User login
Bilateral chylothorax in an AIDS patient with newly diagnosed Kaposi sarcoma
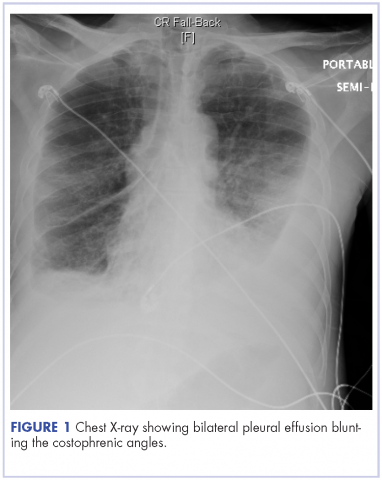
Kaposi sarcoma is an angioproliferative tumor that is associated with human herpes virus-8 (HHV-8). Mucocutaneous disease is the most common site for manifestation of AIDS-related Kaposi sarcoma, commonly affecting the lower extremities, oral mucosa, face, and genitalia. Pleural effusions can occur in 36%-60% of patients with Kaposi sarcoma, and it has been documented that chylothorax is a rare, but plausible presentation in patients with Kaposi sarcoma.1 We present here a case of bilateral chylothorax in a patient with AIDS-related Kaposi sarcoma.
Case presentation and summary
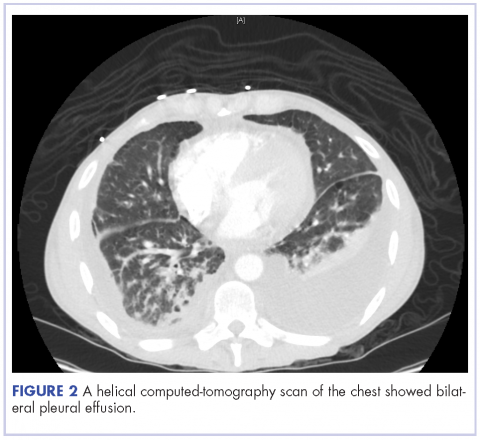
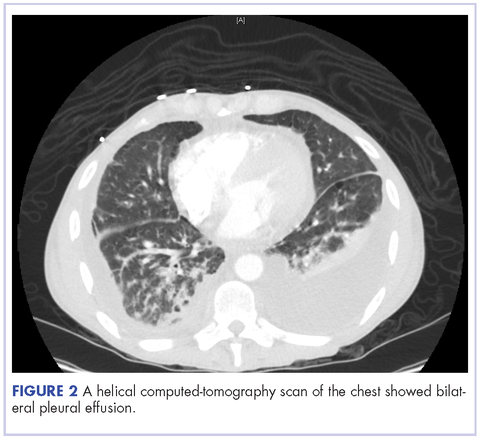
A 52-year-old MSM male with AIDS (CD4, <20 mm3; viral load, 58 copies/ml) presented to the emergency department with complaints of shortness of breath, productive cough, and diarrhea for 2 days prior to presentation. His medical history also included chronic obstructive pulmonary disease, coronary artery disease, and hyperlipidemia. The patient was not on HAART because of his history of noncompliance. The results of a chest X-ray and computed-tomography (CT) scan showed that the patient had bilateral pleural effusion and a spiculated 14-mm nodule in the left upper lobe.
The patient underwent ultrasound-guided placement of a 12-French left-sided chest catheter, and a milky white fluid was aspirated from the left pleural space. Laboratory analysis of the pleural fluid confirmed an exudate with an elevated triglyceride level of 120 mg/dL (chylous, >110 mg/dL) indicating chylothorax.
On close physical examination, the patient was found to have multiple irregular plaques on the back and lower extremities. As described by dermatology, there was a violaceous indurated plaque on the left axillae, violaceous indurated plaques with superficial scale grouped on the left midlateral back, and hyperpigmented lichenified plaques and papules on bilateral shins, with some with plate-like scale. Two punch biopsies were taken of the skin lesions, which confirmed Kaposi sarcoma, plaque stage from the lesion biopsied on the back, and patch stage from the lesion biopsied in the left axilla. Cytology of the pleural fluid was negative for malignant cells. On review by the radiologist of the CT scan of the chest, there was no indication of gross distention of the thoracic duct. Treatment options were offered to the patient, and the patient was considering options for chemotherapy and home hospice given his advanced disease state at the time of discharge.
Discussion
Chylothorax occurs with a thoracic duct obstruction, which results in leakage of lymphatic fluid into the pleural cavity. The two leading causes of chylothorax are trauma and malignancy, with lymphoma being the most common cause of chylothorax among those with malignancy.2 Chylothorax, however, is a rare but documented complication of Kaposi sarcoma. Marais and colleagues reported the case of a 3-year-old HIV-positive patient with newly diagnosed Kaposi sarcoma who was found to have tumor infiltration in the thoracic duct leading to bilateral chylothorax.3 Maradona and colleagues described a 40-year-old man with AIDS-related Kaposi sarcoma who was found to have pleural and pericardial Kaposi sarcoma with chylothorax.4 Priest and colleagues wrote about a 32-year-old patient with AIDS with biopsy-proven Kaposi sarcoma who required multiple therapeutic thoracenteses for rapidly recurrent left chylothorax effusions.5
There are two leading discussions as to the pathophysiology of chylothorax that is related to Kaposi sarcoma: chylothorax developing secondary to metastatic disease or the development of chylothorax secondary to primary Kaposi sarcoma arising from the pleural region.6 One case report examined pleural and lung biopsies in a 34-year-old patient with AIDS-related Kaposi sarcoma that showed immunohistochemical staining that was suggestive of early-stage Kaposi sarcoma of lymphatic endothelial origin. The authors were attempting to illustrate that Kaposi sarcoma may have a stem-cell origin which can differentiate into lymph cells. Kontantinopoulos and colleagues postulated that in situ Kaposi sarcoma can arise from the lymphatic system with a resultant clinical presentation of chylothorax.7 The more mainstream thought however, is that chylothorax has been found to develop secondary to metastatic disease. The present case, therefore, illustrates an unusual presentation of cytology negative chylothorax in a patient with AIDS-related Kaposi sarcoma.
1. Sridar S, Garza EG, Cox J, Rumbak MJ. Serosanguineous pleural effusions in a patient with HIV and Kaposi sarcoma: pleuroscopic findings. J Bronchology Interv Pulmonol. 2011;18(4):337-339.
2. Light RW. Chylothorax and pseudochylothorax. In: Light RW, ed. Pleural diseases. 6th ed. Philadelphia: Lippincott Williams & Wilkins, 2013:412-426.
3. Marais BJ, Pienaar J, Gie RP. Kaposi sarcoma with upper airway obstruction and bilateral chylothoraces. Pediatr Infect Dis J. 2003;22:926-928.
4. Maradona JA, Carton JA, Asensi V, Rodriguez-Guardado A. AIDS-related Kaposi sarcoma with chylothorax and pericardial involvement satisfactorily treated with liposomal doxorubicin. AIDS. 2002;16(5):806.
5. Priest ER, Weiss R. Chylothorax with Kaposi sarcoma. South Med J. 1991;84:806-807.
6. Pantanowitz L, Dezube BJ. Kaposi sarcoma in unusual locations. BMC Cancer. 2008;8:190.
7. Konstantinopoulos PA, Dezube BJ, Pantanowitz L. Morphologic and immunophenotypic evidence of in situ Kaposi sarcoma. BMC Clin Pathol. 2006;30:6:7.
Kaposi sarcoma is an angioproliferative tumor that is associated with human herpes virus-8 (HHV-8). Mucocutaneous disease is the most common site for manifestation of AIDS-related Kaposi sarcoma, commonly affecting the lower extremities, oral mucosa, face, and genitalia. Pleural effusions can occur in 36%-60% of patients with Kaposi sarcoma, and it has been documented that chylothorax is a rare, but plausible presentation in patients with Kaposi sarcoma.1 We present here a case of bilateral chylothorax in a patient with AIDS-related Kaposi sarcoma.
Case presentation and summary
A 52-year-old MSM male with AIDS (CD4, <20 mm3; viral load, 58 copies/ml) presented to the emergency department with complaints of shortness of breath, productive cough, and diarrhea for 2 days prior to presentation. His medical history also included chronic obstructive pulmonary disease, coronary artery disease, and hyperlipidemia. The patient was not on HAART because of his history of noncompliance. The results of a chest X-ray and computed-tomography (CT) scan showed that the patient had bilateral pleural effusion and a spiculated 14-mm nodule in the left upper lobe.
The patient underwent ultrasound-guided placement of a 12-French left-sided chest catheter, and a milky white fluid was aspirated from the left pleural space. Laboratory analysis of the pleural fluid confirmed an exudate with an elevated triglyceride level of 120 mg/dL (chylous, >110 mg/dL) indicating chylothorax.
On close physical examination, the patient was found to have multiple irregular plaques on the back and lower extremities. As described by dermatology, there was a violaceous indurated plaque on the left axillae, violaceous indurated plaques with superficial scale grouped on the left midlateral back, and hyperpigmented lichenified plaques and papules on bilateral shins, with some with plate-like scale. Two punch biopsies were taken of the skin lesions, which confirmed Kaposi sarcoma, plaque stage from the lesion biopsied on the back, and patch stage from the lesion biopsied in the left axilla. Cytology of the pleural fluid was negative for malignant cells. On review by the radiologist of the CT scan of the chest, there was no indication of gross distention of the thoracic duct. Treatment options were offered to the patient, and the patient was considering options for chemotherapy and home hospice given his advanced disease state at the time of discharge.
Discussion
Chylothorax occurs with a thoracic duct obstruction, which results in leakage of lymphatic fluid into the pleural cavity. The two leading causes of chylothorax are trauma and malignancy, with lymphoma being the most common cause of chylothorax among those with malignancy.2 Chylothorax, however, is a rare but documented complication of Kaposi sarcoma. Marais and colleagues reported the case of a 3-year-old HIV-positive patient with newly diagnosed Kaposi sarcoma who was found to have tumor infiltration in the thoracic duct leading to bilateral chylothorax.3 Maradona and colleagues described a 40-year-old man with AIDS-related Kaposi sarcoma who was found to have pleural and pericardial Kaposi sarcoma with chylothorax.4 Priest and colleagues wrote about a 32-year-old patient with AIDS with biopsy-proven Kaposi sarcoma who required multiple therapeutic thoracenteses for rapidly recurrent left chylothorax effusions.5
There are two leading discussions as to the pathophysiology of chylothorax that is related to Kaposi sarcoma: chylothorax developing secondary to metastatic disease or the development of chylothorax secondary to primary Kaposi sarcoma arising from the pleural region.6 One case report examined pleural and lung biopsies in a 34-year-old patient with AIDS-related Kaposi sarcoma that showed immunohistochemical staining that was suggestive of early-stage Kaposi sarcoma of lymphatic endothelial origin. The authors were attempting to illustrate that Kaposi sarcoma may have a stem-cell origin which can differentiate into lymph cells. Kontantinopoulos and colleagues postulated that in situ Kaposi sarcoma can arise from the lymphatic system with a resultant clinical presentation of chylothorax.7 The more mainstream thought however, is that chylothorax has been found to develop secondary to metastatic disease. The present case, therefore, illustrates an unusual presentation of cytology negative chylothorax in a patient with AIDS-related Kaposi sarcoma.
Kaposi sarcoma is an angioproliferative tumor that is associated with human herpes virus-8 (HHV-8). Mucocutaneous disease is the most common site for manifestation of AIDS-related Kaposi sarcoma, commonly affecting the lower extremities, oral mucosa, face, and genitalia. Pleural effusions can occur in 36%-60% of patients with Kaposi sarcoma, and it has been documented that chylothorax is a rare, but plausible presentation in patients with Kaposi sarcoma.1 We present here a case of bilateral chylothorax in a patient with AIDS-related Kaposi sarcoma.
Case presentation and summary
A 52-year-old MSM male with AIDS (CD4, <20 mm3; viral load, 58 copies/ml) presented to the emergency department with complaints of shortness of breath, productive cough, and diarrhea for 2 days prior to presentation. His medical history also included chronic obstructive pulmonary disease, coronary artery disease, and hyperlipidemia. The patient was not on HAART because of his history of noncompliance. The results of a chest X-ray and computed-tomography (CT) scan showed that the patient had bilateral pleural effusion and a spiculated 14-mm nodule in the left upper lobe.
The patient underwent ultrasound-guided placement of a 12-French left-sided chest catheter, and a milky white fluid was aspirated from the left pleural space. Laboratory analysis of the pleural fluid confirmed an exudate with an elevated triglyceride level of 120 mg/dL (chylous, >110 mg/dL) indicating chylothorax.
On close physical examination, the patient was found to have multiple irregular plaques on the back and lower extremities. As described by dermatology, there was a violaceous indurated plaque on the left axillae, violaceous indurated plaques with superficial scale grouped on the left midlateral back, and hyperpigmented lichenified plaques and papules on bilateral shins, with some with plate-like scale. Two punch biopsies were taken of the skin lesions, which confirmed Kaposi sarcoma, plaque stage from the lesion biopsied on the back, and patch stage from the lesion biopsied in the left axilla. Cytology of the pleural fluid was negative for malignant cells. On review by the radiologist of the CT scan of the chest, there was no indication of gross distention of the thoracic duct. Treatment options were offered to the patient, and the patient was considering options for chemotherapy and home hospice given his advanced disease state at the time of discharge.
Discussion
Chylothorax occurs with a thoracic duct obstruction, which results in leakage of lymphatic fluid into the pleural cavity. The two leading causes of chylothorax are trauma and malignancy, with lymphoma being the most common cause of chylothorax among those with malignancy.2 Chylothorax, however, is a rare but documented complication of Kaposi sarcoma. Marais and colleagues reported the case of a 3-year-old HIV-positive patient with newly diagnosed Kaposi sarcoma who was found to have tumor infiltration in the thoracic duct leading to bilateral chylothorax.3 Maradona and colleagues described a 40-year-old man with AIDS-related Kaposi sarcoma who was found to have pleural and pericardial Kaposi sarcoma with chylothorax.4 Priest and colleagues wrote about a 32-year-old patient with AIDS with biopsy-proven Kaposi sarcoma who required multiple therapeutic thoracenteses for rapidly recurrent left chylothorax effusions.5
There are two leading discussions as to the pathophysiology of chylothorax that is related to Kaposi sarcoma: chylothorax developing secondary to metastatic disease or the development of chylothorax secondary to primary Kaposi sarcoma arising from the pleural region.6 One case report examined pleural and lung biopsies in a 34-year-old patient with AIDS-related Kaposi sarcoma that showed immunohistochemical staining that was suggestive of early-stage Kaposi sarcoma of lymphatic endothelial origin. The authors were attempting to illustrate that Kaposi sarcoma may have a stem-cell origin which can differentiate into lymph cells. Kontantinopoulos and colleagues postulated that in situ Kaposi sarcoma can arise from the lymphatic system with a resultant clinical presentation of chylothorax.7 The more mainstream thought however, is that chylothorax has been found to develop secondary to metastatic disease. The present case, therefore, illustrates an unusual presentation of cytology negative chylothorax in a patient with AIDS-related Kaposi sarcoma.
1. Sridar S, Garza EG, Cox J, Rumbak MJ. Serosanguineous pleural effusions in a patient with HIV and Kaposi sarcoma: pleuroscopic findings. J Bronchology Interv Pulmonol. 2011;18(4):337-339.
2. Light RW. Chylothorax and pseudochylothorax. In: Light RW, ed. Pleural diseases. 6th ed. Philadelphia: Lippincott Williams & Wilkins, 2013:412-426.
3. Marais BJ, Pienaar J, Gie RP. Kaposi sarcoma with upper airway obstruction and bilateral chylothoraces. Pediatr Infect Dis J. 2003;22:926-928.
4. Maradona JA, Carton JA, Asensi V, Rodriguez-Guardado A. AIDS-related Kaposi sarcoma with chylothorax and pericardial involvement satisfactorily treated with liposomal doxorubicin. AIDS. 2002;16(5):806.
5. Priest ER, Weiss R. Chylothorax with Kaposi sarcoma. South Med J. 1991;84:806-807.
6. Pantanowitz L, Dezube BJ. Kaposi sarcoma in unusual locations. BMC Cancer. 2008;8:190.
7. Konstantinopoulos PA, Dezube BJ, Pantanowitz L. Morphologic and immunophenotypic evidence of in situ Kaposi sarcoma. BMC Clin Pathol. 2006;30:6:7.
