User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Virus and Booster Apathy Could Be Fueling Long COVID
A celebrity makeup artist, the 55-year-old New Yorker had been boosted and vaccinated at every opportunity since vaccines were approved at the end of 2020, until the fall of 2023, when she skipped the shot.
“I really started subscribing to the mindset that you have an immune system and your immune system is supposed to work for you,” she said. “That was the stupidest thing I’ve ever done.”
Maio was not the only person to skip the latest booster: A recent study reported that while nearly 80% of adults in the United States said they’d received their first series of vaccines, barely 20% were up to date on boosters. Nor was Maio alone in getting long COVID 4 years after the start of the deadliest pandemic in a century.
It’s tempting, this far out from the shutdowns of 2020, to think the virus is over, that we’re immune, and nobody’s going to get sick anymore. But while fewer people are getting COVID, it is still very much a part of our lives. And as Maio and others are learning the hard way, long COVID is, too — and it can be deadly.
For those who have recently contracted long COVID, it can feel as if the whole world has moved on from the pandemic, and they are being left behind.
Too Easy to Let Our Guard Down
“It’s really difficult to prevent exposure to COVID no matter how careful you are and no matter how many times you are vaccinated,” said Akiko Iwasaki, an immunology professor at Yale School of Medicine, New Haven, Connecticut, and pioneer in long COVID research. Iwasaki was quick to point out that “we should never blame anybody for getting long COVID because there is no bulletproof way of preventing long COVID from happening” — although research shows you can increase your protection through vaccination, masking, and increasing ventilation indoors.
Also, just because you didn’t get long COVID after catching the virus once, doesn’t mean you’ll dodge the bullet if you get sick again, as Maio has now learned twice. She had long COVID in 2022 after her second bout with the virus, with breathing problems and brain fog that lasted for several months.
Subsequent long COVID experiences won’t necessarily mimic previous ones. Although Maio developed brain fog again, this time she didn’t have the breathing problems that plagued her in 2022. Instead, she had headaches so excruciating she thought she was dying of a brain aneurysm.
A Journal of the American Medical Association study released in May identified the 37 most common symptoms of long COVID, including symptom subgroups that occurred in 80% of the nearly 10,000 study participants. But the symptoms that patients with long COVID are experiencing now are slightly different from earlier in the pandemic or at least that’s what doctors are finding at the Post-COVID Recovery Clinic affiliated with the University of Pittsburgh Medical Center.
Michael Risbano, MD, the clinic’s codirector, said fewer patients have pulmonary or lung damage now than in the past, but a steady stream report problems with brain fog, forgetfulness, exercise intolerance (shortness of breath and fatigue with exercise and difficulty performing any kind of exertional activity), and post-exertional malaise (feeling wiped out or fatigued for hours or even days after physical or mental activity).
Long COVID Treatments Showing Improvement — Slowly
“There still isn’t a great way to treat any of this,” said Risbano, whose clinic is involved with the National Institute of Health’s RECOVER-VITAL trial, which is evaluating potential treatments including Paxlovid and exercise to treat autonomic dysfunction with similarities to myalgic encephalomyelitis/chronic fatigue syndrome and POTS, exercise intolerance, and neurocognitive effects such as brain fog.
Risbano and colleagues have found that physical therapy and exercise training have helped patients with exercise intolerance and neurocognitive problems. “It’s not a quick thing where they go through one visit and are better the next day,” he stressed. “It takes a little bit of time, a little bit of effort, a little bit of homework — there are no silver bullets, no magic medications.”
A quick fix was definitely not in the cards for Dean Jones, PhD, who could barely move when he developed long COVID in May 2023. A 74-year-old biochemist and professor of medicine at Emory University in Atlanta, Georgia, he’d recovered fully the first time he had COVID, in August 2022, but had a completely different experience the second time. He had been vaccinated four times when he began experiencing chronic fatigue, intense exertion-induced migraines, severe airway congestion, brain fog, and shortness of breath. The symptoms began after Memorial Day and worsened over the next month.
His resting heart rate began racing even when he was sleeping, jumping from 53 to 70 beats per minute. “It was almost as though the virus had hit my heart rather than the lungs alone,” he said.
Doctors prescribed multiple inhalers and glucocorticoids to calm Jones’s immune system. The worst symptoms began to abate after a few weeks. The bad ones continued for fully 2 months, severely limiting Jones’s activity. Although he no longer slept all day, just walking from one room to another was exhausting. A dedicated scientist who typically worked 10-15 hours a day before getting sick, he was lucky to focus on work-related tasks for a fraction of that time.
Although the migraines went away early on, the headaches remained until well into the fall. Jones’s energy level gradually returned, and by Christmas, he was beginning to feel as healthy as he had before getting COVID in May.
Still, he’s not complaining that it took so long to get better. “At 74, there’s a lot of colleagues who have already passed away,” he said. “I respect the realities of my age. There are so many people who died from COVID that I’m thankful I had those vaccines. I’m thankful that I pulled through it and was able to rebound.”
Time Helps Healing — But Prompt Care Still Needed
Recovery is the case for most patients with long COVID, said Lisa Sanders, MD, medical director of the Yale New Haven Health Systems Long COVID Consultation Clinic, which opened in March 2023. Although the clinic has a small segment of patients who have had the condition since 2020, “people who recover, who are most people, move on,” she said. “Even the patients who sometimes have to wait a month or so to see me, some of them say, ‘I’m already starting to get better. I wasn’t sure I should come.’”
Maio, too, is recovering but only after multiple visits to the emergency room and a neurologist in late December and early January. The third emergency room trip was prompted after a brief episode in which she lost the feeling in her legs, which began convulsing. A CAT scan showed severely constricted blood vessels in her brain, leading the medical team to speculate she might have reversible cerebral vasoconstriction syndrome (RCVS), which can trigger the thunderclap headaches that had been causing her such misery.
After her third such headache prompted a fourth emergency room visit, further tests confirmed RCVS, which doctors said was related to inflammation caused by COVID. Maio was then admitted to the hospital, where she spent 4 days starting on a regimen of blood pressure medication, magnesium for the headaches, and oxycodone for pain management.
The TV show Maio works on went back into production after the holidays. She went back at the end of January. She’s still having headaches, though they’re less intense, and she’s still taking medication. She was scheduled for another test to look at her blood vessels in February.
Maio has yet to forgive herself for skipping the last booster, even though there’s no guarantee it would have prevented her from getting sick. Her message for others: it’s better to be safe than to be as sorry as she is.
“I’ll never, ever be persuaded by people who don’t believe in vaccines because I believe in science, and I believe in vaccines — that’s why people don’t die at the age of 30 anymore,” she said. “I really think that people need to know about this and what to expect. Because it is horrendous. It is very painful. I would never want anyone to go through this. Ever.”
A version of this article appeared on Medscape.com.
A celebrity makeup artist, the 55-year-old New Yorker had been boosted and vaccinated at every opportunity since vaccines were approved at the end of 2020, until the fall of 2023, when she skipped the shot.
“I really started subscribing to the mindset that you have an immune system and your immune system is supposed to work for you,” she said. “That was the stupidest thing I’ve ever done.”
Maio was not the only person to skip the latest booster: A recent study reported that while nearly 80% of adults in the United States said they’d received their first series of vaccines, barely 20% were up to date on boosters. Nor was Maio alone in getting long COVID 4 years after the start of the deadliest pandemic in a century.
It’s tempting, this far out from the shutdowns of 2020, to think the virus is over, that we’re immune, and nobody’s going to get sick anymore. But while fewer people are getting COVID, it is still very much a part of our lives. And as Maio and others are learning the hard way, long COVID is, too — and it can be deadly.
For those who have recently contracted long COVID, it can feel as if the whole world has moved on from the pandemic, and they are being left behind.
Too Easy to Let Our Guard Down
“It’s really difficult to prevent exposure to COVID no matter how careful you are and no matter how many times you are vaccinated,” said Akiko Iwasaki, an immunology professor at Yale School of Medicine, New Haven, Connecticut, and pioneer in long COVID research. Iwasaki was quick to point out that “we should never blame anybody for getting long COVID because there is no bulletproof way of preventing long COVID from happening” — although research shows you can increase your protection through vaccination, masking, and increasing ventilation indoors.
Also, just because you didn’t get long COVID after catching the virus once, doesn’t mean you’ll dodge the bullet if you get sick again, as Maio has now learned twice. She had long COVID in 2022 after her second bout with the virus, with breathing problems and brain fog that lasted for several months.
Subsequent long COVID experiences won’t necessarily mimic previous ones. Although Maio developed brain fog again, this time she didn’t have the breathing problems that plagued her in 2022. Instead, she had headaches so excruciating she thought she was dying of a brain aneurysm.
A Journal of the American Medical Association study released in May identified the 37 most common symptoms of long COVID, including symptom subgroups that occurred in 80% of the nearly 10,000 study participants. But the symptoms that patients with long COVID are experiencing now are slightly different from earlier in the pandemic or at least that’s what doctors are finding at the Post-COVID Recovery Clinic affiliated with the University of Pittsburgh Medical Center.
Michael Risbano, MD, the clinic’s codirector, said fewer patients have pulmonary or lung damage now than in the past, but a steady stream report problems with brain fog, forgetfulness, exercise intolerance (shortness of breath and fatigue with exercise and difficulty performing any kind of exertional activity), and post-exertional malaise (feeling wiped out or fatigued for hours or even days after physical or mental activity).
Long COVID Treatments Showing Improvement — Slowly
“There still isn’t a great way to treat any of this,” said Risbano, whose clinic is involved with the National Institute of Health’s RECOVER-VITAL trial, which is evaluating potential treatments including Paxlovid and exercise to treat autonomic dysfunction with similarities to myalgic encephalomyelitis/chronic fatigue syndrome and POTS, exercise intolerance, and neurocognitive effects such as brain fog.
Risbano and colleagues have found that physical therapy and exercise training have helped patients with exercise intolerance and neurocognitive problems. “It’s not a quick thing where they go through one visit and are better the next day,” he stressed. “It takes a little bit of time, a little bit of effort, a little bit of homework — there are no silver bullets, no magic medications.”
A quick fix was definitely not in the cards for Dean Jones, PhD, who could barely move when he developed long COVID in May 2023. A 74-year-old biochemist and professor of medicine at Emory University in Atlanta, Georgia, he’d recovered fully the first time he had COVID, in August 2022, but had a completely different experience the second time. He had been vaccinated four times when he began experiencing chronic fatigue, intense exertion-induced migraines, severe airway congestion, brain fog, and shortness of breath. The symptoms began after Memorial Day and worsened over the next month.
His resting heart rate began racing even when he was sleeping, jumping from 53 to 70 beats per minute. “It was almost as though the virus had hit my heart rather than the lungs alone,” he said.
Doctors prescribed multiple inhalers and glucocorticoids to calm Jones’s immune system. The worst symptoms began to abate after a few weeks. The bad ones continued for fully 2 months, severely limiting Jones’s activity. Although he no longer slept all day, just walking from one room to another was exhausting. A dedicated scientist who typically worked 10-15 hours a day before getting sick, he was lucky to focus on work-related tasks for a fraction of that time.
Although the migraines went away early on, the headaches remained until well into the fall. Jones’s energy level gradually returned, and by Christmas, he was beginning to feel as healthy as he had before getting COVID in May.
Still, he’s not complaining that it took so long to get better. “At 74, there’s a lot of colleagues who have already passed away,” he said. “I respect the realities of my age. There are so many people who died from COVID that I’m thankful I had those vaccines. I’m thankful that I pulled through it and was able to rebound.”
Time Helps Healing — But Prompt Care Still Needed
Recovery is the case for most patients with long COVID, said Lisa Sanders, MD, medical director of the Yale New Haven Health Systems Long COVID Consultation Clinic, which opened in March 2023. Although the clinic has a small segment of patients who have had the condition since 2020, “people who recover, who are most people, move on,” she said. “Even the patients who sometimes have to wait a month or so to see me, some of them say, ‘I’m already starting to get better. I wasn’t sure I should come.’”
Maio, too, is recovering but only after multiple visits to the emergency room and a neurologist in late December and early January. The third emergency room trip was prompted after a brief episode in which she lost the feeling in her legs, which began convulsing. A CAT scan showed severely constricted blood vessels in her brain, leading the medical team to speculate she might have reversible cerebral vasoconstriction syndrome (RCVS), which can trigger the thunderclap headaches that had been causing her such misery.
After her third such headache prompted a fourth emergency room visit, further tests confirmed RCVS, which doctors said was related to inflammation caused by COVID. Maio was then admitted to the hospital, where she spent 4 days starting on a regimen of blood pressure medication, magnesium for the headaches, and oxycodone for pain management.
The TV show Maio works on went back into production after the holidays. She went back at the end of January. She’s still having headaches, though they’re less intense, and she’s still taking medication. She was scheduled for another test to look at her blood vessels in February.
Maio has yet to forgive herself for skipping the last booster, even though there’s no guarantee it would have prevented her from getting sick. Her message for others: it’s better to be safe than to be as sorry as she is.
“I’ll never, ever be persuaded by people who don’t believe in vaccines because I believe in science, and I believe in vaccines — that’s why people don’t die at the age of 30 anymore,” she said. “I really think that people need to know about this and what to expect. Because it is horrendous. It is very painful. I would never want anyone to go through this. Ever.”
A version of this article appeared on Medscape.com.
A celebrity makeup artist, the 55-year-old New Yorker had been boosted and vaccinated at every opportunity since vaccines were approved at the end of 2020, until the fall of 2023, when she skipped the shot.
“I really started subscribing to the mindset that you have an immune system and your immune system is supposed to work for you,” she said. “That was the stupidest thing I’ve ever done.”
Maio was not the only person to skip the latest booster: A recent study reported that while nearly 80% of adults in the United States said they’d received their first series of vaccines, barely 20% were up to date on boosters. Nor was Maio alone in getting long COVID 4 years after the start of the deadliest pandemic in a century.
It’s tempting, this far out from the shutdowns of 2020, to think the virus is over, that we’re immune, and nobody’s going to get sick anymore. But while fewer people are getting COVID, it is still very much a part of our lives. And as Maio and others are learning the hard way, long COVID is, too — and it can be deadly.
For those who have recently contracted long COVID, it can feel as if the whole world has moved on from the pandemic, and they are being left behind.
Too Easy to Let Our Guard Down
“It’s really difficult to prevent exposure to COVID no matter how careful you are and no matter how many times you are vaccinated,” said Akiko Iwasaki, an immunology professor at Yale School of Medicine, New Haven, Connecticut, and pioneer in long COVID research. Iwasaki was quick to point out that “we should never blame anybody for getting long COVID because there is no bulletproof way of preventing long COVID from happening” — although research shows you can increase your protection through vaccination, masking, and increasing ventilation indoors.
Also, just because you didn’t get long COVID after catching the virus once, doesn’t mean you’ll dodge the bullet if you get sick again, as Maio has now learned twice. She had long COVID in 2022 after her second bout with the virus, with breathing problems and brain fog that lasted for several months.
Subsequent long COVID experiences won’t necessarily mimic previous ones. Although Maio developed brain fog again, this time she didn’t have the breathing problems that plagued her in 2022. Instead, she had headaches so excruciating she thought she was dying of a brain aneurysm.
