User login
Biomechanical Consequences of Anterior Femoral Notching in Cruciate-Retaining Versus Posterior-Stabilized Total Knee Arthroplasty
Although rare, periprosthetic fractures remain a significant complication after total knee arthroplasty (TKA), occurring in 0.3% to 2.5% of cases.1-4 Hirsh and colleagues5 were among the first to suggest that anterior femoral notching during TKA was a potential risk factor for postoperative periprosthetic femoral fracture because notching may weaken the anterior femoral cortex. Anterior femoral notching, a cortex violation occurring during an anterior bone cut, occurs in up to 30% of cases.6 Using a theoretical biomechanical model, Culp and colleagues1 found that increasing the depth of the notch defect into the cortex led to reduced torsional strength. In more recent, cadaveric biomechanical studies, notching of the anterior femoral cortex decreased torsional strength by up to 39%.7,8 Contrary to these biomechanical studies, a retrospective study evaluating 1089 TKAs using 2 implant designs (Anatomic Graduated Component, Biomet and Legacy, Zimmer) demonstrated no significant effect of anterior femoral notching with respect to incidence of supracondylar femur fractures.6 That study, however, did not address whether implant design is associated with a differential risk for fracture in the presence of anterior notching.
Previous biomechanical studies have primarily investigated cruciate-retaining (CR) femoral components and properties with respect to anterior notching, even though the posterior-stabilized (PS) design is used more often in the United States.1,7 According to a Mayo Clinic survey, TKAs with a PS design increased from <10% in 1990 to almost 75% by 1997.9 Today, there is little to no consensus about which implant is better, and use of one or the other depends largely on the surgeon and varies widely between countries and regions.10 PS designs require more bone resection and demonstrate prosthesis-controlled rollback during flexion, whereas CR designs preserve more bone and achieve posterior stabilization via the posterior cruciate ligament.11 Despite these differences in design and mechanics, a 2013 Cochrane review of TKA design found no clinically significant differences between CR and PS with respect to pain, range of motion, or clinical and radiologic outcomes.10 The reviewers did not specifically address periprosthetic fractures associated with either femoral notching or TKA design, as they could not quantitatively analyze postoperative complications because of the diversity of reports. Given the limited number of reported cases, a review of radiographic findings pertaining to the characteristics of supracondylar fractures in anterior femoral notching was unsuccessful.12 As the previous biomechanical studies of anterior notching used primarily CR models or no prostheses at all, a study of biomechanical differences between CR and PS designs in the presence of anterior notching is warranted.1,7,8 Therefore, we conducted a study to assess the effect of anterior femoral notching on torsional strength and load to failure in CR and PS femoral components.
Materials and Methods
Twelve fourth-generation composite adult left femur synthetic sawbones (Sawbones; Pacific Research Laboratories) were selected for their consistent biomechanical properties, vs those of cadaveric specimens; in addition, low intersample variability made them preferable to cadaveric bones given the small sample used in this study.13,14 All bones were from the same lot. All were visually inspected for defects and found to be acceptable. In each sample, an anterior cortical defect was created by making an anterior cut with an undersized (size 4) posterior referencing guide. In addition, the distance from the proximal end of the notch to the implant fell within 15 mm, as that is the maximum distance from the implant a notch can be placed using a standard femoral cutting jig.15 Six femora were instrumented with CR implants and 6 with PS implants (DePuy Synthes). Implants were placed using standardized cuts. Before testing, each implant was inspected for proper fit and found to be securely fastened to the femur. In addition, precision calipers were used to measure notch depth and distance from notch to implant before loading. A custom polymethylmethacrylate torsion jig was used to fix each instrumented femur proximally and distally on the femoral implant (Figure 1). Care was taken to ensure the distal jig engaged only the implant, thus isolating the notch as a stress riser. Each femur was loaded in external rotation through the proximal femoral jig along the anatomical axis. Use of external rotation was based on study findings implicating external rotation of the tibia as the most likely mechanism for generating a fracture in the event of a fall.12 Furthermore, distal femur fractures are predominantly spiral as opposed to butterfly or bending—an indication that torsion is the most likely mechanism of failure.16 With no axial rotation possible within the prosthesis, increased torsional stress is undoubtedly generated within adjacent bone. Each specimen underwent torsional stiffness testing and then load to failure. Torsional stiffness was measured by slowly loading each femur in external rotation, from 1 to 18 Nm for 3 cycles at a displacement rate of 0.5° per second. Each specimen then underwent torsional load-to-failure testing on an Instron 5800R machine at a rate of 0.5° per second. Failure was defined as the moment of fracture and subsequent decrease in torsional load—determined graphically by the peak torsional load followed immediately by a sharp decrease in load. Stiffness was determined as the slope of torque to the displacement curve for each cycle, and torque to failure was the highest recorded torque before fracture. Fracture pattern was noted after failure. A sample size of 6 specimens per group provided 80% power to detect a between-group difference of 1 Nm per degree in stiffness, using an estimated SD of 0.7 Nm per degree. In our statistical analysis, continuous variables are reported as means and SDs. Data from our torsional stiffness and load-to-failure testing were analyzed with unpaired 2-sample t tests, and P < .05 was considered statistically significant.
Results
We did not detect a statistical difference in notch depth, notch-to-implant distance, or femoral length between the CR and PS groups. Mean (SD) notch depth was 6.0 (1.3) mm for CR and 4.9 (1.0) mm for PS (P = .13); mean (SD) distance from the proximal end of the notch to the implant was 13.8 (1.7) mm for CR and 11.1 (3.2) mm for PS (P = .08); and mean (SD) femoral length was 46.2 (0.1) cm for CR and 46.2 (0.1) cm for PS (P = .60).
Mean (SD) torsional stiffness for the first 3 precycles was 6.2 (1.2), 8.7 (1.5), and 8.8 (1.4) Nm per degree for the CR group and 6.0 (0.7), 8.4 (1.4), and 8.6 (1.4) Nm per degree for the PS group; the differences were not statistically significant (Figure 2A). In addition, there were no statistically significant differences in mean (SD) stiffness at failure between CR, 6.5 (0.7) Nm per degree, and PS, 7.1 (0.9) Nm per degree (P = .24; Figure 2B) or in mean (SD) final torque at failure between CR, 62.4 (9.4) Nm, and PS, 62.7 (12.2) Nm (P = .95; Figure 2C).
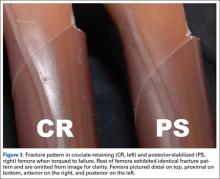
All fractures in both groups were oblique fractures originating at the proximal angle of the notch and extended proximally. None extended distally into the box. Fracture locations and patterns were identical in the CR and PS groups of femurs (Figure 3).
Discussion
Periprosthetic fractures after TKA remain rare. However, these fractures can significantly increase morbidity and complications. Anterior femoral notching occurs inadvertently in 30% to 40% of TKAs.6,17 The impact of femoral notching on supracondylar femur fracture is inconsistent between biomechanical and retrospective clinical studies. Retrospective studies failed to find a significant correlation between anterior femoral notching and supracondylar femur fractures.6,17 However, findings of biomechanical studies have suggested that a notch 3 mm deep will reduce the torsional strength of the femur by 29%.7 Another study, using 3-dimensional finite element analysis, showed a significant increase in local stress with a notch deeper than 3 mm.15
To our knowledge, no clinical studies, including the aforementioned Cochrane review,10 have specifically evaluated the difference in risk for periprosthetic fracture between different TKA models in the presence of notching.11 The biomechanical differences between implant designs could be a confounding factor in the results of past studies. More bone resection is required in PS designs than in CR designs. The position of the PS intercondylar cutout, much lower than the top of the patella flange, should not increase susceptibility to fractures more than in CR designs, but this hypothesis, though accepted, has not been validated biomechanically or addressed specifically in prospective or retrospective clinical analysis. In the present study, we used a biomechanical model to replicate an external rotation failure mechanism and quantify the differences in torsional strength and load to failure between CR TKA and PS TKA models in the presence of anterior femoral notching. Our results showed no significant differences in torsional stiffness, stiffness at failure, or torque at failure between the CR and PS design groups in the presence of anterior femoral notching.
In this study, all femoral fractures were oblique, and they all originated at the site of the cortical defect, not the notch—a situation markedly different from having bending forces applied to the femur. Previous biomechanical data indicated that bending forces applied to a notched femur cause fractures originating at the notch, whereas torsional forces applied to a notched femur cause fractures originating at the anterior aspect of the bone–component interface.7 The difference is attributable to study design. Our femurs were held fixed at their proximal end, which may have exacerbated any bending forces applied during external rotation, but we thought constraining the proximal femur would better replicate a fall involving external rotation.
More important for our study, an oblique fracture pattern was noted for both design groups (CR and PS), indicating the fracture pattern was unrelated to the area from which bone was resected for the PS design. All femur fractures in both design groups occurred proximal to a well-fixed prosthesis, indicating they should be classified as Vancouver C fractures. This is significant because intercondylar fossa resection (PS group) did not convert the fractures into Vancouver B2 fractures, which involve prosthesis loosening caused by pericomponent fracture.18 This simple observation validated our hypothesis that there would be no biomechanical differences between CR and PS designs with respect to the effects of anterior femoral notching. This lack of a significant difference may be attributed to the PS intercondylar cutout being much lower than the top of the anterior flange shielding the resected bone deep to the anterior flange.7 In addition, given the rarity of supracondylar fractures and the lack of sufficient relevant clinical data, it is difficult to speculate on the fracture patterns observed in clinical cases versus biomechanical studies.12
The use of synthetic bone models instead of cadaveric specimens could be seen as a limitation. Although synthetic bones may not reproduce the mechanism of failure in living and cadaveric femurs, the mechanical properties of synthetic bones have previously been found to fall within the range of those of cadaveric bones under axial loading, bending, and torsion testing.13,14 As a uniform testing material, synthetic bones allow removal of the confounding variations in bone size and quality that plague biomechanical studies in cadaveric bones.13,14 Interfemoral variability was 20 to 200 times higher in cadaveric femurs than in synthetic bones, which makes synthetic femurs preferable to cadaveric femurs, especially in studies with a small sample size.13,14 In addition, a uniform specimen provides consistent, reproducible osteotomies, which were crucial for consistent mechanical evaluation of each configuration in this study.
The long-term clinical significance of anterior femoral notching in periprosthetic fractures is equivocal, possibly because most studies predominantly use CR implants.6 This may not be an issue if it is shown that CR and PS implants have the same mechanical properties. Despite the differences between clinical studies and our biomechanical study, reevaluation of clinical data is not warranted given the biomechanical data we present here. Results of biomechanical studies like ours still suggest an increased immediate postoperative risk for supracondylar fracture after anterior cortical notching of the femur.5,7 Ultimately, this study found that, compared with a CR design, a PS design did not alter the torsional biomechanical properties or fracture pattern of an anteriorly notched femur.
1. Culp RW, Schmidt RG, Hanks G, Mak A, Esterhai JL Jr, Heppenstall RB. Supracondylar fracture of the femur following prosthetic knee arthroplasty. Clin Orthop Relat Res. 1987;(222):212-222.
2. Delport PH, Van Audekercke R, Martens M, Mulier JC. Conservative treatment of ipsilateral supracondylar femoral fracture after total knee arthroplasty. J Trauma. 1984;24(9):846-849.
3. Figgie MP, Goldberg VM, Figgie HE 3rd, Sobel M. The results of treatment of supracondylar fracture above total knee arthroplasty. J Arthroplasty. 1990;5(3):267-276.
4. Rorabeck CH, Taylor JW. Periprosthetic fractures of the femur complicating total knee arthroplasty. Orthop Clin North Am. 1999;30(2):265-277.
5. Hirsh DM, Bhalla S, Roffman M. Supracondylar fracture of the femur following total knee replacement. Report of four cases. J Bone Joint Surg Am. 1981;63(1):162-163.
6. Ritter MA, Thong AE, Keating EM, et al. The effect of femoral notching during total knee arthroplasty on the prevalence of postoperative femoral fractures and on clinical outcome. J Bone Joint Surg Am. 2005;87(11):2411-2414.
7. Lesh ML, Schneider DJ, Deol G, Davis B, Jacobs CR, Pellegrini VD Jr. The consequences of anterior femoral notching in total knee arthroplasty. A biomechanical study. J Bone Joint Surg Am. 2000;82(8):1096-1101.
8. Shawen SB, Belmont PJ Jr, Klemme WR, Topoleski LD, Xenos JS, Orchowski JR. Osteoporosis and anterior femoral notching in periprosthetic supracondylar femoral fractures: a biomechanical analysis. J Bone Joint Surg Am. 2003;85(1):115-121.
9. Scuderi GR, Pagnano MW. Review article: the rationale for posterior cruciate substituting total knee arthroplasty. J Orthop Surg (Hong Kong). 2001;9(2):81-88.
10. Verra WC, van den Boom LG, Jacobs W, Clement DJ, Wymenga AA, Nelissen RG. Retention versus sacrifice of the posterior cruciate ligament in total knee arthroplasty for treating osteoarthritis. Cochrane Database Syst Rev. 2013;10:CD004803.
11. Kolisek FR, McGrath MS, Marker DR, et al. Posterior-stabilized versus posterior cruciate ligament-retaining total knee arthroplasty. Iowa Orthop J. 2009;29:23-27.
12. Dennis DA. Periprosthetic fractures following total knee arthroplasty. Instr Course Lect. 2001;50:379-389.
13. Cristofolini L, Viceconti M, Cappello A, Toni A. Mechanical validation of whole bone composite femur models. J Biomech. 1996;29(4):525-535.
14. Heiner AD, Brown TD. Structural properties of a new design of composite replicate femurs and tibias. J Biomech. 2001;34(6):773-781.
15. Beals RK, Tower SS. Periprosthetic fractures of the femur. An analysis of 93 fractures. Clin Orthop Relat Res. 1996;(327):238-246.
16. Gujarathi N, Putti AB, Abboud RJ, MacLean JG, Espley AJ, Kellett CF. Risk of periprosthetic fracture after anterior femoral notching. Acta Orthop. 2009;80(5):553-556.
17. Zalzal P, Backstein D, Gross AE, Papini M. Notching of the anterior femoral cortex during total knee arthroplasty: characteristics that increase local stresses. J Arthroplasty. 2006;21(5):737-743.
18. Gaski GE, Scully SP. In brief: classifications in brief: Vancouver classification of postoperative periprosthetic femur fractures. Clin Orthop Relat Res. 2011;469(5):1507-1510.
