User login
Drug-coated balloons: The future of hemodialysis access?
CHICAGO – Drug-coated balloons show promise of being a long-sought major advance in the endovascular treatment of stenotic arteriovenous fistulae and grafts for hemodialysis access, Syed M. Hussain, MD, said at a symposium on vascular surgery sponsored by Northwestern University.
Something significantly better than today’s standard treatment options is needed, according to Dr. Hussain. Medicare pays out more than $50 billion annually for the treatment of patients with end-stage renal disease, and a hefty chunk of that money goes for oft-repeated procedures aimed at preserving the patency of the access sites.
“Primary patency rates leave much room for improvement,” observed Dr. Hussain, a vascular surgeon at the Christie Clinic in Champaign, Ill.
Indeed, the 50% primary patency rate at 6 months that was optimistically declared a “reasonable goal” in the 2006 Kidney Disease Outcomes Quality Initiative is actually far-fetched using the conventional tools.
“That 50% patency at 6 months would be a tall order to try to meet. Anybody in this room that does fistulography and angioplasty knows the numbers are actually a lot lower than 50%,” said Dr. Hussain.
Plain old balloon angioplasty, the standard first-line intervention for stenotic hemodialysis access sites, has a 6-month patency rate of about 30%. Bare metal stents push the rate up to about 39%. Covered stent grafts are the most effective of the conventional treatment modalities, with a 6-month patency of 51%-53%; however, they are widely considered too expensive for routine use.
Drug-coated balloons (DCBs) have been available for close to 3 years for treatment of lower extremity peripheral artery disease, where they have achieved considerable success. The Food and Drug Administration has approved three commercially available DCBs for this purpose: Bard’s Lutonix 035 AV, Medtronic’s IN.PACT Admiral, and most recently the Stellarex DCB.
In addition, the Lutonix DCB is approved for treatment of dysfunctional or stenotic arteriovenous (AV) fistulae on the strength of the positive results of the first prospective randomized multicenter trial of a DCB versus balloon angioplasty for AV access stenosis as reported at a conference in Leipzig, Germany, in 2017 and summarized by Dr. Hussain.
The pathophysiology of arterial atherosclerotic stenosis is very different from the stenosis that plagues AV access for dialysis. Arterial atherosclerotic stenosis is due to neointimal hyperplasia caused by inflammation and barotrauma secondary to angioplasty. In contrast, the neointimal hyperplasia in AV access stenosis is due to smooth muscle cell proliferation in response to nonphysiologic blood flow dynamics and shear forces between a high-pressure arterial system and the low-pressure venous system to which it has been connected, with resultant stenosis at the venous outflow anastomosis and often at the cephalic arch, Dr. Hussain explained.
Other contributors to the high rate of early stenosis in AV fistulae and grafts include traumatic balloon dilation, uremia, and repetitive traumatic needle insertion.
The breakthrough for DCBs as a potential game changer in dialysis access stenosis came with the discovery that venous smooth muscle cells are much more sensitive to paclitaxel and other antiproliferative drugs than are arterial smooth muscle cells. All three commercially available DCBs utilize paclitaxel as their active agent.
Multiple small single-center studies involving off-label use of the DCBs for dialysis access stenosis strongly suggested 6-month patency rates were higher than with balloon angioplasty. Then came the core lab-adjudicated Lutonix multicenter trial, in which 285 hemodialysis patients at 23 sites were randomized to the DCB or balloon angioplasty. Participants had to have a target lesion less than 10 cm long and had to undergo successful predilatation with high-pressure balloon angioplasty.
“The key thing to remember when we talk about dialysis grafts or fistulae is that we have to look at patency in periods of months. We can’t look at years because it’s pretty unusual to see a fistula stay open that long. So most of the time we’re trying to achieve extra months on these types of circuits,” noted Dr. Hussain.
That being said, the 8-month target lesion primary patency rate was 61.6% in the Lutonix DCB group, compared with 49.4% for percutaneous angioplasty, a statistically significant and clinically meaningful difference. Moreover, 66 interventions were required to maintain target lesion patency during that time frame in the DCB group, versus 94 in the angioplasty group; that translated to a 30% reduction in repeat interventions.
“This clearly has the potential to save a lot of money for the health care system,” he said.
The two forms of treatment were equally safe.
The expanded indication for the Lutonix DCB that resulted from this large randomized trial has triggered considerable research interest in DCBs for AV access stenosis around the world. Major ongoing randomized trials include the PAVE trial in the United Kingdom, the Spanish FISBOL trial, the APERTO trial in the Netherlands, and an Israeli randomized trial restricted to patients with cephalic arch stenosis.
Dr. Hussain is particularly excited about the ongoing 330-patient, prospective, multicenter, single-blinded clinical trial of the IN.PACT Admiral DCB versus plain balloon angioplasty. The Medtronic DCB employs a higher dosage of paclitaxel: 3.5 mcg/mm2, compared with 2.0 mcg/mm2 for the Lutonix DCB. Also, due to differences in the excipients used in the two DCBs, the paclitaxel from the IN.PACT device stays in the media of blood vessels for up to 180 days, compared with 60 days following drug delivery with the Lutonix balloon. Whether this longer period of close range antiproliferative activity will translate into a higher patency rate remains to be seen.
Dr. Hussain reported having no financial conflicts of interest regarding his presentation.
Costs associated with the management of patients with chronic kidney disease, particularly those with end-stage renal disease requiring hemodialysis, is a huge component of the Medicare budget. The maintenance of functional vascular access in these patients remains an ongoing challenge and reduction of costs related to access failure is critical to the continued funding of the program. Traditional methods of maintaining access patency such as balloon angioplasty and bare metal stents have poor long-term outcomes, and moderate improvement is seen with the use of covered stents.
Dr. Hussain reviews the current status of drug-coated balloons (DCB) in the endovascular treatment of dysfunctional hemodialysis fistulas and grafts. Safety and efficacy data from a prospective randomized multicenter trial comparing the Bard Lutonix DCB and plain old balloon angioplasty demonstrated a significant improvement in primary patency and a 30% reduction in repeat interventions with the DCB. This led to FDA approval for the Lutonix DCB in the treatment of dysfunctional or stenotic arteriovenous fistulas. Encouraged by these results, researchers conducting ongoing international randomized trials are attempting to clarify the potential expanded indications for DCB in access stenosis. Of particular interest is the ongoing 330-patient prospective, multicenter IN-PACT trial comparing Admiral DCB to balloon angioplasty in failing arteriovenous fistulas. Both the Admiral DCB and Lutonix DCB utilize paclitaxel as the antiproliferative agent. Dr. Hussain describes the increased sensitivity of venous smooth muscle cells to paclitaxel and other antiproliferative drugs when compared with arterial smooth muscle cells. This exciting finding may explain the improved outcomes in the treatment of dialysis access lesions where pathology frequently occurs at the venous anastomosis or in the venous conduit.
Although early results with the use of DCBs are promising, ongoing clinical trials and careful analysis of data and cost effectiveness are critical to optimize outcomes in treating dialysis access dysfunction. Dr. Hussain appropriately expresses cautious optimism regarding the future of hemodialysis access with this new tool available to interventionists treating these complex patients.
Larry A. Scher, MD, is a vascular surgeon at Montefiore Einstein Center for Heart and Vascular Care, New York, and an associate medical editor for Vascular Specialist.
Costs associated with the management of patients with chronic kidney disease, particularly those with end-stage renal disease requiring hemodialysis, is a huge component of the Medicare budget. The maintenance of functional vascular access in these patients remains an ongoing challenge and reduction of costs related to access failure is critical to the continued funding of the program. Traditional methods of maintaining access patency such as balloon angioplasty and bare metal stents have poor long-term outcomes, and moderate improvement is seen with the use of covered stents.
Dr. Hussain reviews the current status of drug-coated balloons (DCB) in the endovascular treatment of dysfunctional hemodialysis fistulas and grafts. Safety and efficacy data from a prospective randomized multicenter trial comparing the Bard Lutonix DCB and plain old balloon angioplasty demonstrated a significant improvement in primary patency and a 30% reduction in repeat interventions with the DCB. This led to FDA approval for the Lutonix DCB in the treatment of dysfunctional or stenotic arteriovenous fistulas. Encouraged by these results, researchers conducting ongoing international randomized trials are attempting to clarify the potential expanded indications for DCB in access stenosis. Of particular interest is the ongoing 330-patient prospective, multicenter IN-PACT trial comparing Admiral DCB to balloon angioplasty in failing arteriovenous fistulas. Both the Admiral DCB and Lutonix DCB utilize paclitaxel as the antiproliferative agent. Dr. Hussain describes the increased sensitivity of venous smooth muscle cells to paclitaxel and other antiproliferative drugs when compared with arterial smooth muscle cells. This exciting finding may explain the improved outcomes in the treatment of dialysis access lesions where pathology frequently occurs at the venous anastomosis or in the venous conduit.
