that the likelihood of missed work or school increased by 190% (OR, 2.90; 95% CI, 2.56-3.29), the chances of missing household work increased by 237% (OR, 3.37; 95% CI, 3.06-3.72) and the odds of missing other leisure or social activity increased by 228% (OR, 3.28; 95% CI, 2.97-3.64).
Tracking multiple variables
“We encourage all of our patients to monitor their headaches; there are just too many variables to try to keep it in your head,” Robert Cowan, MD, professor of neurology and chief of the division of headache medicine at Stanford (Calif.) University, said in an interview. He referenced a previous study from the University of Washington where patients were asked to track their headaches; that data was then compared against their self-reported headaches at a quarterly physician visit.
“What they found was there was absolutely no correlation with reported frequency of headache at the visit and what was seen in the tracker,” he said. “If patients had a headache in the previous 3 days before their visit, they felt that their headaches were poorly controlled. If they hadn’t, they thought their headaches were under good control. So the value of tracking is pretty clear.”
He added that, while not every headache sufferer needs to track their daily routines and symptoms, once those symptoms interfere with your life on a day-to-day basis, it’s probably time to consider keeping tabs on yourself with a tool of some sort. And while this study’s calculated HDS score supports the idea of migraine’s complexity, it also leaves unanswered the question of how to treat patients with severe symptoms.
“Frequently,” he said, “we’ll see patients who say: ‘I can deal with the pain, but the nausea makes it impossible to work, or the light sensitivity makes it impossible to go outside.’ The big question within the headache community is, can you treat migraine and have it address the whole spectrum, from dizziness to light sensitivity to sound sensitivity to vertigo, or should you be going after individual symptoms? That’s a controversy that rages on; I think most of us go for a combination. We’re in a polypharmacy phase: ‘If nausea is a big problem, take this, but we also try to prevent the whole migraine complex, so take this as well.’ ”
The authors acknowledged their study’s limitations, including the inability to determine how many participants’ migraines were formally diagnosed by a trained medical professional and the lack of generalizability of data from a convenience sample, though they added that patients who independently track their own headaches “may be representative of those who would participate in a clinical trial.” In addition, as seven of the nine features were collected in N1‑Headache on a yes/no scale, they recognized that “increasing the number of response options for each item may improve our ability to measure HDS.”
The study was funded by Amgen through the Competitive Grant Program in Migraine Research. The authors declared several potential conflicts of interest, including receiving funding, research support, salary, and honoraria from various pharmaceutical companies.
according to an analysis of data from thousands of headache sufferers who recorded variables like pain and duration in a daily digital diary.
“Our hope is that this work serves as foundational basis for better understanding the complexity of headache as a symptom-based condition,” James S. McGinley, PhD, of Vector Psychometric Group in Chapel Hill, N.C., and coauthors wrote. The study was published in Cephalalgia.
To evaluate whether keeping track of daily headache features can produce a useful, predictive score, the researchers reviewed data from migraine patients that were collected via N1‑Headache, a commercial digital health platform. Ultimately, information from 4,380 adults with a self-reported migraine diagnosis was analyzed; the sample was 90% female and their mean age was 37 years. Study participants reported an average of 33 headaches per month over the last 3 months. Nine patient-reported variables were initially considered in calculating the Headache Day Severity (HDS) score: pain intensity, headache duration, aura, pulsating/throbbing pain, unilateral pain, pain aggravation by activity, nausea/vomiting, photophobia, and phonophobia.
After determining that unilateral pain was not a meaningful variable, the researchers’ model found that, for every 1 standard deviation increase in HDS, the patient’s odds of physician visit increased by 71% (odds ratio, 1.71; 95% confidence interval, 1.32-2.21) and the odds of an ED visit increased by 342% (OR, 4.42; 95% CI, 2.23-7.60). They also found that the likelihood of missed work or school increased by 190% (OR, 2.90; 95% CI, 2.56-3.29), the chances of missing household work increased by 237% (OR, 3.37; 95% CI, 3.06-3.72) and the odds of missing other leisure or social activity increased by 228% (OR, 3.28; 95% CI, 2.97-3.64).
Tracking multiple variables
“We encourage all of our patients to monitor their headaches; there are just too many variables to try to keep it in your head,” Robert Cowan, MD, professor of neurology and chief of the division of headache medicine at Stanford (Calif.) University, said in an interview. He referenced a previous study from the University of Washington where patients were asked to track their headaches; that data was then compared against their self-reported headaches at a quarterly physician visit.
“What they found was there was absolutely no correlation with reported frequency of headache at the visit and what was seen in the tracker,” he said. “If patients had a headache in the previous 3 days before their visit, they felt that their headaches were poorly controlled. If they hadn’t, they thought their headaches were under good control. So the value of tracking is pretty clear.”
He added that, while not every headache sufferer needs to track their daily routines and symptoms, once those symptoms interfere with your life on a day-to-day basis, it’s probably time to consider keeping tabs on yourself with a tool of some sort. And while this study’s calculated HDS score supports the idea of migraine’s complexity, it also leaves unanswered the question of how to treat patients with severe symptoms.
“Frequently,” he said, “we’ll see patients who say: ‘I can deal with the pain, but the nausea makes it impossible to work, or the light sensitivity makes it impossible to go outside.’ The big question within the headache community is, can you treat migraine and have it address the whole spectrum, from dizziness to light sensitivity to sound sensitivity to vertigo, or should you be going after individual symptoms? That’s a controversy that rages on; I think most of us go for a combination. We’re in a polypharmacy phase: ‘If nausea is a big problem, take this, but we also try to prevent the whole migraine complex, so take this as well.’ ”
The authors acknowledged their study’s limitations, including the inability to determine how many participants’ migraines were formally diagnosed by a trained medical professional and the lack of generalizability of data from a convenience sample, though they added that patients who independently track their own headaches “may be representative of those who would participate in a clinical trial.” In addition, as seven of the nine features were collected in N1‑Headache on a yes/no scale, they recognized that “increasing the number of response options for each item may improve our ability to measure HDS.”
The study was funded by Amgen through the Competitive Grant Program in Migraine Research. The authors declared several potential conflicts of interest, including receiving funding, research support, salary, and honoraria from various pharmaceutical companies.
according to an analysis of data from thousands of headache sufferers who recorded variables like pain and duration in a daily digital diary.
“Our hope is that this work serves as foundational basis for better understanding the complexity of headache as a symptom-based condition,” James S. McGinley, PhD, of Vector Psychometric Group in Chapel Hill, N.C., and coauthors wrote. The study was published in Cephalalgia.
To evaluate whether keeping track of daily headache features can produce a useful, predictive score, the researchers reviewed data from migraine patients that were collected via N1‑Headache, a commercial digital health platform. Ultimately, information from 4,380 adults with a self-reported migraine diagnosis was analyzed; the sample was 90% female and their mean age was 37 years. Study participants reported an average of 33 headaches per month over the last 3 months. Nine patient-reported variables were initially considered in calculating the Headache Day Severity (HDS) score: pain intensity, headache duration, aura, pulsating/throbbing pain, unilateral pain, pain aggravation by activity, nausea/vomiting, photophobia, and phonophobia.
After determining that unilateral pain was not a meaningful variable, the researchers’ model found that, for every 1 standard deviation increase in HDS, the patient’s odds of physician visit increased by 71% (odds ratio, 1.71; 95% confidence interval, 1.32-2.21) and the odds of an ED visit increased by 342% (OR, 4.42; 95% CI, 2.23-7.60). They also found that the likelihood of missed work or school increased by 190% (OR, 2.90; 95% CI, 2.56-3.29), the chances of missing household work increased by 237% (OR, 3.37; 95% CI, 3.06-3.72) and the odds of missing other leisure or social activity increased by 228% (OR, 3.28; 95% CI, 2.97-3.64).
Tracking multiple variables
“We encourage all of our patients to monitor their headaches; there are just too many variables to try to keep it in your head,” Robert Cowan, MD, professor of neurology and chief of the division of headache medicine at Stanford (Calif.) University, said in an interview. He referenced a previous study from the University of Washington where patients were asked to track their headaches; that data was then compared against their self-reported headaches at a quarterly physician visit.
“What they found was there was absolutely no correlation with reported frequency of headache at the visit and what was seen in the tracker,” he said. “If patients had a headache in the previous 3 days before their visit, they felt that their headaches were poorly controlled. If they hadn’t, they thought their headaches were under good control. So the value of tracking is pretty clear.”
He added that, while not every headache sufferer needs to track their daily routines and symptoms, once those symptoms interfere with your life on a day-to-day basis, it’s probably time to consider keeping tabs on yourself with a tool of some sort. And while this study’s calculated HDS score supports the idea of migraine’s complexity, it also leaves unanswered the question of how to treat patients with severe symptoms.