A Journal of the American Medical Association study released in May identified the 37 most common symptoms of long COVID, including symptom subgroups that occurred in 80% of the nearly 10,000 study participants. But the symptoms that patients with long COVID are experiencing now are slightly different from earlier in the pandemic or at least that’s what doctors are finding at the Post-COVID Recovery Clinic affiliated with the University of Pittsburgh Medical Center.
Michael Risbano, MD, the clinic’s codirector, said fewer patients have pulmonary or lung damage now than in the past, but a steady stream report problems with brain fog, forgetfulness, exercise intolerance (shortness of breath and fatigue with exercise and difficulty performing any kind of exertional activity), and post-exertional malaise (feeling wiped out or fatigued for hours or even days after physical or mental activity).
Long COVID Treatments Showing Improvement — Slowly
“There still isn’t a great way to treat any of this,” said Risbano, whose clinic is involved with the National Institute of Health’s RECOVER-VITAL trial, which is evaluating potential treatments including Paxlovid and exercise to treat autonomic dysfunction with similarities to myalgic encephalomyelitis/chronic fatigue syndrome and POTS, exercise intolerance, and neurocognitive effects such as brain fog.
Risbano and colleagues have found that physical therapy and exercise training have helped patients with exercise intolerance and neurocognitive problems. “It’s not a quick thing where they go through one visit and are better the next day,” he stressed. “It takes a little bit of time, a little bit of effort, a little bit of homework — there are no silver bullets, no magic medications.”
A quick fix was definitely not in the cards for Dean Jones, PhD, who could barely move when he developed long COVID in May 2023. A 74-year-old biochemist and professor of medicine at Emory University in Atlanta, Georgia, he’d recovered fully the first time he had COVID, in August 2022, but had a completely different experience the second time. He had been vaccinated four times when he began experiencing chronic fatigue, intense exertion-induced migraines, severe airway congestion, brain fog, and shortness of breath. The symptoms began after Memorial Day and worsened over the next month.
His resting heart rate began racing even when he was sleeping, jumping from 53 to 70 beats per minute. “It was almost as though the virus had hit my heart rather than the lungs alone,” he said.
Doctors prescribed multiple inhalers and glucocorticoids to calm Jones’s immune system. The worst symptoms began to abate after a few weeks. The bad ones continued for fully 2 months, severely limiting Jones’s activity. Although he no longer slept all day, just walking from one room to another was exhausting. A dedicated scientist who typically worked 10-15 hours a day before getting sick, he was lucky to focus on work-related tasks for a fraction of that time.
Although the migraines went away early on, the headaches remained until well into the fall. Jones’s energy level gradually returned, and by Christmas, he was beginning to feel as healthy as he had before getting COVID in May.
Still, he’s not complaining that it took so long to get better. “At 74, there’s a lot of colleagues who have already passed away,” he said. “I respect the realities of my age. There are so many people who died from COVID that I’m thankful I had those vaccines. I’m thankful that I pulled through it and was able to rebound.”
Time Helps Healing — But Prompt Care Still Needed
Recovery is the case for most patients with long COVID, said Lisa Sanders, MD, medical director of the Yale New Haven Health Systems Long COVID Consultation Clinic, which opened in March 2023. Although the clinic has a small segment of patients who have had the condition since 2020, “people who recover, who are most people, move on,” she said. “Even the patients who sometimes have to wait a month or so to see me, some of them say, ‘I’m already starting to get better. I wasn’t sure I should come.’”
Maio, too, is recovering but only after multiple visits to the emergency room and a neurologist in late December and early January. The third emergency room trip was prompted after a brief episode in which she lost the feeling in her legs, which began convulsing. A CAT scan showed severely constricted blood vessels in her brain, leading the medical team to speculate she might have reversible cerebral vasoconstriction syndrome (RCVS), which can trigger the thunderclap headaches that had been causing her such misery.
After her third such headache prompted a fourth emergency room visit, further tests confirmed RCVS, which doctors said was related to inflammation caused by COVID. Maio was then admitted to the hospital, where she spent 4 days starting on a regimen of blood pressure medication, magnesium for the headaches, and oxycodone for pain management.
The TV show Maio works on went back into production after the holidays. She went back at the end of January. She’s still having headaches, though they’re less intense, and she’s still taking medication. She was scheduled for another test to look at her blood vessels in February.
Maio has yet to forgive herself for skipping the last booster, even though there’s no guarantee it would have prevented her from getting sick. Her message for others: it’s better to be safe than to be as sorry as she is.
“I’ll never, ever be persuaded by people who don’t believe in vaccines because I believe in science, and I believe in vaccines — that’s why people don’t die at the age of 30 anymore,” she said. “I really think that people need to know about this and what to expect. Because it is horrendous. It is very painful. I would never want anyone to go through this. Ever.”
A version of this article appeared on Medscape.com.
Diabetes Tech Falls Short as Hypoglycemic Challenges Persist
TOPLINE:
Despite diabetes technology, many with type 1 diabetes (T1D) miss glycemic targets and experience severe hypoglycemia and impaired awareness of hypoglycemia (IAH).
METHODOLOGY:
- The clinical management of T1D through technology is now recognized as the standard of care, but its real-world impact on glycemic targets and severe hypoglycemic events and IAH is unclear.
- Researchers assessed the self-reported prevalence of glycemic metrics, severe hypoglycemia, and hypoglycemia awareness according to the use of continuous glucose monitoring (CGM) and automated insulin delivery (AID) systems.
- They enrolled 2044 individuals diagnosed with T1D for at least 2 years (mean age, 43.0 years; 72.1% women; 95.4% White) from the T1D Exchange Registry and online communities who filled an online survey.
- Participants were stratified on the basis of the presence or absence of CGM and different insulin delivery methods (multiple daily injections, conventional pumps, or AID systems).
- The primary outcome was the proportion of participants who achieved glycemic targets (self-reported A1c), had severe hypoglycemia (any low glucose incidence in 12 months), and/or IAH (a modified Gold score on a seven-point Likert scale).
TAKEAWAY:
- Most participants (91.7%) used CGM, and 50.8% of CGM users used an AID system.
- Despite advanced interventions, only 59.6% (95% CI, 57.3%-61.8%) of CGM users met the glycemic target (A1c < 7%), while nearly 40% of CGM users and 35.6% of AID users didn’t reach the target.
- At least one event of severe hypoglycemia in the previous 12 months was reported in 10.8% of CGM users and 16.6% of those using an AID system.
- IAH prevalence was seen in 31.1% (95% CI, 29.0%-33.2%) and 30.3% (95% CI, 17.5%-33.3%) of participants using CGM and CGM + AID, respectively.
IN PRACTICE:
“Educational initiatives continue to be important for all individuals with type 1 diabetes, and the development of novel therapeutic options and strategies, including bihormonal AID systems and beta-cell replacement, will be required to enable more of these individuals to meet treatment goals,” the authors wrote.
SOURCE:
The study, published online in Diabetes Care, was led by Jennifer L. Sherr, MD, PhD, Yale School of Medicine, New Haven, Connecticut.
LIMITATIONS:
The survey participants in this study were from the T1D Exchange online community, who tend to be highly involved, have technology experience, and are more likely to achieve glycemic targets. The data reported as part of the survey were based on self-reports by participants and may be subject to recall bias. Notably, severe hypoglycemic events may be overreported by individuals using CGM and AID systems due to sensor alarms.
DISCLOSURES:
The study was funded by Vertex Pharmaceuticals. Several authors disclosed financial relationships, including grants, consulting fees, honoraria, stock ownership, and employment with pharmaceutical and device companies and other entities.
A version of this article appeared on Medscape.com.
TOPLINE:
Despite diabetes technology, many with type 1 diabetes (T1D) miss glycemic targets and experience severe hypoglycemia and impaired awareness of hypoglycemia (IAH).
METHODOLOGY:
- The clinical management of T1D through technology is now recognized as the standard of care, but its real-world impact on glycemic targets and severe hypoglycemic events and IAH is unclear.
- Researchers assessed the self-reported prevalence of glycemic metrics, severe hypoglycemia, and hypoglycemia awareness according to the use of continuous glucose monitoring (CGM) and automated insulin delivery (AID) systems.
- They enrolled 2044 individuals diagnosed with T1D for at least 2 years (mean age, 43.0 years; 72.1% women; 95.4% White) from the T1D Exchange Registry and online communities who filled an online survey.
- Participants were stratified on the basis of the presence or absence of CGM and different insulin delivery methods (multiple daily injections, conventional pumps, or AID systems).
- The primary outcome was the proportion of participants who achieved glycemic targets (self-reported A1c), had severe hypoglycemia (any low glucose incidence in 12 months), and/or IAH (a modified Gold score on a seven-point Likert scale).
TAKEAWAY:
- Most participants (91.7%) used CGM, and 50.8% of CGM users used an AID system.
- Despite advanced interventions, only 59.6% (95% CI, 57.3%-61.8%) of CGM users met the glycemic target (A1c < 7%), while nearly 40% of CGM users and 35.6% of AID users didn’t reach the target.
- At least one event of severe hypoglycemia in the previous 12 months was reported in 10.8% of CGM users and 16.6% of those using an AID system.
- IAH prevalence was seen in 31.1% (95% CI, 29.0%-33.2%) and 30.3% (95% CI, 17.5%-33.3%) of participants using CGM and CGM + AID, respectively.
IN PRACTICE:
“Educational initiatives continue to be important for all individuals with type 1 diabetes, and the development of novel therapeutic options and strategies, including bihormonal AID systems and beta-cell replacement, will be required to enable more of these individuals to meet treatment goals,” the authors wrote.
SOURCE:
The study, published online in Diabetes Care, was led by Jennifer L. Sherr, MD, PhD, Yale School of Medicine, New Haven, Connecticut.
LIMITATIONS:
The survey participants in this study were from the T1D Exchange online community, who tend to be highly involved, have technology experience, and are more likely to achieve glycemic targets. The data reported as part of the survey were based on self-reports by participants and may be subject to recall bias. Notably, severe hypoglycemic events may be overreported by individuals using CGM and AID systems due to sensor alarms.
DISCLOSURES:
The study was funded by Vertex Pharmaceuticals. Several authors disclosed financial relationships, including grants, consulting fees, honoraria, stock ownership, and employment with pharmaceutical and device companies and other entities.
A version of this article appeared on Medscape.com.
TOPLINE:
Despite diabetes technology, many with type 1 diabetes (T1D) miss glycemic targets and experience severe hypoglycemia and impaired awareness of hypoglycemia (IAH).
METHODOLOGY:
- The clinical management of T1D through technology is now recognized as the standard of care, but its real-world impact on glycemic targets and severe hypoglycemic events and IAH is unclear.
- Researchers assessed the self-reported prevalence of glycemic metrics, severe hypoglycemia, and hypoglycemia awareness according to the use of continuous glucose monitoring (CGM) and automated insulin delivery (AID) systems.
- They enrolled 2044 individuals diagnosed with T1D for at least 2 years (mean age, 43.0 years; 72.1% women; 95.4% White) from the T1D Exchange Registry and online communities who filled an online survey.
- Participants were stratified on the basis of the presence or absence of CGM and different insulin delivery methods (multiple daily injections, conventional pumps, or AID systems).
- The primary outcome was the proportion of participants who achieved glycemic targets (self-reported A1c), had severe hypoglycemia (any low glucose incidence in 12 months), and/or IAH (a modified Gold score on a seven-point Likert scale).
TAKEAWAY:
- Most participants (91.7%) used CGM, and 50.8% of CGM users used an AID system.
- Despite advanced interventions, only 59.6% (95% CI, 57.3%-61.8%) of CGM users met the glycemic target (A1c < 7%), while nearly 40% of CGM users and 35.6% of AID users didn’t reach the target.
- At least one event of severe hypoglycemia in the previous 12 months was reported in 10.8% of CGM users and 16.6% of those using an AID system.
- IAH prevalence was seen in 31.1% (95% CI, 29.0%-33.2%) and 30.3% (95% CI, 17.5%-33.3%) of participants using CGM and CGM + AID, respectively.
IN PRACTICE:
“Educational initiatives continue to be important for all individuals with type 1 diabetes, and the development of novel therapeutic options and strategies, including bihormonal AID systems and beta-cell replacement, will be required to enable more of these individuals to meet treatment goals,” the authors wrote.
SOURCE:
The study, published online in Diabetes Care, was led by Jennifer L. Sherr, MD, PhD, Yale School of Medicine, New Haven, Connecticut.
LIMITATIONS:
The survey participants in this study were from the T1D Exchange online community, who tend to be highly involved, have technology experience, and are more likely to achieve glycemic targets. The data reported as part of the survey were based on self-reports by participants and may be subject to recall bias. Notably, severe hypoglycemic events may be overreported by individuals using CGM and AID systems due to sensor alarms.
DISCLOSURES:
The study was funded by Vertex Pharmaceuticals. Several authors disclosed financial relationships, including grants, consulting fees, honoraria, stock ownership, and employment with pharmaceutical and device companies and other entities.
A version of this article appeared on Medscape.com.
Bivalent Vaccines Protect Even Children Who’ve Had COVID
This transcript has been edited for clarity.
It was only 3 years ago when we called the pathogen we now refer to as the coronavirus “nCOV-19.” It was, in many ways, more descriptive than what we have today. The little “n” there stood for “novel” — and it was really that little “n” that caused us all the trouble.
You see, coronaviruses themselves were not really new to us. Understudied, perhaps, but with four strains running around the globe at any time giving rise to the common cold, these were viruses our bodies understood.
But Instead of acting like a cold, it acted like nothing we had seen before, at least in our lifetime. The story of the pandemic is very much a bildungsroman of our immune systems — a story of how our immunity grew up.
The difference between the start of 2020 and now, when infections with the coronavirus remain common but not as deadly, can be measured in terms of immune education. Some of our immune systems were educated by infection, some by vaccination, and many by both.
When the first vaccines emerged in December 2020, the opportunity to educate our immune systems was still huge. Though, at the time, an estimated 20 million had been infected in the US and 350,000 had died, there was a large population that remained immunologically naive. I was one of them.
If 2020 into early 2021 was the era of immune education, the postvaccine period was the era of the variant. From one COVID strain to two, to five, to innumerable, our immune memory — trained on a specific version of the virus or its spike protein — became imperfect again. Not naive; these variants were not “novel” in the way COVID-19 was novel, but they were different. And different enough to cause infection.
Following the playbook of another virus that loves to come dressed up in different outfits, the flu virus, we find ourselves in the booster era — a world where yearly doses of a vaccine, ideally matched to the variants circulating when the vaccine is given, are the recommendation if not the norm.
But questions remain about the vaccination program, particularly around who should get it. And two populations with big question marks over their heads are (1) people who have already been infected and (2) kids, because their risk for bad outcomes is so much lower.
This week, we finally have some evidence that can shed light on these questions. The study under the spotlight is this one, appearing in JAMA, which tries to analyze the ability of the bivalent vaccine — that’s the second one to come out, around September 2022 — to protect kids from COVID-19.
Now, right off the bat, this was not a randomized trial. The studies that established the viability of the mRNA vaccine platform were; they happened before the vaccine was authorized. But trials of the bivalent vaccine were mostly limited to proving immune response, not protection from disease.
Nevertheless, with some good observational methods and some statistics, we can try to tease out whether bivalent vaccines in kids worked.
The study combines three prospective cohort studies. The details are in the paper, but what you need to know is that the special sauce of these studies was that the kids were tested for COVID-19 on a weekly basis, whether they had symptoms or not. This is critical because asymptomatic infections can transmit COVID-19.