Although early results with the use of DCBs are promising, ongoing clinical trials and careful analysis of data and cost effectiveness are critical to optimize outcomes in treating dialysis access dysfunction. Dr. Hussain appropriately expresses cautious optimism regarding the future of hemodialysis access with this new tool available to interventionists treating these complex patients.
Larry A. Scher, MD, is a vascular surgeon at Montefiore Einstein Center for Heart and Vascular Care, New York, and an associate medical editor for Vascular Specialist.
Costs associated with the management of patients with chronic kidney disease, particularly those with end-stage renal disease requiring hemodialysis, is a huge component of the Medicare budget. The maintenance of functional vascular access in these patients remains an ongoing challenge and reduction of costs related to access failure is critical to the continued funding of the program. Traditional methods of maintaining access patency such as balloon angioplasty and bare metal stents have poor long-term outcomes, and moderate improvement is seen with the use of covered stents.
Dr. Hussain reviews the current status of drug-coated balloons (DCB) in the endovascular treatment of dysfunctional hemodialysis fistulas and grafts. Safety and efficacy data from a prospective randomized multicenter trial comparing the Bard Lutonix DCB and plain old balloon angioplasty demonstrated a significant improvement in primary patency and a 30% reduction in repeat interventions with the DCB. This led to FDA approval for the Lutonix DCB in the treatment of dysfunctional or stenotic arteriovenous fistulas. Encouraged by these results, researchers conducting ongoing international randomized trials are attempting to clarify the potential expanded indications for DCB in access stenosis. Of particular interest is the ongoing 330-patient prospective, multicenter IN-PACT trial comparing Admiral DCB to balloon angioplasty in failing arteriovenous fistulas. Both the Admiral DCB and Lutonix DCB utilize paclitaxel as the antiproliferative agent. Dr. Hussain describes the increased sensitivity of venous smooth muscle cells to paclitaxel and other antiproliferative drugs when compared with arterial smooth muscle cells. This exciting finding may explain the improved outcomes in the treatment of dialysis access lesions where pathology frequently occurs at the venous anastomosis or in the venous conduit.
Although early results with the use of DCBs are promising, ongoing clinical trials and careful analysis of data and cost effectiveness are critical to optimize outcomes in treating dialysis access dysfunction. Dr. Hussain appropriately expresses cautious optimism regarding the future of hemodialysis access with this new tool available to interventionists treating these complex patients.
Larry A. Scher, MD, is a vascular surgeon at Montefiore Einstein Center for Heart and Vascular Care, New York, and an associate medical editor for Vascular Specialist.
CHICAGO – Drug-coated balloons show promise of being a long-sought major advance in the endovascular treatment of stenotic arteriovenous fistulae and grafts for hemodialysis access, Syed M. Hussain, MD, said at a symposium on vascular surgery sponsored by Northwestern University.
Something significantly better than today’s standard treatment options is needed, according to Dr. Hussain. Medicare pays out more than $50 billion annually for the treatment of patients with end-stage renal disease, and a hefty chunk of that money goes for oft-repeated procedures aimed at preserving the patency of the access sites.
“Primary patency rates leave much room for improvement,” observed Dr. Hussain, a vascular surgeon at the Christie Clinic in Champaign, Ill.
Indeed, the 50% primary patency rate at 6 months that was optimistically declared a “reasonable goal” in the 2006 Kidney Disease Outcomes Quality Initiative is actually far-fetched using the conventional tools.
“That 50% patency at 6 months would be a tall order to try to meet. Anybody in this room that does fistulography and angioplasty knows the numbers are actually a lot lower than 50%,” said Dr. Hussain.
Plain old balloon angioplasty, the standard first-line intervention for stenotic hemodialysis access sites, has a 6-month patency rate of about 30%. Bare metal stents push the rate up to about 39%. Covered stent grafts are the most effective of the conventional treatment modalities, with a 6-month patency of 51%-53%; however, they are widely considered too expensive for routine use.
Drug-coated balloons (DCBs) have been available for close to 3 years for treatment of lower extremity peripheral artery disease, where they have achieved considerable success. The Food and Drug Administration has approved three commercially available DCBs for this purpose: Bard’s Lutonix 035 AV, Medtronic’s IN.PACT Admiral, and most recently the Stellarex DCB.
In addition, the Lutonix DCB is approved for treatment of dysfunctional or stenotic arteriovenous (AV) fistulae on the strength of the positive results of the first prospective randomized multicenter trial of a DCB versus balloon angioplasty for AV access stenosis as reported at a conference in Leipzig, Germany, in 2017 and summarized by Dr. Hussain.
The pathophysiology of arterial atherosclerotic stenosis is very different from the stenosis that plagues AV access for dialysis. Arterial atherosclerotic stenosis is due to neointimal hyperplasia caused by inflammation and barotrauma secondary to angioplasty. In contrast, the neointimal hyperplasia in AV access stenosis is due to smooth muscle cell proliferation in response to nonphysiologic blood flow dynamics and shear forces between a high-pressure arterial system and the low-pressure venous system to which it has been connected, with resultant stenosis at the venous outflow anastomosis and often at the cephalic arch, Dr. Hussain explained.
Other contributors to the high rate of early stenosis in AV fistulae and grafts include traumatic balloon dilation, uremia, and repetitive traumatic needle insertion.
The breakthrough for DCBs as a potential game changer in dialysis access stenosis came with the discovery that venous smooth muscle cells are much more sensitive to paclitaxel and other antiproliferative drugs than are arterial smooth muscle cells. All three commercially available DCBs utilize paclitaxel as their active agent.
Multiple small single-center studies involving off-label use of the DCBs for dialysis access stenosis strongly suggested 6-month patency rates were higher than with balloon angioplasty. Then came the core lab-adjudicated Lutonix multicenter trial, in which 285 hemodialysis patients at 23 sites were randomized to the DCB or balloon angioplasty. Participants had to have a target lesion less than 10 cm long and had to undergo successful predilatation with high-pressure balloon angioplasty.
“The key thing to remember when we talk about dialysis grafts or fistulae is that we have to look at patency in periods of months. We can’t look at years because it’s pretty unusual to see a fistula stay open that long. So most of the time we’re trying to achieve extra months on these types of circuits,” noted Dr. Hussain.
That being said, the 8-month target lesion primary patency rate was 61.6% in the Lutonix DCB group, compared with 49.4% for percutaneous angioplasty, a statistically significant and clinically meaningful difference. Moreover, 66 interventions were required to maintain target lesion patency during that time frame in the DCB group, versus 94 in the angioplasty group; that translated to a 30% reduction in repeat interventions.
“This clearly has the potential to save a lot of money for the health care system,” he said.
The two forms of treatment were equally safe.
The expanded indication for the Lutonix DCB that resulted from this large randomized trial has triggered considerable research interest in DCBs for AV access stenosis around the world. Major ongoing randomized trials include the PAVE trial in the United Kingdom, the Spanish FISBOL trial, the APERTO trial in the Netherlands, and an Israeli randomized trial restricted to patients with cephalic arch stenosis.
Dr. Hussain is particularly excited about the ongoing 330-patient, prospective, multicenter, single-blinded clinical trial of the IN.PACT Admiral DCB versus plain balloon angioplasty. The Medtronic DCB employs a higher dosage of paclitaxel: 3.5 mcg/mm2, compared with 2.0 mcg/mm2 for the Lutonix DCB. Also, due to differences in the excipients used in the two DCBs, the paclitaxel from the IN.PACT device stays in the media of blood vessels for up to 180 days, compared with 60 days following drug delivery with the Lutonix balloon. Whether this longer period of close range antiproliferative activity will translate into a higher patency rate remains to be seen.
Dr. Hussain reported having no financial conflicts of interest regarding his presentation.
CHICAGO – Drug-coated balloons show promise of being a long-sought major advance in the endovascular treatment of stenotic arteriovenous fistulae and grafts for hemodialysis access, Syed M. Hussain, MD, said at a symposium on vascular surgery sponsored by Northwestern University.
Something significantly better than today’s standard treatment options is needed, according to Dr. Hussain. Medicare pays out more than $50 billion annually for the treatment of patients with end-stage renal disease, and a hefty chunk of that money goes for oft-repeated procedures aimed at preserving the patency of the access sites.
“Primary patency rates leave much room for improvement,” observed Dr. Hussain, a vascular surgeon at the Christie Clinic in Champaign, Ill.
Indeed, the 50% primary patency rate at 6 months that was optimistically declared a “reasonable goal” in the 2006 Kidney Disease Outcomes Quality Initiative is actually far-fetched using the conventional tools.
“That 50% patency at 6 months would be a tall order to try to meet. Anybody in this room that does fistulography and angioplasty knows the numbers are actually a lot lower than 50%,” said Dr. Hussain.