“Frequently,” he said, “we’ll see patients who say: ‘I can deal with the pain, but the nausea makes it impossible to work, or the light sensitivity makes it impossible to go outside.’ The big question within the headache community is, can you treat migraine and have it address the whole spectrum, from dizziness to light sensitivity to sound sensitivity to vertigo, or should you be going after individual symptoms? That’s a controversy that rages on; I think most of us go for a combination. We’re in a polypharmacy phase: ‘If nausea is a big problem, take this, but we also try to prevent the whole migraine complex, so take this as well.’ ”
The authors acknowledged their study’s limitations, including the inability to determine how many participants’ migraines were formally diagnosed by a trained medical professional and the lack of generalizability of data from a convenience sample, though they added that patients who independently track their own headaches “may be representative of those who would participate in a clinical trial.” In addition, as seven of the nine features were collected in N1‑Headache on a yes/no scale, they recognized that “increasing the number of response options for each item may improve our ability to measure HDS.”
The study was funded by Amgen through the Competitive Grant Program in Migraine Research. The authors declared several potential conflicts of interest, including receiving funding, research support, salary, and honoraria from various pharmaceutical companies.
FROM CEPHALALGIA
Eurocentric standards of beauty are no longer dominant, experts agree
Addressing current standards of beauty at the Skin of Color Update 2021, dermatologists speaking about attitudes within four ethnic groups recounted a similar story: .
This change is relevant to dermatologists consulting with patients for cosmetic procedures. Four dermatologists who recounted the types of procedures their patients are requesting each reported that more patients are seeking cosmetic enhancements that accentuate rather than modify ethnic features.
Lips in Black, Asian, and Arab ethnic groups are just one example.
“Where several years ago, the conversation was really about lip reductions – how we can deemphasize the lip – I am now seeing lots of women of color coming in to ask about lip augmentation, looking to highlight their lips as a point of beauty,” reported Michelle Henry, MD, a dermatologist who practices in New York City.
She is not alone. Others participating on the same panel spoke of a growing interest among their patients to maintain or even emphasize the same ethnic features – including but not limited to lip shape and size that they were once anxious to modify.
In Asian patients, “the goal is not to Westernize,” agreed Annie Chiu, MD, a dermatologist who practices in North Redondo Beach, Calif. For lips, she spoke of the “50-50 ratio” of upper and lower lip symmetry that is consistent with a traditional Asian characteristic.
Like Dr. Henry, Dr. Chiu said that many requests for cosmetic work now involve accentuating Asian features, such as the oval shape of the face, rather than steps to modify this shape. This is a relatively recent change.
“I am finding that more of my patients want to improve the esthetic balance to optimize the appearance within their own ethnicity,” she said.
In the United Arab Emirates (UAE), Hassan Galadari, MD, an American-trained physician who is assistant professor of dermatology at the UAE University in Dubai, recently conducted a poll of his patients. In order of importance, full lips came after wide eyes, a straight nose, and a sharp jaw line. Full cheeks and a round face completed a list that diverges from the California-blond prototype.
Although Angelina Jolie was selected over several Lebanese actresses as a first choice for an icon of beauty in this same poll, Dr. Galadari pointed out that this actress has many of the features, including wide eyes, a straight nose, and full lips, that are consistent with traditional features of Arab beauty.
Perceptions of beauty are not just changing within ethnic groups but reflected in mass culture. Dr. Henry pointed to a published comparison of the “World’s Most Beautiful” list from People magazine in 2017 relative to 1990. Of the 50 celebrities on the list in 1990, 88% were Fitzpatrick skin types I-III. Only 12% were types IV-VI, which increased to almost 30% of the 135 celebrities on the list in 2017 (P = .01). In 1990, just one celebrity (2%) was of mixed race, which increased to 10.4% in 2017.
Among Hispanic women, the changes in attitude are perhaps best captured among younger relative to older patients requesting cosmetic work, according to Maritza I. Perez, MD, professor of dermatology, University of Connecticut, Farmington. She said that her younger patients are less likely to seek rhinoplasty and blepharoplasty relative to her older patients, a reflection perhaps of comfort with their natural looks.
However, “the celebration of Latinas as beautiful, seductive, and sexual is hardly new,” she said, indicating that younger Hispanic patients are probably not driven to modify their ethnic features because they are already widely admired. “Six of the 10 women crowned Miss Universe in the last decade were from Latin American countries,” she noted.
The general willingness of patients within ethnic groups and society as a whole to see ethnic features as admirable and attractive was generally regarded by all the panelists as a positive development.
Dr. Henry, who said she was “encouraged” by such trends as “the natural hair movement” and diminishing interest among her darker patients in lightening skin pigment, said, “I definitely see a change among my patients in regard to their goals.”
For clinicians offering consults to patients seeking cosmetic work, Dr. Henry recommended being aware and sensitive to this evolution in order to offer appropriate care.
Dr. Chiu, emphasizing the pride that many of her patients take in their Asian features, made the same recommendation. She credited globalization and social media for attitudes that have allowed an embrace of what are now far more inclusive standards of beauty.
Dr. Henry reports financial relationships with Allergan and Merz. Dr. Chiu has financial relationships with AbbVie, Cynosure, Merz, Revance, and Solta. Dr. Galadari reports financial relationships with nine pharmaceutical companies, including Allergan, Merz, Revance, and Fillmed Laboratories. Dr. Perez reports no relevant conflicts of interest.
Addressing current standards of beauty at the Skin of Color Update 2021, dermatologists speaking about attitudes within four ethnic groups recounted a similar story: .
This change is relevant to dermatologists consulting with patients for cosmetic procedures. Four dermatologists who recounted the types of procedures their patients are requesting each reported that more patients are seeking cosmetic enhancements that accentuate rather than modify ethnic features.
Lips in Black, Asian, and Arab ethnic groups are just one example.
“Where several years ago, the conversation was really about lip reductions – how we can deemphasize the lip – I am now seeing lots of women of color coming in to ask about lip augmentation, looking to highlight their lips as a point of beauty,” reported Michelle Henry, MD, a dermatologist who practices in New York City.
She is not alone. Others participating on the same panel spoke of a growing interest among their patients to maintain or even emphasize the same ethnic features – including but not limited to lip shape and size that they were once anxious to modify.
In Asian patients, “the goal is not to Westernize,” agreed Annie Chiu, MD, a dermatologist who practices in North Redondo Beach, Calif. For lips, she spoke of the “50-50 ratio” of upper and lower lip symmetry that is consistent with a traditional Asian characteristic.
Like Dr. Henry, Dr. Chiu said that many requests for cosmetic work now involve accentuating Asian features, such as the oval shape of the face, rather than steps to modify this shape. This is a relatively recent change.
“I am finding that more of my patients want to improve the esthetic balance to optimize the appearance within their own ethnicity,” she said.
In the United Arab Emirates (UAE), Hassan Galadari, MD, an American-trained physician who is assistant professor of dermatology at the UAE University in Dubai, recently conducted a poll of his patients. In order of importance, full lips came after wide eyes, a straight nose, and a sharp jaw line. Full cheeks and a round face completed a list that diverges from the California-blond prototype.
Although Angelina Jolie was selected over several Lebanese actresses as a first choice for an icon of beauty in this same poll, Dr. Galadari pointed out that this actress has many of the features, including wide eyes, a straight nose, and full lips, that are consistent with traditional features of Arab beauty.
Perceptions of beauty are not just changing within ethnic groups but reflected in mass culture. Dr. Henry pointed to a published comparison of the “World’s Most Beautiful” list from People magazine in 2017 relative to 1990. Of the 50 celebrities on the list in 1990, 88% were Fitzpatrick skin types I-III. Only 12% were types IV-VI, which increased to almost 30% of the 135 celebrities on the list in 2017 (P = .01). In 1990, just one celebrity (2%) was of mixed race, which increased to 10.4% in 2017.
Among Hispanic women, the changes in attitude are perhaps best captured among younger relative to older patients requesting cosmetic work, according to Maritza I. Perez, MD, professor of dermatology, University of Connecticut, Farmington. She said that her younger patients are less likely to seek rhinoplasty and blepharoplasty relative to her older patients, a reflection perhaps of comfort with their natural looks.
However, “the celebration of Latinas as beautiful, seductive, and sexual is hardly new,” she said, indicating that younger Hispanic patients are probably not driven to modify their ethnic features because they are already widely admired. “Six of the 10 women crowned Miss Universe in the last decade were from Latin American countries,” she noted.
The general willingness of patients within ethnic groups and society as a whole to see ethnic features as admirable and attractive was generally regarded by all the panelists as a positive development.
Dr. Henry, who said she was “encouraged” by such trends as “the natural hair movement” and diminishing interest among her darker patients in lightening skin pigment, said, “I definitely see a change among my patients in regard to their goals.”
For clinicians offering consults to patients seeking cosmetic work, Dr. Henry recommended being aware and sensitive to this evolution in order to offer appropriate care.
Dr. Chiu, emphasizing the pride that many of her patients take in their Asian features, made the same recommendation. She credited globalization and social media for attitudes that have allowed an embrace of what are now far more inclusive standards of beauty.
Dr. Henry reports financial relationships with Allergan and Merz. Dr. Chiu has financial relationships with AbbVie, Cynosure, Merz, Revance, and Solta. Dr. Galadari reports financial relationships with nine pharmaceutical companies, including Allergan, Merz, Revance, and Fillmed Laboratories. Dr. Perez reports no relevant conflicts of interest.
Addressing current standards of beauty at the Skin of Color Update 2021, dermatologists speaking about attitudes within four ethnic groups recounted a similar story: .