1. Sridar S, Garza EG, Cox J, Rumbak MJ. Serosanguineous pleural effusions in a patient with HIV and Kaposi sarcoma: pleuroscopic findings. J Bronchology Interv Pulmonol. 2011;18(4):337-339.
2. Light RW. Chylothorax and pseudochylothorax. In: Light RW, ed. Pleural diseases. 6th ed. Philadelphia: Lippincott Williams & Wilkins, 2013:412-426.
3. Marais BJ, Pienaar J, Gie RP. Kaposi sarcoma with upper airway obstruction and bilateral chylothoraces. Pediatr Infect Dis J. 2003;22:926-928.
4. Maradona JA, Carton JA, Asensi V, Rodriguez-Guardado A. AIDS-related Kaposi sarcoma with chylothorax and pericardial involvement satisfactorily treated with liposomal doxorubicin. AIDS. 2002;16(5):806.
5. Priest ER, Weiss R. Chylothorax with Kaposi sarcoma. South Med J. 1991;84:806-807.
6. Pantanowitz L, Dezube BJ. Kaposi sarcoma in unusual locations. BMC Cancer. 2008;8:190.
7. Konstantinopoulos PA, Dezube BJ, Pantanowitz L. Morphologic and immunophenotypic evidence of in situ Kaposi sarcoma. BMC Clin Pathol. 2006;30:6:7.
The Princess and the Pea-sized Nodule
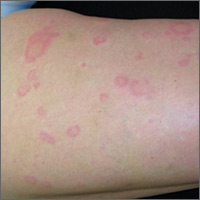
For years, this 33-year-old woman has had a firm, pea-sized nodule on her left upper back. It was never a problem until recently, when it suddenly enlarged and became red, swollen, and tender.
She was prescribed antibiotics (trimethoprim and sulfa) by a provider at her local urgent care center. Dubious of the diagnosis—carbuncle—she sought referral to dermatology.
The patient claims to be in otherwise excellent health, with no history of similar problems. She denies manual manipulation of the lesion.
EXAMINATION
The patient is afebrile and in no distress. On her upper left back is a round, cystic lesion measuring 3.5 cm. It appears swollen and red. The erythema, though impressive, is confined to
the area immediately around the margins. Palpation reveals increased warmth and modest tenderness. A central punctum can be seen in the center of the fluctuant lesion.

After a brief discussion of options, the lesion is incised and drained under sterile conditions with lidocaine and epinephrine. Cheesy, odoriferous material is expressed, effectively flattening the lesion.
What is the diagnosis?
DISCUSSION
Epidermal cysts are utterly common, especially in oil-rich areas such as the upper back. They are often mistakenly called sebaceous cysts; however, true sebaceous cysts are quite rare and contain only pure, clear oil.
When ruptured by trauma, epidermal cysts become inflamed and cheesy material leaks into deep tissues. This activates an immune response in which the body sends out white cells to clean up the leakage—what we call inflammation.
There are many types of cysts (eg, acne cysts, ganglion cysts, Bartholin gland cysts) but none resemble epidermal cysts. Providers often mistake epidermal cysts for carbuncles or boils and may prescribe oral or systemic antibiotics, or incise the cysts and pack the space. The truth is that inflamed epidermal cysts do not represent infection—hence the limited area of redness. By contrast, cellulitis or carbuncle would manifest with tenderness and blush of erythema over the entire area.
The fact that this lesion arose from a longstanding antecedent nodule is consistent with the diagnosis, as are the cheesy, odoriferous contents liberated by incision and drainage. Liberating the contents merely buys the patient relief from the pressure and pain, since the cyst wall is still present and will almost certainly fill up again. The ultimate solution is excision after the emptied cyst has had time to shrink back to its original size, which will minimize the scarring. There is no need to pack such cysts after incision and drainage, though they will continue to drain for a few days.
TAKE-HOME LEARNING POINTS
- Epidermal cysts are common, especially in oil-rich areas such as the face, back, chest, and neck.
- Epidermal cysts are often mistakenly called sebaceous cysts, but these are actually quite rare, containing clear sebum (oil) and no cheesy material.
- Even though such inflamed cysts are often mistaken for boils, carbuncles, and abscesses, they have nothing to do with bacteria or infection and therefore do not require antibiotics or packing after incision and drainage.
- For recurrent inflamed cysts, the treatment of choice is complete excision, done only after the cyst has shrunk to its smallest size.
For years, this 33-year-old woman has had a firm, pea-sized nodule on her left upper back. It was never a problem until recently, when it suddenly enlarged and became red, swollen, and tender.
She was prescribed antibiotics (trimethoprim and sulfa) by a provider at her local urgent care center. Dubious of the diagnosis—carbuncle—she sought referral to dermatology.
The patient claims to be in otherwise excellent health, with no history of similar problems. She denies manual manipulation of the lesion.
EXAMINATION
The patient is afebrile and in no distress. On her upper left back is a round, cystic lesion measuring 3.5 cm. It appears swollen and red. The erythema, though impressive, is confined to
the area immediately around the margins. Palpation reveals increased warmth and modest tenderness. A central punctum can be seen in the center of the fluctuant lesion.

After a brief discussion of options, the lesion is incised and drained under sterile conditions with lidocaine and epinephrine. Cheesy, odoriferous material is expressed, effectively flattening the lesion.
What is the diagnosis?
DISCUSSION
Epidermal cysts are utterly common, especially in oil-rich areas such as the upper back. They are often mistakenly called sebaceous cysts; however, true sebaceous cysts are quite rare and contain only pure, clear oil.
When ruptured by trauma, epidermal cysts become inflamed and cheesy material leaks into deep tissues. This activates an immune response in which the body sends out white cells to clean up the leakage—what we call inflammation.
There are many types of cysts (eg, acne cysts, ganglion cysts, Bartholin gland cysts) but none resemble epidermal cysts. Providers often mistake epidermal cysts for carbuncles or boils and may prescribe oral or systemic antibiotics, or incise the cysts and pack the space. The truth is that inflamed epidermal cysts do not represent infection—hence the limited area of redness. By contrast, cellulitis or carbuncle would manifest with tenderness and blush of erythema over the entire area.
The fact that this lesion arose from a longstanding antecedent nodule is consistent with the diagnosis, as are the cheesy, odoriferous contents liberated by incision and drainage. Liberating the contents merely buys the patient relief from the pressure and pain, since the cyst wall is still present and will almost certainly fill up again. The ultimate solution is excision after the emptied cyst has had time to shrink back to its original size, which will minimize the scarring. There is no need to pack such cysts after incision and drainage, though they will continue to drain for a few days.
TAKE-HOME LEARNING POINTS
- Epidermal cysts are common, especially in oil-rich areas such as the face, back, chest, and neck.
- Epidermal cysts are often mistakenly called sebaceous cysts, but these are actually quite rare, containing clear sebum (oil) and no cheesy material.
- Even though such inflamed cysts are often mistaken for boils, carbuncles, and abscesses, they have nothing to do with bacteria or infection and therefore do not require antibiotics or packing after incision and drainage.
- For recurrent inflamed cysts, the treatment of choice is complete excision, done only after the cyst has shrunk to its smallest size.
For years, this 33-year-old woman has had a firm, pea-sized nodule on her left upper back. It was never a problem until recently, when it suddenly enlarged and became red, swollen, and tender.
She was prescribed antibiotics (trimethoprim and sulfa) by a provider at her local urgent care center. Dubious of the diagnosis—carbuncle—she sought referral to dermatology.
The patient claims to be in otherwise excellent health, with no history of similar problems. She denies manual manipulation of the lesion.
EXAMINATION
The patient is afebrile and in no distress. On her upper left back is a round, cystic lesion measuring 3.5 cm. It appears swollen and red. The erythema, though impressive, is confined to
the area immediately around the margins. Palpation reveals increased warmth and modest tenderness. A central punctum can be seen in the center of the fluctuant lesion.

After a brief discussion of options, the lesion is incised and drained under sterile conditions with lidocaine and epinephrine. Cheesy, odoriferous material is expressed, effectively flattening the lesion.
What is the diagnosis?
DISCUSSION
Epidermal cysts are utterly common, especially in oil-rich areas such as the upper back. They are often mistakenly called sebaceous cysts; however, true sebaceous cysts are quite rare and contain only pure, clear oil.
When ruptured by trauma, epidermal cysts become inflamed and cheesy material leaks into deep tissues. This activates an immune response in which the body sends out white cells to clean up the leakage—what we call inflammation.
There are many types of cysts (eg, acne cysts, ganglion cysts, Bartholin gland cysts) but none resemble epidermal cysts. Providers often mistake epidermal cysts for carbuncles or boils and may prescribe oral or systemic antibiotics, or incise the cysts and pack the space. The truth is that inflamed epidermal cysts do not represent infection—hence the limited area of redness. By contrast, cellulitis or carbuncle would manifest with tenderness and blush of erythema over the entire area.
The fact that this lesion arose from a longstanding antecedent nodule is consistent with the diagnosis, as are the cheesy, odoriferous contents liberated by incision and drainage. Liberating the contents merely buys the patient relief from the pressure and pain, since the cyst wall is still present and will almost certainly fill up again. The ultimate solution is excision after the emptied cyst has had time to shrink back to its original size, which will minimize the scarring. There is no need to pack such cysts after incision and drainage, though they will continue to drain for a few days.
TAKE-HOME LEARNING POINTS
- Epidermal cysts are common, especially in oil-rich areas such as the face, back, chest, and neck.
- Epidermal cysts are often mistakenly called sebaceous cysts, but these are actually quite rare, containing clear sebum (oil) and no cheesy material.
- Even though such inflamed cysts are often mistaken for boils, carbuncles, and abscesses, they have nothing to do with bacteria or infection and therefore do not require antibiotics or packing after incision and drainage.
- For recurrent inflamed cysts, the treatment of choice is complete excision, done only after the cyst has shrunk to its smallest size.
Improving cancer care through modern portfolio theory
We struggle daily to improve cancer care – to improve our therapeutic outcomes in cancer – as individual physicians and as researchers. We work collectively to disseminate information and collaborate, and there are welcome calls for open data sharing to accelerate progress.1 We enroll patients on clinical trials, or we work in a basic science lab to discover mechanisms of carcinogenesis and potential therapeutic targets. We discuss “n of 1” trials and the “paradigm shift of precision oncology,” and we are optimistic about the future of cancer care.
Leaving the world of biology and clinical trials for a minute, we also can apply economic theory in our never-ending quest to improve cancer outcomes. One area of interest may be modern portfolio theory (MPT), which the economist Harry Markowitz introduced in an essay in 1952 and later won the Nobel Prize for his work.
At least 71 billionaires live in the San Francisco Bay Area, where I live, but 14,000 children (13%) in the area live below the poverty line.3 When there is a range of asset allocations in health care, results can vary not on the basis of the underlying disease state or the quality of the provider, but on access to care. As an example, most pediatric cancers are curable, yet a recent retrospective analysis of data in the SEER-Medicare registry showed that mortality within 1 month of diagnosis of childhood cancer related in part to socioeconomic factors – those patients with a lower socioeconomic status (which correlates with being an ethnic minority in the United States) were more likely to die within a month of diagnosis of their cancer than were patients with a higher socioeconomic status.3 Here is where MPT can transform the cancer outcomes landscape at no additional investment in basic science or costly precision medicine5: by triaging these patients according to their disease state rather than their ability to pay, they could be administered curative chemotherapy, placed on the appropriate clinical trial, and be cured of their cancer like other children of higher socioeconomic status.
My colleagues and I observed a similar trend when we looked at treatment of diffuse large-cell non-Hodgkin lymphoma in Medicare recipients.6 Although the cure rate is as high as 60%-80% with the use of CHOP (cyclophosphamide, doxorubicin, vincristine, prednisolone) or R (rituxin)-CHOP chemotherapy, we found that many patients had received suboptimal chemotherapy. Upon closer examination, we found that there were variations in care by socioeconomic status even in a single-payer system. Thus aspects of cultural literacy and additional efforts for triage need to be developed, but again, application of MPT could be instrumental in improving cancer cure rates by reducing disparities in care by allocating assets to solve access-to-care issues, and curing these patients of their non-Hodgkin lymphoma.
A physician at a Bay Area health care system notes that the open slots in his schedule are triaged by his employer by the patient’s ability to pay – well-insured patients are seen within a few days, but there are very few slots for Medicaid patients, who have to wait weeks or longer to be seen. During this time, their malignancies have time to grow, and potentially metastasize. This may provide suboptimal outcomes for some patients in his community.
We solved this problem at a local hospital where all patients were on Medicaid or uninsured. We triaged patients according to severity of illness, with patients with rapidly growing cancers, particularly curable ones, were brought in as soon as possible and patients with stable benign hematologic conditions seen on a less urgent basis. A social worker and I saw patients together. She would find them resources such as transportation, food, copay assistance to help them through their treatment, and I would optimize their cancer care clinically. On a small scale, this application of MPT (or asset allocation) worked quite well. Perhaps it can be reproduced on a much larger scale. Return on investment relates largely to how you allocate your assets. What’s nice about these applications of MPT is that the return on investment – increasing the cure rate of cancer - is quite large for just a minimal change in asset allocation.
1. Bertagnolli M, Sartor O, Chabner BA, et al. Advantages of a truly open-access data-sharing model. N Engl J Med. 2017;376(12):1178-1181.
2. Baum M. Justice. In: The scepticaemic surgeon: how not to win friends and influence people. New York: Nova Science Pubkishers; November 30, 2014.
3. Glaeser E. Gentfrification and its discontents. Wall Street Journal. May 5, 2017.
4. Green AL, Furutani E, Riberio KB, Galindo CR. Death within 1 month of diagnosis in childhood cancer : analysis of risk factors and scope of the problem. J Clin Oncol. 2017;35(12):1320-1327.
5. McCartney M. Are we too captivated by precision medicine? http://www.bmj.com/content/356/bmj.j1168.long. Published March 9, 2017. Accessed May 12, 2017.
6. Griffiths R, Gleeson M, Knopf K, Danese M. Racial differences in treatment and survival in older patients with diffuse large B-cell lymphoma. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2995801/. Published November 12, 2010. Accessed May 12, 2017.
We struggle daily to improve cancer care – to improve our therapeutic outcomes in cancer – as individual physicians and as researchers. We work collectively to disseminate information and collaborate, and there are welcome calls for open data sharing to accelerate progress.1 We enroll patients on clinical trials, or we work in a basic science lab to discover mechanisms of carcinogenesis and potential therapeutic targets. We discuss “n of 1” trials and the “paradigm shift of precision oncology,” and we are optimistic about the future of cancer care.
Leaving the world of biology and clinical trials for a minute, we also can apply economic theory in our never-ending quest to improve cancer outcomes. One area of interest may be modern portfolio theory (MPT), which the economist Harry Markowitz introduced in an essay in 1952 and later won the Nobel Prize for his work.
At least 71 billionaires live in the San Francisco Bay Area, where I live, but 14,000 children (13%) in the area live below the poverty line.3 When there is a range of asset allocations in health care, results can vary not on the basis of the underlying disease state or the quality of the provider, but on access to care. As an example, most pediatric cancers are curable, yet a recent retrospective analysis of data in the SEER-Medicare registry showed that mortality within 1 month of diagnosis of childhood cancer related in part to socioeconomic factors – those patients with a lower socioeconomic status (which correlates with being an ethnic minority in the United States) were more likely to die within a month of diagnosis of their cancer than were patients with a higher socioeconomic status.3 Here is where MPT can transform the cancer outcomes landscape at no additional investment in basic science or costly precision medicine5: by triaging these patients according to their disease state rather than their ability to pay, they could be administered curative chemotherapy, placed on the appropriate clinical trial, and be cured of their cancer like other children of higher socioeconomic status.
My colleagues and I observed a similar trend when we looked at treatment of diffuse large-cell non-Hodgkin lymphoma in Medicare recipients.6 Although the cure rate is as high as 60%-80% with the use of CHOP (cyclophosphamide, doxorubicin, vincristine, prednisolone) or R (rituxin)-CHOP chemotherapy, we found that many patients had received suboptimal chemotherapy. Upon closer examination, we found that there were variations in care by socioeconomic status even in a single-payer system. Thus aspects of cultural literacy and additional efforts for triage need to be developed, but again, application of MPT could be instrumental in improving cancer cure rates by reducing disparities in care by allocating assets to solve access-to-care issues, and curing these patients of their non-Hodgkin lymphoma.