Although rare, periprosthetic fractures remain a significant complication after total knee arthroplasty (TKA), occurring in 0.3% to 2.5% of cases.1-4 Hirsh and colleagues5 were among the first to suggest that anterior femoral notching during TKA was a potential risk factor for postoperative periprosthetic femoral fracture because notching may weaken the anterior femoral cortex. Anterior femoral notching, a cortex violation occurring during an anterior bone cut, occurs in up to 30% of cases.6 Using a theoretical biomechanical model, Culp and colleagues1 found that increasing the depth of the notch defect into the cortex led to reduced torsional strength. In more recent, cadaveric biomechanical studies, notching of the anterior femoral cortex decreased torsional strength by up to 39%.7,8 Contrary to these biomechanical studies, a retrospective study evaluating 1089 TKAs using 2 implant designs (Anatomic Graduated Component, Biomet and Legacy, Zimmer) demonstrated no significant effect of anterior femoral notching with respect to incidence of supracondylar femur fractures.6 That study, however, did not address whether implant design is associated with a differential risk for fracture in the presence of anterior notching.
Previous biomechanical studies have primarily investigated cruciate-retaining (CR) femoral components and properties with respect to anterior notching, even though the posterior-stabilized (PS) design is used more often in the United States.1,7 According to a Mayo Clinic survey, TKAs with a PS design increased from <10% in 1990 to almost 75% by 1997.9 Today, there is little to no consensus about which implant is better, and use of one or the other depends largely on the surgeon and varies widely between countries and regions.10 PS designs require more bone resection and demonstrate prosthesis-controlled rollback during flexion, whereas CR designs preserve more bone and achieve posterior stabilization via the posterior cruciate ligament.11 Despite these differences in design and mechanics, a 2013 Cochrane review of TKA design found no clinically significant differences between CR and PS with respect to pain, range of motion, or clinical and radiologic outcomes.10 The reviewers did not specifically address periprosthetic fractures associated with either femoral notching or TKA design, as they could not quantitatively analyze postoperative complications because of the diversity of reports. Given the limited number of reported cases, a review of radiographic findings pertaining to the characteristics of supracondylar fractures in anterior femoral notching was unsuccessful.12 As the previous biomechanical studies of anterior notching used primarily CR models or no prostheses at all, a study of biomechanical differences between CR and PS designs in the presence of anterior notching is warranted.1,7,8 Therefore, we conducted a study to assess the effect of anterior femoral notching on torsional strength and load to failure in CR and PS femoral components.
Materials and Methods
Twelve fourth-generation composite adult left femur synthetic sawbones (Sawbones; Pacific Research Laboratories) were selected for their consistent biomechanical properties, vs those of cadaveric specimens; in addition, low intersample variability made them preferable to cadaveric bones given the small sample used in this study.13,14 All bones were from the same lot. All were visually inspected for defects and found to be acceptable. In each sample, an anterior cortical defect was created by making an anterior cut with an undersized (size 4) posterior referencing guide. In addition, the distance from the proximal end of the notch to the implant fell within 15 mm, as that is the maximum distance from the implant a notch can be placed using a standard femoral cutting jig.15 Six femora were instrumented with CR implants and 6 with PS implants (DePuy Synthes). Implants were placed using standardized cuts. Before testing, each implant was inspected for proper fit and found to be securely fastened to the femur. In addition, precision calipers were used to measure notch depth and distance from notch to implant before loading. A custom polymethylmethacrylate torsion jig was used to fix each instrumented femur proximally and distally on the femoral implant (Figure 1). Care was taken to ensure the distal jig engaged only the implant, thus isolating the notch as a stress riser. Each femur was loaded in external rotation through the proximal femoral jig along the anatomical axis. Use of external rotation was based on study findings implicating external rotation of the tibia as the most likely mechanism for generating a fracture in the event of a fall.12 Furthermore, distal femur fractures are predominantly spiral as opposed to butterfly or bending—an indication that torsion is the most likely mechanism of failure.16 With no axial rotation possible within the prosthesis, increased torsional stress is undoubtedly generated within adjacent bone. Each specimen underwent torsional stiffness testing and then load to failure. Torsional stiffness was measured by slowly loading each femur in external rotation, from 1 to 18 Nm for 3 cycles at a displacement rate of 0.5° per second. Each specimen then underwent torsional load-to-failure testing on an Instron 5800R machine at a rate of 0.5° per second. Failure was defined as the moment of fracture and subsequent decrease in torsional load—determined graphically by the peak torsional load followed immediately by a sharp decrease in load. Stiffness was determined as the slope of torque to the displacement curve for each cycle, and torque to failure was the highest recorded torque before fracture. Fracture pattern was noted after failure. A sample size of 6 specimens per group provided 80% power to detect a between-group difference of 1 Nm per degree in stiffness, using an estimated SD of 0.7 Nm per degree. In our statistical analysis, continuous variables are reported as means and SDs. Data from our torsional stiffness and load-to-failure testing were analyzed with unpaired 2-sample t tests, and P < .05 was considered statistically significant.
Results
We did not detect a statistical difference in notch depth, notch-to-implant distance, or femoral length between the CR and PS groups. Mean (SD) notch depth was 6.0 (1.3) mm for CR and 4.9 (1.0) mm for PS (P = .13); mean (SD) distance from the proximal end of the notch to the implant was 13.8 (1.7) mm for CR and 11.1 (3.2) mm for PS (P = .08); and mean (SD) femoral length was 46.2 (0.1) cm for CR and 46.2 (0.1) cm for PS (P = .60).
Mean (SD) torsional stiffness for the first 3 precycles was 6.2 (1.2), 8.7 (1.5), and 8.8 (1.4) Nm per degree for the CR group and 6.0 (0.7), 8.4 (1.4), and 8.6 (1.4) Nm per degree for the PS group; the differences were not statistically significant (Figure 2A). In addition, there were no statistically significant differences in mean (SD) stiffness at failure between CR, 6.5 (0.7) Nm per degree, and PS, 7.1 (0.9) Nm per degree (P = .24; Figure 2B) or in mean (SD) final torque at failure between CR, 62.4 (9.4) Nm, and PS, 62.7 (12.2) Nm (P = .95; Figure 2C).
All fractures in both groups were oblique fractures originating at the proximal angle of the notch and extended proximally. None extended distally into the box. Fracture locations and patterns were identical in the CR and PS groups of femurs (Figure 3).
Discussion
Periprosthetic fractures after TKA remain rare. However, these fractures can significantly increase morbidity and complications. Anterior femoral notching occurs inadvertently in 30% to 40% of TKAs.6,17 The impact of femoral notching on supracondylar femur fracture is inconsistent between biomechanical and retrospective clinical studies. Retrospective studies failed to find a significant correlation between anterior femoral notching and supracondylar femur fractures.6,17 However, findings of biomechanical studies have suggested that a notch 3 mm deep will reduce the torsional strength of the femur by 29%.7 Another study, using 3-dimensional finite element analysis, showed a significant increase in local stress with a notch deeper than 3 mm.15
To our knowledge, no clinical studies, including the aforementioned Cochrane review,10 have specifically evaluated the difference in risk for periprosthetic fracture between different TKA models in the presence of notching.11 The biomechanical differences between implant designs could be a confounding factor in the results of past studies. More bone resection is required in PS designs than in CR designs. The position of the PS intercondylar cutout, much lower than the top of the patella flange, should not increase susceptibility to fractures more than in CR designs, but this hypothesis, though accepted, has not been validated biomechanically or addressed specifically in prospective or retrospective clinical analysis. In the present study, we used a biomechanical model to replicate an external rotation failure mechanism and quantify the differences in torsional strength and load to failure between CR TKA and PS TKA models in the presence of anterior femoral notching. Our results showed no significant differences in torsional stiffness, stiffness at failure, or torque at failure between the CR and PS design groups in the presence of anterior femoral notching.
In this study, all femoral fractures were oblique, and they all originated at the site of the cortical defect, not the notch—a situation markedly different from having bending forces applied to the femur. Previous biomechanical data indicated that bending forces applied to a notched femur cause fractures originating at the notch, whereas torsional forces applied to a notched femur cause fractures originating at the anterior aspect of the bone–component interface.7 The difference is attributable to study design. Our femurs were held fixed at their proximal end, which may have exacerbated any bending forces applied during external rotation, but we thought constraining the proximal femur would better replicate a fall involving external rotation.
More important for our study, an oblique fracture pattern was noted for both design groups (CR and PS), indicating the fracture pattern was unrelated to the area from which bone was resected for the PS design. All femur fractures in both design groups occurred proximal to a well-fixed prosthesis, indicating they should be classified as Vancouver C fractures. This is significant because intercondylar fossa resection (PS group) did not convert the fractures into Vancouver B2 fractures, which involve prosthesis loosening caused by pericomponent fracture.18 This simple observation validated our hypothesis that there would be no biomechanical differences between CR and PS designs with respect to the effects of anterior femoral notching. This lack of a significant difference may be attributed to the PS intercondylar cutout being much lower than the top of the anterior flange shielding the resected bone deep to the anterior flange.7 In addition, given the rarity of supracondylar fractures and the lack of sufficient relevant clinical data, it is difficult to speculate on the fracture patterns observed in clinical cases versus biomechanical studies.12
The use of synthetic bone models instead of cadaveric specimens could be seen as a limitation. Although synthetic bones may not reproduce the mechanism of failure in living and cadaveric femurs, the mechanical properties of synthetic bones have previously been found to fall within the range of those of cadaveric bones under axial loading, bending, and torsion testing.13,14 As a uniform testing material, synthetic bones allow removal of the confounding variations in bone size and quality that plague biomechanical studies in cadaveric bones.13,14 Interfemoral variability was 20 to 200 times higher in cadaveric femurs than in synthetic bones, which makes synthetic femurs preferable to cadaveric femurs, especially in studies with a small sample size.13,14 In addition, a uniform specimen provides consistent, reproducible osteotomies, which were crucial for consistent mechanical evaluation of each configuration in this study.
The long-term clinical significance of anterior femoral notching in periprosthetic fractures is equivocal, possibly because most studies predominantly use CR implants.6 This may not be an issue if it is shown that CR and PS implants have the same mechanical properties. Despite the differences between clinical studies and our biomechanical study, reevaluation of clinical data is not warranted given the biomechanical data we present here. Results of biomechanical studies like ours still suggest an increased immediate postoperative risk for supracondylar fracture after anterior cortical notching of the femur.5,7 Ultimately, this study found that, compared with a CR design, a PS design did not alter the torsional biomechanical properties or fracture pattern of an anteriorly notched femur.
Although rare, periprosthetic fractures remain a significant complication after total knee arthroplasty (TKA), occurring in 0.3% to 2.5% of cases.1-4 Hirsh and colleagues5 were among the first to suggest that anterior femoral notching during TKA was a potential risk factor for postoperative periprosthetic femoral fracture because notching may weaken the anterior femoral cortex. Anterior femoral notching, a cortex violation occurring during an anterior bone cut, occurs in up to 30% of cases.6 Using a theoretical biomechanical model, Culp and colleagues1 found that increasing the depth of the notch defect into the cortex led to reduced torsional strength. In more recent, cadaveric biomechanical studies, notching of the anterior femoral cortex decreased torsional strength by up to 39%.7,8 Contrary to these biomechanical studies, a retrospective study evaluating 1089 TKAs using 2 implant designs (Anatomic Graduated Component, Biomet and Legacy, Zimmer) demonstrated no significant effect of anterior femoral notching with respect to incidence of supracondylar femur fractures.6 That study, however, did not address whether implant design is associated with a differential risk for fracture in the presence of anterior notching.
Previous biomechanical studies have primarily investigated cruciate-retaining (CR) femoral components and properties with respect to anterior notching, even though the posterior-stabilized (PS) design is used more often in the United States.1,7 According to a Mayo Clinic survey, TKAs with a PS design increased from <10% in 1990 to almost 75% by 1997.9 Today, there is little to no consensus about which implant is better, and use of one or the other depends largely on the surgeon and varies widely between countries and regions.10 PS designs require more bone resection and demonstrate prosthesis-controlled rollback during flexion, whereas CR designs preserve more bone and achieve posterior stabilization via the posterior cruciate ligament.11 Despite these differences in design and mechanics, a 2013 Cochrane review of TKA design found no clinically significant differences between CR and PS with respect to pain, range of motion, or clinical and radiologic outcomes.10 The reviewers did not specifically address periprosthetic fractures associated with either femoral notching or TKA design, as they could not quantitatively analyze postoperative complications because of the diversity of reports. Given the limited number of reported cases, a review of radiographic findings pertaining to the characteristics of supracondylar fractures in anterior femoral notching was unsuccessful.12 As the previous biomechanical studies of anterior notching used primarily CR models or no prostheses at all, a study of biomechanical differences between CR and PS designs in the presence of anterior notching is warranted.1,7,8 Therefore, we conducted a study to assess the effect of anterior femoral notching on torsional strength and load to failure in CR and PS femoral components.
Materials and Methods
Twelve fourth-generation composite adult left femur synthetic sawbones (Sawbones; Pacific Research Laboratories) were selected for their consistent biomechanical properties, vs those of cadaveric specimens; in addition, low intersample variability made them preferable to cadaveric bones given the small sample used in this study.13,14 All bones were from the same lot. All were visually inspected for defects and found to be acceptable. In each sample, an anterior cortical defect was created by making an anterior cut with an undersized (size 4) posterior referencing guide. In addition, the distance from the proximal end of the notch to the implant fell within 15 mm, as that is the maximum distance from the implant a notch can be placed using a standard femoral cutting jig.15 Six femora were instrumented with CR implants and 6 with PS implants (DePuy Synthes). Implants were placed using standardized cuts. Before testing, each implant was inspected for proper fit and found to be securely fastened to the femur. In addition, precision calipers were used to measure notch depth and distance from notch to implant before loading. A custom polymethylmethacrylate torsion jig was used to fix each instrumented femur proximally and distally on the femoral implant (Figure 1). Care was taken to ensure the distal jig engaged only the implant, thus isolating the notch as a stress riser. Each femur was loaded in external rotation through the proximal femoral jig along the anatomical axis. Use of external rotation was based on study findings implicating external rotation of the tibia as the most likely mechanism for generating a fracture in the event of a fall.12 Furthermore, distal femur fractures are predominantly spiral as opposed to butterfly or bending—an indication that torsion is the most likely mechanism of failure.16 With no axial rotation possible within the prosthesis, increased torsional stress is undoubtedly generated within adjacent bone. Each specimen underwent torsional stiffness testing and then load to failure. Torsional stiffness was measured by slowly loading each femur in external rotation, from 1 to 18 Nm for 3 cycles at a displacement rate of 0.5° per second. Each specimen then underwent torsional load-to-failure testing on an Instron 5800R machine at a rate of 0.5° per second. Failure was defined as the moment of fracture and subsequent decrease in torsional load—determined graphically by the peak torsional load followed immediately by a sharp decrease in load. Stiffness was determined as the slope of torque to the displacement curve for each cycle, and torque to failure was the highest recorded torque before fracture. Fracture pattern was noted after failure. A sample size of 6 specimens per group provided 80% power to detect a between-group difference of 1 Nm per degree in stiffness, using an estimated SD of 0.7 Nm per degree. In our statistical analysis, continuous variables are reported as means and SDs. Data from our torsional stiffness and load-to-failure testing were analyzed with unpaired 2-sample t tests, and P < .05 was considered statistically significant.