Plain old balloon angioplasty, the standard first-line intervention for stenotic hemodialysis access sites, has a 6-month patency rate of about 30%. Bare metal stents push the rate up to about 39%. Covered stent grafts are the most effective of the conventional treatment modalities, with a 6-month patency of 51%-53%; however, they are widely considered too expensive for routine use.
Drug-coated balloons (DCBs) have been available for close to 3 years for treatment of lower extremity peripheral artery disease, where they have achieved considerable success. The Food and Drug Administration has approved three commercially available DCBs for this purpose: Bard’s Lutonix 035 AV, Medtronic’s IN.PACT Admiral, and most recently the Stellarex DCB.
In addition, the Lutonix DCB is approved for treatment of dysfunctional or stenotic arteriovenous (AV) fistulae on the strength of the positive results of the first prospective randomized multicenter trial of a DCB versus balloon angioplasty for AV access stenosis as reported at a conference in Leipzig, Germany, in 2017 and summarized by Dr. Hussain.
The pathophysiology of arterial atherosclerotic stenosis is very different from the stenosis that plagues AV access for dialysis. Arterial atherosclerotic stenosis is due to neointimal hyperplasia caused by inflammation and barotrauma secondary to angioplasty. In contrast, the neointimal hyperplasia in AV access stenosis is due to smooth muscle cell proliferation in response to nonphysiologic blood flow dynamics and shear forces between a high-pressure arterial system and the low-pressure venous system to which it has been connected, with resultant stenosis at the venous outflow anastomosis and often at the cephalic arch, Dr. Hussain explained.
Other contributors to the high rate of early stenosis in AV fistulae and grafts include traumatic balloon dilation, uremia, and repetitive traumatic needle insertion.
The breakthrough for DCBs as a potential game changer in dialysis access stenosis came with the discovery that venous smooth muscle cells are much more sensitive to paclitaxel and other antiproliferative drugs than are arterial smooth muscle cells. All three commercially available DCBs utilize paclitaxel as their active agent.
Multiple small single-center studies involving off-label use of the DCBs for dialysis access stenosis strongly suggested 6-month patency rates were higher than with balloon angioplasty. Then came the core lab-adjudicated Lutonix multicenter trial, in which 285 hemodialysis patients at 23 sites were randomized to the DCB or balloon angioplasty. Participants had to have a target lesion less than 10 cm long and had to undergo successful predilatation with high-pressure balloon angioplasty.
“The key thing to remember when we talk about dialysis grafts or fistulae is that we have to look at patency in periods of months. We can’t look at years because it’s pretty unusual to see a fistula stay open that long. So most of the time we’re trying to achieve extra months on these types of circuits,” noted Dr. Hussain.
That being said, the 8-month target lesion primary patency rate was 61.6% in the Lutonix DCB group, compared with 49.4% for percutaneous angioplasty, a statistically significant and clinically meaningful difference. Moreover, 66 interventions were required to maintain target lesion patency during that time frame in the DCB group, versus 94 in the angioplasty group; that translated to a 30% reduction in repeat interventions.
“This clearly has the potential to save a lot of money for the health care system,” he said.
The two forms of treatment were equally safe.
The expanded indication for the Lutonix DCB that resulted from this large randomized trial has triggered considerable research interest in DCBs for AV access stenosis around the world. Major ongoing randomized trials include the PAVE trial in the United Kingdom, the Spanish FISBOL trial, the APERTO trial in the Netherlands, and an Israeli randomized trial restricted to patients with cephalic arch stenosis.
Dr. Hussain is particularly excited about the ongoing 330-patient, prospective, multicenter, single-blinded clinical trial of the IN.PACT Admiral DCB versus plain balloon angioplasty. The Medtronic DCB employs a higher dosage of paclitaxel: 3.5 mcg/mm2, compared with 2.0 mcg/mm2 for the Lutonix DCB. Also, due to differences in the excipients used in the two DCBs, the paclitaxel from the IN.PACT device stays in the media of blood vessels for up to 180 days, compared with 60 days following drug delivery with the Lutonix balloon. Whether this longer period of close range antiproliferative activity will translate into a higher patency rate remains to be seen.
Dr. Hussain reported having no financial conflicts of interest regarding his presentation.
EXPERT ANALYSIS FROM THE NORTHWESTERN VASCULAR SYMPOSIUM
NHLBI seeks to accelerate hemostasis/thrombosis research
SAN DIEGO – The National Heart, Lung, and Blood Institute is looking to jump-start research into hemostasis and thrombosis. Donna DiMichele, MD, offered two reasons for the research push.
“The first is to stimulate research in areas that are undersubscribed through investigator-initiated research,” Dr. DiMichele, deputy director of the NHLBI’s Division of Blood Diseases and Resources, said at the biennial summit of the Thrombosis & Hemostasis Societies of North America. “We also do it sometimes to steer research in directions that are not developing organically through investigator-initiated efforts. In hemostasis and thrombosis, 50% of the investigator-led research is basic in nature. Nonetheless, there were several basic research initiatives that NHLBI released in the last few years that stimulated the field in a new direction.”
The centers will be required to look beyond current active science disciplines in this field, to include emerging sciences and technologies not currently being exploited in this research area. The initiative is also intended to cross-train the next generation of physicians/scientists with interdisciplinary skill sets. “The research teams with successful applications are going to begin their work by the summer of 2018, and hopefully we’ll soon gain some new insights into Factor VIII immunogenicity,” Dr. DiMichele said.
One novel scientific area that successful applicants will be able to take advantage of is the Trans-Omics for Precision Medicine Program (TOPMed). Launched in 2014, this program facilitates whole-genome sequencing across cohorts in heart, lung, blood, and sleep science. TOPMed now includes more than 120,000 whole genomes, with more than 5,000 of those in hemophilia.
In basic science, two NHLBI-funded initiatives aim to improve the understanding of thrombosis.
One, Sex Hormone Induced Thromboembolism in Premenopausal Women (R61/R33), is meant to elucidate the mechanisms by which female sex hormones and sex hormone-based therapies can increase the risk of venous and arterial thromboembolism in premenopausal women. The other initiative, Consortium Linking Oncology with Thrombosis (U01), is aimed at encouraging studies in individual cancer types that expand investigation into the intersection between cancer and thrombotic pathways. It also aims to help researchers identify and develop biomarkers of thrombotic risk or cancer progression, and new strategies for preventing or treating the deleterious interplay between cancer, cancer therapy, and hemostasis/thrombosis.
In the translational research arena, the NHLBI released an initiative on Perinatal Stroke (R01) in 2017. It solicits applications that propose basic and/or translational research studies related to the developing neurovascular unit, perinatal injury/repair response, and/or stroke-related etiologies and risk factors. The purpose is to stimulate research that will identify therapeutic targets in perinatal stroke.
Other NHLBI-sponsored efforts include the Trans-Agency Research Consortium for Trauma-Induced Coagulopathy (TACTIC), as well as the Translational Research Centers in Thrombotic and Hemostatic Disorders program, which set out to enhance the translation of basic research discoveries that could lead to improved prevention, diagnosis, and treatment for thrombotic and hemostatic disorders. This program ended but it led to the development of several projects that are moving into the commercialization phase.
Finally, to further stimulate research in FVIII immunogenicity, on May 15-16, 2018, the NHBLI will host a free workshop entitled “Factor VIII Inhibitors: Generating a Blueprint for Future Research.” For information, visit https://factorviiinhibitors.eventbrite.com.
Dr. DiMichele reported having no financial disclosures.
SAN DIEGO – The National Heart, Lung, and Blood Institute is looking to jump-start research into hemostasis and thrombosis. Donna DiMichele, MD, offered two reasons for the research push.
“The first is to stimulate research in areas that are undersubscribed through investigator-initiated research,” Dr. DiMichele, deputy director of the NHLBI’s Division of Blood Diseases and Resources, said at the biennial summit of the Thrombosis & Hemostasis Societies of North America. “We also do it sometimes to steer research in directions that are not developing organically through investigator-initiated efforts. In hemostasis and thrombosis, 50% of the investigator-led research is basic in nature. Nonetheless, there were several basic research initiatives that NHLBI released in the last few years that stimulated the field in a new direction.”
The centers will be required to look beyond current active science disciplines in this field, to include emerging sciences and technologies not currently being exploited in this research area. The initiative is also intended to cross-train the next generation of physicians/scientists with interdisciplinary skill sets. “The research teams with successful applications are going to begin their work by the summer of 2018, and hopefully we’ll soon gain some new insights into Factor VIII immunogenicity,” Dr. DiMichele said.
One novel scientific area that successful applicants will be able to take advantage of is the Trans-Omics for Precision Medicine Program (TOPMed). Launched in 2014, this program facilitates whole-genome sequencing across cohorts in heart, lung, blood, and sleep science. TOPMed now includes more than 120,000 whole genomes, with more than 5,000 of those in hemophilia.
