User login
OMP and SNAPPS for Inpatient Teaching
Hospitalists who teach in the clinical environment face challenges that include increased workload,[1] perception among trainees that there is less time to teach,[2] and competition with electronic devices for teaching engagement.[3, 4] In view of these and other challenges, we believe there is potentially much to gain from considering and adapting educational techniques that have been successful in nonhospital and even nonmedical domains. Innovative teaching methods include those designed for the grade‐school classroom (Courage to Teach,[5] Teaching With Love and Logic[6]), and the business world (Teaching Smart People How to Learn,[7] The Back of the Napkin[8]), among other nonmedical professions. Within medicine, we can also re‐examine strategies long utilized in the ambulatory setting. Pascoe and colleagues offer an important example of this in their review of one‐minute preceptor (OMP) and SNAPPS, techniques developed by our colleagues in the outpatient setting but with great potential for framing discussion of clinical reasoning in the inpatient space.[9]
Applying OMP and SNAPPS to inpatient teaching presents some challenges but also genuine opportunities not found in traditional outpatient teaching. As noted by the authors, unlike the solitary learner typical of the outpatient setting, in the inpatient setting the attending is more commonly working with a group of learners of multiple levels and sometimes multiple disciplines. Furthermore, the supervising resident typical of inpatient teams is a learner who inhabits the roles of both trainee and teacher. One can imagine that if OMP and SNAPPs are applied with absolute fidelity to the inpatient setting, without reflection on venue, the teaching encounter might be overly focused on the presenting learner, leaving the rest of the team unattended to, disengaged, and not benefitting from the models. Therefore, attention to group engagement in the process is necessary for successful adaptation. Both models have the potential to help organize the group dynamic during rounds to promote broad participation. The authors describe some examples of how to engage various group members in different steps. It is worth highlighting a few key themes that enable successful use of these models in the inpatient setting.
One key theme is to teach the model to the supervising resident at the beginning of the rotation and agree, before rounds, how the attending and resident will interact as coleaders of the discussion. Because these models offer a stepwise approach to going through a case with a learner, they have the potential to demystify the teaching process, offering an accessible framework for supervising residents to learn teaching both by practicing and by comprehending what their attending is doing to lead a team through a case discussion. With attending support, the supervising resident can be encouraged to manage the team discussion, leading the team using either approach. It can be helpful to touch base briefly before rounds each day to define the teaching roles, giving the resident progressively more responsibility leading the discussion as the rotation progresses.
Another key theme is to use graduated participation. As the authors note, the group must be engaged in the discussion, and the example scenarios illustrate each step of the models being applied to the group. To ensure that the entire group remains eager to partake, the leader must maintain a nonthreatening teaching atmosphere, organizing participation in a way that does not shame learners or undermine the roles people inhabit. To this end, it can be helpful to direct questions to particular members or levels of the group at a time. When expanding participation around a specific question or concept, always work from junior members to senior members, never imposing the reverse. This principle is clearly not exclusive to using these models, but is requisite to successful adaptation of these traditionally dyadic models, in which there is no particular attention to group dynamics within the framework.
A third key theme is to utilize the unique expertise of the other health professionals on the team in steps 4, 5, and 6 of SNAPPS and step 3 of OMP. In step 4 and 5 of SNAPPS, when the teaching attending introduces the team to the model, it is important to encourage them to probe not just the teacher but other disciplines on the team for input. In the inpatient setting, these steps provide an organized point in the discussion in which to involve the other members of the professional team, modeling collaborative interdisciplinary practice.
As Pascoe et al. point out, there are limited studies of OMP and SNAPPS as teaching models in the inpatient environment. This should stimulate academic hospitalists with interest in medical education research to consider how these models might be studied. For example, in comparison to traditional inpatient teaching rounds, do these approaches provide equivalent content coverage? How do they impact the efficiency of teaching rounds? Are attendings who consistently apply these models more effective in providing feedback or assessing training milestones? How much training and practice is required to incorporate these teaching models in the inpatient environment?
Given the time pressure and increasing complexity of medical care in the hospital, coupled with the evolving needs and resources of our learners, we must seek innovative educational practices from sources outside our hospitals to provide the best possible training in hospital medicine. An outstanding recent review by Martin et al. provided an overview of other strategies for teaching in today's environment.[10] We also have much to learn from our colleagues in outpatient medicine, not only in clinical care, but also in medical education. And we have much that we have learned about teaching as hospitalists that needs to be more broadly disseminated.
ACKNOWLEDGMENTS
Disclosure: Nothing to report.
- , , , et al. Effect of the 2011 vs 2003 duty hour regulation‐compliant models on sleep duration, trainee education, and continuity of patient care among internal medicine house staff: a randomized trial. JAMA Intern Med. 2013;173(8):649–655.
- , , , , . Impact of duty‐hour restriction on resident inpatient teaching. J Hosp Med. 2009;4(8):476–480.
- . Culture shock—patient as icon, icon as patient. N Engl J Med. 2008;359(26):2748–2751.
- , , , . Smartphone use during inpatient attending rounds: prevalence, patterns and potential for distraction. J Hosp Med. 2012;7(8):595–599.
- . The Courage to Teach: Exploring the Inner Landscape of a Teacher's Life. San Francisco, CA: Jossey‐Bass; 2007.
- , . Teaching With Love 1995.
- . Teaching Smart People How to Learn. Boston, MA: Harvard Business Press; 2008.
- . The Back of the Napkin: Solving Problems and Selling Ideas With Pictures. New York, NY: Portfolio; 2008.
- , , . Maximizing teaching on the wards: review and application of the one‐minute preceptor and SNAPPS models. J Hosp Med. 2015;10(2):125–130.
- , , . Future: new strategies for hospitalists to overcome challenges in teaching on today's wards. J Hosp Med. 2013;8(7):409–413.
Hospitalists who teach in the clinical environment face challenges that include increased workload,[1] perception among trainees that there is less time to teach,[2] and competition with electronic devices for teaching engagement.[3, 4] In view of these and other challenges, we believe there is potentially much to gain from considering and adapting educational techniques that have been successful in nonhospital and even nonmedical domains. Innovative teaching methods include those designed for the grade‐school classroom (Courage to Teach,[5] Teaching With Love and Logic[6]), and the business world (Teaching Smart People How to Learn,[7] The Back of the Napkin[8]), among other nonmedical professions. Within medicine, we can also re‐examine strategies long utilized in the ambulatory setting. Pascoe and colleagues offer an important example of this in their review of one‐minute preceptor (OMP) and SNAPPS, techniques developed by our colleagues in the outpatient setting but with great potential for framing discussion of clinical reasoning in the inpatient space.[9]
Applying OMP and SNAPPS to inpatient teaching presents some challenges but also genuine opportunities not found in traditional outpatient teaching. As noted by the authors, unlike the solitary learner typical of the outpatient setting, in the inpatient setting the attending is more commonly working with a group of learners of multiple levels and sometimes multiple disciplines. Furthermore, the supervising resident typical of inpatient teams is a learner who inhabits the roles of both trainee and teacher. One can imagine that if OMP and SNAPPs are applied with absolute fidelity to the inpatient setting, without reflection on venue, the teaching encounter might be overly focused on the presenting learner, leaving the rest of the team unattended to, disengaged, and not benefitting from the models. Therefore, attention to group engagement in the process is necessary for successful adaptation. Both models have the potential to help organize the group dynamic during rounds to promote broad participation. The authors describe some examples of how to engage various group members in different steps. It is worth highlighting a few key themes that enable successful use of these models in the inpatient setting.
One key theme is to teach the model to the supervising resident at the beginning of the rotation and agree, before rounds, how the attending and resident will interact as coleaders of the discussion. Because these models offer a stepwise approach to going through a case with a learner, they have the potential to demystify the teaching process, offering an accessible framework for supervising residents to learn teaching both by practicing and by comprehending what their attending is doing to lead a team through a case discussion. With attending support, the supervising resident can be encouraged to manage the team discussion, leading the team using either approach. It can be helpful to touch base briefly before rounds each day to define the teaching roles, giving the resident progressively more responsibility leading the discussion as the rotation progresses.
Another key theme is to use graduated participation. As the authors note, the group must be engaged in the discussion, and the example scenarios illustrate each step of the models being applied to the group. To ensure that the entire group remains eager to partake, the leader must maintain a nonthreatening teaching atmosphere, organizing participation in a way that does not shame learners or undermine the roles people inhabit. To this end, it can be helpful to direct questions to particular members or levels of the group at a time. When expanding participation around a specific question or concept, always work from junior members to senior members, never imposing the reverse. This principle is clearly not exclusive to using these models, but is requisite to successful adaptation of these traditionally dyadic models, in which there is no particular attention to group dynamics within the framework.
A third key theme is to utilize the unique expertise of the other health professionals on the team in steps 4, 5, and 6 of SNAPPS and step 3 of OMP. In step 4 and 5 of SNAPPS, when the teaching attending introduces the team to the model, it is important to encourage them to probe not just the teacher but other disciplines on the team for input. In the inpatient setting, these steps provide an organized point in the discussion in which to involve the other members of the professional team, modeling collaborative interdisciplinary practice.
As Pascoe et al. point out, there are limited studies of OMP and SNAPPS as teaching models in the inpatient environment. This should stimulate academic hospitalists with interest in medical education research to consider how these models might be studied. For example, in comparison to traditional inpatient teaching rounds, do these approaches provide equivalent content coverage? How do they impact the efficiency of teaching rounds? Are attendings who consistently apply these models more effective in providing feedback or assessing training milestones? How much training and practice is required to incorporate these teaching models in the inpatient environment?
Given the time pressure and increasing complexity of medical care in the hospital, coupled with the evolving needs and resources of our learners, we must seek innovative educational practices from sources outside our hospitals to provide the best possible training in hospital medicine. An outstanding recent review by Martin et al. provided an overview of other strategies for teaching in today's environment.[10] We also have much to learn from our colleagues in outpatient medicine, not only in clinical care, but also in medical education. And we have much that we have learned about teaching as hospitalists that needs to be more broadly disseminated.
ACKNOWLEDGMENTS
Disclosure: Nothing to report.
Hospitalists who teach in the clinical environment face challenges that include increased workload,[1] perception among trainees that there is less time to teach,[2] and competition with electronic devices for teaching engagement.[3, 4] In view of these and other challenges, we believe there is potentially much to gain from considering and adapting educational techniques that have been successful in nonhospital and even nonmedical domains. Innovative teaching methods include those designed for the grade‐school classroom (Courage to Teach,[5] Teaching With Love and Logic[6]), and the business world (Teaching Smart People How to Learn,[7] The Back of the Napkin[8]), among other nonmedical professions. Within medicine, we can also re‐examine strategies long utilized in the ambulatory setting. Pascoe and colleagues offer an important example of this in their review of one‐minute preceptor (OMP) and SNAPPS, techniques developed by our colleagues in the outpatient setting but with great potential for framing discussion of clinical reasoning in the inpatient space.[9]
Applying OMP and SNAPPS to inpatient teaching presents some challenges but also genuine opportunities not found in traditional outpatient teaching. As noted by the authors, unlike the solitary learner typical of the outpatient setting, in the inpatient setting the attending is more commonly working with a group of learners of multiple levels and sometimes multiple disciplines. Furthermore, the supervising resident typical of inpatient teams is a learner who inhabits the roles of both trainee and teacher. One can imagine that if OMP and SNAPPs are applied with absolute fidelity to the inpatient setting, without reflection on venue, the teaching encounter might be overly focused on the presenting learner, leaving the rest of the team unattended to, disengaged, and not benefitting from the models. Therefore, attention to group engagement in the process is necessary for successful adaptation. Both models have the potential to help organize the group dynamic during rounds to promote broad participation. The authors describe some examples of how to engage various group members in different steps. It is worth highlighting a few key themes that enable successful use of these models in the inpatient setting.
One key theme is to teach the model to the supervising resident at the beginning of the rotation and agree, before rounds, how the attending and resident will interact as coleaders of the discussion. Because these models offer a stepwise approach to going through a case with a learner, they have the potential to demystify the teaching process, offering an accessible framework for supervising residents to learn teaching both by practicing and by comprehending what their attending is doing to lead a team through a case discussion. With attending support, the supervising resident can be encouraged to manage the team discussion, leading the team using either approach. It can be helpful to touch base briefly before rounds each day to define the teaching roles, giving the resident progressively more responsibility leading the discussion as the rotation progresses.
Another key theme is to use graduated participation. As the authors note, the group must be engaged in the discussion, and the example scenarios illustrate each step of the models being applied to the group. To ensure that the entire group remains eager to partake, the leader must maintain a nonthreatening teaching atmosphere, organizing participation in a way that does not shame learners or undermine the roles people inhabit. To this end, it can be helpful to direct questions to particular members or levels of the group at a time. When expanding participation around a specific question or concept, always work from junior members to senior members, never imposing the reverse. This principle is clearly not exclusive to using these models, but is requisite to successful adaptation of these traditionally dyadic models, in which there is no particular attention to group dynamics within the framework.
A third key theme is to utilize the unique expertise of the other health professionals on the team in steps 4, 5, and 6 of SNAPPS and step 3 of OMP. In step 4 and 5 of SNAPPS, when the teaching attending introduces the team to the model, it is important to encourage them to probe not just the teacher but other disciplines on the team for input. In the inpatient setting, these steps provide an organized point in the discussion in which to involve the other members of the professional team, modeling collaborative interdisciplinary practice.
As Pascoe et al. point out, there are limited studies of OMP and SNAPPS as teaching models in the inpatient environment. This should stimulate academic hospitalists with interest in medical education research to consider how these models might be studied. For example, in comparison to traditional inpatient teaching rounds, do these approaches provide equivalent content coverage? How do they impact the efficiency of teaching rounds? Are attendings who consistently apply these models more effective in providing feedback or assessing training milestones? How much training and practice is required to incorporate these teaching models in the inpatient environment?
Given the time pressure and increasing complexity of medical care in the hospital, coupled with the evolving needs and resources of our learners, we must seek innovative educational practices from sources outside our hospitals to provide the best possible training in hospital medicine. An outstanding recent review by Martin et al. provided an overview of other strategies for teaching in today's environment.[10] We also have much to learn from our colleagues in outpatient medicine, not only in clinical care, but also in medical education. And we have much that we have learned about teaching as hospitalists that needs to be more broadly disseminated.
ACKNOWLEDGMENTS
Disclosure: Nothing to report.
- , , , et al. Effect of the 2011 vs 2003 duty hour regulation‐compliant models on sleep duration, trainee education, and continuity of patient care among internal medicine house staff: a randomized trial. JAMA Intern Med. 2013;173(8):649–655.
- , , , , . Impact of duty‐hour restriction on resident inpatient teaching. J Hosp Med. 2009;4(8):476–480.
- . Culture shock—patient as icon, icon as patient. N Engl J Med. 2008;359(26):2748–2751.
- , , , . Smartphone use during inpatient attending rounds: prevalence, patterns and potential for distraction. J Hosp Med. 2012;7(8):595–599.
- . The Courage to Teach: Exploring the Inner Landscape of a Teacher's Life. San Francisco, CA: Jossey‐Bass; 2007.
- , . Teaching With Love 1995.
- . Teaching Smart People How to Learn. Boston, MA: Harvard Business Press; 2008.
- . The Back of the Napkin: Solving Problems and Selling Ideas With Pictures. New York, NY: Portfolio; 2008.
- , , . Maximizing teaching on the wards: review and application of the one‐minute preceptor and SNAPPS models. J Hosp Med. 2015;10(2):125–130.
- , , . Future: new strategies for hospitalists to overcome challenges in teaching on today's wards. J Hosp Med. 2013;8(7):409–413.
- , , , et al. Effect of the 2011 vs 2003 duty hour regulation‐compliant models on sleep duration, trainee education, and continuity of patient care among internal medicine house staff: a randomized trial. JAMA Intern Med. 2013;173(8):649–655.
- , , , , . Impact of duty‐hour restriction on resident inpatient teaching. J Hosp Med. 2009;4(8):476–480.
- . Culture shock—patient as icon, icon as patient. N Engl J Med. 2008;359(26):2748–2751.
- , , , . Smartphone use during inpatient attending rounds: prevalence, patterns and potential for distraction. J Hosp Med. 2012;7(8):595–599.
- . The Courage to Teach: Exploring the Inner Landscape of a Teacher's Life. San Francisco, CA: Jossey‐Bass; 2007.
- , . Teaching With Love 1995.
- . Teaching Smart People How to Learn. Boston, MA: Harvard Business Press; 2008.
- . The Back of the Napkin: Solving Problems and Selling Ideas With Pictures. New York, NY: Portfolio; 2008.
- , , . Maximizing teaching on the wards: review and application of the one‐minute preceptor and SNAPPS models. J Hosp Med. 2015;10(2):125–130.
- , , . Future: new strategies for hospitalists to overcome challenges in teaching on today's wards. J Hosp Med. 2013;8(7):409–413.
PCPs Who Adopted the Hospitalist Model
Although primary care physicians (PCPs) have traditionally treated patients in both ambulatory and hospital settings, many relinquished inpatient duties to hospitalists in recent decades.[1] Little is known about the PCPs who relinquished inpatient care duties or how the transition to the hospitalist model occurred. For example, what are the characteristics of PCPs who change? Do PCPs adopt the hospitalist model enthusiastically or cautiously? Characterizing PCPs who adopted the hospitalist model can help hospitalists understand their specialty's history and also inform health services research.
Much of the interest in the hospitalist model has been generated by studies reporting improved outcomes and lower hospital lengths of stay associated with hospitalist care.[2, 3, 4, 5] Conversely, detractors of the model point to reports of higher postacute care utilization among hospitalist patients.[6] Although these studies usually adjusted for differences among patients and hospitals, they did not account for PCP characteristics. As patients' access to PCPs and their PCP's capabilities are both plausible factors that could influence hospital length of stay (eg, decisions to complete more or less of a workup in the hospital), quality of care transitions, and postdischarge utilization, it is important to determine if PCPs who use hospitalists differ systematically from those who do not to correctly interpret health system utilization patterns that currently are attributed only to hospitalists.[7, 8]
We conducted this study to determine if observable PCP factors are associated with patients' use of hospitalists and to describe the trajectory by which PCPs referred their patients to hospitalists over time.
METHODS
Source of Data
We used claims data from 100% of Texas Medicare beneficiaries from 2000 to 2009, including Medicare beneficiary summary files, Medicare Provider Analysis and Review (MedPAR) files, Outpatient Standard Analytical Files (OutSAF), and Medicare Carrier files. Diagnosis related group (DRG)‐associated information, including weights, and Major Diagnostic Categories, were obtained from Centers for Medicare & Medicaid Services (
Establishment of the Study Cohort
Using the MedPAR file, we first selected hospital admissions from acute care hospitals in Texas for each year of the study period. We excluded beneficiaries younger than 66 years old, with incomplete Medicare Parts A and B enrollment, or with any health maintenance organization enrollment in the 12 months prior to the admission of interest. For patients with more than 1 admission in a given year, we randomly selected 1 admission. We then attempted to assign each patient to a PCP. We defined a PCP as a generalist (general practitioner, family physician, internist, or geriatrician) who saw a given beneficiary on 3 or more occasions in an outpatient setting in the year prior to the admission of interest.[9] We identified outpatient visits using Current Procedural Terminology (CPT) codes 99201 to 99205 (new patient encounters), and 99211 to 99215 (established patient encounters) from Carrier files. If more than 1 generalist physician saw the beneficiary on 3 or more occasions in a given year, the physician with more than 75% of the total outpatient evaluation and management (E&M) billings was classified as the beneficiary's PCP. Using these criteria, approximately 66% of patients were assigned to a PCP.
For cross‐sectional analyses, we restricted our cohort to beneficiaries whose PCPs were associated with at least 20 inpatients in a given year. To study trends in PCP practice patterns over time, we further restricted the cohort to beneficiaries whose PCPs were associated with at least 20 inpatients in every year of the study period, resulting in 1172 PCPs for the trajectory analyses. The reliability of PCPs' practice profiles increases as the number of patients in their panel increases. We chose 20 inpatients as the minimum because PCPs with 20 hospitalized patients per study year would achieve a reliability of 0.9 for estimating the proportion of their patients that received care from hospitalists.[10]
Identification of Hospitalists
We defined hospitalists as generalists who had at least 100 E&M billings in a given year and generated at least 90% of their total E&M billings in the year from inpatient services.[1] Inpatient E&M billings were identified by CPT codes 99221 to 99223 (new or established patient encounters), 99231 to 99233 (subsequent hospital care), and 99251 to 99255 (inpatient consultations).[1]
Patient Measures
Patient demographic information including, age at admission, gender, race/ethnicity, and Medicaid eligibility were obtained from Medicare beneficiary summary files. We used the Medicaid indicator as a proxy for low socioeconomic status. Information on weekday versus weekend admission, emergent admission, and DRG were obtained from MedPAR files. The DRG category (circulatory system, digestive system, infectious disease, nervous system, respiratory system, or other) was determined based on its Major Diagnostic Category. We determined residence in a nursing facility in the 3 months before the admission of interest from the MedPAR files and by E&M codes 99304 to 99318 (nursing facility services) from Carrier files.[11] Comorbidities were identified using the claims from MedPAR, Carrier, and OutSAF files in the year prior to the admission of interest.[12] Total hospitalizations and outpatient visits in the prior year were identified from MedPAR files and Carrier files, respectively.
PCP Measures
We categorized PCPs by specialty (general practice, gamily practice, geriatric medicine, or internal medicine), years in practice, gender, US‐ versus foreign‐trained, metropolitan statistical area (MSA) of their practice location, and board certification status. The specialty was identified from Carrier files and the other information from AMA data. For each PCP, the total number of outpatient visits and total number of patients seen as outpatients in each year was calculated based on E&M codes (9920199205, 9921199215) from Carrier files. For each year, we computed the average outpatient age, gender, race, and outpatient comorbidity for each PCP's patient panel. We computed hospital volumes using the number of hospitalized patients associated with each PCP in the study cohort.
Study Outcome
To determine whether hospitalized patients received care from hospitalists during a given hospitalization, we identified all inpatient E&M bills from generalist physicians during the admission of interest by linking MedPAR and Carrier files. If more than 50% of the generalist inpatient E&M billings from generalist physicians were from 1 or more hospitalists, the patient was considered to have received care from hospitalists.
Statistical Analyses
Multilevel analyses were used to account for the clustering of patients within PCPs. All multilevel models were adjusted for patient characteristics including age, race/ethnicity, gender, Medicaid eligibility, emergency admission, weekend admission, DRG weight, DRG category, any nursing home stay in the prior 3 months, number of comorbidities, number of hospitalizations, and number of physician visits in the year prior to the admission of interest. To analyze trends in practice patterns, we first used multilevel models to calculate the proportions of inpatients cared for by hospitalists each year for each of the 1172 PCPs with at least 20 patients. Then we employed an SAS procedure (PROC TRAJ) developed by Jones et al. to classify these PCPs into groups based on their trajectories.[13] This group‐based trajectory modeling allowed us to identify relatively homogeneous clusters within a heterogeneous sample population.[14] We chose a model that classified the PCPs into 4 groups.[15] With 4 groups, the average of the posterior probabilities of group membership for the PCPs assigned to each group exceeded 0.93, indicating a low rate of misclassification among these 4 distinct groups. For the 1172 PCPs, we tested interactions between year of hospitalization and PCP characteristics while adjusting for patient characteristics in order to investigate whether or not the impacts of PCP characteristics on how likely their patients being cared for by hospitalists differed with time. All analyses were performed with SAS version 9.2 (SAS Institute Inc., Cary, NC).
RESULTS
During the 2001 through 2009 study period, between 2252 and 2848 PCPs were associated with at least 20 hospitalized beneficiaries in any single year. Among these, 1172 PCPs were associated with at least 20 hospitalized beneficiaries in every year of the study period. These 1172 PCPs were associated with 608,686 hospitalizations over the 9 years.
Table 1 presents the characteristics of the PCPs who contributed to the cross‐sectional analyses in 2001 (N=2252) and 2009 (N=2387), as well as the 1172 PCPs for whom we had data for all 9 years for the longitudinal analyses. Most PCPs were male, trained in the United States, and were board certified. The average number of Medicare patients seen by these PCPs and number of outpatient Medicare visits went up about 7% between 2001 and 2009.
| PCP Characteristics | Cross‐Sectional Analysis | Trajectory Analysis, 20012009 | |
|---|---|---|---|
| 2001 | 2009 | ||
| |||
| Overall, no. (%) | 2,252 (100%) | 2,387 (100%) | 1,172 (100%) |
| Specialty, no. (%) | |||
| General practice | 39 (1.7%) | 34 (1.4%) | 15 (1.3%) |
| Family practice | 948 (42.1%) | 1,089 (45.6%) | 466 (39.8%) |
| Internal medicine | 1,255 (55.7%) | 1,249 (52.3%) | 688 (58.7%) |
| Geriatrics | 10 (0.4%) | 15 (0.6%) | 3 (0.3%) |
| Gender, no. (%) | |||
| Male | 1,990 (88.4%) | 2,015 (84.4%) | 1,072 (91.5%) |
| Female | 262 (11.6%) | 372 (15.6%) | 100 (8.5%) |
| Trained in the United States, no. (%) | |||
| Yes | 1,669 (74.1%) | 1,738 (72.8%) | 844 (72.0%) |
| No | 583 (25.9%) | 649 (27.2%) | 328 (28.0%) |
| Metropolitan statistical area, no. (%) | |||
| 99,999 or less | 417 (17.5) | 237 (20.2) | |
| 100,000249,000 | 438 (18.3) | 234 (20.0) | |
| 250,000999,999 | 381 (16.0) | 216 (18.4) | |
| 1,000,000 or more | 1,151 (48.2) | 485 (41.4) | |
| Board certification, no. (%) | |||
| Yes | 1,657 (69.4%) | 800 (68.3%) | |
| No | 730 (30.6%) | 372 (31.7%) | |
| Years in practice, 2001, meanSD (Q1Q3) | 22.310.6 (15.028.0) | 21.28.9 (15.027.0) | |
| Years in practice, 2009, meanSD (Q1Q3) | 25.010.2 (17.032.0) | 29.28.9 (23.035.0) | |
| Total no. of Medicare outpatient visits, 2001, meanSD (Q1Q3) | 1,624.8879.2 (1,057.51,970.0) | 1,883.39,48.5 (1,236.52,240.5) | |
| Total no. of Medicare outpatient visits, 2009, meanSD (Q1Q3) | 1,733.81,053.3 (1,080.02,048.0) | 2,020.51,200.9 (1,334.52,373.0) | |
| Total no. of Medicare outpatients, 2001, meanSD (Q1Q3) | 418.6186.9 (284.0522.0) | 473.4189.5 (338.0580.5) | |
| Total no. of Medicare outpatients, 2009, meanSD (Q1Q3) | 448.7217.8 (300.0548.0) | 508.7238.2 (350.5615.0) | |
| No. of hospitalized patients, 2001, meanSD (Q1Q3) | 46.025.0 (27.057.0) | 53.028.0 (32.066.0) | |
| No. of hospitalized patients, 2009, meanSD (Q1Q3) | 44.024.0 (26.052.0) | 52.027.0 (33.065.0) | |
| Average outpatient age, 2001, meanSD (Q1Q3) | 72.82.3 (71.574.2) | 72.82.1 (71.774.1) | |
| Average outpatient age, 2009, meanSD (Q1Q3) | 72.12.8 (70.673.9) | 72.82.7 (71.474.5) | |
| Average outpatient gender (% male), 2001, meanSD (Q1Q3) | 38.17.0 (35.542.3) | 38.56.4 (36.242.3) | |
| Average outpatient gender (% male), 2009, meanSD (Q1Q3) | 40.27.6 (37.644.8) | 41.06.5 (38.644.8) | |
| Average outpatient race (% white), 2001, meanSD (Q1Q3) | 84.316.4 (79.295.5) | 85.414.3 (79.995.7) | |
| Average outpatient race (% white), 2009, meanSD (Q1Q3) | 85.214.4 (79.895.2) | 86.312.9 (80.895.6) | |
| Average outpatient comorbidity, 2001, meanSD (Q1Q3)a | 1.60.5 (1.21.8) | 1.60.4 (1.21.8) | |
| Average outpatient comorbidity, 2009, meanSD (Q1Q3)a | 2.20.6 (1.82.5) | 2.20.6 (1.72.5) | |
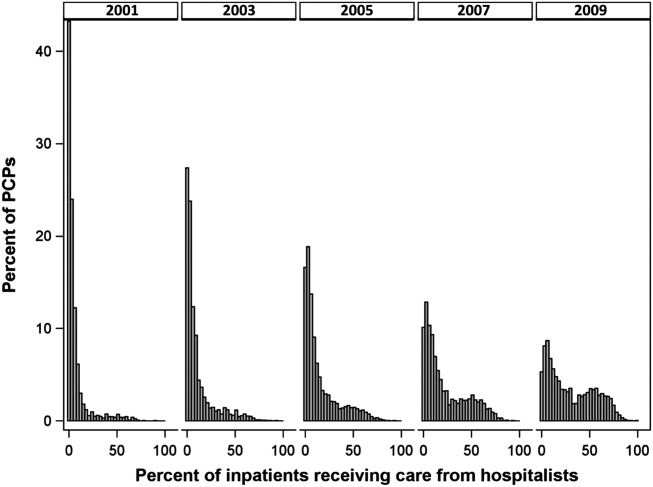
Figure 1 graphs the percentage of PCPs as a function of what percent of their hospitalized patients received care from hospitalists, and how that changed from 2001 to 2009. For 70.9% of PCPs, fewer than 5% of their hospitalized patients received hospitalist care in 2001. By 2009, the percent of PCPs in this category had decreased to 15.2%. In contrast, in 2001, more than half of the patients for 2.1% of PCPs received hospitalist care, and the percent of PCPs in this category increased to 26.3% by 2009.
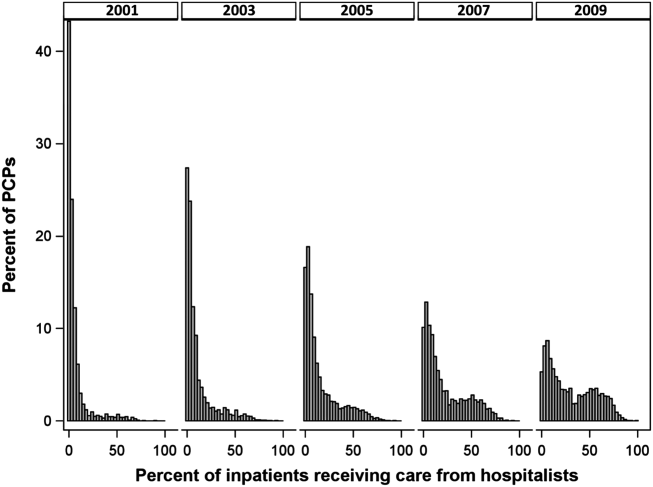
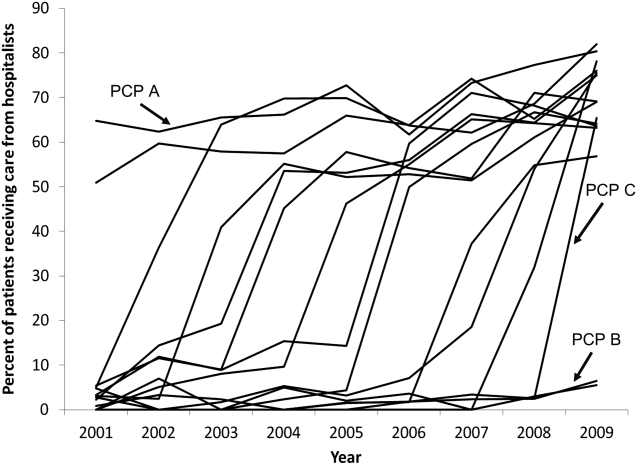
The pattern in Figure 1 shows that PCPs' use of hospitalists changed continuously and gradually over time. However, this pattern describes the PCPs as a group. When examined at the individual PCP level, different patterns emerge. Figure 2, which presents selected individual PCP's use of hospitalists over time, shows several distinct subpatterns of PCP practice behaviors. First, there are PCPs whose use of hospitalists was high in 2001 and stayed high or increased over time (eg, PCP A). There also were PCPs whose use of hospitalists stayed low over the entire study period (eg, PCP B). Finally, there were PCPs whose use of hospitalists was low in 2001 but high in 2009 (eg, PCP C). For this last group, the pattern of change in hospitalist utilization over time was discontinuous; that is, most of the increase occurred over a 1‐ or 2‐year period, instead of increasing gradually over time.
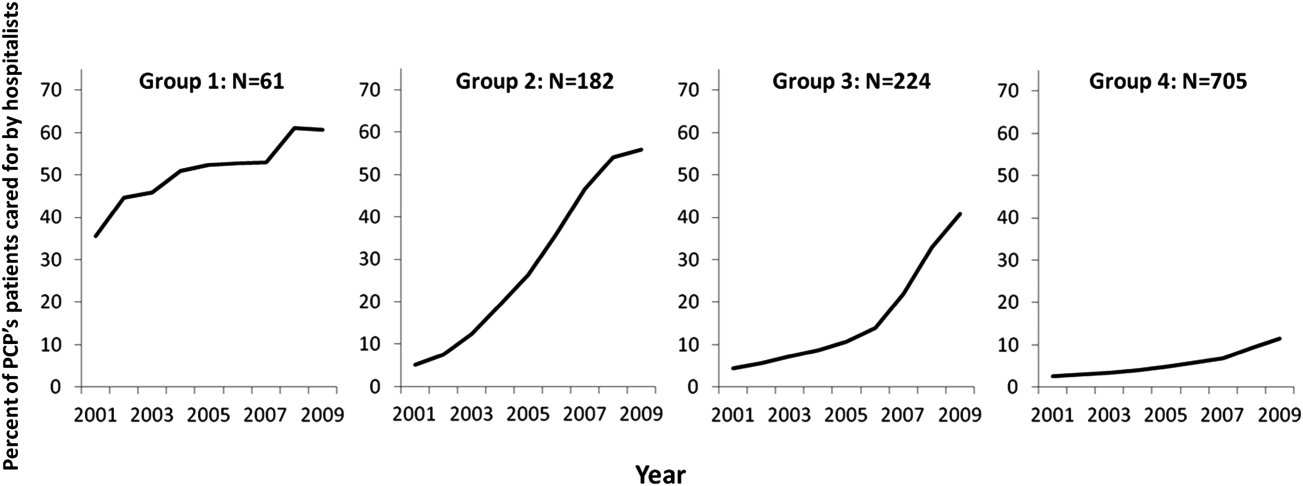
Among the 1172 PCPs associated with 20 hospitalized beneficiaries each year in all 9 years of the study period, group‐based trajectory modeling classified their practice patterns into 4 distinct trajectories (Figure 3). Among PCPs in group 1, more than one‐third of their hospitalized patients were cared for by hospitalists in 2001, and this increased to 60% by 2009. PCPs in groups 2 and 3 rarely used hospitalist care in 2001 but increased their use over time. The increase started early in the period for PCPs in group 2 and later for those in group 3. PCPs in group 4 were associated with little hospitalist use throughout the study period.
We constructed a model to describe the odds of a patient receiving care from hospitalists during the study period using patients associated with these 1172 PCPs. After adjusting for patient characteristics, the residual intraclass correlation coefficient for PCP level was 0.334, which indicates that 33.4% of the variance in whether a hospitalized patient received care from a hospitalist is explained by which PCP the patient saw. When adjusting for both patient and PCP characteristics, the overall odds of a patient receiving hospitalist care increased by 30% (95% confidence interval [CI]: 1.29‐1.30) per year from 2001 through 2009.
There were also significant interactions between year of hospitalization and several PCP characteristics. These interactions are illustrated in Table 2, which stratifies each of those PCP characteristics by 3 time periods: 2001 to 2003, 2004 to 2006, and 2007 to 2009. In all time periods, patients were more likely to receive hospitalist care if their PCP was US trained (US vs international medical graduate: odds ratio [OR]: 1.42, 95% CI: 1.19‐1.69 in 20012003; OR: 1.46, 95% CI: 1.23‐1.73 in 20072009), or specialized in family medicine (family medicine vs internal medicine: OR: 1.46, 95% CI: 1.25‐1.72 in 20012003; OR: 1.46, 95% CI: 1.25‐1.70 in 20072009). Over time, the relative odds of a patient receiving care from hospitalists decreased if their PCP was female (female vs male: OR: 1.91, 95% CI: 1.46‐2.50 in 20012003 vs OR: 1.50, 95% CI: 1.15‐1.95 in 20072009) or practiced in an urban area (largest vs smallest MSA: OR: 3.34, 95% CI: 2.72‐4.09 in 20012003; OR: 2.22, 95% CI: 1.82‐2.71 in 20072009). Although the longest‐practicing PCPs were most likely to use hospitalists in the early 2000s, this effect disappeared by 2007 to 2009 (most vs least years in practice: OR: 1.35, 95% CI: 1.06‐1.72 in 20012003 vs OR: 0.92, 95% CI: 0.73‐1.17 in 20072009).
| PCP Characteristics | 20012003, OR (95% CI) | 20042006, OR (95% CI) | 20072009, OR (95% CI) |
|---|---|---|---|
| |||
| Family practicea vs. internal medicineb | 1.46 (1.251.72) | 1.50 (1.281.76) | 1.46 (1.251.70) |
| Female vs male | 1.91 (1.462.50) | 1.43 (1.091.86) | 1.50 (1.151.95) |
| United States trained (yes vs no) | 1.42 (1.191.69) | 1.53 (1.281.81) | 1.46 (1.231.73) |
| Metropolitan statistical area | |||
| 99,999 or less | 1.00 | 1.00 | 1.00 |
| 100,000249,000 | 0.83 (0.651.05) | 1.00 (0.791.25) | 1.13 (0.901.41) |
| 250,000999,999 | 0.92 (0.721.17) | 1.03 (0.821.31) | 0.98 (0.771.23) |
| 1,000,000 or more | 3.34 (2.724.09) | 2.90 (2.373.54) | 2.22 (1.822.71) |
| Years in practice, 2001 | |||
| Q1 (lowest) | 1.00 | 1.00 | 1.00 |
| Q2 | 0.89 (0.711.12) | 0.83 (0.671.04) | 0.92 (0.741.14) |
| Q3 | 1.06 (0.841.34) | 0.99 (0.791.24) | 1.03 (0.821.29) |
| Q4 | 1.25 (0.991.59) | 1.13 (0.891.42) | 1.15 (0.921.45) |
| Q5 (highest) | 1.35 (1.061.72) | 1.05 (0.831.33) | 0.92 (0.731.17) |
| Total no. of outpatient visitsc | |||
| Q1 (lowest) | 1.00 | 1.00 | 1.00 |
| Q2 | 1.21 (1.121.30) | 1.07 (1.001.14) | 1.13 (1.071.19) |
| Q3 | 1.42 (1.301.54) | 1.18 (1.091.27) | 1.14 (1.071.22) |
| Q4 | 1.34 (1.211.47) | 1.34 (1.231.46) | 1.25 (1.161.35) |
| Q5 (highest) | 1.46 (1.301.63) | 1.33 (1.211.47) | 1.32 (1.201.44) |
| No. of hospitalized patientsc | |||
| Q1 (lowest) | 1.00 | 1.00 | 1.00 |
| Q2 | 1.07 (1.001.15) | 0.91 (0.860.96) | 0.85 (0.810.89) |
| Q3 | 1.00 (0.921.08) | 0.87 (0.820.93) | 0.74 (0.700.79) |
| Q4 | 0.89 (0.810.97) | 0.76 (0.710.82) | 0.62 (0.580.67) |
| Q5 (highest) | 1.05 (0.951.18) | 0.67 (0.610.73) | 0.55 (0.510.60) |
| Average outpatient agec | |||
| Q1 (lowest) | 1.00 | 1.00 | 1.00 |
| Q2 | 0.94 (0.871.01) | 1.15 (1.081.23) | 1.18 (1.111.25) |
| Q3 | 0.82 (0.760.90) | 1.05 (0.971.13) | 1.17 (1.091.25) |
| Q4 | 0.71 (0.650.79) | 1.03 (0.951.12) | 1.10 (1.021.19) |
| Q5 (highest) | 0.72 (0.640.81) | 1.12 (1.011.23) | 1.15 (1.051.26) |
| Average outpatient gender (% male)c | |||
| Q1 (lowest) | 1.00 | 1.00 | 1.00 |
| Q2 | 1.10 (1.021.18) | 1.19 (1.101.27) | 1.27 (1.181.37) |
| Q3 | 1.12 (1.031.22) | 1.27 (1.171.37) | 1.43 (1.321.54) |
| Q4 | 1.36 (1.251.48) | 1.49 (1.371.61) | 1.52 (1.401.65) |
| Q5 (highest) | 1.47 (1.341.61) | 1.84 (1.682.00) | 1.68 (1.541.83) |
| Average outpatient race (% white)c | |||
| Q1 (lowest) | 1.00 | 1.00 | 1.00 |
| Q2 | 1.08 (0.981.20) | 1.01 (0.921.10) | 1.23 (1.131.34) |
| Q3 | 1.27 (1.131.43) | 1.06 (0.951.18) | 1.21 (1.091.34) |
| Q4 | 1.47 (1.291.67) | 0.97 (0.861.09) | 1.33 (1.181.48) |
| Q5 (highest) | 1.39 (1.211.59) | 1.18 (1.041.34) | 1.25 (1.101.42) |
| Average outpatient comorbidityc | |||
| Q1 (lowest) | 1.00 | 1.00 | 1.00 |
| Q2 | 1.26 (1.191.35) | 1.23 (1.161.31) | 1.22 (1.141.30) |
| Q3 | 1.62 (1.491.75) | 1.61 (1.501.72) | 1.43 (1.341.54) |
| Q4 | 1.96 (1.792.15) | 1.86 (1.722.02) | 1.59 (1.471.72) |
| Q5 (highest) | 1.79 (1.592.01) | 2.20 (2.002.41) | 2.03 (1.852.22) |
In terms of PCP workload, patients of PCPs with high outpatient activity were more likely to receive hospitalists care throughout the study period, although the association had decreased by 2007 to 2009 (highest vs lowest outpatient volume: OR: 1.46, 95% CI: 1.30‐1.63 in 20012003 vs OR: 1.32, 95% CI: 1.20‐1.44 in 20072009). In contrast, PCPs with the lowest inpatient volumes became more likely to use hospitalists by the end of the study period (highest vs lowest inpatient volume: OR: 1.05, 95% CI: 0.95‐1.18 in 20012003 vs OR: 0.55, 95% CI: 0.51‐0.60 in 20072009).
The characteristics of PCPs' practice panels also were associated with patients' likelihood of receiving care from hospitalists. PCPs whose practice panels consisted of patients who were predominantly male, white, or with more outpatient comorbidities were consistently more likely to use hospitalists throughout the study period. PCPs with older patient panels were less likely to use hospitalists in 2001 to 2003, but by 2007 to 2009, they were slightly more likely to do so (oldest vs youngest average outpatient panel age: OR: 0.72, 95% CI: 0.64‐0.81 in 20012003 vs OR: 1.15, 95% CI: 1.05‐1.26 in 20072009).
CONCLUSIONS
Prior studies of the hospitalist model have shown that the likelihood of a patient receiving inpatient care from hospitalists is associated with patient characteristics, hospital characteristics, geographic region, and type of admission.[1, 16, 17] We found that PCP characteristics also predict whether patients receive care from hospitalists and that their use of hospitalists developed dynamically between 2001 to 2009. Although many factors (such as whether patients were admitted to a hospital where their PCP had admitting privileges) can influence the decision to use hospitalists, we found that over one‐third of the variance in whether a hospitalized patient received care from a hospitalist is explained by which PCP the patient saw. In showing that systemic differences exist among PCPs who use hospitalists and those who do not, our study suggests that future research on the hospitalist model should, if possible, adjust for PCP characteristics in addition to hospital and patient factors.
Although this study identifies the existence and magnitude of differences in whether or not PCPs use hospitalists, it cannot explain why the differences exist. We only can offer hypotheses. For example, our finding that PCPs with the most years of practice experience were more likely to use hospitalists in the early 2000s but not in more recent years suggests that in hospital medicine's early years, long‐practicing generalist physicians were choosing between practicing traditionalist medicine and adopting the hospitalists model, but by 2009, experienced generalist physicians had already specialized to either inpatient or outpatient settings earlier in their careers. On the other hand, the decreasing odds of urban PCPs using hospitalists may reflect a relative growth in hospitalist use in less populated areas rather than a change in urban PCPs' practice patterns.
PCPs trained in family medicine have reported less inpatient training and less comfort with providing hospital care,[18, 19] thus it is unsurprising that family physicians were more likely to refer patients to hospitalists. Although a recent study reported that family physicians' inpatient volumes remained constant, whereas those of outpatient internists declined between 2003 and 2012, the analysis used University Health Consortium data and thus reflects practice patterns in academic medical centers.[20] Our data suggest that outside of academia, family physicians have embraced the hospitalists as clinical partners.
Meltzer and Chung had previously proposed an economic model to describe the growing use of hospitalists in the United States. They posited that decisions to adopt the hospitalist model are governed by trade‐offs between coordination costs (eg, time and effort spent coordinating multiple providers across different settings) and switching costs (eg, time spent traveling between the office and the hospital or the effort of adjusting to different work settings).[16] The authors hypothesized that empirical testing of this model would show PCPs are more likely to use hospitalists if they have less available professional time (ie, work fewer hours per week), are female (due to competing demands from domestic responsibilities), have relatively few hospitalized patients, or live in areas with high traffic congestion. Our findings provide empirical evidence to support their division‐of‐labor model in showing that patients were more likely to receive hospitalist care if their PCP was female, practiced in an urban location, had higher outpatient practice volumes, or had lower inpatient volumes.
At first glance, some of our findings appear to contradict our earlier study, which showed that younger, black, male patients are more likely to receive inpatient care from hospitalists.[1] However, that study included patients regardless of whether they had a PCP. This study shows that when patients have a PCP, their PCPs are more likely to refer them to hospitalists if they are older, white, male, and have more comorbid conditions. A potential explanation for this finding is that PCPs may preferentially use hospitalists when caring for older and sicker hospitalized patients. For example, commentators often cite hospitalists' constant availability in the hospital as a valuable resource when caring for acutely ill patients.[21, 22]
Another potential explanation is that despite their preferences, PCPs who care for younger, minority patients lack access to hospitalist services. One large study of Medicare beneficiaries reported that physicians who care for black patients are less well‐trained clinically and often lack access to important clinical resources such as diagnostic imaging and nonemergency hospital admissions.[23] Similarly, international medical graduates are more likely than their US‐trained counterparts to care for underserved patients and to practice in small, independent offices.[24, 25, 26] As hospitalist groups often rely on cross‐subsidization from sources within a large healthcare organization, independent PCPs may have less access to their services when compared with PCPs in managed care organizations or large integrated groups. Viewed in this context, our findings imply that although hospitalists often care for socioeconomically vulnerable patients (eg, younger, uninsured, black men) who lack access to primary care services,[1] they also appear to share care responsibilities for more complex hospitalized patients with PCPs in more affluent communities. Further research may determine if the availability of hospitalists influences racial disparities in hospital care.
Our study has limitations. It is an observational study and thus subject to bias and confounding. As our cohort was formed using fee‐for‐service Medicare data in a single, large state, it may not be generalizable to PCPs who practice in other states, who care for a younger population, or who do not accept Medicare. Our findings also may not reflect the practice patterns of physicians‐in‐training, PCP populations with high board‐certification rates, those employed in temporary positions, or those who interrupt their practices for personal reasons, as we restricted our study to established PCPs who had been in practice long and consistently enough to be associated with 20 hospitalized patients during every year of the study. For example, the lower proportion of female PCPs in our cohort (15.6% in our study in 2009 vs 27.5% reported in a nationally representative 2008 survey[27]) may be explained by our exclusion of women who take prolonged time off for childcare duties. We also did not establish whether patient outcomes or healthcare costs differ between PCPs who adopted the hospitalist model and traditionalists. Finally, we could not examine the effect of a number of PCP factors that could plausibly influence whether or not PCPs relinquish inpatient care to hospitalists, such as their comfort with providing inpatient care, having hospital admitting privileges, having office‐based access to hospitals' electronic medical records, or the distance between their office and the hospital. However, this study lays the groundwork for future studies to explore these factors.
In summary, this study is the first, to our knowledge, to characterize PCPs who relinquished inpatient responsibilities to hospitalists. Our findings suggest that some groups of PCPs are more likely to refer patient to hospitalists, that the relationship between hospitalists and PCPs has evolved over time, and that the hospitalist model still has ample room to grow.
ACKNOWLEDGMENTS
Disclosures: This study was supported by grants from the National Institute on Aging (1RO1‐AG033134 and P30‐AG024832) and the National Cancer Institute (K05‐CA124923). The authors have no financial conflicts of interest to disclose. An oral abstract of this article was presented on May 18, 2013 at the Society of Hospital Medicine Annual Meeting in National Harbor, Maryland.
- , , , . Growth in the care of older patients by hospitalists in the United States. N Engl J Med. 2009;360(11):1102–1112.
- , . Effect of hospitalists on length of stay in the medicare population: variation according to hospital and patient characteristics. J Am Geriatr Soc. 2010;58(9):1649–1657.
- , , , , , . Outcomes of care by hospitalists, general internists, and family physicians. N Engl J Med. 2007;357(25):2589–2600.
- , , , , . Hospitalist care and length of stay in patients requiring complex discharge planning and close clinical monitoring. Arch Intern Med. 2007;167(17):1869–1874.
- , . The impact of hospitalists on the cost and quality of inpatient care in the United States: a research synthesis. Med Care Res Rev. 2005;62(4):379–406.
- , . Association of hospitalist care with medical utilization after discharge: evidence of cost shift from a cohort study. Ann Intern Med. 2011;155(3):152–159.
- , . Hospital care and medical utilization after discharge. Ann Intern Med. 2011;155(10):719–720; author reply 722.
- . Hospital care and medical utilization after discharge. Ann Intern Med. 2011;155(10):721; author reply 722.
- , , , , , . Administrative data algorithms can describe ambulatory physician utilization. Health Serv Res. 2007;42:1783–1796.
- , . Estimating the reliability of continuous measures with Cronbach's alpha or the intraclass correlation coefficient: toward the integration of two traditions. J Clin Epidemiol. 1991;44(4–5):381–390.
- , , . Ability of Medicare claims data to identify nursing home patients: a validation study. Med Care. 2008;46(11):1184–1187.
- , , , . Comorbidity measures for use with administrative data. Med Care. 1998;36(1):8–27.
- , , . A SAS procedure based on mixture models for estimating developmental trajectories. Sociol Methods Res. 2001;29(3):374–393.
- . Group‐Based Modeling of Development. Cambridge, MA: Harvard University Press; 2005.
- , . Group‐based trajectory modeling in clinical research. Annu Rev Clin Psychol. 2010;6:109–138.
- , . Coordination, switching costs and the division of labor in general medicine: an economic explanation for the emergence of hospitalists in the United States. National Bureau of Economic Research Working Paper Series No. 16040. Cambridge, MA: National Bureau of Economic Research; 2010.
- , , , , , . Continuity of outpatient and inpatient care by primary care physicians for hospitalized older adults. JAMA. 2009;301(16):1671–1680.
- . Hospitalists and family physicians: understanding opportunities and risks. J Fam Pract. 2004;53(6):473–481.
- , , , , , . Preparedness of internal medicine and family practice residents for treating common conditions. JAMA. 2002;288(20):2609–2614.
- , , , . The status of adult inpatient care by family physicians at US academic medical centers and affiliated teaching hospitals 2003 to 2012: the impact of the hospitalist movement. Fam Med. 2014;46(2):94–99.
- . Hospitalists and the hospital medicine system of care are good for patient care. Arch Intern Med. 2008;168(12):1254–1256; discussion 1259–1260.
- . Hospitalists in the United States—mission accomplished or work in progress? N Engl J Med. 2004;350(19):1935–1936.
- , , , , . Primary care physicians who treat blacks and whites. N Engl J Med. 2004;351(6):575–584.
- , , , . International medical graduates and the primary care workforce for rural underserved areas. Health Aff (Millwood). 2003;22(2):255–262.
- , , . Medical migration and the physician workforce. International medical graduates and American medicine. JAMA. 1995;273(19):1521–1527.
- , , , , . International medical graduates in family medicine in the United States of America: an exploration of professional characteristics and attitudes. Hum Resour Health. 2006;4:17.
- , , . A snapshot of U.S. physicians: key findings from the 2008 Health Tracking Physician Survey. Data Bull (Cent Stud Health Syst Change). 2009(35):1–11.
Although primary care physicians (PCPs) have traditionally treated patients in both ambulatory and hospital settings, many relinquished inpatient duties to hospitalists in recent decades.[1] Little is known about the PCPs who relinquished inpatient care duties or how the transition to the hospitalist model occurred. For example, what are the characteristics of PCPs who change? Do PCPs adopt the hospitalist model enthusiastically or cautiously? Characterizing PCPs who adopted the hospitalist model can help hospitalists understand their specialty's history and also inform health services research.
Much of the interest in the hospitalist model has been generated by studies reporting improved outcomes and lower hospital lengths of stay associated with hospitalist care.[2, 3, 4, 5] Conversely, detractors of the model point to reports of higher postacute care utilization among hospitalist patients.[6] Although these studies usually adjusted for differences among patients and hospitals, they did not account for PCP characteristics. As patients' access to PCPs and their PCP's capabilities are both plausible factors that could influence hospital length of stay (eg, decisions to complete more or less of a workup in the hospital), quality of care transitions, and postdischarge utilization, it is important to determine if PCPs who use hospitalists differ systematically from those who do not to correctly interpret health system utilization patterns that currently are attributed only to hospitalists.[7, 8]
We conducted this study to determine if observable PCP factors are associated with patients' use of hospitalists and to describe the trajectory by which PCPs referred their patients to hospitalists over time.
METHODS
Source of Data
We used claims data from 100% of Texas Medicare beneficiaries from 2000 to 2009, including Medicare beneficiary summary files, Medicare Provider Analysis and Review (MedPAR) files, Outpatient Standard Analytical Files (OutSAF), and Medicare Carrier files. Diagnosis related group (DRG)‐associated information, including weights, and Major Diagnostic Categories, were obtained from Centers for Medicare & Medicaid Services (
Establishment of the Study Cohort
Using the MedPAR file, we first selected hospital admissions from acute care hospitals in Texas for each year of the study period. We excluded beneficiaries younger than 66 years old, with incomplete Medicare Parts A and B enrollment, or with any health maintenance organization enrollment in the 12 months prior to the admission of interest. For patients with more than 1 admission in a given year, we randomly selected 1 admission. We then attempted to assign each patient to a PCP. We defined a PCP as a generalist (general practitioner, family physician, internist, or geriatrician) who saw a given beneficiary on 3 or more occasions in an outpatient setting in the year prior to the admission of interest.[9] We identified outpatient visits using Current Procedural Terminology (CPT) codes 99201 to 99205 (new patient encounters), and 99211 to 99215 (established patient encounters) from Carrier files. If more than 1 generalist physician saw the beneficiary on 3 or more occasions in a given year, the physician with more than 75% of the total outpatient evaluation and management (E&M) billings was classified as the beneficiary's PCP. Using these criteria, approximately 66% of patients were assigned to a PCP.
For cross‐sectional analyses, we restricted our cohort to beneficiaries whose PCPs were associated with at least 20 inpatients in a given year. To study trends in PCP practice patterns over time, we further restricted the cohort to beneficiaries whose PCPs were associated with at least 20 inpatients in every year of the study period, resulting in 1172 PCPs for the trajectory analyses. The reliability of PCPs' practice profiles increases as the number of patients in their panel increases. We chose 20 inpatients as the minimum because PCPs with 20 hospitalized patients per study year would achieve a reliability of 0.9 for estimating the proportion of their patients that received care from hospitalists.[10]
Identification of Hospitalists
We defined hospitalists as generalists who had at least 100 E&M billings in a given year and generated at least 90% of their total E&M billings in the year from inpatient services.[1] Inpatient E&M billings were identified by CPT codes 99221 to 99223 (new or established patient encounters), 99231 to 99233 (subsequent hospital care), and 99251 to 99255 (inpatient consultations).[1]
Patient Measures
Patient demographic information including, age at admission, gender, race/ethnicity, and Medicaid eligibility were obtained from Medicare beneficiary summary files. We used the Medicaid indicator as a proxy for low socioeconomic status. Information on weekday versus weekend admission, emergent admission, and DRG were obtained from MedPAR files. The DRG category (circulatory system, digestive system, infectious disease, nervous system, respiratory system, or other) was determined based on its Major Diagnostic Category. We determined residence in a nursing facility in the 3 months before the admission of interest from the MedPAR files and by E&M codes 99304 to 99318 (nursing facility services) from Carrier files.[11] Comorbidities were identified using the claims from MedPAR, Carrier, and OutSAF files in the year prior to the admission of interest.[12] Total hospitalizations and outpatient visits in the prior year were identified from MedPAR files and Carrier files, respectively.
PCP Measures
We categorized PCPs by specialty (general practice, gamily practice, geriatric medicine, or internal medicine), years in practice, gender, US‐ versus foreign‐trained, metropolitan statistical area (MSA) of their practice location, and board certification status. The specialty was identified from Carrier files and the other information from AMA data. For each PCP, the total number of outpatient visits and total number of patients seen as outpatients in each year was calculated based on E&M codes (9920199205, 9921199215) from Carrier files. For each year, we computed the average outpatient age, gender, race, and outpatient comorbidity for each PCP's patient panel. We computed hospital volumes using the number of hospitalized patients associated with each PCP in the study cohort.
Study Outcome
To determine whether hospitalized patients received care from hospitalists during a given hospitalization, we identified all inpatient E&M bills from generalist physicians during the admission of interest by linking MedPAR and Carrier files. If more than 50% of the generalist inpatient E&M billings from generalist physicians were from 1 or more hospitalists, the patient was considered to have received care from hospitalists.
Statistical Analyses
Multilevel analyses were used to account for the clustering of patients within PCPs. All multilevel models were adjusted for patient characteristics including age, race/ethnicity, gender, Medicaid eligibility, emergency admission, weekend admission, DRG weight, DRG category, any nursing home stay in the prior 3 months, number of comorbidities, number of hospitalizations, and number of physician visits in the year prior to the admission of interest. To analyze trends in practice patterns, we first used multilevel models to calculate the proportions of inpatients cared for by hospitalists each year for each of the 1172 PCPs with at least 20 patients. Then we employed an SAS procedure (PROC TRAJ) developed by Jones et al. to classify these PCPs into groups based on their trajectories.[13] This group‐based trajectory modeling allowed us to identify relatively homogeneous clusters within a heterogeneous sample population.[14] We chose a model that classified the PCPs into 4 groups.[15] With 4 groups, the average of the posterior probabilities of group membership for the PCPs assigned to each group exceeded 0.93, indicating a low rate of misclassification among these 4 distinct groups. For the 1172 PCPs, we tested interactions between year of hospitalization and PCP characteristics while adjusting for patient characteristics in order to investigate whether or not the impacts of PCP characteristics on how likely their patients being cared for by hospitalists differed with time. All analyses were performed with SAS version 9.2 (SAS Institute Inc., Cary, NC).
RESULTS
During the 2001 through 2009 study period, between 2252 and 2848 PCPs were associated with at least 20 hospitalized beneficiaries in any single year. Among these, 1172 PCPs were associated with at least 20 hospitalized beneficiaries in every year of the study period. These 1172 PCPs were associated with 608,686 hospitalizations over the 9 years.
Table 1 presents the characteristics of the PCPs who contributed to the cross‐sectional analyses in 2001 (N=2252) and 2009 (N=2387), as well as the 1172 PCPs for whom we had data for all 9 years for the longitudinal analyses. Most PCPs were male, trained in the United States, and were board certified. The average number of Medicare patients seen by these PCPs and number of outpatient Medicare visits went up about 7% between 2001 and 2009.
| PCP Characteristics | Cross‐Sectional Analysis | Trajectory Analysis, 20012009 | |
|---|---|---|---|
| 2001 | 2009 | ||
| |||
| Overall, no. (%) | 2,252 (100%) | 2,387 (100%) | 1,172 (100%) |
| Specialty, no. (%) | |||
| General practice | 39 (1.7%) | 34 (1.4%) | 15 (1.3%) |
| Family practice | 948 (42.1%) | 1,089 (45.6%) | 466 (39.8%) |
| Internal medicine | 1,255 (55.7%) | 1,249 (52.3%) | 688 (58.7%) |
| Geriatrics | 10 (0.4%) | 15 (0.6%) | 3 (0.3%) |
| Gender, no. (%) | |||
| Male | 1,990 (88.4%) | 2,015 (84.4%) | 1,072 (91.5%) |
| Female | 262 (11.6%) | 372 (15.6%) | 100 (8.5%) |
| Trained in the United States, no. (%) | |||
| Yes | 1,669 (74.1%) | 1,738 (72.8%) | 844 (72.0%) |
| No | 583 (25.9%) | 649 (27.2%) | 328 (28.0%) |
| Metropolitan statistical area, no. (%) | |||
| 99,999 or less | 417 (17.5) | 237 (20.2) | |
| 100,000249,000 | 438 (18.3) | 234 (20.0) | |
| 250,000999,999 | 381 (16.0) | 216 (18.4) | |
| 1,000,000 or more | 1,151 (48.2) | 485 (41.4) | |
| Board certification, no. (%) | |||
| Yes | 1,657 (69.4%) | 800 (68.3%) | |
| No | 730 (30.6%) | 372 (31.7%) | |
| Years in practice, 2001, meanSD (Q1Q3) | 22.310.6 (15.028.0) | 21.28.9 (15.027.0) | |
| Years in practice, 2009, meanSD (Q1Q3) | 25.010.2 (17.032.0) | 29.28.9 (23.035.0) | |
| Total no. of Medicare outpatient visits, 2001, meanSD (Q1Q3) | 1,624.8879.2 (1,057.51,970.0) | 1,883.39,48.5 (1,236.52,240.5) | |
| Total no. of Medicare outpatient visits, 2009, meanSD (Q1Q3) | 1,733.81,053.3 (1,080.02,048.0) | 2,020.51,200.9 (1,334.52,373.0) | |
| Total no. of Medicare outpatients, 2001, meanSD (Q1Q3) | 418.6186.9 (284.0522.0) | 473.4189.5 (338.0580.5) | |
| Total no. of Medicare outpatients, 2009, meanSD (Q1Q3) | 448.7217.8 (300.0548.0) | 508.7238.2 (350.5615.0) | |
| No. of hospitalized patients, 2001, meanSD (Q1Q3) | 46.025.0 (27.057.0) | 53.028.0 (32.066.0) | |
| No. of hospitalized patients, 2009, meanSD (Q1Q3) | 44.024.0 (26.052.0) | 52.027.0 (33.065.0) | |
| Average outpatient age, 2001, meanSD (Q1Q3) | 72.82.3 (71.574.2) | 72.82.1 (71.774.1) | |
| Average outpatient age, 2009, meanSD (Q1Q3) | 72.12.8 (70.673.9) | 72.82.7 (71.474.5) | |
| Average outpatient gender (% male), 2001, meanSD (Q1Q3) | 38.17.0 (35.542.3) | 38.56.4 (36.242.3) | |
| Average outpatient gender (% male), 2009, meanSD (Q1Q3) | 40.27.6 (37.644.8) | 41.06.5 (38.644.8) | |
| Average outpatient race (% white), 2001, meanSD (Q1Q3) | 84.316.4 (79.295.5) | 85.414.3 (79.995.7) | |
| Average outpatient race (% white), 2009, meanSD (Q1Q3) | 85.214.4 (79.895.2) | 86.312.9 (80.895.6) | |
| Average outpatient comorbidity, 2001, meanSD (Q1Q3)a | 1.60.5 (1.21.8) | 1.60.4 (1.21.8) | |
| Average outpatient comorbidity, 2009, meanSD (Q1Q3)a | 2.20.6 (1.82.5) | 2.20.6 (1.72.5) | |
Figure 1 graphs the percentage of PCPs as a function of what percent of their hospitalized patients received care from hospitalists, and how that changed from 2001 to 2009. For 70.9% of PCPs, fewer than 5% of their hospitalized patients received hospitalist care in 2001. By 2009, the percent of PCPs in this category had decreased to 15.2%. In contrast, in 2001, more than half of the patients for 2.1% of PCPs received hospitalist care, and the percent of PCPs in this category increased to 26.3% by 2009.

The pattern in Figure 1 shows that PCPs' use of hospitalists changed continuously and gradually over time. However, this pattern describes the PCPs as a group. When examined at the individual PCP level, different patterns emerge. Figure 2, which presents selected individual PCP's use of hospitalists over time, shows several distinct subpatterns of PCP practice behaviors. First, there are PCPs whose use of hospitalists was high in 2001 and stayed high or increased over time (eg, PCP A). There also were PCPs whose use of hospitalists stayed low over the entire study period (eg, PCP B). Finally, there were PCPs whose use of hospitalists was low in 2001 but high in 2009 (eg, PCP C). For this last group, the pattern of change in hospitalist utilization over time was discontinuous; that is, most of the increase occurred over a 1‐ or 2‐year period, instead of increasing gradually over time.

Among the 1172 PCPs associated with 20 hospitalized beneficiaries each year in all 9 years of the study period, group‐based trajectory modeling classified their practice patterns into 4 distinct trajectories (Figure 3). Among PCPs in group 1, more than one‐third of their hospitalized patients were cared for by hospitalists in 2001, and this increased to 60% by 2009. PCPs in groups 2 and 3 rarely used hospitalist care in 2001 but increased their use over time. The increase started early in the period for PCPs in group 2 and later for those in group 3. PCPs in group 4 were associated with little hospitalist use throughout the study period.