Results
We did not detect a statistical difference in notch depth, notch-to-implant distance, or femoral length between the CR and PS groups. Mean (SD) notch depth was 6.0 (1.3) mm for CR and 4.9 (1.0) mm for PS (P = .13); mean (SD) distance from the proximal end of the notch to the implant was 13.8 (1.7) mm for CR and 11.1 (3.2) mm for PS (P = .08); and mean (SD) femoral length was 46.2 (0.1) cm for CR and 46.2 (0.1) cm for PS (P = .60).
Mean (SD) torsional stiffness for the first 3 precycles was 6.2 (1.2), 8.7 (1.5), and 8.8 (1.4) Nm per degree for the CR group and 6.0 (0.7), 8.4 (1.4), and 8.6 (1.4) Nm per degree for the PS group; the differences were not statistically significant (Figure 2A). In addition, there were no statistically significant differences in mean (SD) stiffness at failure between CR, 6.5 (0.7) Nm per degree, and PS, 7.1 (0.9) Nm per degree (P = .24; Figure 2B) or in mean (SD) final torque at failure between CR, 62.4 (9.4) Nm, and PS, 62.7 (12.2) Nm (P = .95; Figure 2C).
All fractures in both groups were oblique fractures originating at the proximal angle of the notch and extended proximally. None extended distally into the box. Fracture locations and patterns were identical in the CR and PS groups of femurs (Figure 3).
Discussion
Periprosthetic fractures after TKA remain rare. However, these fractures can significantly increase morbidity and complications. Anterior femoral notching occurs inadvertently in 30% to 40% of TKAs.6,17 The impact of femoral notching on supracondylar femur fracture is inconsistent between biomechanical and retrospective clinical studies. Retrospective studies failed to find a significant correlation between anterior femoral notching and supracondylar femur fractures.6,17 However, findings of biomechanical studies have suggested that a notch 3 mm deep will reduce the torsional strength of the femur by 29%.7 Another study, using 3-dimensional finite element analysis, showed a significant increase in local stress with a notch deeper than 3 mm.15
To our knowledge, no clinical studies, including the aforementioned Cochrane review,10 have specifically evaluated the difference in risk for periprosthetic fracture between different TKA models in the presence of notching.11 The biomechanical differences between implant designs could be a confounding factor in the results of past studies. More bone resection is required in PS designs than in CR designs. The position of the PS intercondylar cutout, much lower than the top of the patella flange, should not increase susceptibility to fractures more than in CR designs, but this hypothesis, though accepted, has not been validated biomechanically or addressed specifically in prospective or retrospective clinical analysis. In the present study, we used a biomechanical model to replicate an external rotation failure mechanism and quantify the differences in torsional strength and load to failure between CR TKA and PS TKA models in the presence of anterior femoral notching. Our results showed no significant differences in torsional stiffness, stiffness at failure, or torque at failure between the CR and PS design groups in the presence of anterior femoral notching.
In this study, all femoral fractures were oblique, and they all originated at the site of the cortical defect, not the notch—a situation markedly different from having bending forces applied to the femur. Previous biomechanical data indicated that bending forces applied to a notched femur cause fractures originating at the notch, whereas torsional forces applied to a notched femur cause fractures originating at the anterior aspect of the bone–component interface.7 The difference is attributable to study design. Our femurs were held fixed at their proximal end, which may have exacerbated any bending forces applied during external rotation, but we thought constraining the proximal femur would better replicate a fall involving external rotation.
More important for our study, an oblique fracture pattern was noted for both design groups (CR and PS), indicating the fracture pattern was unrelated to the area from which bone was resected for the PS design. All femur fractures in both design groups occurred proximal to a well-fixed prosthesis, indicating they should be classified as Vancouver C fractures. This is significant because intercondylar fossa resection (PS group) did not convert the fractures into Vancouver B2 fractures, which involve prosthesis loosening caused by pericomponent fracture.18 This simple observation validated our hypothesis that there would be no biomechanical differences between CR and PS designs with respect to the effects of anterior femoral notching. This lack of a significant difference may be attributed to the PS intercondylar cutout being much lower than the top of the anterior flange shielding the resected bone deep to the anterior flange.7 In addition, given the rarity of supracondylar fractures and the lack of sufficient relevant clinical data, it is difficult to speculate on the fracture patterns observed in clinical cases versus biomechanical studies.12
The use of synthetic bone models instead of cadaveric specimens could be seen as a limitation. Although synthetic bones may not reproduce the mechanism of failure in living and cadaveric femurs, the mechanical properties of synthetic bones have previously been found to fall within the range of those of cadaveric bones under axial loading, bending, and torsion testing.13,14 As a uniform testing material, synthetic bones allow removal of the confounding variations in bone size and quality that plague biomechanical studies in cadaveric bones.13,14 Interfemoral variability was 20 to 200 times higher in cadaveric femurs than in synthetic bones, which makes synthetic femurs preferable to cadaveric femurs, especially in studies with a small sample size.13,14 In addition, a uniform specimen provides consistent, reproducible osteotomies, which were crucial for consistent mechanical evaluation of each configuration in this study.
The long-term clinical significance of anterior femoral notching in periprosthetic fractures is equivocal, possibly because most studies predominantly use CR implants.6 This may not be an issue if it is shown that CR and PS implants have the same mechanical properties. Despite the differences between clinical studies and our biomechanical study, reevaluation of clinical data is not warranted given the biomechanical data we present here. Results of biomechanical studies like ours still suggest an increased immediate postoperative risk for supracondylar fracture after anterior cortical notching of the femur.5,7 Ultimately, this study found that, compared with a CR design, a PS design did not alter the torsional biomechanical properties or fracture pattern of an anteriorly notched femur.
1. Culp RW, Schmidt RG, Hanks G, Mak A, Esterhai JL Jr, Heppenstall RB. Supracondylar fracture of the femur following prosthetic knee arthroplasty. Clin Orthop Relat Res. 1987;(222):212-222.
2. Delport PH, Van Audekercke R, Martens M, Mulier JC. Conservative treatment of ipsilateral supracondylar femoral fracture after total knee arthroplasty. J Trauma. 1984;24(9):846-849.
3. Figgie MP, Goldberg VM, Figgie HE 3rd, Sobel M. The results of treatment of supracondylar fracture above total knee arthroplasty. J Arthroplasty. 1990;5(3):267-276.
4. Rorabeck CH, Taylor JW. Periprosthetic fractures of the femur complicating total knee arthroplasty. Orthop Clin North Am. 1999;30(2):265-277.
5. Hirsh DM, Bhalla S, Roffman M. Supracondylar fracture of the femur following total knee replacement. Report of four cases. J Bone Joint Surg Am. 1981;63(1):162-163.
6. Ritter MA, Thong AE, Keating EM, et al. The effect of femoral notching during total knee arthroplasty on the prevalence of postoperative femoral fractures and on clinical outcome. J Bone Joint Surg Am. 2005;87(11):2411-2414.
7. Lesh ML, Schneider DJ, Deol G, Davis B, Jacobs CR, Pellegrini VD Jr. The consequences of anterior femoral notching in total knee arthroplasty. A biomechanical study. J Bone Joint Surg Am. 2000;82(8):1096-1101.
8. Shawen SB, Belmont PJ Jr, Klemme WR, Topoleski LD, Xenos JS, Orchowski JR. Osteoporosis and anterior femoral notching in periprosthetic supracondylar femoral fractures: a biomechanical analysis. J Bone Joint Surg Am. 2003;85(1):115-121.
9. Scuderi GR, Pagnano MW. Review article: the rationale for posterior cruciate substituting total knee arthroplasty. J Orthop Surg (Hong Kong). 2001;9(2):81-88.
10. Verra WC, van den Boom LG, Jacobs W, Clement DJ, Wymenga AA, Nelissen RG. Retention versus sacrifice of the posterior cruciate ligament in total knee arthroplasty for treating osteoarthritis. Cochrane Database Syst Rev. 2013;10:CD004803.
11. Kolisek FR, McGrath MS, Marker DR, et al. Posterior-stabilized versus posterior cruciate ligament-retaining total knee arthroplasty. Iowa Orthop J. 2009;29:23-27.
12. Dennis DA. Periprosthetic fractures following total knee arthroplasty. Instr Course Lect. 2001;50:379-389.
13. Cristofolini L, Viceconti M, Cappello A, Toni A. Mechanical validation of whole bone composite femur models. J Biomech. 1996;29(4):525-535.
14. Heiner AD, Brown TD. Structural properties of a new design of composite replicate femurs and tibias. J Biomech. 2001;34(6):773-781.
15. Beals RK, Tower SS. Periprosthetic fractures of the femur. An analysis of 93 fractures. Clin Orthop Relat Res. 1996;(327):238-246.
16. Gujarathi N, Putti AB, Abboud RJ, MacLean JG, Espley AJ, Kellett CF. Risk of periprosthetic fracture after anterior femoral notching. Acta Orthop. 2009;80(5):553-556.
17. Zalzal P, Backstein D, Gross AE, Papini M. Notching of the anterior femoral cortex during total knee arthroplasty: characteristics that increase local stresses. J Arthroplasty. 2006;21(5):737-743.
18. Gaski GE, Scully SP. In brief: classifications in brief: Vancouver classification of postoperative periprosthetic femur fractures. Clin Orthop Relat Res. 2011;469(5):1507-1510.
1. Culp RW, Schmidt RG, Hanks G, Mak A, Esterhai JL Jr, Heppenstall RB. Supracondylar fracture of the femur following prosthetic knee arthroplasty. Clin Orthop Relat Res. 1987;(222):212-222.
2. Delport PH, Van Audekercke R, Martens M, Mulier JC. Conservative treatment of ipsilateral supracondylar femoral fracture after total knee arthroplasty. J Trauma. 1984;24(9):846-849.
3. Figgie MP, Goldberg VM, Figgie HE 3rd, Sobel M. The results of treatment of supracondylar fracture above total knee arthroplasty. J Arthroplasty. 1990;5(3):267-276.
4. Rorabeck CH, Taylor JW. Periprosthetic fractures of the femur complicating total knee arthroplasty. Orthop Clin North Am. 1999;30(2):265-277.
5. Hirsh DM, Bhalla S, Roffman M. Supracondylar fracture of the femur following total knee replacement. Report of four cases. J Bone Joint Surg Am. 1981;63(1):162-163.
6. Ritter MA, Thong AE, Keating EM, et al. The effect of femoral notching during total knee arthroplasty on the prevalence of postoperative femoral fractures and on clinical outcome. J Bone Joint Surg Am. 2005;87(11):2411-2414.
7. Lesh ML, Schneider DJ, Deol G, Davis B, Jacobs CR, Pellegrini VD Jr. The consequences of anterior femoral notching in total knee arthroplasty. A biomechanical study. J Bone Joint Surg Am. 2000;82(8):1096-1101.
8. Shawen SB, Belmont PJ Jr, Klemme WR, Topoleski LD, Xenos JS, Orchowski JR. Osteoporosis and anterior femoral notching in periprosthetic supracondylar femoral fractures: a biomechanical analysis. J Bone Joint Surg Am. 2003;85(1):115-121.
9. Scuderi GR, Pagnano MW. Review article: the rationale for posterior cruciate substituting total knee arthroplasty. J Orthop Surg (Hong Kong). 2001;9(2):81-88.
10. Verra WC, van den Boom LG, Jacobs W, Clement DJ, Wymenga AA, Nelissen RG. Retention versus sacrifice of the posterior cruciate ligament in total knee arthroplasty for treating osteoarthritis. Cochrane Database Syst Rev. 2013;10:CD004803.
11. Kolisek FR, McGrath MS, Marker DR, et al. Posterior-stabilized versus posterior cruciate ligament-retaining total knee arthroplasty. Iowa Orthop J. 2009;29:23-27.
12. Dennis DA. Periprosthetic fractures following total knee arthroplasty. Instr Course Lect. 2001;50:379-389.
13. Cristofolini L, Viceconti M, Cappello A, Toni A. Mechanical validation of whole bone composite femur models. J Biomech. 1996;29(4):525-535.
14. Heiner AD, Brown TD. Structural properties of a new design of composite replicate femurs and tibias. J Biomech. 2001;34(6):773-781.
15. Beals RK, Tower SS. Periprosthetic fractures of the femur. An analysis of 93 fractures. Clin Orthop Relat Res. 1996;(327):238-246.
16. Gujarathi N, Putti AB, Abboud RJ, MacLean JG, Espley AJ, Kellett CF. Risk of periprosthetic fracture after anterior femoral notching. Acta Orthop. 2009;80(5):553-556.
17. Zalzal P, Backstein D, Gross AE, Papini M. Notching of the anterior femoral cortex during total knee arthroplasty: characteristics that increase local stresses. J Arthroplasty. 2006;21(5):737-743.
18. Gaski GE, Scully SP. In brief: classifications in brief: Vancouver classification of postoperative periprosthetic femur fractures. Clin Orthop Relat Res. 2011;469(5):1507-1510.
Sleep doctor: Less than 7 hours can worsen diabetes
SAN DIEGO – A sleep specialist told an audience of diabetes educators that quality sleep is “profoundly important” to the health of their patients, and regularly sleeping fewer than 7 hours a night can wreak havoc on glucose levels and insulin resistance.
There’s even evidence of a link between lack of sleep, diabetes, and heart disease, according to Terese C. Hammond, MD, medical director of the Keck Hospital of the University of Southern California Sleep Disorders Center in Los Angeles. “We don’t know which way the cause goes, but we know there’s a connection,” she said.
Dr. Hammond, who spoke at the annual meeting of the American Association of Diabetes Educators, noted that the vast majority of adults need 7-9 hours of sleep a night.
If you repeatedly don’t reach 7 hours, “there is ample evidence that some metabolic and biologic things happen to your body. Glucose goes up and insulin is secreted. Leptin decreases – the protein that tells us when we’re sated, when our hunger is satisfied. It’s a pretty potent combination, and this is associated with increases in weight and carbohydrate intake,” Dr. Hammond said.
Indeed, findings from one small study suggest a link between chronic insufficient sleep to consumption of more calories, mostly carbohydrates. The study authors speculated this may be because people eat extra food to account for being awake more but take in more than they need (Proc Natl Acad Sci USA. 2013 Apr;110[14]:5695-700).
It is possible for people to pay off a “sleep debt” by catching up on sleep, Dr. Hammond said, but only if the sleep is lost temporarily, as during a heavy medical rotation or after a personal tragedy. When sleep loss is chronic, she said, “it’s becoming apparent that the end-organ consequences are not fully recoverable.”
In regard to diabetes specifically, she said, research has linked insufficient sleep to higher rates of central obesity, higher rates of diabetes in blacks and whites, impaired glucose tolerance, decreased insulin, metabolic syndrome, and high lipids. Too much sleep has also been linked to poor health outcomes.
How can medical professionals help patients improve sleep? “The most important thing to remember about sleep is that it’s a very primitive process,” according to Dr. Hammond. “You can’t think yourself into better sleep. You have to act yourself into better sleep through light, temperature, sound, and repetitive behaviors.”