This change is relevant to dermatologists consulting with patients for cosmetic procedures. Four dermatologists who recounted the types of procedures their patients are requesting each reported that more patients are seeking cosmetic enhancements that accentuate rather than modify ethnic features.
Lips in Black, Asian, and Arab ethnic groups are just one example.
“Where several years ago, the conversation was really about lip reductions – how we can deemphasize the lip – I am now seeing lots of women of color coming in to ask about lip augmentation, looking to highlight their lips as a point of beauty,” reported Michelle Henry, MD, a dermatologist who practices in New York City.
She is not alone. Others participating on the same panel spoke of a growing interest among their patients to maintain or even emphasize the same ethnic features – including but not limited to lip shape and size that they were once anxious to modify.
In Asian patients, “the goal is not to Westernize,” agreed Annie Chiu, MD, a dermatologist who practices in North Redondo Beach, Calif. For lips, she spoke of the “50-50 ratio” of upper and lower lip symmetry that is consistent with a traditional Asian characteristic.
Like Dr. Henry, Dr. Chiu said that many requests for cosmetic work now involve accentuating Asian features, such as the oval shape of the face, rather than steps to modify this shape. This is a relatively recent change.
“I am finding that more of my patients want to improve the esthetic balance to optimize the appearance within their own ethnicity,” she said.
In the United Arab Emirates (UAE), Hassan Galadari, MD, an American-trained physician who is assistant professor of dermatology at the UAE University in Dubai, recently conducted a poll of his patients. In order of importance, full lips came after wide eyes, a straight nose, and a sharp jaw line. Full cheeks and a round face completed a list that diverges from the California-blond prototype.
Although Angelina Jolie was selected over several Lebanese actresses as a first choice for an icon of beauty in this same poll, Dr. Galadari pointed out that this actress has many of the features, including wide eyes, a straight nose, and full lips, that are consistent with traditional features of Arab beauty.
Perceptions of beauty are not just changing within ethnic groups but reflected in mass culture. Dr. Henry pointed to a published comparison of the “World’s Most Beautiful” list from People magazine in 2017 relative to 1990. Of the 50 celebrities on the list in 1990, 88% were Fitzpatrick skin types I-III. Only 12% were types IV-VI, which increased to almost 30% of the 135 celebrities on the list in 2017 (P = .01). In 1990, just one celebrity (2%) was of mixed race, which increased to 10.4% in 2017.
Among Hispanic women, the changes in attitude are perhaps best captured among younger relative to older patients requesting cosmetic work, according to Maritza I. Perez, MD, professor of dermatology, University of Connecticut, Farmington. She said that her younger patients are less likely to seek rhinoplasty and blepharoplasty relative to her older patients, a reflection perhaps of comfort with their natural looks.
However, “the celebration of Latinas as beautiful, seductive, and sexual is hardly new,” she said, indicating that younger Hispanic patients are probably not driven to modify their ethnic features because they are already widely admired. “Six of the 10 women crowned Miss Universe in the last decade were from Latin American countries,” she noted.
The general willingness of patients within ethnic groups and society as a whole to see ethnic features as admirable and attractive was generally regarded by all the panelists as a positive development.
Dr. Henry, who said she was “encouraged” by such trends as “the natural hair movement” and diminishing interest among her darker patients in lightening skin pigment, said, “I definitely see a change among my patients in regard to their goals.”
For clinicians offering consults to patients seeking cosmetic work, Dr. Henry recommended being aware and sensitive to this evolution in order to offer appropriate care.
Dr. Chiu, emphasizing the pride that many of her patients take in their Asian features, made the same recommendation. She credited globalization and social media for attitudes that have allowed an embrace of what are now far more inclusive standards of beauty.
Dr. Henry reports financial relationships with Allergan and Merz. Dr. Chiu has financial relationships with AbbVie, Cynosure, Merz, Revance, and Solta. Dr. Galadari reports financial relationships with nine pharmaceutical companies, including Allergan, Merz, Revance, and Fillmed Laboratories. Dr. Perez reports no relevant conflicts of interest.
FROM SOC 2021
How could this happen? Judge forces doctors to give ivermectin
The judge’s order was a major affront to many clinical ethicists. A county judge in Ohio ordered a hospital to give ivermectin to a COVID-19 patient on a ventilator. This order occurred against the advice and judgment of the local physicians. It occurred in spite of the hospital’s lawyers fighting the order. How could such a situation occur?
This column is not the appropriate forum to debate the use of ivermectin. The Food and Drug Administration has not approved the drug for treating COVID-19. Indeed, the FDA has specifically recommended against its use.1 So has the Centers for Disease Control and Prevention.2 Poison control centers report a large uptick in exposures this summer because of self-medication, sometimes from veterinary sources.3
Fortunately for this case, the judge who overruled the order, Judge Michael A. Oster, wrote in his decision a summary of facts presented by both sides. The topic here is how a judge could order a medical institution and its staff to provide care against medical judgment. A key tenet of clinical ethics consultation is that the consultant needs to do their own investigation. Most veteran consultants have a litany of anecdotes wherein the initial story changed markedly as new facts were uncovered. The more outrageous the initial story, the more likely a major distortion is found. Therefore, most clinical ethics consultants are reluctant to discuss case studies based solely on publicly available information. Often, it is nearly impossible to obtain further information. One side of the story may be gagged by privacy laws. However, cases must sometimes be discussed based on the limited information available because, without that discussion, egregious violations of medical ethics would not be brought to light.
Fortunately for this case, Judge Osler’s decision contains a summary of facts presented by both sides. In August 2021, a 51-year-old patient with severe COVID-19 is in an Ohio intensive care unit on a ventilator. His wife seeks and obtains a prescription for ivermectin from a physician who has an Ohio state medical license but lives elsewhere, has no clinical privileges at the involved hospital, and has never examined the patient. The wife, as a surrogate decision maker, demands her husband receive the medication. The medical staff involved do not consider it a valid treatment. The wife seeks an injunction. A county judge orders the hospital to administer a specified dose of ivermectin daily for 21 days.4 That judge further grants an emergency preliminary injunction for 14 days that orders administration of the medication while legal appeals are made. Two weeks later, a second county judge hearing the case rules that the wife has not presented convincing evidence that she is likely to ultimately win the case on the merits.5 Therefore, the second judge reverses the preliminary injunction. The hospital need not continue to give the medication while further legal proceedings take place.
Cases like this are uncommon. Judges generally defer the authority for medical decisions to physicians. Various attitudes combine to make such an event happen. The judge may view the hospital as a local monopoly of health care and the patient may be too unstable to transport elsewhere. A judge in that situation, combined with a “the consumer is always right” mentality, and a sympathetic plaintiff, may seek to make miracles happen.
Judges overriding science are more likely to manifest when they see the science as ambiguous. Scientists have lost some of the gravitas they had when men walked on the moon. The spectacular success of the mRNA vaccines has surprisingly not reversed that loss. Science has been tainted by mercenary scientists, biased researchers seeking publications, and the large volume of published medical research that is false.
But there is more going on here. In the United States there has been a significant rebellion against any form of expertise and any form of authority. The echo chambers of misinformation on social media have led to polarization, conspiracy theories, and loyalty to political tribe rather than truth; hence the battle over masks and vaccines. This breakdown in authority is accompanied by losses in virtues such as civic duty and loving one’s neighbor. This is a failure of modern moral institutions. When major medical journals print opinion pieces portraying physicians as interchangeable automatons,6 it should not be surprising to see judges tempted by similar imagery.
One part of the solution is accountability in peer review. With 30,000 county judges scattered in 50 states, there will always be a few rogue and maverick attitudes among judges. The judiciary has a means of reassigning rebels to less impactful tasks. Similarly, if the physician who counseled the wife to use ivermectin had privileges at the admitting hospital, then peer review and credential committees could discipline behaviors that were too far outside accepted norms. Even when a consensus on best practice is hard to establish, damage can be mitigated by creating consequences for promoting aberrant care.
Dr. Powell is a retired pediatric hospitalist and clinical ethics consultant living in St. Louis. Email him at [email protected].
References
1. “Why you should not use ivermectin to treat or prevent COVID-19,” FDA Consumer Updates, Sept. 3, 2021.
2. “Rapid increase in ivermectin prescriptions and reports of severe illness associated with use of products containing ivermectin to prevent or treat COVID-19,” CDC Health Advisory, Aug. 26, 2021.
3. National Poison Data System Bulletin: COVID-19 (Ivermectin), American Association of Poison Control Centers, 2021.
4. Smith v West Chester Hosptial, LLC, DBA West Chester Hospital, Butler County Clerk of Courts, Aug. 23, 2021.
5. Smith v West Chester Hosptial, LLC, Decision denying plaintiff’s action for a preliminary injunction, Butler County Clerk of Courts, Sept. 6, 2021.
6. “Conscientious objection in medicine,” BMJ 2006 Feb 2. doi: 10.1136/bmj.332.7536.294.
The judge’s order was a major affront to many clinical ethicists. A county judge in Ohio ordered a hospital to give ivermectin to a COVID-19 patient on a ventilator. This order occurred against the advice and judgment of the local physicians. It occurred in spite of the hospital’s lawyers fighting the order. How could such a situation occur?