A physician at a Bay Area health care system notes that the open slots in his schedule are triaged by his employer by the patient’s ability to pay – well-insured patients are seen within a few days, but there are very few slots for Medicaid patients, who have to wait weeks or longer to be seen. During this time, their malignancies have time to grow, and potentially metastasize. This may provide suboptimal outcomes for some patients in his community.
We solved this problem at a local hospital where all patients were on Medicaid or uninsured. We triaged patients according to severity of illness, with patients with rapidly growing cancers, particularly curable ones, were brought in as soon as possible and patients with stable benign hematologic conditions seen on a less urgent basis. A social worker and I saw patients together. She would find them resources such as transportation, food, copay assistance to help them through their treatment, and I would optimize their cancer care clinically. On a small scale, this application of MPT (or asset allocation) worked quite well. Perhaps it can be reproduced on a much larger scale. Return on investment relates largely to how you allocate your assets. What’s nice about these applications of MPT is that the return on investment – increasing the cure rate of cancer - is quite large for just a minimal change in asset allocation.
We struggle daily to improve cancer care – to improve our therapeutic outcomes in cancer – as individual physicians and as researchers. We work collectively to disseminate information and collaborate, and there are welcome calls for open data sharing to accelerate progress.1 We enroll patients on clinical trials, or we work in a basic science lab to discover mechanisms of carcinogenesis and potential therapeutic targets. We discuss “n of 1” trials and the “paradigm shift of precision oncology,” and we are optimistic about the future of cancer care.
Leaving the world of biology and clinical trials for a minute, we also can apply economic theory in our never-ending quest to improve cancer outcomes. One area of interest may be modern portfolio theory (MPT), which the economist Harry Markowitz introduced in an essay in 1952 and later won the Nobel Prize for his work.
At least 71 billionaires live in the San Francisco Bay Area, where I live, but 14,000 children (13%) in the area live below the poverty line.3 When there is a range of asset allocations in health care, results can vary not on the basis of the underlying disease state or the quality of the provider, but on access to care. As an example, most pediatric cancers are curable, yet a recent retrospective analysis of data in the SEER-Medicare registry showed that mortality within 1 month of diagnosis of childhood cancer related in part to socioeconomic factors – those patients with a lower socioeconomic status (which correlates with being an ethnic minority in the United States) were more likely to die within a month of diagnosis of their cancer than were patients with a higher socioeconomic status.3 Here is where MPT can transform the cancer outcomes landscape at no additional investment in basic science or costly precision medicine5: by triaging these patients according to their disease state rather than their ability to pay, they could be administered curative chemotherapy, placed on the appropriate clinical trial, and be cured of their cancer like other children of higher socioeconomic status.
My colleagues and I observed a similar trend when we looked at treatment of diffuse large-cell non-Hodgkin lymphoma in Medicare recipients.6 Although the cure rate is as high as 60%-80% with the use of CHOP (cyclophosphamide, doxorubicin, vincristine, prednisolone) or R (rituxin)-CHOP chemotherapy, we found that many patients had received suboptimal chemotherapy. Upon closer examination, we found that there were variations in care by socioeconomic status even in a single-payer system. Thus aspects of cultural literacy and additional efforts for triage need to be developed, but again, application of MPT could be instrumental in improving cancer cure rates by reducing disparities in care by allocating assets to solve access-to-care issues, and curing these patients of their non-Hodgkin lymphoma.
A physician at a Bay Area health care system notes that the open slots in his schedule are triaged by his employer by the patient’s ability to pay – well-insured patients are seen within a few days, but there are very few slots for Medicaid patients, who have to wait weeks or longer to be seen. During this time, their malignancies have time to grow, and potentially metastasize. This may provide suboptimal outcomes for some patients in his community.
We solved this problem at a local hospital where all patients were on Medicaid or uninsured. We triaged patients according to severity of illness, with patients with rapidly growing cancers, particularly curable ones, were brought in as soon as possible and patients with stable benign hematologic conditions seen on a less urgent basis. A social worker and I saw patients together. She would find them resources such as transportation, food, copay assistance to help them through their treatment, and I would optimize their cancer care clinically. On a small scale, this application of MPT (or asset allocation) worked quite well. Perhaps it can be reproduced on a much larger scale. Return on investment relates largely to how you allocate your assets. What’s nice about these applications of MPT is that the return on investment – increasing the cure rate of cancer - is quite large for just a minimal change in asset allocation.
1. Bertagnolli M, Sartor O, Chabner BA, et al. Advantages of a truly open-access data-sharing model. N Engl J Med. 2017;376(12):1178-1181.
2. Baum M. Justice. In: The scepticaemic surgeon: how not to win friends and influence people. New York: Nova Science Pubkishers; November 30, 2014.
3. Glaeser E. Gentfrification and its discontents. Wall Street Journal. May 5, 2017.
4. Green AL, Furutani E, Riberio KB, Galindo CR. Death within 1 month of diagnosis in childhood cancer : analysis of risk factors and scope of the problem. J Clin Oncol. 2017;35(12):1320-1327.
5. McCartney M. Are we too captivated by precision medicine? http://www.bmj.com/content/356/bmj.j1168.long. Published March 9, 2017. Accessed May 12, 2017.
6. Griffiths R, Gleeson M, Knopf K, Danese M. Racial differences in treatment and survival in older patients with diffuse large B-cell lymphoma. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2995801/. Published November 12, 2010. Accessed May 12, 2017.
1. Bertagnolli M, Sartor O, Chabner BA, et al. Advantages of a truly open-access data-sharing model. N Engl J Med. 2017;376(12):1178-1181.
2. Baum M. Justice. In: The scepticaemic surgeon: how not to win friends and influence people. New York: Nova Science Pubkishers; November 30, 2014.
3. Glaeser E. Gentfrification and its discontents. Wall Street Journal. May 5, 2017.
4. Green AL, Furutani E, Riberio KB, Galindo CR. Death within 1 month of diagnosis in childhood cancer : analysis of risk factors and scope of the problem. J Clin Oncol. 2017;35(12):1320-1327.
5. McCartney M. Are we too captivated by precision medicine? http://www.bmj.com/content/356/bmj.j1168.long. Published March 9, 2017. Accessed May 12, 2017.
6. Griffiths R, Gleeson M, Knopf K, Danese M. Racial differences in treatment and survival in older patients with diffuse large B-cell lymphoma. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2995801/. Published November 12, 2010. Accessed May 12, 2017.
Metastatic Kaposi sarcoma with osseous involvement in a patient with AIDS
Kaposi sarcoma is an AIDS-defining illness associated with human herpes virus-8 (HHV-8) co-infection. It was described in 1872 by the Hungarian dermatologist Mortiz Kaposi, and was an isolated and sporadic occurrence before the emergence of HIV infection and AIDS.1 It was first affiliated as an AIDS-associated neoplasm in 1981.1 Kaposi sarcoma is a systemic disease that can present with cutaneous lesions with or without internal involvement. There are four subtypes: Classic, African endemic, AIDS-related (CD4 count, <200), and Kaposi sarcoma in iatrogenically immunosuppressed patients. The disease has the propensity to manifest in the skin and gastro-intestinal and respiratory tracts, and osseous involvement is rarely encountered. We present here the case of an AIDS-positive man with generalized bone pain as a result of metastasis from Kaposi sarcoma. Our discussion includes the epidemiological, clinical, pathological, and radiological facets of AIDS-related Kaposi sarcoma, and the anomaly of osseous involvement.
Case presentation and summary
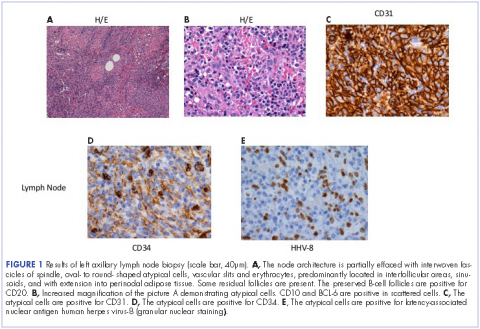
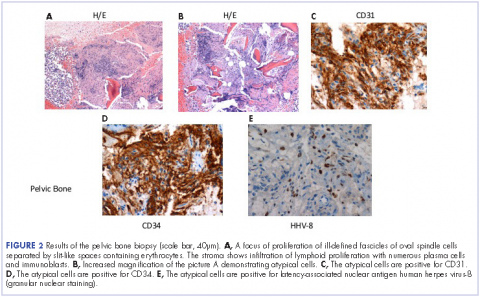
He restarted his previous HAART regimen in March 2016, and was subsequently started on chemotherapy with liposomal doxorubicin (50 mg [20 mg/m2] in 250 ml D5W IV every 2 weeks) because of his extensive disease.2 He completed 6 cycles by June 2016. However, he returned in July 2016 with worsening back pain. A repeat CT scan revealed significant improvement in the disseminated lymphadenopathy, but worsening osseous metastatic disease was seen in the lumbar, thoracic, and pelvic regions. A pelvic lytic lesion biopsy revealed Kaposi sarcoma; pathology showed spindle cells positive for CD34, CD31, and HHV-8 (Figure 2). The patient received palliative radiation to the spine, aiding in pain management and ambulatory dysfunction. He continued with his noncompliance with all medications and outpatient follow-ups, and succumbed to his disease burden.
Discussion
Kaposi sarcoma is a low-grade mesenchymal tumor that involves the blood and lymphatic vessels.3 Its association with AIDS was revealed in the early 1980s at the start of the HIV epidemic in the United States. In 1994, Chang and colleagues discovered the association between Karposi sarcoma and HHV-8 by isolating DNA fragments of HHV in Kaposi sarcoma tumors from AIDS patients.4 The mode of transmission of HHV-8 has not been fully decoded. It has been presumed that adult homosexual contact continues to be an important route of transmission, inferring a common route of infection. In 1990, the overall risk of developing Kaposi sarcoma in AIDS patients was 20,000 times greater than it was in the general population, and 300 times greater than in other immunosuppressed patients.5 This suggests an increase in incidence, in direct relation, with a decrease in the CD4 count.
Kaposi sarcoma can present with a range of clinical features, from negligible cutaneous lesions to a hastily progressing neoplasm. Involvement in the musculoskeletal system is infrequent, but encountered increasingly in the AIDS-related subtype. Moreover, it is recurrently observed in the African population.6 In one of the largest reviews to date exploring Kaposi sarcoma involving the musculoskeletal system, Caponetti and colleagues observed the greatest osseous involvement distinctly in patients with CD4 and T-cell counts below 100 cells/mm3.6
Kaposi sarcoma musculoskeletal involvement, specifically bone, is atypical. If it does occur, it usually manifests as a result of contiguous invasion from an adjacent nonosseous lesion. Caponetti and colleagues that isolated osseous Kaposi sarcoma lesions (with no overlying skin lesion) were found to be more likely to be associated with AIDS in the review by Caponetti and colleagues.6 As in our patient, it is also typically a manifestation of more widely disseminated disease.7
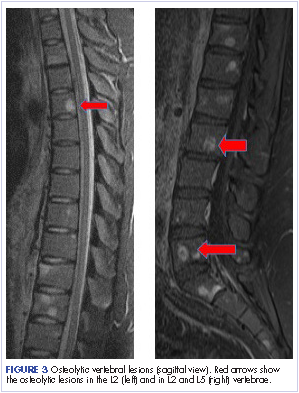
Most of the osseous lytic lesions in AIDS patients are located in the axial skeleton. Radiological features of musculoskeletal Kaposi sarcoma are variable. As observed by Caponetti and colleagues, Kaposi sarcoma lesions can appear as a periosteal reaction, cortical erosions, osteolysis, or osseous destruction, with irregular-shaped cortical erosions being most typical.6 Despite their osteolytic features, Kaposi sarcoma lesions are often not visualized by conventional radiography.6 The preferred imaging for identification of lytic bone changes is CT (Figure 3). Magnetic resonance imaging can also help distinguish marrow abnormalities as well as adjacent soft tissues masses. Radiologically, Kaposi sarcoma osseous lesions have parallel features to bacillary angiomatosis, tuberculosis, or lymphoma.8 Therefore, biopsy of the lesion is essential in establishing the diagnosis of Kaposi sarcoma.
In theory, there should be clinical improvement in Kaposi sarcoma when immunity is restored. Cancers caused by the Epstein-Barr virus and Kaposi sarcoma-associated herpes virus may eventually also be preventable with vaccines.10
There is rarely bone involvement without the foreshadowing of a poor prognosis. Erroneous patient care may inevitably arise from Kaposi sarcoma in uncharacteristic sites. A differential of Kaposi sarcoma should be included if a patient with AIDS presents with osteolytic lesions on imaging. Biopsying the lesion cements the diagnosis and eliminates the possibility of mimicry conditions such as bacillary angiomatosis, benign vascular lesions, and angiosarcoma. As of today, a HAART regimen remains the standard initial care for patients with Kaposi sarcoma.
1. Radu O, Pantanowitz L. Kaposi sarcoma. Arch Pathol Lab Med. 2013;137:289-294.
2. Northfelt DW, Dezube BJ, Thommes JA, et al. Pegylated-liposomal doxorubicin versus doxorubicin, bleomycin, and vincristine in the treatment of AIDS-related Kaposi sarcoma: results of a randomized phase III clinical trial. J Clin Oncol. 1998;16(7):2445-2451.
3. Restrepo CS, Martinez S, Lemos JA, et al. Imaging manifestations of Kaposi sarcoma. RadioGraphics. 2006;26:1169-1185.
4. Chang Y, Cesarman E, Pessin MS, et al. Identification of herpes virus-like DNA sequences in AIDS-associated Kaposi sarcoma. Science. 1994;266:1865-1869.
5. Beral V, Peterman TA, Berkelman RL, Jaffe HW. Kaposi sarcoma among persons with AIDS: a sexually transmitted infection? Lancet. 1990;335:123-128.
6. Caponetti G, Dezube BJ, Restrepo CS, Pantanowitz I. Kaposi sarcoma of the musculoskeletal system: a review of 66 patients. Cancer. 2007;109(6):1040-1052.
7. Krishna G, Chitkara RK. Osseous Kaposi sarcoma. JAMA. 2003;286(9):1106.
8. Thanos L, Mylona S, Kalioras V, Pomoni M, Batakis N. Osseous Kaposi sarcoma in an HIV-positive patient. Skeletal Radiol. 2004;33(4):241-243.
9. Guiholt A, Dupin N, Marcelin AG, et al. Low T-cell response to human herpesvirus 8 in patients with AIDS-related and classic Kaposi sarcoma. J Infect Dis. 2006;194(8):1078-1088.
10. Gopal S, Achenbach CJ, Yanik EL, Dither DP, Eron JJ, Engels EA. Moving forward in HIV-associated cancer. J Clin Oncol. 2014;32(9):876-880.
Kaposi sarcoma is an AIDS-defining illness associated with human herpes virus-8 (HHV-8) co-infection. It was described in 1872 by the Hungarian dermatologist Mortiz Kaposi, and was an isolated and sporadic occurrence before the emergence of HIV infection and AIDS.1 It was first affiliated as an AIDS-associated neoplasm in 1981.1 Kaposi sarcoma is a systemic disease that can present with cutaneous lesions with or without internal involvement. There are four subtypes: Classic, African endemic, AIDS-related (CD4 count, <200), and Kaposi sarcoma in iatrogenically immunosuppressed patients. The disease has the propensity to manifest in the skin and gastro-intestinal and respiratory tracts, and osseous involvement is rarely encountered. We present here the case of an AIDS-positive man with generalized bone pain as a result of metastasis from Kaposi sarcoma. Our discussion includes the epidemiological, clinical, pathological, and radiological facets of AIDS-related Kaposi sarcoma, and the anomaly of osseous involvement.
Case presentation and summary
He restarted his previous HAART regimen in March 2016, and was subsequently started on chemotherapy with liposomal doxorubicin (50 mg [20 mg/m2] in 250 ml D5W IV every 2 weeks) because of his extensive disease.2 He completed 6 cycles by June 2016. However, he returned in July 2016 with worsening back pain. A repeat CT scan revealed significant improvement in the disseminated lymphadenopathy, but worsening osseous metastatic disease was seen in the lumbar, thoracic, and pelvic regions. A pelvic lytic lesion biopsy revealed Kaposi sarcoma; pathology showed spindle cells positive for CD34, CD31, and HHV-8 (Figure 2). The patient received palliative radiation to the spine, aiding in pain management and ambulatory dysfunction. He continued with his noncompliance with all medications and outpatient follow-ups, and succumbed to his disease burden.
