User login
Topical treatments for rosacea to be reviewed at this year’s meeting
This year’s SDEF Hawaii Dermatology Seminar is set to showcase the latest research and expert tips, advice, and perspective on a range of topics.
Linda Stein Gold, MD, director of dermatology research, Henry Ford Health System, Detroit, and a codirector of the meeting, shared a taste of the topics she will be presenting.
In the field of rosacea, “topical ivermectin has proven to be a very effective and safe treatment,” she said in an interview. “On the horizon we will have a new treatment for the erythema of rosacea which seems very well tolerated. Also, topical minocycline foam appears to be very promising in phase II studies.”
See our video interview with Dr. Stein Gold from last year’s Hawaii Dermatology Seminar, where she discussed new approaches for the topical treatment of acne that are on the horizon.
The Hawaii Dermatology Seminar is provided by Global Academy for Medical Education/Skin Disease Education Foundation. SDEF and this news organization are owned by the same parent company. It is being held in Maui, Jan. 29-Feb. 3.
Dr. Stein Gold disclosed relationships with companies including Galderma, Leo, Novan, Valeant, Dermira, Novartis, Celgene, Allergan, Foamix, and Promius.
This year’s SDEF Hawaii Dermatology Seminar is set to showcase the latest research and expert tips, advice, and perspective on a range of topics.
Linda Stein Gold, MD, director of dermatology research, Henry Ford Health System, Detroit, and a codirector of the meeting, shared a taste of the topics she will be presenting.
In the field of rosacea, “topical ivermectin has proven to be a very effective and safe treatment,” she said in an interview. “On the horizon we will have a new treatment for the erythema of rosacea which seems very well tolerated. Also, topical minocycline foam appears to be very promising in phase II studies.”
See our video interview with Dr. Stein Gold from last year’s Hawaii Dermatology Seminar, where she discussed new approaches for the topical treatment of acne that are on the horizon.
The Hawaii Dermatology Seminar is provided by Global Academy for Medical Education/Skin Disease Education Foundation. SDEF and this news organization are owned by the same parent company. It is being held in Maui, Jan. 29-Feb. 3.
Dr. Stein Gold disclosed relationships with companies including Galderma, Leo, Novan, Valeant, Dermira, Novartis, Celgene, Allergan, Foamix, and Promius.
This year’s SDEF Hawaii Dermatology Seminar is set to showcase the latest research and expert tips, advice, and perspective on a range of topics.
Linda Stein Gold, MD, director of dermatology research, Henry Ford Health System, Detroit, and a codirector of the meeting, shared a taste of the topics she will be presenting.
In the field of rosacea, “topical ivermectin has proven to be a very effective and safe treatment,” she said in an interview. “On the horizon we will have a new treatment for the erythema of rosacea which seems very well tolerated. Also, topical minocycline foam appears to be very promising in phase II studies.”
See our video interview with Dr. Stein Gold from last year’s Hawaii Dermatology Seminar, where she discussed new approaches for the topical treatment of acne that are on the horizon.
The Hawaii Dermatology Seminar is provided by Global Academy for Medical Education/Skin Disease Education Foundation. SDEF and this news organization are owned by the same parent company. It is being held in Maui, Jan. 29-Feb. 3.
Dr. Stein Gold disclosed relationships with companies including Galderma, Leo, Novan, Valeant, Dermira, Novartis, Celgene, Allergan, Foamix, and Promius.
Improving the efficacy of obinutuzumab

Preclinical research suggests that immune stimulation through Toll-like receptor 7 (TLR7) agonism can enhance the efficacy of obinutuzumab in lymphoma.
Researchers found that combining the anti-CD20 monoclonal antibody obinutuzumab with the TLR7 agonist R848 improved survival in lab mice with lymphoma.
The combination also demonstrated efficacy against chronic lymphocytic leukemia (CLL) cells in vitro.
Tim Illidge, PhD, MBBS, of the University of Manchester in the UK, and his colleagues reported these findings in the journal Leukemia.
The research was funded by the Kay Kendall Leukaemia Fund and Cancer Research UK in collaboration with Roche Pharmaceutical Research and Early Development.
The researchers said they initially found that R848 activates immune cells in vivo and enhances obinutuzumab-mediated antitumor effector mechanisms in vitro.
The team therefore went on to test R848 and obinutuzumab in C57Bl/6 mice bearing human CD20+ lymphoma (EL4). The mice received obinutuzumab modified to express the murine glycoengineered IgG2a Fc region (m2a) starting 1 day after tumor inoculation and systemic R848 once weekly for 4 weeks.
The researchers found that monotherapy with either obinutuzumab or R848 significantly improved survival compared to control (P<0.0001), but only 8% to 15% of mice that received monotherapy were long-term survivors (living more than 90 days).
Mice that received obinutuzumab in combination with R848 had significantly better survival than mice that received either monotherapy (P<0.0001). And about 70% of mice receiving the combination remained tumor-free out to 95 days.
Furthermore, long-term survivors that had received the combination treatment were protected from tumor re-challenge.
The researchers also tested the combination in a second model—human CD20 transgenic mice, which express the human CD20 antigen on normal B cells. The team said this model is more akin to the clinical situation.
The mice received treatment 7 days after the inoculation of EL4hCD20 cells. Mice that received obinutuzumab monotherapy had significantly better survival than control mice (P=0.02), but there were no long-term survivors. For mice that received R848 monotherapy, survival was not significantly different from that of controls.
Mice that received R848 in combination with obinutuzumab had significantly better survival than mice that received obinutuzumab alone (P=0.003).
In fact, 6 of the 12 mice that received the combination were long-term survivors. And 5 of these mice rejected tumor re-challenge.
“We were excited when we discovered that combining obinutuzumab with TLR7 activation significantly enhanced survival of animals with lymphoma by effectively eradicating tumors,” Dr Illidge said. “Clearly, more work needs to be done to assess the impact of this combination on humans, but this study is, nevertheless, very promising.”
The researchers said the primary antitumor activity of the combination is dependent on natural killer cells and CD4 helper T cells but not on CD8 killer T cells.
“While the combination therapy was highly effective, CD8 killer T cells did not play a major role in the therapy,” said Eleanor Cheadle, PhD, also of the University of Manchester.
“Given the important role that killer T cells can play in long-term protection from tumor regrowth, we are looking at ways to enhance activation of these cells after obinutuzumab therapy.”
The researchers also found that, in vitro, R848 significantly enhanced natural killer cell-mediated antibody-dependent cellular cytotoxicity against obinutuzumab-opsonized CLL cells and significantly increased non-specific, antibody-independent killing of CLL cells. ![]()

Preclinical research suggests that immune stimulation through Toll-like receptor 7 (TLR7) agonism can enhance the efficacy of obinutuzumab in lymphoma.
Researchers found that combining the anti-CD20 monoclonal antibody obinutuzumab with the TLR7 agonist R848 improved survival in lab mice with lymphoma.
The combination also demonstrated efficacy against chronic lymphocytic leukemia (CLL) cells in vitro.
Tim Illidge, PhD, MBBS, of the University of Manchester in the UK, and his colleagues reported these findings in the journal Leukemia.