This column is not the appropriate forum to debate the use of ivermectin. The Food and Drug Administration has not approved the drug for treating COVID-19. Indeed, the FDA has specifically recommended against its use.1 So has the Centers for Disease Control and Prevention.2 Poison control centers report a large uptick in exposures this summer because of self-medication, sometimes from veterinary sources.3
Fortunately for this case, the judge who overruled the order, Judge Michael A. Oster, wrote in his decision a summary of facts presented by both sides. The topic here is how a judge could order a medical institution and its staff to provide care against medical judgment. A key tenet of clinical ethics consultation is that the consultant needs to do their own investigation. Most veteran consultants have a litany of anecdotes wherein the initial story changed markedly as new facts were uncovered. The more outrageous the initial story, the more likely a major distortion is found. Therefore, most clinical ethics consultants are reluctant to discuss case studies based solely on publicly available information. Often, it is nearly impossible to obtain further information. One side of the story may be gagged by privacy laws. However, cases must sometimes be discussed based on the limited information available because, without that discussion, egregious violations of medical ethics would not be brought to light.
Fortunately for this case, Judge Osler’s decision contains a summary of facts presented by both sides. In August 2021, a 51-year-old patient with severe COVID-19 is in an Ohio intensive care unit on a ventilator. His wife seeks and obtains a prescription for ivermectin from a physician who has an Ohio state medical license but lives elsewhere, has no clinical privileges at the involved hospital, and has never examined the patient. The wife, as a surrogate decision maker, demands her husband receive the medication. The medical staff involved do not consider it a valid treatment. The wife seeks an injunction. A county judge orders the hospital to administer a specified dose of ivermectin daily for 21 days.4 That judge further grants an emergency preliminary injunction for 14 days that orders administration of the medication while legal appeals are made. Two weeks later, a second county judge hearing the case rules that the wife has not presented convincing evidence that she is likely to ultimately win the case on the merits.5 Therefore, the second judge reverses the preliminary injunction. The hospital need not continue to give the medication while further legal proceedings take place.
Cases like this are uncommon. Judges generally defer the authority for medical decisions to physicians. Various attitudes combine to make such an event happen. The judge may view the hospital as a local monopoly of health care and the patient may be too unstable to transport elsewhere. A judge in that situation, combined with a “the consumer is always right” mentality, and a sympathetic plaintiff, may seek to make miracles happen.
Judges overriding science are more likely to manifest when they see the science as ambiguous. Scientists have lost some of the gravitas they had when men walked on the moon. The spectacular success of the mRNA vaccines has surprisingly not reversed that loss. Science has been tainted by mercenary scientists, biased researchers seeking publications, and the large volume of published medical research that is false.
But there is more going on here. In the United States there has been a significant rebellion against any form of expertise and any form of authority. The echo chambers of misinformation on social media have led to polarization, conspiracy theories, and loyalty to political tribe rather than truth; hence the battle over masks and vaccines. This breakdown in authority is accompanied by losses in virtues such as civic duty and loving one’s neighbor. This is a failure of modern moral institutions. When major medical journals print opinion pieces portraying physicians as interchangeable automatons,6 it should not be surprising to see judges tempted by similar imagery.
One part of the solution is accountability in peer review. With 30,000 county judges scattered in 50 states, there will always be a few rogue and maverick attitudes among judges. The judiciary has a means of reassigning rebels to less impactful tasks. Similarly, if the physician who counseled the wife to use ivermectin had privileges at the admitting hospital, then peer review and credential committees could discipline behaviors that were too far outside accepted norms. Even when a consensus on best practice is hard to establish, damage can be mitigated by creating consequences for promoting aberrant care.
Dr. Powell is a retired pediatric hospitalist and clinical ethics consultant living in St. Louis. Email him at [email protected].
References
1. “Why you should not use ivermectin to treat or prevent COVID-19,” FDA Consumer Updates, Sept. 3, 2021.
2. “Rapid increase in ivermectin prescriptions and reports of severe illness associated with use of products containing ivermectin to prevent or treat COVID-19,” CDC Health Advisory, Aug. 26, 2021.
3. National Poison Data System Bulletin: COVID-19 (Ivermectin), American Association of Poison Control Centers, 2021.
4. Smith v West Chester Hosptial, LLC, DBA West Chester Hospital, Butler County Clerk of Courts, Aug. 23, 2021.
5. Smith v West Chester Hosptial, LLC, Decision denying plaintiff’s action for a preliminary injunction, Butler County Clerk of Courts, Sept. 6, 2021.
6. “Conscientious objection in medicine,” BMJ 2006 Feb 2. doi: 10.1136/bmj.332.7536.294.
The judge’s order was a major affront to many clinical ethicists. A county judge in Ohio ordered a hospital to give ivermectin to a COVID-19 patient on a ventilator. This order occurred against the advice and judgment of the local physicians. It occurred in spite of the hospital’s lawyers fighting the order. How could such a situation occur?
This column is not the appropriate forum to debate the use of ivermectin. The Food and Drug Administration has not approved the drug for treating COVID-19. Indeed, the FDA has specifically recommended against its use.1 So has the Centers for Disease Control and Prevention.2 Poison control centers report a large uptick in exposures this summer because of self-medication, sometimes from veterinary sources.3
Fortunately for this case, the judge who overruled the order, Judge Michael A. Oster, wrote in his decision a summary of facts presented by both sides. The topic here is how a judge could order a medical institution and its staff to provide care against medical judgment. A key tenet of clinical ethics consultation is that the consultant needs to do their own investigation. Most veteran consultants have a litany of anecdotes wherein the initial story changed markedly as new facts were uncovered. The more outrageous the initial story, the more likely a major distortion is found. Therefore, most clinical ethics consultants are reluctant to discuss case studies based solely on publicly available information. Often, it is nearly impossible to obtain further information. One side of the story may be gagged by privacy laws. However, cases must sometimes be discussed based on the limited information available because, without that discussion, egregious violations of medical ethics would not be brought to light.
Fortunately for this case, Judge Osler’s decision contains a summary of facts presented by both sides. In August 2021, a 51-year-old patient with severe COVID-19 is in an Ohio intensive care unit on a ventilator. His wife seeks and obtains a prescription for ivermectin from a physician who has an Ohio state medical license but lives elsewhere, has no clinical privileges at the involved hospital, and has never examined the patient. The wife, as a surrogate decision maker, demands her husband receive the medication. The medical staff involved do not consider it a valid treatment. The wife seeks an injunction. A county judge orders the hospital to administer a specified dose of ivermectin daily for 21 days.4 That judge further grants an emergency preliminary injunction for 14 days that orders administration of the medication while legal appeals are made. Two weeks later, a second county judge hearing the case rules that the wife has not presented convincing evidence that she is likely to ultimately win the case on the merits.5 Therefore, the second judge reverses the preliminary injunction. The hospital need not continue to give the medication while further legal proceedings take place.
Cases like this are uncommon. Judges generally defer the authority for medical decisions to physicians. Various attitudes combine to make such an event happen. The judge may view the hospital as a local monopoly of health care and the patient may be too unstable to transport elsewhere. A judge in that situation, combined with a “the consumer is always right” mentality, and a sympathetic plaintiff, may seek to make miracles happen.
Judges overriding science are more likely to manifest when they see the science as ambiguous. Scientists have lost some of the gravitas they had when men walked on the moon. The spectacular success of the mRNA vaccines has surprisingly not reversed that loss. Science has been tainted by mercenary scientists, biased researchers seeking publications, and the large volume of published medical research that is false.
But there is more going on here. In the United States there has been a significant rebellion against any form of expertise and any form of authority. The echo chambers of misinformation on social media have led to polarization, conspiracy theories, and loyalty to political tribe rather than truth; hence the battle over masks and vaccines. This breakdown in authority is accompanied by losses in virtues such as civic duty and loving one’s neighbor. This is a failure of modern moral institutions. When major medical journals print opinion pieces portraying physicians as interchangeable automatons,6 it should not be surprising to see judges tempted by similar imagery.
One part of the solution is accountability in peer review. With 30,000 county judges scattered in 50 states, there will always be a few rogue and maverick attitudes among judges. The judiciary has a means of reassigning rebels to less impactful tasks. Similarly, if the physician who counseled the wife to use ivermectin had privileges at the admitting hospital, then peer review and credential committees could discipline behaviors that were too far outside accepted norms. Even when a consensus on best practice is hard to establish, damage can be mitigated by creating consequences for promoting aberrant care.
Dr. Powell is a retired pediatric hospitalist and clinical ethics consultant living in St. Louis. Email him at [email protected].
References
1. “Why you should not use ivermectin to treat or prevent COVID-19,” FDA Consumer Updates, Sept. 3, 2021.
2. “Rapid increase in ivermectin prescriptions and reports of severe illness associated with use of products containing ivermectin to prevent or treat COVID-19,” CDC Health Advisory, Aug. 26, 2021.
