User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
nav[contains(@class, 'nav-ce-stack nav-ce-stack__large-screen')]
header[@id='header']
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
Few Women Know Uterine Fibroid Risk, Treatment Options
Most women (72%) are not aware they are at risk for developing uterine fibroids, though up to 77% of women will develop them in their lifetime, results of a new survey indicate.
Data from The Harris Poll, conducted on behalf of the Society of Interventional Radiology, also found that 17% of women mistakenly think a hysterectomy is the only treatment option, including more than one in four women (27%) who are between the ages of 18 and 34. Results were shared in a press release. The survey included 1,122 US women, some who have been diagnosed with uterine fibroids.
Fibroids may not cause symptoms for some, but some women may have heavy, prolonged, debilitating bleeding. Some women experience pelvic pain, a diminished sex life, and declining energy. However, the growths do not spread to other body regions and typically are not dangerous.
Hysterectomy Is Only One Option
Among the women in the survey who had been diagnosed with fibroids, 53% were presented the option of hysterectomy and 20% were told about other, less-invasive options, including over-the-counter NSAIDs (19%); uterine fibroid embolization (UFE) (17%); oral contraceptives (17%); and endometrial ablation (17%).
“Women need to be informed about the complete range of options available for treating their uterine fibroids, not just the surgical options as is most commonly done by gynecologists,” John C. Lipman, MD, founder and medical director of the Atlanta Fibroid Center in Smyrna, Georgia, said in the press release.
The survey also found that:
- More than half of women ages 18-34 (56%) and women ages 35-44 (51%) were either not familiar with uterine fibroids or never heard of them.
- Awareness was particularly low among Hispanic women, as 50% of Hispanic women say they’ve never heard of or aren’t familiar with the condition, compared with 37% of Black women who answered that way.
- More than one third (36%) of Black women and 22% of Hispanic women mistakenly think they are not at risk for developing fibroids, yet research has shown that uterine fibroids are three times more common in Black women and two times more common in Hispanic women than in White women.
For this study, the full sample data is accurate to within +/– 3.2 percentage points using a 95% confidence level. The data are part of the report “The Fibroid Fix: What Women Need to Know,” published on July 9 by the Society of Interventional Radiology.
Linda Fan, MD, chief of gynecology at Yale University in New Haven, Connecticut, said she is not surprised by those numbers. She says many patients are referred to her department who have not been given the full array of medical options for their fibroids or have not had thorough discussions with their providers, such as whether they want to preserve their fertility, or how they feel about an incision, undergoing anesthesia, or having their uterus removed.
Sometimes the hysterectomy choice is clear, she said — for instance, if there are indications of the rare cancer leiomyosarcoma, or if a postmenopausal woman has rapid growth of fibroids or heavy bleeding. Fibroids should not start growing after menopause, she said.
Additional options include radiofrequency ablation, performed while a patient is under anesthesia, by laparoscopy or hysteroscopy. The procedure uses ultrasound to watch a probe as it shrinks the fibroids with heat.
Currently, if a woman wants large fibroids removed and wants to keep her fertility options open, Dr. Fan says, myomectomy or medication are best “because we have the most information or data on (those options).”
When treating patients who don’t prioritize fertility, she said, UFE is a good option that doesn’t need incisions or anesthesia. But patients sometimes require a lot of pain medication afterward, Dr. Fan said. With radiofrequency ablation, specifically the Acessa and Sonata procedures, she said, “patients don’t experience a lot of pain after the procedure because the shrinking happens when they’re asleep under anesthesia.”
Uterine Fibroid Embolization a Nonsurgical Option
The report describes how UFE works but the Harris Poll showed that 60% of women who have heard of UFE did not hear about it first from a healthcare provider.
“UFE is a nonsurgical treatment, performed by interventional radiologists, that has been proven to significantly reduce heavy menstrual bleeding, relieve uterine pain, and improve energy levels,” the authors write. “Through a tiny incision in the wrist or thigh, a catheter is guided via imaging to the vessels leading to the fibroids. Through this catheter, small clear particles are injected to block the blood flow leading to the fibroids causing them to shrink and disappear.”
After UFE, most women leave the hospital the day of or the day after treatment, according to the report authors, who add that many patients also report they can resume normal activity in about 2 weeks, more quickly than with surgical treatments.
In some cases, watchful waiting will be the best option, the report notes, and that may require repeated checkups and scans.
Dr. Lipman is an adviser on The Fibroid Fix report.
Most women (72%) are not aware they are at risk for developing uterine fibroids, though up to 77% of women will develop them in their lifetime, results of a new survey indicate.
Data from The Harris Poll, conducted on behalf of the Society of Interventional Radiology, also found that 17% of women mistakenly think a hysterectomy is the only treatment option, including more than one in four women (27%) who are between the ages of 18 and 34. Results were shared in a press release. The survey included 1,122 US women, some who have been diagnosed with uterine fibroids.
Fibroids may not cause symptoms for some, but some women may have heavy, prolonged, debilitating bleeding. Some women experience pelvic pain, a diminished sex life, and declining energy. However, the growths do not spread to other body regions and typically are not dangerous.
Hysterectomy Is Only One Option
Among the women in the survey who had been diagnosed with fibroids, 53% were presented the option of hysterectomy and 20% were told about other, less-invasive options, including over-the-counter NSAIDs (19%); uterine fibroid embolization (UFE) (17%); oral contraceptives (17%); and endometrial ablation (17%).
“Women need to be informed about the complete range of options available for treating their uterine fibroids, not just the surgical options as is most commonly done by gynecologists,” John C. Lipman, MD, founder and medical director of the Atlanta Fibroid Center in Smyrna, Georgia, said in the press release.
The survey also found that:
- More than half of women ages 18-34 (56%) and women ages 35-44 (51%) were either not familiar with uterine fibroids or never heard of them.
- Awareness was particularly low among Hispanic women, as 50% of Hispanic women say they’ve never heard of or aren’t familiar with the condition, compared with 37% of Black women who answered that way.
- More than one third (36%) of Black women and 22% of Hispanic women mistakenly think they are not at risk for developing fibroids, yet research has shown that uterine fibroids are three times more common in Black women and two times more common in Hispanic women than in White women.
For this study, the full sample data is accurate to within +/– 3.2 percentage points using a 95% confidence level. The data are part of the report “The Fibroid Fix: What Women Need to Know,” published on July 9 by the Society of Interventional Radiology.
Linda Fan, MD, chief of gynecology at Yale University in New Haven, Connecticut, said she is not surprised by those numbers. She says many patients are referred to her department who have not been given the full array of medical options for their fibroids or have not had thorough discussions with their providers, such as whether they want to preserve their fertility, or how they feel about an incision, undergoing anesthesia, or having their uterus removed.
Sometimes the hysterectomy choice is clear, she said — for instance, if there are indications of the rare cancer leiomyosarcoma, or if a postmenopausal woman has rapid growth of fibroids or heavy bleeding. Fibroids should not start growing after menopause, she said.
Additional options include radiofrequency ablation, performed while a patient is under anesthesia, by laparoscopy or hysteroscopy. The procedure uses ultrasound to watch a probe as it shrinks the fibroids with heat.
Currently, if a woman wants large fibroids removed and wants to keep her fertility options open, Dr. Fan says, myomectomy or medication are best “because we have the most information or data on (those options).”
When treating patients who don’t prioritize fertility, she said, UFE is a good option that doesn’t need incisions or anesthesia. But patients sometimes require a lot of pain medication afterward, Dr. Fan said. With radiofrequency ablation, specifically the Acessa and Sonata procedures, she said, “patients don’t experience a lot of pain after the procedure because the shrinking happens when they’re asleep under anesthesia.”
Uterine Fibroid Embolization a Nonsurgical Option
The report describes how UFE works but the Harris Poll showed that 60% of women who have heard of UFE did not hear about it first from a healthcare provider.
“UFE is a nonsurgical treatment, performed by interventional radiologists, that has been proven to significantly reduce heavy menstrual bleeding, relieve uterine pain, and improve energy levels,” the authors write. “Through a tiny incision in the wrist or thigh, a catheter is guided via imaging to the vessels leading to the fibroids. Through this catheter, small clear particles are injected to block the blood flow leading to the fibroids causing them to shrink and disappear.”
After UFE, most women leave the hospital the day of or the day after treatment, according to the report authors, who add that many patients also report they can resume normal activity in about 2 weeks, more quickly than with surgical treatments.
In some cases, watchful waiting will be the best option, the report notes, and that may require repeated checkups and scans.
Dr. Lipman is an adviser on The Fibroid Fix report.
Most women (72%) are not aware they are at risk for developing uterine fibroids, though up to 77% of women will develop them in their lifetime, results of a new survey indicate.
Data from The Harris Poll, conducted on behalf of the Society of Interventional Radiology, also found that 17% of women mistakenly think a hysterectomy is the only treatment option, including more than one in four women (27%) who are between the ages of 18 and 34. Results were shared in a press release. The survey included 1,122 US women, some who have been diagnosed with uterine fibroids.
Fibroids may not cause symptoms for some, but some women may have heavy, prolonged, debilitating bleeding. Some women experience pelvic pain, a diminished sex life, and declining energy. However, the growths do not spread to other body regions and typically are not dangerous.
Hysterectomy Is Only One Option
Among the women in the survey who had been diagnosed with fibroids, 53% were presented the option of hysterectomy and 20% were told about other, less-invasive options, including over-the-counter NSAIDs (19%); uterine fibroid embolization (UFE) (17%); oral contraceptives (17%); and endometrial ablation (17%).
“Women need to be informed about the complete range of options available for treating their uterine fibroids, not just the surgical options as is most commonly done by gynecologists,” John C. Lipman, MD, founder and medical director of the Atlanta Fibroid Center in Smyrna, Georgia, said in the press release.
The survey also found that:
- More than half of women ages 18-34 (56%) and women ages 35-44 (51%) were either not familiar with uterine fibroids or never heard of them.
- Awareness was particularly low among Hispanic women, as 50% of Hispanic women say they’ve never heard of or aren’t familiar with the condition, compared with 37% of Black women who answered that way.
- More than one third (36%) of Black women and 22% of Hispanic women mistakenly think they are not at risk for developing fibroids, yet research has shown that uterine fibroids are three times more common in Black women and two times more common in Hispanic women than in White women.
For this study, the full sample data is accurate to within +/– 3.2 percentage points using a 95% confidence level. The data are part of the report “The Fibroid Fix: What Women Need to Know,” published on July 9 by the Society of Interventional Radiology.
Linda Fan, MD, chief of gynecology at Yale University in New Haven, Connecticut, said she is not surprised by those numbers. She says many patients are referred to her department who have not been given the full array of medical options for their fibroids or have not had thorough discussions with their providers, such as whether they want to preserve their fertility, or how they feel about an incision, undergoing anesthesia, or having their uterus removed.
Sometimes the hysterectomy choice is clear, she said — for instance, if there are indications of the rare cancer leiomyosarcoma, or if a postmenopausal woman has rapid growth of fibroids or heavy bleeding. Fibroids should not start growing after menopause, she said.
Additional options include radiofrequency ablation, performed while a patient is under anesthesia, by laparoscopy or hysteroscopy. The procedure uses ultrasound to watch a probe as it shrinks the fibroids with heat.
Currently, if a woman wants large fibroids removed and wants to keep her fertility options open, Dr. Fan says, myomectomy or medication are best “because we have the most information or data on (those options).”
When treating patients who don’t prioritize fertility, she said, UFE is a good option that doesn’t need incisions or anesthesia. But patients sometimes require a lot of pain medication afterward, Dr. Fan said. With radiofrequency ablation, specifically the Acessa and Sonata procedures, she said, “patients don’t experience a lot of pain after the procedure because the shrinking happens when they’re asleep under anesthesia.”
Uterine Fibroid Embolization a Nonsurgical Option
The report describes how UFE works but the Harris Poll showed that 60% of women who have heard of UFE did not hear about it first from a healthcare provider.
“UFE is a nonsurgical treatment, performed by interventional radiologists, that has been proven to significantly reduce heavy menstrual bleeding, relieve uterine pain, and improve energy levels,” the authors write. “Through a tiny incision in the wrist or thigh, a catheter is guided via imaging to the vessels leading to the fibroids. Through this catheter, small clear particles are injected to block the blood flow leading to the fibroids causing them to shrink and disappear.”
After UFE, most women leave the hospital the day of or the day after treatment, according to the report authors, who add that many patients also report they can resume normal activity in about 2 weeks, more quickly than with surgical treatments.
In some cases, watchful waiting will be the best option, the report notes, and that may require repeated checkups and scans.
Dr. Lipman is an adviser on The Fibroid Fix report.
Managing Cancer in Pregnancy: Improvements and Considerations
Introduction: Tremendous Progress on Cancer Extends to Cancer in Pregnancy
The biomedical research enterprise that took shape in the United States after World War II has had numerous positive effects, including significant progress made during the past 75-plus years in the diagnosis, prevention, and treatment of cancer.
President Franklin D. Roosevelt’s 1944 request of Dr. Vannevar Bush, director of the then Office of Scientific Research and Development, to organize a program that would advance and apply scientific knowledge for times of peace — just as it been advanced and applied in times of war — culminated in a historic report, Science – The Endless Frontier. Presented in 1945 to President Harry S. Truman, this report helped fuel decades of broad, bold, and coordinated government-sponsored biomedical research aimed at addressing disease and improving the health of the American people (National Science Foundation, 1945).
Discoveries made from research in basic and translational sciences deepened our knowledge of the cellular and molecular underpinnings of cancer, leading to advances in chemotherapy, radiotherapy, and other treatment approaches as well as continual refinements in their application. Similarly, our diagnostic armamentarium has significantly improved.
As a result, we have reduced both the incidence and mortality of cancer. Today, some cancers can be prevented. Others can be reversed or put in remission. Granted, progress has been variable, with some cancers such as ovarian cancer still having relatively low survival rates. Much more needs to be done. Overall, however, the positive effects of the U.S. biomedical research enterprise on cancer are evident. According to the National Cancer Institute’s most recent report on the status of cancer, death rates from cancer fell 1.9% per year on average in females from 2015 to 2019 (Cancer. 2022 Oct 22. doi: 10.1002/cncr.34479).
It is not only patients whose cancer occurs outside of pregnancy who have benefited. When treatment is appropriately selected and timing considerations are made, patients whose cancer is diagnosed during pregnancy — and their children — can have good outcomes.
To explain how the management of cancer in pregnancy has improved, we have invited Gautam G. Rao, MD, gynecologic oncologist and associate professor of obstetrics, gynecology, and reproductive sciences at the University of Maryland School of Medicine, to write this installment of the Master Class in Obstetrics. As Dr. Rao explains, radiation is not as dangerous to the fetus as once thought, and the safety of many chemotherapeutic regimens in pregnancy has been documented. Obstetricians can and should counsel patients, he explains, about the likelihood of good maternal and fetal outcomes.
E. Albert Reece, MD, PhD, MBA, a maternal-fetal medicine specialist, is dean emeritus of the University of Maryland School of Medicine, former university executive vice president; currently the endowed professor and director of the Center for Advanced Research Training and Innovation (CARTI), and senior scientist in the Center for Birth Defects Research. Dr. Reece reported no relevant disclosures. He is the medical editor of this column. Contact him at [email protected].
Managing Cancer in Pregnancy
Cancer can cause fear and distress for any patient, but when cancer is diagnosed during pregnancy, an expectant mother fears not only for her own health but for the health of her unborn child. Fortunately, ob.gyn.s and multidisciplinary teams have good reason to reassure patients about the likelihood of good outcomes.
Cancer treatment in pregnancy has improved with advancements in imaging and chemotherapy, and while maternal and fetal outcomes of prenatal cancer treatment are not well reported, evidence acquired in recent years from case series and retrospective studies shows that most imaging studies and procedural diagnostic tests – and many treatments – can be performed safely in pregnancy.
Decades ago, we avoided CT scans during pregnancy because of concerns about radiation exposure to the fetus, leaving some patients without an accurate staging of their cancer. Today, we have evidence that a CT scan is generally safe in pregnancy. Similarly, the safety of many chemotherapeutic regimens in pregnancy has been documented in recent decades,and the use of chemotherapy during pregnancy has increased progressively. Radiation is also commonly utilized in the management of cancers that may occur during pregnancy, such as breast cancer.1
Considerations of timing are often central to decision-making; chemotherapy and radiotherapy are generally avoided in the first trimester to prevent structural fetal anomalies, for instance, and delaying cancer treatment is often warranted when the patient is a few weeks away from delivery. On occasion, iatrogenic preterm birth is considered when the risks to the mother of delaying a necessary cancer treatment outweigh the risks to the fetus of prematurity.1
Pregnancy termination is rarely indicated, however, and information gathered over the past 2 decades suggests that fetal and placental metastases are rare.1 There is broad agreement that prenatal treatment of cancer in pregnancy should adhere as much as possible to protocols and guidelines for nonpregnant patients and that treatment delays driven by fear of fetal anomalies and miscarriage are unnecessary.
Cancer Incidence, Use of Diagnostic Imaging
Data on the incidence of cancer in pregnancy comes from population-based cancer registries, and unfortunately, these data are not standardized and are often incomplete. Many studies include cancer diagnosed up to 1 year after pregnancy, and some include preinvasive disease. Estimates therefore vary considerably (see Table 1 for a sampling of estimates incidences.)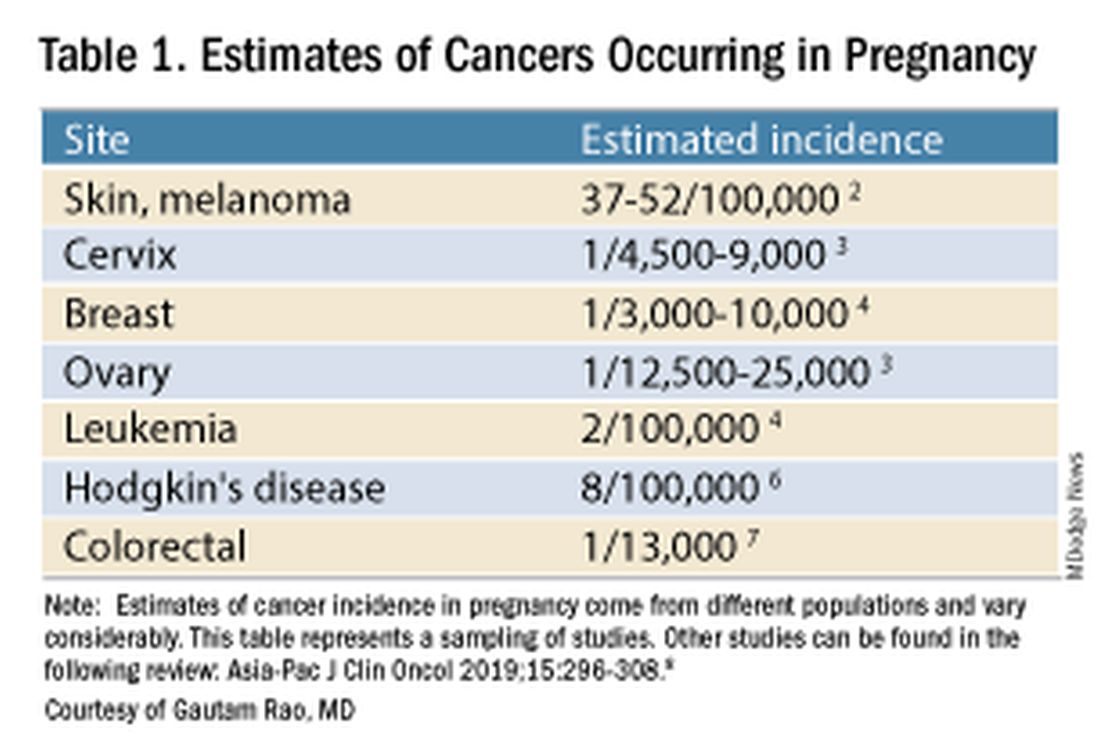
It has been reported, and often cited in the literature, that invasive malignancy complicates one in 1,000 pregnancies and that the incidence of cancer in pregnancy (invasive and noninvasive malignancies) has been rising over time.8 Increasing maternal age is believed to be playing a role in this rise; as women delay childbearing, they enter the age range in which some cancers become more common. Additionally, improvements in screening and diagnostics have led to earlier cancer detection. The incidence of ovarian neoplasms found during pregnancy has increased, for instance, with the routine use of diagnostic ultrasound in pregnancy.1
Among the studies showing an increased incidence of pregnancy-associated cancer is a population-based study in Australia, which found that from 1994 to 2007 the crude incidence of pregnancy-associated cancer increased from 112.3 to 191.5 per 100,000 pregnancies (P < .001).9 A cohort study in the United States documented an increase in incidence from 75.0 per 100,000 pregnancies in 2002 to 138.5 per 100,000 pregnancies in 2012.10
Overall, the literature shows us that the skin, cervix, and breast are also common sites for malignancy during pregnancy.1 According to a 2022 review, breast cancer during pregnancy is less often hormone receptor–positive and more frequently triple negative compared with age-matched controls.11 The frequencies of other pregnancy-associated cancers appear overall to be similar to that of cancer occurring in all women across their reproductive years.1
Too often, diagnosis is delayed because cancer symptoms can be masked by or can mimic normal physiological changes in pregnancy. For instance, breast cancer can be difficult to diagnose during pregnancy and lactation due to anatomic changes in the breast parenchyma. Several studies published in the 1990s showed that breast cancer presents at a more advanced stage in pregnant patients than in nonpregnant patients because of this delay.1 Skin changes suggestive of melanoma can be attributed to hyperpigmentation of pregnancy, for instance. Several observational studies have suggested that thicker melanomas found in pregnancy may be because of delayed diagnosis.8
It is important that we thoroughly investigate signs and symptoms suggestive of a malignancy and not automatically attribute these symptoms to the pregnancy itself. Cervical biopsy of a mass or lesion suspicious for cervical cancer can be done safely during pregnancy and should not be delayed or deferred.
Fetal radiation exposure from radiologic examinations has long been a concern, but we know today that while the imaging modality should be chosen to minimize fetal radiation exposure, CT scans and even PET scans should be performed if these exams are deemed best for evaluation. Embryonic exposure to a dose of less than 50 mGy is rarely if at all associated with fetal malformations or miscarriage and a radiation dose of 100 mGy may be considered a floor for consideration of therapeutic termination of pregnancy.1,8
CT exams are associated with a fetal dose far less than 50 mGy (see Table 2 for radiation doses).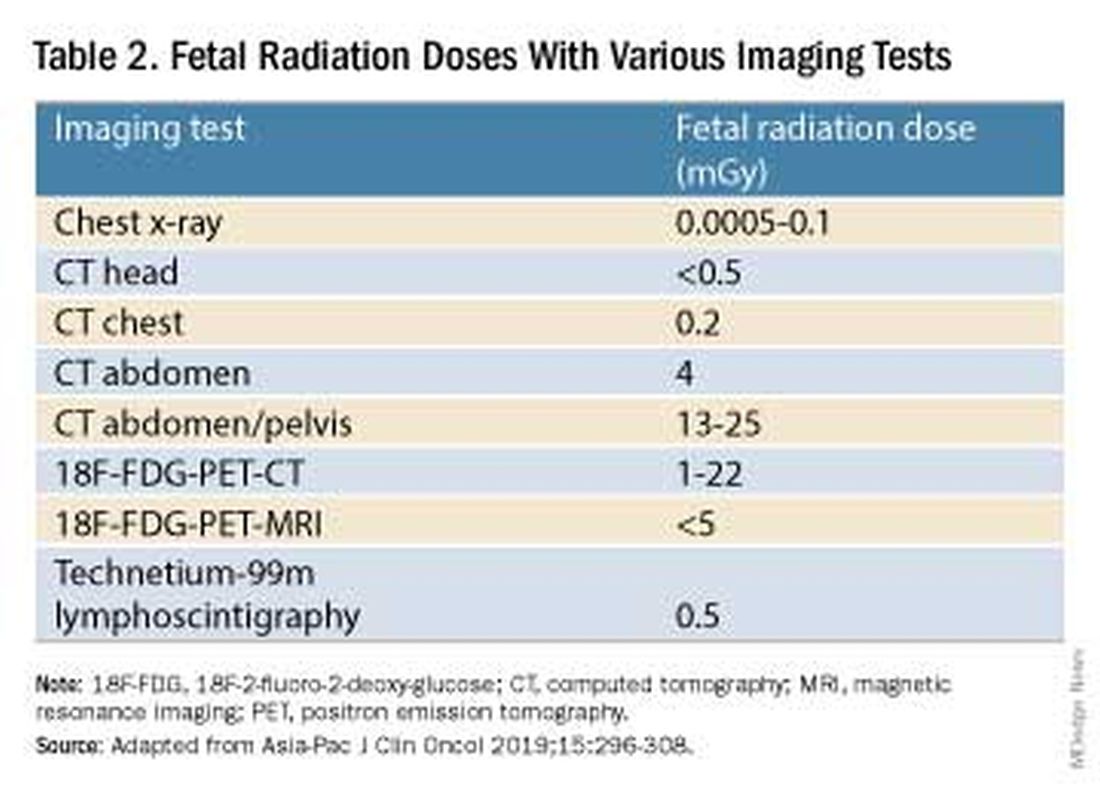
Magnetic resonance imaging with a magnet strength of 3 Tesla or less in any trimester is not associated with an increased risk of harm to the fetus or in early childhood, but the contrast agent gadolinium should be avoided in pregnancy as it has been associated with an increased risk of stillbirth, neonatal death, and childhood inflammatory, rheumatologic, and infiltrative skin lesions.1,8,12
Chemotherapy, Surgery, and Radiation in Pregnancy
The management of cancer during pregnancy requires a multidisciplinary team including medical, gynecologic, or radiation oncologists, and maternal-fetal medicine specialists (Figure 1). Prematurity and low birth weight are frequent complications for fetuses exposed to chemotherapy, although there is some uncertainty as to whether the treatment is causative. However, congenital anomalies no longer are a major concern, provided that drugs are appropriately selected and that fetal exposure occurs during the second or third trimester.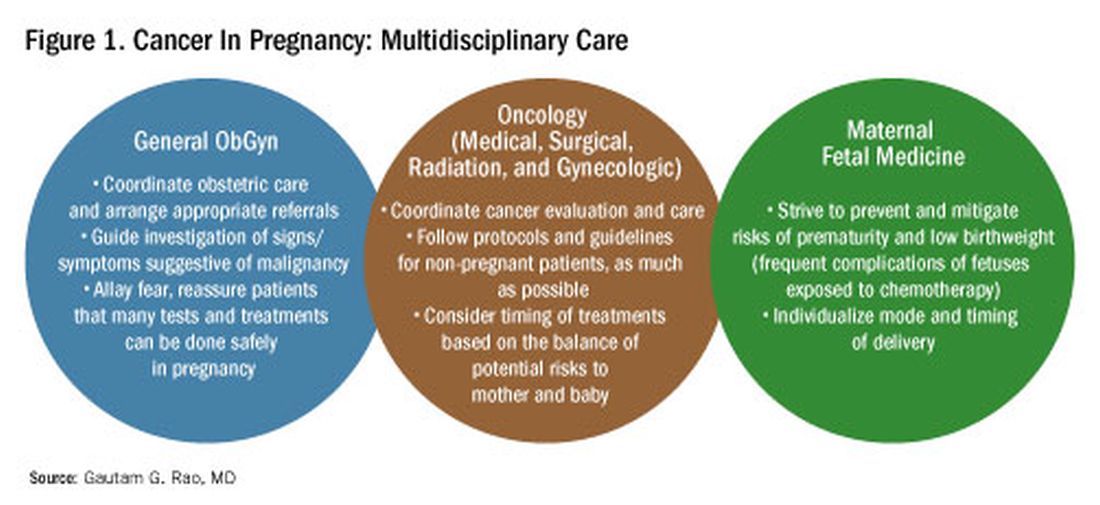
For instance, alkylating agents including cisplatin (an important drug in the management of gynecologic malignancies) have been associated with congenital anomalies in the first trimester but not in the second and third trimesters, and a variety of antimetabolites — excluding methotrexate and aminopterin — similarly have been shown to be relatively safe when used after the first trimester.1
Small studies have shown no long-term effects of chemotherapy exposure on postnatal growth and long-term neurologic/neurocognitive function,1 but this is an area that needs more research.
Also in need of investigation is the safety of newer agents in pregnancy. Data are limited on the use of new targeted treatments, monoclonal antibodies, and immunotherapies in pregnancy and their effects on the fetus, with current knowledge coming mainly from single case reports.13
Until more is learned — a challenge given that pregnant women are generally excluded from clinical trials — management teams are generally postponing use of these therapies until after delivery. Considering the pace of new developments revolutionizing cancer treatment, this topic will likely get more complex and confusing before we begin acquiring sufficient knowledge.
The timing of surgery for malignancy in pregnancy is similarly based on the balance of maternal and fetal risks, including the risk of maternal disease progression, the risk of preterm delivery, and the prevention of fetal metastases. In general, the safest time is the second trimester.
Maternal surgery in the third trimester may be associated with a risk of premature labor and altered uteroplacental perfusion. A 2005 systematic review of 12,452 women who underwent nonobstetric surgery during pregnancy provides some reassurance, however; compared with the general obstetric population, there was no increase in the rate of miscarriage or major birth defects.14
Radiotherapy used to be contraindicated in pregnancy but many experts today believe it can be safely utilized provided the uterus is out of field and is protected from scattered radiation. The head, neck, and breast, for instance, can be treated with newer radiotherapies, including stereotactic ablative radiation therapy.8 Patients with advanced cervical cancer often receive chemotherapy during pregnancy to slow metastatic growth followed by definitive treatment with postpartum radiation or surgery.
More research is needed, but available data on maternal outcomes are encouraging. For instance, there appear to be no significant differences in short- and long-term complications or survival between women who are pregnant and nonpregnant when treated for invasive cervical cancer.8 Similarly, while earlier studies of breast cancer diagnosed during pregnancy suggested a poor prognosis, data now show similar prognoses for pregnant and nonpregnant patients when controlled for stage.1
Dr. Rao is a gynecologic oncologist and associate professor of obstetrics, gynecology, and reproductive sciences at the University of Maryland School of Medicine, Baltimore. He reported no relevant disclosures.
References
1. Rao GG. Chapter 42. Clinical Obstetrics: The Fetus & Mother, 4th ed. Reece EA et al. (eds): 2021.
2. Bannister-Tyrrell M et al. Aust N Z J Obstet Gynaecol. 2014;55:116-122.
3. Oehler MK et al. Aust N Z J Obstet Gynaecol. 2003;43(6):414-420.
4. Ruiz R et al. Breast. 2017;35:136-141. doi: 10.1016/j.breast.2017.07.008.
5. Nolan S et al. Am J Obstet Gynecol. 2019;220(1):S480. doi: 10.1016/j.ajog.2018.11.752.
6. El-Messidi A et al. J Perinat Med. 2015;43(6):683-688. doi: 10.1515/jpm-2014-0133.
7. Pellino G et al. Eur J Gastroenterol Hepatol. 2017;29(7):743-753. doi: 10.1097/MEG.0000000000000863.
8. Eastwood-Wilshere N et al. Asia-Pac J Clin Oncol. 2019;15:296-308.
9. Lee YY et al. BJOG. 2012;119(13):1572-1582.
10. Cottreau CM et al. J Womens Health (Larchmt). 2019 Feb;28(2):250-257.
11. Boere I et al. Best Pract Res Clin Obstet Gynaecol. 2022;82:46-59.
12. Ray JG et al. JAMA 2016;316(9):952-961.
13. Schwab R et al. Cancers. (Basel) 2021;13(12):3048.
14. Cohen-Kerem et al. Am J Surg. 2005;190(3):467-473.
Introduction: Tremendous Progress on Cancer Extends to Cancer in Pregnancy
The biomedical research enterprise that took shape in the United States after World War II has had numerous positive effects, including significant progress made during the past 75-plus years in the diagnosis, prevention, and treatment of cancer.
President Franklin D. Roosevelt’s 1944 request of Dr. Vannevar Bush, director of the then Office of Scientific Research and Development, to organize a program that would advance and apply scientific knowledge for times of peace — just as it been advanced and applied in times of war — culminated in a historic report, Science – The Endless Frontier. Presented in 1945 to President Harry S. Truman, this report helped fuel decades of broad, bold, and coordinated government-sponsored biomedical research aimed at addressing disease and improving the health of the American people (National Science Foundation, 1945).
Discoveries made from research in basic and translational sciences deepened our knowledge of the cellular and molecular underpinnings of cancer, leading to advances in chemotherapy, radiotherapy, and other treatment approaches as well as continual refinements in their application. Similarly, our diagnostic armamentarium has significantly improved.
As a result, we have reduced both the incidence and mortality of cancer. Today, some cancers can be prevented. Others can be reversed or put in remission. Granted, progress has been variable, with some cancers such as ovarian cancer still having relatively low survival rates. Much more needs to be done. Overall, however, the positive effects of the U.S. biomedical research enterprise on cancer are evident. According to the National Cancer Institute’s most recent report on the status of cancer, death rates from cancer fell 1.9% per year on average in females from 2015 to 2019 (Cancer. 2022 Oct 22. doi: 10.1002/cncr.34479).
It is not only patients whose cancer occurs outside of pregnancy who have benefited. When treatment is appropriately selected and timing considerations are made, patients whose cancer is diagnosed during pregnancy — and their children — can have good outcomes.
To explain how the management of cancer in pregnancy has improved, we have invited Gautam G. Rao, MD, gynecologic oncologist and associate professor of obstetrics, gynecology, and reproductive sciences at the University of Maryland School of Medicine, to write this installment of the Master Class in Obstetrics. As Dr. Rao explains, radiation is not as dangerous to the fetus as once thought, and the safety of many chemotherapeutic regimens in pregnancy has been documented. Obstetricians can and should counsel patients, he explains, about the likelihood of good maternal and fetal outcomes.
E. Albert Reece, MD, PhD, MBA, a maternal-fetal medicine specialist, is dean emeritus of the University of Maryland School of Medicine, former university executive vice president; currently the endowed professor and director of the Center for Advanced Research Training and Innovation (CARTI), and senior scientist in the Center for Birth Defects Research. Dr. Reece reported no relevant disclosures. He is the medical editor of this column. Contact him at [email protected].
Managing Cancer in Pregnancy
Cancer can cause fear and distress for any patient, but when cancer is diagnosed during pregnancy, an expectant mother fears not only for her own health but for the health of her unborn child. Fortunately, ob.gyn.s and multidisciplinary teams have good reason to reassure patients about the likelihood of good outcomes.
Cancer treatment in pregnancy has improved with advancements in imaging and chemotherapy, and while maternal and fetal outcomes of prenatal cancer treatment are not well reported, evidence acquired in recent years from case series and retrospective studies shows that most imaging studies and procedural diagnostic tests – and many treatments – can be performed safely in pregnancy.
Decades ago, we avoided CT scans during pregnancy because of concerns about radiation exposure to the fetus, leaving some patients without an accurate staging of their cancer. Today, we have evidence that a CT scan is generally safe in pregnancy. Similarly, the safety of many chemotherapeutic regimens in pregnancy has been documented in recent decades,and the use of chemotherapy during pregnancy has increased progressively. Radiation is also commonly utilized in the management of cancers that may occur during pregnancy, such as breast cancer.1
Considerations of timing are often central to decision-making; chemotherapy and radiotherapy are generally avoided in the first trimester to prevent structural fetal anomalies, for instance, and delaying cancer treatment is often warranted when the patient is a few weeks away from delivery. On occasion, iatrogenic preterm birth is considered when the risks to the mother of delaying a necessary cancer treatment outweigh the risks to the fetus of prematurity.1
Pregnancy termination is rarely indicated, however, and information gathered over the past 2 decades suggests that fetal and placental metastases are rare.1 There is broad agreement that prenatal treatment of cancer in pregnancy should adhere as much as possible to protocols and guidelines for nonpregnant patients and that treatment delays driven by fear of fetal anomalies and miscarriage are unnecessary.
Cancer Incidence, Use of Diagnostic Imaging
Data on the incidence of cancer in pregnancy comes from population-based cancer registries, and unfortunately, these data are not standardized and are often incomplete. Many studies include cancer diagnosed up to 1 year after pregnancy, and some include preinvasive disease. Estimates therefore vary considerably (see Table 1 for a sampling of estimates incidences.)
It has been reported, and often cited in the literature, that invasive malignancy complicates one in 1,000 pregnancies and that the incidence of cancer in pregnancy (invasive and noninvasive malignancies) has been rising over time.8 Increasing maternal age is believed to be playing a role in this rise; as women delay childbearing, they enter the age range in which some cancers become more common. Additionally, improvements in screening and diagnostics have led to earlier cancer detection. The incidence of ovarian neoplasms found during pregnancy has increased, for instance, with the routine use of diagnostic ultrasound in pregnancy.1
Among the studies showing an increased incidence of pregnancy-associated cancer is a population-based study in Australia, which found that from 1994 to 2007 the crude incidence of pregnancy-associated cancer increased from 112.3 to 191.5 per 100,000 pregnancies (P < .001).9 A cohort study in the United States documented an increase in incidence from 75.0 per 100,000 pregnancies in 2002 to 138.5 per 100,000 pregnancies in 2012.10
Overall, the literature shows us that the skin, cervix, and breast are also common sites for malignancy during pregnancy.1 According to a 2022 review, breast cancer during pregnancy is less often hormone receptor–positive and more frequently triple negative compared with age-matched controls.11 The frequencies of other pregnancy-associated cancers appear overall to be similar to that of cancer occurring in all women across their reproductive years.1
Too often, diagnosis is delayed because cancer symptoms can be masked by or can mimic normal physiological changes in pregnancy. For instance, breast cancer can be difficult to diagnose during pregnancy and lactation due to anatomic changes in the breast parenchyma. Several studies published in the 1990s showed that breast cancer presents at a more advanced stage in pregnant patients than in nonpregnant patients because of this delay.1 Skin changes suggestive of melanoma can be attributed to hyperpigmentation of pregnancy, for instance. Several observational studies have suggested that thicker melanomas found in pregnancy may be because of delayed diagnosis.8
It is important that we thoroughly investigate signs and symptoms suggestive of a malignancy and not automatically attribute these symptoms to the pregnancy itself. Cervical biopsy of a mass or lesion suspicious for cervical cancer can be done safely during pregnancy and should not be delayed or deferred.
Fetal radiation exposure from radiologic examinations has long been a concern, but we know today that while the imaging modality should be chosen to minimize fetal radiation exposure, CT scans and even PET scans should be performed if these exams are deemed best for evaluation. Embryonic exposure to a dose of less than 50 mGy is rarely if at all associated with fetal malformations or miscarriage and a radiation dose of 100 mGy may be considered a floor for consideration of therapeutic termination of pregnancy.1,8
CT exams are associated with a fetal dose far less than 50 mGy (see Table 2 for radiation doses).
Magnetic resonance imaging with a magnet strength of 3 Tesla or less in any trimester is not associated with an increased risk of harm to the fetus or in early childhood, but the contrast agent gadolinium should be avoided in pregnancy as it has been associated with an increased risk of stillbirth, neonatal death, and childhood inflammatory, rheumatologic, and infiltrative skin lesions.1,8,12
Chemotherapy, Surgery, and Radiation in Pregnancy
The management of cancer during pregnancy requires a multidisciplinary team including medical, gynecologic, or radiation oncologists, and maternal-fetal medicine specialists (Figure 1). Prematurity and low birth weight are frequent complications for fetuses exposed to chemotherapy, although there is some uncertainty as to whether the treatment is causative. However, congenital anomalies no longer are a major concern, provided that drugs are appropriately selected and that fetal exposure occurs during the second or third trimester.
For instance, alkylating agents including cisplatin (an important drug in the management of gynecologic malignancies) have been associated with congenital anomalies in the first trimester but not in the second and third trimesters, and a variety of antimetabolites — excluding methotrexate and aminopterin — similarly have been shown to be relatively safe when used after the first trimester.1
Small studies have shown no long-term effects of chemotherapy exposure on postnatal growth and long-term neurologic/neurocognitive function,1 but this is an area that needs more research.
Also in need of investigation is the safety of newer agents in pregnancy. Data are limited on the use of new targeted treatments, monoclonal antibodies, and immunotherapies in pregnancy and their effects on the fetus, with current knowledge coming mainly from single case reports.13
Until more is learned — a challenge given that pregnant women are generally excluded from clinical trials — management teams are generally postponing use of these therapies until after delivery. Considering the pace of new developments revolutionizing cancer treatment, this topic will likely get more complex and confusing before we begin acquiring sufficient knowledge.
The timing of surgery for malignancy in pregnancy is similarly based on the balance of maternal and fetal risks, including the risk of maternal disease progression, the risk of preterm delivery, and the prevention of fetal metastases. In general, the safest time is the second trimester.
Maternal surgery in the third trimester may be associated with a risk of premature labor and altered uteroplacental perfusion. A 2005 systematic review of 12,452 women who underwent nonobstetric surgery during pregnancy provides some reassurance, however; compared with the general obstetric population, there was no increase in the rate of miscarriage or major birth defects.14
Radiotherapy used to be contraindicated in pregnancy but many experts today believe it can be safely utilized provided the uterus is out of field and is protected from scattered radiation. The head, neck, and breast, for instance, can be treated with newer radiotherapies, including stereotactic ablative radiation therapy.8 Patients with advanced cervical cancer often receive chemotherapy during pregnancy to slow metastatic growth followed by definitive treatment with postpartum radiation or surgery.
More research is needed, but available data on maternal outcomes are encouraging. For instance, there appear to be no significant differences in short- and long-term complications or survival between women who are pregnant and nonpregnant when treated for invasive cervical cancer.8 Similarly, while earlier studies of breast cancer diagnosed during pregnancy suggested a poor prognosis, data now show similar prognoses for pregnant and nonpregnant patients when controlled for stage.1
Dr. Rao is a gynecologic oncologist and associate professor of obstetrics, gynecology, and reproductive sciences at the University of Maryland School of Medicine, Baltimore. He reported no relevant disclosures.
References
1. Rao GG. Chapter 42. Clinical Obstetrics: The Fetus & Mother, 4th ed. Reece EA et al. (eds): 2021.
2. Bannister-Tyrrell M et al. Aust N Z J Obstet Gynaecol. 2014;55:116-122.
3. Oehler MK et al. Aust N Z J Obstet Gynaecol. 2003;43(6):414-420.
4. Ruiz R et al. Breast. 2017;35:136-141. doi: 10.1016/j.breast.2017.07.008.
5. Nolan S et al. Am J Obstet Gynecol. 2019;220(1):S480. doi: 10.1016/j.ajog.2018.11.752.
6. El-Messidi A et al. J Perinat Med. 2015;43(6):683-688. doi: 10.1515/jpm-2014-0133.
7. Pellino G et al. Eur J Gastroenterol Hepatol. 2017;29(7):743-753. doi: 10.1097/MEG.0000000000000863.
8. Eastwood-Wilshere N et al. Asia-Pac J Clin Oncol. 2019;15:296-308.
9. Lee YY et al. BJOG. 2012;119(13):1572-1582.
10. Cottreau CM et al. J Womens Health (Larchmt). 2019 Feb;28(2):250-257.
11. Boere I et al. Best Pract Res Clin Obstet Gynaecol. 2022;82:46-59.
12. Ray JG et al. JAMA 2016;316(9):952-961.
13. Schwab R et al. Cancers. (Basel) 2021;13(12):3048.
14. Cohen-Kerem et al. Am J Surg. 2005;190(3):467-473.
Introduction: Tremendous Progress on Cancer Extends to Cancer in Pregnancy
The biomedical research enterprise that took shape in the United States after World War II has had numerous positive effects, including significant progress made during the past 75-plus years in the diagnosis, prevention, and treatment of cancer.
President Franklin D. Roosevelt’s 1944 request of Dr. Vannevar Bush, director of the then Office of Scientific Research and Development, to organize a program that would advance and apply scientific knowledge for times of peace — just as it been advanced and applied in times of war — culminated in a historic report, Science – The Endless Frontier. Presented in 1945 to President Harry S. Truman, this report helped fuel decades of broad, bold, and coordinated government-sponsored biomedical research aimed at addressing disease and improving the health of the American people (National Science Foundation, 1945).
Discoveries made from research in basic and translational sciences deepened our knowledge of the cellular and molecular underpinnings of cancer, leading to advances in chemotherapy, radiotherapy, and other treatment approaches as well as continual refinements in their application. Similarly, our diagnostic armamentarium has significantly improved.
As a result, we have reduced both the incidence and mortality of cancer. Today, some cancers can be prevented. Others can be reversed or put in remission. Granted, progress has been variable, with some cancers such as ovarian cancer still having relatively low survival rates. Much more needs to be done. Overall, however, the positive effects of the U.S. biomedical research enterprise on cancer are evident. According to the National Cancer Institute’s most recent report on the status of cancer, death rates from cancer fell 1.9% per year on average in females from 2015 to 2019 (Cancer. 2022 Oct 22. doi: 10.1002/cncr.34479).
It is not only patients whose cancer occurs outside of pregnancy who have benefited. When treatment is appropriately selected and timing considerations are made, patients whose cancer is diagnosed during pregnancy — and their children — can have good outcomes.
To explain how the management of cancer in pregnancy has improved, we have invited Gautam G. Rao, MD, gynecologic oncologist and associate professor of obstetrics, gynecology, and reproductive sciences at the University of Maryland School of Medicine, to write this installment of the Master Class in Obstetrics. As Dr. Rao explains, radiation is not as dangerous to the fetus as once thought, and the safety of many chemotherapeutic regimens in pregnancy has been documented. Obstetricians can and should counsel patients, he explains, about the likelihood of good maternal and fetal outcomes.
E. Albert Reece, MD, PhD, MBA, a maternal-fetal medicine specialist, is dean emeritus of the University of Maryland School of Medicine, former university executive vice president; currently the endowed professor and director of the Center for Advanced Research Training and Innovation (CARTI), and senior scientist in the Center for Birth Defects Research. Dr. Reece reported no relevant disclosures. He is the medical editor of this column. Contact him at [email protected].
Managing Cancer in Pregnancy
Cancer can cause fear and distress for any patient, but when cancer is diagnosed during pregnancy, an expectant mother fears not only for her own health but for the health of her unborn child. Fortunately, ob.gyn.s and multidisciplinary teams have good reason to reassure patients about the likelihood of good outcomes.
Cancer treatment in pregnancy has improved with advancements in imaging and chemotherapy, and while maternal and fetal outcomes of prenatal cancer treatment are not well reported, evidence acquired in recent years from case series and retrospective studies shows that most imaging studies and procedural diagnostic tests – and many treatments – can be performed safely in pregnancy.
Decades ago, we avoided CT scans during pregnancy because of concerns about radiation exposure to the fetus, leaving some patients without an accurate staging of their cancer. Today, we have evidence that a CT scan is generally safe in pregnancy. Similarly, the safety of many chemotherapeutic regimens in pregnancy has been documented in recent decades,and the use of chemotherapy during pregnancy has increased progressively. Radiation is also commonly utilized in the management of cancers that may occur during pregnancy, such as breast cancer.1
Considerations of timing are often central to decision-making; chemotherapy and radiotherapy are generally avoided in the first trimester to prevent structural fetal anomalies, for instance, and delaying cancer treatment is often warranted when the patient is a few weeks away from delivery. On occasion, iatrogenic preterm birth is considered when the risks to the mother of delaying a necessary cancer treatment outweigh the risks to the fetus of prematurity.1
Pregnancy termination is rarely indicated, however, and information gathered over the past 2 decades suggests that fetal and placental metastases are rare.1 There is broad agreement that prenatal treatment of cancer in pregnancy should adhere as much as possible to protocols and guidelines for nonpregnant patients and that treatment delays driven by fear of fetal anomalies and miscarriage are unnecessary.
Cancer Incidence, Use of Diagnostic Imaging
Data on the incidence of cancer in pregnancy comes from population-based cancer registries, and unfortunately, these data are not standardized and are often incomplete. Many studies include cancer diagnosed up to 1 year after pregnancy, and some include preinvasive disease. Estimates therefore vary considerably (see Table 1 for a sampling of estimates incidences.)
It has been reported, and often cited in the literature, that invasive malignancy complicates one in 1,000 pregnancies and that the incidence of cancer in pregnancy (invasive and noninvasive malignancies) has been rising over time.8 Increasing maternal age is believed to be playing a role in this rise; as women delay childbearing, they enter the age range in which some cancers become more common. Additionally, improvements in screening and diagnostics have led to earlier cancer detection. The incidence of ovarian neoplasms found during pregnancy has increased, for instance, with the routine use of diagnostic ultrasound in pregnancy.1
Among the studies showing an increased incidence of pregnancy-associated cancer is a population-based study in Australia, which found that from 1994 to 2007 the crude incidence of pregnancy-associated cancer increased from 112.3 to 191.5 per 100,000 pregnancies (P < .001).9 A cohort study in the United States documented an increase in incidence from 75.0 per 100,000 pregnancies in 2002 to 138.5 per 100,000 pregnancies in 2012.10
Overall, the literature shows us that the skin, cervix, and breast are also common sites for malignancy during pregnancy.1 According to a 2022 review, breast cancer during pregnancy is less often hormone receptor–positive and more frequently triple negative compared with age-matched controls.11 The frequencies of other pregnancy-associated cancers appear overall to be similar to that of cancer occurring in all women across their reproductive years.1
Too often, diagnosis is delayed because cancer symptoms can be masked by or can mimic normal physiological changes in pregnancy. For instance, breast cancer can be difficult to diagnose during pregnancy and lactation due to anatomic changes in the breast parenchyma. Several studies published in the 1990s showed that breast cancer presents at a more advanced stage in pregnant patients than in nonpregnant patients because of this delay.1 Skin changes suggestive of melanoma can be attributed to hyperpigmentation of pregnancy, for instance. Several observational studies have suggested that thicker melanomas found in pregnancy may be because of delayed diagnosis.8
It is important that we thoroughly investigate signs and symptoms suggestive of a malignancy and not automatically attribute these symptoms to the pregnancy itself. Cervical biopsy of a mass or lesion suspicious for cervical cancer can be done safely during pregnancy and should not be delayed or deferred.
Fetal radiation exposure from radiologic examinations has long been a concern, but we know today that while the imaging modality should be chosen to minimize fetal radiation exposure, CT scans and even PET scans should be performed if these exams are deemed best for evaluation. Embryonic exposure to a dose of less than 50 mGy is rarely if at all associated with fetal malformations or miscarriage and a radiation dose of 100 mGy may be considered a floor for consideration of therapeutic termination of pregnancy.1,8
CT exams are associated with a fetal dose far less than 50 mGy (see Table 2 for radiation doses).
Magnetic resonance imaging with a magnet strength of 3 Tesla or less in any trimester is not associated with an increased risk of harm to the fetus or in early childhood, but the contrast agent gadolinium should be avoided in pregnancy as it has been associated with an increased risk of stillbirth, neonatal death, and childhood inflammatory, rheumatologic, and infiltrative skin lesions.1,8,12
Chemotherapy, Surgery, and Radiation in Pregnancy
The management of cancer during pregnancy requires a multidisciplinary team including medical, gynecologic, or radiation oncologists, and maternal-fetal medicine specialists (Figure 1). Prematurity and low birth weight are frequent complications for fetuses exposed to chemotherapy, although there is some uncertainty as to whether the treatment is causative. However, congenital anomalies no longer are a major concern, provided that drugs are appropriately selected and that fetal exposure occurs during the second or third trimester.
For instance, alkylating agents including cisplatin (an important drug in the management of gynecologic malignancies) have been associated with congenital anomalies in the first trimester but not in the second and third trimesters, and a variety of antimetabolites — excluding methotrexate and aminopterin — similarly have been shown to be relatively safe when used after the first trimester.1
Small studies have shown no long-term effects of chemotherapy exposure on postnatal growth and long-term neurologic/neurocognitive function,1 but this is an area that needs more research.
Also in need of investigation is the safety of newer agents in pregnancy. Data are limited on the use of new targeted treatments, monoclonal antibodies, and immunotherapies in pregnancy and their effects on the fetus, with current knowledge coming mainly from single case reports.13
Until more is learned — a challenge given that pregnant women are generally excluded from clinical trials — management teams are generally postponing use of these therapies until after delivery. Considering the pace of new developments revolutionizing cancer treatment, this topic will likely get more complex and confusing before we begin acquiring sufficient knowledge.
The timing of surgery for malignancy in pregnancy is similarly based on the balance of maternal and fetal risks, including the risk of maternal disease progression, the risk of preterm delivery, and the prevention of fetal metastases. In general, the safest time is the second trimester.
Maternal surgery in the third trimester may be associated with a risk of premature labor and altered uteroplacental perfusion. A 2005 systematic review of 12,452 women who underwent nonobstetric surgery during pregnancy provides some reassurance, however; compared with the general obstetric population, there was no increase in the rate of miscarriage or major birth defects.14
Radiotherapy used to be contraindicated in pregnancy but many experts today believe it can be safely utilized provided the uterus is out of field and is protected from scattered radiation. The head, neck, and breast, for instance, can be treated with newer radiotherapies, including stereotactic ablative radiation therapy.8 Patients with advanced cervical cancer often receive chemotherapy during pregnancy to slow metastatic growth followed by definitive treatment with postpartum radiation or surgery.
More research is needed, but available data on maternal outcomes are encouraging. For instance, there appear to be no significant differences in short- and long-term complications or survival between women who are pregnant and nonpregnant when treated for invasive cervical cancer.8 Similarly, while earlier studies of breast cancer diagnosed during pregnancy suggested a poor prognosis, data now show similar prognoses for pregnant and nonpregnant patients when controlled for stage.1
Dr. Rao is a gynecologic oncologist and associate professor of obstetrics, gynecology, and reproductive sciences at the University of Maryland School of Medicine, Baltimore. He reported no relevant disclosures.
References
1. Rao GG. Chapter 42. Clinical Obstetrics: The Fetus & Mother, 4th ed. Reece EA et al. (eds): 2021.
2. Bannister-Tyrrell M et al. Aust N Z J Obstet Gynaecol. 2014;55:116-122.
3. Oehler MK et al. Aust N Z J Obstet Gynaecol. 2003;43(6):414-420.
4. Ruiz R et al. Breast. 2017;35:136-141. doi: 10.1016/j.breast.2017.07.008.
5. Nolan S et al. Am J Obstet Gynecol. 2019;220(1):S480. doi: 10.1016/j.ajog.2018.11.752.
6. El-Messidi A et al. J Perinat Med. 2015;43(6):683-688. doi: 10.1515/jpm-2014-0133.
7. Pellino G et al. Eur J Gastroenterol Hepatol. 2017;29(7):743-753. doi: 10.1097/MEG.0000000000000863.
8. Eastwood-Wilshere N et al. Asia-Pac J Clin Oncol. 2019;15:296-308.
9. Lee YY et al. BJOG. 2012;119(13):1572-1582.
10. Cottreau CM et al. J Womens Health (Larchmt). 2019 Feb;28(2):250-257.
11. Boere I et al. Best Pract Res Clin Obstet Gynaecol. 2022;82:46-59.
12. Ray JG et al. JAMA 2016;316(9):952-961.
13. Schwab R et al. Cancers. (Basel) 2021;13(12):3048.
14. Cohen-Kerem et al. Am J Surg. 2005;190(3):467-473.
Benefit of Massage Therapy for Pain Unclear
The effectiveness of massage therapy for a range of painful adult health conditions remains uncertain. Despite hundreds of randomized clinical trials and dozens of systematic reviews, few studies have offered conclusions based on more than low-certainty evidence, a systematic review in JAMA Network Open has shown (doi: 10.1001/jamanetworkopen.2024.22259).
Some moderate-certainty evidence, however, suggested massage therapy may alleviate pain related to such conditions as low-back problems, labor, and breast cancer surgery, concluded a group led by Selene Mak, PhD, MPH, program manager in the Evidence Synthesis Program at the Veterans Health Administration Greater Los Angeles Healthcare System in Los Angeles, California.
“More high-quality randomized clinical trials are needed to provide a stronger evidence base to assess the effect of massage therapy on pain,” Dr. Mak and colleagues wrote.
The review updates a previous Veterans Affairs evidence map covering reviews of massage therapy for pain published through 2018.
To categorize the evidence base for decision-making by policymakers and practitioners, the VA requested an updated evidence map of reviews to answer the question: “What is the certainty of evidence in systematic reviews of massage therapy for pain?”
The Analysis
The current review included studies published from 2018 to 2023 with formal ratings of evidence quality or certainty, excluding other nonpharmacologic techniques such as sports massage therapy, osteopathy, dry cupping, dry needling, and internal massage therapy, and self-administered techniques such as foam rolling.
Of 129 systematic reviews, only 41 formally rated evidence quality, and 17 were evidence-mapped for pain across 13 health states: cancer, back, neck and mechanical neck issues, fibromyalgia, labor, myofascial, palliative care need, plantar fasciitis, postoperative, post breast cancer surgery, and post cesarean/postpartum.
The investigators found no conclusions based on a high certainty of evidence, while seven based conclusions on moderate-certainty evidence. All remaining conclusions were rated as having low- or very-low-certainty evidence.
The priority, they added, should be studies comparing massage therapy with other recommended, accepted, and active therapies for pain and should have sufficiently long follow-up to allow any nonspecific outcomes to dissipate, At least 6 months’ follow-up has been suggested for studies of chronic pain.
While massage therapy is considered safe, in patients with central sensitizations more aggressive treatments may cause a flare of myofascial pain.
This study was funded by the Department of Veterans Affairs Health Services Research and Development. The authors had no conflicts of interest to disclose.
The effectiveness of massage therapy for a range of painful adult health conditions remains uncertain. Despite hundreds of randomized clinical trials and dozens of systematic reviews, few studies have offered conclusions based on more than low-certainty evidence, a systematic review in JAMA Network Open has shown (doi: 10.1001/jamanetworkopen.2024.22259).
Some moderate-certainty evidence, however, suggested massage therapy may alleviate pain related to such conditions as low-back problems, labor, and breast cancer surgery, concluded a group led by Selene Mak, PhD, MPH, program manager in the Evidence Synthesis Program at the Veterans Health Administration Greater Los Angeles Healthcare System in Los Angeles, California.
“More high-quality randomized clinical trials are needed to provide a stronger evidence base to assess the effect of massage therapy on pain,” Dr. Mak and colleagues wrote.
The review updates a previous Veterans Affairs evidence map covering reviews of massage therapy for pain published through 2018.
To categorize the evidence base for decision-making by policymakers and practitioners, the VA requested an updated evidence map of reviews to answer the question: “What is the certainty of evidence in systematic reviews of massage therapy for pain?”
The Analysis
The current review included studies published from 2018 to 2023 with formal ratings of evidence quality or certainty, excluding other nonpharmacologic techniques such as sports massage therapy, osteopathy, dry cupping, dry needling, and internal massage therapy, and self-administered techniques such as foam rolling.
Of 129 systematic reviews, only 41 formally rated evidence quality, and 17 were evidence-mapped for pain across 13 health states: cancer, back, neck and mechanical neck issues, fibromyalgia, labor, myofascial, palliative care need, plantar fasciitis, postoperative, post breast cancer surgery, and post cesarean/postpartum.
The investigators found no conclusions based on a high certainty of evidence, while seven based conclusions on moderate-certainty evidence. All remaining conclusions were rated as having low- or very-low-certainty evidence.
The priority, they added, should be studies comparing massage therapy with other recommended, accepted, and active therapies for pain and should have sufficiently long follow-up to allow any nonspecific outcomes to dissipate, At least 6 months’ follow-up has been suggested for studies of chronic pain.
While massage therapy is considered safe, in patients with central sensitizations more aggressive treatments may cause a flare of myofascial pain.
This study was funded by the Department of Veterans Affairs Health Services Research and Development. The authors had no conflicts of interest to disclose.
The effectiveness of massage therapy for a range of painful adult health conditions remains uncertain. Despite hundreds of randomized clinical trials and dozens of systematic reviews, few studies have offered conclusions based on more than low-certainty evidence, a systematic review in JAMA Network Open has shown (doi: 10.1001/jamanetworkopen.2024.22259).
Some moderate-certainty evidence, however, suggested massage therapy may alleviate pain related to such conditions as low-back problems, labor, and breast cancer surgery, concluded a group led by Selene Mak, PhD, MPH, program manager in the Evidence Synthesis Program at the Veterans Health Administration Greater Los Angeles Healthcare System in Los Angeles, California.
“More high-quality randomized clinical trials are needed to provide a stronger evidence base to assess the effect of massage therapy on pain,” Dr. Mak and colleagues wrote.
The review updates a previous Veterans Affairs evidence map covering reviews of massage therapy for pain published through 2018.
To categorize the evidence base for decision-making by policymakers and practitioners, the VA requested an updated evidence map of reviews to answer the question: “What is the certainty of evidence in systematic reviews of massage therapy for pain?”
The Analysis
The current review included studies published from 2018 to 2023 with formal ratings of evidence quality or certainty, excluding other nonpharmacologic techniques such as sports massage therapy, osteopathy, dry cupping, dry needling, and internal massage therapy, and self-administered techniques such as foam rolling.
Of 129 systematic reviews, only 41 formally rated evidence quality, and 17 were evidence-mapped for pain across 13 health states: cancer, back, neck and mechanical neck issues, fibromyalgia, labor, myofascial, palliative care need, plantar fasciitis, postoperative, post breast cancer surgery, and post cesarean/postpartum.
The investigators found no conclusions based on a high certainty of evidence, while seven based conclusions on moderate-certainty evidence. All remaining conclusions were rated as having low- or very-low-certainty evidence.
The priority, they added, should be studies comparing massage therapy with other recommended, accepted, and active therapies for pain and should have sufficiently long follow-up to allow any nonspecific outcomes to dissipate, At least 6 months’ follow-up has been suggested for studies of chronic pain.
While massage therapy is considered safe, in patients with central sensitizations more aggressive treatments may cause a flare of myofascial pain.
This study was funded by the Department of Veterans Affairs Health Services Research and Development. The authors had no conflicts of interest to disclose.
FROM JAMA NETWORK OPEN
Medicare Rates in 2025 Would Cut Pay For Docs by 3%
Federal officials on July 11 proposed Medicare rates that effectively would cut physician pay by about 3% in 2025, touching off a fresh round of protests from medical associations.
, the Centers for Medicare & Medicaid Services said.
The American Medical Association (AMA), the American Academy of Family Physicians (AAFP) and other groups on July 10 reiterated calls on Congress to revise the law on Medicare payment for physicians and move away from short-term tweaks.
This proposed cut is mostly due to the 5-year freeze in the physician schedule base rate mandated by the 2015 Medicare Access and CHIP Reauthorization Act (MACRA). Congress designed MACRA with an aim of shifting clinicians toward programs that would peg pay increases to quality measures.
Lawmakers have since had to soften the blow of that freeze, acknowledging flaws in MACRA and inflation’s significant toll on medical practices. Yet lawmakers have made temporary fixes, such as a 2.93% increase in current payment that’s set to expire.
“Previous quick fixes have been insufficient — this situation requires a bold, substantial approach,” Bruce A. Scott, MD, the AMA president, said in a statement. “A Band-Aid goes only so far when the patient is in dire need.”
Dr. Scott noted that the Medicare Economic Index — a measure of practice cost inflation — is expected to rise by 3.6% in 2025.
“As a first step, Congress must enact an annual inflationary update to help physician payment rates keep pace with rising practice costs,” Steven P. Furr, MD, AAFP’s president, said in a statement released July 10. “Any payment reductions will threaten practices and exacerbate workforce shortages, preventing patients from accessing the primary care, behavioral health care, and other critical preventive services they need.”
Many medical groups, including the AMA, AAFP, and the Medical Group Management Association, are pressing Congress to pass a law that would tie the conversion factor of the physician fee schedule to inflation.
Influential advisory groups also have backed the idea of increasing the conversion factor. For example, the Medicare Payment Advisory Commission in March recommended to Congress that it increase the 2025 conversion factor, suggesting a bump of half of the projected increase in the Medicare Economic Index.
Congress seems unlikely to revamp the physician fee schedule this year, with members spending significant time away from Washington ahead of the November election.
That could make it likely that Congress’ next action on Medicare payment rates would be another short-term tweak — instead of long-lasting change.
A version of this article first appeared on Medscape.com.
Federal officials on July 11 proposed Medicare rates that effectively would cut physician pay by about 3% in 2025, touching off a fresh round of protests from medical associations.
, the Centers for Medicare & Medicaid Services said.
The American Medical Association (AMA), the American Academy of Family Physicians (AAFP) and other groups on July 10 reiterated calls on Congress to revise the law on Medicare payment for physicians and move away from short-term tweaks.
This proposed cut is mostly due to the 5-year freeze in the physician schedule base rate mandated by the 2015 Medicare Access and CHIP Reauthorization Act (MACRA). Congress designed MACRA with an aim of shifting clinicians toward programs that would peg pay increases to quality measures.
Lawmakers have since had to soften the blow of that freeze, acknowledging flaws in MACRA and inflation’s significant toll on medical practices. Yet lawmakers have made temporary fixes, such as a 2.93% increase in current payment that’s set to expire.
“Previous quick fixes have been insufficient — this situation requires a bold, substantial approach,” Bruce A. Scott, MD, the AMA president, said in a statement. “A Band-Aid goes only so far when the patient is in dire need.”
Dr. Scott noted that the Medicare Economic Index — a measure of practice cost inflation — is expected to rise by 3.6% in 2025.
“As a first step, Congress must enact an annual inflationary update to help physician payment rates keep pace with rising practice costs,” Steven P. Furr, MD, AAFP’s president, said in a statement released July 10. “Any payment reductions will threaten practices and exacerbate workforce shortages, preventing patients from accessing the primary care, behavioral health care, and other critical preventive services they need.”
Many medical groups, including the AMA, AAFP, and the Medical Group Management Association, are pressing Congress to pass a law that would tie the conversion factor of the physician fee schedule to inflation.
Influential advisory groups also have backed the idea of increasing the conversion factor. For example, the Medicare Payment Advisory Commission in March recommended to Congress that it increase the 2025 conversion factor, suggesting a bump of half of the projected increase in the Medicare Economic Index.
Congress seems unlikely to revamp the physician fee schedule this year, with members spending significant time away from Washington ahead of the November election.
That could make it likely that Congress’ next action on Medicare payment rates would be another short-term tweak — instead of long-lasting change.
A version of this article first appeared on Medscape.com.
Federal officials on July 11 proposed Medicare rates that effectively would cut physician pay by about 3% in 2025, touching off a fresh round of protests from medical associations.
, the Centers for Medicare & Medicaid Services said.
The American Medical Association (AMA), the American Academy of Family Physicians (AAFP) and other groups on July 10 reiterated calls on Congress to revise the law on Medicare payment for physicians and move away from short-term tweaks.
This proposed cut is mostly due to the 5-year freeze in the physician schedule base rate mandated by the 2015 Medicare Access and CHIP Reauthorization Act (MACRA). Congress designed MACRA with an aim of shifting clinicians toward programs that would peg pay increases to quality measures.
Lawmakers have since had to soften the blow of that freeze, acknowledging flaws in MACRA and inflation’s significant toll on medical practices. Yet lawmakers have made temporary fixes, such as a 2.93% increase in current payment that’s set to expire.
“Previous quick fixes have been insufficient — this situation requires a bold, substantial approach,” Bruce A. Scott, MD, the AMA president, said in a statement. “A Band-Aid goes only so far when the patient is in dire need.”
Dr. Scott noted that the Medicare Economic Index — a measure of practice cost inflation — is expected to rise by 3.6% in 2025.
“As a first step, Congress must enact an annual inflationary update to help physician payment rates keep pace with rising practice costs,” Steven P. Furr, MD, AAFP’s president, said in a statement released July 10. “Any payment reductions will threaten practices and exacerbate workforce shortages, preventing patients from accessing the primary care, behavioral health care, and other critical preventive services they need.”
Many medical groups, including the AMA, AAFP, and the Medical Group Management Association, are pressing Congress to pass a law that would tie the conversion factor of the physician fee schedule to inflation.
Influential advisory groups also have backed the idea of increasing the conversion factor. For example, the Medicare Payment Advisory Commission in March recommended to Congress that it increase the 2025 conversion factor, suggesting a bump of half of the projected increase in the Medicare Economic Index.
Congress seems unlikely to revamp the physician fee schedule this year, with members spending significant time away from Washington ahead of the November election.
That could make it likely that Congress’ next action on Medicare payment rates would be another short-term tweak — instead of long-lasting change.
A version of this article first appeared on Medscape.com.
Eribulin Similar to Taxane When Paired With Dual HER2 Blockade in BC
The results of this multicenter, randomized, open-label, parallel-group, phase 3 Japanese trial suggest that patients who cannot tolerate the standard taxane-based regimen have a new option for treatment.
“Our study is the first to show the non-inferiority of eribulin to a taxane, when used in combination with dual HER2 blockade as first-line treatment for this population,” lead author Toshinari Yamashita, MD, PhD, from the Kanagawa Cancer Center, in Kanagawa, Japan, said at the annual meeting of the American Society of Clinical Oncology.
“To our knowledge, noninferiority of eribulin to a taxane when used in combination with dual HER2 blockade has not been investigated,” Dr. Yamashita said.
“The combination of trastuzumab, pertuzumab, and taxane is a current standard first-line therapy for recurrent or metastatic HER2-positive breast cancer,” explained Dr. Yamashita. “However, because of taxane-induced toxicity, the development of less toxic but equally effective alternatives are needed.
“Because its efficacy is comparable to that of the current standard regimen, the combination of eribulin, trastuzumab, and pertuzumab is one of the options for first-line treatment of how to fight locally advanced or metastatic breast cancer,” he continued.
Study Results and Methods
The trial enrolled 446 patients, mean age 56 years, all of whom had locally advanced or metastatic breast cancer and no prior use of chemotherapy, excluding T-DM1. Patients who had received hormonal or HER2 therapy alone or the combination, as treatment for recurrence, were also eligible.
They were randomized 1:1 to receive a 21-day chemotherapy cycle of either (i) eribulin (1.4 mg/m2 on days 1 and 8), or (ii) a taxane (docetaxel 75 mg/m2 on day 1 or paclitaxel 80 mg/m2 on days 1, 8 and 15), each being administered in combination with a dual HER2 blockade of trastuzumab plus pertuzumab.
Baseline characteristics of both groups were well balanced, with 257 (57.6%) having ER-positive disease, 292 (65.5%) visceral metastasis, and 263 (59%) with de novo stage 4 disease, explained Dr. Yamashita.
For the primary endpoint, the median progression-free survival (PFS) was 14 versus 12.9 months in the eribulin and taxane group, respectively (hazard ratio [HR] 0.95, P = .6817), confirming non-inferiority of the study regimen, he reported.
The clinical benefit rate was similar between the two groups, with an objective response rate of 76.8% in the eribulin group and 75.2% in the taxane group.
Median OS was 65.3 months in the taxane group, but has not been reached in the study group (HR 1.09).
In terms of side-effects, the incidence of treatment-emergent adverse events was similar between the eribulin and taxane groups (58.9% vs 59.2%, respectively, for grade 3 or higher).
“Skin-related adverse events (62.4% vs 40.6%), diarrhea (54.1% vs 36.6%), and edema (42.2% vs 8.5%) tend to be more common with taxane, whereas neutropenia (61.6% vs 30.7%) and peripheral neuropathy (61.2% vs 52.8%) tend to be more common with eribulin use,” he said.
Overall, “these results suggest that eribulin is less toxic chemotherapeutic partner for dual HER2 blockade and can be used for a longer,” he said.
Findings Are a ‘Clinical Pearl’
Harold Burstein, MD, PhD, a breast cancer expert at Dana-Farber Cancer Institute and professor at Harvard Medical School in Boston, described the findings as “a nice clinical pearl,” because some patients do not tolerate taxane therapy. “In such cases, you can substitute eribulin, which is usually tolerated without allergic hypersensitivity issues,” he said in an interview.
Eribulin has specific properties that “could make it a perfect candidate” as an adjunct to standard treatment regimens across different breast cancer subtypes, observed Wynne Wijaya, MD an oncology researcher at the University of Oxford, England, and Universitas Gadjah Mada, in Yogyakarta, Indonesia, in a recent review (World J Exp Med. 2024;14[2]:92558).
Dr. Wijaya, who was not involved in this study, said in an interview that the findings have important implications.
“This encouraging result adds eribulin as another option in the first line treatment regimen for patients with HER2-positive, locally advanced or metastatic breast cancer, especially in terms of side effects/toxicities,” she said. “As clinicians, we can offer to tailor the choice of therapy between eribulin versus taxane in the regimen based on [which side effects patients are better able to tolerate]. It would also be interesting and worthwhile to conduct similar trials in different types of populations to provide more robust evidence.”
Eisai Co. funded the research. Dr. Yamashita disclosed ties with AstraZeneca, Chugai Pharma, Daiichi Sankyo, Eisai, Kyowa Hakko Kiri, Lilly, MSD, Pfizer, Taiho, Gilead Sciences, Nihonkayaku, Ono Yakuhin, and Seagen. Dr. Burstein disclosed a research grant from National Cancer Institute. Dr. Wijaya had no relevant disclosures.
The results of this multicenter, randomized, open-label, parallel-group, phase 3 Japanese trial suggest that patients who cannot tolerate the standard taxane-based regimen have a new option for treatment.
“Our study is the first to show the non-inferiority of eribulin to a taxane, when used in combination with dual HER2 blockade as first-line treatment for this population,” lead author Toshinari Yamashita, MD, PhD, from the Kanagawa Cancer Center, in Kanagawa, Japan, said at the annual meeting of the American Society of Clinical Oncology.
“To our knowledge, noninferiority of eribulin to a taxane when used in combination with dual HER2 blockade has not been investigated,” Dr. Yamashita said.
“The combination of trastuzumab, pertuzumab, and taxane is a current standard first-line therapy for recurrent or metastatic HER2-positive breast cancer,” explained Dr. Yamashita. “However, because of taxane-induced toxicity, the development of less toxic but equally effective alternatives are needed.
“Because its efficacy is comparable to that of the current standard regimen, the combination of eribulin, trastuzumab, and pertuzumab is one of the options for first-line treatment of how to fight locally advanced or metastatic breast cancer,” he continued.
Study Results and Methods
The trial enrolled 446 patients, mean age 56 years, all of whom had locally advanced or metastatic breast cancer and no prior use of chemotherapy, excluding T-DM1. Patients who had received hormonal or HER2 therapy alone or the combination, as treatment for recurrence, were also eligible.
They were randomized 1:1 to receive a 21-day chemotherapy cycle of either (i) eribulin (1.4 mg/m2 on days 1 and 8), or (ii) a taxane (docetaxel 75 mg/m2 on day 1 or paclitaxel 80 mg/m2 on days 1, 8 and 15), each being administered in combination with a dual HER2 blockade of trastuzumab plus pertuzumab.
Baseline characteristics of both groups were well balanced, with 257 (57.6%) having ER-positive disease, 292 (65.5%) visceral metastasis, and 263 (59%) with de novo stage 4 disease, explained Dr. Yamashita.
For the primary endpoint, the median progression-free survival (PFS) was 14 versus 12.9 months in the eribulin and taxane group, respectively (hazard ratio [HR] 0.95, P = .6817), confirming non-inferiority of the study regimen, he reported.
The clinical benefit rate was similar between the two groups, with an objective response rate of 76.8% in the eribulin group and 75.2% in the taxane group.
Median OS was 65.3 months in the taxane group, but has not been reached in the study group (HR 1.09).
In terms of side-effects, the incidence of treatment-emergent adverse events was similar between the eribulin and taxane groups (58.9% vs 59.2%, respectively, for grade 3 or higher).
“Skin-related adverse events (62.4% vs 40.6%), diarrhea (54.1% vs 36.6%), and edema (42.2% vs 8.5%) tend to be more common with taxane, whereas neutropenia (61.6% vs 30.7%) and peripheral neuropathy (61.2% vs 52.8%) tend to be more common with eribulin use,” he said.
Overall, “these results suggest that eribulin is less toxic chemotherapeutic partner for dual HER2 blockade and can be used for a longer,” he said.
Findings Are a ‘Clinical Pearl’
Harold Burstein, MD, PhD, a breast cancer expert at Dana-Farber Cancer Institute and professor at Harvard Medical School in Boston, described the findings as “a nice clinical pearl,” because some patients do not tolerate taxane therapy. “In such cases, you can substitute eribulin, which is usually tolerated without allergic hypersensitivity issues,” he said in an interview.
Eribulin has specific properties that “could make it a perfect candidate” as an adjunct to standard treatment regimens across different breast cancer subtypes, observed Wynne Wijaya, MD an oncology researcher at the University of Oxford, England, and Universitas Gadjah Mada, in Yogyakarta, Indonesia, in a recent review (World J Exp Med. 2024;14[2]:92558).
Dr. Wijaya, who was not involved in this study, said in an interview that the findings have important implications.
“This encouraging result adds eribulin as another option in the first line treatment regimen for patients with HER2-positive, locally advanced or metastatic breast cancer, especially in terms of side effects/toxicities,” she said. “As clinicians, we can offer to tailor the choice of therapy between eribulin versus taxane in the regimen based on [which side effects patients are better able to tolerate]. It would also be interesting and worthwhile to conduct similar trials in different types of populations to provide more robust evidence.”
Eisai Co. funded the research. Dr. Yamashita disclosed ties with AstraZeneca, Chugai Pharma, Daiichi Sankyo, Eisai, Kyowa Hakko Kiri, Lilly, MSD, Pfizer, Taiho, Gilead Sciences, Nihonkayaku, Ono Yakuhin, and Seagen. Dr. Burstein disclosed a research grant from National Cancer Institute. Dr. Wijaya had no relevant disclosures.
The results of this multicenter, randomized, open-label, parallel-group, phase 3 Japanese trial suggest that patients who cannot tolerate the standard taxane-based regimen have a new option for treatment.
“Our study is the first to show the non-inferiority of eribulin to a taxane, when used in combination with dual HER2 blockade as first-line treatment for this population,” lead author Toshinari Yamashita, MD, PhD, from the Kanagawa Cancer Center, in Kanagawa, Japan, said at the annual meeting of the American Society of Clinical Oncology.
“To our knowledge, noninferiority of eribulin to a taxane when used in combination with dual HER2 blockade has not been investigated,” Dr. Yamashita said.
“The combination of trastuzumab, pertuzumab, and taxane is a current standard first-line therapy for recurrent or metastatic HER2-positive breast cancer,” explained Dr. Yamashita. “However, because of taxane-induced toxicity, the development of less toxic but equally effective alternatives are needed.
“Because its efficacy is comparable to that of the current standard regimen, the combination of eribulin, trastuzumab, and pertuzumab is one of the options for first-line treatment of how to fight locally advanced or metastatic breast cancer,” he continued.
Study Results and Methods
The trial enrolled 446 patients, mean age 56 years, all of whom had locally advanced or metastatic breast cancer and no prior use of chemotherapy, excluding T-DM1. Patients who had received hormonal or HER2 therapy alone or the combination, as treatment for recurrence, were also eligible.
They were randomized 1:1 to receive a 21-day chemotherapy cycle of either (i) eribulin (1.4 mg/m2 on days 1 and 8), or (ii) a taxane (docetaxel 75 mg/m2 on day 1 or paclitaxel 80 mg/m2 on days 1, 8 and 15), each being administered in combination with a dual HER2 blockade of trastuzumab plus pertuzumab.
Baseline characteristics of both groups were well balanced, with 257 (57.6%) having ER-positive disease, 292 (65.5%) visceral metastasis, and 263 (59%) with de novo stage 4 disease, explained Dr. Yamashita.
For the primary endpoint, the median progression-free survival (PFS) was 14 versus 12.9 months in the eribulin and taxane group, respectively (hazard ratio [HR] 0.95, P = .6817), confirming non-inferiority of the study regimen, he reported.
The clinical benefit rate was similar between the two groups, with an objective response rate of 76.8% in the eribulin group and 75.2% in the taxane group.
Median OS was 65.3 months in the taxane group, but has not been reached in the study group (HR 1.09).
In terms of side-effects, the incidence of treatment-emergent adverse events was similar between the eribulin and taxane groups (58.9% vs 59.2%, respectively, for grade 3 or higher).
“Skin-related adverse events (62.4% vs 40.6%), diarrhea (54.1% vs 36.6%), and edema (42.2% vs 8.5%) tend to be more common with taxane, whereas neutropenia (61.6% vs 30.7%) and peripheral neuropathy (61.2% vs 52.8%) tend to be more common with eribulin use,” he said.
Overall, “these results suggest that eribulin is less toxic chemotherapeutic partner for dual HER2 blockade and can be used for a longer,” he said.
Findings Are a ‘Clinical Pearl’
Harold Burstein, MD, PhD, a breast cancer expert at Dana-Farber Cancer Institute and professor at Harvard Medical School in Boston, described the findings as “a nice clinical pearl,” because some patients do not tolerate taxane therapy. “In such cases, you can substitute eribulin, which is usually tolerated without allergic hypersensitivity issues,” he said in an interview.
Eribulin has specific properties that “could make it a perfect candidate” as an adjunct to standard treatment regimens across different breast cancer subtypes, observed Wynne Wijaya, MD an oncology researcher at the University of Oxford, England, and Universitas Gadjah Mada, in Yogyakarta, Indonesia, in a recent review (World J Exp Med. 2024;14[2]:92558).
Dr. Wijaya, who was not involved in this study, said in an interview that the findings have important implications.
“This encouraging result adds eribulin as another option in the first line treatment regimen for patients with HER2-positive, locally advanced or metastatic breast cancer, especially in terms of side effects/toxicities,” she said. “As clinicians, we can offer to tailor the choice of therapy between eribulin versus taxane in the regimen based on [which side effects patients are better able to tolerate]. It would also be interesting and worthwhile to conduct similar trials in different types of populations to provide more robust evidence.”
Eisai Co. funded the research. Dr. Yamashita disclosed ties with AstraZeneca, Chugai Pharma, Daiichi Sankyo, Eisai, Kyowa Hakko Kiri, Lilly, MSD, Pfizer, Taiho, Gilead Sciences, Nihonkayaku, Ono Yakuhin, and Seagen. Dr. Burstein disclosed a research grant from National Cancer Institute. Dr. Wijaya had no relevant disclosures.
FROM ASCO 2024
Survey: Ob.Gyn.s Cite Burnout as Major Practice Issue
Nearly half (46%) of ob.gyn.s, and physicians generally, said in a new survey their employers were not paying enough attention to the extent of burnout among their physicians.
Findings of the survey were reported in Medscape’s Ob/Gyn Practice Issues Report 2024.
“It’s a big problem,” said Nigel Girgrah, MD, PhD, chief wellness officer at Ochsner Health in New Orleans, Louisiana. But he added he expects that 46% level to drop in the next few years.
“There’s an increasing awareness among executives in healthcare that well-being is a leading quality indicator, if not the indicator, of favorable outcomes in patient care, staff turnover, and other vital areas,” he said.
However, there are structural barriers that threaten work-life balance, says Eve Espey, MD, MPH, chair of the Department of Obstetrics and Gynecology at the University of New Mexico in Albuquerque. In a specialty made up mostly of women, she notes, there is no federal policy on paid medical leave, leading to women ob.gyn.s pulling double duty at work and at home.
“To me, the most important way to engage on burnout is not offering yoga lessons; it’s dealing with these big structural issues,” she said.
What’s Driving the Burnout
Ob.gyn. survey respondents said the top two reasons for burnout are too many bureaucratic tasks, such as paperwork and charting, (cited by 62%) and too many hours at work (cited by 53%).
More than half (55%) said the paperwork burden had increased in the past year; 34% said it stayed the same; and 11% said it had decreased.
“I think the regulatory burden has gotten higher for most of my colleagues,” Dr. Espey, said. “Coding and billing rules are pretty opaque to most ob.gyn.s and a lot of the tasks that used to be done by medical assistants are now done by physicians.”
‘Big Hope’ Is AI
“The big hope on the horizon is AI and programs that do basically what scribes do,” Dr. Espey said. “People feel like that will be a game-changer when it comes to paperwork.”
She said the root problem is that EHRs are used mostly for billing, not for communication. Two thirds of the respondents said they sometimes or always copy and paste from charts, which can lead to note bloat.
“You can’t write concise notes in EHRs,” Dr. Espey said. “I think this is an area where the promise of AI would be very helpful.”
Nearly 40% of ob.gyn.s were enthusiastic about the potential for AI in the survey; 43% were neutral on the subject; and the remainder were apprehensive. Asked about how their practices have most commonly used AI so far, 23% said it was used for researching conditions. The next highest usage was for help in diagnosing conditions (mentioned by 13%).
More than half (53%) of the ob.gyn.s said they thought AI would free up more time for patient conversations and care while only 38% of doctors generally thought it would.
Hard to Spend More Time With One Patient
Nearly three in 10 ob.gyn.s said they have a patient quota at their workplace, compared with one in four for physicians generally.
“Clearly, there can be thresholds (for ob.gyn.s) in the office and ambulatory care settings,” said Catherine Cansino, MD, MPH, a Sacramento ob.gyn. affiliated with University of California–Davis Medical Center. She added that on the inpatient side, patient quotas come from the allotted time an ob.gyn. can spend with a patient. “There are definitely hoops to jump through when we want to spend more time with a patient, and it can be tough for them to get a follow-up appointment.”
Most ob.gyn.s (62%) in the survey would give their employer good ratings for offering an engaging and welcoming atmosphere. But nearly 40% said their employer emphasizes patient quotas and CPT codes more than care quality.
Data for this report were drawn from several Medscape surveys performed between March and October of 2023.
Nearly half (46%) of ob.gyn.s, and physicians generally, said in a new survey their employers were not paying enough attention to the extent of burnout among their physicians.
Findings of the survey were reported in Medscape’s Ob/Gyn Practice Issues Report 2024.
“It’s a big problem,” said Nigel Girgrah, MD, PhD, chief wellness officer at Ochsner Health in New Orleans, Louisiana. But he added he expects that 46% level to drop in the next few years.
“There’s an increasing awareness among executives in healthcare that well-being is a leading quality indicator, if not the indicator, of favorable outcomes in patient care, staff turnover, and other vital areas,” he said.
However, there are structural barriers that threaten work-life balance, says Eve Espey, MD, MPH, chair of the Department of Obstetrics and Gynecology at the University of New Mexico in Albuquerque. In a specialty made up mostly of women, she notes, there is no federal policy on paid medical leave, leading to women ob.gyn.s pulling double duty at work and at home.
“To me, the most important way to engage on burnout is not offering yoga lessons; it’s dealing with these big structural issues,” she said.
What’s Driving the Burnout
Ob.gyn. survey respondents said the top two reasons for burnout are too many bureaucratic tasks, such as paperwork and charting, (cited by 62%) and too many hours at work (cited by 53%).
More than half (55%) said the paperwork burden had increased in the past year; 34% said it stayed the same; and 11% said it had decreased.
“I think the regulatory burden has gotten higher for most of my colleagues,” Dr. Espey, said. “Coding and billing rules are pretty opaque to most ob.gyn.s and a lot of the tasks that used to be done by medical assistants are now done by physicians.”
‘Big Hope’ Is AI
“The big hope on the horizon is AI and programs that do basically what scribes do,” Dr. Espey said. “People feel like that will be a game-changer when it comes to paperwork.”
She said the root problem is that EHRs are used mostly for billing, not for communication. Two thirds of the respondents said they sometimes or always copy and paste from charts, which can lead to note bloat.
“You can’t write concise notes in EHRs,” Dr. Espey said. “I think this is an area where the promise of AI would be very helpful.”
Nearly 40% of ob.gyn.s were enthusiastic about the potential for AI in the survey; 43% were neutral on the subject; and the remainder were apprehensive. Asked about how their practices have most commonly used AI so far, 23% said it was used for researching conditions. The next highest usage was for help in diagnosing conditions (mentioned by 13%).
More than half (53%) of the ob.gyn.s said they thought AI would free up more time for patient conversations and care while only 38% of doctors generally thought it would.
Hard to Spend More Time With One Patient
Nearly three in 10 ob.gyn.s said they have a patient quota at their workplace, compared with one in four for physicians generally.
“Clearly, there can be thresholds (for ob.gyn.s) in the office and ambulatory care settings,” said Catherine Cansino, MD, MPH, a Sacramento ob.gyn. affiliated with University of California–Davis Medical Center. She added that on the inpatient side, patient quotas come from the allotted time an ob.gyn. can spend with a patient. “There are definitely hoops to jump through when we want to spend more time with a patient, and it can be tough for them to get a follow-up appointment.”
Most ob.gyn.s (62%) in the survey would give their employer good ratings for offering an engaging and welcoming atmosphere. But nearly 40% said their employer emphasizes patient quotas and CPT codes more than care quality.
Data for this report were drawn from several Medscape surveys performed between March and October of 2023.
Nearly half (46%) of ob.gyn.s, and physicians generally, said in a new survey their employers were not paying enough attention to the extent of burnout among their physicians.
Findings of the survey were reported in Medscape’s Ob/Gyn Practice Issues Report 2024.
“It’s a big problem,” said Nigel Girgrah, MD, PhD, chief wellness officer at Ochsner Health in New Orleans, Louisiana. But he added he expects that 46% level to drop in the next few years.
“There’s an increasing awareness among executives in healthcare that well-being is a leading quality indicator, if not the indicator, of favorable outcomes in patient care, staff turnover, and other vital areas,” he said.
However, there are structural barriers that threaten work-life balance, says Eve Espey, MD, MPH, chair of the Department of Obstetrics and Gynecology at the University of New Mexico in Albuquerque. In a specialty made up mostly of women, she notes, there is no federal policy on paid medical leave, leading to women ob.gyn.s pulling double duty at work and at home.
“To me, the most important way to engage on burnout is not offering yoga lessons; it’s dealing with these big structural issues,” she said.
What’s Driving the Burnout
Ob.gyn. survey respondents said the top two reasons for burnout are too many bureaucratic tasks, such as paperwork and charting, (cited by 62%) and too many hours at work (cited by 53%).
More than half (55%) said the paperwork burden had increased in the past year; 34% said it stayed the same; and 11% said it had decreased.
“I think the regulatory burden has gotten higher for most of my colleagues,” Dr. Espey, said. “Coding and billing rules are pretty opaque to most ob.gyn.s and a lot of the tasks that used to be done by medical assistants are now done by physicians.”
‘Big Hope’ Is AI
“The big hope on the horizon is AI and programs that do basically what scribes do,” Dr. Espey said. “People feel like that will be a game-changer when it comes to paperwork.”
She said the root problem is that EHRs are used mostly for billing, not for communication. Two thirds of the respondents said they sometimes or always copy and paste from charts, which can lead to note bloat.
“You can’t write concise notes in EHRs,” Dr. Espey said. “I think this is an area where the promise of AI would be very helpful.”
Nearly 40% of ob.gyn.s were enthusiastic about the potential for AI in the survey; 43% were neutral on the subject; and the remainder were apprehensive. Asked about how their practices have most commonly used AI so far, 23% said it was used for researching conditions. The next highest usage was for help in diagnosing conditions (mentioned by 13%).
More than half (53%) of the ob.gyn.s said they thought AI would free up more time for patient conversations and care while only 38% of doctors generally thought it would.
Hard to Spend More Time With One Patient
Nearly three in 10 ob.gyn.s said they have a patient quota at their workplace, compared with one in four for physicians generally.
“Clearly, there can be thresholds (for ob.gyn.s) in the office and ambulatory care settings,” said Catherine Cansino, MD, MPH, a Sacramento ob.gyn. affiliated with University of California–Davis Medical Center. She added that on the inpatient side, patient quotas come from the allotted time an ob.gyn. can spend with a patient. “There are definitely hoops to jump through when we want to spend more time with a patient, and it can be tough for them to get a follow-up appointment.”
Most ob.gyn.s (62%) in the survey would give their employer good ratings for offering an engaging and welcoming atmosphere. But nearly 40% said their employer emphasizes patient quotas and CPT codes more than care quality.
Data for this report were drawn from several Medscape surveys performed between March and October of 2023.
PFS Benefits Seen With Palbociclib + Endocrine Therapy in Breast Cancer
“The combination of palbociclib plus exemestane plus leuprolide showed a consistent significant improvement in PFS [progression-free survival] compared to the capecitabine arm,” Yeon Hee Park, MD, PhD, from Samsung Medical Center, Sungkyunkwan University, Seoul, South Korea, reported at the annual meeting of the American Society of Clinical Oncology.
Study Methods and Results
Young-PEARL, a prospective, multicenter, open-label, randomized phase 2 study, included 184 patients, median age 44 years, who had relapsed or progressed during previous tamoxifen therapy, with one line of previous chemotherapy for mBC allowed. Patients were randomized to palbociclib plus endocrine therapy (oral palbociclib 125 mg per day for 21 days every 4 weeks, oral exemestane 25 mg per day for 28 days, plus leuprolide 3.75 mg subcutaneously every 4 weeks) or chemotherapy (oral capecitabine 1250 mg/m2, twice daily for 2 weeks every 3 weeks).
Previously published initial results (Lancet Oncol. 2019 Dec;20[12]:1750-1759) for the primary endpoint showed a median PFS of 20.1 months in the palbociclib group versus 14.4 months in the capecitabine group, (hazard ratio [HR] 0.659, P = .0235) after median follow-up of 17 months.
Updated results showed this benefit was maintained after a median of 54 months, with a PFS of 19.5 months in the palbociclib arm, versus 14 months in capecitabine arm (HR 0.744, P = .0357), Dr. Park reported. However, this PFS benefit did not lead to an overall survival (OS) benefit, with median OS being similar: 54.8 versus 57.8 months in the palbociclib and capecitabine groups, respectively (HR = 1.02, P = .92).
To explore why PFS — but not OS — was better in the palbociclib arm, the researchers conducted a multivariate analysis which showed that going on to an additional CDK4/6 inhibitor treatment after the end of the study was as an independent variable favoring OS. Because more patients in the capecitabine arm received a post-study CDK4/6 inhibitor (49.3%) compared with in the palbociclib group (15%), this weighted the OS to the capecitabine arm, Dr. Park explained in an interview.
“In the capecitabine arm, excluding post-study CDK4/6 inhibitor use, the median OS was 38.8 months.” This was inferior to the 49 months OS seen in the palbociclib arm (P = .065), she said.
“As expected, hematologic toxicity was more common in the palbociclib arm compared with in the capecitabine arm,” Dr. Park said (92% vs 86%), with neutropenia topping the list [of all adverse events] (65.2% vs 27.9%, all grades). However, “most [adverse events] were not that serious,” Dr. Park said. Arthralgia was more common in the palbociclib arm (25% vs 7%), and diarrhea and hand-foot syndrome were more common in the capecitabine arm (15.2% vs 39.5% and 79.1% vs 2.2%).
Study Validates Endocrine Therapy + CDK4/6 Inhibitor as First Line
Commenting on Young-PEARL in an interview, Harold Burstein, MD, PhD, said, “The point of this study was to compare whether upfront chemotherapy would be better than upfront hormonal therapy for patients who had metastatic ER positive breast cancer.”
“This is the first study in probably 20 years that has compared these two approaches, and it validated that for the vast majority of patients with ER positive metastatic breast cancer, the appropriate first treatment is endocrine therapy with a CDK4/6 inhibitor,” continued Dr. Burstein, a breast cancer expert at Dana-Farber Cancer Institute, and professor at Harvard Medical School in Boston.
Dr. Park disclosed honoraria from AstraZeneca, Daiichi Sankyo, Eisai, Lilly, MSD, Novartis, Pfizer, and Roche; consulting or advisory roles for AstraZeneca, Boryung, Daiichi Sankyo, Eisai, Gilead Sciences, Lilly, Menarini, MSD, Novartis, Pfizer, and Roche; research funding from AstraZeneca, Gencurix, Genome Insight, NGeneBio, Pfizer; and Roche; and travel/accommodations/expenses from Gilead. Dr. Burstein disclosed a research grant from the National Cancer Institute.
“The combination of palbociclib plus exemestane plus leuprolide showed a consistent significant improvement in PFS [progression-free survival] compared to the capecitabine arm,” Yeon Hee Park, MD, PhD, from Samsung Medical Center, Sungkyunkwan University, Seoul, South Korea, reported at the annual meeting of the American Society of Clinical Oncology.
Study Methods and Results
Young-PEARL, a prospective, multicenter, open-label, randomized phase 2 study, included 184 patients, median age 44 years, who had relapsed or progressed during previous tamoxifen therapy, with one line of previous chemotherapy for mBC allowed. Patients were randomized to palbociclib plus endocrine therapy (oral palbociclib 125 mg per day for 21 days every 4 weeks, oral exemestane 25 mg per day for 28 days, plus leuprolide 3.75 mg subcutaneously every 4 weeks) or chemotherapy (oral capecitabine 1250 mg/m2, twice daily for 2 weeks every 3 weeks).
Previously published initial results (Lancet Oncol. 2019 Dec;20[12]:1750-1759) for the primary endpoint showed a median PFS of 20.1 months in the palbociclib group versus 14.4 months in the capecitabine group, (hazard ratio [HR] 0.659, P = .0235) after median follow-up of 17 months.
Updated results showed this benefit was maintained after a median of 54 months, with a PFS of 19.5 months in the palbociclib arm, versus 14 months in capecitabine arm (HR 0.744, P = .0357), Dr. Park reported. However, this PFS benefit did not lead to an overall survival (OS) benefit, with median OS being similar: 54.8 versus 57.8 months in the palbociclib and capecitabine groups, respectively (HR = 1.02, P = .92).
To explore why PFS — but not OS — was better in the palbociclib arm, the researchers conducted a multivariate analysis which showed that going on to an additional CDK4/6 inhibitor treatment after the end of the study was as an independent variable favoring OS. Because more patients in the capecitabine arm received a post-study CDK4/6 inhibitor (49.3%) compared with in the palbociclib group (15%), this weighted the OS to the capecitabine arm, Dr. Park explained in an interview.
“In the capecitabine arm, excluding post-study CDK4/6 inhibitor use, the median OS was 38.8 months.” This was inferior to the 49 months OS seen in the palbociclib arm (P = .065), she said.
“As expected, hematologic toxicity was more common in the palbociclib arm compared with in the capecitabine arm,” Dr. Park said (92% vs 86%), with neutropenia topping the list [of all adverse events] (65.2% vs 27.9%, all grades). However, “most [adverse events] were not that serious,” Dr. Park said. Arthralgia was more common in the palbociclib arm (25% vs 7%), and diarrhea and hand-foot syndrome were more common in the capecitabine arm (15.2% vs 39.5% and 79.1% vs 2.2%).
Study Validates Endocrine Therapy + CDK4/6 Inhibitor as First Line
Commenting on Young-PEARL in an interview, Harold Burstein, MD, PhD, said, “The point of this study was to compare whether upfront chemotherapy would be better than upfront hormonal therapy for patients who had metastatic ER positive breast cancer.”
“This is the first study in probably 20 years that has compared these two approaches, and it validated that for the vast majority of patients with ER positive metastatic breast cancer, the appropriate first treatment is endocrine therapy with a CDK4/6 inhibitor,” continued Dr. Burstein, a breast cancer expert at Dana-Farber Cancer Institute, and professor at Harvard Medical School in Boston.
Dr. Park disclosed honoraria from AstraZeneca, Daiichi Sankyo, Eisai, Lilly, MSD, Novartis, Pfizer, and Roche; consulting or advisory roles for AstraZeneca, Boryung, Daiichi Sankyo, Eisai, Gilead Sciences, Lilly, Menarini, MSD, Novartis, Pfizer, and Roche; research funding from AstraZeneca, Gencurix, Genome Insight, NGeneBio, Pfizer; and Roche; and travel/accommodations/expenses from Gilead. Dr. Burstein disclosed a research grant from the National Cancer Institute.
“The combination of palbociclib plus exemestane plus leuprolide showed a consistent significant improvement in PFS [progression-free survival] compared to the capecitabine arm,” Yeon Hee Park, MD, PhD, from Samsung Medical Center, Sungkyunkwan University, Seoul, South Korea, reported at the annual meeting of the American Society of Clinical Oncology.
Study Methods and Results
Young-PEARL, a prospective, multicenter, open-label, randomized phase 2 study, included 184 patients, median age 44 years, who had relapsed or progressed during previous tamoxifen therapy, with one line of previous chemotherapy for mBC allowed. Patients were randomized to palbociclib plus endocrine therapy (oral palbociclib 125 mg per day for 21 days every 4 weeks, oral exemestane 25 mg per day for 28 days, plus leuprolide 3.75 mg subcutaneously every 4 weeks) or chemotherapy (oral capecitabine 1250 mg/m2, twice daily for 2 weeks every 3 weeks).
Previously published initial results (Lancet Oncol. 2019 Dec;20[12]:1750-1759) for the primary endpoint showed a median PFS of 20.1 months in the palbociclib group versus 14.4 months in the capecitabine group, (hazard ratio [HR] 0.659, P = .0235) after median follow-up of 17 months.
Updated results showed this benefit was maintained after a median of 54 months, with a PFS of 19.5 months in the palbociclib arm, versus 14 months in capecitabine arm (HR 0.744, P = .0357), Dr. Park reported. However, this PFS benefit did not lead to an overall survival (OS) benefit, with median OS being similar: 54.8 versus 57.8 months in the palbociclib and capecitabine groups, respectively (HR = 1.02, P = .92).
To explore why PFS — but not OS — was better in the palbociclib arm, the researchers conducted a multivariate analysis which showed that going on to an additional CDK4/6 inhibitor treatment after the end of the study was as an independent variable favoring OS. Because more patients in the capecitabine arm received a post-study CDK4/6 inhibitor (49.3%) compared with in the palbociclib group (15%), this weighted the OS to the capecitabine arm, Dr. Park explained in an interview.
“In the capecitabine arm, excluding post-study CDK4/6 inhibitor use, the median OS was 38.8 months.” This was inferior to the 49 months OS seen in the palbociclib arm (P = .065), she said.
“As expected, hematologic toxicity was more common in the palbociclib arm compared with in the capecitabine arm,” Dr. Park said (92% vs 86%), with neutropenia topping the list [of all adverse events] (65.2% vs 27.9%, all grades). However, “most [adverse events] were not that serious,” Dr. Park said. Arthralgia was more common in the palbociclib arm (25% vs 7%), and diarrhea and hand-foot syndrome were more common in the capecitabine arm (15.2% vs 39.5% and 79.1% vs 2.2%).
Study Validates Endocrine Therapy + CDK4/6 Inhibitor as First Line
Commenting on Young-PEARL in an interview, Harold Burstein, MD, PhD, said, “The point of this study was to compare whether upfront chemotherapy would be better than upfront hormonal therapy for patients who had metastatic ER positive breast cancer.”
“This is the first study in probably 20 years that has compared these two approaches, and it validated that for the vast majority of patients with ER positive metastatic breast cancer, the appropriate first treatment is endocrine therapy with a CDK4/6 inhibitor,” continued Dr. Burstein, a breast cancer expert at Dana-Farber Cancer Institute, and professor at Harvard Medical School in Boston.
Dr. Park disclosed honoraria from AstraZeneca, Daiichi Sankyo, Eisai, Lilly, MSD, Novartis, Pfizer, and Roche; consulting or advisory roles for AstraZeneca, Boryung, Daiichi Sankyo, Eisai, Gilead Sciences, Lilly, Menarini, MSD, Novartis, Pfizer, and Roche; research funding from AstraZeneca, Gencurix, Genome Insight, NGeneBio, Pfizer; and Roche; and travel/accommodations/expenses from Gilead. Dr. Burstein disclosed a research grant from the National Cancer Institute.
FROM ASCO 2024
New Canadian BC Guidelines Emphasize Personal Choice
The draft guidelines stem from a review of more than 165 recent randomized controlled trials, observational studies, mathematical models, and other data.
The guideline working group included four breast cancer experts (a medical oncologist, a radiation oncologist, a surgical oncologist, and a radiologist), three patient partners, six family physicians, a nurse practitioner, evidence review teams, and other experts.
To avoid potential conflicts of interest, the oncologists provided input but did not vote on the final recommendations, Guylène Thériault, MD, a family physician and chair of the task force and Breast Cancer Working Group, said in an interview.
The guideline recommends that, after the potential benefits and harms of screening have been considered, mammography should be accessible every 2-3 years to women (ie, people assigned female at birth) between ages 40 and 74 years who are at average or moderately increased risk.
Women with a personal or extensive family history of breast cancer or genetic mutations that would increase breast cancer risk; those who have symptoms, such as a lump; those who feel they may be at high risk; and those who are transgender women should consult a healthcare provider about appropriate options, according to the updated guidelines, which do not apply to these patients.
The draft guidelines were published online on May 30 and are open for public comment until August 30.
‘Three Big Questions’
To develop the guidelines, the work group asked “three big questions,” said Dr. Thériault. The first was the effectiveness of breast cancer screening for women aged 40 years and over. For this question, this systematic review, unlike the 2018 guideline update, included not only randomized trials but also observational data to ensure that the work group considered all available data.
“The second question was about comparative effectiveness,” which is something the United States considered for the latest US Preventive Services Task Force (USPSTF) update, said Dr. Thériault. The USPSTF asked questions such as “What happens if we start screening patients at age 40 years? Or at age 50 years? What happens if we stop at age 74 years? Or if we use different tests such as 3D versus digital mammography?”
The Canadian Task Force relied on the evidence that the USPSTF found after grading it with its own criteria, she said. The results were similar, and so are the recommendations in this area. “For example, we don’t recommend supplementary screening for women with dense breasts because there are no studies to inform patient-oriented benefits.”
The third question was about the values and preferences of women regarding breast cancer screening, which is something the United States didn’t examine. “We had looked at that issue in 2018, and this time around, even though we expanded the type of studies, we got the same message: That there are differences between women in their 40s and those who are age 50 years and over.”
“The majority of women in their 40s think that the harms outweigh the benefits and are not interested in screening,” said Dr. Thériault. “But when I say the majority, that’s not every woman. So, we had to recognize that there is variability. And the majority, but not all, of women ages 50-74 years thinks the benefits are higher than the harms. That’s why we say in our recommendation that from ages 40 to 74, it’s a personal choice.”
Responding to Objections
Not surprisingly, the task force has heard objections to its draft guidelines. The first is that women aged 40-49 years are being denied mammograms, said Michelle Nadler, MD, a medical oncologist at Princess Margaret Cancer Centre in Toronto, Canada. “This [objection] has attained a lot of media coverage, which is unfortunate, because people who have not read the guidelines may believe this is true. The guidelines clearly state that an eligible, informed woman of this age group who wants a screening mammogram should receive one.”
The second commonly heard objection is that the task force is overestimating the harms of screening, such as anxiety and overdiagnosis, she said. But an outcome of “anxiety” was not factored into the guideline. Overdiagnosis was calculated on the basis of the literature, and estimates were converted to a common denominator so that they could be compared, said Dr. Nadler. The same was true of benefits.
Another objection was that screening could mean less need for chemotherapy or full axillary dissection, Dr. Nadler said. However, the task force did not find any primary studies that evaluated these outcomes.
Critics also said that the recommendations do not account for racial or ethnic variations. Although more research is likely needed in this area, “the task force states that individuals should be informed of all of their breast cancer risk factors, including race/ethnicity, and that this should be factored into decisions about screening,” said Dr. Nadler.
“I was very surprised that the task force was accused by some parties of paternalism,” added René Wittmer, MD, adjunct clinical professor of family medicine at the University of Montreal and chair of Choosing Wisely Quebec, Montreal, Canada. “In my opinion, the importance they place on shared decision-making is contrary to medical paternalism and aims to empower women to make a decision that fits with their values and preferences.”
Nevertheless, the inclusion of modeling studies and observational trials “may cause the potential benefits to be amplified, compared with what is seen in randomized controlled trials,” he said in an interview.
Decision Aids Help
Once the guidelines are finalized, decision aids will be available to patients and providers to help guide screening discussions, said Dr. Nadler. “Primary care providers need to be aware of an individual’s personal risk factors for breast cancer to know if they are at average, above average, or high lifetime risk of breast cancer. These guidelines do not apply to those with > 20% lifetime risk of breast cancer.”
“The standards for risk communication are in absolute numbers over a common denominator,” she noted. “This is how primary care providers discuss other important primary care topics like smoking cessation, cardiovascular disease (and decisions about statin medications), and osteoporosis risk. These same standards should apply for breast cancer screening.”
Furthermore, she said, providers “should be aware that individuals from marginalized communities may benefit from more than one conversation until they are able to make a decision about screening that is right for them.”
“There is good evidence showing that most advances we’ve seen in breast cancer outcomes (ie, reduction in breast cancer mortality) are likely due to improvements in treatment, not screening,” said Dr. Wittmer. “In fact, mortality reductions are seen even in age groups or countries where there is no routine screening. This means that women benefit from advances in treatments, whether they choose to get screened or not.”
‘Mammography Saves Lives’
Commenting on the updated guidelines, Janie Lee, MD, professor of radiology at the University of Washington School of Medicine and director of breast imaging at the Fred Hutchinson Cancer Center, both in Seattle, said: “For the USPSTF, benefits of life years gained were also considered, in addition to breast cancer deaths averted. To save more lives from breast cancer, guidelines may focus on screening women at older ages, when annual rates of breast cancer are higher.” By contrast, when thinking in terms of years of life saved, focusing on screening younger women, who have more years of life left, increases benefits. “Both are important outcomes that we want to improve with effective screening.”
That said, “we should follow the guidelines of our specific national organizations,” she continued. “Overall populations and healthcare systems are different between the US and Canada.”
For example, “the USPSTF specifically highlighted the potential for reducing breast cancer mortality in Black women, who are more likely to develop biologically aggressive tumors that are diagnosed at more advanced stages, when making updated recommendations earlier this year,” she said. “The Canadian guidelines did not make specific recommendations by race or ethnicity group, instead highlighting the need for more research on the impact of screening in these groups.”
In addition, “screening every year versus every other year is more routine in the US compared with Canada,” she noted. And nonmedical factors that influence health and that may influence access to medical care and timely diagnosis of breast cancer “may be different between our two countries.”
“The most important take-home message is that the scientific evidence is strong that screening mammography saves lives,” said Dr. Lee. “These new recommendations will hopefully result in more early diagnoses of breast cancer and save more lives. Screening works best when it’s used regularly, regardless of how frequently you return. Once you start screening, please urge your patients to plan to return.”
Dr. Nadler disclosed speaker honoraria and consulting fees from Novartis and Exact Sciences outside the scope of this interview and innovation funding from the NSH/UHN AMO Innovation Fund Competition for Developing and Implementing a Consensus Recommendation for Breast Cancer Screening Best Practices. Dr. Thériault is chair of the task force and chair of the working group for the draft guidelines. Dr. Wittmer is chair of Choosing Wisely Quebec. Dr. Lee reported no relevant financial relationships related to her interview.
A version of this article appeared on Medscape.com.
The draft guidelines stem from a review of more than 165 recent randomized controlled trials, observational studies, mathematical models, and other data.
The guideline working group included four breast cancer experts (a medical oncologist, a radiation oncologist, a surgical oncologist, and a radiologist), three patient partners, six family physicians, a nurse practitioner, evidence review teams, and other experts.
To avoid potential conflicts of interest, the oncologists provided input but did not vote on the final recommendations, Guylène Thériault, MD, a family physician and chair of the task force and Breast Cancer Working Group, said in an interview.
The guideline recommends that, after the potential benefits and harms of screening have been considered, mammography should be accessible every 2-3 years to women (ie, people assigned female at birth) between ages 40 and 74 years who are at average or moderately increased risk.
Women with a personal or extensive family history of breast cancer or genetic mutations that would increase breast cancer risk; those who have symptoms, such as a lump; those who feel they may be at high risk; and those who are transgender women should consult a healthcare provider about appropriate options, according to the updated guidelines, which do not apply to these patients.
The draft guidelines were published online on May 30 and are open for public comment until August 30.
‘Three Big Questions’
To develop the guidelines, the work group asked “three big questions,” said Dr. Thériault. The first was the effectiveness of breast cancer screening for women aged 40 years and over. For this question, this systematic review, unlike the 2018 guideline update, included not only randomized trials but also observational data to ensure that the work group considered all available data.
“The second question was about comparative effectiveness,” which is something the United States considered for the latest US Preventive Services Task Force (USPSTF) update, said Dr. Thériault. The USPSTF asked questions such as “What happens if we start screening patients at age 40 years? Or at age 50 years? What happens if we stop at age 74 years? Or if we use different tests such as 3D versus digital mammography?”
The Canadian Task Force relied on the evidence that the USPSTF found after grading it with its own criteria, she said. The results were similar, and so are the recommendations in this area. “For example, we don’t recommend supplementary screening for women with dense breasts because there are no studies to inform patient-oriented benefits.”
The third question was about the values and preferences of women regarding breast cancer screening, which is something the United States didn’t examine. “We had looked at that issue in 2018, and this time around, even though we expanded the type of studies, we got the same message: That there are differences between women in their 40s and those who are age 50 years and over.”
“The majority of women in their 40s think that the harms outweigh the benefits and are not interested in screening,” said Dr. Thériault. “But when I say the majority, that’s not every woman. So, we had to recognize that there is variability. And the majority, but not all, of women ages 50-74 years thinks the benefits are higher than the harms. That’s why we say in our recommendation that from ages 40 to 74, it’s a personal choice.”
Responding to Objections
Not surprisingly, the task force has heard objections to its draft guidelines. The first is that women aged 40-49 years are being denied mammograms, said Michelle Nadler, MD, a medical oncologist at Princess Margaret Cancer Centre in Toronto, Canada. “This [objection] has attained a lot of media coverage, which is unfortunate, because people who have not read the guidelines may believe this is true. The guidelines clearly state that an eligible, informed woman of this age group who wants a screening mammogram should receive one.”
The second commonly heard objection is that the task force is overestimating the harms of screening, such as anxiety and overdiagnosis, she said. But an outcome of “anxiety” was not factored into the guideline. Overdiagnosis was calculated on the basis of the literature, and estimates were converted to a common denominator so that they could be compared, said Dr. Nadler. The same was true of benefits.
Another objection was that screening could mean less need for chemotherapy or full axillary dissection, Dr. Nadler said. However, the task force did not find any primary studies that evaluated these outcomes.
Critics also said that the recommendations do not account for racial or ethnic variations. Although more research is likely needed in this area, “the task force states that individuals should be informed of all of their breast cancer risk factors, including race/ethnicity, and that this should be factored into decisions about screening,” said Dr. Nadler.
“I was very surprised that the task force was accused by some parties of paternalism,” added René Wittmer, MD, adjunct clinical professor of family medicine at the University of Montreal and chair of Choosing Wisely Quebec, Montreal, Canada. “In my opinion, the importance they place on shared decision-making is contrary to medical paternalism and aims to empower women to make a decision that fits with their values and preferences.”
Nevertheless, the inclusion of modeling studies and observational trials “may cause the potential benefits to be amplified, compared with what is seen in randomized controlled trials,” he said in an interview.
Decision Aids Help
Once the guidelines are finalized, decision aids will be available to patients and providers to help guide screening discussions, said Dr. Nadler. “Primary care providers need to be aware of an individual’s personal risk factors for breast cancer to know if they are at average, above average, or high lifetime risk of breast cancer. These guidelines do not apply to those with > 20% lifetime risk of breast cancer.”
“The standards for risk communication are in absolute numbers over a common denominator,” she noted. “This is how primary care providers discuss other important primary care topics like smoking cessation, cardiovascular disease (and decisions about statin medications), and osteoporosis risk. These same standards should apply for breast cancer screening.”
Furthermore, she said, providers “should be aware that individuals from marginalized communities may benefit from more than one conversation until they are able to make a decision about screening that is right for them.”
“There is good evidence showing that most advances we’ve seen in breast cancer outcomes (ie, reduction in breast cancer mortality) are likely due to improvements in treatment, not screening,” said Dr. Wittmer. “In fact, mortality reductions are seen even in age groups or countries where there is no routine screening. This means that women benefit from advances in treatments, whether they choose to get screened or not.”
‘Mammography Saves Lives’
Commenting on the updated guidelines, Janie Lee, MD, professor of radiology at the University of Washington School of Medicine and director of breast imaging at the Fred Hutchinson Cancer Center, both in Seattle, said: “For the USPSTF, benefits of life years gained were also considered, in addition to breast cancer deaths averted. To save more lives from breast cancer, guidelines may focus on screening women at older ages, when annual rates of breast cancer are higher.” By contrast, when thinking in terms of years of life saved, focusing on screening younger women, who have more years of life left, increases benefits. “Both are important outcomes that we want to improve with effective screening.”
That said, “we should follow the guidelines of our specific national organizations,” she continued. “Overall populations and healthcare systems are different between the US and Canada.”
For example, “the USPSTF specifically highlighted the potential for reducing breast cancer mortality in Black women, who are more likely to develop biologically aggressive tumors that are diagnosed at more advanced stages, when making updated recommendations earlier this year,” she said. “The Canadian guidelines did not make specific recommendations by race or ethnicity group, instead highlighting the need for more research on the impact of screening in these groups.”
In addition, “screening every year versus every other year is more routine in the US compared with Canada,” she noted. And nonmedical factors that influence health and that may influence access to medical care and timely diagnosis of breast cancer “may be different between our two countries.”
“The most important take-home message is that the scientific evidence is strong that screening mammography saves lives,” said Dr. Lee. “These new recommendations will hopefully result in more early diagnoses of breast cancer and save more lives. Screening works best when it’s used regularly, regardless of how frequently you return. Once you start screening, please urge your patients to plan to return.”
Dr. Nadler disclosed speaker honoraria and consulting fees from Novartis and Exact Sciences outside the scope of this interview and innovation funding from the NSH/UHN AMO Innovation Fund Competition for Developing and Implementing a Consensus Recommendation for Breast Cancer Screening Best Practices. Dr. Thériault is chair of the task force and chair of the working group for the draft guidelines. Dr. Wittmer is chair of Choosing Wisely Quebec. Dr. Lee reported no relevant financial relationships related to her interview.
A version of this article appeared on Medscape.com.
The draft guidelines stem from a review of more than 165 recent randomized controlled trials, observational studies, mathematical models, and other data.
The guideline working group included four breast cancer experts (a medical oncologist, a radiation oncologist, a surgical oncologist, and a radiologist), three patient partners, six family physicians, a nurse practitioner, evidence review teams, and other experts.
To avoid potential conflicts of interest, the oncologists provided input but did not vote on the final recommendations, Guylène Thériault, MD, a family physician and chair of the task force and Breast Cancer Working Group, said in an interview.
The guideline recommends that, after the potential benefits and harms of screening have been considered, mammography should be accessible every 2-3 years to women (ie, people assigned female at birth) between ages 40 and 74 years who are at average or moderately increased risk.
Women with a personal or extensive family history of breast cancer or genetic mutations that would increase breast cancer risk; those who have symptoms, such as a lump; those who feel they may be at high risk; and those who are transgender women should consult a healthcare provider about appropriate options, according to the updated guidelines, which do not apply to these patients.
The draft guidelines were published online on May 30 and are open for public comment until August 30.
‘Three Big Questions’
To develop the guidelines, the work group asked “three big questions,” said Dr. Thériault. The first was the effectiveness of breast cancer screening for women aged 40 years and over. For this question, this systematic review, unlike the 2018 guideline update, included not only randomized trials but also observational data to ensure that the work group considered all available data.
“The second question was about comparative effectiveness,” which is something the United States considered for the latest US Preventive Services Task Force (USPSTF) update, said Dr. Thériault. The USPSTF asked questions such as “What happens if we start screening patients at age 40 years? Or at age 50 years? What happens if we stop at age 74 years? Or if we use different tests such as 3D versus digital mammography?”
The Canadian Task Force relied on the evidence that the USPSTF found after grading it with its own criteria, she said. The results were similar, and so are the recommendations in this area. “For example, we don’t recommend supplementary screening for women with dense breasts because there are no studies to inform patient-oriented benefits.”
The third question was about the values and preferences of women regarding breast cancer screening, which is something the United States didn’t examine. “We had looked at that issue in 2018, and this time around, even though we expanded the type of studies, we got the same message: That there are differences between women in their 40s and those who are age 50 years and over.”
“The majority of women in their 40s think that the harms outweigh the benefits and are not interested in screening,” said Dr. Thériault. “But when I say the majority, that’s not every woman. So, we had to recognize that there is variability. And the majority, but not all, of women ages 50-74 years thinks the benefits are higher than the harms. That’s why we say in our recommendation that from ages 40 to 74, it’s a personal choice.”
Responding to Objections
Not surprisingly, the task force has heard objections to its draft guidelines. The first is that women aged 40-49 years are being denied mammograms, said Michelle Nadler, MD, a medical oncologist at Princess Margaret Cancer Centre in Toronto, Canada. “This [objection] has attained a lot of media coverage, which is unfortunate, because people who have not read the guidelines may believe this is true. The guidelines clearly state that an eligible, informed woman of this age group who wants a screening mammogram should receive one.”
The second commonly heard objection is that the task force is overestimating the harms of screening, such as anxiety and overdiagnosis, she said. But an outcome of “anxiety” was not factored into the guideline. Overdiagnosis was calculated on the basis of the literature, and estimates were converted to a common denominator so that they could be compared, said Dr. Nadler. The same was true of benefits.
Another objection was that screening could mean less need for chemotherapy or full axillary dissection, Dr. Nadler said. However, the task force did not find any primary studies that evaluated these outcomes.
Critics also said that the recommendations do not account for racial or ethnic variations. Although more research is likely needed in this area, “the task force states that individuals should be informed of all of their breast cancer risk factors, including race/ethnicity, and that this should be factored into decisions about screening,” said Dr. Nadler.
“I was very surprised that the task force was accused by some parties of paternalism,” added René Wittmer, MD, adjunct clinical professor of family medicine at the University of Montreal and chair of Choosing Wisely Quebec, Montreal, Canada. “In my opinion, the importance they place on shared decision-making is contrary to medical paternalism and aims to empower women to make a decision that fits with their values and preferences.”
Nevertheless, the inclusion of modeling studies and observational trials “may cause the potential benefits to be amplified, compared with what is seen in randomized controlled trials,” he said in an interview.
Decision Aids Help
Once the guidelines are finalized, decision aids will be available to patients and providers to help guide screening discussions, said Dr. Nadler. “Primary care providers need to be aware of an individual’s personal risk factors for breast cancer to know if they are at average, above average, or high lifetime risk of breast cancer. These guidelines do not apply to those with > 20% lifetime risk of breast cancer.”
“The standards for risk communication are in absolute numbers over a common denominator,” she noted. “This is how primary care providers discuss other important primary care topics like smoking cessation, cardiovascular disease (and decisions about statin medications), and osteoporosis risk. These same standards should apply for breast cancer screening.”
Furthermore, she said, providers “should be aware that individuals from marginalized communities may benefit from more than one conversation until they are able to make a decision about screening that is right for them.”
“There is good evidence showing that most advances we’ve seen in breast cancer outcomes (ie, reduction in breast cancer mortality) are likely due to improvements in treatment, not screening,” said Dr. Wittmer. “In fact, mortality reductions are seen even in age groups or countries where there is no routine screening. This means that women benefit from advances in treatments, whether they choose to get screened or not.”
‘Mammography Saves Lives’
Commenting on the updated guidelines, Janie Lee, MD, professor of radiology at the University of Washington School of Medicine and director of breast imaging at the Fred Hutchinson Cancer Center, both in Seattle, said: “For the USPSTF, benefits of life years gained were also considered, in addition to breast cancer deaths averted. To save more lives from breast cancer, guidelines may focus on screening women at older ages, when annual rates of breast cancer are higher.” By contrast, when thinking in terms of years of life saved, focusing on screening younger women, who have more years of life left, increases benefits. “Both are important outcomes that we want to improve with effective screening.”
That said, “we should follow the guidelines of our specific national organizations,” she continued. “Overall populations and healthcare systems are different between the US and Canada.”
For example, “the USPSTF specifically highlighted the potential for reducing breast cancer mortality in Black women, who are more likely to develop biologically aggressive tumors that are diagnosed at more advanced stages, when making updated recommendations earlier this year,” she said. “The Canadian guidelines did not make specific recommendations by race or ethnicity group, instead highlighting the need for more research on the impact of screening in these groups.”
In addition, “screening every year versus every other year is more routine in the US compared with Canada,” she noted. And nonmedical factors that influence health and that may influence access to medical care and timely diagnosis of breast cancer “may be different between our two countries.”
“The most important take-home message is that the scientific evidence is strong that screening mammography saves lives,” said Dr. Lee. “These new recommendations will hopefully result in more early diagnoses of breast cancer and save more lives. Screening works best when it’s used regularly, regardless of how frequently you return. Once you start screening, please urge your patients to plan to return.”
Dr. Nadler disclosed speaker honoraria and consulting fees from Novartis and Exact Sciences outside the scope of this interview and innovation funding from the NSH/UHN AMO Innovation Fund Competition for Developing and Implementing a Consensus Recommendation for Breast Cancer Screening Best Practices. Dr. Thériault is chair of the task force and chair of the working group for the draft guidelines. Dr. Wittmer is chair of Choosing Wisely Quebec. Dr. Lee reported no relevant financial relationships related to her interview.
A version of this article appeared on Medscape.com.
A Doctor’s Guide to Relocation
Moving for any new opportunity in medicine can feel like starting a new life, not just a new job. This is especially true for residency or fellowships, as taking a step forward in your career is exciting. But in the process, you may be leaving family and friends for an unknown city or region where you will need to find a community. And the changes could be long-term. According to the Association of American Medical Colleges’ 2023 Report on Residents, 57.1% of the individuals who completed residency training between 2013 and 2022 are still practicing in the state where they completed their residency.
The process of planning out the right timeline; securing a comfortable, convenient, and affordable place to live; and meeting people while working long hours in an unfamiliar location can be overwhelming. And in the case of many residency programs and healthcare settings, financial assistance, relocation information, and other resources are scarce.
This news organization spoke to recent residents and medical school faculty members about how to navigate a medical move and set yourself up for success.
1. Find Relocation Resources
First things first. Find out what your program or hospital has to offer.
Some institutions help incoming residents by providing housing options or information. The Icahn School of Medicine at Mount Sinai’s Real Estate Division, for example, provides off-campus housing resources that guide new residents and faculty toward safe, convenient places to live in New York City. It also guarantees on-campus or block-leased housing offers to all incoming residents who apply.
Michael Leitman, MD, FACS, professor of surgery and medical education and dean for Graduate Medical Education at the Icahn School of Medicine at Mount Sinai in New York City, recommends connecting with colleagues at your program for guidance on navigating a new city and a new healthcare setting. He encourages incoming residents to use the contact information they receive during the interview and orientation processes to reach out to co-residents and faculty members.
Other residency programs offer partial reimbursement or need-based financial aid to help with the expense of relocation. But this is unlikely to cover all or even most of the cost of a cross-country move.
When Morgen Owens, MD, moved from Alabama to New York City for a physical medicine and rehabilitation residency at Mount Sinai in 2021, her program offered subsidized housing options. But there was little reimbursement for relocation. She paid around $3000 for a one-way rental truck, gas, one night in a hotel, and movers to unload her belongings. She says driving herself kept the price down because full-service movers would have cost her between $4000 and $6000.
If this will strain your finances, several banks offer loans specifically for medical school graduates to cover residency and internship expenses. But be aware that these loans tend to have higher interest rates than federal student loans because they are based on credit score rather than fixed.
2. Reach Out and Buddy Up
Reaching out to more senior residents is essential, and some programs facilitate a buddy system for relocation advice.
Family physician Mursal Sekandari, MD, known as “Dr. Mursi,” attended a residency program at St. Luke’s University Hospital–Bethlehem Campus, in Bethlehem, Pennsylvania. The program’s official buddy system paired her with a senior resident who advised her on the area and gave tips for her apartment search.
On the other hand, when America Revere, MD, moved from Texas to Georgia for a surgery residency, she found that her program offered little relocation assistance, financial or otherwise. She leaned on her co-residents, and especially senior ones, for support while she settled in.
Dr. Revere also discovered the importance of accepting invitations to events hosted by both her fellow residents and her program itself, especially in the early stages of residency. “Accepting social invitations is really the only way to get to know people,” she said. “Sure, you’ll meet people at work and get to know their ‘work’ personalities.” But Dr. Revere’s attendings also threw parties, which she says were a great way to connect with a wider group and build a community.
To meet people both within and beyond her own residency program, Dr. Owens joined a group chat for physical medicine and rehab residents in the New York City area. She suggests looking into GroupMe or WhatsApp groups specific to your specialty.
3. Play the ‘Doctor Card’
Finding a place to live in an unfamiliar and competitive housing market can be one of the biggest challenges of any move. Dr. Owens’ options were limited by owning a dog, which wouldn’t be allowed in her hospital’s subsidized housing. Instead, she opted to find her own apartment in New York City. Her strategy: Playing the “doctor card.”
“I explained my situation: ‘I’m a doctor moving from out of state,’ ” Owens said. “Own that! These companies and brokers will look at you as a student and think, ‘Oh, she has no money, she has no savings, she’s got all of these loans, how is she going to pay for this apartment?’ But you have to say, ‘I’m a doctor. I’m an incoming resident who has X amount of years of job security. I’m not going to lose my job while living here.’ ”
4. Move Early
Dr. Revere found it important to move into her new home 2 weeks before the start of her residency program. Moving in early allowed her to settle in, get to know her area, neighbors, and co-residents, and generally prepare for her first day. It also gave her time to put furniture together — her new vanity alone took 12 hours.
Having a larger window of time before residency can also benefit those who hire movers or have their furniture shipped. When it comes to a cross-country move, it can take a few days to a few weeks for the truck to arrive — which could translate to a few nights or a few weeks without a bed.
“When residency comes, it comes fast,” Dr. Revere said. “It’s very confusing, and the last thing you need is to have half of your stuff unpacked or have no idea where you are or know nobody around you.”
5. Make Your New Home Your Sanctuary
During the stress of residency, your home can be a source of peace, and finding that might require trade-offs.
Dr. Sekandari’s parents urged her to live with roommates to save money on rent, but she insisted that spending more for solitude would be worth it. For her first year of residency, she barely saw her apartment. But when she did, she felt grateful to be in such a tranquil place to ease some of the stress of studying. “If you feel uncomfortable while you’re dealing with something stressful, the stress just exponentially increases,” she said. Creating an environment where you can really relax “makes a difference in how you respond to everything else around you.”
Dr. Revere agrees, urging medical professionals — and particularly residents — to invest in the most comfortable mattresses and bedding they can. Whether you are working nights, she also recommends blackout curtains to help facilitate daytime naps or better sleep in general, especially among the bright lights of bigger cities.
“You’re going to need somewhere to decompress,” she said. “That will look different for everyone. But I would definitely invest in your apartment to make it a sanctuary away from work.”
6. Consider a ‘Live’ Stress Reliever
When it comes to crucial stress relief during residency, “I like mine live,” Dr. Revere said in a YouTube vlog while petting her cat, Calyx.
Taking on the added responsibility of a pet during residency or any medical role may seem counterintuitive. But Revere has zero regrets about bringing Calyx along on her journey. “Cats are very easy,” she said. “I have nothing but wonderful things to say about having a cat during my difficult surgical residency.”
Dr. Owens admits that moving to New York City with her dog was difficult during her first years of residency. She worked an average of 80 hours each week and had little time for walks. She made room in her budget for dog walkers. Thankfully, her hours have eased up as she has progressed through her program, and she can now take her dog on longer walks every day. “He definitely has a better life now that I work fewer hours,” she said.
Once you’ve prepared, made the move, and found your village, it’s time for the real work to begin. “The first couple of months are certainly a challenge of adjusting to a new hospital, a new electronic medical record, a new culture, and a new geographic location,” said Dr. Leitman, who has relocated several times. “But at the end of the day ... it’s you and the patient.” By minimizing stress and getting the support you need, it can even be “a fun process,” Dr. Mursi added, “so make it an exciting chapter in your life.”
A version of this article first appeared on Medscape.com.
Moving for any new opportunity in medicine can feel like starting a new life, not just a new job. This is especially true for residency or fellowships, as taking a step forward in your career is exciting. But in the process, you may be leaving family and friends for an unknown city or region where you will need to find a community. And the changes could be long-term. According to the Association of American Medical Colleges’ 2023 Report on Residents, 57.1% of the individuals who completed residency training between 2013 and 2022 are still practicing in the state where they completed their residency.
The process of planning out the right timeline; securing a comfortable, convenient, and affordable place to live; and meeting people while working long hours in an unfamiliar location can be overwhelming. And in the case of many residency programs and healthcare settings, financial assistance, relocation information, and other resources are scarce.
This news organization spoke to recent residents and medical school faculty members about how to navigate a medical move and set yourself up for success.
1. Find Relocation Resources
First things first. Find out what your program or hospital has to offer.
Some institutions help incoming residents by providing housing options or information. The Icahn School of Medicine at Mount Sinai’s Real Estate Division, for example, provides off-campus housing resources that guide new residents and faculty toward safe, convenient places to live in New York City. It also guarantees on-campus or block-leased housing offers to all incoming residents who apply.
Michael Leitman, MD, FACS, professor of surgery and medical education and dean for Graduate Medical Education at the Icahn School of Medicine at Mount Sinai in New York City, recommends connecting with colleagues at your program for guidance on navigating a new city and a new healthcare setting. He encourages incoming residents to use the contact information they receive during the interview and orientation processes to reach out to co-residents and faculty members.
Other residency programs offer partial reimbursement or need-based financial aid to help with the expense of relocation. But this is unlikely to cover all or even most of the cost of a cross-country move.
When Morgen Owens, MD, moved from Alabama to New York City for a physical medicine and rehabilitation residency at Mount Sinai in 2021, her program offered subsidized housing options. But there was little reimbursement for relocation. She paid around $3000 for a one-way rental truck, gas, one night in a hotel, and movers to unload her belongings. She says driving herself kept the price down because full-service movers would have cost her between $4000 and $6000.
If this will strain your finances, several banks offer loans specifically for medical school graduates to cover residency and internship expenses. But be aware that these loans tend to have higher interest rates than federal student loans because they are based on credit score rather than fixed.
2. Reach Out and Buddy Up
Reaching out to more senior residents is essential, and some programs facilitate a buddy system for relocation advice.
Family physician Mursal Sekandari, MD, known as “Dr. Mursi,” attended a residency program at St. Luke’s University Hospital–Bethlehem Campus, in Bethlehem, Pennsylvania. The program’s official buddy system paired her with a senior resident who advised her on the area and gave tips for her apartment search.
On the other hand, when America Revere, MD, moved from Texas to Georgia for a surgery residency, she found that her program offered little relocation assistance, financial or otherwise. She leaned on her co-residents, and especially senior ones, for support while she settled in.
Dr. Revere also discovered the importance of accepting invitations to events hosted by both her fellow residents and her program itself, especially in the early stages of residency. “Accepting social invitations is really the only way to get to know people,” she said. “Sure, you’ll meet people at work and get to know their ‘work’ personalities.” But Dr. Revere’s attendings also threw parties, which she says were a great way to connect with a wider group and build a community.
To meet people both within and beyond her own residency program, Dr. Owens joined a group chat for physical medicine and rehab residents in the New York City area. She suggests looking into GroupMe or WhatsApp groups specific to your specialty.
3. Play the ‘Doctor Card’
Finding a place to live in an unfamiliar and competitive housing market can be one of the biggest challenges of any move. Dr. Owens’ options were limited by owning a dog, which wouldn’t be allowed in her hospital’s subsidized housing. Instead, she opted to find her own apartment in New York City. Her strategy: Playing the “doctor card.”
“I explained my situation: ‘I’m a doctor moving from out of state,’ ” Owens said. “Own that! These companies and brokers will look at you as a student and think, ‘Oh, she has no money, she has no savings, she’s got all of these loans, how is she going to pay for this apartment?’ But you have to say, ‘I’m a doctor. I’m an incoming resident who has X amount of years of job security. I’m not going to lose my job while living here.’ ”
4. Move Early
Dr. Revere found it important to move into her new home 2 weeks before the start of her residency program. Moving in early allowed her to settle in, get to know her area, neighbors, and co-residents, and generally prepare for her first day. It also gave her time to put furniture together — her new vanity alone took 12 hours.
Having a larger window of time before residency can also benefit those who hire movers or have their furniture shipped. When it comes to a cross-country move, it can take a few days to a few weeks for the truck to arrive — which could translate to a few nights or a few weeks without a bed.
“When residency comes, it comes fast,” Dr. Revere said. “It’s very confusing, and the last thing you need is to have half of your stuff unpacked or have no idea where you are or know nobody around you.”
5. Make Your New Home Your Sanctuary
During the stress of residency, your home can be a source of peace, and finding that might require trade-offs.
Dr. Sekandari’s parents urged her to live with roommates to save money on rent, but she insisted that spending more for solitude would be worth it. For her first year of residency, she barely saw her apartment. But when she did, she felt grateful to be in such a tranquil place to ease some of the stress of studying. “If you feel uncomfortable while you’re dealing with something stressful, the stress just exponentially increases,” she said. Creating an environment where you can really relax “makes a difference in how you respond to everything else around you.”
Dr. Revere agrees, urging medical professionals — and particularly residents — to invest in the most comfortable mattresses and bedding they can. Whether you are working nights, she also recommends blackout curtains to help facilitate daytime naps or better sleep in general, especially among the bright lights of bigger cities.
“You’re going to need somewhere to decompress,” she said. “That will look different for everyone. But I would definitely invest in your apartment to make it a sanctuary away from work.”
6. Consider a ‘Live’ Stress Reliever
When it comes to crucial stress relief during residency, “I like mine live,” Dr. Revere said in a YouTube vlog while petting her cat, Calyx.
Taking on the added responsibility of a pet during residency or any medical role may seem counterintuitive. But Revere has zero regrets about bringing Calyx along on her journey. “Cats are very easy,” she said. “I have nothing but wonderful things to say about having a cat during my difficult surgical residency.”
Dr. Owens admits that moving to New York City with her dog was difficult during her first years of residency. She worked an average of 80 hours each week and had little time for walks. She made room in her budget for dog walkers. Thankfully, her hours have eased up as she has progressed through her program, and she can now take her dog on longer walks every day. “He definitely has a better life now that I work fewer hours,” she said.
Once you’ve prepared, made the move, and found your village, it’s time for the real work to begin. “The first couple of months are certainly a challenge of adjusting to a new hospital, a new electronic medical record, a new culture, and a new geographic location,” said Dr. Leitman, who has relocated several times. “But at the end of the day ... it’s you and the patient.” By minimizing stress and getting the support you need, it can even be “a fun process,” Dr. Mursi added, “so make it an exciting chapter in your life.”
A version of this article first appeared on Medscape.com.
Moving for any new opportunity in medicine can feel like starting a new life, not just a new job. This is especially true for residency or fellowships, as taking a step forward in your career is exciting. But in the process, you may be leaving family and friends for an unknown city or region where you will need to find a community. And the changes could be long-term. According to the Association of American Medical Colleges’ 2023 Report on Residents, 57.1% of the individuals who completed residency training between 2013 and 2022 are still practicing in the state where they completed their residency.
The process of planning out the right timeline; securing a comfortable, convenient, and affordable place to live; and meeting people while working long hours in an unfamiliar location can be overwhelming. And in the case of many residency programs and healthcare settings, financial assistance, relocation information, and other resources are scarce.
This news organization spoke to recent residents and medical school faculty members about how to navigate a medical move and set yourself up for success.
1. Find Relocation Resources
First things first. Find out what your program or hospital has to offer.
Some institutions help incoming residents by providing housing options or information. The Icahn School of Medicine at Mount Sinai’s Real Estate Division, for example, provides off-campus housing resources that guide new residents and faculty toward safe, convenient places to live in New York City. It also guarantees on-campus or block-leased housing offers to all incoming residents who apply.
Michael Leitman, MD, FACS, professor of surgery and medical education and dean for Graduate Medical Education at the Icahn School of Medicine at Mount Sinai in New York City, recommends connecting with colleagues at your program for guidance on navigating a new city and a new healthcare setting. He encourages incoming residents to use the contact information they receive during the interview and orientation processes to reach out to co-residents and faculty members.
Other residency programs offer partial reimbursement or need-based financial aid to help with the expense of relocation. But this is unlikely to cover all or even most of the cost of a cross-country move.
When Morgen Owens, MD, moved from Alabama to New York City for a physical medicine and rehabilitation residency at Mount Sinai in 2021, her program offered subsidized housing options. But there was little reimbursement for relocation. She paid around $3000 for a one-way rental truck, gas, one night in a hotel, and movers to unload her belongings. She says driving herself kept the price down because full-service movers would have cost her between $4000 and $6000.
If this will strain your finances, several banks offer loans specifically for medical school graduates to cover residency and internship expenses. But be aware that these loans tend to have higher interest rates than federal student loans because they are based on credit score rather than fixed.
2. Reach Out and Buddy Up
Reaching out to more senior residents is essential, and some programs facilitate a buddy system for relocation advice.
Family physician Mursal Sekandari, MD, known as “Dr. Mursi,” attended a residency program at St. Luke’s University Hospital–Bethlehem Campus, in Bethlehem, Pennsylvania. The program’s official buddy system paired her with a senior resident who advised her on the area and gave tips for her apartment search.
On the other hand, when America Revere, MD, moved from Texas to Georgia for a surgery residency, she found that her program offered little relocation assistance, financial or otherwise. She leaned on her co-residents, and especially senior ones, for support while she settled in.
Dr. Revere also discovered the importance of accepting invitations to events hosted by both her fellow residents and her program itself, especially in the early stages of residency. “Accepting social invitations is really the only way to get to know people,” she said. “Sure, you’ll meet people at work and get to know their ‘work’ personalities.” But Dr. Revere’s attendings also threw parties, which she says were a great way to connect with a wider group and build a community.
To meet people both within and beyond her own residency program, Dr. Owens joined a group chat for physical medicine and rehab residents in the New York City area. She suggests looking into GroupMe or WhatsApp groups specific to your specialty.