The research was funded by the Kay Kendall Leukaemia Fund and Cancer Research UK in collaboration with Roche Pharmaceutical Research and Early Development.
The researchers said they initially found that R848 activates immune cells in vivo and enhances obinutuzumab-mediated antitumor effector mechanisms in vitro.
The team therefore went on to test R848 and obinutuzumab in C57Bl/6 mice bearing human CD20+ lymphoma (EL4). The mice received obinutuzumab modified to express the murine glycoengineered IgG2a Fc region (m2a) starting 1 day after tumor inoculation and systemic R848 once weekly for 4 weeks.
The researchers found that monotherapy with either obinutuzumab or R848 significantly improved survival compared to control (P<0.0001), but only 8% to 15% of mice that received monotherapy were long-term survivors (living more than 90 days).
Mice that received obinutuzumab in combination with R848 had significantly better survival than mice that received either monotherapy (P<0.0001). And about 70% of mice receiving the combination remained tumor-free out to 95 days.
Furthermore, long-term survivors that had received the combination treatment were protected from tumor re-challenge.
The researchers also tested the combination in a second model—human CD20 transgenic mice, which express the human CD20 antigen on normal B cells. The team said this model is more akin to the clinical situation.
The mice received treatment 7 days after the inoculation of EL4hCD20 cells. Mice that received obinutuzumab monotherapy had significantly better survival than control mice (P=0.02), but there were no long-term survivors. For mice that received R848 monotherapy, survival was not significantly different from that of controls.
Mice that received R848 in combination with obinutuzumab had significantly better survival than mice that received obinutuzumab alone (P=0.003).
In fact, 6 of the 12 mice that received the combination were long-term survivors. And 5 of these mice rejected tumor re-challenge.
“We were excited when we discovered that combining obinutuzumab with TLR7 activation significantly enhanced survival of animals with lymphoma by effectively eradicating tumors,” Dr Illidge said. “Clearly, more work needs to be done to assess the impact of this combination on humans, but this study is, nevertheless, very promising.”
The researchers said the primary antitumor activity of the combination is dependent on natural killer cells and CD4 helper T cells but not on CD8 killer T cells.
“While the combination therapy was highly effective, CD8 killer T cells did not play a major role in the therapy,” said Eleanor Cheadle, PhD, also of the University of Manchester.
“Given the important role that killer T cells can play in long-term protection from tumor regrowth, we are looking at ways to enhance activation of these cells after obinutuzumab therapy.”
The researchers also found that, in vitro, R848 significantly enhanced natural killer cell-mediated antibody-dependent cellular cytotoxicity against obinutuzumab-opsonized CLL cells and significantly increased non-specific, antibody-independent killing of CLL cells. ![]()

Preclinical research suggests that immune stimulation through Toll-like receptor 7 (TLR7) agonism can enhance the efficacy of obinutuzumab in lymphoma.
Researchers found that combining the anti-CD20 monoclonal antibody obinutuzumab with the TLR7 agonist R848 improved survival in lab mice with lymphoma.
The combination also demonstrated efficacy against chronic lymphocytic leukemia (CLL) cells in vitro.
Tim Illidge, PhD, MBBS, of the University of Manchester in the UK, and his colleagues reported these findings in the journal Leukemia.
The research was funded by the Kay Kendall Leukaemia Fund and Cancer Research UK in collaboration with Roche Pharmaceutical Research and Early Development.
The researchers said they initially found that R848 activates immune cells in vivo and enhances obinutuzumab-mediated antitumor effector mechanisms in vitro.
The team therefore went on to test R848 and obinutuzumab in C57Bl/6 mice bearing human CD20+ lymphoma (EL4). The mice received obinutuzumab modified to express the murine glycoengineered IgG2a Fc region (m2a) starting 1 day after tumor inoculation and systemic R848 once weekly for 4 weeks.
The researchers found that monotherapy with either obinutuzumab or R848 significantly improved survival compared to control (P<0.0001), but only 8% to 15% of mice that received monotherapy were long-term survivors (living more than 90 days).
Mice that received obinutuzumab in combination with R848 had significantly better survival than mice that received either monotherapy (P<0.0001). And about 70% of mice receiving the combination remained tumor-free out to 95 days.
Furthermore, long-term survivors that had received the combination treatment were protected from tumor re-challenge.
The researchers also tested the combination in a second model—human CD20 transgenic mice, which express the human CD20 antigen on normal B cells. The team said this model is more akin to the clinical situation.
The mice received treatment 7 days after the inoculation of EL4hCD20 cells. Mice that received obinutuzumab monotherapy had significantly better survival than control mice (P=0.02), but there were no long-term survivors. For mice that received R848 monotherapy, survival was not significantly different from that of controls.
Mice that received R848 in combination with obinutuzumab had significantly better survival than mice that received obinutuzumab alone (P=0.003).
In fact, 6 of the 12 mice that received the combination were long-term survivors. And 5 of these mice rejected tumor re-challenge.
“We were excited when we discovered that combining obinutuzumab with TLR7 activation significantly enhanced survival of animals with lymphoma by effectively eradicating tumors,” Dr Illidge said. “Clearly, more work needs to be done to assess the impact of this combination on humans, but this study is, nevertheless, very promising.”
The researchers said the primary antitumor activity of the combination is dependent on natural killer cells and CD4 helper T cells but not on CD8 killer T cells.
“While the combination therapy was highly effective, CD8 killer T cells did not play a major role in the therapy,” said Eleanor Cheadle, PhD, also of the University of Manchester.
“Given the important role that killer T cells can play in long-term protection from tumor regrowth, we are looking at ways to enhance activation of these cells after obinutuzumab therapy.”
The researchers also found that, in vitro, R848 significantly enhanced natural killer cell-mediated antibody-dependent cellular cytotoxicity against obinutuzumab-opsonized CLL cells and significantly increased non-specific, antibody-independent killing of CLL cells. ![]()
CMA report reveals successes and shortcomings

The European Medicines Agency (EMA) has released a report showing both successes and room for improvement regarding conditional marketing authorizations (CMAs).
CMA is one of the tools available to regulators to support the development of and early access to drugs that address unmet medical needs of patients in the European Union.
Drugs are granted CMA if the public health benefit of their immediate availability is thought to outweigh the risk of an authorization on the basis of less comprehensive data than normally required.
A CMA is valid for 1 year. As part of the authorization, the drug’s developer is obliged to carry out further studies to obtain complete data.
The EMA’s Committee for Medicinal Products for Human Use (CHMP) assesses the data generated by these specific post-authorization obligations at least annually to ensure the balance of benefits and risks of the drug continues to remain positive.
At the end of its assessment, the CHMP issues a recommendation regarding the renewal of the CMA or its conversion into a standard marketing authorization.
Overview
The EMA’s report summarizes the experience with CMAs from the first use of this authorization type in 2006 until June 30, 2016.
During this time, a total of 30 drugs have received a CMA, including several
hematology drugs—Adcetris (brentuximab vedotin),
Arzerra (ofatumumab), Blincyto (blinatumomab), Bosulif (bosutinib), Darzalex (daratumumab), and Pixuvri (pixantrone).