We constructed a model to describe the odds of a patient receiving care from hospitalists during the study period using patients associated with these 1172 PCPs. After adjusting for patient characteristics, the residual intraclass correlation coefficient for PCP level was 0.334, which indicates that 33.4% of the variance in whether a hospitalized patient received care from a hospitalist is explained by which PCP the patient saw. When adjusting for both patient and PCP characteristics, the overall odds of a patient receiving hospitalist care increased by 30% (95% confidence interval [CI]: 1.29‐1.30) per year from 2001 through 2009.
There were also significant interactions between year of hospitalization and several PCP characteristics. These interactions are illustrated in Table 2, which stratifies each of those PCP characteristics by 3 time periods: 2001 to 2003, 2004 to 2006, and 2007 to 2009. In all time periods, patients were more likely to receive hospitalist care if their PCP was US trained (US vs international medical graduate: odds ratio [OR]: 1.42, 95% CI: 1.19‐1.69 in 20012003; OR: 1.46, 95% CI: 1.23‐1.73 in 20072009), or specialized in family medicine (family medicine vs internal medicine: OR: 1.46, 95% CI: 1.25‐1.72 in 20012003; OR: 1.46, 95% CI: 1.25‐1.70 in 20072009). Over time, the relative odds of a patient receiving care from hospitalists decreased if their PCP was female (female vs male: OR: 1.91, 95% CI: 1.46‐2.50 in 20012003 vs OR: 1.50, 95% CI: 1.15‐1.95 in 20072009) or practiced in an urban area (largest vs smallest MSA: OR: 3.34, 95% CI: 2.72‐4.09 in 20012003; OR: 2.22, 95% CI: 1.82‐2.71 in 20072009). Although the longest‐practicing PCPs were most likely to use hospitalists in the early 2000s, this effect disappeared by 2007 to 2009 (most vs least years in practice: OR: 1.35, 95% CI: 1.06‐1.72 in 20012003 vs OR: 0.92, 95% CI: 0.73‐1.17 in 20072009).
| PCP Characteristics | 20012003, OR (95% CI) | 20042006, OR (95% CI) | 20072009, OR (95% CI) |
|---|---|---|---|
| |||
| Family practicea vs. internal medicineb | 1.46 (1.251.72) | 1.50 (1.281.76) | 1.46 (1.251.70) |
| Female vs male | 1.91 (1.462.50) | 1.43 (1.091.86) | 1.50 (1.151.95) |
| United States trained (yes vs no) | 1.42 (1.191.69) | 1.53 (1.281.81) | 1.46 (1.231.73) |
| Metropolitan statistical area | |||
| 99,999 or less | 1.00 | 1.00 | 1.00 |
| 100,000249,000 | 0.83 (0.651.05) | 1.00 (0.791.25) | 1.13 (0.901.41) |
| 250,000999,999 | 0.92 (0.721.17) | 1.03 (0.821.31) | 0.98 (0.771.23) |
| 1,000,000 or more | 3.34 (2.724.09) | 2.90 (2.373.54) | 2.22 (1.822.71) |
| Years in practice, 2001 | |||
| Q1 (lowest) | 1.00 | 1.00 | 1.00 |
| Q2 | 0.89 (0.711.12) | 0.83 (0.671.04) | 0.92 (0.741.14) |
| Q3 | 1.06 (0.841.34) | 0.99 (0.791.24) | 1.03 (0.821.29) |
| Q4 | 1.25 (0.991.59) | 1.13 (0.891.42) | 1.15 (0.921.45) |
| Q5 (highest) | 1.35 (1.061.72) | 1.05 (0.831.33) | 0.92 (0.731.17) |
| Total no. of outpatient visitsc | |||
| Q1 (lowest) | 1.00 | 1.00 | 1.00 |
| Q2 | 1.21 (1.121.30) | 1.07 (1.001.14) | 1.13 (1.071.19) |
| Q3 | 1.42 (1.301.54) | 1.18 (1.091.27) | 1.14 (1.071.22) |
| Q4 | 1.34 (1.211.47) | 1.34 (1.231.46) | 1.25 (1.161.35) |
| Q5 (highest) | 1.46 (1.301.63) | 1.33 (1.211.47) | 1.32 (1.201.44) |
| No. of hospitalized patientsc | |||
| Q1 (lowest) | 1.00 | 1.00 | 1.00 |
| Q2 | 1.07 (1.001.15) | 0.91 (0.860.96) | 0.85 (0.810.89) |
| Q3 | 1.00 (0.921.08) | 0.87 (0.820.93) | 0.74 (0.700.79) |
| Q4 | 0.89 (0.810.97) | 0.76 (0.710.82) | 0.62 (0.580.67) |
| Q5 (highest) | 1.05 (0.951.18) | 0.67 (0.610.73) | 0.55 (0.510.60) |
| Average outpatient agec | |||
| Q1 (lowest) | 1.00 | 1.00 | 1.00 |
| Q2 | 0.94 (0.871.01) | 1.15 (1.081.23) | 1.18 (1.111.25) |
| Q3 | 0.82 (0.760.90) | 1.05 (0.971.13) | 1.17 (1.091.25) |
| Q4 | 0.71 (0.650.79) | 1.03 (0.951.12) | 1.10 (1.021.19) |
| Q5 (highest) | 0.72 (0.640.81) | 1.12 (1.011.23) | 1.15 (1.051.26) |
| Average outpatient gender (% male)c | |||
| Q1 (lowest) | 1.00 | 1.00 | 1.00 |
| Q2 | 1.10 (1.021.18) | 1.19 (1.101.27) | 1.27 (1.181.37) |
| Q3 | 1.12 (1.031.22) | 1.27 (1.171.37) | 1.43 (1.321.54) |
| Q4 | 1.36 (1.251.48) | 1.49 (1.371.61) | 1.52 (1.401.65) |
| Q5 (highest) | 1.47 (1.341.61) | 1.84 (1.682.00) | 1.68 (1.541.83) |
| Average outpatient race (% white)c | |||
| Q1 (lowest) | 1.00 | 1.00 | 1.00 |
| Q2 | 1.08 (0.981.20) | 1.01 (0.921.10) | 1.23 (1.131.34) |
| Q3 | 1.27 (1.131.43) | 1.06 (0.951.18) | 1.21 (1.091.34) |
| Q4 | 1.47 (1.291.67) | 0.97 (0.861.09) | 1.33 (1.181.48) |
| Q5 (highest) | 1.39 (1.211.59) | 1.18 (1.041.34) | 1.25 (1.101.42) |
| Average outpatient comorbidityc | |||
| Q1 (lowest) | 1.00 | 1.00 | 1.00 |
| Q2 | 1.26 (1.191.35) | 1.23 (1.161.31) | 1.22 (1.141.30) |
| Q3 | 1.62 (1.491.75) | 1.61 (1.501.72) | 1.43 (1.341.54) |
| Q4 | 1.96 (1.792.15) | 1.86 (1.722.02) | 1.59 (1.471.72) |
| Q5 (highest) | 1.79 (1.592.01) | 2.20 (2.002.41) | 2.03 (1.852.22) |
In terms of PCP workload, patients of PCPs with high outpatient activity were more likely to receive hospitalists care throughout the study period, although the association had decreased by 2007 to 2009 (highest vs lowest outpatient volume: OR: 1.46, 95% CI: 1.30‐1.63 in 20012003 vs OR: 1.32, 95% CI: 1.20‐1.44 in 20072009). In contrast, PCPs with the lowest inpatient volumes became more likely to use hospitalists by the end of the study period (highest vs lowest inpatient volume: OR: 1.05, 95% CI: 0.95‐1.18 in 20012003 vs OR: 0.55, 95% CI: 0.51‐0.60 in 20072009).
The characteristics of PCPs' practice panels also were associated with patients' likelihood of receiving care from hospitalists. PCPs whose practice panels consisted of patients who were predominantly male, white, or with more outpatient comorbidities were consistently more likely to use hospitalists throughout the study period. PCPs with older patient panels were less likely to use hospitalists in 2001 to 2003, but by 2007 to 2009, they were slightly more likely to do so (oldest vs youngest average outpatient panel age: OR: 0.72, 95% CI: 0.64‐0.81 in 20012003 vs OR: 1.15, 95% CI: 1.05‐1.26 in 20072009).
CONCLUSIONS
Prior studies of the hospitalist model have shown that the likelihood of a patient receiving inpatient care from hospitalists is associated with patient characteristics, hospital characteristics, geographic region, and type of admission.[1, 16, 17] We found that PCP characteristics also predict whether patients receive care from hospitalists and that their use of hospitalists developed dynamically between 2001 to 2009. Although many factors (such as whether patients were admitted to a hospital where their PCP had admitting privileges) can influence the decision to use hospitalists, we found that over one‐third of the variance in whether a hospitalized patient received care from a hospitalist is explained by which PCP the patient saw. In showing that systemic differences exist among PCPs who use hospitalists and those who do not, our study suggests that future research on the hospitalist model should, if possible, adjust for PCP characteristics in addition to hospital and patient factors.
Although this study identifies the existence and magnitude of differences in whether or not PCPs use hospitalists, it cannot explain why the differences exist. We only can offer hypotheses. For example, our finding that PCPs with the most years of practice experience were more likely to use hospitalists in the early 2000s but not in more recent years suggests that in hospital medicine's early years, long‐practicing generalist physicians were choosing between practicing traditionalist medicine and adopting the hospitalists model, but by 2009, experienced generalist physicians had already specialized to either inpatient or outpatient settings earlier in their careers. On the other hand, the decreasing odds of urban PCPs using hospitalists may reflect a relative growth in hospitalist use in less populated areas rather than a change in urban PCPs' practice patterns.
PCPs trained in family medicine have reported less inpatient training and less comfort with providing hospital care,[18, 19] thus it is unsurprising that family physicians were more likely to refer patients to hospitalists. Although a recent study reported that family physicians' inpatient volumes remained constant, whereas those of outpatient internists declined between 2003 and 2012, the analysis used University Health Consortium data and thus reflects practice patterns in academic medical centers.[20] Our data suggest that outside of academia, family physicians have embraced the hospitalists as clinical partners.
Meltzer and Chung had previously proposed an economic model to describe the growing use of hospitalists in the United States. They posited that decisions to adopt the hospitalist model are governed by trade‐offs between coordination costs (eg, time and effort spent coordinating multiple providers across different settings) and switching costs (eg, time spent traveling between the office and the hospital or the effort of adjusting to different work settings).[16] The authors hypothesized that empirical testing of this model would show PCPs are more likely to use hospitalists if they have less available professional time (ie, work fewer hours per week), are female (due to competing demands from domestic responsibilities), have relatively few hospitalized patients, or live in areas with high traffic congestion. Our findings provide empirical evidence to support their division‐of‐labor model in showing that patients were more likely to receive hospitalist care if their PCP was female, practiced in an urban location, had higher outpatient practice volumes, or had lower inpatient volumes.
At first glance, some of our findings appear to contradict our earlier study, which showed that younger, black, male patients are more likely to receive inpatient care from hospitalists.[1] However, that study included patients regardless of whether they had a PCP. This study shows that when patients have a PCP, their PCPs are more likely to refer them to hospitalists if they are older, white, male, and have more comorbid conditions. A potential explanation for this finding is that PCPs may preferentially use hospitalists when caring for older and sicker hospitalized patients. For example, commentators often cite hospitalists' constant availability in the hospital as a valuable resource when caring for acutely ill patients.[21, 22]
Another potential explanation is that despite their preferences, PCPs who care for younger, minority patients lack access to hospitalist services. One large study of Medicare beneficiaries reported that physicians who care for black patients are less well‐trained clinically and often lack access to important clinical resources such as diagnostic imaging and nonemergency hospital admissions.[23] Similarly, international medical graduates are more likely than their US‐trained counterparts to care for underserved patients and to practice in small, independent offices.[24, 25, 26] As hospitalist groups often rely on cross‐subsidization from sources within a large healthcare organization, independent PCPs may have less access to their services when compared with PCPs in managed care organizations or large integrated groups. Viewed in this context, our findings imply that although hospitalists often care for socioeconomically vulnerable patients (eg, younger, uninsured, black men) who lack access to primary care services,[1] they also appear to share care responsibilities for more complex hospitalized patients with PCPs in more affluent communities. Further research may determine if the availability of hospitalists influences racial disparities in hospital care.
Our study has limitations. It is an observational study and thus subject to bias and confounding. As our cohort was formed using fee‐for‐service Medicare data in a single, large state, it may not be generalizable to PCPs who practice in other states, who care for a younger population, or who do not accept Medicare. Our findings also may not reflect the practice patterns of physicians‐in‐training, PCP populations with high board‐certification rates, those employed in temporary positions, or those who interrupt their practices for personal reasons, as we restricted our study to established PCPs who had been in practice long and consistently enough to be associated with 20 hospitalized patients during every year of the study. For example, the lower proportion of female PCPs in our cohort (15.6% in our study in 2009 vs 27.5% reported in a nationally representative 2008 survey[27]) may be explained by our exclusion of women who take prolonged time off for childcare duties. We also did not establish whether patient outcomes or healthcare costs differ between PCPs who adopted the hospitalist model and traditionalists. Finally, we could not examine the effect of a number of PCP factors that could plausibly influence whether or not PCPs relinquish inpatient care to hospitalists, such as their comfort with providing inpatient care, having hospital admitting privileges, having office‐based access to hospitals' electronic medical records, or the distance between their office and the hospital. However, this study lays the groundwork for future studies to explore these factors.
In summary, this study is the first, to our knowledge, to characterize PCPs who relinquished inpatient responsibilities to hospitalists. Our findings suggest that some groups of PCPs are more likely to refer patient to hospitalists, that the relationship between hospitalists and PCPs has evolved over time, and that the hospitalist model still has ample room to grow.
ACKNOWLEDGMENTS
Disclosures: This study was supported by grants from the National Institute on Aging (1RO1‐AG033134 and P30‐AG024832) and the National Cancer Institute (K05‐CA124923). The authors have no financial conflicts of interest to disclose. An oral abstract of this article was presented on May 18, 2013 at the Society of Hospital Medicine Annual Meeting in National Harbor, Maryland.
Although primary care physicians (PCPs) have traditionally treated patients in both ambulatory and hospital settings, many relinquished inpatient duties to hospitalists in recent decades.[1] Little is known about the PCPs who relinquished inpatient care duties or how the transition to the hospitalist model occurred. For example, what are the characteristics of PCPs who change? Do PCPs adopt the hospitalist model enthusiastically or cautiously? Characterizing PCPs who adopted the hospitalist model can help hospitalists understand their specialty's history and also inform health services research.
Much of the interest in the hospitalist model has been generated by studies reporting improved outcomes and lower hospital lengths of stay associated with hospitalist care.[2, 3, 4, 5] Conversely, detractors of the model point to reports of higher postacute care utilization among hospitalist patients.[6] Although these studies usually adjusted for differences among patients and hospitals, they did not account for PCP characteristics. As patients' access to PCPs and their PCP's capabilities are both plausible factors that could influence hospital length of stay (eg, decisions to complete more or less of a workup in the hospital), quality of care transitions, and postdischarge utilization, it is important to determine if PCPs who use hospitalists differ systematically from those who do not to correctly interpret health system utilization patterns that currently are attributed only to hospitalists.[7, 8]
We conducted this study to determine if observable PCP factors are associated with patients' use of hospitalists and to describe the trajectory by which PCPs referred their patients to hospitalists over time.
METHODS
Source of Data
We used claims data from 100% of Texas Medicare beneficiaries from 2000 to 2009, including Medicare beneficiary summary files, Medicare Provider Analysis and Review (MedPAR) files, Outpatient Standard Analytical Files (OutSAF), and Medicare Carrier files. Diagnosis related group (DRG)‐associated information, including weights, and Major Diagnostic Categories, were obtained from Centers for Medicare & Medicaid Services (
Establishment of the Study Cohort
Using the MedPAR file, we first selected hospital admissions from acute care hospitals in Texas for each year of the study period. We excluded beneficiaries younger than 66 years old, with incomplete Medicare Parts A and B enrollment, or with any health maintenance organization enrollment in the 12 months prior to the admission of interest. For patients with more than 1 admission in a given year, we randomly selected 1 admission. We then attempted to assign each patient to a PCP. We defined a PCP as a generalist (general practitioner, family physician, internist, or geriatrician) who saw a given beneficiary on 3 or more occasions in an outpatient setting in the year prior to the admission of interest.[9] We identified outpatient visits using Current Procedural Terminology (CPT) codes 99201 to 99205 (new patient encounters), and 99211 to 99215 (established patient encounters) from Carrier files. If more than 1 generalist physician saw the beneficiary on 3 or more occasions in a given year, the physician with more than 75% of the total outpatient evaluation and management (E&M) billings was classified as the beneficiary's PCP. Using these criteria, approximately 66% of patients were assigned to a PCP.
For cross‐sectional analyses, we restricted our cohort to beneficiaries whose PCPs were associated with at least 20 inpatients in a given year. To study trends in PCP practice patterns over time, we further restricted the cohort to beneficiaries whose PCPs were associated with at least 20 inpatients in every year of the study period, resulting in 1172 PCPs for the trajectory analyses. The reliability of PCPs' practice profiles increases as the number of patients in their panel increases. We chose 20 inpatients as the minimum because PCPs with 20 hospitalized patients per study year would achieve a reliability of 0.9 for estimating the proportion of their patients that received care from hospitalists.[10]
Identification of Hospitalists
We defined hospitalists as generalists who had at least 100 E&M billings in a given year and generated at least 90% of their total E&M billings in the year from inpatient services.[1] Inpatient E&M billings were identified by CPT codes 99221 to 99223 (new or established patient encounters), 99231 to 99233 (subsequent hospital care), and 99251 to 99255 (inpatient consultations).[1]
Patient Measures
Patient demographic information including, age at admission, gender, race/ethnicity, and Medicaid eligibility were obtained from Medicare beneficiary summary files. We used the Medicaid indicator as a proxy for low socioeconomic status. Information on weekday versus weekend admission, emergent admission, and DRG were obtained from MedPAR files. The DRG category (circulatory system, digestive system, infectious disease, nervous system, respiratory system, or other) was determined based on its Major Diagnostic Category. We determined residence in a nursing facility in the 3 months before the admission of interest from the MedPAR files and by E&M codes 99304 to 99318 (nursing facility services) from Carrier files.[11] Comorbidities were identified using the claims from MedPAR, Carrier, and OutSAF files in the year prior to the admission of interest.[12] Total hospitalizations and outpatient visits in the prior year were identified from MedPAR files and Carrier files, respectively.
PCP Measures
We categorized PCPs by specialty (general practice, gamily practice, geriatric medicine, or internal medicine), years in practice, gender, US‐ versus foreign‐trained, metropolitan statistical area (MSA) of their practice location, and board certification status. The specialty was identified from Carrier files and the other information from AMA data. For each PCP, the total number of outpatient visits and total number of patients seen as outpatients in each year was calculated based on E&M codes (9920199205, 9921199215) from Carrier files. For each year, we computed the average outpatient age, gender, race, and outpatient comorbidity for each PCP's patient panel. We computed hospital volumes using the number of hospitalized patients associated with each PCP in the study cohort.
Study Outcome
To determine whether hospitalized patients received care from hospitalists during a given hospitalization, we identified all inpatient E&M bills from generalist physicians during the admission of interest by linking MedPAR and Carrier files. If more than 50% of the generalist inpatient E&M billings from generalist physicians were from 1 or more hospitalists, the patient was considered to have received care from hospitalists.
Statistical Analyses
Multilevel analyses were used to account for the clustering of patients within PCPs. All multilevel models were adjusted for patient characteristics including age, race/ethnicity, gender, Medicaid eligibility, emergency admission, weekend admission, DRG weight, DRG category, any nursing home stay in the prior 3 months, number of comorbidities, number of hospitalizations, and number of physician visits in the year prior to the admission of interest. To analyze trends in practice patterns, we first used multilevel models to calculate the proportions of inpatients cared for by hospitalists each year for each of the 1172 PCPs with at least 20 patients. Then we employed an SAS procedure (PROC TRAJ) developed by Jones et al. to classify these PCPs into groups based on their trajectories.[13] This group‐based trajectory modeling allowed us to identify relatively homogeneous clusters within a heterogeneous sample population.[14] We chose a model that classified the PCPs into 4 groups.[15] With 4 groups, the average of the posterior probabilities of group membership for the PCPs assigned to each group exceeded 0.93, indicating a low rate of misclassification among these 4 distinct groups. For the 1172 PCPs, we tested interactions between year of hospitalization and PCP characteristics while adjusting for patient characteristics in order to investigate whether or not the impacts of PCP characteristics on how likely their patients being cared for by hospitalists differed with time. All analyses were performed with SAS version 9.2 (SAS Institute Inc., Cary, NC).
RESULTS
During the 2001 through 2009 study period, between 2252 and 2848 PCPs were associated with at least 20 hospitalized beneficiaries in any single year. Among these, 1172 PCPs were associated with at least 20 hospitalized beneficiaries in every year of the study period. These 1172 PCPs were associated with 608,686 hospitalizations over the 9 years.
Table 1 presents the characteristics of the PCPs who contributed to the cross‐sectional analyses in 2001 (N=2252) and 2009 (N=2387), as well as the 1172 PCPs for whom we had data for all 9 years for the longitudinal analyses. Most PCPs were male, trained in the United States, and were board certified. The average number of Medicare patients seen by these PCPs and number of outpatient Medicare visits went up about 7% between 2001 and 2009.
| PCP Characteristics | Cross‐Sectional Analysis | Trajectory Analysis, 20012009 | |
|---|---|---|---|
| 2001 | 2009 | ||
| |||
| Overall, no. (%) | 2,252 (100%) | 2,387 (100%) | 1,172 (100%) |
| Specialty, no. (%) | |||
| General practice | 39 (1.7%) | 34 (1.4%) | 15 (1.3%) |
| Family practice | 948 (42.1%) | 1,089 (45.6%) | 466 (39.8%) |
| Internal medicine | 1,255 (55.7%) | 1,249 (52.3%) | 688 (58.7%) |
| Geriatrics | 10 (0.4%) | 15 (0.6%) | 3 (0.3%) |
| Gender, no. (%) | |||
| Male | 1,990 (88.4%) | 2,015 (84.4%) | 1,072 (91.5%) |
| Female | 262 (11.6%) | 372 (15.6%) | 100 (8.5%) |
| Trained in the United States, no. (%) | |||
| Yes | 1,669 (74.1%) | 1,738 (72.8%) | 844 (72.0%) |
| No | 583 (25.9%) | 649 (27.2%) | 328 (28.0%) |
| Metropolitan statistical area, no. (%) | |||
| 99,999 or less | 417 (17.5) | 237 (20.2) | |
| 100,000249,000 | 438 (18.3) | 234 (20.0) | |
| 250,000999,999 | 381 (16.0) | 216 (18.4) | |
| 1,000,000 or more | 1,151 (48.2) | 485 (41.4) | |
| Board certification, no. (%) | |||
| Yes | 1,657 (69.4%) | 800 (68.3%) | |
| No | 730 (30.6%) | 372 (31.7%) | |
| Years in practice, 2001, meanSD (Q1Q3) | 22.310.6 (15.028.0) | 21.28.9 (15.027.0) | |
| Years in practice, 2009, meanSD (Q1Q3) | 25.010.2 (17.032.0) | 29.28.9 (23.035.0) | |
| Total no. of Medicare outpatient visits, 2001, meanSD (Q1Q3) | 1,624.8879.2 (1,057.51,970.0) | 1,883.39,48.5 (1,236.52,240.5) | |
| Total no. of Medicare outpatient visits, 2009, meanSD (Q1Q3) | 1,733.81,053.3 (1,080.02,048.0) | 2,020.51,200.9 (1,334.52,373.0) | |
| Total no. of Medicare outpatients, 2001, meanSD (Q1Q3) | 418.6186.9 (284.0522.0) | 473.4189.5 (338.0580.5) | |
| Total no. of Medicare outpatients, 2009, meanSD (Q1Q3) | 448.7217.8 (300.0548.0) | 508.7238.2 (350.5615.0) | |
| No. of hospitalized patients, 2001, meanSD (Q1Q3) | 46.025.0 (27.057.0) | 53.028.0 (32.066.0) | |
| No. of hospitalized patients, 2009, meanSD (Q1Q3) | 44.024.0 (26.052.0) | 52.027.0 (33.065.0) | |
| Average outpatient age, 2001, meanSD (Q1Q3) | 72.82.3 (71.574.2) | 72.82.1 (71.774.1) | |
| Average outpatient age, 2009, meanSD (Q1Q3) | 72.12.8 (70.673.9) | 72.82.7 (71.474.5) | |
| Average outpatient gender (% male), 2001, meanSD (Q1Q3) | 38.17.0 (35.542.3) | 38.56.4 (36.242.3) | |
| Average outpatient gender (% male), 2009, meanSD (Q1Q3) | 40.27.6 (37.644.8) | 41.06.5 (38.644.8) | |
| Average outpatient race (% white), 2001, meanSD (Q1Q3) | 84.316.4 (79.295.5) | 85.414.3 (79.995.7) | |
| Average outpatient race (% white), 2009, meanSD (Q1Q3) | 85.214.4 (79.895.2) | 86.312.9 (80.895.6) | |
| Average outpatient comorbidity, 2001, meanSD (Q1Q3)a | 1.60.5 (1.21.8) | 1.60.4 (1.21.8) | |
| Average outpatient comorbidity, 2009, meanSD (Q1Q3)a | 2.20.6 (1.82.5) | 2.20.6 (1.72.5) | |
Figure 1 graphs the percentage of PCPs as a function of what percent of their hospitalized patients received care from hospitalists, and how that changed from 2001 to 2009. For 70.9% of PCPs, fewer than 5% of their hospitalized patients received hospitalist care in 2001. By 2009, the percent of PCPs in this category had decreased to 15.2%. In contrast, in 2001, more than half of the patients for 2.1% of PCPs received hospitalist care, and the percent of PCPs in this category increased to 26.3% by 2009.

The pattern in Figure 1 shows that PCPs' use of hospitalists changed continuously and gradually over time. However, this pattern describes the PCPs as a group. When examined at the individual PCP level, different patterns emerge. Figure 2, which presents selected individual PCP's use of hospitalists over time, shows several distinct subpatterns of PCP practice behaviors. First, there are PCPs whose use of hospitalists was high in 2001 and stayed high or increased over time (eg, PCP A). There also were PCPs whose use of hospitalists stayed low over the entire study period (eg, PCP B). Finally, there were PCPs whose use of hospitalists was low in 2001 but high in 2009 (eg, PCP C). For this last group, the pattern of change in hospitalist utilization over time was discontinuous; that is, most of the increase occurred over a 1‐ or 2‐year period, instead of increasing gradually over time.

Among the 1172 PCPs associated with 20 hospitalized beneficiaries each year in all 9 years of the study period, group‐based trajectory modeling classified their practice patterns into 4 distinct trajectories (Figure 3). Among PCPs in group 1, more than one‐third of their hospitalized patients were cared for by hospitalists in 2001, and this increased to 60% by 2009. PCPs in groups 2 and 3 rarely used hospitalist care in 2001 but increased their use over time. The increase started early in the period for PCPs in group 2 and later for those in group 3. PCPs in group 4 were associated with little hospitalist use throughout the study period.

We constructed a model to describe the odds of a patient receiving care from hospitalists during the study period using patients associated with these 1172 PCPs. After adjusting for patient characteristics, the residual intraclass correlation coefficient for PCP level was 0.334, which indicates that 33.4% of the variance in whether a hospitalized patient received care from a hospitalist is explained by which PCP the patient saw. When adjusting for both patient and PCP characteristics, the overall odds of a patient receiving hospitalist care increased by 30% (95% confidence interval [CI]: 1.29‐1.30) per year from 2001 through 2009.
There were also significant interactions between year of hospitalization and several PCP characteristics. These interactions are illustrated in Table 2, which stratifies each of those PCP characteristics by 3 time periods: 2001 to 2003, 2004 to 2006, and 2007 to 2009. In all time periods, patients were more likely to receive hospitalist care if their PCP was US trained (US vs international medical graduate: odds ratio [OR]: 1.42, 95% CI: 1.19‐1.69 in 20012003; OR: 1.46, 95% CI: 1.23‐1.73 in 20072009), or specialized in family medicine (family medicine vs internal medicine: OR: 1.46, 95% CI: 1.25‐1.72 in 20012003; OR: 1.46, 95% CI: 1.25‐1.70 in 20072009). Over time, the relative odds of a patient receiving care from hospitalists decreased if their PCP was female (female vs male: OR: 1.91, 95% CI: 1.46‐2.50 in 20012003 vs OR: 1.50, 95% CI: 1.15‐1.95 in 20072009) or practiced in an urban area (largest vs smallest MSA: OR: 3.34, 95% CI: 2.72‐4.09 in 20012003; OR: 2.22, 95% CI: 1.82‐2.71 in 20072009). Although the longest‐practicing PCPs were most likely to use hospitalists in the early 2000s, this effect disappeared by 2007 to 2009 (most vs least years in practice: OR: 1.35, 95% CI: 1.06‐1.72 in 20012003 vs OR: 0.92, 95% CI: 0.73‐1.17 in 20072009).
| PCP Characteristics | 20012003, OR (95% CI) | 20042006, OR (95% CI) | 20072009, OR (95% CI) |
|---|---|---|---|
| |||
| Family practicea vs. internal medicineb | 1.46 (1.251.72) | 1.50 (1.281.76) | 1.46 (1.251.70) |
| Female vs male | 1.91 (1.462.50) | 1.43 (1.091.86) | 1.50 (1.151.95) |
| United States trained (yes vs no) | 1.42 (1.191.69) | 1.53 (1.281.81) | 1.46 (1.231.73) |
| Metropolitan statistical area | |||
| 99,999 or less | 1.00 | 1.00 | 1.00 |
| 100,000249,000 | 0.83 (0.651.05) | 1.00 (0.791.25) | 1.13 (0.901.41) |
| 250,000999,999 | 0.92 (0.721.17) | 1.03 (0.821.31) | 0.98 (0.771.23) |
| 1,000,000 or more | 3.34 (2.724.09) | 2.90 (2.373.54) | 2.22 (1.822.71) |
| Years in practice, 2001 | |||
| Q1 (lowest) | 1.00 | 1.00 | 1.00 |
| Q2 | 0.89 (0.711.12) | 0.83 (0.671.04) | 0.92 (0.741.14) |
| Q3 | 1.06 (0.841.34) | 0.99 (0.791.24) | 1.03 (0.821.29) |
| Q4 | 1.25 (0.991.59) | 1.13 (0.891.42) | 1.15 (0.921.45) |
| Q5 (highest) | 1.35 (1.061.72) | 1.05 (0.831.33) | 0.92 (0.731.17) |
| Total no. of outpatient visitsc | |||
| Q1 (lowest) | 1.00 | 1.00 | 1.00 |
| Q2 | 1.21 (1.121.30) | 1.07 (1.001.14) | 1.13 (1.071.19) |
| Q3 | 1.42 (1.301.54) | 1.18 (1.091.27) | 1.14 (1.071.22) |
| Q4 | 1.34 (1.211.47) | 1.34 (1.231.46) | 1.25 (1.161.35) |
| Q5 (highest) | 1.46 (1.301.63) | 1.33 (1.211.47) | 1.32 (1.201.44) |
| No. of hospitalized patientsc | |||
| Q1 (lowest) | 1.00 | 1.00 | 1.00 |
| Q2 | 1.07 (1.001.15) | 0.91 (0.860.96) | 0.85 (0.810.89) |
| Q3 | 1.00 (0.921.08) | 0.87 (0.820.93) | 0.74 (0.700.79) |
| Q4 | 0.89 (0.810.97) | 0.76 (0.710.82) | 0.62 (0.580.67) |
| Q5 (highest) | 1.05 (0.951.18) | 0.67 (0.610.73) | 0.55 (0.510.60) |
| Average outpatient agec | |||
| Q1 (lowest) | 1.00 | 1.00 | 1.00 |
| Q2 | 0.94 (0.871.01) | 1.15 (1.081.23) | 1.18 (1.111.25) |
| Q3 | 0.82 (0.760.90) | 1.05 (0.971.13) | 1.17 (1.091.25) |
| Q4 | 0.71 (0.650.79) | 1.03 (0.951.12) | 1.10 (1.021.19) |
| Q5 (highest) | 0.72 (0.640.81) | 1.12 (1.011.23) | 1.15 (1.051.26) |
| Average outpatient gender (% male)c | |||
| Q1 (lowest) | 1.00 | 1.00 | 1.00 |
| Q2 | 1.10 (1.021.18) | 1.19 (1.101.27) | 1.27 (1.181.37) |
| Q3 | 1.12 (1.031.22) | 1.27 (1.171.37) | 1.43 (1.321.54) |
| Q4 | 1.36 (1.251.48) | 1.49 (1.371.61) | 1.52 (1.401.65) |
| Q5 (highest) | 1.47 (1.341.61) | 1.84 (1.682.00) | 1.68 (1.541.83) |
| Average outpatient race (% white)c | |||
| Q1 (lowest) | 1.00 | 1.00 | 1.00 |
| Q2 | 1.08 (0.981.20) | 1.01 (0.921.10) | 1.23 (1.131.34) |
| Q3 | 1.27 (1.131.43) | 1.06 (0.951.18) | 1.21 (1.091.34) |
| Q4 | 1.47 (1.291.67) | 0.97 (0.861.09) | 1.33 (1.181.48) |
| Q5 (highest) | 1.39 (1.211.59) | 1.18 (1.041.34) | 1.25 (1.101.42) |
| Average outpatient comorbidityc | |||
| Q1 (lowest) | 1.00 | 1.00 | 1.00 |
| Q2 | 1.26 (1.191.35) | 1.23 (1.161.31) | 1.22 (1.141.30) |
| Q3 | 1.62 (1.491.75) | 1.61 (1.501.72) | 1.43 (1.341.54) |
| Q4 | 1.96 (1.792.15) | 1.86 (1.722.02) | 1.59 (1.471.72) |
| Q5 (highest) | 1.79 (1.592.01) | 2.20 (2.002.41) | 2.03 (1.852.22) |
In terms of PCP workload, patients of PCPs with high outpatient activity were more likely to receive hospitalists care throughout the study period, although the association had decreased by 2007 to 2009 (highest vs lowest outpatient volume: OR: 1.46, 95% CI: 1.30‐1.63 in 20012003 vs OR: 1.32, 95% CI: 1.20‐1.44 in 20072009). In contrast, PCPs with the lowest inpatient volumes became more likely to use hospitalists by the end of the study period (highest vs lowest inpatient volume: OR: 1.05, 95% CI: 0.95‐1.18 in 20012003 vs OR: 0.55, 95% CI: 0.51‐0.60 in 20072009).
The characteristics of PCPs' practice panels also were associated with patients' likelihood of receiving care from hospitalists. PCPs whose practice panels consisted of patients who were predominantly male, white, or with more outpatient comorbidities were consistently more likely to use hospitalists throughout the study period. PCPs with older patient panels were less likely to use hospitalists in 2001 to 2003, but by 2007 to 2009, they were slightly more likely to do so (oldest vs youngest average outpatient panel age: OR: 0.72, 95% CI: 0.64‐0.81 in 20012003 vs OR: 1.15, 95% CI: 1.05‐1.26 in 20072009).
CONCLUSIONS
Prior studies of the hospitalist model have shown that the likelihood of a patient receiving inpatient care from hospitalists is associated with patient characteristics, hospital characteristics, geographic region, and type of admission.[1, 16, 17] We found that PCP characteristics also predict whether patients receive care from hospitalists and that their use of hospitalists developed dynamically between 2001 to 2009. Although many factors (such as whether patients were admitted to a hospital where their PCP had admitting privileges) can influence the decision to use hospitalists, we found that over one‐third of the variance in whether a hospitalized patient received care from a hospitalist is explained by which PCP the patient saw. In showing that systemic differences exist among PCPs who use hospitalists and those who do not, our study suggests that future research on the hospitalist model should, if possible, adjust for PCP characteristics in addition to hospital and patient factors.
Although this study identifies the existence and magnitude of differences in whether or not PCPs use hospitalists, it cannot explain why the differences exist. We only can offer hypotheses. For example, our finding that PCPs with the most years of practice experience were more likely to use hospitalists in the early 2000s but not in more recent years suggests that in hospital medicine's early years, long‐practicing generalist physicians were choosing between practicing traditionalist medicine and adopting the hospitalists model, but by 2009, experienced generalist physicians had already specialized to either inpatient or outpatient settings earlier in their careers. On the other hand, the decreasing odds of urban PCPs using hospitalists may reflect a relative growth in hospitalist use in less populated areas rather than a change in urban PCPs' practice patterns.
PCPs trained in family medicine have reported less inpatient training and less comfort with providing hospital care,[18, 19] thus it is unsurprising that family physicians were more likely to refer patients to hospitalists. Although a recent study reported that family physicians' inpatient volumes remained constant, whereas those of outpatient internists declined between 2003 and 2012, the analysis used University Health Consortium data and thus reflects practice patterns in academic medical centers.[20] Our data suggest that outside of academia, family physicians have embraced the hospitalists as clinical partners.
Meltzer and Chung had previously proposed an economic model to describe the growing use of hospitalists in the United States. They posited that decisions to adopt the hospitalist model are governed by trade‐offs between coordination costs (eg, time and effort spent coordinating multiple providers across different settings) and switching costs (eg, time spent traveling between the office and the hospital or the effort of adjusting to different work settings).[16] The authors hypothesized that empirical testing of this model would show PCPs are more likely to use hospitalists if they have less available professional time (ie, work fewer hours per week), are female (due to competing demands from domestic responsibilities), have relatively few hospitalized patients, or live in areas with high traffic congestion. Our findings provide empirical evidence to support their division‐of‐labor model in showing that patients were more likely to receive hospitalist care if their PCP was female, practiced in an urban location, had higher outpatient practice volumes, or had lower inpatient volumes.
At first glance, some of our findings appear to contradict our earlier study, which showed that younger, black, male patients are more likely to receive inpatient care from hospitalists.[1] However, that study included patients regardless of whether they had a PCP. This study shows that when patients have a PCP, their PCPs are more likely to refer them to hospitalists if they are older, white, male, and have more comorbid conditions. A potential explanation for this finding is that PCPs may preferentially use hospitalists when caring for older and sicker hospitalized patients. For example, commentators often cite hospitalists' constant availability in the hospital as a valuable resource when caring for acutely ill patients.[21, 22]
Another potential explanation is that despite their preferences, PCPs who care for younger, minority patients lack access to hospitalist services. One large study of Medicare beneficiaries reported that physicians who care for black patients are less well‐trained clinically and often lack access to important clinical resources such as diagnostic imaging and nonemergency hospital admissions.[23] Similarly, international medical graduates are more likely than their US‐trained counterparts to care for underserved patients and to practice in small, independent offices.[24, 25, 26] As hospitalist groups often rely on cross‐subsidization from sources within a large healthcare organization, independent PCPs may have less access to their services when compared with PCPs in managed care organizations or large integrated groups. Viewed in this context, our findings imply that although hospitalists often care for socioeconomically vulnerable patients (eg, younger, uninsured, black men) who lack access to primary care services,[1] they also appear to share care responsibilities for more complex hospitalized patients with PCPs in more affluent communities. Further research may determine if the availability of hospitalists influences racial disparities in hospital care.
Our study has limitations. It is an observational study and thus subject to bias and confounding. As our cohort was formed using fee‐for‐service Medicare data in a single, large state, it may not be generalizable to PCPs who practice in other states, who care for a younger population, or who do not accept Medicare. Our findings also may not reflect the practice patterns of physicians‐in‐training, PCP populations with high board‐certification rates, those employed in temporary positions, or those who interrupt their practices for personal reasons, as we restricted our study to established PCPs who had been in practice long and consistently enough to be associated with 20 hospitalized patients during every year of the study. For example, the lower proportion of female PCPs in our cohort (15.6% in our study in 2009 vs 27.5% reported in a nationally representative 2008 survey[27]) may be explained by our exclusion of women who take prolonged time off for childcare duties. We also did not establish whether patient outcomes or healthcare costs differ between PCPs who adopted the hospitalist model and traditionalists. Finally, we could not examine the effect of a number of PCP factors that could plausibly influence whether or not PCPs relinquish inpatient care to hospitalists, such as their comfort with providing inpatient care, having hospital admitting privileges, having office‐based access to hospitals' electronic medical records, or the distance between their office and the hospital. However, this study lays the groundwork for future studies to explore these factors.
In summary, this study is the first, to our knowledge, to characterize PCPs who relinquished inpatient responsibilities to hospitalists. Our findings suggest that some groups of PCPs are more likely to refer patient to hospitalists, that the relationship between hospitalists and PCPs has evolved over time, and that the hospitalist model still has ample room to grow.
ACKNOWLEDGMENTS
Disclosures: This study was supported by grants from the National Institute on Aging (1RO1‐AG033134 and P30‐AG024832) and the National Cancer Institute (K05‐CA124923). The authors have no financial conflicts of interest to disclose. An oral abstract of this article was presented on May 18, 2013 at the Society of Hospital Medicine Annual Meeting in National Harbor, Maryland.
- , , , . Growth in the care of older patients by hospitalists in the United States. N Engl J Med. 2009;360(11):1102–1112.
- , . Effect of hospitalists on length of stay in the medicare population: variation according to hospital and patient characteristics. J Am Geriatr Soc. 2010;58(9):1649–1657.
- , , , , , . Outcomes of care by hospitalists, general internists, and family physicians. N Engl J Med. 2007;357(25):2589–2600.
- , , , , . Hospitalist care and length of stay in patients requiring complex discharge planning and close clinical monitoring. Arch Intern Med. 2007;167(17):1869–1874.
- , . The impact of hospitalists on the cost and quality of inpatient care in the United States: a research synthesis. Med Care Res Rev. 2005;62(4):379–406.
- , . Association of hospitalist care with medical utilization after discharge: evidence of cost shift from a cohort study. Ann Intern Med. 2011;155(3):152–159.
- , . Hospital care and medical utilization after discharge. Ann Intern Med. 2011;155(10):719–720; author reply 722.
- . Hospital care and medical utilization after discharge. Ann Intern Med. 2011;155(10):721; author reply 722.
- , , , , , . Administrative data algorithms can describe ambulatory physician utilization. Health Serv Res. 2007;42:1783–1796.
- , . Estimating the reliability of continuous measures with Cronbach's alpha or the intraclass correlation coefficient: toward the integration of two traditions. J Clin Epidemiol. 1991;44(4–5):381–390.
- , , . Ability of Medicare claims data to identify nursing home patients: a validation study. Med Care. 2008;46(11):1184–1187.
- , , , . Comorbidity measures for use with administrative data. Med Care. 1998;36(1):8–27.
- , , . A SAS procedure based on mixture models for estimating developmental trajectories. Sociol Methods Res. 2001;29(3):374–393.
- . Group‐Based Modeling of Development. Cambridge, MA: Harvard University Press; 2005.
- , . Group‐based trajectory modeling in clinical research. Annu Rev Clin Psychol. 2010;6:109–138.
- , . Coordination, switching costs and the division of labor in general medicine: an economic explanation for the emergence of hospitalists in the United States. National Bureau of Economic Research Working Paper Series No. 16040. Cambridge, MA: National Bureau of Economic Research; 2010.
- , , , , , . Continuity of outpatient and inpatient care by primary care physicians for hospitalized older adults. JAMA. 2009;301(16):1671–1680.
- . Hospitalists and family physicians: understanding opportunities and risks. J Fam Pract. 2004;53(6):473–481.
- , , , , , . Preparedness of internal medicine and family practice residents for treating common conditions. JAMA. 2002;288(20):2609–2614.
- , , , . The status of adult inpatient care by family physicians at US academic medical centers and affiliated teaching hospitals 2003 to 2012: the impact of the hospitalist movement. Fam Med. 2014;46(2):94–99.
- . Hospitalists and the hospital medicine system of care are good for patient care. Arch Intern Med. 2008;168(12):1254–1256; discussion 1259–1260.
- . Hospitalists in the United States—mission accomplished or work in progress? N Engl J Med. 2004;350(19):1935–1936.
- , , , , . Primary care physicians who treat blacks and whites. N Engl J Med. 2004;351(6):575–584.
- , , , . International medical graduates and the primary care workforce for rural underserved areas. Health Aff (Millwood). 2003;22(2):255–262.
- , , . Medical migration and the physician workforce. International medical graduates and American medicine. JAMA. 1995;273(19):1521–1527.
- , , , , . International medical graduates in family medicine in the United States of America: an exploration of professional characteristics and attitudes. Hum Resour Health. 2006;4:17.
- , , . A snapshot of U.S. physicians: key findings from the 2008 Health Tracking Physician Survey. Data Bull (Cent Stud Health Syst Change). 2009(35):1–11.
- , , , . Growth in the care of older patients by hospitalists in the United States. N Engl J Med. 2009;360(11):1102–1112.
- , . Effect of hospitalists on length of stay in the medicare population: variation according to hospital and patient characteristics. J Am Geriatr Soc. 2010;58(9):1649–1657.
- , , , , , . Outcomes of care by hospitalists, general internists, and family physicians. N Engl J Med. 2007;357(25):2589–2600.
- , , , , . Hospitalist care and length of stay in patients requiring complex discharge planning and close clinical monitoring. Arch Intern Med. 2007;167(17):1869–1874.
- , . The impact of hospitalists on the cost and quality of inpatient care in the United States: a research synthesis. Med Care Res Rev. 2005;62(4):379–406.
- , . Association of hospitalist care with medical utilization after discharge: evidence of cost shift from a cohort study. Ann Intern Med. 2011;155(3):152–159.
- , . Hospital care and medical utilization after discharge. Ann Intern Med. 2011;155(10):719–720; author reply 722.
- . Hospital care and medical utilization after discharge. Ann Intern Med. 2011;155(10):721; author reply 722.
- , , , , , . Administrative data algorithms can describe ambulatory physician utilization. Health Serv Res. 2007;42:1783–1796.
- , . Estimating the reliability of continuous measures with Cronbach's alpha or the intraclass correlation coefficient: toward the integration of two traditions. J Clin Epidemiol. 1991;44(4–5):381–390.
- , , . Ability of Medicare claims data to identify nursing home patients: a validation study. Med Care. 2008;46(11):1184–1187.
- , , , . Comorbidity measures for use with administrative data. Med Care. 1998;36(1):8–27.
- , , . A SAS procedure based on mixture models for estimating developmental trajectories. Sociol Methods Res. 2001;29(3):374–393.
- . Group‐Based Modeling of Development. Cambridge, MA: Harvard University Press; 2005.
- , . Group‐based trajectory modeling in clinical research. Annu Rev Clin Psychol. 2010;6:109–138.
- , . Coordination, switching costs and the division of labor in general medicine: an economic explanation for the emergence of hospitalists in the United States. National Bureau of Economic Research Working Paper Series No. 16040. Cambridge, MA: National Bureau of Economic Research; 2010.
- , , , , , . Continuity of outpatient and inpatient care by primary care physicians for hospitalized older adults. JAMA. 2009;301(16):1671–1680.
- . Hospitalists and family physicians: understanding opportunities and risks. J Fam Pract. 2004;53(6):473–481.
- , , , , , . Preparedness of internal medicine and family practice residents for treating common conditions. JAMA. 2002;288(20):2609–2614.
- , , , . The status of adult inpatient care by family physicians at US academic medical centers and affiliated teaching hospitals 2003 to 2012: the impact of the hospitalist movement. Fam Med. 2014;46(2):94–99.
- . Hospitalists and the hospital medicine system of care are good for patient care. Arch Intern Med. 2008;168(12):1254–1256; discussion 1259–1260.
- . Hospitalists in the United States—mission accomplished or work in progress? N Engl J Med. 2004;350(19):1935–1936.
- , , , , . Primary care physicians who treat blacks and whites. N Engl J Med. 2004;351(6):575–584.
- , , , . International medical graduates and the primary care workforce for rural underserved areas. Health Aff (Millwood). 2003;22(2):255–262.
- , , . Medical migration and the physician workforce. International medical graduates and American medicine. JAMA. 1995;273(19):1521–1527.
- , , , , . International medical graduates in family medicine in the United States of America: an exploration of professional characteristics and attitudes. Hum Resour Health. 2006;4:17.
- , , . A snapshot of U.S. physicians: key findings from the 2008 Health Tracking Physician Survey. Data Bull (Cent Stud Health Syst Change). 2009(35):1–11.
© 2015 Society of Hospital Medicine
Maximizing Teaching on the Wards
An important role of the hospitalist educator is to teach residents and medical students how to diagnose and manage acute medical problems. However, clinical reasoning is complex and nuanced, and there are many challenges to teaching this important process. Medical inpatients are increasingly complex, older, and more seriously ill.[1] Documentation requirements and productivity obligations compete with teaching time. Hospitalists must adjust their teaching for learners from different professions and at various levels of training. In addition, hospitalists tend to be less experienced, and must balance the need to learn their roles as clinicians with developing their own skills as educators.[2]
Despite the challenges inherent to the setting, inpatient rotations provide tremendous teaching and learning opportunities. Patients with undifferentiated complaints or known diagnoses in need of management decisions are available to stimulate discussion. Hospitalist educators have the opportunity to assess residents' progress along the developmental milestones, which residency programs are now required to report for accreditation,[3] and provide role modeling for residents who are developing their own teaching skills.
To maximize these opportunities, attendings must engage trainees to practice clinical reasoning and identify their own knowledge gaps. Various strategies for facilitating the clinical reasoning discussion exist, but two frameworksthe One‐Minute Preceptor (OMP) and SNAPPShave been well studied, albeit mainly in the outpatient setting. Both models offer ways to maximize teaching and assess clinical reasoning, but they have different methods and strengths. This article provides a narrative review of the two frameworks and discusses how they can be applied to the inpatient teaching environment. Hospitalists can utilize these models or components of each framework to facilitate teaching on inpatient teams and enhance their roles as educators.
ONE‐MINUTE PRECEPTOR
The OMP was first described in 1992 by Neher and colleagues as an alternative to the traditional model of precepting.[4] It gives preceptors a method to facilitate learners presentation of their thought process and then for the preceptor to provide targeted teaching points.[4] The OMP helps diagnose both learner and patient, whereas the traditional model focuses on diagnosing the patient.[5] In the traditional model, the attending questions the learner to diagnose the patient, which does not often make clear the learner's thinking process. Thus, there may be a mismatch between the teaching points the preceptor makes and what the learner really needs to know.[5] There are several key benefits to the OMP compared to the traditional model; broadly, these relate to improved ability to assess the learner and provide targeted teaching,[4, 5, 6, 7] improved integration of feedback,[4, 8, 9, 10] learner preference,[11] and ease with which it is learned by faculty members.[4]
The OMP model consists of five steps outlined in Table 1. Step 1, getting a commitment, can involve any aspect of the casediagnosis, treatment, or follow‐upand learners should be challenged to make intellectual commitments just beyond their level of comfort.[12] Steps 1 and 2 bring to light the learner's individual learning needs,[11] then the preceptor follows up with personalized teaching. The OMP is efficient; no increase in time was needed to precept a case in an outpatient study.[9] In a separate outpatient study, the OMP led preceptors to be more likely to teach about disease‐specific points and differential diagnosis, as compared to generic items such as history taking and presentation skills with the traditional model.[5]
| A 5‐step framework in which the preceptor does the following: |
| 1. Get a commitment |
| 2. Probe for supporting evidence |
| 3. Provide general rules |
| 4. Reinforce what was done correctly |
| 5. Correct mistakes |
Faculty feel better prepared to assess learners and provide feedback with the OMP model.[6, 9] Aagaard and colleagues provided 116 mostly ambulatory preceptors with scripted, videotaped encounters of the OMP and traditional models. The OMP improved preceptors' confidence at rating students' presentation skills, clinical reasoning, and fund of knowledge. It was rated more efficient and effective, and preceptors were able to diagnose the patient with the same or improved accuracy compared to the traditional model.[6] In a pre‐post study assessing the efficacy of a faculty development workshop, students rated ambulatory teaching encounters incorporating the OMP model as having increased quantity and quality of feedback. Furthermore, faculty reported improved ability to evaluate students and were more likely to let students reach their own conclusions and create their own postencounter learning plans.[9]
The OMP is also well‐received by trainees. Teherani and colleagues analyzed medical students' responses to videotaped teaching encounters of the OMP and traditional models. Students gave higher mean ratings for all studied items (including feedback, involving the student in decision‐making, and overall effectiveness) to the OMP model, and preferred it over the traditional model.[11]
Several studies have evaluated the OMP for use by residents as teachers,[10, 13, 14] and it is one of the most common models taught to residents.[13] One study evaluated the impact of a one‐day workshop for 276 residents that included the five‐step microskills model (also known as the OMP).[10] Residents felt more prepared to teach, set expectations, and provide feedback.[10] The OMP model, despite brief training, is effective in improving residents' teaching effectiveness and confidence.[13]
The only study we found that exclusively evaluated the OMP in the inpatient setting was a randomized trial[8] involving 57 internal medicine residents. Interns and students rated OMP‐trained residents more highly in 4 of 5 behaviors. The behavior that showed no difference from the control group was teaching general rules.[8] However, there was no difference in ratings of overall teaching effectiveness between groups.[8]
Our review of the literature on the OMP shows it is a quickly learned, easily implemented framework for teaching clinical reasoning. It has been used across specialties and settings, provides a built‐in mechanism for feedback, and allows educators to assess trainees' reasoning while extracting the clinical information needed to work efficiently.
SNAPPS
SNAPPS was first described in 2003 by Wolpaw and colleagues. It is a six‐step learner‐centered model as outlined in Table 2.[15] Unlike the OMP, SNAPPS requires both trainee and teacher to learn the framework. In doing so, the responsibility for directing the teaching encounter is shifted toward the learner.[15] Consequently, this model may be best suited to advanced or motivated learners. Like the OMP, SNAPPS was originally described for the ambulatory environment. However, it has been studied in the inpatient setting as well.
| A 6‐step framework in which the learner does the following: |
| 1. Summarize briefly the history and findings |
| 2. Narrow the differential to 2 or 3 possibilities |
| 3. Analyze the differential by comparing/contrasting the possibilities |
| 4. Probe the preceptor by asking questions |
| 5. Plan management for the patient's medical issues |
| 6. Select a case‐related issue for self‐directed learning |
With SNAPPS, the teaching encounter is learner driven. The trainee presents the case and directs the discussion of differential diagnosis. The educator does not have an active role until the fourth step, where the learner asks questions or identifies areas of uncertainty. But even at this stage, the discussion is learner driven. Step 5, planning management, is collaborative, with trainees suggesting management plans with appropriate attending guidance. Depending on learner skill level or case difficulty, the preceptor may need to play more or less of an active role. The final step, picking a case‐related issue to examine, extends the learning beyond the initial encounter, and ensures that it is individualized and relevant. This step also encourages learner progression toward the Accreditation Council for Graduate Medical Education (ACGME) competency of practice‐based learning and improvement.[3]
A handful of studies have evaluated the SNAPPS model. A randomized comparison group trial found that SNAPPS‐trained students outperformed students trained to elicit feedback and students who received the usual and customary preparation.[16] Notably, SNAPPS students expressed more than twice as many differential diagnoses, justified their reasoning more than five times as often, and expressed more questions and uncertainties. The SNAPPS students' presentations were no longer than in the usual and customary group, and were just one minute longer than in the group trained to elicit feedback.[16] A follow‐up analysis found that 100% of the SNAPPS students expressed an uncertainty (i.e. step 4) compared with 54% of the comparison group, and that most of these uncertainties related to diagnostic reasoning.[17]
A study of medicine clerkship students evaluated the impact of extending SNAPPS to the inpatient setting and including educational prescriptions.[18] The goal was to facilitate the formulation and answering of clinical questions by using the patient, intervention, comparison, outcome (PICO) format for step 6 (selecting a case‐based issue to learn about). Dubbing this SNAPPS‐Plus, the authors found that 99% of cases included a question, and 93% of those were answered. Most questions related to therapeutics, and there was a positive correlation between questions more closely corresponding to the PICO format and higher quality answers.[18]
As with the OMP, SNAPPS does not require additional time for case presentations compared to the usual method.[16] From the perspective of a busy hospitalist, this model takes some responsibility for education away from faculty and places it on the learner. This is an important process for fostering self‐directed learning. As with the OMP, SNAPPS appears easily translatable from the outpatient to inpatient setting. Its main downside is the training time required for both parties to implement it.
TRANSLATING THE MODELS TO THE INPATIENT SETTING
The OMP and SNAPPS have largely been used in the outpatient setting. However, we propose that hospitalists can adapt either model for teaching on ward rotations, as the steps of each framework are not exclusive to one clinical setting.
Although the OMP is typically used between a preceptor and single trainee, it is well suited to engaging the entire group on inpatient rounds (Table 3). For example, a student could commit to and support a diagnosis (steps 1 and 2), whereas the intern could commit to and provide evidence for a treatment or management option. Attendings can repeat steps 1 and 2 for patients' secondary problems, encouraging learners to commit to other items on the problem list.
| Attending/ Senior Resident | Learner | Practical Tips | |
|---|---|---|---|
| |||
| Active listening. | Ms. Weinstein is a 60 year old with a history of alcohol abuse and osteoarthritis, admitted with 1 day of epigastric pain and coffee ground emesis. Workup revealed normal vital signs, mild epigastric tenderness, and mild anemia, with normal pancreatic and liver enzymes. | Learners may end their presentation here and expect you to fill in with your assessment and plan. Rather than jumping in, turn it back to the learner following the OMP model. | |
| Get a commitment | What do you think is going on? | The most likely diagnoses are upper GI bleed due to peptic ulcer disease, gastritis, or Mallory‐Weiss tear. | If the diagnosis is already established or the leaner prematurely closes the differential, ask What else could this be? |
| If the student does not expand the differential, direct this question to the intern. | |||
| Probe for supporting evidence | Why do you think this? | Peptic ulcer disease is most likely because of her alcohol abuse and her daily use of NSAIDs for arthritis pain. Gastritis is equally likely for the same reasons. Mallory‐Weiss tear is less likely, as she was not retching prior to the episode of bleeding. | Learners should use the key findings to argue for or against each diagnostic hypothesis. Novice learners often need reminders that vital signs and negative findings (e.g. absence of tachycardia) are often key findings. |
| Provide general rules | When a patient with a history of alcohol abuse has a GI bleed, you should consider whether she has underlying liver disease or a coagulopathy. If she did have liver disease, what other sources of bleeding should you consider? | Esophageal varices? | This is the step the residents tend to struggle with when teaching.[8] If your senior resident is leading the case discussion, be prepared to step in with some clinical pearls. |
| Reinforce what was done correctly | You did a nice job considering her predisposing factors, including NSAIDS and alcohol. This helped you prioritize the most likely diagnoses. | Thank you. | Tell them what they did right and the effect it had. |
| Correct mistakes | You did not address her risk for alcohol withdrawal. This increases in patients who are hospitalized for a medical illness. Next time be sure to include substance abuse in your problem list. | I'll make sure to do that. | Tell them what they did not do right and how to improve for the next time. If the student is presenting, consider asking the intern or senior resident for a management plan. |
While teaching general rules (step 3) in the group setting, hospitalists should emphasize basic principles for students (which will serve as reinforcement for residents) as well as discuss more complex rules for the edification of all team members. Hospitalists should encourage senior residents to speak up during this step and share their knowledge with the group. This is an opportunity for residents to practice their role as teachers, and for faculty to assess their clinical acumen. However, residents struggled with teaching general rules in Furney and colleagues' randomized trial.[8] Successful clinical teachers use a mix of improvisational teaching and curriculum scripts developed through years of experience.[19] Hospitalists can model this method of instruction for residents who are learning to teach. For more junior hospitalists who may still be developing their own teaching scripts, the OMP provides an opportunity to regularly integrate these scripts into rounds.
The OMP teaching encounter ends with feedback. Providing real‐time feedback to an individual in the group setting could feel awkward. Reassuringly, in Furney and colleagues' study, some of the greatest gains were in the realm of feedback, as reported by both the senior residents providing the feedback and the interns and students on the receiving end.[8] Although the OMP builds in a space for feedback, it does not teach one how to give feedback. Although it is possible that not all feedback is beneficial, trainees are eager to receive constructive input, and hospitalists should not fear providing this in front of the group. Thoughtful critique of one trainee can provide learning opportunities for others listening in.
SNAPPS is also well suited to inpatient education (Table 4). Because it emphasizes a discussion of differential diagnosis, it works well for new admissions. Because hospitalized patients usually have multiple problems, learners may repeat steps 2 and 3 for each problem, or just for the primary issue. On subsequent days, a standard presentation may work better, but if new problems arise (e.g. fever), hospitalists can ask learners to go through the SNAPPS steps for the new issue.
| Learner | Attending/ Senior Resident | Practical Tips | |
|---|---|---|---|
| |||
| 1. Summarize | Ms. Weinstein is a 60 year old with a history of alcohol abuse and osteoarthritis, admitted with 1 day of epigastric pain and coffee ground emesis. Workup revealed normal vital signs, mild epigastric tenderness, and mild anemia, with normal pancreatic and liver enzymes. | Active listening. | Rather than a complete, detailed history and physical, we emphasize tailoring the oral presentation to include only those components relevant to this admission. Then, transition to the SNAPPS presentation with a summary statement as presented here. |
| 2. Narrow the differential | The most likely diagnoses are upper GI bleed due to peptic ulcer disease, gastritis, or Mallory‐Weiss tear. | If the diagnosis is already established or the leaner prematurely closes the differential, ask What else could this be? | Hospitalized patients often have multiple problems. Learners can go through this process of SNAPPS for each problem or only the primary problem. |
| 3. Analyze the differential | Peptic ulcer disease is most likely because of her alcohol abuse and her daily use of NSAIDs for arthritis pain. Gastritis is equally likely for the same reasons. Mallory‐Weiss tear is less likely, as she was not retching prior to the episode of bleeding. | That's a very reasonable differential. You did a nice job considering her predisposing factors. What do her vital signs tell you about how much blood she has lost? | Learners should use the key findings to argue for or against each diagnostic hypothesis. Novice learners often need reminders that vital signs and negative findings (eg, absence of tachycardia) are often key findings. |
| 4. Probe the preceptor | I know alcohol increases the risk of esophageal cancer, but I was not sure if that could present like this. | You are right that she has a higher risk of cancer. Because most tumors are slow‐growing, what kind of symptoms do you think a mass in the esophagus might cause? | Guide learners to the correct answer, helping them connect pre‐existing knowledge to the question at hand. This is also a good spot to provide real‐time feedback. |
| NOTE: This is a great place for learners to ask questions that might be harder to look up, or to ask about physical findings (eg, I thought I heard crackles but was not sure. Could somebody check this with me?) | Does anyone else on the team have thoughts about this question? | Alternatively, give the senior resident an opportunity to address the question. This allows the attending to assess the senior resident's clinical reasoning and gives him or her an opportunity to practice teaching. | |
| 5. Plan management | For the suspected GI bleed, I would like to start a proton pump inhibitor, call a GI consult for an EGD, and check the hematocrit every 8 hours. We can use sequential compression devices for DVT prophylaxis. We will also counsel on alcohol cessation and monitor for withdrawal. | Good start. Does anyone else on the team want to add to the management plan? We have a pharmacist rounding with us today. Is there a difference in outcomes or costs with BID dosing versus continuous infusion of a proton pump inhibitor? | If a student is presenting, offer the intern and/or senior resident an opportunity to add to the plan.Incorporate the expertise of ancillary providers rounding with the team. |
| 6. Select a case‐related issue for self‐directed learning | I would like to look up the best way to treat her alcohol withdrawal if she develops it. | Great! We do have a protocol at the hospital, but it is a good idea to review the literature behind it. | Set aside 10 minutes before rounds each day for learners to present their findings. |
| Consider having learners write educational prescriptions following the PICO format. | |||
Step 6 of SNAPPS provides trainees an opportunity to search for and present relevant information to guide patient management. To incorporate more formal teaching time each day, set aside 10 minutes before rounds for learners to present their answers to the team. Also, because SNAPPS has the learner ask about uncertainties, faculty can use their on‐the‐fly teaching time to answer questions for which trainees do not know the answer. In the era of problem‐based learning (PBL) and medical school curricula that foster self‐directed learning from day one, many students should find SNAPPS a natural extension of PBL‐style learning from the preclinical into the clinical years.
Unlike the OMP, SNAPPS does not build in a step for feedback. Therefore, preceptors should focus on step 4 as an opportunity for this. Because feedback is paired with discussion of an uncertainty, it focuses on a trainee's immediate needs and can maximize learning opportunities.[17]
Clinical educators must simultaneously diagnose and manage patients as well as assess learners' abilities.[20] Workplace‐based assessment is particularly important for residents, and hospitalists play a pivotal role in determining their progression along the developmental milestones for achieving the ACGME competencies in medical knowledge, patient care, and practice‐based learning and improvement.[3] Both the OMP and SNAPPS frameworks encourage trainees to think out loud, providing some transparency to their thought process and enabling faculty to more accurately assess their clinical reasoning.
CONCLUSION
Many hospitalists may already use a teaching approach resembling the OMP. It has a familiar, back‐and‐forth rhythm. By explicitly following its steps, however, attendings can ensure they are providing feedback and individualized teaching with each case. SNAPPS, on the other hand, relieves faculty of their familiar role of leading the thought process and imparting teaching points. Instead, the trainee directs the encounter, leaving the attending in the role of guide.[15] SNAPPS aims to help students and residents take charge of their education and develop lifelong learning skills.
Both frameworks can be transferred from the ambulatory to inpatient setting with little modification. The OMP is older and better studied. It is easy to learn, and can be utilized by attendings and residents as teachers. In contrast, SNAPPS requires both teacher and trainee to learn the framework. Typically, this means that SNAPPS needs to be implemented systematically, via a clerkship or residency program. However, if a team was motivated, they could learn and apply it for their time together on service. Though it requires more effort to put in place, SNAPPS provides a novel approach to teaching clinical reasoning. Finally, hospitalists need not implement all steps of either framework for every teaching encounter, but can use components of either model, depending on the individual learners, team composition, time available, or clinical case.
Additional studies examining both frameworks' use for inpatient teaching and assessment would be helpful. Potential questions to address include how the team structure of inpatient rotations impacts the effectiveness of either model (e.g. which trainees benefit when committing to diagnoses or getting feedback in front of a group?), whether either model improves senior residents' ability to lead rounds and teach, whether written faculty assessments of residents are more specific and accurate with either model, and the impact of not following all steps of either model. Higher level outcomes for both models would be another area for investigation, including change in clinical performance, exam performance of students and residents, or patient outcomes, such as length of stay, cost per case, or need for rapid response/emntensive care unit transfer.
ACKNOWLEDGMENTS
Disclosure: Nothing to report.
- , , , . 2006 national hospital discharge survey. Natl Health Stat Report. 2008;(5):1–20.
- , , , , , . Challenges and opportunities in academic hospital medicine: report from the academic hospital medicine summit. J Hosp Med. 2009;4(4):240–246.
- Accreditation Council for Graduate Medical Education. Program and institutional accreditation: next accreditation system: Milestones. Available at: https://www.acgme.org/acgmeweb/tabid/430/ProgramandInstitutionalAccreditation/NextAccreditationSystem/Milestones.aspx. Accessed July 28, 2014.
- , , , . A five‐step "microskills" model of clinical teaching. J Am Board Fam Pract. 1992;5(4):419–424.
- , , . Teaching points identified by preceptors observing one‐minute preceptor and traditional preceptor encounters. Acad Med. 2004;79(1):50–55.
- , , . Effectiveness of the one‐minute preceptor model for diagnosing the patient and the learner: proof of concept. Acad Med. 2004;79(1):42–49.
- , , . Measuring outcomes of a one‐minute preceptor faculty development workshop. J Gen Intern Med. 2006;21(5):410–414.
- , , , , , . Teaching the one‐minute preceptor. A randomized controlled trial. J Gen Intern Med. 2001;16(9):620–624.
- , , , , , . Faculty development seminars based on the one‐minute preceptor improve feedback in the ambulatory setting. J Gen Intern Med. 2002;17(10):779–787.
- , , , , . Change in residents' perceptions of teaching: following a one day "residents as teachers" (RasT) workshop. South Med J. 2008;101(5):495–502.
- , , , , . Student perceptions of the one minute preceptor and traditional preceptor models. Med Teach. 2007;29(4):323–327.
- , . The one‐minute preceptor: shaping the teaching conversation. Fam Med. 2003;35(6):391–393.
- , , . Residents‐as‐teachers curricula: a critical review. Acad Med. 2009;84(3):374–380.
- , , . Teaching to teach in Toronto. Acad Psychiatry. 2010;34(4):277–281.
- , , . SNAPPS: a learner‐centered model for outpatient education. Acad Med. 2003;78(9):893–898.
- , , . Using SNAPPS to facilitate the expression of clinical reasoning and uncertainties: A randomized comparison group trial. Acad Med. 2009;84(4):517–524.
- , , , . Student uncertainties drive teaching during case presentations: more so with SNAPPS. Acad Med. 2012;87(9):1210–1217.
- , , , , , . SNAPPS‐plus: an educational prescription for students to facilitate formulating and answering clinical questions. Acad Med. 2014;89(8):1174–1179.
- . How attending physicians make instructional decisions when conducting teaching rounds. Acad Med. 1992;67(10):630–638.
- . Educational strategies to promote clinical diagnostic reasoning. N Engl J Med. 2006;355(21):2217–2225.
An important role of the hospitalist educator is to teach residents and medical students how to diagnose and manage acute medical problems. However, clinical reasoning is complex and nuanced, and there are many challenges to teaching this important process. Medical inpatients are increasingly complex, older, and more seriously ill.[1] Documentation requirements and productivity obligations compete with teaching time. Hospitalists must adjust their teaching for learners from different professions and at various levels of training. In addition, hospitalists tend to be less experienced, and must balance the need to learn their roles as clinicians with developing their own skills as educators.[2]
Despite the challenges inherent to the setting, inpatient rotations provide tremendous teaching and learning opportunities. Patients with undifferentiated complaints or known diagnoses in need of management decisions are available to stimulate discussion. Hospitalist educators have the opportunity to assess residents' progress along the developmental milestones, which residency programs are now required to report for accreditation,[3] and provide role modeling for residents who are developing their own teaching skills.
To maximize these opportunities, attendings must engage trainees to practice clinical reasoning and identify their own knowledge gaps. Various strategies for facilitating the clinical reasoning discussion exist, but two frameworksthe One‐Minute Preceptor (OMP) and SNAPPShave been well studied, albeit mainly in the outpatient setting. Both models offer ways to maximize teaching and assess clinical reasoning, but they have different methods and strengths. This article provides a narrative review of the two frameworks and discusses how they can be applied to the inpatient teaching environment. Hospitalists can utilize these models or components of each framework to facilitate teaching on inpatient teams and enhance their roles as educators.
ONE‐MINUTE PRECEPTOR
The OMP was first described in 1992 by Neher and colleagues as an alternative to the traditional model of precepting.[4] It gives preceptors a method to facilitate learners presentation of their thought process and then for the preceptor to provide targeted teaching points.[4] The OMP helps diagnose both learner and patient, whereas the traditional model focuses on diagnosing the patient.[5] In the traditional model, the attending questions the learner to diagnose the patient, which does not often make clear the learner's thinking process. Thus, there may be a mismatch between the teaching points the preceptor makes and what the learner really needs to know.[5] There are several key benefits to the OMP compared to the traditional model; broadly, these relate to improved ability to assess the learner and provide targeted teaching,[4, 5, 6, 7] improved integration of feedback,[4, 8, 9, 10] learner preference,[11] and ease with which it is learned by faculty members.[4]
The OMP model consists of five steps outlined in Table 1. Step 1, getting a commitment, can involve any aspect of the casediagnosis, treatment, or follow‐upand learners should be challenged to make intellectual commitments just beyond their level of comfort.[12] Steps 1 and 2 bring to light the learner's individual learning needs,[11] then the preceptor follows up with personalized teaching. The OMP is efficient; no increase in time was needed to precept a case in an outpatient study.[9] In a separate outpatient study, the OMP led preceptors to be more likely to teach about disease‐specific points and differential diagnosis, as compared to generic items such as history taking and presentation skills with the traditional model.[5]
| A 5‐step framework in which the preceptor does the following: |
| 1. Get a commitment |
| 2. Probe for supporting evidence |
| 3. Provide general rules |
| 4. Reinforce what was done correctly |
| 5. Correct mistakes |
Faculty feel better prepared to assess learners and provide feedback with the OMP model.[6, 9] Aagaard and colleagues provided 116 mostly ambulatory preceptors with scripted, videotaped encounters of the OMP and traditional models. The OMP improved preceptors' confidence at rating students' presentation skills, clinical reasoning, and fund of knowledge. It was rated more efficient and effective, and preceptors were able to diagnose the patient with the same or improved accuracy compared to the traditional model.[6] In a pre‐post study assessing the efficacy of a faculty development workshop, students rated ambulatory teaching encounters incorporating the OMP model as having increased quantity and quality of feedback. Furthermore, faculty reported improved ability to evaluate students and were more likely to let students reach their own conclusions and create their own postencounter learning plans.[9]
The OMP is also well‐received by trainees. Teherani and colleagues analyzed medical students' responses to videotaped teaching encounters of the OMP and traditional models. Students gave higher mean ratings for all studied items (including feedback, involving the student in decision‐making, and overall effectiveness) to the OMP model, and preferred it over the traditional model.[11]
Several studies have evaluated the OMP for use by residents as teachers,[10, 13, 14] and it is one of the most common models taught to residents.[13] One study evaluated the impact of a one‐day workshop for 276 residents that included the five‐step microskills model (also known as the OMP).[10] Residents felt more prepared to teach, set expectations, and provide feedback.[10] The OMP model, despite brief training, is effective in improving residents' teaching effectiveness and confidence.[13]
The only study we found that exclusively evaluated the OMP in the inpatient setting was a randomized trial[8] involving 57 internal medicine residents. Interns and students rated OMP‐trained residents more highly in 4 of 5 behaviors. The behavior that showed no difference from the control group was teaching general rules.[8] However, there was no difference in ratings of overall teaching effectiveness between groups.[8]
Our review of the literature on the OMP shows it is a quickly learned, easily implemented framework for teaching clinical reasoning. It has been used across specialties and settings, provides a built‐in mechanism for feedback, and allows educators to assess trainees' reasoning while extracting the clinical information needed to work efficiently.
SNAPPS
SNAPPS was first described in 2003 by Wolpaw and colleagues. It is a six‐step learner‐centered model as outlined in Table 2.[15] Unlike the OMP, SNAPPS requires both trainee and teacher to learn the framework. In doing so, the responsibility for directing the teaching encounter is shifted toward the learner.[15] Consequently, this model may be best suited to advanced or motivated learners. Like the OMP, SNAPPS was originally described for the ambulatory environment. However, it has been studied in the inpatient setting as well.
| A 6‐step framework in which the learner does the following: |
| 1. Summarize briefly the history and findings |
| 2. Narrow the differential to 2 or 3 possibilities |
| 3. Analyze the differential by comparing/contrasting the possibilities |
| 4. Probe the preceptor by asking questions |
| 5. Plan management for the patient's medical issues |
| 6. Select a case‐related issue for self‐directed learning |
With SNAPPS, the teaching encounter is learner driven. The trainee presents the case and directs the discussion of differential diagnosis. The educator does not have an active role until the fourth step, where the learner asks questions or identifies areas of uncertainty. But even at this stage, the discussion is learner driven. Step 5, planning management, is collaborative, with trainees suggesting management plans with appropriate attending guidance. Depending on learner skill level or case difficulty, the preceptor may need to play more or less of an active role. The final step, picking a case‐related issue to examine, extends the learning beyond the initial encounter, and ensures that it is individualized and relevant. This step also encourages learner progression toward the Accreditation Council for Graduate Medical Education (ACGME) competency of practice‐based learning and improvement.[3]
A handful of studies have evaluated the SNAPPS model. A randomized comparison group trial found that SNAPPS‐trained students outperformed students trained to elicit feedback and students who received the usual and customary preparation.[16] Notably, SNAPPS students expressed more than twice as many differential diagnoses, justified their reasoning more than five times as often, and expressed more questions and uncertainties. The SNAPPS students' presentations were no longer than in the usual and customary group, and were just one minute longer than in the group trained to elicit feedback.[16] A follow‐up analysis found that 100% of the SNAPPS students expressed an uncertainty (i.e. step 4) compared with 54% of the comparison group, and that most of these uncertainties related to diagnostic reasoning.[17]
A study of medicine clerkship students evaluated the impact of extending SNAPPS to the inpatient setting and including educational prescriptions.[18] The goal was to facilitate the formulation and answering of clinical questions by using the patient, intervention, comparison, outcome (PICO) format for step 6 (selecting a case‐based issue to learn about). Dubbing this SNAPPS‐Plus, the authors found that 99% of cases included a question, and 93% of those were answered. Most questions related to therapeutics, and there was a positive correlation between questions more closely corresponding to the PICO format and higher quality answers.[18]
As with the OMP, SNAPPS does not require additional time for case presentations compared to the usual method.[16] From the perspective of a busy hospitalist, this model takes some responsibility for education away from faculty and places it on the learner. This is an important process for fostering self‐directed learning. As with the OMP, SNAPPS appears easily translatable from the outpatient to inpatient setting. Its main downside is the training time required for both parties to implement it.
TRANSLATING THE MODELS TO THE INPATIENT SETTING
The OMP and SNAPPS have largely been used in the outpatient setting. However, we propose that hospitalists can adapt either model for teaching on ward rotations, as the steps of each framework are not exclusive to one clinical setting.
Although the OMP is typically used between a preceptor and single trainee, it is well suited to engaging the entire group on inpatient rounds (Table 3). For example, a student could commit to and support a diagnosis (steps 1 and 2), whereas the intern could commit to and provide evidence for a treatment or management option. Attendings can repeat steps 1 and 2 for patients' secondary problems, encouraging learners to commit to other items on the problem list.
| Attending/ Senior Resident | Learner | Practical Tips | |
|---|---|---|---|
| |||
| Active listening. | Ms. Weinstein is a 60 year old with a history of alcohol abuse and osteoarthritis, admitted with 1 day of epigastric pain and coffee ground emesis. Workup revealed normal vital signs, mild epigastric tenderness, and mild anemia, with normal pancreatic and liver enzymes. | Learners may end their presentation here and expect you to fill in with your assessment and plan. Rather than jumping in, turn it back to the learner following the OMP model. | |
| Get a commitment | What do you think is going on? | The most likely diagnoses are upper GI bleed due to peptic ulcer disease, gastritis, or Mallory‐Weiss tear. | If the diagnosis is already established or the leaner prematurely closes the differential, ask What else could this be? |
| If the student does not expand the differential, direct this question to the intern. | |||
| Probe for supporting evidence | Why do you think this? | Peptic ulcer disease is most likely because of her alcohol abuse and her daily use of NSAIDs for arthritis pain. Gastritis is equally likely for the same reasons. Mallory‐Weiss tear is less likely, as she was not retching prior to the episode of bleeding. | Learners should use the key findings to argue for or against each diagnostic hypothesis. Novice learners often need reminders that vital signs and negative findings (e.g. absence of tachycardia) are often key findings. |
| Provide general rules | When a patient with a history of alcohol abuse has a GI bleed, you should consider whether she has underlying liver disease or a coagulopathy. If she did have liver disease, what other sources of bleeding should you consider? | Esophageal varices? | This is the step the residents tend to struggle with when teaching.[8] If your senior resident is leading the case discussion, be prepared to step in with some clinical pearls. |
| Reinforce what was done correctly | You did a nice job considering her predisposing factors, including NSAIDS and alcohol. This helped you prioritize the most likely diagnoses. | Thank you. | Tell them what they did right and the effect it had. |
| Correct mistakes | You did not address her risk for alcohol withdrawal. This increases in patients who are hospitalized for a medical illness. Next time be sure to include substance abuse in your problem list. | I'll make sure to do that. | Tell them what they did not do right and how to improve for the next time. If the student is presenting, consider asking the intern or senior resident for a management plan. |
While teaching general rules (step 3) in the group setting, hospitalists should emphasize basic principles for students (which will serve as reinforcement for residents) as well as discuss more complex rules for the edification of all team members. Hospitalists should encourage senior residents to speak up during this step and share their knowledge with the group. This is an opportunity for residents to practice their role as teachers, and for faculty to assess their clinical acumen. However, residents struggled with teaching general rules in Furney and colleagues' randomized trial.[8] Successful clinical teachers use a mix of improvisational teaching and curriculum scripts developed through years of experience.[19] Hospitalists can model this method of instruction for residents who are learning to teach. For more junior hospitalists who may still be developing their own teaching scripts, the OMP provides an opportunity to regularly integrate these scripts into rounds.
The OMP teaching encounter ends with feedback. Providing real‐time feedback to an individual in the group setting could feel awkward. Reassuringly, in Furney and colleagues' study, some of the greatest gains were in the realm of feedback, as reported by both the senior residents providing the feedback and the interns and students on the receiving end.[8] Although the OMP builds in a space for feedback, it does not teach one how to give feedback. Although it is possible that not all feedback is beneficial, trainees are eager to receive constructive input, and hospitalists should not fear providing this in front of the group. Thoughtful critique of one trainee can provide learning opportunities for others listening in.
SNAPPS is also well suited to inpatient education (Table 4). Because it emphasizes a discussion of differential diagnosis, it works well for new admissions. Because hospitalized patients usually have multiple problems, learners may repeat steps 2 and 3 for each problem, or just for the primary issue. On subsequent days, a standard presentation may work better, but if new problems arise (e.g. fever), hospitalists can ask learners to go through the SNAPPS steps for the new issue.
| Learner | Attending/ Senior Resident | Practical Tips | |
|---|---|---|---|
| |||
| 1. Summarize | Ms. Weinstein is a 60 year old with a history of alcohol abuse and osteoarthritis, admitted with 1 day of epigastric pain and coffee ground emesis. Workup revealed normal vital signs, mild epigastric tenderness, and mild anemia, with normal pancreatic and liver enzymes. | Active listening. | Rather than a complete, detailed history and physical, we emphasize tailoring the oral presentation to include only those components relevant to this admission. Then, transition to the SNAPPS presentation with a summary statement as presented here. |
| 2. Narrow the differential | The most likely diagnoses are upper GI bleed due to peptic ulcer disease, gastritis, or Mallory‐Weiss tear. | If the diagnosis is already established or the leaner prematurely closes the differential, ask What else could this be? | Hospitalized patients often have multiple problems. Learners can go through this process of SNAPPS for each problem or only the primary problem. |
| 3. Analyze the differential | Peptic ulcer disease is most likely because of her alcohol abuse and her daily use of NSAIDs for arthritis pain. Gastritis is equally likely for the same reasons. Mallory‐Weiss tear is less likely, as she was not retching prior to the episode of bleeding. | That's a very reasonable differential. You did a nice job considering her predisposing factors. What do her vital signs tell you about how much blood she has lost? | Learners should use the key findings to argue for or against each diagnostic hypothesis. Novice learners often need reminders that vital signs and negative findings (eg, absence of tachycardia) are often key findings. |
| 4. Probe the preceptor | I know alcohol increases the risk of esophageal cancer, but I was not sure if that could present like this. | You are right that she has a higher risk of cancer. Because most tumors are slow‐growing, what kind of symptoms do you think a mass in the esophagus might cause? | Guide learners to the correct answer, helping them connect pre‐existing knowledge to the question at hand. This is also a good spot to provide real‐time feedback. |
| NOTE: This is a great place for learners to ask questions that might be harder to look up, or to ask about physical findings (eg, I thought I heard crackles but was not sure. Could somebody check this with me?) | Does anyone else on the team have thoughts about this question? | Alternatively, give the senior resident an opportunity to address the question. This allows the attending to assess the senior resident's clinical reasoning and gives him or her an opportunity to practice teaching. | |
| 5. Plan management | For the suspected GI bleed, I would like to start a proton pump inhibitor, call a GI consult for an EGD, and check the hematocrit every 8 hours. We can use sequential compression devices for DVT prophylaxis. We will also counsel on alcohol cessation and monitor for withdrawal. | Good start. Does anyone else on the team want to add to the management plan? We have a pharmacist rounding with us today. Is there a difference in outcomes or costs with BID dosing versus continuous infusion of a proton pump inhibitor? | If a student is presenting, offer the intern and/or senior resident an opportunity to add to the plan.Incorporate the expertise of ancillary providers rounding with the team. |
| 6. Select a case‐related issue for self‐directed learning | I would like to look up the best way to treat her alcohol withdrawal if she develops it. | Great! We do have a protocol at the hospital, but it is a good idea to review the literature behind it. | Set aside 10 minutes before rounds each day for learners to present their findings. |
| Consider having learners write educational prescriptions following the PICO format. | |||
Step 6 of SNAPPS provides trainees an opportunity to search for and present relevant information to guide patient management. To incorporate more formal teaching time each day, set aside 10 minutes before rounds for learners to present their answers to the team. Also, because SNAPPS has the learner ask about uncertainties, faculty can use their on‐the‐fly teaching time to answer questions for which trainees do not know the answer. In the era of problem‐based learning (PBL) and medical school curricula that foster self‐directed learning from day one, many students should find SNAPPS a natural extension of PBL‐style learning from the preclinical into the clinical years.
Unlike the OMP, SNAPPS does not build in a step for feedback. Therefore, preceptors should focus on step 4 as an opportunity for this. Because feedback is paired with discussion of an uncertainty, it focuses on a trainee's immediate needs and can maximize learning opportunities.[17]
Clinical educators must simultaneously diagnose and manage patients as well as assess learners' abilities.[20] Workplace‐based assessment is particularly important for residents, and hospitalists play a pivotal role in determining their progression along the developmental milestones for achieving the ACGME competencies in medical knowledge, patient care, and practice‐based learning and improvement.[3] Both the OMP and SNAPPS frameworks encourage trainees to think out loud, providing some transparency to their thought process and enabling faculty to more accurately assess their clinical reasoning.
CONCLUSION
Many hospitalists may already use a teaching approach resembling the OMP. It has a familiar, back‐and‐forth rhythm. By explicitly following its steps, however, attendings can ensure they are providing feedback and individualized teaching with each case. SNAPPS, on the other hand, relieves faculty of their familiar role of leading the thought process and imparting teaching points. Instead, the trainee directs the encounter, leaving the attending in the role of guide.[15] SNAPPS aims to help students and residents take charge of their education and develop lifelong learning skills.
Both frameworks can be transferred from the ambulatory to inpatient setting with little modification. The OMP is older and better studied. It is easy to learn, and can be utilized by attendings and residents as teachers. In contrast, SNAPPS requires both teacher and trainee to learn the framework. Typically, this means that SNAPPS needs to be implemented systematically, via a clerkship or residency program. However, if a team was motivated, they could learn and apply it for their time together on service. Though it requires more effort to put in place, SNAPPS provides a novel approach to teaching clinical reasoning. Finally, hospitalists need not implement all steps of either framework for every teaching encounter, but can use components of either model, depending on the individual learners, team composition, time available, or clinical case.
Additional studies examining both frameworks' use for inpatient teaching and assessment would be helpful. Potential questions to address include how the team structure of inpatient rotations impacts the effectiveness of either model (e.g. which trainees benefit when committing to diagnoses or getting feedback in front of a group?), whether either model improves senior residents' ability to lead rounds and teach, whether written faculty assessments of residents are more specific and accurate with either model, and the impact of not following all steps of either model. Higher level outcomes for both models would be another area for investigation, including change in clinical performance, exam performance of students and residents, or patient outcomes, such as length of stay, cost per case, or need for rapid response/emntensive care unit transfer.
ACKNOWLEDGMENTS
Disclosure: Nothing to report.
An important role of the hospitalist educator is to teach residents and medical students how to diagnose and manage acute medical problems. However, clinical reasoning is complex and nuanced, and there are many challenges to teaching this important process. Medical inpatients are increasingly complex, older, and more seriously ill.[1] Documentation requirements and productivity obligations compete with teaching time. Hospitalists must adjust their teaching for learners from different professions and at various levels of training. In addition, hospitalists tend to be less experienced, and must balance the need to learn their roles as clinicians with developing their own skills as educators.[2]
Despite the challenges inherent to the setting, inpatient rotations provide tremendous teaching and learning opportunities. Patients with undifferentiated complaints or known diagnoses in need of management decisions are available to stimulate discussion. Hospitalist educators have the opportunity to assess residents' progress along the developmental milestones, which residency programs are now required to report for accreditation,[3] and provide role modeling for residents who are developing their own teaching skills.
To maximize these opportunities, attendings must engage trainees to practice clinical reasoning and identify their own knowledge gaps. Various strategies for facilitating the clinical reasoning discussion exist, but two frameworksthe One‐Minute Preceptor (OMP) and SNAPPShave been well studied, albeit mainly in the outpatient setting. Both models offer ways to maximize teaching and assess clinical reasoning, but they have different methods and strengths. This article provides a narrative review of the two frameworks and discusses how they can be applied to the inpatient teaching environment. Hospitalists can utilize these models or components of each framework to facilitate teaching on inpatient teams and enhance their roles as educators.
ONE‐MINUTE PRECEPTOR
The OMP was first described in 1992 by Neher and colleagues as an alternative to the traditional model of precepting.[4] It gives preceptors a method to facilitate learners presentation of their thought process and then for the preceptor to provide targeted teaching points.[4] The OMP helps diagnose both learner and patient, whereas the traditional model focuses on diagnosing the patient.[5] In the traditional model, the attending questions the learner to diagnose the patient, which does not often make clear the learner's thinking process. Thus, there may be a mismatch between the teaching points the preceptor makes and what the learner really needs to know.[5] There are several key benefits to the OMP compared to the traditional model; broadly, these relate to improved ability to assess the learner and provide targeted teaching,[4, 5, 6, 7] improved integration of feedback,[4, 8, 9, 10] learner preference,[11] and ease with which it is learned by faculty members.[4]
The OMP model consists of five steps outlined in Table 1. Step 1, getting a commitment, can involve any aspect of the casediagnosis, treatment, or follow‐upand learners should be challenged to make intellectual commitments just beyond their level of comfort.[12] Steps 1 and 2 bring to light the learner's individual learning needs,[11] then the preceptor follows up with personalized teaching. The OMP is efficient; no increase in time was needed to precept a case in an outpatient study.[9] In a separate outpatient study, the OMP led preceptors to be more likely to teach about disease‐specific points and differential diagnosis, as compared to generic items such as history taking and presentation skills with the traditional model.[5]
| A 5‐step framework in which the preceptor does the following: |
| 1. Get a commitment |
| 2. Probe for supporting evidence |
| 3. Provide general rules |
| 4. Reinforce what was done correctly |
| 5. Correct mistakes |
Faculty feel better prepared to assess learners and provide feedback with the OMP model.[6, 9] Aagaard and colleagues provided 116 mostly ambulatory preceptors with scripted, videotaped encounters of the OMP and traditional models. The OMP improved preceptors' confidence at rating students' presentation skills, clinical reasoning, and fund of knowledge. It was rated more efficient and effective, and preceptors were able to diagnose the patient with the same or improved accuracy compared to the traditional model.[6] In a pre‐post study assessing the efficacy of a faculty development workshop, students rated ambulatory teaching encounters incorporating the OMP model as having increased quantity and quality of feedback. Furthermore, faculty reported improved ability to evaluate students and were more likely to let students reach their own conclusions and create their own postencounter learning plans.[9]
The OMP is also well‐received by trainees. Teherani and colleagues analyzed medical students' responses to videotaped teaching encounters of the OMP and traditional models. Students gave higher mean ratings for all studied items (including feedback, involving the student in decision‐making, and overall effectiveness) to the OMP model, and preferred it over the traditional model.[11]
Several studies have evaluated the OMP for use by residents as teachers,[10, 13, 14] and it is one of the most common models taught to residents.[13] One study evaluated the impact of a one‐day workshop for 276 residents that included the five‐step microskills model (also known as the OMP).[10] Residents felt more prepared to teach, set expectations, and provide feedback.[10] The OMP model, despite brief training, is effective in improving residents' teaching effectiveness and confidence.[13]
The only study we found that exclusively evaluated the OMP in the inpatient setting was a randomized trial[8] involving 57 internal medicine residents. Interns and students rated OMP‐trained residents more highly in 4 of 5 behaviors. The behavior that showed no difference from the control group was teaching general rules.[8] However, there was no difference in ratings of overall teaching effectiveness between groups.[8]
Our review of the literature on the OMP shows it is a quickly learned, easily implemented framework for teaching clinical reasoning. It has been used across specialties and settings, provides a built‐in mechanism for feedback, and allows educators to assess trainees' reasoning while extracting the clinical information needed to work efficiently.
SNAPPS
SNAPPS was first described in 2003 by Wolpaw and colleagues. It is a six‐step learner‐centered model as outlined in Table 2.[15] Unlike the OMP, SNAPPS requires both trainee and teacher to learn the framework. In doing so, the responsibility for directing the teaching encounter is shifted toward the learner.[15] Consequently, this model may be best suited to advanced or motivated learners. Like the OMP, SNAPPS was originally described for the ambulatory environment. However, it has been studied in the inpatient setting as well.
| A 6‐step framework in which the learner does the following: |
| 1. Summarize briefly the history and findings |
| 2. Narrow the differential to 2 or 3 possibilities |
| 3. Analyze the differential by comparing/contrasting the possibilities |
| 4. Probe the preceptor by asking questions |
| 5. Plan management for the patient's medical issues |
| 6. Select a case‐related issue for self‐directed learning |
With SNAPPS, the teaching encounter is learner driven. The trainee presents the case and directs the discussion of differential diagnosis. The educator does not have an active role until the fourth step, where the learner asks questions or identifies areas of uncertainty. But even at this stage, the discussion is learner driven. Step 5, planning management, is collaborative, with trainees suggesting management plans with appropriate attending guidance. Depending on learner skill level or case difficulty, the preceptor may need to play more or less of an active role. The final step, picking a case‐related issue to examine, extends the learning beyond the initial encounter, and ensures that it is individualized and relevant. This step also encourages learner progression toward the Accreditation Council for Graduate Medical Education (ACGME) competency of practice‐based learning and improvement.[3]
A handful of studies have evaluated the SNAPPS model. A randomized comparison group trial found that SNAPPS‐trained students outperformed students trained to elicit feedback and students who received the usual and customary preparation.[16] Notably, SNAPPS students expressed more than twice as many differential diagnoses, justified their reasoning more than five times as often, and expressed more questions and uncertainties. The SNAPPS students' presentations were no longer than in the usual and customary group, and were just one minute longer than in the group trained to elicit feedback.[16] A follow‐up analysis found that 100% of the SNAPPS students expressed an uncertainty (i.e. step 4) compared with 54% of the comparison group, and that most of these uncertainties related to diagnostic reasoning.[17]
A study of medicine clerkship students evaluated the impact of extending SNAPPS to the inpatient setting and including educational prescriptions.[18] The goal was to facilitate the formulation and answering of clinical questions by using the patient, intervention, comparison, outcome (PICO) format for step 6 (selecting a case‐based issue to learn about). Dubbing this SNAPPS‐Plus, the authors found that 99% of cases included a question, and 93% of those were answered. Most questions related to therapeutics, and there was a positive correlation between questions more closely corresponding to the PICO format and higher quality answers.[18]
As with the OMP, SNAPPS does not require additional time for case presentations compared to the usual method.[16] From the perspective of a busy hospitalist, this model takes some responsibility for education away from faculty and places it on the learner. This is an important process for fostering self‐directed learning. As with the OMP, SNAPPS appears easily translatable from the outpatient to inpatient setting. Its main downside is the training time required for both parties to implement it.
TRANSLATING THE MODELS TO THE INPATIENT SETTING
The OMP and SNAPPS have largely been used in the outpatient setting. However, we propose that hospitalists can adapt either model for teaching on ward rotations, as the steps of each framework are not exclusive to one clinical setting.
Although the OMP is typically used between a preceptor and single trainee, it is well suited to engaging the entire group on inpatient rounds (Table 3). For example, a student could commit to and support a diagnosis (steps 1 and 2), whereas the intern could commit to and provide evidence for a treatment or management option. Attendings can repeat steps 1 and 2 for patients' secondary problems, encouraging learners to commit to other items on the problem list.
| Attending/ Senior Resident | Learner | Practical Tips | |
|---|---|---|---|
| |||
| Active listening. | Ms. Weinstein is a 60 year old with a history of alcohol abuse and osteoarthritis, admitted with 1 day of epigastric pain and coffee ground emesis. Workup revealed normal vital signs, mild epigastric tenderness, and mild anemia, with normal pancreatic and liver enzymes. | Learners may end their presentation here and expect you to fill in with your assessment and plan. Rather than jumping in, turn it back to the learner following the OMP model. | |
| Get a commitment | What do you think is going on? | The most likely diagnoses are upper GI bleed due to peptic ulcer disease, gastritis, or Mallory‐Weiss tear. | If the diagnosis is already established or the leaner prematurely closes the differential, ask What else could this be? |
| If the student does not expand the differential, direct this question to the intern. | |||
| Probe for supporting evidence | Why do you think this? | Peptic ulcer disease is most likely because of her alcohol abuse and her daily use of NSAIDs for arthritis pain. Gastritis is equally likely for the same reasons. Mallory‐Weiss tear is less likely, as she was not retching prior to the episode of bleeding. | Learners should use the key findings to argue for or against each diagnostic hypothesis. Novice learners often need reminders that vital signs and negative findings (e.g. absence of tachycardia) are often key findings. |
| Provide general rules | When a patient with a history of alcohol abuse has a GI bleed, you should consider whether she has underlying liver disease or a coagulopathy. If she did have liver disease, what other sources of bleeding should you consider? | Esophageal varices? | This is the step the residents tend to struggle with when teaching.[8] If your senior resident is leading the case discussion, be prepared to step in with some clinical pearls. |
| Reinforce what was done correctly | You did a nice job considering her predisposing factors, including NSAIDS and alcohol. This helped you prioritize the most likely diagnoses. | Thank you. | Tell them what they did right and the effect it had. |
| Correct mistakes | You did not address her risk for alcohol withdrawal. This increases in patients who are hospitalized for a medical illness. Next time be sure to include substance abuse in your problem list. | I'll make sure to do that. | Tell them what they did not do right and how to improve for the next time. If the student is presenting, consider asking the intern or senior resident for a management plan. |
While teaching general rules (step 3) in the group setting, hospitalists should emphasize basic principles for students (which will serve as reinforcement for residents) as well as discuss more complex rules for the edification of all team members. Hospitalists should encourage senior residents to speak up during this step and share their knowledge with the group. This is an opportunity for residents to practice their role as teachers, and for faculty to assess their clinical acumen. However, residents struggled with teaching general rules in Furney and colleagues' randomized trial.[8] Successful clinical teachers use a mix of improvisational teaching and curriculum scripts developed through years of experience.[19] Hospitalists can model this method of instruction for residents who are learning to teach. For more junior hospitalists who may still be developing their own teaching scripts, the OMP provides an opportunity to regularly integrate these scripts into rounds.
The OMP teaching encounter ends with feedback. Providing real‐time feedback to an individual in the group setting could feel awkward. Reassuringly, in Furney and colleagues' study, some of the greatest gains were in the realm of feedback, as reported by both the senior residents providing the feedback and the interns and students on the receiving end.[8] Although the OMP builds in a space for feedback, it does not teach one how to give feedback. Although it is possible that not all feedback is beneficial, trainees are eager to receive constructive input, and hospitalists should not fear providing this in front of the group. Thoughtful critique of one trainee can provide learning opportunities for others listening in.
SNAPPS is also well suited to inpatient education (Table 4). Because it emphasizes a discussion of differential diagnosis, it works well for new admissions. Because hospitalized patients usually have multiple problems, learners may repeat steps 2 and 3 for each problem, or just for the primary issue. On subsequent days, a standard presentation may work better, but if new problems arise (e.g. fever), hospitalists can ask learners to go through the SNAPPS steps for the new issue.
| Learner | Attending/ Senior Resident | Practical Tips | |
|---|---|---|---|
| |||
| 1. Summarize | Ms. Weinstein is a 60 year old with a history of alcohol abuse and osteoarthritis, admitted with 1 day of epigastric pain and coffee ground emesis. Workup revealed normal vital signs, mild epigastric tenderness, and mild anemia, with normal pancreatic and liver enzymes. | Active listening. | Rather than a complete, detailed history and physical, we emphasize tailoring the oral presentation to include only those components relevant to this admission. Then, transition to the SNAPPS presentation with a summary statement as presented here. |
| 2. Narrow the differential | The most likely diagnoses are upper GI bleed due to peptic ulcer disease, gastritis, or Mallory‐Weiss tear. | If the diagnosis is already established or the leaner prematurely closes the differential, ask What else could this be? | Hospitalized patients often have multiple problems. Learners can go through this process of SNAPPS for each problem or only the primary problem. |
| 3. Analyze the differential | Peptic ulcer disease is most likely because of her alcohol abuse and her daily use of NSAIDs for arthritis pain. Gastritis is equally likely for the same reasons. Mallory‐Weiss tear is less likely, as she was not retching prior to the episode of bleeding. | That's a very reasonable differential. You did a nice job considering her predisposing factors. What do her vital signs tell you about how much blood she has lost? | Learners should use the key findings to argue for or against each diagnostic hypothesis. Novice learners often need reminders that vital signs and negative findings (eg, absence of tachycardia) are often key findings. |
| 4. Probe the preceptor | I know alcohol increases the risk of esophageal cancer, but I was not sure if that could present like this. | You are right that she has a higher risk of cancer. Because most tumors are slow‐growing, what kind of symptoms do you think a mass in the esophagus might cause? | Guide learners to the correct answer, helping them connect pre‐existing knowledge to the question at hand. This is also a good spot to provide real‐time feedback. |
| NOTE: This is a great place for learners to ask questions that might be harder to look up, or to ask about physical findings (eg, I thought I heard crackles but was not sure. Could somebody check this with me?) | Does anyone else on the team have thoughts about this question? | Alternatively, give the senior resident an opportunity to address the question. This allows the attending to assess the senior resident's clinical reasoning and gives him or her an opportunity to practice teaching. | |
| 5. Plan management | For the suspected GI bleed, I would like to start a proton pump inhibitor, call a GI consult for an EGD, and check the hematocrit every 8 hours. We can use sequential compression devices for DVT prophylaxis. We will also counsel on alcohol cessation and monitor for withdrawal. | Good start. Does anyone else on the team want to add to the management plan? We have a pharmacist rounding with us today. Is there a difference in outcomes or costs with BID dosing versus continuous infusion of a proton pump inhibitor? | If a student is presenting, offer the intern and/or senior resident an opportunity to add to the plan.Incorporate the expertise of ancillary providers rounding with the team. |
| 6. Select a case‐related issue for self‐directed learning | I would like to look up the best way to treat her alcohol withdrawal if she develops it. | Great! We do have a protocol at the hospital, but it is a good idea to review the literature behind it. | Set aside 10 minutes before rounds each day for learners to present their findings. |
| Consider having learners write educational prescriptions following the PICO format. | |||
Step 6 of SNAPPS provides trainees an opportunity to search for and present relevant information to guide patient management. To incorporate more formal teaching time each day, set aside 10 minutes before rounds for learners to present their answers to the team. Also, because SNAPPS has the learner ask about uncertainties, faculty can use their on‐the‐fly teaching time to answer questions for which trainees do not know the answer. In the era of problem‐based learning (PBL) and medical school curricula that foster self‐directed learning from day one, many students should find SNAPPS a natural extension of PBL‐style learning from the preclinical into the clinical years.
Unlike the OMP, SNAPPS does not build in a step for feedback. Therefore, preceptors should focus on step 4 as an opportunity for this. Because feedback is paired with discussion of an uncertainty, it focuses on a trainee's immediate needs and can maximize learning opportunities.[17]
Clinical educators must simultaneously diagnose and manage patients as well as assess learners' abilities.[20] Workplace‐based assessment is particularly important for residents, and hospitalists play a pivotal role in determining their progression along the developmental milestones for achieving the ACGME competencies in medical knowledge, patient care, and practice‐based learning and improvement.[3] Both the OMP and SNAPPS frameworks encourage trainees to think out loud, providing some transparency to their thought process and enabling faculty to more accurately assess their clinical reasoning.
CONCLUSION
Many hospitalists may already use a teaching approach resembling the OMP. It has a familiar, back‐and‐forth rhythm. By explicitly following its steps, however, attendings can ensure they are providing feedback and individualized teaching with each case. SNAPPS, on the other hand, relieves faculty of their familiar role of leading the thought process and imparting teaching points. Instead, the trainee directs the encounter, leaving the attending in the role of guide.[15] SNAPPS aims to help students and residents take charge of their education and develop lifelong learning skills.
Both frameworks can be transferred from the ambulatory to inpatient setting with little modification. The OMP is older and better studied. It is easy to learn, and can be utilized by attendings and residents as teachers. In contrast, SNAPPS requires both teacher and trainee to learn the framework. Typically, this means that SNAPPS needs to be implemented systematically, via a clerkship or residency program. However, if a team was motivated, they could learn and apply it for their time together on service. Though it requires more effort to put in place, SNAPPS provides a novel approach to teaching clinical reasoning. Finally, hospitalists need not implement all steps of either framework for every teaching encounter, but can use components of either model, depending on the individual learners, team composition, time available, or clinical case.
Additional studies examining both frameworks' use for inpatient teaching and assessment would be helpful. Potential questions to address include how the team structure of inpatient rotations impacts the effectiveness of either model (e.g. which trainees benefit when committing to diagnoses or getting feedback in front of a group?), whether either model improves senior residents' ability to lead rounds and teach, whether written faculty assessments of residents are more specific and accurate with either model, and the impact of not following all steps of either model. Higher level outcomes for both models would be another area for investigation, including change in clinical performance, exam performance of students and residents, or patient outcomes, such as length of stay, cost per case, or need for rapid response/emntensive care unit transfer.
ACKNOWLEDGMENTS
Disclosure: Nothing to report.
- , , , . 2006 national hospital discharge survey. Natl Health Stat Report. 2008;(5):1–20.
- , , , , , . Challenges and opportunities in academic hospital medicine: report from the academic hospital medicine summit. J Hosp Med. 2009;4(4):240–246.
- Accreditation Council for Graduate Medical Education. Program and institutional accreditation: next accreditation system: Milestones. Available at: https://www.acgme.org/acgmeweb/tabid/430/ProgramandInstitutionalAccreditation/NextAccreditationSystem/Milestones.aspx. Accessed July 28, 2014.
- , , , . A five‐step "microskills" model of clinical teaching. J Am Board Fam Pract. 1992;5(4):419–424.
- , , . Teaching points identified by preceptors observing one‐minute preceptor and traditional preceptor encounters. Acad Med. 2004;79(1):50–55.
- , , . Effectiveness of the one‐minute preceptor model for diagnosing the patient and the learner: proof of concept. Acad Med. 2004;79(1):42–49.
- , , . Measuring outcomes of a one‐minute preceptor faculty development workshop. J Gen Intern Med. 2006;21(5):410–414.
- , , , , , . Teaching the one‐minute preceptor. A randomized controlled trial. J Gen Intern Med. 2001;16(9):620–624.
- , , , , , . Faculty development seminars based on the one‐minute preceptor improve feedback in the ambulatory setting. J Gen Intern Med. 2002;17(10):779–787.
- , , , , . Change in residents' perceptions of teaching: following a one day "residents as teachers" (RasT) workshop. South Med J. 2008;101(5):495–502.
- , , , , . Student perceptions of the one minute preceptor and traditional preceptor models. Med Teach. 2007;29(4):323–327.
- , . The one‐minute preceptor: shaping the teaching conversation. Fam Med. 2003;35(6):391–393.
- , , . Residents‐as‐teachers curricula: a critical review. Acad Med. 2009;84(3):374–380.
- , , . Teaching to teach in Toronto. Acad Psychiatry. 2010;34(4):277–281.
- , , . SNAPPS: a learner‐centered model for outpatient education. Acad Med. 2003;78(9):893–898.
- , , . Using SNAPPS to facilitate the expression of clinical reasoning and uncertainties: A randomized comparison group trial. Acad Med. 2009;84(4):517–524.
- , , , . Student uncertainties drive teaching during case presentations: more so with SNAPPS. Acad Med. 2012;87(9):1210–1217.
- , , , , , . SNAPPS‐plus: an educational prescription for students to facilitate formulating and answering clinical questions. Acad Med. 2014;89(8):1174–1179.
- . How attending physicians make instructional decisions when conducting teaching rounds. Acad Med. 1992;67(10):630–638.
- . Educational strategies to promote clinical diagnostic reasoning. N Engl J Med. 2006;355(21):2217–2225.
- , , , . 2006 national hospital discharge survey. Natl Health Stat Report. 2008;(5):1–20.
- , , , , , . Challenges and opportunities in academic hospital medicine: report from the academic hospital medicine summit. J Hosp Med. 2009;4(4):240–246.
- Accreditation Council for Graduate Medical Education. Program and institutional accreditation: next accreditation system: Milestones. Available at: https://www.acgme.org/acgmeweb/tabid/430/ProgramandInstitutionalAccreditation/NextAccreditationSystem/Milestones.aspx. Accessed July 28, 2014.
- , , , . A five‐step "microskills" model of clinical teaching. J Am Board Fam Pract. 1992;5(4):419–424.
- , , . Teaching points identified by preceptors observing one‐minute preceptor and traditional preceptor encounters. Acad Med. 2004;79(1):50–55.
- , , . Effectiveness of the one‐minute preceptor model for diagnosing the patient and the learner: proof of concept. Acad Med. 2004;79(1):42–49.
- , , . Measuring outcomes of a one‐minute preceptor faculty development workshop. J Gen Intern Med. 2006;21(5):410–414.
- , , , , , . Teaching the one‐minute preceptor. A randomized controlled trial. J Gen Intern Med. 2001;16(9):620–624.
- , , , , , . Faculty development seminars based on the one‐minute preceptor improve feedback in the ambulatory setting. J Gen Intern Med. 2002;17(10):779–787.
- , , , , . Change in residents' perceptions of teaching: following a one day "residents as teachers" (RasT) workshop. South Med J. 2008;101(5):495–502.
- , , , , . Student perceptions of the one minute preceptor and traditional preceptor models. Med Teach. 2007;29(4):323–327.
- , . The one‐minute preceptor: shaping the teaching conversation. Fam Med. 2003;35(6):391–393.
- , , . Residents‐as‐teachers curricula: a critical review. Acad Med. 2009;84(3):374–380.
- , , . Teaching to teach in Toronto. Acad Psychiatry. 2010;34(4):277–281.
- , , . SNAPPS: a learner‐centered model for outpatient education. Acad Med. 2003;78(9):893–898.
- , , . Using SNAPPS to facilitate the expression of clinical reasoning and uncertainties: A randomized comparison group trial. Acad Med. 2009;84(4):517–524.
- , , , . Student uncertainties drive teaching during case presentations: more so with SNAPPS. Acad Med. 2012;87(9):1210–1217.
- , , , , , . SNAPPS‐plus: an educational prescription for students to facilitate formulating and answering clinical questions. Acad Med. 2014;89(8):1174–1179.
- . How attending physicians make instructional decisions when conducting teaching rounds. Acad Med. 1992;67(10):630–638.
- . Educational strategies to promote clinical diagnostic reasoning. N Engl J Med. 2006;355(21):2217–2225.
Acute Kidney Injury for Hospitalists
Acute kidney injury (AKI) is a clinical syndrome broadly defined as an abrupt decline in renal function occurring over a period of hours to days resulting in the retention of nitrogenous and metabolic waste products. Although the initial clinical manifestation of AKI may be oliguria, urine volume can remain normal or even increase. Patients may be asymptomatic, especially early in the course of AKI. The diagnosis is often made in hospitalized patients when biochemical screening reveals a recent increase in serum creatinine and/or blood urea nitrogen concentrations, or when there is a dramatic decrease in urine output.
Older studies looking at the incidence of AKI in hospitalized patients are difficult to interpret due to variable definitions of AKI. Those based on administrative databases were limited by lack of clinical context and/or variation in coding for AKI.[1]
There is no universally accepted operational definition of AKI, and more than 30 different criteria have been employed in various clinical studies. Difficulty in defining AKI lies in the lag time in the rise and fall of the serum creatinine concentration with injury and recovery, the variability of oliguria, and in the heterogeneity of patterns of renal injury. Two classification systems that attempt to capture the spectrum of AKI are the RIFLE (Risk, Injury, Failure, Loss, End Stage) criteria and the AKIN (Acute Kidney Injury Network) criteria.[2, 3] The AKIN criteria parallel the risk, injury, and failure stages of the RIFLE criteria and are the most applicable to characterizing AKI in the hospital (Table 1). AKI is commonly classified by daily urine output as anuric (50 mL/day), oliguric (500 mL/day), or nonoliguric.
| Stage | Creatinine Criteria | Urine Output Criteria |
|---|---|---|
| 1 | Increase in serum creatinine of 0.3 mg/dL (26.4 mol/L) or increase of 150%200% (1.5‐fold to 2‐fold) above baseline | 0.5 mL/kg/hr for >6 hours |
| 2 | Increase in serum creatinine of >200%300% (>2‐fold to 3‐fold) above baseline | 0.5 mL/kg/hr for >12 hours |
| 3 | Increase in serum creatinine of >300% (3‐fold) above baseline or serum creatinine 5.0 mg/dL (354 mol/L) with an acute rise of 0.5 mg/dL (44 mol/L) | 0.3 mL/kg/hr 24 hours or anuria 12 hours |
With a move toward standardized definitions, recent studies have shown a rising incidence of AKI in hospitalized patients.[4, 5, 6] According to these series, AKI develops in up to 7% of hospitalized patients and in about 30% of those admitted to intensive care units. In one study of consecutive hospital admissions, patients classified by the RIFLE criteria had a sharp rise in the rate of in‐hospital mortality whether they had no change or improvement in creatinine (4.4%), or fell into a risk (15.1%), injury (29.2%), or failure (41.1%) class.[7] The in‐hospital mortality of critically ill patients with AKI is higher than 50%. AKI increases length of stay and hospital costs, and affects the clinical course after discharge.[8, 9] Small increases in serum creatinine during an intensive care unit stay predict increased 10‐year mortality above a critical illness alone.[10]
Risk factors for AKI include advanced age, male gender, African American ethnicity, and diabetes mellitus.[11] The most important risk factor, however, is preexisting chronic kidney disease (CKD).[12] AKI and CKD are tightly linked, each increasing the risk of the other.[13, 14, 15] Preexisting renal insufficiency is a key predictor of postoperative AKI and poor surgical outcomes.[16, 17]
AKI AND CLINICAL CONTEXT
The causes of AKI can be broadly divided into 3 categories: prerenal azotemia (a disorder characterized by renal hypoperfusion in which renal parenchymal tissue integrity is preserved), intrinsic kidney injury with parenchymal tissue injury, and postrenal AKI (dysfunction due to acute obstruction of the urinary tract). Table 2 lists several clinical scenarios sorted into these 3 categories.[18] The general epidemiology of AKI varies based on whether it was acquired in the community or in a hospital setting. Prerenal azotemia accounts for the bulk of community‐acquired AKI, followed in lesser frequency by postrenal and intrinsic etiologies. Prerenal azotemia continues to be the major cause of hospital‐acquired AKI, but intrinsic kidney injury becomes more common.[5, 19]
| Prerenal | Intrinsic | Postrenal |
|---|---|---|
| ||
| Hemorrhage | Acute tubular necrosis | Bilateral upper tract obstruction |
| Surgical | Ischemic | Nephrolithiasis |
| Gastrointestinal | Postoperative | Papillary necrosis |
| Retroperitoneal | Prolonged hypotension | Retroperitoneal fibrosis |
| Gastrointestinal losses | Sepsis | Retroperitoneal lymphadenopathy |
| Diarrhea | Nephrotoxins | Obstruction of solitary functioning kidney |
| Vomiting | Myoglobin | Lower tract obstruction |
| Nasogastric suction | Hemoglobin | Prostatic hypertrophy |
| Enteral fistula | Radiocontrast agents | Urethral stricture |
| Renal losses | Aminoglycosides | Bladder mass or stone |
| Diuretics | Intratubular obstruction | Obstructed urinary catheter |
| Glucosuria | Tumor lysis/uric acid | Urinary retention |
| Skin losses | Oxalosis/ethylene glycol ingestion | Neurogenic bladder |
| Excessive sweating | Phosphate nephropathy | Constipation |
| Burns | Light chain nephropathy | Medications |
| Erythroderma | Acyclovir | Anticholinergics |
| Third‐spacing | Indinavir | Antihistamines |
| Hypoalbuminemia | Methotrexate | Alpha1‐agonists |
| Pancreatitis | Acute glomerulonephritis | ‐Blockers |
| Capillary leak | Acute interstitial nephritis | Opiates |
| Reduced effective arterial volume | Proton pump inhibitors | Tricyclic antidepressants |
| Congestive heart failure | Penicillins | |
| Cirrhosis | Fluoroquinolones | |
| Renal vasoconstriction | Atheroembolic disease | |
| Hypercalcemia | Acute vascular syndrome | |
| NSAIDs | Aortic dissection | |
| ACEI/ARB | Bilateral renal artery thromboembolism | |
| Calcineurin inhibitors | Bilateral renal vein thrombosis | |
| Vasopressors | Thrombotic microangiopathy | |
| Iodinated contrast | ||
MEDICAL HISTORY
The initial goal of history taking is to establish whether the patient has AKI rather than the acute discovery of a more chronic process. A recent serum creatinine measurement can be valuable in this regard. In some cases the clinician must make a presumptive diagnosis of AKI while simultaneously reviewing past medical history and family history to assess for underlying CKD. A diagnosis of AKI is more readily established when it occurs during a hospitalization through review of urine output and serial laboratory values.
Symptoms of poor oral intake as well as salt and fluid losses from diarrhea or vomiting suggest a prerenal etiology. Subjective symptoms of lightheadedness, visual clouding, and near‐syncope with standing also suggest volume depletion. Patients should be asked about recent nonsteroidal anti‐inflammatory drug (NSAID) use, as these agents can exacerbate renal hypoperfusion through loss of prostaglandin‐mediated afferent arteriole dilatation. Angiotensin‐converting enzyme inhibitors and angiotensin receptor blockers, especially when combined with diuretics, can generate a hypoperfusion state. Heart failure and liver disease regularly result in an expanded extracellular fluid (ECF) compartment yet a reduced effective arterial volume and predispose to renal hypoperfusion.
A history of decreased urine output or anuria suggests postrenal AKI, but its absence does not rule out urinary tract obstruction. Voiding symptoms such as urinary frequency, hesitancy, or incontinence also raise the possibility of obstructive uropathy. Flank pain and hematuria often accompany obstruction from nephrolithiasis.
Symptoms of fever, skin rash, arthralgias, sinusitis, and/or hemoptysis raise the possibility of glomerulonephritis from infection, collagen vascular disease, or vasculitis. Risk factors for viral hepatitis and human immunodeficiency virus are important to clarify as are systemic symptoms of autoimmunity (eg, dry eyes, dry mouth, eye pain/emnflammation, or visual changes). The recent start of any new medication, including NSAIDs, antibiotics, or proton‐pump inhibitors, raises the possibility of a drug‐induced interstitial nephritis.[20] Statins are direct myotoxins, and the risk of rhabdomyolysis with renal injury increases with dose. Patients may not associate intravenous (IV) contrast or phosphate‐containing bowel preparations (eg, Fleet Phospho Soda), with the development of AKI, thus the clinician must carefully review for recent exposures that could result in intrinsic renal injury.[21]
PHYSICAL EXAMINATION
Estimation of the ECF volume and effective arterial volume are central to assessing the likelihood of renal hypoperfusion. Overt hypotension is the strongest indicator of hypoperfusion, and a careful review of initial blood pressure prior to worsening of renal function can provide significant information. Normal blood pressure does not exclude renal hypoperfusion, as acute tubular necrosis (ATN) may develop in chronically hypertensive patients whose blood pressures are acutely reduced.[22] Less‐severe volume depletion is suggested by an orthostatic pulse increase of more than 30 beats/minute, measured 1 minute after standing. Orthostatic hypotension, defined as a drop in systolic pressure of more than 20 mm Hg after standing, is less helpful, as it occurs in 10% of normovolemic subjects.[23] Dry axillae and mucous membranes with a furrowed tongue are useful signs of volume depletion. Poor skin turgor and slow capillary refill have not been shown to be reliable signs of hypovolemia in adults. The neck veins are usually flat when volume contraction exists, though engorged neck veins in the setting of elevated right‐sided pressures from heart failure or pulmonary hypertension may obscure this sign. Similarly, pulmonary rales, ascites, and peripheral edema may confound the exam in patients with underlying heart failure and/or cirrhosis.
Flank tenderness or a bladder palpable or percussable above the pelvic brim suggests possible urinary tract obstruction. Prostate exam should be performed on all men with AKI and a bimanual pelvic exam considered in women with changes in usual voiding pattern or with suspected gynecologic disease. Postvoid residual can be assessed at the bedside with either straight catheterization or bladder scan where available.
Signs of systemic disease associated with intrinsic AKI include fever, skin and joint findings of connective tissue disease, a new or changing heart murmur, purpura, and petechiae. Cholesterol emboli, disrupted by interarterial catheterization (eg, cardiac catheterization, angiography), cardiac or aortic surgeries, or, rarely, by systemic anticoagulation can shower throughout the vasculature, causing organ dysfunction and local inflammation. Kidney injury due to atheroemboli often has a stuttering course and may be separated in time from the vascular procedure by days to weeks. Physical exam findings of atheroembolic disease include livedo reticularis, blue toes, purpura, painful skin nodules, and gangrene. Retinal examination may reveal atheroembolic emboli (Hollenhorst plaques).[24, 25]
LABORATORY TESTING
Initial testing in AKI aims to assess the severity of injury as well as the likely mechanism of the injury. Estimation of glomerular filtration rate (GFR) gives an approximate measure of the number of functioning nephrons and hence an overall measure of renal function. Mathematical estimates of GFR, however, assume a steady state, and AKI, by definition, is not a steady state. This makes GFR estimates based on plasma creatinine unreliable. A rising serum creatinine concentration indicates that the renal injury is persistent or worsening, whereas a stable or falling creatinine concentration suggests recovery. Interventions that expand the ECF (eg, volume resuscitation with normal saline) will dilute the plasma creatinine concentration and must be considered when interpreting a falling creatinine concentration. A daily rise in the serum creatinine concentration of more than 1 mg/dL nearly always implies a GFR of 10 mL/min. Any change in serum creatinine must be interpreted with the nonlinear relationship of GFR and serum creatinine in mind (Figure 1).[26]