Let’s do the variables of interest. First and foremost, the bivalent vaccine. Some of these kids got the bivalent vaccine, some didn’t. Other key variables include prior vaccination with the monovalent vaccine. Some had been vaccinated with the monovalent vaccine before, some hadn’t. And, of course, prior infection. Some had been infected before (based on either nasal swabs or blood tests).
Let’s focus first on the primary exposure of interest: getting that bivalent vaccine. Again, this was not randomly assigned; kids who got the bivalent vaccine were different from those who did not. In general, they lived in smaller households, they were more likely to be White, less likely to have had a prior COVID infection, and quite a bit more likely to have at least one chronic condition.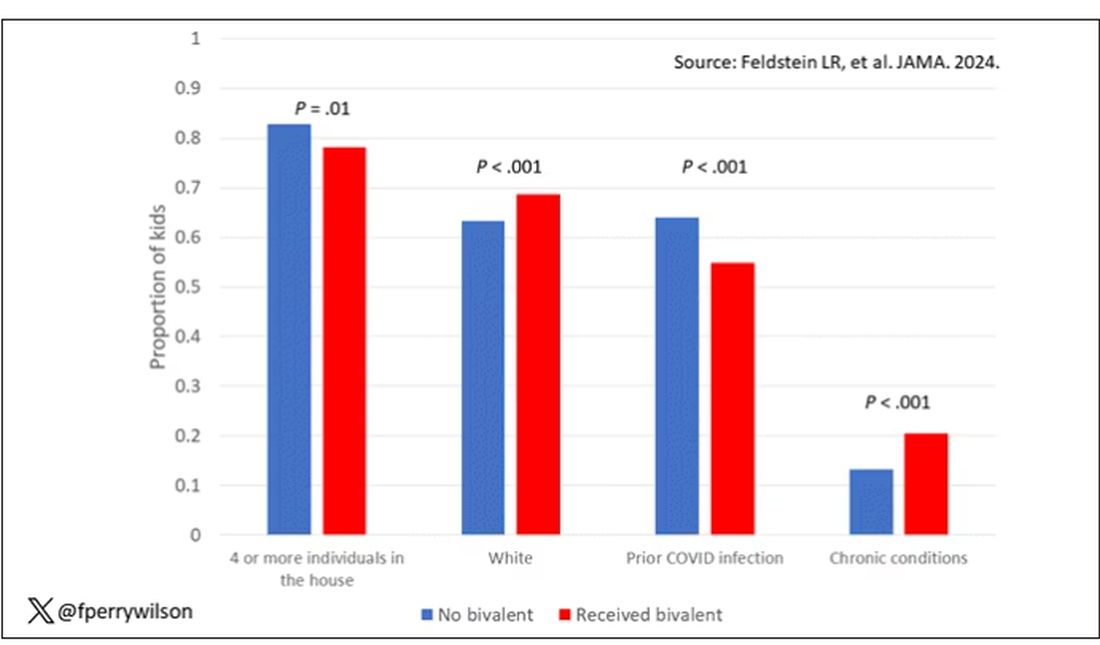
To me, this constellation of factors describes a slightly higher-risk group; it makes sense that they were more likely to get the second vaccine.
Given those factors, what were the rates of COVID infection? After nearly a year of follow-up, around 15% of the kids who hadn’t received the bivalent vaccine got infected compared with 5% of the vaccinated kids. Symptomatic infections represented roughly half of all infections in both groups.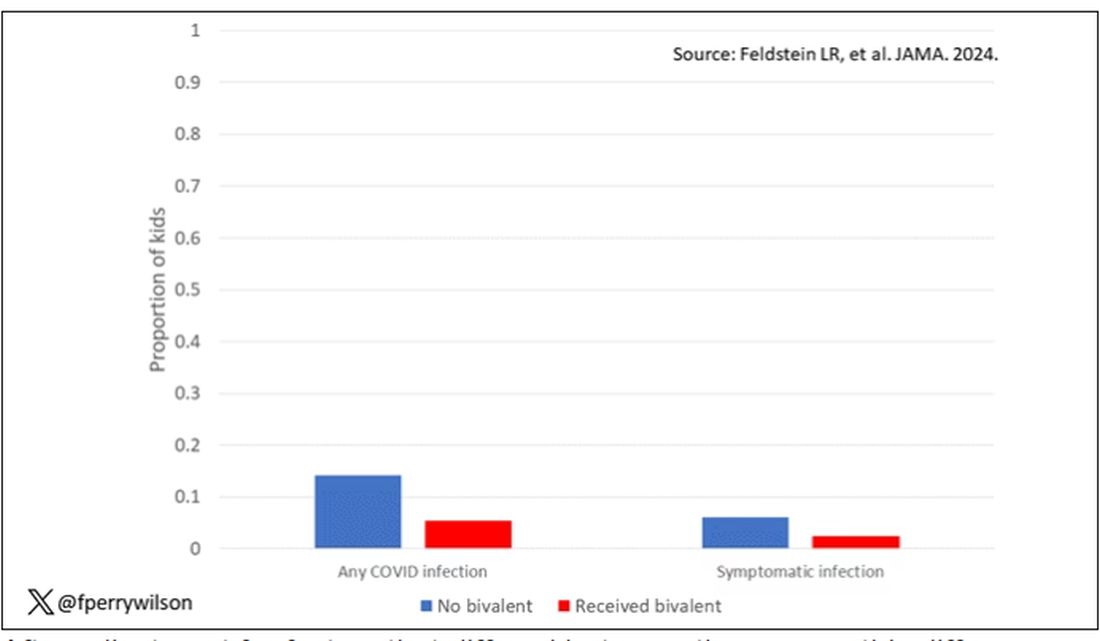
After adjustment for factors that differed between the groups, this difference translated into a vaccine efficacy of about 50% in this population. That’s our first data point. Yes, the bivalent vaccine worked. Not amazingly, of course. But it worked.
What about the kids who had had a prior COVID infection? Somewhat surprisingly, the vaccine was just as effective in this population, despite the fact that their immune systems already had some knowledge of COVID. Ten percent of unvaccinated kids got infected, even though they had been infected before. Just 2.5% of kids who received the bivalent vaccine got infected, suggesting some synergy between prior infection and vaccination.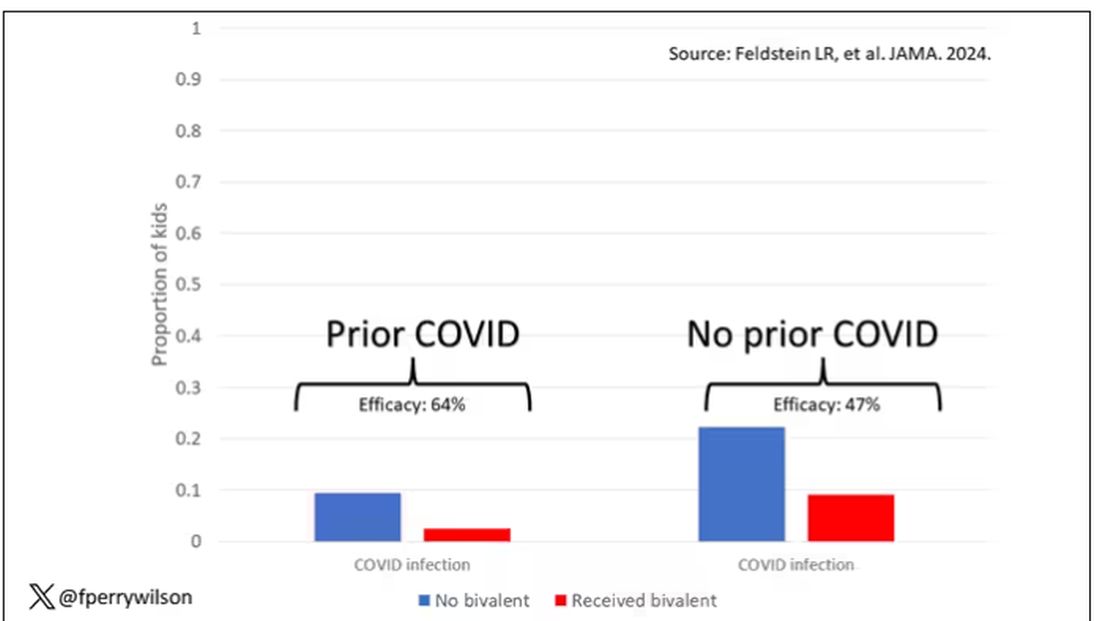
These data suggest that the bivalent vaccine did reduce the risk for COVID infection in kids. All good. But the piece still missing is how severe these infections were. It doesn’t appear that any of the 426 infections documented in this study resulted in hospitalization or death, fortunately. And no data are presented on the incidence of multisystem inflammatory syndrome of children, though given the rarity, I’d be surprised if any of these kids have this either.
So where are we? Well, it seems that the narrative out there that says “the vaccines don’t work” or “the vaccines don’t work if you’ve already been infected” is probably not true. They do work. This study and others in adults show that. If they work to reduce infections, as this study shows, they will also work to reduce deaths. It’s just that death is fortunately so rare in children that the number needed to vaccinate to prevent one death is very large. In that situation, the decision to vaccinate comes down to the risks associated with vaccination. So far, those risk seem very minimal.
Perhaps falling into a flu-like yearly vaccination schedule is not simply the result of old habits dying hard. Maybe it’s actually not a bad idea.
Dr. F. Perry Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
It was only 3 years ago when we called the pathogen we now refer to as the coronavirus “nCOV-19.” It was, in many ways, more descriptive than what we have today. The little “n” there stood for “novel” — and it was really that little “n” that caused us all the trouble.
You see, coronaviruses themselves were not really new to us. Understudied, perhaps, but with four strains running around the globe at any time giving rise to the common cold, these were viruses our bodies understood.
But Instead of acting like a cold, it acted like nothing we had seen before, at least in our lifetime. The story of the pandemic is very much a bildungsroman of our immune systems — a story of how our immunity grew up.
The difference between the start of 2020 and now, when infections with the coronavirus remain common but not as deadly, can be measured in terms of immune education. Some of our immune systems were educated by infection, some by vaccination, and many by both.
When the first vaccines emerged in December 2020, the opportunity to educate our immune systems was still huge. Though, at the time, an estimated 20 million had been infected in the US and 350,000 had died, there was a large population that remained immunologically naive. I was one of them.
If 2020 into early 2021 was the era of immune education, the postvaccine period was the era of the variant. From one COVID strain to two, to five, to innumerable, our immune memory — trained on a specific version of the virus or its spike protein — became imperfect again. Not naive; these variants were not “novel” in the way COVID-19 was novel, but they were different. And different enough to cause infection.
Following the playbook of another virus that loves to come dressed up in different outfits, the flu virus, we find ourselves in the booster era — a world where yearly doses of a vaccine, ideally matched to the variants circulating when the vaccine is given, are the recommendation if not the norm.
But questions remain about the vaccination program, particularly around who should get it. And two populations with big question marks over their heads are (1) people who have already been infected and (2) kids, because their risk for bad outcomes is so much lower.
This week, we finally have some evidence that can shed light on these questions. The study under the spotlight is this one, appearing in JAMA, which tries to analyze the ability of the bivalent vaccine — that’s the second one to come out, around September 2022 — to protect kids from COVID-19.
Now, right off the bat, this was not a randomized trial. The studies that established the viability of the mRNA vaccine platform were; they happened before the vaccine was authorized. But trials of the bivalent vaccine were mostly limited to proving immune response, not protection from disease.
Nevertheless, with some good observational methods and some statistics, we can try to tease out whether bivalent vaccines in kids worked.
The study combines three prospective cohort studies. The details are in the paper, but what you need to know is that the special sauce of these studies was that the kids were tested for COVID-19 on a weekly basis, whether they had symptoms or not. This is critical because asymptomatic infections can transmit COVID-19.
Let’s do the variables of interest. First and foremost, the bivalent vaccine. Some of these kids got the bivalent vaccine, some didn’t. Other key variables include prior vaccination with the monovalent vaccine. Some had been vaccinated with the monovalent vaccine before, some hadn’t. And, of course, prior infection. Some had been infected before (based on either nasal swabs or blood tests).
Let’s focus first on the primary exposure of interest: getting that bivalent vaccine. Again, this was not randomly assigned; kids who got the bivalent vaccine were different from those who did not. In general, they lived in smaller households, they were more likely to be White, less likely to have had a prior COVID infection, and quite a bit more likely to have at least one chronic condition.
To me, this constellation of factors describes a slightly higher-risk group; it makes sense that they were more likely to get the second vaccine.
Given those factors, what were the rates of COVID infection? After nearly a year of follow-up, around 15% of the kids who hadn’t received the bivalent vaccine got infected compared with 5% of the vaccinated kids. Symptomatic infections represented roughly half of all infections in both groups.
After adjustment for factors that differed between the groups, this difference translated into a vaccine efficacy of about 50% in this population. That’s our first data point. Yes, the bivalent vaccine worked. Not amazingly, of course. But it worked.
What about the kids who had had a prior COVID infection? Somewhat surprisingly, the vaccine was just as effective in this population, despite the fact that their immune systems already had some knowledge of COVID. Ten percent of unvaccinated kids got infected, even though they had been infected before. Just 2.5% of kids who received the bivalent vaccine got infected, suggesting some synergy between prior infection and vaccination.
These data suggest that the bivalent vaccine did reduce the risk for COVID infection in kids. All good. But the piece still missing is how severe these infections were. It doesn’t appear that any of the 426 infections documented in this study resulted in hospitalization or death, fortunately. And no data are presented on the incidence of multisystem inflammatory syndrome of children, though given the rarity, I’d be surprised if any of these kids have this either.
So where are we? Well, it seems that the narrative out there that says “the vaccines don’t work” or “the vaccines don’t work if you’ve already been infected” is probably not true. They do work. This study and others in adults show that. If they work to reduce infections, as this study shows, they will also work to reduce deaths. It’s just that death is fortunately so rare in children that the number needed to vaccinate to prevent one death is very large. In that situation, the decision to vaccinate comes down to the risks associated with vaccination. So far, those risk seem very minimal.
Perhaps falling into a flu-like yearly vaccination schedule is not simply the result of old habits dying hard. Maybe it’s actually not a bad idea.
Dr. F. Perry Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
It was only 3 years ago when we called the pathogen we now refer to as the coronavirus “nCOV-19.” It was, in many ways, more descriptive than what we have today. The little “n” there stood for “novel” — and it was really that little “n” that caused us all the trouble.
You see, coronaviruses themselves were not really new to us. Understudied, perhaps, but with four strains running around the globe at any time giving rise to the common cold, these were viruses our bodies understood.
But Instead of acting like a cold, it acted like nothing we had seen before, at least in our lifetime. The story of the pandemic is very much a bildungsroman of our immune systems — a story of how our immunity grew up.
The difference between the start of 2020 and now, when infections with the coronavirus remain common but not as deadly, can be measured in terms of immune education. Some of our immune systems were educated by infection, some by vaccination, and many by both.
When the first vaccines emerged in December 2020, the opportunity to educate our immune systems was still huge. Though, at the time, an estimated 20 million had been infected in the US and 350,000 had died, there was a large population that remained immunologically naive. I was one of them.
If 2020 into early 2021 was the era of immune education, the postvaccine period was the era of the variant. From one COVID strain to two, to five, to innumerable, our immune memory — trained on a specific version of the virus or its spike protein — became imperfect again. Not naive; these variants were not “novel” in the way COVID-19 was novel, but they were different. And different enough to cause infection.