Controversial research has linked sleeping pills to higher mortality rates, she says.“I try to avoid them long term for insomnia, and not only because there’s probably an increased risk. They stop working eventually.”
Another approach, cognitive behavioral therapy (CBT), is “the most potent way to change behavior,” she said.
“CBT improves sleep efficiency in a vast majority of chronic medical diseases,” she said. “It does just as well as drugs and better than most sleeping pills.”
According to Dr. Hammond, CBT focuses on several strategies:
• Education about sleep hygiene, such as limiting the bedroom to nighttime sleep and intimacy, going to bed at the same time every night, and focusing on a dark, cool, quiet environment.
• Increasing the “sleep drive” through temporary sleep deprivation.
• Relaxation training through techniques like guided imagery, biofeedback, progressive muscle relaxation, and self-hypnosis.
Dr. Hammond reported having no relevant financial disclosures.
SAN DIEGO – A sleep specialist told an audience of diabetes educators that quality sleep is “profoundly important” to the health of their patients, and regularly sleeping fewer than 7 hours a night can wreak havoc on glucose levels and insulin resistance.
There’s even evidence of a link between lack of sleep, diabetes, and heart disease, according to Terese C. Hammond, MD, medical director of the Keck Hospital of the University of Southern California Sleep Disorders Center in Los Angeles. “We don’t know which way the cause goes, but we know there’s a connection,” she said.
Dr. Hammond, who spoke at the annual meeting of the American Association of Diabetes Educators, noted that the vast majority of adults need 7-9 hours of sleep a night.
If you repeatedly don’t reach 7 hours, “there is ample evidence that some metabolic and biologic things happen to your body. Glucose goes up and insulin is secreted. Leptin decreases – the protein that tells us when we’re sated, when our hunger is satisfied. It’s a pretty potent combination, and this is associated with increases in weight and carbohydrate intake,” Dr. Hammond said.
Indeed, findings from one small study suggest a link between chronic insufficient sleep to consumption of more calories, mostly carbohydrates. The study authors speculated this may be because people eat extra food to account for being awake more but take in more than they need (Proc Natl Acad Sci USA. 2013 Apr;110[14]:5695-700).
It is possible for people to pay off a “sleep debt” by catching up on sleep, Dr. Hammond said, but only if the sleep is lost temporarily, as during a heavy medical rotation or after a personal tragedy. When sleep loss is chronic, she said, “it’s becoming apparent that the end-organ consequences are not fully recoverable.”
In regard to diabetes specifically, she said, research has linked insufficient sleep to higher rates of central obesity, higher rates of diabetes in blacks and whites, impaired glucose tolerance, decreased insulin, metabolic syndrome, and high lipids. Too much sleep has also been linked to poor health outcomes.
How can medical professionals help patients improve sleep? “The most important thing to remember about sleep is that it’s a very primitive process,” according to Dr. Hammond. “You can’t think yourself into better sleep. You have to act yourself into better sleep through light, temperature, sound, and repetitive behaviors.”
Controversial research has linked sleeping pills to higher mortality rates, she says.“I try to avoid them long term for insomnia, and not only because there’s probably an increased risk. They stop working eventually.”
Another approach, cognitive behavioral therapy (CBT), is “the most potent way to change behavior,” she said.
“CBT improves sleep efficiency in a vast majority of chronic medical diseases,” she said. “It does just as well as drugs and better than most sleeping pills.”
According to Dr. Hammond, CBT focuses on several strategies:
• Education about sleep hygiene, such as limiting the bedroom to nighttime sleep and intimacy, going to bed at the same time every night, and focusing on a dark, cool, quiet environment.
• Increasing the “sleep drive” through temporary sleep deprivation.
• Relaxation training through techniques like guided imagery, biofeedback, progressive muscle relaxation, and self-hypnosis.
Dr. Hammond reported having no relevant financial disclosures.
SAN DIEGO – A sleep specialist told an audience of diabetes educators that quality sleep is “profoundly important” to the health of their patients, and regularly sleeping fewer than 7 hours a night can wreak havoc on glucose levels and insulin resistance.
There’s even evidence of a link between lack of sleep, diabetes, and heart disease, according to Terese C. Hammond, MD, medical director of the Keck Hospital of the University of Southern California Sleep Disorders Center in Los Angeles. “We don’t know which way the cause goes, but we know there’s a connection,” she said.
Dr. Hammond, who spoke at the annual meeting of the American Association of Diabetes Educators, noted that the vast majority of adults need 7-9 hours of sleep a night.
If you repeatedly don’t reach 7 hours, “there is ample evidence that some metabolic and biologic things happen to your body. Glucose goes up and insulin is secreted. Leptin decreases – the protein that tells us when we’re sated, when our hunger is satisfied. It’s a pretty potent combination, and this is associated with increases in weight and carbohydrate intake,” Dr. Hammond said.
Indeed, findings from one small study suggest a link between chronic insufficient sleep to consumption of more calories, mostly carbohydrates. The study authors speculated this may be because people eat extra food to account for being awake more but take in more than they need (Proc Natl Acad Sci USA. 2013 Apr;110[14]:5695-700).
It is possible for people to pay off a “sleep debt” by catching up on sleep, Dr. Hammond said, but only if the sleep is lost temporarily, as during a heavy medical rotation or after a personal tragedy. When sleep loss is chronic, she said, “it’s becoming apparent that the end-organ consequences are not fully recoverable.”
In regard to diabetes specifically, she said, research has linked insufficient sleep to higher rates of central obesity, higher rates of diabetes in blacks and whites, impaired glucose tolerance, decreased insulin, metabolic syndrome, and high lipids. Too much sleep has also been linked to poor health outcomes.
How can medical professionals help patients improve sleep? “The most important thing to remember about sleep is that it’s a very primitive process,” according to Dr. Hammond. “You can’t think yourself into better sleep. You have to act yourself into better sleep through light, temperature, sound, and repetitive behaviors.”
Controversial research has linked sleeping pills to higher mortality rates, she says.“I try to avoid them long term for insomnia, and not only because there’s probably an increased risk. They stop working eventually.”
Another approach, cognitive behavioral therapy (CBT), is “the most potent way to change behavior,” she said.
“CBT improves sleep efficiency in a vast majority of chronic medical diseases,” she said. “It does just as well as drugs and better than most sleeping pills.”
According to Dr. Hammond, CBT focuses on several strategies:
• Education about sleep hygiene, such as limiting the bedroom to nighttime sleep and intimacy, going to bed at the same time every night, and focusing on a dark, cool, quiet environment.
• Increasing the “sleep drive” through temporary sleep deprivation.
• Relaxation training through techniques like guided imagery, biofeedback, progressive muscle relaxation, and self-hypnosis.
Dr. Hammond reported having no relevant financial disclosures.
AT AADE 16
Predisposition to overlapping AIH and PBC can exist within families
A genetic predisposition to autoimmune liver disease can cause overlapping cases of autoimmune hepatitis (AIH) and primary biliary cholangitis (PBC) within the same family, according to a case report from Japanese researchers.
A 68-year-old woman and her 49-year-old daughter were admitted to the hospital for liver dysfunction, 4 years apart. The mother had been diagnosed with PBC at a previous hospital, but was moved after treatment with 600 mg ursodeoxycholic acid (UDCA) was ineffective. The daughter had no previous diagnosis and had a variety of symptoms at admission, including jaundice, general fatigue, and darkness of the urine.
Both the mother and the daughter were negative for hepatitis A, B, and C. A simplified International Autoimmune Hepatitis Group score revealed probable AIH for both patients, and both patients were diagnosed with autoimmune liver disease with overlapping features of PBC and AIH. Treatment with UDCA and prednisolone was effective for both patients.
In a familial study, the mother and her two daughters were tested for liver function, autoantibodies, and human leukocyte antigen (HLA) haplotype. The second daughter, who was healthy with no history of liver dysfunction, had normal liver function, negative ANA, and positive AMA-M2 antibody, but had an HLA haplotype different from that of the mother and the first daughter. No difference in medication use, smoking status, alcohol consumption, or obstetric history was seen.
“The exact mechanism underlying the onset of PBC and AIH overlap within the same family remains unclear. Therefore, it is very important to monitor the healthy second daughter closely as she was positive for the AMA-M2 antibody, as this might yield further knowledge with regard to what factors influence the onset of PBC and AIH overlap within the same family,” the investigators noted.
Find the full study in the Journal of Clinical Gastroenterology (doi: 10.1007/s12328-016-0676-1)
A genetic predisposition to autoimmune liver disease can cause overlapping cases of autoimmune hepatitis (AIH) and primary biliary cholangitis (PBC) within the same family, according to a case report from Japanese researchers.
A 68-year-old woman and her 49-year-old daughter were admitted to the hospital for liver dysfunction, 4 years apart. The mother had been diagnosed with PBC at a previous hospital, but was moved after treatment with 600 mg ursodeoxycholic acid (UDCA) was ineffective. The daughter had no previous diagnosis and had a variety of symptoms at admission, including jaundice, general fatigue, and darkness of the urine.
Both the mother and the daughter were negative for hepatitis A, B, and C. A simplified International Autoimmune Hepatitis Group score revealed probable AIH for both patients, and both patients were diagnosed with autoimmune liver disease with overlapping features of PBC and AIH. Treatment with UDCA and prednisolone was effective for both patients.
In a familial study, the mother and her two daughters were tested for liver function, autoantibodies, and human leukocyte antigen (HLA) haplotype. The second daughter, who was healthy with no history of liver dysfunction, had normal liver function, negative ANA, and positive AMA-M2 antibody, but had an HLA haplotype different from that of the mother and the first daughter. No difference in medication use, smoking status, alcohol consumption, or obstetric history was seen.
“The exact mechanism underlying the onset of PBC and AIH overlap within the same family remains unclear. Therefore, it is very important to monitor the healthy second daughter closely as she was positive for the AMA-M2 antibody, as this might yield further knowledge with regard to what factors influence the onset of PBC and AIH overlap within the same family,” the investigators noted.
Find the full study in the Journal of Clinical Gastroenterology (doi: 10.1007/s12328-016-0676-1)
A genetic predisposition to autoimmune liver disease can cause overlapping cases of autoimmune hepatitis (AIH) and primary biliary cholangitis (PBC) within the same family, according to a case report from Japanese researchers.
A 68-year-old woman and her 49-year-old daughter were admitted to the hospital for liver dysfunction, 4 years apart. The mother had been diagnosed with PBC at a previous hospital, but was moved after treatment with 600 mg ursodeoxycholic acid (UDCA) was ineffective. The daughter had no previous diagnosis and had a variety of symptoms at admission, including jaundice, general fatigue, and darkness of the urine.
Both the mother and the daughter were negative for hepatitis A, B, and C. A simplified International Autoimmune Hepatitis Group score revealed probable AIH for both patients, and both patients were diagnosed with autoimmune liver disease with overlapping features of PBC and AIH. Treatment with UDCA and prednisolone was effective for both patients.
In a familial study, the mother and her two daughters were tested for liver function, autoantibodies, and human leukocyte antigen (HLA) haplotype. The second daughter, who was healthy with no history of liver dysfunction, had normal liver function, negative ANA, and positive AMA-M2 antibody, but had an HLA haplotype different from that of the mother and the first daughter. No difference in medication use, smoking status, alcohol consumption, or obstetric history was seen.
“The exact mechanism underlying the onset of PBC and AIH overlap within the same family remains unclear. Therefore, it is very important to monitor the healthy second daughter closely as she was positive for the AMA-M2 antibody, as this might yield further knowledge with regard to what factors influence the onset of PBC and AIH overlap within the same family,” the investigators noted.
Find the full study in the Journal of Clinical Gastroenterology (doi: 10.1007/s12328-016-0676-1)
FROM THE JOURNAL OF CLINICAL GASTROENTEROLOGY
Inadequate diversity snags hypertrophic cardiomyopathy genetic linkages
The genetic tests used for more than a decade to identify patients or family members who carry genetic mutations linked to hypertrophic cardiomyopathy are seriously flawed.
The tests have been erroneously flagging people as genetically positive for hypertrophic cardiomyopathy (HC) when they actually carried benign genetic variants, according to a new reassessment of the genetic linkages by researchers using a more genetically diverse database. Five genetic variants now reclassified as benign were collectively responsible for flagging 74% of people flagged at genetic risk for HC in the more than 8,500 cases examined.
The results call into question any genetic diagnosis of HC made since genetic testing entered the mainstream in 2003, especially among African Americans who seem to have been disproportionately affected by these mislabeled genetic markers because of inadequate population diversity when the markers were first established.
In addition, the results more broadly cast a shadow over the full spectrum of genetic tests for disease-linked variants now in routine medical practice because of the possibility that other linkage determinations derived from an inadequately-representative reference population, reported Arjun K. Manrai, PhD, and his associates (N Engl J Med. 2016 Aug 18;375[7]:655-65). The researchers call the HC experience they document a “cautionary tale of broad relevance to genetic diagnosis.”
The findings “powerfully illustrate the importance of racial and genetic diversity” when running linkage studies aimed at validating genetic markers for widespread clinical use, Isaac S. Kohane, MD, senior author of the study, said in a written statement. “Racial and ethnic inclusiveness improves the validity and accuracy” of genetic tests, said Dr. Kohane, professor of biomedical informatics and pediatrics at Harvard Medical School in Boston.
“We believe that what we’re seeing in the case of hypertrophic cardiomyopathy may be the tip of the iceberg of a larger problem that transcends a single genetic disease,” Dr. Manrai, a biomedical informatics researcher at Harvard, said in the same statement. “Much genetic assessment today relies on historical links between a disorder and variant, sometimes decades old. We believe our findings illustrate the critical need to systematically reevaluate prior assertions about genetic variants,” Dr. Manrai added in an interview.
The two researchers and their associates reexamined the link between genetic variants and HC in three genetic databases that involved a total of more than 8,500 people. One database included 4,300 white Americans and 2,203 black Americans. A second database included genetic data from 1,092 people from 14 worldwide populations, and the third had genetic data from 938 people from 51 worldwide populations.
The analysis showed that although 94 distinct genetic variants that had previously been reported as associated with HC were confirmed as linked, just 5 met the study’s definition of a “high-frequency” variant with an allele frequent of more than 1%. These five variants together accounted for 74% of the overall total of linkages seen in these 8,533 people. Further analysis classified all five of these high-frequency genetic variants as benign with no discernible link to HC.
These five high-frequency variants occurred disproportionately higher among black Americans, and the consequences of this showed up in the patient records the researchers reviewed from one large U.S. genetic testing laboratory. They examined in detail HC genetic test results during 2004-2013 from 2,912 unrelated people. The records showed seven people had been labeled as carrying either a pathogenic or “presumed pathogenic” variant when in fact they had one of the five high-frequency variants now declared benign. Five of the seven mislabeled people were of African ancestry; the other two had unknown ancestry.