The fractional excretion of sodium (FENa) has been used to differentiate prerenal azotemia from intrinsic renal injury in patients with oligoanuria. Specifically, an FENa of 1% implies a prerenal cause for the oliguric AKI, whereas if it is >1%, then intrinsic renal injury is more likely. Unfortunately, there are significant limitations to this laboratory measure.[27] The FENa may be low (1%) in any intrinsic process that causes tissue ischemia, such as vasculitis, acute glomerulonephritis, atheroembolic disease, or from intense vasoconstriction such as after IV contrast administration. Patients with severe heart failure or portal hypertension often have avid sodium retention, and can have a FENa 1% even in the setting of ATN. Alternatively, the FENa may be elevated (>1%) in prerenal patients on diuretics, with osmotic diuresis, or in the setting of aldosterone deficiency.
Examination of the urinalysis and urine sediment provides valuable information about the etiology of the AKI. Prerenal and postrenal AKI typically present with a bland urine, without evidence of blood, protein, or leukocyte esterase on urinalysis and few cells or hyaline casts in the sediment. The urinalysis typically has a high specific gravity in prerenal AKI, reflecting intact tubules producing a concentrated urine. An active urinary sediment suggests intrinsic renal injury that is either the mechanism of the current AKI or indicative of underlying CKD. ATN, the most common cause of intrinsic renal injury, often produces a dirty urinalysis with many epithelial cells and muddy brown granular and epithelial cell casts. The urine is generally isosthenuric (ie, specific gravity of 1.010) due to loss of tubular function. A urinalysis positive for heme pigment but without red cells on microscopic analysis suggests the presence of either myoglobin from rhabdomyolysis or hemoglobin from hemolysis. Acute glomerulonephritis disrupts the usual glomerular barrier to large proteins and red cells and results in proteinuria and hematuria. Red cells that weather the journey from the glomerulus through the nephron often become dysmorphic with Mickey Mouse ear blebs in their membrane or are bound together by Tamm‐Horsfall protein into red cell casts. Acute interstitial nephritis results in pyuria, proteinuria, and white cell casts. Urinary eosinophils are neither sensitive nor specific for interstitial nephritis and have little utility in its diagnosis.[28, 29]
Given the limitations of serum creatinine as a marker of renal injury, a number of new urinary biomarkers have been recognized over the past decade.[30, 31, 32] These molecules are normal constituents of renal tubular cells that are upregulated and released into the urine in response to renal injury. Early measurement of these biomarkers might allow for detection of AKI within hours of the insult. The 2 biomarkers with the most promise include kidney injury molecule‐1 (KIM‐1) and neutrophil gelatinase‐associated lipocalin (NGAL). KIM‐1 is expressed by proximal tubular cells, and its production is sharply upregulated in response to ischemic injury. NGAL is a protein expressed primarily in immune cells, but also by renal tubular cells. Urinary NGAL levels rapidly rise in response to renal ischemia, and return to baseline following resolution of the injury. Although these urinary biomarkers are promising, they have a relatively low (70%75%) sensitivity and specificity, and have not yet been adopted into routine clinical practice.[33]
IMAGING
Renal ultrasound is useful both in the assessment of AKI as well as in the investigation for underlying CKD. Patients with long‐standing kidney disease frequently have small, echogenic kidneys consistent with fibrosis and nephron loss, or markedly distorted renal architecture in cystic diseases. Hydronephrosis and/or hydroureter suggest an acute or chronic urinary tract obstruction. However, this may not be present in the setting of early obstruction or ureteric encasement. Doppler ultrasonography of the renal vasculature can assess patency when vascular obstruction is suspected. The use of computerized tomography, magnetic resonance imaging, or angiography may be helpful in selected clinical circumstances, but their use is often limited due to the potential risk of contrast nephrotoxicity. Nuclear renal scans use less radiation than computerized tomography and are a preferred imaging modality for pediatric patients. When volume status is uncertain, echocardiography to assess both inferior vena cava volume and change in volume with respiration may be helpful.
MANAGEMENT
The general principles for management of AKI are to limit further injury and prevent systemic complications. Management of the patient with AKI greatly depends on which category of AKI is suspected, namely prerenal, intrinsic renal injury, or a postrenal (obstructive) cause. If a prerenal etiology due to true ECF volume depletion is suspected, volume resuscitation to replace baseline and ongoing losses is imperative. Careful attention to intake and output as well as serial volume assessment should dictate the strategy for resuscitation. Hyperchloremic acidosis is an expected consequence of normal saline resuscitation but is irrelevant to clinical outcomes.[34] NSAIDs, antihypertensives, especially those that affect the angiotensin/aldosterone system, and diuretics should be discontinued. Ongoing hypotension despite volume resuscitation suggests the possibility of blood loss, infection, or autonomic nervous system dysfunction. If this occurs, the patient may need to be transferred to an intensive care unit for pressor support to keep the mean arterial pressure >70 mm Hg. When prerenal AKI from reduced effective circulating volume is suspected, as in decompensated heart failure or cirrhosis, management must be tailored to the underlying pathophysiology.
If judicious volume resuscitation produces no improvement in renal function or if oliguria develops, repeat urinalysis and urine microscopy should be considered to assess for intrinsic renal injury. Aggressive volume resuscitation in the face of oliguria will not speed recovery from the intrinsic injury and may cause signs or symptoms of volume overload. This could also potentially necessitate renal replacement therapy earlier than anticipated.
In patients where an obstructive etiology for the AKI is identified, the obstruction must be relieved as soon and as safely as possible. In this regard, a timely urologic consultation may be helpful in assuring that urethral and/or ureteral conduits are placed rapidly. Interventional radiology can also assist in those patients who need percutaneous nephrostomies for the relief of the obstruction. In many patients with obstructive nephropathy, a timely intervention will avoid the need for renal replacement therapy.
The suspected mechanism of injury influences the management of intrinsic AKI. The management of ATN is primarily supportive, paying close attention to optimizing volume status, correcting electrolyte abnormalities, avoiding further nephrotoxic agents, and adjusting medication doses to the low GFR present. Over the last several decades, multiple studies have explored treatment strategies for established ATN using various drugs and biologic agents. All have been uniformly disappointing.
When the trajectory of AKI is uncertain and the creatinine continues to rise, all medication dosing should be adjusted for GFR 10 mL/min. Antibiotics routinely will require dose reduction, but all current medications should be reviewed for risk of accumulation in renal failure. Because the half‐life of oral hypoglycemic medications is unpredictable in AKI, these medications should be discontinued and replaced with insulin. Vigilance for hypoglycemia is necessary, as renal clearance of insulin is also reduced. Narcotics such as morphine and oxycodone, which are renally cleared, can produce unwanted sedation and respiratory depression if not discontinued. Fentanyl, methadone, and hydromorphone are safer choices for controlling pain in a patient with AKI.[35] Gabapentin is regularly used to treat symptoms of neuropathic pain, but can produce encephalopathy and myoclonus if not dose reduced in renal failure.[36] Clinicians should weigh the risk of overdose with underdose for each medication, namely antibiotics in critically ill patients.
TIMING OF NEPHROLOGY CONSULTATION
The optimal timing for nephrology consultation in hospital‐acquired AKI is uncertain, though several studies have suggested better outcomes, including shorter length of stay and reduced mortality, with early consultation.[37, 38, 39] A renal consult is indicated when intrinsic ATN does not reverse in a timely fashion. Renal replacement therapy should be instituted to limit the systemic complications of prolonged AKI and to allow time for the renal injury to improve or resolve over time. If acute glomerulonephritis or interstitial nephritis is suspected, an urgent consultation may be required for consideration of biopsy, immunosuppression, and guidance for further management. Early consultation may help limit drug toxicities and volume overload in the setting of decreased renal clearance. Guidance on vascular access (eg, peripherally inserted central catheter placement) may prevent future complications with hemodialysis access if the patient ultimately develops end‐stage renal disease (ESRD).[40]
PREVENTION OF AKI
Most studies of AKI prevention have focused on clinical scenarios where the likelihood of ATN was substantial such as in vascular or open heart surgery, or with the use of intravenous contrast agents.[41, 42] This topic remains controversial, though generally supported strategies include judicious volume expansion, avoidance of hypotension, and, when using contrast, limiting the volume of contrast and using iso‐osmolar formulations. As recent studies have shown uncertain benefit, the role for pretreatment with n‐acetylcysteine remains uncertain. Many clinicians, however, continue to use it as a preventive strategy as there are few side effects with this medication.
TAKE HOME POINTS
- AKI is common in hospitalized patients, with pre‐renal azotemia being the dominant etiology in both community‐acquired and hospital‐acquired AKI.
- CKD is an important risk factor for AKI. AKI increases the long‐term risk of developing CKD and ESRD.
- The diagnosis of AKI hinges on detailed medical history, careful physical exam, and key laboratory parameters including the urinalysis and urinary sediment.
- The management of AKI is tailored to the likely mechanism of injury. Reconsideration of the likely etiology is imperative if AKI fails to respond to initial attempts to reverse or limit injury.
- Early renal consultation for AKI is indicated when the etiology remains uncertain, AKI persists despite initial management, or acute glomerulonephritis or interstitial nephritis are suspected.
- , , . Acute kidney injury. Lancet. 2012;380(9843):756–766.
- , , , , . Acute renal failure—definition, outcome measures, animal models, fluid therapy and information technology needs: the Second International Consensus Conference of the Acute Dialysis Quality Initiative (ADQI) Group. Crit Care. 2004;8(4):R204–R212.
- , , , et al. Improving outcomes from acute kidney injury: report of an initiative. Am J Kidney Dis. 2007;50(1):1–4.
- , , , et al. Incidence and mortality of acute renal failure in Medicare beneficiaries, 1992 to 2001. J Am Soc Nephrol. 2006;17(4):1135–1142.
- , , . Hospital‐acquired renal insufficiency. Am J Kidney Dis. 2002;39(5):930–936.
- , , , . Acute renal failure: factors influencing nephrology referral and outcome. QJM. 1997;90(12):781–785.
- , , , , . An assessment of the RIFLE criteria for acute renal failure in hospitalized patients. Crit Care Med. 2006;34(7):1913–1917.
- , , , , . Acute kidney injury, mortality, length of stay, and costs in hospitalized patients. J Am Soc Nephrol. 2005;16(11):3365–3370.
- , , , , . The prognostic importance of a small acute decrement in kidney function in hospitalized patients: a systematic review and meta‐analysis. Am J Kidney Dis. 2007;50(5):712–720.
- , , , , , . Small acute increases in serum creatinine are associated with decreased long‐term survival in the critically ill. Am J Respir Crit Care Med. 2014;189(9):1075–1081.
- , , , et al. Acute kidney injury increases risk of ESRD among elderly. J Am Soc Nephrol. 2009;20(1):223–228.
- , , , , , . The risk of acute renal failure in patients with chronic kidney disease. Kidney Int. 2008;74(1):101–107.
- , , . Chronic kidney disease after acute kidney injury: a systematic review and meta‐analysis. Kidney Int. 2012;81(5):442–448.
- , , , et al. Risk of chronic dialysis and death following acute kidney injury. Am J Med. 2012;125(6):585–593.
- , , , et al. Chronic dialysis and death among survivors of acute kidney injury requiring dialysis. JAMA. 2009;302(11):1179–1185.
- , , , et al. Development and validation of an acute kidney injury risk index for patients undergoing general surgery: results from a national data set. Anesthesiology. 2009;110(3):505–515.
- , , , et al. Preoperative estimates of glomerular filtration rate as predictors of outcome after surgery: a systematic review and meta‐analysis. Anesthesiology. 2013;118(4):809–824.
- , . Brenner 2008.
- , , , , . Hospital‐acquired renal insufficiency: a prospective study. Am J Med. 1983;74(2):243–248.
- , , , . A nationwide nested case‐control study indicates an increased risk of acute interstitial nephritis with proton pump inhibitor use. Kidney Int. 2014;86(4):837–844.
- , , , . Acute phosphate nephropathy following oral sodium phosphate bowel purgative: an underrecognized cause of chronic renal failure. J Am Soc Nephrol. 2005;16(11):3389–3396.
- . Normotensive ischemic acute renal failure. N Engl J Med. 2007;357(8):797–805.
- , , . The rational clinical examination. Is this patient hypovolemic? JAMA. 1999;281(11):1022–1029.
- , . Atheroembolic renal disease. Lancet. 2010;375(9726):1650–1660.
- , , , et al. The challenge of diagnosing atheroembolic renal disease: clinical features and prognostic factors. Circulation. 2007;116(3):298–304.
- , , , , , . A more accurate method to estimate glomerular filtration rate from serum creatinine: a new prediction equation. Modification of Diet in Renal Disease Study Group. Ann Intern Med. 1999;130(6):461–470.
- , . Traditional urinary biomarkers in the assessment of hospital‐acquired AKI. Clin J Am Soc Nephrol. 2012;7(1):167–174.
- , . Urinary eosinophils in AIN: farewell to an old biomarker? Clin J Am Soc Nephrol. 2013;8(11):1841–1843.
- , , . Utility of urine eosinophils in the diagnosis of acute interstitial nephritis. Clin J Am Soc Nephrol. 2013;8(11):1857–1862.
- . Diagnosis of acute kidney injury: from classic parameters to new biomarkers. Contrib Nephrol. 2007;156:213–219.
- , . Biomarkers in nephrology: Core Curriculum 2013. Am J Kidney Dis. 2013;62(1):165–178.
- , , , et al. Urinary biomarkers in the clinical prognosis and early detection of acute kidney injury. Clin J Am Soc Nephrol. 2010;5(12):2154–2165.
- , , , et al. Biomarkers for early diagnosis of AKI in the ICU: ready for prime time use at the bedside? Ann Intensive Care. 2012;2(1):24.
- , . The case for 0.9% NaCl: is the undefendable, defensible? Kidney Int. 2014;86(6):1087–1095.
- , , , , . A systematic review of the use of opioid medication for those with moderate to severe cancer pain and renal impairment: a European Palliative Care Research Collaborative opioid guidelines project. Palliat Med. 2011;25(5):525–552.
- , , . Gabapentin toxicity in patients with chronic kidney disease: a preventable cause of morbidity. Am J Med. 2010;123(4):367–373.
- , , , et al. Timing of initiation of dialysis in critically ill patients with acute kidney injury. Clin J Am Soc Nephrol. 2006;1(5):915–919.
- , , , et al. Nephrology consultation in acute renal failure: does timing matter? Am J Med. 2002;113(6):456–461.
- , , , et al. Early nephrologist involvement in hospital‐acquired acute kidney injury: a pilot study. Am J Kidney Dis. 2011;57(2):228–234.
- , , , et al. Association between prior peripherally inserted central catheters and lack of functioning arteriovenous fistulas: a case‐control study in hemodialysis patients. Am J Kidney Dis. 2012;60(4):601–608.
- , , , , , . Update on clinical trials for the prevention of acute kidney injury in patients undergoing cardiac surgery. Am J Surg. 2013;206(1):86–95.
- , , , et al. Strategies to reduce the risk of contrast‐induced nephropathy. Am J Cardiol. 2006;98(6A):59K–77K.
Acute kidney injury (AKI) is a clinical syndrome broadly defined as an abrupt decline in renal function occurring over a period of hours to days resulting in the retention of nitrogenous and metabolic waste products. Although the initial clinical manifestation of AKI may be oliguria, urine volume can remain normal or even increase. Patients may be asymptomatic, especially early in the course of AKI. The diagnosis is often made in hospitalized patients when biochemical screening reveals a recent increase in serum creatinine and/or blood urea nitrogen concentrations, or when there is a dramatic decrease in urine output.
Older studies looking at the incidence of AKI in hospitalized patients are difficult to interpret due to variable definitions of AKI. Those based on administrative databases were limited by lack of clinical context and/or variation in coding for AKI.[1]
There is no universally accepted operational definition of AKI, and more than 30 different criteria have been employed in various clinical studies. Difficulty in defining AKI lies in the lag time in the rise and fall of the serum creatinine concentration with injury and recovery, the variability of oliguria, and in the heterogeneity of patterns of renal injury. Two classification systems that attempt to capture the spectrum of AKI are the RIFLE (Risk, Injury, Failure, Loss, End Stage) criteria and the AKIN (Acute Kidney Injury Network) criteria.[2, 3] The AKIN criteria parallel the risk, injury, and failure stages of the RIFLE criteria and are the most applicable to characterizing AKI in the hospital (Table 1). AKI is commonly classified by daily urine output as anuric (50 mL/day), oliguric (500 mL/day), or nonoliguric.
| Stage | Creatinine Criteria | Urine Output Criteria |
|---|---|---|
| 1 | Increase in serum creatinine of 0.3 mg/dL (26.4 mol/L) or increase of 150%200% (1.5‐fold to 2‐fold) above baseline | 0.5 mL/kg/hr for >6 hours |
| 2 | Increase in serum creatinine of >200%300% (>2‐fold to 3‐fold) above baseline | 0.5 mL/kg/hr for >12 hours |
| 3 | Increase in serum creatinine of >300% (3‐fold) above baseline or serum creatinine 5.0 mg/dL (354 mol/L) with an acute rise of 0.5 mg/dL (44 mol/L) | 0.3 mL/kg/hr 24 hours or anuria 12 hours |
With a move toward standardized definitions, recent studies have shown a rising incidence of AKI in hospitalized patients.[4, 5, 6] According to these series, AKI develops in up to 7% of hospitalized patients and in about 30% of those admitted to intensive care units. In one study of consecutive hospital admissions, patients classified by the RIFLE criteria had a sharp rise in the rate of in‐hospital mortality whether they had no change or improvement in creatinine (4.4%), or fell into a risk (15.1%), injury (29.2%), or failure (41.1%) class.[7] The in‐hospital mortality of critically ill patients with AKI is higher than 50%. AKI increases length of stay and hospital costs, and affects the clinical course after discharge.[8, 9] Small increases in serum creatinine during an intensive care unit stay predict increased 10‐year mortality above a critical illness alone.[10]
Risk factors for AKI include advanced age, male gender, African American ethnicity, and diabetes mellitus.[11] The most important risk factor, however, is preexisting chronic kidney disease (CKD).[12] AKI and CKD are tightly linked, each increasing the risk of the other.[13, 14, 15] Preexisting renal insufficiency is a key predictor of postoperative AKI and poor surgical outcomes.[16, 17]
AKI AND CLINICAL CONTEXT
The causes of AKI can be broadly divided into 3 categories: prerenal azotemia (a disorder characterized by renal hypoperfusion in which renal parenchymal tissue integrity is preserved), intrinsic kidney injury with parenchymal tissue injury, and postrenal AKI (dysfunction due to acute obstruction of the urinary tract). Table 2 lists several clinical scenarios sorted into these 3 categories.[18] The general epidemiology of AKI varies based on whether it was acquired in the community or in a hospital setting. Prerenal azotemia accounts for the bulk of community‐acquired AKI, followed in lesser frequency by postrenal and intrinsic etiologies. Prerenal azotemia continues to be the major cause of hospital‐acquired AKI, but intrinsic kidney injury becomes more common.[5, 19]
| Prerenal | Intrinsic | Postrenal |
|---|---|---|
| ||
| Hemorrhage | Acute tubular necrosis | Bilateral upper tract obstruction |
| Surgical | Ischemic | Nephrolithiasis |
| Gastrointestinal | Postoperative | Papillary necrosis |
| Retroperitoneal | Prolonged hypotension | Retroperitoneal fibrosis |
| Gastrointestinal losses | Sepsis | Retroperitoneal lymphadenopathy |
| Diarrhea | Nephrotoxins | Obstruction of solitary functioning kidney |
| Vomiting | Myoglobin | Lower tract obstruction |
| Nasogastric suction | Hemoglobin | Prostatic hypertrophy |
| Enteral fistula | Radiocontrast agents | Urethral stricture |
| Renal losses | Aminoglycosides | Bladder mass or stone |
| Diuretics | Intratubular obstruction | Obstructed urinary catheter |
| Glucosuria | Tumor lysis/uric acid | Urinary retention |
| Skin losses | Oxalosis/ethylene glycol ingestion | Neurogenic bladder |
| Excessive sweating | Phosphate nephropathy | Constipation |
| Burns | Light chain nephropathy | Medications |
| Erythroderma | Acyclovir | Anticholinergics |
| Third‐spacing | Indinavir | Antihistamines |
| Hypoalbuminemia | Methotrexate | Alpha1‐agonists |
| Pancreatitis | Acute glomerulonephritis | ‐Blockers |
| Capillary leak | Acute interstitial nephritis | Opiates |
| Reduced effective arterial volume | Proton pump inhibitors | Tricyclic antidepressants |
| Congestive heart failure | Penicillins | |
| Cirrhosis | Fluoroquinolones | |
| Renal vasoconstriction | Atheroembolic disease | |
| Hypercalcemia | Acute vascular syndrome | |
| NSAIDs | Aortic dissection | |
| ACEI/ARB | Bilateral renal artery thromboembolism | |
| Calcineurin inhibitors | Bilateral renal vein thrombosis | |
| Vasopressors | Thrombotic microangiopathy | |
| Iodinated contrast | ||
MEDICAL HISTORY
The initial goal of history taking is to establish whether the patient has AKI rather than the acute discovery of a more chronic process. A recent serum creatinine measurement can be valuable in this regard. In some cases the clinician must make a presumptive diagnosis of AKI while simultaneously reviewing past medical history and family history to assess for underlying CKD. A diagnosis of AKI is more readily established when it occurs during a hospitalization through review of urine output and serial laboratory values.
Symptoms of poor oral intake as well as salt and fluid losses from diarrhea or vomiting suggest a prerenal etiology. Subjective symptoms of lightheadedness, visual clouding, and near‐syncope with standing also suggest volume depletion. Patients should be asked about recent nonsteroidal anti‐inflammatory drug (NSAID) use, as these agents can exacerbate renal hypoperfusion through loss of prostaglandin‐mediated afferent arteriole dilatation. Angiotensin‐converting enzyme inhibitors and angiotensin receptor blockers, especially when combined with diuretics, can generate a hypoperfusion state. Heart failure and liver disease regularly result in an expanded extracellular fluid (ECF) compartment yet a reduced effective arterial volume and predispose to renal hypoperfusion.
A history of decreased urine output or anuria suggests postrenal AKI, but its absence does not rule out urinary tract obstruction. Voiding symptoms such as urinary frequency, hesitancy, or incontinence also raise the possibility of obstructive uropathy. Flank pain and hematuria often accompany obstruction from nephrolithiasis.
Symptoms of fever, skin rash, arthralgias, sinusitis, and/or hemoptysis raise the possibility of glomerulonephritis from infection, collagen vascular disease, or vasculitis. Risk factors for viral hepatitis and human immunodeficiency virus are important to clarify as are systemic symptoms of autoimmunity (eg, dry eyes, dry mouth, eye pain/emnflammation, or visual changes). The recent start of any new medication, including NSAIDs, antibiotics, or proton‐pump inhibitors, raises the possibility of a drug‐induced interstitial nephritis.[20] Statins are direct myotoxins, and the risk of rhabdomyolysis with renal injury increases with dose. Patients may not associate intravenous (IV) contrast or phosphate‐containing bowel preparations (eg, Fleet Phospho Soda), with the development of AKI, thus the clinician must carefully review for recent exposures that could result in intrinsic renal injury.[21]
PHYSICAL EXAMINATION
Estimation of the ECF volume and effective arterial volume are central to assessing the likelihood of renal hypoperfusion. Overt hypotension is the strongest indicator of hypoperfusion, and a careful review of initial blood pressure prior to worsening of renal function can provide significant information. Normal blood pressure does not exclude renal hypoperfusion, as acute tubular necrosis (ATN) may develop in chronically hypertensive patients whose blood pressures are acutely reduced.[22] Less‐severe volume depletion is suggested by an orthostatic pulse increase of more than 30 beats/minute, measured 1 minute after standing. Orthostatic hypotension, defined as a drop in systolic pressure of more than 20 mm Hg after standing, is less helpful, as it occurs in 10% of normovolemic subjects.[23] Dry axillae and mucous membranes with a furrowed tongue are useful signs of volume depletion. Poor skin turgor and slow capillary refill have not been shown to be reliable signs of hypovolemia in adults. The neck veins are usually flat when volume contraction exists, though engorged neck veins in the setting of elevated right‐sided pressures from heart failure or pulmonary hypertension may obscure this sign. Similarly, pulmonary rales, ascites, and peripheral edema may confound the exam in patients with underlying heart failure and/or cirrhosis.
Flank tenderness or a bladder palpable or percussable above the pelvic brim suggests possible urinary tract obstruction. Prostate exam should be performed on all men with AKI and a bimanual pelvic exam considered in women with changes in usual voiding pattern or with suspected gynecologic disease. Postvoid residual can be assessed at the bedside with either straight catheterization or bladder scan where available.
Signs of systemic disease associated with intrinsic AKI include fever, skin and joint findings of connective tissue disease, a new or changing heart murmur, purpura, and petechiae. Cholesterol emboli, disrupted by interarterial catheterization (eg, cardiac catheterization, angiography), cardiac or aortic surgeries, or, rarely, by systemic anticoagulation can shower throughout the vasculature, causing organ dysfunction and local inflammation. Kidney injury due to atheroemboli often has a stuttering course and may be separated in time from the vascular procedure by days to weeks. Physical exam findings of atheroembolic disease include livedo reticularis, blue toes, purpura, painful skin nodules, and gangrene. Retinal examination may reveal atheroembolic emboli (Hollenhorst plaques).[24, 25]
LABORATORY TESTING
Initial testing in AKI aims to assess the severity of injury as well as the likely mechanism of the injury. Estimation of glomerular filtration rate (GFR) gives an approximate measure of the number of functioning nephrons and hence an overall measure of renal function. Mathematical estimates of GFR, however, assume a steady state, and AKI, by definition, is not a steady state. This makes GFR estimates based on plasma creatinine unreliable. A rising serum creatinine concentration indicates that the renal injury is persistent or worsening, whereas a stable or falling creatinine concentration suggests recovery. Interventions that expand the ECF (eg, volume resuscitation with normal saline) will dilute the plasma creatinine concentration and must be considered when interpreting a falling creatinine concentration. A daily rise in the serum creatinine concentration of more than 1 mg/dL nearly always implies a GFR of 10 mL/min. Any change in serum creatinine must be interpreted with the nonlinear relationship of GFR and serum creatinine in mind (Figure 1).[26]