Eleven CMAs have been converted into standard marketing authorizations (including Arzerra’s CMA), 2 have been withdrawn for commercial reasons, and 17 are still conditional authorizations.
None of the drugs that still have CMAs have been authorized for more than 5 years. And none of the CMAs issued since 2006 have had to be revoked or suspended.
Successes
According to the EMA’s analysis, marketing authorization holders comply with the specific obligations imposed by the agency.
More than 90% of completed specific obligations did not result in major changes of scope, and about 70% of specific obligations did not require an extension to the originally specified timelines.
The report shows that it took an average of 4 years to generate the additional data needed and to convert a CMA into a full marketing authorization.
This suggests patients with life-threatening or seriously debilitating conditions had access to promising drugs much earlier than they would have under standard authorization.
Areas for improvement
The EMA’s analysis also revealed room for improvement.
The report showed that, relatively frequently, CMA was first
considered only during the assessment of the drug application, which meant granting a CMA took longer than intended.
Therefore, the EMA recommends that drug developers engage in early dialogue with the EMA
and prospectively plan to apply for a CMA.
The agency said this should support
prompt assessment of such applications and could also facilitate prompt
completion of additional studies and timely availability of
comprehensive data.
The EMA said another area for improvement is engaging other stakeholders involved in bringing drugs to patients—in particular, Health Technology Assessment bodies—to facilitate the generation of all data needed for decision-making through one development program. ![]()

The European Medicines Agency (EMA) has released a report showing both successes and room for improvement regarding conditional marketing authorizations (CMAs).
CMA is one of the tools available to regulators to support the development of and early access to drugs that address unmet medical needs of patients in the European Union.
Drugs are granted CMA if the public health benefit of their immediate availability is thought to outweigh the risk of an authorization on the basis of less comprehensive data than normally required.
A CMA is valid for 1 year. As part of the authorization, the drug’s developer is obliged to carry out further studies to obtain complete data.
The EMA’s Committee for Medicinal Products for Human Use (CHMP) assesses the data generated by these specific post-authorization obligations at least annually to ensure the balance of benefits and risks of the drug continues to remain positive.
At the end of its assessment, the CHMP issues a recommendation regarding the renewal of the CMA or its conversion into a standard marketing authorization.
Overview
The EMA’s report summarizes the experience with CMAs from the first use of this authorization type in 2006 until June 30, 2016.
During this time, a total of 30 drugs have received a CMA, including several
hematology drugs—Adcetris (brentuximab vedotin),
Arzerra (ofatumumab), Blincyto (blinatumomab), Bosulif (bosutinib), Darzalex (daratumumab), and Pixuvri (pixantrone).
Eleven CMAs have been converted into standard marketing authorizations (including Arzerra’s CMA), 2 have been withdrawn for commercial reasons, and 17 are still conditional authorizations.
None of the drugs that still have CMAs have been authorized for more than 5 years. And none of the CMAs issued since 2006 have had to be revoked or suspended.
Successes
According to the EMA’s analysis, marketing authorization holders comply with the specific obligations imposed by the agency.
More than 90% of completed specific obligations did not result in major changes of scope, and about 70% of specific obligations did not require an extension to the originally specified timelines.
The report shows that it took an average of 4 years to generate the additional data needed and to convert a CMA into a full marketing authorization.
This suggests patients with life-threatening or seriously debilitating conditions had access to promising drugs much earlier than they would have under standard authorization.
Areas for improvement
The EMA’s analysis also revealed room for improvement.
The report showed that, relatively frequently, CMA was first
considered only during the assessment of the drug application, which meant granting a CMA took longer than intended.
Therefore, the EMA recommends that drug developers engage in early dialogue with the EMA
and prospectively plan to apply for a CMA.
The agency said this should support
prompt assessment of such applications and could also facilitate prompt
completion of additional studies and timely availability of
comprehensive data.
The EMA said another area for improvement is engaging other stakeholders involved in bringing drugs to patients—in particular, Health Technology Assessment bodies—to facilitate the generation of all data needed for decision-making through one development program. ![]()

The European Medicines Agency (EMA) has released a report showing both successes and room for improvement regarding conditional marketing authorizations (CMAs).
CMA is one of the tools available to regulators to support the development of and early access to drugs that address unmet medical needs of patients in the European Union.
Drugs are granted CMA if the public health benefit of their immediate availability is thought to outweigh the risk of an authorization on the basis of less comprehensive data than normally required.
A CMA is valid for 1 year. As part of the authorization, the drug’s developer is obliged to carry out further studies to obtain complete data.
The EMA’s Committee for Medicinal Products for Human Use (CHMP) assesses the data generated by these specific post-authorization obligations at least annually to ensure the balance of benefits and risks of the drug continues to remain positive.
At the end of its assessment, the CHMP issues a recommendation regarding the renewal of the CMA or its conversion into a standard marketing authorization.
Overview
The EMA’s report summarizes the experience with CMAs from the first use of this authorization type in 2006 until June 30, 2016.
During this time, a total of 30 drugs have received a CMA, including several
hematology drugs—Adcetris (brentuximab vedotin),
Arzerra (ofatumumab), Blincyto (blinatumomab), Bosulif (bosutinib), Darzalex (daratumumab), and Pixuvri (pixantrone).
Eleven CMAs have been converted into standard marketing authorizations (including Arzerra’s CMA), 2 have been withdrawn for commercial reasons, and 17 are still conditional authorizations.
None of the drugs that still have CMAs have been authorized for more than 5 years. And none of the CMAs issued since 2006 have had to be revoked or suspended.
Successes
According to the EMA’s analysis, marketing authorization holders comply with the specific obligations imposed by the agency.
More than 90% of completed specific obligations did not result in major changes of scope, and about 70% of specific obligations did not require an extension to the originally specified timelines.
The report shows that it took an average of 4 years to generate the additional data needed and to convert a CMA into a full marketing authorization.
This suggests patients with life-threatening or seriously debilitating conditions had access to promising drugs much earlier than they would have under standard authorization.
Areas for improvement
The EMA’s analysis also revealed room for improvement.
The report showed that, relatively frequently, CMA was first
considered only during the assessment of the drug application, which meant granting a CMA took longer than intended.
Therefore, the EMA recommends that drug developers engage in early dialogue with the EMA
and prospectively plan to apply for a CMA.
The agency said this should support
prompt assessment of such applications and could also facilitate prompt
completion of additional studies and timely availability of
comprehensive data.
The EMA said another area for improvement is engaging other stakeholders involved in bringing drugs to patients—in particular, Health Technology Assessment bodies—to facilitate the generation of all data needed for decision-making through one development program. ![]()
Iron-fortified nutrition bars combat anemia in India

Consuming an iron-fortified nutrition bar daily for a few months can fight anemia without producing side effects, according to research published in the American Journal of Clinical Nutrition.
Anemic
women in India who consumed an iron-fortified nutrition bar each day for 90 days
were much more likely to experience increases in hemoglobin and
hematocrit and to be cured of their anemia than women who did not consume
such bars.
Rajvi Mehta, a medical student at Duke University in Durham, North Carolina, developed the nutrition bars used in this study, known as GudNesS bars.