Following the playbook of another virus that loves to come dressed up in different outfits, the flu virus, we find ourselves in the booster era — a world where yearly doses of a vaccine, ideally matched to the variants circulating when the vaccine is given, are the recommendation if not the norm.
But questions remain about the vaccination program, particularly around who should get it. And two populations with big question marks over their heads are (1) people who have already been infected and (2) kids, because their risk for bad outcomes is so much lower.
This week, we finally have some evidence that can shed light on these questions. The study under the spotlight is this one, appearing in JAMA, which tries to analyze the ability of the bivalent vaccine — that’s the second one to come out, around September 2022 — to protect kids from COVID-19.
Now, right off the bat, this was not a randomized trial. The studies that established the viability of the mRNA vaccine platform were; they happened before the vaccine was authorized. But trials of the bivalent vaccine were mostly limited to proving immune response, not protection from disease.
Nevertheless, with some good observational methods and some statistics, we can try to tease out whether bivalent vaccines in kids worked.
The study combines three prospective cohort studies. The details are in the paper, but what you need to know is that the special sauce of these studies was that the kids were tested for COVID-19 on a weekly basis, whether they had symptoms or not. This is critical because asymptomatic infections can transmit COVID-19.
Let’s do the variables of interest. First and foremost, the bivalent vaccine. Some of these kids got the bivalent vaccine, some didn’t. Other key variables include prior vaccination with the monovalent vaccine. Some had been vaccinated with the monovalent vaccine before, some hadn’t. And, of course, prior infection. Some had been infected before (based on either nasal swabs or blood tests).
Let’s focus first on the primary exposure of interest: getting that bivalent vaccine. Again, this was not randomly assigned; kids who got the bivalent vaccine were different from those who did not. In general, they lived in smaller households, they were more likely to be White, less likely to have had a prior COVID infection, and quite a bit more likely to have at least one chronic condition.
To me, this constellation of factors describes a slightly higher-risk group; it makes sense that they were more likely to get the second vaccine.
Given those factors, what were the rates of COVID infection? After nearly a year of follow-up, around 15% of the kids who hadn’t received the bivalent vaccine got infected compared with 5% of the vaccinated kids. Symptomatic infections represented roughly half of all infections in both groups.
After adjustment for factors that differed between the groups, this difference translated into a vaccine efficacy of about 50% in this population. That’s our first data point. Yes, the bivalent vaccine worked. Not amazingly, of course. But it worked.
What about the kids who had had a prior COVID infection? Somewhat surprisingly, the vaccine was just as effective in this population, despite the fact that their immune systems already had some knowledge of COVID. Ten percent of unvaccinated kids got infected, even though they had been infected before. Just 2.5% of kids who received the bivalent vaccine got infected, suggesting some synergy between prior infection and vaccination.
These data suggest that the bivalent vaccine did reduce the risk for COVID infection in kids. All good. But the piece still missing is how severe these infections were. It doesn’t appear that any of the 426 infections documented in this study resulted in hospitalization or death, fortunately. And no data are presented on the incidence of multisystem inflammatory syndrome of children, though given the rarity, I’d be surprised if any of these kids have this either.
So where are we? Well, it seems that the narrative out there that says “the vaccines don’t work” or “the vaccines don’t work if you’ve already been infected” is probably not true. They do work. This study and others in adults show that. If they work to reduce infections, as this study shows, they will also work to reduce deaths. It’s just that death is fortunately so rare in children that the number needed to vaccinate to prevent one death is very large. In that situation, the decision to vaccinate comes down to the risks associated with vaccination. So far, those risk seem very minimal.
Perhaps falling into a flu-like yearly vaccination schedule is not simply the result of old habits dying hard. Maybe it’s actually not a bad idea.
Dr. F. Perry Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
OTC Topical Scar Products May Contain Allergens, Study Finds
TOPLINE:
METHODOLOGY:
- OTC topical scar treatments have the potential to cause an allergic reaction, but the prevalence of North American Contact Dermatitis Group (NACDG) core allergens in these products is unclear.
- Researchers used the word scar in a query of Amazon.com and four other retail websites to identify topical scar products for consumers and noted the list of ingredients.
- The investigators also surveyed the American Contact Dermatitis Society’s Contact Allergen Management Program (CAMP), a resource that helps patients with allergies find personal care products that are safe to use, for pertinent products.
TAKEAWAY:
- The search query identified 156 products. Of these, 119 (76.2%) were gels, creams, or oils and 37 (23.7%) were sheets, strips, or tape.
- Of the 125 products that had a list of ingredients, 69 (55.2%) contained at least one NACDG allergen and 45 (36%) contained more than one.
- The top six most common allergens listed in the ingredients were fragrance (16.8%), phenoxyethanol (16.8%), parabens (14.4%), panthenol (12.8%), sodium benzoate (9.60%), and ethylhexylglycerin (8%).
- Analysis of CAMP revealed that the program only had five unique scar products in its list, suggesting that CAMP might not be a reliable source of scar product information for patients with known allergies to pertinent NACDG allergens.
IN PRACTICE:
“Patients can consider trying a ‘use test’ on the inner forearm before applying to the surgical site,” the authors wrote. “It may reveal they are sensitive or sensitized by a product.
SOURCE:
First author Meera Kattapuram, MD, of the Department of Internal Medicine at Mount Sinai Hospital, New York, led the study, published in the February issue of Dermatologic Surgery.
LIMITATIONS:
Limitations include the selection of five retailers and the top 100 products from each website and the potential for ingredient list inaccuracies.
DISCLOSURES:
The authors reported having no financial conflicts of interest. The research was supported by a grant from the National Institutes of Health/National Cancer Institute.
A version of this article appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- OTC topical scar treatments have the potential to cause an allergic reaction, but the prevalence of North American Contact Dermatitis Group (NACDG) core allergens in these products is unclear.
- Researchers used the word scar in a query of Amazon.com and four other retail websites to identify topical scar products for consumers and noted the list of ingredients.
- The investigators also surveyed the American Contact Dermatitis Society’s Contact Allergen Management Program (CAMP), a resource that helps patients with allergies find personal care products that are safe to use, for pertinent products.
TAKEAWAY:
- The search query identified 156 products. Of these, 119 (76.2%) were gels, creams, or oils and 37 (23.7%) were sheets, strips, or tape.
- Of the 125 products that had a list of ingredients, 69 (55.2%) contained at least one NACDG allergen and 45 (36%) contained more than one.
- The top six most common allergens listed in the ingredients were fragrance (16.8%), phenoxyethanol (16.8%), parabens (14.4%), panthenol (12.8%), sodium benzoate (9.60%), and ethylhexylglycerin (8%).
- Analysis of CAMP revealed that the program only had five unique scar products in its list, suggesting that CAMP might not be a reliable source of scar product information for patients with known allergies to pertinent NACDG allergens.
IN PRACTICE:
“Patients can consider trying a ‘use test’ on the inner forearm before applying to the surgical site,” the authors wrote. “It may reveal they are sensitive or sensitized by a product.
SOURCE:
First author Meera Kattapuram, MD, of the Department of Internal Medicine at Mount Sinai Hospital, New York, led the study, published in the February issue of Dermatologic Surgery.
LIMITATIONS:
Limitations include the selection of five retailers and the top 100 products from each website and the potential for ingredient list inaccuracies.
DISCLOSURES:
The authors reported having no financial conflicts of interest. The research was supported by a grant from the National Institutes of Health/National Cancer Institute.
A version of this article appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- OTC topical scar treatments have the potential to cause an allergic reaction, but the prevalence of North American Contact Dermatitis Group (NACDG) core allergens in these products is unclear.
- Researchers used the word scar in a query of Amazon.com and four other retail websites to identify topical scar products for consumers and noted the list of ingredients.
- The investigators also surveyed the American Contact Dermatitis Society’s Contact Allergen Management Program (CAMP), a resource that helps patients with allergies find personal care products that are safe to use, for pertinent products.
TAKEAWAY:
- The search query identified 156 products. Of these, 119 (76.2%) were gels, creams, or oils and 37 (23.7%) were sheets, strips, or tape.
- Of the 125 products that had a list of ingredients, 69 (55.2%) contained at least one NACDG allergen and 45 (36%) contained more than one.
- The top six most common allergens listed in the ingredients were fragrance (16.8%), phenoxyethanol (16.8%), parabens (14.4%), panthenol (12.8%), sodium benzoate (9.60%), and ethylhexylglycerin (8%).
- Analysis of CAMP revealed that the program only had five unique scar products in its list, suggesting that CAMP might not be a reliable source of scar product information for patients with known allergies to pertinent NACDG allergens.
IN PRACTICE:
“Patients can consider trying a ‘use test’ on the inner forearm before applying to the surgical site,” the authors wrote. “It may reveal they are sensitive or sensitized by a product.
SOURCE:
First author Meera Kattapuram, MD, of the Department of Internal Medicine at Mount Sinai Hospital, New York, led the study, published in the February issue of Dermatologic Surgery.
LIMITATIONS:
Limitations include the selection of five retailers and the top 100 products from each website and the potential for ingredient list inaccuracies.
DISCLOSURES:
The authors reported having no financial conflicts of interest. The research was supported by a grant from the National Institutes of Health/National Cancer Institute.
A version of this article appeared on Medscape.com.
When Babies ‘Stop Breathing,’ Who Needs Admission and a Workup?
Many infants have experienced an episode of apnea, defined as a pause in respiration of 20 seconds or more. Most episodes remain unexplained, and no underlying cause can be found. Historically, these were referred to as “near-miss SIDS,” episodes, but that label suggested that all of these events would have ended in death had someone not intervened. New descriptive terminology was needed.
In the mid-1980s, the term “apparent life-threatening event” (ALTE) was adopted. But that term, too, was an overstatement, because although scary for parents, these brief apnea episodes were not, in most cases, truly life-threatening.
In 2013, authors of a systematic review coined the term “brief resolved unexplained event” (BRUE). This review also addressed the history and physical exam features associated with risk for a subsequent episode. It was felt that hospitalization and testing might be warranted if certain infants could be identified as high risk for recurrence.
What Is Considered a BRUE?
In the current working definition of BRUE, the child must be < 1 year old. The episode must be a sudden, brief, and resolved, with one or more of these characteristics:
- Cyanosis or pallor (but not turning red)
- A change in breathing (absent, decreased, or irregular)
- A change in tone (hypertonia or hypotonia)
- A change in responsiveness.
Furthermore, to qualify as a BRUE, no explanation can be found for the event based on the history and physical examination but before any laboratory testing is done. The definition also excludes children with known potential explanatory diagnoses (such as gastroesophageal reflux or bronchiolitis) and those who are otherwise symptomatically ill at the time of the event.
Decision to Admit and Recurrence Risk
An apnea event in an otherwise healthy infant, regardless of what it’s called, puts providers and parents in a difficult position. Should the infant be hospitalized for further monitoring and potentially more invasive testing to determine the cause of the episode? And what are the chances that the episode will be repeated?
A clinical practice guideline (CPG) for BRUE, widely adopted in 2016, resulted in significant reductions in healthcare utilization. The CPG attempted to identify low-risk infants who could safely be discharged from the emergency department. Although the CPG improved outcomes, experts acknowledged that an underlying problem was not likely to be identified even among infants deemed high risk, and these infants would be hospitalized unnecessarily.
Available data were simply insufficient to support this decision. So, with the goal of identifying factors that could help predict recurrent BRUE risk, a 15-hospital collaborative study was undertaken, followed by the development and validation of a clinical decision rule for predicting the risk for a serious underlying diagnosis or event recurrence among infants presenting with BRUE.
Here’s what we learned from more than 3000 cases of BRUE.
First, it turns out that it’s not easy to determine whether an infant is at low or high risk for recurrence of BRUE. Initially, 91.5% of patients enrolled in the study would have been labeled high risk.
Furthermore, a BRUE recurred in 14.3% of the cohort, and 4.8% of high-risk infants were found to have a serious undiagnosed condition. Seizures, airway anomalies, and gastroesophageal reflux were the top three causes of BRUE, but the spectrum of underlying pathology was quite considerable.
The problem was that 4.6% of the entire cohort were found to have a serious underlying condition, nearly identical to the proportion of high-risk infants with these conditions. This prompted the question of whether simply labeling infants “high risk” was really appropriate any longer.
Revised BRUE Management
Although it hasn’t been possible to group infants neatly in low and high-risk categories, the data from that large cohort led to the development of the BRUE 2.0 criteria, which enabled more focused risk assessment of an infant who experienced a BRUE. With an app on MDCalc, these criteria allow providers to ascertain, and show families, a visual representation of their infant’s individualized risk for a subsequent BRUE and of having a serious underlying condition.
The cohort study also identified red flags from the history or physical exam of infants who experienced a BRUE: weight loss, failure to thrive, or a history of feeding problems. Exam findings such as a bulging fontanelle, forceful or bilious emesis, and evidence of gastrointestinal (GI) bleeding suggest a medical diagnosis rather than a BRUE. If GI-related causes are high on the differential, a feeding evaluation can be helpful. A feeding evaluation can be done in the outpatient setting and does not require hospitalization.
For suspicion of an underlying neurological condition (such as seizures), experts recommend obtaining a short EEG, which is highly sensitive for detecting infantile spasms and encephalopathy. They recommend reserving MRI for infants with abnormalities on EEG or physical exam. Metabolic or genetic testing should be done only if the infant looks ill, because most patients with genetic or inborn errors of metabolism will continue to have symptoms as they become older.
The approach to BRUE has moved into the realm of shared decision-making with families. The likelihood of identifying a serious diagnosis is low for most of these children. And unfortunately, no single test can diagnose the full spectrum of potential explanatory diagnoses. For example, data from 2023 demonstrate that only 1.1% of lab tests following a BRUE contributed to a diagnosis, and most of the time that was a positive viral test. Similarly, imaging was helpful in only 1.5% of cases. So, explaining the evidence and deciding along with parents what is reasonable to do (or not do) is the current state of affairs.
My Take
As I reflect back on two and a half decades of caring for these patients, I believe that recent data have helped us a great deal. We do less testing and admit fewer infants to the hospital than we did 20 years ago, and that’s a good thing. Nevertheless, looking for a few red flags, having a high index of suspicion when the clinical exam is abnormal, and engaging in shared decision-making with families can help make the caring for these challenging patients more bearable and lead to better outcomes for all involved.
Dr. Basco is Professor, Department of Pediatrics, Medical University of South Carolina (MUSC); Director, Division of General Pediatrics, Department of Pediatrics, MUSC Children’s Hospital, Charleston, South Carolina. He has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Many infants have experienced an episode of apnea, defined as a pause in respiration of 20 seconds or more. Most episodes remain unexplained, and no underlying cause can be found. Historically, these were referred to as “near-miss SIDS,” episodes, but that label suggested that all of these events would have ended in death had someone not intervened. New descriptive terminology was needed.
In the mid-1980s, the term “apparent life-threatening event” (ALTE) was adopted. But that term, too, was an overstatement, because although scary for parents, these brief apnea episodes were not, in most cases, truly life-threatening.
In 2013, authors of a systematic review coined the term “brief resolved unexplained event” (BRUE). This review also addressed the history and physical exam features associated with risk for a subsequent episode. It was felt that hospitalization and testing might be warranted if certain infants could be identified as high risk for recurrence.
What Is Considered a BRUE?