In basic science, two NHLBI-funded initiatives aim to improve the understanding of thrombosis.
One, Sex Hormone Induced Thromboembolism in Premenopausal Women (R61/R33), is meant to elucidate the mechanisms by which female sex hormones and sex hormone-based therapies can increase the risk of venous and arterial thromboembolism in premenopausal women. The other initiative, Consortium Linking Oncology with Thrombosis (U01), is aimed at encouraging studies in individual cancer types that expand investigation into the intersection between cancer and thrombotic pathways. It also aims to help researchers identify and develop biomarkers of thrombotic risk or cancer progression, and new strategies for preventing or treating the deleterious interplay between cancer, cancer therapy, and hemostasis/thrombosis.
In the translational research arena, the NHLBI released an initiative on Perinatal Stroke (R01) in 2017. It solicits applications that propose basic and/or translational research studies related to the developing neurovascular unit, perinatal injury/repair response, and/or stroke-related etiologies and risk factors. The purpose is to stimulate research that will identify therapeutic targets in perinatal stroke.
Other NHLBI-sponsored efforts include the Trans-Agency Research Consortium for Trauma-Induced Coagulopathy (TACTIC), as well as the Translational Research Centers in Thrombotic and Hemostatic Disorders program, which set out to enhance the translation of basic research discoveries that could lead to improved prevention, diagnosis, and treatment for thrombotic and hemostatic disorders. This program ended but it led to the development of several projects that are moving into the commercialization phase.
Finally, to further stimulate research in FVIII immunogenicity, on May 15-16, 2018, the NHBLI will host a free workshop entitled “Factor VIII Inhibitors: Generating a Blueprint for Future Research.” For information, visit https://factorviiinhibitors.eventbrite.com.
Dr. DiMichele reported having no financial disclosures.
SAN DIEGO – The National Heart, Lung, and Blood Institute is looking to jump-start research into hemostasis and thrombosis. Donna DiMichele, MD, offered two reasons for the research push.
“The first is to stimulate research in areas that are undersubscribed through investigator-initiated research,” Dr. DiMichele, deputy director of the NHLBI’s Division of Blood Diseases and Resources, said at the biennial summit of the Thrombosis & Hemostasis Societies of North America. “We also do it sometimes to steer research in directions that are not developing organically through investigator-initiated efforts. In hemostasis and thrombosis, 50% of the investigator-led research is basic in nature. Nonetheless, there were several basic research initiatives that NHLBI released in the last few years that stimulated the field in a new direction.”
The centers will be required to look beyond current active science disciplines in this field, to include emerging sciences and technologies not currently being exploited in this research area. The initiative is also intended to cross-train the next generation of physicians/scientists with interdisciplinary skill sets. “The research teams with successful applications are going to begin their work by the summer of 2018, and hopefully we’ll soon gain some new insights into Factor VIII immunogenicity,” Dr. DiMichele said.
One novel scientific area that successful applicants will be able to take advantage of is the Trans-Omics for Precision Medicine Program (TOPMed). Launched in 2014, this program facilitates whole-genome sequencing across cohorts in heart, lung, blood, and sleep science. TOPMed now includes more than 120,000 whole genomes, with more than 5,000 of those in hemophilia.
In basic science, two NHLBI-funded initiatives aim to improve the understanding of thrombosis.
One, Sex Hormone Induced Thromboembolism in Premenopausal Women (R61/R33), is meant to elucidate the mechanisms by which female sex hormones and sex hormone-based therapies can increase the risk of venous and arterial thromboembolism in premenopausal women. The other initiative, Consortium Linking Oncology with Thrombosis (U01), is aimed at encouraging studies in individual cancer types that expand investigation into the intersection between cancer and thrombotic pathways. It also aims to help researchers identify and develop biomarkers of thrombotic risk or cancer progression, and new strategies for preventing or treating the deleterious interplay between cancer, cancer therapy, and hemostasis/thrombosis.
In the translational research arena, the NHLBI released an initiative on Perinatal Stroke (R01) in 2017. It solicits applications that propose basic and/or translational research studies related to the developing neurovascular unit, perinatal injury/repair response, and/or stroke-related etiologies and risk factors. The purpose is to stimulate research that will identify therapeutic targets in perinatal stroke.
Other NHLBI-sponsored efforts include the Trans-Agency Research Consortium for Trauma-Induced Coagulopathy (TACTIC), as well as the Translational Research Centers in Thrombotic and Hemostatic Disorders program, which set out to enhance the translation of basic research discoveries that could lead to improved prevention, diagnosis, and treatment for thrombotic and hemostatic disorders. This program ended but it led to the development of several projects that are moving into the commercialization phase.
Finally, to further stimulate research in FVIII immunogenicity, on May 15-16, 2018, the NHBLI will host a free workshop entitled “Factor VIII Inhibitors: Generating a Blueprint for Future Research.” For information, visit https://factorviiinhibitors.eventbrite.com.
Dr. DiMichele reported having no financial disclosures.
EXPERT ANALYSIS FROM THSNA 2018
Managing menopausal symptoms in breast cancer survivors
MDedge Daily News: Fewer smokes mean fewer strokes
Are biopsy’s days numbered for diagnosing celiac disease? The perils of endoscopic therapy for Barrett’s esophagus. And is your EHR preventing breastfeeding?
Listen to the MDedge Daily News podcast for all the details on today’s top news.
Are biopsy’s days numbered for diagnosing celiac disease? The perils of endoscopic therapy for Barrett’s esophagus. And is your EHR preventing breastfeeding?
Listen to the MDedge Daily News podcast for all the details on today’s top news.
Are biopsy’s days numbered for diagnosing celiac disease? The perils of endoscopic therapy for Barrett’s esophagus. And is your EHR preventing breastfeeding?
Listen to the MDedge Daily News podcast for all the details on today’s top news.
What Makes Squamous Cell Cancers Different? Genomics May Explain
Squamous cell carcinomas (SCCs) associated with smoking and human papillomavirus (HPV) have distinct genomic signatures, say researchers from a National Institutes of Health-supported study. That is one of the findings that may help distinguish SCCs from other cancers and point the way to new research and treatment.
The researchers used new analytic tools and data from the recently completed PanCancer Atlas to investigate similarities and differences among SCCs in the head and neck, lung, esophagus, cervix, and bladder. The PanCancer Atlas is a detailed analysis from a dataset containing molecular and clinical information on more than 10,000 tumors from 33 forms of cancer.
The researchers combined multiple platforms of genomic data from 1,400 SCC samples into integrated analyses, creating visual clusters of tumors based on genomic characteristics.
Squamous cell carcinomas had genomic features that set them apart from other cancers, the researchers found. The most common were gains or losses of the sections of certain chromosomes, making it likely that those regions harbor genes important to the development of SCCs.
The current study expands on research reported in 2014 and 2015, which compared genomic features of SCCs in head and neck cancer associated with smoking (a risk factor for head and neck cancer [HNC]) and HPV (a risk factor for cervical and some HNCs). Certain features were present in tumors associated with both, whereas others were exclusive to only 1 of the 2. The researchers also found similarities in the genomic characteristics of HNCs with lung cancers, some bladder cancers, and cervical cancer.
Squamous cell carcinomas (SCCs) associated with smoking and human papillomavirus (HPV) have distinct genomic signatures, say researchers from a National Institutes of Health-supported study. That is one of the findings that may help distinguish SCCs from other cancers and point the way to new research and treatment.
The researchers used new analytic tools and data from the recently completed PanCancer Atlas to investigate similarities and differences among SCCs in the head and neck, lung, esophagus, cervix, and bladder. The PanCancer Atlas is a detailed analysis from a dataset containing molecular and clinical information on more than 10,000 tumors from 33 forms of cancer.
The researchers combined multiple platforms of genomic data from 1,400 SCC samples into integrated analyses, creating visual clusters of tumors based on genomic characteristics.
Squamous cell carcinomas had genomic features that set them apart from other cancers, the researchers found. The most common were gains or losses of the sections of certain chromosomes, making it likely that those regions harbor genes important to the development of SCCs.
The current study expands on research reported in 2014 and 2015, which compared genomic features of SCCs in head and neck cancer associated with smoking (a risk factor for head and neck cancer [HNC]) and HPV (a risk factor for cervical and some HNCs). Certain features were present in tumors associated with both, whereas others were exclusive to only 1 of the 2. The researchers also found similarities in the genomic characteristics of HNCs with lung cancers, some bladder cancers, and cervical cancer.
Squamous cell carcinomas (SCCs) associated with smoking and human papillomavirus (HPV) have distinct genomic signatures, say researchers from a National Institutes of Health-supported study. That is one of the findings that may help distinguish SCCs from other cancers and point the way to new research and treatment.
The researchers used new analytic tools and data from the recently completed PanCancer Atlas to investigate similarities and differences among SCCs in the head and neck, lung, esophagus, cervix, and bladder. The PanCancer Atlas is a detailed analysis from a dataset containing molecular and clinical information on more than 10,000 tumors from 33 forms of cancer.
The researchers combined multiple platforms of genomic data from 1,400 SCC samples into integrated analyses, creating visual clusters of tumors based on genomic characteristics.
Squamous cell carcinomas had genomic features that set them apart from other cancers, the researchers found. The most common were gains or losses of the sections of certain chromosomes, making it likely that those regions harbor genes important to the development of SCCs.
The current study expands on research reported in 2014 and 2015, which compared genomic features of SCCs in head and neck cancer associated with smoking (a risk factor for head and neck cancer [HNC]) and HPV (a risk factor for cervical and some HNCs). Certain features were present in tumors associated with both, whereas others were exclusive to only 1 of the 2. The researchers also found similarities in the genomic characteristics of HNCs with lung cancers, some bladder cancers, and cervical cancer.
Single injection could treat hemophilia B long-term
Cell therapy could produce lasting effects in hemophilia B, according to a group of researchers.
They genetically modified induced pluripotent cells (iPSCs) derived from patients with hemophilia B and converted those iPSCs into hepatocyte-like cells (HLCs).
When transplanted into mouse models of hemophilia B, the HLCs remained functional for months, increasing levels of factor IX (FIX) and clotting efficiency.
However, the HLCs were substantially less effective than cryopreserved human hepatocytes (hHeps), which were able to correct clotting defects in mice long-term.
“The appeal of a cell-based approach is that you minimize the number of treatments that a patient needs,” explained Suvasini Ramaswamy, PhD, of The Boston Consulting Group in Massachusetts.
“Rather than constant injections, you can do this in one shot.”
Dr Ramaswamy and her colleagues described their results with this approach in Cell Reports. Study authors include employees of Shire Therapeutics, Vertex Pharmaceuticals, and Thermo Fisher Scientific.
The researchers first developed a quadruple knockout mouse model of hemophilia B that allows for the engraftment and expansion of hHeps. The team crossed transgenic FIX-/--deficient mice with Rag2-/- IL2rg -/- Fah-/- mice to create this model.
The researchers transplanted cryopreserved hHeps into the mice and observed a sustained increase in circulating levels of human albumin, human FIX, and clusters of FIX- or Fah-positive cells in the liver.
The team said the hHeps were able to restore clotting function to wild-type levels in the mice—at least 10-fold higher than levels needed for a significant improvement in hemophilia B.
The hHeps remained functional for up to a year, and the researchers believe the cells could persist even longer. The team noted that there was no difference in the efficacy of hHeps from different donors or vendors.
The researchers then tested the cell therapy they developed using iPSCs. They collected blood samples from 2 patients with severe hemophilia B, reprogrammed peripheral blood-derived mononuclear cells into iPSCs, and used CRISPR/Cas9 to repair the mutations in each patient’s FIX gene.
The team coaxed those repaired cells into HLCs and tested the HLCs in the mouse model of hemophilia B. The HLCs were transplanted through the spleen so the cells were distributed uniformly in the liver.
The researchers said the HLCs were present and functional in the liver for up to a year, but they were less effective than hHeps. Mice that received HLCs experienced “modest” increases in clotting efficiency, from less than 10% to about 25% of wild-type activity.
The researchers believe this work demonstrates the value of combining stem cell reprogramming and gene-modifying approaches to treat genetic diseases. However, more work is needed to optimize this approach for hemophilia B.
Cell therapy could produce lasting effects in hemophilia B, according to a group of researchers.
They genetically modified induced pluripotent cells (iPSCs) derived from patients with hemophilia B and converted those iPSCs into hepatocyte-like cells (HLCs).
When transplanted into mouse models of hemophilia B, the HLCs remained functional for months, increasing levels of factor IX (FIX) and clotting efficiency.
However, the HLCs were substantially less effective than cryopreserved human hepatocytes (hHeps), which were able to correct clotting defects in mice long-term.
“The appeal of a cell-based approach is that you minimize the number of treatments that a patient needs,” explained Suvasini Ramaswamy, PhD, of The Boston Consulting Group in Massachusetts.
“Rather than constant injections, you can do this in one shot.”
Dr Ramaswamy and her colleagues described their results with this approach in Cell Reports. Study authors include employees of Shire Therapeutics, Vertex Pharmaceuticals, and Thermo Fisher Scientific.
The researchers first developed a quadruple knockout mouse model of hemophilia B that allows for the engraftment and expansion of hHeps. The team crossed transgenic FIX-/--deficient mice with Rag2-/- IL2rg -/- Fah-/- mice to create this model.
The researchers transplanted cryopreserved hHeps into the mice and observed a sustained increase in circulating levels of human albumin, human FIX, and clusters of FIX- or Fah-positive cells in the liver.
The team said the hHeps were able to restore clotting function to wild-type levels in the mice—at least 10-fold higher than levels needed for a significant improvement in hemophilia B.
The hHeps remained functional for up to a year, and the researchers believe the cells could persist even longer. The team noted that there was no difference in the efficacy of hHeps from different donors or vendors.
The researchers then tested the cell therapy they developed using iPSCs. They collected blood samples from 2 patients with severe hemophilia B, reprogrammed peripheral blood-derived mononuclear cells into iPSCs, and used CRISPR/Cas9 to repair the mutations in each patient’s FIX gene.
The team coaxed those repaired cells into HLCs and tested the HLCs in the mouse model of hemophilia B. The HLCs were transplanted through the spleen so the cells were distributed uniformly in the liver.
The researchers said the HLCs were present and functional in the liver for up to a year, but they were less effective than hHeps. Mice that received HLCs experienced “modest” increases in clotting efficiency, from less than 10% to about 25% of wild-type activity.
The researchers believe this work demonstrates the value of combining stem cell reprogramming and gene-modifying approaches to treat genetic diseases. However, more work is needed to optimize this approach for hemophilia B.
Cell therapy could produce lasting effects in hemophilia B, according to a group of researchers.
They genetically modified induced pluripotent cells (iPSCs) derived from patients with hemophilia B and converted those iPSCs into hepatocyte-like cells (HLCs).
When transplanted into mouse models of hemophilia B, the HLCs remained functional for months, increasing levels of factor IX (FIX) and clotting efficiency.
However, the HLCs were substantially less effective than cryopreserved human hepatocytes (hHeps), which were able to correct clotting defects in mice long-term.
“The appeal of a cell-based approach is that you minimize the number of treatments that a patient needs,” explained Suvasini Ramaswamy, PhD, of The Boston Consulting Group in Massachusetts.
“Rather than constant injections, you can do this in one shot.”
Dr Ramaswamy and her colleagues described their results with this approach in Cell Reports. Study authors include employees of Shire Therapeutics, Vertex Pharmaceuticals, and Thermo Fisher Scientific.
The researchers first developed a quadruple knockout mouse model of hemophilia B that allows for the engraftment and expansion of hHeps. The team crossed transgenic FIX-/--deficient mice with Rag2-/- IL2rg -/- Fah-/- mice to create this model.
The researchers transplanted cryopreserved hHeps into the mice and observed a sustained increase in circulating levels of human albumin, human FIX, and clusters of FIX- or Fah-positive cells in the liver.
The team said the hHeps were able to restore clotting function to wild-type levels in the mice—at least 10-fold higher than levels needed for a significant improvement in hemophilia B.
The hHeps remained functional for up to a year, and the researchers believe the cells could persist even longer. The team noted that there was no difference in the efficacy of hHeps from different donors or vendors.
The researchers then tested the cell therapy they developed using iPSCs. They collected blood samples from 2 patients with severe hemophilia B, reprogrammed peripheral blood-derived mononuclear cells into iPSCs, and used CRISPR/Cas9 to repair the mutations in each patient’s FIX gene.
The team coaxed those repaired cells into HLCs and tested the HLCs in the mouse model of hemophilia B. The HLCs were transplanted through the spleen so the cells were distributed uniformly in the liver.
The researchers said the HLCs were present and functional in the liver for up to a year, but they were less effective than hHeps. Mice that received HLCs experienced “modest” increases in clotting efficiency, from less than 10% to about 25% of wild-type activity.
The researchers believe this work demonstrates the value of combining stem cell reprogramming and gene-modifying approaches to treat genetic diseases. However, more work is needed to optimize this approach for hemophilia B.
FDA issues CRL for proposed biosimilar rituximab
The US Food and Drug Administration (FDA) has issued a complete response letter (CRL) saying the agency cannot approve Sandoz’s proposed biosimilar rituximab.
Sandoz submitted the biologics licensing application for the product, known as GP2013, in September 2017.
The company was seeking approval for GP2013 to treat follicular lymphoma (FL), diffuse large B-cell lymphoma, chronic lymphocytic leukemia, rheumatoid arthritis, granulomatosis with polyangiitis, and microscopic polyangiitis.
The drug already has approval for these indications in Europe. The European Commission approved GP2013 (Rixathon) in June 2017.
As for US approval, Sandoz said it is evaluating the content of the FDA’s CRL and “remains committed to further discussions with FDA in order to bring this important medicine to US patients as soon as possible.”
The company said it “stands behind the robust body of evidence included in the regulatory submission” for GP2013.
Part of this evidence is the ASSIST-FL trial, in which researchers compared GP2013 to the reference product, Roche’s MabThera. Results from this trial were published in The Lancet Haematology and presented at ESMO 2017 Congress.
This phase 3 trial included adults with previously untreated, advanced stage FL. Patients received 8 cycles of cyclophosphamide, vincristine, and prednisone with either GP2013 or reference rituximab. Responders then received GP2013 or rituximab monotherapy as maintenance for up to 2 years.
At a median follow-up of 11.6 months, the overall response rate was 87% (271/311) in the GP2013 arm and 88% in the rituximab arm (274/313). Complete response rates were 15% (n=46) and 13% (n=42), respectively.
Rates of adverse events (AEs) were 93% in the GP2013 arm and 91% in the rituximab arm. Rates of serious AEs were 23% and 20%, respectively. The rate of discontinuation due to AEs was 7% in both arms.
The most common AE was neutropenia, which occurred in 26% of patients in the GP2013 arm and 30% of those in the rituximab arm in the combination phase. Rates of neutropenia in the maintenance phase were 10% and 6%, respectively.
The US Food and Drug Administration (FDA) has issued a complete response letter (CRL) saying the agency cannot approve Sandoz’s proposed biosimilar rituximab.
Sandoz submitted the biologics licensing application for the product, known as GP2013, in September 2017.
The company was seeking approval for GP2013 to treat follicular lymphoma (FL), diffuse large B-cell lymphoma, chronic lymphocytic leukemia, rheumatoid arthritis, granulomatosis with polyangiitis, and microscopic polyangiitis.
The drug already has approval for these indications in Europe. The European Commission approved GP2013 (Rixathon) in June 2017.
As for US approval, Sandoz said it is evaluating the content of the FDA’s CRL and “remains committed to further discussions with FDA in order to bring this important medicine to US patients as soon as possible.”
The company said it “stands behind the robust body of evidence included in the regulatory submission” for GP2013.
Part of this evidence is the ASSIST-FL trial, in which researchers compared GP2013 to the reference product, Roche’s MabThera. Results from this trial were published in The Lancet Haematology and presented at ESMO 2017 Congress.
This phase 3 trial included adults with previously untreated, advanced stage FL. Patients received 8 cycles of cyclophosphamide, vincristine, and prednisone with either GP2013 or reference rituximab. Responders then received GP2013 or rituximab monotherapy as maintenance for up to 2 years.
At a median follow-up of 11.6 months, the overall response rate was 87% (271/311) in the GP2013 arm and 88% in the rituximab arm (274/313). Complete response rates were 15% (n=46) and 13% (n=42), respectively.
Rates of adverse events (AEs) were 93% in the GP2013 arm and 91% in the rituximab arm. Rates of serious AEs were 23% and 20%, respectively. The rate of discontinuation due to AEs was 7% in both arms.
The most common AE was neutropenia, which occurred in 26% of patients in the GP2013 arm and 30% of those in the rituximab arm in the combination phase. Rates of neutropenia in the maintenance phase were 10% and 6%, respectively.
The US Food and Drug Administration (FDA) has issued a complete response letter (CRL) saying the agency cannot approve Sandoz’s proposed biosimilar rituximab.
Sandoz submitted the biologics licensing application for the product, known as GP2013, in September 2017.
The company was seeking approval for GP2013 to treat follicular lymphoma (FL), diffuse large B-cell lymphoma, chronic lymphocytic leukemia, rheumatoid arthritis, granulomatosis with polyangiitis, and microscopic polyangiitis.
The drug already has approval for these indications in Europe. The European Commission approved GP2013 (Rixathon) in June 2017.
As for US approval, Sandoz said it is evaluating the content of the FDA’s CRL and “remains committed to further discussions with FDA in order to bring this important medicine to US patients as soon as possible.”
The company said it “stands behind the robust body of evidence included in the regulatory submission” for GP2013.
Part of this evidence is the ASSIST-FL trial, in which researchers compared GP2013 to the reference product, Roche’s MabThera. Results from this trial were published in The Lancet Haematology and presented at ESMO 2017 Congress.
This phase 3 trial included adults with previously untreated, advanced stage FL. Patients received 8 cycles of cyclophosphamide, vincristine, and prednisone with either GP2013 or reference rituximab. Responders then received GP2013 or rituximab monotherapy as maintenance for up to 2 years.
At a median follow-up of 11.6 months, the overall response rate was 87% (271/311) in the GP2013 arm and 88% in the rituximab arm (274/313). Complete response rates were 15% (n=46) and 13% (n=42), respectively.
Rates of adverse events (AEs) were 93% in the GP2013 arm and 91% in the rituximab arm. Rates of serious AEs were 23% and 20%, respectively. The rate of discontinuation due to AEs was 7% in both arms.
The most common AE was neutropenia, which occurred in 26% of patients in the GP2013 arm and 30% of those in the rituximab arm in the combination phase. Rates of neutropenia in the maintenance phase were 10% and 6%, respectively.
JAK inhibitor reduces GVHD in mice
An investigational JAK1/2 inhibitor can fight graft-versus-host disease (GVHD), according to preclinical research published in Leukemia.
The inhibitor, baricitinib, reduced GVHD in mice while preserving T-cell expansion and the graft-versus-leukemia (GVL) effect.
Baricitinib proved more effective than ruxolitinib for the treatment and prevention of GVHD and enabled 100% survival in a mouse model of severe GVHD.
“We were surprised to achieve 100% survival of mice with the most severe model of graft-versus-host disease,” said study author Jaebok Choi, PhD, of the Washington University School of Medicine in St. Louis, Missouri.
“We are now studying the multi-pronged ways this drug behaves in an effort to develop an even better version for eventual use in clinical trials.”
For the current study, Dr Choi and his colleagues tested baricitinib and ruxolitinib in murine recipients of allogeneic hematopoietic stem cell transplants (allo-HSCTs). Mice received either drug for 31 days post-HSCT.
Mice treated with baricitinib had a significant reduction in intestinal GVHD compared to both ruxolitinib recipients and vehicle-treated controls (P=0.037).
In addition, 100% of baricitinib recipients were still alive at 60 days after HSCT, compared to about 60% of ruxolitinib recipients (P=0.0025) and almost none of the vehicle-treated controls.
The researchers found that baricitinib recipients had significantly better blood cell count recovery than control mice. Baricitinib recipients also had full donor chimerism and significantly higher percentages of donor bone marrow-derived B and T cells.
Furthermore, baricitinib recipients had higher levels of donor-derived regulatory T cells (Tregs) compared to vehicle- or ruxolitinib-treated mice.
However, the researchers noted that baricitinib recipients had a survival rate of about 70% even in the absence of donor Tregs. The team said this suggests baricitinib fights GVHD independently of the enhanced expansion of Tregs.
Further investigation revealed that baricitinib decreases helper T-cell 1 and 2 differentiation and reduces the expression of MHC II, CD80/86, and PD-L1 on allogeneic antigen-presenting cells.
The researchers also found that baricitinib could reverse ongoing GVHD. The team withheld baricitinib until mice developed GVHD, then tested the drug at doses of 200 μg and 400 μg per day.
Both doses reduced clinical GVHD scores and enabled 100% overall survival rates.
Finally, the researchers found that baricitinib “preserves and enhances” GVL effects. The team infused A20 cells and T-cell-depleted bone marrow cells into lethally irradiated mice, waited for the B-cell lymphoma to become established, and infused donor T cells.
The researchers found that baricitinib alone did not inhibit tumor growth, but it enhanced the GVL effects of donor T cells, significantly lowering the tumor burden.
“We don’t know yet exactly how this happens, but we’re working to understand it,” Dr Choi said. “We think at least part of the explanation is the drug strips the leukemia cells of their immune defenses, making them more vulnerable to attack by the donor T cells.”
“At the same time, the drug also stops the donor T cells from being able to make their way to important healthy tissues, such as the skin, liver, and gastrointestinal tract, where they often do the most damage.”
An investigational JAK1/2 inhibitor can fight graft-versus-host disease (GVHD), according to preclinical research published in Leukemia.
The inhibitor, baricitinib, reduced GVHD in mice while preserving T-cell expansion and the graft-versus-leukemia (GVL) effect.
Baricitinib proved more effective than ruxolitinib for the treatment and prevention of GVHD and enabled 100% survival in a mouse model of severe GVHD.
“We were surprised to achieve 100% survival of mice with the most severe model of graft-versus-host disease,” said study author Jaebok Choi, PhD, of the Washington University School of Medicine in St. Louis, Missouri.
“We are now studying the multi-pronged ways this drug behaves in an effort to develop an even better version for eventual use in clinical trials.”
For the current study, Dr Choi and his colleagues tested baricitinib and ruxolitinib in murine recipients of allogeneic hematopoietic stem cell transplants (allo-HSCTs). Mice received either drug for 31 days post-HSCT.
Mice treated with baricitinib had a significant reduction in intestinal GVHD compared to both ruxolitinib recipients and vehicle-treated controls (P=0.037).
In addition, 100% of baricitinib recipients were still alive at 60 days after HSCT, compared to about 60% of ruxolitinib recipients (P=0.0025) and almost none of the vehicle-treated controls.
The researchers found that baricitinib recipients had significantly better blood cell count recovery than control mice. Baricitinib recipients also had full donor chimerism and significantly higher percentages of donor bone marrow-derived B and T cells.
Furthermore, baricitinib recipients had higher levels of donor-derived regulatory T cells (Tregs) compared to vehicle- or ruxolitinib-treated mice.
However, the researchers noted that baricitinib recipients had a survival rate of about 70% even in the absence of donor Tregs. The team said this suggests baricitinib fights GVHD independently of the enhanced expansion of Tregs.
Further investigation revealed that baricitinib decreases helper T-cell 1 and 2 differentiation and reduces the expression of MHC II, CD80/86, and PD-L1 on allogeneic antigen-presenting cells.
The researchers also found that baricitinib could reverse ongoing GVHD. The team withheld baricitinib until mice developed GVHD, then tested the drug at doses of 200 μg and 400 μg per day.
Both doses reduced clinical GVHD scores and enabled 100% overall survival rates.
Finally, the researchers found that baricitinib “preserves and enhances” GVL effects. The team infused A20 cells and T-cell-depleted bone marrow cells into lethally irradiated mice, waited for the B-cell lymphoma to become established, and infused donor T cells.
The researchers found that baricitinib alone did not inhibit tumor growth, but it enhanced the GVL effects of donor T cells, significantly lowering the tumor burden.
“We don’t know yet exactly how this happens, but we’re working to understand it,” Dr Choi said. “We think at least part of the explanation is the drug strips the leukemia cells of their immune defenses, making them more vulnerable to attack by the donor T cells.”
“At the same time, the drug also stops the donor T cells from being able to make their way to important healthy tissues, such as the skin, liver, and gastrointestinal tract, where they often do the most damage.”
An investigational JAK1/2 inhibitor can fight graft-versus-host disease (GVHD), according to preclinical research published in Leukemia.
The inhibitor, baricitinib, reduced GVHD in mice while preserving T-cell expansion and the graft-versus-leukemia (GVL) effect.
Baricitinib proved more effective than ruxolitinib for the treatment and prevention of GVHD and enabled 100% survival in a mouse model of severe GVHD.
“We were surprised to achieve 100% survival of mice with the most severe model of graft-versus-host disease,” said study author Jaebok Choi, PhD, of the Washington University School of Medicine in St. Louis, Missouri.
“We are now studying the multi-pronged ways this drug behaves in an effort to develop an even better version for eventual use in clinical trials.”
For the current study, Dr Choi and his colleagues tested baricitinib and ruxolitinib in murine recipients of allogeneic hematopoietic stem cell transplants (allo-HSCTs). Mice received either drug for 31 days post-HSCT.
Mice treated with baricitinib had a significant reduction in intestinal GVHD compared to both ruxolitinib recipients and vehicle-treated controls (P=0.037).
In addition, 100% of baricitinib recipients were still alive at 60 days after HSCT, compared to about 60% of ruxolitinib recipients (P=0.0025) and almost none of the vehicle-treated controls.
The researchers found that baricitinib recipients had significantly better blood cell count recovery than control mice. Baricitinib recipients also had full donor chimerism and significantly higher percentages of donor bone marrow-derived B and T cells.
Furthermore, baricitinib recipients had higher levels of donor-derived regulatory T cells (Tregs) compared to vehicle- or ruxolitinib-treated mice.
However, the researchers noted that baricitinib recipients had a survival rate of about 70% even in the absence of donor Tregs. The team said this suggests baricitinib fights GVHD independently of the enhanced expansion of Tregs.
Further investigation revealed that baricitinib decreases helper T-cell 1 and 2 differentiation and reduces the expression of MHC II, CD80/86, and PD-L1 on allogeneic antigen-presenting cells.
The researchers also found that baricitinib could reverse ongoing GVHD. The team withheld baricitinib until mice developed GVHD, then tested the drug at doses of 200 μg and 400 μg per day.
Both doses reduced clinical GVHD scores and enabled 100% overall survival rates.
Finally, the researchers found that baricitinib “preserves and enhances” GVL effects. The team infused A20 cells and T-cell-depleted bone marrow cells into lethally irradiated mice, waited for the B-cell lymphoma to become established, and infused donor T cells.
The researchers found that baricitinib alone did not inhibit tumor growth, but it enhanced the GVL effects of donor T cells, significantly lowering the tumor burden.
“We don’t know yet exactly how this happens, but we’re working to understand it,” Dr Choi said. “We think at least part of the explanation is the drug strips the leukemia cells of their immune defenses, making them more vulnerable to attack by the donor T cells.”
“At the same time, the drug also stops the donor T cells from being able to make their way to important healthy tissues, such as the skin, liver, and gastrointestinal tract, where they often do the most damage.”
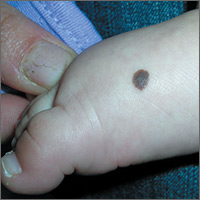
Brown spot on right foot

The FP recognized this as a benign congenital nevus.
While most congenital nevi are visible at birth, there are some that appear in the first year of life and are known as tardive congenital nevi. The FP used a dermatoscope to look at this nevus and found that its features were benign.
The parents wondered whether this needed to be removed to prevent it from becoming skin cancer in the future. The FP reassured them that the risk of melanoma from this one nevus was too small to warrant a prophylactic surgical excision. The parents agreed to the standard 6-month immunizations.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Smith, M. Congenital nevi. In: Usatine R, Smith M, Mayeaux EJ, et al. Color Atlas of Family Medicine. 2nd ed. New York, NY: McGraw-Hill; 2013:953-957.
To learn more about the Color Atlas of Family Medicine, see: www.amazon.com/Color-Family-Medicine-Richard-Usatine/dp/0071769641/.
You can now get the second edition of the Color Atlas of Family Medicine as an app by clicking on this link: usatinemedia.com.

The FP recognized this as a benign congenital nevus.
While most congenital nevi are visible at birth, there are some that appear in the first year of life and are known as tardive congenital nevi. The FP used a dermatoscope to look at this nevus and found that its features were benign.
The parents wondered whether this needed to be removed to prevent it from becoming skin cancer in the future. The FP reassured them that the risk of melanoma from this one nevus was too small to warrant a prophylactic surgical excision. The parents agreed to the standard 6-month immunizations.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Smith, M. Congenital nevi. In: Usatine R, Smith M, Mayeaux EJ, et al. Color Atlas of Family Medicine. 2nd ed. New York, NY: McGraw-Hill; 2013:953-957.
To learn more about the Color Atlas of Family Medicine, see: www.amazon.com/Color-Family-Medicine-Richard-Usatine/dp/0071769641/.
You can now get the second edition of the Color Atlas of Family Medicine as an app by clicking on this link: usatinemedia.com.

The FP recognized this as a benign congenital nevus.
While most congenital nevi are visible at birth, there are some that appear in the first year of life and are known as tardive congenital nevi. The FP used a dermatoscope to look at this nevus and found that its features were benign.
The parents wondered whether this needed to be removed to prevent it from becoming skin cancer in the future. The FP reassured them that the risk of melanoma from this one nevus was too small to warrant a prophylactic surgical excision. The parents agreed to the standard 6-month immunizations.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Smith, M. Congenital nevi. In: Usatine R, Smith M, Mayeaux EJ, et al. Color Atlas of Family Medicine. 2nd ed. New York, NY: McGraw-Hill; 2013:953-957.
To learn more about the Color Atlas of Family Medicine, see: www.amazon.com/Color-Family-Medicine-Richard-Usatine/dp/0071769641/.
You can now get the second edition of the Color Atlas of Family Medicine as an app by clicking on this link: usatinemedia.com.
No benefits from bath emollients for childhood eczema
In a trial of 482 children with atopic dermatitis who were randomized to either use of prescribed emollient bath additives regularly for 12 months or no additives, in addition to usual care, there was no significant difference between the two groups in the patient-oriented eczema measure (POEM) – a score of symptoms in the previous week – for the first 16 weeks of treatment, reported Miriam Santer, MD, of the University of Southampton, England, and her coauthors. The mean POEM score was 7.5 in the bath additives group and 8.4 in the no bath additives group.
There also was no significant effect of emollient bath additives on the secondary outcomes, which included POEM over 52 weeks, dermatitis family impact, generic quality of life, number of eczema exacerbations, or topical corticosteroid or topical calcineurin inhibitor use.
The authors noted that there was the suggestion of benefit in children who bathed five or more times a week, and in children aged under 5 years, but in both cases, the numbers were small.
“Although there is evidence for the need for leave-on emollients and widespread clinical consensus around soap substitutes, there is less agreement about the potential additional benefits of bath additives and a dearth of evidence on their effectiveness,” wrote Dr. Santer and her associates. “Bath additives are, however, widely prescribed at a cost of more than 23 million pounds ($33 million US; 26 million euros) annually to the National Health Service in England.”
The bath emollients used in the study included Aveeno bath oil, and Oilatum bath additive and Balneum bath oil – products available largely in the United Kingdom – along with others. Adherence was reasonable, with 93% of participants in the emollient group using bath additives more than half or all the time, and 92% of those in the no bath additives group using them less than half the time or never.
The authors stressed that there was strong evidence that supported regular use of leave-on emollients, and clinical consensus around the use of emollients as soap substitutes. “These findings are timely for clinicians and prescribing advisers, as prescribing guidelines vary widely in their advice on the use of bath additives, and pressure on budgets has led to formularies becoming increasingly restrictive.”
The study was supported by the National Institute for Health Research. No conflicts of interest were declared.
SOURCE: Santer M et al. BMJ 2018 May 2. doi: 10.1136/bmj.k1332
The National Institute for Health and Care Excellence in the United Kingdom currently recommends regular use of “emollient wash products” for children with eczema, despite the fact that little good evidence of their benefit exists. It is therefore heartening that this study has largely addressed that evidence gap: There was no statistically significant difference between those children prescribed one of three bath additives in addition to standard care and those who received standard care alone.
However, there is still the question of whether younger children – particularly those who are bathed daily – might still benefit from bath emollients. Likewise, children who experience recurrent skin infections may derive some additional benefit from emollient bath additives with antiseptic properties.
Carsten Flohr, MD, is with the St. John’s Institute of Dermatology at Kings College London, and Amina Ahmed is a patient panel member at the Centre of Evidence-Based Dermatology in Nottingham, England. These comments are taken from an editorial accompanying the article by Santer et al. (BMJ. 2018 May 2. doi: 10.1136/bmj.k1791.) No conflicts of interest were declared.
The National Institute for Health and Care Excellence in the United Kingdom currently recommends regular use of “emollient wash products” for children with eczema, despite the fact that little good evidence of their benefit exists. It is therefore heartening that this study has largely addressed that evidence gap: There was no statistically significant difference between those children prescribed one of three bath additives in addition to standard care and those who received standard care alone.
However, there is still the question of whether younger children – particularly those who are bathed daily – might still benefit from bath emollients. Likewise, children who experience recurrent skin infections may derive some additional benefit from emollient bath additives with antiseptic properties.
Carsten Flohr, MD, is with the St. John’s Institute of Dermatology at Kings College London, and Amina Ahmed is a patient panel member at the Centre of Evidence-Based Dermatology in Nottingham, England. These comments are taken from an editorial accompanying the article by Santer et al. (BMJ. 2018 May 2. doi: 10.1136/bmj.k1791.) No conflicts of interest were declared.
The National Institute for Health and Care Excellence in the United Kingdom currently recommends regular use of “emollient wash products” for children with eczema, despite the fact that little good evidence of their benefit exists. It is therefore heartening that this study has largely addressed that evidence gap: There was no statistically significant difference between those children prescribed one of three bath additives in addition to standard care and those who received standard care alone.
However, there is still the question of whether younger children – particularly those who are bathed daily – might still benefit from bath emollients. Likewise, children who experience recurrent skin infections may derive some additional benefit from emollient bath additives with antiseptic properties.
Carsten Flohr, MD, is with the St. John’s Institute of Dermatology at Kings College London, and Amina Ahmed is a patient panel member at the Centre of Evidence-Based Dermatology in Nottingham, England. These comments are taken from an editorial accompanying the article by Santer et al. (BMJ. 2018 May 2. doi: 10.1136/bmj.k1791.) No conflicts of interest were declared.
In a trial of 482 children with atopic dermatitis who were randomized to either use of prescribed emollient bath additives regularly for 12 months or no additives, in addition to usual care, there was no significant difference between the two groups in the patient-oriented eczema measure (POEM) – a score of symptoms in the previous week – for the first 16 weeks of treatment, reported Miriam Santer, MD, of the University of Southampton, England, and her coauthors. The mean POEM score was 7.5 in the bath additives group and 8.4 in the no bath additives group.
There also was no significant effect of emollient bath additives on the secondary outcomes, which included POEM over 52 weeks, dermatitis family impact, generic quality of life, number of eczema exacerbations, or topical corticosteroid or topical calcineurin inhibitor use.
The authors noted that there was the suggestion of benefit in children who bathed five or more times a week, and in children aged under 5 years, but in both cases, the numbers were small.
“Although there is evidence for the need for leave-on emollients and widespread clinical consensus around soap substitutes, there is less agreement about the potential additional benefits of bath additives and a dearth of evidence on their effectiveness,” wrote Dr. Santer and her associates. “Bath additives are, however, widely prescribed at a cost of more than 23 million pounds ($33 million US; 26 million euros) annually to the National Health Service in England.”
The bath emollients used in the study included Aveeno bath oil, and Oilatum bath additive and Balneum bath oil – products available largely in the United Kingdom – along with others. Adherence was reasonable, with 93% of participants in the emollient group using bath additives more than half or all the time, and 92% of those in the no bath additives group using them less than half the time or never.
The authors stressed that there was strong evidence that supported regular use of leave-on emollients, and clinical consensus around the use of emollients as soap substitutes. “These findings are timely for clinicians and prescribing advisers, as prescribing guidelines vary widely in their advice on the use of bath additives, and pressure on budgets has led to formularies becoming increasingly restrictive.”
The study was supported by the National Institute for Health Research. No conflicts of interest were declared.
SOURCE: Santer M et al. BMJ 2018 May 2. doi: 10.1136/bmj.k1332
In a trial of 482 children with atopic dermatitis who were randomized to either use of prescribed emollient bath additives regularly for 12 months or no additives, in addition to usual care, there was no significant difference between the two groups in the patient-oriented eczema measure (POEM) – a score of symptoms in the previous week – for the first 16 weeks of treatment, reported Miriam Santer, MD, of the University of Southampton, England, and her coauthors. The mean POEM score was 7.5 in the bath additives group and 8.4 in the no bath additives group.
There also was no significant effect of emollient bath additives on the secondary outcomes, which included POEM over 52 weeks, dermatitis family impact, generic quality of life, number of eczema exacerbations, or topical corticosteroid or topical calcineurin inhibitor use.
The authors noted that there was the suggestion of benefit in children who bathed five or more times a week, and in children aged under 5 years, but in both cases, the numbers were small.
“Although there is evidence for the need for leave-on emollients and widespread clinical consensus around soap substitutes, there is less agreement about the potential additional benefits of bath additives and a dearth of evidence on their effectiveness,” wrote Dr. Santer and her associates. “Bath additives are, however, widely prescribed at a cost of more than 23 million pounds ($33 million US; 26 million euros) annually to the National Health Service in England.”
The bath emollients used in the study included Aveeno bath oil, and Oilatum bath additive and Balneum bath oil – products available largely in the United Kingdom – along with others. Adherence was reasonable, with 93% of participants in the emollient group using bath additives more than half or all the time, and 92% of those in the no bath additives group using them less than half the time or never.
The authors stressed that there was strong evidence that supported regular use of leave-on emollients, and clinical consensus around the use of emollients as soap substitutes. “These findings are timely for clinicians and prescribing advisers, as prescribing guidelines vary widely in their advice on the use of bath additives, and pressure on budgets has led to formularies becoming increasingly restrictive.”
The study was supported by the National Institute for Health Research. No conflicts of interest were declared.
SOURCE: Santer M et al. BMJ 2018 May 2. doi: 10.1136/bmj.k1332
FROM THE BMJ
Key clinical point: Emollient bath additives do not improve eczema scores in children.
Major finding: There was no significant difference in eczema scores between bath emollients or no bath emollients: the mean POEM score was 7.5 in the bath additives group and 8.4 in the no bath additives group.
Study details: An open-label randomized trial in 482 children with atopic dermatitis.
Disclosures: The study was supported by the National Institute for Health Research. No conflicts of interest were declared.
Source: Santer M et al. BMJ 2018 May 2. doi: 10.1136/bmj.k1332