Discussion
Kaposi sarcoma is a low-grade mesenchymal tumor that involves the blood and lymphatic vessels.3 Its association with AIDS was revealed in the early 1980s at the start of the HIV epidemic in the United States. In 1994, Chang and colleagues discovered the association between Karposi sarcoma and HHV-8 by isolating DNA fragments of HHV in Kaposi sarcoma tumors from AIDS patients.4 The mode of transmission of HHV-8 has not been fully decoded. It has been presumed that adult homosexual contact continues to be an important route of transmission, inferring a common route of infection. In 1990, the overall risk of developing Kaposi sarcoma in AIDS patients was 20,000 times greater than it was in the general population, and 300 times greater than in other immunosuppressed patients.5 This suggests an increase in incidence, in direct relation, with a decrease in the CD4 count.
Kaposi sarcoma can present with a range of clinical features, from negligible cutaneous lesions to a hastily progressing neoplasm. Involvement in the musculoskeletal system is infrequent, but encountered increasingly in the AIDS-related subtype. Moreover, it is recurrently observed in the African population.6 In one of the largest reviews to date exploring Kaposi sarcoma involving the musculoskeletal system, Caponetti and colleagues observed the greatest osseous involvement distinctly in patients with CD4 and T-cell counts below 100 cells/mm3.6
Kaposi sarcoma musculoskeletal involvement, specifically bone, is atypical. If it does occur, it usually manifests as a result of contiguous invasion from an adjacent nonosseous lesion. Caponetti and colleagues that isolated osseous Kaposi sarcoma lesions (with no overlying skin lesion) were found to be more likely to be associated with AIDS in the review by Caponetti and colleagues.6 As in our patient, it is also typically a manifestation of more widely disseminated disease.7
Most of the osseous lytic lesions in AIDS patients are located in the axial skeleton. Radiological features of musculoskeletal Kaposi sarcoma are variable. As observed by Caponetti and colleagues, Kaposi sarcoma lesions can appear as a periosteal reaction, cortical erosions, osteolysis, or osseous destruction, with irregular-shaped cortical erosions being most typical.6 Despite their osteolytic features, Kaposi sarcoma lesions are often not visualized by conventional radiography.6 The preferred imaging for identification of lytic bone changes is CT (Figure 3). Magnetic resonance imaging can also help distinguish marrow abnormalities as well as adjacent soft tissues masses. Radiologically, Kaposi sarcoma osseous lesions have parallel features to bacillary angiomatosis, tuberculosis, or lymphoma.8 Therefore, biopsy of the lesion is essential in establishing the diagnosis of Kaposi sarcoma.
In theory, there should be clinical improvement in Kaposi sarcoma when immunity is restored. Cancers caused by the Epstein-Barr virus and Kaposi sarcoma-associated herpes virus may eventually also be preventable with vaccines.10
There is rarely bone involvement without the foreshadowing of a poor prognosis. Erroneous patient care may inevitably arise from Kaposi sarcoma in uncharacteristic sites. A differential of Kaposi sarcoma should be included if a patient with AIDS presents with osteolytic lesions on imaging. Biopsying the lesion cements the diagnosis and eliminates the possibility of mimicry conditions such as bacillary angiomatosis, benign vascular lesions, and angiosarcoma. As of today, a HAART regimen remains the standard initial care for patients with Kaposi sarcoma.
Kaposi sarcoma is an AIDS-defining illness associated with human herpes virus-8 (HHV-8) co-infection. It was described in 1872 by the Hungarian dermatologist Mortiz Kaposi, and was an isolated and sporadic occurrence before the emergence of HIV infection and AIDS.1 It was first affiliated as an AIDS-associated neoplasm in 1981.1 Kaposi sarcoma is a systemic disease that can present with cutaneous lesions with or without internal involvement. There are four subtypes: Classic, African endemic, AIDS-related (CD4 count, <200), and Kaposi sarcoma in iatrogenically immunosuppressed patients. The disease has the propensity to manifest in the skin and gastro-intestinal and respiratory tracts, and osseous involvement is rarely encountered. We present here the case of an AIDS-positive man with generalized bone pain as a result of metastasis from Kaposi sarcoma. Our discussion includes the epidemiological, clinical, pathological, and radiological facets of AIDS-related Kaposi sarcoma, and the anomaly of osseous involvement.
Case presentation and summary
He restarted his previous HAART regimen in March 2016, and was subsequently started on chemotherapy with liposomal doxorubicin (50 mg [20 mg/m2] in 250 ml D5W IV every 2 weeks) because of his extensive disease.2 He completed 6 cycles by June 2016. However, he returned in July 2016 with worsening back pain. A repeat CT scan revealed significant improvement in the disseminated lymphadenopathy, but worsening osseous metastatic disease was seen in the lumbar, thoracic, and pelvic regions. A pelvic lytic lesion biopsy revealed Kaposi sarcoma; pathology showed spindle cells positive for CD34, CD31, and HHV-8 (Figure 2). The patient received palliative radiation to the spine, aiding in pain management and ambulatory dysfunction. He continued with his noncompliance with all medications and outpatient follow-ups, and succumbed to his disease burden.
Discussion
Kaposi sarcoma is a low-grade mesenchymal tumor that involves the blood and lymphatic vessels.3 Its association with AIDS was revealed in the early 1980s at the start of the HIV epidemic in the United States. In 1994, Chang and colleagues discovered the association between Karposi sarcoma and HHV-8 by isolating DNA fragments of HHV in Kaposi sarcoma tumors from AIDS patients.4 The mode of transmission of HHV-8 has not been fully decoded. It has been presumed that adult homosexual contact continues to be an important route of transmission, inferring a common route of infection. In 1990, the overall risk of developing Kaposi sarcoma in AIDS patients was 20,000 times greater than it was in the general population, and 300 times greater than in other immunosuppressed patients.5 This suggests an increase in incidence, in direct relation, with a decrease in the CD4 count.
Kaposi sarcoma can present with a range of clinical features, from negligible cutaneous lesions to a hastily progressing neoplasm. Involvement in the musculoskeletal system is infrequent, but encountered increasingly in the AIDS-related subtype. Moreover, it is recurrently observed in the African population.6 In one of the largest reviews to date exploring Kaposi sarcoma involving the musculoskeletal system, Caponetti and colleagues observed the greatest osseous involvement distinctly in patients with CD4 and T-cell counts below 100 cells/mm3.6
Kaposi sarcoma musculoskeletal involvement, specifically bone, is atypical. If it does occur, it usually manifests as a result of contiguous invasion from an adjacent nonosseous lesion. Caponetti and colleagues that isolated osseous Kaposi sarcoma lesions (with no overlying skin lesion) were found to be more likely to be associated with AIDS in the review by Caponetti and colleagues.6 As in our patient, it is also typically a manifestation of more widely disseminated disease.7
Most of the osseous lytic lesions in AIDS patients are located in the axial skeleton. Radiological features of musculoskeletal Kaposi sarcoma are variable. As observed by Caponetti and colleagues, Kaposi sarcoma lesions can appear as a periosteal reaction, cortical erosions, osteolysis, or osseous destruction, with irregular-shaped cortical erosions being most typical.6 Despite their osteolytic features, Kaposi sarcoma lesions are often not visualized by conventional radiography.6 The preferred imaging for identification of lytic bone changes is CT (Figure 3). Magnetic resonance imaging can also help distinguish marrow abnormalities as well as adjacent soft tissues masses. Radiologically, Kaposi sarcoma osseous lesions have parallel features to bacillary angiomatosis, tuberculosis, or lymphoma.8 Therefore, biopsy of the lesion is essential in establishing the diagnosis of Kaposi sarcoma.
In theory, there should be clinical improvement in Kaposi sarcoma when immunity is restored. Cancers caused by the Epstein-Barr virus and Kaposi sarcoma-associated herpes virus may eventually also be preventable with vaccines.10
There is rarely bone involvement without the foreshadowing of a poor prognosis. Erroneous patient care may inevitably arise from Kaposi sarcoma in uncharacteristic sites. A differential of Kaposi sarcoma should be included if a patient with AIDS presents with osteolytic lesions on imaging. Biopsying the lesion cements the diagnosis and eliminates the possibility of mimicry conditions such as bacillary angiomatosis, benign vascular lesions, and angiosarcoma. As of today, a HAART regimen remains the standard initial care for patients with Kaposi sarcoma.
1. Radu O, Pantanowitz L. Kaposi sarcoma. Arch Pathol Lab Med. 2013;137:289-294.
2. Northfelt DW, Dezube BJ, Thommes JA, et al. Pegylated-liposomal doxorubicin versus doxorubicin, bleomycin, and vincristine in the treatment of AIDS-related Kaposi sarcoma: results of a randomized phase III clinical trial. J Clin Oncol. 1998;16(7):2445-2451.
3. Restrepo CS, Martinez S, Lemos JA, et al. Imaging manifestations of Kaposi sarcoma. RadioGraphics. 2006;26:1169-1185.
4. Chang Y, Cesarman E, Pessin MS, et al. Identification of herpes virus-like DNA sequences in AIDS-associated Kaposi sarcoma. Science. 1994;266:1865-1869.
5. Beral V, Peterman TA, Berkelman RL, Jaffe HW. Kaposi sarcoma among persons with AIDS: a sexually transmitted infection? Lancet. 1990;335:123-128.
6. Caponetti G, Dezube BJ, Restrepo CS, Pantanowitz I. Kaposi sarcoma of the musculoskeletal system: a review of 66 patients. Cancer. 2007;109(6):1040-1052.
7. Krishna G, Chitkara RK. Osseous Kaposi sarcoma. JAMA. 2003;286(9):1106.
8. Thanos L, Mylona S, Kalioras V, Pomoni M, Batakis N. Osseous Kaposi sarcoma in an HIV-positive patient. Skeletal Radiol. 2004;33(4):241-243.
9. Guiholt A, Dupin N, Marcelin AG, et al. Low T-cell response to human herpesvirus 8 in patients with AIDS-related and classic Kaposi sarcoma. J Infect Dis. 2006;194(8):1078-1088.
10. Gopal S, Achenbach CJ, Yanik EL, Dither DP, Eron JJ, Engels EA. Moving forward in HIV-associated cancer. J Clin Oncol. 2014;32(9):876-880.
1. Radu O, Pantanowitz L. Kaposi sarcoma. Arch Pathol Lab Med. 2013;137:289-294.
2. Northfelt DW, Dezube BJ, Thommes JA, et al. Pegylated-liposomal doxorubicin versus doxorubicin, bleomycin, and vincristine in the treatment of AIDS-related Kaposi sarcoma: results of a randomized phase III clinical trial. J Clin Oncol. 1998;16(7):2445-2451.
3. Restrepo CS, Martinez S, Lemos JA, et al. Imaging manifestations of Kaposi sarcoma. RadioGraphics. 2006;26:1169-1185.
4. Chang Y, Cesarman E, Pessin MS, et al. Identification of herpes virus-like DNA sequences in AIDS-associated Kaposi sarcoma. Science. 1994;266:1865-1869.
5. Beral V, Peterman TA, Berkelman RL, Jaffe HW. Kaposi sarcoma among persons with AIDS: a sexually transmitted infection? Lancet. 1990;335:123-128.
6. Caponetti G, Dezube BJ, Restrepo CS, Pantanowitz I. Kaposi sarcoma of the musculoskeletal system: a review of 66 patients. Cancer. 2007;109(6):1040-1052.
7. Krishna G, Chitkara RK. Osseous Kaposi sarcoma. JAMA. 2003;286(9):1106.
8. Thanos L, Mylona S, Kalioras V, Pomoni M, Batakis N. Osseous Kaposi sarcoma in an HIV-positive patient. Skeletal Radiol. 2004;33(4):241-243.
9. Guiholt A, Dupin N, Marcelin AG, et al. Low T-cell response to human herpesvirus 8 in patients with AIDS-related and classic Kaposi sarcoma. J Infect Dis. 2006;194(8):1078-1088.
10. Gopal S, Achenbach CJ, Yanik EL, Dither DP, Eron JJ, Engels EA. Moving forward in HIV-associated cancer. J Clin Oncol. 2014;32(9):876-880.
T-cell product improves outcomes of haplo-HSCT
MADRID—Updated results of a phase 1/2 study suggest the T-cell product BPX-501 lowers the risks associated with haploidentical hematopoietic stem cell transplant (haplo-HSCT).
In this ongoing study, researchers are testing BPX-501 in pediatric patients undergoing haplo-HSCT to treat a range of hematologic disorders.
Patients treated thus far have experienced rapid engraftment and early hospital discharge, a low rate of acute graft-versus-host disease (GHVD), no extensive chronic GVHD, and a low rate of transplant-related mortality at 180 days.
“The combination of haploidentical transplantation and BPX-501 infusion is an effective strategy for children in need of an allograft lacking a compatible donor,” said study investigator Mattia Algeri, MD, of Ospedale Pediatrico Bambino Gesù in Rome, Italy.
Dr Algeri presented these results during the Presidential Symposium at the 22nd Congress of the European Hematology Association (EHA) as abstract S146.
The research was sponsored by Bellicum Pharmaceuticals, Inc., the company developing BPX-501.
About BPX-501
BPX-501 consists of genetically modified donor T cells incorporating the CaspaCIDe safety switch, which is designed to eliminate the T cells in the event of toxicity.
Rimiducid is used to activate the CaspaCIDe safety switch, which consists of the CID-binding domain coupled to the signaling domain of caspase-9, an enzyme that is part of the apoptotic pathway.
The goal of this therapy is to allow physicians to more safely perform haplo-HSCTs.
Patients
Dr Algeri and his colleagues have tested BPX-501 in 98 pediatric patients treated at centers in Europe and the US.
Fifty-nine patients had non-malignant conditions, including primary immune deficiency (n=26), thalassemia major (n=8), sickle cell disease (n=5), Diamond-Blackfan anemia (n=2), Swachman-Diamond syndrome (n=1), Fanconi anemia (n=9), hemophagocytic lymphohistiocytosis (n=6), aplastic anemia (n=1), and osteoporosis (n=1).
Thirty-nine patients had malignant conditions, including acute lymphoblastic leukemia (ALL, n=21), acute myeloid leukemia (AML, n=14), myelodysplastic syndromes (MDS, n=3), and non-Hodgkin lymphoma (NHL, n=1).
The patients received BPX-501 after an alpha/beta T-cell-depleted haplo-HSCT. All patients had at least 6 months of follow-up.
Overall results
Ninety-five percent of the patients engrafted (93/98), and the researchers said they observed rapid recovery of T cells, B cells, and immunoglobulins.
At 180 days, the incidence of transplant-related mortality was 5%, and there were no cases of post-transplant lymphoproliferative disorder.
The cumulative incidence of grade 2-4 acute GVHD was 14%. For patients with at least 1 year of follow-up, the cumulative incidence of chronic GVHD at 1 year was 3%.
Eleven patients received rimiducid—10 who had uncontrollable acute GVHD and 1 who developed late acute GVHD. In all of these patients, GVHD resolved and has not recurred.
There were no adverse events associated with BPX-501 or rimiducid.
European cohort
Dr Algeri presented more detailed data on the 61 patients treated at centers in Europe.
Fifteen of these patients had ALL, 10 had AML, 16 had primary immune deficiency, 7 had thalassemia major, 1 had sickle cell disease, 2 had Diamond-Blackfan anemia, 5 had Fanconi anemia, 4 had hemophagocytic lymphohistiocytosis, and 1 had osteoporosis.
Their median age was 4.8 (range, 0.25-17), and 56% were male. The patients received busulfan-based conditioning (41%), total body irradiation (36%), treosulfan-based conditioning (18%), and other conditioning (5%).
Ninety-five percent of the patients had a parent donor, and the other 5% had a sibling donor. The median donor age was 36 (range, 19-50).
The patients’ median time to neutrophil recovery was 15 days (range, 9-75), and their median time to platelet recovery was 10 days (range, 4-64). Their median time to discharge was 25 days (range, 14-122).
The cumulative incidence of acute grade 2-4 GVHD was 9.9%, and the cumulative incidence of acute grade 3-4 GVHD was 3.3%.
There were no cases of transplant-related mortality at 180 days and no cases of extensive chronic GVHD.
“These preliminary data compare favorably to previously published data on matched, unrelated donor transplantation,” Dr Algeri said. “And for this reason, an observational matched, unrelated donor study is being initiated to enable comparison of the safety and efficacy of haploidentical transplantation and BPX-501 infusion to the standard of care for patients without a matched sibling donor.” ![]()
MADRID—Updated results of a phase 1/2 study suggest the T-cell product BPX-501 lowers the risks associated with haploidentical hematopoietic stem cell transplant (haplo-HSCT).
In this ongoing study, researchers are testing BPX-501 in pediatric patients undergoing haplo-HSCT to treat a range of hematologic disorders.
Patients treated thus far have experienced rapid engraftment and early hospital discharge, a low rate of acute graft-versus-host disease (GHVD), no extensive chronic GVHD, and a low rate of transplant-related mortality at 180 days.
“The combination of haploidentical transplantation and BPX-501 infusion is an effective strategy for children in need of an allograft lacking a compatible donor,” said study investigator Mattia Algeri, MD, of Ospedale Pediatrico Bambino Gesù in Rome, Italy.
Dr Algeri presented these results during the Presidential Symposium at the 22nd Congress of the European Hematology Association (EHA) as abstract S146.
The research was sponsored by Bellicum Pharmaceuticals, Inc., the company developing BPX-501.
About BPX-501
BPX-501 consists of genetically modified donor T cells incorporating the CaspaCIDe safety switch, which is designed to eliminate the T cells in the event of toxicity.
Rimiducid is used to activate the CaspaCIDe safety switch, which consists of the CID-binding domain coupled to the signaling domain of caspase-9, an enzyme that is part of the apoptotic pathway.
The goal of this therapy is to allow physicians to more safely perform haplo-HSCTs.
Patients
Dr Algeri and his colleagues have tested BPX-501 in 98 pediatric patients treated at centers in Europe and the US.
Fifty-nine patients had non-malignant conditions, including primary immune deficiency (n=26), thalassemia major (n=8), sickle cell disease (n=5), Diamond-Blackfan anemia (n=2), Swachman-Diamond syndrome (n=1), Fanconi anemia (n=9), hemophagocytic lymphohistiocytosis (n=6), aplastic anemia (n=1), and osteoporosis (n=1).
Thirty-nine patients had malignant conditions, including acute lymphoblastic leukemia (ALL, n=21), acute myeloid leukemia (AML, n=14), myelodysplastic syndromes (MDS, n=3), and non-Hodgkin lymphoma (NHL, n=1).
The patients received BPX-501 after an alpha/beta T-cell-depleted haplo-HSCT. All patients had at least 6 months of follow-up.
Overall results
Ninety-five percent of the patients engrafted (93/98), and the researchers said they observed rapid recovery of T cells, B cells, and immunoglobulins.
At 180 days, the incidence of transplant-related mortality was 5%, and there were no cases of post-transplant lymphoproliferative disorder.
The cumulative incidence of grade 2-4 acute GVHD was 14%. For patients with at least 1 year of follow-up, the cumulative incidence of chronic GVHD at 1 year was 3%.
Eleven patients received rimiducid—10 who had uncontrollable acute GVHD and 1 who developed late acute GVHD. In all of these patients, GVHD resolved and has not recurred.
There were no adverse events associated with BPX-501 or rimiducid.
European cohort
Dr Algeri presented more detailed data on the 61 patients treated at centers in Europe.
Fifteen of these patients had ALL, 10 had AML, 16 had primary immune deficiency, 7 had thalassemia major, 1 had sickle cell disease, 2 had Diamond-Blackfan anemia, 5 had Fanconi anemia, 4 had hemophagocytic lymphohistiocytosis, and 1 had osteoporosis.
Their median age was 4.8 (range, 0.25-17), and 56% were male. The patients received busulfan-based conditioning (41%), total body irradiation (36%), treosulfan-based conditioning (18%), and other conditioning (5%).
Ninety-five percent of the patients had a parent donor, and the other 5% had a sibling donor. The median donor age was 36 (range, 19-50).
The patients’ median time to neutrophil recovery was 15 days (range, 9-75), and their median time to platelet recovery was 10 days (range, 4-64). Their median time to discharge was 25 days (range, 14-122).
The cumulative incidence of acute grade 2-4 GVHD was 9.9%, and the cumulative incidence of acute grade 3-4 GVHD was 3.3%.
There were no cases of transplant-related mortality at 180 days and no cases of extensive chronic GVHD.
“These preliminary data compare favorably to previously published data on matched, unrelated donor transplantation,” Dr Algeri said. “And for this reason, an observational matched, unrelated donor study is being initiated to enable comparison of the safety and efficacy of haploidentical transplantation and BPX-501 infusion to the standard of care for patients without a matched sibling donor.” ![]()
MADRID—Updated results of a phase 1/2 study suggest the T-cell product BPX-501 lowers the risks associated with haploidentical hematopoietic stem cell transplant (haplo-HSCT).
In this ongoing study, researchers are testing BPX-501 in pediatric patients undergoing haplo-HSCT to treat a range of hematologic disorders.
Patients treated thus far have experienced rapid engraftment and early hospital discharge, a low rate of acute graft-versus-host disease (GHVD), no extensive chronic GVHD, and a low rate of transplant-related mortality at 180 days.
“The combination of haploidentical transplantation and BPX-501 infusion is an effective strategy for children in need of an allograft lacking a compatible donor,” said study investigator Mattia Algeri, MD, of Ospedale Pediatrico Bambino Gesù in Rome, Italy.
Dr Algeri presented these results during the Presidential Symposium at the 22nd Congress of the European Hematology Association (EHA) as abstract S146.
The research was sponsored by Bellicum Pharmaceuticals, Inc., the company developing BPX-501.
About BPX-501
BPX-501 consists of genetically modified donor T cells incorporating the CaspaCIDe safety switch, which is designed to eliminate the T cells in the event of toxicity.
Rimiducid is used to activate the CaspaCIDe safety switch, which consists of the CID-binding domain coupled to the signaling domain of caspase-9, an enzyme that is part of the apoptotic pathway.
The goal of this therapy is to allow physicians to more safely perform haplo-HSCTs.
Patients
Dr Algeri and his colleagues have tested BPX-501 in 98 pediatric patients treated at centers in Europe and the US.
Fifty-nine patients had non-malignant conditions, including primary immune deficiency (n=26), thalassemia major (n=8), sickle cell disease (n=5), Diamond-Blackfan anemia (n=2), Swachman-Diamond syndrome (n=1), Fanconi anemia (n=9), hemophagocytic lymphohistiocytosis (n=6), aplastic anemia (n=1), and osteoporosis (n=1).
Thirty-nine patients had malignant conditions, including acute lymphoblastic leukemia (ALL, n=21), acute myeloid leukemia (AML, n=14), myelodysplastic syndromes (MDS, n=3), and non-Hodgkin lymphoma (NHL, n=1).
The patients received BPX-501 after an alpha/beta T-cell-depleted haplo-HSCT. All patients had at least 6 months of follow-up.
Overall results
Ninety-five percent of the patients engrafted (93/98), and the researchers said they observed rapid recovery of T cells, B cells, and immunoglobulins.
At 180 days, the incidence of transplant-related mortality was 5%, and there were no cases of post-transplant lymphoproliferative disorder.
The cumulative incidence of grade 2-4 acute GVHD was 14%. For patients with at least 1 year of follow-up, the cumulative incidence of chronic GVHD at 1 year was 3%.
Eleven patients received rimiducid—10 who had uncontrollable acute GVHD and 1 who developed late acute GVHD. In all of these patients, GVHD resolved and has not recurred.
There were no adverse events associated with BPX-501 or rimiducid.
European cohort
Dr Algeri presented more detailed data on the 61 patients treated at centers in Europe.
Fifteen of these patients had ALL, 10 had AML, 16 had primary immune deficiency, 7 had thalassemia major, 1 had sickle cell disease, 2 had Diamond-Blackfan anemia, 5 had Fanconi anemia, 4 had hemophagocytic lymphohistiocytosis, and 1 had osteoporosis.
Their median age was 4.8 (range, 0.25-17), and 56% were male. The patients received busulfan-based conditioning (41%), total body irradiation (36%), treosulfan-based conditioning (18%), and other conditioning (5%).
Ninety-five percent of the patients had a parent donor, and the other 5% had a sibling donor. The median donor age was 36 (range, 19-50).
The patients’ median time to neutrophil recovery was 15 days (range, 9-75), and their median time to platelet recovery was 10 days (range, 4-64). Their median time to discharge was 25 days (range, 14-122).
The cumulative incidence of acute grade 2-4 GVHD was 9.9%, and the cumulative incidence of acute grade 3-4 GVHD was 3.3%.
There were no cases of transplant-related mortality at 180 days and no cases of extensive chronic GVHD.
“These preliminary data compare favorably to previously published data on matched, unrelated donor transplantation,” Dr Algeri said. “And for this reason, an observational matched, unrelated donor study is being initiated to enable comparison of the safety and efficacy of haploidentical transplantation and BPX-501 infusion to the standard of care for patients without a matched sibling donor.” ![]()
Single-dose NEPA found non-inferior to aprepitant/granisetron
WASHINGTON, DC—In a head-to-head study comparing a single-dose oral antiemetic to a 3-day oral regimen, the single dose has shown itself to be non-inferior to the multi-day regimen in preventing chemotherapy-induced nausea and vomiting (CINV).
The investigators evaluated netupitant/palonosetron (NEPA) against aprepitant/granisetron (APR/GRAN) in patients on highly emetogenic chemotherapy.
They found the data suggest “that NEPA, in a single dose, had equivalent efficacy to a 3-day oral aprepitant/granisetron regimen,” according to the lead investigator and abstract presenter.
Li Zhang, MD, of Sun Yat-sen University Cancer Center in Guangzhou, China, presented the data at the Multinational Association of Supportive Care in Cancer/International Society of Oral Oncology (MASCC/ISOO) Congress (abstract PS049, pp S55 – S56).
NEPA is a combination of the selective NK1RA netupitant (300 mg) and the clinically and pharmacologically active 5-HT3RA, palonosetron (0.5 mg) for the prevention of CINV.
Oral palonosetron prevents nausea and vomiting during the acute phase of treatment.
Netupitant prevents nausea and vomiting during both the acute and delayed phase after cancer chemotherapy.
It is formulated into a single oral capsule.
Study design
The study was a phase 3 randomized, double-blind, double-dummy study conducted in 828 chemotherapy-naïve Asian patients receiving cisplatin-based highly emetogenic chemotherapy (HEC) agents.
Patients received a single oral dose of NEPA on day 1 or a 3-day oral APR/GRAN regimen (days 1-3).
All patients received oral dexamethasone on days 1-4.
The primary efficacy endpoint was complete response (CR), defined as no emesis or rescue medication needed during the overall (0-120 hour) phase.
The investigators defined non-inferiority to be the lower limit of the two-sided 95% confidence interval greater than the non-inferiority margin set at ̶ 10%.
Secondary endpoints included no emesis, no rescue medication, and no significant nausea (NSN), defined as <25 mm on 100 mm visual analog scale (VAS).
Results
The baseline demographics were comparable between the NEPA (n=413) and APR/GRAN (n=416) arms: 71% of the patients were male, a mean age of 55 years, and a little more than half were ECOG performance status 1.
The most common cancer types were lung and head and neck cancer.
Patients had received a median cisplatin dose of 73 and 72 mg/m2 in the NEPA and APR/GRAN arms, respectively.
Within the first 24 hours (acute phase), NEPA was non-inferior to APR/GRAN. NEPA had a CR rate of 84.5% and APR/GRAN had a CR rate of 87.0%. The risk difference between the 2 agents was -2.5% (range, -7.2%, 2.3%).
In the delayed phase (25-120 hours), NEPA had a CR rate of 77.9% and APR/GRAN, 74.3%. The risk difference was 3.7% (range, -2.1%, 9.5%).
Overall, for both phases, the CR rate was 73.8% for NEPA and 72.4% for APR/GRAN. The risk difference was 1.5% (range, -4.5%, 7.5%).
Dr Zhang pointed out that although the overall CR rates were similar, the daily rates of patients experiencing CINV remained in the range of 13% - 15% for patients in the APR/GRAN arm.
However, daily rates of CINV for patients receiving NEPA declined from 16% to 8% over the 5 days. The investigators believe this suggests a benefit for delayed CINV.
Regarding secondary endpoints, significantly more patients receiving NEPA did not require rescue medication in the delayed phase and overall than patients in the APR/GRAN arm.
Treatment-emergent adverse events (TEAEs) were comparable between the arms—58.1% in the NEPA arm and 57.5% in the APR/GRAN arm, as were serious TEAS, at 4.5% and 4.6% for NEPA and APR/GRAN, respectively. And the no emesis and no significant nausea rates favored NEPA.
The most common treatment-emergent adverse events occurring in 2% or more of the patients in both arms were constipation and hiccups.
Two serious treatment-related adverse events occurred in each arm, 1 leading to discontinuation in the NEPA arm.
The investigators concluded that NEPA, as a convenient capsule administered once per cycle, is at least as effective as the 3-day regimen of APR/GRAN in patients receiving HEC.
NEPA (Akynzeo®) is approved by the US Food and Drug Administration and marketed globally by Helsinn, Lugano, Switzerland, the sponsor of the trial.
For the full US prescribing information, see the package insert.
WASHINGTON, DC—In a head-to-head study comparing a single-dose oral antiemetic to a 3-day oral regimen, the single dose has shown itself to be non-inferior to the multi-day regimen in preventing chemotherapy-induced nausea and vomiting (CINV).
The investigators evaluated netupitant/palonosetron (NEPA) against aprepitant/granisetron (APR/GRAN) in patients on highly emetogenic chemotherapy.
They found the data suggest “that NEPA, in a single dose, had equivalent efficacy to a 3-day oral aprepitant/granisetron regimen,” according to the lead investigator and abstract presenter.
Li Zhang, MD, of Sun Yat-sen University Cancer Center in Guangzhou, China, presented the data at the Multinational Association of Supportive Care in Cancer/International Society of Oral Oncology (MASCC/ISOO) Congress (abstract PS049, pp S55 – S56).
NEPA is a combination of the selective NK1RA netupitant (300 mg) and the clinically and pharmacologically active 5-HT3RA, palonosetron (0.5 mg) for the prevention of CINV.
Oral palonosetron prevents nausea and vomiting during the acute phase of treatment.
Netupitant prevents nausea and vomiting during both the acute and delayed phase after cancer chemotherapy.
It is formulated into a single oral capsule.
Study design
The study was a phase 3 randomized, double-blind, double-dummy study conducted in 828 chemotherapy-naïve Asian patients receiving cisplatin-based highly emetogenic chemotherapy (HEC) agents.
Patients received a single oral dose of NEPA on day 1 or a 3-day oral APR/GRAN regimen (days 1-3).
All patients received oral dexamethasone on days 1-4.
The primary efficacy endpoint was complete response (CR), defined as no emesis or rescue medication needed during the overall (0-120 hour) phase.
The investigators defined non-inferiority to be the lower limit of the two-sided 95% confidence interval greater than the non-inferiority margin set at ̶ 10%.
Secondary endpoints included no emesis, no rescue medication, and no significant nausea (NSN), defined as <25 mm on 100 mm visual analog scale (VAS).
Results
The baseline demographics were comparable between the NEPA (n=413) and APR/GRAN (n=416) arms: 71% of the patients were male, a mean age of 55 years, and a little more than half were ECOG performance status 1.
The most common cancer types were lung and head and neck cancer.
Patients had received a median cisplatin dose of 73 and 72 mg/m2 in the NEPA and APR/GRAN arms, respectively.
Within the first 24 hours (acute phase), NEPA was non-inferior to APR/GRAN. NEPA had a CR rate of 84.5% and APR/GRAN had a CR rate of 87.0%. The risk difference between the 2 agents was -2.5% (range, -7.2%, 2.3%).
In the delayed phase (25-120 hours), NEPA had a CR rate of 77.9% and APR/GRAN, 74.3%. The risk difference was 3.7% (range, -2.1%, 9.5%).
Overall, for both phases, the CR rate was 73.8% for NEPA and 72.4% for APR/GRAN. The risk difference was 1.5% (range, -4.5%, 7.5%).
Dr Zhang pointed out that although the overall CR rates were similar, the daily rates of patients experiencing CINV remained in the range of 13% - 15% for patients in the APR/GRAN arm.
However, daily rates of CINV for patients receiving NEPA declined from 16% to 8% over the 5 days. The investigators believe this suggests a benefit for delayed CINV.
Regarding secondary endpoints, significantly more patients receiving NEPA did not require rescue medication in the delayed phase and overall than patients in the APR/GRAN arm.
Treatment-emergent adverse events (TEAEs) were comparable between the arms—58.1% in the NEPA arm and 57.5% in the APR/GRAN arm, as were serious TEAS, at 4.5% and 4.6% for NEPA and APR/GRAN, respectively. And the no emesis and no significant nausea rates favored NEPA.