In the current working definition of BRUE, the child must be < 1 year old. The episode must be a sudden, brief, and resolved, with one or more of these characteristics:
- Cyanosis or pallor (but not turning red)
- A change in breathing (absent, decreased, or irregular)
- A change in tone (hypertonia or hypotonia)
- A change in responsiveness.
Furthermore, to qualify as a BRUE, no explanation can be found for the event based on the history and physical examination but before any laboratory testing is done. The definition also excludes children with known potential explanatory diagnoses (such as gastroesophageal reflux or bronchiolitis) and those who are otherwise symptomatically ill at the time of the event.
Decision to Admit and Recurrence Risk
An apnea event in an otherwise healthy infant, regardless of what it’s called, puts providers and parents in a difficult position. Should the infant be hospitalized for further monitoring and potentially more invasive testing to determine the cause of the episode? And what are the chances that the episode will be repeated?
A clinical practice guideline (CPG) for BRUE, widely adopted in 2016, resulted in significant reductions in healthcare utilization. The CPG attempted to identify low-risk infants who could safely be discharged from the emergency department. Although the CPG improved outcomes, experts acknowledged that an underlying problem was not likely to be identified even among infants deemed high risk, and these infants would be hospitalized unnecessarily.
Available data were simply insufficient to support this decision. So, with the goal of identifying factors that could help predict recurrent BRUE risk, a 15-hospital collaborative study was undertaken, followed by the development and validation of a clinical decision rule for predicting the risk for a serious underlying diagnosis or event recurrence among infants presenting with BRUE.
Here’s what we learned from more than 3000 cases of BRUE.
First, it turns out that it’s not easy to determine whether an infant is at low or high risk for recurrence of BRUE. Initially, 91.5% of patients enrolled in the study would have been labeled high risk.
Furthermore, a BRUE recurred in 14.3% of the cohort, and 4.8% of high-risk infants were found to have a serious undiagnosed condition. Seizures, airway anomalies, and gastroesophageal reflux were the top three causes of BRUE, but the spectrum of underlying pathology was quite considerable.
The problem was that 4.6% of the entire cohort were found to have a serious underlying condition, nearly identical to the proportion of high-risk infants with these conditions. This prompted the question of whether simply labeling infants “high risk” was really appropriate any longer.
Revised BRUE Management
Although it hasn’t been possible to group infants neatly in low and high-risk categories, the data from that large cohort led to the development of the BRUE 2.0 criteria, which enabled more focused risk assessment of an infant who experienced a BRUE. With an app on MDCalc, these criteria allow providers to ascertain, and show families, a visual representation of their infant’s individualized risk for a subsequent BRUE and of having a serious underlying condition.
The cohort study also identified red flags from the history or physical exam of infants who experienced a BRUE: weight loss, failure to thrive, or a history of feeding problems. Exam findings such as a bulging fontanelle, forceful or bilious emesis, and evidence of gastrointestinal (GI) bleeding suggest a medical diagnosis rather than a BRUE. If GI-related causes are high on the differential, a feeding evaluation can be helpful. A feeding evaluation can be done in the outpatient setting and does not require hospitalization.
For suspicion of an underlying neurological condition (such as seizures), experts recommend obtaining a short EEG, which is highly sensitive for detecting infantile spasms and encephalopathy. They recommend reserving MRI for infants with abnormalities on EEG or physical exam. Metabolic or genetic testing should be done only if the infant looks ill, because most patients with genetic or inborn errors of metabolism will continue to have symptoms as they become older.
The approach to BRUE has moved into the realm of shared decision-making with families. The likelihood of identifying a serious diagnosis is low for most of these children. And unfortunately, no single test can diagnose the full spectrum of potential explanatory diagnoses. For example, data from 2023 demonstrate that only 1.1% of lab tests following a BRUE contributed to a diagnosis, and most of the time that was a positive viral test. Similarly, imaging was helpful in only 1.5% of cases. So, explaining the evidence and deciding along with parents what is reasonable to do (or not do) is the current state of affairs.
My Take
As I reflect back on two and a half decades of caring for these patients, I believe that recent data have helped us a great deal. We do less testing and admit fewer infants to the hospital than we did 20 years ago, and that’s a good thing. Nevertheless, looking for a few red flags, having a high index of suspicion when the clinical exam is abnormal, and engaging in shared decision-making with families can help make the caring for these challenging patients more bearable and lead to better outcomes for all involved.
Dr. Basco is Professor, Department of Pediatrics, Medical University of South Carolina (MUSC); Director, Division of General Pediatrics, Department of Pediatrics, MUSC Children’s Hospital, Charleston, South Carolina. He has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Many infants have experienced an episode of apnea, defined as a pause in respiration of 20 seconds or more. Most episodes remain unexplained, and no underlying cause can be found. Historically, these were referred to as “near-miss SIDS,” episodes, but that label suggested that all of these events would have ended in death had someone not intervened. New descriptive terminology was needed.
In the mid-1980s, the term “apparent life-threatening event” (ALTE) was adopted. But that term, too, was an overstatement, because although scary for parents, these brief apnea episodes were not, in most cases, truly life-threatening.
In 2013, authors of a systematic review coined the term “brief resolved unexplained event” (BRUE). This review also addressed the history and physical exam features associated with risk for a subsequent episode. It was felt that hospitalization and testing might be warranted if certain infants could be identified as high risk for recurrence.
What Is Considered a BRUE?
In the current working definition of BRUE, the child must be < 1 year old. The episode must be a sudden, brief, and resolved, with one or more of these characteristics:
- Cyanosis or pallor (but not turning red)
- A change in breathing (absent, decreased, or irregular)
- A change in tone (hypertonia or hypotonia)
- A change in responsiveness.
Furthermore, to qualify as a BRUE, no explanation can be found for the event based on the history and physical examination but before any laboratory testing is done. The definition also excludes children with known potential explanatory diagnoses (such as gastroesophageal reflux or bronchiolitis) and those who are otherwise symptomatically ill at the time of the event.
Decision to Admit and Recurrence Risk
An apnea event in an otherwise healthy infant, regardless of what it’s called, puts providers and parents in a difficult position. Should the infant be hospitalized for further monitoring and potentially more invasive testing to determine the cause of the episode? And what are the chances that the episode will be repeated?
A clinical practice guideline (CPG) for BRUE, widely adopted in 2016, resulted in significant reductions in healthcare utilization. The CPG attempted to identify low-risk infants who could safely be discharged from the emergency department. Although the CPG improved outcomes, experts acknowledged that an underlying problem was not likely to be identified even among infants deemed high risk, and these infants would be hospitalized unnecessarily.
Available data were simply insufficient to support this decision. So, with the goal of identifying factors that could help predict recurrent BRUE risk, a 15-hospital collaborative study was undertaken, followed by the development and validation of a clinical decision rule for predicting the risk for a serious underlying diagnosis or event recurrence among infants presenting with BRUE.
Here’s what we learned from more than 3000 cases of BRUE.
First, it turns out that it’s not easy to determine whether an infant is at low or high risk for recurrence of BRUE. Initially, 91.5% of patients enrolled in the study would have been labeled high risk.
Furthermore, a BRUE recurred in 14.3% of the cohort, and 4.8% of high-risk infants were found to have a serious undiagnosed condition. Seizures, airway anomalies, and gastroesophageal reflux were the top three causes of BRUE, but the spectrum of underlying pathology was quite considerable.
The problem was that 4.6% of the entire cohort were found to have a serious underlying condition, nearly identical to the proportion of high-risk infants with these conditions. This prompted the question of whether simply labeling infants “high risk” was really appropriate any longer.
Revised BRUE Management
Although it hasn’t been possible to group infants neatly in low and high-risk categories, the data from that large cohort led to the development of the BRUE 2.0 criteria, which enabled more focused risk assessment of an infant who experienced a BRUE. With an app on MDCalc, these criteria allow providers to ascertain, and show families, a visual representation of their infant’s individualized risk for a subsequent BRUE and of having a serious underlying condition.
The cohort study also identified red flags from the history or physical exam of infants who experienced a BRUE: weight loss, failure to thrive, or a history of feeding problems. Exam findings such as a bulging fontanelle, forceful or bilious emesis, and evidence of gastrointestinal (GI) bleeding suggest a medical diagnosis rather than a BRUE. If GI-related causes are high on the differential, a feeding evaluation can be helpful. A feeding evaluation can be done in the outpatient setting and does not require hospitalization.
For suspicion of an underlying neurological condition (such as seizures), experts recommend obtaining a short EEG, which is highly sensitive for detecting infantile spasms and encephalopathy. They recommend reserving MRI for infants with abnormalities on EEG or physical exam. Metabolic or genetic testing should be done only if the infant looks ill, because most patients with genetic or inborn errors of metabolism will continue to have symptoms as they become older.
The approach to BRUE has moved into the realm of shared decision-making with families. The likelihood of identifying a serious diagnosis is low for most of these children. And unfortunately, no single test can diagnose the full spectrum of potential explanatory diagnoses. For example, data from 2023 demonstrate that only 1.1% of lab tests following a BRUE contributed to a diagnosis, and most of the time that was a positive viral test. Similarly, imaging was helpful in only 1.5% of cases. So, explaining the evidence and deciding along with parents what is reasonable to do (or not do) is the current state of affairs.
My Take
As I reflect back on two and a half decades of caring for these patients, I believe that recent data have helped us a great deal. We do less testing and admit fewer infants to the hospital than we did 20 years ago, and that’s a good thing. Nevertheless, looking for a few red flags, having a high index of suspicion when the clinical exam is abnormal, and engaging in shared decision-making with families can help make the caring for these challenging patients more bearable and lead to better outcomes for all involved.
Dr. Basco is Professor, Department of Pediatrics, Medical University of South Carolina (MUSC); Director, Division of General Pediatrics, Department of Pediatrics, MUSC Children’s Hospital, Charleston, South Carolina. He has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Study: Lifetime Cost of Vyjuvek Gene Therapy for DEB Could Be $15-$22 Million
according to the authors of a new study.
The US Food and Drug Administration (FDA) approved Vyjuvek (Krystal Biotech) in May 2023 for the treatment of wounds in patients ages 6 months and older with dystrophic epidermolysis bullosa (DEB), which includes two types, the most severe form (autosomal recessive, or RDEB) and the autosomal dominant form of DEB (DDEB), which tends to be milder.
Treatment with Vyjuvek “represents an important advance in the treatment of RDEB,” wrote Adam J.N. Raymakers, PhD, and colleagues at the Program on Regulation, Therapeutics, and Law; the Department of Dermatology; and the Division of Pulmonary and Critical Care Medicine at Brigham & Women’s Hospital in Boston, Massachusetts, in their paper, published in JAMA Dermatology. But the price “will be high, potentially limiting patients’ access to it,” they added. Evidence to support it in DDEB “is less conclusive,” they wrote, noting that the pivotal phase 3 study that led to approval included one patient with DDEB.
“The wider indication granted by the FDA may lead to friction between payers on the one hand and patients and physicians on the other,” they wrote, noting a potential minimum price of $300,000 per patient a year, which was based on Krystal’s regulatory filings.
There is no cure for DEB. Vyjuvek, applied as a gel on an ongoing basis, uses a nonreplicating herpes simplex virus type 1 vector to deliver the COL7A1 gene directly to skin cells, restoring the COL7 protein fibrils that stabilize skin structure.
The authors estimated that in the United States, 894 individuals – largely children – with both forms of the disease would be eligible for Vyjuvek treatment in the first year. Based on the $300,000 price, spending on gene therapy could range from $179 million to $357 million for those 894 patients, they reported in the study.
Over the first 3 years, spending could range as high as $1 billion, the authors estimated. Even if patients with only the most severe disease (RDEB) — an estimated 442 patients — received treatment, spending could be $132 million and up to $400 million or more over the first 3 years, they wrote.
Some media outlets have reported that Vyjuvek could cost as much as $600,000, said Dr. Raymakers, a research fellow. That price “would double all of our estimates,” he told this news organization.
The study assumed that patients with RDEB would only live to age 50, which led to a lifetime cost estimate of $15 million. But that is likely a conservative estimate, he and his coauthors wrote, noting that many patients with RDEB die from squamous cell carcinoma, but that Vyjuvek could, by attenuating skin damage, also potentially prevent skin cancer.
Dr. Raymakers said he and his colleagues began their study when Vyjuvek was approved, and thus they did not have any real-world data on the price or payer responses. Their estimates also did not include differing dosing regimens, which also could change their spending figures.
Krystal Biotech recently reported that in its third quarter of 2023 – representing just 1 month of Vyjuvek availability – it received requests to begin treatment for 284 patients from 136 unique clinicians. Twenty percent of the start requests were for patients with the milder form (DDEB), and a third of all the requests were for patients 10 years of age or younger. The company also said that it had “received positive coverage determinations from all major commercial national health plans” and that it was on track to receive approval from most state Medicaid plans.
In 1 month, Krystal reported net Vyjuvek revenues of $8.6 million.
The authors suggested that one way to evaluate Vyjuvek’s value — especially for those with DDEB — would be through a cost-effectiveness study. While important, a cost-effectiveness study would not get at the impact on a payer, said Dr. Raymakers. “Something can be cost-effective but unaffordable to the system,” he said.
“When there’s one of these very expensive therapies, that’s one thing,” he said. “But when there’s more and more coming to market, you wonder how much can be tolerated,” said Dr. Raymakers.
CMS Launching Gene Therapy Program
The Biden administration recently announced that it was launching a program aimed at increasing access, curbing costs, and ensuring value of gene therapies, starting with sickle cell disease. The program will begin in early 2025. Among other aspects, the federal government will negotiate the price of the product with the manufacturer.
“The goal of the Cell and Gene Therapy Access Model is to increase access to innovative cell and gene therapies for people with Medicaid by making it easier for states to pay for these therapies,” said Liz Fowler, CMS Deputy Administrator and Director of the CMS Innovation Center, in a statement announcing the program.
Whether the new program takes a look at Vyjuvek – and when – is not clear.
But the authors of the study noted that the lifetime costs of treating a patient with Vyjuvek “exceed the costs of all other one-time gene therapies for other diseases.” And they wrote, even at the most conservative estimates, Vyjuvek “will be the most expensive gene therapy currently marketed in the US.”
The study was funded by a grant from Arnold Ventures, grants from the Kaiser Permanente Institute for Health Policy, the Commonwealth Fund, and the National Heart, Lung, and Blood Institute. Dr. Raymakers and co-authors reported no financial relationships relevant to the work.
according to the authors of a new study.
The US Food and Drug Administration (FDA) approved Vyjuvek (Krystal Biotech) in May 2023 for the treatment of wounds in patients ages 6 months and older with dystrophic epidermolysis bullosa (DEB), which includes two types, the most severe form (autosomal recessive, or RDEB) and the autosomal dominant form of DEB (DDEB), which tends to be milder.
Treatment with Vyjuvek “represents an important advance in the treatment of RDEB,” wrote Adam J.N. Raymakers, PhD, and colleagues at the Program on Regulation, Therapeutics, and Law; the Department of Dermatology; and the Division of Pulmonary and Critical Care Medicine at Brigham & Women’s Hospital in Boston, Massachusetts, in their paper, published in JAMA Dermatology. But the price “will be high, potentially limiting patients’ access to it,” they added. Evidence to support it in DDEB “is less conclusive,” they wrote, noting that the pivotal phase 3 study that led to approval included one patient with DDEB.