3. National Poison Data System Bulletin: COVID-19 (Ivermectin), American Association of Poison Control Centers, 2021.
4. Smith v West Chester Hosptial, LLC, DBA West Chester Hospital, Butler County Clerk of Courts, Aug. 23, 2021.
5. Smith v West Chester Hosptial, LLC, Decision denying plaintiff’s action for a preliminary injunction, Butler County Clerk of Courts, Sept. 6, 2021.
6. “Conscientious objection in medicine,” BMJ 2006 Feb 2. doi: 10.1136/bmj.332.7536.294.
Gut microbiome could make weight loss easier for some
Researchers found that the gut microbiome – the bacteria that help digest food and absorb nutrients in the intestines – can influence attempts at weight loss.
They identified genes within these bacteria that determine how quickly the bacteria grow, how well people can take advantage of nutrients in food, and whether starches and fiber, in particular, get broken down into sugars too quickly to aid weight loss.
“Some people have a harder time losing weight than others,” study author Sean Gibbons, PhD, told this news organization. “For example, some people are able to control their weight through basic lifestyle interventions, while others may not.”
Furthermore, it is difficult to predict which individuals will respond to changes in diet or exercise and who might require more intense strategies.
The study, which was published online September 14 in mSystems, a journal of the American Society for Microbiology, could bring us closer to an answer.
“We’ve identified specific genetic signatures in the gut microbiome that were predictive of weight loss response in a small cohort of patients following a healthy lifestyle intervention,” explained Dr. Gibbons, Washington Research Foundation distinguished investigator and assistant professor at the Institute for Systems Biology in Seattle.
Weight loss takes guts?
Differences in 31 functional genes emerged from the gut microbiome in 48 people who lost 1% or more of their weight each month compared with 57 others whose weight remained the same. These findings came from stool samples taken 6 to 12 months after people started a commercial weight loss coaching program.
In contrast, lead author Christian Diener, PhD, also of the Institute for Systems Biology, and colleagues found only one factor in the blood that differed between the weight loss and weight maintenance groups. (They specifically evaluated proteins associated with obesity in the blood and genetic data from stool samples in a subset of 25 participants.)
Their findings align with previous research showing different types of bacteria in the gut microbiome can affect the success of weight loss interventions, but they took it a step further to determine how this works.
“We know that the gut microbiome plays an important role in weight management and can also influence a response to weight loss interventions. However, specific gut microbiome features that can explain this observation in more detail are still to be discovered,” Hana Kahleova, MD, PhD, MBA, director of clinical research at the Physicians Committee for Responsible Medicine in Washington, D.C., told this news organization when asked for comment.
Good versus bad players
On the plus side, genes that help bacteria grow more rapidly were associated with weight loss. These bacteria take more of the nutrients in food for themselves, leaving less to go toward human weight gain compared with slower growing bacteria.
In fact, prior evidence points to a particular gut bacteria, Prevotella, as being beneficial for weight loss. “In our study,” Dr. Gibbons said, “we found that some of the fastest-growing microbes in the weight-loss responder group were from the genus Prevotella.”
On the other hand, bacteria that produce more enzymes to breakdown starches or fiber quickly into sugars, for example, were linked with making people more resistant to weight loss.
“By understanding these functional patterns, we may one day be able to engineer resistant microbiomes to be more permissive to weight loss,” Dr. Gibbons said.
Dr. Kahleova agreed. “These findings expand our understanding of the specific features of the gut microbiome that play a role in weight loss,” she said.
Moving beyond BMI
Interestingly, the researchers controlled for baseline body mass index (BMI) and other factors that could affect weight loss. People who start off with a higher BMI tend to lose more weight than others, a phenomenon known as ‘regression to the mean.” This factor confounded some earlier research, they noted.
“The vast majority of features associated with weight loss, independent of BMI, were functional genes within the gut metagenome,” Dr. Gibbons said.
“This tells us that the gut microbiome is an important modulator of weight loss, independent of your underlying metabolic health state, baseline diet, or BMI status.”
“This study described several metagenomic functional features that were associated with weight loss after controlling for potential confounders, such as age, sex, and baseline BMI,” Dr. Kahleova said. “These findings ... may help optimize the weight-loss protocols in future studies.”
Fecal microbiota transplants?
What do the findings mean for people willing to adjust their diet – or undergo a fecal transplant – to include more of the gut bacteria that facilitate weight loss?
It could be too soon for such interventions, Dr. Gibbons said. “It is still very difficult to rationally engineer your gut microbiome.”
“Interestingly, a recent study suggests that fecal transplants from a high-Prevotella donor may be able to flip low-Prevotella recipients to high-Prevotella,” Dr. Gibbons said.
More research is required, however, to understand whether or not these fecal microbial transplant-flipped individuals are also more capable of weight loss, he added.
Beyond that, “I can’t give any specific recommendations, other than that [people] should eat more fiber-rich, plant-based, whole foods and reduce their consumption of red meat. That’s well-supported.”
“Also, prepare your own meals, rather than relying on sugar and sodium-rich processed foods,” Dr. Gibbons said.
Dr. Gibbons and his team hope to validate their work in larger human studies “and perhaps develop clinical diagnostics or interventions for people trying to lose weight.”
A version of this article first appeared on Medscape.com.
Researchers found that the gut microbiome – the bacteria that help digest food and absorb nutrients in the intestines – can influence attempts at weight loss.
They identified genes within these bacteria that determine how quickly the bacteria grow, how well people can take advantage of nutrients in food, and whether starches and fiber, in particular, get broken down into sugars too quickly to aid weight loss.
“Some people have a harder time losing weight than others,” study author Sean Gibbons, PhD, told this news organization. “For example, some people are able to control their weight through basic lifestyle interventions, while others may not.”
Furthermore, it is difficult to predict which individuals will respond to changes in diet or exercise and who might require more intense strategies.
The study, which was published online September 14 in mSystems, a journal of the American Society for Microbiology, could bring us closer to an answer.
“We’ve identified specific genetic signatures in the gut microbiome that were predictive of weight loss response in a small cohort of patients following a healthy lifestyle intervention,” explained Dr. Gibbons, Washington Research Foundation distinguished investigator and assistant professor at the Institute for Systems Biology in Seattle.
Weight loss takes guts?
Differences in 31 functional genes emerged from the gut microbiome in 48 people who lost 1% or more of their weight each month compared with 57 others whose weight remained the same. These findings came from stool samples taken 6 to 12 months after people started a commercial weight loss coaching program.
In contrast, lead author Christian Diener, PhD, also of the Institute for Systems Biology, and colleagues found only one factor in the blood that differed between the weight loss and weight maintenance groups. (They specifically evaluated proteins associated with obesity in the blood and genetic data from stool samples in a subset of 25 participants.)
Their findings align with previous research showing different types of bacteria in the gut microbiome can affect the success of weight loss interventions, but they took it a step further to determine how this works.
“We know that the gut microbiome plays an important role in weight management and can also influence a response to weight loss interventions. However, specific gut microbiome features that can explain this observation in more detail are still to be discovered,” Hana Kahleova, MD, PhD, MBA, director of clinical research at the Physicians Committee for Responsible Medicine in Washington, D.C., told this news organization when asked for comment.
Good versus bad players
On the plus side, genes that help bacteria grow more rapidly were associated with weight loss. These bacteria take more of the nutrients in food for themselves, leaving less to go toward human weight gain compared with slower growing bacteria.
In fact, prior evidence points to a particular gut bacteria, Prevotella, as being beneficial for weight loss. “In our study,” Dr. Gibbons said, “we found that some of the fastest-growing microbes in the weight-loss responder group were from the genus Prevotella.”
On the other hand, bacteria that produce more enzymes to breakdown starches or fiber quickly into sugars, for example, were linked with making people more resistant to weight loss.
“By understanding these functional patterns, we may one day be able to engineer resistant microbiomes to be more permissive to weight loss,” Dr. Gibbons said.
Dr. Kahleova agreed. “These findings expand our understanding of the specific features of the gut microbiome that play a role in weight loss,” she said.
Moving beyond BMI
Interestingly, the researchers controlled for baseline body mass index (BMI) and other factors that could affect weight loss. People who start off with a higher BMI tend to lose more weight than others, a phenomenon known as ‘regression to the mean.” This factor confounded some earlier research, they noted.
“The vast majority of features associated with weight loss, independent of BMI, were functional genes within the gut metagenome,” Dr. Gibbons said.
“This tells us that the gut microbiome is an important modulator of weight loss, independent of your underlying metabolic health state, baseline diet, or BMI status.”
“This study described several metagenomic functional features that were associated with weight loss after controlling for potential confounders, such as age, sex, and baseline BMI,” Dr. Kahleova said. “These findings ... may help optimize the weight-loss protocols in future studies.”
Fecal microbiota transplants?
What do the findings mean for people willing to adjust their diet – or undergo a fecal transplant – to include more of the gut bacteria that facilitate weight loss?
It could be too soon for such interventions, Dr. Gibbons said. “It is still very difficult to rationally engineer your gut microbiome.”
“Interestingly, a recent study suggests that fecal transplants from a high-Prevotella donor may be able to flip low-Prevotella recipients to high-Prevotella,” Dr. Gibbons said.
More research is required, however, to understand whether or not these fecal microbial transplant-flipped individuals are also more capable of weight loss, he added.