The researchers called for reevaluating known variants for all genetic diseases in more diverse populations and to immediately release the results of updated linkage assessments. This has the potential to meaningfully rewrite current gospel for many genetic variants and linkages.
“Our findings point to the value of patients staying in contact with their genetic counselors and physicians, even years after genetic testing,” Dr. Manrai said. Reassessments using more diverse populations will take time, he acknowledged, but tools are available to allow clinical geneticists to update old variants and apply new ones in real time, as soon as a new assessment completes.
Dr. Manrai and Dr. Kohane had no disclosures.
On Twitter @mitchelzoler
The genetic tests used for more than a decade to identify patients or family members who carry genetic mutations linked to hypertrophic cardiomyopathy are seriously flawed.
The tests have been erroneously flagging people as genetically positive for hypertrophic cardiomyopathy (HC) when they actually carried benign genetic variants, according to a new reassessment of the genetic linkages by researchers using a more genetically diverse database. Five genetic variants now reclassified as benign were collectively responsible for flagging 74% of people flagged at genetic risk for HC in the more than 8,500 cases examined.
The results call into question any genetic diagnosis of HC made since genetic testing entered the mainstream in 2003, especially among African Americans who seem to have been disproportionately affected by these mislabeled genetic markers because of inadequate population diversity when the markers were first established.
In addition, the results more broadly cast a shadow over the full spectrum of genetic tests for disease-linked variants now in routine medical practice because of the possibility that other linkage determinations derived from an inadequately-representative reference population, reported Arjun K. Manrai, PhD, and his associates (N Engl J Med. 2016 Aug 18;375[7]:655-65). The researchers call the HC experience they document a “cautionary tale of broad relevance to genetic diagnosis.”
The findings “powerfully illustrate the importance of racial and genetic diversity” when running linkage studies aimed at validating genetic markers for widespread clinical use, Isaac S. Kohane, MD, senior author of the study, said in a written statement. “Racial and ethnic inclusiveness improves the validity and accuracy” of genetic tests, said Dr. Kohane, professor of biomedical informatics and pediatrics at Harvard Medical School in Boston.
“We believe that what we’re seeing in the case of hypertrophic cardiomyopathy may be the tip of the iceberg of a larger problem that transcends a single genetic disease,” Dr. Manrai, a biomedical informatics researcher at Harvard, said in the same statement. “Much genetic assessment today relies on historical links between a disorder and variant, sometimes decades old. We believe our findings illustrate the critical need to systematically reevaluate prior assertions about genetic variants,” Dr. Manrai added in an interview.
The two researchers and their associates reexamined the link between genetic variants and HC in three genetic databases that involved a total of more than 8,500 people. One database included 4,300 white Americans and 2,203 black Americans. A second database included genetic data from 1,092 people from 14 worldwide populations, and the third had genetic data from 938 people from 51 worldwide populations.
The analysis showed that although 94 distinct genetic variants that had previously been reported as associated with HC were confirmed as linked, just 5 met the study’s definition of a “high-frequency” variant with an allele frequent of more than 1%. These five variants together accounted for 74% of the overall total of linkages seen in these 8,533 people. Further analysis classified all five of these high-frequency genetic variants as benign with no discernible link to HC.
These five high-frequency variants occurred disproportionately higher among black Americans, and the consequences of this showed up in the patient records the researchers reviewed from one large U.S. genetic testing laboratory. They examined in detail HC genetic test results during 2004-2013 from 2,912 unrelated people. The records showed seven people had been labeled as carrying either a pathogenic or “presumed pathogenic” variant when in fact they had one of the five high-frequency variants now declared benign. Five of the seven mislabeled people were of African ancestry; the other two had unknown ancestry.
The researchers called for reevaluating known variants for all genetic diseases in more diverse populations and to immediately release the results of updated linkage assessments. This has the potential to meaningfully rewrite current gospel for many genetic variants and linkages.
“Our findings point to the value of patients staying in contact with their genetic counselors and physicians, even years after genetic testing,” Dr. Manrai said. Reassessments using more diverse populations will take time, he acknowledged, but tools are available to allow clinical geneticists to update old variants and apply new ones in real time, as soon as a new assessment completes.
Dr. Manrai and Dr. Kohane had no disclosures.
On Twitter @mitchelzoler
The genetic tests used for more than a decade to identify patients or family members who carry genetic mutations linked to hypertrophic cardiomyopathy are seriously flawed.
The tests have been erroneously flagging people as genetically positive for hypertrophic cardiomyopathy (HC) when they actually carried benign genetic variants, according to a new reassessment of the genetic linkages by researchers using a more genetically diverse database. Five genetic variants now reclassified as benign were collectively responsible for flagging 74% of people flagged at genetic risk for HC in the more than 8,500 cases examined.
The results call into question any genetic diagnosis of HC made since genetic testing entered the mainstream in 2003, especially among African Americans who seem to have been disproportionately affected by these mislabeled genetic markers because of inadequate population diversity when the markers were first established.
In addition, the results more broadly cast a shadow over the full spectrum of genetic tests for disease-linked variants now in routine medical practice because of the possibility that other linkage determinations derived from an inadequately-representative reference population, reported Arjun K. Manrai, PhD, and his associates (N Engl J Med. 2016 Aug 18;375[7]:655-65). The researchers call the HC experience they document a “cautionary tale of broad relevance to genetic diagnosis.”
The findings “powerfully illustrate the importance of racial and genetic diversity” when running linkage studies aimed at validating genetic markers for widespread clinical use, Isaac S. Kohane, MD, senior author of the study, said in a written statement. “Racial and ethnic inclusiveness improves the validity and accuracy” of genetic tests, said Dr. Kohane, professor of biomedical informatics and pediatrics at Harvard Medical School in Boston.
“We believe that what we’re seeing in the case of hypertrophic cardiomyopathy may be the tip of the iceberg of a larger problem that transcends a single genetic disease,” Dr. Manrai, a biomedical informatics researcher at Harvard, said in the same statement. “Much genetic assessment today relies on historical links between a disorder and variant, sometimes decades old. We believe our findings illustrate the critical need to systematically reevaluate prior assertions about genetic variants,” Dr. Manrai added in an interview.
The two researchers and their associates reexamined the link between genetic variants and HC in three genetic databases that involved a total of more than 8,500 people. One database included 4,300 white Americans and 2,203 black Americans. A second database included genetic data from 1,092 people from 14 worldwide populations, and the third had genetic data from 938 people from 51 worldwide populations.
The analysis showed that although 94 distinct genetic variants that had previously been reported as associated with HC were confirmed as linked, just 5 met the study’s definition of a “high-frequency” variant with an allele frequent of more than 1%. These five variants together accounted for 74% of the overall total of linkages seen in these 8,533 people. Further analysis classified all five of these high-frequency genetic variants as benign with no discernible link to HC.
These five high-frequency variants occurred disproportionately higher among black Americans, and the consequences of this showed up in the patient records the researchers reviewed from one large U.S. genetic testing laboratory. They examined in detail HC genetic test results during 2004-2013 from 2,912 unrelated people. The records showed seven people had been labeled as carrying either a pathogenic or “presumed pathogenic” variant when in fact they had one of the five high-frequency variants now declared benign. Five of the seven mislabeled people were of African ancestry; the other two had unknown ancestry.
The researchers called for reevaluating known variants for all genetic diseases in more diverse populations and to immediately release the results of updated linkage assessments. This has the potential to meaningfully rewrite current gospel for many genetic variants and linkages.
“Our findings point to the value of patients staying in contact with their genetic counselors and physicians, even years after genetic testing,” Dr. Manrai said. Reassessments using more diverse populations will take time, he acknowledged, but tools are available to allow clinical geneticists to update old variants and apply new ones in real time, as soon as a new assessment completes.
Dr. Manrai and Dr. Kohane had no disclosures.
On Twitter @mitchelzoler
Key clinical point: Five high-frequency genetic variants that collectively had been linked to 74% of hypertrophic cardiomyopathy cases are actually benign with no detectable pathologic linkage.
Major finding: Inaccurate linkage data mislabeled seven people as having a hypertrophic cardiomyopathy–causing genetic variant.
Data source: Three genomic sequence databases that included 8,533 people and a genetic laboratory’s records for 2,912 clients.
Disclosures: Dr. Manrai and Dr. Kohane had no disclosures.
Multiarterial grafting survival exceeds conventional CABG, PCI
A large, 16-year single-center study of patients with multivessel disease has determined that multivessel coronary artery bypass grafting achieved longer survival than not only percutaneous coronary interventions, but also conventional coronary artery bypass grafting, researchers from the Mayo Clinic reported in the August issue of the Journal of Thoracic and Cardiovascular Surgery (J Thorac Cardiovasc Surg. 2016;152:369-79).
Lead author Chaim Locker, MD, and his colleagues said the use of what they called MultiArt, for multivessel arterial grafting, also known as MAG, “must increase.”
The evolution of bare-metal and then drug-eluting stents may have favored percutaneous coronary interventions (PCI) over coronary artery bypass grafting (CABG), but, Dr. Locker and his coauthors said, “Evidence is accumulating that late outcome of surgical revascularization is improved when at least two arterial grafts are used.”
The study analyzed results of 12,615 patients who had either isolated primary CABG (6,667) or PCI (5,948) from 1993 to 2009. Among the CABG patients, 5,712 had the more conventional approach involving arterial grafts into the left internal thoracic artery/saphenous vein (ITA/SV) and 955 had MAG. Patients in the PCI group had three different procedures: balloon angioplasty (1,020), drug-eluting stent (1,686), or bare-metal stent (3,242). The study excluded patients who had revascularization procedures after a heart attack.
While the overall 15-year survival for patients with CABG was lower than it was for those who had PCI (36% vs. 46%), the survival for those who had MAG was significantly higher: 65% vs. 31% for those who had left ITA/SV revascularization. 8-year survival for the MAG subgroup was also significantly higher than all other subgroups: 87% vs. 69% for left ITA/SV, 75% for bare-metal stent, 73% for balloon angioplasty, and 70% for drug-eluting stent.
Propensity matching found similar survivability for balloon angioplasty and left ITA/SV when compared with MAG: 66% for MAG vs. 57% for the former; and 64% for MAG vs. 56% for the latter. The researchers also estimated the hazard ratio during the first 5 years of follow-up and found that those who had bare-metal stents had “significantly worse” survival, compared with MAG, but that survival evened out after that. Survival in the bare-metal stent group was similar to that of the left ITA/SV group, but “significantly worse” during the first 5 years for those who had balloon angioplasty.
Dr. Locker and his colleagues acknowledged that multiple randomized studies have compared CABG and PCI over the years, but they said that in most of those studies “the enrolled patients were highly selected and likely did not represent the broader population of patients with MVD [multivessel disease] undergoing revascularization.” With the exception of the SYNTAX trial (Synergy Between PCI With Taxus and Cardiac Surgery) (N Engl J Med. 2009;360:961-72; Lancet. 2013;381:629-38), those studies did not report on the frequency of MAG within the study population. The Mayo study, on the other hand, included all treated patients, excluding those who had a previous heart attack.
However, MAG is used infrequently, Dr. Locker and his colleagues said. The average annual rate of MAG in their Mayo practice was 15.2%, higher than the 5% annual rate the Society of Thoracic Surgeons National Cardiac Surgery Database (J Thorac Cardiovasc Surg. 2012;143:273-81) reported, and higher than the 12% rate in Europe (Eur J Cardiothorac Surg. 2006;29: 486-91). The SYNTAX trial reported an annual MAG rate of 27.6% for all CABG cases.
“It seems clear that use of MultiArt should be more frequent in patients with MVD undergoing CABG,” Dr. Locker and his coauthors said. “MultiArt can be used in most patients with MVD, including diabetic patients and elderly patients, and this strategy will improve outcomes of surgical revascularization.”
Dr. Locker and his coauthors had no financial relationships to disclose.
One might wonder about the validity of another retrospective, single-center study comparing revascularization techniques, but the study by Dr. Locker and his colleagues is “compelling” for two reasons, Paul Kurlansky, MD, of Columbia University, New York, pointed out in his invited commentary (J Thorac Cardiovasc Surg. 2016;152:380-1).

|
Dr. Paul Kurlansky |
Dr. Kurlansky noted the finding of equivalent survival at up to 8 years among propensity-matched patients who had left coronary artery bypass grafting with the use of a single internal thoracic artery with supplemental vein grafts (left ITA/SV) and those who received drug-eluting stents was “a bit more provocative” than some of the expected study results; and the reversal of the survival benefit of left ITA/SV, compared with both balloon angioplasty and bare-metal stents after 7 to 10 years was “more perturbing for the surgical community.”
This study underscores that increased use of multiple arterial grafting is essential to give patients the best revascularization option, even in the age of growing percutaneous interventions, Dr. Kurlansky said.
“Limitations notwithstanding, the message for the surgical community is clear – if we wish to have the opportunity to treat patients with advanced, multivessel coronary artery disease, we will need to more fully embrace a strategy of MAG,” he said.
Dr. Kurlansky had no financial relationships to disclose.
One might wonder about the validity of another retrospective, single-center study comparing revascularization techniques, but the study by Dr. Locker and his colleagues is “compelling” for two reasons, Paul Kurlansky, MD, of Columbia University, New York, pointed out in his invited commentary (J Thorac Cardiovasc Surg. 2016;152:380-1).

|
Dr. Paul Kurlansky |
Dr. Kurlansky noted the finding of equivalent survival at up to 8 years among propensity-matched patients who had left coronary artery bypass grafting with the use of a single internal thoracic artery with supplemental vein grafts (left ITA/SV) and those who received drug-eluting stents was “a bit more provocative” than some of the expected study results; and the reversal of the survival benefit of left ITA/SV, compared with both balloon angioplasty and bare-metal stents after 7 to 10 years was “more perturbing for the surgical community.”
This study underscores that increased use of multiple arterial grafting is essential to give patients the best revascularization option, even in the age of growing percutaneous interventions, Dr. Kurlansky said.
“Limitations notwithstanding, the message for the surgical community is clear – if we wish to have the opportunity to treat patients with advanced, multivessel coronary artery disease, we will need to more fully embrace a strategy of MAG,” he said.
Dr. Kurlansky had no financial relationships to disclose.
One might wonder about the validity of another retrospective, single-center study comparing revascularization techniques, but the study by Dr. Locker and his colleagues is “compelling” for two reasons, Paul Kurlansky, MD, of Columbia University, New York, pointed out in his invited commentary (J Thorac Cardiovasc Surg. 2016;152:380-1).

|
Dr. Paul Kurlansky |
Dr. Kurlansky noted the finding of equivalent survival at up to 8 years among propensity-matched patients who had left coronary artery bypass grafting with the use of a single internal thoracic artery with supplemental vein grafts (left ITA/SV) and those who received drug-eluting stents was “a bit more provocative” than some of the expected study results; and the reversal of the survival benefit of left ITA/SV, compared with both balloon angioplasty and bare-metal stents after 7 to 10 years was “more perturbing for the surgical community.”