“The wider indication granted by the FDA may lead to friction between payers on the one hand and patients and physicians on the other,” they wrote, noting a potential minimum price of $300,000 per patient a year, which was based on Krystal’s regulatory filings.
There is no cure for DEB. Vyjuvek, applied as a gel on an ongoing basis, uses a nonreplicating herpes simplex virus type 1 vector to deliver the COL7A1 gene directly to skin cells, restoring the COL7 protein fibrils that stabilize skin structure.
The authors estimated that in the United States, 894 individuals – largely children – with both forms of the disease would be eligible for Vyjuvek treatment in the first year. Based on the $300,000 price, spending on gene therapy could range from $179 million to $357 million for those 894 patients, they reported in the study.
Over the first 3 years, spending could range as high as $1 billion, the authors estimated. Even if patients with only the most severe disease (RDEB) — an estimated 442 patients — received treatment, spending could be $132 million and up to $400 million or more over the first 3 years, they wrote.
Some media outlets have reported that Vyjuvek could cost as much as $600,000, said Dr. Raymakers, a research fellow. That price “would double all of our estimates,” he told this news organization.
The study assumed that patients with RDEB would only live to age 50, which led to a lifetime cost estimate of $15 million. But that is likely a conservative estimate, he and his coauthors wrote, noting that many patients with RDEB die from squamous cell carcinoma, but that Vyjuvek could, by attenuating skin damage, also potentially prevent skin cancer.
Dr. Raymakers said he and his colleagues began their study when Vyjuvek was approved, and thus they did not have any real-world data on the price or payer responses. Their estimates also did not include differing dosing regimens, which also could change their spending figures.
Krystal Biotech recently reported that in its third quarter of 2023 – representing just 1 month of Vyjuvek availability – it received requests to begin treatment for 284 patients from 136 unique clinicians. Twenty percent of the start requests were for patients with the milder form (DDEB), and a third of all the requests were for patients 10 years of age or younger. The company also said that it had “received positive coverage determinations from all major commercial national health plans” and that it was on track to receive approval from most state Medicaid plans.
In 1 month, Krystal reported net Vyjuvek revenues of $8.6 million.
The authors suggested that one way to evaluate Vyjuvek’s value — especially for those with DDEB — would be through a cost-effectiveness study. While important, a cost-effectiveness study would not get at the impact on a payer, said Dr. Raymakers. “Something can be cost-effective but unaffordable to the system,” he said.
“When there’s one of these very expensive therapies, that’s one thing,” he said. “But when there’s more and more coming to market, you wonder how much can be tolerated,” said Dr. Raymakers.
CMS Launching Gene Therapy Program
The Biden administration recently announced that it was launching a program aimed at increasing access, curbing costs, and ensuring value of gene therapies, starting with sickle cell disease. The program will begin in early 2025. Among other aspects, the federal government will negotiate the price of the product with the manufacturer.
“The goal of the Cell and Gene Therapy Access Model is to increase access to innovative cell and gene therapies for people with Medicaid by making it easier for states to pay for these therapies,” said Liz Fowler, CMS Deputy Administrator and Director of the CMS Innovation Center, in a statement announcing the program.
Whether the new program takes a look at Vyjuvek – and when – is not clear.
But the authors of the study noted that the lifetime costs of treating a patient with Vyjuvek “exceed the costs of all other one-time gene therapies for other diseases.” And they wrote, even at the most conservative estimates, Vyjuvek “will be the most expensive gene therapy currently marketed in the US.”
The study was funded by a grant from Arnold Ventures, grants from the Kaiser Permanente Institute for Health Policy, the Commonwealth Fund, and the National Heart, Lung, and Blood Institute. Dr. Raymakers and co-authors reported no financial relationships relevant to the work.
according to the authors of a new study.
The US Food and Drug Administration (FDA) approved Vyjuvek (Krystal Biotech) in May 2023 for the treatment of wounds in patients ages 6 months and older with dystrophic epidermolysis bullosa (DEB), which includes two types, the most severe form (autosomal recessive, or RDEB) and the autosomal dominant form of DEB (DDEB), which tends to be milder.
Treatment with Vyjuvek “represents an important advance in the treatment of RDEB,” wrote Adam J.N. Raymakers, PhD, and colleagues at the Program on Regulation, Therapeutics, and Law; the Department of Dermatology; and the Division of Pulmonary and Critical Care Medicine at Brigham & Women’s Hospital in Boston, Massachusetts, in their paper, published in JAMA Dermatology. But the price “will be high, potentially limiting patients’ access to it,” they added. Evidence to support it in DDEB “is less conclusive,” they wrote, noting that the pivotal phase 3 study that led to approval included one patient with DDEB.
“The wider indication granted by the FDA may lead to friction between payers on the one hand and patients and physicians on the other,” they wrote, noting a potential minimum price of $300,000 per patient a year, which was based on Krystal’s regulatory filings.
There is no cure for DEB. Vyjuvek, applied as a gel on an ongoing basis, uses a nonreplicating herpes simplex virus type 1 vector to deliver the COL7A1 gene directly to skin cells, restoring the COL7 protein fibrils that stabilize skin structure.
The authors estimated that in the United States, 894 individuals – largely children – with both forms of the disease would be eligible for Vyjuvek treatment in the first year. Based on the $300,000 price, spending on gene therapy could range from $179 million to $357 million for those 894 patients, they reported in the study.
Over the first 3 years, spending could range as high as $1 billion, the authors estimated. Even if patients with only the most severe disease (RDEB) — an estimated 442 patients — received treatment, spending could be $132 million and up to $400 million or more over the first 3 years, they wrote.
Some media outlets have reported that Vyjuvek could cost as much as $600,000, said Dr. Raymakers, a research fellow. That price “would double all of our estimates,” he told this news organization.
The study assumed that patients with RDEB would only live to age 50, which led to a lifetime cost estimate of $15 million. But that is likely a conservative estimate, he and his coauthors wrote, noting that many patients with RDEB die from squamous cell carcinoma, but that Vyjuvek could, by attenuating skin damage, also potentially prevent skin cancer.
Dr. Raymakers said he and his colleagues began their study when Vyjuvek was approved, and thus they did not have any real-world data on the price or payer responses. Their estimates also did not include differing dosing regimens, which also could change their spending figures.
Krystal Biotech recently reported that in its third quarter of 2023 – representing just 1 month of Vyjuvek availability – it received requests to begin treatment for 284 patients from 136 unique clinicians. Twenty percent of the start requests were for patients with the milder form (DDEB), and a third of all the requests were for patients 10 years of age or younger. The company also said that it had “received positive coverage determinations from all major commercial national health plans” and that it was on track to receive approval from most state Medicaid plans.
In 1 month, Krystal reported net Vyjuvek revenues of $8.6 million.
The authors suggested that one way to evaluate Vyjuvek’s value — especially for those with DDEB — would be through a cost-effectiveness study. While important, a cost-effectiveness study would not get at the impact on a payer, said Dr. Raymakers. “Something can be cost-effective but unaffordable to the system,” he said.
“When there’s one of these very expensive therapies, that’s one thing,” he said. “But when there’s more and more coming to market, you wonder how much can be tolerated,” said Dr. Raymakers.
CMS Launching Gene Therapy Program
The Biden administration recently announced that it was launching a program aimed at increasing access, curbing costs, and ensuring value of gene therapies, starting with sickle cell disease. The program will begin in early 2025. Among other aspects, the federal government will negotiate the price of the product with the manufacturer.
“The goal of the Cell and Gene Therapy Access Model is to increase access to innovative cell and gene therapies for people with Medicaid by making it easier for states to pay for these therapies,” said Liz Fowler, CMS Deputy Administrator and Director of the CMS Innovation Center, in a statement announcing the program.
Whether the new program takes a look at Vyjuvek – and when – is not clear.
But the authors of the study noted that the lifetime costs of treating a patient with Vyjuvek “exceed the costs of all other one-time gene therapies for other diseases.” And they wrote, even at the most conservative estimates, Vyjuvek “will be the most expensive gene therapy currently marketed in the US.”
The study was funded by a grant from Arnold Ventures, grants from the Kaiser Permanente Institute for Health Policy, the Commonwealth Fund, and the National Heart, Lung, and Blood Institute. Dr. Raymakers and co-authors reported no financial relationships relevant to the work.
FROM JAMA DERMATOLOGY
Youth Mental Health in ‘Dire Straits’
, suggests a new report that shines a light on the global mental health crisis among young people.
The burden is high in this population, with around one-fifth of all disease-related disability attributable to mental disorders. The data, drawn from the 2019 Global Burden of Disease (GBD) study, examines mental health in the 293 million people worldwide in this age group.
“This concentration of disability burden at an early age raises concern about the potential lifetime impact of these conditions,” wrote the authors, led by Christian Kieling, MD, PhD, Universidade Federal do Rio Grande do Sul, Porto Alegre, Brazil.
The study was published online in JAMA Psychiatry.
State of Emergency
Soaring rates of mental health disorders among young people, intensified by the COVID-19 pandemic, have led the American Academy of Child and Adolescent Psychiatry and the American Academy of Pediatrics to declare a state of emergency.
Using the GBD study, Dr. Kieling and colleagues estimated the global prevalence and years lived with disability associated with mental disorders and substance use disorders in people aged 5-24 years.
In 2019, individuals in this age range had at least one mental disorder and 31 million had a substance use disorder — an average prevalence of 11.6% and 1.2%, respectively.
The prevalence of mental disorders doubled from the age range of 5-9 years (6.8%) to 20-24 years (13.6%).
Among mental disorders analyzed, anxiety disorders were most common in the overall population (84 million; 3.35%) and schizophrenia the least common (2 million; 0.08%).
Notably, the researchers said, there was a steep increase in mood disorders, particularly anxiety and substance use disorders, across early to late adolescence and from late adolescence to young adulthood.
Mental disorders and substance use disorders were the leading cause of nonfatal disability in children and youths in 2019, accounting for 31 million and 4.3 million years lived with disability (YLDs), respectively. That represents roughly 20% and 3% of YLDs, respectively, from all causes.
Youth Mental Health Is Not a Monolith
“That youth mental health is in such dire straits is particularly striking given that many measures of global physical health in young people are improving,” wrote the authors of an accompanying editorial.
In their editorial, Jeremy Veenstra-VanderWeele, MD, Department of Psychiatry, Columbia University, New York, and co-authors noted that these and other age- and gender-related findings “represent a meaningful contribution to the literature.”
The granular data underscore that youth mental health is “not a monolith” but rather involves considerable variation in prevalence and morbidity across both age and gender, they wrote.
Therefore, mental health screening, promotion, and prevention efforts may benefit from an age-based approach that targets specific disorders during “high prevalence developmental intervals, with keen attention also paid to gender,” they suggested.
On the basis of the findings in this analysis, healthcare and education resource allocation may need to be adjusted for specific disorders, they added.
“One might propose a community- or school-based mental health initiative that screens for and educates parents and teachers on ADHD and anxiety disorders from kindergarten through third grade (ages 5-9 years, when prevalence and resulting disability grow markedly),” Dr. Veenstra-VanderWeele and colleagues wrote. “Later initiatives could then focus on mood and substance use disorders during high school and college (ages 15-19 years and 20-24 years).”
The study was partially funded by a research grant from the Cundill Centre for Child and Youth Depression. Dr. Kieling is the founder of Wida. Dr. Veenstra-VanderWeele reported receiving grants from the National Institutes of Health and Simon’s Foundation and research support/advisory board/editorial fees from Autism Speaks, Agency for Healthcare Research and Quality, Health Resources and Services Administration Maternal and Child Health Bureau, American Academy of Child and Adolescent Psychiatry, Forest, Janssen, Yamo, MapLight, Acadia, Roche, Novartis, Seaside Therapeutics, Springer, SynapDx, and Wiley.
A version of this article appeared on Medscape.com.
, suggests a new report that shines a light on the global mental health crisis among young people.
The burden is high in this population, with around one-fifth of all disease-related disability attributable to mental disorders. The data, drawn from the 2019 Global Burden of Disease (GBD) study, examines mental health in the 293 million people worldwide in this age group.
“This concentration of disability burden at an early age raises concern about the potential lifetime impact of these conditions,” wrote the authors, led by Christian Kieling, MD, PhD, Universidade Federal do Rio Grande do Sul, Porto Alegre, Brazil.
The study was published online in JAMA Psychiatry.
State of Emergency
Soaring rates of mental health disorders among young people, intensified by the COVID-19 pandemic, have led the American Academy of Child and Adolescent Psychiatry and the American Academy of Pediatrics to declare a state of emergency.
Using the GBD study, Dr. Kieling and colleagues estimated the global prevalence and years lived with disability associated with mental disorders and substance use disorders in people aged 5-24 years.
In 2019, individuals in this age range had at least one mental disorder and 31 million had a substance use disorder — an average prevalence of 11.6% and 1.2%, respectively.
The prevalence of mental disorders doubled from the age range of 5-9 years (6.8%) to 20-24 years (13.6%).
Among mental disorders analyzed, anxiety disorders were most common in the overall population (84 million; 3.35%) and schizophrenia the least common (2 million; 0.08%).
Notably, the researchers said, there was a steep increase in mood disorders, particularly anxiety and substance use disorders, across early to late adolescence and from late adolescence to young adulthood.
Mental disorders and substance use disorders were the leading cause of nonfatal disability in children and youths in 2019, accounting for 31 million and 4.3 million years lived with disability (YLDs), respectively. That represents roughly 20% and 3% of YLDs, respectively, from all causes.
Youth Mental Health Is Not a Monolith
“That youth mental health is in such dire straits is particularly striking given that many measures of global physical health in young people are improving,” wrote the authors of an accompanying editorial.
In their editorial, Jeremy Veenstra-VanderWeele, MD, Department of Psychiatry, Columbia University, New York, and co-authors noted that these and other age- and gender-related findings “represent a meaningful contribution to the literature.”
The granular data underscore that youth mental health is “not a monolith” but rather involves considerable variation in prevalence and morbidity across both age and gender, they wrote.
Therefore, mental health screening, promotion, and prevention efforts may benefit from an age-based approach that targets specific disorders during “high prevalence developmental intervals, with keen attention also paid to gender,” they suggested.
On the basis of the findings in this analysis, healthcare and education resource allocation may need to be adjusted for specific disorders, they added.
“One might propose a community- or school-based mental health initiative that screens for and educates parents and teachers on ADHD and anxiety disorders from kindergarten through third grade (ages 5-9 years, when prevalence and resulting disability grow markedly),” Dr. Veenstra-VanderWeele and colleagues wrote. “Later initiatives could then focus on mood and substance use disorders during high school and college (ages 15-19 years and 20-24 years).”
The study was partially funded by a research grant from the Cundill Centre for Child and Youth Depression. Dr. Kieling is the founder of Wida. Dr. Veenstra-VanderWeele reported receiving grants from the National Institutes of Health and Simon’s Foundation and research support/advisory board/editorial fees from Autism Speaks, Agency for Healthcare Research and Quality, Health Resources and Services Administration Maternal and Child Health Bureau, American Academy of Child and Adolescent Psychiatry, Forest, Janssen, Yamo, MapLight, Acadia, Roche, Novartis, Seaside Therapeutics, Springer, SynapDx, and Wiley.
A version of this article appeared on Medscape.com.
, suggests a new report that shines a light on the global mental health crisis among young people.
The burden is high in this population, with around one-fifth of all disease-related disability attributable to mental disorders. The data, drawn from the 2019 Global Burden of Disease (GBD) study, examines mental health in the 293 million people worldwide in this age group.