The most common treatment-emergent adverse events occurring in 2% or more of the patients in both arms were constipation and hiccups.
Two serious treatment-related adverse events occurred in each arm, 1 leading to discontinuation in the NEPA arm.
The investigators concluded that NEPA, as a convenient capsule administered once per cycle, is at least as effective as the 3-day regimen of APR/GRAN in patients receiving HEC.
NEPA (Akynzeo®) is approved by the US Food and Drug Administration and marketed globally by Helsinn, Lugano, Switzerland, the sponsor of the trial.
For the full US prescribing information, see the package insert.
WASHINGTON, DC—In a head-to-head study comparing a single-dose oral antiemetic to a 3-day oral regimen, the single dose has shown itself to be non-inferior to the multi-day regimen in preventing chemotherapy-induced nausea and vomiting (CINV).
The investigators evaluated netupitant/palonosetron (NEPA) against aprepitant/granisetron (APR/GRAN) in patients on highly emetogenic chemotherapy.
They found the data suggest “that NEPA, in a single dose, had equivalent efficacy to a 3-day oral aprepitant/granisetron regimen,” according to the lead investigator and abstract presenter.
Li Zhang, MD, of Sun Yat-sen University Cancer Center in Guangzhou, China, presented the data at the Multinational Association of Supportive Care in Cancer/International Society of Oral Oncology (MASCC/ISOO) Congress (abstract PS049, pp S55 – S56).
NEPA is a combination of the selective NK1RA netupitant (300 mg) and the clinically and pharmacologically active 5-HT3RA, palonosetron (0.5 mg) for the prevention of CINV.
Oral palonosetron prevents nausea and vomiting during the acute phase of treatment.
Netupitant prevents nausea and vomiting during both the acute and delayed phase after cancer chemotherapy.
It is formulated into a single oral capsule.
Study design
The study was a phase 3 randomized, double-blind, double-dummy study conducted in 828 chemotherapy-naïve Asian patients receiving cisplatin-based highly emetogenic chemotherapy (HEC) agents.
Patients received a single oral dose of NEPA on day 1 or a 3-day oral APR/GRAN regimen (days 1-3).
All patients received oral dexamethasone on days 1-4.
The primary efficacy endpoint was complete response (CR), defined as no emesis or rescue medication needed during the overall (0-120 hour) phase.
The investigators defined non-inferiority to be the lower limit of the two-sided 95% confidence interval greater than the non-inferiority margin set at ̶ 10%.
Secondary endpoints included no emesis, no rescue medication, and no significant nausea (NSN), defined as <25 mm on 100 mm visual analog scale (VAS).
Results
The baseline demographics were comparable between the NEPA (n=413) and APR/GRAN (n=416) arms: 71% of the patients were male, a mean age of 55 years, and a little more than half were ECOG performance status 1.
The most common cancer types were lung and head and neck cancer.
Patients had received a median cisplatin dose of 73 and 72 mg/m2 in the NEPA and APR/GRAN arms, respectively.
Within the first 24 hours (acute phase), NEPA was non-inferior to APR/GRAN. NEPA had a CR rate of 84.5% and APR/GRAN had a CR rate of 87.0%. The risk difference between the 2 agents was -2.5% (range, -7.2%, 2.3%).
In the delayed phase (25-120 hours), NEPA had a CR rate of 77.9% and APR/GRAN, 74.3%. The risk difference was 3.7% (range, -2.1%, 9.5%).
Overall, for both phases, the CR rate was 73.8% for NEPA and 72.4% for APR/GRAN. The risk difference was 1.5% (range, -4.5%, 7.5%).
Dr Zhang pointed out that although the overall CR rates were similar, the daily rates of patients experiencing CINV remained in the range of 13% - 15% for patients in the APR/GRAN arm.
However, daily rates of CINV for patients receiving NEPA declined from 16% to 8% over the 5 days. The investigators believe this suggests a benefit for delayed CINV.
Regarding secondary endpoints, significantly more patients receiving NEPA did not require rescue medication in the delayed phase and overall than patients in the APR/GRAN arm.
Treatment-emergent adverse events (TEAEs) were comparable between the arms—58.1% in the NEPA arm and 57.5% in the APR/GRAN arm, as were serious TEAS, at 4.5% and 4.6% for NEPA and APR/GRAN, respectively. And the no emesis and no significant nausea rates favored NEPA.
The most common treatment-emergent adverse events occurring in 2% or more of the patients in both arms were constipation and hiccups.
Two serious treatment-related adverse events occurred in each arm, 1 leading to discontinuation in the NEPA arm.
The investigators concluded that NEPA, as a convenient capsule administered once per cycle, is at least as effective as the 3-day regimen of APR/GRAN in patients receiving HEC.
NEPA (Akynzeo®) is approved by the US Food and Drug Administration and marketed globally by Helsinn, Lugano, Switzerland, the sponsor of the trial.
For the full US prescribing information, see the package insert.
Lower dose of rivaroxaban receives priority review
The US Food and Drug Administration (FDA) has accepted for priority review a supplemental new drug application (sNDA) for rivaroxaban (XARELTO®).
The sNDA is for a 10 mg once-daily dose of rivaroxaban to reduce the risk of venous thromboembolism (VTE) in patients who have received at least 6 months of standard anticoagulant therapy.
The FDA grants priority review to applications for products that may provide significant improvements in the treatment, diagnosis, or prevention of serious conditions.
The agency’s goal is to take action on a priority review application within 6 months of receiving it, rather than the standard 10 months.
The FDA plans to make a decision on the rivaroxaban sNDA by October 28, 2017.
The sNDA is supported by data from the EINSTEIN CHOICE study. Patients enrolled in this study had confirmed VTE and were treated initially with standard anticoagulant therapy for 6 to 12 months.
During EINSTEIN CHOICE, 3365 patients received rivaroxaban at 10 mg, rivaroxaban at 20 mg, or aspirin at 100 mg once daily for up to 12 months of extended treatment.
Both rivaroxaban doses were superior to aspirin in preventing fatal or non-fatal recurrent VTE, the study’s primary efficacy endpoint.
The rate of recurrent VTE was 1.2% in the 10 mg rivaroxaban arm (hazard ratio [HR]=0.26; 95% CI, 0.14 to 0.47; P<0.001), 1.5% in the 20 mg rivaroxaban arm (HR=0.34; 95% CI, 0.20 to 0.59; P<0.001), and 4.4% in the aspirin arm. Fatal VTE occurred in 0%, 0.2%, and 0.2% of patients, respectively.
The primary safety endpoint was major bleeding as defined by the International Society on Thrombosis and Haemostasis.
The rate of major bleeding was 0.4% for the 10 mg rivaroxaban group (HR=1.64; 95% CI, 0.39 to 6.84; P=0.50), 0.5% for the 20 mg rivaroxaban group (HR=2.01; 95% CI, 0.50 to 8.04; P=0.32), and 0.3% for the aspirin group. ![]()
The US Food and Drug Administration (FDA) has accepted for priority review a supplemental new drug application (sNDA) for rivaroxaban (XARELTO®).
The sNDA is for a 10 mg once-daily dose of rivaroxaban to reduce the risk of venous thromboembolism (VTE) in patients who have received at least 6 months of standard anticoagulant therapy.
The FDA grants priority review to applications for products that may provide significant improvements in the treatment, diagnosis, or prevention of serious conditions.
The agency’s goal is to take action on a priority review application within 6 months of receiving it, rather than the standard 10 months.
The FDA plans to make a decision on the rivaroxaban sNDA by October 28, 2017.
The sNDA is supported by data from the EINSTEIN CHOICE study. Patients enrolled in this study had confirmed VTE and were treated initially with standard anticoagulant therapy for 6 to 12 months.
During EINSTEIN CHOICE, 3365 patients received rivaroxaban at 10 mg, rivaroxaban at 20 mg, or aspirin at 100 mg once daily for up to 12 months of extended treatment.
Both rivaroxaban doses were superior to aspirin in preventing fatal or non-fatal recurrent VTE, the study’s primary efficacy endpoint.
The rate of recurrent VTE was 1.2% in the 10 mg rivaroxaban arm (hazard ratio [HR]=0.26; 95% CI, 0.14 to 0.47; P<0.001), 1.5% in the 20 mg rivaroxaban arm (HR=0.34; 95% CI, 0.20 to 0.59; P<0.001), and 4.4% in the aspirin arm. Fatal VTE occurred in 0%, 0.2%, and 0.2% of patients, respectively.
The primary safety endpoint was major bleeding as defined by the International Society on Thrombosis and Haemostasis.
The rate of major bleeding was 0.4% for the 10 mg rivaroxaban group (HR=1.64; 95% CI, 0.39 to 6.84; P=0.50), 0.5% for the 20 mg rivaroxaban group (HR=2.01; 95% CI, 0.50 to 8.04; P=0.32), and 0.3% for the aspirin group. ![]()
The US Food and Drug Administration (FDA) has accepted for priority review a supplemental new drug application (sNDA) for rivaroxaban (XARELTO®).
The sNDA is for a 10 mg once-daily dose of rivaroxaban to reduce the risk of venous thromboembolism (VTE) in patients who have received at least 6 months of standard anticoagulant therapy.
The FDA grants priority review to applications for products that may provide significant improvements in the treatment, diagnosis, or prevention of serious conditions.
The agency’s goal is to take action on a priority review application within 6 months of receiving it, rather than the standard 10 months.
The FDA plans to make a decision on the rivaroxaban sNDA by October 28, 2017.
The sNDA is supported by data from the EINSTEIN CHOICE study. Patients enrolled in this study had confirmed VTE and were treated initially with standard anticoagulant therapy for 6 to 12 months.
During EINSTEIN CHOICE, 3365 patients received rivaroxaban at 10 mg, rivaroxaban at 20 mg, or aspirin at 100 mg once daily for up to 12 months of extended treatment.
Both rivaroxaban doses were superior to aspirin in preventing fatal or non-fatal recurrent VTE, the study’s primary efficacy endpoint.
The rate of recurrent VTE was 1.2% in the 10 mg rivaroxaban arm (hazard ratio [HR]=0.26; 95% CI, 0.14 to 0.47; P<0.001), 1.5% in the 20 mg rivaroxaban arm (HR=0.34; 95% CI, 0.20 to 0.59; P<0.001), and 4.4% in the aspirin arm. Fatal VTE occurred in 0%, 0.2%, and 0.2% of patients, respectively.
The primary safety endpoint was major bleeding as defined by the International Society on Thrombosis and Haemostasis.
The rate of major bleeding was 0.4% for the 10 mg rivaroxaban group (HR=1.64; 95% CI, 0.39 to 6.84; P=0.50), 0.5% for the 20 mg rivaroxaban group (HR=2.01; 95% CI, 0.50 to 8.04; P=0.32), and 0.3% for the aspirin group. ![]()
Persistent rash on extremities
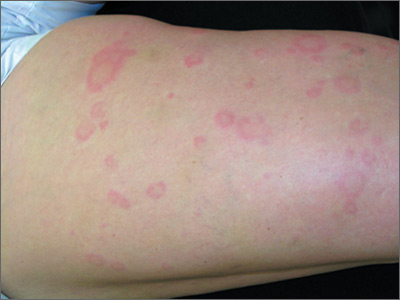
Given the history that the rash persisted for longer than 3 months, the FP made the diagnosis of chronic urticaria. The cause of chronic urticaria (>6 weeks duration) is determined in less than 20% of cases. Because no cause was found in this case, the patient’s urticaria was determined to be chronic idiopathic urticaria.
Chronic urticaria is twice as common in women than in men. Urticaria is a dynamic process in which new wheals evolve as old ones resolve. The wheals result from localized capillary vasodilation, followed by transudation of protein-rich fluid into the surrounding skin. The wheals resolve when the fluid is slowly reabsorbed. Patients may benefit from avoiding potential urticarial precipitants such as aspirin, nonsteroidal anti-inflammatory drugs, opiates, and alcohol.
Second-generation H1 antihistamines, such as cetirizine, should be prescribed as first-line treatment for chronic urticaria. Increasing the dose of cetirizine from 10 mg/d to 20 mg/d produces a significant improvement in the severity of wheals and itching in urticaria refractory to the standard doses of antihistamines. The National Institute for Health and Care Excellence (NICE) notes in its 2014 evidence summary that international guidelines suggest that non-sedating antihistamines may be used at up to quadruple the manufacturers’ recommended dosages before changing to an alternative therapy. NICE recommends waiting up to 4 weeks to allow full effectiveness of the antihistamines before considering referral to a specialist.
The patient in this case was started on cetirizine 10 mg twice daily with written instructions to gradually increase up to 20 mg twice daily if the lower doses were not working. With cetirizine 20 mg twice daily, the chronic urticaria subsided and the patient was satisfied with the results.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Usatine R. Urticaria and angioedema. In: Usatine R, Smith M, Mayeaux EJ, et al, eds. Color Atlas of Family Medicine. 2nd ed. New York, NY: McGraw-Hill; 2013: 863-870.
To learn more about the Color Atlas of Family Medicine, see: www.amazon.com/Color-Family-Medicine-Richard-Usatine/dp/0071769641/
You can now get the second edition of the Color Atlas of Family Medicine as an app by clicking on this link: usatinemedia.com

Given the history that the rash persisted for longer than 3 months, the FP made the diagnosis of chronic urticaria. The cause of chronic urticaria (>6 weeks duration) is determined in less than 20% of cases. Because no cause was found in this case, the patient’s urticaria was determined to be chronic idiopathic urticaria.
Chronic urticaria is twice as common in women than in men. Urticaria is a dynamic process in which new wheals evolve as old ones resolve. The wheals result from localized capillary vasodilation, followed by transudation of protein-rich fluid into the surrounding skin. The wheals resolve when the fluid is slowly reabsorbed. Patients may benefit from avoiding potential urticarial precipitants such as aspirin, nonsteroidal anti-inflammatory drugs, opiates, and alcohol.
Second-generation H1 antihistamines, such as cetirizine, should be prescribed as first-line treatment for chronic urticaria. Increasing the dose of cetirizine from 10 mg/d to 20 mg/d produces a significant improvement in the severity of wheals and itching in urticaria refractory to the standard doses of antihistamines. The National Institute for Health and Care Excellence (NICE) notes in its 2014 evidence summary that international guidelines suggest that non-sedating antihistamines may be used at up to quadruple the manufacturers’ recommended dosages before changing to an alternative therapy. NICE recommends waiting up to 4 weeks to allow full effectiveness of the antihistamines before considering referral to a specialist.
The patient in this case was started on cetirizine 10 mg twice daily with written instructions to gradually increase up to 20 mg twice daily if the lower doses were not working. With cetirizine 20 mg twice daily, the chronic urticaria subsided and the patient was satisfied with the results.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Usatine R. Urticaria and angioedema. In: Usatine R, Smith M, Mayeaux EJ, et al, eds. Color Atlas of Family Medicine. 2nd ed. New York, NY: McGraw-Hill; 2013: 863-870.
To learn more about the Color Atlas of Family Medicine, see: www.amazon.com/Color-Family-Medicine-Richard-Usatine/dp/0071769641/
You can now get the second edition of the Color Atlas of Family Medicine as an app by clicking on this link: usatinemedia.com

Given the history that the rash persisted for longer than 3 months, the FP made the diagnosis of chronic urticaria. The cause of chronic urticaria (>6 weeks duration) is determined in less than 20% of cases. Because no cause was found in this case, the patient’s urticaria was determined to be chronic idiopathic urticaria.
Chronic urticaria is twice as common in women than in men. Urticaria is a dynamic process in which new wheals evolve as old ones resolve. The wheals result from localized capillary vasodilation, followed by transudation of protein-rich fluid into the surrounding skin. The wheals resolve when the fluid is slowly reabsorbed. Patients may benefit from avoiding potential urticarial precipitants such as aspirin, nonsteroidal anti-inflammatory drugs, opiates, and alcohol.
Second-generation H1 antihistamines, such as cetirizine, should be prescribed as first-line treatment for chronic urticaria. Increasing the dose of cetirizine from 10 mg/d to 20 mg/d produces a significant improvement in the severity of wheals and itching in urticaria refractory to the standard doses of antihistamines. The National Institute for Health and Care Excellence (NICE) notes in its 2014 evidence summary that international guidelines suggest that non-sedating antihistamines may be used at up to quadruple the manufacturers’ recommended dosages before changing to an alternative therapy. NICE recommends waiting up to 4 weeks to allow full effectiveness of the antihistamines before considering referral to a specialist.