The fractional excretion of sodium (FENa) has been used to differentiate prerenal azotemia from intrinsic renal injury in patients with oligoanuria. Specifically, an FENa of 1% implies a prerenal cause for the oliguric AKI, whereas if it is >1%, then intrinsic renal injury is more likely. Unfortunately, there are significant limitations to this laboratory measure.[27] The FENa may be low (1%) in any intrinsic process that causes tissue ischemia, such as vasculitis, acute glomerulonephritis, atheroembolic disease, or from intense vasoconstriction such as after IV contrast administration. Patients with severe heart failure or portal hypertension often have avid sodium retention, and can have a FENa 1% even in the setting of ATN. Alternatively, the FENa may be elevated (>1%) in prerenal patients on diuretics, with osmotic diuresis, or in the setting of aldosterone deficiency.
Examination of the urinalysis and urine sediment provides valuable information about the etiology of the AKI. Prerenal and postrenal AKI typically present with a bland urine, without evidence of blood, protein, or leukocyte esterase on urinalysis and few cells or hyaline casts in the sediment. The urinalysis typically has a high specific gravity in prerenal AKI, reflecting intact tubules producing a concentrated urine. An active urinary sediment suggests intrinsic renal injury that is either the mechanism of the current AKI or indicative of underlying CKD. ATN, the most common cause of intrinsic renal injury, often produces a dirty urinalysis with many epithelial cells and muddy brown granular and epithelial cell casts. The urine is generally isosthenuric (ie, specific gravity of 1.010) due to loss of tubular function. A urinalysis positive for heme pigment but without red cells on microscopic analysis suggests the presence of either myoglobin from rhabdomyolysis or hemoglobin from hemolysis. Acute glomerulonephritis disrupts the usual glomerular barrier to large proteins and red cells and results in proteinuria and hematuria. Red cells that weather the journey from the glomerulus through the nephron often become dysmorphic with Mickey Mouse ear blebs in their membrane or are bound together by Tamm‐Horsfall protein into red cell casts. Acute interstitial nephritis results in pyuria, proteinuria, and white cell casts. Urinary eosinophils are neither sensitive nor specific for interstitial nephritis and have little utility in its diagnosis.[28, 29]
Given the limitations of serum creatinine as a marker of renal injury, a number of new urinary biomarkers have been recognized over the past decade.[30, 31, 32] These molecules are normal constituents of renal tubular cells that are upregulated and released into the urine in response to renal injury. Early measurement of these biomarkers might allow for detection of AKI within hours of the insult. The 2 biomarkers with the most promise include kidney injury molecule‐1 (KIM‐1) and neutrophil gelatinase‐associated lipocalin (NGAL). KIM‐1 is expressed by proximal tubular cells, and its production is sharply upregulated in response to ischemic injury. NGAL is a protein expressed primarily in immune cells, but also by renal tubular cells. Urinary NGAL levels rapidly rise in response to renal ischemia, and return to baseline following resolution of the injury. Although these urinary biomarkers are promising, they have a relatively low (70%75%) sensitivity and specificity, and have not yet been adopted into routine clinical practice.[33]
IMAGING
Renal ultrasound is useful both in the assessment of AKI as well as in the investigation for underlying CKD. Patients with long‐standing kidney disease frequently have small, echogenic kidneys consistent with fibrosis and nephron loss, or markedly distorted renal architecture in cystic diseases. Hydronephrosis and/or hydroureter suggest an acute or chronic urinary tract obstruction. However, this may not be present in the setting of early obstruction or ureteric encasement. Doppler ultrasonography of the renal vasculature can assess patency when vascular obstruction is suspected. The use of computerized tomography, magnetic resonance imaging, or angiography may be helpful in selected clinical circumstances, but their use is often limited due to the potential risk of contrast nephrotoxicity. Nuclear renal scans use less radiation than computerized tomography and are a preferred imaging modality for pediatric patients. When volume status is uncertain, echocardiography to assess both inferior vena cava volume and change in volume with respiration may be helpful.
MANAGEMENT
The general principles for management of AKI are to limit further injury and prevent systemic complications. Management of the patient with AKI greatly depends on which category of AKI is suspected, namely prerenal, intrinsic renal injury, or a postrenal (obstructive) cause. If a prerenal etiology due to true ECF volume depletion is suspected, volume resuscitation to replace baseline and ongoing losses is imperative. Careful attention to intake and output as well as serial volume assessment should dictate the strategy for resuscitation. Hyperchloremic acidosis is an expected consequence of normal saline resuscitation but is irrelevant to clinical outcomes.[34] NSAIDs, antihypertensives, especially those that affect the angiotensin/aldosterone system, and diuretics should be discontinued. Ongoing hypotension despite volume resuscitation suggests the possibility of blood loss, infection, or autonomic nervous system dysfunction. If this occurs, the patient may need to be transferred to an intensive care unit for pressor support to keep the mean arterial pressure >70 mm Hg. When prerenal AKI from reduced effective circulating volume is suspected, as in decompensated heart failure or cirrhosis, management must be tailored to the underlying pathophysiology.
If judicious volume resuscitation produces no improvement in renal function or if oliguria develops, repeat urinalysis and urine microscopy should be considered to assess for intrinsic renal injury. Aggressive volume resuscitation in the face of oliguria will not speed recovery from the intrinsic injury and may cause signs or symptoms of volume overload. This could also potentially necessitate renal replacement therapy earlier than anticipated.
In patients where an obstructive etiology for the AKI is identified, the obstruction must be relieved as soon and as safely as possible. In this regard, a timely urologic consultation may be helpful in assuring that urethral and/or ureteral conduits are placed rapidly. Interventional radiology can also assist in those patients who need percutaneous nephrostomies for the relief of the obstruction. In many patients with obstructive nephropathy, a timely intervention will avoid the need for renal replacement therapy.
The suspected mechanism of injury influences the management of intrinsic AKI. The management of ATN is primarily supportive, paying close attention to optimizing volume status, correcting electrolyte abnormalities, avoiding further nephrotoxic agents, and adjusting medication doses to the low GFR present. Over the last several decades, multiple studies have explored treatment strategies for established ATN using various drugs and biologic agents. All have been uniformly disappointing.
When the trajectory of AKI is uncertain and the creatinine continues to rise, all medication dosing should be adjusted for GFR 10 mL/min. Antibiotics routinely will require dose reduction, but all current medications should be reviewed for risk of accumulation in renal failure. Because the half‐life of oral hypoglycemic medications is unpredictable in AKI, these medications should be discontinued and replaced with insulin. Vigilance for hypoglycemia is necessary, as renal clearance of insulin is also reduced. Narcotics such as morphine and oxycodone, which are renally cleared, can produce unwanted sedation and respiratory depression if not discontinued. Fentanyl, methadone, and hydromorphone are safer choices for controlling pain in a patient with AKI.[35] Gabapentin is regularly used to treat symptoms of neuropathic pain, but can produce encephalopathy and myoclonus if not dose reduced in renal failure.[36] Clinicians should weigh the risk of overdose with underdose for each medication, namely antibiotics in critically ill patients.
TIMING OF NEPHROLOGY CONSULTATION
The optimal timing for nephrology consultation in hospital‐acquired AKI is uncertain, though several studies have suggested better outcomes, including shorter length of stay and reduced mortality, with early consultation.[37, 38, 39] A renal consult is indicated when intrinsic ATN does not reverse in a timely fashion. Renal replacement therapy should be instituted to limit the systemic complications of prolonged AKI and to allow time for the renal injury to improve or resolve over time. If acute glomerulonephritis or interstitial nephritis is suspected, an urgent consultation may be required for consideration of biopsy, immunosuppression, and guidance for further management. Early consultation may help limit drug toxicities and volume overload in the setting of decreased renal clearance. Guidance on vascular access (eg, peripherally inserted central catheter placement) may prevent future complications with hemodialysis access if the patient ultimately develops end‐stage renal disease (ESRD).[40]
PREVENTION OF AKI
Most studies of AKI prevention have focused on clinical scenarios where the likelihood of ATN was substantial such as in vascular or open heart surgery, or with the use of intravenous contrast agents.[41, 42] This topic remains controversial, though generally supported strategies include judicious volume expansion, avoidance of hypotension, and, when using contrast, limiting the volume of contrast and using iso‐osmolar formulations. As recent studies have shown uncertain benefit, the role for pretreatment with n‐acetylcysteine remains uncertain. Many clinicians, however, continue to use it as a preventive strategy as there are few side effects with this medication.
TAKE HOME POINTS
- AKI is common in hospitalized patients, with pre‐renal azotemia being the dominant etiology in both community‐acquired and hospital‐acquired AKI.
- CKD is an important risk factor for AKI. AKI increases the long‐term risk of developing CKD and ESRD.
- The diagnosis of AKI hinges on detailed medical history, careful physical exam, and key laboratory parameters including the urinalysis and urinary sediment.
- The management of AKI is tailored to the likely mechanism of injury. Reconsideration of the likely etiology is imperative if AKI fails to respond to initial attempts to reverse or limit injury.
- Early renal consultation for AKI is indicated when the etiology remains uncertain, AKI persists despite initial management, or acute glomerulonephritis or interstitial nephritis are suspected.
Acute kidney injury (AKI) is a clinical syndrome broadly defined as an abrupt decline in renal function occurring over a period of hours to days resulting in the retention of nitrogenous and metabolic waste products. Although the initial clinical manifestation of AKI may be oliguria, urine volume can remain normal or even increase. Patients may be asymptomatic, especially early in the course of AKI. The diagnosis is often made in hospitalized patients when biochemical screening reveals a recent increase in serum creatinine and/or blood urea nitrogen concentrations, or when there is a dramatic decrease in urine output.
Older studies looking at the incidence of AKI in hospitalized patients are difficult to interpret due to variable definitions of AKI. Those based on administrative databases were limited by lack of clinical context and/or variation in coding for AKI.[1]
There is no universally accepted operational definition of AKI, and more than 30 different criteria have been employed in various clinical studies. Difficulty in defining AKI lies in the lag time in the rise and fall of the serum creatinine concentration with injury and recovery, the variability of oliguria, and in the heterogeneity of patterns of renal injury. Two classification systems that attempt to capture the spectrum of AKI are the RIFLE (Risk, Injury, Failure, Loss, End Stage) criteria and the AKIN (Acute Kidney Injury Network) criteria.[2, 3] The AKIN criteria parallel the risk, injury, and failure stages of the RIFLE criteria and are the most applicable to characterizing AKI in the hospital (Table 1). AKI is commonly classified by daily urine output as anuric (50 mL/day), oliguric (500 mL/day), or nonoliguric.
| Stage | Creatinine Criteria | Urine Output Criteria |
|---|---|---|
| 1 | Increase in serum creatinine of 0.3 mg/dL (26.4 mol/L) or increase of 150%200% (1.5‐fold to 2‐fold) above baseline | 0.5 mL/kg/hr for >6 hours |
| 2 | Increase in serum creatinine of >200%300% (>2‐fold to 3‐fold) above baseline | 0.5 mL/kg/hr for >12 hours |
| 3 | Increase in serum creatinine of >300% (3‐fold) above baseline or serum creatinine 5.0 mg/dL (354 mol/L) with an acute rise of 0.5 mg/dL (44 mol/L) | 0.3 mL/kg/hr 24 hours or anuria 12 hours |
With a move toward standardized definitions, recent studies have shown a rising incidence of AKI in hospitalized patients.[4, 5, 6] According to these series, AKI develops in up to 7% of hospitalized patients and in about 30% of those admitted to intensive care units. In one study of consecutive hospital admissions, patients classified by the RIFLE criteria had a sharp rise in the rate of in‐hospital mortality whether they had no change or improvement in creatinine (4.4%), or fell into a risk (15.1%), injury (29.2%), or failure (41.1%) class.[7] The in‐hospital mortality of critically ill patients with AKI is higher than 50%. AKI increases length of stay and hospital costs, and affects the clinical course after discharge.[8, 9] Small increases in serum creatinine during an intensive care unit stay predict increased 10‐year mortality above a critical illness alone.[10]
Risk factors for AKI include advanced age, male gender, African American ethnicity, and diabetes mellitus.[11] The most important risk factor, however, is preexisting chronic kidney disease (CKD).[12] AKI and CKD are tightly linked, each increasing the risk of the other.[13, 14, 15] Preexisting renal insufficiency is a key predictor of postoperative AKI and poor surgical outcomes.[16, 17]
AKI AND CLINICAL CONTEXT
The causes of AKI can be broadly divided into 3 categories: prerenal azotemia (a disorder characterized by renal hypoperfusion in which renal parenchymal tissue integrity is preserved), intrinsic kidney injury with parenchymal tissue injury, and postrenal AKI (dysfunction due to acute obstruction of the urinary tract). Table 2 lists several clinical scenarios sorted into these 3 categories.[18] The general epidemiology of AKI varies based on whether it was acquired in the community or in a hospital setting. Prerenal azotemia accounts for the bulk of community‐acquired AKI, followed in lesser frequency by postrenal and intrinsic etiologies. Prerenal azotemia continues to be the major cause of hospital‐acquired AKI, but intrinsic kidney injury becomes more common.[5, 19]
| Prerenal | Intrinsic | Postrenal |
|---|---|---|
| ||
| Hemorrhage | Acute tubular necrosis | Bilateral upper tract obstruction |
| Surgical | Ischemic | Nephrolithiasis |
| Gastrointestinal | Postoperative | Papillary necrosis |
| Retroperitoneal | Prolonged hypotension | Retroperitoneal fibrosis |
| Gastrointestinal losses | Sepsis | Retroperitoneal lymphadenopathy |
| Diarrhea | Nephrotoxins | Obstruction of solitary functioning kidney |
| Vomiting | Myoglobin | Lower tract obstruction |
| Nasogastric suction | Hemoglobin | Prostatic hypertrophy |
| Enteral fistula | Radiocontrast agents | Urethral stricture |
| Renal losses | Aminoglycosides | Bladder mass or stone |
| Diuretics | Intratubular obstruction | Obstructed urinary catheter |
| Glucosuria | Tumor lysis/uric acid | Urinary retention |
| Skin losses | Oxalosis/ethylene glycol ingestion | Neurogenic bladder |
| Excessive sweating | Phosphate nephropathy | Constipation |
| Burns | Light chain nephropathy | Medications |
| Erythroderma | Acyclovir | Anticholinergics |
| Third‐spacing | Indinavir | Antihistamines |
| Hypoalbuminemia | Methotrexate | Alpha1‐agonists |
| Pancreatitis | Acute glomerulonephritis | ‐Blockers |
| Capillary leak | Acute interstitial nephritis | Opiates |
| Reduced effective arterial volume | Proton pump inhibitors | Tricyclic antidepressants |
| Congestive heart failure | Penicillins | |
| Cirrhosis | Fluoroquinolones | |
| Renal vasoconstriction | Atheroembolic disease | |
| Hypercalcemia | Acute vascular syndrome | |
| NSAIDs | Aortic dissection | |
| ACEI/ARB | Bilateral renal artery thromboembolism | |
| Calcineurin inhibitors | Bilateral renal vein thrombosis | |
| Vasopressors | Thrombotic microangiopathy | |
| Iodinated contrast | ||
MEDICAL HISTORY
The initial goal of history taking is to establish whether the patient has AKI rather than the acute discovery of a more chronic process. A recent serum creatinine measurement can be valuable in this regard. In some cases the clinician must make a presumptive diagnosis of AKI while simultaneously reviewing past medical history and family history to assess for underlying CKD. A diagnosis of AKI is more readily established when it occurs during a hospitalization through review of urine output and serial laboratory values.
Symptoms of poor oral intake as well as salt and fluid losses from diarrhea or vomiting suggest a prerenal etiology. Subjective symptoms of lightheadedness, visual clouding, and near‐syncope with standing also suggest volume depletion. Patients should be asked about recent nonsteroidal anti‐inflammatory drug (NSAID) use, as these agents can exacerbate renal hypoperfusion through loss of prostaglandin‐mediated afferent arteriole dilatation. Angiotensin‐converting enzyme inhibitors and angiotensin receptor blockers, especially when combined with diuretics, can generate a hypoperfusion state. Heart failure and liver disease regularly result in an expanded extracellular fluid (ECF) compartment yet a reduced effective arterial volume and predispose to renal hypoperfusion.
A history of decreased urine output or anuria suggests postrenal AKI, but its absence does not rule out urinary tract obstruction. Voiding symptoms such as urinary frequency, hesitancy, or incontinence also raise the possibility of obstructive uropathy. Flank pain and hematuria often accompany obstruction from nephrolithiasis.
Symptoms of fever, skin rash, arthralgias, sinusitis, and/or hemoptysis raise the possibility of glomerulonephritis from infection, collagen vascular disease, or vasculitis. Risk factors for viral hepatitis and human immunodeficiency virus are important to clarify as are systemic symptoms of autoimmunity (eg, dry eyes, dry mouth, eye pain/emnflammation, or visual changes). The recent start of any new medication, including NSAIDs, antibiotics, or proton‐pump inhibitors, raises the possibility of a drug‐induced interstitial nephritis.[20] Statins are direct myotoxins, and the risk of rhabdomyolysis with renal injury increases with dose. Patients may not associate intravenous (IV) contrast or phosphate‐containing bowel preparations (eg, Fleet Phospho Soda), with the development of AKI, thus the clinician must carefully review for recent exposures that could result in intrinsic renal injury.[21]
PHYSICAL EXAMINATION
Estimation of the ECF volume and effective arterial volume are central to assessing the likelihood of renal hypoperfusion. Overt hypotension is the strongest indicator of hypoperfusion, and a careful review of initial blood pressure prior to worsening of renal function can provide significant information. Normal blood pressure does not exclude renal hypoperfusion, as acute tubular necrosis (ATN) may develop in chronically hypertensive patients whose blood pressures are acutely reduced.[22] Less‐severe volume depletion is suggested by an orthostatic pulse increase of more than 30 beats/minute, measured 1 minute after standing. Orthostatic hypotension, defined as a drop in systolic pressure of more than 20 mm Hg after standing, is less helpful, as it occurs in 10% of normovolemic subjects.[23] Dry axillae and mucous membranes with a furrowed tongue are useful signs of volume depletion. Poor skin turgor and slow capillary refill have not been shown to be reliable signs of hypovolemia in adults. The neck veins are usually flat when volume contraction exists, though engorged neck veins in the setting of elevated right‐sided pressures from heart failure or pulmonary hypertension may obscure this sign. Similarly, pulmonary rales, ascites, and peripheral edema may confound the exam in patients with underlying heart failure and/or cirrhosis.
Flank tenderness or a bladder palpable or percussable above the pelvic brim suggests possible urinary tract obstruction. Prostate exam should be performed on all men with AKI and a bimanual pelvic exam considered in women with changes in usual voiding pattern or with suspected gynecologic disease. Postvoid residual can be assessed at the bedside with either straight catheterization or bladder scan where available.
Signs of systemic disease associated with intrinsic AKI include fever, skin and joint findings of connective tissue disease, a new or changing heart murmur, purpura, and petechiae. Cholesterol emboli, disrupted by interarterial catheterization (eg, cardiac catheterization, angiography), cardiac or aortic surgeries, or, rarely, by systemic anticoagulation can shower throughout the vasculature, causing organ dysfunction and local inflammation. Kidney injury due to atheroemboli often has a stuttering course and may be separated in time from the vascular procedure by days to weeks. Physical exam findings of atheroembolic disease include livedo reticularis, blue toes, purpura, painful skin nodules, and gangrene. Retinal examination may reveal atheroembolic emboli (Hollenhorst plaques).[24, 25]
LABORATORY TESTING
Initial testing in AKI aims to assess the severity of injury as well as the likely mechanism of the injury. Estimation of glomerular filtration rate (GFR) gives an approximate measure of the number of functioning nephrons and hence an overall measure of renal function. Mathematical estimates of GFR, however, assume a steady state, and AKI, by definition, is not a steady state. This makes GFR estimates based on plasma creatinine unreliable. A rising serum creatinine concentration indicates that the renal injury is persistent or worsening, whereas a stable or falling creatinine concentration suggests recovery. Interventions that expand the ECF (eg, volume resuscitation with normal saline) will dilute the plasma creatinine concentration and must be considered when interpreting a falling creatinine concentration. A daily rise in the serum creatinine concentration of more than 1 mg/dL nearly always implies a GFR of 10 mL/min. Any change in serum creatinine must be interpreted with the nonlinear relationship of GFR and serum creatinine in mind (Figure 1).[26]

The fractional excretion of sodium (FENa) has been used to differentiate prerenal azotemia from intrinsic renal injury in patients with oligoanuria. Specifically, an FENa of 1% implies a prerenal cause for the oliguric AKI, whereas if it is >1%, then intrinsic renal injury is more likely. Unfortunately, there are significant limitations to this laboratory measure.[27] The FENa may be low (1%) in any intrinsic process that causes tissue ischemia, such as vasculitis, acute glomerulonephritis, atheroembolic disease, or from intense vasoconstriction such as after IV contrast administration. Patients with severe heart failure or portal hypertension often have avid sodium retention, and can have a FENa 1% even in the setting of ATN. Alternatively, the FENa may be elevated (>1%) in prerenal patients on diuretics, with osmotic diuresis, or in the setting of aldosterone deficiency.
Examination of the urinalysis and urine sediment provides valuable information about the etiology of the AKI. Prerenal and postrenal AKI typically present with a bland urine, without evidence of blood, protein, or leukocyte esterase on urinalysis and few cells or hyaline casts in the sediment. The urinalysis typically has a high specific gravity in prerenal AKI, reflecting intact tubules producing a concentrated urine. An active urinary sediment suggests intrinsic renal injury that is either the mechanism of the current AKI or indicative of underlying CKD. ATN, the most common cause of intrinsic renal injury, often produces a dirty urinalysis with many epithelial cells and muddy brown granular and epithelial cell casts. The urine is generally isosthenuric (ie, specific gravity of 1.010) due to loss of tubular function. A urinalysis positive for heme pigment but without red cells on microscopic analysis suggests the presence of either myoglobin from rhabdomyolysis or hemoglobin from hemolysis. Acute glomerulonephritis disrupts the usual glomerular barrier to large proteins and red cells and results in proteinuria and hematuria. Red cells that weather the journey from the glomerulus through the nephron often become dysmorphic with Mickey Mouse ear blebs in their membrane or are bound together by Tamm‐Horsfall protein into red cell casts. Acute interstitial nephritis results in pyuria, proteinuria, and white cell casts. Urinary eosinophils are neither sensitive nor specific for interstitial nephritis and have little utility in its diagnosis.[28, 29]
Given the limitations of serum creatinine as a marker of renal injury, a number of new urinary biomarkers have been recognized over the past decade.[30, 31, 32] These molecules are normal constituents of renal tubular cells that are upregulated and released into the urine in response to renal injury. Early measurement of these biomarkers might allow for detection of AKI within hours of the insult. The 2 biomarkers with the most promise include kidney injury molecule‐1 (KIM‐1) and neutrophil gelatinase‐associated lipocalin (NGAL). KIM‐1 is expressed by proximal tubular cells, and its production is sharply upregulated in response to ischemic injury. NGAL is a protein expressed primarily in immune cells, but also by renal tubular cells. Urinary NGAL levels rapidly rise in response to renal ischemia, and return to baseline following resolution of the injury. Although these urinary biomarkers are promising, they have a relatively low (70%75%) sensitivity and specificity, and have not yet been adopted into routine clinical practice.[33]
IMAGING
Renal ultrasound is useful both in the assessment of AKI as well as in the investigation for underlying CKD. Patients with long‐standing kidney disease frequently have small, echogenic kidneys consistent with fibrosis and nephron loss, or markedly distorted renal architecture in cystic diseases. Hydronephrosis and/or hydroureter suggest an acute or chronic urinary tract obstruction. However, this may not be present in the setting of early obstruction or ureteric encasement. Doppler ultrasonography of the renal vasculature can assess patency when vascular obstruction is suspected. The use of computerized tomography, magnetic resonance imaging, or angiography may be helpful in selected clinical circumstances, but their use is often limited due to the potential risk of contrast nephrotoxicity. Nuclear renal scans use less radiation than computerized tomography and are a preferred imaging modality for pediatric patients. When volume status is uncertain, echocardiography to assess both inferior vena cava volume and change in volume with respiration may be helpful.
MANAGEMENT
The general principles for management of AKI are to limit further injury and prevent systemic complications. Management of the patient with AKI greatly depends on which category of AKI is suspected, namely prerenal, intrinsic renal injury, or a postrenal (obstructive) cause. If a prerenal etiology due to true ECF volume depletion is suspected, volume resuscitation to replace baseline and ongoing losses is imperative. Careful attention to intake and output as well as serial volume assessment should dictate the strategy for resuscitation. Hyperchloremic acidosis is an expected consequence of normal saline resuscitation but is irrelevant to clinical outcomes.[34] NSAIDs, antihypertensives, especially those that affect the angiotensin/aldosterone system, and diuretics should be discontinued. Ongoing hypotension despite volume resuscitation suggests the possibility of blood loss, infection, or autonomic nervous system dysfunction. If this occurs, the patient may need to be transferred to an intensive care unit for pressor support to keep the mean arterial pressure >70 mm Hg. When prerenal AKI from reduced effective circulating volume is suspected, as in decompensated heart failure or cirrhosis, management must be tailored to the underlying pathophysiology.
If judicious volume resuscitation produces no improvement in renal function or if oliguria develops, repeat urinalysis and urine microscopy should be considered to assess for intrinsic renal injury. Aggressive volume resuscitation in the face of oliguria will not speed recovery from the intrinsic injury and may cause signs or symptoms of volume overload. This could also potentially necessitate renal replacement therapy earlier than anticipated.
In patients where an obstructive etiology for the AKI is identified, the obstruction must be relieved as soon and as safely as possible. In this regard, a timely urologic consultation may be helpful in assuring that urethral and/or ureteral conduits are placed rapidly. Interventional radiology can also assist in those patients who need percutaneous nephrostomies for the relief of the obstruction. In many patients with obstructive nephropathy, a timely intervention will avoid the need for renal replacement therapy.
The suspected mechanism of injury influences the management of intrinsic AKI. The management of ATN is primarily supportive, paying close attention to optimizing volume status, correcting electrolyte abnormalities, avoiding further nephrotoxic agents, and adjusting medication doses to the low GFR present. Over the last several decades, multiple studies have explored treatment strategies for established ATN using various drugs and biologic agents. All have been uniformly disappointing.
When the trajectory of AKI is uncertain and the creatinine continues to rise, all medication dosing should be adjusted for GFR 10 mL/min. Antibiotics routinely will require dose reduction, but all current medications should be reviewed for risk of accumulation in renal failure. Because the half‐life of oral hypoglycemic medications is unpredictable in AKI, these medications should be discontinued and replaced with insulin. Vigilance for hypoglycemia is necessary, as renal clearance of insulin is also reduced. Narcotics such as morphine and oxycodone, which are renally cleared, can produce unwanted sedation and respiratory depression if not discontinued. Fentanyl, methadone, and hydromorphone are safer choices for controlling pain in a patient with AKI.[35] Gabapentin is regularly used to treat symptoms of neuropathic pain, but can produce encephalopathy and myoclonus if not dose reduced in renal failure.[36] Clinicians should weigh the risk of overdose with underdose for each medication, namely antibiotics in critically ill patients.
TIMING OF NEPHROLOGY CONSULTATION
The optimal timing for nephrology consultation in hospital‐acquired AKI is uncertain, though several studies have suggested better outcomes, including shorter length of stay and reduced mortality, with early consultation.[37, 38, 39] A renal consult is indicated when intrinsic ATN does not reverse in a timely fashion. Renal replacement therapy should be instituted to limit the systemic complications of prolonged AKI and to allow time for the renal injury to improve or resolve over time. If acute glomerulonephritis or interstitial nephritis is suspected, an urgent consultation may be required for consideration of biopsy, immunosuppression, and guidance for further management. Early consultation may help limit drug toxicities and volume overload in the setting of decreased renal clearance. Guidance on vascular access (eg, peripherally inserted central catheter placement) may prevent future complications with hemodialysis access if the patient ultimately develops end‐stage renal disease (ESRD).[40]
PREVENTION OF AKI
Most studies of AKI prevention have focused on clinical scenarios where the likelihood of ATN was substantial such as in vascular or open heart surgery, or with the use of intravenous contrast agents.[41, 42] This topic remains controversial, though generally supported strategies include judicious volume expansion, avoidance of hypotension, and, when using contrast, limiting the volume of contrast and using iso‐osmolar formulations. As recent studies have shown uncertain benefit, the role for pretreatment with n‐acetylcysteine remains uncertain. Many clinicians, however, continue to use it as a preventive strategy as there are few side effects with this medication.
TAKE HOME POINTS
- AKI is common in hospitalized patients, with pre‐renal azotemia being the dominant etiology in both community‐acquired and hospital‐acquired AKI.
- CKD is an important risk factor for AKI. AKI increases the long‐term risk of developing CKD and ESRD.
- The diagnosis of AKI hinges on detailed medical history, careful physical exam, and key laboratory parameters including the urinalysis and urinary sediment.
- The management of AKI is tailored to the likely mechanism of injury. Reconsideration of the likely etiology is imperative if AKI fails to respond to initial attempts to reverse or limit injury.
- Early renal consultation for AKI is indicated when the etiology remains uncertain, AKI persists despite initial management, or acute glomerulonephritis or interstitial nephritis are suspected.
- , , . Acute kidney injury. Lancet. 2012;380(9843):756–766.
- , , , , . Acute renal failure—definition, outcome measures, animal models, fluid therapy and information technology needs: the Second International Consensus Conference of the Acute Dialysis Quality Initiative (ADQI) Group. Crit Care. 2004;8(4):R204–R212.
- , , , et al. Improving outcomes from acute kidney injury: report of an initiative. Am J Kidney Dis. 2007;50(1):1–4.
- , , , et al. Incidence and mortality of acute renal failure in Medicare beneficiaries, 1992 to 2001. J Am Soc Nephrol. 2006;17(4):1135–1142.
- , , . Hospital‐acquired renal insufficiency. Am J Kidney Dis. 2002;39(5):930–936.
- , , , . Acute renal failure: factors influencing nephrology referral and outcome. QJM. 1997;90(12):781–785.
- , , , , . An assessment of the RIFLE criteria for acute renal failure in hospitalized patients. Crit Care Med. 2006;34(7):1913–1917.
- , , , , . Acute kidney injury, mortality, length of stay, and costs in hospitalized patients. J Am Soc Nephrol. 2005;16(11):3365–3370.
- , , , , . The prognostic importance of a small acute decrement in kidney function in hospitalized patients: a systematic review and meta‐analysis. Am J Kidney Dis. 2007;50(5):712–720.
- , , , , , . Small acute increases in serum creatinine are associated with decreased long‐term survival in the critically ill. Am J Respir Crit Care Med. 2014;189(9):1075–1081.
- , , , et al. Acute kidney injury increases risk of ESRD among elderly. J Am Soc Nephrol. 2009;20(1):223–228.
- , , , , , . The risk of acute renal failure in patients with chronic kidney disease. Kidney Int. 2008;74(1):101–107.
- , , . Chronic kidney disease after acute kidney injury: a systematic review and meta‐analysis. Kidney Int. 2012;81(5):442–448.
- , , , et al. Risk of chronic dialysis and death following acute kidney injury. Am J Med. 2012;125(6):585–593.
- , , , et al. Chronic dialysis and death among survivors of acute kidney injury requiring dialysis. JAMA. 2009;302(11):1179–1185.
- , , , et al. Development and validation of an acute kidney injury risk index for patients undergoing general surgery: results from a national data set. Anesthesiology. 2009;110(3):505–515.
- , , , et al. Preoperative estimates of glomerular filtration rate as predictors of outcome after surgery: a systematic review and meta‐analysis. Anesthesiology. 2013;118(4):809–824.
- , . Brenner 2008.
- , , , , . Hospital‐acquired renal insufficiency: a prospective study. Am J Med. 1983;74(2):243–248.
- , , , . A nationwide nested case‐control study indicates an increased risk of acute interstitial nephritis with proton pump inhibitor use. Kidney Int. 2014;86(4):837–844.
- , , , . Acute phosphate nephropathy following oral sodium phosphate bowel purgative: an underrecognized cause of chronic renal failure. J Am Soc Nephrol. 2005;16(11):3389–3396.
- . Normotensive ischemic acute renal failure. N Engl J Med. 2007;357(8):797–805.
- , , . The rational clinical examination. Is this patient hypovolemic? JAMA. 1999;281(11):1022–1029.
- , . Atheroembolic renal disease. Lancet. 2010;375(9726):1650–1660.
- , , , et al. The challenge of diagnosing atheroembolic renal disease: clinical features and prognostic factors. Circulation. 2007;116(3):298–304.
- , , , , , . A more accurate method to estimate glomerular filtration rate from serum creatinine: a new prediction equation. Modification of Diet in Renal Disease Study Group. Ann Intern Med. 1999;130(6):461–470.
- , . Traditional urinary biomarkers in the assessment of hospital‐acquired AKI. Clin J Am Soc Nephrol. 2012;7(1):167–174.
- , . Urinary eosinophils in AIN: farewell to an old biomarker? Clin J Am Soc Nephrol. 2013;8(11):1841–1843.
- , , . Utility of urine eosinophils in the diagnosis of acute interstitial nephritis. Clin J Am Soc Nephrol. 2013;8(11):1857–1862.
- . Diagnosis of acute kidney injury: from classic parameters to new biomarkers. Contrib Nephrol. 2007;156:213–219.
- , . Biomarkers in nephrology: Core Curriculum 2013. Am J Kidney Dis. 2013;62(1):165–178.
- , , , et al. Urinary biomarkers in the clinical prognosis and early detection of acute kidney injury. Clin J Am Soc Nephrol. 2010;5(12):2154–2165.
- , , , et al. Biomarkers for early diagnosis of AKI in the ICU: ready for prime time use at the bedside? Ann Intensive Care. 2012;2(1):24.
- , . The case for 0.9% NaCl: is the undefendable, defensible? Kidney Int. 2014;86(6):1087–1095.
- , , , , . A systematic review of the use of opioid medication for those with moderate to severe cancer pain and renal impairment: a European Palliative Care Research Collaborative opioid guidelines project. Palliat Med. 2011;25(5):525–552.
- , , . Gabapentin toxicity in patients with chronic kidney disease: a preventable cause of morbidity. Am J Med. 2010;123(4):367–373.
- , , , et al. Timing of initiation of dialysis in critically ill patients with acute kidney injury. Clin J Am Soc Nephrol. 2006;1(5):915–919.
- , , , et al. Nephrology consultation in acute renal failure: does timing matter? Am J Med. 2002;113(6):456–461.
- , , , et al. Early nephrologist involvement in hospital‐acquired acute kidney injury: a pilot study. Am J Kidney Dis. 2011;57(2):228–234.
- , , , et al. Association between prior peripherally inserted central catheters and lack of functioning arteriovenous fistulas: a case‐control study in hemodialysis patients. Am J Kidney Dis. 2012;60(4):601–608.
- , , , , , . Update on clinical trials for the prevention of acute kidney injury in patients undergoing cardiac surgery. Am J Surg. 2013;206(1):86–95.
- , , , et al. Strategies to reduce the risk of contrast‐induced nephropathy. Am J Cardiol. 2006;98(6A):59K–77K.
- , , . Acute kidney injury. Lancet. 2012;380(9843):756–766.
- , , , , . Acute renal failure—definition, outcome measures, animal models, fluid therapy and information technology needs: the Second International Consensus Conference of the Acute Dialysis Quality Initiative (ADQI) Group. Crit Care. 2004;8(4):R204–R212.
- , , , et al. Improving outcomes from acute kidney injury: report of an initiative. Am J Kidney Dis. 2007;50(1):1–4.
- , , , et al. Incidence and mortality of acute renal failure in Medicare beneficiaries, 1992 to 2001. J Am Soc Nephrol. 2006;17(4):1135–1142.
- , , . Hospital‐acquired renal insufficiency. Am J Kidney Dis. 2002;39(5):930–936.
- , , , . Acute renal failure: factors influencing nephrology referral and outcome. QJM. 1997;90(12):781–785.
- , , , , . An assessment of the RIFLE criteria for acute renal failure in hospitalized patients. Crit Care Med. 2006;34(7):1913–1917.
- , , , , . Acute kidney injury, mortality, length of stay, and costs in hospitalized patients. J Am Soc Nephrol. 2005;16(11):3365–3370.
- , , , , . The prognostic importance of a small acute decrement in kidney function in hospitalized patients: a systematic review and meta‐analysis. Am J Kidney Dis. 2007;50(5):712–720.
- , , , , , . Small acute increases in serum creatinine are associated with decreased long‐term survival in the critically ill. Am J Respir Crit Care Med. 2014;189(9):1075–1081.
- , , , et al. Acute kidney injury increases risk of ESRD among elderly. J Am Soc Nephrol. 2009;20(1):223–228.
- , , , , , . The risk of acute renal failure in patients with chronic kidney disease. Kidney Int. 2008;74(1):101–107.
- , , . Chronic kidney disease after acute kidney injury: a systematic review and meta‐analysis. Kidney Int. 2012;81(5):442–448.
- , , , et al. Risk of chronic dialysis and death following acute kidney injury. Am J Med. 2012;125(6):585–593.
- , , , et al. Chronic dialysis and death among survivors of acute kidney injury requiring dialysis. JAMA. 2009;302(11):1179–1185.
- , , , et al. Development and validation of an acute kidney injury risk index for patients undergoing general surgery: results from a national data set. Anesthesiology. 2009;110(3):505–515.
- , , , et al. Preoperative estimates of glomerular filtration rate as predictors of outcome after surgery: a systematic review and meta‐analysis. Anesthesiology. 2013;118(4):809–824.
- , . Brenner 2008.
- , , , , . Hospital‐acquired renal insufficiency: a prospective study. Am J Med. 1983;74(2):243–248.
- , , , . A nationwide nested case‐control study indicates an increased risk of acute interstitial nephritis with proton pump inhibitor use. Kidney Int. 2014;86(4):837–844.
- , , , . Acute phosphate nephropathy following oral sodium phosphate bowel purgative: an underrecognized cause of chronic renal failure. J Am Soc Nephrol. 2005;16(11):3389–3396.
- . Normotensive ischemic acute renal failure. N Engl J Med. 2007;357(8):797–805.
- , , . The rational clinical examination. Is this patient hypovolemic? JAMA. 1999;281(11):1022–1029.
- , . Atheroembolic renal disease. Lancet. 2010;375(9726):1650–1660.
- , , , et al. The challenge of diagnosing atheroembolic renal disease: clinical features and prognostic factors. Circulation. 2007;116(3):298–304.
- , , , , , . A more accurate method to estimate glomerular filtration rate from serum creatinine: a new prediction equation. Modification of Diet in Renal Disease Study Group. Ann Intern Med. 1999;130(6):461–470.
- , . Traditional urinary biomarkers in the assessment of hospital‐acquired AKI. Clin J Am Soc Nephrol. 2012;7(1):167–174.
- , . Urinary eosinophils in AIN: farewell to an old biomarker? Clin J Am Soc Nephrol. 2013;8(11):1841–1843.
- , , . Utility of urine eosinophils in the diagnosis of acute interstitial nephritis. Clin J Am Soc Nephrol. 2013;8(11):1857–1862.
- . Diagnosis of acute kidney injury: from classic parameters to new biomarkers. Contrib Nephrol. 2007;156:213–219.
- , . Biomarkers in nephrology: Core Curriculum 2013. Am J Kidney Dis. 2013;62(1):165–178.
- , , , et al. Urinary biomarkers in the clinical prognosis and early detection of acute kidney injury. Clin J Am Soc Nephrol. 2010;5(12):2154–2165.
- , , , et al. Biomarkers for early diagnosis of AKI in the ICU: ready for prime time use at the bedside? Ann Intensive Care. 2012;2(1):24.
- , . The case for 0.9% NaCl: is the undefendable, defensible? Kidney Int. 2014;86(6):1087–1095.
- , , , , . A systematic review of the use of opioid medication for those with moderate to severe cancer pain and renal impairment: a European Palliative Care Research Collaborative opioid guidelines project. Palliat Med. 2011;25(5):525–552.
- , , . Gabapentin toxicity in patients with chronic kidney disease: a preventable cause of morbidity. Am J Med. 2010;123(4):367–373.
- , , , et al. Timing of initiation of dialysis in critically ill patients with acute kidney injury. Clin J Am Soc Nephrol. 2006;1(5):915–919.
- , , , et al. Nephrology consultation in acute renal failure: does timing matter? Am J Med. 2002;113(6):456–461.
- , , , et al. Early nephrologist involvement in hospital‐acquired acute kidney injury: a pilot study. Am J Kidney Dis. 2011;57(2):228–234.
- , , , et al. Association between prior peripherally inserted central catheters and lack of functioning arteriovenous fistulas: a case‐control study in hemodialysis patients. Am J Kidney Dis. 2012;60(4):601–608.
- , , , , , . Update on clinical trials for the prevention of acute kidney injury in patients undergoing cardiac surgery. Am J Surg. 2013;206(1):86–95.
- , , , et al. Strategies to reduce the risk of contrast‐induced nephropathy. Am J Cardiol. 2006;98(6A):59K–77K.
Improving Interhospital Transfers
Mrs. S arrived to the medicine service at our hospital by ambulance transport at 9:00 pm. The intern on call received a page from the nurse, Mrs. S has arrived. She is confused. Please assess. As is often the case, the intern had no prior knowledge of the patient's arrival, and review of medical records indicated that Mrs. S had never been seen at our hospital before.
The intern went to the bedside to assess the patient and found an elderly woman who appeared confused and was unable to provider her medical history, reason for the transfer, or details about her recent hospital course.
A few minutes later, the patient's son arrived at the bedside asking about her plan of care. The intern looked through the stack of papers in the envelope by her chart, and was able to locate reports of a recent chest x‐ray and abdominal computed tomography, as well as copies of brief progress notes, but was unable to find a transfer summary detailing her prior 5 days of hospitalization or reason for transfer. The patient's son was able to give some information, but he had just returned from a business trip and was not up to date on the details of his mother's hospital stay. Based on her son's input, the intern concluded the patient's somnolence was not her baseline; he performed an arterial blood gas and blood work, revealing profound acidemia and hyponatremia of unclear acuity. Mrs. S became hypotensive, requiring transfer to the intensive care unit. Several days later, she died.
This scenario highlights the potential dangers associated with patient transfers between acute care hospitals, known as interhospital transfer (IHT). Unfortunately, the described scenario is not a rare event.[1, 2] Most providers who care for transferred patients can recount similar challenges when caring for IHT patients.[3]
Patient transfers from 1 hospital to another are common, affecting nearly 1 in 20 Medicare patients admitted to the intensive care unit[4] and up to 50% of patients presenting with acute myocardial infarction,[5] although reasons for transfer remain largely unstudied. The Emergency Medical Treatment and Active Labor Act requires a hospital to transfer patients who require a more specialized service unavailable at the subject institution, or when medical benefits outweigh the increased risks to the individual.[6] Yet, this broad standard provides little guidance to clinicians in practice.
Identifying which patients may benefit from transfer is an ambiguous and subjective process. Studies show little agreement between the reasons cited for transfer among patients, transferring physicians, and receiving physicians,[7] and incentives for transfer are often different between each stakeholder. For example, patients or families might initiate transfer for a second opinion from a fresh set of eyes because of a grim or uncertain prognosis or in the hope of a more promising or definitive medical opinion. Similarly, referring physicians may initiate transfer for particular procedures, surgeries, or consultations that the receiving physician may ultimately decide will be of little clinical benefit to the patient. Such heightened expectations and changes to the care plan as agreed on by the patient and referring physician may affect the patient's perceptions of care at the receiving institution, although exactly how remains unknown. Alternatively, patients and families may desire transfer because of previously established relationships with providers at another institution, or they may be dissatisfied with certain aspects of care at the referring institution. Referring institutions may initiate transfer for a variety of reasons, including inability to provide a needed procedure or test, patient/family preference, or protocol, among others. Receiving hospitals usually have an interest in maintaining a large referral base for the sake of both revenue and reputation, but may also view accepting transfers as part of their larger mission to provide expert consultation and specialty services that may not be available at the referring institution. Additional proposed benefits include strengthening provider networks, promoting clinical diversity, and improving the educational experience of trainees often present at the accepting institution. Although patients, providers, and referring and accepting hospitals all undoubtedly benefit from various aspects of the IHT, further research is needed to more clearly identify which patients are most likely to benefit from transfer and why.
Once the decision to transfer/accept a patient has been made, there are no clear guidelines over how this process should be executed. For this reason, care providers at community hospitals describe IHT as frustrating and time consuming.[8] Referring providers may face challenges identifying an accepting hospital due to the limited capacity of the receiving institution, reaching the correct receiving physician, and managing delays in transfer once the patient is accepted.[8] Similarly, accepting physicians may be frustrated by the time waste associated with accepting a patient that ends up transferred to another facility, limited authority to triage the patient to the most appropriate accepting service, inability to predict time of patient arrival, and missing pieces of critical information at time of patient arrival, among other reasons. Furthermore, incompatible electronic health records make access to data from the referring institution difficult. For example, without standards for transferring imaging, patients may undergo unnecessary and costly duplicate imaging leading to delay in needed procedures. Existing guidelines are largely focused on equipment and expertise required for the physical transfer of the patient, but fail to consider other aspects of the transfer process that may be critical for patient safety such as protocols for communication of patient information and transfer of completed imaging. As such, hospitals are largely left to devise their own protocols for IHT, which often differ between hospitals as well as between different services within 1 hospital.[1, 3]
Although it is true that many patients benefit from IHT, the process introduces inherent vulnerability into healthcare delivery. Moving a patient between facilities exposes that individual to risks associated with discontinuity of care, well described in the literature on intrahospital patient handoffs (ie, the transfer of patient care responsibility from 1 provider to another within 1 hospital), which can lead to excessive costs and poor patient outcomes.[9] Presumably, such risks are even greater for patients transferred between hospitals than for those transferred between providers within 1 hospital, because system factors like electronic health records, nursing and ancillary staff continuity, and accessibility of transferring provider are not in place to mitigate communication gaps. Furthermore, unlike discharges home or to subacute care facilities, also known to be error prone and lead to adverse events,[10, 11] in the case of IHT, patients are often more acutely ill and less stable. In fact, limited data suggest that aside from a select subset of patients requiring specialized care, individuals transferred may have increased resource utilization and greater‐than‐expected mortality than those who are not transferred.[1, 2, 12] Moreover, these findings may not be entirely attributable to medical complexity among transferred patients.
Today, the process of IHT varies tremendously across US hospitals,[1] differences that may have significant implications for both cost and patient safety outcomes. Standardization of IHT, including patient selection and information exchange between transferring and accepting providers/emnstitutions, is imperative to improve the quality and safety of this process. As demonstrated with other common, high‐risk care transitions, such as intrahospital patient handoffs and patient discharge, creating basic guidelines of practice (such as including important data elements at time of care transfer)[13, 14] is necessary to improve quality of the care transition.
However, to achieve high‐quality standardization, we must first methodologically conduct rigorous clinical research to understand fundamental issues of the IHT process, including why patients are transferred (from the perspective of patients and transferring and accepting institutions), which patients benefit most from transfer and why, and how various IHT processes impact health outcomes. Interventions such as communication and data transfer tools, feedback mechanisms between referring and accepting institutions, and other evidence‐based guidelines can then be designed to improve IHT based on the findings of this research while still allowing for flexibility of individual patient needs. Additional work is then needed to implement and rigorously evaluate the effects of such interventions on patient and provider outcomes including, but not limited to, length of stay, adverse events, mortality, readmissions, and patient satisfaction measures. In summary, by focusing research and quality improvement initiatives on these vital questions, we can begin to improve the quality of care we provide to patients during this critical transition of care.
Disclosure
Disclosure: Nothing to report.
- , , , . Patients transferred from outside hospitals to academic hospitalists and general internists have higher mortality and costs than patients from the ED. Paper presented at: Society of Hospital Medicine National Conference; May 2013; Washington, DC.
- , , , . Interhospital facility transfers in the united states: a nationwide outcomes study [published online ahead of print November 13, 2014]. J Patient Saf. doi: 10.1097/PTS.0000000000000148.
- , . Physician perspectives on inter‐hospital transfers. Paper presented at: Society of Hospital Medicine National Conference; March 2014; Las Vegas, NV.
- , , , , . The structure of critical care transfer networks. Med Care. 2009;47(7):787–793.
- , , , . Interhospital transfers among Medicare beneficiaries admitted for acute myocardial infarction at nonrevascularization hospitals. Circ Cardiovasc Qual Outcomes. 2010;3(5):468–475.
- U.S. House of Representatives. Office of the Law Revision Counsel. Examination and treatment for emergency medical conditions and women in labor. Title 42 USC §1395dd. Available at: http://www.gpo.gov/fdsys/granule/USCODE‐2010‐title42/USCODE‐2010‐title42‐chap7‐subchapXVIII‐partE‐sec1395dd. Accessed October 29 2014.
- , , . Reasons underlying interhospital transfers to an academic medical intensive care unit. J Crit Care. 2013;28(2):202–208.
- , , . Which patients and where: a qualitative study of patient transfers from community hospitals. Med Care. 2011;49(6):592–598.
- , , , . Conceptualizing handover strategies at change of shift in the emergency department: a grounded theory study. BMC Health Serv Res. 2008;8:256.
- , , , , . The incidence and severity of adverse events affecting patients after discharge from the hospital. Ann Intern Med. 2003;138(3):161–167.
- , , , , . Pending laboratory tests and the hospital discharge summary in patients discharged to sub‐acute care. J Gen Intern Med. 2011;26(4):393–398.
- , , , , . Comparing the hospitalizations of transfer and non‐transfer patients in an academic medical center. Acad Med. 1996;71(3):262–266.
- , , , , , . I‐pass, a mnemonic to standardize verbal handoffs. Pediatrics. 2012;129(2):201–204.
- Hospital Medicine Reengineering Network (HOMERUN) Collaborative. Executive summary. Available at: https://members.aamc.org/eweb/upload/HOMERUN%20summary%202012.pdf. Accessed July 23, 2013.
Mrs. S arrived to the medicine service at our hospital by ambulance transport at 9:00 pm. The intern on call received a page from the nurse, Mrs. S has arrived. She is confused. Please assess. As is often the case, the intern had no prior knowledge of the patient's arrival, and review of medical records indicated that Mrs. S had never been seen at our hospital before.
The intern went to the bedside to assess the patient and found an elderly woman who appeared confused and was unable to provider her medical history, reason for the transfer, or details about her recent hospital course.
A few minutes later, the patient's son arrived at the bedside asking about her plan of care. The intern looked through the stack of papers in the envelope by her chart, and was able to locate reports of a recent chest x‐ray and abdominal computed tomography, as well as copies of brief progress notes, but was unable to find a transfer summary detailing her prior 5 days of hospitalization or reason for transfer. The patient's son was able to give some information, but he had just returned from a business trip and was not up to date on the details of his mother's hospital stay. Based on her son's input, the intern concluded the patient's somnolence was not her baseline; he performed an arterial blood gas and blood work, revealing profound acidemia and hyponatremia of unclear acuity. Mrs. S became hypotensive, requiring transfer to the intensive care unit. Several days later, she died.
This scenario highlights the potential dangers associated with patient transfers between acute care hospitals, known as interhospital transfer (IHT). Unfortunately, the described scenario is not a rare event.[1, 2] Most providers who care for transferred patients can recount similar challenges when caring for IHT patients.[3]
Patient transfers from 1 hospital to another are common, affecting nearly 1 in 20 Medicare patients admitted to the intensive care unit[4] and up to 50% of patients presenting with acute myocardial infarction,[5] although reasons for transfer remain largely unstudied. The Emergency Medical Treatment and Active Labor Act requires a hospital to transfer patients who require a more specialized service unavailable at the subject institution, or when medical benefits outweigh the increased risks to the individual.[6] Yet, this broad standard provides little guidance to clinicians in practice.
Identifying which patients may benefit from transfer is an ambiguous and subjective process. Studies show little agreement between the reasons cited for transfer among patients, transferring physicians, and receiving physicians,[7] and incentives for transfer are often different between each stakeholder. For example, patients or families might initiate transfer for a second opinion from a fresh set of eyes because of a grim or uncertain prognosis or in the hope of a more promising or definitive medical opinion. Similarly, referring physicians may initiate transfer for particular procedures, surgeries, or consultations that the receiving physician may ultimately decide will be of little clinical benefit to the patient. Such heightened expectations and changes to the care plan as agreed on by the patient and referring physician may affect the patient's perceptions of care at the receiving institution, although exactly how remains unknown. Alternatively, patients and families may desire transfer because of previously established relationships with providers at another institution, or they may be dissatisfied with certain aspects of care at the referring institution. Referring institutions may initiate transfer for a variety of reasons, including inability to provide a needed procedure or test, patient/family preference, or protocol, among others. Receiving hospitals usually have an interest in maintaining a large referral base for the sake of both revenue and reputation, but may also view accepting transfers as part of their larger mission to provide expert consultation and specialty services that may not be available at the referring institution. Additional proposed benefits include strengthening provider networks, promoting clinical diversity, and improving the educational experience of trainees often present at the accepting institution. Although patients, providers, and referring and accepting hospitals all undoubtedly benefit from various aspects of the IHT, further research is needed to more clearly identify which patients are most likely to benefit from transfer and why.
Once the decision to transfer/accept a patient has been made, there are no clear guidelines over how this process should be executed. For this reason, care providers at community hospitals describe IHT as frustrating and time consuming.[8] Referring providers may face challenges identifying an accepting hospital due to the limited capacity of the receiving institution, reaching the correct receiving physician, and managing delays in transfer once the patient is accepted.[8] Similarly, accepting physicians may be frustrated by the time waste associated with accepting a patient that ends up transferred to another facility, limited authority to triage the patient to the most appropriate accepting service, inability to predict time of patient arrival, and missing pieces of critical information at time of patient arrival, among other reasons. Furthermore, incompatible electronic health records make access to data from the referring institution difficult. For example, without standards for transferring imaging, patients may undergo unnecessary and costly duplicate imaging leading to delay in needed procedures. Existing guidelines are largely focused on equipment and expertise required for the physical transfer of the patient, but fail to consider other aspects of the transfer process that may be critical for patient safety such as protocols for communication of patient information and transfer of completed imaging. As such, hospitals are largely left to devise their own protocols for IHT, which often differ between hospitals as well as between different services within 1 hospital.[1, 3]
Although it is true that many patients benefit from IHT, the process introduces inherent vulnerability into healthcare delivery. Moving a patient between facilities exposes that individual to risks associated with discontinuity of care, well described in the literature on intrahospital patient handoffs (ie, the transfer of patient care responsibility from 1 provider to another within 1 hospital), which can lead to excessive costs and poor patient outcomes.[9] Presumably, such risks are even greater for patients transferred between hospitals than for those transferred between providers within 1 hospital, because system factors like electronic health records, nursing and ancillary staff continuity, and accessibility of transferring provider are not in place to mitigate communication gaps. Furthermore, unlike discharges home or to subacute care facilities, also known to be error prone and lead to adverse events,[10, 11] in the case of IHT, patients are often more acutely ill and less stable. In fact, limited data suggest that aside from a select subset of patients requiring specialized care, individuals transferred may have increased resource utilization and greater‐than‐expected mortality than those who are not transferred.[1, 2, 12] Moreover, these findings may not be entirely attributable to medical complexity among transferred patients.
Today, the process of IHT varies tremendously across US hospitals,[1] differences that may have significant implications for both cost and patient safety outcomes. Standardization of IHT, including patient selection and information exchange between transferring and accepting providers/emnstitutions, is imperative to improve the quality and safety of this process. As demonstrated with other common, high‐risk care transitions, such as intrahospital patient handoffs and patient discharge, creating basic guidelines of practice (such as including important data elements at time of care transfer)[13, 14] is necessary to improve quality of the care transition.
However, to achieve high‐quality standardization, we must first methodologically conduct rigorous clinical research to understand fundamental issues of the IHT process, including why patients are transferred (from the perspective of patients and transferring and accepting institutions), which patients benefit most from transfer and why, and how various IHT processes impact health outcomes. Interventions such as communication and data transfer tools, feedback mechanisms between referring and accepting institutions, and other evidence‐based guidelines can then be designed to improve IHT based on the findings of this research while still allowing for flexibility of individual patient needs. Additional work is then needed to implement and rigorously evaluate the effects of such interventions on patient and provider outcomes including, but not limited to, length of stay, adverse events, mortality, readmissions, and patient satisfaction measures. In summary, by focusing research and quality improvement initiatives on these vital questions, we can begin to improve the quality of care we provide to patients during this critical transition of care.
Disclosure
Disclosure: Nothing to report.
Mrs. S arrived to the medicine service at our hospital by ambulance transport at 9:00 pm. The intern on call received a page from the nurse, Mrs. S has arrived. She is confused. Please assess. As is often the case, the intern had no prior knowledge of the patient's arrival, and review of medical records indicated that Mrs. S had never been seen at our hospital before.
The intern went to the bedside to assess the patient and found an elderly woman who appeared confused and was unable to provider her medical history, reason for the transfer, or details about her recent hospital course.
A few minutes later, the patient's son arrived at the bedside asking about her plan of care. The intern looked through the stack of papers in the envelope by her chart, and was able to locate reports of a recent chest x‐ray and abdominal computed tomography, as well as copies of brief progress notes, but was unable to find a transfer summary detailing her prior 5 days of hospitalization or reason for transfer. The patient's son was able to give some information, but he had just returned from a business trip and was not up to date on the details of his mother's hospital stay. Based on her son's input, the intern concluded the patient's somnolence was not her baseline; he performed an arterial blood gas and blood work, revealing profound acidemia and hyponatremia of unclear acuity. Mrs. S became hypotensive, requiring transfer to the intensive care unit. Several days later, she died.
This scenario highlights the potential dangers associated with patient transfers between acute care hospitals, known as interhospital transfer (IHT). Unfortunately, the described scenario is not a rare event.[1, 2] Most providers who care for transferred patients can recount similar challenges when caring for IHT patients.[3]
Patient transfers from 1 hospital to another are common, affecting nearly 1 in 20 Medicare patients admitted to the intensive care unit[4] and up to 50% of patients presenting with acute myocardial infarction,[5] although reasons for transfer remain largely unstudied. The Emergency Medical Treatment and Active Labor Act requires a hospital to transfer patients who require a more specialized service unavailable at the subject institution, or when medical benefits outweigh the increased risks to the individual.[6] Yet, this broad standard provides little guidance to clinicians in practice.
Identifying which patients may benefit from transfer is an ambiguous and subjective process. Studies show little agreement between the reasons cited for transfer among patients, transferring physicians, and receiving physicians,[7] and incentives for transfer are often different between each stakeholder. For example, patients or families might initiate transfer for a second opinion from a fresh set of eyes because of a grim or uncertain prognosis or in the hope of a more promising or definitive medical opinion. Similarly, referring physicians may initiate transfer for particular procedures, surgeries, or consultations that the receiving physician may ultimately decide will be of little clinical benefit to the patient. Such heightened expectations and changes to the care plan as agreed on by the patient and referring physician may affect the patient's perceptions of care at the receiving institution, although exactly how remains unknown. Alternatively, patients and families may desire transfer because of previously established relationships with providers at another institution, or they may be dissatisfied with certain aspects of care at the referring institution. Referring institutions may initiate transfer for a variety of reasons, including inability to provide a needed procedure or test, patient/family preference, or protocol, among others. Receiving hospitals usually have an interest in maintaining a large referral base for the sake of both revenue and reputation, but may also view accepting transfers as part of their larger mission to provide expert consultation and specialty services that may not be available at the referring institution. Additional proposed benefits include strengthening provider networks, promoting clinical diversity, and improving the educational experience of trainees often present at the accepting institution. Although patients, providers, and referring and accepting hospitals all undoubtedly benefit from various aspects of the IHT, further research is needed to more clearly identify which patients are most likely to benefit from transfer and why.
Once the decision to transfer/accept a patient has been made, there are no clear guidelines over how this process should be executed. For this reason, care providers at community hospitals describe IHT as frustrating and time consuming.[8] Referring providers may face challenges identifying an accepting hospital due to the limited capacity of the receiving institution, reaching the correct receiving physician, and managing delays in transfer once the patient is accepted.[8] Similarly, accepting physicians may be frustrated by the time waste associated with accepting a patient that ends up transferred to another facility, limited authority to triage the patient to the most appropriate accepting service, inability to predict time of patient arrival, and missing pieces of critical information at time of patient arrival, among other reasons. Furthermore, incompatible electronic health records make access to data from the referring institution difficult. For example, without standards for transferring imaging, patients may undergo unnecessary and costly duplicate imaging leading to delay in needed procedures. Existing guidelines are largely focused on equipment and expertise required for the physical transfer of the patient, but fail to consider other aspects of the transfer process that may be critical for patient safety such as protocols for communication of patient information and transfer of completed imaging. As such, hospitals are largely left to devise their own protocols for IHT, which often differ between hospitals as well as between different services within 1 hospital.[1, 3]
Although it is true that many patients benefit from IHT, the process introduces inherent vulnerability into healthcare delivery. Moving a patient between facilities exposes that individual to risks associated with discontinuity of care, well described in the literature on intrahospital patient handoffs (ie, the transfer of patient care responsibility from 1 provider to another within 1 hospital), which can lead to excessive costs and poor patient outcomes.[9] Presumably, such risks are even greater for patients transferred between hospitals than for those transferred between providers within 1 hospital, because system factors like electronic health records, nursing and ancillary staff continuity, and accessibility of transferring provider are not in place to mitigate communication gaps. Furthermore, unlike discharges home or to subacute care facilities, also known to be error prone and lead to adverse events,[10, 11] in the case of IHT, patients are often more acutely ill and less stable. In fact, limited data suggest that aside from a select subset of patients requiring specialized care, individuals transferred may have increased resource utilization and greater‐than‐expected mortality than those who are not transferred.[1, 2, 12] Moreover, these findings may not be entirely attributable to medical complexity among transferred patients.
Today, the process of IHT varies tremendously across US hospitals,[1] differences that may have significant implications for both cost and patient safety outcomes. Standardization of IHT, including patient selection and information exchange between transferring and accepting providers/emnstitutions, is imperative to improve the quality and safety of this process. As demonstrated with other common, high‐risk care transitions, such as intrahospital patient handoffs and patient discharge, creating basic guidelines of practice (such as including important data elements at time of care transfer)[13, 14] is necessary to improve quality of the care transition.
However, to achieve high‐quality standardization, we must first methodologically conduct rigorous clinical research to understand fundamental issues of the IHT process, including why patients are transferred (from the perspective of patients and transferring and accepting institutions), which patients benefit most from transfer and why, and how various IHT processes impact health outcomes. Interventions such as communication and data transfer tools, feedback mechanisms between referring and accepting institutions, and other evidence‐based guidelines can then be designed to improve IHT based on the findings of this research while still allowing for flexibility of individual patient needs. Additional work is then needed to implement and rigorously evaluate the effects of such interventions on patient and provider outcomes including, but not limited to, length of stay, adverse events, mortality, readmissions, and patient satisfaction measures. In summary, by focusing research and quality improvement initiatives on these vital questions, we can begin to improve the quality of care we provide to patients during this critical transition of care.
Disclosure
Disclosure: Nothing to report.
- , , , . Patients transferred from outside hospitals to academic hospitalists and general internists have higher mortality and costs than patients from the ED. Paper presented at: Society of Hospital Medicine National Conference; May 2013; Washington, DC.
- , , , . Interhospital facility transfers in the united states: a nationwide outcomes study [published online ahead of print November 13, 2014]. J Patient Saf. doi: 10.1097/PTS.0000000000000148.
- , . Physician perspectives on inter‐hospital transfers. Paper presented at: Society of Hospital Medicine National Conference; March 2014; Las Vegas, NV.
- , , , , . The structure of critical care transfer networks. Med Care. 2009;47(7):787–793.
- , , , . Interhospital transfers among Medicare beneficiaries admitted for acute myocardial infarction at nonrevascularization hospitals. Circ Cardiovasc Qual Outcomes. 2010;3(5):468–475.
- U.S. House of Representatives. Office of the Law Revision Counsel. Examination and treatment for emergency medical conditions and women in labor. Title 42 USC §1395dd. Available at: http://www.gpo.gov/fdsys/granule/USCODE‐2010‐title42/USCODE‐2010‐title42‐chap7‐subchapXVIII‐partE‐sec1395dd. Accessed October 29 2014.
- , , . Reasons underlying interhospital transfers to an academic medical intensive care unit. J Crit Care. 2013;28(2):202–208.
- , , . Which patients and where: a qualitative study of patient transfers from community hospitals. Med Care. 2011;49(6):592–598.
- , , , . Conceptualizing handover strategies at change of shift in the emergency department: a grounded theory study. BMC Health Serv Res. 2008;8:256.
- , , , , . The incidence and severity of adverse events affecting patients after discharge from the hospital. Ann Intern Med. 2003;138(3):161–167.
- , , , , . Pending laboratory tests and the hospital discharge summary in patients discharged to sub‐acute care. J Gen Intern Med. 2011;26(4):393–398.
- , , , , . Comparing the hospitalizations of transfer and non‐transfer patients in an academic medical center. Acad Med. 1996;71(3):262–266.
- , , , , , . I‐pass, a mnemonic to standardize verbal handoffs. Pediatrics. 2012;129(2):201–204.
- Hospital Medicine Reengineering Network (HOMERUN) Collaborative. Executive summary. Available at: https://members.aamc.org/eweb/upload/HOMERUN%20summary%202012.pdf. Accessed July 23, 2013.
- , , , . Patients transferred from outside hospitals to academic hospitalists and general internists have higher mortality and costs than patients from the ED. Paper presented at: Society of Hospital Medicine National Conference; May 2013; Washington, DC.
- , , , . Interhospital facility transfers in the united states: a nationwide outcomes study [published online ahead of print November 13, 2014]. J Patient Saf. doi: 10.1097/PTS.0000000000000148.
- , . Physician perspectives on inter‐hospital transfers. Paper presented at: Society of Hospital Medicine National Conference; March 2014; Las Vegas, NV.
- , , , , . The structure of critical care transfer networks. Med Care. 2009;47(7):787–793.
- , , , . Interhospital transfers among Medicare beneficiaries admitted for acute myocardial infarction at nonrevascularization hospitals. Circ Cardiovasc Qual Outcomes. 2010;3(5):468–475.
- U.S. House of Representatives. Office of the Law Revision Counsel. Examination and treatment for emergency medical conditions and women in labor. Title 42 USC §1395dd. Available at: http://www.gpo.gov/fdsys/granule/USCODE‐2010‐title42/USCODE‐2010‐title42‐chap7‐subchapXVIII‐partE‐sec1395dd. Accessed October 29 2014.
- , , . Reasons underlying interhospital transfers to an academic medical intensive care unit. J Crit Care. 2013;28(2):202–208.
- , , . Which patients and where: a qualitative study of patient transfers from community hospitals. Med Care. 2011;49(6):592–598.
- , , , . Conceptualizing handover strategies at change of shift in the emergency department: a grounded theory study. BMC Health Serv Res. 2008;8:256.
- , , , , . The incidence and severity of adverse events affecting patients after discharge from the hospital. Ann Intern Med. 2003;138(3):161–167.
- , , , , . Pending laboratory tests and the hospital discharge summary in patients discharged to sub‐acute care. J Gen Intern Med. 2011;26(4):393–398.
- , , , , . Comparing the hospitalizations of transfer and non‐transfer patients in an academic medical center. Acad Med. 1996;71(3):262–266.
- , , , , , . I‐pass, a mnemonic to standardize verbal handoffs. Pediatrics. 2012;129(2):201–204.
- Hospital Medicine Reengineering Network (HOMERUN) Collaborative. Executive summary. Available at: https://members.aamc.org/eweb/upload/HOMERUN%20summary%202012.pdf. Accessed July 23, 2013.
Glucose Management and Inpatient Mortality
Patients with diabetes currently comprise over 8% of the US population (over 25 million people) and more than 20% of hospitalized patients.[1, 2] Hospitalizations of patients with diabetes account for 23% of total hospital costs in the United States,[2] and patients with diabetes have worse outcomes after hospitalization for a variety of common medical conditions,[3, 4, 5, 6] as well as in intensive care unit (ICU) settings.[7, 8] Individuals with diabetes have historically experienced higher inpatient mortality than individuals without diabetes.[9] However, we recently reported that patients with diabetes at our large academic medical center have experienced a disproportionate reduction in in‐hospital mortality relative to patients without diabetes over the past decade.[10] This surprising trend begs further inquiry.
Improvement in in‐hospital mortality among patients with diabetes may stem from improved inpatient glycemic management. The landmark 2001 study by van den Berghe et al. demonstrating that intensive insulin therapy reduced postsurgical mortality among ICU patients ushered in an era of intensive inpatient glucose control.[11] However, follow‐up multicenter studies have not been able to replicate these results.[12, 13, 14, 15] In non‐ICU and nonsurgical settings, intensive glucose control has not yet been shown to have any mortality benefit, although it may impact other morbidities, such as postoperative infections.[16] Consequently, less stringent glycemic targets are now recommended.[17] Nonetheless, hospitals are being held accountable for certain aspects of inpatient glucose control. For example, the Centers for Medicare & Medicaid Services (CMS) began asking hospitals to report inpatient glucose control in cardiac surgery patients in 2004.[18] This measure is now publicly reported, and as of 2013 is included in the CMS Value‐Based Purchasing Program, which financially penalizes hospitals that do not meet targets.
Outpatient diabetes standards have also evolved in the past decade. The Diabetes Control and Complications Trial in 1993 and the United Kingdom Prospective Diabetes Study in 1997 demonstrated that better glycemic control in type 1 and newly diagnosed type 2 diabetes patients, respectively, improved clinical outcomes, and prompted guidelines for pharmacologic treatment of diabetic patients.[19, 20] However, subsequent randomized clinical trials have failed to establish a clear beneficial effect of intensive glucose control on primary cardiovascular endpoints among higher‐risk patients with longstanding type 2 diabetes,[21, 22, 23] and clinical practice recommendations now accept a more individualized approach to glycemic control.[24] Nonetheless, clinicians are also being held accountable for outpatient glucose control.[25]
To better understand the disproportionate reduction in mortality among hospitalized patients with diabetes that we observed, we first examined whether it was limited to surgical patients or patients in the ICU, the populations that have been demonstrated to benefit from intensive inpatient glucose control. Furthermore, given recent improvements in inpatient and outpatient glycemic control,[26, 27] we examined whether inpatient or outpatient glucose control explained the mortality trends. Results from this study contribute empirical evidence on real‐world effects of efforts to improve inpatient and outpatient glycemic control.
METHODS
Setting
During the study period, YaleNew Haven Hospital (YNHH) was an urban academic medical center in New Haven, Connecticut, with over 950 beds and an average of approximately 32,000 annual adult nonobstetric admissions. YNHH conducted a variety of inpatient glucose control initiatives during the study period. The surgical ICU began an informal medical teamdirected insulin infusion protocol in 2000 to 2001. In 2002, the medical ICU instituted a formal insulin infusion protocol with a target of 100 to 140 mg/dL, which spread to remaining hospital ICUs by the end of 2003. In 2005, YNHH launched a consultative inpatient diabetes management team to assist clinicians in controlling glucose in non‐ICU patients with diabetes. This team covered approximately 10 to 15 patients at a time and consisted of an advanced‐practice nurse practitioner, a supervising endocrinologist and endocrinology fellow, and a nurse educator to provide diabetic teaching. Additionally, in 2005, basal‐boluscorrection insulin order sets became available. The surgical ICU implemented a stringent insulin infusion protocol with target glucose of 80 to 110 mg/dL in 2006, but relaxed it (goal 80150 mg/dL) in 2007. Similarly, in 2006, YNHH made ICU insulin infusion recommendations more stringent in remaining ICUs (goal 90130 mg/dL), but relaxed them in 2010 (goal 120160 mg/dL), based on emerging data from clinical trials and prevailing national guidelines.
Participants and Data Sources
We included all adult, nonobstetric discharges from YNHH between January 1, 2000 and December 31, 2010. Repeat visits by the same patient were linked by medical record number. We obtained data from YNHH administrative billing, laboratory, and point‐of‐care capillary blood glucose databases. The Yale Human Investigation Committee approved our study design and granted a Health Insurance Portability and Accountability Act waiver and a waiver of patient consent.
Variables
Our primary endpoint was in‐hospital mortality. The primary exposure of interest was whether a patient had diabetes mellitus, defined as the presence of International Classification of Diseases, Ninth Revision codes 249.x, 250.x, V4585, V5391, or V6546 in any of the primary or secondary diagnosis codes in the index admission, or in any hospital encounter in the year prior to the index admission.
We assessed 2 effect‐modifying variables: ICU status (as measured by a charge for at least 1 night in the ICU) and service assignment to surgery (including neurosurgery and orthopedics), compared to medicine (including neurology). Independent explanatory variables included time between the start of the study and patient admission (measured as days/365), diabetes status, inpatient glucose control, and long‐term glucose control (as measured by hemoglobin A1c at any time in the 180 days prior to hospital admission in order to have adequate sample size). We assessed inpatient blood glucose control through point‐of‐care blood glucose meters (OneTouch SureStep; LifeScan, Inc., Milipitas, CA) at YNHH. We used 4 validated measures of inpatient glucose control: the proportion of days in each hospitalization in which there was any hypoglycemic episode (blood glucose value <70 mg/dL), the proportion of days in which there was any severely hyperglycemic episode (blood glucose value >299 mg/dL), the proportion of days in which mean blood glucose was considered to be within adequate control (all blood glucose values between 70 and 179 mg/dL), and the standard deviation of mean glucose during hospitalization as a measure of glycemic variability.[28]
Covariates included gender, age at time of admission, length of stay in days, race (defined by hospital registration), payer, Elixhauser comorbidity dummy variables (revised to exclude diabetes and to use only secondary diagnosis codes),[29] and primary discharge diagnosis grouped using Clinical Classifications Software,[30] based on established associations with in‐hospital mortality.
Statistical Analysis
We summarized demographic characteristics numerically and graphically for patients with and without diabetes and compared them using [2] and t tests. We summarized changes in inpatient and outpatient measures of glucose control over time numerically and graphically, and compared across years using the Wilcoxon rank sum test adjusted for multiple hypothesis testing.
We stratified all analyses first by ICU status and then by service assignment (medicine vs surgery). Statistical analyses within each stratum paralleled our previous approach to the full study cohort.[10] Taking each stratum separately (ie, only ICU patients or only medicine patients), we used a difference‐in‐differences approach comparing changes over time in in‐hospital mortality among patients with diabetes compared to those without diabetes. This approach enabled us to determine whether patients with diabetes had a different time trend in risk of in‐hospital mortality than those without diabetes. That is, for each stratum, we constructed multivariate logistic regression models including time in years, diabetes status, and the interaction between time and diabetes status as well as the aforementioned covariates. We calculated odds of death and confidence intervals for each additional year for patients with diabetes by exponentiating the sum of parameter estimates for time and the diabetes‐time interaction term. We evaluated all 2‐way interactions between year or diabetes status and the covariates in a multiple degree of freedom likelihood ratio test. We investigated nonlinearity of the relation between mortality and time by evaluating first and second‐order polynomials.
Because we found a significant decline in mortality risk for patients with versus without diabetes among ICU patients but not among non‐ICU patients, and because service assignment was not found to be an effect modifier, we then limited our sample to ICU patients with diabetes to better understand the role of inpatient and outpatient glucose control in accounting for observed mortality trends. First, we determined the relation between the measures of inpatient glucose control and changes in mortality over time using logistic regression. Then, we repeated this analysis in the subsets of patients who had inpatient glucose data and both inpatient and outpatient glycemic control data, adding inpatient and outpatient measures sequentially. Given the high level of missing outpatient glycemic control data, we compared demographic characteristics for diabetic ICU patients with and without such data using [2] and t tests, and found that patients with data were younger and less likely to be white and had longer mean length of stay, slightly worse performance on several measures of inpatient glucose control, and lower mortality (see Supporting Table 1 in the online version of this article).
| Characteristic | Overall, N=322,939 | Any ICU Stay, N=54,646 | No ICU Stay, N=268,293 | Medical Service, N=196,325 | Surgical Service, N=126,614 |
|---|---|---|---|---|---|
| |||||
| Died during admission, n (%) | 7,587 (2.3) | 5,439 (10.0) | 2,147 (0.8) | 5,705 (2.9) | 1,883 (1.5) |
| Diabetes, n (%) | 76,758 (23.8) | 14,364 (26.3) | 62,394 (23.2) | 55,453 (28.2) | 21,305 (16.8) |
| Age, y, mean (SD) | 55.5 (20.0) | 61.0 (17.0) | 54.4 (21.7) | 60.3 (18.9) | 48.0 (23.8) |
| Age, full range (interquartile range) | 0118 (4273) | 18112 (4975) | 0118 (4072) | 0118 (4776) | 0111 (3266) |
| Female, n (%) | 159,227 (49.3) | 23,208 (42.5) | 134,296 (50.1) | 99,805 (50.8) | 59,422 (46.9) |
| White race, n (%) | 226,586 (70.2) | 41,982 (76.8) | 184,604 (68.8) | 132,749 (67.6) | 93,838 (74.1) |
| Insurance, n (%) | |||||
| Medicaid | 54,590 (16.9) | 7,222 (13.2) | 47,378 (17.7) | 35,229 (17.9) | 19,361 (15.3) |
| Medicare | 141,638 (43.9) | 27,458 (50.2) | 114,180 (42.6) | 100,615 (51.2) | 41,023 (32.4) |
| Commercial | 113,013 (35.0) | 18,248 (33.4) | 94,765 (35.3) | 53,510 (27.2) | 59,503 (47.0) |
| Uninsured | 13,521 (4.2) | 1,688 (3.1) | 11,833 (4.4) | 6,878 (3.5) | 6,643 (5.2) |
| Length of stay, d, mean (SD) | 5.4 (9.5) | 11.8 (17.8) | 4.2 (6.2) | 5.46 (10.52) | 5.42 (9.75) |
| Service, n (%) | |||||
| Medicine | 184,495 (57.1) | 27,190 (49.8) | 157,305 (58.6) | 184,496 (94.0) | |
| Surgery | 126,614 (39.2) | 25,602 (46.9) | 101,012 (37.7) | 126,614 (100%) | |
| Neurology | 11,829 (3.7) | 1,853 (3.4) | 9,976 (3.7) | 11,829 (6.0) | |
To explore the effects of dependence among observations from patients with multiple encounters, we compared parameter estimates derived from a model with all patient encounters (including repeated admissions for the same patient) with those from a model with a randomly sampled single visit per patient, and observed that there was no difference in parameter estimates between the 2 classes of models. For all analyses, we used a type I error of 5% (2 sided) to test for statistical significance using SAS version 9.3 (SAS Institute, Cary, NC) or R software (
RESULTS
We included 322,938 patient admissions. Of this sample, 54,645 (16.9%) had spent at least 1 night in the ICU. Overall, 76,758 patients (23.8%) had diabetes, representing 26.3% of ICU patients, 23.2% of non‐ICU patients, 28.2% of medical patients, and 16.8% of surgical patients (see Table 1 for demographic characteristics).
Mortality Trends Within Strata
Among ICU patients, the overall mortality rate was 9.9%: 10.5% of patients with diabetes and 9.8% of patients without diabetes. Among non‐ICU patients, the overall mortality rate was 0.8%: 0.9% of patients with diabetes and 0.7% of patients without diabetes.
Among medical patients, the overall mortality rate was 2.9%: 3.1% of patients with diabetes and 2.8% of patients without diabetes. Among surgical patients, the overall mortality rate was 1.4%: 1.8% of patients with diabetes and 1.4% of patients without diabetes. Figure 1 shows quarterly in‐hospital mortality for patients with and without diabetes from 2000 to 2010 stratified by ICU status and by service assignment.
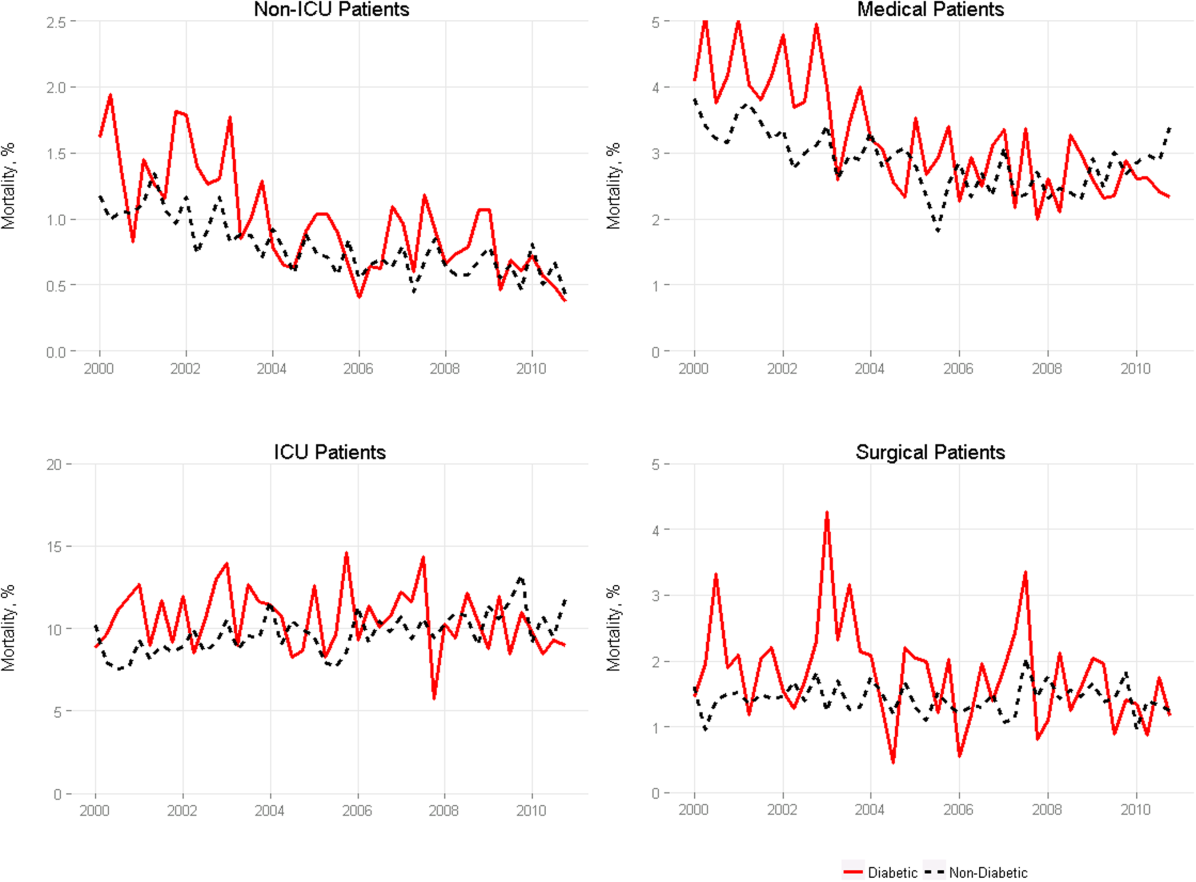
Table 2 describes the difference‐in‐differences regression analyses, stratified by ICU status and service assignment. Among ICU patients (Table 2, model 1), each successive year was associated with a 2.6% relative reduction in the adjusted odds of mortality (odds ratio [OR]: 0.974, 95% confidence interval [CI]: 0.963‐0.985) for patients without diabetes compared to a 7.8% relative reduction for those with diabetes (OR: 0.923, 95% CI: 0.906‐0.940). In other words, patients with diabetes compared to patients without diabetes had a significantly greater decline in odds of adjusted mortality of 5.3% per year (OR: 0.947, 95% CI: 0.927‐0.967). As a result, the adjusted odds of mortality among patients with versus without diabetes decreased from 1.352 in 2000 to 0.772 in 2010.
| Independent Variables | ICU Patients, N=54,646, OR (95% CI) | Non‐ICU Patients, N=268,293, OR (95% CI) | Medical Patients, N=196,325, OR (95% CI) | Surgical Patients, N=126,614, OR (95% CI) |
|---|---|---|---|---|
| Model 1 | Model 2 | Model 3 | Model 4 | |
| ||||
| Year | 0.974 (0.963‐0.985) | 0.925 (0.909‐0.940) | 0.943 (0.933‐0.954) | 0.995 (0.977‐1.103) |
| Diabetes | 1.352 (1.562‐1.171) | 0.958 (0.783‐1.173) | 1.186 (1.037‐1.356) | 1.213 (0.942‐1.563) |
| Diabetes*year | 0.947 (0.927‐0.967) | 0.977 (0.946‐1.008) | 0.961 (0.942‐0.980) | 0.955 (0.918‐0.994) |
| C statistic | 0.812 | 0.907 | 0.880 | 0.919 |
Among non‐ICU patients (Table 2, model 2), each successive year was associated with a 7.5% relative reduction in the adjusted odds of mortality (OR: 0.925, 95% CI: 0.909‐0.940) for patients without diabetes compared to a 9.6% relative reduction for those with diabetes (OR: 0.904, 95% CI: 0.879‐0.929); this greater decline in odds of adjusted mortality of 2.3% per year (OR: 0.977, 95% CI: 0.946‐1.008; P=0.148) was not statistically significant.
We found greater decline in odds of mortality among patients with diabetes than among patients without diabetes over time in both medical patients (3.9% greater decline per year; OR: 0.961, 95% CI: 0.942‐0.980) and surgical patients (4.5% greater decline per year; OR: 0.955, 95% CI: 0.918‐0.994), without a difference between the 2. Detailed results are shown in Table 2, models 3 and 4.
Glycemic Control
Among ICU patients with diabetes (N=14,364), at least 2 inpatient point‐of‐care glucose readings were available for 13,136 (91.5%), with a mean of 4.67 readings per day, whereas hemoglobin A1c data were available for only 5321 patients (37.0%). Both inpatient glucose data and hemoglobin A1c were available for 4989 patients (34.7%). Figure 2 shows trends in inpatient and outpatient glycemic control measures among ICU patients with diabetes over the study period. Mean hemoglobin A1c decreased from 7.7 in 2000 to 7.3 in 2010. Mean hospitalization glucose began at 187.2, reached a nadir of 162.4 in the third quarter (Q3) of 2007, and rose subsequently to 174.4 with loosened glucose control targets. Standard deviation of mean glucose and percentage of patient‐days with a severe hyperglycemic episode followed a similar pattern, though with nadirs in Q4 2007 and Q2 2008, respectively, whereas percentage of patient‐days with a hypoglycemic episode rose from 1.46% in 2000, peaked at 3.00% in Q3 2005, and returned to 2.15% in 2010. All changes in glucose control are significant with P<0.001.
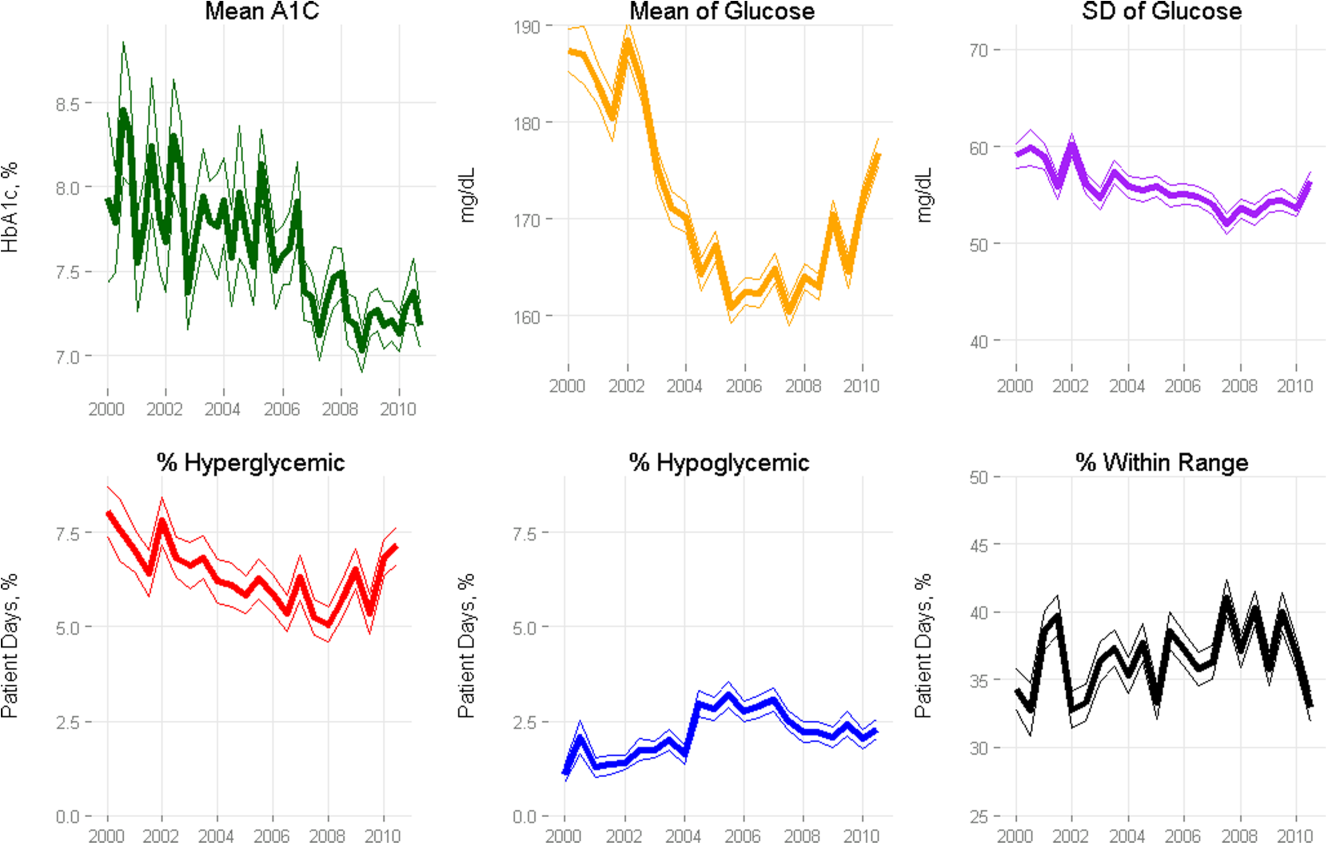
Mortality Trends and Glycemic Control
To determine whether glucose control explained the excess decline in odds of mortality among patients with diabetes in the ICU, we restricted our sample to ICU patients with diabetes and examined the association of diabetes with mortality after including measures of glucose control.
We first verified that the overall adjusted mortality trend among ICU patients with diabetes for whom we had measures of inpatient glucose control was similar to that of the full sample of ICU patients with diabetes. Similar to the full sample, we found that the adjusted excess odds of death significantly declined by a relative 7.3% each successive year (OR: 0.927, 95% CI: 0.907‐0.947; Table 3, model 1). We then included measures of inpatient glucose control in the model and found, as expected, that a higher percentage of days with severe hyperglycemia and with hypoglycemia was associated with an increased odds of death (P<0.001 for both; Table 3, model 2). Nonetheless, after including measures of inpatient glucose control, we found that the rate of change of excess odds of death for patients with diabetes was unchanged (OR: 0.926, 95% CI: 0.905‐0.947).
| Patients With Inpatient Glucose Control Measures, n=13,136 | Patients With Inpatient and Outpatient Glucose Control Measures, n=4,989 | ||||
|---|---|---|---|---|---|
| Independent Variables | Model 1, OR (95% CI) | Model 2, OR (95% CI) | Model 3, OR (95% CI) | Model 4, OR (95% CI) | Model 5, OR (95% CI) |
| |||||
| Year | 0.927 (0.907‐0.947) | 0.926 (0.905‐0.947) | 0.958 (0.919‐0.998) | 0.956 (0.916‐0.997) | 0.953 (0.914‐0.994) |
| % Severe hyperglycemic days | 1.016 (1.010‐1.021) | 1.009 (0.998‐1.020) | 1.010 (0.999‐1.021) | ||
| % Hypoglycemic days | 1.047 (1.040‐1.055) | 1.051 (1.037‐1.065) | 1.049 (1.036‐1.063) | ||
| % Normoglycemic days | 0.997 (0.994‐1.000) | 0.994 (0.989‐0.999) | 0.993 (0.988‐0.998) | ||
| SD of mean glucose | 0.996 (0.992‐1.000) | 0.993 (0.986‐1.000) | 0.994 (0.987‐1.002) | ||
| Mean HbA1c | 0.892 (0.828‐0.961) | ||||
| C statistic | 0.806 | 0.825 | 0.825 | 0.838 | 0.841 |
We then restricted our sample to patients with diabetes with both inpatient and outpatient glycemic control data and found that, in this subpopulation, the adjusted excess odds of death among patients with diabetes relative to those without significantly declined by a relative 4.2% each progressive year (OR: 0.958, 95% CI: 0.918‐0.998; Table 3, model 3). Including measures of inpatient glucose control in the model did not significantly change the rate of change of excess odds of death (OR: 0.956, 95% CI: 0.916‐0.997; Table 3, model 4), nor did including both measures of inpatient and outpatient glycemic control (OR: 0.953, 95% CI: 0.914‐0.994; Table 3, model 5).
DISCUSSION
We conducted a difference‐in‐difference analysis of in‐hospital mortality rates among adult patients with diabetes compared to patients without diabetes over 10 years, stratifying by ICU status and service assignment. For patients with any ICU stay, we found that the reduction in odds of mortality for patients with diabetes has been 3 times larger than the reduction in odds of mortality for patients without diabetes. For those without an ICU stay, we found no significant difference between patients with and without diabetes in the rate at which in‐hospital mortality declined. We did not find stratification by assignment to a medical or surgical service to be an effect modifier. Finally, despite the fact that our institution achieved better aggregate inpatient glucose control, less severe hyperglycemia, and better long‐term glucose control over the course of the decade, we did not find that either inpatient or outpatient glucose control explained the trend in mortality for patients with diabetes in the ICU. Our study is unique in its inclusion of all hospitalized patients and its ability to simultaneously assess whether both inpatient and outpatient glucose control are explanatory factors in the observed mortality trends.
The fact that improved inpatient glucose control did not explain the trend in mortality for patients with diabetes in the ICU is consistent with the majority of the literature on intensive inpatient glucose control. In randomized trials, intensive glucose control appears to be of greater benefit for patients without diabetes than for patients with diabetes.[31] In fact, in 1 study, patients with diabetes were the only group that did not benefit from intensive glucose control.[32] In our study, it is possible that the rise in hypoglycemia nullified some of the benefits of glucose control. Nationally, hospital admissions for hypoglycemia among Medicare beneficiaries now outnumber admissions for hyperglycemia.[27]
We also do not find that the decline in hemoglobin A1c attenuated the reduction in mortality in the minority of patients for whom these data were available. This is concordant with evidence from 3 randomized clinical trials that have failed to establish a clear beneficial effect of intensive outpatient glucose control on primary cardiovascular endpoints among older, high‐risk patients with type 2 diabetes using glucose‐lowering agents.[21, 22, 23] It is notable, however, that the population for whom we had available hemoglobin A1c results was not representative of the overall population of ICU patients with diabetes. Consequently, there may be an association of outpatient glucose control with inpatient mortality in the overall population of ICU patients with diabetes that we were not able to detect.
The decline in mortality among ICU patients with diabetes in our study may stem from factors other than glycemic control. It is possible that patients were diagnosed earlier in their course of disease in later years of the study period, making the population of patients with diabetes younger or healthier. Of note, however, our risk adjustment models were very robust, with C statistics from 0.82 to 0.92, suggesting that we were able to account for much of the mortality risk attributable to patient clinical and demographic factors. More intensive glucose management may have nonglycemic benefits, such as closer patient observation, which may themselves affect mortality. Alternatively, improved cardiovascular management for patients with diabetes may have decreased the incidence of cardiovascular events. During the study period, evidence from large clinical trials demonstrated the importance of tight blood pressure and lipid management in improving outcomes for patients with diabetes,[33, 34, 35, 36] guidelines for lipid management for patients with diabetes changed,[37] and fewer patients developed cardiovascular complications.[38] Finally, it is possible that our findings can be explained by an improvement in treatment of complications for which patients with diabetes previously have had disproportionately worse outcomes, such as percutaneous coronary intervention.[39]
Our findings may have important implications for both clinicians and policymakers. Changes in inpatient glucose management have required substantial additional resources on the part of hospitals. Our evidence regarding the questionable impact of inpatient glucose control on in‐hospital mortality trends for patients with diabetes is disappointing and highlights the need for multifaceted evaluation of the impact of such quality initiatives. There may, for instance, be benefits from tighter blood glucose control in the hospital beyond mortality, such as reduced infections, costs, or length of stay. On the outpatient side, our more limited data are consistent with recent studies that have not been able to show a mortality benefit in older diabetic patients from more stringent glycemic control. A reassessment of prevailing diabetes‐related quality measures, as recently called for by some,[40, 41] seems reasonable.
Our study must be interpreted in light of its limitations. It is possible that the improvements in glucose management were too small to result in a mortality benefit. The overall reduction of 25 mg dL achieved at our institution is less than the 33 to 50 mg/dL difference between intensive and conventional groups in those randomized clinical trials that have found reductions in mortality.[11, 42] In addition, an increase in mean glucose during the last 1 to 2 years of the observation period (in response to prevailing guidelines) could potentially have attenuated any benefit on mortality. The study does not include other important clinical endpoints, such as infections, complications, length of stay, and hospital costs. Additionally, we did not examine postdischarge mortality, which might have shown a different pattern. The small proportion of patients with hemoglobin A1c results may have hampered our ability to detect an effect of outpatient glucose control. Consequently, our findings regarding outpatient glucose control are only suggestive. Finally, our findings represent the experience of a single, large academic medical center and may not be generalizable to all settings.
Overall, we found that patients with diabetes in the ICU have experienced a disproportionate reduction in in‐hospital mortality over time that does not appear to be explained by improvements in either inpatient or outpatient glucose control. Although improved glycemic control may have other benefits, it does not appear to impact in‐hospital mortality. Our real‐world empirical results contribute to the discourse among clinicians and policymakers with regards to refocusing the approach to managing glucose in‐hospital and readjudication of diabetes‐related quality measures.
Acknowledgments
The authors would like to acknowledge the YaleNew Haven Hospital diabetes management team: Gael Ulisse, APRN, Helen Psarakis, APRN, Anne Kaisen, APRN, and the Yale Endocrine Fellows.
Disclosures: Design and conduct of the study: N. B., J. D., S. I., T. B., L. H. Collection, management, analysis, and interpretation of the data: N. B., B. J., J. D., J. R., J. B., S. I., L. H. Preparation, review, or approval of the manuscript: N. B., B. J., J. D., J. R., S. I., T. B., L. H. Leora Horwitz, MD, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. Dr. Horwitz is supported by the National Institute on Aging (K08 AG038336) and by the American Federation for Aging Research through the Paul B. Beeson Career Development Award Program. This publication was also made possible by CTSA grant number UL1 RR024139 from the National Center for Research Resources and the National Center for Advancing Translational Science, components of the National Institutes of Health (NIH), and NIH roadmap for Medical Research. Its contents are solely the responsibility of the authors and do not necessarily represent the official view of the NIH. No funding source had any role in design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; or decision to submit the manuscript for publication. Silvio E. Inzucchi, MD, serves on a Data Safety Monitoring Board for Novo Nordisk, a manufacturer of insulin products used in the hospital setting. The remaining authors declare no conflicts of interest.
- National Diabetes Information Clearinghouse. National Diabetes Statistics; 2011. Available at: http://diabetes.niddk.nih.gov/dm/pubs/america/index.aspx. Accessed November 12, 2013.
- Healthcare Cost and Utilization Project. Statistical brief #93; 2010. Available at: http://www.hcup‐us.ahrq.gov/reports/statbriefs/sb93.pdf. Accessed November 12, 2013.
- , , , et al. Association between diabetes mellitus and post‐discharge outcomes in patients hospitalized with heart failure: findings from the EVEREST trial. Eur J Heart Fail. 2013;15(2):194–202.
- , , , et al. Influence of diabetes mellitus on clinical outcome in the thrombolytic era of acute myocardial infarction. GUSTO‐I Investigators. Global Utilization of Streptokinase and Tissue Plasminogen Activator for Occluded Coronary Arteries. J Am Coll Cardiol. 1997;30(1):171–179.
- , , , , , . Type 2 diabetes and pneumonia outcomes: a population‐based cohort study. Diabetes Care. 2007;30(9):2251–2257.
- , , , . Prevalence and outcomes of diabetes, hypertension and cardiovascular disease in COPD. Eur Respir J. 2008;32(4):962–969.
- , , , , . The role of body mass index and diabetes in the development of acute organ failure and subsequent mortality in an observational cohort. Crit Care. 2006;10(5):R137.
- , , , , , . Type 2 diabetes and 1‐year mortality in intensive care unit patients. Eur J Clin Invest. 2013;43(3):238–247.
- , , . Excess mortality during hospital stays among patients with recorded diabetes compared with those without diabetes. Diabet Med. 2013;30(12):1393–1402.
- , , , et al. Decade‐long trends in mortality among patients with and without diabetes mellitus at a major academic medical center. JAMA Intern Med. 2014;174(7):1187–1188.
- , , , et al. Intensive insulin therapy in critically ill patients. N Engl J Med. 2001;345(19):1359–1367.
- , , , et al. Intensive versus conventional glucose control in critically ill patients. N Engl J Med. 2009;360(13):1283–1297.
- , , , et al. A prospective randomised multi‐centre controlled trial on tight glucose control by intensive insulin therapy in adult intensive care units: the Glucontrol study. Intensive Care Med. 2009;35(10):1738–1748.
- , , , et al. Intensive versus conventional insulin therapy: a randomized controlled trial in medical and surgical critically ill patients. Crit Care Med. 2008;36(12):3190–3197.
- , , , et al. Intensive insulin therapy in the medical ICU. N Engl J Med. 2006;354(5):449–461.
- , , , et al. Glycemic control in non‐critically ill hospitalized patients: a systematic review and meta‐analysis. J Clin Endocrinol Metab. 2012;97(1):49–58.
- , , , et al. American Association of Clinical Endocrinologists and American Diabetes Association consensus statement on inpatient glycemic control. Diabetes Care. 2009;32(6):1119–1131.
- Agency for Healthcare Research and Quality National Quality Measures Clearinghouse. Percent of cardiac surgery patients with controlled 6 A.M. postoperative blood glucose; 2012. Available at: http://www.qualitymeasures.ahrq.gov/content.aspx?id=35532. Accessed November 12, 2013.
- The effect of intensive treatment of diabetes on the development and progression of long‐term complications in insulin‐dependent diabetes mellitus. The Diabetes Control and Complications Trial Research Group. N Engl J Med. 1993;329(14):977–986.
- , , , et al. Intensive blood‐glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). Lancet. 1998;352(9131):837–853.
- Effects of intensive glucose lowering in type 2 diabetes. N Engl J Med. 2008;358(24):2545–2559.
- , , , et al. Glucose control and vascular complications in veterans with type 2 diabetes. N Engl J Med. 2009;360(2):129–139.
- , , , et al. Intensive blood glucose control and vascular outcomes in patients with type 2 diabetes. N Engl J Med. 2008;358(24):2560–2572.
- . Standards of medical care in diabetes—2014. Diabetes Care. 2014;37(suppl 1):S14–S80.
- National Committee for Quality Assurance. HEDIS 2013. Available at: http://www.ncqa.org/HEDISQualityMeasurement.aspx. Accessed November 12, 2013.
- , , , . Is glycemic control improving in US adults? Diabetes Care. 2008;31(1):81–86.
- , , , et al. National trends in US hospital admissions for hyperglycemia and hypoglycemia among medicare beneficiaries, 1999 to 2011. JAMA Intern Med. 2014;174(7):1116–1124.
- , , , et al. "Glucometrics"—assessing the quality of inpatient glucose management. Diabetes Technol Ther. 2006;8(5):560–569.
- , , , , . A modification of the Elixhauser comorbidity measures into a point system for hospital death using administrative data. Med Care. 2009;47(6):626–633.
- Healthcare Cost and Utilization Project. Clinical Classifications Software (CCS) for ICD‐9‐CM; 2013. Available at: http://www.hcup‐us.ahrq.gov/toolssoftware/ccs/ccs.jsp. Accessed November 12, 2013.
- , , , , . The impact of premorbid diabetic status on the relationship between the three domains of glycemic control and mortality in critically ill patients. Curr Opin Clin Nutr Metab Care. 2012;15(2):151–160.
- , , , et al. Intensive insulin therapy in mixed medical/surgical intensive care units: benefit versus harm. Diabetes. 2006;55(11):3151–3159.
- Tight blood pressure control and risk of macrovascular and microvascular complications in type 2 diabetes: UKPDS 38. UK Prospective Diabetes Study Group. BMJ. 1998;317(7160):703–713.
- , , , et al. Effects of a fixed combination of perindopril and indapamide on macrovascular and microvascular outcomes in patients with type 2 diabetes mellitus (the ADVANCE trial): a randomised controlled trial. Lancet. 2007;370(9590):829–840.
- , , , , . MRC/BHF heart protection study of cholesterol‐lowering with simvastatin in 5963 people with diabetes: a randomised placebo‐controlled trial. Lancet. 2003;361(9374):2005–2016.
- , , , et al. Primary prevention of cardiovascular disease with atorvastatin in type 2 diabetes in the Collaborative Atorvastatin Diabetes Study (CARDS): multicentre randomised placebo‐controlled trial. Lancet. 2004;364(9435):685–696.
- , , , . Expert panel on detection, evaluation and treatment of high blood cholesterol in adults. Executive summary of the third report of the national cholesterol education program (NCEP) adult treatment panel (atp III). JAMA. 2001;285(19):2486–2497.
- , , , et al. Changes in diabetes‐related complications in the United States, 1990–2010. N Engl J Med. 2014;370(16):1514–1523.
- , , . Coronary heart disease in patients with diabetes: part II: recent advances in coronary revascularization. J Am Coll Cardiol. 2007;49(6):643–656.
- , , , et al. Management of hyperglycemia in type 2 diabetes: a patient‐centered approach position statement of the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetes Care. 2012;35(6):1364–1379.
- , , , , . Assessing potential glycemic overtreatment in persons at hypoglycemic risk. JAMA Intern Med. 2013;174(2):259–268.
- , , , . Glycometabolic state at admission: important risk marker of mortality in conventionally treated patients with diabetes mellitus and acute myocardial infarction: long‐term results from the Diabetes and Insulin‐Glucose Infusion in Acute Myocardial Infarction (DIGAMI) study. Circulation. 1999;99(20):2626–2632.
Patients with diabetes currently comprise over 8% of the US population (over 25 million people) and more than 20% of hospitalized patients.[1, 2] Hospitalizations of patients with diabetes account for 23% of total hospital costs in the United States,[2] and patients with diabetes have worse outcomes after hospitalization for a variety of common medical conditions,[3, 4, 5, 6] as well as in intensive care unit (ICU) settings.[7, 8] Individuals with diabetes have historically experienced higher inpatient mortality than individuals without diabetes.[9] However, we recently reported that patients with diabetes at our large academic medical center have experienced a disproportionate reduction in in‐hospital mortality relative to patients without diabetes over the past decade.[10] This surprising trend begs further inquiry.
Improvement in in‐hospital mortality among patients with diabetes may stem from improved inpatient glycemic management. The landmark 2001 study by van den Berghe et al. demonstrating that intensive insulin therapy reduced postsurgical mortality among ICU patients ushered in an era of intensive inpatient glucose control.[11] However, follow‐up multicenter studies have not been able to replicate these results.[12, 13, 14, 15] In non‐ICU and nonsurgical settings, intensive glucose control has not yet been shown to have any mortality benefit, although it may impact other morbidities, such as postoperative infections.[16] Consequently, less stringent glycemic targets are now recommended.[17] Nonetheless, hospitals are being held accountable for certain aspects of inpatient glucose control. For example, the Centers for Medicare & Medicaid Services (CMS) began asking hospitals to report inpatient glucose control in cardiac surgery patients in 2004.[18] This measure is now publicly reported, and as of 2013 is included in the CMS Value‐Based Purchasing Program, which financially penalizes hospitals that do not meet targets.
Outpatient diabetes standards have also evolved in the past decade. The Diabetes Control and Complications Trial in 1993 and the United Kingdom Prospective Diabetes Study in 1997 demonstrated that better glycemic control in type 1 and newly diagnosed type 2 diabetes patients, respectively, improved clinical outcomes, and prompted guidelines for pharmacologic treatment of diabetic patients.[19, 20] However, subsequent randomized clinical trials have failed to establish a clear beneficial effect of intensive glucose control on primary cardiovascular endpoints among higher‐risk patients with longstanding type 2 diabetes,[21, 22, 23] and clinical practice recommendations now accept a more individualized approach to glycemic control.[24] Nonetheless, clinicians are also being held accountable for outpatient glucose control.[25]
To better understand the disproportionate reduction in mortality among hospitalized patients with diabetes that we observed, we first examined whether it was limited to surgical patients or patients in the ICU, the populations that have been demonstrated to benefit from intensive inpatient glucose control. Furthermore, given recent improvements in inpatient and outpatient glycemic control,[26, 27] we examined whether inpatient or outpatient glucose control explained the mortality trends. Results from this study contribute empirical evidence on real‐world effects of efforts to improve inpatient and outpatient glycemic control.
METHODS
Setting
During the study period, YaleNew Haven Hospital (YNHH) was an urban academic medical center in New Haven, Connecticut, with over 950 beds and an average of approximately 32,000 annual adult nonobstetric admissions. YNHH conducted a variety of inpatient glucose control initiatives during the study period. The surgical ICU began an informal medical teamdirected insulin infusion protocol in 2000 to 2001. In 2002, the medical ICU instituted a formal insulin infusion protocol with a target of 100 to 140 mg/dL, which spread to remaining hospital ICUs by the end of 2003. In 2005, YNHH launched a consultative inpatient diabetes management team to assist clinicians in controlling glucose in non‐ICU patients with diabetes. This team covered approximately 10 to 15 patients at a time and consisted of an advanced‐practice nurse practitioner, a supervising endocrinologist and endocrinology fellow, and a nurse educator to provide diabetic teaching. Additionally, in 2005, basal‐boluscorrection insulin order sets became available. The surgical ICU implemented a stringent insulin infusion protocol with target glucose of 80 to 110 mg/dL in 2006, but relaxed it (goal 80150 mg/dL) in 2007. Similarly, in 2006, YNHH made ICU insulin infusion recommendations more stringent in remaining ICUs (goal 90130 mg/dL), but relaxed them in 2010 (goal 120160 mg/dL), based on emerging data from clinical trials and prevailing national guidelines.
Participants and Data Sources
We included all adult, nonobstetric discharges from YNHH between January 1, 2000 and December 31, 2010. Repeat visits by the same patient were linked by medical record number. We obtained data from YNHH administrative billing, laboratory, and point‐of‐care capillary blood glucose databases. The Yale Human Investigation Committee approved our study design and granted a Health Insurance Portability and Accountability Act waiver and a waiver of patient consent.
Variables
Our primary endpoint was in‐hospital mortality. The primary exposure of interest was whether a patient had diabetes mellitus, defined as the presence of International Classification of Diseases, Ninth Revision codes 249.x, 250.x, V4585, V5391, or V6546 in any of the primary or secondary diagnosis codes in the index admission, or in any hospital encounter in the year prior to the index admission.
We assessed 2 effect‐modifying variables: ICU status (as measured by a charge for at least 1 night in the ICU) and service assignment to surgery (including neurosurgery and orthopedics), compared to medicine (including neurology). Independent explanatory variables included time between the start of the study and patient admission (measured as days/365), diabetes status, inpatient glucose control, and long‐term glucose control (as measured by hemoglobin A1c at any time in the 180 days prior to hospital admission in order to have adequate sample size). We assessed inpatient blood glucose control through point‐of‐care blood glucose meters (OneTouch SureStep; LifeScan, Inc., Milipitas, CA) at YNHH. We used 4 validated measures of inpatient glucose control: the proportion of days in each hospitalization in which there was any hypoglycemic episode (blood glucose value <70 mg/dL), the proportion of days in which there was any severely hyperglycemic episode (blood glucose value >299 mg/dL), the proportion of days in which mean blood glucose was considered to be within adequate control (all blood glucose values between 70 and 179 mg/dL), and the standard deviation of mean glucose during hospitalization as a measure of glycemic variability.[28]
Covariates included gender, age at time of admission, length of stay in days, race (defined by hospital registration), payer, Elixhauser comorbidity dummy variables (revised to exclude diabetes and to use only secondary diagnosis codes),[29] and primary discharge diagnosis grouped using Clinical Classifications Software,[30] based on established associations with in‐hospital mortality.
Statistical Analysis
We summarized demographic characteristics numerically and graphically for patients with and without diabetes and compared them using [2] and t tests. We summarized changes in inpatient and outpatient measures of glucose control over time numerically and graphically, and compared across years using the Wilcoxon rank sum test adjusted for multiple hypothesis testing.
We stratified all analyses first by ICU status and then by service assignment (medicine vs surgery). Statistical analyses within each stratum paralleled our previous approach to the full study cohort.[10] Taking each stratum separately (ie, only ICU patients or only medicine patients), we used a difference‐in‐differences approach comparing changes over time in in‐hospital mortality among patients with diabetes compared to those without diabetes. This approach enabled us to determine whether patients with diabetes had a different time trend in risk of in‐hospital mortality than those without diabetes. That is, for each stratum, we constructed multivariate logistic regression models including time in years, diabetes status, and the interaction between time and diabetes status as well as the aforementioned covariates. We calculated odds of death and confidence intervals for each additional year for patients with diabetes by exponentiating the sum of parameter estimates for time and the diabetes‐time interaction term. We evaluated all 2‐way interactions between year or diabetes status and the covariates in a multiple degree of freedom likelihood ratio test. We investigated nonlinearity of the relation between mortality and time by evaluating first and second‐order polynomials.
Because we found a significant decline in mortality risk for patients with versus without diabetes among ICU patients but not among non‐ICU patients, and because service assignment was not found to be an effect modifier, we then limited our sample to ICU patients with diabetes to better understand the role of inpatient and outpatient glucose control in accounting for observed mortality trends. First, we determined the relation between the measures of inpatient glucose control and changes in mortality over time using logistic regression. Then, we repeated this analysis in the subsets of patients who had inpatient glucose data and both inpatient and outpatient glycemic control data, adding inpatient and outpatient measures sequentially. Given the high level of missing outpatient glycemic control data, we compared demographic characteristics for diabetic ICU patients with and without such data using [2] and t tests, and found that patients with data were younger and less likely to be white and had longer mean length of stay, slightly worse performance on several measures of inpatient glucose control, and lower mortality (see Supporting Table 1 in the online version of this article).
| Characteristic | Overall, N=322,939 | Any ICU Stay, N=54,646 | No ICU Stay, N=268,293 | Medical Service, N=196,325 | Surgical Service, N=126,614 |
|---|---|---|---|---|---|
| |||||
| Died during admission, n (%) | 7,587 (2.3) | 5,439 (10.0) | 2,147 (0.8) | 5,705 (2.9) | 1,883 (1.5) |
| Diabetes, n (%) | 76,758 (23.8) | 14,364 (26.3) | 62,394 (23.2) | 55,453 (28.2) | 21,305 (16.8) |
| Age, y, mean (SD) | 55.5 (20.0) | 61.0 (17.0) | 54.4 (21.7) | 60.3 (18.9) | 48.0 (23.8) |
| Age, full range (interquartile range) | 0118 (4273) | 18112 (4975) | 0118 (4072) | 0118 (4776) | 0111 (3266) |
| Female, n (%) | 159,227 (49.3) | 23,208 (42.5) | 134,296 (50.1) | 99,805 (50.8) | 59,422 (46.9) |
| White race, n (%) | 226,586 (70.2) | 41,982 (76.8) | 184,604 (68.8) | 132,749 (67.6) | 93,838 (74.1) |
| Insurance, n (%) | |||||
| Medicaid | 54,590 (16.9) | 7,222 (13.2) | 47,378 (17.7) | 35,229 (17.9) | 19,361 (15.3) |
| Medicare | 141,638 (43.9) | 27,458 (50.2) | 114,180 (42.6) | 100,615 (51.2) | 41,023 (32.4) |
| Commercial | 113,013 (35.0) | 18,248 (33.4) | 94,765 (35.3) | 53,510 (27.2) | 59,503 (47.0) |
| Uninsured | 13,521 (4.2) | 1,688 (3.1) | 11,833 (4.4) | 6,878 (3.5) | 6,643 (5.2) |
| Length of stay, d, mean (SD) | 5.4 (9.5) | 11.8 (17.8) | 4.2 (6.2) | 5.46 (10.52) | 5.42 (9.75) |
| Service, n (%) | |||||
| Medicine | 184,495 (57.1) | 27,190 (49.8) | 157,305 (58.6) | 184,496 (94.0) | |
| Surgery | 126,614 (39.2) | 25,602 (46.9) | 101,012 (37.7) | 126,614 (100%) | |
| Neurology | 11,829 (3.7) | 1,853 (3.4) | 9,976 (3.7) | 11,829 (6.0) | |
To explore the effects of dependence among observations from patients with multiple encounters, we compared parameter estimates derived from a model with all patient encounters (including repeated admissions for the same patient) with those from a model with a randomly sampled single visit per patient, and observed that there was no difference in parameter estimates between the 2 classes of models. For all analyses, we used a type I error of 5% (2 sided) to test for statistical significance using SAS version 9.3 (SAS Institute, Cary, NC) or R software (
RESULTS
We included 322,938 patient admissions. Of this sample, 54,645 (16.9%) had spent at least 1 night in the ICU. Overall, 76,758 patients (23.8%) had diabetes, representing 26.3% of ICU patients, 23.2% of non‐ICU patients, 28.2% of medical patients, and 16.8% of surgical patients (see Table 1 for demographic characteristics).
Mortality Trends Within Strata
Among ICU patients, the overall mortality rate was 9.9%: 10.5% of patients with diabetes and 9.8% of patients without diabetes. Among non‐ICU patients, the overall mortality rate was 0.8%: 0.9% of patients with diabetes and 0.7% of patients without diabetes.
Among medical patients, the overall mortality rate was 2.9%: 3.1% of patients with diabetes and 2.8% of patients without diabetes. Among surgical patients, the overall mortality rate was 1.4%: 1.8% of patients with diabetes and 1.4% of patients without diabetes. Figure 1 shows quarterly in‐hospital mortality for patients with and without diabetes from 2000 to 2010 stratified by ICU status and by service assignment.

Table 2 describes the difference‐in‐differences regression analyses, stratified by ICU status and service assignment. Among ICU patients (Table 2, model 1), each successive year was associated with a 2.6% relative reduction in the adjusted odds of mortality (odds ratio [OR]: 0.974, 95% confidence interval [CI]: 0.963‐0.985) for patients without diabetes compared to a 7.8% relative reduction for those with diabetes (OR: 0.923, 95% CI: 0.906‐0.940). In other words, patients with diabetes compared to patients without diabetes had a significantly greater decline in odds of adjusted mortality of 5.3% per year (OR: 0.947, 95% CI: 0.927‐0.967). As a result, the adjusted odds of mortality among patients with versus without diabetes decreased from 1.352 in 2000 to 0.772 in 2010.
| Independent Variables | ICU Patients, N=54,646, OR (95% CI) | Non‐ICU Patients, N=268,293, OR (95% CI) | Medical Patients, N=196,325, OR (95% CI) | Surgical Patients, N=126,614, OR (95% CI) |
|---|---|---|---|---|
| Model 1 | Model 2 | Model 3 | Model 4 | |
| ||||
| Year | 0.974 (0.963‐0.985) | 0.925 (0.909‐0.940) | 0.943 (0.933‐0.954) | 0.995 (0.977‐1.103) |
| Diabetes | 1.352 (1.562‐1.171) | 0.958 (0.783‐1.173) | 1.186 (1.037‐1.356) | 1.213 (0.942‐1.563) |
| Diabetes*year | 0.947 (0.927‐0.967) | 0.977 (0.946‐1.008) | 0.961 (0.942‐0.980) | 0.955 (0.918‐0.994) |
| C statistic | 0.812 | 0.907 | 0.880 | 0.919 |
Among non‐ICU patients (Table 2, model 2), each successive year was associated with a 7.5% relative reduction in the adjusted odds of mortality (OR: 0.925, 95% CI: 0.909‐0.940) for patients without diabetes compared to a 9.6% relative reduction for those with diabetes (OR: 0.904, 95% CI: 0.879‐0.929); this greater decline in odds of adjusted mortality of 2.3% per year (OR: 0.977, 95% CI: 0.946‐1.008; P=0.148) was not statistically significant.
We found greater decline in odds of mortality among patients with diabetes than among patients without diabetes over time in both medical patients (3.9% greater decline per year; OR: 0.961, 95% CI: 0.942‐0.980) and surgical patients (4.5% greater decline per year; OR: 0.955, 95% CI: 0.918‐0.994), without a difference between the 2. Detailed results are shown in Table 2, models 3 and 4.
Glycemic Control
Among ICU patients with diabetes (N=14,364), at least 2 inpatient point‐of‐care glucose readings were available for 13,136 (91.5%), with a mean of 4.67 readings per day, whereas hemoglobin A1c data were available for only 5321 patients (37.0%). Both inpatient glucose data and hemoglobin A1c were available for 4989 patients (34.7%). Figure 2 shows trends in inpatient and outpatient glycemic control measures among ICU patients with diabetes over the study period. Mean hemoglobin A1c decreased from 7.7 in 2000 to 7.3 in 2010. Mean hospitalization glucose began at 187.2, reached a nadir of 162.4 in the third quarter (Q3) of 2007, and rose subsequently to 174.4 with loosened glucose control targets. Standard deviation of mean glucose and percentage of patient‐days with a severe hyperglycemic episode followed a similar pattern, though with nadirs in Q4 2007 and Q2 2008, respectively, whereas percentage of patient‐days with a hypoglycemic episode rose from 1.46% in 2000, peaked at 3.00% in Q3 2005, and returned to 2.15% in 2010. All changes in glucose control are significant with P<0.001.

Mortality Trends and Glycemic Control
To determine whether glucose control explained the excess decline in odds of mortality among patients with diabetes in the ICU, we restricted our sample to ICU patients with diabetes and examined the association of diabetes with mortality after including measures of glucose control.
We first verified that the overall adjusted mortality trend among ICU patients with diabetes for whom we had measures of inpatient glucose control was similar to that of the full sample of ICU patients with diabetes. Similar to the full sample, we found that the adjusted excess odds of death significantly declined by a relative 7.3% each successive year (OR: 0.927, 95% CI: 0.907‐0.947; Table 3, model 1). We then included measures of inpatient glucose control in the model and found, as expected, that a higher percentage of days with severe hyperglycemia and with hypoglycemia was associated with an increased odds of death (P<0.001 for both; Table 3, model 2). Nonetheless, after including measures of inpatient glucose control, we found that the rate of change of excess odds of death for patients with diabetes was unchanged (OR: 0.926, 95% CI: 0.905‐0.947).
| Patients With Inpatient Glucose Control Measures, n=13,136 | Patients With Inpatient and Outpatient Glucose Control Measures, n=4,989 | ||||
|---|---|---|---|---|---|
| Independent Variables | Model 1, OR (95% CI) | Model 2, OR (95% CI) | Model 3, OR (95% CI) | Model 4, OR (95% CI) | Model 5, OR (95% CI) |
| |||||
| Year | 0.927 (0.907‐0.947) | 0.926 (0.905‐0.947) | 0.958 (0.919‐0.998) | 0.956 (0.916‐0.997) | 0.953 (0.914‐0.994) |
| % Severe hyperglycemic days | 1.016 (1.010‐1.021) | 1.009 (0.998‐1.020) | 1.010 (0.999‐1.021) | ||
| % Hypoglycemic days | 1.047 (1.040‐1.055) | 1.051 (1.037‐1.065) | 1.049 (1.036‐1.063) | ||
| % Normoglycemic days | 0.997 (0.994‐1.000) | 0.994 (0.989‐0.999) | 0.993 (0.988‐0.998) | ||
| SD of mean glucose | 0.996 (0.992‐1.000) | 0.993 (0.986‐1.000) | 0.994 (0.987‐1.002) | ||
| Mean HbA1c | 0.892 (0.828‐0.961) | ||||
| C statistic | 0.806 | 0.825 | 0.825 | 0.838 | 0.841 |
We then restricted our sample to patients with diabetes with both inpatient and outpatient glycemic control data and found that, in this subpopulation, the adjusted excess odds of death among patients with diabetes relative to those without significantly declined by a relative 4.2% each progressive year (OR: 0.958, 95% CI: 0.918‐0.998; Table 3, model 3). Including measures of inpatient glucose control in the model did not significantly change the rate of change of excess odds of death (OR: 0.956, 95% CI: 0.916‐0.997; Table 3, model 4), nor did including both measures of inpatient and outpatient glycemic control (OR: 0.953, 95% CI: 0.914‐0.994; Table 3, model 5).
DISCUSSION
We conducted a difference‐in‐difference analysis of in‐hospital mortality rates among adult patients with diabetes compared to patients without diabetes over 10 years, stratifying by ICU status and service assignment. For patients with any ICU stay, we found that the reduction in odds of mortality for patients with diabetes has been 3 times larger than the reduction in odds of mortality for patients without diabetes. For those without an ICU stay, we found no significant difference between patients with and without diabetes in the rate at which in‐hospital mortality declined. We did not find stratification by assignment to a medical or surgical service to be an effect modifier. Finally, despite the fact that our institution achieved better aggregate inpatient glucose control, less severe hyperglycemia, and better long‐term glucose control over the course of the decade, we did not find that either inpatient or outpatient glucose control explained the trend in mortality for patients with diabetes in the ICU. Our study is unique in its inclusion of all hospitalized patients and its ability to simultaneously assess whether both inpatient and outpatient glucose control are explanatory factors in the observed mortality trends.
The fact that improved inpatient glucose control did not explain the trend in mortality for patients with diabetes in the ICU is consistent with the majority of the literature on intensive inpatient glucose control. In randomized trials, intensive glucose control appears to be of greater benefit for patients without diabetes than for patients with diabetes.[31] In fact, in 1 study, patients with diabetes were the only group that did not benefit from intensive glucose control.[32] In our study, it is possible that the rise in hypoglycemia nullified some of the benefits of glucose control. Nationally, hospital admissions for hypoglycemia among Medicare beneficiaries now outnumber admissions for hyperglycemia.[27]
We also do not find that the decline in hemoglobin A1c attenuated the reduction in mortality in the minority of patients for whom these data were available. This is concordant with evidence from 3 randomized clinical trials that have failed to establish a clear beneficial effect of intensive outpatient glucose control on primary cardiovascular endpoints among older, high‐risk patients with type 2 diabetes using glucose‐lowering agents.[21, 22, 23] It is notable, however, that the population for whom we had available hemoglobin A1c results was not representative of the overall population of ICU patients with diabetes. Consequently, there may be an association of outpatient glucose control with inpatient mortality in the overall population of ICU patients with diabetes that we were not able to detect.
The decline in mortality among ICU patients with diabetes in our study may stem from factors other than glycemic control. It is possible that patients were diagnosed earlier in their course of disease in later years of the study period, making the population of patients with diabetes younger or healthier. Of note, however, our risk adjustment models were very robust, with C statistics from 0.82 to 0.92, suggesting that we were able to account for much of the mortality risk attributable to patient clinical and demographic factors. More intensive glucose management may have nonglycemic benefits, such as closer patient observation, which may themselves affect mortality. Alternatively, improved cardiovascular management for patients with diabetes may have decreased the incidence of cardiovascular events. During the study period, evidence from large clinical trials demonstrated the importance of tight blood pressure and lipid management in improving outcomes for patients with diabetes,[33, 34, 35, 36] guidelines for lipid management for patients with diabetes changed,[37] and fewer patients developed cardiovascular complications.[38] Finally, it is possible that our findings can be explained by an improvement in treatment of complications for which patients with diabetes previously have had disproportionately worse outcomes, such as percutaneous coronary intervention.[39]
Our findings may have important implications for both clinicians and policymakers. Changes in inpatient glucose management have required substantial additional resources on the part of hospitals. Our evidence regarding the questionable impact of inpatient glucose control on in‐hospital mortality trends for patients with diabetes is disappointing and highlights the need for multifaceted evaluation of the impact of such quality initiatives. There may, for instance, be benefits from tighter blood glucose control in the hospital beyond mortality, such as reduced infections, costs, or length of stay. On the outpatient side, our more limited data are consistent with recent studies that have not been able to show a mortality benefit in older diabetic patients from more stringent glycemic control. A reassessment of prevailing diabetes‐related quality measures, as recently called for by some,[40, 41] seems reasonable.
Our study must be interpreted in light of its limitations. It is possible that the improvements in glucose management were too small to result in a mortality benefit. The overall reduction of 25 mg dL achieved at our institution is less than the 33 to 50 mg/dL difference between intensive and conventional groups in those randomized clinical trials that have found reductions in mortality.[11, 42] In addition, an increase in mean glucose during the last 1 to 2 years of the observation period (in response to prevailing guidelines) could potentially have attenuated any benefit on mortality. The study does not include other important clinical endpoints, such as infections, complications, length of stay, and hospital costs. Additionally, we did not examine postdischarge mortality, which might have shown a different pattern. The small proportion of patients with hemoglobin A1c results may have hampered our ability to detect an effect of outpatient glucose control. Consequently, our findings regarding outpatient glucose control are only suggestive. Finally, our findings represent the experience of a single, large academic medical center and may not be generalizable to all settings.
Overall, we found that patients with diabetes in the ICU have experienced a disproportionate reduction in in‐hospital mortality over time that does not appear to be explained by improvements in either inpatient or outpatient glucose control. Although improved glycemic control may have other benefits, it does not appear to impact in‐hospital mortality. Our real‐world empirical results contribute to the discourse among clinicians and policymakers with regards to refocusing the approach to managing glucose in‐hospital and readjudication of diabetes‐related quality measures.
Acknowledgments
The authors would like to acknowledge the YaleNew Haven Hospital diabetes management team: Gael Ulisse, APRN, Helen Psarakis, APRN, Anne Kaisen, APRN, and the Yale Endocrine Fellows.
Disclosures: Design and conduct of the study: N. B., J. D., S. I., T. B., L. H. Collection, management, analysis, and interpretation of the data: N. B., B. J., J. D., J. R., J. B., S. I., L. H. Preparation, review, or approval of the manuscript: N. B., B. J., J. D., J. R., S. I., T. B., L. H. Leora Horwitz, MD, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. Dr. Horwitz is supported by the National Institute on Aging (K08 AG038336) and by the American Federation for Aging Research through the Paul B. Beeson Career Development Award Program. This publication was also made possible by CTSA grant number UL1 RR024139 from the National Center for Research Resources and the National Center for Advancing Translational Science, components of the National Institutes of Health (NIH), and NIH roadmap for Medical Research. Its contents are solely the responsibility of the authors and do not necessarily represent the official view of the NIH. No funding source had any role in design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; or decision to submit the manuscript for publication. Silvio E. Inzucchi, MD, serves on a Data Safety Monitoring Board for Novo Nordisk, a manufacturer of insulin products used in the hospital setting. The remaining authors declare no conflicts of interest.
Patients with diabetes currently comprise over 8% of the US population (over 25 million people) and more than 20% of hospitalized patients.[1, 2] Hospitalizations of patients with diabetes account for 23% of total hospital costs in the United States,[2] and patients with diabetes have worse outcomes after hospitalization for a variety of common medical conditions,[3, 4, 5, 6] as well as in intensive care unit (ICU) settings.[7, 8] Individuals with diabetes have historically experienced higher inpatient mortality than individuals without diabetes.[9] However, we recently reported that patients with diabetes at our large academic medical center have experienced a disproportionate reduction in in‐hospital mortality relative to patients without diabetes over the past decade.[10] This surprising trend begs further inquiry.
Improvement in in‐hospital mortality among patients with diabetes may stem from improved inpatient glycemic management. The landmark 2001 study by van den Berghe et al. demonstrating that intensive insulin therapy reduced postsurgical mortality among ICU patients ushered in an era of intensive inpatient glucose control.[11] However, follow‐up multicenter studies have not been able to replicate these results.[12, 13, 14, 15] In non‐ICU and nonsurgical settings, intensive glucose control has not yet been shown to have any mortality benefit, although it may impact other morbidities, such as postoperative infections.[16] Consequently, less stringent glycemic targets are now recommended.[17] Nonetheless, hospitals are being held accountable for certain aspects of inpatient glucose control. For example, the Centers for Medicare & Medicaid Services (CMS) began asking hospitals to report inpatient glucose control in cardiac surgery patients in 2004.[18] This measure is now publicly reported, and as of 2013 is included in the CMS Value‐Based Purchasing Program, which financially penalizes hospitals that do not meet targets.
Outpatient diabetes standards have also evolved in the past decade. The Diabetes Control and Complications Trial in 1993 and the United Kingdom Prospective Diabetes Study in 1997 demonstrated that better glycemic control in type 1 and newly diagnosed type 2 diabetes patients, respectively, improved clinical outcomes, and prompted guidelines for pharmacologic treatment of diabetic patients.[19, 20] However, subsequent randomized clinical trials have failed to establish a clear beneficial effect of intensive glucose control on primary cardiovascular endpoints among higher‐risk patients with longstanding type 2 diabetes,[21, 22, 23] and clinical practice recommendations now accept a more individualized approach to glycemic control.[24] Nonetheless, clinicians are also being held accountable for outpatient glucose control.[25]
To better understand the disproportionate reduction in mortality among hospitalized patients with diabetes that we observed, we first examined whether it was limited to surgical patients or patients in the ICU, the populations that have been demonstrated to benefit from intensive inpatient glucose control. Furthermore, given recent improvements in inpatient and outpatient glycemic control,[26, 27] we examined whether inpatient or outpatient glucose control explained the mortality trends. Results from this study contribute empirical evidence on real‐world effects of efforts to improve inpatient and outpatient glycemic control.
METHODS
Setting
During the study period, YaleNew Haven Hospital (YNHH) was an urban academic medical center in New Haven, Connecticut, with over 950 beds and an average of approximately 32,000 annual adult nonobstetric admissions. YNHH conducted a variety of inpatient glucose control initiatives during the study period. The surgical ICU began an informal medical teamdirected insulin infusion protocol in 2000 to 2001. In 2002, the medical ICU instituted a formal insulin infusion protocol with a target of 100 to 140 mg/dL, which spread to remaining hospital ICUs by the end of 2003. In 2005, YNHH launched a consultative inpatient diabetes management team to assist clinicians in controlling glucose in non‐ICU patients with diabetes. This team covered approximately 10 to 15 patients at a time and consisted of an advanced‐practice nurse practitioner, a supervising endocrinologist and endocrinology fellow, and a nurse educator to provide diabetic teaching. Additionally, in 2005, basal‐boluscorrection insulin order sets became available. The surgical ICU implemented a stringent insulin infusion protocol with target glucose of 80 to 110 mg/dL in 2006, but relaxed it (goal 80150 mg/dL) in 2007. Similarly, in 2006, YNHH made ICU insulin infusion recommendations more stringent in remaining ICUs (goal 90130 mg/dL), but relaxed them in 2010 (goal 120160 mg/dL), based on emerging data from clinical trials and prevailing national guidelines.
Participants and Data Sources
We included all adult, nonobstetric discharges from YNHH between January 1, 2000 and December 31, 2010. Repeat visits by the same patient were linked by medical record number. We obtained data from YNHH administrative billing, laboratory, and point‐of‐care capillary blood glucose databases. The Yale Human Investigation Committee approved our study design and granted a Health Insurance Portability and Accountability Act waiver and a waiver of patient consent.
Variables
Our primary endpoint was in‐hospital mortality. The primary exposure of interest was whether a patient had diabetes mellitus, defined as the presence of International Classification of Diseases, Ninth Revision codes 249.x, 250.x, V4585, V5391, or V6546 in any of the primary or secondary diagnosis codes in the index admission, or in any hospital encounter in the year prior to the index admission.
We assessed 2 effect‐modifying variables: ICU status (as measured by a charge for at least 1 night in the ICU) and service assignment to surgery (including neurosurgery and orthopedics), compared to medicine (including neurology). Independent explanatory variables included time between the start of the study and patient admission (measured as days/365), diabetes status, inpatient glucose control, and long‐term glucose control (as measured by hemoglobin A1c at any time in the 180 days prior to hospital admission in order to have adequate sample size). We assessed inpatient blood glucose control through point‐of‐care blood glucose meters (OneTouch SureStep; LifeScan, Inc., Milipitas, CA) at YNHH. We used 4 validated measures of inpatient glucose control: the proportion of days in each hospitalization in which there was any hypoglycemic episode (blood glucose value <70 mg/dL), the proportion of days in which there was any severely hyperglycemic episode (blood glucose value >299 mg/dL), the proportion of days in which mean blood glucose was considered to be within adequate control (all blood glucose values between 70 and 179 mg/dL), and the standard deviation of mean glucose during hospitalization as a measure of glycemic variability.[28]
Covariates included gender, age at time of admission, length of stay in days, race (defined by hospital registration), payer, Elixhauser comorbidity dummy variables (revised to exclude diabetes and to use only secondary diagnosis codes),[29] and primary discharge diagnosis grouped using Clinical Classifications Software,[30] based on established associations with in‐hospital mortality.
Statistical Analysis
We summarized demographic characteristics numerically and graphically for patients with and without diabetes and compared them using [2] and t tests. We summarized changes in inpatient and outpatient measures of glucose control over time numerically and graphically, and compared across years using the Wilcoxon rank sum test adjusted for multiple hypothesis testing.
We stratified all analyses first by ICU status and then by service assignment (medicine vs surgery). Statistical analyses within each stratum paralleled our previous approach to the full study cohort.[10] Taking each stratum separately (ie, only ICU patients or only medicine patients), we used a difference‐in‐differences approach comparing changes over time in in‐hospital mortality among patients with diabetes compared to those without diabetes. This approach enabled us to determine whether patients with diabetes had a different time trend in risk of in‐hospital mortality than those without diabetes. That is, for each stratum, we constructed multivariate logistic regression models including time in years, diabetes status, and the interaction between time and diabetes status as well as the aforementioned covariates. We calculated odds of death and confidence intervals for each additional year for patients with diabetes by exponentiating the sum of parameter estimates for time and the diabetes‐time interaction term. We evaluated all 2‐way interactions between year or diabetes status and the covariates in a multiple degree of freedom likelihood ratio test. We investigated nonlinearity of the relation between mortality and time by evaluating first and second‐order polynomials.
Because we found a significant decline in mortality risk for patients with versus without diabetes among ICU patients but not among non‐ICU patients, and because service assignment was not found to be an effect modifier, we then limited our sample to ICU patients with diabetes to better understand the role of inpatient and outpatient glucose control in accounting for observed mortality trends. First, we determined the relation between the measures of inpatient glucose control and changes in mortality over time using logistic regression. Then, we repeated this analysis in the subsets of patients who had inpatient glucose data and both inpatient and outpatient glycemic control data, adding inpatient and outpatient measures sequentially. Given the high level of missing outpatient glycemic control data, we compared demographic characteristics for diabetic ICU patients with and without such data using [2] and t tests, and found that patients with data were younger and less likely to be white and had longer mean length of stay, slightly worse performance on several measures of inpatient glucose control, and lower mortality (see Supporting Table 1 in the online version of this article).
| Characteristic | Overall, N=322,939 | Any ICU Stay, N=54,646 | No ICU Stay, N=268,293 | Medical Service, N=196,325 | Surgical Service, N=126,614 |
|---|---|---|---|---|---|
| |||||
| Died during admission, n (%) | 7,587 (2.3) | 5,439 (10.0) | 2,147 (0.8) | 5,705 (2.9) | 1,883 (1.5) |
| Diabetes, n (%) | 76,758 (23.8) | 14,364 (26.3) | 62,394 (23.2) | 55,453 (28.2) | 21,305 (16.8) |
| Age, y, mean (SD) | 55.5 (20.0) | 61.0 (17.0) | 54.4 (21.7) | 60.3 (18.9) | 48.0 (23.8) |
| Age, full range (interquartile range) | 0118 (4273) | 18112 (4975) | 0118 (4072) | 0118 (4776) | 0111 (3266) |
| Female, n (%) | 159,227 (49.3) | 23,208 (42.5) | 134,296 (50.1) | 99,805 (50.8) | 59,422 (46.9) |
| White race, n (%) | 226,586 (70.2) | 41,982 (76.8) | 184,604 (68.8) | 132,749 (67.6) | 93,838 (74.1) |
| Insurance, n (%) | |||||
| Medicaid | 54,590 (16.9) | 7,222 (13.2) | 47,378 (17.7) | 35,229 (17.9) | 19,361 (15.3) |
| Medicare | 141,638 (43.9) | 27,458 (50.2) | 114,180 (42.6) | 100,615 (51.2) | 41,023 (32.4) |
| Commercial | 113,013 (35.0) | 18,248 (33.4) | 94,765 (35.3) | 53,510 (27.2) | 59,503 (47.0) |
| Uninsured | 13,521 (4.2) | 1,688 (3.1) | 11,833 (4.4) | 6,878 (3.5) | 6,643 (5.2) |
| Length of stay, d, mean (SD) | 5.4 (9.5) | 11.8 (17.8) | 4.2 (6.2) | 5.46 (10.52) | 5.42 (9.75) |
| Service, n (%) | |||||
| Medicine | 184,495 (57.1) | 27,190 (49.8) | 157,305 (58.6) | 184,496 (94.0) | |
| Surgery | 126,614 (39.2) | 25,602 (46.9) | 101,012 (37.7) | 126,614 (100%) | |
| Neurology | 11,829 (3.7) | 1,853 (3.4) | 9,976 (3.7) | 11,829 (6.0) | |
To explore the effects of dependence among observations from patients with multiple encounters, we compared parameter estimates derived from a model with all patient encounters (including repeated admissions for the same patient) with those from a model with a randomly sampled single visit per patient, and observed that there was no difference in parameter estimates between the 2 classes of models. For all analyses, we used a type I error of 5% (2 sided) to test for statistical significance using SAS version 9.3 (SAS Institute, Cary, NC) or R software (
RESULTS
We included 322,938 patient admissions. Of this sample, 54,645 (16.9%) had spent at least 1 night in the ICU. Overall, 76,758 patients (23.8%) had diabetes, representing 26.3% of ICU patients, 23.2% of non‐ICU patients, 28.2% of medical patients, and 16.8% of surgical patients (see Table 1 for demographic characteristics).
Mortality Trends Within Strata
Among ICU patients, the overall mortality rate was 9.9%: 10.5% of patients with diabetes and 9.8% of patients without diabetes. Among non‐ICU patients, the overall mortality rate was 0.8%: 0.9% of patients with diabetes and 0.7% of patients without diabetes.
Among medical patients, the overall mortality rate was 2.9%: 3.1% of patients with diabetes and 2.8% of patients without diabetes. Among surgical patients, the overall mortality rate was 1.4%: 1.8% of patients with diabetes and 1.4% of patients without diabetes. Figure 1 shows quarterly in‐hospital mortality for patients with and without diabetes from 2000 to 2010 stratified by ICU status and by service assignment.

Table 2 describes the difference‐in‐differences regression analyses, stratified by ICU status and service assignment. Among ICU patients (Table 2, model 1), each successive year was associated with a 2.6% relative reduction in the adjusted odds of mortality (odds ratio [OR]: 0.974, 95% confidence interval [CI]: 0.963‐0.985) for patients without diabetes compared to a 7.8% relative reduction for those with diabetes (OR: 0.923, 95% CI: 0.906‐0.940). In other words, patients with diabetes compared to patients without diabetes had a significantly greater decline in odds of adjusted mortality of 5.3% per year (OR: 0.947, 95% CI: 0.927‐0.967). As a result, the adjusted odds of mortality among patients with versus without diabetes decreased from 1.352 in 2000 to 0.772 in 2010.
| Independent Variables | ICU Patients, N=54,646, OR (95% CI) | Non‐ICU Patients, N=268,293, OR (95% CI) | Medical Patients, N=196,325, OR (95% CI) | Surgical Patients, N=126,614, OR (95% CI) |
|---|---|---|---|---|
| Model 1 | Model 2 | Model 3 | Model 4 | |
| ||||
| Year | 0.974 (0.963‐0.985) | 0.925 (0.909‐0.940) | 0.943 (0.933‐0.954) | 0.995 (0.977‐1.103) |
| Diabetes | 1.352 (1.562‐1.171) | 0.958 (0.783‐1.173) | 1.186 (1.037‐1.356) | 1.213 (0.942‐1.563) |
| Diabetes*year | 0.947 (0.927‐0.967) | 0.977 (0.946‐1.008) | 0.961 (0.942‐0.980) | 0.955 (0.918‐0.994) |
| C statistic | 0.812 | 0.907 | 0.880 | 0.919 |
Among non‐ICU patients (Table 2, model 2), each successive year was associated with a 7.5% relative reduction in the adjusted odds of mortality (OR: 0.925, 95% CI: 0.909‐0.940) for patients without diabetes compared to a 9.6% relative reduction for those with diabetes (OR: 0.904, 95% CI: 0.879‐0.929); this greater decline in odds of adjusted mortality of 2.3% per year (OR: 0.977, 95% CI: 0.946‐1.008; P=0.148) was not statistically significant.
We found greater decline in odds of mortality among patients with diabetes than among patients without diabetes over time in both medical patients (3.9% greater decline per year; OR: 0.961, 95% CI: 0.942‐0.980) and surgical patients (4.5% greater decline per year; OR: 0.955, 95% CI: 0.918‐0.994), without a difference between the 2. Detailed results are shown in Table 2, models 3 and 4.
Glycemic Control
Among ICU patients with diabetes (N=14,364), at least 2 inpatient point‐of‐care glucose readings were available for 13,136 (91.5%), with a mean of 4.67 readings per day, whereas hemoglobin A1c data were available for only 5321 patients (37.0%). Both inpatient glucose data and hemoglobin A1c were available for 4989 patients (34.7%). Figure 2 shows trends in inpatient and outpatient glycemic control measures among ICU patients with diabetes over the study period. Mean hemoglobin A1c decreased from 7.7 in 2000 to 7.3 in 2010. Mean hospitalization glucose began at 187.2, reached a nadir of 162.4 in the third quarter (Q3) of 2007, and rose subsequently to 174.4 with loosened glucose control targets. Standard deviation of mean glucose and percentage of patient‐days with a severe hyperglycemic episode followed a similar pattern, though with nadirs in Q4 2007 and Q2 2008, respectively, whereas percentage of patient‐days with a hypoglycemic episode rose from 1.46% in 2000, peaked at 3.00% in Q3 2005, and returned to 2.15% in 2010. All changes in glucose control are significant with P<0.001.

Mortality Trends and Glycemic Control
To determine whether glucose control explained the excess decline in odds of mortality among patients with diabetes in the ICU, we restricted our sample to ICU patients with diabetes and examined the association of diabetes with mortality after including measures of glucose control.
We first verified that the overall adjusted mortality trend among ICU patients with diabetes for whom we had measures of inpatient glucose control was similar to that of the full sample of ICU patients with diabetes. Similar to the full sample, we found that the adjusted excess odds of death significantly declined by a relative 7.3% each successive year (OR: 0.927, 95% CI: 0.907‐0.947; Table 3, model 1). We then included measures of inpatient glucose control in the model and found, as expected, that a higher percentage of days with severe hyperglycemia and with hypoglycemia was associated with an increased odds of death (P<0.001 for both; Table 3, model 2). Nonetheless, after including measures of inpatient glucose control, we found that the rate of change of excess odds of death for patients with diabetes was unchanged (OR: 0.926, 95% CI: 0.905‐0.947).
| Patients With Inpatient Glucose Control Measures, n=13,136 | Patients With Inpatient and Outpatient Glucose Control Measures, n=4,989 | ||||
|---|---|---|---|---|---|
| Independent Variables | Model 1, OR (95% CI) | Model 2, OR (95% CI) | Model 3, OR (95% CI) | Model 4, OR (95% CI) | Model 5, OR (95% CI) |
| |||||
| Year | 0.927 (0.907‐0.947) | 0.926 (0.905‐0.947) | 0.958 (0.919‐0.998) | 0.956 (0.916‐0.997) | 0.953 (0.914‐0.994) |
| % Severe hyperglycemic days | 1.016 (1.010‐1.021) | 1.009 (0.998‐1.020) | 1.010 (0.999‐1.021) | ||
| % Hypoglycemic days | 1.047 (1.040‐1.055) | 1.051 (1.037‐1.065) | 1.049 (1.036‐1.063) | ||
| % Normoglycemic days | 0.997 (0.994‐1.000) | 0.994 (0.989‐0.999) | 0.993 (0.988‐0.998) | ||
| SD of mean glucose | 0.996 (0.992‐1.000) | 0.993 (0.986‐1.000) | 0.994 (0.987‐1.002) | ||
| Mean HbA1c | 0.892 (0.828‐0.961) | ||||
| C statistic | 0.806 | 0.825 | 0.825 | 0.838 | 0.841 |
We then restricted our sample to patients with diabetes with both inpatient and outpatient glycemic control data and found that, in this subpopulation, the adjusted excess odds of death among patients with diabetes relative to those without significantly declined by a relative 4.2% each progressive year (OR: 0.958, 95% CI: 0.918‐0.998; Table 3, model 3). Including measures of inpatient glucose control in the model did not significantly change the rate of change of excess odds of death (OR: 0.956, 95% CI: 0.916‐0.997; Table 3, model 4), nor did including both measures of inpatient and outpatient glycemic control (OR: 0.953, 95% CI: 0.914‐0.994; Table 3, model 5).
DISCUSSION
We conducted a difference‐in‐difference analysis of in‐hospital mortality rates among adult patients with diabetes compared to patients without diabetes over 10 years, stratifying by ICU status and service assignment. For patients with any ICU stay, we found that the reduction in odds of mortality for patients with diabetes has been 3 times larger than the reduction in odds of mortality for patients without diabetes. For those without an ICU stay, we found no significant difference between patients with and without diabetes in the rate at which in‐hospital mortality declined. We did not find stratification by assignment to a medical or surgical service to be an effect modifier. Finally, despite the fact that our institution achieved better aggregate inpatient glucose control, less severe hyperglycemia, and better long‐term glucose control over the course of the decade, we did not find that either inpatient or outpatient glucose control explained the trend in mortality for patients with diabetes in the ICU. Our study is unique in its inclusion of all hospitalized patients and its ability to simultaneously assess whether both inpatient and outpatient glucose control are explanatory factors in the observed mortality trends.
The fact that improved inpatient glucose control did not explain the trend in mortality for patients with diabetes in the ICU is consistent with the majority of the literature on intensive inpatient glucose control. In randomized trials, intensive glucose control appears to be of greater benefit for patients without diabetes than for patients with diabetes.[31] In fact, in 1 study, patients with diabetes were the only group that did not benefit from intensive glucose control.[32] In our study, it is possible that the rise in hypoglycemia nullified some of the benefits of glucose control. Nationally, hospital admissions for hypoglycemia among Medicare beneficiaries now outnumber admissions for hyperglycemia.[27]
We also do not find that the decline in hemoglobin A1c attenuated the reduction in mortality in the minority of patients for whom these data were available. This is concordant with evidence from 3 randomized clinical trials that have failed to establish a clear beneficial effect of intensive outpatient glucose control on primary cardiovascular endpoints among older, high‐risk patients with type 2 diabetes using glucose‐lowering agents.[21, 22, 23] It is notable, however, that the population for whom we had available hemoglobin A1c results was not representative of the overall population of ICU patients with diabetes. Consequently, there may be an association of outpatient glucose control with inpatient mortality in the overall population of ICU patients with diabetes that we were not able to detect.
The decline in mortality among ICU patients with diabetes in our study may stem from factors other than glycemic control. It is possible that patients were diagnosed earlier in their course of disease in later years of the study period, making the population of patients with diabetes younger or healthier. Of note, however, our risk adjustment models were very robust, with C statistics from 0.82 to 0.92, suggesting that we were able to account for much of the mortality risk attributable to patient clinical and demographic factors. More intensive glucose management may have nonglycemic benefits, such as closer patient observation, which may themselves affect mortality. Alternatively, improved cardiovascular management for patients with diabetes may have decreased the incidence of cardiovascular events. During the study period, evidence from large clinical trials demonstrated the importance of tight blood pressure and lipid management in improving outcomes for patients with diabetes,[33, 34, 35, 36] guidelines for lipid management for patients with diabetes changed,[37] and fewer patients developed cardiovascular complications.[38] Finally, it is possible that our findings can be explained by an improvement in treatment of complications for which patients with diabetes previously have had disproportionately worse outcomes, such as percutaneous coronary intervention.[39]
Our findings may have important implications for both clinicians and policymakers. Changes in inpatient glucose management have required substantial additional resources on the part of hospitals. Our evidence regarding the questionable impact of inpatient glucose control on in‐hospital mortality trends for patients with diabetes is disappointing and highlights the need for multifaceted evaluation of the impact of such quality initiatives. There may, for instance, be benefits from tighter blood glucose control in the hospital beyond mortality, such as reduced infections, costs, or length of stay. On the outpatient side, our more limited data are consistent with recent studies that have not been able to show a mortality benefit in older diabetic patients from more stringent glycemic control. A reassessment of prevailing diabetes‐related quality measures, as recently called for by some,[40, 41] seems reasonable.
Our study must be interpreted in light of its limitations. It is possible that the improvements in glucose management were too small to result in a mortality benefit. The overall reduction of 25 mg dL achieved at our institution is less than the 33 to 50 mg/dL difference between intensive and conventional groups in those randomized clinical trials that have found reductions in mortality.[11, 42] In addition, an increase in mean glucose during the last 1 to 2 years of the observation period (in response to prevailing guidelines) could potentially have attenuated any benefit on mortality. The study does not include other important clinical endpoints, such as infections, complications, length of stay, and hospital costs. Additionally, we did not examine postdischarge mortality, which might have shown a different pattern. The small proportion of patients with hemoglobin A1c results may have hampered our ability to detect an effect of outpatient glucose control. Consequently, our findings regarding outpatient glucose control are only suggestive. Finally, our findings represent the experience of a single, large academic medical center and may not be generalizable to all settings.
Overall, we found that patients with diabetes in the ICU have experienced a disproportionate reduction in in‐hospital mortality over time that does not appear to be explained by improvements in either inpatient or outpatient glucose control. Although improved glycemic control may have other benefits, it does not appear to impact in‐hospital mortality. Our real‐world empirical results contribute to the discourse among clinicians and policymakers with regards to refocusing the approach to managing glucose in‐hospital and readjudication of diabetes‐related quality measures.
Acknowledgments
The authors would like to acknowledge the YaleNew Haven Hospital diabetes management team: Gael Ulisse, APRN, Helen Psarakis, APRN, Anne Kaisen, APRN, and the Yale Endocrine Fellows.
Disclosures: Design and conduct of the study: N. B., J. D., S. I., T. B., L. H. Collection, management, analysis, and interpretation of the data: N. B., B. J., J. D., J. R., J. B., S. I., L. H. Preparation, review, or approval of the manuscript: N. B., B. J., J. D., J. R., S. I., T. B., L. H. Leora Horwitz, MD, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. Dr. Horwitz is supported by the National Institute on Aging (K08 AG038336) and by the American Federation for Aging Research through the Paul B. Beeson Career Development Award Program. This publication was also made possible by CTSA grant number UL1 RR024139 from the National Center for Research Resources and the National Center for Advancing Translational Science, components of the National Institutes of Health (NIH), and NIH roadmap for Medical Research. Its contents are solely the responsibility of the authors and do not necessarily represent the official view of the NIH. No funding source had any role in design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; or decision to submit the manuscript for publication. Silvio E. Inzucchi, MD, serves on a Data Safety Monitoring Board for Novo Nordisk, a manufacturer of insulin products used in the hospital setting. The remaining authors declare no conflicts of interest.
- National Diabetes Information Clearinghouse. National Diabetes Statistics; 2011. Available at: http://diabetes.niddk.nih.gov/dm/pubs/america/index.aspx. Accessed November 12, 2013.
- Healthcare Cost and Utilization Project. Statistical brief #93; 2010. Available at: http://www.hcup‐us.ahrq.gov/reports/statbriefs/sb93.pdf. Accessed November 12, 2013.
- , , , et al. Association between diabetes mellitus and post‐discharge outcomes in patients hospitalized with heart failure: findings from the EVEREST trial. Eur J Heart Fail. 2013;15(2):194–202.
- , , , et al. Influence of diabetes mellitus on clinical outcome in the thrombolytic era of acute myocardial infarction. GUSTO‐I Investigators. Global Utilization of Streptokinase and Tissue Plasminogen Activator for Occluded Coronary Arteries. J Am Coll Cardiol. 1997;30(1):171–179.
- , , , , , . Type 2 diabetes and pneumonia outcomes: a population‐based cohort study. Diabetes Care. 2007;30(9):2251–2257.
- , , , . Prevalence and outcomes of diabetes, hypertension and cardiovascular disease in COPD. Eur Respir J. 2008;32(4):962–969.
- , , , , . The role of body mass index and diabetes in the development of acute organ failure and subsequent mortality in an observational cohort. Crit Care. 2006;10(5):R137.
- , , , , , . Type 2 diabetes and 1‐year mortality in intensive care unit patients. Eur J Clin Invest. 2013;43(3):238–247.
- , , . Excess mortality during hospital stays among patients with recorded diabetes compared with those without diabetes. Diabet Med. 2013;30(12):1393–1402.
- , , , et al. Decade‐long trends in mortality among patients with and without diabetes mellitus at a major academic medical center. JAMA Intern Med. 2014;174(7):1187–1188.
- , , , et al. Intensive insulin therapy in critically ill patients. N Engl J Med. 2001;345(19):1359–1367.
- , , , et al. Intensive versus conventional glucose control in critically ill patients. N Engl J Med. 2009;360(13):1283–1297.
- , , , et al. A prospective randomised multi‐centre controlled trial on tight glucose control by intensive insulin therapy in adult intensive care units: the Glucontrol study. Intensive Care Med. 2009;35(10):1738–1748.
- , , , et al. Intensive versus conventional insulin therapy: a randomized controlled trial in medical and surgical critically ill patients. Crit Care Med. 2008;36(12):3190–3197.
- , , , et al. Intensive insulin therapy in the medical ICU. N Engl J Med. 2006;354(5):449–461.
- , , , et al. Glycemic control in non‐critically ill hospitalized patients: a systematic review and meta‐analysis. J Clin Endocrinol Metab. 2012;97(1):49–58.
- , , , et al. American Association of Clinical Endocrinologists and American Diabetes Association consensus statement on inpatient glycemic control. Diabetes Care. 2009;32(6):1119–1131.
- Agency for Healthcare Research and Quality National Quality Measures Clearinghouse. Percent of cardiac surgery patients with controlled 6 A.M. postoperative blood glucose; 2012. Available at: http://www.qualitymeasures.ahrq.gov/content.aspx?id=35532. Accessed November 12, 2013.
- The effect of intensive treatment of diabetes on the development and progression of long‐term complications in insulin‐dependent diabetes mellitus. The Diabetes Control and Complications Trial Research Group. N Engl J Med. 1993;329(14):977–986.
- , , , et al. Intensive blood‐glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). Lancet. 1998;352(9131):837–853.
- Effects of intensive glucose lowering in type 2 diabetes. N Engl J Med. 2008;358(24):2545–2559.
- , , , et al. Glucose control and vascular complications in veterans with type 2 diabetes. N Engl J Med. 2009;360(2):129–139.
- , , , et al. Intensive blood glucose control and vascular outcomes in patients with type 2 diabetes. N Engl J Med. 2008;358(24):2560–2572.
- . Standards of medical care in diabetes—2014. Diabetes Care. 2014;37(suppl 1):S14–S80.
- National Committee for Quality Assurance. HEDIS 2013. Available at: http://www.ncqa.org/HEDISQualityMeasurement.aspx. Accessed November 12, 2013.
- , , , . Is glycemic control improving in US adults? Diabetes Care. 2008;31(1):81–86.
- , , , et al. National trends in US hospital admissions for hyperglycemia and hypoglycemia among medicare beneficiaries, 1999 to 2011. JAMA Intern Med. 2014;174(7):1116–1124.
- , , , et al. "Glucometrics"—assessing the quality of inpatient glucose management. Diabetes Technol Ther. 2006;8(5):560–569.
- , , , , . A modification of the Elixhauser comorbidity measures into a point system for hospital death using administrative data. Med Care. 2009;47(6):626–633.
- Healthcare Cost and Utilization Project. Clinical Classifications Software (CCS) for ICD‐9‐CM; 2013. Available at: http://www.hcup‐us.ahrq.gov/toolssoftware/ccs/ccs.jsp. Accessed November 12, 2013.
- , , , , . The impact of premorbid diabetic status on the relationship between the three domains of glycemic control and mortality in critically ill patients. Curr Opin Clin Nutr Metab Care. 2012;15(2):151–160.
- , , , et al. Intensive insulin therapy in mixed medical/surgical intensive care units: benefit versus harm. Diabetes. 2006;55(11):3151–3159.
- Tight blood pressure control and risk of macrovascular and microvascular complications in type 2 diabetes: UKPDS 38. UK Prospective Diabetes Study Group. BMJ. 1998;317(7160):703–713.
- , , , et al. Effects of a fixed combination of perindopril and indapamide on macrovascular and microvascular outcomes in patients with type 2 diabetes mellitus (the ADVANCE trial): a randomised controlled trial. Lancet. 2007;370(9590):829–840.
- , , , , . MRC/BHF heart protection study of cholesterol‐lowering with simvastatin in 5963 people with diabetes: a randomised placebo‐controlled trial. Lancet. 2003;361(9374):2005–2016.
- , , , et al. Primary prevention of cardiovascular disease with atorvastatin in type 2 diabetes in the Collaborative Atorvastatin Diabetes Study (CARDS): multicentre randomised placebo‐controlled trial. Lancet. 2004;364(9435):685–696.
- , , , . Expert panel on detection, evaluation and treatment of high blood cholesterol in adults. Executive summary of the third report of the national cholesterol education program (NCEP) adult treatment panel (atp III). JAMA. 2001;285(19):2486–2497.
- , , , et al. Changes in diabetes‐related complications in the United States, 1990–2010. N Engl J Med. 2014;370(16):1514–1523.
- , , . Coronary heart disease in patients with diabetes: part II: recent advances in coronary revascularization. J Am Coll Cardiol. 2007;49(6):643–656.
- , , , et al. Management of hyperglycemia in type 2 diabetes: a patient‐centered approach position statement of the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetes Care. 2012;35(6):1364–1379.
- , , , , . Assessing potential glycemic overtreatment in persons at hypoglycemic risk. JAMA Intern Med. 2013;174(2):259–268.
- , , , . Glycometabolic state at admission: important risk marker of mortality in conventionally treated patients with diabetes mellitus and acute myocardial infarction: long‐term results from the Diabetes and Insulin‐Glucose Infusion in Acute Myocardial Infarction (DIGAMI) study. Circulation. 1999;99(20):2626–2632.
- National Diabetes Information Clearinghouse. National Diabetes Statistics; 2011. Available at: http://diabetes.niddk.nih.gov/dm/pubs/america/index.aspx. Accessed November 12, 2013.
- Healthcare Cost and Utilization Project. Statistical brief #93; 2010. Available at: http://www.hcup‐us.ahrq.gov/reports/statbriefs/sb93.pdf. Accessed November 12, 2013.
- , , , et al. Association between diabetes mellitus and post‐discharge outcomes in patients hospitalized with heart failure: findings from the EVEREST trial. Eur J Heart Fail. 2013;15(2):194–202.
- , , , et al. Influence of diabetes mellitus on clinical outcome in the thrombolytic era of acute myocardial infarction. GUSTO‐I Investigators. Global Utilization of Streptokinase and Tissue Plasminogen Activator for Occluded Coronary Arteries. J Am Coll Cardiol. 1997;30(1):171–179.
- , , , , , . Type 2 diabetes and pneumonia outcomes: a population‐based cohort study. Diabetes Care. 2007;30(9):2251–2257.
- , , , . Prevalence and outcomes of diabetes, hypertension and cardiovascular disease in COPD. Eur Respir J. 2008;32(4):962–969.
- , , , , . The role of body mass index and diabetes in the development of acute organ failure and subsequent mortality in an observational cohort. Crit Care. 2006;10(5):R137.
- , , , , , . Type 2 diabetes and 1‐year mortality in intensive care unit patients. Eur J Clin Invest. 2013;43(3):238–247.
- , , . Excess mortality during hospital stays among patients with recorded diabetes compared with those without diabetes. Diabet Med. 2013;30(12):1393–1402.
- , , , et al. Decade‐long trends in mortality among patients with and without diabetes mellitus at a major academic medical center. JAMA Intern Med. 2014;174(7):1187–1188.
- , , , et al. Intensive insulin therapy in critically ill patients. N Engl J Med. 2001;345(19):1359–1367.
- , , , et al. Intensive versus conventional glucose control in critically ill patients. N Engl J Med. 2009;360(13):1283–1297.
- , , , et al. A prospective randomised multi‐centre controlled trial on tight glucose control by intensive insulin therapy in adult intensive care units: the Glucontrol study. Intensive Care Med. 2009;35(10):1738–1748.
- , , , et al. Intensive versus conventional insulin therapy: a randomized controlled trial in medical and surgical critically ill patients. Crit Care Med. 2008;36(12):3190–3197.
- , , , et al. Intensive insulin therapy in the medical ICU. N Engl J Med. 2006;354(5):449–461.
- , , , et al. Glycemic control in non‐critically ill hospitalized patients: a systematic review and meta‐analysis. J Clin Endocrinol Metab. 2012;97(1):49–58.
- , , , et al. American Association of Clinical Endocrinologists and American Diabetes Association consensus statement on inpatient glycemic control. Diabetes Care. 2009;32(6):1119–1131.
- Agency for Healthcare Research and Quality National Quality Measures Clearinghouse. Percent of cardiac surgery patients with controlled 6 A.M. postoperative blood glucose; 2012. Available at: http://www.qualitymeasures.ahrq.gov/content.aspx?id=35532. Accessed November 12, 2013.
- The effect of intensive treatment of diabetes on the development and progression of long‐term complications in insulin‐dependent diabetes mellitus. The Diabetes Control and Complications Trial Research Group. N Engl J Med. 1993;329(14):977–986.
- , , , et al. Intensive blood‐glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). Lancet. 1998;352(9131):837–853.
- Effects of intensive glucose lowering in type 2 diabetes. N Engl J Med. 2008;358(24):2545–2559.
- , , , et al. Glucose control and vascular complications in veterans with type 2 diabetes. N Engl J Med. 2009;360(2):129–139.
- , , , et al. Intensive blood glucose control and vascular outcomes in patients with type 2 diabetes. N Engl J Med. 2008;358(24):2560–2572.
- . Standards of medical care in diabetes—2014. Diabetes Care. 2014;37(suppl 1):S14–S80.
- National Committee for Quality Assurance. HEDIS 2013. Available at: http://www.ncqa.org/HEDISQualityMeasurement.aspx. Accessed November 12, 2013.
- , , , . Is glycemic control improving in US adults? Diabetes Care. 2008;31(1):81–86.
- , , , et al. National trends in US hospital admissions for hyperglycemia and hypoglycemia among medicare beneficiaries, 1999 to 2011. JAMA Intern Med. 2014;174(7):1116–1124.
- , , , et al. "Glucometrics"—assessing the quality of inpatient glucose management. Diabetes Technol Ther. 2006;8(5):560–569.
- , , , , . A modification of the Elixhauser comorbidity measures into a point system for hospital death using administrative data. Med Care. 2009;47(6):626–633.
- Healthcare Cost and Utilization Project. Clinical Classifications Software (CCS) for ICD‐9‐CM; 2013. Available at: http://www.hcup‐us.ahrq.gov/toolssoftware/ccs/ccs.jsp. Accessed November 12, 2013.
- , , , , . The impact of premorbid diabetic status on the relationship between the three domains of glycemic control and mortality in critically ill patients. Curr Opin Clin Nutr Metab Care. 2012;15(2):151–160.
- , , , et al. Intensive insulin therapy in mixed medical/surgical intensive care units: benefit versus harm. Diabetes. 2006;55(11):3151–3159.
- Tight blood pressure control and risk of macrovascular and microvascular complications in type 2 diabetes: UKPDS 38. UK Prospective Diabetes Study Group. BMJ. 1998;317(7160):703–713.
- , , , et al. Effects of a fixed combination of perindopril and indapamide on macrovascular and microvascular outcomes in patients with type 2 diabetes mellitus (the ADVANCE trial): a randomised controlled trial. Lancet. 2007;370(9590):829–840.
- , , , , . MRC/BHF heart protection study of cholesterol‐lowering with simvastatin in 5963 people with diabetes: a randomised placebo‐controlled trial. Lancet. 2003;361(9374):2005–2016.
- , , , et al. Primary prevention of cardiovascular disease with atorvastatin in type 2 diabetes in the Collaborative Atorvastatin Diabetes Study (CARDS): multicentre randomised placebo‐controlled trial. Lancet. 2004;364(9435):685–696.
- , , , . Expert panel on detection, evaluation and treatment of high blood cholesterol in adults. Executive summary of the third report of the national cholesterol education program (NCEP) adult treatment panel (atp III). JAMA. 2001;285(19):2486–2497.
- , , , et al. Changes in diabetes‐related complications in the United States, 1990–2010. N Engl J Med. 2014;370(16):1514–1523.
- , , . Coronary heart disease in patients with diabetes: part II: recent advances in coronary revascularization. J Am Coll Cardiol. 2007;49(6):643–656.
- , , , et al. Management of hyperglycemia in type 2 diabetes: a patient‐centered approach position statement of the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetes Care. 2012;35(6):1364–1379.
- , , , , . Assessing potential glycemic overtreatment in persons at hypoglycemic risk. JAMA Intern Med. 2013;174(2):259–268.
- , , , . Glycometabolic state at admission: important risk marker of mortality in conventionally treated patients with diabetes mellitus and acute myocardial infarction: long‐term results from the Diabetes and Insulin‐Glucose Infusion in Acute Myocardial Infarction (DIGAMI) study. Circulation. 1999;99(20):2626–2632.
© 2015 Society of Hospital Medicine
Inpatient Mammography
Testing for breast cancer is traditionally offered in outpatient settings, and screening mammography rates have plateaued since 2000.[1] Current data suggest that the mammography utilization gap by race has narrowed; however, disparity remains among low‐income, uninsured, and underinsured populations.[2, 3] The lowest compliance with screening mammography recommendations have been reported among women with low income (63.2%), uninsured (50.4%), and those without a usual source of healthcare (43.6%).[4] Although socioeconomic status, access to the healthcare system, and awareness about screening benefits can all influence women's willingness to have screening, the most common reason that women report for not having mammograms were that no one recommended the test.[5, 6] These findings support previous reports that physicians' recommendations about the need for screening mammography is an influential factor in determining women's decisions related to compliance.[7] Hence, the role of healthcare providers in all clinical care settings is pivotal in reducing mammography utilization disparities.
A recent study evaluating the breast cancer screening adherence among the hospitalized women aged 50 to 75 years noted that many (60%) were low income (annual household income <$20,000), 39% were nonadherent, and 35% were at high risk of developing breast cancer.[8] Further, a majority of these hospitalized women were amenable to inpatient screening mammography if due and offered during the hospital stay.[8] As a follow‐up, the purpose of the current study was to explore how hospitalists feel about getting involved in breast cancer screening and ordering screening mammograms for hospitalized women. We hypothesized that a greater proportion of hospitalists would order mammography for hospitalized women who were both overdue for screening and at high risk for developing breast cancer if they fundamentally believe that they have a role in breast cancer screening. This study also explored anticipated barriers that may be of concern to hospitalists when ordering inpatient screening mammography.
METHODS
Study Design and Sample
All hospitalist providers within 4 groups affiliated with Johns Hopkins Medical Institution (Johns Hopkins Hospital, Johns Hopkins Bayview Medical Center, Howard County General Hospital, and Suburban Hospital) were approached for participation in this‐cross sectional study. The hospitalists included physicians, nurse practitioners, and physician assistants. All hospitalists were eligible to participate in the study, and there was no monetary incentive attached to the study participation. A total of 110 hospitalists were approached for study participation. Of these, 4 hospitalists (3.5%) declined to participate, leaving a study population of 106 hospitalists.
Data Collection and Measures
Participants were sent the survey via email using SurveyMonkey. The survey included questions regarding demographic information such as age, gender, race, and clinical experience in hospital medicine. To evaluate for potential personal sources of bias related to mammography, study participants were asked if they have had a family member diagnosed with breast cancer.
A central question asked whether respondents agreed with the following: I believe that hospitalists should be involved in breast cancer screening. The questionnaire also evaluated hospitalists' practical approaches to 2 clinical scenarios by soliciting decision about whether they would order an inpatient screening mammogram. These clinical scenarios were designed using the Gail risk prediction score for probability of developing breast cancer within the next 5 years according to the National Cancer Institute Breast Cancer Risk Tool.[9] Study participants were not provided with the Gail scores and had to infer the risk from the clinical information provided in scenarios. One case described a woman at high risk, and the other with a lower‐risk profile. The first question was: Would you order screening mammography for a 65‐year‐old African American female with obesity and family history for breast cancer admitted to the hospital for cellulitis? She has never had a mammogram and is willing to have it while in hospital. Based on the information provided in the scenario, the 5‐year risk prediction for developing breast cancer using the Gail risk model was high (2.1%). The second scenario asked: Would you order a screening mammography for a 62‐year‐old healthy Hispanic female admitted for presyncope? Patient is uninsured and requests a screening mammogram while in hospital [assume that personal and family histories for breast cancer are negative]. Based on the information provided in the scenario, the 5‐year risk prediction for developing breast cancer using the Gail risk model was low (0.6%).
Several questions regarding potential barriers to inpatient screening mammography were also asked. Some of these questions were based on barriers mentioned in our earlier study of patients,[8] whereas others emerged from a review of the literature and during focus group discussions with hospitalist providers. Pilot testing of the survey was conducted on hospitalists outside the study sample to enhance question clarity. This study was approved by our institutional review board.
Statistical Methods
Respondent characteristics are presented as proportions and means. Unpaired t tests and [2] tests were used to look for associations between demographic characteristics and responses to the question about whether they believe that they should be involved in breast cancer screening. The survey data were analyzed using the Stata statistical software package version 12.1 (StataCorp, College Station, TX).
RESULTS
Out of 106 study subjects willing to participate, 8 did not respond, yielding a response rate of 92%. The mean age of the study participants was 37.6 years, and 55% were female. Almost two‐thirds of study participants (59%) were faculty physicians at an academic hospital, and the average clinical experience as a hospitalist was 4.6 years. Study participants were diverse with respect to ethnicity, and only 30% reported having a family member with breast cancer (Table 1). Because breast cancer is a disease that affects primarily women, stratified analysis by gender showed that most of these characteristic were similar across genders, except fewer women were full time (76% vs 93%, P=0.04) and on the faculty (44% vs 77%, P=0.003).
| Characteristics* | All Participants (n=98) |
|---|---|
| |
| Age, y, mean (SD) | 37.6 (5.5) |
| Female, n (%) | 54 (55) |
| Race, n (%) | |
| Caucasian | 35 (36) |
| African American | 12 (12) |
| Asian | 32 (33) |
| Other | 13 (13) |
| Hospitalist experience, y, mean (SD) | 4.6 (3.5) |
| Full time, n (%) | 82 (84) |
| Family history of breast cancer, n (%) | 30 (30) |
| Faculty physician, n (%) | 58 (59) |
| Believe that hospitalists should be involved in breast cancer screening, n (%) | 35 (38) |
Only 38% believed that hospitalists should be involved with breast cancer screening. The most commonly cited concern related to ordering an inpatient screening mammography was follow‐up of the results of the mammography, followed by the test may not be covered by patient's insurance. As shown in Table 2, these concerns were not perceived differently among providers who believed that hospitalists should be involved in breast cancer screening as compared to those who do not. Demographic variables from Table 1 failed to discern any significant associations related to believing that hospitalists should be involved with breast cancer screening or with concerns about the barriers to screening presented in Table 2 (data not shown). As shown in Table 2, overall, 32% hospitalists were willing to order a screening mammography during a hospital stay for the scenario of the woman at high risk for developing breast cancer (5‐year risk prediction using Gail model 2.1%) and 33% for the low‐risk scenario (5‐year risk prediction using Gail model 0.6%).
| Concern About Screening* | Believe That Hospitalists Should Be Involved in Breast Cancer Screening (n=35) | Do Not Believe That Hospitalists Should Be Involved in Breast Cancer Screening (n=58) | P Value |
|---|---|---|---|
| |||
| Result follow‐up, agree/strongly agree, n (%) | 34 (97) | 51 (88) | 0.25 |
| Interference with patient care, agree/strongly agree, n (%) | 23 (67) | 27 (47) | 0.07 |
| Cost, agree/strongly agree, n (%) | 23 (66) | 28 (48) | 0.10 |
| Concern that the test will not be covered by patient's insurance, agree/strongly agree, n (%) | 23 (66) | 34 (59) | 0.50 |
| Not my responsibility to do cancer prevention, agree/strongly agree, n (%) | 7 (20) | 16 (28) | 0.57 |
| Response to clinical scenarios | |||
| Would order a screening mammogram in the hospital for a high‐risk woman [scenario 1: Gail risk model: 2.1%], n (%) | 23 (66) | 6 (10) | 0.0001 |
| Would order a screening mammography in the hospital for a low‐risk woman [scenario 2: Gail risk model: 0.6%], n (%) | 18 (51) | 13 (22) | 0.004 |
DISCUSSION
Our study suggests that most hospitalists do not believe that they should be involved in breast cancer screening for their hospitalized patients. This perspective was not influenced by either the physician gender, family history for breast cancer, or by the patient's level of risk for developing breast cancer. When patients are in the hospital, both the setting and the acute illness are known to promote reflection and consideration of self‐care.[10] With major healthcare system changes on the horizon and the passing of the Affordable Care Act, we are becoming teams of providers who are collectively responsible for optimal care delivery. It may be possible to increase breast cancer screening rates by educating our patients and offering inpatient screening mammography while they are in the hospital, particularly to those who are at high risk of developing breast cancer.
Physician recommendations for preventive health and screening have consistently been found to be among the strongest predictors of screening utilization.[11] This is the first study to our knowledge that has attempted to understand hospitalists' views and concerns about ordering screening tests to detect occult malignancy. Although addressing preventive care during a hospitalization may seem complex and difficult, helping these women understand their personal risk profile (eg, family history of breast cancer, use of estrogen, race, age, and genetic risk factors) may be what is needed for beginning to influence perspective that might ultimately translate into a willingness to undergo screening.[12, 13, 14] Such delivery of patient‐centered care is built on a foundation of shared decision‐making, which takes into account the patient's preferences, values, and wishes.[15]
Ordering screening mammography for hospitalized patients will require a deeper understanding of hospitalists' attitudes, because the way that these physicians feel about the tests utility will dramatically influence the way that this opportunity is presented to patients, and ultimately the patients' preference to have or forego testing. Our study results are consistent with another publication that highlighted incongruence between physicians' views and patients' preferences for screening practices.[8, 11] Concerns cited, such as interference with patient's acute care, deserve attention, because it may be possible to carry out the screening in ways and at times that do not interfere with treatment or prolong length of stay. Exploring this with a feasibility study will be necessary. Such an approach has been advocated by Trimble et al. for inpatient cervical cancer screening as an efficient strategy to target high‐risk, nonadherent women.[16]
The inpatient setting allows for the elimination of major barriers to screening (like transportation and remembering to get to screening appointments),[8] thereby actively facilitating this needed service. Costs associated with inpatient screening mammography may deter both hospitalists and patients from screening; however, some insurers and Medicare pay for the full cost of screening tests, irrespective of the clinical setting.[17] Further, as hospitals or accountable care organizations become responsible for total cost per beneficiary, screening costs will be preferable when compared with the expenses associated with later detection of pathology and caring for advanced disease states.
One might question whether the mortality benefit of screening mammography is comparable among hospitalized women (who are theoretically sicker and with shorter life expectancy) and those cared for in outpatient practices. Unfortunately, we do not yet know the answer to this question, because data for inpatient screening mammography are nonexistent, and currently this is not considered as a standard of care. However, one can expect the benefits to be similar, if not greater, when performed in the outpatient setting, if preliminary efforts are directed at those who are both nonadherent and at high risk for breast cancer. According to 1 study, increasing mammography utilization by 5% in our country would prevent 560 deaths from breast cancer each year.[18]
Several limitations of this study should be considered. First, this cross‐sectional study was conducted at hospitals associated with a single institution and the results may not be generalizable. Second, although physicians' concerns were explored in this study, we did not solicit input about the potential impact of prevention and screening on the nursing staff. Third, there may be concerns about the hypothetical nature of anchoring and possible framing effects with the 2 clinical scenarios. Finally, it is possible that the hospitalists' response may have been subject to social desirability bias. That said, the response to the key question Do you think hospitalists should be involved in breast cancer screening? do not support a socially desirable bias.
Given the current policy emphasis on reducing disparities in cancer screening, it may be reasonable to expand the role of all healthcare providers and healthcare facilities in screening high‐risk populations. Screening tests that may seem difficult to coordinate in hospitals currently may become easier as our hospitals evolve to become more patient centered. Future studies are needed to evaluate the feasibility and potential barriers to inpatient screening mammography.
Disclosure
Disclosures: Dr. Wright is a Miller‐Coulson Family Scholar, and this support comes from Hopkins Center for Innovative Medicine. This work was made possible in part by the Maryland Cigarette Restitution Fund Research Grant at Johns Hopkins. The authors report no conflicts of interest.
- Centers for Disease Control and Prevention (CDC). Vital signs: breast cancer screening among women aged 50–74 years—United States, 2008. MMWR Morb Mortal Wkly Rep. 2010;59(26):813–816.
- American Cancer Society. Breast Cancer Facts 2013.
- , , , et al. Impact of socioeconomic status on cancer incidence and stage at diagnosis: selected findings from the surveillance, epidemiology, and end results: National Longitudinal Mortality Study. Cancer Causes Control. 2009;20:417–435.
- , , , ; Centers for Disease Control and Prevention. Breast cancer screening among adult women—behavioral risk factor surveillance system, United States, 2010. MMWR Morb Mortal Wkly Rep. 2012;61(suppl):46–50.
- , . Disparities in breast cancer. Curr Probl Cancer. 2007;31(3):134–156.
- , , . Factors associated with mammography utilization: a systematic quantitative review of the literature. J Womens Health (Larchmt). 2008;17:1477–1498.
- , , , et al. Processes of care in cervical and breast cancer screening and follow‐up: the importance of communication. Prev Med. 2004;39:81–90.
- , , , . Breast cancer screening preferences among hospitalized women. J Womens Health (Larchmt). 2013;22(7):637–642.
- , , , et al. Projecting individualized probabilities of developing breast cancer for white females who are being examined annually. J Natl Cancer Inst. 1989;8:1879–1886.
- , , , . Expanding the roles of hospitalist physicians to include public health. J Hosp Med. 2007;2:93–101.
- , , , et al. Colorectal cancer screening: conjoint analysis of consumer preferences and physicians' perceived consumer preferences in the US and Canada. Paper presented at: 27th Annual Meeting of the Society for Medical Decision Making; October 21–24, 2005; San Francisco, CA.
- , , . Family history of breast cancer: impact on the disease experience. Cancer Pract. 2000;8:135–142.
- , , , . Breast cancer knowledge and attitudes toward mammography as predictors of breast cancer preventive behavior in Kazakh, Korean, and Russian women in Kazakhstan. Int J Public Health. 2008;53:123–130.
- , , , , . The relation between projected breast cancer risk, perceived cancer risk, and mammography use. Results from the National Health Interview Survey. J Gen Intern Med. 2006;21:158–164.
- , . Patient‐centered communication in cancer care: promoting healing and reducing suffering. NIH publication no. 07‐6225. Bethesda, MD: National Cancer Institute, 2007.
- , , , , , . Effectiveness of screening for cervical cancer in an inpatient hospital setting. Obstet Gynecol. 2004;103(2):310–316.
- Centers for Medicare 38:600–609.
Testing for breast cancer is traditionally offered in outpatient settings, and screening mammography rates have plateaued since 2000.[1] Current data suggest that the mammography utilization gap by race has narrowed; however, disparity remains among low‐income, uninsured, and underinsured populations.[2, 3] The lowest compliance with screening mammography recommendations have been reported among women with low income (63.2%), uninsured (50.4%), and those without a usual source of healthcare (43.6%).[4] Although socioeconomic status, access to the healthcare system, and awareness about screening benefits can all influence women's willingness to have screening, the most common reason that women report for not having mammograms were that no one recommended the test.[5, 6] These findings support previous reports that physicians' recommendations about the need for screening mammography is an influential factor in determining women's decisions related to compliance.[7] Hence, the role of healthcare providers in all clinical care settings is pivotal in reducing mammography utilization disparities.
A recent study evaluating the breast cancer screening adherence among the hospitalized women aged 50 to 75 years noted that many (60%) were low income (annual household income <$20,000), 39% were nonadherent, and 35% were at high risk of developing breast cancer.[8] Further, a majority of these hospitalized women were amenable to inpatient screening mammography if due and offered during the hospital stay.[8] As a follow‐up, the purpose of the current study was to explore how hospitalists feel about getting involved in breast cancer screening and ordering screening mammograms for hospitalized women. We hypothesized that a greater proportion of hospitalists would order mammography for hospitalized women who were both overdue for screening and at high risk for developing breast cancer if they fundamentally believe that they have a role in breast cancer screening. This study also explored anticipated barriers that may be of concern to hospitalists when ordering inpatient screening mammography.
METHODS
Study Design and Sample
All hospitalist providers within 4 groups affiliated with Johns Hopkins Medical Institution (Johns Hopkins Hospital, Johns Hopkins Bayview Medical Center, Howard County General Hospital, and Suburban Hospital) were approached for participation in this‐cross sectional study. The hospitalists included physicians, nurse practitioners, and physician assistants. All hospitalists were eligible to participate in the study, and there was no monetary incentive attached to the study participation. A total of 110 hospitalists were approached for study participation. Of these, 4 hospitalists (3.5%) declined to participate, leaving a study population of 106 hospitalists.
Data Collection and Measures
Participants were sent the survey via email using SurveyMonkey. The survey included questions regarding demographic information such as age, gender, race, and clinical experience in hospital medicine. To evaluate for potential personal sources of bias related to mammography, study participants were asked if they have had a family member diagnosed with breast cancer.
A central question asked whether respondents agreed with the following: I believe that hospitalists should be involved in breast cancer screening. The questionnaire also evaluated hospitalists' practical approaches to 2 clinical scenarios by soliciting decision about whether they would order an inpatient screening mammogram. These clinical scenarios were designed using the Gail risk prediction score for probability of developing breast cancer within the next 5 years according to the National Cancer Institute Breast Cancer Risk Tool.[9] Study participants were not provided with the Gail scores and had to infer the risk from the clinical information provided in scenarios. One case described a woman at high risk, and the other with a lower‐risk profile. The first question was: Would you order screening mammography for a 65‐year‐old African American female with obesity and family history for breast cancer admitted to the hospital for cellulitis? She has never had a mammogram and is willing to have it while in hospital. Based on the information provided in the scenario, the 5‐year risk prediction for developing breast cancer using the Gail risk model was high (2.1%). The second scenario asked: Would you order a screening mammography for a 62‐year‐old healthy Hispanic female admitted for presyncope? Patient is uninsured and requests a screening mammogram while in hospital [assume that personal and family histories for breast cancer are negative]. Based on the information provided in the scenario, the 5‐year risk prediction for developing breast cancer using the Gail risk model was low (0.6%).
Several questions regarding potential barriers to inpatient screening mammography were also asked. Some of these questions were based on barriers mentioned in our earlier study of patients,[8] whereas others emerged from a review of the literature and during focus group discussions with hospitalist providers. Pilot testing of the survey was conducted on hospitalists outside the study sample to enhance question clarity. This study was approved by our institutional review board.
Statistical Methods
Respondent characteristics are presented as proportions and means. Unpaired t tests and [2] tests were used to look for associations between demographic characteristics and responses to the question about whether they believe that they should be involved in breast cancer screening. The survey data were analyzed using the Stata statistical software package version 12.1 (StataCorp, College Station, TX).
RESULTS
Out of 106 study subjects willing to participate, 8 did not respond, yielding a response rate of 92%. The mean age of the study participants was 37.6 years, and 55% were female. Almost two‐thirds of study participants (59%) were faculty physicians at an academic hospital, and the average clinical experience as a hospitalist was 4.6 years. Study participants were diverse with respect to ethnicity, and only 30% reported having a family member with breast cancer (Table 1). Because breast cancer is a disease that affects primarily women, stratified analysis by gender showed that most of these characteristic were similar across genders, except fewer women were full time (76% vs 93%, P=0.04) and on the faculty (44% vs 77%, P=0.003).
| Characteristics* | All Participants (n=98) |
|---|---|
| |
| Age, y, mean (SD) | 37.6 (5.5) |
| Female, n (%) | 54 (55) |
| Race, n (%) | |
| Caucasian | 35 (36) |
| African American | 12 (12) |
| Asian | 32 (33) |
| Other | 13 (13) |
| Hospitalist experience, y, mean (SD) | 4.6 (3.5) |
| Full time, n (%) | 82 (84) |
| Family history of breast cancer, n (%) | 30 (30) |
| Faculty physician, n (%) | 58 (59) |
| Believe that hospitalists should be involved in breast cancer screening, n (%) | 35 (38) |
Only 38% believed that hospitalists should be involved with breast cancer screening. The most commonly cited concern related to ordering an inpatient screening mammography was follow‐up of the results of the mammography, followed by the test may not be covered by patient's insurance. As shown in Table 2, these concerns were not perceived differently among providers who believed that hospitalists should be involved in breast cancer screening as compared to those who do not. Demographic variables from Table 1 failed to discern any significant associations related to believing that hospitalists should be involved with breast cancer screening or with concerns about the barriers to screening presented in Table 2 (data not shown). As shown in Table 2, overall, 32% hospitalists were willing to order a screening mammography during a hospital stay for the scenario of the woman at high risk for developing breast cancer (5‐year risk prediction using Gail model 2.1%) and 33% for the low‐risk scenario (5‐year risk prediction using Gail model 0.6%).
| Concern About Screening* | Believe That Hospitalists Should Be Involved in Breast Cancer Screening (n=35) | Do Not Believe That Hospitalists Should Be Involved in Breast Cancer Screening (n=58) | P Value |
|---|---|---|---|
| |||
| Result follow‐up, agree/strongly agree, n (%) | 34 (97) | 51 (88) | 0.25 |
| Interference with patient care, agree/strongly agree, n (%) | 23 (67) | 27 (47) | 0.07 |
| Cost, agree/strongly agree, n (%) | 23 (66) | 28 (48) | 0.10 |
| Concern that the test will not be covered by patient's insurance, agree/strongly agree, n (%) | 23 (66) | 34 (59) | 0.50 |
| Not my responsibility to do cancer prevention, agree/strongly agree, n (%) | 7 (20) | 16 (28) | 0.57 |
| Response to clinical scenarios | |||
| Would order a screening mammogram in the hospital for a high‐risk woman [scenario 1: Gail risk model: 2.1%], n (%) | 23 (66) | 6 (10) | 0.0001 |
| Would order a screening mammography in the hospital for a low‐risk woman [scenario 2: Gail risk model: 0.6%], n (%) | 18 (51) | 13 (22) | 0.004 |
DISCUSSION
Our study suggests that most hospitalists do not believe that they should be involved in breast cancer screening for their hospitalized patients. This perspective was not influenced by either the physician gender, family history for breast cancer, or by the patient's level of risk for developing breast cancer. When patients are in the hospital, both the setting and the acute illness are known to promote reflection and consideration of self‐care.[10] With major healthcare system changes on the horizon and the passing of the Affordable Care Act, we are becoming teams of providers who are collectively responsible for optimal care delivery. It may be possible to increase breast cancer screening rates by educating our patients and offering inpatient screening mammography while they are in the hospital, particularly to those who are at high risk of developing breast cancer.
Physician recommendations for preventive health and screening have consistently been found to be among the strongest predictors of screening utilization.[11] This is the first study to our knowledge that has attempted to understand hospitalists' views and concerns about ordering screening tests to detect occult malignancy. Although addressing preventive care during a hospitalization may seem complex and difficult, helping these women understand their personal risk profile (eg, family history of breast cancer, use of estrogen, race, age, and genetic risk factors) may be what is needed for beginning to influence perspective that might ultimately translate into a willingness to undergo screening.[12, 13, 14] Such delivery of patient‐centered care is built on a foundation of shared decision‐making, which takes into account the patient's preferences, values, and wishes.[15]
Ordering screening mammography for hospitalized patients will require a deeper understanding of hospitalists' attitudes, because the way that these physicians feel about the tests utility will dramatically influence the way that this opportunity is presented to patients, and ultimately the patients' preference to have or forego testing. Our study results are consistent with another publication that highlighted incongruence between physicians' views and patients' preferences for screening practices.[8, 11] Concerns cited, such as interference with patient's acute care, deserve attention, because it may be possible to carry out the screening in ways and at times that do not interfere with treatment or prolong length of stay. Exploring this with a feasibility study will be necessary. Such an approach has been advocated by Trimble et al. for inpatient cervical cancer screening as an efficient strategy to target high‐risk, nonadherent women.[16]
The inpatient setting allows for the elimination of major barriers to screening (like transportation and remembering to get to screening appointments),[8] thereby actively facilitating this needed service. Costs associated with inpatient screening mammography may deter both hospitalists and patients from screening; however, some insurers and Medicare pay for the full cost of screening tests, irrespective of the clinical setting.[17] Further, as hospitals or accountable care organizations become responsible for total cost per beneficiary, screening costs will be preferable when compared with the expenses associated with later detection of pathology and caring for advanced disease states.
One might question whether the mortality benefit of screening mammography is comparable among hospitalized women (who are theoretically sicker and with shorter life expectancy) and those cared for in outpatient practices. Unfortunately, we do not yet know the answer to this question, because data for inpatient screening mammography are nonexistent, and currently this is not considered as a standard of care. However, one can expect the benefits to be similar, if not greater, when performed in the outpatient setting, if preliminary efforts are directed at those who are both nonadherent and at high risk for breast cancer. According to 1 study, increasing mammography utilization by 5% in our country would prevent 560 deaths from breast cancer each year.[18]
Several limitations of this study should be considered. First, this cross‐sectional study was conducted at hospitals associated with a single institution and the results may not be generalizable. Second, although physicians' concerns were explored in this study, we did not solicit input about the potential impact of prevention and screening on the nursing staff. Third, there may be concerns about the hypothetical nature of anchoring and possible framing effects with the 2 clinical scenarios. Finally, it is possible that the hospitalists' response may have been subject to social desirability bias. That said, the response to the key question Do you think hospitalists should be involved in breast cancer screening? do not support a socially desirable bias.
Given the current policy emphasis on reducing disparities in cancer screening, it may be reasonable to expand the role of all healthcare providers and healthcare facilities in screening high‐risk populations. Screening tests that may seem difficult to coordinate in hospitals currently may become easier as our hospitals evolve to become more patient centered. Future studies are needed to evaluate the feasibility and potential barriers to inpatient screening mammography.
Disclosure
Disclosures: Dr. Wright is a Miller‐Coulson Family Scholar, and this support comes from Hopkins Center for Innovative Medicine. This work was made possible in part by the Maryland Cigarette Restitution Fund Research Grant at Johns Hopkins. The authors report no conflicts of interest.
Testing for breast cancer is traditionally offered in outpatient settings, and screening mammography rates have plateaued since 2000.[1] Current data suggest that the mammography utilization gap by race has narrowed; however, disparity remains among low‐income, uninsured, and underinsured populations.[2, 3] The lowest compliance with screening mammography recommendations have been reported among women with low income (63.2%), uninsured (50.4%), and those without a usual source of healthcare (43.6%).[4] Although socioeconomic status, access to the healthcare system, and awareness about screening benefits can all influence women's willingness to have screening, the most common reason that women report for not having mammograms were that no one recommended the test.[5, 6] These findings support previous reports that physicians' recommendations about the need for screening mammography is an influential factor in determining women's decisions related to compliance.[7] Hence, the role of healthcare providers in all clinical care settings is pivotal in reducing mammography utilization disparities.
A recent study evaluating the breast cancer screening adherence among the hospitalized women aged 50 to 75 years noted that many (60%) were low income (annual household income <$20,000), 39% were nonadherent, and 35% were at high risk of developing breast cancer.[8] Further, a majority of these hospitalized women were amenable to inpatient screening mammography if due and offered during the hospital stay.[8] As a follow‐up, the purpose of the current study was to explore how hospitalists feel about getting involved in breast cancer screening and ordering screening mammograms for hospitalized women. We hypothesized that a greater proportion of hospitalists would order mammography for hospitalized women who were both overdue for screening and at high risk for developing breast cancer if they fundamentally believe that they have a role in breast cancer screening. This study also explored anticipated barriers that may be of concern to hospitalists when ordering inpatient screening mammography.
METHODS
Study Design and Sample
All hospitalist providers within 4 groups affiliated with Johns Hopkins Medical Institution (Johns Hopkins Hospital, Johns Hopkins Bayview Medical Center, Howard County General Hospital, and Suburban Hospital) were approached for participation in this‐cross sectional study. The hospitalists included physicians, nurse practitioners, and physician assistants. All hospitalists were eligible to participate in the study, and there was no monetary incentive attached to the study participation. A total of 110 hospitalists were approached for study participation. Of these, 4 hospitalists (3.5%) declined to participate, leaving a study population of 106 hospitalists.
Data Collection and Measures
Participants were sent the survey via email using SurveyMonkey. The survey included questions regarding demographic information such as age, gender, race, and clinical experience in hospital medicine. To evaluate for potential personal sources of bias related to mammography, study participants were asked if they have had a family member diagnosed with breast cancer.
A central question asked whether respondents agreed with the following: I believe that hospitalists should be involved in breast cancer screening. The questionnaire also evaluated hospitalists' practical approaches to 2 clinical scenarios by soliciting decision about whether they would order an inpatient screening mammogram. These clinical scenarios were designed using the Gail risk prediction score for probability of developing breast cancer within the next 5 years according to the National Cancer Institute Breast Cancer Risk Tool.[9] Study participants were not provided with the Gail scores and had to infer the risk from the clinical information provided in scenarios. One case described a woman at high risk, and the other with a lower‐risk profile. The first question was: Would you order screening mammography for a 65‐year‐old African American female with obesity and family history for breast cancer admitted to the hospital for cellulitis? She has never had a mammogram and is willing to have it while in hospital. Based on the information provided in the scenario, the 5‐year risk prediction for developing breast cancer using the Gail risk model was high (2.1%). The second scenario asked: Would you order a screening mammography for a 62‐year‐old healthy Hispanic female admitted for presyncope? Patient is uninsured and requests a screening mammogram while in hospital [assume that personal and family histories for breast cancer are negative]. Based on the information provided in the scenario, the 5‐year risk prediction for developing breast cancer using the Gail risk model was low (0.6%).
Several questions regarding potential barriers to inpatient screening mammography were also asked. Some of these questions were based on barriers mentioned in our earlier study of patients,[8] whereas others emerged from a review of the literature and during focus group discussions with hospitalist providers. Pilot testing of the survey was conducted on hospitalists outside the study sample to enhance question clarity. This study was approved by our institutional review board.
Statistical Methods
Respondent characteristics are presented as proportions and means. Unpaired t tests and [2] tests were used to look for associations between demographic characteristics and responses to the question about whether they believe that they should be involved in breast cancer screening. The survey data were analyzed using the Stata statistical software package version 12.1 (StataCorp, College Station, TX).
RESULTS
Out of 106 study subjects willing to participate, 8 did not respond, yielding a response rate of 92%. The mean age of the study participants was 37.6 years, and 55% were female. Almost two‐thirds of study participants (59%) were faculty physicians at an academic hospital, and the average clinical experience as a hospitalist was 4.6 years. Study participants were diverse with respect to ethnicity, and only 30% reported having a family member with breast cancer (Table 1). Because breast cancer is a disease that affects primarily women, stratified analysis by gender showed that most of these characteristic were similar across genders, except fewer women were full time (76% vs 93%, P=0.04) and on the faculty (44% vs 77%, P=0.003).
| Characteristics* | All Participants (n=98) |
|---|---|
| |
| Age, y, mean (SD) | 37.6 (5.5) |
| Female, n (%) | 54 (55) |
| Race, n (%) | |
| Caucasian | 35 (36) |
| African American | 12 (12) |
| Asian | 32 (33) |
| Other | 13 (13) |
| Hospitalist experience, y, mean (SD) | 4.6 (3.5) |
| Full time, n (%) | 82 (84) |
| Family history of breast cancer, n (%) | 30 (30) |
| Faculty physician, n (%) | 58 (59) |
| Believe that hospitalists should be involved in breast cancer screening, n (%) | 35 (38) |
Only 38% believed that hospitalists should be involved with breast cancer screening. The most commonly cited concern related to ordering an inpatient screening mammography was follow‐up of the results of the mammography, followed by the test may not be covered by patient's insurance. As shown in Table 2, these concerns were not perceived differently among providers who believed that hospitalists should be involved in breast cancer screening as compared to those who do not. Demographic variables from Table 1 failed to discern any significant associations related to believing that hospitalists should be involved with breast cancer screening or with concerns about the barriers to screening presented in Table 2 (data not shown). As shown in Table 2, overall, 32% hospitalists were willing to order a screening mammography during a hospital stay for the scenario of the woman at high risk for developing breast cancer (5‐year risk prediction using Gail model 2.1%) and 33% for the low‐risk scenario (5‐year risk prediction using Gail model 0.6%).
| Concern About Screening* | Believe That Hospitalists Should Be Involved in Breast Cancer Screening (n=35) | Do Not Believe That Hospitalists Should Be Involved in Breast Cancer Screening (n=58) | P Value |
|---|---|---|---|
| |||
| Result follow‐up, agree/strongly agree, n (%) | 34 (97) | 51 (88) | 0.25 |
| Interference with patient care, agree/strongly agree, n (%) | 23 (67) | 27 (47) | 0.07 |
| Cost, agree/strongly agree, n (%) | 23 (66) | 28 (48) | 0.10 |
| Concern that the test will not be covered by patient's insurance, agree/strongly agree, n (%) | 23 (66) | 34 (59) | 0.50 |
| Not my responsibility to do cancer prevention, agree/strongly agree, n (%) | 7 (20) | 16 (28) | 0.57 |
| Response to clinical scenarios | |||
| Would order a screening mammogram in the hospital for a high‐risk woman [scenario 1: Gail risk model: 2.1%], n (%) | 23 (66) | 6 (10) | 0.0001 |
| Would order a screening mammography in the hospital for a low‐risk woman [scenario 2: Gail risk model: 0.6%], n (%) | 18 (51) | 13 (22) | 0.004 |
DISCUSSION
Our study suggests that most hospitalists do not believe that they should be involved in breast cancer screening for their hospitalized patients. This perspective was not influenced by either the physician gender, family history for breast cancer, or by the patient's level of risk for developing breast cancer. When patients are in the hospital, both the setting and the acute illness are known to promote reflection and consideration of self‐care.[10] With major healthcare system changes on the horizon and the passing of the Affordable Care Act, we are becoming teams of providers who are collectively responsible for optimal care delivery. It may be possible to increase breast cancer screening rates by educating our patients and offering inpatient screening mammography while they are in the hospital, particularly to those who are at high risk of developing breast cancer.
Physician recommendations for preventive health and screening have consistently been found to be among the strongest predictors of screening utilization.[11] This is the first study to our knowledge that has attempted to understand hospitalists' views and concerns about ordering screening tests to detect occult malignancy. Although addressing preventive care during a hospitalization may seem complex and difficult, helping these women understand their personal risk profile (eg, family history of breast cancer, use of estrogen, race, age, and genetic risk factors) may be what is needed for beginning to influence perspective that might ultimately translate into a willingness to undergo screening.[12, 13, 14] Such delivery of patient‐centered care is built on a foundation of shared decision‐making, which takes into account the patient's preferences, values, and wishes.[15]
Ordering screening mammography for hospitalized patients will require a deeper understanding of hospitalists' attitudes, because the way that these physicians feel about the tests utility will dramatically influence the way that this opportunity is presented to patients, and ultimately the patients' preference to have or forego testing. Our study results are consistent with another publication that highlighted incongruence between physicians' views and patients' preferences for screening practices.[8, 11] Concerns cited, such as interference with patient's acute care, deserve attention, because it may be possible to carry out the screening in ways and at times that do not interfere with treatment or prolong length of stay. Exploring this with a feasibility study will be necessary. Such an approach has been advocated by Trimble et al. for inpatient cervical cancer screening as an efficient strategy to target high‐risk, nonadherent women.[16]
The inpatient setting allows for the elimination of major barriers to screening (like transportation and remembering to get to screening appointments),[8] thereby actively facilitating this needed service. Costs associated with inpatient screening mammography may deter both hospitalists and patients from screening; however, some insurers and Medicare pay for the full cost of screening tests, irrespective of the clinical setting.[17] Further, as hospitals or accountable care organizations become responsible for total cost per beneficiary, screening costs will be preferable when compared with the expenses associated with later detection of pathology and caring for advanced disease states.
One might question whether the mortality benefit of screening mammography is comparable among hospitalized women (who are theoretically sicker and with shorter life expectancy) and those cared for in outpatient practices. Unfortunately, we do not yet know the answer to this question, because data for inpatient screening mammography are nonexistent, and currently this is not considered as a standard of care. However, one can expect the benefits to be similar, if not greater, when performed in the outpatient setting, if preliminary efforts are directed at those who are both nonadherent and at high risk for breast cancer. According to 1 study, increasing mammography utilization by 5% in our country would prevent 560 deaths from breast cancer each year.[18]
Several limitations of this study should be considered. First, this cross‐sectional study was conducted at hospitals associated with a single institution and the results may not be generalizable. Second, although physicians' concerns were explored in this study, we did not solicit input about the potential impact of prevention and screening on the nursing staff. Third, there may be concerns about the hypothetical nature of anchoring and possible framing effects with the 2 clinical scenarios. Finally, it is possible that the hospitalists' response may have been subject to social desirability bias. That said, the response to the key question Do you think hospitalists should be involved in breast cancer screening? do not support a socially desirable bias.
Given the current policy emphasis on reducing disparities in cancer screening, it may be reasonable to expand the role of all healthcare providers and healthcare facilities in screening high‐risk populations. Screening tests that may seem difficult to coordinate in hospitals currently may become easier as our hospitals evolve to become more patient centered. Future studies are needed to evaluate the feasibility and potential barriers to inpatient screening mammography.
Disclosure
Disclosures: Dr. Wright is a Miller‐Coulson Family Scholar, and this support comes from Hopkins Center for Innovative Medicine. This work was made possible in part by the Maryland Cigarette Restitution Fund Research Grant at Johns Hopkins. The authors report no conflicts of interest.
- Centers for Disease Control and Prevention (CDC). Vital signs: breast cancer screening among women aged 50–74 years—United States, 2008. MMWR Morb Mortal Wkly Rep. 2010;59(26):813–816.
- American Cancer Society. Breast Cancer Facts 2013.
- , , , et al. Impact of socioeconomic status on cancer incidence and stage at diagnosis: selected findings from the surveillance, epidemiology, and end results: National Longitudinal Mortality Study. Cancer Causes Control. 2009;20:417–435.
- , , , ; Centers for Disease Control and Prevention. Breast cancer screening among adult women—behavioral risk factor surveillance system, United States, 2010. MMWR Morb Mortal Wkly Rep. 2012;61(suppl):46–50.
- , . Disparities in breast cancer. Curr Probl Cancer. 2007;31(3):134–156.
- , , . Factors associated with mammography utilization: a systematic quantitative review of the literature. J Womens Health (Larchmt). 2008;17:1477–1498.
- , , , et al. Processes of care in cervical and breast cancer screening and follow‐up: the importance of communication. Prev Med. 2004;39:81–90.
- , , , . Breast cancer screening preferences among hospitalized women. J Womens Health (Larchmt). 2013;22(7):637–642.
- , , , et al. Projecting individualized probabilities of developing breast cancer for white females who are being examined annually. J Natl Cancer Inst. 1989;8:1879–1886.
- , , , . Expanding the roles of hospitalist physicians to include public health. J Hosp Med. 2007;2:93–101.
- , , , et al. Colorectal cancer screening: conjoint analysis of consumer preferences and physicians' perceived consumer preferences in the US and Canada. Paper presented at: 27th Annual Meeting of the Society for Medical Decision Making; October 21–24, 2005; San Francisco, CA.
- , , . Family history of breast cancer: impact on the disease experience. Cancer Pract. 2000;8:135–142.
- , , , . Breast cancer knowledge and attitudes toward mammography as predictors of breast cancer preventive behavior in Kazakh, Korean, and Russian women in Kazakhstan. Int J Public Health. 2008;53:123–130.
- , , , , . The relation between projected breast cancer risk, perceived cancer risk, and mammography use. Results from the National Health Interview Survey. J Gen Intern Med. 2006;21:158–164.
- , . Patient‐centered communication in cancer care: promoting healing and reducing suffering. NIH publication no. 07‐6225. Bethesda, MD: National Cancer Institute, 2007.
- , , , , , . Effectiveness of screening for cervical cancer in an inpatient hospital setting. Obstet Gynecol. 2004;103(2):310–316.
- Centers for Medicare 38:600–609.
- Centers for Disease Control and Prevention (CDC). Vital signs: breast cancer screening among women aged 50–74 years—United States, 2008. MMWR Morb Mortal Wkly Rep. 2010;59(26):813–816.
- American Cancer Society. Breast Cancer Facts 2013.
- , , , et al. Impact of socioeconomic status on cancer incidence and stage at diagnosis: selected findings from the surveillance, epidemiology, and end results: National Longitudinal Mortality Study. Cancer Causes Control. 2009;20:417–435.
- , , , ; Centers for Disease Control and Prevention. Breast cancer screening among adult women—behavioral risk factor surveillance system, United States, 2010. MMWR Morb Mortal Wkly Rep. 2012;61(suppl):46–50.
- , . Disparities in breast cancer. Curr Probl Cancer. 2007;31(3):134–156.
- , , . Factors associated with mammography utilization: a systematic quantitative review of the literature. J Womens Health (Larchmt). 2008;17:1477–1498.
- , , , et al. Processes of care in cervical and breast cancer screening and follow‐up: the importance of communication. Prev Med. 2004;39:81–90.
- , , , . Breast cancer screening preferences among hospitalized women. J Womens Health (Larchmt). 2013;22(7):637–642.
- , , , et al. Projecting individualized probabilities of developing breast cancer for white females who are being examined annually. J Natl Cancer Inst. 1989;8:1879–1886.
- , , , . Expanding the roles of hospitalist physicians to include public health. J Hosp Med. 2007;2:93–101.
- , , , et al. Colorectal cancer screening: conjoint analysis of consumer preferences and physicians' perceived consumer preferences in the US and Canada. Paper presented at: 27th Annual Meeting of the Society for Medical Decision Making; October 21–24, 2005; San Francisco, CA.
- , , . Family history of breast cancer: impact on the disease experience. Cancer Pract. 2000;8:135–142.
- , , , . Breast cancer knowledge and attitudes toward mammography as predictors of breast cancer preventive behavior in Kazakh, Korean, and Russian women in Kazakhstan. Int J Public Health. 2008;53:123–130.
- , , , , . The relation between projected breast cancer risk, perceived cancer risk, and mammography use. Results from the National Health Interview Survey. J Gen Intern Med. 2006;21:158–164.
- , . Patient‐centered communication in cancer care: promoting healing and reducing suffering. NIH publication no. 07‐6225. Bethesda, MD: National Cancer Institute, 2007.
- , , , , , . Effectiveness of screening for cervical cancer in an inpatient hospital setting. Obstet Gynecol. 2004;103(2):310–316.
- Centers for Medicare 38:600–609.
© 2015 Society of Hospital Medicine
Safe Discharge in Bronchiolitis
Bronchiolitis is the most common cause of hospitalization in infancy, with estimated annual US costs of over $1.7 billion.[1] The last 2 decades have seen numerous thoughtful and well‐designed research studies but little improvement in the value of care.[1, 2, 3, 4] The diagnosis and treatment section of the recently released 2014 American Academy of Pediatrics (AAP) Clinical Practice Guideline for bronchiolitis contains 7 should not's and 3 should's,[3] with the only clear affirmative recommendations related to the history and physical and to the use of supplemental fluids. As supported by several systematic reviews and randomized controlled trials, the use of respiratory treatments, including ‐agonists, racemic epinephrine, and hypertonic saline, was discouraged. There continues to be significant variation in care for patients with bronchiolitis[5, 6] and rigorous evidence was lacking on when a child could be safely discharged home.
Mansbach and colleagues in the Multicenter Airway Research Collaboration (MARC‐30) provide the best evidence to date on the clinical course of bronchiolitis and present multicenter data upon which to build evidence‐based discharge criteria.[7] In their prospective cohort study of 16 US children's hospitals, Mansbach et al. sought to answer 3 research questions: (1) In infants hospitalized with bronchiolitis, what is the time to clinical improvement? (2) What is the risk of clinical worsening after standardized improvement criteria are met? (3) What discharge criteria might balance both timely discharge and very low readmission risk? In an analytic cohort of 1916 children 2 years of age with a physician diagnosis of bronchiolitis, the time from onset of difficulty breathing until clinical improvement was a median of 4 days, with a 75th percentile of 7.5 days. Of the 1702 children who clinically improved before discharge, only 76 (4%) then worsened. Although there are some limitations to how these criteria were assessed, the authors' work supports discharge criteria of (1) no or mild and stable or improving retractions, (2) stable or improving respiratory rate that is below the 90th percentile for age, (3) estimated room air saturation of 90% without any points 88%, and (4) clinician assessment of the child maintaining adequate oral hydration, regardless of use of intravenous fluids.
Three limitations warrant consideration when interpreting the study results. First, the MARC‐30 investigators oversampled from the intensive care unit and excluded 109 children with a hospital length of stay (LOS) 1 day. Although it is uncertain what effect these decisions would have on worsening after improving, both would overestimate the LOS in the sampled population at study hospitals. It is likely that the median LOS and 75th percentile of 4 and 7.5 days, respectively, are higher than what hospital medicine physicians saw at these hospitals. Second, the study team did not use a scoring tool. The authors note that the holistic assessments clinicians used to estimate respiratory rate and oxygen saturation may be more similar to standard clinical practice more than a calculated mean. This raises an important question: If less numerous data might lead to more information and knowledge, might they also lead to reliability and validity concerns? Given an absence of a structured, validated assessment of these severity indicators, it seems possible clinicians worked backward from the holistic assessment of this child is ready to go home and then entered data to support their larger assessment. This would tend to bias toward lower proportions of worsening after clinical improvement. Third, the once‐daily review of the medical record led to less precise estimates of each event including time from difficulty breathing to improvement and LOS. In addition to the absence of a scoring tool, this likely adds a modest bias toward underdetection of clinical worsening after improvement, because observations from discharged children were effectively censored from analysis. Importantly the low readmission rates suggest neither of those biases is substantial.
Several of the findings in this article support recent changes to the recommendations in the 2014 AAP Bronchiolitis Clinical Practice Guideline.[3] Although there is no recommendation on discharge readiness, Mansbach and colleagues found that an operationalization of the core criteria outlined in the 2006 version of the AAP Bronchiolitis Clinical Practice Guideline would result in a low proportion of subsequent clinical worsening.[8] This study also informs and supports an additional change to the AAP's 2006 guideline recommendation on continuous pulse oximetry. Key Action Statement 6b in the 2014 guideline notes Clinicians may choose not to use continuous pulse oximetry for infants and children with a diagnosis of bronchiolitis, expanding the recommendation from the 2006 guideline discouraging continuous pulse oximetry as the child's clinical course improves.[3, 8] Mansbach and colleagues found that removing the lower desaturation threshold of 88% improved the percentage of children who met criteria, with no changes in proportion subsequently worsening. With an improvement criterion of average oxygen saturation threshold of 95%, less than half of the children met this criteria before discharge, and an increased percentage (5%) clinically worsened, presumably due to clinically inconsequential desaturations to 94%. The less stringent the pulse oximetry criteria, the better their improvement criteria performed. This study adds to the modest literature on how overuse of continuous pulse oximetry may prolong hospitalization, leading to nonvalue‐added care and potentially increasing the risk of iatrogenic harm.[9, 10, 11]
Another strength of this study is the extensive viral testing on nasal aspirates. The absence of an association between individual viral pathogen or coinfection on the risk of worsening after improving further supports the recommendation against viral testing. The authors also identified a large group of children with a very low risk of worsening after an improving course: children 2 months, born at term, and who did not present with severe retractions. This finding, which will resonate with clinicians who care for patients with bronchiolitis, provides additional data on a group likely to have short hospitalization and unlikely to benefit from therapies. It also identifies a group of children with increased risk of worsening, which could be targeted for future research efforts on therapies such as hypertonic saline and high‐flow nasal cannula, where the evidence is mixed and inconclusive.
Both the MARC‐30 study and the 2014 AAP guidelines are tremendous contributions to the scientific literature on this common, costly, and often frustrating disease for clinicians and families alike. More important, however, will be implementation and dissemination efforts to ensure children benefit from this new knowledge. After the 2006 AAP guidelines, there was some evidence of improved care[12] but remaining profound hospital‐level variation.[5] Immediate next steps to improve bronchiolitis care should include interventions to standardize evidence‐based discharge criteria and reduce the overuse of nonevidence‐based care. Local clinical practice guidelines aid in the early phases of standardization, but without work and willpower in the implementation and sustain phase, their effect may be modest.[13] This study and the new guideline raise several important T3[14] or how questions for pediatric hospital medicine clinicians, researchers, and improvers. First, how can evidence‐based discharge criteria, such as those presented here, be applied reliably and broadly at the point of care? White and colleagues at Cincinnati shared a strategy that will benefit from further testing and adaptation.[15] Second, how can continuous pulse oximetry be either greatly reduced or have its data put in a broader context to inform decision making? Relatedly, which strategy is more effective and for whom? Finally, what incentives at the hospital and policy level are most effective in helping physicians to choose wisely[16] and do less?
Answering these questions will be crucial to ensure the knowledge produced from Mansbach and colleagues benefits the hundreds of thousands of children hospitalized with bronchiolitis each year.
Disclosure
Nothing to report.
- , , , , . Trends in bronchiolitis hospitalizations in the United States, 2000–2009. Pediatrics. 2013;132(1):28–36.
- , , , , , . Bronchiolitis‐associated hospitalizations among US children, 1980–1996. JAMA. 1999;282(15):1440–1446.
- , , , et al. Clinical practice guideline: the diagnosis, management, and prevention of bronchiolitis. Pediatrics. 2014;134(5):e1474–e1502.
- , , , , . Bronchiolitis‐associated mortality and estimates of respiratory syncytial virus‐associated deaths among US children, 1979–1997. J Infect Dis. 2001;183(1):16–22.
- , , , , , . Variation in the management of infants hospitalized for bronchiolitis persists after the 2006 American Academy of Pediatrics bronchiolitis guidelines. J Pediatr. 2014;165(4):786–792.e781.
- , , , , . Population variation in admission rates and duration of inpatient stay for bronchiolitis in England. Arch Dis Child. 2013;98(1):57–59.
- , , , et al.; MARC‐30 Investigators. Hospital course and discharge criteria for children hospitalized with bronchiolitis. J Hosp Med. 2015;10(4):205–211.
- American Academy of Pediatrics Subcommittee on Diagnosis and Management of Bronchiolitis. Diagnosis and management of bronchiolitis. Pediatrics. 2006;118(4):1774–1793.
- , , , . Impact of pulse oximetry and oxygen therapy on length of stay in bronchiolitis hospitalizations. Arch Pediatr Adolesc Med. 2004;158(6):527–530.
- , . Observational study of two oxygen saturation targets for discharge in bronchiolitis. Arch Dis Child. 2012;97(4):361–363.
- , , , . Preventable adverse events in infants hospitalized with bronchiolitis. Pediatrics. 2005;116(3):603–608.
- , , . Bronchiolitis management before and after the AAP guidelines. Pediatrics. 2014;133(1):e1–e7.
- , , , et al. Impact of inpatient bronchiolitis clinical practice guideline implementation on testing and treatment. J Pediatr. 2014;165(3):570–576.e573.
- , . The "3T's" road map to transform US health care: the "how" of high‐quality care. JAMA. 2008;299(19):2319–2321.
- , , , et al. Using quality improvement to optimise paediatric discharge efficiency. BMJ Qual Saf. 2014;23(5):428–436.
- , , , et al. Choosing wisely in pediatric hospital medicine: five opportunities for improved healthcare value. J Hosp Med. 2013;8(9):479–485.
Bronchiolitis is the most common cause of hospitalization in infancy, with estimated annual US costs of over $1.7 billion.[1] The last 2 decades have seen numerous thoughtful and well‐designed research studies but little improvement in the value of care.[1, 2, 3, 4] The diagnosis and treatment section of the recently released 2014 American Academy of Pediatrics (AAP) Clinical Practice Guideline for bronchiolitis contains 7 should not's and 3 should's,[3] with the only clear affirmative recommendations related to the history and physical and to the use of supplemental fluids. As supported by several systematic reviews and randomized controlled trials, the use of respiratory treatments, including ‐agonists, racemic epinephrine, and hypertonic saline, was discouraged. There continues to be significant variation in care for patients with bronchiolitis[5, 6] and rigorous evidence was lacking on when a child could be safely discharged home.
Mansbach and colleagues in the Multicenter Airway Research Collaboration (MARC‐30) provide the best evidence to date on the clinical course of bronchiolitis and present multicenter data upon which to build evidence‐based discharge criteria.[7] In their prospective cohort study of 16 US children's hospitals, Mansbach et al. sought to answer 3 research questions: (1) In infants hospitalized with bronchiolitis, what is the time to clinical improvement? (2) What is the risk of clinical worsening after standardized improvement criteria are met? (3) What discharge criteria might balance both timely discharge and very low readmission risk? In an analytic cohort of 1916 children 2 years of age with a physician diagnosis of bronchiolitis, the time from onset of difficulty breathing until clinical improvement was a median of 4 days, with a 75th percentile of 7.5 days. Of the 1702 children who clinically improved before discharge, only 76 (4%) then worsened. Although there are some limitations to how these criteria were assessed, the authors' work supports discharge criteria of (1) no or mild and stable or improving retractions, (2) stable or improving respiratory rate that is below the 90th percentile for age, (3) estimated room air saturation of 90% without any points 88%, and (4) clinician assessment of the child maintaining adequate oral hydration, regardless of use of intravenous fluids.
Three limitations warrant consideration when interpreting the study results. First, the MARC‐30 investigators oversampled from the intensive care unit and excluded 109 children with a hospital length of stay (LOS) 1 day. Although it is uncertain what effect these decisions would have on worsening after improving, both would overestimate the LOS in the sampled population at study hospitals. It is likely that the median LOS and 75th percentile of 4 and 7.5 days, respectively, are higher than what hospital medicine physicians saw at these hospitals. Second, the study team did not use a scoring tool. The authors note that the holistic assessments clinicians used to estimate respiratory rate and oxygen saturation may be more similar to standard clinical practice more than a calculated mean. This raises an important question: If less numerous data might lead to more information and knowledge, might they also lead to reliability and validity concerns? Given an absence of a structured, validated assessment of these severity indicators, it seems possible clinicians worked backward from the holistic assessment of this child is ready to go home and then entered data to support their larger assessment. This would tend to bias toward lower proportions of worsening after clinical improvement. Third, the once‐daily review of the medical record led to less precise estimates of each event including time from difficulty breathing to improvement and LOS. In addition to the absence of a scoring tool, this likely adds a modest bias toward underdetection of clinical worsening after improvement, because observations from discharged children were effectively censored from analysis. Importantly the low readmission rates suggest neither of those biases is substantial.
Several of the findings in this article support recent changes to the recommendations in the 2014 AAP Bronchiolitis Clinical Practice Guideline.[3] Although there is no recommendation on discharge readiness, Mansbach and colleagues found that an operationalization of the core criteria outlined in the 2006 version of the AAP Bronchiolitis Clinical Practice Guideline would result in a low proportion of subsequent clinical worsening.[8] This study also informs and supports an additional change to the AAP's 2006 guideline recommendation on continuous pulse oximetry. Key Action Statement 6b in the 2014 guideline notes Clinicians may choose not to use continuous pulse oximetry for infants and children with a diagnosis of bronchiolitis, expanding the recommendation from the 2006 guideline discouraging continuous pulse oximetry as the child's clinical course improves.[3, 8] Mansbach and colleagues found that removing the lower desaturation threshold of 88% improved the percentage of children who met criteria, with no changes in proportion subsequently worsening. With an improvement criterion of average oxygen saturation threshold of 95%, less than half of the children met this criteria before discharge, and an increased percentage (5%) clinically worsened, presumably due to clinically inconsequential desaturations to 94%. The less stringent the pulse oximetry criteria, the better their improvement criteria performed. This study adds to the modest literature on how overuse of continuous pulse oximetry may prolong hospitalization, leading to nonvalue‐added care and potentially increasing the risk of iatrogenic harm.[9, 10, 11]
Another strength of this study is the extensive viral testing on nasal aspirates. The absence of an association between individual viral pathogen or coinfection on the risk of worsening after improving further supports the recommendation against viral testing. The authors also identified a large group of children with a very low risk of worsening after an improving course: children 2 months, born at term, and who did not present with severe retractions. This finding, which will resonate with clinicians who care for patients with bronchiolitis, provides additional data on a group likely to have short hospitalization and unlikely to benefit from therapies. It also identifies a group of children with increased risk of worsening, which could be targeted for future research efforts on therapies such as hypertonic saline and high‐flow nasal cannula, where the evidence is mixed and inconclusive.
Both the MARC‐30 study and the 2014 AAP guidelines are tremendous contributions to the scientific literature on this common, costly, and often frustrating disease for clinicians and families alike. More important, however, will be implementation and dissemination efforts to ensure children benefit from this new knowledge. After the 2006 AAP guidelines, there was some evidence of improved care[12] but remaining profound hospital‐level variation.[5] Immediate next steps to improve bronchiolitis care should include interventions to standardize evidence‐based discharge criteria and reduce the overuse of nonevidence‐based care. Local clinical practice guidelines aid in the early phases of standardization, but without work and willpower in the implementation and sustain phase, their effect may be modest.[13] This study and the new guideline raise several important T3[14] or how questions for pediatric hospital medicine clinicians, researchers, and improvers. First, how can evidence‐based discharge criteria, such as those presented here, be applied reliably and broadly at the point of care? White and colleagues at Cincinnati shared a strategy that will benefit from further testing and adaptation.[15] Second, how can continuous pulse oximetry be either greatly reduced or have its data put in a broader context to inform decision making? Relatedly, which strategy is more effective and for whom? Finally, what incentives at the hospital and policy level are most effective in helping physicians to choose wisely[16] and do less?
Answering these questions will be crucial to ensure the knowledge produced from Mansbach and colleagues benefits the hundreds of thousands of children hospitalized with bronchiolitis each year.
Disclosure
Nothing to report.
Bronchiolitis is the most common cause of hospitalization in infancy, with estimated annual US costs of over $1.7 billion.[1] The last 2 decades have seen numerous thoughtful and well‐designed research studies but little improvement in the value of care.[1, 2, 3, 4] The diagnosis and treatment section of the recently released 2014 American Academy of Pediatrics (AAP) Clinical Practice Guideline for bronchiolitis contains 7 should not's and 3 should's,[3] with the only clear affirmative recommendations related to the history and physical and to the use of supplemental fluids. As supported by several systematic reviews and randomized controlled trials, the use of respiratory treatments, including ‐agonists, racemic epinephrine, and hypertonic saline, was discouraged. There continues to be significant variation in care for patients with bronchiolitis[5, 6] and rigorous evidence was lacking on when a child could be safely discharged home.
Mansbach and colleagues in the Multicenter Airway Research Collaboration (MARC‐30) provide the best evidence to date on the clinical course of bronchiolitis and present multicenter data upon which to build evidence‐based discharge criteria.[7] In their prospective cohort study of 16 US children's hospitals, Mansbach et al. sought to answer 3 research questions: (1) In infants hospitalized with bronchiolitis, what is the time to clinical improvement? (2) What is the risk of clinical worsening after standardized improvement criteria are met? (3) What discharge criteria might balance both timely discharge and very low readmission risk? In an analytic cohort of 1916 children 2 years of age with a physician diagnosis of bronchiolitis, the time from onset of difficulty breathing until clinical improvement was a median of 4 days, with a 75th percentile of 7.5 days. Of the 1702 children who clinically improved before discharge, only 76 (4%) then worsened. Although there are some limitations to how these criteria were assessed, the authors' work supports discharge criteria of (1) no or mild and stable or improving retractions, (2) stable or improving respiratory rate that is below the 90th percentile for age, (3) estimated room air saturation of 90% without any points 88%, and (4) clinician assessment of the child maintaining adequate oral hydration, regardless of use of intravenous fluids.
Three limitations warrant consideration when interpreting the study results. First, the MARC‐30 investigators oversampled from the intensive care unit and excluded 109 children with a hospital length of stay (LOS) 1 day. Although it is uncertain what effect these decisions would have on worsening after improving, both would overestimate the LOS in the sampled population at study hospitals. It is likely that the median LOS and 75th percentile of 4 and 7.5 days, respectively, are higher than what hospital medicine physicians saw at these hospitals. Second, the study team did not use a scoring tool. The authors note that the holistic assessments clinicians used to estimate respiratory rate and oxygen saturation may be more similar to standard clinical practice more than a calculated mean. This raises an important question: If less numerous data might lead to more information and knowledge, might they also lead to reliability and validity concerns? Given an absence of a structured, validated assessment of these severity indicators, it seems possible clinicians worked backward from the holistic assessment of this child is ready to go home and then entered data to support their larger assessment. This would tend to bias toward lower proportions of worsening after clinical improvement. Third, the once‐daily review of the medical record led to less precise estimates of each event including time from difficulty breathing to improvement and LOS. In addition to the absence of a scoring tool, this likely adds a modest bias toward underdetection of clinical worsening after improvement, because observations from discharged children were effectively censored from analysis. Importantly the low readmission rates suggest neither of those biases is substantial.
Several of the findings in this article support recent changes to the recommendations in the 2014 AAP Bronchiolitis Clinical Practice Guideline.[3] Although there is no recommendation on discharge readiness, Mansbach and colleagues found that an operationalization of the core criteria outlined in the 2006 version of the AAP Bronchiolitis Clinical Practice Guideline would result in a low proportion of subsequent clinical worsening.[8] This study also informs and supports an additional change to the AAP's 2006 guideline recommendation on continuous pulse oximetry. Key Action Statement 6b in the 2014 guideline notes Clinicians may choose not to use continuous pulse oximetry for infants and children with a diagnosis of bronchiolitis, expanding the recommendation from the 2006 guideline discouraging continuous pulse oximetry as the child's clinical course improves.[3, 8] Mansbach and colleagues found that removing the lower desaturation threshold of 88% improved the percentage of children who met criteria, with no changes in proportion subsequently worsening. With an improvement criterion of average oxygen saturation threshold of 95%, less than half of the children met this criteria before discharge, and an increased percentage (5%) clinically worsened, presumably due to clinically inconsequential desaturations to 94%. The less stringent the pulse oximetry criteria, the better their improvement criteria performed. This study adds to the modest literature on how overuse of continuous pulse oximetry may prolong hospitalization, leading to nonvalue‐added care and potentially increasing the risk of iatrogenic harm.[9, 10, 11]
Another strength of this study is the extensive viral testing on nasal aspirates. The absence of an association between individual viral pathogen or coinfection on the risk of worsening after improving further supports the recommendation against viral testing. The authors also identified a large group of children with a very low risk of worsening after an improving course: children 2 months, born at term, and who did not present with severe retractions. This finding, which will resonate with clinicians who care for patients with bronchiolitis, provides additional data on a group likely to have short hospitalization and unlikely to benefit from therapies. It also identifies a group of children with increased risk of worsening, which could be targeted for future research efforts on therapies such as hypertonic saline and high‐flow nasal cannula, where the evidence is mixed and inconclusive.
Both the MARC‐30 study and the 2014 AAP guidelines are tremendous contributions to the scientific literature on this common, costly, and often frustrating disease for clinicians and families alike. More important, however, will be implementation and dissemination efforts to ensure children benefit from this new knowledge. After the 2006 AAP guidelines, there was some evidence of improved care[12] but remaining profound hospital‐level variation.[5] Immediate next steps to improve bronchiolitis care should include interventions to standardize evidence‐based discharge criteria and reduce the overuse of nonevidence‐based care. Local clinical practice guidelines aid in the early phases of standardization, but without work and willpower in the implementation and sustain phase, their effect may be modest.[13] This study and the new guideline raise several important T3[14] or how questions for pediatric hospital medicine clinicians, researchers, and improvers. First, how can evidence‐based discharge criteria, such as those presented here, be applied reliably and broadly at the point of care? White and colleagues at Cincinnati shared a strategy that will benefit from further testing and adaptation.[15] Second, how can continuous pulse oximetry be either greatly reduced or have its data put in a broader context to inform decision making? Relatedly, which strategy is more effective and for whom? Finally, what incentives at the hospital and policy level are most effective in helping physicians to choose wisely[16] and do less?
Answering these questions will be crucial to ensure the knowledge produced from Mansbach and colleagues benefits the hundreds of thousands of children hospitalized with bronchiolitis each year.
Disclosure
Nothing to report.
- , , , , . Trends in bronchiolitis hospitalizations in the United States, 2000–2009. Pediatrics. 2013;132(1):28–36.
- , , , , , . Bronchiolitis‐associated hospitalizations among US children, 1980–1996. JAMA. 1999;282(15):1440–1446.
- , , , et al. Clinical practice guideline: the diagnosis, management, and prevention of bronchiolitis. Pediatrics. 2014;134(5):e1474–e1502.
- , , , , . Bronchiolitis‐associated mortality and estimates of respiratory syncytial virus‐associated deaths among US children, 1979–1997. J Infect Dis. 2001;183(1):16–22.
- , , , , , . Variation in the management of infants hospitalized for bronchiolitis persists after the 2006 American Academy of Pediatrics bronchiolitis guidelines. J Pediatr. 2014;165(4):786–792.e781.
- , , , , . Population variation in admission rates and duration of inpatient stay for bronchiolitis in England. Arch Dis Child. 2013;98(1):57–59.
- , , , et al.; MARC‐30 Investigators. Hospital course and discharge criteria for children hospitalized with bronchiolitis. J Hosp Med. 2015;10(4):205–211.
- American Academy of Pediatrics Subcommittee on Diagnosis and Management of Bronchiolitis. Diagnosis and management of bronchiolitis. Pediatrics. 2006;118(4):1774–1793.
- , , , . Impact of pulse oximetry and oxygen therapy on length of stay in bronchiolitis hospitalizations. Arch Pediatr Adolesc Med. 2004;158(6):527–530.
- , . Observational study of two oxygen saturation targets for discharge in bronchiolitis. Arch Dis Child. 2012;97(4):361–363.
- , , , . Preventable adverse events in infants hospitalized with bronchiolitis. Pediatrics. 2005;116(3):603–608.
- , , . Bronchiolitis management before and after the AAP guidelines. Pediatrics. 2014;133(1):e1–e7.
- , , , et al. Impact of inpatient bronchiolitis clinical practice guideline implementation on testing and treatment. J Pediatr. 2014;165(3):570–576.e573.
- , . The "3T's" road map to transform US health care: the "how" of high‐quality care. JAMA. 2008;299(19):2319–2321.
- , , , et al. Using quality improvement to optimise paediatric discharge efficiency. BMJ Qual Saf. 2014;23(5):428–436.
- , , , et al. Choosing wisely in pediatric hospital medicine: five opportunities for improved healthcare value. J Hosp Med. 2013;8(9):479–485.
- , , , , . Trends in bronchiolitis hospitalizations in the United States, 2000–2009. Pediatrics. 2013;132(1):28–36.
- , , , , , . Bronchiolitis‐associated hospitalizations among US children, 1980–1996. JAMA. 1999;282(15):1440–1446.
- , , , et al. Clinical practice guideline: the diagnosis, management, and prevention of bronchiolitis. Pediatrics. 2014;134(5):e1474–e1502.
- , , , , . Bronchiolitis‐associated mortality and estimates of respiratory syncytial virus‐associated deaths among US children, 1979–1997. J Infect Dis. 2001;183(1):16–22.
- , , , , , . Variation in the management of infants hospitalized for bronchiolitis persists after the 2006 American Academy of Pediatrics bronchiolitis guidelines. J Pediatr. 2014;165(4):786–792.e781.
- , , , , . Population variation in admission rates and duration of inpatient stay for bronchiolitis in England. Arch Dis Child. 2013;98(1):57–59.
- , , , et al.; MARC‐30 Investigators. Hospital course and discharge criteria for children hospitalized with bronchiolitis. J Hosp Med. 2015;10(4):205–211.
- American Academy of Pediatrics Subcommittee on Diagnosis and Management of Bronchiolitis. Diagnosis and management of bronchiolitis. Pediatrics. 2006;118(4):1774–1793.
- , , , . Impact of pulse oximetry and oxygen therapy on length of stay in bronchiolitis hospitalizations. Arch Pediatr Adolesc Med. 2004;158(6):527–530.
- , . Observational study of two oxygen saturation targets for discharge in bronchiolitis. Arch Dis Child. 2012;97(4):361–363.
- , , , . Preventable adverse events in infants hospitalized with bronchiolitis. Pediatrics. 2005;116(3):603–608.
- , , . Bronchiolitis management before and after the AAP guidelines. Pediatrics. 2014;133(1):e1–e7.
- , , , et al. Impact of inpatient bronchiolitis clinical practice guideline implementation on testing and treatment. J Pediatr. 2014;165(3):570–576.e573.
- , . The "3T's" road map to transform US health care: the "how" of high‐quality care. JAMA. 2008;299(19):2319–2321.
- , , , et al. Using quality improvement to optimise paediatric discharge efficiency. BMJ Qual Saf. 2014;23(5):428–436.
- , , , et al. Choosing wisely in pediatric hospital medicine: five opportunities for improved healthcare value. J Hosp Med. 2013;8(9):479–485.
Bronchiolitis and Discharge Criteria
Although bronchiolitis is the leading cause of hospitalization for US infants,[1] there is a lack of basic prospective data about the expected inpatient clinical course and ongoing uncertainty about when a hospitalized child is ready for discharge to home.[2] This lack of data about children's readiness for discharge may result in variable hospital length‐of‐stay (LOS).[3, 4, 5]
One specific source of variability in discharge readiness and LOS variability may be the lack of consensus about safe threshold oxygen saturation values for discharge in children hospitalized with bronchiolitis.[6, 7] In 2006, the Scottish Intercollegiate Guidelines Network recommended a discharge room air oxygen (RAO2) saturation threshold of 95%.[8] The same year, the American Academy of Pediatrics (AAP) bronchiolitis clinical practice guideline stated that oxygen is not needed for children with RAO2 saturations 90% who are feeding well and have minimal respiratory distress.[9] There is a need for prospective studies to help clinicians make evidenced‐based discharge decisions for this common condition.
We performed a prospective, multicenter, multiyear study[10, 11, 12] to examine the typical inpatient clinical course of and to develop hospital discharge guidelines for children age <2 years hospitalized with bronchiolitis. We hypothesized that children would not worsen clinically and would be safe to discharge home once their respiratory status improved and they were able to remain hydrated.
METHODS
Study Design and Population
We conducted a prospective, multicenter cohort study for 3 consecutive years during the 2007 to 2010 winter seasons, as part of the Multicenter Airway Research Collaboration (MARC), a program of the Emergency Medicine Network (
All patients were treated at the discretion of the treating physician. Inclusion criteria were an attending physician's diagnosis of bronchiolitis, age <2 years, and the ability of the parent/guardian to give informed consent. The exclusion criteria were previous enrollment and transfer to a participating hospital >48 hours after the original admission time. Therefore, children with comorbid conditions were included in this study. All consent and data forms were translated into Spanish. The institutional review board at each of the 16 participating hospitals approved the study.
Of the 2207 enrolled children, we excluded 109 (5%) children with a hospital LOS <1 day due to inadequate time to capture the required data for the present analysis. Among the 2098 remaining children, 1916 (91%) had daily inpatient data on all factors used to define clinical improvement and clinical worsening. Thus, the analytic cohort was comprised of 1916 children hospitalized for bronchiolitis.
Data Collection
Investigators conducted detailed structured interviews. Chart reviews were conducted to obtain preadmission and daily hospital clinical data including respiratory rates, daily respiratory rate trends, degree of retractions, oxygen saturation, daily oxygen saturation trends, medical management, and disposition. These data were manually reviewed, and site investigators were queried about missing data and discrepancies. A follow‐up telephone interview was conducted with families 1 week after discharge to examine relapse events at both 24 hours and 7 days.
We used the question: How long ago did the following symptoms [eg, difficulty breathing] begin [for the] current illness? to estimate the onset of the current illness. Pulse was categorized as low, normal, or high based on age‐related heart rate values.[13] Presence of apnea was recorded daily by site investigators.[14]
Nasopharyngeal Aspirate Collection and Virology Testing
As described previously, site teams used a standardized protocol to collect nasopharyngeal aspirates,[11] which were tested for respiratory syncytial virus (RSV) types A and B; rhinovirus (RV); parainfluenza virus types 1, 2, and 3; influenza virus types A and B; 2009 novel H1N1; human metapneumovirus; coronaviruses NL‐63, HKU1, OC43, and 229E; enterovirus, and adenovirus using polymerase chain reaction.[11, 15, 16, 17]
Defining Clinical Improvement and Worsening
Clinical improvement criteria were based on the 2006 AAP guidelines.[9] For respiratory rate and oxygen saturation, clinicians estimated average daily respiratory rate and oxygen saturation based on the recorded readings from the previous 24 hours. This estimation reflects the process clinicians use when rounding on their hospitalized patients, and thus may be more similar to standard clinical practice than a calculated mean. The respiratory rate criteria are adjusted for age.[18, 19] For daily estimated average oxygen saturation we used the AAP criteria of RAO2 saturation of 90%. Considering that oxygen saturation is the main determinant of LOS,[20] healthy infants age <6 months may have transient oxygen saturations of around 80%,[21] and that errors in estimation may occur, we included a lowest RAO2 of 88% in our improvement criteria. By combining the dichotomized estimated oxygen saturation (90% or not) with the lower limit of 88%, there was little room for erroneous conclusions. A child was considered clinically improved on the earliest date he/she met all of the following criteria: (1) none or mild retractions and improved or stable retractions compared with the previous inpatient day; (2) daily estimated average respiratory rate (RR) <60 breaths per minute for age <6 months, <55 breaths/minute for age 6 to 11 months, and <45 breaths/minute for age 12 months with a decreasing or stable trend over the course of the current day; (3) daily estimated average RAO2 saturation 90%, lowest RAO2 saturation 88%[21]; and (4) not receiving intravenous (IV) fluids or for children receiving IV fluids a clinician report of the child maintaining oral hydration. Children who reached the clinical improvement criteria were considered clinically worse if they required intensive care or had the inverse of 1 of the improvement criteria: moderate/severe retractions that were worse compared with the previous inpatient day, daily average RR 60 with an increasing trend over the current day, need for oxygen, or need for IV fluids.
Statistical Analyses
All analyses were performed using Stata 12.0 (StataCorp, College Station, TX). Data are presented as proportions with 95% confidence intervals (95% CIs), means with standard deviations, and medians with interquartile ranges (IQR). To examine potential factors associated with clinical worsening after reaching clinical improvement, we used 2, Fisher exact, Student t test, and Kruskall‐Wallis tests, as appropriate.
Adjusted analyses used generalized linear mixed models with a logit link to identify independent risk factors for worsening after reaching clinical improvement. Fixed effects for patient‐level factors and a random site effect were used. Factors were tested for inclusion in the multivariable model if they were found to be associated with worsening in unadjusted analyses (P<0.20) or were considered clinically important. Results are reported as odds ratios with 95% CIs.
We performed several sensitivity analyses to evaluate these improvement criteria: (1) we excluded the lowest RAO2 saturation requirement of 88%, (2) we examined a 94% daily estimated average RAO2 saturation threshold,[22] (3) we examined a 95% daily estimated average RAO2 saturation threshold,[8] and (4) we examined children age <12 months with no history of wheeze.
RESULTS
There were 1916 children hospitalized with bronchiolitis with data on all factors used to define clinical improvement and clinical worsening. The median number of days from the beginning of difficulty breathing until admission was 2 days (IQR, 15.5 days; range, 18 days) and from the beginning of difficulty breathing until clinical improvement was 4 days (IQR, 37.5 days; range, 133 days) (Figure 1). The variance for days to admission was significantly less than the variance for days to clinical improvement (P<0.001).
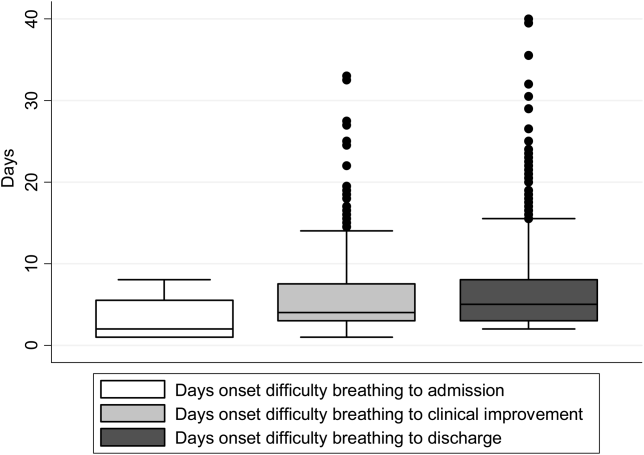
In this observational study, clinicians discharged 214 (11%) of the 1916 children before meeting the definition of clinical improvement. Thus, 1702 (89%; 95% CI: 87%‐90%) children reached the clinical improvement criteria, had a LOS >1 day, and had data on all factors (Figure 2).
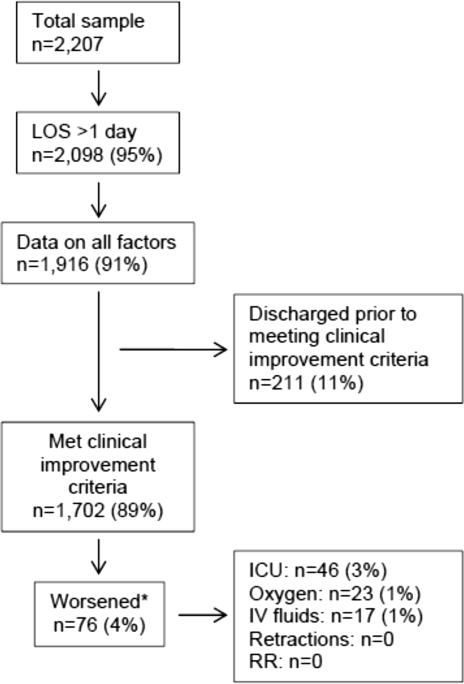
Of the 1702 children who met the clinical improvement criteria, there were 76 children (4%; 95% CI: 3%5%) who worsened (Figure 2). The worsening occurred within a median of 1 day (IQR, 13 days) of clinical improvement. Forty‐six (3%) of the children required transfer to the ICU (1 required intubation, 1 required continuous positive airway pressure, and 4 had apnea), 23 (1%) required oxygen, and 17 (1%) required IV fluids. Eight percent of children met multiple criteria for worsening. A comparison between children who did and did not worsen is shown in Table 1. In general, children who worsened after improvement were younger and born earlier. These children also presented in more severe respiratory distress, had moderate or severe retractions, oxygen saturation <85% at hospitalization, inadequate oral intake, and apnea documented during the hospitalization. Neither viral etiology nor site of care influenced whether the children worsened after improving. However, stratified analysis of children based on initial location of admission (ie, ICU or ward) showed that among the children admitted to the ICU from the emergency department (ED), 89% met the improvement criteria and 19% clinically worsened. In contrast, among children admitted to the ward from the ED, 89% met the improvement criteria, and only 2% clinically worsened. Stratified multivariable models based on the initial location of admission from the ED (ie, ICU or ward) were not possible due to small sample sizes after stratification. None of these children had relapse events requiring rehospitalization within either 24 hours or 7 days of discharge.
| Did Not Worsen, n=1,626 | Worsened, n=76 | P Value | |
|---|---|---|---|
| |||
| Demographic characteristics | |||
| Age <2 months, % | 29 | 57 | <0.001 |
| Month of birth, % | 0.02 | ||
| OctoberMarch | 61 | 75 | |
| AprilSeptember | 39 | 25 | |
| Sex, % | 0.51 | ||
| Male | 59 | 55 | |
| Female | 41 | 45 | |
| Race, % | 0.050 | ||
| White | 63 | 58 | |
| Black | 23 | 34 | |
| Other or missing | 14 | 8 | |
| Hispanic ethnicity, % | 37 | 22 | 0.01 |
| Insurance, % | 0.87 | ||
| Nonprivate | 68 | 67 | |
| Private | 32 | 33 | |
| Medical history | |||
| Gestational age <37 weeks, % | 23 | 39 | 0.002 |
| Birth weight, % | 0.52 | ||
| <5 lbs | 13 | 12 | |
| 5 lbs | 34 | 41 | |
| 7 lbs | 53 | 47 | |
| Mother's age, median (IQR) | 27 (2333) | 27 (2233) | 0.54 |
| Is or was breastfed, % | 61 | 51 | 0.10 |
| Smoked during pregnancy, % | 15 | 20 | 0.22 |
| Exposure to smoke, % | 13 | 20 | 0.11 |
| Family history of asthma, % | 0.89 | ||
| Neither parent | 68 | 64 | |
| Either mother or father | 27 | 30 | |
| Both parents | 4 | 4 | |
| Do not know/missing | 2 | 1 | |
| History of wheezing, % | 23 | 17 | 0.24 |
| History of eczema, % | 16 | 7 | 0.04 |
| History of intubation, % | 9 | 12 | 0.50 |
| Major, relevant, comorbid medical disorder, % | 20 | 24 | 0.46 |
| Current illness | |||
| When difficulty breathing began, preadmission, % | 0.63 | ||
| 1 day | 70 | 75 | |
| <1 day | 28 | 23 | |
| No difficulty preadmission | 2 | 3 | |
| Weight, lbs, median (IQR) | 12.3 (8.817.4) | 9.0 (6.613.2) | 0.001 |
| Temperature, F, median (IQR) | 99.5 (98.6100.6) | 99.4 (98.1100.4) | 0.06 |
| Pulse, beats per minute by age | 0.82 | ||
| Low | 0.3 | 0 | |
| Normal | 48 | 46 | |
| High | 51 | 54 | |
| Respiratory rate, breaths per minute, median (IQR) | 48 (4060) | 48 (3864) | 0.28 |
| Retractions, % | 0.001 | ||
| None | 22 | 25 | |
| Mild | 43 | 24 | |
| Moderate | 26 | 33 | |
| Severe | 4 | 12 | |
| Missing | 5 | 7 | |
| Oxygen saturation by pulse oximetry or ABG, % | 0.001 | ||
| <85 | 4 | 12 | |
| 8587.9 | 3 | 4 | |
| 8889.9 | 5 | 0 | |
| 9093.9 | 18 | 11 | |
| 94 | 72 | 73 | |
| Oral intake, % | <0.001 | ||
| Adequate | 45 | 22 | |
| Inadequate | 42 | 63 | |
| Missing | 13 | 14 | |
| Presence of apnea, % | 7 | 24 | <0.001 |
| RSV‐A, % | 44 | 41 | 0.54 |
| RSV‐B, % | 30 | 25 | 0.36 |
| HRV, % | 24 | 24 | 0.88 |
| Chest x‐ray results during ED/preadmission visit | |||
| Atelectasis | 12 | 13 | 0.77 |
| Infiltrate | 13 | 11 | 0.50 |
| Hyperinflated | 18 | 21 | 0.47 |
| Peribronchial cuffing/thickening | 23 | 17 | 0.32 |
| Normal | 14 | 16 | 0.75 |
| White blood count, median (IQR) | 11.2 (8.714.4) | 11.9 (9.214.4) | 0.60 |
| Platelet count, median (IQR) | 395 (317490) | 430 (299537) | 0.56 |
| Sodium, median (IQR) | 138 (136140) | 137 (135138) | 0.19 |
| Hospital length of stay, median (IQR) | 2 (14) | 4.5 (28) | <0.001 |
| One‐week follow‐up | |||
| Relapse within 24 hours of hospital discharge requiring hospital admission, % | 0.5 | 0 | 0.56 |
| Relapse within 7 days of hospital discharge requiring hospital admission, % | 1 | 0 | 0.35 |
On multivariable analysis (Table 2), independent risk factors for worsening after reaching the clinical improvement criteria were young age, preterm birth, and presenting to care with more severe bronchiolitis represented by severe retractions, inadequate oral intake, or apnea. To further evaluate the improvement criteria in the current analysis, multiple sensitivity analyses were conducted. The frequency of clinical worsening after reaching the improvement criteria was stable when we examined different RA02 criteria in sensitivity analyses: (1) excluding RA02 as a criterion for improvement: 90% met improvement criteria and 4% experienced clinical worsening, (2) changing the average RA02 threshold for clinical improvement to 94%: 62% met improvement criteria and 6% experienced clinical worsening, and (3) changing the average RA02 threshold for clinical improvement to 95%: 47% met improvement criteria and 5% experienced clinical worsening. Furthermore, stratifying by age <2 months and restricting to more stringent definitions of bronchiolitis (ie, age <1 year or age <1 year+no history of wheezing) also did not materially change the results (see Supporting Figure 1 in the online version of this article).
| Odds Ratio | 95% CI | P Value | |
|---|---|---|---|
| |||
| Age <2 months | 3.51 | 2.07‐5.94 | <0.001 |
| Gestational age <37 weeks | 1.94 | 1.13‐3.32 | 0.02 |
| Retractions | |||
| None | 1.30 | 0.80‐3.23 | 0.19 |
| Mild | 1.0 | Reference | |
| Moderate | 1.91 | 0.99‐3.71 | 0.06 |
| Severe | 5.55 | 2.1214.50 | <0.001 |
| Missing | 1.70 | 0.53‐5.42 | 0.37 |
| Oral intake | |||
| Adequate | 1.00 | Reference | |
| Inadequate | 2.54 | 1.39‐4.62 | 0.002 |
| Unknown/missing | 1.88 | 0.79‐4.44 | 0.15 |
| Presence of apnea | 2.87 | 1.45‐5.68 | 0.003 |
We compared the 214 children who were discharged prior to reaching clinical improvement with the 1702 children who reached the clinical improvement criteria. The 214 children were less likely to be age <2 months (22% vs 30%; P=0.02). These 2 groups (214 vs 1702) were similar with respect to severe retractions (2% vs 4%; P=0.13), median respiratory rate (48 vs 48; P=0.42), oxygen saturation <90% (15% vs 11%; P=0.07), inadequate oral intake (50% vs 43%; P=0.13), and rates of relapse events requiring rehospitalization within both 24 hours (0.6% vs 0.6%; P=0.88) and 7 days (1% vs 1%; P=0.90) of discharge.
DISCUSSION
In this large, multicenter, multiyear study of children hospitalized with bronchiolitis, we found that children present to a hospital in a relatively narrow time frame, but their time to recovery in the hospital is highly variable. Nonetheless, 96% of children continued to improve once they had: (1) improving or stable retractions rated as none/mild, (2) a decreasing or stable RR by age, (3) estimated average RAO2 saturation 90% and lowest RAO2 saturation of 88%, and (4) were hydrated. The 4% of children who worsened after clinically improving were more likely to be age <2 months, born <37 weeks, and present with more severe distress (ie, severe retractions, inadequate oral intake, or apnea). Based on the low risk of worsening after clinical improvement, especially among children admitted to the regular ward (2%), we believe these 4 clinical criteria could be used as discharge criteria for this common pediatric illness with a predominantly monophasic clinical course.
Variability in hospital LOS for children with bronchiolitis exists in the United States[3] and internationally.[4, 5] Cheung and colleagues analyzed administrative data from over 75,000 children admitted for bronchiolitis in England between April 2007 and March 2010 and found sixfold variation in LOS between sites. They concluded that this LOS variability was due in part to providers' clinical decision making.[5] Srivastava and colleagues[23] addressed variable clinician decision making in bronchiolitis and 10 other common pediatric conditions by embedding discharge criteria developed by expert consensus into admission order sets. They found that for children with bronchiolitis, the embedded discharge criteria reduced the median LOS from 1.91 to 1.87 days. In contrast to the single‐center data presented by White and colleagues,[24] the prospective, multicenter MARC‐30 data provide a clear understanding of the normal clinical course for children hospitalized with bronchiolitis, determine if children clinically worsen after clinical improvement, and provide data about discharge criteria for children hospitalized with bronchiolitis. Although there is a lack of rigorous published data, the lower tract symptoms of bronchiolitis (eg, cough, retractions) are said to peak on days 5 to 7 of illness and then gradually resolve.[25] In the present study, we found that the time from the onset of difficulty breathing until hospital admission is less variable than the time from the onset of difficulty breathing until either clinical improvement or discharge. Although 75% of children have clinically improved within 7.5 days of difficulty breathing based on the IQR results, the remaining 25% may have a more prolonged recovery in the hospital of up to 3 weeks. Interestingly, prolonged recovery times from bronchiolitis have also been noted in children presenting to the ED[26] and in an outpatient population.[27] It is unclear why 20% to 25% of children at different levels of severity of illness have prolonged recovery from bronchiolitis, but this group of children requires further investigation.
Given the variability of recovery times, clinicians may have difficulty knowing when a child is ready for hospital discharge. One of the main stumbling blocks for discharge readiness in children with bronchiolitis is the interpretation of the oxygen saturation value.[6, 8, 9, 20, 28] However, it should be considered that interpreting the oxygen saturation in a child who is clinically improving in the hospital setting is different than interpreting the oxygen saturation of a child in the ED or the clinic whose clinical course is less certain.[22] In the hospital setting, using the oxygen saturation value in in the AAP guideline,[9] 4% of children clinically worsened after they met the improvement criteria, a clinical pattern observed previously with supplemental oxygen.[28] This unpredictability may explain some of the variation in providers' clinical decision making.[5] The children who worsened, and therefore deserve more cautious discharge planning, were young (<2 months), premature (<37 weeks gestational age), and presented in more severe distress. Those children admitted to the ICU from the ED worsened more commonly than children admitted to the ward (19% vs 2%). Interestingly, the viral etiology of the child's bronchiolitis did not influence whether a child worsened after reaching the improvement criteria. Therefore, although children with RV bronchiolitis have a shorter hospital LOS than children with RSV bronchiolitis,[11] the pattern of recovery did not differ by viral etiology.
In addition to unsafe discharges, clinicians may be concerned about the possibility of readmissions. Although somewhat controversial, hospital readmission is being used as a quality of care metric.[29, 30, 31] One response to minimize readmissions would be for clinicians to observe children for longer than clinically indicated.[32] However, shorter LOS is not necessarily associated with increased readmission rates.[33] Given that the geometric mean of hospital charges per child with bronchiolitis increased from $6380 in 2000 to $8530 in 2009,[34] the potential for safely reducing hospital LOS by using the discharge criteria proposed in the current study instead of other criteria[8] may net substantial cost savings. Furthermore, reducing LOS would decrease the time children expose others to these respiratory viruses and possibly reduce medical errors.[35]
Our study has some potential limitations. Because the study participants were all hospitalized, these data do not inform admission or discharge decisions from either the ED or the clinic; but other data address those clinical scenarios.[22] Also, the 16 sites that participated in this study were large, urban teaching hospitals. Consequently, these results are not necessarily generalizable to smaller community hospitals. Although numerous data points were required to enter the analytic cohort, only 9% of the sample was excluded for missing data. There were 214 children who did not meet our improvement criteria by the time of discharge. Although the inability to include these children in the analysis may be seen as a limitation, this practice variability underscores the need for more data about discharging hospitalized children with bronchiolitis. Last, site teams reviewed medical records daily. More frequent recording of the clinical course would have yielded more granular data, but the current methodology replicates how data are generally presented during patient care rounds, when decisions about suitability for discharge are often considered.
CONCLUSION
We documented in this large multicenter study that most children hospitalized with bronchiolitis had a wide range of time to recovery, but the vast majority continued to improve once they reached the identified clinical criteria that predict a safe discharge to home. The children who worsened after clinical improvement were more likely to be younger, premature infants presenting in more severe distress. Although additional prospective validation of these hospital discharge criteria is warranted, these data may help clinicians make more evidence‐based discharge decisions for a common pediatric illness with high practice variation, both in the United States[3] and in other countries.[4, 5]
Acknowledgements
Collaborators in the MARC‐30 Study: Besh Barcega, MD, Loma Linda University Children's Hospital, Loma Linda, CA; John Cheng, MD, Children's Healthcare of Atlanta at Egleston, Atlanta, GA; Dorothy Damore, MD, New York Presbyterian Hospital‐Cornell, New York, NY; Carlos Delgado, MD, Children's Healthcare of Atlanta at Egleston, Atlanta, GA; Haitham Haddad, MD, Rainbow Babies & Children's Hospital, Cleveland, OH; Paul Hain, MD, Monroe Carell Jr. Children's Hospital at Vanderbilt, Nashville, TN; Frank LoVecchio, DO, Maricopa Medical Center, Phoenix, AZ; Charles Macias, MD MPH, Texas Children's Hospital, Houston, TX; Jonathan Mansbach, MD, MPH, Boston Children's Hospital, Boston, MA; Eugene Mowad, MD, Akron Children's Hospital, Akron, OH; Brian Pate, MD, Children's Mercy Hospital, Kansas City, MO; Mark Riederer, MD, Monroe Carell Jr. Children's Hospital at Vanderbilt, Nashville, TN; M. Jason Sanders, MD, Children's Memorial Hermann Hospital, Houston, TX; Alan R. Schroeder, MD, Santa Clara Valley Medical Center, San Jose, CA; Nikhil Shah, MD, New York Presbyterian Hospital‐Cornell, New York, NY; Michelle Stevenson, MD, MS, Kosair Children's Hospital, Louisville, KY; Erin Stucky Fisher, MD, Rady Children's Hospital, San Diego, CA; Stephen Teach, MD, MPH, Children's National Medical Center, Washington, DC; Lisa Zaoutis, MD, Children's Hospital of Philadelphia, Philadelphia, PA.
Disclosures: This study was supported by grants U01 AI‐67693 and K23 AI‐77801 from the National Institutes of Health (Bethesda, MD). The content of this manuscript is solely the responsibility of the authors and does not necessarily represent the official views of the National Institute of Allergy and Infectious Diseases or the National Institutes of Health. Drs. Mansbach and Piedra have provided consultation to Regeneron Pharmaceuticals. Otherwise, no authors report any potential conflicts of interest, including relevant financial interests, activities, relationships, and affiliations.
- , , , , . Infectious disease hospitalizations among infants in the United States. Pediatrics. 2008;121(2):244–252.
- . “A hospital is no place to be sick” Samuel Goldwyn (1882–1974). Arch Dis Child. 2009;94(8):565–566.
- , , , , , Variation in inpatient diagnostic testing and management of bronchiolitis. Pediatrics. 2005;115(4):878–884.
- , , , International variation in the management of infants hospitalized with respiratory syncytial virus. International RSV Study Group. Eur J Pediatr. 1998;157(3):215–220.
- , , , , . Population variation in admission rates and duration of inpatient stay for bronchiolitis in England. Arch Dis Child. 2013;98(1):57–59.
- , , , . Impact of pulse oximetry and oxygen therapy on length of stay in bronchiolitis hospitalizations. Arch Pediatr Adolesc Med. 2004;158(6):527–530.
- , , . Pulse oximetry in pediatric practice. Pediatrics. 2011;128(4):740–752.
- Scottish Intercollegiate Guidelines Network. Bronchiolitis in children (SIGN 91). In: NHS Quality Improvement Scotland. Edinburgh, Scotland: Scottish Intercollegiate Guidelines Network; 2006.
- , , , et al. Diagnosis and management of bronchiolitis. Pediatrics. 2006;118(4):1774–1793.
- , , , et al. Prospective multicenter study of children with bronchiolitis requiring mechanical ventilation. Pediatrics. 2012;130(3):e492–e500.
- , , , et al. Prospective multicenter study of viral etiology and hospital length of stay in children with severe bronchiolitis. Arch Pediatr Adolesc Med. 2012;166(8):700–706.
- , , , et al. Apnea in children hospitalized with bronchiolitis. Pediatrics. 2013;132(5):e1194–e1201.
- . Evaluation of the cardiovascular system: history and physical evaluation. In: Kliegman RM, Stanton BF, St. Geme JW III, Schor NF, Behrman RF, eds. Nelson Textbook of Pediatrics. Philadelphia, PA: Elsevier Saunders; 2011:1529–1536.
- , , , et al. Apnea in children hospitalized with bronchiolitis. Pediatrics. 2013;132(5):e1194–e1201.
- , , , et al. Respiratory viral infections in patients with chronic, obstructive pulmonary disease. J Infect. 2005;50(4):322–330.
- , , , . Evaluation of real‐time PCR for diagnosis of Bordetella pertussis infection. BMC Infect Dis. 2006;6:62.
- , , , , . Evaluation of three real‐time PCR assays for detection of Mycoplasma pneumoniae in an outbreak investigation. J Clin Microbiol. 2008;46(9):3116–3118.
- , , , et al. Normal ranges of heart rate and respiratory rate in children from birth to 18 years of age: a systematic review of observational studies. Lancet. 2011;377(9770):1011–1018.
- , , , , , . Development of heart and respiratory rate percentile curves for hospitalized children. Pediatrics. 2013;131(4):e1150–e1157.
- , . Effect of oxygen supplementation on length of stay for infants hospitalized with acute viral bronchiolitis. Pediatrics. 2008;121(3):470–475.
- , , , et al. Longitudinal assessment of hemoglobin oxygen saturation in healthy infants during the first 6 months of age. Collaborative Home Infant Monitoring Evaluation (CHIME) Study Group. J Pediatr. 1999;135(5):580–586.
- , , , et al. Prospective multicenter study of bronchiolitis: predicting safe discharges from the emergency department. Pediatrics. 2008;121(4):680–688.
- , , , et al. Delays in discharge in a tertiary care pediatric hospital. J Hosp Med. 2009;4(8):481–485.
- , , , et al. Using quality improvement to optimise paediatric discharge efficiency. BMJ Qual Saf. 2014;23(5):428–436.
- , . Bronchiolitis in infants and children: treatment; outcome; and prevention. In: Torchia M, ed. UpToDate. Alphen aan den Rijn, the Netherlands; Wolters Kluwer Health; 2013.
- , . Duration of illness in infants with bronchiolitis evaluated in the emergency department. Pediatrics. 2010;126(2):285–290.
- , , . Duration of illness in ambulatory children diagnosed with bronchiolitis. Arch Pediatr Adolesc Med. 2000;154(10):997–1000.
- , , , et al. A clinical pathway for bronchiolitis is effective in reducing readmission rates. J Pediatr. 2005;147(5):622–626.
- , , , et al. Measuring hospital quality using pediatric readmission and revisit rates. Pediatrics. 2013;132(3):429–436.
- , , , et al. Pediatric readmission prevalence and variability across hospitals. JAMA. 2013;309(4):372–380.
- , , , , , . Preventability of early readmissions at a children's hospital. Pediatrics. 2013;131(1):e171–e181.
- , . Hospital readmission: quality indicator or statistical inevitability? Pediatrics. 2013;132(3):569–570.
- , , , et al. Children's hospitals with shorter lengths of stay do not have higher readmission rates. J Pediatr. 2013;163(4):1034–1038.e1.
- , , , , . Trends in bronchiolitis hospitalizations in the United States, 2000–2009. Pediatrics. 2013;132(1):28–36.
- , , , . Preventable adverse events in infants hospitalized with bronchiolitis. Pediatrics. 2005;116(3):603–608.
Although bronchiolitis is the leading cause of hospitalization for US infants,[1] there is a lack of basic prospective data about the expected inpatient clinical course and ongoing uncertainty about when a hospitalized child is ready for discharge to home.[2] This lack of data about children's readiness for discharge may result in variable hospital length‐of‐stay (LOS).[3, 4, 5]
One specific source of variability in discharge readiness and LOS variability may be the lack of consensus about safe threshold oxygen saturation values for discharge in children hospitalized with bronchiolitis.[6, 7] In 2006, the Scottish Intercollegiate Guidelines Network recommended a discharge room air oxygen (RAO2) saturation threshold of 95%.[8] The same year, the American Academy of Pediatrics (AAP) bronchiolitis clinical practice guideline stated that oxygen is not needed for children with RAO2 saturations 90% who are feeding well and have minimal respiratory distress.[9] There is a need for prospective studies to help clinicians make evidenced‐based discharge decisions for this common condition.
We performed a prospective, multicenter, multiyear study[10, 11, 12] to examine the typical inpatient clinical course of and to develop hospital discharge guidelines for children age <2 years hospitalized with bronchiolitis. We hypothesized that children would not worsen clinically and would be safe to discharge home once their respiratory status improved and they were able to remain hydrated.
METHODS
Study Design and Population
We conducted a prospective, multicenter cohort study for 3 consecutive years during the 2007 to 2010 winter seasons, as part of the Multicenter Airway Research Collaboration (MARC), a program of the Emergency Medicine Network (
All patients were treated at the discretion of the treating physician. Inclusion criteria were an attending physician's diagnosis of bronchiolitis, age <2 years, and the ability of the parent/guardian to give informed consent. The exclusion criteria were previous enrollment and transfer to a participating hospital >48 hours after the original admission time. Therefore, children with comorbid conditions were included in this study. All consent and data forms were translated into Spanish. The institutional review board at each of the 16 participating hospitals approved the study.
Of the 2207 enrolled children, we excluded 109 (5%) children with a hospital LOS <1 day due to inadequate time to capture the required data for the present analysis. Among the 2098 remaining children, 1916 (91%) had daily inpatient data on all factors used to define clinical improvement and clinical worsening. Thus, the analytic cohort was comprised of 1916 children hospitalized for bronchiolitis.
Data Collection
Investigators conducted detailed structured interviews. Chart reviews were conducted to obtain preadmission and daily hospital clinical data including respiratory rates, daily respiratory rate trends, degree of retractions, oxygen saturation, daily oxygen saturation trends, medical management, and disposition. These data were manually reviewed, and site investigators were queried about missing data and discrepancies. A follow‐up telephone interview was conducted with families 1 week after discharge to examine relapse events at both 24 hours and 7 days.
We used the question: How long ago did the following symptoms [eg, difficulty breathing] begin [for the] current illness? to estimate the onset of the current illness. Pulse was categorized as low, normal, or high based on age‐related heart rate values.[13] Presence of apnea was recorded daily by site investigators.[14]
Nasopharyngeal Aspirate Collection and Virology Testing
As described previously, site teams used a standardized protocol to collect nasopharyngeal aspirates,[11] which were tested for respiratory syncytial virus (RSV) types A and B; rhinovirus (RV); parainfluenza virus types 1, 2, and 3; influenza virus types A and B; 2009 novel H1N1; human metapneumovirus; coronaviruses NL‐63, HKU1, OC43, and 229E; enterovirus, and adenovirus using polymerase chain reaction.[11, 15, 16, 17]
Defining Clinical Improvement and Worsening
Clinical improvement criteria were based on the 2006 AAP guidelines.[9] For respiratory rate and oxygen saturation, clinicians estimated average daily respiratory rate and oxygen saturation based on the recorded readings from the previous 24 hours. This estimation reflects the process clinicians use when rounding on their hospitalized patients, and thus may be more similar to standard clinical practice than a calculated mean. The respiratory rate criteria are adjusted for age.[18, 19] For daily estimated average oxygen saturation we used the AAP criteria of RAO2 saturation of 90%. Considering that oxygen saturation is the main determinant of LOS,[20] healthy infants age <6 months may have transient oxygen saturations of around 80%,[21] and that errors in estimation may occur, we included a lowest RAO2 of 88% in our improvement criteria. By combining the dichotomized estimated oxygen saturation (90% or not) with the lower limit of 88%, there was little room for erroneous conclusions. A child was considered clinically improved on the earliest date he/she met all of the following criteria: (1) none or mild retractions and improved or stable retractions compared with the previous inpatient day; (2) daily estimated average respiratory rate (RR) <60 breaths per minute for age <6 months, <55 breaths/minute for age 6 to 11 months, and <45 breaths/minute for age 12 months with a decreasing or stable trend over the course of the current day; (3) daily estimated average RAO2 saturation 90%, lowest RAO2 saturation 88%[21]; and (4) not receiving intravenous (IV) fluids or for children receiving IV fluids a clinician report of the child maintaining oral hydration. Children who reached the clinical improvement criteria were considered clinically worse if they required intensive care or had the inverse of 1 of the improvement criteria: moderate/severe retractions that were worse compared with the previous inpatient day, daily average RR 60 with an increasing trend over the current day, need for oxygen, or need for IV fluids.
Statistical Analyses
All analyses were performed using Stata 12.0 (StataCorp, College Station, TX). Data are presented as proportions with 95% confidence intervals (95% CIs), means with standard deviations, and medians with interquartile ranges (IQR). To examine potential factors associated with clinical worsening after reaching clinical improvement, we used 2, Fisher exact, Student t test, and Kruskall‐Wallis tests, as appropriate.
Adjusted analyses used generalized linear mixed models with a logit link to identify independent risk factors for worsening after reaching clinical improvement. Fixed effects for patient‐level factors and a random site effect were used. Factors were tested for inclusion in the multivariable model if they were found to be associated with worsening in unadjusted analyses (P<0.20) or were considered clinically important. Results are reported as odds ratios with 95% CIs.
We performed several sensitivity analyses to evaluate these improvement criteria: (1) we excluded the lowest RAO2 saturation requirement of 88%, (2) we examined a 94% daily estimated average RAO2 saturation threshold,[22] (3) we examined a 95% daily estimated average RAO2 saturation threshold,[8] and (4) we examined children age <12 months with no history of wheeze.
RESULTS
There were 1916 children hospitalized with bronchiolitis with data on all factors used to define clinical improvement and clinical worsening. The median number of days from the beginning of difficulty breathing until admission was 2 days (IQR, 15.5 days; range, 18 days) and from the beginning of difficulty breathing until clinical improvement was 4 days (IQR, 37.5 days; range, 133 days) (Figure 1). The variance for days to admission was significantly less than the variance for days to clinical improvement (P<0.001).

In this observational study, clinicians discharged 214 (11%) of the 1916 children before meeting the definition of clinical improvement. Thus, 1702 (89%; 95% CI: 87%‐90%) children reached the clinical improvement criteria, had a LOS >1 day, and had data on all factors (Figure 2).

Of the 1702 children who met the clinical improvement criteria, there were 76 children (4%; 95% CI: 3%5%) who worsened (Figure 2). The worsening occurred within a median of 1 day (IQR, 13 days) of clinical improvement. Forty‐six (3%) of the children required transfer to the ICU (1 required intubation, 1 required continuous positive airway pressure, and 4 had apnea), 23 (1%) required oxygen, and 17 (1%) required IV fluids. Eight percent of children met multiple criteria for worsening. A comparison between children who did and did not worsen is shown in Table 1. In general, children who worsened after improvement were younger and born earlier. These children also presented in more severe respiratory distress, had moderate or severe retractions, oxygen saturation <85% at hospitalization, inadequate oral intake, and apnea documented during the hospitalization. Neither viral etiology nor site of care influenced whether the children worsened after improving. However, stratified analysis of children based on initial location of admission (ie, ICU or ward) showed that among the children admitted to the ICU from the emergency department (ED), 89% met the improvement criteria and 19% clinically worsened. In contrast, among children admitted to the ward from the ED, 89% met the improvement criteria, and only 2% clinically worsened. Stratified multivariable models based on the initial location of admission from the ED (ie, ICU or ward) were not possible due to small sample sizes after stratification. None of these children had relapse events requiring rehospitalization within either 24 hours or 7 days of discharge.
| Did Not Worsen, n=1,626 | Worsened, n=76 | P Value | |
|---|---|---|---|
| |||
| Demographic characteristics | |||
| Age <2 months, % | 29 | 57 | <0.001 |
| Month of birth, % | 0.02 | ||
| OctoberMarch | 61 | 75 | |
| AprilSeptember | 39 | 25 | |
| Sex, % | 0.51 | ||
| Male | 59 | 55 | |
| Female | 41 | 45 | |
| Race, % | 0.050 | ||
| White | 63 | 58 | |
| Black | 23 | 34 | |
| Other or missing | 14 | 8 | |
| Hispanic ethnicity, % | 37 | 22 | 0.01 |
| Insurance, % | 0.87 | ||
| Nonprivate | 68 | 67 | |
| Private | 32 | 33 | |
| Medical history | |||
| Gestational age <37 weeks, % | 23 | 39 | 0.002 |
| Birth weight, % | 0.52 | ||
| <5 lbs | 13 | 12 | |
| 5 lbs | 34 | 41 | |
| 7 lbs | 53 | 47 | |
| Mother's age, median (IQR) | 27 (2333) | 27 (2233) | 0.54 |
| Is or was breastfed, % | 61 | 51 | 0.10 |
| Smoked during pregnancy, % | 15 | 20 | 0.22 |
| Exposure to smoke, % | 13 | 20 | 0.11 |
| Family history of asthma, % | 0.89 | ||
| Neither parent | 68 | 64 | |
| Either mother or father | 27 | 30 | |
| Both parents | 4 | 4 | |
| Do not know/missing | 2 | 1 | |
| History of wheezing, % | 23 | 17 | 0.24 |
| History of eczema, % | 16 | 7 | 0.04 |
| History of intubation, % | 9 | 12 | 0.50 |
| Major, relevant, comorbid medical disorder, % | 20 | 24 | 0.46 |
| Current illness | |||
| When difficulty breathing began, preadmission, % | 0.63 | ||
| 1 day | 70 | 75 | |
| <1 day | 28 | 23 | |
| No difficulty preadmission | 2 | 3 | |
| Weight, lbs, median (IQR) | 12.3 (8.817.4) | 9.0 (6.613.2) | 0.001 |
| Temperature, F, median (IQR) | 99.5 (98.6100.6) | 99.4 (98.1100.4) | 0.06 |
| Pulse, beats per minute by age | 0.82 | ||
| Low | 0.3 | 0 | |
| Normal | 48 | 46 | |
| High | 51 | 54 | |
| Respiratory rate, breaths per minute, median (IQR) | 48 (4060) | 48 (3864) | 0.28 |
| Retractions, % | 0.001 | ||
| None | 22 | 25 | |
| Mild | 43 | 24 | |
| Moderate | 26 | 33 | |
| Severe | 4 | 12 | |
| Missing | 5 | 7 | |
| Oxygen saturation by pulse oximetry or ABG, % | 0.001 | ||
| <85 | 4 | 12 | |
| 8587.9 | 3 | 4 | |
| 8889.9 | 5 | 0 | |
| 9093.9 | 18 | 11 | |
| 94 | 72 | 73 | |
| Oral intake, % | <0.001 | ||
| Adequate | 45 | 22 | |
| Inadequate | 42 | 63 | |
| Missing | 13 | 14 | |
| Presence of apnea, % | 7 | 24 | <0.001 |
| RSV‐A, % | 44 | 41 | 0.54 |
| RSV‐B, % | 30 | 25 | 0.36 |
| HRV, % | 24 | 24 | 0.88 |
| Chest x‐ray results during ED/preadmission visit | |||
| Atelectasis | 12 | 13 | 0.77 |
| Infiltrate | 13 | 11 | 0.50 |
| Hyperinflated | 18 | 21 | 0.47 |
| Peribronchial cuffing/thickening | 23 | 17 | 0.32 |
| Normal | 14 | 16 | 0.75 |
| White blood count, median (IQR) | 11.2 (8.714.4) | 11.9 (9.214.4) | 0.60 |
| Platelet count, median (IQR) | 395 (317490) | 430 (299537) | 0.56 |
| Sodium, median (IQR) | 138 (136140) | 137 (135138) | 0.19 |
| Hospital length of stay, median (IQR) | 2 (14) | 4.5 (28) | <0.001 |
| One‐week follow‐up | |||
| Relapse within 24 hours of hospital discharge requiring hospital admission, % | 0.5 | 0 | 0.56 |
| Relapse within 7 days of hospital discharge requiring hospital admission, % | 1 | 0 | 0.35 |
On multivariable analysis (Table 2), independent risk factors for worsening after reaching the clinical improvement criteria were young age, preterm birth, and presenting to care with more severe bronchiolitis represented by severe retractions, inadequate oral intake, or apnea. To further evaluate the improvement criteria in the current analysis, multiple sensitivity analyses were conducted. The frequency of clinical worsening after reaching the improvement criteria was stable when we examined different RA02 criteria in sensitivity analyses: (1) excluding RA02 as a criterion for improvement: 90% met improvement criteria and 4% experienced clinical worsening, (2) changing the average RA02 threshold for clinical improvement to 94%: 62% met improvement criteria and 6% experienced clinical worsening, and (3) changing the average RA02 threshold for clinical improvement to 95%: 47% met improvement criteria and 5% experienced clinical worsening. Furthermore, stratifying by age <2 months and restricting to more stringent definitions of bronchiolitis (ie, age <1 year or age <1 year+no history of wheezing) also did not materially change the results (see Supporting Figure 1 in the online version of this article).
| Odds Ratio | 95% CI | P Value | |
|---|---|---|---|
| |||
| Age <2 months | 3.51 | 2.07‐5.94 | <0.001 |
| Gestational age <37 weeks | 1.94 | 1.13‐3.32 | 0.02 |
| Retractions | |||
| None | 1.30 | 0.80‐3.23 | 0.19 |
| Mild | 1.0 | Reference | |
| Moderate | 1.91 | 0.99‐3.71 | 0.06 |
| Severe | 5.55 | 2.1214.50 | <0.001 |
| Missing | 1.70 | 0.53‐5.42 | 0.37 |
| Oral intake | |||
| Adequate | 1.00 | Reference | |
| Inadequate | 2.54 | 1.39‐4.62 | 0.002 |
| Unknown/missing | 1.88 | 0.79‐4.44 | 0.15 |
| Presence of apnea | 2.87 | 1.45‐5.68 | 0.003 |
We compared the 214 children who were discharged prior to reaching clinical improvement with the 1702 children who reached the clinical improvement criteria. The 214 children were less likely to be age <2 months (22% vs 30%; P=0.02). These 2 groups (214 vs 1702) were similar with respect to severe retractions (2% vs 4%; P=0.13), median respiratory rate (48 vs 48; P=0.42), oxygen saturation <90% (15% vs 11%; P=0.07), inadequate oral intake (50% vs 43%; P=0.13), and rates of relapse events requiring rehospitalization within both 24 hours (0.6% vs 0.6%; P=0.88) and 7 days (1% vs 1%; P=0.90) of discharge.
DISCUSSION
In this large, multicenter, multiyear study of children hospitalized with bronchiolitis, we found that children present to a hospital in a relatively narrow time frame, but their time to recovery in the hospital is highly variable. Nonetheless, 96% of children continued to improve once they had: (1) improving or stable retractions rated as none/mild, (2) a decreasing or stable RR by age, (3) estimated average RAO2 saturation 90% and lowest RAO2 saturation of 88%, and (4) were hydrated. The 4% of children who worsened after clinically improving were more likely to be age <2 months, born <37 weeks, and present with more severe distress (ie, severe retractions, inadequate oral intake, or apnea). Based on the low risk of worsening after clinical improvement, especially among children admitted to the regular ward (2%), we believe these 4 clinical criteria could be used as discharge criteria for this common pediatric illness with a predominantly monophasic clinical course.
Variability in hospital LOS for children with bronchiolitis exists in the United States[3] and internationally.[4, 5] Cheung and colleagues analyzed administrative data from over 75,000 children admitted for bronchiolitis in England between April 2007 and March 2010 and found sixfold variation in LOS between sites. They concluded that this LOS variability was due in part to providers' clinical decision making.[5] Srivastava and colleagues[23] addressed variable clinician decision making in bronchiolitis and 10 other common pediatric conditions by embedding discharge criteria developed by expert consensus into admission order sets. They found that for children with bronchiolitis, the embedded discharge criteria reduced the median LOS from 1.91 to 1.87 days. In contrast to the single‐center data presented by White and colleagues,[24] the prospective, multicenter MARC‐30 data provide a clear understanding of the normal clinical course for children hospitalized with bronchiolitis, determine if children clinically worsen after clinical improvement, and provide data about discharge criteria for children hospitalized with bronchiolitis. Although there is a lack of rigorous published data, the lower tract symptoms of bronchiolitis (eg, cough, retractions) are said to peak on days 5 to 7 of illness and then gradually resolve.[25] In the present study, we found that the time from the onset of difficulty breathing until hospital admission is less variable than the time from the onset of difficulty breathing until either clinical improvement or discharge. Although 75% of children have clinically improved within 7.5 days of difficulty breathing based on the IQR results, the remaining 25% may have a more prolonged recovery in the hospital of up to 3 weeks. Interestingly, prolonged recovery times from bronchiolitis have also been noted in children presenting to the ED[26] and in an outpatient population.[27] It is unclear why 20% to 25% of children at different levels of severity of illness have prolonged recovery from bronchiolitis, but this group of children requires further investigation.
Given the variability of recovery times, clinicians may have difficulty knowing when a child is ready for hospital discharge. One of the main stumbling blocks for discharge readiness in children with bronchiolitis is the interpretation of the oxygen saturation value.[6, 8, 9, 20, 28] However, it should be considered that interpreting the oxygen saturation in a child who is clinically improving in the hospital setting is different than interpreting the oxygen saturation of a child in the ED or the clinic whose clinical course is less certain.[22] In the hospital setting, using the oxygen saturation value in in the AAP guideline,[9] 4% of children clinically worsened after they met the improvement criteria, a clinical pattern observed previously with supplemental oxygen.[28] This unpredictability may explain some of the variation in providers' clinical decision making.[5] The children who worsened, and therefore deserve more cautious discharge planning, were young (<2 months), premature (<37 weeks gestational age), and presented in more severe distress. Those children admitted to the ICU from the ED worsened more commonly than children admitted to the ward (19% vs 2%). Interestingly, the viral etiology of the child's bronchiolitis did not influence whether a child worsened after reaching the improvement criteria. Therefore, although children with RV bronchiolitis have a shorter hospital LOS than children with RSV bronchiolitis,[11] the pattern of recovery did not differ by viral etiology.
In addition to unsafe discharges, clinicians may be concerned about the possibility of readmissions. Although somewhat controversial, hospital readmission is being used as a quality of care metric.[29, 30, 31] One response to minimize readmissions would be for clinicians to observe children for longer than clinically indicated.[32] However, shorter LOS is not necessarily associated with increased readmission rates.[33] Given that the geometric mean of hospital charges per child with bronchiolitis increased from $6380 in 2000 to $8530 in 2009,[34] the potential for safely reducing hospital LOS by using the discharge criteria proposed in the current study instead of other criteria[8] may net substantial cost savings. Furthermore, reducing LOS would decrease the time children expose others to these respiratory viruses and possibly reduce medical errors.[35]
Our study has some potential limitations. Because the study participants were all hospitalized, these data do not inform admission or discharge decisions from either the ED or the clinic; but other data address those clinical scenarios.[22] Also, the 16 sites that participated in this study were large, urban teaching hospitals. Consequently, these results are not necessarily generalizable to smaller community hospitals. Although numerous data points were required to enter the analytic cohort, only 9% of the sample was excluded for missing data. There were 214 children who did not meet our improvement criteria by the time of discharge. Although the inability to include these children in the analysis may be seen as a limitation, this practice variability underscores the need for more data about discharging hospitalized children with bronchiolitis. Last, site teams reviewed medical records daily. More frequent recording of the clinical course would have yielded more granular data, but the current methodology replicates how data are generally presented during patient care rounds, when decisions about suitability for discharge are often considered.
CONCLUSION
We documented in this large multicenter study that most children hospitalized with bronchiolitis had a wide range of time to recovery, but the vast majority continued to improve once they reached the identified clinical criteria that predict a safe discharge to home. The children who worsened after clinical improvement were more likely to be younger, premature infants presenting in more severe distress. Although additional prospective validation of these hospital discharge criteria is warranted, these data may help clinicians make more evidence‐based discharge decisions for a common pediatric illness with high practice variation, both in the United States[3] and in other countries.[4, 5]
Acknowledgements
Collaborators in the MARC‐30 Study: Besh Barcega, MD, Loma Linda University Children's Hospital, Loma Linda, CA; John Cheng, MD, Children's Healthcare of Atlanta at Egleston, Atlanta, GA; Dorothy Damore, MD, New York Presbyterian Hospital‐Cornell, New York, NY; Carlos Delgado, MD, Children's Healthcare of Atlanta at Egleston, Atlanta, GA; Haitham Haddad, MD, Rainbow Babies & Children's Hospital, Cleveland, OH; Paul Hain, MD, Monroe Carell Jr. Children's Hospital at Vanderbilt, Nashville, TN; Frank LoVecchio, DO, Maricopa Medical Center, Phoenix, AZ; Charles Macias, MD MPH, Texas Children's Hospital, Houston, TX; Jonathan Mansbach, MD, MPH, Boston Children's Hospital, Boston, MA; Eugene Mowad, MD, Akron Children's Hospital, Akron, OH; Brian Pate, MD, Children's Mercy Hospital, Kansas City, MO; Mark Riederer, MD, Monroe Carell Jr. Children's Hospital at Vanderbilt, Nashville, TN; M. Jason Sanders, MD, Children's Memorial Hermann Hospital, Houston, TX; Alan R. Schroeder, MD, Santa Clara Valley Medical Center, San Jose, CA; Nikhil Shah, MD, New York Presbyterian Hospital‐Cornell, New York, NY; Michelle Stevenson, MD, MS, Kosair Children's Hospital, Louisville, KY; Erin Stucky Fisher, MD, Rady Children's Hospital, San Diego, CA; Stephen Teach, MD, MPH, Children's National Medical Center, Washington, DC; Lisa Zaoutis, MD, Children's Hospital of Philadelphia, Philadelphia, PA.
Disclosures: This study was supported by grants U01 AI‐67693 and K23 AI‐77801 from the National Institutes of Health (Bethesda, MD). The content of this manuscript is solely the responsibility of the authors and does not necessarily represent the official views of the National Institute of Allergy and Infectious Diseases or the National Institutes of Health. Drs. Mansbach and Piedra have provided consultation to Regeneron Pharmaceuticals. Otherwise, no authors report any potential conflicts of interest, including relevant financial interests, activities, relationships, and affiliations.
Although bronchiolitis is the leading cause of hospitalization for US infants,[1] there is a lack of basic prospective data about the expected inpatient clinical course and ongoing uncertainty about when a hospitalized child is ready for discharge to home.[2] This lack of data about children's readiness for discharge may result in variable hospital length‐of‐stay (LOS).[3, 4, 5]
One specific source of variability in discharge readiness and LOS variability may be the lack of consensus about safe threshold oxygen saturation values for discharge in children hospitalized with bronchiolitis.[6, 7] In 2006, the Scottish Intercollegiate Guidelines Network recommended a discharge room air oxygen (RAO2) saturation threshold of 95%.[8] The same year, the American Academy of Pediatrics (AAP) bronchiolitis clinical practice guideline stated that oxygen is not needed for children with RAO2 saturations 90% who are feeding well and have minimal respiratory distress.[9] There is a need for prospective studies to help clinicians make evidenced‐based discharge decisions for this common condition.
We performed a prospective, multicenter, multiyear study[10, 11, 12] to examine the typical inpatient clinical course of and to develop hospital discharge guidelines for children age <2 years hospitalized with bronchiolitis. We hypothesized that children would not worsen clinically and would be safe to discharge home once their respiratory status improved and they were able to remain hydrated.
METHODS
Study Design and Population
We conducted a prospective, multicenter cohort study for 3 consecutive years during the 2007 to 2010 winter seasons, as part of the Multicenter Airway Research Collaboration (MARC), a program of the Emergency Medicine Network (
All patients were treated at the discretion of the treating physician. Inclusion criteria were an attending physician's diagnosis of bronchiolitis, age <2 years, and the ability of the parent/guardian to give informed consent. The exclusion criteria were previous enrollment and transfer to a participating hospital >48 hours after the original admission time. Therefore, children with comorbid conditions were included in this study. All consent and data forms were translated into Spanish. The institutional review board at each of the 16 participating hospitals approved the study.
Of the 2207 enrolled children, we excluded 109 (5%) children with a hospital LOS <1 day due to inadequate time to capture the required data for the present analysis. Among the 2098 remaining children, 1916 (91%) had daily inpatient data on all factors used to define clinical improvement and clinical worsening. Thus, the analytic cohort was comprised of 1916 children hospitalized for bronchiolitis.
Data Collection
Investigators conducted detailed structured interviews. Chart reviews were conducted to obtain preadmission and daily hospital clinical data including respiratory rates, daily respiratory rate trends, degree of retractions, oxygen saturation, daily oxygen saturation trends, medical management, and disposition. These data were manually reviewed, and site investigators were queried about missing data and discrepancies. A follow‐up telephone interview was conducted with families 1 week after discharge to examine relapse events at both 24 hours and 7 days.
We used the question: How long ago did the following symptoms [eg, difficulty breathing] begin [for the] current illness? to estimate the onset of the current illness. Pulse was categorized as low, normal, or high based on age‐related heart rate values.[13] Presence of apnea was recorded daily by site investigators.[14]
Nasopharyngeal Aspirate Collection and Virology Testing
As described previously, site teams used a standardized protocol to collect nasopharyngeal aspirates,[11] which were tested for respiratory syncytial virus (RSV) types A and B; rhinovirus (RV); parainfluenza virus types 1, 2, and 3; influenza virus types A and B; 2009 novel H1N1; human metapneumovirus; coronaviruses NL‐63, HKU1, OC43, and 229E; enterovirus, and adenovirus using polymerase chain reaction.[11, 15, 16, 17]
Defining Clinical Improvement and Worsening
Clinical improvement criteria were based on the 2006 AAP guidelines.[9] For respiratory rate and oxygen saturation, clinicians estimated average daily respiratory rate and oxygen saturation based on the recorded readings from the previous 24 hours. This estimation reflects the process clinicians use when rounding on their hospitalized patients, and thus may be more similar to standard clinical practice than a calculated mean. The respiratory rate criteria are adjusted for age.[18, 19] For daily estimated average oxygen saturation we used the AAP criteria of RAO2 saturation of 90%. Considering that oxygen saturation is the main determinant of LOS,[20] healthy infants age <6 months may have transient oxygen saturations of around 80%,[21] and that errors in estimation may occur, we included a lowest RAO2 of 88% in our improvement criteria. By combining the dichotomized estimated oxygen saturation (90% or not) with the lower limit of 88%, there was little room for erroneous conclusions. A child was considered clinically improved on the earliest date he/she met all of the following criteria: (1) none or mild retractions and improved or stable retractions compared with the previous inpatient day; (2) daily estimated average respiratory rate (RR) <60 breaths per minute for age <6 months, <55 breaths/minute for age 6 to 11 months, and <45 breaths/minute for age 12 months with a decreasing or stable trend over the course of the current day; (3) daily estimated average RAO2 saturation 90%, lowest RAO2 saturation 88%[21]; and (4) not receiving intravenous (IV) fluids or for children receiving IV fluids a clinician report of the child maintaining oral hydration. Children who reached the clinical improvement criteria were considered clinically worse if they required intensive care or had the inverse of 1 of the improvement criteria: moderate/severe retractions that were worse compared with the previous inpatient day, daily average RR 60 with an increasing trend over the current day, need for oxygen, or need for IV fluids.
Statistical Analyses
All analyses were performed using Stata 12.0 (StataCorp, College Station, TX). Data are presented as proportions with 95% confidence intervals (95% CIs), means with standard deviations, and medians with interquartile ranges (IQR). To examine potential factors associated with clinical worsening after reaching clinical improvement, we used 2, Fisher exact, Student t test, and Kruskall‐Wallis tests, as appropriate.
Adjusted analyses used generalized linear mixed models with a logit link to identify independent risk factors for worsening after reaching clinical improvement. Fixed effects for patient‐level factors and a random site effect were used. Factors were tested for inclusion in the multivariable model if they were found to be associated with worsening in unadjusted analyses (P<0.20) or were considered clinically important. Results are reported as odds ratios with 95% CIs.
We performed several sensitivity analyses to evaluate these improvement criteria: (1) we excluded the lowest RAO2 saturation requirement of 88%, (2) we examined a 94% daily estimated average RAO2 saturation threshold,[22] (3) we examined a 95% daily estimated average RAO2 saturation threshold,[8] and (4) we examined children age <12 months with no history of wheeze.
RESULTS
There were 1916 children hospitalized with bronchiolitis with data on all factors used to define clinical improvement and clinical worsening. The median number of days from the beginning of difficulty breathing until admission was 2 days (IQR, 15.5 days; range, 18 days) and from the beginning of difficulty breathing until clinical improvement was 4 days (IQR, 37.5 days; range, 133 days) (Figure 1). The variance for days to admission was significantly less than the variance for days to clinical improvement (P<0.001).

In this observational study, clinicians discharged 214 (11%) of the 1916 children before meeting the definition of clinical improvement. Thus, 1702 (89%; 95% CI: 87%‐90%) children reached the clinical improvement criteria, had a LOS >1 day, and had data on all factors (Figure 2).