Beyond that, “I can’t give any specific recommendations, other than that [people] should eat more fiber-rich, plant-based, whole foods and reduce their consumption of red meat. That’s well-supported.”
“Also, prepare your own meals, rather than relying on sugar and sodium-rich processed foods,” Dr. Gibbons said.
Dr. Gibbons and his team hope to validate their work in larger human studies “and perhaps develop clinical diagnostics or interventions for people trying to lose weight.”
A version of this article first appeared on Medscape.com.
Researchers found that the gut microbiome – the bacteria that help digest food and absorb nutrients in the intestines – can influence attempts at weight loss.
They identified genes within these bacteria that determine how quickly the bacteria grow, how well people can take advantage of nutrients in food, and whether starches and fiber, in particular, get broken down into sugars too quickly to aid weight loss.
“Some people have a harder time losing weight than others,” study author Sean Gibbons, PhD, told this news organization. “For example, some people are able to control their weight through basic lifestyle interventions, while others may not.”
Furthermore, it is difficult to predict which individuals will respond to changes in diet or exercise and who might require more intense strategies.
The study, which was published online September 14 in mSystems, a journal of the American Society for Microbiology, could bring us closer to an answer.
“We’ve identified specific genetic signatures in the gut microbiome that were predictive of weight loss response in a small cohort of patients following a healthy lifestyle intervention,” explained Dr. Gibbons, Washington Research Foundation distinguished investigator and assistant professor at the Institute for Systems Biology in Seattle.
Weight loss takes guts?
Differences in 31 functional genes emerged from the gut microbiome in 48 people who lost 1% or more of their weight each month compared with 57 others whose weight remained the same. These findings came from stool samples taken 6 to 12 months after people started a commercial weight loss coaching program.
In contrast, lead author Christian Diener, PhD, also of the Institute for Systems Biology, and colleagues found only one factor in the blood that differed between the weight loss and weight maintenance groups. (They specifically evaluated proteins associated with obesity in the blood and genetic data from stool samples in a subset of 25 participants.)
Their findings align with previous research showing different types of bacteria in the gut microbiome can affect the success of weight loss interventions, but they took it a step further to determine how this works.
“We know that the gut microbiome plays an important role in weight management and can also influence a response to weight loss interventions. However, specific gut microbiome features that can explain this observation in more detail are still to be discovered,” Hana Kahleova, MD, PhD, MBA, director of clinical research at the Physicians Committee for Responsible Medicine in Washington, D.C., told this news organization when asked for comment.
Good versus bad players
On the plus side, genes that help bacteria grow more rapidly were associated with weight loss. These bacteria take more of the nutrients in food for themselves, leaving less to go toward human weight gain compared with slower growing bacteria.
In fact, prior evidence points to a particular gut bacteria, Prevotella, as being beneficial for weight loss. “In our study,” Dr. Gibbons said, “we found that some of the fastest-growing microbes in the weight-loss responder group were from the genus Prevotella.”
On the other hand, bacteria that produce more enzymes to breakdown starches or fiber quickly into sugars, for example, were linked with making people more resistant to weight loss.
“By understanding these functional patterns, we may one day be able to engineer resistant microbiomes to be more permissive to weight loss,” Dr. Gibbons said.
Dr. Kahleova agreed. “These findings expand our understanding of the specific features of the gut microbiome that play a role in weight loss,” she said.
Moving beyond BMI
Interestingly, the researchers controlled for baseline body mass index (BMI) and other factors that could affect weight loss. People who start off with a higher BMI tend to lose more weight than others, a phenomenon known as ‘regression to the mean.” This factor confounded some earlier research, they noted.
“The vast majority of features associated with weight loss, independent of BMI, were functional genes within the gut metagenome,” Dr. Gibbons said.
“This tells us that the gut microbiome is an important modulator of weight loss, independent of your underlying metabolic health state, baseline diet, or BMI status.”
“This study described several metagenomic functional features that were associated with weight loss after controlling for potential confounders, such as age, sex, and baseline BMI,” Dr. Kahleova said. “These findings ... may help optimize the weight-loss protocols in future studies.”
Fecal microbiota transplants?
What do the findings mean for people willing to adjust their diet – or undergo a fecal transplant – to include more of the gut bacteria that facilitate weight loss?
It could be too soon for such interventions, Dr. Gibbons said. “It is still very difficult to rationally engineer your gut microbiome.”
“Interestingly, a recent study suggests that fecal transplants from a high-Prevotella donor may be able to flip low-Prevotella recipients to high-Prevotella,” Dr. Gibbons said.
More research is required, however, to understand whether or not these fecal microbial transplant-flipped individuals are also more capable of weight loss, he added.
Beyond that, “I can’t give any specific recommendations, other than that [people] should eat more fiber-rich, plant-based, whole foods and reduce their consumption of red meat. That’s well-supported.”
“Also, prepare your own meals, rather than relying on sugar and sodium-rich processed foods,” Dr. Gibbons said.
Dr. Gibbons and his team hope to validate their work in larger human studies “and perhaps develop clinical diagnostics or interventions for people trying to lose weight.”
A version of this article first appeared on Medscape.com.
Hormone agonist therapy disrupts bone density in transgender youth
The use of gonadotropin-releasing hormone agonists has a negative effect on bone mass in transgender youth, according to data from 172 individuals.
The onset of puberty and pubertal hormones contributes to the development of bone mass and body composition in adolescence, wrote Behdad Navabi, MD, and colleagues at Children’s Hospital of Eastern Ontario, Canada. Although the safety and efficacy of gonadotropin-releasing hormone agonists (GnRHa) has been described in short-term studies of youth with gender dysphoria, concerns persist about suppression of bone mass accrual from extended use of GnRHas in this population, they noted.
In a study published in Pediatrics, the researchers reviewed data from 172 youth younger than 18 years of age who were treated with GNRHa and underwent at least one baseline dual-energy radiograph absorptiometry (DXA) measurement between January 2006 and April 2017 at a single center. The standard treatment protocol started with three doses of 7.5 mg leuprolide acetate, given intramuscularly every 4 weeks, followed by 11.25 mg intramuscularly every 12 weeks after puberty suppression was confirmed both clinically and biochemically. Areal bone mineral density (aBMD) measurement z scores were based on birth-assigned sex, age, and ethnicity, and assessed at baseline and every 12 months. In addition, volumetric bone mineral density was calculated as bone mineral apparent density (BMAD) at the lower spine, and the z score based on age-matched, birth-assigned gender BMAD.
Overall, 55.2% of the youth were vitamin D deficient or insufficient at baseline, but 87.3% were sufficient by the time of a third follow-up visit after treatment with 1,000-2,000 IU of vitamin D daily; no cases of vitamin D toxicity were reported.
At baseline, transgender females had lower z scores for the LS aBMD and BMAD compared to transgender males, reflecting a difference seen in previous studies of transgender youth and adult females, the researchers noted.
The researchers analyzed pre- and posttreatment DXA data in a subgroup of 36 transgender females and 80 transgender males to identify any changes associated with GnRHa. The average time between the DXA scans was 407 days. In this population, aBMD z scores at the lower lumbar spine (LS), left total hip (LTH), and total body less head (TBLH) decreased significantly from baseline in transgender males and females.
Among transgender males, LS bone mineral apparent density (BMAD) z scores also decreased significantly from baseline, but no such change occurred among transgender females. The most significant decrease in z scores occurred in the LS aBMD and BMAD of transgender males, with changes that reflect findings from previous studies and may be explained by decreased estrogen, the researchers wrote.
In terms of body composition, no significant changes occurred in body mass index z score from baseline to follow-up in transgender males or females, the researchers noted, and changes in both gynoid and android fat percentages were consistent with the individuals’ affirmed genders. No vertebral fractures were detected.
However, GnRHa was significantly associated with a decrease in total body fat percentage and a decrease in lean body mass (LBM) in transgender females.
The study findings were limited by several factors, including the lack of consistent baseline physical activity records, and limited analysis at follow-up of the possible role of physical activity in bone health and body composition, the researchers noted. However, the results were strengthened by the relatively large study population with baseline assessments, and by the pre- and posttreatment analysis, they added.
“Evidence on GnRHa-associated changes in body composition and BMD will help health care professionals involved in the care of youth with GD [gender dysphoria] to counsel appropriately and optimize their bone health,” the researchers said. “Given the absence of vertebral fractures detected in those with significant decreases in their LS z scores, the significance of BMD effects of GnRHa in transgender youth needs further study, as well as whether future spine radiographs are needed on the basis of BMD trajectory,” they concluded.
Balance bone health concerns with potential benefits
The effect of estrogen and testosterone on bone geometry in puberty varies, and the increase in the use of GnRHa as part of a multidisciplinary gender transition plan makes research on the skeletal impact of this therapy in transgender youth a top priority, Laura K. Bachrach, MD, of Stanford (Calif.) University, and Catherine M. Gordon of Harvard Medical School, Boston, wrote in an accompanying editorial.
The decrease in areal bone mineral density and in bone mineral apparent density (BMAD) z scores in the current study is not unexpected, but the key question is how much bone density recovers once the suppression therapy ends and transgender sex steroid use begins, they said. “Follow-up studies of young adults treated with GnRHa for precocious puberty in childhood are reassuring,” they wrote. “It is premature, however, to extrapolate from these findings to transgender youth,” because the impact of gender-affirming sex steroid therapy on the skeleton at older ages and stages of maturity are unclear, they emphasized.