The bars are made with iron-rich, natural, local (to India), and culturally accepted ingredients. They contain the World Health Organization’s daily recommended dose of iron.
In 2011, Mehta worked with nutritionists and physicians in India to establish a social venture there called Let’s Be Well Red (LBWR) to begin large-scale production of the bars.
The study, conducted from March to August 2014 in Mumbai and Navi Mumbai, India, involved 179 anemic, non-pregnant participants of reproductive age (18-35) at 10 demographically diverse sites.
The sites were randomly placed in either a control group or an intervention group.
Women in the intervention group received 1 iron-fortified nutrition bar (containing 14 mg Fe) daily for 90 days, and women in the control group received nothing. Baseline characteristics were comparable between the groups.
Each group underwent 3 blood tests during the 90-day follow-up period—at 15 days, 45 days, and 90 days—to measure their hemoglobin and hematocrit.
Seventy-six percent of subjects (n=136) completed all follow-up assessments (65 intervention and 71 control subjects).
The primary outcomes were 90-day changes from baseline in hemoglobin concentrations and hematocrit percentages.
The researchers said the mean hemoglobin and hematocrit increases after 90 days were greater for the intervention group than the control group, at 1.4 g/dL and 2.7%, respectively.
And subjects in the intervention group had a much greater decrease in anemia than those in the control group. At 90 days, 29.2% of subjects in the intervention group still had anemia, compared to 98.6% of those in the control group. The odds ratio was 0.007.
The researchers said no side effects were reported.
“We are encouraged by the results of this study, which show a positive connection between consuming an iron-fortified nutrition bar and a reduction in anemia prevalence,” said study author Elizabeth Turner, PhD, of Duke University.
“It appears to be a practical and well-tolerated solution to a significant health challenge in India.”
Let’s Be Well Red is currently operating in 3 locations in India and produces 100,000 bars each year that it distributes throughout the country.
“Anemia is a debilitating condition that can have severe health consequences,” Mehta said. “I am thrilled that my colleagues and I were able to develop a solution that has proven to be effective among a high-risk population. Making an impact in global health has long been a goal of mine.” ![]()

Consuming an iron-fortified nutrition bar daily for a few months can fight anemia without producing side effects, according to research published in the American Journal of Clinical Nutrition.
Anemic
women in India who consumed an iron-fortified nutrition bar each day for 90 days
were much more likely to experience increases in hemoglobin and
hematocrit and to be cured of their anemia than women who did not consume
such bars.
Rajvi Mehta, a medical student at Duke University in Durham, North Carolina, developed the nutrition bars used in this study, known as GudNesS bars.
The bars are made with iron-rich, natural, local (to India), and culturally accepted ingredients. They contain the World Health Organization’s daily recommended dose of iron.
In 2011, Mehta worked with nutritionists and physicians in India to establish a social venture there called Let’s Be Well Red (LBWR) to begin large-scale production of the bars.
The study, conducted from March to August 2014 in Mumbai and Navi Mumbai, India, involved 179 anemic, non-pregnant participants of reproductive age (18-35) at 10 demographically diverse sites.
The sites were randomly placed in either a control group or an intervention group.
Women in the intervention group received 1 iron-fortified nutrition bar (containing 14 mg Fe) daily for 90 days, and women in the control group received nothing. Baseline characteristics were comparable between the groups.
Each group underwent 3 blood tests during the 90-day follow-up period—at 15 days, 45 days, and 90 days—to measure their hemoglobin and hematocrit.
Seventy-six percent of subjects (n=136) completed all follow-up assessments (65 intervention and 71 control subjects).
The primary outcomes were 90-day changes from baseline in hemoglobin concentrations and hematocrit percentages.
The researchers said the mean hemoglobin and hematocrit increases after 90 days were greater for the intervention group than the control group, at 1.4 g/dL and 2.7%, respectively.
And subjects in the intervention group had a much greater decrease in anemia than those in the control group. At 90 days, 29.2% of subjects in the intervention group still had anemia, compared to 98.6% of those in the control group. The odds ratio was 0.007.
The researchers said no side effects were reported.
“We are encouraged by the results of this study, which show a positive connection between consuming an iron-fortified nutrition bar and a reduction in anemia prevalence,” said study author Elizabeth Turner, PhD, of Duke University.
“It appears to be a practical and well-tolerated solution to a significant health challenge in India.”
Let’s Be Well Red is currently operating in 3 locations in India and produces 100,000 bars each year that it distributes throughout the country.
“Anemia is a debilitating condition that can have severe health consequences,” Mehta said. “I am thrilled that my colleagues and I were able to develop a solution that has proven to be effective among a high-risk population. Making an impact in global health has long been a goal of mine.” ![]()

Consuming an iron-fortified nutrition bar daily for a few months can fight anemia without producing side effects, according to research published in the American Journal of Clinical Nutrition.
Anemic
women in India who consumed an iron-fortified nutrition bar each day for 90 days
were much more likely to experience increases in hemoglobin and
hematocrit and to be cured of their anemia than women who did not consume
such bars.
Rajvi Mehta, a medical student at Duke University in Durham, North Carolina, developed the nutrition bars used in this study, known as GudNesS bars.
The bars are made with iron-rich, natural, local (to India), and culturally accepted ingredients. They contain the World Health Organization’s daily recommended dose of iron.
In 2011, Mehta worked with nutritionists and physicians in India to establish a social venture there called Let’s Be Well Red (LBWR) to begin large-scale production of the bars.
The study, conducted from March to August 2014 in Mumbai and Navi Mumbai, India, involved 179 anemic, non-pregnant participants of reproductive age (18-35) at 10 demographically diverse sites.
The sites were randomly placed in either a control group or an intervention group.
Women in the intervention group received 1 iron-fortified nutrition bar (containing 14 mg Fe) daily for 90 days, and women in the control group received nothing. Baseline characteristics were comparable between the groups.
Each group underwent 3 blood tests during the 90-day follow-up period—at 15 days, 45 days, and 90 days—to measure their hemoglobin and hematocrit.
Seventy-six percent of subjects (n=136) completed all follow-up assessments (65 intervention and 71 control subjects).
The primary outcomes were 90-day changes from baseline in hemoglobin concentrations and hematocrit percentages.
The researchers said the mean hemoglobin and hematocrit increases after 90 days were greater for the intervention group than the control group, at 1.4 g/dL and 2.7%, respectively.
And subjects in the intervention group had a much greater decrease in anemia than those in the control group. At 90 days, 29.2% of subjects in the intervention group still had anemia, compared to 98.6% of those in the control group. The odds ratio was 0.007.
The researchers said no side effects were reported.
“We are encouraged by the results of this study, which show a positive connection between consuming an iron-fortified nutrition bar and a reduction in anemia prevalence,” said study author Elizabeth Turner, PhD, of Duke University.
“It appears to be a practical and well-tolerated solution to a significant health challenge in India.”
Let’s Be Well Red is currently operating in 3 locations in India and produces 100,000 bars each year that it distributes throughout the country.