This study underscores that increased use of multiple arterial grafting is essential to give patients the best revascularization option, even in the age of growing percutaneous interventions, Dr. Kurlansky said.
“Limitations notwithstanding, the message for the surgical community is clear – if we wish to have the opportunity to treat patients with advanced, multivessel coronary artery disease, we will need to more fully embrace a strategy of MAG,” he said.
Dr. Kurlansky had no financial relationships to disclose.
A large, 16-year single-center study of patients with multivessel disease has determined that multivessel coronary artery bypass grafting achieved longer survival than not only percutaneous coronary interventions, but also conventional coronary artery bypass grafting, researchers from the Mayo Clinic reported in the August issue of the Journal of Thoracic and Cardiovascular Surgery (J Thorac Cardiovasc Surg. 2016;152:369-79).
Lead author Chaim Locker, MD, and his colleagues said the use of what they called MultiArt, for multivessel arterial grafting, also known as MAG, “must increase.”
The evolution of bare-metal and then drug-eluting stents may have favored percutaneous coronary interventions (PCI) over coronary artery bypass grafting (CABG), but, Dr. Locker and his coauthors said, “Evidence is accumulating that late outcome of surgical revascularization is improved when at least two arterial grafts are used.”
The study analyzed results of 12,615 patients who had either isolated primary CABG (6,667) or PCI (5,948) from 1993 to 2009. Among the CABG patients, 5,712 had the more conventional approach involving arterial grafts into the left internal thoracic artery/saphenous vein (ITA/SV) and 955 had MAG. Patients in the PCI group had three different procedures: balloon angioplasty (1,020), drug-eluting stent (1,686), or bare-metal stent (3,242). The study excluded patients who had revascularization procedures after a heart attack.
While the overall 15-year survival for patients with CABG was lower than it was for those who had PCI (36% vs. 46%), the survival for those who had MAG was significantly higher: 65% vs. 31% for those who had left ITA/SV revascularization. 8-year survival for the MAG subgroup was also significantly higher than all other subgroups: 87% vs. 69% for left ITA/SV, 75% for bare-metal stent, 73% for balloon angioplasty, and 70% for drug-eluting stent.
Propensity matching found similar survivability for balloon angioplasty and left ITA/SV when compared with MAG: 66% for MAG vs. 57% for the former; and 64% for MAG vs. 56% for the latter. The researchers also estimated the hazard ratio during the first 5 years of follow-up and found that those who had bare-metal stents had “significantly worse” survival, compared with MAG, but that survival evened out after that. Survival in the bare-metal stent group was similar to that of the left ITA/SV group, but “significantly worse” during the first 5 years for those who had balloon angioplasty.
Dr. Locker and his colleagues acknowledged that multiple randomized studies have compared CABG and PCI over the years, but they said that in most of those studies “the enrolled patients were highly selected and likely did not represent the broader population of patients with MVD [multivessel disease] undergoing revascularization.” With the exception of the SYNTAX trial (Synergy Between PCI With Taxus and Cardiac Surgery) (N Engl J Med. 2009;360:961-72; Lancet. 2013;381:629-38), those studies did not report on the frequency of MAG within the study population. The Mayo study, on the other hand, included all treated patients, excluding those who had a previous heart attack.
However, MAG is used infrequently, Dr. Locker and his colleagues said. The average annual rate of MAG in their Mayo practice was 15.2%, higher than the 5% annual rate the Society of Thoracic Surgeons National Cardiac Surgery Database (J Thorac Cardiovasc Surg. 2012;143:273-81) reported, and higher than the 12% rate in Europe (Eur J Cardiothorac Surg. 2006;29: 486-91). The SYNTAX trial reported an annual MAG rate of 27.6% for all CABG cases.
“It seems clear that use of MultiArt should be more frequent in patients with MVD undergoing CABG,” Dr. Locker and his coauthors said. “MultiArt can be used in most patients with MVD, including diabetic patients and elderly patients, and this strategy will improve outcomes of surgical revascularization.”
Dr. Locker and his coauthors had no financial relationships to disclose.
A large, 16-year single-center study of patients with multivessel disease has determined that multivessel coronary artery bypass grafting achieved longer survival than not only percutaneous coronary interventions, but also conventional coronary artery bypass grafting, researchers from the Mayo Clinic reported in the August issue of the Journal of Thoracic and Cardiovascular Surgery (J Thorac Cardiovasc Surg. 2016;152:369-79).
Lead author Chaim Locker, MD, and his colleagues said the use of what they called MultiArt, for multivessel arterial grafting, also known as MAG, “must increase.”
The evolution of bare-metal and then drug-eluting stents may have favored percutaneous coronary interventions (PCI) over coronary artery bypass grafting (CABG), but, Dr. Locker and his coauthors said, “Evidence is accumulating that late outcome of surgical revascularization is improved when at least two arterial grafts are used.”
The study analyzed results of 12,615 patients who had either isolated primary CABG (6,667) or PCI (5,948) from 1993 to 2009. Among the CABG patients, 5,712 had the more conventional approach involving arterial grafts into the left internal thoracic artery/saphenous vein (ITA/SV) and 955 had MAG. Patients in the PCI group had three different procedures: balloon angioplasty (1,020), drug-eluting stent (1,686), or bare-metal stent (3,242). The study excluded patients who had revascularization procedures after a heart attack.
While the overall 15-year survival for patients with CABG was lower than it was for those who had PCI (36% vs. 46%), the survival for those who had MAG was significantly higher: 65% vs. 31% for those who had left ITA/SV revascularization. 8-year survival for the MAG subgroup was also significantly higher than all other subgroups: 87% vs. 69% for left ITA/SV, 75% for bare-metal stent, 73% for balloon angioplasty, and 70% for drug-eluting stent.
Propensity matching found similar survivability for balloon angioplasty and left ITA/SV when compared with MAG: 66% for MAG vs. 57% for the former; and 64% for MAG vs. 56% for the latter. The researchers also estimated the hazard ratio during the first 5 years of follow-up and found that those who had bare-metal stents had “significantly worse” survival, compared with MAG, but that survival evened out after that. Survival in the bare-metal stent group was similar to that of the left ITA/SV group, but “significantly worse” during the first 5 years for those who had balloon angioplasty.
Dr. Locker and his colleagues acknowledged that multiple randomized studies have compared CABG and PCI over the years, but they said that in most of those studies “the enrolled patients were highly selected and likely did not represent the broader population of patients with MVD [multivessel disease] undergoing revascularization.” With the exception of the SYNTAX trial (Synergy Between PCI With Taxus and Cardiac Surgery) (N Engl J Med. 2009;360:961-72; Lancet. 2013;381:629-38), those studies did not report on the frequency of MAG within the study population. The Mayo study, on the other hand, included all treated patients, excluding those who had a previous heart attack.
However, MAG is used infrequently, Dr. Locker and his colleagues said. The average annual rate of MAG in their Mayo practice was 15.2%, higher than the 5% annual rate the Society of Thoracic Surgeons National Cardiac Surgery Database (J Thorac Cardiovasc Surg. 2012;143:273-81) reported, and higher than the 12% rate in Europe (Eur J Cardiothorac Surg. 2006;29: 486-91). The SYNTAX trial reported an annual MAG rate of 27.6% for all CABG cases.
“It seems clear that use of MultiArt should be more frequent in patients with MVD undergoing CABG,” Dr. Locker and his coauthors said. “MultiArt can be used in most patients with MVD, including diabetic patients and elderly patients, and this strategy will improve outcomes of surgical revascularization.”
Dr. Locker and his coauthors had no financial relationships to disclose.
FROM THE JOURNAL OF THORACIC AND CARDIOVASCULAR SURGERY
Key clinical point: Cardiac surgeons should use multiple arterial coronary artery bypass grafting (MAG) more frequently because it achieves superior survival, compared with conventional bypass surgery or percutaneous coronary interventions.
Major finding: The overall 8-year survival of those who had MAG was 87% vs. 69% for conventional coronary bypass surgery and 70%-75% for percutaneous procedures.
Data source: Retrospective, single-institution study of 12,615 patients with multivessel disease who had revascularization procedures at the Mayo Clinic from 1993 to 2009.
Disclosures: Dr. Locker and his coauthors had no financial relationships to disclose.
Local Zika virus transmission identified in second area of Miami
The Centers for Disease Control and Prevention has confirmed that a new pocket of local Zika virus transmission has emerged in Miami Beach, adding yet another area in southern Florida for travelers to avoid.
“We now recommend [that] pregnant women should avoid travel to the designated area of Miami Beach, in addition to the designated area of Wynwood,” CDC Director Tom Frieden, MD, MPH, said during a conference call.
The announcement of a second Miami neighborhood experiencing ongoing local transmission came within 24 hours of the Miami Beach area being identified as a potential hazard, according to Dr. Frieden. Public health officials notified the public in late July about Zika virus transmission in the Wynwood neighborhood; in addition, there have been at least four other cases of independent mosquito-borne transmission of the Zika virus, but the CDC noted that these are not indicative of the disease spreading or becoming locally transmitted to a significant extent.
For those living in or near the affected areas, Dr. Frieden urged that they “do everything they can to prevent mosquito bites,” including wearing clothing that covers as much of the body as possible and using bug repellent, among other things. Those who traveled to the Miami area on or after July 14 of this year should use protection while having sex with their partners to prevent transmitting the virus that way. Women should wait until at least 8 weeks after onset of Zika virus symptoms before attempting to get pregnant.
“More broadly and not just with respect to Florida, all pregnant women anywhere in the U.S. should be evaluated for possible Zika virus exposure during each prenatal care visit,” said Dr. Frieden. “These evaluations should include an assessment of the symptoms of Zika virus disease such as fever, rash, arthralgia, and conjunctivitis, their travel history, and their potential partners’ exposure to Zika virus.”
Dr. Frieden said that the CDC will continue monitoring the situation on a daily basis and make any further announcements, along with changes to current guidelines and recommendations, as necessary.
The Centers for Disease Control and Prevention has confirmed that a new pocket of local Zika virus transmission has emerged in Miami Beach, adding yet another area in southern Florida for travelers to avoid.
“We now recommend [that] pregnant women should avoid travel to the designated area of Miami Beach, in addition to the designated area of Wynwood,” CDC Director Tom Frieden, MD, MPH, said during a conference call.
The announcement of a second Miami neighborhood experiencing ongoing local transmission came within 24 hours of the Miami Beach area being identified as a potential hazard, according to Dr. Frieden. Public health officials notified the public in late July about Zika virus transmission in the Wynwood neighborhood; in addition, there have been at least four other cases of independent mosquito-borne transmission of the Zika virus, but the CDC noted that these are not indicative of the disease spreading or becoming locally transmitted to a significant extent.
For those living in or near the affected areas, Dr. Frieden urged that they “do everything they can to prevent mosquito bites,” including wearing clothing that covers as much of the body as possible and using bug repellent, among other things. Those who traveled to the Miami area on or after July 14 of this year should use protection while having sex with their partners to prevent transmitting the virus that way. Women should wait until at least 8 weeks after onset of Zika virus symptoms before attempting to get pregnant.
“More broadly and not just with respect to Florida, all pregnant women anywhere in the U.S. should be evaluated for possible Zika virus exposure during each prenatal care visit,” said Dr. Frieden. “These evaluations should include an assessment of the symptoms of Zika virus disease such as fever, rash, arthralgia, and conjunctivitis, their travel history, and their potential partners’ exposure to Zika virus.”
Dr. Frieden said that the CDC will continue monitoring the situation on a daily basis and make any further announcements, along with changes to current guidelines and recommendations, as necessary.
The Centers for Disease Control and Prevention has confirmed that a new pocket of local Zika virus transmission has emerged in Miami Beach, adding yet another area in southern Florida for travelers to avoid.
“We now recommend [that] pregnant women should avoid travel to the designated area of Miami Beach, in addition to the designated area of Wynwood,” CDC Director Tom Frieden, MD, MPH, said during a conference call.
The announcement of a second Miami neighborhood experiencing ongoing local transmission came within 24 hours of the Miami Beach area being identified as a potential hazard, according to Dr. Frieden. Public health officials notified the public in late July about Zika virus transmission in the Wynwood neighborhood; in addition, there have been at least four other cases of independent mosquito-borne transmission of the Zika virus, but the CDC noted that these are not indicative of the disease spreading or becoming locally transmitted to a significant extent.
For those living in or near the affected areas, Dr. Frieden urged that they “do everything they can to prevent mosquito bites,” including wearing clothing that covers as much of the body as possible and using bug repellent, among other things. Those who traveled to the Miami area on or after July 14 of this year should use protection while having sex with their partners to prevent transmitting the virus that way. Women should wait until at least 8 weeks after onset of Zika virus symptoms before attempting to get pregnant.
“More broadly and not just with respect to Florida, all pregnant women anywhere in the U.S. should be evaluated for possible Zika virus exposure during each prenatal care visit,” said Dr. Frieden. “These evaluations should include an assessment of the symptoms of Zika virus disease such as fever, rash, arthralgia, and conjunctivitis, their travel history, and their potential partners’ exposure to Zika virus.”
Dr. Frieden said that the CDC will continue monitoring the situation on a daily basis and make any further announcements, along with changes to current guidelines and recommendations, as necessary.
Endocrinologist links nighttime hypoglycemia to many ills
SAN DIEGO – A top endocrinologist cautioned diabetes educators that research is linking nighttime hypoglycemia to a variety of ills, and technology isn’t providing much hope – yet.
Patients with nocturnal low blood sugar “say this is the hardest thing they have to deal with. It upsets their whole day and they feel terrible,” said Anthony L. McCall, MD, PhD, James M. Moss Professor of Diabetes at the University of Virginia, Charlottesville, and vice president of clinical science with the Endocrine Society.
Dr. McCall told an audience at the annual meeting of the American Association of Diabetes Educators that half of hypoglycemia is nocturnal and unrecognized despite its dangers. According to him, hypoglycemia represents a blood glucose level of at or under 70 mg/dL (3.9 mmol/L). This is higher than the threshold for hypoglycemia in nondiabetics and those with well controlled diabetes.
Even as few as two values in a week in the range of the 60s (mg/dL) can go unrecognized and lead to full-blown hypoglycemia-associated autonomic failure, he said. There are other possible risks: “impaired sleep quality, daytime drowsiness, mood changes, risk for nocturnal falls,” he said.
Cognitive dysfunction is possible, especially in children, he added. “Neurological dysfunction may be temporary, but those who can answer simple questions may not be OK.”
There’s a potential for a vicious cycle here, he said, because people with diabetes can also develop impaired hypoglycemia awareness, making it less likely they’ll notice the low blood sugar levels that contribute to autonomic failure.
Dr. McCall reported that nighttime hypoglycemia may also:
• Trigger neurologic symptoms like those of strokes or temporary ischemic attacks. “Someone’s got check to their blood sugar,” he says.
• Lengthen the QT interval and boost the risk of irregular heartbeats.
• Contribute to “dead in bed” syndrome in which young people with type 1 diabetes are discovered dead despite not having any complications or showing signs of convulsion.
What can be done to help these patients? One approach is to combat impaired hypoglycemia awareness. Bedtime snacks, caffeine, and uncooked cornstarch are among the many nutrition supplements (and medications) that have shown inconsistent results at best on this front, Dr. McCall said. If they work, he said, they often lead to hyperglycemia.
Another strategy is to look for factors that raise the risk of nighttime hypoglycemia, such as basal insulin overtreatment, long periods between meals, delayed effects of exercise, and higher insulin sensitivity overnight.
Insulin pumps may be helpful, he said, and he generally favors their use. However, he cautioned that it’s hard to show that they reduce hypoglycemia, and some patients don’t use them properly.
Data have been mixed until recently regarding real-time continuous glucose monitoring, he said, and the devices must be worn 75%-85% of the time to show benefit. As for sensor-augmented insulin pumps, he said they’ve shown mixed results.
Dr. McCall said the artificial pancreas, once it makes it to market, could mark the beginning of a new era. “This was around the corner 40 years ago. But it’s closer now,” he said. “I have great hope that we’re going to do better.”
Dr. McCall reported being a consultant to Sanofi regarding new insulin studies and serving on the advisory board of DexCom/Google regarding the use of continuous glucose monitoring.
SAN DIEGO – A top endocrinologist cautioned diabetes educators that research is linking nighttime hypoglycemia to a variety of ills, and technology isn’t providing much hope – yet.
Patients with nocturnal low blood sugar “say this is the hardest thing they have to deal with. It upsets their whole day and they feel terrible,” said Anthony L. McCall, MD, PhD, James M. Moss Professor of Diabetes at the University of Virginia, Charlottesville, and vice president of clinical science with the Endocrine Society.
Dr. McCall told an audience at the annual meeting of the American Association of Diabetes Educators that half of hypoglycemia is nocturnal and unrecognized despite its dangers. According to him, hypoglycemia represents a blood glucose level of at or under 70 mg/dL (3.9 mmol/L). This is higher than the threshold for hypoglycemia in nondiabetics and those with well controlled diabetes.
Even as few as two values in a week in the range of the 60s (mg/dL) can go unrecognized and lead to full-blown hypoglycemia-associated autonomic failure, he said. There are other possible risks: “impaired sleep quality, daytime drowsiness, mood changes, risk for nocturnal falls,” he said.
Cognitive dysfunction is possible, especially in children, he added. “Neurological dysfunction may be temporary, but those who can answer simple questions may not be OK.”
There’s a potential for a vicious cycle here, he said, because people with diabetes can also develop impaired hypoglycemia awareness, making it less likely they’ll notice the low blood sugar levels that contribute to autonomic failure.
Dr. McCall reported that nighttime hypoglycemia may also:
• Trigger neurologic symptoms like those of strokes or temporary ischemic attacks. “Someone’s got check to their blood sugar,” he says.
• Lengthen the QT interval and boost the risk of irregular heartbeats.
• Contribute to “dead in bed” syndrome in which young people with type 1 diabetes are discovered dead despite not having any complications or showing signs of convulsion.
What can be done to help these patients? One approach is to combat impaired hypoglycemia awareness. Bedtime snacks, caffeine, and uncooked cornstarch are among the many nutrition supplements (and medications) that have shown inconsistent results at best on this front, Dr. McCall said. If they work, he said, they often lead to hyperglycemia.
Another strategy is to look for factors that raise the risk of nighttime hypoglycemia, such as basal insulin overtreatment, long periods between meals, delayed effects of exercise, and higher insulin sensitivity overnight.
Insulin pumps may be helpful, he said, and he generally favors their use. However, he cautioned that it’s hard to show that they reduce hypoglycemia, and some patients don’t use them properly.
Data have been mixed until recently regarding real-time continuous glucose monitoring, he said, and the devices must be worn 75%-85% of the time to show benefit. As for sensor-augmented insulin pumps, he said they’ve shown mixed results.
Dr. McCall said the artificial pancreas, once it makes it to market, could mark the beginning of a new era. “This was around the corner 40 years ago. But it’s closer now,” he said. “I have great hope that we’re going to do better.”
Dr. McCall reported being a consultant to Sanofi regarding new insulin studies and serving on the advisory board of DexCom/Google regarding the use of continuous glucose monitoring.
SAN DIEGO – A top endocrinologist cautioned diabetes educators that research is linking nighttime hypoglycemia to a variety of ills, and technology isn’t providing much hope – yet.
Patients with nocturnal low blood sugar “say this is the hardest thing they have to deal with. It upsets their whole day and they feel terrible,” said Anthony L. McCall, MD, PhD, James M. Moss Professor of Diabetes at the University of Virginia, Charlottesville, and vice president of clinical science with the Endocrine Society.
Dr. McCall told an audience at the annual meeting of the American Association of Diabetes Educators that half of hypoglycemia is nocturnal and unrecognized despite its dangers. According to him, hypoglycemia represents a blood glucose level of at or under 70 mg/dL (3.9 mmol/L). This is higher than the threshold for hypoglycemia in nondiabetics and those with well controlled diabetes.
Even as few as two values in a week in the range of the 60s (mg/dL) can go unrecognized and lead to full-blown hypoglycemia-associated autonomic failure, he said. There are other possible risks: “impaired sleep quality, daytime drowsiness, mood changes, risk for nocturnal falls,” he said.
Cognitive dysfunction is possible, especially in children, he added. “Neurological dysfunction may be temporary, but those who can answer simple questions may not be OK.”
There’s a potential for a vicious cycle here, he said, because people with diabetes can also develop impaired hypoglycemia awareness, making it less likely they’ll notice the low blood sugar levels that contribute to autonomic failure.
Dr. McCall reported that nighttime hypoglycemia may also:
• Trigger neurologic symptoms like those of strokes or temporary ischemic attacks. “Someone’s got check to their blood sugar,” he says.
• Lengthen the QT interval and boost the risk of irregular heartbeats.
• Contribute to “dead in bed” syndrome in which young people with type 1 diabetes are discovered dead despite not having any complications or showing signs of convulsion.
What can be done to help these patients? One approach is to combat impaired hypoglycemia awareness. Bedtime snacks, caffeine, and uncooked cornstarch are among the many nutrition supplements (and medications) that have shown inconsistent results at best on this front, Dr. McCall said. If they work, he said, they often lead to hyperglycemia.
Another strategy is to look for factors that raise the risk of nighttime hypoglycemia, such as basal insulin overtreatment, long periods between meals, delayed effects of exercise, and higher insulin sensitivity overnight.
Insulin pumps may be helpful, he said, and he generally favors their use. However, he cautioned that it’s hard to show that they reduce hypoglycemia, and some patients don’t use them properly.
Data have been mixed until recently regarding real-time continuous glucose monitoring, he said, and the devices must be worn 75%-85% of the time to show benefit. As for sensor-augmented insulin pumps, he said they’ve shown mixed results.
Dr. McCall said the artificial pancreas, once it makes it to market, could mark the beginning of a new era. “This was around the corner 40 years ago. But it’s closer now,” he said. “I have great hope that we’re going to do better.”
Dr. McCall reported being a consultant to Sanofi regarding new insulin studies and serving on the advisory board of DexCom/Google regarding the use of continuous glucose monitoring.
AT AADE 16
What doctors want for their kids
Editor’s note: This is Dr. Chan’s last post for her “Rheum in Bloom” column. She is saying goodbye to New England, leaving the private practice setting for a different challenge.
All too often these days, I find myself fidgeting by the doorway to my exam room, trying to conclude an office visit with one of my patients. When I look at my career at midlife, I realize that in many ways I have become the kind of doctor I never thought I’d be: impatient, occasionally indifferent, at times dismissive or paternalistic.
–Sandeep Jauhar, MD
For as long as I’ve been a doctor, doctors have lamented how much medicine has changed. Columnist Charles Krauthammer, MD, wrote a widely-circulated piece in 2015 with the catchy title “Why Doctors Quit,” attributing physician dissatisfaction to the wasteful and frustrating electronic health record mandate. It is worth noting here that Dr. Krauthammer is a political partisan making a point about government overreach and not really about the state of medicine, but the piece certainly resonated with doctors, judging by the number of times it was shared with me. In Medscape’s physician compensation poll of 2016, only 64% of doctors would choose medicine again. In a separate Medscape poll on burnout, under 30% of physicians felt happy at work.
In Dr. Sandeep Jauhar’s book, “Doctored,” (from which the above quote is taken), he cites several issues: the lack of respect that the profession is afforded, the practice of defensive medicine, inadequate compensation, the lack of independence. He goes further: “... Surveys have shown that 30%-40% of practicing physicians would not choose to enter the medical profession if they were deciding on a career again, and an even higher percentage would not encourage their children to pursue a medical career.” He does not say where he got that information, but it sounds plausible.
I asked peers, mostly people I went through training with and therefore generally belonging to my age group. This small, nonrepresentative sample of young physician parents, many of whom are foreign graduates, seemed to belie Dr. Jauhar’s pessimism. Most of my friends thought medicine is still rewarding. They see our profession as emotionally meaningful and intellectually fulfilling – this despite a surprising number of them having been named in ultimately unsuccessful lawsuits. “The headaches are many, but most professions that make decent money have their own set of headaches and problems,” my allergist friend says. In addition, a career in medicine, they feel, would provide their children financial stability. The consensus was that if their children wanted to, my peers would be nothing but supportive.
I asked my boss about it, too. He is older and very vocal about what he perceives as a hostile working environment. If there was just one demographic that Dr. Krauthammer was speaking for, that is exactly the demographic that my boss belongs to. So I was certain that he had similarly dissuaded his son from going into medicine. But I was wrong.
It isn’t that my boss discouraged his son; it’s that his son was not interested. He says if he thought his son might have enjoyed the work he absolutely would have encouraged it. Because as onerous as our profession has become, it is still a meaningful and rewarding one. And as long as we derive meaning from the work that we do, it is much easier to put up with the unsavory parts.
Dr. Chan practices rheumatology in Pawtucket, R.I.
Editor’s note: This is Dr. Chan’s last post for her “Rheum in Bloom” column. She is saying goodbye to New England, leaving the private practice setting for a different challenge.
All too often these days, I find myself fidgeting by the doorway to my exam room, trying to conclude an office visit with one of my patients. When I look at my career at midlife, I realize that in many ways I have become the kind of doctor I never thought I’d be: impatient, occasionally indifferent, at times dismissive or paternalistic.
–Sandeep Jauhar, MD
For as long as I’ve been a doctor, doctors have lamented how much medicine has changed. Columnist Charles Krauthammer, MD, wrote a widely-circulated piece in 2015 with the catchy title “Why Doctors Quit,” attributing physician dissatisfaction to the wasteful and frustrating electronic health record mandate. It is worth noting here that Dr. Krauthammer is a political partisan making a point about government overreach and not really about the state of medicine, but the piece certainly resonated with doctors, judging by the number of times it was shared with me. In Medscape’s physician compensation poll of 2016, only 64% of doctors would choose medicine again. In a separate Medscape poll on burnout, under 30% of physicians felt happy at work.
In Dr. Sandeep Jauhar’s book, “Doctored,” (from which the above quote is taken), he cites several issues: the lack of respect that the profession is afforded, the practice of defensive medicine, inadequate compensation, the lack of independence. He goes further: “... Surveys have shown that 30%-40% of practicing physicians would not choose to enter the medical profession if they were deciding on a career again, and an even higher percentage would not encourage their children to pursue a medical career.” He does not say where he got that information, but it sounds plausible.
I asked peers, mostly people I went through training with and therefore generally belonging to my age group. This small, nonrepresentative sample of young physician parents, many of whom are foreign graduates, seemed to belie Dr. Jauhar’s pessimism. Most of my friends thought medicine is still rewarding. They see our profession as emotionally meaningful and intellectually fulfilling – this despite a surprising number of them having been named in ultimately unsuccessful lawsuits. “The headaches are many, but most professions that make decent money have their own set of headaches and problems,” my allergist friend says. In addition, a career in medicine, they feel, would provide their children financial stability. The consensus was that if their children wanted to, my peers would be nothing but supportive.
I asked my boss about it, too. He is older and very vocal about what he perceives as a hostile working environment. If there was just one demographic that Dr. Krauthammer was speaking for, that is exactly the demographic that my boss belongs to. So I was certain that he had similarly dissuaded his son from going into medicine. But I was wrong.
It isn’t that my boss discouraged his son; it’s that his son was not interested. He says if he thought his son might have enjoyed the work he absolutely would have encouraged it. Because as onerous as our profession has become, it is still a meaningful and rewarding one. And as long as we derive meaning from the work that we do, it is much easier to put up with the unsavory parts.
Dr. Chan practices rheumatology in Pawtucket, R.I.
Editor’s note: This is Dr. Chan’s last post for her “Rheum in Bloom” column. She is saying goodbye to New England, leaving the private practice setting for a different challenge.
All too often these days, I find myself fidgeting by the doorway to my exam room, trying to conclude an office visit with one of my patients. When I look at my career at midlife, I realize that in many ways I have become the kind of doctor I never thought I’d be: impatient, occasionally indifferent, at times dismissive or paternalistic.
–Sandeep Jauhar, MD
For as long as I’ve been a doctor, doctors have lamented how much medicine has changed. Columnist Charles Krauthammer, MD, wrote a widely-circulated piece in 2015 with the catchy title “Why Doctors Quit,” attributing physician dissatisfaction to the wasteful and frustrating electronic health record mandate. It is worth noting here that Dr. Krauthammer is a political partisan making a point about government overreach and not really about the state of medicine, but the piece certainly resonated with doctors, judging by the number of times it was shared with me. In Medscape’s physician compensation poll of 2016, only 64% of doctors would choose medicine again. In a separate Medscape poll on burnout, under 30% of physicians felt happy at work.
In Dr. Sandeep Jauhar’s book, “Doctored,” (from which the above quote is taken), he cites several issues: the lack of respect that the profession is afforded, the practice of defensive medicine, inadequate compensation, the lack of independence. He goes further: “... Surveys have shown that 30%-40% of practicing physicians would not choose to enter the medical profession if they were deciding on a career again, and an even higher percentage would not encourage their children to pursue a medical career.” He does not say where he got that information, but it sounds plausible.
I asked peers, mostly people I went through training with and therefore generally belonging to my age group. This small, nonrepresentative sample of young physician parents, many of whom are foreign graduates, seemed to belie Dr. Jauhar’s pessimism. Most of my friends thought medicine is still rewarding. They see our profession as emotionally meaningful and intellectually fulfilling – this despite a surprising number of them having been named in ultimately unsuccessful lawsuits. “The headaches are many, but most professions that make decent money have their own set of headaches and problems,” my allergist friend says. In addition, a career in medicine, they feel, would provide their children financial stability. The consensus was that if their children wanted to, my peers would be nothing but supportive.
I asked my boss about it, too. He is older and very vocal about what he perceives as a hostile working environment. If there was just one demographic that Dr. Krauthammer was speaking for, that is exactly the demographic that my boss belongs to. So I was certain that he had similarly dissuaded his son from going into medicine. But I was wrong.
It isn’t that my boss discouraged his son; it’s that his son was not interested. He says if he thought his son might have enjoyed the work he absolutely would have encouraged it. Because as onerous as our profession has become, it is still a meaningful and rewarding one. And as long as we derive meaning from the work that we do, it is much easier to put up with the unsavory parts.
Dr. Chan practices rheumatology in Pawtucket, R.I.
Should risk-reducing gynecologic surgery for BRCA mutation carriers include hysterectomy?
EXPERT COMMENTARY
Although RRSO is routinely recommended to decrease the risk of ovarian as well as breast cancer in women who harbor BRCA mutations, whether or not surgery should include hysterectomy has not been clear. Shu and colleagues prospectively evaluated the risk of uterine cancer in BRCA1 and BRCA2 mutation carriers who underwent RRSO without hysterectomy at 1 of 9 centers in the United States or the United Kingdom, comparing it with rates expected from national surveillance data.
Details of the study
Among 1,083 women (median age, 45.6 years at the time of RRSO; median follow-up, 5.1 years), 8 incident uterine cancers were observed (4.3 were expected; observed to expected [O:E] ratio, 1.9; P = .09). In an analysis stratified by tumor subtype, no elevated risk for endometrioid endometrial cancer or sarcoma was noted. Five serous and/or serous-like endometrial cancers were observed after RRSO. Four were found in BRCA1 carriers and 1 in a BRCA2 carrier, with the O:E ratio of 22.2 (P<.001) for BRCA1 and 6.4 (P = .15) for BRCA2 carriers.
Details of the accompanying editorial
Commenting on the study, Leath and colleagues noted that while the number of cases was small, the study demonstrated a link between the presence of BRCA mutations (particularly BRCA1 mutations) and a "small but not null risk of endometrial cancer." Many of these uterine cancers have a serous histology, a concern due to the likelihood of worse outcomes compared with the more common endometrioid variant.
The authors suggest that clinicians make patients with BRCA mutations undergoing RRSO aware of the potential risks and benefits of concomitant hysterectomy and work to make an individualized decision based on the limitations of available data. They state, "Perhaps it is time to consider that the line for risk-reducing gynecologic surgery in patients with BRCA mutations not stop at the ovaries and fallopian tubes. Thus, concomitant hysterectomy with RRSO, when performed with a minimally invasive surgical approach, particularly for women with a BRCA1 mutation, should be able to be performed with minimum morbidity and allow for use of estrogen-only hormone therapy after surgery, if needed."
Although adding hysterectomy to RRSO increases the perioperative risks, these risks should be attenuated by the use of minimally invasive surgical approaches. Women diagnosed with serous endometrial cancer have worse outcomes than those with the more common endometrioid endometrial tumors, even when they are diagnosed with early stage disease. Future risk of any uterine malignancy is essentially eliminated when hysterectomy is performed.
Risk-reducing gynecologic surgery in BRCA carriers is optimally performed prior to age 40, meaning that unless hormone therapy is used, severe menopausal symptoms likely will occur, and women are at elevated risk for osteoporosis and cardiovascular and neurodegenerative diseases. Given that hysterectomy allows estrogen (little or no impact on breast cancer risk) to be prescribed without progestin (small elevation in breast cancer risk), hysterectomy has particular advantages for younger mutation carriers undergoing risk-reducing gynecologic surgery.
I agree with the editorialists who provided an accompanying comment on the study that—provided a minimally invasive approach is used—we should encourage hysterectomy as part of risk-reducing surgery for women with BRCA mutations.
—ANDREW M. KAUNITZ, MD
Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
EXPERT COMMENTARY
Although RRSO is routinely recommended to decrease the risk of ovarian as well as breast cancer in women who harbor BRCA mutations, whether or not surgery should include hysterectomy has not been clear. Shu and colleagues prospectively evaluated the risk of uterine cancer in BRCA1 and BRCA2 mutation carriers who underwent RRSO without hysterectomy at 1 of 9 centers in the United States or the United Kingdom, comparing it with rates expected from national surveillance data.
Details of the study
Among 1,083 women (median age, 45.6 years at the time of RRSO; median follow-up, 5.1 years), 8 incident uterine cancers were observed (4.3 were expected; observed to expected [O:E] ratio, 1.9; P = .09). In an analysis stratified by tumor subtype, no elevated risk for endometrioid endometrial cancer or sarcoma was noted. Five serous and/or serous-like endometrial cancers were observed after RRSO. Four were found in BRCA1 carriers and 1 in a BRCA2 carrier, with the O:E ratio of 22.2 (P<.001) for BRCA1 and 6.4 (P = .15) for BRCA2 carriers.
Details of the accompanying editorial
Commenting on the study, Leath and colleagues noted that while the number of cases was small, the study demonstrated a link between the presence of BRCA mutations (particularly BRCA1 mutations) and a "small but not null risk of endometrial cancer." Many of these uterine cancers have a serous histology, a concern due to the likelihood of worse outcomes compared with the more common endometrioid variant.
The authors suggest that clinicians make patients with BRCA mutations undergoing RRSO aware of the potential risks and benefits of concomitant hysterectomy and work to make an individualized decision based on the limitations of available data. They state, "Perhaps it is time to consider that the line for risk-reducing gynecologic surgery in patients with BRCA mutations not stop at the ovaries and fallopian tubes. Thus, concomitant hysterectomy with RRSO, when performed with a minimally invasive surgical approach, particularly for women with a BRCA1 mutation, should be able to be performed with minimum morbidity and allow for use of estrogen-only hormone therapy after surgery, if needed."
Although adding hysterectomy to RRSO increases the perioperative risks, these risks should be attenuated by the use of minimally invasive surgical approaches. Women diagnosed with serous endometrial cancer have worse outcomes than those with the more common endometrioid endometrial tumors, even when they are diagnosed with early stage disease. Future risk of any uterine malignancy is essentially eliminated when hysterectomy is performed.
Risk-reducing gynecologic surgery in BRCA carriers is optimally performed prior to age 40, meaning that unless hormone therapy is used, severe menopausal symptoms likely will occur, and women are at elevated risk for osteoporosis and cardiovascular and neurodegenerative diseases. Given that hysterectomy allows estrogen (little or no impact on breast cancer risk) to be prescribed without progestin (small elevation in breast cancer risk), hysterectomy has particular advantages for younger mutation carriers undergoing risk-reducing gynecologic surgery.
I agree with the editorialists who provided an accompanying comment on the study that—provided a minimally invasive approach is used—we should encourage hysterectomy as part of risk-reducing surgery for women with BRCA mutations.
—ANDREW M. KAUNITZ, MD
Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
EXPERT COMMENTARY
Although RRSO is routinely recommended to decrease the risk of ovarian as well as breast cancer in women who harbor BRCA mutations, whether or not surgery should include hysterectomy has not been clear. Shu and colleagues prospectively evaluated the risk of uterine cancer in BRCA1 and BRCA2 mutation carriers who underwent RRSO without hysterectomy at 1 of 9 centers in the United States or the United Kingdom, comparing it with rates expected from national surveillance data.
Details of the study
Among 1,083 women (median age, 45.6 years at the time of RRSO; median follow-up, 5.1 years), 8 incident uterine cancers were observed (4.3 were expected; observed to expected [O:E] ratio, 1.9; P = .09). In an analysis stratified by tumor subtype, no elevated risk for endometrioid endometrial cancer or sarcoma was noted. Five serous and/or serous-like endometrial cancers were observed after RRSO. Four were found in BRCA1 carriers and 1 in a BRCA2 carrier, with the O:E ratio of 22.2 (P<.001) for BRCA1 and 6.4 (P = .15) for BRCA2 carriers.
Details of the accompanying editorial
Commenting on the study, Leath and colleagues noted that while the number of cases was small, the study demonstrated a link between the presence of BRCA mutations (particularly BRCA1 mutations) and a "small but not null risk of endometrial cancer." Many of these uterine cancers have a serous histology, a concern due to the likelihood of worse outcomes compared with the more common endometrioid variant.
The authors suggest that clinicians make patients with BRCA mutations undergoing RRSO aware of the potential risks and benefits of concomitant hysterectomy and work to make an individualized decision based on the limitations of available data. They state, "Perhaps it is time to consider that the line for risk-reducing gynecologic surgery in patients with BRCA mutations not stop at the ovaries and fallopian tubes. Thus, concomitant hysterectomy with RRSO, when performed with a minimally invasive surgical approach, particularly for women with a BRCA1 mutation, should be able to be performed with minimum morbidity and allow for use of estrogen-only hormone therapy after surgery, if needed."
Although adding hysterectomy to RRSO increases the perioperative risks, these risks should be attenuated by the use of minimally invasive surgical approaches. Women diagnosed with serous endometrial cancer have worse outcomes than those with the more common endometrioid endometrial tumors, even when they are diagnosed with early stage disease. Future risk of any uterine malignancy is essentially eliminated when hysterectomy is performed.
Risk-reducing gynecologic surgery in BRCA carriers is optimally performed prior to age 40, meaning that unless hormone therapy is used, severe menopausal symptoms likely will occur, and women are at elevated risk for osteoporosis and cardiovascular and neurodegenerative diseases. Given that hysterectomy allows estrogen (little or no impact on breast cancer risk) to be prescribed without progestin (small elevation in breast cancer risk), hysterectomy has particular advantages for younger mutation carriers undergoing risk-reducing gynecologic surgery.
I agree with the editorialists who provided an accompanying comment on the study that—provided a minimally invasive approach is used—we should encourage hysterectomy as part of risk-reducing surgery for women with BRCA mutations.
—ANDREW M. KAUNITZ, MD
Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
Vigilance in prescribing narcotics: Unavoidable collateral damage?
Every 3 months I get a letter from the state, showing me how my controlled prescriptions stack up against those of other neurologists here.
I’ve always been in the “normal” range. Which is good, I guess. In this era, no one wants to be seen as running a pill mill.
But when I compare them over the last year, I notice that my narcotic prescriptions have been gradually declining. Am I reducing my scripts subconsciously, knowing that I’m being watched by regulatory agencies?
It’s quite possible. While I haven’t intentionally been cutting back on narcotics, I have been paying closer attention to who I’m writing them for. This has likely led me to use them more sparingly.
The trouble is that pain is a legitimate problem for many, and NSAIDs have numerous safety issues that limit their use. Acetaminophen has hepatic issues. My previous noncontrolled drug of choice, Tramadol, was reclassified as controlled in 2014.
Narcotic abuse and diversion are serious problems that need attention. But there are also people with legitimate noncancer pain who require them to function with a reasonable quality of life. We all have them in our practice. Sorting them out from abusers is never easy.
Unfortunately, the increased vigilance also affects those who need our help. A recent National Public Radio article noted the difficulty of finding a doctor in Montana who is willing to take on pain patients, with the result that some have to travel out of state to get help.
I’m sure they’re not the only ones, especially in states that have a low population density. And not all patients are going to have the financial resources to travel. Or afford the rates of the dwindling number of physicians willing to frequently prescribe narcotics. These people sadly become collateral damage in the drug wars.
Does this mean I’m going to increase my use of narcotics to help all who come to me? No.
Because, in a world where my licensure (and therefore livelihood) is potentially affected by my prescribing habits, I have to put my family first. This doesn’t mean I’m abandoning any of my current or even future patients, but it does mean I’ll be more vigilant on every controlled script I write.
I don’t know anyone who’d do otherwise in today’s climate.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
Every 3 months I get a letter from the state, showing me how my controlled prescriptions stack up against those of other neurologists here.
I’ve always been in the “normal” range. Which is good, I guess. In this era, no one wants to be seen as running a pill mill.
But when I compare them over the last year, I notice that my narcotic prescriptions have been gradually declining. Am I reducing my scripts subconsciously, knowing that I’m being watched by regulatory agencies?
It’s quite possible. While I haven’t intentionally been cutting back on narcotics, I have been paying closer attention to who I’m writing them for. This has likely led me to use them more sparingly.
The trouble is that pain is a legitimate problem for many, and NSAIDs have numerous safety issues that limit their use. Acetaminophen has hepatic issues. My previous noncontrolled drug of choice, Tramadol, was reclassified as controlled in 2014.
Narcotic abuse and diversion are serious problems that need attention. But there are also people with legitimate noncancer pain who require them to function with a reasonable quality of life. We all have them in our practice. Sorting them out from abusers is never easy.
Unfortunately, the increased vigilance also affects those who need our help. A recent National Public Radio article noted the difficulty of finding a doctor in Montana who is willing to take on pain patients, with the result that some have to travel out of state to get help.
I’m sure they’re not the only ones, especially in states that have a low population density. And not all patients are going to have the financial resources to travel. Or afford the rates of the dwindling number of physicians willing to frequently prescribe narcotics. These people sadly become collateral damage in the drug wars.
Does this mean I’m going to increase my use of narcotics to help all who come to me? No.
Because, in a world where my licensure (and therefore livelihood) is potentially affected by my prescribing habits, I have to put my family first. This doesn’t mean I’m abandoning any of my current or even future patients, but it does mean I’ll be more vigilant on every controlled script I write.
I don’t know anyone who’d do otherwise in today’s climate.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
Every 3 months I get a letter from the state, showing me how my controlled prescriptions stack up against those of other neurologists here.
I’ve always been in the “normal” range. Which is good, I guess. In this era, no one wants to be seen as running a pill mill.
But when I compare them over the last year, I notice that my narcotic prescriptions have been gradually declining. Am I reducing my scripts subconsciously, knowing that I’m being watched by regulatory agencies?
It’s quite possible. While I haven’t intentionally been cutting back on narcotics, I have been paying closer attention to who I’m writing them for. This has likely led me to use them more sparingly.
The trouble is that pain is a legitimate problem for many, and NSAIDs have numerous safety issues that limit their use. Acetaminophen has hepatic issues. My previous noncontrolled drug of choice, Tramadol, was reclassified as controlled in 2014.
Narcotic abuse and diversion are serious problems that need attention. But there are also people with legitimate noncancer pain who require them to function with a reasonable quality of life. We all have them in our practice. Sorting them out from abusers is never easy.
Unfortunately, the increased vigilance also affects those who need our help. A recent National Public Radio article noted the difficulty of finding a doctor in Montana who is willing to take on pain patients, with the result that some have to travel out of state to get help.
I’m sure they’re not the only ones, especially in states that have a low population density. And not all patients are going to have the financial resources to travel. Or afford the rates of the dwindling number of physicians willing to frequently prescribe narcotics. These people sadly become collateral damage in the drug wars.
Does this mean I’m going to increase my use of narcotics to help all who come to me? No.
Because, in a world where my licensure (and therefore livelihood) is potentially affected by my prescribing habits, I have to put my family first. This doesn’t mean I’m abandoning any of my current or even future patients, but it does mean I’ll be more vigilant on every controlled script I write.
I don’t know anyone who’d do otherwise in today’s climate.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.