In the absence of definitive answers, the editorial authors advised clinicians treating youth with gender dysphoria to provide a balanced view of the risks and benefits of hormone therapy, and encourage adequate intake of dietary vitamin D and calcium, along with weight-bearing physical activity, to promote general bone health. “Transgender teenagers and their parents should be reassured that some recovery from decreases in aBMD during pubertal suppression with GnRHa is likely,” the authors noted. Bone health should be monitored throughout all stages of treatment in transgender youth, but concerns about transient bone loss should not discourage gender transition therapy, they emphasized. “In this patient group, providing a pause in pubertal development offers a life-changing and, for some, a life-saving intervention,” they concluded.
Comparison to cisgender controls would add value
“This study is important because one of the major side effects of GnRH agonists is decreased bone density, especially the longer that patients are on them,” M. Brett Cooper, MD, of UT Southwestern Medical Center, said in an interview. The findings add to existing data to underscore the importance of screening for low bone density and low vitamin D levels, Dr. Cooper added.
Dr. Cooper said that he was not surprised by the study findings. “I think that this study supported what clinicians already knew, which is that GnRH agonists do potentially cause a decline in bone mineral density and thus, you need to support these patients as best you can with calcium, vitamin D, and weight-bearing exercise,” he noted.
Dr. Cooper emphasized two main take-home points from the study. “First, clinicians who prescribe GnRH agonists need to ensure that they are checking bone density and vitamin D measurements, and then optimizing these appropriately,” he said. “Second, when a bone density is found to be low or a vitamin level is low, clinicians need to ensure that they are monitored and treated appropriately.” Clinicians need to use these data when deciding when to start gender-affirming hormones so their patients have the best chance to recover bone density, he added.
“I think one confounding factor on this study is the ranges they used for vitamin D deficiency,” Dr. Cooper noted. “This study was done in Canada, and the scale used was in nmol/L, while most labs in the U.S. use ng/mL,” he said. “Most pediatric and adolescent societies in the United States use < 20 ng/mL as an indicator of vitamin D deficient and between 20 and 29 ng/mL as insufficient,” he explained, citing the position statement on recommended vitamin D intake for adolescents published by The Society for Adolescent Health and Medicine. In this study, the results converted to < 12 ng/mL as deficient and between 12 and 20 ng/mL as insufficient, respectively, on the U.S. scale, said Dr. Cooper.
“Therefore, I can see that there are cases where someone may have been labeled vitamin D insufficient in this study using their range, whereas in the U.S. these patients would be labeled as vitamin D deficient and treated with higher-dose supplementation,” he said. In addition, individuals with levels between 20 ng/mL and 29 ng/mL in the U.S. would still be treated with vitamin D supplementation, “whereas in their study those individuals would have been labeled as normal,” he noted.
As for future research, it would be useful to study whether bone mass in transgender young people differs from age- and gender-matched controls who are not gender diverse (cisgender), Dr. Cooper added. “It may be possible that the youth in this study are not different from their peers and maybe the GnRH agonist is not the culprit,” he said.
The study received no outside funding. The researchers, editorial authors, and Dr. Cooper had no financial conflicts to disclose.
The use of gonadotropin-releasing hormone agonists has a negative effect on bone mass in transgender youth, according to data from 172 individuals.
The onset of puberty and pubertal hormones contributes to the development of bone mass and body composition in adolescence, wrote Behdad Navabi, MD, and colleagues at Children’s Hospital of Eastern Ontario, Canada. Although the safety and efficacy of gonadotropin-releasing hormone agonists (GnRHa) has been described in short-term studies of youth with gender dysphoria, concerns persist about suppression of bone mass accrual from extended use of GnRHas in this population, they noted.
In a study published in Pediatrics, the researchers reviewed data from 172 youth younger than 18 years of age who were treated with GNRHa and underwent at least one baseline dual-energy radiograph absorptiometry (DXA) measurement between January 2006 and April 2017 at a single center. The standard treatment protocol started with three doses of 7.5 mg leuprolide acetate, given intramuscularly every 4 weeks, followed by 11.25 mg intramuscularly every 12 weeks after puberty suppression was confirmed both clinically and biochemically. Areal bone mineral density (aBMD) measurement z scores were based on birth-assigned sex, age, and ethnicity, and assessed at baseline and every 12 months. In addition, volumetric bone mineral density was calculated as bone mineral apparent density (BMAD) at the lower spine, and the z score based on age-matched, birth-assigned gender BMAD.
Overall, 55.2% of the youth were vitamin D deficient or insufficient at baseline, but 87.3% were sufficient by the time of a third follow-up visit after treatment with 1,000-2,000 IU of vitamin D daily; no cases of vitamin D toxicity were reported.
At baseline, transgender females had lower z scores for the LS aBMD and BMAD compared to transgender males, reflecting a difference seen in previous studies of transgender youth and adult females, the researchers noted.
The researchers analyzed pre- and posttreatment DXA data in a subgroup of 36 transgender females and 80 transgender males to identify any changes associated with GnRHa. The average time between the DXA scans was 407 days. In this population, aBMD z scores at the lower lumbar spine (LS), left total hip (LTH), and total body less head (TBLH) decreased significantly from baseline in transgender males and females.
Among transgender males, LS bone mineral apparent density (BMAD) z scores also decreased significantly from baseline, but no such change occurred among transgender females. The most significant decrease in z scores occurred in the LS aBMD and BMAD of transgender males, with changes that reflect findings from previous studies and may be explained by decreased estrogen, the researchers wrote.
In terms of body composition, no significant changes occurred in body mass index z score from baseline to follow-up in transgender males or females, the researchers noted, and changes in both gynoid and android fat percentages were consistent with the individuals’ affirmed genders. No vertebral fractures were detected.
However, GnRHa was significantly associated with a decrease in total body fat percentage and a decrease in lean body mass (LBM) in transgender females.
The study findings were limited by several factors, including the lack of consistent baseline physical activity records, and limited analysis at follow-up of the possible role of physical activity in bone health and body composition, the researchers noted. However, the results were strengthened by the relatively large study population with baseline assessments, and by the pre- and posttreatment analysis, they added.
“Evidence on GnRHa-associated changes in body composition and BMD will help health care professionals involved in the care of youth with GD [gender dysphoria] to counsel appropriately and optimize their bone health,” the researchers said. “Given the absence of vertebral fractures detected in those with significant decreases in their LS z scores, the significance of BMD effects of GnRHa in transgender youth needs further study, as well as whether future spine radiographs are needed on the basis of BMD trajectory,” they concluded.
Balance bone health concerns with potential benefits
The effect of estrogen and testosterone on bone geometry in puberty varies, and the increase in the use of GnRHa as part of a multidisciplinary gender transition plan makes research on the skeletal impact of this therapy in transgender youth a top priority, Laura K. Bachrach, MD, of Stanford (Calif.) University, and Catherine M. Gordon of Harvard Medical School, Boston, wrote in an accompanying editorial.
The decrease in areal bone mineral density and in bone mineral apparent density (BMAD) z scores in the current study is not unexpected, but the key question is how much bone density recovers once the suppression therapy ends and transgender sex steroid use begins, they said. “Follow-up studies of young adults treated with GnRHa for precocious puberty in childhood are reassuring,” they wrote. “It is premature, however, to extrapolate from these findings to transgender youth,” because the impact of gender-affirming sex steroid therapy on the skeleton at older ages and stages of maturity are unclear, they emphasized.
In the absence of definitive answers, the editorial authors advised clinicians treating youth with gender dysphoria to provide a balanced view of the risks and benefits of hormone therapy, and encourage adequate intake of dietary vitamin D and calcium, along with weight-bearing physical activity, to promote general bone health. “Transgender teenagers and their parents should be reassured that some recovery from decreases in aBMD during pubertal suppression with GnRHa is likely,” the authors noted. Bone health should be monitored throughout all stages of treatment in transgender youth, but concerns about transient bone loss should not discourage gender transition therapy, they emphasized. “In this patient group, providing a pause in pubertal development offers a life-changing and, for some, a life-saving intervention,” they concluded.
Comparison to cisgender controls would add value
“This study is important because one of the major side effects of GnRH agonists is decreased bone density, especially the longer that patients are on them,” M. Brett Cooper, MD, of UT Southwestern Medical Center, said in an interview. The findings add to existing data to underscore the importance of screening for low bone density and low vitamin D levels, Dr. Cooper added.
Dr. Cooper said that he was not surprised by the study findings. “I think that this study supported what clinicians already knew, which is that GnRH agonists do potentially cause a decline in bone mineral density and thus, you need to support these patients as best you can with calcium, vitamin D, and weight-bearing exercise,” he noted.
Dr. Cooper emphasized two main take-home points from the study. “First, clinicians who prescribe GnRH agonists need to ensure that they are checking bone density and vitamin D measurements, and then optimizing these appropriately,” he said. “Second, when a bone density is found to be low or a vitamin level is low, clinicians need to ensure that they are monitored and treated appropriately.” Clinicians need to use these data when deciding when to start gender-affirming hormones so their patients have the best chance to recover bone density, he added.