“This concentration of disability burden at an early age raises concern about the potential lifetime impact of these conditions,” wrote the authors, led by Christian Kieling, MD, PhD, Universidade Federal do Rio Grande do Sul, Porto Alegre, Brazil.
The study was published online in JAMA Psychiatry.
State of Emergency
Soaring rates of mental health disorders among young people, intensified by the COVID-19 pandemic, have led the American Academy of Child and Adolescent Psychiatry and the American Academy of Pediatrics to declare a state of emergency.
Using the GBD study, Dr. Kieling and colleagues estimated the global prevalence and years lived with disability associated with mental disorders and substance use disorders in people aged 5-24 years.
In 2019, individuals in this age range had at least one mental disorder and 31 million had a substance use disorder — an average prevalence of 11.6% and 1.2%, respectively.
The prevalence of mental disorders doubled from the age range of 5-9 years (6.8%) to 20-24 years (13.6%).
Among mental disorders analyzed, anxiety disorders were most common in the overall population (84 million; 3.35%) and schizophrenia the least common (2 million; 0.08%).
Notably, the researchers said, there was a steep increase in mood disorders, particularly anxiety and substance use disorders, across early to late adolescence and from late adolescence to young adulthood.
Mental disorders and substance use disorders were the leading cause of nonfatal disability in children and youths in 2019, accounting for 31 million and 4.3 million years lived with disability (YLDs), respectively. That represents roughly 20% and 3% of YLDs, respectively, from all causes.
Youth Mental Health Is Not a Monolith
“That youth mental health is in such dire straits is particularly striking given that many measures of global physical health in young people are improving,” wrote the authors of an accompanying editorial.
In their editorial, Jeremy Veenstra-VanderWeele, MD, Department of Psychiatry, Columbia University, New York, and co-authors noted that these and other age- and gender-related findings “represent a meaningful contribution to the literature.”
The granular data underscore that youth mental health is “not a monolith” but rather involves considerable variation in prevalence and morbidity across both age and gender, they wrote.
Therefore, mental health screening, promotion, and prevention efforts may benefit from an age-based approach that targets specific disorders during “high prevalence developmental intervals, with keen attention also paid to gender,” they suggested.
On the basis of the findings in this analysis, healthcare and education resource allocation may need to be adjusted for specific disorders, they added.
“One might propose a community- or school-based mental health initiative that screens for and educates parents and teachers on ADHD and anxiety disorders from kindergarten through third grade (ages 5-9 years, when prevalence and resulting disability grow markedly),” Dr. Veenstra-VanderWeele and colleagues wrote. “Later initiatives could then focus on mood and substance use disorders during high school and college (ages 15-19 years and 20-24 years).”
The study was partially funded by a research grant from the Cundill Centre for Child and Youth Depression. Dr. Kieling is the founder of Wida. Dr. Veenstra-VanderWeele reported receiving grants from the National Institutes of Health and Simon’s Foundation and research support/advisory board/editorial fees from Autism Speaks, Agency for Healthcare Research and Quality, Health Resources and Services Administration Maternal and Child Health Bureau, American Academy of Child and Adolescent Psychiatry, Forest, Janssen, Yamo, MapLight, Acadia, Roche, Novartis, Seaside Therapeutics, Springer, SynapDx, and Wiley.
A version of this article appeared on Medscape.com.
Dupilumab Improves AD Affecting the Hands, Feet
TOPLINE:
compared with placebo.
METHODOLOGY:
- The multinational phase 3 LIBERTY-AD-HAFT trial of adults and adolescents with moderate to severe chronic atopic dermatitis (AD) of the hands, feet, or both included 67 participants at 48 sites randomized to dupilumab monotherapy and 66 to placebo.
- The primary endpoint was the proportion of patients scoring 0 or 1 on Hand and Foot Investigator’s Global Assessment (HF-IGA) at week 16.
- Secondary endpoints were severity and extent of signs, symptom intensity (itch and pain), sleep, and quality of life.
TAKEAWAY:
- At week 16, 27 patients receiving dupilumab vs 11 receiving placebo achieved an HF-IGA score of 0 or 1 (40.3% vs 16.7%; P = .003).
- At week 16, 35 participants receiving dupilumab vs nine receiving placebo improved at least four points in the weekly average of daily HF-Peak Pruritus Numeric Rating Scale (52.2% vs 13.6%; P < .0001).
- At week 16, Quality of Life Hand Eczema Questionnaire results improved in the dupilumab group compared with controls (P < .0001), and weekly average of daily Sleep Numeric Rating Scale results improved in the dupilumab group compared with controls (P < .05).
- The safety profile was similar to the known profile in adults and adolescents with moderate to severe AD.
IN PRACTICE:
The results of the study “support dupilumab” as an “efficacious systemic therapy for moderate to severe H/F AD,” the authors wrote.
SOURCE:
The study, led by Eric L. Simpson, MD, MCR, professor of dermatology at the Oregon Health & Science University in Portland, was published on January 29, 2024, in the Journal of the American Academy of Dermatology.
LIMITATIONS:
The short duration of the study and the large proportion of patients with positive patch tests (31 of 133) suggested that some participants may have had concurrent AD and allergic contact dermatitis, so the effect of dupilumab on those patients needs further evaluation.
DISCLOSURES:
The study was sponsored by Sanofi and Regeneron. All but one author had financial relationships with Sanofi, Regeneron, or both. Several authors were employees of, and may hold stocks or stock options in, Sanofi or Regeneron.
TOPLINE:
compared with placebo.
METHODOLOGY:
- The multinational phase 3 LIBERTY-AD-HAFT trial of adults and adolescents with moderate to severe chronic atopic dermatitis (AD) of the hands, feet, or both included 67 participants at 48 sites randomized to dupilumab monotherapy and 66 to placebo.
- The primary endpoint was the proportion of patients scoring 0 or 1 on Hand and Foot Investigator’s Global Assessment (HF-IGA) at week 16.
- Secondary endpoints were severity and extent of signs, symptom intensity (itch and pain), sleep, and quality of life.
TAKEAWAY:
- At week 16, 27 patients receiving dupilumab vs 11 receiving placebo achieved an HF-IGA score of 0 or 1 (40.3% vs 16.7%; P = .003).
- At week 16, 35 participants receiving dupilumab vs nine receiving placebo improved at least four points in the weekly average of daily HF-Peak Pruritus Numeric Rating Scale (52.2% vs 13.6%; P < .0001).
- At week 16, Quality of Life Hand Eczema Questionnaire results improved in the dupilumab group compared with controls (P < .0001), and weekly average of daily Sleep Numeric Rating Scale results improved in the dupilumab group compared with controls (P < .05).
- The safety profile was similar to the known profile in adults and adolescents with moderate to severe AD.
IN PRACTICE:
The results of the study “support dupilumab” as an “efficacious systemic therapy for moderate to severe H/F AD,” the authors wrote.
SOURCE:
The study, led by Eric L. Simpson, MD, MCR, professor of dermatology at the Oregon Health & Science University in Portland, was published on January 29, 2024, in the Journal of the American Academy of Dermatology.
LIMITATIONS:
The short duration of the study and the large proportion of patients with positive patch tests (31 of 133) suggested that some participants may have had concurrent AD and allergic contact dermatitis, so the effect of dupilumab on those patients needs further evaluation.
DISCLOSURES:
The study was sponsored by Sanofi and Regeneron. All but one author had financial relationships with Sanofi, Regeneron, or both. Several authors were employees of, and may hold stocks or stock options in, Sanofi or Regeneron.
TOPLINE:
compared with placebo.
METHODOLOGY:
- The multinational phase 3 LIBERTY-AD-HAFT trial of adults and adolescents with moderate to severe chronic atopic dermatitis (AD) of the hands, feet, or both included 67 participants at 48 sites randomized to dupilumab monotherapy and 66 to placebo.
- The primary endpoint was the proportion of patients scoring 0 or 1 on Hand and Foot Investigator’s Global Assessment (HF-IGA) at week 16.
- Secondary endpoints were severity and extent of signs, symptom intensity (itch and pain), sleep, and quality of life.
TAKEAWAY:
- At week 16, 27 patients receiving dupilumab vs 11 receiving placebo achieved an HF-IGA score of 0 or 1 (40.3% vs 16.7%; P = .003).
- At week 16, 35 participants receiving dupilumab vs nine receiving placebo improved at least four points in the weekly average of daily HF-Peak Pruritus Numeric Rating Scale (52.2% vs 13.6%; P < .0001).
- At week 16, Quality of Life Hand Eczema Questionnaire results improved in the dupilumab group compared with controls (P < .0001), and weekly average of daily Sleep Numeric Rating Scale results improved in the dupilumab group compared with controls (P < .05).
- The safety profile was similar to the known profile in adults and adolescents with moderate to severe AD.
IN PRACTICE:
The results of the study “support dupilumab” as an “efficacious systemic therapy for moderate to severe H/F AD,” the authors wrote.
SOURCE:
The study, led by Eric L. Simpson, MD, MCR, professor of dermatology at the Oregon Health & Science University in Portland, was published on January 29, 2024, in the Journal of the American Academy of Dermatology.
LIMITATIONS:
The short duration of the study and the large proportion of patients with positive patch tests (31 of 133) suggested that some participants may have had concurrent AD and allergic contact dermatitis, so the effect of dupilumab on those patients needs further evaluation.
DISCLOSURES:
The study was sponsored by Sanofi and Regeneron. All but one author had financial relationships with Sanofi, Regeneron, or both. Several authors were employees of, and may hold stocks or stock options in, Sanofi or Regeneron.
Survey: Dermatology Residents Shortchanged on Sensitive Skin Education
Although sensitive skin affects an estimated 40%-70% of the population, knowledge of the pathophysiology of sensitive skin is incomplete, and consensus is lacking as to the best diagnosis and treatment strategies, and the inclusion of sensitive skin education in dermatology curricula has not been examined, according to Erika T. McCormick, BS, and Adam Friedman, MD, of George Washington University, Washington, DC.
For the study, published in the Journal of Drugs in Dermatology, they developed a 26-question survey for dermatology residents that asked about sensitive skin in dermatology residency training. Participants came from the Orlando Dermatology, Aesthetic, and Surgical Conference email list.
Survey respondents included 214 residents at various levels of training at programs across the United States; 67.1% were female, 92.1% were aged 25-34 years, and 85.5% were in academic or university programs.
Overall, 99% of respondents believed that sensitive skin issues should be part of their residency training to some extent, and 84% reported experiences with patients for whom the chief presenting complaint was sensitive skin.
However, fewer than half (48%) of the residents reported specific resident education in sensitive skin, while 51% reported nonspecific education about sensitive skin education in the context of other skin diseases, and 1% reported no education about sensitive skin.
Less than one-quarter of the respondents who received any sensitive skin education reported feeling comfortable in their ability to diagnose, evaluate, and manage sensitive skin, while those with sensitive skin–specific education were significantly more likely to describe themselves as “very knowledgeable.”
As for treatment approaches, residents with specific sensitive skin education were more likely than were those without sensitive skin–specific training to ask patients about allergies and past reactions to skin products, and to counsel them about environmental triggers.
Notably, 96% of the respondents were not familiar with the Sensitive Skin (SS) Scale–10, a validated measure of sensitive skin severity.
The most common challenges in care of patients with sensitive skin were assessing improvement over time, reported by 25% of respondents, recommending products (23%), and prescribing/medical management (22%). The topics residents expressed most interest in learning about were product recommendations (78%), patient counseling (77%), reviewing research on sensitive skin (70%), diagnosing sensitive skin (67%), using the SS-10 (48%), and clinical research updates (40%).
The findings were limited by several factors including the reliance on self-reports, the researchers noted. However, the results highlight the lack of consensus in treatment of sensitive skin and the need to address this knowledge gap at the residency level, they said.
Improving Tools for Practice
“Many practice patterns and approaches are forged in the fires of training,” corresponding author Dr. Friedman, professor and chair of dermatology and residency program director at George Washington University, said in an interview. “Identifying gaps, especially for heavily prevalent issues, questions, and concerns such as sensitive skin that residents will encounter in practice is important to ensure an educated workforce,” he said.
Education on sensitive skin is lacking because, until recently, research and clinical guidance have been lacking, Dr. Friedman said. The root of the problem is that sensitive skin is mainly considered a symptom, rather than an independent condition, he explained. “Depending on the study, the prevalence of sensitive skin has been reported as high as 70%, with roughly 40% of these patients having no primary skin condition,” he said. This means sensitive skin can be both a symptom and a condition, which causes confusion for clinicians and patients, he added.
“Therefore, in order to overcome this gap, the condition itself at a minimum needs a standard definition and a way to diagnosis, which we fortunately have in the validated research tool known as the SS-10,” said Dr. Friedman.
Almost all residents surveyed in the current study had never heard of the SS-10, but more than half found it to be useful after learning of it through the study survey, he noted.
Looking ahead, greater elucidation of the pathophysiology of sensitive skin is needed to effectively pursue studies of products and treatments for these patients, but the SS-10 can be used to define and monitor the condition to evaluate improvement, he added.
The study was funded by an independent fellowship grant from Galderma. Ms. McCormick is supported by an unrestricted fellowship grant funded by Galderma. Dr. Friedman has served as a consultant for Galderma.
Although sensitive skin affects an estimated 40%-70% of the population, knowledge of the pathophysiology of sensitive skin is incomplete, and consensus is lacking as to the best diagnosis and treatment strategies, and the inclusion of sensitive skin education in dermatology curricula has not been examined, according to Erika T. McCormick, BS, and Adam Friedman, MD, of George Washington University, Washington, DC.
For the study, published in the Journal of Drugs in Dermatology, they developed a 26-question survey for dermatology residents that asked about sensitive skin in dermatology residency training. Participants came from the Orlando Dermatology, Aesthetic, and Surgical Conference email list.
Survey respondents included 214 residents at various levels of training at programs across the United States; 67.1% were female, 92.1% were aged 25-34 years, and 85.5% were in academic or university programs.
Overall, 99% of respondents believed that sensitive skin issues should be part of their residency training to some extent, and 84% reported experiences with patients for whom the chief presenting complaint was sensitive skin.
However, fewer than half (48%) of the residents reported specific resident education in sensitive skin, while 51% reported nonspecific education about sensitive skin education in the context of other skin diseases, and 1% reported no education about sensitive skin.
Less than one-quarter of the respondents who received any sensitive skin education reported feeling comfortable in their ability to diagnose, evaluate, and manage sensitive skin, while those with sensitive skin–specific education were significantly more likely to describe themselves as “very knowledgeable.”
As for treatment approaches, residents with specific sensitive skin education were more likely than were those without sensitive skin–specific training to ask patients about allergies and past reactions to skin products, and to counsel them about environmental triggers.
Notably, 96% of the respondents were not familiar with the Sensitive Skin (SS) Scale–10, a validated measure of sensitive skin severity.
The most common challenges in care of patients with sensitive skin were assessing improvement over time, reported by 25% of respondents, recommending products (23%), and prescribing/medical management (22%). The topics residents expressed most interest in learning about were product recommendations (78%), patient counseling (77%), reviewing research on sensitive skin (70%), diagnosing sensitive skin (67%), using the SS-10 (48%), and clinical research updates (40%).
The findings were limited by several factors including the reliance on self-reports, the researchers noted. However, the results highlight the lack of consensus in treatment of sensitive skin and the need to address this knowledge gap at the residency level, they said.