The patient in this case was started on cetirizine 10 mg twice daily with written instructions to gradually increase up to 20 mg twice daily if the lower doses were not working. With cetirizine 20 mg twice daily, the chronic urticaria subsided and the patient was satisfied with the results.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Usatine R. Urticaria and angioedema. In: Usatine R, Smith M, Mayeaux EJ, et al, eds. Color Atlas of Family Medicine. 2nd ed. New York, NY: McGraw-Hill; 2013: 863-870.
To learn more about the Color Atlas of Family Medicine, see: www.amazon.com/Color-Family-Medicine-Richard-Usatine/dp/0071769641/
You can now get the second edition of the Color Atlas of Family Medicine as an app by clicking on this link: usatinemedia.com
A rare case of hypoglycemia induced by a classic gastrointestinal stromal tumor
Hypoglycemia, a frequently encountered medical emergency, is usually seen in patients with diabetes, most commonly as a result of iatrogenesis. However, it can also be encountered in nondiabetic patients. Various causes, such as pancreatic islet cell tumors producing insulin, primary or secondary adrenal insufficiency, advanced liver disease, pheochromocytoma and hypothyroidism, have been found to contribute to the condition in the nondiabetic population.1 In rare cases, an excessive production of insulin-like growth factor (IGF-2) – a condition known as nonislet cell tumor-induced hypoglycemia (NICTH) – has also been found to cause hypoglycemia. Hypoinsulinemic hypoglycemia, with low IGF-1 levels and an IGF-2-IgF1 ratio of greater than 10, is found to be suggestive of NICTH.
Case presentation and summary
An 81-year-old man with a history of diabetes mellitus, systolic heart failure, chronic kidney disease, and metastatic classical gastrointestinal spindle cell sarcoma presented to the emergency department with an acute change in mental status resulting from a new onset hypoglycemia. He was admitted, and during his hospital stay, he experienced severe hypoglycemic episodes with symptomatic presentations of diaphoresis on multiple occasions. A detailed history revealed that for diabetes, the patient had been on insulin for the first 12 years after his diagnosis, after which he was switched to metformin 500 mg twice daily for about 2 years, and as a satisfactory glycemic control was attained, eventually metformin had also been stopped 3 years prior to the current presentation.
The patient’s past medical records were obtained from the hospital at which he had been diagnosed gastrointestinal spindle cell sarcoma. Patient had not received treatment for the cancer as the disease was too widespread to be treated. The gastrointestinal spindle cell sarcoma, which had initially been surgically resected 7 years before the current presentation, had a recurrence 3 years later with abdominal and pulmonary metastasis, but no liver metastasis. No further intervention was carried out because the widely metastasized disease would not have benefited from any more surgical intervention and chemotherapy was not initiated because of the patient’s comorbid illnesses.
A blood sample drawn from the patient at the time of one hypoglycemic event, revealed low serum insulin <0.1 U/ml (normal, 2-19.6 U/ml); low C-peptide level, 0.59 ng/ml (0.8-3.85 ng/ml); low IGF-1, 16 ng/ml (5-4 ng/ml); and IGF-3, 0.9 ng/ml (2.2-4.5 ng/ml). IGF-2 levels were found to be markedly elevated at 945 ng/ml (47-350 ng/ml). The calculated IGF-2-IGF-1 ratio was 59.06 (normal, <10), suggesting NICTH as the etiology for the patient’s hypoglycemia.
The hypoglycemic episodes were initially treated with a continuous dextrose infusion followed by diazoxide treatment. However, diazoxide did not prevent his hypoglycemic episodes, so dexamethasone was considered as an alternative for his condition. The dexamethasone treatment resulted in the normalization of the patient’s serum glucose levels and resolution of his symptoms. The patient was discharged in a satisfactory state few days later and followed up thereafter. No recurrence of hypoglycemic episodes was found, and he was continued on dexamethasone therapy.
Discussion
Hypoglycemia due to NICTH is rare, with a prevalence of four times less than that of insulinoma.3 In most cases, NICTH occurs in patients with solid tumors of mesenchymal and epithelial origins such as hepatocellular carcinoma, gastric carcinoma or mesothelioma.4 In NICTH, the serum levels of insulin, C-peptide, and IGF-1 are usually decreased or undetectable. However, the circulating levels of total IGF2 may be increased, decreased, or normal. Concurrent normal to high morning cortisol and normal response to cosyntropin stimulation can rule out adrenal insufficiency and suggest NICTH. An IGF-2: IGF-1 ratio of >10 is considered to be clinically significant and highly suggestive of NICTH.5 Hypoglycemia in NICTH can be managed by administration of oral glucose, intravenous dextrose or glucagon. In some cases, diazoxide, a potent inhibitor of insulin secretion, has been found to be useful.6 Diazoxide directly inhibits the release of insulin through stimulation of adrenergic receptors and also has an extra pancreatic hyperglycemic effect, probably by inhibiting cyclic adenosine monophosphate phosphodiesterase, resulting in higher plasma levels of cyclic AMP and enhanced glycogenolysis.
Glucocorticoid therapy has been shown to suppress IGF-2 in a dose dependent manner and also by increasing gluconeogenesis.7 Surgical resection of the tumor whenever possible is the treatment of choice followed by radiotherapy and chemotherapy for inoperable disease and if successful, usually results in resolution of hypoglycemia. Imatinib, is the chemotherapeutic drug of choice for metastatic GIST, but many case reports have suggested worsening of hypoglycemia in advanced GIST with the use of the drug.8 The patient described in our report was not on any chemotherapy, hence hypoglycemia could not be attributed to it. On the basis of findings among 24 patients with GIST, Rikhof and colleagues have recommended monitoring plasma levels of pro-IGF-IIE to identify patients at high risk for developing hypoglycemia, especially those with progressive disease.9 Furthermore, over expression of IGF-2 as a predictor of potential relapse may be an area for potential research and further study.10
1. Marks V, Teale JD. Tumours producing hypoglycaemia. Diabetes Metab Rev. 1991;7:79-91.
2. Dutta P, Aggarwal A, Gogate Y, Nahar U, Shah VN, Singla M. Non-islet cell tumor-induced hypoglycemia: a report of five cases and brief review of the literature. Endocrinol Diabetes Metab Case Rep. 2013;2013:130046
3. de Groot JW, Rikhof B, van Doorn J, et al. Non-islet cell tumour-induced hypoglycaemia: a review of the literature including two new cases. Endocr Relat Cancer. 2007;14:979-93.
4. Fukuda I, Hizuka N, Ishikawa Y, et al. Clinical features of insulin-like growth factor II producing non-islet-cell tumor hypoglycemia
5. Marks V, Teale JD: Tumours producing hypoglycaemia. Endocr Relat Cancer. 1998;5:111-129.
6. Le Roith D. Tumor-induced hypoglycemia. N Engl J Med. 1999;341:757-758.
7. Teale JD, Marks V. Glucocorticoid therapy suppresses abnormal secretion of big IGF-II by non-islet cell tumours inducing hypoglycaemia (NICTH). Clin Endocrinol .1998;49:491-498.
8. Hamberg P, De Jong FA, Boonstra JG, et al. Non-islet-cell tumor induced hypoglycemia in patients with advanced gastrointestinal stromal tumor possibly worsened by imatinib. J Clin Oncol. 2006;24:e30-e31.
9. Rikhof B, van Doorn J, Suurmeijer AJ, et al. Insulin-like growth factors and insulin-like growth factor-binding proteins in relation to disease status and incidence of hypoglycaemia in patients with a gastrointestinal stromal tumour. Ann Oncol. 2009;20:1582-1588.
10. Braconi C, Bracci R, Bearzi I, et al. Insulin-like growth factor (IGF) 1 and 2 help to predict disease outcome in GIST patients. Ann Oncol. 2008;19:1293-1298.
Hypoglycemia, a frequently encountered medical emergency, is usually seen in patients with diabetes, most commonly as a result of iatrogenesis. However, it can also be encountered in nondiabetic patients. Various causes, such as pancreatic islet cell tumors producing insulin, primary or secondary adrenal insufficiency, advanced liver disease, pheochromocytoma and hypothyroidism, have been found to contribute to the condition in the nondiabetic population.1 In rare cases, an excessive production of insulin-like growth factor (IGF-2) – a condition known as nonislet cell tumor-induced hypoglycemia (NICTH) – has also been found to cause hypoglycemia. Hypoinsulinemic hypoglycemia, with low IGF-1 levels and an IGF-2-IgF1 ratio of greater than 10, is found to be suggestive of NICTH.
Case presentation and summary
An 81-year-old man with a history of diabetes mellitus, systolic heart failure, chronic kidney disease, and metastatic classical gastrointestinal spindle cell sarcoma presented to the emergency department with an acute change in mental status resulting from a new onset hypoglycemia. He was admitted, and during his hospital stay, he experienced severe hypoglycemic episodes with symptomatic presentations of diaphoresis on multiple occasions. A detailed history revealed that for diabetes, the patient had been on insulin for the first 12 years after his diagnosis, after which he was switched to metformin 500 mg twice daily for about 2 years, and as a satisfactory glycemic control was attained, eventually metformin had also been stopped 3 years prior to the current presentation.
The patient’s past medical records were obtained from the hospital at which he had been diagnosed gastrointestinal spindle cell sarcoma. Patient had not received treatment for the cancer as the disease was too widespread to be treated. The gastrointestinal spindle cell sarcoma, which had initially been surgically resected 7 years before the current presentation, had a recurrence 3 years later with abdominal and pulmonary metastasis, but no liver metastasis. No further intervention was carried out because the widely metastasized disease would not have benefited from any more surgical intervention and chemotherapy was not initiated because of the patient’s comorbid illnesses.
A blood sample drawn from the patient at the time of one hypoglycemic event, revealed low serum insulin <0.1 U/ml (normal, 2-19.6 U/ml); low C-peptide level, 0.59 ng/ml (0.8-3.85 ng/ml); low IGF-1, 16 ng/ml (5-4 ng/ml); and IGF-3, 0.9 ng/ml (2.2-4.5 ng/ml). IGF-2 levels were found to be markedly elevated at 945 ng/ml (47-350 ng/ml). The calculated IGF-2-IGF-1 ratio was 59.06 (normal, <10), suggesting NICTH as the etiology for the patient’s hypoglycemia.
The hypoglycemic episodes were initially treated with a continuous dextrose infusion followed by diazoxide treatment. However, diazoxide did not prevent his hypoglycemic episodes, so dexamethasone was considered as an alternative for his condition. The dexamethasone treatment resulted in the normalization of the patient’s serum glucose levels and resolution of his symptoms. The patient was discharged in a satisfactory state few days later and followed up thereafter. No recurrence of hypoglycemic episodes was found, and he was continued on dexamethasone therapy.
Discussion
Hypoglycemia due to NICTH is rare, with a prevalence of four times less than that of insulinoma.3 In most cases, NICTH occurs in patients with solid tumors of mesenchymal and epithelial origins such as hepatocellular carcinoma, gastric carcinoma or mesothelioma.4 In NICTH, the serum levels of insulin, C-peptide, and IGF-1 are usually decreased or undetectable. However, the circulating levels of total IGF2 may be increased, decreased, or normal. Concurrent normal to high morning cortisol and normal response to cosyntropin stimulation can rule out adrenal insufficiency and suggest NICTH. An IGF-2: IGF-1 ratio of >10 is considered to be clinically significant and highly suggestive of NICTH.5 Hypoglycemia in NICTH can be managed by administration of oral glucose, intravenous dextrose or glucagon. In some cases, diazoxide, a potent inhibitor of insulin secretion, has been found to be useful.6 Diazoxide directly inhibits the release of insulin through stimulation of adrenergic receptors and also has an extra pancreatic hyperglycemic effect, probably by inhibiting cyclic adenosine monophosphate phosphodiesterase, resulting in higher plasma levels of cyclic AMP and enhanced glycogenolysis.
Glucocorticoid therapy has been shown to suppress IGF-2 in a dose dependent manner and also by increasing gluconeogenesis.7 Surgical resection of the tumor whenever possible is the treatment of choice followed by radiotherapy and chemotherapy for inoperable disease and if successful, usually results in resolution of hypoglycemia. Imatinib, is the chemotherapeutic drug of choice for metastatic GIST, but many case reports have suggested worsening of hypoglycemia in advanced GIST with the use of the drug.8 The patient described in our report was not on any chemotherapy, hence hypoglycemia could not be attributed to it. On the basis of findings among 24 patients with GIST, Rikhof and colleagues have recommended monitoring plasma levels of pro-IGF-IIE to identify patients at high risk for developing hypoglycemia, especially those with progressive disease.9 Furthermore, over expression of IGF-2 as a predictor of potential relapse may be an area for potential research and further study.10
Hypoglycemia, a frequently encountered medical emergency, is usually seen in patients with diabetes, most commonly as a result of iatrogenesis. However, it can also be encountered in nondiabetic patients. Various causes, such as pancreatic islet cell tumors producing insulin, primary or secondary adrenal insufficiency, advanced liver disease, pheochromocytoma and hypothyroidism, have been found to contribute to the condition in the nondiabetic population.1 In rare cases, an excessive production of insulin-like growth factor (IGF-2) – a condition known as nonislet cell tumor-induced hypoglycemia (NICTH) – has also been found to cause hypoglycemia. Hypoinsulinemic hypoglycemia, with low IGF-1 levels and an IGF-2-IgF1 ratio of greater than 10, is found to be suggestive of NICTH.
Case presentation and summary
An 81-year-old man with a history of diabetes mellitus, systolic heart failure, chronic kidney disease, and metastatic classical gastrointestinal spindle cell sarcoma presented to the emergency department with an acute change in mental status resulting from a new onset hypoglycemia. He was admitted, and during his hospital stay, he experienced severe hypoglycemic episodes with symptomatic presentations of diaphoresis on multiple occasions. A detailed history revealed that for diabetes, the patient had been on insulin for the first 12 years after his diagnosis, after which he was switched to metformin 500 mg twice daily for about 2 years, and as a satisfactory glycemic control was attained, eventually metformin had also been stopped 3 years prior to the current presentation.
The patient’s past medical records were obtained from the hospital at which he had been diagnosed gastrointestinal spindle cell sarcoma. Patient had not received treatment for the cancer as the disease was too widespread to be treated. The gastrointestinal spindle cell sarcoma, which had initially been surgically resected 7 years before the current presentation, had a recurrence 3 years later with abdominal and pulmonary metastasis, but no liver metastasis. No further intervention was carried out because the widely metastasized disease would not have benefited from any more surgical intervention and chemotherapy was not initiated because of the patient’s comorbid illnesses.
A blood sample drawn from the patient at the time of one hypoglycemic event, revealed low serum insulin <0.1 U/ml (normal, 2-19.6 U/ml); low C-peptide level, 0.59 ng/ml (0.8-3.85 ng/ml); low IGF-1, 16 ng/ml (5-4 ng/ml); and IGF-3, 0.9 ng/ml (2.2-4.5 ng/ml). IGF-2 levels were found to be markedly elevated at 945 ng/ml (47-350 ng/ml). The calculated IGF-2-IGF-1 ratio was 59.06 (normal, <10), suggesting NICTH as the etiology for the patient’s hypoglycemia.
The hypoglycemic episodes were initially treated with a continuous dextrose infusion followed by diazoxide treatment. However, diazoxide did not prevent his hypoglycemic episodes, so dexamethasone was considered as an alternative for his condition. The dexamethasone treatment resulted in the normalization of the patient’s serum glucose levels and resolution of his symptoms. The patient was discharged in a satisfactory state few days later and followed up thereafter. No recurrence of hypoglycemic episodes was found, and he was continued on dexamethasone therapy.
Discussion
Hypoglycemia due to NICTH is rare, with a prevalence of four times less than that of insulinoma.3 In most cases, NICTH occurs in patients with solid tumors of mesenchymal and epithelial origins such as hepatocellular carcinoma, gastric carcinoma or mesothelioma.4 In NICTH, the serum levels of insulin, C-peptide, and IGF-1 are usually decreased or undetectable. However, the circulating levels of total IGF2 may be increased, decreased, or normal. Concurrent normal to high morning cortisol and normal response to cosyntropin stimulation can rule out adrenal insufficiency and suggest NICTH. An IGF-2: IGF-1 ratio of >10 is considered to be clinically significant and highly suggestive of NICTH.5 Hypoglycemia in NICTH can be managed by administration of oral glucose, intravenous dextrose or glucagon. In some cases, diazoxide, a potent inhibitor of insulin secretion, has been found to be useful.6 Diazoxide directly inhibits the release of insulin through stimulation of adrenergic receptors and also has an extra pancreatic hyperglycemic effect, probably by inhibiting cyclic adenosine monophosphate phosphodiesterase, resulting in higher plasma levels of cyclic AMP and enhanced glycogenolysis.