3. Play the ‘Doctor Card’
Finding a place to live in an unfamiliar and competitive housing market can be one of the biggest challenges of any move. Dr. Owens’ options were limited by owning a dog, which wouldn’t be allowed in her hospital’s subsidized housing. Instead, she opted to find her own apartment in New York City. Her strategy: Playing the “doctor card.”
“I explained my situation: ‘I’m a doctor moving from out of state,’ ” Owens said. “Own that! These companies and brokers will look at you as a student and think, ‘Oh, she has no money, she has no savings, she’s got all of these loans, how is she going to pay for this apartment?’ But you have to say, ‘I’m a doctor. I’m an incoming resident who has X amount of years of job security. I’m not going to lose my job while living here.’ ”
4. Move Early
Dr. Revere found it important to move into her new home 2 weeks before the start of her residency program. Moving in early allowed her to settle in, get to know her area, neighbors, and co-residents, and generally prepare for her first day. It also gave her time to put furniture together — her new vanity alone took 12 hours.
Having a larger window of time before residency can also benefit those who hire movers or have their furniture shipped. When it comes to a cross-country move, it can take a few days to a few weeks for the truck to arrive — which could translate to a few nights or a few weeks without a bed.
“When residency comes, it comes fast,” Dr. Revere said. “It’s very confusing, and the last thing you need is to have half of your stuff unpacked or have no idea where you are or know nobody around you.”
5. Make Your New Home Your Sanctuary
During the stress of residency, your home can be a source of peace, and finding that might require trade-offs.
Dr. Sekandari’s parents urged her to live with roommates to save money on rent, but she insisted that spending more for solitude would be worth it. For her first year of residency, she barely saw her apartment. But when she did, she felt grateful to be in such a tranquil place to ease some of the stress of studying. “If you feel uncomfortable while you’re dealing with something stressful, the stress just exponentially increases,” she said. Creating an environment where you can really relax “makes a difference in how you respond to everything else around you.”
Dr. Revere agrees, urging medical professionals — and particularly residents — to invest in the most comfortable mattresses and bedding they can. Whether you are working nights, she also recommends blackout curtains to help facilitate daytime naps or better sleep in general, especially among the bright lights of bigger cities.
“You’re going to need somewhere to decompress,” she said. “That will look different for everyone. But I would definitely invest in your apartment to make it a sanctuary away from work.”
6. Consider a ‘Live’ Stress Reliever
When it comes to crucial stress relief during residency, “I like mine live,” Dr. Revere said in a YouTube vlog while petting her cat, Calyx.
Taking on the added responsibility of a pet during residency or any medical role may seem counterintuitive. But Revere has zero regrets about bringing Calyx along on her journey. “Cats are very easy,” she said. “I have nothing but wonderful things to say about having a cat during my difficult surgical residency.”
Dr. Owens admits that moving to New York City with her dog was difficult during her first years of residency. She worked an average of 80 hours each week and had little time for walks. She made room in her budget for dog walkers. Thankfully, her hours have eased up as she has progressed through her program, and she can now take her dog on longer walks every day. “He definitely has a better life now that I work fewer hours,” she said.
Once you’ve prepared, made the move, and found your village, it’s time for the real work to begin. “The first couple of months are certainly a challenge of adjusting to a new hospital, a new electronic medical record, a new culture, and a new geographic location,” said Dr. Leitman, who has relocated several times. “But at the end of the day ... it’s you and the patient.” By minimizing stress and getting the support you need, it can even be “a fun process,” Dr. Mursi added, “so make it an exciting chapter in your life.”
A version of this article first appeared on Medscape.com.
Confronting Healthcare Disinformation on Social Media
More than 90% of internet users are active on social media, which had 4.76 billion users worldwide in January 2023. The digital revolution has reshaped the news landscape and changed how users interact with information. Social media has fostered an active relationship with the media, including the ability to interact directly with the content presented. It also has augmented media’s ability to reach a large audience with tight deadlines.
These developments suggest that social media can be a useful tool in everyday medical practice for professionals and patients. But social media also can spread misinformation, as happened during the COVID-19 pandemic.
This characteristic is the focus of the latest research by Fabiana Zollo, a computer science professor at Ca’ Foscari University of Venice, Italy, and coordinator of the Data Science for Society laboratory. The research was published in The BMJ. Ms. Zollo’s research group aims to assess the effect of social media on misinformation and consequent behaviors related to health. “The study results focus primarily on two topics, the COVID-19 pandemic and vaccinations, but can also be applied to other health-related behaviors such as smoking and diet,” Ms. Zollo told Univadis Italy.
Social media has become an important tool for public health organizations to inform and educate citizens. Institutions can use it to monitor choices and understand which topics are being discussed most at a given time, thus comprehending how the topics evolve and take shape in public discourse. “This could lead to the emergence of people’s perceptions, allowing us to understand, among other things, what the population’s needs might be, including informational needs,” said Ms. Zollo.
Tenuous Causal Link
While social media offers public health organizations the opportunity to inform and engage the public, it also raises concerns about misinformation and the difficulty of measuring its effect on health behavior. Although some studies have observed correlations between exposure to misinformation on social media and levels of adherence to vaccination campaigns, establishing a causal link is complex. As the authors emphasize, “despite the importance of the effect of social media and misinformation on people’s behavior and the broad hypotheses within public and political debates, the current state of the art cannot provide definitive conclusions on a clear causal association between social media and health behaviors.” Establishing a clear causal link between information obtained from social media and offline behavior is challenging due to methodologic limitations and the complexity of connections between online and offline behaviors. Studies often rely on self-reported data, which may not accurately reflect real behaviors, and struggle to isolate the effect of social media from other external influences. Moreover, many studies primarily focus on Western countries, limiting the generalizability of the results to other cultural and geographical conditions.
Another issue highlighted by Ms. Zollo and colleagues is the lack of complete and representative data. Studies often lack detailed information about participants, such as demographic or geolocation data, and rely on limited samples. This lack makes it difficult to assess the effect of misinformation on different segments of the population and in different geographic areas.
“The main methodologic difficulty concerns behavior, which is difficult to measure because it would require tracking a person’s actions over time and having a shared methodology to do so. We need to understand whether online stated intentions do or do not translate into actual behaviors,” said Ms. Zollo. Therefore, despite the recognized importance of the effect of social media and misinformation on people’s general behavior and the broad hypotheses expressed within public and political debates, the current state of the art cannot provide definitive conclusions on a causal association between social media and health behaviors.
Institutions’ Role
Social media is a fertile ground for the formation of echo chambers (where users find themselves dialoguing with like-minded people, forming a distorted impression of the real prevalence of that opinion) and for reinforcing polarized positions around certain topics. “We know that on certain topics, especially those related to health, there is a lot of misinformation circulating precisely because it is easy to leverage factors such as fear and beliefs, even the difficulties in understanding the technical aspects of a message,” said Ms. Zollo. Moreover, institutions have not always provided timely information during the pandemic. “Often, when there is a gap in response to a specific informational need, people turn elsewhere, where those questions find answers. And even if the response is not of high quality, it sometimes confirms the idea that the user had already created in their mind.”
The article published in The BMJ aims primarily to provide information and evaluation insights to institutions rather than professionals or healthcare workers. “We would like to spark the interest of institutions and ministries that can analyze this type of data and integrate it into their monitoring system. Social monitoring (the observation of what happens on social media) is a practice that the World Health Organization is also evaluating and trying to integrate with more traditional tools, such as questionnaires. The aim is to understand as well as possible what a population thinks about a particular health measure, such as a vaccine: Through data obtained from social monitoring, a more realistic and comprehensive view of the problem could be achieved,” said Ms. Zollo.
A Doctor’s Role
And this is where the doctor comes in: All the information thus obtained allows for identifying the needs that the population expresses and that “could push a patient to turn elsewhere, toward sources that provide answers even if of dubious quality or extremely oversimplified.” The doctor can enter this landscape by trying to understand, even with the data provided by institutions, what needs the patients are trying to fill and what drives them to seek elsewhere and to look for a reference community that offers the relevant confirmations.
From the doctor’s perspective, therefore, it can be useful to understand how these dynamics arise and evolve because they could help improve interactions with patients. At the institutional level, social monitoring would be an excellent tool for providing services to doctors who, in turn, offer a service to patients. If it were possible to identify areas where a disinformation narrative is developing from the outset, both the doctor and the institutions would benefit.
Misinformation vs Disinformation
The rapid spread of false or misleading information on social media can undermine trust in healthcare institutions and negatively influence health-related behaviors. Ms. Zollo and colleagues, in fact, speak of misinformation in their discussion, not disinformation. “In English, a distinction is made between misinformation and disinformation, a distinction that we are also adopting in Italian. When we talk about misinformation, we mean information that is generally false, inaccurate, or misleading but has not been created with the intention to harm, an intention that is present in disinformation,” said Ms. Zollo.
The distinction is often not easy to define even at the operational level, but in her studies, Ms. Zollo is mainly interested in understanding how the end user interacts with content, not the purposes for which that content was created. “This allows us to focus on users and the relationships that are created on various social platforms, thus bypassing the author of that information and focusing on how misinformation arises and evolves so that it can be effectively combated before it translates into action (ie, into incorrect health choices),” said Ms. Zollo.
This story was translated from Univadis Italy, which is part of the Medscape Professional Network, using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
More than 90% of internet users are active on social media, which had 4.76 billion users worldwide in January 2023. The digital revolution has reshaped the news landscape and changed how users interact with information. Social media has fostered an active relationship with the media, including the ability to interact directly with the content presented. It also has augmented media’s ability to reach a large audience with tight deadlines.
These developments suggest that social media can be a useful tool in everyday medical practice for professionals and patients. But social media also can spread misinformation, as happened during the COVID-19 pandemic.
This characteristic is the focus of the latest research by Fabiana Zollo, a computer science professor at Ca’ Foscari University of Venice, Italy, and coordinator of the Data Science for Society laboratory. The research was published in The BMJ. Ms. Zollo’s research group aims to assess the effect of social media on misinformation and consequent behaviors related to health. “The study results focus primarily on two topics, the COVID-19 pandemic and vaccinations, but can also be applied to other health-related behaviors such as smoking and diet,” Ms. Zollo told Univadis Italy.
Social media has become an important tool for public health organizations to inform and educate citizens. Institutions can use it to monitor choices and understand which topics are being discussed most at a given time, thus comprehending how the topics evolve and take shape in public discourse. “This could lead to the emergence of people’s perceptions, allowing us to understand, among other things, what the population’s needs might be, including informational needs,” said Ms. Zollo.
Tenuous Causal Link
While social media offers public health organizations the opportunity to inform and engage the public, it also raises concerns about misinformation and the difficulty of measuring its effect on health behavior. Although some studies have observed correlations between exposure to misinformation on social media and levels of adherence to vaccination campaigns, establishing a causal link is complex. As the authors emphasize, “despite the importance of the effect of social media and misinformation on people’s behavior and the broad hypotheses within public and political debates, the current state of the art cannot provide definitive conclusions on a clear causal association between social media and health behaviors.” Establishing a clear causal link between information obtained from social media and offline behavior is challenging due to methodologic limitations and the complexity of connections between online and offline behaviors. Studies often rely on self-reported data, which may not accurately reflect real behaviors, and struggle to isolate the effect of social media from other external influences. Moreover, many studies primarily focus on Western countries, limiting the generalizability of the results to other cultural and geographical conditions.
Another issue highlighted by Ms. Zollo and colleagues is the lack of complete and representative data. Studies often lack detailed information about participants, such as demographic or geolocation data, and rely on limited samples. This lack makes it difficult to assess the effect of misinformation on different segments of the population and in different geographic areas.
“The main methodologic difficulty concerns behavior, which is difficult to measure because it would require tracking a person’s actions over time and having a shared methodology to do so. We need to understand whether online stated intentions do or do not translate into actual behaviors,” said Ms. Zollo. Therefore, despite the recognized importance of the effect of social media and misinformation on people’s general behavior and the broad hypotheses expressed within public and political debates, the current state of the art cannot provide definitive conclusions on a causal association between social media and health behaviors.
Institutions’ Role
Social media is a fertile ground for the formation of echo chambers (where users find themselves dialoguing with like-minded people, forming a distorted impression of the real prevalence of that opinion) and for reinforcing polarized positions around certain topics. “We know that on certain topics, especially those related to health, there is a lot of misinformation circulating precisely because it is easy to leverage factors such as fear and beliefs, even the difficulties in understanding the technical aspects of a message,” said Ms. Zollo. Moreover, institutions have not always provided timely information during the pandemic. “Often, when there is a gap in response to a specific informational need, people turn elsewhere, where those questions find answers. And even if the response is not of high quality, it sometimes confirms the idea that the user had already created in their mind.”
The article published in The BMJ aims primarily to provide information and evaluation insights to institutions rather than professionals or healthcare workers. “We would like to spark the interest of institutions and ministries that can analyze this type of data and integrate it into their monitoring system. Social monitoring (the observation of what happens on social media) is a practice that the World Health Organization is also evaluating and trying to integrate with more traditional tools, such as questionnaires. The aim is to understand as well as possible what a population thinks about a particular health measure, such as a vaccine: Through data obtained from social monitoring, a more realistic and comprehensive view of the problem could be achieved,” said Ms. Zollo.
A Doctor’s Role
And this is where the doctor comes in: All the information thus obtained allows for identifying the needs that the population expresses and that “could push a patient to turn elsewhere, toward sources that provide answers even if of dubious quality or extremely oversimplified.” The doctor can enter this landscape by trying to understand, even with the data provided by institutions, what needs the patients are trying to fill and what drives them to seek elsewhere and to look for a reference community that offers the relevant confirmations.
From the doctor’s perspective, therefore, it can be useful to understand how these dynamics arise and evolve because they could help improve interactions with patients. At the institutional level, social monitoring would be an excellent tool for providing services to doctors who, in turn, offer a service to patients. If it were possible to identify areas where a disinformation narrative is developing from the outset, both the doctor and the institutions would benefit.
Misinformation vs Disinformation
The rapid spread of false or misleading information on social media can undermine trust in healthcare institutions and negatively influence health-related behaviors. Ms. Zollo and colleagues, in fact, speak of misinformation in their discussion, not disinformation. “In English, a distinction is made between misinformation and disinformation, a distinction that we are also adopting in Italian. When we talk about misinformation, we mean information that is generally false, inaccurate, or misleading but has not been created with the intention to harm, an intention that is present in disinformation,” said Ms. Zollo.
The distinction is often not easy to define even at the operational level, but in her studies, Ms. Zollo is mainly interested in understanding how the end user interacts with content, not the purposes for which that content was created. “This allows us to focus on users and the relationships that are created on various social platforms, thus bypassing the author of that information and focusing on how misinformation arises and evolves so that it can be effectively combated before it translates into action (ie, into incorrect health choices),” said Ms. Zollo.
This story was translated from Univadis Italy, which is part of the Medscape Professional Network, using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
More than 90% of internet users are active on social media, which had 4.76 billion users worldwide in January 2023. The digital revolution has reshaped the news landscape and changed how users interact with information. Social media has fostered an active relationship with the media, including the ability to interact directly with the content presented. It also has augmented media’s ability to reach a large audience with tight deadlines.
These developments suggest that social media can be a useful tool in everyday medical practice for professionals and patients. But social media also can spread misinformation, as happened during the COVID-19 pandemic.
This characteristic is the focus of the latest research by Fabiana Zollo, a computer science professor at Ca’ Foscari University of Venice, Italy, and coordinator of the Data Science for Society laboratory. The research was published in The BMJ. Ms. Zollo’s research group aims to assess the effect of social media on misinformation and consequent behaviors related to health. “The study results focus primarily on two topics, the COVID-19 pandemic and vaccinations, but can also be applied to other health-related behaviors such as smoking and diet,” Ms. Zollo told Univadis Italy.
Social media has become an important tool for public health organizations to inform and educate citizens. Institutions can use it to monitor choices and understand which topics are being discussed most at a given time, thus comprehending how the topics evolve and take shape in public discourse. “This could lead to the emergence of people’s perceptions, allowing us to understand, among other things, what the population’s needs might be, including informational needs,” said Ms. Zollo.
Tenuous Causal Link
While social media offers public health organizations the opportunity to inform and engage the public, it also raises concerns about misinformation and the difficulty of measuring its effect on health behavior. Although some studies have observed correlations between exposure to misinformation on social media and levels of adherence to vaccination campaigns, establishing a causal link is complex. As the authors emphasize, “despite the importance of the effect of social media and misinformation on people’s behavior and the broad hypotheses within public and political debates, the current state of the art cannot provide definitive conclusions on a clear causal association between social media and health behaviors.” Establishing a clear causal link between information obtained from social media and offline behavior is challenging due to methodologic limitations and the complexity of connections between online and offline behaviors. Studies often rely on self-reported data, which may not accurately reflect real behaviors, and struggle to isolate the effect of social media from other external influences. Moreover, many studies primarily focus on Western countries, limiting the generalizability of the results to other cultural and geographical conditions.
Another issue highlighted by Ms. Zollo and colleagues is the lack of complete and representative data. Studies often lack detailed information about participants, such as demographic or geolocation data, and rely on limited samples. This lack makes it difficult to assess the effect of misinformation on different segments of the population and in different geographic areas.
“The main methodologic difficulty concerns behavior, which is difficult to measure because it would require tracking a person’s actions over time and having a shared methodology to do so. We need to understand whether online stated intentions do or do not translate into actual behaviors,” said Ms. Zollo. Therefore, despite the recognized importance of the effect of social media and misinformation on people’s general behavior and the broad hypotheses expressed within public and political debates, the current state of the art cannot provide definitive conclusions on a causal association between social media and health behaviors.
Institutions’ Role
Social media is a fertile ground for the formation of echo chambers (where users find themselves dialoguing with like-minded people, forming a distorted impression of the real prevalence of that opinion) and for reinforcing polarized positions around certain topics. “We know that on certain topics, especially those related to health, there is a lot of misinformation circulating precisely because it is easy to leverage factors such as fear and beliefs, even the difficulties in understanding the technical aspects of a message,” said Ms. Zollo. Moreover, institutions have not always provided timely information during the pandemic. “Often, when there is a gap in response to a specific informational need, people turn elsewhere, where those questions find answers. And even if the response is not of high quality, it sometimes confirms the idea that the user had already created in their mind.”
The article published in The BMJ aims primarily to provide information and evaluation insights to institutions rather than professionals or healthcare workers. “We would like to spark the interest of institutions and ministries that can analyze this type of data and integrate it into their monitoring system. Social monitoring (the observation of what happens on social media) is a practice that the World Health Organization is also evaluating and trying to integrate with more traditional tools, such as questionnaires. The aim is to understand as well as possible what a population thinks about a particular health measure, such as a vaccine: Through data obtained from social monitoring, a more realistic and comprehensive view of the problem could be achieved,” said Ms. Zollo.
A Doctor’s Role
And this is where the doctor comes in: All the information thus obtained allows for identifying the needs that the population expresses and that “could push a patient to turn elsewhere, toward sources that provide answers even if of dubious quality or extremely oversimplified.” The doctor can enter this landscape by trying to understand, even with the data provided by institutions, what needs the patients are trying to fill and what drives them to seek elsewhere and to look for a reference community that offers the relevant confirmations.
From the doctor’s perspective, therefore, it can be useful to understand how these dynamics arise and evolve because they could help improve interactions with patients. At the institutional level, social monitoring would be an excellent tool for providing services to doctors who, in turn, offer a service to patients. If it were possible to identify areas where a disinformation narrative is developing from the outset, both the doctor and the institutions would benefit.
Misinformation vs Disinformation
The rapid spread of false or misleading information on social media can undermine trust in healthcare institutions and negatively influence health-related behaviors. Ms. Zollo and colleagues, in fact, speak of misinformation in their discussion, not disinformation. “In English, a distinction is made between misinformation and disinformation, a distinction that we are also adopting in Italian. When we talk about misinformation, we mean information that is generally false, inaccurate, or misleading but has not been created with the intention to harm, an intention that is present in disinformation,” said Ms. Zollo.
The distinction is often not easy to define even at the operational level, but in her studies, Ms. Zollo is mainly interested in understanding how the end user interacts with content, not the purposes for which that content was created. “This allows us to focus on users and the relationships that are created on various social platforms, thus bypassing the author of that information and focusing on how misinformation arises and evolves so that it can be effectively combated before it translates into action (ie, into incorrect health choices),” said Ms. Zollo.
This story was translated from Univadis Italy, which is part of the Medscape Professional Network, using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.