“Anemia is a debilitating condition that can have severe health consequences,” Mehta said. “I am thrilled that my colleagues and I were able to develop a solution that has proven to be effective among a high-risk population. Making an impact in global health has long been a goal of mine.” ![]()
New recommendations for pediatric AMKL
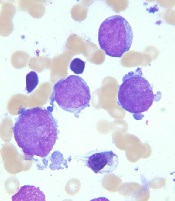
Image courtesy of St. Jude
Children’s Research Hospital
and Tina Motroni
Research has revealed genetic alterations that may prove useful for predicting treatment outcomes in pediatric patients with acute megakaryoblastic leukemia (AMKL) who do not have Down syndrome.
The study suggests that 3
genetic alterations can be used to identify high-risk patients who may benefit
from allogeneic stem cell transplant, and 1 alteration may identify
low-risk patients who require less chemotherapy than their peers.
Researchers said these findings, published in Nature Genetics, support revised diagnostic screening and treatment recommendations for pediatric AMKL.
“Because long-term survival for pediatric AMKL patients without Down syndrome is poor, just 14% to 34%, the standard recommendation by many pediatric oncologists has been to treat all patients with allogeneic stem cell transplantation during their first remission,” said study author Tanja Gruber, MD, PhD, of St. Jude Children’s Research Hospital in Memphis, Tennessee.
“In this study, we identified several genetic alterations that are important predictors of treatment success. All newly identified pediatric AMKL patients without Down syndrome should be screened for these prognostic indicators at diagnosis. The results will help identify which patients need allogeneic stem cell transplants during their first remission and which do not.”
This study involved next-generation sequencing of the whole exome or RNA of 99 AMKL patients without Down syndrome (75 pediatric and 24 adult patients). Along with the sequencing data, researchers analyzed patients’ gene expression and long-term survival.
Results
The results showed that non-Down syndrome pediatric AMKL can be divided into 7 subgroups based on the underlying genetic alteration, pattern of gene expression, and treatment outcome.
The subgroups include the newly identified HOX subgroup. About 15% of the pediatric patients in this study were in the HOX subgroup, which is characterized by several different HOX fusion genes.
The researchers also identified cooperating mutations that help fuel AMKL in different subgroups. The cooperating mutations include changes in the RB1 gene and recurring mutations in the RAS and JAK pathways.
In addition, the study showed that 3 genetic alterations—CBFA2T3-GLIS2, KMT2A rearrangements, and NUP98-KDM5A—are associated with reduced survival in pediatric AMKL subtypes.
The researchers said patients with these alterations may benefit from allogeneic stem cell transplants, so non-Down syndrome pediatric AMKL patients should be screened for these alterations at diagnosis.
The team also recommended testing AMKL patients for mutations in the GATA1 gene. GATA1 mutations are a hallmark of AMKL in children with Down syndrome, who almost always survive the leukemia. In this study, AMKL patients with GATA1 mutations and no fusion gene had the same favorable outcomes.
“The results raise the possibility that pediatric AMKL patients without Down syndrome who have mutations in GATA1 may benefit from the same reduced chemotherapy used to treat the leukemia in patients with Down syndrome,” Dr Gruber said.
These revised diagnostic screening and treatment recommendations are being implemented at St. Jude.
This study also showed that adults with AMKL lacked recurrent fusion genes. The most common mutations found in the adult patients were in TP53 (21%), cohesin genes (17%), splicing factor genes (17%), ASXL genes (17%), and DNMT3A (13%). About 4% had GATA1 mutations. ![]()

Image courtesy of St. Jude
Children’s Research Hospital
and Tina Motroni
Research has revealed genetic alterations that may prove useful for predicting treatment outcomes in pediatric patients with acute megakaryoblastic leukemia (AMKL) who do not have Down syndrome.
The study suggests that 3
genetic alterations can be used to identify high-risk patients who may benefit
from allogeneic stem cell transplant, and 1 alteration may identify
low-risk patients who require less chemotherapy than their peers.
Researchers said these findings, published in Nature Genetics, support revised diagnostic screening and treatment recommendations for pediatric AMKL.
“Because long-term survival for pediatric AMKL patients without Down syndrome is poor, just 14% to 34%, the standard recommendation by many pediatric oncologists has been to treat all patients with allogeneic stem cell transplantation during their first remission,” said study author Tanja Gruber, MD, PhD, of St. Jude Children’s Research Hospital in Memphis, Tennessee.
“In this study, we identified several genetic alterations that are important predictors of treatment success. All newly identified pediatric AMKL patients without Down syndrome should be screened for these prognostic indicators at diagnosis. The results will help identify which patients need allogeneic stem cell transplants during their first remission and which do not.”
This study involved next-generation sequencing of the whole exome or RNA of 99 AMKL patients without Down syndrome (75 pediatric and 24 adult patients). Along with the sequencing data, researchers analyzed patients’ gene expression and long-term survival.
Results
The results showed that non-Down syndrome pediatric AMKL can be divided into 7 subgroups based on the underlying genetic alteration, pattern of gene expression, and treatment outcome.
The subgroups include the newly identified HOX subgroup. About 15% of the pediatric patients in this study were in the HOX subgroup, which is characterized by several different HOX fusion genes.
The researchers also identified cooperating mutations that help fuel AMKL in different subgroups. The cooperating mutations include changes in the RB1 gene and recurring mutations in the RAS and JAK pathways.
In addition, the study showed that 3 genetic alterations—CBFA2T3-GLIS2, KMT2A rearrangements, and NUP98-KDM5A—are associated with reduced survival in pediatric AMKL subtypes.
The researchers said patients with these alterations may benefit from allogeneic stem cell transplants, so non-Down syndrome pediatric AMKL patients should be screened for these alterations at diagnosis.
The team also recommended testing AMKL patients for mutations in the GATA1 gene. GATA1 mutations are a hallmark of AMKL in children with Down syndrome, who almost always survive the leukemia. In this study, AMKL patients with GATA1 mutations and no fusion gene had the same favorable outcomes.
“The results raise the possibility that pediatric AMKL patients without Down syndrome who have mutations in GATA1 may benefit from the same reduced chemotherapy used to treat the leukemia in patients with Down syndrome,” Dr Gruber said.
These revised diagnostic screening and treatment recommendations are being implemented at St. Jude.
This study also showed that adults with AMKL lacked recurrent fusion genes. The most common mutations found in the adult patients were in TP53 (21%), cohesin genes (17%), splicing factor genes (17%), ASXL genes (17%), and DNMT3A (13%). About 4% had GATA1 mutations. ![]()

Image courtesy of St. Jude
Children’s Research Hospital
and Tina Motroni
Research has revealed genetic alterations that may prove useful for predicting treatment outcomes in pediatric patients with acute megakaryoblastic leukemia (AMKL) who do not have Down syndrome.
The study suggests that 3
genetic alterations can be used to identify high-risk patients who may benefit
from allogeneic stem cell transplant, and 1 alteration may identify
low-risk patients who require less chemotherapy than their peers.
Researchers said these findings, published in Nature Genetics, support revised diagnostic screening and treatment recommendations for pediatric AMKL.
“Because long-term survival for pediatric AMKL patients without Down syndrome is poor, just 14% to 34%, the standard recommendation by many pediatric oncologists has been to treat all patients with allogeneic stem cell transplantation during their first remission,” said study author Tanja Gruber, MD, PhD, of St. Jude Children’s Research Hospital in Memphis, Tennessee.