“I think one confounding factor on this study is the ranges they used for vitamin D deficiency,” Dr. Cooper noted. “This study was done in Canada, and the scale used was in nmol/L, while most labs in the U.S. use ng/mL,” he said. “Most pediatric and adolescent societies in the United States use < 20 ng/mL as an indicator of vitamin D deficient and between 20 and 29 ng/mL as insufficient,” he explained, citing the position statement on recommended vitamin D intake for adolescents published by The Society for Adolescent Health and Medicine. In this study, the results converted to < 12 ng/mL as deficient and between 12 and 20 ng/mL as insufficient, respectively, on the U.S. scale, said Dr. Cooper.
“Therefore, I can see that there are cases where someone may have been labeled vitamin D insufficient in this study using their range, whereas in the U.S. these patients would be labeled as vitamin D deficient and treated with higher-dose supplementation,” he said. In addition, individuals with levels between 20 ng/mL and 29 ng/mL in the U.S. would still be treated with vitamin D supplementation, “whereas in their study those individuals would have been labeled as normal,” he noted.
As for future research, it would be useful to study whether bone mass in transgender young people differs from age- and gender-matched controls who are not gender diverse (cisgender), Dr. Cooper added. “It may be possible that the youth in this study are not different from their peers and maybe the GnRH agonist is not the culprit,” he said.
The study received no outside funding. The researchers, editorial authors, and Dr. Cooper had no financial conflicts to disclose.
The use of gonadotropin-releasing hormone agonists has a negative effect on bone mass in transgender youth, according to data from 172 individuals.
The onset of puberty and pubertal hormones contributes to the development of bone mass and body composition in adolescence, wrote Behdad Navabi, MD, and colleagues at Children’s Hospital of Eastern Ontario, Canada. Although the safety and efficacy of gonadotropin-releasing hormone agonists (GnRHa) has been described in short-term studies of youth with gender dysphoria, concerns persist about suppression of bone mass accrual from extended use of GnRHas in this population, they noted.
In a study published in Pediatrics, the researchers reviewed data from 172 youth younger than 18 years of age who were treated with GNRHa and underwent at least one baseline dual-energy radiograph absorptiometry (DXA) measurement between January 2006 and April 2017 at a single center. The standard treatment protocol started with three doses of 7.5 mg leuprolide acetate, given intramuscularly every 4 weeks, followed by 11.25 mg intramuscularly every 12 weeks after puberty suppression was confirmed both clinically and biochemically. Areal bone mineral density (aBMD) measurement z scores were based on birth-assigned sex, age, and ethnicity, and assessed at baseline and every 12 months. In addition, volumetric bone mineral density was calculated as bone mineral apparent density (BMAD) at the lower spine, and the z score based on age-matched, birth-assigned gender BMAD.
Overall, 55.2% of the youth were vitamin D deficient or insufficient at baseline, but 87.3% were sufficient by the time of a third follow-up visit after treatment with 1,000-2,000 IU of vitamin D daily; no cases of vitamin D toxicity were reported.
At baseline, transgender females had lower z scores for the LS aBMD and BMAD compared to transgender males, reflecting a difference seen in previous studies of transgender youth and adult females, the researchers noted.
The researchers analyzed pre- and posttreatment DXA data in a subgroup of 36 transgender females and 80 transgender males to identify any changes associated with GnRHa. The average time between the DXA scans was 407 days. In this population, aBMD z scores at the lower lumbar spine (LS), left total hip (LTH), and total body less head (TBLH) decreased significantly from baseline in transgender males and females.
Among transgender males, LS bone mineral apparent density (BMAD) z scores also decreased significantly from baseline, but no such change occurred among transgender females. The most significant decrease in z scores occurred in the LS aBMD and BMAD of transgender males, with changes that reflect findings from previous studies and may be explained by decreased estrogen, the researchers wrote.
In terms of body composition, no significant changes occurred in body mass index z score from baseline to follow-up in transgender males or females, the researchers noted, and changes in both gynoid and android fat percentages were consistent with the individuals’ affirmed genders. No vertebral fractures were detected.
However, GnRHa was significantly associated with a decrease in total body fat percentage and a decrease in lean body mass (LBM) in transgender females.
The study findings were limited by several factors, including the lack of consistent baseline physical activity records, and limited analysis at follow-up of the possible role of physical activity in bone health and body composition, the researchers noted. However, the results were strengthened by the relatively large study population with baseline assessments, and by the pre- and posttreatment analysis, they added.
“Evidence on GnRHa-associated changes in body composition and BMD will help health care professionals involved in the care of youth with GD [gender dysphoria] to counsel appropriately and optimize their bone health,” the researchers said. “Given the absence of vertebral fractures detected in those with significant decreases in their LS z scores, the significance of BMD effects of GnRHa in transgender youth needs further study, as well as whether future spine radiographs are needed on the basis of BMD trajectory,” they concluded.
Balance bone health concerns with potential benefits
The effect of estrogen and testosterone on bone geometry in puberty varies, and the increase in the use of GnRHa as part of a multidisciplinary gender transition plan makes research on the skeletal impact of this therapy in transgender youth a top priority, Laura K. Bachrach, MD, of Stanford (Calif.) University, and Catherine M. Gordon of Harvard Medical School, Boston, wrote in an accompanying editorial.
The decrease in areal bone mineral density and in bone mineral apparent density (BMAD) z scores in the current study is not unexpected, but the key question is how much bone density recovers once the suppression therapy ends and transgender sex steroid use begins, they said. “Follow-up studies of young adults treated with GnRHa for precocious puberty in childhood are reassuring,” they wrote. “It is premature, however, to extrapolate from these findings to transgender youth,” because the impact of gender-affirming sex steroid therapy on the skeleton at older ages and stages of maturity are unclear, they emphasized.
In the absence of definitive answers, the editorial authors advised clinicians treating youth with gender dysphoria to provide a balanced view of the risks and benefits of hormone therapy, and encourage adequate intake of dietary vitamin D and calcium, along with weight-bearing physical activity, to promote general bone health. “Transgender teenagers and their parents should be reassured that some recovery from decreases in aBMD during pubertal suppression with GnRHa is likely,” the authors noted. Bone health should be monitored throughout all stages of treatment in transgender youth, but concerns about transient bone loss should not discourage gender transition therapy, they emphasized. “In this patient group, providing a pause in pubertal development offers a life-changing and, for some, a life-saving intervention,” they concluded.
Comparison to cisgender controls would add value
“This study is important because one of the major side effects of GnRH agonists is decreased bone density, especially the longer that patients are on them,” M. Brett Cooper, MD, of UT Southwestern Medical Center, said in an interview. The findings add to existing data to underscore the importance of screening for low bone density and low vitamin D levels, Dr. Cooper added.
Dr. Cooper said that he was not surprised by the study findings. “I think that this study supported what clinicians already knew, which is that GnRH agonists do potentially cause a decline in bone mineral density and thus, you need to support these patients as best you can with calcium, vitamin D, and weight-bearing exercise,” he noted.
Dr. Cooper emphasized two main take-home points from the study. “First, clinicians who prescribe GnRH agonists need to ensure that they are checking bone density and vitamin D measurements, and then optimizing these appropriately,” he said. “Second, when a bone density is found to be low or a vitamin level is low, clinicians need to ensure that they are monitored and treated appropriately.” Clinicians need to use these data when deciding when to start gender-affirming hormones so their patients have the best chance to recover bone density, he added.
“I think one confounding factor on this study is the ranges they used for vitamin D deficiency,” Dr. Cooper noted. “This study was done in Canada, and the scale used was in nmol/L, while most labs in the U.S. use ng/mL,” he said. “Most pediatric and adolescent societies in the United States use < 20 ng/mL as an indicator of vitamin D deficient and between 20 and 29 ng/mL as insufficient,” he explained, citing the position statement on recommended vitamin D intake for adolescents published by The Society for Adolescent Health and Medicine. In this study, the results converted to < 12 ng/mL as deficient and between 12 and 20 ng/mL as insufficient, respectively, on the U.S. scale, said Dr. Cooper.
“Therefore, I can see that there are cases where someone may have been labeled vitamin D insufficient in this study using their range, whereas in the U.S. these patients would be labeled as vitamin D deficient and treated with higher-dose supplementation,” he said. In addition, individuals with levels between 20 ng/mL and 29 ng/mL in the U.S. would still be treated with vitamin D supplementation, “whereas in their study those individuals would have been labeled as normal,” he noted.
As for future research, it would be useful to study whether bone mass in transgender young people differs from age- and gender-matched controls who are not gender diverse (cisgender), Dr. Cooper added. “It may be possible that the youth in this study are not different from their peers and maybe the GnRH agonist is not the culprit,” he said.
The study received no outside funding. The researchers, editorial authors, and Dr. Cooper had no financial conflicts to disclose.
FROM PEDIATRICS
Sacral nerve root endometriosis

Additional videos from SGS are available here, including these recent offerings:

Additional videos from SGS are available here, including these recent offerings:

Additional videos from SGS are available here, including these recent offerings: