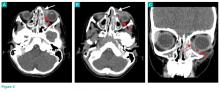
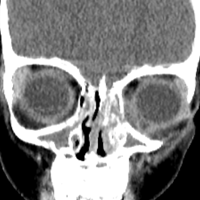
User login
Working up patients with allergic contact dermatitis
When working up patients with allergic contact dermatitis (ACD), the patch test used may depend on how frequently testing is performed in the practice, and the type of allergies that are being evaluated, according to Joseph Fowler Jr., MD, of the department of dermatology at the University of Louisville (Ky).
T.R.U.E. TEST is more convenient than standard patch testing is but misses allergic contact dermatitis up to 40% of the time, he pointed out. The benefit of the T.R.U.E. TEST is that it’s easy to use, allergens come preapplied to a gel-based tape so there’s very little prep time, and they are well standardized with the same quantity on each patch every time.
T.R.U.E. TEST seems to work well for when testing for metal allergies, as well as allergies to topical antibiotics, steroids, and rubber, but not as well for dental implants, fragrances, newer preservatives, surfactants, acrylates, and some industrial and cosmetic allergens. It’s not so effective in many occupational settings, but even so, T.R.U.E. TEST is a good option when testing is performed infrequently, and “is much better than no patch testing at all,” according to Dr. Fowler, who spoke at the Annual Coastal Dermatology Symposium, jointly presented by the University of Louisville and Global Academy for Medical Education.
In a presentation on contact dermatitis and itch, he pointed out that what appears to be atopic dermatitis (AD) in a patient might actually be ACD and that ACD is common in patients with AD and complicates its treatment. Metals, fragrances, and topical components – namely lanolin and neomycin – are the most likely allergens to cause trouble in AD. Nickel allergy can be particularly problematic, causing severe lesions beyond the point of contact (Dermatitis. 2012 Nov-Dec;23[6]:275-80).
“Strongly consider patch testing any chronic, difficult to control atopic patient,” especially when AD is not affecting the typical areas – or spreads beyond them – and when it doesn’t respond to the usual treatments. The onset of AD beyond age 5 years is another clue that contact dermatitis might be at work. Patch testing atopic patients is “more likely to be helpful in disease management than scratch or RAST [radioallergosorbent] testing,” Dr. Fowler said.
It’s best if patch testing is done while patients are off immunosuppressants, but current immunosuppressive therapy should not be an absolute contraindication to testing, he said. Not all of them throw off the results. “You do not need to worry about patch testing a patient who is on antihistamines, tumor necrosis factor–alpha inhibitors, NSAIDs, or methotrexate.” However, when it comes to patch testing a patient on cyclosporine, tacrolimus, azathioprine, and mycophenolate mofetil, he said, “probably not” (Dermatitis. 2012 Nov-Dec;23[6]:301-3).
Pruritus might or might not be related to the skin issues. For itch caused by skin diseases such as scabies, dermatitis, or psoriasis, “treat the dermatosis to treat the itch,” Dr. Fowler said.
Several topicals can help while the skin problems are being tamed, including hypochlorous acid to stabilize mast cells; strontium 4% hydrogel; and compounded topical ketamine, amitriptyline, and lidocaine, which seems to be particularly helpful (J Am Acad Dermatol. 2017 Apr;76[4]:760-1). Other than for urticaria, antihistamines are of little use, except to provide sedation.
Renal disease, liver disease, lymphoma, and neurologic abnormalities are among the systemic problems that can cause itch; the giveaway is that there’s no primary skin disease, Dr. Fowler said. While systemic problems are being addressed, gabapentin, tricyclic antidepressants, and anxiolytics can help. For generalized pruritus, with no primary skin disease, a referral to a neurologist is essential, he said.
This publication and the Global Academy for Medical Education are owned by Frontline Medical News. Dr. Fowler is a consultant, speaker, and/or researcher for a number of companies, including AbbVie, Regeneron/Sanofi, Allergan, Galderma, and Merck.
When working up patients with allergic contact dermatitis (ACD), the patch test used may depend on how frequently testing is performed in the practice, and the type of allergies that are being evaluated, according to Joseph Fowler Jr., MD, of the department of dermatology at the University of Louisville (Ky).
T.R.U.E. TEST is more convenient than standard patch testing is but misses allergic contact dermatitis up to 40% of the time, he pointed out. The benefit of the T.R.U.E. TEST is that it’s easy to use, allergens come preapplied to a gel-based tape so there’s very little prep time, and they are well standardized with the same quantity on each patch every time.
T.R.U.E. TEST seems to work well for when testing for metal allergies, as well as allergies to topical antibiotics, steroids, and rubber, but not as well for dental implants, fragrances, newer preservatives, surfactants, acrylates, and some industrial and cosmetic allergens. It’s not so effective in many occupational settings, but even so, T.R.U.E. TEST is a good option when testing is performed infrequently, and “is much better than no patch testing at all,” according to Dr. Fowler, who spoke at the Annual Coastal Dermatology Symposium, jointly presented by the University of Louisville and Global Academy for Medical Education.
In a presentation on contact dermatitis and itch, he pointed out that what appears to be atopic dermatitis (AD) in a patient might actually be ACD and that ACD is common in patients with AD and complicates its treatment. Metals, fragrances, and topical components – namely lanolin and neomycin – are the most likely allergens to cause trouble in AD. Nickel allergy can be particularly problematic, causing severe lesions beyond the point of contact (Dermatitis. 2012 Nov-Dec;23[6]:275-80).
“Strongly consider patch testing any chronic, difficult to control atopic patient,” especially when AD is not affecting the typical areas – or spreads beyond them – and when it doesn’t respond to the usual treatments. The onset of AD beyond age 5 years is another clue that contact dermatitis might be at work. Patch testing atopic patients is “more likely to be helpful in disease management than scratch or RAST [radioallergosorbent] testing,” Dr. Fowler said.
It’s best if patch testing is done while patients are off immunosuppressants, but current immunosuppressive therapy should not be an absolute contraindication to testing, he said. Not all of them throw off the results. “You do not need to worry about patch testing a patient who is on antihistamines, tumor necrosis factor–alpha inhibitors, NSAIDs, or methotrexate.” However, when it comes to patch testing a patient on cyclosporine, tacrolimus, azathioprine, and mycophenolate mofetil, he said, “probably not” (Dermatitis. 2012 Nov-Dec;23[6]:301-3).
Pruritus might or might not be related to the skin issues. For itch caused by skin diseases such as scabies, dermatitis, or psoriasis, “treat the dermatosis to treat the itch,” Dr. Fowler said.
Several topicals can help while the skin problems are being tamed, including hypochlorous acid to stabilize mast cells; strontium 4% hydrogel; and compounded topical ketamine, amitriptyline, and lidocaine, which seems to be particularly helpful (J Am Acad Dermatol. 2017 Apr;76[4]:760-1). Other than for urticaria, antihistamines are of little use, except to provide sedation.
Renal disease, liver disease, lymphoma, and neurologic abnormalities are among the systemic problems that can cause itch; the giveaway is that there’s no primary skin disease, Dr. Fowler said. While systemic problems are being addressed, gabapentin, tricyclic antidepressants, and anxiolytics can help. For generalized pruritus, with no primary skin disease, a referral to a neurologist is essential, he said.
This publication and the Global Academy for Medical Education are owned by Frontline Medical News. Dr. Fowler is a consultant, speaker, and/or researcher for a number of companies, including AbbVie, Regeneron/Sanofi, Allergan, Galderma, and Merck.
When working up patients with allergic contact dermatitis (ACD), the patch test used may depend on how frequently testing is performed in the practice, and the type of allergies that are being evaluated, according to Joseph Fowler Jr., MD, of the department of dermatology at the University of Louisville (Ky).
T.R.U.E. TEST is more convenient than standard patch testing is but misses allergic contact dermatitis up to 40% of the time, he pointed out. The benefit of the T.R.U.E. TEST is that it’s easy to use, allergens come preapplied to a gel-based tape so there’s very little prep time, and they are well standardized with the same quantity on each patch every time.
T.R.U.E. TEST seems to work well for when testing for metal allergies, as well as allergies to topical antibiotics, steroids, and rubber, but not as well for dental implants, fragrances, newer preservatives, surfactants, acrylates, and some industrial and cosmetic allergens. It’s not so effective in many occupational settings, but even so, T.R.U.E. TEST is a good option when testing is performed infrequently, and “is much better than no patch testing at all,” according to Dr. Fowler, who spoke at the Annual Coastal Dermatology Symposium, jointly presented by the University of Louisville and Global Academy for Medical Education.
In a presentation on contact dermatitis and itch, he pointed out that what appears to be atopic dermatitis (AD) in a patient might actually be ACD and that ACD is common in patients with AD and complicates its treatment. Metals, fragrances, and topical components – namely lanolin and neomycin – are the most likely allergens to cause trouble in AD. Nickel allergy can be particularly problematic, causing severe lesions beyond the point of contact (Dermatitis. 2012 Nov-Dec;23[6]:275-80).
“Strongly consider patch testing any chronic, difficult to control atopic patient,” especially when AD is not affecting the typical areas – or spreads beyond them – and when it doesn’t respond to the usual treatments. The onset of AD beyond age 5 years is another clue that contact dermatitis might be at work. Patch testing atopic patients is “more likely to be helpful in disease management than scratch or RAST [radioallergosorbent] testing,” Dr. Fowler said.
It’s best if patch testing is done while patients are off immunosuppressants, but current immunosuppressive therapy should not be an absolute contraindication to testing, he said. Not all of them throw off the results. “You do not need to worry about patch testing a patient who is on antihistamines, tumor necrosis factor–alpha inhibitors, NSAIDs, or methotrexate.” However, when it comes to patch testing a patient on cyclosporine, tacrolimus, azathioprine, and mycophenolate mofetil, he said, “probably not” (Dermatitis. 2012 Nov-Dec;23[6]:301-3).
Pruritus might or might not be related to the skin issues. For itch caused by skin diseases such as scabies, dermatitis, or psoriasis, “treat the dermatosis to treat the itch,” Dr. Fowler said.
Several topicals can help while the skin problems are being tamed, including hypochlorous acid to stabilize mast cells; strontium 4% hydrogel; and compounded topical ketamine, amitriptyline, and lidocaine, which seems to be particularly helpful (J Am Acad Dermatol. 2017 Apr;76[4]:760-1). Other than for urticaria, antihistamines are of little use, except to provide sedation.
Renal disease, liver disease, lymphoma, and neurologic abnormalities are among the systemic problems that can cause itch; the giveaway is that there’s no primary skin disease, Dr. Fowler said. While systemic problems are being addressed, gabapentin, tricyclic antidepressants, and anxiolytics can help. For generalized pruritus, with no primary skin disease, a referral to a neurologist is essential, he said.
This publication and the Global Academy for Medical Education are owned by Frontline Medical News. Dr. Fowler is a consultant, speaker, and/or researcher for a number of companies, including AbbVie, Regeneron/Sanofi, Allergan, Galderma, and Merck.
FROM THE COASTAL DERMATOLOGY SYMPOSIUM
Whose nurse is she/he?
I suspect that there is at least one person in your office or on your team whose name is followed by the initials “RN.” How do you refer to that individual? Do you introduce her as “My nurse Louise”? Or do you say “I would like you to meet Lance, who is one of our nurses”? How often do you say “Rachel will be your nurse today”?
Is there really much difference between “my,” “our,” and “your” in this context? I suspect that most of us unconsciously avoid “my.” But, back in the era when solo practitioner owner/operators walked the earth, “my nurse” was a more frequent descriptor. The system was male dominated and hierarchical. And, of course, the doctor was paying the nurse’s salary.
However, a recent Ethics Rounds in the September 2017 Pediatrics titled “Physician-Nurse Interactions in Critical Care” has gotten me thinking more about what may seem to be semantic hairsplitting between “our nurse” and “your nurse” (doi: 10.1542/peds.2017-0352). The scenario revolves around a young neonatal ICU nurse in her first clinical position who is criticized by her supervisor for advocating for a young mother by questioning the doctor. A good part of the discussion focuses on the ethical dilemma faced by someone whose training has emphasized her obligation to advocate for her patients suddenly finding herself in a situation in which she sees the doctor’s care plan as flawed or at best inadequate. In this particular case, a more experienced nurse would probably already have acquired strategies and a vocabulary that could minimize or avert the conflict. However,
I hope that you have fostered a professional atmosphere that leaves room in which – as well as a process by which – a nurse can question your management of a patient without fear of retribution. Although it is never easy to have your actions questioned, it is certainly easier when the process takes place in a retrospective review rather than when the issue presents itself in the glare of real time and the nurse feels he must speak up now to advocate for the patient adequately.
When the call comes in from a panicked parent at 4 p.m., pleading to have her sick child seen, how does the nurse balance his commitment to the health of the patients against his concern for the doctor’s well being. Occasionally, I hear a nurse erring on the side of being zealous guardians of the doctor’s free time. However, I sense that, day in and day out, it is the nurse’s obligation to the patient that prevails most of the time. I hope I am correct.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.”
Email him at [email protected].
I suspect that there is at least one person in your office or on your team whose name is followed by the initials “RN.” How do you refer to that individual? Do you introduce her as “My nurse Louise”? Or do you say “I would like you to meet Lance, who is one of our nurses”? How often do you say “Rachel will be your nurse today”?
Is there really much difference between “my,” “our,” and “your” in this context? I suspect that most of us unconsciously avoid “my.” But, back in the era when solo practitioner owner/operators walked the earth, “my nurse” was a more frequent descriptor. The system was male dominated and hierarchical. And, of course, the doctor was paying the nurse’s salary.
However, a recent Ethics Rounds in the September 2017 Pediatrics titled “Physician-Nurse Interactions in Critical Care” has gotten me thinking more about what may seem to be semantic hairsplitting between “our nurse” and “your nurse” (doi: 10.1542/peds.2017-0352). The scenario revolves around a young neonatal ICU nurse in her first clinical position who is criticized by her supervisor for advocating for a young mother by questioning the doctor. A good part of the discussion focuses on the ethical dilemma faced by someone whose training has emphasized her obligation to advocate for her patients suddenly finding herself in a situation in which she sees the doctor’s care plan as flawed or at best inadequate. In this particular case, a more experienced nurse would probably already have acquired strategies and a vocabulary that could minimize or avert the conflict. However,
I hope that you have fostered a professional atmosphere that leaves room in which – as well as a process by which – a nurse can question your management of a patient without fear of retribution. Although it is never easy to have your actions questioned, it is certainly easier when the process takes place in a retrospective review rather than when the issue presents itself in the glare of real time and the nurse feels he must speak up now to advocate for the patient adequately.
When the call comes in from a panicked parent at 4 p.m., pleading to have her sick child seen, how does the nurse balance his commitment to the health of the patients against his concern for the doctor’s well being. Occasionally, I hear a nurse erring on the side of being zealous guardians of the doctor’s free time. However, I sense that, day in and day out, it is the nurse’s obligation to the patient that prevails most of the time. I hope I am correct.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.”
Email him at [email protected].
I suspect that there is at least one person in your office or on your team whose name is followed by the initials “RN.” How do you refer to that individual? Do you introduce her as “My nurse Louise”? Or do you say “I would like you to meet Lance, who is one of our nurses”? How often do you say “Rachel will be your nurse today”?
Is there really much difference between “my,” “our,” and “your” in this context? I suspect that most of us unconsciously avoid “my.” But, back in the era when solo practitioner owner/operators walked the earth, “my nurse” was a more frequent descriptor. The system was male dominated and hierarchical. And, of course, the doctor was paying the nurse’s salary.
However, a recent Ethics Rounds in the September 2017 Pediatrics titled “Physician-Nurse Interactions in Critical Care” has gotten me thinking more about what may seem to be semantic hairsplitting between “our nurse” and “your nurse” (doi: 10.1542/peds.2017-0352). The scenario revolves around a young neonatal ICU nurse in her first clinical position who is criticized by her supervisor for advocating for a young mother by questioning the doctor. A good part of the discussion focuses on the ethical dilemma faced by someone whose training has emphasized her obligation to advocate for her patients suddenly finding herself in a situation in which she sees the doctor’s care plan as flawed or at best inadequate. In this particular case, a more experienced nurse would probably already have acquired strategies and a vocabulary that could minimize or avert the conflict. However,
I hope that you have fostered a professional atmosphere that leaves room in which – as well as a process by which – a nurse can question your management of a patient without fear of retribution. Although it is never easy to have your actions questioned, it is certainly easier when the process takes place in a retrospective review rather than when the issue presents itself in the glare of real time and the nurse feels he must speak up now to advocate for the patient adequately.
When the call comes in from a panicked parent at 4 p.m., pleading to have her sick child seen, how does the nurse balance his commitment to the health of the patients against his concern for the doctor’s well being. Occasionally, I hear a nurse erring on the side of being zealous guardians of the doctor’s free time. However, I sense that, day in and day out, it is the nurse’s obligation to the patient that prevails most of the time. I hope I am correct.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.”
Email him at [email protected].
In the reemergence of typhus, the challenge is early diagnosis
SAN DIEGO – Typhus in many forms, particularly scrub typhus, has reemerged worldwide, but none of these rickettsial infections poses a significant public health threat if promptly diagnosed, according to an update presented at an annual scientific meeting on infectious diseases.
Scrub typhus, which is spread by several species of trombiculid mites or chiggers, poses a large threat in regard to typhus epidemics, particularly in Asia, but sporadic cases of different types of typhus are being seen everywhere, including in the United States, according to George M. Varghese, MD, department of infectious diseases, Christian Medical College, Vellore, India.
“The typhus diseases are clinically similar but epidemiologically and etiologically distinct,” reported Dr. Varghese, “but doxycycline is the drug of choice for almost all of the rickettsial infections.”

The bacteria responsible for scrub typhus is Orientia tsutsugamushi, which is no longer included in the genus Rickettsia, but Dr. Varghese, who has published frequently on the epidemiology of scrub typhus, said that it is still appropriately grouped among rickettsial infections. It shares many features with the other rickettsioses, which were considered to be fading but are now resurging after the large epidemics that occurred prior to the introduction of antibiotics.
In the World Wars, Rickettsia prowazekii – which is carried and spread by body lice – was the most well known typhus threat. According to Dr. Varghese, this bacterium may have killed more soldiers in these conflicts than did firearms. Although R. prowazekii has not disappeared as a source of typhus outbreaks, particularly in South America and Africa, there are current epidemics produced from rickettsial infections carried by fleas, such as R. typhi, or ticks, like R. rickettsii, or mice, like R. felis.
For clinical detection of these forms of typhus, there are differences. Although all are associated with a rapid onset of fever, headache, and myalgia, subtle signs can be helpful in making a diagnosis while waiting for laboratory confirmation. For example, scrub typhus, unlike Rocky Mountain Fever, which is caused by tick bites, does not generally include a rash. Rather, eschars, which are small patches of necrotic skin, are far more characteristic.
“Serological tests are the most common diagnostic tool for typhus, but serology may not allow early diagnosis. You can obtain a false positive in the early stages of disease,” Dr. Varghese warned. To speed the diagnosis, he said that looking for the clinical clues characteristic of the suspected form of typhus, such as the scrub typhus-associated eschar, “is valuable.” However, he also emphasized that even with positive serology results, “good epidemiology and history is helpful for laboratory interpretation.”
A variety of serological tests can identify typhus pathogens, but ELISA is now the most widely used, according to Dr. Varghese, noting that this test offers a sensitivity of 93% and a specificity of 91%. Both are higher than those provided by alternatives. As a result of improved sensitivity of diagnostic tests, prevalence rates of some forms of typhus have proved to be unexpectedly high. For example, in a study undertaken in his region of India, the seroprevalence of scrub typhus was 31.8% (Trop Med Int Health. 2017;22:576-82. doi: 10.1111/tmi.12853).
Of unmet needs in the clinical management of typhus, Dr. Varghese listed better strategies for point-of-care diagnosis and treatment and better data on how to manage patients who are severely ill. Advanced disease, which is common to rural areas with limited access to health care, is the source of almost all typhus mortality, according to Dr. Varghese. He described a trial now being initiated in severe disease that will compare intravenous doxycycline to IV azithromycin and to a combination of both IV doxycycline and azithromycin.
Although Dr. Varghese cautioned that reports of resistant typhus infections, particularly in Thailand, might prove to be the next big clinical challenge in typhus, he said that progress is being made toward reducing the burden of this disease in his area of the world. In a disease associated with a mortality of 50% if left untreated, he attributes gains to earlier diagnosis and prompt treatment.
At his medical center, “we have been working with this disease for a decade and a half,” he said, referring to scrub typhus. “When we started off, the mortality was around 15% after diagnosis. Today, the mortality is about 5%-7%.”
Dr. Varghese reported that he has no financial relationships relevant to this topic. The event was the combined annual meetings of the Infectious Diseases Society of America, the Society for Healthcare Epidemiology of America, the HIV Medicine Association, and the Pediatric Infectious Diseases Society.
SAN DIEGO – Typhus in many forms, particularly scrub typhus, has reemerged worldwide, but none of these rickettsial infections poses a significant public health threat if promptly diagnosed, according to an update presented at an annual scientific meeting on infectious diseases.
Scrub typhus, which is spread by several species of trombiculid mites or chiggers, poses a large threat in regard to typhus epidemics, particularly in Asia, but sporadic cases of different types of typhus are being seen everywhere, including in the United States, according to George M. Varghese, MD, department of infectious diseases, Christian Medical College, Vellore, India.
“The typhus diseases are clinically similar but epidemiologically and etiologically distinct,” reported Dr. Varghese, “but doxycycline is the drug of choice for almost all of the rickettsial infections.”

The bacteria responsible for scrub typhus is Orientia tsutsugamushi, which is no longer included in the genus Rickettsia, but Dr. Varghese, who has published frequently on the epidemiology of scrub typhus, said that it is still appropriately grouped among rickettsial infections. It shares many features with the other rickettsioses, which were considered to be fading but are now resurging after the large epidemics that occurred prior to the introduction of antibiotics.
In the World Wars, Rickettsia prowazekii – which is carried and spread by body lice – was the most well known typhus threat. According to Dr. Varghese, this bacterium may have killed more soldiers in these conflicts than did firearms. Although R. prowazekii has not disappeared as a source of typhus outbreaks, particularly in South America and Africa, there are current epidemics produced from rickettsial infections carried by fleas, such as R. typhi, or ticks, like R. rickettsii, or mice, like R. felis.
For clinical detection of these forms of typhus, there are differences. Although all are associated with a rapid onset of fever, headache, and myalgia, subtle signs can be helpful in making a diagnosis while waiting for laboratory confirmation. For example, scrub typhus, unlike Rocky Mountain Fever, which is caused by tick bites, does not generally include a rash. Rather, eschars, which are small patches of necrotic skin, are far more characteristic.
“Serological tests are the most common diagnostic tool for typhus, but serology may not allow early diagnosis. You can obtain a false positive in the early stages of disease,” Dr. Varghese warned. To speed the diagnosis, he said that looking for the clinical clues characteristic of the suspected form of typhus, such as the scrub typhus-associated eschar, “is valuable.” However, he also emphasized that even with positive serology results, “good epidemiology and history is helpful for laboratory interpretation.”
A variety of serological tests can identify typhus pathogens, but ELISA is now the most widely used, according to Dr. Varghese, noting that this test offers a sensitivity of 93% and a specificity of 91%. Both are higher than those provided by alternatives. As a result of improved sensitivity of diagnostic tests, prevalence rates of some forms of typhus have proved to be unexpectedly high. For example, in a study undertaken in his region of India, the seroprevalence of scrub typhus was 31.8% (Trop Med Int Health. 2017;22:576-82. doi: 10.1111/tmi.12853).
Of unmet needs in the clinical management of typhus, Dr. Varghese listed better strategies for point-of-care diagnosis and treatment and better data on how to manage patients who are severely ill. Advanced disease, which is common to rural areas with limited access to health care, is the source of almost all typhus mortality, according to Dr. Varghese. He described a trial now being initiated in severe disease that will compare intravenous doxycycline to IV azithromycin and to a combination of both IV doxycycline and azithromycin.
Although Dr. Varghese cautioned that reports of resistant typhus infections, particularly in Thailand, might prove to be the next big clinical challenge in typhus, he said that progress is being made toward reducing the burden of this disease in his area of the world. In a disease associated with a mortality of 50% if left untreated, he attributes gains to earlier diagnosis and prompt treatment.
At his medical center, “we have been working with this disease for a decade and a half,” he said, referring to scrub typhus. “When we started off, the mortality was around 15% after diagnosis. Today, the mortality is about 5%-7%.”
Dr. Varghese reported that he has no financial relationships relevant to this topic. The event was the combined annual meetings of the Infectious Diseases Society of America, the Society for Healthcare Epidemiology of America, the HIV Medicine Association, and the Pediatric Infectious Diseases Society.
SAN DIEGO – Typhus in many forms, particularly scrub typhus, has reemerged worldwide, but none of these rickettsial infections poses a significant public health threat if promptly diagnosed, according to an update presented at an annual scientific meeting on infectious diseases.
Scrub typhus, which is spread by several species of trombiculid mites or chiggers, poses a large threat in regard to typhus epidemics, particularly in Asia, but sporadic cases of different types of typhus are being seen everywhere, including in the United States, according to George M. Varghese, MD, department of infectious diseases, Christian Medical College, Vellore, India.
“The typhus diseases are clinically similar but epidemiologically and etiologically distinct,” reported Dr. Varghese, “but doxycycline is the drug of choice for almost all of the rickettsial infections.”

The bacteria responsible for scrub typhus is Orientia tsutsugamushi, which is no longer included in the genus Rickettsia, but Dr. Varghese, who has published frequently on the epidemiology of scrub typhus, said that it is still appropriately grouped among rickettsial infections. It shares many features with the other rickettsioses, which were considered to be fading but are now resurging after the large epidemics that occurred prior to the introduction of antibiotics.
In the World Wars, Rickettsia prowazekii – which is carried and spread by body lice – was the most well known typhus threat. According to Dr. Varghese, this bacterium may have killed more soldiers in these conflicts than did firearms. Although R. prowazekii has not disappeared as a source of typhus outbreaks, particularly in South America and Africa, there are current epidemics produced from rickettsial infections carried by fleas, such as R. typhi, or ticks, like R. rickettsii, or mice, like R. felis.
For clinical detection of these forms of typhus, there are differences. Although all are associated with a rapid onset of fever, headache, and myalgia, subtle signs can be helpful in making a diagnosis while waiting for laboratory confirmation. For example, scrub typhus, unlike Rocky Mountain Fever, which is caused by tick bites, does not generally include a rash. Rather, eschars, which are small patches of necrotic skin, are far more characteristic.
“Serological tests are the most common diagnostic tool for typhus, but serology may not allow early diagnosis. You can obtain a false positive in the early stages of disease,” Dr. Varghese warned. To speed the diagnosis, he said that looking for the clinical clues characteristic of the suspected form of typhus, such as the scrub typhus-associated eschar, “is valuable.” However, he also emphasized that even with positive serology results, “good epidemiology and history is helpful for laboratory interpretation.”
A variety of serological tests can identify typhus pathogens, but ELISA is now the most widely used, according to Dr. Varghese, noting that this test offers a sensitivity of 93% and a specificity of 91%. Both are higher than those provided by alternatives. As a result of improved sensitivity of diagnostic tests, prevalence rates of some forms of typhus have proved to be unexpectedly high. For example, in a study undertaken in his region of India, the seroprevalence of scrub typhus was 31.8% (Trop Med Int Health. 2017;22:576-82. doi: 10.1111/tmi.12853).
Of unmet needs in the clinical management of typhus, Dr. Varghese listed better strategies for point-of-care diagnosis and treatment and better data on how to manage patients who are severely ill. Advanced disease, which is common to rural areas with limited access to health care, is the source of almost all typhus mortality, according to Dr. Varghese. He described a trial now being initiated in severe disease that will compare intravenous doxycycline to IV azithromycin and to a combination of both IV doxycycline and azithromycin.
Although Dr. Varghese cautioned that reports of resistant typhus infections, particularly in Thailand, might prove to be the next big clinical challenge in typhus, he said that progress is being made toward reducing the burden of this disease in his area of the world. In a disease associated with a mortality of 50% if left untreated, he attributes gains to earlier diagnosis and prompt treatment.
At his medical center, “we have been working with this disease for a decade and a half,” he said, referring to scrub typhus. “When we started off, the mortality was around 15% after diagnosis. Today, the mortality is about 5%-7%.”
Dr. Varghese reported that he has no financial relationships relevant to this topic. The event was the combined annual meetings of the Infectious Diseases Society of America, the Society for Healthcare Epidemiology of America, the HIV Medicine Association, and the Pediatric Infectious Diseases Society.
AT IDWEEK 2017
Key clinical point:
Major finding: Almost all of the estimated 15,000 annual global deaths attributed to rickettsial infections could be eliminated with prompt doxycycline therapy.
Data source: Topic review.
Disclosures: Dr. Varghese reported that he has no relevant conflicts to disclose.
Study: Macrolide treatment lowers risk of failure in pediatric CAP patients
SAN DIEGO – Macrolide use showed lower treatment failure rates than did amoxicillin or beta-lactam treatment for pediatric community acquired pneumonia (CAP) patients, according to a study presented at an annual scientific meeting on infectious diseases.
While guidelines recommend amoxicillin as the first-line therapy against CAP, investigators have noticed an increase in macrolide prescriptions to pediatric outpatients, despite reported shortcomings in its use against atypical pneumonia.
“Macrolides are probably prescribed out of proportion to the presence of atypical pneumonia in that practice setting,” said Lori Handy, MD, of Children’s Hospital of Philadelphia. This could be an issue, according to Dr. Handy: “We also know that depending on the study, up to 40% of Streptococcus pneumoniae is resistant to macrolides, meaning there are children out there who may have S. pneumoniae who are receiving therapy not targeted at their disease pathogen.”
To examine the possible impact of an increase in macrolide prescriptions, the investigators conducted a retrospective cohort study of 10,470 CAP pediatric patients across 31 primary care practices in the Children’s Hospital of Philadelphia network who were diagnosed between January 2009 and December 2013.
The studied cohort was split into three groups based on treatment options: amoxicillin monotherapy (4,252, 40.6%), macrolide monotherapy (4,459, 42.6%), and broad-spectrum beta-lactams (1,759, 16.8%).
Patient age ranged from 3 months to 18 years, the majority were white, with a roughly equal number of each sex. Of the children studied, 634 (6.1%) experienced treatment failure, defined as a change in antibiotics, an emergency department visit for related symptoms, or hospitalization for pneumonia, all of which had to occur more than 24 hours after a pediatric visit, according to Dr. Handy.
Of the children who failed treatment, 341 (54%) were in the amoxicillin group, 145 (23%) were in the macrolide group, and 147 (23%) were in the broad-spectrum group.
Patients younger than 5 years old who received macrolide therapy were half as likely to experience treatment failure compared with those given amoxicillin (odds ratio [OR] .52 [95% confidence interval (CI), 0.34-0.78]).
“What this translates to in practice is that about 32 children would need to treated with macrolides to prevent one failure in the amoxicillin group,” said Dr. Handy.
Patients 5 years and older showed even lower odds of treatment failure, at approximately one-third the rate of amoxicillin treated patients (OR .31 [95% CI, 0.23-0.92]).
Dr. Handy stated that the retrospective nature of the study and the possibility of changes in the epidemiology of CAP occurring since 2013 should be considered when evaluating the findings.
In addition, she pointed out, CAP is a clinical diagnosis, and there is generally no microbiological data associated with it in order to determine the etiology of the infection.
Overall, in healthy children with CAP, it would be better to use macrolide antibiotics compared with amoxicillin, Dr. Handy concluded. However, without the microbiological data, a more randomized, controlled trial would be needed to determine how to best treat these patients, she added.
During discussion, members of the audience asked about the appropriateness of measuring a change in antibiotics as an endpoint, especially in children with viral pneumonia, who may have had parents request stronger medication when their children did not improve quickly enough.
The 47 patients who were hospitalized would not have provided enough control to properly test the results, Dr. Handy replied, although she did acknowledge the potential issue of viral infections.
She stated the need for further study to assess its possible impact, saying she didn’t know whether viral infections may have skewed their results. “Either they’ve done nothing because they’re equally distributed among the groups or they’ve pushed them one way or the other way,” she said.
Dr. Handy and her colleagues reported having no relevant financial disclosures. The event was the combined annual meetings of the Infectious Diseases Society of America, the Society for Healthcare Epidemiology of America, the HIV Medicine Association, and the Pediatric Infectious Diseases Society.
[email protected]
On Twitter @eaztweets
SAN DIEGO – Macrolide use showed lower treatment failure rates than did amoxicillin or beta-lactam treatment for pediatric community acquired pneumonia (CAP) patients, according to a study presented at an annual scientific meeting on infectious diseases.
While guidelines recommend amoxicillin as the first-line therapy against CAP, investigators have noticed an increase in macrolide prescriptions to pediatric outpatients, despite reported shortcomings in its use against atypical pneumonia.
“Macrolides are probably prescribed out of proportion to the presence of atypical pneumonia in that practice setting,” said Lori Handy, MD, of Children’s Hospital of Philadelphia. This could be an issue, according to Dr. Handy: “We also know that depending on the study, up to 40% of Streptococcus pneumoniae is resistant to macrolides, meaning there are children out there who may have S. pneumoniae who are receiving therapy not targeted at their disease pathogen.”
To examine the possible impact of an increase in macrolide prescriptions, the investigators conducted a retrospective cohort study of 10,470 CAP pediatric patients across 31 primary care practices in the Children’s Hospital of Philadelphia network who were diagnosed between January 2009 and December 2013.
The studied cohort was split into three groups based on treatment options: amoxicillin monotherapy (4,252, 40.6%), macrolide monotherapy (4,459, 42.6%), and broad-spectrum beta-lactams (1,759, 16.8%).
Patient age ranged from 3 months to 18 years, the majority were white, with a roughly equal number of each sex. Of the children studied, 634 (6.1%) experienced treatment failure, defined as a change in antibiotics, an emergency department visit for related symptoms, or hospitalization for pneumonia, all of which had to occur more than 24 hours after a pediatric visit, according to Dr. Handy.
Of the children who failed treatment, 341 (54%) were in the amoxicillin group, 145 (23%) were in the macrolide group, and 147 (23%) were in the broad-spectrum group.
Patients younger than 5 years old who received macrolide therapy were half as likely to experience treatment failure compared with those given amoxicillin (odds ratio [OR] .52 [95% confidence interval (CI), 0.34-0.78]).
“What this translates to in practice is that about 32 children would need to treated with macrolides to prevent one failure in the amoxicillin group,” said Dr. Handy.
Patients 5 years and older showed even lower odds of treatment failure, at approximately one-third the rate of amoxicillin treated patients (OR .31 [95% CI, 0.23-0.92]).
Dr. Handy stated that the retrospective nature of the study and the possibility of changes in the epidemiology of CAP occurring since 2013 should be considered when evaluating the findings.
In addition, she pointed out, CAP is a clinical diagnosis, and there is generally no microbiological data associated with it in order to determine the etiology of the infection.
Overall, in healthy children with CAP, it would be better to use macrolide antibiotics compared with amoxicillin, Dr. Handy concluded. However, without the microbiological data, a more randomized, controlled trial would be needed to determine how to best treat these patients, she added.
During discussion, members of the audience asked about the appropriateness of measuring a change in antibiotics as an endpoint, especially in children with viral pneumonia, who may have had parents request stronger medication when their children did not improve quickly enough.
The 47 patients who were hospitalized would not have provided enough control to properly test the results, Dr. Handy replied, although she did acknowledge the potential issue of viral infections.
She stated the need for further study to assess its possible impact, saying she didn’t know whether viral infections may have skewed their results. “Either they’ve done nothing because they’re equally distributed among the groups or they’ve pushed them one way or the other way,” she said.
Dr. Handy and her colleagues reported having no relevant financial disclosures. The event was the combined annual meetings of the Infectious Diseases Society of America, the Society for Healthcare Epidemiology of America, the HIV Medicine Association, and the Pediatric Infectious Diseases Society.
[email protected]
On Twitter @eaztweets
SAN DIEGO – Macrolide use showed lower treatment failure rates than did amoxicillin or beta-lactam treatment for pediatric community acquired pneumonia (CAP) patients, according to a study presented at an annual scientific meeting on infectious diseases.
While guidelines recommend amoxicillin as the first-line therapy against CAP, investigators have noticed an increase in macrolide prescriptions to pediatric outpatients, despite reported shortcomings in its use against atypical pneumonia.
“Macrolides are probably prescribed out of proportion to the presence of atypical pneumonia in that practice setting,” said Lori Handy, MD, of Children’s Hospital of Philadelphia. This could be an issue, according to Dr. Handy: “We also know that depending on the study, up to 40% of Streptococcus pneumoniae is resistant to macrolides, meaning there are children out there who may have S. pneumoniae who are receiving therapy not targeted at their disease pathogen.”
To examine the possible impact of an increase in macrolide prescriptions, the investigators conducted a retrospective cohort study of 10,470 CAP pediatric patients across 31 primary care practices in the Children’s Hospital of Philadelphia network who were diagnosed between January 2009 and December 2013.
The studied cohort was split into three groups based on treatment options: amoxicillin monotherapy (4,252, 40.6%), macrolide monotherapy (4,459, 42.6%), and broad-spectrum beta-lactams (1,759, 16.8%).
Patient age ranged from 3 months to 18 years, the majority were white, with a roughly equal number of each sex. Of the children studied, 634 (6.1%) experienced treatment failure, defined as a change in antibiotics, an emergency department visit for related symptoms, or hospitalization for pneumonia, all of which had to occur more than 24 hours after a pediatric visit, according to Dr. Handy.
Of the children who failed treatment, 341 (54%) were in the amoxicillin group, 145 (23%) were in the macrolide group, and 147 (23%) were in the broad-spectrum group.
Patients younger than 5 years old who received macrolide therapy were half as likely to experience treatment failure compared with those given amoxicillin (odds ratio [OR] .52 [95% confidence interval (CI), 0.34-0.78]).
“What this translates to in practice is that about 32 children would need to treated with macrolides to prevent one failure in the amoxicillin group,” said Dr. Handy.
Patients 5 years and older showed even lower odds of treatment failure, at approximately one-third the rate of amoxicillin treated patients (OR .31 [95% CI, 0.23-0.92]).
Dr. Handy stated that the retrospective nature of the study and the possibility of changes in the epidemiology of CAP occurring since 2013 should be considered when evaluating the findings.
In addition, she pointed out, CAP is a clinical diagnosis, and there is generally no microbiological data associated with it in order to determine the etiology of the infection.
Overall, in healthy children with CAP, it would be better to use macrolide antibiotics compared with amoxicillin, Dr. Handy concluded. However, without the microbiological data, a more randomized, controlled trial would be needed to determine how to best treat these patients, she added.
During discussion, members of the audience asked about the appropriateness of measuring a change in antibiotics as an endpoint, especially in children with viral pneumonia, who may have had parents request stronger medication when their children did not improve quickly enough.
The 47 patients who were hospitalized would not have provided enough control to properly test the results, Dr. Handy replied, although she did acknowledge the potential issue of viral infections.
She stated the need for further study to assess its possible impact, saying she didn’t know whether viral infections may have skewed their results. “Either they’ve done nothing because they’re equally distributed among the groups or they’ve pushed them one way or the other way,” she said.
Dr. Handy and her colleagues reported having no relevant financial disclosures. The event was the combined annual meetings of the Infectious Diseases Society of America, the Society for Healthcare Epidemiology of America, the HIV Medicine Association, and the Pediatric Infectious Diseases Society.
[email protected]
On Twitter @eaztweets
AT ID WEEK 2017
Key clinical point:
Major finding: Macrolide treatment was associated with treatment failure OR of .52 in patients younger than 5 years and .31 among patients older than 5 years.
Data source: Retrospective study of 10,460 pediatric patients receiving antibiotics for community acquired pneumonia during 2009-2013.
Disclosures: Dr. Handy and her colleagues reported having no relevant financial disclosures.
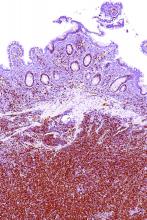
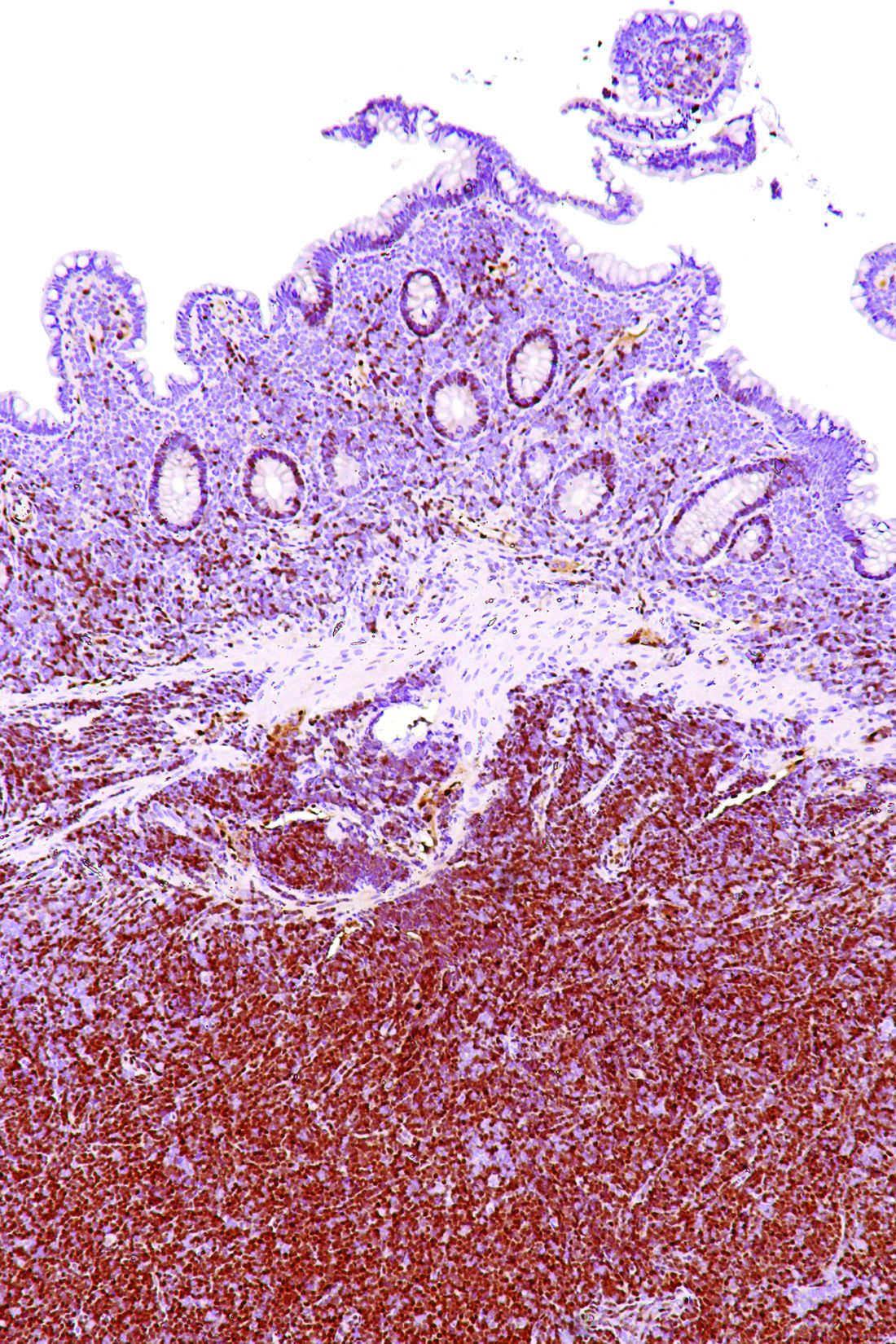
Rare type of MCL mimics Castleman disease
A rare type of mantle cell lymphoma (MCL) has features that are similar to those of Castleman disease, according to a recent report based on three patient cases.
Lymph node biopsies for these patients initially indicated histologic features consistent with those of plasma cell (PC)-type Castleman disease, reported Takuro Igawa, MD, PhD, of Okayama (Japan) University Graduate School of Medicine, Dentistry, and Pharmaceutical Sciences, and his coauthors. Further work-up, including flow cytometric analysis and cyclin D1 immunostaining, showed features consistent with those of MCL.
“This rare type of MCL can mimic Castleman disease in the clinical setting and upon histological examination,” Dr. Igawa and his colleagues wrote (Pathol Res Pract. 2017 Sep 18. pii: S0344-0338[17]30684-2. doi: 10.1016/j.prp.2017.09.015). “These confusing characteristics can make the diagnosis challenging, and careful flow cytometric analysis is recommended when a histopathological diagnosis is made.”
The patients in the study, all male, were 51, 74, and 81 years of age. Each presented with systemic lymphadenopathy, along with abnormal laboratory findings that according to the investigators are not usually associated with B-cell lymphomas such as MCL, including anemia, polyclonal IgG hypergammaglobulinemia, and elevated levels of C-reactive protein.
In lymph node biopsy specimens, the MCL component was “masked by histological features of PC-type Castleman disease” such as interfollicular plasmacytosis and atrophic germinal centers, the researchers wrote.
However, further pathologic investigations revealed features that were “essential to distinguish these 3 cases of MCL from PC-type Castleman disease,” they added.
In particular, an abnormal B-cell population was found using flow cytometric analysis, while subsequent cyclin D1 immunostaining in all three cases showed abnormal B-cells primarily in the mantle zone that were positive for CD20 and CD5, both typically expressed by MCL, along with SOX11, which is an “excellent diagnostic marker for MCL, including atypical MCL,” the investigators wrote.
These case reports also provide some evidence that interleukin-6 (IL-6), which is thought to be a driver of Castleman disease, might also be implicated in the pathogenesis of this rare MCL variant. the researchers found that all three cases had positive IL-6 staining in the interfollicular areas.
If further studies confirm the role of IL-6 in this rare setting, “specific treatments other than chemotherapy could potentially be used for patients with MCL with features of Castleman disease, such as an anti-IL-6 receptor antibody (tocilizumab), which is already used for patients with Castleman disease,” they said.
A rare type of mantle cell lymphoma (MCL) has features that are similar to those of Castleman disease, according to a recent report based on three patient cases.
Lymph node biopsies for these patients initially indicated histologic features consistent with those of plasma cell (PC)-type Castleman disease, reported Takuro Igawa, MD, PhD, of Okayama (Japan) University Graduate School of Medicine, Dentistry, and Pharmaceutical Sciences, and his coauthors. Further work-up, including flow cytometric analysis and cyclin D1 immunostaining, showed features consistent with those of MCL.
“This rare type of MCL can mimic Castleman disease in the clinical setting and upon histological examination,” Dr. Igawa and his colleagues wrote (Pathol Res Pract. 2017 Sep 18. pii: S0344-0338[17]30684-2. doi: 10.1016/j.prp.2017.09.015). “These confusing characteristics can make the diagnosis challenging, and careful flow cytometric analysis is recommended when a histopathological diagnosis is made.”
The patients in the study, all male, were 51, 74, and 81 years of age. Each presented with systemic lymphadenopathy, along with abnormal laboratory findings that according to the investigators are not usually associated with B-cell lymphomas such as MCL, including anemia, polyclonal IgG hypergammaglobulinemia, and elevated levels of C-reactive protein.
In lymph node biopsy specimens, the MCL component was “masked by histological features of PC-type Castleman disease” such as interfollicular plasmacytosis and atrophic germinal centers, the researchers wrote.
However, further pathologic investigations revealed features that were “essential to distinguish these 3 cases of MCL from PC-type Castleman disease,” they added.
In particular, an abnormal B-cell population was found using flow cytometric analysis, while subsequent cyclin D1 immunostaining in all three cases showed abnormal B-cells primarily in the mantle zone that were positive for CD20 and CD5, both typically expressed by MCL, along with SOX11, which is an “excellent diagnostic marker for MCL, including atypical MCL,” the investigators wrote.
These case reports also provide some evidence that interleukin-6 (IL-6), which is thought to be a driver of Castleman disease, might also be implicated in the pathogenesis of this rare MCL variant. the researchers found that all three cases had positive IL-6 staining in the interfollicular areas.
If further studies confirm the role of IL-6 in this rare setting, “specific treatments other than chemotherapy could potentially be used for patients with MCL with features of Castleman disease, such as an anti-IL-6 receptor antibody (tocilizumab), which is already used for patients with Castleman disease,” they said.
A rare type of mantle cell lymphoma (MCL) has features that are similar to those of Castleman disease, according to a recent report based on three patient cases.
Lymph node biopsies for these patients initially indicated histologic features consistent with those of plasma cell (PC)-type Castleman disease, reported Takuro Igawa, MD, PhD, of Okayama (Japan) University Graduate School of Medicine, Dentistry, and Pharmaceutical Sciences, and his coauthors. Further work-up, including flow cytometric analysis and cyclin D1 immunostaining, showed features consistent with those of MCL.
“This rare type of MCL can mimic Castleman disease in the clinical setting and upon histological examination,” Dr. Igawa and his colleagues wrote (Pathol Res Pract. 2017 Sep 18. pii: S0344-0338[17]30684-2. doi: 10.1016/j.prp.2017.09.015). “These confusing characteristics can make the diagnosis challenging, and careful flow cytometric analysis is recommended when a histopathological diagnosis is made.”
The patients in the study, all male, were 51, 74, and 81 years of age. Each presented with systemic lymphadenopathy, along with abnormal laboratory findings that according to the investigators are not usually associated with B-cell lymphomas such as MCL, including anemia, polyclonal IgG hypergammaglobulinemia, and elevated levels of C-reactive protein.
In lymph node biopsy specimens, the MCL component was “masked by histological features of PC-type Castleman disease” such as interfollicular plasmacytosis and atrophic germinal centers, the researchers wrote.
However, further pathologic investigations revealed features that were “essential to distinguish these 3 cases of MCL from PC-type Castleman disease,” they added.
In particular, an abnormal B-cell population was found using flow cytometric analysis, while subsequent cyclin D1 immunostaining in all three cases showed abnormal B-cells primarily in the mantle zone that were positive for CD20 and CD5, both typically expressed by MCL, along with SOX11, which is an “excellent diagnostic marker for MCL, including atypical MCL,” the investigators wrote.
These case reports also provide some evidence that interleukin-6 (IL-6), which is thought to be a driver of Castleman disease, might also be implicated in the pathogenesis of this rare MCL variant. the researchers found that all three cases had positive IL-6 staining in the interfollicular areas.
If further studies confirm the role of IL-6 in this rare setting, “specific treatments other than chemotherapy could potentially be used for patients with MCL with features of Castleman disease, such as an anti-IL-6 receptor antibody (tocilizumab), which is already used for patients with Castleman disease,” they said.
FROM PATHOLOGY – RESEARCH AND PRACTICE
Key clinical point:
Major finding: Lymph node biopsy revealed histologic features consistent with plasma cell (PC)-type Castleman disease, but cyclin D1 immunostaining and flow cytometric analysis showed features consistent with a diagnosis of MCL.
Data source: A report on three patient cases of MCL with features of PC-type Castleman disease retrieved from surgical pathology consultation files.
Disclosures: The authors reported no conflicts of interest.
Plenary sessions at ANA 2017 cover wide spectrum of neurologic topics
The six plenary sessions of the annual meeting of the American Neurological Association, taking place Oct. 15-17 in San Diego, promise to cover a broad range of research areas, including neuronal circuits and behavior, global neurology, precision medicine, antisense oligonucleotide therapies, and molecular imaging.
The morning of Oct. 15 starts off with the plenary session, “Linking Circuits to Behavior: Promise & Peril,” which seeks to impart how technologies such as optogenetics enable manipulation of discrete neural populations but require careful consideration of the methods for interpreting the resulting data in order to translate it to human functional neuroimaging for potential therapeutic use.
Later, in the afternoon of Oct. 15, the traditional Derek Denny-Brown Young Neurological Scholar Symposium will showcase the presentations from the two clinical science winners and one basic science winner of the Derek Denny-Brown Young Neurological Scholar Awards, as well as the 2017 Distinguished Neurology Teacher Award, the 2017 Grass Foundation ANA Award in Neuroscience, and the 2017 Wolfe Neuropathy Research Prize. The Derek Denny-Brown Young Neurological Scholar Award recognizes neurologists and neuroscientists in the first 10 years of their career at the assistant/associate faculty (equivalent) level who have made outstanding basic and clinical scientific advances toward the prevention, diagnosis, treatment, and cure of neurologic diseases. This year, award winner Keven N. Sheth, MD, of Yale University, New Haven, Conn., will present on “Instructive, Pragmatic, and Successful Trials in Acute Brain Injury: Making Intracerebral Hemorrhage the LEAST Devastating Form of Stroke”; Leslie E. Skolarus, MD, of the University of Michigan, Ann Arbor, will present on “Reducing the Burden of Stroke in a Disadvantaged Community”; and Conrad Chris Weihl, MD, PhD, of Washington University in St. Louis will present on “Connecting Protein Quality Control Pathways in Skeletal Muscle and Muscle Disease.” The 2017 Distinguished Neurology Teacher Award goes to Zachary Nathaniel London, MD, of the University of Michigan, Ann Arbor. The winner of this year’s Grass Foundation ANA Award in Neuroscience, which goes to an outstanding young physician-scientist conducting research in basic or clinical neuroscience, is Clotilde Lagier-Tourenne, MD, PhD, of Massachusetts General Hospital, Boston, who will discuss “Modeling C9ORF72 Disease: A Crucial Step for Therapeutic Development in ALS and Frontotemporal Dementia.” The symposium’s final presentation will have Stefanie Geisler, MD, of Washington University in St. Louis, talk about “Targeting a Core Axonal Degeneration Program to Treat Vincristine and Bortezomib-Induced Axonal Degeneration.” Dr. Geisler won the Wolfe Neuropathy Research Prize, which honors outstanding investigators who identify a new cause or treatment of axonal peripheral neuropathy.
The morning plenary session on Oct. 16 will focus on translational neuroscience efforts that are paying off with discoveries and insights into neurologic disorders that have higher prevalence or greater relevance to low- and middle-income countries. Presentations on these efforts will include discussion of the causation and prevention of Konzo, a distinct upper–motor neuron disease associated with cassava cyanogenic poisoning in sub-Saharan Africa; a case-control study on the impact of multiple mycotoxins on the development of Nodding syndrome in northern Uganda; efforts to address neurologic manifestations of sexually transmitted virus infections in Peru; a longitudinal cohort study of neurologic sequelae in Ebola virus disease survivors in Liberia; efforts to protect against cerebral malaria; the epidemiology of peripheral neuropathy in urban and rural Bangladeshi type 2 diabetes patients; and the use of smartphones and teleconsultations to improve care for people with epilepsy in low- and middle-income countries.
“Precision Medicine in Neurologic Disease” is the theme of four presentations in the afternoon plenary session on Oct. 16. Huda Y. Zoghbi, MD, of Baylor University, and Texas Children’s Hospital in Houston will talk about how her work in animal models of disease has enabled new insights into the effect that certain regulator proteins have on levels of disease-driving proteins such as tau and alpha-synuclein in neurodegenerative diseases. Amy Wagers, PhD, of Harvard Medical School, Boston, will describe her lab’s use of the gene-editing potential of the CRISPR-Cas9 system to fix frame-disrupting mutations in the Duchenne muscular dystrophy gene, DMD, which encodes dystrophin, and produce functional dystrophin expression in muscle stem cells in a mouse model of the disease, which partially recovered functional deficiencies of dystrophic muscle. Donald Berry, PhD, of the University of Texas, M.D. Anderson Cancer Center in Houston plans to discuss the importance of adaptive platform trials – which match therapies to patients – from oncology to neurologic therapy trials and the lessons learned from two major ongoing oncology treatment trials. Cristina Sampaio, MD, PhD, of the CHDI Foundation, aims to inform attendees of the power of prognostic and predictive biomarker-guided trials in neurology to improve the likelihood of success of drug development. Three high-scoring abstracts in the field of precision medicine also will be presented.
The final day of the meeting brings a morning plenary session on “Antisense Oligonucleotide Treatment of Genetic Neurological Diseases” that will focus on the use of antisense oligonucleotides (ASOs) to silence specific genes or alter their pre-mRNA splicing in Duchenne muscular dystrophy, spinal muscular atrophy, Huntington’s disease, amyotrophic lateral sclerosis, and tauopathies. Additional presentations will focus on abstracts about blood and salivary biomarkers in Huntington’s disease and the early efficacy and safety results of an ASO in patients with hereditary transthyretin amyloidosis with polyneuropathy.
The expanding use and development of methods to assess brain pathology in vivo sets the scene for the meeting’s final plenary session, “Molecular Imaging in Neurologic Disease” in the afternoon of Oct. 17. The use of positron emission tomography and single-photon emission computed tomography (SPECT) tracers for glucose metabolism, the dopamine system, amyloid-beta, tau, synaptic markers, and activated microglia has grown substantially to investigate disease mechanisms, develop new therapeutics, and provide diagnostic and prognostic clinical care. Reisa Sperling, MD, of Harvard Medical School, Boston, will provide an overview of the direction of PET ligand use and development in diagnosing early Alzheimer’s disease. Nicolaas I. Bohnen, MD, PhD, of the University of Michigan, Ann Arbor, will describe a hypothesis for how hypercholinergic activity in the brain of Parkinson’s disease patients may for a time compensate for the loss of striatal dopamine and influence the appearance of a tremor-predominant motor phenotype in patients. Richard E. Carson, PhD, of Yale University will focus on the development of PET ligands to monitor synaptic density loss in neuropsychiatric disorders. Noninvasive imaging has also begun to influence research in the detection of neuroinflammation in a wide variety of conditions, with most research focusing on tracers for activated microglia and astrocytes, according to speaker Martin Pomper, MD, PhD, of Johns Hopkins University, Baltimore. The session will conclude with three molecular imaging abstract presentations.
The six plenary sessions of the annual meeting of the American Neurological Association, taking place Oct. 15-17 in San Diego, promise to cover a broad range of research areas, including neuronal circuits and behavior, global neurology, precision medicine, antisense oligonucleotide therapies, and molecular imaging.
The morning of Oct. 15 starts off with the plenary session, “Linking Circuits to Behavior: Promise & Peril,” which seeks to impart how technologies such as optogenetics enable manipulation of discrete neural populations but require careful consideration of the methods for interpreting the resulting data in order to translate it to human functional neuroimaging for potential therapeutic use.
Later, in the afternoon of Oct. 15, the traditional Derek Denny-Brown Young Neurological Scholar Symposium will showcase the presentations from the two clinical science winners and one basic science winner of the Derek Denny-Brown Young Neurological Scholar Awards, as well as the 2017 Distinguished Neurology Teacher Award, the 2017 Grass Foundation ANA Award in Neuroscience, and the 2017 Wolfe Neuropathy Research Prize. The Derek Denny-Brown Young Neurological Scholar Award recognizes neurologists and neuroscientists in the first 10 years of their career at the assistant/associate faculty (equivalent) level who have made outstanding basic and clinical scientific advances toward the prevention, diagnosis, treatment, and cure of neurologic diseases. This year, award winner Keven N. Sheth, MD, of Yale University, New Haven, Conn., will present on “Instructive, Pragmatic, and Successful Trials in Acute Brain Injury: Making Intracerebral Hemorrhage the LEAST Devastating Form of Stroke”; Leslie E. Skolarus, MD, of the University of Michigan, Ann Arbor, will present on “Reducing the Burden of Stroke in a Disadvantaged Community”; and Conrad Chris Weihl, MD, PhD, of Washington University in St. Louis will present on “Connecting Protein Quality Control Pathways in Skeletal Muscle and Muscle Disease.” The 2017 Distinguished Neurology Teacher Award goes to Zachary Nathaniel London, MD, of the University of Michigan, Ann Arbor. The winner of this year’s Grass Foundation ANA Award in Neuroscience, which goes to an outstanding young physician-scientist conducting research in basic or clinical neuroscience, is Clotilde Lagier-Tourenne, MD, PhD, of Massachusetts General Hospital, Boston, who will discuss “Modeling C9ORF72 Disease: A Crucial Step for Therapeutic Development in ALS and Frontotemporal Dementia.” The symposium’s final presentation will have Stefanie Geisler, MD, of Washington University in St. Louis, talk about “Targeting a Core Axonal Degeneration Program to Treat Vincristine and Bortezomib-Induced Axonal Degeneration.” Dr. Geisler won the Wolfe Neuropathy Research Prize, which honors outstanding investigators who identify a new cause or treatment of axonal peripheral neuropathy.
The morning plenary session on Oct. 16 will focus on translational neuroscience efforts that are paying off with discoveries and insights into neurologic disorders that have higher prevalence or greater relevance to low- and middle-income countries. Presentations on these efforts will include discussion of the causation and prevention of Konzo, a distinct upper–motor neuron disease associated with cassava cyanogenic poisoning in sub-Saharan Africa; a case-control study on the impact of multiple mycotoxins on the development of Nodding syndrome in northern Uganda; efforts to address neurologic manifestations of sexually transmitted virus infections in Peru; a longitudinal cohort study of neurologic sequelae in Ebola virus disease survivors in Liberia; efforts to protect against cerebral malaria; the epidemiology of peripheral neuropathy in urban and rural Bangladeshi type 2 diabetes patients; and the use of smartphones and teleconsultations to improve care for people with epilepsy in low- and middle-income countries.
“Precision Medicine in Neurologic Disease” is the theme of four presentations in the afternoon plenary session on Oct. 16. Huda Y. Zoghbi, MD, of Baylor University, and Texas Children’s Hospital in Houston will talk about how her work in animal models of disease has enabled new insights into the effect that certain regulator proteins have on levels of disease-driving proteins such as tau and alpha-synuclein in neurodegenerative diseases. Amy Wagers, PhD, of Harvard Medical School, Boston, will describe her lab’s use of the gene-editing potential of the CRISPR-Cas9 system to fix frame-disrupting mutations in the Duchenne muscular dystrophy gene, DMD, which encodes dystrophin, and produce functional dystrophin expression in muscle stem cells in a mouse model of the disease, which partially recovered functional deficiencies of dystrophic muscle. Donald Berry, PhD, of the University of Texas, M.D. Anderson Cancer Center in Houston plans to discuss the importance of adaptive platform trials – which match therapies to patients – from oncology to neurologic therapy trials and the lessons learned from two major ongoing oncology treatment trials. Cristina Sampaio, MD, PhD, of the CHDI Foundation, aims to inform attendees of the power of prognostic and predictive biomarker-guided trials in neurology to improve the likelihood of success of drug development. Three high-scoring abstracts in the field of precision medicine also will be presented.
The final day of the meeting brings a morning plenary session on “Antisense Oligonucleotide Treatment of Genetic Neurological Diseases” that will focus on the use of antisense oligonucleotides (ASOs) to silence specific genes or alter their pre-mRNA splicing in Duchenne muscular dystrophy, spinal muscular atrophy, Huntington’s disease, amyotrophic lateral sclerosis, and tauopathies. Additional presentations will focus on abstracts about blood and salivary biomarkers in Huntington’s disease and the early efficacy and safety results of an ASO in patients with hereditary transthyretin amyloidosis with polyneuropathy.
The expanding use and development of methods to assess brain pathology in vivo sets the scene for the meeting’s final plenary session, “Molecular Imaging in Neurologic Disease” in the afternoon of Oct. 17. The use of positron emission tomography and single-photon emission computed tomography (SPECT) tracers for glucose metabolism, the dopamine system, amyloid-beta, tau, synaptic markers, and activated microglia has grown substantially to investigate disease mechanisms, develop new therapeutics, and provide diagnostic and prognostic clinical care. Reisa Sperling, MD, of Harvard Medical School, Boston, will provide an overview of the direction of PET ligand use and development in diagnosing early Alzheimer’s disease. Nicolaas I. Bohnen, MD, PhD, of the University of Michigan, Ann Arbor, will describe a hypothesis for how hypercholinergic activity in the brain of Parkinson’s disease patients may for a time compensate for the loss of striatal dopamine and influence the appearance of a tremor-predominant motor phenotype in patients. Richard E. Carson, PhD, of Yale University will focus on the development of PET ligands to monitor synaptic density loss in neuropsychiatric disorders. Noninvasive imaging has also begun to influence research in the detection of neuroinflammation in a wide variety of conditions, with most research focusing on tracers for activated microglia and astrocytes, according to speaker Martin Pomper, MD, PhD, of Johns Hopkins University, Baltimore. The session will conclude with three molecular imaging abstract presentations.
The six plenary sessions of the annual meeting of the American Neurological Association, taking place Oct. 15-17 in San Diego, promise to cover a broad range of research areas, including neuronal circuits and behavior, global neurology, precision medicine, antisense oligonucleotide therapies, and molecular imaging.
The morning of Oct. 15 starts off with the plenary session, “Linking Circuits to Behavior: Promise & Peril,” which seeks to impart how technologies such as optogenetics enable manipulation of discrete neural populations but require careful consideration of the methods for interpreting the resulting data in order to translate it to human functional neuroimaging for potential therapeutic use.
Later, in the afternoon of Oct. 15, the traditional Derek Denny-Brown Young Neurological Scholar Symposium will showcase the presentations from the two clinical science winners and one basic science winner of the Derek Denny-Brown Young Neurological Scholar Awards, as well as the 2017 Distinguished Neurology Teacher Award, the 2017 Grass Foundation ANA Award in Neuroscience, and the 2017 Wolfe Neuropathy Research Prize. The Derek Denny-Brown Young Neurological Scholar Award recognizes neurologists and neuroscientists in the first 10 years of their career at the assistant/associate faculty (equivalent) level who have made outstanding basic and clinical scientific advances toward the prevention, diagnosis, treatment, and cure of neurologic diseases. This year, award winner Keven N. Sheth, MD, of Yale University, New Haven, Conn., will present on “Instructive, Pragmatic, and Successful Trials in Acute Brain Injury: Making Intracerebral Hemorrhage the LEAST Devastating Form of Stroke”; Leslie E. Skolarus, MD, of the University of Michigan, Ann Arbor, will present on “Reducing the Burden of Stroke in a Disadvantaged Community”; and Conrad Chris Weihl, MD, PhD, of Washington University in St. Louis will present on “Connecting Protein Quality Control Pathways in Skeletal Muscle and Muscle Disease.” The 2017 Distinguished Neurology Teacher Award goes to Zachary Nathaniel London, MD, of the University of Michigan, Ann Arbor. The winner of this year’s Grass Foundation ANA Award in Neuroscience, which goes to an outstanding young physician-scientist conducting research in basic or clinical neuroscience, is Clotilde Lagier-Tourenne, MD, PhD, of Massachusetts General Hospital, Boston, who will discuss “Modeling C9ORF72 Disease: A Crucial Step for Therapeutic Development in ALS and Frontotemporal Dementia.” The symposium’s final presentation will have Stefanie Geisler, MD, of Washington University in St. Louis, talk about “Targeting a Core Axonal Degeneration Program to Treat Vincristine and Bortezomib-Induced Axonal Degeneration.” Dr. Geisler won the Wolfe Neuropathy Research Prize, which honors outstanding investigators who identify a new cause or treatment of axonal peripheral neuropathy.
The morning plenary session on Oct. 16 will focus on translational neuroscience efforts that are paying off with discoveries and insights into neurologic disorders that have higher prevalence or greater relevance to low- and middle-income countries. Presentations on these efforts will include discussion of the causation and prevention of Konzo, a distinct upper–motor neuron disease associated with cassava cyanogenic poisoning in sub-Saharan Africa; a case-control study on the impact of multiple mycotoxins on the development of Nodding syndrome in northern Uganda; efforts to address neurologic manifestations of sexually transmitted virus infections in Peru; a longitudinal cohort study of neurologic sequelae in Ebola virus disease survivors in Liberia; efforts to protect against cerebral malaria; the epidemiology of peripheral neuropathy in urban and rural Bangladeshi type 2 diabetes patients; and the use of smartphones and teleconsultations to improve care for people with epilepsy in low- and middle-income countries.
“Precision Medicine in Neurologic Disease” is the theme of four presentations in the afternoon plenary session on Oct. 16. Huda Y. Zoghbi, MD, of Baylor University, and Texas Children’s Hospital in Houston will talk about how her work in animal models of disease has enabled new insights into the effect that certain regulator proteins have on levels of disease-driving proteins such as tau and alpha-synuclein in neurodegenerative diseases. Amy Wagers, PhD, of Harvard Medical School, Boston, will describe her lab’s use of the gene-editing potential of the CRISPR-Cas9 system to fix frame-disrupting mutations in the Duchenne muscular dystrophy gene, DMD, which encodes dystrophin, and produce functional dystrophin expression in muscle stem cells in a mouse model of the disease, which partially recovered functional deficiencies of dystrophic muscle. Donald Berry, PhD, of the University of Texas, M.D. Anderson Cancer Center in Houston plans to discuss the importance of adaptive platform trials – which match therapies to patients – from oncology to neurologic therapy trials and the lessons learned from two major ongoing oncology treatment trials. Cristina Sampaio, MD, PhD, of the CHDI Foundation, aims to inform attendees of the power of prognostic and predictive biomarker-guided trials in neurology to improve the likelihood of success of drug development. Three high-scoring abstracts in the field of precision medicine also will be presented.
The final day of the meeting brings a morning plenary session on “Antisense Oligonucleotide Treatment of Genetic Neurological Diseases” that will focus on the use of antisense oligonucleotides (ASOs) to silence specific genes or alter their pre-mRNA splicing in Duchenne muscular dystrophy, spinal muscular atrophy, Huntington’s disease, amyotrophic lateral sclerosis, and tauopathies. Additional presentations will focus on abstracts about blood and salivary biomarkers in Huntington’s disease and the early efficacy and safety results of an ASO in patients with hereditary transthyretin amyloidosis with polyneuropathy.
The expanding use and development of methods to assess brain pathology in vivo sets the scene for the meeting’s final plenary session, “Molecular Imaging in Neurologic Disease” in the afternoon of Oct. 17. The use of positron emission tomography and single-photon emission computed tomography (SPECT) tracers for glucose metabolism, the dopamine system, amyloid-beta, tau, synaptic markers, and activated microglia has grown substantially to investigate disease mechanisms, develop new therapeutics, and provide diagnostic and prognostic clinical care. Reisa Sperling, MD, of Harvard Medical School, Boston, will provide an overview of the direction of PET ligand use and development in diagnosing early Alzheimer’s disease. Nicolaas I. Bohnen, MD, PhD, of the University of Michigan, Ann Arbor, will describe a hypothesis for how hypercholinergic activity in the brain of Parkinson’s disease patients may for a time compensate for the loss of striatal dopamine and influence the appearance of a tremor-predominant motor phenotype in patients. Richard E. Carson, PhD, of Yale University will focus on the development of PET ligands to monitor synaptic density loss in neuropsychiatric disorders. Noninvasive imaging has also begun to influence research in the detection of neuroinflammation in a wide variety of conditions, with most research focusing on tracers for activated microglia and astrocytes, according to speaker Martin Pomper, MD, PhD, of Johns Hopkins University, Baltimore. The session will conclude with three molecular imaging abstract presentations.
VCR regimen showed efficacy in mantle cell and indolent lymphomas
The combination of bortezomib, cladribine, and rituximab (VCR) was an effective treatment regimen for patients with CD20-positive mantle cell lymphoma (MCL) and indolent non-Hodgkin’s lymphoma (iNHL), based on results of a recent phase 2, open-label study.
The overall response rate was 92% in the single-center, 24-patient study. The 2-year progression-free survival (PFS) was 82% and 54%, respectively, for MCL and iNHL patients; PFS was 80% for treatment-naive patients and 57% for those with refractory/recalcitrant disease, according to Soham D. Puvvada, MD, of the University of Arizona Cancer Center in Tucson, and her associates.
Two-year overall survival was 91% for MCL and 69% for iNHL patients. Median time to progression was 34.5 months, and median PFS had not been reached at 2 years, according to the researchers.
While the study (NCT00980395) was small and limited by its single-center design, the VCR combination “has encouraging activity in both MCL and iNHL and could be compared to standard therapies in future studies,” the researchers wrote. “For MCL in particular, we believe a noninferiority comparison to standard therapies would be justified by our results.”
Adverse events were most commonly hematologic, and three patients experienced febrile neutropenia, data show.
“Although hematological toxicity can be an issue, the regimen provides an alternative option in transplant ineligible relapsed/refractory MCL and iNHL,” wrote Dr. Puvvada and her colleagues. The study was published in Clinical Lymphoma, Myeloma & Leukemia (doi: 10.1016/j.clml.2017.09.001).
The researchers studied the combination of bortezomib, the proteasome inhibitor initially approved for relapsed/refractory MCL, cladribine, which has shown activity and promising response rates in patients with indolent lymphomas, and rituximab in patients with CD20-positive mantle cell or indolent lymphoma.
Patients with follicular lymphomas were eligible to be included in the study if they had received at least one previous line of therapy. All other participants could be treatment naive or have relapsed after previous treatment.
Of the 24 patients enrolled, 11 had MCL, 5 had follicular lymphoma, 4 had marginal zone lymphoma, 3 had lymphoplasmacytic lymphoma, and 1 had small lymphocytic lymphoma.
The VCR regimen, given every 28 days for no more than six cycles, included rituximab at 375 mg/m2 given intravenously on day 1 of each cycle, cladribine 4 mg/m2 given intravenously over 2 hours on days 1 through 5, and bortezomib 1.3 mg/m2 given intravenously on days 1 and 4. Patients received a median of five cycles of therapy.
Adverse events of grade 3 or greater occurred in 14 patients (58%); 8 patients had leukopenia, 6 had thrombocytopenia, 5 had fatigue, and 5 had neutropenia, which included febrile neutropenia in 3 patients.
With a median follow-up of 38.5 months, overall response rate for VCR was 96%. Complete responses occurred in 8 of 23 evaluable patients (35%) and partial responses in 14 more patients (61%).
The combination of bortezomib, cladribine, and rituximab (VCR) was an effective treatment regimen for patients with CD20-positive mantle cell lymphoma (MCL) and indolent non-Hodgkin’s lymphoma (iNHL), based on results of a recent phase 2, open-label study.
The overall response rate was 92% in the single-center, 24-patient study. The 2-year progression-free survival (PFS) was 82% and 54%, respectively, for MCL and iNHL patients; PFS was 80% for treatment-naive patients and 57% for those with refractory/recalcitrant disease, according to Soham D. Puvvada, MD, of the University of Arizona Cancer Center in Tucson, and her associates.
Two-year overall survival was 91% for MCL and 69% for iNHL patients. Median time to progression was 34.5 months, and median PFS had not been reached at 2 years, according to the researchers.
While the study (NCT00980395) was small and limited by its single-center design, the VCR combination “has encouraging activity in both MCL and iNHL and could be compared to standard therapies in future studies,” the researchers wrote. “For MCL in particular, we believe a noninferiority comparison to standard therapies would be justified by our results.”
Adverse events were most commonly hematologic, and three patients experienced febrile neutropenia, data show.
“Although hematological toxicity can be an issue, the regimen provides an alternative option in transplant ineligible relapsed/refractory MCL and iNHL,” wrote Dr. Puvvada and her colleagues. The study was published in Clinical Lymphoma, Myeloma & Leukemia (doi: 10.1016/j.clml.2017.09.001).
The researchers studied the combination of bortezomib, the proteasome inhibitor initially approved for relapsed/refractory MCL, cladribine, which has shown activity and promising response rates in patients with indolent lymphomas, and rituximab in patients with CD20-positive mantle cell or indolent lymphoma.
Patients with follicular lymphomas were eligible to be included in the study if they had received at least one previous line of therapy. All other participants could be treatment naive or have relapsed after previous treatment.
Of the 24 patients enrolled, 11 had MCL, 5 had follicular lymphoma, 4 had marginal zone lymphoma, 3 had lymphoplasmacytic lymphoma, and 1 had small lymphocytic lymphoma.
The VCR regimen, given every 28 days for no more than six cycles, included rituximab at 375 mg/m2 given intravenously on day 1 of each cycle, cladribine 4 mg/m2 given intravenously over 2 hours on days 1 through 5, and bortezomib 1.3 mg/m2 given intravenously on days 1 and 4. Patients received a median of five cycles of therapy.
Adverse events of grade 3 or greater occurred in 14 patients (58%); 8 patients had leukopenia, 6 had thrombocytopenia, 5 had fatigue, and 5 had neutropenia, which included febrile neutropenia in 3 patients.
With a median follow-up of 38.5 months, overall response rate for VCR was 96%. Complete responses occurred in 8 of 23 evaluable patients (35%) and partial responses in 14 more patients (61%).
The combination of bortezomib, cladribine, and rituximab (VCR) was an effective treatment regimen for patients with CD20-positive mantle cell lymphoma (MCL) and indolent non-Hodgkin’s lymphoma (iNHL), based on results of a recent phase 2, open-label study.
The overall response rate was 92% in the single-center, 24-patient study. The 2-year progression-free survival (PFS) was 82% and 54%, respectively, for MCL and iNHL patients; PFS was 80% for treatment-naive patients and 57% for those with refractory/recalcitrant disease, according to Soham D. Puvvada, MD, of the University of Arizona Cancer Center in Tucson, and her associates.
Two-year overall survival was 91% for MCL and 69% for iNHL patients. Median time to progression was 34.5 months, and median PFS had not been reached at 2 years, according to the researchers.
While the study (NCT00980395) was small and limited by its single-center design, the VCR combination “has encouraging activity in both MCL and iNHL and could be compared to standard therapies in future studies,” the researchers wrote. “For MCL in particular, we believe a noninferiority comparison to standard therapies would be justified by our results.”
Adverse events were most commonly hematologic, and three patients experienced febrile neutropenia, data show.
“Although hematological toxicity can be an issue, the regimen provides an alternative option in transplant ineligible relapsed/refractory MCL and iNHL,” wrote Dr. Puvvada and her colleagues. The study was published in Clinical Lymphoma, Myeloma & Leukemia (doi: 10.1016/j.clml.2017.09.001).
The researchers studied the combination of bortezomib, the proteasome inhibitor initially approved for relapsed/refractory MCL, cladribine, which has shown activity and promising response rates in patients with indolent lymphomas, and rituximab in patients with CD20-positive mantle cell or indolent lymphoma.
Patients with follicular lymphomas were eligible to be included in the study if they had received at least one previous line of therapy. All other participants could be treatment naive or have relapsed after previous treatment.
Of the 24 patients enrolled, 11 had MCL, 5 had follicular lymphoma, 4 had marginal zone lymphoma, 3 had lymphoplasmacytic lymphoma, and 1 had small lymphocytic lymphoma.
The VCR regimen, given every 28 days for no more than six cycles, included rituximab at 375 mg/m2 given intravenously on day 1 of each cycle, cladribine 4 mg/m2 given intravenously over 2 hours on days 1 through 5, and bortezomib 1.3 mg/m2 given intravenously on days 1 and 4. Patients received a median of five cycles of therapy.
Adverse events of grade 3 or greater occurred in 14 patients (58%); 8 patients had leukopenia, 6 had thrombocytopenia, 5 had fatigue, and 5 had neutropenia, which included febrile neutropenia in 3 patients.
With a median follow-up of 38.5 months, overall response rate for VCR was 96%. Complete responses occurred in 8 of 23 evaluable patients (35%) and partial responses in 14 more patients (61%).
FROM LYMPHOMA, MYELOMA & LEUKEMIA
Key clinical point:
Major finding: The overall response rate was 92%, with a 2-year PFS of 82% and 54% for patients with mantle cell lymphoma (MCL) and indolent non-Hodgkin’s lymphoma (iNHL), respectively. Adverse events were most commonly hematologic, and three patients experienced febrile neutropenia.
Data source: A phase 2, open-label study including 24 patients with mantle cell or indolent lymphomas.
Disclosures: No disclosures were reported in the accepted manuscript.
FDA approves test to screen donated blood for Zika virus
The cobas Zika test has been approved for detecting the virus in whole blood, blood components, and donated organs, the U.S. Food and Drug Administration announced in a press release.
“Today’s action represents the first approval of a Zika virus detection test for use with screening the nation’s blood supply,” Peter Marks, MD, PhD, director of the FDA’s Center for Biologics Evaluation and Research, said in a press release. “Screening blood donations for the Zika virus is critical to preventing infected donations from entering the U.S. blood supply.”
In August 2016, the FDA issued a final guidance document recommending that all individual units of whole blood and blood components be screened with an investigational blood screening test available under an investigational new drug application. Data obtained on the cobas Zika test under its investigational new drug application and from additional studies performed by the manufacturer demonstrated that the cobas Zika test is effective. Testing individual samples from blood donations at five external laboratory sites resulted in a clinical specificity exceeding 99%.
The cobas Zika test is intended for use on the fully automated cobas 6800 and cobas 8800 systems. The cobas Zika test, cobas 6800, and cobas 8800 systems are manufactured by Roche Molecular Systems.
The cobas Zika test has been approved for detecting the virus in whole blood, blood components, and donated organs, the U.S. Food and Drug Administration announced in a press release.
“Today’s action represents the first approval of a Zika virus detection test for use with screening the nation’s blood supply,” Peter Marks, MD, PhD, director of the FDA’s Center for Biologics Evaluation and Research, said in a press release. “Screening blood donations for the Zika virus is critical to preventing infected donations from entering the U.S. blood supply.”
In August 2016, the FDA issued a final guidance document recommending that all individual units of whole blood and blood components be screened with an investigational blood screening test available under an investigational new drug application. Data obtained on the cobas Zika test under its investigational new drug application and from additional studies performed by the manufacturer demonstrated that the cobas Zika test is effective. Testing individual samples from blood donations at five external laboratory sites resulted in a clinical specificity exceeding 99%.
The cobas Zika test is intended for use on the fully automated cobas 6800 and cobas 8800 systems. The cobas Zika test, cobas 6800, and cobas 8800 systems are manufactured by Roche Molecular Systems.
The cobas Zika test has been approved for detecting the virus in whole blood, blood components, and donated organs, the U.S. Food and Drug Administration announced in a press release.
“Today’s action represents the first approval of a Zika virus detection test for use with screening the nation’s blood supply,” Peter Marks, MD, PhD, director of the FDA’s Center for Biologics Evaluation and Research, said in a press release. “Screening blood donations for the Zika virus is critical to preventing infected donations from entering the U.S. blood supply.”
In August 2016, the FDA issued a final guidance document recommending that all individual units of whole blood and blood components be screened with an investigational blood screening test available under an investigational new drug application. Data obtained on the cobas Zika test under its investigational new drug application and from additional studies performed by the manufacturer demonstrated that the cobas Zika test is effective. Testing individual samples from blood donations at five external laboratory sites resulted in a clinical specificity exceeding 99%.
The cobas Zika test is intended for use on the fully automated cobas 6800 and cobas 8800 systems. The cobas Zika test, cobas 6800, and cobas 8800 systems are manufactured by Roche Molecular Systems.
Emergency Imaging: Left Periorbital Swelling
Case
A 3-year-old boy was brought to the ED by his parents for evaluation of left periorbital swelling. A few days prior to presentation, the child was seen at an outpatient center where he was diagnosed with preseptal cellulitis and given an oral antibiotic. However, even after receiving three doses of the antibiotic, the periorbital swelling and redness around the child’s eye worsened, prompting this visit to the ED.
Physical examination revealed edema and erythema both above and below the left eye, with associated tenderness to palpation. A contrast-enhanced maxillofacial computed tomography (CT) scan, with special attention to the orbits, was ordered; representative images are shown (Figure 1a-1c).
What is the diagnosis?
Answer
The CT images of the orbits demonstrated edema in the superficial left eyelid (white arrows, Figure 2a and 2b) and left deep orbital septum (red arrows, Figure 2a-2c). A peripherally enhancing fluid collection centered in the left nasolacrimal gland was present (red asterisks, Figure 2b and 2c) with mild mass effect on the left globe. Opacification was also noted within the paranasal sinuses (white asterisks, Figure 2a-2c). Together these findings indicated sinusitis with dacryocystitis and orbital cellulitis.
Dacryocystitis
Dacryocystitis is an infection or inflammation of the lacrimal sac, usually developing secondary to blockage of the nasolacrimal duct. Orbital cellulitis is an infection involving the contents of the orbit, including the fat and ocular muscles. Orbital cellulitis should not be confused with preseptal cellulitis, which is an infection involving the eyelid occurring posterior to the orbital septum. While both of these conditions are more common in children than in adults, preseptal cellulitis is much more common than orbital cellulitis.
Preseptal Cellulitis
Preseptal cellulitis is typically due to local trauma, local skin infection, or dacryocystitis.1 Preseptal cellulitis rarely extends into the orbit, though a minority of cases have been reported in patients with concomitant dacryocystitis.2 Orbital cellulitis most commonly results from paranasal sinus disease, particularly of the ethmoid sinus, which is only separated from the orbit by the thin lamina papyracea.3 While both preseptal cellulitis and orbital cellulitis can cause eyelid swelling and erythema, preseptal cellulitis is typically a mild condition. Orbital cellulitis, however, is a serious medical emergency that requires prompt diagnosis and treatment to avoid loss of vision and intracranial complications, such as venous thrombosis and empyema.3
Imaging Studies
Although the clinical features of orbital cellulitis (eg, proptosis, ophthalmoplegia, pain with ocular movement) can sometimes distinguish it from preseptal cellulitis, imaging studies are helpful to confirm the diagnosis.4 As previously noted, prompt recognition, diagnosis, and treatment of orbital cellulitis are essential to avoid serious complications.
Computed tomography has a high specificity and sensitivity in detecting the extension of infection into the orbit and associated complications such as subperiosteal or intracranial abscess. For patients in whom intravenous (IV) contrast is contraindicated or who wish to avoid ionizing radiation, magnetic resonance imaging is a useful alternate modality, and diffusion-weighted imaging is particularly sensitive in diagnosing abscess.5
Treatment
1. Baring DE, Hilmi OJ. An evidence based review of periorbital cellulitis. Clin Otolaryngol. 2011;36(1):57-64. doi:10.1111/j.1749-4486.2011.02258.x.
2. Kikkawa DO, Heinz GW, Martin RT, Nunery WN, Eiseman AS. Orbital cellulitis and abscess secondary to dacryocystitis. Arch Ophthalmol. 2002;120(8):1096-1099.
3. Mathew AV, Craig E, Al-Mahmoud R, et al. Paediatric post-septal and pre-septal cellulitis: 10 years’ experience at a tertiary-level children’s hospital. Br J Radiol. 2014;87(1033):20130503. doi:10.1259/bjr.20130503.
4. Rudloe TF, Harper MB, Prabhu SP, Rahbar R, Vanderveen D, Kimia AA. Acute periorbital infections: who needs emergent imaging? Pediatrics. 2010;125(4):e719-e726. doi:10.1542/peds.2009-1709.
5. Sepahdari AR, Aakalu VK, Kapur R, et al. MRI of orbital cellulitis and orbital abscess: the role of diffusion-weighted imaging. AJR Am J Roentgenol. 2009;193(3):W244-W250. doi:10.2214/AJR.08.1838.
6. Ho CF, Huang YC, Wang CJ, Chiu CH, Lin TY. Clinical analysis of computed tomography-staged orbital cellulitis in children. J Microbiol Immunol Infect. 2007;40(6):518-524.
Case
A 3-year-old boy was brought to the ED by his parents for evaluation of left periorbital swelling. A few days prior to presentation, the child was seen at an outpatient center where he was diagnosed with preseptal cellulitis and given an oral antibiotic. However, even after receiving three doses of the antibiotic, the periorbital swelling and redness around the child’s eye worsened, prompting this visit to the ED.
Physical examination revealed edema and erythema both above and below the left eye, with associated tenderness to palpation. A contrast-enhanced maxillofacial computed tomography (CT) scan, with special attention to the orbits, was ordered; representative images are shown (Figure 1a-1c).
What is the diagnosis?
Answer
The CT images of the orbits demonstrated edema in the superficial left eyelid (white arrows, Figure 2a and 2b) and left deep orbital septum (red arrows, Figure 2a-2c). A peripherally enhancing fluid collection centered in the left nasolacrimal gland was present (red asterisks, Figure 2b and 2c) with mild mass effect on the left globe. Opacification was also noted within the paranasal sinuses (white asterisks, Figure 2a-2c). Together these findings indicated sinusitis with dacryocystitis and orbital cellulitis.
Dacryocystitis
Dacryocystitis is an infection or inflammation of the lacrimal sac, usually developing secondary to blockage of the nasolacrimal duct. Orbital cellulitis is an infection involving the contents of the orbit, including the fat and ocular muscles. Orbital cellulitis should not be confused with preseptal cellulitis, which is an infection involving the eyelid occurring posterior to the orbital septum. While both of these conditions are more common in children than in adults, preseptal cellulitis is much more common than orbital cellulitis.
Preseptal Cellulitis
Preseptal cellulitis is typically due to local trauma, local skin infection, or dacryocystitis.1 Preseptal cellulitis rarely extends into the orbit, though a minority of cases have been reported in patients with concomitant dacryocystitis.2 Orbital cellulitis most commonly results from paranasal sinus disease, particularly of the ethmoid sinus, which is only separated from the orbit by the thin lamina papyracea.3 While both preseptal cellulitis and orbital cellulitis can cause eyelid swelling and erythema, preseptal cellulitis is typically a mild condition. Orbital cellulitis, however, is a serious medical emergency that requires prompt diagnosis and treatment to avoid loss of vision and intracranial complications, such as venous thrombosis and empyema.3
Imaging Studies
Although the clinical features of orbital cellulitis (eg, proptosis, ophthalmoplegia, pain with ocular movement) can sometimes distinguish it from preseptal cellulitis, imaging studies are helpful to confirm the diagnosis.4 As previously noted, prompt recognition, diagnosis, and treatment of orbital cellulitis are essential to avoid serious complications.
Computed tomography has a high specificity and sensitivity in detecting the extension of infection into the orbit and associated complications such as subperiosteal or intracranial abscess. For patients in whom intravenous (IV) contrast is contraindicated or who wish to avoid ionizing radiation, magnetic resonance imaging is a useful alternate modality, and diffusion-weighted imaging is particularly sensitive in diagnosing abscess.5
Treatment
Case
A 3-year-old boy was brought to the ED by his parents for evaluation of left periorbital swelling. A few days prior to presentation, the child was seen at an outpatient center where he was diagnosed with preseptal cellulitis and given an oral antibiotic. However, even after receiving three doses of the antibiotic, the periorbital swelling and redness around the child’s eye worsened, prompting this visit to the ED.
Physical examination revealed edema and erythema both above and below the left eye, with associated tenderness to palpation. A contrast-enhanced maxillofacial computed tomography (CT) scan, with special attention to the orbits, was ordered; representative images are shown (Figure 1a-1c).
What is the diagnosis?
Answer
The CT images of the orbits demonstrated edema in the superficial left eyelid (white arrows, Figure 2a and 2b) and left deep orbital septum (red arrows, Figure 2a-2c). A peripherally enhancing fluid collection centered in the left nasolacrimal gland was present (red asterisks, Figure 2b and 2c) with mild mass effect on the left globe. Opacification was also noted within the paranasal sinuses (white asterisks, Figure 2a-2c). Together these findings indicated sinusitis with dacryocystitis and orbital cellulitis.
Dacryocystitis
Dacryocystitis is an infection or inflammation of the lacrimal sac, usually developing secondary to blockage of the nasolacrimal duct. Orbital cellulitis is an infection involving the contents of the orbit, including the fat and ocular muscles. Orbital cellulitis should not be confused with preseptal cellulitis, which is an infection involving the eyelid occurring posterior to the orbital septum. While both of these conditions are more common in children than in adults, preseptal cellulitis is much more common than orbital cellulitis.
Preseptal Cellulitis
Preseptal cellulitis is typically due to local trauma, local skin infection, or dacryocystitis.1 Preseptal cellulitis rarely extends into the orbit, though a minority of cases have been reported in patients with concomitant dacryocystitis.2 Orbital cellulitis most commonly results from paranasal sinus disease, particularly of the ethmoid sinus, which is only separated from the orbit by the thin lamina papyracea.3 While both preseptal cellulitis and orbital cellulitis can cause eyelid swelling and erythema, preseptal cellulitis is typically a mild condition. Orbital cellulitis, however, is a serious medical emergency that requires prompt diagnosis and treatment to avoid loss of vision and intracranial complications, such as venous thrombosis and empyema.3
Imaging Studies
Although the clinical features of orbital cellulitis (eg, proptosis, ophthalmoplegia, pain with ocular movement) can sometimes distinguish it from preseptal cellulitis, imaging studies are helpful to confirm the diagnosis.4 As previously noted, prompt recognition, diagnosis, and treatment of orbital cellulitis are essential to avoid serious complications.
Computed tomography has a high specificity and sensitivity in detecting the extension of infection into the orbit and associated complications such as subperiosteal or intracranial abscess. For patients in whom intravenous (IV) contrast is contraindicated or who wish to avoid ionizing radiation, magnetic resonance imaging is a useful alternate modality, and diffusion-weighted imaging is particularly sensitive in diagnosing abscess.5
Treatment
1. Baring DE, Hilmi OJ. An evidence based review of periorbital cellulitis. Clin Otolaryngol. 2011;36(1):57-64. doi:10.1111/j.1749-4486.2011.02258.x.
2. Kikkawa DO, Heinz GW, Martin RT, Nunery WN, Eiseman AS. Orbital cellulitis and abscess secondary to dacryocystitis. Arch Ophthalmol. 2002;120(8):1096-1099.
3. Mathew AV, Craig E, Al-Mahmoud R, et al. Paediatric post-septal and pre-septal cellulitis: 10 years’ experience at a tertiary-level children’s hospital. Br J Radiol. 2014;87(1033):20130503. doi:10.1259/bjr.20130503.
4. Rudloe TF, Harper MB, Prabhu SP, Rahbar R, Vanderveen D, Kimia AA. Acute periorbital infections: who needs emergent imaging? Pediatrics. 2010;125(4):e719-e726. doi:10.1542/peds.2009-1709.
5. Sepahdari AR, Aakalu VK, Kapur R, et al. MRI of orbital cellulitis and orbital abscess: the role of diffusion-weighted imaging. AJR Am J Roentgenol. 2009;193(3):W244-W250. doi:10.2214/AJR.08.1838.
6. Ho CF, Huang YC, Wang CJ, Chiu CH, Lin TY. Clinical analysis of computed tomography-staged orbital cellulitis in children. J Microbiol Immunol Infect. 2007;40(6):518-524.
1. Baring DE, Hilmi OJ. An evidence based review of periorbital cellulitis. Clin Otolaryngol. 2011;36(1):57-64. doi:10.1111/j.1749-4486.2011.02258.x.
2. Kikkawa DO, Heinz GW, Martin RT, Nunery WN, Eiseman AS. Orbital cellulitis and abscess secondary to dacryocystitis. Arch Ophthalmol. 2002;120(8):1096-1099.
3. Mathew AV, Craig E, Al-Mahmoud R, et al. Paediatric post-septal and pre-septal cellulitis: 10 years’ experience at a tertiary-level children’s hospital. Br J Radiol. 2014;87(1033):20130503. doi:10.1259/bjr.20130503.
4. Rudloe TF, Harper MB, Prabhu SP, Rahbar R, Vanderveen D, Kimia AA. Acute periorbital infections: who needs emergent imaging? Pediatrics. 2010;125(4):e719-e726. doi:10.1542/peds.2009-1709.
5. Sepahdari AR, Aakalu VK, Kapur R, et al. MRI of orbital cellulitis and orbital abscess: the role of diffusion-weighted imaging. AJR Am J Roentgenol. 2009;193(3):W244-W250. doi:10.2214/AJR.08.1838.
6. Ho CF, Huang YC, Wang CJ, Chiu CH, Lin TY. Clinical analysis of computed tomography-staged orbital cellulitis in children. J Microbiol Immunol Infect. 2007;40(6):518-524.
Malpractice Counsel: A Pain in the…Scrotum
Case
A 52-year-old man presented to the ED for evaluation of right scrotal pain and swelling. The patient stated that the pain started several hours prior to presentation and had gradually worsened. He denied any trauma or inciting event to the affected area; he further denied abdominal pain, nausea, vomiting, dysuria, polyuria, or fever. The patient’s remote medical history was significant for type 2 diabetes mellitus (DM), which he managed through dietary modification-only as he had refused pharmacological therapy. The patient admitted to smoking one half-pack of cigarettes per week, but denied alcohol or illicit drug use.
At presentation, the patient’s vital signs were all within normal range. The physical examination was remarkable only for right testicular tenderness and mild scrotal swelling, and there were no hernias or lymphadenopathy present.
The emergency physician (EP) ordered a urinalysis and color-flow Doppler ultrasound study of both testes, which the radiologist interpreted as an enlarged right epididymis with hyperemia; the left testicle was normal. The urinalysis was normal.
The patient was diagnosed with epididymitis and discharged home with a prescription for oral levofloxacin 500 mg daily for 10 days. He also was instructed to take ibuprofen for pain, apply ice to the affected area, keep the scrotal area elevated, and follow-up with a urologist in 1 week.
Approximately 8 hours after discharge, the patient returned to the same ED with complaints of increasing right testicular pain and swelling. The history and physical examination at this visit were essentially unchanged from his initial presentation. No laboratory evaluation, imaging studies, or other tests were ordered at the second visit.
The patient was discharged home with a prescription for a narcotic analgesic, which he was instructed to take in addition to the ibuprofen; he was also instructed to follow-up with a urologist within the next 2 to 3 days, instead of in 1 week.
The patient returned the following morning to the same ED with complaints of increased swelling and pain of the right testicle. In addition to the worsening testicular pain and swelling, he also had right inguinal pain, nausea, vomiting, and fever. Vital signs at this third presentation were: blood pressure (BP), 124/64 mm Hg; heart rate (HR), 110 beats/min; respiratory rate, 20 breaths/min; and temperature, 99.8o F. Oxygen saturation was 98% on room air.
The patient was tachycardic on heart examination, but with regular rhythm and no murmurs, rubs, or gallops. The lung and abdominal examinations were normal. The genital examination revealed marked right scrotal swelling and tenderness, as well as tender right inguinal lymphadenopathy.
The EP ordered an intravenous (IV) bolus of 1 L normal saline and laboratory studies, which included lactic acid, blood cultures, urinalysis, and urine culture and sensitivity. The EP was concerned for a scrotal abscess and ordered a testicular Doppler color-flow ultrasound study. The laboratory studies revealed an elevated white blood count of 16.5 K/uL, elevated blood glucose of 364 mg/dL, and elevated lactate of 2.8 mg/dL. As demonstrated on the ultrasound study performed at the patient’s first presentation, the ultrasound again showed an enlarged right epididymis, but without orchitis or abscess. The scrotal wall had significant thickening, consistent with cellulitis. The EP ordered broad spectrum IV antibiotics and admitted the patient to the hospitalist with a consult request for urology services.
The patient continued to receive IV fluids and antibiotics throughout the evening. In the morning, he was seen by the same hospitalist/admitting physician from the previous evening. Upon physical examination, the hospitalist noted tenderness, swelling, and erythema in the patient’s perineal area. The patient’s BP had dropped to 100/60 mm Hg, and his HR had increased to 115 beats/min despite receiving nearly 2 L of normal saline IV throughout the previous evening and night.
The urologist examined the patient soon after the consult request and diagnosed him with Fournier’s gangrene. He started the patient on aggressive IV fluid resuscitation, after which the patient was immediately taken to the operating room for extensive surgical debridement and scrotectomy. The patient’s postoperative course was complicated by acute kidney injury, respiratory failure requiring ventilator support, and sepsis. After a lengthy hospital stay, the patient was discharged home, but required a scrotal skin graft, and experienced erectile dysfunction and depression.
The patient sued all of the EPs involved in his care, the hospital, the hospitalist/admitting physician, and the urologist for negligence. The plaintiff’s attorney argued that since the patient progressively deteriorated over the 24 to 36 hours during his three presentations to the ED, urology services should have been consulted earlier, and that the urologist should have seen the patient immediately at the time of hospital admission.
The attorneys for the defendants claimed the patient denied dysuria, penile lesions, or urethral discharge and that the history, physical examination, and testicular ultrasound were all consistent with the diagnosis of epididymitis. For this reason, they argued, there was no indication for an emergent consultation with urology services. The jury returned a defense verdict.
Discussion
It is easy for a busy EP to have a differential diagnosis of only two disorders when evaluating a patient for unilateral testicular pain and swelling—in this case, testicular torsion and epididymitis. While these are the most common causes of testicular pain and swelling, this case emphasizes the need to also consider Fournier’s gangrene in the differential. A thorough history and physical examination, coupled with appropriate testing, will usually identify the correct diagnosis. While the differential diagnosis is broader than just these three disease processes (see the Box), we will review the evaluation and management of the three most serious: epididymitis, testicular torsion, and Fournier’s gangrene.
Noninfectious and Bacterial Epididymitis
Epididymitis is the most common cause of acute scrotal pain among US adults, accounting for approximately 600,000 cases each year.1 Infectious epididymitis is typically classified as acute (symptom duration of <6 weeks) or chronic (symptom duration of ≥6 weeks).2
Cases of noninfectious epididymitis are typically due to a chronic condition, such as autoimmune disease, cancer, or vasculitis. Although not as common, noninfectious epididymitis can also occur due to testicular trauma or amiodarone therapy.3,4
Patients with acute bacterial epididymitis typically present with scrotal pain and swelling ranging from mild to marked. These patients may also exhibit fever and chills, along with dysuria, frequency, and urgency, if associated with a urinary tract infection.2 The chronic presentation is more common though, and usually not associated with voiding issues.
Chronic epididymis is frequently seen in postpubertal boys and men following sexual activity, heavy physical exertion, and bicycle/motorcycle riding.2 On physical examination, palpation reveals induration and swelling of the involved epididymis with exquisite tenderness.2 Testicular swelling and pain, along with scrotal wall erythema, may be present in more advanced cases.2 The cremasteric reflex should be intact (ie, scratching the medial proximal thigh will cause ipsilateral testicle retraction). Similarly, the lie of both testicles while the patient is standing should be equal and symmetrical—ie, both testicles descended equally. However, in the presence of moderate-to-severe scrotal swelling, both of these physical findings may be impossible to confirm.
A urinalysis and urine culture should be ordered if there is any suspicion of epididymitis; pyuria will be present in approximately 50% of cases. However, since pyuria is neither sensitive nor specific for epididymitis, in most cases, a testicular ultrasound with Doppler flow is required to exclude testicular torsion. In cases of epididymitis, ultrasound usually demonstrates increased flow on the affected side, whereas in testicular torsion, there is decreased or absent blood flow.
The treatment for epididymitis involves antibiotics and symptomatic care. If epididymitis from chlamydia and/or gonorrhea is the suspected cause, or if the patient is younger than age 35 years, he should be given ceftriaxone 250 mg intramuscularly plus oral doxycycline 100 mg twice a day for 10 days. Patients who practice insertive anal sex should be treated with ceftriaxone, plus either oral ofloxacin 300 mg twice a day or oral levofloxacin 500 mg daily for 10 days.
In cases in which enteric organisms are suspected, the patient is older than age 35 years, or if patient status is posturinary tract instrumentation or vasectomy, he should be treated with either oral ofloxacin 300 mg twice a day or oral levofloxacin 500 mg daily for 10 days.2
For symptomatic relief, scrotal elevation, ice application, and nonsteroidal anti-inflammatory drugs are recommended.
Patients with epididymitis, regardless of etiology, should be instructed to follow-up with a urologist within 1 week. If the patient appears ill, septic, or in significant pain, admission to the hospital with IV antibiotics, IV fluids, and an urgent consult with urology services is required.
Testicular Torsion
Testicular torsion is a time-sensitive issue, requiring early diagnosis and rapid treatment to preserve the patient’s fertility. Most clinicians recommend detorsion within 6 hours of torsion onset because salvage rates are excellent when performed within this timeframe; after 12 hours, the testis will likely suffer irreversible damage due to ischemia.5,6
Testicular torsion can occur at any age, but is most commonly seen in a bimodal distribution—ie, neonates and postpubertal boys. The prevalence of testicular torsion in adult patients hospitalized with acute scrotal pain is approximately 25% to 50%.2
Patients with testicular torsion usually describe a sudden onset of severe, acute pain. The pain frequently occurs a few hours after vigorous physical activity or minor testicular trauma.2 Occasionally, the patient may complain of lower quadrant abdominal pain rather than testicular or scrotal pain. Nausea with vomiting can also be present.
On physical examination, significant testicular swelling is usually present. Examining the patient in the standing position will often reveal an asymmetrical, high-riding testis with a transverse lie on the affected side. The cremasteric reflex is usually absent in patients with testicular torsion.
Because of the significant overlap in history and physical examination findings for epididymitis and testicular torsion, a testicular ultrasound with color Doppler should be ordered. Multiple studies have confirmed the high sensitivity and specificity of ultrasound in the diagnosis of testicular torsion.
The treatment for suspected or confirmed testicular torsion is immediate surgical exploration with intraoperative detorsion and fixation of the testes. The EP can attempt manual detorsion (ie, performed in a medial to lateral motion, similar to opening a book). However, this should not delay the EP from consulting with urology services.
Pediatric patients with testicular torsion usually have a more favorable outcome than do adults. In one retrospective study, patients younger than age 21 years had a 70% testicular salvage rate compared to only 41% of patients aged 21 years and older.7 Regardless of age, better outcomes are associated with shorter periods of torsion.
Fournier’s Gangrene
Fournier’s gangrene is a polymicrobial necrotizing fasciitis of the perineum and scrotum that typically develops initially as a benign infection or abscess but quickly spreads. Risk factors for Fournier’s gangrene include DM, alcohol abuse, and any immunocompromised state (eg, HIV, cancer).
If the patient presents early in onset, there may be only mild tenderness, erythema, or swelling of the affected area; however, this infection progresses rapidly. Later findings include marked tenderness, swelling, crepitus, blisters, and ecchymoses. Patients with Fournier’s gangrene also develop systemic signs of infection, including fever, tachycardia, tachypnea, and hypotension. The key to diagnosis is careful examination of the perineal and scrotal area in any patient presenting with acute scrotal pain.
In the majority of cases, the diagnosis of Fournier’s gangrene is made clinically. Once the diagnosis is made, patients require immediate and aggressive IV fluid resuscitation, broad-spectrum IV antibiotics (typically vancomycin and piperacillin/tazobactam), and emergent evaluation by a urologist. It is essential that these patients undergo early and aggressive surgical exploration and debridement of necrotic tissue.2 Antibiotic therapy alone is associated with a 100% mortality rate, emphasizing the need for urgent surgery.2 Even with optimal medical and surgical management, the mortality rate remains significant.
Summary
This case emphasizes several important teaching points. The EP should be mindful of the patient who keeps returning to the ED with the same complaint—despite “appropriate” treatment—as the initial diagnosis may not be the correct one. Such returning patients require greater, not less, scrutiny. As with any patient, the EP should always take a complete history and perform a thorough physical examination at each presentation—as one would with a de novo patient. Finally, the EP should consider Fournier’s gangrene in addition to testicular torsion and epididymitis in the differential diagnosis for acute scrotal pain.
1. Trojian TH, Lishnak TS, Heiman D. Epididymitis and orchitis: an overview. Am Fam Physician. 2009;79(7):583-587.
2. Eyre RC. Evaluation of acute scrotal pain in adults. UpToDate Web site. https://www.uptodate.com/contents/evaluation-of-acute-scrotal-pain-in-adults. Updated July 31, 2017. Accessed September 7, 2017.
3. Shen Y, Liu H, Cheng J, Bu P. Amiodarone-induced epididymitis: a pathologically confirmed case report and review of the literature. Cardiology. 2014;128(4):349-351. doi:10.1159/000361038.
4. Tracy CR, Steers WD, Costabile R. Diagnosis and management of epididymitis. Urol Clin North Am. 2008;35(1):101-108. doi:10.1016/j.ucl.2007.09.013.
5. Wampler SM, Llanes M. Common scrotal and testicular problems. Prim Care. 2010;37(3):613-626. doi:10.1016/j.pop.2010.04.009.
6. Dunne PJ, O’Loughlin BS. Testicular torsion: time is the enemy. Aust NZ J Surg. 2000;70(6):441-442.
7. Cummings JM, Boullier JA, Sekhon D, Bose K. Adult testicular torsion. J Urol. 2002;167(5):2109-2110.
Case
A 52-year-old man presented to the ED for evaluation of right scrotal pain and swelling. The patient stated that the pain started several hours prior to presentation and had gradually worsened. He denied any trauma or inciting event to the affected area; he further denied abdominal pain, nausea, vomiting, dysuria, polyuria, or fever. The patient’s remote medical history was significant for type 2 diabetes mellitus (DM), which he managed through dietary modification-only as he had refused pharmacological therapy. The patient admitted to smoking one half-pack of cigarettes per week, but denied alcohol or illicit drug use.
At presentation, the patient’s vital signs were all within normal range. The physical examination was remarkable only for right testicular tenderness and mild scrotal swelling, and there were no hernias or lymphadenopathy present.
The emergency physician (EP) ordered a urinalysis and color-flow Doppler ultrasound study of both testes, which the radiologist interpreted as an enlarged right epididymis with hyperemia; the left testicle was normal. The urinalysis was normal.
The patient was diagnosed with epididymitis and discharged home with a prescription for oral levofloxacin 500 mg daily for 10 days. He also was instructed to take ibuprofen for pain, apply ice to the affected area, keep the scrotal area elevated, and follow-up with a urologist in 1 week.
Approximately 8 hours after discharge, the patient returned to the same ED with complaints of increasing right testicular pain and swelling. The history and physical examination at this visit were essentially unchanged from his initial presentation. No laboratory evaluation, imaging studies, or other tests were ordered at the second visit.
The patient was discharged home with a prescription for a narcotic analgesic, which he was instructed to take in addition to the ibuprofen; he was also instructed to follow-up with a urologist within the next 2 to 3 days, instead of in 1 week.
The patient returned the following morning to the same ED with complaints of increased swelling and pain of the right testicle. In addition to the worsening testicular pain and swelling, he also had right inguinal pain, nausea, vomiting, and fever. Vital signs at this third presentation were: blood pressure (BP), 124/64 mm Hg; heart rate (HR), 110 beats/min; respiratory rate, 20 breaths/min; and temperature, 99.8o F. Oxygen saturation was 98% on room air.
The patient was tachycardic on heart examination, but with regular rhythm and no murmurs, rubs, or gallops. The lung and abdominal examinations were normal. The genital examination revealed marked right scrotal swelling and tenderness, as well as tender right inguinal lymphadenopathy.
The EP ordered an intravenous (IV) bolus of 1 L normal saline and laboratory studies, which included lactic acid, blood cultures, urinalysis, and urine culture and sensitivity. The EP was concerned for a scrotal abscess and ordered a testicular Doppler color-flow ultrasound study. The laboratory studies revealed an elevated white blood count of 16.5 K/uL, elevated blood glucose of 364 mg/dL, and elevated lactate of 2.8 mg/dL. As demonstrated on the ultrasound study performed at the patient’s first presentation, the ultrasound again showed an enlarged right epididymis, but without orchitis or abscess. The scrotal wall had significant thickening, consistent with cellulitis. The EP ordered broad spectrum IV antibiotics and admitted the patient to the hospitalist with a consult request for urology services.
The patient continued to receive IV fluids and antibiotics throughout the evening. In the morning, he was seen by the same hospitalist/admitting physician from the previous evening. Upon physical examination, the hospitalist noted tenderness, swelling, and erythema in the patient’s perineal area. The patient’s BP had dropped to 100/60 mm Hg, and his HR had increased to 115 beats/min despite receiving nearly 2 L of normal saline IV throughout the previous evening and night.
The urologist examined the patient soon after the consult request and diagnosed him with Fournier’s gangrene. He started the patient on aggressive IV fluid resuscitation, after which the patient was immediately taken to the operating room for extensive surgical debridement and scrotectomy. The patient’s postoperative course was complicated by acute kidney injury, respiratory failure requiring ventilator support, and sepsis. After a lengthy hospital stay, the patient was discharged home, but required a scrotal skin graft, and experienced erectile dysfunction and depression.
The patient sued all of the EPs involved in his care, the hospital, the hospitalist/admitting physician, and the urologist for negligence. The plaintiff’s attorney argued that since the patient progressively deteriorated over the 24 to 36 hours during his three presentations to the ED, urology services should have been consulted earlier, and that the urologist should have seen the patient immediately at the time of hospital admission.
The attorneys for the defendants claimed the patient denied dysuria, penile lesions, or urethral discharge and that the history, physical examination, and testicular ultrasound were all consistent with the diagnosis of epididymitis. For this reason, they argued, there was no indication for an emergent consultation with urology services. The jury returned a defense verdict.
Discussion
It is easy for a busy EP to have a differential diagnosis of only two disorders when evaluating a patient for unilateral testicular pain and swelling—in this case, testicular torsion and epididymitis. While these are the most common causes of testicular pain and swelling, this case emphasizes the need to also consider Fournier’s gangrene in the differential. A thorough history and physical examination, coupled with appropriate testing, will usually identify the correct diagnosis. While the differential diagnosis is broader than just these three disease processes (see the Box), we will review the evaluation and management of the three most serious: epididymitis, testicular torsion, and Fournier’s gangrene.
Noninfectious and Bacterial Epididymitis
Epididymitis is the most common cause of acute scrotal pain among US adults, accounting for approximately 600,000 cases each year.1 Infectious epididymitis is typically classified as acute (symptom duration of <6 weeks) or chronic (symptom duration of ≥6 weeks).2
Cases of noninfectious epididymitis are typically due to a chronic condition, such as autoimmune disease, cancer, or vasculitis. Although not as common, noninfectious epididymitis can also occur due to testicular trauma or amiodarone therapy.3,4
Patients with acute bacterial epididymitis typically present with scrotal pain and swelling ranging from mild to marked. These patients may also exhibit fever and chills, along with dysuria, frequency, and urgency, if associated with a urinary tract infection.2 The chronic presentation is more common though, and usually not associated with voiding issues.
Chronic epididymis is frequently seen in postpubertal boys and men following sexual activity, heavy physical exertion, and bicycle/motorcycle riding.2 On physical examination, palpation reveals induration and swelling of the involved epididymis with exquisite tenderness.2 Testicular swelling and pain, along with scrotal wall erythema, may be present in more advanced cases.2 The cremasteric reflex should be intact (ie, scratching the medial proximal thigh will cause ipsilateral testicle retraction). Similarly, the lie of both testicles while the patient is standing should be equal and symmetrical—ie, both testicles descended equally. However, in the presence of moderate-to-severe scrotal swelling, both of these physical findings may be impossible to confirm.
A urinalysis and urine culture should be ordered if there is any suspicion of epididymitis; pyuria will be present in approximately 50% of cases. However, since pyuria is neither sensitive nor specific for epididymitis, in most cases, a testicular ultrasound with Doppler flow is required to exclude testicular torsion. In cases of epididymitis, ultrasound usually demonstrates increased flow on the affected side, whereas in testicular torsion, there is decreased or absent blood flow.
The treatment for epididymitis involves antibiotics and symptomatic care. If epididymitis from chlamydia and/or gonorrhea is the suspected cause, or if the patient is younger than age 35 years, he should be given ceftriaxone 250 mg intramuscularly plus oral doxycycline 100 mg twice a day for 10 days. Patients who practice insertive anal sex should be treated with ceftriaxone, plus either oral ofloxacin 300 mg twice a day or oral levofloxacin 500 mg daily for 10 days.
In cases in which enteric organisms are suspected, the patient is older than age 35 years, or if patient status is posturinary tract instrumentation or vasectomy, he should be treated with either oral ofloxacin 300 mg twice a day or oral levofloxacin 500 mg daily for 10 days.2
For symptomatic relief, scrotal elevation, ice application, and nonsteroidal anti-inflammatory drugs are recommended.
Patients with epididymitis, regardless of etiology, should be instructed to follow-up with a urologist within 1 week. If the patient appears ill, septic, or in significant pain, admission to the hospital with IV antibiotics, IV fluids, and an urgent consult with urology services is required.
Testicular Torsion
Testicular torsion is a time-sensitive issue, requiring early diagnosis and rapid treatment to preserve the patient’s fertility. Most clinicians recommend detorsion within 6 hours of torsion onset because salvage rates are excellent when performed within this timeframe; after 12 hours, the testis will likely suffer irreversible damage due to ischemia.5,6
Testicular torsion can occur at any age, but is most commonly seen in a bimodal distribution—ie, neonates and postpubertal boys. The prevalence of testicular torsion in adult patients hospitalized with acute scrotal pain is approximately 25% to 50%.2
Patients with testicular torsion usually describe a sudden onset of severe, acute pain. The pain frequently occurs a few hours after vigorous physical activity or minor testicular trauma.2 Occasionally, the patient may complain of lower quadrant abdominal pain rather than testicular or scrotal pain. Nausea with vomiting can also be present.