Glucocorticoid therapy has been shown to suppress IGF-2 in a dose dependent manner and also by increasing gluconeogenesis.7 Surgical resection of the tumor whenever possible is the treatment of choice followed by radiotherapy and chemotherapy for inoperable disease and if successful, usually results in resolution of hypoglycemia. Imatinib, is the chemotherapeutic drug of choice for metastatic GIST, but many case reports have suggested worsening of hypoglycemia in advanced GIST with the use of the drug.8 The patient described in our report was not on any chemotherapy, hence hypoglycemia could not be attributed to it. On the basis of findings among 24 patients with GIST, Rikhof and colleagues have recommended monitoring plasma levels of pro-IGF-IIE to identify patients at high risk for developing hypoglycemia, especially those with progressive disease.9 Furthermore, over expression of IGF-2 as a predictor of potential relapse may be an area for potential research and further study.10
1. Marks V, Teale JD. Tumours producing hypoglycaemia. Diabetes Metab Rev. 1991;7:79-91.
2. Dutta P, Aggarwal A, Gogate Y, Nahar U, Shah VN, Singla M. Non-islet cell tumor-induced hypoglycemia: a report of five cases and brief review of the literature. Endocrinol Diabetes Metab Case Rep. 2013;2013:130046
3. de Groot JW, Rikhof B, van Doorn J, et al. Non-islet cell tumour-induced hypoglycaemia: a review of the literature including two new cases. Endocr Relat Cancer. 2007;14:979-93.
4. Fukuda I, Hizuka N, Ishikawa Y, et al. Clinical features of insulin-like growth factor II producing non-islet-cell tumor hypoglycemia
5. Marks V, Teale JD: Tumours producing hypoglycaemia. Endocr Relat Cancer. 1998;5:111-129.
6. Le Roith D. Tumor-induced hypoglycemia. N Engl J Med. 1999;341:757-758.
7. Teale JD, Marks V. Glucocorticoid therapy suppresses abnormal secretion of big IGF-II by non-islet cell tumours inducing hypoglycaemia (NICTH). Clin Endocrinol .1998;49:491-498.
8. Hamberg P, De Jong FA, Boonstra JG, et al. Non-islet-cell tumor induced hypoglycemia in patients with advanced gastrointestinal stromal tumor possibly worsened by imatinib. J Clin Oncol. 2006;24:e30-e31.
9. Rikhof B, van Doorn J, Suurmeijer AJ, et al. Insulin-like growth factors and insulin-like growth factor-binding proteins in relation to disease status and incidence of hypoglycaemia in patients with a gastrointestinal stromal tumour. Ann Oncol. 2009;20:1582-1588.
10. Braconi C, Bracci R, Bearzi I, et al. Insulin-like growth factor (IGF) 1 and 2 help to predict disease outcome in GIST patients. Ann Oncol. 2008;19:1293-1298.
1. Marks V, Teale JD. Tumours producing hypoglycaemia. Diabetes Metab Rev. 1991;7:79-91.
2. Dutta P, Aggarwal A, Gogate Y, Nahar U, Shah VN, Singla M. Non-islet cell tumor-induced hypoglycemia: a report of five cases and brief review of the literature. Endocrinol Diabetes Metab Case Rep. 2013;2013:130046
3. de Groot JW, Rikhof B, van Doorn J, et al. Non-islet cell tumour-induced hypoglycaemia: a review of the literature including two new cases. Endocr Relat Cancer. 2007;14:979-93.
4. Fukuda I, Hizuka N, Ishikawa Y, et al. Clinical features of insulin-like growth factor II producing non-islet-cell tumor hypoglycemia
5. Marks V, Teale JD: Tumours producing hypoglycaemia. Endocr Relat Cancer. 1998;5:111-129.
6. Le Roith D. Tumor-induced hypoglycemia. N Engl J Med. 1999;341:757-758.
7. Teale JD, Marks V. Glucocorticoid therapy suppresses abnormal secretion of big IGF-II by non-islet cell tumours inducing hypoglycaemia (NICTH). Clin Endocrinol .1998;49:491-498.
8. Hamberg P, De Jong FA, Boonstra JG, et al. Non-islet-cell tumor induced hypoglycemia in patients with advanced gastrointestinal stromal tumor possibly worsened by imatinib. J Clin Oncol. 2006;24:e30-e31.
9. Rikhof B, van Doorn J, Suurmeijer AJ, et al. Insulin-like growth factors and insulin-like growth factor-binding proteins in relation to disease status and incidence of hypoglycaemia in patients with a gastrointestinal stromal tumour. Ann Oncol. 2009;20:1582-1588.
10. Braconi C, Bracci R, Bearzi I, et al. Insulin-like growth factor (IGF) 1 and 2 help to predict disease outcome in GIST patients. Ann Oncol. 2008;19:1293-1298.
VIDEO: Hip, knee replacements fall in Danish RA patients
MADRID – The rates of both total hip and total knee replacement surgeries dropped among Danish patients with rheumatoid arthritis since the mid-1990s, reductions that were coincident with more widespread use of biologic drugs as well as with other improvements in care, according to analyses of Danish national health records.
“The introduction of guidelines [on biologic drug use] in 2002 and increasing use of biologic drugs [as a result] may have contributed to this positive development,” Lene Dreyer, MD, said at the European Congress of Rheumatology. Other factors that may have also contributed include widespread use of conventional disease-modifying antirheumatic drugs (DMARDs) and adoption of a treat-to-target strategy by many clinicians.
In 1996, the first year studied and before any biologic DMARDs were routinely used for rheumatoid arthritis, the rate of total knee replacement was nearly 6/1,000 person-years among RA patients, compared with a 0.42/1,000 person-years rate in the general adult Danish population, a roughly 14-fold excess among the RA patients, Dr. Dreyer reported. But by 2016, ”this gap had almost disappeared,” she said in a video interview. “It seems like rheumatologists in Denmark are doing a good job” treating RA patients.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
That may have been especially true subsequent to 2002, when the Danish Institute for Rational Pharmacotherapy issued recommendations that opened the door to wider use of biologic DMARDs, such as tumor necrosis factor inhibitors, to treat RA patients, noted Dr. Dreyer of Gentofte University Hospital, Copenhagen. During 2003-2011, use of total knee replacement surgery in RA patients fell by an average annualized rate of 0.2 surgeries/1,000 person-years. But among the general Danish population the average annualized rate of knee surgeries rose by 0.08/1,000 person-years.
“This is a very important finding,” commented Robert Landewé, MD, PhD, professor of rheumatology at the Academic Medical Center in Amsterdam. “It is extremely difficult to test the effect of the introduction of the [biologic DMARD] guidelines,” he cautioned. But he highlighted the positive finding that the excess of hip and knee replacement surgeries in patients with RA, compared with the general population, had recently narrowed.
Dr. Dreyer and her associates used records from the Danish National Patient Register to compare 29,427 patients with incident RA during 1996-2011 with more than 290,000 matched control individuals. All people studied had not undergone knee or hip replacement surgery prior to their entry into the study. The researchers used an “interrupted time series analysis” to examine the possible impact of the introduction of widespread access to biologic DMARDs starting in 2003.
The analysis showed that the rate of total hip replacements in 1996 was nearly 9 surgeries/1,000 person-years among RA patients and nearly 3/1,000 person-years in the general population, a threefold excess for RA patients. This rate fell by an average annual rate of 0.38/1,000 person-years among RA patients both before and after 2002, so that by 2011 the rate was roughly half the 1996 rate, about 4.5/1,000 patient-years. The rate in the general population rose during 1996-2011, and by 2011 was nearly 4/1,000 person-years and so nearly the same as RA patients. Wider availability of biologic DMARDs for RA patients starting in 2003 did not have an apparent impact on the rate of total hip replacement.
In contrast, wider use of biologic DMARDs appeared to have an effect on the rate of total knee surgeries among RA patients. During 1996-2001, the rate rose by an annual average of 0.19/1,000 person-years, very similar to the 0.21/1,000 person-years annual rise in the general Danish population. However, during 2003-2011, the average annual rate of total knee surgery fell by 0.20/1,000 person-years in the RA patients but continued to rise at an annual average rate of 0.08/1,000 person-years in the general population, Dr. Dreyer reported.
Additional Danish registry data exist for patients who received biologic DMARDs, and Dr. Dreyer said that she and her associates hope to use this to further examine the impact of these drugs on patient outcomes.
Dr. Dreyer has received lecture fees from Merck Sharp & Dohme and UCB. Dr. Landewé has received consulting fees from several drug companies.
[email protected]
On Twitter @mitchelzoler
MADRID – The rates of both total hip and total knee replacement surgeries dropped among Danish patients with rheumatoid arthritis since the mid-1990s, reductions that were coincident with more widespread use of biologic drugs as well as with other improvements in care, according to analyses of Danish national health records.
“The introduction of guidelines [on biologic drug use] in 2002 and increasing use of biologic drugs [as a result] may have contributed to this positive development,” Lene Dreyer, MD, said at the European Congress of Rheumatology. Other factors that may have also contributed include widespread use of conventional disease-modifying antirheumatic drugs (DMARDs) and adoption of a treat-to-target strategy by many clinicians.
In 1996, the first year studied and before any biologic DMARDs were routinely used for rheumatoid arthritis, the rate of total knee replacement was nearly 6/1,000 person-years among RA patients, compared with a 0.42/1,000 person-years rate in the general adult Danish population, a roughly 14-fold excess among the RA patients, Dr. Dreyer reported. But by 2016, ”this gap had almost disappeared,” she said in a video interview. “It seems like rheumatologists in Denmark are doing a good job” treating RA patients.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
That may have been especially true subsequent to 2002, when the Danish Institute for Rational Pharmacotherapy issued recommendations that opened the door to wider use of biologic DMARDs, such as tumor necrosis factor inhibitors, to treat RA patients, noted Dr. Dreyer of Gentofte University Hospital, Copenhagen. During 2003-2011, use of total knee replacement surgery in RA patients fell by an average annualized rate of 0.2 surgeries/1,000 person-years. But among the general Danish population the average annualized rate of knee surgeries rose by 0.08/1,000 person-years.
“This is a very important finding,” commented Robert Landewé, MD, PhD, professor of rheumatology at the Academic Medical Center in Amsterdam. “It is extremely difficult to test the effect of the introduction of the [biologic DMARD] guidelines,” he cautioned. But he highlighted the positive finding that the excess of hip and knee replacement surgeries in patients with RA, compared with the general population, had recently narrowed.
Dr. Dreyer and her associates used records from the Danish National Patient Register to compare 29,427 patients with incident RA during 1996-2011 with more than 290,000 matched control individuals. All people studied had not undergone knee or hip replacement surgery prior to their entry into the study. The researchers used an “interrupted time series analysis” to examine the possible impact of the introduction of widespread access to biologic DMARDs starting in 2003.
The analysis showed that the rate of total hip replacements in 1996 was nearly 9 surgeries/1,000 person-years among RA patients and nearly 3/1,000 person-years in the general population, a threefold excess for RA patients. This rate fell by an average annual rate of 0.38/1,000 person-years among RA patients both before and after 2002, so that by 2011 the rate was roughly half the 1996 rate, about 4.5/1,000 patient-years. The rate in the general population rose during 1996-2011, and by 2011 was nearly 4/1,000 person-years and so nearly the same as RA patients. Wider availability of biologic DMARDs for RA patients starting in 2003 did not have an apparent impact on the rate of total hip replacement.
In contrast, wider use of biologic DMARDs appeared to have an effect on the rate of total knee surgeries among RA patients. During 1996-2001, the rate rose by an annual average of 0.19/1,000 person-years, very similar to the 0.21/1,000 person-years annual rise in the general Danish population. However, during 2003-2011, the average annual rate of total knee surgery fell by 0.20/1,000 person-years in the RA patients but continued to rise at an annual average rate of 0.08/1,000 person-years in the general population, Dr. Dreyer reported.
Additional Danish registry data exist for patients who received biologic DMARDs, and Dr. Dreyer said that she and her associates hope to use this to further examine the impact of these drugs on patient outcomes.
Dr. Dreyer has received lecture fees from Merck Sharp & Dohme and UCB. Dr. Landewé has received consulting fees from several drug companies.
[email protected]
On Twitter @mitchelzoler
MADRID – The rates of both total hip and total knee replacement surgeries dropped among Danish patients with rheumatoid arthritis since the mid-1990s, reductions that were coincident with more widespread use of biologic drugs as well as with other improvements in care, according to analyses of Danish national health records.
“The introduction of guidelines [on biologic drug use] in 2002 and increasing use of biologic drugs [as a result] may have contributed to this positive development,” Lene Dreyer, MD, said at the European Congress of Rheumatology. Other factors that may have also contributed include widespread use of conventional disease-modifying antirheumatic drugs (DMARDs) and adoption of a treat-to-target strategy by many clinicians.
In 1996, the first year studied and before any biologic DMARDs were routinely used for rheumatoid arthritis, the rate of total knee replacement was nearly 6/1,000 person-years among RA patients, compared with a 0.42/1,000 person-years rate in the general adult Danish population, a roughly 14-fold excess among the RA patients, Dr. Dreyer reported. But by 2016, ”this gap had almost disappeared,” she said in a video interview. “It seems like rheumatologists in Denmark are doing a good job” treating RA patients.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
That may have been especially true subsequent to 2002, when the Danish Institute for Rational Pharmacotherapy issued recommendations that opened the door to wider use of biologic DMARDs, such as tumor necrosis factor inhibitors, to treat RA patients, noted Dr. Dreyer of Gentofte University Hospital, Copenhagen. During 2003-2011, use of total knee replacement surgery in RA patients fell by an average annualized rate of 0.2 surgeries/1,000 person-years. But among the general Danish population the average annualized rate of knee surgeries rose by 0.08/1,000 person-years.
“This is a very important finding,” commented Robert Landewé, MD, PhD, professor of rheumatology at the Academic Medical Center in Amsterdam. “It is extremely difficult to test the effect of the introduction of the [biologic DMARD] guidelines,” he cautioned. But he highlighted the positive finding that the excess of hip and knee replacement surgeries in patients with RA, compared with the general population, had recently narrowed.
Dr. Dreyer and her associates used records from the Danish National Patient Register to compare 29,427 patients with incident RA during 1996-2011 with more than 290,000 matched control individuals. All people studied had not undergone knee or hip replacement surgery prior to their entry into the study. The researchers used an “interrupted time series analysis” to examine the possible impact of the introduction of widespread access to biologic DMARDs starting in 2003.
The analysis showed that the rate of total hip replacements in 1996 was nearly 9 surgeries/1,000 person-years among RA patients and nearly 3/1,000 person-years in the general population, a threefold excess for RA patients. This rate fell by an average annual rate of 0.38/1,000 person-years among RA patients both before and after 2002, so that by 2011 the rate was roughly half the 1996 rate, about 4.5/1,000 patient-years. The rate in the general population rose during 1996-2011, and by 2011 was nearly 4/1,000 person-years and so nearly the same as RA patients. Wider availability of biologic DMARDs for RA patients starting in 2003 did not have an apparent impact on the rate of total hip replacement.
In contrast, wider use of biologic DMARDs appeared to have an effect on the rate of total knee surgeries among RA patients. During 1996-2001, the rate rose by an annual average of 0.19/1,000 person-years, very similar to the 0.21/1,000 person-years annual rise in the general Danish population. However, during 2003-2011, the average annual rate of total knee surgery fell by 0.20/1,000 person-years in the RA patients but continued to rise at an annual average rate of 0.08/1,000 person-years in the general population, Dr. Dreyer reported.
Additional Danish registry data exist for patients who received biologic DMARDs, and Dr. Dreyer said that she and her associates hope to use this to further examine the impact of these drugs on patient outcomes.
Dr. Dreyer has received lecture fees from Merck Sharp & Dohme and UCB. Dr. Landewé has received consulting fees from several drug companies.
[email protected]
On Twitter @mitchelzoler
AT THE EULAR 2017 CONGRESS
Key clinical point:
Major finding: RA patient hip replacements fell from nearly 9/1,000 person-years in 1996 to about 4.5/1,000 person-years in 2011.
Data source: Records from more than 300,000 people in the Danish National Patient Register.
Disclosures: Dr. Dreyer has received lecture fees from Merck Sharp & Dohme and UCB. Dr. Landewé has received consulting fees from several drug companies.