“In this study, we identified several genetic alterations that are important predictors of treatment success. All newly identified pediatric AMKL patients without Down syndrome should be screened for these prognostic indicators at diagnosis. The results will help identify which patients need allogeneic stem cell transplants during their first remission and which do not.”
This study involved next-generation sequencing of the whole exome or RNA of 99 AMKL patients without Down syndrome (75 pediatric and 24 adult patients). Along with the sequencing data, researchers analyzed patients’ gene expression and long-term survival.
Results
The results showed that non-Down syndrome pediatric AMKL can be divided into 7 subgroups based on the underlying genetic alteration, pattern of gene expression, and treatment outcome.
The subgroups include the newly identified HOX subgroup. About 15% of the pediatric patients in this study were in the HOX subgroup, which is characterized by several different HOX fusion genes.
The researchers also identified cooperating mutations that help fuel AMKL in different subgroups. The cooperating mutations include changes in the RB1 gene and recurring mutations in the RAS and JAK pathways.
In addition, the study showed that 3 genetic alterations—CBFA2T3-GLIS2, KMT2A rearrangements, and NUP98-KDM5A—are associated with reduced survival in pediatric AMKL subtypes.
The researchers said patients with these alterations may benefit from allogeneic stem cell transplants, so non-Down syndrome pediatric AMKL patients should be screened for these alterations at diagnosis.
The team also recommended testing AMKL patients for mutations in the GATA1 gene. GATA1 mutations are a hallmark of AMKL in children with Down syndrome, who almost always survive the leukemia. In this study, AMKL patients with GATA1 mutations and no fusion gene had the same favorable outcomes.
“The results raise the possibility that pediatric AMKL patients without Down syndrome who have mutations in GATA1 may benefit from the same reduced chemotherapy used to treat the leukemia in patients with Down syndrome,” Dr Gruber said.
These revised diagnostic screening and treatment recommendations are being implemented at St. Jude.
This study also showed that adults with AMKL lacked recurrent fusion genes. The most common mutations found in the adult patients were in TP53 (21%), cohesin genes (17%), splicing factor genes (17%), ASXL genes (17%), and DNMT3A (13%). About 4% had GATA1 mutations. ![]()
What Do Parasites Have to Do With Leukemia?
Parasites have been shown to have both pro- and antitumor effects. Malaria parasites (Plasmodium spp) are among those known to have this possible “bidirectional role” in carcinogenesis, say researchers from Aix-Marseille Université in France. They reviewed the current thinking on whether malaria—a worldwide killer—can be useful in cancer prevention and treatment.
Positive relationships between malaria and virus-associated cancers are relatively well documented, the researchers say. Evidence suggests that malaria can alter immune responses by modulating both humoral and cell-mediated immunity. Plasmodium-related cancers are primarily lymphoproliferative, vulnerable to virus reactivation. Epstein-Barr virus (EBV), for example, has been observed in lymphatic and hematologic tumors such as Hodgkin disease and T cell lymphoma, and malaria can reactivate EBV.
In animal studies, malarial infection with Plasmodium berghei (P berghei) increased the rate of spontaneous leukemia. In one study, concurrent infection with P berghei increased the incidence of malignant lymphoma in mice injected with Moloney leukemogenic virus.
On the other hand, Plasmodium spp also produces proteins that demonstrate certain anti-oncogenic effects, they note. The researchers suggest that using proteins in cancer treatment should be explored, adding that it’s a “safer approach than the inoculation of wild type Plasmodium.” Positive parasite-induced effects against cancers of the hematopoietic and lymphoid tissues are mentioned only for 2 species and those only in a decades-old study. Based on current knowledge, the researchers say, the antitumor effects observed are attributable to modifications to the host immune response. Thus, their characteristics and locations within the host can be highly diverse.
All in all, the researchers conclude, the growing evidence is opening intriguing pathways for using one ill to cure another.
Source:
Faure E. Parasitology. 2016;143(14):1811-1823.
Parasites have been shown to have both pro- and antitumor effects. Malaria parasites (Plasmodium spp) are among those known to have this possible “bidirectional role” in carcinogenesis, say researchers from Aix-Marseille Université in France. They reviewed the current thinking on whether malaria—a worldwide killer—can be useful in cancer prevention and treatment.
Positive relationships between malaria and virus-associated cancers are relatively well documented, the researchers say. Evidence suggests that malaria can alter immune responses by modulating both humoral and cell-mediated immunity. Plasmodium-related cancers are primarily lymphoproliferative, vulnerable to virus reactivation. Epstein-Barr virus (EBV), for example, has been observed in lymphatic and hematologic tumors such as Hodgkin disease and T cell lymphoma, and malaria can reactivate EBV.
In animal studies, malarial infection with Plasmodium berghei (P berghei) increased the rate of spontaneous leukemia. In one study, concurrent infection with P berghei increased the incidence of malignant lymphoma in mice injected with Moloney leukemogenic virus.
On the other hand, Plasmodium spp also produces proteins that demonstrate certain anti-oncogenic effects, they note. The researchers suggest that using proteins in cancer treatment should be explored, adding that it’s a “safer approach than the inoculation of wild type Plasmodium.” Positive parasite-induced effects against cancers of the hematopoietic and lymphoid tissues are mentioned only for 2 species and those only in a decades-old study. Based on current knowledge, the researchers say, the antitumor effects observed are attributable to modifications to the host immune response. Thus, their characteristics and locations within the host can be highly diverse.
All in all, the researchers conclude, the growing evidence is opening intriguing pathways for using one ill to cure another.
Source:
Faure E. Parasitology. 2016;143(14):1811-1823.
Parasites have been shown to have both pro- and antitumor effects. Malaria parasites (Plasmodium spp) are among those known to have this possible “bidirectional role” in carcinogenesis, say researchers from Aix-Marseille Université in France. They reviewed the current thinking on whether malaria—a worldwide killer—can be useful in cancer prevention and treatment.
Positive relationships between malaria and virus-associated cancers are relatively well documented, the researchers say. Evidence suggests that malaria can alter immune responses by modulating both humoral and cell-mediated immunity. Plasmodium-related cancers are primarily lymphoproliferative, vulnerable to virus reactivation. Epstein-Barr virus (EBV), for example, has been observed in lymphatic and hematologic tumors such as Hodgkin disease and T cell lymphoma, and malaria can reactivate EBV.
In animal studies, malarial infection with Plasmodium berghei (P berghei) increased the rate of spontaneous leukemia. In one study, concurrent infection with P berghei increased the incidence of malignant lymphoma in mice injected with Moloney leukemogenic virus.
On the other hand, Plasmodium spp also produces proteins that demonstrate certain anti-oncogenic effects, they note. The researchers suggest that using proteins in cancer treatment should be explored, adding that it’s a “safer approach than the inoculation of wild type Plasmodium.” Positive parasite-induced effects against cancers of the hematopoietic and lymphoid tissues are mentioned only for 2 species and those only in a decades-old study. Based on current knowledge, the researchers say, the antitumor effects observed are attributable to modifications to the host immune response. Thus, their characteristics and locations within the host can be highly diverse.