On physical examination, significant testicular swelling is usually present. Examining the patient in the standing position will often reveal an asymmetrical, high-riding testis with a transverse lie on the affected side. The cremasteric reflex is usually absent in patients with testicular torsion.
Because of the significant overlap in history and physical examination findings for epididymitis and testicular torsion, a testicular ultrasound with color Doppler should be ordered. Multiple studies have confirmed the high sensitivity and specificity of ultrasound in the diagnosis of testicular torsion.
The treatment for suspected or confirmed testicular torsion is immediate surgical exploration with intraoperative detorsion and fixation of the testes. The EP can attempt manual detorsion (ie, performed in a medial to lateral motion, similar to opening a book). However, this should not delay the EP from consulting with urology services.
Pediatric patients with testicular torsion usually have a more favorable outcome than do adults. In one retrospective study, patients younger than age 21 years had a 70% testicular salvage rate compared to only 41% of patients aged 21 years and older.7 Regardless of age, better outcomes are associated with shorter periods of torsion.
Fournier’s Gangrene
Fournier’s gangrene is a polymicrobial necrotizing fasciitis of the perineum and scrotum that typically develops initially as a benign infection or abscess but quickly spreads. Risk factors for Fournier’s gangrene include DM, alcohol abuse, and any immunocompromised state (eg, HIV, cancer).
If the patient presents early in onset, there may be only mild tenderness, erythema, or swelling of the affected area; however, this infection progresses rapidly. Later findings include marked tenderness, swelling, crepitus, blisters, and ecchymoses. Patients with Fournier’s gangrene also develop systemic signs of infection, including fever, tachycardia, tachypnea, and hypotension. The key to diagnosis is careful examination of the perineal and scrotal area in any patient presenting with acute scrotal pain.
In the majority of cases, the diagnosis of Fournier’s gangrene is made clinically. Once the diagnosis is made, patients require immediate and aggressive IV fluid resuscitation, broad-spectrum IV antibiotics (typically vancomycin and piperacillin/tazobactam), and emergent evaluation by a urologist. It is essential that these patients undergo early and aggressive surgical exploration and debridement of necrotic tissue.2 Antibiotic therapy alone is associated with a 100% mortality rate, emphasizing the need for urgent surgery.2 Even with optimal medical and surgical management, the mortality rate remains significant.
Summary
This case emphasizes several important teaching points. The EP should be mindful of the patient who keeps returning to the ED with the same complaint—despite “appropriate” treatment—as the initial diagnosis may not be the correct one. Such returning patients require greater, not less, scrutiny. As with any patient, the EP should always take a complete history and perform a thorough physical examination at each presentation—as one would with a de novo patient. Finally, the EP should consider Fournier’s gangrene in addition to testicular torsion and epididymitis in the differential diagnosis for acute scrotal pain.
Case
A 52-year-old man presented to the ED for evaluation of right scrotal pain and swelling. The patient stated that the pain started several hours prior to presentation and had gradually worsened. He denied any trauma or inciting event to the affected area; he further denied abdominal pain, nausea, vomiting, dysuria, polyuria, or fever. The patient’s remote medical history was significant for type 2 diabetes mellitus (DM), which he managed through dietary modification-only as he had refused pharmacological therapy. The patient admitted to smoking one half-pack of cigarettes per week, but denied alcohol or illicit drug use.
At presentation, the patient’s vital signs were all within normal range. The physical examination was remarkable only for right testicular tenderness and mild scrotal swelling, and there were no hernias or lymphadenopathy present.
The emergency physician (EP) ordered a urinalysis and color-flow Doppler ultrasound study of both testes, which the radiologist interpreted as an enlarged right epididymis with hyperemia; the left testicle was normal. The urinalysis was normal.
The patient was diagnosed with epididymitis and discharged home with a prescription for oral levofloxacin 500 mg daily for 10 days. He also was instructed to take ibuprofen for pain, apply ice to the affected area, keep the scrotal area elevated, and follow-up with a urologist in 1 week.
Approximately 8 hours after discharge, the patient returned to the same ED with complaints of increasing right testicular pain and swelling. The history and physical examination at this visit were essentially unchanged from his initial presentation. No laboratory evaluation, imaging studies, or other tests were ordered at the second visit.
The patient was discharged home with a prescription for a narcotic analgesic, which he was instructed to take in addition to the ibuprofen; he was also instructed to follow-up with a urologist within the next 2 to 3 days, instead of in 1 week.
The patient returned the following morning to the same ED with complaints of increased swelling and pain of the right testicle. In addition to the worsening testicular pain and swelling, he also had right inguinal pain, nausea, vomiting, and fever. Vital signs at this third presentation were: blood pressure (BP), 124/64 mm Hg; heart rate (HR), 110 beats/min; respiratory rate, 20 breaths/min; and temperature, 99.8o F. Oxygen saturation was 98% on room air.
The patient was tachycardic on heart examination, but with regular rhythm and no murmurs, rubs, or gallops. The lung and abdominal examinations were normal. The genital examination revealed marked right scrotal swelling and tenderness, as well as tender right inguinal lymphadenopathy.
The EP ordered an intravenous (IV) bolus of 1 L normal saline and laboratory studies, which included lactic acid, blood cultures, urinalysis, and urine culture and sensitivity. The EP was concerned for a scrotal abscess and ordered a testicular Doppler color-flow ultrasound study. The laboratory studies revealed an elevated white blood count of 16.5 K/uL, elevated blood glucose of 364 mg/dL, and elevated lactate of 2.8 mg/dL. As demonstrated on the ultrasound study performed at the patient’s first presentation, the ultrasound again showed an enlarged right epididymis, but without orchitis or abscess. The scrotal wall had significant thickening, consistent with cellulitis. The EP ordered broad spectrum IV antibiotics and admitted the patient to the hospitalist with a consult request for urology services.
The patient continued to receive IV fluids and antibiotics throughout the evening. In the morning, he was seen by the same hospitalist/admitting physician from the previous evening. Upon physical examination, the hospitalist noted tenderness, swelling, and erythema in the patient’s perineal area. The patient’s BP had dropped to 100/60 mm Hg, and his HR had increased to 115 beats/min despite receiving nearly 2 L of normal saline IV throughout the previous evening and night.
The urologist examined the patient soon after the consult request and diagnosed him with Fournier’s gangrene. He started the patient on aggressive IV fluid resuscitation, after which the patient was immediately taken to the operating room for extensive surgical debridement and scrotectomy. The patient’s postoperative course was complicated by acute kidney injury, respiratory failure requiring ventilator support, and sepsis. After a lengthy hospital stay, the patient was discharged home, but required a scrotal skin graft, and experienced erectile dysfunction and depression.
The patient sued all of the EPs involved in his care, the hospital, the hospitalist/admitting physician, and the urologist for negligence. The plaintiff’s attorney argued that since the patient progressively deteriorated over the 24 to 36 hours during his three presentations to the ED, urology services should have been consulted earlier, and that the urologist should have seen the patient immediately at the time of hospital admission.
The attorneys for the defendants claimed the patient denied dysuria, penile lesions, or urethral discharge and that the history, physical examination, and testicular ultrasound were all consistent with the diagnosis of epididymitis. For this reason, they argued, there was no indication for an emergent consultation with urology services. The jury returned a defense verdict.
Discussion
It is easy for a busy EP to have a differential diagnosis of only two disorders when evaluating a patient for unilateral testicular pain and swelling—in this case, testicular torsion and epididymitis. While these are the most common causes of testicular pain and swelling, this case emphasizes the need to also consider Fournier’s gangrene in the differential. A thorough history and physical examination, coupled with appropriate testing, will usually identify the correct diagnosis. While the differential diagnosis is broader than just these three disease processes (see the Box), we will review the evaluation and management of the three most serious: epididymitis, testicular torsion, and Fournier’s gangrene.
Noninfectious and Bacterial Epididymitis
Epididymitis is the most common cause of acute scrotal pain among US adults, accounting for approximately 600,000 cases each year.1 Infectious epididymitis is typically classified as acute (symptom duration of <6 weeks) or chronic (symptom duration of ≥6 weeks).2
Cases of noninfectious epididymitis are typically due to a chronic condition, such as autoimmune disease, cancer, or vasculitis. Although not as common, noninfectious epididymitis can also occur due to testicular trauma or amiodarone therapy.3,4
Patients with acute bacterial epididymitis typically present with scrotal pain and swelling ranging from mild to marked. These patients may also exhibit fever and chills, along with dysuria, frequency, and urgency, if associated with a urinary tract infection.2 The chronic presentation is more common though, and usually not associated with voiding issues.
Chronic epididymis is frequently seen in postpubertal boys and men following sexual activity, heavy physical exertion, and bicycle/motorcycle riding.2 On physical examination, palpation reveals induration and swelling of the involved epididymis with exquisite tenderness.2 Testicular swelling and pain, along with scrotal wall erythema, may be present in more advanced cases.2 The cremasteric reflex should be intact (ie, scratching the medial proximal thigh will cause ipsilateral testicle retraction). Similarly, the lie of both testicles while the patient is standing should be equal and symmetrical—ie, both testicles descended equally. However, in the presence of moderate-to-severe scrotal swelling, both of these physical findings may be impossible to confirm.
A urinalysis and urine culture should be ordered if there is any suspicion of epididymitis; pyuria will be present in approximately 50% of cases. However, since pyuria is neither sensitive nor specific for epididymitis, in most cases, a testicular ultrasound with Doppler flow is required to exclude testicular torsion. In cases of epididymitis, ultrasound usually demonstrates increased flow on the affected side, whereas in testicular torsion, there is decreased or absent blood flow.
The treatment for epididymitis involves antibiotics and symptomatic care. If epididymitis from chlamydia and/or gonorrhea is the suspected cause, or if the patient is younger than age 35 years, he should be given ceftriaxone 250 mg intramuscularly plus oral doxycycline 100 mg twice a day for 10 days. Patients who practice insertive anal sex should be treated with ceftriaxone, plus either oral ofloxacin 300 mg twice a day or oral levofloxacin 500 mg daily for 10 days.
In cases in which enteric organisms are suspected, the patient is older than age 35 years, or if patient status is posturinary tract instrumentation or vasectomy, he should be treated with either oral ofloxacin 300 mg twice a day or oral levofloxacin 500 mg daily for 10 days.2
For symptomatic relief, scrotal elevation, ice application, and nonsteroidal anti-inflammatory drugs are recommended.
Patients with epididymitis, regardless of etiology, should be instructed to follow-up with a urologist within 1 week. If the patient appears ill, septic, or in significant pain, admission to the hospital with IV antibiotics, IV fluids, and an urgent consult with urology services is required.
Testicular Torsion
Testicular torsion is a time-sensitive issue, requiring early diagnosis and rapid treatment to preserve the patient’s fertility. Most clinicians recommend detorsion within 6 hours of torsion onset because salvage rates are excellent when performed within this timeframe; after 12 hours, the testis will likely suffer irreversible damage due to ischemia.5,6
Testicular torsion can occur at any age, but is most commonly seen in a bimodal distribution—ie, neonates and postpubertal boys. The prevalence of testicular torsion in adult patients hospitalized with acute scrotal pain is approximately 25% to 50%.2
Patients with testicular torsion usually describe a sudden onset of severe, acute pain. The pain frequently occurs a few hours after vigorous physical activity or minor testicular trauma.2 Occasionally, the patient may complain of lower quadrant abdominal pain rather than testicular or scrotal pain. Nausea with vomiting can also be present.
On physical examination, significant testicular swelling is usually present. Examining the patient in the standing position will often reveal an asymmetrical, high-riding testis with a transverse lie on the affected side. The cremasteric reflex is usually absent in patients with testicular torsion.
Because of the significant overlap in history and physical examination findings for epididymitis and testicular torsion, a testicular ultrasound with color Doppler should be ordered. Multiple studies have confirmed the high sensitivity and specificity of ultrasound in the diagnosis of testicular torsion.
The treatment for suspected or confirmed testicular torsion is immediate surgical exploration with intraoperative detorsion and fixation of the testes. The EP can attempt manual detorsion (ie, performed in a medial to lateral motion, similar to opening a book). However, this should not delay the EP from consulting with urology services.
Pediatric patients with testicular torsion usually have a more favorable outcome than do adults. In one retrospective study, patients younger than age 21 years had a 70% testicular salvage rate compared to only 41% of patients aged 21 years and older.7 Regardless of age, better outcomes are associated with shorter periods of torsion.
Fournier’s Gangrene
Fournier’s gangrene is a polymicrobial necrotizing fasciitis of the perineum and scrotum that typically develops initially as a benign infection or abscess but quickly spreads. Risk factors for Fournier’s gangrene include DM, alcohol abuse, and any immunocompromised state (eg, HIV, cancer).
If the patient presents early in onset, there may be only mild tenderness, erythema, or swelling of the affected area; however, this infection progresses rapidly. Later findings include marked tenderness, swelling, crepitus, blisters, and ecchymoses. Patients with Fournier’s gangrene also develop systemic signs of infection, including fever, tachycardia, tachypnea, and hypotension. The key to diagnosis is careful examination of the perineal and scrotal area in any patient presenting with acute scrotal pain.
In the majority of cases, the diagnosis of Fournier’s gangrene is made clinically. Once the diagnosis is made, patients require immediate and aggressive IV fluid resuscitation, broad-spectrum IV antibiotics (typically vancomycin and piperacillin/tazobactam), and emergent evaluation by a urologist. It is essential that these patients undergo early and aggressive surgical exploration and debridement of necrotic tissue.2 Antibiotic therapy alone is associated with a 100% mortality rate, emphasizing the need for urgent surgery.2 Even with optimal medical and surgical management, the mortality rate remains significant.
Summary
This case emphasizes several important teaching points. The EP should be mindful of the patient who keeps returning to the ED with the same complaint—despite “appropriate” treatment—as the initial diagnosis may not be the correct one. Such returning patients require greater, not less, scrutiny. As with any patient, the EP should always take a complete history and perform a thorough physical examination at each presentation—as one would with a de novo patient. Finally, the EP should consider Fournier’s gangrene in addition to testicular torsion and epididymitis in the differential diagnosis for acute scrotal pain.
1. Trojian TH, Lishnak TS, Heiman D. Epididymitis and orchitis: an overview. Am Fam Physician. 2009;79(7):583-587.
2. Eyre RC. Evaluation of acute scrotal pain in adults. UpToDate Web site. https://www.uptodate.com/contents/evaluation-of-acute-scrotal-pain-in-adults. Updated July 31, 2017. Accessed September 7, 2017.
3. Shen Y, Liu H, Cheng J, Bu P. Amiodarone-induced epididymitis: a pathologically confirmed case report and review of the literature. Cardiology. 2014;128(4):349-351. doi:10.1159/000361038.
4. Tracy CR, Steers WD, Costabile R. Diagnosis and management of epididymitis. Urol Clin North Am. 2008;35(1):101-108. doi:10.1016/j.ucl.2007.09.013.
5. Wampler SM, Llanes M. Common scrotal and testicular problems. Prim Care. 2010;37(3):613-626. doi:10.1016/j.pop.2010.04.009.
6. Dunne PJ, O’Loughlin BS. Testicular torsion: time is the enemy. Aust NZ J Surg. 2000;70(6):441-442.
7. Cummings JM, Boullier JA, Sekhon D, Bose K. Adult testicular torsion. J Urol. 2002;167(5):2109-2110.
1. Trojian TH, Lishnak TS, Heiman D. Epididymitis and orchitis: an overview. Am Fam Physician. 2009;79(7):583-587.
2. Eyre RC. Evaluation of acute scrotal pain in adults. UpToDate Web site. https://www.uptodate.com/contents/evaluation-of-acute-scrotal-pain-in-adults. Updated July 31, 2017. Accessed September 7, 2017.
3. Shen Y, Liu H, Cheng J, Bu P. Amiodarone-induced epididymitis: a pathologically confirmed case report and review of the literature. Cardiology. 2014;128(4):349-351. doi:10.1159/000361038.
4. Tracy CR, Steers WD, Costabile R. Diagnosis and management of epididymitis. Urol Clin North Am. 2008;35(1):101-108. doi:10.1016/j.ucl.2007.09.013.
5. Wampler SM, Llanes M. Common scrotal and testicular problems. Prim Care. 2010;37(3):613-626. doi:10.1016/j.pop.2010.04.009.
6. Dunne PJ, O’Loughlin BS. Testicular torsion: time is the enemy. Aust NZ J Surg. 2000;70(6):441-442.
7. Cummings JM, Boullier JA, Sekhon D, Bose K. Adult testicular torsion. J Urol. 2002;167(5):2109-2110.