Of the 1702 children who met the clinical improvement criteria, there were 76 children (4%; 95% CI: 3%5%) who worsened (Figure 2). The worsening occurred within a median of 1 day (IQR, 13 days) of clinical improvement. Forty‐six (3%) of the children required transfer to the ICU (1 required intubation, 1 required continuous positive airway pressure, and 4 had apnea), 23 (1%) required oxygen, and 17 (1%) required IV fluids. Eight percent of children met multiple criteria for worsening. A comparison between children who did and did not worsen is shown in Table 1. In general, children who worsened after improvement were younger and born earlier. These children also presented in more severe respiratory distress, had moderate or severe retractions, oxygen saturation <85% at hospitalization, inadequate oral intake, and apnea documented during the hospitalization. Neither viral etiology nor site of care influenced whether the children worsened after improving. However, stratified analysis of children based on initial location of admission (ie, ICU or ward) showed that among the children admitted to the ICU from the emergency department (ED), 89% met the improvement criteria and 19% clinically worsened. In contrast, among children admitted to the ward from the ED, 89% met the improvement criteria, and only 2% clinically worsened. Stratified multivariable models based on the initial location of admission from the ED (ie, ICU or ward) were not possible due to small sample sizes after stratification. None of these children had relapse events requiring rehospitalization within either 24 hours or 7 days of discharge.
| Did Not Worsen, n=1,626 | Worsened, n=76 | P Value | |
|---|---|---|---|
| |||
| Demographic characteristics | |||
| Age <2 months, % | 29 | 57 | <0.001 |
| Month of birth, % | 0.02 | ||
| OctoberMarch | 61 | 75 | |
| AprilSeptember | 39 | 25 | |
| Sex, % | 0.51 | ||
| Male | 59 | 55 | |
| Female | 41 | 45 | |
| Race, % | 0.050 | ||
| White | 63 | 58 | |
| Black | 23 | 34 | |
| Other or missing | 14 | 8 | |
| Hispanic ethnicity, % | 37 | 22 | 0.01 |
| Insurance, % | 0.87 | ||
| Nonprivate | 68 | 67 | |
| Private | 32 | 33 | |
| Medical history | |||
| Gestational age <37 weeks, % | 23 | 39 | 0.002 |
| Birth weight, % | 0.52 | ||
| <5 lbs | 13 | 12 | |
| 5 lbs | 34 | 41 | |
| 7 lbs | 53 | 47 | |
| Mother's age, median (IQR) | 27 (2333) | 27 (2233) | 0.54 |
| Is or was breastfed, % | 61 | 51 | 0.10 |
| Smoked during pregnancy, % | 15 | 20 | 0.22 |
| Exposure to smoke, % | 13 | 20 | 0.11 |
| Family history of asthma, % | 0.89 | ||
| Neither parent | 68 | 64 | |
| Either mother or father | 27 | 30 | |
| Both parents | 4 | 4 | |
| Do not know/missing | 2 | 1 | |
| History of wheezing, % | 23 | 17 | 0.24 |
| History of eczema, % | 16 | 7 | 0.04 |
| History of intubation, % | 9 | 12 | 0.50 |
| Major, relevant, comorbid medical disorder, % | 20 | 24 | 0.46 |
| Current illness | |||
| When difficulty breathing began, preadmission, % | 0.63 | ||
| 1 day | 70 | 75 | |
| <1 day | 28 | 23 | |
| No difficulty preadmission | 2 | 3 | |
| Weight, lbs, median (IQR) | 12.3 (8.817.4) | 9.0 (6.613.2) | 0.001 |
| Temperature, F, median (IQR) | 99.5 (98.6100.6) | 99.4 (98.1100.4) | 0.06 |
| Pulse, beats per minute by age | 0.82 | ||
| Low | 0.3 | 0 | |
| Normal | 48 | 46 | |
| High | 51 | 54 | |
| Respiratory rate, breaths per minute, median (IQR) | 48 (4060) | 48 (3864) | 0.28 |
| Retractions, % | 0.001 | ||
| None | 22 | 25 | |
| Mild | 43 | 24 | |
| Moderate | 26 | 33 | |
| Severe | 4 | 12 | |
| Missing | 5 | 7 | |
| Oxygen saturation by pulse oximetry or ABG, % | 0.001 | ||
| <85 | 4 | 12 | |
| 8587.9 | 3 | 4 | |
| 8889.9 | 5 | 0 | |
| 9093.9 | 18 | 11 | |
| 94 | 72 | 73 | |
| Oral intake, % | <0.001 | ||
| Adequate | 45 | 22 | |
| Inadequate | 42 | 63 | |
| Missing | 13 | 14 | |
| Presence of apnea, % | 7 | 24 | <0.001 |
| RSV‐A, % | 44 | 41 | 0.54 |
| RSV‐B, % | 30 | 25 | 0.36 |
| HRV, % | 24 | 24 | 0.88 |
| Chest x‐ray results during ED/preadmission visit | |||
| Atelectasis | 12 | 13 | 0.77 |
| Infiltrate | 13 | 11 | 0.50 |
| Hyperinflated | 18 | 21 | 0.47 |
| Peribronchial cuffing/thickening | 23 | 17 | 0.32 |
| Normal | 14 | 16 | 0.75 |
| White blood count, median (IQR) | 11.2 (8.714.4) | 11.9 (9.214.4) | 0.60 |
| Platelet count, median (IQR) | 395 (317490) | 430 (299537) | 0.56 |
| Sodium, median (IQR) | 138 (136140) | 137 (135138) | 0.19 |
| Hospital length of stay, median (IQR) | 2 (14) | 4.5 (28) | <0.001 |
| One‐week follow‐up | |||
| Relapse within 24 hours of hospital discharge requiring hospital admission, % | 0.5 | 0 | 0.56 |
| Relapse within 7 days of hospital discharge requiring hospital admission, % | 1 | 0 | 0.35 |
On multivariable analysis (Table 2), independent risk factors for worsening after reaching the clinical improvement criteria were young age, preterm birth, and presenting to care with more severe bronchiolitis represented by severe retractions, inadequate oral intake, or apnea. To further evaluate the improvement criteria in the current analysis, multiple sensitivity analyses were conducted. The frequency of clinical worsening after reaching the improvement criteria was stable when we examined different RA02 criteria in sensitivity analyses: (1) excluding RA02 as a criterion for improvement: 90% met improvement criteria and 4% experienced clinical worsening, (2) changing the average RA02 threshold for clinical improvement to 94%: 62% met improvement criteria and 6% experienced clinical worsening, and (3) changing the average RA02 threshold for clinical improvement to 95%: 47% met improvement criteria and 5% experienced clinical worsening. Furthermore, stratifying by age <2 months and restricting to more stringent definitions of bronchiolitis (ie, age <1 year or age <1 year+no history of wheezing) also did not materially change the results (see Supporting Figure 1 in the online version of this article).
| Odds Ratio | 95% CI | P Value | |
|---|---|---|---|
| |||
| Age <2 months | 3.51 | 2.07‐5.94 | <0.001 |
| Gestational age <37 weeks | 1.94 | 1.13‐3.32 | 0.02 |
| Retractions | |||
| None | 1.30 | 0.80‐3.23 | 0.19 |
| Mild | 1.0 | Reference | |
| Moderate | 1.91 | 0.99‐3.71 | 0.06 |
| Severe | 5.55 | 2.1214.50 | <0.001 |
| Missing | 1.70 | 0.53‐5.42 | 0.37 |
| Oral intake | |||
| Adequate | 1.00 | Reference | |
| Inadequate | 2.54 | 1.39‐4.62 | 0.002 |
| Unknown/missing | 1.88 | 0.79‐4.44 | 0.15 |
| Presence of apnea | 2.87 | 1.45‐5.68 | 0.003 |
We compared the 214 children who were discharged prior to reaching clinical improvement with the 1702 children who reached the clinical improvement criteria. The 214 children were less likely to be age <2 months (22% vs 30%; P=0.02). These 2 groups (214 vs 1702) were similar with respect to severe retractions (2% vs 4%; P=0.13), median respiratory rate (48 vs 48; P=0.42), oxygen saturation <90% (15% vs 11%; P=0.07), inadequate oral intake (50% vs 43%; P=0.13), and rates of relapse events requiring rehospitalization within both 24 hours (0.6% vs 0.6%; P=0.88) and 7 days (1% vs 1%; P=0.90) of discharge.
DISCUSSION
In this large, multicenter, multiyear study of children hospitalized with bronchiolitis, we found that children present to a hospital in a relatively narrow time frame, but their time to recovery in the hospital is highly variable. Nonetheless, 96% of children continued to improve once they had: (1) improving or stable retractions rated as none/mild, (2) a decreasing or stable RR by age, (3) estimated average RAO2 saturation 90% and lowest RAO2 saturation of 88%, and (4) were hydrated. The 4% of children who worsened after clinically improving were more likely to be age <2 months, born <37 weeks, and present with more severe distress (ie, severe retractions, inadequate oral intake, or apnea). Based on the low risk of worsening after clinical improvement, especially among children admitted to the regular ward (2%), we believe these 4 clinical criteria could be used as discharge criteria for this common pediatric illness with a predominantly monophasic clinical course.
Variability in hospital LOS for children with bronchiolitis exists in the United States[3] and internationally.[4, 5] Cheung and colleagues analyzed administrative data from over 75,000 children admitted for bronchiolitis in England between April 2007 and March 2010 and found sixfold variation in LOS between sites. They concluded that this LOS variability was due in part to providers' clinical decision making.[5] Srivastava and colleagues[23] addressed variable clinician decision making in bronchiolitis and 10 other common pediatric conditions by embedding discharge criteria developed by expert consensus into admission order sets. They found that for children with bronchiolitis, the embedded discharge criteria reduced the median LOS from 1.91 to 1.87 days. In contrast to the single‐center data presented by White and colleagues,[24] the prospective, multicenter MARC‐30 data provide a clear understanding of the normal clinical course for children hospitalized with bronchiolitis, determine if children clinically worsen after clinical improvement, and provide data about discharge criteria for children hospitalized with bronchiolitis. Although there is a lack of rigorous published data, the lower tract symptoms of bronchiolitis (eg, cough, retractions) are said to peak on days 5 to 7 of illness and then gradually resolve.[25] In the present study, we found that the time from the onset of difficulty breathing until hospital admission is less variable than the time from the onset of difficulty breathing until either clinical improvement or discharge. Although 75% of children have clinically improved within 7.5 days of difficulty breathing based on the IQR results, the remaining 25% may have a more prolonged recovery in the hospital of up to 3 weeks. Interestingly, prolonged recovery times from bronchiolitis have also been noted in children presenting to the ED[26] and in an outpatient population.[27] It is unclear why 20% to 25% of children at different levels of severity of illness have prolonged recovery from bronchiolitis, but this group of children requires further investigation.
Given the variability of recovery times, clinicians may have difficulty knowing when a child is ready for hospital discharge. One of the main stumbling blocks for discharge readiness in children with bronchiolitis is the interpretation of the oxygen saturation value.[6, 8, 9, 20, 28] However, it should be considered that interpreting the oxygen saturation in a child who is clinically improving in the hospital setting is different than interpreting the oxygen saturation of a child in the ED or the clinic whose clinical course is less certain.[22] In the hospital setting, using the oxygen saturation value in in the AAP guideline,[9] 4% of children clinically worsened after they met the improvement criteria, a clinical pattern observed previously with supplemental oxygen.[28] This unpredictability may explain some of the variation in providers' clinical decision making.[5] The children who worsened, and therefore deserve more cautious discharge planning, were young (<2 months), premature (<37 weeks gestational age), and presented in more severe distress. Those children admitted to the ICU from the ED worsened more commonly than children admitted to the ward (19% vs 2%). Interestingly, the viral etiology of the child's bronchiolitis did not influence whether a child worsened after reaching the improvement criteria. Therefore, although children with RV bronchiolitis have a shorter hospital LOS than children with RSV bronchiolitis,[11] the pattern of recovery did not differ by viral etiology.
In addition to unsafe discharges, clinicians may be concerned about the possibility of readmissions. Although somewhat controversial, hospital readmission is being used as a quality of care metric.[29, 30, 31] One response to minimize readmissions would be for clinicians to observe children for longer than clinically indicated.[32] However, shorter LOS is not necessarily associated with increased readmission rates.[33] Given that the geometric mean of hospital charges per child with bronchiolitis increased from $6380 in 2000 to $8530 in 2009,[34] the potential for safely reducing hospital LOS by using the discharge criteria proposed in the current study instead of other criteria[8] may net substantial cost savings. Furthermore, reducing LOS would decrease the time children expose others to these respiratory viruses and possibly reduce medical errors.[35]
Our study has some potential limitations. Because the study participants were all hospitalized, these data do not inform admission or discharge decisions from either the ED or the clinic; but other data address those clinical scenarios.[22] Also, the 16 sites that participated in this study were large, urban teaching hospitals. Consequently, these results are not necessarily generalizable to smaller community hospitals. Although numerous data points were required to enter the analytic cohort, only 9% of the sample was excluded for missing data. There were 214 children who did not meet our improvement criteria by the time of discharge. Although the inability to include these children in the analysis may be seen as a limitation, this practice variability underscores the need for more data about discharging hospitalized children with bronchiolitis. Last, site teams reviewed medical records daily. More frequent recording of the clinical course would have yielded more granular data, but the current methodology replicates how data are generally presented during patient care rounds, when decisions about suitability for discharge are often considered.
CONCLUSION
We documented in this large multicenter study that most children hospitalized with bronchiolitis had a wide range of time to recovery, but the vast majority continued to improve once they reached the identified clinical criteria that predict a safe discharge to home. The children who worsened after clinical improvement were more likely to be younger, premature infants presenting in more severe distress. Although additional prospective validation of these hospital discharge criteria is warranted, these data may help clinicians make more evidence‐based discharge decisions for a common pediatric illness with high practice variation, both in the United States[3] and in other countries.[4, 5]
Acknowledgements
Collaborators in the MARC‐30 Study: Besh Barcega, MD, Loma Linda University Children's Hospital, Loma Linda, CA; John Cheng, MD, Children's Healthcare of Atlanta at Egleston, Atlanta, GA; Dorothy Damore, MD, New York Presbyterian Hospital‐Cornell, New York, NY; Carlos Delgado, MD, Children's Healthcare of Atlanta at Egleston, Atlanta, GA; Haitham Haddad, MD, Rainbow Babies & Children's Hospital, Cleveland, OH; Paul Hain, MD, Monroe Carell Jr. Children's Hospital at Vanderbilt, Nashville, TN; Frank LoVecchio, DO, Maricopa Medical Center, Phoenix, AZ; Charles Macias, MD MPH, Texas Children's Hospital, Houston, TX; Jonathan Mansbach, MD, MPH, Boston Children's Hospital, Boston, MA; Eugene Mowad, MD, Akron Children's Hospital, Akron, OH; Brian Pate, MD, Children's Mercy Hospital, Kansas City, MO; Mark Riederer, MD, Monroe Carell Jr. Children's Hospital at Vanderbilt, Nashville, TN; M. Jason Sanders, MD, Children's Memorial Hermann Hospital, Houston, TX; Alan R. Schroeder, MD, Santa Clara Valley Medical Center, San Jose, CA; Nikhil Shah, MD, New York Presbyterian Hospital‐Cornell, New York, NY; Michelle Stevenson, MD, MS, Kosair Children's Hospital, Louisville, KY; Erin Stucky Fisher, MD, Rady Children's Hospital, San Diego, CA; Stephen Teach, MD, MPH, Children's National Medical Center, Washington, DC; Lisa Zaoutis, MD, Children's Hospital of Philadelphia, Philadelphia, PA.
Disclosures: This study was supported by grants U01 AI‐67693 and K23 AI‐77801 from the National Institutes of Health (Bethesda, MD). The content of this manuscript is solely the responsibility of the authors and does not necessarily represent the official views of the National Institute of Allergy and Infectious Diseases or the National Institutes of Health. Drs. Mansbach and Piedra have provided consultation to Regeneron Pharmaceuticals. Otherwise, no authors report any potential conflicts of interest, including relevant financial interests, activities, relationships, and affiliations.
- , , , , . Infectious disease hospitalizations among infants in the United States. Pediatrics. 2008;121(2):244–252.
- . “A hospital is no place to be sick” Samuel Goldwyn (1882–1974). Arch Dis Child. 2009;94(8):565–566.
- , , , , , Variation in inpatient diagnostic testing and management of bronchiolitis. Pediatrics. 2005;115(4):878–884.
- , , , International variation in the management of infants hospitalized with respiratory syncytial virus. International RSV Study Group. Eur J Pediatr. 1998;157(3):215–220.
- , , , , . Population variation in admission rates and duration of inpatient stay for bronchiolitis in England. Arch Dis Child. 2013;98(1):57–59.
- , , , . Impact of pulse oximetry and oxygen therapy on length of stay in bronchiolitis hospitalizations. Arch Pediatr Adolesc Med. 2004;158(6):527–530.
- , , . Pulse oximetry in pediatric practice. Pediatrics. 2011;128(4):740–752.
- Scottish Intercollegiate Guidelines Network. Bronchiolitis in children (SIGN 91). In: NHS Quality Improvement Scotland. Edinburgh, Scotland: Scottish Intercollegiate Guidelines Network; 2006.
- , , , et al. Diagnosis and management of bronchiolitis. Pediatrics. 2006;118(4):1774–1793.
- , , , et al. Prospective multicenter study of children with bronchiolitis requiring mechanical ventilation. Pediatrics. 2012;130(3):e492–e500.
- , , , et al. Prospective multicenter study of viral etiology and hospital length of stay in children with severe bronchiolitis. Arch Pediatr Adolesc Med. 2012;166(8):700–706.
- , , , et al. Apnea in children hospitalized with bronchiolitis. Pediatrics. 2013;132(5):e1194–e1201.
- . Evaluation of the cardiovascular system: history and physical evaluation. In: Kliegman RM, Stanton BF, St. Geme JW III, Schor NF, Behrman RF, eds. Nelson Textbook of Pediatrics. Philadelphia, PA: Elsevier Saunders; 2011:1529–1536.
- , , , et al. Apnea in children hospitalized with bronchiolitis. Pediatrics. 2013;132(5):e1194–e1201.
- , , , et al. Respiratory viral infections in patients with chronic, obstructive pulmonary disease. J Infect. 2005;50(4):322–330.
- , , , . Evaluation of real‐time PCR for diagnosis of Bordetella pertussis infection. BMC Infect Dis. 2006;6:62.
- , , , , . Evaluation of three real‐time PCR assays for detection of Mycoplasma pneumoniae in an outbreak investigation. J Clin Microbiol. 2008;46(9):3116–3118.
- , , , et al. Normal ranges of heart rate and respiratory rate in children from birth to 18 years of age: a systematic review of observational studies. Lancet. 2011;377(9770):1011–1018.
- , , , , , . Development of heart and respiratory rate percentile curves for hospitalized children. Pediatrics. 2013;131(4):e1150–e1157.
- , . Effect of oxygen supplementation on length of stay for infants hospitalized with acute viral bronchiolitis. Pediatrics. 2008;121(3):470–475.
- , , , et al. Longitudinal assessment of hemoglobin oxygen saturation in healthy infants during the first 6 months of age. Collaborative Home Infant Monitoring Evaluation (CHIME) Study Group. J Pediatr. 1999;135(5):580–586.
- , , , et al. Prospective multicenter study of bronchiolitis: predicting safe discharges from the emergency department. Pediatrics. 2008;121(4):680–688.
- , , , et al. Delays in discharge in a tertiary care pediatric hospital. J Hosp Med. 2009;4(8):481–485.
- , , , et al. Using quality improvement to optimise paediatric discharge efficiency. BMJ Qual Saf. 2014;23(5):428–436.
- , . Bronchiolitis in infants and children: treatment; outcome; and prevention. In: Torchia M, ed. UpToDate. Alphen aan den Rijn, the Netherlands; Wolters Kluwer Health; 2013.
- , . Duration of illness in infants with bronchiolitis evaluated in the emergency department. Pediatrics. 2010;126(2):285–290.
- , , . Duration of illness in ambulatory children diagnosed with bronchiolitis. Arch Pediatr Adolesc Med. 2000;154(10):997–1000.
- , , , et al. A clinical pathway for bronchiolitis is effective in reducing readmission rates. J Pediatr. 2005;147(5):622–626.
- , , , et al. Measuring hospital quality using pediatric readmission and revisit rates. Pediatrics. 2013;132(3):429–436.
- , , , et al. Pediatric readmission prevalence and variability across hospitals. JAMA. 2013;309(4):372–380.
- , , , , , . Preventability of early readmissions at a children's hospital. Pediatrics. 2013;131(1):e171–e181.
- , . Hospital readmission: quality indicator or statistical inevitability? Pediatrics. 2013;132(3):569–570.
- , , , et al. Children's hospitals with shorter lengths of stay do not have higher readmission rates. J Pediatr. 2013;163(4):1034–1038.e1.
- , , , , . Trends in bronchiolitis hospitalizations in the United States, 2000–2009. Pediatrics. 2013;132(1):28–36.
- , , , . Preventable adverse events in infants hospitalized with bronchiolitis. Pediatrics. 2005;116(3):603–608.
- , , , , . Infectious disease hospitalizations among infants in the United States. Pediatrics. 2008;121(2):244–252.
- . “A hospital is no place to be sick” Samuel Goldwyn (1882–1974). Arch Dis Child. 2009;94(8):565–566.
- , , , , , Variation in inpatient diagnostic testing and management of bronchiolitis. Pediatrics. 2005;115(4):878–884.
- , , , International variation in the management of infants hospitalized with respiratory syncytial virus. International RSV Study Group. Eur J Pediatr. 1998;157(3):215–220.
- , , , , . Population variation in admission rates and duration of inpatient stay for bronchiolitis in England. Arch Dis Child. 2013;98(1):57–59.
- , , , . Impact of pulse oximetry and oxygen therapy on length of stay in bronchiolitis hospitalizations. Arch Pediatr Adolesc Med. 2004;158(6):527–530.
- , , . Pulse oximetry in pediatric practice. Pediatrics. 2011;128(4):740–752.
- Scottish Intercollegiate Guidelines Network. Bronchiolitis in children (SIGN 91). In: NHS Quality Improvement Scotland. Edinburgh, Scotland: Scottish Intercollegiate Guidelines Network; 2006.
- , , , et al. Diagnosis and management of bronchiolitis. Pediatrics. 2006;118(4):1774–1793.
- , , , et al. Prospective multicenter study of children with bronchiolitis requiring mechanical ventilation. Pediatrics. 2012;130(3):e492–e500.
- , , , et al. Prospective multicenter study of viral etiology and hospital length of stay in children with severe bronchiolitis. Arch Pediatr Adolesc Med. 2012;166(8):700–706.
- , , , et al. Apnea in children hospitalized with bronchiolitis. Pediatrics. 2013;132(5):e1194–e1201.
- . Evaluation of the cardiovascular system: history and physical evaluation. In: Kliegman RM, Stanton BF, St. Geme JW III, Schor NF, Behrman RF, eds. Nelson Textbook of Pediatrics. Philadelphia, PA: Elsevier Saunders; 2011:1529–1536.
- , , , et al. Apnea in children hospitalized with bronchiolitis. Pediatrics. 2013;132(5):e1194–e1201.
- , , , et al. Respiratory viral infections in patients with chronic, obstructive pulmonary disease. J Infect. 2005;50(4):322–330.
- , , , . Evaluation of real‐time PCR for diagnosis of Bordetella pertussis infection. BMC Infect Dis. 2006;6:62.
- , , , , . Evaluation of three real‐time PCR assays for detection of Mycoplasma pneumoniae in an outbreak investigation. J Clin Microbiol. 2008;46(9):3116–3118.
- , , , et al. Normal ranges of heart rate and respiratory rate in children from birth to 18 years of age: a systematic review of observational studies. Lancet. 2011;377(9770):1011–1018.
- , , , , , . Development of heart and respiratory rate percentile curves for hospitalized children. Pediatrics. 2013;131(4):e1150–e1157.
- , . Effect of oxygen supplementation on length of stay for infants hospitalized with acute viral bronchiolitis. Pediatrics. 2008;121(3):470–475.
- , , , et al. Longitudinal assessment of hemoglobin oxygen saturation in healthy infants during the first 6 months of age. Collaborative Home Infant Monitoring Evaluation (CHIME) Study Group. J Pediatr. 1999;135(5):580–586.
- , , , et al. Prospective multicenter study of bronchiolitis: predicting safe discharges from the emergency department. Pediatrics. 2008;121(4):680–688.
- , , , et al. Delays in discharge in a tertiary care pediatric hospital. J Hosp Med. 2009;4(8):481–485.
- , , , et al. Using quality improvement to optimise paediatric discharge efficiency. BMJ Qual Saf. 2014;23(5):428–436.
- , . Bronchiolitis in infants and children: treatment; outcome; and prevention. In: Torchia M, ed. UpToDate. Alphen aan den Rijn, the Netherlands; Wolters Kluwer Health; 2013.
- , . Duration of illness in infants with bronchiolitis evaluated in the emergency department. Pediatrics. 2010;126(2):285–290.
- , , . Duration of illness in ambulatory children diagnosed with bronchiolitis. Arch Pediatr Adolesc Med. 2000;154(10):997–1000.
- , , , et al. A clinical pathway for bronchiolitis is effective in reducing readmission rates. J Pediatr. 2005;147(5):622–626.
- , , , et al. Measuring hospital quality using pediatric readmission and revisit rates. Pediatrics. 2013;132(3):429–436.
- , , , et al. Pediatric readmission prevalence and variability across hospitals. JAMA. 2013;309(4):372–380.
- , , , , , . Preventability of early readmissions at a children's hospital. Pediatrics. 2013;131(1):e171–e181.
- , . Hospital readmission: quality indicator or statistical inevitability? Pediatrics. 2013;132(3):569–570.
- , , , et al. Children's hospitals with shorter lengths of stay do not have higher readmission rates. J Pediatr. 2013;163(4):1034–1038.e1.
- , , , , . Trends in bronchiolitis hospitalizations in the United States, 2000–2009. Pediatrics. 2013;132(1):28–36.
- , , , . Preventable adverse events in infants hospitalized with bronchiolitis. Pediatrics. 2005;116(3):603–608.
© 2015 Society of Hospital Medicine
Stranger than Fiction
A 65‐year‐old man suffered a myocardial infarction (MI) while traveling in Thailand. After 7 days of recovery, the patient departed for his home in the United States. He developed substernal, nonexertional, inspiratory chest pain and shortness of breath during his return flight and presented directly to an emergency room after arrival.
Initially, the evaluation should focus on life‐threatening diagnoses and not be distracted by the travel history. The immediate diagnostic concerns are active cardiac ischemia, complications of MI, and pulmonary embolus. Other cardiac causes of dyspnea include ischemic mitral regurgitation, postinfarction pericarditis with or without pericardial effusion, and heart failure. Mechanical complications of infarction, such as left ventricular free wall rupture or rupture of the interventricular septum, can occur in this time frame and are associated with significant morbidity. Pneumothorax may be precipitated by air travel, especially in patients with underlying lung disease. The immobilization associated with long airline flights is a risk factor for thromboembolic disease, which is classically associated with pleuritic chest pain. Inspiratory chest pain is also associated with inflammatory processes involving the pericardium or pleura. If pneumonia, pericarditis, or pleural effusion is present, details of his travel history will become more important in his evaluation.
The patient elaborated that he spent 10 days in Thailand. On the third day of his trip he developed severe chest pain while hiking toward a waterfall in a rural northern district. He was transferred to a large private hospital, where he received a stent in the proximal left anterior descending coronary artery 4 hours after symptom onset. At discharge he was prescribed ticagrelor 90 mg twice daily and daily doses of losartan 50 mg, furosemide 20 mg, spironolactone 12.5 mg, aspirin 81 mg, ivabradine 2.5 mg, and pravastatin 40 mg. He had also been taking doxycycline for malaria prophylaxis since departing the United States.
His past medical history was notable for hypertension and hyperlipidemia. The patient was a lifelong nonsmoker, did not use illicit substances, and consumed no more than 2 alcoholic beverages per day. He denied cough, fevers, chills, diaphoresis, weight loss, recent upper respiratory infection, abdominal pain, hematuria, and nausea. However, he reported exertional dyspnea following his MI and nonbloody diarrhea that occurred a few days prior to his return flight and resolved without intervention.
The remainder of his past medical history confirms that he received appropriate post‐MI care, but does not substantially alter the high priority concerns in his differential diagnosis. Diarrhea may occur in up to 50% of international travelers, and is especially common when returning from Southeast Asia or the Indian subcontinent. Disease processes that may explain diarrhea and subsequent dyspnea include intestinal infections that spread to the lung (eg, ascariasis and Loeffler syndrome), infection that precipitates neuromuscular weakness (eg, Campylobacter and Guillain‐Barr syndrome), or infection that precipitates heart failure (eg, coxsackievirus, myocarditis).
On admission, his temperature was 36.2C, heart rate 91 beats per minute, blood pressure 135/81 mm Hg, respiratory rate 16 breaths per minute, and oxygen saturation 98% on room air. Cardiac exam revealed a regular rhythm without rubs, murmurs, or diastolic gallops. He had no jugular venous distention, and no lower extremity edema. His distal pulses were equal and palpable throughout. Pulmonary exam was notable for decreased breath sounds at both bases without wheezing, rhonchi, or crackles noted. He had no rashes, joint effusions, or jaundice. Abdominal and neurologic examinations were unremarkable.
Diminished breath sounds may suggest atelectasis or pleural effusion; the latter could account for the patient's inspiratory chest pain. A chest radiograph is essential to evaluate this finding further. The physical examination is not suggestive of decompensated heart failure; measurement of serum brain natriuretic peptide level would further exclude that diagnosis.
Laboratory evaluation revealed a leukocytosis of 16,000/L, with 76% polymorphonuclear cells and 12% lymphocytes without eosinophils or band forms; a hematocrit of 38%; and a platelet count of 363,000/L. The patient had a creatinine of 1.6 mg/dL, potassium of 2.7 mEq/L, and a troponin‐I of 1.0 ng/mL (normal 0.40 ng/mL), with the remainder of the routine serum chemistries within normal limits. An electrocardiogram (ECG) showed QS complexes in the anteroseptal leads, and a chest radiograph showed bibasilar consolidations and a left pleural effusion. A ventilation‐perfusion scan of the chest was performed to evaluate for pulmonary embolism, and was interpreted as low probability. Transthoracic echocardiography demonstrated severe left ventricular systolic dysfunction with anterior wall akinesis, and an aneurysmal left ventricle with an apical thrombus. No significant valvular pathology or other structural defects were noted.
The ECG and echocardiogram confirm the history of a large anteroseptal infarction with severe left ventricular dysfunction. Serial troponin testing would be reasonable. However, the absence of any acute ischemic ECG changes, typical angina symptoms, and a relatively normal troponin level all suggest his chest pain does not represent active ischemia. His low abnormal troponin‐I is consistent with slow resolution after a large ischemic event in the recent past, and his anterior wall akinesis is consistent with prior infarction in the territory of his culprit left anterior descending coronary artery.
Although acute cardiac conditions appear less likely, the brisk leukocytosis in a returned traveler prompts consideration of infection. His lung consolidations could represent either new or resolving pneumonia. The complete absence of cough and fever is unusual for pneumonia, yet clinical findings are not as sensitive as chest radiograph for this diagnosis. At this point, typical organisms as well as uncommon pathogens associated with diarrhea or his travel history should be included in the differential.
After 24 hours, the patient was discharged on warfarin to treat the apical thrombus and moxifloxacin for a presumed community‐acquired pneumonia. Eight days after discharge, the patient visited his primary care physician with improving, but not resolved, shortness of breath and pleuritic pain despite completing the 7‐day course of moxifloxacin. A chest radiograph showed a large posterior left basal pleural fluid collection, increased from previous.
In the setting of a recent infection, the symptoms and radiographic findings suggest a complicated parapneumonic effusion or empyema. Failure to drain a previously seeded fluid collection leaves bacterial pathogens susceptible to moxifloxacin on the differential, including Streptococcus pneumoniae, Staphylococcus aureus, Legionella species, and other enterobacteriaciae (eg, Klebsiella pneumoniae).
The indolent course should also prompt consideration of more unusual pathogens, including roundworms (such as Ascaris) or lung flukes (Paragonimus), either of which can cause a lung infection without traditional pneumonia symptoms. Tuberculosis tends to present months (or years) after exposure. Older adults may manifest primary pulmonary tuberculosis with lower lobe infiltrates, consistent with this presentation. However, moxifloxacin is quite active against tuberculosis, and although single drug therapy would not be expected to cure the patient, it would be surprising for him to progress this quickly on moxifloxacin.
In northern Thailand, Burkholderia pseudomallei is a common cause of bacteremic pneumonia. The organism often has high‐level resistance to fluoroquinolones, and may present in a more insidious fashion than other causes of community‐acquired pneumonia. Although infection with B pseudomallei (melioidosis) can occasionally mimic apical pulmonary tuberculosis and may present after a prolonged latent period, it most commonly manifests as an acute pneumonia.
The patient was prescribed 10 days of amoxicillin‐clavulanic acid and clindamycin, and decubitus films were ordered to assess the effusion. These films, obtained 5 days later, showed a persistent pleural effusion. Subsequent ultrasound demonstrated loculated fluid, but a thoracentesis was not performed at that time due to the patient's therapeutic international normalized ratio and dual antiplatelet therapy.
The loculation further suggests a complicated parapneumonic effusion or empyema. Clindamycin adds very little to amoxicillin‐clavulanate as far as coverage of oral anaerobes or common pneumonia pathogens and may add to the risk of antibiotic side effects. A susceptible organism might not clear because of failure to drain this collection; if undertreated bacterial infection is suspected, tube thoracentesis is the established standard of care. However, the protracted course of illness makes untreated pyogenic bacterial infections unlikely.
At this point, the top 2 diagnostic considerations are Paragonimus westermani and B pseudomallei. P westermani is initially ingested, usually from an undercooked freshwater crustacean. Infected patients may experience a brief diarrheal illness, as this patient reported. However, infected patients typically have a brisk peripheral eosinophilia.
Melioidosis is thus the leading concern. Amoxicillin‐clavulanate is active against many strains of B pseudomallei, so the failure of the patient to worsen could be seen as a partial treatment and supports this diagnosis. However, as prolonged therapy is necessary for complete eradication of B pseudomallei, a definitive, culture‐based diagnosis should be established before committing the patient to months of antibiotics.
After completing 10 days of clindamycin and amoxicillin‐clavulanate, the patient reported improvement of his pleuritic pain, and repeat physical exam suggested interval decrease in the size of the effusion. Two days later, the patient began experiencing dysuria that persisted despite 3 days of nitrofurantoin.
Melioidosis can also involve the genitourinary tract. Hematogenous spread of B pseudomallei can seed a number of visceral organs including the bladder, joints, and bones. Men with suspected urinary infection should be evaluated for the possibility of prostatitis, an infection with considerable morbidity that requires extended therapy. This gentleman should have a prostate exam, and blood and urine cultures should be collected if prostatitis is suspected. Empiric antibiotics are not recommended without culture in a patient with complicated urinary tract infection.
Prostate exam was unremarkable. A urine culture grew a gram‐negative rod identified as B pseudomallei. Because B pseudomallei can cause fulminant sepsis, the infectious disease consultant requested that he return for admission, further evaluation, and initiation of intravenous antibiotics. Computed tomography (CT) of the chest, abdomen, and pelvis revealed multiple pulmonary nodules, a persistent left pleural effusion, and a rim‐enhancing hypodensity in the prostate consistent with abscess (Figure 1). Blood and pleural fluid cultures were negative.
Initial treatment for a patient with severe or metastatic B pseudomallei infection requires high‐dose intravenous antibiotic therapy. Ceftazidime, imipenem, and meropenem are the best studied agents for this purpose. Surgical drainage should be considered for the abscess. Following the completion of intensive intravenous therapy, relapse rates are high unless a longer‐term, consolidation therapy is pursued. Trimethoprim‐sulfamethoxazole is the recommended agent.
The patient was treated with high‐dose ceftazidime for 2 weeks, followed by 6 months of high‐dose oral trimethoprim‐sulfamethoxazole. His symptoms resolved, and 7 months after presentation, he continued to feel well.
DISCUSSION
Melioidosis refers to any infection caused by B pseudomallei, a gram‐negative bacillus found in soil and water, most commonly in Southeast Asia and Australia.[1] It is an important cause of pneumonia in endemic regions; in Thailand, the incidence is as high as 12 cases per 100,000 people, and it is the third leading infectious cause of death, following human immunodeficiency virus and tuberculosis.[2] However, it occurs only as an imported infection in the United States and remains an unfamiliar infection for many US medical practitioners. Melioidosis should be considered in patients returning from endemic regions presenting with sepsis, pneumonia, urinary symptoms, or abscesses.
B pseudomallei can be transmitted to humans through exposure to contaminated soil or water via ingestion, inhalation, or percutaneous inoculation.[1] Outbreaks typically occur during the rainy season and after typhoons.[1, 3] Presumably, this patient's exposure to B pseudomallei occurred while hiking and wading in freshwater lakes and waterfalls. Although hospital‐acquired melioidosis has not been reported, and isolation precautions are not necessary, rare cases of disease acquired via laboratory exposure have been reported among US healthcare workers. Clinicians suspecting melioidosis should alert the receiving laboratory.[4]
The treatment course for melioidosis is lengthy and should involve consultation with an infectious disease specialist. B pseudomallei is known to be resistant to penicillin, first‐ and second‐generation cephalosporins, and moxifloxacin. The standard treatment includes 10 to 14 days of intravenous ceftazidime, meropenem, or imipenem, and then trimethoprim‐sulfamethoxazole for 3 to 6 months.[1] Treatment should be guided by culture susceptibility data when available. There are reports of B pseudomallei having different resistance patterns within the same host; clinicians should culture all drained fluid collections and tailor antibiotics to the most resistant strain recovered.[5, 6] Although melioidosis is a life‐threatening infection, previously healthy patients have an excellent prognosis assuming prompt diagnosis and treatment are provided.[3]
After excluding common causes of chest pain, the discussant identified the need to definitively establish a microbiologic diagnosis by obtaining pleural fluid. Although common clinical scenarios can often be treated with guideline‐supported empiric antibiotics, the use of serial courses of empiric antibiotics should be carefully questioned and is generally discouraged. Specific data to prove or disprove the presence of infection should be obtained before exposing a patient to the risks of multiple drugs or prolonged antibiotic therapy, as well as the risks of delayed (or missed) diagnosis. Unfortunately, a complete evaluation was delayed by clinical contraindications to diagnostic thoracentesis, and a definitive diagnosis was reached only after development of more widespread symptoms.
This patient's protean presentation is not surprising given his ultimate diagnosis. B pseudomallei has been termed the great mimicker, as disease presentation and organ involvement can vary from an indolent localized infection to acute severe sepsis.[7] Pneumonia and genitourinary infections are the most common manifestations, although skin infections, bacteremia, septic arthritis, and neurologic disease are also possible.[1, 3] In addition, melioidosis may develop after a lengthy incubation. In a case series, confirmed incubation periods ranged from 1 to 21 days (mean, 9 days); however, cases of chronic (>2 months) infection, mimicking tuberculosis, are estimated to occur in about 12% of cases.[4] B pseudomallei is also capable of causing reactivation disease, similar to tuberculosis. It was referred to as the Vietnamese time bomb when US Vietnam War veterans, exposed to the disease when helicopters aerosolized the bacteria in the soil, developed the disease only after their return to the United States.[8] Fortunately, only a tiny fraction of the quarter‐million soldiers with serologically confirmed exposure to the bacteria ultimately developed disease.
In The Adventure of the Dying Detective, Sherlock Holmes fakes a serious illness characterized by shortness of breath and weakness to trick an adversary into confessing to murder. The abrupt, crippling infection mimicked by Holmes is thought by some to be melioidosis.[9, 10] Conan Doyle's story was published in 1913, a year after melioidosis was first reported in the medical literature, and the exotic, protean infection may well have sparked Doyle's imagination. However, this patient's case of melioidosis proved stranger than fiction in its untimely concomitant development with an MI. Cracking our case required imagination and nimble thinking to avoid a number of cognitive pitfalls. The patient's recent MI anchored reasoning at his initial presentation, and the initial diagnosis of community‐acquired pneumonia raised the danger of premature closure. Reaching the correct diagnosis required an open mind, a detailed travel history, and firm microbiologic evidence. Hospitalists need not be expert in the health risks of travel to specific foreign destinations, but investigating those risks can hasten proper diagnosis and treatment.
TEACHING POINTS
- Melioidosis should be considered in patients returning from endemic regions who present with sepsis, pneumonia, urinary symptoms, or an abscess.
- For patients with a loculated parapneumonic effusion, tube thoracentesis for culture and drainage is the standard of care for diagnosis and treatment.
- Culture identification and antibiotic sensitivities are critical for management of B pseudomallei, because prolonged antibiotic treatment is needed.
Disclosure
Nothing to report.
- , , . Melioidosis. N Engl J Med. 2012;367(11):1035–1044.
- , , , et al. Increasing incidence of human melioidosis in Northeast Thailand. Am J Trop Med Hyg. 2010;82(6):1113–1117.
- , , . The epidemiology and clinical spectrum of melioidosis: 540 cases from the 20 year Darwin prospective study. PLoS Negl Trop Dis. 2010;4(11):e900.
- , , , et al. Management of accidental laboratory exposure to Burkholderia pseudomallei and B. mallei. Emerg Infect Dis. 2008;14(7):e2.
- , , . Variations in ceftazidime and amoxicillin‐clavulanate susceptibilities within a clonal infection of Burkholderia pseudomallei. J Clin Microbiol. 2009;47(5):1556–1558.
- , , , et al. Within‐host evolution of Burkholderia pseudomallei in four cases of acute melioidosis. PLoS Pathog. 2010;6(1):e1000725.
- , , , , . Melioidosis: insights into the pathogenicity of Burkholderia pseudomallei. Nat Rev Microbiol. 2006;4(4):272–282.
- , . Melioidosis. In: Dembeck ZF, ed. Medical Aspects of Biological Warfare. 2nd ed. Washington, DC: Office of the Surgeon General; 2007:146–166.
- . Sherlock Holmes and a biological weapon. J R Soc Med. 2002;95(2):101–103.
- . Sherlock Holmes and tropical medicine: a centennial appraisal. Am J Trop Med Hyg. 1994;50:99–101.
A 65‐year‐old man suffered a myocardial infarction (MI) while traveling in Thailand. After 7 days of recovery, the patient departed for his home in the United States. He developed substernal, nonexertional, inspiratory chest pain and shortness of breath during his return flight and presented directly to an emergency room after arrival.
Initially, the evaluation should focus on life‐threatening diagnoses and not be distracted by the travel history. The immediate diagnostic concerns are active cardiac ischemia, complications of MI, and pulmonary embolus. Other cardiac causes of dyspnea include ischemic mitral regurgitation, postinfarction pericarditis with or without pericardial effusion, and heart failure. Mechanical complications of infarction, such as left ventricular free wall rupture or rupture of the interventricular septum, can occur in this time frame and are associated with significant morbidity. Pneumothorax may be precipitated by air travel, especially in patients with underlying lung disease. The immobilization associated with long airline flights is a risk factor for thromboembolic disease, which is classically associated with pleuritic chest pain. Inspiratory chest pain is also associated with inflammatory processes involving the pericardium or pleura. If pneumonia, pericarditis, or pleural effusion is present, details of his travel history will become more important in his evaluation.
The patient elaborated that he spent 10 days in Thailand. On the third day of his trip he developed severe chest pain while hiking toward a waterfall in a rural northern district. He was transferred to a large private hospital, where he received a stent in the proximal left anterior descending coronary artery 4 hours after symptom onset. At discharge he was prescribed ticagrelor 90 mg twice daily and daily doses of losartan 50 mg, furosemide 20 mg, spironolactone 12.5 mg, aspirin 81 mg, ivabradine 2.5 mg, and pravastatin 40 mg. He had also been taking doxycycline for malaria prophylaxis since departing the United States.
His past medical history was notable for hypertension and hyperlipidemia. The patient was a lifelong nonsmoker, did not use illicit substances, and consumed no more than 2 alcoholic beverages per day. He denied cough, fevers, chills, diaphoresis, weight loss, recent upper respiratory infection, abdominal pain, hematuria, and nausea. However, he reported exertional dyspnea following his MI and nonbloody diarrhea that occurred a few days prior to his return flight and resolved without intervention.
The remainder of his past medical history confirms that he received appropriate post‐MI care, but does not substantially alter the high priority concerns in his differential diagnosis. Diarrhea may occur in up to 50% of international travelers, and is especially common when returning from Southeast Asia or the Indian subcontinent. Disease processes that may explain diarrhea and subsequent dyspnea include intestinal infections that spread to the lung (eg, ascariasis and Loeffler syndrome), infection that precipitates neuromuscular weakness (eg, Campylobacter and Guillain‐Barr syndrome), or infection that precipitates heart failure (eg, coxsackievirus, myocarditis).
On admission, his temperature was 36.2C, heart rate 91 beats per minute, blood pressure 135/81 mm Hg, respiratory rate 16 breaths per minute, and oxygen saturation 98% on room air. Cardiac exam revealed a regular rhythm without rubs, murmurs, or diastolic gallops. He had no jugular venous distention, and no lower extremity edema. His distal pulses were equal and palpable throughout. Pulmonary exam was notable for decreased breath sounds at both bases without wheezing, rhonchi, or crackles noted. He had no rashes, joint effusions, or jaundice. Abdominal and neurologic examinations were unremarkable.
Diminished breath sounds may suggest atelectasis or pleural effusion; the latter could account for the patient's inspiratory chest pain. A chest radiograph is essential to evaluate this finding further. The physical examination is not suggestive of decompensated heart failure; measurement of serum brain natriuretic peptide level would further exclude that diagnosis.
Laboratory evaluation revealed a leukocytosis of 16,000/L, with 76% polymorphonuclear cells and 12% lymphocytes without eosinophils or band forms; a hematocrit of 38%; and a platelet count of 363,000/L. The patient had a creatinine of 1.6 mg/dL, potassium of 2.7 mEq/L, and a troponin‐I of 1.0 ng/mL (normal 0.40 ng/mL), with the remainder of the routine serum chemistries within normal limits. An electrocardiogram (ECG) showed QS complexes in the anteroseptal leads, and a chest radiograph showed bibasilar consolidations and a left pleural effusion. A ventilation‐perfusion scan of the chest was performed to evaluate for pulmonary embolism, and was interpreted as low probability. Transthoracic echocardiography demonstrated severe left ventricular systolic dysfunction with anterior wall akinesis, and an aneurysmal left ventricle with an apical thrombus. No significant valvular pathology or other structural defects were noted.
The ECG and echocardiogram confirm the history of a large anteroseptal infarction with severe left ventricular dysfunction. Serial troponin testing would be reasonable. However, the absence of any acute ischemic ECG changes, typical angina symptoms, and a relatively normal troponin level all suggest his chest pain does not represent active ischemia. His low abnormal troponin‐I is consistent with slow resolution after a large ischemic event in the recent past, and his anterior wall akinesis is consistent with prior infarction in the territory of his culprit left anterior descending coronary artery.
Although acute cardiac conditions appear less likely, the brisk leukocytosis in a returned traveler prompts consideration of infection. His lung consolidations could represent either new or resolving pneumonia. The complete absence of cough and fever is unusual for pneumonia, yet clinical findings are not as sensitive as chest radiograph for this diagnosis. At this point, typical organisms as well as uncommon pathogens associated with diarrhea or his travel history should be included in the differential.
After 24 hours, the patient was discharged on warfarin to treat the apical thrombus and moxifloxacin for a presumed community‐acquired pneumonia. Eight days after discharge, the patient visited his primary care physician with improving, but not resolved, shortness of breath and pleuritic pain despite completing the 7‐day course of moxifloxacin. A chest radiograph showed a large posterior left basal pleural fluid collection, increased from previous.
In the setting of a recent infection, the symptoms and radiographic findings suggest a complicated parapneumonic effusion or empyema. Failure to drain a previously seeded fluid collection leaves bacterial pathogens susceptible to moxifloxacin on the differential, including Streptococcus pneumoniae, Staphylococcus aureus, Legionella species, and other enterobacteriaciae (eg, Klebsiella pneumoniae).
The indolent course should also prompt consideration of more unusual pathogens, including roundworms (such as Ascaris) or lung flukes (Paragonimus), either of which can cause a lung infection without traditional pneumonia symptoms. Tuberculosis tends to present months (or years) after exposure. Older adults may manifest primary pulmonary tuberculosis with lower lobe infiltrates, consistent with this presentation. However, moxifloxacin is quite active against tuberculosis, and although single drug therapy would not be expected to cure the patient, it would be surprising for him to progress this quickly on moxifloxacin.
In northern Thailand, Burkholderia pseudomallei is a common cause of bacteremic pneumonia. The organism often has high‐level resistance to fluoroquinolones, and may present in a more insidious fashion than other causes of community‐acquired pneumonia. Although infection with B pseudomallei (melioidosis) can occasionally mimic apical pulmonary tuberculosis and may present after a prolonged latent period, it most commonly manifests as an acute pneumonia.
The patient was prescribed 10 days of amoxicillin‐clavulanic acid and clindamycin, and decubitus films were ordered to assess the effusion. These films, obtained 5 days later, showed a persistent pleural effusion. Subsequent ultrasound demonstrated loculated fluid, but a thoracentesis was not performed at that time due to the patient's therapeutic international normalized ratio and dual antiplatelet therapy.
The loculation further suggests a complicated parapneumonic effusion or empyema. Clindamycin adds very little to amoxicillin‐clavulanate as far as coverage of oral anaerobes or common pneumonia pathogens and may add to the risk of antibiotic side effects. A susceptible organism might not clear because of failure to drain this collection; if undertreated bacterial infection is suspected, tube thoracentesis is the established standard of care. However, the protracted course of illness makes untreated pyogenic bacterial infections unlikely.
At this point, the top 2 diagnostic considerations are Paragonimus westermani and B pseudomallei. P westermani is initially ingested, usually from an undercooked freshwater crustacean. Infected patients may experience a brief diarrheal illness, as this patient reported. However, infected patients typically have a brisk peripheral eosinophilia.
Melioidosis is thus the leading concern. Amoxicillin‐clavulanate is active against many strains of B pseudomallei, so the failure of the patient to worsen could be seen as a partial treatment and supports this diagnosis. However, as prolonged therapy is necessary for complete eradication of B pseudomallei, a definitive, culture‐based diagnosis should be established before committing the patient to months of antibiotics.
After completing 10 days of clindamycin and amoxicillin‐clavulanate, the patient reported improvement of his pleuritic pain, and repeat physical exam suggested interval decrease in the size of the effusion. Two days later, the patient began experiencing dysuria that persisted despite 3 days of nitrofurantoin.
Melioidosis can also involve the genitourinary tract. Hematogenous spread of B pseudomallei can seed a number of visceral organs including the bladder, joints, and bones. Men with suspected urinary infection should be evaluated for the possibility of prostatitis, an infection with considerable morbidity that requires extended therapy. This gentleman should have a prostate exam, and blood and urine cultures should be collected if prostatitis is suspected. Empiric antibiotics are not recommended without culture in a patient with complicated urinary tract infection.
Prostate exam was unremarkable. A urine culture grew a gram‐negative rod identified as B pseudomallei. Because B pseudomallei can cause fulminant sepsis, the infectious disease consultant requested that he return for admission, further evaluation, and initiation of intravenous antibiotics. Computed tomography (CT) of the chest, abdomen, and pelvis revealed multiple pulmonary nodules, a persistent left pleural effusion, and a rim‐enhancing hypodensity in the prostate consistent with abscess (Figure 1). Blood and pleural fluid cultures were negative.

Initial treatment for a patient with severe or metastatic B pseudomallei infection requires high‐dose intravenous antibiotic therapy. Ceftazidime, imipenem, and meropenem are the best studied agents for this purpose. Surgical drainage should be considered for the abscess. Following the completion of intensive intravenous therapy, relapse rates are high unless a longer‐term, consolidation therapy is pursued. Trimethoprim‐sulfamethoxazole is the recommended agent.
The patient was treated with high‐dose ceftazidime for 2 weeks, followed by 6 months of high‐dose oral trimethoprim‐sulfamethoxazole. His symptoms resolved, and 7 months after presentation, he continued to feel well.
DISCUSSION
Melioidosis refers to any infection caused by B pseudomallei, a gram‐negative bacillus found in soil and water, most commonly in Southeast Asia and Australia.[1] It is an important cause of pneumonia in endemic regions; in Thailand, the incidence is as high as 12 cases per 100,000 people, and it is the third leading infectious cause of death, following human immunodeficiency virus and tuberculosis.[2] However, it occurs only as an imported infection in the United States and remains an unfamiliar infection for many US medical practitioners. Melioidosis should be considered in patients returning from endemic regions presenting with sepsis, pneumonia, urinary symptoms, or abscesses.
B pseudomallei can be transmitted to humans through exposure to contaminated soil or water via ingestion, inhalation, or percutaneous inoculation.[1] Outbreaks typically occur during the rainy season and after typhoons.[1, 3] Presumably, this patient's exposure to B pseudomallei occurred while hiking and wading in freshwater lakes and waterfalls. Although hospital‐acquired melioidosis has not been reported, and isolation precautions are not necessary, rare cases of disease acquired via laboratory exposure have been reported among US healthcare workers. Clinicians suspecting melioidosis should alert the receiving laboratory.[4]
The treatment course for melioidosis is lengthy and should involve consultation with an infectious disease specialist. B pseudomallei is known to be resistant to penicillin, first‐ and second‐generation cephalosporins, and moxifloxacin. The standard treatment includes 10 to 14 days of intravenous ceftazidime, meropenem, or imipenem, and then trimethoprim‐sulfamethoxazole for 3 to 6 months.[1] Treatment should be guided by culture susceptibility data when available. There are reports of B pseudomallei having different resistance patterns within the same host; clinicians should culture all drained fluid collections and tailor antibiotics to the most resistant strain recovered.[5, 6] Although melioidosis is a life‐threatening infection, previously healthy patients have an excellent prognosis assuming prompt diagnosis and treatment are provided.[3]
After excluding common causes of chest pain, the discussant identified the need to definitively establish a microbiologic diagnosis by obtaining pleural fluid. Although common clinical scenarios can often be treated with guideline‐supported empiric antibiotics, the use of serial courses of empiric antibiotics should be carefully questioned and is generally discouraged. Specific data to prove or disprove the presence of infection should be obtained before exposing a patient to the risks of multiple drugs or prolonged antibiotic therapy, as well as the risks of delayed (or missed) diagnosis. Unfortunately, a complete evaluation was delayed by clinical contraindications to diagnostic thoracentesis, and a definitive diagnosis was reached only after development of more widespread symptoms.
This patient's protean presentation is not surprising given his ultimate diagnosis. B pseudomallei has been termed the great mimicker, as disease presentation and organ involvement can vary from an indolent localized infection to acute severe sepsis.[7] Pneumonia and genitourinary infections are the most common manifestations, although skin infections, bacteremia, septic arthritis, and neurologic disease are also possible.[1, 3] In addition, melioidosis may develop after a lengthy incubation. In a case series, confirmed incubation periods ranged from 1 to 21 days (mean, 9 days); however, cases of chronic (>2 months) infection, mimicking tuberculosis, are estimated to occur in about 12% of cases.[4] B pseudomallei is also capable of causing reactivation disease, similar to tuberculosis. It was referred to as the Vietnamese time bomb when US Vietnam War veterans, exposed to the disease when helicopters aerosolized the bacteria in the soil, developed the disease only after their return to the United States.[8] Fortunately, only a tiny fraction of the quarter‐million soldiers with serologically confirmed exposure to the bacteria ultimately developed disease.
In The Adventure of the Dying Detective, Sherlock Holmes fakes a serious illness characterized by shortness of breath and weakness to trick an adversary into confessing to murder. The abrupt, crippling infection mimicked by Holmes is thought by some to be melioidosis.[9, 10] Conan Doyle's story was published in 1913, a year after melioidosis was first reported in the medical literature, and the exotic, protean infection may well have sparked Doyle's imagination. However, this patient's case of melioidosis proved stranger than fiction in its untimely concomitant development with an MI. Cracking our case required imagination and nimble thinking to avoid a number of cognitive pitfalls. The patient's recent MI anchored reasoning at his initial presentation, and the initial diagnosis of community‐acquired pneumonia raised the danger of premature closure. Reaching the correct diagnosis required an open mind, a detailed travel history, and firm microbiologic evidence. Hospitalists need not be expert in the health risks of travel to specific foreign destinations, but investigating those risks can hasten proper diagnosis and treatment.
TEACHING POINTS
- Melioidosis should be considered in patients returning from endemic regions who present with sepsis, pneumonia, urinary symptoms, or an abscess.
- For patients with a loculated parapneumonic effusion, tube thoracentesis for culture and drainage is the standard of care for diagnosis and treatment.
- Culture identification and antibiotic sensitivities are critical for management of B pseudomallei, because prolonged antibiotic treatment is needed.
Disclosure
Nothing to report.
A 65‐year‐old man suffered a myocardial infarction (MI) while traveling in Thailand. After 7 days of recovery, the patient departed for his home in the United States. He developed substernal, nonexertional, inspiratory chest pain and shortness of breath during his return flight and presented directly to an emergency room after arrival.
Initially, the evaluation should focus on life‐threatening diagnoses and not be distracted by the travel history. The immediate diagnostic concerns are active cardiac ischemia, complications of MI, and pulmonary embolus. Other cardiac causes of dyspnea include ischemic mitral regurgitation, postinfarction pericarditis with or without pericardial effusion, and heart failure. Mechanical complications of infarction, such as left ventricular free wall rupture or rupture of the interventricular septum, can occur in this time frame and are associated with significant morbidity. Pneumothorax may be precipitated by air travel, especially in patients with underlying lung disease. The immobilization associated with long airline flights is a risk factor for thromboembolic disease, which is classically associated with pleuritic chest pain. Inspiratory chest pain is also associated with inflammatory processes involving the pericardium or pleura. If pneumonia, pericarditis, or pleural effusion is present, details of his travel history will become more important in his evaluation.
The patient elaborated that he spent 10 days in Thailand. On the third day of his trip he developed severe chest pain while hiking toward a waterfall in a rural northern district. He was transferred to a large private hospital, where he received a stent in the proximal left anterior descending coronary artery 4 hours after symptom onset. At discharge he was prescribed ticagrelor 90 mg twice daily and daily doses of losartan 50 mg, furosemide 20 mg, spironolactone 12.5 mg, aspirin 81 mg, ivabradine 2.5 mg, and pravastatin 40 mg. He had also been taking doxycycline for malaria prophylaxis since departing the United States.
His past medical history was notable for hypertension and hyperlipidemia. The patient was a lifelong nonsmoker, did not use illicit substances, and consumed no more than 2 alcoholic beverages per day. He denied cough, fevers, chills, diaphoresis, weight loss, recent upper respiratory infection, abdominal pain, hematuria, and nausea. However, he reported exertional dyspnea following his MI and nonbloody diarrhea that occurred a few days prior to his return flight and resolved without intervention.
The remainder of his past medical history confirms that he received appropriate post‐MI care, but does not substantially alter the high priority concerns in his differential diagnosis. Diarrhea may occur in up to 50% of international travelers, and is especially common when returning from Southeast Asia or the Indian subcontinent. Disease processes that may explain diarrhea and subsequent dyspnea include intestinal infections that spread to the lung (eg, ascariasis and Loeffler syndrome), infection that precipitates neuromuscular weakness (eg, Campylobacter and Guillain‐Barr syndrome), or infection that precipitates heart failure (eg, coxsackievirus, myocarditis).
On admission, his temperature was 36.2C, heart rate 91 beats per minute, blood pressure 135/81 mm Hg, respiratory rate 16 breaths per minute, and oxygen saturation 98% on room air. Cardiac exam revealed a regular rhythm without rubs, murmurs, or diastolic gallops. He had no jugular venous distention, and no lower extremity edema. His distal pulses were equal and palpable throughout. Pulmonary exam was notable for decreased breath sounds at both bases without wheezing, rhonchi, or crackles noted. He had no rashes, joint effusions, or jaundice. Abdominal and neurologic examinations were unremarkable.
Diminished breath sounds may suggest atelectasis or pleural effusion; the latter could account for the patient's inspiratory chest pain. A chest radiograph is essential to evaluate this finding further. The physical examination is not suggestive of decompensated heart failure; measurement of serum brain natriuretic peptide level would further exclude that diagnosis.
Laboratory evaluation revealed a leukocytosis of 16,000/L, with 76% polymorphonuclear cells and 12% lymphocytes without eosinophils or band forms; a hematocrit of 38%; and a platelet count of 363,000/L. The patient had a creatinine of 1.6 mg/dL, potassium of 2.7 mEq/L, and a troponin‐I of 1.0 ng/mL (normal 0.40 ng/mL), with the remainder of the routine serum chemistries within normal limits. An electrocardiogram (ECG) showed QS complexes in the anteroseptal leads, and a chest radiograph showed bibasilar consolidations and a left pleural effusion. A ventilation‐perfusion scan of the chest was performed to evaluate for pulmonary embolism, and was interpreted as low probability. Transthoracic echocardiography demonstrated severe left ventricular systolic dysfunction with anterior wall akinesis, and an aneurysmal left ventricle with an apical thrombus. No significant valvular pathology or other structural defects were noted.
The ECG and echocardiogram confirm the history of a large anteroseptal infarction with severe left ventricular dysfunction. Serial troponin testing would be reasonable. However, the absence of any acute ischemic ECG changes, typical angina symptoms, and a relatively normal troponin level all suggest his chest pain does not represent active ischemia. His low abnormal troponin‐I is consistent with slow resolution after a large ischemic event in the recent past, and his anterior wall akinesis is consistent with prior infarction in the territory of his culprit left anterior descending coronary artery.
Although acute cardiac conditions appear less likely, the brisk leukocytosis in a returned traveler prompts consideration of infection. His lung consolidations could represent either new or resolving pneumonia. The complete absence of cough and fever is unusual for pneumonia, yet clinical findings are not as sensitive as chest radiograph for this diagnosis. At this point, typical organisms as well as uncommon pathogens associated with diarrhea or his travel history should be included in the differential.
After 24 hours, the patient was discharged on warfarin to treat the apical thrombus and moxifloxacin for a presumed community‐acquired pneumonia. Eight days after discharge, the patient visited his primary care physician with improving, but not resolved, shortness of breath and pleuritic pain despite completing the 7‐day course of moxifloxacin. A chest radiograph showed a large posterior left basal pleural fluid collection, increased from previous.
In the setting of a recent infection, the symptoms and radiographic findings suggest a complicated parapneumonic effusion or empyema. Failure to drain a previously seeded fluid collection leaves bacterial pathogens susceptible to moxifloxacin on the differential, including Streptococcus pneumoniae, Staphylococcus aureus, Legionella species, and other enterobacteriaciae (eg, Klebsiella pneumoniae).
The indolent course should also prompt consideration of more unusual pathogens, including roundworms (such as Ascaris) or lung flukes (Paragonimus), either of which can cause a lung infection without traditional pneumonia symptoms. Tuberculosis tends to present months (or years) after exposure. Older adults may manifest primary pulmonary tuberculosis with lower lobe infiltrates, consistent with this presentation. However, moxifloxacin is quite active against tuberculosis, and although single drug therapy would not be expected to cure the patient, it would be surprising for him to progress this quickly on moxifloxacin.
In northern Thailand, Burkholderia pseudomallei is a common cause of bacteremic pneumonia. The organism often has high‐level resistance to fluoroquinolones, and may present in a more insidious fashion than other causes of community‐acquired pneumonia. Although infection with B pseudomallei (melioidosis) can occasionally mimic apical pulmonary tuberculosis and may present after a prolonged latent period, it most commonly manifests as an acute pneumonia.
The patient was prescribed 10 days of amoxicillin‐clavulanic acid and clindamycin, and decubitus films were ordered to assess the effusion. These films, obtained 5 days later, showed a persistent pleural effusion. Subsequent ultrasound demonstrated loculated fluid, but a thoracentesis was not performed at that time due to the patient's therapeutic international normalized ratio and dual antiplatelet therapy.
The loculation further suggests a complicated parapneumonic effusion or empyema. Clindamycin adds very little to amoxicillin‐clavulanate as far as coverage of oral anaerobes or common pneumonia pathogens and may add to the risk of antibiotic side effects. A susceptible organism might not clear because of failure to drain this collection; if undertreated bacterial infection is suspected, tube thoracentesis is the established standard of care. However, the protracted course of illness makes untreated pyogenic bacterial infections unlikely.
At this point, the top 2 diagnostic considerations are Paragonimus westermani and B pseudomallei. P westermani is initially ingested, usually from an undercooked freshwater crustacean. Infected patients may experience a brief diarrheal illness, as this patient reported. However, infected patients typically have a brisk peripheral eosinophilia.
Melioidosis is thus the leading concern. Amoxicillin‐clavulanate is active against many strains of B pseudomallei, so the failure of the patient to worsen could be seen as a partial treatment and supports this diagnosis. However, as prolonged therapy is necessary for complete eradication of B pseudomallei, a definitive, culture‐based diagnosis should be established before committing the patient to months of antibiotics.
After completing 10 days of clindamycin and amoxicillin‐clavulanate, the patient reported improvement of his pleuritic pain, and repeat physical exam suggested interval decrease in the size of the effusion. Two days later, the patient began experiencing dysuria that persisted despite 3 days of nitrofurantoin.
Melioidosis can also involve the genitourinary tract. Hematogenous spread of B pseudomallei can seed a number of visceral organs including the bladder, joints, and bones. Men with suspected urinary infection should be evaluated for the possibility of prostatitis, an infection with considerable morbidity that requires extended therapy. This gentleman should have a prostate exam, and blood and urine cultures should be collected if prostatitis is suspected. Empiric antibiotics are not recommended without culture in a patient with complicated urinary tract infection.
Prostate exam was unremarkable. A urine culture grew a gram‐negative rod identified as B pseudomallei. Because B pseudomallei can cause fulminant sepsis, the infectious disease consultant requested that he return for admission, further evaluation, and initiation of intravenous antibiotics. Computed tomography (CT) of the chest, abdomen, and pelvis revealed multiple pulmonary nodules, a persistent left pleural effusion, and a rim‐enhancing hypodensity in the prostate consistent with abscess (Figure 1). Blood and pleural fluid cultures were negative.

Initial treatment for a patient with severe or metastatic B pseudomallei infection requires high‐dose intravenous antibiotic therapy. Ceftazidime, imipenem, and meropenem are the best studied agents for this purpose. Surgical drainage should be considered for the abscess. Following the completion of intensive intravenous therapy, relapse rates are high unless a longer‐term, consolidation therapy is pursued. Trimethoprim‐sulfamethoxazole is the recommended agent.
The patient was treated with high‐dose ceftazidime for 2 weeks, followed by 6 months of high‐dose oral trimethoprim‐sulfamethoxazole. His symptoms resolved, and 7 months after presentation, he continued to feel well.
DISCUSSION
Melioidosis refers to any infection caused by B pseudomallei, a gram‐negative bacillus found in soil and water, most commonly in Southeast Asia and Australia.[1] It is an important cause of pneumonia in endemic regions; in Thailand, the incidence is as high as 12 cases per 100,000 people, and it is the third leading infectious cause of death, following human immunodeficiency virus and tuberculosis.[2] However, it occurs only as an imported infection in the United States and remains an unfamiliar infection for many US medical practitioners. Melioidosis should be considered in patients returning from endemic regions presenting with sepsis, pneumonia, urinary symptoms, or abscesses.
B pseudomallei can be transmitted to humans through exposure to contaminated soil or water via ingestion, inhalation, or percutaneous inoculation.[1] Outbreaks typically occur during the rainy season and after typhoons.[1, 3] Presumably, this patient's exposure to B pseudomallei occurred while hiking and wading in freshwater lakes and waterfalls. Although hospital‐acquired melioidosis has not been reported, and isolation precautions are not necessary, rare cases of disease acquired via laboratory exposure have been reported among US healthcare workers. Clinicians suspecting melioidosis should alert the receiving laboratory.[4]
The treatment course for melioidosis is lengthy and should involve consultation with an infectious disease specialist. B pseudomallei is known to be resistant to penicillin, first‐ and second‐generation cephalosporins, and moxifloxacin. The standard treatment includes 10 to 14 days of intravenous ceftazidime, meropenem, or imipenem, and then trimethoprim‐sulfamethoxazole for 3 to 6 months.[1] Treatment should be guided by culture susceptibility data when available. There are reports of B pseudomallei having different resistance patterns within the same host; clinicians should culture all drained fluid collections and tailor antibiotics to the most resistant strain recovered.[5, 6] Although melioidosis is a life‐threatening infection, previously healthy patients have an excellent prognosis assuming prompt diagnosis and treatment are provided.[3]
After excluding common causes of chest pain, the discussant identified the need to definitively establish a microbiologic diagnosis by obtaining pleural fluid. Although common clinical scenarios can often be treated with guideline‐supported empiric antibiotics, the use of serial courses of empiric antibiotics should be carefully questioned and is generally discouraged. Specific data to prove or disprove the presence of infection should be obtained before exposing a patient to the risks of multiple drugs or prolonged antibiotic therapy, as well as the risks of delayed (or missed) diagnosis. Unfortunately, a complete evaluation was delayed by clinical contraindications to diagnostic thoracentesis, and a definitive diagnosis was reached only after development of more widespread symptoms.
This patient's protean presentation is not surprising given his ultimate diagnosis. B pseudomallei has been termed the great mimicker, as disease presentation and organ involvement can vary from an indolent localized infection to acute severe sepsis.[7] Pneumonia and genitourinary infections are the most common manifestations, although skin infections, bacteremia, septic arthritis, and neurologic disease are also possible.[1, 3] In addition, melioidosis may develop after a lengthy incubation. In a case series, confirmed incubation periods ranged from 1 to 21 days (mean, 9 days); however, cases of chronic (>2 months) infection, mimicking tuberculosis, are estimated to occur in about 12% of cases.[4] B pseudomallei is also capable of causing reactivation disease, similar to tuberculosis. It was referred to as the Vietnamese time bomb when US Vietnam War veterans, exposed to the disease when helicopters aerosolized the bacteria in the soil, developed the disease only after their return to the United States.[8] Fortunately, only a tiny fraction of the quarter‐million soldiers with serologically confirmed exposure to the bacteria ultimately developed disease.
In The Adventure of the Dying Detective, Sherlock Holmes fakes a serious illness characterized by shortness of breath and weakness to trick an adversary into confessing to murder. The abrupt, crippling infection mimicked by Holmes is thought by some to be melioidosis.[9, 10] Conan Doyle's story was published in 1913, a year after melioidosis was first reported in the medical literature, and the exotic, protean infection may well have sparked Doyle's imagination. However, this patient's case of melioidosis proved stranger than fiction in its untimely concomitant development with an MI. Cracking our case required imagination and nimble thinking to avoid a number of cognitive pitfalls. The patient's recent MI anchored reasoning at his initial presentation, and the initial diagnosis of community‐acquired pneumonia raised the danger of premature closure. Reaching the correct diagnosis required an open mind, a detailed travel history, and firm microbiologic evidence. Hospitalists need not be expert in the health risks of travel to specific foreign destinations, but investigating those risks can hasten proper diagnosis and treatment.
TEACHING POINTS
- Melioidosis should be considered in patients returning from endemic regions who present with sepsis, pneumonia, urinary symptoms, or an abscess.
- For patients with a loculated parapneumonic effusion, tube thoracentesis for culture and drainage is the standard of care for diagnosis and treatment.
- Culture identification and antibiotic sensitivities are critical for management of B pseudomallei, because prolonged antibiotic treatment is needed.
Disclosure
Nothing to report.
- , , . Melioidosis. N Engl J Med. 2012;367(11):1035–1044.
- , , , et al. Increasing incidence of human melioidosis in Northeast Thailand. Am J Trop Med Hyg. 2010;82(6):1113–1117.
- , , . The epidemiology and clinical spectrum of melioidosis: 540 cases from the 20 year Darwin prospective study. PLoS Negl Trop Dis. 2010;4(11):e900.
- , , , et al. Management of accidental laboratory exposure to Burkholderia pseudomallei and B. mallei. Emerg Infect Dis. 2008;14(7):e2.
- , , . Variations in ceftazidime and amoxicillin‐clavulanate susceptibilities within a clonal infection of Burkholderia pseudomallei. J Clin Microbiol. 2009;47(5):1556–1558.
- , , , et al. Within‐host evolution of Burkholderia pseudomallei in four cases of acute melioidosis. PLoS Pathog. 2010;6(1):e1000725.
- , , , , . Melioidosis: insights into the pathogenicity of Burkholderia pseudomallei. Nat Rev Microbiol. 2006;4(4):272–282.
- , . Melioidosis. In: Dembeck ZF, ed. Medical Aspects of Biological Warfare. 2nd ed. Washington, DC: Office of the Surgeon General; 2007:146–166.
- . Sherlock Holmes and a biological weapon. J R Soc Med. 2002;95(2):101–103.
- . Sherlock Holmes and tropical medicine: a centennial appraisal. Am J Trop Med Hyg. 1994;50:99–101.
- , , . Melioidosis. N Engl J Med. 2012;367(11):1035–1044.
- , , , et al. Increasing incidence of human melioidosis in Northeast Thailand. Am J Trop Med Hyg. 2010;82(6):1113–1117.
- , , . The epidemiology and clinical spectrum of melioidosis: 540 cases from the 20 year Darwin prospective study. PLoS Negl Trop Dis. 2010;4(11):e900.
- , , , et al. Management of accidental laboratory exposure to Burkholderia pseudomallei and B. mallei. Emerg Infect Dis. 2008;14(7):e2.
- , , . Variations in ceftazidime and amoxicillin‐clavulanate susceptibilities within a clonal infection of Burkholderia pseudomallei. J Clin Microbiol. 2009;47(5):1556–1558.
- , , , et al. Within‐host evolution of Burkholderia pseudomallei in four cases of acute melioidosis. PLoS Pathog. 2010;6(1):e1000725.
- , , , , . Melioidosis: insights into the pathogenicity of Burkholderia pseudomallei. Nat Rev Microbiol. 2006;4(4):272–282.
- , . Melioidosis. In: Dembeck ZF, ed. Medical Aspects of Biological Warfare. 2nd ed. Washington, DC: Office of the Surgeon General; 2007:146–166.
- . Sherlock Holmes and a biological weapon. J R Soc Med. 2002;95(2):101–103.
- . Sherlock Holmes and tropical medicine: a centennial appraisal. Am J Trop Med Hyg. 1994;50:99–101.