Improving Tools for Practice
“Many practice patterns and approaches are forged in the fires of training,” corresponding author Dr. Friedman, professor and chair of dermatology and residency program director at George Washington University, said in an interview. “Identifying gaps, especially for heavily prevalent issues, questions, and concerns such as sensitive skin that residents will encounter in practice is important to ensure an educated workforce,” he said.
Education on sensitive skin is lacking because, until recently, research and clinical guidance have been lacking, Dr. Friedman said. The root of the problem is that sensitive skin is mainly considered a symptom, rather than an independent condition, he explained. “Depending on the study, the prevalence of sensitive skin has been reported as high as 70%, with roughly 40% of these patients having no primary skin condition,” he said. This means sensitive skin can be both a symptom and a condition, which causes confusion for clinicians and patients, he added.
“Therefore, in order to overcome this gap, the condition itself at a minimum needs a standard definition and a way to diagnosis, which we fortunately have in the validated research tool known as the SS-10,” said Dr. Friedman.
Almost all residents surveyed in the current study had never heard of the SS-10, but more than half found it to be useful after learning of it through the study survey, he noted.
Looking ahead, greater elucidation of the pathophysiology of sensitive skin is needed to effectively pursue studies of products and treatments for these patients, but the SS-10 can be used to define and monitor the condition to evaluate improvement, he added.
The study was funded by an independent fellowship grant from Galderma. Ms. McCormick is supported by an unrestricted fellowship grant funded by Galderma. Dr. Friedman has served as a consultant for Galderma.
Although sensitive skin affects an estimated 40%-70% of the population, knowledge of the pathophysiology of sensitive skin is incomplete, and consensus is lacking as to the best diagnosis and treatment strategies, and the inclusion of sensitive skin education in dermatology curricula has not been examined, according to Erika T. McCormick, BS, and Adam Friedman, MD, of George Washington University, Washington, DC.
For the study, published in the Journal of Drugs in Dermatology, they developed a 26-question survey for dermatology residents that asked about sensitive skin in dermatology residency training. Participants came from the Orlando Dermatology, Aesthetic, and Surgical Conference email list.
Survey respondents included 214 residents at various levels of training at programs across the United States; 67.1% were female, 92.1% were aged 25-34 years, and 85.5% were in academic or university programs.
Overall, 99% of respondents believed that sensitive skin issues should be part of their residency training to some extent, and 84% reported experiences with patients for whom the chief presenting complaint was sensitive skin.
However, fewer than half (48%) of the residents reported specific resident education in sensitive skin, while 51% reported nonspecific education about sensitive skin education in the context of other skin diseases, and 1% reported no education about sensitive skin.
Less than one-quarter of the respondents who received any sensitive skin education reported feeling comfortable in their ability to diagnose, evaluate, and manage sensitive skin, while those with sensitive skin–specific education were significantly more likely to describe themselves as “very knowledgeable.”
As for treatment approaches, residents with specific sensitive skin education were more likely than were those without sensitive skin–specific training to ask patients about allergies and past reactions to skin products, and to counsel them about environmental triggers.
Notably, 96% of the respondents were not familiar with the Sensitive Skin (SS) Scale–10, a validated measure of sensitive skin severity.
The most common challenges in care of patients with sensitive skin were assessing improvement over time, reported by 25% of respondents, recommending products (23%), and prescribing/medical management (22%). The topics residents expressed most interest in learning about were product recommendations (78%), patient counseling (77%), reviewing research on sensitive skin (70%), diagnosing sensitive skin (67%), using the SS-10 (48%), and clinical research updates (40%).
The findings were limited by several factors including the reliance on self-reports, the researchers noted. However, the results highlight the lack of consensus in treatment of sensitive skin and the need to address this knowledge gap at the residency level, they said.
Improving Tools for Practice
“Many practice patterns and approaches are forged in the fires of training,” corresponding author Dr. Friedman, professor and chair of dermatology and residency program director at George Washington University, said in an interview. “Identifying gaps, especially for heavily prevalent issues, questions, and concerns such as sensitive skin that residents will encounter in practice is important to ensure an educated workforce,” he said.
Education on sensitive skin is lacking because, until recently, research and clinical guidance have been lacking, Dr. Friedman said. The root of the problem is that sensitive skin is mainly considered a symptom, rather than an independent condition, he explained. “Depending on the study, the prevalence of sensitive skin has been reported as high as 70%, with roughly 40% of these patients having no primary skin condition,” he said. This means sensitive skin can be both a symptom and a condition, which causes confusion for clinicians and patients, he added.
“Therefore, in order to overcome this gap, the condition itself at a minimum needs a standard definition and a way to diagnosis, which we fortunately have in the validated research tool known as the SS-10,” said Dr. Friedman.
Almost all residents surveyed in the current study had never heard of the SS-10, but more than half found it to be useful after learning of it through the study survey, he noted.
Looking ahead, greater elucidation of the pathophysiology of sensitive skin is needed to effectively pursue studies of products and treatments for these patients, but the SS-10 can be used to define and monitor the condition to evaluate improvement, he added.
The study was funded by an independent fellowship grant from Galderma. Ms. McCormick is supported by an unrestricted fellowship grant funded by Galderma. Dr. Friedman has served as a consultant for Galderma.
FROM THE JOURNAL OF DRUGS IN DERMATOLOGY
Freedom of Speech and Gender-Affirming Care
Blue Hill is a small idyllic town a little less than two and a half hours Down East the coast from where I am sitting here in Harpswell. Thanks to gentrification it tends to lean left politically, but like the rest of Maine most folks in the surrounding communities often don’t know or care much about their neighbor’s party affiliation. Its library, founded in 1796, is well funded and a source of civic pride.
One day a couple of years ago, the library director received a donated book from a patron. Although he personally didn’t agree with the book’s message, he felt it deserved a space in their collection dealing with the subject. What happened in the wake of this donation is an ugly tale. Some community members objected to the book and asked that it be removed from the shelves, or at least kept under the desk and loaned out only on request.
The objectors, many of whom knew the director, were confrontational. The collections committee unanimously supported his decision. Some committee members also received similar responses from community members. Remember, this is a small town.
A request for support sent to the American Library Association was basically ignored. Over the next 2 years things have quieted, but fractured friendships and relationships in this quiet coastal Maine town have not been repaired. However, as the librarian has observed, “intellectual freedom or the freedom of speech isn’t there just to protect the ideas that we like.”
While the title of the book may feel inflammatory to some, every publisher hopes to grab the market’s attention with a hot title. The cause of this sad situation in Blue Hill was not a white supremacist’s polemic offering specific ways to create genocide. This was a book suggesting that gender dysphoria presenting in adolescence may have multiple causes and raises concerns about the wisdom of the pace of some gender-affirming care.
Clearly the topic of gender dysphoria in adolescence has become a third rail that must be approached with caution or completely avoided. A recent opinion piece in the New York Times provides even more concerning examples of this peril. Again, the eye-catching title of the article — As Kids, They Thought They Were Trans. They No Longer Do — draws in the audience eager to read about some unfortunate individuals who have regretted their decision to transition and are now detransitioning.
If you are interested in hearing anecdotal evidence and opinions supporting the notion that there is such a thing as rapid-onset gender dysphoria, I suggest you read the entire piece. However, the article’s most troubling message for me comes when I read about the professionals who were former gender-related care providers who left the field because of “pushback, the accusations of being transphobic, from being pro-assessment and wanting a more thorough process.”
One therapist trained in gender-affirming care who began to have doubts about the model and spoke out in favor of a more measured approach was investigated by her licensing board after transgender advocates threatened to report her. Ultimately, her case was dismissed, but she continues to fear for her safety.
Gender-related healthcare is another sad example of how in this country it is the noise coming from the advocates on the extremes of the issue that is drowning out the “vast ideological middle” that is seeking civil and rational discussions.
In this situation there are those who want to make it illegal for the healthcare providers to help patients who might benefit from transitioning. On the other end of the spectrum are those advocates who are unwilling to acknowledge that there may be some adolescents with what has been called by some “rapid-onset gender dysphoria.”
The landscape on which this tragedy is being played out is changing so quickly that there will be no correct answers in the short term. There just isn’t enough data. However, there is enough anecdotal evidence from professionals who were and still are practicing gender-related care to raise a concern that something is happening in the adolescent population that suggests some individuals with gender dysphoria should be managed in a different way than the currently accepted gender-affirming model. The size of this subgroup is up for debate and we may never learn it because of reporting bias and privacy concerns.
The American Academy of Pediatrics has recently authorized a systematic review of gender-affirming care. I hope that, like the librarian in Blue Hill, it will have the courage to include all the evidence available even though, as we have seen here in Maine, some of it may spark a firestorm of vehement responses.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected].
Blue Hill is a small idyllic town a little less than two and a half hours Down East the coast from where I am sitting here in Harpswell. Thanks to gentrification it tends to lean left politically, but like the rest of Maine most folks in the surrounding communities often don’t know or care much about their neighbor’s party affiliation. Its library, founded in 1796, is well funded and a source of civic pride.
One day a couple of years ago, the library director received a donated book from a patron. Although he personally didn’t agree with the book’s message, he felt it deserved a space in their collection dealing with the subject. What happened in the wake of this donation is an ugly tale. Some community members objected to the book and asked that it be removed from the shelves, or at least kept under the desk and loaned out only on request.
The objectors, many of whom knew the director, were confrontational. The collections committee unanimously supported his decision. Some committee members also received similar responses from community members. Remember, this is a small town.
A request for support sent to the American Library Association was basically ignored. Over the next 2 years things have quieted, but fractured friendships and relationships in this quiet coastal Maine town have not been repaired. However, as the librarian has observed, “intellectual freedom or the freedom of speech isn’t there just to protect the ideas that we like.”
While the title of the book may feel inflammatory to some, every publisher hopes to grab the market’s attention with a hot title. The cause of this sad situation in Blue Hill was not a white supremacist’s polemic offering specific ways to create genocide. This was a book suggesting that gender dysphoria presenting in adolescence may have multiple causes and raises concerns about the wisdom of the pace of some gender-affirming care.
Clearly the topic of gender dysphoria in adolescence has become a third rail that must be approached with caution or completely avoided. A recent opinion piece in the New York Times provides even more concerning examples of this peril. Again, the eye-catching title of the article — As Kids, They Thought They Were Trans. They No Longer Do — draws in the audience eager to read about some unfortunate individuals who have regretted their decision to transition and are now detransitioning.
If you are interested in hearing anecdotal evidence and opinions supporting the notion that there is such a thing as rapid-onset gender dysphoria, I suggest you read the entire piece. However, the article’s most troubling message for me comes when I read about the professionals who were former gender-related care providers who left the field because of “pushback, the accusations of being transphobic, from being pro-assessment and wanting a more thorough process.”
One therapist trained in gender-affirming care who began to have doubts about the model and spoke out in favor of a more measured approach was investigated by her licensing board after transgender advocates threatened to report her. Ultimately, her case was dismissed, but she continues to fear for her safety.
Gender-related healthcare is another sad example of how in this country it is the noise coming from the advocates on the extremes of the issue that is drowning out the “vast ideological middle” that is seeking civil and rational discussions.
In this situation there are those who want to make it illegal for the healthcare providers to help patients who might benefit from transitioning. On the other end of the spectrum are those advocates who are unwilling to acknowledge that there may be some adolescents with what has been called by some “rapid-onset gender dysphoria.”
The landscape on which this tragedy is being played out is changing so quickly that there will be no correct answers in the short term. There just isn’t enough data. However, there is enough anecdotal evidence from professionals who were and still are practicing gender-related care to raise a concern that something is happening in the adolescent population that suggests some individuals with gender dysphoria should be managed in a different way than the currently accepted gender-affirming model. The size of this subgroup is up for debate and we may never learn it because of reporting bias and privacy concerns.
The American Academy of Pediatrics has recently authorized a systematic review of gender-affirming care. I hope that, like the librarian in Blue Hill, it will have the courage to include all the evidence available even though, as we have seen here in Maine, some of it may spark a firestorm of vehement responses.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected].
Blue Hill is a small idyllic town a little less than two and a half hours Down East the coast from where I am sitting here in Harpswell. Thanks to gentrification it tends to lean left politically, but like the rest of Maine most folks in the surrounding communities often don’t know or care much about their neighbor’s party affiliation. Its library, founded in 1796, is well funded and a source of civic pride.
One day a couple of years ago, the library director received a donated book from a patron. Although he personally didn’t agree with the book’s message, he felt it deserved a space in their collection dealing with the subject. What happened in the wake of this donation is an ugly tale. Some community members objected to the book and asked that it be removed from the shelves, or at least kept under the desk and loaned out only on request.
The objectors, many of whom knew the director, were confrontational. The collections committee unanimously supported his decision. Some committee members also received similar responses from community members. Remember, this is a small town.
A request for support sent to the American Library Association was basically ignored. Over the next 2 years things have quieted, but fractured friendships and relationships in this quiet coastal Maine town have not been repaired. However, as the librarian has observed, “intellectual freedom or the freedom of speech isn’t there just to protect the ideas that we like.”
While the title of the book may feel inflammatory to some, every publisher hopes to grab the market’s attention with a hot title. The cause of this sad situation in Blue Hill was not a white supremacist’s polemic offering specific ways to create genocide. This was a book suggesting that gender dysphoria presenting in adolescence may have multiple causes and raises concerns about the wisdom of the pace of some gender-affirming care.
Clearly the topic of gender dysphoria in adolescence has become a third rail that must be approached with caution or completely avoided. A recent opinion piece in the New York Times provides even more concerning examples of this peril. Again, the eye-catching title of the article — As Kids, They Thought They Were Trans. They No Longer Do — draws in the audience eager to read about some unfortunate individuals who have regretted their decision to transition and are now detransitioning.
If you are interested in hearing anecdotal evidence and opinions supporting the notion that there is such a thing as rapid-onset gender dysphoria, I suggest you read the entire piece. However, the article’s most troubling message for me comes when I read about the professionals who were former gender-related care providers who left the field because of “pushback, the accusations of being transphobic, from being pro-assessment and wanting a more thorough process.”
One therapist trained in gender-affirming care who began to have doubts about the model and spoke out in favor of a more measured approach was investigated by her licensing board after transgender advocates threatened to report her. Ultimately, her case was dismissed, but she continues to fear for her safety.
Gender-related healthcare is another sad example of how in this country it is the noise coming from the advocates on the extremes of the issue that is drowning out the “vast ideological middle” that is seeking civil and rational discussions.
In this situation there are those who want to make it illegal for the healthcare providers to help patients who might benefit from transitioning. On the other end of the spectrum are those advocates who are unwilling to acknowledge that there may be some adolescents with what has been called by some “rapid-onset gender dysphoria.”
The landscape on which this tragedy is being played out is changing so quickly that there will be no correct answers in the short term. There just isn’t enough data. However, there is enough anecdotal evidence from professionals who were and still are practicing gender-related care to raise a concern that something is happening in the adolescent population that suggests some individuals with gender dysphoria should be managed in a different way than the currently accepted gender-affirming model. The size of this subgroup is up for debate and we may never learn it because of reporting bias and privacy concerns.
The American Academy of Pediatrics has recently authorized a systematic review of gender-affirming care. I hope that, like the librarian in Blue Hill, it will have the courage to include all the evidence available even though, as we have seen here in Maine, some of it may spark a firestorm of vehement responses.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected].