All in all, the researchers conclude, the growing evidence is opening intriguing pathways for using one ill to cure another.
Source:
Faure E. Parasitology. 2016;143(14):1811-1823.
Treat Hypothyroid Conditions
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Finding the Sweet Spot: The Diabetic Kidney
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Dabigatran has less bleeding than rivaroxaban in atrial fibrillation
Clinical question: Does dabigatran or rivaroxaban have more bleeding episodes?
Background: Alternatives to warfarin exist for stroke prevention in nonvalvular atrial fibrillation (AF). The RE-LY and ROCKET-AF trials demonstrated noninferiority to warfarin for both dabigatran (a direct thrombin inhibitor) and rivaroxaban (a factor Xa inhibitor), respectively. Although indirect comparisons have been done using data from these trials, direct, head-to-head comparisons are not available.
Study design: New-user cohort study.
Setting: Medicare beneficiaries 65 years or older with AF and a prescription for either dabigatran or rivaroxaban.
Intracranial hemorrhage and extracranial major bleeding events were significantly greater in the rivaroxaban group than the dabigatran group. There was no significant difference in thromboembolic stroke events.
Limitations include short treatment and follow-up times. Additionally, the study is not generalizable to younger populations.
Bottom line: In elderly patients with non-valvular AF, rivaroxaban was associated with more adverse bleeding events than dabigatran, with no difference in stroke prevention.
Citation: Graham DJ, Reichman ME, Wernecke M, et al. Stroke, bleeding, and mortality risks in elderly Medicare beneficiaries treated with dabigatran or rivaroxaban for nonvalvular atrial fibrillation. JAMA Intern Med. 2016;176:1662-71.
Dr. Graves is an assistant professor at the University of Utah School of Medicine and associate program director of quality and patient safety for the University of Utah Internal Medicine residency training program.
Clinical question: Does dabigatran or rivaroxaban have more bleeding episodes?
Background: Alternatives to warfarin exist for stroke prevention in nonvalvular atrial fibrillation (AF). The RE-LY and ROCKET-AF trials demonstrated noninferiority to warfarin for both dabigatran (a direct thrombin inhibitor) and rivaroxaban (a factor Xa inhibitor), respectively. Although indirect comparisons have been done using data from these trials, direct, head-to-head comparisons are not available.
Study design: New-user cohort study.
Setting: Medicare beneficiaries 65 years or older with AF and a prescription for either dabigatran or rivaroxaban.
Intracranial hemorrhage and extracranial major bleeding events were significantly greater in the rivaroxaban group than the dabigatran group. There was no significant difference in thromboembolic stroke events.
Limitations include short treatment and follow-up times. Additionally, the study is not generalizable to younger populations.
Bottom line: In elderly patients with non-valvular AF, rivaroxaban was associated with more adverse bleeding events than dabigatran, with no difference in stroke prevention.
Citation: Graham DJ, Reichman ME, Wernecke M, et al. Stroke, bleeding, and mortality risks in elderly Medicare beneficiaries treated with dabigatran or rivaroxaban for nonvalvular atrial fibrillation. JAMA Intern Med. 2016;176:1662-71.
Dr. Graves is an assistant professor at the University of Utah School of Medicine and associate program director of quality and patient safety for the University of Utah Internal Medicine residency training program.
Clinical question: Does dabigatran or rivaroxaban have more bleeding episodes?
Background: Alternatives to warfarin exist for stroke prevention in nonvalvular atrial fibrillation (AF). The RE-LY and ROCKET-AF trials demonstrated noninferiority to warfarin for both dabigatran (a direct thrombin inhibitor) and rivaroxaban (a factor Xa inhibitor), respectively. Although indirect comparisons have been done using data from these trials, direct, head-to-head comparisons are not available.
Study design: New-user cohort study.
Setting: Medicare beneficiaries 65 years or older with AF and a prescription for either dabigatran or rivaroxaban.
Intracranial hemorrhage and extracranial major bleeding events were significantly greater in the rivaroxaban group than the dabigatran group. There was no significant difference in thromboembolic stroke events.
Limitations include short treatment and follow-up times. Additionally, the study is not generalizable to younger populations.
Bottom line: In elderly patients with non-valvular AF, rivaroxaban was associated with more adverse bleeding events than dabigatran, with no difference in stroke prevention.
Citation: Graham DJ, Reichman ME, Wernecke M, et al. Stroke, bleeding, and mortality risks in elderly Medicare beneficiaries treated with dabigatran or rivaroxaban for nonvalvular atrial fibrillation. JAMA Intern Med. 2016;176:1662-71.
Dr. Graves is an assistant professor at the University of Utah School of Medicine and associate program director of quality and patient safety for the University of Utah Internal Medicine residency training program.
Unique microbiota mix found in guts of T1D patients
potentially offering early insight into possible links between the disease and gut germs.
The findings by an Italian team don’t confirm any connection between bacteria in the digestive system and diabetes. Still, “this study is probably the best example to date in the literature of inflammatory events happening in the gut that are correlated with type 1 diabetes,” said Aleksandar Kostic, PhD, of the department of microbiology and immunobiology at Harvard Medical School, Boston, who conducts similar research.
At issue: What role, if any, does the gut play in the development of type 1 diabetes (T1D)? Scientists already believe that the gut microbiome directly affects metabolism and the development of type 2 diabetes, according to Dr. Kostic. But T1D is an autoimmune disease, not a metabolic one, he said, “and the mechanisms are very different. For type 1, we don’t know a whole lot. We’re in the very early days.”
Still, “there’s a theory that inflammatory stimulus in the gut that is somehow partially responsible for causing T1D. The idea is that the microbiome is less diverse, which means that it loses its integrity in some way and loses the ability to crowd out inflammatory organisms,” he said in an interview.
For the new study, researchers led by scientists at Milan’s IRCCS San Raffaele Scientific Institute measured inflammation and the microbiome in the duodenal mucosa of 19 patients with T1D, 19 with celiac disease, and 16 healthy controls. They reported their findings online Jan. 19 (J Clin Endocrinol Metab. 2017. doi: 10.1210/jc.2016-3222).
The researchers found a unique inflammation profile through an analysis of gene expression in the patients with T1D. They called it a “peculiar signature” that’s notable for increased numbers of infiltration from the monocyte/macrophage lineage.
“In T1D patients, we didn’t observe any correlation between gene expression and [hemoglobin A1c] level, duration of diabetes, presence of secondary complications or the reason that led to endoscopy, indicating that gene expression was not influenced by these variables,” the researchers write.
They also found a “specific microbiota composition” featuring a reduction in the role of Proteobacteria and an increase in Firmicutes; this was unique to the T1D patients. Bacteroidetes “showed a trend to reduction” in both T1D and celiac patients compared to the controls.
“The expression of genes specific for T1D inflammation was associated with the abundance of specific bacteria in duodenum,” the researchers added.
Elena Barengolts, MD, of the division of endocrinology, diabetes, and metabolism at the University of Illinois, Chicago, who’s familiar with the study, said it appears to be valid. However, the methods used have limited powers to define specific types of bacteria, making it difficult to know if the germs in question are “bad” or “good,” she said in an interview.
For his part, Dr. Kostic said the findings are “really neat” and consistent with previous findings regarding the role of the gut microbiome and T1D. He pointed to a study he led that found less-diverse microbiomes in the guts of Finnish infants with T1D (Cell Host Microbe. 2015 Feb 11;17[2]:260-73).
As a result, the gut microbiome is “functionally capable of doing fewer things, and the community gets overrun by certain pathogens,” he said. “We saw that a lot of organisms were capable of promoting inflammation in the gut.”
Dr. Kostic, Dr. Barengolts, and the study authors report no relevant disclosures.
potentially offering early insight into possible links between the disease and gut germs.
The findings by an Italian team don’t confirm any connection between bacteria in the digestive system and diabetes. Still, “this study is probably the best example to date in the literature of inflammatory events happening in the gut that are correlated with type 1 diabetes,” said Aleksandar Kostic, PhD, of the department of microbiology and immunobiology at Harvard Medical School, Boston, who conducts similar research.
At issue: What role, if any, does the gut play in the development of type 1 diabetes (T1D)? Scientists already believe that the gut microbiome directly affects metabolism and the development of type 2 diabetes, according to Dr. Kostic. But T1D is an autoimmune disease, not a metabolic one, he said, “and the mechanisms are very different. For type 1, we don’t know a whole lot. We’re in the very early days.”
Still, “there’s a theory that inflammatory stimulus in the gut that is somehow partially responsible for causing T1D. The idea is that the microbiome is less diverse, which means that it loses its integrity in some way and loses the ability to crowd out inflammatory organisms,” he said in an interview.
For the new study, researchers led by scientists at Milan’s IRCCS San Raffaele Scientific Institute measured inflammation and the microbiome in the duodenal mucosa of 19 patients with T1D, 19 with celiac disease, and 16 healthy controls. They reported their findings online Jan. 19 (J Clin Endocrinol Metab. 2017. doi: 10.1210/jc.2016-3222).
The researchers found a unique inflammation profile through an analysis of gene expression in the patients with T1D. They called it a “peculiar signature” that’s notable for increased numbers of infiltration from the monocyte/macrophage lineage.
“In T1D patients, we didn’t observe any correlation between gene expression and [hemoglobin A1c] level, duration of diabetes, presence of secondary complications or the reason that led to endoscopy, indicating that gene expression was not influenced by these variables,” the researchers write.
They also found a “specific microbiota composition” featuring a reduction in the role of Proteobacteria and an increase in Firmicutes; this was unique to the T1D patients. Bacteroidetes “showed a trend to reduction” in both T1D and celiac patients compared to the controls.
“The expression of genes specific for T1D inflammation was associated with the abundance of specific bacteria in duodenum,” the researchers added.
Elena Barengolts, MD, of the division of endocrinology, diabetes, and metabolism at the University of Illinois, Chicago, who’s familiar with the study, said it appears to be valid. However, the methods used have limited powers to define specific types of bacteria, making it difficult to know if the germs in question are “bad” or “good,” she said in an interview.
For his part, Dr. Kostic said the findings are “really neat” and consistent with previous findings regarding the role of the gut microbiome and T1D. He pointed to a study he led that found less-diverse microbiomes in the guts of Finnish infants with T1D (Cell Host Microbe. 2015 Feb 11;17[2]:260-73).
As a result, the gut microbiome is “functionally capable of doing fewer things, and the community gets overrun by certain pathogens,” he said. “We saw that a lot of organisms were capable of promoting inflammation in the gut.”
Dr. Kostic, Dr. Barengolts, and the study authors report no relevant disclosures.
potentially offering early insight into possible links between the disease and gut germs.
The findings by an Italian team don’t confirm any connection between bacteria in the digestive system and diabetes. Still, “this study is probably the best example to date in the literature of inflammatory events happening in the gut that are correlated with type 1 diabetes,” said Aleksandar Kostic, PhD, of the department of microbiology and immunobiology at Harvard Medical School, Boston, who conducts similar research.
At issue: What role, if any, does the gut play in the development of type 1 diabetes (T1D)? Scientists already believe that the gut microbiome directly affects metabolism and the development of type 2 diabetes, according to Dr. Kostic. But T1D is an autoimmune disease, not a metabolic one, he said, “and the mechanisms are very different. For type 1, we don’t know a whole lot. We’re in the very early days.”
Still, “there’s a theory that inflammatory stimulus in the gut that is somehow partially responsible for causing T1D. The idea is that the microbiome is less diverse, which means that it loses its integrity in some way and loses the ability to crowd out inflammatory organisms,” he said in an interview.
For the new study, researchers led by scientists at Milan’s IRCCS San Raffaele Scientific Institute measured inflammation and the microbiome in the duodenal mucosa of 19 patients with T1D, 19 with celiac disease, and 16 healthy controls. They reported their findings online Jan. 19 (J Clin Endocrinol Metab. 2017. doi: 10.1210/jc.2016-3222).
The researchers found a unique inflammation profile through an analysis of gene expression in the patients with T1D. They called it a “peculiar signature” that’s notable for increased numbers of infiltration from the monocyte/macrophage lineage.
“In T1D patients, we didn’t observe any correlation between gene expression and [hemoglobin A1c] level, duration of diabetes, presence of secondary complications or the reason that led to endoscopy, indicating that gene expression was not influenced by these variables,” the researchers write.
They also found a “specific microbiota composition” featuring a reduction in the role of Proteobacteria and an increase in Firmicutes; this was unique to the T1D patients. Bacteroidetes “showed a trend to reduction” in both T1D and celiac patients compared to the controls.
“The expression of genes specific for T1D inflammation was associated with the abundance of specific bacteria in duodenum,” the researchers added.
Elena Barengolts, MD, of the division of endocrinology, diabetes, and metabolism at the University of Illinois, Chicago, who’s familiar with the study, said it appears to be valid. However, the methods used have limited powers to define specific types of bacteria, making it difficult to know if the germs in question are “bad” or “good,” she said in an interview.
For his part, Dr. Kostic said the findings are “really neat” and consistent with previous findings regarding the role of the gut microbiome and T1D. He pointed to a study he led that found less-diverse microbiomes in the guts of Finnish infants with T1D (Cell Host Microbe. 2015 Feb 11;17[2]:260-73).
As a result, the gut microbiome is “functionally capable of doing fewer things, and the community gets overrun by certain pathogens,” he said. “We saw that a lot of organisms were capable of promoting inflammation in the gut.”
Dr. Kostic, Dr. Barengolts, and the study authors report no relevant disclosures.
FROM THE JOURNAL OF CLINICAL ENDOCRINOLOGY & METABOLISM
Key clinical point: Patients with type 1 diabetes (T1D) show signs of unique inflammation and microbiota in the duodenal mucosa, compared with controls and celiac patients.
Major finding: T1D patients had a “peculiar” inflammation signature and a unique microbiota composition, and there’s a sign of a link between inflammation and bacteria levels.
Data source: An analysis of 19 patients with T1D, 19 with celiac disease, and 16 healthy controls.
Disclosures: The study was supported by institutional funds, and the authors report no relevant disclosures.





